our trade secrets and unpatented know-how. We require our employees, consultants and significant scientific collaborators and sponsored researchers to execute confidentiality agreements upon the commencement of an employment or consulting relationship with us. These agreements may, however, fail to provide meaningful protection or adequate remedies for us in the event of unauthorized use, transfer or disclosure of such information or technology.
If we are unable to obtain necessary licenses to third-party patents and other rights, we may not be able to commercially develop our expected products.
A number of pharmaceutical, biotechnology and other companies, universities and research institutions have filed patent applications or have received patents relating to cell therapy, stem and progenitor cells and other technologies potentially relevant to, or necessary for, our expected products. We cannot predict which, if any, of these applications will issue as patents or how many of these issued patents will be found valid and enforceable. There may also be existing issued patents which we are currently unaware of which would be infringed by the commercialization of one or more of our product candidates. If so, we may be prevented from commercializing these products unless the third party is willing to grant a license to us. We may be unable to obtain licenses to the relevant patents at a reasonable cost, if at all, and may also be unable to develop or obtain alternative non-infringing technology. If we are unable to obtain such licenses or develop non-infringing technology at a reasonable cost, our business could be significantly harmed. Also, any infringement lawsuits commenced against us may result in significant costs, divert our management’s attention and result in an award against us for substantial damages, or potentially prevent us from continuing certain operations.
We are aware of intellectual property rights held by third parties that relate to products or technologies we are developing. For example, some aspects of our cell-based therapeutic product candidates involve the use of growth factors, antibodies and other reagents that may, in certain cases, be the subject of third party rights. Before we commercialize any product using these growth factors, antibodies or reagents, we may need to obtain license rights from third parties or use alternative growth factors, antibodies and reagents that are not then the subject of third party patent rights. We currently believe that the commercialization of our products as currently planned will not infringe these third party rights, or, alternatively, that we will be able to obtain necessary licenses or otherwise use alternative non-infringing technology. However, third parties may nonetheless bring suit against us claiming infringement. If we are unable to prove that our technology does not infringe their patents, or if we are unable to obtain necessary licenses or otherwise use alternative non-infringing technology, we may not be able to commercialize any products.
We have obtained rights from companies, universities and research institutions to technologies, processes and compounds that we believe may be important to the development of our products. These licensors, however, may cancel our licenses or convert them to non-exclusive licenses if we fail to use the relevant technology or otherwise breach these agreements. Loss of these licenses could expose us to the risk that our technology infringes the rights of third parties. We can give no assurance that any of these licenses will provide effective protection against our competitors.
21
We compete with companies that have significant advantages over us.
The market for therapeutic products to treat diseases of, or injuries to, the central nervous system (CNS) is large and competition is intense. The majority of the products currently on the market or in development are small molecule pharmaceutical compounds, and many pharmaceutical companies have made significant commitments to the CNS field. We believe cellular therapies, if proven safe and effective, will have unique properties that will make them desirable over small molecule drugs, none of which currently replace damaged tissue. However, any cell-based therapeutic to treat diseases of, or injuries to, the CNS is likely to face intense competition from the small molecule sector, biologics, as well as medical devices. We expect to compete with a host of companies, some of which are privately owned and some of which have resources far greater than ours.
In the liver field, there are no broad-based therapies for the treatment of liver disease at present. The primary therapy is liver transplantation, which is limited by the availability of matched donor organs. Liver-assist devices, when and if they become available, could also be used to help patients while they await suitably matched organs for
23
transplantation. Liver transplantation may remain the standard of care even if we successfully develop a cellular therapy. In addition, new therapies may become available before we successfully develop a cell-based therapy for liver disease.
Development of our technologies is subject to, and restricted by, extensive government regulation, which could impede our business.
Our research and development efforts, as well as any ongoing or future clinical trials, and the manufacturing and marketing of any products we may develop, will be subject to, and restricted by, extensive regulation by governmental authorities in the United States and other countries. The process of obtaining FDA and other necessary regulatory approvals is lengthy, expensive and uncertain. FDA and other legal and regulatory requirements applicable to the development and manufacture of the cells and cell lines required for our preclinical and clinical products could substantially delay or prevent us from producing the cells needed to initiate additional clinical trials. We or our collaborators may fail to obtain the necessary approvals to commence or continue clinical testing or to manufacture or market our potential products in reasonable time frames, if at all. In addition, the U.S. Congress and other legislative bodies may enact regulatory reforms or restrictions on the development of new therapies that could adversely affect the regulatory environment in which we operate or the development of any products we may develop.
We base our research and development on the use of human stem and progenitor cells obtained from human tissue, including fetal tissue. The U.S. federal and state governments and other jurisdictions impose restrictions on the acquisition and use of fetal tissue, including those incorporated in federal Good Tissue Practice, or cGTP,GTP, regulations. These regulatory and other constraints could prevent us from obtaining cells and other components of our products in the quantity or quality needed for their development or commercialization. These restrictions change from time to time and may become more onerous. Additionally, we may not be able to identify or develop reliable sources for the cells necessary for our potential products — that is, sources that follow all state and federal laws and guidelines for cell procurement. Certain components used to manufacture our stem and progenitor cell product candidates will need to be manufactured in compliance with the FDA’s Good Manufacturing Practices, or cGMP.GMP. Accordingly, we will need to enter into supply agreements with companies that manufacture these components to cGMPGMP standards.
Noncompliance with applicable requirements both before and after approval, if any, can subject us, our third party suppliers and manufacturers, and our other collaborators to administrative and judicial sanctions, such as, among other things, warning letters, fines and other monetary payments, recall or seizure of products, criminal proceedings, suspension or withdrawal of regulatory approvals, interruption or cessation of clinical trials, total or partial suspension of production or distribution, injunctions, limitations on or the elimination of claims we can make for our products, and refusal of the government to enter into supply contracts or fund research, or delay in approving or refusal to approve new drug applications.
We are dependent on the services of key personnel.
We are highly dependent on the principal members of our management and scientific staff and some of our outside consultants, including the members of our scientific advisory board, our chief executive officer, our chief operating officer, our vice presidents, and the heads of key departments or functions within the company. Although we have entered into employment agreements with some of these individuals, they may terminate their agreements at any time. In addition, our operations are dependent upon our ability to attract and retain additional qualified scientific and management personnel. We may not be able to attract and retain the personnel we need on acceptable terms given the competition for experienced personnel among pharmaceutical, biotechnology and health care companies, universities and research institutions.
22
Our activities involve hazardous materials and experimental animal testing; improper handling of these animals and materials by our employees or agents could expose us to significant legal and financial penalties.
Our research and development activities involve the controlled use of test animals as well as hazardous chemicals and potentially hazardous biological materials such as human tissue. Their use subjects us to
24
environmental and safety laws and regulations such as those governing laboratory procedures, exposure to blood-borne pathogens, use of laboratory animals, and the handling of biohazardous materials. Compliance with current or future laws and regulations may be expensive and the cost of compliance could adversely affect us.
Although we believe that our safety procedures for using, handling, storing, and disposing of hazardous and potentially hazardous materials comply with the standards prescribed by California and federal regulations, the risk of accidental contamination or injury from these materials cannot be eliminated. In the event of such an accident or of any violation of these or future laws and regulations, state or federal authorities could curtail our use of these materials; we could be liable for any civil damages that result, the cost of which could be substantial; and we could be subjected to substantial fines or penalties. In addition, any failure by us to control the use, disposal, removal, or storage, or to adequately restrict the discharge, or to assist in the cleanup, of hazardous chemicals or hazardous, infectious or toxic substances could subject us to significant liability. Any such liability could exceed our resources and could have a material adverse effect on our business, financial condition and results of operations. Moreover, an accident could damage our research and manufacturing facilities and operations and result in serious adverse effects on our business.
The development, manufacturing and commercialization of cell-based therapeutic products expose us to product liability claims, which could lead to substantial liability.
By developing and, ultimately, commercializing medical products, we are exposed to the risk of product liability claims. Product liability claims against us could result in substantial litigation costs and damage awards against us. We have obtained liability insurance that covers our clinical trials, and we will need to increase our insurance coverage if and when we begin commercializing products. We may not be able to obtain insurance on acceptable terms, if at all, and the policy limits on our insurance policies may be insufficient to cover our liability.
The manufacture of cell-based therapeutic products is novel, highly regulated, critical to our business, and dependent upon specialized key materials.
The proliferation and manufacture of cell-based therapeutic products are complicated and difficult processes, dependent upon substantial know-how and subject to the need for continual process improvements to be competitive. Our manufacturing experience is limited and the technologies are comparatively new. In addition, our ability toscale-up manufacturing to satisfy the various requirements of our planned clinical trials, such as GTP, GMP and release testing requirements, is uncertain. Manufacturing disruptions may occur and despite efforts to regulate and control all aspects of manufacturing, the potential for human or system failure remains. Manufacturing irregularities or lapses in quality control could have a serious adverse effect on our reputation and business, which could cause a significant loss of stockholder value. Many of the materials that we use to prepare our cell-based products are highly specialized, complex and available from only a limited number of suppliers.suppliers or derived from a biological origin. At present, some of our material requirements are single sourced, and the loss of one or more of these sources may adversely affect our business if we are unable to obtain alternatives or alternative sources at all or upon terms that are acceptable to us.
Because health care insurers and other organizations may not pay for our products or may impose limits on reimbursements, our ability to become profitable could be adversely affected.
In both domestic and foreign markets, sales of potential products are likely to depend in part upon the availability and amounts of reimbursement from third-party health care payor organizations, including government agencies, private health care insurers and other health care payors, such as health maintenance organizations and self-insured employee plans. There is considerable pressure to reduce the cost of therapeutic products. Government and other third party payors are increasingly attempting to contain health care costs by limiting both coverage and the level of reimbursement for new therapeutic products and by refusing, in some cases, to provide any coverage for
23
uses of approved products for disease indications for which the FDA or other relevant authority has not granted marketing approval. Moreover, in some cases, government and other third party payors have refused to provide reimbursement for uses of approved products for disease indications for which the FDA or other relevant authorityhasgranted marketing approval. Significant uncertainty exists as to the reimbursement status of newly approved health care products or novel therapies such as ours. Even if we obtain regulatory approval to market our products, we can give no assurance that reimbursement will be provided by such payors at all or without substantial delay or,
25
if such reimbursement is provided, that the approved reimbursement amounts will be sufficient to enable us to sell products we develop on a profitable basis. Changes in reimbursement policies could also adversely affect the willingness of pharmaceutical companies to collaborate with us on the development of our cellular technologies. In certain foreign markets, pricing or profitability of prescription pharmaceuticals is subject to government control. We also expect that there will continue to be a number of federal and state proposals to implement government control over health care costs. Efforts to change regulatory and reimbursement standards are likely to continue in future legislative sessions. We do not know what legislative proposals federal or state governments will adopt or what actions federal, state or private payors for health care goods and services may take in response to such proposals or legislation. We cannot predict the effect of government control and health care reimbursement practices on our business.
Ethical and other concerns surrounding the use of stem or progenitor-based cell therapy may negatively affect regulatory approval or public perception of our product candidates, which could reduce demand for our products or depress our stock price.
The use of stem cells for research and therapy has been the subject of debate regarding related ethical, legal and social issues. Although these concerns have mainly been directed to the use of embryonic stem cells, which we presently do not use, the distinction between embryonic and non-embryonic stem cells is frequently overlooked; moreover, our use of human stem or progenitor cells from fetal sources might raise these or similar concerns. Also, upon completion of the Acquisition of the assets of Stem Cell Sciences, we intend to continue the development of embryonic stem cells and iPS cells as potential research tools, and we may in the future explore their applicability as cell-based therapeutic products. Negative public attitudes toward stem cell therapy could result in greater governmental regulation of stem cell therapies, which could harm our business. For example, concerns regarding such possible regulation could impact our ability to attract collaborators and investors. Also, existing regulatory constraints on the use of embryonic stem cells may in the future be extended to use of fetal stem cells, and these constraints might prohibit or restrict us from conducting research or from commercializing products. Existing and potential U.S. government regulation of embryonic tissue may lead researchers to leave the field of stem cell research or the country altogether, in order to assure that their careers will not be impeded by restrictions on their work. Similarly, these factors may induce graduate students to choose other fields less vulnerable to changes in regulatory oversight, thus exacerbating the risk that we may not be able to attract and retain the scientific personnel we need in face of the competition among pharmaceutical, biotechnology and health care companies, universities and research institutions for what may become a shrinking class of qualified individuals.
Our corporate documents and Delaware law contain provisions that could make it difficult for us to be acquired in a transaction that might be beneficial to our stockholders.
Our board of directors has the authority to issue shares of preferred stock and to fix the rights, preferences, privileges, and restrictions of these shares without stockholder approval. In addition, we have adopted a rights plan that generally permits our existing stockholders to acquire additional shares at a substantial discount to the market price in the event of certain attempts by third parties to acquire us. These rights, along with certain provisions in our corporate documents, andalong with certain provisions under Delaware law, may make it more difficult for a third party to acquire us or discourage a third party from attempting to acquire us, even if the acquisition might be beneficial to our stockholders.
24
Risks Related to the Securities Market
Our stock price has been, and will likely continue to be, highly volatile, which may negatively affect our ability to obtain additional financing in the future.
The market price per share of our common stock has been and is likely to continue to be highly volatile due to the risks and uncertainties described in this section of this Annual Report onForm 10-K, as well as other factors, including:
| • | our ability to develop and test our technologies; | |
| • | our ability to patent or obtain licenses to necessary technologies; | |
| • | conditions and publicity regarding the industry in which we operate, as well as the specific areas our product candidates seek to address; |
26
| • | competition in our industry; | |
| • | economic and other external factors or other disasters or crises; | |
| • | price and volume fluctuations in the stock market at large that are unrelated to our operating performance; and | |
| • | comments by securities analysts, or our failure to meet market expectations. |
Over the two-year period ended December 31, 2007,2008, the trading price of our common stock as reported on the Nasdaq Global Market ranged from a high of $4.06$3.63 to a low of $1.40.$0.66 per share. As a result of this volatility, youran investment in our stock is subject to substantial risk. Furthermore, the volatility of our stock price could negatively impact our ability to raise capital or acquire businesses or technologies.
We are contractually obligated to issue shares in the future, diluting the interest of current stockholders.
As of December 31, 2007,2008, there were outstanding warrants to purchase 1,355,00011,599,828 shares of our common stock, at a weighted average exercise price of $1.85$2.05 per share. Also as of December 31, 2007, there wereshare, outstanding options to purchase 9,028,8108,340,530 shares of our common stock, at a weighted average exercise price of $2.36$2.32 per share.share, and outstanding restricted stock units for 1,650,000 shares of our common stock. In March 2009, we entered into an asset purchase agreement with Stem Cell Sciences Plc (“SCS”) to acquire substantially all of the operating assets and liabilities of SCS (the “Acquisition”). The Acquisition is subject to customary closing conditions, including the approval of the stockholders of SCS, and is expected to close shortly after the SCS extraordinary general meeting scheduled for March 27, 2009. As partial consideration for the operating assets and liabilities to be acquired, we will issue to SCS 2,650,000 shares of our common stock. Moreover, we expect to issue additional options to purchase shares of our common stock to compensate employees, consultants and directors, and may issue additional shares to raise capital, to acquire other companies or technologies, to pay for services, or for other corporate purposes. Any such issuances will have the effect of diluting the interest of current stockholders.
| Item 1B. | UNRESOLVED STAFF COMMENTS |
| Item 2. | PROPERTIES |
We entered into a5-year lease, as of February 1, 2001, for a 40,000 square foot facility, located in the Stanford Research Park in Palo Alto, California. This facility includes space for animals as well as laboratories, offices and a suite designed to be used to manufacture materials for clinical trials. Effective July 1, 2006, under an agreement that extends the lease through March 31, 2010, we leased the remainder of the building, adding approximately 27,500 square feet to our leased premises. We have a space-sharing agreement with Stanford University for part of the animal facility not needed for our own use.
25
We continue to lease the following facilities in Lincoln, Rhode Island obtained in connection with our former encapsulated cell technology: our former research laboratory and corporate headquarters building which contains 62,500 square feet of wet labs, specialty research areas and administrative offices held on a lease agreement that goes through June 2013, as well as a 21,000 square-foot pilot manufacturing facility and a 3,000 square-foot cell processing facility financed by bonds issued by the Rhode Island Industrial Facilities Corporation. We have subleased small portions of the 62,500 square foot facility, amounting to approximately 21 percent of the total space. We are actively seeking to sublease, assign or sell our remaining interests in these properties.
| Item 3. | LEGAL PROCEEDINGS |
In July 2006, we filed suit against Neuralstem, Inc., in the Federal District Court for the District of Maryland, alleging that Neuralstem’s activities violate claims in four of the patents we exclusively licensed from NeuroSpheres. Neuralstem has filed a motion for dismissal or summary judgment in the alternative, citing Title 35, Section 271(e)(1) of the United States Code, which says that it is not an act of patent infringement to make, use or sell a patented invention “solely for uses reasonably related to the development and submission of information” to the FDA. Neuralstem argues that because it does not have any therapeutic products on the market yet, the activities complained of fall within the protection of Section 271(e)(1) — that is, basically, that the suit is premature. This
27
issue will be decided after discovery is complete. Subsequent to filing its motion to dismiss, in December 2006, Neuralstem petitioned the U.S. Patent and Trademark Office (PTO) to reexamine two of the patents in our infringement action against Neuralstem, namely U.S. Patent No. 6,294,346 (claiming the use of human neural stem cells for drug screening) and U.S. Patent No. 7,101,709 (claiming the use of human neural stem cells for screening biological agents). In April 2007, Neuralstem petitioned the PTO to reexamine the remaining two patents in the suit, namely U.S. Patent No. 5,851,832 (claiming methods for proliferating human neural stem cells) and U.S. Patent No. 6,497,872 (claiming methods for transplanting human neural stem cells). These requests were granted by the PTO and, in June 2007, the parties voluntarily agreed to stay the pending litigation while the PTO considers these reexamination requests. In October 2007, Neuralstem petitioned the PTO to reexamine a fifth patent, namely U.S. Patent No. 6,103,530, which claims a culture medium for proliferating mammalian neural stem cells. In September 2007,April 2008, the PTO upheld the ’832 and ’872 patents, as amended, and issued first office actions in eachNotices of Intent to Issue anEx ParteReexamination Certificate for both. In August 2008, the first four reexaminations.PTO upheld the ’530 patent, as amended, and issued a Notice of Intent to Issue anEx ParteReexamination Certificate. The Company has since filed its first responses to each of these, and expects all fourremaining two patents to re-issue in 2008.are still under review by the PTO.
In 2003, Geron CorporationMay 2008, we filed an opposition toa second patent infringement suit against Neuralstem and its two founders, Karl Johe and Richard Garr. In this suit, which we filed in the Federal District Court for the Northern District of our issued European patent cases, namely EP0594669California, we allege that Neuralstem’s activities infringe claims in two patents we exclusively license from NeuroSpheres, specifically U.S. Patent No. 7,361,505 (claiming among other things, methods for proliferating and using human neural stem cells as therapeutic and drug screening agents) and EP0669973 (claiming, among other things, methods for proliferating and differentiating human neural stem cells). Both oppositions were heard in 2005, and the patents were maintained in somewhat altered form by the Opposition Divisioncomposition of the European Patent Office. In essence the scope of each patent was limited to proliferation using specific growth factors and each had to disclaim derivationmatter of human neural stem cells derived from any source material) and U.S. Patent No. 7,115,418 (claiming methods for proliferating human embryonic tissueneural stem cells). In addition, we allege various state law causes of action against Neuralstem arising out of its repeated derogatory statements to the public about our patent portfolio. Also in order to comply withMay 2008, Neuralstem filed suit against us and NeuroSpheres in the European law which precludesFederal District Court for the patentingDistrict of embryonic stem cells. The time for appeal has run in each case. U.S. counterparts to theseMaryland seeking a declaratory judgment that the ‘505 and ‘418 patents are part of our issued patent portfolio; theyeither invalid or are not subjectinfringed by Neuralstem and that Neuralstem has not violated California state law. In August 2008, the California court transferred our lawsuit against Neuralstem to opposition, becauseMaryland for resolution on the merits. We anticipate that procedure does not exist under U.S. patent law, although other types of proceedings may be availablethe Maryland District Court will consolidate these actions in some manner prior to third parties to contest our U.S. patents.trial.
| Item 4. | SUBMISSION OF MATTERS TO A VOTE OF SECURITY HOLDERS |
None.
26
PART II
| Item 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED SHAREHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
| (a) | Market price and dividend information |
Our stock is traded on the Nasdaq Global Market under the symbol STEM. The quarterly ranges of high and low bid prices per share for the last two fiscal years as reported by Nasdaq are shown below:
2007 | High | Low | ||||||
| First Quarter | $ | 3.63 | $ | 2.36 | ||||
| Second Quarter | $ | 3.09 | $ | 2.27 | ||||
| Third Quarter | $ | 2.45 | $ | 1.90 | ||||
| Fourth Quarter | $ | 2.53 | $ | 1.40 | ||||
2006 | ||||||||||||||||
| High | Low | |||||||||||||||
2008 | ||||||||||||||||
| First Quarter | $ | 4.06 | $ | 3.45 | $ | 1.90 | $ | 1.00 | ||||||||
| Second Quarter | $ | 3.58 | $ | 1.77 | $ | 1.75 | $ | 1.11 | ||||||||
| Third Quarter | $ | 2.55 | $ | 1.90 | $ | 1.43 | $ | 1.00 | ||||||||
| Fourth Quarter | $ | 3.49 | $ | 2.05 | $ | 2.48 | $ | 0.66 | ||||||||
2007 | ||||||||||||||||
| First Quarter | $ | 3.63 | $ | 2.36 | ||||||||||||
| Second Quarter | $ | 3.09 | $ | 2.27 | ||||||||||||
| Third Quarter | $ | 2.45 | $ | 1.90 | ||||||||||||
| Fourth Quarter | $ | 2.53 | $ | 1.40 | ||||||||||||
No cash dividends have been declared on our common stock since our inception.
2728
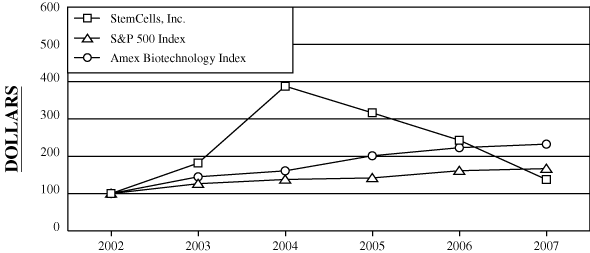
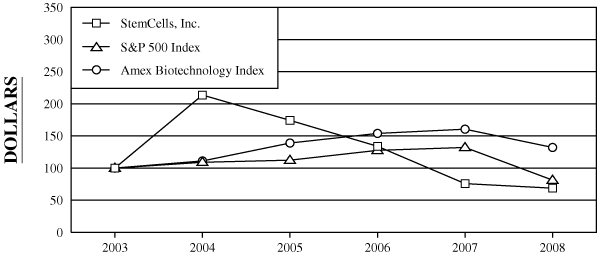
PERFORMANCE GRAPH
We show below the cumulative total return to our stockholders during the period from December 31, 20022003 through December 31, 2007200812 in comparison to the cumulative return on the Standard & Poor’s 500 Index and the Amex Biotechnology Index during that same period.
The stock price performance shown on the graph below is not necessarily indicative of future stock price performance.
| December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | |||||||||||||||||||||||||||||||||||||||||||||||||
| 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | |||||||||||||||||||||||||||||||||||||||||||||||||
| StemCells, Inc. | $ | 100.00 | $ | 181.65 | $ | 388.07 | $ | 316.51 | $ | 243.12 | $ | 137.61 | $ | 100.00 | $ | 213.64 | $ | 174.24 | $ | 133.84 | $ | 75.76 | $ | 68.69 | ||||||||||||||||||||||||||||||||||||
| S&P 500 Index | $ | 100.00 | $ | 126.38 | $ | 137.75 | $ | 141.88 | $ | 161.20 | $ | 166.89 | $ | 100.00 | $ | 108.99 | $ | 112.26 | $ | 127.55 | $ | 132.06 | $ | 81.23 | ||||||||||||||||||||||||||||||||||||
| Amex Biotechnology Index | $ | 100.00 | $ | 144.91 | $ | 160.92 | $ | 201.32 | $ | 223.01 | $ | 232.54 | $ | 100.00 | $ | 111.05 | $ | 138.93 | $ | 153.9 | $ | 160.48 | $ | 132.05 | ||||||||||||||||||||||||||||||||||||
The information under “Performance Graph” is not deemed filed with the Securities and Exchange Commission and is not to be incorporated by reference in any filing of StemCells, Inc. under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this10-K and irrespective of any general incorporation language in those filings.
| (b) | Approximate Number of Holders of Common Stock |
As of February 29, 2008,27, 2009, there were approximately 582590 holders of record of our common stock and the closing price per share of our common stock on the Nasdaq Global Market was $1.51.$1.56.
The number of record holders is based upon the actual number of holders registered on the books of our transfer agent at such date and does not include holders of shares in “street names” or persons, partnerships, associations, corporations or other entities identified in security position listings maintained by depository trust companies.
| (c) | Recent Sales of Unregistered Securities (last three years ending December 31, 2008) |
We issued the following unregistered securities in 2008:
| • | In September 2008, we issued 6,924 shares of common stock to the California Institute of Technology (Cal Tech) for payment of annual fees of $5,000 for each of two patent families to which we hold a license from |
2 Cumulative total returns assumes a hypothetical investment of $100 on December 31, 2007)2003.
29
| Cal Tech, payable in cash or stock at our choice. We elected to pay these fees in stock. The shares were issued in a transaction not involving any public offering pursuant to Section 4(2) of the Securities Act of 1933, as amended. |
We issued the following unregistered securities in 2007:
| • | In June 2007, we issued 3,865 shares of common stock to the California Institute of Technology (Cal Tech) for payment of annual fees of $5,000 for each of two |
28
We issued the following unregistered securities in 2006:
| • | In August 2006, we issued 3,848 shares of common stock to the California Institute of Technology (Cal Tech) as payment of annual fees of $5,000 for each of two |
Equity Compensation Plan Information
The following table provides certain information with respect to all of our equity compensation plans in effect as of December 31, 2007.2008.
| Equity Compensation Plan Information | ||||||||||||
| Number of Securities | ||||||||||||
| Number of Securities to | Remaining Available for | |||||||||||
| be Issued Upon | Weighted-average | Future Issuance Under Equity | ||||||||||
| Exercise of | Exercise Price of | Compensation Plans | ||||||||||
| Outstanding Options, | Outstanding Options, | (Excluding Securities | ||||||||||
| Warrants and Rights | Warrants and rights | Reflected in Column(a)) | ||||||||||
Plan category | (a) | (b) | (c) | |||||||||
| Equity compensation plans approved by security holders(1) | 9,028,810 | $ | 2.36 | 3,024,408 | ||||||||
| Equity compensation arrangements not approved by security holders(2) | 100,000 | $ | 1.20 | N/A | ||||||||
| Totals | 9,128,810 | $ | 2.33 | 3,024,408 | ||||||||
| Equity Compensation Plan Information | ||||||||||||
| Number of Securities to | Number of Securities | |||||||||||
| be Issued Upon | Weighted-Average | Remaining Available for | ||||||||||
| Exercise of | Exercise Price of | Future Issuance Under Equity | ||||||||||
| Outstanding Stock | Outstanding Stock | Compensation Plans | ||||||||||
| Options, | Options, | (Excluding Securities | ||||||||||
| Warrants and Rights | Warrants and Rights | Reflected in Column(a)) | ||||||||||
Plan Category | (a) | (b) | (c) | |||||||||
| Equity compensation plans approved by security holders(1) | 9,990,530 | $ | 1.93 | 4,571,429 | ||||||||
| (1) | Consists of stock options issued to employees and directors, restricted stock units issued to employees and stock options issued as compensation to consultants for consultation services. These stock options and restricted stock units were issued under our 1992 Equity Incentive Plan, Directors’ Stock Option Plan, StemCells, Inc. Stock Option Plan, or our 2001, 2004 and 2006 Equity Incentive Plans. | |
30
| Item 6. | SELECTED FINANCIAL DATA |
The following selected financial and operating data are derived from our audited consolidated financial statements. The selected financial and operating data should be read in conjunction with “Item 7. Management’s
29
Discussion and Analysis of Financial Condition and Results of Operation” and the consolidated financial statements and notes thereto contained elsewhere in thisForm 10-K.
