UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| (Mark One) | |
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the Fiscal Year Ended: December 31, 20162019
Or
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the transition period from to
Commission File Number: 001-35060

PACIRA PHARMACEUTICALS,BIOSCIENCES, INC.
(Exact Name of Registrant as Specified in its Charter)
| Delaware | 51-0619477 | |
(State or Other Jurisdiction of Incorporation or Organization) | (I.R.S. Employer Identification No.) | |
5 Sylvan Way, Suite 300 | ||
| Parsippany, | New Jersey | |
| 07054 | ||
| (Address and Zip Code of Principal Executive Offices) | ||
| (973) | 254-3560 | |
| (Registrant’s Telephone Number, Including Area Code) | ||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol | Name of each exchange on which registered | ||
| Common Stock, | PCRX | Nasdaq Global Select Market | ||
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes x No o
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports) and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” and “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer | ☒ | Accelerated filer | ☐ | |||
Non-accelerated filer | ☐ | Smaller reporting company | ☐ | |||
| Emerging growth company | ☐ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes o☐ No x
The aggregate market value of the Registrant’sregistrant’s voting stock held by non-affiliates of the registrant, based upon the closing sale price of the common stock as reported on the NASDAQNasdaq Global Select Market on June 30, 2016,28, 2019, the last businesstrading day of the registrant’s most recently completed second fiscal quarter, of $33.73$43.49 per share was $949approximately $848.3 million. Shares of common stock held by each director and executive officer (and their respective affiliates) and by each person who owns 10 percent or more of the outstanding common stock or who is otherwise believed by the registrant to be in a control position have been excluded. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of February 22, 2017, 37,525,10816, 2020, 42,033,039 shares of the registrant’s common stock, $0.001 par value per share, were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Part III of this Annual Report on Form 10-K incorporates certain information by reference from the registrant’s proxy statement for the 20172020 annual meeting of stockholders to be filed no later than 120 days after the end of the registrant’s fiscal year ended December 31, 2016.2019.
i
ANNUAL REPORT ON FORM 10-K
FOR THE YEAR ENDED DECEMBER 31, 2019
TABLE OF CONTENTS
| Page | ||
Forward-Looking Statements
This Annual Report on Form 10-K (the “Annual Report”) and certain other communications made by us contain forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), including statements about our growth and future operating results, discovery and development of products, strategic alliances and intellectual property. For this purpose, any statement that is not a statement of historical fact should be considered a forward-looking statement. We often use the words “believe,” “anticipate,” “plan,” “estimate,” “expect,” “intend,” “may”“may,” “will,” “would,” “could,” “can” and similar expressions to help identify forward-looking statements. We cannot assure you that our estimates, assumptions and expectations will prove to have been correct. These forward-looking statements include, among others, statements about: the success of our sales and manufacturing efforts in support of the commercialization of EXPAREL®EXPAREL® (bupivacaine liposome injectable suspension) and our other products;; the rate and degree of market acceptance of EXPAREL; the size and growth of the potential markets for EXPAREL and our ability to serve those markets; the Company’sour plans to expand the use of EXPAREL to additional indications and opportunities, and the timing and success of any related clinical trials; our ability to realize the anticipated benefits and synergies from the acquisition of MyoScience, Inc., or MyoScience; the ability to successfully integrate iovera°® and MyoScience into the Company’s existing business; the commercial success of iovera°; the related timing and success of United States Food and Drug Administration, or FDA, supplemental New Drug Applications;Applications, or sNDAs, and premarket notification 510(k)s; the outcome of the U.S. Department of Justice, or DOJ, inquiry; the Company’s plans to evaluate, develop and pursue additional DepoFoam®DepoFoam®-based product candidates; clinical trials in support of an existing or potential DepoFoam-based product; the Company’s plans to continue to manufacture and provide support services for its commercial partners who have licensed DepoCyt(e); our commercialization and marketing capabilities and the Company’s and Patheon UK Limited’sour ability to successfully and timely construct an additional EXPAREL manufacturing suites.suite in Swindon, England and assumptions associated with contingent consideration payments. Important factors could cause our actual results to differ materially from those indicated or implied by forward-looking statements, including those discussed below in Part I-Item 1A. Risk Factors.statements. We undertake no intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, and readers should not rely on the forward-looking statements as representing our views as of any date subsequent to the date of the filing of this Annual Report on Form 10-K.Report.
These forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause the Company’sour actual results, levels of activity, performance or achievements to differ materially from those expressed or implied by these statements. These factors include the matters discussed and referenced in Part I-Item 1A. Risk Factors.
PART I
Item 1. Business
References
Pacira Pharmaceuticals,BioSciences, Inc., a Delaware corporation, changed its name from Pacira Pharmaceuticals, Inc. upon completing its acquisition of MyoScience, a privately-held medical technology company, in April 2019 (referred to herein as the “MyoScience Acquisition”) in order to better reflect a broadening portfolio of innovative non-opioid pain management and regenerative health solutions. Pacira BioSciences, Inc. is the holding company for our California operating subsidiary of the same name,named Pacira Pharmaceuticals, Inc., or Pacira California. In March 2007, we acquired Pacira California from SkyePharma Holdings, Inc. (now a subsidiary of Vectura Group plc), or Skyepharma (referred to in this Annual Report on Form 10-Kherein as the “Acquisition”“Skyepharma Acquisition”). Pacira California retained the name Pacira Pharmaceuticals, Inc. upon the completion of the MyoScience Acquisition. Unless the context requires otherwise, references to “Pacira,” “we,” the “Company,” “us” and “our” in this Annual Report on Form 10-K refers to Pacira Pharmaceuticals,BioSciences, Inc., a Delaware corporation, and its subsidiaries.
Corporate Information
We were incorporated in Delaware under the name Blue Acquisition Corp. in December 2006 and changed our name to Pacira, Inc. in June 2007. In October 2010, we changed our name to Pacira Pharmaceuticals, Inc. and in April 2019 changed our name to Pacira BioSciences, Inc. upon the completion of the MyoScience Acquisition. Our principal executive offices are located in Parsippany, New Jersey.
Pacira®, EXPAREL®, iovera°®, DepoFoam®, DepoCyt® (United States (US)(U.S.) registration), DepoCyte® (European Union (EU)(E.U.) registration), DepoTXA®, the Pacira logo and other trademarks or service marks of Pacira appearing in this Annual Report on Form 10-K are the property of Pacira. In addition, references in this Annual Report on Form 10-K to DepoCyt(e) mean DepoCyt when discussed in the context of the United StatesU.S. and Canada and DepoCyte when discussed in the context of the EU.E.U.
This Annual Report on Form 10-K contains additional trade names, trademarks and service marks of other companies.
Overview
We are a specialty pharmaceutical companyleading provider of non-opioid pain management options to advance and improve outcomes for health care practitioners and their patients, focused on becoming a global leader in delivering innovative non-opioid pain management and
regenerative health solutions to surgeons and anesthesiologists. Our corporate mission is to provide an opioid alternative to as many appropriate patients as possible. To that end, we are advancing a three-part growth strategy focusing on: (i) expanding the development, manufacture and commercializationuse of pharmaceutical products, based on our proprietary DepoFoam extended release drug delivery technology, for use primarily in hospitals and ambulatory surgery centers. We operate in one reportable segment. On October 28, 2011, the US Food and Drug Administration, or FDA, approved our New Drug Application, or NDA, for EXPAREL (bupivacaine liposome injectable suspension).and iovera°, our non-opioid pain therapies; (ii) pursuing innovative opioid-sparing options through in-licensing and acquisition; and (iii) advancing a pipeline of non-opioid opportunities for acute and chronic pain management.
In April 2019, we completed the MyoScience Acquisition and added the iovera° system to our commercial offering. The iovera° system is a non-opioid handheld cryoanalgesia device used to alleviate pain.
EXPAREL was approved by the FDA in October 2011 and was commercially launched in April 2012. EXPAREL is currently indicated for single-dose infiltration in adults to produce postsurgical local analgesia and as an interscalene brachial plexus nerve block to produce postsurgical regional analgesia. Since its initial approval in 2011 for single-dose infiltration, more than six million patients have been treated with EXPAREL. EXPAREL consists of bupivacaine, an amide-type local anesthetic, encapsulated in DepoFoam, and is indicatedour proprietary extended release drug delivery technology, that delivers bupivacaine over time for single-dose infiltration into the surgical site to produce postsurgicalextended analgesia. We believe that EXPAREL addresses a significant medical need for a long-acting non-opioid postsurgical analgesic resulting in simplified postsurgical pain management and reduced opioid consumption, and has the potential to result in improved patient outcomes and enhanced hospital economics. We have developed an internal sales force entirely dedicated to commercializing EXPAREL, which we commercially launched in the United States in April 2012. Our net sales for EXPAREL in 2016 were $265.8 million. We believe EXPAREL is playingplays a significant role in opioid minimization strategies.
EXPAREL is designed for recovery with minimal opioid use by (i) delivering targeted local analgesia at the surgical site; (ii) reliably releasing bupivacaine over time for prolonged analgesia; (iii) eliminating the need for catheters and pumps that may hinder recovery and (iv) providing long-lasting pain control while reducing the need for opioids. Our net product sales of EXPAREL in 2019 were $407.9 million. For the years ended December 31, 2019, 2018 and 2017, net product sales of EXPAREL accounted for 97%, 98% and 99% of our total revenues, respectively. In addition to EXPAREL, DepoFoam is also the basis for our otherfuture clinical candidates.
The iovera° system is an FDA-approved commercial product, DepoCyt(e),medical device used to deliver precise, controlled doses of cold temperature only to targeted nerves, which we manufacturehas been FDA 510(k) cleared for our commercial partners,use in pain applications since March 2014. The iovera° system is highly complementary to EXPAREL as well as our product candidates.a non-opioid therapy that alleviates pain by disrupting pain signals being transmitted to the brain from the site of injury or surgery. For the yearsyear ended December 31, 2016, 2015 and 2014,2019, net product sales of EXPAREL accounted for 96%, 96% and 95%iovera° were $7.9 million. We began recognizing sales of our total revenues, respectively.iovera° after completing the MyoScience Acquisition in April 2019.
Our current product portfolio and product candidate pipeline, along with expectedanticipated milestones over the next 12 to 18 months, are summarized in the table below:
| PROPRIETARY PIPELINE | ||||
| Product / Product Candidates | Status | Next Expected Milestone | ||
EXPAREL (bupivacaine liposome injectable suspension): | ||||
| Surgical infiltration | Approved | |||
| | Approved (U.S.) | Publish results / geographic expansion | ||
C-section TAP field block 1 | Phase 4 | |||
C-section TAP field block follow-on study 1 | Phase 4 | Publish results in peer-reviewed journal | ||
| Spine | Phase 4 | |||
| | ||||
| | ||||
| | ||||
| | Phase 3 | |||
| | ||||
| Phase 3 | ||||
| | ||||
| Approved | Initiate studies | |||
| Blocking of pain | Approved | Initiate studies | ||
| DepoFoam-based local anesthetic | Preclinical | Advance preclinical activities | ||
| DepoDexmedetomidine | Preclinical | Initiate clinical study | ||
| | Approved | |||
| Marketed by Aratana Therapeutics, Inc. | ||||
1 - Trial being conducted at MD Anderson Cancer Center under TAP block is a Pacira Granttransversus abdominis plane field block
2 - Formerly Sigma-Tau Rare Disease Ltd.
NOCITA® is a registered trademark of Aratana Therapeutics, Inc., a wholly owned subsidiary of Elanco Animal Health, Inc.
Our Strategy
We continue to advance our goal to be a global leader in delivering innovative non-opioid pain management and regenerative health solutions. To achieve this, we are advancing a three-pronged strategy:
| • | Expanding the use of EXPAREL and iovera° for opioid-sparing pain management: As the only opioid-free, long-acting local and regional analgesic approved for infiltration, field blocks and interscalene brachial plexus nerve block, we believe EXPAREL is well-positioned to continue delivering strong sustainable growth from multiple sources. We are seeing increased use within the anesthesiology community through EXPAREL-based regional approaches that are enabling the shift of complex, painful procedures to the hospital outpatient and ambulatory settings. Our partnership with DePuy Synthes Sales Inc., or DePuy Synthes, part of the Johnson & Johnson family of companies, has established the role of EXPAREL as the foundation of opioid-sparing protocols for painful orthopedic procedures, including shoulder, hip fracture, joint reconstruction, and spine surgeries. We are expanding the body of clinical evidence through Phase 4 studies in key surgical settings, such as C-section and spine. In addition, we are advancing clinical and regulatory activities to expand the EXPAREL label to include the pediatric and lower extremity nerve block settings. For iovera°, we are focusing on iovera° plus EXPAREL as a multimodal procedural solution for total knee arthroplasty, or TKA, as well as drug-free, opioid-free, surgery-free pain management for osteoarthritis of the knee. |
| • | Pursuing innovative acquisition targets that align with our strategy: We believe EXPAREL, iovera° and the DepoFoam platform offer a strong foundation to address the opioid epidemic. Building on these company assets, we are also pursuing innovative acquisition targets ranging from devices, therapeutics, cell therapies and regenerative |
medicines. Our goal is to bebuild a leading specialty pharmaceutical company focused onportfolio of customer-focused non-opioid pain and regenerative health solutions to improve patients’ journeys along the development, manufacture and commercialization of proprietary pharmaceutical products primarily for use in hospitals and ambulatory surgery centers. We plan to achieve this by:neural pain pathway.
| • | Advancing a pipeline of new clinical candidates: We are developing a pipeline based on our DepoFoam platform, our established safe and effective multivesicular liposomal drug delivery technology. DepoFoam consists of microscopic, spherical, lipid-based particles composed of a honeycomb of numerous, non-concentric, internal aqueous chambers containing the encapsulated drug. DepoFoam provides flexible delivery and can be designed to offer an immediate release dose followed by sustained delivery. We are advancing a program for the intrathecal delivery of a DepoFoam-based local anesthetic as a potential alternative to the use of subarachnoid opioids delivered by pumps and catheters and DepoDexmedetomidine for end-of-life pain and painful conditions in the elderly. |
EXPAREL
Opioid addiction in the United States for postsurgical analgesia by infiltration;
| • | Expanding the use of EXPAREL in key surgical settings. We are expanding the clinical evidence for EXPAREL through Phase 4 clinical trials across several surgical specialties. We have published positive results from a Phase 4 multicenter, randomized, controlled trial, or RCT, in TKA in the Journal of Arthroplasty. Positive findings from a multicenter RCT in C-section were presented at the most recent annual meeting of the Society for Obstetric Anesthesia and Perinatology (SOAP), and we recently reported positive topline results from a follow-on Phase 4 study in C-section procedures that compared an opioid-free EXPAREL arm to an opioid-based standard of care arm. EXPAREL is being incorporated into an increasing number of Enhanced Recovery After Surgery, or ERAS, protocols from major academic centers for a wide range of procedures. In addition, we are advancing clinical and regulatory activities to support the future expansion of EXPAREL to the pediatric and lower extremity nerve block settings. Regulatory initiatives are also advancing in new global markets, such as Europe, Canada and China. |
| • | Expanding access to EXPAREL and driving education and awareness around the need for opioid-sparing strategies. New payer policies and benefits are supporting this migration to realize cost savings while enhancing patient care through the use of opioid-sparing protocols. The Centers for Medicare and Medicaid Services, or CMS, is providing Medicare reimbursement for EXPAREL when administered in an Ambulatory Surgery Center, or ASC, through the product-specific Healthcare Common Procedure Coding System (HCPCS) code of C9290. Effective January 1, 2020, CMS removed total hip arthroplasty and six spinal procedures from its inpatient-only list, making these procedures eligible for payment by Medicare in the hospital outpatient setting. The final CMS rule for 2020 also added TKA and several other procedures to its listing of ASC covered procedures. In addition, we continue to advance our Choices Matter national educational campaign, aimed at empowering patients to proactively discuss postsurgical pain management, including non-opioid options, with their healthcare providers. |
| • | Partnering with those who share our commitment to innovative opioid-sparing procedural solutions. We have a growing network of strategic collaborations to expand education on the importance of non-opioid multimodal alternatives for post-surgical pain management and broaden our commercial reach. These include agreements with industry partners, as well as healthcare providers and hospital systems to support their implementation of opioid-sparing enhanced recovery protocols. In January 2017, we formed a partnership with DePuy Synthes to support the promotion, education and training of EXPAREL in orthopedics. We are collaborating on national and regional training initiatives with large anesthesia physician practices, such as MEDNAX, Inc. and Envision Physician Services. Our growing coalition of collaborators also includes Aetna, the American Association of Oral and Maxillofacial Surgeons, or AAOMS, the American College of Surgeons, the American Society for Enhanced Recovery, Cancer Treatment Centers of America, the Illinois Surgical Quality Improvement Collaborative, WellStar Health System and Shatterproof.org. |
EXPAREL Clinical Benefits
We believe EXPAREL can replace the use of bupivacaine via elastomeric pumps as the foundation of a multimodal regimen for long-acting postsurgical pain management. Based on our clinical data, EXPAREL:
provides long-lasting postsurgicallocal or regional analgesia;
is a ready-to-use formulation;
expands easily with saline or lactated Ringer’s solution to reach a desired volume;
leverages existing postsurgicalinterscalene brachial plexus nerve block, field block and infiltration administration techniques; and
facilitates treatment of both small and large surgical sites.
We believe EXPAREL can become the foundationis a key component of a long-acting postsurgical pain management regimen in order toregimens that reduce and delay opioid usage.the need for opioids. Based on the clinical data from our Phase 3 hemorrhoidectomy trialand Phase 4 clinical studies as well as ourdata from retrospective health outcomes studies, data, EXPAREL significantly delays and reduces opioid usage while improving postsurgical pain management.
In our Phase 3 hemorrhoidectomy trial, EXPAREL:
delayed the median time to rescue analgesic use (opioids) to 15 hours for patients treated with EXPAREL andversus one hour for patients treated with placebo;
significantly increased the percentage of patients requiring no opioid rescue medication through 72 hours post-surgery to 28%, compared to 10% for placebo;
resulted in 45% less opioid usage through 72 hours post-surgery compared to placebo; and
increased the percentage of patients who arewere pain free at 24 hours post-surgery compared to placebo.
In our Phase 3 trial as an interscalene brachial plexus nerve block for upper extremity surgeries, EXPAREL:
decreased total opioid consumption by 78% (p<0.0001) from zero to 48 hours after surgery;
reduced pain scores by 46% versus placebo (p<0.0001); and
allowed 13% of patients who received EXPAREL to remain opioid-free for 48 hours after surgery (p<0.01) compared to zero patients in the placebo arm.
In our Phase 4 trial of EXPAREL versus bupivacaine HCl in TKA, EXPAREL:
decreased total opioid consumption by 78% (p=0.0048) from zero to 48 hours after surgery;
reduced pain scores by 14% (p=0.0381) from 12 to 48 hours after surgery; and
allowed 10% of patients in the EXPAREL arm to remain opioid-free through 48 and 72 hours (compared to zero patients in the bupivacaine arm; p<0.01).
In our Phase 4 trial of EXPAREL versus bupivacaine administered via a transversus abdominis plane block in C-section procedures:
Comparable pain control with a reduction in total opioid consumption with EXPAREL plus bupivacaine HCl versus bupivacaine HCl:
| ◦ | 52% reduction through 72 hours, (p=0.0117) |
| ◦ | 49% reduction at one week (p=0.0175) |
| ◦ | 41% reduction at two weeks (trending toward significance; p=0.0542) |
More than a two-fold increase in the percentage of opioid-spared patients with EXPAREL versus bupivacaine HCl (defined as patients who took no more than one oxycodone 10mg tablet, or equivalent, with no opioid-related side effects through 72 hours; p=0.0012)
EXPAREL can improve patient satisfaction and outcomes. We believe EXPAREL:
provides effective pain control without the need for expensive and difficult-to-use delivery technologies that extend the duration of action for bupivacaine, such as elastomeric bags, or opioids administered through patient-controlled analgesia, or PCA, when used as part of a multimodal postsurgical pain regimen;
reduces the need for patients to be constrained by elastomeric bags and PCA systems, which are barriers to earlier ambulation and may introduce catheter-related issues, including infection; and
promotes maintenance of early postsurgical pain management, which may reduce the time spent in the intensive care unit.
EXPAREL Health Economic Benefits
In addition to being efficacious and safe, we believe that EXPAREL provides health economic benefits that play an important role in formulary decision-making and that these health economic benefits are an often overlooked factor in planning for the commercial success of a pharmaceutical product.overlooked. Several members of our management team have extensive experience applying health economic outcomes research to support the successful launch of commercial products.success. Our strategy is to work directly with the senior leadership of our hospital customers, group purchasing organizations, integrated health networks, quality improvement organizations, key opinion leaders, or KOLs, in the field of postsurgical pain management and leading influencer hospitals to provide them with retrospective and prospective studies to demonstrate the economic benefits of EXPAREL.
Retrospective chart review data were captured for 645 consecutive Medicare patients who underwent primary inpatient TKA (337 patients) or THA (308 patients) between June 1, 2015, and November 16, 2017. All patients followed a procedure-specific ERAS protocol which included EXPAREL (Van Horne A, Van Horne J. Patient-optimizing enhanced recovery pathways for total knee and hip arthroplasty in conjunctionMedicare patients: implication for transition to ambulatory surgery centers. Arthroplasty Today 2019). Key findings included:
84% did not require any additional opioid prescriptions beyond the initial seven-day prescription provided at discharge (while nationally, 38% of knee replacement patients are still taking opioids two months after surgery)
84% of patients were same-day discharged to home, following their joint replacement
Patients reported high satisfaction with hospital customer groups utilizing their own hospital databases, revealed thatperioperative experience (98% were highly satisfied with their pain management)
Comparable or lower complication rates to nationally reported rates
In June 2019, we reported new data on the use of opioids for postsurgical pain control isEXPAREL following THA. The findings show that patients receiving EXPAREL had a significant driverreduction in opioid use, hospital length of hospital resource consumption, including higherstay, or LOS, and total hospitalization costs longercompared to THA patients who did not receive the product. The results were published in The Journal of Medical Economics.
This retrospective analysis utilized data from the Premier Healthcare Database from January 2011 through April 2017 for the ten hospitals in the U.S. with the highest number of THA procedures using EXPAREL. Patients undergoing THA who received EXPAREL were matched in a one-to-one ratio to a control group of patients whose pain management strategy did not include the product. The study population included a total of 12,589 patients, with 7,232 Medicare patients and 5,357 commercial insurance patients.
Results showed that patients undergoing THA who received EXPAREL compared to those who did not demonstrated a significant:
Decrease in opioid consumption, expressed in oral morphine equivalent dosing (MED), among Medicare and commercial insurance patients (105 mg MED and 81 mg MED reductions, respectively; p<0.0001)
Decrease in average hospital LOS by 0.7 days in both the Medicare and commercial insurance groups (p<0.0001)
Decrease in total hospitalization costs in the Medicare population (-$561; p<0.0001)
Increase in likelihood to be discharged home in both the Medicare and commercial insurance groups (1.66 and 1.57, respectively; p<0.0001)
Results of this study are consistent with findings from several retrospective studies and RCTs on the use of EXPAREL for total joint replacement procedures, including additional data published in 2018 in The Journal of Medical Economics that found a decrease in opioid consumption, hospital LOS and hospitalization costs for patients receiving EXPAREL following TKA (Asche et al. Impact of treatment with liposomal bupivacaine on hospital costs, length of stay, and discharge status in patients undergoing total knee arthroplasty at high-use institutions, Journal of Medical Economics, DOI: 10.1080/13696998.2018.1543190).
Third Molar Procedures
In September 2017, we announced a collaboration with Aetna, one of the nation’s leading diversified health care benefits companies, with the support of AAOMS. This national program aims to reduce the number of opioid tablets dispensed to patients undergoing impacted third molar (wisdom tooth) extractions by at least 50 percent through the utilization of EXPAREL to provide prolonged non-opioid postsurgical pain control. Aetna now includes the cost of EXPAREL as a covered expense for impacted third molar extractions performed by surgeons who have completed training on use of the product.
According to a Journal of the American Medical Association (JAMA) study, more than two-thirds of patients who underwent surgical tooth extractions reported unused prescription opioids, with the majority also indicating that these medications are neither safely stored nor disposed of. These facts suggest that there is a dangerous accumulation of opioids in the home, which are available for potential diversion or misuse.
In June 2019, investigators reported results from a study confirming that patients who received EXPAREL were prescribed significantly fewer opioids compared to those who did not. In this study, researchers reviewed data from 600 patients who underwent impacted third molar extractions between 2012 and 2018 in two dental clinics; one in Connecticut and one in North Carolina. Data from 300 patients who received EXPAREL were compared to data from 300 patients who did not receive EXPAREL. Patients in the EXPAREL treatment group received:
Fewer total prescribed opioid tablets including refills, compared to patients in the non-EXPAREL group (6.4 tablets vs. 15.5 tablets, respectively; p<0.0001)
Fewer additional opioid prescriptions compared to the non-EXPAREL group (3.3% of patients required a refill vs. 7.7% of patients, respectively)
The research was presented at the June 2019 International Association for opioid-related adverse events.Dental Research meeting in Vancouver, Canada and highlighted in the Aetna Fall 2019 Dental Dialog newsletter.
EXPAREL Dosing, Volume Expansion and Admixing with Bupivacaine HCl
EXPAREL is available as a 266 mg/20 mL single-use vial and a 133 mg/10 mL single-use vial. The recommended dose of EXPAREL is based on (i) the size of the surgical site; (ii) the volume needed to cover the width and depth of the surgical site and (iii) patient-specific factors that could impact safety of an amide-type local anesthetic. The maximum dose should not exceed 266 mg.
EXPAREL can be expanded in volume to optimize results. Physicians consider the size of the surgical site and neuroanatomy to determine dosing and volume expansion. The 266 mg (20 mL) EXPAREL vial can be expanded with up to 280 mL of normal (0.9%) saline or lactated Ringer’s solution for a total volume of 300 mL (a 1:14 ratio). For smaller surgical sites where 20 mL is too much volume, the 133 mg (10 mL) vial should be considered.
To ensure early analgesic activity, EXPAREL can be admixed with bupivacaine HCl so long as the ratio does not exceed 1:2. For example, the 266 mg/20mL vial may be administered with up to 30 mL of 0.5% bupivacaine HCl or up to 60 mL of 0.25% bupivacaine HCl. Bupivacaine HCl may be administered immediately before EXPAREL or admixed in the same syringe.
EXPAREL Label Expansion Studies
Pediatrics
In December 2019, we reported positive topline results from our Phase 3 registration study (known as “PLAY”) of EXPAREL administered as a single-dose infiltration in pediatric patients undergoing spinal or cardiac surgeries. Overall findings were consistent with the pharmacokinetic and safety profiles for adult patients with no safety concerns identified at a dose of 4 mg/kg. We believe the results from this study will provide the foundation for an sNDA submission in the first half of 2020 to the FDA seeking expansion of the EXPAREL label to include children aged six and over. We are pursuing additional indicationsalso working with the FDA to finalize a regulatory pathway to expand the EXPAREL label for EXPAREL. Our most advanced development program is evaluatingto include EXPAREL for nerve block. Nerve block is a general term used to refer to the injection of local anesthetic onto or near nerves for pain control. Traditionally, nerve blocks are single injections of short-acting anesthetics andadministered as a result, have a limited duration of action. When extended pain management is required, a catheter is used to deliver bupivacaine continuously using an external pump. EXPAREL is designed to provide extended pain management using a single injection.
