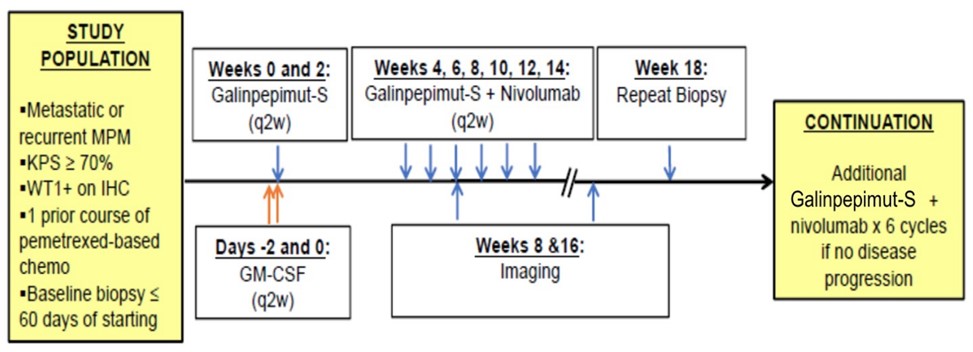
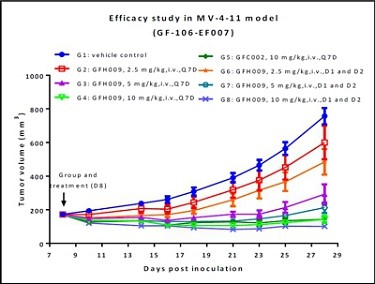
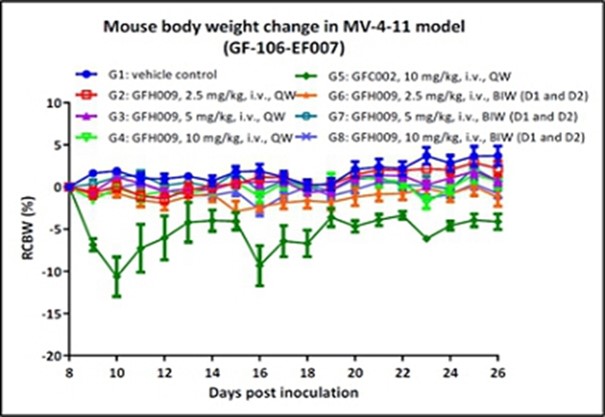
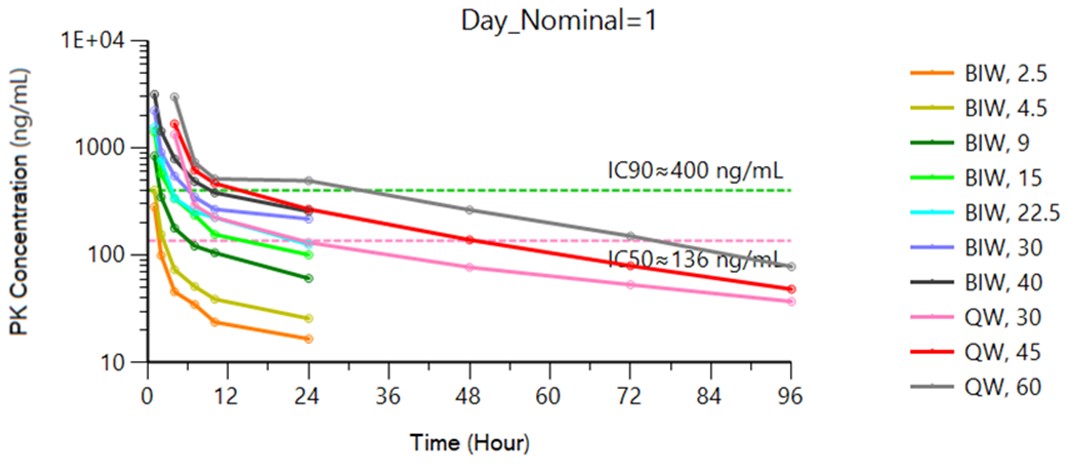
The FDA and other regulatory authorities at federal, state, and local levels, as well as in foreign countries, extensively regulate, among other things, the research, development, testing, manufacture, quality control, import, export, safety, effectiveness, labeling, packaging, storage, distribution, record keeping, approval, advertising, promotion, marketing, post-approval monitoring, and post-approval reporting of drugs and biologics such as those we are developing. Along with our third-party contractors, we will be required to navigate the various preclinical, clinical and commercial approval requirements of the governing regulatory agencies of the countries in which we wish to conduct studies or seek approval or licensure of its current or future product candidates. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources. A company can make
only those claims relating to safety and efficacy, purity and potency that are approved by the FDA and in accordance with the provisions of the approved label.
The testing and approval process requires substantial time, effort and financial resources, and we cannot be certain that any approvals for our current or future product candidates will be granted on a timely basis, if at all.
Before testing any drug or biological product candidate, including our product candidates, in humans, the product candidate must undergo rigorous preclinical testing. TheNonclinical studies during the preclinical developmentaldevelopment stage generally involves laboratory evaluations of drug chemistry, formulation and stability, as well as studies to evaluate toxicity in animals, which support subsequent clinical testing. Preclinical studies include laboratory evaluation of product chemistry and formulation as well asand typically include in vitro and animal studies to assess the potential for adverse events and in some cases to establish a rationale for therapeutic use. The
Prior to beginning the first clinical trial with a product candidate, we must submit an IND to the FDA. An IND is a request for authorization from the FDA to administer an investigational new drug product to humans. The central focus of an IND submission is on the general investigational plan and the protocol(s) for clinical studies. The IND also includes the results of the preclinicalnonclinical studies of the product candidate, together with manufacturing information, analytical data, any available clinical data or literature. An IND must become effective before human clinical trials may begin. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, issues a notice expressly authorizing the proposed trial to proceed or raises safety concerns or questions about the proposed clinical trial. In such a case,If the FDA raises concerns or places the trial on clinical hold, the IND may be placed on clinical hold and the IND sponsor and the FDAagency must resolve any outstanding concerns or questions before the clinicalproposed trial can begin. Submission of an IND therefore may or may not result in FDA authorization to begin a clinical trial.
Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified investigators in accordance with GCP, which include the requirement that all research subjects provide their informed consent for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. A separate submission to the existing IND must be made for each successive clinical trial conducted during product development and for any subsequent protocol amendments. Furthermore, an IRB for each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial and its informed consent form before the clinical trial begins at that site and must monitor the clinical trial until completed. Regulatory authorities, the IRB or the sponsor may suspend a clinical trial at any time on various grounds, including a finding that the subjects are being exposed to an unacceptable health risk or that the trial is unlikely to meet its stated objectives. Some studies also include oversight by an independent data safety monitoring board, or DSMB, organized by the clinical trial sponsor, which provides authorization for whether or not a clinical trial may move forward at designated check points based on access to certain data from the clinical trial and may recommend the sponsor to halt the clinical trial if itthe DSMB determines that there is an unacceptable safety risk for subjects or other grounds, such as no demonstration of efficacy.
Information about certain clinical trials, including details of the protocol and eventually study results, also must be submitted within specific timeframes to the National Institutes of HealthNIH for public dissemination on the ClinicalTrials.gov data registry. Information related to the product, patient population, phase of investigation, study sites and investigators and other aspects of the clinical trial is made public as part of the registration of the clinical trial. Sponsors are also obligated to disclose the results of their clinical trials after completion. Disclosure of the results of these trials can be delayed in some cases for up to two years after the date of completion of the trial. Competitors may use this publicly available information to gain knowledge regarding the progress of development programs. Failure to timely register a covered clinical trial or to submit study results as provided for in the law can give rise to civil monetary penalties and also prevent the non-compliant party from receiving future grant funds from the federal government. The NIH Final Rule on ClinicalTrials.gov registration and reporting requirements became effective in 2017, and both NIH and FDA have brought enforcement actions against non-compliant clinical trial sponsors.
Phase 1, Phase 2 and Phase 3 testing may not be completed successfully within a specified period, if at all, and there can be no assurance that the data collected will support FDA approval or licensure of the product.
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if unexpected serious adverse events, or SAEs, occur. The FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects or patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the clinical protocol, cGCP,GCP, or other IRB requirements or if the drug has been associated with unexpected serious harm to patients.
BLA Submission and Review by the FDA
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, the results of product development, preclinical studies and clinical trials are submitted to the FDA as part of aan NDA or BLA requesting approval to market the product for one or more indications. The NDA or BLA must contain proof of the biological product candidate’s safety purity, potency and efficacysubstantial evidence of effectiveness for its proposed indication or indications in the form of relevant data available from pertinent preclinical and clinical studies, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s chemistry, manufacturing, controls, and proposed labeling, among other things. In particular, a marketing application must demonstrate that the manufacturing methods and quality controls used to produce the drug or biological product are adequate to preserve the drug’s identity, strength, quality, and purity for an NDA or a biologic’s safety, purity, and potency for a BLA. Data can come from company-sponsored clinical studies intended to test the safety and effectiveness of a use of the product, or from a number of alternative sources, including studies initiated by investigators. FDA approval of aan NDA or BLA must be obtained before the corresponding drug or biologic may be marketed in the United States.
Under federal law, the fee for the submission of aan NDA or BLA for which clinical data is submitted and analyzed is substantial, (for example, for FY2021 this application fee exceeds $2.8 million), and the sponsor of an approved NDA or BLA is also subject to an annual program fee, currently more than $330,000 per program.fee. These fees are typically increased annually, but exemptions and waivers may be available under certain circumstances (such as a waiver for the first human drug application submitted by a qualifying small business and exemptions for orphan products).
Once a BLA has beenThe FDA reviews all submitted the FDA’s goal isNDAs and BLAs to review the application within ten months afterdetermine if they are substantially complete before it accepts the applicationthem for filing or, if the application relates to an unmet medical need in a serious or life-threatening indication, six months after the FDA accepts the application for filing. This FDA review typically takes twelve months from the date the BLA is submitted to the FDA (for a standard review) and eight months from the date the BLA is submitted (for a “priority review”) because the FDA has approximately two months, or 60 days, after BLA submission to make a “filing” decision. Furthermore, the review process is often significantly extended by FDA requests for additional information or clarification after the BLA has been accepted for filing. The FDA may also request additional information rather than acceptaccepting a submission for filing. The FDA must make a decision on accepting an NDA or BLA for filing.filing within 60 days of receipt and must inform the sponsor by the 74th day after the FDA’s receipt of the submission whether the application is sufficiently complete to permit substantive review. The FDA may refuse to file any submission that it deems incomplete or not properly reviewable at the time of submission and may request additional information. In this event, the marketing application must be resubmitted with the additional information.information requested by the agency. The resubmitted application is also subject to review before the FDA accepts it for filing.
After aOnce an NDA or BLA is accepted for filing, the FDA’s goal is to review the application within 10 months after it accepts the application for filing, or, if the application meets the criteria for “priority review”, six months after the FDA accepts the application for filing. The review process is often significantly extended by FDA requests for additional information or clarification after the NDA or BLA has been accepted for filing. The review process may be extended by the FDA for three additional months to consider new information or in the case of a clarification provided by the applicant to address an outstanding deficiency identified by the FDA following the original submission.
During the review process, the FDA reviews itthe NDA or BLA to determine, among other things, whether the product is safe, effective, pure and potent and the facility in which it is manufactured, processed, packed, or held meets standards designed to assure the product’s continued safety, purity and potency. The FDA may refer any NDA or BLA, including applications for novel drug or biologic candidates which present difficult questions of safety or efficacy, to an advisory committee to provide clinical insight on application review questions. Typically, an advisory committee is a panel of independent experts, including clinicians and other scientific experts that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it considers such recommendations carefully when making final decisions on approval.
Before approving aan NDA or BLA, the FDA will typically inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving aan NDA or BLA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP.GCP and integrity of the clinical data. If the FDA determines that the application, manufacturing process or manufacturing facilities are not acceptable, it will outline the deficiencies inas part of the submissionreview process and often will request additional testing or information. Notwithstanding the submission of
any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
Under the Pediatric Research Equity Act, or PREA, amendments to the FDCA, aan NDA or BLA or supplement to a BLAsuch applications must contain data that are adequate to assess the safety and efficacy of the product candidate for the claimed indications in all relevant pediatric populations and to support dosing and administration for each pediatric population for which the product is safe and effective. The FDA may grant deferrals for submission of pediatric data or full or partial waivers. The Food and Drug Administration Safety and Innovation Act, or the FDASIA, enacted in 2012, made permanent PREA to requirerequires a sponsor whothat is planning to submit a marketing application for a product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration to submit an initial Pediatric Study Plan, or PSP, within sixty days of an end-of-Phaseend of-Phase 2 meeting or, if there is no such meeting, as early as practicable before the initiation of the Phase 3 or Phase 2/3 clinical trial. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including trial objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. The FDA and the sponsor must reach an agreement on the PSP. A sponsor can submit amendments to an agreed upon initial PSP at any time if changes to the pediatric plan need to be considered based on data collected from pre-clinicalpreclinical studies, early phaseearly-phase clinical trials or other clinical development programs.
The testing and approval process requires substantial time, effort and financial resources, and each may take several years to complete. The FDA may not grant approval on a timely basis, or at all, and we may encounter difficulties or unanticipated costs in our efforts to secure necessary governmental approvals, which could delay or preclude us from marketing itsour products. After the FDA evaluates aan NDA or BLA and conducts inspections of the manufacturing facilities where the investigational product and/or its drug substance will be produced, the FDA may issue an approval letter or a Complete Response Letter.Letter, or CRL. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. A Complete Response LetterCRL indicates that the review cycle of the application is complete and the application iswill not ready for approval.be approved in its present form. A Complete Response LetterCRL generally outlines the deficiencies in the submission and may require substantial additional testing, information or clarification for FDA to reconsider the application. The FDA may delay or refuse approval of aan NDA or BLA if applicable regulatory criteria are not satisfied, require additional testing or information and/or require post-marketing testing and surveillance to monitor safety or efficacy of a product. If a CRL is issued, the applicant may either resubmit the NDA or BLA, addressing all of the deficiencies identified in the letter, or withdraw the application. If and when the deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the marketing application, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in response to an issued CRL in either two or six months depending on the type of information included. Even if such data and information are submitted, the FDA may ultimately decide that the NDA or BLA does not satisfy the criteria for approval.
If regulatory approval of a product is granted, such approval is limited to the conditions of use (e.g., patient population, indication) described in the application and may entail further limitations on the indicated uses for which such product may be marketed. For example, the FDA may approve the NDA or BLA with a Risk Evaluation and Mitigation Strategy, or REMS, plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA determines the requirement for a REMS, as well as the specific REMS provisions, on a case-by-case basis. If the FDA concludes a REMS plan is needed, the sponsor of the NDA or BLA must submit a proposed REMS. The FDA will not approve aan NDA or BLA without a REMS, if one is required. The FDA also may condition approval on, among other things, changes to proposed labeling (e.g., adding contraindications, warnings or precautions) or the development of adequate controls and specifications. Once approved, the FDA may withdraw the product approval if compliance with pre- and post-marketing regulatory standards is not maintained or if problems occur after the product reaches the marketplace. The FDA may require one or more Phase 4 post-market studies and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization and may limit further marketing of the product based on the results of these post-marketing studies. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and FDA review and approval. In addition, new government requirements, including those resulting from new legislation,
may be established, or the FDA’s policies may change, which could delay or prevent regulatory approval of our products under development.
Fast Track, Priority Review, Accelerated Approval, and Breakthrough Therapy Designations
A sponsor may seek approval of its product candidate under programs designed to accelerate FDA’s review and approval of new drugs and biological products that meet certain criteria. Specifically, new drugs and biological products are eligible for Fast Track designation if they are intended to treat a serious or life-threatening condition and demonstrate the potential to address unmet medical needs for the condition. For a product candidate with Fast Track designation provides increased opportunities for sponsor interactions with the FDA during preclinical and clinical development, in addition to the potential for rolling review once a marketing application is filed, meaning that the FDA may consider for review sections of the NDA or BLA for review on a rolling basis before the complete application is submitted, if relevant criteria are met.the sponsor provides a schedule for the submission of the sections of the NDA or BLA, the FDA agrees to accept sections of the marketing application and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application. A Fast Track designated product candidate may also qualify for accelerated approval (described below) or priority review, under which the FDA sets the target date for FDA action on the NDA or BLA at six months after the FDA accepts the application for filing. We have obtained Fast Track designation for GPS in AML, MPM and MM, and for NPSSLS009 in TNBC. r/r AML and r/r PTCL.
Priority review is granted when there is evidence that the proposed product would be a significant improvement in the safety or effectiveness of the treatment, diagnosis, or prevention of a serious condition. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting drug reaction, documented enhancement of patient compliance that may lead to improvement in serious outcomes, or evidence of safety and effectiveness in a new subpopulation. If criteria are not met for priority review, the application is subject to the standard FDA review period of ten months after FDA accepts the application for filing. Priority review designation does not change the scientific/medical standard for approval or the quality of evidence necessary to support approval.
Under the Accelerated Approval program, the FDA may approve a BLA on the basis of either a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. Post-marketing studies or completion of ongoing studies after marketing approval are generally required to verify the biologic’s clinical benefit in relationship to the surrogate endpoint or ultimate outcome in relationship to the clinical benefit. Failure to conduct required post-approval studies, or to confirm the predicted clinical benefit of the product during post-marketing studies, would allow the FDA to withdraw approval of the drug.
In addition, a sponsor may seek FDA designation of its product candidate as a Breakthrough Therapy, if the product candidate is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the therapy may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. IfBreakthrough Therapy designation provides all the features of Fast Track designation in addition to intensive guidance on an efficient development program beginning as early as Phase 1, and FDA designatesorganizational commitment to expedited development, including involvement of senior managers and experienced review and regulatory staff in a breakthrough therapy, it may take actions appropriate to expedite the development andproactive, collaborative, cross-disciplinary review, of the application. Breakthrough designation also allows the sponsor to file sections of the BLA for review on a rolling basis. Drugs or biologicswhere appropriate. A drug designated as breakthrough therapies areBreakthrough Therapy is also eligible for accelerated approval of their respective marketing applications.if the relevant criteria are met.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened. Fast Track, Priority Reviewpriority review and Breakthrough Therapy designations do not change the scientific or medical standards for approval or the quality of evidence necessary to support approval but may expedite the development or approval process.
Accelerated Approval
In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval from the FDA and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The FDA may also grant accelerated approval for such a drug or biologic when it has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality, or IMM, and that is reasonably likely to predict an effect on IMM or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require that a sponsor of a drug or biologic receiving accelerated approval perform post-marketing clinical trials to verify and
describe the predicted effect on IMM or other clinical endpoint, and the product may be subject to expedited withdrawal procedures. Drugs and biologics granted accelerated approval must meet the same statutory standards for safety and effectiveness as those granted traditional approval.
For the purposes of accelerated approval, a surrogate endpoint is a marker, such as a laboratory measurement, radiographic image, physical sign, or other measure that is thought to predict clinical benefit but is not itself a measure of clinical benefit. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. An intermediate clinical endpoint is a measurement of a therapeutic effect that is considered reasonably likely to predict the clinical benefit of a drug or biologic, such as an effect on IMM. The FDA has limited experience with accelerated approvals based on intermediate clinical endpoints but has indicated that such endpoints generally may support accelerated approval when the therapeutic effect measured by the endpoint is not itself a clinical benefit and basis for traditional approval, if there is a basis for concluding that the therapeutic effect is reasonably likely to predict the ultimate long-term clinical benefit of a drug or biologic.
The accelerated approval pathway is most often used in settings in which the course of a disease is long and an extended period of time is required to measure the intended clinical benefit of a drug, even if the effect on the surrogate or intermediate clinical endpoint occurs rapidly. For example, accelerated approval has been used extensively in the development and approval of drugs and biologics for treatment of a variety of cancers in which the goal of therapy is generally to improve survival or decrease morbidity and the duration of the typical disease course requires lengthy and sometimes large clinical trials to demonstrate a clinical or survival benefit.
The accelerated approval pathway is usually contingent on a sponsor’s agreement to conduct, in a diligent manner, additional post-approval confirmatory studies to verify and describe the product candidate’s clinical benefit. As a result, a product candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or to confirm the predicted clinical benefit of the product during post-marketing studies, would allow the FDA to withdraw approval of the product. As part of the Consolidated
Appropriations Act for 2023, Congress provided FDA additional statutory authority to mitigate potential risks to
patients from continued marketing of ineffective drugs or biologics previously granted accelerated approval. Under
the act’s amendments to the FDCA, FDA may require the sponsor of a product granted accelerated approval to have a confirmatory trial underway prior to approval. The sponsor must also submit progress reports on a confirmatory trial every six months until the trial is complete, and such reports are published on FDA’s website. The amendments also give FDA the option of using expedited procedures to withdraw product approval if the sponsor’s confirmatory trial fails to verify the claimed clinical benefits of the product.
All promotional materials for product candidates being considered and approved under the accelerated approval program are subject to prior review by the FDA.
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant Orphan Drug Product Designation to a drug or biologic intended to treat a rare disease or condition, defined as a disease or condition with a patient population of fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 individuals in the United States and when there is no reasonable expectation that the cost of developing and making available the drug or biologic in the United States will be recovered from sales in the United States for that drug or biologic. Orphan Drug Product Designation must be requested before submitting aan NDA or BLA. After the FDA grants Orphan Drug Product Designation, the generic identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA.
If a drug or biologic product that has Orphan Drug Product Designation subsequently receives the first FDA approval for a particular active ingredient for the disease for which it has such designation, the product is entitled to orphan product exclusivity, which means that the FDA may not approve any other applications, including a full NDA or BLA, to market the same drug or biologic for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan product exclusivity or if FDA finds that the holder of the orphan product exclusivity has not shown that it can assure the availability of sufficient
quantities of the orphan product to meet the needs of patients with the disease or condition for which the drug or biologic was designated. Orphan product exclusivity does not prevent the FDA from approving a different drug or biologic for the same disease or condition, or the same drug or biologic for a different disease or condition. Among the other benefits of Orphan Drug Product Designation are tax credits for certain research and a waiver of the BLA application user fee.
A drug or biologic with Orphan Drug Product Designation may not receive orphan product exclusivity if it is approved for a use that is broader than the indication for which it received Orphan Drug Product Designation. In addition, orphan product exclusive marketing rights in the United States may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition.
Recent court cases have challenged FDA’s approach to determining the scope of orphan drug exclusivity; however, at this time the agency continues to apply its long-standing interpretation of the governing regulations and has stated that it does not plan to change any orphan drug implementing regulations.
We have obtained Orphan Drug Product Designation in the United States for GPS in AML, MPM and MM.MM and for SLS009 for AML and PTCL.
Pediatric exclusivity
Pediatric exclusivity is a type of non-patent marketing exclusivity available in the United States and, if granted, it provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity or listed patents. This six-month exclusivity may be granted if aan NDA or BLA sponsor submits pediatric data that fairly respond to a written request from the FDA for such data. The data do not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the FDA’s request, the additional protection is granted. If reports of requested pediatric studies are submitted to and accepted by the FDA within the statutory time limits, whatever statutory or regulatory periods of exclusivity or patent protection cover the product are extended by six months. This is not a patent term extension, but it effectively extends the regulatory period during which the FDA cannot approve another application. The issuance of a written request does not require the sponsor to undertake the described studies.
Patent term restoration
Depending upon the timing, duration and specifics of FDA approval of the use of our product candidates, some of our United States patents may be eligible for limited patent term extension under the Hatch-Waxman Act. The Hatch-Waxman Act permits a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product candidate’s approval date. The patent term restoration period is generally one half of the time between the effective date of an IND and the submission date of an NDA or BLA, plus the time between the submission date of the NDA or BLA and the approval of that application, except that the review period is reduced by any time during which the applicant failed to exercise due diligence. Only one patent applicable to an approved product candidate is eligible for the extension and the application for extension must be made prior to expiration of the patent. The USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we intend to apply for restorations of patent term for some of our currently owned or licensed patents to add patent life beyond their current expiration date, depending on the expected length of clinical trials and other factors involved in the submission of the relevant NDA or BLA.
Abbreviated new drug applications for generic drugs
In 1984, with passage of the Drug Price Competition and Patent Term Restoration Act, or Hatch-Waxman Act, which established an abbreviated regulatory scheme authorizing the FDA to approve generic drugs based on an innovator or “reference” product, Congress also enacted Section 505(b)(2) of the FDCA, which provides a hybrid pathway combining features of a traditional NDA and a generic drug application. To obtain approval of a generic
drug, an applicant must submit an abbreviated new drug application, or ANDA, to the agency. ANDAs are “abbreviated” because they do not include preclinical and clinical data to demonstrate safety and effectiveness.
Instead, in support of such applications, a generic manufacturer may rely on the preclinical and clinical testing previously conducted for a drug product previously approved under an NDA, known as the reference-listed drug, or RLD.
Specifically, in order for an ANDA to be approved, the FDA must find that the generic version is identical to the RLD with respect to the active ingredients, the route of administration, the dosage form, and the strength of the drug. At the same time, the FDA must also determine that the generic drug is “bioequivalent” to the innovator drug. Under the statute, a generic drug is bioequivalent to an RLD if “the rate and extent of absorption of the drug do not show a significant difference from the rate and extent of absorption of the listed drug.”
Upon approval of an ANDA, the FDA indicates whether the generic product is “therapeutically equivalent” to the RLD in its publication Approved Drug Products with Therapeutic Equivalence Evaluations, also referred to as the Orange Book. Clinicians and pharmacists consider a therapeutic equivalent generic drug to be fully substitutable for the RLD. In addition, by operation of certain state laws and numerous health insurance programs, the FDA’s designation of therapeutic equivalence often results in substitution of the generic drug without the knowledge or consent of either the prescribing clinicians or patient.
In contrast, Section 505(b)(2) permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference. A Section 505(b)(2) applicant may eliminate the need to conduct certain preclinical or clinical studies, if it can establish that reliance on studies conducted for a previously approved product is scientifically appropriate. Unlike the ANDA pathway used by developers of bioequivalent versions of innovator drugs, the 505(b)(2) regulatory pathway does not preclude the possibility that a follow-on applicant would need to conduct additional clinical trials or nonclinical studies; for example, they may be seeking approval to market a previously approved drug for new indications or for a new patient population that would require new clinical data to demonstrate safety or effectiveness.
In addition, under the Hatch-Waxman Act, the FDA might not approve an ANDA or 505(b)(2) NDA until any applicable period of non-patent exclusivity for the RLD has expired. These market exclusivity provisions under the FDCA also can delay the submission or the approval of certain applications. The FDCA provides a period of five years of non-patent data exclusivity for a new drug containing a new chemical entity. For the purposes of this provision, a new chemical entity, or NCE, is a drug that contains no active moiety that has previously been approved by the FDA in any other NDA. An active moiety is the molecule or ion responsible for the physiological or pharmacological action of the drug substance. In cases where such NCE exclusivity has been granted, an ANDA or 505(b)(2) NDA may not be filed with the FDA until the expiration of five years unless the submission is accompanied by a Paragraph IV certification (described below), in which case the applicant may submit its application four years following the original product approval.
The FDCA also provides for a period of three years of exclusivity for an NDA, 505(b)(2) NDA or supplement thereto if one or more new clinical investigations, other than bioavailability or bioequivalence studies, that were conducted by or for the applicant are deemed by the FDA to be essential to the approval of the application. This three-year exclusivity period often protects changes to a previously approved drug product, such as a new dosage form, route of administration, combination or indication. The three-year exclusivity covers only the conditions of use associated with the new clinical investigations and does not prohibit the FDA from approving follow-on applications for drugs containing the original active agent. Five-year and three-year exclusivity also will not delay the submission or approval of a traditional NDA filed under Section 505(b)(1) of the FDCA. However, an applicant submitting a traditional NDA would be required to either conduct or obtain a right of reference to all of the preclinical studies and
adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
Hatch-Waxman patent certification and the 30-month stay
Upon approval of an NDA or a supplement thereto, NDA sponsors are required to list with the FDA each patent with claims that cover the applicant’s product or an approved method of using the product. Each of the patents listed by
the NDA sponsor is published in the Orange Book. When an ANDA applicant files its application with the FDA, the applicant is required to certify to the FDA concerning any patents listed for the reference product in the Orange Book, except for patents covering methods of use for which the ANDA applicant is not seeking approval. To the extent that the Section 505(b)(2) NDA applicant is relying on studies conducted for an already approved product, the applicant is required to certify to the FDA concerning any patents listed for the approved product in the Orange Book to the same extent that an ANDA applicant would.
Specifically, the applicant must certify with respect to each patent that:
I.the required patent information has not been filed by the original applicant;
II.the listed patent has expired;
III.the listed patent has not expired, but will expire on a particular date and approval is sought after patent expiration; or
IV.the listed patent is invalid, unenforceable or will not be infringed by the manufacture, use or sale of the new product.
If a Paragraph I or II certification is filed, the FDA may make approval of the application effective immediately upon completion of its review. If a Paragraph III certification is filed, the approval may be made effective on the patent expiration date specified in the application, although a tentative approval may be issued before that time. If an application contains a Paragraph IV certification, a series of events will be triggered, the outcome of which will determine the effective date of approval of the ANDA or 505(b)(2) application.
If the follow-on applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the follow-on application in question has been accepted for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days after the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA or 505(b)(2) NDA until the earlier of 30 months after the receipt of the Paragraph IV notice, expiration of the patent, or a decision in the infringement case that is favorable to the ANDA or 505(b)(2) applicant. Alternatively, if the listed patent holder does not file a patent infringement lawsuit within the required 45-day period, the follow-on applicant’s ANDA or 505(b)(2) NDA will not be subject to the 30-month stay.
Reference a product exclusivity for biological products
In March 2010, the Patient Protection and Affordable Care Act, or ACA, was enacted in the United States and included theThe Biologics Price Competition and Innovation Act of 2009, or the BPCIA. The BPCIA, amended the PHSA to createauthorize the FDA to approve similar versions of innovative biologics such as ours, which are also known as “reference biological products.” The new pathway authorized under the BPCIA allows FDA to approve, under an abbreviated approval pathway forapplication, a biological productsproduct that are biosimilardemonstrated to be “biosimilar” or interchangeable“interchangeable” with an FDA-licensed reference biological product. A federal district court ruling in Texas struck down the ACA in its entirety based on constitutionality in December 2018 after the individual mandate was repealed by Congress as part of the Tax Cuts and Jobs Act, effective January 1, 2019, and in December 2019 the Fifth Circuit Court of Appeals upheld the lower court’s finding that the individual mandate in the law is unconstitutional. However, the Fifth Circuit also reversed and remanded the caseBiosimilarity to the district court to determine if other reforms enacted as part of the ACA, but not specifically related to the individual mandate or health insurance, including the BPCIA, could be severed from the rest of the ACA so as not to be declared invalid. On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case and allocated one hour for oral arguments, which occurred on November 10, 2020. A decision from the Supreme Court is expected to be issued in spring 2021. It is unclear how this litigation and other efforts to repeal and replace the ACA will affect the implementation of that law and our business. To date, the FDA hasan approved a number of biosimilars, and numerous biosimilars have been approved in Europe. The FDA has also issued several guidance documents outlining its approach to reviewing and approving biosimilars and interchangeable biosimilars.
A biosimilar product is defined as one that is highly similar to a reference product notwithstanding minorrequires that there be no differences in clinically inactive componentsmechanism of action for the conditions of use, route of administration, dosage form, and for which there arestrength, and no clinically meaningful differences between the follow-on biological product and the reference product in terms of the safety, purity, and potencypotency. Biosimilarity is demonstrated in steps beginning with rigorous analytical studies or “fingerprinting,” in vitro studies, in vivo animal studies, and generally at least one clinical study, absent a waiver from the FDA. The biosimilarity exercise tests the hypothesis that the investigational product and the reference product are the same. If at any point in the stepwise biosimilarity process a significant difference is observed, then the products are not biosimilar, and the development of a standalone BLA for the product. An interchangeablefollow-on biological product is necessary. In order to meet the higher hurdle of interchangeability, a sponsor must demonstrate that the biosimilar product that can be expected to produce the same clinical resultsresult as the reference product, in any given patient and for productsa product that is administered multiple times to an individual,more than once, that the product andrisk of switching between the reference product may be alternated or switched after one has been previously administered without increasing safety risks or risksand biosimilar product is not greater than the risk of diminished efficacy relative to exclusive use ofmaintaining the patient on the reference product. Complexities associated with the larger, and often more complex, structures of biological product withoutproducts, as well as the process by which such alternation or switch. Upon licensureproducts are manufactured, pose significant hurdles to implementation that are still being evaluated by the FDA, an interchangeable biosimilar may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product, although to date no such products have been approved for marketing in the United States.FDA.
The biosimilar applicant must demonstrate that its product is biosimilar based on data from (1) analytical studies showing thatUnder the biosimilar product is highly similar to the reference product; (2) animal studies (including toxicity); and (3) one or more clinical studies to demonstrate safety, purity and potency in one or more appropriate conditions of use for which the reference product is approved. In addition, the applicant must show that the biosimilar and reference products have the same mechanism of action for the conditions of use on the label, route of administration, dosage and strength, and the production facility must meet standards designed to assure product safety, purity and potency.
ABPCIA, a reference biological product is granted twelve12 years of data exclusivity from the timedate of first licensure of the product, andwhich means that the FDA is barred from approving biosimilar applications for 12 years after the reference biological product receives initial marketing approval. The first approved interchangeable biologic
biological product will be granted an exclusivity period of up to one year after it is first commercially marketed. If pediatric studies are performedmarketed, and accepted byas part of the FDA as responsiveConsolidated Appropriations Act for 2023, Congress amended the PHSA in order to a Written Request,permit multiple interchangeable products approved on the twelve-yearsame day to receive and benefit from this one-year exclusivity period will be extended for an additional six months.period. In addition, the FDA will not accept an application for a biosimilar or interchangeable product based on the reference biological product until four years after the date of first licensure of the reference product. “First licensure” typically means the initial date the particular product at issue was licensed in the United States. Date of first licensure does not include the date of licensure of (and a new period of exclusivity is not available for) a supplement for the reference product for a subsequent application filed by the same sponsor or manufacturer of the reference product (or licensor, predecessor in interest or other related entity) for a change (not including a modification to the structure of the biological product) that results in a new indication, route of administration, dosing schedule, dosage form, delivery system, delivery device or strength or for a modification to the structure of the biological product that does not result in a change in safety, purity or potency. Therefore, one must determine whether a new product includes a modification to the structure of a previously licensed product that results in a change in safety, purity or potency to assess whether the licensure of the new product is a first licensure that triggers its own period of exclusivity. Whether a subsequent application, if approved, warrants exclusivity as the “first licensure” of a biological product is determined on a case-by-case basis with data submitted by the sponsor.
The BPCIA is complex and only beginningcontinues to be interpreted and implemented by the FDA. In addition, recent government proposals have sought to reduce the twelve-year12-year reference product exclusivity period. Other aspects of the BPCIA, some of which may impact the BPCIA exclusivity provisions, have also been the subject of recent litigation. As a result, the ultimate impact, implementation and meaning of the BPCIA continues to be subject to significant uncertainty.
Post-approval requirements
Following approval of a new product, the manufacturer and the approved product are subject to pervasive and continuing regulation by the FDA, including, among other things, monitoring and recordkeeping activities, reporting of adverse experiences with the product, product sampling and distribution restrictions, complying with promotion and advertising requirements, which include restrictions on promoting drugs for unapproved uses or patient populations (i.e., “off-label use”) and limitations on industry-sponsored scientific and educational activities. The manufacturer and its productproducts are also subject to similar post-approval requirements by regulatory authorities comparable to FDA in jurisdictions outside of the United States where the product isproducts are approved. Although physicians may prescribe legally available products for off-label uses, manufacturers may not market or promote such uses. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. If there are any modifications to the product, including changes in indications, labeling or manufacturing processes or facilities, the applicant may be required to submit and obtain FDA approval of a new NDA or BLA or a BLA supplement thereto, which may require the applicant to develop additional data or conduct additional pre-clinicalpreclinical studies and clinical trials. The FDA may also place other conditions on approvals including the requirement for a REMS to assure the safe use of the product. A REMS could include medication guides, physician communication plans or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of products. Product approvals may be withdrawn for non-compliance with regulatory standards or if problems occur following initial marketing.
FDA regulations require that products be manufactured in specific approved facilities and in accordance with cGMPs.cGMP. The cGMP regulations include requirements relating to organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports and returned or salvaged products. The manufacturing facilities for our product candidates must meet applicable cGMP requirements to the FDA'sFDA’s or comparable foreign regulatory authorities'authorities’ satisfaction before any product is approved and our commercial products can be manufactured. We rely, and expect to continue to rely, on third parties for the production of clinical and commercial quantities of our products in accordance with cGMP regulations. These manufacturers must comply with cGMP regulations that require, among other things, quality control and quality assurance, the maintenance of records and documentation and the obligation to investigate and correct any deviations from cGMP. Manufacturers and other entities involved in the manufacture and distribution of approved
drugs or biologics are required to register their establishments with the FDA and certain state agencies and are subject to periodic prescheduled or unannounced inspections by the FDA and certain state agencies for compliance with cGMP and other laws. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain cGMP compliance. Future inspections by the FDA and other regulatory agencies may identify compliance issues at the facilities of our CMOs that may disrupt production or distribution or require substantial resources to correct. In addition, the discovery of conditions that violate these rules, including failure to conform to cGMPs,cGMP, could result in enforcement actions, and the discovery of problems with a product after approval may result in restrictions on a product, manufacturer or holder of an approved BLA, including voluntary recall and regulatory sanctions as described below.
Once an approval or clearance of a drug or biologic is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
•restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
•fines, warning letters or other enforcement-related letters or clinical holds on post-approval clinical trials;
•refusal of the FDA to approve pending NDAs or BLAs or supplements to approved BLAs,marketing authorizations, or suspension or revocation of product approvals;
•product seizure or detention, or refusal to permit the import or export of products;
•injunctions or the imposition of civil or criminal penalties; and
•consent decrees, corporate integrity agreements, debarment, or exclusion from federal health care programs; or mandated modification of promotional materials and labeling and the issuance of corrective information.
In addition, the distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, or PDMA, which regulates the distribution of drugs and drug samples at the federal level and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution. Most recently,Furthermore, the Drug Supply Chain Security Act, or DSCSA, was enacted with the aim of building an electronic system to identify and trace certain prescription drugs distributed in the United States, including most biological products. The DSCSA mandates phased-in and resource-intensive obligations for pharmaceutical manufacturers, wholesale distributors, and dispensers over a ten‑yearten-year period, that is expected to culminatewhich culminated in November 2023. Most recently, the FDA announced a one-year stabilization period to November 2024, giving entities subject to the DSCSA additional time to finalize interoperable tracking systems and to ensure supply chain continuity. From time to time, new legislation and regulations may be implemented that could significantly change the statutory provisions governing the approval, manufacturing and marketing of products regulated by the FDA. It is impossible to predict whether further legislative or regulatory changes will be enacted, whether FDA regulations, guidance or interpretations will be changed or what the impact of such changes, if any, may be.
