
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM ☒ OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 2017 OR ☐ OF THE SECURITIES EXCHANGE ACT OF 1934 For the transition period from___________ to __________ Commission file number 1-37648 OncoCyte Corporation (Exact name of registrant as specified in its charter)10-K☒ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) (Exact
| California | 27-1041563 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
1010 Atlantic Avenue, Suite 102
Alameda, California 94501
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code(510) 775-0515
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Name of exchange on which registered | |
| Common Stock, no par value | NYSE American |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☒ No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant'sregistrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of "large“large accelerated filer," "accelerated” “accelerated filer," "smaller” “smaller reporting company,"” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ | Accelerated filer ☐ |
| Non-accelerated filer ☒ (Do not check if a smaller reporting company) | Smaller reporting company ☐ |
Emerging growth company ☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided to Section 13(a) of the Exchange Act.☒
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act): Yes ☐ No ☒
The approximate aggregate market value of shares of voting common stock held by non-affiliates computed by reference to the price at which shares of common stock were last sold as of June 30, 2017 was $41.4 million. Shares held by each executive officer and director and by each person who beneficially owns more than 5% of the outstanding common stock have been excluded in that such persons may under certain circumstances be deemed to be affiliates. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of March 20, 2018, there were outstanding 31,468,558 shares of common stock, no par value.
DOCUMENTS INCORPORATED BY REFERENCE
None
2
The description or discussion, in this Form 10-K, of any contract or agreement is a summary only and is qualified in all respects by reference to the full text of the applicable contract or agreement.
We are filing this Amendment No. 1 on Form 10-K/A (this “Amendment No. 1”) to amend our Annual Report (“Report”) on Form 10-K contains market datafor the year ended December 31, 2017, originally filed with the Securities and industry forecastsExchange Commission (the “SEC”) on April 2, 2018, to include the information required by Items 10 through 14 of Part III of Form 10-K. We previously omitted this information from our Form 10-K in reliance on General Instruction G(3) to Form 10-K, which permits that were obtainedinformation to be incorporated in the Form 10-K by reference from industry publications, third party marketa definitive proxy statement if the proxy statement is filed no later than 120 days after our fiscal year-end. We are filing this Amendment No. 1 solely to provide the information required in Part III of Form 10-K because our definitive proxy statement containing this information will not be filed until a later date. The reference on the cover of the Form 10-K to the incorporation by reference to portions of our definitive proxy statement into Part III of the Original Form 10-K is hereby deleted.
PART III
Item 10.Directors, Executive Officers, and Corporate Governance
Directors
The names and ages of our directors are:
Ronald Andrews, 58, joined our Board of Directors in April 2018. Mr. Andrews is the founder and principal of the Bethesda Group, a consulting firm that advises companies in the molecular diagnostics and genomics fields. Prior to founding the Bethesda Group in 2015, Mr. Andrews served as President, Genetic Sciences Division of Thermo Fisher Scientific from September 2013 to December 2014, and as President, Medical Sciences Venture for Life Technologies from February 2012 to September 2013 when Life Technologies was acquired by Thermo Fisher. From 2004 to December 2010, Mr. Andrews was the Chief Executive Officer and Vice Chairman of the Board of Clarient, Inc., a cancer diagnostics company, and from December 2010 to February 2012 he served as CEO of GE Molecular Diagnostics after Clarient was acquired by GE Healthcare. Mr. Andrews also held management positions with companies in diagnostics and related medical fields, including Roche Molecular Diagnostics, Immucor, Inc. and Abbott Labs. Mr. Andrews also serves as a director of Orion Health Group Ltd., Oxford ImmunoTec. Mr. Andrews is also a member of the Board of Governors of CancerLinQ LLC, a wholly-owned non-profit subsidiary of the American Society of Clinical Oncology.
Mr. Andrews has over 30 years of experience in the molecular diagnostics and genomics industries, including experience integrating companies acquired in mergers., He also oversaw the transition of Clarient, Inc. into GE Healthcare and established a strategic plan to integrate In Vivo and In Vitro and expand GE’s presence in oncology.
William Annett, 64, joined OncoCyte as Chief Executive Officer in June 2015. Prior to becoming our Chief Executive Officer, Mr. Annett was a Managing Director at Accenture from 2011 to 2014, where he founded, built, and headed Accenture’s West Coast Life Sciences practice with sales, marketing, and delivery responsibilities for the entire territory. His clients included most of the major biotech and pharmaceutical companies in the western United States. At Genentech, from 2003 until 2011, Mr. Annett led the Commercial Strategy group and managed large operational projects with several hundred team members. He also directed the Project Finance function for research and development, which supported all development pipeline products with more than 200 clinical trials. In 2001 Mr. Annett founded and until 2003 served as CEO of Corra Life Sciences, a prenatal diagnostics company, which worked with a consortium of universities to develop blood tests for the major diseases of pregnancy. Mr. Annett also previously served as Chief Executive Officer of BioFX Laboratories, Inc. from 1999 to 2000. Early in his career, Mr. Annett also founded a consumer products company, which he led for six years as Chief Executive Officer. During his tenure, the company became publicly available information. These publications generally statetraded on NASDAQ and was then acquired. Mr. Annett holds an MBA from the Harvard Business School.
Mr. Annett brings to our Board his years of senior management experience overseeing the development and commercialization of products and services in the life sciences, pharmaceutical and diagnostics industries.
| 1 |
Andrew Arno, 58, joined our Board of Directors in June 2015 and has 30 years of experience working with emerging growth companies. He is currently Vice Chairman of “The Special Equities Group” at Chardan Capital Markets, LLC, a privately held investment banking firm, and from June 2013 until July 2015 served as Managing Director of Emerging Growth Equities, an investment bank, and Vice President of Sabr, Inc., a family investment group. He was previously President of LOMUSA Limited, an investment banking firm. From 2009 to 2012, Mr. Arno served as Vice Chairman and Chief Marketing Officer of Unterberg Capital, LLC, an investment advisory firm that he co-founded. He was also Vice Chairman and Head of Equity Capital Markets of Merriman Capital LLC, an investment banking firm, and served on the board of the parent company, Merriman Holdings, Inc. Mr. Arno currently serves on the boards of directors of Smith Micro Software, Inc. and Asterias Biotherapeutics, Inc.
Mr. Arno brings over 30 years’ experience handling a wide range of corporate and financial matters and his background as an investment banker and strategic advisor to emerging growth companies qualifies him to serve on our Board of Directors.
Alfred D. Kingsley, 75, joined the Board of Directors during September 2009 and served as Chairman of the Board from December 2010 until April 2018. Mr. Kingsley is also the Chairman of the Board of Directors of BioTime, Inc. Mr. Kingsley has been general partner of Greenway Partners, L.P., a private investment firm, and President of Greenbelt Corp., a business consulting firm, since 1993. Greenbelt Corp. served as BioTime’s financial advisor from 1998 until June 30, 2009. Mr. Kingsley was Senior Vice-President of Icahn and Company and its affiliated entities for more than 25 years. Mr. Kingsley is also a director of Asterias Biotherapeutics, Inc. Mr. Kingsley holds a BS degree in economics from the Wharton School of the University of Pennsylvania, and a J.D. degree and LLM in taxation from New York University Law School.
Mr. Kingsley’s long career in corporate finance and mergers and acquisitions includes substantial experience in helping companies to improve their management and corporate governance, and to restructure their operations in order to add value for shareholders. As Chairman of the Board of our former parent company BioTime, Mr. Kingsley has been instrumental in structuring BioTime’s equity and debt financings and the business acquisitions that have helped BioTime expand the scope of its business. Mr. Kingsley, along with entities that he controls, is currently one of the largest shareholders of BioTime.
Andrew J. Last, 58, joined the Board of Directors during December 2015.In January of 2018, Dr. Last was appointed as Chief Commercial Officer at Berkeley Lights. Berkeley Lights is enabling cell by cell selection and manipulation to empower development, antibody discovery and engineering for personalized therapies. Previously, he served as Chief Operating Officer of Intrexon Corporation. From 2010 to 2016, Dr. Last was Executive Vice President and Chief Operating Officer of Affymetrix. Before joining Affymetrix, Dr. Last served as Vice President, Global and Strategic Marketing of BD Biosciences and as General Manager of Pharmingen from 2004 to 2010. From 2002 to 2004, Dr. Last held management positions at Applied Biosystems, Inc., including as Vice President and General Manager from 2003-2004 and Vice President of Marking 2002-2003. Earlier in his career, he served in a variety of management positions at other companies, including Incyte Genomics and Monsanto. Dr. Last holds Ph. D. and MS degrees with specialization in Agrochemical Chemicals and Bio-Aeronautics, respectively, from Cranfield University, and a BS degree in Biological Sciences from the University of Leicester in the United Kingdom.
Dr. Last shares with our Board his many years of senior management experience commercializing products internationally in the genomics and life-sciences industries.
Aditya P. Mohanty, 51, joined the Board of Directors during April 2015. Mr. Mohanty was appointed Co-Chief Executive Officer of BioTime, Inc. during October 2015, after serving as BioTime’s Chief Operating Officer since December 2014. Mr. Mohanty is also a member of BioTime’s Board of Directors. Mr. Mohanty previously served in a number of executive positions at Shire plc, including as President/Head Regenerative Medicine from 2013 to 2014, as Senior Vice President, Business and Technical Operations from 2012 to 2013, as Global Franchise Head MPS from 2010 to 2012, and as Vice President of Operations/Product General Manager from 2005 to 2012. Shire plc is a biotechnology company focused on research, development and commercialization of novel biological products for rare diseases. Mr. Mohanty was VP of Manufacturing and Operations at Transkaryotic Therapies, Inc. from 2002 to 2005 when it was acquired by Shire. Before joining Transkaryotic Therapies, Mr. Mohanty held a number of management positions at Baxter Healthcare Corporation, Bioscience Division from 1990 to 2002. Mr. Mohanty also serves as a director of Asterias Biotherapeutics, Inc. Mr. Mohanty received an MBA degree from Saint Mary’s College, an MS in Chemical Engineering from Clarkson University, and a B. Tech in Chemical Engineering from REC Trichy, in India.
| 2 |
Mr. Mohanty brings to our Board his years of experience as an executive in the pharmaceutical industry, with particular emphasis on product development and manufacturing.
Cavan Redmond, 57, joined our Board of Directors in August of 2015 and was appointed Chairman of the Board during April 2018. Since 2014, Mr. Redmond has served as Partner for Zarsy, LLC. Mr. Redmond served as Chief Executive Officer of WebMD from May 2012 until May 2013. From August 2011 until May 2012, Mr. Redmond served as Group President, Animal Health, Consumer Healthcare and Corporate Strategy of Pfizer Inc., a pharmaceutical company. He served as Pfizer’s Group President, Animal Health, Consumer Healthcare, Capsugel and Corporate Strategy from December 2010 until August 2011 and as its Senior Vice President and Group President, Pfizer Diversified Businesses from October 2009 until December 2010. Prior to Pfizer’s acquisition of Wyeth, a pharmaceutical company, Mr. Redmond served as President, Wyeth Consumer Healthcare and Animal Health Business from May 2009 until October 2009. Before that, he held the positions of President, Wyeth Consumer Healthcare from December 2007 until May 2009 and Executive Vice President and General Manager, BioPharma, Wyeth Pharmaceuticals from 2003 until December 2007. Mr. Redmond also serves as a director of BioTime, Inc.
