2
2
PART I
ITEM 1. BUSINESS.
| ITEM 1. | BUSINESS. |
Microparticles can be used to provide sustained drug delivery, allowing patients to receive injections at less frequent intervals (e.g., monthly instead of daily). Similarly, our patented PhotoLink® technology enhances the maneuverability of dilatation catheters and guidewires within the body by improving the lubricity of the device surface.
These clinical trial results, in part, led to the collaborative research and license agreement with Merck & Co., Inc. that we signed in June 2007. Through this agreement, SurModics and Merck will pursue the development and commercialization of the I-vation™ Sustained Drug Delivery System in combination with triamcinolone acetonide and proprietary Merck compounds. Under the terms of our agreement with Merck, we received an up-front license fee of $20 million and may receive up to an additional $288 million in fees and development milestones associated with the successful product development and attainment of appropriate U.S. and EU regulatory approvals for these new combination products. We will also be paid for our activities in researching and developing these combination products. Additionally, under the terms of our agreement with Merck, we will be responsible for the exclusive manufacture and supply of clinical and commercial products. We will also receive royalties on sales of products developed under our collaboration.
3
product.
Since our acquisition of
3
Brookwood Pharmaceuticals generates revenue from research and development fees, polymer sales, and royalties and license fees.
We managefiscal year ending September 30, 2008, we managed our business through the following seven technology- and market-focused business units:
| • | Drug Delivery,creating and supporting site specific drug delivery polymers and coating technologies for use in drug/device combination products in our chosen markets, such as drug-eluting stents for the treatment of vascular disease, ophthalmic implants, orthopedics, urology, oncology, and wound treatment, among others. | |
| • | Ophthalmology,developing drug delivery systems intended to enhance performance, safety, patient convenience and patient compliance for a variety of drugs and other bioactive agents that are being developed by pharmaceutical and ophthalmology companies for the treatment of serious eye diseases. | |
| • | Hydrophilic Technologies,specializing in advanced lubricity (slippery) coatings that can enhance the function of medical devices, facilitating and easing their placement and maneuverability in the body. | |
| • | Regenerative Technologies,developing platforms intended to augment or replace tissue/organ function (e.g., cell encapsulation applications), or to modify medical devices to facilitate tissue/organ recovery through natural repair mechanisms (e.g., prohealing coatings). |
4
Drug Delivery,creating and supporting site specific drug delivery polymers and coating technologies for use in drug/device combination products in our chosen markets, such as drug-eluting stents for the treatment of vascular disease, ophthalmic implants, orthopedics, urology, oncology, and wound treatment, among others.Ophthalmology,developing drug delivery systems intended to enhance performance, safety, patient convenience and patient compliance for a variety of drugs and other bioactive agents that are being developed by pharmaceutical and ophthalmology companies for the treatment of serious eye diseases.Hydrophilic Technologies,specializing in advanced lubricity (slippery) coatings that can enhance the function of medical devices, facilitating and easing their placement and maneuverability in the body.Regenerative Technologies,developing platforms intended to augment or replace tissue/organ function (e.g., cell encapsulation applications), or to modify medical devices to facilitate tissue/organ recovery through natural repair mechanisms (e.g., prohealing coatings).Orthopedics,developing innovative solutions for the treatment of structural defects in patients using proven SurModics technologies, and creating new technology solutions for existing patient care needs in the orthopedics field.
| • | In Vitro Technologies
| |
| • | Orthopedics,developing innovative solutions for the treatment of structural defects in patients using proven SurModics technologies, and creating new technology solutions for existing patient care needs in the orthopedics field. | |
| • | Brookwood Pharmaceuticals,
formulations. |
We believe SurModics is uniquely positioned to exploit the continuing trend of incorporating surface modification and drug delivery technologies into the design of products such as devices and drugs, potentially leading to more efficient and effective products as well as creating entirely new product applications. We have a growing portfolio of proprietary technologies, market expertise and insight, and unique collaborative research and development capabilities – all key ingredients to bring innovation together for the benefit of patients, the Company, and the healthcare industry.
Coatings for Surface Modification and Drug Delivery
Our proprietary PhotoLink coating technology is a versatile, easily applied, coating technology that modifies medical device surfaces by creating covalent bonds between device surfaces and a variety of chemical agents. PhotoLink coatings can impart many performance enhancing characteristics, such as advanced lubricity (slippery) and hemocompatibility (preventing clot formation), by becoming bound onto surfaces of medical devices or other biological materials without materially changing the dimensions or other physical properties of devices. Our PhotoLink technology utilizes proprietary, light activated (photochemical) reagents, which include advanced polymers or active biomolecules having desired surface characteristics and an attached light reactive chemical compound (photogroup). When the reagent is exposed to a direct light source, typically ultraviolet light, a photochemical reaction creates a covalent bond between the photogroup and the surface of the medical device, thereby imparting the desired property to the surface. A covalent bond is a very strong chemical bond that results from the sharing of electrons between carbon atoms of the substrate and the applied coating, making the coating very durable and resilient.
Our proprietary PhotoLink reagents can be applied to a variety of substrates. Our reagents are easily applied to the material surface by dipping, spraying, roll coating, ink jetting or brushing. We continue to expand our portfolio of proprietary reagents for use by our customers. These reagents enable our customers to develop novel surface features for their devices, satisfying the expanding requirements of the healthcare industry. We are also continually working to expand the list of materials that are compatible with our surface modification and drug delivery reagents. Additionally, we develop coating processes and coating equipment to meet the device quality, manufacturing throughput and cost requirements of our customers.
Our drug delivery coating technologies differ from PhotoLink in that they involve non-photochemical reagents. Therapeutic drugs are incorporated within our proprietary polymer matrices to provide controlled, site specific release of the drug into the surrounding environment. The release of the drug can be tuned to elute quickly (in a few days) or slowly (ranging from several months to over a year), illustrating the wide range of release profiles that can be achieved with our coating systems. On a wide range of devices, drug-eluting coatings can help improve device performance, increase patient safety and enable innovative new treatments. We work with companies in the pharmaceutical, biotechnology and medical device industries to develop specialized coatings that allow for the controlled release of drugs from device surfaces. We see at least three primary areas with strong future potential: (1) improving the function of a device which itself is necessary to treat the medical condition; (2) enabling drug delivery in cases where the device serves only as a vehicle to deliver a drug to a specific site in the body; and (3) enhancing the biocompatibility of a medical device to ensure that it continues to function over a long period of time.
6
We offer customers several distinct polymer families for site specific drug delivery. Our Bravo™ Drug Delivery Polymer Matrix is utilized on the CYPHER® Sirolimus-eluting Coronary Stent from Cordis Corporation, a Johnson & Johnson company. CYPHER® is a trademark of Cordis Corporation. The Bravo polymer is a durable coating and is also used on our I-vation™ intravitreal implant within our Ophthalmology business unit. In addition, we offer several biodegradable polymer technologies that can be used for drug delivery applications. Because some biodegradable polymers can deliver proteins and other large molecule therapeutic agents, they have the potential to expand the breadth of drug delivery applications we can pursue. Biodegradable polymers can be combined with one or more drugs and applied to a medical device, and the drug is then released as the polymer degrades in the body over time.
Two key differentiating characteristics of our coatings are their flexibility and ease of use. In terms of flexibility, coatings can be applied to many different kinds of surfaces and can immobilize a variety of chemical, pharmaceutical and biological agents. This flexibility allows customers to be innovative in the design of their products without significantly changing the dimensions or other physical properties of the device. Additionally, the surface modification process can be tailored to provide customers with the ability to improve the performance of their devices by choosing the specific coating properties desired for particular applications. Our surface modification technologies also can be combined to deliver multiple surface-enhancing characteristics on the same device.