The FDA, as a conditionPLAY study enrolled 98 patients to evaluate the pharmacokinetics and safety of EXPAREL approval, has required usfor two patient groups: patients aged 12 to study EXPAREL in pediatric patients. We were granted a deferral for the required pediatric trials in all age groups for EXPAREL in the setting of wound infiltrationless than 17 years and planpatients aged 6 to conduct these pediatric trials as a post-marketing requirement, which was stated in the NDA approval letter for EXPAREL. We recently secured feedback fromless than 12 years. In agreement with the FDA, on the pediatric trial design in all age groupsprimary and we are in the process of finalizing our clinical strategy.
Nerve Block in Lower Extremity Surgery
We have initiated a Phase 3 study for nerve block in lower extremity surgeries (known as “STRIDE”) that is comparing an EXPAREL nerve block in lower extremity surgeries to a bupivacaine lower extremity nerve block in patients undergoing foot and ankle surgeries. We believe positive results from this study would support an sNDA submission seeking label expansion to include lower extremity nerve blocks. We believe the addition of this indication is significant as anesthesia-driven regional approaches using nerve and field blocks continue to expand as institutional protocols.
Global Expansion
We have defined a global expansion strategy for EXPAREL that we believe provides us with the opportunity to increase our revenue and leverage our fixed cost infrastructure. We have prioritized Europe, Canada and China. In Europe, we have secured a positive opinion for our Pediatric Investigation Plan (PIP) and in June 2019 our Marketing Authorization Application, or MAA, was validated by the European Medicines Agency, or EMA. In Canada, which is a concentrated market driven by four provinces, Health Canada has validated our New Drug Submission. We do not intend to pursue a commercial partnership to commercialize EXPAREL in Europe or Canada. In China, we have an agreement with Nuance Biotech Co. Ltd., or Nuance, a China-based specialty pharmaceutical company, for the development and commercialization of EXPAREL. We have received feedback from the National Medical Products Administration, or NMPA, in China and we are preparing for a meeting with the NMPA in 2020 to finalize our regulatory path forward.
The iovera° system
The iovera° system is highly complementary to EXPAREL as a non-opioid therapy that delivers cryoanalgesia via a handheld device to alleviate pain by disrupting pain signals being transmitted to the brain from the site of injury or surgery. Initially, we will focus on two broad patient care opportunities. The ioveraº system is 510(k) cleared in the U.S. for the blocking of pain, pain relief and symptoms associated with osteoarthritis of the knee as well as general surgical use.
Our first priority is iovera° and EXPAREL for opioid-sparing pain management for the TKA patient, with iovera° being administered before surgery and EXPAREL administered during surgery. As many as 30 percent of presurgical patients with end-stage knee osteoarthritis use prescription opioids. With iovera°, our goal is to provide patients with several months of non-opioid pain control to allow them to prepare for surgery with an appropriate regimen. We are advancing a Phase 4 multicenter, randomized, double-blind, controlled trial ofalso believe that EXPAREL for surgical pain control and EXPAREL plus iovera° for postsurgical pain control could support rapid functional recovery.
The second target market is iovera° for osteoarthritis patients who have failed conservative treatments, such as non-steroidal anti-inflammatory drugs or viscosupplementation, and are seeking drug-free, opioid-free, surgery-free pain management infor several months. We are targeting patients undergoing open lumbar spinal fusion surgery. Patients will be randomized to receive local infiltration analgesia with EXPAREL admixed with bupivacaine and expanded in volume or local infiltration analgesia with bupivacaine expanded in volume. The primary objective of this trial is to compare postsurgical pain control and the secondary objectives will compare additional efficacy, safety and health economic outcomes. We expect to report top-line data from this trial in the second half of 2017.
Osteoarthritis of the Knee
There is a growing body of clinical data demonstrating success with the iovera° treatment for osteoarthritis of the knee. There are 14 million individuals in the U.S. who have symptomatic knee osteoarthritis, and nearly two million are under the age of 45. Surgical intervention is typically a last resort for patients suffering from osteoarthritis of the knee. In one study, the majority of the patients suffering from osteoarthritis of the knee experienced pain relief beyond 150 days after being treated with iovera°.
Preliminary findings demonstrated reductions in opioids, including:
The daily morphine equivalent was significantly lower at 72 hours (p<0.05), 6 weeks (p<0.05) and 12 weeks (p<0.05), with an overall 35 percent reduction in daily morphine equivalents across the 12-week postoperative period in the iovera° treatment group.
Patients who were administered iovera° were far less likely to take opioids six weeks after surgery. The number of patients taking opioids six weeks after TKA in the control group was three times the number of patients taking opioids in the cryoanalgesia group (14% vs. 44%, p<0.01).
Patients in the iovera° group demonstrated a statistically significant reduction in pain scores from their baseline pain scores at 72 hours (p<0.05) and at 12 weeks (p<0.05).
We believe these data validate iovera° as a clinically meaningful non-opioid alternative for patients undergoing TKA, and that iovera° offers the opportunity to provide patients with non-opioid pain control well in advance of any necessary surgical intervention through a number of additional efficacy, safetykey product attributes:
iovera° is safe and health economic outcomes.effective with immediate pain relief that can last for several months as the nerve regenerates over time;
iovera° is repeatable;
iovera° can be controlled with conventional cytarabine, but because of the drug’s short half-life, a spinal injection is required twice per week, whereas DepoCyt(e) is dosed once every two weeks indelivered precisely using ultrasound guidance or an outpatient setting. DepoCyt(e) was granted accelerated approval by the FDA in 1999 and full approval in 2007. We recognized revenue from DepoCyt(e) of $7.2 million from our commercial partners in 2016.
We believe the DepoFoam formulation provides several technical, regulatorycombination of iovera° and commercial advantages over competitive technologies, including:
Product Pipeline
Given the needproven safety, flexibility and customizability of our DepoFoam platform for post-operative drains, thereby increasing vigoracute, sub-acute and chronic pain applications, we have several DepoFoam-based products in patients while decreasing overall costspreclinical development. Following data readouts from animal and other feasibility studies for these candidates, we have prioritized two programs for clinical development: (i) the intrathecal delivery of a DepoFoam-based analgesic for acute and chronic pain and (ii) DepoDexmedetomidine, a sedative-analgesic for end-of-life pain and painful conditions in the elderly.
We plan to invest in clinical initiatives to broaden the scope of iovera° applications and improve its functionality for current and future end users. This will be accomplished through enhancements across the product line, which is comprised of single-use disposable units as well as non-disposable handheld devices.
In parallel, our business development team continues to pursue innovative acquisition targets that align with our strategy and are complementary to EXPAREL and iovera° by thoughtfully pursuing additional opportunities that are of great interest to the hospital system.
Sales and Marketing
We have built our marketing and sales organization to commercialize EXPAREL and our product candidates in the US. We intend to out-license commercialization rights for other territories.products. Our goal is to retain significant control over the development process and commercial execution for our product candidates, while participating in a meaningful way in the economics of all products that we bring to the market. The primary target audience for EXPAREL isaudiences are healthcare practitioners who influence pain management decisions including anesthesiologists, surgeons, anesthesiologists, pharmacists and nurses.
Our field team, consisting of both sales representatives, andaccount managers, scientific and medical affairs personnel and reimbursement and market access professionals, executes on a full range of activities to broaden the use of our non-opioid products for EXPAREL,pain management, including:
providing publications and abstracts showing the EXPAREL clinical program efficacy and safety, health outcomes program and review articles on pain management;articles;
working in tandem with hospital staff, such as registered nurses,anesthesiologists, surgeons, heads of quality, pharmacists, executives and executives,registered nurses, to provide access and resources for drug utilization or medication use evaluations and health outcomes studies, which provide retrospective and prospective analyses for our hospital customers using their own hospital data to demonstrate the true cost of opioid-based postsurgical pain control;
working with KOLs and advisory boards to address topics of best practice techniques as well as guidelines and protocols for the use of EXPAREL,our products, meeting the educational and training needs of our physician, surgeon, anesthesiologist, pharmacist and registered nurse customers;
undertaking education initiatives such as center of excellence programs; preceptorship programs; opioid-sparing and ERAS pain protocols and predictive models for enhanced patient care; interactive discussion forums; patient education platforms leveraging public relations, advocacy partnerships and public affairs efforts where appropriate; web-based training and virtual launch programs; and
collaborating with surgeonshealthcare providers towards improving the knowledge and management of pain in surgical and osteoarthritis patients with a focus on opioid risk and non-opioid alternatives and engaging our field-based medical teams in system-wide partnerships to address the national opioid epidemic, with a goal of studying alternative postsurgical pain management options that focus on optimization and opioid alternative strategies.strategies; and
facilitating reimbursement and the shift of procedures to hospital outpatient and ASC sites of care.
DePuy Synthes Sales Inc.
In January 2017, we entered into a co-promotion agreement with DePuy Synthes, Sales Inc., or DePuy Synthes,part of the Johnson & Johnson family of companies, to market and promote the use of EXPAREL for orthopedic procedures in the USU.S. market. Through this collaboration, we believe we can accelerate thehave accelerated EXPAREL growth strategy by quickly leveraging the broad reach of DePuy Synthes and their established relationships and scale within hospitals and ambulatory surgery centers.ASCs.
DePuy Synthes field representatives, specializing in joint reconstruction, spine, sports medicine, trauma and trauma, willcranio-maxillofacial (CMF) procedures, collaborate with, and supplement, our field teams by expanding the reach and frequency of EXPAREL education in the hospital surgical suite and ambulatory surgery center settings. DePuy Synthes willis also includeincluding EXPAREL in their Orthopedic Episode of Care Approach for health systems and surgeons.surgeons, and is including EXPAREL in all of their professional education programs. In addition to supporting DePuy Synthes,orthopedic specialties, we will focusare focusing on soft tissue surgeons in key specialties and anesthesiologists and we continue to act as the overall EXPAREL account manager.
We will also work with DePuy Synthes to develop ERAS protocols to improve procedure-specific patient care and to then rapidly communicate opportunities to utilize EXPAREL-based multimodal pain strategies to minimize opioids and improve patient satisfaction and hospital economics.
DePuy Synthes is entitled toreceives commissions on sales of EXPAREL under the agreement, subject to conditions, limitations and adjustments. The initial term of the agreement commencesbegan on January 24, 2017 and ends on December 31, 2021, with the option to extend the agreement in 12 month12-month increments upon the parties’ mutual agreement, of the parties, subject to certain conditions.
We and DePuy Synthes have mutual termination rights under the agreement, subject to certain terms, conditions, and advance notice requirements;notice; provided that we or DePuy Synthes generallyneither party may not terminate the agreement, without cause, within three years of the effective date of the agreement. We also have additional unilateral termination rights under certain circumstances. The agreement contains customary representations, warranties, covenants and confidentiality provisions, and also contains mutual indemnification obligations. DePuy Synthes is also subject to certain obligations and restrictions, including required compliance with certain laws and regulations and our policies, in connection with fulfilling their obligations under the agreement.
Other Agreements
MyoScience Acquisition
In April 2019, we completed the MyoScience Acquisition. The consideration included an initial cash payment of $120.0 million, reduced by $1.0 million for post-closing purchase price adjustments and indemnification obligations incurred to date, plus contingent milestone payments up to an aggregate of $100.0 million. Upon the completion of the MyoScience Acquisition, we renamed MyoScience to Pacira CryoTech, Inc. For more information on the MyoScience Acquisition, refer to Note 5, MyoScience Acquisition, to our consolidated financial statements included herein.
TELA Bio, Inc.
In October 2017, we made an investment of $15.0 million in TELA Bio, Inc., or TELA Bio, a surgical reconstruction company that markets its proprietary OviTexTM portfolio of products for ventral hernia repair and abdominal wall reconstruction. OviTex Reinforced BioScaffolds (RBSs) are intended for use as a surgical mesh to reinforce and/or repair soft tissue where weakness exists. In 2019, we made an additional cash investment of $1.6 million in TELA Bio. During the year ended December 31, 2019, we received a non-cash stock dividend from our investment in the amount of $2.5 million and recognized a loss in the amount of $5.7 million, recognized in the other, net line in our consolidated statements of operations. For more information, refer to Note 12, Financial Instruments, to our consolidated financial statements included herein.
SkyePharma Holdings, Inc. (Now a Subsidiary of Vectura Group plc)
In connection with the stock purchase agreement related to the Skyepharma Acquisition, we agreed to certain earn-out and milestone payments. Milestone payments are based on a percentage of net sales of DepoBupivacaine products collected, including EXPAREL, and certain other yet-to-be-developed products as well asproducts. For purposes of meeting future potential milestone payments, for DepoBupivacaine products, including EXPARELannual net sales are measured on a rolling quarterly basis. The milestones are as follows:
$4.0 million upon the first commercial sale in the United States (met April 2012);
See Note 6, 9, Goodwill and Intangible Assets, to our consolidated financial statements included herein for further information related to the Skyepharma agreement.Acquisition.
Research Development Foundation
Pursuant to an agreement with one of our stockholders, the Research Development Foundation, or RDF, we are required to pay RDF a low single-digit royalty on the collection of revenues from our DepoFoam-based products for as long as certain patents assigned to us under the agreement remain valid. RDF has the right to terminate the agreement for an uncured material breach by us, in connection with our bankruptcy or insolvency or if we directly or indirectly oppose or dispute the validity of the assigned patent rights.
DepoCyt(e) was a sustained-release liposomal formulation of the chemotherapeutic agent cytarabine that utilized our DepoFoam technology. DepoCyt(e) was indicated for the intrathecal treatment of lymphomatous meningitis, a life-threatening complication of lymphoma, a cancer of the immune system. In December 2002,June 2017, we entered into a supplydiscontinued production of DepoCyt® (U.S. and distribution agreement with Enzon Pharmaceuticals Inc., or Enzon, regarding the promotionCanada) and distribution of DepoCyt. PursuantDepoCyte® (E.U.) due to persistent technical issues specific to the agreement, Enzon was appointed the exclusive distributor of DepoCyt in the United States and Canada for a ten-year term, with successive two year renewal periods. In January 2010, Sigma-Tau Rare Disease, Ltd., or Sigma-Tau, acquired the rights to sell DepoCyt from Enzon for the United States and Canada. In December 2016, Sigma-Tau changed their name to Leadiant Biosciences, Ltd., or Leadiant. We and Leadiant are currently operating under the terms of the agreement. Under the supply and distribution agreement, we supply unlabeled DepoCyt vials to Leadiant. Under these agreements, we receive a fixed payment for the sale of DepoCyt vials, as well as a royalty on their sales in the thirty percent range.
DepoCyt(e) manufacturing process.
Aratana Therapeutics, Inc.
In December 2012, we entered into an Exclusive License, Development and Commercialization Agreement and related Supply Agreement with Aratana Therapeutics, Inc., or Aratana. Under the agreements, we granted Aratana an exclusive royalty-bearing license, including the limited right to grant sublicenses, for the development and commercialization of our bupivacaine liposome injectable suspension product for animal health indications.use in animals. In August 2016, the FDA’s Center for Veterinary Medicine, or CVM, approved NOCITA® (bupivacaine liposome injectable suspension) as a local post-operative analgesia for cranial cruciate ligament surgery in dogs. In August 2018, the FDA’s CVM expanded the NOCITA label to include its use as a peripheral nerve block to provide regional postoperative analgesia following onychectomy in cats. In June 2019, the FDA’s CVM approved a 10mL vial size for NOCITA. Aratana began purchasing our bupivacaine liposome injectable suspension product in the third quarter of 2016.
In connection with its entry into the license agreement, we received a one-time payment of $1.0 million. In December 2013, we received a $0.5 million milestone payment under the agreement. In June 2016, we recorded $1.0 million in milestone revenue for Aratana’s filing of an FDA Administrative New Animal Drug Application, or ANADA, and in August 2016 recorded $1.0 million related to the FDA’s approval of the ANADA. We are eligible to receive up to an additional aggregate $40.0 million upon the achievement of commercial milestones. Aratana is required to pay us a tiered double-digit royalty on certain net sales made in the United States.U.S. If the product is approved by foreign regulatory agencies for sale outside of the United States,U.S., Aratana will be required to pay us a tiered double-digit royalty on such net sales. Royalty rates will be reduced by a certain percentage upon the entry of a generic competitor for animal health indications into a jurisdiction or if Aratana must pay royalties to third parties under certain circumstances.
Either party has the right to terminate the license agreement in connection with (i) an insolvency event involving the other party that is not discharged in a specified period of time; (ii) a material breach of the agreement by the other party that remains uncured for a specified cure period or (iii) the failure to achieve a minimum annual revenue as set forth in the agreement, all on specified notice. We may terminate the agreement in connection with (i) Aratana’s failure to pay any amounts due under the agreement; (ii) Aratana’s failure to achieve regulatory approval in a particular jurisdiction with respect to such jurisdiction or (iii) Aratana’s failure to achieve its first commercial sale within a certain amount of time on a country by country basis after receiving regulatory approval, all on specified notice. Aratana may terminate the license agreement (i) upon the entry of a generic competitor for animal health indications on a country by country basis or (ii) at any time on a country by country basis except with respect to the United StatesU.S. and any country in the European Union,E.U., all on specified notice. The parties may also terminate the license agreement by mutual consent. The license agreement will terminate automatically if we terminate the supply agreement. In the event that the license agreement is terminated, all rights to the product (on a jurisdiction by jurisdiction basis) will be terminated and returned to us.
Unless terminated earlier pursuant to its terms, the license agreement is effective until December 5, 2027,July 2033, after which Aratana has the option to extend the agreement for an additional five (5) yearfive-year term, subject to certain requirements.
NOCITA® is a registered trademark of Aratana Therapeutics, Inc.Aratana.
Nuance Biotech Co. Ltd.
In October 2013, we formedJune 2018, the Company entered into an agreement with Nuance, a five-year arrangement with CrossLink BioScience, LLC, or CrossLink, forChina-based specialty pharmaceutical company, to advance the promotiondevelopment and salecommercialization of EXPAREL pursuant toin China. Under the terms of a Master Distributor Agreement. On June 30, 2016, we provided notice to CrossLink electing to terminate the agreement, (as amended) effectivethe Company agreed to be the sole supplier of EXPAREL to Nuance and has granted Nuance the exclusive rights to develop and commercialize EXPAREL in China. The Company received an upfront payment of $3.0 million in July 2018 and is eligible to receive future milestone payments of up to $60.0 million that are triggered by filing for and securing regulatory approval(s) and annual sales in China exceeding certain levels. The Company is also entitled to tiered royalties as of September 30, 2016. In connection with the termination, a fee based on a percentage of earned performance-based fees is due to CrossLink. This fee of $7.1 million is payable to CrossLink quarterly over two years, and is recorded in selling, general and administrative expense in the consolidated statements of operations.
net sales.
Significant Customers
We had three customerswholesalers each comprising 10% or more of our total revenue for the year ended December 31, 2016:2019: Cardinal Health, Inc., McKesson Drug Company and AmerisourceBergen Health Corporation, which accounted for 32%34%, 28%29% and 26% of our revenues, respectively. These customers are wholesalers that process orders for EXPAREL under a drop-ship program. EXPAREL is delivered directly to end-users without the wholesalers ever taking physical possession of the product.
Manufacturing and Research Facilities
Internal Facilities
We manufacture EXPAREL and DepoCyt(e) in two manufacturing facilitiesat our facility in San Diego, California. These facilities areThis facility is designated as Building 1 and Building 6 and are located within two miles of each other on two separate and distinct sites.1. We also have a research and development facility, Building 2, which sits adjacent to Building 1, and a warehouse, Building 7, located within five miles of our manufacturing facilities. We refer to these fourthree buildings as the Science Center Campus, and together these fourthree buildings consist of approximately 172,000150,000 square feet. Our manufacturing facilities are inspected regularly and approved for pharmaceutical manufacturing by the FDA and the Environmental Protection Agency (EPA). They have been inspected and approved previously by the European Medicines Agency, or EMA, and the Medicines and Healthcare Products Regulatory Agency, or MHRA, but are no longer with the discontinuation of DepoCyt(e). Our iovera° facility in Fremont, California, consists of approximately 20,000 square feet of mixed-use manufacturing, research and the Environmental Protection Agency.development and office space. We also have a lease for our former DepoCyt(e) production facility in San Diego which is currently idle and expires in August 2020.
We purchase raw materials and components from third-party suppliers in order to manufacture EXPAREL.EXPAREL and iovera°. In most instances, alternative sources of supply are available, although switching to an alternative source would, in some instances, take time and could lead to delays in manufacturing our drugproduct candidates. We also purchase raw materials and equipment from third-party suppliers for the manufacture of DepoCyt(e). While we have not experienced shortages of our raw materials in the past, such suppliers may not sell these raw materials to us at the times that we need them or on commercially reasonable terms and we do not have direct control over the availability of these raw materials from our suppliers.
All manufacturing of products, initial product release and stability testing are conducted by us in accordance with current Good Manufacturing Practices, or cGMP.
Building 1 is an approximately 84,000 square foot concrete structure located on a five acrefive-acre site. It was custom built as a pharmaceutical research and development and manufacturing facility in 1995. Activities in this facility include the manufacture of EXPAREL bulk product on dedicated production lines and its fill/finish into vials, microbiological and quality control testing, product storage, development of analytical methods and manufacturing of development products. Prior to 2014, the bulk manufacturing of all EXPAREL product sold to the marketplace had occurred in a manufacturing line housed in what we refer to as Suite A. In 2014, the FDA approved our recently installed manufacturing lines, referred to as Suite C. Suite C significantly increased our manufacturing capacity and ability to meet the growing demand for EXPAREL. In 2017, we expect Suite A to be used for both commercial production of EXPAREL and for the production of clinical material for development products. We are expanding our EXPAREL manufacturing capacity directly and through agreements with a third-party, Thermo Fisher Scientific Pharma Services, or Thermo Fisher (formerly Patheon U.K. Limited, or Patheon,UK Limited), as demand for EXPAREL increases, as explained below.
Building 2 is a recently renovatedan approximately 45,000 square foot steel and concrete structure located adjacent to Building 1, originally built as a pharmaceutical research and development lab and office building located adjacent to Building 1, built in 2003. We moved most ofThis building houses our Science Center related general and administrative functions to this building in 2015, as roughly half of the building is office space.functions. The other half of the building is being used for research and development activities as it includes both laboratories and the building infrastructure necessary to support the formulation, analytical testing, clinical and process development activities for additional commercial product indications and new pipeline products.
Building 7 is an approximately 21,000 square foot concrete panel structurebuilding built in 1988 whichthat serves as the main cGMP warehouse for all of our San Diego operations. It was recently renovated in early 2014 to support the expansion of EXPAREL. The warehouse isoperations, primarily being used for the storage of materials used in the production of our products.materials. It contains ambient as well as cold temperature cGMP warehouse storage for materials used in our manufacturing operations. Itand also features a quality control clean room for sampling incoming materials.
Our Fremont, California facility was built in 1998 and has been leased since 2015. It is dedicated to the iovera° product line and consists of approximately 20,000 square feet of space for manufacturing, quality control, research and development and the warehousing of raw materials and finished goods. The entire iovera° product line of tips, cartridges and handheld systems are produced as well as developed at this site.
Distribution of our DepoFoam products, including EXPAREL, requires cold-chain distribution, whereby a product must be maintained between specified temperatures. We have validated processes for continuous monitoring of temperature from manufacturing through delivery to the end-user. We and our partners utilize similar cold-chain processes for DepoCyt(e).
Co-Production Facilities
In April 2014, we and PatheonThermo Fisher entered into a Strategic Co-Production Agreement, Technical Transfer and Service Agreement and Manufacturing and Supply Agreement (the “Patheon“Thermo Fisher Agreements”) to collaborate in the manufacture of
EXPAREL. Patheon has agreed to undertakeThermo Fisher undertook certain technical transfer activities and construction services needed to prepare Patheon’sThermo Fisher’s Swindon, England facility for the manufacture of EXPAREL in two dedicated manufacturing suites. We will provide Patheonprovided Thermo Fisher with the equipment necessary to manufacture EXPAREL and will pay fees to PatheonThermo Fisher based on Patheon’sThermo Fisher’s achievement of certain technical transfer and construction milestones. We will also reimburse PatheonThermo Fisher for certain nominal expenses and additional services. We also currently expect, subject to receiptIn February 2019, we announced that commercial production of regulatory approvals,EXPAREL is underway at the first commercialThermo Fisher suite, and that we are developing a second dedicated suite that is expected to enable another doubling of EXPAREL manufacturing suite at Patheon’s facilitycapacity and should be available to commencebegin commercial production in late 2018.
The initial term of the Manufacturing and Supply Agreement is 10 years from the date of FDA approval of the initial manufacturing suite.suite, which was received in May 2018. We will pay fees to PatheonThermo Fisher for their operation of the manufacturing suites and the amount of EXPAREL produced by Patheon.Thermo Fisher. We will also reimburse PatheonThermo Fisher for purchases made on our behalf, certain nominal expenses and additional services. We may terminate this agreement upon one month’s notice if a regulatory authority causes the withdrawal of EXPAREL from the United StatesU.S. or any other market that represents 80% of our overall sales, or at any time for convenience by providing between 18 and 36 monthsmonths’ notice (depending on the number of years after the FDA approval date). Either party may terminate the Manufacturing and Supply Agreement in the event of the breach or bankruptcy of the other party.
Intellectual Property and Exclusivity
We seek to protect our products, our product candidates and our technologytechnologies through a combination of patents, trade secrets, proprietary know-how, regulatory exclusivity and contractual restrictions on disclosure. We note that the patents and applications described below are only examples intended to highlight the variety of coverage provided by our existing and constantly developing portfolio.
Patents and Patent Applications
We seek to protect the proprietary position of our products and product candidates by, among other methods, filing USU.S. and foreign patent applications related to our proprietary technology, inventions and improvements that are important to the development of our business. As of December 31, 2016,2019, there are over 14nine families of patents and patent applications relating to various aspects of the DepoFoam delivery technology.technology and 25 families of patents and patent applications relating to various aspects of the technology used by iovera°. Patents have been issued in numerous countries, with an emphasis on the North American, European and Japanese markets. These patents generally have a term of 20 years from the date of the non-provisional filing unless referring to an earlier filed application. Some of our expired USU.S. patents havehad a term fromof 17 years from the grant date. Our issued patents expire at various dates in the future, as discussed below, with the last currently issued patent for the DepoFoam delivery technology expiring in 2018. Certain pending2033 and the last currently issued patent applications may qualify for patent term adjustment that, if granted, would provide patent protectionthe iovera° technology expiring in 2037.
Patents and Patent Applications for EXPAREL beyond 2018. DepoFoam and DepoFoam Products
We received an issue notification from the United States Patent and Trademark Office, or USPTO, stating that a patent relating to product-by-process and process claims in connection with the production of multivesicular liposomes will issuewas issued on March 7, 2017. This patent will beis listed on the Orange Book for EXPAREL and includes a patent term adjustment that equates to an expiration date of December 24, 2021.