Other HealthcareHealth Care Laws and Compliance Requirements
Our sales, promotion, medical education and other activities following product approval will be subject to regulation by numerous regulatory and law enforcement authorities in the United States in addition to FDA, including potentially the Federal Trade Commission, or FTC, the Department of Justice, or DOJ, the Centers for Medicare and Medicaid Services, or CMS, other divisions of the Department of Health and Human Services, or DHHS, and state and local governments. Our promotional and scientific/educational programs must comply with the federal
Anti-Kickback Statute, the Foreign Corrupt Practices Act, the False Claims Act, or FCA, the Veterans Health Care Act, physician payment transparency laws, privacy laws, security laws, and additional state laws similar to the foregoing.
The federal Anti-Kickback Statute prohibits, among other things, the offer, receipt,persons from knowingly and willfully soliciting, offering, receiving, or payment ofpaying remuneration, directly or indirectly, in cash or in kind, in exchange for or to induce or reward either the referral of patients for, or the usepurchase, order or recommendation of, productsany good or servicesservice that wouldmay be paid for in whole or part by Medicare, Medicaid or other federal health care programs. Remuneration has been broadly defined to include anything of value, including cash, improper discounts, and free or reduced pricereduced-price items and services. The government has enforced the Anti-Kickback Statute to reach large settlements with healthcarehealth care companies based on sham research or consulting and other financial arrangements with physicians. Further, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the FCA. Many states have similar laws that apply to their state health care programs as well as private payors.
The FCA imposes liability on persons who, among other things, (i) knowingly present or cause to be presented false or fraudulent claims for payment or approval by a federal health care program.program, (ii) knowingly make, use, or cause to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or (iii) avoid, decrease, or conceal an obligation to pay money to the federal government. The FCA has been used to prosecute persons submitting claims for payment that are inaccurate or fraudulent, that are for services not provided as claimed, or for services that are not medically necessary. Actions under the FCA may be brought by the U.S. Attorney General or as a qui tam action by a private individual in the name of the government. Violations of the FCA can result in significant monetary penalties and treble damages. The federal government is using the FCA, and the accompanying threat of significant liability, in its investigation and prosecution of pharmaceutical and biotechnology companies throughout the country, for example, in connection with the promotion of products for unapproved uses and other sales and marketing practices. The government has obtained multi-million and multi-billion dollar settlements under the FCA in addition to individual criminal convictions under applicable criminal statutes. Conviction or civil judgment for violating the FCA may result in exclusion from federal health care programs, and suspension and debarment from government contracts, and refusal of orders under existing government contracts. In addition, companies have been forced to implement extensive corrective action plans and have often become subject to consent decrees or corporate integrity agreements, restricting the manner in which they conduct their business. The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, also created federal criminal statutes that prohibit, among other things, knowingly and willfully executing a scheme to defraud any healthcarehealth care benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcarehealth care benefits, items or services. Given the significant size of actual and potential settlements, it is expected that the government will continue to devote substantial resources to investigating healthcarehealth care providers’ and manufacturers’ compliance with applicable fraud and abuse laws.
In addition, there has been a recent trend of increased federal and state regulation of payments made to physicians and other healthcarehealth care providers. The ACA,federal Physician Payments Sunshine Act, enacted as amended bypart of the Health CarePatient Protection and Education Reconciliation Act, or collectively, the Affordable Care Act among other things,of 2010, or the ACA, requires manufacturers of FDA-approved drugs, devices, biologics and medical supplies covered by Medicare, Medicaid or Medicaidthe Children’s Health Insurance Program to report, on an annual basis, to the U.S. Department of Health and Human ServicesCMS information related to payments or other transfers of value made by them to U.S.-licensed physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain advanced non-physician health care practitioners and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Failure to submit required information may result in civil monetary penalties. Certain states also mandate implementation of commercial compliance programs, impose restrictions on drug manufacturer marketing practices and/or require the tracking and reporting of gifts, compensation and other remuneration to physicians and other healthcarehealth care professionals.
The federal criminal statutes enacted under HIPAA impose criminal liability for knowingly and willfully executing, or attempting to execute, a scheme to defraud any health care benefit program, including private third-party payors, or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any health care benefit program; knowingly and willfully embezzling or
stealing from a health care benefit program; willfully preventing, obstructing, misleading, or delaying a criminal investigation of a health care offense; and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statements in connection with the delivery of or payment for health care benefits, items or services. Similar to the federal Anti-Kickback Statute, a person or entity need not have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
We may also be subject to data privacy and security regulation by both the federal government and the states in which it conducts its business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their respective implementing regulations imposes specifiedimpose specific requirements on covered entities relating to the privacy, security and transmission of individually identifiable health information, known as protected health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to “business associates,” defined as independent contractors or agents of covered entities that create, receive, maintain or transmit protected health information in connection with providing a service for or on behalf of a covered entity.entity for a function or activity regulated by HIPAA. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions. We are not a covered entity or a business associate under HIPAA; however, we are indirectly affected by HIPAA because the protected health information held by investigators conducting our clinical trials is subject to HIPAA and can only be used for our clinical research consistent with HIPAA requirements imposed on those investigators. In addition, state laws, such as the California Consumer Privacy Act, or the CCPA, govern the privacy and security of healththe personal information of individuals residing in such states, and may in certain circumstances, apply to health information. In addition to California, other states have implemented laws protecting identifiable health and personal information, and many of whichthese laws differ from each other in significant ways and may not have the same effect.be preempted by HIPAA, thus complicating compliance efforts.
PricingWe may also be subject to analogous state and rebate programs mustforeign laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving health care items or services reimbursed by non-governmental third-party payors, including private insurers, and may be broader in scope than their federal equivalents. The laws of some U.S. states and foreign jurisdictions require pharmaceutical companies to comply with the Medicaid rebate requirements ofpharmaceutical industry’s voluntary compliance guidelines and the U.S. Omnibus Budget Reconciliation Act of 1990relevant compliance guidance promulgated by the federal government or otherwise restrict payments that may be made to health care providers. In addition, certain state and more recent requirementsforeign laws and regulations require disclosures to regulatory agencies and/or commercial purchasers with respect to certain price increases that exceed a certain level as identified in the ACA. If products are made availablerelevant statutes, require drug manufacturers to authorized usersreport information related to payments and other transfers of the Federal Supply Schedulevalue to physicians and other health care providers, and restrict marketing practices or require disclosure of the General Services Administration, additional lawsmarketing expenditures and requirements apply. Prescription drug and biologic productspricing information. Some U.S. states also must meet applicable child-resistant packaging requirements under the U.S, Poison Prevention Packaging Act.require registration of pharmaceutical sales representatives.
If our operations are found to be in violation of any of such laws or any other governmental regulations that apply to it, we may be subject to penalties, including, without limitation, civil and criminal penalties, damages, fines, the curtailment or restructuring of our operations, exclusion from participation in federal and state healthcarehealth care programs and imprisonment, any of which could adversely affect our ability to operate our business and our financial results.
Also, the U.S. Foreign Corrupt Practices Act and similar worldwide anti-bribery laws generally prohibit companies and their intermediaries from making improper payments to foreign officials for the purpose of obtaining or retaining business. We cannot assure you that our internal control policies and procedures will protect us from reckless or negligent acts committed by our employees, future distributors, partners, collaborators or agents. Violations of these laws, or allegations of such violations, could result in fines, penalties or prosecution and have a negative impact on our business, results of operations and reputation.
General Data Protection Regulation
The collection, use, disclosure, transfer, or other processing of personal data regarding individuals in the EU, including personal health data, is subject to the EU’s General Data Protection Regulation, or GDPR, which became effective on May 25, 2018. The GDPR is wide-ranging in scope and imposes numerous requirements on companies that process personal data, including requirements relating to processing health and other sensitive data, obtaining
consent of the individuals to whom the personal data relates, providing information to individuals regarding data processing activities, implementing safeguards to protect the security and confidentiality of personal data, providing notification of data breaches, and taking certain measures when engaging third-party processors. The GDPR also imposes strict rules on the transfer of personal data to countries outside the EU, including the U.S., and permits data protection authorities to impose large penalties for violations of the GDPR, including potential fines of up to €20 million or 4% of annual global revenues, whichever is greater. The GDPR also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory authorities, seek judicial remedies, and obtain compensation for damages resulting from violations of the GDPR. The Company must comply with the GDPR in the performance of its clinical trials in the EU and relies on its CROs to implement appropriate safeguards and procedures relating to informed consent in order to ensure that trials are conducted in a manner consistent with the GDPR.
In July 2023, the European Commission adopted an adequacy decision for a new mechanism for transferring personal data from the EU to the United States – the EU-U.S. Data Privacy Framework, which provides EU individuals with several new rights, including the right to obtain access to their data, or obtain correction or deletion of incorrect or unlawfully handled data. In addition, the EU-U.S. Data Privacy Framework offers additional redress avenues for violations, including free of charge independent dispute resolution mechanisms and an arbitration panel. The adequacy decision followed the U.S.' signing of an executive order introducing new binding safeguards to address the points raised by the Court of Justice of the EU in its decision on a case known as Schrems II, which invalidated the previous EU-U.S. Privacy Shield. Notably, the new obligations were geared to ensure that data can be accessed by U.S. intelligence agencies only to the extent necessary and proportionate and to establish an independent and impartial redress mechanism to handle complaints from Europeans concerning the collection of their data for national security purposes. The European Commission will continually review developments in the United States along with its adequacy decision. Adequacy decisions can be adapted or even withdrawn in the event of developments affecting the level of protection in the applicable jurisdiction. Future actions of EU data protection authorities are difficult to predict. Some customers or other service providers may respond to these evolving laws and regulations by asking us to make certain privacy or data-related contractual commitments that we are unable or unwilling to make. This could lead to the loss of current or prospective customers or other business relationships.
Coverage and Reimbursement
Sales of our products approved for marketing by the FDA and foreign regulatory authorities will depend, in part, on the extent to which our products will be covered by third-party payors, such as government health programs, commercial or private insurance and managed care organizations. The process for determining whether a payor will provide coverage for a drug or biological product may be separate from the process for setting the price or reimbursement rate that the payor will pay for the drug or biological product. Third-party payors may limit coverage to specific drug or biological products on an approved list, or formulary, which might not include all of the FDA-approved drug or biological products for a particular indication. Third-party payors are increasingly challenging drug prices and examining the medical necessity and cost-effectiveness of medical products and services, in addition to their safety and efficacy.
In order to secure coverage and reimbursement for any product that might be approved for sale, a company may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of the product, in addition to the costs required to obtain FDA or other comparable regulatory approvals. Our drug or biological candidates may or may not be considered medically necessary or cost-effective or may require prior authorizations before use. A payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. Moreover, eligibility for reimbursement may not be available at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim payments for new products, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the product and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost products and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by third-party payors and by any future relaxation of laws that presently restrict imports of products from countries where they may be sold at lower prices than in the United States. In the United States, no uniformthird-party payors often rely upon CMS coverage policy ofand payment limitations in setting their own reimbursement policies, but they also have their own methods and approval process apart from CMS coverage and reimbursement determinations.
Accordingly, one third-party payor’s determination to provide coverage for drug or biological products exists. Accordingly, decisions regardinga product does not assure that other payors will also provide coverage for the extent of coverage and amount of reimbursement to be provided for any of our products will be made on a payor-by-payor basis. As a result, theproduct.
The coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that coverage and adequate reimbursement will be obtained.applied consistently or granted at all. The process for determining whether a payor will cover and provide reimbursement for a product may be separate from the process of seeking approval for or setting the price of the product. Even if reimbursement is provided, market acceptance of our products may be adversely affected if the amount of payment for our products proves to be unprofitable for health care providers or less profitable than alternative treatments or if administrative burdens make our products less desirable to use.
TheAdditionally, the United States government, state legislatures and foreign governments have shown significant interest in implementing cost containment programs to limit the growth of government-paid health care costs, including price-controls,price controls, restrictions on reimbursement and requirements for substitution of generic products for branded prescription drugs. For example, the ACA contains provisions that may reduce the profitability of drug products through increased rebates for drugs reimbursed by Medicaid programs, extension of Medicaid rebates to Medicaid managed care plans, mandatory discounts for certain Medicare Part D beneficiaries and annual fees based on pharmaceuticalbiopharmaceutical companies’ share of sales to federal health care programs. Adoption of general controls and measures, coupled with the tightening of restrictive policies in jurisdictions with existing controls and measures, could limit payments for pharmaceutical drugs.drugs and biologics. The Medicaid Drug Rebate Program requires pharmaceuticalbiopharmaceutical manufacturers to enter into and have in effect a national rebate agreement with the Secretary of the Department of Health and Human ServicesDHHS as a condition for states to receive federal matching funds for the manufacturer’s outpatient drugstherapeutic products furnished to Medicaid patients. The ACA made several changes to the Medicaid Drug Rebate Program, including increasing pharmaceuticalbiopharmaceutical manufacturers’ rebate liability by raising the minimum basic Medicaid rebate on most branded prescription drugs from 15.1% of average manufacturer price, or AMP, to 23.1% of AMP and adding a new rebate calculation for “line extensions” (i.e., new formulations, such as extended release formulations) of solid oral dosage forms of branded products, as well as potentially impacting their rebate liability by modifying the statutory definition of AMP. The ACA also expanded the universe of Medicaid utilization subject to drug rebates by requiring pharmaceuticalbiopharmaceutical manufacturers to pay rebates on Medicaid managed care utilization and by enlarging the population potentially eligible for Medicaid drug benefits. Since its enactment, there have been judicial and Congressional challenges to certain aspects ofAs another example, the ACA, and as a result certain sections of the ACA have not been fully implemented or effectively repealed. In particular, in December of 2018, a Texas U.S. District Court Judge ruled that the ACA is unconstitutional in its entirety because the individual mandate was repealed by Congress as part of the Tax Cuts and Jobs Act, effective January 1, 2019. On December 18, 2019, the U.S. Court of Appeals for the Fifth Circuit upheld the District Court ruling that the individual mandate was unconstitutional but remanded the case back to the District Court to determine whether other reforms enacted as part of the ACA but not specifically related to the individual mandate or health insurance could be severed from the rest of the ACA so as not to be declared invalid as well. On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case and allocated one hour for oral arguments, which occurred on November 10, 2020. A decision from the Supreme Court is expected to be issued in spring 2021. It is unclear how this litigation and other efforts to repeal and replace the ACA will impact the ACA and the pharmaceutical industry more generally. In addition, other legislative changes have been proposed and adopted since the ACA was enacted. These changes include aggregate reductions to Medicare payments to providers of up to 2% per fiscal year pursuant to the Budget Control Act of 2011, which began in 2013 and will remain in effect through 2030 unless additional Congressional action is taken. The Coronavirus Aid, Relief, and Economic Security Act, or the CARES Act, which was signed into law on March 27, 2020 and was designed to provide financial support and resources to individuals and businesses affected by the COVID-19 pandemic, suspended the 2% Medicare sequester from May 1, 2020 through December 31, 2020, and extended the sequester by one year, through 2030, in order to offset the added expense of the 2020 cancellation. The 2021 Consolidated Appropriations Act, was subsequently signed into law on December 27, 2020, incorporated extensive health care provisions and extendsamendments to existing laws, including a requirement that all manufacturers of drugs and biological products covered under Medicare Part B report the CARES Act suspension periodproduct’s average sales price, or ASP, to March 31, 2021.the DHHS beginning on January 1, 2022, subject to enforcement via civil money penalties.
TheUnder currently applicable U.S. law, certain products that are not self-administered by the patient (including injectable drugs) may be eligible for coverage under Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or the MMA, established thethrough Medicare Part D program to provide a voluntary prescription drug benefit to Medicare beneficiaries. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities that provide coverage of outpatient prescription drugs. UnlikeB. Medicare Part A and B Part D coverage is not standardized. While all Medicare drug plans must give at least a standard level of coverage setadministered by Medicare Part D prescription drug plan sponsors are not requiredAdministrative Contractors, which generally have the responsibility of making coverage decisions. Subject to paycertain payment adjustments and limits, Medicare generally pays for all covered Part D drugs, and each drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any formulary used by a Part D prescriptionB-covered drug plan must be developed and reviewed byor biologic based on a pharmacy and therapeutic committee. Government payment for somepercentage of the costs of prescription drugs may increase demand for products formanufacturer-reported Average Sales Price, which we receive marketing approval. However, any negotiated prices for our products covered by a Part D prescription drug plan likely will be lower than the prices we might otherwise obtain. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payors.
is regularly updated. For a drug or biological product to receive federal reimbursement under the Medicaid or Medicare Part B programs or to be sold directly to U.S. government agencies, the manufacturer must extend discounts to entities eligible to participate in the 340B drug pricing program.Drug Pricing Program. The maximum amount that a manufacturer may charge a 340B covered entity for a given product is the AMP reduced by the rebate amount paid by the manufacturer to Medicaid for each unit of that product. As of 2010, the ACA expanded the types of entities eligible to receive discounted 340B pricing, although, under the current state of the law, with the exception of children’s hospitals, these newly eligible entities will not be eligible to receive discounted 340B pricing on orphan drugs. In addition, as 340B drug pricing is determined based on AMP and Medicaid rebate data, the revisions to the Medicaid rebate formula and AMP definition described above could cause the required 340B discount to increase.
Moreover, thereThere has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products. In MarchFor example, the 2021 Consolidated Appropriations Act signed into law on December 27, 2020 incorporated extensive health care provisions and amendments to existing laws, including new requirements for (1) all manufacturers of drugs and biological products covered under Medicare Part B to report the Trump administration sent “principles” for drug pricingproduct’s ASP, to Congress, calling for legislation that would, among other things, capthe DHHS beginning on January 1, 2022, subject to enforcement via civil money penalties, (2)
certain Medicare plans to develop tools to display Medicare Part D beneficiary out-of-pocketprescription drug benefit information in real time, and (3) for group and health insurance issuers to report information on pharmacy expenses, provide an optionbenefit and drug costs to cap Medicare Part D beneficiary monthly out-of-pocket expenses,the Secretaries of the DHHS, the Department of Labor and place limits on pharmaceutical price increases. In addition, the Trump administration previously released a “Blueprint” to lower drugDepartment of the Treasury.
More recently, in August 2022, President Biden signed into the law the Inflation Reduction Act of 2022, or the IRA. Among other things, the IRA has multiple provisions that may impact the prices and reduce out of pocket costs of drugs that contained proposals to increase manufacturer competition, increase the negotiating power of certain federal healthcare programs, incentivize manufacturers to lower the list price of their products and reduce the out-of-pocket costs of drug products paidthat are both sold into the Medicare program and throughout the United States. Starting in 2023, a manufacturer of a drug or biological product covered by consumers. HHS has solicited feedbackMedicare Parts B or D must pay a rebate to the federal government if the drug product’s price increases faster than the rate of inflation. This calculation is made on some of these measures and has implemented others under its existing authority. For example, in September 2020, the FDA finalized a rulemaking to establish a system whereby state governmental entities could lawfully import and distribute prescription drugs sourced from Canada. Those new regulations became effective on November 30, 2020, although the impact of such future programs is uncertain and lawsuits have been filed challenging the government’s authority to promulgate them. While some of these and other measures may require additional authorization to become effective, Congressdrug product by drug product basis and the Trump administration have each indicatedamount of the rebate owed to the federal government is directly dependent on the volume of a drug product that theyis paid for by Medicare Parts B or D. Additionally, starting in payment year 2026, CMS will continue to seek new legislative and/negotiate drug prices annually for a select number of single-source Part D drugs without generic or administrative measures to controlbiosimilar competition. CMS will also negotiate drug costs. For example, on July 24, 2020, President Trump announced four executive orders related to prescriptionprices for a select number of Part B drugs starting for payment year 2028. If a drug pricingproduct is selected by CMS for negotiation, it is expected that attemptthe revenue generated from such drug will decrease. CMS has begun to implement severalthese new authorities and entered into the first set of agreements with pharmaceutical manufacturers to conduct price negotiations in October 2023. However, the Administration’s proposals, including a policy that would tie Medicare Part B drug prices to international drug prices; one that directed HHS to finalize the Canadian drug importation proposed rule previously issued by HHS (which has since been finalized, as noted above) and made other changes allowing for personal importation of drugs from Canada; one that directed HHS to finalize the rulemaking process on modifying the anti-kickback law safe harbors for plans, pharmacies, and pharmaceutical benefit managers after HHS confirms that the action is not projected to increase federal spending, Medicare beneficiary premiums, or patients’ total out-of-pocket costs (which HHS finalized in November 2020); and one that reduces costs of insulin and epinephrine auto-injectors to patients of federally qualified health centers. President Trump also issued another executive order on September 13, 2020 that directed HHS to undertake rulemaking in order to test an international reference pricing model for prescription drug products, which was also implemented by HHS and then challenged in federal court by industry groups in December 2020. The probability of success of these newly announced policies and theirIRA’s impact on the pharmaceutical industry in the United States remains uncertain, in part because multiple large pharmaceutical companies and other stakeholders (e.g., the U.S. prescription drug marketplaceChamber of Commerce) have initiated federal lawsuits against CMS arguing the program is unknown. There are likely to be continued political and legal challenges associated with implementing these reforms as theyunconstitutional for a variety of reasons, among other complaints. Those lawsuits are currently envisioned, notwithstanding the January 20, 2021 transition to a new presidential administration. ongoing.
At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceuticaldrug and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures.measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. For example, in recent years, several states have formed prescription drug affordability boards, or PDABs. Much like the IRA’s drug price negotiation program, these PDABs have attempted to implement upper payment limits, or UPLs, on drugs sold in their respective states in both public and commercial health plans. For example, in August 2023, Colorado’s PDAB announced a list of five prescription drugs that would undergo an affordability review. The effects of these efforts remain uncertain pending the outcomes of several federal lawsuits challenging state authority to regulate prescription drug payment limits. We expect that federal, state and local governments in the United States will continue to consider legislation directed at lowering the total cost of health care. In December 2020, the U.S. Supreme Court held unanimously that federal law does not preempt the states’ ability to regulate pharmaceuticalpharmacy benefit managers, (“PBMs”)or PBMs, and other members of the health care and pharmaceutical supply chain, an important decision that may lead to further and more aggressive efforts by states in this area. The Federal Trade Commission in mid-2022 also launched sweeping investigations in the practices of the PBM industry that could lead to additional federal and state legislative or regulatory proposals targeting such entities’ operations, pharmacy networks, or financial arrangements. Significant efforts to change the PBM industry as it currently exists in the United States may affect the entire pharmaceutical supply chain and the business of other stakeholders, including biopharmaceutical developers like us.
As noted above, the marketability of any products for which we receive regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. An increasing emphasis on cost containment measures in the United States has increased and we expect will continue to increase the pressure on pharmaceuticaldrug or biological product pricing. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
In some foreign countries, proposed pricing for a drug and biological products must be approved before the product may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. Some countries provide that drug and biological products may be marketed only after agreement on a reimbursement price has been reached. Some countries may require additional studies that compare the cost-effectiveness of our product candidate to currently available therapies (so called health technology assessment, or HTA) in order to obtain reimbursement or pricing approval. For example, the European UnionEU provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicines. A member state may approve a specific price for the product or it may instead adopt a
system of direct or indirect controls on the profitability of the company placing the product on the market. Other member states allow companies to fix their own drug prices but monitor and control prescription volumes and issue guidance to physicians to limit prescriptions. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceuticaldrug and biological products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European UnionEU do not follow price structures of the United States and generally tend to bybe priced significantly lower.
Foreign Regulation
In addition to regulations in the United States, we are and will be subject, either directly or through our distribution partners, to a variety of regulations in other jurisdictions governing, among other things, clinical trials and commercial sales and distribution of our products, if approved in such jurisdiction.
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in non-U.S. countries prior to the commencement of clinical trials or marketing of the product in those countries. The time required to obtain approval in other countries and jurisdictions might differ from and be longer than that required to obtain FDA approval. Regulatory approval in one country or jurisdiction does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country or jurisdiction may negatively impact the regulatory process in others. It is not yet clear how
Similar to the United Kingdom’s withdrawal fromStates, the European Union,various phases of non-clinical and clinical research that takes place in other countries are subject to significant regulatory controls. For example, the EU’s Clinical Trials Regulation, which took place oneffect in January 31, 2020, will affect2022, was enacted to simplify and streamline the approval of medicinal productsclinical trials in the United Kingdom.
Certain countries outsideregion. The main characteristics of the United States have processes that requireregulation include: a streamlined application procedure via a single entry point, the submission“EU portal” or Clinical Trial Information System; a single set of documents to be prepared and submitted for the application as well as simplified reporting procedures for clinical trial sponsors; and a harmonized procedure for the assessment of applications for clinical trials, which is divided in two parts. Part I, which contains scientific and medicinal product documentation, is assessed by the competent authorities of all EU member states in which an application for authorization of a clinical trial application much like an IND priorhas been submitted. Part II, which contains the national and patient-level documentation, will be assessed individually by each such EU member state. Any substantial changes to the commencement of human clinical trials. In Europe, for example, a clinical trial application,protocol or other information submitted with the CTA must be submittednotified to or approved by the relevant competent national health authorityauthorities and to independent ethics committeescommittees. Medicines used in each country in which a company plans to conduct clinical trials. Once the CTA is approvedtrials must be manufactured in accordance with a country’sapplicable cGMP requirements. Other national and EU-wide regulatory requirements clinical trials may proceed in that country.
The E.U. clinical trials legislation currently is undergoing a transition process mainly aimed at harmonizing and streamlining clinical-trial authorization, simplifying adverse-event reporting procedures, improvingalso apply. Currently, the supervision of clinical trials and increasing their transparency. Specifically, in April 2014, the new Clinical Trials Regulation, (E.U.) No 536/2014 (Clinical Trials Regulation) was adopted and it is anticipated to come into application in late 2020 or early 2021. The Clinical Trials Regulation will be directly applicable in all the E.U. Member States, repealing the current Clinical Trials Directive 2001/20/EC. Conduct of all clinical trials performed in the European Union will continue to be bound by currently applicable provisions until the new Clinical Trials Regulation becomes applicable. The extent to which ongoing clinical trials will be governed by the Clinical Trials Regulation will depend on when the Clinical Trials Regulation becomes applicable andclinical trial is initiated or on the duration of an ongoing trial. As of January 2023, all new clinical trials must comply with the individual clinical trial. If aClinical Trials Regulation. In addition, any clinical trial that was already under way as of January 1, 2023, and continues for more than three years from the day on which the Clinical Trials Regulation becomes applicable (i.e., January 31, 2025), the Clinical Trials Regulation will at that time begin to apply to the clinical trial.
The requirements and processes governing the conduct of clinical trials, product licensing, pricing and reimbursement in Europe vary from country to country, even though there is already some degree of legal harmonization in the E.U.EU member states resulting from the national implementation of underlying E.U.EU legislation. In all cases, the clinical trials are conducted in accordance with GCP and other applicable regulatory requirements. To obtain regulatory approval of a new drug or medicinal product in the European Union,EU, a sponsor must obtain approval of a marketing authorization application. The way in which a medicinal product can be approved in the European UnionEU depends on the nature of the medicinal product.
The centralized procedure results in a single marketing authorization granted by the European Commission that is valid across the European Union,EU, as well as in Iceland, Liechtenstein and Norway.Norway, or the European Economic Area. The centralized procedure is compulsory for human drugsmedicines that are: (i) derived from biotechnology processes, such as genetic engineering, (ii) contain a new active substance indicated for the treatment of certain diseases, such as HIV/AIDS, cancer, diabetes, neurodegenerative diseases, autoimmune and other immune dysfunctions and viral diseases, (iii) officially designated as “orphan drugs”orphan medicinal product and (iv) advanced-therapy medicines, such as gene-therapy, somatic cell-therapy or tissue-engineered medicines. The centralized procedure may at the request of the applicant also be used for human drugsmedicines which do not fall within the above mentioned categories if (a) the human drugmedicinal product contains a new active substance which washas not been previously authorized in the European Community before May 20, 2004;Economic
Area, or (b) the applicant shows thatEEA, the medicinal product constitutes a significant therapeutic, scientific or technical innovation, or thatwhere the granting of authorization in the centralized procedure is in the interests of patients or animalpublic health atin the European Community level.EEA.
Under the centralized procedure in the European Union,EU, the maximum timeframe for the evaluation of a marketing authorization application by the EMA is 210 days (excluding clock stops, when additional written or oral information is to be provided by the applicant in response to questions asked by the Committee for Medicinal Products for Human Use, or CHMP),CHMP, with adoption of the actual marketing authorization by the European Commission thereafter. Accelerated evaluation might be granted by the CHMP in exceptional cases, when a medicinal product is expected to be of a major public health interest from the point of view of therapeutic innovation, defined by three cumulative criteria: the seriousness of the disease to be treated; the absence of an appropriate alternative therapeutic approach, and anticipation of exceptional high therapeutic benefit. In this circumstance, EMA ensures that the evaluation for the opinion of the CHMP is completed within 150 days, excluding clock stops, and the opinion issued thereafter.
There are also two other possible routes to authorize medicinal products in several E.U.EU countries, which are available for investigational medicinal products for which the centralized procedure is not obligatory: the decentralized procedure and the mutual recognition procedure, or MRP. Using the decentralized procedure, an applicant may apply for simultaneous authorization in more than one E.U.EU country of a medicinal productsproduct that havehas not yet been authorized in any E.U.EU country and that dodoes not fall within the mandatory scope of the centralized procedure.
The MRP is applicable to the majority of conventional medicinal products and is based on the principle of recognition of an already existing national marketing authorization by one or more member states. The characteristic ofIn the MRP is that the procedure builds on an already existing marketing authorization in an E.U. member state which is used as reference in order to obtain marketing authorizations in other E.U. member states. In the MRP,process, a marketing authorization for a drug already exists in one or more member states of the E.U.EU and subsequently marketing authorization applications are made in other European UnionEU member states by referring to the initial marketing authorization. The member state in which the marketing authorization was first granted will then act as the reference member state. The member states where the marketing authorization is subsequently applied for act as concerned member states.
The MRP is based on the principle of the mutual recognition by E.U. member states of their respective national marketing authorizations. Based on a marketing authorization in After the reference member state the applicant may apply for marketing authorizations in other member states. In such case, the reference member state shall updatecompletes its existingmedicinal product assessment, report about the drug in 90 days. After the assessment is completed, copies of the report are sent to all member states, together with the approved summary of product characteristics, labeling and package leaflet. The concerned member states then have 90 days to recognize the decision of the reference member state and the summary of product characteristics, labeling and package leaflet. National marketing authorizations shall be granted within 30 days after acknowledgement of the agreement.
Should any Member StateEU member state refuse to recognize the marketing authorization by the reference member state, on the grounds of potential serious risk to public health, the issue will be referred to a coordination group. Within a timeframe of 60 days, member states shall, within the coordination group, make all efforts to reach a consensus. If this fails, the procedure is submitted to an EMA scientific committee for arbitration. The opinion of this EMA Committee is then forwarded to the European Commission, for the start of the decision-making process. As in the centralized procedure, this process entails consulting various European Commission Directorates General and the EU member states Standing Committee on Human Medicinal Products or Veterinary Medicinal Products, as appropriate.Products.
In the E.U.,EU, new chemical entities, sometimes referred to as new active substances, qualify for eight years of data exclusivity upon marketing authorization and an additional two years of market exclusivity. The data exclusivity, if granted, prevents regulatory authorities in the E.U.EU from referencing the innovator’s data to assess a generic or biosimilar application for eight years, after which generic marketing authorization can be submitted, and the innovator’s data may be referenced, but not approved for two years. The overall ten-year period can be extended to a maximum of 11 years if, during the first eight years of those 10ten years, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are determined to bring a significant clinical benefit in comparison with currently approved therapies.
The criteria for designating an orphan medicinal product in the European UnionEU are similar in principle to those in the United States. Under Article 3 of Regulation (EC) 141/2000, a medicinal product may be designated as orphan if (1) it is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition; (2) either (a) such condition affects no more than five in 10,000 persons in the European UnionEU when the application is made, or (b) the product, without the benefits derived from orphan status, would not generate sufficient return in the European UnionEU to justify investment; and (3) there exists no satisfactory method of diagnosis, prevention or treatment of such condition authorized for marketing in the European Union,EU, or if such a method exists, the product will be of significant benefit to those
affected by the condition, as defined in Regulation (EC) 847/2000. Orphan medicinal products are eligible for financial incentives such as reduction of fees or fee waivers and are, upon grant of a marketing authorization, entitled to ten years of market exclusivity for the approved therapeutic indication. The application for orphan designation must be submitted before the application for marketing authorization. The applicant will receive a fee reduction for the marketing authorization application if the orphan designation has been granted, but not if the designation is still pending at the time the marketing authorization is submitted. Orphan designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process.
The ten-year10-year market exclusivity for orphan products in the European UnionEU may be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation, for example, if the product is sufficiently profitable not to justify maintenance of market exclusivity. Additionally, marketing authorization may be granted to a similar product for the same indication at any time if:
•the second applicant can establish that its product, although similar, is safer, more effective or otherwise clinically superior;
•the applicant consents to a second orphan medicinal product application; or
•the applicant cannot supply enough orphan medicinal product.
We have obtained Orphan Medicinal Product Designations from the EMA for GPS in AML, MPM and MM.
For other countries outside of the United States and the European Union,EU, such as countries in Eastern Europe, Latin America or Asia, the requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country. In all cases, again, the clinical trials are conducted in accordance with GCP and the other applicable regulatory requirements.
If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension of clinical trials, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Health Care Reform in the U.S. and Potential Changes to Health Care Laws
FDAIn the United States and some foreign jurisdictions, there have been, and continue to be, several legislative and regulatory changes and proposed changes regarding the health care system that could prevent or delay marketing approval of product and therapeutic candidates, restrict or regulate post-approval activities, and affect the ability to profitably sell product and therapeutic candidates that obtain marketing approval. The FDA’s and other regulatory authorityauthorities’ policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our drugproduct and therapeutic candidates. For example, in December 2016, the 21st Century Cures Act, or Cures Act, was signed into law. The Cures Act, among other things, is intended to modernize the regulation of drugs and devices and to spur innovation, but its ultimate implementation is uncertain. In addition, in August 2017, the FDA Reauthorization Act was signed into law, which reauthorized the FDA’s user fee programs and included additional drug and device provisions that build on the Cures Act. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we otherwise may have obtained and we may not achieve or sustain profitability, which would adversely affect our business, prospects, financial condition and results of operations.
As previously mentioned, the primary trend in the US health care industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medical products and services, implementing reductions in Medicare and other health care funding and applying new payment methodologies. For example,In addition to the sweeping reforms contained in March 2010, the ACA was enacted, which, among(summarized above in the section entitled “Coverage and Reimbursement”), other things, increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program; introduced a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care plans; imposed mandatory discounts for certain Medicare Part D beneficiaries as a condition for manufacturers’ outpatient drugs coverage under Medicare Part D; and established a Center for Medicare Innovation at the US Centers for Medicare and Medicaid Services, or CMS, to test innovative payment and service delivery models to lower Medicare and Medicaid spending. The uncertainty related to the Affordable Care Act and the Trump Administration’s regulatory and executive actions pertaining to drug costs and drug pricing matters is described above under “Coverage, Pricing, and Reimbursement.”
Other legislative changes have been proposed and adopted in the United States since the Affordable Care Act that may affect health care expenditures. For example, on December 20, 2019, President Trump signed the 2020 Further Consolidated Appropriations Act for 2020 into law (P.L. 116-94) that includesincluded a piece of bipartisan legislation called the Creating and Restoring Equal Access to Equivalent Samples Act of 2019, or the “CREATESCREATES Act.” The CREATES Act aims to address the concern articulated by both the FDA and others in the industry that some brand manufacturers have improperly restricted the distribution of their products, including by invoking the existence of a REMS for certain products, to deny generic and biosimilar product developers access to samples of brand products. Because generic and biosimilar product developers need samples to conduct certain comparative testing required by the FDA, some have attributed the inability to timely obtain samples as a cause of delay in the entry of generic and biosimilar products. To remedy this concern, the CREATES Act establishes a private cause of action that permits a generic or biosimilar product developer to sue the brand manufacturer to compel it to furnish the necessary samples on “commercially reasonable, market-based terms.” Whether and how generic and biosimilar product developments will use this new pathway, as well as the likely outcome of any legal challenges to provisions of the CREATES Act, remain highly uncertain and its potential effects on our future commercial products are unknown. The Consolidated
Appropriations Act of 2021 signed into law by President Trump on December 27, 2020 also includes, among other things, a new requirement for patent information to be submitted to the FDA and published in a “Purple Book” that contains detailed information about each FDA-licensed biological product, analogous to the Orange Book that provides information about approved small-molecule drug products and their patent and exclusivity information under the Hatch-Waxman Amendments. In addition, the next cycle of Congressional reauthorization for FDA’s prescription drug, biologic, and medical device user fee programs must be completed by mid-2022 and that periodic must-pass legislation is typically used as a vehicle to implement federal policy changes or other substantive amendments to the FDCA.Act.
In addition to the IRA’s drug price negotiation provisions summarized above, President Biden’s Executive Order 14087, issued in October 2022, called for the CMS Innovation Center to prepare and submit a report to the White House on potential payment and delivery modes that would complement the IRA, lower drug costs, and promote access to innovative drugs. In February 2023, CMS published its report which described three potential models focusing on affordability, accessibility and feasibility of implementation for further testing by the CMS Innovation Center. As of January 2024, the CMS Innovation Center continues to test the proposed models and develop implementation plans.
52In the EU, many member states have increased the amount of discounts required on pharmaceuticals and these efforts could continue as countries attempt to manage health care expenditures, especially in light of the severe fiscal and debt crises experienced by many countries in the EU. The downward pressure on health care costs in general, particularly prescription products, has become intense. As a result, increasingly high barriers are being erected to the entry of new medicinal products. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various EU member states, and parallel trade, i.e., arbitrage between low-priced and high-priced member states, can further reduce prices. There can be no assurance that any country that has price controls or reimbursement limitations for medicinal products will allow favorable reimbursement and pricing arrangements for any products, if approved in those countries.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. We expect that additional state and federal health care reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for health care products and services. Moreover, if we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, our therapeutic candidates may lose any marketing approval that may have been obtained and we may not achieve or sustain profitability, which would adversely affect our business.business, prospects, financial condition and results of operations.
Corporate Information
Our principal executive offices are located at 7 Times Square, Suite 2503, New York, NY 10036, and our phone number is (646) 200-5278. Our website address is www.sellaslife.com.www.sellaslifesciences.com. We do not incorporate the information on our website into this annual reportAnnual Report on Form 10-K, and you should not consider such information part of this annual reportAnnual Report on Form 10-K.
We were incorporated on April 3, 2006 in Delaware as Argonaut Pharmaceuticals, Inc. On November 28, 2006, we changed our name to RXi Pharmaceuticals Corporation and began operations January 2007. On September 26, 2011, we changed our name to Galena Biopharma, Inc., or Galena. In December 2017, we completed a business combination, or the Merger, with SELLAS Life Sciences Group, Ltd., a privately held Bermuda exempted company, or Private SELLAS, and changed our name to “SELLAS Life Sciences Group, Inc.”
