UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark one) |
☒ | | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December31, 20202023 |
OR |
☐ | | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to |
Commission file number: 000-24477001-37942

Diffusion PharmaceuticalsCervoMed Inc.
(Exact Name of Registrant as specified in its Charter)
Delaware | 30-0645032
|
| (State or Other Jurisdiction of Incorporation or Organization) | 30-0645032 (I.R.S. Employer Identification No) |
1317 Carlton Avenue, Suite 200 | |
Charlottesville, VA | 22902 |
20 Park Plaza, Suite 424
Boston, Massachusetts
(Address of Principal Executive Offices) | 02116 (Zip Code) |
(434) 220-0718
(617) 744-4400
(Registrant's telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of Each Class | Trading Symbol | Name of Each Exchange on Which Registered |
Common Stock, par value $0.001 per share | DFFNCRVO
| The NasdaqNASDAQ Capital Market
|
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ | Accelerated filer ☐ |
Non-accelerated filer ☒ | Smaller reporting company ☒ |
Emerging growth company ☐ | |
If an emerging growth company, indicate by check mark if the registrant has elected to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicated by check mark whether the registrant has filed a report on and attestation to its management's assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
The aggregate market value of the registrant’s common stock beneficially owned by non-affiliates of the registrant, calculated based upon the closing sale price of the common stock as quoted by the Nasdaq Capital Market on June 30, 20202023 (the last business day of the registrant’s second fiscal quarter), was approximately $62.4$6.8 million.
As of March 12, 2021, 101,903,97926, 2024, 6,170,479 shares of common stock of the registrant were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant's definitive Proxy Statement on Schedule 14A for its 2021 Annual Meeting of StockholdersThe following documents (or parts thereof) are incorporated by reference into the indicatedfollowing parts of this Form 10-K, as specified10-K: Certain information required in the responses to the item numbers involved. Such Proxy Statement will be filed within 120 days after December 31, 2020, the endPart III of the fiscal year to which this Annual Report relates.on Form 10-K is incorporated by reference from the Registrant’s Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission.
TABLE OF CONTENTS
PART I | | 1Page No.
|
| | | |
ITEM 1. | BUSINESSIntroductory Notes
| 1 |
| | |
Part I | | |
| | |
Item 1: | 1Business
| 5 |
Item 1A: | Risk Factors | 45 |
Item 1B: | Unresolved Staff Comments | 89 |
Item 1C: | Cybersecurity | 89 |
Item 2: | Properties | 90 |
Item 3: | Legal Proceedings | 91 |
Item 4: | Mine Safety Disclosures | 91 |
| | | |
ITEM 1A.Part II | | |
| | |
RISK FACTORSItem 5:
| 21Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
| 92 |
Item 6: | [Reserved] | 92 |
Item 7: | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 93 |
Item 7A: | Quantitative and Qualitative Disclosure About Market Risk | 101 |
Item 8: | Financial Statements and Supplementary Data | 102 |
Item 9: | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 124 |
Item 9A: | Controls and Procedures | 124 |
Item 9B: | Other Information | 125 |
Item 9C: | Disclosures Regarding Foreign Jurisdictions that Prevent Inspections | 125 |
| | | |
ITEM 1B.Part III | | |
| | |
UNRESOLVED STAFF COMMENTSItem 10:
| 36Directors, Executive Officers and Corporate Governance
| 126 |
Item 11: | Executive Compensation | 126 |
Item 12: | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 126 |
Item 13: | Certain Relationships and Related Transactions, and Director Independence | 126 |
Item 14: | Principal Accountant Fees and Services | 126 |
| | | |
ITEM 2. | PROPERTIESPart IV
| 36
| |
| | | |
ITEM 3. | LEGAL PROCEEDINGSItem 15:
| 36Exhibit and Financial Statement Schedules
| 127 |
| | |
ITEM 4. | MINE SAFETY DISCLOSURESItem 16:
| 36
|
| | |
PART II | | 37
|
| | |
ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIESForm 10-K Summary
| 37131
|
| | |
ITEM 6. | SELECTED FINANCIAL DATA
| 37
|
| | |
ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
| 38
|
| | |
ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURE ABOUT MARKET RISK
| 45
|
| | |
ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
| 46
|
| | |
ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
| 62
|
| | |
ITEM 9A. | CONTROLS AND PROCEDURES
| 62
|
| | |
ITEM 9B. | OTHER INFORMATION
| 62
|
| | |
PART III | | 63
|
| | |
ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
| 63
|
| | |
ITEM 11. | EXECUTIVE COMPENSATION
| 63
|
| | |
ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
| 63
|
| | |
ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
| 63
|
| | |
ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES
| 63
|
| | |
PART IV | | 64
|
| | |
ITEM 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES
| 64
|
| | |
ITEM 16. | FORM 10-K SUMMARY
| 67
|
INTRODUCTORY NOTES
Note Regarding Company References and Other Defined Terms
UnlessAs previously disclosed in our Current Report on Form 8-K filed on August 17, 2023 with the SEC, on August 16, 2023, the Delaware corporation formerly known as “Diffusion Pharmaceuticals Inc.” completed a merger transaction in accordance with the terms and conditions of the Agreement and Plan of Merger, dated March 30, 2023 (the “Merger Agreement”) by and among Diffusion Pharmaceuticals Inc. (“Diffusion”), Dawn Merger Inc., a wholly-owned subsidiary of Diffusion (“Merger Sub”) and EIP Pharma, Inc. (“EIP “), pursuant to which Merger Sub merged with and into EIP, with EIP surviving the Merger a wholly-owned subsidiary of Diffusion (the “Merger”). Additionally, on August 16, 2023, Diffusion changed its name from “Diffusion Pharmaceuticals Inc.” to “CervoMed Inc.”
Prior to the Effective Time (as defined below), in connection with the transactions contemplated by the Merger Agreement, Diffusion effected a reverse stock split of the Company’s common stock, par value $0.001 per share (“common stock”), at a ratio of 1-for-1.5 (the “Reverse Stock Split”). At the Effective Time, each outstanding share of EIP capital stock was converted into the right to receive 0.1151 shares of Company common stock.
For accounting purposes, the Merger is treated as a reverse recapitalization under US GAAP and EIP is considered the accounting acquirer. Accordingly, EIP’s historical results of operations are deemed the Company’s historical results of operations for all periods prior to the Merger and, for all periods following the Merger, the results of operations of the combined company will be included in the Company’s financial statements. Following the completion of the Merger, the business conducted by the Company became primarily the business conducted by EIP.
Accordingly, unless the context otherwise requires, all references in this Annual Report to (i) references to“CervoMed,” the “Company,” “we,” “our,” or “us”“us,” refer to Diffusion Pharmaceuticals Inc.the business of EIP for all dates and its subsidiariesperiods prior to August 16, 2023 and to the business of CervoMed for all dates and periods subsequent to (and including) August 16, 2023 and (ii) references to “common stock” refer to the common stock, par value $0.001 per share, of the Company. Company, after giving effect to the Reverse Stock Split. Historical share and per share figures of EIP have been retroactively restated based upon the exchange ratio of 0.1151.
We have also used several other defined terms in this Annual Report, many of which are explained or defined below:
Term | Definition |
2015 Equity Plan | Diffusion PharmaceuticalsCervoMed Inc. 2015 Equity Incentive Plan, as amended
|
2017 Tax Act2018 Plan
| Tax CutsCervoMed Inc. 2018 Employee, Director and Jobs ActConsultant Equity Incentive Plan, as amended
|
2020 Notes | the previously outstanding convertible promissory notes of 2017EIP, dated as of December 4, 2020, as amended |
2021 Notes | the previously outstanding convertible promissory notes of EIP, dated as of December 10, 2021, as amended |
2022 Notes Amendment | the amendments to the 2020 Notes entered into in April 2022 |
| 2022 Sales Agreement | our At-The-Market Sales Agreement, dated July 22, 2022, with BTIG, as agent |
2023 Notes Amendment | the amendments to the 2020 Notes and 2021 Notes entered into in June 2023 |
| 2024 Private Placement | our private placement of an aggregate of 2,532,285 units, each consisting of (i) (A) one share of common stock or (B) one Pre-Funded Warrant in lieu thereof and (ii) one Series A Warrant, for aggregate gross proceeds of up to approximately $149.4 million, announced March 28, 2024 and expected to be completed on or about April 1, 2024 |
401(k) Plan | Diffusion PharmaceuticalsCervoMed Inc. 401(k) Defined Contribution Plan
|
Affordable Care ActAD
| U.S. Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act
|
ANDA
| abbreviated new drug applicationAlzheimer’s Disease
|
Annual Report | this Annual Report on Form 10-K |
ACA | Affordable Care Act and the Healthcare and Education Reconciliation Act |
ACR20 | American College of Rheumatology 20 |
AIA | America Invents Act |
AKS | anti-kickback statute |
AMP | average manufacturer price |
ANDA | abbreviated new drug application |
API | active pharmaceutical ingredient |
ARDS
| acute respiratory distress syndrome
|
ASC | Accounting Standard Codification of the FASB |
ASC 740-10 | ASC Subtopic 740-10, Accounting for Uncertainty of Income Taxes |
ASC 815-40AscenD-LB Trial
| ASC 815-40, Derivatives and Hedging, Contractsour Phase 2a clinical trial evaluating neflamapimod for the treatment of patients with DLB, completed in an Entity's Own Equitythe second half of 2021
|
ASUsASU
| Accounting Standards Updates of the FASBUpdate |
ASU 2018-07Bayh-Doyle Act
| ASU 2018-07, Compensation--Stock Compensation (Topic 718): Improvements to Non-employee Share Based Payment AccountingBayh-Dole Act of 1980
|
Black-Scholes ModelBID
| Black-Scholes-Merton derivative investment instrument pricing modeltwice daily
|
BFC | basal forebrain cholinergic |
| BTIG | BTIG LLC |
Board | ourthe board of directors of the Company
|
BylawsCARES Act
| Coronavirus Aid, Relief, and Economic Security Act |
CCPA | the Company's bylaws, as amendedCalifornia Consumer Privacy Act |
COVID-19CPRA
| Corona Virusthe California Privacy Rights Act
|
CDR-SB | Clinical Dementia Rating Sum of Boxes test |
CGIC | the Alzheimer’s Disease 2019, the novel coronavirus disease known as COVID-19, caused by SARS-CoV-2 infectionCooperative Study-Clinician Global Impression of Change |
cGMP | current good manufacturing practices |
ChAT+ neurons | neurons staining positively for choline acetyl transferase |
CMC | chemistry, manufacturing and controls |
CMO | contract manufacturing organization |
CMS | the U.S. Centers for Medicare & Medicaid Services |
Convertible Notes | collectively, the 2020 Notes and the 2021 Notes |
CNS | central nervous system |
Code | the U.S. Internal Revenue Code of 1986, as amended |
CREATES Act | the Creating and Restoring Equal Access to Equivalent Samples Act of 2019 |
CRL | Complete Response Letter |
CRO | contract research organization |
CTACSF
| clinical trial applicationcerebrospinal fluid
|
December 2019 OfferingDSCSA
| our registered direct public offering and sale of 6,266,787 shares of common stock and concurrent private placement of warrants to purchase up to 6,266,787 shares of common stock completed in December 2019Drug Supply Chain Security Act
|
Diffusion LLCDGM
| Diffusion Pharmaceuticals LLC, a Virginia limited liability company and our wholly owned subsidiarydeep grey matter
|
DLCODLB
| diffusion capacity of lung for carbon monoxidedementia with Lewy bodies
|
Dodd-Frank ActDNP
| Dodd-Frank Wall Street Reform and Consumer Protection Actthe FDA’s Division of 2010Neurology Products
|
E.U.EEA
| European UnionEconomic Area |
ETASUEEG
| elementselectroencephalogram
|
Effective Time | the effective time of the Merger on August 16, 2023 |
EIP Common Stock | the common stock, par value $0.001, of EIP issued and outstanding prior to assure safe usethe Merger |
EMA | European Medicines Agency |
EOAD | Early Onset Alzheimer’s Disease |
EOT | end of treatment |
Exchange Act | Securities Exchange Act of 1934, as amended |
Exchange Ratio | the “Exchange Ratio” as defined in the Merger Agreement |
FASB | Financial Accounting Standards Board |
FCPA | the Foreign Corrupt Practices Act |
FDA | U.S. Food and Drug Administration |
FDC ActFDCA
| Federal Food, Drug, and Cosmetic Act |
February 2021 OfferingFDIC
| our public offering and sale of 33,658,538 shares of common stock completed in February 2021Federal Deposit Insurance Corporation
|
FTC | Federal Trade Commission |
FTD | frontotemporal dementia |
G&A | general and administrative |
GAAP
| U.S. generally accepted accounting principles
|
GBM | glioblastoma multiforme brain cancer |
GCP | good clinical practice |
GDPR | European Union General Data Protection Regulation |
GLP | good laboratory practice |
HIPAA | the Health Insurance Portability and Accountability of Act of 1996 |
HITECHHVLT
| Health Information Technology for Economic and Clinical Health Act of 2009Hopkins Verbal Learning Test
|
IMM | irreversible morbidity and mortality |
IND | investigational new drug application |
IPR&DIRA
| in-process research and developmentInflation Reduction Act of 2022
|
IRB | institutional review board |
January 2018 OfferingIT
| our public offering and sale of 1,131,375 shares of common stock and warrants to purchase up to 1,131,375 shares of common stock completed in January 2018information technology
|
May 2019 OfferingLOAD
| our registered direct public offering and sale of 1,317,060 shares of common stock and concurrent private placement of warrants to purchase up to 1,317,060 shares of common stock completed in May 2019late onset AD
|
May 2020 Investor Warrant ExerciseMA
| the exercise of the Prior Warrant in May 2020 pursuant to a warrant exercise agreementmarketing authorization
|
May 2020 OfferingMCI
| our registered direct public offering and sale of 11,428,572 shares of common stock completed in May 2020mild cognitive impairment
|
MRI | magnetic resonance imaging |
MSN | medial septal nucleus |
Nasdaq | Nasdaq Stock Market, LLC |
NbM | Nucleus basalis of Meynert |
NCE | new chemical entity |
NDA | new drug application |
NIIDNGF
| nerve growth factor |
NIA | the National Institute on Aging of the National Institutes of Health |
NIA Grant | the $21 million grant awarded to us by the NIA in January 2023 to support the RewinD-LB Trial |
NIH | National InstituteInstitutes of Infectious Diseases in Bucharest, RomaniaHealth |
NOL | net operating loss |
November 2019 OfferingNTB
| our public offering and sale of 5,104,429 shares of common stock, pre-funded warrants to purchase up to 6,324,143 shares of common stock, and warrants to purchase up to 22,857,144 shares of common stock completed in November 2019Neuropsychological Test Battery
|
PaO2NYSE
| partial pressure of blood oxygenNew York Stock Exchange
|
Planned Phase 2 Hypoxia-related Indication Trialp38α | a Phase 2, controlled, clinical outcome study evaluating TSC in one or more appropriate hypoxiarelated indications that we intend to initiate assuming success in one or more of the TSC Oxygenation Trialsp38 mitogen-activated protein kinase alpha |
Prior WarrantPBM
| a previously outstanding warrantpharmacy benefit manger
|
PD | Parkinson’s disease |
PDAB | prescription drug affordability board |
PDD | Parkinson’s disease dementia |
PDMA | Prescription Drug Marketing Act |
PDUFA | Prescription Drug User Fee Act, as amended |
PET | positron emission tomography |
POC | proof-of-concept |
PPA | primary progressive aphasia |
| Pre-Funded Warrants | the pre-funded warrants each to purchase up to 5,000,000 sharesone share of common stock at an exercisea purchase price of $0.35$0.001 per share expected to be issued in connection with the 2024 Private Placement |
PREA | Pediatric Research Equity Act |
Proxy Statement | ourthe definitive proxy statement on Schedule 14A for our 20212024 Annual Meeting of Stockholders to be filed with the SEC within 120 days after December 31, 2020, the end of the fiscal year to which this Annual Report relates
|
ptau181 | plasma phosphorylated tau at position 181 |
RA | rheumatoid arthritis |
R&D | research and development |
| Registration Statement | Amendment No. 2 to our Registration Statement on Form S-4, filed with the SEC on July 11, 2023, as amended from time to time |
Regulation S-K | Regulation S-K promulgated under the Securities Act |
REMS | risk evaluationRisk Evaluation and mitigation strategyMitigation Strategy
|
SARS-CoV-2RewinD-LB Trial
| severe acute respiratory syndrome coronavirus 2,our Phase 2b clinical trial evaluating neflamapimod for the virus responsible for COVID-19treatment of patients with DLB, initiated in the second quarter of 2023
|
RLD | reference-listed drug |
ROU | right-of-use |
SAB | scientific advisory board |
SAE | serious adverse events |
SEC | U.S. Securities and Exchange Commission |
Securities ActSection 382
| Section 382 of the Code |
| Securities Act | Securities Act of 1933, as amended |
| Series A Warrants | the warrants to purchase an aggregate of 2,532,285 shares of common stock at a purchase price of $39.24 per share expected to be issued in connection with the 2024 Private Placement |
TCJA | Tax Cuts and Jobs Act of 2017 |
SOXTID
| Sarbanes-Oxley Act of 2002, as amended
|
Tax Code
| U.S. Internal Revenue Code of 1986, as amended
|
TCOM
| transcutaneous oxygen measurement
|
| |
tPA
| tissue plasminogen activatorthree times daily
|
TSC | trans sodium crocetinate |
TSC COVID TrialTUG
| our Phase 1b clinical trial evaluating TSC in hospitalized COVID-19 patients, completed in February 2021Timed Up and Go test
|
TSC DLCO TrialUPL
| our planned Phase 2a clinical trial evaluating the effects of TSC through the measure of DLCO through the lungs as a surrogate measure of oxygen transfer efficiency in patients with previously diagnosed interstitial lung disease who have a baseline DLCO test that is abnormal
|
TSC GBM Trial
| our Phase 3 clinical trial evaluating TSC in a newly diagnosed inoperable GBM patient population initiated in December 2017
|
TSC Induced Hypoxia Trial
| our planned Phase 1b clinical trial evaluating the effects of TSC on VO2 and PaO2 in normal healthy volunteers exposed to conditions that induce hypoxia
|
TSC Oxygenation Trials
| collectively, the TSC TCOM Trial, the TSC Induced Hypoxia Trial, and the TSC DLCO Trial
|
TSC Stroke Trial
| our Phase 2 clinical trial evaluating TSC in the treatment of acute ischemic or hemorrhagic stroke, initiated in October 2019
|
TSC TCOM Trial
| our planned Phase 1b clinical trial evaluating the effects of TSC on peripheral tissue oxygenation in healthy normal volunteers using a TCOM device, which we expect to commence before the end of March 2021upper payment limit
|
U.S. | United States of America |
US GAAP | U.S. generally accepted accounting principles |
USPTO | U.S. Patent and Trademark Office |
VO2Vertex
| maximal oxygen consumptionVertex Pharmaceuticals Incorporated
|
Vertex Agreement | the Option and License Agreement, dated as of August 27, 2012, by and between EIP Pharma LLC and Vertex, as amended |
Explanatory Note Regarding 2024 Private Placement
On March 28, 2024, we entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The aggregate upfront gross proceeds from the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and additional gross proceeds of up to approximately $99.4 million may be received if the Series A Warrants are exercised in full for cash.