Mr. Redmond brings to our Board his years of experience as a senior executive in the pharmaceutical and veterinary products industries.
Code of Ethics
We have adopted a Code of Business Conduct and Ethics (“Code of Ethics”) that applies to our principal executive officers, our principal financial officer and accounting officer, our other executive officers, and our directors. The purpose of the Code of Ethics is to promote (i) honest and ethical conduct, including the ethical handling of actual or apparent conflicts of interest between personal and professional relationships; (ii) full, fair, accurate, timely, and understandable disclosure in reports and documents that we file with or submit to the SEC and in our other public communications; (iii) compliance with applicable governmental rules and regulations; (iv) prompt internal reporting of violations of the Code of Ethics to an appropriate person or persons identified in the Code of Ethics; and (v) accountability for adherence to the Code of Ethics. A copy of our Code of Ethics has been posted on our internet website and can be found atwww.oncocyte.com. We intend to disclose any future amendments to certain provisions of our Code of Ethics, and any waivers of those provisions granted to our principal executive officers, principal financial officer, principal accounting officer or controller or persons performing similar functions, by posting the information contained thereinon our website within four business days following the date of the amendment or waiver.
Board Leadership Structure
Our leadership structure bifurcates the roles of Chief Executive Officer and Chairman of the Board. In other words, although William Annett is our Chief Executive Officer and is a member of our Board, Cavan Redmond currently serves as Chairman of the Board. As Chairman of the Board Mr. Redmond will play an active role in the structuring and oversight of our financings and the growth of our business. This structure allows our Chief Executive Officer to focus on innovation in our research and development programs and planning for the completion of development and commercialization of our initial cancer diagnostic tests. The Chairman of the Board serves as an active liaison between the Board and our Chief Executive Officer and our other senior management. The Chairman of the Board also interfaces with our other non-management directors with respect to matters such as the members and chairs of Board committees, other corporate governance matters, financing, strategic planning, and potential business acquisitions.
The Board’s Role in Risk Management
The Board has been obtained from sources believed to be reliable. While we believe thatan active role, as a whole, in overseeing management of the risks of our business. The Board regularly reviews information from these publications is reliable, we have not independently verified such information.
| 3 |
Audit Committee
The Board of Directors has an Audit Committee, the members of which are independent in accordance with Section 803(A) of the NYSE American Company Guides and Section 10A-3 under the Exchange Act. The members of the Audit Committee must also meet the independence tests applicable to members of audit committees under the NYSE American Company Guide.
The members of the Audit Committee are Andrew Arno (Chairman), Ronald Andrews, Andrew J. Last, and Cavan Redmond. Mr. Andrews joined the Audit Committee during April 2018. During 2017, Don M. Bailey served on the Audit Committee. The purpose of the Audit Committee is to recommend the engagement of our independent registered public accountants, to review their performance and the plan, scope, and results of the audit, and to review and approve the fees we pay to our independent registered public accountants. The Audit Committee also will review our accounting and financial reporting procedures and controls.
Our Board of Directors has determined that Andrew Arno meets the criteria of an “audit committee financial expert” within the meaning of the SEC’s regulations based on his many years of experience in the investment banking industry, and his audit committee service at another company, including the evaluation of financial statements.
Executive Officers
William Annett, Mitchell Levine, Lyndal K. Hesterberg, and Kristine C. Mechem are our only executive officers. Russell L. Skibsted served as Chief Financial Officer through November 2017.
Mitchell Levinejoined OncoCyte as Chief Financial Officer in November 2017. Prior to joining OncoCyte, Mr. Levine was the Managing Partner of Kirby Cove Capital Advisors, which provides consulting services to international life sciences investment funds regarding collaboration and investment in US-based life sciences companies. Mr. Levine was the founder and from 2002 to 2017 the managing member of Enable Capital Management, LLC, the general publications, surveyspartner of, Enable Growth Partners, L.P. provided growth capital to technology, life sciences, consumer products, and studies conducted by third parties, someenergy companies. Prior to founding Enable, Mr. Levine was a founding member of which may not be publicly available. Such data involvesThe Shemano Group, a numberleading San Francisco-based investment bank that focused on the capital needs of assumptionsgrowth companies. He has also worked at Bear Stearns and limitationsLehman Brothers. Mr. Levine received his BA from the University of California, Davis.
Lyndal K. Hesterberg, Ph.D. was appointed Senior Vice President-Research and contains projectionsDevelopment during November 2016. Dr. Hesterberg began providing consulting services to OncoCyte in 2015 and estimateswas named Vice President of Development in February of 2016. Dr. Hesterberg continues to provide counsel on clinical trial design, product development and corporate strategy as a consultant to medical and biotech companies. Until 2012, Dr. Hesterberg was the Chief Technology Officer of Crescendo Biosciences where he was responsible for clinical trial, laboratory operations, manufacturing and quality systems and helped bring to market Vectra DA. Previously, he was the president and Chief Executive Officer of Barofold, Inc., where he led the company from product conception through its clinical stage, and recruited a senior leadership team that developed a pipeline of proprietary drug candidates. Dr. Hesterberg received his Ph.D. in biochemistry from the University of St. Louis and a Bachelor of Sciences from the University of Illinois.
Kristine C. Mechem, Ph.D. joined OncoCyte Corporation as Vice President of Marketing in August 2015 after serving as a commercialization consultant assisting in the areas of portfolio management, launch planning and forecasting from April 2015 until joining OncoCyte. Dr. Mechem served as a Director of Business Insights and Analytics at Abbott Diabetes Care, from 2011 until 2014, leading a team of analysts and forecasters supporting the business planning process and developing monthly key performance indicators for its senior management. Dr. Mechem brings to OncoCyte extensive experience within the biotechnology and diagnostics industry. Dr. Mechem’s leadership and industry experience includes: marketing, planning and analytic roles. Her experience spans from smaller early stage diagnostic companies such as Corra Life Sciences, to larger biopharma companies like Abbott Laboratories and Genentech. At Corra Life Sciences, a prenatal diagnostics company, she was a co-founder and served as Vice President of Business Development and Marketing from 2006 until 2007. Dr. Mechem provided business development and marketing support working to secure co-development partners and license intellectual property from three prominent universities. At Genentech from 2009 until 2010, Dr. Mechem supported the portfolio management and led the business planning process for a $400 million annual revenue product. Her increasing roles of responsibility included long range planning, commercial opportunity assessments, and target product profile market research. Dr. Mechem holds a Ph.D. from the University of Chicago, where she was a National Institute of Child Health and Human Development (NICHD) fellow. She also completed the Stanford Business School Executive Program in Strategy and Organization.
| 4 |
Other Key Management Team Members
Lyssa Friedman joined OncoCyte as a consultant in April 2014 and was named Vice President of Clinical and Regulatory Affairs in September 2015. Ms. Friedman started her career as a registered nurse specializing in oncology, and has led clinical research operations teams for more than 15 years. Ms. Friedman was a founding team member at Veracyte, Inc. and worked there from 2008 to 2014, where she executed a 49-site, 4000-subject clinical validation study that resulted in the launch of the future performanceAfirma®Gene Expression Classifier. She later oversaw clinical utility studies that contributed to positive coverage decision from the Centers for Medicare and Medicaid Services and major U.S private insurers. Ms. Friedman is an author of about a dozen peer-reviewed publications and is a frequent speaker on diagnostic development, clinical and regulatory affairs, and reimbursement. Ms. Friedman received her masters in Public Administration from the University of San Francisco.
William K. Seltzer, Ph.D. began providing consulting services to OncoCyte in September 2014 and has since been appointed Vice President of Clinical Services. Dr. Seltzer provides consulting services to companies in the diagnostic industry and is a board certified clinical molecular geneticist with 30 years commercial and academic experience in clinical diagnostic services, clinical diagnostic products development and commercialization, quality assurance and regulatory compliance. As an Associate Professor of Pediatrics at Colorado, he founded and directed the University’s DNA Diagnostic Laboratory. Later, as a member of the industriesexecutive management team at Athena Diagnostics, Inc. and as senior Laboratory Director, Dr. Seltzer was responsible for the successful launch and oversight of clinical services for over 100 laboratory diagnostic tests in the fields of neurology, endocrinology and nephrology.
Dr. Seltzer established the clinical diagnostic laboratory at Veracyte, Inc. which we operate that are subjectdeveloped and launched the first molecular cytology test in the country and later served as laboratory director and site director at Counsyl, Inc. a health technology company. He has published more than 50 articles in peer-reviewed scientific journals and is co-inventor on patents for molecular diagnostics and methods. Dr. Seltzer received his Ph.D. in Pharmacology and Nutrition from the University of Southern California and completed a clinical fellowship in molecular genetics at the University of Colorado School of Medicine.
Michael G. Vicari joined OncoCyte in August 2017 as Vice President of Salesand brings 36 years of successful sales and marketing leadership within the healthcare industry, including the launch of several important diagnostic products. Before joining OncoCyte, he was Vice President of Sales and Marketing at Eurofins Scientific where he established two separate commercial organizations for EGL Labs and NTD Labs. Prior to Eurofins, he was Vice President of Sales for Sequenom, Inc., where he built the commercial organization, led the successful launch of the world’s first noninvasive prenatal test MaterniT21. Mr. Vicari was the Vice President of Sales and Marketing for Oncology/HIV at Monogram Biosciences before it was acquired by LabCorp and managed the launch of HerMark for breast cancer. Mike has served in senior commercial leadership roles at biotechs such as Genentech, Corixa Oncology, and MedImmune.
Compliance with Section 16(A) of the Securities Exchange Act of 1934
Section 16(a) of Exchange Act, requires our directors and executive officers and persons who own more than ten percent (10%) of a high degreeregistered class of uncertainty. We caution you notour equity securities to give undue weight to such projections, assumptionsfile with the SEC initial reports of ownership and estimates.
To our knowledge, based solely on our review of the copies of such reports furnished to us, all Section 16(a) filing requirements applicable to our officers, directors, and greater than ten percent beneficial owners were heldcomplied with during the fiscal year ended December 31, 2017, except that a Form 4 was filed late by our parentAndrew Last.
| 5 |
Item 11.Executive Compensation
Director Compensation
Directors and members of committees of the Board of Directors who are salaried employees of OncoCyte or BioTime Inc. ("BioTime"). Beginning on February 17,are entitled to receive compensation as employees but are not compensated for serving as directors or attending meetings of the Board or committees of the Board. All directors are entitled to reimbursements for their out-of-pocket expenses incurred in attending meetings of the Board or committees of the Board.