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | 2004 | 2003 | 2008 | 2007 | 2006 | 2005 | 2004 | |||||||||||||||||||||||||||||||
| (In thousands, except per share amounts) | (In thousands, except per share amounts) | |||||||||||||||||||||||||||||||||||||||
| Consolidated Statement of Operations | ||||||||||||||||||||||||||||||||||||||||
| Consolidated Statements of Operations | ||||||||||||||||||||||||||||||||||||||||
| Revenue from licensing agreements and grants | $ | 57 | $ | 93 | $ | 206 | $ | 141 | $ | 273 | $ | 232 | $ | 57 | $ | 93 | $ | 206 | $ | 141 | ||||||||||||||||||||
| Research and development expenses | 19,937 | 13,600 | 8,226 | 7,844 | 5,479 | 17,808 | 19,937 | 13,600 | 8,226 | 7,844 | ||||||||||||||||||||||||||||||
| General and administrative expenses | 7,927 | 7,154 | 5,540 | 4,870 | 4,056 | 8,296 | 7,927 | 7,154 | 5,540 | 4,870 | ||||||||||||||||||||||||||||||
| Wind-down expenses | 783 | 709 | 2,827 | 2,827 | 2,885 | 866 | 783 | 709 | 2,827 | 2,827 | ||||||||||||||||||||||||||||||
| License & settlement agreement income, net(2) | 551 | 103 | 3,736 | — | — | |||||||||||||||||||||||||||||||||||
| Write down for other than temporary impairment of marketable securities(3) | 2,083 | — | — | — | — | |||||||||||||||||||||||||||||||||||
| Loss on change in fair value of warrant liability(4) | 937 | — | — | — | — | |||||||||||||||||||||||||||||||||||
| License & settlement agreement income, net(5) | — | 551 | 103 | 3,736 | — | |||||||||||||||||||||||||||||||||||
| Gain on sale of marketable securities | 716 | — | — | — | — | — | 716 | — | — | — | ||||||||||||||||||||||||||||||
| Loss before deemed dividends and cumulative effect of change in accounting principle | (25,023 | ) | (18,948 | ) | (11,738 | ) | (15,330 | ) | (12,291 | ) | ||||||||||||||||||||||||||||||
| Net loss | (25,023 | ) | (18,948 | ) | (11,738 | ) | (15,330 | ) | (14,425 | ) | (29,087 | ) | (25,023 | ) | (18,948 | ) | (11,738 | ) | (15,330 | ) | ||||||||||||||||||||
| Basic and diluted loss per share | $ | (0.31 | ) | $ | (0.25 | ) | $ | (0.18 | ) | $ | (0.31 | ) | $ | (0.45 | ) | $ | (0.35 | ) | $ | (0.31 | ) | $ | (0.25 | ) | $ | (0.18 | ) | $ | (0.31 | ) | ||||||||||
| Shares used in computing basic and diluted loss per share amounts | 79,772 | 74,611 | 63,643 | 49,606 | 32,080 | 82,716 | 79,772 | 74,611 | 63,643 | 49,606 | ||||||||||||||||||||||||||||||
| December 31, | December 31, | |||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | 2004 | 2003 | 2008 | 2007 | 2006 | 2005 | 2004 | |||||||||||||||||||||||||||||||
| (In thousands) | (In thousands) | |||||||||||||||||||||||||||||||||||||||
| Consolidated Balance Sheet | ||||||||||||||||||||||||||||||||||||||||
| Consolidated Balance Sheets | ||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 9,759 | $ | 51,795 | $ | 34,541 | $ | 41,060 | $ | 13,082 | $ | 30,043 | $ | 9,759 | $ | 51,795 | $ | 34,541 | $ | 41,060 | ||||||||||||||||||||
| Marketable securities | 29,847 | 7,266 | 3,721 | — | — | 4,182 | 29,847 | 7,266 | 3,721 | — | ||||||||||||||||||||||||||||||
| Total assets | 48,283 | 66,857 | 44,839 | 47,627 | 19,786 | 41,230 | 48,283 | 66,857 | 44,839 | 47,627 | ||||||||||||||||||||||||||||||
| Accrued wind-down expenses | 6,143 | 6,750 | 7,306 | 5,528 | 3,823 | 5,513 | 6,143 | 6,750 | 7,306 | 5,528 | ||||||||||||||||||||||||||||||
| Fair value of warrant liability(4) | 8,440 | — | — | — | — | |||||||||||||||||||||||||||||||||||
| Long-term debt, including capital leases | 1,034 | 1,145 | 1,351 | 1,646 | 1,850 | 867 | 1,034 | 1,145 | 1,351 | 1,646 | ||||||||||||||||||||||||||||||
| Stockholders’ equity | 35,212 | 54,376 | 32,376 | 36,950 | 10,964 | 21,809 | 35,212 | 54,376 | 32,376 | 36,950 | ||||||||||||||||||||||||||||||
| (1) | Effective January 1, 2006, we adopted Statement of Financial Accounting Standards 123 (revised 2004) (SFAS 123R),Share-Based Payment, in accordance with the provisions of SFAS 123R, we elected to adopt the standard using the modified prospective method. SFAS 123R requires us to recognize in operating expenses, the fair value of our stock-based compensation awards. See Note 7 “Stock-Based Compensation” in the Notes to the Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information. | |
| (2) | Relates to wind-down expenses in respect of our Rhode Island facility. See Note | |
| Relates to | ||
| (4) | Relates to the fair value of warrants issued as part of our financing in November 2008. See Note 10 “Warrant Liability” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information. | |
| (5) | Relates to an agreement with ReNeuron. See Note 2 “Financial Instruments” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information. |
31
| Item 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
This report contains forward looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities Exchange Act that involve substantial risks and uncertainties. Such statements include, without limitation, all statements as to expectation or belief and statements as to our future results of operations; the progress of our research, product development and clinical programs; the need for, and timing of, additional capital and capital expenditures; partnering prospects; costs of manufacture of products; the protection of, and the need for, additional intellectual property rights; effects of regulations; the need for additional facilities; and potential market opportunities. Our actual results may vary materially from those contained in such forward-looking statements because of risks to which we are subject, including uncertainty as to whether the U.S. Food and Drug Administration (FDA) or other regulatory authorities will permit us to proceed with clinical testing of proposed products despite the novel and unproven nature of our technologies; the risk that our initial clinical trial and any other clinical trials or studies could be substantially delayed beyond their expected dates or cause us to incur substantial unanticipated costs; uncertainties in our ability to obtain the capital resources needed to continue our current research and development operations and to conduct the research, preclinical development and clinical trials necessary for regulatory approvals; the uncertainty regarding our ability to obtain a corporate partner or partners, if
30
needed, to support the development and commercialization of our potential cell-based therapeutics products; the uncertainty regarding the outcome of our Phase I clinical trial in NCL and any other clinical trials or studies we may conduct in the future;conduct; the uncertainty regarding the validity and enforceability of our issued patents; the uncertainty whether any products that may be generated in our cell-based therapeutics programs will prove clinically safe and effective; the uncertainty whether we will achieve revenue from product sales or become profitable; uncertainties regarding our obligations with respect to our former encapsulated cell therapy facilities in Rhode Island; obsolescence of our technologies; competition from third parties; intellectual property rights of third parties; litigation risks; and other risks to which we are subject. All forward-looking statements attributable to us or to persons acting on our behalf are expressly qualified in their entirety by the cautionary statements and risk factors set forth in “Risk Factors” in Part I, Item 1A of thisForm 10-K.
Overview
The Company
Our research and development (R&D) programs are focused on identifying and developing potential cell-based therapeutics which can either restore or support organ function. Since we relocated our corporate headquarters and research laboratories to California in 1999 our R&D efforts have primarily been directed at refining our methods for identifying, isolating, culturing, and purifying the human neural stem cell and human liver engrafting cells (hLEC) and developing these as potential cell-based therapeutics for the central nervous system (CNS) and the liver, respectively. We are currently conducting a Phase I clinical trial ofIn our CNS Program, our HuCNS-SC® product candidate (purified human neural stem cells) is in clinical development for two indications. In January 2009, we completed a six patient Phase I clinical trial to evaluate the safety and preliminary efficacy of HuCNS-SC® cells as a treatment for infantile and late infantile neuronal ceroid lipofuscinosis (NCL), two forms of a fatal neurodegenerative diseasegroup of disorders often referred to as Batten disease. We have completed enrollmentexpect to complete data analysis and dosing for this six-patientto report the trial results in mid 2009. In December 2008, the FDA approved our IND to initiate a Phase I clinical trial of HuCNS-SC cells in a second indication, Pelizeaus-Merzbacher Disease (PMD), a fatal myelination disorder in the brain. We expect the PMD trial to begin enrolling patients in 2009 and expect itthat the trial will take12-18 months to be completedcomplete. In addition, our HuCNS-SC cells are in early 2009. Our CNS Program is continuing basic research and preclinical development for additional potential indications in the CNS field. We are targeting to initiate clinical trials to test our HuCNS-SC product candidate for a spinal cord indication by mid-2008injury and for a myelin disorder in the brain by the end of 2008.retinal disorders. In our Liver Program, we are in preclinical development with our human liver engrafting cells (hLEC) and are exploring their applicabilitywe plan to seek the necessary approvals to initiate a clinical study to evaluate hLEC as a potential cellular therapy, with the initial indication likely to restore function to liver tissue by replacing dysfunctional or damaged cells.be liver-based metabolic disorders. See Overview “Research and Development Programs” in the Business Section of Part I, Item 1 of thisForm 10-K for a brief description of our significant research and development programs. We have also conducted research on several other cell types and in other areas, which could lead to other possible product candidates, process improvements or further research activities.
We have not derived any revenue or cash flows from the sale or commercialization of any products except for license revenue for certain of our patented cells and media for use in research. As a result, we have incurred annual operating losses since inception and expect to incur substantial operating losses in the future. Therefore, we are
32
dependent upon external financing from equity and debt offerings and revenue from collaborative research arrangements with corporate sponsors to finance our operations. We have no such collaborative research arrangements at this time and there can be no assurance that such financing or partnering revenue will be available when needed or on terms acceptable to us.
Before we can derive revenue or cash inflows from the commercialization of any of our therapeutic product candidates, we will need to: (i) conduct substantialin vitrotesting and characterization of our proprietary cell types, (ii) undertake preclinical and clinical testing for specific disease indications; (iii) develop, validate andscale-up manufacturing processes to produce these cell-based therapeutics, and (iv) pursue required regulatory approvals. These steps are risky, expensive and time consuming.
Overall, we expect our R&D expenses to be substantial and to increase for the foreseeable future as we continue the development and clinical investigation of our current and future product candidates. However, expenditures on R&D programs are subject to many uncertainties, including whether we develop our product candidates with a partner or independently. We cannot forecast with any degree of certainty which of our current product candidates will be subject to future collaboration, when such collaboration agreements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements. In addition, there are numerous factors associated with the successful commercialization of any of our cell-based therapeutics, including future trial design and regulatory requirements, many of which cannot be determined with accuracy at this
31
time given the stage of our development and the novel nature of stem cell technologies. The regulatory pathways, both in the United States and internationally, are complex and fluid given the novel and, in general, clinically unproven nature of stem cell technologies. At this time, due to such uncertainties and inherent risks, we cannot estimate in a meaningful way the duration of, or the costs to complete, our R&D programs or whether, when or to what extent we will generate revenues or cash inflows from the commercialization and sale of any of our product candidates. While we are currently focused on advancing each of our product development programs, our future R&D expenses will depend on the determinations we make as to the scientific and clinical prospects of each product candidate, as well as our ongoing assessment of the regulatory requirements and each product candidate’s commercial potential. If we are successful in completing the Acquisition of the business of SCS, we would expect our expenses and expenditures to increase.
Given the early stage of development of our product candidates, any estimates of when we may be able to commercialize one or more of these products would not be meaningful. Moreover, any estimate of the time and investment required to develop potential products based upon our proprietary HuCNS-SC and hLEC technologies will change depending on the ultimate approach or approaches we take to pursue them, the results of preclinical and clinical studies, and the content and timing of decisions made by the FDA and other regulatory authorities. There can be no assurance that we will be able to develop any product successfully, or that we will be able to recover our development costs, whether upon commercialization of a developed product or otherwise. We cannot provide assurance that any of these programs will result in products that can be marketed or marketed profitably. If certain of our development-stage programs do not result in commercially viable products, our results of operations could be materially adversely affected.
Significant Events
In January 2008, we completed enrollment and dosing of a six-patient Phase I clinical trial of our HuCNS-SC product candidate as a treatment for infantile and late infantile neuronal ceroid lipofuscinosis (NCL) at Oregon Health & Science University (OHSU) Doernbecher Children’s Hospital.
In January 2008, we entered into a research collaboration with the OHSU Casey Eye Institute to evaluate our HuCNS-SC product candidate as a potential treatment for retinal degeneration, a leading cause of blindness.
In December 2007, we announced that we were exploringApril 2008, the possible acquisition of privately held Progenitor Cell Therapy, LLC (PCT), a provider of cGMP-quality cell processing services headquartered in Hackensack, NJ. PCT agreed to a period of exclusivity to allow for due diligenceU.S. Patent and negotiations, and in consideration thereof, we agreed to make a secured loan of up to $3.8 million to PCT, of which $1.0 million was lent. In March 2008, we terminated discussions to acquire PCT because the parties were unable to reach mutually agreeable terms and conditions. We anticipate the $1.0 million loan will be repaid in accordanceTrademark Office issued U.S. Patent Number 7,361,505 with its terms.
3233
In June 2008, U.S. Patent and Trademark Office issued U.S. Patent Number 7,381,561 claiming the use of additional monoclonal antibodies for the prospective isolation of rare cells from human neural tissue, such as our HuCNS-SC product candidate. The ’561 patent is assigned to us.
In September 2008, Stewart Craig, Ph.D. joined us as Senior Vice President, Development and Operations, with responsibility for process design and engineering, GMP manufacturing operations, regulatory affairs, quality assurance, facilities and supply chain management. Dr. Craig has over twenty-five years of experience in the biotechnology sector, the last 15 of which have been in the cell therapy field.
In October 2008, we were awarded a $305,000 grant from the National Institute of Diabetes and Digestive and Kidney Diseases to research and develop a potential cell-based therapeutic for liver disease arising from infection by the hepatitis C virus. This grant will fund work over the next year to investigate whether our human liver engrafting cells can be made resistant to infection by the hepatitis C virus.
In November 2008, we reported that our HuCNS-SC cells, when transplanted into a well-established animal model, can protect the retina from progressive degeneration and prevent the loss of visual function. Retinal degeneration leads to loss of vision in diseases such as age-related macular degeneration and retinitis pigmentosa.
In November 2008, we raised approximately $20 million in gross proceeds through the sale of approximately 13.8 million units at $1.45 per unit. Each unit consisted of one share of common stock and a warrant to purchase 0.75 shares of common stock at an exercise price of $2.30 per share. We received total proceeds, net of offering expenses and placement agency fees, of approximately $18.6 million.
In December 2008, the FDA approved our IND to initiate a clinical trial of our HuCNS-SC product candidate to treat Pelizaeus-Merzbacher Disease (PMD), a fatal brain disorder that mainly affects young children. This Phase I trial, which is designed to evaluate the safety and preliminary efficacy of HuCNS-SC cells as a treatment for PMD, is expected to begin enrolling patients in 2009.
In January 2007, Desmond H. O’Connell, Jr., joined2009, we completed the six-patient Phase I clinical trial of our Board of Directors. CurrentlyHuCNS-SC product candidate as a treatment for infantile and late infantile NCL.
In March 2009, we entered into an independent management consultant and private investor, Mr. O’Connell is a director of Abiomed, Inc. and was formerly a director, acting chief executive officer and chairmanasset purchase agreement with Stem Cell Sciences Plc (“SCS”) to acquire substantially all of the boardoperating assets and liabilities of Serologicals Corporation until that company was sold in 2006.SCS (the proposed “Acquisition”). The Acquisition is subject to the approval of the stockholders of SCS and other customary closing conditions, and is expected to close shortly after the SCS extraordinary general meeting scheduled for March 27, 2009.
Critical Accounting Policies and the Use of Estimates
The accompanying discussion and analysis of our financial condition and results of operations are based on our Consolidated Financial Statements and the related disclosures, which have been prepared in accordance with U.S. generally accepted accounting principles (GAAP). The preparation of these Consolidated Financial Statements requires management to make estimates, assumptions, and judgments that affect the reported amounts in our Consolidated Financial Statements and accompanying notes. These estimates form the basis for making judgments about the carrying values of assets and liabilities. We base our estimates and judgments on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, and we have established internal controls related to the preparation of these estimates. Actual results and the timing of the results could differ materially from these estimates.
Warrant Liability
We account for our warrants in accordance with Emerging Issues Task Force IssueNo. 00-19,Accounting for Derivative Financial Instruments Indexed to and Potentially Settled in a Company’s Own Stock(EITF 00-19), which defines how freestanding contracts that are indexed to and potentially settled in a company’s own stock should be measured and classified. The general concept underEITF 00-19 is that contracts that could require net-cash settlement should be classified as assets or liabilities and contracts that only provide for settlement in shares should be classified as equity. In order for a contract to be classified as equity, each of the specific conditions enumerated inEITF 00-19 must be met; these conditions are intended to identify situations in which net cash
34
settlement could be forced upon the issuer. As part of our November 2008 financing, we issued warrants with a five year term to purchase 10,344,828 shares of our common stock at $2.30 per share. In accordance withEITF 00-19, we are required to classify the fair value of the warrants issued as a liability, with subsequent changes in fair value to be recorded as income (loss) on change in fair value of warrant liability. The fair value of the warrants is determined using the Black-Scholes-Merton (Black-Scholes) option pricing model and is affected by changes in inputs to that model including our stock price, expected stock price volatility and contractual term. We will continue to classify the fair value of the warrants as a liability until the warrants are exercised, expire or are amended in a way that would no longer require these warrants to be classified as a liability. The estimated fair value of our warrant liability at December 31, 2008, was approximately $8,440,000.
Stock-Based Compensation
On January 1, 2006, we adopted Statement of Financial Accounting Standards No. 123 (revised 2004) (SFAS 123R),Share-Based Payment, which revises SFAS 123,Accounting for Stock-Based Compensation, and supersedes Accounting Principles Board Opinion 25,Accounting for Stock Issued to Employees. SFAS 123R requires us to recognize expense related to the fair value of our stock-based compensation awards, including employee stock options.options and restricted stock units. Under the provisions of SFAS 123R, employee stock-based compensation is estimated at the date of grant based on the award’s fair value using the Black-Scholes-Merton (Black-Scholes)Black-Scholes option-pricing model and is recognized as expense ratably over the requisite service period. The Black-Scholes option-pricing model requires the use of certain assumptions, the most significant of which are our estimates of the expected volatility of the market price of our stock, and the expected term of the award.award, and the risk-free interest rate. Our estimate of the expected volatility is based on historical volatility. We currently useThe expected term represents the simplified methodperiod during which our stock-based awards are expected to be outstanding. In 2008 we estimated this amount based on historical experience of similar awards, giving consideration to the contractual terms of the awards, vesting requirements, and expectation of future employee behavior, including post-vesting terminations. Our estimate of the risk-free interest rate is based on U.S. Treasury debt securities with maturities close to the expected term of the option. Under this method, the expected term is the averageoption as of the contractual lifedate of the option and its vesting period.grant. As required under SFAS 123R, we review our valuation assumptions at each grant date and, as a result, our assumptions in future periods may change. For the year ended December 31, 2007,2008, employee stock-based compensation expense was approximately $3,061,000.$3,755,000. As of December 31, 2007,2008, total compensation cost related to unvested stock options and restricted stock units not yet recognized was approximately $6,956,000,$5,207,000, which is expected to be recognized as expense over a weighted-average period of 1.62.1 years.
Wind-down expenses
In connection with our wind-down of our research and manufacturing operations in Lincoln, Rhode Island, and the relocation of our corporate headquarters and remaining research laboratories to California in October 1999, we provided a reserve for our estimate of the exit cost obligation in accordance withEITF 94-3,Liability Recognition for Certain Employee Termination Benefits and Other Costs to Exit an Activity (Including Certain Costs Incurred in a RestructuringRestructuring). The reserve reflects estimates of the ongoing costs of our former research and administrative facility in Lincoln, which we hold on a lease that terminates on June 30, 2013. We are seeking to sublease, assign, sell, or otherwise divest ourselves of our interest in the facility at the earliest possible time, but we cannot determine with certainty a fixed date by which such events will occur, if at all.
In determining the facility exit cost reserve amount, we are required to consider our lease payments through to the end of the lease term and estimate other relevant factors such as facility operating expenses, real estate market conditions in Rhode Island for similar facilities, occupancy rates, and sublease rental rates projected over the course of the leasehold. We re-evaluate the estimate each quarter, taking into account of changes, if any, in each of the underlying factor.factors. The process is inherently subjective because it involves projections overinto time — from the date of the estimate through the end of the lease — and it is not possible to determine any of the factors except the lease payments with certainty over that period.
33
Management forms its best estimate on a quarterly basis, after considering actual sublease activity, reports from our broker/realtor about current and predicted real estate market conditions in Rhode Island, the likelihood of new subleases in the foreseeable future for the specific facility and significant changes in the actual or projected operating expenses of the property. We discount the projected net outflow over the term of the leaseholdlease to arrive at the
35
present value, and adjust the reserve to that figure. The estimated vacancy rate for the facility is an important assumption in determining the reserve because changes in this assumption have the greatest effect on estimated sublease income. In addition, the vacancy rate estimate is the variable most subject to change, while at the same time it involves the greatest judgment and uncertainty due to the absence of highly predictive information concerning the future of the local economy and future demand for specialized laboratory and office space in that area. The average vacancy rate of the facility over the last fivesix years (2003 through 2007)2008) was approximately 73%74%, varying from 66%62% to 89%. As of December 31, 2007,2008, based on current information available to management, the vacancy rate is projected to be 80%approximately 78% for 2008,2009, and approximately 70% from 20092010 through the end of the lease. These estimates are based on actual occupancy as of December 31, 2007,2008, predicted lead time for acquiring new subtenants, historical vacancy rates for the area and assessments by our broker/realtor of future real estate market conditions. If the assumed vacancy rate for 20092010 to the end of the lease had been five percentage points higher or lower at December 31, 2007,2008, then the reserve would have increased or decreased by approximately $214,000.$178,000. Similarly, a 5% increase or decrease in the operating expenses for the facility from 2008 on would have increased or decreased the reserve by approximately $122,000,$118,000, and a 5% increase or decrease in the assumed average rental charge per square foot would have increased or decreased the reserve by approximately $80,000.$54,000. Management does not wait for specific events to change its estimate, but instead uses its best efforts to anticipate them on a quarterly basis.
For the year ended December 31, 2007,2008, we recorded actual expenses against this reserve net of subtenant income of approximately $1,420,000.$1,293,000. Based on management’s evaluation of the factors mentioned above, and particularly the projected vacancy rates described above, we adjusted the reserve to $6,143,000$5,513,000 at December 31, 20072008 by recording an additional $783,000$866,000 as wind-down expenses for the year ended December 31, 2007.2008.
Income Taxes
We account for income taxes in accordance with SFAS No. 109, Accounting for Income Taxes(SFAS 109) and FASB Interpretation No. 48,Accounting for Uncertainty in Income Taxes, an interpretation of FASB Statement No. 109,as amended by FASB Staff PositionNo. 48-1 (FIN 48). Under SFAS 109 and FIN 48, we must recognize deferred tax assets and liabilities for expected future tax consequences of temporary differences between the carrying amounts and tax bases of assets and liabilities. Income tax receivables and liabilities, and deferred tax assets and liabilities, are recognized based on the amounts that more likely than not would be sustained upon ultimate settlement with taxing authorities.
Developing our provision for income taxes and analyzing our tax positions requires significant judgment and knowledge of federal and state income tax laws, regulations and strategies, including the determination of deferred tax assets and liabilities and, any valuation allowances that may be required for deferred tax assets.
We assess the likelihood of realizing our deferred tax assets to determine whether an income tax valuation allowance is required. Based on such evidence that can be objectively verified, we determine whether it is more likely than not that all or a portion of the deferred tax assets will be realized. The main factors that we consider include:
| • | cumulative losses in recent years; | |
| • | income/losses expected in future years; and | |
| • | the applicable statute of limitations. |
Tax benefits associated with uncertain tax positions are recognized in the period in which one of the following conditions is satisfied: (1) the more likely than not recognition threshold is satisfied; (2) the position is ultimately settled through negotiation or litigation; or (3) the statute of limitations for the taxing authority to examine and challenge the position has expired. Tax benefits associated with an uncertain tax position are reversed in the period in which the more likely than not recognition threshold is no longer satisfied.
34
We concluded that the realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance.
36
Contingencies
We are currently involved in certain legal proceedings. See Note 8,9, “Commitments and Contingencies,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information on these matters.
Results of Operations
Our results of operations have varied significantly from year to year and quarter to quarter and may vary significantly in the future due to the occurrence of material recurring and nonrecurring events, including without limitation the receipt and payment of recurring and nonrecurring licensing payments, the initiation or termination of research collaborations, the on-going expenses to lease and maintain our Rhode Island facilities, other than temporary impairment of our financial assets, changes in estimated fair value of our warrant liability, and the increasing costs associated with operating our California facility. To expandfacility and provide high quality systems and support toexpanding our research and development programs, we will need to hire more personnel, which will lead to higher operating expenses.operations.
Revenue
Revenue totaled approximately $232,000 in 2008, $57,000 in 2007, and $93,000 in 2006, and $206,000 in 2005.2006.
| Change in | Change in | Change in | Change in | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2008 | 2007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Versus 2006 | Versus 2005 | Versus 2007 | Versus 2006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | $ | % | $ | % | 2008 | 2007 | 2006 | $ | % | $ | % | |||||||||||||||||||||||||||||||||||||||||||
| Revenue | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Licensing agreements and grants | $ | 56,722 | $ | 92,850 | $ | 205,914 | $ | (36,128 | ) | (39 | )% | $ | (113,064 | ) | (55 | )% | $ | 231,730 | $ | 56,722 | $ | 92,850 | $ | 175,008 | 309 | % | $ | (36,128 | ) | (39 | )% | |||||||||||||||||||||||||
The increase in licensing and grant revenue in 2008 as compared to 2007 was primarily attributable to the receipt of a $150,000 milestone payment under a license agreement. In addition, in October 2008, we were awarded a $305,000 grant from the National Institute of Diabetes and Digestive and Kidney Diseases to research and develop a potential cell-based therapeutic for liver disease arising from infection by the hepatitis C virus. The award is a Phase I grant under the Small Business Innovation Research (SBIR) Program of the National Institutes of Health. We recognized approximately $26,000 as grant revenue in 2008. The decrease in licensing and grant revenue in 2007 as compared to 2006 was primarily attributable to the completed draw down in 2006 of a $464,000 Small Business Technology Transfer Grant for studies in Alzheimer’s disease that was awarded in September 2004. The grant supported joint work with Dr. George A. Carlson of the McLaughlin Research Institute (MRI) in Great Falls, Montana. The decrease in licensing and grant revenue in 2006 as compared to 2005 was primarily attributable to lesser amounts drawn down in 2006 from the grant as compared to 2005. We received and recognized approximately $38,000 in 2006, $186,000 in 2005, and $26,000 in 2004 as grant revenue the remainder was reimbursed to MRI.for this study.
35
Operating Expenses
Operating expense totaled approximately $26,970,000 in 2008, $28,648,000 in 2007, and $21,464,000 in 2006, and $16,594,000 in 2005.2006.
| Change in | Change in | Change in | Change in | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2008 | 2007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Versus 2006 | Versus 2005 | Versus 2007 | Versus 2006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | $ | % | $ | % | 2008 | 2007 | 2006 | $ | % | $ | % | |||||||||||||||||||||||||||||||||||||||||||
| Operating Expenses | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Research & development | $ | 19,937,426 | $ | 13,600,433 | $ | 8,226,734 | $ | 6,336,993 | 47 | % | $ | 5,373,699 | 65 | % | $ | 17,808,009 | $ | 19,937,426 | $ | 13,600,433 | $ | (2,129,417 | ) | (11 | )% | $ | 6,336,993 | 47 | % | |||||||||||||||||||||||||||
| General & administrative | 7,927,443 | 7,154,042 | 5,539,845 | 773,401 | 11 | % | 1,614,197 | 29 | % | 8,295,554 | 7,927,443 | 7,154,042 | 368,111 | 5 | % | 773,401 | 11 | % | ||||||||||||||||||||||||||||||||||||||
| Wind-down expenses | 783,022 | 709,209 | 2,827,403 | 73,813 | 10 | % | (2,118,194 | ) | (75 | )% | 866,199 | 783,022 | 709,209 | 83,177 | 11 | % | 73,813 | 10 | % | |||||||||||||||||||||||||||||||||||||
| Total expense | $ | 28,647,891 | $ | 21,463,684 | $ | 16,593,982 | $ | 7,184,207 | 33 | % | $ | 4,869,702 | 29 | % | ||||||||||||||||||||||||||||||||||||||||||
| Total operating expenses | $ | 26,969,762 | $ | 28,647,891 | $ | 21,463,684 | $ | (1,678,129 | ) | (6 | )% | $ | 7,184,207 | 33 | % | |||||||||||||||||||||||||||||||||||||||||
Research and Development Expenses
Our R&D expenses consist primarily of salaries and related personnel expenses, costs associated with clinical trials and regulatory submissions; costs associated with preclinical activities such as toxicology studies; costs associated with cell processing and process development; certain patent-related costs such as licensing; facilities-relatedfacilities-
37
related costs such as depreciation; lab equipment and supplies. Clinical trial expenses include payments to vendors such as clinical research organizations, contract manufacturers, clinical trial sites, laboratories for testing clinical samples and consultants. Cumulative R&D costs incurred since we refocused our activities on developing cell-based therapeutics (fiscal years 2000 through 2007)2008) were approximately $75$92 million. Over this period, the majority of these cumulative costs were related to: (i) characterization of our proprietary HuCNS-SC cell, (ii) expenditures for toxicology and other preclinical studies, preparation and submission of our Investigational New Drug (IND) application for our Phase I trial for NCLapplications to the FDA,regulatory agencies to conduct clinical trials and obtaining FDA clearance; and (iii) expenditures in connectionregulatory clearance to initiate such trials, all with respect to our HuCNS-SC Phase I clinical trial.cells, (iii) preclinical studies and development of our human liver engrafting cells; and (iv) costs associated with cell processing and process development.