The information contained in this Annual Report, including our consolidated financial statements set forth in, “Part II — Item 8 — Financial Statements” and the information regarding our liquidity, capital resources and cash runway set forth in, “Part II --- Item 7 – Management’s Discussion and Analysis of Financial Condition and Results of Operations,” does not reflect the anticipated consummation of, or our anticipated receipt of proceeds from, the 2024 Private Placement. For additional information regarding the 2024 Private Placement, the terms thereof (including the conditions to closing), and our expected use of the net proceeds therefrom, refer to our Current Report on Form 8-K filed with the SEC on March 28, 2024.
Note Regarding Forward-Looking Statements
This Annual Report (including, for purposes of this Note Regarding Forward-Looking Statements, any information or documents incorporated herein by reference) includes express and implied forward-looking statements. By their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics and industry change, and depend on the economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated. Although we believe that we have a reasonable basis for each forward-looking statement contained in this Annual Report, we caution you that forward-looking statements are not guarantees of future performance and that our actual results of operations, financial condition, liquidity, and prospects may differ materially from the forward-looking statements contained in this Annual Report. In addition, even if our results of operations, financial condition, liquidity, and prospects are consistent with the forward-looking statements contained in this Annual Report, they may not be predictive of actual results or reflect unanticipated developments in future periods.
Forward-looking statements appear in a number of places throughout this Annual Report. We may, in some cases, use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately,” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Forward-looking statements also include statements regarding our intentions, beliefs, projections, outlook, analyses or expectations including our intentions, beliefs, projections, outlook, analyses, or expectations concerning, among other things:
the success and timing of our clinical and preclinical studies, including our ability to enroll subjects in our ongoing and planned clinical studies at anticipated rates;
our ability to obtain and maintain regulatory approval of our product candidates and, if approved, our products, including the labeling under any approval we may obtain;
our plans and ability to develop and commercialize our product candidates and the outcomes of our research and development activities;
the accuracy of our estimates of the size and characteristics of the potential markets for our product candidates, the rate and degree of market acceptance of any of our product candidates that may be approved in the future, and our ability to serve those markets;
the success of products that are or may become available which also target the potential markets for our product candidates;
obtaining and maintaining intellectual property protection for our product candidates and our proprietary technology;
our ability to operate our business without infringing the intellectual property rights of others and the potential for others to infringe upon our intellectual property rights;
our failure to recruit or retain key scientific or management personnel or to retain our executive officers;
the performance of third parties, including contract research organizations, manufacturers, suppliers, and outside consultants, to whom we outsource certain operational, staff and other functions;
our ability to obtain additional financing in the future and continue as a going concern;
our estimates regarding expenses, future revenues, capital requirements, and needs for additional financing;
regulatory developments in the U.S., E.U., and other foreign jurisdictions;
recently enacted and future legislation related to the healthcare system, including trends towards managed care and healthcare cost containment, the impact of any significant spending reductions or cost controls affecting publicly funded or subsidized healthcare programs, or any replacement, repeal, modification, or invalidation of some or all of the provisions of the Affordable Care Act;
any significant breakdown, infiltration, or interruption of our information technology systems and infrastructure;
our ability to satisfy the continued listing requirements of the NASDAQ Capital Market or any other exchange on which our securities may trade in the future;
uncertainties related to general economic, political, business, industry, and market conditions, including the ongoing COVID-19 pandemic; and
| | •●
| our cash balances and our ability to obtain additional financing in the future and continue as a going concern; |
| ● | the success and timing of our ongoing RewinD-LB Trial and our other clinical and preclinical studies, including our ability to enroll subjects in our studies at anticipated rates and our ability to manufacture an adequate amount of drug supply for our studies; |
| ● | obtaining and maintaining intellectual property protection for our current or future product candidates and our proprietary technology; |
| ● | the performance of third parties, including contract research organizations, manufacturers, suppliers, and outside consultants, to whom we outsource certain operational, staff and other functions; |
| ● | our ability to obtain and maintain regulatory approval of our current or future product candidates and, if approved, our products, including the labeling under any approval we may obtain; |
| ● | our plans and ability to develop and commercialize our current or future product candidates and the outcomes of our research and development activities; |
| ● | our estimates regarding expenses, future revenues, capital requirements, and needs for additional financing; |
| ● | our future obligations under the Vertex Agreement; |
| ● | our failure to recruit or retain key scientific or management personnel or to retain our executive officers; |
| ● | the accuracy of our estimates of the size and characteristics of the potential markets for our current or future product candidates, the rate and degree of market acceptance of any of our current or future product candidates that may be approved in the future, and our ability to serve those markets; |
| ● | the success of products that are or may become available which also target the potential markets for our current or future product candidates; |
| ● | our ability to operate our business without infringing the intellectual property rights of others and the potential for others to infringe upon our intellectual property rights; |
| ● | any significant breakdown, infiltration, or interruption of our information technology systems and infrastructure; |
| ● | our ability to remediate our previously disclosed material weaknesses in our internal controls over financial reporting in a timely manner; |
| ● | our ability to successfully integrate the historical businesses of EIP and Diffusion and realize the anticipated benefits of the Merger; |
| ● | recently enacted and future legislation related to the healthcare system; |
| ● | other regulatory developments in the U.S., European Union, and other foreign jurisdictions; |
| ● | our ability to satisfy the continued listing requirements of the Nasdaq or any other exchange on which our securities may trade in the future; |
| ● | uncertainties related to general economic, political, business, industry, and market conditions, including the continued availability of funding for the NIA to support disbursements under our previously received grant and |
| ● | other risks and uncertainties, including those discussed in Part I - Item 1A. - Risk Factors of this Annual Reportunder the heading "Risk Factors" herein and elsewhere in our other public filings. |
As a result of these and other factors, known and unknown, actual results could differ materially from our intentions, beliefs, projections, outlook, analyses, or expectations expressed in any forward-looking statements in this Annual Report. Accordingly, we cannot assure you that the forward-looking statements contained in this Annual Report will prove to be accurate or that any such inaccuracy will not be material. You should also understand that it is not possible to predict or identify all such factors, and you should not consider any such list to be a complete set of all potential riskrisks or uncertainties. In light of the foregoing and the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame, or at all. For all forward-looking statements, we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995.
Any forward-looking statementstatements that we make in this Annual Report speaksspeak only as of the date of such statement, and, except as required by applicable law or by the rules and regulations of the SEC, we undertake no obligation to update such statements to reflect events or circumstances after the date of this Annual Report or to reflect the occurrence of unanticipated events. Comparisons of current and any prior period results are not intended to express any ongoing or future trends or indications of future performance, unless explicitly expressed as such, and should only be viewed as historical data.
Note Regarding Stock Splits
Unless the context otherwise requires, in this Annual Report, all share and per share amounts related to our common stock give effect to (i) our 1-for-15 reverse stock split effective December 13, 2018 and (ii) our 1-for-10 reverse stock split effective August 17, 2016.
Note Regarding Trademarks, Trade Names, and Service Marks
This Annual Report contains certainincludes trademarks, trade names, and service marks of ours, including “DIFFUSIO2N.”owned by us or other companies. All othertrademarks, service marks and trade names trademarks, and service marks appearingincluded in this Annual Report are to the knowledge of Diffusion, the property of their respective owners. To the extent any such terms appear without the trade name, trademark, or service mark notice, such presentation is for convenience only and should not be construed as being used in a descriptive or generic sense.
PART I
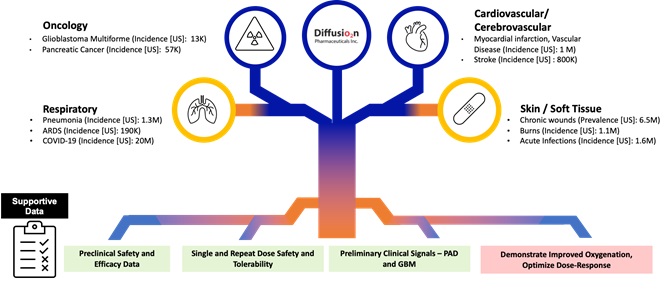
Diffusion Pharmaceuticals: Enhancing Oxygen, Fueling LifeOverview
We are a clinical-stage biotechnology company focused on developing treatments for age-related neurologic disorders. We are currently focused on the development of our lead drug candidate, neflamapimod, an innovative biopharmaceutical company developinginvestigational, orally administered, small molecule brain penetrant that inhibits p38α in the neurons (nerve cells) within the brains of people with neurodegenerative diseases. Neflamapimod has the potential to treat and improve synaptic dysfunction, the reversible aspect of the underlying disease processes in DLB and certain other major neurological disorders, and is currently being evaluated in our ongoing RewinD-LB Trial, a Phase 2b study in patients with DLB funded by a $21.0 million grant from the NIA. We expect to complete enrollment in the RewinD-LB Trial during the second quarter of 2024 and to report initial results from the placebo-controlled portion of the study during the fourth quarter of 2024.
Our novel therapiesapproach focuses on reducing the impact of inflammation in the brain, or neuroinflammation, which we believe is a key factor in the manifestation of degenerative diseases of the brain, including DLB. Chronic activation of the enzyme p38α in the neurons (nerve cells) within the brains of people with neurodegenerative diseases is believed to impair how neurons communicate through synapses (the connections between neurons). This impairment, termed synaptic dysfunction, leads to deterioration of cognitive and motor abilities. Left untreated, synaptic dysfunction can result in neuronal loss that enhance the body’s abilityleads to deliver oxygendevastating disabilities, significant reliance on a caretaker, long term care living, and, ultimately, death. However, before neuronal loss commences, disease progression in major neurodegenerative disorders, including DLB, initially involves a protracted period of functional loss, particularly with respect to the areas wheresynapses. We believe that inhibiting p38α activity in the brain, by interfering with key pathogenic drivers of disease, has the potential to reverse the clinical progression observed in early-stage neurodegenerative diseases, and that it is needed most. Our lead product candidate, TSC,possible to slow further progression by delaying permanent synaptic dysfunction and neuron death.
We believe we are a leader in the industry in developing a treatment for DLB, as we are the only company of which we are aware with an asset that has shown statistically significant improvements compared to placebo in a Phase 2a clinical trial (our AscenD-LB Trial) and has initiated a Phase 2b clinical evaluation (our ongoing RewinD-LB Trial), from which we expect initial results before the end of 2024. The clinical symptoms in DLB are most directly linked to synaptic dysfunction in cholinergic neurons (neurons producing the neurotransmitter acetylcholine) in a part of the brain named the basal forebrain. Based on available preclinical and clinical data, we believe if neflamapimod is being developedgiven in the early stages of certain degenerative diseases of the brain, it may reverse synaptic dysfunction and improve neuron health and function. In preclinical studies, neflamapimod has been shown to enhancereverse the diffusionneurodegenerative process in the BFC system. Following earlier clinical studies demonstrating blood-brain-barrier penetration, target (p38α) engagement, and identification of oxygendose-response, we obtained positive Phase 2a clinical data in patients with DLB in our AscenD-LB Trial. Specifically, statistically significant improvement was observed in patients treated with neflamapimod compared to tissuespatients treated with low oxygen levels, also knownplacebo on measures of dementia severity (as measured by CDR-SB) and functional mobility (i.e., walking ability, as hypoxia,measured by the TUG test) in the primary (intention-to-treat) analysis that includes all patients randomized into the study that had at least one measurement of the endpoint analyzed. In addition, in a serious complicationsecondary analysis, neflamapimod demonstrated statistically significant improvement compared to placebo in a battery of many of medicine’s most intractable and difficult-to-treat conditions.cognitive tests, particularly with respect to tests that measured attention.