Issued patents for EXPAREL in the United StatesU.S. relating to methods for modifying the rate of drug release of the product candidate and the composition of the product candidate expired in January 2017 and will expire in September 2018, respectively. Pursuant to 35 U.S.C. § 156, an application for patent term extension was filed with the USPTO in October 2016 in connection with the regulatory approval of Aratana Therapeutics, Inc.’s NOCITA®. A USAratana’s NOCITA. That application was subsequently withdrawn after the product-by-process patent, relating to compositions including EXPAREL, but not EXPAREL specifically, expired in November 2013. Areferenced above, was issued on March 7, 2017. In the U.S., a patent relating to the composition of the product was issued in September 2014 and will expireexpired in September 2018. A patent relating to the method of treatment using EXPAREL was issued in December 2015 and will expireexpired in September 2018. Two pending US applications relating to the process for making the product candidate, if granted, would expire in November 2018 or later. In Europe, granted patent(s) related to the composition of the product candidate expireEXPAREL expired in September 2018 and certain European patent(s) expired in November 2014. In Europe, a2018. A patent relating to methods of modifying the rate of drug release of the product candidate expiresexpired in January 2018. A pending application in EuropeIn addition, a patent relating to the process for making the product candidate if granted, would expireexpired in November 2018.
In April 2010, a provisional patent was filed relating to a new process to manufacture EXPAREL and other DepoFoam-
Issued patents in the U.S. afford us a wide range of coverage of various aspects of the iovera° technology. For example, several of our earliest filed patents cover the structural aspects of a handheld cryogenic device with single needle and needle arrays, tissue-penetrating needle probes that may be detachable, fused silica tubing fluid delivery paths, methods of applying cryotherapy using the cryogenic device and methods for using replaceable needle probes. These patents are set to protect our two pipeline candidates, DepoMLXexpire between 2025 and DepoTXA. Pending2032. An important patent applications for compositionsfamily specifically directed to systems and methods of treating pain offers both broad and variable coverage of cryogenic device features and methods of using the same for pain management, including single-use needle probes, particular needle sizes and shapes. Patents in this family are set to expire between 2025 and 2028. Another important patent family has broad disclosure and coverage of a variety of indications for treatment by cryogenic devices, including joint function and stiffness, osteoarthritis, occipital neuralgia, spasticity, neuroma and other nerve entrapment indications and is set to expire between 2033 and 2037.
Additionally, there are several patents and pending patent applications directed to other important aspects of DepoMLX, if granted, wouldthe iovera° technology. For example, patents covering needle cladding and the probe filtration system are set to expire in October 2031. In addition,2033 and a patent on the smart tip technology is not set to expire until 2037. Other applications cover methods of using needles with blunt tips and aspects of cryogenic devices coupled with a neurostimulator for locating nerves. We also have three design patent families that cover the current handheld cryogenic device, its charging station dock and combinations thereof. To obtain coverage of our developing next-generation technology, we filed four new provisional patent application for DepoTXA has been filedapplications in 2019, which if converted and if granted, would expire in January 2036.could potentially prevent others from using this next-generation technology until at least 2040.
Trade Secrets and Proprietary Information
Trade secrets play an important role in protecting our DepoFoam-based and iovera° products and provide protection beyond patents and regulatory exclusivity. The scale-up and commercial manufacture of DepoFoamDepoFoam-based and iovera° products involvesinvolve processes, custom equipment and in-process and release analytical techniques that we believe are unique to us. The expertise and knowledge required to understand the critical aspects of DepoFoam manufacturing steps requires knowledge of both traditional and non-traditional emulsion processing and traditional pharmaceutical production, overlaid with all of the challenges presented by aseptic manufacturing. The iovera° system relies on manufacturing techniques that are able to provide the precision and tight tolerances required for a self-contained handheld cryogenic device. Additionally, our device includes proprietary software for device operations during cryotherapy treatments.
We seek to protect our proprietary information, including our trade secrets and proprietary know-how, by requiring our employees, consultants and other advisors to execute proprietary information and confidentiality agreements upon the commencement of their employment or engagement. These agreements generally provide that all confidential information developed or made known during the course of the relationship with us be kept confidential and not be disclosed to third parties except in specific circumstances. In the case of our employees, the agreements also typically provide that all inventions
resulting from work performed for us, utilizing our property or relating to our business and conceived or completed during employment shall be our exclusive property to the extent permitted by law. Where appropriate, agreements we obtain with our consultants also typically contain similar assignment of invention obligations. Further, we require confidentiality agreements from third parties that receive our confidential data or materials.
Competition
EXPAREL
The pharmaceutical and biotechnology industries areindustry is intensely competitive and subject to rapid and significant technological change. Our competitors include organizations such as major multinational pharmaceutical companies, established biotechnology companies, specialty pharmaceutical companies and generic drug companies. Many of our competitors have greater financial and other resources than we have, such as more commercial resources, larger research and development staffs and more extensive marketing and manufacturing organizations. As a result, these companies may obtain marketing approval more rapidly than we are able and may be more effective in developing, selling and marketing their products. Smaller or early stageearly-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies.
Our competitors may succeed in developing, acquiring or licensing on an exclusive basis technologies and drug products that are more effective or less costly than EXPAREL or any other products that we are currently selling through partners or developing or that we may develop, which could render our products obsolete and noncompetitive. We expect any products that we develop and commercialize to compete on the basis of, among other things, efficacy, safety, convenience of administration and delivery, price and the availability of reimbursement from government and other third-party payers.
EXPAREL competes with well-established products with similar indications. Competing products available for postsurgical pain management include opioids such as morphine, fentanyl, meperidine and hydromorphone, each of which is available generically from several manufacturers, and several of which are available as proprietary products using novel delivery systems. Ketorolac, ana non-steroidal anti-inflammatory drug, or NSAID, is also available generically in the United StatesU.S. from several manufacturers, and Caldolor (ibuprofen for injection), an NSAID, has been approved by the FDA for pain management and fever in adults. EXPAREL also competes with currently-marketed non-opioid products such as bupivacaine, marcaine, ropivacaine and other anesthetics/analgesics, all of which are also used in the treatment of postsurgical pain and are available as either oral tablets, injectable dosage forms or administered using novel delivery systems. Additional products may be developed for the treatment of acute pain, including new injectable NSAIDs, novel opioids, new formulations of currently available opioids and NSAIDs, long-acting local anesthetics and new chemical entities as well as alternative delivery forms of various opioids and NSAIDs. Currently EXPAREL also competes with elastomeric pump/catheter devices intended to provide bupivacaine over several days. I-FLOW Corporation (acquired by Kimberly-Clark Corporation in 2009 and spun off into Halyard Health,Avanos Medical, Inc. in 2014) has marketedmarkets these medical devices in the United States since 2004.U.S.
iovera°
The medical device industry is intensely competitive and subject to rapid and significant technological change. The cryotherapy pain management field in particular is a growing industry due to increased attention on opioid usage for pain, which has created a rapidly emerging market and has fueled an increased interest in opioid alternatives. Many of our competitors in our space have greater financial and other resources than we have, such as more commercial resources, larger research and development staffs and more extensive marketing and manufacturing organizations. As a result, these companies may obtain marketing approval more rapidly than we are able and may be more effective in developing, selling and marketing their products. The rise of various small and early-stage companies in the cryotherapy pain management field may also prove to be significant competitors, particularly if they enter into collaborative arrangements with large, established companies.
Our competitors are continuously engaged in trials and attempts to develop new products or approaches in hopes of capturing the pain management market. They may succeed in developing, acquiring or licensing on an exclusive basis, technologies that are more effective or less costly than the iovera° system, which could render the iovera° system obsolete and noncompetitive. As a result, it is critical that we continue to innovate and to increase marketing efforts in our primary markets. We expect any products that we develop and commercialize to compete on the basis of, among other things, efficacy, safety, convenience of administration and delivery, price and the availability of reimbursement from government and other third-party payers.
Besides pharmaceutical products for pain management, iovera° competes with medical devices that ablate or degenerate peripheral nerves to treat indications such as joint pain, neuralgia and osteoarthritis pain. Competing products include cryotherapy devices as well as other devices such as cooled radio-frequency ablation devices that block or degenerate
peripheral nerves involved in conducting pain signals. Avanos Medical, Inc. markets these medical devices in the U.S. Additional non-opioid products or entirely different approaches may also be developed for pain management by one or more of our competitors.
Government Regulation
In the United States,U.S., prescription drug and medical device products are subject to extensive pre- and post-market regulation by the FDA, including regulations that govern the research, development, testing, manufacturing, distribution, safety, efficacy, approval, labeling, storage, record keeping, reporting, advertising and promotion of such products under the Federal Food, Drug and Cosmetic Act, or FDCA, and its implementing regulations. Outside the United States,U.S., prescription drug and medical device products are regulated by comparable agencies, laws and regulations. Failure to comply with applicable regulatory requirements in the United StatesU.S. or elsewhere may result in, among other things, refusal to approve pending applications, withdrawal of an approval, warning letters, clinical holds, civil or criminal penalties, recall or seizure of products, injunction, debarment, partial or total suspension of production or withdrawal of the product from the market. Any agency or judicial enforcement action could have a material adverse effect on the Company.
United States Regulatory Environment
Pharmaceuticals
Generally, the FDA must approve any new drug, including a new use of a previously approved drug, before marketing of the drug occurs in the United States.U.S. This process generally involves:
completion of preclinical laboratory and animal testing and formulation studies in compliance with the FDA’s Good Laboratory Practice regulations (21 CFR 58);
submission to the FDA of an Investigational New Drug, or IND, application for human clinical testing, which must become effective before human clinical trials may begin for unapproved use in the United States;U.S.;
approval by an independent Institutional Review Board, or IRB, at each clinical trial site before each trial may be initiated;
performance of adequate and well-controlled human clinical trials in accordance with the FDA’s Good Clinical Practices, or GCP, to establish the safety and efficacy of the proposed drug product for each intended use;
completion of process validation, quality product release and stability;
submission of ana New Drug Application, or NDA, to the FDA;
satisfactory completion of an FDA pre-approval inspection of the product’s manufacturing facility or facilities to assess compliance with cGMP requirements and to ensure that the facilities, methods and controls are adequate to preserve the drug’s identity, quality and purity;
satisfactory completion of an FDA advisory committee review, if applicable; and
review and approval by the FDA of the NDA.
The preclinical and clinical testing and approval process requires substantial time, effort and financial resources, and we cannot be certain that the FDA will grant approvals for any of our product candidates on a timely basis, if at all. Preclinical tests include laboratory evaluation of product chemistry, formulation and stability, as well as studies to evaluate toxicity in animals. The results of preclinical tests, together with manufacturing information, analytical data and a proposed clinical trial protocol and other information, are submitted as part of an IND application to the FDA. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA places the trial on a clinical hold because of, among other things, concerns about the conduct of the clinical trial or about exposure of human research subjects to unreasonable health risks. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. Thus, submission of an IND does not by itself automatically result in FDA authorization to commence a clinical trial. In addition, the FDA requires us to amend an existing IND for each successive clinical trial conducted during product development. Further, an IRB covering each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial along with informed consent information for subjects before the clinical trial commences at that center. The IRB also must monitor the clinical trial
until it is completed. The FDA, the IRB or the sponsor may suspend a clinical trial at any time, on various grounds, including a finding that the subjects or patients are being exposed to an unacceptable health risk. We may also suspend or terminate a clinical trial based on evolving business objectives and/or the competitive climate.
Clinical trials involve the administration of the product candidate to healthy volunteers or patients having the disease being studied under the supervision of qualified investigators in accordance with GCP requirements, which include the
requirement that all research subjects provide their informed consent for their participation in any clinical trial. Sponsors of clinical trials generally must register at the NIH-maintained website www.clinicaltrials.gov(www.clinicaltrials.gov) and report key findings and parameters. For purposes of an NDA submission and approval, typically, the conduct of human clinical trials occurs in the following three pre-market sequential phases, which may overlap or be combined:
| • | Phase 1: Sponsors initially conduct clinical trials in a limited population, either patients or healthy volunteers, to test the product candidate for safety, dose tolerance, absorption, metabolism, distribution, excretion and clinical pharmacology, and, if possible, to gain early evidence of effectiveness. In the cases of some products for severe or life-threatening diseases, especially when the product may be too inherently toxic to ethically administer to healthy volunteers, the initial human testing often is conducted only on patients having the specific disease. |
| • | Phase 2: Sponsors conduct clinical trials generally in a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted indications and to determine dose tolerance, optimal dosage and dosing schedule. Sponsors may conduct multiple Phase 2 clinical trials to obtain information prior to beginning larger and more extensive Phase 3 clinical trials. |
| • | Phase 3: These include expanded controlled and uncontrolled trials, including pivotal clinical trials. When Phase 2 evaluations suggest the effectiveness of a dose range of the product and acceptability of such product’s safety profile, sponsors undertake Phase 3 clinical trials in larger patient populations to obtain additional information needed to evaluate the overall benefit and risk balance of the drug and to provide an adequate basis to develop labeling. |
Some clinical trials may be overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group provides authorization for whether or not a trial may move forward at designated check points based on access to certain data from the trial. The process of completing clinical testing and obtaining FDA approval for a new drug is likely to take a number of years and requires the expenditure of substantial resources. If an application is submitted, there can be no assurance that the FDA will review and approve the NDA. In addition, sponsors may elect to conduct, or be required by the FDA to, conduct post-approval clinical trials to further assess the drug’s safety or effectiveness after NDA approval.approval, generate new data and best-practice administration techniques. Such post approval trials are typically referred to as Phase 4 clinical trials.
In the U.S., the Medical Device Amendments of 1976 to the FDCA and its subsequent amendments regulate the design, manufacture and marketing of medical devices. Medical devices that require notification submitted as a 510(k) clearance request must be reviewed and cleared by the FDA before we can begin marketing them. To request 510(k) clearance, we must be able to demonstrate that the medical device is substantially equivalent to a previously-cleared and legally marketed 510(k) medical device. Medical devices require extensive clinical testing which consists of safety and efficacy studies, followed by pre-market approval, or PMA, applications for specific surgical indications. The FDA’s Quality System Regulations, or QSRs, set forth standards for our product design and manufacturing processes, require the maintenance of certain records and provide for inspections of our facilities by the FDA. There are also certain requirements of state, local and foreign governments that must be complied with in the manufacture and marketing of our products.
U.S. Review and Approval Process
Pharmaceuticals
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, sponsors submit the results of product development, preclinical studies and clinical trials to the FDA as part of an NDA requesting approval to market the product for one or more indications. NDAs must also contain extensive information relating to the product’s pharmacology, chemistry, manufacture, controls and proposed labeling, among other things. In addition, 505(b)(2) applications must contain a patent certification for each patent listed in FDA’s “Orange Book” that covers the drug
referenced in the application and upon which the third-party studies were conducted. For some drugs, the FDA may require risk evaluationRisk Evaluation and mitigation strategies,Mitigation Strategies, or REMS, which could include medication guides, physician communication plans or restrictions on distribution and use, such as limitations on who may prescribe the drug or where it may be dispensed or administered. Upon receipt of an NDA, the FDA has 60 days to determine whether it is sufficiently complete to initiate a substantive review. If the FDA identifies deficiencies that would preclude substantive review, the FDA will refuse to accept the NDA (“refuse to file”) and will inform the sponsor of the deficiencies that must be corrected prior to resubmission. The resubmitted application is also subject to review before the FDA accepts it for filing. If the FDA accepts the submission for substantive review, the FDA typically reviews the NDA in accordance with established timeframes. Under the Prescription Drug User Fee Act, or PDUFA, the FDA establishes goals for NDA review time through a two-tiered classification system: Priority Review and Standard Review. A Priority Review designation is given to drugs that address andan unmet medical need by offering major advances in treatment or providing a treatment where no adequate therapy currently exists. Standard Review applies to all applications that are not eligible for Priority Review. The FDA aims to complete Standard Reviews of NDAs within ten12 months of submission (ten months after the Day 60 filing date) and Priority Reviews within 6 months.eight months of submission (six months after the Day 60 filing date). Review processes may sometimes extend beyond these target completion dates due to FDA requests for additional information or clarification, difficulties scheduling an advisory committee meeting, negotiations regarding REMS or FDA workload issues, but in general under PDUFA the FDA is supposed to complete its reviews within the target timeframes despite these factors. The FDA may refer the application to an advisory committee for review, evaluation and recommendation as to the application’s approval. The recommendations of an advisory committee do not bind the FDA, but the FDA generally follows such recommendations.
Under PDUFA, NDA applicants must pay significant NDA user fees upon submission. In addition, manufacturers of approved prescription drug products must pay annual establishment and product userprogram fees.
Before approving an NDA, the FDA will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and are adequate to ensure consistent production of the product within required specifications. Additionally, the FDA will typically inspect one or more clinical sites to ensure compliance with GCP before approving an NDA.
After the FDA evaluates the NDA and the manufacturing facilities, it may issue an approval letter or a Complete Response Letter, or CRL, to indicate that the review cycle for an application is complete and that the application is not ready for approval. CRLs generally outline the deficiencies in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application. Even if such additional information is submitted, the FDA may ultimately decide that the NDA does not satisfy the criteria for approval. Data from clinical trials are not always conclusive and the FDA may interpret data differently than we do. The FDA could also require a REMS plan to mitigate risks, which could include medication guides, physician communication plans or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA also may approve an NDA contingent on, among other things, changes to proposed labeling, a commitment to conduct one or more post-market studies or clinical trials and the correction of identified manufacturing deficiencies, including the development of adequate controls and specifications. If and when the deficiencies have been addressed to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Medical Devices
In the U.S., authorization to bring a medical device to market is generally obtained in one of two ways. The first pathway, a pre-market notification (the 510(k) process), requires demonstration that the new device is substantially equivalent to an already legally marketed medical device. The second pathway, known as pre-market approval, or PMA, requires an independent demonstration that a medical device is safe and effective for its intended use. In general, pre-market approvals require a much longer time horizon and can be much more expensive than obtaining clearance through the 510(k) process.
To obtain 510(k) clearance, we must file with the FDA a pre‑market notification demonstrating that our proposed device is substantially equivalent to a previously cleared and legally marketed 510(k) device or a device that was in commercial distribution before May 28, 1976 for which the FDA has not yet called for the submission of a PMA. 510(k) clearance for iovera° was first obtained in March 2009 when the focus of MyoScience was cosmetic applications (i.e. facial wrinkle reduction). The MyoScience business focus shifted to pain management in 2014, and since then there have been a number of advancements that led to three additional 510(k) submissions and clearances to support iovera° and the subsequent growth of the iovera° product line.
A PMA must be submitted to the FDA if it is determined that the device is not eligible for the 510(k) clearance process. A PMA must be supported by extensive data including, but not limited to, technical, preclinical and clinical trials, manufacturing and labeling to demonstrate reasonable evidence of the device’s safety and efficacy to the FDA’s satisfaction.
After a device receives 510(k) clearance or a PMA approval, it may be changed or modified. Any modification that could significantly affect its safety or effectiveness, or that would constitute a significant change in its intended use, will require a new clearance or approval. Regulations provide that the manufacturer initially determines when a specific modification requires notification to FDA. The FDA has issued draft guidance that, if finalized and implemented, will result in manufacturers needing to seek a significant number of new clearances for changes made to legally marketed devices. The FDA reviews the manufacturer’s decision to file a 510(k) or PMA for modifications during facility audits.
Section 505(b)(2) New Drug Applications
Applications under Section 505(b)(2) are subject to any non-patent exclusivity period applicable to the referenced product, which may delay approval of the 505(b)(2) application even if the FDA has completed its substantive review and determined the drug should be approved. In addition, 505(b)(2) applications must include patent certifications to any patents listed in the FDA’s Orange Book as covering the referenced product. If the 505(b)(2) applicant seeks to obtain approval before the expiration of an applicable listed patent, the 505(b)(2) applicant must provide notice to the patent owner and NDA holder of the referenced product. If the patent owner or NDA holder brings a patent infringement lawsuit within 45 days of such notice, the 505(b)(2) application cannot be approved for 30 months or until the 505(b)(2) applicant prevails, whichever is sooner. If the 505(b)(2) applicant loses the patent infringement suit, FDA may not approve the 505(b)(2) application until the patent expires, plus any period of pediatric exclusivity.
In the NDA submissions for our product candidates, we intend to follow the development and approval pathway permitted under the FDCA that we believe will maximize the commercial opportunities for these product candidates.
Post-Approval Requirements
Pharmaceuticals
After approval, the NDA sponsor must comply with comprehensive requirements governing, among other things, drug listing, recordkeeping, manufacturing, marketing activities, product sampling and distribution, annual reporting and adverse event reporting.
If new safety issues are identified following approval, the FDA can require the NDA sponsor to revise the approved labeling to reflect the new safety information; conduct post-market studies or clinical trials to assess the new safety information and implement a REMS program to mitigate newly-identified risks. The FDA may also require post-approval testing, including Phase 4 trials, and surveillance programs to monitor the effect of approved products which have been commercialized, and the FDA has the authority to prevent or limit further marketing of a product based on the results of these post-marketing programs. Drugs may be marketed only for approved indications and in accordance with the provisions of the FDA-approved label. Further, if we modify a drug, including any changes in indications, labeling or manufacturing processes or facilities, the FDA may require us to submit and obtain FDA approval of a new or supplemental NDA, which may require us to develop additional data or conduct additional preclinical studies and clinical trials.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation
and correction of any deviations from cGMP and impose reporting and documentation requirements upon us and any third-party manufacturers that we may decide to use.
If after approval the FDA determines that the product does not meet applicable regulatory requirements or poses unacceptable safety risks, the FDA may take other regulatory actions, including initiating suspension or withdrawal of the NDA approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
fines, warning letters or holds on post-approval clinical trials;
refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals;
product seizure or detention, or refusal to permit the import or export of products; or
injunctions or the imposition of civil or criminal penalties.
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. These regulations include standards and restrictions for direct-to-consumer advertising, industry-sponsored scientific and educational activities, promotional activities involving the internet and off-label promotion. While physicians may prescribe for off-label uses, manufacturers may only promote for the approved indications and in accordance with the provisions of the approved label. The FDA has very broad enforcement authority under the FDCA, and failure to abide by these regulations can result in penalties, including the issuance of a warning letter directing entities to correct deviations from FDA standards, a requirement that future advertising and promotional materials be pre-cleared by the FDA, and state and federal civil and criminal investigations and prosecutions.
In addition, the distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, or PDMA, which regulates the distribution of drugs and drug samples at the federal level and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution, including a drug pedigree which tracks the distribution of prescription drugs.
In December 2015, we announced that we achieved an amicable resolution with the United StatesU.S. in our lawsuit filed in September 2015 against the FDA and other governmental defendants. The resolution confirmsconfirmed that EXPAREL is, and has been since its approval in 2011, broadly indicated for administrationsingle-dose infiltration into the surgical site to provideproduce postsurgical analgesia. In April 2018, the FDA approved an expansion of the label for EXPAREL to include interscalene brachial plexus nerve block. The new indication statement in the label for EXPAREL now reads: “EXPAREL is indicated for single-dose infiltration in adults to produce postsurgical local analgesia and as an interscalene brachial plexus nerve block to produce postsurgical regional analgesia. Safety and efficacy has not been established in other nerve blocks.”
Medical Devices
The FDA has broad post‑market and regulatory obligations that we must adhere to. We are subject to unannounced inspections by the FDA to determine our compliance with QSRs and other rules and regulations.
After a medical device is placed on the market, numerous regulatory requirements apply. These include, but are not limited to:
QSRs, which require manufacturers, including third‑party manufacturers, to follow stringent design, testing, documentation and other quality assurance procedures during product design and throughout the manufacturing process;
Labeling regulations and FDA prohibitions against the promotion of products for uncleared, unapproved or off‑label uses; and
Medical device reporting regulations, which require that manufacturers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if the malfunction were to recur.
Failure to comply with regulatory requirements can result in enforcement action by the FDA, which may include any of the following sanctions:
restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
fines, warning letters or holds on post-approval clinical trials;
the potential withdrawal of 510(k) clearance or other approvals that were previously granted;
refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals;
product seizure or detention, or refusal to permit the import or export of products;
injunctions or the imposition of civil or criminal penalties; or
requiring us to repair, replace and/or refund the cost of any medical device we have manufactured or distributed.
If any of these events were to occur, they could have a material adverse effect on our business.
International Regulation
In addition to regulations in the United States,U.S., we are subject to a variety of foreign regulations governing clinical trials and the commercial sales and distribution of our products. Whether or not we obtain FDA approval for a product, we must obtain approval by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval.
For example, in Europe, there are several tracks for marketing approval for pharmaceuticals, for product approval and post-approval regulatory processes, depending on the type of product for which approval is sought. Under the centralized procedure, a company submits a single application to the EMA. The marketing application is similar to the NDA in the USU.S. and is evaluated by the Committee for Medicinal Products for Human Use, or CHMP, the expert scientific committee of the EMA. If the CHMP determines that the marketing application fulfills the requirements for quality, safety and efficacy, it will submit a favorable opinion to the European Commission, or EC. The CHMP opinion is not binding, but is typically adopted by the EC. A marketing application approved by the EC is valid in all member states. The centralized procedure is required for all biological
products, orphan medicinal products and new treatments for neurodegenerative disorders, and it is available for certain other products, including those which constitute a significant therapeutic, scientific or technical innovation.
As with FDA approval, we may not be able to secure regulatory approvals in Europe in a timely manner, if at all. Additionally, as in the United States,U.S., post-approval regulatory requirements, such as those regarding product manufacture, marketing or distribution would apply to any product that is approved in Europe, and failure to comply with such obligations could have a material adverse effect on our ability to successfully commercialize any product.
In addition to regulations in Europe and the United States,U.S., we will be subject to a variety of foreign regulations governing clinical trials, product approvals, and commercial distribution ofin Canada, China and any other jurisdictions in which EXPAREL, iovera° or any other future products.product is approved.
Third-Party Payer Coverage and Reimbursement
The commercial success of our products and product candidates will depend, in part, upon the availability of coverage and reimbursement from third-party payers at the federal, state and private levels. Government payer programs, including Medicare and Medicaid, private health care insurance companies and managed care plans may deny coverage or reimbursement for a product or therapy in whole or in part if they determine that the product or therapy is not medically appropriate or necessary. Also, third-party payers have attempted to control costs by limiting coverage and the amount of reimbursement for particular
procedures, medical devices or drug treatments. The United States Congress and state legislatures from time to time propose and adopt initiatives aimed at cost containment that could impact our ability to sell our products at a price level high enough to realize an appropriate return on our investment, which would materially impact our results of operations.
In March 2010, President Obama signed into law the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act which we refer to collectively as(collectively, the Affordable“Affordable Care Act,Act”), a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. The Affordable Care Act revised the definition of “average manufacturer price” for reporting purposes, which could increase the amount of Medicaid drug rebates owed to states by pharmaceutical manufacturers for covered outpatient drugs. The Affordable Care Act also established a new Medicare Part D coverage gap discount program, in which drug manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices of applicable brand name drugs to eligible beneficiaries during their coverage gap period as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D. Further, the new law imposed a significant annual, nondeductible fee on companies that manufacture or import branded prescription drug products. Substantial new provisions affecting compliance have also been enacted, which may require us to modify our business practices with healthcare practitioners. There have been proposed in Congress a number of legislative initiatives regarding healthcare, including possible repeal of the Affordable Care Act. At this time, it remains unclear whether there will be any changes made to the Affordable Care Act. The full impact that the Affordable Care and other new laws will have on our business is uncertain. However, such laws appear likely to continue the pressure on pharmaceutical pricing, especially under the Medicare program, and may also increase our regulatory burdens and operating costs. Moreover, in the coming years, additional changes could be made to governmental healthcare programs that could significantly impact the success of our products.