We use and manage our R&D resources, including our employees and facilities, across various projects rather than on aproject-by-project basis for the following reasons. The allocations of time and resources change as the needs and priorities of individual projects and programs change, and many of our researchers are assigned to more than one project at any given time. Furthermore, we are exploring multiple possible uses for each of our proprietary cell types, so much of our R&D effort is complementary to and supportive of each of these projects. Lastly, much of our R&D effort is focused on manufacturing processes, which can result in process improvements useful across cell types. We also use external service providers to assist in the conduct of our clinical trials, to manufacture certain of our product candidates and to provide various other R&D related products and services. Many of these costs and expenses are complementary to and supportive of each of our programs. Because we do not have a development collaborator for any of our product programs, we are currently responsible for all costs incurred with respect to our product candidates.
R&D expense totaled approximately $17,808,000 in 2008, as compared to $19,937,000 in 2007 as compared toand $13,600,000 in 2006 and $8,227,000 in 2005.2006. At December 31, 2007,2008, we had 4943 full-time employees working in research and development and laboratory support services as compared to 49 at December 31, 2007 and 35 at December 31, 20062006.
2008 versus 2007. The decrease in R&D expenses of approximately $2,129,000, or 11%, in 2008 as compared to 2007 was primarily attributable to a decrease in external services of approximately $2,833,000; these external services were mainly related to manufacturing and 33 at December 31, 2005.testing of our cells and to clinical trial expenses. The decrease in clinical trial expenses was due mainly to the completion of enrollment and dosing of our six-patient Phase I clinical trial in January 2008. The decrease in R&D expenses was also attributable to a decrease in business travel expenses of approximately $197,000. These decreased R&D expenses were partially offset by an increase in other operating expenses primarily attributable to (i) an increase in share based compensation expense of $263,000, and (ii) an increase in other operating expenses of approximately $638,000, primarily attributable to supplies.
2007 versus 2006. The increase in R&D expenses of approximately $6,337,000, or 47%, fromin 2007 as compared to 2006 to 2007 was primarily attributable to the continued expansion of our operations in cell processing and clinical development, including an increase indevelopment. A portion of our cell processing and clinical operations are performed by external service providers, so external services and clinical studytrial costs ofwere approximately $3,954,000 andof the increase in R&D expenses. The increase in R&D expenses was also due to an increase in personnel costs of approximately $1,442,000, of which approximately $206,000 was attributable to stock-based compensation expense. The remainder of the increase in R&D expenses in 2007 was due to increases in supplies, rent, and other operating expenses.
36
General and Administrative Expenses
General and administrative (G&A) expenses totaled approximately $8,296,000 in 2008, compared with $7,927,000 in 2007 compared withand $7,154,000 in 2006 and $5,540,000 in 2005.2006.
2008 versus 2007. The increase in G&A expenses of approximately $369,000, or 5%, in 2008 as compared to 2007 was primarily attributable to an increase in share-based compensation expense of $431,000. In addition, operating expenses for our vacant pilot manufacturing facility in Rhode Island increased by approximately $524,000 due to the loss of tenant income to offset operating expenses. These increased expenses were partially offset by a decrease in external fees of $399,000, including legal and recruiting fees, and a decrease in other operating expenses of approximately $187,000.
38
2007 versus 2006. The increase in G&A expenses of approximately $773,000, or 11%, fromin 2007 as compared to 2006 to 2007 was primarily attributable to an increase in external services of approximately $763,000, driven by an increase in legal fees related to patent prosecutions and litigation, and an increase in personnel costs of approximately $425,000, of which approximately $211,000 was attributable to an increase in stock-based compensation expense. These increases were partially offset by a decrease in other G&A expenses.
Wind-down Expenses
In 1999, in connection with exiting our former research facility in Rhode Island, we created a reserve for the estimated lease payments and operating expenses related to it. The reserve has been re-evaluated and adjusted based on assumptions relevant to real estate market conditions and the estimated time until we could either fully sublease, assign or sell our remaining interests in the property. The reserve was approximately $6,145,000$5,513,000 at December 31, 20072008 and $6,750,000$6,143,000 at December 31, 2006.2007. Payments net of subtenant income were recorded against this reserve of $1,293,000 in 2008, $1,420,000 in 2007, and $1,295,000 in 2006, and $1,079,000 in 2005.2006. We re-evaluated the estimate and adjusted the reserve by recording in aggregate, additional wind-down expenses of $866,000 in 2008, $783,000 in 2007, and $709,000 in 2006, and $2,827,000 in 2005.2006. Expenses for this facility will fluctuate based on changes in tenant occupancy rates and other operating expenses related to the lease. Even though it is our intent to sublease, assign, sell, or otherwise divest ourselves of our interests in the facility at the earliest possible time, we cannot determine with certainty a fixed date by which such events will occur. In light of this uncertainty, based on estimates, we will periodically re-evaluate and adjust the reserve, as necessary. See Note 78 “Wind-down and exit costs,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information.
Other Income (Expense)
Other expense totaled approximately $2,349,000 in 2008, compared with other income totaledof approximately $3,568,000 in 2007 compared withand $2,422,000 in 2006 and $4,650,000 in 2005.2006.
37
| Change in | Change in | Change in | Change in | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2008 | 2007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Versus 2006 | Versus 2005 | Versus 2007 | Versus 2006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | $ | % | $ | % | 2008 | 2007 | 2006 | $ | % | $ | % | |||||||||||||||||||||||||||||||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| License and settlement agreement | $ | 550,467 | $ | 103,359 | $ | 3,735,556 | $ | 447,108 | 433 | % | $ | (3,632,197 | ) | (97 | )% | |||||||||||||||||||||||||||||||||||||||||
| License and settlement agreement, net | $ | — | $ | 550,467 | $ | 103,359 | $ | (550,467 | ) | (100 | )% | $ | 447,108 | 433 | % | |||||||||||||||||||||||||||||||||||||||||
| Realized gain on sale of marketable securities | — | 715,584 | — | (715,584 | ) | (100 | )% | 715,584 | *% | |||||||||||||||||||||||||||||||||||||||||||||||
| Other than temporary impairment of marketable securities | (2,082,894 | ) | — | — | (2,082,894 | ) | *% | — | — | |||||||||||||||||||||||||||||||||||||||||||||||
| Change in fair value of warrant liability | (937,241 | ) | — | — | (937,241 | ) | *% | — | — | |||||||||||||||||||||||||||||||||||||||||||||||
| Interest income | 2,459,820 | 2,479,740 | 1,122,963 | (19,920 | ) | (1 | )% | 1,356,777 | 121 | % | 803,095 | 2,459,820 | 2,479,740 | (1,656,725 | ) | (67 | )% | (19,920 | ) | (1 | )% | |||||||||||||||||||||||||||||||||||
| Interest expense | (123,606 | ) | (143,001 | ) | (171,909 | ) | 19,395 | (14 | )% | 28,908 | (17 | )% | (109,762 | ) | (123,606 | ) | (143,001 | ) | 13,844 | (11 | )% | 19,395 | (14 | )% | ||||||||||||||||||||||||||||||||
| Gain on sale of investment | 715,584 | — | — | 715,584 | *% | — | *% | |||||||||||||||||||||||||||||||||||||||||||||||||
| Other expense, net | (33,899 | ) | (17,644 | ) | (36,892 | ) | (16,255 | ) | 92 | % | 19,248 | (52 | )% | (21,943 | ) | (33,899 | ) | (17,644 | ) | 11,956 | (35 | )% | (16,255 | ) | 92 | % | ||||||||||||||||||||||||||||||
| Total other income, net | $ | 3,568,366 | $ | 2,422,454 | $ | 4,649,718 | $ | 1,145,912 | 47 | % | $ | (2,227,264 | ) | (48 | )% | |||||||||||||||||||||||||||||||||||||||||
| Total other income (expense), net | $ | (2,348,745 | ) | $ | 3,568,366 | $ | 2,422,454 | $ | (5,917,111 | ) | (166 | )% | $ | 1,145,912 | 47 | % | ||||||||||||||||||||||||||||||||||||||||
| * | Calculation |
License and Settlement Agreement
In July 2005, we entered into an agreement with ReNeuron Limited, a wholly owned subsidiary of ReNeuron Group plc, a listed UK corporation (collectively referred to as “ReNeuron”). As part of the agreement, we granted ReNeuron a license that allows ReNeuron to exploit their “c-mycER” conditionally immortalized adult human neural stem cell technology for therapy and other purposes. We received a 7.5% fully-diluted equity interest inshares of ReNeuron subject to certain anti-dilution provisions, andcommon stock, as well as a cross-license to the exclusive use of ReNeuron’s technology for certain diseases and conditions, including lysosomal storage diseases, spinal cord injury, cerebral palsy, and multiple sclerosis. The agreement also provides
39
for full settlement of any potential claims that either we or ReNeuron might have had against the other in connection with any putative infringement of certain of each party’s patent rights prior to the effective date of the agreement. In July and August 2005 we received approximately 8,836,000 ordinary shares of ReNeuron common stock (net of approximately 104,000 shares that were transferred to NeuroSpheres), and subsequently, in 2006 and 2007, as a result of certain anti-dilution provisions in the agreement, we received approximately 1,261,000 more shares, net of approximately 18,000 shares that were transferred to NeuroSpheres. In February 2007, we sold 5,275,000 shares for net proceeds of approximately $3,077,000. See Note 15 “Subsequent Events” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information.
Other income from the license and settlement agreement totaled approximately $0 in 2008, $550,000 in 2007, and $103,000 in 2006, and $3,736,000 in 2005, which was the fair value of the ReNeuron shares we received under such agreement, net of legal fees and the value of the shares that were transferred to NeuroSpheres Ltd., an Alberta corporation from which we have licensed some of the patent rights that are the subject of the agreement with ReNeuron. See Note 2 “Financial Instruments — ReNeuron License Agreement” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information regarding this transaction.
38
Gain on Sale of Marketable Equity Securities
The gain on sale of marketable equity securities of approximately $716,000 in 2007 was attributable to sales of ReNeuron shares. See Note 2 “Financial Instruments,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information on this transaction.
Other than temporary impairment of marketable securities
As of December 31, 2008, we determined that our investment in ReNeuron shares (marketable equity securities) was impaired and that such impairment was other than temporary. We considered various criteria, including the duration of the impairment and our intent to liquidate all or part of this investment within a reasonably short period of time. For the year ended December 31, 2008, we recorded a loss of $2,082,894, which is the difference between the investment’s carrying value and its quoted market price at that date. No other than temporary impairment was recognized during the years ended December 31, 2007 and 2006. See Note 2 “Financial Instruments,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information on this transaction.
Interest Income
Interest income totaled approximately $803,000 in 2008, $2,460,000 in 2007, and $2,480,000 in 2006. The decrease in interest income in 2008 as compared to 2007 was primarily attributable to lower average yields and a lower average bank balance in 2008. Interest income in 2007 was relatively flat compared to 2006, as a result of lower average bank balances offset by higher average yields.
Interest Expense
Interest expense was approximately $110,000 in 2008, $124,000 in 2007, and $143,000 in 2006. The decreases in 2008 as compared to 2007 and in 2007 as compared to 2006 were attributable to lower outstanding debt and capital lease balances. See Note 9 “Commitment and Contingencies,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information.
Other Expense, net
Other expense, net for 2008, 2007, wereand 2006 of approximately $22,000, $34,000, which included approximately $34,000 forand $18,000 respectively, primarily relate to the payment of state franchise taxes paid, partially offset by a gain of approximately $1,500 from the disposal of old equipment. Other expense, net for 2006 were approximately $18,000, which included approximately $20,000 for state franchise taxes paid, partially offset by a gain of approximately $2,000 from the disposal of old equipment. Other expense, net for 2005 were approximately $37,000, which included approximately $36,000 for state franchise taxes and approximately $1,000 from a write-off of obsolete equipment.taxes.
40
Liquidity and Capital Resources
Since our inception, we have financed our operations through the sale of common and preferred stock, the issuance of long-term debt and capitalized lease obligations, revenue from collaborative agreements, research grants, license fees, and interest income.
| Change in | Change in | Change in | Change in | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2008 | 2007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Versus 2006 | Versus 2005 | Versus 2007 | Versus 2006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2007 | 2006 | 2005 | $ | % | $ | % | 2008 | 2007 | 2006 | $ | % | $ | % | |||||||||||||||||||||||||||||||||||||||||||
| At December 31: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and highly liquid investments(1) | $ | 37,645,085 | $ | 51,795,529 | $ | 34,540,908 | $ | (14,150,444 | ) | (27 | )% | $ | 17,254,621 | 50 | % | $ | 34,037,775 | $ | 37,645,085 | $ | 51,795,529 | $ | (3,607,310 | ) | (10 | )% | $ | (14,150,444 | ) | (27 | )% | |||||||||||||||||||||||||
| Year ended December 31: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net cash used in operating activities | $ | (20,856,746 | ) | $ | (16,104,120 | ) | $ | (11,870,568 | ) | $ | (4,752,626 | ) | 29 | % | $ | (4,233,552 | ) | 36 | % | $ | (22,740,421 | ) | $ | (20,856,746 | ) | $ | (16,104,120 | ) | $ | 1,883,475 | 9 | % | $ | 4,752,626 | 29 | % | ||||||||||||||||||||
| Net cash used in investing activities | (27,155,656 | ) | (1,297,124 | ) | (847,505 | ) | (25,858,532 | ) | 1994 | % | (449,619 | ) | 53 | % | ||||||||||||||||||||||||||||||||||||||||||
| Net cash provided by (used in) investing activities | $ | 24,223,629 | $ | (27,155,656 | ) | $ | (1,297,124 | ) | $ | 51,379,285 | 189 | % | $ | 25,858,532 | 1994 | % | ||||||||||||||||||||||||||||||||||||||||
| Net cash provided by financing activities | 5,976,042 | 34,655,865 | 6,199,449 | (28,679,823 | ) | (83 | )% | 28,456,416 | 459 | % | $ | 18,800,609 | $ | 5,976,042 | $ | 34,655,865 | $ | 12,824,567 | 215 | % | $ | (28,679,823 | ) | (83 | )% | |||||||||||||||||||||||||||||||
| (1) | Cash and highly liquid investments include unrestricted cash, cash equivalents, and short-term and long-term marketable debt securities. Marketable equity securities, which are comprised of 4,821,924 ordinary shares of ReNeuron, are excluded from the amounts above. See Note 2, “Financial Instruments,” in the Notes to the Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information. |
Total cash and highly liquid investments were approximately $34,038,000 at December 31, 2008, compared with approximately $37,645,000 at December 31, 2007, compared with approximatelyand $51,796,000 at December 31, 2006, and $34,541,000 at December 31, 2005.
Net Cash Used in Operating Activities
39
Net Cash Used in Investing Activities
The increase from 2006of approximately $51,379,000 for net cash provided by investing activities in 2008 as compared to 2007 was primarily attributable to the maturity of marketable debt securities held to maturity in 2008. In 2008, we received net proceeds of $23,859,000 from the maturity of marketable debt securities, while in 2007, we invested approximately $27,862,000 in net purchases of marketable debt securities. In addition, in December 2008, we made a secured loan of £200,000 (approximately $298,000) to SCS in connection with a potential acquisition transaction. The loan accrues interest at 8% per annum and is repayable on June 23, 2009 if the proposed Acquisition does not close beforehand. The increase of approximately $25,859,000 for net cash used in investing activities in 2007 as compared to 2006 was almost entirely due to the redeployment of cash held in money market funds (classified as cash equivalents) to marketable debt securities (classified as marketable securities). In February 2007, we sold 5,275,000 ordinary shares of ReNeuron for net proceeds of approximately $3,077,000.$3,075,000. In addition, cash used in
41
investing activities in 2007 includesincluded a secured loan of $1,000,000 made to PCT in December 2007. See Note 2, “Financial Instruments,” in the Notes to the Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information on our investing activities. The increase from 2005 to 2006 of approximately $450,000 for net cash used in investing activities was primarily attributable to an increase in capital expenditures.
Net Cash Provided by Financing Activities
The decrease from 2006 to 2007 of approximately $28,680,000 and the increase in 2006 from 2005 of approximately $28,456,000 for net cash provided by financing activities of approximately $12,825,000 in 2008 as compared to 2007 was primarily attributable to the sale onin November 2008 of 13,793,104 units to institutional investors at a price of $1.45 per unit. Each unit consisted of one share of our common stock and a warrant to purchase 0.75 shares of our common stock at an exercise price of $2.30 per share. We received approximately $18,637,000, net of offering expenses and placement agency fees. The decrease of approximately $28,680,000 in 2007 as compared to 2006 was primarily attributable to the sale in April 6, 2006 of 11,750,820 shares of our common stock to a limited number of institutional investors at a price of $3.05 per share. We received total proceeds, net of offering expenses and placement agency fees, of approximately $33,422,000.
Listed below are key financing transactions entered into by us in the last three years:
| • | In November 2008, we sold 13,793,104 units to institutional investors at a price of $1.45 per unit, for gross proceeds of $20,000,000. The units, each of which consisted of one share of common stock and a warrant to purchase 0.75 shares of common stock at an exercise price of $2.30 per share, were offered as a registered direct offering under an effective shelf registration statement previously filed with and declared effective by the Securities and Exchange Commission. We received total proceeds net of offering expenses and placement agency fees of approximately $18,637,000. | |
| • | In April 2007, a warrant issued as part of | |
| • | ||
| • | ||
| • | In March 2006, a warrant issued as part of | |
In the first quarter of 2009, we sold in aggregate, 3,325,000 shares of our common stock pursuant to the sales agreement we entered into with Cantor, at an average price per share of $2.10 for gross proceeds of approximately $6,999,000. Cantor is paid compensation equal to 5.0% of the gross proceeds pursuant to the terms of the agreement.
We have incurred significant operating losses and negative cash flows since inception. We have not achieved profitability and may not be able to realize sufficient revenue to achieve or sustain profitability in the future. We do not expect to be profitable in the next several years, but rather expect to incur additional operating losses. We have limited liquidity and capital resources and must obtain significant additional capital resources in order to sustain our product development efforts, for acquisition of technologies and intellectual property rights, for preclinical and clinical testing of our anticipated products, pursuit of regulatory approvals, acquisition of capital equipment, laboratory and office facilities, establishment of production capabilities, for general and administrative expenses and other working capital requirements. We rely on cash balances and proceeds from equity and debt offerings,
42
proceeds from the transfer or sale of our intellectual property rights, equipment, facilities or investments, and government grants and funding from collaborative arrangements, if obtainable, to fund our operations.
40
We intend to pursue opportunities to obtain additional financing in the future through equity and debt financings, grants and collaborative research arrangements. We haveOn June 25, 2008 we filed with the SEC a universal shelf registration statement, declared effective July 18, 2008, which aspermits us to issue up to $100 million worth of December 31, 2007, coveredregistered debt and equity securities. Under this effective shelf registration, we have the flexibility to issue registered securities, from time to time, in one or more separate offerings or other transactions with the size, price and terms to be determined at the time of issuance. Registered securities issued using this shelf may be used to raise additional capital to fund our working capital and other corporate needs, for future acquisitions of assets, programs or businesses, and for other corporate purposes. As of March 10, 2009, we had approximately $71 million under our universal shelf registration statement available for issuing debt or equity securities; approximately $24 million of this $71 million has been reserved for the potential exercise of the warrants issued in connection with our November 2008 financing. In July 2008, we deregistered the remaining unissued shares (approximately $59 million worth of common stock) available under the shelf registration statement we had filed in October 2005. The 2005 shelf permitted the issuance of up to $100 million of registered shares of our common stock up to a value of approximately $59 million that could be available for financings. On December 29, 2006,stock. Also in July 2008, we filed a Prospectus Supplement announcing the entry of aamended our sales agreement with Cantor Fitzgerald & Co.to allow for sales under which up to 10,000,000 shares may be sold from time to time under theour universal shelf registration statement. rather than the 2005 shelf registration.
The source, timing and availability of any future financing will depend principally upon market conditions, interest rates and, more specifically, on our progress in our exploratory, preclinical and future clinical development programs. Funding may not be available when needed — at all, or on terms acceptable to us. Lack of necessary funds may require us, among other things, to delay, scale back or eliminate some or all of our research and product development programs, planned clinical trials,and/or our capital expenditures or to license our potential products or technologies to third parties. In addition, the decline in economic activity, together with the deterioration of the credit and capital markets, could have an adverse impact on potential sources of future financing.
Commitments
See Note 8,9, “Commitments and Contingencies” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information.
Off-Balance Sheet Arrangements
We have certain contractual arrangements that create potential risk for us and are not recognized in our Consolidated Balance Sheets. Discussed below are those off-balance sheet arrangements that have or are reasonably likely to have a material current or future effect on our financial condition, changes in financial condition, revenue or expenses, results of operations, liquidity, capital expenditures or capital resources.
Operating Leases
We lease various real properties under operating leases that generally require us to pay taxes, insurance, maintenance, and minimum lease payments. Some of our leases have options to renew.
We entered into and amended a lease agreement for an approximately 68,000 square foot facility located at the Stanford Research Park in Palo Alto, California. At December 31, 2007,2008, we had a space-sharing agreement covering approximately 10,451 square feet of this facility. We receive base payments plus a proportionate share of the operating expenses based on square footage over the term of the space-sharing agreement. For the year 2008,2009, we expect to receive, in aggregate, approximately $376,000$606,000 as part of the space-sharing agreement. As a result of the above transactions, our estimated net cash outlay for the rent and operating expenses of this facility will be approximately $1,921,000$3,244,000 for 2008.2009.
We continue to have outstanding obligations in regard to our former facilities in Lincoln, Rhode Island. In 1997, we had entered into a fifteen-year lease for a scientific and administrative facility (the SAF) in a sale and leaseback arrangement. The lease includes escalating rent payments. For the year 2008,2009, we expect to pay approximately $1,172,000 in operating lease payments and estimated operating expenses of approximately $550,000,
43
$625,000, before receipt ofsub-tenant income. For the year 2008,2009, we expect to receive, in aggregate, approximately $282,000$212,000 insub-tenant rent. As a result of the above transactions, our estimated cash outlay net ofsub-tenant rent for the SAF will be approximately $1,440,000$1,585,000 for 2008.2009.
With the exception of leases discussed above, we have not entered into any off balance sheet financial arrangements and have not established any special purpose entities. We have not guaranteed any debts or commitments of other entities or entered into any options on non-financial assets.
See Note 8,9, “Commitments and Contingencies,” in the Notes to Consolidated Financial Statements of Part II, Item 8 of thisForm 10-K for further information.
41
Indemnification Agreement
In July 2008, we amended our 1997 and 2000 license agreements with NeuroSpheres. NeuroSpheres is the holder of certain patents exclusively licensed by us, including the six patents that are the basis of our patent infringement suits against Neuralstem. As part of the amendment, we agreed to pay all reasonable litigation costs, expenses and attorney’s fees incurred by NeuroSpheres in the declaratory judgment suit between us and Neuralstem. In return, we are entitled to off-set all litigation costs incurred in that suit against amounts that would otherwise be owed under the license agreements, such as annual maintenance fees, milestones and royalty payments. At this time, we cannot estimate the likely total costs of our pending litigation with Neuralstem, given the unpredictable nature of such proceedings, or the total amount we may ultimately owe under the NeuroSpheres license agreements. However, the ability to apply the offsets will run for the entire term of each license agreement. For these reasons, we have chosen to approximate the potential value of the offset receivable by assuming that all litigation charges actually incurred in the declaratory judgment action as of December 31, 2008, will ultimately be offset against royalties owed. Management will reevaluate this assumption on a quarterly basis based on actual costs and other relevant factors.
Contractual Obligations
In the table below, we set forth our legally binding and enforceable contractual cash obligations:
| Payable in | Payable in | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | 2013 | Total | 2014 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Obligations | Payable in | Payable in | Payable in | Payable in | Payable in | and | Obligations | Payable in | Payable in | Payable in | Payable in | Payable in | and | |||||||||||||||||||||||||||||||||||||||||||
| at 12/31/07 | 2008 | 2009 | 2010 | 2011 | 2012 | Beyond | at 12/31/08 | 2009 | 2010 | 2011 | 2012 | 2013 | Beyond | |||||||||||||||||||||||||||||||||||||||||||
| Operating lease payments(1) | $ | 11,849,336 | $ | 3,469,017 | $ | 3,536,843 | $ | 1,767,304 | $ | 1,171,875 | $ | 1,171,875 | $ | 732,422 | $ | 8,380,319 | $ | 3,536,843 | $ | 1,767,304 | $ | 1,171,875 | $ | 1,171,875 | $ | 732,422 | $ | — | ||||||||||||||||||||||||||||
| Capital lease (equipment) | 46,347 | 19,862 | 19,862 | 6,623 | — | — | — | 26,483 | 19,862 | 6,621 | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Bonds Payable (principal & interest)(2) | 1,589,093 | 244,531 | 244,572 | 242,559 | 242,321 | 240,666 | 374,444 | 1,344,563 | 244,572 | 242,559 | 242,321 | 240,666 | 237,593 | 136,852 | ||||||||||||||||||||||||||||||||||||||||||
| Total contractual cash obligations | $ | 13,484,776 | $ | 3,733,410 | $ | 3,801,277 | $ | 2,016,486 | $ | 1,414,196 | $ | 1,412,541 | $ | 1,106,866 | $ | 9,751,365 | $ | 3,801,277 | $ | 2,016,484 | $ | 1,414,196 | $ | 1,412,541 | $ | 970,015 | $ | 136,852 | ||||||||||||||||||||||||||||
| (1) | Operating lease payments exclude space-sharing andsub-lease income. See “Off-Balance Sheet Arrangements — Operating Leases” above for further information. | |
| (2) | See Note |
Under license agreements with NeuroSpheres, Ltd., we obtained an exclusive patent license covering all uses of certain neural stem cell technology. We made up-front payments to NeuroSpheres of 65,000 shares of our common stock and $50,000, and will make additional cash payments as stated milestones are achieved. Effective in 2004, we began making annual $50,000 payments, creditable against certain royalties.
We do not have any material unconditional purchase obligations or commercial commitments related to capital expenditures, clinical development, clinical manufacturing, or other external services contracts at December 31, 2007.2008.
44
Recent Accounting Pronouncements
In September 2006,February 2008, the FASB issued SFASFASB Staff Position (FSP)No. FAS 157-2,Effective Date of FASB Statement No. 157(FSP 157-2).FSP 157-2 delays the effective date of SFAS 157 for nonfinancial assets and nonfinancial liabilities, except for certain items that are recognized or disclosed at fair value in the financial statements on a recurring basis (at least annually). We are currently evaluating the impact of SFAS 157 on our consolidated financial statements for items within the scope ofFSP 157-2, which will become effective beginning with our first quarter of 2009.
In October 2008, the FASB issued FSPNo. FAS 157-3,Determining the Fair Value Measurementsof a Financial Asset When the Market for That Asset Is Not Active(FSP 157-3).FSP 157-3 clarifies the application of SFAS 157, in a market that is not active and provides an example to illustrate key considerations in determining the fair value of a financial asset when the market for that financial asset is not active. This FSP shall be effective upon issuance, including prior periods for which financial statements have not been issued. Revisions resulting from a change in the valuation technique or its application shall be accounted for as a change in accounting estimate. Adoption ofFSP 157-3 did not have a material impact on our consolidated financial statement.
In April 2008, the FASB issued FSPNo. 142-3,Determination of the Useful Life of Intangible Assets(FSP 142-3).FSP 142-3 amends the factors that should be considered in developing renewal or extension assumptions used to determine the useful life of a recognized intangible asset under FASB Statement No. 142,Goodwill and Other Intangible Assets(SFAS 157)142).FSP 142-3 amends paragraph 11(d) of SFAS 157 defines fair value, establishes a framework and gives guidance regarding142 to require an entity to use its own assumptions about renewal or extension of an arrangement, adjusted for the methods used for measuring fair value, and expands disclosures about fair value measurements.entity-specific factors in paragraph 11 of SFAS 157142, even when there is likely to be substantial cost or material modifications.FSP 142-3 is effective for financial statements issued for fiscal years beginning after NovemberDecember 15, 2007,2008, and interim periods within those fiscal years.years, with early adoption prohibited. We are currently assessingdo not expect that the impact that SFAS 157 mayadoption ofFSP 142-3 on January 1, 2009, will have a material effect on our consolidated financial condition and results of operations and financial position.operations.
In FebruaryDecember 2007, the FASB issued SFAS No. 159,141R,The Fair Value Option for Financial Assets and Financial Liabilities — Including an amendment of FASB Statement No. 115Business Combinations(SFAS 159)141R). SFAS 159141R provides companies with principles and requirements on how an acquirer recognizes and measures in its financial statements the identifiable assets acquired, liabilities assumed, and any non controlling interest in the acquiree as well as the recognition and measurement of goodwill acquired in a business combination. SFAS 141R also requires certain disclosures to enable users of the financial statements to evaluate the nature and financial effects of the business combination. Acquisition costs associated with the business combination will generally be expensed as incurred. SFAS 141R is expected to expand the use of fair value accounting but does not affect existing standards which require certain assets or liabilities to be carried at fair value. The objectiveeffective for business combinations occurring in fiscal years beginning after December 15, 2008. Early adoption of SFAS 159141R is to improve financial reporting by providing companies with the opportunity to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without havingnot permitted. We will be required to apply complex hedge accounting provisions. Underthe guidance in SFAS 159,141R to any future business combinations effective January 1, 2009.