A copy of our Corporate Governance Guidelines, Code of Business Conduct and Ethics and the charters of the Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee are posted on our website, www.sellaslifesciences.com, under “Investors – Corporate Governance.”
ITEM 1A. RISK FACTORS
You should carefully consider carefully the risks and uncertainties described below, together with all of the other information in this annual reportAnnual Report on Form 10-K. We operate in a dynamic and rapidly changing industry that involves numerous risks and uncertainties. The risks and uncertainties described below are not the only ones we face. Other risks and uncertainties, including those that we do not currently consider material, may impair our business.
If any of the risks discussed below actually occur, our business, financial condition, operating results or cash flows could be materially adversely affected. This annual reportAnnual Report on Form 10-K also contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including the risks we face as described below and elsewhere in this annual reportAnnual Report on Form 10-K.
Our business has been and may continue to be adversely affected by COVID-19.
In December 2019, a respiratory disease caused by a novel strain of coronavirus, known as COVID-19, was reported to have surfaced in Wuhan, China. This coronavirus has since spread to other parts of the world, including the United States and Europe, and efforts to contain the spread of this coronavirus have intensified, resulting in travel bans and restrictions, quarantines, shelter-in-place orders and shutdowns. On March 11, 2020, the World Health Organization declared the COVID-19 outbreak as a “pandemic.” We have followed the guidelines from the U.S. Center for Disease Control (CDC) and have implemented recommended safety protocols from the CDC and the State of New York. Since March 2020, the spread of COVID-19 has caused us to modify our business practices (including curtailing employee travel and mandatory work-from-home policies where necessary), and we may need to take further actions as required by government authorities or that we determine are in the best interests of our employees, customers, partners and suppliers.
The COVID-19 pandemic has caused a widespread health crisis that could adversely affect the economies and financial markets of many countries, resulting in an economic downturn that could impact our operating results. During 2020, our business and operations, in particular our clinical development programs, were impacted by the coronavirus pandemic. The extent to which our business and operations could be impacted throughout 2021 will depend on future developments, which are highly uncertain and cannot be predicted, including the duration of the outbreak, the availability and efficacy of vaccines, new information which may emerge concerning the severity of COVID-19, the emergence of new mutations of COVID-19, and the actions to contain COVID-19 or treat its impact, among others. Given such continued uncertainty, we cannot estimate the impact of the COVID-19 pandemic on the Company's financial results or operations.
In particular, the continued spread of COVID-19 globally have adversely impacted and could continue to adversely impact our clinical trial operations, including the REGAL study, and could have an adverse impact on our business and financial results. For example, our clinical trials may suffer from slower than anticipated patient recruitment or enrollment and we may be forced to temporarily delay or pause ongoing trials. Further, if the spread of COVID-19 continues, the operations of manufacturers of our clinical supply of GPS could be significantly delayed as well and we risk a delay, default and/or nonperformance under our existing agreements. While we believe that we currently have sufficient supply of our product candidates to continue our ongoing clinical trials, such delays, defaults or non-performance could materially adversely affect our revenues, financial condition, profitability, and cash flows.
Risks RelatingRelated to Our Financial Position and Capital Needs
We have incurred substantial losses since our inception and anticipate that we will continue to incur substantial and increasing losses for the foreseeable future.
Investment in biopharmaceutical product development is highly speculative because it entails substantial upfront capital expenditures and significant risk that a product candidate will fail to prove effective, gain regulatory approval or become commercially viable. We do not have any products approved by regulatory authorities, have not generated any product revenues to date, and have incurred significant research, development and other expenses related to our ongoing operations. As a result, we have not been profitable and have incurred significant operating losses in every reporting period since our inception. For the years ended December 31, 20202023 and 2019,2022, we reported a net loss of $16.8$37.3 million and $19.3$41.3 million, respectively. As of December 31, 20202023 and 2019,2022, we had an accumulated deficit of $117.9$217.2 million and $101.1$179.9 million, respectively.
We do not expect to generate product revenues for many years, if at all. We expect to continue to incur significant expenses and operating losses for the foreseeable future. We anticipate these losses to increase as we continue to research, develop and seek regulatory approvals for our product candidates and any additional product candidates we may acquire, and potentially begin to commercialize product candidates that may achieve regulatory approval. We may also encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may adversely affect our business. The size of our future net losses will depend, in part, on the rate of future growth of our expenses and our ability to generate revenues. Our expenses will further increase as we:
•conduct additional clinical trials of our lead product, GPS, including the Phase 3 clinical trial evaluating GPS for AML, and our second clinical candidate, SLS009;
•seek marketing approval for any of our product candidates that successfully complete clinical trials;
•develop our outsourced manufacturing activities and establish sales, marketing and distribution capabilities, if we receive, or expect to receive, marketing approval for any product candidates;
•in-license or acquire the Phase 1/2 basket study in combination with pembrolizumab;rights to, and pursue development of, other products, product candidates or technologies;
•maintain, expand and protect our intellectual property portfolio;
•hire additional personnel, including clinical, manufacturing, quality control, quality assurance and other scientific personnel, sales and marketing personnel and general and administrative personnel; and
•seek marketing approval for any product candidates that successfully complete clinical trials;
•develop our outsourced manufacturing and commercial activities and establish sales, marketing and distribution capabilities, if we receive, or expect to receive, marketing approval for any product candidates;
•in-license or acquire the rights to, and pursue development of, other products, product candidates or technologies;
•maintain, expand and protect our intellectual property portfolio; and
•add operational, financial and management information systems and personnel.
We currently have no source of revenues from product sales. We may never generate such revenues or achieve profitability.
Currently, we do not generate any revenues from product sales or otherwise. Even if we are able to successfully achieve regulatory approval for our product candidates, we do not know when we will generate revenues or become profitable, if at all. Our ability to generate revenues from product sales and achieve profitability will depend on our ability to successfully commercialize products, including our current product candidates, and other product candidates that we may develop, in-license or acquire in the future. Our ability to generate revenues and achieve profitability also depends on a number of additional factors, including our ability to:
•successfully complete development activities, including the necessary clinical trials;
•complete and submit BLAs and NDAs to the FDA and obtain U.S. regulatory approval for indications for which there is a commercial market;
•complete and submit applications to foreign regulatory authorities in Europe, Asia and other jurisdictions;
•obtain regulatory approval in territories with viable market sizes;
•obtain coverage and adequate reimbursement from third parties, including government and private payors;
•set commercially viable prices for our products, if any;
•establish and maintain supply and manufacturing relationships with reliable third parties and/or build our own manufacturing facility and ensure adequate, legally globally compliant manufacturing of bulk drug substances and drug products to maintain that supply;
•develop distribution processes for our product candidates;
•develop commercial quantities of our product candidates, once approved, at acceptable cost levels;
•obtain additional funding, if required to develop and commercialize our product candidates;
•develop a commercial organization capable of sales, marketing and distribution for any products we intend to sell ourselves, in the markets in which we choose to commercialize on our own, and successfully enter into arrangements with third parties to sell, market, and distribute our products in markets where we choose not to commercialize on our own;
•achieve market acceptance of our products;
•attract, hire and retain qualified personnel; and
•protect our rights in our intellectual property portfolio.
Our revenues for any product candidate for which regulatory approval is obtained will be dependent, in part, upon the size of the markets in the territories for which it gains regulatory approval, the accepted price for the product, the ability to get reimbursement at any price, and whether we own the commercial rights for that territory. If the number of our addressable disease patients is not as significant as our estimates, the indication approved by regulatory authorities is narrower than we expect, or the reasonably accepted population for treatment is narrowed by competition, physician choice or treatment guidelines, we may not generate significant revenues from sales of such products, even if approved. In addition, we anticipate incurring significant costs associated with commercializing any approved product candidate. As a result, even if we generate revenues, we may not become profitable and may need to obtain additional funding to continue operations. If we fail to become profitable or are unable to sustain profitability on a continuing basis, then we may be unable to continue our operations at planned levels and may be forced to reduce our operations.
We will need additional financing to fund our operations and complete the development and, if approved, the commercialization of our product candidates. If we are unable to raise capital when needed, we could be forced to delay, reduce or eliminate our product development programs or commercialization efforts.
We expect our existing cash as of December 31, 2020 and the proceeds received in the first quarter of 2021 from the exercise of certain warrants to acquire share of common stock purchase will enable us to fund our operating expenses for at least the next 12 months. We expect to expend substantial resources for the foreseeable future to continue the clinical development and manufacturing of GPS, in particular the Phase 3 study of GPS in AML, and the Phase 1/2 basket study of GPS in combination with pembrolizumab.SLS009. Our existing cash will not be sufficient to complete such development activities and obtain regulatory approval for GPSour product candidates and, if we receive regulatory approval for GPS,our product candidates, commence commercialization activities, and we will need to raise significant additional capital to help us do so. In addition, our operating plan may change as a result of factors currently unknown to us, and we may need additional funds sooner than planned. If we are unable to obtain
sufficient funding for our operations, we may be delayed in pursuing our development programprograms for GPS.GPS and SLS009.
Our future capital requirements depend on many factors, including:
•the scope, progress, results and costs of our ongoing and planned development programs for our product candidates, as well as any additional clinical trials we undertake to obtain data sufficient to seek marketing approval for our product candidates in any indication;
•the timing of, and the costs involved in, obtaining regulatory approvals for our product candidates if our clinical trials are successful;
•the cost of commercialization activities for our product candidates, if any of these product candidates are approved for sale, including marketing, sales and distribution costs;
•the cost of manufacturing our product candidates for clinical trials in preparation for regulatory approval, including the cost and timing of process development, manufacturing scale-up and validation activities;
•our ability to establish and maintain strategic licensing or other arrangements and the financial terms of such agreements;
•the costs to in-license future product candidates or technologies;
•the costs involved in preparing, filing, prosecuting, maintaining, expanding, defending and enforcing patent claims, including litigation costs and the outcome of such litigation;
•the costs in defending and resolving future derivative and securities class action litigation;
•our operating expenses; and
•the emergence of competing technologies or other adverse market developments.
Additional funds may not be available when we need them on terms that are acceptable to us, or at all. WeMoreover, global and domestic events, such as public health crises, geopolitical unrest and domestic political events, have no committed source of additional capital. Moreover, the global coronavirus pandemic has caused and could continue to cause uncertainty and volatility in the capital markets which could impact our ability to raise capital. If adequate funds are not available to us on a timely basis, we may not be able to continue as a going concern or we may be required to delay, limit, reduce or terminate preclinical studies, clinical trials or other development activities for one or more of our product candidates or target indications, or delay, limit, reduce or terminate our establishment of sales and marketing capabilities or other activities that may be necessary to commercialize our product candidates.
Raising additional capital may cause dilution to our existing stockholders, restrict our operations or require us to relinquish rights to our product candidates on unfavorable terms to us.
We may seek additional capital through a variety of means, including through private and public equity offerings and debt financings, collaborations, strategic alliances and marketing, distribution or licensing arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, or through the issuance of shares under management or other types of contracts, or upon the exercise or conversion of outstanding derivative securities, the ownership interests of our stockholders will be diluted, and the terms of such financings may include liquidation or other preferences, anti-dilution rights, conversion and exercise price adjustments and other provisions that adversely affect the rights of our stockholders, including rights, preferences and privileges that are senior to those of our holders of common stock in the event of a liquidation. In such event, there is a possibility that once all senior claims are settled, there may be no assets remaining to pay out to the holders of our common stock. Debt financing, if available, could include covenants limiting or restricting our ability to take certain actions, such as incurring additional debt, making capital expenditures, entering into licensing arrangements, or declaring dividends and may require us to grant security interests in our assets, including our
intellectual property, and for our subsidiaries to guarantee our obligations. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to grant rights to develop and market products or product candidates that we would otherwise prefer to develop and market ourselves. If we raise additional funds through collaborations, strategic alliances, or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, products or product candidates or grant licenses on terms that may not be favorable to us.
Our cash and cash equivalents balance as of December 31, 2023 will fund our operations for less than one year.
As of December 31, 2023, we had a cash and cash equivalents balance of approximately $2.5 million. We expect our existing cash and cash equivalents balance as of December 31, 2023, will be insufficient to fund current planned operations for at least the next 12 months from the date of issuance of our consolidated financial statements for the year ended December 31, 2023, and that we will need to raise additional capital in order to continue our operations as currently planned. In the event that we are unable to obtain additional financing, we may be unable to continue as a going concern. There is no guarantee that we will be able to secure additional financing. Changes in our operating plans, our existing and anticipated working capital needs, the acceleration or modification of our development activities, any near-term or future expansion plans, increased expenses, potential acquisitions or other events may further affect our ability to continue as a going concern. See Note 2 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K for additional information on our assessment. Similarly, the report of our independent registered public accounting firm on our consolidated financial statements as of and for the year ended December 31, 2023 includes an Emphasis of Matter paragraph indicating that there is substantial doubt about our ability to continue as a going concern. Our consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty. If we cannot continue as a viable entity, our security holders may lose some or all of their investment in us.
Adverse developments affecting the financial services industry, such as actual events or concerns involving liquidity, defaults or non-performance by financial institutions or transactional counterparties, could adversely affect our current and projected business operations and its financial condition and results of operations.
Actual events involving limited liquidity, defaults, non-performance or other adverse developments that affect financial institutions, transactional counterparties or other companies in the financial services industry or the financial services industry generally, or concerns or rumors about any events of these kinds or other similar risks, have in the past and may in the future lead to market-wide liquidity problems. For example, on March 10, 2023, Silicon Valley Bank, or SVB, was closed by the California Department of Financial Protection and Innovation, which appointed the Federal Deposit Insurance Corporation, or the FDIC, as receiver. Similarly, on March 12, 2023, Signature Bank and Silvergate Capital Corp. were each swept into receivership. If any of our counterparties to any credit agreements, letters of credit or certain other financial instruments that we may enter into in the future were to be placed into receivership, we may be unable to access such funds. In addition, if any parties with whom we conduct business are unable to access funds pursuant to such instruments or lending arrangements with such a financial institution, such parties’ ability to pay their obligations to us or to enter into new commercial arrangements requiring additional payments to us could be adversely affected. Similar impacts have occurred in the past, such as during the 2008-2010 financial crisis.
Inflation and rapid increases in interest rates have led to a decline in the trading value of previously issued government securities with interest rates below current market interest rates. There is no guarantee that the U.S. Department of Treasury, FDIC and Federal Reserve Board will provide access to uninsured funds in the future in the event of the closure of other banks or financial institutions, or that they would do so in a timely fashion.
Although we assess our banking relationships as we believe necessary or appropriate, our access to funding sources and other credit arrangements in amounts adequate to finance or capitalize our current and projected future business operations could be significantly impaired by factors that affect us, the financial institutions with which we have arrangements directly, or the financial services industry or economy in general. These factors could include, among others, events such as liquidity constraints or failures, the ability to perform obligations under various types of financial, credit or liquidity agreements or arrangements, disruptions or instability in the financial
services industry or financial markets, or concerns or negative expectations about the prospects for companies in the financial services industry. These factors could involve financial institutions or financial services industry companies with which we have financial or business relationships, but could also include factors involving financial markets or the financial services industry generally.
In addition, investor concerns regarding the U.S. or international financial systems could result in less favorable commercial financing terms, including higher interest rates or costs and tighter financial and operating covenants, or systemic limitations on access to credit and liquidity sources, thereby making it more difficult for us to acquire financing on acceptable terms or at all. Any decline in available funding or access to our cash and liquidity resources could, among other risks, adversely impact our ability to meet our operating expenses, financial obligations or fulfill our other obligations, result in breaches of our financial and/or contractual obligations or result in violations of federal or state wage and hour laws. Any of these impacts, or any other impacts resulting from the factors described above or other related or similar factors not described above, could have material adverse impacts on our liquidity and our current and/or projected business operations and financial condition and results of operations.
In addition, any further deterioration in the macroeconomic economy or financial services industry could lead to losses or defaults by parties with whom we conduct business, which in turn, could have a material adverse effect on our current and/or projected business operations and results of operations and financial condition. For example, a party with whom we conduct business may fail to make payments when due, default under their agreements with us, become insolvent or declare bankruptcy. Any bankruptcy or insolvency, or the failure to make payments when due, of any counterparty of ours, or the loss of any significant relationships, could have material adverse impacts on our liquidity and our current and/or projected business operations and financial condition and results of operations.
Risks Related to the Development and Regulatory Approval of Our Product Candidates
Clinical-stage biopharmaceutical companies with product candidates in clinical development face a wide range of challenging activities which may entail substantial risk.
The success of our product candidates will depend on several factors, including the following:
•designing, conducting and successfully completing preclinical development activities, including preclinical efficacy and IND-enabling studies, for our product candidates or product candidates we are interested in in-licensing or acquiring;
•designing, conducting and completing clinical trials for our product candidates with positive results;
•receipt of regulatory approvals from applicable authorities;
•obtaining and maintaining patent and trade secret protection and regulatory exclusivity for our product candidates;
•making arrangements with third-party manufacturers, receiving regulatory approval of our manufacturing processes and our third-party manufacturers’ facilities from applicable regulatory authorities and ensuring adequate supply of drug product;
•manufacturing our product candidates at an acceptable cost;
•effectively launching commercial sales of our product candidates, if approved, whether alone or in collaboration with others;
•achieving acceptance of our product candidates, if approved, by patients, the medical community and third-party payors;
•effectively competing with other therapies;
•if our products candidates are approved, obtaining and maintaining coverage and adequate reimbursement by third-party payors, including government payors, for our product candidates;
•complying with all applicable regulatory requirements, including FDA current Good Clinical Practices, or cGCP, current Good Manufacturing Practices, orGCP and cGMP andrequirements, as well as, standards, rules and regulations governing promotional and other marketing activities;
•maintaining a continued acceptable safety profile of the products during development and following approval; and
•maintaining and growing an organization of scientists and business peoplebusinesspeople who can develop and commercialize our product candidates.
If we do not achieve one or more of these factors in a timely manner or at all, we could experience significant delays or an inability to successfully develop and commercialize our product candidates, which could materially harm our business.
Our lead product candidate, GPS, represents a new therapeutic approach that presents significant challenges.
Our future success is substantially dependent on the successful development of WT1 peptide immunotherapies in general and GPS in particular. Because this program represents a new approach to cancer immunotherapy for the treatment of cancer and other diseases, developing and commercializing GPS subjects us to a number of challenges, including:
•obtaining regulatory approval from the FDA and other regulatory authorities, which have very limited experience with the development and commercialization of WT1 cancer immunotherapies;
•obtaining the components required for the administration of GPS (i.e., GPS, GM-CSF, and Montanide) from three separate sources, the subsequent separate storage requirements for each of these components and the separate delivery of these components to the administration location;
•utilizing GPS in combination with other therapies, which may increase the risk of adverse side effects;
•sourcing clinical and, if approved, commercial supplies for the materials used to manufacture and process GPS;
•developing a manufacturing process used in connection with GPS that will yield a satisfactory product that is safe, effective, scalable and profitable;
•establishing sales and marketing capabilities after obtaining any regulatory approval to gain market acceptance; and
•obtaining coverage and adequate reimbursement from third-party payors and government authorities.
Moreover, public perception of safety issues, including adoption of new therapeutics or novel approaches to treatment, may adversely influence the willingness of subjects to participate in clinical trials, or if approved, of physicians to subscribe to the novel treatment mechanics. Physicians, hospitals and third-party payors often are slow to adopt new products, technologies and treatment practices that require additional educational upfront costs and training. Physicians may not be willing to undergo training to adopt this novel therapy, may decide the therapy is too complex to adopt without appropriate training and may choose not to administer the therapy. Based on these and other factors, hospitals and payors may decide that the benefits of this new therapy do not or will not outweigh their costs.
We may find it difficult to enroll patients in our clinical trials due to the impact of COVID-19 and given theThe limited number of patients who have the diseases for which our product candidates are being studied, has made it more difficult to enroll patients in our clinical trials, which could delay or prevent the start of clinical trials for our product candidates.
Identifying and qualifying patients to participate in clinical trials of our current and future product candidates is essential to our success. The timing of our clinical trials depends in part on the rate at which we can recruit patients to participate in clinical trials of our product candidates, and we may experience delays in our clinical trials if we encounter difficulties in enrollment. If we experience delays in our clinical trials, the timeline for obtaining regulatory approval of our product candidates will most likely be delayed.
Many factors may affect our ability to identify, enroll and maintain qualified patients, including the following:
•disruptions caused by the global coronavirus pandemic;shortages of personnel at our clinical sites;
•eligibility criteria of our ongoing and planned clinical trials with specific characteristics appropriate for inclusion in our clinical trials;
•design of the clinical trial;
•size and nature of the patient population;
•patients’ perceptions as to risks and benefits of the product candidate under study and the participation in a clinical trial generally in relation to other available therapies, including any new drugs that may be approved for the indications we are investigating;
•the availability and efficacy of competing therapies and clinical trials;
•pendency of other trials underway in the same patient population;
•willingness of physicians to participate in our planned clinical trials;
•severity of the disease under investigation;
•proximity of patients to clinical sites;
•patients who do not complete the trials for personal reasons; and
•issues with contract research organizations, or CROs, and/or with other vendors that handle our clinical trials.
For example, we have observed that, at certain times and in certain instances, clinical site initiations and patient enrollment may have been delayed due to prioritization of hospital resources towards the COVID-19 pandemic. Clinicians and patients may not have been able to comply with clinical trial protocols if quarantines impeded patient movement or interrupted operations at sites. Certain newly initiated sites have taken longer than expected to be fully operational. Additionally, several European Union countries in which we plan to initiate clinical sites, including Germany, France, and Italy, continue to impose restrictions in response to the continued surge in coronavirus cases throughout the European Union. We believe that these factors have had and could continue to have an impact on the projected timelines for enrollment of patients in the REGAL study.
Moreover, theThe indication being studied in our Phase 3 clinical trial for GPS, i.e., patients with AML who have achieved Crem2,CR2, is an orphan indication. Moreover,In addition, only CRem2those CR2 patients who meet specific inclusion criteria will beare eligible to participate in the study. Primary entry restrictions include demonstrating adequate hematologic recovery and not being candidates for bone marrow transplants and not being eligible for treatments targeted at certain mutations common in significant proportions of AML patients.transplants. The estimated prevalence of newly diagnosed AML patients is 12,000 toapproximately 20,000 cases in the United States annually (across all ages) with only a subset of this group having achieved CRem2CR2 and only a further subset of this groupthe CR2 subset satisfying the enrollment criteria for our AML Phase 3 clinical trial.
We may not be able to initiate or continue to support clinical trials of our product candidates for one or more indications, or any future product candidates if we are unable to locate and enroll a sufficient number of eligible participants in these trials as required by the FDA or other regulatory authorities. Even if we are able to enroll a sufficient number of patients in our clinical trials, if the pace of enrollment is slower than we expect, the development costs for our product candidates may increase and the completion of our trials may be delayed or our trials could become too expensive to complete.
Congress also recently amended the FDCA to require sponsors of a Phase 3 clinical trial, or other “pivotal study” of a new drug or biologic to support marketing authorization, to design and submit a diversity action plan for such clinical trial. The action plan must describe appropriate diversity goals for enrollment, as well as a rationale for the goals and a description of how the sponsor will meet them. Our Phase 3 REGAL trial of GPS for AML patients who
have achieved CR2 was initiated before this requirement became effective, but for any future Phase 3 trials we plan to conduct, including any registrational study for SLS009, we must submit a diversity action plan to the FDA by the time we submit plans for such Phase 3, or pivotal study, protocol to the agency for review as part of an IND, unless we are able to obtain a waiver for some or all of the requirements for a diversity action plan. It is unknown at this time how the diversity action plan may affect the planning and timing of any future Phase 3 trial for our product candidates or what specific information FDA will expect in such plans. However, initiation of such trials may be delayed if the FDA objects to our proposed diversity action plans for any future Phase 3 trial for our product candidates, and we may experience difficulties recruiting a diverse population of patients in attempting to fulfill the requirements of any approved diversity action plan.
If we experience delays in the completion of, or termination of, any clinical trials of our current or future product candidates, the commercial prospects of our product candidates could be harmed, and our ability to generate product revenue from any of these product candidates could be delayed or prevented. In addition, any delays in completing our clinical trials would likely increase our overall costs, impair product candidate development and jeopardize our ability to obtain regulatory approval relative to our current plans. Any of these occurrences may harm our business, financial condition, and prospects significantly.
The results of preclinical studies or earlier clinical trials are not necessarily predictive of future results. Our existing product candidates in clinical trials, and any other product candidates that may advance into clinical trials, may not have favorable results in later clinical trials or receive regulatory approval.
Success in preclinical studies and early clinical trials does not ensure that later clinical trials will generate adequate data to demonstrate the efficacy and safety of an investigational drug. A number of companies in the pharmaceutical and biotechnology industries, including those with greater resources and experience than us, have suffered significant setbacks in clinical trials, even after seeing promising results in earlier preclinical studies or clinical trials. Any of our product candidates that are in, or may advance to, clinical trials may not succeed in clinical trials despite promising preclinical data. For example, with respect to GPS, a broadly similar anti-cancer peptide immunotherapeutic against melanoma-specific antigen being developed by GlaxoSmithKline for advanced unresectable melanoma initially produced positive efficacy data in a Phase 2 clinical study, but subsequently failed to prove more beneficial than placebo in a controlled, blinded and randomized Phase 3, registration-enabling clinical trial in the same indication in patients after tumor resection.
Despite the results reported in earlier preclinical studies or clinical trials for our product candidates, we do not know whether the clinical trials we may conduct will demonstrate adequate efficacy and safety to result in regulatory approval to market GPS or any of our product candidates for a particular indication, either as a monotherapy or in combination, in any particular jurisdiction. Efficacy data from prospectively designed trials may differ significantly from those obtained from retrospective subgroup analyses. If later-stage clinical trials do not produce favorable results, our ability to achieve regulatory approval for GPSour product candidates may be adversely impacted. Even if we believe that we have adequate data to support an application for regulatory approval to market any of our current or future product candidates, the FDA or other regulatory authorities may not agree and may require that we conduct additional clinical trials.
Interim, topline and preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary, interim or topline data from our clinical trials. These interim updates are based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to a particular study or trial. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully evaluate all data. As a result, the topline results or preliminary data that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Topline data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline data should be viewed with caution until the final data are available. In addition, we may report interim analyses of only certain endpoints rather than all
endpoints. Interim data from clinical trials that we complete are subject to the risk that one or more of the clinical outcomes may materially change as more patient data becomes available. Adverse changes between interim data and final data could adversely affect our business and prospects and could result in volatility in the price of our common stock.
We are developing and may continue to develop our programs in combination with other therapies, which exposes us to additional risks.
We are currently investigating SLS009 in combination with aza/ven ina Phase 2a clinical trial and we may continue to develop clinical candidates in combination with one or more currently approved cancer therapies or therapies currently in clinical development. Patients may not be able to tolerate our product candidates in combination with other therapies or dosing of our product candidates in combination with other therapies may have unexpected consequences. Even if any of our product candidates were to receive marketing approval or be commercialized for use in combination with other existing therapies, we would continue to be subject to the risks that the FDA, EMA or other comparable foreign regulatory authorities could revoke approval of the therapy used in combination with any of our product candidates, or safety, efficacy, manufacturing or supply issues could arise with these existing therapies. In addition, it is possible that existing therapies with which our product candidates are approved for use could themselves fall out of favor or be relegated to later lines of treatment. This could result in the need to identify other combination therapies for our product candidates or our own products being less successful commercially.
We may also evaluate our product candidates in combination with one or more other cancer therapies that have not yet been approved for marketing by the FDA, EMA or comparable foreign regulatory authorities. If the FDA, EMA or other comparable foreign regulatory authorities do not approve or revoke their approval of these other therapies, or if safety, efficacy, commercial adoption, manufacturing or supply issues arise with the therapies we choose to evaluate in combination with our product candidates, we may be unable to obtain approval of or successfully market our product candidates.
Additionally, if the third-party providers of therapies or therapies in development used in combination with our product candidates are unable to produce sufficient quantities for clinical trials or commercialization of our product candidates, or if the cost of combination therapies are prohibitive, our development and commercialization efforts would be impaired, which would have an adverse impact on our business, financial condition, results of operations and growth prospects.
Clinical drug development involves a lengthy and expensive process with an uncertain outcome.
Clinical testing is expensive and can take many years to complete, with the outcome inherently uncertain. Failure can occur at any time during the clinical trial process. Before obtaining approval from regulatory authorities for the sale of any product candidate, we must conduct extensive clinical trials to demonstrate the safety and efficacy of our product candidates in humans. Prior to initiating clinical trials, a sponsor must complete extensive preclinical testing of a product candidate, including, in most cases, preclinical efficacy experiments as well IND-enabling toxicology studies. These experiments and studies may be time-consuming and expensive to complete. The necessary preclinical testing may not be completed successfully for a preclinical product candidate and a potentially promising product candidate may therefore never be tested in humans. Once it commences, clinical testing is expensive, difficult to design and implement, can take many years to complete and is uncertain as to outcome. A failure of one or more clinical trials can occur at any stage of testing. The outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results. Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless failed to obtain marketing approval of their products. We may experience numerous unforeseen events during drug development that could delay or prevent our ability to receive marketing approval or commercialize our product candidates. In particular, clinical trials of our product candidates may produce inconclusive or negative results. We have limited data regarding the safety, tolerability and efficacy of GPS administered as monotherapy or in combination with PD-1 inhibitors.inhibitors or for SLS009 as monotherapy and in combination with other therapeutics, including aza/ven. For a further discussion of the safety risks in our trials, see the risk factor herein entitled "Our current and future product candidates, the methods used to deliver them or their dosage levels may cause undesirable side effects or have other properties that could delay or
prevent their regulatory approval, limit the commercial profile of an approved label or result in significant negative consequences following any regulatory approval."
Clinical trials also require the review and oversight of an institutional review board, or IRB. An inability or delay in obtaining IRB approval could prevent or delay the initiation and completion of clinical trials, and the FDA may decide not to consider any data or information derived from a clinical investigation not subject to initial and continuing IRB review and approval.
We may experience delays in our ongoing or future clinical trials, and we do not know whether planned clinical trials will begin or enroll subjects on time, will need to be redesigned or will be completed on schedule, if at all. There can be no assurance that the FDA will not put clinical trials of any of our product candidates on clinical hold in the future. Clinical trials may be delayed, suspended or prematurely terminated for a variety of reasons, such as:
•delay or failure in reaching agreement with the FDA or a comparable foreign regulatory authority on a clinical trial design that we are able to execute;
•delay or failure in obtaining authorization to commence a trial or inability to comply with conditions imposed by a regulatory authority regarding the scope or design of a trial;
•delay or failure in reaching agreement on acceptable terms with prospective contract research organizations, or CROs, and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites;
•delay or failure in obtaining IRB approval or the approval of other reviewing entities, including comparable foreign regulatory authorities, to conduct a clinical trial at each site;
•withdrawal of clinical trial sites from our clinical trials or the ineligibility of a site to participate in our clinical trials;
•the impact of COVID-19 on the operations of clinical sites;
•clinical sites and investigators deviating from trial protocol, failing to conduct the trial in accordance with regulatory requirements, or dropping out of a trial;
•inability to identify and maintain a sufficient number of trial sites, many of which may already be engaged in other clinical trial programs, including some that may be for the same indication;
•failure of our third-party clinical trial managers, CROs, clinical trial sites, contracted laboratories or other third-party vendors to satisfy their contractual duties, meet expected deadlines or return trustworthy data;
•delay or failure in adding new trial sites;
•delay or failure in recruiting and enrolling suitable subjects to participate in a trial;
•delay or failure in subjects completing a trial or returning for post-treatment follow-up;
•interim results or data that are ambiguous or negative or are inconsistent with earlier results or data;
•alteration of trial design necessitated by re-evaluation of design assumptions based upon observed data;
•feedback from the FDA, the IRB, DSMB or a comparable foreign regulatory authority, the IRB, or DSMB, or results from earlier stage or concurrent preclinical studies and clinical trials, that might require modification to the protocol for a trial;
•a decision by the FDA the IRB, a comparableor foreign regulatory authority, the IRB, or us, or a recommendation by a DSMB or comparable foreign regulatory authority, to suspend or terminate clinical trials at any time for safety issues or for any other reason;
•unacceptable risk-benefit profile, unforeseen safety issues or adverse side effects;
•failure to demonstrate a benefit from using a product candidate;
•difficulties in manufacturing or obtaining from third parties sufficient quantities of a product candidate to start or to use in clinical trials;
•lack of adequate funding to continue a trial, including the incurrence of unforeseen costs due to enrollment delays, requirements to conduct additional studies or increased expenses associated with the services of our CROs and other third parties; or
•changes in governmental regulations or administrative actions or lack of adequate funding to continue a clinical trial.
If we experience delays in the completion or termination of any clinical trial of our product candidates, the approval and commercial prospects of such product candidates will be harmed, delaying our ability to generate product revenues from such product candidate and our costs will most likely increase. The required regulatory approvals may also be delayed, thereby jeopardizing our ability to commence product sales and generate revenues and the period of commercial exclusivity for our products may be decreased. Regulatory approval of our product candidates may be denied for the same reasons that caused the delay.
Risks associated with operating in foreign countries could materially adversely affect our product development.
We may conduct future studiesFor certain of our clinical trials, we have clinical sites in countries outside of the United States. Consequently, we may be subject to risks related to operating in foreign countries. Risks associated with conducting operations in foreign countries include:
•differing regulatory requirements for drug approvals and regulation of approved drugs in foreign countries; more stringent privacy requirements for data to be supplied to our operations in the United States, e.g., General Data Protection RegulationGDPR in the European Union;EU;
•unexpected changes in tariffs, trade barriers and regulatory requirements; economic weakness, including inflation, or political instability in particular foreign economies and markets; compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; foreign taxes, including withholding of payroll taxes;
•differing payor reimbursement regimes, governmental payors or patient self-pay systems and price controls;
•foreign currency fluctuations, which could result in increased operating expenses or reduced revenues, and other obligations incident to doing business or operating in another country;
•workforce uncertainty in countries where labor unrest is more common than in the United States;
•continued uncertainties related to the withdrawal of the United Kingdom from the European UnionEU (known as "Brexit") and its financial, trade, regulatory and legal implications, which could lead to legal uncertainty and potentially divergent national laws and regulations as the United Kingdom determines which EU laws to replace or replicate, and which may further create global economic uncertainty, which could materially adversely affect our business, business opportunities, results of operations, financial condition, and cash flows;
•the COVID-19 pandemic, which has resulted in global travel restrictions;
•production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad, including those that may result from the recent coronavirus outbreak; and
•business interruptions resulting from geopolitical actions, including war and terrorism.
Global, market and economic conditions may negatively impact our business, financial condition and share price.
The results of our operations could be adversely affected by general conditions in the global economy, the global financial markets and the global political conditions. The United States and global economies have recently faced growing inflation, higher interest rates and a potential recession. Furthermore, a prolonged economic downturn, including a recession or depression resulting from public health crises such as a pandemic or ongoing political disruption such as the war between Ukraine and Russia and the conflict involving Israel and Hamas could result in a variety of risks to our business, including weakened demand for our programs and development candidates, if approved, relationships with any vendors or business partners located in affected geographies and our ability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy or political disruption, including any international trade disputes, particularly between the United States and China, could also strain our existing or future partnerships, manufacturers or suppliers, possibly resulting in disruption to our clinical trials or supply, or cause our customers to delay making payments for our potential products. Any of the foregoing could seriously harm our business, and we cannot anticipate all of the ways in which the political or economic climate and financial market conditions could seriously harm our business.
Although we do not currently have any clinical study sites in Russia or Ukraine, economic, political and social conditions resulting from Russia’s invasion of Ukraine could materially disrupt our clinical trials, increase our costs and may disrupt planned clinical development activities. For example, we currently have clinical sites for our REGAL study in Poland, a country that borders Ukraine and has been impacted by an influx of Ukrainian refugees resulting from Russia’s invasion of Ukraine. Furthermore, we rely on suppliers in the EU. To the extent the conflict between Ukraine and Russia adversely impacts the ability of our suppliers to distribute the supplies we need for our clinical trials, or such distribution cannot be done on a timely basis, the timing for completing our clinical trials may be adversely impacted.
Continued increases in inflation could raise our costs for commodities, labor, materials and services and other costs required to grow and operate our business, and failure to secure these on reasonable terms may adversely impact our financial condition. Additionally, increases in inflation, along with the uncertainties surrounding geopolitical developments and global supply chain disruptions, have caused, and may in the future cause, global economic uncertainty and uncertainty about the interest rate environment. A failure to adequately respond to these risks could have a material adverse impact on our financial condition, results of operations or cash flows. In response to high levels of inflation and recession fears, the U.S. Federal Reserve, the European Central Bank, and the Bank of England have raised, and may continue to raise, interest rates and implement fiscal policy interventions. Even if these interventions lower inflation, they may also reduce economic growth rates, create a recession, and have other similar effects.
The U.S. debt ceiling and budget deficit concerns have increased the possibility of credit-rating downgrades and economic slowdowns, or a recession in the United States. Although U.S. lawmakers have previously passed legislation to raise the federal debt ceiling on multiple occasions, there is a history of ratings agencies lowering or threatening to lower the long-term sovereign credit rating on the United States given such uncertainty. On August 1, 2023, Fitch Ratings downgraded the United States’ long-term foreign currency issuer default rating to AA+ from AAA as a result of these repeated debt ceiling and budget deficit concerns. The impact of this or any further downgrades to the U.S. government’s sovereign credit rating or its perceived creditworthiness could adversely affect the U.S. and global financial markets and economic conditions.
If the equity and credit markets deteriorate, it may make any necessary equity or debt financing more difficult to secure, more costly or more dilutive. Failure to secure any necessary financing in a timely manner and on favorable terms could harm our growth strategy, financial performance and stock price and could require us to delay or abandon plans with respect to our business, including clinical development plans. Further, recent developments in the banking industry could adversely affect our business. If the financial institutions with which we do business enter receivership or become insolvent in the future, there is no guarantee that the Department of the Treasury, the Federal Reserve and the FDIC will intercede to provide us and other depositors with access to balances in excess of the $250,000 FDIC insurance limit, that we would be able to access our existing cash, cash equivalents and investments, that we would be able to maintain any required letters of credit or other credit support arrangements, or that we would be able to adequately fund our business for a prolonged period of time or at all, any of which could have a material adverse effect on our business, financial condition and results of operations. We cannot predict the impact that the high market volatility and instability of the banking sector more broadly could have on economic
activity and our business in particular. In addition, there is a risk that one or more of our current service providers, manufacturers or other third parties with which we conduct business may not survive difficult economic times, the ongoing conflict between Russia and Ukraine, the war between Israel and Hamas, the instability of the banking sector, and the uncertainty associated with current worldwide economic conditions, which could directly affect our ability to attain our operating goals on schedule and on budget.
Our partnerships in China subject us to risks and uncertainties relating to the laws and regulations of China and the changes in relations between the United States and China.
