DOCUMENTS INCORPORATED BY REFERENCE
ACCELERON PHARMA INC.
FORM 10-K
INDEX
| Page | ||
FORWARD-LOOKING STATEMENTS
This Annual Report filed on Form 10-K and the information incorporated herein by reference includes statements that are, or may be deemed, "forward-looking statements." In some cases, these forward-looking statements can be identified by the use of forward-looking terminology. The terms "anticipate", "believe", "contemplate", "continue", "could", "estimate", "expect", "forecast", "goal", "intend", "may", "plan", "potential", "predict", "project", "should", "strategy", "target", "will", "would", "vision","believe," "contemplate," "continue," "could," "estimate," "expect," "forecast," "goal," "intend," "may," "plan," "potential," "predict," "project," "should," "strategy," "target," "will," "would," "vision," or, in each case, the negative or other variations thereon or other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
The forward-looking statements in this Annual Report on Form 10-K include, among other things, statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things:
our ongoing and planned preclinical studies and clinical trials;
clinical trial data and the timing of results of our ongoing clinical trials;
our plans to develop and commercialize dalanterceptACE-083, ACE-2494 and ACE-083, and our other preclinical therapeutic candidates;
our and Celgene's plans to develop and commercialize luspatercept and sotatercept;
the potential benefits of strategic partnership agreements and our ability to enter into selective strategic partnership arrangements;
the timing of anticipated milestone payments under our collaboration agreements with Celgene;
the timing of, and our and Celgene's ability to, obtain and maintain regulatory approvals for our therapeutic candidates;
the rate and degree of market acceptance and clinical utility of any approved therapeutic candidate, particularly in specific patient populations;
our ability to quickly and efficiently identify and develop therapeutic candidates;
our commercialization, marketing and manufacturing capabilities and strategy;
our plans for commercialization and marketing;
our intellectual property position; and
our estimates regarding our results of operations, financial condition, liquidity, capital requirements, prospects, growth and strategies.
By their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics, and industry change and depend on the economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated. We caution you that forward-looking statements are not guarantees of future performance and that our actual results of operations, financial condition and liquidity, and events in the industry in which we operate may differ materially from the forward-looking statements contained herein.
Any forward-looking statements that we make in this Annual Report on Form 10-K speak only as of the date of such statement, and we undertake no obligation to update such statements to reflect events or circumstances after the date of this Annual Report on Form 10-K or to reflect the occurrence of unanticipated events.
You should also read carefully the factors described in the "Risk Factors" section of this Annual Report on Form 10-K to better understand the risks and uncertainties inherent in our business and underlying any forward-looking statements. You are advised, however, to consult any further disclosures we make on related subjects in our Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, press releases, and our website.
Trademarks
We own or have rights to trademarks, service marks and trade names that we use in connection with the operation of our business, including our corporate name, logos and website names. Other trademarks, service marks and trade names appearing in this report are the property of their respective owners. The trademarks that we own include Acceleron Pharma® and IntelliTrap™. Solely for convenience, some of the trademarks, service marks and trade names referred to in this report are listed without the ® and ™ symbols, but we will assert, to the fullest extent under applicable law, our rights to our trademarks, service marks and trade names.
PART I
Item 1. Business
We are a clinical stageleading biopharmaceutical company focused onin the discovery and development of TGF-beta therapeutics to treat serious and commercialization of novel therapeutic candidates that are based on the mechanisms that the human body uses to regulate the growth and repair of its cells and tissues.rare diseases. Our research focuses on key natural regulators of cellular growth and repair, particularly the Transforming Growth Factor-Beta, or TGF-beta, protein superfamily. We are a leading company in discovering and developing therapeutic candidates that regulate cellular growth and repair. By combining our discovery and development expertise, including our proprietary knowledge of the TGF-beta superfamily, and our internal protein engineering and manufacturing capabilities, we have built a highly productive discovery and development platform that has generated several innovative therapeutic candidates, withall of which encompass novel potential first-in-class mechanisms of action. These differentiatedWe have focused and prioritized our research and development activities within three key therapeutic areas: hematologic, neuromuscular and pulmonary. If successful, these candidates could have the potential to significantly improve clinical outcomes for patients across many fieldsthese areas of medicine, and we have focused our discovery and development efforts on treatments for cancer and rare diseases.
Luspatercept, our lead program, and sotatercept, are partnered with Celgene Corporation, or Celgene. Luspatercept is an erythroid maturation agent designed to promote red blood cell production through a novel mechanism, and we are developing luspatercept with Celgeneis being developed to treat chronic anemia and associated complications in myelodysplastic syndromes, (MDS)or MDS, beta-thalassemia, and beta-thalassemia. In 2015,myelofibrosis. Celgene initiatedis currently conducting two Phase 3 clinical trials for luspaterceptwith luspatercept; one for the treatment of patients with lower-risk MDS, known as the "MEDALIST" trial, and beta-thalassemia.another for the treatment of patients with beta-thalassemia, also known as the "BELIEVE" trial. Celgene has recently initiated a Phase 2 trial in non-transfusion-dependent beta-thalassemia patients, referred to as the "BEYOND" trial. We further expect Celgene to initiate a Phase 3 clinical trial, the "COMMANDS" trial, in first-line, lower-risk MDS patients in the first half of 2018. Enrollment is also currently ongoing in a Phase 2 clinical trial for the treatment of patients with myelofibrosis, a rare bone marrow disorder. If luspatercept were to receive regulatory approval for each of these indications in the United States and Europe, we believe that there is an aggregate sales opportunity for this product in excess of $2 billion.
For sotatercept, we announced in September 2017 that Celgene are developinggranted us the rights to fund, develop, and lead the global commercialization of sotatercept in pulmonary hypertension, including pulmonary arterial hypertension, or PAH. PAH is a rare and chronic, rapidly progressing disorder characterized by the constriction of small pulmonary arteries, resulting in abnormally high blood pressure in the pulmonary arteries. If sotatercept is commercialized to treat PAH and we recognize such revenue, then Celgene will be eligible to receive a royalty in the low 20% range on global net sales. In certain circumstances Celgene may recognize revenue related to the commercialization of sotatercept in PAH, and in this scenario we will be eligible to receive a royalty from Celgene such that the economic position of the parties is equivalent to the scenario in which we recognize such revenue. We expect to initiate a Phase 2 clinical trial for the treatment of patients with chronic kidney disease. Sotatercept hasPAH in the potential to treat several complicationsfirst half of chronic kidney disease including mineral-bone disorder, vascular calcification2018.
For luspatercept and, anemia.outside of pulmonary hypertension, sotatercept, Celgene is responsible for paying 100% of the development costs for all clinical trials for luspatercept and sotatercept, including our ongoing earlier stage clinical trials for these therapeutic candidates.trials. We may receive up to an additional $560a maximum of $545.0 million offor the potential development, regulatory and commercial milestone paymentspayments. If luspatercept and, if these therapeutic candidatesoutside of pulmonary hypertension, sotatercept are commercialized, we willare eligible to receive a royalty on net sales in the low-to-mid 20% range. We will co-promote luspaterceptrange and sotatercept, if approved,we have a co-promotion right in North America, for which our commercialization costs will be entirely funded by Celgene.
We wholly own dalantercept and ACE-083, and we are independently developing these therapeutic candidates. We are currently evaluating dalantercept in a Phase 2 clinical trial for the treatment of patients with renal cell carcinoma.our wholly-owned neuromuscular candidate, ACE-083. ACE-083 is designed for the treatment of focal muscle disorders, such as facioscapulohumeral dystrophy, and we are currently conducting a Phase 2 clinical trials with ACE-083 in patients with facioscapulohumeral dystrophy, or FSHD, as well as in patients with Charcot-Marie-Tooth disease, or CMT. In January 2018, we announced preliminary results for the first two cohorts in part 1 of the Phase 2 clinical trial with ACE-083 in healthy volunteers. In 2015, we reported data from the Phase 1 clinical trial of ACE-083patients with FSHD showing marked increases in the mean total muscle volume of the muscles treated with ACE-083.ACE-083 measured using magnetic resonance imaging, or MRI. We expect to initiate part 2 of the ACE-083 FSHD Phase 2 trial during the second quarter of this year, and we expect to report preliminary results from all dose-escalation cohorts of part 1 in our FSHD and CMT Phase 2 clinical trials with ACE-083 in the second half of this year.
In addition to our mid- to late-stage clinical programs, we initiated a Phase 1 healthy volunteer study in early 2018 with ACE-2494, our wholly-owned systemic muscle agent from our proprietary platform technology, IntelliTrap™, and we expect to report initial results from this healthy volunteer study in the first half of 2019. We are also conducting research within our three focused disease areas—hematologic, neuromuscular and pulmonary—in order to identify new therapeutic candidates to bring forwardadvance into clinical trials. To this end, in 2015 we implemented a new platform technology, IntelliTrap™, that is accelerating our discovery efforts. We have nominated an IntelliTrap™ molecule, ACE-2494, as a candidate for clinical development and will initiate IND-enabling activities in 2016. ACE-2494 is designed to treat systemic muscle disorders.
As of December 31, 20152017 our operations have been funded primarily by $105.1 million in equity investments from venture investors, $219.3$539.7 million from public investors, $64.2$123.7 million in equity investments from our collaboration partners and $233.5$273.7 million in upfront payments, milestones, and net research and development payments from our collaboration partners. We estimate that we have spent approximately $145.4$89.7 million, $68.6 million, and $58.4 million on research and development for the three year period from 2013 through 2015.years ended December 31, 2017, 2016, and 2015, respectively.
Our Goals and Objectives forin the Year 20162018
By building on the milestones achieved in 2015,2017, we intend to advance and expand our pipeline in 20162018 and achieve the following goals and objectives:
Hematology
Luspatercept in Rare Blood Disorders
Myelodysplastic syndromes (MDS):
| ◦ |
| ◦ |
Beta-thalassemia:
| ◦ | Announce BELIEVE Phase 3 clinical trial top-line results |
| ◦ |
| ◦ |
Neuromuscular
| ◦ |
| ◦ |
| ◦ | Initiate part 2 of the Phase 2 clinical trial in CMT by |
| ◦ |
Pulmonary
Sotatercept in Chronic Kidney Disease
| ◦ |
The Acceleron Discovery Platform: Novel Approaches to Potent Biology
Since our founding, we have focused on developing therapeutic candidates that regulate cellular growth and repair. We have targeted a group of approximately 30 secreted proteins, or ligands, that are collectively referred to as the TGF-beta superfamily. These ligands bind to subsets of 12 different receptors on the surface of cells, triggering intra-cellular changes in gene expression that guide cell growth and differentiation. The TGF-beta superfamily ligands and their receptors represent a diverse and underexplored set of drug targets with the potential to yield potent therapeutics for the growth and repair of diseased cells and tissues.
Applying our proprietary discovery and development platform, including our knowledge of the biology of the TGF-beta superfamily and its receptors, we have generated multiple Fc-fusion protein ligand traps, our novel IntelliTrap™ platform technology and a robust pipeline of innovative clinical and preclinical therapeutic candidates targeting key mechanisms underlying cancerserious and rare diseases. Additionally, we are conducting a multi-target antibody discovery collaboration with Adimab LLC, or Adimab, a leading antibody discovery company, under which Adimab is generating human antibodies against undisclosed targets that we select. We expect that thisselect.This collaboration willcould expand our biologics platform and provide us with enhanced access to antibody therapeutic candidates.candidates in the future.
We use our integrated platform of research, development and manufacturing technologies to rapidly and cost-effectively create, test and advance our therapeutic candidates. Our robust clinical and preclinical pipeline is focused on areas of high-unmet medical need, particularlyleadership in the areasunderstanding of cancerTGF-beta biology and rare diseases.protein engineering generates innovative compounds that engage the body's ability to regulate cellular growth and repair.
Our Product Pipeline

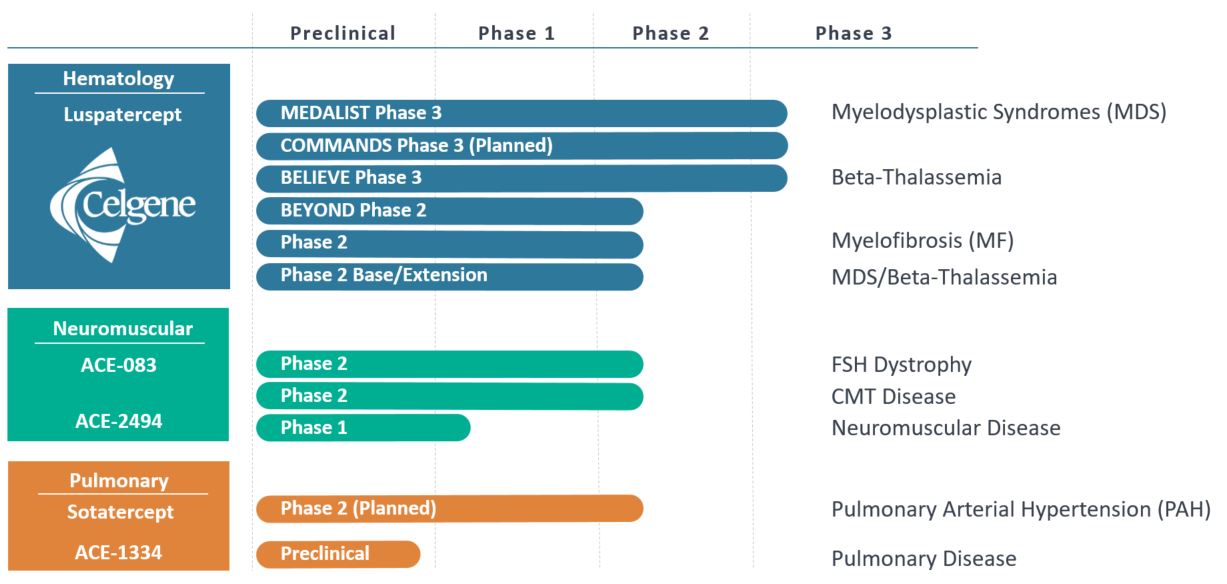
Luspatercept
Luspatercept is an erythroid maturation agent designed to promote red blood cell production through a novel mechanism. We are developing luspatercept, through our collaborations with Celgene, as a treatment for chronic anemia and associated complications in diseases in which erythropoiesis-stimulating agents, or ESAs, are either not approved or are not well-suited to treat the underlying anemia, such as beta-thalassemia and MDS.
With respect to MDS, both our and Celgene's objective is to develop luspatercept as a treatment to increase hemoglobin levels and decrease red blood cell transfusion burden, with patients ultimately becoming transfusion independent.
MDS is a group of heterogeneous hematologic diseases characterized by abnormal proliferation and differentiation of blood precursor cells, including red blood cell precursors, in the bone marrow. This leads to peripheral reductions in red blood cells, often accompanied by decreases in white blood cells and platelets, as well as a risk of disease progression to acute myeloid leukemia. Although MDS patients may have varying forms of the disease, anemia is present in the vast majority of MDS patients at the time of diagnosis. MDS is primarily a disease of the elderly, with 88% of cases diagnosed in individuals 60 years of age or older. Cancer surveillance databases estimate the annual incidence of MDS in the United States at 10,000 toover 15,000 cases and the overall U.S./EU prevalence at approximatelyleast 125,000 patients.
Hematopoietic stem cell transplantation represents the only treatment modality with curative potential, although the relatively high morbidity and mortality of this approach limits its use. Approximately 70%75% of the MDS patients in the U.S. and EU are classified as lower risk and 30%25% are classified as higher risk. High risk patients are typically treated with inhibitors of DNA methyltransferase such as Vidaza® or Dacogen®, or generic versions that are now available in some countries. The patientsMany categorized as low risklower-risk typically receive erythropoiesis stimulating agentsESAs as first-line therapy, though erythropoiesis stimulating agentsESAs are not approved by the FDA or the EMA for the treatment of anemia in MDS patients. Our internal market research estimates that erythropoiesis stimulating agentsESAs generate $500 to $700 million in annual U.S. sales from their use in this disease. AfterTherapeutic options following failure on erythropoiesis stimulating agents,ESAs are limited, and most patients are treated withend up receiving red blood cell transfusion and/or Revlimid®, Vidaza® or Dacogen®.transfusions to manage their anemia. Across the disease, approximately 15% of patients have a specific chromosomal mutation and are treated with Revlimid® (2015 U.S. sales of $152 million for MDS).
The chronic anemia in MDS is primarily due to ineffective erythropoiesis, and a significant number of MDS patients have serum erythropoietin levels substantially above the normal range, indicating that the chronic anemia in these MDS patients is not a consequence of erythropoietin deficiency. The ineffective erythropoiesis of MDS may be caused by excess signaling by members of the TGF-beta superfamily, which signaling inhibits red blood cell maturation. For this reason, we believe that blocking this excess signaling by luspatercept may reverse this inhibition. Approximately 50%More than half of MDS patients are unresponsive to the administration of recombinant erythropoietin and instead require red blood cell transfusions, which can increase the risk of infection and iron-overload related toxicities. Treatment-resistant chronic anemia resulting from ineffective erythropoiesis is a major cause of morbidity in MDS patients.
Celgene is expressed at an elevated level in a mouse model of MDS, leading to elevated levels of an activated transcription factor, P-SMAD2/3, and ineffective erythropoiesis. Treatment with a mouse version of luspatercept, referred to as RAP-536, reduced P-SMAD2/3 levels and caused statistically significant increases in red blood cell count, hemoglobin levels and hematocrit compared to controls. Additionally, RAP-536 reduced the ineffective erythropoiesis as evidenced by the improvement in the ratio of red blood cell precursors to other cells in the bone marrow.
In addition to the Phase 3 clinical trial, we are currently conducting two Phase 2 clinical trials of luspatercept in patients with MDS. The first clinical trial, a 3-month base study, is designed as a two-part trial, with an ascending dose part to evaluate the safety and efficacy in patients with low or intermediate risk MDS per the International Prognostic Scoring System, and an expansion part in which additional patients are enrolled at a selected dose level (3-month base study).level. We have currently completed patient enrollment in all of the dose escalation cohorts and we have completed enrollment of patients inas well as the initialfirst two expansion cohortcohorts of the trial, for a total of 58107 patients.
We have expanded the trial to include two additional cohorts of patients to further evaluate the effects of luspatercept in selected MDS patient populations. Enrollment in expansion cohort 1 is completed and includes patients with serum erythropoietin, or EPO, levels > 500 U/L, or, if ≤ 500 U/L, patients that are non-responsive, refractory, or intolerant to ESAs, or ESAs are contraindicated or unavailable. Enrollment in expansion cohort 2 is completed and includes patients with either (i) RS+ (≥ 15% ring sideroblasts in bone marrow), low transfusion burden (< 4U RBC/8 weeks), no prior ESA treatment, and EPO levels ≤ 200 U/L or (ii) RS- (< 15% ring sideroblasts in bone marrow), low transfusion burden (< 6U RBC/8 weeks), any prior ESA treatment and any EPO level. Patients are treated every 3 weeks by subcutaneous injection for up to 5 doses (titration allowed from 1.0 to 1.75 mg/kg). All patients enrolled in the base study are eligible to enroll in a second Phase 2 trial (extension(long-term extension study) that permits dosing with luspatercept for up to an additional twofive years. These trials are being conducted at sites in Germany.
We believe that preliminary results from the long-term Phase 2 MDS extension study, including the two additional cohorts, are encouraging. We presented these results using a data cut-off date of August 31, 2015,September 8, 2017 at the 57th58th American Society of Hematology (ASH) Annual Meeting and Exposition in December 2015.2017. As of the cut-off date, a total of 3299 patients were treated in the extensionbase study in whichat dose levels of luspatercept was administered subcutaneouslyof at least 0.75 mg/kg, once every 3three weeks. Of these 32All 99 patients 13 had a lowwere evaluable according to the International Working Group, or IWG, Hematologic Improvement Erythroid, or HI-E, response criterion and 67 patients were evaluable for whether they achieved red blood cell, (RBC)or RBC, transfusion independence, or RBC-TI. For patients evaluable for HI-E, the median hemoglobin was 8.4 grams per deciliter (g/dL) (range 6-10 g/dL) and the median RBC transfusion burden (LTB; < 4was 2 units of RBC during each 8 week period (2U RBC/8 weeks) (range 0-18 RBC units). Among patients evaluable for RBC-TI, the median RBC transfusion burden was 4U RBC/8 weeks (range 4-18 RBC units). A total of 63% of patients were ring sideroblast positive, or RS+, 35% were ring sideroblast negative, or RS–, and 19 hadthe remainder were indeterminate as to ring sideroblast status.
Patient response was evaluated using the IWG HI-E response criterion. To achieve IWG HI-E criterion, a low transfusion burden (< 4 U RBC/8 week, Hb <10 g/dL) patient must have a hemoglobin increase of ≥ 1.5 g/dL for ≥ 8 weeks, and a high transfusion burden (HTB; ≥4 units(> 4 U RBC/8 weeks). 59% of patients had been treated previously with erythropoiesis stimulating agents (ESA) and 19% of patients had previously been treated with lenalidomide. With regard to LTB patients, 9 of 13 (69%) LTB patients achieved the International Working Group (IWG) Hematologic Improvement Erythroid (HI-E) response criterion of a hemoglobin increase ≥1.5 g/dL for ≥8 weeks. With regard to HTB patients, 13 of 19 (68%) HTB patients achieved the IWG HI-E criterion of patient must have a reduction of ≥4≥ 4 units RBC over 8 weeks, and 8 of 19 (42%) HTBweeks. The preliminary IWG HI-E response rates for patients treated with luspatercept dose levels of ≥ 0.75 mg/kg were 64% (25/39) for RS+ patients with erythropoietin, or EPO, levels < 200 U/L, 54% (7/13) for RS– patients with EPO levels < 200 U/L, 71% (10/14) for RS+ patients with EPO levels 200-500 U/L, and 50% (4/8) for RS– patients with EPO levels 200-500 U/L.
Patients were also evaluated on the basis of whether they achieved RBC transfusion independence, or RBC-TI, for ≥8≥ 8 weeks. An additional 3The RBC-TI rates for patients treated with luspatercept dose levels of 3 (100%) LTB≥ 0.75 mg/kg were 67% (16/24) for RS+ patients with 2 units/8 weeksEPO levels < 200 U/L, 43% (3/7) for RS– patients with EPO levels < 200 U/L, 44% (4/9) for RS+ patients with EPO levels 200-500 U/L, and 60% (3/5) for RS– patients with EPO levels 200-500 U/L.
With regard to safety, as reported at baseline achieved RBC transfusion independence for ≥8 weeks. A substantialASH in December 2017, the majority of the patients in the Phase 2 trial had a bone marrow cell morphology referred to as ring sideroblasts and given the encouraging response rates in these patients, the Phase 3 trial has been designed to focus on patients with this particular cellular morphology. The most common adverse events observed in this extension study,(AEs) were grade 1 or 2 and luspatercept was generally well-tolerated. There were eight possibly related grade 3, non-serious adverse events (in one patient each): ascites, blast cell count increase, blood bilirubin increase, bone pain, hypertension, mucosal inflammation, platelet count increase and pleural effusion. There were four possibly related serious adverse events (in three patients): general physical health deterioration, muscular weakness and musculoskeletal pain, and myalgia. Adverse events which may be related to luspatercept observed in > 2 patients in these studies were headache, hypertension, fatigue, bone pain, headache, hypotonia,diarrhea, arthralgia, injection site erythema, myalgia, and nausea. Thereperipheral edema.
Based on these data, we concluded that lower risk MDS patients treated with luspatercept continue to demonstrate increases in hemoglobin and decreases in transfusion burden (per IWG HI-E) and a high rate of RBC transfusion independence, and encouraging response rates were no drug-related serious adverse events.observed across all baseline EPO levels. Response rates were similar in patients who received prior ESA compared to those who were ESA naïve. The IWG HI-E response rates were similar in RS+ and RS– when EPO levels were ≤ 500 U/L. These data continue to support the initiation of a new Phase 3 trial in first line, lower-risk MDS patients, referred to as the COMMANDS trial.
The COMMANDS trial is designed as a Phase 3 trial to evaluate luspatercept versus standard-of-care in the first-line treatment setting for lower-risk MDS patients. We expect that the COMMANDS trial will be initiated in the first half of 2018.
Beta-thalassemia
With respect to beta-thalassemia, both our and Celgene's objective is to develop luspatercept as a treatment to increase hemoglobin levels, decrease transfusion burden, decrease iron overload, improve symptoms associated with anemia, and alleviate other disease complications, such as leg ulcers.
The beta-thalassemiasthalassemias comprise a heterogeneous group of disorders arising from defects in the genes that encode the proteins that comprise hemoglobin. Hemoglobin is a four-subunit protein complex formed of two alpha-subunits and two beta-subunits, each with an iron-containing heme group that binds to and carries oxygen molecules within red blood cells. There are two main classifications of thalassemia, alpha-thalassemia and beta-thalassemia, depending on whether the genetic defect lies in the gene encoding the alpha-subunit or the beta-subunit.beta-subunit, respectively. Beta-thalassemia is particularly prevalent throughout the Mediterranean region, Middle East, and Southeast Asia, and, due to migration and immigration, is nowincreasingly a global disease. The Thalassaemia International Federation estimates that there are approximately 300,000 patients worldwide with beta-thalassemia, approximately 20,000 of which are in the United States and Europe, who are dependent on frequent blood transfusions. We estimate that there are at least as many beta-thalassemia patients in the same regions who are not transfusion dependent and not included in these estimates. Many of these patients have hemoglobin levels that are approximately half that of normal individuals and experience significant complications of the disease. Beta-thalassemia is treated primarily by red blood cell transfusions that, over time, cause a toxic accumulation of iron in the body. A central challenge for managing patients with beta-thalassemia is to restore the red blood cell levels while avoiding iron overload. Iron chelation therapy alone costs between $40,000 and $60,000$80,000 per year in countries such as the United States and Italycertain countries in Europe and yet does not treat the underlying anemia. The course of the disease depends largely on whether patients are maintained on an adequate transfusion and iron chelation regimen. Poor compliance with transfusion and/or iron chelation is associated with a poor prognosis and shortened survival. However, even with the standard of care, patients are at risk of infection from transfusions as well as toxicities related to iron chelation therapy.
No drug is approved to treat the anemia of beta-thalassemia. Hematopoietic stem cell transplantation is viewed as the only curative approach for beta-thalassemia, although this option is limited by the availability of appropriate donors and by risks, including death, associated with the bone marrow transplant procedure. Consequently this treatment is used only in the most severely affected patients.
In addition to the Phase 3 clinical trial, we are currently conducting two Phase 2 clinical trials of luspatercept in patients with beta-thalassemia. The first clinical trial, a 3-month base study, is designed as a two-part trial, with an ascending dose part to evaluate the safety and efficacy of luspatercept in patients with beta-thalassemia, and an expansion part in which additional patients are enrolled at a selected dose level (3-month base study).level. We have currently completed enrollment and treatment of all of the dose escalation cohorts as well as the expansion cohort of the base trial. Patients enrolled in the initial 3-monthbase trial are eligible to enroll in a second Phase 2 trial (extension study) that permits dosing with luspatercept for up to an additional twofive years. This trial is currently being conducted at sites in Italy and Greece.
We believe that the preliminary results from the Phase 2 clinical trials are encouraging. We presented these results, using a data cut-off date of September 25, 2015,April 13, 2017, at the 57th ASH Annual Meeting and Exposition22nd Congress of the EHA in December 2015.June 2017. As of the cut-off date, a total of 6463 patients werehad been treated in thewith luspatercept dose escalation and expansion cohorts of this study, in which luspaterceptlevels ≥ 0.6 mg/kg. Luspatercept was administered subcutaneously, once every
3 weeks. A total of 59 patients were evaluable for efficacy (5 patients were ongoing with <12 weeks treatment). Of these 5963 patients, 32 were transfusion dependent and 31 were non-transfusion dependent and 28 were transfusion dependent. Specifically, 22 of 28 (79%)For the transfusion dependent patients treated with luspatercept dose levels ≥ 0.6 mg/kg, 69% of patients had a ≥20%≥ 33% reduction in transfusion burden 21 of 28 (75%) had a ≥33% reduction,over any 12-week period compared to baseline.
For the non-transfusion dependent patients treated with luspatercept dose levels ≥ 0.6 mg/kg, 71%, respectively, achieved ≥ 1.0 g/dL increases, and 16 of 28 (57%) had a ≥50% reduction52% achieved ≥ 1.5 g/dL increases, in mean hemoglobin over aany 12-week period.period compared to baseline.
A trend of reduction in liver iron concentration, or LIC, was observed in the majority of non-transfusion dependent patients with or without iron chelation therapy, and in the majority of transfusion dependent patients receiving iron chelation therapy. Improvement in patient-reported quality of life in non-transfusion dependent patients correlated with increaseincreases in hemoglobin. Rapid healing and partial healing of leg ulcers, a serious complication of beta-thalassemia, was observed in some patients. To date, we have received no reports of related serious adverse events and we have received reports of related grade 3 patients, with 2 additional patients experiencing partial healing.adverse events consisting of bone pain, asthenia, and headache. The most common related adverse events which may be related to luspatercept observed in ≥ 10% of patients were bone pain, myalgia, headache, arthralgia, musculoskeletal pain, asthenia, injection site pain, back pain and pain in jaw. There were no drug-related serious adverse events. 6 of 59 (10%) patients discontinued early with an associated adverse event: bone pain (2 patients) and arthralgia, asthenia, cerebrovascular accident and headache (1 patient each).injection site pain.
Myelofibrosis is an acquired disease of the bone marrow that results in replacement of the bone marrow with fibrotic tissue leading to bone marrow failure and inability to make new blood cells, including red blood cells, which leads to anemia. Epidemiological databases suggest that there are approximately 30,000 myelofibrosis patients in the United States and Europe. Approximately 30% of myelofibrosis patients present primarily with anemia when diagnosed and nearly all patients will develop anemia with progression of the disease. There is no approved drug therapy to treat chronic anemia in myelofibrosis. Our and Celgene's objective is to develop luspatercept as a treatment for the chronic anemia in myelofibrosis patients, and Celgene has initiated a Phase 2 clinical trial in patients with myelofibrosis and enrollment is ongoing.
Sotatercept
Sotatercept is an activin receptor type IIA fusion protein that acts as a ligand trap for members in the TGF-beta protein superfamily involved in the remodeling of a variety of different tissues, including the vasculature and fibrotic tissue. In Phase 1 and Phase 2 clinical trials involving approximately 400 patients, sotatercept has been shown in humans to promote the formation of red blood cells, increase bone mineral density and reduce calcified blockages in the vasculature (vascular calcification). We have recently discovered that sotatercept has a potent effect in improving cardiovascular health in mouse models of a disease termed pulmonary arterial hypertension, or PAH.
Pulmonary hypertension is a group of diseases characterized by elevated blood pressure in the pulmonary circulation resulting from a variety of causes. PAH is a type of pulmonary hypertension. It is a rare, chronic, rapidly progressing disorder characterized by the constriction of small pulmonary arteries, resulting in abnormally high blood pressure in these vessels. Patients typically present with exercise fatigue and shortness of breath. Progressive obstruction and constriction of the pulmonary vasculature leads to elevated blood pressure in the pulmonary circulation and strain on the right side of the heart. In later stages of disease, right heart failure may develop, and heart failure is the major cause of death in PAH patients. The major classes of drugs used to treat PAH are: the phosphodiesterase 5 inhibitors (PDE5i), the endothelin receptor antagonists (ERAs), the soluble guanylate cyclase stimulators (sGCs) and the prostacyclins. Despite the availability of therapeutic options, the disease progresses rapidly and the five-year survival rate for patients is approximately 57%. All classes of currently approved therapies are designed to promote the dilation of blood vessels, a mechanism referred to as vasodilation. As evident from the high mortality rate, therapies that promote vasodilation improve many aspects of the disease, but do not fundamentally modify or halt the progression of the disease. Accordingly, there is significant unmet need for therapies that address more fundamental aspects of the disease.
Heritable forms of PAH are known, and the underlying genetic defects have been identified. In most cases, the underlying mutations map to a gene that encodes the BMP receptor type II (BMPRII) or genes encoding other proteins that participate in the BMPRII signaling pathway. BMPRII is a key receptor for ligands referred to as the BMPs, and these are part of the TGF-beta superfamily. The BMP-BMPRII signaling pathway activates transcription factors called Smad1/5/8 that coordinate changes in the expression of certain genes. A deficiency in BMP-BMPRII signaling, and the attendant activation of Smad1/5/8, is thought to lead to increased proliferation of cell types that line the vessels, including endothelial cells, smooth muscle cells and fibroblasts. This increased proliferation leads to an overgrowth of smooth muscle and fibrotic tissue around the small arterioles, leading to the formation of muscularized vessels and so-called plexiform lesions. The vessels become constricted and obstructed. Plexiform lesions are arterioles that have become almost completely obstructed by an over-proliferation of
endothelial cells, smooth muscle cells and fibrotic tissue. Although the association between BMP-BMPRII signaling and PAH was first identified through the study of patients with heritable forms of the disease, it is now understood that BMP-BMPRII signaling is deficient in patients with non-heritable forms of the disease, including idiopathic and associated forms of PAH. For these reasons, it is expected that a therapeutic agent that improves the signaling deficits in the BMP-BMPRII pathway, activating Smad1/5/8, could rectify key defective molecular signaling events causing the disease, with the potential to modify the course of disease.
It has been reported that a group of proteins targeted by sotatercept, the activins, are present at increased levels in PAH patients. Recently, we and others have found that activins can suppress signaling through the BMP-BMPRII pathway, as measured by assessing Smad1/5/8 activation. We further found that sotatercept, by inhibiting activins, can stimulate the BMP-BMPRII signaling pathway in isolated cells. We evaluated sotatercept for its effects on the establishment of PAH in two mouse models of the disease, and we found that sotatercept had a more profound benefit than representatives of any of the classes of standard-of-care therapies. As a result, we believe that sotatercept has the potential to correct the BMP-BMPRII signaling deficit that is fundamental to disease in PAH patients and thereby modify the course of disease. Further, because sotatercept works by a distinct mechanism from the available vasodilator agents, we believe that sotatercept can be used in combination with these other agents.
The worldwide PAH patient population is expanding, with approximately 30,000 patients in the United States and 40,000 patients in the European Union. PAH represents one of the largest specialty pharmaceutical markets with more than $4 billion in sales in the United States in 2016.
In September, 2017, we amended our agreement with Celgene to secure the right to develop and commercialize sotatercept in patients with pulmonary hypertension, and we expect to initiate a Phase 2 clinical trial with sotatercept in patients with PAH during the first half of 2018. In addition to other patents and patent applications related to sotatercept, we have filed patent applications relating to the use of sotatercept in the treatment of pulmonary hypertension. The expected expiration date for these method of treatment patents is 2037, exclusive of possible patent term extensions.
Celgene has the rights to continue to develop and commercialize sotatercept outside the pulmonary hypertension field. Sotatercept is being studied through investigator-initiated clinical trials in multiple myeloma, Diamond-Blackfan anemia and myelofibrosis. Investigators at the MD Anderson Cancer Center are conducting a trial to determine the safety and efficacy of sotatercept in patients with myeloproliferative neoplasm-associated myelofibrosis and anemia.
ACE-083
Our fourththird clinical stage therapeutic candidate, ACE-083, is designed to promote muscle growth and function in specific, targeted muscles. In 2014, we initiatedACE-083 uses the myostatin+ approach to promote muscle growth, meaning that ACE-083 inhibits myostatin, a Phase 1 clinical trial withwell-characterized regulator of muscle size, as well as other factors that suppress muscle growth. Through the inhibition of multiple suppressors of muscle growth, ACE-083 in healthy volunteers. ACE-083 has been well tolerated and no serious adverse events have been reported. Initial data from the Phase 1 trial showed that, at the highest dose level tested, ACE-083 generatedpromotes a meansubstantially greater muscle mass increase in muscle volume of approximately 14.5% in the treated muscle.than selective myostatin antagonists. We have completed enrollment for the ACE-083 Phase 1 clinical trial, and we expect to initiate aare currently conducting Phase 2 clinical trialtrials with ACE-083 in patients with facioscapulohumeral dystrophy, or FSHD, as well as in the second half of 2016.patients with Charcot-Marie-Tooth disease, or CMT.
FSHD
FSHD is a severe, disabling, and painful skeletal muscle disease that presents with muscle-by-muscle progression. Muscle weakness can be heterogeneous, but is highly focal and primarily asymmetric. Typical onset occurs between the ages 20 and 40. Most FSHD patients live a normal lifespan, but many will suffer from disability, pain, and depression. Current treatment is largely limited to orthotic or surgical intervention to provide or maintain functionality. FSHD is one of the most prevalent forms of muscular dystrophy affecting roughly 19,00020,000 patients in the United States.
We recently announced preliminary results for the first two cohorts in part 1 of the Phase 2 clinical trial with ACE-083 in patients with facioscapulohumeral dystrophy, or FSHD. These preliminary results included data from 23 patients evaluable for magnetic resonance imaging, or MRI, among two different cohorts consisting of 11 patients with tibialis anterior weakness and 12 patients with biceps brachii weakness. Each patient received ACE-083 (150 mg or 200 mg) as a unilateral intramuscular injection once every three weeks for 12 weeks. Total muscle volume changes were measured by MRI relative to baseline at 3 weeks after the last injection of ACE-083. Based on overlap in dosing on a milligram per gram muscle analysis, dose cohorts were pooled for the analyses of each muscle. The tibialis anterior part 1 cohorts consisting of 11 patients receiving either 150 mg or 200 mg of ACE-083 generated a mean total muscle volume increase of 12.6% and a mean decrease, or improvement, in fat fraction of 5.3%. The biceps brachii part 1 cohorts consisting of 12 patients receiving either 150 mg or 200 mg of ACE-083 generated a mean total muscle volume increase of 13.2% and a mean decrease, or improvement, of fat fraction of 0.6%. In the
biceps brachii cohorts, the majority of patients had less intramuscular fat at baseline relative to the patients in the tibialis anterior cohorts. Patients with higher fat fraction in the biceps brachii cohorts at baseline demonstrated larger decreases in fat fraction with treatment.
ACE-083 has been well tolerated in the FSHD Phase 2 clinical trial and no serious adverse events were reported. The most common adverse events were injection site related and grades 1-2. One patient experienced a related grade 3 non-serious adverse event of lower leg intramuscular swelling. This patient fully recovered and was discontinued from the study. We expect to initiate part 2 of the ACE-083 FSHD Phase 2 trial during the second quarter of this year.
CMT
CMT is one of the most common inherited neurologic diseases estimated to affect more than 125,000 people in the United States. The primary clinical manifestations of CMT include muscle weakness in the lower legs and arms. The lower leg muscle weakness can result in foot drop and a high-stepped gait leading to frequent tripping or falls. The disease is typically diagnosed by the presence of a characteristic pattern of muscle weakness and other clinical signs and symptoms, as well as through genetic testing.
We are currently conducting a Phase 2 clinical trial with ACE-083 in patients with CMT and expect to report preliminary results from part 1 of all dose-escalation cohorts in the second half of this year.
ACE-2494
In addition to our mid- to late-stage clinical programs, we initiated a Phase 1 healthy volunteer study in early 2018 with ACE-2494, our wholly-owned systemic muscle agent from our proprietary platform technology, IntelliTrap™, and we expect to report initial results from this healthy volunteer study in the first half of 2019.
Preclinical Programs
In addition to our clinical development activities, we are expanding our research capabilities in order to increase the rate at which our highly productive research group can identify and advance new, internally discovered, therapeutic candidates for clinical development. Our discovery efforts are primarily focused on identifying new protein therapeutic candidates from our IntelliTrap™fusion protein platform, which includes the IntelliTrap™ platform, and identifying novel antibodies. We have selected our first IntelliTrap™ therapeutic candidate, ACE-2494, for pre-clinical evaluation and advancement to clinical trials by the end of 2016. We are alsocurrently evaluating ACE-3891 as a candidate therapeutic for the treatment of muscle disease, ACE-1332,ACE-1334, a selective TGF-beta antagonist, for treatment of disorders with a fibrotic component, and ACE-2798, ACE-2536 and ACE-2395additional molecules from our IntelliTrap™ platform for undisclosed therapeutic areas.
Our Strategic Partnerships
Collaborations with corporate partners have provided us with significant funding and access to our partners' scientific, development, regulatory and commercial capabilities. We have received more than $297.7$397.5 million from our collaborations with Celgene and our prior collaborations with Alkermes plc (Alkermes) and our terminated collaboration with Shire AG (Shire).
Celgene
On February 20, 2008 we entered into an agreement with Celgene relating to sotatercept, which we refer to as the Original Sotatercept Agreement. We amended the Original Sotatercept Agreement with Celgene Corporation, underon August 2, 2011, which we grantedrefer to Celgene worldwide rightsas the Amended Sotatercept Agreement. We further amended and restated the Amended Sotatercept Agreement in its entirety on September 18, 2017, and refer to sotatercept.the amended and restated agreement as the Restated Sotatercept Agreement. On August 2, 2011 we entered into a second agreement with Celgene for luspatercept, which we refer to as the Luspatercept Agreement.
Restated Sotatercept Agreement. The Restated Sotatercept Agreement under which we grantedprovides Celgene with an exclusive license to Celgenesotatercept outside of the field of pulmonary hypertension, referred to as the PH field, and provides us with the worldwide rights to luspaterceptdevelop and also amended certain terms ofcommercialize sotatercept in the Sotatercept Agreement. These agreements provide Celgene exclusive licenses for these therapeutic candidates in all indications, as well as exclusive rights to obtain a license to certain future compounds.