In June 2008, the FASB issued EITF IssueNo. 07-05,Determining Whether an Instrument (or Embedded Feature) Is Indexed to an Entity’s Own Stock. EITF IssueNo. 07-05 clarifies the determination of whether an instrument (or an embedded feature) is indexed to an entity’s own stock, which would qualify as a company may choose, at specified election dates, to measure eligible items at fair valuescope exception under SFAS No. 133,Accounting for Derivative Instruments and report unrealized gains and losses on items for which the fair value option has been elected in earnings at each subsequent reporting date. SFAS 159Hedging Activities. EITF IssueNo. 07-05 is effective for financial statements issued for fiscal years beginning after NovemberDecember 15, 2007, and interim periods within those fiscal years.2008. Early adoption for an existing instrument is not permitted. We are currently assessingdo not expect the adoption of EITF IssueNo. 07-05 to have a material impact that SFAS 159 may have on our results of operations andconsolidated financial position.statements.
42
| Item 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Interest Rate and Credit RiskRisks
Our interest-bearing assets, or interest-bearing portfolio, consists of cash, cash equivalents, restricted cash, and marketable debt securities. The balance of our interest-bearing portfolio, was approximately $34,031,000, or 85%, of total assets at December 31, 2008 and $38,414,000, or 79%, of total assets at December 31, 2007 and $52,567,000, or 79%, of total assets at December 31, 2006.2007. Interest income earned on these assets was approximately $803,000 in 2008 and $2,460,000 in 2007 and $2,478,000 in 2006.2007. Our interest income is sensitive to changes in the general level of interest rates, primarily U.S. interest rates. OurAt December 31, 2008, our debt securities arewere primarily composed of money market accounts comprised of commercial paper, corporate debtUS Treasuries and asset-backed securities.repurchase
45
agreements that are backed by US Treasuries. Generally, corporate obligations must have senior credit ratings of A2/A or the equivalent. See Note 1, “Summary of Significant Accounting Policies — Financial Instruments” and Note 2 “Financial Instruments” section in the Notes to Consolidated Financial Statements in Part II, Item 8 of thisForm 10-K for further information.
Our long-term debt is comprised of industrial revenue bonds issued by the State of Rhode Island to finance the construction of our pilot manufacturing facility in Rhode Island. See Note 8,9, “Commitments and Contingencies,” section in the Notes to Consolidated Financial Statements in Part II, Item 8 of thisForm 10-K for further information.
Equity Security and Foreign Exchange Risks
In July 2005, we entered into a license and settlementan agreement with ReNeuron Limited, a wholly owned subsidiary of ReNeuron Group plc, a publicly listed UK corporation (collectively referred to as “ReNeuron”). As part of the agreement, we granted ReNeuron a license that allows ReNeuron to exploit their “c-mycER” conditionally immortalized adult human neural stem cell technology for therapy and other purposes. In return for the license, weWe received a 7.5% fully-diluted equity interest inshares of ReNeuron subject to certain anti-dilution provisions, andcommon stock, as well as a cross-license to the exclusive use of ReNeuron’s technology for certain diseases and conditions, including lysosomal storage diseases, spinal cord injury, cerebral palsy, and multiple sclerosis. The agreement also provides for full settlement of any potential claims that either we or ReNeuron might have had against the other in connection with any putative infringement of certain of each party’s patent rights prior to the effective date of the agreement. In February 2007,July and August 2005 we sold 5,275,000received approximately 8,836,000 ordinary shares of ReNeuron for net proceedscommon stock (net of approximately $3,075,000. We recorded approximately $716,000 as realized gain for this transaction. In February 2007, ReNeuron issued additional104,000 shares of common stockthat were transferred to NeuroSpheres), and subsequently, as a consequenceresult of certain anti-dilution provisions in the agreement. We were entitled toagreement, we received approximately 822,0001,261,000 more shares, net of approximately 12,00018,000 shares that were transferred to NeuroSpheres. We recordedIn February 2007, we sold 5,275,000 shares for net proceeds of approximately $551,000$3,077,000. In the first quarter of 2009, we sold in aggregate, approximately 2,900,000 more shares and received net proceeds of approximately $512,000. As of March 10, 2009, we held approximately 1,922,000 shares of ReNeuron as other income for the fair value of the additional shares received.marketable equity securities.
Changes in market value as a result of changes in market price per share or the exchange rate between the U.S. dollar and the British pound are accounted for under “other comprehensive income (loss)” if deemed temporary and are not recorded as “other income or loss” until the shares are disposed of and a gain or loss realized. The unrealized loss asrealized or an impairment is determined to be other than temporary. After considering various criteria, including, the duration of the impairment and our intent to liquidate all or part of this investment within a reasonably short period of time, we determined that the impairment of our investment in ReNeuron was other than temporary. For the year ended December 31, 2007 was approximately $249,000.2008, we recorded, on our “Consolidated Statements of Operations” under “Other Income (expense), a loss of $2,082,894, which is the difference between the investment’s carrying value and its quoted market price at that date.
| Share | Share | |||||||||||||||||||||||||||||||||||||||||||||
| No. of | price at | Exchange | Market | No. of | Price at | Exchange | Market | |||||||||||||||||||||||||||||||||||||||
| Shares | December 31, | Rate at | Value | Expected | Shares | December 31, | Rate at | Value | Expected | |||||||||||||||||||||||||||||||||||||
| at | 2007 | December 31, | in USD at | Future | at | 2008 | December 31, | in USD at | Future | |||||||||||||||||||||||||||||||||||||
| Associated | December 31, | in | 2007 | December 31, | Cash | Associated | December 31, | in | 2008 | December 31, | Cash | |||||||||||||||||||||||||||||||||||
Company/Stock Symbol | Exchange | Risks | 2007 | GBP(£) | 1 GBP = USD | 2007 | Flows | Exchange | Risks | 2008 | GBP(£) | 1 GBP = USD | 2008 | Flows | ||||||||||||||||||||||||||||||||
| ReNeuron Group plc/RENE | AIM (AIM is the London Stock Exchange’s Alternative Investment Market) | — Lower share price — Foreign currency translation — Liquidity — Bankruptcy | 4,821,924 | 0.205 | 1.9843 | $ | 1,961,469 | (1) | AIM (AIM is the London Stock Exchange’s Alternative Investment Market) | — Lower share price — Foreign currency translation — Liquidity — Bankruptcy | 4,821,924 | 0.0265 | 1.4619 | $ | 186,803 | (1 | ) | |||||||||||||||||||||||||||||
| (1) | It is our intention to liquidate this investment when we can do so at prices acceptable to us. Although we are not legally restricted from selling the stock, the share price is subject to change and the volume traded has often been very small since the stock was listed on the AIM on August 12, 2005. The performance of ReNeuron Group plc stock since its listing does not predict its future value. |
4346
44
| Item 8. | FINANCIAL STATEMENTS AND |
STEMCELLS, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| Page | ||||
4547
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of Directors and Stockholders
StemCells, Inc.
We have audited the accompanying consolidated balance sheets of StemCells, Inc. (a Delaware corporation) and subsidiary (collectively, the “Company”) as of December 31, 20072008 and 2006,2007, and the related consolidated statements of operations, changes in stockholders’ equity, and cash flows for each of the three years in the period ended December 31, 2007.2008. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of StemCells, Inc. and subsidiary as of December 31, 20072008 and 2006,2007, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2007,2008, in conformity with accounting principles generally accepted in the United States of America.
We also have audited, in accordance with standards of the Public Company Accounting Oversight Board (United States), StemCells, Inc.’s and subsidiary’s internal control over financial reporting as of December 31, 2007,2008, based on criteria established inInternal Control — Integrated Frameworkissued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO), and our report dated March 7, 200811, 2009 expressed an unqualified opinion thereon.
/s/ GRANT THORNTON LLP
San Francisco, California
March 7, 200811, 2009
4648
StemCells, Inc.
Consolidated Balance Sheets
| December 31, | ||||||||
| 2007 | 2006 | |||||||
ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 9,759,169 | $ | 51,795,529 | ||||
| Marketable securities, current | 26,696,413 | 4,132,646 | ||||||
| Other receivables | 264,631 | 482,850 | ||||||
| Note receivable | 1,000,000 | — | ||||||
| Prepaid assets | 1,032,482 | 1,119,467 | ||||||
| Total current assets | 38,752,695 | 57,530,492 | ||||||
| Marketable securities, non current | 3,150,971 | 3,133,632 | ||||||
| Property, plant and equipment, net | 3,905,404 | 3,596,150 | ||||||
| Other assets, non-current | 1,710,829 | 1,720,361 | ||||||
| Intangible assets, net | 762,667 | 876,182 | ||||||
| Total assets | $ | 48,282,566 | $ | 66,856,817 | ||||
LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 1,813,595 | $ | 620,765 | ||||
| Accrued expenses and other liabilities | 2,462,252 | 2,053,902 | ||||||
| Accrued wind-down expenses, current | 1,374,632 | 1,252,483 | ||||||
| Deferred revenue, current | 43,909 | 16,826 | ||||||
| Capital lease obligation, current | 17,530 | — | ||||||
| Bonds payable, current | 136,250 | 205,833 | ||||||
| Total current liabilities | 5,848,168 | 4,149,809 | ||||||
| Capital lease obligation, non-current | 25,269 | — | ||||||
| Bonds payable, non-current | 1,009,166 | 1,145,416 | ||||||
| Deposits and other long-term liabilities | 527,804 | 547,392 | ||||||
| Accrued wind-down expenses, non-current | 4,768,859 | 5,497,774 | ||||||
| Deferred rent | 727,535 | 959,732 | ||||||
| Deferred revenue, non-current | 163,865 | 180,691 | ||||||
| Total liabilities | 13,070,666 | 12,480,814 | ||||||
| Commitments and contingencies (Note 8) | ||||||||
| Stockholders’ equity: | ||||||||
| Common stock, $.01 par value; 125,000,000 shares authorized; issued and outstanding 80,681,087 at December 31, 2007 and 78,046,304 at December 31, 2006 | 806,810 | 780,462 | ||||||
| Additional paid-in capital | 264,603,711 | 255,299,508 | ||||||
| Accumulated deficit | (229,914,747 | ) | (204,891,945 | ) | ||||
| Accumulated other comprehensive income (loss) | (283,874 | ) | 3,187,978 | |||||
| Total stockholders’ equity | 35,211,900 | 54,376,003 | ||||||
| Total liabilities and stockholders’ equity | $ | 48,282,566 | $ | 66,856,817 | ||||
47
| Year Ended December 31, | ||||||||||||
| 2007 | 2006 | 2005 | ||||||||||
| Revenue: | ||||||||||||
| Revenue from licensing agreements and grants | $ | 56,722 | $ | 92,850 | $ | 205,914 | ||||||
| Operating Expenses: | ||||||||||||
| Research and development | 19,937,426 | 13,600,433 | 8,226,734 | |||||||||
| General and administrative | 7,927,443 | 7,154,042 | 5,539,845 | |||||||||
| Wind-down expenses | 783,022 | 709,209 | 2,827,403 | |||||||||
| Total operating expenses | 28,647,891 | 21,463,684 | 16,593,982 | |||||||||
| Operating losses | (28,591,169 | ) | (21,370,834 | ) | (16,388,068 | ) | ||||||
| Other Income (expense): | ||||||||||||
| License and settlement agreement, net | 550,467 | 103,359 | 3,735,556 | |||||||||
| Realized gain on sale of marketable securities | 715,584 | — | — | |||||||||
| Interest income | 2,459,820 | 2,479,740 | 1,122,963 | |||||||||
| Interest expense | (123,606 | ) | (143,001 | ) | (171,909 | ) | ||||||
| Other income (expense) | (33,898 | ) | (17,644 | ) | (36,892 | ) | ||||||
| Total other income, net | 3,568,367 | 2,422,454 | 4,649,718 | |||||||||
| Net loss | $ | (25,022,802 | ) | $ | (18,948,380 | ) | $ | (11,738,350 | ) | |||
| Basic and diluted net loss per share | $ | (0.31 | ) | $ | (0.25 | ) | $ | (0.18 | ) | |||
| Shares used to compute basic and diluted loss per share | 79,772,351 | 74,611,196 | 63,643,176 | |||||||||
48
| Accumulated | ||||||||||||||||||||||||||||
| Additional | Other | Total | ||||||||||||||||||||||||||
| Common Stock | Paid-in | Accumulated | Comprehensive | Deferred | Stockholders’ | |||||||||||||||||||||||
| Shares | Amount | Capital | Deficit | Income (Loss) | Compensation | Equity | ||||||||||||||||||||||
Balances, December 31, 2004 | 62,129,407 | $ | 621,294 | $ | 211,419,300 | $ | (174,205,215 | ) | $ | — | $ | (885,832 | ) | $ | 36,949,547 | |||||||||||||
| Comprehensive loss | ||||||||||||||||||||||||||||
| Net loss | — | — | — | (11,738,350 | ) | — | — | (11,738,350 | ) | |||||||||||||||||||
| Change in unrealized loss on securities available-for-sale | — | — | — | — | (254,147 | ) | — | (254,147 | ) | |||||||||||||||||||
| Comprehensive loss | (11,992,497 | ) | ||||||||||||||||||||||||||
| Expenses related to equity financing | — | — | (193,946 | ) | — | — | — | (193,946 | ) | |||||||||||||||||||
| Common stock issued for external services | 2,022 | 20 | 8,310 | — | — | — | 8,330 | |||||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 28,459 | 285 | 110,772 | — | — | — | 111,057 | |||||||||||||||||||||
| Compensation expense from grant of options and stock (fair value) | — | — | 461,675 | — | — | — | 461,675 | |||||||||||||||||||||
| Exercise of employee and consultant stock options | 393,509 | 3,935 | 733,753 | — | — | — | 737,688 | |||||||||||||||||||||
| Exercise of warrants | 2,842,625 | 28,426 | 5,910,680 | — | — | — | 5,939,106 | |||||||||||||||||||||
| Deferred compensation | — | — | (531,208 | ) | — | — | 531,208 | — | ||||||||||||||||||||
| Amortization of deferred compensation | — | — | — | — | — | 354,624 | 354,624 | |||||||||||||||||||||
Balances, December 31, 2005 | 65,396,022 | 653,960 | 217,919,336 | (185,943,565 | ) | (254,147 | ) | — | 32,375,584 | |||||||||||||||||||
| Comprehensive loss | ||||||||||||||||||||||||||||
| Net loss | — | — | — | (18,948,380 | ) | — | — | (18,948,380 | ) | |||||||||||||||||||
| Change in unrealized gain on securities available-for-sale | — | — | — | — | 3,442,125 | — | 3,442,125 | |||||||||||||||||||||
| Comprehensive loss | (15,506,255 | ) | ||||||||||||||||||||||||||
| Issuance of common stock related to equity financing net of issuance cost of $2,418,467 | 11,750,820 | 117,508 | 33,304,026 | — | — | — | 33,421,534 | |||||||||||||||||||||
| Common stock issued for licensing agreements | 3,848 | 38 | 9,962 | — | — | — | 10,000 | |||||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 50,120 | 501 | 121,955 | — | — | — | 122,456 | |||||||||||||||||||||
| Compensation expense from grant of options and stock (fair value) | — | — | 2,409,509 | — | — | — | 2,409,509 | |||||||||||||||||||||
| Exercise of employee and consultant stock options | 319,094 | 3,191 | 545,088 | — | — | — | 548,279 | |||||||||||||||||||||
| Exercise of warrants | 526,400 | 5,264 | 989,632 | — | — | — | 994,896 | |||||||||||||||||||||
Balances, December 31, 2006 | 78,046,304 | 780,462 | 255,299,508 | (204,891,945 | ) | 3,187,978 | — | 54,376,003 | ||||||||||||||||||||
| Comprehensive loss | ||||||||||||||||||||||||||||
| Net loss | — | — | — | (25,022,802 | ) | — | — | (25,022,802 | ) | |||||||||||||||||||
| Change in unrealized loss on securities available-for-sale | — | — | — | — | (3,471,852 | ) | — | (3,471,852 | ) | |||||||||||||||||||
| Comprehensive loss | (28,494,654 | ) | ||||||||||||||||||||||||||
| Issuance of common stock related to equity financing net of issuance cost of $297,465 | 1,807,000 | 18,070 | 4,816,983 | — | — | — | 4,835,053 | |||||||||||||||||||||
| Common stock issued for licensing agreements | 3,865 | 39 | 9,961 | — | — | — | 10,000 | |||||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 73,074 | 731 | 172,429 | — | — | — | 173,160 | |||||||||||||||||||||
| Compensation expense from grant of options and stock (fair value) | — | — | 3,008,315 | — | — | — | 3,008,315 | |||||||||||||||||||||
| Exercise of employee stock options | 175,186 | 1,752 | 208,521 | — | — | — | 210,273 | |||||||||||||||||||||
| Exercise of warrants | 575,658 | 5,756 | 1,087,994 | — | — | — | 1,093,750 | |||||||||||||||||||||
Balances, December 31, 2007 | 80,681,087 | $ | 806,810 | $ | 264,603,711 | $ | (229,914,747 | ) | $ | (283,874 | ) | $ | — | $ | 35,211,900 | |||||||||||||
| December 31, | ||||||||
| 2008 | 2007 | |||||||
ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 30,042,986 | $ | 9,759,169 | ||||
| Marketable securities, current | 4,181,592 | 26,696,413 | ||||||
| Other receivables | 164,204 | 264,631 | ||||||
| Note receivable | 298,032 | 1,000,000 | ||||||
| Prepaid assets | 645,242 | 1,032,482 | ||||||
| Total current assets | 35,332,056 | 38,752,695 | ||||||
| Marketable securities, non current | — | 3,150,971 | ||||||
| Property, plant and equipment, net | 3,173,468 | 3,905,404 | ||||||
| Other assets, non-current | 2,079,278 | 1,710,829 | ||||||
| Intangible assets, net | 645,538 | 762,667 | ||||||
| Total assets | $ | 41,230,340 | $ | 48,282,566 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 1,078,123 | $ | 1,813,595 | ||||
| Accrued expenses and other liabilities | 2,261,245 | 2,462,252 | ||||||
| Accrued wind-down expenses, current | 1,420,378 | 1,374,632 | ||||||
| Deferred revenue, current | 43,909 | 43,909 | ||||||
| Capital lease obligation, current | 18,739 | 17,530 | ||||||
| Deferred rent, current | 346,930 | 290,391 | ||||||
| Bonds payable, current | 149,167 | 136,250 | ||||||
| Total current liabilities | 5,318,491 | 6,138,559 | ||||||
| Capital lease obligation, non-current | 6,529 | 25,269 | ||||||
| Bonds payable, non-current | 860,000 | 1,009,166 | ||||||
| Fair value of warrant liability | 8,439,931 | — | ||||||
| Deposits and other long-term liabilities | 466,211 | 527,804 | ||||||
| Accrued wind-down expenses, non-current | 4,092,939 | 4,768,859 | ||||||
| Deferred rent, non-current | 90,215 | 437,144 | ||||||
| Deferred revenue, non-current | 147,039 | 163,865 | ||||||
| Total liabilities | 19,421,355 | 13,070,666 | ||||||
| Commitments and contingencies (Note 9) | ||||||||
| Stockholders’ equity: | ||||||||
| Common stock, $.01 par value; 250,000,000 shares authorized; issued and outstanding 94,945,603 at December 31, 2008 and 80,681,087 at December 31, 2007 | 949,455 | 806,810 | ||||||
| Additional paid-in capital | 279,868,802 | 264,603,711 | ||||||
| Accumulated deficit | (259,001,524 | ) | (229,914,747 | ) | ||||
| Accumulated other comprehensive loss | (7,748 | ) | (283,874 | ) | ||||
| Total stockholders’ equity | 21,808,985 | 35,211,900 | ||||||
| Total liabilities and stockholders’ equity | $ | 41,230,340 | $ | 48,282,566 | ||||
See Notes to Consolidated Financial Statements.
49
StemCells, Inc.
Consolidated Statements of Operations
| Year Ended December 31, | ||||||||||||
| 2008 | 2007 | 2006 | ||||||||||
| Revenue: | ||||||||||||
| Revenue from licensing agreements and grants | $ | 231,730 | $ | 56,722 | $ | 92,850 | ||||||
| Operating expenses: | ||||||||||||
| Research and development | 17,808,009 | 19,937,426 | 13,600,433 | |||||||||
| General and administrative | 8,295,554 | 7,927,443 | 7,154,042 | |||||||||
| Wind-down expenses | 866,199 | 783,022 | 709,209 | |||||||||
| Total operating expenses | 26,969,762 | 28,647,891 | 21,463,684 | |||||||||
| Operating loss | (26,738,032 | ) | (28,591,169 | ) | (21,370,834 | ) | ||||||
| Other income (expense): | ||||||||||||
| License and settlement agreement, net | — | 550,467 | 103,359 | |||||||||
| Realized gain on sale of marketable securities | — | 715,584 | — | |||||||||
| Other than temporary impairment of marketable securities | (2,082,894 | ) | — | — | ||||||||
| Change in fair value of warrant liability | (937,241 | ) | — | — | ||||||||
| Interest income | 803,095 | 2,459,820 | 2,479,740 | |||||||||
| Interest expense | (109,762 | ) | (123,606 | ) | (143,001 | ) | ||||||
| Other expense, net | (21,943 | ) | (33,898 | ) | (17,644 | ) | ||||||
| Total other income (expense), net | (2,348,745 | ) | 3,568,367 | 2,422,454 | ||||||||
| Net loss | $ | (29,086,777 | ) | $ | (25,022,802 | ) | $ | (18,948,380 | ) | |||
| Basic and diluted net loss per share | $ | (0.35 | ) | $ | (0.31 | ) | $ | (0.25 | ) | |||
| Shares used to compute basic and diluted loss per share | 82,716,455 | 79,772,351 | 74,611,196 | |||||||||
See Notes to Consolidated Financial Statements.
50
StemCells, Inc.
Consolidated Statements of Stockholders’ Equity
| Accumulated | ||||||||||||||||||||||||
| Additional | Other | Total | ||||||||||||||||||||||
| Common Stock | Paid-in | Accumulated | Comprehensive | Stockholders’ | ||||||||||||||||||||
| Shares | Amount | Capital | Deficit | Income (Loss) | Equity | |||||||||||||||||||
Balances, December 31, 2005 | 65,396,022 | $ | 653,960 | $ | 217,919,336 | $ | (185,943,565 | ) | $ | (254,147 | ) | $ | 32,375,584 | |||||||||||
| Comprehensive loss | ||||||||||||||||||||||||
| Net loss | — | — | — | (18,948,380 | ) | — | (18,948,380 | ) | ||||||||||||||||
| Change in unrealized gain on securities available-for-sale | — | — | — | — | 3,442,125 | 3,442,125 | ||||||||||||||||||
| Comprehensive loss | (15,506,255 | ) | ||||||||||||||||||||||
| Issuance of common stock related to equity financing net of issuance cost of $2,418,467 | 11,750,820 | 117,508 | 33,304,026 | — | — | 33,421,534 | ||||||||||||||||||
| Common stock issued for licensing agreements | 3,848 | 38 | 9,962 | — | — | 10,000 | ||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 50,120 | 501 | 121,955 | — | — | 122,456 | ||||||||||||||||||
| Compensation expense from grant of options and stock (fair value) | — | — | 2,409,509 | — | — | 2,409,509 | ||||||||||||||||||
| Exercise of employee and consultant stock options | 319,094 | 3,191 | 545,088 | — | — | 548,279 | ||||||||||||||||||
| Exercise of warrants | 526,400 | 5,264 | 989,632 | — | — | 994,896 | ||||||||||||||||||
Balances, December 31, 2006 | 78,046,304 | 780,462 | 255,299,508 | (204,891,945 | ) | 3,187,978 | 54,376,003 | |||||||||||||||||
| Comprehensive loss | ||||||||||||||||||||||||
| Net loss | — | — | — | (25,022,802 | ) | — | (25,022,802 | ) | ||||||||||||||||
| Change in unrealized loss on securities available-for-sale | — | — | — | — | (3,471,852 | ) | (3,471,852 | ) | ||||||||||||||||
| Comprehensive loss | (28,494,654 | ) | ||||||||||||||||||||||
| Issuance of common stock related to equity financing net of issuance cost of $297,465 | 1,807,000 | 18,070 | 4,816,983 | — | — | 4,835,053 | ||||||||||||||||||
| Common stock issued for licensing agreements | 3,865 | 39 | 9,961 | — | — | 10,000 | ||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 73,074 | 731 | 172,429 | — | — | 173,160 | ||||||||||||||||||
| Compensation expense from grant of options and stock (fair value) | — | — | 3,008,315 | — | — | 3,008,315 | ||||||||||||||||||
| Exercise of employee stock options | 175,186 | 1,752 | 208,521 | — | — | 210,273 | ||||||||||||||||||
| Exercise of warrants | 575,658 | 5,756 | 1,087,994 | — | — | 1,093,750 | ||||||||||||||||||
Balances, December 31, 2007 | 80,681,087 | 806,810 | 264,603,711 | (229,914,747 | ) | (283,874 | ) | 35,211,900 | ||||||||||||||||
| Comprehensive loss | ||||||||||||||||||||||||
| Net loss | — | — | — | (29,086,777 | ) | — | (29,086,777 | ) | ||||||||||||||||
| Change in unrealized loss on securities available-for-sale | — | — | — | — | 276,126 | 276,126 | ||||||||||||||||||
| Comprehensive loss | (28,810,651 | ) | ||||||||||||||||||||||
| Issuance of common stock and warrants, net of issuance cost of $1,432,539 | 13,998,704 | 139,987 | 11,184,188 | — | — | 11,324,175 | ||||||||||||||||||
| Common stock issued for licensing agreements | 6,924 | 69 | 9,931 | — | — | 10,000 | ||||||||||||||||||
| Common stock issued pursuant to employee benefit plan | 144,188 | 1,442 | 189,724 | — | — | 191,166 | ||||||||||||||||||
| Compensation expense from grant of options, restricted stock units and stock (fair value) | — | — | 3,754,871 | — | — | 3,754,871 | ||||||||||||||||||
| Exercise of employee and director stock options | 114,700 | 1,147 | 126,377 | — | — | 127,524 | ||||||||||||||||||
Balances, December 31, 2008 | 94,945,603 | $ | 949,455 | $ | 279,868,802 | $ | (259,001,524 | ) | $ | (7,748 | ) | $ | 21,808,985 | |||||||||||
See Notes to Consolidated Financial Statements.
51
StemCells, Inc.