Under its current leadership, the government of China has been pursuing economic reform policies, including by encouraging foreign trade and investment. However, there is no assurance that the Chinese government will continue to pursue such policies, that such policies will be successfully implemented, that such policies will not be significantly altered, or that such policies will be beneficial to our partnerships in China. China’s system of laws can be unpredictable, especially with respect to foreign investment and foreign trade. The U.S. government has called for substantial changes to foreign trade policy with China and has raised, and has proposed to further raise in the future, tariffs on several Chinese goods. China has retaliated with increased tariffs on U.S. goods. Moreover, China’s legislature has adopted a national security law to substantially change the way Hong Kong has been governed since the territory was handed over by the United Kingdom to China in 1997. This law increases the power of the central government in Beijing over Hong Kong, limit the civil liberties of residents of Hong Kong and could restrict the ability of businesses in Hong Kong to continue to conduct business or to continue to with business as previously conducted. The U.S. State Department has indicated that the United States no longer considers Hong Kong to have significant autonomy from China. The U.S. State Department also previously enacted sanctions related to China’s governing of Hong Kong, and the United States may impose the same tariffs and other trade restrictions on exports from Hong Kong that it places on goods from mainland China. Any further changes in U.S. trade policy could trigger retaliatory actions by affected countries, including China, resulting in trade wars. For example, the Uyghur Forced Labor Prevention Act bans imports from China’s Xinjiang Uyghur Autonomous Region unless it can be shown that the goods were not produced using forced labor and this legislation may have an adverse effect on global supply chains which could adversely impact our business and results of operations. Additionally, the biopharmaceutical industry in particular in China is strictly regulated by the Chinese government. Changes to Chinese regulations affecting biopharmaceutical companies are also unpredictable. Any regulatory changes and changes in United States and China relations may have a material adverse effect on our partnerships in China which could materially harm our business and financial condition.
Climate change or legal, regulatory or market measures to address climate change may negatively affect our business, results of operations, cash flows and prospects.
Climate change may potentially negatively affect our business and results of operations, cash flows and prospects in the future. We could be exposed to physical risks (such as extreme weather conditions or rising sea levels), risks in transitioning to a low-carbon economy (such as additional legal or regulatory requirements, changes in technology, market risk and reputational risk), and social and human effects (such as population dislocations and harm to health and well-being) associated with climate change. These risks can be either acute (short-term) or chronic (long-term).
Extreme weather and sea-level rise could pose physical risks to facilities of our suppliers. Such risks include losses incurred as a result of physical damage to facilities, loss or spoilage of inventory, and business interruption caused by such natural disasters and extreme weather events which could disrupt our operations and supply chains that may result in increased costs.
New legal or regulatory requirements may be enacted to prevent, mitigate, or adapt to the implications of a changing climate and its effects on the environment. These regulations, which may differ across jurisdictions, could result in us being subject to new or expanded carbon pricing or taxes, increased compliance costs, restrictions on greenhouse gas emissions, investment in new technologies, increased carbon disclosure and transparency, upgrade of facilities to meet new building codes, and the redesign of utility systems, which could increase our operating costs, including the cost of electricity and energy used by us. Our supply chain would likely be subject to these same transitional risks and would likely pass along any increased costs to us.
Our current and future product candidates, the methods used to deliver them or their dosage levels may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label or result in significant negative consequences following any regulatory approval.
Undesirable side effects caused by our current or future product candidates, their delivery methods or dosage levels could cause us, the IRB, DSMB, or the FDA or comparable regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval or termination of clinical trials by the FDA or other comparable foreign regulatory authority; an independent DSMB that is governing our clinical trials; or an IRB, that approves and, monitors biomedical research to protect the rights and welfare of human subjects.trials. For example, although no high-grade delayed type hypersensitivity in the skin or systemic anaphylaxis events have been noted after GPS administration in patients treated in our clinical studies to date, it is theoretically possible that such toxicities, or other type of adverse events, may occur in future clinical studies. As a result of safety or toxicity issues that we may experience in our clinical trials, or negative or inconclusive results from the clinical trials of others for drug candidates similar to our own, we may not receive approval to market any product candidates, which could prevent us from ever generating revenues or achieving profitability. Results of our trials could reveal an unacceptably high severity and incidence of side effects. In such an event, our trials could be suspended or terminated, and the FDA or comparable foreign regulatory authorities could order us to cease further development of or deny approval of our product candidates for any or all targeted indications. The drug-related side effects could also affect patient recruitment or the ability of enrolled subjects to complete the trial or result in potential product liability claims. Any of these occurrences may have a material adverse effect on our business, results of operations, financial condition, cash flows and future prospects.
Additionally, if any of our product candidates receives regulatory approval, and we or others later identify undesirable side effects caused by such product, a number of potentially significant negative consequences could result, including that:
•we may be forced to suspend marketing of such product;
•regulatory authorities may withdraw their approvals of such product;
•regulatory authorities may require additional warnings on the label that could diminish the usage or otherwise limit the commercial success of such products;product;
•we may be required to conduct post-marketing studies;
•we may be required to change or the health care setting in which the way the product is administered;
•we could be sued and held liable for harm caused to subjects or patients; and
•our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of the particular product candidate, if approved.
Our product development program may not uncover all possible adverse events that patients who take our product candidates may experience. The number of subjects exposed to product candidates and the average exposure time in the clinical development program may be inadequate to detect rare adverse events or chance findings that may only be detected once the product is administered to more patients and for greater periods of time.
Clinical trials by their nature utilize a sample of the potential patient population. However, with a limited number of subjects and limited duration of exposure, we cannot be fully assured that rare and severe side effects of our product candidates will be uncovered. Such rare and severe side effects may only be uncovered with a significantly larger number of patients exposed to our product candidates. If such safety problems occur or are identified after any of our product candidates reaches the market, the FDA may require that we amend the labeling of the product or recall the product, or may even withdraw approval for the product.product, any of which could subject us to substantial product liability claims and related litigation.
Our future success is dependent on the regulatory approval of our product candidates.
Our business is dependent on our ability to obtain regulatory approval for our product candidates in a timely manner. We cannot commercialize product candidates in the United States without first obtaining regulatory approval for the product from the FDA. Similarly, we cannot commercialize product candidates outside of the United States without obtaining regulatory approval from comparable foreign regulatory authorities. Before obtaining regulatory approvals for the commercial sale of any product candidate for a target indication, we must demonstrate with substantial evidence gathered infrom preclinical studies and clinical trials, generally including well-controlled Phase 3clinical trials that the product candidate is safe and effective for use for that target indication and that the manufacturing facilities, processes and controls are adequate with respect to such product candidate.
The time required to obtain approval by the FDA and comparable foreign regulatory authorities is unpredictable but typically takes many years following the commencement of preclinical studies and clinical trials and depends upon numerous factors, including the substantial discretion and available resources of the regulatory authorities. In addition, approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions.
Even if a product candidate were to successfully obtain approval from the FDA and comparable foreign regulatory authorities, any approval might contain significant limitations related to use restrictions for specified age groups, warnings, precautions or contraindications, or may be subject to burdensome post-approval study or risk management requirements. Also, any regulatory approval of our current or future product candidates, once obtained, may be withdrawn.
Our current and future product candidates could fail to receive regulatory approval from the FDA.FDA or comparable foreign regulatory authorities.
We have not obtained regulatory approval for any product candidate and it is possible that our existing product candidates or any future product candidates will not obtain regulatory approval, for many reasons, including:
•disagreement with the regulatory authorities regarding the scope, design or implementation of our clinical trials;
•failure to demonstrate that a product candidate is safe and effective for our proposed indication;
•failure of clinical trials to meet the level of statistical significance required for approval;
•failure to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks;
•disagreement with our interpretation of data from preclinical studies or clinical trials;
•the insufficiency of data collected from clinical trials of our product candidates to support the submission and filing of a BLA, NDA or other submission or to obtain regulatory approval;
•the insufficiency of a single Phase 3 clinical trial of GPS in AML for regulatory approval in that indication;
•failure to obtain approval of our manufacturing processes or facilities of third-party manufacturers with whom we contract for clinical and commercial supplies or our own manufacturing facility; or
•changes in the approval policies or regulations that render our preclinical and clinical data insufficient for approval.
The FDA or a comparable foreign regulatory authority may require more information, including additional preclinical or clinical data to support approval or additional studies, to support a marketing approval decision, which may delay or prevent approval and our commercialization plans, or we may decide to abandon the development program. If we were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited
indications than we request (including failing to approve the most commercially promising indications), may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve a product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate.
If we are unable to obtain regulatory approval for one of our product candidates in one or more jurisdictions, or any approval contains significant limitations, we may not be able to obtain sufficient funding to continue the development of that product or generate revenues attributable to that product candidate.
We currently have Orphan Drug ProductODD designation for certain product candidates,GPS and may seek Orphan Drug Product designationSLS009 for additional product candidates orcertain indications, which might not be received or provide the intended benefit thereof.
Regulatory authorities in some jurisdictions, including the United States and Europe, may designate drugs for relatively small patient populations as Orphan Drug Products. Under the Orphan Drug Act, the FDA may designate a product as an Orphan Drug Product if it is a drug intended to treat a rare disease or condition, which is generally defined as a patient population of fewer than 200,000 individuals annually in the United States. We have received Orphan Drug Product designationsODD from the FDA for GPS in AML, MPM and MM as well as Orphan Medicinal Product designations from the EMA for GPS in AML, MPM and MM. In addition, we have received ODD from the FDA for SLS009 for the treatment of AML and PTCL. Although we have received Orphan Drug Product designationODD for GPS and SLS009, there is no guarantee that any of these indications for GPS or SLS009 will be successfully approved by the FDA or the EMA, that GPS or SLS009 will be commercially successful in the marketplace, or that another product will not be approved for the same indication ahead of our product candidate.
Even if we obtain Orphan Drug Productorphan product exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs can be approved for the same disease or condition. Even after an Orphan Drug Product is approved, the FDA can subsequently approve another drug or biologic for the same disease or condition if the FDA concludes that the later drugproduct is clinically superior in that it is shown to be safer, more effective or makes a major contribution to patient care. In addition, Orphan Drug Productproduct exclusivity may be lost if the FDA or EMA determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug to meet the needs of patients with the rare disease or condition.
We currently have Fast Track designation for certain product candidatesGPS and SLS009 and may seek Fast Track designation for additional product candidates, or indications, which might not be received or provide the intended benefits thereof.
If a product candidate is intended for the treatment of a serious condition and nonclinical or clinical data demonstrate the potential to address unmet medical need for this condition, a product sponsor may apply to the FDA for Fast Track designation, which may or may not be granted by the FDA. The FDA has given usWe have received Fast Track designation from the FDA for GPS in AML, MPM and MM and Fast Track designation for NPSSLS009 for the adjuvant treatment of patients with early stage breast cancer with low to intermediate HER2 expression following standard of care upfront therapy (surgery plus chemotherapy +/- radiotherapy).r/r AML and r/r PTCL.
However, Fast Track designation does not ensure that we will receive marketing approval or that approval will be granted within any particular timeframe. We may not experience a faster development or regulatory review or approval process with Fast Track designation compared to conventional FDA procedures. In addition, the FDA may withdraw Fast Track designation if it believes that the designation is no longer supported by data from our clinical development program. Fast Track designation alone does not guarantee qualification for the FDA’s accelerated approval or priority review procedures.
Failure to obtain regulatory approval in international jurisdictions would prevent our product candidates from being marketed abroad.
In addition to regulations in the United States, to market and sell our product candidates in the European Union,EU, United Kingdom, many Asian countries and other jurisdictions, we must obtain separate regulatory approvals and comply with numerous and varying regulatory requirements. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions or by the FDA. The regulatory
approval process outside the United States generally includes all of the risks associated with obtaining FDA approval as well as risks attributable to the satisfaction of local regulations in foreign jurisdictions. The approval procedure variesprocedures vary among countries and can involve additional nonclinical or clinical testing. The time required to obtain approval may differ substantially from that required to obtain FDA approval. We may not be able to obtain approvals from regulatory authorities outside the United States on a timely basis, if at all. Clinical trials accepted in one country may not be accepted by regulatory authorities in other countries. In addition, many countries outside the United States require that a product be approved for reimbursement before it can be approved for sale in that country. A product candidate that has been approved for sale in a particular country may not receive reimbursement approval in that country, or may receive reimbursement at a level that is not commercially viable.
We may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize our products in any market. If we are unable to obtain approval of any of our current or future product candidates by regulatory authorities in the European Union,EU, United Kingdom, Asia or elsewhere, the commercial prospects of that product candidate may be significantly diminished, our business prospects could decline and this could materially adversely affect our business, results of operations and financial condition.
Even if our current and future product candidates receive regulatory approval, they may still face future development and regulatory difficulties.
Even ifAny regulatory approvals we obtain regulatory approvalreceive for aany of our product candidates may be subject to limitations on the approved indicated uses for which the product candidate thatmay be marketed or to the conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase 4 clinical trials. In addition, any such regulatory approvals would be subject to ongoing requirements by the FDA and comparable foreign regulatory authorities governing the manufacture, quality control, further development, labeling, packaging, storage, distribution, adverse event reporting, safety surveillance, import, export, advertising, promotion, recordkeeping and reporting of safety and other post-marketing information. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance by us and/or our contractCMOs with respect to product quality and manufacturing organizations, or CMOs,operations and our CROs for any post-approval clinical trials that we may conduct. The safety profile of any product will continue to be closely monitored by the FDA and comparable foreign regulatory authorities after approval. If the FDA or comparable foreign regulatory authorities become aware of new safety information after approval of any of our product candidates, they may require labeling changes or establishment of a risk evaluation and mitigation strategy,REMS, impose significant restrictions on a product’s indicated uses or marketing or impose ongoing requirements for potentially costly post-approval studies or post-market surveillance. Any new legislation addressing drug safety issues could result in delays in product development or commercialization, or increased costs to assure compliance. If our original marketing approval for a product candidate was obtained through an accelerated approval pathway, we could be required to conduct a successful post-marketing clinical trial to confirm the clinical benefit for our products. An unsuccessful post-marketing clinical trial or failure to complete such a trial could result in the withdrawal of marketing approval.
In addition, manufacturers of drug products and theirmanufacturers’ facilities are required to continuously comply with FDA and comparable foreign regulatory authority requirements, including ensuring quality control and manufacturing procedures conform to cGMP, regulations and corresponding foreign regulatory manufacturing requirements. Accordingly, we and our contract manufacturers will be subject to continual review and periodic inspections by the FDA and other regulatory authorities forto assess compliance with cGMP cGCP, and adherence to commitments made in any BLA or NDA submission to the FDA or any other regulations.type of domestic or foreign marketing authorization application. We or our third-party manufacturers may not be able to comply with cGMP regulations or similar regulatory requirements outside of the United States. If any of our third-party suppliers fails to comply with cGMP or other applicable manufacturing regulations, our ability to develop and commercialize our product candidates could suffer significant interruptions. If we or a regulatory agency discover previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions on that product, the manufacturing facility or us, including requiring recall or withdrawal of the product from the market or suspension of manufacturing. If we, our product candidates or the manufacturing facilities for our product candidates fail to comply with applicable regulatory requirements, a regulatory agency may:may take the following actions, any of which could significantly and adversely affect supplies of our products:
•issue Form 483 notices of observations, warning letters or untitled letters;
•mandate modifications to promotional materials or require us to provide corrective information to healthcarehealth care practitioners;
•require us to enter into a consent decree, which can include imposition of various fines, reimbursements for inspection costs, required due dates for specific actions and penalties for noncompliance;
•seek an injunction or impose civil or criminal penalties or monetary fines;
•suspend or withdraw regulatory approval;
•suspend any ongoing clinical trials;
•refuse to approve pending applications or supplements to applications filed by us;
•suspend or impose restrictions on operations, including costly new manufacturing requirements; or
•seize or detain products, refuse to permit the import or export of products, or require us to initiate a product recall.
Any government investigation of alleged violations of law would require us to expend significant time and resources in response and could generate adverse publicity. The occurrence of any event or penalty described above may inhibit our ability to successfully commercialize our products and generate revenues.
Advertising and promotion of any product candidate that obtains approval in the United States will be heavily scrutinized by the FDA, the DOJ, the Office of Inspector General offor the U.S. Department of Health and Human Services, or HHS,DHHS, state attorneys general, members of Congress and the public. A company can make only those claims relating to safety and efficacy, purity and potency that are approved by the FDA and in accordance with the provisions of the approved label. Additionally, advertising and promotion of any product candidate that obtains approval outside of the United States will be heavily scrutinized by comparable foreign regulatory authorities. Violations, including actual or alleged promotion of our products for unapproved or off-label uses, are subject to enforcement letters, inquiries and investigations, and civil and criminal sanctions by the FDA, as well as potential prosecution under the federal False Claims Act. Any actual or alleged failure to comply with labeling and promotion requirements may have a negative impact on our business.
Even if we obtain regulatory approval for a product, we will remain subject to ongoing regulatory requirements.
Even if our product candidates are approved, we will be subject to ongoing regulatory requirements with respect to manufacturing, labeling, packaging, storage, advertising, promotion, sampling, record-keeping, conduct of post-marketing clinical trials and submission of safety, efficacy and other post-approval information, including both federal and state requirements in the United States and requirements of comparable foreign regulatory authorities.
Manufacturers and manufacturers’ facilities are required to continuously comply with FDA and comparable foreign regulatory authority requirements, including ensuring quality control and manufacturing procedures conform to cGMP, regulations and corresponding foreign regulatory manufacturing requirements. Accordingly, we and our contract manufacturers will be subject to continual review and inspections to assess compliance with cGMP and adherence to commitments made in any BLA submission to the FDA or any other type of domestic or foreign marketing authorization application.
Any regulatory approvals we receive for any of our product candidates may be subject to limitations on the approved indicated uses for which the product candidate may be marketed or to the conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase 4 clinical trials, and surveillance to monitor the safety and efficacy of the product candidate. We will be required to report adverse reactions and production problems, if any, to the FDA and comparable foreign regulatory authorities. Any new legislation addressing drug safety issues could result in delays in product development or commercialization, or increased costs to assure compliance. If our original marketing approval for a product candidate was obtained through an accelerated approval pathway, we could be required to conduct a successful post-marketing clinical trial to confirm the clinical benefit for our products. An unsuccessful post-marketing clinical trial or failure to complete such a trial could result in the withdrawal of marketing approval.
If a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, or it disagrees with the promotion, marketing or labeling of a product, the regulatory agency may impose restrictions on that product or us, including requiring withdrawal of the product from the market. If we fail to comply with applicable regulatory requirements, a regulatory agency or enforcement authority may, among other things:
•issue warning letters;
•impose civil or criminal penalties;
•suspend or withdraw regulatory approval;
•suspend any of our ongoing clinical trials;
•refuse to approve pending applications or supplements to approved applications submitted by us;
•impose restrictions on our operations, including closing our contract manufacturers' facilities; or
•require a product recall.
Any government investigation of alleged violations of law would require us to expend significant time and resources in response and could generate adverse publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to develop and commercialize our products and the value of our business and our operating results would be adversely affected.
Risks Related to Our Manufacturing
We have limited to no manufacturing sales, marketing or distribution capability and must rely upon third parties for such.
We currently have direct or indirect agreements or arrangements with various third-party manufacturing facilities for production of our product candidates for research and development and testing purposes. For example, for SLS009 we are party to a supply agreement with GenFleet who has agreements with third-party manufacturers for the manufacture of SLS009. We depend on these manufacturers to meet our deadlines, quality standards and specifications. Reliance on third-party providers may expose us to more risk than if we were to manufacture our product candidates ourselves. We do not control the manufacturing processes of the CMOs we rely on and are dependent on those third parties for the production of our product candidates in accordance with relevant applicable regulations, such as cGMP, which includes, among other things, quality control, quality assurance and the maintenance of records and documentation. Our reliance on third parties for the manufacture of our active pharmaceutical ingredientingredients and investigational drug productproducts and, in the future, any approved products, creates a dependency that could severely disrupt our research and development, our clinical testing, and ultimately our sales and marketing efforts if the source of such supply proves to be unreliable or unavailable. If the contracted manufacturing source is unreliable or unavailable, we may not be able to manufacture clinical drug supplies of our product candidates, and our preclinical and clinical testing programs may not be able to move forward and our entire business plan could fail.
The third-party manufacturers we rely on for the manufacture of our product candidates are subject to inspection and approval by regulatory authorities before we can commence the manufacture and sale of any of our product
candidates, and thereafter are subject to ongoing inspection from time to time. Our third-party manufacturers may not be able to comply with applicable cGMP regulations or similar regulatory requirements outside of the United States. In complying with the manufacturing regulations of the FDA and other comparable foreign regulatory authorities, we and our third-party suppliers must spend significant time, money and effort in the areas of design and development, testing, production, record-keeping and quality control to assure that the products meet applicable specifications and other regulatory requirements. If either we or the CMOs we rely on fail to comply with these requirements, our ability to develop and commercialize our product candidates could suffer significant interruptions, and we may be subject to regulatory enforcement action, including the seizure of products and shutting down of production.
Both the active pharmaceutical ingredient and drug product for our product candidates are currently single sourced.single-sourced. We believe these single sources are currently capable of supplying all anticipated needs of our proposed clinical studies, as well as initial commercial introduction.introduction should such product candidates received regulatory approval. If we are able to commercialize our products in the future, there is no assurance that our manufacturers will be able to meet commercialized scale production requirements in a timely manner or in accordance with applicable standards or cGMP. Once the nature and scope of additional indications and their commensurate drug product demands are established, we will seek secondary suppliers of both the active pharmaceutical ingredientingredients and drug productproducts for our product candidates, but we cannot assure that such secondary suppliers will be found on terms acceptable to us, or in a timely manner, or at all.
We are subject to a multitude of manufacturing risks, any of which could substantially increase our costs and limit supply of our product candidates.
We and ourthe CMOs we rely on will need to conduct significant development work for each product candidate for each target indication for studies, trials and commercial launch readiness. We intend to improve the existing processes for GPS in connection with more advanced clinical trials or commercialization efforts we may undertake in the future. Developing commercially viable manufacturing processes is a difficult, expensive and uncertain task, and there are risks associated with scaling to the level required for advanced clinical trials or commercialization, including cost overruns, potential problems with process scale-up, process reproducibility, stability issues, storage issues, consistency and timely availability of reagents or raw materials. The manufacturing facilities in which our product candidates will be made could be adversely affected by the recent coronavirus outbreak, other pandemics, earthquakes and other natural disasters, equipment failures, labor shortages, lack of adequate temperature controls, power failures, and numerous other factors. We currently estimate that we have sufficient clinical supplies to support our clinical trials, for at least the next 12 months, however, this estimate is dependent on patient enrollment rates and a number of other factors and, accordingly, could change. Moreover, current clinical supplies may not be adequate for future clinical studies.
Additionally, the process of manufacturing our product candidates is complex, highly regulated and subject to several risks, including but not limited to:
•product loss due to contamination, equipment failure or improper installation or operation of equipment, or vendor or operator error;
•product loss or manufacturing failure due to failure of temperature controls in production, storage or transit;
•product loss, which may not be covered by insurance, due to global conflict and unrest, including related inoperability of shipping lanes;
•reduced production yields, product defects, and other supply disruptions due to deviations, even minor, from normal manufacturing and distribution processes;
•inability to procure or delay in procuring raw materials and reagents for manufacturing products;
•unexpected product defects;
•microbial, viral, or other contaminations in our product candidates or in the manufacturing facilities in which our product candidates are made, which may result in the closure of such manufacturing facilities for an extended period of time to allow for the investigation and remediation of the contamination;
•adverse impact on the active ingredient of GPS as a result of potential contamination from the presence of heavy metals which can lead to higher than acceptable rates of impurities resulting in the active ingredient being unacceptable for use; and
•adverse impact on the manufacturing of GPS as a result of potential contamination from excess waterhumidity and oxygen which can lead to higher than acceptable levels of impurities resulting in the drug product being unacceptable for use.
Any adverse developments affecting manufacturing operations for our product candidates may result in shipment delays, inventory shortages, lot failures, withdrawals or recalls or other interruptions in the supply of our drug substance and drug product, which could delay the development of our product candidates. We may also have to write off inventory, incur other charges and expenses for supply of drug product that fails to meet specifications, undertake costly remediation efforts, or seek more costly manufacturing alternatives. Inability to meet the demand for our product candidates could damage our reputation and the reputation of our products among physicians, healthcarehealth care payors, patients or the medical community, and cancer treatment centers, which could adversely affect our ability to operate our business and our results of operations.
In theour GPS clinical trials, usingGM-CSF and Montanide are also administered in addition to GPS and NPS, GM-CSF is also administered and itstheir availability is dependent upon a third-party manufacturer,manufacturers, which may or may not reliably provide GM-CSF or Montanide, thus jeopardizing the completion of the trials.
Both GPS and NPS areis administered in combination with GM-CSF, which is available in both liquid and lyophilized forms exclusively from one manufacturer.manufacturer, and Montanide, which is exclusively available from another supplier. We will continue to be dependent on that manufacturerthese manufacturers for our supply of GM-CSFthese materials in connection with the ongoing GPS and NPS trials and the potential commercial manufacture of these programs.GPS. We have not entered into a dedicated supply agreement with the manufacturermanufacturers for GM-CSF or Montanide, and instead rely on purchase orders to meet our supply needs. Any temporary interruptions or discontinuation of the availability of GM-CSF,these materials, or any determination by us to change the GM-CSF usedtheir use with GPS, or NPS, could have a material adverse effect on our clinical trials and any commercialization of the assets. Similarly, for GPS, Montanide is also administered in combination with GM-CSF and GPS. Any temporary interruptions or discontinuation of the availability of Montanide could have a material adverse effect on our clinical trials for GPS and any commercialization of the asset.
If any of our CMOs’the clinical manufacturing facilities of CMOs we rely on for clinical supply are damaged or destroyed or production at such facilities is otherwise interrupted, our business and prospects would be negatively affected.
If our CMOs’the manufacturing facilities of the CMOs we rely on for clinical supply or the equipment in them is damaged or destroyed, we may not be able, to quickly or inexpensively, to replace oursuch manufacturing capacity or replace it at all. In the event of a temporary or protracted loss of thisa facility or equipment, we might not be able to transfer manufacturing to another CMO. Even if we could transfer manufacturing to another CMO, the shift would likely be expensive and time-consuming, particularly because the new facility would need to comply with the necessary regulatory requirements, and we would need FDA approval before selling any products manufactured at that facility. Such an event could delay our clinical trials or reduce our product sales.
Although we currently maintain insurance coverage against damage to our property and to cover business interruption and research and development restoration expenses, our insurance coverage may not reimburse us, or may not be sufficient to reimburse us, for any expenses or losses we may suffer. In addition, our clinical trials insurance coverage has exclusions for global conflict and unrest of the type currently ongoing in Ukraine. We may be unable to meet our requirements for our product candidates if there were a catastrophic event or failure of our current manufacturing facility or processes.
Risks Related to Our Dependence on Third Parties and Our License Agreements
We rely on third parties to conduct our preclinical studies and clinical trials. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, or if we lose any of our CROs or
other key third-party vendors, we may not be able to obtain regulatory approval for or commercialize our current or future product candidates on a timely basis, if at all.
Our internal capacity for clinical trial execution and management is limited and therefore we rely heavily on third parties. We have relied upon and plan to continue to rely upon third-party CROs, vendors and contractors to monitor and manage data for our ongoing preclinical and clinical programs. We currently rely on and plan to continue to rely on a CRO for our Phase 3 trial for GPS in AML and well as all of our ongoing and contemplated clinical studies, with services to be rendered by such CROs and vendors ranging from in the case of assorted Phase 2 trials, specific and need-tailored (e.g., data management and biostatistics) only to, in the case of our immune combination (PD1 blocker) Phase 2 trials and our Phase 3 trial for GPS in AML, all-encompassing. We rely on these parties for the execution of our preclinical studies and clinical trials, including the proper and timely conduct of our clinical trials, and we control only some aspects of their activities. Outsourcing these functions involves risk that third parties may not perform to our standards, may not produce results or data in a timely manner or may fail to perform at all.
While we have agreements governing the commitments of our third-party vendor services, we have limited influence over their actual performance. Nevertheless, we are responsible for ensuring that each of our trials is conducted in accordance with the applicable protocol and legal, regulatory and scientific standards, and our reliance on the CROs does not relieve us of our regulatory responsibilities.
If our company,we or any of our partners or CROs fail to comply with applicable regulations and good clinical practices,GCP, the clinical data generated in our clinical trials may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our regulatory applications. Upon inspection by a given regulatory authority, such regulatory authority could determine that any of our clinical trials are not in compliance with applicable requirements. In addition, our clinical trials must be conducted with product candidates produced under cGMP and other requirements. We are also required to register ongoing clinical trials and post the results of completed clinical trials on a government-sponsored database, clinicaltrials.gov,ClinicalTrials.gov, within a specified timeframe. Failure to comply also would violate federal requirements in the United States and could result in other penalties, which would delay the regulatory approval process and result in adverse publicity.
Our CROs, third-party vendors and contractors are not our employees, and except for remedies available to us under our agreements with such CROs, third-party vendors and contractors, we cannot control whether or not they devote sufficient time and resources, including experienced staff, to our ongoing clinical, nonclinical and preclinical programs. They may also have relationships with other entities, some of which may be our competitors. If CROs, third-party vendors and contractors do not successfully carry out their contractual duties or obligations or meet expected deadlines or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our current or future product candidates. CRO, vendor or contractor errors could cause our results of operations and the commercial prospects for our current or future product candidates to be harmed, our costs to increase and our ability to generate revenues to be delayed.
In addition, the use of third-party service providers requires us to disclose our proprietary information to these parties, which could increase the risk that this information will be misappropriated. We currently have a small number of employees, which limits the internal resources we have available to identify and monitor our third-party providers. To the extent we are unable to identify and successfully manage the performance of third-party service providers in the future, our business may be adversely affected. Though we carefully manage our relationships with our CROs, there can be no assurance that we will not encounter challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition and prospects.
If any of our relationships with our third-party CROs, third-party vendors or contractors terminate, we may not be able to enter into arrangements with alternative CROs, third-party vendors or contractors on a timely basis, on commercially reasonable terms or at all.
Our CROs, third-party vendors and contractors have the right to terminate their agreements with us in the event of an uncured material breach. In addition, some of our CROs, third-party vendors and contractors have an ability to terminate their respective agreements with us if it can be reasonably demonstrated that the safety of the subjects
participating in our clinical trials warrants such termination, if we make a general assignment for the benefit of our creditors or if we are liquidated. Identifying, qualifying and managing performance of third-party service providers can be difficult, time consuming and cause delays in our development programs. In addition, there is a natural transition period when a new CRO, third-party vendor or contractor commences work and the new CRO, third-party vendor or contractor may not provide the same type or level of services as the original provider.
We are dependent on technologies we license, and if we lose the right to license such technologies or we fail to license new technologies in the future, our ability to develop new products would be harmed, and if we fail to meet our obligations under our license agreements, we may lose the ability to develop our product candidates.
We currently are dependent on licenses from third parties for technologies relating to our product candidates. Our current licenses impose, and any future licenses we enter into are likely to impose, various development, funding, royalty, diligence, sublicensing, insurance and other obligations on us. If our license with respect to any of these technologies is terminated for any reason, the development of the products contemplated by the licenses would be delayed, or suspended altogether, while we seek to license similar technology or develop new non-infringing technology. The costs of obtaining new licenses are high. For example, we are entirely dependent on our license from MSK to allow us to develop and commercialize our lead product candidate, GPS, and any loss of or challenge to our license agreement with MSK could have a material and adverse effect on our business and result of operations.
In addition, our business depends on our ability to license therapeutic compounds from third parties. If we fail to meet our obligations under our license agreements, we may lose the ability to develop our product candidates, which would adversely affect our business.
We have in-licensed a significant portion of our intellectual property from MSK. If we breach our license agreement with MSK, we could lose the ability to continue the development and potential commercialization of GPS.
GPS is licensed-inin-licensed from MSK and includes an exclusive license to United StatesU.S. and foreign patent applications. Under the MSK license agreement, we are subject to various obligations, including diligence obligations with respect to funding, development and commercialization activities, payment obligations upon achievement of certain milestones and royalties on product sales, as well as other material obligations. If there is any conflict, dispute, disagreement or issue of nonperformance between us and MSK regarding our rights or obligations under the license agreements, including any such conflict, dispute or disagreement arising from our failure to satisfy diligence or payment obligations under any such agreement, we may be liable to pay damages and MSK may have a right to terminate the affected license. The loss of our license agreement with MSK could materially adversely affect our ability to proceed to utilize the affected intellectual property in our development efforts, our ability to enter into future collaboration, licensing and/or marketing agreements for GPS and our ability to commercialize GPS.
We rely on a license agreement with GenFleet for the development of SLS009, and if this license is breached or otherwise terminated, we could lose the ability to continue the development and potential commercialization of SLS009.
We have entered into a license agreement with GenFleet under which we have an exclusive license to develop and commercialize SLS009 worldwide, other than in mainland China, Hong Kong, Macau and Taiwan. Under the license agreement, we are subject to various obligations, including diligence obligations with respect to development and commercialization activities, payment obligations upon achievement of certain milestones, and royalties on annual net sales (if the product candidate is ultimately commercialized), as well as other material obligations. If there is any conflict, dispute, disagreement, or issue of nonperformance between us and GenFleet regarding our rights or obligations under the license agreement, including any such conflict, dispute, or disagreement arising from our failure to satisfy diligence or payment obligations under the license agreement, we may be liable to pay damages and GenFleet may have a right to terminate the license. The loss of the license agreement could prevent us from developing, commercializing, or entering into future strategic transactions relating to SLS009.
The risks described elsewhere pertaining to our patents and other intellectual property rights also apply to the intellectual property rights that we license, and any failure by us or our licensors to obtain, maintain and enforce these rights could have a material adverse effect on our business.
We In addition, our business depends on our ability to license additional therapeutic compounds from third parties. If we fail to meet our obligations under our current license agreements, we may not be able to identify any suitable acquiror, licensing or collaboration partner for NPS, or if we are able to find such a suitable acquiror or partner,lose the ability to enter into a transaction with such acquiror or partner on favorable terms,licenses for the development of additional product candidates in the future, which will impairwould adversely affect our ability to derive value from the NPS asset.business.
We have determined that we will not engage in further development of NPS. As a result of such determination, the primary path available to derive value from the NPS asset is to find a suitable acquiror, licensing or collaboration partner for the asset. We currently have no agreements or commitments to engage in any specific transactions, and our exploration of various strategic alternatives may not result in any specific action or transaction. To the extent that this effort results in a transaction, our business objectives may change depending upon the nature of the transaction. There can be no assurance that we will enter into any transaction as a result of this effort or that a transaction will be able to be consummated upon favorable terms, including up-front, milestone, royalty and/or license payments as a result of numerous factors, many of which are outside of our control. Furthermore, if we enter into a transaction relating to NPS, we cannot predict the impact that such transaction might have on our stock price. We also cannot predict the impact on our stock price if we fail to enter into such a transaction. If we do enter into a transaction to out-license NPS, there is no assurance that the licensee will be successful in the development and commercialization of the asset as any potential licensee would be subject to many of the same risks associated with clinical development set forth in this “Risk Factors” section.
We may not realize the benefits of our strategic alliances that we may form in the future.
We may form strategic alliances, create joint ventures or collaborations or enter into licensing arrangements with third parties that we believe will complement or augment our existing business, such as our License Agreementlicense agreement with 3D Medicines.Medicines and our license agreement with GenFleet. These relationships, or those like them, may require us to incur nonrecurring and other charges, increase our near- and long-term expenditures, issue securities that dilute our existing stockholders or disrupt our management and business. In addition, we face significant competition in seeking appropriate strategic alliances and the negotiation process is time-consuming and complex. Moreover, we may not be successful in our efforts to establish a strategic alliance or other alternative arrangements for any future product candidates and programs because our research and development pipeline may be insufficient, our product candidates and programs may be deemed to be at too early a stage of development for collaborative effort and third parties may not view our product candidates and programs as having the requisite potential to demonstrate safety and efficacy. If we license products or acquire businesses, we may not be able to realize the benefit of such
transactions if we are unable to successfully integrate them with our existing operations and company culture. We cannot be certain that, following a strategic transaction or license, we will achieve the revenues or specific net income that justifies such transaction. Any delays in entering into new strategic alliances or license agreements related to our product candidates could also delay the development and commercialization of our product candidates and reduce their competitiveness even if they reach the market.
Our business involves the use of hazardous materials and we and our third-party manufacturers and suppliers must comply with environmental, health and safety laws and regulations, which can be expensive and restrict how we do business.
Our third-party manufacturers’manufacturers and suppliers’ activities involve the controlled storage, use and disposal of hazardous materials. We and our manufacturers and suppliers are subject to laws and regulations governing the use, manufacture, storage, handling and disposal of these hazardous materials even after we sell or otherwise dispose of the products. In some cases, these hazardous materials and various wastes resulting from their use will be stored at our contractors or manufacturers’ facilities pending use and disposal. We cannot completely eliminate the risk of contamination, which could cause injury to our employees and others, environmental damage resulting in costly cleanup and liabilities under applicable laws and regulations governing the use, storage, handling and disposal of these materials and specified waste products. Although we expect that the safety procedures utilized by our third-partythird party contractors and manufacturers for handling and disposing of these materials will generally comply with the standards prescribed by these laws and regulations, we cannot guarantee that this will be the case or eliminate the risk of accidental contamination or injury from these materials. In such an event, we may be held liable for any resulting damages and such liability could exceed our resources. We do not currently carry biological or hazardous waste insurance coverage and our property and casualty, and general liability insurance policies specifically exclude coverage for damages and fines arising from biological or hazardous waste exposure or contamination.
We may not be able to establish or maintain the third-party relationships that are necessary to develop or potentially commercialize some or all of our product candidates.candidates, including our relationship with 3D Medicines.
We expect to depend on collaborators, partners, licensees, clinical research organizations and other third parties to support our discovery efforts, to formulate product candidates, to manufacture our product candidates, to conduct clinical trials for some or all of our product candidates and to commercialize our product candidates if approved. For example, in December 2020 we entered into an Exclusive License Agreement with 3D Medicines pursuant to which we granted commercialization rights in Greater China to 3D Medicines. In accordance with the 3D Medicines Agreement and Side Letter, we expected that 3D Medicines would begin enrolling patients in mainland China in the REGAL study in the second half of 2023. To date, no patients have been enrolled in mainland China. However, patients were enrolled in the REGAL study in Taiwan, which is part of the Greater China Territoryterritory, prior to the second half of 2023. Thus, we and 3D Medicines.Medicines are currently engaged in a dispute regarding, among other things, the trigger and payment of the relevant milestone payments due to us as well as 3D Medicines’ failure to use commercially reasonable best efforts to develop GPS in the Greater China territory, and particularly in mainland China. Over the last three to four months of 2023, we attempted to resolve the aforementioned matters in good faith under the dispute resolution provisions of the 3D Medicines Agreement with 3D Medicines but we were unable to reach a resolution. Accordingly, we commenced a binding arbitration proceeding administered by the Hong Kong International Arbitration Centre governed by New York State law as per the 3D Medicines Agreement in December 2023. We are unable at this time to predict with certainty the outcome of the arbitration proceeding, or the timing of the receipt of any milestone payments and other damages it is seeking in the arbitration proceeding, if at all.