In connection with the Restated Sotatercept Agreement, Celgene agreed not to develop or commercialize in the field of pulmonary hypertension any compound developed under the Restated Sotatercept Agreement or the Luspatercept Agreement, and we agreed not to develop or commercialize any compound developed under the Restated Sotatercept Agreement or the Luspatercept Agreement in any field outside of pulmonary hypertension. We retained responsibilityhave the right to license, transfer or sell our rights to develop and commercialize sotatercept in pulmonary hypertension, subject to Celgene’s right of first negotiation.
We are responsible for research,100% of the costs related to our development throughand commercialization of sotatercept in the endPH field. If sotatercept is commercialized to treat pulmonary hypertension and we recognize such revenue, then Celgene will be eligible
to receive a royalty in the low 20% range on global net sales. In certain circumstances Celgene may recognize revenue related to the commercialization of sotatercept in pulmonary hypertension, and in this scenario, we will be eligible to receive a royalty from Celgene such that the economic position of the parties is equivalent to the scenario in which we recognize such revenue. With respect to the development and commercialization of sotatercept outside of the PH field or the development and commercialization of any other compound under the Restated Sotatercept Agreement, the terms of the Amended Sotatercept Agreement remained unchanged.
Since January 1, 2013, Celgene has been responsible for paying 100% of worldwide development costs for the sotatercept program outside of the PH field and Celgene will be responsible for all commercialization costs worldwide. We will be eligible to receive tiered royalty payments in the low-to-mid 20% range on net sales of sotatercept outside of the PH field. We are obligated to co-promote sotatercept outside of the PH field and future products in all fields, in each case if approved, in North America, and Celgene will pay all costs related thereto. Outside the PH field, Celgene will be responsible for any sotatercept Phase 2a3 clinical trials, as well as manufacturing the clinical supplies for these trials. These activities are substantially complete. Celgene is conducting the currentany additional Phase 2 clinical trials, and is responsible for beta-thalassemia, MDSmanufacturing or overseeing the manufacture of Phase 3 and chronic kidney diseasecommercial supplies.
Pursuant to the Restated Sotatercept Agreement, Celgene will provide us with certain quantities of Celgene’s existing clinical supply of sotatercept for development in the PH field at no cost to us. For clinical or commercial supply of sotatercept in excess of that which is agreed to under the Restated Sotatercept Agreement, Celgene can elect to provide us with such clinical and commercial supply of sotatercept at a negotiated price or provide a tech transfer to us to enable us to manufacture on our own behalf. The conduct of the collaboration is managed by a Joint Development Committee and Joint Commercialization Committee. In the event of a deadlock of a committee, we shall determine the resolution of issues specifically related to the PH field (other than pricing which shall be determined by consensus), and Celgene shall determine the resolution of all other issues. The Joint Commercialization Committee will oversee commercialization of sotatercept and sotatercept pricing will be responsibledetermined by mutual agreement of us and Celgene in the Joint Commercialization Committee.
The Restated Sotatercept Agreement will expire on a country-by-country basis on the occurrence of the latest to occur of the following: (1) the expiration of the royalty term with respect to all license products outside the PH field in such country, (2) the expiration of the royalty term with respect to all sotatercept licensed products in the PH field in such country, and (3) the exercise or forfeiture by Celgene of its option with regard to each option compound. In the PH field, the royalty term for any futureeach licensed product in each country is the period commencing with first commercial sale of the applicable licensed product in the applicable country and ending on the latest of expiration of specified patent coverage or a specified period of years. Outside the PH field, the royalty term for each licensed product in each country outside North America is the period commencing with first commercial sale of the applicable licensed product in the applicable country and ending on the latest of expiration of specified patent coverage or a specified period of years, and the royalty term for each licensed product in North America is the period commencing with the first commercial sale in North America and ending, on a licensed product and country-by-country basis on the date which commercialization of such licensed product has ceased. The term for each option compound runs for a specified period of years unless Celgene exercises its option, in which case the compound becomes a licensed product, or forfeits its option by failing to make certain payments following the achievement of certain milestones in early clinical trialsdevelopment of the option compound.
The Restated Sotatercept Agreement is terminable by either party upon a breach that is uncured and continuing or by Celgene for convenience on a country-by-country or product-by-product basis, or in its entirety. Celgene may also terminate the Restated Sotatercept Agreement, in its entirety or on a product-by-product basis, for failure of a product to meet a development or clinical trial endpoint. Termination for cause by us or termination by Celgene for convenience or failure to meet an endpoint will have the effect of terminating the applicable license to Celgene and the rights granted to us with respect to the development of sotatercept as well asin the PH field shall become irrevocable. Termination for all future manufacturecause by either party shall result in reducing the remaining royalties due to the breaching party by a certain percentage. Upon termination by Celgene for convenience or for failure to meet an endpoint, we and Celgene will enter into a termination agreement pursuant to which, among other things, Celgene will continue to be eligible to receive a royalty in the low 20% range on global net sales of sotatercept. sotatercept in the PH field.
We are eligible to receive future development, regulatory and commercial milestones of up to $360.0 million for the sotatercept program outside of the PH field and up to an additional $348.0 million for each of the three discovery stage programs. None of the three discovery stage programs has advanced to the stage to achieve payment of a milestone, nor do we expect any such milestone payments in the near future. We were not required to make any upfront payments to Celgene upon execution of the Restated Sotatercept Agreement, and we will not be required to make any milestone payments to Celgene in connection with our development and commercialization of sotatercept in the PH field.
Luspatercept Agreement. Under the terms of theThe Luspatercept Agreement provides Celgene with an exclusive license to luspatercept in all indications and we and Celgene are collaborating in the development and commercialization of luspatercept. We also granted Celgene an option to license products for which Acceleron files an investigational new drug application for the treatment of anemia. Celgene paid $25.0 million to us upon execution of the Luspatercept Agreement in August 2011 and, as of
December 31, 2015,2017, we have received $60.3$99.4 million in research and development funding and milestone payments for the luspatercept program.
Under this agreement, we retained responsibility for research, development through the end of Phase 1 and the two ongoing Phase 2 clinical trials in MDS and beta-thalassemia, as well as manufacturing the clinical supplies for these studies. Celgene will conduct subsequent Phase 2 and Phase 3 clinical trials. Acceleron will manufacture luspatercept for all Phase 1 and Phase 2 clinical trials, and Celgene will have responsibility for the manufacture of luspatercept for Phase 3 clinical trials and commercial supplies. We are eligible to receive future development, regulatory and commercial milestones of up to $200.0 million for the luspatercept program.
In November 2013, the Company agreed to conduct additional development activities for luspatercept including clinical and mid-single digit royaltiesnon-clinical services, which are reimbursed under the same terms and rates of the existing Luspatercept Agreement. Please refer to Note 10 to the financial statements in this Annual Report on product salesForm 10-K for each drug developed and commercialized by Alkermes using this technology. To our knowledge, Alkermes is not currently pursuing technology licensed under this collaboration. Rights pertaining to a lead program, referred to ACE-771, were returnedthe revenue recognition accounting, including changes in estimates, pursuant to the terms of the agreement, and we are not pursuing development of ACE-771 at this point in time.
Competition
The development and commercialization of new drugs is highly competitive. We and our collaborators will face competition with respect to all therapeutic candidates we may develop or commercialize in the future from pharmaceutical and biotechnology companies worldwide. Many of the entities developing and marketing potentially competing products have significantly greater financial resources and expertise than we do in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing. Our commercial opportunity will be reduced or eliminated if our competitors develop and commercialize products that are more effective, have fewer side effects, are more convenient or are less expensive than any products that we may develop.
If our clinical stage therapeutic candidates are approved, they will compete with currently marketed drugs and therapies used for treatment of the following indications, and potentially with drug candidates currently in development for the same indications:
MDS
If either luspatercept or sotatercept is approved for the treatment of patients with MDS, it would compete with the following:
Recombinant erythropoietin and other erythropoiesis stimulating agents. Although these agents are not approved to treat anemia in MDS in the United States, current practice guidelines include the use of erythropoiesis stimulating agents and granulocyte colony stimulating factor agents (G-CSF) to treat patients with MDS. Additionally, Amgen is currently studying erythropoiesis stimulating agent, Aranesp® andEprex® from Janssen Pharmaceuticals is studying erythropoiesis stimulating agent Eprex® in Phase 3 clinical trialswas recently approved in Europe for treatment ofto treat symptomatic anemia in certain patients with lower risk MDS.
| • | Red blood cell transfusions, which are used to treat anemia in patients with MDS, and iron chelation therapy, such as Novartis's oral iron chelating agents Exjade® and JadenuTM, which are used to treat iron overload in patients with MDS. |
Immunomodulators, including Celgene's approved product, Revlimid® (lenalidomide), for the treatment of anemia of certain MDS patients.
Other therapies in development, including: an oral form of the hypomethylating agent azacitidine, known as CC-486, being developed by Celgene to treat patients with transfusion dependent anemia and thrombocytopenia due to lower risk MDS, which is currently in Phase 3 clinical trials in the United States and Europe; an anti-cancer therapy being developed by Onconova to treat patients with MDS; a telemorase inhibitor, imetelstat, being studied by Geron and Janssen in a Phase 2/3 study in lower risk MDS patients; and a CD95 ligand inhibitor, APG101, being studied by Apogenix, incompleted a Phase 1 study in transfusion dependent, lower risk MDS patients.
Beta-thalassemia
If luspatercept is approved for the treatment of patients with beta-thalassemia, it would compete with:
| • | Red blood cell transfusions, which are used to treat anemia in patients with beta-thalessemia, and iron chelation therapy, such as Novartis's oral iron chelating agents Exjade® and JadenuTM, which are used to treat iron overload in patients with beta-thalessemia. |
Fetal hemoglobin stimulating agents, such as hydroxyurea, which are primarily used to treat patients with anemia from sickle cell disease, are sometimes used to treat patients with beta-thalassemia.
Hematopoietic stem cell transplant treatment is given to a small percentage of patients with beta-thalassemia, since it requires a sufficiently well-matched source of donor cells. Certain academic centers around the world are seeking to develop improvements to this approach.approach
Bluebird bio’s gene therapy program, LentiGlobin®, is currently in Phase 3 clinical trials in patients with transfusion dependent beta-thalassemia.
Other therapies in development, including gene therapy and genome editing are being developed by several different groups, including bluebird bio, Inc., Memorial Sloan Kettering Cancer Center, GlaxoSmithKline plc, and Sangamo BioSciences Inc. in collaboration with Biogen Idec.Bioverativ, and CRISPR Therapeutics in collaboration with Vertex.
If sotaterceptluspatercept is approved to treat certain aspects of chronic kidney disease-mineral and bone disorder (CKD-MBD), other drugs approved for the treatment of patients with myelofibrosis, it would compete with:
Insight Biotechnology's JAK2 kinase inhibitor, Jakafi®, for the treatment of intermediate or high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis and post-essential thrombocythemia myelofibrosis.
Recombinant erythropoietin, other erythropoiesis stimulating agents and immunomodulatory drugs. Although these patients could be considered competitive. CKD-MBD is a systemic disorder encompassing mineral, bone, and calcific cardiovascular abnormalities that develop as a complication of CKD and that contribute to CKD-related cardiovascular disease and high mortality rates. Therapiesagents are not approved to treat aspects of CKD-MBD include phosphate binders to address hyperphosphatemia, including Sanofi’s Renagel® and Renvela® , Shire’s Fosrenol®,and Keryx’s newly approved product Auryxia®. Amgen’s oral calcimimetic product Sensipar®/Mimpara® is approved to treat secondary hyperparathyroidismanemia in CKD patients on dialysis. Amgen is also studying etelcalcitide (AMG-416), an intravenously administered calcium sensing receptor agonist, and has filed for marketing approvalmyelofibrosis in the United States, and Europecurrent practice guidelines include the use of these agents to treat secondary hyperparathyroidism in chronic kidney disease patients on dialysis. Additionally, if sotatercept is studied and approvedanemia in patients with pre-dialysis kidney disease, other drugs that potentially slow progression of kidney disease could be considered competitors. Currently, angiotensin converting enzyme (ACE) inhibitorsmyelofibrosis.
A telemorase inhibitor, imetelstat, being studied by Geron and angiotensin receptor blockers (ARB) therapies are standard of careJanssen in a Phase 2 study to treat intermediate-2 or high-risk in patients with chronic kidney disease and have been shown to lower blood pressure, reduce proteinuria, and slow the progression of kidney disease. There are also a number of drugs in late-stage clinical trials to slow progression of diabetic nephropathy, a form of kidney disease in diabetic patients. Janssen Pharmaceuticals. NephroGenex, Abbvie, and Bayer have programs in ongoing Phase 3 clinical trials, and Mitsubishi Pharma, Lilly, and Vascular Pharmaceuticals, have programs in ongoing or completed Phase 2 clinical trials, to slow progression of diabetic nephropathy.
Therapies for Treating Muscle Loss
We are currently studying ACE-083 in a Phase 12 clinical trial in healthy volunteers and plan to develop ACE-083 for the treatment of neuromuscular disorders and other diseases characterized bypatients with FSHD, as well as a loss of muscle function including facioscapulohumeral muscular dystrophy (FSHD).Phase 2 clinical trial with ACE-083 in patients with CMT. One potential competitor of ACE-083 in FSHD is Resolaris® (ATYR1940), which is an investigational protein inbeing developed by aTyr Pharma, which completed Phase 1b/2 studies to treat adult patients with FSHDFSHD. A potential competitor of ACE-083 in CMT is PXT3003, which is an investigational pleotherapy being developed by aTyr Pharma.Pharnext to treat CMT1A and is currently in a Phase 3 clinical trial.
We are also aware of approaches to treat other muscle loss diseases that are in clinical trials. Novartis is developing bimagrumab (BYM338), a monoclonal antibody targeting the activin receptor type IIB (ActRIIB), in various Phase 2 clinical trials to treat pathological muscle loss and weaknessweakness. Regeneron is developing two drugs in combination, REGN2477, an Activin A antibody, and Trevogrumab, a myostatin antibody, in a Phase a 2/31 clinical trial to treat patients with sporadic inclusion body myositis (sIBM). Regeneronmuscle wasting diseases. Roche is developingstudying RG-6206 a myostatin monoclonal antibody, REGN1033 (SAR391786), which has completedfusion protein, in a Phase 1/2 clinical trial for treatment of sarcopenia.Duchenne muscular dystrophy (DMD). Pfizer is conducting a Phase 2 clinical study for PF-06252616, a myostatin antibody, in patients with Duchenne muscular dystrophy (DMD).DMD. Nationwide Children's Hospital, in collaboration with The Myositis Association, Parent Project Muscular Dystrophy and Milo Biotechnology, are conducting ahave completed Phase 1 clinical trial of a gene therapy delivery of follistatin (FS344) to muscle in patients with Becker muscular dystrophy (BMD) and sporadic inclusion body myositis (sIBM). and have a second clinical trial in Duchenne muscular dystrophy underway. There are also other programs in early stage development that focus on gene editing and next generation antisense that could compete with our therapies for treating muscle loss.
Pulmonary Arterial Hypertension
We expect to initiate a Phase 2 clinical trial with sotatercept for the treatment of patients with pulmonary arterial hypertension, or PAH, in the first half of 2018. PAH is a rare and chronic, rapidly progressing disorder characterized by the constriction of small pulmonary arteries, resulting in abnormally high blood pressure in the pulmonary arteries.
If sotatercept is approved for the treatment of patients with PAH, it would compete with four distinct classes of vasodilator therapies:
Phosphodiesterase type 5 (PDE5) inhibitors Adcira® (tadalafil, Eli Lilly) and Revatio® (sildenafil, Pfizer), which are orally available small molecules. PDE5 inhibitors prevent degradation of intracellular cGMP released in response to nitric oxide (NO), resulting in relaxation of pulmonary vessels.
Soluble guanylate cylase (sGC) stimulator Adempas® (riociguat, Bayer), which targets different molecules in the same biological pathway as PDE5 inhibitors and is also an oral small molecule.
Endothelin receptor antagonists (ERA) Letairis® (ambrisentan, Gilead), Opsumit® (macitentan, Actelion/J&J), and Tracleer® (bosentan, Actelion/J&J)), which are all orally available small molecules. ERAs block activity of endothelin-1, which causes constriction of pulmonary vessels.
| • | Prostacyclin analogues relax pulmonary vessels, prevent platelet aggregation, and inhibit smooth muscle proliferation. They are the most potent therapies for treating PAH and are available orally (Orenitram® (trepostinil, United Therapeutics), Uptravi® (selexipag, Actelion/J&J)); for inhalation (Tyvaso® (trepostinil, United Therapeutics), Ventavis® (iloprost, Actelion/J&J)); and for infusion (Remodulin® (trepostinil, United Therapeutics), Veletri® (epoprostenol, Actelion/J&J)). |
The key competitive factors affecting the success of any approved product will be its efficacy, safety profile, price, method of administration and level of promotional activity.
Commercialization
We retain co-promotion rights in North America with our collaboration partner, Celgene, for both sotatercept outside the PH field and luspatercept, in North America, and under the terms of our agreements with Celgene, our commercialization costs will be entirely funded by Celgene. We also currently retain worldwide commercialization rights for our oncology therapeutic candidate, dalantercept,ACE-083, sotatercept in the PH field, and our muscle therapeutic candidate, ACE-083.ACE-2494. We intend to build hematology, neuromuscular and neuromuscularpulmonary disorder focused, specialty sales forces in North America and, possibly, other markets to effectively support the commercialization of these and future products. We believe that a specialty sales force will be sufficient to target key prescribing physicians in these areas. We currently do not have any sales or marketing capabilities or experience. We will establish the required capabilities within an appropriate time frame ahead of any product approval and commercialization to support a product launch. If we are not able to establish sales and marketing capabilities or are not successful in commercializing our future products, either on our own or through collaborations with Celgene, any future product revenue will be materially adversely affected.
Intellectual Property
Our commercial success depends in part on our ability to obtain and maintain proprietary protection for our therapeutic candidates, novel biological discoveries, screening and drug development technology, to operate without infringing on the proprietary rights of others and to prevent others from infringing our proprietary rights. Our policy is to seek to protect our proprietary position by, among other methods, filing U.S. and foreign patent applications related to our proprietary technology, inventions and improvements that are important to the development and implementation of our business. We also rely on trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our proprietary position. Additionally, we expect to benefit from a variety of statutory frameworks in the United States, Europe and other countries that relate to the regulation of biosimilar molecules and orphan drug status. These statutory frameworks provide periods of non-patent-based exclusivity for qualifying molecules. See "Government Regulations"Regulation".
Our patenting strategy is focused on our therapeutic candidates. We seek composition-of-matter and method-of-treatment patents for each such protein in key therapeutic areas. We also seek patent protection with respect to companion diagnostic methods and compositions and treatments for targeted patient populations. We have sought patent protection alone or jointly with our collaborators, as dictated by our collaboration agreements.
Our patent estate, on a worldwide basis, includes approximately 160635 issued patents and approximately 390492 pending patent applications, with pending and issued claims relating to all of our current clinical stage therapeutic candidates,
sotatercept, luspatercept, dalanterceptACE-083 and ACE-083.ACE-2494. These figures include in-licensed patents and patent applications to which we hold exclusive commercial rights.
Individual patents extend for varying periods of time depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, patents issued from applications filed in the United States are effective for twenty years from the earliest non-provisional filing date. In addition, in certain instances, a patent term can be extended to recapture a portion of the term effectively lost as a result of the FDA regulatory review period, however, the restoration period cannot be longer than five years and the total patent term including the restoration period must not exceed 14 years following FDA approval. The duration of foreign patents varies in accordance with provisions of applicable local law, but typically is also twenty years from the earliest international filing date. Our issued patents and pending applications with respect to our clinical stage therapeutic candidates will expire on dates ranging from 2026 to 2035,2037, exclusive of possible patent term extensions, However, the actual protection afforded by a patent varies on a product by productproduct-by-product basis, from country to country and depends upon many factors, including the type of patent, the scope of its coverage, the availability of extensions of patent term, the availability of legal remedies in a particular country and the validity and enforceability of the patent.
National and international patent laws concerning therapeutic candidates remain highly unsettled. No consistent policy regarding the patent-eligibility or the breadth of claims allowed in such patents has emerged to date in the United States, Europe or other countries. Changes in either the patent laws or in interpretations of patent laws in the United States and other countries can diminish our ability to protect our inventions and enforce our intellectual property rights. Accordingly, we cannot predict the breadth or enforceability of claims that may be granted in our patents or in third-party patents. The biotechnology and pharmaceutical industries are characterized by extensive litigation regarding patents and other intellectual property rights. Our ability to maintain and solidify our proprietary position for our drugs and technology will depend on our success in obtaining effective claims and enforcing those claims once granted. We do not know whether any of the patent applications that we may file or license from third parties will result in the issuance of any patents. The issued patents that we own or may receive in the future, may be challenged, invalidated or circumvented, and the rights granted under any issued patents may not provide us with sufficient protection or competitive advantages against competitors with similar technology. Furthermore, our competitors may be able to independently develop and commercialize similar drugs or duplicate our technology, business model or strategy without infringing our patents. Because of the extensive time required for clinical development and regulatory review of a drug we may develop, it is possible that, before any of our drugs can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby reducing any advantage of any such patent. The patent positions for our most advanced programs are summarized below:
Luspatercept Patent Coverage
We hold two issued patents covering the luspatercept composition of matter in the United States, one issued patent in Europe (registered in most countries of the European Patent Convention) and additional patents issued or pending in many other major jurisdictions worldwide, including Europe, Australia, Canada, Japan, China, South Korea, Brazil, Mexico, Russia and India. The expected expiration dates for these composition of matter patents are 2028 and 2029, exclusive of possible patent term extensions.
We hold two issued patents covering the treatment of anemia by administration of luspatercept in the United States and similar patents issued or pending in other major jurisdictions worldwide, including Europe, Australia, Canada, Japan, China, South Korea, Brazil, Mexico, Russia and India. The expected expiration date for these method of treatment patents is 2029, exclusive of possible patent term extensions.
We also hold patent applications directed to a variety of other uses for luspatercept, including the treatment of complications of thalassemia, such as iron overload and ulcers, and treatment of patient sub-groups with MDS.luspatercept. The expected expiration date for these method of treatment patents ranges from 2029 to 20352037 exclusive of possible patent term extensions.
Sotatercept Patent Coverage
We hold two issued patents covering the sotatercept composition of matter in the United States, one issued patent in Europe (registered in most countries of the European Patent Convention) and additional patents issued or pending in many other major jurisdictions worldwide, including Australia, Canada, Japan, China, South Korea, Brazil, Mexico, Russia, Israel and India. The expected expiration date for these composition of matter patents is 2026, exclusive of possible patent term extensions.
We hold three issued patentsone pending patent application covering the treatment of anemiapulmonary arterial hypertension by administration of sotatercept in the United States, and similar patentswhich if issued or pendingis expected to expire in many other major jurisdictions worldwide, including Europe, Australia, Canada, Japan, China, South Korea, Brazil, Mexico, Russia, Israel and India. The expected expiration date for these method of treatment patents is 2027,2037, exclusive of possible patent term extensions. This patent application is also eligible for filing in major jurisdictions worldwide.
We also hold patents and patent applications directed to a variety of other uses for sotatercept, including the treatment of multiple myeloma and the treatment of bone loss in patients with chronic kidney disease.
ACE-083 Patent Coverage
We hold two pending patent applications covering the ACE-083 composition of matter in the United States, which, if issued are expected to expire in 2029 and 2035, respectively, exclusive of possible patent term extensions. TheseWe hold additional pending patent applications are also eligible for filingcovering the ACE-083 composition of matter in many other major jurisdictions worldwide.worldwide, including Europe, Australia, Canada, Japan, China, South Korea, Brazil, Mexico, Russia and India. The expected expiration date for these patent applications, should they issue as patents, is 2035, exclusive of possible patent term extensions.
We also hold patent applications directed to a variety of uses for ACE-083, including the treatment of muscular dystrophies, such as facioscapulohumeral muscular dystrophy and Charcot-Marie-Tooth disease. The expected expiration date for these method of treatment patent applications, should they issue as patents, is 2035, exclusive of possible patent term extensions.
ACE-2494 Patent Coverage
We hold two pending patent applications covering the ACE-2494 composition of matter in the United States, which, if issued, are expected to expire in 2036 and 2037, respectively, exclusive of possible patent term extensions. We hold additional pending patent applications covering the ACE-2494 composition of matter in many other major jurisdictions worldwide, including Europe, Australia, Canada, Japan, China, South Korea, Brazil, Mexico and India. The expected expiration date for these patent applications, should they issue as patents, ranges from 2036 to 2037, exclusive of possible patent term extensions.
We also hold patent applications directed to a variety of uses for ACE-2494, including the treatment of systemic muscle disorders. The expected expiration date for these method of treatment patent applications, should they issue as patents, ranges from 2036 to 2037, exclusive of possible patent term extensions.
Trade Secrets
In addition to patents, we rely upon unpatented trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality agreements with our commercial partners, collaborators, employees and consultants and invention assignment agreements with our employees. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. These agreements may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
In-Licenses
In August 2010, we entered into two amended and restated license agreements with the Salk Institute for Biological Studies, or Salk, providing rights under U.S. patent filings solely owned by Salk. The agreements for the licensed patent rights relate to the first cloning of the type II activin receptors, human ActRIIA and frog ActRIIB, respectively, and include claims to
vertebrate homolog nucleic acids and proteins with respect to each of the foregoing. These patent rights expire between the years 2016 and 2017. One of these agreements relates to ActRIIA and sotatercept; the other agreement relates to ActRIIB, luspatercept and the discontinued program ACE-031, which we refer to as the ActRIIB Agreement. The licenses granted are exclusive as to the therapeutic products that are covered by the patents and non-exclusive as to diagnostic products and other products that are developed using the Salk patent rights. If we sublicense the Salk patent rights, we will owe Salk a percentage of sublicensing revenue, excluding payments based on sales. Under the agreements, Salk retained rights, on behalf of itself and other non-profit academic institutions, to practice under the licensed rights for non-profit purposes. We agreed to pay Salk specified development milestone payments totaling up to $2.0 million for sotatercept and $0.7 million for luspatercept. In addition, we are required to pay Salk royalties in the low single-digits on worldwide net product sales by us or our sublicensees of products claimed in the licensed patents, or derived from use of the licensed patent rights, with royalty obligations continuing at a reduced rate for a period of time after patent expiration. The agreements terminate upon the expiration of royalty obligations. We may terminate either agreement at any time by giving Salk advance written notice. Either agreement may also be terminated by Salk in the event of a material breach by us or in the event we become subject to bankruptcy or similar circumstances.
In October 2012, Salk filed a lawsuit against us alleging that we breached the ActRIIB Agreement. In July 2014, we settled the Salk lawsuit and entered into an amendment to the ActRIIB Agreement with Salk. Pursuant to the settlement, we made a one-time total payment of $5 million, inclusive of interest, to Salk and we agreed to pay Salk 6% of future development milestone payments received under the agreement with Celgene relating to luspatercept. Finally, we and Salk have further agreed that the royalty percentage on net sales of luspatercept will remain at 1% as provided in the ActRIIB Agreement with Salk, and that such royalty will be payable until June 2022.
Government Regulation
The preclinical studies and clinical testing, manufacture, labeling, storage, record keeping, advertising, promotion, export, marketing and sales, among other things, of our therapeutic candidates and future products, are subject to extensive regulation by governmental authorities in the United States and other countries. In the United States, pharmaceutical products are regulated by the FDA under the Federal Food, Drug, and Cosmetic Act and other laws, including, in the case of biologics, the Public Health Service Act. We expect sotatercept, luspatercept, dalantercept, ACE-083 and ACE-2494 to be regulated by the FDA as biologics and to be reviewed by the Center for Drug Evaluation and Research (CDER) as proteins intended for therapeutic use. Therapeutic candidates require the submission of a Biologics License Application, or BLA, and approval by the FDA prior to being marketed in the U.S. Manufacturers of therapeutic candidates may also be subject to state regulation. Failure to comply with FDA requirements, both before and after product approval, may subject us or our partners, contract manufacturers, and suppliers to administrative or judicial sanctions, including FDA refusal to approve applications, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, fines and/or criminal prosecution.
The steps required before a biologic may be approved for marketing of an indication in the United States generally include:
completion of preclinical laboratory tests, animal studies and formulation studies conducted according to Good Laboratory Practices, or GLPs, and other applicable regulations;
submission to the FDA of an Investigational New Drug application or IND, which must become effective before human clinical trials may commence;
completion of adequate and well-controlled human clinical trials in accordance with Good Clinical Practices, or GCPs, to establish that the biological product is "safe, pure and potent", which is analogous to the safety and efficacy approval standard for a chemical drug product for its intended use;
submission to the FDA of a BLA;
satisfactory completion of an FDA pre-approval inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with applicable current Good Manufacturing Practice requirements, or cGMPs; and
FDA review of the BLA and issuance of a biologics license which is the approval necessary to market a therapeutic candidate.
Preclinical studies include laboratory evaluation of product chemistry, toxicity and formulation as well as animal studies to assess the potential safety and efficacy of the biologic candidate. Preclinical studies must be conducted in compliance with FDA regulations regarding GLPs. The results of the preclinical tests, together with manufacturing information and analytical
data, are submitted to the FDA as part of an IND. Some preclinical testing may continue even after the IND is submitted. In addition to including the results of the preclinical testing, the IND will also include a protocol detailing, among other things, the objectives of the clinical trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated if the first phase or phases of the clinical trial lends themselves to an efficacy determination. The IND will automatically become effective 30 days after receipt by the FDA, unless the FDA within the 30-day time period places the IND on clinical hold because of its concerns about the drug candidate or the conduct of the trial described in the clinical protocol included in the IND. The IND sponsor and the FDA must resolve any outstanding concerns before clinical trials can proceed.
All clinical trials must be conducted under the supervision of one or more qualified principal investigators in accordance with GCPs. They must be conducted under protocols detailing the objectives of the applicable phase of the trial, dosing procedures, research subject selection and exclusion criteria and the safety and effectiveness criteria to be evaluated. Each protocol must be submitted to the FDA as part of the IND, and progress reports detailing the status of the clinical trials must be submitted to the FDA annually. Sponsors also must timely report to the FDA serious and unexpected adverse reactions, any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator's brochure, or any findings from other studies or animal or in vitro testing that suggest a significant risk in humans exposed to the drug. An institutional review board, or IRB, at each institution participating in the clinical trial must review and approve the
protocol before a clinical trial commences at that institution, approve the information regarding the trial and the consent form that must be provided to each research subject or the subject's legal representative, and monitor the study until completed.
Clinical trials are typically conducted in three sequential phases, but the phases may overlap and different trials may be initiated with the same drug candidate within the same phase of development in similar or differing patient populations. Phase 1 trials may be conducted in a limited number of patients, but are usually conducted in healthy volunteer subjects. The drug candidate is initially tested for safety and, as appropriate, for absorption, metabolism, distribution, excretion, pharmacodynamics and pharmacokinetics.
Phase 2 trials usually involve a larger, but still limited, patient population to evaluate preliminarily the efficacy of the drug candidate for specific, targeted indications to determine dosage tolerance and optimal dosage and to identify possible short-term adverse effects and safety risks.
Phase 3 trials are undertaken to further evaluate clinical efficacy of a specific endpoint and to test further for safety within an expanded patient population at geographically dispersed clinical trial sites. Phase 1, Phase 2, or Phase 3 testing might not be completed successfully within any specific time period, if at all, with respect to any of our therapeutic candidates. Results from one trial are not necessarily predictive of results from later trials. Furthermore, the FDA or the sponsor may suspend clinical trials at any time on various grounds, including a finding that the subjects or patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB's requirements or if the drug candidate has been associated with unexpected serious harm to patients.
The results of the preclinical studies and clinical trials, together with other detailed information, including information on the manufacture and composition of the product, are submitted to the FDA as part of a BLA requesting approval to market the drug candidate for a proposed indication. Under the Prescription Drug User Fee Act (PDUFA), as re-authorized most recently in July 2012,August 2017 (PDUFA VI), the fees payable to the FDA for reviewing a BLA, as well as annual program fees for commercial manufacturing establishments and for approved products, can be substantial. The feeFor fiscal years 2018-2022, user fees for review of an application that requires clinical data, such as a BLA, for the one year period ending September 30, 2016, isplus prescription drug program fees, are estimated at approximately $2.4$2.7 million, subject to certain limited deferrals, waivers, and reductions that may be available.available, including waiver of the application fee for a product designated for an orphan indication. The fees typically increase each year. Each BLA submitted to the FDA for approval is reviewed for administrative completeness and reviewability within 60 days following receipt by the FDA of the application. If the BLA is found complete, the FDA will file the BLA, triggering a full review of the application. The FDA may refuse to file any BLA that it deems incomplete or not properly reviewable at the time of submission. The FDA's established goal is to review 90% of priority BLA applications within six months after the application is accepted for filing and 90% of standard BLA applications within 10 months of the acceptance date, whereupon a review decision is to be made. The FDA, however, may not approve a drug candidate within these established goals and its review goals are subject to change from time to time. Further, the outcome of the review, even if generally favorable, may not be an actual approval but a "complete response letter" that describes additional work that must be done before the application can be approved. Before approving a BLA, the FDA may inspect the facility or facilities at which the product is manufactured and will not approve the product unless the facility complies with cGMPs. The FDA may deny approval of a BLA if applicable statutory or regulatory criteria are not satisfied, or may require additional testing or information, which can extend the review process. FDA approval of any application may include many delays or never be granted. If a product is approved, the approval may impose limitations on the uses for which the product may be marketed, may require that warning statements be included in the product labeling, may require that additional studies be conducted following approval as a condition of the approval, and may impose restrictions and conditions on product distribution, prescribing, or dispensing in the form of a Risk Evaluation and Mitigation Strategy, or REMS, or
otherwise limit the scope of any approval. The FDA must approve a BLA supplement or a new BLA before a product may be marketed for other uses or before certain manufacturing or other changes may be made. Further post-marketing testing and surveillance to monitor the safety or efficacy of a product is required. Also, product approvals may be withdrawn if compliance with regulatory standards is not maintained or if safety or manufacturing problems occur following initial marketing. In addition, new government requirements may be established that could delay or prevent regulatory approval of our therapeutic candidates under development.
As part of the Patient Protection and Affordable Care Act of 2010, under the subtitle of Biologics Price Competition and Innovation Act of 2009, or the BPCI, a statutory pathway has been created for licensure, or approval, of biological products that are biosimilar to, and possibly interchangeable with, earlier biological products licensed under the Public Health Service Act. Also under the BPCI, innovator manufacturers of original reference biological products are granted 12 years of exclusivity before biosimilars can be approved for marketing in the United States. The objectives of the BPCI are conceptually similar to those of the Drug Price Competition and Patent Term Restoration Act of 1984, commonly referred to as the "Hatch-Waxman Act", which established abbreviated pathways for the approval of drug products. The implementation of an abbreviated approval pathway for biological products is under the direction of the FDA and is currently being developed. In late 2010,FDA. Since the enactment of the BPCI, the FDA held a hearing to receive comments from a broad group of stakeholders regarding the implementation of the BPCI. Since that hearing in 2010, the FDA, in February 2012 and February 2013, has
issued several draft guidances for industry related to the BPCI, addressing scientific, quality and procedural issues relevant to an abbreviated application for a biosimilar product.product and has approved fewer than ten such products to date. The approval of a biologic product biosimilar to one of our products could have a material adverse impact on our business as it may be significantly less costly to bring to market and may be priced significantly lower than our products.
Both before and after the FDA approves a product, the manufacturer and the holder or holders of the BLA for the product are subject to comprehensive regulatory oversight. For example, quality control and manufacturing procedures must conform, on an ongoing basis, to cGMP requirements, and the FDA periodically inspects manufacturing facilities to assess compliance with cGMPs. Accordingly, manufacturers must continue to spend time, money and effort to maintain cGMP compliance.
Orphan Drug Act
The Orphan Drug Act provides incentives to manufacturers to develop and market drugs for rare diseases and conditions affecting fewer than 200,000 persons in the United States at the time of application for orphan drug designation. Orphan drug designation must be requested before submitting a BLA. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. If a product that has orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the holder of the approval is entitled to a seven-year exclusive marketing period in the United States for that product except in very limited circumstances. For example, a drug that the FDA considers to be clinically superior to, or different from, another approved orphan drug, even though for the same indication, may also obtain approval in the United States during the seven-year exclusive marketing period. In addition, holders of exclusivity for orphan drugs are expected to assure the availability of sufficient quantities of their orphan drugs to meet the needs of patients. Failure to do so could result in the withdrawal of marketing exclusivity for the drug. Luspatercept has orphan drug designation in the United States for the treatment of beta-thalassemia and for the treatment of MDS.
Legislation similar to the Orphan Drug Act has been enacted outside the U.S., including in the EU. The orphan legislation in the EU is available for therapies addressing chronic debilitating or life-threatening conditions that affect five or fewer out of 10,000 persons or are financially not viable to develop. The market exclusivity period is for ten years, although that period can be reduced to six years if, at the end of the fifth year, available evidence establishes that the product is sufficiently profitable not to justify maintenance of market exclusivity. The market exclusivity may be extended to 12 years if sponsors complete a pediatric investigation plan agreed upon with the relevant committee of the EMA.
Expedited Review and Approval
The FDA has various programs, including Fast Track, priority review, and accelerated approval, which are intended to expedite or simplify the process for reviewing drugs, and/or provide for the approval of a drug on the basis of a surrogate endpoint. Even if a drug qualifies for one or more of these programs, the FDA may later decide that the drug no longer meets the conditions for qualification or that the time period for FDA review or approval will be shortened. Generally, drugs that are eligible for these programs are those for serious or life-threatening conditions, those with the potential to address unmet medical needs and those that offer meaningful benefits over existing treatments. For example, Fast Track is a process designed to facilitate the development and expedite the review of drugs to treat serious or life- threatening diseases or conditions and fill unmet medical needs. Priority review is designed to give drugs that offer major advances in treatment or provide a treatment where no adequate therapy exists an initial review within six months as compared to a standard review time of ten months. Although Fast Track and priority review do not affect the standards for approval, the FDA will attempt to facilitate early and frequent meetings with a sponsor of a Fast Track designated drug and expedite review of the application for a drug designated for priority review. Accelerated approval provides for an earlier approval for a new drug that is intended to treat a serious or
life-threatening disease or condition and that fills an unmet medical need based on a surrogate endpoint. A surrogate endpoint is a laboratory measurement or physical sign used as an indirect or substitute measurement representing a clinically meaningful outcome. As a condition of approval, the FDA may require that a sponsor of a drug candidate receiving accelerated approval perform post-marketing clinical trials to confirm the clinically meaning full outcome as predicted by the surrogate marker trial.
In the Food and Drug Administration Safety and Innovation Act, or FDASIA, which was signed into law in July 2012, Congress encouraged the FDA to utilize innovative and flexible approaches to the assessment of products under accelerated approval. In May 2014, the FDA published a Guidance for Industry entitled, "Expedited Programs for Serious Conditions—Drugs and Biologics" which provides guidance on FDA programs that are intended to facilitate and expedite development and review of new drugs as well as threshold criteria generally applicable to concluding that a drug is a candidate for these expedited development and review programs. In addition to the Fast Track, accelerated approval and priority review programs discussed above, the FDA also provided guidance on a new program for Breakthrough Therapy designation. A request for Breakthrough Therapy designation should be submitted concurrently with, or as an amendment to an IND. FDA has already granted this designation to over 100nearly 200 new drugs and has approved more than 2550 Breakthrough Therapy designated drugs.
Pediatric Exclusivity and Pediatric Use
Under the Best Pharmaceuticals for Children Act, or BPCA, certain drugs may obtain an additional six months of exclusivity, if the sponsor submits information requested in writing by the FDA, or a Written Request, relating to the use of the active moiety of the drug in children. The FDA may not issue a Written Request for studies on unapproved or approved indications or where it determines that information relating to the use of a drug in a pediatric population, or part of the pediatric population, may not produce health benefits in that population.
We have not received a Written Request for such pediatric studies, although we may ask the FDA to issue a Written Request for such studies in the future. To receive the six-month pediatric market exclusivity, we would have to receive a Written Request from the FDA, conduct the requested studies in accordance with a written agreement with the FDA or, if there is no written agreement, in accordance with commonly accepted scientific principles, and submit reports of the studies. A Written Request may include studies for indications that are not currently in the labeling if the FDA determines that such information will benefit the public health. The FDA will accept the reports upon its determination that the studies were conducted in accordance with and are responsive to the original Written Request or commonly accepted scientific principles, as appropriate, and that the reports comply with the FDA's filing requirements.
In addition, the Pediatric Research Equity Act, or PREA, requires a sponsor to conduct pediatric studies for most drugs and biologicals, for a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration. Under PREA, original NDAs, BLAs and supplements thereto must contain a pediatric assessment unless the sponsor has received a deferral or waiver. The required assessment must include the evaluation of the safety and effectiveness of the product for the claimed indications in all relevant pediatric subpopulations and support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The sponsor or FDA may request a deferral of pediatric studies for some or all of the pediatric subpopulations. A deferral may be granted for several reasons, including a finding that the drug or biologic is ready for approval for use in adults before pediatric studies are complete or that additional safety or effectiveness data needs to be collected before the pediatric studies begin. After April 2013, the FDA must send a non-compliance letter to any sponsor that fails to submit the required assessment, keep a deferral current or fails to submit a request for approval of a pediatric formulation.
