UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Washington, D.C. 20549
Form 10-K
| (Mark One) | ||
þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the fiscal year ended December 31, | ||
-OR- | ||
o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the transition period from to | ||
Commission File No. 001-33958
RXi PHARMACEUTICALS CORPORATION
(Exact name of registrant as specified in its charter)
| Delaware | ||
(State or other jurisdiction of incorporation or organization) | 20-8099512 (I.R.S. Employer Identification Number) | |
60 Prescott Street Worcester, Massachusetts (Address of principal executive offices) | 01605 (Zip Code) |
Registrant’s telephone number, including area code:
(508) 767-3861
SECURITIES REGISTERED PURSUANT TO SECTION 12(b) OF THE ACT:
Title of Each Class | Name of Exchange on Which Registered | |
| Common Stock, $0.0001 Par Value Per Share | NASDAQ Capital Market |
SECURITIES REGISTERED PURSUANT TO SECTION 12(g) OF THE ACT:
None
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No þ
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No þ
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 ofRegulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of thisForm 10-K or any amendment to thisForm 10-K. Yes o No þ
Indicate by check mark whether the registrant has submitted electronically and posted on it corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 ofRegulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter time that the registrant was required to submit and post such files). Yes o No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” inRule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer o | Accelerated filer o | Non-accelerated filer o | Smaller reporting company þ |
(Do not check if a smaller reporting company)Smaller reporting company þ
Indicate by check mark whether the registrant is a shell company (as defined inRule 12b-2 of the Act). Yes o No þ
The aggregate market value of the voting Common Stock held by non-affiliates of the registrant, based on the last sale price of the registrant’s Common Stock at the close of business on June 30, 2009,2010, was $34,131,897.$39,725,083
As of March 29, 2010,April 14, 2011 the registrant had 18,281,12524,995,428 shares of Common Stock, $0.0001 par value per share, outstanding.
DOCUMENTS INCORPORATED BY REFERENCE:
Portions of the registrant’s definitive proxy statement for its 20102011 annual meeting of stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission not later than 120 days after the registrant’s fiscal year end of December 31, 2009,2010, are incorporated by reference into thisForm 10-K.
RXi PHARMACEUTICALS CORPORATION
FORM 10-K — FISCAL YEAR ENDED DECEMBER 31, 20092010
Table of Contents
All references in thisForm 10-K to “RXi,” the “Company,” “we,” “us,” or “our” mean RXi Pharmaceuticals Corporation, unless we state otherwise or the context otherwise requires.
i
| ITEM 1. | BUSINESS |
Overview
We were incorporated as Argonaut Pharmaceuticals, Inc. in Delaware on April 3, 2006, changed our name to RXi Pharmaceuticals Corporation on November 28, 2006, and began operations in January 2007. Our principal executive offices are located at 60 Prescott Street, Worcester, MA 01605, and our phone number is(508) 767-3861. As described below under “Recent Developments,” on April 13, 2011, we acquired Apthera, Inc. (“Apthera”), a private biotechnology company. In connection with the Apthera acquisition, we issued 4,974,090 shares of our common stock to former stockholders of Apthera and agreed to make certain contingent payments to such stockholders upon the achievement of certain milestones. Unless otherwise specified, the information contained in this annual report on Form 10-K gives effect to the acquisition of Apthera.
RXi is a biotechnology company focussed on discovering, developing and commercializing innovative therapies addressing major unmet medical needs using RNAi-targeted and immunotherapy technologies. We are a discovery-stage biopharmaceutical company pursuing (1) cancer therapies utilizing peptide-based immunotherapy products, including our main product candidate NeuVaxtm, for the treatment of various cancers and (2) proprietary therapeutics based on RNA interference, or “RNAi”, a naturally occurring cellular mechanism that has the potential to effectively and selectively interfere with, or “silence”, expression of targeted disease-associated genes. It is widely accepted by the scientific community that this specific silencing can be used to potentially treat human diseases by “turning off” genes that lead to disease. While no therapeutic RNAi products have been approved for marketing to date, there has been significant growth in the field of RNAi development and potential therapeutic applications in this field.
| • | Immunotherapy Products. Our main product candidate is NeuVax, which is a peptide-based immunotherapy to reduce the recurrence of breast cancer inlow-to-intermediate HER2-positive breast cancer patients not eligible for Herceptin®. We expect NeuVax to enter Phase III clinical trials in this breast cancer patient population during the first half of 2012 if we are able to satisfy certain United States Food and Drug Administration (“FDA”) information requirements to be released from a clinical hold to commence the trial. In addition, based on our clinical trials, we believe that NeuVax has the potential to treat other cancers, including prostate, bladder and ovarian cancers. | |
| • | RNAi Products. We believe that certain human diseases can potentially be treated by silencing targeted genes that lead to disease. While no therapeutic RNAi products have been approved by the FDA to date, there has been significant interest and growth in the field of RNAi therapeutic development. This growth is driven by the potential ability to use RNAi to rapidly develop lead compounds that specifically and selectively inhibit a target gene, many of which are thought to be undruggable by other modalities. RXI-109, our first RNAi product candidate, is a dermal anti-scarring therapy that targets CTGF (connective tissue growth factor). We are currently working towards filing an investigational new drug application (“IND”) for RXI-109 in the second half of 2011 and commencing a Phase I clinical trial in the first half of 2012. We intend to maintain our core RNAi discovery and development capability to advance current collaborations, as well as enable alliances. We believe that RXI-109 may be able to treat other indications, including pulmonary fibrosis, liver fibrosis, acute spinal injury, ocular scarring and restinosis. |
1
The chart below summarizes the current status of our drug development programs, with the dark shading indicating completed stages of development and the light shading indicating development activities we intend to prioritize in the near-term.
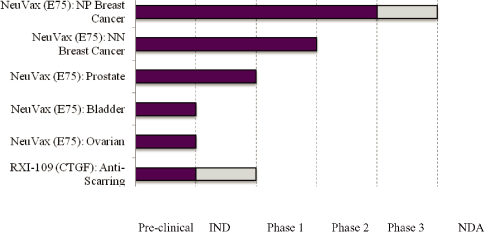
Our Therapeutic Programs
Immunotherapy Program
We are developing a pipeline of immunotherapy product candidates for the treatment of various cancers based on the E75 peptide, the most advanced of which is NeuVax, which is targeted at preventing the recurrence of breast cancer. NeuVax has had positive Phase I/II clinical trial results for the prevention of breast cancer recurrence in patients who have had breast cancer and received the standard of care treatment (e.g., surgery, chemotherapy, radiotherapy and hormonal therapy). We intend to conduct a Phase III clinical trial of NeuVax for the prevention of breast cancer recurrence in early-stage (node-positive) HER2-positive breast cancer patients. We project that this Phase III clinical trial will begin in the first half of 2012 subject to satisfying certain FDA information requirements to be released from a clinical hold to commence the trial.
NeuVax is an immunotherapy that stimulates the immune system to actively seek out and selectively kill cancer cells. NeuVax directs “killer” T-cells to target and destroy cancer cells that express HER2/neu, a protein associated with epithelial tumors in breast, ovarian, pancreatic, colon, bladder and prostate cancers. NeuVax is comprised of two components: a HER2/neu-derived peptide called E75 and the immune adjuvant GM-CSF. E75 is a small9-amino acid sequence that is immunogenic (produces an immune response) and GM-CSF is a commercially available protein that acts to stimulate and activate components of the immune system such as macrophages and dendritic cells.
NeuVax has been shown to be most effective in patients withlow-to-intermediate HER2/neu expressors with HLA type A2+ or A3+. We believe that approximately 25,000 of the approximately 200,000 women diagnosed with breast cancer in the U.S. each year meet these criteria. We believe that NeuVax’s specificity provides for a highly targeted therapy to prevent breast cancer recurrence for a selected subset of breast cancer patients and we believe it will increase the chance of a successful treatment outcome for these patients.
In addition to the lead early-stage breast cancer indication for NeuVax, we are pursuing additional therapeutic indications for NeuVax that are currently in Phase I/II clinical trials. Under our IND, open protocols for the treatment of prostate cancer, ovarian cancer and bladder cancer exist for patient populations with the same general criteria for eligibility as in breast cancer (i.e.,early-stage disease and adjuvant treatment setting after surgery with immunologic competence). We may also explore whether NeuVax provides clinical benefits in other areas, such as a prophylactic vaccine against breast cancer occurrence in healthy women with a high likelihood for developing breast cancer based on genetic assays or biomarkers and a strong positive familial history of breast cancer. Clinical trials conducted on NeuVax have providedproof-of-principle data in
2
early-stage node-negative breast cancer, although such data is preliminary and not statistically significant as the trials were not designed to provide statistically significant efficacy data. Both the early-stage node-negative breast cancer indication and the high risk patient indication are longer-term areas of interest that we currently expect we will only explore with support from corporate partners for these programs.
RNAi Program
By utilizing our expertise in RNAi and the comprehensive RNAi therapeutic platform that we have established, we believe we will be able to discover and develop lead compounds and progress them into and through clinical development for potential commercialization more efficiently than traditional drug development approaches.
commercialization. Our proprietary therapeutic platform is comprised of two main components:
| • | Novel RNAi Compounds,referred to as rxRNAtm compounds, that are distinct from, and we believe convey significant advantages over, classic siRNA (conventionally-designed “small interfering RNA” compounds), and offer many of the properties that we believe are important to the clinical development of RNAi-based drugs. We have developed a number of unique forms of rxRNA compounds, all of which have been shown | |
| • | Advanced Delivery Technologiesthat |
We are currently focusing our internal therapeutic development areas in fibrosis-dermal anti-scarring. RXI-109, our first RNAi product candidate, is a dermal anti-scarring therapy that targets CTGF (connective tissue growth factor). Approximately 42 million surgical procedures are performed annually, with many patients experiencing hypertrophic scarring and keloids. We believe that RXI-109 will inhibit connective tissue formation in human fibrotic disease.
Data obtained from pre-clinical studies of our sd-rxRNA compounds in preliminary pre-clinical models using local administration to the skin have shown robust delivery and effective target gene silencing. We have targeted filing an IND forRXI-109 in the second half of 2011. If clinical studies of RXI-109 produce successful results in anti-scarring, we may explore opportunities in other dermatology applications as well as in other anti-fibrotic indications, including pulmonary fibrosis, liver fibrosis, acute spinal cord injury, ocular scarring and restenosis.
Recent Developments
During the fourth quarter of 2010 and the first quarter of 2011, the Company announced several important developments which are outlined below.
On November 2, 2010, we announced that the United States Internal Revenue Service awarded us four Therapeutic Discovery Project (TDP) grants totaling $977,917 as part of the Patient Protection and Affordable Care Act of 2010. The TDP grants were awarded in four equal amounts for developing (1) self-delivering RNAi therapeutic for fibrotic disease, (2) self-delivering RNAi therapeutic for age-related macular degeneration, (3) self-delivering RNAi for ALS (Lou Gehrig’s disease), and (4) oral delivery of glucose encapsulated siRNAs for rheumatoid arthritis.
13
On December 17, 2010, we announced the selection of RXI-109 as our first RNAi therapeutic product candidate to advance into development. We have begun manufacturing activities and we are preparing a pre-IND package to submit to the FDA. Pending FDA review, we intend to use our RNAi therapeutic platformemploy a clinical trial design to study safety and our expertisetolerability as well as initial efficacy in RNAi to identify lead compounds against multiple target genes, and advance them towards pre-clinical anda first clinical development in therapeutic areas that address broad unmet medical needs, in both acute and chronic settings. There are many well-studied genes that have been associated with numerous diseases but have been difficult to target with conventional medicinal chemistry or traditional modalities involving both large and small molecules. We believe RNAi technology may play an important role in targeting these genes and potentially treating the related diseases and disorders. Wetrial targeted for 2012. In this clinical trial, we plan to pursue select disease areas withevaluate RXI-109 for the goalreduction of creating multiple clinical development program opportunities, either independently or with partners through various collaborations and partnerships with academic institutions or pharmaceutical or biotechnology companies.dermal scarring in planned surgeries.
On January 27, 2011, we announced positive initial results as part of our collaboration with miRagen Therapeutics, Inc., in creating microRNA mimics, or artificial copies of microRNAs, using our sd-rxRNA technology. In particular, the collaboration demonstrated efficacy in down-regulating a reporter gene (in vitro) whose expression is controlled by the microRNA in cell culture model systems develop by miRagen. Increasing the level of particular microRNAs by using therapeutic mimics may treat certain diseases, including cardiovascular, cancer, inflammatory, fibrotic and metabolic disorders.
On March 31, 2011, we announced that we have createdhad entered into an Agreement and establishedPlan of Merger (the “Merger Agreement”) with our wholly-owned subsidiary, Diamondback Acquisition Corp., a strong intellectual property portfolio. We have secured exclusiveDelaware corporation (“Merger Sub”), Apthera and nonexclusive licenses from both academic institutionsRobert E. Kennedy, in his capacity as representative of Apthera’s stockholders. On April 13, 2011, pursuant to the terms of the Merger Agreement, Merger Sub merged with and commercial entitiesinto Apthera, with Apthera surviving as a wholly-owned subsidiary of RXi (the “Merger”). In connection with the Merger, we issued 4,974,090 shares of our common stock to former stockholders of Apthera and agreed to make certain issuedcontingent payments to such stockholders upon the achievement of certain milestones. The terms of the Merger Agreement are described inForm 8-Ks, filed by RXi with the Securities and pending patentsExchange Commission (“SEC”) on April 5, 2011 and patent applications covering RNAi technologies in the following three categories: (i) therapeutic targets, (ii) chemistry and configurations of RNAi compounds and (iii) formulation and delivery of RNAi compounds. We have also filed patents based on our internal discoveries in theApril 14, 2011, each of the areas mentioned above, which should enable us to further strengthen our intellectual property portfolio.is incorporated herein by reference.
We were formed in 2006 by CytRx Corporation (“CytRx”) (Nasdaq: CYTR) and four prominent RNAi researchers, including Dr. Craig Mello, who was awarded the 2006 Nobel Prize in Medicine for his co-discovery of RNAi. From 2003 through 2006, CytRx sponsored therapeutic RNAi research at the University of Massachusetts Medical School (“UMMS”) and Massachusetts General Hospital. We commenced operations in January 2007 after CytRx contributed to us its portfolio of RNAi therapeutic assets in exchange for approximately 7.04 million shares of our common stock. These assets consisted primarily of RNAi licenses and related intellectual property and a nominal amount of equipment.
2
4
Our Board of Directors continually evaluates our strategic alternatives and recently determined that it was in the best interests of our stockholders to diversify our development programs with additional development candidates at various stages of development. Our acquisition of Apthera followed from this determination to broaden our strategic direction. We believe that acquiring Apthera will enhance our long-term prospects by giving us access to a late stage product candidate, NeuVax, which is expected to enter Phase III clinical trials under an FDA-approved Special Protocol Assessment (“SPA”) for the treatment of breast cancer in the first half of 2012 if we are able to satisfy certain FDA information requirements to be released from a clinical hold to commence the trial. Based on our clinical trials, we also believe that NeuVax has the potential to treat other cancers, including prostate, bladder and ovarian cancers. In addition, we believe that reducing the scope of our RNAi activities will enable us to commit more resources to RXI-109, our lead RNAi-product, while maintaining our core RNAi discovery and development capability to advance current collaborations, as well as enable alliances.
Management and Scientific Team
In connection with the Apthera acquisition, we reorganized our management and scientific teams. Our Board of Directors believes that the following personnel possess the experience and skills necessary to lead RXi into its next stage of development.
We have a senior management team with experience in developing and commercializing biopharmaceutical and healthcare products consisting of:
| • | Mark J. Ahn, Ph.D., President and Chief Executive Officer. Dr. Ahn served as a director on RXi’s board from 2007 until his appointment as President and Chief Executive Officer in 2011. He brings more than 20 years of experience in the biopharmaceutical industry, including as founder, President and Chief Executive Officer for Hana Biosciences, Inc. Prior to joining Hana, he served as Vice President, Hematology and corporate officer at Genentech, Inc., and held positions of increasing responsibility in strategy, general management, sales and marketing, business development, and finance with Amgen Inc. and Bristol-Myers Squibb Company. | |
| • | Mark W. Schwartz, Ph.D., Executive Vice President and Chief Operating Officer. Dr. Schwartz joins RXi as part of our acquisition of Apthera, where he had been the President and Chief Executive Officer. Prior to joining Apthera, Dr. Schwartz served for five years as President and Chief Executive Officer of Bayhill Therapeutics Inc., a company developing an innovative DNA vaccine platform for the treatment of autoimmune diseases, where he completed a successful partnership with Genentech for the development of the company’s type 1 diabetes vaccine. He had also served as President and Chief Executive Officer of Calyx Therapeutics, Inc., which doubled its size, nurtured a successful working relationship with the FDA, and completed key phase I and phase II international clinical trials of novel anti-inflammatory compounds during his tenure. | |
| • | Robert E. Kennedy, Treasurer and Chief Financial Officer. Robert E. Kennedy co-founded Apthera in 2005, where he served as Director, Secretary, Treasurer and Chief Financial Officer. Previously, Mr. Kennedy served as Director and Chief Financial Officer for Blue Dot Services, Inc., a nationwide heating, ventilation, air-conditioning and plumbing construction and services company. Prior to his work at Blue Dot Services, he was the managing director for Koch Ventures, Inc., the venture capital arm of Koch Industries, Inc., the second largest privately-held company in the United States. Mr. Kennedy has held finance and accounting management roles at Sterling House Corporation, Thorn Americas, Inc., Raytheon Aircraft Corporation, and F.B. Kubik & Company, CPAs; he serves on the board of directors of Immunologix, Inc. and Arizona BioIndustry Association, and is a member of the American Institute of Certified Public Accountants and the Arizona Society of CPAs. | |
| • | Anastasia Khvorova, Ph.D., Chief Scientific Officer. Dr. Khvorova has been our Chief Scientific Officer since October 2008. Dr. Khvorova has contributed significantly to the RNAi field. While at Dharmacon (ThermoFisher Scientific, Inc.), she made major technology advances in RNAi and microRNA. Dr. Khvorova was also responsible for establishing and managing several drug discovery/development collaborations with major pharmaceutical companies, including Abbott Laboratories and |
5
| Alcon Inc. Her groundbreaking work has allowed her to author more than 150 abstracts, 30 patents and patent applications, several book chapters and over 40 peer reviewed publications. Dr. Khvorova received her Ph.D. in Biochemistry from the Russian Academia of Sciences in Moscow in 1994 and after 10 years of working in academia and industry she joined Dharmacon in 2002, where she served as the Chief Scientific Officer for 6 years. |
| • | Pamela Pavco, Ph.D., Vice President Pharmaceutical Development. Dr. Pavco has been our Vice President of Pharmaceutical Development since March 2007. Dr. Pavco brings over 20 years of research and development experience in oligonucleotides to us. From 2002 to 2006, Dr. Pavco was Senior Director, R&D Project Management at Sirna Therapeutics, Inc., previously known as Ribozyme Pharmaceuticals, Inc., where she was responsible for the discovery research and development of Sirna-027, the first chemically modified siRNA to enter into clinical trials. Dr. Pavco also managed the alliance with Allergan, Inc. that was initiated to continue discovery research in the area of ophthalmology and take Sirna-027 forward into Phase 2 clinical studies. Dr. Pavco received a Ph.D. in Biochemistry from Virginia Commonwealth University in 1983 and did her post-doctoral work at Duke University prior to joining Sirna Therapeutics. She is a member of the American Association of Cancer Research and the Association for Research and Vision in Ophthalmology. |
Scientific Advisory Board
We have an accomplished Scientific Advisory Board (“SAB”) which includes Craig C. Mello, Ph.D. (the Chairman of the SAB), George Peoples, Ph.D. (an expert in cancer vaccines and the principal investigator in the Phase I/II clinical trials for NeuVax, who joined our SAB in connection with the Apthera acquisition), Gregory Hannon, Ph.D., Michael Czech, Ph.D., Victor Ambros, Ph.D. and Nassim Usman, Ph.D., together known as the SAB members. The SAB members participate in scientific planning meetings which are typically held every three to six months. During such meetings, our management team and the SAB members review the progress of our research and licensing efforts and provide technological input, including suggestions for new experiments, suggestions regarding the therapeutic relevance of target genes and suggestions regarding new technologies we may want to consider licensingand/or developing internally. Further, certain of our SAB members periodically assist us in business-related activities, such as discussions with potential strategic partners and introductions to potential key consultants and collaborators.
Financial Condition
We have not generated revenue to date and may not generate product revenue in the foreseeable future, if ever. We expect to incur significant operating losses as we advance our product candidates through the drug development and regulatory process. In addition to increasing research and development expenses, we expect general and administrative costs to increase as we add personnel.personnel and integrate Apthera. We will need to generate significant revenues to achieve profitability and might never do so. In the absence of product revenues, our potential sources of operational funding are expected to be the proceeds from the sale of equity, funded research and development payments and payments received under partnership and collaborative agreements.
We had cash and cash equivalents of approximately $5.7$6.9 million as of December 31, 20092010 and approximately $14.4$11.0 million as of March 31, 2010.2011.
We believe that our existing cash and cash equivalents should be sufficient to fund our operations through at least the first halfquarter of 2011. In addition, we also have available to us the SEDA which expires on January 30, 2011.2012. In the future, we will be dependent on obtaining funding from third parties, such as proceeds from the sale of equity, funded research and development payments and payments under partnership and collaborative agreements, in order to maintain our operations and meet our obligations to licensors. There is no guarantee that debt, additional equity or other funding will be available to us on acceptable terms, or at all. If we fail to obtain additional funding when needed, we
36
Recent Financing Activities
On March 4, 2011, we closed an underwritten public offering of 6,000,000 units at a price to the public of $1.35 per unit for gross proceeds of $8.1 million. The financing provided approximately $7.3 million to the Company after deducting the underwriting fee and offering expenses. Each unit consisted of (i) one share of common stock, (ii) a thirteen-month warrant to purchase 0.50 of a share of common stock at an exercise of $1.70 per share (subject to anti-dilution adjustment) and (iii) a five-year warrant to purchase 0.50 of a share of common stock at an exercise price of $1.87 per share (subject to anti-dilution adjustment). As a result of the April 15, 2011 offering, the exercise price of these warrants will be reduced to $1.00 per share.
On April 15, 2011, we priced an underwritten public offering of 11,950,000 units at a price to the public of $1.00 per unit for gross proceeds of $12 million. The financing provided approximately $11 million to the Company after deducting the underwriting fee and offering expenses. The common stock and warrants will be sold in units, with each unit consisting of one share of common stock and one warrant to purchase a share of common stock at an exercise price of $1.00. The warrants are exercisable beginning one year and one day from the date of issuance, but only if our stockholders approve an increase in the number of our authorized shares of common stock, and expire on the sixth anniversary of the date of issuance.
Our Competitive Strengths
We believe we are well positioned to compete successfully in the cancer immunotherapy and RNAi-based therapeutics marketmarkets due to the following competitive strengths:
| • | The Apthera acquisition provides RXi with a late stage product candidate, NeuVax, a peptide-based immunotherapy forlow-to-intermediate HER2+ breast cancer, not eligible for Herceptin, which is expected to enter Phase III clinical trials in the first half of 2012; | |
| • | Novel, proprietary technology platform with the potential to generate multiple RNAi therapeutic product opportunities, comprised of: |
| • | Our rxRNA compound platform that includes multiple distinct approaches all of which offer novelty and potential high | |
| • | Multiple delivery technologies, including |
| • | Accomplished scientific and business team with significant experience in | |
| • | Scientific advisors who are recognized leaders in | |
| • | Strong |
| • | ||
| • | proprietary, formulation and delivery of active RNAi compounds, and | |
| • | key therapeutic target genes; and |
| • | A focus on unmet medical needs and significant market opportunity. |
7
Summary of NeuVax
General
NeuVax is a cytotoxic T-cell activating, epitope-specific immunotherapy comprised of a peptide called E75 and an immune adjuvant called GM-CSF (sargramostim). E75 is derived from HER2/neu, a185-Kd transmembrane glycoprotein that is part of the epidermal growth factor (EGF) family of tyrosine kinases.GM-CSF is recombinant human granulocyte-macrophage colony stimulating factor, a stimulator and activator of macrophages and dendritic cells. HER2/neuis expressed at very low levels in a number of normal epithelial tissues but is amplified and overexpressed in many epithelial tumors. HER2/neuis key to growth of cancer and has proven to be a profitable target for such drugs as Herceptin and Tykerb.
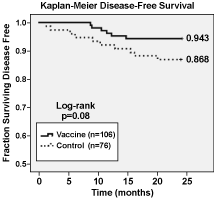
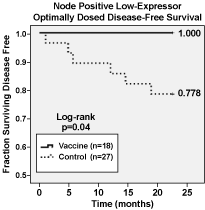
Apthera’s studies have shown that NeuVax boosts a pre-existing immune response found in most cancer patients. Its active component, the9-amino acid peptide, E75 (derived from amino acids369-377 in the extracellular domain of the HER2/neuprotein), is the synthetic version of an epitope recognized by cytotoxic T-lymphocytes (CTLs) and was originally found in tumor-infiltrating lymphocytes of breast and ovarian cancer patients. In contrast to previous clinical studies with peptides, E75 induces a 1,000-fold increased T-cell response; typically 1-2% of circulating T-cells become reactive to the E75 peptide. Such E75-reactive T-cells are therapeutically active as measured by a reduction in disease recurrence in early-stage breast cancer patients. Figure 1 below shows the24-monthfollow-up data for all patients treated during the Phase II clinical trial, including both node positive and node negative patients, HER2 1+, 2+, and 3+ patients, as well as optimally dosed andsub-optimally dosed. The Kaplan-Meier Disease Free Survival rate improved for the patients receiving NeuVax (N=106) compared to the control group (N=76). An even greater improvement can be seen when we focus on Phase II patients who meet the proposed Phase III clinical protocols (i.e., node-positive, low and intermediate HER2+ expressor, optimally-dosed patients) (N=18) compared to the control (N=27). Figure 2 below shows the24-monthfollow-up data for such patients, and the results are statistically significant.
Figure 1 — Kaplan-Meier Disease-Free Survival for All Patients
8
Figure 2 — Kaplan-Meier Disease-Free Survival for Node-Positive Low-Expressor Optimally-Dosed Patients
NeuVax is an epitope-specific immunotherapy. This class of therapies has the highest specificity for tumor cells compared to antigen-specific and polyvalent/whole-cell cancer vaccines and do not contain “self” epitopes or tolerogenic antigens, which are often found in antigen-specific and polyvalent/whole-cell vaccines. NeuVax differs from “cancer vaccines” which utilize partial or entire tumor antigens or polyvalent/whole-cell vaccines (e.g., autologous or patient-specific vaccines) that require presentation on cytokine-activated, dendritic cells, derived from the patient, cultured for a period of time and then re-administered to the patient. Vaccination with self epitopes can theoretically result in the immune system breaking tolerance to “self” and lead to autoimmune reactions against normal tissues while tolerogenic antigens can dampen the immune response to the tumor. In the case of NeuVax, we believe that the presence of a single, immunodominantT-cell epitope will confer both high tumor-specificity, lack of self epitopes and high immunogenicity.
Another potential significant advantage of NeuVax is that the process to manufacture it is relatively simple and can be accomplished using standard peptide synthesis techniques and automated methods. This results in cost of goods that are potentially significantly lower than both antigen-specific and polyvalent/whole-cell vaccines, which have increasingly complex manufacturing schemes.
Apthera and its academic collaborators have developed this therapy using a paradigm that focuses on treating early-stage cancer patients with no/low tumor burden and relatively healthy immune systems compared to patients with advanced/metastatic disease. Also, we intend to use a clinical trial endpoint focused on disease recurrence and disease-free survival in its Phase III trial for breast cancer as the basis for conditional approval of NeuVax rather than an overall survival endpoint. In contrast, cancer vaccines to date have been evaluated in patients with advanced/metastatic disease, resulting in equivocal survival data in clinical trials and several clinical development failures.
9
Potential Advantages ofNeuVax Versus Cancer Vaccines
Parameter | Neuvax | Antigen-Specific Vaccines | Polyvalent/Whole-Cell Vaccines | |||
Tumor Specificity | Highest | Less specific than peptide vaccines | Lowest | |||
Presence of Self and/or Tolerogenic Antigens | None — E75 peptide contains a single, immunodominant epitope | Potential for multiple self epitopes that may be immunodominant over E75 | Mostly self antigens that may be immunodominant over E75 | |||
Cost/Ease of Manufacturing | Relatively easy and inexpensive automated peptide synthesis | More complex; requires recombinant or DNA plasmid; often requires autologous dendritic cells for presentation | Very complex, multi-step and expensive process for culturing cells; fraught with batch-to-batch inconsistency | |||
Clinical Development Path | Early-stage patientswith low tumor burden and robust immune systems | Advanced/metastatic patients with high tumor burden and poorly functioning immune systems | Advanced/metastatic patients with high tumor burden and poorly functioning immune systems | |||
Trial Endpoints | Disease recurrence | Survival and disease recurrence | Survival and disease recurrence |
NeuVax has been evaluated in Phase I/II clinical trials in early-stage breast cancer patients and aPhase I/II trial in prostate cancer patients at high risk for disease recurrence. Based on these Phase I/II results and feedback from Apthera’sEnd-of-Phase-II meeting with the FDA, we are preparing to launch a multi-center Phase III trial in breast cancer under an SPA in early 2012 pending a release from a partial clinical hold imposed on us by the FDA until we provide the FDA with Chemical Manufacturing and Controls (“CMC”) information that is required to commence such clinical trials. If we obtain strong interim analysis, we expect to commence a second Phase III trial immediately following completion of patient enrollment into the first Phase III trial and would then expect to conclude both trials in 2018.
In addition to breast cancer, we intend to further develop NeuVax for the treatment of prostate cancer patients at high risk for disease recurrence, as well as for other solid tumor types that express HER2.
Market Opportunity for NeuVax
We intend to develop NeuVax for the treatment of node-positive (NP) breast cancer and secondarily for high-risk prostate cancer patients that possess the HLA-2 or HLA-3 haplotype and have HER2-expressing tumors. As summarized in the table below, of the approximate 182,500 individuals diagnosed with breast and prostate cancer annually in the United States, we expect that approximately 18,400, or around 10%, will be eligible for NeuVax treatment translating into a potential market opportunity of $1.06 billion in the U.S. alone.
U.S. Market Opportunities for NeuVax
| Est. Annual U.S. | ||||||||||||
| Newly Diagnosed | Number of NeuVax | Market | ||||||||||
Indication | Cases/Year | Eligible Patients | Opportunity | |||||||||
| ($ millions)1 | ||||||||||||
| Breast cancer | 182,500 | 18,400 | $ | 1,060 | ||||||||
| (1) | U.S. insurance reimbursement assumed at $35,000 for initial course of therapy. |
Introduction to the Field of RNAi Therapeutics
RNAi is a naturally-occurring phenomenon where short double-stranded RNA molecules interfere with the expression of targeted genes. RNAi technology takes advantage of this phenomenon and potentially allows
10
us to effectively interfere with particular genes within living cells by designing RNA-derived molecules targeting those genes. RNAi is regarded as a significant advancement in the scientific community, as evidenced by the journalScience’s selection of RNAi as the “Breakthrough of the Year” in 2002, and by the awarding of the 2006 Nobel Prize in Medicine to the co-discoverers of RNAi, including Dr. Craig Mello, an RXi founder and our SAB Chairman.
RNAi offers a novel approach to the drug development process because, as described below under “The RNAi Mechanism,” RNAi compounds can potentially be designed to target any one of the thousands of human genes, many of which are undruggable by other modalities. In contrast, an article published in the December 2005 edition ofDrug Discovery Todayby Andreas P. Russ and Stefan Lampel has demonstrated that only a subset of the proteins encoded in the human genetic code (human genome) are able to be targeted efficiently by traditional medicinal chemistry or antibody-based approaches. The specificity of RNA interference is achieved by an intrinsic well-understood biological mechanism based on designing the sequence of an RNAi compound to match the sequence of the targeted gene. The specificity of RNAi may be sufficient to permit therapeutic targeting of only a single gene and, importantly, may even selectively reduce or eliminate expression from a single abnormal copy of a gene while preserving expression from a normal copy (“allele-specific” targeting). This is critical in diseases such as cancer and neurodegenerative disorders that are often caused by abnormal copies of genes.