Additionally, we cannot guarantee that we will be able to successfully negotiate future agreements for or maintain relationships with collaborators, partners, licensees, clinical investigators, vendors and other third parties on favorable terms, if at all. Our ability to successfully negotiate such agreements will depend on, among other things, potential partners’ evaluation of the superiority of our technology over competing technologies and the quality of the preclinical and clinical data that we have generated, and the perceived risks specific to developing our product candidates. If we are unable to obtain or maintain these agreements, we may not be able to clinically develop, formulate, manufacture, obtain regulatory approvals for or commercialize our product candidates. We cannot necessarily control the amount or timing of resources that our contract partners, including 3D Medicines, will devote to our research and development programs, product candidates or potential product candidates, and we cannot
guarantee that these parties will fulfill their obligations to us under these arrangements in a timely fashion.fashion, if at all. We may not be able to readily terminate any such agreements with contract partners even if such contract partners do not fulfill their obligations to us.
In addition, we may receive notices from third parties from time to time alleging that our technology or product candidates infringe upon the intellectual property rights of those third parties. Any assertion by third parties that our activities or product candidates infringe upon the intellectual property rights of third parties may adversely affect our ability to secure strategic partners or licensees for our technology or product candidates or our ability to secure or maintain manufacturers for our compounds.
Risks Related to Our Intellectual Property
We may not be able to obtain and enforce patent rights or other intellectual property rights that cover our product candidates and that are of sufficient breadth to prevent third parties from competing against us.us.
Our success with respect to our product candidates will depend in part on our ability to obtain and maintain patent protection in the United States and abroad, to preserve our trade secrets, and to prevent third parties from infringing upon our proprietary rights. We seek to protect our proprietary position by filing in the United States and in certain foreign jurisdictions patent applications related to our novel technologies and product candidates that are important to our business. The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. In addition, we may not pursue or obtain patent protection in all major markets. Moreover, in some circumstances, we do not have the right to control the preparation, filing or prosecution of patent applications, or to maintain the patents, covering technology that we license from third parties or covering technology that a collaboration or commercialization partner may develop. In some circumstances, our licensors have the right to prosecute and/or enforce the licensed patents without our involvement or consent, or to decide not to enforce or to allow us to enforce the licensed patents. Therefore, these patents and applications may not be prosecuted and enforced in a manner consistent with the best interests of our business. If any such licensors fail to maintain such patents, or lose rights to those patents, the rights that we have licensed may be reduced or eliminated and our ability to develop and commercialize any of our products that are the subject of such licensed rights could be adversely affected.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. In addition, the laws of foreign jurisdictions may not protect our rights to the same extent as the laws of the United States. For example, European patent law restricts the patentability of methods of treatment of the human body more than U.S. law does. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore, we cannot be certain that we or our licensors were the first to make the inventions claimed in our owned or licensed patents or pending patent applications, or that we or our licensors were the first to file for patent protection of such inventions. Moreover, the U.S. Patent and Trademark Office, or USPTO, might require that the term of a patent issuing from a pending patent application be disclaimed and limited to the term of another patent that is commonly owned or names a common inventor. As a result, the issuance, scope, validity, term, enforceability and commercial value of our patent rights are highly uncertain.
Our pending and future patent applications, and any collaboration or commercialization partner’s pending and future patent applications, may not result in patents being issued which protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive technologies and products.
During prosecution of any patent application, the issuance of any patents based on the application may depend upon our or theirour partners’ ability to generate additional preclinical or clinical data that support the patentability of our proposed claims. We or any collaboration or commercialization partner may not be able to generate sufficient additional data on a timely basis, or at all. Moreover, changes in either the patent laws or interpretation of the patent
laws in the United States or other countries may diminish the value of our or a collaboration or commercialization partner’s patents or narrow the scope of our or their patent protection.
Changes in either the patent laws, regulations, or in the interpretations of patent lawsthereof in the United States or abroad may diminish the value of our intellectual property.
On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to the U.S. patent law. These include provisionsfollowing are potential factors that could affect the way patent applications will be prosecuted and may also affect patent litigation. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation ofscope our business. However, the Leahy-Smith Act, in particular the first-to-file provision and our implementation, could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement of or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents.intellectual property:
In addition, U.S. Supreme Court rulings have narrowedPatent Reform Legislation: The United States Congress periodically considers patent reform legislation aimed at modifying the standards for patentability, patent enforcement procedures, or the rights of patent holders. Such legislative changes could introduce stricter requirements for patent eligibility, limit the scope of patent protection, available in certain circumstances in certain situations. From timeor streamline patent challenges. Any of these changes may weaken the enforceability or value of our existing patents or make it more difficult to time, the U.S. Supreme Court, other federal courts, the U.S. Congress,obtain new patents for our products or interpretation by the USPTO may change the standards of patentability and any such changes could have a negative impact on our business.technologies.
Some cases decided byJudicial Interpretations: Court decisions, particularly those from the U.S.United States Supreme Court have involved questions of when claims reciting abstract ideas, laws of nature, natural phenomena and/or natural products are eligibleand the Federal Circuit, play a crucial role in shaping patent law and practice. Shifts in judicial interpretations, such as alterations to the criteria for a patent, regardless of whether the claimed subject matter is otherwise novel and inventive. These cases include Association for Molecular Pathology v. Myriad Genetics, Inc., 569 U.S. 576 (2013), also known as the Myriad decision; Alice Corp. v. CLS Bank International, 573 U.S. 13-298 (2014), also known as the Alice decision; and Mayo Collaborative Services v. Prometheus Laboratories, Inc., also known as the Prometheus decision, 566 U.S. 66 (2012). The full impact of these decisions is not yet known. In view of these and subsequent court decisions, the USPTO has issued materials to patent examiners providing guidance for determining the patent eligibility of claims reciting laws of nature, natural phenomena, or natural products.
Our current product candidates include products, or components, derived to various extents from nature; therefore, these decisions and their interpretation by the courts and the USPTOstandard for proving patent infringement, may impact prosecution, defense, and enforcement of certain types of patent claims in our patent portfolio. In addition to increasing uncertainty with regard to
International Harmonization: Changes in patent laws or regulations in foreign jurisdictions where we hold or seek patent protection may also impact the value of our intellectual property. Harmonization efforts or shifts in international standards for patentability criteria, enforcement mechanisms, or patent term extensions could affect our ability to obtain future patents, this combinationprotect and monetize our intellectual property in key markets outside the United States.
Patent Regulations and Patent Office Practices: Alterations in patent examination procedures or policies at the United States Patent and Trademark Office, or USPTO, or foreign patent offices could influence the strength and scope of events has created uncertainty with respectour patent rights. Changes in patent office practices related to patent eligibility or patentability standards, or the valuehandling of patents, once obtained. Depending on these and other decisions by U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could changepost-grant proceedings such as inter partes reviews, or be interpreted in unpredictable ways that wouldIPRs, may weaken our ability to obtain some patent claims or to enforce patents that may issue to us in the future. In addition, these eventsMoreover, changes in regulations governing patent rights, such as those related to government-funded programs, may affect our current or future intellectual property rights.
While we continuously monitor developments in patent laws and regulations, there can be no assurance that changes in patent laws, regulations, or interpretations thereof will not adversely affect the value of our ability to defend patents that may issue in procedures in the USPTO or in U.S. courts.intellectual property.
While we intend to take actions reasonably necessary to enforce our patent rights, we may not be able to detect infringement of our own or in-licensed patents, which may be especially difficult for methods of manufacturing or formulation products.
We depend, in part, on our licensors and collaborators to protect a substantial portion of our proprietary rights. In addition, third parties may challenge our in-licensed patents and any of our own patents that we may obtain, which could result in the invalidation or unenforceability of some or all of the relevant patent claims. Litigation or other proceedings to enforce or defend intellectual property rights is very complex, expensive, and may divert our management’s attention from our core business and may result in unfavorable results that could adversely affect our ability to prevent third parties from competing with us.
If another party has reason to assert a substantial new question of patentability against any of our claims in our own and in-licensed patents, the third party can request that the patent claims be reexamined, which may result in a loss of scope of some claims or a loss of the entire patent. In addition to potential infringement suits, and interference and reexamination proceedings, we may become a party to inter partes and post-grant review proceedings in the United States and patent opposition proceedings outside the United States, where either the patentability of the inventions subject of our patents areis challenged, or we are challenging the patents of others. The costs of these proceedings could be substantial, and it is possible that such efforts would be unsuccessful. As the medical device, biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that others may assert our commercial product and/or product candidates infringe their patent rights. If a third-party’s patents were found to
cover our commercial product and product candidates, proprietary technologies, or our uses, we or our collaborators could be enjoined by a court and required to pay damages and could be unable to continue to commercialize our products or use our proprietary technologies unless we or itsuch collaborators obtained a license to the patent. A license may not be available to us or our collaborators on acceptable terms, if at all. In addition, during litigation, the patent holder could obtain a preliminary injunction or other equitable relief, which could prohibit us from making, using or selling our commercial product and product candidates pending a trial on the merits, which could be years away.
Our product candidatesSLS009 may face biosimilargeneric competition sooner than expected afterbefore the expiration of our composition of matter patent protection.
Even if we are successful in achieving regulatory approval to commercialize SLS009, our product candidate may face generic competition. Once an NDA is approved, the product covered thereby becomes a “reference listed drug” in the FDA’s Orange Book. In the United States, manufacturers may seek approval of generic versions of reference listed drugs through submission of an ANDA or approval through a 505(b)(2) NDA. In support of an ANDA, a generic manufacturer need not conduct clinical trials to assess safety and efficacy. Rather, the applicant generally must show that its product has the same active ingredient(s), dosage form, strength, route of administration and conditions of use or labelling as the reference listed drug and that the generic version is bioequivalent to the reference listed drug, meaning it is absorbed in the body at the same rate and to the same extent. Generic products may be significantly less costly to bring to market than the reference listed drug and companies that produce generic products are generally able to offer them at lower prices. Thus, following introduction of a generic drug, a significant percentage of the sales of any branded drug is typically lost to the generic product. In contrast, Section 505(b)(2) enables the applicant to rely, in part, on the FDA’s prior findings of safety and efficacy data for an existing product, or published literature, in support of its application. Section 505(b)(2) provides an alternate path to FDA approval for new or improved formulations or new uses of previously approved products; for example, a follow-on applicant may be seeking approval to market a previously approved drug for new indications or for a new patient population that would require new clinical data to demonstrate safety or effectiveness. Competition that our products could face from follow-on versions of our products could materially and adversely affect our future revenue, profitability and cash flows and substantially limit our ability to obtain a return on the investments we have made in those products. Because SLS009 has not been previously approved as an active ingredient, we expect the Hatch-Waxman Act to provide a five-year period of new chemical entity, or NCE, exclusivity following its approval during which time generic competitors cannot file an ANDA for a generic version of SLS009 or a 505(b)(2) NDA for SLS009, unless the submission contains a Paragraph IV Certification that one or more patents listed in the Orange Book for SLS009 are invalid, unenforceable or will not be infringed by a proposed ANDA product, in which case the submission may be made four years following the original drug approval. If a Paragraph IV Certification is made, the follow-on applicant is required to provide a Paragraph IV Notice Letter advising of the certification. If that occurs, we will have the opportunity to bring a patent infringement action against the follow-on applicant. If such products.a suit is filed within the 45-day period following receipt of the Paragraph IV Notice Letter, the Hatch-Waxman Act provides for a 30-month stay on FDA’s ability to grant final approval of the proposed follow-on product. The 30-month stay generally runs from the date the Paragraph IV Notice Letter is received. However, when a Paragraph IV certification is received during the five-year period of NCE exclusivity following the date of first NDA approval, the 30-month stay extends from five years after the date that product was first approved. The 30-month stay may be shortened or lengthened, including due to a settlement of a lawsuit, a court order (including a decision by the district court on the merits of the case), or patent expiration. The party filing the ANDA or 505(b)(2) NDA may also counterclaim in the litigation that one or more of our patents are invalid, unenforceable, and/or not infringed. If all of the asserted SLS009 patents were found invalid, enforceable, and/or not infringed, a competing generic product could be marketed prior to expiration of those patents, our business could be harmed.
Settlements and related licensing agreements resulting from Hatch-Waxman litigation can be challenged and have the potential to generate additional litigation which can be costly. The success of such litigation depends on the strength of the patents covering our branded products and our ability to prove that the follow-on applicant’s product would infringe one or more such patents. The outcome of such litigation is inherently uncertain and may result in potential loss of market exclusivity for SLS009, which may have a significant financial impact on our business. Furthermore, the Federal Trade Commission, or FTC, has brought successful lawsuits challenging Hatch-Waxman litigation settlements as anti-competitive, and such decisions have been upheld by federal circuit courts. If we engage in Hatch-Waxman litigation, we may also face an FTC challenge with respect to any proposed settlement related to such litigation, which may result in additional expense or penalty. The FTC also has more recently been questioning pharmaceutical company patent listings in the Orange Book and raising concerns about “improper”
listings that may be intended to discourage competition by follow-on drug developers, and certain members of Congress have been investigating similar issues. Accordingly, there could be future changes to federal laws, regulations, or guidelines related to Hatch-Waxman requirements or procedures that could have a material adverse impact on all pharmaceutical innovators, including us.
If the FDA, EMA or other foreign regulatory authorities approve generic or biosimilar versions of any of our product candidates that receive marketing approval, or such authorities do not grant our products appropriate periods of exclusivity before approving generic or biosimilar versions of those products, the sales of our products, if approved, could be adversely affected.
Even if we are successful in achieving regulatory approval to commercialize a biologic product candidate ahead of our competitors, our product candidates may face competition from biosimilar and generic products. Most biological products are licensed for marketing by FDA via a BLA, under authorities in the Public Health Service Act, or PHSA. ToAssuming that we receive positive data from the REGAL trial, we will file a BLA in order to obtain licensure or marketing approvalauthorization for a new biologic, the sponsor (generally, the manufacturer) must demonstrate in the BLA that the biological product, and that the facility in which it is manufactured, processed, packed, or held, meets standards to assure that the product is safe, pure, and potent. As with other FDA-approved products, any subsequent change to the manufacturing process requires a demonstration to FDA of the comparability of the product’s attributes before and after the change to ensure that the safety and effectiveness of the product is maintained.GPS. In 2010, the Biologics Price Competition and Innovation Act, or BPCIA, enacted as Title VII of the ACA, established an abbreviated pathway under the PHSA for licensure of biosimilar biologics (i.e., biosimilars, sometimes referred to as follow-on biologics). A biosimilar is a biological product that is demonstrated to be “highly similar” (i.e., biosimilar), but not identical, to an FDA-licensed biological product (i.e., the reference product).
The BPCIA also establishes periods of exclusivity for a brand-name biologic (the reference product), one with a duration of 4four years and the other with a duration of 12 years. These periods of regulatory exclusivity initiate upon licensure of the new biological product if certain requirements are met. During the four-year exclusivity period, an abbreviated BLA for a biosimilar referencing the protected brand-name biologic may not be submitted to FDA. During the 12-year exclusivity period, approval of an abbreviated BLA for a biosimilar referencing the protected brand-name biologic may not be made effective, which means FDA may not approve the biosimilar application until 12 years after the date on which the reference product was first licensed.
In addition, the BPCIA provides for a process for disclosure and negotiation between the biosimilar applicant and reference product sponsor, sometimes referred to as the “patent dance”.dance.” Although not mandatory on the party of the biosimilar applicant, the dance involves several rounds of informational exchanges concerning potential disputes over the biosimilar applicant’s infringement of the reference product sponsor’s patents. Also, biosimilar licensure under the BPCIA is not contingent upon resolution of patent disputes. Therefore, the FDA may approve a biosimilar despite unresolved patent issues between the reference product sponsor and the biosimilar applicant.
Our composition of matter patents for certain of our product candidates have expired or will expire prior to any product marketing approval. We intend to seek data exclusivity and market exclusivity for GPS, which, like NPS, is expected to be regulated by the FDA as a biological product, provided under the PHSA, and similar laws in other countries. We believe that these product candidatesGPS will qualify for four years of data exclusivity and 12 years of market exclusivity under the BPCIA. The law is complex and continues to be interpreted and implemented by the FDA. As a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when such processes intended to implement BPCIA may be fully adopted by the FDA, any such processes could have a material adverse effect on the future commercial prospects for our product candidates. There is also a risk that the U.S. Congress could amend the BPCIA to shorten the 12-year market exclusivity period or that the FDA will not consider our product candidates to be reference biological products pursuant to its interpretation of the exclusivity provisions of the BPCIA for competing products, potentially creating the opportunity for biosimilar competition sooner than anticipated after the expiration of our patent protection. Moreover, the extent to which a biosimilar, once approved, will be substituted for any reference product in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing. Under the BPCIA as well as state pharmacy laws, only interchangeable biosimilar products are considered substitutable for the reference biological product without the intervention of the health care provider who prescribed the original biological product. However, as with all prescribing decisions made in the context of a patient-provider relationship and a patient’s specific medical needs, health care providers are not restricted from prescribing biosimilar products in an off-label manner.
Even if, as we expect, GPS and NPS areis considered to be reference products eligible for 12 years of exclusivity under the BPCIA, a competitor could decide to forego the abbreviated approval pathway available for biosimilar products and to submit a full BLA for product licensure after completing its own preclinical studies and clinical trials. In such a situation, any exclusivity to which we may be eligible under the BPCIA would not prevent the competitor from marketing its biological product as soon as it is approved.
In Europe, the European Commission has granted marketing authorizations for several biosimilar products pursuant to a set of general and product class-specific guidelines for biosimilar approvals issued over the past few years. In addition, companies may be developing biosimilar products in other countries that could compete with our products, if approved.
The regulatory, or non-patent, exclusivity available to drugs or biologics in some countries is less than that provided by the United States. On January 29, 2020, the United States signed the United States-Mexico-Canada Agreement (USMCA), which is to replace the North American Free Trade Agreement (NAFTA) of 1994. The current version does not include a minimum 10-year market exclusivity periodFor instance, Canada currently provides for biologics that was part of earlier versions of the Agreement. The 10-year period in the USMCA would have increased Canada’s 8-yearan eight-year period of exclusivity for new biological products, and Mexico’s 5-yearMexico provides for a five-year period of exclusivity. Furthermore, in some countries outside of the United States, peptide vaccines, such as GPS, and NPS are regulated as chemical drugs rather than as biologics and may or may not be eligible for non-patent exclusivity.
If competitors are able to obtain marketing approval for biosimilars referencing our therapeutic candidates, if approved, our future products may become subject to competition from such biosimilars, whether or not they are designated as interchangeable, with the attendant competitive pressure and potential adverse consequences. Such competitive products may be able to immediately compete with us in each indication for which our therapeutic candidates may have received approval.
If we are sued for infringing the intellectual property rights of third parties, such litigation could be costly and time-consuming and could prevent or delay our development and commercialization efforts.
Our commercial success depends, in part, on us and our collaborators not infringing the patents and proprietary rights of third parties. There is a substantial amount of litigation and other adversarial proceedings, both within and outside the United States, involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interference or derivation proceedings, oppositions, and inter partes and post-grant review proceedings before the USPTO and non-U.S. patent offices. Numerous U.S. and non-U.S. issued patents and pending patent applications owned by third parties exist in the fields in which we are developing and may develop our current and future product candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, and as our product pipeline grows, the risk increases that our product candidates may be subject to claims of infringement of third parties’ patent rights as it may not always be clear to industry participants, including us, which patents cover various types of products or methods of use. The coverage of patents is subject to interpretation by the courts, and the interpretation is not always uniform or predictable.
If we are sued for patent infringement, we would need to demonstrate that our product candidates, products and methods either do not infringe the patent claims of the relevant patent or that the patent claims are invalid, and we may not be able to do this. Proving that a patent is invalid is difficult. If any issued third-party patents were held by a court of competent jurisdiction to cover aspects of our materials, formulations, methods of manufacture or methods for treatment, we could be forced, including by court order, to cease developing, manufacturing or commercializing the relevant product candidate until such patent expired. Alternatively, we may be required to obtain a license from such third party in order to use the infringing technology and to continue developing, manufacturing or marketing the infringing product candidate. We could be prevented from commercializing a product candidate or be forced to cease some aspect of our business operations, if, as a result of actual or threatened patent infringement claims, we are unable to enter into licenses on acceptable terms. In addition, parties making claims against us may also obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our product candidates.
Defending against claims of patent infringement or misappropriation of trade secrets could be costly and time consuming, regardless of the outcome. Thus, even if we were to ultimately prevail, or to settle at an early stage, such litigation could burden us with substantial unanticipated costs. In addition, litigation or threatened litigation could result in significant demands on the time and attention of our management team, distracting them from the pursuit of other company business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees if we are found to have willfully infringed a patent, or to redesign our infringing product candidates, which may be impossible or require substantial time and monetary expenditure. We may also elect to enter into license agreements in order to settle patent infringement claims prior to litigation, and any such license agreement may require us to pay royalties and other fees that could
be significant. During the course of any patent or other intellectual property litigation, there could be public announcements of the results of hearings, rulings on motions, and other interim proceedings in the litigation. If securities analysts or investors regard these announcements as negative, the perceived value of our product candidates, programs or intellectual property could be diminished. Accordingly, the market price of our shares of common stock may decline.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting, enforcing and defending patents on our current and future product candidates in all countries throughout the world would be prohibitively expensive. We or our licensors’ intellectual property rights in certain countries outside the United States may be less extensive than those in the United States. In addition, the laws of certain foreign countries do not protect intellectual property rights to the same extent as laws in the United States. Consequently, we and our licensors may not be able to prevent third parties from practicing our and our licensors’ inventions in countries outside the United States, or from selling or importing infringing products made using our and our licensors’ inventions in and into the United States or other jurisdictions. Competitors may use our and our licensors’ technologies in jurisdictions where we have not obtained patent protection or where we do not have exclusive rights under the relevant patent(s) to develop their own products and, further, may export otherwise infringing products to territories where we and our licensors have patent protection but where enforcement is not as strong as that in the United States. These infringing products may compete with our product candidates in jurisdictions where we or our licensors have no issued patents or where we do not have exclusive rights under the relevant patent(s), or our patent claims and other intellectual property rights may not be effective or sufficient to prevent them from so competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals,drugs and biologics, which could make it difficult for us and our licensors to stop the infringement of our and our licensors’ patents or marketing of competing products in violation of our and our licensors’ proprietary rights generally. Certain governments outside the United States have indicated that compulsory licenses to patents may be sought to further their domestic policies or on the basis of national emergencies. Proceedings to enforce our and our licensors’ patent rights in foreign jurisdictions could result in substantial costs and divert our attention from other aspects of our business, could put our and our licensors’ patents at risk of being invalidated or interpreted narrowly, could put our and our licensors’ patent applications at risk of not issuing, and could provoke third parties to assert claims against us or our licensors. We or our licensors may not prevail in any lawsuit that we or our licensors initiate, and even if we or our licensors are successful the damages or other remedies awarded, if any, may not be commercially meaningful.
We may become involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time-consuming and unsuccessful and have a material adverse effect on the success of our business and on our stock price.
Third parties may infringe our patents, the patents of our licensors, or misappropriate or otherwise violate our or our licensors’ intellectual property rights. We and our licensors’ patent applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issuesis issued from such applications, and then only to the extent the issued claims cover the technology. In the future, we or our licensors may elect to initiate legal proceedings to enforce or defend our or our licensors’ intellectual property rights, to protect our or our licensors’ trade secrets or to determine the validity or scope of intellectual property rights we own or control. Any claims that we assert against perceived infringers could also provoke these parties to assert counterclaims against us alleging that we infringe their intellectual property rights or that our intellectual property rights are invalid. In addition, third parties may initiate legal proceedings against us or our licensors to challenge the validity or scope of intellectual property rights we own or control. The proceedings can be expensive and time-consuming. Many of our or our licensors’ adversaries in these proceedings may have the ability to dedicate substantially greater resources to prosecuting these legal actions than we or our licensors can. Accordingly, despite our or our licensors’ efforts, we or our licensors may not be able to prevent third parties from infringing upon or misappropriating intellectual property rights we own or control, particularly in countries where the laws may not protect our rights as fully as in the United States. Litigation could result in substantial costs and diversion of
management resources, which could harm our business and financial results. In addition, in an infringement proceeding, a court may decide that a patent owned by or licensed to us is invalid or unenforceable, in whole or in part, or may refuse to stop the other party from using the technology at issue on the grounds that our or our licensors’ patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our or our licensors’ patents at risk of being invalidated, held unenforceable or interpreted narrowly.
Interference or derivation proceedings provoked by third parties, brought by us or our licensors or collaborators, or brought by the USPTO or any non-U.S. patent authority may be necessary to determine the priority of inventions or matters of inventorship with respect to our or our licensors’ patents or patent applications. We may also become involved in other proceedings, such as reexamination, reissue, or opposition proceedings, inter partes review, post-grant review or other pre-issuance or post-grant proceedings in the USPTO or its foreign counterparts relating to our intellectual property or the intellectual property of others. An unfavorable outcome in any such proceeding could require us or our licensors to cease using the related technology and commercializing the affected product candidate, or to attempt to license rights to it from the prevailing party.
Our business could be harmed if the prevailing party does not offer us or our licensors a license on commercially reasonable terms if any license is offered at all. Even if we or our licensors obtain a license, it may be non-exclusive, thereby giving our competitors access to the same technologies licensed to us or our licensors. In addition, if the breadth or strength of protection provided by our or our licensor’s patents and patent applications is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize current and future product candidates. Even if we successfully defend such litigation or proceeding, we may incur substantial costs and it may distract our management and other employees. We could be found liable for monetary damages, including treble damages and attorneys’ fees, if we are found to have willfully infringed a patent.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of shares of our common stock. Furthermore, under Title XI of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or MMA, certain agreements, including patent litigation settlement agreements between brand and generic drug companies, must be filed with the FTC and DOJ. The Patient Right to Know Drug Prices Act amended MMA Title XI, expanding the reporting requirements to include agreements between biosimilar product applicants and biologic companies.
Although we have taken steps to protect our trade secrets and unpatented know-how by entering into confidentiality agreements with third parties and proprietary information and invention agreements with our employees and certain employees, consultants and advisors, third parties may still obtain this information or we may be unable to protect our rights.
Proprietary trade secrets and unpatented know-how are also very important to our business. We also have limited control over the protection of trade secrets used by our licensors, collaborators and suppliers. There can be no assurance that binding agreements will not be breached, that we would have adequate remedies for any breach, or that our trade secrets and unpatented know-how will not otherwise become known or be independently discovered by our competitors. If trade secrets are independently discovered, we would not be able to prevent their use. Enforcing a claim that a third party illegally obtained and is using our trade secrets or unpatented know-how is expensive and time consuming,time-consuming, and the outcome is unpredictable.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed to us alleged trade secrets of their other clients or former employers. As is common in the biotechnology and pharmaceutical industry, certain of our employees were formerly employed by other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Moreover, we engage the services of consultants to assist us in the development of our commercial product and product candidates, many of whom were previously employed at or may have previously been or are currently providing consulting services to, other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may be subject to claims that these employees and consultants or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers or their former or current customers. Litigation may be necessary to defend against these types of claims. Even if we are successful in defending against any such
claims, any such litigation would likely be protracted, expensive, a distraction to our management team, not viewed favorably by investors and other third parties, and may potentially result in an unfavorable outcome.
If we are unable to protect the confidentiality of our trade secrets and other proprietary information, the value of our technology could be materially adversely affected, and our business could be harmed.
In additionProprietary trade secrets and unpatented know-how are also very important to seekingour business. We have limited control over the protection affordedof trade secrets used by patents, weour licensors, collaborators and suppliers. We rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce, and other elements of our technology, discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. Any disclosure to or misappropriation by third parties of our confidential proprietary information could enable competitors to quickly duplicate or surpass our technological achievements, including by enabling them to develop and commercialize products substantially similar to or competitive with our current or future product candidates, thus eroding our competitive position in the market. Trade secrets can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements and invention assignment agreements with our employees, consultants, and outside scientific advisors, contractors and collaborators. These agreements are designed to protect our proprietary information. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, or outside scientific advisors might intentionally or inadvertently disclose our trade secrets or confidential, proprietary information to competitors. In addition, competitors may otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. If any of our confidential proprietary information were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent such competitor from using that technology or information to compete with us, which could harm our competitive position.
Enforcing a claim that a third party illegally obtained and is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, the laws of certain foreign countries do not protect proprietary rights such as trade secrets to the same extent or in the same manner as the laws of the United States. Misappropriation or unauthorized disclosure of our trade secrets to third parties could impair our competitive advantage in the market and could materially adversely affect our business, results of operations and financial condition.
Some intellectual property that we have in-licensed, if created as a result of government funded programs, may be subject to certain federal regulations.
Some of the agreements covering the intellectual property rights we have licensed provide that to the extent that such rights are derived from the use of U.S. government funding, those rights may therefore be subject to certain federal regulations. As a result, the U.S. government may have certain rights to intellectual property embodied in our current or future product candidates pursuant to the Bayh-Dole Act of 1980, or Bayh-Dole Act. These U.S. government rights in certain inventions developed under a government-funded program include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any governmental purpose. In addition, the U.S. government has the right to require us to grant exclusive, partially exclusive or non-exclusive licenses to any of these inventions to a third party if it determines that: (i) adequate steps have not been taken to commercialize the invention, (ii) government action is necessary to meet public health or safety needs or (iii) government action is necessary to meet requirements for public use under federal regulations (also referred to as "march-in rights"). The U.S. government also has the right to take title to these inventions if we, or the applicable licensor, fail to disclose the invention to the government and fail to file an application to register the intellectual property within specified time limits. Intellectual property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require us or the applicable licensor to expend substantial resources. In addition, the U.S. government requires that any products embodying the subject invention or produced through the use of the subject invention be manufactured substantially in the United States. The manufacturing preference requirement can be waived if the owner of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the United States or that under the circumstances domestic manufacture is not commercially feasible. This preference for U.S. manufacturers may limit our ability to contract with non-U.S. product manufacturers for products covered by such intellectual property. To the extent any of our current or future intellectual property is generated through the use of U.S. government funding, the provisions of the Bayh-Dole Act
may similarly apply. Moreover, changes in regulations related to government-funded programs may affect how the government may exercise march-in rights and affect any of our current or future intellectual property rights derived from U.S. government funding.
Risks Related to Competition and Commercialization of Our Current and Future Product Candidates
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
Our future success depends on our ability to demonstrate and maintain a competitive advantage with respect to the design, development, and commercialization of our product candidates. Our competitors may succeed in developing competing products before we do for the same indications we are pursuing, obtaining regulatory approval for products, or gaining acceptance for the same markets that we are targeting. If we are not “first to market” with a product candidate, thereby effecting our order of entry, our competitive position could be compromised via reduced market share and higher hurdles to regulatory approval.
We expect any product candidate which we commercialize will compete with products from other companies in the biotechnology and pharmaceutical industries. There are several biopharmaceutical companies which have approved treatments options in the United States for AML broadly, with different mechanisms of action, including AbbVie/Genentech (Venclexta), Pfizer (Mylotarg), Daiichi-Sankyo (Vanflyta), Rigel Pharmaceuticals (Rezlidhia) and Bristol Myers Squibb (Vidaza).
Key late-stage pipeline agents that are different from GPS have been granted ODD or Fast Track designation due to the unmet need in AML. Therefore, clinical late-stage companies developing late-stage clinical candidates to treat r/r AML may enter the market before our potential products, such as GlycoMimetics (uproleselan), Delta-Fly Pharma (DFP-10917), and AROG Pharmaceuticals (crenolanib). A key competitor may be Iomab-b, which is being developed by Actinium Pharmaceuticals. In late 2022 / early 2023, Actinium announced positive results in its pivotal Phase 3 trial in adults aged 55 and above with active r/r AML.
With respect to WT1-targeting therapies, we do not believe GPS currently has direct competition in AML in the maintenance setting after CR2. There are companies engaged in the clinical development of WT-1 targeting therapies; however, they are not focused on AML at this time, including Astellas (ASP7517) and Cue Biopharma (CUE-102), or are in earlier stages, such as NexImmune (NEXI-001).
With respect to our SLS009 program, we anticipate competition with MEI Pharma, whose voruciclib is in a Phase 1 clinical trial as a monotherapy and in combination with venetoclax for adults with r/r AML. There may also be competition in one of our other indications, PTCL, from enitociclib, a CDK9 inhibitor under development by Vincerx Pharma as a monotherapy and in combination with venetoclax for the treatment of PTCL. Furthermore, there are other companies which are in early development stages for their CDK9 inhibitors and targeting other hematological malignancies or solid tumors, including Kronos Bio (KB-0742), Sumitomo Dainippon Pharma (TP-1287),Adastra Pharmaceuticals (zotiraciclib), and Prelude Therapeutics (PRT2527).
Many of our competitors have substantially greater commercial infrastructures and financial, technical and personnel resources than we have. In addition, some are farther along in their clinical development programs or in collaboration with larger, established pharmaceutical companies. We may not be able to compete unless we successfully:
•design and develop products that are superior to other products in the market;
•conduct successful preclinical and clinical trials;
•attract qualified scientific, medical, sales and marketing and commercial personnel;
•obtain patent and/or other proprietary protection for our processes and product candidates;
•obtain required regulatory approvals; and
•collaborate with others in the design, development, and commercialization of new products.
Established competitors may invest heavily to quickly discover and develop novel compounds that could make our product candidates obsolete. In addition, any new product that competes with an approved product must
demonstrate compelling advantages in efficacy, convenience, tolerability, and safety to overcome price competition and to be commercially successful. If we are not able to compete effectively against our current and future competitors, our business will not grow, and our financial condition and operations will suffer.
If we obtain marketing approval, our commercial success depends on establishing or implementing our own sales, marketing, access and reimbursement, and distribution capabilities or entering into licensing or collaboration agreements for these purposes, and the timing of these.
With the exception of very few employees, including our executive officers, we have not yet built a broader team with any significant sales, marketing, access and reimbursement, or distribution experience. If we progress toward regulatory approval, we will take an efficient and measured approach to building an appropriately sized commercial infrastructure depending on whether we choose to commercialize our current and any future product candidates on our own or through licensing, distribution or collaboration agreements. We will have to invest significant amounts of financial and management resources, some of which will be committed prior to the receipt of positive data if we choose to commercialize on our own. We may need to successfully recruit, retain and train a broad complement of effective commercial staff, including, but not limited to, sales, access and reimbursement, business analytics, and diagnostics. As AML is a highly competitive space, we would expect some personnel to be sought by our competitors. Any delays in hiring an adequate number of experienced sales personnel (including support staff), inability to obtain access to key markets, and unforeseen time, cost and expenses associated with creating a separate and high performing sales and marketing organization could adversely impact commercialization of any product for which we obtain marketing approval.
We may elect to utilize other means of commercialization should the economics of the approach be deemed appropriate. This could include contract sales forces or strategic partners to support in the commercialization of our product candidates. If we enter into arrangements with third parties to perform sales, marketing and distribution services for our products, the resulting revenues or the profitability from these revenues to us are likely to be lower than if we had sold, marketed and distributed our products ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell, market and distribute our product candidates, or do so in a timely manner, or may be unable to do so on terms that are favorable to us. We may also need the infrastructure and resources to maintain the contractual relationships and external support. If we are not able to timely and properly establish a commercial organization on our own or in collaboration with third parties, then we may not be profitable.
Our commercial success depends upon attaining significant market acceptance of our current and future product candidates, if approved, among physicians, patients, healthcarehealth care providers, third-party payors and operators of major cancer treatment centers.clinics.
Even if we obtain regulatory approval for any of our current or future product candidates, the products may not gain market acceptance among physicians, healthcarethird-party payors, patients or the medical community, includingcommunity. For example, current cancer treatment centers. Marketsuch as chemotherapy and radiation therapy are well-established in the medical community, and health care providers may continue to rely on these treatments. The degree of market acceptance of any product candidates for which we receive approval depends on a number of factors, including:
•the efficacy and safety of such product candidates as demonstrated in clinical trials;trials, and acceptance of such by physicians, major cancer treatment centers, and patients;
•the potential and perceived advantages and disadvantages of product candidates over alternative treatments, including the degree of clinically meaningful improvement in care, ease of administration and prevalence and severity of side effects;
•the clinical indications and patient populations for which the product candidate is approved;approved and the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies;
•acceptance by physicians, major cancer treatment centers and patients of the drug as a safe and effective treatment;
•the adoptionability to garner placement of novel immunotherapies by physicians, hospitals and third-party payors;our therapeutics in widely accepted clinical practice treatment guidelines;
•the potentialplacement, or the lack of placement, of our therapeutics in reputable and perceived advantages of product candidates over alternative treatments;highly regarded clinical treatment guidelines;
•the safety of product candidates seen in a broader patient group, including our use outside the approved indications;
•any restrictions on use together with other medications;
•the prevalence and severity of any side effects;
•product labeling or product insert requirements of the FDA or other regulatory authorities;authorities and any restrictions on use with other medications;
•the timing of market introduction of our products as well as competitive products;
•the development of manufacturing and distribution processes for commercial scale manufacturing for our novel WT1 peptide cancer immunotherapy product candidate;
•the cost of treatment in relation to alternative treatments;and coverage and reimbursement status;
•the availability of coveragedevelopment and adequate reimbursement from third-party payors and government authorities;
•relative convenience and ease of administration; and
•the effectiveness of our sales and marketing, manufacturing and distribution efforts for commercial scale; and those
•new healthcare legislation that could adversely affect the payor profile of our collaborators.marketed assets.
If any of our current and future product candidates are approved but fail to achieve market acceptance, among physicians, patients, healthcare payors or cancer treatment centers, we will not be able to generate significant revenues, which would compromise our ability to become profitable.
Even if we are able to commercialize our current or future product candidates, the products may not receive coverage and adequatebecome subject to unfavorable pricing regulations, third-party reimbursement from third-party payors in the United States and in other countries in which we seek to commercialize our products,practices or health care reform initiatives, which could harm our business.
Significant uncertainty exists as to the coverage and reimbursement status of any drug or biological candidates for which we obtain regulatory approval. The regulations that govern marketing approvals, pricing and reimbursement (public and private) for new drug products vary widely from country to country. In the United States, recently passed legislation may significantly change the approval requirements in ways that could involve additional costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we might obtain marketing approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product, possibly for lengthy time periods, and negatively impact the revenues we are able to generate from the sale of the product in that country. Adverse pricing limitations may hinder our ability to recoup our investment in one or more product candidates, even if our product candidates obtain marketing approval.
Our ability to commercialize any product successfully will also depend, in part, on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from third-party payors, including government health administration authorities, private health insurers and other organizations.