In terms of ease of use, unlike competitive coating processes, the PhotoLink coating process is relatively simple and is easily integrated into the customer’s manufacturing process. In addition, it does not subject the coated products to harsh chemical or temperature conditions, produces no hazardous byproducts, and does not require lengthy processing or curing time. Further, our Photolink coatings are compatible with generally accepted sterilization processes, so the surface attributes are not lost when the medical device is sterilized.
Systemic and Local Drug Delivery Through Injectable Microparticles and Implants
Through our acquisition of Brookwood Pharmaceuticals, as well as internal development and acquisition of our biodegradable materials, we offer customers drug delivery systems based on polymer-based microparticles and implants. These systems enable the controlled delivery of a broad variety of drugs, ranging in size from small molecule drugs to larger molecule drugs such as peptides and proteins. Depending on the drug and application, our microparticles and implants can incorporate drugs for delivery over days to weeks to months.
Brookwood Pharmaceuticals scientists have developed an extensive body of experience, know-how and patented capability in the field of microparticle drug delivery, working with a very wide range of drug classes. Our microparticles incorporate a customer’s drug and our polymers into very small particles that are measured in microns (1,000 microns equals one millimeter). Using our extensive technology base, we can develop long-acting, injectable microparticles for systemic, local, and cellular delivery of active pharmaceutical ingredients and vaccines. A variety of commercially viable microencapsulation processes are used including: solvent extraction, solvent evaporation, phase separation, fluid bed coating, and spray drying. Based on the desired product specifications, our development team will select the appropriate microencapsulation process, as well as the formulation variables to achieve dose, duration and other product specifications.
Injectable solid implants are rod, coil or other-shaped devices with drug dispersed throughout a polymer matrix. They are designed to release the drug at a programmed rate for days, weeks, or months. This type of drug delivery dosage form is especially suitable when efficacy is dependent on delivering a relatively large dose of a drug over a long duration. The polymer matrix controls the rate of release of drug from the implant. We are developing long-acting implants with biodegradable and non-biodegradable polymers. One of our biodegradable drug delivery implant systems has shape memory properties. This capability allows the implant to be delivered in one shape so that it can be placed through a catheter or other delivery device, after which the implant returns to its original shape once delivered to the desired site in the body.
Through our Brookwood Pharmaceuticals business unit, we are also collaborating with Genzyme Pharmaceuticals, a business unit of Genzyme Corporation, to develop novel drug delivery solutions, with an initial focus on peptide delivery. The relationship offers customized solutions for parenteral formulations by combining expertise in design for peptide delivery, peptide synthesis, and drug delivery technologies.
7
SurModics’ Surface Modification and Drug Delivery Technologies – Clinical Benefits
- Drug Delivery and Surface Modification Markets
5
6
7
| • | Drug | |
| • | ||
| • |
8
| to form a stable endothelial |
| • | Hemo/ | |
| • | Tissue
| |
| • | ||
| • | DNA and Protein |
| Desired Surface | ||
Market Segment Served | Examples of Applications
| |
Interventional cardiology and vascular access | Lubricity: catheters, guidewires | |
Hemocompatibility: vascular stents, catheters, distal protection devices | ||
Drug/biologics delivery: vascular stents, catheters | ||
Prohealing: vascular stents, vascular grafts |
9
| Desired Surface Property and | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Market Segment Served | Examples of Applications | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cardiac rhythm management | Lubricity: pacemaker and defibrillator leads, electrophysiology devices | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Hemocompatibility: electrophysiology devices | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prohealing: pacemaker and defibrillator leads | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug/biologics delivery: pacemaker and defibrillator leads | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cardiothoracic surgery | Prohealing: heart valves, septal defect repair devices | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Hemocompatibility: minimally invasive bypass devices, vascular grafts, ventricular assist devices | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In VitroDiagnostics | Lubricity: microfluidic devices | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Hemocompatibility: blood/glucose monitoring devices, biosensors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Biomolecule immobilization: DNA and protein arrays, protein attachment to synthetic nanofibrillar extracellular matrix for cell culture applications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cell culture growth and tissue integration: cell culture products,in vitro applications using synthetic nanofibrillar extracellular matrix to provide a more ‘‘in vivo-like” surface | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Interventional neurology and neurosurgery | Lubricity: catheters, guidewires | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prohealing: neuroembolic devices | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tissue engineering: aneurysm repair devices | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Urology and gynecology | Lubricity: urinary catheters, incontinence devices, ureteral stents, fertility devices | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug/biologics delivery: prostatic stents, microparticle injections | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tissue engineering: female sterilization devices | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ophthalmology | Drug/biologics delivery: sustained drug delivery | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Orthopedics | Cell growth and
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
10
Other customers, particularly in the pharmaceutical and biotechnology industries, prefer to outsource the manufacturing of drug delivery formulations to partners. Accordingly, we are investing in our Brookwood Pharmaceuticals manufacturing facility in Alabama in order to meet the Current Good Manufacturing Practice (CGMP) manufacturing needs of our customers.
The increase in research and development revenue reflects the addition of our Brookwood Pharmaceuticals business unit.
10
We currently have 100September 30, 2008, we had 103 licensed productsproduct classes (customer products utilizing SurModics technology) already on the market generating royalties and 94105 customer productsproduct classes incorporating our technology pending regulatory approval. These 194 products208 product classes are being sold or developed by 92101 licensed customers. We signed a record 2723 new licenses in fiscal 2007, up from 21 new licenses signed2008, compared with 27 in fiscal 2006.
2007.
11
During fiscal 1999, we launched our 3D-Link® Activated Slide to the genomics market. Coated glass slides are used by genomics researchers to prepare microarrays for DNA analysis. General Electric Company, through GE Healthcare, had an exclusive license to our coated glass slide technology that, in fiscal 2007, became a non-exclusive license. In addition to license fees, we generate revenue under this license from the manufacture and sale of coated glass slides to GE Healthcare, who markets the slides under their CodeLink® brand.
Stabilization Products
Substrates
Through
11
Ultra-Web™
12
will not receive any additional diagnostic royalty revenue.
In order to better serve our customers, in November 2008 we announced the creation of a new centralized R&D function in order to serve the needs of the Company’s clinically and market focused business units. Our Brookwood Pharmaceuticals business unit will continue to have its own R&D operations, which will remain unchanged.
12
to meet these needs across multiple medical markets. In addition, we are expanding our drug delivery and surface modification technology expertise to capture more of the final product value. We are doing this by, in selected cases, developing or acquiring technologies or devices to develop from feasibility stage up to and including animal and human clinical tests.testing stage. There can be no assurance that we will be successful in developing or acquiring additional technologies or devices.
13
of which 42 were exclusively licensed from others.
revenue.
13
14
On July 10, In fiscal 2007 SurModics acquired Brookwood and BioFX for $42.3 million and $11.6 million, respectively. There are contingent consideration features associated with each acquisition whereby SurModics may be required to pay up to an additional $22 million in cash related to Brookwood and $11.4 million in cash related to BioFX. In fiscal 2008 both entities achieved earnout targets resulting in additional cash consideration paid of $2 million and $1.1 million for Brookwood and BioFX, respectively. The additional cash consideration was recorded as an increase to goodwill.
On August 1, 2007, See Note 2 to the Consolidated Financial Statements for discussion of SurModics’ accounting for this investment.
On August 13, 2007, we announced our acquisition of BioFX Laboratories, Inc., a provider of substrates to thein vitro diagnostics industry, for $11.3 million in cash at closing and up to an additional $11.4 million in cash upon the successful achievement of specified revenue targets. We will account for our acquisition of BioFX under the purchase method of accounting. BioFX Laboratories is a leading manufacturer of substrates, a critical component of diagnostic test kits used to detect and signal that a certain reaction has taken place. We expect our acquisition of BioFX to broaden our product portfolio in thein vitro diagnostics market and expand marketing opportunities for each company’s products by way of complementary customer bases.
release formulations.