In additionOctober 2023, the major clinical neurology journal, Neurology, published additional analyses of the AscenD-LB Trial data that further strengthened these conclusions regarding neflamapimod’s potential efficacy and identified the DLB patient population most responsive to TSC,neflamapimod treatment. In these analyses, the results were stratified by pre-treatment levels of plasma ptau181, which recent scientific literature has identified as a biomarker to differentiate DLB patients with AD-associated co-pathology – a form of mixed dementia which we sometimes refer to as “DLB+AD” – from DLB patients without AD-associated co-pathology – which we sometimes refer to as “pure DLB.” In pure DLB patients, who generally represent early-stage patients with limited neurodegeneration in the hippocampus, the treatment response to neflamapimod in the AscenD-LB Trial was substantial (Cohen’s d effect size ≥ 0.7 and statistically significant vs. placebo on the CDR-SB, TUG, cognitive tests of attention and working memory) and greater than the overall patient population. In a February 2024 publication in the Journal of Prevention of Alzheimer’s Disease, results from our product candidate DFN-529, a novel, allosteric PI3K/Akt/mTOR pathway inhibitor, is in early-stage development. We previously completed two Phase 1prior clinical trials evaluating DFN-529of neflamapimod in age-related macular degeneration. DFN-529 was also previouslyAD and DLB were integrated to show not only the demonstrated effects of neflamapimod on cognition and function, but on other biomarkers such as EEG and brain volume and functional connectivity in preclinical development in oncology, specifically GBM.the basal forebrain.
Highlights from 2020 and Early 2021
| •
| Board & Management Additions and Changes - During the second half of 2020, our leadership team changed significantly. We appointed our new chief executive officer, Robert J. Cobuzzi, Jr., Ph.D., and our new general counsel, William Elder, in September 2020, followed by our new chief medical officer, Christopher Galloway, M.D., in October 2020. We also welcomed a new director, Jane Hollingsworth, to our Board in August 2020. Dr. Cobuzzi joined the Board in January 2020.
|
| •
| Initiation and Completion of TSC COVID Trial - In September 2020, we announced the dosing of the first two patients in our TSC COVID Trial evaluating TSC in hospitalized COVID-19 patients at the NIID in Bucharest, Romania. On February 9, 2021, we completed dosing of the twenty-fourth and final patient in the TSC COVID Trial. No dose-limiting toxicities or serious adverse events were observed among any patients in the study, including those who received the highest dose of 1.5 mg/kg every 6 hours. Evaluation of secondary endpoint data is ongoing and we anticipate this data will be available early in the second quarter of 2021.
|
| •
| February 2021 Equity Offering & Selected Unaudited Financial Information - During the first quarter of 2021, we completed the February 2021 Offering resulting in aggregate gross proceeds to the Company of approximately $34.5 million, before deducting underwriting discounts and commissions and offering expenses payable by us. In addition, during the period from January 1, 2021 through March 15, 2021, certain holders of warrants to purchase common stock cash exercised those warrants resulting in aggregate gross proceeds to the Company of approximately $2.2 million. As a result, combined with our cash and cash equivalents as of December 31, 2020 of $18.5 million, we expect that our existing cash and cash equivalents will enable us to fund our operating expenses and capital expenditures (including our planned clinical trials) through 2023.
|
Our Strategic Prioritiesongoing RewinD-LB Trial is a double-blind, placebo-controlled, 16-week Phase 2b study in 160 patients with pure DLB funded by a $21.0 million grant from the NIA. The trial is intended to confirm the efficacy findings from the AscenD-LB Trial and definitively demonstrate proof-of-concept. We have utilized our subsequent analyses of the AscenD-LB data and the other information described above to optimize the RewinD-LB Trial’s design and bolster the trial’s statistical power. Critically, the RewinD-LB Trial will exclude patients with Alzheimer’s disease related co-pathology as evaluated by plasma ptau181 levels (i.e., the study will only enroll patients with pure DLB) and, to enrich for 2021such patients, the global CDR-SB score at entry will be limited to 0.5 or 1.0. Together with additional modifications to the Phase 2a design related to dosing regimen and primary endpoint, sample size calculations indicate that the RewinD-LB Phase Trial has greater than 95% statistical power (approaching 100%) to meet its primary objective of demonstrating improvement relative to placebo on change in CDR-SB over the course of the study.
SinceWe expect to complete enrollment in the foundingRewinD-LB Trial during the second quarter of Diffusion LLC,2024 and to report initial results from the placebo-controlled portion of the study during the fourth quarter of 2024. The results of the RewinD-LB Trial are intended to provide the data necessary to finalize our design of a Phase 3 clinical trial, the general framework of which, including a 24-week treatment duration, has been agreed upon with the FDA.
In addition to neflamapimod’s potential to treat DLB, we believe the benefit of targeting neuroinflammation-induced synaptic dysfunction in the BFC system can be applied to other neurologic indications in which treatment of BFC dysfunction and degeneration would be expected to be clinically beneficial, including as treatment promoting recovery in the three months after ischemic stroke, as a disease-modifying treatment for early-stage Alzheimer’s disease, and as a treatment for certain forms of frontotemporal dementia.
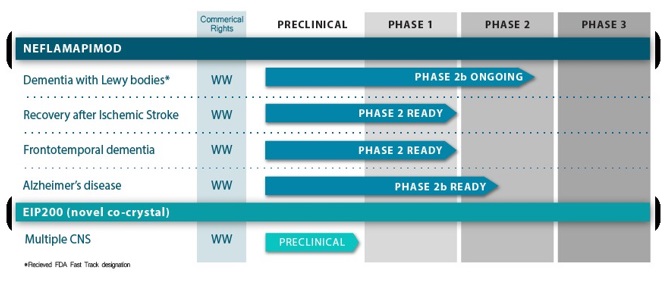
Our Pipeline
Set forth below is a table presenting our clinical pipeline:
Our Team
We have been principally focused onassembled a diverse team of experienced company builders and drug developers, complemented by an experienced Board and world-class scientific advisors. This group shares a long-term commitment to execute our strategy, advance the development of TSCneflamapimod, and improve treatment outcomes and quality of life for patients suffering from age-related neurologic disorders. Moreover, we benefit from the significant pharmaceutical development experience of our management team members and directors, several of whom have worked on neflamapimod in the past at Vertex and are well acquainted with the unique properties of the compound for application in DLB and other potential target indications.
| ● | Our Co-Founder, President and Chief Executive Officer, John Alam, M.D., is a biotech industry veteran with more than 30 years’ experience and is an industry leader in translational medicine. He has a proven track record of creating value through clinical development success, including having played major roles during the clinical development of five innovative drugs that are now on the market, and is an emerging drug development leader in neurodegenerative diseases, including having been the global head of all R&D activities directed towards neurodegenerative diseases at Sanofi S.A. (Nasdaq: SNY), a top ten global pharmaceutical company. Dr. Alam also has direct experience with neflamapimod from his time at Vertex, where he was Executive Vice President, Medicines Development and Chief Medical Officer. Dr. Alam also led the clinical development of Biogen’s first approved drug for the treatment of multiple sclerosis, Avonex. |
| ● | Our Co-Founder and Director, Dr. Sylvie Grégoire, PharmD., is also an industry veteran with more than 30 years’ experience who previously held executive leadership posts in several multinational life sciences firms. Dr. Grégoire has extensive experience with corporate governance and board operations and is currently also on the board of directors at of two public life sciences companies, Novo Nordisk A/S (NYSE: NVO) and Revvity (Nasdaq: RVTY) (formerly known as PerkinElmer, Inc. (NYSE: PKI)), and one private company, F2G; and she previously was chair of Corvidia Therapeutics (acquired by Novo Nordisk), and member of the board of directors of ViFor Pharma (acquired by CSL) and Cubist Pharmaceuticals (acquired by Merck). |
| ● | The Chair of our Board, Joshua S. Boger, Ph.D., is an industry veteran who has served in multiple scientific and business leadership roles during his multi-decade career. Dr. Boger founded Vertex in 1989 and served as its Chief Executive Officer from 1992 until 2009, and currently serves as the Executive Chairman of Alkeus Pharmaceuticals. Prior to founding Vertex, Dr. Boger was Senior Director of Basic Chemistry at Merck Sharp & Dohme Research Laboratories in Rahway, NJ, where he headed both the Departments of Biophysical Chemistry and Medicinal Chemistry of Immunology & Inflammation. |
| ● | Our Chief Financial Officer, William Tanner, Ph.D., through his more than 20 years’ experience as a healthcare research analyst at well recognized investment banks, has expertise and relevant industry experience. |
| ● | Our Chief Operating Officer, Robert J. Cobuzzi, Jr., Ph.D., has over 25 years of cross-functional executive and operational leadership experience in the pharmaceutical and biotechnology industries across the areas of corporate development, research & development, and operations, at Endo International Plc, Adolor Corporation, Diffusion Pharmaceuticals, Centocor and AstraMerck. Dr. Cobuzzi also currently serves as a Venture Partner for Sunstone Life Science Ventures and also is Chairman of Sunstone’s Business Development Advisory Board. |
| ● | Our SVP of Clinical Development, Kelly Blackburn, MHA, has more than 30 years of experience in clinical development operations, including senior management positions at aTyr Pharma and Vertex where she held senior global clinical operational responsibility for three major novel therapeutics: Kalydeco® for the treatment of cystic fibrosis, Incivek® for hepatitis C, and Velcade® for multiple myeloma. |
| ● | In addition, to provide a strong scientific underpinning for the neflamapimod program, we have surrounded ourselves with thought leaders in the fields of cell biology, intracellular signal transduction, neurotherapeutics, and translational neuroscience. Our SAB is chaired by Dr. Ole Isacson, who serves as Professor of Neurology at Harvard Medical School and is a Founding Director of the Neuroregeneration Research Institute at McLean Hospital. Other members of our SAB include Dr. Lewis Cantley, Professor of Cell Biology at the Dana Farber Cancer Institute, and who previously served as the Director of the Sandra and Edward Meyer Cancer Center at the Weill Cornell Medical Center; Dr. Jeffrey Cummings is the Joy Chambers-Grundy Professor of Brain Science at the UNLV Integrated School of Health Sciences and Director of the Chambers-Grundy Center for Transformative Neuroscience, and Director Emeritus of the Cleveland Clinic Lou Ruvo Center for Brain Health and Professor at the Cleveland Clinic Lerner College of Medicine of Case Western University; and Dr. Heidi McBride, Canada Research Chair in Mitochondrial Cell Biology and as Professor in the Department of Neurology and Neurosurgery at McGill University. |
Our Strategy
Our mission is to develop and commercialize innovative medicines that change the course of the disease of patients who suffer from age-related neurologic disorders.
The key elements of our strategy are:
| ● | Advance clinical development of neflamapimod for treatment of DLB with a focus on moving the program through to Phase 3 initiation in mid-2025. We initiated our Phase 2b RewinD-LB Trial in the second quarter of 2023 and anticipate completing enrollment in the second quarter of 2024. The efficacy data, which would come at the end of the four-month placebo-controlled portion of the trial, are expected in the fourth quarter of 2024. With those results in hand, we plan to meet with the FDA in an end-of-Phase 2 meeting to finalize the design of a single 24-week treatment duration Phase 3 clinical trial, which we are targeting to initiate in mid-2025. As the design of the Phase 3 clinical trial will largely replicate the RewinD-LB Trial design, we believe that success in the RewinD-LB Trial will be a meaningful predictor of the potential for a successful clinical outcome in our planned Phase 3 trial. |
| ● | Advance clinical development of neflamapimod for other disease indications. Neflamapimod’s mechanism of action with respect to treating neuro-inflammation and, more specifically, cholinergic dysfunction and degeneration provides opportunities to advance our drug in a range of neurologic disorders, in addition to DLB, in which targeting and treating BFC dysfunction and degeneration would be expected to provide substantial clinical benefit. Our anticipated second indication is as a three-month treatment following ischemic stroke to promote neurologic recovery, particularly of motor function. A potential third indication is as disease-modifying treatment early-stage AD, when the BFC degeneration is a major driver of disease progression. In addition, we believe there is strong scientific basis for evaluating neflamapimod in certain forms of frontotemporal dementia. |
| ● | Commercialize neflamapimod ourselves and/or in collaboration with one or more partners. If neflamapimod receives regulatory approval, we intend to be prepared to commercialize as soon as practicable in the market(s) where it is first approved, if at all, which we expect to be in North America and/or Europe. In the future, we may seek partners to seek approval and commercialize our products in other regions. |
| ● | Expand our pipeline through in-licensing and acquisitions. In the future, we intend to leverage our expertise in drug development and business development, as well as our understanding of translational neuroscience with respect to synaptic dysfunction, to opportunistically evaluate product candidates that are complementary to neflamapimod in our pursuit of novel therapies for DLB, AD and other age-related neurologic disorders. |
Neflamapimod in Dementia with Lewy Bodies
Our Approach
Our approach is based on an understanding of the mechanism by which neuroinflammation leads to the initiation and establishment of the neurodegenerative process. The process of neurodegeneration starts with dysfunction of synapses, i.e., the interconnections between neurons. Treating synaptic dysfunction has emerged as a major therapeutic objective to address progression of neurodegenerative diseases, particularly in the early stages prior to the onset of significant cell death. Importantly, in animal models, while neurodegeneration is irreversible, synaptic dysfunction has been observed to be reversible. In addition, even in animal models of rapidly progressive neurodegeneration (e.g., prion disease), interventions that reverse synaptic dysfunction both improve function and “arrest” the neurodegenerative process. Thus, therapeutic interventions that target synaptic dysfunction have the potential to both reverse and slow disease progression in the early stages of neurodegenerative dementias.
The basal forebrain, and specifically nerve cells producing the neurotransmitter acetylcholine (i.e., “cholinergic neurons”), play critical roles in controlling and optimizing a wide range of cognitive, motor, and visual tasks. Synaptic dysfunction in the basal forebrain cholinergic system is the primary pathogenic driver of disease expression and progression of DLB. Basal forebrain cholinergic dysfunction also plays a major role in disease progression in the early stages of AD, and basal forebrain cholinergic dysfunction is rate limiting for optimal recovery after ischemic stroke.
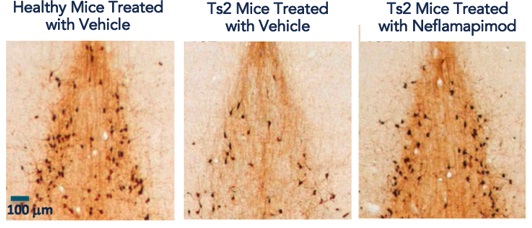
In collaborative work conducted with the New York University Langone Medical Center, and as published in the journal Nature Communications, we have demonstrated that neflamapimod targets the specific molecular mechanisms underlying basal forebrain cholinergic dysfunction, and eventually degeneration, and, as discussed in subsequent sections, can successfully reverse disease progression in animals with basal forebrain cholinergic dysfunction and degeneration.