Third-party Government authorities and third-party payors, determinesuch as private health insurers and health maintenance organizations, decide which medications they will cover and establish reimbursement levels.how much they will pay. A primary trend in the healthcareU.S. health care industry and elsewhere is cost containment. Third-party payorsGovernment authorities and third-party have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. Third-party payors may also seek additional clinical evidence, beyond the data required to obtain regulatory approval, demonstrating clinical benefit and value in specific patient populations before covering our products for those patients. We cannot be sure that coverage and adequate reimbursement will be available for any product that we commercialize and, if coveragereimbursement is available, what the level of reimbursement will be. Coverage and reimbursementreimbursement. Reimbursement may impact the demand for, or the price of, any product candidate for which we obtain regulatorymarketing approval. Obtaining reimbursement for our products may be particularly difficult because of the higher prices often associated with drugs administered under the supervision of a physician. If reimbursement is not available or is available only atto limited levels, we may not be able to successfully commercialize any product candidate for which we obtain regulatorymarketing approval.
There may be significant delays in obtaining coverage and reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA or comparablesimilar foreign regulatory authorities.authorities outside the United States. Moreover, eligibility for coverage and reimbursement does not imply that any drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient to cover our costs and may only be temporary. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may be based on
reimbursement levels already set for lower cost drugs and may be incorporated into existing payments for other services. Net prices for drugs may be reduced by mandatory discounts or rebates required by third-partygovernment health care programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the United States. No uniform policy for coverage and reimbursement exists in the United States, and coverage and reimbursement can differ significantly from payor to payor. Third-party commercial payors often rely upon Medicarefollow CMS coverage policy and payment limitations in setting their own reimbursement policies, but also have their own methods and approval process apart from Medicare determinations.policies. Our inability to promptly obtain coverage and profitable reimbursementpayment rates from both government-funded and private payors for any approved products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
Further, there have been, and may continue to be, legislative and regulatory proposals at the U.S. federal and state levels and in foreign jurisdictions directed at broadening the availability and containing or lowering the cost of health care. The continuing efforts of the government, insurance companies, managed care organizations and other third-party payors to contain or reduce costs of health care may adversely affect our ability to set prices for our products that would allow us to achieve or sustain profitability. In addition, governments may impose price controls on any of our products that obtain marketing approval, which may adversely affect our future profitability.
More recently, in August 2022, President Biden signed into the law the IRA. Among other things, the IRA has multiple provisions that may impact the prices of drug products that are both sold into the Medicare program and throughout the United States. Starting in 2023, a manufacturer of drugs or biological products covered by Medicare Parts B or D must pay a rebate to the federal government if their drug product’s price increases faster than the rate of inflation. This calculation is made on a drug product-by-drug product basis and the amount of the rebate owed to the federal government is directly dependent on the volume of a drug product that is paid for by Medicare Parts B or D. Additionally, starting for payment year 2026, CMS will negotiate drug prices annually for a select number of single source Part D drugs without generic or biosimilar competition. CMS will also negotiate drug prices for a select number of Part B drugs starting for payment year 2028. If a drug product is selected by CMS for negotiation, it is expected that the revenue generated from such drug will decrease. Additional state and federal health care reform measures are expected to be adopted in the future, any of which could limit the amounts that federal and state governments will pay for health care products and services, which could result in reduced demand for certain biopharmaceutical products or additional pricing pressures.
In some foreign countries, particularly the member states of the EU, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can be a long and expensive process after the receipt of marketing approval for a drug candidate. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various EU member states and parallel distribution, or arbitrage between low-priced and high-priced member states, can further reduce prices. In some countries, we may be required to conduct additional clinical trials that compare the cost-effectiveness of our drug candidates to other available therapies in order to obtain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on prices or reimbursement levels within the country of publication and other countries. If reimbursement of our products is unavailable or limited in scope or amount in a particular country, or if pricing is set at unsatisfactory levels, we may be unable to successfully commercialize and achieve or sustain profitability for sales of any of our drug candidates that are approved for marketing in that country and our business could be adversely affected.
Health care policy changes may have a material adverse effect on our business and results of operations.
Our business may be affected by the efforts of government and third-party payors to contain or reduce the cost of health care through various means. For example, the recently enacted IRA requires drug manufacturers to pay a rebate to the federal government if prices for single-source drugs and biologicals covered under Medicare Part B and nearly all covered drugs under Part D increase faster than the rate of inflation (CPI-U Penalty). The Patient Protection and Affordable Care Act, and the Health Care and Education Affordability Reconciliation Act of 2010, or collectively, the ACA, enacted in March 2010, substantially changed the way health care is financed by both governmental and private insurers, and significantly impacted the pharmaceutical industry, by among other things, increasing the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program;
introducing a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; extending the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care plans; imposing mandatory discounts for certain Medicare Part D beneficiaries as a condition for manufacturers’ outpatient drugs coverage under Medicare Part D; and establishing a Center for Medicare Innovation at the CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending. Regardless of whether or not the ACA is changed or modified by Congress or the U.S. Supreme Court, we expect both government and private health plans to continue to require health care providers, including health care providers that may one day purchase our products, to contain costs and demonstrate the value of the therapies they provide.
Recently enacted and future legislation, including potentially unfavorable pricing regulations, may increase
In addition to the difficulty and cost for usIRA’s drug price negotiation provisions summarized above (see the risk factor herein entitled “Even if we are able to obtain regulatory approval of and commercialize our current or future product candidates, the products may become subject to unfavorable pricing regulations, third-party reimbursement practices or health care reform initiatives, which could harm our business.”), President Biden’s Executive Order 14087, issued in October 2022, called for the CMS Innovation Center to prepare and affectsubmit a report to the prices weWhite House on potential payment and delivery modes that would complement the IRA, lower drug costs, and promote access to innovative drugs. In February 2023, CMS published its report which described three potential models focusing on affordability, accessibility and feasibility of implementation for further testing by the CMS Innovation Center. As of January 2024, the CMS Innovation Center continues to test the proposed models and develop implementation plans. Such models, if implemented, may obtain.have a significant impact on the distribution of and reimbursement for our product candidates that receive FDA approval, if any.
The regulations that govern,Additionally, there has been heightened governmental scrutiny in the United States of pharmaceutical pricing practices considering the rising cost of prescription drugs and biologics. Such scrutiny has resulted in several congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, regulatory approvals, coverage,bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for new drug products vary widely from country to country. In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay regulatory approval of our current or future product candidates, restrict or regulate post-approval activities and affect our ability to successfully sell any product candidates for which we obtain regulatory approval. In the United States, the European Union, United Kingdom and other potentially significant markets for our current and future product candidates, government authorities and third-party payors are increasingly attempting to limit or regulate the price of medical products and services, particularly for new and innovative products and therapies, which has resulted in lower average selling prices. Legislative and regulatory proposals have also been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. For example, onin May 8, 2019, CMS issued a final rule that would have required direct-to-consumer television advertisements of prescriptionto allow Medicare Advantage plans the option to use step therapy for Part B drugs and biological products to include the Wholesale Acquisition Cost, or list price, of the product; however, the U.S. District Court for the District of Columbia invalidated thebeginning January 1, 2020. This final rule preventing it from taking effect. The District Court’s rulingcodified CMS’s policy change that was upheld by the U.S. Court of Appeals for the DC Circuit.effective January 1, 2019. In addition, the 2021 Consolidated Appropriations Act signed into law on December 27, 2020, incorporated extensive health care provisions and amendments to existing laws, including a requirement that all manufacturers of drug products covered under Medicare Part B report the Trump Administration’s Safe Importation Plan, atproduct’s average sales price to CMS beginning on January 1, 2022, subject to enforcement via civil money penalties.
Individual states in the end of December 2019, the FDA issued a notice of proposed rulemaking to establish a system whereby state governmental entities could lawfully import and distribute prescription drugs sourced from Canada, although the impact of such future programs is uncertain. Congress has indicated that it will continue to seek additional new legislative and/or administrative measures to control drug costs. At the state level, legislatures areUnited States have also increasingly passingpassed legislation and implementingimplemented regulations designed to control pharmaceutical and biologicalbiopharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. For example, in recent years, several states have formed PDABs. Much like the IRA’s drug price negotiation program, these PDABs have attempted to implement upper payment limits on drugs sold in their respective states in both public and commercial health plans. For example, in August 2023, Colorado’s PDAB announced a list of five prescription drugs that would undergo an affordability review. The effects of these efforts remain uncertain pending the outcomes of several federal lawsuits challenging state authority to regulate prescription drug payment limits. In December 2020, the U.S. Supreme Court held unanimously that federal law does not preempt the states’ ability to regulate pharmacy benefit managers, or PBMs, and other members of the health care and pharmaceutical supply chain, an important decision that may lead to further and more aggressive efforts by states in this area. In addition, the FTC in mid-2022 also launched sweeping investigations into the practices of the PBM industry that could lead to additional federal and state legislative or regulatory proposals targeting such entities’ operations, pharmacy networks, or financial arrangements. Significant efforts to change the PBM industry as it currently exists in the United States may affect the entire pharmaceutical supply chain and the business of other stakeholders, including biopharmaceutical developers like us.
We expect that additional statethese and federal healthcareother health care reform measures willthat may be adopted in the future any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which couldmay result in reduced demand for our product candidates or additional pricing pressures. We cannot be sure whetherreductions in Medicare and other health care funding, more rigorous coverage criteria, new payment methodologies and additional legislative changes will be enacted, or whether the FDA regulations, guidance or interpretations will be changed, or what the impact of such changesdownward pressure on the regulatory approvals of our product candidates, ifprice that we receive for any may be.
Healthcare legislative measures aimed at reducing healthcare costs may have a material adverse effect on our business and results of operations.
Third-party payors, whether domestic or foreign, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In both the United States and certain international jurisdictions, there have been a number of legislative and regulatory changes to the health care system that could impact our ability to sell our products profitably. In particular, in 2010, the ACA was enacted, which, among other things, subjected biologic products to potential competition by lower-cost biosimilars; addressed a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program; extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care organizations; subjected manufacturers to new annual fees and taxes for certain branded prescription drugs; and provided incentives to programs that increase the federal government’s comparative effectiveness research. Since its enactment, there have been judicial and Congressional challenges to certain aspects of the ACA, as well as recent efforts to repeal or repeal and replace certain aspects of the ACA. On December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, or the Texas District Court Judge, ruled that the individual mandate is a critical and inseverable feature of the ACA, and therefore, because it was repealed as a part of the Tax Act, the remaining provisions of the ACA are invalid as well. In December 2019, the Fifth Circuit Court of Appeals upheld the lower court’s finding that the individual mandate in the law is unconstitutional. However, the Fifth Circuit also reversed and remanded the case to the district court to determine if other reforms enacted as part of the ACA, but not specifically related to the individual mandate or health insurance, including the BPCIA, could be severed from the rest of the ACA so as not to be declared invalid. On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case and allocated one hour for oral arguments, which occurred on November 10, 2020. A decision from the Supreme Court is expected to be issued in spring 2021. It is unclear how this litigation and other efforts to repeal and replace the ACA will affect the implementation of that law. Until there is more certainty concerning the future of the ACA, it will be difficult to predict its full impact and influence on our business.
Further changes to and under the ACA remain possible, although the new administration under President Joseph Biden has signaled that it plans to build on the ACA and expand the number of people who are eligible for subsidies under it. President Biden indicated that he may use executive orders to undo changes to the ACA made by the former administration and would advocate for legislation to build on the ACA. It is unknown what form any such changes or any law proposed to replace the ACA would take, and how or whether it may affect our business in the future. We expect that changes to the ACA, the Medicare and Medicaid programs, changes allowing the federal government to directly negotiate drug prices and changes stemming from other healthcare reform measures, especially with regard to healthcare access, financing or other legislation in individual states, could have a material adverse effect on the healthcare industry.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA was enacted. In August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation’s automatic reduction to several government programs. This includes aggregate reductions of Medicare payments to providers of 2% per fiscal year, which went into effect in 2013, and will remain in effect through 2027 unless additional Congressional action is taken. The American Taxpayer Relief Act of 2012 further reduced Medicare payments to several providers, including hospitals and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign, federal and state levels directed at containing or lowering the cost of healthcare, including facilitation of generic entry and biosimilars.approved product. We cannot predict the initiatives that may be adopted in the future. The continuing efforts of the government, insurance
companies, managed care organizations and other payors of healthcarehealth care services to contain or reduce costs of healthcarehealth care and/or impose price controls may adversely affect:
•the demand for our product candidates, if we obtain regulatory approval;
•our ability to receive or set a price that we believe is fair for our products;
•our ability to generate revenue and achieve or maintain profitability;
•our ability to enjoy or maintain market exclusivity;
•the level of taxes that we are required to pay; and
•the availability of capital.
We expect thatOn January 5, 2024, the ACA, as well as other healthcare reform measures that mayFDA granted approval to Florida for the importation of specific prescription medications from Canada. This initiative aims to provide residents of Florida with access to more affordable drug prices comparable to those paid by Canadians. The noteworthy aspect of this announcement lies in it being the inaugural instance where the FDA has officially sanctioned a state to securely import prescription drugs from an international source. Should this trend of parallel importing grow within the U.S. markets, net revenues could be adopted in the future, may result in additional reductions in Medicare and other healthcare funding, more rigorous coverage criteria, lower reimbursement and new payment methodologies. This could lower the price that we receive for any approved product. Any denial in coverage or reduction in reimbursement from Medicare or other government-funded programs may result in a similar denial or reduction in payments from private payors, which may prevent us from being able to generate sufficient revenue, attain profitability or commercialize our product candidates, if approved.adversely affected.
Price controls may be imposed in foreign markets, which may adversely affect our future profitability.
In some countries, particularly member states of the European Union,EU, the pricing of prescription drugs is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after receipt of regulatory approval for a product. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. ReferenceGlobal reference pricing used by various European Unionmost (if not all) EU member states and parallel distribution, or arbitrage between low-priced and high-priced member states, can further reduce prices. In some countries, we or our collaborators may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our product candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be adversely affected.
Risks Related to HealthcareHealth Care Compliance Regulations
Our relationships with customers and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcarehealth care laws and regulations, which could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings. If we or they are unable to comply with these provisions, we may become subject to civil and criminal investigations and proceedings that could have a material adverse effect on our business, financial condition and prospects.
HealthcareHealth care providers, physicians and third-party payors will play a primary role in the recommendation and prescription of any product candidates for which we obtain regulatory approval. Our current and future arrangements with healthcarehealth care providers, healthcarehealth care entities, third-party payors and customers may expose us to broadly applicable fraud and abuse and other healthcarehealth care laws and regulations that may constrain the business or financial arrangements and relationships through which we research, develop and will market, sell and distribute our products. As a pharmaceuticalbiopharmaceutical company, even though we do not and will not control referrals of healthcarehealth care services or bill directly to Medicare, Medicaid or other third-party payors, federal and state healthcarehealth care laws and regulations pertaining to fraud and abuse and patients’ rights are applicable to our business. Restrictions under applicable federal and state healthcarehealth care laws and regulations that may affect our ability to operate include the following:
•the federal healthcarehealth care Anti-Kickback Statute which prohibits, among other things, individuals and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made, in whole or in part, under a federal healthcarehealth care program such as Medicare or Medicaid;
•federal civil and criminal false claims laws, including the federal False Claims ActFCA that can be enforced through civil whistleblower or qui tam actions, and civil monetary penalty laws, prohibit individuals or entities from knowingly presenting, or causing to be presented, to the federal government, including the Medicare and Medicaid programs, claims for payment or approval that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government;
•the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which imposes criminal and civil liability for executing a scheme to defraud any healthcarehealth care benefit program and also created federal criminal laws that prohibit knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statements in connection with the delivery of or payment for healthcarehealth care benefits, items or services, and further, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, which imposes obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information on entities subject to the law, such as certain healthcarehealth care providers, health plans, and healthcarehealth care clearinghouses, known as covered entities, and their respective business associates that perform services for them that involve the creation, use, maintenance or disclosure of, individually identifiable health information;
•the federal physician sunshine requirements under the ACA which requires certain manufacturers of drugs, devices, biologics and medical supplies, with certain exceptions, to report annually to HHS information related to payments and other transfers of value to physicians, other healthcare providers,certain advanced non-physician health care practitioners, and teaching hospitals, and ownership and investment interests held by physicians and other healthcarehealth care providers and their immediate family members and applicable group purchasing organizations;
•analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcarehealth care items or services reimbursed by non-governmental third-party payors, including private insurers; some state laws which require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government and may require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcarehealth care providers, marketing expenditures or pricing information; and certain state and local laws which require the registration of pharmaceutical sales representatives; and
•state and foreign laws govern the privacy and security of health information in specified circumstances, including the GDPR, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcarehealth care laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcarehealth care laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, disgorgement, exclusion from government funded healthcarehealth care programs, such as Medicare and Medicaid, integrity oversight and reporting obligations, and the curtailment or restructuring of our operations. If any physicians or other healthcarehealth care providers or entities with whom we expect to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcarehealth care programs. Many healthcarehealth care laws and regulations are rapidly changing and legislative bodies and regulatory agencies are regularly considering amendments and supplements to existing laws and regulations, and as a result interpretations of rules and confirmation of our compliance with such rules can be ambiguous.
We may be subject to, or may in the future become subject to, U.S. federal and state, and international laws and regulations imposing obligations on how we collect, use, disclose, store and process personal information. Our actual or perceived failure to comply with such evolving privacy and data protection laws could adversely affect our business, results of operations, and financial condition.
New privacy and data security laws have been proposed in more than half of the states in the United States and in the U.S. Congress, reflecting a trend toward more stringent privacy legislation in the U.S., which trend may accelerate with increasing concerns about individual privacy. The existence of comprehensive privacy laws in different states in the U.S. may make our compliance obligations more complex and costly, may require us to modify our data processing practices and policies, and may require us to incur substantial costs and potential liability in an effort to comply.
In California, the CCPA, which became effective in 2020, broadly defines personal information, gives California residents expanded individual privacy rights and protections, provides for civil penalties for violations, and gives California residents a private right of action for data breaches in certain cases. Further, the California Privacy Rights Act, or the CPRA, which became effective in 2023 and amends the CCPA, imposes additional obligations on covered businesses, including additional consumer rights processes, limitations on data uses, new audit requirements for higher risk data, and opt outs for certain uses of sensitive data. It also created a new California Privacy Protection Agency authorized to issue substantive regulations and is expected to result in increased privacy and information security enforcement. The CPRA also extends the provisions of both the CCPA and the CPRA to the personal information of California-based employees. While there is an exception for certain health information, including protected health information that is subject to HIPAA, and clinical trial data, the CCPA may impact our business activities if we become a “Business” regulated by the CCPA. Further, there continues to be some uncertainly about how certain provisions of the CCPA will be interpreted and how some areas of the law will be enforced. We will continue to monitor developments related to the CCPA and anticipate additional costs and expenses associated with compliance.
In addition to the CCPA, broad consumer privacy laws recently went into effect in Virginia on January 1, 2023, in Colorado and Connecticut on July 1, 2023, and in Utah on December 31, 2023. New privacy laws will also become effective in Florida, Montana, Oregon, and Texas in 2024, in Delaware, Iowa, New Hampshire, New Jersey, and Tennessee in 2025, and in Indiana in 2026. In addition, numerous other states are considering new comprehensive privacy laws.
Other U.S. states, such as New York and Massachusetts have enacted stringent data security laws and numerous other states have proposed similar laws. Additionally, various states, such as California and Massachusetts, have implemented similar privacy laws and regulations, such as the California Confidentiality of Medical Information Act, that impose restrictive requirements regulating the use and disclosure of health information and other personally identifiable information. These laws and regulations are not necessarily preempted by HIPAA, particularly if a state affords greater protection to individuals than HIPAA. Where state laws are more protective, we have to comply with the stricter provisions. In addition to fines and penalties imposed upon violators, some of these state laws also afford private rights of action to individuals who believe their personal information has been misused. California’s patient privacy laws, for example, provide for penalties of up to $250,000 and permit injured parties to sue for damages. Similarly, as discussed above, the CCPA allows consumers a private right of action when certain personal information is subject to unauthorized access and exfiltration, theft or disclosure due to a business’ failure to implement and maintain reasonable security procedures.
Furthermore, over the past few years, the number of privacy-related enforcement actions in the U.S., and in many cases the fines, have steadily increased. Failure to comply with these current and future laws, policies, industry standards, or legal obligations. or any data breach involving personal information, may result in government enforcement actions, litigation, fines, and penalties, private litigation, or adverse publicity, and could cause our customers, business partners, and investors to lose trust in us which could have a material adverse impact on our business, results of our operations, and our financial condition. We continue to face uncertainty as to the exact interpretation of the new requirements on our clinical trials and we may be unsuccessful in implementing all measures required by data protection authorities or courts in interpretation of the new law.
The interplay of federal and state laws may be subject to varying interpretations by courts and government agencies, creating complex compliance issues for us and data we receive, use and share, potentially exposing us to additional expense, adverse publicity and liability. Further, as regulatory focus on privacy issues continues to increase and laws and regulations concerning the protection of personal information expand and become more complex, these potential risks to our business could intensify. Changes in laws or regulations associated with the enhanced protection of certain types of sensitive data, for the treatment of genetic data, along with increased customer demands for enhanced data security infrastructure, could greatly increase our cost of providing our products, decrease demand for our products, reduce our revenues and/or subject us to additional liabilities.
In many activities, including the conduct of clinical trials and our regulatory and commercial operations in the EEA and the United Kingdom, or UK, we are subject to international laws and regulations governing data privacy and the protection of health-related and other personal information. The regulatory framework for collecting, using, safeguarding, sharing, transferring and other processing of information worldwide is rapidly evolving and is likely to remain uncertain for the foreseeable future. The withdrawal of the UK from the EU and the subsequent separation of the data protection regimes of these territories means we are required to comply with separate data protection laws in the EU and the UK, which may lead to additional compliance costs and could increase our overall risk. Similar laws and regulations govern our processing of personal data, including the collection, access, use, analysis, modification, storage, transfer, security breach notification, destruction and disposal of personal data. For example, the collection, use, disclosure, transfer, or other processing of personal data regarding individuals in the EU, including personal health data, is subject to the GDPR, which took effect across all Member States of the EEA on May 25, 2018, and as still in effect in the UK as the UK GDPR. On June 28, 2021, the EU Commission adopted decisions on the UK’s adequacy under the EU GDPR, and the UK continues to operate under this adequacy decision. The GDPR applies to any company established in the EU as well as to those outside the EU that process personal data in connection with the offering of goods or services to individuals in the EU or the monitoring of their behavior. We currently conduct clinical trials and engage in regulatory and commercial operations in the EEA and the UK. As a result, we are subject to privacy laws, including the GDPR and UK GDPR. The GDPR imposes a broad range of data protection obligations on controllers and/or processors, as applicable, that must be complied with when processing personal data subject to the GDPR, including, for example, providing expanded disclosures about how their personal data will be used; higher standards for organizations to demonstrate that they have obtained valid consent or have another legal basis in place to justify their data processing activities; the obligation to appoint data protection officers in certain circumstances; new rights for individuals to be “forgotten” and rights to data portability, as well as enhanced current rights (e.g., access requests); the principal of accountability and demonstrating compliance through policies, procedures, training and audit; limitations on retention of information; mandatory data breach notification requirements; ; safeguards to protect the security and confidentiality of personal data; restrictions on transfers of personal data outside of the EU to third countries deemed to lack adequate privacy protections (such as the U.S.), and onerous new obligations and liabilities on services providers or data processors. .In particular, medical or health data, genetic data and biometric data are all classified as “special category” data under the GDPR and afford greater protection and require additional compliance obligations. Further, the UK and EU member states have a broad right to impose additional conditions—including restrictions—on these data categories. This is because the GDPR allows EU member states to derogate from the requirements of the GDPR mainly in regard to specific processing situations (including special category data and processing for scientific or statistical purposes). Non-compliance with the GDPR may result in monetary penalties of up to €20 million or 4% of worldwide revenue, whichever is greater. Moreover, data subjects can claim damages resulting from infringement of the GDPR. The GDPR further grants non-profit organizations and consumer organizations the right to bring claims on behalf of data subjects. The GDPR and other changes in laws or regulations associated with the enhanced protection of certain types of personal data, such as healthcare data or other sensitive information, could greatly increase our cost of providing our products and services or even prevent us from offering certain services in jurisdictions that we may operate in. The GDPR may increase our responsibility and liability in relation to personal data that we process where such processing is subject to the GDPR, and we may be required to put in place additional mechanisms to ensure compliance with the GDPR, including as implemented by individual EU Member States. Compliance with the GDPR is a rigorous and time-intensive process that may increase our cost of doing business or require us to change our business practices, and despite those efforts, there is a risk that we may be subject to fines and penalties, litigation, and reputational harm in connection with our EU activities.
Further, as referenced above, following the UK’s withdrawal from the EU (i.e., Brexit), and the expiry of the Brexit transition period, which ended on December 31, 2020, the EU GDPR has been implemented in the UK (as the UK
GDPR). The UK GDPR sits alongside the UK Data Protection Act 2018 which implements certain derogations in the EU GDPR into UK law. Under the UK GDPR, companies not established in the UK but who process personal data in relation to the offering of goods or services to individuals in the UK, or to monitor their behavior will be subject to the UK GDPR – the requirements of which are (at this time) largely aligned with those under the EU GDPR and as such, may lead to similar compliance and operational costs. Non-compliance with the UK GDPR may result in monetary penalties of up to £17.5 million or 4% of worldwide revenue, whichever is higher.
In addition, we may be unable to transfer personal data from the EU, UK, and other jurisdictions to U.S or other countries due to limitations on cross-border data flows. In particular, the EEA and the UK have significantly regulated the transfer of personal data to the U.S and other countries whose privacy laws it believes are inadequate. Other jurisdictions may adopt similarly stringent interpretations of their data localization and cross-border data transfer laws. Although there are currently various mechanisms that may be used to transfer personal data from the EEA and the UK to the U.S. in compliance with law, such as the EEA and UK’s standard contractual clauses and the newly-adopted Data Privacy Framework, these mechanisms are subject to legal challenges, and there is no assurance that we can satisfy or rely on these measures to lawfully transfer personal data to the U.S.. If there is no lawful manner for us to transfer personal data from the EEA, the UK or other jurisdictions to the U.S., or if the requirements for a legally-compliant transfer are too onerous, we could face significant adverse consequences, including the interruption or degradation of our operations, the need to relocate part of or all of our business or data processing activities to other jurisdictions at significant expense, increased exposure to regulatory actions, substantial fines and penalties, the inability to transfer data and work with partners, vendors and other third parties, and injunctions against our processing or transferring of personal data necessary to operate our business. Additionally, companies that transfer personal data out of the EEA and the UK to other jurisdictions, particularly to the U.S., are subject to increased scrutiny from regulators, individual litigants, and activist groups.
If we are investigated by an EEA or UK data protection authority, we may face fines and other penalties, which could have a negative effect on our existing business and on our ability to attract and retain new clients or pharmaceutical partners. We may also experience hesitancy, reluctance, or refusal by EEA, UK, or multi-national clients or pharmaceutical partners to continue to use our products due to the potential risk exposure because of the current (and future) data protection obligations imposed on them by certain data protection authorities in interpretation of current law, including the GDPR and UK GDPR. Such clients or pharmaceutical partners may also view any alternative approaches to compliance as being too costly, too burdensome, too legally uncertain, or otherwise objectionable and therefore decide not to do business with us. Any of the foregoing could materially harm our business, prospects, financial condition, and results of operations.
In addition, many jurisdictions outside of the EEA and the UK are also considering and/or enacting comprehensive data protection legislation. For example, as of August 2020, the Brazilian General Data Protection Law imposes stringent requirements similar to GDPR with respect to personal information collected from individuals in Brazil.
In China, there have also been recent significant developments concerning privacy and data security. The Data Security Law of the People’s Republic of China (Data Security Law), which took effect on September 1, 2021, requires data processing (which includes the collection, storage, use, processing, transmission, provision and publication of data), to be conducted in a legitimate and proper manner. The Data Security Law imposes data security and privacy obligations on entities and individuals carrying out data processing activities and also introduces a data classification and hierarchical protection system based on the importance of data in economic and social development and the degree of harm it may cause to national security, public interests, or legitimate rights and interests of individuals or organizations if such data are tampered with, destroyed, leaked, illegally acquired or illegally used. The appropriate level of protection measures is required to be taken for each respective category of data.
Also in China, the Personal Information Protection Law, which took effect on November 1, 2021, introduced stringent protection requirements for processing personal information, which are in many ways akin to the requirements of the GDPR. We may be required to make further significant adjustments to our business practices to comply with the personal information protection laws and regulations in China including the Personal Information Protection Law.
We also continue to see jurisdictions imposing data localization laws. These regulations may interfere with our intended business activities, inhibit our ability to expand into those markets or prohibit us from continuing to offer services in those markets without significant additional costs.
Because the interpretation and application of many domestic and international privacy and data protection laws, commercial frameworks, and standards are uncertain, it is possible that these laws, frameworks, and standards may be interpreted and applied in a manner that is inconsistent with our existing data management practices and policies. It is also possible that by complying with one law, we may be violating another. In addition to the possibility of fines, lawsuits, breach of contract claims, and other claims and penalties, we could be required to fundamentally change our business activities and practices or modify our solutions, which could have an adverse effect on our business. Failure to comply with current and future privacy and data protection laws and regulations could result in government enforcement actions (including the imposition of significant penalties), criminal and civil liability for us and our officers and directors, private litigation and/or adverse publicity that negatively affects our business. Any inability to adequately respond to privacy and security concerns, even if unfounded, or to comply with applicable privacy and data protection laws, regulations, and policies, could result in additional cost and liability to us, damage our reputation, inhibit our ability to conduct trials, and adversely affect our business.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could cause significant liability for us and harm our reputation.
We are exposed to the risk of employee fraud or other misconduct, including intentional failures to comply with FDA regulations or similar regulations of comparable foreign regulatory authorities, provide accurate information to the FDA or comparable foreign regulatory authorities, comply with manufacturing standards we have established, comply with federal and state healthcarehealth care fraud and abuse laws and regulations and similar laws and regulations established and enforced by comparable foreign regulatory authorities, report financial information or data accurately or disclose unauthorized activities to us. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion from government funded healthcarehealth care programs, such as Medicare and Medicaid, and integrity oversight and reporting obligations.
We have been involved in multiple legal and governmental proceedings, including securities class action litigation, and may in the future be involved in proceedings, relating to the commercial activities of our predecessor that could divert management’s attention and adversely affect our financial condition and our business.
Our predecessor, Galena, was involved in multiple legal and governmental proceedings, including stockholder class actions, both state and federal, some of which are ongoing. These legal and governmental actions, or the Galena Legacy Matters, included allegations relating to federal securities law violations, claims under the False Claims Act and Anti-Kickback Statute, claims regarding breaches of contract, and other stockholder allegations, including claims of breaches of fiduciary duty by our former directors, and fentanyl related litigation. Additionally, securities class action or stockholder derivative litigation has become common in our industry following the announcement of negative data or adverse events. We have in the past, and may in the future, become involved in this type of litigation. Litigation often is expensive and diverts management’s attention and resources, which could adversely affect the continuing company’s business.
In December 2015, Galena announced that it had received a subpoena from the U.S. Attorney’s Office for the District of New Jersey, or the USAO NJ, requesting the production of a broad range of documents pertaining to marketing and promotional practices related to Abstral, a fentanyl or synthetic opioid product, that was sold to Sentynl Therapeutics Inc., or Sentynl, in November 2015. In January 2016, Galena announced that the USAO NJ and the DOJ were conducting a criminal and civil investigation of Galena, which came to involve criminal investigations with respect to possibly one or more then-current and/or former employees. On September 8, 2017, the DOJ announced a civil settlement agreement with Galena regarding certain of the marketing and promotional practices at issue in the USAO NJ and DOJ’s investigation. The settlement involved a civil resolution agreement and a civil payment of approximately $7.551 million, plus interest accrued since the date of reaching an agreement in principle in return for a release of federal government claims against Galena in connection with the covered conduct in investigation. The civil payment was fully paid on or about December 29, 2017. The settlement did not include releases of criminal claims by the USAO NJ and DOJ or claims by state agencies or administrative claims HHS but each of these government authorities indicated that they had no present intention to pursue claims in connection with the investigation. A qui tam action had been filed against Galena and others as described in the settlement agreement with DOJ and USAO NJ. As set forth in that settlement agreement, for a release of all claims against Galena and its former officers and directors and dismissal with prejudice of the qui tam lawsuit, the relator received a portion of the $7.551 million payment to the federal government. As a result of the payment of the settlement amount, the federal government and the relator filed a stipulation of dismissal with prejudice as to their claims against Galena in the qui tam lawsuit. In a separate settlement agreement, Galena paid $0.3 million in cash to the relator’s counsel for the statutorily mandated attorney’s fees.
A federal investigation led by the U.S. Attorney’s Office for the Southern District of Alabama, or the SDAL, of two of the high-prescribing physicians for Abstral resulted in the criminal prosecution of the two physicians for alleged violations of the federal False Claims Act and other federal statutes. On April 28, 2016, a second superseding indictment was filed in the criminal case, which added additional information about the defendant physicians and provided information regarding the facts and circumstances involving a rebate agreement between us and the defendant physicians’ pharmacy as well as their ownership of our common stock. The criminal trial, which began on January 4, 2017, concluded with a jury verdict on February 23, 2017 finding these physicians guilty on 19 of 20 counts. In May 2017, one physician was sentenced to 20 years in prison, and the other physician was sentenced to 21 years in prison. At the end of the SDAL case, SDAL dismissed count 18 of the indictment charging that the physicians conspired, through the C&R Pharmacy, to receive illegal kickbacks in exchange for prescribing Abstral. To our knowledge, we were not a target or subject of that investigation.
We received a subpoena from the U.S. Attorney’s Office for the Southern District of New York, or USAO SDNY, in February 2018, seeking documents related to specific prescribing physicians for Abstral (fentanyl) sublingual tablets, who have been subsequently indicted, to which we responded. To our knowledge, we are not a target or subject of that investigation and have had no further interaction with the USAO SDNY with regard to the matter after responding to the subpoena.
There continues to be significant litigation and governmental activity generally in the fentanyl and opioid area, and this activity is expected to continue and may increase in the future. We cannot assure you we will not become subject to additional significant legal or governmental proceedings relating to Galena’s former Abstral business in the future. Moreover, in addition to these ongoing and prior matters, we may be exposed to claims, or other legal or governmental actions in the future relating to violations of the False Claims Act, Anti-Kickback Statute, the ACA, or any other applicable state or federal statutes or regulations, and thereby be subject to penalties, such as civil and criminal penalties, damages, fines, or an administrative action of exclusion from government health care reimbursement programs. Since DOJ published a memorandum in 2016 formally instructing prosecutors to focus on individual accountability when dealing with corporate misconduct, individual prosecutions have increased. As a result, former individual executives or other former employees of Galena could be subjected to prosecution relating to Galena’s former Abstral business in the future.
In February 2017, Galena, and certain of its then officers and directors, were named in a purported consolidated putative class action lawsuit that generally alleges that Galena and certain of its then officers and directors violated Sections 10(b) and/or 20(a) of the Securities Exchange Act of 1934 and Rule 10b-5 promulgated thereunder by failing to disclose, among other things, that certain of Galena’s promotional practices were allegedly improper and that these alleged failures rendered Galena’s statements about its business misleading. This action is currently pending in the U.S. District Court for the District of New Jersey. We deny any and all allegations of wrongdoing and are vigorously defending against this lawsuits. We are, however, unable to predict the outcome of this matter at this time. This lawsuit could adversely impact our reputation and divert management attention and resources away from other priorities. Three related derivative lawsuits were filed with respect to which we have reached a settlement in principle subject to final documentation and court approval.
As a result of a cease and desist order issued by the SEC on April 10, 2017 and our related settlement with the SEC, or the SEC Settlement, we are currently an “ineligible issuer” as the term is defined under Rule 405 promulgated under the Securities Act. This could make it more difficult for us to raise necessary financing in the future. If we fail to comply with the terms of the SEC Settlement in the future, it could have significant additional adverse consequences to us.
These Galena Legacy Matters have required and continue to require our management and board of directors to devote a significant amount of time and resources to defending such claims and addressing such allegations, rather than focusing on executing on our business plans and operations. We may, in the future, become subject to additional legal and governmental actions that will also require us to expend time and resources. The settlement of the Galena Legacy Matters has resulted in substantial payments, some of which have not been fully covered by our insurance policies. We may continue to incur substantial unreimbursed legal fees and other expenses in connection with the Galena Legacy Matters. These ongoing and other future legal and governmental proceedings may not qualify for coverage under, or may exceed the limit of, our applicable directors and officers liability insurance policies and could have a material adverse effect on our financial condition, liquidity, and results of operations. An unfavorable outcome in any of these matters could damage our business and reputation or result in additional claims or proceedings against us. Moreover, in addition to these ongoing and prior matters, we may be exposed to other legal or governmental actions in the future, which could result in the payment of additional amounts and have a material adverse effect on our financial condition and results of operations. We can make no assurances as to the time or resources that will need to be devoted to the Galena Legacy Matters, or any new or future matters or their outcome, or the impact, if any, that these matters or any resulting legal or governmental proceedings may have on our business or financial condition but any further action in respect of any such matter by a governmental agency could have a material adverse effect on our results of operation and our business and prospects. See “Business-Legal Proceedings” for more information regarding our legal and governmental proceedings.
Product liability lawsuits against us could cause us to incur substantial liabilities and to limit commercialization of any products that we may develop.
We face an inherent risk of product liability exposure related to the testing of our current or future product candidates in human clinical trials and will face an even greater risk if we commercially sell any products that we may develop. Product liability claims may be brought against us by subjects enrolled in our clinical trials, patients, healthcarehealth care providers or others using, administering or selling our products. If we cannot successfully defend ourselves against claims that our product candidates or products caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
•decreased demand for any product candidates or products that we may develop;
•termination of clinical trial sites or entire clinical trial programs;
•injury to our reputation and significant negative media attention;
•withdrawal of clinical trial participants;
•significant costs to defend the related litigation;
•substantial monetary awards to trial subjects or patients;
•loss of revenue;
•diversion of management and scientific resources from our business operations; and
•the inability to commercialize any products that we may develop.
We currently hold product liability insurance coverage at a level that we believe is customary for similarly situated companies and adequate to provide us with insurance coverage for foreseeable risks, but which may not be adequate to cover all liabilities that we may incur. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. We intend to expand our insurance coverage for products to include the sale of commercial products if we obtain regulatory approval for our product candidates in development, but we may be unable to obtain commercially reasonable product liability insurance for any products that receive regulatory approval. Large judgments have been awarded in class action lawsuits based on drugs that had unanticipated side effects. A successful product liability claim or series of claims brought against us, particularly if judgments exceed our insurance coverage, could decrease our cash and adversely affect our business. In addition, even in instances where we have insurance coverage, our insurance carriers may deny coverage, which could lead to the inability to recover for certain losses and costly insurance coverage disputes with our carriers.
Risks Related to our Business Operations
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, stockholders could lose confidence in our financial and other public reports, which would harm our business, the trading price of our common stock and our ability to raise additional capital in the future.