14
of appropriate U.S. and EU regulatory approvals for these new combination products. We will also be paid for our activities in researching and developing these combination products. Additionally, under the terms of our agreement with Merck, we will be responsible for the exclusive manufacture and supply of clinical and commercial products. We will also receive royalties on sales of products developed under our collaboration. The loss of one or more of our largest customers could have a material adverse effect on our business, financial condition, results of operations, and cash flow as discussed in more detail below.
15
15
technology. In order to fulfill our commitments to Merck, however, our facilities will need to be upgraded. In this regard,construction, which we expect to make investments in our operations to add capacity and to bring our facilities into compliance with GMP and other applicable regulatory standards. We may enter into similar arrangements with other of our customers where a clear, strategic rationale exists.
be completed by Fall 2009.
16
genes.
Although we are not currently regulated by Good Manufacturing Practices (GMP), we
16
has been approved by the FDA. The burden of obtaining FDA approval of the NDA or BLA rests with our customers.
17
the Company’s significant reliance on its relationship with Cordis, which causes our financial results andstock price to be subject to factors affecting Cordis and its CYPHER®stent program, including amongothers, the rate of market penetration by Cordis, the timing of market introduction of competing products,product safety or efficacy concerns and intellectual property litigation generally and specifically the litigation involving Boston Scientific Scimed, Inc. and Cordis in the U.S. District Court for the District of Delaware in which each was reported in June and July 2005 to have infringed the patent rights of the other;
| • | the Company’s significant reliance on its relationship with Cordis, which causes our financial results and stock price to be subject to factors affecting Cordis and its CYPHER® stent program, including among others, the rate of market penetration by Cordis, the timing of market introduction of competing products, product safety or efficacy concerns and intellectual property litigation the outcome of which could adversely affect the royalty revenue we derive based on the sales of licensed products. | |
| • | General economic conditions we are subject to which are beyond our control, including the impact of recession, business investment and changes in consumer confidence; | |
| • | frequent intellectual property litigation in the medical device industry that may directly or indirectly adversely affect our customers’ ability to market their products incorporating our technologies; | |
| • | our ability to protect our own intellectual property; | |
| • | healthcare reform efforts and reimbursement rates for medical device products that may adversely affect our customers’ ability to cost-effectively market and sell devices incorporating our technologies; |
17
frequent intellectual property litigation in the medical device industry that may directly or indirectlyadversely affect our customers’ ability to market their products incorporating our technologies;our ability to protect our own intellectual property;healthcare reform efforts and reimbursement rates for medical device products that may adversely affect ourcustomers’ ability to cost-effectively market and sell devices incorporating our technologies;the Company’s ability to attract new licensees and to enter into agreements for additional productapplications with existing licensees, the willingness of potential licensees to sign license agreements underthe terms offered by the Company, and the Company’s ability to maintain satisfactory relationships with itslicensees;the Company’s ability to increase the number of market segments and applications that use its coatingtechnologies through its sales and marketing and research and development efforts;the Company’s ability to facilitate through strategic investment and research and development support thecreation of new medical device market segments and applications that incorporate its coating technologies;market acceptance of products sold by customers incorporating our technologies and the timing of newproduct introductions by licensees;market acceptance of products sold by customers’ competitors and the timing and pricing of new productintroductions by customers’ competitors;the difficulties and uncertainties associated with the lengthy and costly new product development andforeign and domestic regulatory approval processes, such as delays, difficulties or failures in achievingacceptable clinical results or obtaining foreign or FDA marketing clearances, which may result in lost marketopportunities or postpone or preclude product commercialization by licensees;efficacy or safety concerns with respect to products marketed by us and our licensees, whether scientificallyjustified or not, that may lead to product recalls, withdrawals or declining sales;the ability to secure raw materials for reagents the Company sells;the Company’s ability to manage successfully clinical trials and related foreign and domestic regulatoryprocesses for the I-vation™ intravitreal implant or other acquired products from InnoRx under developmentby the Company’s Ophthalmology business unit, whether delays, difficulties or failures in achievingacceptable clinical results or obtaining foreign or FDA marketing clearances postpone or preclude productcommercialization of the intravitreal implant or other acquired products, and whether the intravitreal implantand any other acquired products remain viable commercial prospects;product liability claims not covered by insurance;the development of new products or technologies by competitors, technological obsolescence and otherchanges in competitive factors;the trend of consolidation in the medical device industry, resulting in more significant, complex and longterm contracts than in the past and potentially greater pricing pressures;the Company’s ability to identify suitable businesses to acquire or with whom to form strategic relationshipsto expand its technology development and commercialization, its ability to successfully integrate theoperations of companies it may acquire from time to time and its ability to create synergies from acquisitionsand other strategic relationships;the Company’s ability to successfully internally perform certain product development activities andgovernmental and regulatory compliance activities with respect to acquired technology, including InnoRxtechnology, which activities the Company has not previously undertaken in any significant manner;the Company’s ability to improve and qualify its facilities to accommodate its obligations to manufactureand supply clinical and commercial quantities of the I-vation™ intravitreal implant for Merck and otherpotential licensees of that technology;
18
economic and other factors over which the Company has no control, including changes in inflation andconsumer confidence;acts of God or terrorism which impact the Company’s personnel or facilities; andother factors described below in “Risk Factors.”
| • | the Company’s ability to attract new licensees and to enter into agreements for additional product applications with existing licensees, the willingness of potential licensees to sign license agreements under the terms offered by the Company, changes in the development and marketing priorities of our licensees and development partners and the Company’s ability to maintain satisfactory relationships with its licensees; | |
| • | the Company’s ability to increase the number of market segments and applications that use its technologies through its sales and marketing and research and development efforts; | |
| • | the Company’s ability to facilitate the creation of new market segments and applications that incorporate its technologies through strategic investment and research and development support; | |
| • | market acceptance of products sold by customers incorporating our technologies and the timing of new product introductions by licensees; | |
| • | market acceptance of products sold by customers’ competitors and the timing and pricing of new product introductions by customers’ competitors; | |
| • | the difficulties and uncertainties associated with the lengthy and costly new product development and foreign and domestic regulatory approval processes, such as delays, difficulties or failures in achieving acceptable clinical results or obtaining foreign or FDA marketing clearances or approvals, which may result in lost market opportunities or postpone or preclude product commercialization by licensees; | |
| • | efficacy or safety concerns with respect to products marketed by us and our licensees, whether scientifically justified or not, that may lead to product recalls, withdrawals or declining sales; | |
| • | the ability to secure raw materials for reagents the Company sells; | |
| • | the Company’s ability to successfully manage clinical trials and related foreign and domestic regulatory processes for the I-vationtm intravitreal implant or other products under development by the Company, whether delays, difficulties or failures in achieving acceptable clinical results or obtaining foreign or FDA marketing clearances or approvals postpone or preclude product commercialization of the intravitreal implant or other products, and whether the intravitreal implant and any other products remain viable commercial prospects; | |
| • | product liability claims not covered by insurance; | |
| • | the development of new products or technologies by competitors, technological obsolescence and other changes in competitive factors; | |
| • | the trend of consolidation in the medical device industry, resulting in more significant, complex and long term contracts than in the past and potentially greater pricing pressures; | |
| • | the Company’s ability to identify suitable businesses to acquire or with whom to form strategic relationships to expand its technology development and commercialization, its ability to successfully integrate the operations of companies it may acquire from time to time and its ability to create synergies from acquisitions and other strategic relationships; | |
| • | the Company’s ability to successfully internally perform certain product development activities and governmental and regulatory compliance activities which the Company has not previously undertaken in any significant manner; | |
| • | acts of God or terrorism which impact the Company’s personnel or facilities; and | |
| • | other factors described below in “Risk Factors.” |
19
ITEM 1A. RISK FACTORS.
| ITEM 1A. | RISK FACTORS. |
20
19
production and marketing capabilities than us. Competitors may succeed in developing competing technologies or obtaining governmental approval for products before us. Products incorporating our competitors’ technologies may gain market acceptance more rapidly than products using ours. Developments by competitors may render our existing and potential products noncompetitive or obsolete. Furthermore, there can be no assurance that new products or technologies developed by others, or the emergence of new industry standards, will not render our products or technologies or licensees’ products incorporating our technologies noncompetitive or obsolete. Any new technologies whichthat make our drug delivery or surface modification or drug delivery technologies less competitive or obsolete would have a material adverse effect on our business, financial condition and results of operations.