The marketability of our products may suffer if the government and third-party payers fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care in the United StatesU.S. has increased, and we expect will continue to increase, the pressure on pharmaceutical and medical device pricing. Some third-party payers require pre-approval of coverage for new or innovative devices or drug therapies before they will reimburse healthcare providers that use such therapies, or place limits on the amount of reimbursement. Coverage policies and third-party payer reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for our products, less favorable coverage policies and reimbursement rates may be implemented in the future.
In international markets, reimbursement and healthcare payment systems vary significantly by country, and many countries have instituted price ceilings on specific products and therapies. There can be no assurance that our products will be considered medically reasonable and necessary for a specific indication, that our products will be considered cost-effective by
third-party payers or that an adequate level orof reimbursement will be available so that the third-party payers’ reimbursement policies will not adversely affect our ability to sell our products profitably.
Marketing/Data Exclusivity
The FDA may grant three or five years of marketing exclusivity in the United StatesU.S. for the approval of new or supplemental NDAs, including Section 505(b)(2) NDAs, for, among other things, new indications, dosages or dosage forms of an existing drug, if new clinical investigations that were conducted or sponsored by the applicant are essential to the approval of the application. Additionally, six months of marketing exclusivity in the United StatesU.S. is available under Section 505A of the FDCA if, in response to a written request from the FDA, a sponsor submits and the agency accepts requested information relating to the use of the approved drug in the pediatric population. This six monthsix-month pediatric exclusivity period is not a standalone exclusivity period, but rather is added to any existing patent or non-patent exclusivity period for which the drug product is eligible. BasedIn the past, based on our clinical trial program for EXPAREL, the FDA granted three years of marketing exclusivity to EXPAREL, which expired onin October 28, 2014.
Manufacturing Requirements
We must comply with the FDA’s cGMP requirements and comparable regulations in other countries. The cGMP provisions include requirements relating to the organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports and returned or salvaged products. The manufacturing facilities for our products must meet cGMP requirements to the satisfaction of the FDA and other authorities pursuant to a pre-approval inspection before we can use them to manufacture our products. We and any third-party manufacturers we engage or with which we partner are also subject to periodic inspections of facilities by the FDA and other authorities, including procedures and operations used in the testing and manufacture of our products to assess our compliance with applicable regulations. Failure to comply with these and other statutory and regulatory requirements subjects a manufacturer to possible
legal or regulatory action, including warning letters, the seizure or recall of products, injunctions, consent decrees placing significant restrictions on or suspending manufacturing operations and civil and criminal penalties. Adverse experiences with the product or product complaints must be reported and could result in the imposition of market restrictions through labeling changes or in product removal. Product approvals may be withdrawn if compliance with regulatory requirements is not maintained or if problems concerning safety or efficacy of the product occur following approval.
Regulations Pertaining to Sales and Marketing
We are subject to various federal and state laws pertaining to health care “fraud and abuse,” including anti-kickback laws and false claims laws. Anti-kickback laws generally prohibit a prescription drug manufacturer from soliciting, offering, receiving, or paying any remuneration to generate business, including the purchase or prescription of a particular drug.drug or medical device. Although the specific provisions of these laws vary, their scope is generally broad and there may be no regulations, guidance or court decisions that clarify how the laws apply to particular industry practices. There is therefore a possibility that our practices might be challenged under the anti-kickback or similar laws. False claims laws prohibit anyone from knowingly and willingly presenting, or causing to be presented for payment to third-party payers (including Medicare and Medicaid) claims for reimbursed drugs, procedures or services that are false or fraudulent, claims for items or services not provided as claimed, or claims for medically unnecessary items or services. Our activities relating to the sale and marketing of our products may be subject to scrutiny under these laws. Violations of fraud and abuse laws may be punishable by criminal or civil sanctions, including fines and civil monetary penalties and exclusion from federal health care programs (including Medicare and Medicaid). In the US,U.S., federal and state authorities are paying increased attention to enforcement of these laws within the pharmaceutical industryand medical device industries and private individuals have been active in alleging violations of the laws and bringing suits on behalf of the government under the federal civil False Claims Act. If we were subject to allegations concerning, or were convicted of violating, these laws, our business could be harmed.
Laws and regulations have been enacted by the federal government and various states to regulate the sales and marketing practices of pharmaceutical and medical device manufacturers. The laws and regulations generally limit financial interactions between manufacturers and health care providers or require disclosure to the government and public of such interactions. The laws include the federal Physician Payment Sunshine Act, or “sunshine” provisions, enacted in 2010 as part of the Affordable Care Act. The sunshine provisions apply to pharmaceutical and medical device manufacturers with products reimbursed under certain government programs and require those manufacturers to disclose annually to the federal government (for re-disclosure to the public) certain payments made to physicians and certain other healthcare practitioners or to teaching hospitals. State laws may also require disclosure of pharmaceutical and medical device pricing information and marketing expenditures. Many of these laws and regulations contain ambiguous requirements. Given the lack of clarity in laws and their implementation, our reporting actions could be
subject to the penalty provisions of the pertinent federal and state laws and regulations. Outside the US,U.S., other countries have implemented requirements for disclosure of financial interactions with healthcare providers and additional countries may consider or implement such laws.
In April 2015, we received a subpoena from the USU.S. Department of Justice, USU.S. Attorney’s Office for the District of New Jersey, requiring the production of a broad range of documents pertaining to marketing and promotional practices related to EXPAREL. We are cooperating with the government’s inquiry. We can make no assurances asRefer to the time or resources that will need to be devotedItem 3, Legal Proceedings, for an update related to this inquiry or its final outcome, or the impact, if any, of this inquiry or any proceedings on our business, financial condition, results of operations and cash flows.matter.
Healthcare Privacy and Security Laws
We may be subject to, or our marketing activities may be limited by the Health Insurance Portability and Accountability Act, or HIPAA and its implementing regulations, which established uniform standards for certain “covered entities” (healthcare providers, health plans and healthcare clearinghouses) governing the conduct of certain electronic healthcare transactions and protecting the security and privacy of protected health information. The American Recovery and Reinvestment Act of 2009, commonly referred to as the economic stimulus package, included sweeping expansion of HIPAA’s privacy and security standards called the Health Information Technology for Economic and Clinical Health Act, or HITECH, which became effective on February 17, 2010. Among other things, the new law makes HIPAA’s privacy and security standards directly applicable to “business associates”—independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions.
Environmental Matters
Our research and development processes and our manufacturing processes involve the controlled use of hazardous materials and chemicals and produce waste products. We are subject to federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous materials and waste products. We do not expect the cost of complying with these laws and regulations to be material. While we believe we are in compliance with applicable environmental regulations, the failure to fully comply with any such regulations could result in the imposition of penalties, fines and/or sanctions which could have a material adverse effect on our business.
Employees
As of December 31, 2016,2019, we had 503606 employees. All of our employees are located in the United StatesU.S. except for sixnine located in England.England and one located in the Netherlands. None of our employees are represented by a labor union, and we consider our current employee relations to be good.
Available Information
Our corporate website is located at www.pacira.com. We file reports and other information with the United States Securities and Exchange Commission, or SEC, as required by the Exchange Act.Act, which are accessible on the SEC’s website at www.sec.gov. We also make available free of charge through our website (http://www.pacira.com) our annual report on Form 10-K,Annual Report, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements and any amendments to those reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Exchange Act. We make these reports available through our website as soon as reasonably practicable after we electronically file such reports with, or furnish such reports to, the SEC. In addition, we regularly use our corporate website to post information regarding our business, product development programs and governance, and we encourage investors to use our website, particularly the information in the sectionsections entitled “Investors & Media,“Investors” and “News,” as a source of information about us. The foregoing references to our corporate website are not intended to, nor shall they be deemed to, incorporate information on our website into this Annual Report on Form 10-K by reference.
Item 1A. Risk Factors
In addition to the other information in this Annual Report, on Form 10-K, any of the factors set forth below could significantly and negatively affect our business, financial condition, results of operations or prospects. The trading price of our common stock may decline due to these risks. This section contains forward-looking statements. You should refer to the explanation of the qualifications and limitations on forward-looking statements beginning on page 1.
Risks Related to the Development and Commercialization of Ourour Products and Product Candidates
Our success depends primarily on our ability to successfully commercialize EXPAREL.
We have invested a significant portion of our efforts and financial resources in the development and commercialization of our lead product, EXPAREL, which was approved by the FDA on October 28, 2011 and commercially launched in April 2012. During 2016,2019, sales of EXPAREL constituted the vast majoritysubstantially all of our total revenue, and we expect it will do so for the foreseeable future. Our success depends on our ability to continue to effectively commercialize EXPAREL. Our ability to effectively generate revenues from EXPAREL will depend on our ability to, among other things:
create market demand for EXPAREL through our marketing and sales activities and other arrangements established for the promotion of EXPAREL;
train, deploy and support a qualified sales force;
secure formulary approvals for EXPAREL at a substantial number of targeted hospitals;hospitals and ASCs;
manufacture EXPAREL in sufficient quantities in compliance with requirements of the FDA and similar foreign regulatory agencies and at acceptable quality and pricing levels in order to meet commercial demand;
implement and maintain agreements with wholesalers distributors and group purchasing organizationsdistributors on commercially reasonable terms;
receive adequate levels of coverage and reimbursement for EXPAREL from commercial health plans and governmental health programs;
maintain compliance with regulatory requirements;
obtain regulatory approvals for additional indications for the use of EXPAREL;
ensure that our entire supply chain efficiently and consistently delivers EXPAREL to our customers; and
maintain and defend our patent protection and regulatory exclusivity for EXPAREL.
Any disruption in our ability to generate revenues from the sale of EXPAREL will have a material and adverse impact on our results of operations.
Our efforts to successfully commercialize EXPAREL are subject to many internal and external challenges and if we cannot overcome these challenges in a timely manner, our future revenues and profits could be materially and adversely impacted.
EXPAREL has been a commercialized drug for less than five years. As a result, wesince 2012. We continue to expend significant time and resources to train our sales force to be credible and persuasive in convincing physicians, hospitals and hospitalsASCs to use EXPAREL. In addition, we also must train our sales force to ensure that a consistent and appropriate message about EXPAREL is delivered to our potential customers. If we are unable to effectively train our sales force and equip them with effective materials, including medical and sales literature to help them inform and educate potential customers about the benefits and risks of EXPAREL and its proper administration, our efforts to successfully commercialize EXPAREL could be put in jeopardy, which could have a material adverse effect on our future revenues and profits.
In addition to our extensive internal efforts, the successful commercialization of EXPAREL will require many third parties, over whom we have no control, to choose to utilize EXPAREL. These third parties include physicians and hospital pharmacy and therapeutics committees, which we refer to as P&T committees. Generally, before we can attempt to sell EXPAREL in a hospital, EXPAREL must be approved for addition to that hospital’s list of approved drugs, or formulary list, by the hospital’s P&T committee. A hospital’s P&T committee typically governs all matters pertaining to the use of medications within the institution, including the review of medication formulary data and recommendations for the appropriate use of drugs within the institution to the medical staff. The frequency of P&T committee meetings at hospitals varies considerably, and P&T committees often require additional information to aid in their decision-making process. Therefore, we may experience substantial delays in obtaining formulary approvals. Additionally, hospital pharmacists may be concerned that the cost of
acquiring EXPAREL for use in their institutions will adversely impact their overall pharmacy budgets, which could cause pharmacists to resist efforts to add EXPAREL to the formulary, or to implement restrictions on the usage of EXPAREL or to encourage use of a lower cost dose than a surgeon or anesthesiologist would otherwise choose in order to control costs. We cannot guarantee that we will be successful in obtaining the approvals we need from enough P&T committees quickly enough to optimize hospital sales of EXPAREL.
Even if we obtain hospital formulary approval for EXPAREL, physicians must still prescribe EXPAREL for its commercialization to be successful. Because EXPAREL is a relatively new drug with a limited track record of sales in the United States, any inability to timely supply EXPAREL to our customers, or any unexpected side effects that develop from use of the drug, particularly early in product launch, may lead physicians to not accept EXPAREL as a viable treatment alternative.
If EXPAREL does not achieve broad market acceptance, the revenues that we generate from its sales will be limited. The degree of market acceptance of EXPAREL also depends on a number of other factors, including:
changes in the standard of care for the targeted indications for EXPAREL, which could reduce the marketing impact of any claims that we can make;
the relative efficacy, convenience and ease of administration of EXPAREL;
the prevalence and severity of adverse events associated with EXPAREL;
cost of treatment versus economic and clinical benefit, both in absolute terms and in relation to alternative treatments;
the availability of adequate coverage or reimbursement by third parties, such as insurance companies and other healthcare payers, and by government healthcare programs, including Medicare and Medicaid;
the extent and strength of our marketing and distribution of EXPAREL;
the safety, efficacy and other potential advantages over, and availability of, alternative treatments, including, in the case of EXPAREL, a number of products already used to treat pain in the hospital setting; and
distribution and use restrictions imposed by the FDA or to which we agree as part of a mandatory risk evaluation and mitigation strategy or voluntary risk management plan.
Our ability to effectively promote and sell EXPAREL and any product candidates that we may develop, license or acquire in the hospital or ASC marketplace will also depend on pricing and cost effectiveness, including our ability to produce a product at a competitive price and therefore achieve acceptance of the product onto hospital formularies, and our ability to obtain sufficient third-party coverage or reimbursement. Since many hospitals are members of group purchasing organizations, which leverage the purchasing power of a group of entities to obtain discounts based on the collective buying power of the group, our ability to attract customers in the hospital marketplace will also depend on our ability to effectively promote our product candidates to group purchasing organizations. We will also need to demonstrate acceptable evidence of safety and efficacy, as well as relative convenience and ease of administration. Market acceptance could be further limited depending on the prevalence and severity of any expected or unexpected adverse side effects associated with our product candidates.
In addition, the labeling approved by the FDA does not contain claims that EXPAREL is safer or more effective than competitive products and does not permit us to promote EXPAREL as being superior to competing products. Further, the availability of inexpensive generic forms of postsurgical pain management products may also limit acceptance of EXPAREL among physicians, patients and third-party payers. If EXPAREL does not achieve an adequate level of acceptance among physicians, patients and third-party payers, we may not generate meaningful revenues from EXPAREL, and we may not become profitable.return to profitability.
We face significant competition from other pharmaceutical and biotechnology companies. Our operating results will suffer if we fail to compete effectively.
The pharmaceutical and biotechnology industries are intensely competitive and subject to rapid and significant technological change. Our major competitors include organizations such as major multinational pharmaceutical companies, established biotechnology companies and specialty pharmaceutical and generic drug companies. Many of our competitors have greater financial and other resources than we have, such as larger research and development staff, more extensive marketing, distribution, sales and manufacturing organizations and experience, more extensive clinical trial and regulatory experience, expertise in prosecution of intellectual property rights and access to development resources like personnel and technology. As a result, these companies may obtain regulatory approval more rapidly than we are able to and may be more effective in selling and marketing their products. Smaller or early stageearly-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. Our competitors may succeed in developing, acquiring or licensing on an exclusive basis technologies, and drug products and medical devices that are more effective or less costly than EXPAREL, iovera° or any product candidate that we are currently developing or that we may develop, which could render our products obsolete and noncompetitive or significantly harm the commercial opportunity for EXPAREL, iovera° or our product candidates.
As a result of these factors, our competitors may obtain patent protection or other intellectual property rights that may limit our ability to develop other indications for, or commercialize, EXPAREL.EXPAREL, iovera° or our product candidates. Our competitors may also develop drugs or medical devices that are safer, more effective, useful or less costly than ours and may be more successful than us in manufacturing and marketing their products.
EXPAREL competes with well-established products with similar indications. Competing products available for postsurgical pain management include opioids such as morphine, fentanyl, meperidine and hydromorphone, each of which is available generically from several manufacturers, and several of which are available as proprietary products using novel delivery systems. Ketorolac, an NSAID is also available generically in the United StatesU.S. from several manufacturers, and Caldolor (ibuprofen for injection), an NSAID, has been approved by the FDA for pain management and fever in adults. In addition, EXPAREL competes with non-opioid products such as bupivacaine, marcaine, ropivacaine and other anesthetics/analgesics, all of which are also used in the treatment of postsurgical pain and are available as either oral tablets, injectable dosage forms or administered using novel delivery systems. Additional products may be developed for the treatment of acute pain, including new injectable NSAIDs, novel opioids, new formulations of currently available opioids and NSAIDs, long-acting local anesthetics and new chemical entities as well as alternative delivery forms of various opioids and NSAIDs.
EXPAREL also competes with elastomeric bag/catheter devices intended to provide bupivacaine over several days. I-FLOW Corporation (acquired by Kimberly-Clark Corporation in 2009 and spun off into Halyard Health, Inc. in 2014) has marketed these medical devices in the United States since 2004.
Regulatory approval for any approved product is limited by the FDA to those specific indications and conditions for which clinical safety and efficacy have been demonstrated, and allegations of our failure to comply with such approved indications could limit our sales efforts and have a material adverse effect on our business.
The FDA strictly regulates marketing, labeling, advertising and promotion of prescription drugs.drugs and medical devices. These regulations include standards and restrictions for direct-to-consumer advertising, industry-sponsored scientific and educational activities, promotional activities involving the internet and off-label promotion. Any regulatory approval that the FDA grants is limited to those specific diseases and indications for which a product is deemed to be safe and effective by the FDA. For example, the FDA-approved label for EXPAREL does not include an indication in obstetrical paracervical block anesthesia. In addition to the FDA approval required for new formulations, any new indication for an approved product also requires FDA approval. If we are not able to obtain FDA approval for any desired future indications for our products and product candidates, our ability to effectively market and sell our products may be reduced and our business may be adversely affected.
While physicians in the United StatesU.S. may choose, and are generally permitted to prescribe drugs or treatments for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical trials and approved by the regulatory authorities, our ability to promote the products is narrowly limited to those indications that are specifically approved by the FDA. These “off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. Regulatory authorities in the United StatesU.S. generally do not regulate the behavior of physicians in their choice of treatments. Regulatory authorities do, however, restrict communications by pharmaceutical and medical device companies on the subject of off-label use. Although recent court decisions suggest that certain off-label promotional activities may be protected under the First Amendment of the U.S. Constitution, the scope of any such protection is unclear. If our promotional activities fail to comply with the FDA’s regulations or guidelines, we may be subject to warnings from, or enforcement action by, these authorities. In addition, our failure to follow FDA rules and guidelines relating to promotion and advertising may cause the FDA to issue warning letters or untitled letters, bring an enforcement action against us, suspend or withdraw an approved product from the market, require a recall or institute fines or civil fines, or could result in disgorgement of money, operating restrictions, injunctions or criminal prosecution, any of which could harm our reputation and our business.
If we are unable to establish and maintain effective marketing and sales capabilities or enter into agreements with third parties to market and sell EXPAREL, we may be unable to generate product revenues.
We are continuing to build our commercial infrastructure for the marketing, sale and distribution of pharmaceutical products. In order to continue commercializing EXPAREL effectively, we must continue to build our marketing, sales and distribution capabilities. We entered into an agreement with Quintiles for the outsourcing of our specialty sales force, which we then hired as direct employees in January 2013. The establishment, development and training of our sales force and related compliance plans to market EXPAREL is expensive and time consuming. In the event we are not successful in developing our
marketing and sales infrastructure, we may not be able to successfully commercialize EXPAREL, which would limit our ability to generate product revenues.
In addition to our internal marketing and sales efforts, we have entered into agreements with third-party distributors to promote and sell EXPAREL in certain territories. For example, in January 2017, we entered into a co-promotion agreement with DePuy Synthes to market and promote the use of EXPAREL for orthopedic procedures in the US market. The initial termU.S. market, and in June 2018, we entered into an agreement with Nuance to advance the development and commercialization of the agreement commences on January 24, 2017EXPAREL in China. There can be no assurance that such distributors and ends on December 31, 2021, with the option to extend the agreementpromoters will be successful in 12 month increments upon mutual agreement of the parties, subject to certain conditions. marketing and promoting EXPAREL.
We may seek additional distribution arrangements in the future, including arrangements with third-party distributors to commercialize and sell EXPAREL in certain foreign countries. The use of distributors involves certain risks, including risks that such distributors will:
not effectively distribute or support our products;
not provide us with accurate or timely information regarding their inventories, the number of accounts using our products or complaints about our products;
fail to comply with their obligations to us;
fail to comply with laws and regulations to which they are subject, whether in the USU.S. or in foreign jurisdictions;
reduce or discontinue their efforts to sell or promote our products; or
cease operations.
Any such failure may result in decreased sales, which would have an adverse effect on our business.
We rely on third parties to perform many essential services for EXPAREL and iovera° and will rely on third parties for any other products that we commercialize, including services related to customer service support, warehousing and inventory program services, distribution services, contract administration and chargeback processing services, accounts receivable management and cash application services, and financial management and information technology services.commercialize. If these third parties fail to perform as expected or to comply with legal and regulatory requirements, our ability to commercialize EXPAREL and iovera° will be significantly impacted and we may be subject to regulatory sanctions.
We have entered into agreements with third-party service providers to perform a variety of functions related to the sale and distribution of EXPAREL and iovera°, key aspects of which are out of our direct control. These service providers provide key services related to customer service support, warehousing and inventory program services, distribution services, contract administration and chargeback processing services, accounts receivable management and cash application services, financial management and information technology services. In addition, our inventory is stored at two warehouses maintained by two service providers. We substantially rely on these providers as well as other third-party providers that perform services for us, including entrusting our inventories of products to their care and handling. If these third-party service providers fail to comply with applicable laws and regulations, fail to meet expected deadlines or otherwise do not carry out their contractual duties to us, or encounter physical or natural damage at their facilities, our ability to deliver product to meet commercial demand would be significantly impaired. In addition, we may engage third parties to perform various other services for us relating to adverse event reporting, safety database management, fulfillment of requests for medical information regarding our product candidates and related services. If the quality or accuracy of the data maintained by these service providers is insufficient, we could be subject to regulatory sanctions.
Distribution of our DepoFoam-based products, including EXPAREL, requires cold-chain distribution provided by third parties, whereby the product must be maintained between specified temperatures. We and our partners have utilized similar cold-chain processes for DepoCyt(e) and, when it was produced, DepoDur. If a problem occurs in our cold-chain distribution processes, whether through our failure to maintain our products or product candidates between specified temperatures or because of a failure of one of our distributors or partners to maintain the temperature of the products or product candidates, the product or product candidate could be adulterated and rendered unusable. We have obtained limited inventory and cargo insurance coverage for our products. However, our insurance coverage may not reimburse us or may not be sufficient to reimburse us for any expenses or losses we may suffer. This could have a material adverse effect on our business, financial condition, results of operations and reputation.
We willmay need to increase the size of our organization and effectively manage our sales force, and we may experience difficulties in managing growth.
As of December 31, 2016,2019, we had 503606 employees. We may need to expand our personnel resources in order to manage our operations and sales of EXPAREL.EXPAREL and iovera°. Our management, personnel, systems and facilities currently in place may not be
adequate to support this future growth. In addition, we may not be able to recruit and retain qualified personnel in the future, particularly marketing positions, due to competition for personnel among pharmaceutical and medical device businesses, and the failure to do so could have a significant negative impact on our future product revenues and business results. Our need to effectively manage our operations, growth and various projects requires that we:
continue the hiring and training of an effective commercial organization for the commercialization of EXPAREL and iovera°, and establish appropriate systems, policies and infrastructure to support that organization;
continue to establish and maintain effective relationships with distributors and commercial partners for the promotion and sale of our products;
ensure that our distributors, partners, suppliers, consultants and other service providers successfully carry out their contractual obligations, provide high quality results and meet expected deadlines;
manage our development efforts and clinical trials effectively;
expand our manufacturing capabilities and effectively manage our co-production arrangement with Patheon;Thermo Fisher;
continue to carry out our own contractual obligations to our licensors and other third parties; and
continue to improve our operational, financial and management controls, reporting systems and procedures.
We may be unable to successfully implement these tasks on a larger scale and, accordingly, may not achieve our development and commercialization goals. Additionally, these tasks may impose a strain on our administrative and operational infrastructure. If we are unable to effectively manage our growth, our product sales and resulting revenues will be negatively impacted.
We may not be able to manage our business effectively if we are unable to attract and retain key personnel.
We may not be able to attract or retain qualified management and commercial, scientific and clinical personnel due to the intense competition for qualified personnel among biotechnology, pharmaceutical, medical device and other businesses, as well as universities, non-profit research organizations and government entities, particularly in the San Diego, California, the San Francisco Bay Area and northern New Jersey areas.Jersey. If we are not able to attract and retain necessary personnel to accomplish our business objectives, we may experience constraints that will significantly impede the achievement of our development objectives, our ability to raise additional capital and our ability to implement our business strategy.
Our industry has experienced a high rate of turnover of management personnel in recent years. We are highly dependent on the development and manufacturing expertise for our DepoFoam delivery technology and the commercialization expertise of certain members of our senior management. In particular, we are highly dependent on the skills and leadership of our senior management team, including David Stack, our Chief Executive Officer and Chairman, James Scibetta, our President and Charles A. Reinhart, III, our Chief Financial Officer.team. If we lose one or more of these key employees, our ability to successfully implement our business strategy could be seriously harmed. Replacing key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to develop, gain regulatory approval of and commercialize products successfully. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate additional key personnel.
We face potential product liability exposure, and if successful claims are brought against us, we may incur substantial liability for DepoCyt(e)EXPAREL, iovera°, EXPARELDepoCyt(e) or product candidates that we may develop and may have to limit their commercialization.
The use of DepoCyt(e)EXPAREL, iovera°, EXPARELDepoCyt(e) and any product candidates that we may develop, license or acquire in clinical trials and the sale of any products for which we obtain regulatory approval expose us to the risk of product liability claims. Product liability claims might be brought against us by consumers, health care providers or others using, administering or selling our products. We have been a party of these suits in the past and may be again in the future. If we cannot successfully defend ourselves against these claims, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
loss of revenue from decreased demand for our products and/or product candidates;
impairment of our business reputation or financial stability;
costs of related litigation;
substantial monetary awards to patients or other claimants;
diversion of management attention;
loss of revenues;
withdrawal of clinical trial participants and potential termination of clinical trial sites or entire clinical programs; and
the inability to commercialize our products and/or product candidates.
We have obtained limited product liability insurance coverage for our products and our clinical trials with a $10.0 million annual aggregate coverage limit. However, our insurance coverage may not reimburse us, or may not be sufficient to reimburse us, for any expenses or losses we may suffer, including our indemnification obligations to other parties. Moreover, insurance coverage is becoming increasingly expensive, and, in the future, we may not be able to maintain insurance coverage on acceptable terms, at a reasonable cost or in sufficient amounts to protect us against losses due to liability. We intend to expand our insurance coverage to include the sale of additional commercial products upon FDA approval for our product candidates in development, but we may be unable to obtain commercially reasonable product liability insurance for any products approved for marketing, or at all. On occasion, large judgments have been awarded in class action lawsuits based on drugs or medical devices that had unanticipated side effects. A successful product liability claim or series of claims brought against us could cause our stock price to fall and, if judgments exceed our insurance coverage, could decrease our cash and adversely affect our business.
