UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 10-K
(Mark One)
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the fiscal year ended December 31, 20182019
or
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the transition period from to
Commission File Number: 001-33500
JAZZ PHARMACEUTICALS PUBLIC LIMITED COMPANY
(Exact name of registrant as specified in its charter)
| Ireland | 98-1032470 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
Fifth Floor, Waterloo Exchange
Waterloo Road, Dublin 4, IrelandD04 E5W7
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Ordinary shares, nominal value $0.0001 per share | JAZZ | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yesý☒ No ¨☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨☐Noý☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yesý☒ No ¨☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yesý☒ No ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | ☒ | Accelerated filer | ☐ | Non-accelerated filer | ☐ | Smaller reporting company | ☐ | Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨☐ No ý☒
The aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant, as of June 30, 2018,28, 2019, the last business day of the registrant’s most recently completed second fiscal quarter, was approximately $7,515,311,963$7,869,781,340 based upon the last sale price reported for the registrant’s ordinary shares on such date on The Nasdaq Global Select Market. The calculation of the aggregate market value of voting and non-voting common equity excludes 16,768,9831,418,007 ordinary shares of the registrant held by executive officers, directors and shareholders that the registrant concluded were affiliates of the registrant on that date. Exclusion of such shares should not be construed to indicate that any such person possesses the power, direct or indirect, to direct or cause the direction of the management or policies of the registrant or that such person is controlled by or under common control with the registrant.
As of February 19, 201918, 2020, a total of 57,055,73156,133,306 ordinary shares, nominal value $0.0001 per share, of the registrant were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Certain information required by Part III, Items 10-14 of this Form 10-K10‑K is incorporated by reference to the registrant’s definitive Proxy Statement for the 20192020 Annual General Meeting of Shareholders to be filed with the Securities and Exchange Commission pursuant to Regulation 14A not later than 120 days after the end of the fiscal year covered by this Form 10-K,10‑K, provided that if such Proxy Statement is not filed within such period, such information will be included in an amendment to this Form 10-K10‑K to be filed within such 120-day period.
1
JAZZ PHARMACEUTICALS PLC
TABLE OF CONTENTS
| Page | ||
| PART I | ||
| Item 1. | ||
| Item 1A. | ||
| Item 1B. | ||
| Item 2. | ||
| Item 3. | ||
| Item 4. | ||
| PART II | ||
| Item 5. | ||
| Item 6. | ||
| Item 7. | ||
| Item 7A. | ||
| Item 8. | ||
| Item 9. | ||
| Item 9A. | ||
| Item 9B. | ||
| PART III | ||
| Item 10. | ||
| Item 11. | ||
| Item 12. | ||
| Item 13. | ||
| Item 14. | ||
| PART IV | ||
| Item 15. | ||
| Item 16. | ||
We own or have rights to various copyrights, trademarks, and trade names used in our business in the U.S. and/or other countries, including the following: Jazz Pharmaceuticals®, Xyrem® (sodium oxybate) oral solution, ErwinazeSunosi® (asparaginase Erwinia chrysanthemi) (solriamfetol), ErwinaseDefitelio®,Defitelio® (defibrotide sodium), Defitelio® (defibrotide), CombiPlexErwinaze® (asparaginase Erwinia chrysanthemi), Erwinase®, CombiPlex®, Vyxeos® (daunorubicin and cytarabine) liposome for injection and Vyxeos® liposomal 44 mg/100 mg powder for concentrate for solution for infusion and FazaClo® (clozapine, USP).infusion. This report also includes trademarks, service marks and trade names of other companies. Trademarks, service marks and trade names appearing in this Annual Report on Form 10-K10‑K are the property of their respective owners.
2
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K10‑K contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, which are subject to the “safe harbor” created by those sections. Forward-looking statements are based on our management’s beliefs and assumptions and on information currently available to our management. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “could,” “would,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “predict,” “propose,” “intend,” “continue,” “potential,” “possible,” “foreseeable,” “likely,” “unforeseen” and similar expressions intended to identify forward-looking statements. These statements involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance, time frames or achievements to be materially different from any future results, performance, time frames or achievements expressed or implied by the forward-looking statements. We discuss many of these risks, uncertainties and other factors in this Annual Report on Form 10‑K in greater detail under the heading “Risk Factors.” Given these risks, uncertainties and other factors, you should not place undue reliance on these forward-looking statements. Also, these forward-looking statements represent our estimates and assumptions only as of the date of this filing. You should read this Annual Report on Form 10-K10‑K completely and with the understanding that our actual future results may be materially different from what we expect. We hereby qualify our forward-looking statements by our cautionary statements. Except as required by law, we assume no obligation to update our forward-looking statements publicly, or to update the reasons that actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future.
NOTE REGARDING COMPANY REFERENCE
In this report, unless otherwise indicated or the context otherwise requires, all references to “Jazz Pharmaceuticals,” “the registrant,” “we,” “us,” and “our” refer to Jazz Pharmaceuticals plc and its consolidated subsidiaries.
PART I
| Item 1. | Business |
Overview
Jazz Pharmaceuticals plc is a global biopharmaceutical company dedicated to developing life-changing medicines for people with serious diseases – often with limited or no options. As a leader in sleep medicine and with a growing hematology/oncology portfolio, weWe have a diverse portfolio of productsmarketed medicines and novel product candidates, from early- to late-stage development, in development.key therapeutic areas. Our focus is in neuroscience, including sleep medicine and movement disorders, and in oncology, including hematologic and solid tumors.
Our lead marketed products are:
| • | Xyrem® (sodium oxybate) oral solution, the only product approved by the U.S. Food and Drug Administration, or FDA, and marketed in the U.S. for the treatment of both cataplexy and excessive daytime sleepiness, or EDS, in both adult and pediatric patients with narcolepsy; |
| • |
| • | Defitelio® (defibrotide sodium), a product approved in the U.S. for the treatment of adult and pediatric patients with hepatic veno-occlusive disease, or VOD, also known as sinusoidal obstruction syndrome, or SOS, with renal or pulmonary dysfunction following hematopoietic stem cell transplantation, or HSCT, and in Europe (where it is marketed as Defitelio® (defibrotide)) for the treatment of severe VOD in adults and children undergoing HSCT therapy; |
| • | Erwinaze® (asparaginase Erwinia chrysanthemi), a treatment approved in the U.S. and in certain markets in Europe (where it is marketed as Erwinase®) for patients with acute lymphoblastic leukemia, or ALL, who have developed hypersensitivity to E. coli-derived asparaginase; and |
| • | Vyxeos® (daunorubicin and cytarabine) liposome for injection, a product approved in the U.S. and in Europe (where it is marketed as Vyxeos® liposomal 44 mg/100 mg powder for concentrate for solution for infusion) for the treatment of adults with newly-diagnosed therapy-related acute myeloid leukemia, or t-AML, or |
3
commencement of the rolling launch of Sunosi in the U.S. and Europe for solriamfetol as a treatment to improve wakefulness and reduce EDS in adult patients with narcolepsy or obstructive sleep apnea, or OSA. Theby mid-2020. In February 2020, the FDA accepted our solriamfetolfor filing with priority review the new drug application, or NDA, for filinglurbinectedin for the treatment of relapsed small cell lung cancer, or SCLC, a product candidate for which we recently acquired exclusive U.S. development and commercialization rights. In January 2020, we submitted an NDA to the FDA seeking marketing approval for JZP-258, an oxybate product candidate that contains 92%, or approximately 1,000 to 1,500 milligrams per day, less sodium than Xyrem, for the treatment of cataplexy and EDS in narcolepsy patients seven years of age and older. We also have in development JZP-458, a recombinant Erwinia asparaginase product candidate, for the treatment of pediatric and adult patients with ALL or lymphoblastic lymphoma, or LBL, who are hypersensitive to E. coli-derived asparaginase products, and expect to submit a standard review in early 2018 and the current Prescription Drug User Fee Act, or PDUFA, date is March 20, 2019. We submitted a solriamfetol marketing authorizationbiologics license application, or MAA,BLA, to the European Medicines Agency, or EMA, inFDA for JZP-458 as early as the fourth quarter of 2018.
2020.
Our strategy to create shareholder value is focused on:
Strong financial execution through growth in sales of our current lead marketed products;
Building a diversified product portfolio and development pipeline through a combination of our internal research and development efforts and obtaining rights to clinically meaningful and differentiated on- or near-market products and early- to late-stage product candidates through acquisitions, collaborations, licensing arrangements, partnerships and venture investments; and
Maximizing the value of our products and product candidates by continuing to implement our comprehensive global development plans, including through generating additional clinical data and seeking regulatory approval for new indications.indications and new geographies.
In 2018, in support of2019, consistent with our strategy, we continued to expand and advance our research and development pipeline in our sleepsleep/neuroscience and hematology/oncology therapeutic areas, both by conducting activities internally and by leveraging partnerships with third parties. For a summary of our ongoing research and development activities, see “Business—Research and Development” in this Part I, Item 1.
Our Commercialized Products
Xyrem. Xyrem is the only treatmentproduct approved by the FDA and marketed in the U.S. for the treatment of both cataplexy and EDS in both adult and pediatric patients with narcolepsy. Sodium oxybate, the active pharmaceutical ingredient, or API, in Xyrem, is a formulation of the sodium salt of gamma-hydroxybutyrate, an endogenous neurotransmitter and metabolite of gamma-aminobutyric acid.
Narcolepsy is a chronic, debilitating neurological disorder causedcharacterized by a loss of neurons that produceEDS and the neurotransmitter hypocretin (also known as orexin)inability to regulate sleep-wake cycles normally. It affects an estimated one in 2,000 people in the U.S., which is hypothesized to stabilize sleep-wake states. Thewith symptoms typically appearing in childhood. There are five primary symptoms of narcolepsy, includeincluding EDS, cataplexy, disrupted nighttime sleep, paralysis, hypnogogicsleep-related hallucinations, and disrupted nighttime sleep.sleep paralysis. While patients with narcolepsy may not experience all five symptoms, EDS is an essential symptom of narcolepsy, is present in all narcolepsy patients and is characterized by chronic, pervasive sleepiness as well as sudden irresistible and overwhelming urges to sleep (inadvertent naps and sleep attacks). Narcolepsy may affect many areas of life, including limiting a patient’s education and employment opportunities, and may lead to difficulties at work, school, or in daily life activities like driving, operating machinery or caring for children. Patients with narcolepsy may also suffer from significant medical comorbidities, including cardiac disorders, depression, suicide risk, anxiety, diseases of the digestive system and respiratory diseases.
Cataplexy, the sudden loss of muscle tone with retained consciousness, can be one of the most debilitating symptoms of narcolepsy. Cataplexy is present in approximately 70% of patients with narcolepsy. Cataplexy can range from slight weakness or a drooping of facial muscles to the complete loss of muscle tone resulting in postural collapse. It may also impair a patient’s vision or speech. Cataplexy is often triggered by strong emotions such as laughter, anger or surprise. Cataplexy can severely impair a patient’s quality of life and ability to function.
Xyrem was approved in the U.S. for the treatment of cataplexy in adult patients with narcolepsy in 2002 and was approved for EDS in adult patients with narcolepsy in 2005. In October 2018, Xyrem was also approved in the U.S. for the treatment of cataplexy or EDS in pediatric narcolepsy patients ages seven and older. The American Academy of Sleep Medicine recommends Xyrem as a standard of care for the treatment of both cataplexy and EDS associated with narcolepsy.
4
who treat narcolepsy, physician/healthcare provider education, enhanced patient and physician support services related to Xyrem since 2002. Our current agreement with Express Scripts expires on July 1, 2019, but we expect to exercise our right to extend the agreement for an additional year. The agreement may be terminated by either party at any time without cause on 180 days’ prior written notice to the other party. We own all standard operating procedures, business rules and the related intellectual propertyunbranded disease awareness programs for the services Express Scripts provides related to patient support programs. The agreement provides for Express Scripts to assist in the orderly transfer of the services that Express Scripts provides to us and the related intellectual property, including intellectual property related to the patient database, to any new pharmacy that we may engage.public.
Our marketing, sales and distribution of Xyrem in the U.S. are subject to a risk evaluation and mitigation strategy, or REMS, which is required by the FDA to mitigate the risks of serious adverse outcomes resulting from inappropriate prescribing, abuse, misuse and diversion of Xyrem. Under the Xyrem REMS, all of the Xyrem sold in the U.S. must be
dispensed and shipped directly to patients or caregivers through a central pharmacy. Xyrem may not be stocked in retail pharmacies. Physicians and patients must complete an enrollment process prior to fulfillment of Xyrem prescriptions, and each physician and patient must receive materials concerning the serious risks associated with Xyrem before the physician can prescribe, or a patient can receive, the product. The central certified pharmacy must monitor and report instances of patient or prescriber behavior giving rise to a reasonable suspicion of abuse, misuse or diversion of Xyrem, and maintains enrollment and prescription monitoring information in a central database. The central pharmacy ships the product directly to the patient (or caregiver) by a courier service.
In 2018,2019, net product sales of Xyrem were $1.4$1.6 billion, which represented 75%77% of our total net product sales.
OSA, commonly referred to as sleep apnea, is a highly prevalent disease, and EDS, a major symptom of OSA, is characterized by the inability to stay awake and alert during the day resulting in unplanned lapses into sleep or drowsiness. Although positive airway pressure therapy, with its most common form being continuous positive airway pressure, or CPAP, has been shown to be an effective therapy for Health. PBL also manufactures the product for ussleep apnea that frequently results in improvement in EDS in many patients, not all patients tolerate CPAP therapy and among those who tolerate CPAP, usage is our sole supplier for Erwinaze. We are obligated to make tiered royalty payments to PBL based on worldwide net sales of Erwinaze. Our license and supply agreementhighly variable. EDS may persist in people with PBL, which includes our license for Erwinaze, expires on December 31, 2020. We and PBL had been engaged in discussions related to entry into a replacement agreement to extend the term of our commercial relationship with respect to Erwinaze past 2020, but we did not reach agreement. Unless we and PBL enter into a new agreement, we will lose our license to Erwinaze after December 31, 2020, other than our right to sell certain Erwinaze inventory for a post-termination sales period of 12 months. Either party also has the right to terminate the agreement prior to its expiration in the event of the other party’s uncured material breach or insolvency.
In 2018,2019, net product sales of ErwinazeSunosi were $174.7 million, which represented 9% of our total net product sales.$3.7 million.
Defitelio. Defibrotide, the API in Defitelio, has been approved for the treatment of VOD, a potentially life-threatening complication of HSCT, and is in development for other complications following HSCT, including prevention of VOD, prevention of acute Graft versus Host Disease, or aGvHD, and treatment of transplant-associated thrombotic microangiopathy, or TA-TMA, as well as complications following anti-cancer treatment, including prevention of CAR-T-associatedchimeric antigen receptor T-cell, or CAR T-cell, therapy-associated neurotoxicity. Defibrotide is the sodium salt of a complex mixture of single-stranded oligodeoxyribonucleotides derived from porcine DNA. Defibrotide mediates its effects via interaction with endothelial cells. Non-clinical data suggest that defibrotide stabilizes endothelial cells by reducing endothelial cell activation and by protecting them from further damage.
Stem cell transplantation is a frequently used treatment modality for hematologic cancers and other conditions in both adults and children. Certain conditioning regimens used as part of HSCT can damage the cells that line the hepatic vessels, which is thought to lead to the development of VOD, also referred to as SOS, a blockage of the small vessels in the liver, that can lead to liver failure and potentially result in significant dysfunction in other organs such as the kidneys and lungs. Severe VOD is the most extreme form of VOD and is associated with multi-organ failure and high rates of morbidity and mortality.
An analysis of retrospective data, prospective cohort studies and clinical trials published between 1979 and 2007 found that the 100-day mortality rate in severe VOD cases is greater than 80%.
The European Commission, or EC granted marketing authorization under exceptional circumstances for Defitelio for the treatment of severe VOD in adults and children undergoing HSCT in 2013. We commenced a rolling launch of Defitelio in European countries in 2014. In countries where we currently commercialize Defitelio, we are working to maintain current levels of market access.
5
In 2016, the FDA approved our NDA for Defitelio for the treatment of adult and pediatric patients with VOD with renal or pulmonary dysfunction following HSCT. We launched Defitelio in the U.S. shortly after FDA approval. We acquired the rights to defibrotide for the treatment and prevention of VOD in North America, Central America and South America from Sigma-Tau Pharmaceuticals, Inc., or Sigma-Tau, in 2014. In exchange for these rights, we made an upfront payment of $75.0 million to Sigma-Tau and also made milestone payments of $175.0 million comprised of (i) $25.0 million upon the acceptance for filing by the FDA of the first NDA for defibrotide for VOD, paid in the fourth quarter of 2015; and (ii) an additional final payment of $150.0 million upon FDA approval of defibrotide for VOD, paid in the second quarter of 2016.
In 2018,2019, Defitelio/defibrotide product sales were $149.4$172.9 million, which represented 8% of our total net product sales.
For ALL patients with hypersensitivity to E. coli-derived asparaginase, Erwinaze can be a crucial component of their therapeutic regimen. Current treatment guidelines and protocols recommend switching a patient receiving E. coli-derived asparaginase to treatment with Erwinaze if the patient’s hypersensitivity reaction to the E. coli-derived asparaginase is clinically meaningful, indicating that the hypersensitivity reaction has resulted in an intervention or interruption in infusion occurring in the patient’s treatment regimen. While treatment protocols for pediatric, adolescent and young adult (up to age 39) patients commonly include asparaginase, adult protocols do not.
First approved by the FDA under a BLA for administration via intramuscular injection in conjunction with chemotherapy, Erwinaze was launched in the U.S. in 2011. In 2014, the FDA approved a supplemental BLA for administration of Erwinaze via intravenous infusion in conjunction with chemotherapy.
Erwinaze is exclusively licensed to us for worldwide marketing, sales and distribution by Porton Biopharma Limited, or PBL, a company that is wholly owned by the UK Department of Health and Social Care. PBL also manufactures the product for us and is our sole supplier for Erwinaze. We are obligated to make tiered royalty payments to PBL based on worldwide net sales of Erwinaze. Our license and supply agreement with PBL, which includes our license to Erwinaze trademarks and manufacturing know-how, expires on December 31, 2020. Unless we and PBL enter into a new agreement, we will lose our rights to exclusively market Erwinaze effective December 31, 2020, other than our right to sell certain Erwinaze inventory for a post-termination sales period of 12 months and certain other post-termination rights, including but not limited to intellectual property and data ownership. Either party also has the right to terminate the agreement prior to December 31, 2020 in the event of the other party’s uncured material breach or insolvency.
In 2019, net product sales of Erwinaze were $177.5 million, which represented 8% of our total net product sales.
Vyxeos. Vyxeos is a liposomeliposomal formulation of a fixed ratio combination of daunorubicin and cytarabine for intravenous infusion that is indicated for the treatment of adults with newly-diagnosed t-AML or AML-MRC and has been shown to have synergistic effects at killing leukemia cells in vitro and in animal models. Vyxeos is the first drug delivery combination product based on our CombiPlex technology platform to be approved by the FDA and the EC.
AML is a rapidly progressing and life-threatening blood cancer that begins in the bone marrow, which produces most of the body's new blood cells. AML cells crowd out healthy cells and move aggressively into the bloodstream to spread cancer to other parts of the body. AML is a relatively rare disease representing about 1% of all new cancer cases and has the lowest survival rate of any form of leukemia. Patients with newly diagnosed t-AML or AML-MRC may have a particularly poor prognosis.
In 2017, we launched Vyxeos in the U.S. after the FDA approved our NDA for the treatment of adults with newly-diagnosed t-AML or AML-MRC. In August 2018, the EC granted marketing authorization for Vyxeos, and, shortly thereafter, we commenced aas part of our rolling launch of Vyxeos in theEurope, we are continuing to make pricing and reimbursement submissions in European Union, or EU.
In 2018,2019, Vyxeos product sales were $100.8$121.4 million, which represented 5%6% of our total net product sales.
Research and Development
A key aspect of our growth strategy is our continued investment in our evolving and expanding research and development pipeline.activities. We actively explore new options for patients including novel compounds, small molecule advancements, biologics and innovative product candidates ranging from small molecules to biologics.delivery technologies. While we are primarily focused on opportunities within our sleepsleep/neuroscience and hematology/oncology therapeutic areas, such as our recent expansion into movement disorders and solid tumors, we are also interestedexploring and investing in and exploring adjacent therapeutic areas including central nervous system disorders and solid tumors.that could further diversify our portfolio.
6
Our development activities encompass all stages of development and currently include clinical testing of new product candidates and activities related to clinical improvements of, or additional indications or new clinical data for, our commercializedexisting marketed products. We have also recently expanded into preclinical exploration of novel therapies, including precision medicines in hematology and oncology. We conduct most of these activities by leveraging our growing internal research and development function, but we have also entered into collaborations with third parties for the research and development of early-
Our current and planned development activities in our sleep and neuroscience therapeutic area are primarily focused on two product candidates, solriamfetolJZP-258, JZP-324 and JZP-258.
JZP-258. JZP-258 is an oxybate product candidate that contains 90%with a unique composition of cations resulting in 92%, or approximately 1,000 to 1,500 milligrams per day, less sodium than Xyrem and is being developedXyrem. In January 2020, we submitted an NDA for JZP-258 for the potential treatment of both cataplexy and EDS in patients with narcolepsy and potential treatment of idiopathic hypersomnia,in connection with this submission, redeemed the priority review voucher, or IH, a chronic neurological disorder that is primarily characterized by EDS.PRV, we acquired in May 2018. Given the well-accepted relationship between dietary sodium and blood pressure as well as published hypertension guidelines underscoring that excessive consumption of sodium is independently associated with an increased risk of stroke, cardiovascular disease and other adverse outcomes, we believe that the lower sodium intake would be beneficial for patients and thereforecontent of JZP-258 wouldhas the potential to offer a clinically meaningful benefit to patients compared to Xyrem. We have conductedare also conducting a Phase 3 clinical trial of JZP-258 in patients for the treatment of both cataplexyidiopathic hypersomnia, a chronic neurological disorder that is primarily characterized by EDS and EDS in narcolepsy and, subject to the results of our Phase 3 clinical trial, we expect to submit an NDA by as early as the end of 2019. We are conducting a Phase 3 clinical trial for the treatment of IH, which currently has no approved therapies in the U.S.
JZP-324. We are also pursuing early-stage activities related to the potential development of an extended releaseJZP-324, a low sodium, oxybate formulation with the potential for once-nightly dosing that we believe could provide a clinically meaningful option for some narcolepsy patients.
JZP-385. JZP-385 is a T-type calcium channel modulator that is a small molecule currently in development for the treatment of essential tremor. We acquired JZP-385 in our acquisition of Cavion, Inc., or Cavion, a clinical-stage biotechnology company, in August 2019. We expect to initiate a Phase 2b study of JZP-385 in the fourth quarter of 2020.
Our current and planned development activities in our hematology/hematology and oncology therapeutic area are focused on JZP-458, lurbinectedin and exploring additional indications for Defitelio and Vyxeos, generating additional clinical data for Vyxeos, including in combination with other therapeutic agents, and the research and development of new product candidates.
JZP-458. JZP-458 is a recombinant Erwinia asparaginase that uses a novel Pseudomonas fluorescens expression platform, which is being developed for use as a component of a multi-agent chemotherapeutic regimen in the treatment of pediatric and adult patients with ALL or LBL, who are hypersensitive to E. coli-derived asparaginase products. JZP-458 was granted Fast Track designation by the FDA in October 2019 for the treatment of this patient population, and in December 2019, the first patient was enrolled in the pivotal Phase 2/3 clinical study for JZP-458 conducted in collaboration with the Children’s Oncology Group. We expect to submit a BLA to the FDA for JZP-458 as early as the fourth quarter of 2020.
Lurbinectedin. In furtherance of our interest in and efforts to expand our oncology therapeutic area, in December 2019, we entered into an exclusive license agreement with Pharma Mar, S.A., or PharmaMar, pursuant to which we obtained exclusive U.S. development and commercialization rights to lurbinectedin, a product candidate under clinical investigation for the treatment of patients with relapsed SCLC. Lurbinectedin was granted orphan drug designation for SCLC by the FDA in August 2018, and PharmaMar submitted an NDA to the FDA in December 2019 for accelerated approval of lurbinectedin for relapsed SCLC based on data from a Phase 2 trial. In February 2020, the FDA accepted the NDA for filing with priority review with a Prescription Drug User Fee Act, or PDUFA, date of August 16, 2020. The term of our license agreement with PharmaMar extends until the latest of: (i) expiration of the last PharmaMar patent covering lurbinectedin in the U.S. (subject to certain exclusions), (ii) expiration of regulatory exclusivity for lurbinectedin in the U.S. and (iii) 12 years after the first commercial sale of lurbinectedin in the U.S. We also have the right to terminate the agreement at will upon a specified notice period, provided that the effective date of such termination is not within one year of the signing of the agreement. Either party can terminate the agreement for the other party’s uncured material breach or bankruptcy.
Defitelio. Our Defitelio clinical development strategy generallygenerally focuses on the prevention and treatment of serious diseases associated with stem cell transplantation and endothelial cell damage. In addition to clinical trials we are sponsoring, there are more than 20 investigator-sponsored trials ongoing in the U.S. and EU to evaluateevaluating defibrotide in multiple conditions.
Vyxeos. Our Vyxeos clinical development strategy is designed to target potential new patient segments across the AML landscape, to pursue indications related to myelodysplastic syndrome, or MDS,landscape and to generate clinical data on Vyxeos when used in combination with other therapeutic agents. As reflected in the table below, we are pursuing this strategy by sponsoring clinical trials, working with cooperative groups who are conducting clinical trials, and partnering with The University of Texas MD Anderson Cancer Center, or MD Anderson. In August 2018, we announced a five-year collaboration with MD Anderson to evaluate potential treatment options for hematologic malignancies,
7
with a near-term focus on Vyxeos, and shortly thereafter, commenced development activities under this collaboration. In addition, there are multiple ongoing investigator-sponsored trials studying Vyxeos.
CombiPlex Platform. We are also evaluating the use of our CombiPlex delivery technology platform in a number of therapeutic combinations in oncology as part of our internal oncology research and development activities. CombiPlex enables the design and rapid evaluation of various combinations of therapies to deliver enhanced anti-cancer activity by identifying an optimal synergistic ratio of drugs in vitro and fixing this ratio in a nanoscale delivery complex that maintains and then coordinates the release of the synergistic combination after administration. CombiPlex utilizes two proprietary nanoscale delivery platforms: liposomes to control the release and distribution of water-soluble drugs and drugs that are both water- and fat-soluble (amphipathic), and nanoparticles to control the release and distribution of non-water-soluble (hydrophobic) drugs. We are conducting preclinical activities using our CombiPlex platform to develop a novel combination of therapeutic agents that are designed to target an undisclosed solid tumor candidate, as well as other hematology/oncology exploratory activities.
Through third parties, we are also pursuing preclinical and clinical research and development activities in hematology and in precision oncology under a number of licensing and collaboration agreements, including with:
ImmunoGen, Inc., or ImmunoGen, for opt-in rights to license twoa hematology-related antibody-drug conjugate or ADC, product candidatescandidate granted orphan drug designation by the FDA, as well as an additional ADC product candidate;
FDA;
Codiak BioSciences, Inc., or Codiak, for an exclusive, worldwide, royalty-bearing license to develop, manufacture and commercialize potential therapeutic candidates directed at five targets to be developed using Codiak's engEx™ precision engineering platform for exosome therapeutics;
| • | Pfenex, Inc., or Pfenex, for rights to an early-stage long-acting Erwinia asparaginase and an option to negotiate a license for a recombinant pegaspargase product candidate; |
| • | XL-protein GmbH, or XLp, for rights to use XLp’s PASylation® technology to extend the plasma half-life of selected asparaginase product |
Redx Pharma, or Redx, for pre-clinical collaboration activities related to the pan-RAF inhibitor program that we purchased from Redx for the potential treatment of RAF and RAS mutant tumors.
Below is a summary of our key ongoing and planned development projects related to our products and pipeline and their corresponding current stages of development:
Sleep and Neuroscience
| Product Candidates | Description | |
| Submitted for Regulatory Approval | ||
| JZP-258 (oxybate; | Cataplexy and EDS in narcolepsy | |
| Idiopathic hypersomnia | ||
| Sunosi | EDS in | |
| Phase 2b | ||
| JZP-385 | Essential tremor (planned study) | |
| Preclinical | ||
| JZP-324 | Oxybate once-nightly formulation | |
| Product Candidates | Description |
| Lurbinectedin | Relapsed SCLC (exclusive U.S. license) |
| Phase 3 | |
| Defitelio | Prevention of VOD in high- and very high-risk patients following HSCT |
| Vyxeos | AML or high-risk Myelodysplastic Syndrome, or MDS |
| Vyxeos | Newly diagnosed adults with standard- and high-risk AML (AML Study Group cooperative group study) |
| Vyxeos | Newly diagnosed pediatric patients with AML |
| Relapsed SCLC (ATLANTIS) (exclusive U.S. license) | |
8
| Product Candidates | Description |
| Phase 2/3 | |
JZP-458 (recombinant Erwinia asparaginase) | ALL/LBL |
| Phase 2 | |
| Defitelio | Prevention of aGvHD following allogeneic HSCT |
| Defitelio | |
| Prevention of | |
| Vyxeos + venetoclax | De novo or relapsed/refractory, or R/R, AML (MD Anderson collaboration study) |
| Vyxeos | High-risk MDS (European Myelodysplastic Syndromes Cooperative Group cooperative group study) |
| Vyxeos | Newly diagnosed older adults with high-risk AML (planned cooperative group study) |
| Vyxeos + venetoclax | |
| Vyxeos + gemtuzumab | R/R AML or hypomethylating agent failure MDS (MD Anderson collaboration study) |
| Vyxeos + venetoclax | Low intensity |
| Vyxeos | Low intensity dosing for higher risk MDS (MD Anderson collaboration study) |
| IMGN632 | R/R CD123+ hematological malignancies (Jazz opt-in opportunity with ImmunoGen) |
| IMGN632 +/- venetoclax/azacitidine | CD123+ |
| Preclinical | |
| CombiPlex | Solid tumors candidate |
| CombiPlex | Hematology/oncology exploratory activities |
JZP-341 (long-acting Erwinia asparaginase) | ALL and other hematological malignancies (collaboration with Pfenex) |
| Recombinant | Hematological malignancies (Jazz opt-in opportunity with Pfenex) |
| Defitelio | Exploratory activities |
| Exosome NRAS candidate | Hematological malignancies (collaboration with Codiak) |
| Exosome STAT3 candidate | Hematological malignancies (collaboration with Codiak) |
| Exosome-based candidates | Solid tumors/hematological malignancies (collaboration with Codiak) |
| Pan-RAF inhibitor program | RAF and RAS mutant tumors (acquired from Redx, which is continuing development) |
In 20192020 and beyond, we expect that our research and development expenses will continue to increase from historicalprevious levels, particularly as we prepare for anticipated regulatory submissions and trial data read-outs from clinical trials, initiate and undertake additional clinical trials and related development work and potentially acquire rights to additional product candidates.
We have commercial operations primarily in the U.S., Europe and Canada. In the U.S., our products are marketedcommercialized through our commerciala number of teams, including a team of experienced, trained sales professionals who provide education and promote Xyrem, Sunosi, Defitelio, Erwinaze Defitelio and Vyxeos directly to physicianshealthcare providers in the appropriate specialties appropriate for each product.product, a team that interacts with payors and institutions to ensure access and coverage for the products, and a team that distributes the products throughout the U.S. healthcare system (wholesalers, pharmacies, hospitals, and community and academic institutions) and provides patient services.
In Canada and in approved markets in Europe where we commercialize Erwinaze, Defitelio, Erwinase and Vyxeos, we have a field force of hematology field specialists comprised of sales personnel and medical affairs personnel.specialists. In markets where these products either are not approved or are unable to be promoted under
9
local regulation, we have medical science liaisons and medical directorsaffairs personnel responsible for responding to medical information requests and for providing information consistent with local treatment protocols with respect to such products. In certain European markets, we have a team of medical science liaisons in preparation for our rolling launch of Sunosi in Europe. We are currently in the process of recruiting a sales team in some of those markets. Outside the U.S., we directly market Xyrem in Canada for the treatment of cataplexy in patients with narcolepsy. We also utilize distributors in certain markets outside the U.S. where we do not market our products directly.
Our commercial activities include marketing related services, distribution services and commercial support services. We employ third party vendors, such as advertising agencies, market research firms and suppliers of marketing and other sales support-related services, to assist with our commercial activities. We also provide reimbursement support for our U.S. markets.
Competition
The biopharmaceutical industry is highly competitive. Our products compete, and our product candidates may in the future compete, with currently existing therapies, product candidates currently under development by us and others and/or future product candidates, including new chemical entities that may turn out to be safer, more effective or more effectiveconvenient than our products. Any products that we develop may be commercialized in competitive markets, and our competitors, which include large global pharmaceutical companies and small research-based companies and institutions, may succeed in developing products that render our products obsolete or noncompetitive.
In particular, our products and most advanced product candidates face or may face competition as described below:
| • | Xyrem. While Xyrem is currently the only product approved by the FDA and marketed in the U.S. for the treatment of both cataplexy and EDS in both adult and pediatric patients with narcolepsy, we and others have launched products to treat EDS in narcolepsy and may in the future launch products to treat cataplexy in narcolepsy that are competitive with or disrupt the market for Xyrem. In the future, we expect Xyrem to face competition from authorized generic and generic versions of sodium oxybate. For a description of generic versions of sodium oxybate and/or new products for treatment of cataplexy and/or EDS that could compete with, or otherwise disrupt the market for, Xyrem, as well as a description of our settlement agreements with abbreviated new drug application, or ANDA, filers, see the risk factor under the heading “The introduction of new products in the U.S. market that compete with, or otherwise disrupt the market for, our oxybate products and product candidates would adversely affect sales of our oxybate products and product candidates” in Part I, Item 1A of this Annual Report on Form 10-K. |
In addition to generic competition, Xyrem may face competition in the U.S.future from our JZP-258 product candidate, if approved, as well as from other new sodium oxybate formulations for the treatment of both cataplexy and EDS in patients with narcolepsy, we and others may launch products as treatment options in cataplexy and/or EDS in narcolepsy, including other branded sodium oxybate products and other new and existing branded market entrants. In addition, Xyrem will face competition from generics and authorized generics.