We
an inability to assimilate acquired operations, personnel, technology, information systems, and internalcontrol systems and products;diversion of management’s attention;difficulties and uncertainties in transitioning the customers or other business relationships from the acquiredentity to us; andthe loss of key employees of acquired companies.
| • | an inability to assimilate acquired operations, personnel, technology, information systems, and internal control systems and products; | |
| • | diversion of management’s attention, including the need to manage several remote locations with a limited management team; | |
| • | difficulties and uncertainties in transitioning the customers or other business relationships from the acquired entity to us; and | |
| • | the loss of key employees of acquired companies. |
Our acquisition of Brookwood Pharmaceuticals was the largest in our Company’s history. The process of integrating any acquired business, technology, or product into our business and operations may result in unforeseen operating difficulties and expenditures, including those described above. Our ability to realize the anticipated benefits of our acquisition of Brookwood will require the integration of our sales and marketing efforts to certain customers, integration of information technology and other administration systems. Additional operating difficulties may arise as a result of our having to manage a large, remote location with a limited management team. Failure to successfully integrate Brookwood into our operationsnew technologies may adversely affect our operating results.
21
20
The success of our business depends on a number of factors, including our continued research and development of new technologies for future commercialization. In researching and developing such new technologies, we may incur significant expenses that may adversely affect our operating results, including our profitability. Additionally, these activities are subject to risks of failure that are inherent in the development of new medical technologies and as a result, may never result in commercially viable technologies.
We recognize revenue in accordance with various complex accounting standards, and changes in circumstances or interpretations may lead to accounting adjustments.
21
marketing efforts may be unsuccessful, resulting in delayed or discontinued products. Hence, the amount and timing of royalty payments received by us will fluctuate, and such fluctuations could have a material adverse effect on our business, financial condition and results of operations.
22
22
instituted by a device manufacturer or us or required by a regulatory agency. A product liability claim, recall or other claim with respect to uninsured liabilities or for amounts in excess of insured liabilities could have a material adverse effect on our business, financial condition and results of operations.
Our manufacture and supply of pharmaceutical products may subject us to product liability claims directly against us that could result in costly litigation and significant liability
Our involvement in the manufacture and supply of clinical and commercial quantities of the I-vation intravitreal implant for Merck, and potentially other licensees, may expose us to significant risk of product liability claims filed directly against us. Any product liability claims, with or without merit, could result in costly litigation, reduced sales, significant liabilities and diversion of our management’s time, attention and resources. We have obtained a level of liability insurance coverage that we believe is adequate in scope and coverage given our current stage of development. However,development, however we cannot be sure that our product liability insurance coverage is adequate or that it will continue to be available to us on acceptable terms,
23
results of operations.
24
Although surface modification and drug delivery technologies themselves are not directly regulated, the medical
23
products typically rests with our licensees, (thethe medical device or pharmaceutical manufacturers).manufacturers. However, we have prepared Drug Master Files and Device Master Files which may be accessed by the FDA and other regulatory authorities to assist them in their review of the applications filed by our licensees.
25
We are currently collaborating with Merck in conducting a Phase I safety trial for our I-vation™ intravitreal implant. The Phase I trial is intended to help assess the safety and tolerability of the implant in patients with diabetic macular edema (DME), and is being conducted under an investigational new drug application with the U.S. Food and Drug Administration. A total of thirty subjects were enrolled in this Phase I trial, which enrollment was completed in March 2006, and will be subject to follow-up monitoring for three years. Merck’s ability to commercialize this implant in a timely manner will depend upon the success of this Phase I safety trial, as well as future required clinical trials that will further evaluate and document the safety profile and therapeutic benefit in targeted patient populations. Although the early results of the Phase I trial have not identified any significant safety issues, we cannot be certain the implant will perform as expected in additional clinical tests. Problems in connection with our Phase I trials or in any subsequent phases of required clinical trials may prevent or delay our or our partner’s obtaining necessary regulatory approvals and threaten our ability to timely or cost-effectively commercialize the implant, if at all. Our Phase I trial is being conducted on a statistically insignificant number of human patients and is not intended to evaluate aspects of the effectiveness of the implant. Because the initial number of tests performed in humans has been relatively small, there is no assurance that the Phase I trials will identify problems that may become evident from a larger base of tests or after a longer period of observation of the patients. We will be able to accurately evaluate the performance of the implant in humans only after extensive testing in large numbers of patients over a period of years.
We may face liability if we mishandle or improperly dispose of the hazardous materials used in some of our research, development and manufacturing processes.
24
RISKS RELATING TO OUR INTELLECTUAL PROPERTY
If we cannot adequately protect our technologies and proprietary information, we may be unable to sustain a competitive advantage.
Our success depends, in large part, on our ability to obtain and maintain patents, maintain trade secret protection, operate without infringing on the proprietary rights of third parties and protect our proprietary rights against infringement by third parties. We have been granted U.S. and foreign patents and have U.S. and foreign patent applications pending related to our proprietary technologies. There can be no assurance that any pending patent application will be approved, that we will develop additional proprietary technologies that are patentable, that any patents issued will provide us with competitive advantages or will not be challenged or invalidated by third parties, or that the patents of others will not prevent the commercialization of products incorporating our technologies. Furthermore, there can be no assurance that others will not independently develop similar technologies, duplicate any of our technologies or design around our patents.
We may become involved in expensive and unpredictable patent litigation or other intellectual property proceedings which could result in liability for damages, or impair our development and commercialization efforts.
Our commercial success also will depend, in part, on our ability to avoid infringing patent or other intellectual property rights of third parties. There has been substantial litigation regarding patent and other intellectual property rights in the medical device and pharmaceutical industries, and intellectual property litigation may be used against us as a means of gaining a competitive advantage. Intellectual property litigation is complex, time consuming and expensive, and the outcome of such litigation is difficult to predict. If we were found to be infringing any third party patent or other intellectual property right, we could be required to pay significant damages, alter our products or processes, obtain licenses from others, which we may not be able to do on commercially reasonable terms, if at all, or cease commercialization of our products and processes. Any of these outcomes could have a material adverse effect on our business, financial condition and results of operations.
Patent litigation or U.S. Patent and Trademark Office interference proceedings may also be necessary to enforce any patents issued or licensed to us or to determine the scope and validity of third party proprietary rights. These activities could result in substantial cost to us, even if the eventual outcome is favorable to us. An adverse outcome of any such litigation or interference proceeding could subject us to significant liabilities to third parties, require disputed rights to be licensed from third parties or require us to cease using our technology. Any action to defend or prosecute intellectual property would be costly and result in significant diversion of the efforts of our management and technical personnel, regardless of outcome, and could have a material adverse effect on our business, financial condition and results of operations.
If we are unable to keep our trade secrets confidential, our technology and proprietary information may be used by others to compete against us.
We rely significantly upon proprietary technology, information, processes and know-how that are not subject to patent protection. We seek to protect this information through trade secret or confidentiality agreements with our employees, consultants, potential licensees, or other parties as well as through other security measures. There can be no assurance that these agreements or any security measure will provide meaningful protection for our unpatented proprietary information. In addition, our trade secrets may otherwise become known or be independently developed by competitors.