If we fail to manufacture EXPARELour products in sufficient quantities and at acceptable quality and pricing levels, or to fully comply with cGMP regulations, we may face delays in the commercialization of this productthese products or be unable to meet market demand, and may lose potential revenues.
The manufacture of EXPAREL requires significant expertise and capital investment, including the development of advanced manufacturing techniques, process controls and the use of specialized processing equipment. We must comply with federal, state and foreign regulations, including the FDA’s regulations governing cGMP, enforced by the FDA through its facilities inspection program and by similar regulatory authorities in other jurisdictions where we do business. These requirements include, among other things, quality control, quality assurance and the maintenance of records and documentation. The FDA or similar foreign regulatory authorities at any time may implement new standards or change their interpretation and enforcement of existing standards for manufacture, packaging or testing of our products. Any failure by us or our manufacturing partner to comply with applicable regulations may result in fines and civil penalties, suspension of production, product seizure or recall, operating restrictions, imposition of a consent decree, modification or withdrawal of product approval or criminal prosecution and would limit the availability of our product. Any manufacturing defect or error discovered after products have been produced and distributed also could result in significant consequences, including costly recall procedures, re-stocking costs, damage to our reputation and potential for product liability claims.
The FDA requires manufacturers of medical devices to adhere to certain regulations, including the FDA’s QSRs, which requires periodic audits, design controls, quality control testing and documentation procedures, as well as complaint evaluations and investigations. Regulations regarding the development, manufacture and sale of medical products are evolving and are subject to change in the future.
If we are unable to produce the required commercial quantities of EXPARELour products to meet market demand for EXPARELthose products on a timely basis or at all, or if we fail to comply with applicable laws for the manufacturing of EXPAREL,our products, we will suffer damage to our reputation and commercial prospects, and we will lose potential revenues.revenues and we may be required to expend significant resources to resolve any such issues.
We will need to expand our manufacturing operations or outsource such operations to third parties.
To successfully meet future customer demand for EXPAREL and iovera°, we will need to expand our existing commercial manufacturing facilities or establish large-scale commercial manufacturing capabilities. In addition, as our drug development pipeline increases and matures, we will have a greater need for clinical trial and commercial manufacturing capacity. As a result, we must continue to improve our manufacturing processes to allow us to reduce our production costs. We may not be able to manufacture our drugs and/or medical devices at a cost or in quantities necessary to be commercially successful.
The build-up or other expansion of our internal manufacturing capabilities for EXPAREL production in San Diego, California and co-production capabilities at Thermo Fisher’s Swindon, England site, exposes us to significant up-front fixed costs. If market demand for EXPAREL does not align with our expanded manufacturing capacity, we may be unable to offset these costs and to achieve economies of scale, and our operating results may be adversely affected as a result of high operating expenses. Alternatively, if we experience demand for EXPAREL in excess of our estimates, our facilities may be insufficient to support higher production volumes, which could harm our customer relationships and overall reputation. Our ability to meet such excess demand could also depend on our ability to raise additional capital and effectively scale our manufacturing operations.
In addition, the procurement time for the equipment that we use to manufacture EXPAREL requires long lead times. Therefore, we may experience delays, additional or unexpected costs and other adverse events in connection with our capacity expansion projects, including those associated with potential delays in the procurement of manufacturing equipment required to manufacture EXPAREL, including the equipment for the construction of manufacturing suites at Patheon.EXPAREL.
In addition to expanding our internal manufacturing facilities, we may enter into arrangements with third parties to supply, manufacture, package, test and/or store EXPAREL, iovera° or our other products, such as our manufacturing arrangement with Patheon.Thermo Fisher. Entering into such arrangements requires testing and compliance inspections, FDA approvals and development of the processes and facilities necessary for the production of our products. Such arrangements also involve additional risks, many of which would be outside of our control. Such risks include disruptions or delays in production, manufactured products that do not meet our required specifications, the failure of such third-party manufacturers to comply with cGMP regulations or other regulatory requirements, protection of our intellectual property and manufacturing process, loss of control of our complex manufacturing process, inabilities to fulfill our commercial needs and financial risks in connection with our investment in setting up a third-party manufacturing process, such as the substantial capital outlays that were required by us to assist in setting up our manufacturing process at Patheon’s facilities.Thermo Fisher’s facility.
If we are unable to timely achieve and maintain satisfactory production yields and quality, whether through our internal manufacturing capabilities or arrangements with contract manufacturers, our relationships with potential customers and overall reputation may be harmed and our revenues could decrease.
For example, in 2012June 2017, we temporarily ceased the manufacturing of DepoCyt(e) for sales in the European Union to implement a remediation plan to address certain issues noted in an inspection report issued by the MHRA, in July 2012 regarding our DepoCyt(e) manufacturing facility, which is located in a separate building from our EXPAREL manufacturing facility. The assessment report also recommended a selective recall of DepoCyt(e) in European Union member states where DepoCyt(e) is not considered to be an “essential medicinal product,” which contributed to a reduction in product sales of DepoCyt(e) during fiscal year 2012. Although we received notice from the MHRA in January 2013 that our remediation efforts were successful and that we could resumediscontinued production of DepoCyt(e) for sale in Europe, we may be required indue to persistent technical issues specific to the future to ceaseDepoCyt(e) manufacturing operations at our facilities in response to inspection reports or other regulatory actions, and such temporary cessations could result in additional costs or delays in the production and sale of our products.process.
Our San Diego and Fremont facilities in California and the Thermo Fisher facility in Swindon, England are also subject to the risks of a natural or man-made disaster, including earthquakes, floods and fires, or other business disruption.disruptions. In addition, we have obtained limited property and business interruption insurance coverage for our facilitiesmanufacturing sites in San Diego.Diego, Fremont and England. However, our insurance coverage may not reimburse us, or may not be sufficient to reimburse us, for any expenses or losses we may suffer. There can be no assurance that we would be able to meet our requirements for EXPAREL and DepoCyt(e)or iovera° if there were a catastrophic event or failure of our current manufacturing system.systems. If we are required to change or add a new manufacturer or supplier, the process would likely require prior FDA and/or equivalent foreign regulatory authority approval and would be very time consuming. An inability to continue manufacturing adequate supplies of EXPAREL and DepoCyt(e) at our facilities in San Diego, California or at the Thermo Fisher facility in Swindon, England or iovera° at our facility in Fremont, California could result in a disruption in the supply of EXPAREL and DepoCyt(e), respectively,or iovera° to our customers and partners and a breach of our contractual obligations to such counterparties.
Our co-production and other agreements with PatheonThermo Fisher may involve unanticipated expenses and delays, including the need for the PatheonThermo Fisher facilities to receive regulatory approvals required for manufacturing to commence at the PatheonThermo Fisher suites.
We and PatheonThermo Fisher have entered into a Co-Production Agreement, Technical Transfer and Service Agreement and Manufacturing and Supply Agreement. Under these agreements, Patheon will undertakeThermo Fisher undertook certain technical transfer activities and construction services to prepare Patheon’sThermo Fisher’s Swindon, England facility for the manufacture of EXPAREL in two dedicated manufacturing suites.suites, of which one suite received FDA approval in May 2018 and began commercial production in February 2019. We have agreed with Patheon,Thermo Fisher, among other things, to provide them with the process equipment necessary to manufacture EXPAREL in these suites. We have anticipated and budgeted for capital expenditures associated with the two PatheonThermo Fisher suites, including the equipment purchase and construction of the suites as well as payments to be made to Patheon.Thermo Fisher.
The PatheonThermo Fisher facilities must be approved by therequire FDA approval prior to any production and manufacturing of EXPAREL. We currently expect, subject to receipt of regulatory approvals, that the first commercial manufacturing suite at Patheon’s facility will commence commercial production in late 2018. If the construction of the Patheon suitessecond Thermo Fisher suite is delayed, if PatheonThermo Fisher experiences unanticipated cost overruns, or if the Patheon suites doadditional Thermo Fisher suite does not receive or maintain regulatory approvals in the timeframe anticipated (if at all), this could have a material adverse effect on our business, financial position and results of operations.
Further, if and when the Patheon facilities are constructed and have received the required FDA approvals, the production under these agreements involve additional risks, many of which would be outside of our control, such as disruptions or delays in production, manufactured products that do not meet our required specifications, the failure of PatheonThermo Fisher to comply with cGMP regulations or other regulatory requirements, protection of our intellectual property and manufacturing process, loss of control of our complex manufacturing process and inabilities to fulfill our commercial needs.
We rely on third parties for the timely supply of specified raw materials and equipment for the manufacture of DepoCyt(e)EXPAREL and EXPAREL.iovera°. Although we actively manage these third-party relationships to provide continuity and quality, some events which are beyond our control could result in the complete or partial failure of these goods and services. Any such failure could have a material adverse effect on our financial condition and operations.
We purchase certain raw materials and equipment from various suppliers in order to manufacture our products. The acquisition of certain of these materials may require considerable lead times, and our ability to source such materials is also dependent on logistics providers. If we are unable to source the required raw materials and equipment from our suppliers on a timely basis and in accordance with our specifications, we may experience delays in manufacturing and may not be able to meet our customers’ or partners’ demands for our products. In addition, we and our third-party suppliers must comply with federal, state and foreign regulations, including cGMP regulations, and any failure to comply with applicable regulations, or failure of government agencies to provide necessary authorizations, may harm our ability to manufacture and commercialize our products on a timely and competitive basis, which could result in decreased product sales and lower revenues.
Our future growth depends on our ability to identify, develop, acquire or in-license products and if we do not successfully identify, develop, acquire or in-license related product candidates or integrate them into our operations, we may have limited growth opportunities.
An important part of our business strategy is to continue to develop a pipeline of product candidates by developing, acquiring or in-licensing products, businesses or technologies that we believe are a strategic fit with our focus on the hospital marketplace, including our current product candidates DepoMLX and DepoTXA.marketplace. However, these business activities may entail numerous operational and financial risks, including:
significant capital expenditures;
difficulty or inability to secure financing to fund development activities for such development, acquisition or in-licensed products or technologies;
incurrence of substantial debt or dilutive issuances of securities to pay for development, acquisition or in-licensing of new products;
the successful integration of acquired products, businesses or technologies into our operations, and achieving the expected benefits and synergies from such acquisitions;
disruption of our business and diversion of our management’s time and attention;
higher than expected development, acquisition or in-license and integration costs;
exposure to unknown liabilities;
difficulty and cost in combining the operations and personnel of any acquired businesses with our operations and personnel;
inability to retain key employees of any acquired businesses;
difficulty entering markets in which we have limited or no direct experience;
difficulty in managing multiple product development programs; and
inability to successfully develop new products or clinical failure.
We have limited resources to identify and execute the development, acquisition or in-licensing of products, businesses and technologies and integrate them into our current infrastructure. We may compete with larger pharmaceutical and medical device companies and other competitors, including public and private research organizations, academic institutions and government agencies, in our efforts to establish new collaborations and in-licensing opportunities. These competitors may have access to greater financial resources, research and development staffs and facilities than us and may have greater expertise in identifying and evaluating new opportunities. We may not be successful in locating and acquiring or in-licensing additional desirable product candidates on acceptable terms or at all. We may also not be successful in developing or commercializing our current product candidates
Our business involves the use of hazardous materials and we must comply with environmental laws and regulations, which can be expensive and restrict how we do business.
Our manufacturing activities involve the controlled storage, use and disposal of hazardous materials, including the components of our products, product candidates and other hazardous compounds. We are subject to federal, state and local laws and regulations governing the use, manufacture, storage, handling, release and disposal of, and exposure to, these hazardous materials. Violation of these laws and regulations could lead to substantial fines and penalties. Although we believe that our safety procedures for handling and disposing of these materials comply with the standards prescribed by these laws and regulations, we cannot eliminate the risk of accidental contamination or injury from these materials or unintended failure to comply with these laws and regulations. In the event of an accident or failure to comply with these laws and regulations, state or federal authorities may curtail our use of these materials and interrupt our business operations. In addition, we could become subject to potentially material liabilities relating to the investigation and cleanup of any contamination, whether currently unknown or caused by future releases.
Our business and operations would suffer in the event of system failures.
Despite the implementation of security measures, our internal computer systems are vulnerable to damage from computer viruses, human error, unauthorized access, natural disasters, intentional acts of vandalism, terrorism, war and telecommunication and electrical failures. Any system failure, accident or security breach that causes interruptions in our operations could result in a material disruption of our product development programs. For example, the loss of clinical trial data from completed clinical trials for EXPAREL could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we may incur liability, reputation damage and harm to our business operations.
Any collaboration arrangements that we may enter into in the future may not be successful, which could adversely affect our ability to develop and commercialize our product candidates.
Our business model is to commercialize our product candidatesproducts in the United StatesU.S. and generally to seekabroad, occasionally seeking collaboration arrangements with pharmaceutical or biotechnology companies for the development or commercialization of our product candidatesproducts in the rest of the world.other countries. Accordingly, we may enter into collaboration arrangements in the future on a selective basis. Any future collaboration arrangements that we enter into may not be successful. The success of our collaboration arrangements will depend heavily on the efforts and activities of our collaborators. Collaborators generally have significant discretion in determining the efforts and resources that they will apply to these collaboration arrangements.
Disagreements between parties to a collaboration arrangement regarding clinical development and commercialization matters can lead to delays in the development process or commercializing the applicable product candidate and, in some cases, termination of the collaboration arrangement. These disagreements can be difficult to resolve if neither of the parties has final decision makingdecision-making authority.
Collaborations with pharmaceutical and/or medical device companies and other third parties often are terminated or allowed to expire by the other party. Any such termination or expiration would adversely affect us financially and could harm our business reputation.
Clinical trials may fail to demonstrate the safety and efficacy of our drug products or medical devices, which could prevent or significantly delay obtaining regulatory approval.
Prior to receiving approval to commercialize any of our drug products or medical devices, we must demonstrate with substantialscientifically appropriate and statistically sound evidence from well-controlled clinical trials, and to the satisfaction of the FDA, other regulatory authorities in the United States,U.S., and other countries, that each of the products is both safe and effective. For each drug product, we will need to demonstrate its efficacy and monitor its safety throughout the process. If such development is unsuccessful, our business and reputation would be harmed and our stock price would be adversely affected.
All of our drug and medical device products are prone to the risks of failure inherent in drug development. Clinical trials of new drug and medical device products sufficient to obtain regulatory marketing approval are expensive and take years to complete. We may not be able to successfully complete clinical testing within the time frame we have planned, or at all. We may experience numerous unforeseen events during, or as a result of, the clinical trial process which could delay or prevent us from receiving regulatory approval or commercializing our drug products. In addition, the results of pre-clinical studies and early-stage clinical trials of our drug products do not necessarily predict the results of later-stage clinical trials. Later-stage clinical trials may fail to demonstrate that a drug product is safe and effective despite having progressed through initial clinical testing. Even if we
believe the data collected from clinical trials of our drug products is promising, such data may not be sufficient to support approval by the FDA or any other USU.S. or foreign regulatory approval authority. Pre-clinical and clinical data can be interpreted in different ways.
Accordingly, the FDA officialsor other regulatory authorities could interpret such data in different ways than we or our partners do, which could delay, limit or prevent regulatory approval. The FDA, other regulatory authorities, our institutional review boards, our contract research organizations or we ourselves may suspend or terminate our clinical trials for our drug products.products and medical devices. Any failure or significant delay in completing clinical trials for our drug products or medical devices, or in receiving regulatory approval for the sale of any drugs or medical devices resulting from our drug products, may severely harm our business and reputation. Even if we receive FDA and other regulatory approvals, our drug and medical device products may later exhibit adverse effects that may limit or prevent their widespread use, may cause the FDA to revoke, suspend or limit their approval, or may force us to withdraw products derived from those drug or medical device products from the market.
Our dependence on contract research organizations could result in delays in and additional costs for our drug development efforts.
We may rely on contract research organizations, or CROs, to perform preclinical testing and clinical trials for drug candidates that we choose to develop without a collaborator. If the CROs that we hire to perform our preclinical testing and clinical trials or our collaborators or licensees do not meet deadlines, do not follow proper procedures or a conflict arises between us and our CROs, our preclinical testing and clinical trials may take longer than expected, may be delayed or may be terminated. If we were forced to find a replacement CRO to perform any of our preclinical testing or clinical trials, we may not be able to find a suitable replacement on favorable terms, if at all. Even if we were able to find another CRO to perform a preclinical test or clinical trial, any material delay in a test or clinical trial may result in significant additional expenditures that could adversely affect our operating results. Events such as these may also delay regulatory approval for our drug candidates or our ability to commercialize our products.
We depend on clinical investigators and clinical sites to enroll patients in our clinical trials and sometimes other third parties to manage the trials and to perform related data collection and analysis, and, as a result, we may face costs and delays outside of our control.
We rely on clinical investigators and clinical sites to enroll patients and sometimes third parties to manage our trials and to perform related data collection and analysis. However, we may be unable to control the amount and timing of resources that the clinical sites which conduct the clinical testing may devote to our clinical trials.
Our clinical trials may be delayed or terminated due to the inability of our clinical investigators to enroll enough patients. Patient enrollment depends on many factors, including the size of the patient population, the nature of the trial protocol, the proximity of patients to clinical sites and the eligibility criteria for the trial. If our clinical investigators and clinical sites fail to enroll a sufficient number of patients in our clinical trials or fail to enroll them on our planned schedule, we may face increased costs, delays or termination of the trials, which could delay or prevent us from obtaining regulatory approvals for our product candidates.
Our agreements with clinical investigators and clinical sites for clinical testing and for trial management services place substantial responsibilities on these parties, which could result in delays in, or termination of, our clinical trials if these parties fail to perform as expected. For example, if any of our clinical trial sites fail to comply with FDA-approved GCPs, we may be unable to use the data gathered at those sites. If these clinical investigators, clinical sites or other third parties do not carry out their contractual duties or obligations or fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to their failure to adhere to our clinical protocols or for other reasons, our clinical trials may be extended, delayed or terminated, and we may be unable to obtain regulatory approval for, or successfully commercialize, our product candidates.
We are subject to periodic litigation, which could result in losses or unexpected expense of time and resources.
From time to time, we are called upon to defend ourselves against lawsuits relating to our business. Due to the inherent uncertainties of litigation, we cannot accurately predict the ultimate outcome of any such proceedings. See Item 3 Legal Proceedings in Part I of this Form 10-K.Annual Report. An unfavorable outcome in either of these or other proceedings could have an adverse impact on our business, financial condition and results of operations. In addition, any significant litigation in the future, regardless of its merits, could divert management’s attention from our operations and result in substantial legal fees. In addition, if our stock price is volatile, we may become involved in additional securities class action lawsuits in the future. Any litigation could result in substantial costs and a diversion of management’s attention and resources that are needed to successfully run our business.
Regulatory Risks
We
In April 2015, we received a subpoena from the USU.S. Department of Justice, USU.S. Attorney’s Office for the District of New Jersey, requiring the production of a broad range of documents pertaining to marketing and promotional practices related to EXPAREL. We are cooperating with the government’s inquiry. We cannot estimate what impactcan make no assurances as to the time or resources that will need to be devoted to this inquiry andor its final outcome, or the impact, if any, results fromof this inquiry or any proceedings could have on our business, financial condition, results of operations or cash flows. Cooperation with this inquiry may divert the attention of management and require the devotion of a substantial amount of time and resources. The existence of the inquiry could also adversely impact our sales activity or our customers’ perception of us or EXPAREL. Any of these impacts could have a material adverse effect on our business, financial condition, results of operations and cash flows.
In December 2019, we reached an agreement in principle with the Department of Justice and more than one state Attorney General’s office (the “Plaintiffs”) on a proposal for a global civil settlement in the amount of $3.5 million, subject to accrual of interest on the settlement amount from the date of the agreement in principle, negotiation of a definitive settlement agreement and other contingencies. As part of the settlement, Pacira will admit no wrongdoing and will explicitly deny the Plaintiffs’ allegations. Pacira has been given assurances that, if the parties can agree to negotiation of the settlement, this will conclude the investigation that originated from the U.S. Department of Justice subpoena in April 2015.
If a final settlement cannot be reached, and as a result of this inquiry that started with the April 2015 subpoena, proceedings are initiated and we are found to have violated one or more applicable laws, we may be subject to significant liability, including without limitation, civil fines, criminal fines and penalties, civil damages and exclusion from federal funded healthcare programs such as Medicare and Medicaid, as well as potential liability under the federal False Claims Act and state false claims acts, and/or be required to enter into a corporate integrity or other settlement with the government, any of which could materially affect our reputation, business, financial condition, results of operations and cash flows. Conduct giving rise to such liability could also form the basis for private civil litigation by third-party payors or other persons allegedly harmed by such conduct. In addition, if some of our existing business practices are challenged as unlawful, we may have to change those practices, including changes and impacts on the practices of our sales force, which could also have a material adverse effect on our business, financial condition, results of operations and cash flows.
Our business could be materially adversely affected if the FDA determines that we are promoting or have in the past promoted the “Off-label” use of drugs.our products.
The FDA strictly regulates marketing, labeling, advertising and promotion of prescription drugs.drugs and medical devices. These regulations include standards and restrictions for direct-to-consumer advertising, industry-sponsored scientific and educational activities, promotional activities involving the internet and off-label promotion. According to these regulations, companies may not promote drugs or medical devices for “Off-label” uses—that is, uses that are not described inconsistent with the product’s labeling and that differ from those that were approved by the FDA. For example, the FDA-approved label for EXPAREL does not include an indication in obstetrical paracervical block anesthesia. In addition to the FDA approval required for new formulations or device enhancements, any new indication for an approved product also requires FDA approval. If we are not able to obtain FDA approval for any desired future indications for our products and product candidates, our ability to effectively market and sell our products may be reduced and our business may be adversely affected.
While physicians in the United StatesU.S. may choose, and are generally permitted to prescribe drugs and/or medical devices for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical trials and approved by the regulatory authorities, under the FDA’s regulations our ability to promote the products is narrowly limited to those indications that are approved by the FDA. “Off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. Regulatory authorities in the United StatesU.S. generally do not regulate the behavior of physicians in their choice of treatments. Regulatory authorities do, however, restrict communications by pharmaceutical and medical device companies on the subject of off-label use. Although recent court decisions suggest that certain off-label promotional activities may be protected under the First Amendment of the U.S. Constitution, the scope of such protection is unclear. Moreover, while we promote our products consistent with what we believe to be the approved indication for our drugs and medical devices, the FDA may disagree. If the FDA determines that our promotional activities fail to comply with the FDA’s regulations or guidelines, we may be subject to warnings from, or enforcement action by, these authorities. In addition, our failure to follow FDA rules and guidelines relating to promotion and advertising may cause the FDA to issue warning letters or untitled letters, bring an enforcement action against us, suspend or withdraw an approved product from the market, require a recall or institute fines or civil fines, or could result in disgorgement of money, operating restrictions, injunctions or criminal prosecution, any of which could harm our reputation and our business.
In September 2014, we received a warning letter from the OPDP pertaining to certain promotional aspects of EXPAREL. We took actions to immediately address the FDA’s concerns and minimize further disruption to our business. Ultimately, however, in September 2015, we, along with two independent physicians, filed a lawsuit in federal court against the FDA and other governmental defendants seeking to exercise our lawful rights to communicate truthful and non-misleading information about EXPAREL. The complaint outlined our belief that the FDA’s warning letter received in September 2014 and regulations restricting our truthful and non-misleading speech about EXPAREL violateviolated the Administrative Procedure Act and the First and
Fifth Amendments of the USU.S. Constitution. The lawsuit sought a declaration and injunctive relief to permit us to promote EXPAREL consistent with its approved indication and pivotal trials that supported FDA approval. On December 15, 2015, we announced that the FDA had formally withdrawn the September 2014 Warning Letter via a “Rescission Letter,” and that the FDA and Pacira had reached an amicable resolution of the lawsuit. As part of the resolution of this matter, the FDA confirmed that EXPAREL was broadly approved for “administration into the surgical site to productproduce postsurgical analgesia” in a variety of surgeries not limited to those studied in its pivotal trials. The FDA also approved a labeling supplement for EXPAREL that further clarified that EXPAREL was not limited to any specific surgery type or site, that the proper dosage and administration of EXPAREL is based on various patient and procedure-specific factors, that there was a significant treatment effect for EXPAREL compared to placebo over the first 72 hours in the pivotal hemorrhoidectomy trial and that EXPAREL may be admixed with bupivacaine, provided certain medication ratios are observed. The Warning Letter and labeling supplement only applied to the infiltration indication that was approved at that time, and does not apply to the interscalene brachial plexus nerve block indication approved in April 2018. We and the FDA have agreed that, in future interactions, the parties will deal with each other in an open, forthright and fair manner.
We are unable to predict whether any future regulatory actions will have an effect on EXPARELour product sales, and even if such actions are ultimately resolved favorably, our sales may suffer due to reputational or other concerns. We can make no assurances that we will not receive FDA warning letters in the future or be subject to other regulatory action. As noted above, any regulatory violation or allegations of a violation may have a material adverse effect on our reputation and business.
We may not receive regulatory approval for any of our product candidates, or the approval may be delayed for various reasons, including successful challenges to the FDA’s interpretation of Section 505(b)(2), which would have a material adverse effect on our business and financial condition.
We may experience delays in our efforts to obtain regulatory approval from the FDA for any of our product candidates, and there can be no assurance that such approval will not be delayed, or that the FDA will ultimately approve these product candidates. Although the FDA’s longstanding position has been that the Agency may rely upon prior findings of safety or effectiveness to support approval of a 505(b)(2) application, this policy has been controversial and subject to challenge in the past. If the FDA’s policy is successfully challenged administratively or in court, we may be required to seek approval of our products via full NDAs that contain a complete data package demonstrating the safety and effectiveness of our product candidates, which would be time-consuming, expensive and would have a material adverse effect on our business and financial condition.
The FDA, as a condition of the EXPAREL NDA approval on October 28, 2011, has required us to study EXPAREL in pediatric patients.patients as a post-marketing requirement. We have agreed to a trial timeline where over several years, we will study successive pediatric patient populationssubpopulations. In December 2019, we announced positive results for our extended pharmacokinetic and safety study for local analgesia in descending order startingchildren aged 6 to 17 undergoing cardiovascular or spine surgeries. Those positive results will provide the foundation for an sNDA. We are also working with 12-18 year olds and ending with children under two yearsthe FDA to define a program to study the administration of age.EXPAREL as a nerve block in the pediatric setting. These trials will be expensive and time consuming and we are required to meet the timelines for submission of protocols and data and for completion as agreed with the FDA, and we may be delayed in meeting such timelines. We are required to conduct these trials even if we believe that the costs and potential benefits of conducting the trials are not warranted from a scientific or financial perspective. The failure to conduct these pediatric trials or to meet applicable deadlines could result in the imposition of sanctions, including, among other things, issuance of warnings letters or imposition of seizures or injunctions.
For iovera° and any other potential medical device, we must obtain clearance or approval from the FDA or other regulatory authorities prior to introducing a new product or a modification to an existing product. The regulatory clearance process may result in substantial delays, unexpected or additional costs and other unforeseen factors and limitations on the types and uses of products we would be able to commercialize, any of which could have a material adverse effect on our business and financial condition.