4
The RNAi Mechanism
The genome is made of a double-strand of DNA (the double helix) that acts as an instruction manual for the production of the roughly 30,000 to 50,000 human proteins. Proteins are important molecules that allow cells and organisms to live and function. With rare exceptions, each cell in the human body has the entire complement of genes. However, only a subset of these genes directs the production of proteins in any particular cell type. For example, a muscle cell produces muscle-specific protein, whereas a skin cell does not.
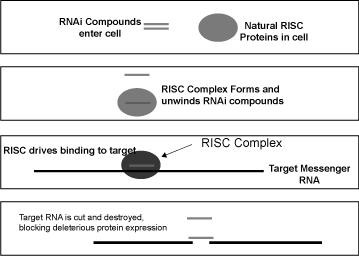
In order for a gene to guide the production of a protein, it must first be copied into a single-stranded chemical messenger (messenger RNA or mRNA), which is then translated into protein. RNA interference is a naturally occurring process by which a particular messenger RNA can be destroyed before it is translated into protein. The process of RNAi can be artificially induced by introducing a small double-stranded fragment of RNA corresponding to a particular messenger RNA into a cell. A protein complex within the cell called RISC (RNA-Induced Silencing Complex) recognizes this double-stranded RNA fragment and splits thedouble-strands apart, retaining one strand in the RISC complex. The RISC then helps this guide strand of RNA bind to and destroy its corresponding cellular messenger RNA target. Thus, RNAi provides a method to potentially block the creation of the proteins that cause disease, as depicted in the following figure.
11
Figure 1 — Mechanism of RNA interference within a cell
Since gene expression controls most cellular processes, the ability to inhibit gene expression provides a potentially powerful tool to treat human diseases. Furthermore, since the human genome has already been decoded, and based on numerous gene-silencing reports, we believe that RNAi compounds can readily be designed to interfere with the expression of any specific gene. Based on our internal research and our review of certain scientific literature we also believe that our RNAi platform may allow us to develop create therapeutics with significant potential advantages over traditional drug development methods, including:
| • | High specificity for targeted | |
| • | High potency (low doses) | |
| • | Ability to interfere with the expression of potentially any | |
| • | Accelerated generation of lead | |
| • | Low toxicity, natural mechanism of action. |
5
RXi’s RNAi Therapeutic Platform
RNAi Compound Design
RNAi compounds are made from a strand or strands of RNA that are manufactured by a nucleic acid synthesizer. The synthesizer is programmed to assemble a strand of RNA of a particular sequence using the four kinds of nucleotide units (Adenine (“A”), Uracil (“U”), Cytidine (“C”) and Guanosine (“G”)) that match a small segment of the targeted gene. The hallmark of an RNAi compound is that it has a double stranded
12
region. The compounds can be of various lengths of nucleotide units (nt). As seen in Figure 2 below, the two strands can have overhangs (as shown on the far left), or they can have blunt ends (as shown in the middle and right). A single strand can form an RNAi compound by forming a structure referred to as a hairpin.
Figure 2 — Types of RNAi Compounds

The length and shape of the compound can affect the activity and hence the potency of the RNAi in cells. The first design of RNAi compounds to be pursued for development as a human therapeutic was a short double-stranded RNA that included at least one overhanging single-stranded region, known as small interfering RNA, or siRNA which we also refer to as classic siRNA and can be seen in Figure 2 above.
Figure 3 — First generation of RNAi pursued for human therapeutics: classic siRNA
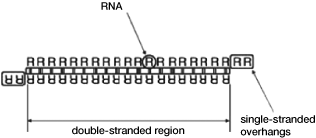
In the case of classic siRNA, double-stranded RNA with single-stranded overhangs is used. The two strands comprising the RNA have bases that are complementary to each other in order to create double-stranded regions; that is, an “A” on one strand is paired with a “U” on the other, and a “C” on one strand is paired with a “G” on the other, creating double-stranded regions. The pairing holds the two strands together creating double-stranded RNA. The overhangs that are at the ends of the double-stranded RNA do not have a matching partner and thus these single-stranded bases in the overhang area are exposed to nucleases in the environment which can degrade the molecule. Classic siRNA therapeutics are about 19 to 23 base pairs long.
We believe that classic siRNAs have drawbacks that may limit the usefulness of those agents as human therapeutics, and that we may be able to utilize the technologies we have licensed and developed internally to optimize RNAi compounds for use as human therapeutic agents. It is the combination of the length, the nucleotide sequence, and the configuration of chemical modifications that are important for effective RNAi therapeutics. For example, the RNA can be chemically modified in a manner that reduces its sensitivity to nucleases, which are enzymes that attack and degrade RNA. Likewise, it is our expectation that removing the single-stranded overhang regions will be a way of reducing the rate of spurious degradation of the RNAi, as single-stranded RNA is more susceptible to degradation than double-stranded RNA. The length range of 19 to 23 nucleotides can also be varied to yield more potent RNAi compounds. Introducing “mismatches” in the
6
double-stranded region, that is, discrete internal portions of the duplex region that do not form good base pairs between the two strands, also may be a useful way of improving the potency of the resulting RNA.
Depending on the delivery method selected, stabilizing RNAi compounds by chemical modification may be critical for RNAi activity in animal models and in humans. The stabilization may be necessary to protect the RNAi compounds from being degraded by enzymes that exist in bodily fluids. Many of our employees and SAB Members are accomplished in the field of chemically modified RNAi design. For example, Dr. Craig
13
Mello, an expert in the field, is an inventor of the Fire-Mello seminal family of patents (“Fire-Mello”), which we have a license to, Dr. Tod Woolf, our former President and CEO, was a co-inventor of the Stealthtm RNAi brand of chemically modified RNAi compounds and Dr. Anastasia Khvorova, our Chief Scientific Officer, was a co-inventor of OnTarget Plustm, siStable® and Accelltm brands of chemically modified RNAi compounds. We will continue to employ their collective expertise to design chemically modified RNAi compounds. We have in-licensed technology on chemically stabilized RNAi compounds that will serve as a foundation for our chemical modification strategy.
Our internal research leads us to believe that next generation rxRNA compounds offer significant advantages over classic siRNA used by other companies developing RNAi therapeutics, highlighted by the following characteristics:
| • | Up to 100 times more active than classic | |
| • | More resistant to nuclease | |
| • | Readily | |
| • | Potentially more specific for the target | |
| • | More reliable at blocking immune side effects than classic | |
| • | In the case ofsd-rxRNA, |
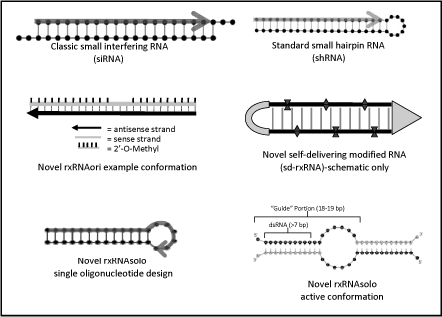
Based on our own research we have developed a variety of novel siRNA configurations with potential advantages for therapeutic use. The first of these has been termed rxRNAori. This configuration has some similarities to classic siRNA in that it is composed of two, short RNA strands. We have found that by using a somewhat longer length (25 bp), removing the overhangs and using proprietary chemical modification patterns we achieve a higher hit rate of very potent (picomolar potency) compounds in a given target sequence. These rxRNAoricompounds are modified to increase resistance to nucleases and to prevent off-target effects including induction of an immune response. These novel RNAi compounds are distinct from the siRNA compounds used by many other companies developing RNAi therapeutics in that they are designed specifically for therapeutic use and offer many of the properties that we believe are important to the clinical development of RNAi-based drugs.
The second novel configuration has been termed rxRNAsoloto indicate the fact that it is composed of a single RNA oligonucleotide strand. This configuration also makes use of carefully placed and selected chemical modifications to introduce properties for therapeutic use as described above. Conventional shRNA (short hairpin RNA) and siRNA compounds with duplex regions of19-27 nucleotides are efficient substrates for RNAi machinery in mammalian cells. Efficacy of single-stranded oligonucleotides is substantially lower (by 3-4 orders of magnitude). We have developed a single-oligo (27 nt long)long ) construct with silencing potency equal to conventional RNAi triggers. These molecules are designed to have at least 16 bases complementary to the target mRNA and are then extended at the 3'3’ end to allow efficient self annealing (dimerization). This new class of RNAi compounds has the potential to further reduce off-target effects and manufacturing costs and may thus offer additional advantages for use in research and therapeutic applications.
A third novel configuration has been called “sd-rxRNA” to indicate its novel “self-delivering” properties which do not require additional delivery vehicles for efficient cellular uptake and RISC-mediated silencing. A combination of at least three characteristics is required for activity: 1)(a) specific, proprietary chemical modifications, 2)(b) a precise number of chemical modifications and 3)(c) reduction in oligonucleotide content.
7
Kineteic analyses of fluorescently-labeled compounds demonstrate that efficient cellular internalization is observed within minutes of exposure. These molecules are taken up efficiently and cause target gene silencing in diverse cell types (cell lines and primary cells).cells. This novel class of RNAi compounds may afford a broad opportunity for therapeutic development.
14
Figure 4 — Classic and Novel RNAi Compound Configurations
8
Delivery
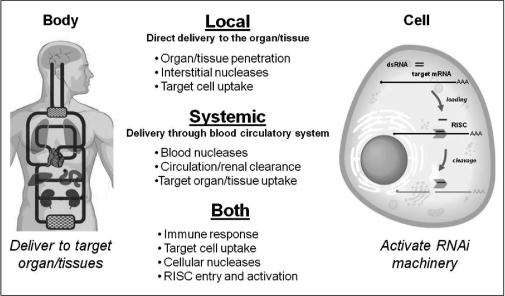
Figure 5 — Principles and Challenges of RNAiin vivo Delivery
We believe both chemical modification and formulation of RNAi compounds may be utilized to develop RNA drugs suitable for therapeutic use. A series of delivery hurdles must be overcome to achieve in vivo efficacy: (1) delivery to the target tissue or organ (2) tissue penetration and distribution (3) crossing of the
15
plasma membrane (4) intracellular trafficking to the RISC (RNAi machinery complex) (5) incorporation into and activation of the RISC. Different cell types and tissues may each require unique approaches. Three categories of tools currently exist: variation in administration route, selection of delivery vehicle, and chemical modification of the RNA compound. A combination of some or all of these is likely to be required for successful delivery.
The route by which an RNAi therapeutic is brought into contact with the body depends on the intended organ or tissue to be treated. Delivery routes can be simplified into two major categories: local (when a drug is delivered directly to the tissue of interest) or systemic (when a drug accesses the tissue of interest through the circulatory system). Local delivery may avoid some hurdles associated with systemic approaches such as circulation clearance and tissue extravasation (crossing the endothelial barrier from the blood stream). However, this approach can only be applied to a limited number of organs or tissues (e.g.(e.g., skin, eye, lung, and potentially, the central nervous system).
RNAi delivery vehicles, a large and diverse group of particles (e.g.(e.g., polymer-based particles, lipoplexes, other), can contribute in additional ways to successful delivery. Formulation can help prevent nuclease degradation, improve nucleotide retention in circulation and alter tissue and cell. In some cases, a formulation can result in more efficient cellular uptake and intracellular release.
RNA chemical modifications as described above of thesd-rxRNA compounds as described above, can include base, backbone and sugar modifications, as well as covalently bound moieties such as cholesterol, antibody fragments or peptides. Some of these modifications can be utilized to enhance stability, reduce immunogenicity, improve circulation properties (presumably through binding to blood transporter proteins), increase tissue access and improve uptake to cells and the RNAi machinery. A combination of chemical modifications, delivery vehicles or both might be dictated by the target organs/tissues and specific requirements for the therapeutic application, including the intended administration route.
Our founding scientists recognized that the key to therapeutic success with RNAi lies in delivering intact RNAi compounds to the target tissue and the interior of the target cells. To accomplish this, we have
9
developed a comprehensive platform that includes local, systemic and oral delivery approaches. We work with chemically synthesized RNAi compounds that are optimized for stability and efficacy, and combine delivery at the site of action and formulation with delivery agents to achieve optimal delivery to specific target tissues.
Local Delivery
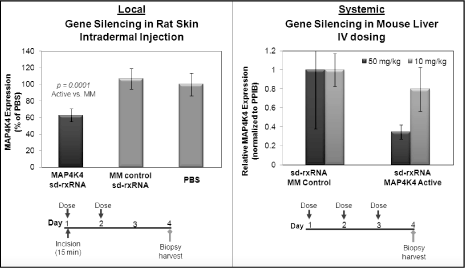
sd-rxRNA molecules have unique properties which improve tissue and cell uptake. Delivery ofsd-rxRNA by a local route of administration may avoid hurdles associated with systemic approaches such as rapid clearance from the bloodstream and inefficient extravasation (e.g.(e.g., crossing the endothelial barrier from the blood stream). We have studiedsd-rxRNA molecules in a rat model of dermal delivery. Direct application ofsd-rxRNA with no additional delivery vehicle to the skin (incision introduced) demonstrates that target gene silencing can be measured after topical delivery. Figure 6 illustrates that direct injection ofsd-rxRNA into the dermis layers of the skin with no additional delivery vehicle resulted in efficient uptake and significant target gene silencing. The dose levels required for these direct injection methods are small and suitable for clinical development suggesting that local delivery indications will be very accessible with thesd-rxRNA technology platform. Target tissues that are potentially accessible for local delivery usingsd-rxRNA compounds include lung, eye, skin, CNS, mucosal tissues, sites of inflammation, and tumors (direct administration).
Systemic Delivery
Systemic delivery occurs when a drug accesses the tissue of interest through the circulatory system. In some cases, such as in targeting a treatment to the liver, the optimal route of delivery may be by a systemic route. We have developed a portfolio of systemic delivery solutions utilizing our RNAi therapeutic platforms. One novel approach involves the use ofsd-rxRNA compounds. The self-delivering technology introduces properties required forin vivoefficacy such as cell and tissue penetration and improved blood clearance and distribution properties. Systemic delivery of these compounds to mice has resulted in gene specific inhibition with no additional delivery vehicle required as shown in Figure 6 below . In addition, we have developed
16
novel nanotransporter formulations to aid in transport of RNAi compounds to both liver and various other target tissues in the body. These nanotransporters are chemically synthesized molecules that form nanometer-sized particles when mixed with RNAi compounds and alter the clearance, distribution and tissue penetration properties of the RNAi compounds. Delivery of RNAi compounds to the liver might be critical for the treatment of many diseases and using rxRNA in conjunction with such delivery vehicles has enabled us to demonstrate gene specific inhibition at low doses in a mouse model after intravenous, systemic delivery. Target tissues that are potentially accessible using rxRNA compounds by systemic delivery include liver, lung, adipocytes, cardiomyocytes, bone marrow, sites of inflammation, tumors, vascular endothelium, and kidney.
10
Figure 6: Data demonstratingin vivo gene silencing with sd-rxRNA in local and systemic settings
Oral Delivery
Most RNAi therapeutic products being developed today require recurring intravenous injections or other forms of administration which are not patient friendly. To address the desire for RNAi therapeutics with improved modes of administration, we are testing a novel formulation technology, Glucan Encapsulated RNAi Particles (GeRPs), that may allow our rxRNA compounds to be incorporated into orally administered pills. Early data to date suggest that the GeRP delivery system appears to be more potent than previous methods used for systemic delivery of RNAi therapeutics by intravenous injection. Additional studies will need to be conducted to clearly establish the flexibility of the GeRP system and to determine whether they can either be used to administer a single RNAi compound, multiple RNAi compounds, or could potentially allow co-delivery of RNAi, DNA, protein and small molecule combinations.
11

Alliance Partners in Therapeutic Areas
We are actively seeking to leverage our technology platformplatforms by seeking to work with pharmaceutical and biotechnology partners in the partners’ fields of interest. Our team has experience targeting genes in virtually every major therapeutic area, and based on this experience, we believe we can discover many more drug candidates by working with partners than we can develop with our own resources. We are seeking to work with partners in the discovery and development of rxRNA based drugs in a number of therapeutic areas.
12
Intellectual Property and Proprietary Rights
We actively seek protection for our proprietary information by means of United States and foreign patents, trademarks, and copyrights, as appropriate.copyrights. In addition, we rely upon trade secret protection and contractual arrangements to protect certain of our proprietary information and products. We have pending patent
17
applications that relate to potential drug targets, compounds we are developing to modulate those targets (described throughout herein as rxRNA), methods of making or using those compounds and proprietary elements of our drug discovery platform.
Much of our technology and many of our processes depend upon the knowledge, experience and skills of key scientific and technical personnel. To protect our rights to our proprietary know-how and technology, we require all employees, as well as our consultants and advisors when feasible, to enter into confidentiality agreements that require disclosure and assignment to us of ideas, developments, discoveries and inventions made by these employees, consultants and advisors in the course of their service to us.
We have also obtained rights to various patents and patent applications under licenses with third parties, which provide forrequire us to the payment of milestonespay royalties and royalties by us.milestone payments. The ultimate degree of patent protection that will be afforded tofor biotechnology products and processes, including ours, remains uncertain, both in the United States and in other important markets, remains uncertain and is dependent uponbecause the scope of protection decided upon by thedepends on decisions of patent offices, courts and lawmakers in these countries. There is no certainty that our existing patents or others, if obtained, will afford us substantial protection or commercial benefit. Similarly, there is no assurance that our pending patent applications or patent applications licensed from third parties will ultimately be granted as patents or that those patents that have been issued or are issued in the future will stand if they are challenged in court. We assess our license agreements on an ongoing basis, and may from time to time terminate licenses to technology that we do not intend to employ in our immunotherapy or RNAi technology platform,platforms, or in our product discovery or development activities.
We have 12 pending patent applications 10 of which were filed as PCTs to date, encompassing what we believe to be important new compounds and their use as therapeutics in RNAi, chemical modifications of these and existing RNAi compounds that improve the compounds’ suitability for therapeutic uses (including delivery), and compounds directed to specific targets (that address specific disease states). Any patents that may issue from these pending patent applications will be set to expire between 2028 and 2031, not including any patent term extensions that may be afforded to the patents that encompass products (or processes for making or using the same) that are human drug products subject to regulation under the Federal Food, Drug and Cosmetic Act (and the equivalent provisions in foreign jurisdictions) for any delays incurred during the regulatory approval process of therelating to human drug product.products (or processes for making or using human drug products).
Immunotherapy-related Licenses
As a result of our acquisition of Apthera, we acquired exclusive and non-exclusive rights to develop NeuVax for the treatment of cancer by licensing key patent rights from third parties. These rights include composition of matter on E75, the active peptide component of NeuVax, and methods of use thereof.
The Board of Regents, University of Texas and Henry Jackson Foundation
We obtained, through the completion of the merger with Apthera, an exclusive license from the University of Texas through Apthera’s Patent and Technology License AgreementsAgreement with The Board of Regents of the University of Texas System (the “Texas License”). The Texas License provides an exclusive right to use the E75 peptide in humans for therapeutic purposes, under Issued U.S. Patent No. 6,514,942, titled “Methods and Compositions for Stimulating T-lymphocytes”. This patent expires in 2015, without taking into account any patent extensions that may be available, and we do not expect to commence commercialization of NeuVax prior to 2018, if at all.
We have also secured a royalty fee, non-exclusive irrevocable license to practice the inventions described in PCT Published Patent Application WO/2008/15057, titled “Vaccine for the Prevention of Breast Cancer Relapse” from the Henry Jackson Foundation. WO/2008/15057 provides protection on improved methods for treating breast cancer using the E75 peptide as identified in clinical trials. We are currently negotiating the terms of an exclusive license to these patents, although we may not successfully conclude such negotiations.
18
The issued United States patent and any patents that may issue from the licensed or pending patent applications will be set to expire between 2015 for our composition of matter patent and we have no equivalent protection outside of the United States and 2028 for other patent applications covering methods of treating cancer patients, not including any patent term extensions that may be afforded under the Federal Food, Drug and Cosmetic Act (and the equivalent provisions in foreign jurisdictions). We are currently negotiating for exclusive rights to a patent application to which we already have non-exclusive rights that cover certain aspects of the method of treatment using NeuVax that could provide additional patent protection in major countries around the world through 2027.
In connection with the Texas License, we are obligated to pay specified milestones and royalties on sales of products covered by the licensed patents, including royalties possibly extending beyond the expiration date of a patent.
We are preparing to apply for Orphan Drug status for NeuVax, which, if granted, could provide seven years or ten years of data exclusivity, or the inability of another company to use our clinical data to support their application for regulatory approval in the United States or European Union, respectively. We also anticipate that NeuVax will qualify for 12 years of data exclusivity under the Patient Protection and Affordable Care Act.
RNAi-related Licenses
We have also secured exclusive and non-exclusive rights to develop RNAi therapeutics by licensing key RNAi technologies and patent rights from third parties. These rights relate to chemistry and configurationsconfiguration of RNAi compounds, delivery technologies of RNAi compounds to cells, and therapeutic targets.
13
University of Massachusetts Medical School
Under these licenses, UMMS granted to us exclusive, worldwide licenses, with the right tosub-license, to three different patent families and a non-exclusive, worldwide license to a fourth patent family. As
14
These licensesrights will
19
expire upon the expiration of all patents licensed under the licenses,agreement, are terminable by either party upon an uncured breach by the other party, and may be terminated by us for any reason following a specified notice period. We are generally required to indemnify UMMS for losses incurred by UMMS based on the exercise of the licensed patents by us.
In connection with all of our licenses with UMMS, including those assigned to us by CytRx, as well as those entered into directly between us and UMMS, we are obligated to pay specified royalties on net sales of products covered by the licensed patents, subject to minimum annual royalties.
Other License Agreements
Fire-Mello.We recently terminatedhave a non-exclusive license to the Fire-Mello (US 6,506,559, set to expire in 2018) and related applications covering the use of double stranded RNA to induce gene silencing that describes RNAi products, compositions and therapeutic RNAi methods.
TriLink Biotechnologies. In August 2007, we entered into a license agreement with TriLink Biotechnologies, Inc. for three RNAi chemistry technologies for all therapeutic RNAi applications, for which we paid an up-front fee and agreed to pay yearly maintenance fees, as well as future clinical milestone payments and royalty payments based on sales of therapeutic products developed from the licensed technologies.
Dharmacon. In October 2007, we entered into a license agreement with Dharmacon, Inc. (now part of Thermo Fisher Scientific Inc.), pursuant to which we obtained an exclusive license to certain RNAi sequences to a number of these UMMS licenses and still hold licensestarget genes for the development of our rxRNA compounds. Further, we have obtained the right to patents andlicense additional RNAi sequences, under the same terms, disclosed by Thermo Fisher Scientific Inc. in its pending patent applications that belongagainst target genes and have received an option for exclusivity for other siRNA configurations. As consideration for this license, we paid an up-front fee and agreed to six distinct familiespay future clinical milestone payments and royalty payments based on sales of patent applicationssiRNA compositions developed in connection with the licensed technology.
Life Technologies. In November 2007, we entered into a license agreement with Life Technologies, Inc., pursuant to which we were granted rights under four patents relating to RNA target sequences, RNA chemical modifications, RNA configurationsand/or RNA delivery to cells. As consideration for this license, we paid an up-front fee of $250,000 and agreed to pay yearly maintenance fees of the same amount beginning in 2008. Further, we are obligated to pay a fee for each additional gene target added to the license as well as a fee on the first and second anniversaries of the date we were granted consent to add the gene target to the list of those covered by the license. We have also been granted, for each gene target, an option to secure pre-clinical rightsand/or the clinical rights, for which we would be required to pay additional fees. Further, we are required to make future clinical milestone payments and royalty payments based on sales of therapeutic products developed from the original thirteen.licensed technologies.
15
Competition
A number of multinational pharmaceutical companies, as well as large biotechnology companies, including Roche Laboratories, Inc., Pfizer Inc., Bayer HealthCare AG, Sanofi-Aventis, US, LLC, Amgen, Inc. GlaxoSmithKline plc, and Genentech are pursuing the development or are currently marketing pharmaceuticals that target oncology pathways on which we are focusing. It is probable that the number of companies seeking to develop products and therapies for the treatment of unmet needs in oncology will increase.
20
There are also a number of medical institutions and pharmaceutical companies are seeking to develop therapeutic products using RNAi technologies. Companies working in this area include: Alnylam Pharmaceuticals, MDRNA, Cequent Pharmaceuticals,Inc., Tacere Therapeutics, Inc., Benitec Limited, OPKO Health, Inc., Silence Therapeutics plc, Quark Pharmaceuticals, Inc., Rosetta Genomics Ltd., Lorus Therapeutics, Inc., Tekmira Pharmaceuticals Corporation, Calando Pharmaceuticals, Inc., Regulus Therapeutics Inc. and Santaris Pharmaceuticals, as well as a number of large pharmaceutical companies. A number of the large pharmaceutical companies also either have in-house RNAi development programs or are collaborating with smaller biopharmaceutical companies. This competition will manifest itself in the discovery and development of RNAi technology, in recruiting and retaining key scientific and management personnel, in securing strategic alliances, and in obtaining rights to key intellectual property.
Our RNAi-focused competitors as well as major pharmaceutical companies may be targeting the same diseases we intend to target. Competitors both in and outside of the oncology immunotherapeutic and RNAi fieldfields have financial resources, research and development staffs, and facilities that are, in most cases, substantially greater than ours or potentially those of our strategic partners or licensees and are engaged in the research and development of pharmaceutical products that could compete with our potential products. The industry is characterized by rapid technological advances and competitors may develop products more rapidly and such products may be more effective than those currently under development or that may be developed in the future by our strategic partners or licensees.
Government Regulation
The United States and other developed countries extensively regulate the pre-clinical and clinical testing, manufacturing, labeling, storage, record-keeping, advertising, promotion, export, marketing and distribution of drugs and biologic products. The United States Food and Drug Administration (“FDA”)FDA regulates pharmaceutical and biologic products under the Federal Food, Drug, and Cosmetic Act, the Public Health Service Act and other federal statutes and regulations.
To obtain approval of our future product candidates from the FDA, we must, among other requirements, submit data supporting safety and efficacy for the intended indication as well as detailed information on the manufacture and composition of the product candidate. In most cases, this will require extensive laboratory tests and pre-clinical and clinical trials. The collection of these data, as well as the preparation of applications for review by the FDA involve significant time and expense. The FDA also may require post-marketing testing
16
to monitor the safety and efficacy of approved products or place conditions on any approvals that could restrict the therapeutic claims and commercial applications of these products. Regulatory authorities may withdraw product approvals if we fail to comply with regulatory standards or if we encounter problems at any time following initial marketing of our products.
The first stage of the FDA approval process for a new biologic or drug involves completion of pre-clinical studies and the submission of the results of these studies to the FDA. ThisThese data, together with proposed clinical protocols, manufacturing information, analytical data and other information submitted to the FDA, in an investigational new drug application, or IND, must become effective before human clinical trials may commence. Pre-clinical studies generally involve FDA regulated laboratory evaluation of product characteristics and animal studies to assess the efficacy and safety of the product candidate.
After the IND becomes effective, a company may commence human clinical trials. These are typically conducted in three sequential phases, but the phases may overlap. Phase I trials consist of testing the product candidate in a small number of patients or healthy volunteers, primarily for safety at one or more doses. Phase II trials, in addition to safety, evaluate the efficacy of the product candidate in a patient population somewhat larger than Phase I trials. Phase III trials typically involve additional testing for safety and clinical efficacy in an expanded population at multiple test sites. A company must submit to the FDA a clinical protocol, accompanied by the approval of the Institutional Review Boards at the institutions participating in the trials, prior to commencement of each clinical trial.
To obtain FDA marketing authorization, a company must submit to the FDA the results of the pre-clinical and clinical testing, together with, among other things, detailed information on the manufacture and
21
composition of the product candidate, in the form of a new drug application, or NDA, or, in the case of a biologic, a biologics license application, or BLA.
The amount of time taken by the FDA for approval of an NDA or BLA will depend upon a number of factors, including whether the product candidate has received priority review, the quality of the submission and studies presented, the potential contribution that the compound will make in improving the treatment of the disease in question, and the workload at the FDA.
The FDA may, in some cases, confer upon an investigational product the status of a fast track product. A fast track product is defined as a new drug or biologic intended for the treatment of a serious or life threatening condition that demonstrates the potential to address unmet medical needs for this condition. The FDA can base approval of an NDA or BLA for a fast track product on an effect on a surrogate endpoint, or on another endpoint that is reasonably likely to predict clinical benefit. If a preliminary review of clinical data suggests that a fast track product may be effective, the FDA may initiate review of entire sections of a marketing application for a fast track product before the sponsor completes the application.
We anticipate that our products will be manufactured by our strategic partners, licensees or other third parties. Before approving an NDA or BLA, the FDA will inspect the facilities at which the product is manufactured and will not approve the product unless the manufacturing facilities are in compliance with the FDA’s current good manufacturing practice (“cGMP”), which are regulations that govern the manufacture, holding and distribution of a product. Manufacturers of biologics also must comply with the FDA’s general biological product standards. Our manufacturers also will be subject to regulation under the Occupational Safety and Health Act, the Environmental Protection Act, the Nuclear Energy and Radiation Control Act, the Toxic Substance Control Act and the Resource Conservation and Recovery Act.Act and other applicable environmental statutes. Following approval, the FDA periodically inspects drug and biologic manufacturing facilities to ensure continued compliance with the good manufacturing practices regulations. Our manufacturers will have to continue to comply with those requirements. Failure to comply with these requirements subjects the manufacturer to possible legal or regulatory action, such as suspension of manufacturing or recall or seizure of product. Adverse patient experiences with the product must be reported to the FDA and could result in the imposition of marketing restrictions through labeling changes or market removal. Product approvals may be withdrawn if compliance with regulatory requirements is not maintained or if problems concerning safety or efficacy of the product occur following approval.
17
The labeling, advertising, promotion, marketing and distribution of a drug or biologic product also must be in compliance with FDA and Federal Trade Commission requirements which include, among others, standards and regulations for off-label promotion, industry sponsored scientific and educational activities, promotional activities involving the internet, anddirect-to-consumer advertising. We also will be subject to a variety of federal, state and local regulations relating to the use, handling, storage and disposal of hazardous materials, including chemicals and radioactive and biological materials. In addition, we will be subject to various laws and regulations governing laboratory practices and the experimental use of animals. In each of these areas, as above, the FDA has broad regulatory and enforcement powers, including the ability to levy fines and civil penalties, suspend or delay issuance of product approvals, seize or recall products, and deny or withdraw approvals.
We will also be subject to a variety of regulations governing clinical trials and sales of our products outside the United States. Whether or not FDA approval has been obtained, approval of a product candidate by the comparable regulatory authorities of foreign countries and regions must be obtained prior to the commencement of marketing the product in those countries. The approval process varies from one regulatory authority to another and the time may be longer or shorter than that required for FDA approval. In the European Union, Canada and Australia, regulatory requirements and approval processes are similar, in principle, to those in the United States.
Environmental Compliance
Our research and development activities involve the controlled use of potentially harmful biological materials as well as hazardous materials, chemicals and various radioactive compounds. We are subject to
22
federal, state and local laws and regulations governing the use, storage, handling and disposal of these materials and specific waste products. We are also subject to numerous environmental, health and workplace safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of bio-hazardous materials. The cost of compliance with these laws and regulations could be significant and may adversely affect capital expenditures to the extent we are required to procure expensive capital equipment to meet regulatory requirements.
Human Resources
As of March 29, 2010,31, 2011, we had 28 employees, 27 of whom were32 full-time employees and one of whom was a part-time employee. 19employees. 22 of our employees are engaged in research and development and 910 of our employees are engaged in management, administration and finance. None of our employees are represented by a labor union or covered by a collective bargaining agreement, nor have we experienced any work stoppages.
Insurance
| ITEM 1A. | RISK FACTORS |
Our business is subject to numerous risks. We caution you that the following important factors, among others, could cause our actual results to differ materially from those expressed in forward-looking statements made by us or on our behalf in filings with the SEC, press releases, communications with investors and oral statements. Any or all of our forward-looking statements in this annual report onForm 10-K and in any other public statements we make may turn out to be wrong. They can be affected by inaccurate assumptions or by known or unknown risks and uncertainties. Many factors mentioned in the discussion below will be important in determining future results. Consequently, no forward-looking statement can be guaranteed. Actual future results may vary materially from those anticipated in forward-looking statements. When used in thisForm 10-K, the words “believe,” “anticipate,” “expect,” “estimate,” “intend,” “plan,” “project,” “will be,” “will continue,” “will result,” “seek,” “could,” “may,” “might” and similar expressions are intended to identify forward-looking statements. We explicitly disclaim any obligation to update any forward-looking statements to
18
reflect events or circumstances that arise after the date hereof. You are advised, however, to consult any further disclosure we make in our reports filed with the SEC.