25
If we or any of our licensees breach any of the agreements under which we have in-licensed intellectual property from others, we could be deprived of important intellectual property rights and future revenue.
We are a party to various agreements through which we have in-licensed or otherwise acquired from third parties rights to certain technologies that are important to our business. In exchange for the rights granted to us under these agreements, we agree to meet certain research, development, commercialization, sublicensing, royalty, indemnification, insurance, and other obligations. If we or one of our licensees fails to comply with these obligations set forth in the relevant agreement through which we have acquired rights, we may be unable to effectively use, license, or otherwise exploit the relevant intellectual property rights and may be deprived of current or future revenues that are associated with such intellectual property.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS. |
26
ITEM 1B. UNRESOLVED STAFF COMMENTS.
None.
ITEM 2. PROPERTIES.
| ITEM 2. | PROPERTIES. |
use to accommodate our growth needs.
ITEM 3. LEGAL PROCEEDINGS.
The information in
| ITEM 3. | LEGAL PROCEEDINGS. |
ITEM 4. SUBMISSION OF MATTERS TO A VOTE OF SECURITY HOLDERS.
contingencies.
| ITEM 4. | SUBMISSION OF MATTERS TO A VOTE OF SECURITY HOLDERS. |
26
2008.
The
Name | Age | Position | ||||
| Bruce J Barclay | 52 | President and Chief Executive Officer | ||||
| Aron B. Anderson, Ph.D. | 45 | Vice President and Chief Scientific Officer | ||||
| Philip D. Ankeny | 45 | Senior Vice President and Chief Financial Officer | ||||
| Douglas P. Astry | 56 | General Manager, In Vitro Technologies | ||||
| Lise W. Duran, Ph.D. | 53 | Vice President of Research | ||||
| Paul A. Lopez | 52 | Vice President and President — Ophthalmology Division | ||||
| Charles W. Olson | 44 | Vice President and General Manager | ||||
| Bryan K. Phillips | 37 | Vice President, General Counsel and | ||||
| Brian L. Robey | 45 | Vice President of Product Development and | ||||
| Michael J. Shoup | 48 | Vice President of Quality, Regulatory and Clinical | ||||
| Affairs | ||||||
| Arthur J. Tipton, Ph.D. | 51 | Vice President, and President of Brookwood | ||||
| Pharmaceuticals | ||||||
| Jan M. Webster | 49 | Vice President of Human Resources | ||||
27
27
over five years with IBM and Shiva in sales, marketing and business development roles. Mr. Ankeny also serves on the Board of Directors of Innovex, Inc., which designs and manufactures flexible circuit interconnect solutions to original equipment manufacturers in the electronics industry. Mr. Ankeny received an A.B. degree in economics and engineering from Dartmouth College in 1985 and an M.B.A. from Harvard Business School in 1989.
Steven J. Keoughjoined the Company as its Senior Vice President and Chief Intellectual Property Counsel in January 2004 and added the duties of Vice President and General Manager of the New Ventures business unit in April of that year. The current Orthopedics business unit emerged in October 2005 from New Ventures, and is led by Mr. Keough. Before joining SurModics, Mr. Keough practiced law at Minneapolis-based Fredrikson & Byron, P.A. from 2000-2003, where he was a senior member and past chairman of the intellectual property department. He previously served as president and co-founder of the intellectual property law firm Patterson & Keough, P.A. from 1991-2000. He was also Manager of Asia-Pacific at the Minneapolis law firm of Merchant & Gould, from 1987-1991. Mr. Keough has extensive business and legal experience involving medical technologies, technology transfer, strategic planning, licensing and high technology business management. Mr. Keough earned a J.D. from Boston College in 1987, an M.A. from the Catholic University of America in 1982, and a Bachelor of Science degree from the United States Naval Academy in 1977.
Paul A. Lopezjoined the Company in July 2005 as Vice President and President of the Company’s Ophthalmology business unit. Before joining SurModics, Mr. Lopez was President and CEO of Valley Forge Pharmaceuticals, an early stage pharmaceutical company from March 2001 to July 2005. Prior to Valley Forge, Mr. Lopez served in various senior level positions at Bausch & Lomb, including President, North America Surgical; Vice President, Commercial Operations, Americas and Asia Pacific Regions; and Vice President, Business Integration from January 1999 to March 2001. Mr. Lopez has also held roles at Monsanto Company, Pharmacia and Upjohn, Inc. and Iolab Corporation. Mr. Lopez serves on the Board of Directors of Alliance Medical Products, a private company located in Irvine, California. Mr. Lopez received an M.B.A. from California State Polytechnic University in 1984and a B.S. in Business Administration from California State University, Long Beach in 1979.
1979 and an M.B.A. from California State Polytechnic University in 1984.
28
2005, the position of Vice President, Sales was added to his responsibilities. In November 2008 following the change in our organizational structure, Mr. Olson was named Vice President of our
28
in 1995.
1987.
29
29
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES. |
| Fiscal Quarter Ended: | High | Low | |||
| September 30, 2007 | 52.68 | 44.72 | |||
| June 30, 2007 | 50.00 | 35.30 | |||
| March 31, 2007 | 38.49 | 31.11 | |||
| December 31, 2006 | 35.38 | 31.10 | |||
| September 30, 2006 | 38.00 | 33.36 | |||
| June 30, 2006 | 39.65 | 31.92 | |||
| March 31, 2006 | 40.22 | 32.90 | |||
| December 31, 2005 | 43.37 | 36.46 | |||
High Low September 30, 2008 45.06 28.05 June 30, 2008 47.88 42.00 March 31, 2008 55.40 38.17 December 31, 2007 56.09 48.35 September 30, 2007 52.68 44.72 June 30, 2007 50.00 35.30 March 31, 2007 38.49 31.11 December 31, 2006 35.38 31.10 7, 2007,8, 2008, there were 295284 holders of record of our Common Stock and approximately 5,78413,826 beneficial owners of shares registered in nominee or street name.2007,2008, by the Company or on behalf of the Company or any “affiliated purchaser” of the Company, as defined inRule 10b-18(a)(3) under the Exchange Act.
____________________ (c) (d) Total Number Maximum of Shares Number of Purchased Shares that as Part of May Yet Be (a) (b) Publicly Purchased Total Number Average Announced Under the of Shares Price Paid Plans or Plans or Period Purchased(1) Per Share(1) Programs Programs 7/1/07 - 7/31/07 6,390 $ 50.87 N/A N/A 8/1/07 - 8/31/07 33,400 $ 49.48 N/A N/A 9/1/07 - 9/30/07 7,810 $ 48.00 N/A N/A Total 47,600 $ 49.42 N/A N/A (c) (d) Total Number Dollar Maximum of Shares Value of Purchased Shares That as Part of May Yet be (a) (b) Publicly Purchased Total Number Average Announced Under the of Shares Price Paid Plans or Plans or Purchased(1) per Share(1) Programs Programs(2) 7/1/08 — 7/31/08 153,277 $ 43.17 105,000 $ 23,781,637 8/1/08 — 8/31/08 35,400 $ 40.95 35,400 $ 22,332,097 9/1/08 — 9/30/08 26,730 $ 37.50 0 $ 22,332,097 Total 215,407 $ 42.10 140,400 $ 22,332,097 (1) AllThe purchases in this column include (i) shares repurchased as part of theour publicly announced program, (ii) 30,619 shares that were repurchased by the Company to pay the exercise price and/or to satisfy tax withholding obligations in connection with so-called “stock swap exercises” related to the vesting of employee stock options or restricted stock awards, and (iii) and 40,388 shares that had previously been issued to seven employees.former stockholders of InnoRx, Inc.(2) On November 15, 2007, our Board of Directors announced the authorization of the repurchase of $35 million of our outstanding common stock. As of September 30, 2008, we have repurchased 297,900 shares at an average price of $42.52 per share. Under the current authorization, the Company has $22.3 million available for authorized share repurchases as of September 30, 2008. The repurchase authorization does not have an expiration date.