As part of the FDASIA, Congress made a few revisions to BPCA and PREA, which were slated to expire on September 30, 2012, and made both laws permanent.
Reimbursement
In both domestic and foreign markets, sales and reimbursement of any approved products will depend, in part, on the extent to which the costs of such products will be covered by third-party payers, such as government health programs, commercial insurance and managed healthcare organizations. These third-party payers are increasingly challenging the prices charged for medical products and services and imposing controls to manage costs. The containment of healthcare costs has become a priority of federal and state governments and the prices of drugs have been a focus in this effort. Governments have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our net revenue and results. In addition, there is significant uncertainty regarding the reimbursement status of newly approved healthcare products. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the cost-effectiveness of our products. If third-party payers do not consider our products to be cost-effective compared to other therapies, the payers may not cover our
products after approved as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products on a profitable basis.
Within the United States, if we obtain appropriate approval in the future to market any of our current therapeutic candidates, we may seek approval and coverage for those products under Medicaid, Medicare and the Public Health Service (PHS) pharmaceutical pricing program and also seek to sell the products to federal agencies.
Medicaid is a joint federal and state program that is administered by the states for low income and disabled beneficiaries. Under the Medicaid Drug Rebate Program, manufacturers are required to pay a rebate for each unit of product reimbursed by the state Medicaid programs. The amount of the rebate for each product is set by law and may be subject to an additional discount if certain pricing increases more than inflation.
Medicare is a federal program administered by the federal government that covers individuals age 65 and over as well as those with certain disabilities. Medicare Part D provides coverage to enrolled Medicare patients for self-administered drugs (i.e., drugs that do not need to be administered by a physician). Medicare Part D is administered by private prescription drug plans approved by the U.S. government and each drug plan establishes its own Medicare Part D formulary for prescription drug coverage and pricing, which the drug plan may modify from time-to-time.
Medicare Part B covers most injectable drugs given in an in-patient setting, and some drugs administered by a licensed medical provider in hospital outpatient departments and doctorsdoctors' offices. Medicare Part B is administered by Medicare Administrative Contractors, which generally have the responsibility of making coverage decisions. Subject to certain payment adjustments and limits, Medicare generally pays for Part B covered drugs based on a percentage of manufacturer-reported average sales price.
Drug products are subject to discounted pricing when purchased by federal agencies via the Federal Supply Schedule (FSS). FSS participation is required for a drug product to be covered and paid for by certain federal agencies and for coverage under Medicaid, Medicare Part B and the PHS pharmaceutical pricing program. FSS pricing is negotiated periodically with the Department of Veterans Affairs. FSS pricing is intended to not exceed the price that a manufacturer charges its most-favored non-federal customer for its product. In addition, prices for drugs purchased by the Veterans Administration, Department of Defense (including drugs purchased by military personnel and dependents through the TRICARE retail pharmacy program), Coast Guard, and PHS are subject to a cap on pricing (known as the "federal ceiling price") and may be subject to an additional discount if pricing increases more than inflation.
To maintain coverage of drugs under the Medicaid Drug Rebate Program, manufacturers are required to extend discounts to certain purchasers under the PHS pharmaceutical pricing program. Purchasers eligible for discounts include hospitals that serve a disproportionate share of financially needy patients, community health clinics and other entities that receive health services grants from the PHS.
The American Recovery and Reinvestment Act of 2009 provides funding for the federal government to compare the effectiveness of different treatments for the same illness. A plan for the research will be developed by the Department of Health and Human Services, the Agency for Healthcare Research and Quality and the National Institutes for Health, and periodic reports on the status of the research and related expenditures will be made to Congress. Although the results of the comparative effectiveness studies are not intended to mandate coverage policies for public or private payers, it is not clear what effect, if any, the research will have on the sales of any product, if any such product or the condition that it is intended to treat is the subject of a study. It is also possible that comparative effectiveness research demonstrating benefits in a competitor's product could adversely affect the sales of any of our approved therapeutic candidates. If third-party payers do not consider our products to be cost-effective compared to other available therapies, they may not cover our products as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products on a profitable basis.
Recently, there has been considerable public and government scrutiny in the U.S. of pharmaceutical pricing and proposals to address the perceived high cost of pharmaceuticals. There have also been several recent state legislative efforts to address drug costs, which generally have focused on increasing transparency around drug costs or limiting drug prices or price increases. Adoption of other new legislation at the federal or state level could further limit reimbursementaffect demand for, pharmaceuticals.or pricing of, our product candidates if approved for sale.
We cannot predict the ultimate content, timing or effect of any changes to the Healthcare Reform Act or other federal and state reform efforts. There is no assurance that federal or state health care reform will not adversely affect our future business and financial results.
Outside the United States, ensuring adequate coverage and payment for our products will face challenges. Pricing of prescription pharmaceuticals is subject to governmental control in many countries. Pricing negotiations with governmental authorities can extend well beyond the receipt of regulatory marketing approval for a product and may require us to conduct a clinical trial that compares the cost effectiveness of our therapeutic candidates or products to other available therapies. The conduct of such a clinical trial could be expensive and result in delays in our commercialization efforts. Third-party payers are challenging the prices charged for medical products and services, and many third-party payers limit reimbursement for newly-approved healthcare products. Recent budgetary pressures in many European Union countries are also causing governments to
consider or implement various cost-containment measures, such as price freezes, increased price cuts and rebates. If budget pressures continue, governments may implement additional cost-containment measures. Cost-control initiatives could decrease the price we might establish for products that we may develop or sell, which would result in lower product revenues or royalties payable to us. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products.
Foreign Regulation
In addition to regulations in the United States, we will be subject to a variety of foreign regulations governing clinical trials and commercial sales and distribution of our therapeutic candidates. Whether or not we obtain FDA approval for a therapeutic candidate, we must obtain approval from the comparable regulatory authorities of foreign countries or economic areas, such as the European Union, before we may commence clinical trials or market products in those countries or areas. The approval process and requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from place to place, and the time may be longer or shorter than that required for FDA approval.
Certain countries outside of the United States have a process that requires the submission of a clinical trial application much like an IND prior to the commencement of human clinical trials. In Europe, for example, a clinical trial application, or CTA, must be submitted to the competent national health authority and to independent ethics committees in each country in which a company intends to conduct clinical trials. Once the CTA is approved in accordance with a country's requirements, clinical trial development may proceed in that country. In all cases, the clinical trials must be conducted in accordance with good clinical practices, or GCPs and other applicable regulatory requirements.
Under European Union regulatory systems, a company may submit marketing authorization applications either under a centralized or decentralized procedure. The centralized procedure is compulsory for medicinal products produced by biotechnology or those medicinal products containing new active substances for specific indications such as the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, viral diseases and designated orphan medicines, and optional for other medicines which are highly innovative. Under the centralized procedure, a marketing application is submitted to the European Medicines Agency where it will be evaluated by the Committee for Medicinal Products for Human Use and a favorable opinion typically results in the grant by the European Commission of a single marketing authorization that is valid for all European Union member states within 67 days of receipt of the opinion. The initial marketing authorization is valid for five years, but once renewed is usually valid for an unlimited period. The decentralized procedure provides for approval by one or more "concerned" member states based on an assessment of an application performed by one member state, known as the "reference" member state. Under the decentralized approval procedure, an applicant submits an application, or dossier, and related materials to the reference member state and concerned member states. The reference member state prepares a draft assessment and drafts of the related materials within 120 days after receipt of a valid application. Within 90 days of receiving the reference member state's assessment report, each concerned member state must decide whether to approve the assessment report and related materials. If a member state does not recognize the marketing authorization, the disputed points are eventually referred to the European Commission, whose decision is binding on all member states.
As in the United States, we may apply for designation of a product as an orphan drug for the treatment of a specific indication in the European Union before the application for marketing authorization is made. Orphan drugs in Europe enjoy economic and marketing benefits, including up to 10 years of market exclusivity for the approved indication unless another applicant can show that its product is safer, more effective or otherwise clinically superior to the orphan designated product.
Fraud and Abuse Laws
Laws and regulations have been enacted by the federal government and various states to regulate the sales and marketing practices of pharmaceutical manufacturers with marketed products. The laws and regulations generally limit financial interactions between manufacturers and health care providers and/or knowingly making,require disclosure to the government and public of such interactions. Many of these laws and regulations contain ambiguous requirements or causingrequire administrative guidance for implementation. Given the lack of clarity in laws and their implementation, any future activities (if we obtain approval and/or reimbursement from federal healthcare programs for our product candidates) could be subject to be made, a false statement
Additional Regulation
We are also subject to regulation under the Occupational Safety and Health Act, the Environmental Protection Act, the Toxic Substances Control Act, the Resource Conservation and Recovery Act and other present and potential federal, state or local regulations. These and other laws govern our use, handling and disposal of various biological and chemical substances used in, and waste generated by our operations. Our research and development involves the controlled use of hazardous materials, chemicals and viruses. Although we believe that our safety procedures for handling and disposing of such materials comply with the standards prescribed by state and federal regulations, the risk of accidental contamination or injury from these materials cannot be completely eliminated. In the event of such an accident, we could be held liable for any damages that result and any such liability could exceed our resources.
There have been a number of federal and state proposals during the last few years regarding the pricing of pharmaceutical and biological products, government control and other changes to the healthcare system of the U.S. It is uncertain what legislative proposals will be adopted or what actions federal, state or private payers for medical goods and services may take in response to any healthcare reform proposals or legislation. We cannot predict the effect medical or healthcare reforms may have on our business, and no assurance can be given that any such reforms will not have a material adverse effect.
Manufacturing
We currently manufacture drug substance for our preclinical studies, Phase 1 clinical trials and Phase 2 clinical trials of luspatercept, dalantercept, ACE-083 and ACE-2494. We manufacture material compliant to U.S. and European cGMP at our 12,000 square foot multi-product facility located at our corporate headquarters in Cambridge, Massachusetts. We have the capabilities to manufacture receptor fusion proteins, monoclonal antibodies, and other therapeutic candidates.
Our manufacturing facility is based on single use, disposable technology to maximize the focus of personnel and other resources on the production process, minimizing the need for cleaning and sterilization while optimizing the efficiency of product change-over. The facility consists of four independent clean rooms totaling 4,000 square feet. The facility includes one 250 liter and one 1,000 liter single use bioreactor and has space for two additional 1,000 liter bioreactors.
Approximately 2234 full time employees focus on our process development and manufacturing activities. We believe that our strategic investment in manufacturing capabilities allows us to advance our therapeutic candidates at a more rapid pace and provides us with more portfolio flexibility than if we used a contract manufacturer. The facility produces drug substance in a cost-effective manner while allowing us to retain control over the process and provides an ability to balance the requirements of multiple programs and avoid costly commitments of funds before clinical data are available.
Our manufacturing capabilities encompass the full manufacturing process through quality control and quality assurance. These groups are integrated with our project teams from discovery through development. This structure enables us to efficiently transfer preclinical stage lead molecules into manufacturing. We have designed our manufacturing facility and processes to provide maximum flexibility and rapid change over for the manufacture of different therapeutic candidates. We outsource fill-finish, packaging, labeling, shipping, and distribution.
We manufacture our therapeutic candidates using readily available raw materials and well established manufacturing procedures based on a standardized process modified for each of our therapeutic candidates. We produce our proteins in bioreactors using Chinese hamster ovary cells that have been genetically engineered to produce our specific therapeutic candidates. We then purify the proteins using industry standard methods, which include affinity chromatography and ultrafiltration operations. Processes developed within our facility have been successfully transferred to commercial facilities based on stainless steel bioreactors. We have conducted comparability characterization on sotatercept between our Phase 2 material and material made at a commercial manufacturer and found them to be comparable.
We believe that we can scale our manufacturing processes to support our clinical development programs and the potential commercialization of our therapeutic candidates. For our early phase therapeutic candidates, we intend to continue to manufacture drug substance for preclinical testing and Phase 1 and Phase 2 clinical development at our current facilities. AsFor the luspatercept progresses to Phase 3 clinical trials, we intend to transferhave transferred the process for Phase 3 production to Celgene, under the terms of our collaboration agreements. Celgene uses contract manufacturers for Phase 3 supply of luspatercept, and we expect Celgene will use a contract manufacturer for commercial supply of luspatercept. We have alreadyalso previously successfully transferred the manufacturing process for sotatercept to Celgene, and we expect Celgene will use a contract manufacturer for Phase 3additional clinical supply and any future commercial supply of sotatercept and luspatercept.sotatercept. We intend to contract with a third party manufacturer for the supply of dalanterceptACE-083 and ACE-083ACE-2494 for any Phase 3 clinical trials.
Employees
As of December 31, 2015,2017, we had 102139 full-time employees, 76108 of whom are involved in research, development or manufacturing, and 2735 of whom have Ph.D. or M.D. degrees. We have no collective bargaining agreements with our employees and we have not experienced any work stoppages. We consider our relations with our employees to be good.
Corporate Information
We were incorporated in the state of Delaware in June 2003 as Phoenix Pharma, Inc., and we subsequently changed our name to Acceleron Pharma Inc. and commenced operations in February 2004. Our principal executive offices are located at 128 Sidney Street, Cambridge, Massachusetts 02139, and our telephone number is (617) 649-9200. Our Internet address is www.acceleronpharma.com. The contents of our website are not part of this Annual Report on Form 10-K. We make available free of charge through our website our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to these reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended, or the Exchange Act, as soon as reasonably practicable after we electronically file or furnish such materials to the Securities and Exchange Commission.
Item 1A. Risk Factors
Investing in our common stock involves a high degree of risk. You should carefully consider the risks and uncertainties described below in addition to the other information included or incorporated by reference in this Annual Report on Form 10-K before purchasing our common stock. If any of the following risks actually occurs, our business, financial condition or results of operations would likely suffer, possibly materially. In that case, the trading price of our common stock could fall, and you may lose all or part of the money you paid to buy our common stock. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently believe to be immaterial may also adversely affect our business.
Risks Related to Our Financial Position and Need for Additional Capital
We have incurred net operating losses since our inception and anticipate that we will continue to incur substantial operating losses for the foreseeable future. We may never achieve or sustain profitability.
We have incurred net losses during most fiscal periods since our inception. As of December 31, 2015,2017, we had an accumulated deficit of $307.5$473.0 million. We do not know whether or when we will become profitable. To date, we have not commercialized any products or generated any revenues from the sale of products, and we do not expect to generate any product revenues in the foreseeable future. Our losses have resulted principally from costs incurred in our discovery and development activities.
We anticipate that our expenses will increase in the future as we expand our discovery, research, development, manufacturing and commercialization activities. However, we also anticipate that these increased expenses will be partially offset by milestone payments we expect to receive under our agreements with Celgene and potentially by payments we may receive under new collaboration arrangements we may enter into with third parties for dalantercept, ACE-083, ACE-2494 or other therapeutic candidates. If we do not receive the anticipated milestone payments or do not enter into partnerships for dalantercept, ACE-083, ACE-2494 or other therapeutic candidates on acceptable terms, our operating losses will substantially increase over the next several years as we execute our plan to expand our discovery, research, development, manufacturing and commercialization activities. There can be no assurance that we will enter into a new collaboration or achieve milestones and, therefore, no assurance our losses will not increase prohibitively in the future.
To become and remain profitable, we or our partners must succeed in developing our therapeutic candidates, obtaining regulatory approval for them, and manufacturing, marketing and selling those products for which we or our partners may obtain regulatory approval. We or they may not succeed in these activities, and we may never generate revenue from product sales that is significant enough to achieve profitability. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Our failure to become or remain profitable would depress our market value and could impair our ability to raise capital, expand our business, discover or develop other therapeutic candidates or continue our operations. A decline in the value of our company could cause you to lose all or part of your investment.
We will require substantial additional financing to achieve our goals, and a failure to obtain this necessary capital when needed could force us to delay, limit, reduce or terminate our product development or commercialization efforts.
As of December 31, 2015,2017, our cash, cash equivalents and investments were $136.0$372.9 million. We believe that we will continue to expend substantial resources for the foreseeable future developing dalantercept,sotatercept in the PH field, ACE-083, ACE-2494 and new therapeutic candidates. These expenditures will include costs associated with research and development, potentially acquiring new technologies, conducting preclinical studies and clinical trials, potentially obtaining regulatory approvals and manufacturing products, as well as marketing and selling products approved for sale, if any. In addition, other unanticipated costs may arise. Because the outcome of our planned and anticipated clinical trials is highly uncertain, we cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of our therapeutic candidates.
Celgene generally pays development, manufacturing and commercialization and certain patent costs for luspatercept and for sotatercept and luspatercept.outside of the PH field. Other than those costs, our future capital requirements depend on many factors, particularly in connection with the development of our other therapeutic candidates including dalanterceptsotatercept in the PH field, ACE-083 and ACE-083:ACE-2494:
the scope, progress, results and costs of researching and developing our other therapeutic candidates, and conducting preclinical studies and clinical trials;
the timing of, and the costs involved in, obtaining regulatory approvals for our other therapeutic candidates if clinical trials are successful;
the cost of commercialization activities for our other therapeutic candidates, if any of these therapeutic candidates is approved for sale, including marketing, sales and distribution costs;
the cost of manufacturing our other therapeutic candidates for clinical trials in preparation for regulatory approval and in preparation for commercialization;
our ability to establish and maintain strategic partnerships, licensing or other arrangements and the financial terms of such agreements;
the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims, including litigation costs and the outcome of such litigation; and
the timing, receipt, and amount of sales of, or royalties on, our future products, if any.
Based on our current operating plan, we believe that our current cash, cash equivalents and investments together with the net proceeds we received from our follow-on public offering in January 2016 and receipt of anticipated milestone payments, will be sufficient to fund our projected operating requirements into the second half of 2019.2021. However, our operating plan may change as a result of many factors currently unknown to us, and we may need additional funds sooner than planned. In addition, we may seek additional capital due to favorable market conditions or strategic considerations even if we believe that we have sufficient funds for our current or future operating plans. Additional funds may not be available when we need them on terms that are acceptable to us, or at all. If adequate funds are not available to us on a timely basis, we may be required to delay, limit, reduce or terminate preclinical studies, clinical trials or other development activities for one or more of our therapeutic candidates or delay, limit, reduce or terminate our establishment of sales and marketing capabilities or other activities that may be necessary to commercialize our therapeutic candidates.
Raising additional capital may cause dilution to our existing stockholders, restrict our operations or require us to relinquish rights to our technologies or therapeutic candidates on terms unfavorable terms to us.
We may seek additional capital through a variety of means, including through private and public equity offerings and debt financings. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms may include liquidation or other preferences that adversely affect your rights as a stockholder. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take certain actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through strategic partnerships with third parties, we may have to relinquish valuable rights to our technologies or therapeutic candidates, or grant licenses on terms that are not favorable to us. If we are unable to raise additional funds through equity or debt financing when needed, we may be required to delay, limit, reduce or terminate our product development or commercialization efforts for dalantercept, ACE-083, sotatercept in the PH field, ACE-2494 or any therapeutic candidates other than luspatercept or sotatercept outside of the PH field, or grant rights to develop and market therapeutic candidates that we would otherwise prefer to develop and market ourselves.
Risks Related to Regulatory Review and Approval of Our Therapeutic Candidates
If we or our partners do not obtain regulatory approval for our current and future therapeutic candidates, our business will be adversely affected.
Our therapeutic candidates will be subject to extensive governmental regulations relating to, among other things, development, clinical trials, manufacturing and commercialization. In order to obtain regulatory approval for the commercial sale of any therapeutic candidates, we or our partners must demonstrate through extensive preclinical studies and clinical trials that the therapeutic candidate is safe and effective for use in each target indication. Clinical testing is expensive, time-consuming and uncertain as to outcome. We or our partners may gain regulatory approval for sotatercept, luspatercept, dalantercept, ACE-083, ACE-2494 or any other therapeutic candidate in some but not all of the territories available or some but not all of the target indications or may receive approval with limited labeling or boxed warnings, resulting in limited commercial opportunity for the approved therapeutic candidates, or we or they may never obtain regulatory approval for these therapeutic candidates.
Delays in the commencement, enrollment or completion of clinical trials of our therapeutic candidates could result in increased costs to us as well as a delay or failure in obtaining regulatory approval, or prevent us from commercializing our therapeutic candidates on a timely basis, or at all.
We cannot guarantee that clinical trials will be conducted as planned or completed on schedule, if at all. A failure of one or more clinical trials can occur at any stage of testing. Events that may prevent successful or timely commencement, enrollment or completion of clinical development include:
delays by us or our current or future partners in reaching a consensus with regulatory agencies on trial design;
delays in reaching agreement on acceptable terms with prospective clinical research organizations, or CROs, and clinical trial sites;
delays in obtaining required Institutional Review Board, or IRB, approval at each clinical trial site;
delays in recruiting suitable patients to participate in clinical trials;
imposition of a clinical hold by regulatory agencies for any reason, including safety or manufacturing concerns or after an inspection of clinical operations or trial sites;
failure by CROs, other third parties or us or our current or future partners to adhere to clinical trial requirements;
failure to perform in accordance with the FDA's good clinical practices, or GCP, or applicable regulatory guidelines in other countries;
delays in the testing, validation, manufacturing and delivery of the therapeutic candidates to the clinical sites;
delays caused by patients not completing participation in a trial or not returning for post-treatment follow-up;
clinical trial sites or patients dropping out of a trial;
occurrence of serious adverse events in clinical trials that are associated with the therapeutic candidates that are viewed to outweigh its potential benefits; or
changes in regulatory requirements and guidance that require amending or submitting new clinical protocols.
Delays, including delays caused by the above factors, can be costly and could negatively affect our or Celgene's ability to complete a clinical trial. If we or Celgene are not able to successfully complete clinical trials of our therapeutic candidates, we will not be able to obtain regulatory approval and will not be able to commercialize our therapeutic candidates.
There is a high risk of clinical failure at any stage of clinical development, and we may never succeed in developing marketable products or generating product revenue.
Our encouraging preclinical and clinical results to date for sotatercept, luspatercept, dalantercept and ACE-083, and our encouraging preclinical results to date for ACE-2494, are not necessarily predictive of the results of our ongoing or future clinical trials. Promising results in preclinical studies of a drug candidate may not be predictive of similar results in humans during clinical trials, and successful results from early clinical trials of a drug candidate may not be replicated in later and larger clinical trials or in clinical trials for different indications. If the results of our or our current or future partners' ongoing or future clinical trials are inconclusive with respect to the efficacy of our therapeutic candidates or if we or they do not meet the clinical endpoints with statistical significance or if there are safety concerns or adverse events associated with our therapeutic candidates, we or our partner may be prevented or delayed in obtaining marketing approval for our therapeutic candidates. We routinely develop our therapeutic candidates in multiple indications and our therapeutic candidates are at multiple stages of development at any given time, and any data, including safety or efficacy data, from any of our clinical trials could have a negative effect on our other clinical trials for the same or different therapeutic candidates. For example, if we or Celgene observe negative data in any one or more of our luspatercept clinical trials, this data could have an adverse effect on our ability to complete the MEDALIST or BELIEVE Phase 3 clinical trials or obtain regulatory approval for luspatercept in any indication.
In addition, data obtained from trials and studies are susceptible to varying interpretations, and regulators may not interpret our data as favorably as we do, which may delay or prevent regulatory approval. Alternatively, evenregulators may ask us to conduct more rigorous studies or trials or studies with endpoints that are different from the studies we are currently conducting. Even if we or our partners obtain regulatory approval, that approval may be for indications or patient populations that are not as broad as intended or desired or may require labeling that includes significant use or distribution restrictions or safety warnings. We or our partners may also be required to perform additional or unanticipated clinical trials to obtain approval or be subject to additional post-marketing testing requirements to maintain regulatory approval. In addition, regulatory authorities may withdraw their approval of a product or impose restrictions on its distribution, such as in the form of a risk evaluation and mitigation strategy.
If we or our current or future partners fail to obtain regulatory approval in jurisdictions outside the United States, we and they will not be able to market our products in those jurisdictions.
We and our current or future partners intend to market our therapeutic candidates, if approved, in international markets. Such marketing will require separate regulatory approvals in each market and compliance with numerous and varying regulatory requirements. The approval procedures vary from country-to-country and may require additional testing. Moreover, the time required to obtain approval may differ from that required to obtain FDA approval. In addition, in many countries outside the United States, a therapeutic candidate must be approved for reimbursement before it can be approved for sale in that country. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and
approval by one foreign regulatory authority does not ensure approval by regulatory authorities in other foreign countries or by the FDA. The foreign regulatory approval process may include all of the risks associated with obtaining FDA approval. We or our current or future partners may not obtain foreign regulatory approvals on a timely basis, if at all. We or our partners may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize our products in any market.
We and, for our Celgene partnered programs, Celgene, regularly request and receive guidance from the FDA and foreign regulators regarding the design or conduct of clinical trials with our therapeutic candidates. This guidance is not binding on these agencies and their policies and guidance could change substantially and unpredictably, potentially in a way that makes our clinical trials or our path to regulatory approval longer, more expensive or otherwise more difficult.
Any guidance that we or Celgene receive from the FDA or foreign regulators regarding the design or conduct of our or Celgene's clinical trials is not necessarily indicative of what these regulators will eventually require from us or Celgene to obtain regulatory approval of our therapeutic candidates. These regulators typically caution that any guidance received from them represents their then-current thinking, does not create or confer any rights to us or Celgene, and does not operate to bind the regulator. If later guidance that we or Celgene receive from the FDA or foreign regulators regarding our clinical trial design or conduct is materially different than the current guidance we have received from these regulators, we may need to change our clinical development plans and it may take longer, be more expensive or otherwise be more difficult to obtain FDA or foreign regulatory approval of our therapeutic candidates and our business may be materially harmed.
We undertake no obligation to disclose guidance that we or Celgene may receive from the FDA or foreign regulators. Any guidance from the FDA or foreign regulators that we may disclose publicly speaks only as of the date of such disclosure. We undertake no obligation to update any disclosure we make regarding regulator guidance to reflect additional regulatory guidance received after the date of such disclosure or to reflect the occurrence of unanticipated events that may affect the guidance.
Even if we or our current or future partners receive regulatory approval for our therapeutic candidates, such products will be subject to ongoing regulatory review, which may result in significant additional expense. Additionally, our therapeutic candidates, if approved, could be subject to labeling and other restrictions, and we or our current or future partners may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our products.
Any regulatory approvals that we or our current or future partners receive for our therapeutic candidates may also be subject to limitations on the approved indicated uses for which the product may be marketed or to conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase 4 clinical trials, and surveillance to monitor safety and efficacy. In addition, if the FDA approves any of our therapeutic candidates, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for the product will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with current good manufacturing practice, or cGMP, and GCP, for any clinical trials that we or our partners conduct post-approval.
Later discovery of previously unknown problems with an approved therapeutic candidate, including adverse events of unanticipated severity or frequency, or with manufacturing operations or processes, or failure to comply with regulatory requirements, may result in, among other things:
restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, or voluntary or mandatory product recalls;
fines, warning letters, or holds on clinical trials;
refusal by the FDA to approve pending applications or supplements to approved applications filed by us or our partners, or suspension or revocation of product license approvals;
product seizure or detention, or refusal to permit the import or export of products; and
injunctions or the imposition of civil or criminal penalties.
The FDA's policies of the FDA and other regulatory agencies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our therapeutic candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we or our partners are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or not able to
maintain regulatory compliance, we or our partners may lose any marketing approval that may have been obtained and we may not achieve or sustain profitability, which would adversely affect our business.
A Fast Track designation by the FDA may not actually lead to a faster development or regulatory review or approval process.
In the United States, luspatercept received Fast Track designation for the treatment of anemia in patients with lower-risk myelodysplastic syndromes, or MDS, for the treatment of patients with transfusion-dependent beta-thalassemia, and for the treatment of patients with non-transfusion-dependent beta-thalassemia. The FDA grants Fast Track designation to therapies that are considered capable of addressing unmet medical needs and possess the potential to treat serious or life-threatening disease conditions in order to facilitate its development and expedite the review procedure. The FDA has broad discretion in granting Fast Track designation, so even if we believe that a particular product candidate is eligible for such designation, the FDA could decide not to grant it. Even though luspatercept has received Fast Track designation, for multiple indications, we may not experience a faster development process, review or approval compared to conventional FDA procedures. The FDA may also withdraw Fast Track designation if it believes that the designation is no longer supported by data from our clinical development program.
The Phase 3 MEDALIST and BELIEVE clinical trials may be delayed, suspended or terminated, or may not lead to marketing approval.
If we or any of our current or future partners violate the guidelines pertaining to promotion and advertising of any of our therapeutic candidates, if approved, we or they may be subject to disciplinary action by the FDA's Office of Prescription Drug Promotion (OPDP) or other regulatory authorities.
The FDA's Office of Prescription Drug Promotion, or OPDP, is responsible for reviewing prescription drug advertising and promotional labeling to ensure that the information contained in these materials is not false or misleading. There are specific disclosure requirements, and the applicable regulations mandate that advertisements cannot be false or misleading or omit material facts about the product. Prescription drug promotional materials must present a fair balance between the drug's effectiveness and the risks associated with its use. Most warning letters from OPDP cite inadequate disclosure of risk information.
OPDP prioritizes its actions based on the degree of risk to the public health, and often focuses on newly introduced drugs and those associated with significant health risks. There are two types of letters that OPDP typically sends to companies that violate its drug advertising and promotional guidelines: notice of violation letters, or untitled letters and warning letters. In the case of an untitled letter, OPDP typically alerts the drug company of the violation and issues a directive to refrain from future violations, but does not
typically demand other corrective action. A warning letter is typically issued in cases that are more serious or where the company is a repeat offender. Although we have not received any such letters from OPDP, we or any partnerof our current or future partners may inadvertently violate OPDP's guidelines in the future and be subject to a OPDP untitled letter or warning letter, which may have a negative impact on our business.
Risks Related to Our Reliance on Third Parties
We are dependent on Celgene for the successful development and commercialization of sotatercept and our most advanced therapeutic candidate, luspatercept. If Celgene does not devote sufficient resources to the development of these candidates,luspatercept, discontinues development of these candidates,luspatercept, is unsuccessful in its efforts, or chooses to terminate its agreementsthe luspatercept agreement with us, our business will be materially harmed.
We have entered into collaboration agreements with Celgene to develop and commercialize sotatercept and luspatercept. Pursuant to the sotatercept agreement, responsibility for all clinical and other product development activities for all indications other than for pulmonary hypertension and for manufacturing sotatercept has been transferred to Celgene. For luspatercept, we are responsible for conducting ongoing Phase 2 clinical trials in MDS and beta-thalassemia, and we are also responsible for manufacturing supplies for our Phase 1 and Phase 2 studies. Celgene will be responsible for all clinical development and manufacturing activities after such studies are completed. As of January 1, 2013, Celgene becameis responsible for paying 100% of worldwide development costs for luspatercept and for sotatercept and luspatercept.outside of pulmonary hypertension. We will co-promote luspatercept and sotatercept and luspatercept,outside of pulmonary hypertension, if approved by the FDA and its counterparties, in North America. Celgene will be responsible for all commercialization costs, including the cost of our promotion activities.
Celgene is obligated to use commercially reasonable efforts to develop and commercialize luspatercept and sotatercept and luspatercept.outside of pulmonary hypertension. Celgene may determine that it is commercially reasonable to develop and commercialize only luspatercept or sotatercept and discontinue the development or commercialization of the other therapeutic candidate, or Celgene may determine that it is not commercially reasonable to continue development of one or both of sotatercept and luspatercept. For example, Celgene may elect not to undertake the development of sotatercept for the treatment of pre-dialysis patients. This may occur for many reasons, including internal business reasons or because of unfavorable regulatory feedback. Further, on review of the safety and efficacy data available to date, the FDA may impose requirements on thea clinical trial program that would render such athe program commercially nonviable. In the event of any such decision, we would be unable to progress sotatercept for this or other indicationsadvance such program ourselves. In addition, under
Under our collaboration agreements, oncewhen Celgene takes over development activities of a therapeutic candidate, it may determine the development plan and activities for that therapeutic candidate. We may disagree with Celgene about the development strategy it employs, but we will have no rights to impose our development strategy on Celgene. Similarly, Celgene may decide to seek regulatory approval for, and limit commercialization of, either or both of sotatercept outside of pulmonary hypertension and luspatercept to narrower indications than we would pursue. We would be prevented from developing or commercializing a candidate in an indication that Celgene has chosen not to pursue.pursue, other than sotatercept in pulmonary hypertension. More broadly, if Celgene elects to discontinue the development of both sotatercept andoutside of pulmonary hypertension and/or luspatercept, we may be unable to advance the products ourselves.
Pursuant to our collaboration agreement with Celgene for the development of sotatercept in pulmonary hypertension, we will rely on Celgene for many aspects of our development of sotatercept in pulmonary hypertension, including, for example, drug supply, manufacturing (if elected by Celgene), and regulatory support. If Celgene fails to perform its obligations under the sotatercept agreement, our ability to develop sotatercept in pulmonary hypertension may be materially harmed.
This partnership may not be scientifically or commercially successful due to a number of important factors, including the following:
Celgene has wide discretion in determining the efforts and resources that it will apply to its partnership with us. The timing and amount of any development milestones, and downstream commercial milestones and royalties that we may receive under such partnership will depend on, among other things, the efforts, allocation of resources and successful development and commercialization of these therapeutic candidates by Celgene.
Celgene may develop and commercialize, either alone or with others, products that are similar to or competitive with the therapeutic candidates that are the subject of its partnerships with us. For example, Celgene is currently commercializing and/or developing certain of its existing products, lenalidomide and azacitidine, for certain MDS patients for which luspatercept is also being developed.
Celgene may terminate its partnership with us without cause and for circumstances outside of our control, which could make it difficult for us to attract new strategic partners or adversely affect how we are perceived in scientific and financial communities.
Celgene may develop or commercialize our therapeutic candidates in such a way as to elicit litigation that could jeopardize or invalidate our intellectual property rights or expose us to potential liability.
Celgene may not comply with all applicable regulatory requirements, or may fail to report safety data in accordance with all applicable regulatory requirements.
If Celgene were to breach its arrangements with us, we may need to enforce our right to terminate the agreement in legal proceedings, which could be costly and cause delay in our ability to receive rights back to the relevant therapeutic candidates. If we were to terminate an agreement with Celgene due to Celgene's breach or Celgene terminated the agreement without cause, the development and commercialization of sotatercept and luspatercept could be delayed, curtailed or terminated because we may not have sufficient financial resources or capabilities to continue
development and commercialization of these candidates on our own if we choose not to, or are unable to, enter into a new collaboration for these candidates.
Celgene may enter into one or more transactions with third parties, including a merger, consolidation, reorganization, sale of substantial assets, sale of substantial stock or other change in control, which could divert the attention of its management and adversely affect Celgene's ability to retain and motivate key personnel who are important to the continued development of the programs under the strategic partnership with us. In addition, the third-party to any such transaction could determine to reprioritize Celgene's development programs such that Celgene ceases to diligently pursue the development of our programs and/or cause the respective partnership with us to terminate.
We and Celgene routinely publicly disclose information about luspatercept and sotatercept, and if our and Celgene’s public disclosures are inconsistent, this could materially harm public and market perception of luspatercept and sotatercept and the value of our common stock.
Through a variety of public forums and media, such as press releases, conference calls, filings with the Securities and Exchange Commission, and scientific/medical conferences, we and Celgene routinely disclose information about luspatercept and sotatercept. We and Celgene may not agree on information that is disclosed or the interpretation of that information, and our disclosures may be inconsistent with Celgene's disclosures. If our and Celgene's public disclosures are inconsistent, public and market perception of luspatercept and sotatercept and the value of our common stock could be materially adversely affected.
We currently have limited marketing, sales and distribution experience and capabilities and, for luspatercept and, outside of pulmonary hypertension, sotatercept, we will be dependent upon Celgene to commercializefor commercialization.
For luspatercept and, outside of pulmonary hypertension, sotatercept, outside the United States.
We and Celgene rely on third parties to conduct preclinical studies and clinical trials for our therapeutic candidates, and if they do not properly and successfully perform their obligations to us, we may not be able to obtain regulatory approvals for our therapeutic candidates.
We design the clinical trials for dalanterceptACE-083 and ACE-083sotatercept outside of pulmonary hypertension and will do so for any future unpartnered therapeutic candidates such as ACE-2494, and we will continue to work with Celgene on any ongoing or future trials for sotatercept and luspatercept. However, we and Celgene rely on CROs and other third parties to assist in managing, monitoring and otherwise carrying out many of these trials. We and Celgene compete with many other companies for the resources of these third parties. The third parties on whom we and Celgene rely generally may terminate their engagements at any time, and having to enter into alternative arrangements would delay development and commercialization of our therapeutic candidates.
The FDA and foreign regulatory authorities require compliance with regulations and standards, including GCP, for designing, conducting, monitoring, recording, analyzing, and reporting the results of clinical trials to assure that the data and
results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. Although we and Celgene rely on third parties to conduct many of our and their clinical trials, we and Celgene are responsible for ensuring that each of these clinical trials is conducted in accordance with its general investigational plan, protocol and other requirements.
If these third parties do not successfully carry out their duties under their agreements, if the quality or accuracy of the data they obtain is compromised due to their failure to adhere to clinical trial protocols or to regulatory requirements, or if they otherwise fail to comply with clinical trial protocols or meet expected deadlines, the clinical trials of our therapeutic candidates may not meet regulatory requirements or may be delayed. If clinical trials do not meet regulatory requirements or if these third parties need to be replaced, preclinical development activities or clinical trials may be extended, delayed, suspended or terminated. If any of these events occur, we or Celgene may not be able to obtain regulatory approval of our therapeutic candidates on a timely basis or at all.
Manufacturing biologic drugs is complicated and is tightly regulated by the FDA, the European Medicines Agency, or EMA, and comparable regulatory authorities around the world. We currently manufacture drug substance for our preclinical studies, Phase 1 clinical trials and Phase 2 clinical trials of luspatercept, dalantercept, ACE-083 and ACE-2494. For Phase 3 and commercial supply of our products that we have not partnered, we expect to use contract manufacturing organizations. Successfully transferring complicated manufacturing techniques to contract manufacturing organizations and scaling up these techniques for commercial quantities will be time consuming and we may not be able to achieve such transfer. Moreover, the market for contract manufacturing services for therapeutic candidates is highly cyclical, with periods of relatively abundant capacity alternating with periods in which there is little available capacity. If any need we have for contract manufacturing services increases during a period of industry-wide tight capacity, we may not be able to access the required capacity on a timely basis or on commercially viable terms.
In addition, we contract with fill & finishing providers with the appropriate expertise, facilities and scale to meet our needs. Failure to maintain cGMP can result in a contractor receiving FDA sanctions, which can impact our contractors' ability to operate or lead to delays in our clinical development programs. We believe that our current fill & finish contractors are operating in accordance with cGMP, but we can give no assurance that FDA or other regulatory agencies will not conclude that a lack of compliance exists.agree. In addition, any delay in contracting for fill & finish services, or failure of the contract manufacturer to perform the services as needed, may delay clinical trials, registration and launches. Any such issues may have a substantial negative effect on our business.
For sotatercept and our most advanced therapeutic candidate, luspatercept, we rely on our collaboration partner Celgene to produce,manufacture, or contract for the productionmanufacture of, bulk drug substance and finished drug product for use in late stage clinical trials and for commercial supplies of any approved candidates.these product candidates, if approved. Any failure by Celgene or by third-parties with which Celgene contracts may delay or impair the ability to complete late stage clinical trials or commercialize either or both of sotatercept and luspatercept, if approved.
Celgene is now responsible for manufacturing sotatercept outside of pulmonary hypertension and luspatercept for future late-stageall Celgene-sponsored clinical trials.trials, and may elect to be responsible for manufacturing sotatercept for clinical trials in pulmonary hypertension. Celgene generally does not perform the manufacture of the drug substance or drug product for either sotatercept or luspatercept itself. Celgene has used and may continue to use contract manufacturers for the manufacture of drug substance and drug product for sotatercept and we have no expectation that Celgene plans to perform the manufacture of bulk drug substance or drug product for either sotatercept or luspatercept in the future. However, Celgene would have the right to manufacture sotatercept or luspatercept, itself or through the use of contract manufacturers. We understand that they haveCelgene has entered into manufacturing arrangements for clinical and commercial supplies of sotatercept and luspatercept bulk drug substance with contract manufacturers with considerable biotherapeutics manufacturing experience, including manufacturing monoclonal antibodies through processes similar to those used for sotatercept. If any of these manufacturers is unwilling or unable to manufacture sufficient quantities of sotatercept and/or luspatercept to meet clinical or commercial demand, either for technical or business reasons, the development and commercialization of sotatercept and/or luspatercept may be delayed.
We may not be successful in establishing and maintaining additional strategic partnerships, which could adversely affect our ability to develop and commercialize products, negatively impacting our operating results.
In addition to our current collaborations with Celgene, part of our strategy is to evaluate and enter into additional partnerships in the future for our other product candidates when strategically attractive, including potentially with major biotechnology or pharmaceutical companies. We face significant competition in seeking appropriate partners for our therapeutic
candidates, and the negotiation process is time-consuming and complex. In order for us to successfully partner our therapeutic candidates, potential partners must view these therapeutic candidates as economically valuable in markets they determine to be attractive in light of the terms that we are seeking and other available products for licensing by other companies. Even if we are successful in our efforts to establish new strategic partnerships, the terms that we agree upon may not be favorable to us, and we may not be able to maintain such strategic partnerships if, for example, development or approval of a therapeutic candidate is delayed or sales of an approved product are disappointing. Any delay in entering into new strategic partnership agreements related to our other therapeutic candidates could delay the development and commercialization of these therapeutic candidates and reduce their competitiveness even if they reach the market.