Non-employee directors, other than the Chairman of the Board of Directors, received an annual fee of $30,000 in cash during 2017, plus $1,000 for each regular or special meeting of the shares heldBoard of Directors attended or $500 for each meeting attended by BioTime account for less than 50% of our total common stock outstanding. Accordingly, effective February 17, 2017, we are a no longer a consolidated subsidiary of BioTime. See Note 1 of our financial statements included elsewhere in this Report.
The annual fee of cash was paid, and the stock as if it had occurred atoptions granted vested and became exercisable, in four equal quarterly installments, provided that the beginningnon-employee director remained a director on the last day of the earliest period presented.
Directors who served on the Audit Committee, the Compensation Committee, the Nominating/Corporate Governance Committee, Science and Technology Committee or the Finance Committee during 2017, received, in addition to other fees payable to them as directors, the following annual fees:
| ● | Audit Committee Chairman: $10,000 in the first quarter and increased to $15,000 effective April 24, 2017. | |
| ● | Audit Committee Member other than Chairman: $7,000 in the first quarter and increased to $7,500 effective April 24, 2017 | |
| ● | Compensation Committee Chairman: $7,500 in the first quarter and increased to $10,000 effective April 24, 2017 | |
| ● | Compensation Committee Member other than Chairman: $5,000 | |
| ● | Nominating/Corporate Governance Committee Chairman: $7,500 in the first quarter and increased to $10,000 effective April 24, 2017 | |
| ● | Nominating/Corporate Governance Committee Member other than Chairman: $5,000 | |
| ● | Science and Technology Committee Chairman: $7,500 in the first quarter and increased to $10,000 effective April 24, 2017 | |
| ● | Science and Technology Committee Member other than Chairman: $5,000 | |
| ● | Finance Committee Chairman: $10,000 effective July 15, 2017 | |
| ● | Finance Committee Member other than Chairman: $5,000 effective July 15, 2017 |
Members of those committees also received $1,000 for each committee meeting attended in person, and $500 for each committee meeting attended by telephone. Per meeting fees were eliminated as of April 24, 2017.
The following table summarizes certain information concerning the compensation paid during the past fiscal year to develop highly accurate, easy to administer, non-invasive molecular diagnostic tests to improve the standard of care for cancer diagnosis to better meet the needs of patients, physicians and payers. Our initial focus will be confirmatory diagnostics, utilizing novel liquid biopsy technology, for use in conjunction with imaging to confirm initial suspicious imaging results such as lung nodules and breast lesions within certain oncology indications. Our lead product is DetermaVu™, which we are developing as a confirmatory diagnostic test for lung cancer.
| Name | Fees Earned Or Paid in Cash | Option Awards(1) | Total | |||||||||
| Andrew Arno | $ | 65,750 | $ | 83,281 | (2) | $ | 149,031 | |||||
| Don M. Bailey | $ | 41,250 | $ | 83,281 | (3) | $ | 124,531 | |||||
| Alfred D. Kingsley | $ | 68,000 | $ | 210,344 | (4) | $ | 278,344 | |||||
| Andrew J. Last | $ | 70,875 | $ | 83,281 | (2) | $ | 154,156 | |||||
| Cavan Redmond | $ | 62,750 | $ | 83,281 | (2) | $ | 146,031 | |||||
 | |
| (2) | Messrs. Arno, Bailey, Last, and Redmond each received 26,520 stock options. We used the following variables to compute the value of the options: stock price and exercise price of $5.10, expected option term of 5.3 years, volatility of 73.42%, and bond equivalent yield discount rate of 1.79%. |
| (3) | Mr. Bailey resigned from the Board of Directors during November 2017. |
| (4) | Mr. Kingsley received 66,300 stock options. We used the following variables to compute the value of the options: stock price and exercise price of $5.10, expected option term of 5.3 years, volatility of 73.42%, and bond equivalent yield discount rate of 1.79%. |
Emerging Growth Company
We are an "emerging“emerging growth company"company” as defined in the Jumpstart Our Business Startups Act of 2012. We will remain an "emerging growth company" until the earliest of (i) the last day of the fiscal year in which we have total annual gross revenues of $1.0 billion or more; (ii) the last day of our fiscal year following the fifth anniversary of the first sale of our common equity securities pursuant to an effective registration statement under the Securities Act of 1933, as amended (the "Securities Act"); (iii) the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the Securities and Exchange Commission, or the SEC. We refer to the Jumpstart Our Business Startups Act of 2012 herein as the "JOBS Act," and references herein to "emerging growth company" shall have the meaning associated with it in the JOBS Act.
Compensation Committee Interlocks and Insider Participation in Compensation Decisions
The members of our Compensation Committee are Ronald Andrews, Andrew Arno, Andrew J. Last, and Cavan Redmond, each of whom qualify as “independent” in accordance with Section 803(A) and Section 805(c) of the NYSE American Company Guide. Mr. Andrews, Mr. Arno, Mr. Last and Mr. Redmond are not current or former officers or employees of OncoCyte or BioTime or any of BioTime’s subsidiaries, nor did any of them have any relationship with OncoCyte, BioTime or any of BioTime’s subsidiaries requiring disclosure in this Report under Item 404 of SEC Regulation S-K.
During last fiscal year, none of these exemptions for up to five years or such earlier time that we are no longer an emerging growth company.
 |
 |
 |
 |
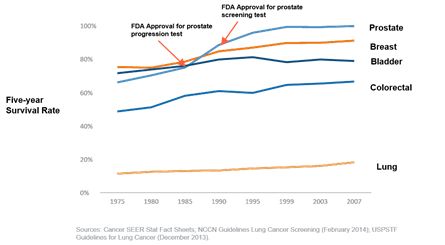 |
Executive Employment Agreements and Change of cancer, has not increased significantly in the last 30 years. The low probability of surviving lung cancer is significantly affected by the late diagnosis – with more than half of all patients diagnosed after the point that the cancer has spread. Poor survival rates for lung cancer was one of the drivers for the development of the USPSTF guidelines. Annual screening with LDCTs is projected to increase the probability of detecting lung cancer in earlier stages such as Stage I where it is treatable and where survival rates could be significantly improved. The number of Stage I patients has been projected to almost double as LDCT becomes part of the high risk patients’ annual check-ups.
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
We have entered into Employment Agreements with our Chief Executive Officer William Annett, our Chief Financial Officer Mitchell Levine, our Senior Vice President of Research and Development Lyndal Hesterberg, and our Vice President of Marketing Kristine C. Mechem.
During March 2017, the Compensation Committee reviewed the base salaries of Mr. Annett and Dr. Mechem. The Compensation Committee determined, and recommended to the Board of Directors, that during 2017 the base salaries for Mr. Annett and Dr. Mechem should increase from $380,000 and $210,000 to $400,000 and $228,000, respectively, and the Board of Directors approved those salary increases. The Board of Directors, in consultation with the Compensation Committee, awarded limited bonuses to our executive officers for 2017, as shown in the Summary Compensation Table.
Pursuant to Mr. Annett’s Employment Agreement, he is eligible to receive annual cash incentive bonus awards determined by the Board of Directors, with a License Agreement with Wistar that entitles ustarget bonus of not less than 35% of his base salary, based on his achievement of specific, objectively determinable, performance goals at target levels for the year. If the specified performance goals are achieved at maximum levels, the amount of the annual bonus will be up to use certain patents, know-how and data belonging150% of Mr. Annett’s base salary as determined by the Board of Directors in its sole discretion.
Mr. Annett’s employment agreement contains provisions entitling him to Wistar.
“Change of Control” means (A) the acquisition of our voting securities by a person or an Affiliated Group entitling the holder to elect a majority of our directors; provided, that an increase in the amount of cumulative net sales. The amountvoting securities held by a person or Affiliated Group who on the date of royalties payable to Wistar will be reducedthe Employment Agreement beneficially owned (as defined in Section 13(d) of the Exchange Act, and the regulations thereunder) more than 10% of our voting securities shall not constitute a Change of Control; and provided, further, that an acquisition of voting securities by the amountone or more persons acting as an underwriter in connection with a sale or distribution of any royalties that we must pay to any third parties onvoting securities shall not constitute a Change of Control, (B) the sale of all or substantially all of our assets; or (C) a merger or consolidation in which we merge or consolidate into another corporation or entity in which our shareholders immediately before the licensed products, butmerger or consolidation do not own, in the aggregate, voting securities of the surviving corporation or entity (or the ultimate parent of the surviving corporation or entity) entitling them, in the aggregate (and without regard to whether they constitute an Affiliated Group) to elect a majority of the directors or persons holding similar powers of the surviving corporation or entity (or the ultimate parent of the surviving corporation or entity). A Change of Control shall not be deemed to have occurred if all of the persons acquiring our voting securities or assets, or merging or consolidating with us, are one or more of our direct or indirect subsidiaries or parent corporations. “Affiliated Group” means (A) a person and one or more other persons in control of, controlled by, or under common control with, such person; and (B) two or more persons who, by written agreement among them, act in concert to acquire voting securities entitling them to elect a majority of our directors. “Person” includes both people and entities.
| 8 |
Mr. Annett’s Employment Agreement also provides that if any of the payments to him would constitute “parachute payments” under applicable provisions of the Internal Revenue Code and would be subject to a maximum reduction of 50%. Our obligationexcise tax, we will use our best efforts to pay royalties to Wistar will terminate on a licensed product by-licensed product and country-by-country basis until the later of (i) the date a valid claim of a licensed patent covering the licensed product no longer exists, or (ii) the tenth (10th) anniversaryobtain shareholder approval of the first commercial salepayment, or Mr. Annett may elect to accept a reduced amount that would not be subject to the excise tax on parachute payments.
Pursuant to Mr. Levine’s Employment Agreement entered into on November 15, 2017, he is eligible to receive annual cash incentive bonus awards determined by the Board of Directors, with a target bonus of not less than 40% of his base salary, based on his achievement of specific, objectively determinable, performance goals at target levels for the licensed productyear.
Mr. Levine’s employment agreement contains provisions entitling him to severance benefits under certain circumstances. If we terminate Mr. Levine’s employment on or after August 1, 2018 without “cause” he will be entitled to six months base salary. If we terminate Mr. Levine on or after August 1, 2018 following a “Change of Control,” he will be entitled to twelve months base salary. The severance compensation may be paid in each country.a lump sum or, at our election, in installments consistent with the payment of his salary while employed by us. In order to receive the severance benefits, Mr. Levine must execute a general release of all claims against us and must return all our property in his possession.
Pursuant to his Employment Agreement, Dr. Hesterberg received a base annual salary of $210,750 for 2017. He is eligible to receive annual cash incentive bonus awards determined by the Board of Directors, with a target bonus of 35% of his base salary, based on his performance and achievement of goals or milestones set by the Board of Directors.