Avadel Pharmaceuticals plc or Avadel, is conducting a Phase 3 clinical trial of a once-nightly formulaformulation of sodium oxybate which uses its proprietary technology for the treatment of EDS and cataplexy in patients with narcolepsy.narcolepsy, and that Avadel has indicated that it intends to seek approval using an NDA approval pathway under Section 505(b)(2) ofand referencing the Federal Food, Drug and Cosmetic Act, or FDCA, by referencing Xyrem and relying, to some degree, on the FDA’s approval of Xyrem and related determinations of safety and efficacy.efficacy data for Xyrem. Xyrem may also face competition from new branded entrants to treat EDS in narcolepsy such as pitolisant. Other companies may develop a sodium oxybate or similarhave announced that they have product using, for example, an alternative formulation or a different delivery technology, and seek approvalcandidates in various phases of development to treat the U.S. under Section 505(b)(2) or otherwise. For a further descriptionsymptoms of the approval process for ANDAs and NDAs under the FDA’s Section 505(b)(2) approval pathway, see “Business—Government Regulation—Marketing Exclusivity—The Hatch-Waxman Act” in this Part I, Item 1.narcolepsy, such as Axsome Therapeutics, Inc.’s reboxetine.
10
| • | Sunosi. Sunosi faces competition from existing branded and generic products that treat EDS or improve wakefulness in adult patients with narcolepsy or OSA in a competitive retail pharmacy market. To successfully commercialize Sunosi, we need to differentiate Sunosi from other branded and generic products that treat EDS in patients with narcolepsy, including stimulants, wake-promoting agents, such as Provigil and Nuvigil, and generic versions of stimulants and wake-promoting agents. We are also aware that stimulants are prescribed off-label for patients to treat excessive sleepiness in OSA. Like Xyrem, Sunosi may face competition from new branded entrants such as pitolisant, a drug that was approved by the FDA in August 2019 for the treatment of EDS in adult patients with narcolepsy and became commercially available in the U.S. in the fourth quarter of 2019, and that has also been approved and marketed in Europe to treat adult patients with narcolepsy with or without cataplexy. Sunosi may also face competition from other products in development as potential treatments for EDS in patients with narcolepsy or OSA. |
| • | JZP-258. We expect that, if approved, JZP-258 will face competition similar to that described above for Xyrem, including from new branded entrants in narcolepsy and/or from generic or authorized generic sodium oxybate products. |
| • | Defitelio. While there is currently no direct competition to Defitelio to treat severe VOD, changes in the types of conditioning regimens used as part of HSCT may affect the incidence of VOD diagnosis and demand for Defitelio. |
| • | Erwinaze.While there is currently no direct competition to Erwinaze to treat ALL patients with hypersensitivity to E. coli-derived asparaginase, we and other companies have developed or are developing new treatments for ALL. Some new asparaginase treatments could reduce the rate of hypersensitivity in patients with ALL, and new treatment protocols are being developed and approved for ALL that may not include asparaginase-containing regimens, including some for the treatment of relapsed or refractory ALL patients. We have experienced frequent intermittent shortages of the product that have impacted prescribing habits for Erwinaze, including prescribers’ use of alternate methods to address hypersensitivity reactions. As a biologic product, Erwinaze also faces potential competition from biosimilar products. |
| • | Vyxeos. With respect to Vyxeos, there are a number of alternative established therapies in AML. A key consideration in the treatment of AML patients is the patient’s suitability for chemotherapy. The AML patient population studied in the Vyxeos Phase 3 clinical trial supporting our NDA included 60-75 year old fit patients, or those deemed able to tolerate intensive induction chemotherapy. Prior to Vyxeos, the most widely recognized option for the treatment of newly-diagnosed t-AML and AML-MRC in fit patients was cytarabine in combination with daunorubicin, known as 7+3, which is still used today in this population, along with other intensive chemotherapy regimens, particularly in patients under the age of 60. Also, since Vyxeos was approved, several other products have been approved by the FDA or are in development as treatment options for newly diagnosed AML patients eligible for intensive chemotherapy, such as targeted agents (e.g. midostaurin, enasidenib and ivosidenib), immunotherapies (e.g., gemtuzumab ozogamicin and CAR-T‑cell therapy), and agents disrupting leukemia cell survival (e.g., glasdegib). We are also aware of the increasing use of venetoclax combined with either a hypomethylating agent or low-dose cytarabine, a treatment approved by the FDA in newly diagnosed AML patients who are age 75 years or older, or who have comorbidities that preclude use of intensive induction chemotherapy. |
| • | Lurbinectedin. We expect that, if approved, lurbinectedin will face competition from topotecan, which is currently the only approved treatment in second line SCLC in the U.S., as well as other regimens for relapsed SCLC currently recommended in compendia guidelines. There are also a number of products and immunotherapies in development for the treatment of second line SCLC in various phases of development. |
An important part of our corporate strategy is to build a diversified product pipeline, including by acquiring or in-licensing and developing, or partnering to license and develop, additional products and product candidates that we believe are highly differentiated and have significant commercial potential. Our ability to continue to grow our product portfolio requires that we compete successfully with other pharmaceutical companies, many of which may have substantially greater financial sales and marketing resources, to acquire or in-license products and product candidates. These competitors include established companies that may have a competitive advantage over us due to their size and financial resources.
Customers
In the U.S., our lead marketed product, Xyrem is sold to one specialty pharmacy, a subsidiary of Express Scripts,ESSDS, which ships Xyrem directly to patients. Also in the U.S., Sunosi is distributed through a retail channel consisting of numerous distributors who sell Sunosi to retail pharmacies. Defitelio, Erwinaze Defitelio and Vyxeos are sold to hospital customers through subsidiary specialty distributors of McKesson Corporation, or McKesson. We have distribution services agreements made in the ordinary course of business with McKesson and a pharmacy services agreement with Express ScriptsESSDS that provides for the distribution of Xyrem to patients. For more information
11
regarding our relationship with Express Scripts,ESSDS, see “Business—Our Commercialized Products” in this Part I, Item 1. Purchases are made on a purchase order basis.
In certain countries in Europe, Defitelio, Erwinase Defitelio and Vyxeos are sold pursuant to marketing authorizations. We distribute these products through Durbin PLC, a UK-based wholesaler and distributor, and O&M Movianto Nederland BV, our centralized European logistics services provider, to hospitals and local wholesalers in Europe where we market these products directly and, in other markets in Europe and elsewhere where we do not market these products directly, to local distributors and wholesalers. In countries where there is no marketing authorization, Defitelio, Erwinase Defitelio and Vyxeos are sold pursuant to named patient programs, temporary use authorizations or similar authorizations.
We directly market Xyrem in Canada for the treatment of cataplexy in patients with narcolepsy. Xyrem is also sold in 2122 countries by UCB Pharma Limited, or UCB (which has rights to market Xyrem in 54 countries).
Information on our total revenues by product and revenues attributed to customers who represented at least 10% of our total revenues in each of 2019, 2018 2017 and 20162017 is included in Note 17,18, Revenues, of the Notes to Consolidated Financial Statements included in Part IV of this Annual Report on Form 10-K.10‑K.
Manufacturing
We have a manufacturing and development facility in Athlone, Ireland where we manufacture Xyrem and certain development-stage oxybate product candidates, including JZP-258.candidates. We also have a manufacturing plant in Italy where we produce the defibrotide drug substance. Other than these two facilities, we currently do not have our own commercial manufacturing or packaging capability for our other products, and product candidates or their APIs. As a result, our ability to develop and supply products in a timely and competitive manner depends primarily on our third party suppliers in most cases single source suppliers, being able to meet our ongoing commercial and clinical trial needs.needs for API, other raw materials, packaging materials and finished products.
Lead Marketed Products
Xyrem. Xyrem is manufactured by us in our Athlone facility and by Patheon Pharmaceuticals Inc., which we refer to together with its affiliates as Patheon, under a Master Manufacturing Services Agreement, or the Patheon Agreement, entered into with Patheon in 2015. The Patheon Agreement establishes the general terms and conditions pursuant to which Patheon provides manufacturing services for multiple drug products, including Xyrem, as specified by us in product agreements we enter into from time to time. Although weWe manufacture Xyrem in our Athlone facility we expect tofor most of our U.S. commercial supply and rely on Patheon as our sole U.S.-based supplier ofto supply Xyrem for the foreseeable future. However,other markets, though we are not required to purchase Xyrem exclusively from Patheon. The current term of the Patheon Agreement expireswill expire on December 31, 2020 and may be extended for additional two-year terms2022, subject to further automatic two-yearly extensions if Patheon is then providing manufacturing services for any product, unless either party provides 18 months’ prior notice of termination.termination at least 18 months prior to the end of the then current term. In addition, we may terminate the Patheon Agreement for any reason upon 12 months’ prior written notice, and each party has the right to terminate the agreement in the event of the other party’s uncured material breach.
Siegfried USA, LLC and its European affiliates, or Siegfried, suppliessupply sodium oxybate, the API of Xyrem, to Patheon and through a Siegfried affiliate in Europe, to our Athlone facility. Although Siegfried has been our only supplier of sodium oxybate since 2012, we have the right to purchase a portion of our worldwide requirements of sodium oxybate from other suppliers. Under our agreement, entered into in 2010, we provide periodic rolling forecasts to Siegfried, and a portion of each rolling forecast constitutes a firm purchase order. The agreement with Siegfried expires in April 2021,2024, subject to automatic three-year extensions until either party provides notice to the other of its intent to terminate the agreement at least 18 months before the end of the then-current term. Each party also has the right to terminate the agreement in the event of the other party’s uncured material breach or insolvency. During the term of the agreement and, under certain circumstances for 18 months after the agreement terminates, Siegfried is not permitted to manufacture sodium oxybate for any other company.
Xyrem is a Schedule III controlled substance in the U.S., and the API of Xyrem is the sodium salt of gamma-hydroxybutyric acid, which is a Schedule I controlled substance in the U.S. As a result, Xyrem is subject to regulation by the U.S. Drug Enforcement Administration, or DEA, under the Controlled Substances Act, or CSA. As a result,CSA, and its manufacturing and distribution are highly restricted. Quotas from the DEA are required in order to manufacture and package sodium oxybate and Xyrem in the U.S. For information related to DEA quota requirements, see “Business—Government Regulation—Other Post-Approval Pharmaceutical Product Regulation—Controlled Substance Regulations” in this Part I, Item 1.
12
Erwinaze. Erwinaze is exclusively licensed to us, and manufactured for us, by PBL, which is our sole supplier for Erwinaze. Our license and supply agreement with PBL, which includes an exclusive right to market, sell or distribute Erwinaze, an exclusive license to Erwinaze trademarks, and a non-exclusive license to PBL’s manufacturing know-how, will expire on December 31, 2020. For information related to our agreement with PBL, see “Business—Our Commercialized Products—Erwinaze” in this Part I, Item 1.
A continuing and significant challenge to maintaining sales of Erwinaze and a barrier to increasing sales is PBL’s inability to consistently supply product that meets specifications in quantities that are adequate to meet market demand. We experienced limited product availability of Erwinaze and supply disruptions globally in 2019 and may experience continued supply disruptions in 2020. Such supply instability will continue to adversely impact our ability to generate sales of and revenues from Erwinaze and our business, financial condition, results of operations and growth prospects could be materially adversely affected. For a more complete description of supply issues related to Erwinaze, see the risk factor under the heading “Delays or problems in the supply of our products for sale or for use in clinical trials, loss of our single source suppliers or failure to comply with manufacturing regulations could materially and adversely affect our business, financial condition, results of operations and growth prospects” in Part I, Item 1A of this Annual Report on Form 10‑K.
Vyxeos. Vyxeos is manufactured by Baxter Oncology GmbH, or Baxter, which is a sole source supplier from a single site location, using our CombiPlex technology platform. CombiPlex products represent formulations with increased manufacturing complexities associated with producing drug delivery vehicles encapsulating two or more drugs that are maintained at a fixed ratio and, in the case of Vyxeos, two drugs that are co-encapsulated in a freeze-dried format. There have been batch failures at Baxter due to mechanical, component and other issues, and batches have been produced that have otherwise not been in compliance with applicable specifications. We are continuing to work with Baxter to address manufacturing complexities. Our manufacturing agreement with Baxter expires in August 2022, subject to automatic three-year renewal terms, unless terminated by either party 24 months prior to the end of the initial term or any renewal term. Each party has the right to terminate the agreement for breach, subject to customary cure periods, and each party may terminate the agreement immediately in the event of the other party’s insolvency. While other contract manufacturers may be able to produce Vyxeos, the proprietary technology that supports the manufacture of Vyxeos is not easily transferable. The marketing authorization in the European Union, or EU, for Vyxeos also requires us to comply with certain manufacturing-related post-approval commitments.
Product Candidates
JZP-258 is currently manufactured at our Athlone facility, and we expect to manufacture this product commercially at our Athlone facility should this candidate proceed through development and receive regulatory approval.
Clinical development supply of lurbinectedin is currently manufactured by Baxter and GP Pharm S.A., and its API is manufactured by PharmaMar. PharmaMar retains manufacturing rights for the API for potential future U.S. commercial supply of lurbinectedin. If lurbinectedin is approved by the FDA, launch quantities of lurbinectedin and ongoing supply of the API will be supplied to us by PharmaMar under a separate agreement to be entered into between us and PharmaMar. We also expect to enter into a manufacturing agreement for ongoing commercial supply of the drug product lurbinectedin with Baxter, GP Pharm S.A. and/or another comparable manufacturer.
JZP-458 is currently manufactured by Patheon, and the API of JZP-458 is manufactured by AGC Biologics A/S.
For further discussion of the challenges we face with respect to supply of our products and product candidates, see the risk factor under the heading “Delays or problems in the supply of our products for sale or our for use in clinical trials, loss of our single source suppliers or failure to comply with manufacturing regulations could materially and adversely affect our business, financial condition, results of operations and growth prospects” in Part I, Item 1A of this Annual Report on Form 10‑K.
13
Patents and Proprietary Rights
We actively seek to patent, or to acquire or obtain licenses to third party patents, to protect our products and product candidates and related inventions and improvements that we consider important to our business. We own a portfolio of U.S and non-U.S. patents and patent applications and have licensed rights to a number of issued patents and patent applications. Our owned and licensed patents and patent applications cover or relate to our products and product candidates, including certain formulations, used to treat particular conditions, distribution methods and methods of administration, drug delivery technologies and delivery profiles and methods of production.making and use. Patents extend for varying periods according to the date of the patent filing or grant and the legal term of patents in the various countries where patent protection is obtained. The patent laws of non-U.S. countries differ from those in U.S., and the degree of protection afforded by non-U.S. patents may be different from the protection offered by U.S. patents. In addition to patents, our products and product candidates are in some instances protected by various marketingregulatory exclusivities. For a description of those exclusivities and their regulatory background, see “Business—Government Regulation—Marketing Exclusivity—The Hatch-Waxman Act” in this Part I, Item 1 and in the risk factor under the heading “It is difficult and costly to protect our proprietary rights, and we may not be able to ensure their protection” in Part I, Item 1A of this Annual Report on Form 10-K.
1.
The patents, patent applications and marketingregulatory exclusivities that relate to our marketed products and product candidates include:
| • | Xyrem. We currently have nine issued, unexpired patents in the U.S. relating to Xyrem. All but two of these patents are listed in the FDA’s publication “Approved Drug Products with Therapeutic Equivalence Evaluations,” or the Orange Book. Our patents relate to Xyrem’s stable and microbially resistant formulation, |
Some of our Orange Book-listed patents that have not been invalidated.
A Xyrem formulation patent hasthat had issued in multiple non-U.S. countries and will expireexpired in December 2019. The European Patent Office has issued a method of administration patent relating to the DDI between Xyrem and divalproex sodium that will expire in February 2034. Those patents areThat patent is licensed to UCB as the marketing authorization holder outside of the U.S. and Canada, and UCB has the right to enforce them.it. In addition to our issued patents, we have patent applications relating to Xyrem pending in the U.S. and other countries.
| • | Sunosi. We acquired worldwide development, manufacturing and commercial rights to solriamfetol from Aerial BioPharma LLC, or Aerial, in 2014, including Aerial’s patent rights relating to solriamfetol, other than in certain jurisdictions in Asia where SK Biopharmaceuticals Co., Ltd. retains rights. We have a portfolio of U.S. and non-U.S. patents and patent applications for solriamfetol relating to various compositions, formulations and methods of use. Four of our U.S. patents are method of use patents covering treatment of sleep-related conditions expiring between June 2026 and August 2027. Two other U.S. patents cover, respectively, the formulation of solriamfetol and the method of treating select conditions with formulations of solriamfetol (both expiring in September 2037). A request for a patent term extension for one of the above method of use patents has been filed. Sunosi has also been granted orphan drug exclusivity for narcolepsy in the U.S. |
| • | Defitelio. The unique process of deriving defibrotide from porcine DNA is extensive and uses both chemical and biological processes that rely on complex characterization methods. We have U.S. and non-U.S. patents and patent applications relating to various compositions, methods of use and methods of characterization, expiring at various times between April 2021 and November 2035. None of these patents are listed in the Orange Book. Defibrotide has been granted orphan drug exclusivity by the FDA to treat and prevent VOD until March 2023. Defibrotide has also been granted orphan drug designation by the EC and the Korean Ministry of Food and Drug Safety to treat and prevent VOD, by the Commonwealth of Australia-Department of Health for the treatment of VOD and by the EC for the prevention of aGvHD. We acquired the rights to defibrotide for the treatment and prevention of VOD in North America, Central America and South America from Sigma-Tau Pharmaceuticals, Inc. in 2014. |
| • | Erwinaze. Erwinaze has no patent protection. It had been granted orphan drug exclusivity by the FDA for the treatment of ALL in the U.S. until November 2018, and as a biological product approved under a BLA, we believe |
that it is protected by exclusivity that prevents approval of a biosimilar in the U.S. through late 2023 under the U.S. Biologics Price Competition and Innovation Act, or BPCIA.
| • | Vyxeos. We have a portfolio of U.S. and non-U.S. patents and patent applications for Vyxeos and the CombiPlex technology platform relating to various compositions and methods of making and use. These include six U.S. patents covering Vyxeos compositions and methods of use expiring between April 2025 and September 2034 and two U.S. patents covering CombiPlex (which also cover Vyxeos) expiring in January 2027. These patents are listed in the Orange Book. Vyxeos has been granted orphan drug exclusivity by the FDA until August 2024, seven years from its FDA approval, for the treatment of adults with newly-diagnosed t-AML or AML-MRC. In addition, Vyxeos has been granted orphan drug designation by the EC until August 2028, ten years from its EC approval for the treatment of adults with newly-diagnosed t-AML or AML-MRC. |
We also rely on trade secrets and other unpatented proprietary information to protect our products and commercial position, particularly with respect to our products with limited or no patent protection, such as Erwinaze.
The patents and/or patent applications that relate to our product candidates include:
| • | JZP-258. We have U.S. patents and patent applications that relate to our product candidate JZP-258. These patents expire December 2033. |
| • | JZP-385. Through the acquisition of Cavion in 2019, we obtained a portfolio of U.S. and non-U.S. patents and patent applications, including rights relating to compositions and methods of using JZP-385. The portfolio includes a U.S. composition of matter patent relating to JZP-385, which expires in 2027. |
| • | JZP-458. We obtained worldwide rights from Pfenex, including Pfenex’s patent rights relating to JZP-458, in 2016 to develop and commercialize multiple early-stage hematology product candidates, including a license to a U.S. process patent relating to JZP-458, which expires in 2026. |
| • | Lurbinectedin. In December of 2019, we entered into an exclusive license agreement with PharmaMar pursuant to which we obtained exclusive U.S. development and commercialization rights to lurbinectedin, including a license to a U.S. composition of matter patent, which expires in 2024. |
In addition, we have rights to a number of trademarks and service marks, and pending trademark and service mark applications, in the U.S. and elsewhere in the world to further protect the proprietary position of our products. For furthera discussion of the challenges we face in obtaining or maintaining patent and/or trade secret protection, see the risk factors under the heading “Risks Related to Our Intellectual Property” in Part I, Item 1A of this Annual Report on Form 10‑K.
Government Regulation
As a global pharmaceutical company, our activities are subject to extensive regulation in the U.S., the EU and other countries where we do business. Regulatory requirements encompass the entire life cycle of pharmaceutical products, from research and development activities to marketing approval, manufacturing, labeling, packaging, adverse event and safety reporting, storage, advertising, promotion, sale, pricing and reimbursement, recordkeeping, distribution, importing and exporting. Regulations differ from country to country and are constantly evolving.
Testing and Approval of Pharmaceutical Products
We are not permitted to market a product in a country until we receive approval from the relevant regulatory authority, such as the FDA in the U.S. and the EC or the competent authorities of the EU member states. An application for marketing approval must contain information generated by the applicant, also called a sponsor, demonstrating the quality, safety and efficacy of the product candidate, including data from preclinical and clinical trials, proposed product packaging and labeling and information pertaining to product formulation and the manufacture and analytical testing of the API and the finished product.
In the U.S., the FDA reviews and, if warranted, approves applications for marketing approval. The process for obtaining marketing approval in the U.S. for a drug or biologic product candidate generally includes:
conducting preclinical laboratory and animal testing and submitting the results to the FDA in an investigational new drug, or IND, application requesting approval to test the product candidate in human clinical trials;
conducting adequate and well-controlled human clinical trials to establish the safety and efficacy of the product candidate in the desired indication;
15
submitting an NDA, supplemental NDA, or sNDA, or BLA, as appropriate, to the FDA seeking approval for a specific indication; and
completing inspections by the FDA of the facilities where the product candidate is manufactured, analyzed and stored to demonstrate compliance with current Good Manufacturing Practices, or cGMP, and any requested FDA audits of the clinical trial sites that generated the data supporting the application.
Human clinical trials conducted before approval of a product generally proceed in three sequential phases, although the phases may overlap. In Phase 1, the initial introduction of the product candidate in humans, the product candidate is typically tested to assess metabolism, pharmacokinetics, pharmacological actions and side effects associated with increasing doses. Phase 2 usually involves clinical trials in a limited patient population to determine the effectiveness of the product candidate for a particular indication or indications, dosage tolerance and optimum dosage and to identify common adverse effects and safety risks. If a product candidate demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2, Phase 3 clinical trials are undertaken to obtain additional information about clinical efficacy and safety in a larger number of patients. Clinical trials must be conducted in accordance with specific protocols, as well as FDA requirements related to conducting the trials and recording and reporting the results, commonly referred to as good clinical practices, to ensure that the resulting data are credible and accurate and that the trial participants are adequately protected. The FDA enforces good clinical practices through periodic inspections of trial sponsors, clinical investigators and trial sites.
Once an NDA, sNDA or BLA has been compiled and submitted, the FDA performs an initial review before it accepts the application for filing. The FDA may refuse to file an application and/or request additional information before acceptance. Once accepted for filing, the FDA begins an in-depth review of the application. Under the current goals and policies agreed to by the FDA under PDUFA for a new molecular entity, the FDA has ten months from the filing decision in which to complete its initial review of a standard application and respond to the applicant, and eight months for a priority application. The FDA does not always meet its PDUFA goal dates, and in certain circumstances, the PDUFA goal date may be extended. For example, in December 2018, the FDA determined that a submission we made during the course of discussions regarding draft labeling in solriamfetol constituted a major amendment to the NDA, resulting in a three-month extension of the PDUFA goal date to March 20, 2019.
The FDA also has various programs, including Fast Track, Priority Review, Breakthrough Therapy and Accelerated Approval (Subpart H and E), that are intended to expedite the process for reviewing certain applications and/or provide for approval on the basis of surrogate endpoints or restricted distribution. Generally, products may be eligible for one or more of these programs if they are intended for serious or life-threatening diseases or conditions, have potential to address unmet medical needs, or may provide meaningful benefit over existing treatments. For example, the FDA granted Vyxeos
Breakthrough Therapy and Fast Track designations and also granted Priority Review with respect to our NDA for Vyxeos for the treatment of t-AML and AML-MRC that was approved in August 2017. In addition, a priority review voucher, or PRV such as the PRV that we acquired in May 2018, may be used to obtain priority review by the FDA for one of our future regulatory submissions. We have used the PRV we acquired in May 2018 to obtain priority review for our JZP-258 NDA, which is under review by the FDA.
During its review of an application, the FDA evaluates whether the product demonstrates the required level of safety and efficacy for the indication for which approval is sought and also conducts the inspections and audits described above. The FDA may also refer an application to an advisory committee, typically a panel of clinicians, for review, evaluation and a non-binding recommendation as to whether the application should be approved. When the FDA completes its evaluation, it issues either an approval letter or a complete response letter. A complete response letter generally outlines what the FDA considers to be the deficiencies in the application and may indicate that substantial additional testing or information is required in order for the FDA to approve the product. If and when identified deficiencies have been addressed to the FDA’s satisfaction inafter a review of the resubmission of the application, or if the decision is reversed through an administrative appeal, the FDA will issue an approval letter.
Even if a product is approved, the approval may be subject to limitations based on the FDA’s interpretation of the data submitted in the application. For example, as a condition of approval, the FDA may require the sponsor to agree to certain post-marketing commitments,requirements, such as conducting Phase 4, or post-approval, clinical trials to gain additional safety data or to document a clinical benefit in the case of products approved under Accelerated Approval regulations. The FDA’s approval of the BLA for Erwinaze includes a number of post-marketing commitments related to the manufacture of Erwinaze.and requirements. Several post-marketing commitments and requirements were also mandated by the FDA in connection with its approvalsapproval of Defitelio, including the requirement that we conduct a clinical trial to analyze the safety of defibrotide versus best supportive care in the prevention of VOD in adult and pediatric patients, and its approval of Vyxeos, including the requirement that we conduct a safety study to characterize infusion-related reactions in patients treated with Vyxeos and a clinical trial to determine dosing to minimize toxicity in patients with moderate and severe renal impairment.
In addition, if the FDA determines that a REMS is necessary to ensure that the benefits of the product outweigh the risks, a sponsor may be required to include a proposed REMS (either as part of the application or after approval), which may include a patient package insert or a medication guide to provide information to consumers about the product’s risks and benefits; a plan for communication to healthcare providers; or conditions on the product’s prescribing or distribution referred to as elements to assure safe use. Xyrem is required to have a REMS. For more discussion regarding the Xyrem REMS, see the risk factors under the headings “The distribution and sale of Xyremour oxybate products are subject to significant regulatory restrictions,
16
including the requirements of a REMS, and these regulatory requirements subject us to risks and uncertainties, any of which could negatively impact sales of Xyrem and if approved, JZP-258” and “Risks Related to Our Intellectual Property” in Part I, Item 1A of this Annual Report on Form 10‑K.
The EU and many individual countries have regulatory structures similar to the U.S. for conducting preclinical and clinical testing and applying for marketing approval or authorization, although specifics may vary widely from country to country. Clinical trials in the EU must be conducted in accordance with the requirements of the EU Clinical Trials Directive, which may be replaced with the new EU Clinical Trials Regulation in 2019,2022, and applicable good clinical practice standards. In the EU, there are several procedures for requesting marketing authorization which can be more efficient than applying for authorization on a country-by-country basis. There is a “centralized” procedure allowing submission of a single marketing authorization application to the European Medicines Agency, or EMA. If the EMA issues a positive opinion, the EC will grant a centralized marketing authorization that is valid in all EU member states and three of the four European Free Trade Association countries (Iceland, Liechtenstein and Norway). The centralized procedure is mandatory for certain medicinal products, including orphan medicinal products and biotechnology-derived medicinal products, and optional for others. There is also a “decentralized” procedure allowing companies to file identical applications to several EU member states simultaneously for product candidates that have not yet been authorized in any EU member state and a “mutual recognition” procedure allowing companies that have a product already authorized in one EU member state to apply for that authorization to be recognized by the competent authorities in other EU member states. The UK’s planned withdrawal from the EU on January 31, 2020, commonly referred to as Brexit, has, however, created significant uncertainty concerning the future relationship between the UK and the EU, and theEU. The impact of Brexit on the on-going validity in the UK of current EU authorizations for medicinal products, whether granted through the centralized procedure, decentralized procedure, or mutual recognition, and on the future process for obtaining or maintaining marketing authorization for pharmaceutical products manufactured or sold in the UK is unknown.
The maximum timeframe for the evaluation of an application in the EU under the centralized procedure is 210 days, subject to certain exceptions.exceptions and clock stops. An initial marketing authorization granted in the EU is valid for five years, with renewal subject to re-evaluation of the risk-benefit profile of the product. Once renewed, the authorization is usually valid for an unlimited period unless the national competent authority or the EC decides on justified grounds to proceed with one additional five-year renewal.
In the EU, if an applicant can demonstrate that comprehensive data on the efficacy and safety of the product under normal conditions of use cannot be provided due to certain specified objective and verifiable reasons, products may be granted marketing authorization “under exceptional circumstances”.circumstances.” A marketing authorization granted under exceptional
circumstances is valid for five years, subject to an annual reassessment of conditions imposed by the EC. The marketing authorization in the EU for Defitelio was granted under exceptional circumstances because it was not possible to obtain complete information about the product due to the rarity of the disease and because ethical considerations prevented conducting a study directly comparing Defitelio with best supportive care or a placebo. As a result, the marketing authorization requires us to comply with a number of post-marketing obligations, including obligations relating to the manufacture of the drug substance and finished product, the submission of data concerning patients treated with the product collected through a third-party patient registry and the establishment of a multi-center, multinational and prospective observational patient registry to investigate the long-term safety, health outcomes and patterns of utilization of Defitelio during normal use. We are in the process of conducting the post-authorization study in the EU to provide further data on long-term safety, health outcomes and patterns of utilization of Defitelio in normal use.
Similar to the use of REMS in the U.S. to ensure that the benefits of a product outweigh its risks, in the EU and other countries we are required and may, in the future in relation to new products, be required to agree to post-marketing obligations in the marketing authorization for our products, to include a patient package insert or a medication guide to provide information to consumers about the product’s risks and benefits, to implement a plan for communication to healthcare providers, and to impose restrictions on the product’s distribution. For example, the marketing authorization in the EU for Vyxeos requires us to comply with certain manufacturing-related post-approval commitments.
After approval, certain changes to the approved product, such as adding new indications, making certain manufacturing changes, modifying a REMS, or making certain additional labeling claims, are subject to further regulatory review and approval. Obtaining approval for a new indication generally requires that additional clinical studies be conducted to demonstrate that the product is safe and effective for the new intended use. Such regulatory reviews can result in denial or modification of the planned changes, or requirements to conduct additional tests or evaluations that can substantially delay or increase the cost of the planned changes.
Manufacture of Pharmaceutical Products
The manufacturing process for pharmaceutical products is highly regulated, and regulators may shut down manufacturing facilities that they believe do not comply with regulations. We and the third party suppliers of our products are subject to cGMP, which are extensive regulations governing manufacturing processes, stability testing, recordkeeping and quality
17
standards as defined by the FDA, the EC, the EMA, competent authorities of EU member states and other regulatory authorities. The FDA also periodically inspects manufacturing facilities and the sponsor’s and manufacturer’s records related to manufacturing, and assesses compliance with cGMP. Following such inspections, the FDA may issue notices on Form FDA 483 and warning letters. For example, the FDA issued a warning letter to PBL, ourthe Erwinaze manufacturer, in January 2017 indicating that it was not satisfied with PBL’s responses to a Form 483 issued to PBL and citing significant violations of cGMP for finished pharmaceuticals and significant deviations from cGMP for APIs. As recently as August 2018, the FDA conducted an inspection of the PBL manufacturing facility and issued an FDA Form 483 to PBL citing observations related to items referenced in the existing warning letter as well as other manufacturing practices, including data and records management. In addition to Form FDA 483 notices and warning letters, failure to comply with the statutory and regulatory requirements may result in suspension of manufacturing, product seizure, withdrawal of the product from the market, administrative, civil and criminal penalties, among other enforcement remedies both in the U.S. and in non-U.S. countries.
In the EU, a manufacturing authorization is required to manufacture medicinal products, and the manufacturing authorization holder must comply with various requirements set out in applicable EU laws, regulations and guidance. These requirements include compliance with EU cGMP standards when manufacturing products and their APIs, including APIs manufactured outside of the EU with the intention of importing them into the EU. In this regard, in the UK, where PBL’s manufacturing facilities are located, PBL is subject to inspections conducted by the MHRA. Following a site inspection of PBL by the MHRA in December 2017, MHRA issued an inspection report listing several major findings, including major deficiencies and failures by PBL to comply with cGMP. In addition to inspection reports, manufacturers and marketing authorization holders may be subject to civil, criminal or administrative sanctions, including suspension of manufacturing authorization, in cases of non-compliance with the EU or EU member states’ requirements applicable to manufacturing.
Sales and Marketing of Pharmaceutical Products
Advertising and Promotional Activities
The FDA regulates advertising and promotional activities for products in the U.S., requiring advertising, promotional materials and labeling to be truthful and not misleading, and products to be marketed only for their approved indications and in accordance with the provisions of the approved label. The FDA actively investigates allegations of off-label promotion in order to enforce regulations prohibiting these types of activities. The FDA routinely issues informal and more formal communications such as untitled letters or warning letters interpreting its authority over these matters. While such communications may not be considered final agency decisions, many companies may decide not to contest the agency’s
interpretations so as to avoid disputes with the FDA, even if they believe the claims they were making to be truthful, not misleading and otherwise lawful.
In the EU, the advertising and promotion of our products are subject to laws governing promotion of medicinal products, interactions with physicians, misleading and comparative advertising and unfair commercial practices. For example, applicable laws require that promotional materials and advertising in relation to medicinal products comply with the product’s Summary of Product Characteristics, or SmPC, as approved by the competent authorities in connection with a marketing authorization approval. The SmPC is the document that provides information to physicians concerning the safe and effective use of the product. Promotional activity that does not comply with the SmPC is considered off-label and is prohibited in the EU. Other applicable laws at the EU level and in the individual EU member states also apply to the advertising and promotion of medicinal products, including laws that prohibit the direct-to-consumer advertising of prescription-only medicinal products and further limit or restrict the advertising and promotion of our products to the general public and to health care professionals. Violations of the rules governing the promotion of medicinal products in the EU could be penalized by administrative measures, fines and imprisonment.
We are also subject to numerous anti-fraudfraud and abuse laws and regulations globally. In the U.S., there are a variety of federal and state laws restricting certain marketing practices in the pharmaceutical industry pertaining to healthcare fraud and abuse, including anti-kickback laws and false claims laws. Our sales, marketing, patient support and medical activities may be subject to scrutiny under these laws. The U.S. federal healthcare program anti-kickback statuteAnti-Kickback Statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving anything of value to induce (or in return for) the referral of business, including the purchase, recommendation or prescription of a particular drug reimbursable under Medicare, Medicaid or other federally financed healthcare programs. The statute has been interpreted to apply to arrangements between pharmaceutical companies on one hand and patients, prescribers, purchasers and formulary managers on the other. Violations of the federal anti-kickback statute may be punished by civil and criminal fines, imprisonment, and/or exclusion from participation in federal healthcare programs.
18
criminal, and administrative fines and penalties, damages, imprisonment, and/or exclusion from participation in federal healthcare programs.
The federal civil False Claims Act prohibits, among other things, any person from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment of federal funds, or knowingly making, or causing to be made, a false statement to get a false claim paid. A claim resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim. The False Claims Act also permits a private individual acting as a “whistleblower” to bring actions on behalf of themselves and the federal government alleging violations of the statute and to share in any monetary recovery. Violations of the False Claims Act may result in significant financial penalties (on(including mandatory penalties on a per claim or statement basis), treble damages and exclusion from participation in federal health care programs.