Risks Relating to RXi’s Business and Industry
The approach we are taking to discover and develop novel therapeutics using RNAi is unproven andanticipated benefits of our Apthera acquisition may never lead to marketable products.not be realized.
1923
In addition, the actual integration may result in unanticipated adverse effects and unforeseen expenses, and the lossanticipated benefits of the technology licensed. We cannot necessarily control the amount or timing of resources that our contract partners will devote to our research and development programs, product candidates or potential product candidates, and we cannot guarantee that these parties will fulfill their obligations to us under these arrangements in a timely fashion. Weintegration plan may not be ablerealized. Actual cost synergies, if achieved at all, may be lower than expected and may take longer to readily terminate any such agreements with contract partners even if such contract partners doachieve than anticipated. If these challenges are not fulfilladequately addressed, RXi and Apthera may be unable to successfully integrate their obligationsoperations, or to us.realize the anticipated benefits of the integration of the two companies.
We are largely dependent on technologies we license, and if we lose the right to license such technologiessuccess of our two leading drug candidates neither of which may receive regulatory approval or we fail to license new technologies in the future, our ability to develop new products would be harmed.successfully commercialized.
We currentlyhave identified and are dependent on licenses from third parties for our key technologies relating to fundamental RNAi technologies. Our current licenses impose, and any future licenses we enter into are likely to impose, various development, funding, royalty, diligence, sublicensing, insurance and other obligations on us. If our license with respect to any of these technologies is terminated for any reason, the development of the products contemplated by the licenses would be delayed, or suspended altogether, while we seek to license similar technology or develop new non-infringing technology. The costs of obtaining new licenses are high, and many patents in the RNAi field have already been exclusively licensed to third parties, including our competitors. If any of our existing licenses are terminated, the development of the products contemplated by the licenses could be delayed or terminated and we may not be able to negotiate additional licenses on acceptable terms, if at all, which would have a material adverse effect on our business.
20
A number of different factors could prevent us from obtaining regulatory approval or commercializing our product candidates on a timely basis, or at all.
We, the FDA or other applicable regulatory authorities, or an institutional review board (“IRB”), which is an independent committee under the oversight of the United States Department of Health and Human Services (“HHS”), which that has been formally registered with HHS and functions to approve, monitor and review biomedical and behavioral research involving humans, may suspend clinical trials of a drug candidate at any time for various reasons, including if we or they believe the subjects or patients participating in such trials are being exposed to unacceptable health risks. Among other reasons, adverse side effects of a drug candidate on subjects or patients in a clinical trial could result in the FDA or other regulatory authorities suspending or terminating the trial and refusing to approve a particular drug candidate for any or all indications of use.
Clinical trials of a new drug candidate require the enrollment of a sufficient number of patients, including patients who are suffering from the disease the drug candidate is intended to treat and who meet other eligibility criteria. Rates of patient enrollment are affected by many factors, and delays in patient enrollment can result in increased costs and longer development times.
Clinical trials also require the review and oversight of IRBs, which approve and continually review clinical investigations and protect the rights and welfare of human subjects. An inability or delay in obtaining IRB approval could prevent or delay the initiation and completion of clinical trials, and the FDA may decide
24
not to consider any data or information derived from a clinical investigation not subject to initial and continuing IRB review and approval.
In addition, cancer vaccines are a relatively new form of therapeutic and a very limited number of such products have received regulatory approval. Therefore, the FDA or other regulatory authority may apply standards for approval of a new cancer vaccine that is different from past experience.
Numerous factors could affect the timing, cost or outcome of our drug development efforts, including the following:
| • | ||
| • | ||
| • | difficulty in securing centers to conduct trials, | |
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • |
21
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • |
It is possible that none of the product candidates that we develop will obtain the appropriate regulatory approvals necessary for us to begin selling them or that any regulatory approval to market a product may be subject to limitations on the indicated uses for which we may market the product. The time required to obtain FDA and other approvals is unpredictable but often can take years following the commencement of clinical trials, depending upon the complexity of the drug candidate. Any analysis we perform of data from clinical activities is subject to confirmation and interpretation by regulatory authorities, which could delay, limit or
25
prevent regulatory approval. Any delay or failure in obtaining required approvals could have a material adverse effect on our ability to generate revenue from the particular drug candidate.
We are also subject to numerous foreign regulatory requirements governing the conduct of clinical trials, manufacturing and marketing authorization, pricing and third-party reimbursement. The foreign regulatory approval process includes all of the risks associated with the FDA approval described above as well as risks attributable to the satisfaction of local regulations in foreign jurisdictions. Approval by the FDA does not assure approval by regulatory authorities outside of the United States.
The FDA has placed the IND for NeuVax on a partial clinical hold which precludes us from entering into a Phase III clinical trial of NeuVax.
On May 8, 2009, we submitted an SPA for a Phase III clinical trial for NeuVax. The FDA requires certain CMC information to be submitted prior to the FDA granting its approval to proceed with a Phase III trial. We did not include such CMC information in our SPA application. Although we received notification from the FDA of its acceptance of our SPA in June 2009, in July 2009, the FDA informed us that our IND application had been placed on “partial clinical hold” pending our submission and the FDA’s acceptance of the required CMC information. As a result, while we are allowed to continue semi-annual treatments of patients enrolled active Phase I/II trials, we are prohibited from initiating a Phase III clinical study until we have completed certain product manufacturing activities, submitted the required CMC information to the FDA and the FDA has approved such information and removed the partial clinical hold from the IND. Such actions will require us to expend additional funds to meet the FDA’s Phase III requirements, currently estimated by us to be approximately $2.5 million.
The approach we are taking to discover and develop novel therapeutics using RNAi is unproven and may never lead to marketable products.
RNA interference is a relatively new scientific discovery. The RNAi technologies that we have licensed or have created internally and that we intend to develop have not yet been clinically tested by us, nor are we aware of any clinical trials for efficacy having been completed by third parties involving these technologies. To date, neither we nor any other company has received regulatory approval to market therapeutics utilizing RNAi and a number of clinical trials of third parties’ RNAi technology have been unsuccessful. The scientific evidence to support the feasibility of developing drugs based on these discoveries is both preliminary and limited. To successfully develop RNAi-based products we must solve a number of issues, such as providing suitable methods of stabilizing the RNAi material and delivering it into target cells in the human body. We may spend large amounts of money trying to solve these issues and never succeed in doing so. In addition, any compounds that we develop may not demonstrate in patients the chemical and pharmacological properties ascribed to them in laboratory studies, and they may interact with human biological systems in unforeseen, ineffective or even harmful ways.
The FDA could impose a unique regulatory regime for RNAi therapeutics.
The substances we are intending to develop may represent a new class of drug, and the FDA has not yet established any definitive policies, practices or guidelines in relation to these drugs. While we expect any product candidates that we develop will be regulated as a new drug under the Federal Food, Drug, and Cosmetic Act, the FDA could decide to regulate them or other products we may develop as biologics under the Public Health Service Act. The lack of policies, practices or guidelines may hinder or slow review by the FDA of any regulatory filings that we may submit. Moreover, the FDA may respond to these submissions by defining requirements that we may not have anticipated.
The FDA approval process may be delayed for any drugs we develop that require the use of specialized drug delivery devices or vehicles.
Some drug candidates that we develop may need to be administered using specialized vehicles that deliver RNAi therapeutics directly to diseased parts of the body. For example, we may use an implantable
26
pump to deliver certain potential drug candidates to the nervous system. The drug delivery vehicles that we expect to deliver our drug candidates have not been approved by the FDA or other regulatory agencies. In addition, the FDA may regulate the product as a combination product of a drug and a device or require additional approvals or clearances for the modified delivery.
Further, to the extent the specialized delivery vehicle is owned by another company, we would need that company’s cooperation to implement the necessary changes to the vehicle, or its labeling, and to obtain any additional approvals or clearances. Any delays in finding suitable drug delivery vehicles to administer RNAi therapeutics directly to diseased parts of the body could negatively affect our ability to successfully develop our RNAi therapeutics.
22
We do not successful in developing pre-clinicalhave the facilities or expertise to manufacture supplies of any of our potential product candidates for clinical trials. Accordingly, we will be dependent upon contract manufacturers for these supplies. We currently manufacture limited quantities of our RXi-based product candidates for our research activities at our facility. There can be no assurance that we will be able to secure needed supply arrangements on attractive terms, or at all. Our failure to secure these arrangements as needed could have a materially adverse effect on our ability to complete the development of our product candidates or, if we obtain regulatory approval for our product candidates, to commercialize them.
Our current plans call for the manufacture of our compounds and, as necessary, any delivery vehicles that may be used to deliver our compounds by contract manufacturers offering research grade, Good Laboratory grade and Good Manufacturing Practices grade materials for preclinical studies (e.g. toxicology studies) and for clinical use. We anticipate the chemistry, manufacturing and controls for each active pharmaceutical ingredient will be addressed by our clinical development team in close collaboration with a contract manufacturer with extensive experience in drug synthesis. Certain of our product candidates are complex molecules requiring many synthesis steps, which may lead to challenges with purification andscale-up. These challenges could result in increased costs and delays in manufacturing.
Production and utilization of products using our technologies may require the development of new manufacturing technologies and expertise. We or our collaborators may be unable to successfully meet any of these technological challenges, or others that may arise in the course of development.
We may not be able to commence clinical trials in humansestablish or obtain approval formaintain the third party relationships that are necessary to develop or potentially commercialize some or all of our product candidates.
We are inexpect to depend on collaborators, partners, licensees, clinical research organizations and other third parties to support our discovery efforts, to formulate product candidates, to manufacture our product candidates, and to conduct clinical trials for some or all of our product candidates. We cannot guarantee that we will be able to successfully negotiate agreements for or maintain relationships with collaborators, partners, licensees, clinical investigators and other third parties on favorable terms, if at all. Our ability to successfully negotiate such agreements will depend on, among other things, potential partners’ evaluation of the new drug discovery phasesuperiority of our technology over competing technologies and the quality of the pre-clinical and clinical data that we have not yet identified any lead compounds for therapeutic development. RNA interference is a relatively new scientific field,generated, and the technologiesperceived risks specific to developing our product candidates. In addition, we recently reduced the scale of our RNAi operations, which could affect our ability to maintain or enter into new alliances. If we are still in the early stage of development. We have no compounds in pre-clinical toxicology studies, andunable to obtain or maintain these agreements, we may not be able to advanceclinically develop, formulate, manufacture, obtain regulatory approvals for or commercialize our product candidates. Under certain license agreements that we have already entered into, we have minimum dollar amounts per year that we are obligated to spend on the development of the technology we have licensed from our contract partners and other obligations to maintain certain licenses. If we fail to meet this requirement under any product candidate through the pre-clinical stage into clinical trials. Additionally,of our development effortslicenses that contain such requirements or any other obligations under these licenses, we may neverbe in breach of our obligations under such agreement, which may result in the identificationloss of the technology licensed. We cannot necessarily control the amount or timing of resources that our contract partners will devote to our research and development programs, product candidates or potential product candidates, and we cannot guarantee that these
27
parties will fulfill their obligations to us under these arrangements in a pre-clinical candidate which we are able to successfully develop into a drug. Even if we are able to designate a lead candidate, wetimely fashion. We may not be able to identify data that would support enteringreadily terminate any such a candidate into clinical trials. Furthermore,agreements with contract partners even if such contract partners do not fulfill their obligations to us.
In addition, we successfully enter into clinical studies,and other drug development companies receive notices from third parties from time to time that our or such other companies’ technology or product candidates infringe or may infringe the results from pre-clinical testingintellectual property rights of a drug candidatethose third parties. The assertion by third parties that our activities or product candidates infringe upon their intellectual property rights may not predict the results that will be obtained on human clinical trials.adversely affect our ability to secure strategic partners or licensees for our technology or product candidates or our ability to secure or maintain manufacturers for our compounds.
Even if we obtain regulatory approvals, our marketed drugs will be subject to ongoing regulatory review. If we fail to comply with continuing U.S. and foreign regulations, we could lose our approvals to market drugs and our business would be materially adversely affected.
Following any initial regulatory approval of any drugs we may develop, we will also beremain subject to continuing regulatory review, including the review of adverse drug experiences and clinical results that are reported after our drug products are made available to patients. This would include results from any post marketing tests or vigilance required as a condition of approval. The manufacturer and manufacturing facilities we use to make any of our drug candidatesproducts will also be subject to periodic review and inspection by the FDA. The discovery of any new or previously unknown problems with the product, manufacturer or facility may result in restrictions on the drug or manufacturer or facility, including withdrawal of the drug from the market. Our product promotion and advertising also willWe would continue to be subject to regulatorythe FDA requirements governing the labeling, packaging, storage, advertising, promotion, recordkeeping, and continuing regulatory review.submission of safety and other post-market information for all of our product candidates, even those which the FDA had approved. If we fail to comply with applicable continuing regulatory requirements, we may be subject to fines, suspension or withdrawal of regulatory approval, product recalls and seizures, operating restrictions and other adverse consequences.
Even if we receive regulatory approval to market our product candidates, our product candidates may not be accepted commercially, which may prevent us from becoming profitable.
The RNAi product candidates that we are developing are based on new technologies and therapeutic approaches. RNAi products may be more expensive to manufacture than traditional small molecule drugs, which may make them more costly than competing small molecule drugs. Additionally, for various applications, RNAi products are likely to require injection or implantation, and do not readily cross the so-called blood brain barrier, which will make them less convenient to administer than drugs administered orally. Key participants in the pharmaceutical marketplace, such as physicians, medical professionals working in large reference laboratories, public health laboratories and hospitals, third-partythird- party payors and consumers, may not accept products intended to improve therapeutic results based on RNAi technology. As a result, it may be more difficult for us to convince the medical community and third-party payors to accept and use our product, or to provide favorable reimbursement. And if medical professionals working with large reference laboratories, public health laboratories and hospitals choose not to adopt and use our RNAi technology, our products may not achieve broader market acceptance.
NeuVax and our other cancer-targeted product candidates will face many of the same commercial challenges facing our RNAi product candidates.
Other factors that we believe will materially affect market acceptance of our product candidates include:
| • | ||
| • | ||
| • | ||
| • | ||
2328
| • | ||
| • | the availability of government and third-party payor reimbursement, | |
| • | ||
| • |
We will be subject to competition and may not be able to compete successfully.
The biotechnology industry, including the cancer therapy vaccines market and RNAi research sector, is intensely competitive and involves a high degree of risk. We compete with other companies that have far greater experience and financial, research and technical resources than us. Potential competitors in the United States and worldwide are numerous and include pharmaceutical and biotechnology companies, educational institutions and research foundations, many of which have substantially greater capital resources, marketing experience, research and development staffs and facilities than us. Some of our competitors may develop and commercialize products that compete directly with those incorporating our technology, introduce products to market earlier than such products or on a more cost effective basis. We may be unable to effectively develop our technology or any other applications on a cost effective basis or otherwise. In addition, our technology may be subject to competition from other technology or methods developed using techniques other than those developed by traditional biotechnology methods. Our competitors compete with us in recruiting and retaining qualified scientific and management personnel as well as in acquiring technologies complementary to our technology. Our collaborators or we will face competition with respect to product efficacy and safety, ease of use and adaptability to various modes of administration, acceptance by physicians, the timing and scope of regulatory approvals, availability of resources, reimbursement coverage, price and patent position, including potentially dominant patent positions of others. An inability to successfully complete our product development could lead to us having limited prospects for establishing market share or generating revenues from our technology.
For patients with early stage breast cancer, adjuvant therapy is often given to prevent recurrence and increase the chance of long-term disease free survival. Adjuvant therapy for breast cancer can include chemotherapy, hormonal therapy, radiation therapy, or combinations thereof. In addition, the HER2 targeted drug trastuzumab (Herceptin®) may be given to patients with tumors with high expression of HER2 (IHC 3+).
There are a number of cancer vaccines in development for breast cancer, including but not limited to Lapuleucel-T (Dendreon), AE-37 (Antigen Express) and Stimuvax (Merck KgA). While these development candidates are aimed at a number of different targets, there is no guarantee that any of the these compounds will not in the future be indicated for treatment of low to intermediate HER 2 breast cancer patients and become directly competitive with NeuVax.
Similarly, a number of companies are using RNAi technologies, including for at least some of the disease indications we have been focusing our efforts on to date. Companies working in the RNAi area include: Alnylam Pharmaceuticals, Inc., Marina Biotech, Inc., Tacere Therapeutics, Inc., Benitec Limited, OPKO Health, Inc., Silence Therapeutics plc, Quark Pharmaceuticals, Inc., Rosetta Genomics Ltd., Lorus Therapeutics, Inc., Tekmira Pharmaceuticals Corporation, Calando Pharmaceuticals, Inc., Regulus Therapeutics Inc., and Santaris Pharmaceuticals, as well as a number of the large pharmaceutical companies.
Further, a number of companies are developing therapeutics for the same diseases we are targeting, including anti-scarring for which we are developing our first RNAi product candidate, using technologies other than RNA interference, and, for some of these diseases, there are existing therapeutics currently on the market. Most of these competitors have substantially greater research and development capabilities and financial, scientific, technical, manufacturing, marketing, distribution, and other resources than us, and we may not be able to successfully compete with them. In addition, even if we are successful in developing our product candidates, in order to compete successfully we may need to be first to market or to demonstrate that our RNAi based products are superior to therapies based on different technologies. A number of our competitors have already commenced clinical testing of RNAi product candidates and may be more advanced than are we
29
in the process of developing products. If we are not first to market or are unable to demonstrate such superiority, any products for which we are able to obtain approval may not be successful.
We are dependent on technologies we license, and if we lose the right to license such technologies or we fail to license new technologies in the future, our ability to develop new products would be harmed.
We currently are dependent on licenses from third parties for technologies relating to our product candidates. Our current licenses impose, and any future licenses we enter into are likely to impose, various development, funding, royalty, diligence, sublicensing, insurance and other obligations on us. If our license with respect to any of these technologies is terminated for any reason, the development of the products contemplated by the licenses would be delayed, or suspended altogether, while we seek to license similar technology or develop new non-infringing technology. The costs of obtaining new licenses are high, and many patents in the RNAi field have already been exclusively licensed to third parties, including our competitors. If any of our existing licenses are terminated, the development of the products contemplated by the licenses could be delayed or terminated and we may not be able to negotiate additional licenses on acceptable terms, if at all, which would have a material adverse effect on our business.
We may be unable to protect our intellectual property rights licensed from others parties, our intellectual property rights may be inadequate to prevent third parties from using our technologies or developing competing products, and we may need to license additional intellectual property from others.
We have a non-exclusive license to the Fire-Mello patent owned by UMMS and the Carnegie Institution of Washington, which claims various aspects of RNAi or genetic inhibition by double stranded RNA. This license continues to be available to third parties, and as such it does not provide us with the ability to exclude others from its use or protect us from competition. Therapeutic applications of gene silencing technologies, delivery methods, and other technologies that we license from third parties are claimed in a number of pending patent applications, but there can be no assurance that these applications will result in any issued patents or that those patents would withstand possible legal challenges or protect our technologies from competition. The United States Patent and Trademark Office and patent granting authorities in other countries have upheld stringent standards for the RNAi patents that have been prosecuted so far. Consequently, pending patents that we have licensed and those that we own may continue to experience long and difficult prosecution challenges and may ultimately issue with much narrower claims than those in the pending applications. We are aware of a number of issued patents covering various particular forms and compositions of RNAi-mediating molecules and therapeutic methods. Third parties may hold or seek to obtain additional patents that could make it more difficult or impossible for us to develop products based on RNAi technology without obtaining a license to such patents, which licenses may not be available on attractive terms or at all.
In addition, others may challenge the patents or patent applications that we currently license or may license in the future or that we own and, as a result, these patents could be narrowed, invalidated or rendered unenforceable, which would negatively affect our ability to exclude others from use ofusing RNAi technologies described in these patents. There can be no assurance that these patent or other pending applications or issued patents we license or that we own will withstand possible legal challenges. Moreover, the laws of some foreign countries may not protect our proprietary rights to the same extent as do the laws of the United States. Any patents issued to us or our licensors may not provide us with any competitive advantages, and there can be no assurance that the patents of others will not have an adverse effect on our ability to do business or to continue to use our technologies freely. Our efforts to enforce and maintain our intellectual property rights may not be successful and may result in substantial costs and diversion of management time. Even if our rights are valid, enforceable and broad in scope, competitors may develop products based on technology that is not covered by our licenses or patents or patent application that we own.
We may needhave received a letter from a third party claiming that we require access to such third party’s patents and patent applications and demanding that we stop engaging in unspecified alleged infringing activities unless we obtain a license additional intellectual property rights from such third partiesparty. We understand that other companies working in order to be able to complete the development or enhanceRNAi area have received similar letters from this third party. Although we do not believe, based on the efficacyadvice of our product candidates or avoid possible infringement of thepatent counsel, that our current and planned activities infringe any valid patent rights of others. Additionally, many of our UMMS licenses are limited to ALS, obesity, diabetes and cancer, and in order to pursue other diseases against proprietary gene targets, we may need licenses from othersuch third parties that mayparty, there can be unavailable. There is
30
no assurance that we will not need to alter our development candidates or products or obtain a license to such third party rights to avoid any such infringement.
There is no guarantee that future licenses will be able to acquire any additional intellectual property rightsavailable from third parties for either of our product candidates on satisfactory terms, or at all. To the extent that we are required and are able to obtain multiple licenses from third parties to develop or commercialize a product candidate, the aggregate licensing fees and milestones and royalty payments made to these parties may materially reduce our economic returns or even cause us to abandon development or commercialization of a product candidate.
There is also a risk that the products incorporating our peptide-based immunotherapy technology or otherwise marketed by us might infringe the patent, trademark or other intellectual property rights of third parties. We have not received any notice of any claims or threats of litigation based on any third party patent, trademark or other intellectual property rights; however, the lack of such a notice to date does not guarantee that we will not receive such a notice in the future, as frequently patent holders do not asset infringement until an alleged infringer is commercializing a product.
In addition to our licenses, we also rely on copyright and trademark protection, trade secrets, know-how, continuing technological innovation and licensing opportunities. In an effort to maintain the confidentiality and ownership of our trade secrets and proprietary information, we require our employees, consultants, advisors and others to whom we disclose confidential information to execute confidentiality and proprietary information agreements. However, it is possible that these agreements may be breached, invalidated or rendered unenforceable, and if so, there may not be an adequate corrective remedy available. Furthermore, like many companies
24
in our industry, we may from time to time hire scientific personnel formerly employed by other companies involved in one or more areas similar to the activities we conduct. In some situations, our confidentiality and proprietary information agreements may conflict with, or be subject to, the rights of third parties with whom our employees, consultants or advisors have prior employment or consulting relationships. Although we require our employees and consultants to maintain the confidentiality of all confidential information of previous employers, we or these individuals may be subject to allegations of trade secret misappropriation or other similar claims as a result of their prior affiliations. Finally, others may independently develop substantially equivalent proprietary information and techniques, or otherwise gain access to our trade secrets. Our failure to protect our proprietary information and techniques may inhibit or limit our ability to exclude certain competitors from the market and execute our business strategies.
Our success depends upon our ability to obtain and maintain intellectual property protection for our products and technologies.
Our success will depend on our ability to obtain and maintain adequate protection of our intellectual property covering our product candidates and technologies. The ultimate degree of patent protection that will be afforded to biotechnology products and processes, including ours, in the United States and in other important markets remains uncertain and is dependent upon the scope of protection decided upon by the patent offices, courts and lawmakers in these countries. There is no certainty that our existing patents, or patent applications if obtained, will afford us substantial protection or commercial benefit. Similarly, there is no assurance that our pending patent applications or patent applications licensed from third parties will ultimately be granted as patents or that those patents that have been issued or are issued in the future will stand if they are challenged in court. TheseThe applications based on RNAi technologies claim many different methods, compositions and processes relating to the discovery, development, delivery and commercialization of RNAi therapeutics. Because thethis field is so new, very few of these patent applications have been fully processed by government patent offices around the world, and there is a great deal of uncertainty about which patents will issue, when, to whom, and with what claims. While we are not aware of any litigation, threatened litigation or challenge to our intellectual property rights, itIt is likely that there will be significant litigation and other proceedings, such as interference and opposition proceedings in various patent offices, relating to patent rights in the RNAi field.field and that we may be a party to such proceedings.
There may be patent or other intellectual property rights belonging to others that require us to alter our products, pay licensing fees or cease certain activities. If our products infringe patent or other intellectual property rights of others, the owners of those rights could bring legal actions against us claiming damages and
31
seeking to enjoin manufacturingmanufacture, use, marketing and marketingsales of the affected products. If these legal actions are successful, in addition to any potential liability for damages, we could be required to obtain a license in order to continue to manufacture or market the affected products. We may not prevail in any action brought against us, and any license required under any rights that we infringe may not be available on acceptable terms or at all. Others may attempt to invalidate our intellectual property rights or those of our licensors. Even if our rights, or those of our licensors, are not directly challenged, disputes among third parties could lead to the weakening or invalidation of our intellectual property rights. Any attempt by third parties to circumventundermine or invalidate our intellectual property rights could be costly to defend, require significant time and attention of our management and have a material adverse effect on our business.
In addition, we anticipate that the issued United States patent covering the composition of matter of NeuVax that we have exclusively licensed will expire in 2015, and we have no equivalent patent protection outside of the United States. We are currently negotiating for exclusive rights to a patent application to which we already have non-exclusive rights that covers certain aspects of the method of treatment using NeuVax that could provide additional patent protection in major countries around the world through 2027, but there can be no assurance that we will successfully negotiate such a license.
If we are unable to obtain regulatory exclusivity for NeuVax, our business would be adversely affected and such exclusivity may not provide sufficient protection to prevent competitors from entering our markets.
Because our intellectual property rights to the composition of matter of NeuVax expire prior to commercialization, we expect to rely substantially on orphan drug designation, if granted for NeuVax, and data exclusivity provided under the Federal Food, Drug, and Cosmetic Act and similar laws in other countries. We are preparing to apply for Orphan Drug status for NeuVax which, if granted, could provide seven years or ten years of data exclusivity in the US or EU, respectively. However, there is no assurance that our Orphan Drug Application will be approved by either the FDA or EMEA. While we also anticipate that NeuVax will qualify for 12 years of data exclusivity, or the inability of another company to use our clinical data to support their application for regulatory approval, under the Patient Protection and Affordable Care Act; there can be no assurance that the 12 years of exclusivity provided for under the Patient Protection and Affordable Care Act will remain law, or that NeuVax will meet the qualifications of a “biological product” to receive the specified period of exclusivity.
While the orphan drug designation for NeuVax, if granted, will provide seven years of market exclusivity in the United States, we will not be able to exclude other companies from manufacturingand/or selling E75 beyond that timeframe. Even if we have orphan drug designation for a particular drug indication, we cannot guarantee that another company also holding orphan drug designation will not receive FDA approval for the same indication before we do. If that were to happen, our applications for that indication may not be approved until the competing company’s seven-year period of exclusivity expired. Even if we are the first to obtain FDA approval for an orphan drug indication, there are circumstances under which a competing product may be approved for the same indication during our seven-year period of marketing exclusivity, such as if the later product is shown to be clinically superior to the orphan product. Further, the seven-year marketing exclusivity would not prevent competitors from obtaining approval of the same compound for other indications or the use of other types of drugs for the same use as the orphan drug. In addition, data exclusivity does not prevent another company from completing its own clinical trials with NeuVax and obtaining regulatory approval for the same indication for which NeuVax may be approved. Consequently, we may not be able to prevent competitors from entering the market prior to the end of any applicable data exclusivity period. If we are not able to prevent competitors from entering the market with a similar product to NeuVax, our ability to achieve profits from sales of NeuVax will be dramatically limited.
We are subject to potential liabilities from clinical testing and future product liability claims.
If any of our future products are alleged to be defective, they may expose us to claims for personal injury by patients in clinical trials of our products or by patients using our commercially marketed products. Even if the marketing of one or more of our products is approved by the FDA, users may claim that such products
32
caused unintended adverse effects. We will seek to obtain clinical trial insurance for clinical trials that we conduct, as well as liability insurance for any products that we market. There can be no assurance that we will be able to obtain insurance in the amounts we seek, or at all. We anticipate that licensees who develop our products will carry liability insurance covering the clinical testing and marketing of those products. There is no assurance, however, that any insurance maintained by us or our licensees will prove adequate in the event
25
of a claim against us. Even if claims asserted against us are unsuccessful, they may divert management’s attention from our operations and we may have to incur substantial costs to defend such claims.
Any drugs we develop may become subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, which could have a material adverse effect on our business.
We intend to sell our products primarily to hospitals which receive reimbursement for the health care services they provide to their patients from third-party payors, such as Medicare, Medicaid and other domestic and international government programs, private insurance plans and managed care programs. Most third-party payors may deny reimbursement if they determine that a medical product was not used in accordance with cost-effective treatment methods, as determined by the third-party payor, or was used for an unapproved indication. Third-party payors also may refuse to reimburse for experimental procedures and devices. Furthermore, because our programs are in the early stages of development, we are unable at this time to determine their cost-effectiveness and the level or method of reimbursement for them. Increasingly, the third-party payors who reimburse patients are requiring that drug companies provide them with predetermined discounts from list prices, and are challenging the prices charged for medical products. If the price we are able to charge for any products we develop is inadequate in light of our development and other costs, our profitability could be adversely effected.
We currently expect that any drugs we develop may need to be administered under the supervision of a physician. Under currently applicable law, drugs that are not usually self-administered may be eligible for coverage by the Medicare program if:
| • | ||
| • | ||
| • | ||
| • |
There may be significant delays in obtaining insurance coverage for newly-approved drugs, and insurance coverage may be more limited than the purpose for which the drug is approved by the FDA. Moreover, eligibility for insurance coverage does not imply that any drug will be reimbursed in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim payments for new drugs, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement may be based on payments for other services and may reflect budgetary constraints or imperfections in Medicare data. Net prices for drugs may be reduced by mandatory discounts or rebates required by government health care programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement rates. Our inability to promptly obtain coverage and profitable reimbursement rates from both government-funded and private payors for new drugs that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to develop products, and our overall financial condition.
Additionally, third-party payors are increasingly attempting to contain health care costs by limiting both coverage and the level of reimbursement for medical products and services. Levels of reimbursement may decrease in the future, and future legislation, regulation or reimbursement policies of third-party payors may adversely affect the demand for and price levels of our products. If our customers are not reimbursed for our
33
products, they may reduce or discontinue purchases of our products, which could have a material adverse effect on our business, financial condition and results of operations.
Comprehensive health care reform legislation, which was recently adopted by Congress and was subsequently signed into law, could adversely affect our business and financial condition. Among other provisions, the legislation provides that a “biosimilar” product may be approved by the FDA on the basis of
26
analytical tests and certain clinical studies demonstrating that such product is highly similar to an existing, approved product and that switching between an existing product and the biosimilar product will not result in diminished safety or efficacy. This abbreviated regulatory approval process may result in increased competition if we are able to bring a product to market. The legislation also includes more stringent compliance programs for companies in various sectors of the life sciences industry with which we may need to comply and enhanced penalties for non-compliance with the new health care regulations. Complying with new regulations may divert management resources, and inadvertent failure to comply with new regulations may result in penalties being imposed on us.
Some states and localities have established drug importation programs for their citizens, and federal drug import legislation has been introduced in Congress. The Medicare Prescription Drug Plan legislation, which became law in December 2003, required the Secretary of Health and Human Services to promulgate regulations for drug reimportation from Canada into the United States under some circumstances, including when the drugs are sold at a lower price than in the United States. The Secretary, however, retained the discretion not to implement a drug reimportation plan if he finds that the benefits do not outweigh the costs, and has so far declined to approve a reimportation plan. Proponents of drug reimportation may attempt to pass legislation that would directly allow reimportation under certain circumstances. Legislation or regulations allowing the reimportation of drugs, if enacted, could decrease the price we receive for any products that we may develop and adversely affect our future revenues and prospects for profitability.