Consolidated Statements of Cash Flows
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||||
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
Cash flows from operating activities: | ||||||||||||||||||||||||
| Net loss | $ | (25,022,802 | ) | $ | (18,948,380 | ) | $ | (11,738,350 | ) | $ | (29,086,777 | ) | $ | (25,022,802 | ) | $ | (18,948,380 | ) | ||||||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||||||||||||||
| Depreciation and amortization | 1,174,510 | 1,044,688 | 1,082,793 | 1,186,428 | 1,174,510 | 1,044,688 | ||||||||||||||||||
| Amortization of deferred compensation | — | — | 354,624 | |||||||||||||||||||||
| Issue of shares and options in exchange for services | 3,181,475 | 2,531,966 | 581,062 | 3,946,037 | 3,181,475 | 2,531,966 | ||||||||||||||||||
| (Gain) loss on disposal of fixed assets | (1,500 | ) | 1,573 | 1,377 | — | (1,500 | ) | 1,573 | ||||||||||||||||
| Non-cash income from license and settlement agreement, net | — | (550,467 | ) | (103,359 | ) | |||||||||||||||||||
| Gain on sale of marketable securities | (715,584 | ) | — | — | — | (715,584 | ) | — | ||||||||||||||||
| Non-cash income from license and settlement agreement, net | (550,467 | ) | (103,359 | ) | (3,974,941 | ) | ||||||||||||||||||
| Other than temporary impairment of marketable securities | 2,082,894 | — | — | |||||||||||||||||||||
| Change in fair value of warrant liability | 937,241 | — | — | |||||||||||||||||||||
| Changes in operating assets and liabilities: | ||||||||||||||||||||||||
| Accrued interest and other receivables | 218,219 | (280,931 | ) | (20,956 | ) | |||||||||||||||||||
| Other receivables | 100,427 | 218,219 | (280,931 | ) | ||||||||||||||||||||
| Prepaid assets | 86,985 | (732,501 | ) | (177,892 | ) | 387,240 | 86,985 | (732,501 | ) | |||||||||||||||
| Intangible and other assets, net | 19,532 | 56,270 | (47,053 | ) | ||||||||||||||||||||
| Other assets, net | (358,449 | ) | 19,532 | 56,270 | ||||||||||||||||||||
| Accounts payable and accrued expenses | 1,601,180 | 554,245 | 48,135 | (936,479 | ) | 1,601,180 | 554,245 | |||||||||||||||||
| Accrued wind-down expenses | (606,766 | ) | (555,469 | ) | 1,777,697 | (630,174 | ) | (606,766 | ) | (555,469 | ) | |||||||||||||
| Deferred revenue | 10,257 | 197,517 | — | (16,826 | ) | 10,257 | 197,517 | |||||||||||||||||
| Deferred rent | (232,198 | ) | 105,735 | 330,196 | (290,390 | ) | (232,198 | ) | 105,735 | |||||||||||||||
| Deposits and other long-term liabilities | (19,587 | ) | 24,526 | (87,260 | ) | (61,593 | ) | (19,587 | ) | 24,526 | ||||||||||||||
| Net cash used in operating activities | (20,856,746 | ) | (16,104,120 | ) | (11,870,568 | ) | (22,740,421 | ) | (20,856,746 | ) | (16,104,120 | ) | ||||||||||||
Cash flows from investing activities: | ||||||||||||||||||||||||
| Purchase of marketable securities | (27,861,561 | ) | — | — | ||||||||||||||||||||
| Proceeds from sale of marketable securities | 3,074,654 | — | — | |||||||||||||||||||||
| Purchase of marketable debt securities | (4,822,684 | ) | (37,029,744 | ) | — | |||||||||||||||||||
| Sales or maturity of marketable debt securities | 28,681,708 | 9,168,183 | — | |||||||||||||||||||||
| Proceeds from sale of marketable equity securities | — | 3,074,654 | — | |||||||||||||||||||||
| Repayment received under note receivable | 1,000,000 | — | — | |||||||||||||||||||||
| Advance made under note receivable | (1,000,000 | ) | — | — | (298,032 | ) | (1,000,000 | ) | — | |||||||||||||||
| Purchases of property, plant and equipment, net | (1,319,374 | ) | (1,258,749 | ) | (817,505 | ) | ||||||||||||||||||
| Purchase of other assets | (49,375 | ) | (38,375 | ) | (30,000 | ) | ||||||||||||||||||
| Purchases of property, plant and equipment | (312,988 | ) | (1,319,374 | ) | (1,258,749 | ) | ||||||||||||||||||
| Purchase of intangibles and other assets | (24,375 | ) | (49,375 | ) | (38,375 | ) | ||||||||||||||||||
| Net cash used in investing activities | (27,155,656 | ) | (1,297,124 | ) | (847,505 | ) | ||||||||||||||||||
| Net cash provided by (used in) investing activities | 24,223,629 | (27,155,656 | ) | (1,297,124 | ) | |||||||||||||||||||
Cash flows from financing activities: | ||||||||||||||||||||||||
| Proceeds (expense) from issuance of common stock, net | 4,835,053 | 33,421,534 | (193,946 | ) | ||||||||||||||||||||
| Proceeds from issuance of common stock, net | 18,826,865 | 4,835,053 | 33,421,534 | |||||||||||||||||||||
| Proceeds from the exercise of stock options | 210,273 | 548,279 | 737,688 | 127,524 | 210,273 | 548,279 | ||||||||||||||||||
| Proceeds from the exercise of warrants | 1,093,750 | 994,896 | 5,939,106 | — | 1,093,750 | 994,896 | ||||||||||||||||||
| Proceeds (repayments) of capital lease obligations | 42,799 | (54,676 | ) | (39,232 | ) | (17,531 | ) | 42,799 | (54,676 | ) | ||||||||||||||
| Repayments of debt obligations | (205,833 | ) | (254,168 | ) | (244,167 | ) | ||||||||||||||||||
| Repayments of bonds payable | (136,249 | ) | (205,833 | ) | (254,168 | ) | ||||||||||||||||||
| Net cash provided by financing activities | 5,976,042 | 34,655,865 | 6,199,449 | 18,800,609 | 5,976,042 | 34,655,865 | ||||||||||||||||||
| Increase (decrease) in cash and cash equivalents | (42,036,360 | ) | 17,254,621 | (6,518,624 | ) | 20,283,817 | (42,036,360 | ) | 17,254,621 | |||||||||||||||
| Cash and cash equivalents at beginning of year | 51,795,529 | 34,540,908 | 41,059,532 | 9,759,169 | 51,795,529 | 34,540,908 | ||||||||||||||||||
| Cash and cash equivalents at end of the year | $ | 9,759,169 | $ | 51,795,529 | $ | 34,540,908 | $ | 30,042,986 | $ | 9,759,169 | $ | 51,795,529 | ||||||||||||
| Supplemental disclosure of cash flow information: | ||||||||||||||||||||||||
| Interest paid | $ | 123,606 | $ | 143,001 | $ | 171,909 | $ | 109,762 | $ | 123,606 | $ | 143,001 | ||||||||||||
| Supplemental schedule of non-cash investing and financing activities: | ||||||||||||||||||||||||
Stock issued for licensing agreements(1) | $ | 10,000 | $ | 10,000 | — | |||||||||||||||||||
| Stock issued for licensing agreements(1) | $ | 10,000 | $ | 10,000 | $ | 10,000 | ||||||||||||||||||
| (1) | Under terms of a license agreement with the California Institute of Technology (Cal Tech), annual fees of $5,000 were due on each of two patents to which StemCells holds a license from Cal Tech, payable in cash or stock at our choice. We elected to pay the fees in common stock and issued shares of 6,924 in 2008, 3,865 in 2007 and 3,848 in 2006 to Cal Tech. |
See Notes to Consolidated Financial Statements.
5052
StemCells, Inc.
Notes to Consolidated Financial Statements
December 31, 20072008
| Note 1. | Summary of Significant Accounting Policies |
Nature of Business
StemCells, Inc., a Delaware corporation, is a biopharmaceutical company that operates in one segment, the development of novel cell-based therapeutics designed to treat human diseases and disorders.
The accompanying consolidated financial statements have been prepared on the basis that we will continue as a going concern. Since inception, we have incurred annual losses and negative cash flows from operations and have an accumulated deficit of approximately $230$259 million at December 31, 2007.2008. We have not derived revenue from the sale of products, and do not expect to receive revenue from product sales for at least several years. We may never be able to realize sufficient revenue to achieve or sustain profitability in the future.
We expect to incur additional operating losses over the foreseeable future. We have limited liquidity and capital resources and must obtain significant additional capital and other resources in order to sustain our product development efforts, to provide funding for the acquisition of technologies and intellectual property rights, preclinical and clinical testing of our anticipated products, pursuit of regulatory approvals, acquisition of capital equipment, laboratory and office facilities, establishment of production capabilities, general and administrative expenses and other working capital requirements. We rely on our cash reserves, proceeds from equity and debt offerings, proceeds from the transfer or sale of intellectual property rights, equipment, facilities or investments, government grants and funding from collaborative arrangements, to fund our operations. If we exhaust our cash reserves and are unable to obtain adequate financing, we may be unable to meet our operating obligations and we may be required to initiate bankruptcy proceedings. The financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the outcome of this uncertainty.
Principles of Consolidation
The consolidated financial statements include the accounts of StemCells, Inc., and our wholly owned subsidiary, StemCells California, Inc. Material intercompany accounts and transactions have been eliminated.
Use of Estimates
The preparation of financial statements in conformity with generally accepted accounting principles requires management to make judgments, assumptions and estimates that affect the amounts reported in our consolidated financial statements and accompanying notes. Actual results could differ materially from those estimates.
Significant estimates include the following:
| • | Accrued wind-down expenses (See Note | |
| • | The | |
| • | Valuation allowance against net deferred tax assets (See Note | |
| • | The fair value of warrants recorded as a liability in accordance with Emerging Issues Task Force IssueNo. 00-19,Accounting for Derivative Financial Instruments Indexed to and Potentially Settled in a Company’s Own StockEITF 00-19. The warrants were issued as part of our November 2008 financing (See Note 10). |
53
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Financial Instruments
Cash Equivalents and Marketable Securities
All money market and highly liquid investments with a maturity of 90 days or less at the date of purchase are classified as cash equivalents. Highly liquid investments with maturities of 365 days or less not previously classified as cash equivalents are classified as marketable securities, current. Investments with maturities greater than 365 days
51
are classified as marketable securities, non-current. Our marketable debt and equity securities have been classified and accounted for as available-for-sale. Management determines the appropriate classification of its investments in marketable debt and equity securities at the time of purchase and reevaluates the available-for-sale designations as of each balance sheet date. These securities are carried at fair value (see Note 2, “Financial Instruments,” below), with the unrealized gains and losses reported as a component of stockholders’ equity. The cost of securities sold is based upon the specific identification method.
If the estimated fair value of a security is below its carrying value, we evaluate whether we have the intent and ability to retain our investment for a period of time sufficient to allow for any anticipated recovery to the cost of the investment, and whether evidence indicating that the cost of the investment is recoverable within a reasonable period of time outweighs evidence to the contrary. Other-than-temporary declines in estimated fair value of all marketable securities are charged to “other income (expense), net.” After considering various criteria, including the duration of the impairment and our intent to liquidate all or part of our investment within a reasonably short period of time, we determined that the impairment of our investment in ordinary shares of ReNeuron (marketable equity securities) (see Note 2, “Financial Instruments,” below), was other than temporary. For the year ended December 31, 2008, we recorded on our “Consolidated Statements of Operations” under “Other Income (expense)” a loss of $2,082,894, which is the difference between the investment’s carrying value and its quoted market price at that date. No suchother than temporary impairment was recognized during the years ended December 31, 2007 2006 and 2005.2006.
Other Receivables
Our non-trade receivables generally consist of interest income on our financial instruments, revenue from licensing agreements and rent from our sub-lease tenants.
Estimated Fair Value of Financial Instruments
The estimated fair value of cash and cash equivalents, other receivables, accounts payable and the current portion of the bonds payable approximates their carrying values due to the short maturities of these instruments. The estimated fair value of our marketable debt securities approximates its carrying value based on current rates available to us for similar debt securities.
Property, Plant and Equipment
Property, plant, and equipment, including those held under capital lease, are stated at cost. Depreciation is computed by use of the straight-line method over the estimated useful lives of the assets, or the lease term if shorter, as follows:
| Building and improvements | 3 - 20 years | |||||||
| Machinery and equipment | 3 - 10 years | |||||||
| Furniture and fixtures | 3 - 10 years |
Repairs and maintenance costs are expensed as incurred.
Intangible Assets (Patent and License Costs)
Prior to fiscal year 2001, we capitalized certain patent costs, which are being amortized over the estimated life of the patent and would be expensed at the time such patents are deemed to have no continuing value. Since 2001, all
54
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
patent costs are expensed as incurred. License costs are capitalized as incurred and amortized over the estimated life of the license agreement.
Impairment of Long-Lived Assets
We review property, plant, and equipment and certain identifiable intangibles for impairment in accordance with Financial Accounting Standards Board (FASB) Statement of Financial Accounting Standards (SFAS) No. 144,Accounting for the Impairment of Long-Lived Assets and for Long-Lived Assets to Be Disposed Of.Long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate the carrying amount of an asset may not be recoverable. Recoverability of these assets is measured by comparing the carrying amount to future undiscounted cash flows the assets are expected to generate. If property, plant, and equipment and patents are
52
considered to be impaired, the impairment to be recognized equals the amount by which the carrying value of the assets exceeds its estimated fair market value. No such impairment was recognized during the years ended December 31, 2008, 2007 2006 and 2005.2006.
Warrant Liability
We account for our warrants in accordance withEITF 00-19, which defines how freestanding contracts that are indexed to and potentially settled in a company’s own stock should be measured and classified. The general concept underEITF 00-19 is that contracts that could require net-cash settlement should be classified as assets or liabilities and contracts that only provide for settlement in shares should be classified as equity. In order for a contract to be classified as equity, each of the specific conditions enumerated inEITF 00-19 must be met; these conditions are intended to identify situations in which net cash settlement could be forced upon the issuer. As part of our November 2008 financing, we issued warrants with a five year term to purchase 10,344,828 shares of our common stock at $2.30 per share. In accordance withEITF 00-19, we are required to classify the fair value of the warrants issued as a liability, with subsequent changes in fair value to be recorded as income (loss) on change in fair value of warrant liability. The fair value of the warrants is determined using the Black-Scholes-Merton (Black-Scholes) option pricing model and is affected by changes in inputs to that model including our stock price, expected stock price volatility and contractual term. We will continue to classify the fair value of the warrants as a liability until the warrants are exercised, expire or are amended in a way that would no longer require these warrants to be classified as a liability.
Revenue Recognition
We currently recognize revenue resulting from the licensing and use of our technology and intellectual property. Such licensing agreements may contain multiple elements, such as upfrontup-front fees, payments related to the achievement of particular milestones and royalties. Revenue from upfrontup-front fees for licensing agreements that contain multiple elements are generally deferred and recognized on a straight-line basis over the term of the agreement. Fees associated with substantive at risk performance-based milestones are recognized as revenue upon completion of the scientific or regulatory event specified in the agreement, and royalties received are recognized as earned. Revenue from collaborative agreements and grants are recognized as earned upon either the incurring of reimbursable expenses directly related to the particular research plan or the completion of certain development milestones as defined within the terms of the relevant collaborative agreement or grant.
Research and Development Costs
Our research and development expenses consist primarily of salaries and related personnel expenses, costs associated with clinical trials and regulatory submissions; costs associated with preclinical activities such as toxicology studies; certain patent-related costs such as licensing; facilities-related costs such as depreciation; lab equipment and supplies. Clinical trial expenses include payments to vendors such as clinical research organizations,
55
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
contract manufacturers, clinical trial sites, laboratories for testing clinical samples and consultants. All research and development costs are expensed as incurred.
Stock-Based Compensation
On January 1, 2006, we adopted SFAS No. 123 (revised 2004) (SFAS 123R),Share-Based Payment,, which revises SFAS 123,Accounting for Stock-Based Compensation, and supersedes Accounting Principles Board Opinion 25 (APB 25),Accounting for Stock Issued to Employees. SFAS 123R requires us to expense the fair value of our stock-based compensation awards to employees. We elected to use the modified prospective transition method as permitted by SFAS 123R and therefore have not restated our financial results for prior periods. Under this transition method, we apply SFAS 123R to new awards, as well as to awards that vest, are modified, repurchased, or cancelled after January 1, 2006.the date of adoption. The compensation cost we record for these awards are based on their grant-date fair value as calculated and amortized over their vesting period. See Note 6,7, “Stock-Based Compensation” for further information.
We account for stock options granted to non-employees in accordance with SFAS 123 and Emerging Issues Task Force (EITF)96-18Accounting For Equity Instruments That Are Issued To Other Than Employees For Acquiring, Or In Conjunction With Selling, Goods Or Services, and accordingly, expense the estimated fair value of such options as calculated using the Black-Scholes model over the service period. The estimated fair value is re-measured at each reporting date and is amortized over the remaining service period.
53
Income Taxes
We account for income taxes in accordance with SFAS No. 109, Accounting for Income Taxes(SFAS 109) and FASB Interpretation No. 48,Accounting for Uncertainty in Income Taxes, an interpretation of FASB Statement No. 109,as amended by FASB Staff PositionNo. 48-1 (FIN 48). This approach requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the carrying amounts and the tax bases of assets and liabilities. Income tax receivables and liabilities and deferred tax assets and liabilities are recognized based on the amounts that more likely than not will be sustained upon ultimate settlement with taxing authorities.
Developing our provision for income taxes and analyzing our uncertain tax positions requires significant judgment and knowledge of federal and state income tax laws, regulations and strategies, including the determination of deferred tax assets and liabilities and, any valuation allowances that may be required for deferred tax assets.
We assess the realization of our deferred tax assets to determine whether an income tax valuation allowance is required. Based on such evidence that can be objectively verified, we determine whether it is more likely than not that all or a portion of the deferred tax assets will be realized. The main factors that we consider include:
| • | Cumulative losses in recent years; | |
| • | Income/losses expected in future years; | |
| • | The applicable statute of limitations. |
Tax benefits associated with uncertain tax positions are recognized in the period in which one of the following conditions is satisfied: (1) the more likely than not recognition threshold is satisfied; (2) the position is ultimately settled through negotiation or litigation; or (3) the statute of limitations for the taxing authority to examine and challenge the position has expired. Tax benefits associated with an uncertain tax position are derecognized in the period in which the more likely than not recognition threshold is no longer satisfied.
We concluded that the realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance.
56
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Net Loss per Share
Basic net loss per share is computed based on the weighted-average number of shares of our common stock outstanding during the period. Diluted net loss per share is computed based on the weighted-average number of shares of our common stock and other dilutive securities.
The following are the basic and dilutive net loss per share computations for the last three fiscal years:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| Net loss | $ | (25,022,802 | ) | $ | (18,948,380 | ) | $ | (11,738,350 | ) | $ | (29,086,777 | ) | $ | (25,022,802 | ) | $ | (18,948,380 | ) | ||||||
| Weighted average shares outstanding used to compute basic and diluted net loss per share | 79,772,351 | 74,611,196 | 63,643,176 | 82,716,455 | 79,772,351 | 74,611,196 | ||||||||||||||||||
| Basic and diluted net loss per share | $ | (0.31 | ) | $ | (0.25 | ) | $ | (0.18 | ) | $ | (0.35 | ) | $ | (0.31 | ) | $ | (0.25 | ) | ||||||
54
Outstanding options, restricted stock units and warrants to purchase shares of our common stock were excluded from the computation of diluted net loss per share because the effect would have been anti-dilutive for all periods presented below:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| Outstanding options | 9,028,810 | 8,501,503 | 6,608,109 | 8,340,530 | 9,028,810 | 8,501,503 | ||||||||||||||||||
| Restricted stock units | 1,650,000 | — | — | |||||||||||||||||||||
| Outstanding warrants | 1,355,000 | 1,930,658 | 2,521,400 | 11,599,828 | 1,355,000 | 1,930,658 | ||||||||||||||||||
| Total | 10,383,810 | 10,432,161 | 9,129,509 | 21,590,358 | 10,383,810 | 10,432,161 | ||||||||||||||||||
Comprehensive Income (Loss)
Comprehensive income (loss) is comprised of net losses and other comprehensive income (or “OCI”). OCI includes certain changes in stockholders’ equity that are excluded from net losses. Specifically, we include in OCI changes in unrealized gains and losses on our marketable securities. Comprehensive incomeloss for the years ended December 31, 2008, 2007 2006 and 20052006 has been reflected in the Consolidated Statements of Stockholders’ Equity.
The activity in OCI is as follows:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| (Decrease) increase in unrealized gains/(losses) on marketable securities | $ | (2,756,268 | ) | $ | 3,442,125 | $ | (254,147 | ) | ||||||||||||||||
| (Decrease) increase in unrealized gains(losses) on marketable securities | $ | (1,806,768 | ) | $ | (2,756,268 | ) | $ | 3,442,125 | ||||||||||||||||
| Recognition in net loss, other than temporary impairment of marketable securities | 2,082,894 | — | — | |||||||||||||||||||||
| Reclassification adjustment for gains on marketable securities included in net income | (715,584 | ) | — | — | — | (715,584 | ) | — | ||||||||||||||||
| Other comprehensive income (loss) | $ | (3,471,852 | ) | $ | 3,442,125 | $ | (254,147 | ) | $ | 276,126 | $ | (3,471,852 | ) | $ | 3,442,125 | |||||||||
Recent Accounting Pronouncements
In September 2006,February 2008, the FASB issued SFASFASB Staff Position (FSP)No. FAS 157-2,Effective Date of FASB Statement No. 157(FSP 157-2).FSP 157-2 delays the effective date of SFAS 157 for nonfinancial assets and nonfinancial liabilities, except for certain items that are recognized or disclosed at fair value in the financial statements on a recurring basis (at least annually). We are currently evaluating the impact of SFAS 157 on our consolidated financial statements for items within the scope ofFSP 157-2, which will become effective beginning with our first quarter of 2009.
57
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
In October 2008, the FASB issued FSPNo. FAS 157-3,Determining the Fair Value Measurementsof a Financial Asset When the Market for That Asset Is Not Active(FSP 157-3).FSP 157-3 clarifies the application of SFAS 157, in a market that is not active and provides an example to illustrate key considerations in determining the fair value of a financial asset when the market for that financial asset is not active. This FSP shall be effective upon issuance, including prior periods for which financial statements have not been issued. Revisions resulting from a change in the valuation technique or its application shall be accounted for as a change in accounting estimate. Adoption ofFSP 157-3 did not have a material impact on our consolidated financial statements.
In April 2008, the FASB issued FSPNo. 142-3,Determination of the Useful Life of Intangible Assets(FSP 142-3).FSP 142-3 amends the factors that should be considered in developing renewal or extension assumptions used to determine the useful life of a recognized intangible asset under FASB Statement No. 142,Goodwill and Other Intangible Assets(SFAS 157)142).FSP 142-3 amends paragraph 11(d) of SFAS 157 defines fair value, establishes a framework and gives guidance regarding142 to require an entity to use its own assumptions about renewal or extension of an arrangement, adjusted for the methods used for measuring fair value, and expands disclosures about fair value measurements.entity-specific factors in paragraph 11 of SFAS 157142, even when there is likely to be substantial cost or material modifications.FSP 142-3 is effective for financial statements issued for fiscal years beginning after NovemberDecember 15, 2007,2008, and interim periods within those fiscal years.years, with early adoption prohibited. We are currently assessingdo not expect that the impact that SFAS 157 mayadoption ofFSP 142-3 on January 1, 2009, to have a material effect on our consolidated financial condition and results of operations and financial position.operations.
In FebruaryDecember 2007, the FASB issued SFAS No. 159,141R,The Fair Value Option for Financial Assets and Financial Liabilities — Including an amendment of FASB Statement No. 115Business Combinations(SFAS 159)141R). SFAS 159141R provides companies with principles and requirements on how an acquirer recognizes and measures in its financial statements the identifiable assets acquired, liabilities assumed, and any non controlling interest in the acquiree as well as the recognition and measurement of goodwill acquired in a business combination. SFAS 141R also requires certain disclosures to enable users of the financial statements to evaluate the nature and financial effects of the business combination. Acquisition costs associated with the business combination will generally be expensed as incurred. SFAS 141R is expected to expand the use of fair value accounting but does not affect existing standards which require certain assets or liabilities to be carried at fair value. The objectiveeffective for business combinations occurring in fiscal years beginning after December 15, 2008. Early adoption of SFAS 159141R is to improve financial reporting by providing companies with the opportunity to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without havingnot permitted. We will be required to apply complex hedge accounting provisions. Underthe guidance in SFAS 159,141R to any future business combinations effective January 1, 2009
In June 2008, the FASB issued EITF IssueNo. 07-05,Determining Whether an Instrument (or Embedded Feature) Is Indexed to an Entity’s Own Stock. EITF IssueNo. 07-05 clarifies the determination of whether an instrument (or an embedded feature) is indexed to an entity’s own stock, which would qualify as a company may choose, at specified election dates, to measure eligible items at fair valuescope exception under SFAS No. 133,Accounting for Derivative Instruments and report unrealized gains and losses on items for which the fair value option has been elected in earnings at each subsequent reporting date. SFAS 159Hedging Activities. EITF IssueNo. 07-05 is effective for financial statements issued for fiscal years beginning after NovemberDecember 15, 2007, and interim periods within those fiscal years.2008. Early adoption for an existing instrument is not permitted. We are currently assessingdo not expect the adoption of EITF IssueNo. 07-05 to have a material impact that SFAS 159 may have on our results of operations andconsolidated financial position.
5558
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
| Note 2. | Financial Instruments |
Cash, cash equivalents and marketable securities
The following table summarizes the fair value of our cash, cash equivalents and available-for-sale securities held in our investment portfolio:
| Gross Unrealized | Gross Unrealized | Gross | Gross | |||||||||||||||||||||||||||||
| Amortized Cost | Gains | Losses | Fair Value | Amortized | Unrealized | Unrealized | ||||||||||||||||||||||||||
| Cost | Gains | Losses | Fair Value | |||||||||||||||||||||||||||||
December 31, 2008 | ||||||||||||||||||||||||||||||||
| Cash | $ | 243,883 | $ | — | $ | — | $ | 243,883 | ||||||||||||||||||||||||
| Cash equivalents (money market accounts) | 29,799,103 | — | — | 29,799,103 | ||||||||||||||||||||||||||||
| Marketable debt securities, current (maturity within 1 year) | 4,002,537 | — | (7,748 | ) | 3,994,789 | |||||||||||||||||||||||||||
| Marketable equity securities, current | 186,803 | — | — | 186,803 | ||||||||||||||||||||||||||||
| Total cash, cash equivalents, and marketable securities | $ | 34,232,326 | $ | — | $ | (7,748 | ) | $ | 34,224,578 | |||||||||||||||||||||||
December 31, 2007 | December 31, 2007 | |||||||||||||||||||||||||||||||
| Cash | $ | 549,544 | $ | — | $ | — | $ | 549,544 | $ | 549,544 | $ | — | $ | — | $ | 549,544 | ||||||||||||||||
| Money market accounts | 5,079,564 | — | — | 5,079,564 | 5,079,564 | — | — | 5,079,564 | ||||||||||||||||||||||||
| Marketable debt securities (maturity within 90 days) | 4,130,404 | — | (343 | ) | 4,130,061 | 4,130,404 | — | (343 | ) | 4,130,061 | ||||||||||||||||||||||
| Total cash equivalents | 9,209,968 | — | (343 | ) | 9,209,625 | 9,209,968 | — | (343 | ) | 9,209,625 | ||||||||||||||||||||||
| Marketable debt securities (maturity within 1 year) | 26,680,824 | 19,137 | (3,548 | ) | 26,696,413 | 26,680,824 | 19,137 | (3,548 | ) | 26,696,413 | ||||||||||||||||||||||
| Total marketable securities, current | 26,680,824 | 19,137 | (3,548 | ) | 26,696,413 | 26,680,824 | 19,137 | (3,548 | ) | 26,696,413 | ||||||||||||||||||||||
| Marketable debt securities | 1,180,394 | 9,109 | — | 1,189,503 | 1,180,394 | 9,109 | — | 1,189,503 | ||||||||||||||||||||||||
| Marketable equity securities | 2,269,697 | — | (308,229 | ) | 1,961,468 | 2,269,697 | — | (308,229 | ) | 1,961,468 | ||||||||||||||||||||||
| Total marketable securities, non-current | 3,450,091 | 9,109 | (308,229 | ) | 3,150,971 | 3,450,091 | 9,109 | (308,229 | ) | 3,150,971 | ||||||||||||||||||||||
| Total cash, cash equivalents, and marketable securities | $ | 39,890,427 | $ | 28,246 | $ | (312,120 | ) | $ | 39,606,553 | $ | 39,890,427 | $ | 28,246 | $ | (312,120 | ) | $ | 39,606,553 | ||||||||||||||
December 31, 2006 | ||||||||||||||||||||||||||||||||
| Cash | $ | 162,685 | $ | — | $ | — | $ | 162,685 | ||||||||||||||||||||||||
| Money market accounts | 51,632,844 | — | — | 51,632,844 | ||||||||||||||||||||||||||||
| Total cash and cash equivalents | 51,795,529 | — | — | 51,795,529 | ||||||||||||||||||||||||||||
| Marketable equity securities, current | 2,319,505 | 1,813,141 | — | 4,132,646 | ||||||||||||||||||||||||||||
| Marketable equity securities, non-current | 1,758,795 | 1,374,837 | — | 3,133,632 | ||||||||||||||||||||||||||||
| Total cash, cash equivalents, and marketable securities | $ | 55,873,829 | $ | 3,187,978 | $ | — | $ | 59,061,807 | ||||||||||||||||||||||||
Our investment in marketable equity securities consists of ordinary shares inof ReNeuron Group plc, a publicly listed UK corporation. In July 2005, we entered into a license and settlementan agreement with ReNeuron Limited, a wholly owned subsidiary of ReNeuron Group plc, (collectively referred to as “ReNeuron”).ReNeuron. As part of the agreement, we granted ReNeuron a license that allows ReNeuron to exploit their “c-mycER” conditionally immortalized adult
56
human neural stem cell technology for therapy and other purposes. In return for the license, weWe received a 7.5% fully-diluted equity interest inshares of ReNeuron subject to certain anti-dilution provisions, andcommon stock, as well as a cross-license to the exclusive use of ReNeuron’s technology for certain diseases and conditions, including lysosomal storage diseases, spinal cord injury, cerebral palsy, and multiple sclerosis. The agreement also provides for full settlement of any potential claims that either we or ReNeuron might have had against the other in connection with any putative infringement of certain of each party’s patent rights prior to the effective date of the agreement. In July and August 2005 we received approximately 8,836,000 ordinary shares of ReNeuron common stock (net of approximately 104,000 shares that were transferred to NeuroSpheres), and subsequently, as a result of
59
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
certain anti-dilution provisions in the agreement, we received approximately 1,261,000 more shares, net of approximately 18,000 shares that were transferred to NeuroSpheres. In February 2007, we sold 5,275,000 ordinary shares of ReNeuron for net proceeds of approximately $3,075,000. We recognized approximately $716,000 as realized gain from this transaction. In February 2007, as a consequence of certain anti-dilution provisions in the agreement, ReNeuron issued us an additional 822,000 shares of common stock net of approximately 12,000 shares which were transferred to NeuroSpheres Ltd., (NeuroSpheres) an Alberta corporation from which we have licensed some of the patent rights that are subject to the agreement with ReNeuron. We recorded approximately $550,000 as other income for the additional shares. We owned 4,821,924approximately 4,822,000 ordinary shares of ReNeuron at December 31, 2008 and 2007.
If the fair value of a security is below its carrying value, we evaluate whether we have the intent and ability to retain our investment for a period of time sufficient to allow for any anticipated recovery to the cost of the investment, and whether evidence indicating that the cost of the investment is recoverable within a reasonable period of time outweighs evidence to the contrary. Other-than-temporary declines in estimated fair value of all marketable securities are charged to “other income (expense), net.” After considering various criteria, including the duration of the impairment and our intent to liquidate all or part of our investment within a reasonably short period of time, we determined that the impairment of our investment in ordinary shares of ReNeuron (marketable equity securities) (see Note 2, “Financial Instruments,” below), was other than temporary. For the year ended December 31, 2008, we recorded on our “Consolidated Statements of Operations” under “Other Income (expense)” a loss of $2,082,894, which is the difference between the investment’s carrying value and its quoted market price at that date. No other than temporary impairment was recognized during the years ended December 31, 2007 and 9,274,837 at December 31, 2006.