Pharmaceutical companies are subject to other federal false claim and statements laws, some of which extend to non-government health benefit programs. For example, the healthcare fraud provisions under the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act, or HIPAA, impose criminal liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any health care benefit program, including private third party payors, or falsifying or covering up a material fact or making any materially false or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services. Violations of HIPAA fraud provisions may result in criminal, civil and administrative penalties, fines and damages, including exclusion from participation in federal healthcare programs.
The majority of individual states also have statutes or regulations similar to the federal anti-kickback law and the False Claims Act, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Other states restrict whether and when pharmaceutical companies may provide meals to health care professionals or engage in other marketing-related activities, and certain states and cities require identification or licensing of sales representatives.
Other Post-Approval Pharmaceutical Product Regulation
Safety Reporting/Pharmacovigilance
The FDA, the EMA and other governmental authorities track information on side effects and adverse events reported during clinical studies and after marketing approval. We are required to file periodic safety update reports with the authorities concerning adverse events. If, upon review, an authority determines that any events and/or reports indicate a trend or signal, they can require a change in a product label, restrict sales and marketing, require post-approval safety studies, require a labor intensive collection of data regarding the risks and benefits of marketed products and ongoing assessments of those risks and benefits and/or require or conduct other actions, potentially including withdrawal or suspension of the product from the market. For example, if the EMA has concerns that the risk-benefit profile of a product has changed, it can, following an investigation procedure, adopt an opinion advising that the existing marketing authorization for the product be varied or suspended and requiring the marketing authorization holder to conduct
post-authorization safety studies. The opinion is then submitted for approval by the EC. Also, from time to time, the FDA issues drug safety communications on its adverse event reporting system based on its review of reported adverse events.
The FDA and the competent authorities of the EU member states on behalf of the EMA also periodically inspect our records related to safety reporting. Following such inspections, the FDA may issue notices on FDA Form 483 and warning letters that could cause us to modify certain activities. An FDA Form 483 notice, if issued, can list conditions the FDA investigators believe may have violated relevant FDA regulations or guidance. Failure to adequately and promptly correct the observation(s) can result in a warning letter or other regulatory enforcement action. Similarly, the EMA’s Pharmacovigilance Risk Assessment Committee may propose to the Committee for Medicinal Products for Human Use that the marketing authorization holder be required to take specific steps. Non-compliance in all cases can lead to the variation, suspension or withdrawal of marketing authorization or imposition of financial penalties or other enforcement measures.
Sunshine Act and Transparency Laws
The Physician Payment Sunshine Act requires extensive tracking of payments and transfers of value to physicians and teaching hospitals and ownership interests held by physicians and their families, and provides for reporting to the federal government and public disclosure of these data. Beginning in 2022, reporting will also be required of information regarding payments and transfers of value provided to physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, and certified nurse-midwives. A number of states now require pharmaceutical companies to report expenses relating to the marketing and promotion of pharmaceutical products and to report gifts and payments to individual physicianshealthcare providers in the states. Government agencies and private entities may inquire about our marketing practices or pursue other enforcement activities based on the disclosures in those public reports. Other states restrict when pharmaceutical companies may provide meals to prescribers or engage in other marketing-related activities.
Outside the U.S., interactions between pharmaceutical companies and physicians are also governed by strict laws, regulations, industry self-regulation codes of conduct and physicians’ codes of professional conduct. The provision of benefits
19
or advantages to physicians to induce or encourage the prescription, recommendation, endorsement, purchase, supply, order or use of medicinal products, which is prohibited in the EU, is governed by the national anti-bribery laws of the EU member states, as described below in “Business—Government Regulation—Anti-Corruption Legislation” in this Part I, Item 1. Violation of these laws could result in substantial fines and imprisonment. Certain EU member states require that payments made to physicians be publicly disclosed. Moreover, agreements with physicians must often be the subject of prior notification and approval by the physician’s employer, his/her competent professional organization, and/or the competent authorities of the individual EU member states. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment.
Controlled Substance Regulations
A drug product approved by the FDA may be subject to scheduling as a controlled substance under the CSA depending on the drug’s potential for abuse. Controlled substances that are pharmaceutical products are subject to a high degree of regulation under the CSA, which establishes, among other things, certain registration, manufacturing quotas, security, recordkeeping, reporting, import, export and other requirements administered by the DEA. The DEA classifies controlled substances into five schedules. Schedule I substances by definition have a high potential for abuse, have no currently “accepted medical use” in the U.S., lack accepted safety for use under medical supervision, and may not be prescribed, marketed or sold in the U.S. Pharmaceutical products approved for use in the U.S. may be listed as Schedule II, III, IV or V, with Schedule II substances considered to present the highest potential for abuse or dependence and Schedule V substances the lowest relative risk of abuse. The active ingredientAPI of Xyrem, sodium oxybate, is regulated by the DEA as a Schedule I controlled substance.substance, and Xyrem as an FDA-approved drug product, is regulated as a Schedule III controlled substance. We expect thatThe API of Sunosi, solriamfetol, will be subject to scheduling under the CSA, which will need to be completed after any NDA approval and before commercial launch.is regulated as a Schedule IV controlled substance. Individual states also impose similar requirements for controlled substances.
The DEA limits the quantity of certain Schedule I controlled substances that may be manufactured and procured in the U.S. in any given calendar year through a quota system and, as a result, quotas from the DEA are required in order to manufacture and package sodium oxybate and Xyrem in the U.S. Accordingly, we require DEA quotas for Siegfried, our U.S.‑based sodium oxybate supplier, to procure sodium oxybate and for Patheon, our U.S.-based Xyrem supplier, to obtain the sodium oxybate from Siegfried in order to manufacture and supply us with Xyrem. Xyrem manufactured at our plant in Ireland enters the U.S. as a Schedule III drug and thus does not require a manufacturing quota.
As a Schedule III drug, Xyrem is also subject to DEA and state regulations relating to manufacturing, storage, distribution and physician prescription procedures, including limitations on prescription refills. In addition, the third parties who perform our clinical and commercial manufacturing, distribution, dispensing and clinical studies for Xyrem are required to maintain necessary DEA registrations and state licenses. The DEA periodically inspects facilities for compliance with its rules and regulations.
Other Regulations
There are many other requirements and restrictions in the U.S. and elsewhere imposed on pharmaceutical companies and their activities, including those related to the posting of information relating to clinical studies and their outcomes, the export and importation of products, required authorizations for distributors, the identification or licensing of sales representatives, restrictions on the ability of manufacturers to offer co-pay support to patients for certain prescription drugs, implementation of required compliance programs or marketing codes of conduct, protection of the environment, taxation and work safety. Non-compliance with such requirements may result in civil, criminal or administrative sanctions.
Anti-Corruption Legislation
Our business activities outside of the U.S. are subject to the U.S. Foreign Corrupt Practices Act, or FCPA, and similar anti-bribery or anti-corruption laws, regulations, industry self-regulation codes of conduct and physicians’ codes of professional conduct or rules of other countries in which we operate, including the UK Bribery Act of 2010, or the UK Bribery Act. The FCPA and similar anti-corruption laws in other countries generally prohibit the offering, promising, giving, or authorizing others to give anything of value, either directly or indirectly, to U.S. or non-U.S. government officials in order to improperly influence any act or decision, secure an improper advantage, or obtain or retain business. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect the transactions of the company and to devise and maintain an adequate system of internal accounting controls. The UK Bribery Act prohibits giving, offering, or promising bribes to any person, including UK and non-UK government officials and private persons, as well as requesting, agreeing to receive, or accepting bribes from any person. In addition, under the UK Bribery Act, companies that carry on a business or part of a business in the UK may be held liable for bribes given, offered or promised to any person, including UK and non-UK government officials and private persons in any country, by employees and persons associated with the company in order to obtain or retain business or a business advantage for the company. Liability is strict, with no element of a corrupt state of mind, but a defense of having in place adequate procedures designed to prevent bribery is available. As described above, our business is heavily regulated and therefore involves significant interaction with government officials in many countries. Additionally, in certain countries, the health care providers who prescribe pharmaceuticals are employed by their government, and the
20
purchasers of pharmaceuticals are government entities; therefore, our dealings with these prescribers and purchasers may be subject to the FCPA, the UK Bribery Act and similar laws. Recently the Securities and Exchange Commission, or SEC, and the DOJ have increased their FCPA enforcement activities with respect to pharmaceutical companies. In addition, under the Dodd-Frank Wall Street Reform and Consumer Protection Act, private individuals who report to the SEC original information that leads to successful enforcement actions may be eligible for a monetary award.We engage in ongoing efforts designed to ensure our compliance with these laws, including due diligence, training, policies, procedures, and internal controls. However, there is no certainty that all employees and third party business partners (including our distributors, wholesalers, agents, contractors, and other partners) will comply with anti-bribery laws. In particular, we do not control the actions of our suppliers and other third party agents, although we may be liable for their actions. Violation of these laws may result in civil or criminal sanctions, which could include monetary fines, criminal penalties, and disgorgement of past profits.
Data Protection and Privacy
We are also subject to data protection and privacy laws and regulations governing the processing ofglobally, which may place restrictions on our ability to transfer, access and use personal data.data across our business. The legislative and regulatory landscape for privacy and data security is subject to increasing focus and continues to evolve. For example,There has been increased attention to privacy and data security issues that could potentially affect our business, including the EU General Data Protection Regulation, or GDPR, which became effective inwent into effect on May 25, 2018 introduced new data protection requirements for all individuals within the EU and the European Economic Area, or EEA. It also addresses the exportimposes penalties up to 4% of personal data outside the EU and EEA areas. Substantial fines may be imposed for violations of GDPR.annual global turnover. In addition, certain EU member states have adopted more stringent data protection standards, which add to the complexity of processing personal datalaws and regulations enacted in the EU. TheUnited States, Europe, Asia and Latin America, including the new California Consumer Privacy Act of 2018, effective beginningwhich went into effect January 1, 2020, mirrorsincreases potential enforcement and litigation activity. In order to manage these evolving risks, we have adopted a number of the key provisions in the GDPR.
Marketing Exclusivity
The Hatch-Waxman Act
The marketing approval process described above for the U.S. is premised on the applicant being the owner of, or having obtained a right of reference to, all of the data required to prove the safety and effectiveness of a drug product. This type of marketing application, sometimes referred to as a “full” or “stand-alone” NDA, is governed by Section 505(b)(1) of the United States Federal Food, Drug, and Cosmetic Act, or FDCA. A Section 505(b)(1) NDA contains full reports of investigations of safety and effectiveness, which includes the results of preclinical and clinical trials, together with detailed information on the manufacture and composition of the product, in addition to other information. As an alternative, the Drug Price Competition and Patent Term Restoration Act of 1984, or the Hatch-Waxman Act, provides two abbreviated approval pathways for certain drug products.
The first path, under Section 505(b)(2) of the FDCA, usually is used for the approval of a product that is similar, but not identical, to a previously-approved brand-name product, referred to as the reference listed drug, or RLD. Under this path, the applicant is permitted to rely to some degree on the FDA’s finding that the RLD is safe and effective and must submit its own product-specific data on safety and effectiveness only to the extent necessary to bridge the differences between the products. The second abbreviated path established under the Hatch-Waxman Act is for the approval of generic drugs. Section 505(j) of the FDCA permits the submission of an ANDA for a generic version of an approved, brand-name drug. Generally, an ANDA must contain data and information showing that the proposed generic product and the RLD (i) have the same active ingredient, in the same strength and dosage form, to be delivered via the same route of administration, (ii) are intended for the same uses, and (iii) are bioequivalent. This data and information are provided instead of data and information independently demonstrating the proposed generic product’s safety and effectiveness.
The Hatch-Waxman Act requires an ANDA or a Section 505(b)(2) NDA applicant to certify that there are no patents listed for that product in the Orange Book, or that for each Orange Book-listed patent either the listed patent has expired, the listed patent will expire on a particular date and approval is sought after patent expiration, or the listed patent is invalid or will not be infringed by the manufacture, use or sale of the new product. A certification that approval is sought after patent expiration is called a “Paragraph III Certification.” A certification that the new product will not infringe the RLD’s Orange Book-listed patents, or that such patents are invalid, is called a “Paragraph IV Certification.” If a relevant patent covers an approved method of use, an ANDA or Section 505(b)(2) NDA applicant can also file a statement, called, in the case of an ANDA, a “section viii statement,” that the application does not seek approval of the method of use covered by the listed patent. With such a statement, the applicant must “carve out” the protected method of use (typically an indication and related material) from the proposed product’s labeling. If the applicant makes a Paragraph III Certification, the ANDA or the Section 505(b)(2) NDA will not be approved until the listed patents claiming the RLD have expired.
If the applicant has provided a Paragraph IV Certification to the FDA, the applicant must also send a notice of that certification to the NDA holder and the relevant patent holders once the FDA accepts the ANDA or the Section 505(b)(2) NDA for filing. The NDA and patent holders then have 45 days to initiate a patent infringement lawsuit. Filing the lawsuit triggers an automatic stay on FDA’s approval of the ANDA or the Section 505(b)(2) NDA until the earliest of 30 months after the NDA holder’s receipt of the notice of Paragraph IV Certification, expiration of the patent, certain settlements of the lawsuit, or a
21
decision in the infringement case that is favorable to the applicant. The FDA may issue tentative approval of an application if the application meets all conditions for approval but cannot receive effective approval because the 30-month stay or aanother period of regulatory exclusivity has not expired. If an ANDA or Section 505(b)(2) NDA is approved before conclusion of any relevant patent litigation, the applicant can choose to launch the product, but does so “at risk” of being liable for damages, and potentially treble damages, if the RLD sponsor or patent holder ultimately prevails in patent litigation.
Under the Hatch-Waxman Act, newly approved drugs and indications may benefit from statutory periods of non-patent marketing exclusivity that can potentially delay review or approval of an ANDA or Section 505(b)(2) application. For example, the Hatch-Waxman Act provides five-year marketing exclusivity to the first applicant to gain approval of an NDA for a new chemical entity, meaning a drug containing an active moiety that the FDA has not previously approved. During this period, the FDA cannot accept for review an ANDA or a Section 505(b)(2) NDA for a product containing the same moiety, except that an application containing a Paragraph IV Certification may be submitted after four years, which may trigger the litigation and stay described above. The Hatch-Waxman Act also provides three years of marketing exclusivity with the approval of an NDA, including a Section 505(b)(2) NDA, for a product containing a previously-approved moiety but that incorporates a change (such as a new indication, dosage form or strength) from an approved product with the same moiety, if the change required clinical data from new investigations that were conducted or sponsored by the applicant. This three-year exclusivity does not preclude submission of the ANDA or Section 505(b)(2) NDA for such a product, but prevents the FDA from giving final approval to such product.
The Hatch-Waxman Act also permits a patent term extension of up to five years (but not beyond 14 years from the date of approval) for an NDA, including a Section 505(b)(2) NDA, that is approved for a product that contains an active ingredient that has not previously been approved. The extension, which compensates for patent term lost during product development and the FDA regulatory review process, is generally equal to the sum of one-half the time between the effective date of an IND application and the submission date of an NDA, and all of the time between the submission date of an NDA and the approval of that application. It is available for only one patent for a given product, and it must be a patent that claims the product or a method of using or manufacturing the product. The USPTO, in consultation with the FDA, reviews and approves applications for patent term extension.
Orphan Drug and Other Exclusivities
Some jurisdictions, including the U.S., may designate drugs or biologics for relatively small patient populations as orphan drugs. The FDA grants orphan drug designation to drugs or biologics intended to treat a rare disease or condition, which is one that affects fewer than 200,000 individuals in the U.S., or more than 200,000 individuals, but for which there is no reasonable expectation that the cost of developing the product and making it available in the U.S. for the disease or condition will be recovered from U.S. sales of the product. Orphan drug designation does not shorten the duration of the regulatory review process or lower the approval standards, but can provide important benefits, including consultation with the FDA. If a product is approved for its orphan designated use, it may be entitled to orphan drug exclusivity, which blocks the FDA from approving for seven years any other application for a product that is the same drug for the same indication. If there is a previously-approved product that is the same drug for the same indication, orphan drug designation requires the sponsor to provide a plausible hypothesis of clinical superiority over the approved product, whereas orphan drug exclusivity requires the sponsor to actually demonstrate clinical superiority. Clinical superiority can be established by way of greater efficacy, greater safety, or making a major contribution to patient care. Additionally, a later product can be approved if the sponsor holding orphan drug exclusivity consents, or cannot adequately supply the market. Orphan drug exclusivity does not prevent approval of another sponsor’s application for different indications or uses of the same drug, or for different drugs for the same indication. Defibrotide has been granted orphan drug exclusivity by the FDA to treat and prevent VOD until March 2023. Vyxeos has been granted orphan drug exclusivity by the FDA for the treatment of AML until August 2024.
Biologic products approved under a BLA are subject to the BPCIA, which authorizes an abbreviated approval pathway for a biological product that is “biosimilar” to an already approved biologic, or reference product. The BPCIA provides periods of exclusivity that protect a reference product from competition by biosimilars. The FDA may not accept a biosimilar application for review until four years after the date of first licensure of the reference product, and the biosimilar cannot be licensed until 12 years after the reference product was first licensed. We believe that Erwinaze, which was approved under a BLA in November 2011, is subject to an exclusivity period that will prevent approval of a biosimilar in the U.S. into November 2023.
Under certain circumstances, the exclusivity periods applicable to drugs and biologics and the patent-related protections applicable to drugs may be eligible for a six-month extension if the sponsor submits pediatric data that fairly respond to a written request from the FDA for such data. This exclusivity may be granted even if the data does not support a pediatric indication. We will consider seeking pediatric exclusivity for our products whenever appropriate. For example, in response to a written request from the FDA, we conducted a Phase 3 clinical trial to assess the safety and efficacy of Xyrem in children and adolescents aged seven to 17 who have narcolepsy with cataplexy, and submitted study results in a supplement to the Xyrem NDA, seeking approval for this indication. In October 2018, FDA approved the sNDA and notified us that we had been
22
granted pediatric exclusivity, extending by six months the preclusive effect of our Orange Book-listed patents for Xyrem, that have not been invalidated, as well as the three-year regulatory exclusivity period granted to the Xyrem pediatric indication because of the clinical studies that were necessary for approval of the sNDA.
In the EU, orphan drug designation may be granted to products that can be used to treat life-threatening diseases or chronically debilitating conditions with an incidence of no more than five in 10,000 people or that, for economic reasons, would be unlikely to be developed without incentives. Orphan designated medicinal products are entitled to a range of benefits during the development and regulatory review process and ten years of market exclusivity in all EU member states upon approval. Similar toAs in the U.S., a similar medicinal product with the same orphan indication may be approved, notwithstanding orphan product exclusivity, if the exclusivity holder gives consent or if the manufacturer of the original orphan medicinal product is unable to supply sufficient quantities. Marketing authorization may also be granted to a similar medicinal product with the same orphan indication if the similar product is deemed safer, more effective or otherwise clinically superior to the original orphan medicinal product. The period of market exclusivity granted in relation to the original orphan medicinal product may, in addition, be reduced to six years if it can be demonstrated on the basis of available evidence that the original orphan medicinal product is sufficiently profitable not to justify maintenance of market exclusivity. Defibrotide has been granted orphan drug designation by the EC and the Korean Ministry of Food and Drug Safety to treat and prevent VOD, by the Commonwealth of Australia-Department of Health for the treatment of VOD and by the EC for the prevention of aGvHD. Vyxeos has been granted orphan drug designation by the EC until August 2028.
Pharmaceutical Pricing, Reimbursement by Government and Private Payors and Patient Access
Pricing and Reimbursement
Successful commercialization of our products depends in significant part on adequate financial coverage and reimbursement from third party payors, including governmental payors (such as the Medicaid and Medicare programs in the U.S.), managed care organizations and private health insurers. Third party payors decide which drugs will be reimbursed and establish reimbursement and co-pay levels and conditions for reimbursement. Third party payors are increasingly challenging the prices charged for medical products and services by examining their cost effectiveness, as demonstrated in pharmacoeconomic and/or clinical studies, in addition to their safety and efficacy. In some cases, for example, third party payors try to encourage the use of less expensive products, when available, through their prescription benefits coverage and reimbursement, co-pay and prior authorization policies. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the price or reimbursement rate that the payor will pay for the product once coverage is approved. Third party payors may require prior approval before covering a specific product, or may require patients and health care providers to try other covered products first. Third party payors may also limit coverage to specific products on an approved list, or formulary, which might not include all of the approved products for a particular indication. In many instances,For certain categories of products, third party payors, now requireprincipally through contracted pharmacy benefit managers, or PBMs, negotiate rebates with some drug manufacturers in exchange for including a specific productinclusion of products on their formularies. Informularies in specific positions or coverage criteria. Beginning in the past,third quarter of 2019, we have not enteredbeen entering into such arrangementsagreements with third party payorscertain PBMs to provide rebates for any of our products. The increasing pressure of the pharmaceuticalproducts where coverage environment may lead uswas provided and products were listed in certain formulary positions, among other conditions. We expect to do soenter into additional agreements in the future.2020.
Medicaid is a joint federal and state program that is administered by the states for low‑income and disabled beneficiaries. Medicare is a federal program that is administered by the federal government covering individuals age 65 and over as well as those with certain disabilities. Medicare Part B pays physicians who administer our products. Under the Medicaid Drug Rebate program, as a condition of having federal funds being made available to the states for our drugs under Medicare Part B, we are required to pay a rebate to each state Medicaid program for our covered outpatient drugs that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program. Medicaid rebates are based on pricing data we report on a monthly and quarterly basis to the U.S. Centers for Medicare & Medicaid Services, or CMS, the federal agency that administers the Medicaid Drug Rebate program, several state Medicaid supplemental rebate programs and other governmental pricing programs. These data include the average manufacturer price and, in the case of innovator products, the best price for each drug which, in general, represents the lowest price available from the manufacturer to any entity in the U.S. in any pricing structure, calculated to include all applicable sales and associated rebates, discounts and other price concessions. For the federal government to determine Medicare Part B payments to physicians, we are required to provide average sales price, or ASP, information for certain of our products to the CMS on a quarterly basis. The average sales priceASP is calculated based on a statutorily defined formula as well as regulations and interpretations of the statute by CMS. This information is used to compute Medicare payment rates, with rates for Medicare Part B drugs outside the hospital outpatient setting and in the hospital outpatient setting consisting of ASP plus a specified percentage. If we become aware that our reporting for a prior quarter was incorrect, or has changed as a result of recalculation of the pricing data, we are obligated to resubmit the corrected data for up to three years after those data originally were due.
23
Federal law requires that any company that participates in the Medicaid Drug Rebate program also participate in the Public Health Service’s 340B program, or the 340B program, in order for federal funds to be available for the manufacturer’s drugs under Medicaid and Medicare Part B. The 340B program, which is administered by the Health Resources and Services Administration, or HRSA, requires participating manufacturers to agree to charge statutorily defined covered entities no more than the 340B “ceiling price” for the manufacturer’s covered drugs used in an outpatient setting. These 340B covered entities include a variety of community health clinics and other entities that receive health services grants from the Public Health Service, as well as hospitals that serve a disproportionate share of low-income patients. The 340B ceiling price is calculated using a statutory formula, which is based on the average manufacturer price and rebate amount for the covered outpatient drug as calculated under the Medicaid Drug Rebate program, and in general, products subject to Medicaid price reporting and rebate liability are also subject to the 340B ceiling price calculation and discount requirement. A new regulation regarding the calculation of the 340B ceiling price and the imposition of civil monetary penalties on manufacturers that knowingly and intentionally overcharge covered entities became effective on January 1, 2019. We also are required to report our 340B ceiling prices to HRSA also has announced that it will begin to implementon a ceiling price reporting requirement related to the 340B program during the first quarter of 2019.quarterly basis. In addition, legislation may be introduced that, if passed, would further expand the 340B program to additional covered entities or would require participating manufacturers to agree to provide 340B discounted pricing on drugs used in an inpatient setting.
In order to be eligible to have our products paid for with federal funds under the Medicaid and Medicare Part B programs and purchased by certain federal agencies and grantees, we also participate in the U.S. Department of Veterans Affairs, or VA, Federal Supply Schedule, or FSS, pricing program. Under this program, we are obligated to make our products available for procurement on an FSS contract under which we must comply with standard government terms and conditions and charge a price to certain federal agencies that is no higher than the statutory Federal Ceiling Price, or FCP. The FCP is based on the non-
In addition, in the U.S., drug pricing by pharmaceutical companies is currently, and is expected to continue to be, under close scrutiny, including with respect to companies that have increased the price of products after acquiring those products from other companies. There are numerous ongoing efforts at the federal and state level seeking to indirectly or directly regulate drug prices to reduce overall healthcare costs using tools such as price ceilings, value-based pricing and increased transparency and disclosure obligations. The House Oversight Committee of the 116th Congress has announced an investigation into the actions of a number of pharmaceutical companies in raising prescription drug prices in the U.S. Several states have passed or are considering legislation that requirespurports to require companies to report proprietary pricing information. For example, in 2017, California adopted a prescription drug price transparency state bill requiring advance notice and an explanation for price increases of certain drugs that exceed a specified threshold. Similar bills have been previously introduced at the federal level and additional legislation could be introduced this year.
Similar to what is occurring in the U.S., political, economic and regulatory developments outside of the U.S. are also subjecting the healthcare industry to fundamental changes and challenges. Pressure by governments and other stakeholders on prices and reimbursement levels continue to exist. In various EU member states we expect to be subject to continuous cost-cutting measures, such as lower maximum prices, lower or lack of reimbursement coverage and incentives to use cheaper, usually generic, products as an alternative. Health technology assessment, or HTA, of medicinal products is becoming an increasingly common part of the pricing and reimbursement procedures in some EU member states, including countries representing major markets. The HTA process, which is governed by the national laws of these countries, is the procedure according to which the assessment of the public health impact, therapeutic impact and the economic and societal impact of use of a given medicinal product in the national healthcare systems of the individual country is conducted. HTA generally compares attributes of individual medicinal products, as compared with other treatment options available on the market. The outcome of HTA regarding specific medicinal products will often influence the pricing and reimbursement status granted to these medicinal products by the competent authorities of individual EU member states. On January 31, 2018, the EC adopted a proposal for an HTA regulation intended to boost cooperation among EU member states in assessing health technologies, including new medicinal products. The proposal provides that EU member states will be able to use common HTA tools, methodologies, and procedures across the EU. Individual EU member states will continue to be responsible for assessing non-clinical (e.g., economic, social and ethical) aspects of health technologies, and making decisions on pricing and reimbursement.
In the EU, our products are marketed through various channels and within different legal frameworks. The making available or placing on the EU market of unauthorized medicinal products is generally prohibited. However, the competent authorities of the EU member states may exceptionally and temporarily allow and reimburse the supply of such unauthorized products, either on a named patient basis or through a compassionate use process, to individual patients or a group of patients with a chronically or seriously debilitating disease or whose disease is considered to be life-threatening, and who cannot be treated satisfactorily by an authorized medicinal product. Such reimbursement may no longer be available if authorization for named patient or compassionate use programs expire or areis terminated or if marketing authorization is granted for the product.
24
In othersome EU member states, authorization and reimbursement policies may also delay commercialization of our products, or may adversely affect our ability to sell our products on a profitable basis. After initial price and reimbursement approvals, reductions in prices and changes in reimbursement levels can be triggered by multiple factors, including reference pricing systems and publication of discounts by third party payors or authorities in other countries. In the EU, prices can be reduced further by parallel distribution and parallel trade, or arbitrage between low-priced and high-priced EU member states.
For more information, see the risk factors under the headings “Adequate coverage and reimbursement from third party payors may not be available for our products, which could diminish our sales or affect our ability to sell our products profitably,” “The pricing of pharmaceutical products has come under increasing scrutiny as part of a global trend toward healthcare cost containment and resulting changes in healthcare law and policy may impact our business in ways that we cannot currently predict, which could have a material adverse effect on our business and financial condition” and “If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate program or other governmental pricing programs, we could be subject to additional reimbursement requirements, penalties, sanctions and fines” fines, which could have a material adverse effect on our business, financial condition, results of operations and growth prospects” in Part I, Item 1A of this Annual Report on Form 10‑K.
Patient Assistance Programs
We have various patient assistance programs to help patients access our products, including patient assistance programs, which include co-pay coupons for certain of our products, services that help patients determine their insurance coverage for our products, and a free product program. We also make grants to independent charitable foundations that help financially needy patients with their premium, and co-pay and co-insurance obligations. There has been enhanced scrutiny of company-sponsored patient assistance
programs, including insurance premium and co-pay assistance programs and donations to third-party charities that provide such assistance, as well as reimbursement support offerings.
The OIG has established guidelines that permitfor pharmaceutical manufacturers towho make donations to charitable organizations providing co-pay assistance to Medicare patients,patients. Such donations are unlikely to run afoul of the anti-kickback laws provided that suchthe organizations receiving donations, among other things, are bona fide charities, are entirely independent of and not controlled by the manufacturer, provide aid to applicants on a first-come basis according to consistent financial criteria, and do not link aid to use of a donor’s product. In 2016 and 2017, we received subpoenas from the U.S. Attorney’s Office for the District of Massachusetts requesting documents related to our support of charitable organizations that provide financial assistance to Medicare patients. In April 2018,2019, we reached anfinalized our civil settlement agreement in principle with the DOJ on a proposal for a civil settlement of potential claims by the DOJ in the amount of $57.0 million, subject to accrual of interest on the settlement amount from the date of the agreement in principle, negotiation of a definitive settlement agreement and other contingencies, which we expect will include entryOIG, and entered into a corporate integrity agreement requiring us to maintain our ongoing corporate compliance program and obligating us to implement or continue, as applicable, a set of defined corporate integrity activities to ensure compliance with OIG’s policies around charitable contributions for a period of five years from the OIG. For more information, see Note 12, Commitments and Contingencies—Legal Proceedingseffective date of the Notes to Consolidated Financial Statements included in Part IV of this Annual Report on Form 10-K.corporate integrity agreement.
U.S. Healthcare Reform
The Patient Protection and Affordable Care Act, as amended by the Healthcare and Education Reconciliation Act of 2010, which we refer to together as the Healthcare Reform Act, was intended to expand healthcare coverage within the U.S., primarily through the imposition of health insurance mandates on employers and individuals, the provision of subsidies to eligible individuals enrolled in plans offered on the health insurance exchanges, and the expansion of the Medicaid program. This law has substantially changed the way healthcare is financed by both governmental and private insurers and significantly impacts the pharmaceutical industry. Changes that may affect our business include those governing enrollment in federal healthcare programs, reimbursement changes, benefits for patients within a coverage gap in the Medicare Part D prescription drug program (commonly known as the “donut hole”), rules regarding prescription drug benefits under the health insurance exchanges, changes to the Medicaid Drug Rebate program, expansion of the 340B program, and fraud and abuse and enforcement. These changes have impacted previously existing government healthcare programs and have resulted in the development of new programs, including Medicare payment for performance initiatives and improvementsinitiatives.
Certain provisions of the Healthcare Reform Act have been subject to the physician quality reporting system and feedback program.
Employees
As of February 19, 2019,18, 2020, we had approximately 1,3601,620 employees worldwide. We consider our employee relations to be good.
25
Environment, Health and Safety
Our operations are subject to complex and increasingly stringent environmental, health and safety laws and regulations in the countries where we operate and, in particular, in Italy and Ireland where we have manufacturing facilities. Our manufacturing activities involve the controlled storage, use and disposal of chemicals and solvents. Environmental and health and safety authorities in Italy and Ireland administer laws governing, among other matters, the emission of pollutants into the air (including the workplace), the discharge of pollutants into bodies of water, the storage, use, handling and disposal of hazardous substances, the exposure of persons to hazardous substances, and the general health, safety and welfare of employees and members of the public. In certain cases, such laws, directives and regulations may impose strict liability for pollution of the environment and contamination resulting from spills, disposals or other releases of hazardous substances or waste. Costs, damages and/or fines may result from the presence, investigation and remediation of such contamination at properties currently or formerly owned, leased or operated by us or at off-site locations, including where we have arranged for the disposal of hazardous substances or waste. In addition, we may be subject to third party claims, including for natural resource damages, personal injury and property damage, in connection with such contamination.
About Jazz Pharmaceuticals plc
Jazz Pharmaceuticals plc was formed under the laws of Ireland (registered number 399192) as a private limited liability company in March 2005 under the name Azur Pharma Limited and was subsequently re-registered as a public limited company under the name Azur Pharma Public Limited Company, or Azur Pharma, in October 2011. On January 18, 2012, the businesses of Jazz Pharmaceuticals, Inc. and Azur Pharma were combined in a merger transaction, in connection with which Azur Pharma was re-named Jazz Pharmaceuticals plc and we became the parent company of and successor to Jazz Pharmaceuticals, Inc.
Our predecessor, Jazz Pharmaceuticals, Inc., was incorporated in California in March 2003 and was reincorporated in Delaware in January 2004.
Available Information
The mailing address of our headquarters is Fifth Floor, Waterloo Exchange, Waterloo Road, Dublin 4, Ireland, and our telephone number at that location is 353-1-634-7800. Our website is www.jazzpharmaceuticals.com.
We file or furnish pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended, as applicable, our Annual Reports on Form 10-K,10‑K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, amendments to those reports, proxy statements and other information electronically with the SEC. Through a link on our website, we make copies of our periodic and current reports, amendments to those reports, proxy statements and other information available, free of charge, as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Information found on, or accessible through, our website is not a part of, and is not incorporated into, this Annual Report on Form 10-K.10‑K.
| Item 1A. | Risk Factors |
We have identified the following risks and uncertainties that may have a material adverse effect on our business, financial condition or results of operations. The risks described below are not the only ones we face. Additional risks not presently known to us or that we currently believe are immaterial may also significantly impair our business operations. Our business could be harmed by any of these risks. The trading price of our ordinary shares could decline due to any of these risks, and you may lose all or part of your investment. In assessing these risks, you should also refer to the other information contained in this Annual Report on Form 10-K,10‑K, including our consolidated financial statements and accompanying notes.
Risks Related to XyremOur Lead Products and Our Other Marketed ProductsProduct Candidates
Our current business is substantially dependent on Xyrem® (sodium oxybate) oral solution, is the only product approved by the U.S. Food and Drug Administration, or FDA, and marketed in the U.S. for the treatment of both cataplexy and excessive daytime sleepiness, or EDS, in adult and pediatric patients with narcolepsy. Xyrem is our largest selling product, and our financial results are significantly influenced by sales of Xyrem, which accounted for 75% and 74% of our net product sales for the years ended December 31, 2018 and 2017, respectively. Our future plans assume thatXyrem. A significant decline in sales of Xyrem will increase, but there is no guarantee that we can maintain sales of Xyrem at or near current levels, or that Xyrem sales will continue to grow. We have periodically increased the price of Xyrem, most recently in January 2019, and there is no guarantee that price adjustments we have taken or may take in the future will not negatively affect Xyrem sales volumes and revenues from Xyrem.