Pursuant to her Employment Agreement, Dr. Mechem is eligible to receive annual cash incentive bonus awards determined by the Board of Directors, with a target bonus of not less than 30% of her base salary, based on her achievement of objectively determinable performance goals for the year
If we terminate the employment of Dr. Hesterberg or Dr. Mechem without “cause” as defined in their respective Employment Agreements, the terminated employee will be entitled to receive, as a severance benefit, payment of three months base salary if he or she has been employed by us for one year or less, or six months base salary, if he or she has been employed by us for more than one year. The severance compensation may be paid in a lump sum or, at our election, in installments consistent with the payment of his or her salary while employed by us. In order to receive the severance benefits, the terminated employee must execute a general release of all claims against us and must return all our property in his or her possession. If Dr. Hesterberg’s employment is terminated by OncoCyte without “cause” or if he resigns for “good reason” within twelve months following a “Change of Control,” he will be entitled to twelve months base salary and accelerated vesting of 100% of any then unvested stock options as may have been granted to him by us.
| 9 |
The following tables show certain information relating to the compensation of our Chief Executive Officer and the three highest paid individuals who were serving as executive officers at year whichend and in each case whose total compensation exceeded $100,000 during 2017. We refer to such executive officers referred to as our “Named Executive Officers
SUMMARY COMPENSATION TABLE
| Name and principal position | Year | Salary | Bonus | Option Awards(1) | All other Compensation (2) | Total | ||||||||||||||||||
| William Annett | 2017 | $ | 397,308 | $ | 40,000 | $ | 740,276 | (3) | $ | 13,500 | $ | 1,191,084 | ||||||||||||
| President and Chief Executive Officer | 2016 | $ | 380,000 | $ | 171,000 | $ | 481,518 | (4) | $ | 12,108 | $ | 1,044,626 | ||||||||||||
| 2015 | $ | 173,333 | $ | 93,333 | $ | 852,206 | (5) | $ | 21,262 | (6) | $ | 1,140,134 | ||||||||||||
| Mitchell Levine(7) | 2017 | $ | 41,885 | $ | 8,684 | $ | 578,991 | (7) | $ | — | $ | 629,560 | ||||||||||||
| Chief Financial Officer | ||||||||||||||||||||||||
| Kristine C. Mechem | 2017 | $ | 225,577 | $ | 27,132 | $ | 222,083 | (8) | $ | 13,490 | $ | 448,282 | ||||||||||||
| VP of Marketing | 2016 | $ | 210,000 | $ | 66,150 | $ | 115,565 | (9) | $ | 11,750 | $ | 403,465 | ||||||||||||
| 2015 | $ | 83,333 | $ | 25,000 | $ | 201,633 | (10) | $ | 3,333 | $ | 310,299 | |||||||||||||
| Lyndal K. Hesterberg(11) | 2017 | $ | 210,750 | $ | 33,720 | $ | 47,378 | (12) | $ | 7,521 | $ | 299,369 | ||||||||||||
| Senior VP of Research and Development | 2016 | $ | 35,125 | $ | 12,625 | $ | 418,873 | (13) | $ | 208,837 | (14) | $ | 671,460 | |||||||||||
| (1) | Option awards were granted under the Option Plan and are valued at the aggregate grant date fair value, as if all options were fully vested and exercisable at the date of grant. Except as otherwise indicated below, one quarter of the options will vest upon completion of 12 full months of continuous employment measured from the grant date, and the balance of the options shall vest in 36 equal monthly installments commencing on the first anniversary of the date of grant, based upon the completion of each month of continuous employment. Option awards are accounted for in accordance with FASB Accounting Standards Codification (ASC) Topic 718. We used the Black-Sholes-Merton Pricing Model to compute option fair values based on applicable exercise and stock prices, an expected option term of six to nine years, volatility ranging from 55.12% to 76.58%, and risk-free interest rate ranging from 1.18% to 2.12%, depending on the date of the grant and based upon observed interest rates on United States government securities appropriate for the expected term. |
| (2) | Except as otherwise indicated below, other compensation consists entirely of employer contributions to employee accounts under BioTime’s 401(k) plan in which employees of BioTime subsidiaries, including OncoCyte, are entitled to participate. |
| (3) | Mr. Annett was granted 225,000 stock options exercisable at an exercise price of $4.85 per share. |
| (4) | Mr. Annett was granted 250,000 stock options exercisable at an exercise price of $3.06 per share. |
| (5) | Mr. Annett was appointed as our Chief Executive Officer on June 16, 2015. Mr. Annett was granted 605,000 stock options exercisable at an exercise price of $2.20 per share of which 5,000 options vested on the date of grant. |
| (6) | Other compensation also includes 10,000 stock options, with a fair market value of $13,262, granted as partial compensation for serving on the Board of Directors of which 5,000 options were forfeited before Mr. Annett was appointed President and Chief Executive Officer. |
| (7) | Mr. Levine was appointed as our Chief Financial Officer on November 15, 2017. Mr. Levine was granted 200,000 stock options exercisable at an exercise price of $5.90 per share. |
| (8) | Dr. Mechem was granted 67,500 stock options exercisable at an exercise price of $4.70 per share |
| (9) | Dr. Mechem was granted 60,000 stock options exercisable at an exercise price of $3.06 per share. |
| (10) | Dr. Mechem was granted 100,000 stock options exercisable at an exercise price of $3.16 per share. |
| (11) | Dr. Hesterberg was appointed Senior Vice President-Research and Development during November 2016. Dr. Hesterberg began providing consulting services to OncoCyte in 2015 and was named Vice President of Development in February of 2016. |
| (12) | Dr. Hesterberg was granted 14,400 stock options exercisable at an exercise price of $4.70 per share |
| (13) | In February 2016, Dr. Hesterberg in the capacity of a consultant received 25,000 stock options exercisable at an exercise price of $3.06 per share. In November 2016, Dr. Hesterberg received 125,000 stock options exercisable at an exercise price of $4.05 per share as part of his employment |
| (14) | Other compensation entirely represents cash compensation paid to Dr. Hesterberg for consulting services prior to the date he became an OncoCyte employee. |
| 10 |
Stock Options Outstanding at Year End
The following table summarizes certain information concerning stock options granted by us under the Option Plan, and held as of December 31, 2017 by our Named Executive Officers:
OUTSTANDING EQUITY AWARDS AT YEAR-END
| Option Awards | ||||||||||||||
| Name | Number of Securities Underlying Unexercised Options Exercisable | Number of Securities Underlying Unexercised Options Unexercisable(1) | Option Exercise Price | Option Expiration | ||||||||||
| William Annett | 5,000 | — | $ | 2.20 | January 8, 2025 | |||||||||
| 5,000 | — | $ | 2.20 | June 15, 2025 | ||||||||||
| 375,000 | 225,000 | (2) | $ | 2.20 | June 15, 2025 | |||||||||
| 114,583 | 135,417 | (3) | $ | 3.06 | February 15, 2026 | |||||||||
| — | 225,000 | (4) | $ | 4.70 | February 16, 2027 | |||||||||
| Mitchell Levine | — | 200,000 | (5) | $ | 5.90 | November 15, 2027 | ||||||||
| Kristine C. Mechem | 58,333 | 41,667 | (6) | $ | 3.16 | August 3, 2025 | ||||||||
| 27,500 | 32,5000 | (3) | $ | 3.06 | February 15, 2026 | |||||||||
| — | 67,500 | (4) | $ | 4.70 | February 16, 2027 | |||||||||
| Lyndal K. Hesterberg | 6,250 | 18,750 | (7) | $ | 3.06 | February 15, 2026 | ||||||||
| 33,854 | 91,146 | (8) | $ | 4.05 | October 31, 2026 | |||||||||
| — | 14,400 | (4) | $ | 4.70 | February 16, 2027 | |||||||||
| (1) | Except as otherwise indicated below, one quarter of the options shall vest upon completion of 12 full months of continuous employment measured from the date of grant, and the balance of the options will vest in 36 equal monthly installments commencing on the first anniversary of the date of grant, based upon the completion of each month of continuous employment. |
| (2) | The date of grant was June 15, 2015. |
| (3) | The date of grant was February 16, 2016. |
| (4) | The date of grant was February 17, 2017. |
| (5) | These options were granted to Mr. Levine upon his appointment as Chief Financial Officer on November 16, 2017. |
| (6) | The date of grant was August 4, 2015. |
| (7) | These options were granted to Dr. Hesterberg in the capacity of a consultant on February 16, 2016. One quarter of the options shall vest upon completion of 12 full months of continuous service provided to the company measured from the date of grant, the second and third quarterly installment vest upon achievement of certain milestones, and the last one quarter shall vest on the second anniversary from the option grant date. |
| (8) | The date of grant was November 1, 2016 at which time Dr. Hesterberg became an employee of OncoCyte. |
| 11 |
Stock Option Plan
We have adopted the Option Plan pursuant to which we may grant options to purchase, or we may sell as restricted stock, up to a total of 5,200,000 shares of common stock. The following summary of the Option Plan is qualified in all respects by reference to the full text of the Option Plan, a copy of which has been filed as an exhibit to our registration statement on Form 10.
Administration of the Option Plan
The Option Plan is administered by the Compensation Committee, which recommends to the Board of Directors which of our officers, directors, employees, consultants, and independent contractors should be credited against total royalties dueawarded options or restricted stock, the number of shares subject to the options granted or shares of restricted stock to be sold, the exercise price of the options or purchase price of restricted stock, and certain other terms and conditions of the options and restricted stock.
No options may be granted under the Option Plan more than ten years after the date the Option Plan was adopted by the Board of Directors, and no options granted under the Option Plan may be exercised after the expiration of ten years from the date of grant.
Terms of the Options
Options granted under the Option Plan may be either “incentive stock options” within the meaning of Section 422(b) of the Internal Revenue Code of 1986, as amended (the “Code”), or non-qualified stock options. Incentive stock options may be granted only to our employees and employees any parent or subsidiary company. The exercise price of incentive stock options granted under the Option Plan must be equal to not less than 85% of the fair market of our common stock on net salesthe date the option is granted. In the case of licensed products duringan optionee who, at the year in whichtime of grant, owns more than 10% of the minimum royalty is paid. We will also be obligated to pay Wistar an annual license maintenance fee incombined voting power of all classes of our capital stock, the mid-five figures.
The options’ exercise price may be payable in cash or in shares of common stock having a fair market value equal to the exercise price, or in a combination of cash and common stock.
Options granted under the Option Plan are nontransferable (except by will pay Wistar (a) milestone paymentsor the laws of descent and distribution) and may vest upon the occurrencesatisfaction of certain milestone events in the development and commercialization of a licensed product, and (b) all past or ongoing costs incurred or to be incurred by Wistar, including government fees and attorneys' fees, in the course of prosecuting the licensed patents.
 |
 |
 |
Certain Adjustments to Number of an expenditure or use of financial or other resources by us, BioTime as our parent company,Shares and the BioTime subsidiaries among which the allocations are made.
The need to obtain regulatory approval to market a new diagnostic test means that:
| 12 |
Corporate Reorganization or Liquidation
In the event of the membersdissolution or liquidation of OncoCyte, or in the event of a reorganization, merger, or consolidation of OncoCyte as a result of which OncoCyte common stock is changed into or exchanged for cash or property or securities not issued by us, or upon a sale of substantially all of our Boardproperty to, or the acquisition of Directors and to influence our management. Twostock representing more than eighty percent 80% of the seven members of our Board of Directors are also officers or directors of BioTime and one of our directors is our Chief Executive Officer. This commonality of directors means that representatives of BioTime and our management are participating in making business decisions on our behalf.