Changes in marketfair value as a result of changes in market price per share or the exchange rate between the US dollar and the British pound are accounted for under “other comprehensive income (loss)” if deemed temporary and are not recorded as “other income or loss” until the shares are disposed of and a gain or loss realized.realized or an impairment is considered other than temporary. After considering various criteria, including, the duration of the impairment and our intent to sell within a reasonably short period of time, we determined that the impairment of our investment in shares of ReNeuron (marketable equity securities) was other than temporary. For the year ended December 31, 2008, we recorded, on our “Consolidated Statements of Operations” under “Other Income (expense)”, a loss of $2,082,894, which is the difference between the investment’s carrying value and its quoted market price at that date. No other than temporary impairment was recognized during the years ended December 31, 2007 and 2006.
In accordance with FASB Staff PositionFAS 115-1 andFAS 124-1,The Meaning of Other-Than-Temporary Impairment and Its Application to Certain Investments, the following table shows the gross unrealized losses and fair value for those investments that were in an unrealized loss position as of December 31, 2007,2008, aggregated by investment category and the length of time that individual securities have been in a continuous loss position:
| Less than 12 Months | 12 Months of Greater | Total | Less than 12 Months | 12 Months of Greater | Total | |||||||||||||||||||||||||||||||||||||||||||
| Fair Value | Unrealized Loss | Fair Value | Unrealized Loss | Fair Value | Unrealized Loss | Fair | Unrealized | Fair | Unrealized | Fair | Unrealized | |||||||||||||||||||||||||||||||||||||
| Value | Loss | Value | Loss | Value | Loss | |||||||||||||||||||||||||||||||||||||||||||
December 31, 2007 | ||||||||||||||||||||||||||||||||||||||||||||||||
December 31, 2008 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Marketable debt securities | $ | 9,418,713 | $ | (3,891 | ) | $ | — | $ | (— | ) | $ | 9,418,713 | $ | (3,891 | ) | $ | 3,994,789 | $ | (7,748 | ) | $ | — | $ | — | $ | 3,994,789 | $ | (7,748 | ) | |||||||||||||||||||
| Marketable equity securities | 1,961,468 | (308,229 | ) | 1,961,468 | (308,229 | ) | 186,803 | — | — | — | 186,803 | — | ||||||||||||||||||||||||||||||||||||
| Total | $ | 11,380,181 | $ | (312,120 | ) | $ | — | $ | (— | ) | $ | 11,380,181 | $ | (312,120 | ) | $ | 4,181,592 | $ | (7,748 | ) | $ | — | $ | — | $ | 4,181,592 | $ | (7,748 | ) | |||||||||||||||||||
Unrealized losses in our marketable debt securities portfolio are due to eightfour U.S. corporate debt securities primarily consisting of commercial paper. For these securities, the unrealized losses are primarily due to a change in interest rates. Because we have the ability and intent to hold these investments until a forecasted recovery of carrying value, which may be maturity or call date, we do not consider these investments to be other-than-temporarily impaired as of December 31, 2007.2008. See Note 1, “Summary of Significant Accounting Policies — Cash Equivalents and Marketable Securities,” for further discussion of the criteria used to determine impairment of our marketable securities.
60
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Note Receivable
In December 2007, we committed to make a secured loan of up to $3.8 million to Progenitor Cell Therapy, LLC (PCT) in return for a period of exclusivity to allow for due diligence and negotiation of a possible acquisition transaction. Of this amount, $1.0 million was lent and outstanding at December 31, 2007 with the maturity date within twelve months from the effective date of the loan. In March 2008, we terminated discussions to acquire PCT. We anticipateIn April 2008 we were repaid the $1.0 million loan will be repaid in accordance with its terms.
In December 2008, we made a secured loan of £200,000 (approximately $298,000) to Stem Cell Sciences Plc in connection with a potential acquisition transaction. The loan accrues interest at 8% per annum and is repayable on June 23, 2009 if the acquisition of SCS does not occur before then.
| Note 3. | Fair Value Measurement |
Effective January 1, 2008, we adopted SFAS 157, except as it applies to the nonfinancial assets and nonfinancial liabilities subject to FSPSFAS 157-2. SFAS 157 clarifies that fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or a liability. As a basis for considering such assumptions, SFAS 157 establishes a three-tier value hierarchy, which prioritizes the inputs used in the valuation methodologies in measuring fair value:
Level 1 — Observable inputs that reflect quoted prices (unadjusted) for identical assets or liabilities in active markets.
Level 2 — Directly or indirectly observable inputs other than in Level 1, that include quoted prices for similar assets or liabilities in active markets or quoted prices for identical or similar assets or liabilities in markets that are not active.
Level 3 — Unobservable inputs which are supported by little or no market activity that reflects the reporting entity’s own assumptions about the assumptions that market participants would use in pricing the asset or liability
The fair value hierarchy also requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value.
In accordance with SFAS 157, we measure our financial assets and liabilities at fair value. Our cash equivalents and marketable securities are classified within Level 1 or Level 2. This is because our cash equivalents and marketable securities are valued primarily using quoted market prices or alternative pricing sources and models utilizing market observable inputs. We currently do not have any Level 3 financial assets or liabilities.
5761
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
The following table presents assets and liabilities measured at fair value:
| Fair Value Measurement | ||||||||||||
| at Reporting Date Using | ||||||||||||
| Quoted Prices | ||||||||||||
| in Active Markets for | Significant Other | As of | ||||||||||
| Identical Assets | Observable Inputs | December 31, | ||||||||||
| (Level 1) | (Level 2) | 2008 | ||||||||||
| Assets | ||||||||||||
| Cash Equivalents: | ||||||||||||
| Money market funds | $ | 356,000 | $ | — | $ | 356,000 | ||||||
| U.S. Treasury obligations | 29,443,103 | — | 29,443,103 | |||||||||
| Marketable Securities: | ||||||||||||
| Equity securities | 186,803 | — | 186,803 | |||||||||
| Corporate bonds | — | 2,798,580 | 2,798,580 | |||||||||
| Asset-backed securities | — | 1,196,209 | 1,196,209 | |||||||||
| Total assets | $ | 29,985,906 | $ | 3,994,789 | $ | 33,980,695 | ||||||
| Liabilities | ||||||||||||
| Bond obligation | $ | — | $ | 1,009,166 | $ | 1,009,166 | ||||||
| Note | Property, Plant and Equipment |
Property, plant and equipment balances at December 31 are summarized below:
| 2007 | 2006 | 2008 | 2007 | |||||||||||||
| Building and improvements | $ | 3,397,639 | $ | 3,369,775 | $ | 3,404,969 | $ | 3,397,639 | ||||||||
| Machinery and equipment | 6,002,945 | 4,712,950 | 6,308,603 | 6,002,945 | ||||||||||||
| Furniture and fixtures | 369,068 | 366,053 | 369,068 | 369,068 | ||||||||||||
| 9,769,652 | 8,448,778 | 10,082,640 | 9,769,652 | |||||||||||||
| Less accumulated depreciation and amortization | (5,864,248 | ) | (4,852,628 | ) | (6,909,172 | ) | (5,864,248 | ) | ||||||||
| Net property, plant and equipment, net | $ | 3,905,404 | $ | 3,596,150 | ||||||||||||
| Property, plant and equipment, net | $ | 3,173,468 | $ | 3,905,404 | ||||||||||||
Depreciation expense was approximately $1,045,000 in 2008, $1,012,000 in 2007, and $944,000 in 2006, and $958,000 in 2005.2006.
62
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
| Note | Intangible and Other Assets |
The components of our intangible assets at December 31 are summarized below:
| Gross Carrying | Accumulated | Gross Carrying | Accumulated | Net Carrying | ||||||||||||||||||||
Intangible Asset Class | amount | Amortization | Net Carrying Amount | Amount | Amortization | Amount | ||||||||||||||||||
2008 | ||||||||||||||||||||||||
| Patents | $ | 979,612 | $ | (515,255 | ) | $ | 464,357 | |||||||||||||||||
| License agreements | 1,785,998 | (1,604,817 | ) | 181,181 | ||||||||||||||||||||
| Total intangible assets | $ | 2,765,610 | $ | (2,120,072 | ) | $ | 645,538 | |||||||||||||||||
2007 | ||||||||||||||||||||||||
| Patents | $ | 979,612 | $ | (459,452 | ) | $ | 520,160 | $ | 979,612 | $ | (459,452 | ) | $ | 520,160 | ||||||||||
| License agreements | 1,761,623 | (1,519,116 | ) | 242,507 | 1,761,623 | (1,519,116 | ) | 242,507 | ||||||||||||||||
| Total intangible assets | $ | 2,741,235 | $ | (1,978,568 | ) | $ | 762,667 | $ | 2,741,235 | $ | (1,978,568 | ) | $ | 762,667 | ||||||||||
2006 | ||||||||||||||||||||||||
| Patents | $ | 979,612 | $ | (403,650 | ) | $ | 575,962 | |||||||||||||||||
| License agreements | 1,712,248 | (1,412,028 | ) | 300,220 | ||||||||||||||||||||
| Total intangible assets | $ | 2,691,860 | $ | (1,815,678 | ) | $ | 876,182 | |||||||||||||||||
Amortization expense was approximately $142,000 in 2008, $163,000 in 2007, and $101,000 in 2006, and $125,000 in 2005.2006.
The expected future annual amortization expense based on current balances of our intangible assets is as follows:
For the year ending December 31: | ||||||||||||
| 2008 | $ | 107,249 | ||||||||||
| 2009 | $ | 107,249 | $ | 119,687 | ||||||||
| 2010 | $ | 107,249 | $ | 107,499 | ||||||||
| 2011 | $ | 69,468 | $ | 69,718 | ||||||||
| 2012 | $ | 68,295 | $ | 68,545 | ||||||||
| 2013 | $ | 66,212 | ||||||||||
Other assets at December 31 are summarized below:
| 2007 | 2006 | 2008 | 2007 | |||||||||||||
| Prepaid royalties | $ | 180,250 | $ | 189,782 | $ | 551,199 | $ | 180,250 | ||||||||
| Security deposit (building lease) | 752,500 | 752,500 | 750,000 | 752,500 | ||||||||||||
| Restricted cash (letter of credit) | 778,079 | 778,079 | 778,079 | 778,079 | ||||||||||||
| Total other long-term assets | $ | 1,710,829 | $ | 1,720,361 | ||||||||||||
| Total other non-current assets | $ | 2,079,278 | $ | 1,710,829 | ||||||||||||
58
| Note | Accrued Expenses and Other |
Accrued expenses at December 31 are summarized below:
| 2007 | 2006 | 2008 | 2007 | |||||||||||||
| External services | $ | 360,340 | $ | 323,162 | $ | 466,360 | $ | 360,340 | ||||||||
| Employee compensation | 1,885,249 | 1,570,915 | 1,526,115 | 1,885,249 | ||||||||||||
| Other | 216,663 | 159,825 | 268,770 | 216,663 | ||||||||||||
| Total other accrued liabilities | $ | 2,462,252 | $ | 2,053,902 | ||||||||||||
| Total accrued expenses and other liabilities | $ | 2,261,245 | $ | 2,462,252 | ||||||||||||
| Note | Stock-Based Compensation |
We currently grant options under three equity incentive plans and as of December 31, 2007,2008, we had 12,000,00015,227,243 shares authorized under these three plans. However, atAt our annual stockholders meeting held on June 12,
63
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
2007, our stockholders approved an amendment to our 2006 Equity Incentive Plan to provide for an annual increase in the number of shares of common stock available for issuance under the plan each January 1 (beginning January 1, 2008) equal to 4% of the outstanding common shares as of that date. The amendment further provided an aggregate limit of 30,000,000 shares issuable pursuant to incentive stock optionsbased awards under the plan. Under these three plans we may grant incentive stock options, nonqualified stock options, stock appreciation rights, restricted stock, restricted stock units and performance-based shares to our employees, directors and consultants, at prices determined by our Board of Directors. Incentive stock options may only be granted to employees under these plans with a grant price not less than the fair market value on the date of grant.
Generally, stock options and restricted stock units granted to employees have a maximum term of ten years, and vest over a four year period from the date of grant; 25% vest at the end of one year, and 75% vest monthly over the remaining three-year service period. We may grant options with different vesting terms from time to time. Upon employee termination of service, any unexercised vested option will be forfeited three months following termination or the expiration of the option, whichever is earlier.
Our compensation expense for stock options and restricted stock units issued from our equity incentive plans for the last twothree fiscal years was as follows:
| 2007 | 2006 | 2008 | 2007 | 2006 | ||||||||||||||||
| Research and development expense | $ | 1,347,239 | $ | 1,048,697 | $ | 1,845,523 | $ | 1,347,239 | $ | 1,048,697 | ||||||||||
| General and administrative expense | 1,558,056 | 1,236,334 | 1,909,348 | 1,558,056 | 1,236,334 | |||||||||||||||
| Total employee stock-based compensation expense and effect on net loss | $ | 2,905,295 | $ | 2,285,031 | ||||||||||||||||
| Total stock-based compensation expense and effect on net loss | $ | 3,754,871 | $ | 2,905,295 | $ | 2,285,031 | ||||||||||||||
| Effect on basic and diluted net loss per common share | $ | (0.04 | ) | $ | (0.03 | ) | ||||||||||||||
As of December 31, 2007,2008, we have approximately $6,956,000$5,207,000 of total unrecognized compensation expense related to unvested awards granted under our various share-based plans that we expect to recognize over a weighted-average period of 1.62.1 years.
59
| 2005 | ||||
| Net loss: | ||||
| Net loss, as reported | $ | (11,738,350 | ) | |
| Deduct: Stock-based employee compensation expense determined under fair value-based method for all awards | (1,019,120 | ) | ||
| Pro forma net loss | $ | (12,757,470 | ) | |
| Basic and diluted earnings per common share: | ||||
| As reported | $ | (0.18 | ) | |
| Pro forma | $ | (0.20 | ) | |
| Shares used in computing basic and diluted loss per share | 63,643,176 | |||
The fair value of options granted is estimated as of the date of grant using the Black-Scholes option pricing model and expensed on a pro-rata straight-line basis over the period in which the stock options vest. The Black-Scholes option pricing model requires certain assumptions as of the date of grant. The weighted-average assumptions used for the last three fiscal years are as follows:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| Expected life (years)(1) | 6.25 | 6.25 | 5.00 | 7.24 | 6.25 | 6.25 | ||||||||||||||||||
| Risk-free interest rate(2) | 4.36 | % | 4.72 | % | 4.14 | % | 3.23 | % | 4.36 | % | 4.72 | % | ||||||||||||
| Expected volatility(3) | 95.2 | % | 109.0 | % | 100.7 | % | 94.0 | % | 95.2 | % | 109.0 | % | ||||||||||||
| Expected dividend yield(4) | 0 | % | 0 | % | 0 | % | 0 | % | 0 | % | 0 | % | ||||||||||||
| (1) | The expected term represents the period during which our stock-based awards are expected to be outstanding. In 2008 we estimated this amount based on historical experience of similar awards, giving consideration to the contractual terms of the awards, vesting requirements, and expectation of future employee behavior, including post-vesting terminations. The expected term in 2007 and 2006 is equal to the average of the contractual life of the stock option and its vesting period as of the date of grant. | |
| (2) | The risk-free interest rate is based on U.S. Treasury debt securities with maturities close to the expected term of the option as of the date of grant. | |
| (3) | Expected volatility is based on historical volatility over the most recent historical period equal to the length of the expected term of the option as of the date of grant. | |
| (4) | We have neither declared nor paid dividends on any share of common stock and we do not expect to do so in the foreseeable future. |
64
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
At the end of each reporting period we estimate forfeiture rates based on our historical experience within separate groups of employees and adjust the stock-based compensation expense accordingly.
60
A summary of our stock option activity and related information for the last three fiscal years is as follows:
| Outstanding Options | Outstanding Options | |||||||||||||||||||||||||||||||||||
| Weighted- | Weighted-Average | Aggregate | Weighted- | Weighted-Average | Aggregate | |||||||||||||||||||||||||||||||
| Shares Available | Number | Average | remaining | Intrinsic | Number | Average | Remaining | Intrinsic | ||||||||||||||||||||||||||||
| for Grant | of Shares | Exercise Price | contractual term | Value(1) | of Shares | Exercise Price | Contractual Term | Value(1) | ||||||||||||||||||||||||||||
Balance at December 31, 2004 | 6,339,406 | 6,682,201 | $ | 2.67 | ||||||||||||||||||||||||||||||||
| Granted | (1,075,481 | ) | 1,075,481 | $ | 4.75 | |||||||||||||||||||||||||||||||
| Exercised | (423,989 | ) | $ | 1.76 | ||||||||||||||||||||||||||||||||
| Cancelled (forfeited and expired) | 725,584 | (725,584 | ) | $ | 3.11 | |||||||||||||||||||||||||||||||
Balance at December 31, 2005 | 5,989,509 | 6,608,109 | $ | 3.02 | 6,608,109 | $ | 3.02 | |||||||||||||||||||||||||||||
| Granted | (2,818,684 | ) | 2,818,684 | $ | 2.38 | 2,818,684 | $ | 2.38 | ||||||||||||||||||||||||||||
| Exercised | (369,214 | ) | $ | 1.82 | (369,214 | ) | $ | 1.82 | ||||||||||||||||||||||||||||
| Cancelled (forfeited and expired) | 556,076 | (556,076 | ) | $ | 2.82 | (556,076 | ) | $ | 2.82 | |||||||||||||||||||||||||||
Balance at December 31, 2006 | 3,726,901 | 8,501,503 | $ | 2.88 | 8,501,503 | $ | 2.88 | |||||||||||||||||||||||||||||
| Granted | (2,484,100 | ) | 2,484,100 | $ | 2.33 | 2,484,100 | $ | 2.33 | ||||||||||||||||||||||||||||
| Exercised | (175,186 | ) | $ | 1.20 | (175,186 | ) | $ | 1.20 | ||||||||||||||||||||||||||||
| Cancelled (forfeited and expired) | 1,781,607 | (1,781,607 | ) | $ | 4.91 | (1,781,607 | ) | $ | 4.91 | |||||||||||||||||||||||||||
Balance at December 31, 2007 | 3,024,408 | 9,028,810 | $ | 2.36 | 7.26 | $ | 826,558 | 9,028,810 | $ | 2.36 | 7.26 | $ | 826,558 | |||||||||||||||||||||||
| Granted | 353,000 | $ | 1.24 | |||||||||||||||||||||||||||||||||
| Exercised | (114,700 | ) | $ | 1.11 | ||||||||||||||||||||||||||||||||
| Cancelled (forfeited and expired) | (926,580 | ) | $ | 2.44 | ||||||||||||||||||||||||||||||||
| Exercisable at December 31, 2007 | 4,600,618 | $ | 2.24 | 5.68 | $ | 826,558 | ||||||||||||||||||||||||||||||
Balance at December 31, 2008 | 8,340,530 | $ | 2.32 | 6.55 | $ | 692,739 | ||||||||||||||||||||||||||||||
| Exercisable at December 31, 2008 | 5,726,441 | $ | 2.33 | 5.76 | $ | 635,969 | ||||||||||||||||||||||||||||||
| Vested and expected to vest(2) | 8,337,077 | $ | 2.34 | 7.34 | $ | 826,558 | 7,927,918 | $ | 2.32 | 6.46 | $ | 685,157 | ||||||||||||||||||||||||
| (1) | Aggregate intrinsic value represents the value of the closing price per share of our common stock on the last trading day of the fiscal period in excess of the exercise price multiplied by the number of options outstanding or exercisable. | |
| (2) | Shares include options vested and those expected to vest net of estimated forfeitures. |
The estimated weighted average fair value per share of options granted was approximately $1.00 in 2008, $1.85 in 2007, and $2.37 in 2006, and $3.75 in 2005, based on the assumptions in the Black-Scholes model discussed above. Total intrinsic value of options exercised at time of exercise was approximately $396,700$39,000 in 2008, $397,000 in 2007, and $453,000 in 2006, and $519,000 in 2005.2006.
The following is a summary of changes in unvested options:
| Weighted average | Weighted Average | |||||||||||||||
| grant date fair | Grant Date Fair | |||||||||||||||
Unvested Options | Number of options | value | Number of Options | Value | ||||||||||||
| Unvested options at December 31, 2006 | 3,598,784 | $ | 2.16 | |||||||||||||
| Unvested options at December 31, 2007 | 4,428,209 | $ | 2.01 | |||||||||||||
| Granted | 2,484,100 | 1.85 | 353,000 | 1.00 | ||||||||||||
| Vested | (1,485,573 | ) | 2.14 | (1,792,976 | ) | 2.05 | ||||||||||
| Cancelled | (169,119 | ) | 1.88 | (374,144 | ) | 1.95 | ||||||||||
| Unvested options at December 31, 2007 | 4,428,192 | $ | 2.00 | |||||||||||||
| Unvested options at December 31, 2008 | 2,614,089 | $ | 1.85 | |||||||||||||
The estimated fair value of options vested were approximately $3,671,000 in 2008, $3,173,000 in 2007 and $2,292,000 in 2006.
6165
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
The following table presents weighted average exercise price and term information about significant option groups outstanding at December 31, 2007:2008:
| Options outstanding at December 31, 2007 | ||||||||||||||||||||||||||||||||
| Options Outstanding at December 31, 2008 | Options Outstanding at December 31, 2008 | |||||||||||||||||||||||||||||||
| Weighted Average | Weighted Average | Weighted Average | Weighted Average | Aggregate Intrinsic | ||||||||||||||||||||||||||||
| Range of | Remaining | Exercise | Remaining | Exercise | Value at December 31, | |||||||||||||||||||||||||||
Exercise Prices | Number Outstanding | Term (Yrs.) | Price | Aggregate Intrinsic Value | Number Outstanding | Term (Yrs.) | Price | 2008 | ||||||||||||||||||||||||
| Less than $2.00 | 2,383,594 | 5.4 | $ | 1.19 | $ | 826,558 | 2,340,086 | 5.5 | $ | 1.17 | $ | 692,739 | ||||||||||||||||||||
| $2.00 — $3.99 | 5,882,631 | 8.1 | $ | 2.45 | — | 5,312,661 | 7.1 | $ | 2.44 | — | ||||||||||||||||||||||
| $4.00 — $5.99 | 762,585 | 7.0 | $ | 5.23 | — | 687,783 | 6.1 | $ | 5.27 | — | ||||||||||||||||||||||
| 9,028,810 | $ | 826,558 | 8,340,530 | $ | 2.32 | $ | 692,739 | |||||||||||||||||||||||||
| Options vested at December 31, 2007 | ||||||||||||||||||||||||||||||||
| Weighted Average | Weighted Average | |||||||||||||||||||||||||||||||
| Range of | Remaining | Exercise | ||||||||||||||||||||||||||||||
Exercise Prices | Number Outstanding | Term (Yrs.) | Price | Aggregate Intrinsic Value | ||||||||||||||||||||||||||||
| Less than $2.00 | 2,100,174 | 5.1 | $ | 1.14 | $ | 826,558 | ||||||||||||||||||||||||||
| $2.00 — $3.99 | 1,999,056 | 6.1 | $ | 2.65 | — | |||||||||||||||||||||||||||
| $4.00 — $5.99 | 501,388 | 6.7 | $ | 5.17 | — | |||||||||||||||||||||||||||
| 4,600,618 | $ | 826,558 | ||||||||||||||||||||||||||||||
| Options expected to vest after at December 31, 2007 | ||||||||||||||||||||||||||||||||
| Weighted Average | Weighted Average | |||||||||||||||||||||||||||||||
| Range of | Remaining | Exercise | ||||||||||||||||||||||||||||||
Exercise Prices | Number Outstanding | Term (Yrs.) | Price | Aggregate Intrinsic Value | ||||||||||||||||||||||||||||
| Less than $2.00 | 232,886 | 7.5 | $ | 1.61 | $ | — | ||||||||||||||||||||||||||
| $2.00 — $3.99 | 3,278,971 | 9.1 | $ | 2.35 | — | |||||||||||||||||||||||||||
| $4.00 — $5.99 | 224,602 | 7.7 | $ | 5.34 | — | |||||||||||||||||||||||||||
| 3,736,459 | $ | — | ||||||||||||||||||||||||||||||
| Vested Options Outstanding at December 31, 2008 | ||||||||
Range of Exercise Prices | Number Outstanding | Weighted Average Exercise Price | ||||||
| Less than $2.00 | 1,981,126 | $ | 1.16 | |||||
| $2.00 — $3.99 | 3,161,619 | $ | 2.53 | |||||
| $4.00 — $5.99 | 583,696 | $ | 5.26 | |||||
| 5,726,441 | $ | 2.33 | ||||||
Restricted Stock Units
In March 2008, we granted restricted stock units to certain employees that entitle the holders to receive shares of our common stock upon vesting. These restricted stock units vest over a three-year period from the date of grant: one-third of the award will vest on each grant date anniversary over the following three years. The fair value of restricted stock units granted are based upon the market price of the underlying common stock as if it were vested and issued on the date of grant.
A summary of our restricted stock unit activity for the year ended December 31, 2008 is as follows:
| Weighted-Average | ||||||||
| Number of RSUs | Grant Date Fair Value | |||||||
| Outstanding at January 1, 2008 | — | — | ||||||
| Granted | 1,650,000 | $ | 1.26 | |||||
| Exercised | — | — | ||||||
| Cancelled | — | — | ||||||
| Outstanding at December 31, 2008 | 1,650,000 | $ | 1.26 | |||||
| Vested RSUs outstanding at December 31, 2008 | — | — | ||||||
Stock Appreciation Rights
In July 2006, we granted cash-settled Stock Appreciation Rights (SARs) to certain employees under the 2006 Equity Incentive Plan. The SARs give the holder the right, upon exercise, to the difference between the price per share of our common stock at the time of exercise and the exercise price of the SAR. The exercise price of the SAR is equal to the market price of our common stock at the date of grant. The SARs vest 25% on the first anniversary of the grant date and 75% vest monthly over the remaining three-year service period. Compensation expense is based on the fair value of SARs which is calculated using the Black-Scholes option pricing model. The share-based compensation expenses and liability are re-measured at each reporting date through the date of settlement. The share-based compensation liability as re-measured at December 31, 2008 was $500,720.
6266
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
The following is a summary of the changes in non-vested SARs for the last twothree fiscal years:
| 2007 | 2006 | |||||||||||||||
| Weighted | Weighted | |||||||||||||||
| Average Exercise | Average Exercise | |||||||||||||||
| Number | Price | Number | Price | |||||||||||||
| Outstanding at January 1 | 1,564,599 | $ | 2.00 | — | — | |||||||||||
| Granted | — | — | 1,564,599 | $ | 2.00 | |||||||||||
| Exercised | — | — | — | — | ||||||||||||
| Forfeited and expired | (86,380 | ) | — | — | — | |||||||||||
| Outstanding SARs at December 31 | 1,478,219 | $ | 2.00 | 1,564,599 | $ | 2.00 | ||||||||||
| SARs exercisable at December 31 | 47,370 | $ | 2.00 | — | — | |||||||||||
| 2008 | 2007 | 2006 | ||||||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||||||
| Average | Average | Average | ||||||||||||||||||||||
| Exercise | Exercise | Exercise | ||||||||||||||||||||||
| Number | Price | Number | Price | Number | Price | |||||||||||||||||||
| Outstanding at January 1, | 1,478,219 | $ | 2.00 | 1,564,599 | $ | 2.00 | — | — | ||||||||||||||||
| Granted | — | — | — | — | 1,564,599 | $ | 2.00 | |||||||||||||||||
| Exercised | — | — | — | — | — | — | ||||||||||||||||||
| Forfeited | (47,390 | ) | (86,380 | ) | $ | 2.00 | — | — | ||||||||||||||||
| Outstanding at December 31, | 1,430,829 | $ | 2.00 | 1,478,219 | $ | 2.00 | 1,564,599 | $ | 2.00 | |||||||||||||||
| Exercisable at December 31, | 864,467 | $ | 2.00 | 506,754 | $ | 2.00 | — | — | ||||||||||||||||
The total compensation expense related to SARs was approximately $73,000 in 2008, $135,000 in 2007 and $294,000 in 2006. At December 31, 2007,2008, approximately $752,000$318,000 of unrecognized compensation expense related to SARs is expected to be recognized over a weighted average period of approximately 1.5 years.1 year. The resulting effect on net loss and net loss per share attributable to common stockholders is not likely to be representative of the effects in future periods, due to changes in the fair value calculation which is dependent on the stock price, volatility, interest and forfeiture rates, additional grants and subsequent periods of vesting.
| Note | Wind-down and exit costs |
In October 1999, we relocated to California from Rhode Island and established a wind downwind-down reserve for the estimated lease payments and operating costs of the Rhode Island facilities through an expected disposal date of June 30, 2000. We did not fully sublet the Rhode Island facilities in 2000. Even though we intend to dispose of the facility at the earliest possible time, we cannot determine with certainty a fixed date by which such disposal will occur. In light of this uncertainty, we periodically re-evaluate and adjust the reserve. We consider various factors such as our lease payments through to the end of the lease, operating expenses, the current real estate market in Rhode Island, and estimated subtenant income based on actual and projected occupancy.