26
sales is subject to a number of risks and uncertainties as discussed in greater detail below, including those related to the introduction of authorized generic and generic versions of sodium oxybate and/or new products for treatment of cataplexy and/or excessive daytime sleepiness, or EDS, in narcolepsy in the U.S. market, increased pricing pressure from, changes in policies by, or restrictions on reimbursement imposed by, third party payors, challenges to our intellectual property around Xyrem, and continued acceptance of Xyrem by physicians and patients.
As for other products and product candidates in our sleep and neuroscience therapeutic area, we obtained approval of Sunosi® (solriamfetol) in 2019 in the U.S. and in January 2020 in the European Union, or EU, for the treatment of EDS associated with narcolepsy or obstructive sleep apnea, or OSA. Our ability to realize the anticipated benefits from our investment in Sunosi is subject to a number of risks and uncertainties, including market acceptance of Sunosi; our ability, in a competitive retail pharmacy market, to differentiate Sunosi from other products that are prescribed to treat excessive sleepiness in patients with OSA or EDS in patients with narcolepsy; the availability of adequate formulary positions and pricing and adequate coverage and reimbursement by government programs and other third party payors, including the impact of future coverage decisions by payors; restrictions on permitted promotional activities based on any additional limitations on the labeling for the product that may be required by the FDA in the future and any such limitations that may be required by the European Commission, or the EC, or other regulatory authority on any approved labeling;and our ability to satisfy the FDA’s post-marketing requirements.
We also submitted a new drug application, or NDA, in January 2020 for marketing approval of JZP-258, an oxybate product candidate that contains 92%, or approximately 1,000 to 1,500 milligrams per day, less sodium than Xyrem, for the treatment of cataplexy and EDS in narcolepsy patients seven years of age and older. Our future plans assume that, if approved, JZP-258 may be prescribed to patients as a safer and clinically superior alternative to Xyrem as well as being prescribed to patients who may otherwise be ineligible to take Xyrem based on its high sodium content. Our ability to realize the anticipated benefits from our investment in JZP-258 is subject to a number of risks and uncertainties, including delay or failure in obtaining approval of JZP-258; our receipt of approval for narrower indications than sought or burdens in the approved label; obtaining FDA approval of a risk evaluation and mitigation strategy, or REMS; obtaining and maintaining adequate coverage and reimbursement for JZP-258; the introduction of new products in the U.S. market that competescompete with JZP-258 in the treatment of cataplexy and/or EDS in narcolepsy, including generic or authorized generic versions of sodium oxybate or new sodium oxybate products; and acceptance of JZP-258 by payors, physicians and patients.
If we are unable to successfully commercialize Sunosi and/or JZP-258 (if approved), or if sales of Sunosi and JZP-258 do not reach the levels we expect, our anticipated revenue from our sleep therapeutic area will be negatively affected, which would have a material adverse effect on our business, financial condition, results of operations and growth prospects.
The introduction of new products in the U.S. market that compete with, or otherwise disruptsdisrupt the market for, Xyremour oxybate products and product candidates would adversely affect sales of Xyrem.our oxybate products and product candidates.
While Xyrem is currently the only product approved by the U.S. Food and Drug Administration, or FDA, and marketed in the U.S. for the treatment of both cataplexy and EDS in both adult and pediatric patients with narcolepsy, we and others have launched and may in the future launch products as treatment optionsthat are competitive with or disrupt the market for Xyrem.
For example, in cataplexy and/or EDS in narcolepsy, including other branded sodium oxybate products and other new and existing branded market entrants. In addition,the future, we expect Xyrem willto face competition from generics and authorized generics. We expect that the approval and launch of any other sodium oxybate or alternative product that treats narcolepsy, or the launch of an authorized generic product, or AG Product, or otherand generic versionversions of Xyrem, couldsodium oxybate. Nine companies have a material adverse effect on our sales of and revenues from Xyrem and on our business, financial condition, results of operations and growth prospects.
27
Products and maintain FDA approval of its ANDA. In January 2017, the FDA approved West-Ward’s ANDA and tentatively approved two additional ANDAs for generic sodium oxybate products and we believe that it is likely that the FDA will approve or tentatively approve the additional ANDAs that have been filed.under our settlement agreements are subject to acceleration under certain circumstances.
Any ANDA holder launching an AG Product or another generic sodium oxybate product will independently establish the price of the AG Product and/or its own generic sodium oxybate product. Generic competition often results in decreases in the prices at which branded products can be sold. After any introduction of a generic product, whether or not it is an AG Product, a significant percentage of the prescriptions written for Xyrem maywill likely be filled with the generic product. Certain U.S. state laws allow for, and in some instances in the absence of specific instructions from the prescribing physician mandate, the dispensing of generic products rather than branded products when a generic version is available. This would result in reduction in sales of, and revenue from, Xyrem, although we would continue to receive royaltyroyalties and other revenue based on sales of an AG Product in
accordance with the terms of our settlement agreements. For more information on the impact of generic competition, see the risk factors under the heading “Adequate coverage and reimbursement from third party payors may not be available for our products, which could diminish our sales or affect our ability to sell our products profitably” and “The pricing of pharmaceutical products has come under increasing scrutiny as part of a global trend toward healthcare cost containment and resulting changes in healthcare law and policy may impact our business in ways that we cannot currently predict, which could have a material adverse effect on our business and financial condition” in this Part I, Item 1A.
It is possible that additional companies may file ANDAs seeking to market a generic version of Xyrem which could lead to additional patent litigation or challenges with respect to Xyrem. Such patent litigation or challenges could potentially trigger acceleration of the launch dates in our settlement agreements. Foragreements if, for example, the launch dates in our settlement agreements would be accelerated if a new ANDA filerpatents covering Xyrem were to obtain a final decision prior to January 1, 2023 that all unexpired claims of the Xyrem patents are invalid and/or unenforceable.invalidated. Alternatively, the launch dates in our settlement agreements could be accelerated if a new ANDA filer were to obtain FDA approval for its sodium oxybate product, and launch its generic product through a generic sodium oxybate REMS before the entry dates specified in our settlement agreements, if, for example, we are unable to obtain an injunction or because that party launches “at risk” of being held liable for damages for patent infringement.agreements. It is also possible that we could enter into a settlement agreement with a future ANDA filer that would permit such filer to enter the market on or prior to the launch date(s) in our settlement agreements. If a company launches a generic or authorized generic sodium oxybate product in any of these scenarios, except in limited circumstances related to an “at risk” launch, the launch date for West-Ward’sHikma’s AG Product would be accelerated to a date on or prior to the date of such entry, which could lead to acceleration of the other settling ANDA filers’ AG Product and generic sodium oxybate product launch dates as described above. For further discussion of Xyrem-related patent matters, see the risk factors under the heading “Risks Related to Our Intellectual Property” in this Part 1, Item 1A.
Another circumstance that could trigger acceleration of West-Ward’sHikma’s launch date for an AG Product, which would also accelerate Amneal, Lupin and Par’s launch dates for their AG Products and ultimately could lead to acceleration of the other settling ANDA filers’ launch dates for their generic sodium oxybate products, is a substantial reduction in Xyrem net sales. Such a reduction could occur under various circumstances, including if we introduce, or a third party introduces, a product to treat EDS or cataplexy in narcolepsy that leads to a substantial decline in Xyrem net sales prior to January 1, 2023. For example, we are developing JZP-258, an oxybate product candidate that contains 90% less sodium than Xyrem. Given the well-accepted relationship between dietary sodium and blood pressure as well as published hypertension guidelines underscoring that excessive consumption of sodium is independently associated with an increased risk of stroke, cardiovascular disease and other adverse outcomes, we believe that lower sodium intake would be beneficial for patients. JZP-258 is being developed for the treatment of both cataplexy and EDS in narcolepsy as well as for other conditions, and, subject to the results of our Phase 3 clinical trial in narcolepsy, we expect to file an NDA for approval of this product by as early as the end of 2019. Other companies may similarly develop a sodium oxybate product for treatment of narcolepsy, using an alternative formulation or a different delivery technology, and seek approval in the U.S. using an NDA approval pathway under Section 505(b)(2) and referencing the safety and efficacy data for Xyrem, which could lead to additional patent litigation or challenges.Xyrem. We are aware that a company called Avadel Pharmaceuticals plc, or Avadel, is conducting a Phase 3 clinical trial of a once-nightly formulaformulation of sodium oxybate which uses its proprietary technology for the treatment of EDS and cataplexy in patients with narcolepsy, and that Avadel has indicated that it intends to seek approval using the Section 505(b)(2) approval pathway. Approvalpathway referencing the safety and successful commercializationefficacy data for Xyrem. Xyrem may also face competition from new branded entrants to treat EDS in narcolepsy such as pitolisant. Other companies have announced that they have product candidates in various phases of development to treat the symptoms of narcolepsy, such as Axsome Therapeutics, Inc.’s reboxetine.
We expect that, if approved, JZP-258, will face competition similar to that described above for Xyrem, including from generic or Avadel’sauthorized generic sodium oxybate formulation,product or any other new non-genericbranded entrants in narcolepsy such as Avadel. Avadel has announced that it has obtained an orphan drug designation from the FDA for its once-nightly sodium oxybate formulation. To obtain orphan drug exclusivity upon approval, Avadel will have to show clinical superiority to Xyrem and possibly to JZP-258, if approved. If the FDA approves Avadel’s product and grants it orphan drug exclusivity before we obtain approval for JZP-258, there is a risk that JZP-258 will not be approvable for a narcolepsy indication for seven years unless it can establish clinical superiority to Avadel’s product. We cannot predict the timing of Avadel’s submission or other producthow the FDA will evaluate any clinical superiority arguments that either company may make, but a delay in approval or inability to obtain approval for JZP-258 could materially and adversely affect our business, financial condition, results of operations and growth prospects.
Moreover, non-oxybate products intended for the treatment of EDS or cataplexy in narcolepsy, patientsincluding new market entrants, even if not directly competitive with Xyrem or JZP-258 (if approved), could negatively impact our ability to maintainhave the effect of changing treatment regimens and growpayor or formulary coverage of Xyrem or JZP-258 in favor of other products, and indirectly materially and adversely affect sales of Xyrem.
28
The distribution and sale of Xyremour oxybate products are subject to significant regulatory restrictions, including the requirements of a REMS, and these regulatory requirements subject us to risks and uncertainties, any of which could negatively impact sales of Xyrem.Xyrem and if approved, JZP-258.
The active pharmaceutical ingredient, or API, of Xyrem, sodium oxybate, is the sodium salt of gamma-hydroxybutyric acid, or GHB, a central nervous system depressant known to be associated with facilitated sexual assault as well as with respiratory depression and other serious side effects. As a result, the FDA requires that we maintain a REMS with elements to assure safe use, or ETASU, for Xyrem to help ensure that the benefits of the drug in the treatment of cataplexy and EDS in narcolepsy outweigh the serious risks of the drug. The REMS imposes extensive controls and restrictions on the sales and marketing of Xyrem that we are responsible for implementing. For example, under the Xyrem REMS, all of the Xyrem sold in the U.S. must be dispensed and shipped directly to patients or caregivers through a central pharmacy, and may not be stocked in retail pharmacies. Physicians and patients must complete an enrollment process prior to fulfillment of Xyrem prescriptions, and each physician and patient must receive materials concerning the serious risks associated with Xyrem before the physician can prescribe, or a patient can receive, the product. The central certified pharmacy must monitor and report instances of patient or prescriber behavior giving rise to a reasonable suspicion of abuse, misuse or diversion of Xyrem, and maintains enrollment and prescription monitoring information in a central database. Any failure to complydemonstrate our substantial compliance with our REMS obligations, or a determination by the FDA that the Xyrem REMS is not meeting its goals, could result in enforcement action by the FDA, lead to changes in our Xyrem REMS obligations, negatively affect sales of Xyrem, result in additional costs and expenses for us and/or require us to invest a significant amount of resources, any of which could materially and adversely affect our business, financial condition, results of operations and growth prospects.
The FDA has stated that it will evaluate the Xyrem REMS on an ongoing basis and will require modifications as may be appropriate. We cannot predict whether the FDA will request, seek to require or ultimately require modifications to, or impose additional requirements on, the Xyrem REMS, including in connection with the submission of applications for new oxybate products including JZP-258, new oxybate indications, or the introduction of authorized generics, or to accommodate generics, or whether the FDA will approve modifications to the Xyrem REMS that we consider warranted in connection with the submission of applications for new oxybate products.products including JZP-258. Any modifications approved, required or rejected by the FDA could change the safety profile of Xyrem, and have a significant negative impact in terms of product liability, public acceptance of Xyrem as a treatment for cataplexy and EDS in narcolepsy, and prescribers’ willingness to prescribe, and patients’ willingness to take, Xyrem, any of which could have a material adverse effect on our Xyrem business. Modifications approved, required or rejected by the FDA could also make it more difficult or expensive for us to distribute Xyrem, make distribution easier for sodium oxybate competitors, disrupt continuity of care for Xyrem patients and/or negatively affect sales of Xyrem.
We depend on outside vendors, including the central certified pharmacy, to implement the requirements of the Xyrem REMS. We have an exclusive agreement with Express Scripts Specialty Distribution Services, Inc., the central pharmacy for Xyrem, which expires on July 1, 2019, but we expect to exercise our right to extend the agreement for an additional year. The agreement may be terminated by either party at any time without cause on 180 days’ prior written notice to the other party. If the central pharmacy fails to meet the requirements of the Xyrem REMS applicable to the central pharmacy or otherwise does not fulfill its contractual obligations to us, provides timely notice that it wantsmoves to terminate our agreement, refuses or fails to adequately serve patients, or fails to promptly and adequately address operational challenges or challenges in implementing REMS modifications, the fulfillment of Xyrem prescriptions and our sales would be adversely affected. If we change to a new central pharmacy, new contracts might be required with government payors and other insurers who pay for Xyrem, and the terms of any new contracts could be less favorable to us than current agreements. In addition, any new central pharmacy would need to be registered with the U.S. Drug Enforcement Administration, or DEA, and certified and would also need to implement
the particular processes, procedures and activities necessary to distribute Xyrem under the Xyrem REMS. Transitioning to a new pharmacy could result in product shortages, which would negatively affect sales of Xyrem, result in additional costs and expenses for us and/or take a significant amount of time, any of which could materially and adversely affect our business, financial condition, results of operations and growth prospects.
29
We may face pressure to further modify the Xyrem REMS or to license or share intellectual property pertinent to the Xyrem REMS, including proprietary data required for the safe distribution of sodium oxybate, in connection with the FDA’s approval of the generic sodium oxybate REMS or otherwise. Our settlement agreements with ANDA filers do not directly impact the FDA’s waiver of the single shared system REMS requirement, any other ANDA or NDA filer’s ability to develop and implement the generic sodium oxybate REMS for its generic sodium oxybate product, or our ability to take any action with respect to the safety of the generic sodium oxybate REMS. We cannot predict the outcome or impact on our business of any future action that we may take with respect to the FDA’s waiver of the single shared system REMS requirement, its approval and tentative approval of generic versions of Xyremsodium oxybate or the consequences of distribution of sodium oxybate through the generic sodium oxybate REMS approved by the FDA or another separate REMS.
REMS programs have increasingly drawn public scrutiny from the U.S. Congress, the Federal Trade Commission, or FTC, and the FDA, with allegations that such programs are used as a means of improperly blocking or delaying competition. TheIn December 2019, as part of the Further Consolidated Appropriations Act of 2020, the U.S. Congress for example, has introduced proposedpassed legislation aimed at preventingknown as the Creating and Restoring Equal Access To Equivalent Samples Act, or CREATES. CREATES is intended to prevent companies from using REMS and other restricted distribution programs as a means to deny potential competitors access to product samples needed for bioequivalence testing. The FDA has stated that it will seekare reasonably necessary to coordinate withconduct testing in support of an application that references a listed drug or biologic, and provides such potential competitors a potential private right of action if the FTC in identifying and publicizing practices the FTC findsinnovator fails to be anticompetitive and has further stated thattimely provide samples upon request. CREATES also grants the FDA has concerns related to the role of REMS programs in delayingadditional authority regarding approval of generic products. For example, in May 2018, FDA published a list of companies that it said had potentially been blocking access to the samples of their branded products including one of our subsidiaries that sells FazaClo® (clozapine, USP) through a REMS program. with REMS.
It is possible that the FTC, the FDA, other governmental authorities or other third parties could claim that, or launch an investigation into whether, we are using our REMS programs in an anticompetitive manner or have engaged in other anticompetitive practices. The Federal Food, Drug and Cosmetic Act further states that a REMS ETASU shall not be used by an NDA holder to block or delay generic drugs or drugs covered by an application under Section 505(b)(2) from entering the market. In its 2015 letter approving the Xyrem REMS, the FDA expressed concern that we were aware that the Xyrem REMS could have the effect of blocking or delaying generic competition. We cannot predict whether we would face a government investigation or a complaint by a third party premised on a claim that the Xyrem REMS is blocking competition, or the outcome or impact of any such claim.
Pharmaceutical companies, including their agents and employees, are required to monitor adverse events occurring during the use of their products and report them to the FDA. The patient counseling and monitoring requirements of the Xyrem REMS provide more extensive information about adverse events experienced by patients taking Xyrem, including deaths, than is generally available for other products that are not subject to similar REMS requirements. As required by the FDA and other regulatory agencies, the adverse
event information that we collect for Xyrem is regularly reported to the FDA and could result in the FDA requiring changes to Xyrem labeling, including additional warnings or additional boxed warnings, or requiring us to take other actions that could have an adverse effect on patient and prescriber acceptance of Xyrem. As required by the FDA, Xyrem’s current labeling includes a boxed warning regarding the risk of central nervous system depression and misuse and abuse.
Any failure to demonstrate our substantial compliance with the REMS or any other applicable regulatory requirements to the satisfaction of the FDA or another regulatory authority could result in such regulatory authorities taking actions in the future which could have a material adverse effect on Xyrem sales and therefore on our business, financial condition, results of operations and growth prospects. For more information, see the risk factor under the heading “In addition to those specifically described in other risk factors, we are subject to significant ongoing regulatory obligations and oversight, which may result in significant additional expense and limit our ability to commercialize our products” in this Part I, Item 1A.
While Xyrem remains our largest product, our success also depends on our ability to effectively commercialize products outside our other products.sleep and neuroscience therapeutic area.
In addition to Xyrem and our other sleep and neuroscience products and product candidates, we are commercializing a portfolio of products, including our other lead marketed products, Defitelio, Erwinaze Defitelio and Vyxeos, and we are making significant investments in maximizing the value and therapeutic reach of Defitelio and Vyxeos by conducting additional research and development activities, which include generating additional supportive clinical data and seeking regulatory approval for new indications, as appropriate. OurVyxeos. An inability to effectively commercialize our lead marketed productsDefitelio and Vyxeos and to maximize their potential where possible through successful research and development activities, and an inability to retain marketing rights to Erwinaze after 2020, could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
Defitelio
Our ability to maintain and successfully and sustainably grow sales of and to realize the anticipated benefits from our investment in Defitelio®(defibrotide sodium) is subject to a number of risks and uncertainties, including the following as well as other risks and uncertainties described elsewhere in this Part I, Item 1A:
;the limited experience of, and need to educate, physicians in recognizing, diagnosing and treating hepatic veno-occlusive disease, or VOD, particularly in adults;
the possibility that physicians recognizing VOD symptoms may not initiate or may delay initiation of treatment while waiting for those symptoms to improve, or may terminate treatment before the end of the recommended dosing schedule;
the limited size of the population of VOD patients who are indicated for treatment with Defitelio (particularly if changes in HSCT hematopoietic stem cell transplantation treatment protocols reduce the incidence of VOD diagnosis);
30
anticipated revenue from the product will be negatively affected and treatmentour business, financial condition, results of serious diseases associated with endothelial cell damage, including an ongoing Phase 3 clinical trial in prevention of VOD in high-risk patients following HSCT, ongoing Phase 2 trials in prevention of acute Graft versus Host Disease following allogeneic HSCT,operations and planned Phase 2 trials in the treatment of transplant-associated thrombotic microangiopathy and the prevention of chimeric antigen receptor T-cell therapy-, or CAR-T-, associated neurotoxicity. In addition to clinical trials we are sponsoring, there are more than 20 investigator-sponsored trials ongoing in the U.S. and EU to evaluate defibrotide in multiple conditions. If these development activities are unsuccessful, our growth prospects could be materially adversely affected.
Erwinaze
Erwinaze® (asparaginase Erwinia chrysanthemi), which is approved to treat a limited population of patients with acute lymphoblastic leukemia, or ALL, who have developed hypersensitivity to E. coli-derived asparaginase, is licensed from, and manufactured by, a single source, Porton Biopharma Limited, or PBL, a company that is wholly owned by the UK Department of Health and Social Care. Our license and supply agreement with PBL, which includes an exclusive right to market, sell or distribute Erwinaze, an exclusive license to Erwinaze trademarks, and a non-exclusive license to PBL’s manufacturing know-how, will expire on December 31, 2020. Unless we and PBL enter into a new agreement, we will lose our licensed rights to exclusively market Erwinaze effective December 31, 2020, other than our right to sell certain Erwinaze inventory for a post-termination sales period of Defitelio do12 months and certain other post-termination rights, including but not reach the levelslimited to intellectual property and data ownership. In such event, we expect, our anticipated revenue frommay not be able to replace the product sales we would be negatively affected, which could have a material adverse effect onlose from Erwinaze, and our business, financial condition, results of operations and growth prospects.
Erwinaze and a barrier to increasing sales is PBL’s inability to consistently supply product that meets specifications in quantities that are adequate to meet market demand, as discussed elsewhere in these risk factors. Such supply instability will continue to adversely impact our ability to generate sales of and revenues from Erwinaze and our business, financial condition, results of operations and growth prospects could be materially adversely affected. Other challenges facing Erwinaze include the limited population of patients with ALL, and the incidence of hypersensitivity reactions to E. coli-derived asparaginase within that population; the development and/or approval of new asparaginase treatments or treatment protocols for ALL that may not include asparaginase-containing regimens and prescribers’ use of alternate methods to address hypersensitivity reactions; difficulties with obtaining and maintaining favorable pricing and reimbursement arrangements; and potential competition from future biosimilar products.
Vyxeos
our ability to differentiate Vyxeos from other liposomal chemotherapies and generically available chemotherapy combinations with which physicians and treatment centers are more familiar;
countries; the increasing complexity of the acute myeloid leukemia, or AML, landscape requiring changes in patient identification and treatment selection, including diagnostic tests and monitoring that clinicians may find challenging to incorporate;
the use of new and novel compounds in AML that are either used off-label or are only approved for use in combination with other agents and that have not been tested in combination with Vyxeos;
the limited size of the population of high-risk AML patients who may potentially be indicated for treatment with Vyxeos, particularly given Vyxeos; the ongoing clinical trials by other companies with the same patient population; and
If sales of Vyxeos do not reach the levels we expect, our anticipated revenue from the product will be negatively affected, which could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
We face substantial competition from other companies, including companies with larger sales organizations and more experience working with large and diverse product portfolios.
Our products compete, and our product candidates may in the future compete, with currently existing therapies, including generic drugs, product candidates currently under development by us and others and/or future product candidates, including new chemical entities that may turn out to be safer or more effective or more convenient than our products. Any products that we develop may be commercialized in competitive markets, and our competitors, which include large global pharmaceutical companies and small research-based companies and institutions, may succeed in developing products that render our products obsolete or noncompetitive.
For a discussiondescription of specific risks relating to the launch of newcompetition that our lead marketed products that treat cataplexy and/and most advanced product candidates face or EDS in narcolepsy, including generic versions of Xyrem or other sodium oxybate products,may face, see the discussion in “Business—Competition” in Part I, Item 1 of this Annual Report on Form 10-K and the risk
31
factor under the heading “The introduction of a new productproducts in the U.S. market that competescompete with, or otherwise disruptsdisrupt the market for, Xyremour oxybate products and product candidates would adversely affect sales of Xyrem” our oxybate products and product candidates” in this Part I, Item 1A. We expect that the approval and launch of any other sodium oxybate or alternative product that treats narcolepsy, or the launch of an AG Product or other generic version of Xyrem, could have a material adverse effect on our sales of and revenues from Xyrem and on our business, financial condition, results of operations and growth prospects.
Adequate coverage and reimbursement from third party payors may not be available for our products, which could diminish our sales or affect our ability to sell our products profitably.
In both U.S. and non-U.S. markets, our ability to successfully commercialize and achieve market acceptance of our products depends in significant part on adequate financial coverage and reimbursement from third party payors. Third party payors, includeincluding governmental payors (such as the Medicare and Medicaid programs in the U.S.), managed care organizations and private health insurers. Without third party payor support,reimbursement, patients may not be able to obtain or afford prescribed medications duemedications. In addition, reimbursement guidelines and incentives provided to barriersprescribing physicians by third party payors may have a significant impact on the prescribing physicians’ willingness and ability to access, includingprescribe our products. The demand for, and the inability to affordprofitability of, our products could be materially harmed if the medication.Medicaid program, Medicare program, other federal healthcare program, or other third party commercial payors in the U.S. or elsewhere deny reimbursement for our products, limit the indications for which our products will be reimbursed, or provide reimbursement only on unfavorable terms.
Third party payors increasingly examine the cost effectivenesscost-effectiveness of pharmaceutical products, in addition to their safety and efficacy, when making coverage and reimbursement decisions. We may need to conduct expensive pharmacoeconomic and/or clinical studies in order to demonstrate the cost-effectiveness of our products. Even with such studies, our products may be considered less safe, less effective or less cost-effective than other products, and third party payors may not provide and maintain coverage and reimbursement for our products. If our competitors offer their products at prices that provide purportedly lower treatment costs than our products, or otherwise suggest that their products are safer, more effective or more cost-effective than our products, this may result in a greater level of access for their products relative to our products, which would reduce our sales and harm our results of operations. In some cases, for example, third party payors try to encourage the use of less expensive generic products through their prescription benefits coverage and reimbursement and co-pay policies. Because some of our products compete in a market with both branded and generic products, obtaining and maintaining access and reimbursement coverage for our products may be more challenging than for products that are new chemical entities for which no therapeutic alternatives exist.
Payors could decide to enter into rebate agreements in order to ensure that patients continue to have access to Xyrem, and to support the long-term success of our sleep franchise, which might result in lower net revenues for Xyrem.
In many countries outside the U.S., procedures to obtain price approvals, coverage and reimbursement can take considerable time after the receipt of marketing approval. Many European countries periodically review their reimbursement of medicinal products, which could have an adverse impact on reimbursement status. In addition, if approved, new products indicated for the treatment of symptoms of narcolepsy, like solriamfetol or pitolisant, could impact access to Xyrem, particularly for newly diagnosed narcolepsy patients, if, for example, payors impose a step edit requiring a narcolepsy patient to try solriamfetol before authorizing payment for Xyrem. For more information on solriamfetol, see the risk factor under the heading “Our future success depends on our ability to successfully developwe expect that legislators, policymakers and obtain and maintain regulatory approvalhealthcare insurance funds in the U.S.EU member states will continue to propose and Europe for our late-stage product candidatesimplement cost-containing measures, such as lower maximum prices, lower or lack of reimbursement coverage and if approved,incentives to successfully launch and commercialize those product candidates.”use cheaper, usually
32
generic, products as an alternative to branded products, to keep healthcare costs down. Moreover, in this Part I, Item 1A.
The pricing of pharmaceutical products has come under increasing scrutiny as part of a global trend toward healthcare cost containment and resulting changes in healthcare law and policy may impact our business in ways that we cannot currently predict, which could have a material adverse effect on our business and financial condition.
Political, economic and regulatory influences are subjecting the healthcare industry in the U.S. to fundamental changes, particularly given the current atmosphere of mounting criticism of prescription drug costs in the U.S. We expect there will
continue to be legislative and regulatory proposals to change the healthcare system in ways that could impact our ability to sell our products profitably. TheFor example, we anticipate that the U.S. Congress, state legislatures, and regulators may adopt or accelerate adoption of new healthcare policies and reforms intended to curb healthcare costs, such as federal and state controls on government-funded reimbursement for drugs (including Medicare, Medicaid) and commercial health plans, new or increased requirements to pay prescription drug rebates and penalties to government health care programs, and additional pharmaceutical cost transparency bills that aim to require drug companies to justify their prices through required disclosures. Additionally, proposals made part of proposed legislation and executive rule-making seek to utilize an “international pricing index” as a benchmark to determine the costs and potentially limit the reimbursement of drugs under Medicare Part B to more closely align with international drug prices. If the U.S. were to move to such a pricing system that were to apply to any of our products, our revenues from U.S. sales of such products could decrease.
Legislative and regulatory proposals that have recently been considered include the potential authorization of prescription drug importation from other countries, legislative proposals to limit the terms of patent litigation settlements with generic sponsors, and proposals to define certain conduct around patenting and new product development as unfair competition. All such considerations may adversely affect our business and industry in ways that we cannot accurately predict.
There is also ongoing activity related to the Patient Protection and Affordable Care Act, as amended by the Healthcare and Education Reconciliation Act of 2010, together, the Healthcare Reform Act. The Healthcare Reform Act has substantially changed the way healthcare is financed by both governmental and private insurers. These changes have impacted previously existing government healthcare programs and have resulted in the development of new programs, including Medicare payment-for-performance initiatives. Additional legislativeSome of the provisions of the Healthcare Reform Act have yet to be fully implemented, and regulatory changes remain possible and appear likely. Their nature and extent are uncertain and they will becertain provisions have been subject to judicial and other challenges. For example, while “repeal and replace”Congressional challenges, as well as efforts by President Trump and the U.S. Congress have failed,current presidential administration to repeal or replace certain aspects of the Healthcare Reform Act have been changed legislatively, such asand to alter the repeal of the requirement that certain individuals who fail to maintain qualifying health coverage for all or part of a year make a tax-based shared responsibility payment commonly referred to as the “individual mandate.” In addition, there have been delays in the implementation of key provisions of the Healthcare Reform Act including the excise tax on generous employer-based health plans.and related laws. We expect that the Healthcare Reform Act as currently enactedand its implementation, efforts to repeal or as it may be amended inreplace, or invalidate, the future,Healthcare Reform Act or portions thereof and other healthcare reform measures that may be adopted in the future could have a material adverse effect on our industry generally and on our ability to maintain or increase sales of our products.
If healthcare policies or reforms intended to curb healthcare costs are adopted or if we experience negative publicity with respect to pricing of our products or the pricing of pharmaceutical drugs generally, the prices that we charge for our products, including Xyrem, may be affected, our commercial opportunity may be limited and/or our revenues from sales of our products may be negatively impacted. We have periodically increased the price of Xyrem, most recently in January 2019,2020, and may do so againthere is no guarantee that we will be able to make similar price adjustments in the future.future or that price adjustments we have taken or may take in the future will not negatively affect Xyrem sales volumes and revenues from Xyrem. We also have made and may in the future make similar price increases on our other products. There is no guarantee that such price adjustmentsincreases will not negatively affect our reputation and our ability to secure and maintain reimbursement coverage for our products, which could limit the prices that we charge for our products, including Xyrem, limit the commercial opportunities for our products and/or negatively impact revenues from sales of our products.
If we become the subject of any future government investigation or U.S. Congressional hearing with respect to drug pricing or other business practices, we could incur significant expense and could be distracted from operation of our business and execution of our strategy. Any such investigation or hearing could also result in reduced market acceptance and demand for our products, could harm our reputation and our ability to market our products in the future, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects. For more information, see Note 12, Commitments and Contingencies—Legal Proceedings of the Notes to Consolidated Financial Statements included in Part IV of this Annual Report on Form 10-K and the risk factor under the heading “In addition to those specifically described in other risk
We expect that legislators, policymakers and healthcare insurance funds in the EUEurope will continue to propose and implement cost-containing measures to keep healthcare costs down. SuchThese measures could include limitations on the prices we will be able to charge for our products or the amountslevel of reimbursement available for these products from governmental agenciesauthorities or third party payors, may increase the tax obligations on pharmaceutical companies, or may facilitate the introduction of generic competition with respect to our products.payors. Further, an increasing number of EU member statesEuropean and other foreign countries use prices for medicinal products established in other countries as “reference prices” to help determine the price of the product in their own territory. Consequently, a downward trend in prices of medicinal products in some countries could contribute to similar downward trends elsewhere. Moreover, in order to obtain reimbursement for our products in some countries, including some EU member states, we may be required to conduct clinical trials that compare the cost-effectiveness of our products to other available therapies. There can be no assurance that our products will obtain favorable reimbursement status in any country. Any cost containment measures, including those listed above, or other healthcare system reforms that are adopted, could negatively affect our growth prospects in Europe.
In addition to access, coverage and reimbursement, the commercial success of our products depends upon their market acceptance by physicians, patients, third party payors and the medical community.
If physicians do not prescribe our products, we cannot generate the revenues we anticipate from product sales. Market acceptance of each of our products by physicians, patients, third party payors and the medical community depends on:
the clinical indications for which a product is approved and any restrictions placed upon the product in connection with its approval, such as a REMS, patient registry requirements or labeling restrictions;
the prevalence of the disease or condition for which the product is approved and its diagnosis;
the severity of side effects;effects and other risks in relation to the benefits of our products;
acceptance by physicians and patients of each product as a safe and effective treatment;
availability of sufficient product inventory to meet demand, particularly with respect to Erwinaze;
physicians’ decisions relating to treatment practices based on availability of product, particularly with respect to Erwinaze;
perceived advantages over alternative treatments;
relative convenience and ease of administration;
with respect to Xyrem, physician and patient assessment of the burdens associated with obtaining or maintaining the certifications required under the Xyrem REMS;
the cost of treatment in relation to alternative treatments, including generic products; and
the availability of financial or other assistance for patients who are uninsured or underinsured.
Because of our dependence upon market acceptance of our products, any adverse publicity associated with harm to patients or other adverse events resulting from the use or misuse of any of our products or any similar products distributed by other companies, including generic versions of our products, could materially and adversely affect our business, financial condition, results of operations and growth prospects. For example, from time to time, there is negative publicity about illicit GHB and its effects, including with respect to illegal use, overdoses, serious injury and death. Because sodium oxybate, the API in Xyrem, is a derivative of GHB, Xyrem sometimes also receives negative mention in publicity relating to GHB. JZP-258 includes the same API as Xyrem, but uses a different mixture of salts. Patients, physicians and regulators may therefore view Xyrem or JZP 258, if approved, as the same as or similar to illicit GHB. In addition, there are regulators and some law enforcement agencies that oppose the prescription and use of Xyrem, and potentially other oxybate products generally because of itstheir connection to GHB. Xyrem’s label includes information about adverse events from GHB.