If we fail to establish and maintain additional strategic partnerships related to dalantercept, ACE-083, ACE-2494 or other therapeutic candidates, we will bear all of the risk and costs related to the development of any such therapeutic candidate, and we may need to seek additional financing, hire additional employees and otherwise develop expertise for which we have not budgeted. This could negatively affect the development of any unpartnered therapeutic candidate.
Risks Related to Our Intellectual Property
If we are unable to obtain or protect intellectual property rights related to our therapeutic candidates, we may not be able to compete effectively.
We rely upon a combination of patents, trade secret protection and confidentiality agreements to protect the intellectual property related to our platform technology and therapeutic candidates. The patent position of biotechnology companies is generally uncertain because it involves complex legal and factual considerations. The standards applied by the United States Patent and Trademark Office, or USPTO, and foreign patent offices in granting patents are not always applied uniformly or predictably. For example, there is no uniform worldwide policy regarding patentable subject matter or the scope of claims allowable in biotechnology patents. The patent applications that we own or in-license may fail to result in issued patents with claims that cover our therapeutic candidates in the United States or in other countries. There is no assurance that all potentially relevant prior art relating to our patents and patent applications has been found. We may be unaware of prior art that could be used to invalidate an issued patent or prevent our pending patent applications from issuing as patents. Even if patents do
successfully issue and even if such patents cover our therapeutic candidates, third parties may challenge their validity, enforceability or scope, which may result in such patents being narrowed or invalidated. Furthermore, even if they are unchallenged, our patents and patent applications may not adequately protect our intellectual property, provide exclusivity for our therapeutic candidates or prevent others from designing around our claims. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
If patent applications we hold or have in-licensed with respect to our platform or therapeutic candidates fail to issue, if their breadth or strength of protection is threatened, or if they fail to provide meaningful exclusivity for our therapeutic candidates, it could dissuade companies from collaborating with us. SeveralWe regularly file patent applications coveringto cover our therapeutic candidates have been filed recently.candidates. We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patents or whether any issued patents will be found invalid and unenforceable or will be threatened by third parties. Any successful challenge to these patents or any other patents owned by or licensed to us could deprive us of rights necessary for the successful commercialization of any therapeutic candidate that we or our current or future partners may develop. Since patent applications in the United States and most other countries are confidential for a period of time after filing, and some remain so until issued, we cannot be certain that we were the first to file any patent application related to a therapeutic candidate. Furthermore, if third parties have filed such patent applications, an interference proceeding in the United States can be initiated by the USPTO or a third party to determine who was the first to invent any of the subject matter covered by the patent claims of our applications. In addition, patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years after it is filed. Various extensions may be available; however, the life of a patent and the protection it affords is limited. If we encounter delays in obtaining regulatory approvals, the period of time during which we could market a therapeutic candidate under patent protection could be reduced. Even if patents covering our therapeutic candidates are obtained, once the patent life has expired for a product, we may be open to competition from biosimilar products.
Any loss of patent protection could have a material adverse impact on our business. We and our current or future partners may be unable to prevent competitors from entering the market with a product that is similar to or the same as our therapeutic candidates. In addition, the royalty we would receive under our collaboration agreements with Celgene for sotatercept and luspatercept would be reduced by 50% if such product ceases to be covered by a valid claim of our patents even if no competitor with a similar product has entered the market.
Third-party claims of intellectual property infringement or misappropriation may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on us and our current or future partners not infringing the patents and proprietary rights of third parties. There is a substantial amount of litigation, both within and outside the United States, involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, oppositions and inter partes reexamination proceedings before the USPTO and corresponding foreign patent offices. Numerous U.S. and foreign issued patents and pending patent applications owned by third parties exist in the fields in which we and our current or future partners are developing and may develop our therapeutic candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our therapeutic candidates may be subject to claims of infringement of the patent rights of third parties.
Third parties may assert that we are employing their proprietary technology without authorization. There may be third-party patents or patent applications with claims to materials, formulations, methods of manufacture or methods for treatment related to the use or manufacture of our therapeutic candidates, that we failed to identify. For example, applications filed before November 29, 2000 and certain applications filed after that date that will not be filed outside the United States remain confidential until issued as patents. Except for the preceding exceptions, patent applications in the United States and elsewhere are generally published only after a waiting period of approximately 18 months after the earliest filing. Therefore, patent applications covering our platform technology or our therapeutic candidates could have been filed by others without our knowledge. Additionally, pending patent applications which have been published can, subject to certain limitations, be later amended in a manner that could cover our platform technologies, our therapeutic candidates or the use or manufacture of our therapeutic candidates.
If any third-party patents were held by a court of competent jurisdiction to cover aspects of our materials, formulations, methods of manufacture or methods for treatment, the holders of any such patents would be able to block our ability to develop and commercialize the applicable therapeutic candidate until such patent expired or unless we or our partners obtain a license. These licenses may not be available on acceptable terms, if at all. Even if we or our partners were able to obtain a license, the rights may be nonexclusive, which could result in our competitors gaining access to the same intellectual property. Ultimately, we or our partners could be prevented from commercializing a product, or be forced to cease some aspect of our business operations, if, as a result of actual or threatened patent infringement claims, we or our partners are unable to enter into licenses on acceptable terms. If Celgene is required to enter a license agreement with a third party in order to import, develop,
manufacture or commercialize sotatercept or luspatercept, the royalty rate and sales milestone payments that we could receive may be reduced by up to 50%. This could harm our business significantly.
Parties making claims against us or our partners may obtain injunctive or other equitable relief, which could effectively block our or our partners' ability to further develop and commercialize one or more of our therapeutic candidates. Defending against claims of patent infringement or misappropriation of trade secrets could be costly and time consuming, regardless of the outcome. Thus, even if we were to ultimately prevail, or to settle at an early stage, such litigation could burden us with substantial unanticipated costs. In addition, litigation or threatened litigation could result in significant demands on the time and attention of our management team, distracting them from the pursuit of other company business. In the event of a successful claim of infringement against us or our partners, we may have to pay substantial damages, including treble damages and attorneys' fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
We may face a claim of misappropriation if a third party believes that we inappropriately obtained and used trade secrets of such third party. If we are found to have misappropriated a third party's trade secrets, we may be prevented from further using such trade secrets, limiting our ability to develop our therapeutic candidates, and we may be required to pay damages.
During the course of any patent or other intellectual property litigation, there could be public announcements of the results of hearings, rulings on motions, and other interim proceedings in the litigation. If securities analysts or investors regard these announcements as negative, the perceived value of our therapeutic candidates, programs, or intellectual property could be diminished. Accordingly, the market price of our common stock could decline.
We have in-licensed, and may in the future in-license, a portion of our intellectual property, and, if we fail to comply with our obligations under these arrangements, we could lose such intellectual property rights or owe damages to the licensor of such intellectual property.
We are a party to a number of license agreements that are important to our business, and we may enter into additional license agreements in the future. OurAs part of our discovery and development platform is built, in part, around patents exclusively in-licensedactivities, we routinely evaluate in-licenses from academic orand research institutions. Certain of our in-licensed intellectual property also covers sotatercept and dalantercept. See "Business—"Business–Intellectual Property—Property–In-Licenses" for a description of our license agreements with the Beth Israel Deaconess Medical Center, the Ludwig Institute for Cancer Research and the Salk Institute for Biological Studies.agreements.
Our existing license agreements impose, and we expect that future license agreements will impose, various diligence, milestone payment, royalty and other obligations on us. If there is any conflict, dispute, disagreement or issue of non-performance between us and our licensing partners regarding our rights or obligations under the license agreements, including any such conflict, dispute or disagreement arising from our failure to satisfy payment obligations under any such agreement, we may owe damages, our licensor may have a right to terminate the affected license, and our and our partners' ability to utilize the affected intellectual property in our drug discovery and development efforts, and our ability to enter into collaboration or marketing agreements for an affected therapeutic candidate, may be adversely affected.
Confidentiality agreements with employees and third parties may not prevent unauthorized disclosure of trade secrets and other proprietary information.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce and any other elements of our platform technology and discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. However, trade secrets can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, and outside scientific advisors, contractors and collaborators. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, or outside scientific advisors might intentionally or inadvertently disclose our trade secret information to competitors. In addition, competitors may otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques.
Enforcing a claim that a third party illegally obtained and is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States sometimes are less willing than U.S. courts to protect trade secrets. Misappropriation or unauthorized disclosure of our trade secrets could impair our competitive position and may have a material adverse effect on our business.
Risks Related to Development and Commercialization of Our Therapeutic Candidates
Our future commercial success depends upon attaining significant market acceptance of our therapeutic candidates, if approved, among physicians, patients, healthcare payers and acceptance by the operators of major medical providers.
Even if we or our current or future partners obtain regulatory approval for sotatercept, luspatercept, dalantercept, ACE-083any of our existing therapeutic candidates or any other therapeutic candidates that we may develop or acquire in the future, the product may not gain market acceptance among physicians, healthcare payers, patients and the medical community. Market acceptance of any approved products depends on a number of factors, including:
the efficacy and safety of the product, as demonstrated in clinical trials;
the clinical indications for which the product is approved and the label approved by regulatory authorities for use with the product, including any warnings that may be required on the label;
acceptance by physicians and patients of the product as a safe and effective treatment;
decisions by healthcare organizations to utilize the product;
the cost, safety and efficacy of treatment in relation to alternative treatments;
the availability of adequate reimbursement and pricing by third party payers and government authorities;
the continued projected growth of drug markets in our various indications;
relative convenience and ease of administration;
the prevalence and severity of adverse side effects; and
the effectiveness of our and our current or future partners' sales and marketing efforts.
Market acceptance is critical to our ability to generate significant revenue. Any therapeutic candidate, if approved and commercialized, may be accepted in only limited capacities or not at all. If any approved products are not accepted by the market to the extent that we expect, we may not be able to generate significant revenue and our business would suffer.
Reimbursement may be limited or unavailable in certain market segments for our therapeutic candidates, which could make it difficult for us to sell our products profitably.
Market acceptance and sales of any approved therapeutic candidates will depend significantly on the availability of adequate coverage and reimbursement from third-party payers and may be affected by existing and future healthcare reform measures. Government authorities and third-party payers, such as private health insurers and health maintenance organizations, decide which drugs they will pay for and establish reimbursement levels. Reimbursement by a third-party payer may depend upon a number of factors, including the third-party payer's determination that use of a product is:
a covered benefit under its health plan;
safe, effective and medically necessary;
appropriate for the specific patient;
cost-effective; and
neither experimental nor investigational.
Obtaining coverage and reimbursement approval for a product from a government or other third party payer is a time consuming and costly process that could require us to provide supporting scientific, clinical and cost-effectiveness data for the use of our products to the payer. We or our current or future partners may not be able to provide data sufficient to gain acceptance with respect to coverage and reimbursement. We cannot be sure that coverage or adequate reimbursement will be available for any of our therapeutic candidates. Also, we cannot be sure that reimbursement amounts will not reduce the demand for, or the price of, our products. If reimbursement is not available or is available only to limited levels, we may not be able to commercialize certain of our products. In addition in the United States, third-party payers are increasingly attempting to contain healthcare costs by limiting both coverage and the level of reimbursement of new drugs. As a result, significant uncertainty exists as to whether and how much third-party payers will reimburse patients for their use of newly approved drugs, which in turn will put pressure on the pricing of drugs.
Price controls and price pressure may be imposed in foreign and U.S. markets, which may adversely affect our future profitability.
In some countries, particularly member states of the European Union, the pricing of prescription drugs is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after receipt of marketing approval for a product. In addition, there can be considerable pressure by governments and other stakeholders, including those in the United States, on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various European Union member states and parallel distribution, or arbitrage between low-priced and high-priced member states, can further reduce prices. In some countries, we or our current or future partners may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our therapeutic candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payers or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be adversely affected.
Recent and future healthcare reform legislation and other changes in the healthcare industry and in healthcare spending may adversely affect our business model.
Our revenue prospects could be affected by changes in healthcare spending and policy in the United States and abroad. We operate in a highly regulated industry and new laws, regulations or judicial decisions, or new interpretations of existing laws, regulations or decisions, related to healthcare availability, the method of delivery or payment for healthcare products and services could negatively impact our business, operations and financial condition. There is significant interest in promoting healthcare reform, as evidenced by the enactment in the United States of the Patient Protection and Affordable Care Act and the Health Care and Education Reconciliation Act in 2010.2010, and as evidenced by public discourse on the topic. See "Business–Government Regulation" for a detailed description on recent developments in healthcare regulations. It is likely that federal and state legislatures within the United States and foreign governments will continue to consider changes to existing healthcare legislation. We cannot predict the reform initiatives that may be adopted in the future or whether initiatives that have been adopted will be repealed or modified. The continuing efforts of the government, insurance companies, managed care organizations and other payers of healthcare services to contain or reduce costs of healthcare may adversely affect:
public and market perceptions of our future business prospects, including future revenue and profitability;
the demand for any drug products for which we may obtain regulatory approval;
our ability to set a price that we believe is fair for our products;
our ability to obtain coverage and reimbursement approval for a product;
our ability to generate revenues and achieve or maintain profitability; and
the level of taxes that we are required to pay.
We face substantial competition, which may result in others discovering, developing or commercializing products before, or more successfully, than we do.
Our future success depends on our or our partners' ability to demonstrate and maintain a competitive advantage with respect to the design, development and commercialization of our therapeutic candidates. Our objective is to design, develop and commercialize new products with superior efficacy, convenience, tolerability and safety. In many cases, the therapeutic candidates that we commercialize with our strategic partners or on our own will compete with existing, market-leading products. See "Business–Competition" for a detailed description of the competitive landscape for our therapeutic candidates.
There are products currently approved to treat patients with MDS, including iron chelation therapy, immunomodulators and various chemotherapeutic agents. In addition, erythropoiesis stimulating agents and red blood cell transfusions are extensively used to treat anemia in MDS. Luspatercept, or sotatercept, if approved, will compete with these therapies. In addition, one or more products not currently approved for the treatment of anemia in MDS may in the future be granted marketing approval for the treatment of anemia in MDS or other conditions for which luspatercept or sotatercept might be approved, including Aranesp®, being developed by Amgen, which is in Phase 3 trials.approved. While there are currently no drug products approved for the treatment of anemia in beta-thalassemia, red blood cell transfusions are extensively used and luspatercept, if approved, would compete with this therapy. Further, the future approval, in one or more regions, of a biosimilar product to one of our products could create substantial competition and have a material impact on our business. In addition, the success of gene and/or cell therapy in beta-thalassemia patients could materially reduce the potential patient population for luspatercept, especially in transfusion dependent patients.
If ACE-083 is approved for the treatment of neuromuscular disorders or other diseases characterized by a loss of muscle function, it could compete with a variety of other approaches to treating neuromuscular disorders or muscle loss that are currently in clinical trials, including, among others, a monoclonal antibody targeting the activin receptor type IIB, bimagrumab, being studied by Novartis to treat pathological muscle loss and weakness, and various myostatin monoclonal antibodies being studied to treat disuse muscle atrophy, cancer-related cachexia, and sarcopenia. We intend to conduct our first phaseare currently conducting Phase 2 trial ofclinical trials with ACE-083 in patients with facioscapulohumeral muscular dystrophy, or FSHD. We are aware of competitors also developing products to treat thisFSHD, as well as in patients with Charcot-Marie-Tooth disease, including aTyr Pharma.or CMT. If competitive products prove to be superior to ACE-083, our business may be harmed. (See "Competition").
Many of our potential competitors have significantly greater financial, manufacturing, marketing, drug development, technical and human resources than we do. Large pharmaceutical companies, in particular, have extensive experience in clinical testing, obtaining regulatory approvals, recruiting patients and in manufacturing pharmaceutical products. These companies also have significantly greater research and marketing capabilities than we do and may also have products that have been approved or are in late stages of development, and have collaborative arrangements in our target markets with leading companies and research institutions. Established pharmaceutical companies may also invest heavily to accelerate discovery and development of novel compounds or to in-license novel compounds that could make the therapeutic candidates that we develop obsolete. As a result of all of these factors, our competitors may succeed in obtaining patent protection and/or FDA approval or discovering, developing and commercializing therapeutic candidates before we do. In addition, any new product that competes with an approved product must demonstrate compelling advantages in efficacy, convenience, tolerability and safety in order to overcome price competition and to be commercially successful. If we are not able to compete effectively against potential competitors, our business will not grow and our financial condition and operations will suffer.
If our clinical trials fail to demonstrate the safety and efficacy of our therapeutic candidates to the satisfaction of regulatory authorities or do not otherwise produce positive results, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our therapeutic candidates.
Undesirable side effects caused by our therapeutic candidates could cause us, Celgene or regulatory authorities to interrupt, delay or halt clinical trials and could result in the denial of regulatory approval by the FDA or other regulatory authorities and potential products liability claims. We and Celgene are currently conducting a number of clinical trials for our clinical stage therapeutic candidates. Serious adverse events deemed to be caused by our therapeutic candidates could have a
material adverse effect on the development of our therapeutic candidates and our business as a whole. In the case of placebo-controlled clinical trials, if the rate of an adverse event or serious adverse event observed in the treatment group exceeds the rate of such adverse event or serious adverse event observed in the placebo-controlled group, then the FDA or other regulatory authorities may require the delay or termination of such clinical trial, or require the conduct of one or more additional clinical trials to further demonstrate the safety of the therapeutic candidate.
For a more complete description of the safety profile for our therapeutic candidates, see the description of each of our therapeutic candidates in the "Business" section of this Annual Report on Form 10-K.
Our understanding of the relationship between our therapeutic candidates and these events may change as we gather more information, and additional unexpected adverse events may occur. There can be no assurance that additional adverse events associated with our therapeutic candidates will not be observed. As is typical in drug development, we have a program of ongoing toxicology studies in animals for our clinical stage therapeutic candidates and cannot provide assurance that the findings from such studies or any ongoing or future clinical trials will not adversely affect our clinical development activities.
Before obtaining marketing approval from regulatory authorities for the sale of our therapeutic candidates, we must complete preclinical development and then conduct extensive clinical trials to demonstrate the safety and efficacy of our therapeutic candidates in humans. Clinical testing is expensive, difficult to design and implement, can take many years to complete and is uncertain as to outcome. A failure of one or more clinical trials can occur at any stage of testing. The outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results. In addition, preclinical and clinical data are often susceptible to varying interpretations and analyses. Many companies that have believed their therapeutic candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless failed to obtain marketing approval of their products.
We or our current or future partners may experience numerous unforeseen events during, or as a result of, clinical trials that could delay or prevent our ability to receive marketing approval or commercialize our therapeutic candidates, including:
regulators or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site;
we may have delays in reaching or fail to reach agreement on acceptable clinical trial contracts or clinical trial protocols with prospective trial sites;
clinical trials of our therapeutic candidates may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical trials or abandon product development programs;
the number of patients required for clinical trials of our therapeutic candidates may be larger than we anticipate; enrollment in these clinical trials may be slower than we anticipate; or participants may drop out of these clinical trials at a higher rate than we anticipate;
third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all;
we might have to suspend or terminate clinical trials of our therapeutic candidates for various reasons, including a finding that the participants are being exposed to unacceptable health risks;
regulators, institutional review boards, or the data safety monitoring board for such trials may require that we or our investigators suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health risks;
the cost of clinical trials of our therapeutic candidates may be greater than we anticipate;
the supply or quality of our therapeutic candidates or other materials necessary to conduct clinical trials of our therapeutic candidates may be insufficient or inadequate; and
our therapeutic candidates may have undesirable side effects or other unexpected characteristics, causing us or our investigators, regulators or institutional review boards to suspend or terminate the trials.
If we or our current or future partners are required to conduct additional clinical trials or other testing of our therapeutic candidates beyond those that we currently contemplate, if we or our current or future partners are unable to successfully complete clinical trials of our therapeutic candidates or other testing, if the results of these trials or tests are not positive or are only modestly positive or if we or others identify undesirable side effects caused by our therapeutic candidates either before or after receipt of marketing approval, then a number of potentially significant negative consequences could result, including that we or our current or future partners may:
be delayed in obtaining or be unable to obtain marketing approval for our therapeutic candidates;
obtain approval for indications or patient populations that are not as broad as intended or desired;
be required to provide a medication guide outlining the risks of such side effects for distribution to patients;
obtain approval with labeling that includes significant use or distribution restrictions or safety warnings, including boxed warnings;
suffer reputational harm;
be sued and held liable for harm caused to patients;
be subject to additional post-marketing testing requirements; or
have the product removed from the market after obtaining marketing approval.
Product development costs will also increase if we or our current or future partners experience delays in testing or marketing approvals. We do not know whether any clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. Significant clinical trial delays also could shorten any periods during which we may have the exclusive right to commercialize our therapeutic candidates, could allow our competitors to bring products to market before we do, and could impair our ability to successfully commercialize our therapeutic candidates, any of which may harm our business and results of operations.
Our results to date do not guarantee that any of our therapeutic candidates will be safe or effective, or receive regulatory approval.
The risk of failure of our current therapeutic candidates is high. To date, the data supporting our clinical development strategy for our therapeutic candidates are derived solely from laboratory and preclinical studies and limited early-to-mid-stage clinical trials. Later clinical trials may not yield data consistent with earlier clinical trials. Similarly, clinical responses seen in patients enrolled at early stages of a clinical trial may not be replicated in patients enrolled in that trial at a later time. In addition, adverse events not observed in early clinical trials may be seen for the first time in later studies, or adverse events observed in a small number of patients in early trials may be seen in a greater number of patients in later studies and have greater statistical significance than previously anticipated. In the event that our clinical trials do not yield data consistent with earlier experience, it may be necessary for us to change our development strategy or abandon development of that therapeutic candidate, either of which could result in delays, additional costs and a decrease in our stock price.
Our Phase 2 clinical trial results for luspatercept are not necessarily indicative or predictive of the results of Celgene's Phase 3 clinical trials. It is impossible to predict when or if any of our therapeutic candidates will prove safe or effective in humans or receive regulatory approval. These therapeutic candidates may not demonstrate in patients the chemical and pharmacological properties ascribed to them in laboratory studies or early-to-mid-stage clinical trials, and they may interact with human biological systems or other drugs in unforeseen, ineffective or harmful ways. If we are unable to discover or successfully develop drugs that are safe and effective in humans, we will not have a viable business.
We manufacture ACE-083, ACE-2494 and earlier stagefor our Acceleron-sponsored trials, luspatercept, at our manufacturing facility. If our manufacturing facility is damaged or destroyed or production at this facility is otherwise interrupted, our business and prospects would be negatively affected.
If the manufacturing facility at our corporate headquarters or the equipment in it is damaged or destroyed, or if production at this facility is otherwise interrupted, we may not be able to quickly or economically replace our manufacturing capacity or replace it at all. In the event of a temporary or protracted loss of this facility or equipment, we might not be able to transfer manufacturing to a third party. Even if we could transfer manufacturing to a third party, the shift would likely be expensive and time-consuming, particularly since the new facility would need to comply with the necessary regulatory requirements and we
would need approval from the FDA and foreign regulators before administering any products manufactured at that facility to patients. Such an event could delay our clinical trials or, if our therapeutic candidates are approved by the FDA or foreign regulators, reduce our product sales.trials.
Our expanded research activities may not identify new therapeutic candidates, and we may not be successful in developing any new therapeutic candidates that are identified.
Discovery and development of new therapeutic candidates is an unpredictable activity. We may not succeed in identifying new therapeutic candidates, and if we are unable to do so, our pipeline of clinical stage therapeutic candidates will be reduced in size, potentially harming our business. Our discovery efforts are primarily focused on IntelliTrap™IntelliTrap™ therapeutic candidates and antibodies. We have not previously manufactured or developedlimited experience manufacturing and developing antibodies or IntelliTrap™and IntelliTrap™ proteins, and we may not be successful at doing so.so in the future. Manufacturing biologic therapeutic candidates is complex and often requires extensive process development to achieve suitable quality and quantity of drug substance for clinical development and commercialization, and we may not succeed in achieving the quality and quantity of drug substance that is required at any particular stage. We may be unable to manufacture these candidate therapeutics,therapeutic candidates, these candidate therapeuticstherapeutic candidates may show unacceptable toxicity or pharmacokinetic properties, or these therapeutic candidates may not be safe or effective in clinical trials.
Risks Related to Our Business and Industry
If we undertake business combinations, collaborations or similar strategic transactions, they may disrupt our business, divert management’s attention, dilute stockholder value or be difficult to integrate.
On a regular basis, we consider various business combination transactions, collaborations, license agreements and strategic transactions with third parties, including transactions which may result in us acquiring, or being acquired by, a third party. The consummation or performance of any future business combination, collaboration or strategic transaction may involve risks, such as:
diversion of managerial attention and resources from day-to-day operations;
challenges associated with integrating acquired technologies and operations of acquired companies;
exposure to unforeseen liabilities or increased regulatory and compliance obligations;
difficulties in the assimilation of different cultures and practices, as well as in the assimilation and retention of broad and geographically dispersed personnel and operations;
misjudgment with respect to value, return on investment or strategic fit;
higher than expected transaction costs; and
additional dilution to our existing stockholders if we issue equity securities as consideration for any acquisitions.
As a result of these risks, we may not be able to achieve the expected benefits of any such transaction. Future business combinations could involve the acquisition of significant intangible assets. We may need to record write-downs from future impairments of identified intangible assets and goodwill. These accounting charges would increase a reported loss or reduce any future reported earnings. In addition, we could use substantial portions of our available cash to pay the purchase price for company or product candidate acquisitions. We could also incur debt or issue additional equity securities as consideration for these acquisitions, which could cause our stockholders to suffer significant dilution.
If we fail to attract and keep senior management and key personnel, we may be unable to successfully develop our therapeutic candidates, conduct our clinical trials and commercialize our therapeutic candidates.
We are highly dependent on members of our senior management, including John L. Knopf, Ph.D., ourmanagement. We recently hired a new Chief ExecutiveCommercial Officer and President and oneare in the process of recruiting for a new Chief Medical Officer. If we fail to successfully recruit a new Chief Medical Officer or if we fail to successfully manage the transition of our founders. Thenew Chief Commercial Officer or Chief Medical Officer, our clinical and commercial operations could be harmed. In addition, the loss or transition of the services from any member of any of these personsour senior management could impede the achievement of our research, development andor commercialization objectives. We do not maintain "key person" insurance for any of our executives or other employees.
Recruiting and retaining qualified scientific, clinical, regulatory, manufacturing, sales and marketing personnel will also be critical to our success. We may not be able to attract and retain these personnel on acceptable terms given the competition
among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. In addition, we rely on consultants and advisors, including scientific and clinical advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities that may limit their availability to us.
We may encounter difficulties in managing our organizational changes and successfully adjusting our operations.
As we seek to advance our therapeutic candidates through clinical trials and commercialization, we will need to expand our development, regulatory, manufacturing, marketing and sales capabilities or contract with third parties to provide these capabilities for us. As our operations expand, we expect that we will need to manage additional relationships with various strategic partners, suppliers and other third parties. Future growth will impose significant added responsibilities on members of management. Our future financial performance and our ability to commercialize our therapeutic candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To that end, we must be able to manage our development efforts and clinical trials effectively and hire, train and integrate additional management, administrative and, if necessary, sales and marketing personnel. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing our company.
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit commercialization of our therapeutic candidates.
We face an inherent risk of product liability as a result of the clinical testing of our therapeutic candidates and will face an even greater risk if we commercialize any products. For example, we may be sued if any product we develop allegedly causes injury or is found to be otherwise unsuitable during product testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability, and a breach of warranties. Claims could also be asserted under state consumer protection acts. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our therapeutic candidates. Even a successful defense would require significant financial and management resources. Regardless of the merits or eventual outcome, liability claims may result in:
injury to our reputation;
withdrawal of clinical trial participants;
costs to defend the related litigations;
a diversion of management's time and our resources;
substantial monetary awards to trial participants or patients;
product recalls, withdrawals, or labeling, marketing or promotional restrictions;
loss of revenue;
the inability to commercialize our therapeutic candidates; and
a decline in our stock price.
Failure to obtain and retain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of products we develop. We currently carry product liability
insurance covering our clinical trials in the amount of $10.0 million in the aggregate.trials. Although we maintain such insurance, any claim that may be brought against us could result in a court judgment or settlement in an amount that is not covered, in whole or in part, by our insurance or that is in excess of the limits of our insurance coverage. Our insurance policies also have various exclusions, and we may be subject to a product liability claim for which we have no coverage. We will have to pay any amounts awarded by a court or negotiated in a settlement that exceed our coverage limitations or that are not covered by our insurance, and we may not have, or be able to obtain, sufficient capital to pay such amounts.
We could be subject to securities class action litigation.
In the past, securities class action litigation has often been brought against companies following a decline in the market price of their securities. This risk is especially relevant for us because biopharmaceutical companies have experienced significant stock price volatility in recent years. If we face such litigation, it could result in substantial costs and a diversion of management’s attention and resources, which could harm our business.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Under Section 382 of the Internal Revenue Code of 1986, as amended, if a corporation undergoes an “ownership change,” generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period, the corporation’s ability to use its pre-change net operating loss carryforwards, or NOLs, and other pre-change tax attributes (such as research tax credits) to offset its post-change income may be limited. We have completed several financings since our inception which may have resulted in a change in control as defined by IRC Section 382. We may also experience ownership changes in the future as a result of subsequent shifts in our stock ownership. As a result, if we earn net taxable income, our ability to use our pre-change net operating loss carryforwards to offset U.S. federal taxable income may be subject to limitations, which could potentially result in increased future tax liability to us. In addition, at the state level, there may be periods during which the use of NOLs is suspended or otherwise limited, which could accelerate or permanently increase state taxes owed.
We must comply with environmental laws and regulations, and failure to comply with these laws and regulations could expose us to significant liabilities.
We are subject to a variety of federal, state and local laws and regulations governing the use, generation, manufacture, distribution, storage, handling, treatment and disposal of materials that we use in our manufacturing process. Although we believe our safety procedures for handling and disposing of these materials and waste products comply with these laws and regulations, we cannot eliminate the risk of accidental injury or contamination from the use, manufacture, distribution, storage, handling, treatment or disposal of hazardous materials. In the event of contamination or injury, or failure to comply with environmental, occupational health and safety and export control laws and regulations, we could be held liable for any resulting damages and any such liability could exceed our assets and resources. We are uninsured for third-party contamination injury.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. A severe or prolonged economic downturn could result in a variety of risks to our business, including, weakened demand for our therapeutic candidates and our ability to raise additional capital when needed on acceptable terms, if at all. Weak global economic conditions, especially in Europe, could decrease the number of clinical trial sites available to us and hinder our ability to conduct clinical trials, which would have a material adverse effect on our business and the development of our therapeutic candidates. A weak or declining economy could also strain our suppliers, possibly resulting in supply disruption, or cause our customers to delay making payments for our services. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
Our internal computer systems, or those of our partners, third-party CROs or other contractors or consultants, may fail or suffer securitydata breaches and cyber-attacks, which could compromise our intellectual property or other sensitive information and could result in a material disruption of our therapeutic candidate development programs.
Our network and storage applications and those of our currentclinical research organizations, collaborators and vendors may be subject to unauthorized access by hackers or breached due to operator error, malfeasance or other system disruptions. It is often difficult to anticipate or immediately detect such incidents and the damage caused by such incidents. These data breaches and any future CROs and other contractors, consultants and collaborators are vulnerable to damage from computer viruses, unauthorized access natural disasters, terrorism, waror disclosure of our information or intellectual property could compromise our intellectual property and telecommunicationexpose sensitive business information. A data security breach could also lead to public exposure of personal information of
our employees. Cyber-attacks could cause us to incur significant remediation costs, result in product development delays, disrupt key business operations and electrical failures.divert attention of management and key information technology resources. Our network security and data recovery measures and those of our clinical research organizations, collaborators and vendors may not be adequate to protect against such security breaches and disruptions. These incidents could also subject us to liability, expose us to significant expense and cause significant harm to our reputation and business. While we have not experienced any such material system failure, accidentsecurity breaches or security breachdisruptions to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our or our partners' regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our therapeutic candidates and conduct clinical trials, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our or any of our partners' data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our therapeutic candidate could be delayed.
Risks Related to Our Common Stock
The market price of our common stock may be highly volatile, and you may not be able to resell your shares at or above the price at which you purchased them.
Since our initial public offering, in September 2013, the price of our common stock as reported on The NASDAQ Global Market has ranged from a low of $16.78 on November 6 and 8, 2013 to a high of $57.89 on January 22, 2014. The market price of shares of our common stock could be subject to wide fluctuations in response to many risk factors listed in this section, and others beyond our control, including:
results of clinical trials of our therapeutic candidates, including luspatercept, sotatercept, luspatercept, dalanterceptACE-083 and ACE-083;ACE-2494;
the timing of the release of results of our clinical trials that are being conducted by Celgene;
results of clinical trials of our competitors' products;
regulatory actions with respect to our products or our competitors' products;
actual or anticipated fluctuations in our financial condition and operating results;
publication of research reports by securities analysts about us or our competitors or our industry;
our failure or the failure of our competitors to meet analysts' projections or guidance that we or our competitors may give to the market;
additions and departures of key personnel;
strategic decisions by us or our competitors, such as acquisitions, divestitures, spin-offs, joint ventures, strategic investments or changes in business strategy;
the passage of legislation or other regulatory developments affecting us or our industry;
fluctuations in the valuation of companies perceived by investors to be comparable to us;
sales of our common stock by us, our insiders or our other stockholders;
speculation in the press or investment community;
announcement or expectation of additional financing efforts;
changes in accounting principles;
terrorist acts, acts of war or periods of widespread civil unrest;
natural disasters and other calamities;
actual or perceived changes in current or future market conditions for biopharmaceutical stocks; and
changes in general market and economic conditions.
In addition, the stock market has recently experienced significant volatility, particularly with respect to pharmaceutical, biotechnology and other life sciences company stocks. The volatility of pharmaceutical, biotechnology and other life sciences company stocks often does not relate to the operating performance of the companies represented by the stock. As we operate in a single industry, we are especially vulnerable to these factors to the extent that they affect our industry or our products, or to a lesser extent our markets. In the past, securities class action litigation has often been initiated against companies following periods of volatility in their stock price. This type of litigation could result in substantial costs and divert our management's attention and resources, and could also require us to make substantial payments to satisfy judgments or to settle litigation.
We continue to incur significant costs as a result of operating as a public company and complying with the Sarbanes-Oxley Act, especially now that we are a large accelerated filer and are no longer an ""emerging growth company,"" and our management continues to beis required to devote substantial time to compliance initiatives. If we fail to maintain an effective system of internal control over financial reporting in the future, we may not be able to accurately report our financial condition, results of operations or cash flows, which may adversely affect investor confidence in us and, as a result, the value of our common stock.
As a public company, we incur significant legal, accounting and other expenses that we did not incur as a private company.expenses. In addition, the Sarbanes-Oxley Act, and rules of the SEC and those of NASDAQ have imposed various requirements on public companies including requiring establishment and maintenance of effective disclosure and financial controls. Our management and other personnel devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations have increased and will continue to increase our legal and financial compliance costs and will make some activities more time-consuming and costly.
The Sarbanes-Oxley Act requires, among other things, that we maintain effective internal controls for financial reporting and disclosure controls and procedures. We are required, under Section 404 of the Sarbanes-Oxley Act, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting. This assessment must include disclosure of any material weaknesses identified by our management in our internal control over financial reporting. A material weakness is a control deficiency, or combination of control deficiencies, in internal control over financial reporting that results in more than a reasonable possibility that a material misstatement of annual or interim financial statements will not be prevented or detected on a timely basis. Section 404 of the Sarbanes-Oxley Act also generally requires an attestation fromIn addition, we are required to have our independent registered public accounting firm onattest to the effectiveness of our internal control over financial reporting. Now that we no longer qualify as an emerging growth company as defined in the JOBS Act, we are no longer exempted from certain requirements, such as the independent registered public accounting firm attestation.
During the evaluation and testing process, if we identify one or more material weaknesses in our internal control over financial reporting, we will be unable to assert that our internal control over financial reporting is effective. We cannot assure you that there will not be material weaknesses or significant deficiencies in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to accurately report our financial condition, results of operations or cash flows. If we are unable to conclude that our internal control over financial reporting is effective, or if our independent registered public accounting firm determines we have a material weakness or significant deficiency in our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, the market price of our common stock could decline, and we could be subject to sanctions or investigations by the NASDAQ, the SEC or other regulatory authorities. Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
We do not expect to pay any cash dividends for the foreseeable future.
You should not rely on an investment in our common stock to provide dividend income. We do not anticipate that we will pay any cash dividends to holders of our common stock in the foreseeable future. Instead, we plan to retain any earnings to maintain and expand our operations. In addition, any future debt financing arrangement may contain terms prohibiting or limiting the amount of dividends that may be declared or paid on our common stock. Accordingly, investors must rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any return on their investment. As a result, investors seeking cash dividends should not purchase our common stock.
Provisions in our restated certificate of incorporation, our amended and restated by-laws and Delaware law may have anti-takeover effects that could discourage an acquisition of us by others, even if an acquisition would be beneficial to our stockholders, and may prevent attempts by our stockholders to replace or remove our current management.
Our restated certificate of incorporation, amended and restated by-laws and Delaware law contain provisions that may have the effect of delaying or preventing a change in control of us or changes in our management. Our restated certificate of incorporation and by-laws include provisions that:
authorize "blank check" preferred stock, which could be issued by our board of directors without stockholder approval and may contain voting, liquidation, dividend and other rights superior to our common stock;
create a classified board of directors whose members serve staggered three-year terms;
specify that special meetings of our stockholders can be called only by our board of directors;
prohibit stockholder action by written consent;
establish an advance notice procedure for stockholder approvals to be brought before an annual meeting of our stockholders, including proposed nominations of persons for election to our board of directors;
provide that our directors may be removed only for cause;
provide that vacancies on our board of directors may be filled only by a majority of directors then in office, even though less than a quorum;
specify that no stockholder is permitted to cumulate votes at any election of directors;
expressly authorize our board of directors to modify, alter or repeal our amended and restated by-laws; and
require supermajority votes of the holders of our common stock to amend specified provisions of our restated certificate of incorporation and amended and restated by-laws
These provisions, alone or together, could delay or prevent hostile takeovers and changes in control or changes in our management.
In addition, we are subject to the provisions of Section 203 of the Delaware General Corporation Law, which prohibits a publicly-held Delaware corporation from engaging in a "business combination" with an "interested stockholder" for a three-year period following the time that this stockholder becomes an interested stockholder, unless the business combination is approved in a prescribed manner. As a result, you may lose your ability to sell your stock for a price in excess of the prevailing market price due to these protective measures and efforts by stockholders to change the direction or management of the company may be unsuccessful.
Any provision of our restated certificate of incorporation or amended and restated by-laws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock, and could also affect the price that some investors are willing to pay for our common stock.
Our restated certificate of incorporation designates the Court of Chancery of the State of Delaware and federal court within the State of Delaware as the exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, which could limit our stockholders' ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our restated certificate of incorporation provides that, subject to limited exceptions, the Court of Chancery of the State of Delaware and federal court within the State of Delaware will be exclusive forums for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or
other employees to us or our stockholders, (3) any action asserting a claim against us arising pursuant to any provision of the Delaware General Corporation Law, our restated certificate of incorporation or our amended and restated by-laws, or (4) any other action asserting a claim against us that is governed by the internal affairs doctrine. Any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock shall be deemed to have notice of and to have consented to the provisions of our restated certificate of incorporation described above. This choice of forum provision may limit a stockholder's ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and employees. Alternatively, if a court were to find these provisions of our restated certificate of incorporation inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could adversely affect our business and financial condition.
Sales of our common stock by our employees, including our executive officers, could cause our stock price to fall or prevent it from increasing for numerous reasons, and sales by such persons could be viewed negatively by other investors.
In accordance with the guidelines specified under Rule 10b5-1 of the Securities Exchange Act of 1934, as amended, and our policies regarding stock transactions, a number of our employees, including executive officers, have adopted and may continue to adopt stock trading plans pursuant to which they have arranged to sell shares of our common stock from time to time in the
future. Generally, sales under such plans by our executive officers and directors require public filings. Sales of our common stock by such persons could cause the price of our common stock to fall or prevent it from increasing. If sales by employees, executive officers or directors cause a substantial number of shares of our common stock to become available for purchase in the public market, the price of our common stock could fall or may not increase. Also, sales by such persons could be viewed negatively by holders and potential purchasers of our common stock. If securities or industry analysts do not continue to publish research or publish inaccurate or unfavorable research about our business, our share price and trading volume could decline.
If securities analysts do not publish research or reports about our business or if they publish negative, or inaccurate, evaluations of our stock, the price of our stock and trading volume could decline.
The trading market for our common stock may be impacted, in part, by the research and reports that securities or industry analysts publish about us or our business. There can be no assurance that analysts will cover us, continue to cover us or provide favorable coverage. If one or more analysts downgrade our stock or change their opinion of our stock, our share price may decline. In addition, if one or more analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in the financial markets, which could cause our share price or trading volume to decline.
Item 1B. Unresolved Staff Comments
Not applicable.
Item 2. Properties
Our corporate, research and development, manufacturing, and clinical trial operations are located in Cambridge, Massachusetts. We lease approximately 88,00099,000 square feet of office and laboratory space in threefour adjacent buildings with aggregate monthly rent expense of approximately $0.3 million, which will increase to approximately $0.4 million in April 2016. Twomillion. Three of our leases expire in September 20182023 and one lease expires in March 2021.