Effective internal controls over financial reporting are necessary for us to provide reliable financial reports and, together with adequate disclosure controls and procedures, are designed to prevent fraud. Any failure to implement required new or improved controls, or difficulties encountered in their implementation, could cause us to fail to meet our reporting obligations. Ineffective internal controls could also cause investors to lose confidence in our reported financial information, which could have a negative effect on the trading price of our common stock, and which could impact our ability to raise capital in the future. In addition, any future testing by us conducted in connection with Section 404 of the Sarbanes-Oxley Act of 2002, as amended, (“SOX”),or SOX, or any required subsequent testing by our independent registered public accounting firm, may reveal deficiencies in our internal controls over financial reporting that are deemed to be material weaknesses or that may require prospective or retroactive changes to our consolidated financial statements or identify other areas for further attention or improvement.
We are required, pursuant to Section 404 of SOX, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting as of December 31, 2020.2023. However, our independent registered public accounting firm is not required to attest to the effectiveness of our internal control over financial reporting pursuant to Section 404. Under the supervision and with the participation of our Chief Executive Officer and Vice President Finance and Chief AccountingFinancial Officer, our management conducted an evaluation of the effectiveness of our internal control over financial reporting based on the guidelines in the Internal Control-Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on that evaluation, our management concluded that our internal control over financial reporting was effective as of December 31, 2020.2023. An independent assessment of the effectiveness of our internal controls could detect problems that our management’s assessment might not. Undetected material weaknesses in our internal controls could lead to financial statement restatements and require us to incur the expense of remediation.
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
We face competition from numerous pharmaceutical and biotechnology enterprises, as well as from academic institutions, government agencies and private and public research institutions for our current product candidates. Our commercial opportunities will be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer side effects or are less expensive than any products that we may develop. Competition could result in reduced sales and pricing pressure on our current or future product candidates, if approved, which in turn would reduce our ability to generate meaningful revenues and have a negative impact on our results of operations. In addition, significant delays in the development of our product candidates could allow our competitors to bring products to market before we do and impair our ability to commercialize our product candidates. The biotechnology industry, including the cancer immunotherapy market, is intensely competitive and involves a high degree of risk. We compete with other companies that have far greater experience and financial, research and technical resources than us. Potential competitors in the United States and worldwide are numerous and include pharmaceutical and biotechnology companies, educational institutions and research foundations, many of which have substantially greater capital resources, marketing experience, research and development staffs and facilities than ours. Some of our competitors may develop and commercialize products that compete directly with those incorporating our technology or may introduce products to market earlier than our products or on a more cost-effective basis. In addition, our technology may be subject to competition from other technology or methods developed using techniques other than those developed by traditional biotechnology methods. Our competitors compete with us in recruiting and retaining qualified scientific and management personnel as well as in acquiring technologies complementary to our technology. Our company and our collaborators may face competition with respect to product efficacy and safety, ease of use and adaptability to various modes of administration, acceptance by physicians, the timing and scope of regulatory approvals, availability of resources, reimbursement coverage, price and patent position, including the potentially dominant patent positions of others. An inability to successfully complete our product development or commercializing those product candidates could result in our having limited prospects for establishing market share or generating revenue from our technology.
There are several agents in clinical development in potentially comparable settings to our Phase 3 AML clinical development program for GPS. The most advanced of these products is Onureg (oral azacitidine) under development by Bristol-Myers Squibb, or BMS, which was approved by the FDA in September 2020 for maintenance therapy of AML patients after having achieved first complete remission. There are several of other investigational immunotherapies advancing through Phase 2 and Phase 3 trials for target indications that we believe are also potential target indications for GPS. If these or other therapies are successful in their development, it could negatively impact our ability to enroll our clinical trials and could negatively impact the commercial potential of GPS.
Principal competitors for our AML indication include both companies with currently approved products in AML, such as AbbVie/Genentech (the holders of rights to VENCLEXTA), Agios Pharmaceuticals, Inc./Servier (the holder of U.S. rights to TIBSOVO), Novartis AG (the holder of rights to RYDAPT), Astellas Pharmaceuticals (the holder of rights to XOSPATA), BMS (the holder of rights to VIDAZA and IDHIFA), Otsuka Pharmaceutical Co., Ltd. (the holder of rights to DACOGEN), among others, as well as those with front-line chemotherapy drugs and maintenance therapies such as Jazz Pharmaceuticals, Inc. (the holder of rights to VYXEOS), BMS (the holder of rights to ONUREG),as well as Pfizer Inc. (the holder of rights to MYLOTARG and DAURISMO), among others, as well as companies with drugs currently in development in AML, such Daiichi Sankyo (the holder of rights to quizartinib/licensed in Japan under the trade name VANFLYTA), Karyopharm Therapeutics, Inc. (the holder of rights to XPOVIO), Pfizer Inc./AROG Pharmaceuticals, LLC (the holders of rights to crenolanib), Novartis AG (the holder of rights to MBG453), Johnson & Johnson/Janssen Pharmaceuticals, Inc. (the holder of rights to cusatuzumab, or ARGX-110/JNJ-4550), Gilead Sciences, Inc. (the holder of rights to magrolimab, or Hu5F9 G4), Actinium Pharmaceuticals, Inc. (the holder of rights to [131]-iodine-apamistamab, or Iomab-B), and Rafael Pharmaceuticals, Inc. (the holder of rights to devimistat), among others. Companies currently engaged in the clinical development of AML therapies with an immunological/immuno-modulatory mechanism of action include Pfizer Inc./EMD Serono (the holders of rights to BAVENCIO), BMS (the holder of rights to YERVOY), MacroGenics, Inc./Les Laboratoires Servier, SA (the holders of rights to flotetuzumab, or MGD006), Celyad Oncology SA (the holder of rights to CYAD-01), Fortress Biotech, Inc. (the holder of rights to CNDO-109), Glycostem Therapeutics BV (the holder of rights to oNKord), iCell Gene Therapeutics, LLC (the holder of rights to CLL1-CD33 Compound CAR T-cell), among others. Finally, companies currently engaged in the clinical development of WT1-targeting vaccines (not specifically for AML) include Otsuka Pharmaceutical Co., Ltd. (the holder of rights to OCV-501) and Dainippon Sumitomo Pharma Co., Ltd./ Boston Biomedical, Inc. (the holder of rights to DSP-7888/ade-gramotide/nelatimotide).
We are also conducting clinical development programs in combination with cancer checkpoint inhibitors. This is a highly competitive field, with hundreds of such combination trials with various checkpoint inhibitors ongoing. If one or more of these combinations produce positive results in indications that we believe are targets for GPS (either in combination or in stand-alone administration) this could increase the difficulty for us to conduct our trials and could negatively impact our path to regulatory approval and our ability to successfully commercialize our products.
Many of our competitors or potential competitors have significantly greater established presence in the market, financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do, and as a result may have a competitive advantage over us. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies and technology licenses complementary to our programs or potentially advantageous to our business.
As a result of these factors, these competitors may obtain regulatory approval of their products before we are able to obtain patent protection or other intellectual property rights, which will limit our ability to develop or commercialize our current or future product candidates. Our competitors may also develop drugs that are safer, more effective, more widely used and cheaper than ours, and may also be more successful than us in manufacturing and marketing their products. These appreciable advantages could render our product candidates obsolete or noncompetitive before we can recover the expenses of development and commercialization.
We enter into various contracts in the normal course of our business in which we may be required to indemnify the other party to the contract under certain specific scenarios. In the event we have to perform under these indemnification provisions, it could have a material adverse effect on our business, financial condition and results of operations.
In the normal course of business, we periodically enter into academic, commercial, service, collaboration, licensing, consulting and other agreements that contain indemnification provisions. With respect to our academic and other research agreements, we typically agree to indemnify the institution and related parties from losses arising from claims relating to the products, processes or services made, used, sold or performed pursuant to the agreements for which we have secured licenses, and from claims arising from our or our sublicensees’ exercise of rights under the agreement. With respect to our collaboration agreements, we indemnify our collaborators from any third-party product liability claims that could result from the production, use or consumption of the product, as well as for alleged infringements of any patent or other intellectual property right by a third party. With respect to consultants, we indemnify them from claims arising from the good faith performance of their services.
Should our obligations under an indemnification provision exceed applicable insurance coverage or if we were denied insurance coverage for any claim, our business, financial condition and results of operations could be adversely affected. Similarly, if we are relying on a collaborator to indemnify us and the collaborator is denied insurance coverage for the claim or the indemnification obligation exceeds the applicable insurance coverage, and if the collaborator does not have other assets available to indemnify us, our business, financial condition and results of operations could be adversely affected.
Significant disruptions of information technology systems, computer system failures or breaches of information securitycybersecurity incidents could adversely affect our business.
We rely to a large extent upon sophisticated information technology networks and systems to operate our business. In the ordinary course of business, we collect, store and transmit large amounts of confidential information (including, but not limited to, personal information and intellectual property). We also have outsourced significant elements of our operations to third parties, including significant elements of our information technology infrastructure and, as a result, we are managing many independent vendor relationships withrely on third parties who may or could have access tothat process confidential information on our confidential information.behalf. This includes information technology systems of MSK, our CROs, our CMOs, and other business vendors on which we rely. The size and complexity of our information technology and information security systems, and those of our third-party vendors with whom we contract, (andand the large amounts of confidential information that is present on them),and data stored or processed thereon, make such systems potentially vulnerable to service interruptionscomputer viruses, bugs, worms, or to security breaches from inadvertent or intentional actions by our employees or vendors, or fromother malicious codes, malware, including as a result of advanced persistent threat intrusions, and other attacks by third parties.computer hackers, cracking, application security attacks, social engineering, including through phishing attacks, supply chain attacks and vulnerabilities through our third-party service providers, denial-of-service attacks, such as credential stuffing, credential harvesting, personnel misconduct or error, supply-chain attacks, software bugs, server malfunctions, software or hardware failures, loss of data or other information technology assets, adware, telecommunications failures, earthquakes, fires, floods, and other similar threats. Such attacks are of ever-increasing levels of sophistication and are made by groups, including nation states and organized crime, and individuals with a wide range of motives (including, but not limited to, industrial espionage and market manipulation) and expertise. These threats pose a risk to the security of our systems and networks and the confidentiality, availability and integrity of our data. While we have invested significantly in the protection of data and information technology, there can be no assurance that we will be able to detect any such disruption or security breachcybersecurity incident in a timely manner or at all, or that our efforts will prevent service interruptions or security breaches.cybersecurity incidents.
Our internal computer systems, and those of MSK,While we have implemented security measures, there can be no assurances that a cybersecurity incident or any other previously identified threats will not occur. Any such event could have a material adverse effect upon our CROs, our CMOs, and otherreputation, business, vendors on which we rely, are vulnerable to damage from computer viruses, unauthorized access, natural disasters, fire, terrorism, war and telecommunication and electrical failures. We exercise littleoperations, or no control over these third parties, which increases our vulnerability to problems with their systems. Iffinancial condition. For example, if such an event were to occur and cause interruptions in our operations or those of our third-parties on which we rely, it could result in a material disruption of our drug development programs.programs and the development of our services and technologies could be delayed. Any such interruption or breach in our systems could adversely affect our business operations and/or result in the loss of critical or sensitive confidential information or intellectual property, and could result in financial, legal, business and reputational harm to us or allow third parties to gain material, inside information that they use to trade in our securities. For example, the loss of clinical trial data from completed or ongoing clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breachcybersecurity incident results in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability, the further development of our current and future product candidates could be delayed and our business could be otherwise adversely affected.
The costs related to significant cybersecurity incidents or disruptions could be material and could exceed the limits of the cybersecurity insurance we maintain against such risks. If the information technology systems of our third-party vendors and other contractors and consultants become subject to disruptions or cybersecurity incidents, we may have insufficient recourse against such third parties and may have to expend significant resources to mitigate the impact of such an event, and to develop and implement protections to prevent future events of this nature from occurring.
Furthermore, significant disruptions of our internal information technology systems or those of our third-party vendors and other contractors and consultants, or cybersecurity incidents could result in the loss, misappropriation, and/or unauthorized access, use, or disclosure of, or the prevention of access to, confidential information (including trade secrets or other intellectual property, proprietary business information, and personal information), which could result in financial, legal, business, and reputational harm to us. For instance, any such event that leads to unauthorized access to, or use, acquisition, or disclosure of personal information, including personal information regarding our customers, employees or other individuals, could harm our reputation, subject us to liability under and require us to comply with federal and/or state breach notification laws and international law equivalents, domestic or international privacy, data protection, and data security laws such as HIPAA and HITECH, subject us to mandatory corrective action, and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information. Such incidents could also result in legal claims or proceedings. Certain cybersecurity incidents require notice to the affected individuals, contractual partners, regulatory authorities such as the Secretary of HHS and attorneys general, and in some cases, require notice to the media. Such notice could harm our reputation and our ability to compete, result in significant legal and financial exposure and reputational damages, and loss of confidence in us, all of which could potentially have an adverse effect on our business.
Although we have implemented security measures, there is no guarantee we can protect our data from cybersecurity incidents. Cybersecurity incidents could also damage our reputation or disrupt our operations, including our ability to conduct our analyses, conduct research and development activities, collect, process and prepare company financial information, and manage the administrative aspects of our business.
Penalties for violations of these laws vary. For instance, penalties for failure to comply with a requirement of HIPAA and HITECH vary significantly, and include significant civil monetary penalties and, in certain circumstances, criminal penalties with fines up to $250,000 per violation and/or imprisonment. A person who knowingly obtains or discloses individually identifiable health information in violation of HIPAA may face a criminal penalty of up to $50,000 and up to one-year imprisonment. The criminal penalties increase if the wrongful conduct involves false pretenses or the intent to sell, transfer or use identifiable health information for commercial advantage, personal gain or malicious harm.
Artificial intelligence presents risks and challenges that can impact our business including by posing security risks to our confidential information, proprietary information, and personal data.
Issues in the development and use of artificial intelligence, combined with an uncertain regulatory environment, may result in reputational harm, liability, or other adverse consequences to our business operations. As with many technological innovations, artificial intelligence presents risks and challenges that could impact our business. Our vendors may incorporate generative artificial intelligence tools into their offerings without disclosing this use to us, and the providers of these generative artificial intelligence tools may not meet existing or rapidly evolving regulatory or industry standards with respect to privacy and data protection and may inhibit our or our vendors’ ability to maintain an adequate level of service and experience. If we, our vendors, or our third-party partners experience an actual or perceived breach or privacy or security incident because of the use of generative artificial intelligence, we may lose valuable intellectual property and confidential information and our reputation and the public perception of the effectiveness of our security measures could be harmed. Further, bad actors around the world use increasingly sophisticated methods, including the use of artificial intelligence, to engage in illegal activities involving the theft and misuse of personal information, confidential information, and intellectual property. Any of these outcomes could damage our reputation, result in the loss of valuable property and information, and adversely impact our business.
A pandemic, epidemic, or outbreak of an infectious disease, such as the COVID-19 pandemic, could materially and adversely affect our business.
Public health crises such as pandemics or similar outbreaks could adversely impact our business. Previously, our clinical trial operations were directly and indirectly adversely impacted by the COVID-19 pandemic. A new pandemic or a resurgence of the COVID-19 pandemic could have adverse economic impacts to us.
We will likely need to grow the size of our organization in the future, and we may experience difficulties in managing this growth.
As of December 31, 2020,March 1, 2024, we had seven16 full-time employees. As our development and commercialization plans and strategies continue to develop, our need for additional managerial, operational, manufacturing, regulatory, sales, marketing, financial and other resources may increase. We will need to grow the size of our organization in order to support our continued development and potential commercialization of our product candidates. Ascandidates to complement our developmentmanagement and commercialization plans and strategies continue to develop, our need for additional managerial, operational, manufacturing, sales, marketing, financial and other resources may increase. Our management, personnel and systemsemployees currently in place may not be adequateand to support thisour future growth. Future growth would impose significant added responsibilities on members of management, including:
•managing our clinical trials effectively;
•identifying, recruiting, maintaining, motivating, integrating and integratingretaining additional employees;
•managing our internal development efforts effectively while complying with our contractual obligations to licensors, licensees, contractors and other third parties;
•improving our managerial, development, operational, information technology, human resources and finance systems; and
•expanding our facilities.
If our operations expand, we will also need to manage additional relationships with various strategic partners, suppliers and other third parties. Our future financial performance and our ability to commercialize our product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively, as well as our ability to develop a sales and marketing force when appropriate for our company. To that end, we must be able to manage our development efforts and preclinical studies and clinical trials effectively and hire, train and integrate additional management, research and development, manufacturing, administrative and sales and marketing personnel. The failure to accomplish any of these tasks could prevent us from successfully growing our company.
The requirements of being a public company may strain our resources, divert management’s attention and affect our ability to attract and retain qualified board members.
As a public company, we are subject to the reporting requirements of the Exchange Act, the Sarbanes-Oxley Act, the Dodd-Frank Act, the listing requirements of Nasdaq and other applicable securities rules and regulations. Compliance with these rules and regulations has increased, and will likely continue to increase, our legal and financial compliance costs, make some activities more difficult, time-consuming or costly, and place significant strain on our personnel, systems and resources. In addition, changing laws, regulations and standards relating to corporate governance and public disclosure are creating uncertainty for public companies, increasing legal and financial compliance costs and making some activities more time consuming. These laws, regulations and standards are subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time. This could result in continuing uncertainty regarding compliance matters, higher administrative expenses and a diversion of management’s time and attention. Further, if our compliance efforts differ from the activities intended by regulatory or governing bodies due to ambiguities related to practice, regulatory authorities may initiate legal proceedings against us and our business may be harmed. Being a public company that is subject to these rules and regulations also makes it more expensive for us to obtain and retain director and officer liability insurance, and we may in the future be required to accept reduced coverage or incur substantially higher costs to obtain or retain adequate coverage. These factors could also make it more difficult for us to attract and retain qualified members of our board of directors and qualified executive officers.
Our future success depends on our ability to retain our executive officers and to attract, retain and motivate qualified personnel.
We are highly dependent upon our personnel, including Dr. Angelos M. Stergiou (M.D.M.D., Sc.D. h.c.), our President and Chief Executive Officer, and member of our board of directors. Our employment agreement with Dr. Stergiou does not prevent him from terminating his employment with us at any time. The loss of Dr. Stergiou’s services could impede the achievement of our research, development and commercialization objectives. We have not obtained, do not own, nor are we the beneficiary of, key-person life insurance.
Governance changes, becoming subject to enhanced regulatory requirements We employ our executive officers, other than Dr. Stergiou, on an at-will basis and increased responsibilities associated with becoming a public company may influence our management personnel and our employees to terminate their employment with us. To enhance our ability to retain our executive management personnel, we have entered into retention agreements with certain executive officerscan be terminated by them or us at any time, for any reason and may find it beneficial to enter into additional retention agreements with other key personnel in the future, potentially increasing payroll and operating expenses.
Our future growth and success depend on our ability to recruit, retain, manage and motivate our employees.without notice. The loss of any member of our senior management team or the inability to hire or retain experienced senior management personnel could compromise our ability to execute our business plan and harm our operating results. Because
In order to retain valuable employees at our company, in addition to salary and discretionary non-equity incentive plan compensation, we provide stock options and restricted stock units, or RSUs, that vest over time. The value to our employees of the specialized scientificstock options and managerial nature ofRSUs could be significantly affected by movements in our business, we rely heavilystock price that are beyond our control and may at any time be insufficient to counteract offers from other companies.
Our future growth and success depend not only on our ability to retain, manage and motivate our employees but also on our ability to recruit new employees which is key to our growth. We might not be able to attract andor retain qualified scientific, technicalmanagement and managerial personnel. Theother key personnel in the future due to the intense competition for qualified talent among biotechnology, pharmaceutical and other businesses. We could have difficulty attracting experienced personnel to our company and may be required to expend significant financial resources in our employment recruitment and retention efforts. Many pharmaceutical and biotechnology companies with whom we compete for qualified personnel have greater financial and other resources, different risk profiles and longer histories in the pharmaceutical field is intense and as a result,industry than we may be unable to continue to attract and retain qualified personnel necessary for the development of our business.do.
If we and our third-party manufacturers fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
We and our third-party manufacturers are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including chemicals and biological materials. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from us or our third-party manufacturers’ use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties.
Although we maintain workers’ compensation insurance to cover the costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials with a policy limit that we believe is customary for similarly situated companies and adequate to provide us with insurance coverage for foreseeable risks, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological or hazardous materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penaltiesLegislation or other sanctions, which could adversely affect our business, financial condition, results of operations and prospects.
The Tax Cuts and Jobs Actchanges in U.S. tax law could adversely affect our business and financial condition.
H.R. 1, “An ActThe rules dealing with U.S. federal, state and local income taxation are constantly under review by persons involved in the legislative process and by the Internal Revenue Service and the U.S. Treasury Department. Changes to provide for reconciliation pursuanttax laws (which changes may have retroactive application) could adversely affect us or holders of our common stock. In recent years, many changes have been made to title IIapplicable tax laws and V ofchanges are likely to continue to occur in the concurrent resolution on the budget for fiscal year 2018,”future.
For example, legislation enacted in 2017 informally entitledtitled, the Tax Cuts and Jobs Act, or the Tax Act, enacted on December 22, 2017, among other things, containsTCJA, made significant changes to corporate taxation, including the reduction of the corporate tax rate from a top marginal rate of 35% to a singleflat rate of 21%, the limitation of the tax deduction for net interest expense to 30% of adjusted taxable income (except for certain small businesses), the limitation of the deduction for net operating losses carried forward from taxable years beginning after December 31, 2017 to 80% of current year taxable income and the elimination of net operating loss carrybacks one time taxationgenerated in taxable years ending after December 31, 2017 (though any such net operating losses may be carried forward indefinitely) and the modification or repeal of offshore earnings at reduced rates regardless of whether they are repatriated, elimination of U.S. tax on foreign earnings (subject to certain important exceptions), providing immediate deductions for certain new investments instead of deductions for depreciation expense over time, and modifying or repealing many business deductions and credits (including reduction of tax credits under the Orphan Drug Act). Notwithstanding the reduction in the corporate income tax rate, the overall impact of the Tax Act is uncertain and our business and financial condition could be adversely affected.credits. In addition, it is uncertain if and to what extent various states will conform to the Tax Act.
Notwithstanding the reduction in the corporate income tax rate, the overall impact of the Tax Act remains uncertain and our business and financial condition could be adversely affected. In addition, as part of Congress’ response to the COVID-19 pandemic, the Families First Coronavirus Response Act, or the FFCR Act, was enacted on March 18,27, 2020, former President Trump signed into law the Coronavirus“Coronavirus Aid, Relief, and Economic Security Act,Act” or the CARES Act, was enacted on March 27, 2020, and COVID relief provisions werewhich included certain changes in tax law intended to stimulate the Consolidated Appropriations Act, 2021 or CAA, which was enacted on December 27, 2020. All contain numerous tax provisions. Regulatory guidance under the Tax Act, the FFCR Act, the CARES Act, and the CAA is and continues to be forthcoming, and such guidance could ultimately increase or lessen their impact on our business and financial condition. It is also likely that Congress will enact additional legislationU.S. economy in connection withlight of the COVID-19 pandemic,public health emergency, including providing temporary relief from certain aspects of the TCJA that had imposed limitations on the utilization of certain losses, interest expense deductions, and as aminimum tax credits and provided temporary deferral of certain payroll taxes.
It cannot be predicted whether, when, in what form or with what effective dates new tax laws may be enacted, or regulations and rulings may be enacted, promulgated or issued under existing or new tax laws, which could result of thein an increase in our or our shareholders’ tax liability or require changes in the U.S. presidential administration and controlmanner in which we operate in order to minimize or mitigate any adverse effects of changes in tax law or in the U.S. Senate, additional tax legislation may also be enacted; any such additional legislation could have an impact on us. In addition, it is uncertain if and to what extent various states will conform to the TCJA, the FFCR Act, the CARES Act, or the CAA.interpretation thereof.
Our ability to use net operating losses to offset future taxable income may be subject to limitations.
As of December 31, 2020,2023, we had federal and state net operating loss carryforwards of approximately $33.7$57.1 million and $1.2$3.6 million, respectively. Our NOLs generated in tax years ending on or prior to December 31, 2017 are only permitted to be carried forward for 20 years under applicable U.S. tax laws, and will begin to expire, if not utilized, beginning in 2027. These NOL carryforwards could expire unused and be unavailable to offset future income tax liabilities. Under the Tax Act, federal NOLs incurred in tax years ending after December 31, 2017 may be carried forward indefinitely, but the deductibility of such federal NOLs is limited. It is uncertain if and to what extent various states will conform to the Tax Act, or whether any further regulatory changes may be adopted in the future that could minimize its applicability. In addition, under Section 382 of the Internal Revenue Code of 1986, as amended, and certain corresponding provisions of state law, if a corporation undergoes an “ownership change,” which is generally defined as a greater than 50% change, by value, in the ownership of its equity over a three-year period, the corporation’s ability to use its pre-change NOL carryforwards and other pre-change tax attributes to offset its post-change income may be limited. The Merger constituted an ownership change and as such, our ability to use our NOL carryforwards is materially limited, which may harm our future operating results by effectively increasing our future tax obligations.
Risks RelatingRelated to Ownership of Our Common Stock
We will likely need to secure additional capital which may cause dilution to you and our existing stockholders, provide subsequent investors with rights and preference that are senior to yours, restrict our operations or require us to relinquish rights to our product candidates on unfavorable terms to us.
We will likely need to raise additional capital in the future. If we raise funds through the issuance of debt or equity, any debt securities or preferred stock issued will have rights, preferences and privileges senior to those of holders of our common stock in the event of a liquidation. In such event, there is a possibility that once all senior claims are settled, there may be no assets remaining to pay out to the holders of common stock. In addition, if we raise funds through the issuance of additional equity, whether through private placements or additional public offerings, such an issuance would dilute our stockholders and, similar to some of our past financings, may contain terms that could result in additional further significant dilution in the future. Debt financing, if available, could include covenants limiting or restricting our ability to take certain actions, such as incurring additional debt, making capital expenditures, entering into licensing arrangements, or declaring dividends, and may require us to grant security interests in our assets, including our intellectual property and for our subsidiaries to guarantee our obligations.
The market price and trading volume of shares of our common stock may be volatile.
The market price of shares of our common stock has exhibited substantial volatility. Between January 2, 20203, 2023 and December 31, 2020,29, 2023, the daily closing price of shares of our common stock as reported on Nasdaq ranged from a low of $1.53$0.88 to a high of $17.66.$3.86. The market price of shares of our common stock could continue to fluctuate significantly for many reasons, including the following factors:
•reports of the results of our clinical trials regarding the safety or efficacy of our product candidates and surrogate markers;
•announcements of regulatory developments or technological innovations by us or our competitors;
•announcements of business or strategic transactions or our success in finalizing such a transaction;
•announcements of legal or regulatory actions against us or any adverse outcome of any such actions;
•changes in our relationships with our licensors, licensees and other strategic partners;
•low volume in the number of shares of our common stock traded on Nasdaq;
•our quarterly operating results;
•announcements of dilutive financing;
•announcements of additional potential reverse stock split;
•developments in patent or other technology ownership rights;
•additional funds may not be available on terms that are favorable to us and, in the case of equity financings, may result in dilution to our stockholders;
•government regulation of drug pricing; and
•general changes in the economy, the financial marketsmarket conditions and other factors unrelated to our operating performance or the pharmaceutical or biotechnology industries.operating performance of our competitors, including deteriorating market conditions due to investor concerns regarding inflation and hostilities between Russia and Ukraine and Israel and Hamas.
Factors beyond our control may also have an impact on the market price of shares of our common stock. For example, to the extent that other companies within our industry experience declines in their stock prices, the market price of shares of our common stock may decline as well.
Inadequate funding for the FDA, the SEC and other domestic and foreign government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA or foreign regulatory authorities to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years the U.S. government has shut down several times and certain regulatory agencies, such as the FDA and the SEC, have had to furlough critical FDA, SEC and other government employees and stop critical activities. If a prolonged government shutdown were to occur, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business and could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Future sales of substantial amounts of our common stock, or the possibility that such sales could occur, could adversely affect the market price of our common stock.
Future sales in the public market of shares of our common stock, including shares referred to in the foregoing risk factors or shares issued upon exercise of our outstanding stock options or warrants, or the perception by the market that these sales could occur, could lower the market price of our common stock or make it difficult for us to raise additional capital.
As of December 31, 2020,2023, we had reserved for issuance 207,52015,853,773 shares of our common stock issuable upon the exercise of outstanding warrants at a weighted-average exercise price of $1.95 per share, 1,606,803 shares of our common stock issuable upon the exercise of outstanding stock options at a weighted-average exercise price of $13.38$5.92 per share, 170,000and 338,141 shares of our common stock issuable upon the vesting of outstanding restricted stock units with a weighted average grant date fair value of $1.89 per share, and 1,391,650 shares of our common stock issuable upon the exercise of outstanding warrants at a weighted-average exercise price of $27.75$2.99 per share. Upon exercise or conversion, the underlying shares, similar to those issued as the settlement payment, may be resold into the public market. In the case of outstanding securities that have exercise or conversion prices that are below the market price of our common stock from time to time, our stockholders would experience dilution upon the exercise or conversion of these securities.
Certain of our securityholders have registration rights and they can require us, subject to certain limitations, to register their securities for resale and to maintain such registration. Any such resales into the public market could place downward pressure on the price of our common stock.
We have issued and may issue additional preferred stock in the future, and the terms of the preferred stock may reduce the value of our common stock.
We are authorized to issue up to 5,000,000 shares of preferred stock in one or more series. Our board of directors may determine the terms of future preferred stock offerings without further action by our stockholders. If we issue shares of preferred stock, it could affect stockholder rights or reduce the market value of our outstanding common stock. In particular, specific rights granted to future holders of preferred stock may include voting rights, preferences as to dividends and liquidation, conversion and redemption rights, sinking fund provisions, and restrictions on our ability to merge with or sell our assets to a third party.
We have settled in the past and may in the future settle legal claims through the issuance of freely tradable shares of our common stock, which results in dilution to holders of our common stock and may adversely affect the market price of our common stockstock.
We have settled in the past and may in the future settle legal claims through the issuance of freely tradable shares of our common stock. In June 2018, we issued 4,574 unrestricted shares of our common stock (or $1,250,000 based on the volume-weighted average closing price for the 20 trading days immediately preceding June 21, 2018, the day before the transfer of the settlement stock to the settlement fund) to settle the case entitled Patel vs. Galena Biopharma, Inc. et. al. We may issue additional shares of common stock as settlement payments in the future. Payment of these amounts in our common stock could cause significant dilution to our stockholders, and the amount of that dilution will vary depending on the price of our common stock at the time of the payment. In addition, the issuance of such a significant number of shares of our common stock may cause a decrease in the trading price of our common stock.
Anti-takeover provisions of our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws and provisions of Delaware law could delay or prevent a change of control.
Anti-takeover provisions of our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws may discourage, delay or prevent a merger or other change of control that stockholders may consider favorable or may impede the ability of the holders of our common stock to change our management and may be constrained by other contractual agreements with third parties. These provisions of our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws, among other things:
•divide our Board of Directors into three classes, with members of each class to be elected for staggered three-year terms;
•limit the right of securityholders to remove directors;
•prohibit stockholders from acting by written consent;
•regulate how stockholders may present proposals or nominate directors for election at annual meetings of stockholders; and
•authorize our Board to issue preferred stock in one or more series, without stockholder approval.
In addition, Section 203 of the Delaware General Corporation Law provides that, subject to limited exceptions, persons that acquire, or are affiliated with a person that acquires, more than 15% of the outstanding voting stock of a Delaware corporation shall not engage in any business combination with that corporation, including by merger, consolidation or acquisitions of additional shares for a three-year period following the date on which that person or our affiliate crosses the 15% stock ownership threshold. Section 203 could operate to delay or prevent a change of control of us.
If our common stock becomes subject to the penny stock rules, it may be more difficult to sell our common stock.
The SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are generally equity securities with a price of less than $5.00 (other than securities registered on certain national securities exchanges or authorized for quotation on certain automated quotation systems, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or system). The OTC Bulletin Board does not meet such requirements and if the price of our common stock is less than $5.00 and our common stock is no longer listed on a national securities exchange such as Nasdaq, our stock may be deemed a penny stock. The penny stock rules require a broker-dealer, at least two business days prior to a transaction in a penny stock not otherwise exempt from those rules, to deliver to the customer a standardized risk disclosure document containing specified information and to obtain from the customer a signed and date acknowledgment of receipt of that document. In addition, the penny stock rules require that prior to effecting any transaction in a penny stock not otherwise exempt from those rules, a broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive: (i) the purchaser’s written acknowledgment of the receipt of a risk disclosure statement; (ii) a written agreement to transactions involving penny stocks; and (iii) a signed and dated copy of a written suitability statement. These disclosure requirements may have the effect of reducing the trading activity in the secondary market for our common stock, and therefore stockholders may have difficulty selling their shares.
Our common stock may be delisted fromfailure to meet the continued listing requirements of The Nasdaq Capital Market which could negatively impactresult in a delisting of our common stock.
Our shares of common stock are currently listed on The Nasdaq Capital Market. If we fail to satisfy the continued listing requirements of Nasdaq, such as the corporate governance requirements, minimum bid price requirement or the minimum stockholder's equity requirement, The Nasdaq Stock Market LLC may take steps to delist our common stock. A delisting of our common stock liquidity and our ability to access the capital markets.
from The listing standards of the Nasdaq Capital Market provide that a company, in order to qualify for continued listing, must maintain a minimum stock price of $1.00 and satisfy standards relative to minimum stockholders’ equity, minimum market value of publicly held shares and various additional requirements. If we fail to comply with all listing standards applicable to issuers listed on the Nasdaq Capital Market, our common stock may be delisted. If our common stock is delisted, it could materially reduce the priceliquidity of our common stock and the levels of liquidity available to our stockholders. In addition, the delisting of our common stock could materially adversely affect our access to the capital markets and any limitation on liquidity orresult in a corresponding material reduction in the price of our common stockstock. In addition, delisting could materially adversely affectharm our ability to raise capital. Delisting from the Nasdaq Capital Market could alsocapital through alternative financing sources on terms acceptable to us, or at all, and may result in other negative consequences, including the potential loss of confidence by investors, suppliers, customers and employees the loss of institutional investor interest and fewer business development opportunities.
As previously reported, on May 31, 2019,On December 6, 2023, we received a letter from Nasdaq indicatingnotifying us that we no longer met Nasdaq's requirements for continued listing on The Nasdaq Capital Market under Nasdaq Listing Rule 5550(b)(2), or the MVLS Rule, because, for a period of 30 consecutive business days, the market value of our common stock, calculated based upon the most recent total shares outstanding multiplied by the closing bid price per share, had not maintained a minimum of $35.0 million.
In accordance with Nasdaq Listing Rule 5810(c)(3)(C), we were provided a period of 180 calendar days, or until June 3, 2024, in which to regain compliance with the MVLS Rule. To regain compliance, the market value of our common stock must meet or exceed $35.0 million for a minimum of 10 consecutive business days during the 180-day compliance period. If we have not regained compliance with the MVLS Rule by June 3, 2024, Nasdaq will provide notice to us that our securities will be subject to delisting, in which case we may appeal the delisting determination to a Nasdaq Hearings Panel.
On March 5, 2024, we received a letter from Nasdaq stating that we have regained compliance under the MVLS Rule by maintaining a market value of our common stock of greater than $35 million for 10 consecutive business, and that the matter is now closed.
On February 14, 2024, we received a letter from Nasdaq notifying us that we no longer met Nasdaq's requirements for continued listing on The Nasdaq Capital Market under Nasdaq Listing Rule 5550(a)(2), or the Minimum Bid Price Requirement, because, for the preceding 30 consecutive business days, our common stock did not maintain a minimum closing bid price of $1.00 per share.
In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were provided a period of 180 calendar days, or until August 12, 2024, to regain compliance with the Minimum Bid Price Requirement. Compliance can be achieved automatically and without further action if the closing bid price of our common stock is at or above $1.00 for the last 30a minimum of 10 consecutive business days we did not meetat any time during the minimum bid price of $1.00 per share required for continued listing on the180-day compliance period, in which case Nasdaq Capital Market pursuant to the Minimum Bid Price Rule. In connection therewith, we effected a 1-for-50 reverse stock split of our common stock on November 7, 2019, and on November 25, 2019, we were informed by Nasdaq that we regained compliance with the Minimum Bid Price Rule.
Notwithstandingwill notify us of our compliance and the matter will be closed. If, however, we do not achieve compliance with the Minimum Bid Price Requirement by August 12, 2024, we may be eligible for additional time to comply. In order to be eligible for such additional time, we will be required to meet the continued listing requirement for market
value of publicly held shares and all other initial listing standards for The Nasdaq Capital Market, with the exception of the Minimum Bid Price Requirement, and must notify Nasdaq in writing of our intention to cure the deficiency during the second compliance period, by effecting a reverse stock split, and ourif necessary.
On March 11, 2024, we received a letter from Nasdaq stating that we have regained compliance with The Nasdaq Capital market requirements, we cannot be sure that our shareunder the Minimum Bid Price Requirement by maintaining a minimum closing bid price will comply with the requirements for continued listing of our common stock of $1.00 per share for 10 consecutive business days, from February 26, 2024 to March 8, 2024, and that the matter is now closed.
Our common stock currently remains listed on The Nasdaq Capital Market inunder the future, or that we will comply with the other continued listing requirements. If our shares of common stock lose their status on Nasdaq, we believe that they would likely be eligible to be quoted on the inter-dealer electronic quotation and trading system operated by Pink OTC Markets Inc., commonly referred to as the Pink Sheets and now known as the OTCQB market. Our shares of common stock may also be quoted on the Over-the-Counter Bulletin Board, an electronic quotation service maintained by the Financial Industry Regulatory Authority. These markets are generally not considered to be as efficient as, and not as broad as, Nasdaq. Selling our shares on these markets could be more difficult because smaller quantities of shares would likely be bought and sold, and transactions could be delayed. In addition, in the event our shares are delisted, broker-dealers have certain regulatory burdens imposed upon them, which may discourage broker-dealers from effecting transactions in our common stock, further limiting the liquidity of our common stock. These factors could result in lower prices and larger spreads in the bid and ask prices for our common stock.symbol SLS.
We have never declared or paid cash dividends on our common stock and we do not anticipate paying cash dividends on our common stock in the foreseeable future.
Our business requires significant funding. We currently plan to invest all available funds and future earnings in the development and growth of our business and do not anticipate paying any cash dividends on our common stock in the foreseeable future. As a result, capital appreciation, if any, of our common stock will be our stockholders’ sole source of potential gain for the foreseeable future.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 1C. CYBERSECURITY
We recognize the critical importance of maintaining the trust and confidence of business partners, such as CROs and CMOs, clinical trial investigators, patients and employees toward our business and are committed to protecting the confidentiality, integrity and availability of our business operations and systems. Our board of directors is actively involved in oversight of our risk management activities, and cybersecurity represents an important element of our overall approach to risk management. In general, we seek to address cybersecurity risks through a comprehensive, cross-functional approach that is focused on preserving the confidentiality, security and availability of the information that we collect and store by identifying, preventing and mitigating cybersecurity threats and effectively responding to cybersecurity incidents when they occur.