Delays or problems in the supply of our products for sale or for use in clinical trials, loss of our single source suppliers or failure to comply with manufacturing regulations could materially and adversely affect our business, financial condition, results of operations and growth prospects.
The manufacture of pharmaceutical products requires significant expertise and capital investment, including the development of process controls required to consistently produce the API and the finished product in sufficient quantities while meeting detailed product specifications on a repeated basis. We and our suppliers may encounter difficulties in production, including difficulties with production costs and yields, process controls, quality control and quality assurance, including testing of stability, impurities and impurity levels and other product specifications by validated test methods, and compliance with strictly enforced U.S., state and non-U.S. regulations. In addition, we and our suppliers are subject to the FDA’s current Good Manufacturing Practices, or cGMP, requirements, DEA regulations and equivalent rules and regulations prescribed by non-U.S. regulatory authorities. We have cGMP responsibilities for the products we manufacture in our facilities and also have oversight responsibilities for the manufacturing conducted by our third party suppliers operating under contract. If we or any of our suppliers encounter manufacturing, quality or compliance difficulties with respect to any of our products, we may be unable to obtain or maintain regulatory approval or meet commercial demand for such products, which could adversely affect our business, financial condition, results of operations and growth prospects. In addition, thewe could be subject to enforcement action by regulatory authorities for our failure of any of our suppliers to comply with cGMP or other ruleswith respect to the products we manufacture in our facilities as well as for our failure to adequately oversee compliance with cGMP by any of our third party suppliers operating under contract. Moreover, failure to comply with applicable legal and regulations while manufacturing products onregulatory requirements subjects us and our behalf could result insuppliers to possible regulatory action, directed atincluding restrictions on supply or shutdown, which may adversely affect our or a supplier’s ability to supply the adequacy of our oversight of our contract suppliers, which could result in enforcement actions against us by the FDA and other regulatory entities. ingredients or finished products we need.
We have a manufacturing and development facility in Ireland where we manufacture Xyrem and development-stage oxybate products, including JZP-258, and a manufacturing plant in Italy where we produce the defibrotide drug substance. We currently do not have our own commercial manufacturing or packaging capability for our other products, product candidates or
34
their APIs. As a result, our ability to develop and supply products in a timely and competitive manner depends primarily on third party suppliers being able to meet our ongoing commercial and clinical trial needs for API, other raw materials, packaging materials and finished products. For details of our arrangements with our suppliers, see “Business—Manufacturing” in Part I, Item 1 of this Annual Report on Form 10-K.
In part due to the limited market size for our products and product candidates, we have a single source of supply for most of our marketed products, product candidates and their APIs. We are the sole supplier of the defibrotide compound. We have a single source for sodium oxybate, the API for Xyrem, for Erwinaze, for the finished vial form of Defitelio and for Vyxeos. Single sourcing puts us at risk of interruption in supply in the event of manufacturing, quality or compliance difficulties. There is no guarantee that our suppliers can or will continue to supply on a timely basis, or at all, the quantities of API or finished product that we need. If one of our suppliers fails or refuses to supply us for any reason, it would take a significant amount of time and expense to implement and execute the necessary technology transfer to, and to qualify, a new supplier. The FDA and similar international or national regulatory bodies must approve manufacturers of the active and inactive pharmaceutical ingredients and
certain packaging materials used in our products. The loss of one of our suppliers could require us to obtain regulatory clearance in the form of a “prior approval supplement” and to incur validation and other costs associated with the transfer of the API or product manufacturing process. We believe that it could take up to two years, or longer in certain cases, to qualify a new supplier, and we may not be able to obtain APIs or finished products from new suppliers on acceptable terms and at reasonable prices, or at all. If there are delays in qualifying new suppliers or facilities or a new supplier is unable to meet FDA’s or similar international regulatory body’s requirements for approval, there could be a shortage of the affected products for the marketplace or for use in clinical studies, or both, which could negatively impact our anticipated revenues and could potentially cause us to breach contractual obligations with customers or to violate local laws requiring us to deliver the product to those in need.
Erwinaze is licensed from, and manufactured for us by, a single source, PBL. The Erwinaze BLA includes a number of post-marketing commitments relatedA continuing and significant challenge to the manufacturemaintaining sales of Erwinaze and a barrier to increasing sales is PBL’s inability to consistently supply product that meets specifications in quantities that are adequate to meet market demand. All Erwinaze that PBL has been able to supply is currently completely absorbed by PBL.
demand for the product, and erratic supply patterns have prevented us from meeting patient demand in some markets or from being able to expand to new markets or indications. As a consequence, there is no product inventory that can be used to absorb supply disruptions resulting from quality, manufacturing, regulatory or other issues. PBL has experienced and continues to experience product quality and manufacturing issues that have resulted, and continue to result, in disruptions in our ability to supply markets from time to time and have caused, and may in the future cause, us to implement batch-specific, modified product use instructions. We experienced limited product availability of Erwinaze and supply disruptions globally in 2019 and may experience continued supply disruptions in 2020. In January 2017,addition, the FDA has issued a warning letter and FDA Forms 483 to PBL indicating that it was not satisfied with PBL’s response to the FDA Form 483 issued to PBL in March 2016 and citing, among other things, significant violations of cGMP for finished pharmaceuticals and significant deviations from cGMP for APIs. In March 2017,We cannot predict whether the required remediation activities by PBL filed a response to the warning letterin connection with the FDA. In August 2018, the FDA conducted an inspection of the PBL manufacturing facility and issued an FDA Form 483 to PBL citing observations related to items referenced in the warning letter as well as other manufacturing practices, including data and records management. PBL continues to address the issues identified by the FDA in theits prior warning letter and has submitted its response to the August 2018 Form 483.
As capacity constraints and supply disruptions continue, whether as a result of continued quality or manufacturing challenges at PBL, is subjectregulatory issues or an inability to similar inspections conducted byenforce our contractual rights, we will be unable to build product inventory, our ability to supply the MHRA. Following a site inspection of PBL by MHRA in December 2017, MHRA issued an inspection report listing several major findings, including major deficienciesmarket will continue to be compromised and failures by PBLphysicians’ decisions to comply with cGMP.use Erwinaze will continue to be negatively impacted. In January 2018, PBL filed a response to the report with the MHRA.
Vyxeos is manufactured by Baxter Oncology GmbH, or Baxter, which is a sole source supplier from a single site location. Given that our Vyxeos launch is at an early stage, there is limited experience with the complex manufacturing process relating to Vyxeos. Baxter manufactured batches that were used in the Phase 3 clinical trial for Vyxeos; there have since beenhas experienced batch failures due to mechanical, component and other issues in the production of Vyxeos, and batches have been produced that have otherwise not been in compliance with applicable specifications. We are continuing to work with Baxter to address manufacturing complexities.complexities related to Vyxeos. Moreover, the proprietary technology that supports the manufacture of Vyxeos is not easily transferable. Consequently, engaging an alternate manufacturer may be difficult, costly and time-consuming. If we fail to obtain a sufficient supply of Vyxeos in accordance with applicable specifications on a timely basis, for any reason or due to manufacturing or regulatory challenges, our sales of and revenues from Vyxeos, our future maintenance and potential growth of the market for this product, our ability to conduct ongoing and future clinical trials of Vyxeos, and our business, financial condition, results of operations and growth prospects could be materially adversely affected.
In addition, while the APIs in Vyxeos, daunorubicin and cytarabine, are available from a number of suppliers, certain suppliers have received warning letters from the FDA. As a result, we have qualified other suppliers for each API, and we provided the qualification data to the FDA. If the FDA restricts importation of API from either supplier, and we are unable to qualify API from additional suppliers in a timely manner, or at all, our ability to successfully commercialize Vyxeos and generate sales of this product at the level we expect and to conduct ongoing and future clinical trials of Vyxeos could be materially and adversely affected.
35
In addition, in order to conduct our ongoing and any future clinical trials of, complete marketing authorization submissions for, and potentially launch our other product candidates, we also need to have sufficient quantities of product manufactured. For example, Siegfried USA, LLC, or Siegfried, is currently our sole supplier of both the API and finished product for our development activities involving solriamfetol, and we expect that Siegfried will manufacture and supply solriamfetol drug product for commercial sale if solriamfetol receives regulatory approval. If Siegfried does not or is not able to supply us with solriamfetol for any reason, it may take time and resources to implement and execute the necessary technology transfer to another provider, and such delay could negatively impact our anticipated revenues from solriamfetol.
Risks Related to Growth of Our Product Portfolio and Research and Development
Our future success depends on our ability to successfully develop and obtain and maintain regulatory approval in the U.S. and Europe for our late-stage product candidates and, if approved, to successfully launch and commercialize those product candidates.
We may not be able to successfully identify and acquire or in-license additional products or product candidates to grow our business, and, even if we are able to do so, we may otherwise fail to realize the anticipated benefits of these acquisitions.transactions.
In addition to continued investment in our research and development pipeline, we intend to grow our business by acquiring or in-licensing, and developing, including with collaboration partners, additional products and product candidates that we believe are highly differentiated and have significant commercial potential. Future growth throughHowever, we may be unable to identify or consummate suitable acquisition or in-licensing will depend upon the availabilityopportunities, and this inability could impair our ability to grow our business. Other companies, many of suitable productswhich may have substantially greater financial, sales and product candidatesmarketing resources, compete with us for acquisition or in-licensing on acceptable prices, terms and conditions.
these opportunities. Even if appropriate opportunities are available, we may not be able to successfully identify them, or we may not have the financial resources necessary to pursue them. Other companies, many
36
Even if we are able to successfully identify and acquire, in-license or develop additional products or product candidates, we may not be able to successfully manage the risks associated with integrating any products or product candidates into our portfolio or the risks arising from anticipated and unanticipated problems in connection with an acquisition or in-licensing. WeFurther, while we seek to mitigate risks and liabilities of potential acquisitions and in-licensing transactions through, among other things, due diligence, there may be risks and liabilities that such due diligence efforts fail to discover, that are not be abledisclosed to realize the anticipated benefits for a variety of reasons, including if:
us, or that we are unable to obtain and maintain adequate funding to complete the development of, obtain regulatory approval for and commercialize an acquired product candidate;
inadequately assess. Any failure to identifyin identifying and managemanaging these risks, liabilities and uncertainties effectively, could have a material adverse effect on our business.
business, results of operations and financial condition. In addition, product and product candidate acquisitions, create other uncertainties and risks, particularly when the acquisition takes the form of a merger or other business consolidation. Our business acquisitionsconsolidation, have required, and any similar future transactions will also require, significant efforts and expenditures, including with respect to transition activities and integrating the acquired business with our historical business.integration activities. We may encounter unexpected difficulties, or incur unexpectedsubstantial costs, in connection with potential acquisitions and similar transactions, which include:
the need to incur substantial debt or engage in dilutive issuances of equity securities to pay for acquisitions;
the potential disruption of our historical core business;
the strain on, and need to continue to expand, our existing operational, technical, financial and administrative infrastructure;
the difficulties in integrating acquired products and product candidates into our portfolio;
the difficulties in assimilating employees and corporate cultures;
the failure to retain key managers and other personnel;
the need to write down assets or recognize impairment charges;
the diversion of our management’s attention to integration of operations and corporate and administrative infrastructures; and
any unanticipated liabilities for activities of or related to the acquired business or its operations, products or product candidates.
Conducting clinical trials is costly and time-consuming, and the outcomes are uncertain. A failure to prove that our product candidates are safe and effective in clinical trials, or to generate data in clinical trials to support expansion of the therapeutic uses for our existing products, could materially and adversely affect our business, financial condition, results of operations and growth prospects.
As a condition to regulatory approval, each product candidate must undergo extensive and expensive preclinical studies and clinical trials to demonstrate to a statistically significant degree that the product candidate is safe and effective. The results at any stage of the development process may lack the desired safety, efficacy or pharmacokinetic characteristics. Results of limited preclinical studies, including studiesIf the FDA determines that the safety or efficacy data submitted for the NDAs for JZP-258 or lurbinectedin, or to be submitted in animal models,the planned biologics license application, or BLA, for JZP-458, do not warrant marketing approval, we may not predict the results of human clinical trials. Similarly, results from earlybe required to conduct additional clinical trials, may notwhich could be predictive of results obtained in latercostly and larger clinical trials, and later clinical trials may fail to show the desired safety and efficacy of our product candidates despite successful initial clinical testing.time-consuming. Even if we believe we have successfully completed testing, the FDA or any equivalent non-U.S. regulatory agency may determine our data is not sufficiently compelling to warrant marketing approval for the indications sought, if at all, and may require us to engage in additional clinical trials or provide further analysis which may be costly and time-consuming. A number of companies in the pharmaceutical industry, including us, have suffered significant setbacks in clinical trials, even in advanced clinical trials after showing positive results in preclinical studies or earlier clinical trials. If a product candidate fails at any stage of development or the data is otherwise not sufficient for regulatory approval, we will not be able to commercialize it and receive any return on our investment in that product candidate.
Any adverse events or other data generated during the course of clinical trials of our product candidates and/or clinical trials related to additional indications for our commercialized products could result in action by the FDA or a non-U.S. regulatory agency, which may restrict our ability to sell, or adversely affect sales of, currently marketed products, or such events or other data could otherwise have a material adverse effect on a related commercial product, including with respect to its safety profile. Any failure or delay in completing such clinical trials could materially and adversely affect the maintenance and growth of the markets for the related marketed products, which could adversely affect our business, financial condition, results of operations and overall growth prospects.
In addition to issues relating to the results generated in clinical trials, clinical trials can be delayed or halted for a variety of reasons, including:
difficulty identifying, recruiting or enrolling eligible patients, often based on the number of clinical trials, particularly in hematology and oncology, with enrollment criteria targeting the same patient population;
difficulty identifying a clinical development pathway, including viable indications and appropriate clinical trial protocol design, particularly where there is no applicable regulatory precedent;
delays or failures in obtaining regulatory authorization to commence a trial because of safety concerns of regulators relating to our product candidates or similar product candidates of our competitors or failure to follow regulatory guidelines;
37
delays or failures in obtaining clinical materials and manufacturing sufficient quantities of the product candidate for use in trials;
delays or failures in reaching agreement on acceptable terms with prospective study sites;
delays or failures in obtaining approval of our clinical trial protocol from an institutional review board, known as an ethics committee in Europe, to conduct a clinical trial at a prospective study site;
failure of our clinical trials and clinical investigators, including contract research organizations or other third parties assisting us with clinical trials, to be in compliancesatisfactorily perform their contractual duties, meet expected deadlines and comply with the FDA and other regulatory agencies’ requirements, commonly referred to asincluding good clinical practices;
unforeseen safety issues;
inability to monitor patients adequately during or after treatment;
difficulty monitoring multiple study sites;
insufficient funds to complete the trials.
The regulatory approval process is expensive, time-consuming and uncertain and may prevent us or our partners from obtaining and maintaining approvals for the commercialization of some or all of our product candidates.
We are not permitted to market a pharmaceutical product in the U.S. or in the EU member states until we receive approval from the FDA or a marketing authorization from the EC or the competent authorities of the EU member states, as applicable. Submission of an application for marketing authorization does not assure approval for marketing in any jurisdiction, and we may encounter significant difficulties or costs in our efforts to obtain approval to market products.
If the FDA, the EC or the competent authorities of the EU member states determine that our quality, safety or efficacy data do not warrant marketing approval for a product candidate such as JZP-258, we could be required to conduct additional clinical trials as a condition to receiving approval, which could be costly and time-consuming and could delay or preclude the approval of our application. Any delay or failure in obtaining approval of a product candidate, our receipt of approval for narrower indications than sought, or burdens in an approved label such as a boxed warning, could have a negative impact on our ability to recoup our research and development costs and to successfully commercialize that product, any of which could materially and adversely affect our business, financial condition, results of operations and growth prospects.
In May 2018, we purchased a rare pediatric disease priority review voucher for $110.0 million, which we redeemed in connection with the submission of our NDA for JZP-258 in January 2020. However, the redemption of our rare pediatric disease priority review voucher may not result in faster review or approval for JZP-258 compared to products considered for approval under conventional FDA procedures and, in any event, does not assure ultimate approval of JZP-258 by FDA. For example, the FDA could determine that our application is not sufficient to support approval with the label we have requested, and require amendments or additional data, which could delay or preclude the approval of our NDA.
Even if we receive approval itof a product, regulatory authorities may be subject toimpose significant labeling restrictions or requirements, including limitations on the dosing of the product, requirements around the naming or strength of a product, restrictions on indicated uses for which we may market the product, the imposition of a boxed warning or other warnings and precautions, such asand/or the requirement for a REMS to ensure that the benefits of the drug outweigh the risks or the imposition of a boxed warning included in the
For example, we expect that the FDA will require a REMS for approval of JZP-258. Regulatory authorities may also impose post-marketing obligations as part of their approval. Post-marketing obligationsapproval, which may lead to additional costs and burdens associated with commercialization of the drug, and may pose a risk to maintaining approval of the drug. We are subject to certain post-marketing requirements and commitments in connection with the approval of certain of our products, including Defitelio, Erwinaze, DefitelioVyxeos and Vyxeos. For example, for Defitelio, in the U.S. the FDA imposed several post-marketing commitments and requirements in connection with its approval, including the requirement that we conduct a clinical trial to analyze the safety of defibrotide versus best supportive care in the prevention of VOD in adult and pediatric patients, and in the EU marketing authorization was granted under exceptional circumstances and requires us to comply with a number of post-marketing obligations, including a study to provide further data on long-term safety, health outcomes and patterns of utilization of Defitelio in normal use. Similarly, the FDA imposedSunosi. These post-marketing requirements in connection with its approval of our NDA for Vyxeos, including the requirement that we conduct aand commitments include satisfactorily conducting multiple post-marketing clinical trials and safety study to characterize infusion-related reactions in patients treated with Vyxeos and a clinical trial to determine dosing to minimize toxicity in patients with moderate and severe renal impairment, and the marketing authorization in the EU also requires us to comply with certain manufacturing-related post-approval commitments.studies. In the event that we are unable to comply with our post-marketing obligations imposed as part of the marketing approvals in the U.S. or EU, our approval may be varied, suspended or revoked, product supply may be delayed and our sales of and revenues from our products could be materially adversely affected.
Risks Related to Our Intellectual Property
It is difficult and costly to protect our proprietary rights, and we may not be able to ensure their protection.
Our commercial success depends in part on obtaining, maintaining and defending intellectual property protection for our products and product candidates, including protection of their use and methods of manufacturing and distribution. Our ability to protect our products and product candidates from unauthorized making, using, selling, offering to sell or importation by third parties depends on the extent to which we have rights under valid and enforceable patents or have adequately protected trade secrets that cover these activities.
The patent position of pharmaceutical companies can be highly uncertain and involve complex and often changing legal, regulatory and factual questions. The degree of protection to be afforded by our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
our patent applications, or those of our licensors or partners, may not result in issued patents;
38
others may independently develop similar or therapeutically equivalent products without infringing our patents, or those of our licensors, such as products that are not covered by the claims of our patents, or for which we do not have adequate exclusive rights under our license agreements;
our issued patents, or those of our licensors or partners, may be held invalid or unenforceable as a result of legal challenges by third parties or may be vulnerable to legal challenges as a result of changes in applicable law;
we or our licensors or partners might not have been the first to invent or file, as appropriate, subject matters covered by our issued patents or pending patent applications or those of our licensors or partners;
competitors may manufacture products in countries where we have not applied for patent protection or that have a different scope of patent protection or that do not respect our patents; or
others may be issued patents that prevent the sale of our products or require licensing and the payment of significant fees or royalties.
For example, we have several method of use patents listed in the FDA’s publication “Approved Drug Products with Therapeutic Equivalence Evaluations,” or the Orange Book, that expire in 2033 that cover instructions ontreatment methods included in the Xyrem package insert and Xyrem REMSlabel related to a drug-drug interaction, or DDI, with divalproex sodium. Although the FDA has stated, in granting a Citizen Petition we submitted in 2016, that it would not approve any sodium oxybate ANDA referencing Xyrem that does not include the portions of the currently approved Xyrem package insertlabel related to the DDI patents, we cannot predict whether a future ANDA filer, or a company that files a Section 505(b)(2) application for a drug referencing Xyrem, may pursue regulatory strategies to avoid infringing our DDI patents notwithstanding the FDA’s response to the Citizen Petition, or whether any such strategy would be successful. Likewise, we cannot predict whether we will be able to maintain the validity of these patents or will otherwise obtain a judicial determination that a generic or other sodium oxybate product, its package insert or the generic sodium oxybate REMS or another separate REMS will infringe any of our patents or, if we prevail in proving infringement, whether a court will grant an injunction that prevents a future ANDA filer or other company introducing a different sodium oxybate product from marketing its product, or instead require that party to pay damages in the form of lost profits or a reasonable royalty.
Since Xyrem’s regulatory exclusivity has expired in the EU, we are aware that generic or hybrid generic applications have been approved by various EU regulatory authorities, and additional generic or hybrid generic applications may be submitted and approved. We cannot predict whether our licensee in the EU will be able to enforce our existing European patents against generic or hybrid generic filers in the EU.
We also currently rely on trade secretssecret protection for several of our products, including Erwinaze and other unpatented proprietaryDefitelio. Trade secret protection does not protect information or inventions if another party develops that information or invention independently, and establishing that a competitor developed a product through trade secret misappropriation rather than through legitimate means may be difficult to prove. Trade secret protection also requires that information be secret and subject to reasonable efforts to maintain secrecy, and this requirement may come into conflict with requirements to provide information to protect our productsemployees, consultants, business partners, and their commercial position, particularly with respect to our products with limited or no patent protection, such as Erwinaze, which has no patent protection.regulatory bodies. We seek to protect our trade secrets and other unpatented proprietary information in part through confidentiality and invention agreements with our employees, consultants, advisors and partners. Nevertheless, our employees, consultants, advisors and partners may unintentionally or willfully disclose our proprietary information to competitors, and we may not have adequate remedies for such disclosures. EnforcingMoreover, if a claim that a third party illegally obtaineddispute arises with our employees, consultants, advisors or is using anypartners over the ownership of rights to inventions, including jointly developed intellectual property, we could lose patent protection or the confidentiality of our inventionsproprietary information, and possibly also lose the ability to pursue the development of certain new products or trade secrets would be expensive and time-consuming, and the outcome is unpredictable. In addition, courts outsideproduct candidates.
39
In some instances, we also rely on regulatory exclusivity to protect our commercial position. In addition to relying on trade secret protection,For example, Erwinaze was granted orphan drug exclusivity by the FDA for the treatment of ALL in the U.S. for a seven-year period from its FDA approval, which had precluded approval of another product with the same principal molecular structure for the same indication until November 2018. As a biologic product approved under a BLA, Erwinaze is also subject to the U.S. Biologics Price Competition and Innovation Act, or BPCIA. We believe that Erwinaze isBPCIA, and accordingly should be protected by exclusivity that prevents approval of a biosimilar in the U.S. through late 2023 under the BPCIA. However, interpretation of regulatory exclusivity under the BPCIA may evolve over time based on FDA issuance of guidance documents, proposed regulations or decisions made by the FDA in the course of considering specific applications. In addition, the BPCIA exclusivity period does not prevent another company from independently developing a product that is highly similar to Erwinaze, generating all the
data necessary for a full BLA and seeking approval. BPCIA exclusivity only assures that another company cannot rely on the FDA’s prior approvals of Erwinaze to support the biosimilar product’s approval. As a result, it is possible that a potential competing drug product might obtain FDA approval before the expected BPCIA exclusivity period has expired, which would adversely affect our sales of Erwinaze.Erwinaze if we are able to maintain our marketing rights to that product. In the EU, the regulatory data protection that provides an exclusivity period for Erwinase has lapsed. Any new marketing authorizations for Erwinase in other EU member states will not receive any regulatory data protection. If a biosimilar product to Erwinaze is approved as interchangeable to Erwinaze in the U.S. or in other countries where Erwinaze is sold, a significant percentage of the prescriptions that would have been written forcould be filled by Erwinaze, if commercially available, may be filled with the biosimilar version, resulting in a loss in sales of Erwinaze, and there may be a decrease in the price at which Erwinaze can be sold. Competition from a biosimilar product to Erwinaze could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
We have incurred and may in the future incur substantial costs as a result of litigation or other proceedings relating to patents, other intellectual property rights and related matters, and we may be unable to protect our rights to, or commercialize, our products.
Our ability, and that of our partners, to commercialize any approved products will depend, in part, on our ability to obtain patents, enforce those patents and operate without infringing the proprietary rights of third parties. If we choose to go to court to stop a third party from infringing our patents, our licensed patents or our partners’ patents, that third party has the right to ask the court or an administrative agency to rule that these patents are invalid and/or should not be enforced. These lawsuits and administrative proceedings are expensive and consume time and other resources, and we may not be successful in these proceedings or in stopping infringement. In addition, the inter partes review, or IPR, process under the Leahy-Smith America Invents Act permits any person, whether they are accused of infringing the patent at issue or not, to challenge the validity of certain patents. As a result, many types of entities, including ANDA filers, have challenged valuable pharmaceutical patents through the IPR process, and six of our Orange Book-listed patents for Xyrem were invalidated through this process.
There is a risk that a court or USPTO,the PTAB could decide that our patents or certain claims in our patents are not valid or infringed, and that we do not have the right to stop a third party from using the inventions covered by those claims, as happened with the decision of the PTAB that certainsix of our patent claimspatents covering the Xyrem REMS, are invalid.which were invalidated through the IPR process and delisted from the Orange Book. In addition, even if we prevail in establishing that another product infringes a valid claim of one of our patents, a court may determine that we can be compensated for the infringement in damages, and refuse to issue an injunction. As a result, we may not be entitled to stop another party from infringing our patents for their full term. For more information, see the risk factors under the headings “Risks Related to Xyrem and Our Other Marketed Products” and “It is difficult and costly to protect our proprietary rights, and we may not be able to ensure their protection” in this Part I, Item 1A. Lawsuits or proceedings we may file in the future, or our defense against any lawsuits or other proceedings that may be brought against us, may not costly and time-consuming and may not be successful in stopping the infringement of our patents.
Litigation involving patent matters is frequently settled between the parties, rather than continuing to a court ruling, and we have settled patent litigation with all nine Xyrem ANDA filers. The FTC has publicly stated that, in its view, certain types of agreements between branded and generic pharmaceutical companies related to the settlement of patent litigation or the manufacture, marketing and sale of generic versions of branded drugs violate the antitrust laws and has commenced investigations and brought actions against some companies that have entered into such agreements. In particular, the FTC has expressed its intention to take aggressive action to challenge settlements that include an alleged transfer of value from the brand company to the generic company (so-called “pay for delay” patent litigation settlements). The U.S. Congress and state legislatures have also identified pharmaceutical patent settlements as potential impediments to callgeneric competition and have introduced, and in states like California passed, legislation to regulate them. Third party payors have also challenged such settlements on legislators to pass stronger laws prohibiting such settlements.the grounds that they increase drug prices. Because there is currently no precise legal standard with respect to the lawfulness of such settlements, there could be extensive litigation over whether any settlement that we have entered into or might enter into in the future constitutes a reasonable and lawful patent settlement. Parties to such settlement agreements in the U.S. are required by law to file the agreements with the FTC and the U.SU.S. Department of Justice, or DOJ, for review. Accordingly, we have submitted our ANDA litigation settlement agreements to the FTC and the DOJ for review. We may receive formal or informal requests from the FTC regarding our ANDA litigation settlements, and there is a risk that the FTC may commence a
formal investigation or action against us, or a third party may initiate civil litigation regarding such settlements, which could divert the attention of management and cause us to incur significant costs, regardless of the outcome. Any claim or finding that we or our business partners have failed to comply with applicable laws and regulations could be costly to us and could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
40
A third party may claim that we or our manufacturing or commercialization partners are using inventions covered by the third party’s patent rights, or that we or such partners are infringing, misappropriating or otherwise violating other intellectual property rights, and may go to court to stop us from engaging in our normal operations and activities, including making or selling our products. Such lawsuits are costly and could affect our results of operations and divert the attention of management and development personnel. There is a risk that a court could decide that we or our partners are infringing, misappropriating or otherwise violating third party patent or other intellectual property rights, which could be very costly to us and have a material adverse effect on our business.
Other Risks Related to Our Business and Industry
We have substantially expanded our international footprint and operations, and we may expand further in the future, which subjects us to a variety of risks and complexities which, if not effectively managed, could negatively affect our business.
We are headquartered in Dublin, Ireland and have multiple offices in the U.S., Canada, the UK, Italy and other countries in Europe. Our headcount has grown to approximately 1,360 as of February 2019. This includes employees in 15 countries in North America and Europe, a European commercial presence, a complex distribution network for products in Europe and additional territories, and manufacturing facilities in Italy and Ireland. We may further expand our international operations into other countries in the future, either organically or by acquisition. While we have management and other personnel with substantial international experience, conductingConducting our business in multiple countries subjects us to a variety of risks and complexities that may materially and adversely affect our business, results of operations, financial condition and growth prospects. These risks and complexities include:prospects, including:
the diverse regulatory, financial and legal requirements in the countries where we are located or do business, including those related to data security and the use of, or access to, commercial and personal information, taxation, trade laws, including tariffs, export quotas, custom duties or other trade restrictions, and any changes to those requirements;
challenges inherent in efficiently managing employees in diverse geographies, including the need to adapt systems, policies, benefits and compliance programs to differing labor and employment law and other regulations, as well as maintaining positive interactions with our unionized employees;
costs of, and liabilities for, our international operations, products or product candidates; and
In addition, as a result of our international expansion, our business and corporate structure has become substantially more complex. Significant management time and effort is required to effectively manage the increased complexity of our company, and there can be no guarantee that we will effectively manage the increasedincreasing, global complexity of our business without experiencing operating inefficiencies or control deficiencies. Our failure to do so could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
The UK’s planned withdrawal from the EU, commonly referred to as Brexit, may have a negative effect on global economic conditions, financial marketscould increase our cost of doing business, reduce our gross margins or otherwise negatively impact our business and our business.financial results.
Brexit has createdwill continue to create significant uncertainty concerning the future relationship between the UK and the EU, particularly if the recent UK withdrawswithdrawal from the EU withoutin January 2020 is followed by a ratified withdrawal agreement in place. Fromfailure to agree to a regulatory perspective, there is uncertainty about which lawsfuture trading relationship between the EU and regulations will apply. Athe UK. Since a significant portion of the regulatory framework in the UK is derived from EU laws. However, it is unclear which EU laws, the UK will decide to replace or replicate in connection with its withdrawal from the EU. In particular,Brexit could materially impact the regulatory regime applicable to our operations, including with respect to the development, manufacture, importation, approval and commercialization of our product candidates may change, potentially significantly, and the impact on the process for obtaining or maintaining marketing authorization for pharmaceutical products manufactured or sold in the UK is otherwise unknown.
In addition, the laws and regulations that will apply after the UK withdraws from the EU may have implications for manufacturing sites that hold certifications issued by the UK competent authorities. Our capabilityour ability to rely on theseUK manufacturing sites for products intended for the EU market will depend on the terms of the UK's withdrawaltrade agreements concluded between the EU and the UK in the coming months and, potentially, on the ability to obtain relevant exemptions under EU law to supply the EU market with products manufactured at UK-certified sites. There is also the risk that if batch release and quality control testing sites for our products are located only in the UK, manufacturers will need to use sites in other EU member states.states to manufacture products for supply to the EU market. All of these changes, if they occur, could increase our costs and otherwise adversely affect our business.
We have an office in Oxford, England, which is focused on commercialization of our products outside of the U.S. We do not know to what extent, or when, the UK’s recent withdrawal from the EU or any other future changes to membership in the EU will impact our business, particularly our ability to conduct international business from a base of operations in the UK. The UK could lose the benefits of global trade agreements negotiated by the EU on behalf of its members, possibly resulting in increased trade barriers, which could make doing business in Europe more difficult and/or costly. Moreover, in the U.S., tariffs on certain U.S. imports have recently been imposed, and the EU and other countries have responded with retaliatory tariffs on certain U.S. exports. We cannot predict what effects these and potential additional tariffs will have on our business, including in the context of escalating global trade and political tensions. However, these tariffs and other trade restrictions, whether resulting from the UK’s withdrawal from the EU or
41
otherwise, could increase our cost of doing business, reduce our gross margins or otherwise negatively impact our business and our financial results.
If we fail to attract, retain and motivate key personnel or to retain the members of our executive management team, our operations and our future growth may be adversely affected.
Our success and our ability to grow depend in part on our continued ability to attract, retain and motivate highly qualified personnel. We are highly dependent uponpersonnel, including our executive management team and other critical personnel, all of whom work on many complex matters that are essential to our success.team. We do not carry “key person” insurance. The loss of services and institutional knowledge of one or more additional members of our executive management team or other key personnel could delay or prevent the successful completion of some of our vital activities. Any employee may terminate his or her employment at any time without notice or with only short noticeactivities and without cause or good reason. The resulting loss of institutional knowledge may negatively impact our operations and future growth.
In addition, changes in our organization as a result of executive management transition may have a disruptive impact on our ability to implement our strategy. Until we integrate new personnel, and unless they are able to succeed in their positions, we may be unable to successfully manage and grow our company we will need additional personnel. Competition for qualified personnel in the pharmaceutical industry is intense. Ifbusiness. In any event, if we are unable to attract, retain and motivate quality individuals, including in our researchor if there are delays, or if we do not successfully manage personnel and development operations, which are continuing to expand,executive management transitions, our business, financial condition, results of operations and growth prospects could be adversely affected.
Significant disruptions of information technology systems or data security breaches could adversely affect our business.
Our information technology systems, and those of our vendors, are large and complex and store large amounts of confidential information. The size and complexity of these systems make them potentially vulnerable to service interruptions or to security breaches from inadvertent or intentional actions by our employees, third party vendors and/or business partners, or from cyber-attacks by malicious third parties. Attacks of this natureare increasing in frequency, persistence, sophistication and intensity, and are being conducted by sophisticated and organized groups and individuals with a wide range of motives (including, but not limited to, industrial espionage) and expertise, including organized criminal groups, “hacktivists,” nation states and others. In addition to the extraction of important information, such attacks could include the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of our information. Although the aggregate impact on our operations and financial condition has not been material to date, we and our vendors have been the target of events of this nature and expect them to continue.
Significant disruptions of our, our third party vendors’ and/or business partners’ information technology systems or security breaches could adversely affect our business operations and/or result in the loss, misappropriation, and/or unauthorized access, use or disclosure of, or the prevention of access to, confidential information (including trade secrets or other intellectual property, proprietary business information and personal information), and could result in financial, legal, business and reputational harm to us. Any such event that leads to unauthorized access, use or disclosure of personal information, including personal information regarding our patients or employees, could harm our reputation, compel us to comply with federal and/or state breach notification laws and foreign law equivalents, subject us to mandatory corrective action, require us to verify the correctness of database contents and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information, which could disrupt our business, result in increased costs or loss of revenue, and/or result in significant legal and financial exposure. In addition, security breaches and other inappropriate access can be difficult to detect, and any delay in identifying them may further harm us. Moreover, the prevalent use of mobile devices to access confidential information increases the risk of security breaches. While we have implemented security measures to protect our information technology systems and infrastructure, there can be no assurance that such measures will prevent service interruptions or security breaches that could adversely affect our business. In addition, failure to maintain effective internal accounting controls related to security breaches and cybersecurity in general could impact our ability to produce timely and accurate financial statements and subject us to regulatory scrutiny.