In the U.S., before we are able to market a new medical device, or a new use, claim for or significant modification to an existing medical device, we generally must first receive clearance or approval from the FDA and certain other regulatory authorities. Many foreign jurisdictions outside the U.S. also require clearance, approval or compliance with certain standards before a medical device or other product can be marketed. The process of obtaining regulatory clearances and approvals to market a medical device can be costly, time consuming, involve rigorous pre-clinical and clinical testing, require changes in products or result in limitations on the indicated uses of products. There can be no assurance that these clearances and
approvals will be granted on a timely basis, if at all. In addition, once a medical device has been cleared or approved, a new clearance or approval may be required before the medical device may be modified, its labeling changed or marketed for a different use. Medical devices are cleared or approved for one or more specific intended uses and promoting a device for an off-label use could result in government enforcement action. Furthermore, a product approval or clearance can be withdrawn or limited due to unforeseen problems with the medical device or issues relating to its application. The regulatory clearance and approval process may result in, among other things, delayed, if at all, realization of product net sales, substantial additional costs and limitations on the types of products we may bring to market or their indicated uses, any one of which could have a material adverse effect on our business, results of operations, financial condition and cash flows.
The FDA may determine that EXPARELour products or any of our product candidates have undesirable side effects.
If concerns are raised regarding the safety of a new product candidate as a result of undesirable side effects identified during clinical testing, the FDA may decline to approve the drug at the end of the NDA review periodor medical device or issue a letter requesting additional data or information prior to making a final decision regarding whether or not to approve the drug.product. The number of such requests for additional data or information issued by the FDA in recent years has increased, and resulted in substantial delays in the approval of several new drugs.drugs and medical devices. Undesirable side effects caused by EXPARELour products or any product candidate could also result in the inclusion of unfavorable information in our product labeling, imposition of distribution or use restrictions, a requirement to conduct post-market studies or to implement a risk evaluation and mitigation strategy, denial, suspension or withdrawal of regulatory approval by the FDA or other regulatory authorities for any or all targeted indications, and in turn prevent us from commercializing and generating revenues from the sale of EXPAREL, iovera° or any product candidate.
For example, the side effects observed in the EXPAREL clinical trials completed to date include nausea and vomiting. In addition, the class of drugs that EXPAREL belongs to has been associated with nervous system and cardiovascular toxicities at high doses. We cannot be certain that these side effects and others will not be observed in the future, or that the FDA will not require additional trials or impose more severe labeling restrictions due to these side effects or other concerns. The active component of EXPAREL is bupivacaine and bupivacaine infusions have been associated with the destruction of articular cartilage, or chondrolysis. Chondrolysis has not been observed in clinical trials of EXPAREL, but we cannot be certain that this side effect will not be observed in the future.
Following approval of EXPAREL, iovera° or any of our product candidates, if we or others later identify previously unknown undesirable side effects caused by such products, if known side effects are more frequent or severe than in the past, or if we or others detect unexpected safety signals for such products or any products perceived to be similar to such products:
regulatory authorities may require the addition of unfavorable labeling statements, specific warnings or contraindications (including boxed warnings);
regulatory authorities may suspend or withdraw their approval of the product, or require it to be removed from the market;
regulatory authorities may impose restrictions on the distribution or use of the product;
we may be required to change the way the product is administered, conduct additional clinical trials, reformulate the product, change the labeling of the product or change or obtain re-approvals of manufacturing facilities;
sales of the product may be significantly decreased from projected sales;
we may be subject to government investigations, product liability claims and litigation; and
our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of EXPARELour products or any of our product candidates and could substantially increase our commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from its sale.
Regulatory approval for any approved product is limited by the FDA to those specific indications and conditions for which clinical safety and efficacy have been demonstrated.
Any regulatory approval is limited to those specific diseases and indications for which a product is deemed to be safe and effective by the FDA. For example, the FDA-approved label for EXPAREL does not include an indication in obstetrical paracervical block anesthesia. In addition to the FDA approval required for new formulations,products or product enhancements, any new indication for an approved product also requires FDA approval. If we are not able to obtain FDA approval for any desired
future indications for our products and product candidates, our ability to effectively market and sell our products may be reduced and our business may be adversely affected.
While physicians may choose to prescribe drugs and medical devices for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical trials and approved by the regulatory authorities, our ability to promote the products is limited to those indications that are specifically approved by the FDA. These “off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. Regulatory authorities in the United States generally do not regulate the behavior of physicians in their choice of treatments. Regulatory authorities do, however, restrict communications by pharmaceutical companies on the subject of off-label use. Although recent court decisions suggest that certain off-label promotional activities may be protected under the First Amendment, the scope of any such protection is unclear. If our promotional activities fail to comply with the FDA’s regulations or guidelines, we may be subject to warnings from, or enforcement action by, these authorities. In addition, our failure to follow FDA rules and guidelines relating to promotion and advertising may cause the FDA to issue warning letters or untitled letters, suspend or withdraw an approved product from the market, require a recall or institute fines or civil fines or could result in disgorgement of money, operating restrictions, injunctions or criminal prosecution, any of which could harm our business.
If we do not comply with federal, state and foreign laws and regulations relating to the health care business, we could face substantial penalties.
We and our customers are subject to extensive regulation by the federal government, and the governments of the states and foreign countries in which we may conduct our business. In the United States,U.S., the laws that directly or indirectly affect our ability to operate our business include the following:
the Federal Anti-Kickback Law, which prohibits persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce either the referral of an individual or furnishing or arranging for a good or service for which payment may be made under federal health care programs such as Medicare and Medicaid;
other Medicare laws and regulations that prescribe the requirements for coverage and payment for services performed by our customers, including the amount of such payment;
the Federal False Claims Act, which imposes civil and criminal liability on individuals and entities who submit, or cause to be submitted, false or fraudulent claims for payment to the government;
the Federal False Statements Act, which prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement in connection with delivery of or payment for health care benefits, items or services; and
various state laws that impose similar requirements and liability with respect to state healthcare reimbursement and other programs.
If our operations are found to be in violation of any of the laws and regulations described above or any other law or governmental regulation to which we or our customers are or will be subject, we may be subject to civil and criminal penalties, damages, fines, exclusion from the Medicare and Medicaid programs and the curtailment or restructuring of our operations. Similarly, if our customers are found to be non-compliant with applicable laws, they may be subject to sanctions, which could also have a negative impact on us. Any penalties, damages, fines, curtailment or restructuring of our operations would adversely affect our ability to operate our business and our financial results. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses, divert our management’s attention from the operation of our business and damage our reputation.
The design, development, manufacture, supply and distribution of EXPAREL and DepoCyt(e) isare highly regulated and technically complex.
The design, development, manufacture, supply and distribution of EXPAREL and DepoCyt(e) isare all highly regulated. We, along with our third-party providers, must comply with all applicable regulatory requirements of the FDA and foreign authorities. In addition, the facilities used to manufacture, store and distribute our productsEXPAREL are subject to inspection by regulatory authorities at any time to determine compliance with applicable regulations.
The manufacturing techniques and facilities used for the manufacture and supply of our products must be operated in conformity with cGMP and other FDA and MHRA regulations, including potentially prior regulatory approval. In addition, any expansion of our existing manufacturing facilities or the introduction of any new manufacturing facilities, including the manufacturing suites to be constructed at Patheon’sThermo Fisher’s facility, also require conformity with cGMP and other FDA and MHRA regulations. In complying with these requirements, we, along with our co-production partners and suppliers, must continually expend time, money and effort in production, record keeping and quality assurance and control to ensure that our products meet applicable specifications and other requirements for safety, efficacy and quality. In addition, we, along with our co-production partners and suppliers, are subject to unannounced inspections by the FDA, MHRA and other regulatory authorities.
Any failure to comply with regulatory and other legal requirements applicable to the manufacture, supply and distribution of our products could lead to remedial action (such as recalls), civil and criminal penalties and delays in manufacture, supply and distribution of our products. For instance, in July 2012, the MHRA issued its inspection report in which the MHRA noted certain critical and major failures to comply with the Principles and Guidelines of Good Manufacturing Practices related to our DepoCyt(e) manufacturing facility. We responded to the MHRA regarding these inspectional observations, completed implementation of our proposed remediation plan and were reinspected by the MHRA in December 2012. In January 2013, we received notice from the MHRA that our remediation efforts were successful and that we could recommence manufacturing DepoCyt(e) for Europe.
The design, development, manufacture, supply and distribution of EXPAREL and DepoCyt(e) isare all highly complex. As part of our routine stability monitoring that occurred in October 2016, it came to our attention that one of two test batches of EXPAREL made in early 2016 had fallen slightly out of specification for one of the 21 acceptance criteria measured during testing. This test result was unexpected and suggestive of some deviation from a consistency of manufacturing output. As a result, we have been in discussions with the FDA about both a modification of that specification as well as the potential development of a new analytical test for this attribute. Until that process is completed, we have agreed with the FDA that all EXPAREL manufactured beginning in October 2016 will include 12 month expiration dating. In connection with this issue, in 2016, we recorded a $20.7 million charge to cost of goods sold. If we are unable to manufacture EXPAREL in compliance with our highly complex specifications in the future, we may be subject to product exchanges, significant costs and charges, supply constraints or other corrective measures.
If we fail to comply with the extensive regulatory requirements to which we and our products EXPAREL and DepoCyt(e), are subject, such products could be subject to restrictions or withdrawal from the market and we could be subject to penalties.
The testing, manufacturing, quality control, labeling, safety, effectiveness, advertising, promotion, storage, sales, distribution, import, export and marketing, among other things, of EXPAREL, iovera° and our products EXPAREL and DepoCyt(e)product candidates are subject to extensive regulation by governmental authorities in the United StatesU.S. and elsewhere throughout the world. Quality control and manufacturing procedures regarding EXPAREL and DepoCyt(e)our product candidates must conform to cGMP. Regulatory authorities, including the FDA and the MHRA, periodically inspect manufacturing facilities to assess compliance with cGMP. Our failure, or the failure
of any contract manufacturers with whom we may work in the future, to comply with the laws administered by the FDA, the MHRA or other governmental authorities could result in, among other things, any of the following:
product recall or seizure;
suspension or withdrawal of an approved product from the market;
interruption of production;
reputational concerns of our customers or the medical community;
operating restrictions;
warning letters;
injunctions;
refusal to permit import or export of an approved product;
refusal to approve pending applications or supplements to approved applications that we submit;
denial of permission to file an application or supplement in a jurisdiction;
consent decrees;
suspension or termination of ongoing clinical trials;
fines and other monetary penalties;
criminal prosecutions; and
unanticipated expenditures.
If the government or third-party payers fail to provide coverage and adequate coverage and payment rates for EXPAREL, DepoCyt(e)iovera° or any future products, or if hospitals or ASCs choose to use therapies that are less expensive, our revenue and prospects for profitability will be limited.
In both domestic and foreign markets, sales of our existing products and any future products will depend in part upon the availability of coverage and reimbursement from third-party payers. Such third-party payers include government health programs such as Medicare and Medicaid, managed care providers, private health insurers and other organizations. Coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available. Assuming coverage is approved, the resulting reimbursement payment rates might not be adequate. In particular, many USU.S. hospitals and ASCs receive a fixed reimbursement amount per procedure for certain surgeries and other treatment therapies they perform. Because this amount may not be based on the actual expenses the hospital or ASC incurs, hospitalsthese sites may choose to use therapies which are less expensive when compared to our product candidates. Although hospitals currentlyand ASCs may receive separate reimbursement for EXPAREL, used in the hospital outpatient setting, EXPAREL, DepoCyt(e)iovera° or any product candidates that we may develop, in-license or acquire, if approved, will face competition from other therapies and drugs for these limited hospital and ASC financial resources. We may need to conduct post-marketing studies in order to demonstrate the cost-effectiveness of any future products to the satisfaction of hospitals, ASCs, other target
customers and their third-party payers. Such studies might require us to commit a significant amount of management time, financial and other resources. Our future products might not ultimately be considered cost-effective. Adequate third-party coverage and reimbursement might not be available to enable us to maintain price levels sufficient to realize an appropriate return on investment in product development.
Third-party payers, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. For example, third-party payers may limit the indications for which our products will be reimbursed to a smaller set of indications than we believe is appropriate or limit the circumstances under which our products will be reimbursed to a smaller set of circumstances than we believe is appropriate. In addition, in the United States,U.S., no uniform policy of coverage and reimbursement for drug or medical device products exists among third-party payers. Therefore, coverage and reimbursement for drug products can differ significantly from payer to payer.
Further, we believe that future coverage and reimbursement will likely be subject to increased restrictions both in the United StatesU.S. and in international markets, as federal, state and foreign governments continue to propose and pass new legislation designed to reduce or contain the cost of healthcare. Third-party coverage and reimbursement for our products or product candidates for which we receive regulatory approval may not be available or adequate in either the United StatesU.S. or international markets, which could have a negative effect on our business, results of operations, financial condition and prospects.
Public concern regarding the safety of drug products such as EXPAREL and medical device products such as iovera° could result in the inclusion of unfavorable information in our labeling, or require us to undertake other activities that may entail additional costs.
In light of widely publicized events concerning the safety risk of certain drug products, the FDA, members of Congress, the Government Accountability Office, medical professionals and the general public have raised concerns about potential drug and medical device safety issues. These events have resulted in the withdrawal of drug and medical device products, revisions to drug labeling that further limitlimits use of the drug and medical device products and the establishment of risk management programs that may, for example, restrict distribution of drug or medical device products after approval. The Food and Drug Administration Amendments Act of 2007, or FDAAA, grants significant expanded authority to the FDA, much of which is aimed at improving the safety of drug and medical device products before and after approval. In particular, the FDAAA authorizes the FDA to, among other things, require post-approval studies and clinical trials, mandate changes to drugproduct labeling to reflect new safety information and require risk evaluation and mitigation strategies for certain drugs and medical devices, including certain currently approved drugs.drugs and medical devices. The FDAAA also significantly expands the federal government’s clinical trial registry and results databank, which we expect will result in significantly increased government oversight of clinical trials. Under the FDAAA, companies that violate these and other provisions of the new law are subject to substantial civil monetary penalties, among other regulatory, civil and criminal penalties. The increased attention to drug safety issues may result in a more cautious approach by the FDA in its review of data from our clinical trials. Data from clinical trials may receive greater scrutiny, particularly with respect to safety, which may make the FDA or other regulatory authorities more likely to require additional preclinical studies or clinical trials. If the FDA requires us to provide additional clinical or preclinical data for EXPAREL or iovera°, the indications for which this product candidate wasthese products were approved may be limited or there may be specific warnings or limitations on dosing, and our efforts to commercialize EXPAREL or iovera° may be otherwise adversely impacted.
Risks Related to Intellectual Property
The patents and the patent applications that we have covering our products are limited to specific injectable formulations, processes and uses of drugs encapsulated in our DepoFoam drug delivery technology and our market opportunity for our product candidates may be limited by the lack of patent protection for the active ingredient itself and other formulations and delivery technology and systems that may be developed by competitors.
The active ingredientsingredient in EXPAREL and DepoCyt(e) are bupivacaine and cytarabine, respectively.is bupivacaine. Patent protection for the bupivacaine and cytarabine molecules themselves has expired and generic immediate-release products are available. As a result, competitors who obtain the requisite regulatory approval can offer products with the same active ingredientsingredient as EXPAREL and DepoCyt(e) so long as the competitors do not infringe any process, use or formulation patents that we have developed for these drugs encapsulated in our DepoFoam drug delivery technology.
For example, we are aware of at least one long-acting injectable bupivacaine product in development which utilizes an alternative delivery system to EXPAREL. Such a product is similar to EXPAREL in that it also extends the duration of effect of bupivacaine, but achieves this clinical outcome using a completely different drug delivery system as compared to our DepoFoam drug delivery technology.
The number of patents and patent applications covering products in the same field as EXPAREL indicates that competitors have sought to develop and may seek to market competing formulations that may not be covered by our patents and patent applications. The commercial opportunity for EXPAREL could be significantly harmed if competitors are able to develop and commercialize alternative formulations of bupivacaine that are long-acting but outside the scope of our patents.
Because EXPAREL has been approved by the FDA, one or more third parties may challenge the patents covering this product, which could result in the invalidation or unenforceability of some or all of the relevant patent claims. For example, if a third-party files an Abbreviated New Drug Application, or ANDA, for a generic drug product containing bupivacaine and relies in whole or in part on studies conducted by or for us, the third-party will be required to certify to the FDA that either: (i) there is no patent information listed in the FDA’s Orange Book with respect to our NDA for EXPAREL; (ii) the patents listed in the Orange Book have expired; (iii) the listed patents have not expired, but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patents are invalid or will not be infringed by the manufacture, use or sale of the third-party’s generic drug product. A certification that the new product will not infringe the Orange Book-listed patents for EXPAREL, or that such patents are invalid, is called a paragraph IV certification. If the third-party submits a paragraph IV certification to the FDA, a notice of the paragraph IV certification must also be sent to us once the third-party’s ANDA is accepted for filing by the FDA. We may then initiate a lawsuit to defend the patents identified in the notice. The filing of a patent infringement lawsuit within 45 days of receipt of the notice automatically prevents the FDA from approving the third-
The patents and the patent applications that we have covering our iovera° products are primarily limited to specific handheld cryogenic needle devices that are cooled by a cryogen and methods for applying cryotherapy to nerve tissue using the cryogenic devices. Our market opportunity for our product candidates may be limited by gaps in patent coverage for the cryogenic devices, methods of use and other cryotherapy technology and systems that may be developed by competitors.
The iovera° cryogenic device is a compact, self-contained handheld device with a replaceable cryogen cartridge that delivers a cryogen through internal supply tubes to needle lumens of a replaceable needle probe, so as to cool the needle probe and thereby cool a surrounding target nerve tissue. We also have secured patents covering particular cryotherapy methods and pain treatments that provide what we deem to be optimal treatment using the iovera° cryogenic device.
Although we have patents that are broad enough to cover various alternative designs and methods, much of our patent coverage is tailored to cover the iovera° device and methods of use. It is thus possible that competitors may attempt to design around many of our patents. For example, we are aware of competitors developing cryogenic systems that are not self-contained handheld devices, or cryogenic systems that deliver cryotherapy through different mechanisms. It is also possible that competitors may attempt to develop and market cryotherapy devices and methods not covered by our patents, for example, basic cryotherapy treatment systems that are off-patent or cryoanalgesia for other nerve entrapment treatments.
The commercial opportunity for iovera° could be significantly harmed if competitors are able to develop and commercialize alternative designs and methods outside the scope of our patents.
Furthermore, our earliest patent family is scheduled to expire in 2025, thereby opening the door for competitors to copy some of our early technology. This early patent family is primarily focused on treating cosmetic defects that are no longer the focus of iovera°, but the underlying technology is nonetheless relevant enough for there to be appreciable overlap.
Finally, one or more third parties may challenge the patents covering the iovera° product, which could result in the invalidation or unenforceability of some or all of the relevant patent claims. Litigation or other proceedings to defend or enforce intellectual property rights are often very complex in nature, may be very expensive and time-consuming, may divert our management’s attention from our core business and may result in unfavorable results that could adversely impact our ability to prevent third parties from competing with our products.
Because it is difficult and costly to protect our proprietary rights, we may not be able to ensure their protection and all patents will eventually expire.
Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection for EXPAREL, DepoCyt(e)iovera°, DepoFoam and for any product candidates that we may develop, license or acquire and the methods we use to manufacture them, as well as successfully defending these patents and trade secrets against third-party challenges. We will only be able to protect our technologies from unauthorized use by third parties to the extent that valid and enforceable patents or trade secrets cover them.
The patent positions of pharmaceutical, medical device and biotechnology companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in pharmaceutical, medical device or biotechnology patents has emerged to date in the United States.U.S. Patent positions and policies outside the United StatesU.S. are even more uncertain. Changes in either the patent laws or in interpretations of patent
laws in the United StatesU.S. and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents.
The degree of future protection for our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
we may not have been the first to make the inventions covered by each of our pending patent applications and issued patents;
we may not have been the first to file patent applications for these inventions;
others may independently develop similar or alternative technologies or duplicate any of our product candidates or technologies;
it is possible that none of the pending patent applications will result in issued patents;
the issued patents covering our product candidates may not provide a basis for commercially viable active products, may not provide us with any competitive advantages, may not have sufficient scope or strength to protect the technologies they were intended to protect or may be challenged by third parties;
others may design around our patent claims to produce competitive products that fall outside the scope of our patents;
we may not develop or in-license additional proprietary technologies that are patentable;
patents of others may have an adverse effect on our business; or
competitors may infringe our patents and we may not have adequate resources to enforce our patents.
Patent applications in the United StatesU.S. are maintained in confidence for at least 18 months after their earliest effective filing date. Consequently, we cannot be certain we were the first to invent or the first to file patent applications on EXPAREL, iovera°, our DepoFoam drug delivery technology or any product candidates that we may develop, license or acquire. In the event that a third-party has also filed a USU.S. patent application relating to our product candidates or a similar invention, we may have to participate in interference proceedings declared by the USPTO to determine priority of invention in the United States.U.S. The costs of these proceedings could be substantial and it is possible that our efforts would be unsuccessful, resulting in a material adverse effect on our USU.S. patent position. Furthermore, we may not have identified all United StatesU.S. and foreign patents or published applications that affect our business either by blocking our ability to commercialize our drugs or medical devices or by covering similar technologies that affect our drug market.or medical device markets.
In addition, some countries, including many in Europe, do not grant patent claims directed to methods of treating humans, and in these countries patent protection may not be available at all to protect our product candidates. Even if patents are issued, we cannot guarantee that the claims of those patents will be valid and enforceable or provide us with any significant protection
against competitive products, or otherwise be commercially valuable to us. Furthermore, while we generally apply for patents in those countries where we intend to make, have made, use or sell patented products, we may not accurately predict all of the countries where patent protection will ultimately be desirable. If we fail to timely file a patent application in any such country, we may be precluded from doing so at a later date. We also cannot assure you that the patents issuing as a result of our foreign patent applications will have the same scope of coverage as our United StatesU.S. patents.
Some of our older patents have already expired. In the case of DepoCyt(e), keyEXPAREL, the European and U.S. patents providing protection in Europe have expired. Inprotecting the caseformulation of EXPAREL our European and US patent applications have been granted and provide protection through November 2018 and September 2018, respectively.expired in 2018. An existing formulation patent for EXPAREL expired in November 2013. An existing formulation patent for EXPAREL expired in the USU.S. in 2013 and its equivalents in Canada, Germany, France, Spain, Italy and the United Kingdom expired in 2014. Our earliest patent family for iovera° is scheduled to expire in 2025. Once our patents covering EXPAREL and iovera° have expired, we will be more reliant on trade secrets to protect against generic competition.
We also rely on trade secrets to protect our technology, particularly where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. While we use reasonable efforts to protect our trade secrets through confidentiality and non-disclosure agreements, our licensors, employees, consultants, contractors, outside scientific collaborators and other advisors may unintentionally or willfully disclose our information to competitors. Policing unauthorized use of our trade secrets or enforcing a claim that a third-party illegally obtained and is using our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, trade secret laws in other countries may not be as
protective as they are in the United States.U.S. Thus, courts outside the United StatesU.S. are sometimes less willing to protect trade secrets. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
In order to protect the goodwill associated with our company and product names, we rely on trademark protection for our marks. We have registered the “Pacira”, “EXPAREL”, “iovera°”, “DepoCyt”, “DepoCyte” and “DepoTXA”“DepoCyte” marks with the USPTO. A third-party may assert a claim that one of our marks is confusingly similar to its mark, and such claims or the failure to timely register a mark or objections by the FDA could force us to select a new name for one of our product candidates, which could cause us to incur additional expense or delay the commercialization of such product.
If we fail to obtain or maintain patent protection or trade secret protection for EXPAREL, DepoCyt(e)iovera°, DepoFoam or any product candidate that we may develop, license or acquire, third parties could use our proprietary information, which could impair our ability to compete in the market and adversely affect our ability to generate revenues and achieve profitability.
If we are sued for infringing intellectual property rights of third parties, it will be costly and time consuming, and an unfavorable outcome in any litigation would harm our business.
Our ability to develop, manufacture, market and sell EXPAREL, iovera°, our DepoFoam drug delivery technology or any product candidates that we may develop, license or acquire depends upon our ability to avoid infringing the proprietary rights of third parties. Numerous USU.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the general fields of pain management and cancer treatment and cover the use of numerous compounds, formulations and formulationsmedical devices in our targeted markets. Because of the uncertainty inherent in any patent or other litigation involving proprietary rights, we and our licensors may not be successful in defending intellectual property claims by third parties, which could have a material adverse effect on our results of operations. Regardless of the outcome of any litigation, defending the litigation may be expensive, time-consuming and distracting to management. In addition, because patent applications can take many years to issue, there may be currently pending applications, unknown to us, which may later result in issued patents that EXPAREL or DepoCyt(e)iovera° may infringe. There could also be existing patents of which we are not aware that EXPAREL or DepoCyt(e)iovera° may inadvertently infringe.
There is a substantial amount of litigation involving patent and other intellectual property rights in the biotechnology and biopharmaceutical industries in general. If a third-party claims that we infringe on their products or technology, we could face a number of issues, including:
infringement and other intellectual property claims which, with or without merit, can be expensive and time consuming to litigate and can divert management’s attention from our core business;
substantial damages for past infringement which we may have to pay if a court decides that our product infringes on a competitor’s patent;
a court prohibiting us from selling or licensing our product unless the patent holder licenses the patent to us, which it would not be required to do;
if a license is available from a patent holder, we may have to pay substantial royalties or grant cross licenses to our patents; and
redesigning our processes so they do not infringe, which may not be possible or could require substantial funds and time.
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
As is common in the biotechnology, pharmaceutical and pharmaceutical industry,medical device industries, we employ individuals who were previously employed at other biotechnology, or pharmaceutical and medical device companies, including our competitors or potential competitors. Although no claims against us are currently pending, we may be subject to claims that these employees or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
Risks Related to Cybersecurity
If we do not maintain the privacy and security of personal and business information, we could damage our reputation with customers and employees, incur substantial additional costs and become subject to litigation.
We receive, retain and transmit personal information about our customers and employees and entrust that information to third-party suppliers, including cloud service-providers that perform activities for us. Our business depends upon the secure transmission of encrypted confidential information over public networks, including information permitting payments. A compromise of our security systems or defects within our hardware or software, or those of our suppliers, that results in our customers’ or employees’ information being obtained by unauthorized persons, could adversely affect our reputation with our customers and others, as well as our operations, results of operations, financial condition and liquidity, and could result in litigation, government actions, or the imposition of penalties. In addition, a breach could require that we expend significant additional resources related to the security of information systems and could disrupt our operations.
The use of data by our business is regulated at the national and state or local level in all of our operating countries. Privacy and information-security laws and regulations change, and compliance with them may result in cost increases due to, among other things, systems changes and the development of new processes. If we or those with whom we share information fail to comply with these laws and regulations, our reputation could be damaged, possibly resulting in lost future business, and we could be subjected to additional legal risk as a result of non-compliance.