Restricted Stock Sales
In order to facilitate the development, manufacture and commercializationlieu of our diagnostic testsgranting options, we may enter into strategic alliancesrestricted stock purchase agreements with diagnostic, pharmaceutical,employees under which they may purchase common stock subject to certain vesting and repurchase restrictions. We have the right to repurchase unvested shares at the shareholder’s cost upon the occurrence of specified events, such as termination of employment. The price at which shares may be sold under restricted stock purchase agreements will be not less than 85% of fair market value, or medical device companies100% of fair market value in the case of stock sold to advancea person who owns capital stock representing more than 10% of the combined voting power of all classes of our programs and enable us to maintain our financial and operational capacity. We will face significant competition in seeking appropriate alliances.capital stock. We may permit employees or consultants, but not be able to negotiate alliances on acceptable terms, if at all. If we fail to create and maintain suitable alliances, we may have to limit the sizeexecutive officers or scope of, or delay, one or more of our product development or research programs, or we will have to increase our expenditures and will need to obtain additional funding, which may be unavailable or available only on unfavorable terms.
Other Compensation Plans
We do not have any control over these analysts. Four securities analysts cover our sharespension plans, defined benefit plans, or non-qualified deferred compensation plans. We do make contributions to 401(k) plans for participating executive officers and they could issue reportsother employees.
Risk Considerations and Recoupment Policies
The Compensation Committee considers, in establishing and reviewing the executive compensation program, whether the program encourages unnecessary or recommendationsexcessive risk taking. Our executive compensation arrangements include a fixed salary that are unfavorableprovides a steady income so that executives do not feel pressured to focus exclusively on stock price performance or short term financial targets to the pricedetriment of our shares,long-term operational and they could downgrade a previously favorable report or recommendation, and in either case our share price could declinestrategic objectives. We supplement fixed salaries with discretionary bonus awards based on the executive’s performance as a resultwell as the performance of OncoCyte. Most of the report. If onestock options that we have granted to our executive officers under our Option Plan vest over four years, assuring that the executives take a long-term perspective in viewing their equity ownership.
Because we have not adopted compensation plans, or moremade incentive awards, based on quantified financial performance measures, we have not adopted specific policies regarding the adjustment or recovery of these analysts ceases to cover our sharesawards or fails to publish regular reports on our business, we could lose visibilitypayments if the relevant performance measures are restated or otherwise adjusted in a manner that would reduce the financial markets, which could cause our share pricesize of an award or trading volume to decline.
| 13 |
Item 12.Security Ownership of licenses to technology or rights to acquire diagnostic tests in connection with future business acquisitions, or for other business purposes. Certain Beneficial Owners and Management, and Related Stockholder Matters
Security Ownership of Certain Beneficial Owners
The future issuancefollowing table sets forth information as of any such additional common stock or other securities may create downward pressure on the trading price of our common stock.
| Shareholder | Number of Shares | Percent of Total | ||||||
| BioTime, Inc.(1) 1010 Atlantic Avenue, Suite 102 Alameda, California 94501 | 14,674,244 | 38.80 | % | |||||
| Broadwood Partners, L.P.(2) 724 Fifth Avenue, 9th Floor New York, New York 10019 | 8,624,041 | 22.18 | % | |||||
| George Karfunkel 126 East 56th Street/15th Floor New York, New York 10022 | 3,240,812 | 8.57 | % | |||||
| GKarfunkel Family LLC(3) 126 East 56th Street/15th Floor New York, New York 10022 | 3,000,000 | 7.73 | % | |||||
| Bernard Karfunkel 126 East 56th Street/15th Floor New York, New York 10022 | 2,551,666 | 6.75 | % | |||||
| (1) | Excludes 192,644 shares held by BioTime’s former subsidiary Asterias Biotherapeutics, Inc. |
| (2) | Includes 1,055,961 shares that may be acquired upon the exercise of certain warrants. |
| (3) | Includes 1,000,000 shares that may be acquired upon the exercise of certain warrants. |
| 14 |
Security Ownership of Management
The following table sets forth information as of April 9, 2018 concerning beneficial ownership of our common stock less attractive to investors.by each member of the Board of Directors, all Named Executive Officers, and all executive officers and directors as a group.
| Number of Shares | Percent of Total | |||||||
| William Annett(1) | 658,437 | 1.71 | % | |||||
| Mitchell Levine(2) | — | * | ||||||
| Kristine C. Mechem(3) | 123,593 | * | ||||||
| Lyndal K. Hesterberg(4) | 61,979 | * | ||||||
| Alfred D. Kingsley(5) | 690,739 | 1.81 | % | |||||
| Ronald Andrews(6) | — | * | ||||||
| Andrew Arno(7) | 49,890 | * | ||||||
| Andrew J. Last(7) | 49,890 | * | ||||||
| Cavan Redmond(7) | 49,890 | * | ||||||
| Aditya Mohanty | — | * | ||||||
| All executive officers and directors as a group (10 persons)(8) | 1,684,418 | 4.31 | % | |||||
*Less than 1%
| (1) | Includes 613,645 shares that may be acquired through the exercise of stock options that are presently exercisable and 44,792 shares that may be acquired upon the exercise of certain stock options that may become exercisable within 60 days. Excludes 426,563 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (2) | Excludes 200,000 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (3) | Includes 114,114 shares that may be acquired through the exercise of stock options that are presently exercisable and 9,479 shares that may be acquired upon the exercise of certain stock options that may become exercisable within 60 days. Excludes 103,907 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (4) | Includes 56,770 shares that may be acquired through the exercise of stock options that are presently exercisable and 5,209 shares that may be acquired upon the exercise of certain stock options that may become exercisable within 60 days. Excludes 102,421 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| 15 |
| (5) | Includes 299,725 shares that may be acquired through the exercise of stock options that are presently exercisable. Excludes 16,575 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (6) | Excludes 20,000 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (7) | Includes 49,890 shares that may be acquired through the sale of options that are presently exercisable. Excludes 6,630 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
| (8) | Includes 1,293,404 shares that may be acquired upon the exercise of certain stock options that are presently exercisable or that may become exercisable within 60 days. Excludes 889,356 shares that may be acquired upon the exercise of certain stock options that are not presently exercisable and that will not become exercisable within 60 days. |
Item 13.Certain Relationships and Related Transactions, and Director Independence
Shared Facilities Agreement and Relationship with BioTime
Since inception, BioTime has provided us with the JOBS Act,use of office and laboratory facilities, laboratory and office equipment and supplies, utilities, insurance, and the services of its employees and contractors, for which we may to take advantagehave reimbursed BioTime, either through cash payments, shares of certain exemptions from various reporting requirements that are applicable to other public companies that are not "emerging growth companies" including reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We cannot predict if investors will find our common stock, less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile. or delivering convertible promissory notes.
We may take advantage of these reporting exemptions until we are no longer an "emerging growth company." We will remain an "emerging growth company" until the earliest of (i) the last day of the fiscal year in which we have total annual gross revenues of $1.0 billion or more; (ii) the fifth anniversary of the completion of the first sale of our common equity securities pursuant to an effective registration statement under the Securities Act; (iii) the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
| Quarter Ended | High | Low | ||||||
| March 31, 2016 | $ | 10.11 | $ | 2.62 | ||||
| June 30, 2016 | $ | 6.07 | $ | 3.37 | ||||
| September 30, 2016 | $ | 5.32 | $ | 3.25 | ||||
| December 31, 2016 | $ | 7.70 | $ | 3.95 | ||||
| March 31, 2017 | $ | 7.00 | $ | 4.70 | ||||
| June 30, 2017 | $ | 7.35 | $ | 4.95 | ||||
| September 30, 2017 | $ | 7.55 | $ | 3.60 | ||||
| December 31, 2017 | $ | 7.40 | $ | 3.90 | ||||
| Plan Category | Number of Shares to be Issued upon Exercise of Outstanding Options, Warrants, and Rights | Weighted Average Exercise Price of the Outstanding Options, Warrants, and Rights | Number of Shares Remaining Available for Future Issuance under Equity Compensation Plans | ||||||
| OncoCyte Stock Option Plans Approved by Shareholders | 3,390 | $3.25 | 1,384 |
| 12/31/2015 | 3/31/2016 | 6/30/2016 | 9/30/2016 | 12/31/2016 | 3/31/2017 | 6/30/2017 | 9/30/2017 | 12/31/2017 | |||||||||||||||||||||||||||||
| OncoCyte Corporation | Return % | -26.24 | -22.78 | 41.57 | 39.88 | -15.60 | -12.61 | 45.19 | -38.41 | ||||||||||||||||||||||||||||
| Cum $ | 100.00 | 73.76 | 56.96 | 80.64 | 112.80 | 95.20 | 83.20 | 120.80 | 74.40 | ||||||||||||||||||||||||||||
| AMEX Market Value Index (US Companies) | Return % | -0.87 | 5.86 | 5.81 | 1.27 | 3.92 | 2.18 | 0.10 | 3.48 | ||||||||||||||||||||||||||||
| Cum $ | 100.00 | 99.13 | 104.94 | 111.04 | 112.45 | 116.85 | 119.40 | 119.53 | 123.68 | ||||||||||||||||||||||||||||
| NYSE Arca Biotechnology Index | Return % | -22.37 | 2.31 | 11.50 | -8.70 | 16.03 | 8.32 | 8.96 | 0.53 | ||||||||||||||||||||||||||||
| Cum $ | 100.00 | 77.63 | 79.42 | 88.55 | 80.85 | 93.81 | 101.62 | 110.73 | 111.33 | ||||||||||||||||||||||||||||

| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| OPERATING EXPENSES | ||||||||||||
| Research and development | $ | 7,174 | $ | 5,677 | $ | 4,527 | ||||||
| General and administrative | 9,232 | 4,265 | 3,867 | |||||||||
| Sales and marketing | 2,443 | 1,198 | 324 | |||||||||
| Total operating expenses | 18,849 | 11,140 | 8,718 | |||||||||
| Loss from operations | (18,849 | ) | (11,140 | ) | (8,718 | ) | ||||||
| OTHER EXPENSES, NET | ||||||||||||
| Loss on sale of available-for-sale securities and other expenses, net | (309 | ) | - | - | ||||||||
| Interest expense, net | (217 | ) | (28 | ) | (19 | ) | ||||||
| Other income, net | - | - | 2 | |||||||||
| Total other expenses, net | (526 | ) | (28 | ) | (17 | ) | ||||||
| NET LOSS | $ | (19,375 | ) | $ | (11,168 | ) | $ | (8,735 | ) | |||
| Basic and diluted net loss per share | $ | (0.64 | ) | $ | (0.42 | ) | $ | (0.42 | ) | |||
| Weighted average shares outstanding: basic and diluted | 30,195 | 26,529 | 21,009 | |||||||||
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Balance Sheet Data (in thousands): | ||||||||
| Cash and cash equivalents | $ | 7,600 | $ | 10,174 | ||||
| BioTime shares held as available-for-sale securities, at fair value | 760 | 2,237 | ||||||
| Intangible assets, net | 746 | 988 | ||||||
| Total assets | 10,216 | 14,447 | ||||||
| Total liabilities | 5,813 | 4,585 | ||||||
| Total stockholders' equity | $ | 4,403 | $ | 9,862 | ||||
| Year Ended December 31, | $ Increase | % Increase | ||||||||||||||
| 2017 | 2016 | |||||||||||||||
| Research and development expenses | $ | 7,174 | $ | 5,677 | $ | 1,497 | 26.4 | % | ||||||||
| General and administrative expenses | 9,232 | 4,265 | 4,967 | 116.5 | % | |||||||||||
| Sales and marketing expenses | 2,443 | 1,198 | 1,245 | 103.9 | % | |||||||||||
| Year Ended December 31, | $ Increase | % Increase | ||||||||||||||
| 2016 | 2015 | |||||||||||||||
| Research and development expenses | $ | 5,677 | $ | 4,527 | $ | +1,150 | + 25.4 | % | ||||||||
| General and administrative expenses | 4,265 | 3,867 | +398 | +10.3 | % | |||||||||||
| Sales and marketing expenses | 1,198 | 324 | +874 | +269.8 | % | |||||||||||
Amount (1) | Percent | |||||||||||||||
| Program | 2016 | 2015 | 2016 | 2015 | ||||||||||||
| General | $ | 1,649 | $ | 1,687 | 29.1 | % | 37.3 | % | ||||||||
| Lung Cancer Confirmatory Diagnostic | 2,940 | 763 | 51.8 | % | 16.8 | % | ||||||||||
| Bladder Cancer Confirmatory Diagnostic | 376 | 895 | 6.6 | % | 19.8 | % | ||||||||||
| Breast Cancer Confirmatory Diagnostic | 375 | 1,105 | 6.6 | % | 24.4 | % | ||||||||||
| CLIA Lab | 337 | 77 | 5.9 | % | 1.7 | % | ||||||||||
| Total | $ | 5,677 | $ | 4,527 | 100 | % | 100 | % | ||||||||
BioTime shares held as available-for-sale securities. Changes in working capital amounted to an approximate $686,000 of additional use of cash.