If our new management team is not effective or if we fail to attract, hire and retain qualified personnel, we may not be able to design, develop, market or sell our products or successfully manage our business.
Competition for skilled research, product development, regulatory and technical personnel also is intense, and we may not be able to recruit and retain the personnel we need. The loss of the services of any key research, product development, regulatory, and technical personnel, or our inability to hire new personnel with the requisite skills, could restrict our ability to develop our product candidates.
We use biological and hazardous materials and if we do not comply with laws regulating the protection of the environment and health and human safety, our business could be adversely affected.
Our research and development activities involve the controlled use of potentially harmful biological materials as well as hazardous materials, chemicals and various radioactive compounds. We cannot completely eliminate the risk of accidental contamination or injury; we could be held liable for any damages that result, and any liability could exceed our resources. We are subject to federal, state and local laws and regulations governing the use, storage, handling and disposal of these materials and specific waste products. We are also subject to numerous environmental, health and workplace safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of biohazardous materials. The cost of compliance with these laws and regulations could be significant and may adversely affect capital expenditures to the extent we are required to procure expensive capital equipment to meet regulatory requirements.
34
We are also subject to numerous environmental, health and workplace safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of biohazardous materials. We maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of these materials. The limits of our workers’ compensation insurance are mandated by state law, and our workers’ compensation liability is capped at these state-mandated limits. We do not maintain insurance for environmental liability or toxic tort claims
27
that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials. Additional federal, state and local laws and regulations affecting our operations may be adopted in the future. We may incur substantial costs to comply with, and substantial fines or penalties if we violate any of these laws or regulations.
Risks Relating Our Financial Position and Capital Requirements
We may not be able to obtain sufficient financing, and may not be able to develop our product candidates.
We believe that our existing cash and cash equivalentequivalents should be sufficient to fund our operations through at least the first halfquarter of 2011.2012. In the future, we will be dependent on obtaining further financing from third parties in order to maintain our operations and to meet our financial obligations. In addition, until its expiration on January 30, 2011, we also have available to us the SEDA that we entered into on January 30, 2009. However, before we are able to access additional capital from the SEDA, we must satisfy certain conditions, including the requirement that shares of our stock to be sold to YA Global be registered with the U.S. Securities and Exchange Commission (“SEC”), and there is risk of delays in our satisfying these conditions. We cannot assure that additional debt or equity or other funding to maintain our operations and to meet our obligations to our licensors will be available to us in the future on acceptable terms, or at all. If we fail to obtain additional funding when needed, we would be forced to scale back, or terminate, our operations, or to seek to merge with or to be acquired by another company.
We anticipate that we will need to raise substantial amounts of money to fund a variety of future activities integral to the development of our business, which may include but are not limited to the following:
| • | ||
| • | to conduct a Phase III clinical trial for NeuVax, | |
| • | to file the IND for RXI-109 and commence a Phase I clinical trial, | |
| • | to conduct research and development to successfully develop our RNAi technologies, | |
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • |
We cannot assure you that any financing needed for the development of our business will be available to us on acceptable terms or at all. If we cannot obtain additional financing in the future, our operations may be restricted and we may ultimately be unable to continue to develop and potentially commercialize our product candidates.
We expect to continue to incur significant research and development expenses, which may make it difficult for us to attain profitability, and may lead to uncertainty about or as to our ability to continue as a going concern.
Substantial funds were expended to develop our RNAi technologies and product candidates, and additional substantial funds will be required for further research and development, including pre-clinical testing and
35
clinical trials of any product candidates, and to manufacture and market any products that are approved for commercial sale. Because the successful development of our products is uncertain, we are unable to precisely estimate the actual funds we will require to develop and potentially commercialize them. In addition, we may not be able to generate enough revenue, even if we are able to commercialize any of our product candidates, to become profitable.
In the event that we are unable to achieve or sustain profitability or to secure additional financing, we may not be able to meet our obligations as they come due, raising substantial doubts as to our ability to
28
continue as a going concern. Any such inability to continue as a going concern may result in our common stock holders losing their entire investment. There is no guaranty that we will become profitable or secure additional financing. Our financial statements contemplate that we will continue as a going concern and do not contain any adjustments that might result if we were unable to continue as a going concern. Changes in our operating plans, our existing and anticipated working capital needs, the acceleration or modification of our expansion plans, increased expenses, potential acquisitions or other events will all affect our ability to continue as a going concern.
Future financing may be obtained through, and future development efforts may be paid for by, the issuance of debt or equity, which may have an adverse effect on our stockholders or may otherwise adversely affect our business.
If we raise funds through the issuance of debt or equity, any debt securities or preferred stock issued will have rights, preferences and privileges senior to those of holders of our common stock in the event of a liquidation. In such event, there is a possibility that once all senior claims are settled, there may be no assets remaining to pay out to the holders of common stock. In addition, if we raise funds through the issuance of additional equity, whether through private placements or additional public offerings, such an issuance would dilute your ownership in us.
The terms of debt securities may also impose restrictions on our operations, which may include limiting our ability to incur additional indebtedness, to pay dividends on or repurchase our capital stock, or to make certain acquisitions or investments. In addition, we may be subject to covenants requiring us to satisfy certain financial tests and ratios, and our ability to satisfy such covenants may be affected by events outside of our control.
You may have difficulty evaluating our business, because we have limited history and our historical financial information may not be representative of our future results.
We have limited operating experience and may not be able to effectively operate.
We are a discovery-stagedevelopment-stage company with limited operating history. We will focus solely on developing and, if we obtain regulatory approval, forcommercializing our product candidates, commercializing therapeutic products based upon RNAi technologies, and there is no assurance that we will be able to successfully implement our business plan. While our management collectively possesses substantial business and scientific experience, there is no assurance that we will be able to manage our business effectively, or that we will be able to identify, hire and retain any needed additional management or scientific personnel to develop and implement our product development plans, obtain third-party contracts or any needed financing, or achieve the other components of our business plan.
The obligations associated with being an independent public company require significant resources and management attention.
As a publicly traded company, we are subject to the reporting requirements of the U.S. Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the Sarbanes-Oxley Act of 2002. In addition,
29
the Exchange Act requires that we file annual, quarterly and current reports. Our failure to prepare and disclose this information in a timely manner could subject us to penalties under federal securities laws, expose us to lawsuits and restrict our ability to access financing. The Sarbanes-Oxley Act requires that we, among other things, establish and maintain effective internal controls and procedures for financial reporting. From time to time we evaluate our existing internal controls in light of the standards adopted by the Public Company Accounting Oversight Board. It is possible that we or our independent registered public accounting firm may identify significant deficiencies or material weaknesses in our internal control over financial reporting in the future. Any failure or difficulties in implementing and maintaining these controls could cause us to fail to meet the periodic reporting obligations or result in material misstatements in our financial statements.
36
Section 404 of the Sarbanes-Oxley Act requires annual management assessments of the effectiveness of our internal control over financial reporting. Our failure to satisfy the requirements of Section 404 on a timely basis could result in the loss of investor confidence in the reliability of our financial statements, which in turn could have a material adverse effect on our business and our common stock.
Risks Related to Ownership of Our Common Stock
The market price and trading volume of our common stock may be volatilevolatile.
The market price of our common stock could fluctuate significantly for many reasons, including the following factors:
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • | ||
| • |
In addition, factors beyond our control may also have an impact on the price of our stock. For example, to the extent that other large companies within our industry experience declines in their stock price, our stock price may decline as well. In addition, when the market price of a company’s common stock drops significantly, stockholders often institute securities class action lawsuits against the company. A lawsuit against us could cause us to incur substantial costs and could divert the time and attention of our management and other resources.
30
Anti-takeover provisions of our certificate of incorporation and by-laws and provisions of Delaware law could delay or prevent a change of control that you may favor.
Anti-takeover provisions of our certificate of incorporation and by-laws and provisions of Delaware law may discourage, delay or prevent a merger or other change of control that stockholders may consider favorable, or may impede the ability of the holders of our common stock to change our management. These provisions of our certificate of incorporation and by-laws, among other things:
| • | ||
| • | ||
| • | ||
| • |
In addition, Section 203 of the Delaware General Corporation Law provides that, subject to limited exceptions, persons that acquire, or are affiliated with a person that acquires, more than 15% of the outstanding voting stock of a Delaware corporation such as our company shall not engage in any business combination with that corporation, including by merger, consolidation or acquisitions of additional shares for a
37
three-year period following the date on which that person or its affiliate crosses the 15% stock ownership threshold. Section 203 could operate to delay or prevent a change of control of our company.
31
We may acquire other businesses or form joint ventures that may be unsuccessful and could adversely dilute your ownership of our company.
As part of our business strategy, we may pursue future acquisitions of other complementary businesses and technology licensing arrangements. We also may pursue strategic alliances. We have no experience with respect to acquiring other companies and limited experience with respect to the formation of collaborations, strategic alliances and joint ventures. If we wereOur acquisition of Apthera creates significant risks for the Company. See “The anticipated benefits of our Apthera acquisition may not be realized” for risks specific to make any acquisitions, wethe Apthera acquisition. We may not be able to integrate theseApthera or other acquisitions successfully into our existing business and we could assume unknown or contingent liabilities. We also could experience adverse effects on our reported results of operations from acquisition related charges, amortization of acquired technology and other intangibles and impairment charges relating to write-offs of goodwill and other intangible assets from time to time following the acquisition. Integration of an acquired company also may requirerequires management resources that otherwise would be available for ongoing development of our existing business. We may not identify or complete these transactions in a timely manner, on a cost-effective basis, or at all, and we may not realize the anticipated benefits of any acquisition, technology license or strategic alliance.
To finance future acquisitions, we may choose to issue shares of our common stock as consideration, which would dilute your ownership interest in us. Alternatively, it may be necessary for us to raise additional funds through public or private financings. Additional funds may not be available on terms that are favorable to us and, in the case of equity financings, may result in dilution to our stockholders. Any future acquisitions by us also could result in large and immediate write-offs, the incurrence of contingent liabilities or amortization of expenses related to acquired intangible assets, any of which could harm our operating results.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
On September 25, 2007, we entered into a lease agreement with Newgate Properties, LLC (an affiliate of Worcester Polytechnic Institute), for our facility located at 60 Prescott Street, Worcester, Massachusetts. The lease has a term of 20 months. The facility is approximately 6,800 square feet, of which 5,600 square feet is laboratory space used for research and development and the additional 1,200 square feet is used for general and administrative offices. On January 23, 2009, we extended our lease for an additional two years through July 31, 2011. The monthly rental fee is approximately $19,000. We believe that the space is suitable for our current needs.
| ITEM 3. | LEGAL PROCEEDINGS |
Although we are not currently involved in any legal proceedings, from time to time, we may become a party to various legal actions and complaints arising in the ordinary course of business.
| ITEM 4. | RESERVED |
38
PART II
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock has been listed on the Nasdaq Capital Market under the symbol “RXII” since March 12, 2008. Prior to that, there was no established public market for our common stock. The following
32
table sets forth for the periods indicated the high and low sales prices of our common stock on the Nasdaq Capital Market:
Year Ended December 31, 2008 | High | Low | ||||||||||||||
Year Ended December 31, 2009 | High | Low | ||||||||||||||
| First Quarter | $ | 23.95 | $ | 6.01 | $ | 7.19 | $ | 2.71 | ||||||||
| Second Quarter | $ | 10.12 | $ | 5.22 | $ | 7.57 | $ | 4.00 | ||||||||
| Third Quarter | $ | 9.05 | $ | 6.42 | $ | 4.93 | $ | 2.44 | ||||||||
| Fourth Quarter | $ | 12.25 | $ | 5.41 | $ | 4.84 | $ | 1.51 | ||||||||
Year Ended December 31, 2009 | High | Low | ||||||||||||||
Year Ended December 31, 2010 | High | Low | ||||||||||||||
| First Quarter | $ | 7.19 | $ | 2.71 | $ | 8.11 | $ | 3.72 | ||||||||
| Second Quarter | $ | 7.57 | $ | 4.00 | $ | 4.71 | $ | 2.56 | ||||||||
| Third Quarter | $ | 4.93 | $ | 2.44 | $ | 2.85 | $ | 1.75 | ||||||||
| Fourth Quarter | $ | 4.84 | $ | 1.51 | $ | 3.85 | $ | 2.41 | ||||||||
Holders
As of March 29, 2010,April 13, 2011, there were approximately 631628 holders of record of our common stock. Because many of our shares are held by brokers and other institutions on behalf of stockholders, we are unable to estimate the total number of individual stockholders represented by these records.holders of record.
Dividends
We have never paid any dividends and do not anticipate paying any dividends on our common stock in the foreseeable future. We expect to retain future earnings, if any, for use in our development activities and the operation of our business. The payment of any future dividends will be subject to the discretion of our board of directors and will depend, among other things, upon our results of operations, financial condition, cash requirements, prospects and other factors that our board of directors may deem relevant. Additionally, our ability to pay future dividends may be restricted by the terms of any debt financing.
Securities authorized for issuance under equity compensation plans
Information relating to our equity compensation plans will be included in our proxy statement in connection with our 20102011 Annual Meeting of Stockholders, under the caption “Equity Compensation Plan Information”.Information.” The relevant portion of our proxy statement is incorporated herein by reference.
Performance Graph
Because we are a smaller reporting company, we are not required to provide this information.
Recent Sales of Unregistered Securities
Set forth below is information regarding shares of common stock and preferred stock issued, and options and warrants granted, by us during the period covered by this report. Also included is the consideration, if any, received by us for such shares, options and warrants and information relating to the section of the Securities Act, or rule of the SEC under which exemption from registration was claimed.
39
Preferred Stock
There were no unregistered shares of preferred stock issued by us during the period covered by this report.
Common Stock
There were no unregistered shares of common stock issued by us during the period covered by this report.
33
| ITEM 6. | SELECTED FINANCIAL DATA |
Because we are a smaller reporting company, we are not required to provide the information required by this Item.
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
You should read the following discussion in conjunction with the RXi financial statements and the notes to financial statements included elsewhere in this annual report. This “Management’s Discussion and Analysis of Financial Condition and Results of Operations” section contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. For a discussion of indicators of forward-looking statements and specific important factors that could cause actual results to differ materially from those contained in forward-looking statements, see “Risk Factors” under Part I — Item 1A of this Annual Report onForm 10-K for the fiscal year ended December 31, 2009.2010. This Management’s Discussion and Analysis of Financial Condition and Results of Operations should be read and interpreted in light of such factors.
Our actual results and the timing of certain events could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including those discussed below and elsewhere in this annual report.
Overview
We are a discovery-stage biopharmaceuticalbiotechnology company focussed on discovering, developing and commercializing innovative therapies addressing major unmet medical needs using RNAi-targeted and immunotherapy technologies. We are pursuing (1) cancer therapies utilizing peptide-based immunotherapy products, including our main product candidate NeuVaxtm, for the treatment of various cancers and (2) proprietary therapeutics based on RNA interference, or “RNAi,”“RNAi”, a naturally occurring cellular mechanism that has the potential to effectively and selectively interfere with, or “silence,”“silence”, expression of targeted disease-associated genes. It is believed that this specific silencing can be used to potentially treat human diseases by “turning off” genes that lead to disease.
| • | Immunotherapy Products. Our main product candidate is NeuVax, which is a peptide-based immunotherapy to reduce the recurrence of breast cancer inlow-to-intermediate HER2-positive breast cancer patients not eligible for Herceptin®. We expect NeuVax to enter Phase III clinical trials in this breast cancer patient population during the first half of 2012 if we are able to satisfy certain United States Food and Drug Administration (“FDA”) information requirements to be released from a clinical hold to commence the trial. In addition, based on our clinical trials, we believe that NeuVax has the potential to treat other cancers, including prostate, bladder and ovarian cancers. | |
| • | RNAi Products. We believe that certain human diseases can potentially be treated by silencing targeted genes that lead to disease. While no therapeutic RNAi products have been approved by the FDA to date, there has been significant interest and growth in the field of RNAi therapeutic development. This growth is driven by the potential ability to use RNAi to rapidly develop lead compounds that |
40
| specifically and selectively inhibit a target gene, many of which are thought to be undruggable by other modalities. RXI-109, our first RNAi product candidate, is a dermal anti-scarring therapy that targets CTGF (connective tissue growth factor). We are currently working towards filing an investigational new drug application (“IND”) for RXI-109 in the second half of 2011 and commencing a Phase I clinical trial in the first half of 2012. We intend to maintain our core RNAi discovery and development capability to advance current collaborations, as well as enable alliances. We believe that RXI-109 may be able to treat other indications, including pulmonary fibrosis, liver fibrosis, acute spinal injury, ocular scarring and restinosis. |
Background on the Company and our expertiseRecent Change in RNAi to identify lead compounds against multiple target genes, and advance them towards pre-clinical and clinical development in therapeutic areas that address broad unmet medical needs, in both acute and chronic settings. By utilizing our expertise in RNAi and the comprehensive RNAi therapeutic platform that we have established, we believe we will be able to discover and develop lead compounds and progress them into and through clinical development for potential commercialization more efficiently than traditional drug development approaches.Strategic Focus
We were formed in 2006 by CytRx Corporation (“CytRx”) (Nasdaq: CYTR) and four prominent RNAi researchers, including Dr. Craig Mello, who was awarded the 2006 Nobel Prize in Medicine for his co-discovery of RNAi. From 2003 through 2006, CytRx sponsored therapeutic RNAi research at the University of Massachusetts Medical School, or “UMMS,”(“UMMS”) and Massachusetts General Hospital. We commenced operations in January 2007 after CytRx contributed to us its portfolio of RNAi therapeutic assets in exchange for approximately 7.04 million shares of our common stock. These assets consisted primarily of RNAi licenses and related intellectual property and a nominal amount of equipment. The cost of the licenses had previously been expensed by CytRx as in-process research and development and was recorded in the predecessor financial statements at cost.
34
at establishing research and development partnerships with pharmaceutical and larger biotechnology companies.
Research and Development
Following the Apthera acquisition, our researching and development programs will focus on developing (1) cancer therapies utilizing peptide-based immunotherapy products, including our main product candidate NeuVax, for the treatment of various cancers and (2) proprietary therapeutics based on RNA interference, or “RNAi”, a naturally occurring cellular mechanism that has the potential to effectively and selectively interfere with, or “silence”, expression of targeted disease-associated genes.
Since we commenced operations, research and development has comprised a significant proportion of our total operating expenses and is expected to comprise the majority of our spending for the foreseeable future.
There are risks in any new field of drug discovery that preclude certainty regarding the successful development of a product. We cannot reasonably estimate or know the nature, timing and costs of the efforts
41
necessary to complete the development of, or the period in which material net cash inflows are expected to commence from, any product candidate. Our inability to make these estimates results from the uncertainty of numerous factors, including but not limited to:
| • | Our ability to advance product candidates into pre-clinical research and clinical trials; | |
| • | The scope and rate of progress of our pre-clinical program and other research and development activities; | |
| • | The scope, rate of progress and cost of any clinical trials we commence; | |
| • | The cost of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; | |
| • | Clinical trial results; | |
| • | The terms and timing of any collaborative, licensing and other arrangements that we may establish; | |
| • | The cost and timing of regulatory approvals; | |
| • | The cost of establishing clinical and commercial supplies of our product candidates and any products that we may develop; | |
| • | The cost and timing of establishing sales, marketing and distribution capabilities; |
35
| • | The effect of competing technological and market developments; and | |
| • | The effect of government regulation and insurance industry efforts to control healthcare costs through reimbursement policy and other cost management strategies. |
Failure to complete any stage of the development of our product candidates in a timely manner could have a material adverse effect on our operations, financial position and liquidity.
License Agreements
We have entered into licensing relationships with academic institutions, research foundations and commercial entities, and may seek to enter into additional licenses with pharmaceutical and biotechnology companies. We also may enter into strategic alliances to expand our RNAi intellectual property portfolio and to potentially accelerate our development programs by gaining access to technology and funding, including equity sales, license fees and other revenues. For each product that we develop that is covered by the patents licensed to us including theour material licenses discussed below,elsewhere in this annual report, we are obligated to make additional payments upon the attainment of certain specified product development milestones.
36
Critical Accounting Policies and Estimates
Use of Estimates
Management’s discussion and analysis of our financial condition and results of operations include the financial statements as of and for the years ended December 31, 20092010 and 2008.2009. The preparation of these financial statements requires management to make estimates, allocations and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent assets and liabilities. On an ongoing basis, management evaluates its estimates, including those related to impairment of long-lived assets, accrued liabilities and certain expenses. We base our estimates about the carrying values of assets and liabilities that are not readily apparent from other sources on historical experience and on other assumptions believed to be reasonable under the circumstances. Actual results may differ materially from these estimates under different assumptions or conditions. Additionally, the financial information included here may not necessarily reflect the financial position, operating results, changes in our invested equity and cash flows in
42
the future or what they would have been had we been a separate, stand-alone entity during the periods presented.
Our significant accounting policies are summarized in the footnotes to our financial statements. We believe the following critical accounting policies involve significant judgments and estimates used in the preparation of our financial statements.
Research and Development Expenses
Research and development costs are expensed as incurred. Included in research and development costs are wages, benefits and other operating costs, facilities, supplies, external services and overhead directly related to our research and development departments as well as costs to acquire technology licenses.
Stock-Based Compensation
We follow the provisions of the Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 718, ““Compensation — Stock Compensation”Compensation” (“ASC 718”), which requires the measurement and recognition of compensation expense for all share-based payment awards made to employees, non-employee directors, and consultants, including employee stock options. Stock compensation expense based on the grant date fair value estimated in accordance with the provisions of ASC 718 is recognized as an expense over the requisite service period.
For stock options granted as consideration for services rendered by non-employees, we recognize compensation expense in accordance with the requirements of FASB ASC Topic505-50 (“ASC505-50”), ““Equity Based Payments to Non-Employees.Non- Employees.”Non-employee option grants that do not vest immediately upon grant are recorded as an expense over the vesting period of the underlying stock options. At the end of each financial reporting period prior to vesting, the value of these options, as calculated using the Black-Scholes option-pricing model, will be re-measured using the fair value of our common stock and the non-cash compensation recognized during the period will be adjusted accordingly. Since the fair market value of options granted to non-employees is subject to change in the future, the amount of the future compensation expense will include fair value re-measurements until the stock options are fully vested.
37
The fair value of each option grant is estimated using the Black-Scholes option-pricing model, with the following weighted average assumptions:
| 2009 | 2008 | 2010 | 2009 | |||||
| Weighted average risk free interest rate | 1.55% - 3.91% | 1.55% - 3.99% | 1.88% - 3.28% | 1.55% - 3.91% | ||||
| Weighted average volatility | 116.72% - 122.93% | 101.79% - 116.74% | 118.3% - 133.62% | 116.72% - 122.93% | ||||
| Expected lives (years) | 6 - 10 | 6 - 10 | 6 - 10 | 6 - 10 | ||||
| Expected dividend yield | 0% | 0% | 0% | 0% |
Our expected common stock price volatility assumption is based upon the volatility of a basket of comparable companies. The expected life assumptions for employee grants were based upon the simplified method provided for under ASC 718, which averages the contractual term of our options of ten years with the average vesting term of four years for an average of six years. The expected life assumptions for non-employees were based upon the contractual term of the option. The dividend yield assumption of zero is based upon the fact that we have never paid cash dividends and presently have no intention of paying cash dividends in the future. The risk-free interest rate used for each grant was also based upon prevailing short-term interest rates.
We have an estimated an annualized forfeiture rate of 4.0%15.0% for options granted to our employees, 2.1%and 8.0% for options granted to senior management and no forfeiture rate for the directors. We record additional expense if the actual forfeitures are lower than estimated and will record a recovery of prior expense if the actual forfeiture rates are higher than estimated.
43
Valuation of Common Stock
Our common stock was registered and began trading publicly on March 12, 2008. As a result, the actual value of a common share may be materially different than the fair value per share prior to that date.
Derivative Financial Instruments
During the normal course of business, from time to time, we issue warrants and options to vendors as consideration to perform services. We may also issue warrants as part of a debt or equity financing. We do not enter into any derivative contracts for speculative purposes.
We recognize all derivatives as assets or liabilities measured at fair value with changes in fair value of derivatives reflected as current period income or loss unless the derivatives qualify for hedge accounting and are accounted for as such. During the yearyears ended December 31, 2010 and 2009, we issued warrants to purchase 540,000 and 978,142 shares of common stock, respectively, in connection with an equity transaction. In accordance with ASC TopicTopic 815-40, ““Derivatives and Hedging — Contracts in Entity’s Own Stock”(“ASC815-40”), the value of these warrants is required to be recorded as a liability, as the holders have an option to put the warrants back to us in certain events, as defined.
Results of Operations
For the year ended December 31, 2009,2010, our net loss was approximately $18,387,000,$11,993,000, compared with a net loss of $14,373,000$18,387,000 for the year ended December 31, 2008. The loss increased by $4,014,000 or approximately 28%.2009. Reasons for the variations in the losses between the years are discussed below.
Revenue
Since we are a development-stage biopharmaceutical company, we have not generated any revenues since inception through December 31, 2009.2010.
38
Research and Development Expense (in thousands)
| For the Years Ended December 31, | ||||||||||||||||
| 2009 | 2008 | For the Years Ended December 31, | ||||||||||||||
| (In thousands) | 2010 | 2009 | ||||||||||||||
| Research and development expense | $ | 6,728 | $ | 5,105 | $ | 6,046 | $ | 6,728 | ||||||||
| Research and development employee stock-based compensation expense | 867 | 336 | 1,084 | 867 | ||||||||||||
| Research and development non-employee stock-based compensation expense | 1,297 | 1,613 | 743 | 1,297 | ||||||||||||
Total research and development expense | $ | 8,892 | $ | 7,054 | $ | 7,873 | $ | 8,892 | ||||||||
Total research and development expenses for the year ended December 31, 20092010 were approximately $7,873,000 as compared to $8,892,000 or 51% of our total operating expenses incurred. For the year ended December 31, 2008, total research and development expenses were approximately $7,054,000, or 48%, of our total operating expenses incurred, an increase of approximately $1,838,000, or 26%. Research and development expenses increased $1,623,000, or 32%, from $5,105,000 in the year ended December 31, 2008 to $6,728,000 infor the year ended December 31, 2009. This increaseThe decrease of $1,019,000 or 11% was primarily due to increasesa decrease of $554,000 in non-employee non-cash stock based compensation and a $682,000 decrease in research and development cash expenses related to the timing of patent application and prosecution on internal discoveries, offset by an increase of $217,000 in costs associated with employee non-cash stock based compensation resulting from an increase in headcount as well as an increase in patent costsprimarily related to patent applications on internal discoveries.
44
Research and Development Non-Employee Stock-Based Compensation Expense
We issued options to purchase shares of our common stock as compensation to SAB members and consultants. For financial statement purposes, we valued these shares at their fair value. Fluctuations in non-employee stock-based compensation expense results from variations in the number of common stock options issued, vesting schedules and the Black-Scholes fair values of common stock options granted to SAB members.
39
General and Administrative Expense (in thousands)
| For the Years Ended | ||||||||||||||||
| December 31, | For the Years Ended | |||||||||||||||
| 2009 | 2008 | December 31, | ||||||||||||||
| (In thousands) | 2010 | 2009 | ||||||||||||||
| General and administrative expenses | $ | 5,483 | $ | 4,874 | $ | 5,493 | $ | 5,483 | ||||||||
| Common stock warrants issued for general and administrative expense | 826 | 750 | 718 | 826 | ||||||||||||
| Fair value of common stock issued in exchange for general and administrative expenses | 281 | — | — | 281 | ||||||||||||
| Common stock and stock options issued for general and administrative expense | 2,038 | 1,875 | 2,541 | 2,038 | ||||||||||||
Total general and administrative expense | $ | 8,628 | $ | 7,499 | $ | 8,752 | $ | 8,628 | ||||||||
General and administrative expenses include compensation-related costs for our employees dedicated to general and administrative activities, legal fees, audit and tax fees, consultants and professional services, business development and corporate partnership opportunities and general corporate expenses.
From time to time, we expect to issue shares of our common stock or warrants or options to purchase shares of our common stock to consultants and other service providers in exchange for services. For financial statement purposes, we will value these shares of common stock, common stock options, and warrants at their fair value, or at the value of the services received, whichever is more reliably measurable.
Interest Income (Expense)
Interest income (expense) was negligible for both the year ended December 31, 2009, compared with approximately $180,000 for the year ended2010, and December 31, 2008. This decrease was primarily due to current interest rates available to us on our cash and cash equivalents during the twelve months ended December 31, 2009, as compared with the twelve months ended December 31, 2008.2009. The key objectives of our investment policy are to preserve principal and ensure sufficient liquidity, so our invested cash may not earn as high a level of income as longer-term or higher risk securities, which generally have less liquidity and more volatility. The interest rates available on lower risk, shorter-term investments in today’s market are lower than rates available in the prior period. We expect to have interest income in future periods based on our current account balances and potentially from additional capital we may receive in the future.
45
Other ExpenseIncome (expense)
Other expenses were $862,000income (expense) is summarized as follows (in thousands):
| For the Years Ended December 31, | ||||||||
| 2010 | 2009 | |||||||
| Change in fair value of common stock warrants issued | $ | 3,049 | $ | (858 | ) | |||
| Reduction of potential redemption liability | 785 | — | ||||||
| Other income | 793 | (4 | ) | |||||
| Other income (expense), net | $ | 4,627 | $ | (862 | ) | |||
Other income (expense) was $4,627,000 and $(862,000) for the yearyears ended December 31, 2009. Approximately $858,0002010 and 2009, respectively. The overall increase of this expense relates$5,489,000, or 637%, was due to an increase of $3,907,000 attributable to the change in fair value of common stock warrants issued, the reduction of potential redemption liability of $785,000, and an increase of $797,000 of other income, representing primarily an increase in connection with the 2009 Offering.grant income.
Income Taxes
There was no income tax expense for the years ended December 31, 20092010 and 20082009 due to the fact that we have incurred significant tax losses since we began operations. A tax benefit would have been recorded for losses however, due to the uncertainty of realizing these assets, a valuation allowance was recognized which fully offset the deferred income tax assets.
40
Liquidity and Capital Resources
We had cash and cash equivalents of approximately $5.7$6.9 million as of December 31, 20092010 and approximately $14.4$11.0 million as of March 31, 2010.
We have not hadgenerated revenue to date and may not generated any revenues since inception nor are any revenues expected forgenerate product revenue in the foreseeable future.future, if ever. We expect to incur significant operating losses for the foreseeable future whileas we advance our future product candidates from discovery through pre-clinical studiesthe drug development and clinical trials and seek regulatory approval and potential commercialization, even if we are collaborating with pharmaceutical and larger biotechnology companies.process. In addition to these increasing research and development expenses, we expect general and administrative costs to increase as we recruit additional managementadd personnel and administrative personnel.integrate Apthera. We will need to generate significant revenues to achieve profitability and might never do so. In the absence of product revenues, our potential sources of operational funding are expected to be the proceeds from the sale of equity, funded research and development payments and payments received under partnership and collaborative agreements.
As a result of our acquisition of Apthera and the expenses expected to be incurred in connection with the Phase III clinical trial for NeuVax, we expect that our expenses will increase significantly from historic levels for the foreseeable future. We believe that our existing cash and cash equivalents should be sufficient to fund our operations through at least the first halfquarter of 2011. In addition, we also have available to us the SEDA which expires on January 30, 2011.2012. In the future, we will be dependent on obtaining funding from third parties, such as proceeds from the sale of equity, funded research and development payments and payments under partnership and collaborative agreements, in order to maintain our operations and meet our obligations to licensors. There is no guarantee that debt, additional equity or other funding will be available to us on acceptable terms, or at all. If we fail to obtain additional funding when needed, we would be forced to scale back, or terminate, our operations or to seek to merge with or to be acquired by another company.
We believe that our existing cash and cash equivalents should be sufficient to fund our operations through at least the first quarter of 2012. In the future, we will be dependent on obtaining funding from third parties such as proceeds from the sale of equity, funded research and development payments and payments under partnership and collaborative agreements, in order to maintain our operations and meet our obligations to licensors. There is no guarantee that debt, additional equity or other funding will be available to us on
46
acceptable terms, or at all. If we fail to obtain additional funding when needed, we would be forced to scale back, or terminate, our operations or to seek to merge with or to be acquired by another company.