FDA and Equivalent Non-U.S. Regulatory Authorities
Our activities are subject to extensive regulation encompassing the entire life cycle of our products, from research and development activities to marketing approval (including specific post-marketing obligations), manufacturing, labeling, packaging, adverse event and safety reporting, storage, advertising, promotion, sale, pricing and reimbursement, recordkeeping, distribution, importing and exporting. These requirements apply both to us and to third parties we contract with to perform services and supply us with products. The failure by us or any of our third party partners, including our corporate development and collaboration partners, clinical trial sites, suppliers, distributors and our central pharmacy for Xyrem, to comply with
42
applicable requirements could subject us to administrative or judicial sanctions or other negative consequences, such as delays in approval or refusal to approve a product candidate, restrictions on our products, our suppliers, our other partners or us, the withdrawal, suspension or variation of product approval or manufacturing authorizations, untitled letters, warning letters, fines and other monetary penalties, unanticipated expenditures, product recall, withdrawal or seizure, total or partial suspension of production or distribution, interruption of manufacturing or clinical trials, operating restrictions, injunctions, suspension of licenses, civil penalties and/or criminal prosecution, any of which could result in a significant drop in our revenues from the affected products and harm to our reputation and could have a significant impact on our sales, business and financial condition.
We monitor adverse events resulting from the use of our products, as do the regulatory authorities, and we file periodic reports with the authorities concerning adverse events. The authorities review these events and reports, and if they determine
that any events and/or reports indicate a trend or signal, they can require a change in a product label, restrict sales and marketing and/or require or conduct other actions, potentially including variation, withdrawal or suspension of the product from the market,marketing authorization, any of which could result in reduced market acceptance and demand for our products, could harm our reputation and our ability to market our products in the future, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects. The FDA and the competent authorities of the EU member states on behalf of the European Medicines Agency, or EMA, also periodically inspect our records related to safety reporting. Following such inspections, the FDA may issue notices on FDA Form 483 and warning letters that could cause us to modify certain activities. The EMA’s Pharmacovigilance Risk Assessment Committee may propose to the Committee for Medicinal Products for Human Use that the marketing authorization holder be required to take specific steps or advise that the existing marketing authorization be varied, suspended or revoked. Failure to adequately and promptly correct the observation(s) can result in further regulatory enforcement action, which could include the variation, suspension or withdrawal of marketing authorization or imposition of financial penalties or other enforcement measures. The failure to adequately address and promptly correct any matters identified by the FDA or other regulatory agencies could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
The FDA, the competent authorities of the EU member states and other governmental authorities require advertising and promotional labelingmaterials to be truthful and not misleading, and products to be marketed only for their approved indications and in accordance with the provisions of the approved label. Regulatory authorities actively investigate allegations of off-label promotion in order to enforce regulations prohibiting these types of activities. IfA determination that we are found to have promoted an approved product for off-label uses we may becould subject us to significant liability, including civil and administrative financial penalties and other remedies as well as criminal financial penalties, other sanctions and imprisonment. Even if we are not determined to have engaged in off-label promotion, an allegation that we have engaged in such activities could have a significant impact on our sales, business and financial condition. The U.S. government has also required companies to enter into complex corporate integrity agreements and/or non-prosecution agreements that impose significant reporting and other burdens on the affected companies. Failure to maintain a comprehensive and effective compliance program, and to integrate the operations of acquired businesses into a combined comprehensive and effective compliance program on a timely basis, could subject us to a range of regulatory actions and/or civil or criminal penalties that could affect our ability to commercialize our products and could harm or prevent sales of the affected products, or could substantially increase the costs and expenses of commercializing and marketing our products.
Other Regulatory Authorities
We are also subject to regulation by other regional, national, state and local agencies, including the DEA, the DOJ, the FTC, the United States Department of Commerce, the Office of Inspector General or OIG, of the HHSU.S. Department of Health and Human Services, or OIG, and other regulatory bodies, as well as similar governmental authorities in those non-U.S. countries in which we commercialize our products.
We are subject to numerous anti-fraudfraud and abuse laws and regulations globally and our sales, marketing, patient support and medical activities may be subject to scrutiny under these laws and regulations. For example, the U.S. federal anti-kickback statute is broad and activities that involve providing anythingThese laws are described in “Business—Government Regulation” in Part I, Item 1 of value to those who prescribe, purchase, or recommend pharmaceutical products may be subject to scrutiny. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common manufacturer business arrangements and activities, the exemptions and safe harbors are drawn narrowly, and practices or arrangements that involve remuneration may be subject to scrutiny if they do not clearly qualify for an exemption or safe harbor.this Annual Report on Form 10-K. While we maintain a comprehensive compliance program to try to ensure that our practices and the activities of our third-party contractors and employees fall within the scope of suchavailable statutory exceptions and regulatory safe harbors, regulators and enforcement agencies may disagree with our assessment or find fault with the conduct of our employees or contractors. In addition, existing regulations are subject to regulatory revision or changes in interpretation by the DOJ or OIG. Violations of the federal anti-kickback statute may be punished by civil and criminal fines, imprisonment, and/or exclusion from participation in federal healthcare programs.
43
including providing free product to customers expecting that the customers would bill federal programs for the product, providing consulting fees, grants, free travel and other benefits to physicians to induce them to prescribe the company’s products, and inflating prices reported to private price publication services, which are used to set drug reimbursement rates under government healthcare programs. In addition, the government and private whistleblowers have pursued False Claims Act cases against pharmaceutical companies for causing false claims to be submitted as a result of the marketing of their products for unapproved uses. Pharmaceutical and other healthcare companies also are subject to other federal false claim laws, including federal criminal healthcare fraud and false statement statutes that extend to non-government health benefit programs. If we become the subject of a government False Claims Act or other investigation or whistleblower suit, we could incur substantial legal costs (including settlement costs) and business disruption responding to such investigation or suit, regardless of the outcome. Violations of
Public reporting under the False Claims Act may result in significant financial penalties (on a per claim or statement basis), treble damages and exclusion from participation in federal health care programs.
We have various programs to help patients access our products, including patient assistance programs, which include co-pay coupons for certain of our products, services thatassistance to help patients determine their insurance coverage for our products, and a free product program. Co-pay coupon programs for commercially insured patients, including our program for Xyrem, have received negative publicity related to allegations regarding their use to promote branded pharmaceutical products over other less costly alternatives. In the past, payors brought class action lawsuits challenging the legality of manufacturer co-pay programs under a variety of federal and state laws and insurers have taken actions through their network pharmacies and PBMs to restrict manufacturer co-pay programs. In September 2014, the OIG issued a Special Advisory Bulletin warning manufacturers that they may be subject to sanctions under the federal anti-kickback statuteAnti-Kickback Statute and other laws if they do not take appropriate steps to exclude Medicare Part D beneficiaries from using co-pay coupons. It is possible that changes in insurer policies regarding co-pay coupons and/or the introduction and enactment of new legislation or regulatory action could restrict or otherwise negatively affect these patient support programs, which could result in fewer patients using affected products, including Xyrem, and therefore could have a material adverse effect on our sales, business and financial condition.
We have established programs to consider grant applications submitted by independent charitable organizations, including organizations that provide co-pay support to patients who suffer from the diseases treated by our drugs. The OIG has established guidelines that suggest that it is lawfulissued guidance for how pharmaceutical manufacturers tocan lawfully make donations to charitable organizations who provide co-pay assistance to Medicare patients, provided that such organizations, among other things, are bona fide charities, are entirely independent of and not controlled by the manufacturer, provide aid to applicants on a first-come basis according to consistent financial criteria, and do not link aid to use of a donor’s product. IfAlthough we have structured our programs to follow available guidance, if we or our vendors or donation recipients are deemed to fail to comply with relevant laws, regulations or evolving government guidance in the operation of these programs, such facts could be used as the basis for an enforcement action against us by the federal government.government or other enforcement agencies or private litigants.
In 2016 and 2017, we received subpoenas from the U.S. Attorney’s Office for the District of Massachusetts requesting documents related to our support of charitable organizations that provide financial assistance to Medicare patients. In April 2018,2019, we reached anfinalized our civil settlement agreement in principle with the DOJ on a proposal for a civil settlement of potential claims by the DOJ in the amount of $57.0 million, subject to accrual of interest on the settlement amount from the date of the agreement in principle, negotiation of a definitive settlement agreement and other contingencies. We expect any such settlement will involve entryOIG, and entered into a corporate integrity agreement which will impose significant costsrequiring us to maintain our ongoing corporate compliance program and operational burdens on our business. Moreover,obligating us to implement or continue, as applicable, a failure to comply withset of defined corporate integrity activities for a period of five years from the termseffective date of the corporate integrity agreement. In the event of a breach of the corporate integrity agreement, we could result in monetarybecome liable for payment of certain stipulated penalties or a reduction or elimination of coverage for our products by federal health care programs such as Medicare and Medicaid and state health care programs. If we do not reach a final settlement, or if we are unable to successfully negotiate and enter into a corporate integrity agreement, the outcome of this investigation could include an enforcement action against us. If the federal government were to file an enforcement action against us as a result of the investigation and could establish the elements of a violation of relevant laws, we could be subject to damages, fines and penalties, which could be substantial, along with other criminal, civil or administrative sanctions, including exclusionexcluded from participation in federal health care programs. Weprograms, which would expect to incur significant costs in connection with any enforcement action, regardless of the outcome.have a material adverse effect on our sales, business and financial condition.
We may also become subject to similar investigations by other state or federal governmental agencies or offices. Any additional investigationsoffices of our patient assistance programs or other business practices, maywhich could result in damages, fines, penalties or other criminal, civil or administrative sanctions or enforcement actions. Such investigations may also result inactions, as well as negative publicity, or other negative actions that could harm our reputation, impact our business practices, reducereduction in demand for, or patient access to, our products and/or reduce coverage of our products, including by federal health care programs and state health care programs. If any or all of these events occur, our business, financial condition, results of operations and stock price could be materially and adversely affected.
Our business activities outside of the U.S. are subject to the U.S. Foreign Corrupt Practices Act, or FCPA, and similar anti-bribery or anti-corruption laws, regulations or rules of other countries in which we operate, including the UK Bribery Act of 2010, or the UK Bribery Act. Our heavily regulated business involves significant interaction with public officials, including officials of non-U.S. governments. Additionally, inIn certain countries, the health care providers who prescribe pharmaceuticals are employed by their government and the purchasers of pharmaceuticals are government entities; therefore, our dealings with these prescribers and purchasers may be subject to regulation under the FCPA and the UK Bribery Act. Recently the U.S. Securities and
44
Exchange Commission, or SEC, and the DOJ have increased their FCPA enforcement activities with respect to pharmaceutical companies. In addition, under the Dodd-Frank Wall Street Reform and Consumer Protection Act, private individuals who report original information to the SEC that leads to successful enforcement actions may be eligible for a monetary award. There is no certainty that all employees and third party business partners (includingViolation of these laws by us or our distributors, wholesalers, agents, contractors, and other partners) will comply with anti-bribery laws. In particular, we do not control the actions of suppliers and other third party agents althoughfor which we may be liable for their actions. Violation of these laws may result
in civil or criminal sanctions, which could include monetary fines, criminal penalties, and disgorgement of past profits, which could have a material adverse impact on our business and financial condition.
Outside the U.S., interactions between pharmaceutical companies and physicians are also governed by strict laws, such as national anti-bribery laws of European countries, regulations, industry self-regulation codes of conduct and physicians’ codes of professional conduct. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment.
Xyrem and Sunosi are controlled substances under the Controlled Substances Act. Our suppliers, distributors, clinical sites and prescribers, as well as retail pharmacies for Sunosi and the central pharmacy for Xyrem, are subject to DEA and state regulations relating to manufacturing, storage, distribution and physician prescription procedures, including limitations on prescription refills, and are required to maintain DEA registration and state licenses, when handling these drugs and their APIs. The DEA periodically inspects facilities for compliance with its rules and regulations. Failure to comply with current and future regulations of the DEA, relevant state authorities or any comparable international requirements could lead to a variety of sanctions, including revocation or denial of renewal of DEA registrations, fines, injunctions, or civil or criminal penalties, could result in, among other things, additional operating costs to us or delays in shipments outside or into the U.S. and could have an adverse effect on our business and financial condition.
We are also subject to data protection and privacy laws and regulations governing the processing of personal data. The legislative and regulatory landscape for privacy and data security continues to evolve, and there has been an increasing focus on privacy and data security issues which may affectIf we or our business. Failurethird party partners fail to comply with currentapplicable data protection and futureprivacy laws and regulations, such as the EU General Data Protection Regulation that became effective in May 2018 and the California Consumer Privacy Act of 2018 that will become effective beginning January 2020,we could result inbe subject to government enforcement actions (including the imposition ofand significant penalties),penalties against us, criminal and civil liability for us and our officers and directors, private litigation and/or adverse publicity that negatively affects our business. If we or our vendors fail to comply with applicable data privacy laws, or if the legal mechanisms we or our vendors rely upon for the transfer of personal data are ever deemed inadequate, we could be subject to government enforcement actions and significant penalties against us, andpublicity. In addition, our business could be adversely impacted if our ability to transfer personal data outside of the European Economic Area or Switzerland is restricted, which could adversely impact our operating results. In addition, although we are not directly subject to privacy and security requirements under the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act, or HIPAA, other than with respect to providing certain employee benefits, we potentially could be subject to criminal penalties if we, our affiliates or our agents knowingly obtain or disclose individually identifiable health information maintained by a HIPAA-covered entity in a manner that is not authorized or permitted by HIPAA.
If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate program or other governmental pricing programs, we could be subject to additional reimbursement requirements, penalties, sanctions and fines.fines, which could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
Pricing and rebate calculations vary across products and programs, are complex, and are often subject to interpretation by us, governmental or regulatory agencies and the courts.courts, which can change and evolve over time. In the case of our Medicaid pricing data, if we become aware that our reporting for a prior quarter was incorrect, or has changed as a result of recalculation of the pricing data, we are obligated to resubmit the corrected data for up to three years after those data originally were due. Such restatements and recalculations increase our costs for complying with the laws and regulations governing the Medicaid Drug Rebate program and could result in an overage or underage in our rebate liability for past quarters. Price recalculations also may affect the ceiling price at which we are required to offer our products under the 340B program.
Civil monetary penalties can be applied if we are found to have knowingly submitted any false price or product information to the government, if we are found to have made a misrepresentation in the reporting of our average sales price, or if we fail to submit the required price data on a timely basis, or if we are found to have charged 340B covered entities more than the statutorily mandated ceiling price. Such conductCenters for Medicare and Medicaid Services, or CMS, could also could be grounds for CMSdecide to terminate our Medicaid drug rebate agreement, in which case federal payments may not be available under Medicaid or Medicare Part B for our covered outpatient drugs. There is no guarantee that
Our failure to comply with our submissionsreporting and payment obligations under the Medicaid Drug Rebate program and other governmental programs could negatively impact our financial results. CMS issued a final regulation, which became effective on April 1, 2016, to implement the changes to the Medicaid Drug Rebate program under the Healthcare Reform Act. The issuance of the final regulation, as well as any other regulations and coverage expansion by various governmental agencies relating to the Medicaid Drug Rebate program, has increased and will not be found by CMScontinue to increase our costs and the complexity of
45
compliance, has been and will continue to be incompletetime-consuming to implement, and could have a material adverse effect on our results of operations, particularly if CMS challenges the approach we take in our implementation of the final regulation.
The Health Resources and Services Administration, or incorrect.HRSA, issued a final regulation regarding the calculation of the 340B ceiling price and the imposition of civil monetary penalties on manufacturers that knowingly and intentionally overcharge covered entities, which became effective on January 1, 2019. Implementation of this regulation could affect our obligations and potential liability under the 340B program in ways we cannot anticipate. We are also required to report the 340B ceiling prices for our covered outpatient drugs to HRSA, which then publishes them to 340B covered entities. Any charge by HRSA that we have violated the requirements of the program or the regulation could negatively impact our financial results. Further, any additional future changes to the definition of average manufacturer price and the Medicaid rebate amount under the Healthcare Reform Act or otherwise could affect our 340B ceiling price calculations and negatively impact our results of operations.
We participate inhave obligations to report the U.S. Departmentaverage sales price for certain of Veterans Affairs, Federal Supply Schedule,our drugs to the Medicare program. Statutory or FSS, pricing programregulatory changes or CMS guidance could affect the average sales price calculations for our products and the Tricare Retail Pharmacy program, as described in more detail under the heading “Business—Pharmaceutical Pricing, Reimbursement by Governmentresulting Medicare payment rate, and Private Payors and Patient Access” in Part I, Item 1Acould negatively impact our results of this Annual Report on Form 10-K. operations.
Pursuant to applicable law, knowing provision of false information in connection with price reporting under thesethe U.S. Department of Veterans Affairs, FSS or Tricare Retail Pharmacy, or Tricare, programs can subject a manufacturer to civil monetary penalties. These program obligations also contain extensive disclosure and certification requirements.
If we overcharge the government in connection with our arrangements with FSS or Tricare, we are required to refund the difference to the government. Failure to make necessary disclosures and/or to identify contract overcharges can result in allegations against us under the False Claims Act and other laws and regulations. Unexpected refunds to the government, and responding to a government investigation or enforcement action, would be expensive and time-consuming, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
Our business and operations could be negatively affected if we become subject to shareholder activism, which could cause us to incur significant expense, hinder execution of our business strategy and impact our stock price.
Shareholder activism, which takes many forms and arises in a variety of situations, has been increasingly prevalent. If we become the subject of certain forms of shareholder activism, such as proxy contests, the attention of our management and our board of directors may be diverted from execution of our strategy. Such shareholder activism could give rise to perceived uncertainties as to our future strategy, adversely affect our relationships with business partners and make it more difficult to attract and retain qualified personnel. Also, we may incur substantial costs, including significant legal fees and other expenses, related to activist shareholder matters. Our stock price could be subject to significant fluctuation or otherwise be adversely affected by the events, risks and uncertainties of any shareholder activism.
Product liability and product recalls could harm our business.
The development, manufacture, testing, marketing and sale of pharmaceutical products are associated with significant risks of product liability claims or recalls. Side effects or adverse events known or reported to be associated with, or manufacturing defects in, the products sold by us could exacerbate a patient’s condition, or could result in serious injury or impairment or even death. This could result in product liability claims against us and/or recalls of one or more of our products. In many countries, including in EU member states, national laws provide for strict (no-fault) liability which applies even where damages are caused both by a defect in a product and by the act or omission of a third party.
Product liability insurance coverage is expensive, can be difficult to obtain and may not be available in the future on acceptable terms, or at all. Our product liability insurance may not cover all of the future liabilities we might incur in connection with the development, manufacture or sale of our products. In addition, we may not continue to be able to obtain
Product liability insurance coverage is expensive, can be difficult to obtain and may not be available in the future on acceptable terms, or at all. Our product liability insurance may not cover all of the future liabilities we might incur in connection with the development, manufacture or sale of our products. A successful claim or claims brought against us in excess of available insurance coverage could subject us to significant liabilities and could have a material adverse effect on our business, financial condition, results of operations and growth prospects. Such claims could also harm our reputation and the reputation of our products, adversely affecting our ability to market our products successfully.
46
We use hazardous materials in our manufacturing facilities, and any claims relating to the improper handling, storage, release or disposal of these materials could be time-consuming and expensive.
Our operations are subject to complex and increasingly stringent environmental, health and safety laws and regulations in the countries where we operate and, in particular, in Italy and Ireland where we have manufacturing facilities. Environmental and health and safety authorities in Italy and Ireland administer laws governing, among other matters, the emission of pollutants into the air (including the workplace), the discharge of pollutants into bodies of water, the storage, use, handling and disposal of hazardous substances, the exposure of persons to hazardous substances, and the general health, safety and welfare of employees and members of the public. Our manufacturing facilities are involved in the controlled storage, use and disposal of chemicals and solvents. Even if our safety procedures for handling and disposing of these hazardous materials comply with the standards prescribed by EU laws, we cannot completely eliminate the risk of contamination or injury from hazardous materials. If an accident or contamination involving pollutants or hazardous substances occurs, an injured party could seek to hold us liable for any damages that result and any liability could exceed the limits or fall outside the coverage of our insurance. We may not be able to maintain insurance with sufficient coverage on acceptable terms, or at all. In certain cases, laws may impose strict liability for pollution of the environment and contamination resulting from spills, disposals or other releases of hazardous substances or waste or any migration of such hazardous substances or waste. Costs, damages and/or fines may result from the presence, investigation and remediation of such contamination at properties currently or formerly owned, leased or operated by us or at off-site locations, including where we have arranged for the disposal of hazardous substances or waste. In addition, we may be subject to third party claims, including for natural resource damages, personal injury and property damage, in connection with such contamination.
Risks Related to Our Financial Condition and Results
We have incurred substantial debt, which could impair our flexibility and access to capital and adversely affect our financial position.position, and our business would be adversely affected if we are unable to service our debt obligations.
As of December 31, 2018,2019, we had total indebtedness of approximately $1.8 billion, which included $651.0 million in outstanding term loanbillion. Our substantial indebtedness under a secured credit agreement that we entered into in June 2015 and subsequently amended in July 2016 and in June 2018, which we refer to as the amended credit agreement, $575.0 million of outstanding indebtedness under our 1.875% exchangeable senior notes due 2021, or the 2021 Notes, which were issued in August 2014, and $575.0 million of outstanding indebtedness under our 1.50% exchangeable senior notes due 2024, or the 2024 Notes, which were issued in August 2017 and which we refer to, together with the 2021 Notes, as the Exchangeable Senior Notes.
limit our ability to borrow additional funds for working capital, capital expenditures, acquisitions or other general business purposes;
limit our ability to use our cash flow or obtain additional financing for working capital, capital expenditures, acquisitions, investments or other general business purposes;
require us to use a substantial portion of our cash flow from operations to make debt service payments;
limit our flexibility to plan for, or react to, changes in our business and industry;industry, or our ability to take specified actions to take advantage of certain business opportunities that may be presented to us;
result in dilution to our existing shareholders in the event exchanges of the Exchangeable Senior Notesour exchangeable senior notes are settled in our ordinary shares;
place us at a competitive disadvantage compared to our less leveraged competitors; and
increase our vulnerability to the impact of adverse economic and industry conditions.
If our cash flows and capital resources are insufficient to fund our debt service obligations, we may be forced to reduce or delay investments and capital expenditures, seek additional capital or restructure or refinance our debt. These alternative measures may not be successful and may not permit us to meet our scheduled debt service obligations. In the absence of such cash flows and resources, we could face substantial liquidity problems and might be required to dispose of material assets or operations to meet our debt service and other obligations. TheIn addition, if we are unable to repay amounts under our secured credit agreement that we entered into in June 2015 and subsequently amended, which we refer to as the amended credit agreement, restricts our ability to dispose of
Covenants in our amended credit agreement restrict our business and are expectedoperations in many ways and if we do not effectively manage our covenants, our financial conditions and results of operations could be adversely affected.
The amended credit agreement contains various covenants that, among other things, limit our ability and/or our restricted subsidiaries’ ability to:
incur or assume liens or additional debt or provide guarantees in respect of obligations of other persons;
pay dividends or distributions or redeem or repurchase capital stock;
prepay, redeem or repurchase certain debt;
make loans, investments, acquisitions (including acquisitions of exclusive licenses) and capital expenditures;
enter into agreements that restrict distributions from our subsidiaries;
sell assets and capital stock of our subsidiaries; and
consolidate or merge with or into, or sell substantially all of our assets to, continueanother person.
The amended credit agreement also includes certain financial covenants that require us to maintain a maximum secured leverage ratio and a minimum interest coverage ratio. Our failure to comply with any of the covenants could result in a default under the amended credit agreement, which could permit the lenders to declare all or part of any outstanding borrowings to be immediately due and payable, or to refuse to permit additional borrowings under the revolving credit facility. Moreover, our failure to repurchase our exchangeable senior notes at variable ratesa time when the repurchase is required by the indentures governing our exchangeable senior notes or to pay any cash payable on future exchanges of interest and expose usour exchangeable senior notes as required by those indentures would constitute a default under those indentures. A default under those indentures could also lead to interest rate risk. If interest rates increase,a default under other debt agreements or obligations, including the amended credit agreement. Likewise, a default under the amended
47
credit agreement could also lead to a default under other debt agreements or obligations, including the indentures governing our debt service obligations on the variable rate indebtedness would increase even if the amount borrowed remained the same, and our net income would decrease.exchangeable senior notes.
To continue to grow our business, we will need to commit substantial resources, which could result in future losses or otherwise limit our opportunities or affect our ability to operate and grow our business.
The scope of our business and operations has grown substantially since 2012, including through a series of transactions, including the business combination between Jazz Pharmaceuticals, Inc.combinations and Azur Pharma Public Limited Company, which we refer to as the Azur Merger, and our acquisitions of EUSA Pharma Inc., Gentium S.r.l. and Celator Pharmaceuticals, Inc.acquisitions. To continue to grow our business over the longer term, we will needplan to commit substantial additional resources to our businessproduct acquisition and executionin-licensing, product development, clinical trials of our strategy. Our ongoing capital requirements will depend on many factors, including:
We have significant intangible assets and goodwill. Consequently, the future impairment of our intangible assets and goodwill may significantly impact our profitability.
Our intangible assets and goodwill are significant. As of December 31, 2018, we had recorded $3.7 billion of intangible assetssignificant and goodwill related to our past acquisitions. Intangible assets and goodwill are subject to an impairment analysis whenever events or changes in circumstances indicate the carrying amount of the asset may not be recoverable. For example, in connection with entry into an asset purchase agreement in June 2018 to sell substantially all of the assets held by us related to Prialt® (ziconotide) intrathecal infusion, we recognized an impairment charge of $42.9 million in our consolidated statements of income in 2018, primarily related to the carrying balances of intangible assets. Additionally, goodwill and indefinite-lived assets are subject to an impairment test at least annually.
Events giving rise to impairment are an inherent risk in the pharmaceutical industry and cannot be predicted. Our results of operations and financial position in future periods could be negatively impacted should future impairments of intangible assets or goodwill occur.
Our financial results have been and may continue to be adversely affected by foreign currency exchange rate fluctuations.
Changes in our effective tax rates which could adversely affect our business and financial condition, results of operations and growth prospects.
We are incorporated in Ireland and maintain subsidiaries in North America and a number of other foreign jurisdictions. As a result, our effective tax rate is derived from a combination of applicable tax rates in the various jurisdictions where we operate. Our effective tax rate may fluctuate depending on a number of factors, including, but not limited to, the distribution of our profits or losses between the jurisdictions where we operate and changes to or differences in interpretation of tax laws. In addition, the tax laws of any jurisdiction in which we operate may change in the future, which could impact our effective tax rate. We are subject to reviews and audits by the U.S. Internal Revenue Services, or IRS, and other taxing authorities from time to time, and the IRS or other taxing authority may challenge our structure, transfer pricing arrangements and tax positions through an audit or lawsuit. Responding to or defending against challenges from taxing authorities could be expensive and consume time and other resources. If we are unsuccessful, we may be required to pay taxes for prior periods, interest, fines or penalties, and may be obligated to pay increased taxes in the future, any of which could require us to reduce our operating expenses, decrease efforts in support of our products or seek to raise additional funds. Any of these actions could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
The IRS may not agree with the conclusion that we should be treated as a foreign corporation for U.S. federal tax purposes.
Although we are incorporated in Ireland, the IRS may assert that we should be treated as a U.S. corporation (and, therefore, a U.S. tax resident) for U.S. federal tax purposes pursuant to Section 7874 of the U.S. Internal Revenue Code, or the Code. For U.S. federal tax purposes, a corporation generally is considered a tax resident in the jurisdiction of its organization or incorporation. Because we are an Irish incorporated entity, we would be classified as a foreign corporation (and, therefore, a non-U.S. tax resident) under these rules. Section 7874 of the Code provides an exception under which a foreign incorporated entity may, in certain circumstances, be treated as a U.S. corporation for U.S. federal tax purposes. Because we indirectly acquired all of Jazz Pharmaceuticals, Inc.’s assets through the acquisition of the shares of Jazz Pharmaceuticals, Inc. common
48
stock in the Azur Merger, the IRS could assert that we should be treated as a U.S. corporation for U.S. federal tax purposes under Section 7874. The IRS continues to scrutinize transactions that are potentially subject to Section 7874, and has issued several sets of final and temporary regulations under Section 7874 since 2012. We do not expect these regulations to affect the U.S. tax consequences of the Azur Merger. Nevertheless, new statutory and/or regulatory provisions under Section 7874 of the Code or otherwise could be enacted that adversely affect our status as a foreign corporation for U.S. federal tax purposes, and any such provisions could have prospective or retroactive application to us, our shareholders, Jazz Pharmaceuticals, Inc. and/or the Azur Merger.
Our U.S. affiliates’ ability to use their net operating losses to offset potential taxable income and related income taxes that would otherwise be due is limited under Section 7874 of the Code and could be subject to further limitations if we do not generate taxable income in a timely manner or if the “ownership change” provisions of Sections 382 and 383 of the Code result in further annual limitations.
Following certain acquisitions of a U.S. corporation by a foreign corporation, Section 7874 of the Code can limit the ability of the acquired U.S. corporation and its U.S. affiliates to utilize U.S. tax attributes such as net operating losses, or NOLs, to offset U.S. taxable income resulting from certain transactions. Based on the limited guidance available, this limitation applies to us. Our U.S. affiliates have a significant amount of NOLs. As a result of Section 7874 of the Code, after the Azur Merger, our U.S. affiliates have not been able and will continue to be unable, for a period of time, to utilize their U.S. tax attributes to offset their U.S. taxable income, if any, resulting from certain taxable transactions. While we expect to be able to fully utilize our U.S. affiliates’ U.S. NOLs prior to their expiration, as a result of this limitation, it may take our U.S. affiliates longer to use their NOLs.
Our ability to use these NOLs to offset potential future taxable income and related income taxes that would otherwise be due is also dependent upon our generation of future taxable income before the expiration dates of the NOLs, and we cannot predict with certainty when, or whether, our U.S. affiliates will generate sufficient taxable income to use all of the NOLs. In addition, the use of NOLs to offset potential future taxable income and related income taxes that would otherwise be due is subject to annual limitations under the “ownership change” provisions of Sections 382 and 383 of the Code and similar state provisions, which may result in the expiration of additional NOLs before future utilization.
The U.S. Congress, the EU, the Organization for Economic Co-operation and Development, or OECD, and other government agencies in jurisdictions where we and our affiliates do business have had an extended focus on issues related to the taxation of multinational corporations. One example is the OECD’s initiative in the area of “base erosion and profit shifting,” where payments are made between affiliates from a jurisdiction with high tax rates to a jurisdiction with lower tax rates. Some countries are beginning to implement legislation and other guidance to align their international tax rules with the OECD’s recommendation. As a result of the focus on the taxation of multinational corporations, the tax laws in Ireland, the U.S. and other countries in which we and our affiliates do business could change on a prospective or retroactive basis, and any such changes could adversely affect us.
On December 22, 2017, the U.S. Tax Cuts and Jobs Act, or U.S. Tax Act, was signed into law. The U.S. Tax Act made broad and complex changes to the U.S. tax code. The U.S. Department of Treasury has issued limited regulations and other interpretive guidance under the U.S. Tax Act, and is expected to issue additional guidance, the impact of which is uncertain but could change the financial impacts that were previously recorded or are expected to be recorded in future periods. Furthermore, the impact of this tax reform on certain holders of our ordinary shares could be adverse. Among other things, changes to the rules for determining a foreign corporation’s status as a controlled foreign corporation could have an adverse effect on U.S. persons who are treated as owning (directly or indirectly) at least 10% of the value or voting power of our ordinary shares. Investors should consult their own advisers regarding the potential application of these rules to their investments.
A substantial portion of our indebtedness bears interest at variable interest rates based on USD LIBOR and certain of our financial contracts are also indexed to USD LIBOR. Changes in the method of determining LIBOR, or the replacement of LIBOR with an alternative reference rate, may adversely affect interest rates on our current or future indebtedness and may otherwise adversely affect our financial condition and results of operations.
In July 2017, the Financial Conduct Authority, the authority that regulates the London Inter-bank Offered Rate, or LIBOR, announced that it intended to stop compelling banks to submit rates for the calculation of LIBOR after 2021. The Alternative Reference Rates Committee, or ARRC, in the U.S. has proposed that the Secured Overnight Financing Rate, or SOFR, is the rate that represents best practice as the alternative to the U.S. dollar, or USD, LIBOR for use in derivatives and other financial contracts that are currently indexed to USD LIBOR. ARRC has proposed a paced market transition plan to SOFR from USD LIBOR and organizations are currently working on industry-wide and company-specific transition plans as relating to derivatives and cash markets exposed to USD LIBOR. We have certain financial contracts, including the amended credit agreement and our interest rate swaps, that are indexed to USD LIBOR. Changes in the method of determining LIBOR, or the replacement of LIBOR with an alternative
reference rate, may adversely affect interest rates on our current or future indebtedness. Any transition process may involve, among other things, increased volatility or illiquidity in markets for instruments that rely on LIBOR, reductions in the value of certain instruments or the effectiveness of related transactions such as hedges, increased borrowing costs, uncertainty under applicable documentation, or difficult and costly consent processes. We are monitoring this activity and evaluating the related risks, and any such effects of theThe transition away from LIBOR may result in increased expenses, may impair our ability to refinance our indebtedness or hedge our exposure to floating rate instruments, or may result in
49
difficulties, complications or delays in connection with future financing efforts, any of which could adversely affect our financial condition and results of operations.
Risks Related to Our Ordinary Shares
The market price of our ordinary shares has been volatile and may continue to be volatile in the future, and the value of your investment could decline significantly.
The market price for our ordinary shares has fluctuated significantly from time to time for example, varying between a high of $184.00 on June 20, 2018 and a low of $113.52 on December 24, 2018 during 2018. The market price of our ordinary shares is likely to continue to be volatile and subject to significant price and volume fluctuations in response to market, industry and other factors, including the risk factors described above. The stock market in general, including the market for life sciences companies, has experienced extreme price and trading volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies. In particular, negative publicity regarding pricing and price increases by pharmaceutical companies has negatively impacted, and may continue to negatively impact, the market for life sciences companies. These broad market and industry factors have harmed, and in the future may seriously harm, the market price of our ordinary shares, regardless of our operating performance.