We believe our facilities are adequate for our current needs and that suitable additional substitute space would be available if needed.
Item 3. Legal Proceedings
While we are not currently a party to any material legal proceedings, we could become subject to legal proceedings in the ordinary course of business. We do not expect any such potential items to have a significant impact on our financial position.
Item 4. Mine Safety Disclosures
Not applicable.
PART II
Item 5. Market for Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information and Stockholders
Our common stock has been listed on The NASDAQ Global Market under the symbol "XLRN" since September 19, 2013. Prior to that, there was no public market for our common stock. The following table sets forth for the periods indicated the high and low sales prices per share of our common stock as reported on The NASDAQ Global Market:
| Common Stock Price | Common Stock Price | |||||||||||||||||||||||||||||
| 2015 | 2014 | 2017 | 2016 | |||||||||||||||||||||||||||
| High | Low | High | Low | High | Low | High | Low | |||||||||||||||||||||||
| First Quarter | $ | 43.00 | $ | 35.11 | $ | 57.89 | $ | 33.81 | $ | 32.57 | $ | 23.07 | $ | 48.05 | $ | 22.67 | ||||||||||||||
| Second Quarter | $ | 37.90 | $ | 26.94 | $ | 42.24 | $ | 28.53 | $ | 34.91 | $ | 24.37 | $ | 37.67 | $ | 25.49 | ||||||||||||||
| Third Quarter | $ | 36.31 | $ | 20.00 | $ | 35.00 | $ | 23.61 | $ | 40.35 | $ | 29.57 | $ | 40.70 | $ | 28.29 | ||||||||||||||
| Fourth Quarter | $ | 50.86 | $ | 21.93 | $ | 48.50 | $ | 27.64 | $ | 43.68 | $ | 33.37 | $ | 41.69 | $ | 24.77 | ||||||||||||||
As of January 31, 2016,2018, there were approximately 10579 holders of record of our common stock.
Dividends
We have never declared or paid cash dividends on our common stock. We currently intend to retain all available funds and any future earnings, if any, to fund the development and expansion of our business and we do not anticipate paying any cash dividends in the foreseeable future. In addition, any future debt financing arrangement may contain terms prohibiting or limiting the amount of dividends that may be declared or paid on our common stock. Any future determination to pay dividends will be made at the discretion of our board of directors.
Securities Authorized for Issuance Under Equity Compensation Plans
Information about our equity compensation plans is incorporated herein by reference to Item 12 of Part III of this Annual Report on Form 10-K.
Performance Graph
The following graph shows a comparison from September 19, 2013 through December 31, 20152017 of cumulative total return on assumed investment of $100.00 in cash in our common stock, the NASDAQ Composite Index and the NASDAQ Biotechnology Index. Such returns are based on historical results and are not intended to suggest future performance. Data for the NASDAQ Composite Index and the NASDAQ Biotechnology Index assume reinvestment of dividends.
COMPARISON OF 2751 MONTH CUMULATIVE TOTAL RETURN(1)(2)
Among Acceleron Pharma Inc., the NASDAQ Composite Index,
and the NASDAQ Biotechnology Index
| (1) | This performance graph shall not be deemed "soliciting material" or to be "filed" with the SEC for purposes of Section 18 of the Securities and Exchange Act of 1934, as amended, or otherwise subject to the liabilities under that Section, and shall not be deemed incorporated by reference into any filing of Acceleron Pharma Inc. under the Securities Act of 1933, as amended. |
| (2) | $100 invested on September 19, 2013 in stock, or on August 31, 2013 in each index, including reinvestment of dividends. |
Recent Sales of Unregistered Securities
In December 2017, we issued 2,207 shares of Common Stock upon the cashless exercise of warrants to purchase 2,702 shares of Common Stock.
These issuances of shares of our common stock were exempt from registration under the Securities Act of 1933, or the Securities Act, pursuant to Rule 506 of Regulation D of the Securities Act and Section 4(a)(2) of the Securities Act.
Purchases of Equity Securities by the Issuer or Affiliated Purchasers
None.
Item 6. Selected Financial Data
The information set forth below should be read in conjunction with the "Management's Discussion and Analysis of Financial Condition and Results of Operations" section and with our consolidated financial statements and notes thereto included elsewhere in this Annual Report on Form 10-K. The selected financial data in this section is not intended to replace the consolidated financial statements and are qualified in their entirety by the consolidated financial statements and related notes included elsewhere in this Annual Report on Form 10-K.
The selected consolidated statements of operations data for the years ended December 31, 2015, 20142017, 2016 and 20132015 and the consolidated balance sheet data as of December 31, 20152017 and 20142016 have been derived from our audited consolidated financial statements included elsewhere in this Annual Report on Form 10-K. We derived the selected consolidated financial data for the years ended December 31, 20122014 and 20112013 and as of December 31, 2013, 20122015, 2014 and 20112013 from audited financial statements which are not included in this Annual Report on Form 10-K.
Historical results are not necessarily indicative of the results to be expected in future periods.
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||||||||||||||||||
| (in thousands, except per share data) | 2015 | 2014 | 2013 | 2012 | 2011 | 2017 | 2016 | 2015 | 2014 | 2013 | ||||||||||||||||||||||||||||
| Consolidated Statements of Operations Data: | ||||||||||||||||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||||||||||||||||
| Collaboration revenue: | ||||||||||||||||||||||||||||||||||||||
| License and milestone | $ | 1,184 | $ | 1,673 | $ | 43,948 | $ | 9,696 | $ | 74,406 | $ | 541 | $ | 15,550 | $ | 1,184 | $ | 1,673 | $ | 43,948 | ||||||||||||||||||
| Cost-sharing, net | 16,913 | 12,959 | 13,282 | 5,558 | 4,760 | 12,940 | 12,221 | 16,913 | 12,959 | 13,282 | ||||||||||||||||||||||||||||
| Contract manufacturing | — | — | — | — | 1,745 | |||||||||||||||||||||||||||||||||
| Total revenue | 18,097 | 14,632 | 57,230 | 15,254 | 80,911 | 13,481 | 27,771 | 18,097 | 14,632 | 57,230 | ||||||||||||||||||||||||||||
| Costs and expenses: | ||||||||||||||||||||||||||||||||||||||
| Research and development | 58,404 | 50,897 | 36,051 | 35,319 | 32,713 | 89,726 | 68,580 | 58,404 | 50,897 | 36,051 | ||||||||||||||||||||||||||||
| Litigation Settlement | — | 5,000 | — | — | — | |||||||||||||||||||||||||||||||||
| Litigation settlement | — | — | — | 5,000 | — | |||||||||||||||||||||||||||||||||
| General and administrative | 20,572 | 14,199 | 14,227 | 8,824 | 8,142 | 33,738 | 25,297 | 20,572 | 14,199 | 14,227 | ||||||||||||||||||||||||||||
| Cost of contract manufacturing revenue | — | — | — | — | 1,500 | |||||||||||||||||||||||||||||||||
| Total costs and expenses | 78,976 | 70,096 | 50,278 | 44,143 | 42,355 | 123,464 | 93,877 | 78,976 | 70,096 | 50,278 | ||||||||||||||||||||||||||||
| (Loss) income from operations | (60,879 | ) | (55,464 | ) | 6,952 | (28,889 | ) | 38,556 | (109,983 | ) | (66,106 | ) | (60,879 | ) | (55,464 | ) | 6,952 | |||||||||||||||||||||
| Total other (expense) income, net | (3,015 | ) | 4,205 | (28,850 | ) | (3,693 | ) | (2,290 | ) | |||||||||||||||||||||||||||||
| Net (loss) income | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | $ | (32,582 | ) | $ | 36,266 | ||||||||||||||||||||||||
| Net (loss) income per share applicable to common stockholders(1) | ||||||||||||||||||||||||||||||||||||||
| Basic | $ | (1.92 | ) | $ | (1.63 | ) | $ | (4.15 | ) | $ | (24.84 | ) | $ | 0.80 | ||||||||||||||||||||||||
| Diluted | $ | (1.92 | ) | $ | (1.63 | ) | $ | (4.15 | ) | $ | (24.84 | ) | $ | 0.78 | ||||||||||||||||||||||||
| Weighted-average number of common shares used in computing net (loss) income per share applicable to common stockholders | ||||||||||||||||||||||||||||||||||||||
| Basic | 33,303 | 31,515 | 9,407 | 2,401 | 2,328 | |||||||||||||||||||||||||||||||||
| Diluted | 33,303 | 31,515 | 9,407 | 2,401 | 2,716 | |||||||||||||||||||||||||||||||||
| Total other income (expense), net | 1,561 | 9,116 | (3,015 | ) | 4,205 | (28,850 | ) | |||||||||||||||||||||||||||||||
| Loss before income taxes | (108,422 | ) | (56,990 | ) | (63,894 | ) | (51,259 | ) | (21,898 | ) | ||||||||||||||||||||||||||||
| Income tax provision | (32 | ) | (24 | ) | — | — | — | |||||||||||||||||||||||||||||||
| Net loss | $ | (108,454 | ) | $ | (57,014 | ) | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | |||||||||||||||||||||||
| Reconciliation of net loss to net loss applicable to common stockholders: | ||||||||||||||||||||||||||||||||||||||
| Net loss | $ | (108,454 | ) | $ | (57,014 | ) | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | |||||||||||||||||||||||
| Accretion of dividends, interest, redemption value and issuance costs on redeemable convertible preferred stock | — | — | — | — | (19,870 | ) | ||||||||||||||||||||||||||||||||
| Gain on extinguishment of redeemable convertible preferred stock | — | — | — | — | 2,765 | |||||||||||||||||||||||||||||||||
| Net loss applicable to common stockholders-basic and diluted | $ | (108,454 | ) | $ | (57,014 | ) | $ | (63,894 | ) | $ | (51,259 | ) | $ | (39,003 | ) | |||||||||||||||||||||||
| Net loss per share applicable to common stockholders-basic and diluted(1) | $ | (2.68 | ) | $ | (1.52 | ) | $ | (1.92 | ) | $ | (1.63 | ) | $ | (4.15 | ) | |||||||||||||||||||||||
| Weighted-average number of common shares used in computing net loss per share applicable to common stockholders-basic and diluted | 40,420 | 37,430 | 33,303 | 31,515 | 9,407 | |||||||||||||||||||||||||||||||||
| As of December 31, | As of December 31, | |||||||||||||||||||||||||||||||||||||
| (in thousands) | 2015 | 2014 | 2013 | 2012 | 2011 | 2017 | 2016 | 2015 | 2014 | 2013 | ||||||||||||||||||||||||||||
| Consolidated Balance Sheet Data: | ||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 27,783 | $ | 176,460 | $ | 113,163 | $ | 39,611 | $ | 65,037 | $ | 100,150 | $ | 20,950 | $ | 27,783 | $ | 176,460 | $ | 113,163 | ||||||||||||||||||
| Short and long-term investments | 108,198 | — | — | — | — | 272,800 | 213,432 | 108,198 | — | — | ||||||||||||||||||||||||||||
| Total assets | 146,337 | 186,296 | 123,732 | 49,212 | 73,789 | 389,177 | 247,647 | 146,337 | 186,296 | 123,732 | ||||||||||||||||||||||||||||
| Total current liabilities | 14,491 | 9,253 | 18,162 | 38,802 | 23,853 | 16,745 | 16,149 | 14,491 | 9,253 | 18,162 | ||||||||||||||||||||||||||||
| Long-term deferred revenue | 4,239 | 4,816 | 5,620 | 6,760 | 33,350 | 3,161 | 3,704 | 4,239 | 4,816 | 5,620 | ||||||||||||||||||||||||||||
| Long-term deferred rent | 1,157 | 1,818 | 2,337 | 2,837 | 3,335 | 1,818 | 953 | 1,157 | 1,818 | 2,337 | ||||||||||||||||||||||||||||
| Long-term notes payable | — | — | 9,048 | 16,525 | — | — | — | — | — | 9,048 | ||||||||||||||||||||||||||||
| Warrants to purchase redeemable convertible preferred stock | — | — | — | 1,422 | 1,046 | |||||||||||||||||||||||||||||||||
| Warrants to purchase common stock | 17,187 | 14,124 | 30,753 | 5,229 | 3,347 | 2,236 | 1,244 | 17,187 | 14,124 | 30,753 | ||||||||||||||||||||||||||||
| Redeemable convertible preferred stock | — | — | — | 268,610 | 241,549 | |||||||||||||||||||||||||||||||||
| Total stockholder's equity (deficit) | 109,263 | 156,285 | 57,812 | (290,973 | ) | (232,691 | ) | |||||||||||||||||||||||||||||||
| Total stockholder's equity | 365,217 | 225,597 | 109,263 | 156,285 | 57,812 | |||||||||||||||||||||||||||||||||
| (1) | See Note 2 within the notes to our consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K for a description of the method used to calculate basic and diluted net |
Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations
The following discussion of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and the notes to those consolidated financial statements included in Item 15 of this Annual Report on Form 10-K. This discussion contains forward-looking statements that involve significant risks and uncertainties. As a result of many factors, such as those set forth under "Risk Factors" and elsewhere in this Annual Report on Form 10-K, our actual results may differ materially from those anticipated in these forward-looking statements. Please also refer to the section under heading "Forward-Looking Statements."
Overview
We are a clinical stageleading biopharmaceutical company focused onin the discovery and development of TGF-beta therapeutics to treat serious and commercialization of novel therapeutic candidates that are based on the mechanisms that the human body uses to regulate the growth and repair of its cells and tissues.rare diseases. Our research focuses on key natural regulators of cellular growth and repair, particularly the Transforming Growth Factor-Beta, or TGF-beta, protein superfamily. We are a leading company in discovering and developing therapeutic candidates that regulate cellular growth and repair. By combining our discovery and development expertise, including our proprietary knowledge of the TGF-beta superfamily, and our internal protein engineering and manufacturing capabilities, we have built a highly productive discovery and development platform that has generated several innovative therapeutic candidates, withall of which encompass novel potential first-in-class mechanisms of action. These differentiatedWe have focused and prioritized our research and development activities within three key therapeutic areas: hematologic, neuromuscular and pulmonary. If successful, these candidates could have the potential to significantly improve clinical outcomes for patients across many fieldsthese areas of medicine, and we have focused our discovery and development efforts on treatments for cancer and rare diseases.
Luspatercept, our lead program, and sotatercept, are partnered with Celgene Corporation, or Celgene. Luspatercept is an erythroid maturation agent designed to promote red blood cell production through a novel mechanism, and we are developing luspatercept with Celgeneis being developed to treat chronic anemia and associated complications in myelodysplastic syndromes, (MDS)or MDS, beta-thalassemia, and beta-thalassemia. In 2015,myelofibrosis. Celgene initiatedis currently conducting two Phase 3 clinical trials for luspaterceptwith luspatercept; one for the treatment of patients with lower-risk MDS, known as the "MEDALIST" trial, and beta-thalassemia.another for the treatment of patients with beta-thalassemia, also known as the "BELIEVE" trial. Celgene has recently initiated a Phase 2 trial in non-transfusion-dependent beta-thalassemia patients, referred to as the "BEYOND" trial. We further expect Celgene to initiate a Phase 3 clinical trial, the "COMMANDS" trial, in first-line, lower-risk MDS patients in the first half of 2018. Enrollment is also currently ongoing in a Phase 2 clinical trial for the treatment of patients with myelofibrosis, a rare bone marrow disorder. If luspatercept were to receive regulatory approval for each of these indications in the United States and Europe, we believe that there is an aggregate sales opportunity for this product in excess of $2 billion.
For sotatercept, we announced in September 2017 that Celgene are developinggranted us the rights to fund, develop, and lead the global commercialization of sotatercept in pulmonary hypertension, including pulmonary arterial hypertension, or PAH. PAH is a rare and chronic, rapidly progressing disorder characterized by the constriction of small pulmonary arteries, resulting in abnormally high blood pressure in the pulmonary arteries. If sotatercept is commercialized to treat PAH and we recognize such revenue, then Celgene will be eligible to receive a royalty in the low 20% range on global net sales. In certain circumstances Celgene may recognize revenue related to the commercialization of sotatercept in PAH, and in this scenario we will be eligible to receive a royalty from Celgene such that the economic position of the parties is equivalent to the scenario in which we recognize such revenue. We expect to initiate a Phase 2 clinical trial for the treatment of patients with chronic kidney disease. Sotatercept hasPAH in the potential to treat several complicationsfirst half of chronic kidney disease including mineral-bone disorder, vascular calcification2018.
For luspatercept and, anemia.outside of pulmonary hypertension, sotatercept, Celgene is responsible for paying 100% of the development costs for all clinical trials for luspatercept and sotatercept, including our ongoing earlier stage clinical trials for these therapeutic candidates.trials. We may receive up to an additional $560.0a maximum of $545.0 million offor the potential development, regulatory and commercial milestone paymentspayments. If luspatercept and, if these therapeutic candidatesoutside of pulmonary hypertension, sotatercept are commercialized, we willare eligible to receive a royalty on net sales in the low-to-mid 20% range. We will co-promote luspaterceptrange and sotatercept, if approved,we have a co-promotion right in North America, for which our commercialization costs will be entirely funded by Celgene.
We wholly own dalantercept and ACE-083, and we are independently developing these therapeutic candidates. We are currently evaluating dalantercept in a Phase 2 clinical trial for the treatment of patients with renal cell carcinoma.our wholly-owned neuromuscular candidate, ACE-083. ACE-083 is designed for the treatment of focal muscle disorders, such as facioscapulohumeral dystrophy, and we are currently conducting a Phase 2 clinical trials with ACE-083 in patients with facioscapulohumeral dystrophy, or FSHD, as well as in patients with Charcot-Marie-Tooth disease, or CMT. In January 2018, we announced preliminary results for the first two cohorts in part 1 of the Phase 2 clinical trial with ACE-083 in healthy volunteers. In 2015, we reported data from the Phase 1 clinical trial of ACE-083patients with FSHD showing marked increases in the mean total muscle volume of the muscles treated with ACE-083.ACE-083 measured using magnetic resonance imaging, or MRI. We expect to initiate part 2 of the ACE-083 FSHD Phase 2 trial during the second quarter of this year, and we expect to report preliminary results from all dose-escalation cohorts of part 1 in both of our Phase 2 clinical trials with ACE-083 in the second half of this year.
In addition to our mid- to late-stage clinical programs, we initiated a Phase 1 healthy volunteer study in early 2018 with ACE-2494, our wholly-owned systemic muscle agent from our proprietary platform technology, IntelliTrap™, and we expect to report initial results from this healthy volunteer study in the first half of 2019. We are also conducting research within our three
focused disease areas—hematologic, neuromuscular and pulmonary—in order to identify new therapeutic candidates to bring forwardadvance into clinical trials. To this end, in 2015 we implemented a new platform technology, IntelliTrap™, that is accelerating our discovery efforts. We have nominated an IntelliTrap™ molecule, ACE-2494, as a candidate for clinical development and will initiate IND-enabling activities in 2016. ACE-2494 is designed to treat systemic muscle disorders.
As of December 31, 2015,2017, our operations have been funded primarily by $105.1 million in equity investments from venture investors, $219.3$539.7 million from public investors, $64.2$123.7 million in equity investments from our collaboration partners and $233.5$273.7 million in upfront payments, milestones, and net research and development payments from our collaboration partners. We estimate that we have spent approximately $145.4$89.7 million, $68.6 million, and $58.4 million, on research and development for the three year period from 2013 through 2015.years ended December 31, 2017, 2016, and 2015, respectively.
We expect to continue to incur significant expenses and increasing operating losses over at least the next several years. We expect our expenses will increase substantially in connection with our ongoing activities, as we:
conduct clinical trials for dalantercept and ACE-083;ACE-083, sotatercept, ACE-2494 or any future therapeutic candidates;
continue our preclinical studies and potential clinical development efforts of our existing preclinical therapeutic candidates;
continue research activities for the discovery of new therapeutic candidates;
manufacture therapeutic candidates for our preclinical studies and clinical trials;
acquire or in-license other therapeutic candidates and patents;
seek regulatory approval for our therapeutic candidates; and
operate as a public company.
We will not generate revenue from product sales unless and until we or a partner successfully complete development and obtain regulatory approval for one or more of our therapeutic candidates. We expect that this will take a number of years and is subject to significant uncertainty. All current and future development and commercialization costs for sotaterceptluspatercept and, luspaterceptoutside of pulmonary hypertension, sotatercept, are paid by Celgene. If we obtain regulatory approval for dalantercept, ACE-083, sotatercept, ACE-2494 or any future therapeutic candidate, we expect to incur significant commercialization expenses related to product sales, marketing, manufacturing and distribution to the extent that such costs are not paid by future partners. We will seek to fund our operations through the sale of equity, debt financings or other sources, including potential additional collaborations. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms, or at all. If we fail to raise capital or enter into such other arrangements as, and when, needed, we may have to significantly delay, scale back or discontinue the development or commercialization of one or more of our therapeutic candidates.
Our ability to generate product revenue and become profitable depends upon our and our partners' ability to successfully commercialize products. We expect to incur losses for the foreseeable future, and we expect these losses to increase as we continue our development of, and seek regulatory approvals for, our therapeutic candidates and potentially begin to commercialize any approved products. For a description of the numerous risks and uncertainties associated with product development, see "Risk Factors".
Financial Operations Overview
Revenue
Collaboration Revenue
We have not generated any revenue from the sale of products. Our revenue to date has been predominantly derived from collaboration revenue, which includes license and milestone revenues and cost sharingcost-sharing revenue, generated through collaboration and license agreements with partners for the development and commercialization of our therapeutic candidates. Cost sharingCost-sharing revenue represents amounts reimbursed by our collaboration partners for expenses incurred by us for research and development activities and, potentially, co-promotion activities, under our collaboration agreements. Cost sharingCost-sharing revenue is recognized in the period that the related activities are performed. To the extent that we reimburse collaborators for costs incurred in connection with activities performed by them, we record these costs as a reduction of cost-sharing revenue.
Costs and Expenses
Research and Development Expenses
Research and development expenses consist primarily of costs directly incurred by us for the development of our therapeutic candidates, which include:
direct employee-related expenses, including salaries, benefits, travel and stock-based compensation expense of our research and development personnel;
expenses incurred under agreements with clinical research organizations, or CROs, and investigative sites that will conduct our clinical trials;
the cost of acquiring and manufacturing preclinical and clinical study materials and developing manufacturing processes;
allocated facilities, depreciation, and other expenses, which include rent and maintenance of facilities, insurance and other supplies;
expenses associated with obtaining and maintaining patents; and
costs associated with preclinical activities and regulatory compliance.
Research and development costs are expensed as incurred. Costs for certain development activities are recognized based on an evaluation of the progress to completion of specific tasks using information and data provided to us by our vendors and our clinical sites.
We cannot determine with certainty the duration and completion costs of the current or future clinical trials of our therapeutic candidates or if, when, or to what extent we will generate revenues from the commercialization and sale of any of our therapeutic candidates for which we or any partner obtain regulatory approval. We or our partners may never succeed in achieving regulatory approval for any of our therapeutic candidates. The duration, costs and timing of clinical trials and development of our therapeutic candidates will depend on a variety of factors, including:
the scope, rate of progress, and expense of our ongoing, as well as any additional, clinical trials and other research and development activities;
future clinical trial results;
potential changes in government regulation; and
the timing and receipt of any regulatory approvals.
A change in the outcome of any of these variables with respect to the development of a therapeutic candidate could mean a significant change in the costs and timing associated with the development of that therapeutic candidate. For example, if the FDA,U.S. Food and Drug Administration, or another regulatory authority were to require us to conduct clinical trials beyond those that we currently anticipate will be required for the completion of the clinical development of therapeutic candidates, or if we experience significant delays in the enrollment in any clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development.
From inception through December 31, 2015,2017, we have incurred $396.5$554.8 million in research and development expenses. We plan to increase our research and development expenses for the foreseeable future as we continue the development of our TGF-beta platform therapeutic candidates, the discovery and development of preclinical therapeutic candidates, and the development of sotatercept, luspatercept, dalantercept and ACE-083. As of January 1, 2013, expensesour clinical programs. Expenses associated with sotaterceptluspatercept, and luspaterceptoutside of pulmonary hypertension, sotatercept, are reimbursed 100% by Celgene. These reimbursements are recorded as revenue. Of the Phase 2 clinical trials that are underway for sotatercept, luspatercept and dalantercept, we are expensing the costs of seven clinical trials of luspatercept and dalantercept, of which the four for luspatercept are reimbursed by Celgene. We are also expensing the costs of a Phase 1 clinical trial for ACE-083.
We manage certain activities such as clinical trial operations, manufacture of therapeutic candidates, and preclinical animal toxicology studies through third-party CROs. The only costs we track by each therapeutic candidate are external costs such as services provided to us by CROs, manufacturing of preclinical and clinical drug product, and other outsourced research and development expenses. We do not assign or allocate to individual development programs internal costs such as salaries and benefits, facilities costs, lab supplies and the costs of preclinical research and studies. Our external research and development expenses for sotatercept, luspatercept, dalantercept, ACE-031 (for which development was suspended in April 2013) and ACE-083 (for which development commenced in the fourth quarter of 2013) during the years ended December 31, 2017, 2016 and 2015, 2014 and 2013, arewere as follows:
| Year ended December 31, | Year ended December 31, | |||||||||||||||||||||
| (in thousands) | 2015 | 2014 | 2013 | 2017 | 2016 | 2015 | ||||||||||||||||
| Sotatercept(1) | $ | — | $ | — | $ | 2 | $ | 495 | $ | — | $ | — | ||||||||||
| Luspatercept(1) | 9,744 | 7,944 | 5,081 | 6,616 | 6,945 | 9,744 | ||||||||||||||||
| Dalantercept | 8,864 | 7,526 | 4,636 | 4,718 | 6,757 | 8,864 | ||||||||||||||||
| ACE-083 | 3,293 | 5,111 | 105 | 9,797 | 4,973 | 3,293 | ||||||||||||||||
| ACE-031(2) | 3 | 8 | 1,023 | |||||||||||||||||||
| ACE-2494 | 4,382 | 1,045 | — | |||||||||||||||||||
| Total direct research and development expenses | 21,904 | 20,589 | 10,847 | 26,008 | 19,720 | 21,901 | ||||||||||||||||
| Other expenses(3) | 36,500 | 30,308 | 25,204 | 63,718 | 48,860 | 36,503 | ||||||||||||||||
| Total research and development expenses | $ | 58,404 | $ | 50,897 | $ | 36,051 | $ | 89,726 | $ | 68,580 | $ | 58,404 | ||||||||||
| (1) |
| (2) |
| (3) | Other expenses include unallocated employee and contractor-related expenses, facility expenses, lab supplies and miscellaneous expenses. |
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related costs for personnel, including stock-based compensation and travel expenses for our employees in executive, operational, finance and human resource functions and other general and administrative expenses including directors' fees and professional fees for accounting and legal services.
Other Expense,Income (Expense), Net
Other expense,income (expense), net consists primarily of interest expense from our previous venture debt facility, interest income earned on cash and cash equivalents, and the re-measurement gain or loss associated with the change in the fair value of our preferred stock and common stock warrant liabilities.liabilities and interest income earned on cash, cash equivalents and investments.
To estimate the fair value of our liability-classified warrants, we use either the warrants.Monte Carlo simulation framework, which incorporates future financing events over the remaining life of the warrants to purchase common stock, or for certain re-measurement dates, due to the warrants being deeply in the money, the Black-Scholes option pricing model. We base the estimates in the Black-Scholes option pricing model,models, in part, on subjective assumptions, including stock price volatility, risk-free interest rate, dividend yield, and the fair value of the preferred stock or common stock underlying the warrants. The Black-Scholes option pricing model was used at December 31, 2017.
Critical Accounting Policies and Significant Judgments and Estimates
Our management's discussion and analysis of our financial condition and results of operations are based on our consolidated financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, and expenses and the disclosure of contingent assets and liabilities in our consolidated financial statements. On an ongoing basis, we evaluate our estimates and judgments, including those related to revenue recognition, accrued expenses and stock-based compensation. We also utilize significant estimates and assumptions in determining the fair value of our common stock and the fair value of our liability-classified warrants to purchase preferred stock and common stock. We base our estimates on historical experience, known trends and events, and various other factors that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in the notes to our consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K, we believe the following accounting policies to be most critical to the judgments and estimates used in the preparation of our consolidated financial statements.
Revenue Recognition
We have primarily generated revenue through collaboration arrangements with strategic partners for the development and commercialization of our therapeutic candidates.
We recognize revenue in accordance with Accounting Standards Codification (ASC) Topic 605, Revenue Recognition. Accordingly, revenue is recognized for each unit of accounting when all of the following criteria are met: (1) persuasive evidence of an arrangement exists; (2) delivery has occurred or services have been rendered; (3) the fee is fixed or determinable; and (4) collectability is reasonably assured.
Amounts received prior to satisfying the revenue recognition criteria are recorded as deferred revenue on our consolidated balance sheets. Amounts expected to be recognized as revenue within the 12 months following the balance sheet date are classified as deferred revenue, current portion and amounts not expected to be recognized as revenue within the 12 months following the balance sheet date are classified as deferred revenue, net of current portion.
Under collaboration agreements, we may receive payments for non-refundable up-frontupfront fees, milestone payments upon achieving significant development events, research and development reimbursements and royalties on future product sales. These payments are received in connection with the deliverables contained in the arrangements which may include (1) licenses, or options to obtain licenses, to our technology, (2) research and development activities performed for the collaboration partner, (3) participation on joint committees and (4) manufacturing clinical or preclinical material.
Effective January 1, 2011, we adopted Accounting Standards Update (ASU) No. 2009-13, Multiple-Deliverable Revenue Arrangements, which amends ASC Topic 605-25, Revenue Recognition—Multiple Element Arrangements. This guidance applies to new arrangements as well as existing agreements that are significantly modified after January 1, 2011.
The application of the multiple element guidance requires subjective determinations, and requires management to make judgments about the individual deliverables, and whether such deliverables are separable from the other aspects of the contractual relationship. Deliverables are considered separate units of accounting provided that: (1) the delivered item(s) has value to the customer on a stand-alone basis and (2) if the arrangement includes a general right of return relative to the delivered item(s), delivery or performance of the undelivered item(s) is considered probable and substantially in our control. In determining the units of accounting, management evaluates certain criteria, including whether the deliverables have stand-alone value, based on the consideration of the relevant facts and circumstances for each arrangement, such as the research, manufacturing and commercialization capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. In addition, we consider whether the collaboration partner can use the other deliverable(s) for their intended purpose without the receipt of the remaining element(s), whether the value of the deliverable is dependent on the undelivered item(s), and whether there are other vendors that can provide the undelivered element(s).
Arrangement consideration that is fixed or determinable is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria, as described above, are applied to each of the separate units of accounting in determining the appropriate period or pattern of recognition. We determine the estimated selling price for deliverables within each agreement using vendor-specific objective evidence (VSOE) of selling price, if available, third-party evidence (TPE) of selling price if VSOE is not available, or management's best estimate of selling price (BESP) if neither VSOE nor TPE is available. Subsequent to the adoption of ASU 2009-13, we typically use BESP to estimate the selling price of the deliverables. Determining the BESP for a unit of accounting requires significant judgment. In developing the BESP
for a unit of accounting, we consider applicable market conditions and relevant entity-specific factors, including factors that
were contemplated in negotiating the agreement with the customer and estimated costs. We validate the BESP for units of accounting by evaluating whether changes in the key assumptions used to determine the BESP will have a significant effect on the allocation of arrangement consideration between multiple units of accounting.
Our agreements may contain options which provide the collaboration partner the right to obtain additional licenses. Options are considered substantive if, at the inception of the arrangement, we are at risk as to whether the collaboration partner will choose to exercise the option. Factors that we consider in evaluating whether an option is substantive include the overall objective of the arrangement, the benefit the collaborator might obtain from the arrangement without exercising the option, the cost to exercise the option and the likelihood that the option will be exercised. For arrangements under which an option is considered substantive, we do not consider the item underlying the option to be a deliverable at the inception of the arrangement and the associated option fees are not included in allocable arrangement consideration, assuming the option is not priced at a significant and incremental discount. Conversely, for arrangements under which an option is not considered substantive or if an option is priced at a significant and incremental discount, we would consider the item underlying the option to be a deliverable at the inception of the arrangement and a corresponding amount would be included in allocable arrangement consideration.
We typically receive up-front,upfront, non-refundable payments when licensing our intellectual property in conjunction with a research and development agreement. When we believe the license to our intellectual property has stand-alone value, we generally recognize revenue attributed to the license upon delivery. When we believe the license to our intellectual property does not have stand-alone value from the other deliverables to be provided in the arrangement, we generally recognize revenue attributed to the license on a straight-line basis over our contractual or estimated performance period, which is typically the term of our research and development or manufacturing obligations. We continually evaluate these periods, and will adjust the period of revenue recognition if circumstances change.
Research and development funding is recognized as revenue in the period that the related services are performed. When we act as the principal under our collaboration arrangements, we record payments received for the reimbursement of research and development costs as cost-sharing revenue. To the extent that we reimburse the collaborator for costs incurred, we record these costs as a reduction of cost-sharing revenue.
We periodically review the basis for our estimates, and we may change the estimates if circumstances change. These changes can significantly increase or decrease the amount of revenue recognized. As we apply our policy to our collaboration arrangements we make judgments which affected the pattern of revenue recognition. For instance, in our arrangement with Celgene, we are obligated to provide research and development services. We are recognizing revenue related to these research and development services over the estimated period of our performance, which was initially estimated to end in December 2014. The Company re-assessed the duration of its deliverables under the collaboration agreement and now estimates the new term to end in the first quarter of 2016, the expected completion date ofconcluded we had completed the proof-of-concept trials for luspatercept under the Celgene collaboration. Another instance relates to our arrangement with Shire AG, wherecollaboration in April 2013, we and Shire determined not to further advance the developmentfirst quarter of ACE-031 or back-up compounds and Shire terminated our collaboration agreement effective as of June 30, 2013.2016.
In addition to up-frontupfront payments and research and development funding, we may also be entitled to milestone payments that are contingent upon achievement of a predefined objective. At the inception of each arrangement that includes milestone payments, we evaluate whether the milestone is substantive and at-risk. This evaluation includes an assessment of whether (1) the consideration is commensurate with either the entity's performance to achieve the milestone, or the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting at least in part from the entity's performance to achieve the milestone, (2) the consideration relates solely to past performance, and (3) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. We evaluate factors such as the scientific, regulatory, commercial and other risks that must be overcome to achieve the respective milestone, the level of effort and investment required to achieve the respective milestone, and whether the milestone consideration is reasonable relative to all deliverables and payment terms in the arrangement in making this assessment. On the milestone achievement date, assuming all other revenue recognition criteria are met and the milestone is deemed substantive and at-risk, we recognize the payment as license and milestone revenue. For milestones that are not deemed substantive and at-risk, where payment is reasonably assured, we recognize the milestone payment over the remaining service period.
Sales and commercial milestones and royalties will be recognized when and if earned, provided collectability is reasonably assured.
CROs and investigative sites in connection with clinical investigators, based uponstudies;
vendors in connection with preclinical development activities; and
vendors related to product manufacturing, development and distribution of clinical supplies
We base our expenses related to CROs on our estimates made as of the reporting dateservices received and efforts expended pursuant to quotes and contracts with CROs that conduct research and development on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. There may be instances in which payments made to our vendors will exceed the level of services provided and result in a prepayment of the work completedresearch and development expense. In accruing service fees, we estimate the time period over which services will be performed and the lifelevel of effort to be expended in each period. If the actual timing of the individual study in accordance with agreements established with CROsperformance of services or the level of effort varies from our estimate, we adjust the accrual or prepaid accordingly. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and clinical trial sites. Some CROs invoice us on a monthly basis, while others invoice upon achievementtiming of milestones and the expense is recorded as services are rendered. We determine the estimates of clinical activities incurred at the end of each reporting period through discussion with internal personnel and outside service providers asperformed relative to the progress or stageactual status and timing of completion of trials or services as of the end of eachperformed may vary and could result in us reporting period, pursuant to contracts with numerous clinical trial centers and CROs and the agreed upon fee to be paid for such services. The significant factors considered in estimating accruals include the number of patients enrolled and the percentage of work completed to date. Costs of setting up clinical trial sites for participation in the trialsamounts that are paid fortoo high or too low in advance are expensed over the estimated set-upany particular period. While the set-up periods vary from one arrangement to another, such set-up periods generally take approximately three months. Set-up activities include clinical site identification, institutional review board, or IRB, submissions, regulatory submissions, clinical investigator kick-off meetings and pre-study site visits. Clinical trial site costs related to patient enrollments are accrued as patients are entered into the trial.
Stock-Based Compensation
We account for our stock-based awards in accordance with ASC Topic 718, Compensation—Stock Compensation, or ASC 718, which requires all stock-based payments to employees, including grants of employee stock options, modifications to existing stock options, and restricted stock unit awards, to be recognized in the statements of operations and comprehensive income (loss) based on their fair values. We recognize the compensation cost of awards subject to service-based vesting conditions over the requisite service period, which is generally equal to the vesting term. For awards subject to both performance and service-based vesting conditions, we recognize compensation cost using an accelerated recognition method when it is probable that the performance condition will be achieved. If achievement of the performance condition is not probable, but the award will vest based on the service condition, we recognize the expense over the requisite service period. We account for stock-based awards to non-employees using the fair value method. Stock options granted to non-employees are subject to periodic revaluation over their vesting terms and stock-based compensation cost is recognized using an accelerated recognition method.
We estimate the fair value of our stock-based awards to employees and non-employees using the Black-Scholes option pricing model, which requires the input of highly subjective assumptions, including (1) the expected volatility of our stock, (2) the expected term of the award, (3) the risk-free interest rate and (4) expected dividends. Due to the lack of a public market for our common stock prior to the completion of our initial public offering in September 2013, and resulting lack of company-specific historical and implied volatility data, we have based our estimate of expected volatility on the historical volatility of a group of similar companies that are publicly traded.traded companies. For these analyses, we have selected companies with characteristics that we believe are comparable to ours, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected life of the stock-based awards. We compute the historical volatility data using the daily closing prices for the selected companies' shares during the equivalent period as the calculated expected term of our stock-based awards. During 2015 we began to estimate our volatility by using a blend of our stock price history, for the length of time we have market data for our stock and the historical volatility of similar public companies for the expected term of each grant. We have estimated the expected life of our employee stock options using the "simplified" method, whereby, the expected life equals the average of the vesting term and the original contractual term of the option. The risk-free interest rates for periods within the expected life of the option are based on the U.S. Treasury yield curve in effect during the period the options were granted.
Prior to the adoption of ASU 2016-09 on January 1, 2017, we estimated forfeitures at the time of grant, and reviserevised those estimates in subsequent periods if actual forfeitures differdiffered from estimates. We useused historical data to estimate pre-vesting option forfeitures to the extent that actual forfeitures differdiffered from our estimates, the difference iswas recorded as a cumulative adjustment in the period the estimates were revised. Stock-based compensation expense recognized in the consolidated financial statements is based on awards that are ultimately expected to vest. For the years ended December 31, 2015, 20142017, 2016 and 2013, respectively,2015, the Company applied an estimated forfeiture rate of approximately zero, 3%, and 4%. , respectively.
Stock-based compensation totaled approximately $12.1$28.2 million, $4.8$18.6 million and $2.2$12.1 million for the years ended December 31, 2015, 20142017, 2016 and 2013,2015, respectively. We expect the impact of our stock-based compensation expense for stock-based awards granted to employees and non-employees to grow in future periods due to the potential increases in the value of our common stock and headcount.
Warrants to Purchase Preferred Stock and Common Stock
As of December 31, 2015,2017, we had warrants outstanding to purchase 398,01160,594 shares of common stock, all of which warrants to purchase 392,805 shares of our common stock contain a provision requiring an adjustment to the number of shares in the event we issue common stock, or securities convertible into or exercisable for common stock, at a price per share lower than the warrant exercise price. The anti-dilution feature requires the warrants to be classified as liabilities and measured at fair value, with changes in fair value recognized as a component of other income (expense). The fair value of the warrants to purchase common stock on the date of issuance and on each re-measurement date for those warrants to purchase common stock classified as liabilities was estimated using either the Monte Carlo simulation framework, which incorporates future financing events over the remaining life of the warrants to purchase common stock, or for certain re-measurement dates, including December 31, 2015, 2014 and 2013, due towhen the warrants beingare deeply in the money, the Black-Scholes option pricing model was used.model. At each reporting period the companyCompany evaluates the best valuation methodology. At December 31, 2017 the Black-Scholes option pricing model was used, at December 31, 2016 the Monte Carlo simulation framework was used, and at December 31, 2015 the Black-Scholes option pricing model was used. Any modifications to the warrant liabilities are recorded in earnings during the period of the modification. The significant assumptions used in estimating the fair value of our warrant liabilities include the exercise price, volatility of the stock underlying the warrant, risk-free interest rate, estimated fair value of the stock underlying the warrant, and the estimated life of the warrant. The common stock warrant liability was $17.2$2.2 million, $14.1$1.2 million and $30.8$17.2 million as of December 31, 2015, 20142017, 2016 and 2013,2015, respectively. At the end of each reporting period, the Companywe remeasured the fair value of the outstanding warrants, using current assumptions, resulting in an increase (decrease) in fair value of $1.0 million, $(7.3) million and $3.5 million $(5.0) millionfor the years ended December 31, 2017, 2016 and $26.9 million,2015, respectively, which was recorded in other income (expense), net in the accompanying consolidated statements of operations and comprehensive loss for the years ended December 31, 2015, 2014 and 2013.loss. The changes in value of the warrant liability were primarily due to fluctuations in the market price of our common stock.