Net Cash Flow from Operating Activities
Net cash used in operating activities was approximately $11,769,000$10,257,000 for the year ended December 31, 20092010 compared with $9,429,000$11,769,000 net cash used in operating activities for the year ended December 31, 2008.2009. The increasedecrease of approximately $2,340,000$1,512,000 resulted primarily from a net loss of $18,387,000,$11,993,000, less the add back of non-cash items of $6,333,000,$1,424,000, of which $4,202,000$4,368,000 related to stock-based compensation, $826,000$718,000 related to stock warrant expense in exchange for services, $218,000 issued as$785,000 related to a commitment fee in connection with the SEDA, $162,000reduction of potential redemption liability, $172,000 related to depreciation and $285,000$312,000 related to changes in current assets and liabilities and $858,000$3,049,000 that reflects the fair value of warrants and mandatorily redeemable stock obligations issued with the registered direct financings completed by the Company in 2009 Offering.and 2010.
41
Net Cash Flow from Investing Activities
Net cash used in investing activities was approximately $106,000 for the year ended December 31, 2010, compared with net cash used in investing activities of $83,000 for the year ended December 31, 2009, compared with net cash provided by investing activities of $9,600,000 for the year ended December 31, 2008.2009. The decreaseincrease of approximately $9,683,000$23,000 in cash provided byused in investing activities was primarily due to net redemptionpurchases of short-term investmentsequipment and furnishings in 2008.2010.
Net Cash Flow from Financing Activities
Net cash provided by financing activities was $11,570,000 for the year ended December 31, 2010, compared with $7,680,000 for the year ended December 31, 2009, compared with $7,922,000 for the year ended December 31, 2008.2009. This decreaseincrease was primarily due to net proceeds from the issuance of common stock in the amount of $7,918,000$15,235,000 to institutional investors in 2010 offset by $3,849,000 used for the second quarterpurchase of 2008treasury shares compared with net proceeds from the issuance of common stock in the amount of $7,714,000 to institutional investors in the third quarter of 2009.
Recently Issued Accounting Standards
Effective January 1, 2010, the Company adopted Accounting Standards Update (ASU)No. 2010-06,Fair Value Measurements and Disclosures (Topic 820): Improving Disclosures about Fair Value Measurements, or ASU2010-06. A reporting entity should provide additional disclosures about the different classes of assets and liabilities measured at fair value, the valuation techniques and inputs used, the activity in Level 3 fair value measurements, and the transfers between Levels 1, 2, and 3 fair value measurements. The adoption of the additional disclosures for Level 1 and Level 2 fair value measurements did not have anyan impact on our financial position, results of operations or cash flows.
4247
In April 2009,February 2010, the FASB issued the following: (i) FASB ASC TopicUpdate820-10-65,No. 2010-09, “Determining Fair Value When the Volume and Level of Activity for the Asset or Liability have Significantly Decreased and Identifying Transactions That Are Not Orderly”(“ASC820-10-65”), (ii) FASB ASC Topic320-10-65, “Subsequent Events (Topic 855) Amendments to Certain Recognition and Presentation ofDisclosure Requirements (UpdateOther-Than-TemporaryNo. 2010-09). Impairment”(“ASC320-10-65”), This update requires SEC registrants to evaluate subsequent events through the date that the financial statements are issued and (iii) FASB ASC Topic825-10-65, “Interim Disclosures about Fair Value of Financial Instruments”(“ASC825-10-65”),removes the requirement to disclose the date through which management evaluated subsequent events. This guidance was effective for interim and annual periods ending after June 15, 2009. ASC820-10-65 provides guidance on how to determine the fair value of assets and liabilities under ASC820-10 in the current economic environment and reemphasizes that the objective of a fair value measurement remains an exit price. If we were to conclude that there has been a significant decrease in the volume and level of activity of the asset or liability in relation to normal market activities, quoted market values may not be representative of fair value and we may conclude that a change in valuation technique or the use of multiple valuation techniques may be appropriate. ASC820-10-65 modifies the requirements for recognizingother-than-temporarily impaired debt securities and revise the existing impairment model for such securities, by modifying the current intent and ability indicator in determining whether a debt security isother-than-temporarily impaired. ASC825-10-65 enhances the disclosure of instruments under the scope of ASC 820 for both interim and annual periods. We are currently evaluating these staff positions and the impact, if any, that adoption will have on our financial position and results of operation.
In December 2007,2010, the FASB ratifiedissued ASC Update2010-29,Business Combinations (Topic 805) - Disclosure of Supplementary Pro Forma Information for Business Combinations (UpdateNo. 2010-29). This Update requires a public entity to disclose pro forma information for business combinations that occurred in the consensus reached bycurrent reporting period. The disclosures include pro forma revenue and earnings of the EITF on ASC Topic 808, “Accountingcombined entity for Collaborative Agreements” (“ASC 808-10-2”), which provides the definitioncurrent reporting period as though the acquisition date for all business combinations that occurred during the year had been as of a collaborative agreementthe beginning of the annual reporting period. If comparative financial statements are presented, the pro forma revenue and ASC 808-10-15 provides guidelines to assist anearnings of the combined entity in determining whether or not it is a party in a collaborative agreement. ASC 808-10-45 states that costs incurred and revenues generated from transactions with third parties shallfor the comparable prior reporting period should be reported in accordance with ASC Topic 605-45, “Reporting Revenue Gross as a Principal versus Netthough the acquisition date for all business combinations that occurred during the current year had been as of the beginning of the comparable prior annual reporting period. This Update affects any public entity that enters into business combinations that are material on an Agent,” (“ASC 808-10-50”) also provides minimum disclosure requirementsindividual or aggregate basis and is effective prospectively for an entity’s collaboration agreements and transition guidance. The adoptionbusiness combinations for which the acquisition date is on or after the beginning of ASC Topic 808 did not have a material impactthe first annual reporting period beginning on our financial position, results of operations and cash flows.
Off-Balance Sheet Arrangements
In connection with certain license agreement,agreements, we are required to indemnify the licensor for certain damages arising in connection with the intellectual property rights licensed under the agreement. In addition, we are a party to a number of agreements entered into in the ordinary course of business that contain typical provisions that obligate us to indemnify the other parties to such agreements upon the occurrence of certain events. These indemnification obligations are considered off-balance sheet arrangements in accordance with ASC Topic 460 (“ASC 460”),“Guarantor’s Accounting and Disclosure Requirements for Guarantees, Including Indirect Guarantees of Indebtedness of Others.”Others”. To date, we have not encountered material costs as
43
a result of such obligations and have not accrued any liabilities related to such obligations and have not accrued any liabilities related to such obligations in our financial statements. See Note 9 to our financial statements included in this annual report onForm 10-K for further discussion of these indemnification agreements.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURE ABOUT MARKET RISK |
Because we are a smaller reporting company, we are not required to provide the information required by this Item.
4448
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
RXi’s financial information as of December 31, 20082010 and 2009 and for the years then ended and for the cumulative financial information for the period from January 1, 2003 (date of inception) to December 31, 20092010 have been audited by our independent registered public accounting firm, BDO Seidman,USA, LLP.
| Page No. | ||||
Index to Financial Statements | ||||
4549
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of Directors and Stockholders
RXi Pharmaceuticals Corporation
Worcester, Massachusetts
We have audited the accompanying balance sheets of RXi Pharmaceuticals Corporation (a development stage Company) as of December 31, 20092010 and 20082009 and the related statements of expenses, stockholders’ equity, and cash flows for the years then ended and for the period from January 1, 2003 (date of inception) to December 31, 2009.2010. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of RXi Pharmaceuticals Corporation as of December 31, 20092010 and 20082009 and the results of its operations and its cash flows for the years then ended and for the period from January 1, 2003 (date of inception) to December 31, 2009,2010, in conformity with accounting principles generally accepted in the United States of America.
/s/ BDO Seidman,USA, LLP
Boston, Massachusetts
4650
| December 31, | December 31, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
| (Amounts in thousands, except share data) | (Amounts in thousands, except share data) | |||||||||||||||
ASSETS | ASSETS | ASSETS | ||||||||||||||
| Current assets: | ||||||||||||||||
| Cash and cash equivalents | $ | 5,684 | $ | 9,856 | $ | 6,891 | $ | 5,684 | ||||||||
| Prepaid expenses | 120 | 73 | 150 | 120 | ||||||||||||
| Total current assets | 5,804 | 9,929 | 7,041 | 5,804 | ||||||||||||
| Equipment and furnishings, net of accumulated depreciation and amortization of $320 and $158 in 2009 and 2008, respectively | 432 | 414 | ||||||||||||||
| Equipment and furnishings, net of accumulated depreciation and amortization of $491 and $320 in 2010 and 2009, respectively | 419 | 432 | ||||||||||||||
| Deposits | 16 | 16 | 16 | 16 | ||||||||||||
| Total assets | $ | 6,252 | $ | 10,359 | $ | 7,476 | $ | 6,252 | ||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||||||||||
| Current liabilities: | ||||||||||||||||
| Accounts payable | $ | 625 | $ | 394 | $ | 724 | $ | 625 | ||||||||
| Accrued expense and other current liabilities | 1,077 | 976 | 1,113 | 1,077 | ||||||||||||
| Current maturities of capital lease obligations | 52 | 17 | 51 | 52 | ||||||||||||
| Warrants potentially settleable in cash | 3,721 | — | 3,138 | 3,721 | ||||||||||||
| Total current liabilities | 5,475 | 1,387 | 5,026 | 5,475 | ||||||||||||
| Capital lease obligations, net of current maturities | 36 | 4 | 20 | 36 | ||||||||||||
| Total liabilities | 5,511 | 1,391 | 5,046 | 5,511 | ||||||||||||
| Commitments and contingencies (Notes 6, 8 & 14) | ||||||||||||||||
| Commitments and contingencies (Notes 6, 8, 14 & 15) | ||||||||||||||||
| Stockholders’ equity: | ||||||||||||||||
| Preferred stock, $0.0001 par value; 5,000,000 shares authorized; no shares issued and outstanding | — | — | — | — | ||||||||||||
| Common stock, $0.0001 par value; 50,000,000 shares authorized; 16,207,625 and 13,763,231 shares issued and outstanding in 2009 and 2008, respectively | 2 | 1 | ||||||||||||||
| Common stock, $0.0001 par value; 50,000,000 shares authorized; 19,047,759 shares issued and 18,372,759 shares outstanding and 16,207,625 shares issued and outstanding at December 31, 2010 and 2009, respectively | 2 | 2 | ||||||||||||||
| Additional paid-in capital | 44,489 | 34,330 | 62,020 | 44,489 | ||||||||||||
| Deficit accumulated during the developmental stage | (43,750 | ) | (25,363 | ) | (55,743 | ) | (43,750 | ) | ||||||||
| Less treasury shares at cost, 675,000 and 0 shares at December 31, 2010 and December 31, 2009, respectively | (3,849 | ) | — | |||||||||||||
| Total stockholders’ equity | 741 | 8,968 | 2,430 | 741 | ||||||||||||
| Total liabilities and stockholders’ equity | $ | 6,252 | $ | 10,359 | $ | 7,476 | $ | 6,252 | ||||||||
See accompanying notes to financial statements.
4751
RXi PHARMACEUTICALS CORPORATION
(A Development Stage Company)
(A Development Stage Company)
| Period from | Period from | |||||||||||||||||||||||
| January 1, | January 1, | |||||||||||||||||||||||
| 2003 (Date of | 2003 (Date of | |||||||||||||||||||||||
| Year Ended | Year Ended | Inception) to | Year Ended | Year Ended | Inception) to | |||||||||||||||||||
| December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | |||||||||||||||||||
| 2009 | 2008 | 2009 | 2010 | 2009 | 2010 | |||||||||||||||||||
| (Amounts in thousands, except share and per share data) | (Amounts in thousands, except share and per share data) | |||||||||||||||||||||||
| Expenses: | ||||||||||||||||||||||||
| Research and development expense | $ | 6,728 | $ | 5,105 | $ | 20,637 | $ | 6,046 | $ | 6,728 | $ | 26,683 | ||||||||||||
| Research and development employee stock-based compensation expense | 867 | 336 | 1,323 | 1,084 | 867 | 2,407 | ||||||||||||||||||
| Research and development non-employee stock-based compensation expense | 1,297 | 1,613 | 5,320 | 743 | 1,297 | 6,063 | ||||||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | — | — | 3,954 | — | — | 3,954 | ||||||||||||||||||
| Total research and development expense | 8,892 | 7,054 | 31,234 | 7,873 | 8,892 | 39,107 | ||||||||||||||||||
| General and administrative | 5,483 | 4,874 | 15,617 | |||||||||||||||||||||
| Fair value of common stock warrants issued for general and administrative expenses | 826 | 750 | 1,576 | |||||||||||||||||||||
| General and administrative expense | 5,493 | 5,483 | 21,110 | |||||||||||||||||||||
| Fair value of common stock warrants issued for general and administrative expense | 718 | 826 | 2,294 | |||||||||||||||||||||
| Fair value of common stock issued in exchange for general and administrative expenses | 281 | — | 281 | — | 281 | 281 | ||||||||||||||||||
| General and administrative employee stock-based compensation | 2,038 | 1,875 | 4,844 | |||||||||||||||||||||
| General and administrative employee stock-based compensation expense | 2,541 | 2,038 | 7,385 | |||||||||||||||||||||
| Total general and administrative expense | 8,628 | 7,499 | 22,318 | 8,752 | 8,628 | 31,070 | ||||||||||||||||||
| Total operating expenses | (17,520 | ) | (14,553 | ) | (53,552 | ) | (16,625 | ) | (17,520 | ) | (70,177 | ) | ||||||||||||
| Interest income (expense) | (5 | ) | 180 | 623 | 5 | (5 | ) | 628 | ||||||||||||||||
| Other income (expense) | (862 | ) | — | (862 | ) | 4,627 | (862 | ) | 3,765 | |||||||||||||||
| Loss before provision for income taxes | (18,387 | ) | (14,373 | ) | (53,791 | ) | (11,993 | ) | (18,387 | ) | (65,784 | ) | ||||||||||||
| Provision for income taxes | — | — | — | — | — | — | ||||||||||||||||||
| Net loss | $ | (18,387 | ) | $ | (14,373 | ) | $ | (53,791 | ) | $ | (11,993 | ) | $ | (18,387 | ) | $ | (65,784 | ) | ||||||
| Net loss per common share: | ||||||||||||||||||||||||
| Basic and diluted loss per share | $ | (1.24 | ) | $ | (1.09 | ) | $ | (0.67 | ) | $ | (1.24 | ) | ||||||||||||
| Weighted average common shares outstanding: basic and diluted | 14,796,541 | 13,239,942 | 17,883,381 | 14,796,541 | ||||||||||||||||||||
See accompanying notes to financial statements.
4852
RXi PHARMACEUTICALS CORPORATION
(A Development Stage Company)
STATEMENTS OF STOCKHOLDERS’ EQUITY FOR THE PERIOD FROM APRIL 3, 2006 TO
DECEMBER 31,20092010 AND PARENT COMPANY’S NET DEFICIT FOR THE PERIOD
FROM DECEMBER 31, 2003 TO DECEMBER 31, 2006
(A Development Stage Company)
STATEMENTS OF STOCKHOLDERS’ EQUITY FOR THE PERIOD FROM APRIL 3, 2006 TO
DECEMBER 31,
FROM DECEMBER 31, 2003 TO DECEMBER 31, 2006
| Deficit | Deficit | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Accumulated | Accumulated | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Additional | During | Parent | Additional | During | Parent | |||||||||||||||||||||||||||||||||||||||||||||||
| Common Stock | Paid-In | Development | Company’s | Common Stock | Paid-In | Development | Treasury | Company’s | ||||||||||||||||||||||||||||||||||||||||||||
| Shares Issued | Amount | Capital | Stage | Net Deficit | Total | Shares Issued | Amount | Capital | Stage | Stock | Net Deficit | Total | ||||||||||||||||||||||||||||||||||||||||
| (Amounts in thousands, except share data) | (Amounts in thousands, except share and per share data) | |||||||||||||||||||||||||||||||||||||||||||||||||||
Predecessor | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2003 | — | $ | — | $ | — | $ | (89 | ) | $ | (89 | ) | — | $ | — | $ | — | $ | (89 | ) | $ | (89 | ) | ||||||||||||||||||||||||||||||
| Net loss | — | — | — | (3,272 | ) | (3,272 | ) | — | — | — | (3,272 | ) | (3,272 | ) | ||||||||||||||||||||||||||||||||||||||
| Net transactions with Parent | — | — | — | 2,393 | 2,393 | — | — | — | 2,393 | 2,393 | ||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2004 | — | — | — | (968 | ) | (968 | ) | — | — | — | (968 | ) | (968 | ) | ||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | (2,209 | ) | (2,209 | ) | — | — | — | (2,209 | ) | (2,209 | ) | ||||||||||||||||||||||||||||||||||||||
| Net transactions with Parent | — | — | — | 2,727 | 2,727 | — | — | — | 2,727 | 2,727 | ||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2005 | — | — | — | (450 | ) | (450 | ) | — | — | — | (450 | ) | (450 | ) | ||||||||||||||||||||||||||||||||||||||
| Net Loss | — | — | — | (2,405 | ) | (2,405 | ) | |||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | (2,405 | ) | (2,405 | ) | |||||||||||||||||||||||||||||||||||||||||||||
| Net transactions with Parent | — | — | — | 2,587 | 2,587 | — | — | — | 2,587 | 2,587 | ||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2006 | — | $ | — | $ | — | $ | (268 | ) | $ | (268 | ) | — | $ | — | $ | — | $ | (268 | ) | $ | (268 | ) | ||||||||||||||||||||||||||||||
Successor | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at April 3, 2006 | — | $ | — | $ | — | $ | — | $ | — | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||||||||||||||||||||
| Issuance of common stock | 1,624,278 | — | 2 | — | 2 | 1,624,278 | — | 2 | — | — | 2 | |||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2006 | 1,624,278 | — | 2 | — | 2 | 1,624,278 | — | 2 | — | — | 2 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued to CytRx for contribution of RXi and other assets | 7,040,318 | 1 | 47 | — | 48 | 7,040,318 | 1 | 47 | — | — | 48 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued for cash | 3,273,292 | — | 15,348 | — | 15,348 | 3,273,292 | — | 15,348 | — | — | 15,348 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued to CytRx for reimbursement of expenses | 188,387 | — | 978 | — | 978 | 188,387 | — | 978 | — | — | 978 | |||||||||||||||||||||||||||||||||||||||||
| Expenses incurred by CytRx for RXi | — | — | 831 | — | 831 | — | — | 831 | — | — | 831 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued to UMMS for additional intellectual properties | 462,112 | — | 2,311 | — | 2,311 | 462,112 | — | 2,311 | — | — | 2,311 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued to directors | 30,000 | — | 150 | — | 150 | 30,000 | — | 150 | — | — | 150 | |||||||||||||||||||||||||||||||||||||||||
| Common stock issued upon exercise of stock options | 66,045 | — | 331 | — | 331 | 66,045 | — | 331 | 331 | |||||||||||||||||||||||||||||||||||||||||||
| Stock based compensation for directors and employees | — | — | 1,048 | — | 1,048 | — | — | 1,048 | — | — | 1,048 | |||||||||||||||||||||||||||||||||||||||||
| Stock based compensation expense for services | — | — | 766 | — | 766 | — | — | 766 | — | — | 766 | |||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | (10,990 | ) | (10,990 | ) | — | — | — | (10,990 | ) | — | (10,990 | ) | |||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2007 | 12,684,432 | 1 | 21,812 | (10,990 | ) | 10,823 | 12,684,432 | 1 | 21,812 | (10,990 | ) | — | 10,823 | |||||||||||||||||||||||||||||||||||||||
4953
| Deficit | ||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||
| Additional | During | Parent | ||||||||||||||||||||||
| Common Stock | Paid-In | Development | Company’s | |||||||||||||||||||||
| Shares Issued | Amount | Capital | Stage | Net Deficit | Total | |||||||||||||||||||
| (Amounts in thousands, except share data) | ||||||||||||||||||||||||
| Issuance of common stock, net of offering costs of $796 | 1,073,299 | — | 7,918 | — | 7,918 | |||||||||||||||||||
| Common stock issued upon exercise of stock options | 5,500 | — | 26 | — | 26 | |||||||||||||||||||
| Stock based compensation for directors and employees | — | — | 2,211 | — | 2,211 | |||||||||||||||||||
| Stock based compensation expense for services | — | — | 1,613 | — | 1,613 | |||||||||||||||||||
| Common stock warrant expense in exchange for services | 750 | 750 | ||||||||||||||||||||||
| Net loss | — | — | — | (14,373 | ) | (14,373 | ) | |||||||||||||||||
| Balance at December 31, 2008 | 13,763,231 | 1 | 34,330 | (25,363 | ) | 8,968 | ||||||||||||||||||
| Issuance of common stock, net of offering costs of $636 | 2,385,715 | 1 | 7,713 | — | 7,714 | |||||||||||||||||||
| Common stock warrants issued in connection with the 2009 Offering | — | — | (2,863 | ) | — | (2,863 | ) | |||||||||||||||||
| Common stock issued upon exercise of stock options | 281 | — | — | — | — | |||||||||||||||||||
| Common stock issued as commitment fee in connection with SEDA | 58,398 | — | 281 | — | 281 | |||||||||||||||||||
| Stock based compensation for directors and employees | — | — | 2,906 | — | 2,906 | |||||||||||||||||||
| Stock based compensation expense for services | — | — | 1,296 | — | 1,296 | |||||||||||||||||||
| Common stock warrant expense in exchange for services | — | 826 | — | 826 | ||||||||||||||||||||
| Net loss | — | — | — | (18,387 | ) | (18,387 | ) | |||||||||||||||||
| Balance at December 31, 2009 | 16,207,625 | 2 | $ | 44,489 | $ | (43,750 | ) | $ | 741 | |||||||||||||||
| Deficit | ||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||
| Additional | During | Parent | ||||||||||||||||||||||||||
| Common Stock | Paid-In | Development | Treasury | Company’s | ||||||||||||||||||||||||
| Shares Issued | Amount | Capital | Stage | Stock | Net Deficit | Total | ||||||||||||||||||||||
| (Amounts in thousands, except share and per share data) | ||||||||||||||||||||||||||||
| Issuance of common stock, net of offering costs of $796 | 1,073,299 | — | 7,918 | — | — | 7,918 | ||||||||||||||||||||||
| Common stock issued upon exercise of stock options | 5,500 | — | 26 | — | — | 26 | ||||||||||||||||||||||
| Stock based compensation for directors and employees | — | — | 2,211 | — | — | 2,211 | ||||||||||||||||||||||
| Stock based compensation expense for services | — | — | 1,613 | — | — | 1,613 | ||||||||||||||||||||||
| Common stock warrant expense in exchange for services | 750 | — | 750 | |||||||||||||||||||||||||
| Net loss | — | — | — | (14,373 | ) | (14,373 | ) | |||||||||||||||||||||
| Balance at December 31, 2008 | 13,763,231 | 1 | 34,330 | (25,363 | ) | — | 8,968 | |||||||||||||||||||||
| Issuance of common stock, net of offering costs of $636 | 2,385,715 | 1 | 7,713 | — | — | 7,714 | ||||||||||||||||||||||
| Common stock warrants issued in connection with the 2009 Offering | — | — | (2,863 | ) | — | — | (2,863 | ) | ||||||||||||||||||||
| Common stock issued upon exercise of stock options | 281 | — | — | — | — | — | ||||||||||||||||||||||
| Common stock issued as commitment fee in connection with SEDA | 58,398 | — | 281 | — | — | 281 | ||||||||||||||||||||||
| Stock based compensation expense for directors and employees | — | — | 2,906 | — | — | 2,906 | ||||||||||||||||||||||
| Stock based compensation expense for services | — | — | 1,296 | — | — | 1,296 | ||||||||||||||||||||||
| Common stock warrant expense in exchange for services | — | — | 826 | — | 826 | |||||||||||||||||||||||
| Net loss | — | — | — | (18,387 | ) | (18,387 | ) | |||||||||||||||||||||
| Balance at December 31, 2009 | 16,207,625 | 2 | 44,489 | (43,750 | ) | — | 741 | |||||||||||||||||||||
| Issuance of common stock, net of offering costs of $965 | 2,700,000 | — | 15,235 | — | — | 15,235 | ||||||||||||||||||||||
| Purchase of 675,000 shares of treasury stock | — | — | — | — | (3,849 | ) | (3,849 | ) | ||||||||||||||||||||
| Common stock warrants issued in connection with 2010 offering | — | — | (2,466 | ) | — | — | (2,466 | ) | ||||||||||||||||||||
| Fair value of shares mandatorily redeemable for cash upon exercise of warrants | — | — | (785 | ) | — | — | (785 | ) | ||||||||||||||||||||
| Common stock issued upon exercise of stock options | 53,500 | — | 254 | — | — | 254 | ||||||||||||||||||||||
54
| Deficit | ||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||
| Additional | During | Parent | ||||||||||||||||||||||||||
| Common Stock | Paid-In | Development | Treasury | Company’s | ||||||||||||||||||||||||
| Shares Issued | Amount | Capital | Stage | Stock | Net Deficit | Total | ||||||||||||||||||||||
| (Amounts in thousands, except share and per share data) | ||||||||||||||||||||||||||||
| Issuance of restricted stock units | 86,634 | — | 207 | — | — | 207 | ||||||||||||||||||||||
| Stock based compensation expense for directors and employees | — | — | 3,625 | — | — | 3,625 | ||||||||||||||||||||||
| Stock based compensation expense for services | — | — | 743 | — | — | 743 | ||||||||||||||||||||||
| Value of common stock warrants issued in exchange for services | — | — | 718 | — | 718 | |||||||||||||||||||||||
| Net loss | — | — | — | (11,993 | ) | — | (11,993 | ) | ||||||||||||||||||||
| Balance at December 31, 2010 | 19,047,759 | $ | 2 | $ | 62,020 | $ | (55,743 | ) | $ | (3,849 | ) | $ | 2,430 | |||||||||||||||
See accompanying notes to financial statements.
5055
| Period from | Period from | |||||||||||||||||||||||
| January 1, | January 1, | |||||||||||||||||||||||
| 2003 (Date of | 2003 (Date of | |||||||||||||||||||||||
| Inception) | Inception) | |||||||||||||||||||||||
| Year Ended | Year Ended | through | Year Ended | Year Ended | through | |||||||||||||||||||
| December 31, | December 31, | December 31, | December 31, | December 31, | December 31, | |||||||||||||||||||
| 2009 | 2008 | 2009 | 2010 | 2009 | 2010 | |||||||||||||||||||
| (Amounts in thousands) | (Amounts in thousands) | |||||||||||||||||||||||
Cash flows from operating activities: | ||||||||||||||||||||||||
| Net loss | $ | (18,387 | ) | $ | (14,373 | ) | $ | (53,791 | ) | $ | (11,993 | ) | $ | (18,387 | ) | $ | (65,784 | ) | ||||||
| Adjustment to reconcile net loss to net cash used in operating activities: | ||||||||||||||||||||||||
| Depreciation and amortization expense | 162 | 131 | 329 | 172 | 162 | 501 | ||||||||||||||||||
| Loss on disposal of equipment | 4 | 8 | 12 | — | 4 | 12 | ||||||||||||||||||
| Non-cash rent expense | — | 29 | 29 | — | — | 29 | ||||||||||||||||||
| Accretion and receipt of bond discount | — | 207 | 35 | — | — | 35 | ||||||||||||||||||
| Non-cash share based compensation | 4,202 | 3,824 | 11,489 | 4,368 | 4,202 | 15,857 | ||||||||||||||||||
| Fair value of shares mandatorily redeemable for cash upon exercise of warrants | (785 | ) | — | (785 | ) | |||||||||||||||||||
| Fair value of common stock warrants issued in exchange for services | 826 | 750 | 1,576 | 718 | 826 | 2,294 | ||||||||||||||||||
| Fair value of common stock issued as commitment fee in connection with SEDA | 281 | — | 281 | |||||||||||||||||||||
| Change in fair value of common stock warrants issued in connection with the 2009 offering | 858 | — | 858 | |||||||||||||||||||||
| Fair value of common stock issued in exchange for services | — | 281 | 281 | |||||||||||||||||||||
| Change in fair value of common stock warrants issued | (3,049 | ) | 858 | (2,191 | ) | |||||||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | — | — | 3,954 | — | — | 3,954 | ||||||||||||||||||
| Changes in assets and liabilities: | ||||||||||||||||||||||||
| Changes in operating assets and liabilities: | ||||||||||||||||||||||||
| Prepaid expenses | (47 | ) | (51 | ) | (120 | ) | (30 | ) | (47 | ) | (150 | ) | ||||||||||||
| Accounts payable | 231 | 339 | 625 | 99 | 231 | 724 | ||||||||||||||||||
| Due to former parent | — | (207 | ) | (207 | ) | — | — | (207 | ) | |||||||||||||||
| Accrued expenses and other current liabilities | 101 | (86 | ) | 1,077 | 243 | 101 | 1,320 | |||||||||||||||||
Net cash used in operating activities | (11,769 | ) | (9,429 | ) | (33,853 | ) | (10,257 | ) | (11,769 | ) | (44,110 | ) | ||||||||||||
| Cash flows from investing activities: | ||||||||||||||||||||||||
| Purchase of short-term investments | — | (19,785 | ) | (31,542 | ) | (5,990 | ) | — | (37,532 | ) | ||||||||||||||
| Maturities of short-term investments | — | 29,530 | 31,507 | 5,990 | — | 37,497 | ||||||||||||||||||
| Cash paid for purchase of equipment and furnishings | (82 | ) | (166 | ) | (580 | ) | (106 | ) | (82 | ) | (686 | ) | ||||||||||||
| Disposal of equipment and furnishings | (1 | ) | — | (1 | ) | — | (1 | ) | (1 | ) | ||||||||||||||
| Cash refunded (paid) for lease deposit | — | 21 | (45 | ) | — | — | (45 | ) | ||||||||||||||||
Net cash provided by (used in) investing activities | (83 | ) | 9,600 | (661 | ) | |||||||||||||||||||
Net cash used in investing activities | (106 | ) | (83 | ) | (767 | ) | ||||||||||||||||||
| Cash flows from financing activities: | ||||||||||||||||||||||||
| Net proceeds from issuance of common stock | 7,714 | 7,918 | 31,132 | |||||||||||||||||||||
| Net proceeds from 2010 registered direct offering | 15,235 | 7,714 | 46,367 | |||||||||||||||||||||
| Repurchase of treasury stock | (3,849 | ) | (3,849 | ) | ||||||||||||||||||||
| Net proceeds from exercise of common stock options | — | 26 | 356 | 254 | — | 610 | ||||||||||||||||||
| Repayments of capital lease obligations | (34 | ) | (22 | ) | (56 | ) | (70 | ) | (34 | ) | (126 | ) | ||||||||||||
| Cash advances from Parent, net | 8,766 | — | 8,766 | |||||||||||||||||||||
Net cash provided by financing activities | 7,680 | 7,922 | 40,198 | 11,570 | 7,680 | 51,768 | ||||||||||||||||||
| Net increase in cash and cash equivalents | (4,172 | ) | 8,093 | 5,684 | 1,207 | (4,172 | ) | 6,891 | ||||||||||||||||
| Cash and cash equivalents at the beginning of period | 9,856 | 1,763 | — | 5,684 | 9,856 | — | ||||||||||||||||||
| Cash and cash equivalents at end of period | $ | 5,684 | $ | 9,856 | $ | 5,684 | $ | 6,891 | $ | 5,684 | $ | 6,891 | ||||||||||||
Supplemental disclosure of cash flow information: | ||||||||||||||||||||||||
| Cash received during the periods for interest | 1 | $ | 449 | $ | 724 | $ | — | $ | 1 | $ | 724 | |||||||||||||
| Cash paid during the periods for interest | $ | — | $ | 7 | $ | 7 | $ | — | $ | — | $ | 7 | ||||||||||||
5156
| Period from | ||||||||||||
| January 1, | ||||||||||||
| 2003 (Date of | ||||||||||||
| Inception) | ||||||||||||
| Year Ended | Year Ended | through | ||||||||||
| December 31, | December 31, | December 31, | ||||||||||
| 2009 | 2008 | 2009 | ||||||||||
| (Amounts in thousands) | ||||||||||||
Supplemental disclosure of non-cash investing and financing activities: | ||||||||||||
| Settlement of corporate formation expenses in exchange for common stock | $ | — | $ | — | $ | 978 | ||||||
| Fair value of warrants issued in connection with common stock recorded as cost of equity | $ | 2,863 | $ | — | $ | 2,863 | ||||||
| Allocation of management expenses | $ | — | $ | — | $ | 551 | ||||||
| Equipment and furnishings exchanged for common stock | $ | — | $ | — | $ | 48 | ||||||
| Acquisition of equipment and furnishings through accrued liabilities | $ | — | $ | — | $ | — | ||||||
| Equipment and furnishings acquired through capital lease | $ | 101 | $ | 43 | $ | 144 | ||||||
| Non-cash lease deposit | $ | — | $ | — | $ | 50 | ||||||
| Period from | ||||||||||||
| January 1, | ||||||||||||
| 2003 (Date of | ||||||||||||
| Inception) | ||||||||||||
| Year Ended | Year Ended | through | ||||||||||
| December 31, | December 31, | December 31, | ||||||||||
| 2010 | 2009 | 2010 | ||||||||||
| (Amounts in thousands) | ||||||||||||
Supplemental disclosure of non-cash investing and financing activities: | ||||||||||||
| Settlement of corporate formation expenses in exchange for common stock | $ | — | $ | — | $ | 978 | ||||||
| Fair value of warrants issued in connection with common stock recorded as cost of equity | $ | 2,466 | $ | 2,863 | $ | 5,329 | ||||||
| Fair value of shares mandatorily redeemable for cash upon exercise of warrants | $ | 785 | $ | — | $ | 785 | ||||||
| Allocation of management expenses | $ | — | $ | — | $ | 551 | ||||||
| Equipment and furnishings exchanged for common stock | $ | — | $ | — | $ | 48 | ||||||
| Equipment and furnishings acquired through capital lease | $ | 53 | $ | 101 | $ | 197 | ||||||
| Non-cash lease deposit | $ | — | $ | — | $ | 50 | ||||||
| Value of restricted stock units issued in lieu of cash bonuses | $ | 207 | $ | — | $ | 207 | ||||||
See accompanying notes to financial statements.