The components of our wind-down reserve at December 31 are as follows:
| 2007 | 2006 | 2008 | 2007 | |||||||||||||
| Accrued wind-down reserve at beginning of period | $ | 5,512,000 | $ | 6,098,000 | $ | 4,875,000 | $ | 5,512,000 | ||||||||
| Less actual expenses recorded against estimated reserve during the period | (1,420,000 | ) | (1,295,000 | ) | (1,293,000 | ) | (1,420,000 | ) | ||||||||
| Additional expense recorded to revise estimated reserve at period-end | 783,000 | 709,000 | 866,000 | 783,000 | ||||||||||||
| Revised reserve at period-end | 4,875,000 | 5,512,000 | 4,448,000 | 4,875,000 | ||||||||||||
| Add deferred rent at period end | 1,268,000 | 1,238,000 | 1,065,000 | 1,268,000 | ||||||||||||
| Total accrued wind-down expenses at period-end (current and non current) | $ | 6,143,000 | $ | 6,750,000 | $ | 5,513,000 | $ | 6,143,000 | ||||||||
| Accrued wind-down expenses, current | $ | 1,374,000 | $ | 1,252,000 | ||||||||||||
| Non current | 4,769,000 | 5,498,000 | ||||||||||||||
| Accrued wind-down expenses, current portion | $ | 1,420,000 | $ | 1,374,000 | ||||||||||||
| Non current portion | 4,093,000 | 4,769,000 | ||||||||||||||
| Total accrued wind-down expenses | $ | 6,143,000 | $ | 6,750,000 | $ | 5,513,000 | $ | 6,143,000 | ||||||||
6367
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
| Note | Commitments and Contingencies |
Leases
We entered into direct financing transactions with the State of Rhode Island and received proceeds from the issuance of industrial revenue bonds totaling $5,000,000 to finance the construction of Rhode Island’s pilot manufacturing facility. The related lease agreements are structured such that lease payments fully fund all semiannual interest payments and annual principal payments through maturity in August 2014. Interest rates vary with the respective bonds’ maturities, ranging from 8.2% to 9.5%. The outstanding principal and interest owed at December 31, 2008 was approximately $1,345,000. The bonds contain certain restrictive covenants which limit, among other things, the payment of cash dividends and the sale of the related assets.
Operating leases
We entered into a fifteen-year lease agreement for a laboratory facility in Rhode Island in connection with a sale and leaseback arrangement in 1997. The lease term expires June 30, 2013. The lease contains escalating rent payments, which we recognize on a straight-line basis. At December 31, 2007,2008, deferred rent expense was approximately $1,268,000$1,065,000 for this facility and is included as part of the wind-down accrual on the accompanying Consolidated Balance Sheet.
We entered into and amended a lease agreement for an approximately 68,000 square foot facility located at the Stanford Research Park in Palo Alto, California. The facility includes space for animals, laboratories, offices, and a GMP (Good Manufacturing Practices) suite. GMP facilities can be used to manufacture materials for clinical trials. The lease term expires March 31, 2010. Under the term of the agreement we were required to provide a letter of credit for a total of approximately $778,000, which serves as a security deposit for the duration of the lease term. The letter of credit issued by our financial institution is collateralized by a certificate of deposit for the same amount, which is reflected as restricted cash in other assets, non-current on our Consolidated Balance Sheets. The lease contains escalating rent payments, which we recognize on a straight-line basis. At December 31, 2007,2008, deferred rent was approximately $728,000,$437,000 and is reflected as deferred rent on our Consolidated Balance Sheet. At December 31, 2007,2008, we had a space-sharing agreement covering approximately 10,451 square feet of this facility. We receive base payments plus a proportionate share of the operating expenses based on square footage over the term of the agreement.
The table below summarizes the components of rent expense atfor the fiscal year endended December 31, as follows:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| Rent expense | $ | 2,963,339 | $ | 2,967,911 | $ | 2,983,879 | $ | 3,077,430 | $ | 3,077,431 | $ | 2,967,911 | ||||||||||||
| Sublease income | (492,306 | ) | (616,600 | ) | (962,757 | ) | (809,065 | ) | (606,398 | ) | (616,600 | ) | ||||||||||||
| Rent expense, net | $ | 2,471,033 | $ | 2,351,311 | $ | 2,021,122 | $ | 2,268,365 | $ | 2,471,033 | $ | 2,351,311 | ||||||||||||
6468
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Future minimum lease payments under all leases and bonds payable at December 31, 20072008 are as follows:
| Bonds | Capital | Operating | Sublease | Bonds | Capital | Operating | Sublease | |||||||||||||||||||||||||
| Payable | Leases | Leases | Income | Payable | Leases | Leases | Income | |||||||||||||||||||||||||
| 2008 | $ | 244,531 | $ | 19,862 | $ | 3,469,017 | $ | 388,989 | ||||||||||||||||||||||||
| 2009 | 244,572 | 19,862 | 3,536,843 | 387,210 | $ | 244,572 | $ | 19,862 | $ | 3,536,843 | $ | 652,624 | ||||||||||||||||||||
| 2010 | 242,559 | 6,623 | 1,767,304 | 97,508 | 242,559 | 6,623 | 1,767,304 | 97,508 | ||||||||||||||||||||||||
| 2011 | 242,321 | — | 1,171,875 | — | 242,321 | — | 1,171,875 | — | ||||||||||||||||||||||||
| 2012 | 240,666 | — | 1,171,875 | — | 240,666 | — | 1,171,875 | — | ||||||||||||||||||||||||
| 2013 | 237,593 | — | 732,422 | — | ||||||||||||||||||||||||||||
| Thereafter | 374,444 | — | 732,422 | — | 136,852 | — | — | — | ||||||||||||||||||||||||
| Total minimum lease payments | 1,589,093 | 46,347 | $ | 11,849,336 | $ | 873,707 | 1,344,563 | 26,485 | $ | 8,380,319 | $ | 750,132 | ||||||||||||||||||||
| Less amounts representing interest | 443,677 | 3,548 | 335,396 | 1,217 | ||||||||||||||||||||||||||||
| Present value of bonds payable payments | 1,145,416 | 42,799 | ||||||||||||||||||||||||||||||
| Present value of bonds payable and capital lease payments | 1,009,167 | 25,268 | ||||||||||||||||||||||||||||||
| Less current maturities | 136,250 | 17,530 | 149,167 | 18,739 | ||||||||||||||||||||||||||||
| Bonds payable, less current maturities | $ | 1,009,166 | $ | 25,269 | $ | 860,000 | $ | 6,529 | ||||||||||||||||||||||||
Contingencies
In July 2006, we filed suit against Neuralstem, Inc., in the Federal District Court for the District of Maryland, alleging that its Neuralstem’s activities violate claims in four of the patents we exclusively licensed from NeuroSpheres. Neuralstem has filed a motion for dismissal or summary judgment in the alternative, citing Title 35, Section 271(e)(1) of the United States Code, which says that it is not an act of patent infringement to make, use or sell a patented invention “solely for uses reasonably related to the development and submission of information” to the FDA. Neuralstem argues that because it does not have any therapeutic products on the market yet, the activities complained of fall within the protection of Section 271(e)(1) — that is, basically, that the suit is premature. This issue will be decided after discovery is complete. Subsequent to filing its motion to dismiss, in December 2006, Neuralstem petitioned the U.S. Patent and Trademark Office (PTO) to reexamine two of the patents in our infringement action against Neuralstem, namely U.S. Patent No. 6,294,346 (claiming the use of human neural stem
65
cells for drug screening) and U.S. Patent No. 7,101,709 (claiming the use of human neural stem cells for screening biological agents). In April 2007, Neuralstem petitioned the PTO to reexamine the remaining two patents in the suit, namely U.S. Patent No. 5,851,832 (claiming methods for proliferating human neural stem cells) and U.S. Patent No. 6,497,872 (claiming methods for transplanting human neural stem cells). These requests were granted by the PTO and, in June 2007, the parties voluntarily agreed to stay the pending litigation while the PTO considers these reexamination requests. In October 2007, Neuralstem petitioned the PTO to reexamine a fifth patent, namely U.S. Patent No. 6,103,530, which claims a culture medium for proliferating mammalian neural stem cells. In September 2007,April 2008, the PTO upheld the ’832 and ’872 patents, as amended, and issued first officeNotices of Intent to Issue anEx ParteReexamination Certificate for both. In August 2008, the PTO upheld the ’530 patent, as amended, and issued a Notice of Intent to Issue anEx ParteReexamination Certificate. The remaining two patents are still under review by the PTO.
In May 2008, we filed a second patent infringement suit against Neuralstem and its two founders, Karl Johe and Richard Garr. In this suit, which we filed in the Federal District Court for the Northern District of California, we allege that Neuralstem’s activities infringe claims in two patents we exclusively license from NeuroSpheres, specifically U.S. Patent No. 7,361,505 (claiming composition of matter of human neural stem cells derived from any source material) and U.S. Patent No. 7,115,418 (claiming methods for proliferating human neural stem cells). In addition, we allege various state law causes of action against Neuralstem arising out of its repeated derogatory statements to the public about our patent portfolio. Also in May 2008, Neuralstem filed suit against us and
69
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
NeuroSpheres in the Federal District Court for the District of Maryland seeking a declaratory judgment that the ’505 and ’418 patents are either invalid or are not infringed by Neuralstem and that Neuralstem has not violated California state law. In August 2008, the California court transferred our lawsuit against Neuralstem to Maryland for resolution on the merits. We anticipate that the Maryland District Court will consolidate these actions in each of the first four reexaminations. The Company has since filed its first responsessome manner prior to each of these, and expects all four patents to re-issue in 2008.trial.
| Note | Warrant Liability |
In November 2008, we sold 13,793,104 units to institutional investors at a price of $1.45 per unit, for gross proceeds of $20,000,000. The units, each of which consisted of one share of common stock and a warrant to purchase 0.75 shares of common stock at an exercise price of $2.30 per share, were offered as a registered direct offering under an effective shelf registration statement previously filed with and declared effective by the Securities and Exchange Commission. We received total proceeds, net of offering expenses and placement agency fees, of approximately $18,637,000. We recorded the fair value of the warrants to purchase 10,344,828 shares of our common stock as a liability. The fair value of the warrant liability will be revalued at the end of each reporting period, with the change in fair value of the warrant liability recorded as a gain or loss in our Consolidated Statement of Operations. We used the Black-Scholes option pricing model to estimate the fair value of these warrants. In using this model, we make certain assumptions about risk-free interest rates, dividend yields, volatility and expected term of the warrants. Risk-free interest rates are derived from the yield on U.S. Treasury securities. Dividend yields are based on our historical dividend payments, which have been zero to date. Volatility is derived from the historical volatility of our common stock as traded on Nasdaq. The expected term of the warrants is based on the time to expiration of the warrants from the date of measurement.
The assumptions used for the Black-Scholes option pricing model are as follows:
| To Calculate | ||||||||
| To Calculate | Fair Value at | |||||||
| Fair Value on | December 31, | |||||||
| Date of Issuance | 2008 | |||||||
| Expected life (years) | 5.5 | 5.4 | ||||||
| Risk-free interest rate | 2.42 | % | 1.60 | % | ||||
| Expected volatility | 83.8 | % | 84.5 | % | ||||
| Expected dividend yield | 0 | % | 0 | % | ||||
| Change in Fair Value | ||||||||||||
| of Warrant Liability | ||||||||||||
| At December 31, | At November 17, | at December 31, | ||||||||||
| 2008 | 2008 | 2008 | ||||||||||
| Fair value of warrant liability | $ | 8,439,931 | $ | 7,502,690 | $ | 937,241 | ||||||
The fair value of the warrants will continue to be classified as a liability until such time as the warrants are exercised, expire or an amendment of the warrant agreement renders these warrants to be no longer classified as a liability.
| Note 11. | Common Stock |
We have neither declared nor paid dividends on any share of common stock and do not expect to do so in the foreseeable future.
70
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Sale of common stock
Major transactions involving our common stock for the previous three years include the following:
| • | In November 2008, we sold 13,793,104 units to institutional investors at a price of $1.45 per unit, for gross proceeds of $20,000,000. The units, each of which consisted of one share of common stock and a warrant to purchase 0.75 shares of common stock at an exercise price of $2.30 per share, were offered as a registered direct offering under an effective shelf registration statement previously filed with and declared effective by the Securities and Exchange Commission. We received total proceeds net of offering expenses and placement agency fees of approximately $18,637,000. | |
| • | In April 2007, a warrant issued as part of | |
| • | ||
| • | ||
| • | In March 2006, a warrant issued as part of | |
Stock Issued For Technology Licenses
Under license agreements with NeuroSpheres, Ltd., we obtained an exclusive patent license covering all uses of certain neural stem cell technology. We made up-front payments to NeuroSpheres of 65,000 shares of our common stock and $50,000, and will make additional cash payments as stated milestones are achieved. Effective in 2004, we began making annual $50,000 payments, creditable against certain royalties.
Pursuant to the terms of a license agreement with the California Institute of Technology (Cal Tech) and our acquisition of its wholly owned subsidiary, StemCells California, we issued 14,513 shares of common stock to Cal Tech. We issued an additional 12,800 shares of common stock to Cal Tech with a market value of approximately $40,000 in May 2000, upon execution of an amendment adding four families of patent applications to the license
66
agreement. In August 2002, we acquired an additional license from Cal Tech for a different technology, pursuant to which we issued 27,535 shares of our common stock with a market value of approximately $35,000. We also issued 3,865 shares (market(with a market value of approximately $10,000)$10,000 each year), 6,924 shares in 2008, 3,865 shares in 2007, 3,848 shares (market value of approximately $10,000) in 2006, and 9,535 shares (market value of approximately $15,000) in 2004 of our common stock to Cal Tech for the issuance and annual license fees of two patents covered under this additional license.
71
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
Common Stock Reserved
We reserved the following shares of common stock for the exercise of options, warrants and other contingent issuances of common stock, as of December 31, 2007:2008:
| Shares reserved for | ||||
| Shares reserved for warrants related to financing transactions | ||||
| Shares reserved for license agreements | ||||
| Total | ||||
| Note | Grant Revenue |
In October 2008, we were awarded a $305,000 grant from the National Institute of Diabetes and Digestive and Kidney Diseases to research and develop a potential cell-based therapeutic for liver disease arising from infection by the hepatitis C virus. The award is a Phase I grant under the Small Business Innovation Research (SBIR) Program of the National Institutes of Health. Should the objectives of the research funded by this grant be met, we anticipate applying for Phase II and additional funding under the SBIR Program. We recognized approximately $26,000 as grant revenue in 2008 related to this grant.
In September 2004, we were awarded a Small Business Technology Transfer (STTR) grant for approximately $464,000 for studies in Alzheimer’s disease conducted over an 18 month period. The grant supported joint work with Dr. George A. Carlson of the McLaughlin Research Institute (MRI) in Great Falls, Montana. We received and recognized approximately $26,000 in 2006, $186,000 in 2005, and $38,000 in 2004 as grant revenue, the remainder was reimbursed to MRI.
| Note | 401(k) Plan |
Our 401(k) Plan covers substantially all of our employees. Participants in the plan are permitted to contribute a fixed percentage of their total annual cash compensation to the plan (subject to the maximum employee contribution defined by law). We match 50% of employee contributions, up to a maximum of 6% of each employee’s eligible compensation in the form of shares of common stock. We recorded an expense of $181,000 in 2008, $179,000 in 2007, and $157,000 in 2006 and $111,000 in 2005 for our contributions under our 401(k) Plan.
| Note | Income Taxes |
In July 2006, the FASB issued FIN 48 which clarifies the accounting for uncertainty in income taxes by prescribing the recognition threshold a tax position is required to meet before being recognized in the financial statements. We adopted FIN 48 effective January 1, 2007. The adoption of FIN 48 did not impact our consolidated financial condition, results of operations or cash flows. At the adoption date of January 1, 2007 and as of December 31, 2008 and 2007, we have not recorded any unrecognized tax benefits. Deferred income taxes reflect the tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting
6772
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
purposes and the amounts used for income tax purposes. Significant components of our deferred tax assets and liabilities at December 31 are as follows:
| 2007 | 2006 | 2008 | 2007 | |||||||||||||
| Deferred tax assets: | ||||||||||||||||
| Capitalized research and development costs | $ | 31,779,000 | $ | 25,058,000 | $ | 38,670,000 | $ | 31,779,000 | ||||||||
| Net operating losses | 42,730,000 | 41,015,000 | 42,247,000 | 42,716,000 | ||||||||||||
| Research and development credits | 6,103,000 | 5,671,000 | 6,671,000 | 6,103,000 | ||||||||||||
| Accrued wind down cost | 1,950,000 | 2,205,000 | 1,780,000 | 1,950,000 | ||||||||||||
| Stock-based compensation | 245,000 | 180,000 | 465,000 | 245,000 | ||||||||||||
| Impaired asset | 833,000 | — | ||||||||||||||
| Other | 315,000 | 361,000 | 458,000 | 329,000 | ||||||||||||
| 83,122,000 | 74,490,000 | 91,124,000 | 83,122,000 | |||||||||||||
| Valuation allowance | (83,122,000 | ) | (74,490,000 | ) | (91,124,000 | ) | (83,122,000 | ) | ||||||||
| Net deferred tax assets | $ | — | $ | — | $ | — | $ | — | ||||||||
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance increased by approximately $8,002,000 in 2008, $8,632,000 in 2007, and $7,105,000 in 2006, and $5,027,000 in 2005.2006.
As of December 31, 2007,2008, we had the following:
| • | Net operating loss carry forwards for federal income tax purposes of approximately | |
| • | Federal research and development tax credits of approximately | |
| • | Net operating loss carry forwards for state income tax purposes of approximately | |
| • | State research and development tax credits of approximately |
The effective tax rate as a percentage of income before income taxes differs from the statutory federal income tax rate (when applied to income before income taxes) for the years ended December 31 as follows:
| 2007 | 2006 | 2005 | 2008 | 2007 | 2006 | |||||||||||||||||||
| Statutory federal income tax (benefit) rate | (34 | )% | (34 | )% | (34 | )% | (34 | )% | (34 | )% | (34 | )% | ||||||||||||
| State income tax (benefit) rate | (6 | ) | (6 | ) | (6 | ) | (6 | ) | (6 | ) | (6 | ) | ||||||||||||
| Increase (decrease) resulting from: | ||||||||||||||||||||||||
| Increase resulting from: | ||||||||||||||||||||||||
| Expenses not deductible for taxes | 4.9 | 5.3 | 2.8 | 5.8 | 4.9 | 5.3 | ||||||||||||||||||
| Increase in valuation allowance | 35.1 | 34.7 | 37.2 | 34.2 | 35.1 | 34.7 | ||||||||||||||||||
| Effective tax (benefit) rate | 0 | % | 0 | % | 0 | % | 0 | % | 0 | % | 0 | % | ||||||||||||
Our policy is to recognize interest and penalties related to income tax matters in income tax expense. Because we have no tax liabilities, no tax-related interest and penalties have been expensed in our consolidated statements of operations during 20072008 or accrued as a liability in our consolidated balance sheets at December 31, 2007.2008. We do not anticipate any significant changes to total unrecognized tax benefits as a result of settlement of audits or the expiration of statute of limitations within the next twelve months.
6873
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
We file U.S. federal income tax returns, as well as tax returns with the State of California and the State of Rhode Island. Due to the carry forward of unutilized net operating losses and research and development credits, our federal tax returns from 19931994 forward remain subject to examination by the Internal Revenue Service, and our State of California tax returns from 19992000 forward and our State of Rhode Island tax returns from 20022003 forward remain subject to examination by the respective state tax authorities.
| Note 15. | Subsequent Events |
At December 31, 2008, we owned 4,821,924 shares of ReNeuron (marketable equity securities) trading on the Alternative Investment Market (a sub-market of the London Stock Exchange) with a carrying and fair market value of $187,000. In the first quarter of 2009, we sold in aggregate, approximately 2,900,000 shares of ReNeuron and received proceeds of approximately $512,000 for a realized gain of approximately $400,000.
In February 2009, a warrant issued as part of a June 2004 financing arrangement, was exercised to purchase an aggregate of 164,474 shares of our common stock at $1.90 per share. We issued 164,474 shares of our common stock and received proceeds of approximately $312,500.
In the first quarter of 2009, we sold in aggregate, 3,325,000 shares of our common stock pursuant to the sales agreement we entered into with Cantor, at an average price per share of $2.10 for gross proceeds of approximately $6,999,000. Cantor is paid compensation equal to 5.0% of the gross proceeds pursuant to the terms of the agreement.
In March 2009, we entered into an asset purchase agreement with Stem Cell Sciences Plc (“SCS”) to acquire substantially all of the operating assets and liabilities of SCS (the “Acquisition”). The Acquisition is subject to customary closing conditions, including the approval of the stockholders of SCS, and is expected to close shortly after the SCS extraordinary general meeting scheduled for March 27, 2009. As consideration for the operating assets and liabilities to be acquired, we will issue to SCS, except as provided below, 2,650,000 shares of our common stock, plus waive certain commitments of SCS to repay approximately $715,000 in cash made available by us to SCS for working capital purposes. The actual number of shares delivered to SCS at the closing will depend on the SCS operating subsidiaries having a specified minimum amount of working capital. In connection with the Acquisition, we also entered into a loan facility agreement with SCS pursuant to which we agreed to lend up to $415,000 to SCS for working capital prior to the closing of the Acquisition. Upon closing of the Acquisition, we will waive SCS’ obligations to repay any amounts borrowed by SCS under this loan facility agreement as well as £200,000 (approximately $298,000) previously borrowed by SCS from us in December 2008. The principal amounts owed on both of these loans accrue interest at 8% per annum and such principal amounts and accrued interest will become due and payable on June 23, 2009 if the Acquisition does not occur beforehand.
*****
74
StemCells, Inc.
Notes to Consolidated Financial Statements — (Continued)
QUARTERLY FINANCIAL DATA (unaudited)
(in thousands, except per share amounts)
| 2008 Quarter Ended | ||||||||||||||||||||||||||||||||
| 2007 Quarter Ended | December 31 | September 30 | June 30 | March 31 | ||||||||||||||||||||||||||||
| December 31 | September 30 | June 30 | March 31 | (In thousands, except per share amounts) | ||||||||||||||||||||||||||||
| Total revenue | $ | 30 | $ | 13 | $ | 8 | $ | 6 | $ | 172 | $ | 12 | $ | 30 | $ | 17 | ||||||||||||||||
| Operating expenses(1) | 8,353 | 7,749 | 6,041 | 6,505 | 7,270 | 5,857 | 6,929 | 6,914 | ||||||||||||||||||||||||
| Other income, net | 497 | 582 | 609 | 1,880 | ||||||||||||||||||||||||||||
| Other income (expense), net(2) | (2,985 | ) | 101 | 183 | 352 | |||||||||||||||||||||||||||
| Net loss | (7,825 | ) | (7,154 | ) | (5,424 | ) | (4,620 | ) | (10,082 | ) | (5,744 | ) | (6,716 | ) | (6,545 | ) | ||||||||||||||||
| Basic and diluted loss per share | $ | (0.10 | ) | $ | (0.09 | ) | $ | (0.07 | ) | $ | (0.06 | ) | ||||||||||||||||||||
| Basic and diluted net loss per share | $ | (0.11 | ) | $ | (0.07 | ) | $ | (0.08 | ) | $ | (0.09 | ) | ||||||||||||||||||||
| 2007 Quarter Ended | ||||||||||||||||||||||||||||||||
| 2006 Quarter Ended | December 31 | September 30 | June 30 | March 31 | ||||||||||||||||||||||||||||
| December 31 | September 30 | June 30 | March 31 | (In thousands, except per share amounts) | ||||||||||||||||||||||||||||
| Total revenue | $ | 12 | $ | 18 | $ | 21 | $ | 42 | $ | 30 | $ | 13 | $ | 8 | $ | 6 | ||||||||||||||||
| Operating expenses(1) | 6,583 | 5,581 | 4,775 | 4,525 | 8,353 | 7,749 | 6,041 | 6,505 | ||||||||||||||||||||||||
| Other income, net | 668 | 699 | 765 | 291 | 497 | 582 | 609 | 1,880 | ||||||||||||||||||||||||
| Net loss | (5,903 | ) | (4,863 | ) | (3,989 | ) | (4,193 | ) | (7,826 | ) | (7,154 | ) | (5,424 | ) | (4,619 | ) | ||||||||||||||||
| Basic and diluted (loss) per share | $ | (0.08 | ) | $ | (0.06 | ) | $ | (0.05 | ) | $ | (0.06 | ) | ||||||||||||||||||||
| Basic and diluted net loss per share | $ | (0.10 | ) | $ | (0.09 | ) | $ | (0.07 | ) | $ | (0.06 | ) | ||||||||||||||||||||
| (1) | Includes adjustment of wind-down accrual — see Note | |
| (2) | Other expense, net, for the quarter ended December 31, 2008, includes a loss of $937,241 relating to the change in fair value of our warrant liability — see Note 10, and a $2,082,894 other than temporary impairment of marketable securities — see Note 2. |
6975
| Item 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
| Item 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
The Company’s management, with the participation of its chief executive officer and chief financial officer, evaluated the effectiveness of the design and operation of its disclosure controls and procedures (as defined inRules 13a-15(e) and15d-15(e) under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this annual report. Based on this evaluation, the Company’s principal executive officer and principal financial officer concluded that these disclosure controls and procedures are effective to ensure that the information required to be disclosed in our reports filed or submitted under the Securities Exchange Act of 1934 is recorded, processed, summarized and reported within the requisite time periods, and to provide reasonable assurance that information required to be disclosed by the Company in such reports is accumulated and communicated to the Company’s management, including its chief executive officer and chief financial officer, as appropriate to allow timely decisions regarding required disclosure.
Changes in Internal Controls
There have been no changes in the Company’s internal control over financial reporting during the quarter ended December 31, 2007,2008, that have materially affected, or are reasonably likely to materially affect, the Company’s internal control over financial reporting.
Management’s Report on Internal Control Over Financial Reporting
Management of the Company is responsible for establishing and maintaining adequate internal control over financial reporting. The Company’s management, including its principal executive officer and principal financial officer, assessed the effectiveness of its internal control over financial reporting based on the framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The evaluation of the design and operating effectiveness of internal controlscontrol over financial reporting include among others those policies and procedures that:
| • | Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of the assets of the Company; | |
| • | Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and | |
| • | Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements. |
During the fiscal year 2007,2008, the Company periodically tested the design and operating effectiveness of its internal controls.control over financial reporting. Among other matters, the Company sought in its evaluation to determine whether there were any “significant deficiencies” or “material weakness” in its internal control over financial reporting, or whether it had identified any acts of fraud involving management or other employees.
Based on the above evaluation, the Company’s chief executive officer and chief financial officer have concluded that as of December 31, 2007,2008, the Company’s internal controlscontrol over financial reporting were effective. Nonetheless, it is important to acknowledge that due to its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Therefore, even systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
The Company’s internal control over financial reporting as of December 31, 2008 has been audited by Grant Thornton LLP, an independent registered public accounting firm, as stated in their report below.
7076
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
ON INTERNAL CONTROL OVER FINANCIAL REPORTING
ON INTERNAL CONTROL OVER FINANCIAL REPORTING
Board of Directors and Stockholders
StemCells, Inc.
We have audited StemCells, Inc. (a Delaware corporation) and subsidiary’s (collectively, the “Company”) internal control over financial reporting as of December 31, 2008, based on criteria established inInternal Control — Integrated Frameworkissued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, StemCells, Inc. and subsidiary maintained, in all material respects, effective internal control over financial reporting as of December 31, 2008 based on criteria established inInternal Control — Integrated Frameworkissued by COSO.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of StemCells, Inc. and subsidiary as of December 31, 2008 and 2007, and the related consolidated statements of operations, changes in stockholders’ equity, and cash flows for each of the three years in the period ended December 31, 2008 and our report dated March 11, 2009 expressed an unqualified opinion thereon.