Results of Operations
Comparison of the Years Ended December 31, 20152017 and 2014
2016
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| Increase (Decrease) | Increase (Decrease) | |||||||||||||||||||||
| (in thousands) | 2015 | 2014 | 2017 | 2016 | ||||||||||||||||||
| Revenue: | ||||||||||||||||||||||
| Collaboration revenue: | ||||||||||||||||||||||
| License and milestone | $ | 1,184 | $ | 1,673 | $ | (489 | ) | $ | 541 | $ | 15,550 | $ | (15,009 | ) | ||||||||
| Cost-sharing, net | 16,913 | 12,959 | 3,954 | 12,940 | 12,221 | 719 | ||||||||||||||||
| Total revenue | 18,097 | 14,632 | 3,465 | |||||||||||||||||||
| Total revenue (all amounts are with a related party) | 13,481 | 27,771 | (14,290 | ) | ||||||||||||||||||
| Costs and expenses: | ||||||||||||||||||||||
| Research and development | 58,404 | 50,897 | 7,507 | 89,726 | 68,580 | 21,146 | ||||||||||||||||
| Litigation settlement | — | 5,000 | (5,000 | ) | ||||||||||||||||||
| General and administrative | 20,572 | 14,199 | 6,373 | 33,738 | 25,297 | 8,441 | ||||||||||||||||
| Total costs and expenses | 78,976 | 70,096 | 8,880 | 123,464 | 93,877 | 29,587 | ||||||||||||||||
| (Loss) income from operations | (60,879 | ) | (55,464 | ) | (5,415 | ) | ||||||||||||||||
| Other (expense) income, net | (3,015 | ) | 4,205 | (7,220 | ) | |||||||||||||||||
| Loss from operations | (109,983 | ) | (66,106 | ) | (43,877 | ) | ||||||||||||||||
| Other income, net | 1,561 | 9,116 | (7,555 | ) | ||||||||||||||||||
| Loss before income taxes | (108,422 | ) | (56,990 | ) | (51,432 | ) | ||||||||||||||||
| Income tax provision | (32 | ) | (24 | ) | (8 | ) | ||||||||||||||||
| Net loss | $ | (63,894 | ) | $ | (51,259 | ) | $ | (12,635 | ) | $ | (108,454 | ) | $ | (57,014 | ) | $ | (51,440 | ) | ||||
Revenue. We recognized revenue of $18.1$13.5 million in the year ended December 31, 2015,2017, compared to $14.6$27.8 million in the year ended December 31, 2016. All of the revenue in both periods was derived from the Celgene agreements. The $14.3 million decrease was primarily due to the receipt of a $15.0 million milestone payment from Celgene for the initiation of a Phase 3 clinical trial with luspatercept in 2016, offset in part by an increase in cost-sharing revenue of $0.7 million primarily related to an increase in reimbursement for personnel compared to the prior year.
Research and Development Expenses. Research and development expenses were $89.7 million in the year ended December 31, 2017, compared to $68.6 million in the year ended December 31, 2016. The $21.1 million increase was primarily due to an increase in personnel expenses totaling $13.5 million to support development of our wholly-owned therapeutic candidates and preclinical programs, which includes an increase in stock-based compensation expense of $6.1 million. Other increases include clinical trial and toxicology expenses of $6.5 million and miscellaneous research and drug supply of $2.0 million. These increases were partially offset by a decrease in-licensing expense related to payments that we made in connection with the achievement of a milestone in 2016 totaling $0.9 million.
General and Administrative Expenses. General and administrative expenses were $33.7 million in the year ended December 31, 2017, compared to $25.3 million in the year ended December 31, 2016. The $8.4 million increase was primarily due to an increase in personnel expenses of $8.7 million, which includes an increase in stock-based compensation of $3.6 million. The increase in stock-based compensation was primarily due to modification of a former executive officer's equity awards announced in May 2017.
Other Income, Net. Other income, net was $1.6 million in the year ended December 31, 2017, compared to $9.1 million in the year ended December 31, 2016. This $7.5 million decrease was primarily due to an $8.3 million decrease in the gain associated with marking the common warrant liability to market, offset by an increase in interest income of $0.7 million.
Income Tax Provision. Income tax provision is attributable to taxes on interest income from our investment portfolio.
Comparison of the Years Ended December 31, 2016 and 2015
Year Ended December 31, | |||||||||||
Increase (Decrease) | |||||||||||
| (in thousands) | 2016 | 2015 | |||||||||
| Revenue: | |||||||||||
| Collaboration revenue: | |||||||||||
| License and milestone | $ | 15,550 | $ | 1,184 | $ | 14,366 | |||||
| Cost-sharing, net | 12,221 | 16,913 | (4,692 | ) | |||||||
| Total revenue (all amounts are with a related party) | 27,771 | 18,097 | 9,674 | ||||||||
| Costs and expenses: | |||||||||||
| Research and development | 68,580 | 58,404 | 10,176 | ||||||||
| General and administrative | 25,297 | 20,572 | 4,725 | ||||||||
| Total costs and expenses | 93,877 | 78,976 | 14,901 | ||||||||
| Loss from operations | (66,106 | ) | (60,879 | ) | (5,227 | ) | |||||
| Other income (expense), net | 9,116 | (3,015 | ) | 12,131 | |||||||
| Loss before income taxes | (56,990 | ) | (63,894 | ) | 6,904 | ||||||
| Income tax provision | (24 | ) | — | (24 | ) | ||||||
| Net loss | $ | (57,014 | ) | $ | (63,894 | ) | $ | 6,880 | |||
Revenue. We recognized revenue of $27.8 million in the year ended December 31, 2016, compared to $18.1 million in year ended December 31, 2014.2015. All of the revenue in both periods was derived from the Celgene agreements. This $3.5$9.7 million
increase was primarily due to higher cost sharingthe receipt of a $15.0 million milestone payment from Celgene for the initiation of a Phase 3 clinical trial with luspatercept, partially offset by a decrease in cost-sharing revenue of $4.0$4.7 million caused by higherprimarily due to lower expenses for luspatercept clinical trials and manufacturingtoxicology studies compared to the prior year and increased luspatercept expenses during 2015 related to a bulk drug substance during 2015, offsetmanufacturing campaign. Deferred revenue also decreased by a decrease in Celgene deferred revenue of $0.5$0.6 million during 2015 as we complete our deliverables under the collaboration agreement.Celgene agreements.
Research and Development Expenses. Research and development expenses were $68.6 million in the year ended December 31, 2016, compared to $58.4 million in the year ended December 31, 2015, compared to $50.9 million in the year ended December 31, 2014.2015. This $7.5$10.2 million increase was
primarily due to increases in personnel expenses totaling $8.5 million to support development of $3.2 million, including an increase of $2.8 million in stock compensation expense,our wholly-owned therapeutic candidates and preclinical programs, which includes an increase in stock-based compensation expense of $3.3 million. Other increases include licensing expense related to the achievement of our milestone totaling $0.9 million and miscellaneous research and drug supply expenses of $3.8 million. These increases were offset in part by a decrease in clinical and toxicologytrial expenses totaling $1.3 million and an increase of $2.5 million in miscellaneous expenses primarily for drug supply and outsourced research.
General and Administrative Expenses. General and administrative expenses were $25.3 million in the year ended December 31, 2016, compared to $20.6 million in the year ended December 31, 2015, compared to $14.2 million in the year ended December 31, 2014.2015. The $6.4$4.7 million increase was primarily due to an increase in personnel expenses includingof $4.2 million, which includes an increase in stockstock-based compensation expense of $4.5 million.
Year Ended December 31, | |||||||||||
Increase (Decrease) | |||||||||||
| (in thousands) | 2014 | 2013 | |||||||||
| Revenue: | |||||||||||
| Collaboration revenue: | |||||||||||
| License and milestone | $ | 1,673 | $ | 43,948 | $ | (42,275 | ) | ||||
| Cost-sharing, net | 12,959 | 13,282 | (323 | ) | |||||||
| Total revenue | 14,632 | 57,230 | (42,598 | ) | |||||||
| Costs and expenses: | |||||||||||
| Research and development | 50,897 | 36,051 | 14,846 | ||||||||
| Litigation settlement | 5,000 | — | 5,000 | ||||||||
| General and administrative | 14,199 | 14,227 | (28 | ) | |||||||
| Total costs and expenses | 70,096 | 50,278 | 19,818 | ||||||||
| (Loss) income from operations | (55,464 | ) | 6,952 | (62,416 | ) | ||||||
| Other income (expense), net | 4,205 | (28,850 | ) | 33,055 | |||||||
| Net loss | $ | (51,259 | ) | $ | (21,898 | ) | $ | (29,361 | ) | ||
| Year Ended December 31, | |||||||||||
| Increase (Decrease) | |||||||||||
| (in thousands) | 2014 | 2013 | |||||||||
| Collaboration revenue: | |||||||||||
| Celgene: | |||||||||||
| License and milestone | $ | 1,673 | $ | 19,626 | $ | (17,953 | ) | ||||
| Cost-sharing, net | 12,959 | 12,658 | 301 | ||||||||
| Total Celgene | 14,632 | 32,284 | (17,652 | ) | |||||||
| Shire: | |||||||||||
| License and milestone | — | 24,322 | (24,322 | ) | |||||||
| Cost-sharing, net | — | 624 | (624 | ) | |||||||
| Total Shire | — | 24,946 | (24,946 | ) | |||||||
| Total collaboration revenue | 14,632 | 57,230 | (42,598 | ) | |||||||
| Total revenue | $ | 14,632 | $ | 57,230 | $ | (42,598 | ) | ||||
Other Income (Expense), Net. Other income, net was $4.2$9.1 million in the year ended December 31, 2014,2016, compared to expense$3.0 million of $28.9 millionexpense in the year ended December 31, 2013.2015. This $33.1$12.1 million increase was primarily due to a $30.7decrease in the common warrant liability of $10.8 million difference indue to the effect of marking the common stock warrant liability to market in each period. The additional increase was a $1.2 million decrease in interest expense because the outstanding long term debt was retired during March 2014period, and an increase in interest income of $1.3 million for the effect of marking preferred stock warrantsmillion.
Income Tax Provision. Income tax provision is attributable to market prior to their conversion during the initial public offering.taxes on interest income from our investment portfolio.
Liquidity and Capital Resources
We have incurred losses and cumulative negative cash flows from operations since our inception in June 2003, and as of December 31, 2015,2017, we had an accumulated deficit of $307.5$473.0 million. We anticipate that we will continue to incur losses for at least the next several years. We expect that our research and development and general and administrative expenses will continue to increase and, as a result, we will need additional capital to fund our operations, which we may raise through a combination of the sale of equity, debt financings or other sources, including potential additional collaborations.
As of December 31, 2015,2017, our operations have been primarily funded by $105.1 million in equity investments from venture investors prior to theour IPO, $219.3$539.7 million from public investors, $64.2$123.7 million in equity investments from our partners and $233.5$273.7 million in upfront payments, milestones, and net research and development payments from our collaboration partners.
On January 11, 2016, we completed the sale of 3,750,000 shares of our common stock, at a public offering price of $40.00 per share, resulting in net proceeds to us of approximately $140.4 million, after deducting underwriting discounts and offering expenses.
As of December 31, 2015,2017, we had $136.0$372.9 million in cash, cash equivalents and investments. Cash in excess of immediate requirements is invested in accordance with our investment policy, primarily with a view to liquidity and capital preservation. Currently, our funds are held in bank deposits and money market mutual funds consisting of U.S. government-backed securities.
Cash Flows
The following table sets forth the primary sources and uses of cash for each of the years set forth below:
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| (in thousands) | 2015 | 2014 | 2013 | 2017 | 2016 | 2015 | ||||||||||||||||
| Net cash (used in) provided by: | ||||||||||||||||||||||
| Operating activities | $ | (44,211 | ) | $ | (53,220 | ) | $ | (19,650 | ) | $ | (76,730 | ) | $ | (44,695 | ) | $ | (44,211 | ) | ||||
| Investing activities | (108,971 | ) | (514 | ) | (307 | ) | (64,366 | ) | (108,805 | ) | (108,971 | ) | ||||||||||
| Financing activities | 4,505 | 117,031 | 93,509 | 220,296 | 146,667 | 4,505 | ||||||||||||||||
| Net (decrease) increase in cash and cash equivalents | $ | (148,677 | ) | $ | 63,297 | $ | 73,552 | |||||||||||||||
| Net increase (decrease) in cash and cash equivalents | $ | 79,200 | $ | (6,833 | ) | $ | (148,677 | ) | ||||||||||||||
Operating Activities.
Net cash used in operating activities was $76.7 million for the year ended December 31, 2017, and consisted primarily of a net loss of $108.5 million adjusted for non-cash items including: stock-based compensation expense of $28.2 million, depreciation and amortization expense of $2.8 million and an increase in fair value of warrants of $1.0 million.
Net cash used in operating activities was $44.7 million for the year ended December 31, 2016, and consisted primarily of a net loss of $57.0 million adjusted for non-cash items including a decrease in fair value of warrants of $7.3 million, stock-
based compensation expense of $18.6 million, depreciation and amortization of $1.7 million, and a net increase due to changes in operating assets and liabilities of $0.7 million. The significant items in the change in operating assets and liabilities include an increase in prepaid expenses and other assets of $1.4 million, a decrease in collaboration receivables of $0.4 million, an increase in accounts payable of $0.6 million, and a decrease in deferred revenue of $0.5 million.
Net cash used in operating activities was $44.2 million for the year ended December 31, 2015, and consisted primarily of a net loss of $63.9 million adjusted for non-cash items including an increase in fair value of warrants of $3.5 million, stock-based compensation expense of $12.1 million, depreciation and amortization of $1.2 million, and a net increase due to changes in operating assets and liabilities of $3.3 million. The significant items in the change in operating assets and liabilities include a decrease in deferred revenue of $1.2 million for the Celgene Collaboration.collaboration. Other components of the change in operating assets and liabilities include a decrease in deferred rent of $0.5 million and an increase in accrued expenses of $5.0 million.
Investing Activities.
Net cash used in investing activities was $109.0$64.4 million, $0.5$108.8 million and $0.3$109.0 million for the years ended December 31, 2015, 20142017, 2016 and 2013,2015, respectively. The significant increase during 2015 was due to the purchase of investments. During 2014 and 2013decrease in our net cash used in investing activities consisted ofduring the year ended December 31, 2017 compared to the year ended December 31, 2016 was primarily due to a decrease in net purchases of property$58.9 million in connection with managing our investment portfolio to meet our projected cash requirements. Our net cash used in investing activities during the years ended December 31, 2016 and equipment.2015 were due to the implementation of our investment policy pursuant to which we began to invest the money raised in our September 2013, January 2014, and January 2016 public offerings in marketable securities.
Financing Activities.
Net cash provided by financing activities was $220.3 million for the year ended December 31, 2017 and was primarily due to net proceeds of $215.8 million from our September 2017 public offering and the underwriters full exercise of the over-allotment option in the offering, as well as $4.5 million in cash proceeds from the exercise of stock options and the issuance of common stock related to the employee stock purchase plan.
Net cash provided by financing activities was $146.7 million for the year ended December 31, 2016 and was primarily due to net proceeds from our January 2016 public offering of $140.3 million, as well as $6.0 million in cash proceeds from the exercise of stock options and the issuance of commons stock related to the employee stock purchase plan.
Net cash provided by financing activities was $4.5 million for the year ended December 31, 2015 and was primarily due to $4.6 million in proceeds received from the employee stock purchase plan, and the exercise of stock options and warrants to purchase common stock.
Operating Capital Requirements
To date, we have not generated any revenue from product sales. We do not know when, or if, we will generate any revenue from product sales. We will not generate revenue from product sales unless and until we or our partners obtain regulatory approval of and commercialize one of our current or future therapeutic candidates. We anticipate that we will continue to generate losses for the foreseeable future, and we expect the losses to increase as we continue the development of, and seek and obtain regulatory approvals for dalantercept, ACE-083, sotatercept in pulmonary hypertension, ACE-2494 and any future therapeutic candidates, and begin to commercialize any approved products. We are subject to all of the risks incident in the development of therapeutic candidates, and we may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may adversely affect our business. Since the closing of our initial public offering, weWe have incurred, and expect to continue to incur, additional costs associated with operating as a public company. We anticipate that we will need additional funding in connection with our continuing operations.
We believe that our existing cash, cash equivalents and investments including the net proceeds from our January 2016 public offering, will be sufficient to fund our projected operating requirements into the second half of 2019.2021. However, we will require additional capital for the further development of our existing therapeutic candidates and may also need to raise additional funds sooner to pursue other development activities related to additional therapeutic candidates.
Until we can generate a sufficient amount of revenue from our products, if ever, we expect to fund our operations through a combination of equity offerings, debt financings or other sources including potential additional collaborations. Additional capital may not be available on favorable terms, if at all. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue the development or commercialization of one or more of our therapeutic candidates. If we raise additional funds through the issuance of additional debt or equity securities, it could result in dilution to our existing stockholders and increased fixed payment obligations, and these securities may have rights senior to those of our common stock. If we incur indebtedness, we could become subject to covenants that
would restrict our operations and potentially impair our competitiveness, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. We may not be able to enter into new collaboration arrangements for any of our proprietary therapeutic candidates. Any of these events could significantly harm our business, financial condition and prospects.
Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement and involves risks and uncertainties, and actual results could vary as a result of a number of factors. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect. Our future funding requirements, both near and long-term, will depend on many factors, including, but not limited to:
the achievement of milestones under our agreementagreements with Celgene;
the terms and timing of any other collaborative, licensing and other arrangements that we may establish;
the initiation, progress, timing and completion of preclinical studies and clinical trials for our therapeutic candidates and potential therapeutic candidates;
the number and characteristics of therapeutic candidates that we pursue;
the progress, costs and results of our clinical trials;
the outcome, timing and cost of regulatory approvals;
delays that may be caused by changing regulatory requirements;
the cost and timing of hiring new employees to support our continued growth;
the costs involved in filing and prosecuting patent applications and enforcing and defending patent claims;
the costs and timing of procuring clinical and commercial supplies of our therapeutic candidates;
the extent to which we acquire or invest in businesses, products or technologies; and
the costs involved in defending and prosecuting litigation regarding in-licensed intellectual property.
Contractual Obligations and Commitments
The following is a summary of our long-term contractual cash obligations as of December 31, 2015.2017.
| (in thousands) | Total | 2016 | 2017 through 2018 | 2019 through 2020 | After 2020 | Total | 2018 | 2019 through 2020 | 2021 through 2022 | After 2022 | ||||||||||||||||||||||||||||
| Operating lease obligations(1) | $ | 13,971 | $ | 4,386 | $ | 8,124 | $ | 1,296 | $ | 165 | $ | 35,536 | $ | 5,551 | $ | 12,826 | $ | 12,398 | $ | 4,761 | ||||||||||||||||||
| Less: sublease income(2) | (704 | ) | (704 | ) | — | — | — | |||||||||||||||||||||||||||||||
| Total | $ | 13,971 | $ | 4,386 | $ | 8,124 | $ | 1,296 | $ | 165 | $ | 34,832 | $ | 4,847 | $ | 12,826 | $ | 12,398 | $ | 4,761 | ||||||||||||||||||
| (1) | We lease office and lab space at 128 Sidney Street, |
| (2) | In July 2017, we entered into a sublease for 11,825 square feet of office and lab space at 99 Erie Street in Cambridge Massachusetts that expires in July 2018. On January 8, 2018, the sublessee exercised its right to extend the sublease by an additional five months through December 31, 2018. |
We also have obligations to make future payments to third party licensors that become due and payable on the achievement of certain development, regulatory and commercial milestones. We have not included these commitments on our consolidated balance sheet or in the table above because the achievement and timing of these milestones is not fixed or determinable. These commitments include the following:
Under our two license agreements with the Salk Institute for Biological Studies, or Salk, relating to the first cloning of the type II activin receptors, if we sublicense the Salk patent rights, we will owe Salk a percentage of sublicensing revenue, excluding payments based on sales. Under one agreement, we agreed to pay Salk specified development milestone payments totaling up to $2.0 million for sotatercept. Under the other agreement, we agreed to pay Salk specified development milestone payments of up to $0.7 million for luspatercept. In addition, under both agreements, we are required to pay Salk royalties in the low single-digits on worldwide net product sales by us or our sublicensees under the licensed patent rights of products claimed in the licensed patents, or products derived from use of the licensed patent rights, with royalty obligations for sotatercept continuing at a reduced rate for a period of time after patent expiration.
In May 2014, we executed a collaboration agreement with a research technology company. We paid an upfront and research fee of $0.3 million upon execution of the agreement. We also received an option to obtain a commercial license to the molecules developed during the collaboration, which, if exercised, would obligate us to pay royalty and milestone payments.
We enter into contracts in the normal course of business with CROs for clinical trials and clinical supply manufacturing and with vendors for preclinical safety and research studies, research supplies and other services and products for operating purposes. These contracts generally provide for termination on notice, and therefore are cancelable contracts and not included in the table of contractual obligations and commitments.
Net Operating Loss (NOL) Carryforwards
We have deferred tax assets of approximately $109.8$145.7 million as of December 31, 2015,2017, which have been fully offset by a valuation allowance due to uncertainties surrounding our ability to realize these tax benefits. The deferred tax assets are primarily composed of federal and state tax net operating loss, or NOL, carryforwards, research and development tax credit carryforwards, and deferred revenue, accruals and other temporary differences. As of December 31, 2015,2017, we had federal NOL carryforwards of approximately $276.1$438.0 million and state NOL carryforwards of $229.8$393.6 million available to reduce future taxable income, if any. These federal and state NOL carryforwards expire at various times through 2035.2037. In general, if we experience a greater than 50 percent aggregate change in ownership of certain significant stockholders over a three-year period, or a Section 382 ownership change, utilization of our pre-change NOL carryforwards are subject to an annual limitation under Section 382 of the Internal Revenue Code of 1986, as amended, and similar state laws. Such limitations may result in expiration of a portion of the NOL carryforwards before utilization and may be substantial. If we experience a Section 382 ownership change in connection with our public offerings or as a result of future changes in our stock ownership, some of which changes are outside our control, the tax benefits related to the NOL carryforwards may be limited or lost. For additional information about our taxes, see Note 13 to the financial statements in this Annual Report on Form 10-K.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined in the rules and regulations of the Securities and Exchange Commission.
Item 7A. Quantitative and Qualitative Disclosures About Market Risks
We are exposed to market risk related to changes in interest rates. As of December 31, 20152017 and December 31, 2014,2016, we had cash, cash equivalents and investments of $136.0$372.9 million and $176.5$234.4 million, respectively. Our cash equivalents are invested primarily in bank deposits and money market mutual funds. Our primary exposure to market risk is interest rate sensitivity, which is affected by changes in the general level of U.S. interest rates. Our investments are subject to interest rate risk and could fall in value if market interest rates increase. Due to the duration of our investment portfolio and the low risk profile of our investments, we do not believe an immediate 100 basis point change in interest rates would have a material effect on the fair market value of our portfolio. We have the ability to hold our investments until maturity, and therefore we would not expect our operating results or cash flows to be affected to any significant degree by the effect of a change in market interest rates on our investments.
Item 8. Financial Statements and Supplementary Data
All financial statements and supplementary data required to be filed hereunder are filed as listed under Item 15(a) of this Annual Report on Form 10-K and are incorporated herein by this reference.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures
As of December 31, 2015,2017, management, with the participation of our Chief Executive Officer and Chief Financial Officer, performed an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act. Our disclosure controls and procedures are designed to ensure that information required to be disclosed in the reports we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission's rules and forms, and that such information is accumulated and communicated to our management, including the Chief Executive Officer and the Chief Financial Officer, to allow timely decisions regarding required disclosures. Any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objective. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that, as of December 31, 2015,2017, the design and operation of our disclosure controls and procedures were effective.
Management's Report on Internal Control Over Financial Reporting
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rule 13a-15(f) and 15d-15(f) of the Exchange Act. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policy or procedures may deteriorate. Under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, management has conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 20152017 based upon the Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework). Based on this evaluation, management, including our Chief Executive Officer and Chief Financial Officer, has concluded that our internal control over financial reporting was effective as of December 31, 2015.2017.
The effectiveness of our internal control over financial reporting as of December 31, 20152017 has been audited by Ernst & Young LLP, our independent registered public accounting firm, as stated in their report which is included herein.
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting (as defined in Rule 13a-15(f) under the Exchange Act) during the year ended December 31, 2015,2017, that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Report of Independent Registered Public Accounting Firm
The Stockholders and the Board of Directors and Stockholders of
Acceleron Pharma Inc.
Opinion on Internal Control over Financial Reporting
We have audited Acceleron Pharma Inc.’s internal control over financial reporting as of December 31, 2015,2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, Acceleron Pharma Inc.’s (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2017, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of Acceleron Pharma Inc. as of December 31, 2017 and 2016, the related consolidated statements of operations and comprehensive loss, stockholders' equity and cash flows for each of the three years in the period ended December 31, 2017, and the related notes and our report dated February 27, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Overover Financial Reporting. Our responsibility is to express an opinion on the company’sCompany’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States).PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
| /s/ Ernst & Young LLP | ||
Boston, Massachusetts
February 25, 201627, 2018
Item 9B. Other Information
None.
PART III
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this Item 10 will be included in our definitive proxy statement to be filed with the Securities and Exchange Commission, or SEC, with respect to our 2018 Annual Meeting of Stockholders and is incorporated herein by reference to our 2016 Proxy Statement.reference.
Item 11. Executive Compensation
The information required by this Item 11 will be included in our definitive proxy statement to be filed with the SEC with respect to our 2018 Annual Meeting of Stockholders and is incorporated herein by reference to our 2016 Proxy Statement.reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this Item 12 will be included in our definitive proxy statement to be filed with the SEC with respect to our 2018 Annual Meeting of Stockholders and is incorporated herein by reference to our 2016 Proxy Statement.reference.
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this Item 13 will be included in our definitive proxy statement to be filed with the SEC with respect to our 2018 Annual Meeting of Stockholders and is incorporated herein by reference to our 2016 Proxy Statement.reference.
Item 14. Principal Accounting Fees and Services
The information required by this Item 14 will be included in our definitive proxy statement to be filed with the SEC with respect to our 2018 Annual Meeting of Stockholders and is incorporated herein by reference to our 2016 Proxy Statement.reference.
PART IV
Item 15. Exhibits and Financial Statement Schedules
| (a) | The following documents are filed as part of this report: |
| (1) | Financial Statements. |
| (2) | Financial Statement Schedules. |
We are not filing any financial statement schedules as part of this Annual Report on Form 10-K because they are not applicable or the required information is included in the consolidated financial statements or notes thereto.
| (3) | Exhibits. |
The list of Exhibits filed as a part of this Annual Report on Form 10-K is set forth on the Exhibit Index immediately preceding such Exhibits and is incorporated herein by this reference.
| (b) | The list of Exhibits filed as a part of this Annual Report on Form 10-K is set forth on the Exhibit Index immediately preceding such Exhibits and is incorporated herein by this reference. |
| (c) | None. |
Item 16. Form 10-K Summary
Acceleron Pharma Inc.
Index to Consolidated Financial Statements
Report of Independent Registered Public Accounting Firm
To The Stockholders and the Board of Directors and Stockholders of
Acceleron Pharma Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Acceleron Pharma Inc. (the Company) as of December 31, 20152017 and 2014, and2016, the related consolidated statements of operations and comprehensive loss, redeemable convertible preferred stock and stockholders’ equity (deficit) and cash flows for each of the three years in the period ended December 31, 2015. These2017, and the related notes (collectively referred to as the "consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the consolidated financial position of Acceleron Pharma Inc.the Company at December 31, 20152017 and 2014,2016, and the consolidated results of its operations and its cash flows for each of the three years in the period ended December 31, 2015,2017, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), Acceleron Pharma Inc.’sthe Company's internal control over financial reporting as of December 31, 2015,2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework)Framework) and our report dated February 25, 201627, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the US federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
| /s/ Ernst & Young LLP | ||
We have served as the Company's auditor since 2005.
Boston, Massachusetts
February 25, 201627, 2018
Acceleron Pharma Inc.
Consolidated Balance Sheets
(amounts in thousands except share and per share data)
| December 31, | December 31, | |||||||||||||
| 2015 | 2014 | 2017 | 2016 | |||||||||||
| Assets | ||||||||||||||
| Current assets: | ||||||||||||||
| Cash and cash equivalents | $ | 27,783 | $ | 176,460 | $ | 100,150 | $ | 20,950 | ||||||
| Short-term investments | 77,064 | — | 177,077 | 118,740 | ||||||||||
| Collaboration receivables (all amounts are with a related party) | 3,628 | 3,367 | 3,570 | 3,234 | ||||||||||
| Prepaid expenses and other current assets | 2,458 | 2,480 | 4,446 | 3,862 | ||||||||||
| Total current assets | 110,933 | 182,307 | 285,243 | 146,786 | ||||||||||
| Property and equipment, net | 3,106 | 3,087 | 6,966 | 5,201 | ||||||||||
| Long-term investments | 31,134 | — | 95,723 | 94,692 | ||||||||||
| Restricted cash | 796 | 902 | 1,132 | 946 | ||||||||||
| Other assets | 368 | — | 113 | 22 | ||||||||||
| Total assets | $ | 146,337 | $ | 186,296 | $ | 389,177 | $ | 247,647 | ||||||
| Liabilities and stockholders' equity | ||||||||||||||
| Current liabilities: | ||||||||||||||
| Accounts payable | $ | 875 | $ | 724 | $ | 1,086 | $ | 1,590 | ||||||
| Accrued expenses | 12,400 | 6,848 | 14,936 | 13,249 | ||||||||||
| Deferred revenue | 555 | 1,162 | 541 | 541 | ||||||||||
| Deferred rent | 661 | 519 | 182 | 769 | ||||||||||
| Total current liabilities | 14,491 | 9,253 | 16,745 | 16,149 | ||||||||||
| Deferred revenue, net of current portion | 4,239 | 4,816 | 3,161 | 3,704 | ||||||||||
| Deferred rent, net of current portion | 1,157 | 1,818 | 1,818 | 953 | ||||||||||
| Warrants to purchase common stock | 17,187 | 14,124 | 2,236 | 1,244 | ||||||||||
| Total liabilities | 37,074 | 30,011 | 23,960 | 22,050 | ||||||||||
| Commitments and contingencies (Note 8) | — | — | — | — | ||||||||||
| Stockholders' equity: | ||||||||||||||
| Undesignated preferred stock, $0.001 par value: 25,000,000 shares authorized and no shares issued or outstanding | — | — | — | — | ||||||||||
| Common stock, $0.001 par value: 175,000,000 shares authorized; 33,313,355 and 32,432,025, shares issued and outstanding at December 31, 2015 and 2014, respectively | 34 | 33 | ||||||||||||
| Common stock, $0.001 par value: 175,000,000 shares authorized; 45,261,175 and 38,251,826, shares issued and outstanding at December 31, 2017 and 2016, respectively | 46 | 39 | ||||||||||||
| Additional paid-in capital | 416,926 | 399,835 | 839,090 | 590,474 | ||||||||||
| Accumulated deficit | (307,477 | ) | (243,583 | ) | (473,024 | ) | (364,491 | ) | ||||||
| Accumulated other comprehensive loss | (220 | ) | — | (895 | ) | (425 | ) | |||||||
| Total stockholders' equity | 109,263 | 156,285 | 365,217 | 225,597 | ||||||||||
| Total liabilities and stockholders' equity | $ | 146,337 | $ | 186,296 | $ | 389,177 | $ | 247,647 | ||||||
See accompanying notes to consolidated financial statements.
Acceleron Pharma Inc.
Consolidated Statements of Operations and Comprehensive Loss
(amounts in thousands except per share data)
| Year Ended December 31, | |||||||||||
| 2015 | 2014 | 2013 | |||||||||
| Revenue: | |||||||||||
| Collaboration revenue: | |||||||||||
| License and milestone | $ | 1,184 | $ | 1,673 | $ | 43,948 | |||||
| Cost-sharing, net | 16,913 | 12,959 | 13,282 | ||||||||
| Total revenue(1) | 18,097 | 14,632 | 57,230 | ||||||||
| Costs and expenses: | |||||||||||
| Research and development | 58,404 | 50,897 | 36,051 | ||||||||
| Litigation settlement | — | 5,000 | — | ||||||||
| General and administrative | 20,572 | 14,199 | 14,227 | ||||||||
| Total costs and expenses | 78,976 | 70,096 | 50,278 | ||||||||
| (Loss) income from operations | (60,879 | ) | (55,464 | ) | 6,952 | ||||||
| Other (expense) income: | |||||||||||
| Other (expense) income, net | (3,527 | ) | 5,044 | (26,797 | ) | ||||||
| Interest income | 512 | 83 | 39 | ||||||||
| Interest expense | — | (922 | ) | (2,092 | ) | ||||||
| Total other (expense) income, net | (3,015 | ) | 4,205 | (28,850 | ) | ||||||
| Net loss | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | ||
| Other comprehensive loss: | |||||||||||
| Net unrealized holding (losses) on short-term and long-term investments during the period | $ | (220 | ) | $ | — | $ | — | ||||
| Comprehensive loss | $ | (64,114 | ) | $ | (51,259 | ) | $ | (21,898 | ) | ||
| Reconciliation of net loss to net loss applicable to common stockholders: | |||||||||||
| Net loss | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | ||
| Accretion of dividends, interest, redemption value and issuance costs on redeemable convertible preferred stock | — | — | (19,870 | ) | |||||||
| Gain on extinguishment of redeemable convertible preferred stock | — | — | 2,765 | ||||||||
| Net loss applicable to common stockholders—basic and diluted | $ | (63,894 | ) | $ | (51,259 | ) | $ | (39,003 | ) | ||
| Net loss per share applicable to common stockholders-basic and diluted | $ | (1.92 | ) | $ | (1.63 | ) | $ | (4.15 | ) | ||
| Weighted-average number of common shares used in computing net loss per share applicable to common stockholders-basic and diluted | 33,303 | 31,515 | 9,407 | ||||||||
| ___________________________________________________________ | |||||||||||
| (1) Includes related party revenue (Note 15) | $ | 18,097 | $ | 14,632 | $ | 32,284 | |||||
| Year Ended December 31, | |||||||||||
| 2017 | 2016 | 2015 | |||||||||
| Revenue: | |||||||||||
| Collaboration revenue: | |||||||||||
| License and milestone | $ | 541 | $ | 15,550 | $ | 1,184 | |||||
| Cost-sharing, net | 12,940 | 12,221 | 16,913 | ||||||||
| Total revenue (all amounts are with a related party) | 13,481 | 27,771 | 18,097 | ||||||||
| Costs and expenses: | |||||||||||
| Research and development | 89,726 | 68,580 | 58,404 | ||||||||
| General and administrative | 33,738 | 25,297 | 20,572 | ||||||||
| Total costs and expenses | 123,464 | 93,877 | 78,976 | ||||||||
| Loss from operations | (109,983 | ) | (66,106 | ) | (60,879 | ) | |||||
| Other (expense) income, net | (992 | ) | 7,262 | (3,527 | ) | ||||||
| Interest income | 2,553 | 1,854 | 512 | ||||||||
| Total other income (expense), net | 1,561 | 9,116 | (3,015 | ) | |||||||
| Loss before income taxes | (108,422 | ) | (56,990 | ) | (63,894 | ) | |||||
| Income tax provision | (32 | ) | (24 | ) | — | ||||||
| Net loss | $ | (108,454 | ) | $ | (57,014 | ) | $ | (63,894 | ) | ||
| Other comprehensive loss: | |||||||||||
| Net unrealized holding losses on short-term and long-term investments during the period | (470 | ) | (205 | ) | (220 | ) | |||||
| Comprehensive loss | $ | (108,924 | ) | $ | (57,219 | ) | $ | (64,114 | ) | ||
| Net loss per share- basic and diluted | $ | (2.68 | ) | $ | (1.52 | ) | $ | (1.92 | ) | ||
| Weighted-average number of common shares used in computing net loss per share- basic and diluted | 40,420 | 37,430 | 33,303 | ||||||||
See accompanying notes to consolidated financial statements.
Acceleron Pharma Inc.
Consolidated Statements of Redeemable Convertible Preferred Stock and Stockholders' Equity (Deficit)
Series A Redeemable Convertible Preferred Stock | Series B Redeemable Convertible Preferred Stock | Series C Redeemable Convertible Preferred Stock | Series C-1 Redeemable Convertible Preferred Stock | Series D Redeemable Convertible Preferred Stock | Series D-1 Redeemable Convertible Preferred Stock | ||||||||||||||||||||||||||||||||||||
Number of Shares | Value | Number of Shares | Value | Number of Shares | Value | Number of Shares | Value | Number of Shares | Value | Number of Shares | Value | ||||||||||||||||||||||||||||||
| December 31, 2012 | 6,410,976 | $ | 66,665 | 4,204,185 | $ | 67,044 | 2,978,062 | $ | 59,909 | 457,875 | $ | 9,387 | 234,940 | $ | 4,325 | 636,942 | $ | 11,864 | |||||||||||||||||||||||
| Accretion of dividends, interest, redemption value and issuance costs related to redeemable convertible preferred stock | — | 3,408 | — | 4,138 | — | 4,068 | — | 661 | — | 487 | — | 1,273 | |||||||||||||||||||||||||||||
| Repurchase and retirement of redeemable convertible preferred stock | — | — | (139,741 | ) | (2,267 | ) | (21,744 | ) | (445 | ) | — | — | (2,906 | ) | (54 | ) | — | — | |||||||||||||||||||||||
| Exercise of warrants to purchase convertible preferred stock | 46,668 | 678 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Exercise of warrants to purchase common stock | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Compensation expense associated with stock options | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Conversion of redeemable convertible preferred stock into common stock | (6,457,644 | ) | (70,751 | ) | (4,064,444 | ) | (68,915 | ) | (2,956,318 | ) | (63,532 | ) | (457,875 | ) | (10,048 | ) | (232,034 | ) | (4,758 | ) | (636,942 | ) | (13,137 | ) | |||||||||||||||||
| Reclassification of warrants to purchase shares of redeemable convertible preferred stock into warrants to purchase common stock | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Issuance of common stock in connection with initial public offering and private placement, net of issuance costs of $2,692 | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| December 31, 2013 | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Issuance of common stock net of expenses of $554 | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Exercise of warrants to purchase common stock | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Balance at December 31, 2014 | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Issuance of common stock related to ESPP | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Unrealized loss on available-for-sale securities | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Balance at December 31, 2015 | — | $ | — | — | $ | — | — | $ | — | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||||||||||
(amounts in thousands except share and per share data)
Series E Redeemable Convertible Preferred Stock | Series F Redeemable Convertible Preferred Stock | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Total Redeemable Convertible Preferred Stock | Common Stock | Common Stock | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Number of Shares | Value | Number of Shares | Value | Number of Shares | $0.001 Par Value | Additional Paid-In Capital | Accumulated Deficit | Comprehensive Income (Loss) | Total Stockholders' Equity (Deficit) | Number of Shares | $0.001 Par Value | Additional Paid-In Capital | Accumulated Deficit | Comprehensive Loss | Total Stockholders' Equity | |||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2012 | 816,060 | $ | 13,393 | 2,426,171 | $ | 36,023 | $ | 268,610 | 2,432,155 | $ | 3 | $ | — | $ | (290,976 | ) | $ | — | $ | (290,973 | ) | |||||||||||||||||||||||||||||||||||||||||
| Accretion of dividends, interest, redemption value and issuance costs related to redeemable convertible preferred stock | — | 1,802 | — | 4,033 | 19,870 | — | — | (4,230 | ) | (15,640 | ) | — | (19,870 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase and retirement of redeemable convertible preferred stock | (13,103 | ) | (224 | ) | (4,825 | ) | (74 | ) | (3,064 | ) | (722 | ) | — | 2,773 | — | — | 2,773 | |||||||||||||||||||||||||||||||||||||||||||||
| Exercise of warrants to purchase convertible preferred stock | — | — | — | — | 678 | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of warrants to purchase common stock | — | — | — | — | — | 23,735 | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Compensation expense associated with stock options | — | — | — | — | — | — | — | 2,196 | — | — | 2,196 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | 292,802 | — | 653 | — | — | 653 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Conversion of redeemable convertible preferred stock into common stock | (802,957 | ) | (14,971 | ) | (2,421,346 | ) | (39,982 | ) | (286,094 | ) | 18,516,993 | 19 | 149,885 | 136,190 | — | 286,094 | ||||||||||||||||||||||||||||||||||||||||||||||
| Reclassification of warrants to purchase shares of redeemable convertible preferred stock into warrants to purchase common stock | — | — | — | — | — | — | — | 2,012 | — | — | 2,012 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock in connection with initial public offering and private placement, net of issuance costs of $2,692 | — | — | — | — | — | 7,083,667 | 7 | 96,818 | — | — | 96,825 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (21,898 | ) | — | (21,898 | ) | |||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2013 | — | — | — | — | — | 28,348,630 | 29 | 250,107 | (192,324 | ) | — | 57,812 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock net of expenses of $554 | — | — | — | — | — | 2,760,000 | 3 | 129,171 | — | — | 129,174 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | — | — | 4,778 | — | — | 4,778 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | 853,507 | 1 | 3,207 | — | — | 3,208 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | — | — | — | — | — | 303,204 | — | 7,422 | — | — | 7,422 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of warrants to purchase common stock | — | — | — | — | — | 166,684 | — | 5,150 | — | — | 5,150 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (51,259 | ) | — | (51,259 | ) | |||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2014 | — | — | — | — | — | 32,432,025 | 33 | 399,835 | (243,583 | ) | — | 156,285 | 32,432,025 | $ | 33 | $ | 399,835 | $ | (243,583 | ) | $ | — | $ | 156,285 | ||||||||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | — | — | 12,075 | — | — | 12,075 | — | — | 12,075 | — | — | 12,075 | |||||||||||||||||||||||||||||||||||||||||||||
| Exercise of stock options | — | — | — | — | — | 837,361 | 1 | 3,962 | — | — | 3,963 | 837,361 | 1 | 3,962 | — | — | 3,963 | |||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock related to ESPP | — | — | — | — | — | 23,787 | — | 589 | — | — | 589 | 23,787 | — | 589 | — | — | 589 | |||||||||||||||||||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | — | — | — | — | — | 20,182 | — | 465 | — | — | 465 | 20,182 | — | 465 | — | — | 465 | |||||||||||||||||||||||||||||||||||||||||||||
| Unrealized loss on available-for-sale securities | — | — | — | — | — | — | — | — | — | (220 | ) | (220 | ) | — | — | — | — | (220 | ) | (220 | ) | |||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (63,894 | ) | — | (63,894 | ) | — | — | — | (63,894 | ) | — | (63,894 | ) | |||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2015 | — | $ | — | — | $ | — | $ | — | 33,313,355 | $ | 34 | $ | 416,926 | $ | (307,477 | ) | $ | (220 | ) | $ | 109,263 | 33,313,355 | 34 | 416,926 | (307,477 | ) | (220 | ) | 109,263 | |||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | 18,557 | — | — | 18,557 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock, net of expense $665 | 3,750,000 | 4 | 140,340 | — | — | 140,344 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of stock options | 885,075 | 1 | 5,311 | — | — | 5,312 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock related to ESPP | 30,671 | — | 658 | — | — | 658 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | 272,725 | — | 8,682 | — | — | 8,682 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized loss on available-for-sale securities | — | — | — | — | (205 | ) | (205 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | (57,014 | ) | — | (57,014 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2016 | 38,251,826 | 39 | 590,474 | (364,491 | ) | (425 | ) | 225,597 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | 28,248 | — | — | 28,248 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock, net of expenses of $397 | 6,216,216 | 6 | 215,796 | — | — | 215,802 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of stock options | 474,056 | 1 | 3,892 | — | — | 3,893 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vesting of restricted stock units | 282,158 | — | (226 | ) | — | — | (226 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock related to ESPP | 33,698 | — | 827 | — | — | 827 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net exercise of warrants to purchase common stock | 3,221 | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized loss on available-for-sale securities | — | — | — | — | (470 | ) | (470 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Effect of adoption of ASU 2016-09 | — | — | 79 | (79 | ) | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | (108,454 | ) | — | (108,454 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2017 | 45,261,175 | $ | 46 | $ | 839,090 | $ | (473,024 | ) | $ | (895 | ) | $ | 365,217 | |||||||||||||||||||||||||||||||||||||||||||||||||
See accompanying notes to consolidated financial statements.