Capitalizing on Our Strengths
We believe that the following competitive strengths will allow us to execute on our mission to develop and commercialize neflamapimod as a disease modifying innovative drug treatment for patients who suffer from DLB and other neuro-inflammatory age-related neurologic disorders:
| ● | Our approach to degenerative diseases of the brain is highly differentiated and has the potential to be the first to market specific drug therapy for DLB. Our approach focuses on reducing the impact of neuroinflammation. Neuroinflammation is directly linked with the initiation of the neurodegenerative process through synaptic dysfunction, which results in a reduction or elimination of the ability of the affected neurons to transfer information. Neflamapimod targets neuro-inflammation and, particularly, the molecular mechanisms within neurons that lead to synaptic dysfunction, thereby both improving cognitive function and slowing down the process that leads to neuronal loss. Currently, there are no approved therapies for DLB and there is limited drug development in this area, with neflamapimod being, to our knowledge, the only disease-modifying approach that has demonstrated significant improvements on clinical outcome measures in a clinical trial in DLB. |
| ● | Neflamapimod has the potential to meet a significant unmet medical need and achieve substantial commercial return. We believe that neflamapimod can address the high unmet medical need with respect to both the cognitive and motor aspects of DLB. DLB is the third most common chronic degenerative disease of the brain (after Alzheimer’s disease and Parkinson’s disease), with an estimated 700,000 individuals with the disease in each of the U.S. and European Union. Despite this prevalence and high unmet medical need, there are currently no FDA or EMA approved treatments for DLB. Further, patients are referred to neurologists to treat the disease. The specialty nature of neflamapimod, if approved, combined with the prevalence of the disease should present a significant commercial opportunity, including through reimbursement, based on the impact on patients’ quality of life and ability to function, reduction of caregiver burden and reduction of health care costs associated with DLB, among other factors. |
| ● | Neflamapimod has the potential to improve cognitive and motor function (i.e., restore function), providing the opportunity to demonstrate clinical efficacy in Phase 2 and, if successful, provide a meaningful predictor of the potential for a successful clinical outcome in Phase 3. A major challenge in developing effective drug treatments for chronic neurodegenerative diseases, particularly AD, has been that approaches to date do not show improvement in disease outcomes in Phase 2 clinical trials (i.e., trials of less than six-month duration). Instead, demonstration of clinical efficacy depends on clinical trial duration of at least 12 to 18 months and large subject numbers (~1,000 or more), effectively requiring Phase 3 trials designed to show an effect of slowing disease progression relative to placebo treatment. As a result, Phase 2 clinical trials data may not provide a meaningful predictor of the potential for a successful clinical outcome in Phase 3 in AD. In contrast, in early-stage DLB, because there is less extensive neuronal loss and fixed (i.e., irreversible) clinical deficits compared to AD, there is the potential to reverse disease progression and improve function in Phase 2 clinical trials. Neflamapimod has previously been shown to reverse disease progression and restore function in preclinical studies and has demonstrated improvement as compared to placebo on clinically meaningful outcomes in a 16-week Phase 2a clinical trial, particularly in patients with pure DLB. If the results of our AscenD-LB Trial are confirmed in the ongoing RewinD-LB Trial (the placebo-controlled portion of which will also be of 16 weeks duration) with a statistically significant difference between placebo and neflamapimod treatment on the primary endpoint, we believe we will have demonstrated proof-of-concept (i.e., have established the neflamapimod is efficacious in the treatment of DLB). In addition, based on discussions we have had with the FDA, and pending confirmation in an end-of-phase 2 meeting with the FDA that we plan to have after Phase 2b, approval for neflamapimod could be obtained with the conduct of a single 24-week treatment duration Phase 3 study involving a few hundred subjects, although there can be no assurances. As the design of the Phase 3 clinical trial will largely replicate the RewinD-LB Trial design, we believe that success in the RewinD-LB Trial will be a meaningful predictor of the potential for a successful clinical outcome in our planned Phase 3 trial. See section titled “Item 1A. Risk Factors - Risks Related to the Company’s Product Development and Regulatory Approval” for a further description of these factors and uncertainties. |
| ● | Neflamapimod has been extensively tested in animals and humans. The safety and tolerability profile has been extensively evaluated and is well understood. Specifically, long-term toxicology studies of neflamapimod have been completed and the drug has been administered to over 300 volunteers and subjects to date (including over 150 subjects in Phase 2 clinical trials in either DLB or AD), some of whom have received up to 30 times the dose we are using in our ongoing RewinD-LB Trial and currently plan to utilize in our planned Phase 3 trial. |
DLB Background
Unmet Medical Need
Dementia with Lewy bodies is the second most common neurodegenerative dementia (after AD), representing 10-20% of the dementia population. The Lewy Body Dementia Association estimates there are 1.4 million individuals in the United States affected with Lewy body dementia, which includes both PDD and non-Parkinson’s DLB. As non-Parkinson’s DLB and PDD are prevalent in the United States at an approximate ratio of 1:1, there are approximately 700,000 individuals with DLB in the United States. Furthermore, the prevalence in European countries is similar to that in the United States, and so we believe there also are approximately 700,000 individuals with DLB in the European Union as well. Despite this prevalence, there are currently no approved treatments specifically for DLB in the U.S. or the European Union.
DLB is characterized by progressive dementia and fluctuating cognition (particularly deficits in attention), visual hallucination, motor dysfunction (disturbances in gait and balance) and sleep disturbances. With respect to life expectancy, in a large cohort of DLB and AD cases (251 DLB, 222 AD), after controlling for age at diagnosis, comorbidity, and antipsychotic prescribing, the survival for DLB was shorter compared to AD, with a median (average) survival of less than four years with DLB (3.3 years for males and 4.0 for females), as compared to nearly seven years with AD (6.7 years for males and 7.0 years for females). Antecedent to death, the time progression to severe dementia is also shorter by nearly two years with DLB compared to AD.
Separate from survival and progression to severe disease, even in the mild-to-moderate stages, with deficits occurring in both cognitive and motor function, the disease burden with respect to quality of life and caregiver burden, is greater in DLB than in AD. Furthermore, patients with DLB are more frequently admitted to general hospitals and utilize inpatient care to a substantially higher degree than do those with AD or the general elderly population. Most importantly, in a large prospective study, mild dementia patients with DLB were admitted to a nursing home after only a median of 1.8 years from presentation and diagnosis, nearly two years shorter than the 3.7 years in the AD group.
Accordingly, DLB in afflicted persons often progresses quickly and severely impacts not only the daily lives of patients suffering from the disease but that of their caregivers. There are currently no disease-modifying treatments available for DLB, so management of DLB currently focuses on relief of symptoms, including its cognitive and parkinsonian (e.g., tremor) manifestations. No approaches have been shown to clinically slow neuronal loss or prevent cognitive decline, and there are no approved therapies for treating the underlying disease process or disease-modifying drugs in Phase 3 clinical trials. Though not approved for DLB, cholinesterase inhibitors are used in its management, with some limited and transient improvement in cognition and a reduction in the frequency and severity of visual hallucinations. However, despite treatment with cholinesterase inhibitors, the cognitive and functional impairments progress rapidly, caregiver burden remains high, and new treatments are needed for these patients. With respect to the motor component of DLB, dopaminergic medications (e.g., carbidopa/levodopa) work less well in DLB as compared to PD and patients with DLB generally have a limited response to these medications, which are in any case poorly tolerated in this patient population; a reason for the poor response is that DLB is primarily a disease of the cholinergic system, rather than the dopaminergic system.
Scientific Rationale
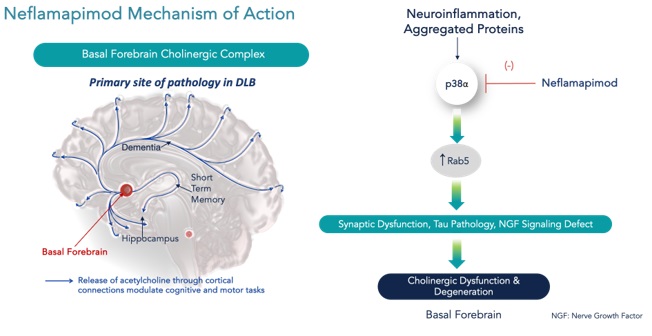
Recent evidence indicates that the primary pathology in DLB is in the basal forebrain cholinergic system, dysfunction and degeneration of which drives neurodegeneration in other regions of the brain. A series of publications, largely from the laboratories and colleagues of Prof. William Mobley at UCSD and Prof. Ralph A. Nixon at NYU Langone and the Nathan Kline Psychiatric Institute, have defined the molecular mechanisms that lead to neurodegeneration of cholinergic neurons. As shown in the figure below, the cholinergic degeneration is believed to result from inflammation and various aggregated proteins that lead to aberrant activation of the protein Rab5, a master regulator of endocytosis and endosomal trafficking, further leading to impaired retrograde axonal transport and a block in NGF signaling from the synapses at the ends of nerve fibers (or “axons”) back to cell body of the cholinergic neuron in the basal forebrain. The resulting loss of support of neuronal health that NGF provides is then believed to lead to dysfunction, and, eventually, degeneration of cholinergic neurons, which are particularly vulnerable to this pathogenic process because of their very long fibers.
Molecular Mechanisms Underlying Cholinergic Neurodegeneration in DLB and Point of Intervention for Neflamapimod
Early-stage patients with pure DLB (i.e., the ~50% of patients without AD-related co-pathology assessed by biomarkers) have relatively limited neurodegeneration and neuronal loss in the cortical regions of the brains, including and particularly in the hippocampus. Moreover, based on a range of animal and human pathology studies, the cholinergic degenerative process in the basal forebrain is believed to be reversible. The cholinergic neurons in that region of the brain do not die, rather they stop functioning normally (i.e., stop producing acetylcholine) and atrophy, or shrink in size. However, as those neurons are still alive, with successful pharmacological treatment they can be rescued and the disease process reversed.
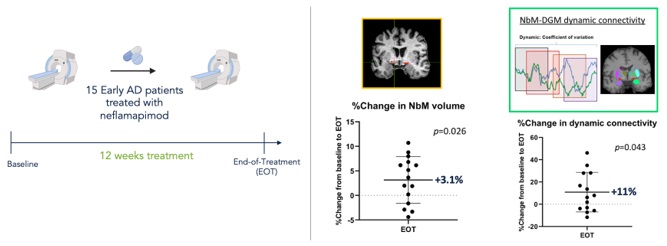
Neflamapimod was hypothesized to reduce Rab5 protein activity – a key therapeutic target in this pathogenic model for cholinergic degeneration in DLB – because of scientific literature showing that the immediate target of neflamapimod, p38α kinase, is the major activator of Rab5. Based on that hypothesis, neflamapimod was evaluated in a preclinical study in an animal model intended to evaluate neflamapimod’s effects on basal forebrain cholinergic atrophy and, later, in our Phase 2a AscenD-LB Trial in patients with DLB. We believe that the results of these studies, through demonstration of reduction in Rab5 activity and reversal cholinergic dysfunction & degeneration, demonstrate neflamapimod’s potential to treat synaptic dysfunction, the reversible aspect of the underlying neurodegenerative processes in the basal forebrain cholinergic system that cause disease in DLB. We also have obtained and published results from a pilot clinical study in patients with early AD that demonstrate neflamapimod treatment increases the volume of the basal forebrain, as well its functional connectivity to the cortex, as assessed by structural and functional MRI, respectively.
Clinical Development Plan
AscenD-LB Trial: Our Completed Phase 2a Trial in Dementia with Lewy Bodies
The AscenD-LB Trial was a Phase 2a double-blind, placebo-controlled, 16-week treatment, exploratory clinical trial of neflamapimod in mild-to-moderate DLB conducted at 22 centers in the United States and two centers in the Netherlands. 91 subjects were enrolled between October 2019 and March 2020 and randomized to receive 40 mg neflamapimod capsules or matching placebo capsules (randomized 1:1) for 16 weeks. The dosing regimen was based on weight, with trial participants weighing less than 80 kg receiving capsules BID and those weighing greater than or equal to 80 kg receiving capsules TID. All subjects had to have already been receiving oral cholinesterase inhibitor therapy for at least three months (stable dose for greater than six weeks) and continued such therapy without dose modification during the trial.
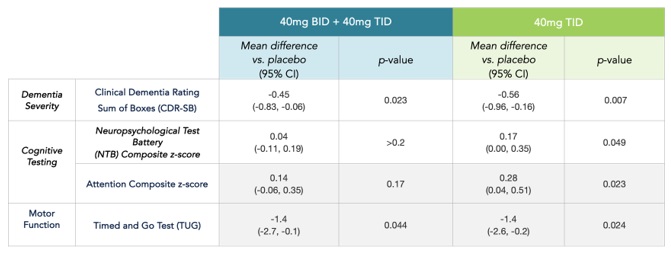
The AscenD-LB Trial was an exploratory clinical trial designed to evaluate the effects of neflamapimod against a range of clinical endpoints. In the primary analysis of the AscenD-LB Trial, which included all patients enrolled and evaluated for treatment effects, neflamapimod demonstrated improvement compared to placebo in dementia severity (assessed by CDR-SB, p=0.023 vs. placebo) and functional mobility (gait or walking ability as assessed by the TUG test, p=0.044 vs. placebo). In additional analyses, at the highest dose (40mg TID), significant improvement on a cognitive test battery, or NTB, was evident as compared to placebo (p=0.049); however, significant improvement compared to placebo on the NTB was not evident in the primary analysis. In addition, encouraging positive trends on the ten-item Neuropsychiatric Inventory were seen, particularly with respect to visual hallucinations, where a significant reduction in frequency relative to placebo was seen.
This primary analysis of the AscenD-LB Trial data showing neflamapimod significantly improved dementia severity and motor function was published in the major scientific journal Nature Communications in September 2022.
Primary Analysis of Major Efficacy Endpoints in AscenD-LB Trial of Neflamapimod in DLB
On-study (all time-points) results; change from baseline analysis utilizing Mixed Model for Repeated Measures. Number of participants: 41 for placebo, 20 each for 40mg BID and 40mg TID.
We believe the lack of significant effect in the primary analysis on the cognitive testing (NTB) results are attributable to the combination of (1) the inclusion of subjects receiving the lower, 40 mg BID dose of neflamapimod, a dose that did not achieve targeted therapeutic blood drug concentrations, and (2) “ceiling effects”, (i.e. that patients with disease have exogenous limits on how much they can improve on a cognitive test) resulting from two separate potential causes. First, all patients in the study were receiving cholinesterase inhibitor therapy, which is known to improve outcomes on cognitive testing in patients with DLB; that is, with having received benefit with cholinesterase inhibitor therapy, there was a limit to how much better perform with neflamapimod treatment, particularly with low dose neflamapimod treatment. Second, the deficits in executive function at baseline were very mild and, as a result, the tests evaluating executive function (two of six in the NTB) could not have demonstrated an effect.
Based on recent scientific literature demonstrating that DLB subjects with abnormally elevated plasma ptau181 (tau protein phosphorylated at residue 181) have AD associated co-pathology (specifically amyloid plaque and/or tau pathology by PET scan or CSF analysis), additional pre-specified analyses of the AscenD-LB data stratified by baseline plasma ptau181 were conducted and identified the pure DLB patient population as the optimal patient population for the RewinD-LB Trial and any future phase 3 clinical trials. Compared to subjects with DLB without elevated plasma ptau181 (i.e., with “pure” DLB), subjects with DLB with elevated plasma ptau181 have more extensive neuronal loss (neurodegeneration) and, therefore, would be expected to be less responsive to treatment. As shown in the table below, patients in the AscenD-LB Trial with pure DLB had an average higher treatment response (evaluated by Cohen’s d effect size), compared to the average response in the overall study, and demonstrated significant improvement in cognitive tests of Attention, the CDR-SB, the TUG test, and in a rest of recognition memory (International Shopping List Test recognition index) with Cohen’s d treatment effect size that was greater than 0.7 for each of these endpoints, indicating clinical effects that are moderate-to-large in magnitude. By comparison, in published studies in the scientific literature, the cholinesterase inhibitors have Cohen’s d effect size of approximately 0.3 in the treatment of AD or DLB.