Our share price may be dependent upon the valuations and recommendations of the analysts who cover our business. If our results do not meet these analysts’ forecasts, the expectations of our investors or the financial guidance we provide to investors in any period, the market price of our ordinary shares could decline. Our ability to meet analysts’ forecasts, investors’ expectations and our financial guidance is substantially dependent on our ability to maintain or increase sales of our marketed products. The risks and uncertainties associated with our ability to maintain or increase sales of our products include those discussed elsewhere in these risk factors. In the past, following periods of volatility in the market or significant price decline, securities class-action litigation has often been instituted against companies. Such litigation, if instituted against us, could result in substantial costs and diversion of management’s attention and resources, which could materially and adversely affect our business, financial condition, results of operations and growth prospects.
In addition, the market price of our ordinary shares may decline if the effects of our strategic transactions including the Celator Acquisition and/or potential future acquisitions, on our financial or operating results are not consistent with the expectations of financial analysts or investors. The market price of our ordinary shares could also be affected by possible sales of our ordinary shares by holders of the Exchangeable Senior Notesour exchangeable senior notes who may view the Exchangeable Senior Notesour exchangeable senior notes as a more attractive means of equity participation in our company and by hedging or arbitrage trading activity involving our ordinary shares by the holders of the Exchangeable Senior Notes.our exchangeable senior notes.
We are subject to Irish law, which differs from the laws in effect in the U.S. and may afford less protection to holders of our securities.
It may not be possible to enforce court judgments obtained in the U.S. against us in Ireland based on the civil liability provisions of the U.S. federal or state securities laws. In addition, there is some uncertainty as to whether the courts of Ireland would recognize or enforce judgments of U.S. courts obtained against us or our directors or officers based on the civil liability provisions of the U.S. federal or state securities laws or hear actions against us or those persons based on those laws. We have been advised that the U.S. currently does not have a treaty with Ireland providing for the reciprocal recognition and enforcement of judgments in civil and commercial matters. Therefore, a final judgment for the payment of money rendered by any U.S. federal or state court based on civil liability, whether or not based solely on U.S. federal or state securities laws, would not automatically be enforceable in Ireland.
As an Irish company, we are governed by the Irish Companies Act 2014, which differs in some material respects from laws generally applicable to U.S. corporations and shareholders, including, among others, differences relating to interested director and officer transactions, mergers, amalgamations and acquisitions, takeovers and shareholder lawsuits. Likewise, theThe duties of directors and officers of an Irish company are generally owed to the company only. Shareholders of Irish companies generally do not have a personal right of action against directors or officers of the company and may exercise such rights of action on behalf of the company only in limited circumstances. Accordingly, holders of our securities may have more difficulty protecting their interests than would holders of securities of a corporation incorporated in a U.S. jurisdiction.
Our articles of association, Irish law and the indentures governing the Exchangeable Senior Notesour exchangeable senior notes contain provisions that could delay or prevent a takeover of us by a third party.
Our articles of association could delay, defer or prevent a third party from acquiring us, despite the possible benefit to our shareholders, or otherwise adversely affect the price of our ordinary shares. For example, our articles of association:
In addition to our articles of association, several mandatory provisions of Irish law could prevent or delay an acquisition of us. For example, Irish law does not permit shareholders of an Irish public limited company to take action by written consent with less than unanimous consent, and the shareholder approval requirements for certain types of transactions differ from those in the U.S., and in some cases are greater, under Irish law. We are also subject to various provisions of Irish law relating to mandatory bids, voluntary bids, requirements to make a cash offer and minimum price requirements, as well as substantial acquisition rules and rules requiring the disclosure of interests in our shares in certain circumstances. Furthermore, the indentures governing the Exchangeable Senior Notesour exchangeable senior notes require us to repurchase the Exchangeable Senior Notesour exchangeable senior notes for cash if we undergo certain fundamental changes and, in certain circumstances, to increase the exchange rate for a holder of 2021 Notes or 2024 Notes.our exchangeable senior notes. A takeover of us may trigger the requirement that we purchase the Exchangeable Senior Notesour exchangeable senior notes and/or increase the exchange rate, which could make it more costly for a potential acquiror to engage in a business combination transaction with us.
These provisions, whether alone or together, may discourage potential takeover attempts, discourage bids for our ordinary shares at a premium over the market price or adversely affect the market price of, and the voting and other rights of the holders
50
of, our ordinary shares. These provisions, whether alone or together, could also discourage proxy contests and make it more difficult for our shareholders to elect directors other than the candidates nominated by our board.
Future sales and issuances of our ordinary shares, securities convertible into our ordinary shares or rights to purchase ordinary shares or convertible securities could result in additional dilution of the percentage ownership of our shareholders and could cause our share price to decline.
We expect to continue to opportunistically seek access to additional capital to license or acquire additional products, product candidates or companies to expand our operations or for general corporate purposes. To the extent we raise additional capital by issuing equity securities or securities convertible into or exchangeable for ordinary shares, our shareholders may experience substantial dilution. We may sell ordinary shares, and we may sell convertible or exchangeable securities or other equity securities in one or more transactions at prices and in a manner we determine from time to time. If we sell such ordinary shares, convertible or exchangeable securities or other equity securities in subsequent transactions, existing shareholders may be materially diluted.
We have never declared or paid dividends on our capital stock and we do not anticipate paying dividends in the foreseeable future.
| Item 1B. | Unresolved Staff Comments |
There are no material unresolved written comments that were received from the SEC staff 180 days or more before the end of our 20182019 fiscal year relating to our periodic or current reports under the Securities Exchange Act of 1934, as amended.
| Item 2. | Properties |
Our corporate headquarters are located in Dublin, Ireland, and our U.S. operations are located in Palo Alto, California, Philadelphia, Pennsylvania and Ewing, New Jersey.
We lease approximately 45,000 square feet of office space in Dublin, Ireland. This lease expires in December 2036, with an option to terminate in December 2024 with no less than one year’s prior written notice and the payment of a termination fee, and a further option to terminate in December 2031 with no less than one year’s prior written notice. We own approximately 58,000 square foot of manufacturing and development facility in Athlone, Ireland, which is primarily used for the manufacture of Xyrem and development-stage products.
In Palo Alto, California, we occupy a total of approximately 143,000198,000 square feet of office space, 99,000 square feet of which is under a lease that expires in October 2029 orand has an option to terminate in October 2027 with no less than one year’s prior written notice and the Palo Alto Lease, and 44,000payment of a termination fee. The remaining 99,000 square feet of which is under a lease that expires in August 2019. We have an option to extend the term of the Palo Alto Lease twice for a period of five years eachJuly 2031 and an option to terminate in October 2027 with no less than one year’s prior written notice and the payment of a termination fee. In September 2017, we entered into an agreement to lease approximately 99,000 additional square feet of office space in Palo Alto, California. We expect to occupy this office space by the end of 2019. This lease has a term of 12 years from commencement, and we have an option to extend the termterms of the leaseboth leases twice for a period of five years each. We also have an option to terminate this lease in October 2029 with no less than one year’s prior written notice and the payment of a termination fee.
We occupy approximately 46,00060,000 square feet of office space in Philadelphia, Pennsylvania under a lease that expires in April 2029, or the Philadelphia Lease. The Philadelphia Lease also provides for another 14,000 square feet, which we expect to occupy by the end of 2019. In addition, we have offices in Canada, the United Kingdom, Italy, France and elsewhere in Europe.2029. We occupy approximately 26,000 square feet of office space in Oxford, United Kingdom under a lease that expires in December 2027.April 2028. We own a manufacturing facility in Villa Guardia (Como), Italy, which is primarily used for the manufacture of Defitelio. The manufacturing facility is approximately 37,00045,000 square feet. We also lease approximately 34,000 square feet of office and laboratory space in Villa Guardia (Como), Italy under a lease that expires in December 2023. In addition, we have offices in Canada, France and elsewhere in Europe.
We believe that our existing properties are in good condition and suitable for the conduct of our business. As we continue to expand our operations, we may need to lease additional or alternative facilities.
51
| Item 3. | Legal Proceedings |
| Item 4. | Mine Safety Disclosures. |
Not applicable.
52
PART II
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Market Information
Our ordinary shares trade on The Nasdaq Global Select Market under the trading symbol “JAZZ.”
Holders of Ordinary Shares
As of February 19, 2019,18, 2020, there were twothree holders of record of our ordinary shares. Because almost all of our ordinary shares are held by brokers, nominees and other institutions on behalf of shareholders, we are unable to estimate the total number of shareholders represented by these record holders.
Dividends
In 20182019 and 2017,2018, we did not declare or pay cash dividends on our common equity and we do not currently plan to pay cash dividends in the foreseeable future. Any future determination as to the payment of dividends will, subject to Irish legal requirements, be at the sole discretion of our board of directors and will depend on our financial condition, results of operations, capital requirements, compliance with the terms of any current credit agreement and other factors our board of directors deems relevant.
Unregistered Sales of Equity Securities
Except as previously reported in our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K filed with the Securities and Exchange Commission, or SEC, during the year ended December 31, 2018,2019, there were no unregistered sales of equity securities by us during the year ended December 31, 2018.2019.
Irish Law Matters
As we are an Irish incorporated company, the following matters of Irish law are relevant to the holders of our ordinary shares.
Irish Restrictions on Import and Export of Capital
Except as indicated below, there are no restrictions on non-residents of Ireland dealing in Irish domestic securities, which includes ordinary shares of Irish companies. Dividends and redemption proceeds also continue to be freely transferable to non-resident holders of such securities. The Financial Transfers Act, 1992 gives power to the Minister for Finance of Ireland to restrict financial transfers between Ireland and other countries and persons. Financial transfers are broadly defined and include all transfers that would be movements of capital or payments within the meaning of the treaties governing the member states of the European Union, or EU.Union. The acquisition or disposal of interests in shares issued by an Irish incorporated company and associated payments falls within this definition. In addition, dividends or payments on redemption or purchase of shares and payments on a liquidation of an Irish incorporated company would fall within this definition. At present the Financial Transfers Act, 1992 prohibits financial transfers involving the late Slobodan Milosevic and associated persons, Belarus, certain persons indicted by the International Criminal Tribunal for the former Yugoslavia, the late Osama bin Laden, Al-Qaida, the Taliban of Afghanistan, Democratic Republic of Congo, Democratic People’s Republic of Korea (North Korea), Iran, Iraq, Côte d’Ivoire, Lebanon, Liberia, Zimbabwe, Sudan, Somalia, Republic of Guinea, Republic of Guinea-Bissau, Afghanistan, Egypt, Eritrea, Libya, Syria, Tunisia, certain known terrorists and terrorist groups, countries that harbor certain terrorist groups and Ukraine without the prior permission of the Central Bank of Ireland.
Any transfer of, or payment in respect of, a share or interest in a share involving the government of any country that is currently the subject of United Nations sanctions, any person or body controlled by any of the foregoing, or by any person acting on behalf of the foregoing, may be subject to restrictions pursuant to such sanctions as implemented into Irish lawlaw.
Irish Taxes Applicable to U.S. Holders
Irish Tax on Capital Gains. A shareholder who is neither resident nor ordinarily resident in Ireland and does not hold our ordinary shares in connection with a trade or business carried on by such shareholder in Ireland through a branch or agency should not be subject to Irish tax on capital gains on a disposal of our ordinary shares.
Capital Acquisitions Tax. Irish capital acquisitions tax, or CAT, is comprised principally of gift tax and inheritance tax. CAT could apply to a gift or inheritance of our ordinary shares irrespective of the place of residence, ordinary residence or domicile of the parties. This is because our ordinary shares are regarded as property situated in Ireland as our share register must be held in Ireland. The person who receives the gift or inheritance has primary liability for CAT.
53
CAT is levied at a rate of 33% above certain tax-free thresholds. The appropriate tax-free threshold is dependent upon (i) the relationship between the donor and the donee and (ii) the aggregation of the values of previous gifts and inheritances received by the donee from persons within the same category of relationship for CAT purposes. Gifts and inheritances passing between spouses are exempt from CAT. Our shareholders should consult their own tax advisers as to any tax consequences of holding our ordinary shares, including whether CAT is creditable or deductible in computing any domestic tax liabilities.
Stamp Duty. Irish stamp duty (if any) may become payable in respect of ordinary share transfers. However, a transfer of our ordinary shares from a seller who holds shares through Depository Trust Company, or DTC, to a buyer who holds the acquired shares through DTC will not be subject to Irish stamp duty. A transfer of our ordinary shares (i) by a seller who holds ordinary shares outside of DTC to any buyer or (ii) by a seller who holds the ordinary shares through DTC to a buyer who holds the acquired ordinary shares outside of DTC, may be subject to Irish stamp duty (currently at the rate of 1% of the price paid or the market value of the ordinary shares acquired, if greater). The person accountable for payment of stamp duty is the buyer or, in the case of a transfer by way of a gift or for less than market value, all parties to the transfer.
A shareholder who holds ordinary shares outside of DTC may transfer those ordinary shares into DTC without giving rise to Irish stamp duty provided that the shareholder would be the beneficial owner of the related book-entry interest in those ordinary shares recorded in the systems of DTC (and in exactly the same proportions) as a result of the transfer and at the time of the transfer into DTC there is no sale of those book-entry interests to a third party being contemplated by the shareholder. Similarly, a shareholder who holds ordinary shares through DTC may transfer those ordinary shares out of DTC without giving rise to Irish stamp duty provided that the shareholder would be the beneficial owner of the ordinary shares (and in exactly the same proportions) as a result of the transfer, and at the time of the transfer out of DTC there is no sale of those ordinary shares to a third party being contemplated by the shareholder. In order for the share registrar to be satisfied as to the application of this Irish stamp duty treatment where relevant, the shareholder must confirm to us that the shareholder would be the beneficial owner of the related book-entry interest in those ordinary shares recorded in the systems of DTC (and in exactly the same proportions) (or vice-versa) as a result of the transfer and there is no agreement being contemplated for the sale of the related book-entry interest or the ordinary shares or an interest in the ordinary shares, as the case may be, by the shareholder to a third party.
Performance Measurement Comparison (1)
The following graph shows the total shareholder return on the last day of each year of an investment of $100 in cash as if made on December 31, 20132014 in (i) our ordinary shares; (ii) the Nasdaq Composite Index; and (iii) the Nasdaq Biotechnology Index through December 31, 2018.2019. The shareholder return shown in the graph below is not necessarily indicative of future performance, and we do not make or endorse any predictions as to future shareholder returns.
COMPARISON OF FIVE YEAR CUMULATIVE TOTAL RETURN (2)
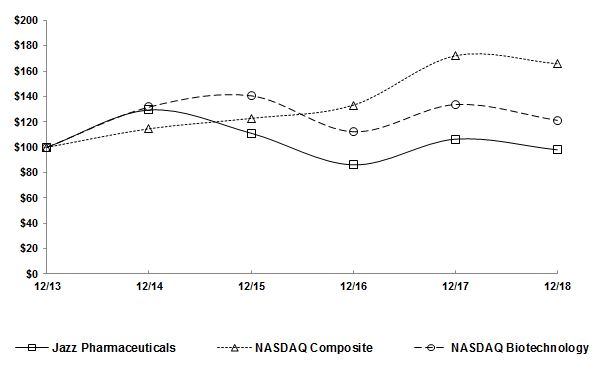
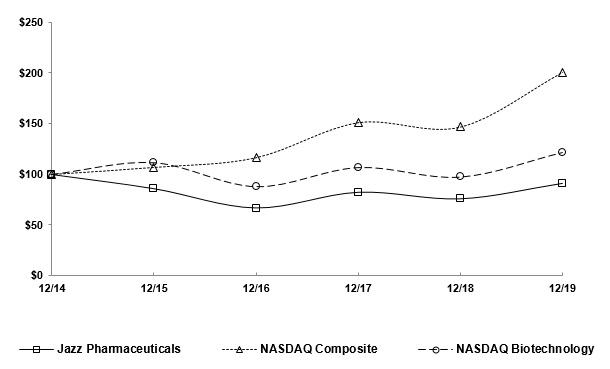
_________________________
| (1) | This section is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference into any of our filings under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, or Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing. |
| (2) | Information used in the graph was obtained from Research Data Group, Inc. |
Issuer Purchases of Equity Securities
The following table summarizes purchases of our ordinary shares made by or on behalf of us or any of our “affiliated purchasers” as defined in Rule 10b-18(a)(3) under the Exchange Act during each fiscal month during the three-month period ended December 31, 2018:2019:
| Total Number of Shares Purchased (1) | Average Price Paid per Share (2) | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs (3) | Maximum Number (or Approximate Dollar Value) of Shares that May Yet Be Purchased Under the Plans or Programs (4) | ||||||||||
| October 1 - October 31, 2018 | 123,399 | $ | 159.86 | 123,000 | $ | 86,074,483 | |||||||
| November 1 - November 30, 2018 | 1,780,091 | $ | 147.70 | 1,780,091 | $ | 143,189,015 | |||||||
| December 1 - December 31, 2018 | 1,127,314 | $ | 145.54 | 1,127,314 | $ | 379,137,805 | |||||||
| Total | 3,030,804 | $ | 147.39 | 3,030,405 | |||||||||
| Total Number of Shares Purchased (1) | Average Price Paid per Share (2) | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs (3) | Maximum Number (or Approximate Dollar Value) of Shares that May Yet Be Purchased Under the Plans or Programs (4) | ||||||||||
| October 1 - October 31, 2019 | 57,300 | $ | 122.96 | 57,300 | $ | 681,006,700 | |||||||
| November 1 - November 30, 2019 | 555,852 | $ | 136.45 | 555,852 | $ | 605,174,166 | |||||||
| December 1 - December 31, 2019 | 183,345 | $ | 149.49 | 183,345 | $ | 577,732,249 | |||||||
| Total | 796,497 | $ | 138.53 | 796,497 | |||||||||
_________________________
| (1) | This column |
| (2) | Average price paid per share includes brokerage commissions. |
| (3) | The ordinary shares reported in this column above were purchased pursuant to our publicly announced share repurchase program. In November 2016, we announced that our board of directors authorized the use of up to $300 million to repurchase our ordinary shares. In November and December 2018, our board of directors increased the existing share repurchase program authorization by $320.0 million and $400.0 million, |
55
directors authorized the additional repurchase of shares having an aggregate purchase price of up to $500.0 million, exclusive of any brokerage commissions. This authorization has no expiration date.
| (4) | The dollar amount shown represents, as of the end of each fiscal month, the approximate dollar value of ordinary shares that may yet be purchased under our publicly announced share repurchase program, exclusive of any brokerage commissions. |
| Item 6. | Selected Financial Data |
The following selected consolidated financial data should be read together with our consolidated financial statements and accompanying notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” appearing elsewhere in this Annual Report on Form 10-K. The selected consolidated financial data in this section is not intended to replace our consolidated financial statements and the accompanying notes. Our historical results are not necessarily indicative of our future results.
We derived the consolidated statements of income data for the years ended December 31, 2019, 2018 2017 and 20162017 and the selected consolidated balance sheet data as of December 31, 20182019 and 20172018 from the audited consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K. The consolidated statements of income data for the years ended December 31, 20152016 and 2014,2015, and the selected consolidated balance sheet data as of December 31, 2017, 2016 2015 and 20142015 are derived from audited consolidated financial statements not included in this Annual Report on Form 10-K.10‑K.
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||||||||||||||||||
| 2018 | 2017 | 2016(1) | 2015 | 2014(2) | 2019 | 2018 | 2017 | 2016(1) | 2015 | |||||||||||||||||||||||||||||
| (In thousands, except per share amounts) | (In thousands, except per share amounts) | |||||||||||||||||||||||||||||||||||||
| Consolidated Statements of Income Data: | Consolidated Statements of Income Data: | Consolidated Statements of Income Data: | ||||||||||||||||||||||||||||||||||||
| Revenues: | ||||||||||||||||||||||||||||||||||||||
| Product sales, net | $ | 1,869,473 | $ | 1,601,399 | $ | 1,477,261 | $ | 1,316,819 | $ | 1,162,716 | $ | 2,135,601 | $ | 1,869,473 | $ | 1,601,399 | $ | 1,477,261 | $ | 1,316,819 | ||||||||||||||||||
| Royalties and contract revenues | 21,449 | 17,294 | 10,712 | 7,984 | 10,159 | 26,160 | 21,449 | 17,294 | 10,712 | 7,984 | ||||||||||||||||||||||||||||
| Total revenues | 1,890,922 | 1,618,693 | 1,487,973 | 1,324,803 | 1,172,875 | 2,161,761 | 1,890,922 | 1,618,693 | 1,487,973 | 1,324,803 | ||||||||||||||||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||||||||||||
| Cost of product sales (excluding amortization of intangible assets) | 121,544 | 110,188 | 105,386 | 102,526 | 117,418 | |||||||||||||||||||||||||||||||||
| Cost of product sales (excluding amortization of acquired developed technologies) | 127,930 | 121,544 | 110,188 | 105,386 | 102,526 | |||||||||||||||||||||||||||||||||
| Selling, general and administrative | 683,530 | 544,156 | 502,892 | 449,119 | 406,114 | 736,942 | 683,530 | 544,156 | 502,892 | 449,119 | ||||||||||||||||||||||||||||
| Research and development | 226,616 | 198,442 | 162,297 | 135,253 | 85,181 | 299,726 | 226,616 | 198,442 | 162,297 | 135,253 | ||||||||||||||||||||||||||||
| Intangible asset amortization | 201,498 | 152,065 | 101,994 | 98,162 | 126,584 | 354,814 | 201,498 | 152,065 | 101,994 | 98,162 | ||||||||||||||||||||||||||||
| Impairment charges | 42,896 | — | — | 31,523 | 39,365 | — | 42,896 | — | — | 31,523 | ||||||||||||||||||||||||||||
| Acquired in-process research and development | — | 85,000 | 23,750 | — | 202,626 | 109,975 | — | 85,000 | 23,750 | — | ||||||||||||||||||||||||||||
| Total operating expenses | 1,276,084 | 1,089,851 | 896,319 | 816,583 | 977,288 | 1,629,387 | 1,276,084 | 1,089,851 | 896,319 | 816,583 | ||||||||||||||||||||||||||||
| Income from operations | 614,838 | 528,842 | 591,654 | 508,220 | 195,587 | 532,374 | 614,838 | 528,842 | 591,654 | 508,220 | ||||||||||||||||||||||||||||
| Interest expense, net | (77,075 | ) | (77,756 | ) | (61,942 | ) | (56,917 | ) | (52,713 | ) | (72,261 | ) | (77,075 | ) | (77,756 | ) | (61,942 | ) | (56,917 | ) | ||||||||||||||||||
| Foreign exchange gain (loss) | (6,875 | ) | (9,969 | ) | 3,372 | 1,445 | 8,683 | (5,811 | ) | (6,875 | ) | (9,969 | ) | 3,372 | 1,445 | |||||||||||||||||||||||
| Loss on extinguishment and modification of debt | (1,425 | ) | — | (638 | ) | (16,815 | ) | — | — | (1,425 | ) | — | (638 | ) | (16,815 | ) | ||||||||||||||||||||||
| Income before income tax provision (benefit) and equity in loss of investees | 529,463 | 441,117 | 532,446 | 435,933 | 151,557 | 454,302 | 529,463 | 441,117 | 532,446 | 435,933 | ||||||||||||||||||||||||||||
| Income tax provision (benefit) | 80,162 | (47,740 | ) | 135,236 | 106,399 | 94,231 | (73,154 | ) | 80,162 | (47,740 | ) | 135,236 | 106,399 | |||||||||||||||||||||||||
| Equity in loss of investees | 2,203 | 1,009 | 379 | — | — | 4,089 | 2,203 | 1,009 | 379 | — | ||||||||||||||||||||||||||||
| Net income | 447,098 | 487,848 | 396,831 | 329,534 | 57,326 | 523,367 | 447,098 | 487,848 | 396,831 | 329,534 | ||||||||||||||||||||||||||||
| Net loss attributable to noncontrolling interests | — | — | — | (1 | ) | (1,061 | ) | — | — | — | — | (1 | ) | |||||||||||||||||||||||||
| Net income attributable to Jazz Pharmaceuticals plc | $ | 447,098 | $ | 487,848 | $ | 396,831 | $ | 329,535 | $ | 58,387 | $ | 523,367 | $ | 447,098 | $ | 487,848 | $ | 396,831 | $ | 329,535 | ||||||||||||||||||
| Net income attributable to Jazz Pharmaceuticals plc per ordinary share: | ||||||||||||||||||||||||||||||||||||||
| Basic | $ | 7.45 | $ | 8.13 | $ | 6.56 | $ | 5.38 | $ | 0.98 | $ | 9.22 | $ | 7.45 | $ | 8.13 | $ | 6.56 | $ | 5.38 | ||||||||||||||||||
| Diluted | $ | 7.30 | $ | 7.96 | $ | 6.41 | $ | 5.23 | $ | 0.93 | $ | 9.09 | $ | 7.30 | $ | 7.96 | $ | 6.41 | $ | 5.23 | ||||||||||||||||||
| Weighted-average ordinary shares used in per share calculations - basic | 59,976 | 60,018 | 60,500 | 61,232 | 59,746 | 56,749 | 59,976 | 60,018 | 60,500 | 61,232 | ||||||||||||||||||||||||||||
| Weighted-average ordinary shares used in per share calculations - diluted | 61,221 | 61,317 | 61,870 | 63,036 | 62,614 | 57,550 | 61,221 | 61,317 | 61,870 | 63,036 | ||||||||||||||||||||||||||||
| As of December 31, | As of December 31, | |||||||||||||||||||||||||||||||||||||
| 2018 | 2017 | 2016(1) | 2015 | 2014(2) | 2019 | 2018 | 2017 | 2016(1) | 2015 | |||||||||||||||||||||||||||||
| (In thousands) | (In thousands) | |||||||||||||||||||||||||||||||||||||
| Consolidated Balance Sheet Data: | ||||||||||||||||||||||||||||||||||||||
| Cash, cash equivalents and investments | $ | 824,622 | $ | 601,035 | $ | 425,963 | $ | 988,785 | $ | 684,042 | $ | 1,077,344 | $ | 824,622 | $ | 601,035 | $ | 425,963 | $ | 988,785 | ||||||||||||||||||
| Working capital | 888,518 | 674,330 | 490,663 | 1,031,025 | 799,044 | 1,265,778 | 888,518 | 674,330 | 490,663 | 1,031,025 | ||||||||||||||||||||||||||||
| Total assets | 5,203,491 | 5,123,672 | 4,800,227 | 3,332,612 | 3,308,617 | 5,538,897 | 5,203,491 | 5,123,672 | 4,800,227 | 3,332,612 | ||||||||||||||||||||||||||||
| Long-term debt, current and non-current | 1,596,412 | 1,581,038 | 2,029,625 | 1,188,444 | 1,313,161 | 1,607,257 | 1,596,412 | 1,581,038 | 2,029,625 | 1,188,444 | ||||||||||||||||||||||||||||
| Retained earnings | 841,050 | 917,956 | 528,907 | 302,686 | 34,704 | 1,067,815 | 841,050 | 917,956 | 528,907 | 302,686 | ||||||||||||||||||||||||||||
| Total Jazz Pharmaceuticals plc shareholders’ equity | 2,757,422 | 2,713,097 | 1,877,339 | 1,598,646 | 1,371,144 | 3,110,981 | 2,757,422 | 2,713,097 | 1,877,339 | 1,598,646 | ||||||||||||||||||||||||||||
__________________________
| (1) | On |
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
The following discussion of our financial condition and results of operations should be read in conjunction with the consolidated financial statements and notes to consolidated financial statements included elsewhere in this Annual Report on Form 10-K.10‑K. This discussion contains forward-looking statements that involve risks and uncertainties. When reviewing the
discussion below, you should keep in mind the substantial risks and uncertainties that impact our business. In particular, we encourage you to review the risks and uncertainties described in “Risk Factors” in Part I, Item 1A in this Annual Report on Form 10-K.10‑K. These risks and uncertainties could cause actual results to differ materially from those projected in forward-looking statements contained in this report or implied by past results and trends.
Overview
Jazz Pharmaceuticals plc is a global biopharmaceutical company dedicated to developing life-changing medicines for people with serious diseases – often with limited or no options. As a leader in sleep medicine and with a growing hematology/oncology portfolio, weWe have a diverse portfolio of productsmarketed medicines and novel product candidates, from early- to late-stage development, in development.key therapeutic areas. Our focus is in neuroscience, including sleep medicine and movement disorders, and in oncology, including hematologic and solid tumors. We actively explore new options for patients including novel compounds, small molecule advancements, biologics and innovative delivery technologies.
Our lead marketed products are:
| • | Xyrem® (sodium oxybate) oral solution, the only product approved by the U.S. Food and Drug Administration, or FDA, and marketed in the U.S. for the treatment of both cataplexy and excessive daytime sleepiness, or EDS, in both adult and pediatric patients with narcolepsy; |
| • |
| • | Defitelio® (defibrotide sodium), a product approved in the U.S. for the treatment of adult and pediatric patients with hepatic veno-occlusive disease, or VOD, also known as sinusoidal obstruction syndrome, with renal or pulmonary dysfunction following hematopoietic stem cell transplantation, or HSCT, and in Europe (where it is marketed as Defitelio® (defibrotide)) for the treatment of severe VOD in adults and children undergoing HSCT therapy; |
| • | Erwinaze® (asparaginase Erwinia chrysanthemi), a treatment approved in the U.S. and in certain markets in Europe (where it is marketed as Erwinase®) for patients with acute lymphoblastic leukemia, or ALL, who have developed hypersensitivity to E. coli-derived asparaginase; and |
| • | Vyxeos® (daunorubicin and cytarabine) liposome for injection, a product approved in the U.S. and in Europe (where it is marketed as Vyxeos® liposomal 44 mg/100 mg powder for concentrate for solution for infusion) for the treatment of adults with newly-diagnosed therapy-related acute myeloid leukemia, or t-AML, or |
58
Over the U.S. and Europe for solriamfetol as a treatment to improve wakefulness and reduce EDS in adult patients with narcolepsy or obstructive sleep apnea, or OSA. Iflast five years, we are successful in obtaining FDA approval, we expect to launchachieved multiple significant regulatory approvals, including most recently the product in the U.S. after scheduling review by the U.S. Drug Enforcement Administration, or DEA, which will need to be completed afterEuropean approval of ourSunosi, and executed on five product launches. Over the next two years, we look forward to three additional potential regulatory approvals and related product launches (lurbinectedin, JZP-258 and JZP-458), as well as the commencement of the rolling launch of Sunosi in Europe by mid-2020. In February 2020, the FDA accepted for filing with priority review the new drug application, or NDA, if any, but before commercial launch.
Our strategy to create shareholder value is focused on:
Strong financial execution through growth in sales of our current lead marketed products;
Building a diversified product portfolio and development pipeline through a combination of our internal research and development efforts and obtaining rights to clinically meaningful and differentiated on- or near-market products and early- to late-stage product candidates through acquisitions, collaborations, licensing arrangements, partnerships and venture investments; and
Maximizing the value of our products and product candidates by continuing to implement our comprehensive global development plans, including through generating additional clinical data and seeking regulatory approval for new indications.indications and new geographies.
Our total net product sales increased by 17%14% in 20182019 compared to 2017,2018, primarily due to an increase in Xyrem net product sales and a full year of net product sales of Vyxeos, which launched in the U.S. in August 2017.sales. We expect total net product sales to increase in 20192020 over 2018,2019, primarily due to expected growth in sales of Xyrem and Vyxeos.Sunosi.
In 2018, in support of2019, consistent with our strategy, we continued to expand and advance our research and development pipeline in our sleepsleep/neuroscience and hematology/oncology therapeutic areas, both by conducting activities internally and by leveraging partnerships with third parties.
While we are focused on opportunities within our sleep/neuroscience and hematology/oncology therapeutic areas, such as our recent expansion into movement disorders and solid tumors, we are also exploring and investing in adjacent therapeutic areas that could further diversify our portfolio. Our development activities encompass all stages of development and currently include clinical testing of new product candidates and activities related to clinical improvements of, or additional indications or new clinical data for, our existing marketed products. We have also expanded into preclinical exploration of novel therapies, including precision medicines in hematology and oncology. A summary of our ongoing development activities is provided under “Business—Research and Development” in Part I, Item 1 of this Annual Report on Form 10-K.10‑K. In 20192020 and beyond, we expect that our research and development expenses will continue to increase from historicalprevious levels, particularly as we prepare for anticipated regulatory submissions and data read-outs from clinical trials, initiate and undertake additional clinical trials and related development work and potentially acquire rights to additional product candidates.
Regulatory Approvals and Launches
In August 2018, the European Commission, or EC, granted marketing authorization for VyxeosMarch 2019, we launched Xyrem for the treatment of adultscataplexy and EDS in pediatric patients with newly-diagnosed t-AMLnarcolepsy, after completing the implementation of the related approved risk evaluation and mitigation strategy, or AML-MRC, and shortly thereafter, we commenced a rolling launch of Vyxeos in the European Union, or EU.
REMS, modification. In October 2018,May 2019, the FDA approved our supplemental NDAconfirmed that as the first sponsor to revise the labelingobtain marketing approval for use of Xyrem to include an indication to treat cataplexy orand EDS in pediatric narcolepsy patients agesaged seven years and older, we are entitled to seven years of orphan drug exclusivity for the pediatric indication.
In March 2019, the FDA approved our NDA for Sunosi as a treatment to improve wakefulness in adult patients with EDS associated with narcolepsy or OSA, and granted Xyrem pediatric exclusivity, adding six monthsrecommended that solriamfetol be scheduled by the U.S. Drug Enforcement Administration, or DEA. In June 2019, the DEA designated solriamfetol as a Schedule IV controlled substance, and, in July 2019, we launched Sunosi in the U.S.
| • | In June 2019, our partner, Nippon Shinyaku Co., Ltd, announced that Japan's Ministry of Health, Labour and Welfare approved the marketing authorization of Defitelio® injection 200mg (defibrotide sodium) for the treatment of sinusoidal obstruction syndrome/hepatic VOD. |
59
In November 2019, the European Medicines Agency recommended the marketing authorization application for Sunosi in Europe, and in January 2020, the EC approved Sunosi to any regulatoryimprove wakefulness in adult patients with EDS associated with narcolepsy or Orange Book patent exclusivity.OSA.
Regulatory Submissions
In March 2018,2019, we announced positive top-line results from our Phase 3 study evaluating the FDA accepted for filing our NDA seeking marketing approval for solriamfetolefficacy and safety of JZP-258 for the treatment of cataplexy and EDS associated with OSA or narcolepsy in adult patients; the target action datepatients with narcolepsy and presented additional results from this study publicly at an international medical conference in September 2019. We submitted an NDA for this product in January 2020 and redeemed our priority review voucher, or PRV, in connection with this submission.
In December 2019, we entered into an exclusive license agreement with Pharma Mar, S.A., or PharmaMar, pursuant to which we obtained exclusive U.S. development and commercialization rights to lurbinectedin, a product candidate under clinical investigation for the treatment of patients with relapsed SCLC. Lurbinectedin was granted orphan drug designation for SCLC by the FDA underin August 2018. In December 2019, PharmaMar submitted an NDA to the FDA for accelerated approval of lurbinectedin for relapsed SCLC based on data from a Phase 2 trial, and in February 2020, the FDA accepted the NDA for filing with priority review with a Prescription Drug User Fee Act, or PDUFA, for a new molecular entity, originally December 20, 2018, was extended by the FDA to March 20, 2019.