We have security measures and controls to protect personal and business information and continue to make investments to secure access to our information technology network. These measures may be undermined, however, due to the actions of outside parties, employee error, internal or external malfeasance, or otherwise, and, as a result, an unauthorized party may obtain access to our data systems and misappropriate business and personal information. Because the techniques used to obtain unauthorized access, disable or degrade service, or sabotage systems change frequently and may not immediately produce signs of intrusion, we may be unable to anticipate these techniques, timely discover or counter them, or implement adequate preventative measures. Any such breach or unauthorized access could result in significant legal and financial exposure, damage to our reputation, and potentially have an adverse effect on our business and results of operations.
Changes in data privacy and protection laws and regulations, particularly in Europe, or any failure to comply with such laws and regulations, could adversely affect our business and financial results.
We are subject to a variety of continuously evolving and developing laws and regulations globally regarding privacy, data protection and data security, including those related to the collection, storage, handling, use, disclosure, transfer and security of personal data. Significant uncertainty exists as privacy and data protection laws may be interpreted and applied differently from country to country and may create inconsistent or conflicting requirements. These laws apply to transfers of information among our affiliates, as well as to transactions we enter into with third-party vendors. For example, the E.U. adopted a comprehensive General Data Privacy Regulation, or GDPR, in May 2016 that replaced the then-current E.U. Data Protection Directive and related country-specific legislation in May 2018. GDPR requires companies to satisfy new requirements regarding the handling of personal and sensitive data, including its use, protection and the ability of persons whose data is stored to correct or delete such data about themselves. Failure to comply with GDPR requirements could result in penalties of up to 4% of worldwide revenue. Complying with the enhanced obligations imposed by the GDPR may result in significant costs to our business and require us to revise certain of our business practices. In addition, legislators and regulators in the U.S. are proposing new and more robust cybersecurity rules in light of the recent broad-based cyberattacks at a number of companies. These and similar initiatives around the world could increase the cost of developing, implementing or securing our servers and require us to allocate more resources to improved technologies, adding to our information technology and compliance costs. In addition, enforcement actions and investigations by regulatory authorities related to data security incidents and privacy violations continue to increase. The enactment of more restrictive laws, rules, regulations, or future enforcement actions or investigations could impact us through increased costs or restrictions on our business, and noncompliance could result in regulatory penalties and significant legal liability.
Risks Related to our Financial Condition and Capital Requirements
Cumulatively, we have incurred significant losses since our inception and may incur additional losses in the future.
iovera°. As a result, we had not been profitable prior to 2015 and werehave not profitable in 2016.been since. Because of the numerous risks and uncertainties associated with developing pharmaceutical products and medical devices, we are unable to predict the extent of any future losses.
We may not return to profitability.
Our ability to return to profitability depends upon our ability to generate revenue from EXPAREL.EXPAREL and iovera°. Our ability to generate revenue depends on a number of factors, including, but not limited to, our ability to:
manufacture commercial quantities of EXPAREL and iovera° at acceptable cost levels; and
continue to develop a commercial organization and the supporting infrastructure required to successfully market and sell EXPAREL.both EXPAREL and iovera°.
We anticipate incurring significant additional costs associated with the commercialization of both EXPAREL and iovera° and are unsure as to whether we will be able to return to profitability. If we are unable to generate additional revenues, we will not be able to do so and may be unable to continue operations without continued funding.
We may need additional funding and may be unable to raise capital when needed, which would force us to delay, reduce or eliminate our product development programs or commercialization efforts.
Developing and commercializing products for use in the hospital setting,or ASC settings, conducting clinical trials, establishing outsourced manufacturing relationships and successfully manufacturing and marketing drugs and medical devices that we may develop is expensive. We may need to raise additional capital to:
continue to fund our operations;
continue our efforts to hire additional personnel and build a commercial infrastructure to commercialize EXPAREL;EXPAREL and iovera°;
qualify, outsource or build additional commercial-scale manufacturing of our products under cGMP;
in-license and develop additional product candidates; and
refinance our current2.375% convertible senior notes, due February 2019.April 2022.
We may not have sufficient financial resources to continue our operations or meet all of our objectives, which could require us to postpone, scale back or eliminate some, or all, of these objectives. Our future funding requirements will depend on many factors, including, but not limited to:
the costs of maintaining a commercial organization to sell, market and distribute EXPAREL;EXPAREL and iovera°;
the success of the commercialization of EXPAREL;EXPAREL and iovera°;
the cost and timing of manufacturing sufficient supplies of EXPAREL and iovera° to meet customer demand, including the cost of expanding our manufacturing facilities to produce EXPAREL;EXPAREL and iovera°;
the rate of progress and costs of our efforts to prepare for the submission of an NDA, sNDA or 510(k) pre-market notification for any product candidates that we may in-license or acquire in the future, and the potential that we may need to conduct additional clinical trials to support applications for regulatory approval;
the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights associated with our product candidates, including any such costs we may be required to expend if our licensors are unwilling or unable to do so;
the effect of competing technological and market developments;
the terms and timing of any collaborative, licensing, co-promotion or other arrangements that we may establish; and
the potential that we may be required to file a lawsuit to defend our patent rights or regulatory exclusivities from challenges by companies seeking to market generic versions of extended-release liposome injection of bupivacaine.bupivacaine or a cryoanalgesic device that infringes on the various patents covering iovera°.
Future capital requirements will also depend on the extent to which we acquire or invest in additional complementary businesses, products and technologies.
Until we can generate a sufficient amount of product revenue, if ever, we expect to finance or supplement future cash needs through public or private equity offerings, debt financings, product supply revenue and royalties, collaboration and licensing arrangements, as well as through interest income earned on cash and investment balances. We cannot be certain that additional funding will be available on acceptable terms, or at all. If adequate funds are not available, we may be required to delay, reduce the scope of, or eliminate one or more of our development programs or our commercialization efforts.
Our quarterly operating results may fluctuate significantly.
We expect our operating results to be subject to quarterly fluctuations. Our operating results will be affected by numerous factors, including:
the level of underlying hospital and ASC demand for EXPAREL and iovera° and end-user buying patterns;
maintaining our existing manufacturing facilities for EXPAREL and iovera°, expanding our manufacturing capacity and constructing facilitiesa second suite for the manufacture of EXPAREL with our co-production partner, Patheon,Thermo Fisher, including installing specialized processing equipment for the manufacturing of EXPAREL;
our execution of other collaborative, licensing, distribution, manufacturing or similar arrangements and the timing of payments we may make or receive under these arrangements;
variations in the level of expenses related to our future development programs;
any product liability or intellectual property infringement lawsuit in which we may become involved; and
regulatory developments, lawsuits and investigations affecting EXPAREL, iovera° or the product candidates of our competitors;
If our quarterly or annual operating results fall below the expectations of our investors or securities analysts, the price of our common stock could decline substantially. Furthermore, any quarterly or annual fluctuations in our operating results may, in
turn, cause the price of our stock to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
We may be unable to successfully integrate the businesses and personnel of acquired companies and businesses, and may not realize the anticipated synergies and benefits of such acquisitions.
From time to time, we may complete acquisitions of companies and certain businesses of companies, and we may not realize the expected benefits from such acquisitions because of integration difficulties or other challenges. For example, on April 9, 2019, we completed the MyoScience Acquisition.
The success of any acquisitions will depend, in part, on our ability to realize all or some of the anticipated synergies and other benefits from integrating the acquired businesses with our existing businesses. The integration process may be complex, costly and time-consuming. The potential difficulties we may face in integrating the operations of our acquisitions include, among others:
failure to implement our business plans for the combined businesses and consolidation or expansion of production capacity as planned and where applicable;
unexpected losses of key employees, customers or suppliers of our acquired companies and businesses;
unanticipated issues in conforming our acquired companies’ and businesses’ standards, processes, procedures and controls with our operations;
coordinating new product and process development;
increasing the scope, geographic diversity and complexity of our operations;
diversion of management’s attention from other business concerns;
adverse effects on our or our acquired companies’ and businesses’ existing business relationships;
unanticipated changes in applicable laws and regulations;
operating risks inherent in our acquired companies’ and businesses’ business and operations;
unanticipated expenses and liabilities;
potential unfamiliarity with our acquired companies and businesses technology, products and markets, which may place us at a competitive disadvantage; and
other difficulties in the assimilation of our acquired companies and businesses operations, technologies, products and systems.
If MyoScience or any other acquired companies and businesses may have unanticipated or larger than anticipated liabilities for patent and trademark infringement claims, violations of laws, commercial disputes, taxes and other known and unknown types of liabilities, there may be liabilities that we underestimated or did not discover in the course of performing our due diligence investigation of our acquired companies and businesses. We may have no recourse or limited recourse under the applicable acquisition-related agreement to recover damages relating to the liabilities of our acquired companies and businesses.
We may not be able to maintain or increase the levels of revenue, earnings or operating efficiency that each of the acquired companies and businesses and us had historically achieved or might achieve separately. In addition, we may not accomplish the integration of any acquired companies and businesses smoothly, successfully or within the anticipated costs or timeframe. If we experience difficulties with the integration process or if the business of any acquired companies or businesses deteriorates, the anticipated cost savings, growth opportunities and other synergies of any acquired companies and businesses may not be realized fully or at all, or may take longer to realize than expected. If any of the above risks occur, our business, financial condition, results of operations and cash flows may be materially and adversely impacted, we may fail to meet the expectations of investors or analysts, and our stock price may decline as a result.
Raising additional funds by issuing securities may cause dilution to existing stockholders and raising funds through lending and licensing arrangements may restrict our operations or require us to relinquish proprietary rights.
To the extent that we raise additional capital by issuing equity securities, our existing stockholders’ ownership willwould be diluted. If we raise additional funds through licensing arrangements, it may be necessary to relinquish potentially valuable rights to our potential products or proprietary technologies, or grant licenses on terms that are not favorable to us. Any debt financing we enter into may involve covenants that restrict our operations. These restrictive covenants may include limitations on additional borrowing and specific restrictions on the use of our assets as well as prohibitions on our ability to create liens, pay dividends, redeem our stock or make investments.
The use of our net operating loss carryforwards and research tax credits will be limited.
We have significant federal and state net operating loss, or NOL, carryforwards and federal and state research and development tax credit carryforwards. Our net operating lossNOL carryforwards and research and development tax credits may expire and not be used. Our net operating lossNOL carryforwards will begin expiring in 20252026 for federal purposes and 2017in 2024 for state purposes if we have not used them prior to that time. For any federal NOLs generated after December 31, 2017, the NOLs will have an indefinite life and utilization will be subject to a limitation of 80% of taxable income. The non-U.S. NOLs do not expire. Additionally, our ability to use certain net operating lossNOLs and credit carryforwards to offset taxable income or tax, respectively, in the future will be limited under Internal Revenue Code Sections 382 and 383 because we experienced cumulative changes in ownership of more than 50% within a three-year period. Such ownership changes were triggered by the cumulative ownership changes arising as a result of the initial acquisition of the Company’s stock in 2007 and the completion of our initial public offering and our other financing transactions. Because of the ownership changes, we will be limited regarding the amount of net operating lossNOL carryforwards and research tax credits that we can utilize annually in the future to offset taxable income or tax, respectively. Such an annual limitation will significantly reduce the utilization of the net operating loss carryforwardsNOLs and research tax credits before they expire. In addition, California and certain states have suspended use of net operating lossNOL carryforwards for certain taxable years, and other states are considering similar measures. As a result, we may incur higher state income tax expense in the future. Depending on our future tax position, continued suspension of our ability to use net operating lossNOL carryforwards in states in which we are subject to income tax could have an adverse impact on our results of operations and financial condition.
Risks Related Ourto our Indebtedness and our Common Stock
Our common stock price may be subject to significant fluctuations and volatility.
Our stock price is volatile, and from February 3, 2011, the first day of trading of our common stock, to February 28, 2017,19, 2020, the trading prices of our stock have ranged from $6.16 to $121.95 per share.
Our stock could be subject to wide fluctuations in price in response to various factors, including the following:
the commercial success of EXPAREL;EXPAREL and iovera°;
results of clinical trials of our products, product candidates or those of our competitors;
changes or developments in laws or regulations applicable to our products or product candidates;
introduction of competitive products or technologies;
failure to meet or exceed financial projections we provide to the public;
actual or anticipated variations in quarterly operating results;
failure to meet or exceed the estimates and projections of the investment community;
the perception of the pharmaceutical and medical device industry by the public, legislatures, regulators and the investment community;
regulatory concerns or government actions
general economic and market conditions and overall fluctuations in USU.S. equity markets;
developments concerning our sources of manufacturing supply;
disputes or other developments relating to patents or other proprietary rights;
additions or departures of key scientific or management personnel;
the extent to which we acquire or invest in products, businesses and technologies;
issuances of debt, equity or convertible securities;
changes in the market valuations of similar companies; and
the other factors described in this “Risk Factors” section.
In addition, the stock market in general, and the market for small pharmaceutical and biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance. Fluctuations in our stock price could, among other things, adversely impact the trading price of our shares.
Servicing our indebtedness requires a significant amount of cash, and we may not have sufficient cash flow from our business to pay our substantial indebtedness.
Our ability to make payments of the principal of, to pay interest on or to refinance our indebtedness, including the 2.375% convertible senior notes due 2022, or 2022 Notes, issued in our private offering completed on January 23, 2013, or Notes,March 13, 2017, as described below, or to make cash payments in connection with any conversion of the 2022 Notes depends on our future performance, which is subject to economic, financial, competitive and other factors beyond our control. Our business may not generate cash flow from operations in the future sufficient to service our indebtedness and make necessary capital expenditures. If we are unable to generate such cash flow, we may be required to adopt one or more alternatives, such as selling assets, restructuring indebtedness or obtaining additional equity capital on terms that may be onerous or highly dilutive. Our ability to refinance our indebtedness will depend on the capital markets and our financial condition at such time. We may not be able to engage in any of these activities or engage in these activities on desirable terms, which could result in a default on our debt obligations.
On January 23, 2013, the CompanyMarch 13, 2017, we completed a private offeringplacement of $120.0$345.0 million in aggregate principal amount of 3.25% convertible senior notes due 2019
our 2022 Notes, and entered into an indenture agreement, or 2022 Indenture, with Wells Fargo Bank, National Association, a national banking association, as trustee, governingrespect to the 2022 Notes. The 2022 Notes accrue interest at a fixed rate of 3.25%2.375% per year, payable semiannually in arrears on FebruaryApril 1 and AugustOctober 1 of each year, beginning on August 1, 2013.year. The 2022 Notes will mature on FebruaryApril 1, 2019.
2022. As of December 31, 2016,2019, our total consolidated gross indebtedness was $118.5$345.0 million, all of which was the principal outstanding on the 2022 Notes and all of which was unsecured indebtedness, andindebtedness. Additionally, our subsidiaries had no indebtedness (in each case, excluding(excluding trade payables, intercompany liabilities and income tax-related liabilities).
We may not have the ability to raise the funds necessary to settle conversions of the 2022 Notes in cash to the extent requiredelected or to repurchase the 2022 Notes upon a fundamental change, and our future indebtedness may contain limitations on our ability to pay cash upon conversion of the 2022 Notes or limitations on our ability to repurchase the 2022 Notes.
Holders of the 2022 Notes will have the right to require us to repurchase their 2022 Notes upon the occurrence of a fundamental change at a repurchase price equal to 100% of their principal amount, plus accrued and unpaid interest, if any. In addition, upon conversion of the 2022 Notes (if we choose to settle the principal amount in cash at our option), we will be required to make cash payments for each $1,000 in principal amount of 2022 Notes converted of at least the lesser of $1,000 and the sum of the daily conversion values. However, we may not have enough available cash or be able to obtain financing at the time we are required to make repurchases of 2022 Notes surrendered therefor or 2022 Notes being converted. Any credit facility or other agreement that we may enter into may limit our ability to make cash payments at the time of a fundamental change or upon conversion of the 2022 Notes. Further, our ability to repurchase the 2022 Notes or to pay cash upon conversions of the 2022 Notes may be limited by law, by regulatory authority or by agreements governing our future indebtedness. Our failure to repurchase 2022 Notes at a time when the repurchase is required by the indenture or to pay any cash payable on future conversions of the 2022 Notes as required by the indenture2022 Indenture would constitute a default under the indenture.2022 Indenture. A default under the indenture or the fundamental change itself could also lead to a default under agreements governing our future indebtedness. If the repayment of the related indebtedness were to be accelerated after any applicable notice or grace periods, we may not have sufficient funds to repay the indebtedness and repurchase the 2022 Notes or make cash payments upon conversions thereof. In
Future sales in the public market or issuances of our common stock could lower the market price for our common stock.
In the future, we may sell additional shares of our common stock to raise capital. Except under limited circumstances, we are not restricted from issuing additional common stock, including securities that are convertible into or exchangeable for, or that represent the right to receive, common stock. The issuance of additional shares of our common stock or convertible securities, including upon exercise of our outstanding options, vesting of our restricted stock units or otherwise, will dilute the ownership interest of our common stockholders. In addition, our greater than 5% stockholders may sell a substantial number of their shares in the public market, which could also affect the market price for our common stock. We cannot predict the size of future sales or issuances of our common stock or the effect, if any, that they may have on the market price for our common stock. The liquidity and trading volume of our common stock is limited. For the three months ended December 31, 2016, the average per day trading volume of our common stock was 857,234 shares. The issuance and/or sale of substantial amounts of common stock, or the perception that such issuances and/or sales may occur, could adversely affect the market price of our common stock and impair our ability to raise capital through the sale of additional equity or debtsdebt securities.
Some provisions of our charter documents and Delaware law may have anti-takeover effects that could discourage an acquisition of us by others, even if an acquisition would be beneficial to our stockholders, and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our restated certificate of incorporation and our bylaws, as well as provisions of the Delaware General Corporation Law, or DGCL, could make it more difficult for a third-party to acquire us or increase the cost of acquiring us, even if doing so would benefit our stockholders, including transactions in which stockholders might otherwise receive a premium for their shares. These provisions include:
authorizing the issuance of “blank check” preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval;
prohibiting stockholder action by written consent, thereby requiring all stockholder actions to be taken at a meeting of our stockholders;
eliminating the ability of stockholders to call a special meeting of stockholders; and
establishing advance notice requirements for nominations for election to the Boardboard of Directorsdirectors or for proposing matters that can be acted upon at stockholder meetings.
These provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our Boardboard of Directors,directors, which is responsible for appointing the members of our management. In addition, we are subject to Section 203 of the DGCL, which generally prohibits a Delaware corporation from engaging in any of a broad range of business combinations with an interested stockholder for a period of three years following the date on which the stockholder became an interested stockholder, unless such transactions are approved by our Boardboard of Directors.directors. This provision could have the effect of delaying or preventing a change of control, whether or not it is desired by or beneficial to our stockholders.
We do not intend to pay dividends on our common stock for the foreseeable future.
We have never declared or paid cash dividends on our common stock. We currently intend to retain our future earnings, if any, to finance the further development and expansion of our business and do not intend to pay cash dividends in the foreseeable future. Any future determination to declare or pay dividends will be at the discretion of our Boardboard of Directorsdirectors and will depend upon our financial condition, results of operations, capital requirements, restrictions contained in future financing instruments, provisions of applicable law and suchany other factors as our Boardboard of Directorsdirectors deems relevant.
Item 1B. Unresolved Staff Comments
None.
Item 2. Properties
We occupy fourthree facilities totaling approximately 172,000150,000 square feet at our Science Center Campus in San Diego, California. We use these facilities for research and development, manufacturing, general and administrative purposes and the storage of inventory and raw materials. All ofOur research and development property lease expires in October 2020, our propertiesEXPAREL manufacturing facility lease expires in San Diego are under leases which expireJune 2030 and our warehouse lease expires in August 2020.2030. Our iovera° facility in Fremont, California, consists of approximately 20,000 square feet of mixed-use manufacturing, research and development and office space, and its lease expires in December 2021. In addition, we maintain our executive offices and our commercial and business development facility in Parsippany, New Jersey, where we occupy approximately 42,00053,000 square feet under a lease expiring in March 2028.2028 and occupy approximately 4,000 square feet of office space in Tampa, Florida under a lease expiring in September 2021. Additionally, we have a lease beginning April 2020 for a new research and development and manufacturing facility consisting of approximately 90,000 square feet at our Science Center Campus in San Diego, California, which will replace our existing approximately 45,000 square foot research and development facility in San Diego whose lease expires in October 2020. We also have a lease for our former DepoCyt(e) production facility in San Diego which is currently idle and expires in August 2020.
We believe that our research and development and manufacturing facilities at our Science Center Campus, Thermo Fisher and yet-to-be completed Patheon facilityFremont sites (as discussed in Item 1-Business1—Business above) will be sufficient for our commercial and pipeline development needs. We also may add new facilities or expand existing facilities as we add employees, expand our geographic markets and if demand for EXPAREL and iovera° increases and we believe that suitable additional or substitute space will be available as needed to accommodate any such expansion of our operations.
Item 3. Legal Proceedings
From time to time, we have been and may again become involved in legal proceedings arising in the ordinary course of our business. Except as described below, we are not presently a party to any litigation that we believe to be material and we are not aware of any pending or threatened litigation against us that we believe could have a material adverse effect on our business, operating results, financial condition or cash flows.
In April 2015, we received a subpoena from the USU.S. Department of Justice, USU.S. Attorney’s Office for the District of New Jersey, requiring the production of a broad range of documents pertaining to marketing and promotional practices related to EXPAREL. We are cooperating with the government’s inquiry. We can make no assurances as to the time or resources that will need to be devoted to this inquiry or its final outcome, or the impact, if any, of this inquiry or any proceedings on our business, financial condition, results of operations and cash flows.
In December 2019, we reached an agreement in principle with the Department of Justice and more than one state Attorney General’s office (the “Plaintiffs”) on a proposal for a global civil settlement in the amount of $3.5 million, subject to accrual of interest on the settlement amount from the date of the agreement in principle, negotiation of a definitive settlement agreement and other contingencies. As part of the settlement, Pacira will admit no wrongdoing and will explicitly deny the Plaintiffs’ allegations. Pacira has been given assurances that, if the parties can agree to negotiation of the settlement, this will conclude the investigation that originated from the U.S. Department of Justice subpoena in April 2015.
Item 4. Mine Safety Disclosures
Not applicable.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our common stock is listed and traded under the ticker symbol “PCRX” on The NASDAQthe Nasdaq Global Select Market. The following table sets forth, for the periods indicated, the high and low intraday sales pricesAs of our common stock as reported by the NASDAQ:
| Year Ended 2016 | High | Low | ||||||
| Fourth Quarter | $ | 38.20 | $ | 29.95 | ||||
| Third Quarter | 46.22 | 32.16 | ||||||
| Second Quarter | 65.64 | 31.08 | ||||||
| First Quarter | 76.75 | 44.15 | ||||||
| Year Ended 2015 | High | Low | ||||||
| Fourth Quarter | $ | 80.25 | $ | 35.78 | ||||
| Third Quarter | 72.98 | 39.29 | ||||||
| Second Quarter | 93.22 | 65.00 | ||||||
| First Quarter | 121.95 | 82.00 | ||||||
Performance Graph
The following graph shows the value of an investment of $100$100.00 made on December 31, 2011,2014, in each of Pacira common stockBioSciences, Inc. (PCRX), the NASDAQNasdaq Composite indexIndex (^IXIC) and the NASDAQNasdaq Biotechnology indexIndex (^NBI). The two Nasdaq indices are included for comparative purposes only and do not necessarily reflect management’s opinion that such indices are an appropriate measure of the relative performance of our common stock. All results assume the reinvestment of dividends, if any, and are calculated as of December 31st of each year. The historical stock price performance of our common stock and the indices shown in thethis performance graph is not necessarily indicative of future stock price performance.
Comparison of Five-Year Cumulative Total Returns
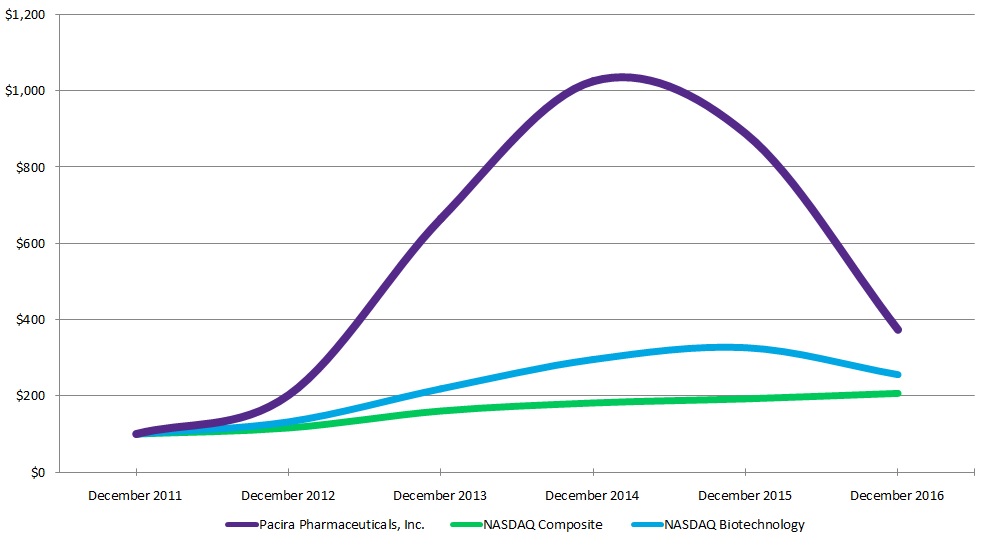
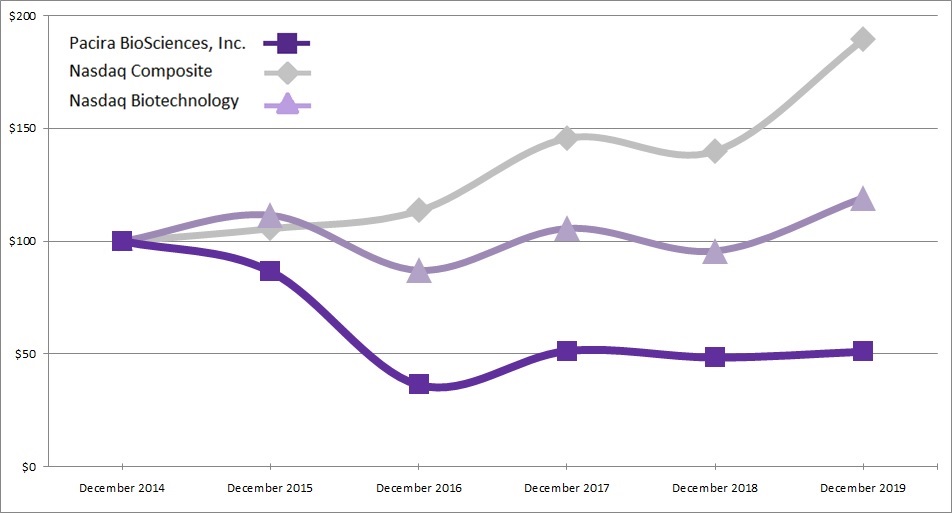
| Cumulative Total Return | |||||||||||||||||||||||
| Dec 31, | Dec 31, | Dec 31, | Dec 31, | Dec 31, | Dec 31, | ||||||||||||||||||
| 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | ||||||||||||||||||
| Pacira Pharmaceuticals, Inc. (PCRX) | $ | 100.00 | $ | 201.97 | $ | 664.62 | $ | 1,024.97 | $ | 887.75 | $ | 373.41 | |||||||||||
| NASDAQ Composite (^IXIC) | 100.00 | 115.91 | 160.32 | 181.44 | 192.21 | 206.63 | |||||||||||||||||
| NASDAQ Biotechnology (^NBI) | 100.00 | 131.91 | 218.45 | 295.37 | 326.39 | 255.62 | |||||||||||||||||
| Cumulative Total Return as of December 31, | |||||||||||||||||||||||
| 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | ||||||||||||||||||
| Pacira BioSciences, Inc. (PCRX) | $ | 100.00 | $ | 86.61 | $ | 36.43 | $ | 51.49 | $ | 48.52 | $ | 51.09 | |||||||||||
| Nasdaq Composite Index (^IXIC) | $ | 100.00 | $ | 105.73 | $ | 113.66 | $ | 145.76 | $ | 140.10 | $ | 189.45 | |||||||||||
| Nasdaq Biotechnology Index (^NBI) | $ | 100.00 | $ | 111.42 | $ | 87.26 | $ | 105.64 | $ | 95.79 | $ | 119.17 | |||||||||||
Dividend Policy
We have never declared or paid any cash dividends on our capital stock. We currently intend to retain our future earnings, if any, to finance the growthfuture development and developmentexpansion of our business, and as such we do not expect to pay any cash dividends on our common stock in the foreseeable future. The payment of future dividends, if any, will be at the discretion of our Boardboard of Directorsdirectors and will depend on our financial condition, results of operations, capital requirements, restrictions contained in future financing instruments, provisions of applicable law and any other factors theour board of directors deems relevant.