Magnitude of 40mg TID Neflamapimod Treatment Effect vs. Placebo in Overall Patient Population and in the Pure DLB Patient Population) of the AscenD-LB Trial*
* By convention the magnitude of a treatment is considered small when the Cohen’s d effect size between 0.2 and, moderate when it is 0.4 to 0.8 and large when it is 0.8 or greater.
In September 2023, the results of these additional analyses of the AscenD-LB Trial were published in Neurology, the medical journal of the American Academy of Neurology. A subsequent publication in Molecular Neurodegeneration provides a combined evaluation of the findings in the Neurology and Nature Communications articles that makes the case for advancing neflamapimod as a treatment for DLB.
RewinD-LB Trial: Our Ongoing Phase 2b Trial in Dementia with Lewy Bodies
In the second quarter of 2023, we initiated our ongoing RewinD-LB Trial, a Phase 2b clinical trial of neflamapimod in subjects with DLB funded by a $21.0 million grant from the NIA, and, in August 2023, we announced dosing of the first patient in the study. We believe the design of the RewinD-LB Trial has positioned the study for success, as it is based on our findings and learnings from the AscenD-LB Trial, including the following:
| ● | Based on the dose response analysis of the AscenD-LB Trial and observations in prior AD studies, the optimal dose was identified as 40 mg TID, which will be the only dosing regimen used in the RewinD-LB Trial. |
| ● | Clinical endpoints that can detect effects on both cognitive and motor function (specifically, CDR-SB and TUG) better distinguish drug treatment from placebo than tests that are purely focused on evaluating cognition. Moreover, in AD, CDR-SB is accepted by regulatory authorities as an approval endpoint. Accordingly, we have chosen CDR-SB as the primary endpoint in the RewinD-LB Trial. |
| ● | Subjects with pure DLB (i.e., those without AD co-pathology as evidenced by increased concentrations of ptau181) appear to have a greater response to treatment. Therefore, we have chosen to exclude subjects with elevated (i.e., abnormal) levels of ptau181 in the RewinD-LB Trial. We believe that excluding subjects with abnormal ptau181 substantially increases the statistical power to demonstrate treatment effects in clinical trials of neflamapimod in DLB. |
Accordingly, in the RewinD-LB Trial, neflamapimod will be administered orally, 40 mg TID, with a second group receiving matching placebo. Each treatment group will include 80 subjects (enrolling a total of 160 subjects) diagnosed with DLB by consensus criteria, including having an abnormal dopamine transporter scan. Subjects with elevated plasma ptau181 (i.e., having evidence of AD co-pathology) will be excluded. Treatments (neflamapimod or placebo) will be administered for 16 weeks in the main trial (i.e., double-blind, placebo-controlled portion of the study), with a 36-week open label treatment extension for subjects completing the initial 16-weeks of the trial. Following completion of informed consent procedures, subjects will enter the screening phase of the trial. Once eligibility is confirmed and before the first dose of study drug, subjects will be randomly assigned on 1:1 basis to placebo or neflamapimod treatment. Dosing will start on day 1 following completion of all baseline procedures. During the placebo-controlled portion of the trial, subjects will return to the clinic at the end of weeks 2, 4, 8, 12 and 16.
The primary objective of the trial is to demonstrate that neflamapimod, compared with placebo, improves dementia severity, as assessed by change from baseline to week 16 in CDR-SB score. The CDR-SB is designed to assess both cognition and function, and is obtained by clinicians rating the severity of symptoms across 6 domains – memory, orientation, judgment & problem solving, community affairs, home & hobbies, and personal care – after a semi-structured interview with the patient and a reliable informant (e.g. family member) on a 0–3 scale for each domain (total range 0–18, with a higher score indicating worse dementia).

Secondary objectives include further evaluation of the safety and tolerability of neflamapimod and treatment effects on (1) cognition, assessed by a DLB-specific cognitive test battery, (2) motor function, as assessed by the TUG test, and (3) global rating of treatment effect, assessed by the CGIC. Tertiary endpoints will examine whether neflamapimod affects neuropsychiatric outcomes as assessed by the NPI-12, effect on fluctuations in cognition as assessed by the Dementia Cognitive Fluctuations Scale, impact on resting-state EEG (as well alpha-reactivity evaluated by EEG) and in a sub-set of subjects, basal forebrain atrophy assessed by structural MRI.
Sample size was calculated via simulations conducted utilizing the data in the Phase 2a study for the major clinical endpoints in the neflamapimod 40mg TID and placebo groups, generating for each patient a change from baseline for each endpoint at individual visits over the course of the simulated clinical study, and then analyzing the result using the linear mixed effects model for repeated measures that will be utilized to analyze the Phase 2b study. Based on the simulation of 100 clinical trials with 80 patients per treatment group, and assuming a 10% dropout rate, the RewinD-LB Trial has approximately 85% power with the NTB, 95% power with TUG, and greater than 95% power (approaching 100%) with the primary endpoint, CDR-SB, to detect a treatment effect at a significance level of 0.05.
We expect to complete enrollment in the RewinD-LB Trial during the second quarter of 2024 and to report initial results from the placebo-controlled portion of the study during the fourth quarter of 2024. The results of the RewinD-LB Trial are intended to provide the data necessary to finalize our design of a Phase 3 clinical trial, the general framework of which has been agreed upon with the FDA.
Planned Phase 3 Development in DLB Based on Success in Phase 2b Clinical Trial
We met with the FDA in January 2020, after completion of the AscenD-LB Trial and availability of the preliminary analysis of the results, in an end-of-phase 2 meeting to discuss potential Phase 3 clinical designs that may support approval of neflamapimod for the treatment of hypoxiaDLB. In that meeting, the FDA stated that a single Phase 3 clinical trial of six months’ treatment duration may be sufficient to support approval of neflamapimod if the trial demonstrated robust, clinically meaningful effects on cognition and on either function or a global measure (e.g., CGIC). Based on those discussions, we believe that if the RewinD-LB Trial demonstrates significant effects on the primary CDR-SB endpoint (a clinically meaningful measure of cognition and function), the result would be highly predictive of success in Phase 3, as the Phase 3 clinical trial would be designed to replicate the Phase 2b findings over six months (an additional two months compared to the four months in Phase 2b). Further, the number of subjects to be enrolled in a Phase 3 trial, which at the time of the January 2020 meeting was proposed to be 250 subjects, would be adjusted based on treatment effect size observed in the Phase 2b results to provide >95% statistical power for the primary efficacy endpoint. We are also evaluating CGIC in our planned Phase 2b trial for incorporation as a platformpotential endpoint in the Phase 3 clinical trial. The size of a Phase 3 clinical trial and certain other aspects of the Phase 3 trial (e.g., choice of secondary endpoints) would be discussed with the FDA in a second end-of-phase 2 meeting that we would expect to enhance standard-of-care treatment for conditions complicated by hypoxia. Prior to 2020, these efforts generated a substantial amount of data related to TSC, including data related to chemistry, manufacturing, and controls, preclinical safety and/orschedule after the primary efficacy data in a wide array of experimental models, single dose safety, tolerability, and pharmacokinetic data, and clinical data and other information related to TSC's potential as a supplement to standard-of-care treatment in GBM, peripheral artery disease, and stroke.
In March 2020,are available from the COVID-19 pandemic acceleratedongoing RewinD-LB Trial, which we anticipate being available in the U.S. resulting in numerous delays and other challenges for biopharmaceutical companies conducting clinical studies in indications not related to COVID-19, including our TSC Stroke Trial. In early April 2020, we announced plans to initiate a clinical research program to evaluate TSC in patients with COVID-19 due to the anticipated persistence of the COVID-19 pandemic coupled with our belief in the potential of TSC to improve low tissue oxygen levels, a common symptom and complicating factor in the treatment of COVID-19. We believe this decision was both opportunistic and a natural extension of the previous development work conducted with TSC, and in September 2020 we commenced the TSC COVID Trial.
In November 2020, we announced that, following changes to our management team in the prior two months, we had initiated a thorough review of our then-existing development program for TSC and commenced plans to modify the program with the intent of accomplishing two principal strategic objectives:
• To optimize the clinical dose and dosing frequency for TSC.
• To evaluate TSC in clinical models designed to establish proof of concept for improvement in oxygenation.
The completion of the TSC COVID Trial in February 2021 provided the first multiple daily dose safety and tolerability data for TSC. We expect corresponding pharmacokinetic and other secondary endpoint data from this trial to be available early in the secondfourth quarter of 2021. Obtaining these data represents the first, major step in our ongoing efforts to implement our modified development strategy, intended to strategically gap-fill, supplement, and expand on our past development work in TSC, with the ultimate goal of maximizing the probability of regulatory approval and, if TSC is approved, commercial success.
During the next twelve months, we intend to continue to execute our strategy and accomplish the following principal, strategic objectives, all of which we expect to be able to fund with cash-on-hand:
• Complete the TSC Oxygenation Trials – As described in more detail under the heading, — Our Lead Product Candidate: Trans Sodium Crocetinate — Our Anticipated Next Steps in the TSC Development Program: The TSC Oxygenation Trials, the next phase in our development plan is to evaluate TSC's effects in clinical models of oxygenation in an effort to establish a dose-response relationship. We expect to commence the TSC TCOM Trial before the end of March 2021 with data expected in the second quarter of 2021, and we intend to commence both the TSC DLCO Trial and TSC Induced Hypoxia Trial in the third quarter of 2021 with data expected from each study expected within two months of their respective completion.2024.
• Initiate NIA Grant
In January 2023, we were awarded a $21.0 million grant from the NIA that is estimated to fully fund development costs associated with the RewinD-LB Trial. The NIA Grant funds will be disbursed over the course of the trial as costs are incurred and, during the year ended December 31, 2023, we received total cash funding of approximately $6.2 million.
In addition, in December 2023, we submitted a request for supplemental funds in the amount of $4.0 million, of which, if approved, $3.9 million would be received in the current year and the remainder would be received in next the funding year. The request for supplemental funds was initially reviewed by the NIA in January 2024 but, due to the NIA currently working under the Continuing Resolution, completion of the review was delayed and the request is currently scheduled to be reviewed for approval in May 2024.
We currently expect to receive the remaining 10%, or $0.8 million, of the previously approved year 2 funding upon U.S. congressional approval of a final appropriations bill, the supplemental amount of $4.0 million following NIA review of our supplement request, and the year 3 funding of $6.2 million in February 2025.
Prior Clinical Studies of Neflamapimod
Phase 2 TrialClinical Trials Evaluating Neflamapimod in Alzheimer’s Disease
Prior to our more recent clinical trials in patients with DLB, two Phase 2a studies of TSCneflamapimod in Hypoxia-related IndicationAD were completed in early 2017. Results from these earlier studies demonstrated that neflamapimod is well tolerated, crosses the blood brain barrier and is pharmacologically active in the brain, including providing us with data around blood-barrier penetration target engagement (biological activity in the brain), and an understanding of dose-response, i.e., the completion of the steps in early clinical studies to successful CNS drug development.
One of these studies, Reverse-SD, was a Phase 2b clinical trial in subjects with AD. 161 subjects were enrolled at 38 sites in the Czech Republic (5 sites), Denmark (3 sites), Netherlands (3 sites), United Kingdom (11 sites) and United States (16 sites) and were randomized 1:1 to receive neflamapimod 40 mg capsules or matching placebo capsules twice daily with food for 24 weeks. Inclusion criteria were as follows: men and women aged 55 to 85 years, with CDR-Global score of 0.5 or 1.0 (i.e., with mild AD); CDR memory sub-score of at least 0.5; MMSE score of 20 to 28, inclusive; positive biomarker for AD, as defined by CSF Aβ1-42 <1000 pg/mL and phospho-tau/Aβ1-42 >0.024 in the Roche Eclesys® immunoassay; receiving either no AD-specific therapy or on a stable dose monotherapy (either cholinesterase inhibitor or memantine; dual therapy excluded).
Including all subjects in the analysis, there was no evident difference between the neflamapimod and placebo groups in the primary clinical efficacy endpoint, the combined change from baseline to week 24 in the z-scores of HVLT of Total Recall and Delayed Recall. However, in the analysis of CSF biomarkers, there were statistically significant effects of neflamapimod treatment, with a reduction relative to placebo, in the change from baseline to week 24 in CSF protein levels of phosphorylated tau (p-tau181, p=0.01 vs. placebo) and total tau (p=0.03 vs. placebo), and a trend on CSF neurogranin (p=0.07 vs. placebo).
Because in the scientific literature tau pathology has been shown to be downstream (is a consequence) of p38α kinase activity, the effect of neflamapimod on CSF levels of ptau181 and total tau demonstrates target engagement, i.e., these CSF results are consistent with “target engagement” within the brains of subjects. Target engagement is the industry term for the drug having the intended pharmacological effect in humans that would be expected based on its mechanism of action; in this case, that neflamapimod is inhibiting p38α activity. Furthermore, as CSF ptau181 and CSF total tau are considered to reflect neurodegeneration and synaptic dysfunction, respectively, we believe the results also provide objective evidence of neflamapimod impacting the neurodegenerative process in patients, including specifically on synaptic dysfunction.
As a single dose of neflamapimod was utilized in the trial, pre-specified pharmacokinetic pharmacodynamic analyses were conducted to evaluate the results for potential dose-dependency. These analyses showed improvement, relative to the placebo group, in tests of episodic memory in neflamapimod-treated subjects with the highest (top quartile) trough plasma drug concentrations; with positive trends evident both for the primary endpoint (combined change in z-scores of HVLT total recall and delayed recall) and the major secondary endpoint of change in Wechsler Memory Scale Combined Immediate and Delayed Recall composites. This analysis provided critical dose-response information as it indicated that 40mg BID was too low a dose, but that a dose of 40mg TID would achieve therapeutically effective drug concentration levels in the blood.
Results of Imaging of Basal Forebrain by MRI in Patients with Early AD after Treatment with Neflamapimod –
With the development and availability of analytic MRI-based techniques to evaluate potential treatment effects on the basal forebrain, the MRI images from patients with mild AD (n=15) from one of our Phase 2a studies were reanalyzed by a specialized neuroimaging group at the Amsterdam Medical Center. The goal of this exploratory analysis, which was presented at the AD/PD meeting in Gothenburg, Sweden in April 2023, was to assess by MRI the treatment effects of neflamapimod on the NbM, the largest cluster of cholinergic neurons in the basal forebrain. Structural and MRI assessments had been conducted as part of the study at baseline and following 12 weeks of treatment with neflamapimod. The additional analysis demonstrated that the NbM volume was statistically significantly higher at EOT (mean 3.1% higher vs. baseline, p=0.026). Eight of 15 subjects had greater than 3% NbM higher volume at EOT, as compared to baseline. Treatment with neflamapimod was also associated with a statistically significantly higher functional dynamic connectivity between the NbM and DGM at EOT (mean 11% higher vs. baseline, p=0.043), with six of 13 subjects showing a greater than 10% higher dynamic NbM-DGM connectivity at EOT, as compared to baseline. We believe the potential reversal of atrophy and recovery of function in neflamapimod-treated subjects in this trial suggests a restoration of cholinergic neurons in the NbM in line with the data generated in previous preclinical studies that demonstrated neflamapimod reversed the neurodegenerative process in the basal forebrain cholinergic system.