Research & Development
In August 2018,July 2019, we announced a five-year collaboration with The University of Texas MD Anderson Cancer Center to evaluatethat we are pursuing development activities for Sunosi for the potential treatment options for hematologic malignancies,of EDS in patients with a near-term focus on Vyxeos, and shortly thereafter, commenced development activities pursuantmajor depressive disorder. We expect to this collaboration.
| • | In October 2019, the FDA granted Fast Track designation to JZP-458, a recombinant Erwinia asparaginase product candidate, for the potential treatment of ALL and LBL and in December 2019, we announced enrollment of the first patient in this study. |
In October 2019, we announced enrollment of the treatment of EDS and cataplexyfirst patient in narcolepsy, (ii) commencing patient enrollment in our Phase 3 clinical trial evaluating JZP-258 for the treatment of idiopathic hypersomnia and (iii) commencing patient enrollment in ouran exploratory Phase 2 clinical trial evaluating the ability of defibrotide to prevent neurotoxicity in patients with relapsed or refractory diffuse large B-cell lymphoma receiving chimeric antigen receptor T-cell therapy.
In October 2019, we completed enrollment in a Phase 2 study for defibrotide in the prevention of acute Graft-versus-Host Disease.graft-vs-host disease.
In December 2019, we activated sites for a Phase 1b master trial of Vyxeos in combination with various targeted agents in first-line, fit AML.
Other Significant Developments
In January 2019, we entered into a strategic collaboration agreement with Codiak BioSciences, Inc., or Codiak, focused on the research, development and commercialization of exosome therapeutics to treat cancer.
In February 2019, we received a contract termination notice from Porton Biopharma Limited, or PBL, the sole manufacturer of Erwinaze. As a result of our receipt of the contract termination notice, our license and supply agreement with PBL, which includes an exclusive right to market, sell or distribute Erwinaze, an exclusive license to Erwinaze trademarks, and a non-exclusive license to PBL’s manufacturing know-how, will expire on December 31, 2020. Unless we and PBL enter into a new agreement, we will lose our licensed rights to exclusively market Erwinaze effective December 31, 2020, other than our right to sell certain Erwinaze inventory for a post-termination sales period of 12 months and certain other post-termination rights, including but not limited to intellectual property and data ownership.
In April 2019, we finalized a settlement agreement with the U.S. Department of Justice, or DOJ, and the Office of Inspector General of the Department of Health and Human Services, or OIG, and we entered into a corporate integrity agreement requiring us to maintain our ongoing corporate compliance program and obligating us to implement or continue, as applicable, a set of defined corporate integrity activities for a period of five years from the effective date of the corporate integrity agreement.
In June 2019, we received notice from ImmunoGen, Inc., or ImmunoGen, that, as a result of portfolio prioritization and restructuring initiatives, ImmunoGen will be discontinuing development of its IMGN779 antibody-drug conjugate, or ADC, product candidate, for which we possess opt-in rights, as well as the programs from which a third opt-in candidate was to be selected. IMGN632, a CD123-targeted ADC product candidate for which we possess opt-in rights, remains under development by ImmunoGen.
In July 2019, we acquired from Redx Pharma plc, or Redx, a pan-RAF inhibitor program for the potential treatment of RAF and RAS mutant tumors. Under the terms of our agreement with Redx, we paid Redx $3.5 million at closing
and Redx is eligible to receive up to $203 million in development, regulatory and commercial milestone payments from us, as well as incremental tiered royalties in mid-single digit percentage based on any future net sales.
In August 2019, we announced the acquisition of Cavion, Inc., a clinical-stage biotechnology company, or Cavion, for an upfront payment of $52.5 million with the potential for additional payments of up to $260.0 million upon the achievement of certain clinical, regulatory and commercial milestones, for a total potential consideration of $312.5 million. As a result of the acquisition, we added CX-8998, now named JZP-385, a modulator of T-type calcium channels, for the potential treatment of essential tremor, to our clinical pipeline.
Beginning in the third quarter of 2019, we have been entering into agreements with commercial payor organizations, including pharmacy benefit managers, or PBMs, to ensure patient access for our products, Sunosi and Xyrem, and to support the long-term success of our sleep and neuroscience therapeutic area. These agreements include terms related to the payment of rebates and/or administrative fees on these products. We expect to enter into additional agreements in 2020 to continue to ensure patient access to and coverage for our products.
In October 2019, our board of directors authorized the additional repurchase of our ordinary shares having an aggregate purchase price of up to $500.0 million, exclusive of any brokerage commissions, under our share repurchase program. During 2019, we repurchased an aggregate of $301.5 million of our ordinary shares under our share repurchase program at an average price of $133.97 per share.
Challenges, Risks and Trends Related to Our Business
As for other products and product candidates in our sleep and neuroscience therapeutic area, we obtained approval of Sunosi in the U.S. and EU, and in January 2020, we submitted an NDA for marketing approval of JZP-258. Our future plans assume that, if approved, JZP-258 may be prescribed to patients as a safer and clinically superior alternative to Xyrem as well as being prescribed to patients who may otherwise be ineligible to take Xyrem based on its high sodium content. We are aware that Avadel Pharmaceuticals plc, or Avadel, is conducting a Phase 3 clinical trial of a once-nightly formulaformulation of sodium oxybate which uses its proprietary technology for the treatment of EDS and cataplexy in patients with narcolepsy, and that Avadel has indicated that it intends to seek approval of its product candidate inusing the U.S. under a Section 505(b)(2) NDA approval pathway. Other companiespathway referencing the safety and efficacy data for Xyrem. If the FDA approves Avadel’s product and grants it orphan drug exclusivity before we obtain approval for JZP-258, there is a risk that JZP-258 will not be approvable for a narcolepsy indication for seven years unless it can establish clinical superiority to Avadel’s product. We cannot predict the timing of Avadel’s submission or how the FDA will evaluate any clinical superiority arguments that either company may also developmake, but a sodium oxybatedelay in approval or similar product using,inability to obtain approval for example, an alternative formulation or a different delivery technology and pursue a similar regulatory approval strategy in the future.
A key aspect of our growth strategy is our continued investment in our evolving and expanding research and development activities. In addition,neuroscience, we cannot predict whether uncertainty relatedare developing JZP-385 for the treatment of essential tremor and expect to rightsinitiate a Phase 2b study of JZP-385 in the fourth quarter of 2020. In our hematology/oncology, we are developing JZP-458,a
61
recombinant Erwinia asparaginase, and we expect to and availabilitysubmit a BLA to the FDA for JZP-458 as early as the fourth quarter of Erwinaze after 2020 will impact2020. If we are not successful in the clinical development of these or other product candidates, if we are unable to obtain regulatory approval for our product candidates in a timely manner, or at all, or if sales of and revenues from this product.
We are increasingly experiencing pressure from third party payors to agree to discounts, rebates or other restrictive pricing terms for Xyrem. As our business becomes more complex, we mayWe also need to enterobtain adequate formulary positions and reimbursement coverage for newly-launched products such as Sunosi and future products, if approved, such as JZP-258, JZP-458 and lurbinectedin. Entering into rebate agreements in orderwith payors or PBMs to ensure that patientspatient access has and will likely continue to haveresult in higher gross to net deductions for future periods for these products. We cannot guarantee we will be able to agree to commercially reasonable terms with PBMs and other third party payors, or that we will be able to ensure patient access to Xyrem,our existing and to support the long-term success of our sleep franchise, which might result in lower net revenues for Xyrem.future products. In addition to increasing pricing pressure from, and restrictions on reimbursement imposed by governmental and private third party payors, due to the attention being paid globally to healthcare cost containment has received global attention, and drug pricing by pharmaceutical companies is currently, and is expected to continue to be, undersubject to close scrutiny by both federal and state governments, includinggovernments. If healthcare policies or reforms intended to curb healthcare costs are adopted or if we experience negative publicity with respect to pricing of our products or the pricing of pharmaceutical drugs generally, the prices that we charge for our products may be affected, our commercial opportunity may be limited and/or our revenues from sales of our products may be negatively impacted.
Finally, business practices by pharmaceutical companies, that have increased the price of products after acquiring those products from other companies. In addition,including product formulation improvements, patent settlements, REMS programs, have increasingly drawn public scrutiny from the U.S. Congress, the Federal Trade Commissionlegislators and the FDA,regulatory agencies, with allegations that such programs are used as a means of improperly blocking or delaying competition. If we become the subject of any future government investigation with respect to drug pricing or otherour business practices, including as they relate to the Xyrem REMS, the launch of JZP-258, our patent settlements or otherwise, we could incur significant expense and could be distracted from operation of our business and execution of our strategy.
Any of these risks and uncertainties could have a material adverse effect on our business, financial condition, results of operations and growth prospects. All of these risks are discussed in greater detail, along with other risks, in “Risk Factors” in Part I, Item 1A of this Annual Report on Form 10-K.10‑K.
Results of Operations
The following table presents revenues and expenses for the years ended December 31, 2019, 2018 2017 and 20162017 (in thousands except percentages):
| 2018 | Change | 2017 | Change | 2016 (1) | 2019 | Change | 2018 | Change | 2017 | |||||||||||||||||||||||||
| Product sales, net | $ | 1,869,473 | 17 | % | $ | 1,601,399 | 8 | % | $ | 1,477,261 | $ | 2,135,601 | 14 | % | $ | 1,869,473 | 17 | % | $ | 1,601,399 | ||||||||||||||
| Royalties and contract revenues | 21,449 | 24 | % | 17,294 | 61 | % | 10,712 | 26,160 | 22 | % | 21,449 | 24 | % | 17,294 | ||||||||||||||||||||
| Cost of product sales (excluding amortization of intangible assets) | 121,544 | 10 | % | 110,188 | 5 | % | 105,386 | |||||||||||||||||||||||||||
| Cost of product sales (excluding amortization of acquired developed technologies) | 127,930 | 5 | % | 121,544 | 10 | % | 110,188 | |||||||||||||||||||||||||||
| Selling, general and administrative | 683,530 | 26 | % | 544,156 | 8 | % | 502,892 | 736,942 | 8 | % | 683,530 | 26 | % | 544,156 | ||||||||||||||||||||
| Research and development | 226,616 | 14 | % | 198,442 | 22 | % | 162,297 | 299,726 | 32 | % | 226,616 | 14 | % | 198,442 | ||||||||||||||||||||
| Intangible asset amortization | 201,498 | 33 | % | 152,065 | 49 | % | 101,994 | 354,814 | 76 | % | 201,498 | 33 | % | 152,065 | ||||||||||||||||||||
| Impairment charges | 42,896 | N/A(2) | — | N/A(2) | — | — | N/A(1) | 42,896 | N/A(1) | — | ||||||||||||||||||||||||
| Acquired in-process research and development | — | N/A(2) | 85,000 | N/A(2) | 23,750 | 109,975 | N/A(1) | — | N/A(1) | 85,000 | ||||||||||||||||||||||||
| Interest expense, net | 77,075 | (1 | )% | 77,756 | 26 | % | 61,942 | 72,261 | (6 | )% | 77,075 | (1 | )% | 77,756 | ||||||||||||||||||||
| Foreign exchange loss (gain) | 6,875 | (31 | )% | 9,969 | (396 | )% | (3,372 | ) | ||||||||||||||||||||||||||
| Foreign exchange loss | 5,811 | (15 | )% | 6,875 | (31 | )% | 9,969 | |||||||||||||||||||||||||||
| Loss on extinguishment and modification of debt | 1,425 | N/A(2) | — | N/A(2) | 638 | — | N/A(1) | 1,425 | N/A(1) | — | ||||||||||||||||||||||||
| Income tax provision (benefit) | 80,162 | (269 | )% | (47,470 | ) | (135 | )% | 135,236 | (73,154 | ) | N/A(1) | 80,162 | N/A(1) | (47,740 | ) | |||||||||||||||||||
| Equity in loss of investees | 2,203 | 118 | % | 1,009 | 166 | % | 379 | 4,089 | 86 | % | 2,203 | 118 | % | 1,009 | ||||||||||||||||||||
_________________________
| (1) |
| Comparison to prior period is not meaningful. |
Revenues
The following table presents product sales, royalties and contract revenues, and total revenues for the years ended December 31, 2019, 2018 2017 and 20162017 (in thousands except percentages):
| 2018 | Change | 2017 | Change | 2016 | 2019 | Change | 2018 | Change | 2017 | |||||||||||||||||||||||||
| Xyrem | $ | 1,404,866 | 18 | % | $ | 1,186,699 | 7 | % | $ | 1,107,616 | $ | 1,642,525 | 17 | % | $ | 1,404,866 | 18 | % | $ | 1,186,699 | ||||||||||||||
| Erwinaze/Erwinase | 174,739 | (11 | )% | 197,340 | (2 | )% | 200,678 | 177,465 | 2 | % | 174,739 | (11 | )% | 197,340 | ||||||||||||||||||||
| Defitelio/defibrotide | 149,448 | 12 | % | 133,650 | 23 | % | 108,952 | 172,938 | 16 | % | 149,448 | 12 | % | 133,650 | ||||||||||||||||||||
| Vyxeos | 100,835 | 198 | % | 33,790 | N/A(1) | — | 121,407 | 20 | % | 100,835 | N/A(1) | 33,790 | ||||||||||||||||||||||
| Prialt | 20,839 | (24 | )% | 27,361 | (6 | )% | 29,120 | |||||||||||||||||||||||||||
| Sunosi | 3,714 | N/A(1) | — | N/A(1) | — | |||||||||||||||||||||||||||||
| Other | 18,746 | (17 | )% | 22,559 | (27 | )% | 30,895 | 17,552 | (56 | )% | 39,585 | (21 | )% | 49,920 | ||||||||||||||||||||
| Product sales, net | 1,869,473 | 17 | % | 1,601,399 | 8 | % | 1,477,261 | 2,135,601 | 14 | % | 1,869,473 | 17 | % | 1,601,399 | ||||||||||||||||||||
| Royalties and contract revenues | 21,449 | 24 | % | 17,294 | 61 | % | 10,712 | 26,160 | 22 | % | 21,449 | 24 | % | 17,294 | ||||||||||||||||||||
| Total revenues | $ | 1,890,922 | 17 | % | $ | 1,618,693 | 9 | % | $ | 1,487,973 | $ | 2,161,761 | 14 | % | $ | 1,890,922 | 17 | % | $ | 1,618,693 | ||||||||||||||
_________________________
| (1) | Comparison to prior period is not meaningful. |
Product Sales, Net
Xyrem product sales increased by 17% in 2019 compared to 2018 primarily due to a higher average net selling price and, to a lesser extent, an increase in sales volume. Price increases were instituted in January and July 2019, January 2018 and in January and July 2017. Xyrem product sales volume increased by 6% in 2019 compared to 2018 primarily driven by an increase in the average number of patients on Xyrem. Xyrem product sales increased by 18% in 2018 compared to 2017 primarily due to an increase in sales volume and, to a lesser extent, a higher average net selling price. Xyrem product sales volume increased by 9% in 2018 compared to 2017 primarily driven by an increase in the average number of patients on Xyrem. Price increases were instituted in January 2018 and in January and July 2017. XyremErwinaze/Erwinase product sales increased by 7% in 20172019 compared to 20162018 primarily due to higher average net selling prices, partially offset by higher gross to net deductions. Sales volumes in 2017 were consistent with 2016; sales growth was impacted by payor mix throughout 2017 and by operational changes that delayed some prescription fulfillment involume as a result of availability of supply from the second half of 2017.manufacturer. Erwinaze/Erwinase product sales decreased in 2018 and 2017 compared to the immediately preceding years2017 primarily due to lower sales volume as a result of limited supply from the manufacturer. Ongoing supply challenges continue to negatively impact our ability to supply the market. We are experiencingexperienced limited product availability of Erwinaze and supply disruptions globally in 2019 and expect furthermay experience continued supply disruptions during 2019.in 2020. Defitelio/defibrotide product sales increased in 2019 compared to 2018, primarily due to higher sales volumes, partially offset by the negative impact of foreign exchange rates. Defitelio/defibrotide product sales increased in 2018 compared to 2017, primarily due to higher sales volumes
63
and, to a lesser extent, the positive impact of foreign exchange rates. Defitelio/defibrotideVyxeos product sales
increased in 20172019 compared to 2016,2018 primarily due to volumes following the impact of a full year of salescommercial launch in the U.S.Europe in 2017.September 2018. Vyxeos product sales in 2018 and 2017 were $100.8 million and $33.8 million respectively,in 2017 following its launch in the U.S. in August 2017 and rolling launch in the EU beginning in August 2018. Prialt2017. Other product sales decreased in 20182019 and 2017in 2018 compared to the immediately preceding years, primarily due to a decrease in volume. We completed the sale of our rights to Prialt to TerSera Therapeutics LLC, or TerSera, in September 2018. Other product sales decreased in 2018 and in 2017 compared to the immediately preceding years, primarily due to a decrease in sales of our psychiatry products due to generic competition. We expect total product sales will increase in 20192020 over 2018,2019, primarily due to expected growth in sales of Xyrem and Vyxeos.Sunosi.
Royalties and Contract Revenues
Royalties and contract revenues increased in 20182019 and in 20172018 compared to the immediately preceding years, primarily due to higher one-time milestonecontract revenues from out-licensing agreements. We expect royalties and contract revenues to decrease in 20192020 compared to 20182019 primarily due to lower milestone revenues from out-licensing arrangements.
Cost of Product Sales
Cost of product sales increased in 20182019 and in 20172018 compared to the immediately preceding years, primarily due to changes in product mix and increases in net product sales. Gross marginsmargin as a percentage of net product sales werewas 94.0%, 93.5%, and 93.1% in 2019, 2018 and 92.9% in 2018, 2017, and 2016, respectively. The increase in the gross margin percentage in 20182019 and in 20172018 compared to the immediately preceding years was primarily due to changes in product mix. We expect that our gross margin as a percentage of net product sales will not change materially in 20192020 compared to 2018.2019.
Selling, General and Administrative Expenses
Selling, general and administrative expenses increased in 20182019 compared to 20172018 primarily due to an accrued estimated loss contingency, including related interest, of $58.2 million. In April 2018, we reached an agreement in principle with the DOJ on a proposal for a civil settlement of potential claims by the DOJ in the amount of $57.0 million, subject to accrual of interest on the settlement amount from the date of the agreement in principle, negotiation of a definitive settlement agreement and other contingencies. For a further description of this matter, see Note 12, Commitments and Contingencies—Legal Proceedings, of the Notes to Consolidated Financial Statements included in Part IV of this Annual Report on Form 10-K. Selling, general and administrative expenses in 2018 also includedhigher expenses related to the potential commercial launch of solriamfetolSunosi in the U.S. and the rolling launch of Vyxeos in the EU, and, an increase in compensation-related expenses driven by higher headcount compared to 2017. Selling, general and administrative expenses increased in 2017 compared to 2016, primarily due to an increase in compensation-related expenses, primarily driven by higher headcount, and an increase in other expenses related to the expansion and support of our business, partially offset by the recognition of a loss contingency including related interest of $58.2 million recorded in 2018 resulting from a settlement agreement with the U.S. Department of Justice and the Office of Inspector General. For a further description of this matter, see the risk factors under the heading “Other Risks Related to Our Business and Industry” in Part I, Item 1A of this Annual Report on Form 10-K. Selling, general and administrative expenses increased in 2018 compared to 2017 primarily due to the recognition of a loss contingency and related interest recorded in 2018, increased expenses related to the commercial launch of Sunosi in the U.S. and the rolling launch of Vyxeos in the U.S., partially offsetEurope, and an increase in compensation-related expenses driven by the impact in 2016 of transaction and integration expenses relatedhigher headcount compared to the Celator Acquisition of $13.1 million and a one-time contract termination fee of $11.6 million to eliminate a potential future royalty obligation related to Vyxeos.2017. We expect selling, general and administrative expenses in 20192020 to increase compared to 2018,2019, primarily due to an increase in compensation-related expenses driven by higher headcount and other expenses related to the expansion and support of our business andincluding an increase in expenses related to the preparation for the potential commercial launch of solriamfetol in the U.S. and the continuation of the commercial launch of VyxeosSunosi in the EU.U.S., the commercial launch of Sunosi in Europe and the planned commercial launch of both lurbinectedin and JZP-258 in the U.S.
Research and Development Expenses
Research and development expenses consist primarily of costs related to clinical studies and outside services, personnel expenses, milestone paymentsexpenses and other research and development costs. Clinical study and outside services costs relate primarily to services performed by clinical research organizations, materials and supplies, and other third party fees. Personnel expenses relate primarily to salaries, benefits and share-based compensation. Other research and development expenses primarily include overhead allocations consisting of various support and facilities-related costs. We do not track fully-burdened research and development expenses on a project-by-project basis. We manage our research and development expenses by identifying the research and development activities that we anticipate will be performed during a given period and then prioritizing efforts based on our assessment of which development activities are important to our business and have a reasonable probability of success, and by dynamically allocating resources accordingly. We also continually review our development pipeline projects and the status of their development and, as necessary, reallocate resources among our development pipeline projects that we believe will best support the future growth of our business.
The following table provides a breakout of our research and development expenses by major categories of expense (in thousands):
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| 2018 | 2017 | 2016 | 2019 | 2018 | 2017 | |||||||||||||||||
| Clinical studies and outside services | $ | 117,903 | $ | 93,317 | $ | 100,165 | $ | 133,042 | $ | 117,903 | $ | 93,317 | ||||||||||
| Personnel expenses | 71,158 | 63,941 | 47,969 | 100,090 | 71,158 | 63,941 | ||||||||||||||||
| Milestone expense | 11,000 | 19,500 | 750 | 26,000 | 11,000 | 19,500 | ||||||||||||||||
| Other | 26,555 | 21,684 | 13,413 | 40,594 | 26,555 | 21,684 | ||||||||||||||||
| Total | $ | 226,616 | $ | 198,442 | $ | 162,297 | $ | 299,726 | $ | 226,616 | $ | 198,442 | ||||||||||
64
Research and development expenses increased by $73.1 million in 2019 compared to 2018. Clinical studies and outside services costs increased in 2019 compared to 2018 primarily due to an increase in expenses related to our ongoing preclinical and clinical development programs and support of partner programs. Personnel expenses increased by $28.9 million in 2019 compared to 2018, primarily due to increased headcount in support of our development programs. Milestone expense of $26.0 million in 2019 related to milestone payments made under our license and option agreement with Pfenex. Research and development expenses increased by $28.2 million in 2018 compared to 2017. Clinical studies and outside services costs increased in 2018 compared to 2017 primarily due to an increase in expenses related to our ongoing preclinical and clinical development programs and support of partner programs, partially offset by lower clinical trial costs following the completion of three Phase 3 clinical trials for solriamfetol.Sunosi. Personnel expenses increased by $7.2 million in 2018 compared to 2017, primarily due to increased headcount in support of our development programs. Milestone expense of $11.0 million in 2018 related to milestone payments following FDA acceptance of our NDA for solriamfetolSunosi in March 2018. Research and development expenses increased by $36.1 million in 2017 compared to 2016. Clinical studies and outside services costs decreased in 2017 compared to 2016 primarily due to lower clinical trial costs following the completion of three Phase 3 clinical trials for solriamfetol, partially offset by an increase in expenses related to other clinical development programs and higher costs in respect of regulatory activities. Personnel expenses increased by $16.0 million in 2017 compared to 2016, primarily driven by increased headcount in support of our development programs. Milestone expense in 2017 related to payments made under the license and option agreement with Pfenex, Inc., or Pfenex, which we entered into in July 2016 and amended in December 2017, for worldwide rights to develop and commercialize multiple early-stage hematology product candidates.
For 20192020 and beyond, we expect that our research and development expenses will continue to increase from historicalprevious levels, particularly as we prepare for anticipated regulatory submissions and data read-outs from clinical trials, initiate and undertake additional clinical trials and related development work and potentially acquire rights to additional product candidates. A discussion of the risks and uncertainties with respect to our research and development activities, including completing the development of and regulatory submissions for our product candidates, and the consequences to our business, financial position and growth prospects can be found in “Risk Factors” in Part I, Item 1A of this Annual Report on Form 10-K.10‑K.
Intangible Asset Amortization
Intangible asset amortization increased in 2018 and in 20172019 compared to 2018 primarily due to the immediately preceding years,amortization of the cost of the PRV of $111.1 million in full following the notification to the FDA of our intention to redeem it in the NDA submission for JZP-258 and the reduction in the estimated remaining useful life of the Erwinaze intangible asset resulting from the contract termination notice we received from PBL in February 2019. Intangible asset amortization increased in 2018 compared to 2017 primarily due to the commencement of amortization of the Vyxeos intangible asset upon FDA approval in August 2017. Intangible asset amortization is expected to increasedecrease in 20192020 compared to 20182019 as a result of the reductionamortization in full of our PRV intangible asset in the estimated remaining useful lifefourth quarter of the Erwinaze intangible asset due to the receipt of a contract termination notice from PBL in February 2019.
Impairment Charges
In June 2018, we entered into an asset purchase agreement, or APA, with TerSera, pursuant to which TerSera agreed to purchase substantially all of the assets held by us related to Prialt. In connection with the entry into the APA, which was subsequently amended, we reclassified the Prialt assets to be transferred to TerSera as assets held for sale and recorded these assets at fair value, less estimated sales costs, resulting in the recognition of an impairment charge of $42.9 million in 2018. The transaction closed onin September 27, 2018.
Acquired In-Process Research and Development
In 2017,2019, acquired in-process research and development, or IPR&D expense primarily related to an upfront payment of $56.0 million to Codiak in connection with our strategic collaboration agreement and the value attributed to JZP-385 in the acquisition of Cavion.
In 2017, IPR&D expense was primarily related to an upfront payment of $75.0 million in connection with aour collaboration and option agreement with ImmunoGen, Inc., or ImmunoGen to acquire rights to opt into exclusive, worldwide licenses to develop and commercialize two hematology-related antibody-drug conjugate, or ADC programs, as well as an additional program to be designated during the term of the agreement.
Interest Expense, Net
Interest expense, net decreased by $4.8 million in 2019 compared to 2018, primarily due to higher interest income. Interest expense, net decreased by $0.7 million in 2018 compared to 2017, primarily due to higher interest income, partially offset by the interest expense on our 1.50% exchangeable senior notes due 2024, or the 2024 Notes, which were issued in August 2017. Interest expense, net increased by $15.8 million in 2017 compared to 2016, primarily due to the increase in
Foreign Exchange Loss (Gain)
The foreign exchange loss (gain) is primarily related to the translation of euro-denominated net monetary liabilities, primarily intercompany balances, held by subsidiaries with a U.S. dollar functional currency and related foreign exchange forward contracts not designated as hedging instruments.
Loss on Extinguishment and Modification of Debt
In 2018, we recorded a loss of $1.4 million in connection with the second amendment of our 2015 credit agreement, in June 2018, related to unamortized debt issuance costs and original issue discount associated with extinguished debt and new third party fees associated with modified debt. In 2016, we recorded a loss
65
Income Tax Provision (Benefit)
Our income tax provisionbenefit was $80.2$73.2 million and $135.2$47.7 million in 20182019 and 2016,2017, respectively, and our income tax benefitprovision was $47.7$80.2 million in 2017.2018. The income tax provisionbenefit for 2019 includes a discrete tax benefit of $112.3 million resulting from an intra-entity intellectual property asset transfer. The income tax benefit, which represents a deferred future benefit, was recorded as a deferred tax asset. The income tax benefit in 2017 included a benefit of $148.8 million relating to the enactment of the U.S. Tax Cuts and Jobs Act, or the U.S. Tax Act. The effective tax rates for 2019, 2018 and 2017 were (16.1)%, 15.1% and 2016 were 15.1%, (10.8)% and 25.4%, respectively. The income tax benefit recognized for 2017 in respect of the enactment of the U.S. Tax Act resulted in a net decrease to our effective tax rate for 2019 was lower than the Irish statutory rate of 33.7%.12.5% primarily due to the impact of the intra-entity intellectual property asset transfer. The effective tax ratesrate for 2018 and 2016 werewas higher than the Irish statutory rate of 12.5%, primarily due to income taxable at a rate higher than the Irish statutory rate and unrecognized tax benefits, partially offset by the release of reserves related to unrecognized tax benefits upon the expiration of a statute of limitation, originating tax credits and the release of a valuation allowance held primarily against certain foreign net operating losses or NOLs. The effective tax rate for 2017 was lower than the Irish statutory rate of 12.5%, primarily due to the impact of the enactment of the U.S. Tax Act. The decrease in the effective tax rate for 2019 compared to 2018 was primarily due to the impact of the intra-entity intellectual property asset transfer. Excluding this effect, the decrease in the effective tax rate for 2019 compared to 2018 was primarily due to the benefit from the application of the Italian patent box incentive regime. The increase in the effective tax rate for 2018 compared to 2017 was primarily due to the impact of the enactment of the U.S. Tax Act. Excluding this effect, the effective rate in 2018 would have decreased compared to 2017 primarily due to a decrease in the U.S. corporate income tax rate. The decrease in the effective tax rate in 2017 compared to 2016 was primarily due to the release of a valuation allowance held against certain foreign NOLs and the release of reserves related to unrecognized tax benefits upon the expiration of a statute of limitation, partially offset by a reduction in deductions available in relation to subsidiary equity.
Equity in Loss of Investees
Equity in loss of investees relates to our share in the net loss of companies in which we have made investments accounted for under the equity method of accounting.
Liquidity and Capital Resources
As of December 31, 2018,2019, we had cash, cash equivalents and investments of $824.6 million,$1.1 billion, borrowing availability under our revolving credit facility of $1.6 billion and a long-term debt principal balance of $1.8 billion. Our long-term debt included $651.0$617.7 million aggregate principal amount term loan, $575.0 million principal amount of theour 1.875% exchangeable senior notes due 2021 Notes and $575.0 million principal amount of the 2024 Notes.our 1.50% exchangeable senior notes due 2024. During 2019, 2018 2017 and 2016,2017, we generated cash flows from operations of $776.4 million, $798.9 million $693.1 million and $592.4$693.1 million, respectively, and we expect to continue to generate positive cash flow from operations.
We believe that our existing cash, cash equivalents and investments balances, cash we expect to generate from operations and funds available under our revolving credit facility will be sufficient to fund our operations and to meet our existing obligations for the foreseeable future. The adequacy of our cash resources depends on many assumptions, including primarily our assumptions with respect to product sales and expenses, as well as the other factors set forth in “Risk Factors” in Part I, Item 1A of this Annual Report on Form 10-K10‑K under the headings “Risks Related to Xyremour Lead Products and Our Other Marketed Products”Product Candidates” and “To continue to grow our business, we will need to commit substantial resources, which could result in future losses or otherwise limit our opportunities or affect our ability to operate our business.” Our assumptions may prove to be wrong or other factors may adversely affect our business, and as a result we could exhaust or significantly decrease our available cash resources, and we may not be able to generate sufficient cash to service our debt obligations which could, among other things, force us to raise additional funds and/or force us to reduce our expenses, either of which could have a material adverse effect on our business.
To continue to grow our business over the longer term, we plan to commit substantial resources to product acquisition and in-licensing, product development, clinical trials of product candidates and expansion of our commercial, development, manufacturing and other operations. In this regard, we have evaluated and expect to continue to evaluate a wide array of
strategic transactions as part of our strategy to acquire or in-license and develop additional products and product candidates. Acquisition opportunities that we pursue could materially affect our liquidity and capital resources and may require us to incur additional indebtedness, seek equity capital or both. In addition, we may pursue new operations or continue the expansion of our existing operations. Accordingly, we expect to continue to opportunistically seek access to additional capital to license or acquire additional products, product candidates or companies to expand our operations or for general corporate purposes. Raising additional capital could be accomplished through one or more public or private debt or equity financings, collaborations or partnering arrangements. Any equity financing would be dilutive to our shareholders, and the consent of the lenders under the amended credit agreement could be required for certain financings.
In November 2015, our board of directors authorized a share repurchase program pursuant to which we were authorized to repurchase a number of ordinary shares having an aggregate purchase price of up to $300 million, exclusive of any brokerage commissions. In September 2016, we completed repurchases under the November 2015 share repurchase program. In November 2016, our board of directors authorized a new share repurchase program pursuant to which we were authorized to repurchase a number of ordinary shares having an aggregate purchase price of up to $300 million, exclusive of any brokerage commissions. In November and December 2018, our board of directors increased the existing share repurchase
66
program authorization by $320.0 million and $400.0 million, respectively, thereby increasingrespectively. In October 2019, our board of directors authorized the total amount authorizedadditional repurchase of shares having an aggregate purchase price of up to $1.02 billion.$500.0 million, exclusive of any brokerage commissions. Under this program, which has no expiration date, we may repurchase ordinary shares from time to time on the open market. The timing and amount of repurchases will depend on a variety of factors, including the price of our ordinary shares, alternative investment opportunities, restrictions under the amended credit agreement, corporate and regulatory requirements and market conditions. The repurchase program may be modified, suspended or discontinued at any time without prior notice. In 2018,2019, we spent a total of $523.7$301.5 million to purchase 3.52.3 million of our ordinary shares under the share repurchase program at an average total purchase price, including brokerage commissions, of $148.33$133.97 per share. In 2017,2018, we spent a total of $98.8$523.7 million to repurchase 0.73.5 million of our ordinary shares at an average total purchase price, including brokerage commissions, of $140.34$148.33 per share. All ordinary shares repurchased were canceled. As of December 31, 2018,2019, the remaining amount authorized under the share repurchase program was $379.1$577.7 million.
The following table shows a summary of our cash flows for the periods indicated (in thousands):
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| 2018 | 2017 | 2016 | 2019 | 2018 | 2017 | |||||||||||||||||
| Net cash provided by operating activities | $ | 798,904 | $ | 693,087 | $ | 592,391 | $ | 776,401 | $ | 798,904 | $ | 693,087 | ||||||||||
| Net cash used in investing activities | (394,487 | ) | (268,950 | ) | (1,751,155 | ) | (155,300 | ) | (394,487 | ) | (268,950 | ) | ||||||||||
| Net cash provided by (used in) financing activities | (479,130 | ) | (409,111 | ) | 540,987 | |||||||||||||||||
| Net cash used in financing activities | (293,745 | ) | (479,130 | ) | (409,111 | ) | ||||||||||||||||
| Effect of exchange rates on cash and cash equivalents | (1,700 | ) | 5,046 | (5,045 | ) | 366 | (1,700 | ) | 5,046 | |||||||||||||
| Net increase (decrease) in cash and cash equivalents | $ | (76,413 | ) | $ | 20,072 | $ | (622,822 | ) | $ | 327,722 | $ | (76,413 | ) | $ | 20,072 | |||||||