5257
| 1. | Nature of Business |
RXi Pharmaceuticals Corporation (“RXi” or the “Company”) was formed by CytRx Corporation (“CytRx” or the “Former Parent”) and four prominent RNAi researchers, including Craig C. Mello, Ph.D., who was awarded the 2006 Nobel Prize in Medicine for his co-discovery of RNAi. The purpose of forming RXi was to pursue the development of proprietary therapeutics based on RNAi for the treatment of human diseases. By utilizing the Company’s expertise in RNAi and the comprehensive RNAi technology platform it has established, the Company believes it will be able to discover and develop lead compounds and progress them into and through clinical development for potential commercialization more efficiently than traditional drug development approaches, primarily in partnerships with pharmaceutical and biotechnology companies.
RXi was incorporated as Argonaut Pharmaceuticals, Inc., in Delaware, on April 3, 2006. The Company changed its name to RXi Pharmaceuticals Corporation on November 28, 2006. From April 3, 2006 (date of incorporation) until January 8, 2007, no business was conducted at the RXi level. On January 8, 2007, RXi entered into a contribution agreement with CytRx under which CytRx assigned and contributed to RXi substantially all of its RNAi-related technologies and assets and commenced operations; these contributed assets were recorded by RXi at the historical cost basis of $48,000.
Because the RNAi activities prior to 2007 were conducted by CytRx, the financial statements of RXi for the periods through December 31, 2006 have been disaggregated, or “carved-out,” of the financial statements of CytRx. These carved-out financial statements form what are referred to herein as the financial statements of the “Predecessor,” and include both direct and indirect expenses. The historical direct expenses consist primarily of the various costs for technology license agreements, sponsored research agreements and fees paid to scientific advisors. Indirect expenses during this period represent expenses incurred by CytRx on behalf of RXi, including salary, benefits, rent, accounting and other general and administrative expenses that have been allocated to RXi based upon estimates of the percentage of time spent by individual CytRx employees working on RXi matters. Management believes the assumptions underlying the allocations of indirect expenses in the carve-out financial information are reasonable; however, RXi’s financial position, results of operations and cash flows may have been materially different if it was operated as a stand-alone entity as of and for the periods ended December 31, 2007. RXi’s financial information from January 8, 2007 is referred to in these financial statements as the financial information of the “Successor” and includes expenses incurred by RXi in its RNAi therapeutic programs, as well as an allocation of indirect expenses relating to corporate services provided by CytRx through December 31, 2007. In addition, the net intercompany activities between Predecessor and CytRx have been accumulated into a single caption entitled “Parent Company’s Net Deficit.”
To date, RXi’s principal activities have consisted of conducting discovery research and pre-clinical development activities utilizing the Company’s RNAi therapeutic platform, acquiring RNAi technologies and patent rights through exclusive, co-exclusive and non-exclusive licenses, recruiting an RNAi-focused management and scientific/clinical advisory team, capital raising activities and conducting business development activities aimed at establishing research and development partnerships with pharmaceutical and larger biotechnology companies.
As the Company has not generated any revenues from inception through December 31, 2009,2010, the Company is considered a development-stage company for accounting purposes.
The Company had cash and cash equivalents of approximately $5.7$6.9 million as of December 31, 20092010.
During 2010 and approximately $14.4 million asto date in 2011, the Company entered into the following significant financing transactions:
On March 26, 2010, the Company closed a registered direct financing pursuant to which the Company sold to certain investors 2,700,000 shares of March 31, 2010.common stock at $6.00 per share and warrants to purchase
5358
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
On March 4, 2011, the Company closed an underwritten public offering of 6,000,000 units at a price to the public of $1.35 per unit for gross proceeds of $8.1 million. The financing provided approximately $7.3 million to the Company estimatesafter deducting the underwriting fee and offering expenses. Each unit consists of (i) one share of common stock, (ii) a thirteen-month warrant to purchase 0.50 of a share of common stock at an exercise of $1.70 per share (subject to anti-dilution adjustment) and (iii) a five-year warrant to purchase 0.50 of a share of common stock at an exercise price of $1.87 per share (subject to anti-dilution adjustment).
On April 15, 2011, the Company announced that it will repurchasehad priced an additional 135,000underwritten public offering of 11,950,000 units at a price to the public of $1.00 per unit for gross proceeds of approximately $12 million. Each unit consists of one share of common stock and a warrant to purchase one share of common stock at an exercise price of $1.00 per share. The shares of its common stock and warrants are immediately separable and will be issued separately such that no units will be issued. The warrants are exercisable beginning one year and one day from CytRx forthe date of issuance, but only if RXi’s stockholders approve an aggregate priceincrease in the number of $0.8 million if allits authorized shares of common stock, and expire on the sixth anniversary of the date of issuance. Net proceeds, after underwriting discounts and commissions and other estimated fees and expenses payable by RXi, and assuming the warrants issued inare not exercised, will be approximately $10.9 million. RXi intends to use the net proceeds of the offering for general corporate purposes, which may include working capital, capital expenditures, research and development expenditures, clinical and pre-clinical trial expenditures, commercial expenditures, acquisitions of new technologies or businesses that are exercised.complementary to its current technologies or business focus, and investments. The offering is expected to close on or about April 20, 2011, subject to satisfaction of customary closing conditions.
5459
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
when needed, we would be forced to scale back, or terminate, the Company’s operations or to seek to merge with or to be acquired by another company.
The Company expects to incur significant operating losses for the foreseeable future while it advances its future product candidates from discovery through pre-clinical studies and clinical trials and seek regulatory approval and potential commercialization, even if the Company is collaborating with pharmaceutical and larger biotechnology companies. In addition to these increasing research and development expenses, the Company expects general and administrative costs to increase as it recruits additional management and administrative personnel. The Company will need to generate significant revenues to achieve profitability and may never do so.
| 2. | Summary of Significant Accounting Policies |
Uses of estimates in preparation of financial statements — The preparation of financial statements in accordance with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ materially from thesethose estimates.
Cash and Cash Equivalents — The Company considers all highly-liquid debt instruments with an original maturity of 90 days or less to be cash equivalents. Cash equivalents consist primarily of amounts invested in money market accounts.
Fair Value of Financial Instruments — The carrying amounts reported in the balance sheet for cash equivalents, accounts payable, and accrued liabilitiescapital leases approximate their fair values due to their short-term nature.nature and market rates of interest.
Equipment and Furnishings — Equipment and furnishings are stated at cost and depreciated using the straight-line method based on the estimated useful lives (generally three to five years for equipment and furniture) of the related assets.
Depreciation and amortization expense for the yearyears ended December 31, 20092010 and 20082009 was approximately $162,000$172,000 and $131,000,$162,000, respectively.
Impairment of Long-Lived Assets — The Company reviews long-lived assets, including finite lived intangible assets, for impairment on an annual basis, as of December 31, or on an interim basis if an event occurs that might reduce the fair value of such assets below their carrying values. An impairment loss would be recognized based on the difference between the carrying value of the asset and its estimated fair value, which would be determined based on either discounted future cash flows or other appropriate fair value methods. The Company believes no impairment existed as of December 31, 2009.2010.
Patents and Patent Application Costs — Although the Company believes that its patents and underlying technology have continuing value, the amount of future benefits to be derived from the patents is uncertain. Patent costs are, therefore, expensed as incurred.
Share-based Compensation — The Company follows the provisions of the Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 718, ““Compensation — Stock Compensation”Compensation” (“ASC 718”), which requires the measurement and recognition of compensation expense for all share-basedstock based payment awards made to employees, non-employee directors, and consultants, including employee stock options. Stock compensation expense based on the grant date fair value estimated in accordance with the provisions of ASC 718 is recognized as an expense over the requisite service period.
60
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
For stock options granted as consideration for services rendered by non-employees, the Company recognizes compensation expense in accordance with the requirements of FASB ASC Topic505-50(“505-50 (“ASC505-50”), ““Equity Based Payments to Non-Employees”Non- Employees.” Non-employee option grants that do not vest immediately upon grant are recorded as an expense over the vesting period of the underlying stock options. At
55
the end of each financial reporting period prior to vesting, the value of these options, as calculated using the Black-Scholes option-pricing model, will be re-measured using the fair value of the Company’s common stock and the non-cash compensation recognized during the period will be adjusted accordingly. Since the fair market value of options granted to non-employees is subject to change in the future, the amount of the future compensation expense will include fair value re-measurements until the stock options are fully vested.
The Company recognized $1.3 million$743,000 and $1.6 million$1,300,000 of stock based compensation expense related to non-employee stock options for the years ended December 31, 20092010 and 2008,2009, respectively.
Derivative Financial Instruments — During the normal course of business, from time to time, the Company issues warrants and options to vendors as consideration to perform services. It may also issue warrants as part of a debt or equity financing. The Company does not enter into any derivative contracts for speculative purposes.
The Company recognizes all derivatives as assets or liabilities measured at fair value with changes in fair value of derivatives reflected as current period income or loss unless the derivatives qualify for hedge accounting and are accounted for as such. During the year ended December 31, 2009, the Company issued warrants to purchase 978,142 shares of common stock in connection with an equity transaction. In accordance with FASB ASC Topic815-40, ““Derivatives and Hedging — Contracts in Entity’s Own Stock”Stock,(“ASC 815-40”),” the value of these warrants is required to be recorded as a liability, as the holders have an option to put the warrants back to the Company inupon the occurrence of certain events as defined.set forth in the warrant agreement.
Research and Development Expenses — Research and development costs are expensed as incurred. Included in research and development costs are wages, benefits and other operating costs, facilities, supplies, external services and overhead directly related to the Company’s research and development departments as well as costs to acquire technology licenses.
Income Taxes — The Company recognizes liabilities or assets for the deferred tax consequences of temporary differences between the tax basis of assets or liabilities and their reported amounts in the financial statements in accordance with FASBASC740-10, ““Accounting for Income Taxes” (“ASC740-10”).These temporary differences will result in taxable or deductible amounts in future years when the reported amounts of the assets or liabilities are recovered or settled.ASC740-10requires that a valuation allowance be established when management determines that it is more likely than not that all or a portion of a deferred asset will not be realized. RXi evaluates the realizability of its net deferred income tax assets and valuation allowances as necessary, at least on an annual basis. During this evaluation, the Company reviews its forecasts of income in conjunction with other positive and negative evidence surrounding the realizability of its deferred income tax assets to determine if a valuation allowance is required. Adjustments to the valuation allowance will increase or decrease the Company’s income tax provision or benefit. The recognition and measurement of benefits related to the Company’s tax positions requires significant judgment, as uncertainties often exist with respect to new laws, new interpretations of existing laws, and rulings by taxing authorities. Differences between actual results and RXi’s assumptions or changes in the Company’s assumptions in future periods are recorded in the period they become known
61
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
Concentrations of Credit Risk — Financial instruments that potentially subject the Company to significant concentrations of credit risk consist principally of cash and cash equivalents. The Company maintains cash balances in several accounts with one bank, which at times are in excess of federally insured limits. As of December 31, 2009,2010, the Company’s cash equivalents were invested in money market mutual funds. The Company’s investment policy disallows investment in any debt securities rated less than “investment grade” by national ratings services. The Company has not experienced any losses on its deposits of cash and cash equivalents or its short-term investments.
56
Comprehensive Loss — The Company’s comprehensive loss is equal to its net loss for all periods presented.
Parent Company’s Net Deficit — The Parent Company’s Net Deficit of the Predecessor consists of CytRx’s initial investment in RXi and subsequent changes in RXi’s net investment resulting from RXi being an integrated part of CytRx. All disbursements for the Predecessor were made by CytRx.
| 3. | Recent Accounting Pronouncements |
5762
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
In February 2010, the FASB issued ASC UpdateNo. 2010-09,Subsequent Events (Topic 855) Amendments to Certain Recognition and Disclosure Requirements (UpdateNo. 2010-09).(“ASC825-10-65”), This update requires SEC registrants to evaluate subsequent events through the date that the financial statements are issued and removes the requirement to disclose the date through which management evaluated subsequent events. This guidance was effective for interim and annual periods ending after June 15, 2009. ASC820-10-65 provides guidance on how to determine the fair value of assets and liabilities under ASC820-10 in the current economic environment and reemphasizes that the objective of a fair value measurement remains an exit price. If RXI were to conclude that there has been a significant decrease in the volume and level of activity of the asset or liability in relation to normal market activities, quoted market values may not be representative of fair value and the Company may conclude that a change in valuation technique or the use of multiple valuation techniques may be appropriate. ASC820-10-65 modifies the requirements for recognizingother-than-temporarily impaired debt securities and revises the existing impairment model for such securities, by modifying the current intent and ability indicator in determining whether a debt security isother-than-temporarily impaired.ASC 825-10-65 enhances the disclosure of instruments under the scope of ASC 820 for both interim and annual periods. The Company is currently evaluating these staff positions and the impact, if any, that adoption will have on its financial position and results of operation.
In December 2007,2010, the FASB ratifiedissued ASC Update2010-29,Business Combinations (Topic 805) - Disclosure of Supplementary Pro Forma Information for Business Combinations (UpdateNo. 2010-29). This Update requires a public entity to disclose pro forma information for business combinations that occurred in the consensus reached bycurrent reporting period. The disclosures include pro forma revenue and earnings of the EITF on ASC Topic 808,“Accountingcombined entity for Collaborative Agreements” (“ASC 808-10-2”), which provides the definitioncurrent reporting period as though the acquisition date for all business combinations that occurred during the year had been as of a collaborative agreementthe beginning of the annual reporting period. If comparative financial statements are presented, the pro forma revenue and ASC 808-10-15 provides guidelines to assist anearnings of the combined entity in determining whether or not it is a party in a collaborative agreement. ASC 808-10-45 states that costs incurred and revenues generated from transactions with third parties shallfor the comparable prior reporting period should be reported in accordance with ASC Topic 605-45,“Reporting Revenue Gross as a Principal versus Netthough the acquisition date for all business combinations that occurred during the current year had been as of the beginning of the comparable prior annual reporting period. This Update affects any public entity that enters into business combinations that are material on an Agent,” (“ASC 808-10-50”) also provides minimum disclosure requirementsindividual or aggregate basis and is effective prospectively for an entity’s collaboration agreements and transition guidance.business combinations for which the acquisition date is on or after the beginning of the first annual reporting period beginning on or after December 15, 2010. The adoption of ASC Topic 808Company did not have a materialelect to adopt this update early as permitted, thus it has no impact on the Company’scurrent financial position, results of operations and cash flows.
| 4. | Fair Value Measurements |
In January 2010, the FASB issued ASU2010-06, “Improving Disclosures about Fair Value Measurements”, (“ASU2010-06”). The standard amends FASB ASC Topic 820, “Fair Value Measurements and Disclosures”, (“ASC 820”) to require additional disclosures related to transfers in and out of Levels 1 and 2 and for activity in Level 3 and clarifies other existing disclosure requirements. The Company adopted ASU2010-06 beginning January 1, 2010. This update had no impact on the Company’s financial statements.
Effective January 1, 2008, the Company implemented FASB ASC Topic 820“Fair Value Measurements and Disclosures” (“ASC 820”) for the Company’s financial assets and liabilities that are re-measured and reported at fair value at each reporting period, and are re-measured and reported at fair value at least annually using a fair value hierarchy that is broken down into three levels. Level inputs are as defined as follows:
Level 1 — quoted prices in active markets for identical assets or liabilities.
Level 2 — other significant observable inputs for the assets or liabilities through corroboration with market data at the measurement date.
58
Level 3 — significant unobservable inputs that reflect management’s best estimate of what market participants would use to price the assets or liabilities at the measurement date.
The Company categorized its cash equivalents as a Level 1 hierarchy. The valuation for Level 1 was determined based on a “market approach” using quoted prices in active markets for identical assets. Valuations of these assets do not require a significant degree of judgment. The Company categorized its warrants potentially settledsettleable in cash as a Level 2 hierarchy. The warrants are measured at market value on a recurring basis using the fixed monetary amount of each warrant that would be received by the Company under the conditions specified in the stock redemption agreement and are being marked to market each quarter-end until they are completely settled. The warrants are valued using the Black-Scholes method, using assumptions consistent with our application of ASC 718. See footnote 9.
63
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
In accordance with the provisions of ASC 820, the Company has elected to defer implementation of ASC 820, as it relates to its financialnon-financial assets and liabilities that are recognized and disclosed at fair value in the financial statements on a nonrecurring basis until January 1,the first quarter of 2010. The Company is evaluating the impact, if any, this standard will have on its financial assets and liabilities. The adoption of ASC 820, as it relates to the Company’s financialnon-financial assets and liabilities that are re-measured and reported at fair value at least annually, did not have an impact on the Company’s financial results.
| Quoted Prices in | Significant Other | |||||||||||||||
| December 31, | Active Markets | Observable Inputs | Unobservable Inputs | |||||||||||||
Description | 2010 | (Level 1) | (Level 2) | (Level 3) | ||||||||||||
| Assets: | ||||||||||||||||
| Cash equivalents | $ | 6,891 | $ | 6,891 | $ | — | $ | — | ||||||||
| Total assets | $ | 6,891 | $ | 6,891 | $ | — | $ | — | ||||||||
| Liabilities: | ||||||||||||||||
| Warrants potentially settleable in cash | $ | 3,138 | $ | — | $ | 3,138 | $ | — | ||||||||
| Total liabilities | $ | 3,138 | $ | — | $ | 3,138 | $ | — | ||||||||
| Quoted Prices in | Significant Other | |||||||||||||||
| December 31, | Active Markets | Observable Inputs | Unobservable Inputs | |||||||||||||
Description | 2019 | (Level 1) | (Level 2) | (Level 3) | ||||||||||||
| Assets: | ||||||||||||||||
| Cash equivalents | $ | 5,684 | $ | 5,684 | $ | — | $ | — | ||||||||
| Total assets | $ | 5,684 | $ | 5,684 | $ | — | $ | — | ||||||||
| Liabilities: | ||||||||||||||||
| Warrants potentially settleable in cash | 3,721 | — | 3,721 | — | ||||||||||||
| Total liabilities | $ | 3,721 | $ | — | $ | 3,721 | $ | — | ||||||||
| 5. | Deposits |
At December 31, 20092010 and 2008,2009, the Company had $16,000 on deposit with its landlords related to leased facilities, all of which are classified as deposits.
| 6. | Capital Lease Obligations |
The Company acquires equipment under capital lease obligations. Accordingly, the Company capitalized approximately $100,000 and $43,000 of equipment during the accompanying years ended December 31, 2009 and 2008, respectively, and thisleases that is included in equipment and furnishings onin the balance sheet. AmortizationThe cost and accumulated amortization of capitalized leased equipment for the years endedwas approximately $196,000 and $56,000 at December 31, 20092010, respectively, and 2008 was approximately $10,000$143,000 and $7,000, respectively. Accumulated amortization of capitalized lease equipment was approximately $17,000 and $7,000 at December 31, 2009, respectively. Amortization expense for capitalized leased equipment was approximately $39,000 in 2010 and 2008, respectively.$10,000 in 2009. Future minimum lease payments under the capital leaseleases including interest are $52,000, $24,000$62,000, $17,000 and $2,000$4,000 for the years ending December 31, 2010, 2011, 2012 and 2012,2013, respectively.
64
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
| 7. | Accrued Expenses and Other Current Liabilities |
Accrued expenses and other current liabilities consist of the following (in thousands):
| For the Years | ||||||||||||||||
| For the Year Ended | Ended | |||||||||||||||
| December 31, | December 31, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
| Professional fees | $ | 390 | $ | 398 | $ | 313 | $ | 390 | ||||||||
| Research and development costs | 28 | 47 | 60 | 28 | ||||||||||||
| Payroll related costs | 659 | 531 | 740 | 659 | ||||||||||||
| Total accrued expenses and other current liabilities | $ | 1,077 | $ | 976 | $ | 1,113 | $ | 1,077 | ||||||||
| 8. | Commitments and Contingencies |
The Company acquires assets still in development and enters into research and development arrangements with third parties that often require milestone and royalty payments based on the progress of the asset through development stages. Milestone payments may be required, for example, upon approval of the product for marketing by a regulatory agency. In certain agreements, RXithe Company is required to make royalty payments based
59
upon a percentage of the sales. Because of the contingent nature of these payments, they are not included in the table of contractual obligations shown below. See footnote 15.below (see also Note 14).
These arrangements may be material individually, and in the unlikely event that milestones for multiple products covered by these arrangements were reached in the same period, the aggregate charge to expense could be material to the results of operations. In addition, these arrangements often give RXithe Company the discretion to unilaterally terminate development of the product, which would allow RXithe Company to avoid making the contingent payments; however, RXithe Company is unlikely to cease development if the compound successfully achieves clinical testing objectives. The Company’s contractual obligations that will require future cash payments as of December 31, 20092010 are as follows (in thousands):
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
Years Ending December 31, | Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | |||||||||||||||
| 2010 | $ | 239 | $ | 1,389 | $ | 1,628 | $ | 2,068 | $ | 3,696 | ||||||||||
| 2011 | 22 | 198 | 220 | 875 | 1,095 | |||||||||||||||
| 2012 | 73 | 73 | 930 | 1,003 | ||||||||||||||||
| 2013 | — | — | — | 1,400 | 1,400 | |||||||||||||||
| 2014 | — | — | — | 735 | 735 | |||||||||||||||
| thereafter | — | — | — | 13,481 | 13,481 | |||||||||||||||
| Total | $ | 261 | $ | 1,660 | $ | 1,921 | $ | 19,489 | $ | 21,410 | ||||||||||
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
| Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| 2011 | $ | 219 | $ | 1,289 | $ | 1,508 | $ | 535 | $ | 2,043 | ||||||||||
| 2012 | 19 | 333 | 352 | 2,068 | 2,420 | |||||||||||||||
| 2013 | 0 | 0 | — | 875 | 875 | |||||||||||||||
| 2014 | 0 | 0 | — | 930 | 930 | |||||||||||||||
| 2015 | 0 | 0 | — | 1,400 | 1,400 | |||||||||||||||
| 2016 and Thereafter | 0 | 0 | — | 14,215 | 14,215 | |||||||||||||||
| Total | $ | 238 | $ | 1,622 | $ | 1,860 | $ | 20,023 | $ | 21,883 | ||||||||||
| (1) | Operating leases are primarily facility and equipment related obligations with third party vendors. Operating lease expenses during the years ended December 31, | |
| (2) | Employment agreement obligations include management contracts, as well as scientific advisory board member compensation agreements. Certain agreements, which have been revised from time to time, provide for minimum salary levels, adjusted annually at the discretion of the Compensation Committee, as well as for minimum bonuses that are payable. | |
| (3) | License agreements generally relate to the Company’s obligations with UMMS associated with RNAi and, for future periods, represent minimum annual royalty and milestone payment obligations, of the total |
65
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
| amount due $2,250,000 can be paid in equity, provided that the securities are registered for resale at the time of such payment. The Company continually assesses the progress of its licensed technology and the progress of its research and development efforts as it relates to its licensed technology and may terminate with notice to the licensor at any time. In the event these licenses are terminated, no amounts will be due. |
The Company applies the disclosure provisions FASB ASC Topic 460 (“ASC 460”), “Guarantor’s Accounting and Disclosure Requirements for Guarantees, Including Indirect Guarantees of Indebtedness of Others”, to its agreements that contain guarantee or indemnification clauses. The Company provides (i) indemnifications of varying scope and size to certain investors and other parties for certain losses suffered or incurred by the indemnified party in connection with various types of third-party claims and (ii) indemnifications of varying scope and size to officers and directors against third party claims arising from the services they provide to us. These indemnifications give rise only to the disclosure provisions of ASC 460. To date, the Company has not incurred costs as a result of these obligations and does not expect to incur material costs in the future. Accordingly, the Company has not accrued any liabilities in its financial statements related to these indemnifications.
| 9. | Stockholders’ Equity |
Preferred Stock — The Company has authorized up to 5,000,000 shares of preferred stock, $0.00001$0.0001 par value per share, for issuance. The preferred stock will have such rights, preferences, privileges and restrictions,
60
including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences, as shall be determined by the Company’s board of directors upon its issuance. At December 31, 2009,2010, there were no shares of preferred stock outstanding.
Common Stock Warrants — On August 7, 2008, the Company issued 190,000 warrants to an investment bank as consideration for investment and business advisory services. The warrants have an exercise price of $7.036 per share and expire 5 years from the date of issuance, on August 7, 2013. The warrantwarrants vested as to 94,000 shares upon issuance, and vested at a rate of 32,000 shares per month starting on the 90 day anniversary of issuance, and isare exercisable for a period of five years. All shares were vested at December 31, 2009. The Company also agreed to give the holder of the warrantwarrants unlimited “piggy back” registration rights with respect to the shares of the Company’s common stock underlying the warrantwarrants in any registration statement the Company files in connection with an underwritten offering of its common stock. The fair value of these warrants has been estimated based on the Black-Scholes options pricing model and changes in the fair value are recorded in the condensed statement of expenses in accordance with the requirements of ASC 718 andASC505-50. There was no expense recorded for these warrants for the year ended December 31, 2010. Total expense related to these warrants was approximately $318,000 and $750,000 during the yearsyear ended December 31, 2009 and 2008, respectively2009.
On October 3, 2008, the Company acquired the rights to license exclusive worldwide technology for the oral delivery of RNAi therapeutics. As consideration for this license, the Company agreed to pay a total license fee of $2.5 million$2,500,000 over a 12 month period, which can be paid in cash, in equity or a combination thereof, provided that a specified amount of the license fee must be made in cash. During 2009, this 12 month period was extended to a date after January 1, 2010 to be agreed upon by the parties. Payments made in equity may only be made if, at the time of such payment, the shares of common stock issuable upon conversion of the warrant have been registered for resale under the Securities Act of 1933. No warrants have been issued under this agreement throughthru the date of this report. The Company continually assesses the progress of its research and development efforts as it relates to its licensed technology and may terminate with notice to the Licensor at any time. Accordingly, the amounts are being expensed, as payments are made. DuringThere was no expense for this license for the year ended December 31, 2010. Total expense for the year ended December 31, 2009 and 2008, the Company paid and expensed $250,000 each year under this agreement.was $250,000.
On January 29, 2009, the Company issued 142,500 warrants to an investment bank as consideration for investment and business advisory services. The warrants have an exercise price of $4.273 per share and expire five years from the date of issuance on January 29, 2014. The warrants vested as to 71,250 shares upon
66
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
issuance, and vested at a rate of 23,750 shares per month starting on the 90 day anniversary of issuance, and are exercisable for a period of five years. All shares were vested at December 31, 2009. The Company has also agreed to give the holder of the warrants unlimited “piggy back” registration rights with respect to the shares of Common Stock underlying the warrants in any registration statement the Company files in connection with an underwritten offering of the common stock. The fair value of these warrants has been estimated based on the Black-Scholes options pricing model and changes in the fair value are recorded in the condensed statement of expenses in accordance with the requirements of ASC Topic 718 and ASC Topic505-50. There was no expense recorded for these warrants for the year ended December 31, 2010. Total expense related to these warrants was approximately $509,000 during the year ended December 31, 2009.
In connection with the 2009 Offering, the Company issued warrants to purchase 978,142 shares of the Company’s common stock. Details of the transaction can be found under the heading 2009 Registered Direct Offering below.
In connection with the 2010 Offering, the Company issued warrants to purchase 540,000 shares of the Company’s common stock. Details of the transaction can be found under the heading 2010 Registered Direct Offering below.
Private Investment in Public Equity — On June 24, 2008, the Company entered into a Securities Purchase Agreement pursuant to which RXi issued and sold to certain investors an aggregate of 1,073,299 shares of common stock in a private placement at a price of $8.12 per share. Net proceeds to the Company were approximately $7.9 million. The Company agreed to file a registration statement covering the resale of all shares issued in the private placement, with all expenses incurred in connection with such registration to be borne by the Company. The registration statement went effective on August 6, 2008.
61
2009 Registered Direct Offering — On March 17, 2009, the Company entered into a placement agency agreement, which was subsequently amended on May 26, 2009 and July 22, 2009, with Rodman & Renshaw, LLC (“Rodman”) as the exclusive placement agent, relating to a proposed offering by the Company of new securities to potential investors. On July 30, 2009, the Company entered into definitive agreements for the sale and issuance by the Company to certain investors of 2,385,715 units, with each unit consisting of one share of the Company’s common stock and a warrant to purchase 0.40 of a share of common stock, at a purchase price of $3.50 per unit (the “2009 Offering”). The 2009 Offering closed on August 4, 2009. The warrants have an exercise price of $4.50 per share and are exercisable for a period beginning on February 3, 2010 until their expiration on August 3, 2014. The Company raised gross proceeds of approximately $8,350,000 in the 2009 Offering and net cash proceeds, after deducting the placement agents’ fees and other offering expenses payable by the Company, of approximately $7.7 million. Total warrants issued in connection with the transaction were 954,285.
As part of the placement agency agreement, the Company issued a warrant to purchase 23,857 shares of the Company’s common stock to Rodman. The warrant has an exercise price of $4.38 per share. The warrant is immediately vested and is exercisable until its expiration on August 3, 2014.
The Company follows the guidance of ASC Topic815-40, as certain warrants issued in connection with the stock offering on August 4, 2009 were determined not to be indexed to the Company’s common stock as they are potentially settleable in cash. The fair value of the warrants at the dates of issuance totaling $2,862,640$2,863,000 was recorded as a liability and a cost of equity and was determined by the Black-Scholes option pricing model. Due to the fact that the Company has limited trading history, the Company’s expected stock volatility assumption is based on a combination of implied volatilities of similar entities whose share or option prices are publically traded. The Company used a weighted average expected stock volatility of 122.69%. The expected life assumption is based on the contract term of five years. The dividend yield of zero is based on the fact that RXi has no present intention to pay cash dividends in the future. The risk free rate of 1.72% used for the warrant is equal to the zero coupon rate in effect at the time of the grant. The increasedecrease in the fair value
67
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
of the warrants from the date of issuance to December 31, 20092010 of $858,000$920,000 has been included as an offset to other expense in the accompanying condensed statements of expenses for the respective period. The fair value of the warrants at December 31, 20092010 of $3,721,000$1,943,000 is included as a current liability in the accompanying balance sheet as of that date and was determined by the Black-Scholes option pricing model. The following assumptions were used to determine the fair value as of December 31, 2009:2010: weighted average expected stock volatility of 119.10%133.62%; an expected life of fivefour years based on the contractual terms;terms and a dividend yield of zero;zero and a risk free rate of 2.69%1.48%.