Neflamapimod treatment was associated with increased basal forebrain volume and functional connectivity
NbM – Nucleus basalis of Meynert, the largest cluster of cholinergic neurons in the basal forebrain; DGM – Deep Grey Matter
Lin C-P, Noteboom S, Bet M, Alam J, Prins N, Barkhof F, Jonkman L, Schoonheim M, Oral Presentation at AD/PD™ 2023, Gothenburg, Sweden, 1 April 2023
Clinical Safety Results
Adverse events seen in all completed Phase 2 clinical trials evaluating neflamapimod in both CNS and non-CNS disorders are shown in the table below. This includes 149 subjects with either AD or DLB who have received neflamapimod for up to 24 weeks at either 40 mg BID or TID or 125 mg BID. Among this cohort of patients with CNS disorders, the most commonly reported adverse events were headache (15 events, 10%), respiratory infection (11 events, 7%), diarrhea (11 events, 7%), fall, (11 events, 7%), and somnolence (seven events, 5%), all mild to moderate in severity. Headache, diarrhea, and somnolence appear to have the strongest association with neflamapimod treatment.
There were five Serious Adverse Events reported in the 149 subjects with AD and DLB treated with neflamapimod (vs. eight who were administered placebo), involving hypokalemia, myeloma, head injury, brain tumor, and brain lesion, none of which were considered related to neflamapimod.
Adverse Events in Neflamapimod Phase 2 Clinical Trials of ≥ 12 weeks duration in AD or DLB
| Placebo (N=128) | Neflamapimod (N=140) |
Falls | 8 (6%) | 11 (8%) |
Diarrhea | 7 (6%) | 10 (7%) |
Headache | 6 (5%) | 9 (6%) |
Common Cold/URI | 8 (6%) | 7 (5%) |
Nausea | 4 (3%) | 6 (4%) |
Somnolence | 3 (2%) | 4 (3%) |
Vomiting | 4 (4%) | 2 (1%) |
Fatigue | 5 (3%) | 1 (1%) |
With respect to liver enzyme abnormalities, during 12 weeks of dosing at 250mg BID (i.e., four-fold higher daily dosing than in the recently initiated Phase 2b trial) in 44 subjects with rheumatoid arthritis, elevations in liver transaminase levels were noted in six subjects (14%). Additionally, in one subject (1%) participating in the Reverse-SD 24-week trial in mild AD who received 40 mg BID neflamapimod, ALT and AST levels increased to three times the upper limit of normal. In each instance, subjects were asymptomatic, there were no associated increases in bilirubin, and the elevations resolved with treatment discontinuation.
In the most recently completed AscenD-LB trial involving 91 subjects with DLB, neflamapimod was well tolerated with no treatment discontinuations due to study drug-related adverse events. There were four SAEs reported in the placebo group (haematochezia, internal bleeding, intraparenchymal hemorrhage, asthma exacerbation) and two among the neflamapimod BID treatment group (brain lesions, head injury), all of which were considered unrelated to treatment. In addition, one SAE (brain tumor diagnosis) was reported 34 days after the last dose in a neflamapimod BID recipient. There were no SAEs or early treatment discontinuations in the neflamapimod TID recipients. Liver enzyme abnormalities were not observed in the AscenD-LB trial.
Preclinical Studies
Ts2 Transgenic Mice
Nearly all individuals who have Down Syndrome, characterized by trisomic chromosome 21, develop AD by their fourth decade of life, and have typical AD pathology when autopsied at death. This may be explained by chromosome 21 containing the gene for amyloid-precursor-protein, which is the gene linked to familial or genetic early onset AD in humans. The Ts2 transgenic mouse model of Down Syndrome utilizes mice that are partially trisomic at chromosome 16, which is the mouse equivalent of chromosome 21. Along with developmental behavioral abnormalities, Ts2 mice develop typical early onset dementia pathology, including endosomal abnormalities and cholinergic neurodegeneration in the basal forebrain cholinergic system. Accordingly, Ts2 mice provide an ideal opportunity to evaluate the effects of drug treatment on basal forebrain cholinergic dysfunction and degeneration.
To evaluate the potential of neflamapimod on the neurodegenerative process, the effects of neflamapimod were evaluated in Ts2 mice. Wild-type mice, referred to as either WT or 2N, and Ts2 mice were treated over 28 days, twice daily, with either vehicle or 3 mg/kg of neflamapimod in vehicle, with nine mice in each group. Treatment was initiated at 6-7 months of age, representing a time point at which endosomal pathology and cholinergic neuronal loss is developing. To assess for effects on cholinergic neurodegeneration, ChAT+ neurons were quantitated in the region of the forebrain that is enriched for cholinergic neurons, which is known as the MSN.
At the end of treatment, consistent with current scientific literature, the number of cholinergic neurons in the MSN region was significantly decreased in vehicle-treated TS2 mice compared to vehicle-treated WT mice (p<0.001). This effect was reversed with neflamapimod treatment, with the number cholinergic neurons in the MSN increased in neflamapimod-treated TS2 mice compared to vehicle-treated TS2 mice, and the number of ChAT+ neurons were similar to those seen in WT mice (p<0.001). Neflamapimod treatment also normalized Rab5 activity and phosphorylated (i.e., activated) p38 MAP kinase and its downstream substrates.
Neflamapimod restores numbers of cholinergic neurons in basal forebrain (i.e., reverses disease progression) in Ts2 transgenic mouse.
Cholinergic neurons, as assessed by staining positive for ChAT+ in the MSN of the basal forebrain, in wild-type treated with vehicle or Ts2 transgenic mice after treatment for four weeks with either vehicle or neflamapimod.
The finding of reversal of disease progression is consistent with studies in the scientific literature that suggest that “loss” of cholinergic neurons in the basal forebrain cholinergic system is not due to cell death. Rather, the “degeneration” and loss of such basal forebrain cholinergic neurons appears to be due to a loss of cholinergic phenotype and functional properties, and neuronal shrinkage, all of which in animal studies can be reversed. That is, the effect of reversing disease progression, evidenced by increased number of cholinergic neurons. This is not a regenerative effect. Rather, we believe it reflects that treatment with neflamapimod is restoring the function of diseased neurons (those that don’t express ChAT), allowing them to express ChAT. There is also evidence from studies in early AD, that cholinergic phenotype loss, rather than frank neuronal death and loss, occurs in the basal forebrain of humans as well. We believe this is consistent with the results obtained from the MRI evaluation of neflamapimod-treated AD patients discussed above in whom an increase in the volume of basal forebrain cholinergic neurons was observed in the NbM.
Aged Rat Model
To obtain preclinical proof-of-principle and confirm the role of p38α in the development of synaptic dysfunction, we tested neflamapimod in a rat model of age-related cognitive decline. When evaluated in the Morris-Water-Maze test of spatial learning, rats show cognitive deficits starting at 20 to 22 months of age, which is equivalent to approximately 60 years of age in humans. Of note, because the deficits in Morris-Water-Maze performance can be fully reversed by implanting healthy cholinergic neurons in the basal forebrain, those deficits are believed to be due to basal forebrain cholinergic dysfunction and degeneration.
The results of these tests showed that treatment with neflamapimod fully reversed the learning deficits in the Morris-Water-Maze test in 20- to 22-month-old rats. Specifically, the performance of aged rats on the last day of testing (day 17) showed that animals treated with neflamapimod at the optimal dose performed significantly better than vehicle–treated aged rats (p=0.007 for latency; p=0.01 for distance). Further, the performance of neflamapimod-treated aged rats was similar to that of young rats (i.e., fully reversed cognitive deficits). The figure below further details the results of these tests, in which two groups of 15 rats each (aged rats with cognitive deficits and a control group of young rats) received vehicle or active drug treatment for 21 days. The Morris-Water-Maze test was conducted on days 4-8 and days 11-17.
Neflamapimod’s Potential in Additional Indications
Acute Indication: Recovery after Ischemic Stroke
We believe the therapeutic benefit of targeting neuroinflammation-induced synaptic dysfunction is not limited to chronic neurodegenerative diseases. A drug that improves synaptic function could also be considered for evaluation of the potential to improve brain function after acute neurological injury. In the future, we may investigate neflamapimod in the treatment of certain acute indications such as ischemia-induced stroke. We have generated preclinical evidence suggesting that neflamapimod could improve recovery after ischemic stroke in an animal model.
A treatment to improve recovery from stroke remains a significant unmet medical need. Every year, more than 795,000 people in the United States suffer a stroke, and approximately 610,000 of these are first or new strokes. About 87% of all strokes are ischemic strokes, in which blood flow to the brain is blocked. The prognosis for recovery from stroke is influenced by a number of different factors, including stroke severity, type of stroke, location of infarct, co-morbidity with other disorders, and other clinical complications. The majority of survivors of an acute stroke demonstrate some level of neurological recovery during the three to six months after the initial event. Despite this initial period of recovery, 40 to 50% of patients exhibit persistent neurological deficits.
During the last 10 years, the medical and scientific communities have gained a better understanding of the mechanisms underlying neuronal recovery following a stroke. The major translational opportunity for therapeutics that target recovery after stroke is the time window in which intervention must be initiated. Rather than just the first few hours after the stroke (as is the case with neuroprotection, i.e., acute stroke therapy to reduce the size of stroke), the window for therapeutics that could improve recovery is days and even weeks after an acute stroke. Waiting to initiate therapy until 48 hours after the stroke allows inclusion of a homogenous patient population as the diagnosis and extent of the stroke can be definitively established by that time in most patients (the exception being the minority who have a “stuttering” stroke). As a result, a POC study in stroke recovery is in the range 50-100 patients per treatment arm, compared to 500+ per treatment arm in neuroprotection trials.
The scientific rationale for evaluating neflamapimod to promote recovery after stroke is that the basal forebrain cholinergic system plays a critical role in recovery after ischemic stroke, particularly motor function recovery. The BFC system is suppressed by residual inflammation in the weeks and months after the acute stroke event. Neflamapimod, through the same mechanisms operating in DLB, would be expected to reverse the suppression of BFC function, leading to improved recovery of motor function. Supporting that concept is our preclinical data with neflamapimod demonstrating significant improvement in neurological recovery vs. vehicle treatment, and TUG results from the AscenD-LB clinical trial where positive effects of neflamapimod on basal forebrain mediated control of movement were observed in the clinic.
In a preclinical study of neflamapimod that evaluated effects on recovery after stroke, which has been published in a peer-reviewed scientific journal, transient ischemia of sufficient duration was induced in rats such that significant neurologic disability developed without mortality, and the neurologic disability did not substantially reverse during follow-up without therapy. These rats were then treated with either vehicle or one of two different doses of neflamapimod. The three groups in the study were: vehicle control (n =18), 1.5 mg/kg neflamapimod (n = 21) and 4.5 mg/kg neflamapimod (n = 21). Six weeks of neflamapimod treatment, starting at 48-hours after stroke, led to substantial improvement on multiple parameters of neurologic function compared to vehicle controls (p<0.001 for each of global neurologic scores; motor and sensory specific tests).
We have no immediate plans to initiate a clinical trial evaluating neflamapimod as a treatment to improve recovery from acute stroke. However, we have had extensive discussions with stroke experts and have designed a 120-patient, 12-week treatment, placebo-controlled Phase 2 POC trial to improve recovery after ischemic trial in which treatment would be initiated between 3 and 7 days after the acute stroke event that could be initiated to evaluate the effects of neflamapimod, subject to available funding.
Early-Stage Sporadic Alzheimer’s Disease
The defining clinical characteristics of early-stage, sporadic AD are deficits in episodic memory (the recollection of everyday events). The driving pathology of sporadic AD is in the hippocampus, the part of the brain in which episodic memory is formed. Accordingly, the amyloid beta therapies have been developed as a treatment for AD based on preclinical data demonstrating that amyloid beta has deleterious effects on synaptic function in the hippocampus. However, scientific literature indicates that degeneration of the basal forebrain cholinergic system also contributes to disease expression and progression in AD, particularly in the early stages, and we believe that a reason for the limited success of amyloid beta directed therapies is that they do not impact disease progression in these basal forebrain cholinergic neurons. In addition to the effects on the BFC system, in experimental studies, p38α expression increased amyloid beta production, while reducing p38α activity decreased amyloid pathology. Further, neflamapimod treatment of transgenic AD mice reduced amyloid beta levels and, in Ts2 mice, neflamapimod reduced the expression of the major enzyme (beta secretase) that produces amyloid beta. Based on these observations, we believe there is a strong rationale for neflamapimod, either as a standalone therapy or in combination with amyloid beta directed therapies.
In addition to the mechanistic and pre-clinical evidence of the potential use of neflamapimod in AD, the AscenD-LB Trial demonstrated clinical outcome results and biomarker results in the CSF and on basal forebrain volume in patients with early AD that we believe suggest neflamapimod’s potential use in treating AD.
We have no current plans to initiate a clinical trial evaluating neflamapimod for treatment of early-stage sporadic AD. Rather, assuming success in our ongoing RewinD-LB Trial, we would likely pursue clinical development in early-stage sporadic AD in parallel with our Phase 3 development of neflamapimod in pure DLB, subject to available funding.
Frontotemporal Dementia
FTD is a neurodegenerative disorder characterized by progressive deterioration in behavior, personality, and language abilities, typically affecting individuals between the ages of 40 and 65 including an estimated 50,000 to 60,000 individuals in the U.S. alone. Unlike AD, which primarily targets memory, FTD primarily affects the frontal and temporal lobes of the brain, leading to changes in social conduct, emotional regulation, and decision-making. There are several subtypes of FTD, including the behavioral variant FTD, the most common subtype (approximately half the patients with FTD) and primary progressive aphasia, or PPA, each presenting with distinct symptom profiles. PPA, a subtype of FTD itself, has three main variants: nonfluent/agrammatic variant PPA, semantic variant PPA, and logopenic variant PPA. The prevalence of these PPA subtypes varies, with approximately 40% of PPA patients being nonfluent/agrammatic variant PPA, 40% being semantic variant PPA, and 20% being logopenic variant PPA. As the disease progresses, individuals with FTD may require increasing levels of care and support, with management focusing on alleviating symptoms and maximizing function.
The rationale for potentially evaluating neflamapimod as a treatment for FTD is based on the atrophy of the BFC system also being a driver of disease and the mechanisms that neflamapimod targets (e.g. defects in axonal transport) being operative in FTD. Specifically, when assessed by MRI, the volume of the basal forebrain is reduced, relative to age-matched healthy control, most prominently in patients who semantic variant PPA and behavioral variant FTD and in patients who have “tauopathies” (i.e., patients at autopsy who have tau pathology, rather than TDP-43 pathology). Moreover, in March 2024, at the AD/PD 2024 scientific conference in Lisbon, Portugal, academic collaborators from University College London presented data that showed that p38 MAPK inhibitors generally, and neflamapimod specifically, enhanced axonal transport in a transgenic mouse model of FTD (rg4510 transgenice harboring P301L mutation). Based, in particular, on the transgenic mouse results, we plan to initiate discussions with experts in field and design a phase 2a study to evaluate neflamapimod in the most appropriate subtype of FTD for the mechanism, subject to available funding.
Additional Neflamapimod Development Background
Discovery and Early Development by Vertex
Neflamapimod was originally discovered at Vertex, which initiated clinical investigations in 1999 to determine the effects of the drug on RA. During its clinical investigations of neflamapimod, Vertex completed single and multi-dose Phase 1 studies and initiated Phase 2a development in rheumatoid arthritis. A total of approximately 150 healthy volunteers and patients received neflamapimod in Vertex-sponsored studies for up to one month at 750 mg twice daily and up to 3 months at a dose of 250 mg twice daily.