Item 6. Selected Financial Data
The following tables provide selected historical consolidated financial data. We have prepared this information using our audited consolidated financial statements as of and for the years ended December 31, 2019, 2018, 2017, 2016 2015, 2014, 2013 and 2012.2015. The following consolidated financial data should be read in conjunction with our consolidated financial statements and related notes and Item 7. “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in this report.
| Year Ended December 31, | |||||||||||||||||||
2019 A | 2018 | 2017 | 2016 | 2015 | |||||||||||||||
| Consolidated Statements of Operations Data | (In thousands, except per share data) | ||||||||||||||||||
| Revenues: | |||||||||||||||||||
| Net product sales | $ | 418,926 | $ | 332,427 | $ | 284,342 | $ | 270,073 | $ | 244,487 | |||||||||
| Collaborative licensing and milestone revenue | — | 3,000 | 387 | 3,426 | 1,426 | ||||||||||||||
| Royalty revenue | 2,100 | 1,850 | 1,901 | 2,872 | 3,084 | ||||||||||||||
| Total revenues | 421,026 | 337,277 | 286,630 | 276,371 | 248,997 | ||||||||||||||
| Operating expenses: | |||||||||||||||||||
| Cost of goods sold | 106,712 | 86,845 | 87,915 | 110,104 | E | 71,837 | |||||||||||||
| Research and development | 72,119 | 55,688 | 57,290 | 45,678 | 28,662 | ||||||||||||||
| Selling, general and administrative | 200,782 | 177,265 | 161,494 | 152,613 | F | 139,043 | |||||||||||||
| Amortization of acquired intangible assets | 5,703 | — | — | — | — | ||||||||||||||
| Acquisition-related charges, product discontinuation and other | 25,230 | B | 1,564 | C | 4,868 | D | — | — | |||||||||||
| Total operating expenses | 410,546 | 321,362 | 311,567 | 308,395 | 239,542 | ||||||||||||||
| Income (loss) from operations | 10,480 | 15,915 | (24,937 | ) | (32,024 | ) | 9,455 | ||||||||||||
| Other (expense) income: | |||||||||||||||||||
| Interest income | 7,376 | 6,497 | 4,078 | 1,323 | 678 | ||||||||||||||
| Interest expense | (23,628 | ) | (21,949 | ) | (18,047 | ) | (7,061 | ) | (7,725 | ) | |||||||||
| Loss on early extinguishment of debt | — | — | (3,732 | ) | — | (52 | ) | ||||||||||||
| Royalty interest obligation | — | — | — | — | (71 | ) | |||||||||||||
| Other, net | (4,976 | ) | (888 | ) | 167 | (82 | ) | (165 | ) | ||||||||||
| Total other expense, net | (21,228 | ) | (16,340 | ) | (17,534 | ) | (5,820 | ) | (7,335 | ) | |||||||||
| Income (loss) before income taxes | (10,748 | ) | (425 | ) | (42,471 | ) | (37,844 | ) | 2,120 | ||||||||||
| Income tax expense | (268 | ) | (46 | ) | (140 | ) | (105 | ) | (264 | ) | |||||||||
| Net income (loss) | $ | (11,016 | ) | $ | (471 | ) | $ | (42,611 | ) | $ | (37,949 | ) | $ | 1,856 | |||||
| Net income (loss) per share: | |||||||||||||||||||
| Basic net income (loss) per common share | $ | (0.27 | ) | $ | (0.01 | ) | $ | (1.07 | ) | $ | (1.02 | ) | $ | 0.05 | |||||
| Diluted net income (loss) per common share | $ | (0.27 | ) | $ | (0.01 | ) | $ | (1.07 | ) | $ | (1.02 | ) | $ | 0.04 | |||||
| Weighted average common shares outstanding: | |||||||||||||||||||
| Basic | 41,513 | 40,911 | 39,806 | 37,236 | 36,540 | ||||||||||||||
| Diluted | 41,513 | 40,911 | 39,806 | 37,236 | 41,301 | ||||||||||||||
A - We completed the MyoScience, Acquisition on April 9, 2019. The acquisition was accounted for using the acquisition method of accounting and, accordingly, the assets acquired, liabilities assumed and results of operations of the acquired business are included in our consolidated financial statements from the date of acquisition through December 31, 2019.
B - Includes charges of $21.6 million related to the MyoScience Acquisition. Of this total, $16.7 million represents increases in the fair value of contingent consideration resulting from the achievement of regulatory milestones and revised commercial forecasts (which are tied to potential future milestone payments), $4.2 million represents advisory costs, including legal, financial, accounting and tax services. The remaining $0.7 million represents separation costs, asset write-downs and other restructuring charges. Charges of $0.2 million were recorded related to the discontinuation of our DepoCyt(e) manufacturing activities for lease costs, asset retirement obligations and other estimated exit costs. Additionally, this includes a charge of $3.5 million related to an agreement in principle with the U.S. Department of Justice on a proposal for a global civil settlement related to an April 2015 inquiry. For further discussion of these charges, see Note 5, MyoScience Acquisition, Note 18, Acquisition-Related Charges and Product Discontinuation, Net and Note 21, Commitments and Contingencies, to our consolidated financial statements included herein.
C - Represents non-recurring charges of $1.6 million related to the discontinuation of our DepoCyt(e) manufacturing activities for lease costs, asset retirement obligations and other estimated exit costs. The charges incurred in 2018 primarily represent additional lease and facility costs due to the fact that we were not able to sub-lease the property where DepoCyt(e) was manufactured considering the short period of time remaining on our existing lease. For further discussion of these charges, see Note 18, Acquisition-Related Charges and Product Discontinuation, Net, to our consolidated financial statements included herein.
| Year Ended December 31, | |||||||||||||||||||
| 2016 | 2015 | 2014 | 2013 | 2012 | |||||||||||||||
| Statement of Operations Data | (In thousands, except per share data) | ||||||||||||||||||
| Revenues: | |||||||||||||||||||
| Net product sales | $ | 270,073 | $ | 244,487 | $ | 193,526 | $ | 81,956 | $ | 18,191 | |||||||||
| Collaborative licensing and milestone revenue | 3,426 | 1,426 | 1,287 | 972 | 18,390 | ||||||||||||||
| Royalty revenue | 2,872 | 3,084 | 2,855 | 2,623 | 2,503 | ||||||||||||||
| Total revenues | 276,371 | 248,997 | 197,668 | 85,551 | 39,084 | ||||||||||||||
| Operating expenses: | |||||||||||||||||||
| Cost of goods sold | 110,104 | 1 | 71,837 | 77,440 | 54,772 | 32,139 | |||||||||||||
| Research and development | 45,678 | 28,662 | 18,731 | 21,560 | 9,937 | ||||||||||||||
| Selling, general and administrative | 152,613 | 2 | 139,043 | 106,662 | 62,508 | 46,306 | |||||||||||||
| Total operating expenses | 308,395 | 239,542 | 202,833 | 138,840 | 88,382 | ||||||||||||||
| Income (loss) from operations | (32,024 | ) | 9,455 | (5,165 | ) | (53,289 | ) | (49,298 | ) | ||||||||||
| Other (expense) income: | |||||||||||||||||||
| Interest income | 1,323 | 678 | 382 | 259 | 275 | ||||||||||||||
| Interest expense | (7,061 | ) | (7,725 | ) | (8,278 | ) | (7,253 | ) | (1,807 | ) | |||||||||
| Loss on early extinguishment of debt | — | (52 | ) | — | (3,398 | ) | (1,062 | ) | |||||||||||
| Royalty interest obligation | — | (71 | ) | (323 | ) | (623 | ) | (278 | ) | ||||||||||
| Other, net | (82 | ) | (165 | ) | (159 | ) | (47 | ) | (111 | ) | |||||||||
| Total other expense, net | (5,820 | ) | (7,335 | ) | (8,378 | ) | (11,062 | ) | (2,983 | ) | |||||||||
| Income (loss) before income taxes | (37,844 | ) | 2,120 | (13,543 | ) | (64,351 | ) | (52,281 | ) | ||||||||||
| Income tax (expense) benefit | (105 | ) | (264 | ) | (173 | ) | 442 | — | |||||||||||
| Net income (loss) | $ | (37,949 | ) | $ | 1,856 | $ | (13,716 | ) | $ | (63,909 | ) | $ | (52,281 | ) | |||||
| Net income (loss) per share: | |||||||||||||||||||
| Basic net income (loss) per common share | $ | (1.02 | ) | $ | 0.05 | $ | (0.39 | ) | $ | (1.93 | ) | $ | (1.72 | ) | |||||
| Diluted net income (loss) per common share | $ | (1.02 | ) | $ | 0.04 | $ | (0.39 | ) | $ | (1.93 | ) | $ | (1.72 | ) | |||||
| Weighted average common shares outstanding: | |||||||||||||||||||
| Basic | 37,236 | 36,540 | 35,299 | 33,182 | 30,332 | ||||||||||||||
| Diluted | 37,236 | 41,301 | 35,299 | 33,182 | 30,332 | ||||||||||||||
E - Includes a $20.7 million charge for inventory and related reserves for the cost of EXPAREL batches impacted by a routine stability test that did not meet required specifications. For further discussion of this charge, see Note 4, Inventories, to our consolidated financial statements included herein.
| December 31, | |||||||||||||||||||
2019 A | 2018 | 2017 | 2016 | 2015 | |||||||||||||||
| Consolidated Balance Sheet Data | (In thousands) | ||||||||||||||||||
| Cash and cash equivalents, short-term and long-term investments | $ | 356,748 | $ | 409,325 | $ | 371,394 | $ | 172,597 | $ | 172,427 | |||||||||
| Working capital | 300,884 | 417,308 | 334,893 | 198,251 | 102,794 | ||||||||||||||
| Total assets | 831,065 | 689,353 | 628,371 | 391,466 | 387,735 | ||||||||||||||
| Long-term liabilities | 368,448 | 307,466 | 292,671 | 127,652 | 19,555 | ||||||||||||||
| Accumulated deficit | (399,398 | ) | (388,226 | ) | (389,136 | ) | (346,238 | ) | (308,289 | ) | |||||||||
| Total stockholders’ equity | 354,944 | 321,226 | 279,483 | 218,976 | 218,392 | ||||||||||||||
A - We completed the MyoScience Acquisition on April 9, 2019. The acquisition was accounted for using the acquisition method of this charge, see Note 15, Commercial Partnersaccounting and, Other Agreements, toaccordingly, the assets acquired, liabilities assumed and results of operations of the acquired business are included in our consolidated financial statements included herein.
| December 31, | |||||||||||||||||||
| 2016 | 2015 | 2014 | 2013 | 2012 | |||||||||||||||
| Balance Sheet Data | (In thousands) | ||||||||||||||||||
| Cash and cash equivalents, restricted cash, short-term and long-term investments | $ | 172,597 | $ | 172,427 | $ | 182,598 | $ | 73,785 | $ | 42,573 | |||||||||
Working capital (deficit) 3 | 198,251 | 102,794 | 71,715 | (18,345 | ) | 46,766 | |||||||||||||
Total assets 3 | 391,466 | 387,735 | 323,540 | 166,668 | 111,722 | ||||||||||||||
Long-term liabilities 3 | 127,652 | 19,555 | 14,917 | 6,628 | 32,376 | ||||||||||||||
| Accumulated deficit | (346,238 | ) | (308,289 | ) | (310,145 | ) | (296,429 | ) | (232,520 | ) | |||||||||
| Total stockholders’ equity | 218,976 | 218,392 | 171,145 | 41,249 | 65,855 | ||||||||||||||
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
Management’s Discussion and Analysis of Financial Condition and Results of Operations is based upon our consolidated financial statements, which have been prepared in accordance with generally accepted accounting principles in the United States of America, or GAAP, and in accordance with the rules and regulations of the United States Securities and Exchange Commission, or SEC. We operate and report our financial information in one segment. The following discussion of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and the notes to those consolidated financial statements appearing elsewhere in Part IV, Item 15, of this Annual Report on Form 10-K.Report. This discussion contains forward-looking statements that involve significant risks and uncertainties. As a result of many factors, such as those set forth under “Risk Factors” in Part I, Item 1A of this Annual Report, on Form 10-K, our actual results may differ materially from those anticipated in these forward-looking statements.
This section of this Annual Report discusses year-to-year comparisons between 2019 and 2018, as well as other discussions of 2019 and 2018 items. We have omitted discussion of the year ended December 31, 2017 (the earliest of the three years covered by our consolidated financial statements presented in this report) as permitted by the SEC’s recent amendments to Regulation S-K. The complete Management’s Discussion and Analysis of Financial Condition and Results of Operations for year-to-year comparisons between 2018 and 2017 and other discussions of 2017 items can be found within Part II, Item 7, to our Annual Report filed with the SEC on February 28, 2019.
Overview
Recent Highlights and Developments
In March 2017,December 2019, we announced positive top-line results from our Phase 3 PLAY study of EXPAREL administered as a single-dose infiltration in pediatric patients undergoing spinal or cardiac surgeries. Overall findings were consistent with the pharmacokinetic and safety profiles for adult patients with no safety concerns identified at a dose of 4 mg/kg. These results will provide the foundation for our sNDA submission in the first half of 2020 to the FDA seeking expansion of the EXPAREL label to include children aged six and over.
In January 2020, we announced a collaboration with Envision Physician Services to train anesthesiology clinicians on ultrasound-guided regional anesthesia techniques utilizing long-acting local anesthetics like EXPAREL via a series of interactive workshops held across the country. The program supports ongoing efforts by both organizations to advance the delivery of high-quality, patient-centered care.
In January 2020, we announced positive results from our Phase 4 multicenter, randomized, double-blind, controlled, parallel group trialopioid-free CHOICE study of EXPAREL in patients undergoing C-section. The study achieved its primary endpoint with a primary unilateral TKA. The trial compared EXPAREL-based local analgesia infiltration to standard bupivacaine-based local analgesia infiltration, each as partstatistically significant reduction in total postsurgical opioid consumption while maintaining pain scores through 72 hours (p≤0.001). EXPAREL demonstrated statistical significance for the key secondary endpoint of a standard multi-modal analgesic protocol. Patients were randomized to receive local infiltration analgesia with EXPAREL admixed with bupivacaine and expanded in volume to local infiltration analgesia with bupivacaine expanded in volume. The trial met its co-primary endpoints for postsurgical pain control (p=0.0381) and opioid reduction (p=0.0048). We plan to report the statistical results for certain key secondary endpoints from this study in the first quarterincidence and severity of 2017. The fullitching for 72 hours after surgery (p≤0.05). Full study results will be submitted for publication in athe peer-reviewed medical journal.literature later this year.
Results of Operations
Comparison of the Years Ended December 31, 2016, 20152019 and 20142018
Revenues
The following table provides information regarding our revenues during the periods indicated, including percent changes (dollar amounts in thousands):
| Year Ended December 31, | 2016 versus 2015 | 2015 versus 2014 | Year Ended December 31, | % Increase / (Decrease) | |||||||||||||||||||||||
| 2016 | 2015 | 2014 | % Increase / (Decrease) | 2019 | 2018 | ||||||||||||||||||||||
| Net product sales: | |||||||||||||||||||||||||||
| EXPAREL | $ | 265,802 | $ | 239,851 | $ | 188,528 | 11 | % | 27 | % | $ | 407,877 | $ | 331,112 | 23 | % | |||||||||||
| DepoCyt(e) and other product sales | 4,271 | 4,636 | 4,998 | (8 | )% | (7 | )% | ||||||||||||||||||||
| Bupivacaine liposome injectable suspension | 3,153 | 1,315 | 100% + | ||||||||||||||||||||||||
| Total EXPAREL / bupivacaine liposome injectable suspension net product sales | 411,030 | 332,427 | 24 | % | |||||||||||||||||||||||
| iovera° | 7,896 | — | N/A | ||||||||||||||||||||||||
| Total net product sales | 270,073 | 244,487 | 193,526 | 10 | % | 26 | % | 418,926 | 332,427 | 26 | % | ||||||||||||||||
| Collaborative licensing and milestone revenue | 3,426 | 1,426 | 1,287 | 140 | % | 11 | % | — | 3,000 | (100 | )% | ||||||||||||||||
| Royalty revenue | 2,872 | 3,084 | 2,855 | (7 | )% | 8 | % | 2,100 | 1,850 | 14 | % | ||||||||||||||||
| Total revenues | $ | 276,371 | $ | 248,997 | $ | 197,668 | 11 | % | 26 | % | $ | 421,026 | $ | 337,277 | 25 | % | |||||||||||
EXPAREL revenue grew 11% and 27%23% in the years ended December 31, 2016 and 2015, respectively,2019 compared to 2018, primarily due to increasesan increase in net product sales volume of 10%EXPAREL units of 27% and 21%a 3% increase in each respective period.gross selling price per unit, partially offset by the mix of EXPAREL product sizes. The demand for EXPAREL has continued to increase as a result of a number of key growth initiatives, such as the expansion of the EXPAREL label in April 2018 to include interscalene brachial plexus nerve block which has resulted in rapid adoption among anesthesiologists, the success of our co-promotion agreement with DePuy Synthes Sales, Inc., or DePuy Synthes, and the
continued implementation of EXPAREL-based Enhanced Recovery After Surgery (ERAS) protocols across a wide range of surgical procedures. All of these factors are driving growth in new accounts and growth within existing accounts which has been driven bydue to the continued adoption of EXPAREL use inas a critical component of multimodal pain management strategies for soft tissue and orthopedic procedures. The remainingIn 2019, there was also an increase in EXPAREL revenue was due to 5% price increases in April 2015 and May 2014, partially offset by lower pricing on government sales from our participation in the Federal Supply Schedule beginning in the third quarter of 2015.
As part of the MyoScience Acquisition, we acquired iovera°. Net product sales decreased 7% in 2015 primarily dueof iovera° were $7.9 million for the year ended December 31, 2019 (attributable to the decreasepost-closing period of April 10, 2019 to December 31, 2019). Thus far, we have seen the greatest iovera° demand as pain relief for patients in the valueadvance of TKA procedures and in chronic pain management, particularly for people with mild to severe osteoarthritis of the Euro reflected in European sales and a decrease in domestic DepoCyt(e) sales volume.knee.
Collaborative licensing and milestone revenue of 140%decreased 100% in 2016 compared2019 versus 2018 due to 2015 was a result of $2.0$3.0 million upfront payment earned in milestones earnedJune 2018 under oura license agreement with Aratana Therapeutics, Inc., or Aratana,Nuance Biotech Co. Ltd. for the development and commercialization of bupivacaine liposome injectable suspension for animal health indications. The increaseEXPAREL in collaborative licensingChina.
In both 2019 and milestone2018, royalty revenue of 11% in 2015 versus 2014 was primarily driven by a full versus a partial year of amortized revenue on an $8.0 million upfront payment received in May 2014 from Mundipharma International Corporation Limited, or Mundipharma. The payment, which is being recognized on a straight-line basis over the contractual term expiring in June 2033, was consideration for extending the term of the existing supply and distribution agreements and expanding the territory where Mundipharma can market and distribute DepoCyte.
Cost of Goods Sold
Cost of goods sold primarily relates to the costs to produce, package and deliver our products to customers. These expenses include labor, raw materials, manufacturing overhead and occupancy costs, depreciation of facilities, royalty payments, quality control and engineering.
The following table provides information regarding cost of goods sold and gross margin during the periods indicated, including our gross margin percentagepercent changes (dollar amounts in thousands):
| Year Ended December 31, | 2016 versus 2015 | 2015 versus 2014 | Year Ended December 31, | % Increase / (Decrease) | |||||||||||||||||||||||
| 2016 | 2015 | 2014 | % Increase / (Decrease) | 2019 | 2018 | ||||||||||||||||||||||
| Cost of goods sold | $ | 110,104 | $ | 71,837 | $ | 77,440 | 53 | % | (7 | )% | $ | 106,712 | $ | 86,845 | 23 | % | |||||||||||
| Gross margin | 60 | % | 71 | % | 61 | % | 75 | % | 74 | % | |||||||||||||||||
Research and Development Expenses
Research and development expenses primarily consist primarily of costs attributablerelated to clinical trials and related outside services, stock-based compensation expensesproduct development and other research and development costs, including Phase 4 trials that we are required as a
The following table provides a breakout of our research and development expenses during the periods indicated, including percent changes (dollar amounts in thousands):
| Year Ended December 31, | 2016 versus 2015 | 2015 versus 2014 | Year Ended December 31, | % Increase / (Decrease) | |||||||||||||||||||||||
| 2016 | 2015 | 2014 | % Increase / (Decrease) | 2019 | 2018 | ||||||||||||||||||||||
| Clinical development | $ | 23,566 | $ | 12,609 | $ | 5,518 | 87 | % | 129 | % | |||||||||||||||||
| Product development and other | 18,815 | 10,919 | 6,723 | 72 | % | 62 | % | ||||||||||||||||||||
| Clinical and preclinical development | $ | 31,055 | $ | 18,630 | 67 | % | |||||||||||||||||||||
| Product development and manufacturing capacity expansion | 29,724 | 28,454 | 4 | % | |||||||||||||||||||||||
| Regulatory and other | 6,226 | 4,670 | 33 | % | |||||||||||||||||||||||
| Stock-based compensation | 3,297 | 5,134 | 6,490 | (36 | )% | (21 | )% | 5,114 | 3,934 | 30 | % | ||||||||||||||||
| Total research and development expense | $ | 45,678 | $ | 28,662 | $ | 18,731 | 59 | % | 53 | % | $ | 72,119 | $ | 55,688 | 30 | % | |||||||||||
| % of total revenue | 17 | % | 12 | % | 9 | % | 17 | % | 17 | % | |||||||||||||||||
Total research and development expensesexpense increased 59%30% in 20162019 versus 2015 largely due to an $11.0 million2018. The 67% increase in clinical and preclinical development expenses driven by theexpense in 2019 versus 2018 was primarily related to completed enrollment of thein our Phase 3 Pediatric (“PLAY”) clinical trial and our Phase 4 infiltration trial in TKA and two Phase 3 nerve block trials, including a femoral nerve block in subjects undergoing TKA and a brachial plexus block in patients undergoing total shoulder arthroplasty, or rotator cuff repair procedures. We also incurred start-up expenses in our spineOpioid Free C-Section (“CHOICE”) clinical trial and costs for planning pediatric trials. Increased costs also include a larger clinical workforce, which is managing our increasing investment in research and development initiatives. The increase in clinical development expense was partially offset by a decrease in research grants. Product development and other expenses increased $7.9 million which reflects our investments in the development of a new EXPAREL DepoFoam spray manufacturing process, DepoMLX and DepoTXA, the latter of which is now in Phase 2 clinical development, along with increased depreciation on our new research and development facility placed into service in August 2015. Expenses for investigational runs and development of a new analytical test for the stability testing attribute are also included in costs for product development and other. Stock-based compensation decreased 36%, which was largely attributable to the requirement to revalue non-employee grants.
Product development and manufacturing capacity expansion expense increased 4% in 2019 versus 2018 due to increased spend in EXPAREL support for in vitro release testing, a significant scale-up of our manufacturing capacity for EXPAREL in Swindon, England in partnership with Thermo Fisher and iovera° development costs to improve the functionality of the device and enhance the iovera° product line.
Regulatory and other expense increased $4.2 million, reflecting33% in 2019 versus 2018 due to activities related to our investmentsEuropean Marketing Authorization Application (MAA) and Health Canada submissions for EXPAREL and the dissemination and publication of EXPAREL data in the development of a new EXPAREL DepoFoam spray manufacturing process, DepoTXA, DepoMLX and additional pre-clinical expenses driven by DepoFoam toxicology trials. response to medical information queries.
Stock-based compensation decreased 21%, which was largely attributableincreased 30% in 2019 versus 2018 primarily due to an increase in personnel as well as the requirement to revalue non-employee grants.number of awards granted during 2019 and the fourth quarter of 2018.
Selling, General and Administrative Expenses
Sales and marketing expenses primarily consist of compensation and benefits for our sales force and personnel that support our sales, marketing, medical and scientific affairs operations, commission payments to our marketing partners for the promotion and sale of EXPAREL and iovera°, expenses related to communicating the health outcome benefits of EXPAREL patients and educational programs for our customers. General and administrative expenses consist of compensation and benefits for legal, finance, regulatory activities related to approved products and indications, compliance, information technology, human resources, business development, executive management and other supporting personnel. It also includes professional fees for legal, audit, tax and consulting services. Stock-based compensation expense relates to the costs of stock option grants, RSU awards and our ESPP.
The following table provides information regarding selling, general and administrative expenses during the periods indicated, including percent changes (dollar amounts in thousands):
| Year Ended December 31, | 2016 versus 2015 | 2015 versus 2014 | Year Ended December 31, | % Increase / (Decrease) | |||||||||||||||||||||||
| 2016 | 2015 | 2014 | % Increase / (Decrease) | 2019 | 2018 | ||||||||||||||||||||||
| Sales and marketing | $ | 89,218 | $ | 77,733 | $ | 65,010 | 15 | % | 20 | % | $ | 129,663 | $ | 107,106 | 21 | % | |||||||||||
| General and administrative | 41,882 | 39,088 | 26,902 | 7 | % | 45 | % | 47,248 | 46,846 | 1 | % | ||||||||||||||||
| Stock-based compensation | 21,513 | 22,222 | 14,750 | (3 | )% | 51 | % | 23,871 | 23,313 | 2 | % | ||||||||||||||||
| Total selling, general and administrative expenses | $ | 152,613 | $ | 139,043 | $ | 106,662 | 10 | % | 30 | % | $ | 200,782 | $ | 177,265 | 13 | % | |||||||||||
| % of total revenue | 55 | % | 56 | % | 54 | % | 48 | % | 53 | % | |||||||||||||||||