| Principal Payments Due by Period | ||||||||||||||||||||
Contractual Obligations (1) | Total | Less Than 1 Year | 1-3 Years | 4-5 Years | After 5 Years | |||||||||||||||
Shared Facilities Agreement (2) | $ | 2,099 | 2,099 | - | - | - | ||||||||||||||
Capital lease (3) | $ | 690 | 383 | 307 | - | - | ||||||||||||||
Loan payable (4) | $ | 2,097 | 876 | 1,221 | - | - | ||||||||||||||
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS | ||||||||
| Cash and cash equivalents | $ | 7,600 | $ | 10,174 | ||||
| BioTime shares held as available-for-sale securities, at fair value | 760 | 2,237 | ||||||
| Prepaid expenses and other current assets | 168 | 285 | ||||||
| Total current assets | 8,528 | 12,696 | ||||||
| NONCURRENT ASSETS | ||||||||
| Intangible assets, net | 746 | 988 | ||||||
| Equipment and furniture, net | 822 | 688 | ||||||
| Deposits | 120 | 75 | ||||||
| TOTAL ASSETS | $ | 10,216 | $ | 14,447 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| CURRENT LIABILITIES | ||||||||
| Amount due to BioTime and affiliates | $ | 2,099 | $ | 2,854 | ||||
| Accounts payable | 175 | 422 | ||||||
| Accrued expenses and other current liabilities | 1,042 | 797 | ||||||
| Loan payable, current | 800 | - | ||||||
| Capital lease liability, current | 338 | 202 | ||||||
| Total current liabilities | 4,454 | 4,275 | ||||||
| LONG-TERM LIABILITIES | ||||||||
| Loan payable, net of deferred financing costs, noncurrent | 1,070 | - | ||||||
| Capital lease liability, noncurrent | 289 | 310 | ||||||
| TOTAL LIABILITIES | 5,813 | 4,585 | ||||||
| Commitments and contingencies (see Note 9) | ||||||||
| STOCKHOLDERS’ EQUITY | ||||||||
| Preferred stock, no par value, 5,000 shares authorized; none issued and outstanding | - | - | ||||||
| Common stock, no par value, 50,000 shares authorized; 31,452 and 28,737 shares issued and outstanding at December 31, 2017 and 2016, respectively | 59,968 | 45,818 | ||||||
| Accumulated other comprehensive loss on available-for-sale securities | (888 | ) | (654 | ) | ||||
| Accumulated deficit | (54,677 | ) | (35,302 | ) | ||||
| Total stockholders’ equity | 4,403 | 9,862 | ||||||
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | $ | 10,216 | $ | 14,447 | ||||
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| OPERATING EXPENSES | ||||||||||||
| Research and development | $ | 7,174 | $ | 5,677 | $ | 4,527 | ||||||
| General and administrative | 9,232 | 4,265 | 3,867 | |||||||||
| Sales and marketing | 2,443 | 1,198 | 324 | |||||||||
| Total operating expenses | 18,849 | 11,140 | 8,718 | |||||||||
| Loss from operations | (18,849 | ) | (11,140 | ) | (8,718 | ) | ||||||
| OTHER EXPENSES, NET | ||||||||||||
| Loss on sale of available-for-sale securities and other expenses, net | (309 | ) | - | - | ||||||||
| Interest expense, net | (217 | ) | (28 | ) | (19 | ) | ||||||
| Other income, net | - | - | 2 | |||||||||
| Total other expenses, net | (526 | ) | (28 | ) | (17 | ) | ||||||
| NET LOSS | $ | (19,375 | ) | $ | (11,168 | ) | $ | (8,735 | ) | |||
| Basic and diluted net loss per share | $ | (0.64 | ) | $ | (0.42 | ) | $ | (0.42 | ) | |||
| Weighted average shares outstanding: basic and diluted | 30,195 | 26,529 | 21,009 | |||||||||
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| NET LOSS | $ | (19,375 | ) | $ | (11,168 | ) | $ | (8,735 | ) | |||
| Other comprehensive loss, net of tax: | ||||||||||||
| Realized loss on sale of available-for-sale securities | 293 | - | 397 | |||||||||
| Unrealized gain (loss) on available-for-sale securities | (527 | ) | (304 | ) | 75 | |||||||
| COMPREHENSIVE LOSS | $ | (19,609 | ) | $ | (11,472 | ) | $ | (8,263 | ) | |||
| Common Stock | Accumulated Other Comprehensive | Accumulated | Total Shareholders’ | |||||||||||||||||
| Shares | Amount | Loss | Deficit | Equity (Deficit) | ||||||||||||||||
| BALANCE AT DECEMBER 31, 2014 | 18,200 | $ | 15,147 | $ | (822 | ) | $ | (15,399 | ) | $ | (1,074 | ) | ||||||||
| Net loss | - | - | - | (8,735 | ) | (8,735 | ) | |||||||||||||
| Unrealized gain on BioTime shares held as available-for-sale securities | - | - | 75 | - | 75 | |||||||||||||||
| Stock-based compensation | - | 1,815 | - | - | 1,815 | |||||||||||||||
| Common stock issued to BioTime for extinguishment of debt | 1,500 | 3,300 | - | - | 3,300 | |||||||||||||||
| Common stock issued to investors for cash | 1,500 | 3,300 | - | - | 3,300 | |||||||||||||||
| Common stock issued to BioTime upon conversion of BioTime convertible note payable and accrued interest | 1,508 | 3,318 | - | - | 3,318 | |||||||||||||||
| Common stock issued to BioTime for cash | 2,711 | 8,349 | - | - | 8,349 | |||||||||||||||
| Exercise of stock options | 3 | 4 | - | - | 4 | |||||||||||||||
| Fair value of contingently issuable warrant | - | 65 | - | - | 65 | |||||||||||||||
| OncoCyte common stock received as a dividend in kind from BioTime | (31 | ) | - | - | - | - | ||||||||||||||
| Transfer of realized loss into equity from sale of BioTime shares | - | (397 | ) | 397 | - | - | ||||||||||||||
| BALANCE AT DECEMBER 31, 2015 | 25,391 | 34,901 | (350 | ) | (24,134 | ) | 10,417 | |||||||||||||
| Net loss | - | - | - | (11,168 | ) | (11,168 | ) | |||||||||||||
| Unrealized loss on BioTime shares held as available-for-sale securities | - | - | (304 | ) | - | (304 | ) | |||||||||||||
| Stock-based compensation | - | 922 | - | - | 922 | |||||||||||||||
| Proceeds from issuance of common stock and warrants, net of discounts and financing costs | 3,246 | 9,777 | - | - | 9,777 | |||||||||||||||
| Exercise of stock options | 100 | 218 | - | - | 218 | |||||||||||||||
| BALANCE AT DECEMBER 31, 2016 | 28,737 | 45,818 | (654 | ) | (35,302 | ) | 9,862 | |||||||||||||
| Net loss | - | - | - | (19,375 | ) | (19,375 | ) | |||||||||||||
| Unrealized loss on BioTime shares held as available-for-sale securities | - | - | (527 | ) | - | (527 | ) | |||||||||||||
| Stock-based compensation | - | 1,630 | - | - | 1,630 | |||||||||||||||
| Issuance of common stock upon exercise of 2016 warrants | 2,392 | 7,774 | - | - | 7,774 | |||||||||||||||
| Exercise of stock options | 323 | 610 | - | - | 610 | |||||||||||||||
| Issuance of warrants for inducement to exercise 2016 warrants | - | 4,074 | - | - | 4,074 | |||||||||||||||
| Issuance of warrants to Silicon Valley Bank | - | 62 | - | - | 62 | |||||||||||||||
| Transfer of realized loss on sale of BioTime shares | - | - | 293 | - | 293 | |||||||||||||||
| BALANCE AT DECEMBER 31, 2017 | 31,452 | $ | 59,968 | $ | (888 | ) | $ | (54,677 | ) | $ | 4,403 | |||||||||
| Year Ended December 31 | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||||||
| Net loss | $ | (19,375 | ) | $ | (11,168 | ) | $ | (8,735 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation expense | 338 | 145 | 41 | |||||||||
| Amortization of intangible assets | 242 | 242 | 242 | |||||||||
| Stock-based compensation | 1,630 | 922 | 1,815 | |||||||||
| Loss on sale of available-for-sale securities, including selling commissions | 309 | - | - | |||||||||
| Warrants issued to certain shareholders as inducement to exercise of warrants | 4,074 | - | - | |||||||||
| Contingently issuable warrant expense to investors | - | - | 65 | |||||||||
| Amortization of debt issuance costs and interest expense | 83 | - | 18 | |||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Amount due to BioTime and affiliates | (753 | ) | 2,007 | 1,557 | ||||||||
| Prepaid expenses and other current assets | 115 | 101 | (274 | ) | ||||||||
| Accounts payable and accrued liabilities | (48 | ) | 229 | 1,042 | ||||||||
| Net cash used in operating activities | (13,385 | ) | (7,522 | ) | (4,229 | ) | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||||||
| Net proceeds from sale of available-for-sale securities | 934 | - | 815 | |||||||||
| Purchase of equipment | (91 | ) | (106 | ) | (500 | ) | ||||||
| Security deposit | - | (75 | ) | - | ||||||||
| Net cash provided by (used in) investing activities | 843 | (181 | ) | 315 | ||||||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||||||
| Proceeds from exercise of options | 610 | 218 | 4 | |||||||||
| Proceeds from exercise of warrants | 7,774 | - | - | |||||||||
| Proceeds from sale of common stock | - | - | 11,649 | |||||||||
| Proceeds from sale of common stock and warrants | - | 10,550 | - | |||||||||
| Financing costs related to sale of common stock and warrants | - | (773 | ) | - | ||||||||
| Proceeds from issuance of loan payable, net of financing costs | 1,982 | - | - | |||||||||
| Repayment of loan payable | (133 | ) | - | - | ||||||||
| Repayment of capital lease obligation | (265 | ) | (114 | ) | - | |||||||
| Net cash provided by financing activities | 9,968 | 9,881 | 11,653 | |||||||||
| NET INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS | (2,574 | ) | 2,178 | 7,739 | ||||||||
| CASH AND CASH EQUIVALENTS: | ||||||||||||
| At beginning of the year | 10,174 | 7,996 | 257 | |||||||||
| At end of the year | $ | 7,600 | $ | 10,174 | $ | 7,996 | ||||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION | ||||||||||||
| Cash paid for interest | $ | 130 | $ | 29 | $ | - | ||||||
| SUPPLEMENTAL SCHEDULE OF NONCASH FINANCING AND INVESTING ACTIVITIES | ||||||||||||
| Equipment purchased under capital leases | $ | 381 | $ | 626 | $ | - | ||||||
| Debt issuance costs | 196 | - | - | |||||||||
| Common stock issued to BioTime for extinguishment of debt | - | - | 3,300 | |||||||||
| Common stock issued to BioTime upon conversion of convertible note payable and accrued interest | - | - | 3,318 | |||||||||
| Realized loss on sale of BioTime shares | - | - | 397 | |||||||||
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Net loss | $ | (19,375 | ) | $ | (11,168 | ) | $ | (8,735 | ) | |||
| Weighted average common shares outstanding – basic and diluted | 30,195 | 26,529 | 21,009 | |||||||||
| Net loss per common share – basic and diluted | $ | (0.64 | ) | $ | (0.42 | ) | $ | (0.42 | ) | |||
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Stock options | 3,390 | 3,017 | 2,240 | |||||||||