2010 Registered Direct Offering — On March 22, 2010, the Company entered into a placement agency agreement relating to a proposed offering by the Company of new securities to potential investors. On March 23, 2010, the Company entered into definitive agreements for the sale and issuance by the Company to certain investors of 2,700,000 units, with each unit consisting of one share of the Company’s common stock and a warrant to purchase 0.20 of a share of the Company’s common stock, at a purchase price of $6.00 per unit (the “2010 Offering”). The 2010 Offering closed on March 26, 2010. The Company issued warrants to purchase 540,000 shares of the Company’s common stock at an exercise price of $6.00 per share and that are exercisable beginning on September 26, 2010 until their expiration on March 26, 2016. The Company raised gross proceeds of approximately $16.2 million in the 2010 Offering and net cash proceeds, after deducting the placement agent fees and other offering expenses payable by the Company, of approximately $15.2 million.
As part of the 2010 Offering, RXi entered in a stock redemption agreement whereby the Company was required to use 25% of the net proceeds from the 2010 Offering to repurchase from CytRx Corporation (“CytRx”) 675,000 shares of the Company’s common stock held by CytRx (“CytRx shares”). The Company repurchased such shares on March 29, 2010. The values of the shares at the date of repurchase totaling $3,849,000 were recorded at cost and have been included in treasury stock in the accompanying balance sheet at December 31, 2010. The Company is also required to use 25% of the proceeds from the exercise of warrants issued in the 2010 Offering to repurchase from CytRx a number of CytRx Shares equal to 25% of shares issued upon the exercise of such warrants. To date, no such warrants have been exercised.
Shares of common stock that are mandatorily redeemable under the stock redemption agreement upon the exercise of warrants issued in the 2010 Offering, were determined to embody an obligation that may require the Company to settle the obligation by transferring assets, and as such, shall be classified as a liability. The fair value of the common stock potentially redeemable under the stock redemption agreement totaling $785,000 was recorded as a liability and a cost of equity and was determined using the fixed monetary amount of each warrant multiplied by assumptions regarding the number and timing of warrants to be exercised. On December 29, 2010, CytRx sold all of their shares held in RXi, thus reducing the potential redemption liability to zero as December 31, 2010.
Certain warrants issued in connection with the 2010 Offering were determined not to be indexed to the Company’s common stock as they are potentially settleable in cash. The fair value of the warrants at the dates of issuance totaling $2,466,000 was recorded as a liability and a cost of equity and was determined by the Black-Scholes option pricing model. Due to the fact that the Company has limited trading history, the Company’s expected stock volatility assumption is based on a combination of implied volatilities of similar entities whose share or option prices are publically traded. The Company used a weighted average expected stock volatility of 119.49%. The expected life assumption is based on the contract term of 6.5 years. The dividend yield of zero is based on the fact that the Company has no present intention to pay cash dividends. The risk free rate of 3.22% used for the warrant is equal to the zero coupon rate in effect at the time of the grant. The decrease in the fair value of the warrants from the date of issuance to December 31, 2010 was $1,271,000 and has been included in other income and expense in the accompanying condensed statements of expenses for the year ended December 31, 2010. The fair value of the warrants at December 31, 2010 of $1,195,000 is included as a current liability in the accompanying balance sheets and was determined by the Black-Scholes option pricing model. In the model, the Company used a weighted average expected stock
6268
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
volatility of 133.62%. The expected life assumption is based on the contract term of 6.0 years. The dividend yield of zero is based on the fact that the Company has no present intention to pay cash dividends. The risk free rate of 2.37% used for the warrant is equal to the zero coupon rate in effect on the date of the re-measurement.
On January 30, 2009, the Company entered into a Standby Equity Distribution Agreement, or SEDA, pursuant to which the Company may, at its option over a two-year period, ending on January 30, 2011, periodically to sell to YA Global shares of the Company’s common stock, for a total purchase price of up to $25.0 million. To date no shares have been sold under the SEDA. The SEDA expired in January 2011 and was not renewed.
On August 4, 2009, the Company closed the 2009 Offering in which it sold 2,385,715 shares of its common stock and warrants to purchase 954,286 shares of its common stock and a at an exercise price of $4.50 per share resulting in approximately $7.8 million in net proceeds after deducting the placement agent fee and offering expenses.
| 10. | Development Stage Supplemental Equity Disclosure |
Summarized below are the Company’s equity (common stock and common stock options) transactions since the Company’s inception through December 31, 20092010 (in thousands except per share data).
| Price per | Price per | |||||||||||||||||||||||||||||||||||||||
| Share or | Share or | |||||||||||||||||||||||||||||||||||||||
| Dollar | Exercise | Dollar | Exercise | |||||||||||||||||||||||||||||||||||||
| Shares of | Amount of | Price per | Counter | Nature of | Shares of | Amount of | Price per | Counter | Nature of | |||||||||||||||||||||||||||||||
| Common | Consideration | Share | Party to | Non-Cash | Basis of | Common | Consideration | Share | Party to | Non-Cash | Basis of | |||||||||||||||||||||||||||||
Type of Security | Date of Issuance | Stock | ($) | ($) | Transaction | Consideration | Assigning Cost | Date of Issuance | Stock | ($) | ($) | Transaction | Consideration | Assigning Cost | ||||||||||||||||||||||||||
| Common Stock | April 3, 2006 | 1,624,278 | 2 | 0.002 | Founders | NA | Cash | April 3, 2006 | 1,624,278 | 2 | 0.002 | Founders | NA | Cash | ||||||||||||||||||||||||||
| Common Stock | January 8, 2007 | 7.040,318 | 48 | (A) | 0.007 | CytRx | Contributed Assets | Predecessor Cost | January 8, 2007 | 7.040,318 | 48 | (A) | 0.007 | CytRx | Contributed Assets | Predecessor Cost | ||||||||||||||||||||||||
| Common Stock | April 30, 2007 | 3,273,292 | 15,348 | (B) | 5.19 | CytRx | NA | Cash | April 30, 2007 | 3,273,292 | 15,348 | (B) | 5.19 | CytRx | NA | Cash | ||||||||||||||||||||||||
| Common Stock | April 30, 2007 | 462,112 | 2,311 | 5.00 | UMMS | Intellectual Properties | Independent Third Party Valuation | April 30, 2007 | 462,112 | 2,311 | 5.00 | UMMS | Intellectual Properties | Independent Third Party Valuation | ||||||||||||||||||||||||||
| Common Stock | August 18, 2007 | 30,000 | 150 | 5.00 | Directors | — | Cash | August 18, 2007 | 30,000 | 150 | 5.00 | Directors | — | Cash | ||||||||||||||||||||||||||
| Common Stock | September 28, 2007 | 188,387 | 978 | 5.19 | CytRx | NA | Independent Third Party Valuation | September 28, 2007 | 188,387 | 978 | 5.19 | CytRx | NA | Independent Third Party Valuation | ||||||||||||||||||||||||||
| Common Stock | November 21, 2007 | 66,045 | 331 | 5.00 | Exercise of Stock Options | NA | Cash | November 21, 2007 | 66,045 | 331 | 5.00 | Exercise of Stock Options | NA | Cash | ||||||||||||||||||||||||||
| Common Stock | June 26, 2008 | 1,073,299 | 7,918 | 8.12 | PIPE | NA | Net Cash | June 26, 2008 | 1,073,299 | 7,918 | 8.12 | PIPE | NA | Net Cash | ||||||||||||||||||||||||||
| Common Stock | October 6, 2008 and November 16, 2008 | 5,500 | 26 | 5.00 | Exercise of Stock Options | NA | Cash | October 6, 2008 and November 16, 2008 | 5,500 | 26 | 5.00 | Exercise of Stock Options | NA | Cash | ||||||||||||||||||||||||||
| Common Stock | January 30, 2009 | 58,398 | NA | NA | NA | Market Value | January 30, 2009 | 58,398 | NA | NA | NA | Market Value | ||||||||||||||||||||||||||||
| Common Stock | May 1, 2009 | 281 | NA | 4.19 | Exercise of Stock Options | NA | Cash | May 1, 2009 | 281 | NA | 4.19 | Exercise of Stock Options | NA | Cash | ||||||||||||||||||||||||||
| Common Stock | August 3, 2009 and August 4, 2009 | 2,385,715 | 7,714 | 3.50 | Register Direct | NA | Net Cash | August 3, 2009 and August 4, 2009 | 2,385,715 | 7,714 | 3.50 | Registered Direct | NA | Net Cash | ||||||||||||||||||||||||||
| Common Stock | March 22, 2010 | 2,700,000 | 15,235 | 6.00 | Registered Direct | NA | Net Cash | |||||||||||||||||||||||||||||||||
| Common Stock | Various — 2010 | 53,500 | 254 | 4.75 | Exercise of Stock Options | NA | Cash | |||||||||||||||||||||||||||||||||
| Common Stock | January 2, 2010 and February 9, 2010 | 86,634 | 207 | NA | Restricted Stock Units | NA | Market Value | |||||||||||||||||||||||||||||||||
69
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
| (A) | Transactions between related parties are accounted for at the historical cost of CytRx, with the intellectual property which was previously expensed on CytRx’s books being recorded at zero cost and equipment and furnishings being recorded at $48,000. | |
| (B) | RXi received gross proceeds of $17.0 million for the issuance of the 3,273,292 shares of common stock which equals $5.19 per share. The gross proceeds were reduced by a reimbursement to CytRx of (1) $1.3 million for RXi’s pro rata share of offering costs related to the April 17, 2007 private placement conducted by CytRx to fund its capital contribution to the Company and (2) $363,000 of expenses incurred on behalf of RXi for the year ended December 31, 2006. Net proceeds to RXi after these charges were $15.3 million or $4.69 a share. |
| 11. | Stock Based Compensation |
Options to Purchase Shares of Common Stock — The Company follows the provisions ASC 718 which requires the measurement and recognition of compensation expense for all share-based payment awards made to employees, non-employee directors, and consultants, including employee stock options. Stock compensation expense based on the grant date fair value estimated in accordance with the provisions of ASC 718 is recognized as an expense over the requisite service period.
For stock options granted as consideration for services rendered by non-employees, the Company recognizes compensation expense in accordance with the requirements of FASB ASC Topic505-50(“ASC 505-50”)505-50 “Equity Based Payments to Non- Employees.” Non-employee option grants that do not vest
63
immediately upon grant are recorded as an expense over the vesting period of the underlying stock options. At the end of each financial reporting period prior to vesting, the value of these options, as calculated using the Black-Scholes option-pricing model, will beis being re-measured using the fair value of the Company’s common stock and the non-cash compensation recognized during the period will be adjusted accordingly. Since the fair market value of options granted to non-employees is subject to change in the future, the amount of the future compensation expense will include fair value re-measurements until the stock options are fully vested.
The Company is currently using the Black-Scholes option-pricing model to determine the fair value of all its option grants. For options grants issued for the year ended December 31, 20092010 and 2008,2009, the following assumptions were used:
| 2009 | 2008 | 2010 | 2009 | |||||
| Weighted average risk free interest rate | 1.55% - 3.91% | 1.55% - 3.99% | 1.88% - 3.28% | 1.55% - 3.91% | ||||
| Weighted average volatility | 116.72% - 122.93% | 101.79% - 116.74% | 118.3% - 133.62% | 116.72% - 122.93% | ||||
| Expected lives (years) | 6 - 10 | 6 - 10 | 6 - 10 | 6 - 10 | ||||
| Expected dividend yield | 0% | 0% | 0% | 0% |
The weighted average fair value of options granted during the years ended December 31, 2010 and 2009 was $4.31 and 2008 was $4.11 and $6.37 per share, respectively.
RXi’s expected common stock price volatility assumption is based upon the volatility of a basket of comparable companies. The expected life assumptions for employee grants were based upon the simplified method provided for under ASC 718, which averages the contractual term of RXi’s options of ten years with the average vesting term of four years for an average of six years. The expected life assumptions for non-employees were based upon the contractual term of the option. The dividend yield assumption of zero is based upon the fact that RXi has never paid cash dividends and presently has no intention of paying cash dividends in the future. The risk-free interest rate used for each grant was also based upon prevailing short-term interest rates. RXi has estimated an annualized forfeiture rate of 4.0%15% for options granted to its employees, 2.1%8% for options granted to senior management and no forfeiture rate for the directors. RXi will record additional expense if the actual forfeitures are lower than estimated and will record a recovery of prior expense if the actual forfeiture rates are higher than estimated.
70
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
RXi recorded approximately $4,202,000$4,368,000 and $3,824,000$4,202,000 of stock-based compensation for the years ended December 31, 20092010 and 2008,2009, respectively. As of December 31, 2009,2010, there was $4.7 million$2,700,000 of unrecognized compensation cost related to outstanding options that is expected to be recognized as a component of RXi’s operating expenses through 2013.
On November 4, 2009, as part of a plan succession in leadership, Tod Woolf, Ph.D., resigned as our President, Chief Executive Officer and a member of our Board of Directors. According to the Separation Agreement between Dr. Woolf and the Company, Dr. Woolf received in one lump sum payment his full severance equivalent to a six (6) month salary ($187,500), six (6) months acceleration of vesting of all of his outstanding unvested Stock Options as of November 4, 2009, and an offer to join the Company’s Scientific Advisory Board (SAB) for 3 years (the “New Position”). In addition, and as part of the Separation Agreement, the Company agreed to extend the exercise period for all of Dr. Woolf’s vested Stock Options as of November 4, 2009, to the later of: (i) a period of two (2) years from his resignation (until November 4, 2011), or (ii) ninety (90) days following the end of the term of the SAB Agreement (February 4, 2013) or such earlier date as the SAB Agreement may be terminated pursuant to the terms of the SAB Agreement provided Dr. Woolf has not violated the non-competition provisions of the SAB Agreement prior to the date of exercise (whether or not the SAB Agreement is still in effect at that time). Notwithstanding any provision of the Company’s 2007 Incentive Plan, the Company also agreed that Dr. Woolf’s previously awarded Stock Options shall continue to vest during his continuing role in the Company in the New Position. The option modification resulted in an incremental value of the options of approximately $153,000 of which $37,000 was expensed during 2009. The remaining $411,000 will be expensed over the remaining vesting periodtotal expense for 2010 was $193,000. As of approximately 3 years.
64
As of December 31, 2009,2010, an aggregate of 4,750,0006,750,000 shares of common stock were reserved for issuance under the RXi Pharmaceuticals Corporation 2007 Incentive Plan, including 3,630,8394,333,136 shares subject to outstanding common stock options and restricted stock units granted under this plan and 1,047,3352,199,497 shares available for future grants. The administrator of the plan determines the times when an option may become exercisable. Vesting periods of options granted to date include vesting upon grant to vesting at the end of a four year period. The options will expire, unless previously exercised, no later than ten years from the grant date.
The following table summarizes the activity of the Company’s stock option plan:
| Weighted | Weighted | |||||||||||||||
| Stock | Average | Stock | Average | |||||||||||||
| Options | Exercise Price | Options | Exercise Price | |||||||||||||
| Outstanding — January 1, 2008 | 1,335,184 | $ | 5.00 | |||||||||||||
| Outstanding — January 1, 2009 | 2,223,452 | $ | 6.11 | |||||||||||||
| Granted | 1,622,546 | 3.84 | ||||||||||||||
| Exercised | (281 | ) | 4.19 | |||||||||||||
| Forfeited | (263,378 | ) | 5.05 | |||||||||||||
| Outstanding — December 31, 2009 | 3,582,339 | 5.16 | ||||||||||||||
| Granted | 899,609 | 7.76 | 926,768 | 4.81 | ||||||||||||
| Exercised | (5,500 | ) | 5.00 | (53,500 | ) | 4.75 | ||||||||||
| Forfeited | (5,841 | ) | 6.03 | (122,471 | ) | 4.85 | ||||||||||
| Outstanding — December 31, 2008 | 2,223,452 | 6.11 | ||||||||||||||
| Granted | 1,622,546 | 3.84 | ||||||||||||||
| Exercised | (281 | ) | 4.19 | |||||||||||||
| Forfeited | (263,378 | ) | 5.05 | |||||||||||||
| Outstanding — December 31, 2009 | 3,582,339 | 5.16 | ||||||||||||||
| Exercisable — December 31, 2008 | 1,238,187 | 5.65 | ||||||||||||||
| Outstanding — December 31, 2010 | 4,333,136 | 5.10 | ||||||||||||||
| Exercisable — December 31, 2009 | 2,131,298 | 5.42 | 2,131,298 | 5.42 | ||||||||||||
| Exercisable — December 31, 2010 | 3,155,900 | 5.22 | ||||||||||||||
71
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The following table summarizes the activity for non-vested stock options:
| Weighted Average | Weighted Average | |||||||||||||||
| Stock | Grant Date Fair | Stock | Grant Date Fair | |||||||||||||
| Options | Value per Share | Options | Value per Share | |||||||||||||
| Non-vested at January 1, 2008 | 839,361 | $ | 3.54 | |||||||||||||
| Granted | 899,609 | 6.37 | ||||||||||||||
| Vested | (740,364 | ) | 4.94 | |||||||||||||
| Exercised | (5,500 | ) | 3.93 | |||||||||||||
| Pre-vested forfeitures | (5,841 | ) | 4.76 | |||||||||||||
| Non-vested at December 31, 2008 | 987,265 | 5.15 | ||||||||||||||
| Non-vested at January 1, 2009 | 987,265 | $ | 5.15 | |||||||||||||
| Granted | 1,622,546 | 3.44 | 1,622,546 | 3.44 | ||||||||||||
| Vested | (895,111 | ) | 4.27 | (895,111 | ) | 4.27 | ||||||||||
| Exercised | (281 | ) | 3.71 | (281 | ) | 3.71 | ||||||||||
| Pre-vested forfeitures | (263,378 | ) | 4.07 | (263,378 | ) | 4.07 | ||||||||||
| Non-vested at December 31, 2009 | 1,451,041 | 3.94 | 1,451,041 | 3.94 | ||||||||||||
| Granted | 926,768 | 4.31 | ||||||||||||||
| Vested | (1,024,602 | ) | 4.06 | |||||||||||||
| Exercised | (53,500 | ) | 3.31 | |||||||||||||
| Pre-vested forfeitures | (122,471 | ) | 4.54 | |||||||||||||
| Non-vested at December 31, 2010 | 1,177,236 | $ | 4.01 | |||||||||||||
The weighted average remaining contractual life of options outstanding and exercisable at December 31, 2010 was 7.35 years and 7.09 years, respectively. The weighted average remaining contractual life of options outstanding and exercisable at December 31, 2009 was 8.47 years and 8.14 years, respectively. The weighted average remaining contractual life of options outstanding and exercisable at December 31, 2008 was 8.832 years and 8.733 years, respectively.
The aggregate intrinsic value of outstanding options as of December 31, 2010 and 2009 is $137,000 and 2008 is $1,262,270 and $653,974,$1,262,000, respectively. The aggregate intrinsic value of exercisable options as of December 31, 2010 and 2009 is $34,000 and 2008 is $138,881 and $654,000,$139,000, respectively. The aggregate intrinsic value is calculated based on the positive difference between the closing fair market value of RXi’sthe Company’s common stock and the exercise price of the underlying options.
65
The aggregate intrinsic value of options exercised during 20092010 and 20082009 was approximately $1,000$164,000 and $18,000,$1,000, respectively.
Restricted Stock Units — In addition to options to purchase shares of common stock, the Company may grant restricted stock units (“RSU”) as part of its compensation package. Each RSU is granted at the fair market value based on the date of grant. Vesting is determined on a grant by grant basis. As of December 31,
In 2010 and 2009, the Company had granted a total of 43,541 and 48,500 RSUs, withrespectively. The RSUs granted in 2010 and 2009 had an aggregate intrinsic value of $222,000$112,000 and recognized total expense$222,000. As of $105,000. TheDecember 31, 2010, all of the RSUs vesthad vested in full on January 2, 2010.full.
| 12. | Net Loss Per Share |
The Company accounts for and discloses net loss per common share in accordance with FASB ASC Topic 260 “Earnings per Share.”Basic net loss per common share is computed by dividing net loss attributable to common stockholders by the weighted average number of common shares outstanding. Diluted net loss per common share is computed by dividing net loss attributable to common stockholders by the weighted average number of common shares that would have been outstanding during the period assuming the issuance of common shares for all potential dilutive common shares outstanding. Potential common shares consist of shares issuable upon the exercise of stock options and warrants. Because the inclusion of potential common shares would be anti-dilutive for all periods presented, diluted net loss per common share is the same as basic net loss per common share.
72
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The following table sets forth the potential common shares excluded from the calculation of net loss per common share because their inclusion would be anti-dilutive:
| December 31, | December 31, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
| Options to purchase common stock | 3,582,339 | 2,223,452 | 4,333,136 | 3,582,339 | ||||||||||||
| Restricted stock units | 48,500 | — | — | 48,500 | ||||||||||||
| Warrants to purchase common stock | 1,310,642 | 190,000 | 2,100,642 | 1,310,642 | ||||||||||||
| Total | 4,941,481 | 2,413,452 | 6,433,778 | 4,941,481 | ||||||||||||
| 13. | Income Taxes |
The components of federal and state income tax expense are as follows (in thousands):
| As of December 31, | As of December 31, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
| Current | ||||||||||||||||
| Federal | $ | — | $ | — | $ | — | $ | — | ||||||||
| State | — | — | — | — | ||||||||||||
| Deferred | ||||||||||||||||
| Federal | (5,533 | ) | (4,466 | ) | 4,853 | (5,533 | ) | |||||||||
| State | (2,257 | ) | (1,513 | ) | 1,283 | (2,257 | ) | |||||||||
| Total deferred | (7,789 | ) | (5,979 | ) | 6,136 | (7,790 | ) | |||||||||
| Valuation allowance | 7,789 | 5,979 | 6,136 | 7,790 | ||||||||||||
| Total income tax expense | $ | — | $ | — | $ | — | $ | — | ||||||||
66
The components of net deferred tax assets are as follows (in thousands):
| As of December 31, | ||||||||||||||||
| 2009 | 2008 | As of December 31, | ||||||||||||||
| 2010 | 2009 | |||||||||||||||
| Net operating loss carryforwards | $ | 10,348 | $ | 6,710 | $ | 13,328 | $ | 10,348 | ||||||||
| Tax credit carryforwards | 753 | 948 | 1,061 | 753 | ||||||||||||
| Non-qualified stock based compensation | 4,222 | 2,471 | ||||||||||||||
| Stock based compensation | 5,864 | 4,222 | ||||||||||||||
| Other | 74 | 28 | 104 | 74 | ||||||||||||
| Licensing deduction deferral | 2,089 | 1,225 | 3,264 | 2,089 | ||||||||||||
| Gross deferred tax assets | 17,486 | 11,382 | 23,621 | 17,486 | ||||||||||||
| Valuation allowance | (17,486 | ) | (11,382 | ) | (23,621 | ) | (17,486 | ) | ||||||||
| Net deferred tax asset | $ | — | $ | — | $ | — | $ | — | ||||||||
73
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The provision for income taxes differs from the provision computed by applying the federal statutory rate to net loss before income taxes as follows (in thousands):
| As of December 31, | As of December 31, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
| Expected federal income tax benefit | $ | (6,252 | ) | $ | (4,887 | ) | $ | (4,078 | ) | $ | (6,252 | ) | ||||
| Non-qualified stock compensation | 621 | 186 | (1,236 | ) | 621 | |||||||||||
| Effect of change in valuation allowance | 6,103 | 6,707 | 6,136 | 6,103 | ||||||||||||
| State income tax credits | 821 | (426 | ) | |||||||||||||
| Income tax credits | (231 | ) | 821 | |||||||||||||
| State income taxes after credits | (324 | ) | (867 | ) | (994 | ) | (324 | ) | ||||||||
| Other | (969 | ) | (713 | ) | 403 | (969 | ) | |||||||||
| $ | — | $ | — | $ | — | $ | — | |||||||||
The Company has incurred net operating losses from inception. At December 31, 2009,2010, the Company had domestic federal and state net operating loss carryforwards of approximately $25.7$34.0 million available to reduce future taxable income, which expire at various dates beginning in 2012 through 2029.2030. The Company also had federal and state research and development tax credit carryforwards of approximately $500,000$705,000 and $400,000,$536,000, respectively, available to reduce future tax liabilities and which expire at various dates beginning in 2022 through 2029.2030.
Under the provisions of the Internal Revenue Code, certain substantial changes in the Company’s ownership may result in a limitation on the amount of net operating loss carryforwards and research and development credit carryforwards which could be utilized annually to offset future taxable income and taxes payable.
Based on an assessment of all available evidence including, but not limited to the fact the RXi operating results have been included in CytRx consolidated U.S. federal and state income tax return for the year ended December 31, 2007, the Company’s limited operating history in its core business and lack of profitability, uncertainties of the commercial viability of its technology, the impact of government regulation and healthcare reform initiatives, and other risks normally associated with biotechnology companies, the Company has concluded that it is more likely than not that these net operating loss carryforwards and credits will not be realized and, as a result, a 100% deferred income tax valuation allowance has been recorded against these assets.
The Company adopted certain provisions of theASC 740,effective January 1, 2007 which clarifies the accounting for uncertainty in income taxes recognized in financial statements and requires the impact of a tax
67
position to be recognized in the financial statements if that position is more likely than not of being sustained by the taxing authority. The adoption ofASC740-10did not have any effect on the Company’s financial position or results of operations.
The Company files income tax returns in the U.S. federal and Massachusetts jurisdictions. The Company is subject to tax examinations throughfor the 20092007 tax year.year and beyond. The Company does not believe there will be any material changes in its unrecognized tax positions over the next 12 months. The Company has not incurred any interest or penalties. In the event that the Company is assessed interest or penalties at some point in the future, they will be classified in the financial statements as general and administrative expense.
| 14. | License Agreements |
As part of its business, the Company enters into numerous licensing agreements. These license agreements with third parties often require milestone and royalty payments based on the progress of the asset through development stages. Milestone payments may be required, for example, upon approval of the product for marketing by a regulatory agency. In certain agreements, RXi is required to make royalty payments based upon a percentage of net sales.
74
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The expenditures required under these arrangements may be material individually in the event that the Company develops product candidates covered by the intellectual property licensed under any such arrangement, and in the unlikely event that milestones for multiple products covered by these arrangements were reached in the same period, the aggregate charge to expense could be material to the results of operations. In addition, these arrangements often give RXi the discretion to unilaterally terminate development of the product, which would allow RXi to avoid making the contingent payments; however, RXi is unlikely to cease development if the compound successfully achieves clinical testing objectives
During the year ended December 31, 2007, RXi entered into a license agreement with Cold Spring Harbor Laboratory (“CSHL”) for small hairpin RNA, or “shRNA”, for which the Company paid $50,000 and agreed to make future milestone and royalty payments upon successful development and commercialization of products. The Company also entered into four exclusive license agreements and an invention disclosure agreement with UMMS for which the Company paid cash of $453,000 and issued 462,112 shares of its common stock valued at $2.3 million, or $5.00 per share. For each RNAi product developed in connection with the license granted by CSHL, the possible aggregate milestone payments equal $2,650,000. The invention disclosure agreement has an initial term of three years and provides the option to negotiate licenses to certain RNAi technologies discovered at UMMS.
On August 29, 2007, RXi entered into a license agreement with TriLink Biotechnologies, Inc. for three RNAi chemistry technologies for all therapeutic RNAi applications, for which the Company paid $100,000 and agreed to pay yearly maintenance fees of $30,000, as well as future clinical milestone payments and royalty payments based on sales of therapeutic products developed from the licensed technologies. There was no expense recorded in 2010. The Company expensed $30,000 for each of the years ended December 31, 2009 and 2008, respectively.in 2009.
In October 2007, RXi entered into a license agreement with Dharmacon, Inc. (now part of Thermo Fisher Scientific Inc.), pursuant to which the Company obtained an exclusive license to certain RNAi sequences to a number of target genes for the development of the Company’s rxRNA compounds. Further, the Company has obtained the right to license additional RNAi sequences, under the same terms, disclosed by Thermo Fisher Scientific Inc. in its pending patent applications against target genes and has received an option for exclusivity for other siRNA configurations. As consideration for this license, the Company paid an up-front fee of $150,000 and agreed to pay future clinical milestone payments and royalty payments based on sales of siRNA compositions developed in connection with the licensed technology. No amounts were expensed in 20082009 and 20092010 related to this license.
In November 2007, RXi entered into a license agreement with Life Technologies, Inc., pursuant to which the Company was granted rights under four patents relating to RNA target sequences, RNA chemical modifications, RNA configurationsand/or RNA delivery to cells. As consideration for this license, RXi paid an up-front fee of $250,000 and agreed to pay yearly maintenance fees of the same amount beginning in 2008. Further, the Company is obligated to pay a fee for each additional gene target added to the license as well as a fee on the first and second anniversaries on the date of which consent to add the gene target to the list of those covered by the license was granted. The Company has also been granted, for each gene target, an option to secure pre-clinical rightsand/or the clinical rights, for which RXi would be required to pay additional fees. Further, the Company is required to make future clinical milestone payments and royalty payments based on sales of therapeutic products developed from the licensed technologies. The Company expensed $62,500 and $250,000 for each of the years ended December 31, 20092010 and 20082009 related to this license.
On October 3, 2008, the Company acquired co-exclusive rights to technology for the oral delivery of RNAi therapeutics from UMMS. As consideration for this license, the Company agreed to pay a total license
68
fee of $2,500,000 over a 12 month period, which can be paid in cash, in equity or a combination thereof, provided that a specified amount of the license fee must be made in cash. This Agreement was amended on July 1, 2009, allowing the Company to extend the periods for which certain milestone payments are due to UMMS. Payments made in equity may only be made if, at the time of such payment, the shares of common
75
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
stock issuable upon conversion of the warrant have been registered for resale under the Securities Act of 1933. No warrants have been issued under this agreement through the date of this report. The Company continually assesses the progress of its research and development efforts as it relates to its licensed technology and may terminate with notice to the licensor at any time. Accordingly, the amounts are being expensed, as payments are made. There were no expenses recorded for the year ended December 31, 2010. The Company expensed $250,000 for each of the yearsyear ended December 31, 2009 and 2008 related to this license2009.
In September, 2009, the Company entered into a Patent and Technology Assignment Agreement with Advirna, LLC (“Advirna”), a Colorado limited liability company co-founded by RXi’s Chief Scientific Officer. Pursuant to the terms of the agreement, Advirna assigned to the Company certain patent and technology rights related to chemically modified polynucleotides (the “Rights”) and the Company granted to Advirna a fullypaid-up license to the Rights in a specified field. Under the terms of the agreement, the Company will pay to Advirna an annual maintenance fee beginning on January 1, 2011, certain payments upon the achievement of regulatory milestones and royalty payments on the sales of certain products. In addition, the Company may terminate the agreement upon 90 days’ prior written notice to Advirna and Advirna may terminate upon 90 days’ prior written notice to the Company in the event the Company ceases to use reasonable efforts to research, develop, license or otherwise commercialize the Rights. If the agreement expires in accordance with its terms or is terminated by a party in the absence of a material breach or for cause in the event that the Company fails to pay Advirna certain fees, the Company will assign the Rights back to Advirna. During the yearsyear ended December 31, 2009, and 2008, the Company paid and expensed $5,000 and $75,000 respectively,for the initial maintenance fee under this agreement. There was no expense recorded for the year ended December 31, 2010.
| 15. | Related Party Transactions |
The Company’s current Chief Scientific Officer or CSO, prior to her employment by the Company, was a consultant to RXi from January 2008 until the date of her employment. This consulting contract resulted in payments to the CSO’s consulting firm of approximately $13,400 which was recorded in the year ended December 31, 2008, in consulting fees and $5,000 recorded as license expense as discussed below. As the
69
CSO is not the sole owner of the consulting firm, the approximate dollar value of her interest in this consulting contract is approximately $9,250.
In addition, RXi and the CSO’s 50% owned Company, Advirna, are parties to an option agreement whereby the Company paid $5,000 in 2008 for consideration to be granted the exclusive worldwide rights to license certain technology by paying anand $75,000 for the initial maintenance fee of $75,000 before June 12, 2009.