30

31
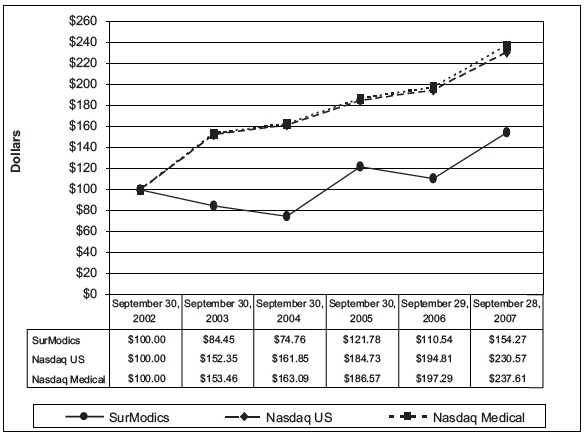
31
ITEM 6. SELECTED FINANCIAL DATA.
| ITEM 6. | SELECTED FINANCIAL DATA. |
| Fiscal Year | ||||||||||||||||
| (Dollars in Thousands) | 2007 | 2006 | 2005 | 2004 | 2003 | |||||||||||
| Statements of Operations Data: | ||||||||||||||||
| Total revenue | $ | 73,164 | $ | 69,884 | $ | 62,381 | $ | 49,738 | $ | 43,232 | ||||||
| Operating income | 9,899 | 36,163 | 2,985 | 10,474 | 20,640 | |||||||||||
| Net income (loss) | 3,347 | 20,334 | (8,246 | ) | 7,242 | 13,936 | ||||||||||
| Diluted net income (loss) per share | 0.18 | 1.09 | (.45 | ) | .41 | .78 | ||||||||||
| Balance Sheet Data: | ||||||||||||||||
| Cash and investments | $ | 26,308 | $ | 58,813 | $ | 24,445 | $ | 19,215 | $ | 6,647 | ||||||
| Total assets | 171,331 | 157,402 | 124,225 | 109,587 | 97,808 | |||||||||||
| Retained earnings | 51,620 | 48,273 | 27,914 | 36,161 | 28,918 | |||||||||||
| Total stockholders’ equity | 130,922 | 145,203 | 115,581 | 94,310 | 86,114 | |||||||||||
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
| Fiscal Year | ||||||||||||||||||||
| 2008 | 2007 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Dollars in thousand, except per share data) | ||||||||||||||||||||
| Statements of Operations Data: | ||||||||||||||||||||
| Total revenue | $ | 97,051 | $ | 73,164 | $ | 69,884 | $ | 62,381 | $ | 49,738 | ||||||||||
| Operating income | 27,261 | 9,899 | 36,163 | 2,985 | 10,474 | |||||||||||||||
| Net income (loss) | 14,739 | 3,347 | 20,334 | (8,246 | ) | 7,242 | ||||||||||||||
| Diluted net income (loss) per share | 0.80 | 0.18 | 1.09 | (0.45 | ) | 0.41 | ||||||||||||||
| Balance Sheet Data: | ||||||||||||||||||||
| Cash | $ | 15,376 | $ | 13,812 | $ | 3,751 | $ | 3,921 | $ | 2,709 | ||||||||||
| Short-term and long-term investments | 56,602 | 56,413 | 102,820 | 69,398 | 60,594 | |||||||||||||||
| Total assets | 191,028 | 171,331 | 157,402 | 124,225 | 109,587 | |||||||||||||||
| Retained earnings | 66,439 | 51,620 | 48,273 | 27,914 | 36,161 | |||||||||||||||
| Total stockholders’ equity | 141,806 | 130,922 | 145,203 | 115,581 | 94,310 | |||||||||||||||
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS. |
32
32
In January 2005, we acquired all We will be reviewing our segment reporting for fiscal 2009 as a result of the assets of InnoRx, Inc. by paying cash and issuing shares of SurModics common stockchanges to InnoRx stockholders. InnoRx was an early-stage company developing drug delivery implants and therapies for the ophthalmology market. The assets we acquired were folded into our newly-created Ophthalmology business unit. Prior to the acquisition, SurModics held an ownership interestorganizational structure in InnoRx of less than 20% and accounted for the investment under the cost method. Upon completion of the InnoRx acquisition, we retroactively adjusted our previously reported results to show the impact of accounting for InnoRx under the equity method. The net impact was an approximate $194,000 reduction in net income for fiscal 2004 from previously reported results.
November 2008.
the triamcinolone acetonide development program. This program terminated 30 days following the notice given in September 2008.
33
Our consolidated financial statements have included the operating results of BioFX since the date of the acquisition.
Our
33
34
35
| Fiscal | Fiscal | |||||||||||||||
(Dollars in thousands) | 2008 | 2007 | Increase | % Change | ||||||||||||
| Revenue: | ||||||||||||||||
| Drug Delivery | $ | 43,938 | $ | 26,488 | $ | 17,450 | 66 | % | ||||||||
| Hydrophilic and Other | 31,864 | 26,493 | 5,371 | 20 | % | |||||||||||
In Vitro | 21,249 | 20,183 | 1,066 | 5 | % | |||||||||||
| Total revenue | $ | 97,051 | $ | 73,164 | $ | 23,887 | 33 | % | ||||||||
36
37
| Fiscal | Fiscal | Increase | |||||||||||||||
| (Dollars in thousands) | 2007 | 2006 | (Decrease) | % Change | |||||||||||||
| Revenue: | |||||||||||||||||
| Drug Delivery | $ | 26,488 | $ | 32,918 | $ | (6,430 | ) | (20)% | |||||||||
| Hydrophilic and Other | 26,493 | 22,233 | 4,260 | 19% | |||||||||||||
| In Vitro | 20,183 | 14,733 | 5,450 | 37% | |||||||||||||
| Total revenue | $ | 73,164 | $ | 69,884 | $ | 3,280 | 5% | ||||||||||
34
Fiscal Fiscal Increase 2007 2006 (Decrease) % Change Revenue: Drug Delivery $ 26,488 $ 32,918 $ (6,430 ) (20 )% Hydrophilic and Other 26,493 22,233 4,260 19 % 20,183 14,733 5,450 37 % Total revenue $ 73,164 $ 69,884 $ 3,280 5 % Brookwood Pharmaceuticals.Brookwood.
38
The decrease in drug delivery royalties and license fees principally reflects decreased royalty revenue from Cordis as a result of lower CYPHER® sales. Partially offsetting the decrease attributable to CYPHER® was an increase in royalties and license fees from ophthalmology customers, as well as an increase in research and development fees from drug delivery and ophthalmology customers. We received a $20 million license fee from Merck in association with the collaborative research and license agreement that we signed in fiscal 2007. However we recognized as revenue only a small portion of this fee, as we will beand are amortizing these paymentsthe remaining amounts over the estimated economic life of the technology we licensed to Merck. As a result of Merck’s September 2008 notice of termination, we anticipate recognizing these amounts in the first quarter of fiscal 2009. Fiscal 2007 sales of reagent chemicals (chemicals that we manufacture and sell to licensees for coating their medical devices) to Cordis decreased slightly when compared with the prior year. The unit volume of reagents sold to Cordis will likely be directly impacted by the proportion of stenting procedures that utilize drug eluting stents, in addition to relative market share positions of drug eluting stent players.