| Warrants | 2,779 | 3,246 | - | |||||||||
| 2017 | 2016 | |||||||
| Accrued bonuses and payroll related expenses | $ | 636 | $ | 549 | ||||
| Other accrued expenses | 406 | 248 | ||||||
| Accrued expenses and other current liabilities | $ | 1,042 | $ | 797 | ||||
| 2017 | 2016 | |||||||
| Intangible assets | $ | 2,419 | $ | 2,419 | ||||
| Accumulated amortization | (1,673 | ) | (1,431 | ) | ||||
| Intangible assets, net | $ | 746 | $ | 988 | ||||
| 2017 | 2016 | |||||||
| Equipment and furniture | $ | 1,479 | $ | 1,007 | ||||
| Accumulated depreciation | (657 | ) | (319 | ) | ||||
| Equipment and furniture, net | $ | 822 | $ | 688 | ||||
The Shared Facilities Agreement will remain in effect from year to year, unless either party gives the other party written notice stating that the Shared Facilities Agreement willthis agreement shall terminate on December 31 of that year, or unless the agreement is otherwise is terminated under another provision of the agreement.
Either party may terminate the Shared Facilities Agreement immediately upon the occurrence of a default by the other party. A default will be deemed to have occurred if a party (i) fails to pay any sum due under the Shared Facilities Agreement, or fails to perform any other obligation under the agreement, and the failure continues for a period of 5 days after written notice from the party seeking to terminate the agreement; (ii) becomes the subject of any order for relief in a proceeding under any Debtor Relief Law; (iii) becomes unable to pay, or admits in writing the party’s inability to pay, its debts as they mature; (iv) makes an assignment for the benefit of creditors; (v) applies for or consents to the appointment of any receiver, trustee, custodian, conservator, liquidator, rehabilitation, or similar officer for the party or for all or any part of the party’s property or assets, or any such officer is appointed for such party or any part of its assets without the party’s consent and such appointment is not dismissed or discharged within 60 calendar days; (vi) institutes or consents to any proceeding under any Debtor Relief Law with respect to the party or all or any part of the party’s property or assets, (vii) becomes subject to any proceeding under any Debtor Relief Law without the consent of the party if such case or proceeding continues undismissed or unstayed for 60 calendar days; or (viii) dissolves or liquidates or takes any action to dissolve or liquidate. As used in the Shared Facilities Agreement, the term Debtor Relief Law means the Bankruptcy Code of the United States of America, as amended, or any other similar debtor relief law affecting the rights of creditors generally.
| 16 |
As of December 31, 2017, and 2016, OncoCyte had $2.1 million and $2.9 million outstanding and payable to BioTime and affiliates included in current liabilities in connection with the costs incurred under the Shared Facilities Agreement. Since these amounts
Cavan Redmond, Alfred D. Kingsley, and Adi Mohanty, who are duemembers of our Board of Directors, are directors of BioTime, and payable in 30 daysMr. Mohanty is the Co-Chief Executive Officer of being invoiced, the payables are classified as current liabilities for all periods presented.
Year Ending December 31, | Loan Payments | ||||
| 2018 | $ | 876 | |||
| 2019 | 835 | ||||
| 2020 | 386 | ||||
| Total payments of principal and interest | 2,097 | ||||
| Less: amounts representing interest | (114 | ) | |||
| Total payments of principal before deferred financing costs | 1,983 | ||||
| Less: deferred financing costs | (113 | ) | |||
| Total loan payable, net of deferred financing costs | $ | 1,870 | |||
Certain Sales of Equity
During August 29, 2016, OncoCytewe sold an aggregate of 3,246,153 immediately separable units,“units,” with each unit consisting of one share of OncoCyteour common stock and one warrant to purchase one share of OncoCyteour common stock, (the “2016 Warrants”), at a price of $3.25 per unit (the “Offering”“Units”). The sales were made pursuant to the termsGeorge Karfunkel and conditionsBroadwood Partners, L.P. (“Broadwood”) who each beneficially own more than 5% of certain Purchase Agreements between OncoCyte and the purchasers in the Offering. The purchasers included certain OncoCyte existing shareholders other than BioTime. At the close of the Offering, BioTime’s percentage ownership of theour outstanding common stock, of OncoCyte declinedpurchased 1,000,000 Units, and purchased 1,538,461 Units. During February 2017, certain investors who purchased Units, including Broadwood, entered into Warrant Exercise Agreements pursuant to 51.2% through which BioTime retained a controlling interest in OncoCyte. OncoCyte received $9.8 million in net proceeds after discounts, commissions and expenses from the Offering. OncoCyte will use the proceeds from the Offering for funding its operations or for working capital or other general corporate purposes.
On July 21, 2017, OncoCyte entered into three forms of Warrant Exercise Agreements (each, an "Exercise Agreement"“Exercise Agreement”) with certain holders of the 2016 Warrants, including Broadwood and George Karfunkel, providing for the cash exercise of their 2016 Warrants and the issuance of new warrants (the "July“July 2017 Warrants"Warrants”) to them.
Pursuant to one form of Exercise Agreement, two investors exercised 2016 Warrants to purchase 226,923 shares of OncoCyte'sOncoCyte’s common stock at the exercise price of $3.25 per share, and OncoCyte issued to them July 2017 Warrants expiring five years from the date of issue, to purchase 226,923 shares of common stock at an exercise price of $5.50 per share.
Pursuant to a second form of Exercise Agreement, one investorBroadwood exercised 2016 Warrants to purchase 540,000 shares of common stock at the exercise price of $3.25 per share, and OncoCyte issued to the investorBroadwood a July 2017 Warrant, expiring five years from the date of issue, to purchase 270,000 shares of common stock at an exercise price of $3.25 per share. In this alternative form of Exercise Agreement, OncoCyte also agreed to use commercially reasonable efforts to file with the SEC a registration statement covering the resale of the shares of common stock issuable upon exercise of the July 2017 Warrant and to keep it continuously effective for up to five years, subject to conditions set forth in the Exercise Agreement.
Pursuant to a third form of Exercise Agreement, one investorGeorge Karfunkel exercised 2016 Warrants to purchase 1,000,000 shares of common stock at the exercise price of $3.25 per share, and OncoCyte issued to the investor (i) a July 2017 Warrant, expiring two years from the date of issue, to purchase 500,000 shares of common stock at an exercise price of $5.50 per share, and (ii) a July 2017 Warrant, expiring two years from the date of issue, to purchase 500,000 shares of common stock at an exercise price of $3.25 per share. In this alternative form of Exercise Agreement, OncoCyte also agreed to use commercially reasonable efforts to file with the SEC a registration statement covering the resale of the shares of common stock issuable upon exercise of the July 2017 Warrant and to keep it continuously effective for up to five years, subject to conditions set forth in the Exercise Agreement.
| 17 |
During March 2018, OncoCyte received gross proceeds of approximately $5.74 millionentered into securities purchase agreements with Broadwood and issued July 2017 WarrantsGeorge Karfunkel pursuant to purchase 1,496,923which Broadwood purchased 3,968,254 shares of common stock, at a weighted average priceand Mr. Karfunkel purchased 2,380,952 shares of $4.34common stock and irrevocably committed to purchase, by April 30, 2018, an additional 1,587,302 shares of common stock, for $1.26 per share.
Approval by the 2016 Warrants. For financial reporting purposes, the issuanceBoard of Directors
All of the July 2017 Warrants is treated as an inducement offer to certain investors to exercise their 2016 Warrants. Accordingly,transactions described above were reviewed directly by the fair valueBoard of Directors, and the Board of Directors or a designated committee of the July 2017 Warrants,Board determined whether to approve or withhold approval of each transaction. The Board of Directors or committee considered such factors as they deemed relevant to the particular transaction and applied such criteria as it determined to be approximately $3.0 million usingappropriate in connection with its evaluation of each proposed transaction on a transaction by transaction basis. The Board of Directors does not have any written guidelines governing the exercise of its discretion other than the Related Person Transaction Policy with respect to transactions with officers, directors, beneficial owners of more than 5% of our outstanding shares of common stock, or any member of their immediate family.
Related Person Transaction PolicyBlack-Scholes option pricing model,
We have adopted a Related Person Transaction Policy that applies to transactions exceeding $120,000 in which any of our officers, directors, beneficial owners of more than 5% of our outstanding shares of common stock, or any member of their immediate family, has a direct or indirect material interest, determined in accordance with the policy (a “Related Person Transaction”). A Related Person Transaction must be reported to our outside legal counsel, our Chief Operating Officer, and our Chief Financial Officer, and will be subject to review and approval by our Nominating/Corporate Governance Committee prior to effectiveness or consummation, to the extent practical. In addition, any Related Person Transaction that is ongoing in nature will be reviewed by the Nominating/Corporate Governance Committee annually to ensure that the transaction has been conducted in accordance with any previous approval and that all required disclosures regarding the transaction are made.