On February 26, 2007, the Company entered into Scientific Advisory Board Agreements (the “SAB Agreements”), with four of its founders. At the time of the execution of the SAB Agreements, each of the founders were beneficial owners of more than five percent of the Company’s outstanding stock. Pursuant to the SAB Agreements, on May 23, 2007, the Company granted to each of the founders a stock option under the 2007 Plan to purchase 52,832 shares of its common stock. In addition, under the SAB Agreements, the Company will grant each of the founders a stock option under the 2007 Plan to purchase 52,832 shares of its common stock on February 26, 2008, June 5, 2009 and June 4, 2010 with a per share exercise price equal to the closing price of such stock on the public market on the date of grant unless a founder terminates a SAB Agreement without good reason (as defined) or the Company terminates a SAB Agreement with cause (as defined therein) in which case no further option grants will be made to the founder. If the Company’s common stock is not publicly available on the dates specified above, its Board of Directors will grant the stock options to the founders at the first scheduled board meeting after such date and the per share exercise price of the options will be determined in good faith by the Company’s board of directors. All options granted pursuant to the SAB Agreements are fully vested on the date of grant and have a term of ten years. The fair value of stock options granted during 20092010 and 20082009 under the SAB Agreement for each founder is
76
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
approximately $245,000$142,000 and $338,000$245,000 which was estimated using the Black-Scholes option-pricing model as more fully discussed above under significant accounting policies and the stock based compensation footnote. Included in the accompanying financial statements for the years ended December 31, 20092010 and 20082009 is approximately $978,000$566,000 and $1,350,000,$978,000, respectively, of expense related to the granting of these stock options.
Additionally, pursuant to a letter agreement between the Company and each founder dated as of April 30, 2007, the “SAB Letters”, in further consideration of the services to be rendered by the founders under the SAB Agreements, the Company granted additional stock options on May 23, 2007 under the 2007 Plan to each of the founders to purchase 26,416 shares of its common stock. Unless a founder terminates a SAB Agreement without good reason (as defined) or the Company terminates a SAB Agreement with cause (as defined therein), the options granted pursuant to the SAB Letters will fully vest from and after April 29, 2012 and will have a term of ten years from the date of grant. At December 31, 2009,2010, the fair market value of stock options under the SAB Agreement for each founder is approximately $60,000$20,500 which was estimated using the Black-Scholes option-pricing model as more fully discussed above under the summary of significant accounting policies and the stock based compensation footnote. Included in the accompanying financial statements for the years ended December 31, 20092010 and 20082009 is approximately $73,000$38,000 of income and $156,000, respectively,$73,000, of expense, respectively, related to these stock options.
| 16. | Employee Benefit Plan |
RXi sponsors a 401(k) retirement savings plan (the “Plan”). Participation in the Plan is available to full-time employees who meet eligibility requirements. Eligible employees may defer a portion of their salary as defined by Internal Revenue Service regulations. The Company may make matching contributions on behalf of all participants in the 401(k) Plan in an amount determined by the Company’s board of directors. The
70
Company may also make additional discretionary profit sharing contributions in amounts as determined by the board of directors, subject t to statutory limitations. Matching and profit-sharing contributions, if any, are subject to a vesting schedule; all other contributions are at all times fully vested. The Company intends the 401(k) Plan, and the accompanying trust, to qualify under Sections 401(k) and 501 of the Internal Revenue Code so that contributions by employees to the 401(k) Plan, and income earned (if any) on plan contributions, are not taxable to employees until withdrawn from the 401(k) Plan, and so that the Company will be able to deduct its contributions, if any, when made. The trustee under the 401(k) Plan, at the direction of each participant, invests the assets of the 401(k) Plan in any of a number of investment options. To date, the Company has not made any matching contributions.
| 17. | Subsequent Events |
On January 20, 2010, the Company granted 43,541 RSUs with a contingent right to receive one share of Company common stock for each restricted stock unit to certain employees. The RSUs vested on February 9, 2010.
On January 29, 2010,February 1, 2011, the Company granted warrants to purchase 250,000150,000 shares of common stock at an exercise price of $5.66$2.10 per share in exchange for business advisory services to the Company for a period of up to twelve months. The warrants vested as to 71,25037,500 shares upon issuance, and then will vest at a rate of 23,75037,500 shares per monthquarter starting on the 90 day anniversary of issuance. The Company has also agreed to give “piggy back” registration rights with respect to the shares of common stock underlying the warrants in any registration statement filed by the company in connection with an underwritten offering of the common stock.
77
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
On March 22, 2010,4, 2011, the Company closed an underwritten public offering of 6,000,000 units at a price to the public of $1.35 per unit for gross proceeds of $8.1 million. The financing provided approximately $7.3 million to the Company after deducting the underwriting fee and offering expenses. Each unit consists of (i) one share of common stock, (ii) a thirteen-month warrant to purchase 0.50 of a share of common stock at an exercise of $1.70 per share (subject to anti-dilution adjustment) and (iii) a five-year warrant to purchase 0.50 of a share of common stock at an exercise price of $1.87 per share (subject to anti-dilution adjustment). As a result of the offering that occurred on April 15, 2011, the exercise price of these warrants will be reduced to $1.00 per share.
On March 15, 2011, the Company granted 147,040 shares of common stock to certain employees at a price of $1.16 per share, which represented the Company’s closing stock price on that date.
On March 15, 2011, the Company granted 220,729 RSUs with a contingent right to receive one share of Company common stock for each restricted stock unit to certain employees. The RSUs vested on March 23, 2011.
On March 25, 2011, the Company granted a total of 900,000 shares of common stock to the members of the Board of Directors at $1.18 per share, which represented the Company’s closing stock price on that date.
On April 1, 2011, the Company granted 33,558 shares of common stock to Tod Woolf, Ph.D., a member of its Scientific Advisory Board (SAB) at $1.49 per share which represented the Company’s closing stock price on that date.
On April 1, 2011, the Company granted 201,342 shares of common stock to the former President and Chief Executive Officer, Noah Beerman as part of a Severance Agreement. The shares were granted at a price of $1.49 per share which represented the Company’s closing stock price on that day.
On March 31, 2011, the Company, Diamondback Acquisition Corp., a Delaware corporation and a wholly-owned subsidiary of the Company (“Merger Sub”), Apthera, Inc., a Delaware corporation (“Apthera”), and Robert E. Kennedy, in his capacity as representative of Apthera’s stockholders (the “Stockholder Representative”), entered into an Agreement and Plan of Merger (the “Merger Agreement”). Subject to the terms and conditions of the Merger Agreement, Merger Sub will merge with and into Apthera, with Apthera surviving as a placement agencywholly-owned subsidiary of the Company (the “Merger”). Pursuant to the Merger Agreement, the aggregate merger consideration that the Company will pay to Apthera’s stockholders consists of (i) 19.9% of the number of shares of the Company’s common stock issued and outstanding as of the date of the Merger Agreement, or approximately 4.9 million shares of common stock (the “Aggregate Stock Consideration”); and (ii) contingent payments of up to $32 million (the “Contingent Consideration”) based on the achievement of certain development and commercial milestones relating to Apthera’s NeuVax product candidate. The payment of the Contingent Consideration will be subject to and in accordance with the terms of a Contingent Value Rights Agreement to be entered into between the Company and the Stockholder Representative in connection with the Merger (the “CVR Agreement”). Under the CVR Agreement, a form of which is attached to the Merger Agreement as Exhibit A, the Contingent Consideration is payable, at the election of the Company, in either cash or additional shares of common stock; provided, however, that the Company may not issue any shares in satisfaction of any Contingent Consideration unless it has first obtained approval of its stockholders in accordance with Rule 5635(a) of the NASDAQ Listing Rules.
The Merger Agreement provides that the Company and the Stockholder Representative will also enter into an escrow agreement (the “Escrow Agreement”), pursuant to which the Company shall deposit with Rodmana third-party escrow agent certificates representing 10% of the Aggregate Stock Consideration (the “Escrow Shares”). Pursuant to the terms of the Escrow Agreement, the Escrow Shares will be available to compensate the Company and related parties for certain indemnifiable losses as described in the Merger Agreement.
78
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The transaction closed as of April 13, 2011. Under the Merger Agreement, the Company has agreed to file, within 10 days of the closing date of the Merger, a Registration Statement onForm S-3 with the Securities and Exchange Commission, registering the resale of the shares representing the Aggregate Stock Consideration exchanged.
Our Board of Directors continually evaluates our strategic alternatives and recently determined that it was in the best interests of our stockholders to diversify our development programs with additional development candidates at various stages of development. Our acquisition of Apthera followed from this determination to broaden our strategic direction. We believe that acquiring Apthera will enhance our long term prospects by giving us access to a late stage product candidate, NeuVax, which is expected to enter Phase III clinical trials under an FDA-approved Special Protocol Assessment (“SPA”) for the treatment of breast cancer in the first half of 2012 if we are able to satisfy certain FDA information requirements to be released from a clinical hold to commence the trial. Based on our clinical trials, we also believe that NeuVax has the potential to treat other cancers, including prostate, bladder and ovarian cancers. In addition, we believe that reducing the scope of our RNAi activities will enable us to commit more resources to RXI-109, our lead RNAi-product, while maintaining our core RNAi discovery and development capability to advance current collaborations, as well as enable alliances.
The purchase price consideration and allocation of purchase price was as follows:
| (in 000’s) | ||||
Calculation of allocable purchase price(i): | ||||
| Fair value of shares issued at closing including escrowed shares expected to be released | $ | 6,317 | (ii) | |
| Estimated value of earn-out | 3,194 | |||
| Total allocable purchase price | $ | 9,511 | ||
Estimated allocation of purchase price(i): | ||||
| Cash | $ | 20 | ||
| Prepaid expenses and other current assets | 7 | |||
| Equipment and furnishings | 13 | |||
| Goodwill | 2,214 | |||
| In-process research and development | 9,637 | |||
| Accounts payable | (1,299 | ) | ||
| Accrued expenses and other current liabilities | (720 | ) | ||
| Notes payable | (361 | ) | ||
| $ | 9,511 | |||
| (i) | The purchase price has not been finalized and is subject to change upon completion of the valuation of intangible assets | |
| (ii) | The value of the Company’s common stock was based upon a per share value of $1.27, the closing price of the Company’s common stock as of the close of business on April 13, 2011. |
79
RXi PHARMACEUTICALS CORPORATION
NOTES TO FINANCIAL STATEMENTS — (Continued)
The following presents the pro forma net loss and net loss per common share for the years ended December 31, 2010 and 2009 of the Company’s acquisition of Apthera assuming the acquisition occurred as of January 1, 2009:
| Year Ended December 31, | ||||||||
| (unaudited) | ||||||||
| 2010 | 2009 | |||||||
| Net loss | (14,244 | ) | (20,275 | ) | ||||
| Net loss per common share | $(0.62 | ) | $(1.03 | ) | ||||
| Weighted average shares outstanding | 22,287,471 | 19,770,631 | ||||||
On April 14, 2011, all of our directors and certain of our executive officers executed agreements with the Company under which they agreed that none of their outstanding stock options will be exercisable unless and until our stockholders increase the number of authorized shares of common stock to a number that is sufficient to permit the exercise or conversion in full of all then outstanding options of the Company (including their stock options), warrants and other securities of the Company that are convertible into shares of common stock, and at that point all of their options shall thereafter be exercisable in accordance with the terms of their options awards. An aggregate of 3,398,256 outstanding stock options are covered by these agreements.
On April 15, 2011, the holders of outstanding warrants issued on March 4, 2011 to purchase an aggregate of 3,450,000 shares of our common stock agreed to exchange such warrants for warrants exercisable for the same number of shares as those being exchanged, but otherwise on the same terms, including the exercise price, as the exclusive placement agent, relating to a proposed offering bywarrants sold in our April, 15 2011 financing. Such exchanged warrants will not be exercisable unless our stockholders approve an increase in the number of authorized shares of common stock.
On April 15, 2011, the Company announced that it had priced an underwritten public offering of new securities11,950,000 units at a price to potential investors. On March 23, 2010, the Company entered into definitive agreementspublic of $1.00 per unit for the sale and issuance by the Company to certain investorsgross proceeds of 2,700,000 units, with eachapproximately $12 million. Each unit consistingconsists of one share of the Company’s common stock and a warrant to purchase 0.20 of aone share of common stock at a purchase price of $6.00 per unit (the “2010 Offering”). The 2010 Offering closed on March 26, 2010. The Company issued 540,000 warrants with an exercise price of $6.00$1.00 per shareshare. The shares of common stock and warrants are immediately separable and will be issued separately such that no units will be issued. The warrants are exercisable for a period beginning on September 26, 2010 until their expiration on March 26, 2016. The Company raised gross proceedsone year and one day from the date of approximately $16.2 millionissuance, but only if RXi’s stockholders approve an increase in the 2010 Offeringnumber of its authorized shares of common stock, and net cashexpire on the sixth anniversary of the date of issuance. Net proceeds, after deducting the placement agentunderwriting discounts and commissions and other estimated fees and other offering expenses payable by RXi, and assuming the Company, ofwarrants are not exercised, will be approximately $15.2$10.9 million.
7180
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURES. |
None
| ITEM | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
Rule 13a-15(e) under the Securities and Exchange Act of 1934, as amended (the “Exchange Act”), defines the term “disclosure controls and procedures” as those controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission rules and forms and that such information is accumulated and communicated to the company’s management, including its principal executive and principal financial officers, or persons performing similar functions, as appropriate to allow timely decisions regarding required disclosure.
Our Chief Executive Officer and Principal Accounting Officer have evaluated the effectiveness of the design and operation of our disclosure controls and procedures (as defined underRules 13a-15(e) and15d-15(e) promulgated under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this report. Based upon that evaluation, our Chief Executive Officer and Principal Accounting Officer have concluded that our disclosure controls and procedures are effective.
There have been no changes in our internal controls over financial reporting during the fourth quarter of the year ended December 31, 20092010 that have materially affected, or are reasonably likely to materially affect, our internal controls over financial reporting.
Evaluation of Disclosure Controls and Procedure Management’s report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange ActRule 13a-15(f). Under the supervision and with the participation of our management, including our Chief Executive Officer and Principal Accounting Officer, we conducted evaluations of the effectiveness of our internal control over financial reporting based on the framework in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”). Based on our evaluations under the framework in Internal Control-Integrated Framework issued by the COSO, our management concluded that our internal control over financial reporting was effective as of December 31, 2009.2010.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
This annual report does not include an attestation report of the company’s registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by the company’sCompany’s registered public accounting firm pursuant to temporary rules of the Securities and Exchange Commission that permit the company to provide only management’s report in this annual report.
| OTHER INFORMATION |
None
7281
PART III
| ITEM 10. | DIRECTORS AND EXECUTIVE OFFICERS OF THE REGISTRANT |
We will file with the SEC a definitive Proxy Statement, which we refer to herein as the Proxy Statement, not later than 120 days after the close of the fiscal year ended December 31, 2009.2010. The information required by this item is incorporated herein by reference to the information contained in the Proxy Statement.
| ITEM 11. | EXECUTIVE COMPENSATION |
The information required by this item is incorporated herein by reference to the information contained in the Proxy Statement.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS. |
The information required by this item is incorporated herein by reference to the information contained in the Proxy Statement.
| ITEM 13. | CERTAIN RELATIONSHIPS, RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE |
The information required by this item is incorporated herein by reference to the information contained in the Proxy Statement.
| ITEM 14. | PRINCIPAL ACCOUNTANT FEES AND SERVICES |
The information required by this item is incorporated herein by reference to the information contained in the Proxy Statement.
PART IV
| ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
(a) (1) Financial Statements
See Item 8 in Part II of this annual report onForm 10-K, “Financial Statements and Supplementary Data”, for an index to the financial statements filed in this annual report.
(2) Financial Statement Schedules
Certain schedules are omitted because they are not applicable, or not required by smaller reporting companies.
(3) Exhibits
The Exhibits listed in the Exhibit Index immediately preceding the Exhibits are filed as a part of this annual report onForm 10-K.
7382
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
RXi PHARMACEUTICALS CORPORATION
| By: | /s/ |
President, Chief Executive Officer and Director
(Principal Executive Officer)
Dated: March 31, 2010April 15, 2011
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
Signatures | Title | Date | ||||
/s/ | President, Chief Executive Officer and Director (Principal Executive Officer) | |||||
/s/ | Principal Accounting Officer (Principal Financial Officer and Accounting Officer) | |||||
/s/ Sanford J. Hillsberg Sanford J. Hillsberg | Director | |||||
/s/ Richard Chin Richard Chin | Director | |||||
/s/ Stephen S. Galliker Stephen S. Galliker | Director | |||||
/s/ Steven A. Kriegsman Steven A. Kriegsman | Director | |||||
/s/ Rudolph Nisi Rudolph Nisi | Director | |||||
7483
EXHIBIT INDEX
| Exhibit | Exhibit | Exhibit | ||||||
Number | Number | Description | Number | Description | ||||
| 2 | .1 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(1) | 2 | .1 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(1) | |||
| 2 | .2 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(1) | 2 | .2 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(1) | |||
| 2 | .3 | Reimbursement Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(1) | 3 | .1 | Form of Amended and Restated Certificate of Incorporation of RXi Pharmaceuticals Corporation(1) | |||
| 3 | .1 | Form of Amended and Restated Certificate of Incorporation of RXi Pharmaceuticals Corporation(1) | 3 | .2 | Form of Amended and Restated By-laws of RXi Pharmaceuticals Corporation(1) | |||
| 3 | .2 | Form of Amended and Restated By-laws of RXi Pharmaceuticals Corporation(1) | 4 | .1 | Specimen common stock certificate(3) | |||
| 4 | .1 | Specimen common stock certificate(3) | 4 | .2 | Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the other Stockholders and the Scientific Advisory Board Members, dated February 23, 2007(1) | |||
| 4 | .2 | Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the other Stockholders and the Scientific Advisory Board Members, dated February 23, 2007(1) | 4 | .3 | Annex I to form of Subscription Agreement — Registration Rights Terms between RXi Pharmaceuticals Corporation and Stephen Galliker, Mark Ahn and Sanford Hillsberg(1) | |||
| 4 | .3 | Exhibit A to Contribution Agreement — Registration Rights Terms between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(1) | 4 | .4 | Form of Securities Purchase Agreement between RXi Pharmaceuticals Corporation and various investors, dated June 24, 2008(5) | |||
| 4 | .4 | Annex I to form of Subscription Agreement — Registration Rights Terms between RXi Pharmaceuticals Corporation and Stephen Galliker, Mark Ahn and Sanford Hillsberg(1) | 4 | .5 | Amendment to Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the Stockholders and the Scientific Advisory Board Members, dated July 28, 2008(6) | |||
| 4 | .5 | Form of Securities Purchase Agreement between RXi Pharmaceuticals Corporation and various investors, dated June 24, 2008(5) | 4 | .6 | WarrantNo. A-1 in favor of J.P. Turner Partners, dated August 7, 2008(8) | |||
| 4 | .6 | Amendment to Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the Stockholders and the Scientific Advisory Board Members, dated July 28, 2008(7) | 4 | .7 | Form of Common Stock Purchase Warrant dated July 31, 2009(11) | |||
| 4 | .7 | Amendment to Exhibit A to Contribution Agreement — Registration Rights Terms between CytRx Corporation and RXi Pharmaceuticals Corporation, dated July 28, 2008(7) | 4 | .8 | Form of Warrant issued in August 2009(11) | |||
| 4 | .8 | Form of Warrant Certificate issued to certain purchasers of the RXI Pharmaceuticals Corporation’s common stock in August 2009(14) | 4 | .9 | Form of Common Stock Purchase Warrant issued in March 2010(13) | |||
| 10 | .1 | Voting Agreement between CytRx Corporation and the University of Massachusetts Medical School, dated January 10, 2007(1) | 4 | .10 | Form of13-Month Common Stock Purchase Warrant issued in March 2011(15) | |||
| 10 | .2 | Invention Disclosure Agreement between the University of Massachusetts Medical School and RXi Pharmaceuticals Corporation, dated January 10, 2007(2) | 4 | .11 | Form ofFive-Year Common Stock Purchase Warrant issued in March 2011(15) | |||
| 10 | .3 | Placement Agency Agreement between RXi Pharmaceuticals Corporation, Jeffries & Company, Inc., Natixis Bleichroeder Inc., Broadpoint Securities Group, Inc. and Griffin Securities, Inc., dated June 24, 2008(5) | 4 | .12 | Form of Common Stock Purchase Warrant issued in April 2011(17) | |||
| 10 | .4 | RXI Pharmaceuticals Corporation’s Amended 2007 Incentive Plan, dated June 5, 2009(13) | 10 | .1 | Voting Agreement between CytRx Corporation and the University of Massachusetts Medical School, dated January 10, 2007(1) | |||
| 10 | .5 | WarrantNo. A-1 in favor of J.P. Turner Partners, dated August 7, 2008(8) | 10 | .2 | Form of Securities Purchase Agreement between RXi Pharmaceuticals Corporation and various investors, dated July 31, 2009(11) | |||
| 10 | .6 | Standby Equity Distribution Agreement between RXi Pharmaceuticals Corporation and YA Global Master SPV Ltd. dated January 30, 2009(10) | 10 | .3 | Form of Contingent Value Rights Agreement among RXi Pharmaceuticals Corporation, Computershare Trust Company, N.A., Computershare Inc., and Robert E Kennedy, dated April 13, 2011(16) | |||
| 10 | .7 | Registration Rights Agreement between RXi Pharmaceuticals Corporation and YA Global Master SPV Ltd. dated January 30, 2009(10) | 10 | .4 | Exclusive License Agreement (No.: UMMC06-21-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | |||
| 10 | .8 | Form of Securities Purchase Agreement between RXi Pharmaceuticals Corporation and various investors, dated July 31, 2009(14) | 10 | .5 | Exclusive License Agreement (No.: UMMC03-75-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | |||
| 10 | .9 | Form of Common Stock Purchase Warrant dated July 31, 2009 | 10 | .6 | Non-Exclusive License Agreement (No.: UMMC06-08-03) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | |||
| 10 | .10 | Exclusive License Agreement (No.: UMMC06-21-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | 10 | .7 | Non-Exclusive License Agreement, between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number01-36, dated April 15, 2003, as amended February 1, 2004+(2) | |||
| 10 | .11 | Exclusive License Agreement (No.: UMMC03-68-02) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | 10 | .8 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number02-01, dated April 15, 2003, as amended September 10, 2004+(2) | |||
| 10 | .12 | Exclusive License Agreement (No.: UMMC03-75-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | 10 | .9 | Amended and Restated Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-05,00-37,01-31,03-134,93-09 and02-38, dated September 15, 2003, as amended September 17, 2003 and February 1, 2004+(2) | |||
| 10 | .13 | Non-Exclusive License Agreement (No.: UMMC06-08-03) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007+(2) | 10 | .10 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-17, dated April 15, 2003, as amended January 7, 2004 and February 1, 2004+(2) | |||
| 10 | .11 | Employment Agreement between RXi Pharmaceuticals Corporation and Pamela Pavco, dated March 7, 2007*(1) | ||||||
| 10 | .12 | Employment Agreement between RXi Pharmaceuticals Corporation and Anastasia Khvorova, dated October 17, 2008*(9) | ||||||
| 10 | .13 | Employment Agreement between RXi Pharmaceuticals and Mark Ahn, dated March 31, 2011*(16) | ||||||
7584
| Exhibit | ||||
Number | Description | |||
| 10 | .14 | Non-Exclusive License Agreement, between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number01-36, dated April 15, 2003, as amended February 1, 2004+(2) | ||
| 10 | .15 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number02-01, dated April 15, 2003, as amended September 10, 2004+(2) | ||
| 10 | .16 | Amended and Restated Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-05,00-37,01-31,03-134,93-09 and02-38, dated September 15, 2003, as amended September 17, 2003 and February 1, 2004+(2) | ||
| 10 | .17 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-17, dated April 15, 2003, as amended January 7, 2004 and February 1, 2004+(2) | ||
| 10 | .18 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-60, dated April 15, 2003 as amended February 1, 2004+(2) | ||
| 10 | .19 | Co-Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-33, and all amendments thereto, dated May 18, 2006+(2) | ||
| 10 | .20 | License Agreement between CytRx Corporation, Imperial College Innovations Limited and Imperial College of Science and Technology, dated May 19, 2004+(2) | ||
| 10 | .21 | Employment Agreement between RXi Pharmaceuticals Corporation and Pamela Pavco, dated March 7, 2007*(1) | ||
| 10 | .22 | Employment Agreement between RXi Pharmaceuticals Corporation and James Warren, dated May 23, 2007*(1) | ||
| 10 | .23 | Employment Agreement between RXi Pharmaceuticals Corporation and Dmitry Samarsky, dated June 25, 2007*(1) | ||
| 10 | .24 | Employment Agreement between RXi Pharmaceuticals Corporation and Stephen J. DiPalma, dated August 28, 2007*(1) | ||
| 10 | .25 | Employment Agreement between RXi Pharmaceuticals Corporation and Anastasia Khvorova, dated October 17, 2008*(11) | ||
| 10 | .26 | Employment Agreement between RXi Pharmaceuticals Corporation and Konstantinos Andrikopoulos, dated November 10, 2008*(11) | ||
| 10 | .27 | Employment Agreement between RXi Pharmaceuticals Corporation and Noah D. Beerman, dated November 5, 2009*(16) | ||
| 10 | .28 | Separation Agreement and General Release between RXi Pharmaceuticals Corporation and Tod Woolf, dated November 5, 2009*(16) | ||
| 10 | .29 | RXi Pharmaceuticals Corporation’s 2007 Incentive Plan*(1) | ||
| 10 | .30 | Form of Incentive Stock Option*(1) | ||
| 10 | .31 | Form of Non-qualified Stock Option*(2) | ||
| 10 | .32 | Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts, 01605, dated September 25, 2007(3) | ||
| 10 | .33 | Amendment to Lease between Xi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts, 01605, dated January 23, 2009(9) | ||
| 10 | .34 | Amendment to Lease between Xi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts, 01605, dated March 5, 2009(12) | ||
| 10 | .35 | Form of Subscription Agreement between RXi Pharmaceuticals Corporation and each of Mark K. Ahn, Ph.D., Stephen S. Galliker and Sanford J. Hillsberg(3) | ||
| 10 | .36 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Tariq Rana, Ph.D., dated February 26, 2007 and corresponding Letter Agreement, dated April 30, 2007(3) | ||
76
| Exhibit | Exhibit | Exhibit | ||||||
Number | Number | Description | Number | Description | ||||
| 10 | .37 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Gregory Hannon, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | 10 | .14 | RXI Pharmaceuticals Corporation’s Amended and Restated 2007 Incentive Plan*(14) | |||
| 10 | .38 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Michael Czech, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | 10 | .15 | Form of Incentive Stock Option*(1) | |||
| 10 | .39 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Craig C. Mello, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | 10 | .16 | Form of Non-qualified Stock Option*(2) | |||
| 10 | .40 | Letter Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated December 27, 2007(3) | 10 | .17 | Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts 01605, dated September 25, 2007(3) | |||
| 10 | .41 | Patent License Agreement between RXi Pharmaceuticals Corporation and Invitrogen IP Holdings, Inc. dated November 1, 2007(4) | 10 | .18 | Amendment to Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts 01605, dated January 23, 2009(8) | |||
| 10 | .42 | Patent and Technology Assignment Agreement between RXi Pharmaceuticals Corporation and Advirna, LLC dated September 21, 2009+(15) | 10 | .19 | Amendment to Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts 01605, dated March 5, 2009(10) | |||
| 14 | .1 | Code of Conduct(5) | 10 | .20 | Form of Subscription Agreement between RXi Pharmaceuticals Corporation and each of Mark K. Ahn, Ph.D., Stephen S. Galliker and Sanford J. Hillsberg(3) | |||
| 23 | .1 | Consent of BDO Seidman, LLP, Independent Registered Public Accounting Firm(16) | 10 | .21 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Tariq Rana, Ph.D., dated February 26, 2007 and corresponding Letter Agreement, dated April 30, 2007(3) | |||
| 31 | .1 | Sarbanes-Oxley Act Section 302 Certification of Noah D. Beerman(16) | 10 | .22 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Gregory Hannon, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | |||
| 31 | .2 | Sarbanes-Oxley Act Section 302 Certification of Amy Tata(16) | 10 | .23 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Michael Czech, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | |||
| 32 | .1 | Sarbanes-Oxley Act Section 906 Certification of Noah D. Beerman and Amy Tata(16) | 10 | .24 | Scientific Advisory Board Agreement between RXi Pharmaceuticals Corporation and Craig C. Mello, Ph.D., dated February 26, 2007 and corresponding Letter Agreement dated April 30, 2007(3) | |||
| 10 | .25 | Letter Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated December 27, 2007(3) | ||||||
| 10 | .26 | Patent License Agreement between RXi Pharmaceuticals Corporation and Invitrogen IP Holdings, Inc. dated November 1, 2007(4) | ||||||
| 10 | .27 | Patent and Technology Assignment Agreement between RXi Pharmaceuticals Corporation and Advirna, LLC dated September 21, 2009+(12) | ||||||
| 14 | .1 | Code of Conduct(5) | ||||||
| 21 | .1 | Subsidiaries of the Registrant | ||||||
| 23 | .1 | Consent of BDO USA, LLP, Independent Registered Public Accounting Firm | ||||||
| 31 | .1 | Sarbanes-Oxley Act Section 302 Certification of Mark J. Ahn | ||||||
| 31 | .2 | Sarbanes-Oxley Act Section 302 Certification of Caitlin Kontulis | ||||||
| 32 | .1 | Sarbanes-Oxley Act Section 906 Certification of Mark J. Ahn and Caitlin Kontulis | ||||||
| (1) | Previously filed as an Exhibit to the Company’s Registration Statement onForm S-1 filed on October 30, 2007 (FileNo. 333-147009) and incorporated by reference herein | |
| (2) | Previously filed as an Exhibit to Amendment No. 1 to the Company’s Registration Statement onForm S-1 filed on November 19, 2007(FileNo. 333-147009) and incorporated by reference herein. | |
| (3) | Previously filed as an Exhibit to Amendment No. 2 to the Company’s Registration Statement onForm S-1 filed on January 20, 2008 (FileNo. 333-147009) and incorporated by reference herein. | |
| (4) | Previously filed as an Exhibit to Amendment No. 3 to the Company’s Registration Statement onForm S-1 filed on February 1, 2008 (FileNo. 333-147009) and incorporated by reference herein. | |
| (5) | Previously filed as an Exhibit to the Company’sForm 8-K filed on June 26, 2008 (File No.001-33958) and incorporated by reference herein. | |
| (6) | Previously filed as an Exhibit to | |
| Previously filed as an Exhibit to the Company’sForm 10-Q filed on November 14, 2008(File No. 001-33958) and incorporated by reference herein. | ||
| Previously filed as an Exhibit to the Company’sForm 8-K filed on January 23, 2009(File No. 001-33958) and incorporated by reference herein. | ||
| Previously filed as an Exhibit to the Company’sForm 10-K filed on March 18, 2009(File No. 001-33958) and incorporated by reference herein. |
85
| Previously filed as an Exhibit to the Company’sForm 10-Q filed on May 15, 2009(File No. 001-33958) and incorporated by reference herein. | ||
| Previously filed as an Exhibit to the Company’sForm 8-K filed on July 31, 2009 (File No.001-33958) and incorporated by reference herein. |
77
| Previously filed as an Exhibit to the Company’sForm 10-Q filed on November 16, 2009(File No. 001-33958) and incorporated by reference herein. | ||
| (13) | Previously filed as an Exhibit to the Company’s Form 8-K filed on March 23, 2010 (FileNo. 001-33958) and incorporated by reference herein. | |
| (14) | Previously filed as Annex A to the Company’s Proxy Statement on Schedule 14A filed on April 23, 2010 (File No. 001-33958) and incorporated by reference herein. | |
| (15) | Previously filed as an Exhibit to the Company’s Form 8-K filed on March 1, 2011 (File No. 001-33958) and incorporated by reference herein. | |
| (16) | ||
| (17) | Previously filed as an Exhibit to the Company’s Form 8-K filed on April 14, 2011 (File No. 001-33958) and incorporated by reference herein. | |
| * | Indicates a management contract or compensatory plan or arrangement. | |
| + | This exhibit was filed separately with the Commission pursuant to an application for confidential treatment. The confidential portions of the exhibit have been omitted and have been marked by an asterisk. |
7886