In a Phase 2a trial in active rheumatoid arthritis conducted by Vertex, a total of 59 healthy volunteers and patients (44 on active drug of 250 mg, and 15 on placebo, twice daily) were enrolled in a 12-week treatment. In this trial, a statistically significant effect of neflamapimod administration on ACR20 response rate was demonstrated (p = 0.027 in the primary endpoint analysis: area-under-the-curve of ACR20 response over the 12-week trial period). In a pharmacokinetic/pharmacodynamic analysis, neflamapimod administration also reduced C-reactive protein and IL-6 levels with increasing cumulative drug exposure.
Neflamapimod was generally well tolerated in this RA Phase 2a trial. The most common adverse events associated with neflamapimod were abdominal pain (21% of the 44 healthy volunteers), diarrhea (18%), infection (16%), headache (14%), increased aspartate aminotransferase (14%) and increased alanine aminotransferase (11%). No treatment-emergent neurologic events were seen. Regarding liver function test abnormalities, transaminase levels returned to normal after treatment discontinuation and were not associated with bilirubin elevations. Liver enzyme elevations are a well-known dose-dependent clinical side effect of p38 MAPK inhibitors. In the case of neflamapimod however, we believe the threshold for inducing liver enzyme elevation is a dose level of 250 mg twice daily when administered for more than 4 weeks, which on a daily dose level is four-fold higher than the 40mg TID dose regiment we are moving forward in DLB and other CNS indications (500 mg per day in RA vs. 120 mg per day in DLB and other CNS indications).
Vertex ultimately discontinued its pursuit of neflamapimod in the early 2000s to focus on the clinical development of a therapy for rheumatoid arthritis with a different p38α inhibitor, which, unlike neflamapimod, does not enter the brain. Neflamapimod lay dormant with Vertex until we expressed our interest in exploring the drug for other indications. See “Vertex Agreement” below for additional information.
Toxicology
A full chronic repeated dose toxicology program has been completed in rodents (rats) and non-rodents (dogs). In the rodent species, in the six-month toxicology study, no human relevant findings were evident at dose levels that provided plasma neflamapimod drug concentration levels approximately ten-fold higher than those achieved in the AD clinical trials. In shorter-term studies, the primary target organ was the liver, with findings commencing at plasma drug concentration levels 20-fold higher than the AD clinical trial exposures. In the non-rodent species, in 9- and 12-month toxicology studies, dose dependent findings were evident beginning at plasma neflamapimod drug concentrations more than ten-fold higher than achieved with 40 mg twice daily in AD clinical trials, with minimal to equivocal findings at that dose level in the liver, bone marrow and CNS. The CNS findings demonstrated damage to axons, or nerve fibers, primarily in the spinal cord. p38α and p38β have been reported to have a role in transport of proteins in axons, and therefore we believe these toxicity findings are related to the inhibition of both p38α and p38β at the very high doses administered in the non-rodent studies. The doses we are using in our clinical trials are at least ten-fold lower than the doses at which these effects were observed.
Regulatory Status
We submitted an IND application to the FDA in February 2015. The FDA cleared our application in March 2015, and the IND remains open and active.
The FDA granted neflamapimod Fast Track designation for the treatment of DLB in October 2019.
Following a review of the long-term animal toxicology studies discussed above, the FDA placed a partial clinical hold on our first Phase 2a Trial in mild AD (Study 303) in August 2015, limiting administration of neflamapimod to doses that lead to plasma drug levels which provide at least a 10-fold safety margin to the plasma drug levels in animals that in long-term animal toxicity studies had previously led to minimal or equivocal findings in the liver, bone marrow and CNS. At the present time, this partial clinical hold effectively limits our clinical dosing in the United States to 40 mg of neflamapimod three times daily in patients with a weight of greater than or equal to 50kg (110 pounds), based on agreements with the FDA and on our current understanding of plasma drug levels achieved with neflamapimod in humans. As our current plans across our indications do not envision surpassing this dose level, we do not expect this partial clinical hold to impact our ongoing and planned clinical trials.
In Europe, clinical trial applications in support of our clinical trials have been reviewed and approved by the national regulatory authorities in each of the Netherlands, United Kingdom, Czech Republic and Denmark. In addition, the Agence Nationale de Sécurité du Médicament et des Produits de Santé (the French national regulatory authority) has reviewed and approved a clinical trial application for an investigator-initiated study of neflamapimod in Toulouse, France.
Vertex Agreement
In August 2012, based on our team’s previous direct experience with this compound and our understanding of its profile and emerging science around p38α in the brain, we entered into the Vertex Agreement, which granted us an option to acquire an exclusive worldwide license to develop and commercialize neflamapimod for the diagnosis, treatment and prevention of AD and other neurodegenerative diseases. In August 2014, we exercised that option to acquire the license to neflamapimod.
The Vertex Agreement contains certain milestone events and the related payments that we would be obligated to make to Vertex if and when such events occur. Each milestone payment is payable only once for each distinct licensed product, upon the first occurrence of the applicable milestone event. The first expected milestone events concern filing of an NDA, with the FDA for marketing approval of neflamapimod, in the U.S., or a similar filing for a non-U.S. major market, as specified in the Vertex Agreement. The Vertex Agreement also provides that we will make royalty payments to Vertex in the event aggregate net sales, as defined in the agreement, for a commercialized licensed product meet specified thresholds. Such royalties will be on a sliding scale of percentages of net sales in the low- to mid-teens, depending on the amount of net sales in the applicable years. We are also obligated to make a milestone payment to Vertex upon net sales reaching a certain specified amount in any 12-month period. The Vertex Agreement states that royalties will be reduced by 50% during any portion of the royalty term when there is no valid claim of an issued patent within specified patent rights covering the licensed product. We also have the right to deduct, on a country by country basis, from royalties otherwise payable to Vertex under the terms of the Vertex Agreement, 50% of all royalties, upfront fees, milestones and other payments paid by us or any of our affiliates or sublicensees to third parties under licenses that are necessary for the development, manufacture, sale or use of a licensed product, provided that in no event will the royalty payable to Vertex be reduced to less than 50% of the rates specified in the Vertex Agreement, subject to certain adjustments specified therein. In the aggregate, our potential milestone payment obligations, all of which relate to development milestones, under the Vertex Agreement are up to $122.0 million. To date, we have made an aggregate of $100,000 in payments to Vertex. In connection with our obligations under the Vertex Agreement, there is no minimum annual expenditure requirement. Our diligence obligations under the Vertex Agreement have included the making of annual expenditures in connection with the development of neflamapimod, commencement of a Phase 2 clinical trial of neflamapimod, and the commercial sale of neflamapimod within six months of market approval.
The Vertex Agreement provides that we may sublicense the rights granted to us by Vertex, in whole or in part, to a third party (through multiple levels of sublicensing) (i) who is providing services to us in connection with the manufacture or development of the licensed product, solely for the purpose of providing such services, or (ii) with the prior written consent of Vertex, which shall not be unreasonably withheld.
The license term under the Vertex Agreement is deemed to have commenced on August 21, 2014, and continues until the expiration of the royalty term, unless sooner terminated in accordance with the terms of the Vertex Agreement. The royalty term commences on the first commercial sale of a licensed product and ends upon the later of (i) the date of expiration, unenforceability or invalidation of the last valid claim of certain specified underlying patent rights, or (ii) ten years after the date of such first commercial sale. Upon the expiration of the royalty term, the license will convert to a perpetual, fully paid-up non-royalty bearing license with the same scope.
The Vertex Agreement may be terminated by us for any reason upon 90 days’ prior written notice to Vertex if such termination occurs before receipt of the first marketing approval of a licensed product, and otherwise upon twelve months’ prior written notice to Vertex. Either party may terminate the Vertex Agreement if the other party is in material breach of its obligations thereunder, following a 60-day notice and cure period, or if the other party files for bankruptcy, reorganization, liquidation, receivership, or an assignment of a substantial portion of assets to creditors. The Vertex Agreement also provides that in the event we materially breach any of certain specified diligence obligations as to a specific major market, Vertex’s sole remedy for such breach, following the applicable notice and cure period, will be to terminate the license as to such specific major market country.
EIP200 – Novel Co-Crystal of Neflamapimod
We have an issued patent, set to expire in 2038, in the United States for novel co-crystals of neflamapimod with identified, specific, Generally Recognized as Safe compounds that have the potential to improve the solubility and other physical properties of neflamapimod. The development of one of these co-crystals as a product would be supported by composition of matter protection afforded by this patent, providing additional patent protection if we developed such a co-crystal product ourselves, the opportunity to license such a product to another pharmaceutical company while retaining the rights to neflamapimod and other potential benefits. The ability to develop one or more of the three TSC Oxygenation Trials would provide definitive evidence of TSC's enhancement of oxygenation, whether through increased uptake in the lungs, enhanced delivery, increased utilization at the tissue level, or some combination thereof. Positive data from any of these studies, if obtained, will guide the subsequent steps of our development strategy focused on demonstrating the clinical and therapeutic benefits of TSC in relevant patient populations on the hypoxia continuum. In particular, if successful in one or moreco-crystal products requires a fuller evaluation of the three TSC Oxygenation Trials, we intendpotential manufacturing processes than has been performed to initiate a Phase 2, controlled, clinical outcome study evaluating TSC in one or more appropriate hypoxia-related indications, which we refer to as our Planned Phase 2 Hypoxia-related Indication Trial, in the first quarter of 2022.date.
• Continue to Improve Our Efficiency and Effectiveness – This will include evaluating, refining, and improving our policies, procedures, and processes related to chemistry, manufacturing, and controls, quality assurance, information technology, intellectual property, human capital, and other areas, including as described in more detail under the headings, — Products, Product Development, and Our Competition and — Our People and Human Capital Resources.
Representation of data supporting the potential use of TSC for the treatment of certain medical conditions complicated by hypoxia. Incidence numbers represent annual incidence in U.S. as reported by U.S. Centers for Disease Control and Prevention and other publicly available sources.
Our Lead Product Candidate: Trans Sodium Crocetinate
Our leadPrior to the Merger in August 2023, Diffusion focused on developing novel therapies that may enhance the body’s ability to deliver oxygen to areas where it is needed most. The most advanced of these product candidate,candidates, TSC, is beinghas been investigated and developed to enhance the diffusion of oxygen to tissues with low oxygen levels, also known as hypoxia. Hypoxia is associated withAlthough we have paused all development activity related to TSC, including the pathophysiologyinitiation of many acute and chronic conditions including cancers, heart attack, stroke, ARDS, skin and soft tissue diseases, neurodegenerative diseases and other intractable and difficult-to-treat conditions, further complicating treatmentDiffusion’s previously announced Phase 2 study of TSC in newly diagnosed GBM patients, we intend to continue to attempt to identify sale or out-licensing transactions for the underlying condition by medical providers.Company’s TSC-related assets.
Hypoxia: A Key Driver of Unmet Medical NeedsSales and Marketing
Oxygen is fuelWe do not currently have any infrastructure for life. The constant uptakethe sales, marketing or distribution of an approved drug product. In order to market and delivery of oxygen within our body is necessary to sustain life whether at restsuccessfully commercialize neflamapimod or under stress, in sickness and in health.
Normally, the body’s supply of oxygen sustains life by meeting the demand for oxygen at the cellular level. This availability of sufficient oxygen enables efficient and sustainable generation of energy to support the metabolic demands of the cells. Low oxygen content in the blood stream, also known as hypoxemia, often leads to hypoxia, a condition in which a region of the body's tissues is deprived of sufficient oxygen to produce the energy it needs to survive. This state can only be sustained for very brief periods of time, sometimes just minutes, before the body tissue experiences ischemia, damage, and, eventually, cellular death.
Under normal conditions, when oxygen is inhaled through the lungs it diffuses from this area of high oxygen concentration in the air sacs of the lungs, or alveoli, into pulmonary circulation where the oxygen concentration is lower. This diffusion process is passive. Oxygen moves across cell layers and vessel walls into the blood stream in a manner that does not require energy and avoids over-oxygenating the body. Once oxygen reaches the blood stream, it is rapidly taken up onto our red blood cells while a very small amount dissolves in the plasma. The hemoglobin within the red blood cells act as mass transport carriers for oxygen. Red blood cells are pumped throughout our circulatory system by the heart, reaching every cell in our body. Upon reaching its cellular destination, the oxygen is released from the hemoglobin and again passively diffuses from the area of high concentration in the bloodstreamany other future product candidate, to the lower concentration areasextent it or they are approved, we must either develop these capabilities internally or make arrangements with third parties to perform these services. We may also collaborate with strategic partners that have experience in the tissues.
Our body'sthese fields. There are significant expenses and risks involved in establishing our own sales, marketing and distribution functions, including our ability to deliverhire, retain and appropriately incentivize qualified individuals, generate sufficient oxygen in this manner is determined by several factors, includingsales leads, provide adequate training to sales and marketing personnel, and effectively manage a geographically dispersed sales and marketing team. Alternatively, to the amountextent that we depend on third parties for such services, any revenues we receive will depend upon the efforts of oxygen in the air we breathe, our cardiac output (i.e., how well the heart circulates the oxygen), our hemoglobin content (i.e., how many transportersthose third parties, and there are to carry the oxygen), and the level of oxygen saturation of hemoglobin, which can carry up to four oxygen molecules. A variety of medical interventions are available to augment or supplement these factors in the oxygen delivery process in order to increase mass oxygen delivery. For example, there are medications and mechanical pumps designed to improve cardiac output, blood transfusions to increase hemoglobin content in the blood, and other passive and mechanical methods to increase the systemic availability of oxygen. However, while these existing strategies can be very effective in certain circumstances, they do not specifically focus on enhancing the diffusion process by which oxygen moves from a high concentration area in the bloodstream to a lower concentration are in hypoxic tissue. Accordingly, we believe there remains a significant unmet need in the treatment of hypoxia.no assurance that such efforts will be successful.
An additional, important factor in oxygen uptake and delivery is how efficiently the oxygen moves or diffuses across the many impediments that can slow the movement of oxygen through the body, such as the red blood cell walls, water molecules in plasma, cellular layers of the blood vessels, and other tissues. Currently, there is no well-known or widely-used medical method to augment or supplement this factor in the oxygen delivery process.
TSC: A Novel Mechanism of Action and Demonstrated Safety Profile
We believe that TSC's novel mechanism of action would be the first therapy specifically designed to enhance the oxygen diffusion process, thereby supporting normal, physiologic levels of oxygen diffusion at the uptake and delivery points.
TSC's Novel Mechanism of Action
Blood plasma is approximately 90.0% water and these water molecules are constantly moving, bound together in a loosely organized matrix by hydrogen bonds. Due to oxygen’s passive diffusion process through the plasma from areas of high oxygen concentration to areas of low oxygen concentration, a treatment that can reduce the factors that resist this movement should make oxygen more available to hypoxic tissues.

Representation of oxygen molecules moving from red blood cells, across the concentration gradient, to oxygen-consuming tissues.
TSC was designed to enhance the level of organization among water molecules by increasing the amount of hydrogen bonding. This creates a less dense matrix of water molecules, reducing the resistance to oxygen diffusion across the concentration gradient. In animal models, this diffusion-enhancing mechanism of action has been observed to affect hypoxic tissue preferentially while avoiding excessive oxygen-related tissue toxicity, also known as hyperoxia.