As appropriate for the circumstances, the Nominating/Corporate Governance Committee will review and consider:
| ● | the interest of the officer, director, beneficial owner of more than 5% of our common stock, or any member of their immediate family (“Related Person”) in the Related Person Transaction; | |
| ● | the approximate dollar value of the amount involved in the Related Person Transaction; | |
| ● | the approximate dollar value of the amount of the Related Person’s interest in the transaction without regard to the amount of any profit or loss; | |
| ● | whether the transaction was undertaken in the ordinary course of our business; | |
| ● | whether the transaction with the Related Person is proposed to be, or was, entered into on terms no less favorable to us than terms that could have been reached with an unrelated third party; | |
| ● | the purpose of, and the potential benefits to the transaction to us; and | |
| ● | any other information regarding the Related Person Transaction or the Related Person in the context of the proposed transaction that would be material to investors in light of the circumstances of the particular transaction. |
The Nominating/Corporate Governance Committee will review all relevant information available to it about a Related Person Transaction. The Nominating/Corporate Governance Committee may approve or ratify the Related Person Transaction only if it determines that, under all of the circumstances, the transaction is in, or is not in conflict with, our best interests. The Nominating/Corporate Governance Committee may, in its sole discretion, impose such conditions as it deems appropriate on us or the Related Person in connection with approval of the Related Person Transaction.
| 18 |
was recordedA copy of our Related Person Transaction Policy can be found on our website atwww.oncocyte.com.
Director Independence
Our Board of Directors has determined that Ronald Andrews, Andrew Arno, Andrew Last, and Cavan Redmond qualify as “independent,” and that Don M. Bailey, who served as a noncash charge to shareholder expense includeddirector until November 2107, qualified as “independent,” in generalaccordance with Section 803(A) of the NYSE American Company Guide. The members of our Audit Committee meet the additional independence standards under Section 803(B)(2) of the NYSE American Company Guide and administrative expenses,Section 10A-3 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and a corresponding increase was recorded to equity on July 21, 2017, the issuance date.members of our Compensation Committee meet the additional independence standards under Section 805(c)(1) of the NYSE American Company Guide. Our independent directors received no compensation or remuneration for serving as directors except as disclosed under “CORPORATE GOVERNANCE—Compensation of Directors.” None of these directors, nor any of the members of their families, have participated in any transaction with us that would disqualify them as “independent” directors under the standards described above.
William Annett does not qualify as independent because he is our full-time employee, Aditya Mohanty does not qualify as “independent” because within the past three years he received compensation from our former parent BioTime as an executive officer, and Alfred D. Kingsley does not qualify as “independent” because within the past three years he received compensation for serving as our Executive Chairman.
Item 14.
OUM & Co., LLP (“OUM”) has served as our independent registered public accountants since the fourth quarter of 2015, and audited our annual financial statements for the fiscal years ended December 31, 2017 OncoCyte has anand 2016.
Audit Fees, Audit Related Fees, Tax Fees and Other Fees
The following table sets forth the aggregate of 2,779,221 warrants issued and outstanding at exercise prices ranging from $3.25 and $5.50 per warrant.
| 2017 | 2016 | |||||||
| Audit Fees(1) | $ | 201,228 | $ | 248,435 | ||||
| Audit Related Fees(2) | — | — | ||||||
| Tax Fees(3) | — | — | ||||||
| Total Fees | $ | 201,228 | $ | 248,435 | ||||
| (1) | Audit Fees consist of fees billed for professional services rendered for the audit of OncoCyte’s annual financial statements included in our Registration Statement on Form 10 and in our Annual Report on Form 10-K, and review of the interim financial statements included in our Registration Statement on Form 10 and our Quarterly Reports on Form 10-Q, as applicable, and services that are normally provided by our independent registered public accountants in connection with statutory and regulatory filings or engagements. |
| (2) | Audit-Related Fees consist of fees billed for assurance and related services that are reasonably related to the performance of the audit or review of our consolidated financial statements and are not reported under “Audit Fees.” This category includes fees related to non-routine SEC filings. |
| (3) | Tax Fees were billed for services including assistance with tax compliance and the preparation of tax returns, tax consultation services, assistance in connection with tax audits and tax advice related to mergers, acquisitions and dispositions. |
| 19 |
Pre-Approval of Audit and 99,496 sharesPermissible Non-Audit Services
Our Audit Committee requires pre-approval of common stock were issued upon the exercise of stock options, from which OncoCyte received $610,000all audit and $218,000 in cash proceeds, respectively.
| Options | Shares Available for Grant | Number of Options Outstanding | Weighted Average Exercise Price | |||||||||
| Total at January 1, 2016 | 1,757 | 2,240 | $ | 2.03 | ||||||||
| Options granted | (962 | ) | 962 | 3.58 | ||||||||
| Options exercised | - | (100 | ) | 2.19 | ||||||||
| Options forfeited, cancelled or expired | 85 | (85 | ) | 2.00 | ||||||||
| Total at December 31, 2016 | 880 | 3,017 | 2.52 | |||||||||
| Increase in pool | 1,200 | - | ||||||||||
| Options granted | (896 | ) | 896 | 5.17 | ||||||||
| Options exercised | - | (323 | ) | 1.89 | ||||||||
| Options forfeited, cancelled or expired | 200 | (200 | ) | 3.11 | ||||||||
| Total at December 31, 2017 | 1,384 | 3,390 | $ | 3.25 | ||||||||
| Exercisable at December 31, 2017 | 1,835 | $ | 2.48 | |||||||||
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Research and development | $ | 668 | $ | 312 | $ | 456 | ||||||
| General and administrative | 841 | 610 | 1,359 | |||||||||
| Sales and marketing | 121 | - | - | |||||||||
| Total stock-based compensation expense | $ | 1,630 | $ | 922 | $ | 1,815 | ||||||
| Year Ended December 31, | |||||||||||||||
| 2017 | 2016 | 2015 | |||||||||||||
| Expected life (in years) | 6.15 | 6.21 | 6.83 | ||||||||||||
| Risk-free interest rates | 2.03 | % | 1.46 | % | 1.87 | % | |||||||||
| Volatility | 66.01 | % | 64.64 | % | 74.15 | % | |||||||||
| Dividend yield | - | % | - | % | - | % | |||||||||
| 2017 | 2016 | |||||||
| Deferred liabilities: | ||||||||
| Available-for-sale securities | $ | - | $ | (761 | ) | |||
| Total deferred tax liabilities | - | (761 | ) | |||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | 11,414 | 11,730 | ||||||
| Research and development credit carryforwards | 2,141 | 1,765 | ||||||
| Patents and fixed assets | 268 | 179 | ||||||
| Stock-based compensation and accrued payroll | 1,260 | 1,041 | ||||||
| Valuation Allowance | (15,083 | ) | (13,954 | ) | ||||
| Total deferred tax assets | - | 761 | ||||||
| Net deferred tax asset (liability) | $ | - | $ | - | ||||
| Year Ended December 31, | |||||||||||||||
| 2017 | 2016 | 2015 | |||||||||||||
| Computed tax benefit at federal statutory rate | 34 | % | 34 | % | 34 | % | |||||||||
| Re-rate of federal net deferred tax assets | (35 | )% | 0 | % | 0 | % | |||||||||
| Permanent differences | (8 | )% | (1 | )% | (9 | )% | |||||||||
| State tax benefit | 3 | % | 2 | % | 15 | % | |||||||||
| Research and development credits | 1 | % | 2 | % | 2 | % | |||||||||
| Other | 0 | % | 7 | % | 3 | % | |||||||||
| Adjust basis for available-for-sale-securities | 11 | % | 0 | % | 0 | % | |||||||||
| Change in valuation allowance | (6 | )% | (44 | )% | (45 | )% | |||||||||
| - | % | - | % | - | % | ||||||||||
Year Ending December 31, | Capital Lease Payments | ||||
| 2018 | $ | 383 | |||
| 2019 | 248 | ||||
| 2020 | 59 | ||||
| Total minimum lease payments | 690 | ||||
| Less amounts representing interest | (63 | ) | |||
| Present value of net minimum lease payments | $ | 627 | |||
Item 15.Exhibits, Financial Statement Schedules
(a-1)
Financial Statements.
The following financial statements of OncoCyte Corporation are filed in the Form 10-K:
Balance Sheets
Statements of Operations
Statements of Comprehensive Loss
Statements of Shareholders'Shareholders’ Equity (Deficit)
Statements of Cash Flows
Exhibit Numbers | Exhibit Description | |
| Form of Director/Consultant Option Agreement | ||
| 20 |
| 10.5 | ||
Employment Agreement, dated August 1, 2015, between OncoCyte Corporation and Kristine Mechem(1) | ||
| 21 |
| 32 | ||
| Section 1350 Certification * | ||
| 101 | Interactive Data Files | |
| 101.INS | XBRL Instance | |
| 101.SCH | XBRL Taxonomy Extension |
| 101.CAL | XBRL Taxonomy Extension Calculation | |
| 101.DEF | XBRL Taxonomy Extension Definition | |
| 101.LAB | XBRL Taxonomy Extension Label | |
| 101.PRE | XBRL Taxonomy Extension Presentation |
| (1) | Incorporated by reference to OncoCyte Corporation’s Form 10 12(b) filed with the Securities and Exchange Commission on November 23, 2015. |
| (2) | Incorporated by reference to OncoCyte Corporation’s Form 10 12(b) A-1 filed with the Securities and Exchange Commission on December 29, 2015. |
| (3) | Incorporated by reference to OncoCyte Corporation’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 30, 2016. |
| (4) | Incorporated by reference to OncoCyte Corporation’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 11, 2016. |
| (5) | Incorporated by reference to OncoCyte Corporation’s Current Report on Form 8-K filed with the Securities and Exchange Commission on August 29, 2016. |
| (6) | Incorporated by reference to OncoCyte |
| (7) | Incorporated by reference to OncoCyte Corporation’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on February 27, 2017. |
| (8) | Incorporated by reference to OncoCyte Corporation’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on April 28, 2017. |
| (9) | Incorporated by reference to Registration Statement on Form S-8, File Number 333-219109 filed with the Securities and Exchange Commission on June 30, 2017. |
| (10) | Incorporated by reference to OncoCyte |
| (11) | Previously filed with OncoCyte’s Annual Report on Form 10-K for the year ended December 31, 2017. |
| * | Filed herewith. |
Item 16. Summary
None.
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this reportAmendment No. 1 on Form 10-K10-K/A to be signed on its behalf by the undersigned, thereunto duly authorized on the 230ndth day of April 2018.
| ONCOCYTE CORPORATION | |||
| By: | /s/ William Annett | ||
| William Annett | |||
| President and Chief Executive Officer | |||
| 23 |