The CYPHER® stent, from which we derive a substantial majority of our Drug Delivery revenue, faces continuing competition from Boston Scientific Corporation’s Taxus drug eluting stent, which is sold within and outside the U.S., and stents from Medtronic, Abbott Vascular and others sold outside the U.S. In addition, drug eluting stents from Medtronic and Abbott are expected to be approved in the U.S. within the next year. These stents compete or will compete directly with the CYPHER® stent. In addition to competition among the various players, the total size of the drug eluting stent market has decreased significantly in the past eighteen months as a result of concerns about product safety, mostly related to potential clotting associated with stents. Therefore, future royalty and reagent sales revenue could decrease because of lower CYPHER® stent sales as a result of the market contraction and the ongoing and expected future competition. We anticipate that quarterly royalty revenue from the CYPHER® stent may be volatile throughout fiscal 2008 and beyond as the various marketers of drug eluting stents continue competing in the marketplace and as others enter the marketplace. Management expects royalties from the CYPHER® stent to continue to constitute a significant portion of our revenue in fiscal 2008. However, whether and the extent to which royalties from the CYPHER® stent continue to constitute a significant source of revenue is subject to a number of risks, including intellectual property litigation generally, and specifically the damages, settlements and mutual agreements that may result from various infringement suits between Boston Scientific and Cordis in which each has been found to have violated certain intellectual property rights of the other.
The inclusion of Brookwood Pharmaceuticals, which contributed to Drug Delivery revenue for only two months in fiscal 2007, will also impact the overall revenue and mix in fiscal 2008. A substantial majority of Brookwood’s revenue is comprised of research and development fees.
35
management.
39
36
Years Ended September 30, 2006 and 2005
| Fiscal | Fiscal | ||||||||||||||||||||
| (Dollars in thousands) | 2006 | 2005 | Increase | % Increase | |||||||||||||||||
| Revenue: | |||||||||||||||||||||
| Drug Delivery | $ | 32,918 | $ | 29,678 | $ | 3,240 | 11 | % | |||||||||||||
| Hydrophilic and Other | 22,233 | 19,065 | 3,168 | 17 | % | ||||||||||||||||
| In Vitro | 14,733 | 13,638 | 1095 | 8 | % | ||||||||||||||||
| Total revenue | $ | 69,884 | $ | 62,381 | $ | 7,503 | 12 | % | |||||||||||||
Revenue.Fiscal 2006 revenue was $69.9 million, an increase of $7.5 million or 12% from fiscal 2005. We experienced growth in all three operating segments as detailed in the table above and further explained in the narrative below.
Drug Delivery.Revenue in the Drug Delivery segment increased 11% to $32.9 million in fiscal 2006. The growth in total revenue reflects increases in royalties and license fees, and research and development revenue related to drug delivery and ophthalmology projects.
Drug Delivery derives a substantial majority of its revenue from royalties and license fees and product sales attributable to Cordis Corporation, a Johnson & Johnson company, on its CYPHER® Sirolimus-eluting Coronary Stent. The CYPHER® stent incorporates a proprietary SurModics polymer coating that delivers a therapeutic drug designed to reduce the occurrence of restenosis in coronary artery lesions.
Over three-fourths of the overall increase in drug delivery revenue reflects increased royalty revenue from Cordis as a result of higher CYPHER® sales. The balance of the fiscal 2006 increase was a result of increased research and development fees from drug delivery and ophthalmology customers. Fiscal 2006 sales of reagent chemicals (chemicals that we manufacture and sell to licensees for coating their medical devices) to Cordis decreased slightly when compared with the prior year.
Hydrophilic and Other.Hydrophilic and Other revenue increased 17% to $22.2 million, primarily as a result of 19% growth in royalties and license fees and 35% growth in reagent sales, partially offset by a 15% decline in research and development revenue. In contrast to our Drug Delivery segment, where a significant percentage of revenue is attributable to Cordis, there are several dozen licensees and an even larger number of coated products generating royalties in our Hydrophilic and Other segment. The growth in royalties principally reflects increased sales of coated products already on the market, and to a lesser extent newly introduced licensed products.
In Vitro.Revenue in the In Vitro segment increased 8% to $14.7 million. Roughly 60% of the increase was attributable to growth in sales of our stabilization products used by diagnostic kit manufacturers in immunoassay diagnostic tests. The balance of the growth resulted from increased royalty revenue from our diagnostic format patents. In Vitro derives a significant percentage of its revenue from GE Healthcare and Abbott Laboratories.
Product costs.Product costs were $3.4 million in fiscal 2006, a 19% increase from the prior year. Overall product margins averaged 70%, on par with the 70% reported for the comparable period in 2005.
Research and development expenses.Research and development expenses were $20.4 million, an increase of 27% compared with fiscal 2005. Approximately $2.5 million of the $4.3 million increase was related to non-cash stock-based compensation charges following the adoption of SFAS No. 123(R). Research and development expenses included no such charge in fiscal 2005. Excluding stock-based compensation, research and development expenses increased 11% in fiscal 2006. The balance of the increase reflects higher costs associated with the clinical trial on our I-vation™ intravitreal implant, increased costs of operating the recently constructed clean rooms and drug coating suites at our Eden Prairie headquarters, and increased personnel costs. These increased costs were partially offset by reduced legal costs.
Selling, general and administrative expenses.Selling, general and administrative expenses were $9.9 million, an increase of 29% compared with fiscal 2005. We recorded approximately $2.9 million in non-cash stock-based compensation charges compared with $588,000 in fiscal 2005. Excluding the impact of stock-based compensation,
37
selling, general and administrative expenses decreased approximately 1% as a result of the cost savings realized since we exited our contract manufacturing facility in Bloomington in April 2006. The majority of the operating costs of the Bloomington facility were reported in selling, general and administrative expenses.
Asset impairment charge.Results in fiscal 2005 included a non-cash asset impairment charge of $2.5 million against our Bloomington, Minnesota, contract manufacturing facility. Results in fiscal 2004 included a non-cash asset impairment charge of $16.5 million against the facility. In September 2005, we entered into an agreement to sell the Bloomington facility and consolidated operations at our Eden Prairie, Minnesota, headquarters in April 2006.
Purchased in-process research and development.In January 2005, the Company acquired all of the assets of InnoRx, Inc. by paying cash and issuing shares of SurModics common stock to InnoRx stockholders. Results in the second quarter of fiscal 2005 include a non-cash in-process research and development charge of $30.3 million. The fair value of the in-process research and development was determined by an independent valuation consultant.
Other income, net.Other income resulted in a loss of $598,000 in fiscal 2006 compared with income of $1.4 million in fiscal 2005, primarily as a result of the $4.7 million impairment loss on our investment in Novocell we recorded in the second quarter of fiscal 2006. Income from investments was $4.2 million in fiscal 2006, an increase of $2.2 million, compared with $2.0 million in fiscal 2005. The increase reflects higher levels of investable cash and higher yields generated from our investment portfolio. Prior year other income results also include a $500,000 loss related to the impact of accounting for the InnoRx acquisition under the equity method. We recorded no such comparable transaction in fiscal 2006.
Income tax expense.The income tax provision was $15.2 million in fiscal 2006 compared with $12.6 million in fiscal 2005. Excluding the impact of the $4.7 million impairment loss (since the Company does not currently foresee offsetting capital gains to offset this capital loss, no tax benefit has been recorded), the effective tax rate was 38.2% in fiscal 2006, compared with 36.8% for fiscal 2005 when the impact of non-tax deductible purchased in-process research and development is excluded. The impact of adopting SFAS No. 123(R) accounts for the bulk of the increase in the effective tax rate from fiscal 2006.