Cybersecurity Risk Management and Strategy; Effect of Risk
We face risks related to cybersecurity, such as unauthorized access, cybersecurity attacks, and other security incidents, including perpetration by hackers and unintentional damage or disruption to hardware and software systems, loss of data, and misappropriation of confidential information. To identify and assess material risks from cybersecurity threats, we, together with our contracted third-party cybersecurity advisors, maintain a comprehensive cybersecurity program to ensure our systems are effective and prepared for information security risks, including regular oversight of our programs for security monitoring of internal and external threats to ensure the confidentiality and integrity of our information assets. We consider risks from cybersecurity threats alongside other company risks as part of our overall risk assessment process. As discussed in more detail under “Cybersecurity Governance” below, our audit committee provides oversight of our cybersecurity risk management and strategy processes, which are led by management.
We, with assistance from our contracted third-party cybersecurity advisors, identify our cybersecurity threat risks by comparing our processes to standards set by the National Institute of Standards and Technology, or NIST. To provide for the availability of critical data and systems, maintain regulatory compliance, manage our material risks from cybersecurity threats, and protect against and respond to cybersecurity incidents, we undertake the following activities:
•monitor emerging data protection laws and implement changes to our processes that are designed to comply with such laws;
•through our policies, practices, and contracts (as applicable), require employees, as well as third parties that provide services on our behalf, to treat confidential information and data with care;
•employ technical safeguards that are designed to protect our information systems from cybersecurity threats, including firewalls, intrusion prevention and detection systems, anti-malware functionality and access controls;
•provide regular, mandatory training for our employees and contractors regarding cybersecurity threats as a means to equip them with effective tools to address cybersecurity threats, and to communicate our evolving information security policies, standards, processes and practices;
•conduct regular phishing email simulations for all employees and contractors with access to our email systems to enhance awareness and responsiveness to possible threats;
•conduct cybersecurity management and incident training for employees involved in our systems and processes that handle sensitive data;
•leverage the NIST incident handling framework to help us identify, protect, detect, respond and recover when there is a potential cybersecurity incident; and
•carry information security risk insurance that provides protection against the potential losses arising from a cybersecurity incident.
Our processes also address cybersecurity threat risks associated with our selection and oversight of third-party service providers, including our suppliers and manufacturers or those who have access to patient and employee data or our systems. We generally require those third parties that could introduce significant cybersecurity risk to us to agree by contract to manage their cybersecurity risks in specified ways.
We describe whether and how risks from identified cybersecurity threats, including as a result of any previous cybersecurity incidents, have materially affected or are reasonably likely to materially affect us, including our business strategy, results of operations, or financial condition, under the heading Significant disruptions of information technology systems, computer system failures or cybersecurity incidents could adversely affect our business, which disclosures are incorporated by reference herein.
In the last three fiscal years, we have not experienced any material cybersecurity incidents.
Cybersecurity Governance; Management
Cybersecurity is an important part of our risk management processes and an area of focus for our Board of Directors and management. Management is responsible for the operational oversight of company-wide cybersecurity strategy, policy, and standards across relevant departments to assess and help prepare us to address cybersecurity risks.
The Audit Committee of the Board of Directors, or the Audit Committee, provides direct oversight over cybersecurity risk and periodically updates our Board of Directors on such matters. The Audit Committee receives periodic updates from management regarding cybersecurity matters, and is notified between such updates regarding any significant new cybersecurity threats or incidents.
Our cybersecurity risk management and strategy processes, which are discussed in greater detail above, are led by our Chief Financial Officer and Vice President, Associate General Counsel and Head of Compliance, with the assistance of our contracted third-party cybersecurity advisors. These management team members are informed about and monitor the prevention, mitigation, detection, and remediation of cybersecurity incidents through their management of, and participation in, the cybersecurity risk management and strategy processes described above, including incident response processes. As discussed above, these management team members periodically report to the Audit Committee about cybersecurity threat risks, among other cybersecurity related matters.
ITEM 2. PROPERTIES
We lease our headquarters in New York, New York. The lease covers approximately 5,1438,455 square feet of office space, which includes additional space beginning on February 22, 2022, and expires in December 2024.September 2025. We believe that our facility is adequate for our current needs.
ITEM 3. LEGAL PROCEEDINGS
Our predecessor, Galena, was involvedIn December 2020, we entered into 3D Medicines Agreement. In November 2022, we announced that we had agreed with 3D Medicines for 3D Medicines to participate in multiple legal proceedings and administrative actions, including stockholder class actions, both state and federal, somethe REGAL study through the inclusion of which are ongoing and to which we are now subject as a resultapproximately 20 patients from mainland China.
In accordance with the terms of the Merger. They are as follows:3D Medicines Agreement and the Side Letter, we had expected that 3D Medicines would begin enrolling patients in mainland China in the REGAL study in the second half of 2023 and subsequently make two development milestone payments totaling $13.0 million. Patients were enrolled in the REGAL study in Taiwan, which is part of the 3DMed Territory, prior to the second half of 2023.
On February 13, 2017, certain putative shareholder securities class action complaints were filed in federal court alleging,December 20, 2023, we commenced a binding arbitration proceeding against 3D Medicines, administered by the Hong Kong International Arbitration Centre and governed by New York State law as per the 3D Medicines
Agreement. The arbitration proceeding involves, among other things, that Galenathe trigger and certainpayment of Galena's former officersthe relevant milestone payments due to us as well as 3D Medicines’ failure to use commercially reasonable best efforts to develop GPS in the 3DMed Territory, and directors failedparticularly in mainland China.
We have engaged an international law firm with expertise in mainland China to disclose that Galena’s promotional practices for Abstral® (fentanyl sublingual tablets) were allegedly improper and that Galena may be subject to civil and criminal liability, and that these alleged failures rendered Galena’s statements about its business misleading. The actions were consolidated, lead plaintiffs were named byassist us with the U.S. District Court for the District of New Jersey and a consolidated complaint was filed. We filed a motion to dismiss the consolidated complaint. On August 21, 2018, our motion to dismiss the consolidated complaint was granted without prejudice to file an amended complaint. On September 20, 2018, the plaintiffs filed an amended complaint. On October 22, 2018,arbitration proceeding. While we filed a motion to dismiss the amended complaint. On November 13, 2019, the U.S. District Court for the District of New Jersey granted our motion to dismiss without prejudice to file an amended complaint. On December 20, 2019, the lead plaintiffs filed a second Amended Consolidated Class Action Complaint. On January 29, 2020, we filed a motion to dismiss the amended complaint. On January 5, 2021, the U.S. District Court for the District of New Jersey granted our motion to dismiss without prejudice to file an amended complaint. On February 18, 2021, the lead plaintiffs filed a third Amended Consolidated Class Action Complaint. We deny the allegations in this action and intend to vigorously defend against them. We are unable however,at this time to predict with certainty the outcome of this matter at this time.
In March 2017, a derivative complaint was filedthe arbitration proceeding, or the timing of the receipt of any milestone payments and other damages it is seeking in the U.S. District Court for the District of New Jersey against Galena’s former directors and Galena, as a nominal defendant. In July 2017, a derivative complaint was filed in California state court against Galena’s former directors and Galena, as a nominal defendant. In January 2018, a derivative complaint was filed in the U.S. District Court for the District of New Jersey against Galena’s former directors, officers and employees, and us as a nominal defendant. These complaints purport to assert derivativearbitration proceeding, if at all, we believe that our claims for breach of fiduciary duty on our behalf against our former directors and, in certain of the complaints, certain of our former officers and former employees, based on substantially similar facts as alleged in the putative shareholder securities class action complaints mentioned above. The derivative lawsuit filed in California state court is currently stayed pending resolution of a motion to dismiss in the referenced securities class action. On July 13, 2020 and July 16, 2020, respectively, we filed motions to dismiss the two complaints filed in the U.S. District Court for the District of New Jersey. We have reached a settlement in principle with the plaintiffs in these three cases which is subject to final documentation and court approval.are meritorious.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
PART II
ITEM 5. MARKET FOR REGISTRANT'S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is listed on The Nasdaq Capital Market under the symbol SLS.
Holders
As of March 16, 2021,27, 2024, there were approximately 1735 holders of record of our common stock. Because many of our shares are held by brokers and other institutions on behalf of stockholders, we are unable to estimate the total number of individual stockholders represented by these holders of record.
Dividends
We have never paid any cash dividends on our common stock and do not anticipate paying any cash dividends on our common stock in the foreseeable future. We expect to retain future earnings, if any, for use in our development activities and the operation of our business. The payment of any future dividends will be subject to the discretion of our Board of Directors and will depend, among other things, upon our results of operations, financial condition, cash requirements, prospects and other factors that our Board of Directors may deem relevant. Our ability to pay future dividends may be restricted by the terms of any future securities we may issue.
Recent Sales of Unregistered Securities
During the period covered by this annual report,Annual Report on Form 10-K, there were no sales by us of unregistered securities that were not previously reported by us in a Quarterly Report on Form 10-Q or Current Report on Form 8-K.
Purchases of Equity Securities
During the year ended December 31, 2020,2023, we did not purchase any of our equity securities. Our Board of Directors has not authorized any repurchase plan or program for the purchase of shares of our common stock or other securities on the open market or otherwise.
Equity Compensation Plan Information
The following table provides information regarding the status of our existing equity compensation plans as of December 31, 2020:2023:
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of Securities to be Issued upon Exercise of Outstanding Options, Warrants and Rights | | Weighted Average Exercise Price of Outstanding Options, Warrants and Rights | | Number of Securities Remaining Available for Future Issuance under Equity Compensation Plans (Excluding Securities Reflected in Previous Columns) |
| Equity compensation plans approved by security holders | | | | | | |
| 2017 Equity Incentive Plan | | 21,520 | | | $ | 112.85 | | | — | |
| 2019 Equity Incentive Plan | | 186,000 | | | $ | 1.87 | | | 100,689 | |
| Restricted Stock Units | | 170,000 | | | N/A | | — | |
| Employee Stock Purchase Plan | | — | | | N/A | | 8,302 | |
| Equity compensation plans not approved by security holders | | | | | | |
| None | | — | | | — | | | — | |
| Total | | 377,520 | | | $ | 13.38 | | | 108,991 | |
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of Securities to be Issued upon Exercise of Outstanding Options, Warrants and Rights | | Weighted Average Exercise Price of Outstanding Options, Warrants and Rights | | Number of Securities Remaining Available for Future Issuance under Equity Compensation Plans (Excluding Securities Reflected in Previous Columns) |
| Equity compensation plans approved by security holders | | | | | | |
| 2017 Equity Incentive Plan | | 21,520 | | | $ | 112.85 | | | — | |
| 2023 Amended and Restated Equity Incentive Plan | | 1,585,283 | | | $ | 4.47 | | | 3,950,528 | |
| Restricted Stock Units | | 338,141 | | | N/A | | — | |
| 2021 Employee Stock Purchase Plan | | — | | | N/A | | 183,457 | |
| Equity compensation plans not approved by security holders | | | | | | |
| None | | — | | | — | | | — | |
| Total | | 1,944,944 | | | $ | 5.92 | | | 4,133,985 | |
ITEM 6. SELECTED FINANCIAL DATA[RESERVED]
Not applicable.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The purpose of this Management's Discussion and Analysis is to better allow our investors to understand and view our company from management's perspective. We are providing an overview of our business and strategy including a discussion of our financial condition and results of operations. You should read the following discussion in conjunction with the consolidated financial statements and the notes to the consolidated financial statements included elsewhere in this annual reportAnnual Report on Form 10-K. This discussion contains forward-looking statements within the meaning of federal securities laws. Such forward-looking statements are subject to risks and uncertainties that could cause actual results to differ materially from those contained in such forward-looking statements, including those discussed in the section “Risk Factors” in Part I — Item 1A of this annual reportAnnual Report on Form 10-K.
Please see Part I, Item 1 “Business— Strategic Collaboration and License Agreements” and Note 5 to our audited consolidated financial statements appearing elsewhere in this annual report on Form 10-K for more information relating to such arrangements.
Overview
We are a late-stage clinical biopharmaceutical company focused on developingthe development of novel cancer immunotherapeuticstherapeutics for a broad range of cancer indications. Our product candidates currently include galinpepimut-S, or GPS, a peptide immunotherapy directed against the Wilms tumor 1, or WT1, antigen, and nelipepimut-S.SLS009 (formerly GFH009), a highly selective small molecule cyclin-dependent kinase 9, or CDK9, inhibitor.
Galinpepimut-S, or GPSGPS: Highly Novel and Engineered Immunotherapy Targeting the WT1 Antigen
Our lead product candidate, galinpepimut-S, or GPS, is a cancer immunotherapeutic agent licensed from Memorial Sloan Kettering Cancer Center, or MSK, that targets the Wilms tumor 1, or WT1 protein, which is present in 20 or more cancer types. Based on its mechanism of action as a directly immunizing agent, GPS has potential as a monotherapy or in combination with other immunotherapeutic agents to address a broad spectrum of hematologic, or blood, cancers and solid tumor indications.
In January 2020, we commenced ain the United States an open label randomized Phase 3 clinical trial, the REGAL study,for GPS monotherapy in patients with acute myeloid leukemia, or AML, in the maintenance setting after achievement of second complete remission, or CRem2,CR2, following successful completion of second-line antileukemic therapy. Patients are randomized (1:1) to receive either GPS or best available treatment, or BAT. We expect this study will be used as the basis for submission of a Biologics License Application, or BLA, subject to a statistically significant and clinically meaningful data outcome and agreement with the U.S. Food &and Drug Administration, or the FDA. The primary endpoint of the REGAL study is overall survival. We expectplanned to enroll approximately 116125 to 140 patients at up to approximately 13595 clinical sites primarily in the United StatesNorth America, Europe and EuropeAsia with a planned interim safety, efficacy and futility analysis after 60 events (deaths). In March 2024, we announced the completion of enrollment. Under our current assumptions with respect to enrollment and the estimated survival times for both the treated and control groups in the study, we believe, after discussions with our external statisticians and experts, that the planned interim analysis after 60 events (deaths) per the protocol will occur in the first half of 2024 and the final analysis after 80 events (deaths).will occur by the end of 2024. Because these analyses are event driven, they are difficult to predict with any certainty and may occur at a different time than currently expected.
In December 2020, we entered into an exclusive license agreement, or 3D Medicines Agreement, with 3D Medicines Inc., or 3D Medicines, a China-based biopharmaceutical company developing next-generation immuno-oncology drugs, for the development and commercialization of GPS, as well as the Company’s next generation heptavalent immunotherapeutic GPS+, which is at preclinical stage, across all therapeutic and diagnostic uses in mainland China, Hong Kong, Macau and Taiwan, which we collectively refer to as Greater China, or the 3DMed Territory. We have retained sole rights to GPS and GPS+ outside of Greater China. In November 2022, we announced that we had agreed with 3D Medicines for 3D Medicines to participate in the REGAL study through the inclusion of approximately 20 patients from mainland China. Although the REGAL study has completed enrollment as announced in March 2024, in accordance with the predetermined statistical analysis plan, 3D Medicines may still enroll patients in mainland China. The timing of such participation and patient enrollment by 3D Medicines, if at all, cannot be predicted with certainty. As of December 31, 2023, we have received an aggregate of $10.5 million in upfront and milestone payments under the 3D Medicines Agreement and a total of $191.5 million in potential future development, regulatory and sales milestones, not including future royalties, remains under the license agreement, which milestones are variable in nature and not under our control. In December 2023, we commenced an arbitration proceeding against 3D Medicines regarding, among other things, the trigger and payment of $13.0 million in milestone payments due to us. See Item 3. Legal Proceedings.
In December 2018, pursuant to a Clinical Trial Collaboration and Supply Agreement, we initiated a Phase 1/2 multi-arm "basket" type clinical study of GPS in combination with Merck & Co., Inc.’s anti-PD-1 therapy, Keytruda® (pembrolizumab)pembrolizumab (Keytruda®). Thetumor type currently being studied isIn 2020, we, together with Merck, determined to focus on ovarian cancer (second or third line). In November 2022, we reported topline clinical and initial immune response data from this study, which showed that treatment with the combination of GPS and pembrolizumab compared favorably to treatment with anti-PD-1 therapy alone in a similar patient population. In November 2023, additional immunobiological and clinical data from the study was presented at the International Gynecologic Cancer Society 2023 Annual Global Meeting which showed a correlation between immune response and PFS.
In February 2020, a Phase I1 open-label investigator-sponsored clinical trial of GPS, in combination with Bristol-Myers Squibb’s anti-PD-1 therapy, nivolumab (Opdivo®), in patients with malignant pleural mesothelioma, or MPM, who harbor relapsed or refractory disease after having received frontline standard of care multimodality therapy was commenced at MSK. Enrollment of a target total of 10 evaluable patients was completed at the end of 2022. We reported positive topline safety and efficacy data from this study in June 2023 and positive follow-up immune response and survival data in December 2023.
GPS was granted Orphan Drug Product Designations, or ODD, from the FDA, as well as Orphan Medicinal Product Designationsorphan medicines designations from the European Medicines Agency, or EMA, for GPS in AML, malignant pleural mesothelioma, or MPM, and multiple myeloma, or MM, as well as Fast Track Designationdesignation for AML, MPM, and MM from the FDA.
Nelipepimut-S or NPSSLS009: Highly Selective Next Generation CDK9 Inhibitor
Nelipepimut-S,On March 31, 2022, we entered into an exclusive license agreement, or NPS, isthe GenFleet Agreement, with GenFleet Therapeutics (Shanghai), Inc., or GenFleet, a cancer immunotherapy targeting the human epidermal growth factor receptor 2, or HER2, expressing cancers. Data presentedclinical-stage biotechnology company developing cutting-edge therapeutics in 2018 from a Phase 2b clinical trial of the combination of trastuzumab (Herceptin®) plus NPS in HER2 low expressing (1+ or 2+ per immunohistochemistry, or IHC) breast cancer patients in the adjuvant settingoncology and immunology, that grants rights to prevent recurrences showed a clinically and statistically significant improvement in the disease-free survival, or DFS, rateus for the triple negativedevelopment and commercialization of SLS009, a highly selective small molecule CDK9 inhibitor, across all therapeutic and diagnostic uses worldwide, except for Greater China.
CDK9 activity has been shown to correlate negatively with overall survival in a number of cancer types, including hematologic cancers, such as AML and lymphomas, as well as solid cancers, such as osteosarcoma, pediatric soft tissue sarcomas, melanoma, endometrial, lung, prostate, breast cancer, or TNBC, cohort at 24 months for patients treated with NPS plus trastuzumab of 92.6% compared to 70.2% for those treated with trastuzumab alone. Following ongoing discussions with the FDA and based upon written feedback from the FDAovarian. As demonstrated in preclinical and on the totality of clinical safety and translational NPS data, to date, we have finalizedSLS009’s high selectivity has the designpotential to reduce toxicity as compared to older CDK9 inhibitors and plan for a Phase 3 registration-enabling study of NPSother next-generation CDK9 inhibitors currently in combination with trastuzumab for the treatment of patients with TNBC in the adjuvant setting after standard treatment. If successful, we believe this study may be considered as the basis for a BLA submission to the FDA. We are seeking out-licensing opportunities to fund and conduct the future clinical development of NPS in orderand to maximize the potential of the program and we do not plan to conduct and fund a Phase 3 program for NPS on our own.potentially be more efficacious.
FBP-targeting bivalent vaccine (GALE-301/-302)We completed a Phase 1 dose-escalating clinical trial in the United States and China for SLS009 in mid-2023 and reported positive safety and efficacy data for both patient cohorts, that is relapsed and/or refractory AML and refractory lymphoma. We also established in the trial a recommended Phase 2 dose, or RP2D, of 60 mg for AML and 100 mg for lymphomas.
In orderthe second quarter of 2023, we commenced an open label, single arm, multi-center Phase 2a clinical trial with SLS009 in combination with venetoclax and azacitidine, or aza/ven, in patients with AML who failed or did not respond to prioritize developmenttreatment with venetoclax-based therapies. The trial is evaluating safety, tolerability, and efficacy at two dose levels of our core assets, we determinedSLS009, 45 mg once weekly, and 60 mg once weekly or 30 mg twice a week, in combination with aza/ven. In addition to cease developmentsafety and tolerability of GALE-301SLS009 in combination with aza/ven, the primary endpoints are complete response composite rate and GALE-302, cancer immunotherapies that targetduration of response. Additional endpoints include event free survival, overall survival, and pharmacokinetic and pharmacodynamic assessments. The trial includes several sites in the E39 peptide derived from the folate binding protein, or FBP, which were licensed in from The Henry M. Jackson Foundation, or HJF,United States, will enroll a minimum of 20 patients and, the MD Anderson Cancer Center, or MDACC and enteredbased on initial results, may be expanded into a Termination Agreement with HJF and MDACC in February 2021.registrational trial.
In the fourth quarter of 2023, we completed enrollment in the 45 mg (safety) dose cohort in the Phase 2a study and reported positive initial topline data. We also commenced enrollment in the 60 mg dose cohort with patients randomized to one of two groups, 60 mg fixed dose once weekly or 30 mg fixed twice weekly. Each group will enroll five to 10 patients.
In March 2024, we announced positive topline data from the Phase 2a clinical trial of SLS009 in combination with aza/ven in r/r/ AML. A total of 21 patients were enrolled in the study as of March 15, 2024: 10 in the 45 mg safety cohort and 11 in the 60 mg cohort (30 mg twice a week or 60 mg once a week). Response rates observed in the three cohorts were 10% in the 45 mg once a week safety dose cohort (dose level below the RP2D),
Financial Position20% in the 60 mg once a week dose cohort, and 50% in the 30 mg twice a week dose cohort. Additionally, we observed strong anti-leukemic activity, which is defined as 50% or more bone marrow blast reduction in 67% of patients across all dose levels. Median OS has not been reached in any of the cohorts and the first patient enrolled in the study who achieved a CR continues on the study and remains leukemia-free 9 months after enrollment. During the trial, we identified potential biomarkers currently undergoing testing as predictive markers in the most recent portion of the study. Patients with the identified biomarkers exhibited significantly higher response rates: 100% response rate at the optimal dose level (30 mg twice a week) and 57% response rate across all dose levels. Furthermore, we have clarified the proposed biological basis and mechanism of action for SLS009 activity in patients with these biomarkers. The relevant biomarkers are present in multiple hematologic and solid cancer indications, with a substantial proportion of patients exhibiting them in additional indications, ranging up to ~50% of patients in some indications.
At December 31, 2020, we had cash and cash equivalents of $35.3 million. We have incurred operating losses since inception and have not generated any product sales revenue or achieved profitable operations. We incurred net losses of $16.8 million and $19.3 millionAdditional topline data for the years ended December 31, 2020 and 2019, respectively. Our accumulated deficit as60 mg dose cohort is expected in the second quarter of December 31, 2020 was $117.9 million, and we expect to continue to incur substantial losses in future periods.2024.
Our operating expenses will increase substantially asIn October 2023, we continue to advance our product candidates assuming we receive sufficient funding to continue our ongoing studies and initiate our planned studies. We anticipateannounced that our expensespartner, GenFleet, dosed the first patient in a Phase Ib/II trial evaluating SLS009 in patients with relapsed/refractory, or r/r, peripheral T-cell lymphoma, or PTCL. The open-label, single-arm trial will increaseenroll up to 95 patients to evaluate safety and efficacy and, based on the results, may serve as we:
•complete our Phase 3 clinical triala registrational study. This initial PTCL study is fully funded by GenFleet and our Phase 1/2 basket study;
•continueis being conducted in China. We expect to report initial topline data from the research, development and scale-upstudy in the second quarter of manufacturing capabilities to optimize products and dose forms for which we may obtain regulatory approval;
•scale up manufacturing for GPS, including manufacturing validation activities;
•maintain, expand and protect our global intellectual property portfolio;
•hire additional personnel, including clinical, manufacturing, and scientific personnel, sales and marketing personnel, and general and administrative personnel.
We intend to use our existing cash and cash equivalents for working capital and to fund the research and development of our product candidates. We believe that our existing cash and cash equivalents as of December 31, 2020 will enable us to fund our operating expenses for at least the next 12 months following the issuance of our financial statements.2024.
ImpactIn March 2024, GenFleet announced that it entered into a collaboration and supply agreement with BeiGene Switzerland GmbH to initiate a combination study of COVID-19SLS009 and Brukinsa (zanubrutinib), a BTK inhibitor, in r/r diffused large B-cell lypmhoma, or DLBCL, and the first patient was dosed in the trial. The open-label, single-arm multi-center trial will be conducted in two parts. In the Phase 1b portion, 6-18 patients will be enrolled. In the Phase 2 portion, approximately 45 patients will be enrolled. This study is funded by GenFleet and is being conducted in China.
On March 11, 2020,SLS009 is also currently being evaluated in pediatric solid tumors and leukemia models through the World Health Organization declaredNCI Pediatric Preclinical in Vivo Testing, or PIVOT, program. Studies are supported through cooperative agreement grants from the outbreak ofNCI to the seven PIVOT research programs performing the PK and efficacy testing in pediatric tumors and a new coronavirus to be a “pandemic”. The COVID-19 pandemic continues to present substantial public health and economic challenges around the world which have impacted, and will continue to impact, millions of individuals and business worldwide. Efforts to contain the spread of the coronavirus since March 2020 have led to travel bans and restrictions, quarantines, shelter-in-place orders and shutdowns. As we have historically functioned operationally as a semi-virtual company, the transition to “work-from-home” for our employees has not materially altered our business operations.centralized coordinating center. We have implemented a return-to-work policy in compliance with federal, state and local requirements and guidance which provides for a hybrid of remote and in-office work, and we expect to operate on such a semi-virtual basis for at leastreport relevant data from the firstprogram in the second half of 2021. We are continuously monitoring2024.
For SLS009, the impact of the pandemic on our clinical development programs. Our Phase 3 REGAL study is progressing, with the necessary work to activate additional sitesFDA granted Orphan Drug Product designations in the United StatesAML and Europe continuing. Throughout 2020PTCL and early 2021, we initiated additional sites as planned. However, we have observed that clinical site initiationsFast Track designations for r/r AML and patient enrollment may be delayed due to prioritization of hospital resources towards the COVID-19 pandemic. Clinicians and patients may not be able to comply with clinical trial protocols if quarantines impede patient movement or interrupt operations at sites. Accordingly, we are uncertain at this time the extent to which these newly initiated sites will be fully operational, which we believe could have an impact on the projected timing of the REGAL study. Additionally, several European Union countries in which we plan to initiate clinical sites, including Germany, France, and Italy, continue to impose restrictions in response to the continued surge in coronavirus cases throughout the European Union. We believe that the COVID-19 pandemic has not materially impacted our efforts to out-license NPS. The full extent to which the COVID-19 pandemic directly or indirectly impacts our business, results of operations and financial condition will depend on future developments that are highly uncertain, subject to change and cannot be predicted with confidence, including the actions taken to contain or treat COVID-19, the overall duration of the outbreak, the availability, effectiveness and uptake of vaccines for COVID-19, the emergence of new variants of COVID-19 and whether existing vaccines are effective with respect to such variants, and the emergence of new geographic hotspots where the coronavirus is spreading more rapidly. In particular, the continued spread of the coronavirus globally could adversely impact our clinical trial operations and could have an adverse impact on our business and the financial results.r/r PTCL.
Components of Results of Operations
LicenseLicensing Revenue
LicenseLicensing revenue consists of revenue recognized pursuant to our Exclusive License Agreement withthe 3D Medicines Inc., or 3DMed, dated December 7, 2020, or the 3DMed Agreement. In the future, we may generate revenue from a combination of reimbursements, up-front payments,regulatory, development, and sales milestone payments and royalties in connection with the 3DMed3D Medicines Agreement.
Cost of Licensing Revenue
Cost of licensing revenue consists of sublicensing fees incurred under our license from MSK in connection with the 3D Medicines Agreement.
Research and Development
Research and development expense consists of expenses incurred in connection with the discovery and development of our product candidates. We expense research and development costs as incurred. These expenses include:
•expenses incurred under agreements with clinical research organizations, or CROs, as well as investigative sites and consultants that conduct our preclinical studies and clinical trials;
•manufacturing and clinical drug supply expenses;
•outsourced professional scientific development services;
•employee-related expenses, which include salaries, benefits and stock-based compensation;
•payments made under our license agreements, under which we acquired certain intellectual property;
•expenses relating to certain regulatory activities, including filing fees paid to regulatory agencies;
•laboratory materials and supplies used to support our research activities; and
•allocated expenses, utilities and other facility-related costs.
The successful development of our current and future product candidates is highly uncertain. At this time, we cannot reasonably estimate or know the nature, timing and costs of the efforts that will be necessary to complete the remainder of the development of, or when, if ever, material net cash inflows may commence from, any current or future product candidates. This uncertainty is due to the numerous risks and uncertainties associated with the duration and cost of our clinical trials, which vary significantly over the life of a project as a result of many factors, including:
•the number and geographical location of clinical sites included in the trials;
•the length of time required to enroll suitable patients;
•the number and geographical location of patients that ultimately participate in the trials;
•the number of doses patients receive;
•the duration of patient follow-up;
•the results of clinical trials;
•the expenses associated with manufacturing;manufacturing and clinical drug supply;
•the receipt of marketing approvals; and
•the commercialization of current and future product candidates.
Research and development activities are central to our business model. Cancer immunotherapyOncology product candidates in the later stages of clinical development generally have higher development costs than those in the earlier stages of clinical development, primarily due to the increased size and duration of the later-stage clinical trials. We expect our research and development expenses to increase for the foreseeable future as we conduct and complete our ongoing early and late stagelate-stage clinical trials, initiate additional clinical trials, and initiate additional clinical trials.expand regulatory activities associated with the preparation and submission of regulatory filings.
Our expenditures are subject to additional uncertainties, including the terms and timing of regulatory approvals. We may never succeed in achieving regulatory approval for any of our current or future product candidates. We may obtain unexpected results from our clinical trials. We may elect to discontinue, delay or modify clinical trials of some product candidates or target indications or focus on others. A change in the outcome of any of these variables with respect to the development of a product candidate could mean a significant change in the costs and timing associated with the development of that product candidate. For example, if the FDA or other regulatory authorities were to require us to conduct clinical trials beyond those that we currently anticipate, or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development.
General and Administrative Expense
General and administrative expenses consist principally of salaries and related costs for personnel in executive, administrative, finance and legal functions, including stock-based compensation, travel expenses and recruiting expenses, fees for outside legal counsel, and director and officer insurance premiums. Other general and administrative expenses include facility related costs, patent filing and prosecution costs, professional fees for business development, accounting, consulting, legal and tax-related services associated with maintaining compliance with our Nasdaq listing and SEC reporting requirements, investor relations costs, and other expenses associated with being a public company.
If and when we believe that regulatory approval of a product candidate appears likely, we anticipate that an increase in general and administrative expenses will occur as a result of our preparation for commercial operations,
particularly as it relates to the sales and marketing of such product candidate. Cancer immunotherapyOncology product commercialization may take several years and millions of dollars in development costs.
In ProcessAcquired In-Process Research and Development Impairment Charge
The impairment charge recognized during the year ended December 31, 2019 was in connection with the abandonment of future development of GALE-301 and GALE-302Acquired in-process research and development consists of costs to acquire or IPR&D, assets.license product candidates from third parties for development with no alternative future use as the technology and know-how acquired are not currently commercially viable.
Non-Operating Income (Expense), Net
Non-operating income (expense), net consists of changes in fair value of our warrant liability, changes in fair value of our contingent consideration, and interest income. Interest income primarily reflects the interest earned from our cash and cash equivalents.
Results of Operations for the Years Ended December 31, 20202023 and 20192022
The following table summarizes our results of operations for the years ended December 31, 20202023 and 2019:2022 (amounts in thousands):
| | | | | | | | | | | | | | | | | |
| (dollars in thousands) | Year ended December 31, | | |
| 2020 | | 2019 | | Change |
| License revenue | $ | 1,900 | | | $ | — | | | $ | 1,900 | |
| Operating expenses: | | | | | |
| Research and development | 9,282 | | | 7,285 | | | 1,997 | |
| General and administrative | 9,600 | | | 9,923 | | | (323) | |
| In-process research and development impairment charge | — | | | 2,833 | | | (2,833) | |
| Total operating expenses | (18,882) | | | (20,041) | | | (1,159) | |
| Loss from operations | (16,982) | | | (20,041) | | | (3,059) | |
| Non-operating income | 208 | | | 668 | | | (460) | |
| Loss before income taxes | (16,774) | | | (19,373) | | | (2,599) | |
| Income tax benefit | (17) | | | (81) | | | 64 | |
| Net loss | $ | (16,757) | | | $ | (19,292) | | | $ | (2,535) | |
For the year ended December 31, 2020, our net loss was $16.8 million compared with a net loss of $19.3 million for the year ended December 31, 2019. The decrease of $2.5 million in net loss was primarily attributable to a decrease in operating loss of $3.1 million, primarily driven by a $1.9 million increase in license revenue, a $0.3 million decrease in general and administrative expenses, and a $2.8 million decrease in non-cash charges for impairments of IPR&D, partially offset by an increase of $2.0 million in research and development expenses, a decrease in non-operating income of $0.5 million and a decrease in income tax benefit of $0.1 million. | | | | | | | | | | | | | | | | | |
| Year ended December 31, | | |
| 2023 | | 2022 | | Change |
| Licensing revenue | $ | — | | | $ | 1,000 | | | $ | (1,000) | |
| Operating expenses: | | | | | |
| Cost of licensing revenue | — | | | 100 | | | (100) | |
| Research and development | 24,007 | | | 20,268 | | | 3,739 | |
| General and administrative | 13,862 | | | 12,582 | | | 1,280 | |
| Acquired in-process research and development | — | | | 10,000 | | | (10,000) | |
| Total operating expenses | 37,869 | | | 42,950 | | | (5,081) | |
| Loss from operations | (37,869) | | | (41,950) | | | 4,081 | |
| Non-operating income | 529 | | | 649 | | | (120) | |
| | | | | |
| | | | | |
| Net loss | $ | (37,340) | | | $ | (41,301) | | | 3,961 | |
Further analysis of the changes and trends in our operating results are discussed below.
LicenseLicensing Revenue
LicenseThere was no licensing revenue for the year ended December 31, 2020 was $1.92023 and $1.0 million and related to the out-licensing of intellectual property rights and transfer of technical know-how associated with the 3DMed Agreement for the development and commercialization of GPS in China, Hong Kong, Macau, and Taiwan. There was no licenselicensing revenue for the year ended December 31, 2019.2022 related to approval by China's National Medical Products Administration, or NMPA, of an Investigational New Drug, or IND, application filed by 3D Medicines for a small Phase 1 clinical trial investigating safety of GPS in China.
Cost of Licensing Revenue
There was no cost of licensing revenue recognized during the year ended December 31, 2023, and $0.1 million of sublicensing fees payable under our license from MSK in connection with the 3D Medicines Agreement during the year ended December 31, 2022.
Research and Development
Research and development expenses were $9.3$24.0 million for the year ended December 31, 20202023 compared to $7.3$20.3 million for the year ended December 31, 2019.2022. As compared to the prior period, the $2.0$3.7 million increase in research and development expenses was primarily attributable to a $0.9$1.9 million increase in clinical trial expenses primarily duerelated to the initiation of our ongoing Phase 3 REGAL clinical trial of GPS in AML in 2020,and our Phase 2a and Phase 1 clinical trials of SLS009, a $0.8$1.3 million increase in manufacturing to support our ongoing clinical programs including initiating a technology transfer to a new drug product manufacturer, a $0.5 million increase in outsourced clinical and regulatory consulting services in supportexpenses due to the advancement of our ongoing clinical programs, and a $0.4$1.1 million increase in personnel related expenses primarily due to increased headcount, and a $0.2 million increase in other research and development expenses.headcount. These increases were partially offset by a $0.8$0.5 million decrease in manufacturing and clinical drug supply costs due to the timing of the manufacture of drug substance and drug product batches, and a $0.1 million decrease in licensing fees perand other research and development costs. We anticipate that our license agreements.research and development expenses will increase in the future as we continue to advance the development of GPS and SLS009, including our Phase 3 REGAL clinical trial of GPS in AML and the ongoing and planned clinical trials of SLS009.
General and Administrative
General and administrative expenses were $9.6$13.9 million for the year ended December 31, 20202023 compared to $9.9$12.6 million for the year ended December 31, 2019.2022. The $0.3$1.3 million decreaseincrease was primarily driven by a $1.0$0.5 million decrease in legal fees due to reduced litigation, a $0.4 million decreaseincrease in personnel related expenses primarily due to reducedincreased headcount, including a $0.3 million increase in non-cash stock-based compensation, a $0.6 million increase in intellectual property fees, a $0.1 million increase in outside services and $0.2public company costs, and a $0.1 million decreaseincrease in other general and administrative expenses. These decreases were partially offset by a $0.9 million increase in insurance premiums due to hardening insurance markets and a $0.4 million increase in outsourced professional services and public company costs.
Acquired In-Process Research and Development Impairment Charge
During the fourth quarter of 2019, we determined that the IPR&D asset associated with the GALE-301There was no acquired in-process research and 302 product candidates was impaired and recorded an impairment charge of approximately $2.8 million fordevelopment expense during the year ended December 31, 2019. Included in2023. During the asset impairment expenses are a non-cash charge of approximately $2.8year ended December 31, 2022, we recognized $10.0 million for impairmentthe acquisition of intangible assets recorded as IPR&Din-process research and an accelerationdevelopment related to the in-licensing of a de minimis amount of prepaid expenses and other current assets.SLS009.
Non-Operating Income Net
Non-operating income (expense), net for the years ended December 31, 20202023 and 2019, respectively,2022 was as follows (dollars in(in thousands):
| | | Years Ended December 31, |
| | 2020 | | 2019 | | Change |
| | | Year ended December 31, | |
| | | Year ended December 31, | |
| | | Year ended December 31, | |
| | | | | | | 2023 | | | | | | | | | 2023 | | 2022 | | Change |
| Change in fair value of warrant liability | Change in fair value of warrant liability | | $ | (97) | | | $ | 1,136 | | | $ | (1,233) | |
| Change in fair value of the contingent consideration | | 279 | | | (586) | | | 865 | |
| Change in fair value of contingent consideration | |
| Interest income | Interest income | | 26 | | | 118 | | | (92) | |
| Total non-operating income, net | | $ | 208 | | | $ | 668 | | | $ | (460) | |
| Total non-operating income | |
Our future operations are highly dependent on a combination of factors, including (i) the timely and successful completion of any additional financing,financings, (ii) our ability to complete revenue-generating partnerships with pharmaceutical and biotechnology companies, (iii) the success of our research and development activities, (iv) the development of competitive therapies by other biotechnology and pharmaceutical companies, and, ultimately, (v) regulatory approval and market acceptance of our proposed future products.product candidates.
The following table provides a reconciliation of the components of cash, cash equivalents, restricted cash, and restricted cash equivalents reported in our consolidated balance sheets to the total of the amount presented in the consolidated statements of cash flows (in thousands):