Acceleron Pharma Inc.
Consolidated Statements of Cash Flows
(amounts in thousands)
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| 2015 | 2014 | 2013 | 2017 | 2016 | 2015 | |||||||||||||||||
| Operating Activities | ||||||||||||||||||||||
| Net loss | $ | (63,894 | ) | $ | (51,259 | ) | $ | (21,898 | ) | $ | (108,454 | ) | $ | (57,014 | ) | $ | (63,894 | ) | ||||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||||||||||||
| Depreciation and amortization | 1,176 | 1,118 | 915 | 2,825 | 1,676 | 1,176 | ||||||||||||||||
| Loss on disposition of property and equipment | 34 | 25 | 32 | |||||||||||||||||||
| Stock-based compensation | 12,075 | 4,778 | 2,196 | 28,248 | 18,557 | 12,075 | ||||||||||||||||
| Accretion of deferred interest | — | (536 | ) | 342 | ||||||||||||||||||
| Amortization of deferred debt issuance costs | — | 36 | 186 | |||||||||||||||||||
| Change in fair value of warrants | 3,528 | (5,037 | ) | 26,875 | 992 | (7,262 | ) | 3,528 | ||||||||||||||
| Gain on retirement of warrants | — | — | (76 | ) | ||||||||||||||||||
| Forgiveness of related party receivable | — | — | 237 | |||||||||||||||||||
| Net amortization of premium on investments | (406 | ) | — | — | ||||||||||||||||||
| Other non-cash items | 176 | (8 | ) | (372 | ) | |||||||||||||||||
| Changes in assets and liabilities: | ||||||||||||||||||||||
| Prepaid expenses and other current assets | 7 | (255 | ) | (823 | ) | (675 | ) | (1,411 | ) | 7 | ||||||||||||
| Collaboration receivables | (261 | ) | 249 | (840 | ) | |||||||||||||||||
| Related party receivable | — | — | (4 | ) | ||||||||||||||||||
| Collaboration receivables (all amounts are with a related party) | (336 | ) | 394 | (261 | ) | |||||||||||||||||
| Accounts payable | 151 | (161 | ) | (53 | ) | (504 | ) | 644 | 151 | |||||||||||||
| Accrued expenses | 4,976 | (17 | ) | 709 | 1,493 | 524 | 4,976 | |||||||||||||||
| Deferred revenue | (1,184 | ) | (1,673 | ) | (26,949 | ) | (543 | ) | (549 | ) | (1,184 | ) | ||||||||||
| Deferred rent | (519 | ) | (499 | ) | (499 | ) | 234 | (96 | ) | (519 | ) | |||||||||||
| Restricted cash | 106 | 11 | — | (186 | ) | (150 | ) | 106 | ||||||||||||||
| Net cash used in operating activities | (44,211 | ) | (53,220 | ) | (19,650 | ) | (76,730 | ) | (44,695 | ) | (44,211 | ) | ||||||||||
| Investing Activities | ||||||||||||||||||||||
| Purchase of investments | (134,697 | ) | — | — | (179,935 | ) | (218,314 | ) | (134,697 | ) | ||||||||||||
| Proceeds from maturities investments | 26,685 | — | — | |||||||||||||||||||
| Proceeds from sales and maturities of investments | 119,965 | 112,889 | 26,685 | |||||||||||||||||||
| Purchases of property and equipment | (959 | ) | (514 | ) | (307 | ) | (4,396 | ) | (3,380 | ) | (959 | ) | ||||||||||
| Net cash used in investing activities | (108,971 | ) | (514 | ) | (307 | ) | (64,366 | ) | (108,805 | ) | (108,971 | ) | ||||||||||
| Financing Activities | ||||||||||||||||||||||
| Proceeds from issuance of common stock from public offering, net of issuance costs | (47 | ) | 129,174 | 86,825 | 215,802 | 140,697 | (47 | ) | ||||||||||||||
| Proceeds from issuance of common stock from private placements | — | — | 10,000 | |||||||||||||||||||
| Payments of long-term debt | — | (16,331 | ) | (3,669 | ) | |||||||||||||||||
| Payments made to repurchase redeemable convertible preferred stock, common stock and warrants to purchase common stock | — | — | (300 | ) | ||||||||||||||||||
| Payments for withholding taxes on restricted stock units | (226 | ) | — | — | ||||||||||||||||||
| Proceeds from issuances of common stock related to employee stock purchase plan | 589 | — | — | 827 | 658 | 589 | ||||||||||||||||
| Proceeds from exercise of stock options and warrants to purchase common stock | 3,963 | 4,188 | 653 | 3,893 | 5,312 | 3,963 | ||||||||||||||||
| Net cash provided by financing activities | 4,505 | 117,031 | 93,509 | 220,296 | 146,667 | 4,505 | ||||||||||||||||
| Net (decrease) increase in cash and cash equivalents | (148,677 | ) | 63,297 | 73,552 | ||||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | 79,200 | (6,833 | ) | (148,677 | ) | |||||||||||||||||
| Cash and cash equivalents at beginning of year | 176,460 | 113,163 | 39,611 | 20,950 | 27,783 | 176,460 | ||||||||||||||||
| Cash and cash equivalents at end of year | $ | 27,783 | $ | 176,460 | $ | 113,163 | $ | 100,150 | $ | 20,950 | $ | 27,783 | ||||||||||
| Supplemental Disclosure of Cash Flow Information: | ||||||||||||||||||||||
| Cash paid for interest | $ | — | $ | 1,574 | $ | 1,657 | ||||||||||||||||
| Supplemental Disclosure of Non-Cash Investing and Financing Activities: | ||||||||||||||||||||||
| Accretion of dividends, interest, redemption value, and issuance costs on preferred stock | $ | — | $ | — | $ | 19,870 | ||||||||||||||||
| Conversion of preferred stock into common stock | $ | — | $ | — | $ | 286,094 | ||||||||||||||||
| Conversion of preferred stock warrants into common stock warrants | $ | — | $ | — | $ | 2,012 | ||||||||||||||||
| Reclassification of warrant liability to additional paid-in capital | $ | 465 | $ | 11,592 | $ | 678 | $ | — | $ | 8,682 | $ | 465 | ||||||||||
| Capitalized follow-on public offering costs included in accrued expenses | $ | 306 | $ | — | $ | 74 | $ | — | $ | — | $ | 306 | ||||||||||
| Purchase of property and equipment included in accounts payable and accrued expenses | $ | 270 | $ | 11 | $ | 297 | $ | 194 | $ | 397 | $ | 270 | ||||||||||
See accompanying notes to consolidated financial statements.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements
Years Ended December 31, 2015, 20142017, 2016 and 20132015
1. Nature of Business
Acceleron Pharma Inc. (Acceleron or the Company) is a Cambridge, Massachusetts-based clinical stage biopharmaceutical company focused ondedicated to the discovery, development and commercialization of novel therapeutic candidates that are based on the mechanisms that the human body usestherapeutics to regulate the growthtreat serious and repair of its cells and tissues.rare diseases. The Company's research focuses on key natural regulatorsleadership in the understanding of TGF-beta biology and protein engineering generates innovative compounds that engage the body's ability to regulate cellular growth and repair, particularly the Transforming Growth Factor-Beta (TGF-beta) protein superfamily. By combining its discovery and development expertise, including its proprietary knowledge of the TGF-beta superfamily, and its internal protein engineering and manufacturing capabilities, the Company has built a highly productive discovery and development platform that has generated innovative therapeutic candidates with novel mechanisms of action. The Company has four internally discovered therapeutic candidates that are currently in clinical trials.repair.
The Company is subject to risks common to companies in the biotechnology industry, including, but not limited to, the risk that the Company never achieves profitability, the need for substantial additional financing, risk of relying on third parties, risks of clinical trial failures, dependence on key personnel, protection of proprietary technology and compliance with government regulations.
2. Summary of Significant Accounting Policies
The accompanying consolidated financial statements reflect the application of certain significant accounting policies as described below and elsewhere in these notes to the consolidated financial statements. The Company believes that a significant accounting policy is one that is both important to the portrayal of the Company's financial condition and results, and requires management's most difficult, subjective, or complex judgments, often as the result of the need to make estimates about the effect of matters that are inherently uncertain.
Principles of Consolidation
The accompanying consolidated financial statements include those of the Company and its wholly-owned subsidiary, Acceleron Securities Corp. All significant intercompany balances and transactions have been eliminated in consolidation.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (GAAP). Any reference in these notes to applicable guidance is meant to refer to the authoritative United States generally accepted accounting principles as found in the Accounting Standards Codification (ASC) and Accounting Standards Update (ASU) of the Financial Accounting Standards Board (FASB).
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts expensed during the reporting period.
Management considers many factors in selecting appropriate financial accounting policies and controls, and in developing the estimates and assumptions that are used in the preparation of these consolidated financial statements. Management must apply significant judgment in this process. In addition, other factors may affect estimates, including: expected business and operational changes, sensitivity and volatility associated with the assumptions used in developing estimates, and whether historical trends are expected to be representative of future trends. The estimation process often may yield a range of potentially reasonable estimates of the ultimate future outcomes and management must select an amount that falls within that range of reasonable estimates. This process may result in actual results differing materially from those estimated amounts used in the preparation of the consolidated financial statements if these results differ from historical experience, or other assumptions do not turn out to be substantially accurate, even if such assumptions are reasonable when made.
In preparing these consolidated financial statements, management used significant estimates in the following areas, among others: revenue recognition, stock-based compensation expense, the determination of the fair value of stock-based awards, the fair value of liability-classified warrants, accrued expenses, and the recoverability of the Company's net deferred tax assets and related valuation allowance.
Collaboration Receivable
Credit is extended to customers based upon an evaluation of the customer's financial condition. Collaboration receivables are recorded at net realizable value. The Company does not charge interest on past due balances. Collaboration receivables are
F-7
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
determined to be past due when the payment due date is exceeded. The Company utilizes a specific identification accounts receivable reserve methodology based on a review of outstanding balances and previous activities to determine the allowance for doubtful accounts. The Company charges off uncollectible receivables at the time the Company determines the receivable is no longer collectible. The Company did not have an allowance for doubtful accounts at December 31, 20152017 or 2014.2016.
Segment Information
Operating segments are identified as components of an enterprise about which separate discrete financial information is available for evaluation by the chief operating decision maker, or decision making group, in making decisions on how to allocate resources and assess performance. The Company's chief operating decision maker is the chief executive officer. The Company and the chief executive officer view the Company's operations and manage its business as one operating segment, which is the discovery, development and commercialization of novel proteinhighly innovative therapeutics for cancerto treat serious and rare diseases. All material long-lived assets of the Company reside in the United States. The Company does use contract research organizations (CROs) and research institutions located outside the United States. Some of these expenses are subject to collaboration reimbursement which is presented as a component of cost sharing,cost-sharing, net in the consolidated statements of operations and comprehensive loss.
Cash, and Cash Equivalents and Restricted CashShort-term and Long-term Investments
The Company considers all highly liquid investments purchased with original maturities of 90 days or less at the date of acquisition to be cash equivalents. Cash and cash equivalents include cash held in banks and amounts held in interest-bearing money market accounts. Cash equivalents are carried at cost, which approximates their fair market value.
The Company determines the appropriate classification of marketable securities at the time of purchase and reevaluates such designation at each balance sheet date. The Company has classified all of its marketable securities at December 31, 2017 and 2016 as “available-for-sale” pursuant to ASC 320, Investments – Debt and Equity Securities. The Company records available-for-sale securities at fair value, with the unrealized gains and losses included in accumulated other comprehensive income (loss) in stockholders’ equity. There were no realized gains or losses on marketable securities for the years ended December 31, 2017, 2016 and 2015.
Investments not classified as cash equivalents are presented as either short-term or long-term investments based on both their maturities as well as the time period the Company intends to hold such securities.
The Company adjusts the cost of available-for-sale debt securities for amortization of premiums and accretion of discounts to maturity. The Company includes such amortization and accretion in interest income. The cost of securities sold is based on the specific identification method. The Company includes interest and dividends on securities classified as available-for-sale in interest income in the accompanying consolidated statements of operations and comprehensive loss.
The Company reviews marketable securities for other-than-temporary impairment whenever the fair value of a marketable security is less than the amortized cost and evidence indicates that a marketable security’s carrying amount is not recoverable within a reasonable period of time. Other-than-temporary impairments of investments are recognized in the consolidated statements of operations if the Company has experienced a credit loss, has the intent to sell the marketable security, or if it is more likely than not that the Company will be required to sell the marketable security before recovery of the amortized cost basis. Evidence considered in this assessment includes reasons for the impairment, compliance with the Company’s investment policy, the severity and the duration of the impairment and changes in value subsequent to the end of the period.
The aggregate fair value of securities held by the Company in an unrealized loss position for less than twelve months as of December 31, 2017 and December 31, 2016 was $193.6 million and $172.2 million, respectively. The aggregate fair value of securities held by the Company in an unrealized loss position for more than twelve months as of December 31, 2017 and December 31, 2016 was $67.0 million and $5.5 million, respectively. The aggregate unrealized loss for those securities in an unrealized loss position for more than twelve months was $0.3 million and $2,000, respectively. The Company evaluated its securities for other-than-temporary impairment and considered the decline in market value for the securities to be primarily attributable to current economic and market conditions. It is not more likely than not that the Company will be required to sell the securities, and the Company does not intend to do so prior to the recovery of the amortized cost basis. Based on this analysis, these marketable securities were not considered to be other-than-temporarily impaired as of December 31, 2017 and December 31, 2016.
Concentrations of Credit Risk and Off-Balance Sheet Risk
F-8
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
The Company has no off-balance sheet risk, such as foreign exchange contracts, option contracts, or other foreign hedging arrangements. Financial instruments that potentially subject the Company to concentrations of credit risk are primarily cash, cash equivalents, restricted cash, short-term and long-term investments and collaboration receivables. The Company maintains its cash and cash equivalent balances in the form of money market accountsand short-term and long-term investments with financial institutions that management believes are creditworthy. Short-term and long-term investments consist of investment grade corporate obligations, treasury notes, asset backed securities, and certificates of deposit. The Company's investment policy includes guidelines on the quality of the institutions and financial instruments and defines allowable investments that the Company believes minimizes the exposure to concentrations of credit risk.
The Company routinely assesses the creditworthiness of its customers and collaboration partners. The Company has not experienced any material losses related to receivables from individual customers and collaboration partners, or groups of customers. The Company does not require collateral. Due to these factors, no additional credit risk beyond amounts provided for collection losses is believed by management to be probable in the Company's collaboration receivables.
Disclosure of Fair Value of Financial Instruments
The carrying amounts of the Company's financial instruments which include cash, cash equivalents, certificates of deposit,short-term and long-term investments, collaboration receivables, common stock warrants, accounts payable, and accrued expenses, approximated their fair values at December 31, 2015 or 2014 due to the short-term nature of these instruments.expenses. See discussion below on the determination of the fair value of the Company's preferred and common stock warrants.
warrants and short-term and long-term investments. The carrying value of the remainder of the Company's financial instruments approximated their fair values at December 31, 2017 and 2016 due to the short-term nature of these instruments. The Company has evaluated the estimated fair value of financial instruments using available market information and management's estimates. The use of different market assumptions and/or estimation methodologies could have a significant effect on the estimated fair value amounts.
Fair Value Measurements
ASC Topic 820, Fair Value Measurement (ASC 820), establishes a fair value hierarchy for instruments measured at fair value that distinguishes between assumptions based on market data (observable inputs) and the Company's own assumptions (unobservable inputs). Observable inputs are inputs that market participants would use in pricing the asset or liability based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company's assumptions about the inputs that market participants would use in pricing the asset or liability, and are developed based on the best information available in the circumstances.
ASC 820 identifies fair value as the exchange price, or exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As a basis for considering market participant assumptions in fair value measurements, ASC 820 establishes a three-tier fair value hierarchy that distinguishes between the following:
Level 1—Quoted market prices in active markets for identical assets or liabilities.
Level 2—Inputs other than Level 1 inputs that are either directly or indirectly observable, such as quoted market prices, interest rates, and yield curves.
Level 3—Unobservable inputs developed using estimates of assumptions developed by the Company, which reflect those that a market participant would use.
To the extent that the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument's level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Items measured at fair value on a recurring basis include warrants to purchase redeemable convertible preferred stock, which were outstanding until the closing of the IPO,short-term and long-term investments (Note 5), and warrants to purchase common stock (Note 7). During the periods presented, the Company has not changed the manner in which it values assets and liabilities that are measured at fair value using Level 3 inputs.
The following tables set forth the Company's financial instruments carried at fair value using the lowest level of input applicable to each financial instrument as of December 31, 20152017 and 2014 and2016 (in thousands):
| December 31, 2015 | |||||||||||||||
| Quoted Prices in Active Markets for Identical Items (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Total | ||||||||||||
| Assets: | |||||||||||||||
| Money market funds | $ | 24,811 | $ | — | $ | — | $ | 24,811 | |||||||
| Corporate obligations | — | 67,706 | — | 67,706 | |||||||||||
| U.S. Treasury securities | — | 10,991 | — | 10,991 | |||||||||||
| Certificates of deposit | — | 16,776 | — | 16,776 | |||||||||||
| Mortgage and other asset backed securities | — | 13,228 | — | 13,228 | |||||||||||
| Restricted cash | 796 | — | — | 796 | |||||||||||
| Total assets | $ | 25,607 | $ | 108,701 | $ | — | $ | 134,308 | |||||||
| Liabilities: | |||||||||||||||
| Warrants to purchase common stock | $ | — | $ | — | $ | 17,187 | $ | 17,187 | |||||||
| Total liabilities | $ | — | $ | — | $ | 17,187 | 17,187 | ||||||||
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
| December 31, 2014 | |||||||||||||||
| Quoted Prices in Active Markets for Identical Items (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Total | ||||||||||||
| Assets: | |||||||||||||||
| Money market funds | $ | 169,679 | $ | — | $ | — | $ | 169,679 | |||||||
| Restricted cash | 902 | — | — | 902 | |||||||||||
| Total assets | $ | 170,581 | $ | — | $ | — | $ | 170,581 | |||||||
| Liabilities: | |||||||||||||||
| Warrants to purchase common stock | $ | — | $ | — | $ | 14,124 | $ | 14,124 | |||||||
| Total liabilities | $ | — | $ | — | $ | 14,124 | $ | 14,124 | |||||||
Year Ended December 31, | |||||||
| 2015 | 2014 | ||||||
| Beginning balance | $ | 14,124 | $ | 30,753 | |||
| Change in fair value | 3,528 | (5,037 | ) | ||||
| Exercises | (465 | ) | (11,592 | ) | |||
| Repurchases | — | — | |||||
| Conversions | — | — | |||||
| Ending balance | $ | 17,187 | $ | 14,124 | |||
| December 31, 2017 | |||||||||||||||
| Quoted Prices in Active Markets for Identical Items (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Total | ||||||||||||
| Assets: | |||||||||||||||
| Money market funds | $ | 90,702 | $ | — | $ | — | $ | 90,702 | |||||||
| Corporate obligations | — | 158,849 | — | 158,849 | |||||||||||
| U.S. Treasury securities | — | 37,813 | — | 37,813 | |||||||||||
| Certificates of deposit | — | 12,244 | — | 12,244 | |||||||||||
| Mortgage and other asset backed securities | — | 67,888 | — | 67,888 | |||||||||||
| Restricted cash | 1,132 | — | — | 1,132 | |||||||||||
| Total assets | $ | 91,834 | $ | 276,794 | $ | — | $ | 368,628 | |||||||
| Liabilities: | |||||||||||||||
| Warrants to purchase common stock | $ | — | $ | — | $ | 2,236 | $ | 2,236 | |||||||
| Total liabilities | $ | — | $ | — | $ | 2,236 | $ | 2,236 | |||||||
| December 31, 2016 | |||||||||||||||
| Quoted Prices in Active Markets for Identical Items (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Total | ||||||||||||
| Assets: | |||||||||||||||
| Money market funds | $ | 19,818 | $ | — | $ | — | $ | 19,818 | |||||||
| Corporate obligations | — | 88,492 | — | 88,492 | |||||||||||
| U.S. Treasury securities | — | 33,968 | — | 33,968 | |||||||||||
| Certificates of deposit | — | 23,373 | — | 23,373 | |||||||||||
| Mortgage and other asset backed securities | — | 67,599 | — | 67,599 | |||||||||||
| Restricted cash | 946 | — | — | 946 | |||||||||||
| Total assets | $ | 20,764 | $ | 213,432 | $ | — | $ | 234,196 | |||||||
| Liabilities: | |||||||||||||||
| Warrants to purchase common stock | $ | — | $ | — | $ | 1,244 | $ | 1,244 | |||||||
| Total liabilities | $ | — | $ | — | $ | 1,244 | $ | 1,244 | |||||||
The money market funds noted above are included in cash and cash equivalents in the accompanying consolidated balance sheets. The Company recognizes transfers between levels of the fair value hierarchy as of the end of the reporting period. There were no transfers within the hierarchy during the years ended December 31, 20152017 and 2014.2016.
The following table sets forth a summary of changes in the fair value of the Company's common stock warrant liabilities, which represent a recurring measurement that is classified within Level 3 of the fair value hierarchy, wherein fair value is estimated using significant unobservable inputs (in thousands):
F-10
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
Year Ended December 31, | |||||||
| 2017 | 2016 | ||||||
| Beginning balance | $ | 1,244 | $ | 17,187 | |||
| Change in fair value | 992 | (7,262 | ) | ||||
| Exercises | — | (8,681 | ) | ||||
| Ending balance | $ | 2,236 | $ | 1,244 | |||
The fair value of the warrants to purchase common stock on the date of issuance and on each re-measurement date for those warrants to purchase common stock classified as liabilities was estimated using either the Monte Carlo simulation framework, which incorporates future financing events over the remaining life of the warrants to purchase common stock, or for certain re-measurement dates, including December 31, 2015 and 2014, due to the warrants being deeply in the money, the Black-Scholes option pricing model was used.model. The Black-Scholes method of valuation involves using inputs such as the fair value of the Company's stock, stock price volatility, the contractual term of the warrants, risk freerisk-free interest rates, and dividend yields. At each reporting period the companyCompany evaluates the best valuation methodology. At December 31, 2017, the Black-Scholes option pricing model was used, at December 31, 2016 the Monte Carlo simulation framework was used, and at December 31, 2015, the Black-Scholes option pricing model was used. Due to the nature of these inputs, the valuation of the warrants is considered a Level 3 measurement.
See Note 7 for further discussions of the accounting for the warrants, as well as for a summary of the significant inputs and assumptions used to determine the fair value of the warrants.
The Company measures eligible assets and liabilities at fair value, with changes in value recognized in earnings. Fair value treatment may be elected either upon initial recognition of an eligible asset or liability or, for an existing asset or liability, if an event triggers a new basis of accounting. The Company did not elect to remeasure any of its existing financial assets or liabilities, and did not elect the fair value option for any financial assets and liabilities transacted in the years ended December 31, 20152017 or 2014.2016.
Property and Equipment
Property and equipment is stated at cost. Maintenance and repairs that do not improve or extend the lives of the respective assets are expensed to operations as incurred. Upon disposal, retirement or sale the related cost and accumulated depreciation is
removed from the accounts and any resulting gain or loss is included in the results of operations. Depreciation and amortization is calculated using the straight-line method over the estimated useful lives of the assets, which are as follows:
| Asset | Estimated Useful Life |
| Computer equipment and software | 3 years |
| Office and laboratory equipment | 3 years |
| Leasehold improvements | Shorter of the useful life or remaining lease term |
The Company reviews long-lived assets when events or changes in circumstances indicate the carrying value of the assets may not be recoverable. Recoverability is measured by comparison of the book values of the assets to future net undiscounted cash flows that the assets are expected to generate. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the book value of the assets exceed their fair value, which is measured based on the projected discounted future net cash flows arising from the assets. No impairment losses have been recorded during the years ended December 31, 2015, 20142017, 2016 and 2013.2015.
Revenue Recognition
The company has primarily generated revenue through collaboration, license and research arrangements with collaboration partners for the development and commercialization of therapeutic candidates.
The Company recognizes revenue in accordance with FASB ASC Topic 605, Revenue Recognition. Accordingly, revenue is recognized for each unit of accounting when all of the following criteria are met: (1) persuasive evidence of an arrangement exists; (2) delivery has occurred or services have been rendered; (3) the fee is fixed or determinable; and (4) collectability is reasonably assured.
F-11
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
Amounts received prior to satisfying the revenue recognition criteria are recorded as deferred revenue in the Company's consolidated balance sheets. Amounts expected to be recognized as revenue within the 12 months following the balance sheet date are classified as deferred revenue, current portion. Amounts not expected to be recognized as revenue within the 12 months following the balance sheet date are classified as deferred revenue, net of current portion.
Multiple Element Revenue Arrangements
The Company enters into collaboration agreements from time to time, which are more fully described in Note 10. The arrangements generally contain multiple elements or deliverables, which may include (1) licenses, or options to obtain licenses, to the Company's technology, (2) research and development activities performed for the collaboration partner, (3) participation on Joint Development Committees, and (4) the manufacturing of clinical or preclinical material. Payments pursuant to these arrangements typically include non-refundable, up-frontupfront payments, milestone payments upon achieving significant development events, research and development reimbursements, sales milestones, and royalties on future product sales.
Effective January 1, 2011, the Company adopted ASU No. 2009-13, Multiple-Deliverable Revenue Arrangements (ASU 2009-13), which amends ASC Topic 605-25, Revenue Recognition—Multiple Element Arrangements (ASC 605-25). The Company applies this guidance to new arrangements as well as existing agreements that are significantly modified after January 1, 2011. For agreements that are significantly modified, the Company determines the estimated selling price for the remaining undelivered elements as of the date of the material modification and allocates arrangement consideration based upon the estimated selling price to the undelivered elements.
The application of the multiple element guidance requires subjective determinations, and requires management to make judgments about the individual deliverables, and whether such deliverables are separable from the other aspects of the contractual relationship. Deliverables are considered separate units of accounting provided that: (1) the delivered item(s) has value to the customer on a stand-alone basis and (2) if the arrangement includes a general right of return relative to the delivered item(s), delivery or performance of the undelivered item(s) is considered probable and substantially in the control of the Company. In determining the units of accounting, management evaluates certain criteria, including whether the deliverables have stand-alone value, based on the consideration of the relevant facts and circumstances for each arrangement, such as the research, manufacturing and commercialization capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. In addition, the Company considers whether the collaboration partner can use the other deliverable(s) for their intended purpose without the receipt of the remaining element(s), whether the value of the deliverable is dependent on the undelivered item(s) and whether there are other vendors that can provide the undelivered element(s).
Arrangement consideration that is fixed or determinable is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria, as described above, are applied to each of the separate units of accounting in determining the appropriate period or pattern of recognition.
The Company determines the estimated selling price for deliverables within each agreement using vendor-specific objective evidence (VSOE) of selling price, if available, third-party evidence (TPE) of selling price if VSOE is not available, or management's best estimate of selling price (BESP) if neither VSOE nor TPE is available. Subsequent to the adoption of ASU 2009-13, the Company typically uses BESP to estimate the selling price of the deliverables. Determining the BESP for a unit of accounting requires significant judgment. In developing the BESP for a unit of accounting, the Company considers applicable market conditions and relevant entity-specific factors, including factors that were contemplated in negotiating the agreement with the customer and estimated costs. The Company validates the BESP for units of accounting by evaluating whether changes in the key assumptions used to determine the BESP will have a significant effect on the allocation of arrangement consideration between multiple units of accounting.
The Company typically receives up-front,upfront, non-refundable payments when licensing its intellectual property in conjunction with a collaboration agreement. When management believes the license to its intellectual property does not have stand-alone value from the other deliverables to be provided in the arrangement, the Company generally recognizes revenue attributed to the license on a straight-line basis over the contractual or estimated performance period, which is typically the term of the Company's research and development or manufacturing obligations. The Company continually evaluates these periods, and will adjust the period of revenue recognition if circumstances change. When management believes the license to its intellectual property has stand-alone value, the Company generally recognizes revenue attributed to the license upon delivery.
Research and development funding is recognized as revenue in the period that the related services are performed. When the Company acts as the principal under its collaboration agreements, it records payments received for the reimbursement of research and development costs as cost-sharing revenue in the consolidated statements of operations and comprehensive loss.
F-12
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
To the extent that the Company reimburses the collaborator for costs incurred, the Company records these costs as a reduction of cost-sharing revenue.
The Company's agreements may contain options which provide the collaboration partner the right to obtain additional licenses. Options are considered substantive if, at the inception of the arrangement, the Company is at risk as to whether the collaboration partner will choose to exercise the option. Factors considered in evaluating whether an option is substantive include the overall objective of the arrangement, the benefit the collaborator might obtain from the arrangement without exercising the option, the cost to exercise the option and the likelihood that the option will be exercised. For arrangements under which an option is considered substantive, the Company does not consider the item underlying the option to be a deliverable at the inception of the arrangement and the associated option fees are not included in allocable arrangement consideration, assuming the option is not priced at a significant and incremental discount. Conversely, for arrangements under which an option is not considered substantive or if an option is priced at a significant and incremental discount, the Company would consider the item underlying the option to be a deliverable at the inception of the arrangement and a corresponding amount would be included in allocable arrangement consideration.
Effective January 1, 2011, the Company adopted ASU No. 2010-17, Revenue Recognition—Milestone Method (ASU 2010-17). At the inception of each arrangement that includes milestone payments, the Company evaluates, with respect to each milestone, whether the milestone is substantive and at-risk. This evaluation includes an assessment of whether (a) the consideration is commensurate with either (1) the entity's performance to achieve the milestone, or (2) the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting at least in part from the entity's performance to achieve the milestone, (b) the consideration relates solely to past performance, and (c) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. The Company evaluates factors such as the scientific, regulatory, commercial, and other risks that must be overcome to achieve the respective milestone, the level of effort and investment required to achieve the respective milestone, and whether the milestone consideration is reasonable relative to all deliverables and payment terms in the arrangement in making this assessment. On the milestone achievement date, assuming all other revenue recognition criteria are met and the milestone is deemed substantive and at-risk, the Company recognizes the payment as license and milestone revenue. For milestones that are not deemed substantive and at-risk, where payment is reasonably assured, the Company recognizes the milestone payment over the remaining service period.
Sales and commercial milestones and royalties will be recognized when and if earned, provided collectability is reasonably assured.
Research and Development Expenses
Research and development costs are charged to expense as costs are incurred in performing research and development activities. Research and development costs include all direct costs, including salaries, stock compensation and benefits for research and development personnel, outside consultants, costs of clinical trials, sponsored research, clinical trials insurance, other outside costs, depreciation and facility costs related to the development of drug candidates. The Company records up-front,upfront, non-refundable payments made to outside vendors, or other payments made in advance of services performed or goods being delivered, as prepaid expenses, which are expensed as services are performed or the goods are delivered.
Certain research and development projects are, or have been, partially funded by collaboration agreements, and the expenses related to these activities are included in research and development costs. The Company records the related reimbursement of research and development costs under these agreements as revenue, as more fully described above and in Note 10.
Stock-Based Compensation
At December 31, 2015,2017, the Company had two stock-based compensation plans, which are more fully described in Note 11. The Company accounts for stock-based compensation in accordance with the provisions of ASC Topic 718, Compensation—Stock Compensation (ASC 718), which requires the recognition of expense related to the fair value of stock-based compensation awards in the consolidated statements of operations and comprehensive loss.
For stock optionsstock-based awards issued to employees and members of the BoardCompany's board of directors (the Board) for their services on the Board and for participation in the employee stock purchase plan, the Company estimates the grant date fair value of each option award using the Black-Scholes option-pricingoption pricing model. The use of the Black-Scholes option-pricingoption pricing model requires management to make assumptions with respect to the expected term of the option, the expected volatility of the common stock consistent with the expected life of the option, risk-free interest rates and expected dividend yields of the
F-13
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
common stock. For awards subject to service-based vesting conditions, the Company recognizes stock-based compensation expense, net of estimated forfeitures, equal to the grant date fair value of stock options on a straight-line basis over the requisite service period, which is generally the vesting term. For awards subject to both performance and service-based vesting conditions, the Company recognizes stock-based compensation expense using an accelerated recognition method when it is probable that the performance condition will be achieved. If achievement of the performance condition is not probable, but the award will vest based on the service condition, expense is recognized over the requisite service period. Forfeitures are required to be estimated at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates.
Share-based payments issued to non-employees are recorded at their fair values, and are periodically revalued as the equity instruments vest and are recognized as expense over the related service period in accordance with the provisions of ASC 718 and ASC Topic 505 (ASC 505), Equity. For stock-based awards granted to non-employees, the Company recognizes stock-based compensation expense using an accelerated recognition method.
Effective January 1, 2017, the Company adopted ASU 2016-09, Compensation - Stock Compensation, Improvements to Employee Share-Based Payment Accounting. In connection with the adoption of this standard, the Company records actual forfeitures as they occur, rather than estimating forfeitures by applying a forfeiture rate.
See Note 11 for a discussion of the assumptions used by the Company in determining the grant date fair value of options granted under the Black-Scholes option pricing model, as well as a summary of the stock option activity under the Company's stock-based compensation plans for the year ended December 31, 2015.2017.
Income Taxes
Income taxes are recorded in accordance with ASC Topic 740, Income Taxes (ASC 740), which provides for deferred taxes using an asset and liability approach. The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or tax returns. Deferred tax assets and liabilities are determined based on the difference between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. Valuation allowances are provided, if based upon the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company accounts for uncertain tax positions in accordance with the provisions of ASC 740. When uncertain tax positions exist, the Company recognizes the tax benefit of tax positions to the extent that the benefit will more likely than not be realized. The determination as to whether the tax benefit will more likely than not be realized is based upon the technical merits
of the tax position as well as consideration of the available facts and circumstances. As of December 31, 20152017 or 2014,2016, the Company does not have any significant uncertain tax positions.
Net Income (Loss)Loss Per Share
The following is a summary of the common stock equivalents which were excluded from the calculation of diluted net loss per share for the periods indicated (in thousands):
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||
| 2015 | 2014 | 2013 | 2017 | 2016 | 2015 | |||||||||||
| Outstanding stock options | 3,191 | 3,230 | 3,709 | 3,452 | 3,316 | 3,191 | ||||||||||
| Common stock warrants | 398 | 422 | 908 | 61 | 64 | 398 | ||||||||||
| Shares issuable under employee stock purchase plan | 15 | 14 | — | 18 | 23 | 15 | ||||||||||
| Restricted stock units | 521 | — | — | 604 | 732 | 521 | ||||||||||
| Preferred stock | — | — | 13,218 | |||||||||||||
| Preferred stock warrants | — | — | 114 | |||||||||||||
| 4,125 | 3,666 | 17,949 | ||||||||||||||
| Total excluded common stock equivalents | 4,135 | 4,135 | 4,125 | |||||||||||||
Comprehensive Income (Loss)Loss
F-14
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
Comprehensive income (loss)loss is defined as the change in equity of a business enterprise during a period from transactions, other events, and circumstances from non-owner sources. Comprehensive loss consists of net loss and other comprehensive loss, which includes certain changes in equity that are excluded from net loss. Comprehensive loss has been disclosed in the accompanying consolidated statements of operations and comprehensive loss. Accumulated other comprehensive loss is presented separately on the consolidated balance sheets and consists entirely of unrealized holdings gains (losses)losses on investments as of December 31, 2015.2017 and 2016.
Subsequent Events
The Company considers events or transactions that occur after the balance sheet date but prior to the issuance of the financial statements to provide additional evidence relative to certain estimates or to identify matters that require additional disclosure. Subsequent events have been evaluated as required. The Company has evaluated all subsequent events and determined that there are no material recognized or unrecognized subsequent events other than as disclosed within the notes to the consolidated financial statements. See Note 16.
Recently Adopted Accounting Pronouncements
From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies and adopted by the Company as of the specified effective date. Unless otherwise discussed, the Company believes that the impact of recently issued standards that are not yet effective will not have a material impact on its financial position or results of operations upon adoption.
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers (Topic 606), which supersedes all existing revenue recognition requirements, including most industry-specific guidance. The new standard requires a company to recognize revenue when it transfers goods or services to customers in an amount that reflects the consideration that the company expects to receive for those goods or services. The new standard will be effective for the Company on January 1, 2018. Topic 606 allows for either a full retrospective application, which the standard is applied to all periods presented, or a modified retrospective application, in which the standard is applied to the most current period presented in the financial statements. As of December 31, 2017, revenue is generated exclusively from the Company's collaboration agreement with Celgene. The Company is currently evaluating the method of adoption and the potential impact that Topic 606 may have on its financial position and results of operations.operations as it relates to this single arrangement, and expects to elect the modified retrospective application as its transition method.
In August 2014,February 2016, the FASB issued ASU No. 2014-15, 2016-02,Presentation of Financial Statements—Going Concern (Subtopic 205-40). The ASU requires all entities to evaluate for the existence of conditions or events that raise substantial doubt about the entity’s ability to continue as a going concern within one year after the issuance date of the financial statements. The accounting standard is effective for interim and annual periods ending after December 15, 2016, and will not have a material impact on the consolidated financial statements, but may impact the Company’s footnote disclosures.
In November 2015,2016, the FASB issued ASU No. 2015-17, Income Taxes2016-18, Statement of Cash Flows - Restricted Cash (Topic 740), Balance Sheet Classification of Deferred Taxes230). TheThis new standard requires that deferred tax assetscompanies to include amounts generally described as restricted cash and liabilities be classified as non-currentrestricted cash equivalents in a classifiedcash and cash equivalents when reconciling beginning-of-period and end-of-period total amounts shown on the statement of financial position. The new standard will becash flows. This guidance is effective for the Company on January 1, 2017.annual and interim reporting periods beginning after December 15, 2017, and requires retrospective application. The Company is currently evaluatingassessing the method of adoption and the potential impact that Topic 740 mayadopting ASU 2016-18 will have on its consolidated financial positionstatements and results of operations.related disclosures.