38
| For the Years Ended | ||||||||
| September30, | ||||||||
| (Dollars in thousands) | 2007 | 2006 | ||||||
| Net income | $ | 3,347 | $ | 20,334 | ||||
| Depreciation and amortization | 4,214 | 3,710 | ||||||
| Stock-based compensation | 10,312 | 5,711 | ||||||
| Purchased in-process research & development | 15,573 | — | ||||||
| Asset impairment charge | — | 4,651 | ||||||
| Net other operating activities | (11,004 | ) | (3,929 | ) | ||||
| Net change in deferred revenue | 19,166 | 2,489 | ||||||
| Net change in other operating assets and liabilities | 9,107 | 2,313 | ||||||
| Net cash provided by operating activities | $ | 50,715 | $ | 35,279 | ||||
For the Years Ended September 30, 2008 2007 (Dollars in thousands) Net income $ 14,739 $ 3,347 Depreciation and amortization 6,071 4,214 Stock-based compensation 9,652 10,312 Purchased in-process research and development — 15,573 Impairment loss on investment 4,314 — Net other operating activities (3,938 ) (11,004 ) Net change in deferred revenue 11,452 19,166 Net change in other operating assets and liabilities (2,468 ) 9,107 Net cash provided by operating activities $ 39,822 $ 50,715
2008.
40
$1.7 million.
In January 2005, we made an equity investment of approximately $3.9 million in OctoPlus, a company based in the Netherlands active in the development of pharmaceutical formulations incorporating novel biodegradable polymers. In May 2006, we made an additional investment of approximately $160,000. As of September 30, 2006 the $4.1 million investment, which is accounted for under the cost method, represented an ownership interest of less than 20%. In October 2006, we made an additional investment of $1.9 million, bringing our total investment to $6.0 million, representing an ownership interest of less than 10%. Also in October 2006, OctoPlus common
39
stock began trading on an international exchange following an initial public offering of its common stock. With a readily determinable fair market value, the Company now treats the investment in OctoPlus as an available-for-sale investment rather than a cost method investment.
In September 2006, our Board of Directors authorized the repurchase of up to $35 million of the Company’s common stock. During fiscal 2007, the Company repurchased 1,007,752 shares of its common stock for $35.0 million at an average price of $34.76 per share.
named Nexeon MedSystems, Inc. We will continue to account for our investment in Paragon and Apollo under the equity method in the first quarter of fiscal 2009, as both entities report results to us on a one-quarter lag. Commencing with the second quarter of fiscal 2009 we expect to account for our investment in Nexeon under the cost method as our ownership is less than 20%.
statements for further information.
41
statements for further information.
We have a current income tax liability of $6.2 million, which will be paid in December 2007. This payment will include taxes payable on the $20 million up-front license fee received from Merck. Because this license fee will be amortized over 16 years under the EITF 00-21 accounting treatment, the related tax expense has been deferred and will be recognized over the same period. Accordingly, going forward, we will carry a deferred tax asset even though the cash taxes will have been paid.
a fixed expiration date. During fiscal 2008, we purchased 297,900 shares of common stock for $12.7 million at an average price of $42.52 per share. Under the current authorization, the Company has $22.3 million remaining available for authorized share repurchases as of September 30, 2008.
current disruption in the global financial markets may negatively impact the Company’s ability to access the capital markets under current and future sources of financing in a timely manner and on attractive terms. Our anticipated liquidity needs for fiscal 2009 include but are not limited to the following: capital expenditures related to the recently acquired Alabama facility in the range of $24 million to $26 million; general capital expenditures in the range of $3 million to $7 million; contingent consideration payments associated with our November 2008 acquisition of certain assets from PR Pharmaceuticals in the range of $3 million to $6 million; contingent consideration payments related to our acquisitions of Brookwood and BioFX; and any amounts associated with the repurchase of common stock under the authorization discussed above.
40
| Less than | More than | |||||||||||||||||||||||
| Total | 1 Year | 1-3Years | 3-5Years | 5 Years | ||||||||||||||||||||
| Operating leases | $ | 2,344 | $ | 684 | $ | 1,660 | $ | — | $— | |||||||||||||||
| Other liabilities reflected on the | ||||||||||||||||||||||||
| balance sheet under GAAP | 1,000 | 1,000 | — | — | — | |||||||||||||||||||
| Total | $ | 3,344 | $ | 1,684 | $ | 1,660 | $ | — | $— | |||||||||||||||
Less than More than Total 1 Year 1-3 Years 3-5 Years 5 Years Operating leases $ 3,116 $ 1,260 $ 1,567 $ 256 $ 33
42
On July 13, 2006, the Financial Accounting Standards Board (“FASB”) Interpretation (“FIN”) No. 48, Accounting for Uncertainty in Income Taxes - an Interpretation of FASB Statement No. 109, was issued. FIN 48 clarifies the accounting for uncertainty in income taxes recognized in an enterprise’s financial statements in accordance with FASB Statement No. 109, Accounting for Income Taxes. FIN 48 also prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. The new FASB interpretation also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosure, and transition. The provisions of FIN 48 are effective for the Company beginning in fiscal 2008. The Company is currently evaluating the effect that the adoption of FIN 48 will have on its results of operations and financial condition.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK. |
$931,000$951,000 decrease in the fair value of the Company’s available-for-sale and held-to-maturity securities as of September 30, 2007,2008, but no material impact on the results of operations or cash flows. Management believes that a reasonable change in raw material prices would not have a material impact on future earnings or cash flows because the Company’s inventory exposure is not material.41ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA. 2006 and the consolidated statements of operations,income, stockholders’ equity and cash flows for each of the three years in the period ended September 30, 2007,2008, together with Report of Independent Registered Public Accounting Firm and related footnotes (including selected unaudited quarterly financial data) begin onpage F-1 of thisForm 10-K.ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE.
43
None.
| ITEM 9A. | CONTROLS AND PROCEDURES. |
1. Disclosure Controls and Procedures.
| 1. | Disclosure Controls and Procedures. |
| 2. | Internal Control over Financial Reporting. |
44
42
45
43
3. Changes in Internal Controls.
| 3. | Changes in Internal Controls. |
| ITEM 9B. | OTHER INFORMATION. |
44
| ITEM 10. |
| ITEM 11. |
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS. |
45
| (c) | |||||||||||||
| Number of securities | |||||||||||||
| (a) | (b) | remaining available for | |||||||||||
| Number of securities to | Weighted-average exercise | future issuance under | |||||||||||
| be issued upon exercise | price of outstanding | equity compensation | |||||||||||
| of outstanding options, | options, warrants and | plans (excluding securities | |||||||||||
| Plan Category | warrants and rights | rights | reflected in column(a)) | ||||||||||
| Equity compensation plans | |||||||||||||
| approved by shareholders | 1,607,611 | (1) | $27.28 | (1) | 730,491 | (2) | |||||||
| Equity compensation plans not | |||||||||||||
| approved by shareholders | 0 | N/A | 0 | ||||||||||
| Total | 1,607,611 | $27.28 | 730,491 | ||||||||||
2008:
| (c) | ||||||||||||
| Number of Securities | ||||||||||||
| (a) | (b) | Remaining Available for | ||||||||||
| Number of Securities to | Weighted-Average | Future Issuance Under | ||||||||||
| be Issued Upon Exercise | Exercise Price of | Equity Compensation | ||||||||||
| of Outstanding Options, | Outstanding Options, | Plans (Excluding Securities | ||||||||||
Plan Category | Warrants and Rights | Warrants and Rights | Reflected in Column (a)) | |||||||||
| Equity compensation plans approved by shareholders | 1,908,067 | (1) | $ | 34.43 | (1) | 315,486 | (2) | |||||
| Equity compensation plans not approved by shareholders | 0 | N/A | 0 | |||||||||
| Total | 1,908,067 | $ | 34.43 | 315,486 | ||||||||
| (1) | Excludes shares that may be issued under the Company’s 1999 Employee Stock Purchase Plan. | |
| (2) | Includes |
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS.
46
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS. |
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES.
| ITEM 14. | PRINCIPAL ACCOUNTANT FEES AND SERVICES. |
46
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES.
| |
| ||||
| Page (s) | ||||
| F-1 | ||||
| F-2 | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| Statements | ||||
| ||||
47