/s/ GRANT THORNTON LLP
San Francisco, California
March 11, 2009
77
| Item 9B. | Other Information |
None
PART III
| Item 10. | DIRECTORS AND EXECUTIVE OFFICERS OF THE REGISTRANT |
Executive Officers
Below are the name, age and principal occupations for the last five years of each executive officer of StemCells, Inc., as of February 29, 2008.28, 2009. All such persons have been elected to serve until their successors are elected and qualified or until their earlier resignation or removal.
| Martin M. McGlynn, President and Chief Executive Officer | Martin M. McGlynn joined the company on January 2001, when he was appointed President and Chief Executive Officer of the company and of its wholly-owned subsidiary, StemCells California, Inc. He was elected to the Board of Directors in February 2001. | |||||
| Ann Tsukamoto, Research and Development | Ann Tsukamoto, Ph.D., joined the company in November 1997 as Senior Director of Scientific Operations; was appointed Vice President, Scientific Operations in June | |||||
| Rodney K.B. Young, Chief Financial Officer and Finance and Administration | Rodney K.B. Young joined the company in September 2005 as Chief Financial Officer and Vice President, Finance. In November 2006 he became CFO and Vice President, Finance and | |||||
| Stewart Craig, Ph.D. Senior Vice President, Development and Operations | 47 | Stewart Craig, Ph.D., joined the company in September 2008 with responsibilities for Development, Manufacturing, Regulatory, Quality Systems and Facilities. From 2005 to 2008, Dr. Craig was Chief Technology Officer and Vice President of Progenitor Cell Therapy, a contract services provider for research, development, manufacture and commercialization of cell-based therapies, prior to which he has held executive positions at Xcyte Therapies, Osiris Therapeutics and SyStemix. | ||||
| Kenneth Stratton, JD General Counsel | 40 | Kenneth Stratton, JD, joined the company in February 2007 as General Counsel, with responsibility for corporate compliance and legal affairs. In March 2008, he assumed responsibilities for the Human Resources function. Prior to StemCells, Mr. Stratton served as Deputy General Counsel for Threshold Pharmaceuticals and as Senior Legal Counsel for Medtronic’s Vascular business unit. |
78
Directors
Below are the name, age and principal occupations for the last five years of each Director of StemCells, Inc., as of February 29, 2008. Directors are elected to staggered three year terms.
| Eric H. Bjerkholt | Eric H. Bjerkholt was elected to the Board of Directors in March 2004. Mr. Bjerkholt joined Sunesis Pharmaceuticals, Inc., in 2004 as Senior Vice President and Chief Financial Officer. Since February 2007, he has served as Senior Vice President, Corporate Development and Finance, and Chief Financial Officer. From 2002 to 2004, Mr. Bjerkholt was Senior Vice President and Chief Financial Officer at IntraBiotics Pharmaceuticals, Inc. | |||||
| Ricardo B. Levy, Ph.D. | Ricardo B. Levy, Ph.D. was elected to the Board of Directors in September 2001. He currently serves on several boards of directors. | |||||
| Martin M. McGlynn | Martin M. McGlynn was elected to the Board of Directors in February 2001. He is President and Chief Executive Officer of the Company, a position he has held since January 2001. | |||||
71
| Roger Perlmutter, M.D., Ph.D. | Roger M. Perlmutter, M.D., Ph.D., was elected to the Board of Directors in December 2000. He is Executive Vice President, Research and Development, of Amgen, Inc., a position he has held since January 2001. | |||||
| John J. Schwartz, Ph.D. | John J. Schwartz, Ph.D., was elected to the Board of Directors in December 1998 and was elected Chairman of the Board at the same time. He is currently President of Quantum Strategies Management Company. | |||||
| Irving Weissman, M.D. | Irving L. Weissman, M.D., was elected to the Board of Directors in September 1997. He is the Virginia and Daniel K. Ludwig Professor of Cancer Research, Professor of Pathology and Professor of Developmental Biology at Stanford. |
Certain other information required by this Item regarding our officers, Directors, and corporate governance is incorporated herein by reference to the information appearing under the headings “Information About Our Directors” and “Information About Ownership of Our Common Stock” in our definitive proxy statement to be filed with the Securities and Exchange Commission within 120 days of December 31, 20072008 (the “2008“2009 Proxy Statement”).
| Item 11. | EXECUTIVE COMPENSATION |
The information required by this Item is incorporated by reference from Item 5 of this Annual Report onForm 10-K and our Proxy Statement for the 20082009 Annual Meeting of Stockholders.
Item 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The information required by this Item is incorporated by reference from Item 5 of this Annual Report onForm 10-K and from our Proxy Statement for the 20082009 Annual Meeting of Stockholders.
| Item 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS |
The information required by this Item is incorporated by reference from our Proxy Statement for the 20082009 Annual Meeting of Stockholders.
79
| Item 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by this Item is incorporated by reference from our Proxy Statement for the 20082009 Annual Meeting of Stockholders.
PART IV
| Item 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
| (a) | The following documents are included as part of this Annual Report on Form10-K. |
(1) Financial Statements:
The financial statements filed as part of this Report are listed and indexed under Item 8 above.
(2) Financial Statement Schedules:
Schedules are not included herein because they are not applicable or the required information appears in the Financial Statements or Notes thereto.
(3) Exhibits.
The documents set forth below are filed herewith or incorporated by reference to the location indicated.
Exhibit No. | Title or Description | |||
| 3 | .1- | Restated Certificate of Incorporation of the Registrant | ||
| 3 | .2-- | Amended and Restated By-Laws of the Registrant | ||
| 4 | .1ˆˆ | Specimen common stock Certificate | ||
| 4 | .2{*} | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .3XXXX | Warrant to Purchase common stock — Cantor Fitzgerald & Co. | ||
| 4 | .4&2 | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .5&4 | Form of Warrant Certificate issued to a certain purchasers of the Registrant’s common stock in November 2008 | ||
| 10 | .1 | Form of at-will Employment Agreement between the Registrant and most of its employees | ||
| 10 | .2* | Form of Agreement for Consulting Services between the Registrant and members of its Scientific Advisory Board | ||
| 10 | .3 | Form of Nondisclosure Agreement between the Registrant and its Contractors | ||
| 10 | .4* | 1992 Equity Incentive Plan | ||
| 10 | .5* | 1992 Stock Option Plan for Non-Employee Directors | ||
| 10 | .6+ | Research Agreement, dated as of March 16, 1994, between NeuroSpheres, Ltd. and Registrant | ||
| 10 | .7+ | Lease Agreement between the Registrant and Rhode Island Industrial Facilities Corporation, dated as of August 1, 1992 | ||
| 10 | .8+ | First Amendment to Lease Agreement between Registrant and The Rhode Island Industrial Facilities Corporation dated as of September 15, 1994 | ||
| 10 | .9# | Lease Agreement, dated as of November 21, 1997, by and between Hub RI Properties Trust, as Landlord, and CytoTherapeutics, Inc., as Tenant | ||
| 10 | .10! | Consulting Agreement, dated as of September 25, 1997, between Dr. Irving Weissman and the Registrant | ||
| 10 | .11!!! | StemCells, Inc. 1996 Stock Option Plan | ||
| 10 | .12!!! | 1997 StemCells Research Stock Option Plan (the “1997 Plan”) | ||
| 10 | .13!!! | Form of Performance-Based Incentive Option Agreement issued under the 1997 Plan | ||
| 10 | .14XX | License Agreement, dated as of October 30, 2000, between the Registrant and NeuroSpheres Ltd. | ||
| 10 | .15XX | Letter Agreement, dated January 2, 2001, between the Registrant and Martin McGlynn | ||
72
80
Exhibit No. | Title or Description | |||
| 3 | .1- | Restated Certificate of Incorporation of the Registrant | ||
| 3 | .2-- | Amended and Restated By-Laws of the Registrant | ||
| 4 | .1^ ^ | Specimen common stock Certificate | ||
| 4 | .2++ | Form of Warrant Certificate issued to a certain purchaser of the Registrant’s common stock in April 1995 | ||
| 4 | .3X | common stock Purchase Warrant | ||
| 4 | .4X | Callable Warrant | ||
| 4 | .5XXX | Callable Warrant, dated June 21, 2001, issued to Millennium Partners, L.P. | ||
| 4 | .6XXX | Common stock Purchase Warrant, Class A, dated June 21, 2001, issued to Millennium Partners, L.P. | ||
| 4 | .7{*} | Certificate of Designations of the Powers, Preferences and Relative, Participating, Optional and other Special Rights of Preferred Stock and Qualifications, Limitations and Restrictions Thereof of 3% Cumulative Convertible Preferred Stock for StemCells, Inc. | ||
| 4 | .8{*} | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .9XXXX | Warrant to Purchase common stock — Cantor Fitzgerald & Co. | ||
| 4 | .10&& | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 10 | .1* | Form of at-will Employment Agreement between the Registrant and most of its employees | ||
| 10 | .2* | Form of Agreement for Consulting Services between the Registrant and members of its Scientific Advisory Board | ||
| 10 | .3* | Form of Nondisclosure Agreement between the Registrant and its Contractors | ||
| 10 | .4* | 1992 Equity Incentive Plan | ||
| 10 | .5* | 1992 Stock Option Plan for Non-Employee Directors | ||
| 10 | .7+ | Research Agreement, dated as of March 16, 1994, between NeuroSpheres, Ltd. and Registrant | ||
| 10 | .8+ | Lease Agreement between the Registrant and Rhode Island Industrial Facilities Corporation, dated as of August 1, 1992 | ||
| 10 | .9+ | First Amendment to Lease Agreement between Registrant and The Rhode Island Industrial Facilities Corporation dated as of September 15, 1994 | ||
| 10 | .10# | Lease Agreement, dated as of November 21, 1997, by and between Hub RI Properties Trust, as Landlord, and CytoTherapeutics, Inc., as Tenant | ||
| 10 | .11!! | CTI individual stockholders option agreement, dated as of July 10, 1996, among the Company and the individuals listed therein | ||
| 10 | .12! | Consulting Agreement, dated as of September 25, 1997, between Dr. Irving Weissman and the Registrant | ||
| 10 | .13!!! | StemCells, Inc. 1996 Stock Option Plan | ||
| 10 | .14!!! | 1997 StemCells Research Stock Option Plan (the “1997 Plan”) | ||
| 10 | .15!!! | Form of Performance-Based Incentive Option Agreement issued under the 1997 Plan | ||
| 10 | .16$** | License Agreement, dated as of October 27, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .17$** | License Agreement, dated as of October 27, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .18$** | License Agreement, dated as of November 20, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .19++** | License Agreement, dated as of June 199,9 between The Scripps Research Institute and the Registrant | ||
| 10 | .20++** | License Agreement, dated as of June 1999, between The Scripps Research Institute and the Registrant | ||
| 10 | .21XX | License Agreement, dated as of October 30, 2000, between the Registrant and NeuroSpheres Ltd. | ||
| 10 | .22XX | Letter Agreement, dated January 2, 2001, between the Registrant and Martin McGlynn | ||
| 10 | .23XX | Lease, dated February 1, 2001, between the Board of Trustees of Stanford University and the Registrant | ||
| 10 | .24$$ | 2001 Equity Incentive Plan | ||
| 10 | .25& | Agreement, dated as of April 9, 2003, between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .26&& | Form of Registration Rights Agreement between the Registrant and Riverview Group, L.L.C. | ||
73
Exhibit No. | Title or Description | |||
| 10 | .27&&& | Securities Purchase Agreement, dated as of May 7, 2003, between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .28% | Securities Purchase Agreement, dated as of December 9, 2003, between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .29^ ^ ^ | Form of Securities Purchase Agreement, dated as of June 16, 2004, between the Registrant and certain Purchasers parties thereto | ||
| 10 | .30^ ^ ^ | Form of Warrant | ||
| 10 | .31^ ^ ^ ^ | Amended and Restated 2004 Equity Incentive Plan of the Registrant | ||
| 10 | .32§ | License Agreement, dated as of July 1, 2005, between the Registrant and ReNeuron Limited | ||
| 10 | .33§§ | Letter Agreement, effective as of September 6, 2005, between the Registrant and Rodney K.B. Young | ||
| 10 | .34§§ | Consulting Agreement, effective as of September 6, 2005, between the Registrant and Judi R. Lum | ||
| 10 | .35^ | Sales Agreement with Cantor Fitzgerald | ||
| 10 | .36XX | Side Letter, dated March 17, 2001, between the Company and Oleh S. Hnatiuk regarding NeuroSpheres License Agreement, dated October 30, 2000 | ||
| 10 | .37 | Note Purchase Agreement with Progenitor Cell Therapy, LLC, dated December 3, 2007. | ||
| 10 | .38@ | License Agreement, dated April 1, 1997, by and among Registrant, NeuroSpheres Ltd. and NeuroSpheres Holdings Ltd. | ||
| 14 | .1%% | Code of Ethics | ||
| 21 | X | Subsidiaries of the Registrant | ||
| 23 | .1 | Consent of Grant Thornton, LLP , Independent Registered Public Accounting Firm | ||
| 31 | .1 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 31 | .2 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| 32 | .1 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 32 | .2 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
Exhibit No. | Title or Description | |||
| 10 | .16XX | Lease, dated February 1, 2001, between the Board of Trustees of Stanford University and the Registrant | ||
| 10 | .17$$ | 2001 Equity Incentive Plan | ||
| 10 | .18ˆˆˆ | Form of Securities Purchase Agreement, dated as of June 16, 2004, between the Registrant and certain Purchasers parties thereto | ||
| 10 | .19ˆˆˆ | Form of Warrant | ||
| 10 | .20ˆˆˆˆ | Amended and Restated 2004 Equity Incentive Plan of the Registrant | ||
| 10 | .21§ | License Agreement, dated as of July 1, 2005, between the Registrant and ReNeuron Limited | ||
| 10 | .22§§ | Letter Agreement, effective as of September 6, 2005, between the Registrant and Rodney K.B. Young | ||
| 10 | .23XX | Side Letter, dated March 17, 2001, between the Company and Oleh S. Hnatiuk regarding NeuroSpheres License Agreement, dated October 30, 2000 | ||
| 10 | .24@ | License Agreement, dated April 1, 1997, by and among Registrant, NeuroSpheres Ltd. and NeuroSpheres Holdings Ltd. | ||
| 10 | .25§§§ | Indemnification Agreement, dated July 9, 2008, by and between registrant and NeuroSpheres Holdings, LTD. | ||
| 10 | .26 | Facility Agreement, dated December 23, 2008, by and among registrant and Stem Cell Sciences Plc | ||
| 10 | .27 | Second Facility Agreement, dated March 1, 2009, by and among registrant, Stem Cell Sciences Plc and Stem Cell Sciences Holdings Limited | ||
| 10 | .28 | Asset Purchase Agreement, dated March 1, 2009, by and between registrant and Stem Cell Sciences Plc | ||
| 14 | .1 | Code of Ethics | ||
| 21 | X | Subsidiaries of the Registrant | ||
| 23 | .1 | Consent of Grant Thornton, LLP , Independent Registered Public Accounting Firm | ||
| 31 | .1 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 31 | .2 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| 32 | .1 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 32 | .2 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| ! | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 1997 and filed on November 14, 1997. | |
| !! | Previously filed with the Commission as an Exhibit to and incorporated by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 1996. | |
| !!! | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-8, FileNo. 333-37313. | |
| $ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s annual report onForm 10-K for the fiscal year ended December 31, 1998 and filed on March 31, 1999. | |
| $$ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s definitive proxy statement filed May 1, 2001. | |
| % | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on December 10, 2003. |
81
| %% | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2003 | |
| &1 | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on April 15, 2003. | |
| &2 | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 13, 2003. | |
| &3 | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 15, 2003. | |
| &4 | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on November 12, 2008. | |
| * | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, Registration Statement onForm S-1, FileNo. 33-45739. | |
| ** | Confidential treatment requested as to certain portions. The term “confidential treatment” and the mark “**” as used throughout the indicated Exhibits mean that material has been omitted and separately filed with the Commission. | |
| ˆ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on December 29, 2006. | |
| ˆˆ | Previously filed with the Commission as an Exhibit to, and incorporated by reference to, the Registrant’s Registration Statement onForm S-3, FileNo. 333-151891. | |
| ˆˆˆ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on June 17, 2004. | |
| ˆˆˆˆ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrants Registration Statement onForm S-8, FileNo. 333-118263. | |
| {*} | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on December 7, 2001. | |
| + | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 33-85494. | |
| ++ | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 33-91228. |
82
| - | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2006 and filed on March 15, 2007. | |
| -- | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 7, 2007. | |
| § | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended June 30, 2005. | |
| §§ | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on September 7, 2005. | |
| §§§ | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 2008 | |
| X | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 333-45496. | |
| XX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2000 and filed on April 2, 2001. | |
| XXX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement filed onForm S-1 as amended toForm S-3, FileNo. 333-61726. | |
| XXXX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement filed onForm S-3, FileNo. 333-75806. | |
| @ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K/A for the fiscal year ended December 31, 2006 and filed on April 1, 1997. | |
| # | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s annual report onForm 10-K for the fiscal year ended December 31, 1997 and filed on March 30, 1998. |
83
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
STEMCELLS, INC.
| By: | /s/ MARTIN MCGLYNN |
Martin McGlynn
PRESIDENT AND CHIEF
EXECUTIVE OFFICER
Dated: March 13, 2009
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
Signature | Capacity | Date | ||||
/s/ Martin McGlynn Martin McGlynn | President and Chief Executive Officer and Director (principal executive officer) | March 13, 2009 | ||||
/s/ Rodney K.B. Young Rodney K.B. Young | Chief Financial Officer (principal financial officer) | March 13, 2009 | ||||
/s/ George Koshy George Koshy | Chief Accounting Officer (principal accounting officer) | March 13, 2009 | ||||
/s/ Eric Bjerkholt Eric Bjerkholt | Director | March 13, 2009 | ||||
/s/ Ricardo B. Levy, Ph.D Ricardo B. Levy, Ph.D. | Director | March 13, 2009 | ||||
/s/ Roger M. Perlmutter, M.D. Roger M. Perlmutter, M.D. | Director | March 13, 2009 | ||||
/s/ John J. Schwartz, Ph. D. John J. Schwartz, Ph.D. | Director, Chairman of the Board | March 13, 2009 | ||||
/s/ Irving L. Weissman, M.D. Irving L. Weissman, M.D. | Director | March 13, 2009 | ||||
84
Exhibit Index
Exhibit No. | Title or Description | |||
| 3 | .1- | Restated Certificate of Incorporation of the Registrant | ||
| 3 | .2-- | Amended and Restated By-Laws of the Registrant | ||
| 4 | .1ˆˆ | Specimen common stock Certificate | ||
| 4 | .2{*} | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .3XXXX | Warrant to Purchase common stock — Cantor Fitzgerald & Co. | ||
| 4 | .4&2 | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .5&4 | Form of Warrant Certificate issued to a certain purchasers of the Registrant’s common stock in November 2008 | ||
| 10 | .1 | Form of at-will Employment Agreement between the Registrant and most of its employees | ||
| 10 | .2* | Form of Agreement for Consulting Services between the Registrant and members of its Scientific Advisory Board | ||
| 10 | .3 | Form of Nondisclosure Agreement between the Registrant and its Contractors | ||
| 10 | .4* | 1992 Equity Incentive Plan | ||
| 10 | .5* | 1992 Stock Option Plan for Non-Employee Directors | ||
| 10 | .6+ | Research Agreement, dated as of March 16, 1994, between NeuroSpheres, Ltd. and Registrant | ||
| 10 | .7+ | Lease Agreement between the Registrant and Rhode Island Industrial Facilities Corporation, dated as of August 1, 1992 | ||
| 10 | .8+ | First Amendment to Lease Agreement between Registrant and The Rhode Island Industrial Facilities Corporation dated as of September 15, 1994 | ||
| 10 | .9# | Lease Agreement, dated as of November 21, 1997, by and between Hub RI Properties Trust, as Landlord, and CytoTherapeutics, Inc., as Tenant | ||
| 10 | .10! | Consulting Agreement, dated as of September 25, 1997, between Dr. Irving Weissman and the Registrant | ||
| 10 | .11!!! | StemCells, Inc. 1996 Stock Option Plan | ||
| 10 | .12!!! | 1997 StemCells Research Stock Option Plan (the “1997 Plan”) | ||
| 10 | .13!!! | Form of Performance-Based Incentive Option Agreement issued under the 1997 Plan | ||
| 10 | .14XX | License Agreement, dated as of October 30, 2000, between the Registrant and NeuroSpheres Ltd. | ||
| 10 | .15XX | Letter Agreement, dated January 2, 2001, between the Registrant and Martin McGlynn | ||
| 10 | .16XX | Lease, dated February 1, 2001, between the Board of Trustees of Stanford University and the Registrant | ||
| 10 | .17$$ | 2001 Equity Incentive Plan | ||
| 10 | .18ˆˆˆ | Form of Securities Purchase Agreement, dated as of June 16, 2004, between the Registrant and certain Purchasers parties thereto | ||
| 10 | .19ˆˆˆ | Form of Warrant | ||
| 10 | .20ˆˆˆˆ | Amended and Restated 2004 Equity Incentive Plan of the Registrant | ||
| 10 | .21§ | License Agreement, dated as of July 1, 2005, between the Registrant and ReNeuron Limited | ||
| 10 | .22§§ | Letter Agreement, effective as of September 6, 2005, between the Registrant and Rodney K.B. Young | ||
| 10 | .23XX | Side Letter, dated March 17, 2001, between the Company and Oleh S. Hnatiuk regarding NeuroSpheres License Agreement, dated October 30, 2000 | ||
| 10 | .24@ | License Agreement, dated April 1, 1997, by and among Registrant, NeuroSpheres Ltd. and NeuroSpheres Holdings Ltd. | ||
| 10 | .25§§§ | Indemnification Agreement, dated July 9, 2008, by and between registrant and NeuroSpheres Holdings, LTD. | ||
| 10 | .26 | Facility Agreement, dated December 23, 2008, by and among registrant and Stem Cell Sciences Plc | ||
Exhibit No. | Title or Description | |||
| 10 | .27 | Second Facility Agreement, dated March 1, 2009, by and among registrant, Stem Cell Sciences Plc and Stem Cell Sciences Holdings Limited | ||
| 10 | .28 | Asset Purchase Agreement, dated March 1, 2009, by and between registrant and Stem Cell Sciences Plc | ||
| 14 | .1 | Code of Ethics | ||
| 21 | X | Subsidiaries of the Registrant | ||
| 23 | .1 | Consent of Grant Thornton, LLP , Independent Registered Public Accounting Firm | ||
| 31 | .1 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 31 | .2 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| 32 | .1 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 32 | .2 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| ! | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 1997 and filed on November 14, 1997. | |
| !! | Previously filed with the Commission as an Exhibit to and incorporated by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 1996. | |
| !!! | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-8, FileNo. 333-37313. | |
| $ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s annual report onForm 10-K for the fiscal year ended December 31, 1998 and filed on March 31, 1999. | |
| $$ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s definitive proxy statement filed May 1, 2001. | |
| % | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on December 10, 2003. | |
| %% | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2003 | |
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on April 15, 2003. | ||
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 13, 2003. |
74
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 15, 2003. | ||
| &4 | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on November 12, 2008. | |
| * | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, Registration Statement onForm S-1, FileNo. 33-45739. |
| ** | Confidential treatment requested as to certain portions. The term “confidential treatment” and the mark “**” as used throughout the indicated Exhibits mean that material has been omitted and separately filed with the Commission. | |
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on December 29, 2006. | ||
| Previously filed with the Commission as an Exhibit to, and incorporated by reference to, the Registrant’s Registration Statement onForm S-3, FileNo. | ||
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on June 17, 2004. | ||
| Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrants Registration Statement onForm S-8, FileNo. 333-118263. | ||
| {*} | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on December 7, 2001. | |
| + | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 33-85494. | |
| ++ | Previously filed with the Commission as Exhibits to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 33-91228. | |
| - | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2006 and filed on March 15, 2007. | |
| -- | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s current report onForm 8-K on May 7, 2007. | |
| § | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended June 30, 2005. | |
| §§ | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s current report onForm 8-K filed on September 7, 2005. | |
| §§§ | Previously filed with the Commission as an Exhibit to and incorporated herein by reference to, the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 2008. | |
| X | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement onForm S-1, FileNo. 333-45496. |
| XX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K for the fiscal year ended December 31, 2000 and filed on April 2, 2001. | |
| XXX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement filed onForm S-1 as amended toForm S-3, FileNo.333-61726. | |
| XXXX | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Registration Statement filed onForm S-3, FileNo. 333-75806. | |
| @ | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s Annual Report onForm 10-K/A for the fiscal year ended December 31, 2006 and filed on April 1, 1997. | |
| # | Previously filed with the Commission as an Exhibit to, and incorporated herein by reference to, the Registrant’s annual report onForm 10-K for the fiscal year ended December 31, 1997 and filed on March 30, 1998. |
75
76
Exhibit No. | Title or Description | |||
| 3 | .1- | Restated Certificate of Incorporation of the Registrant | ||
| 3 | .2-- | Amended and Restated By-Laws of the Registrant | ||
| 4 | .1^ ^ | Specimen common stock Certificate | ||
| 4 | .2++ | Form of Warrant Certificate issued to a certain purchaser of the Registrant’s common stock in April 1995 | ||
| 4 | .3X | common stock Purchase Warrant | ||
| 4 | .4X | Callable Warrant | ||
| 4 | .5XXX | Callable Warrant, dated June 21, 2001, issued to Millennium Partners, L.P. | ||
| 4 | .6XXX | Common stock Purchase Warrant, Class A, dated June 21, 2001, issued to Millennium Partners, L.P. | ||
| 4 | .7{*} | Certificate of Designations of the Powers, Preferences and Relative, Participating, Optional and other Special Rights of Preferred Stock and Qualifications, Limitations and Restrictions Thereof of 3% Cumulative Convertible Preferred Stock for StemCells, Inc. | ||
| 4 | .8{*} | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 4 | .9XXXX | Warrant to Purchase common stock — Cantor Fitzgerald & Co. | ||
| 4 | .10&& | Warrant to Purchase common stock — Riverview Group, LLC | ||
| 10 | .1* | Form of at-will Employment Agreement between the Registrant and most of its employees | ||
| 10 | .2* | Form of Agreement for Consulting Services between the Registrant and members of its Scientific Advisory Board | ||
| 10 | .3* | Form of Nondisclosure Agreement between the Registrant and its Contractors | ||
| 10 | .4* | 1992 Equity Incentive Plan | ||
| 10 | .5* | 1992 Stock Option Plan for Non-Employee Directors | ||
| 10 | .7+ | Research Agreement, dated as of March 16, 1994, between NeuroSpheres, Ltd. and Registrant | ||
| 10 | .8+ | Lease Agreement between the Registrant and Rhode Island Industrial Facilities Corporation, dated as of August 1, 1992 | ||
| 10 | .9+ | First Amendment to Lease Agreement between Registrant and The Rhode Island Industrial Facilities Corporation dated as of September 15, 1994 | ||
| 10 | .10# | Lease Agreement, dated as of November 21, 1997, by and between Hub RI Properties Trust, as Landlord, and CytoTherapeutics, Inc., as Tenant | ||
| 10 | .11!! | CTI individual stockholders option agreement, dated as of July 10, 1996, among the Company and the individuals listed therein | ||
| 10 | .12! | Consulting Agreement, dated as of September 25, 1997, between Dr. Irving Weissman and the Registrant | ||
| 10 | .13!!! | StemCells, Inc. 1996 Stock Option Plan | ||
| 10 | .14!!! | 1997 StemCells Research Stock Option Plan (the “1997 Plan”) | ||
| 10 | .15!!! | Form of Performance-Based Incentive Option Agreement issued under the 1997 Plan | ||
| 10 | .16$** | License Agreement, dated as of October 27, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .17$** | License Agreement, dated as of October 27, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .18$** | License Agreement, dated as of November 20, 1998, between The Scripps Research Institute and the Registrant | ||
| 10 | .19++** | License Agreement, dated as of June 199,9 between The Scripps Research Institute and the Registrant | ||
| 10 | .20++** | License Agreement, dated as of June 1999, between The Scripps Research Institute and the Registrant | ||
| 10 | .21XX | License Agreement, dated as of October 30, 2000, between the Registrant and NeuroSpheres Ltd. | ||
| 10 | .22XX | Letter Agreement, dated January 2, 2001, between the Registrant and Martin McGlynn | ||
| 10 | .23XX | Lease, dated February 1, 2001, between the Board of Trustees of Stanford University and the Registrant | ||
| 10 | .24$$ | 2001 Equity Incentive Plan | ||
| 10 | .25& | Agreement, dated as of April 9, 2003, between the Registrant and Riverview Group, L.L.C. | ||
77
Exhibit No. | Title or Description | |||
| 10 | .26&& | Form of Registration Rights Agreement between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .27&&& | Securities Purchase Agreement, dated as of May 7, 2003, between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .28% | Securities Purchase Agreement, dated as of December 9, 2003, between the Registrant and Riverview Group, L.L.C. | ||
| 10 | .29^ ^ ^ | Form of Securities Purchase Agreement, dated as of June 16, 2004, between the Registrant and certain Purchasers parties thereto | ||
| 10 | .30^ ^ ^ | Form of Warrant | ||
| 10 | .31^ ^ ^ ^ | Amended and Restated 2004 Equity Incentive Plan of the Registrant | ||
| 10 | .32§ | License Agreement, dated as of July 1, 2005, between the Registrant and ReNeuron Limited | ||
| 10 | .33§§ | Letter Agreement, effective as of September 6, 2005, between the Registrant and Rodney K.B. Young | ||
| 10 | .34§§ | Consulting Agreement, effective as of September 6, 2005, between the Registrant and Judi R. Lum | ||
| 10 | .35^ | Sales Agreement with Cantor Fitzgerald | ||
| 10 | .36XX | Side Letter, dated March 17, 2001, between the Company and Oleh S. Hnatiuk regarding NeuroSpheres License Agreement, dated October 30, 2000 | ||
| 10 | .37 | Note Purchase Agreement with Progenitor Cell Therapy, LLC, dated December 3, 2007. | ||
| 10 | .38@ | License Agreement, dated April 1, 1997, by and among Registrant, NeuroSpheres Ltd. and NeuroSpheres Holdings Ltd. | ||
| 14 | .1%% | Code of Ethics | ||
| 21 | X | Subsidiaries of the Registrant | ||
| 23 | .1 | Consent of Grant Thornton, LLP , Independent Registered Public Accounting Firm | ||
| 31 | .1 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 31 | .2 | Certification Pursuant to Securities Exchange ActRule 13(a)-14(a), as Adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
| 32 | .1 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Martin McGlynn, Chief Executive Officer) | ||
| 32 | .2 | Certification Pursuant to 18 U.S.C. Section 1350, As Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Rodney K.B. Young, Chief Financial Officer) | ||
78
79