In March 2017, the FASB issued ASU 2017-08, Receivables - Nonrefundable Fees and Equipment, NetOther Costs (Subtopic 310-20): Premium Amortization on Purchased Callable Debt Securities. This new standard shortens the amortization period for certain callable debt securities held at a premium. Specifically, the amendment requires the premium to be amortized to the earliest call date. The amendment does not require an accounting change for securities held at a discount; the discount continues to be amortized to maturity. The guidance is effective for annual and interim periods beginning after December 15, 2018, and early
| December 31, | |||||||
| 2015 | 2014 | ||||||
| Computer equipment and software | $ | 1,163 | $ | 956 | |||
| Office equipment | 260 | 213 | |||||
| Laboratory equipment | 11,458 | 11,311 | |||||
| Leasehold improvements | 9,990 | 9,930 | |||||
| Construction in progress | 390 | 11 | |||||
| Total property and equipment | 23,261 | 22,421 | |||||
| Accumulated depreciation and amortization | (20,155 | ) | (19,334 | ) | |||
| Property and equipment, net | $ | 3,106 | $ | 3,087 | |||
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
adoption is permitted. The amendment should be applied on a modified retrospective basis, with the cumulative-effect adjustment directly to retained earnings as of the beginning of the period of adoption. The Company is currently assessing the impact that adopting ASU 2017-08 will have on its consolidated financial statements and related disclosures.
3. Property and Equipment, Net
Property and equipment, net, consists of the following (in thousands):
| December 31, | |||||||
| 2017 | 2016 | ||||||
| Computer equipment and software | $ | 1,501 | $ | 1,260 | |||
| Office equipment | 522 | 519 | |||||
| Laboratory equipment | 17,268 | 13,204 | |||||
| Leasehold improvements | 11,501 | 11,126 | |||||
| Construction in progress | 584 | 849 | |||||
| Total property and equipment | 31,376 | 26,958 | |||||
| Accumulated depreciation and amortization | (24,410 | ) | (21,757 | ) | |||
| Property and equipment, net | $ | 6,966 | $ | 5,201 | |||
Depreciation and amortization expense was $2.8 million, $1.7 million and $1.2 million for the years ending December 31, 2017, 2016 and 2015 respectively.
4. Restricted Cash
As of December 31, 2015,2017, and 2014,2016, the Company maintained letters of credit totaling $0.8$1.1 million and $0.9 million respectively, held in the form of certificates of deposit as collateral for the Company's facility lease obligations and its credit cards.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
5. Cash, Cash Equivalents and Short-term and Long-term Investments
The following is a summary of cash, cash equivalents and available-for-sale securities as of December 31, 20152017 and December 31, 20142016 (in thousands):
| December 31, 2015 | December 31, 2017 | |||||||||||||||||||||||||||||
| Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | |||||||||||||||||||||||
| Cash and cash equivalents due in 90 days or less | $ | 27,783 | $ | — | $ | — | $ | 27,783 | $ | 100,150 | $ | — | $ | — | $ | 100,150 | ||||||||||||||
| Available-for-sale securities: | ||||||||||||||||||||||||||||||
| Corporate obligations due in one year or less | 53,243 | — | (81 | ) | 53,162 | 99,792 | — | (219 | ) | 99,573 | ||||||||||||||||||||
| Corporate obligations due in more than one year | 14,112 | — | (72 | ) | 14,040 | 57,537 | — | (261 | ) | 57,276 | ||||||||||||||||||||
| U.S. Treasury securities due in one year or less | 6,016 | — | (4 | ) | 6,012 | 27,987 | — | (93 | ) | 27,894 | ||||||||||||||||||||
| U.S. Treasury securities due in more than one year | 4,995 | — | (15 | ) | 4,980 | 9,968 | — | (48 | ) | 9,920 | ||||||||||||||||||||
| Certificates of deposit due in one year or less | 11,890 | — | — | 11,890 | 10,529 | — | — | 10,529 | ||||||||||||||||||||||
| Certificates of deposit due in more than one year | 4,886 | — | — | 4,886 | 1,715 | — | — | 1,715 | ||||||||||||||||||||||
| Mortgage and other asset backed securities due in one year or less | 6,010 | — | (10 | ) | 6,000 | 39,236 | — | (155 | ) | 39,081 | ||||||||||||||||||||
| Mortgage and other asset backed securities due in more than one year | 7,266 | — | (38 | ) | 7,228 | 26,931 | — | (119 | ) | 26,812 | ||||||||||||||||||||
| Total available-for-sale securities | $ | 108,418 | $ | — | $ | (220 | ) | $ | 108,198 | $ | 273,695 | $ | — | $ | (895 | ) | $ | 272,800 | ||||||||||||
| Total cash, cash equivalents and available-for-sale securities | $ | 136,201 | $ | — | $ | (220 | ) | $ | 135,981 | $ | 373,845 | $ | — | $ | (895 | ) | $ | 372,950 | ||||||||||||
| December 31, 2014 | December 31, 2016 | |||||||||||||||||||||||||||||
| Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | |||||||||||||||||||||||
| Cash and cash equivalents due in 90 days or less | $ | 176,460 | $ | — | $ | — | $ | 176,460 | $ | 20,950 | $ | — | $ | — | $ | 20,950 | ||||||||||||||
| Total cash and cash equivalents | $ | 176,460 | $ | — | $ | — | $ | 176,460 | ||||||||||||||||||||||
| Available-for-sale securities: | ||||||||||||||||||||||||||||||
| Corporate obligations due in one year or less | 45,839 | 1 | (58 | ) | 45,782 | |||||||||||||||||||||||||
| Corporate obligations due in more than one year | 42,895 | — | (185 | ) | 42,710 | |||||||||||||||||||||||||
| U.S. Treasury securities due in one year or less | 22,490 | — | (10 | ) | 22,480 | |||||||||||||||||||||||||
| U.S. Treasury securities due in more than one year | 11,541 | — | (53 | ) | 11,488 | |||||||||||||||||||||||||
| Certificates of deposit due in one year or less | 13,562 | — | — | 13,562 | ||||||||||||||||||||||||||
| Certificates of deposit due in more than one year | 9,811 | — | — | 9,811 | ||||||||||||||||||||||||||
| Mortgage and other asset backed securities due in one year or less | 36,948 | — | (32 | ) | 36,916 | |||||||||||||||||||||||||
| Mortgage and other asset backed securities due in more than one year | 30,771 | — | (88 | ) | 30,683 | |||||||||||||||||||||||||
| Total available-for-sale securities | $ | 213,857 | $ | 1 | $ | (426 | ) | $ | 213,432 | |||||||||||||||||||||
| Total cash, cash equivalents and available-for-sale securities | $ | 234,807 | $ | 1 | $ | (426 | ) | $ | 234,382 | |||||||||||||||||||||
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
6. Accrued Expenses
Accrued expenses consist of the following (in thousands):
| December 31, | December 31, | |||||||||||||
| 2015 | 2014 | 2017 | 2016 | |||||||||||
| Research and development related | $ | 4,938 | $ | 2,291 | $ | 4,014 | $ | 5,238 | ||||||
| Employee compensation | 3,551 | 3,050 | 6,809 | 4,840 | ||||||||||
| Professional services | 1,198 | 426 | 1,183 | 739 | ||||||||||
| Other | 2,713 | 1,081 | 2,930 | 2,432 | ||||||||||
| $ | 12,400 | $ | 6,848 | |||||||||||
| Total accrued expenses | $ | 14,936 | $ | 13,249 | ||||||||||
7. Warrants
Below is a summary of the number of shares issuable upon exercise of outstanding warrants and the terms and accounting treatment for the outstanding warrants (in thousands, except per share data):
| Warrants as of | Warrants as of | |||||||||||||||||||||||||||||
Weighted- Average Exercise | Balance Sheet Classification | Weighted- Average Exercise | Balance Sheet Classification | |||||||||||||||||||||||||||
| December 31, 2015 | December 31, 2014 | Price Per Share | Expiration | December 31, 2015 | December 31, 2014 | December 31, 2017 | December 31, 2016 | Price Per Share | Expiration | December 31, 2017 | December 31, 2016 | |||||||||||||||||||
| Warrants to purchase common stock | 393 | 409 | 5.88 | June 10, 2020 - July 9, 2020 | Liability(2) | Liability(2) | 61 | 61 | $ | 5.88 | June 10, 2020 - July 9, 2020 | Liability | Liability | |||||||||||||||||
| Warrants to purchase common stock | 5 | 13 | 4.00 - 7.40 | March 31, 2016 - December 31, 2017 | Equity(1) (3) (4) | Equity(1) (3) (4) | — | 4 | 4.00 - 7.40 | March 28, 2017 - December 31, 2017 | Equity(1) (2) (3) | Equity(1) (2) (3) | ||||||||||||||||||
| All warrants | 398 | 422 | $ | 5.88 | 61 | 65 | $ | 5.88 | ||||||||||||||||||||||
| (1) | In March Stock. |
| (2) | In |
| (3) |
Warrants to purchase common stock were issued in connection with various debt financing transactions that were consummated our initial public offering. |
In connection with the Series E redeemable convertible preferred stock (Series E Preferred Stock) financing transactions that took place in June 2010 and July 2010, the Company issued warrants to purchase up to 871,580 shares of common stock. Each warrant was immediately exercisable and expires ten years from the original date of issuance. The warrants to purchase shares of the Company's common stock have an exercise price equal to the estimated fair value of the underlying instrument as of the initial date such warrants were issued. Each warrant is exercisable on either a physical settlement or net share settlement basis from the date of issuance. The warrant agreement contains a provision requiring an adjustment to the number of shares in the event the Company issues common stock, or securities convertible into or exercisable for common stock, at a price per share lower than the warrant exercise price. The Company concluded the anti-dilution feature required the warrants to be classified as liabilities under ASC Topic 815, Derivatives and Hedging—Contracts in Entity's Own Equity (ASC 815). The warrants are measured at fair value, with changes in fair value recognized as a gain or loss to other income (expense) in the consolidated statements of operations and comprehensive loss for each reporting period thereafter. The fair value of the common stock warrants werewas recorded as a discount to the preferred stock issued, of $3.0 million, and the preferred stock were beingwas accreted to the redemption value. At the end of each reporting period, the Company remeasured the fair value of the outstanding warrants, using current assumptions, resulting in an increase (decrease) in fair value of $3.5$1.0 million, $(5.0)$(7.3) million and $26.9$3.5 million, respectively, which was recorded in other income (expense), net in the accompanying consolidated statements of operations and comprehensive loss for the years ended December 31, 2015, 20142017, 2016 and 2013.2015. The Company will continue to re-measure the fair value of the liability associated with the warrants to purchase common stock at the end of each reporting period until the earlier of the exercise or the expiration of the applicable warrants. On March 31, 2013, the Company retired 13,994 warrants to purchase common stock as a consequence of a repurchase of shares from an investor. All remaining outstanding warrants were fully vested and exercisable as of December 31, 2015.2017.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
8. Commitments and Contingencies
Operating Leases
The Company leases its facilities under non-cancelable operating leases that expire at various dates through March 2021.September 2023. All of the Company's leases contain escalating rent clauses, which require higher rent payments in future years. The Company expenses rent on a straight-line basis over the term of the lease, including any rent-free periods. In addition, the Company received certain lease incentives, and recorded these incentives as deferred rent, which is amortized as a reduction of rent expense over the life of the lease. Rent expense of approximately $3.4$4.8 million, $3.3$3.8 million and $3.5$3.4 million were incurred during the years ended December 31, 2015, 20142017, 2016 and 2013,2015, respectively.
Future annual minimum lease payments as of December 31, 2015,2017, are as follows (in thousands):
| 2016 | 4,386 | |||||
| 2017 | 4,547 | |||||
| 2018 | 3,577 | $ | 5,551 | |||
| 2019 | 640 | 6,320 | ||||
| 2020 | 656 | 6,506 | ||||
| 2021 | 165 | 6,191 | ||||
| 2022 | 6,207 | |||||
| 2023 | 4,761 | |||||
| Total | $ | 13,971 | $ | 35,536 | ||
Future annual minimum sublease proceeds expected as of credit in the amount of $0.2 million, naming the landlordDecember 31, 2017 are as beneficiary. follows (in thousands):
| 2018 | $ | 704 | |
| Total | $ | 704 | |
Legal Proceedings
Other
The Company is also party to various agreements, principally relating to licensed technology, that require future payments relating to milestones not met at December 31, 20152017 and 2014,2016, or royalties on future sales of specified products. No milestone or royalty payments under these agreements are expected to be payable in the immediate future. See Note 10 for discussion of these arrangements.
The Company enters into standard indemnification agreements in the ordinary course of business. Pursuant to the agreements, the Company indemnifies, holds harmless, and agrees to reimburse the indemnified party for losses suffered or incurred by the indemnified party, generally the Company's business partners or customers, in connection with any U.S. patent or any copyright or other intellectual property infringement claim by any third party with respect to the Company's products. The term of these indemnification agreements is generally perpetual any time after execution of the agreement. The maximum potential amount of future payments the Company could be required to make under these indemnification agreements is unlimited. The Company has never incurred costs to defend lawsuits or settle claims related to these indemnification agreements.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
9. Stockholders' Equity
On January 11, 2016, the Company completed the sale of 3,750,000 shares of common stock at a public offering price of $40.00 per share, resulting in net proceeds to the Company of approximately $140.3 million.
On September 25, 2017, the Company completed the sale of 5,405,406 shares of common stock at a public offering price of $37.00 per share. On October 4, 2017, in connection with the September 2017 public offering, the underwriters fully exercised their option to purchase an additional 810,810 shares of common stock. The total net proceeds to the Company from the September 2017 public offering and the underwriters' exercise of their option to purchase additional shares of common stock was $215.8 million.
Preferred Stock
The Company’s certificate of incorporation authorizes its board of directorsthe Board to issue up to 25,000,000 shares of preferred stock from time to time in one or more series. The rights, preferences, restrictions, qualifications and limitations of such stock are determined by the board of directors.Board. As of December 31, 20152017 no shares are issued or outstanding.
Common Stock
The holders of shares of common stock are entitled to one vote for each share of common stock held at all meetings of stockholders and written actions in lieu of meetings.
The holders of shares of common stock are entitled to receive dividends, if and when declared by the Board. No dividends have been declared or paid by the Company through December 31, 2015.2017.
Common Stock Reserved for Future Issuance
At December 31, 2015,2017, the Company has reserved for future issuance the following number of shares of common stock (in thousands):
| December 31, | ||
| Outstanding stock options to purchase common stock | ||
| Outstanding restricted stock units | 604 | |
| Shares available for future issuance under | ||
| Warrants to purchase common stock | ||
| Shares available for future issuance under the employee stock purchase plan | ||
| Additional shares reserved for unissued, but designated, Preferred Stock | 25,000 | |
| Total shares of authorized common stock reserved for future issuance | ||
10. Significant Agreements
Celgene
Overview
On February 20, 2008 the Company entered into a collaboration, license, and optionan agreement (the Sotatercept Agreement) with Celgene Corporation (Celgene) relating to sotatercept.sotatercept (the Original Sotatercept Agreement), which was amended on August 2, 2011 (as amended, the Amended Sotatercept Agreement). The Company further amended and restated the Original Sotatercept Agreement in its entirety on September 18, 2017, (the Restated Sotatercept Agreement). On August 2, 2011 the Company entered into a second collaboration, license and option agreement with Celgene for luspatercept, (the Luspatercept Agreement), and also amended certain terms.
Restated Sotatercept Agreement
The Restated Sotatercept Agreement provides Celgene with an exclusive license to sotatercept outside of the Sotatercept Agreement. These agreements provide Celgene exclusive licenses for sotaterceptfield of pulmonary hypertension, referred to as the PH field, and luspatercept in all indications, as well as exclusiveprovides the Company with the worldwide rights to obtain a license to certain future compounds. Celgene is a global biopharmaceutical company primarily engageddevelop and commercialize sotatercept in the discovery, development and commercialization of innovative therapies designed to treat cancer and immune-inflammatory related diseases.PH field.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
In connection with entering into the Restated Sotatercept Agreement, Celgene purchased 457,875 shares of Series C-1 Preferred Stock atagreed not to develop or commercialize in PH field any compound developed under the aggregate purchase price of $5.0 million. The Series C-1 Preferred Stock was purchased at an amount that was deemed to represent fair value at the time of purchase. Per our agreement and concurrent with the IPO, Celgene purchased 666,667 shares of Common Stock at the IPO offer price of $15.00 per share for $10.0 million.
The Company is responsible for 100% of the costs related to its development and commercialization of sotatercept in the PH field. If sotatercept is commercialized to treat pulmonary hypertension and the Company recognizes such revenue, then Celgene will be eligible to receive a royalty in the low 20% range on global net sales. In certain circumstances Celgene may recognize revenue related to the commercialization of sotatercept in the PH field, and in this scenario, the Company will be eligible to receive a royalty from Celgene such that the economic position of the parties is equivalent to the scenario in which the Company recognizes such revenue. With respect to the development and commercialization of sotatercept outside of the PH field or the development and commercialization of any other compound under the Restated Sotatercept Agreement, the terms of the Amended Sotatercept Agreement, described below, remained unchanged.
Pursuant to the Restated Sotatercept Agreement, Celgene will provide the Company with certain quantities of Celgene’s existing clinical supply of sotatercept for development in the PH field at no cost. For clinical or commercial supply of sotatercept in excess of that which is agreed to under the Restated Sotatercept Agreement, Celgene can elect to provide the Company with such clinical and commercial supply of sotatercept at a negotiated price or provide a tech transfer to enable the Company to manufacture on its own behalf. The conduct of the collaboration is managed by a Joint Development Committee and Joint Commercialization Committee. In the event of a deadlock of a committee, the Company shall determine the resolution of issues specifically related to the PH field, (other than pricing which shall be determined by consensus), and Celgene shall determine the resolution of all other issues. The Joint Commercialization Committee will oversee commercialization of sotatercept, and sotatercept pricing will be determined by mutual agreement of the Company and Celgene agreed to modifyin the terms of the Sotatercept Agreement. The modified terms included: (1) a change to the responsibility for development costs to align with the Luspatercept Agreement, with Celgene responsible for more than half of the worldwide costs through December 31, 2012, and 100% of the development costs thereafter, (2) future contingent development milestones for sotatercept were amended to a two-category (oncology and non-oncology) structure with potential future clinical milestones of $27.0 million and regulatory milestones of $190.0 million from a four-category (various cancer indications) structure and, (3) future contingent development milestones for option compounds were amended to a two-category (oncology and non-oncology) structure with potential future clinical milestones of $25.5 million and regulatory milestones of $142.5 million from a four-category (various cancer indications) structure, and (4) an option to buy down tiered royalty payments on both sotatercept and luspatercept with a one-time $25.0 million payment on or prior to January 1, 2013. The potential commercial milestones remained unchanged. To date, the Company has received $43.0 million in research and development funding and milestone payments for sotatercept under the original and modified agreements, of which $7.0 million is a clinical milestone payment earned in December 2013 that resulted from Celgene's start of a Phase 2b clinical trial in chronic kidney disease. The next likely clinical milestone payment would be $10.0 million and result from Celgene's start of a Phase 3 study in chronic kidney disease.Joint Commercialization Committee.
The Restated Sotatercept Agreement will expire on a country-by-country basis on the occurrence of boththe latest to occur of the following: (1) the expiration of the royalty term with respect to all license products outside the PH field in such country, (2) the expiration of the royalty term with respect to all sotatercept licensed products in the PH field in such country, and (2)(3) the exercise or forfeiture by Celgene of its option with regard to each option compound. TheIn the PH field, the royalty term for each licensed product in each country is the period commencing with first commercial sale of the applicable licensed product in the applicable country and ending on the latest of expiration of specified patent coverage or a specified period of years. Outside the PH field, the royalty term for each licensed product in each country outside North America is the period commencing with first commercial sale of the applicable licensed product in the applicable country and ending on the latest of expiration of specified patent coverage or a specified period of years. Theyears, and the royalty term for each licensed product in North America is the period commencing with the first commercial sale in North America and ending, on a licensed product and country-by-country basis on the date which commercialization of such licensed product has ceased. The term for each option compound runs for a specified period of years unless Celgene exercises its option, in which case the compound becomes a licensed product, or forfeits its option by failing to make certain payments following the achievement of certain milestones in early clinical development of the option compound.
The Restated Sotatercept Agreement is terminable by either party upon a breach that is uncured and continuing or by Celgene has the right tofor convenience on a country-by-country or product-by-product basis, or in its entirety. Celgene may also terminate the agreementRestated Sotatercept Agreement, in its entirety or on a product-by-product basis, for failure of a product to meet a development or clinical trial endpoint. Termination for cause by the Company or termination by Celgene for convenience or failure to meet an endpoint will have the effect of terminating the applicable license to Celgene and the rights granted to the Company with respect to onethe development of sotatercept in the PH field shall become irrevocable. Termination for cause by either party shall result in reducing the remaining royalties due to the breaching party by a certain percentage. Upon termination by Celgene for convenience or more licensed targets or in its entirety, upon 180 days' notice (or 45 days' notice if the licensed product has failedfor failure to meet certain end point criteria with respect to clinical trials or other development activities). The agreement may also be terminated in its entirety by either Celgene oran endpoint, the Company and Celgene will enter into a termination agreement pursuant to which, among other things, Celgene will continue to be eligible to receive a royalty in the eventlow 20% range on global net sales of a material breach by the other party orsotatercept in the event of a bankruptcy filingPH field.
The Company was not required to make any upfront payments to Celgene upon execution of the other party. There are no cancellation, termination or refund provisionsRestated Sotatercept Agreement, and is not be required to make any milestone payments to Celgene in this arrangement that contain material financial consequences toconnection with its development and commercialization of sotatercept in the Company.PH field.
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2015, 20142017, 2016 and 20132015
As a result of the changes to the economic terms under the Restated Sotatercept Agreement, the Company concluded the modification did not represent a material modification under ASU 2009-13. Further, the Company concluded there are no new deliverables which rise from the agreement. There have been no material changes to the accounting under the Amended Sotatercept Agreement pursuant to the Restated Sotatercept Agreement or the Luspatercept Agreement.
Original and Amended Sotatercept Agreement
Under the Original Sotatercept Agreement, as preserved by the Amended Sotatercept Agreement, the Company granted Celgene an exclusive license to sotatercept in all indications and an option to license discovery stage compounds against three specified targets. Celgene paid $45.0 million of nonrefundable, upfront license and option payments to the Company and bought $5.0 million of equity upon closing in February 2008. Per the Original Sotatercept Agreement, concurrent with the Company's 2013 IPO, Celgene purchased an additional $10.0 million of the Company's common stock.
The Company retained responsibility for research and development of sotatercept through the end of Phase 2a clinical trials, as well as manufacturing the clinical supplies for these trials. These activities were substantially completed in 2011. Celgene will be responsible for any sotatercept Phase 3 clinical trials, as well as any additional Phase 2 clinical trials and is responsible for manufacturing or overseeing the manufacture of Phase 3 and commercial supplies. Commensurate with the execution of the Luspatercept Agreement described below, in August 2011 the Company and Celgene agreed to modify the terms of the collaboration. Outside of the PH field, the significant financial terms of the Amended Sotatercept Agreement, which were preserved in the Restated Sotatercept Agreement, are:
Since January 1, 2013, Celgene has been responsible for paying 100% of worldwide development costs for the sotatercept program;
Celgene will be responsible for all commercialization costs worldwide;
The Company will be eligible to receive tiered royalty payments in the low-to-mid 20% percent range on net sales of sotatercept subject to certain reductions, including for entry of a generic product onto the market; and
The Company is obligated to co-promote sotatercept and future products in all fields, in each case if approved, in North America, and Celgene will pay all costs related thereto;
The Amended Sotatercept Agreement, as preserved in the Restated Sotatercept Agreement, contains a two-category contingent development milestone structure outside of the PH field (oncology and non-oncology) for sotatercept, including future clinical milestones of up to $27.0 million, regulatory milestones of up to $190.0 million and commercial milestones of up to $150.0 million. Additionally, the Company is eligible to receive option fees of up to $30.0 million for each of the three discovery-stage targets, and for all three discovery-stage targets in the aggregate, clinical milestones of up to $25.5 million, regulatory milestones of up to $142.5 million and commercial milestones of up to $150.0 million. None of the three discovery stage programs has advanced to the stage to achieve payment of a milestone, nor does the Company expect any such milestone payments in the near future.
As of December 31, 2017, the Company has received $44.2 million in research and development funding and milestone payments for the sotatercept program. The next likely clinical milestone payment would be $10.0 million and result from Celgene's start of a Phase 3 study with sotatercept outside of the PH field.
Luspatercept Agreement
Under the terms of the Luspatercept Agreement, the Company and Celgene collaborate worldwide for the joint development and commercialization of luspatercept. The Company also granted Celgene an option for future products for which Acceleron files an Investigational New Drug application for the treatment of anemia. Celgene paid $25.0 million on the closing of the Luspatercept Agreement in August 2011.
The Company retainsretained responsibility for research and development through the end of Phase 1 and the Company's initial luspatercept beta-thalassemia and luspatercept MDS Phase 2 clinical trials, as well as manufacturing the clinical supplies for these studies. Celgene will conduct subsequent Phase 2 and Phase 3 clinical studies. Acceleron will manufacture luspatercept for the Phase 1studies and Phase 2 clinical trials and Celgene will be responsible for overseeing the manufacture of Phase 3 and commercial supplies by third party contract manufacturing organizations. The significant financial terms of the Luspatercept Agreement are:
Since January 1, 2013, Celgene has been responsible for paying 100% of worldwide development costs for the luspatercept program
Celgene will be responsible for all commercialization costs worldwide;
F-22
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
The Company will be eligible to receive tiered royalty payments in the low-to-mid 20% percent range on net sales of luspatercept subject to certain reductions, including for entry of a generic product onto the market; and
The Company is obligated to co-promote luspatercept and future products in all fields, in each case if approved, in North America, and Celgene will pay all costs related thereto;
The Company is eligible to receive clinical milestones of up to $32.5 million, regulatory milestones of up to $105.0 million and commercial milestones of up to $80.0 million for luspatercept. The Company will receive additional, lower development, regulatory, and commercial milestones for any additional products for the treatment of anemia on which Celgene exercises an option. Clinical milestone payments are triggered upon initiation of a defined phase of clinical research for a therapeutic candidate. Regulatory milestone payments are triggered upon the acceptance of the marketing application and upon approval to market a therapeutic candidate by the FDA or other global regulatory authorities. Commercial milestone payments are triggered when an approved pharmaceutical product reaches certain defined levels of net sales by Celgene in countries outside of North America. In addition, to the extent luspatercept is commercialized, the Company would be entitled to receive tiered royalty payments in the low-to-mid twenty percent range of net sales from sales generated from all geographies. Royalty payments are subject to certain reductions, including for entry of a generic product onto the market.
The Luspatercept Agreement will expire on a country-by-country basis on the occurrence of both of the following: (1) the expiration of the royalty term with respect to all license products in such country, and (2) the end of the option term. The royalty term for each licensed product in each country outside North America is the period commencing with first commercial sale of the applicable licensed product in the applicable country and ending on the latest of expiration of specified patent coverage or a specified period of years. The royalty term for each licensed product in North America is the period commencing with the first commercial sale in North America and ending, on a licensed product and country-by-country basis on the date which commercialization of such licensed product has ceased. The option term runs until the later of (1) the date on which no development or commercialization activities are ongoing or are expected to commence for any licensed products under the Luspatercept Agreement; (2) the date on which no development or commercialization activities are ongoing or are expected to commence for any licensed products under the Restated Sotatercept Agreement and all option rights under the Restated Sotatercept Agreement have been forfeited with respect to each option compound where Celgene has made a payment with respect to such compound; and (3) the royalty term for all licensed products under the Luspatercept Agreement and the Restated Sotatercept Agreement has ended; provided that if at the time the option term would otherwise end any option compounds under the Luspatercept Agreement are in clinical development the option term shall continue until Celgene's rights to such compound are either exercised or forfeited.
Celgene has the right to terminate the Luspatercept Agreement with respect to one or more licensed targets or in its entirety, upon 180 days' notice (or 45 days' notice if the licensed product has failed to meet certain end point criteria with respect to clinical trials or other development activities), provided that Celgene may not terminate the Luspatercept Agreement prior to the completion of the on-going luspatercept beta-thalassemia and luspatercept MDS Phase 2 clinical trials, except under certain conditions.. The agreement may also be terminated in its entirety by either Celgene or the Company in the event of a material breach by the other party or in the event of a bankruptcy filing of the other party. There are no cancellation, termination or refund provisions in this arrangement that contain material financial consequences to the Company.
Accounting Analysis
Prior to 2011, the Company accounted for the Original Sotatercept Agreement, as a multiple element arrangement under ASC 605-25 (prior to the amendments of ASU 2009-13). The Company identified the following deliverables under the arrangement; (1)the license to the ActRIIA compound, (2) the right to license option program compounds, (3) participation in the joint development committee, (4) participation in the joint commercialization committee and (5) research and development activities. Under the provisions of ASC 605-25, applicable to the arrangement, since the Company could not establish vendor-specific objective evidence (VSOE)VSOE for the undelivered elements, the Company was required to recognize the initial consideration, consisting of the $45.0 million of nonrefundable upfront license and option payments, over the period the undelivered elements were to be delivered, which was initially estimated to be 15 years. As of the date of the modification of the agreement, there was approximately $34.7 million of deferred revenue under the arrangement.
As a result of the materialAmended Sotatercept Agreement in August 2011, the Company concluded the modifications to the cost sharingcost-sharing obligations, milestone payments structure and royalty payment structure the Company concluded the modification represented a significant modification under ASU 2009-13, which required the Company to apply the updated provisions of ASU 2009-13 subsequent to the modification.
Because the Luspatercept Agreement and the amendment to theAmended Sotatercept Agreement were negotiated in contemplation of each other, and the Company had not yet completed all of its obligations pursuant to the Original Sotatercept Agreement, the
F-23
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
agreements were considered one arrangement for accounting purposes. The deliverables under the combined arrangement include: (1) licenses to develop and commercialize sotatercept and luspatercept, (2) performance of research and development services, (3) participation on the joint development committees, and (4) the performance of manufacturing services to provide clinical material to Celgene. The Company has determined the option to future products related to the treatment of anemia represents a substantive option. The Company is under no obligation to discover, develop or deliver any new compounds that modulate anemia and Celgene is not contractually obligated to exercise the option. As a result, the Company is at risk as to whether Celgene will exercise the option.
All of these deliverables identified in the arrangement were deemed to have stand-alone value and to meet the criteria to be accounted for as separate units of accounting under ASC 605-25. Factors considered in making this determination included, among other things, the subject of the licenses, the nature of the research and development services, and the capabilities of Celgene.
The total arrangement consideration of $77.7 million under the Luspatercept Agreement and amendedAmended Sotatercept Agreement consisted of (1) the $25.0 million up-frontupfront payment for the license of luspatercept, (2) the remaining deferred revenue from the Original Sotatercept Agreement of $34.7 million, and (3) estimated payments for development activities and manufacturing services of $18.0 million. The Company used its best estimate of selling price (BESP)BESP for each of the undelivered elements as the Company did not have VSOE or (third-party evidence) TPE of selling price for each deliverable. The Company's BESP considered its development plan for the compounds, expected manufacturing services, and reimbursement from Celgene (reimbursement of more than half of development expenses through December 31, 2012 and 100% thereafter). The Company determined its BESP for each of the undelivered elements under the arrangements as of the arrangement execution date as follows:
$18.8 million for research and development services
$2.9 million for the sotatercept joint development committee
$3.7 million for the ACE 536 joint development committee
$2.8 million for the manufacturing services
After determining the BESP of the undelivered elements, the remaining consideration of $49.5 million was recognized upon execution of the arrangements.Luspatercept Agreement and Amended Sotatercept Agreement. The difference between the estimated payments of $18.0 million and the estimated selling prices which totaled $28.2 million, using BESP, for undelivered elements was $10.2 million. This amount was deferred at inception and will be recognized as the undelivered elements are delivered, using the proportional performance method, or ratably in the case of performance on the Joint Development Committee.
As noted above, the total arrangement consideration includes estimated payments for development activities and manufacturing services identified at the outset of the Luspatercept Agreement and amendedAmended Sotatercept Agreement. At the end of each reporting period, the Company reassesses the estimated payments to be received related to these services and the BESP
of the undelivered elements based upon the Company's current estimates. The Company accounts for such changes as a change in accounting estimate and the cumulative impact of any change is reflected in the period of change.
During 2011, the Company achieved a $7.5 million clinical milestone under its Luspatercept Agreement, related to the dosing of the first patient in a multiple-dose clinical trial. The Company evaluated the milestone and determined that it was not substantive, as there was no significant uncertainty to achieving the milestone upon execution of the Luspatercept Agreement. As such, the Company allocated the $7.5 million payment based on the allocation of arrangement consideration determined at the execution date of the Luspatercept Agreement and amendedAmended Sotatercept Agreement. Based on this allocation, the Company recognized $4.8 million of the payment upon achievement, with the remaining $2.7 million recognized as revenue as the undelivered elements are delivered, consistent with the treatment of the up-frontupfront payment. During January 2013,February 2016, the Company achieved a $10.0$15.0 million clinical milestone under its Luspatercept Agreement, related to the dosinginitiation of the first patient for a Phase 23 clinical trial.trial with Luspatercept. The Company evaluated the milestone and deemeddetermined that it to bewas substantive and consistent with the definition of a milestone included in ASU 2010-17 and, accordingly, recognized the $10.0$15.0 million payment in revenue during the year ended December 31, 2013. During December 2013, the Company achieved a $7.0 million clinical milestone under its Sotatercept Agreement, related to Celgene's start of a Phase 2b clinical trial in chronic kidney disease.2016. The Company evaluated the milestone and deemed it to be substantive and consistent with the definition of a milestone included in ASU 2010-17 and, accordingly, recognized the $7.0 million payment in revenue during the year ended December 31, 2013.The remaining development milestones under the Luspatercept and the Amended Sotatercept Agreements were deemed to be substantive and consistent with the definition of a milestone included in ASU 2010-17 and, accordingly, the Company will recognize payments related to the achievement of such milestones, if any, when such milestone is achieved. Factors considered in this determination included scientific and regulatory risks that must be overcome to achieve the milestones, the level of effort and investment required to achieve each milestone, and the monetary value attributed to each milestone. During the years ended December 31, 2015, 20142017, 2016 and 2013,2015, the Company recognized $1.2$0.5 million, $1.7$0.5 million and $2.6$1.2 million, respectively, of the total deferred revenue as license and milestone revenue in the accompanying consolidated statements of operations and comprehensive loss.
F-24
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and development activities through the completion of Phase 1 and initial Phase 2 clinical trials, as well as manufacturing the clinical supplies for these studies. Celgene is responsible for the conduct of subsequent Phase 2 and Phase 3 clinical studies. 2015
In November, 2013, the Company agreed to conduct additional activities for the benefit of the luspatercept program including certain clinical and non-clinical services such as multiple toxicology studies, and associated assay development and sample testing, clinical extension studies, and market development work. These activities will be reimbursed under the same terms and rates of the existing Agreements.agreements. The Company evaluated the additional services to be provided and determined that as the Company is under no obligation to conduct these additional activities, these services do not represent a deliverable under or modification to the Luspatercept Agreement, but rather, represent a separate services arrangement which should be accounted for as the services are delivered.
Other Agreements
Other
In 2004, the Company entered into a license agreement with a non-profit institution for an exclusive, sublicensable, worldwide, royalty-bearing license to certain patents developed by the institution (Primary Licensed Products). In addition, the Company was granted a non-exclusive, non-sub-licensable license for Secondary Licensed Products. As compensation for the licenses, the Company issued 62,500 shares of its common stock to the institution, the fair value of which was $25,000, and was expensed during 2004, to research and development expense. The Company also agreed to pay specified development milestone payments totaling up to $2.0 million for sotatercept and $0.7 million for luspatercept. In addition, the Company is obligated to pay milestone fees based on the Company's research and development progress, and U.S. sublicensing revenue ranging from 10%-25%, as well as a royalty ranging from 1.0%-3.5% of net sales on any products developed under the licenses. During the years ended December 31, 2015, 20142017, 2016 and 2013,2015, the Company paid and expensed milestones and fees defined under the agreement totaling $0.1 million, $0.1 million and $0.5$0.1 million, respectively, which is recorded as research and development expense.
In May 2014, the Company executed a collaboration agreement with a research technology company. The Company paid an upfront and research fee of $0.3 million upon execution of the agreement and the Company is obligated to pay additional research fees of approximately $0.6 million over approximately the next year, depending on the success of the research program. The Company also received an option to obtain a commercial license to the molecules developed during the collaboration. During the yearyears ended December 31, 20152017, 2016, and December 31, 20142015, the Company expensed milestones and fees of $1.4totaling $1.6 million, $1.0 million, and $0.6$1.4 million, which is recorded as research and development expense.
11. Stock-Based Compensation
At December 31, 2015,2017, the Company had two stock-based compensations plans, which are more fully described below.
The Company's 2003 Stock Option and Restricted Stock Plan (the 2003 Plan) providesprovided for the issuance of stock options restricted stock awards, and restricted stock to employees, officers, directors, consultants and key personnel of the Company as determined by the Board. In conjunction with the effectiveness of the 2013 Equity Incentive Plan (the 2013 Plan) described below, the Company determined that no further stock options or other equity-based awards may be granted under the 2003 Plan.
On September 4, 2013, the Board and stockholders approved the adoption of the 2013 Equity Incentive Plan (the 2013 Plan)., which provides for the issuance of stock options, restricted stock units, and other equity-based awards. The Company has reserved for issuance an aggregate of 1,500,000 shares of common stock under the 2013 Plan which is comprised of (i) the remaining 155,884 shares reserved for issuance under the 2003 Plan and (ii) an additional 1,344,116 shares. The 2013 Plan provides that the number of shares reserved and available for issuance under the plan will automatically increase each January 1, beginning in 2014, by the lesser of (i) 3,150,000 shares, or (ii) 4% of the outstanding number of shares of the Company's common stock on the immediately preceding December 31st. This number is subject to adjustment in the event of a stock split, stock dividend or other change in the Company's capitalization. The number of optionsshares underlying equity awards available for future grant was 1,889,7302,762,770 at December 31, 2015.2017.
The Company has not granted unrestricted stock awards under the 2003 Plan or the 2013 Plan since its inception. Stock options carry an exercise price equal to the estimated fair value of the Company's common stock on the date of grant. Options generally expire 10 years following the date of grant. Stock options and restricted stock awards typically vest over 4 years, and restricted stock units typically vest over 3 years, but vesting provisions can vary based on the discretion of the Board.
F-25
Acceleron Pharma Inc.
Notes to Consolidated Financial Statements (Continued)
Years Ended December 31, 2017, 2016 and 2015
Shares of the Company's common stock underlying any awards that are forfeited, canceled, withheld upon exercise of an option, or settlement of an award to cover the exercise price or tax withholding, reacquired by the Company prior to vesting, satisfied without the issuance of shares of the Company's common stock, or otherwise terminated other than by exercise will be added back to the shares of common stock available for issuance under the 2013 Plan. Shares available for issuance under the 2013 Plan may be authorized but unissued shares of the Company's common stock or shares of the Company's common stock that have been reacquired by the Company.
Additionally, on September 4, 2013, the Board and stockholders approved the adoption of the 2013 Employee Stock Purchase Plan (the 2013 ESPP). Under the 2013 ESPP, 275,000 shares of the Company's common stock will be available for issuance to eligible employees. The per-share purchase price at the end of each offering period is equal to 85% of the closing price of one share of the Company’s common stock at the beginning or end of the offering period, whichever is lower, subject to Internal Revenue Service limits. The 2013 ESPP will terminate on September 4, 2023, the tenth anniversary of the initial adoption of the plan. The Board determined the initial offering period commenced on September 16, 2014 and the initial purchase will occuroccurred on the 6 month anniversary.anniversary with subsequent 6 month purchase periods commencing on the day following the purchase from the prior period. The Company recorded $0.3 million, $0.1$0.3 million, and zero$0.3 million of stock-based compensation expense during the years ended December 31, 2017, 2016, and 2015, 2014respectively related to the 2013 ESPP. The number of shares available for future issuance was 186,844 at December 31, 2017.
In December 2016, the Company entered into a consulting agreement with its former Chief Executive Officer. In accordance with the 2003 Plan and 2013 respectively.Plan, any vested shares remain exercisable and any outstanding and unvested options and restricted stock units will continue to vest in accordance with their terms so long as he continues to provides services as a non-employee consultant. During the years ended December 31, 2017, 2016 and 2015, the Company recognized $4.9 million, $0.5 million, and zero, respectively, of stock-based compensation expense related to the agreement.
In April 2017, the Company amended the employment agreement with its former Chief Operating Officer as a result of his diagnosis of amyotrophic lateral sclerosis (ALS). The amended agreement modified the vesting conditions of his stock options and restricted stock units in the event of his termination of employment as a result of death or disability and extended the post termination exercise period of the options. These modifications resulted in $3.6 million of stock-based compensation, recognized within general and administrative expense, during the year ended December 31, 2017.
The Company recognized stock-based compensation expense under the various Plans totaling $12.1 million, $4.8 million and $2.2 million during the years ended December 31, 2015, 2014 and 2013, respectively.
| Year Ended December 31, | Year Ended December 31, | |||||||||||||||||||||
| 2015 | 2014 | 2013 | 2017 | 2016 | 2015 | |||||||||||||||||
| Research and development | $ | 4,852 | $ | 2,065 | $ | 659 | $ | 14,227 | $ | 8,171 | $ | 4,852 | ||||||||||
| General and administrative | 7,223 | 2,713 | 1,537 | 14,021 | 10,386 | 7,223 | ||||||||||||||||
| $ | 12,075 | $ | 4,778 | $ | 2,196 | $ | 28,248 | $ | 18,557 | $ | 12,075 | |||||||||||