Representation of the indirect course of oxygen molecule movement through “unorganized” or randomly moving water molecules (left panel) versus the more direct path through “organized” molecules allowed by TSC (right panel).
TSC's Clinical Safety Profile
TSC has been observed to be safe and well-tolerated at most doses tested in over 180 subjects included to-date in clinical studies. A maximum tolerated dose was identified in the initial clinical study of TSC conducted in normal healthy volunteers, and subsequent clinical studies have been conducted with lower doses. These studies have included patients with variety of medical conditions often complicated by hypoxia, including our clinical studies conducted in patients afflicted with GBM, peripheral artery disease with intermittent claudication, stroke, and, most recently, COVID-19. In each of these conditions and many others, hypoxia is a significant contributor to morbidity and mortality, and a considerable treatment obstacle for medical providers.
In the TSC COVID Trial, which we completed in February 2021, we evaluated the safety and tolerability of TSC administered on a more frequent dosing regimen than had been previously tested in a clinical trial setting. No dose-limiting toxicities or serious adverse events were observed among any patients in the TSC COVID Trial, including those who received the highest tested dose of TSC, 1.5 mg/kg administered intravenously every six hours for a period of at least five and up to 15 days.
TSC's Potential to Treat Hypoxia-Related Conditions
Hypoxia is a critical, complicating factor in many intractable and difficult-to-treat conditions affecting people of all ages, including cancers, cardiovascular disease, stroke, respiratory disease including ARDS, skin and soft tissue diseases, and neurodegenerative diseases. We have previously evaluated TSC in a variety of preclinical and clinical studies. We believe that the data we have obtained through certain preclinical studies provide support for TSC’s ability to improve oxygenation, while data obtained through the clinical trials we have undertaken to date suggest TSC is safe and well-tolerated at the doses tested, including administration of TSC to more than 180 human subjects and patients with GBM, acute stroke, COVID-19, acute lung injury, and peripheral artery disease with intermittent claudication.
Clinical and Preclinical Development of TSC to Date
Selected Preclinical Experiences
TSC has been evaluated in a variety of preclinical models intended to mimic relevant human conditions known to be complicated by hypoxia. In these studies, a number of positive effects have been observed in connection with TSC administration, including:
• Reducing hypoxia in rat brain tumors without hyper-oxygenation of normal tissue.
Positron emission tomography scans showing reduction in hypoxia in a rat C6 glioma brain tumor model 45 min after TSC administration.
• Improving survival in a rat brain tumor model when added to radiotherapy, whether or not combined with chemotherapy.
• Improving tissue oxygenation without hyper-oxygenation of normal tissue and reducing infarct size in a rat ischemic stroke model.
• Demonstrating a functional benefit in a rabbit ischemic stroke model, with or without tPA at one-hour post-clot infection and with tPA at three hours post-clot infection.
• Improving arterial PaO2 levels in a rat model of ARDS.
Glioblastoma Multiforme
We believe TSC may be able to benefit cancer patients by re-oxygenating hypoxic cancer tissue, making the cells more susceptible to the therapeutic effects of standard-of-care radiation therapy and chemotherapy. In particular, GBM is an especially deadly form of brain cancer that each year affects approximately 12,000 patients in the U.S. and approximately 35,000 patients worldwide, and for which TSC has received an Orphan Drug Designation from the FDA.
We previously completed a Phase 2 clinical trial that evaluated 59 patients with newly diagnosed GBM. This open-label, historically controlled trial demonstrated a favorable safety and tolerability profile for TSC when combined with standard of care treatment for GBM. Although not prospectively defined, a post hoc subgroup analysis of inoperable patients suggested a higher proportion of TSC-treated patients survived at two years compared to those in the historical control group.
Based upon data from the inoperable patient subgroup in the Phase 2 trial and guidance from the FDA received at our End-of-Phase-2 meeting, we initiated the TSC GBM Trial in the newly diagnosed inoperable GBM patient population in December 2017. The TSC GBM Trial was designed to enroll 236 patients in total, with 118 in the treatment arm and 118 in the control arm. The trial began with an FDA-mandated, open-label, dose-escalation safety run-in for which enrollment was completed and has closed. A total of 19 patients were enrolled in an attempt to ensure that at least 8 patients completed the FDA-specified 42-month exposure period. At a meeting in the third quarter of 2019, the data safety monitoring board for the trial concluded that no adverse safety signal was present and unanimously recommended the trial continue as planned, with TSC to be used in combination with temozolomide, an anti-cancer chemotherapy drug, during the adjuvant treatment chemotherapy period. However, we determined we did not have adequate resources at the time to fully support the randomized portion of the TSC GBM Trial and commencement of enrollment in the randomization portion of the TSC GBM Trial was suspended. Further consideration of if, when, and how to continue the development of TSC as a treatment for GBM — including any restart of the TSC GBM Trial — will take place following completion of the TSC Oxygenation Trials.
Stroke
We believe TSC has potential applications in both ischemic and hemorrhagic stroke through mitigation of stroke-induced hypoxia and related damage to brain tissue. A stroke occurs when blood flow to the brain is blocked, referred to as an ischemic stroke, or a blood vessel in the brain ruptures, referred to as a hemorrhagic stroke, causing damage to surrounding brain tissue. According to the U.S. Centers for Disease Control and Prevention, stroke is the fifth leading cause of death and a leading cause of adult disability in the U.S. The hypoxic conditions in the brain of stroke patients may be a significant factor contributing to morbidity and mortality and, based upon certain preclinical safety and efficacy data and additional clinical safety data, we believe TSC could enhance the diffusion of oxygen into brain tissue to reduce stroke-induced hypoxia and neuronal death.
In October 2019, we began enrolling patients in our TSC Stroke Trial, a randomized Phase 2 trial designed to evaluate TSC in the treatment of acute ischemic or hemorrhagic stroke. The TSC Stroke Trial was planned to enroll 160 total patients, evenly split between the TSC treatment arm and the control arm, with all patients to receive TSC or placebo treatment, as applicable, while in the ambulance to ensure treatment as soon as possible after the onset of clinical symptoms. However, due to a variety of factors, including challenges related to the trial's on-ambulance treatment design and overall healthcare system capacity related to the onset of the COVID-19 pandemic during the first half of 2020, we voluntarily terminated the TSC Stroke Study in the second half of 2020 in order to prioritize resources to shorter duration studies, including the TSC COVID Trial and the TSC Oxygenation Trials. Further consideration of if, when, and how to continue the development of TSC as a treatment for stroke will take place following completion of the TSC Oxygenation Trials.
COVID-19
We believe TSC’s oxygen-enhancing mechanism could potentially provide benefit to patients with low oxygen levels at risk of developing ARDS and multiple organ failure, such as patients with COVID-19.
On September 10, 2020, we announced the dosing of the first two patients in our TSC COVID Trial evaluating TSC in hospitalized COVID-19 patients at the NIID in Bucharest, Romania. The primary endpoint of the TSC COVID Trial was to evaluate the safety and tolerability of TSC administered every six hours for at least five and up to 15 days, a more frequent dosing regimen than had been used in our previous clinical studies. Secondary endpoints included pharmacokinetic measurement of TSC levels after dosing, relative improvements in blood oxygen levels, and certain other clinical parameters related to COVID-19.
On February 9, 2021, we completed dosing of the twenty-fourth and final patient in the TSC COVID Trial. No dose-limiting toxicities or serious adverse events were observed among any patients in the study, including those who received the highest dose of 1.5 mg/kg every 6 hours. Evaluation of secondary endpoint data is ongoing and we anticipate this data will be available early in the second quarter of 2021. Further consideration of if, when, and how to continue the development of TSC as a treatment for COVID-19 — including any commencement of enrollment in the previously announced Phase 2b portion of the TSC COVID Trial — will take place following completion of the TSC Oxygenation Trials.
Our Anticipated Next Steps in the TSC Development Program: The TSC Oxygenation Trials
Following completion of the TSC COVID Trial in February 2021, the next step we have planned in the development of TSC is the design and execution of a trilogy of clinical studies using short-term, experimental models to evaluate the clinical effects of TSC on oxygenation. As of March 15, 2021, TSC has been administered to more than 180 subjects in our clinical trials. Data from these clinical trials have contributed significantly to our understanding of the safety, tolerability, and pharmacokinetics of TSC. In addition, post hoc analyses of two of our prior studies involving patients with peripheral artery disease with claudication and unresected GBM tumors have provided preliminary signals suggesting TSC's effect on oxygenation. However, neither of these studies was statistically powered to formally evaluate efficacy, and we therefore believe that further, robust clinical development of TSC requires a prospective exploration of the relationship between the level of exposure (dose) and response (change in oxygenation).
To this end, we plan to execute three short-term clinical trials during 2021, each of which we expect to conduct in the U.S. and fund with cash-on-hand. We believe positive data from any one or more of the three TSC Oxygenation Trials would provide evidence of a definitive effect of TSC on oxygenation, whether through increased uptake in the lungs, enhanced delivery, increased utilization at the tissue level, or some combination thereof. Positive data from any of these studies, if obtained, will also guide the subsequent steps of our development strategy focused on demonstrating the clinical and therapeutic benefits of TSC in relevant patient populations across the hypoxia continuum.
The TSC TCOM Trial: TSC's Effects on Peripheral Tissue Oxygenation
The first of the three Oxygenation Trials is the TSC TCOM Trial, which we expect to initiate before the end of March 2021. The TSC TCOM Trial is designed as a double-blind, randomized, placebo-controlled study in healthy volunteers breathing 100.0% oxygen. The study is designed to evaluate the effects of TSC on peripheral tissue oxygenation using a TCOM device. The TCOM device directly measures the release of oxygen from the blood vessels through the skin and is commonly used to predict the likelihood of wound healing, the potential for success with hyperbaric therapy, and to map the appropriate location for limb amputation. The TSC TCOM Trial will test single, ascending doses of TSC in an attempt to establish the exposure-response relationship between TSC and enhanced oxygen delivery and will be statistically powered to evaluate the difference in tissue oxygenation levels versus placebo. We anticipate this study will be completed in the second quarter of 2021, with topline results available within two months following study completion.
The TSC Induced Hypoxia Trial: TSC's Effects Under Induced Hypoxic Conditions
The second TSC Oxygenation Trial is our planned TSC Induced Hypoxia Trial, which we expect to initiate in the third quarter of 2021. The TSC Induced Hypoxia Trial is expected to be a double-blind, randomized, placebo-controlled study which will evaluate the effects of TSC on maximal oxygen consumption, or VO2, and partial pressure of blood oxygen, or PaO2, in normal healthy volunteers exposed to simulated altitude conditions that induce hypoxia. Subjects in the TSC Induced Hypoxia Trial will be subjected to incremental levels of physical exertion while exposed to hypoxic and hypobaric conditions. The primary endpoints in the TSC Induced Hypoxia Trial will be change from baseline in VO2 and PaO2 after receiving a single intravenous dose of TSC. The study will be statistically powered to evaluate the difference in effect of escalating doses of TSC versus placebo in an attempt to establish a dose-response relationship on oxygen consumption and availability. We anticipate this study will be completed in the second half of 2021, with topline results available within two months of study completion.
The TSC DLCO Trial: TSC's Effects on Oxygen Transfer Efficiency
The third TSC Oxygenation Trial is our planned TSC DLCO Trial, which we expect to initiate in the third quarter of 2021. The TSC DLCO Trial is expected to be a double-blind, randomized, placebo-controlled study which will evaluate the effects of TSC on the diffusion of carbon monoxide through the lungs, also known as DLCO, in patients with previously diagnosed interstitial lung disease who have a baseline DLCO test result that is abnormal. DLCO testing is commonly performed as part of standard pulmonary function testing and aids in the diagnosis or dyspnea, also known as shortness of breath, as well as to track improvement or progression over time on prescribed treatments. In the TSC DLCO Trial, DLCO will act as a surrogate measure of oxygen transfer efficiency, or uptake, from the alveoli of the lungs, through the plasma, and onto hemoglobin within red blood cells. The TSC DLCO Trial TSC will test single, ascending doses of TSC in an attempt to establish the exposure-response relationship between TSC and oxygen transfer efficiency. The study will be statistically powered to evaluate the difference in effect of TSC on improvement in DLCO versus placebo. We anticipate this study will be completed in the second half of 2021, with topline results available within two months of study completion.
Anticipated Next Steps Following Completion of the TSC Oxygenation Trials
Assuming success in one or more of the three TSC Oxygenation Trials, we expect to identify and announce the specific, hypoxia-related indications we will target in advancing TSC's development in the fourth quarter of 2021 and intend to initiate our Planned Phase 2 Hypoxia-related Indication Trial in the first quarter of 2022.
Certain medical conditions complicated by hypoxia.
Our Second Product Candidate: DFN-529
Our product candidate DFN-529 is a novel PI3K/Akt/mTOR pathway inhibitor which has completed two Phase 1 clinical trials for age-related macular degeneration and was previously in preclinical development in oncology, specifically for GBM. DFN-529 has shown activity in both in vitro and in vivo GBM animal models and has been demonstrated to be orally bioavailable and capable of crossing the blood brain barrier. We are currently in discussions with multiple third-party research scientists regarding the potential initiation of further studies to evaluate the effects of DFN-529 in preclinical cancer models. We believe these additional data may help guide us in choosing an appropriate clinical development pathway for DFN-529. We also continue to explore business options for DFN-529, including out-licensing opportunities and other strategic partnerships.
Products, Product Development, and Our Competition
Research and Development
In recent years, the majority of our research and development expenditures have been directed to the development of TSC. For example, during the year ended December 31, 2020, we incurred approximately $9.4 million in costs related to research and development of our products, an increase of approximately $2.8 million compared to the year ended December 31, 2019. The majority of these costs were related to the development of TSC and related personnel, including costs associated with the TSC COVID Trial, which commenced in September 2020 and was completed in February 2021, as well as the wind down of the TSC Stroke Trial, which commenced in October 2019 and was terminated in the second half of 2020.
We expect this trend to continue for the foreseeable future, including planned expenditures related to the TSC Oxygenation Trials and other costs associated with the conduct of additional, supportive preclinical studies and general research and development activities related to TSC. We intend to focus the remainder of our foreseeable research and development expenditures will on the development of DFN-529, including additional preclinical studies evaluating DFN-529 in cancer models that we are in the early stages of planning.
As a development-stage company, we continuously evaluate opportunities to improve the value of our development pipeline, including potential acquisition and in-licensing opportunities. Our efforts include ongoing evaluation and modification of our development plans intended to maximize the probability of technical, developmental, and regulatory success while enhancing patient and stockholder value. These activities have, and we expect they will continue to be for the foreseeable future, focused on opportunities that are synergistic with our overall corporate strategy to develop novel treatments for the treatment of hypoxia and conditions complicated by hypoxia.
Our Intellectual Property
We believe that a strong intellectual property portfolio is critical to our success. We are committed to obtaining and maintaining appropriate patent and other protections for our products candidates and other technologies, preserving and protecting our trade secrets and other confidential and proprietary information, and fiercely defending our intellectual property portfolio against any potential infringement by third parties. We attempt to protect our intellectual property through among other things, the filing of applications for patent, trademark, and other appropriate intellectual property protections, the use of confidentiality agreements with consultants, contractors and other third parties, our employee policies regarding confidentiality, invention disclosure, and the assignment of inventions, as well as regular meetings of members of our internal development and legal teams, which contains key members of our management team. We are also committed to operating our business without infringing on the intellectual property of others.
In general, patents extend for varying periods according to the date of patent filing or grant and the legal term of patents in various countries where patent protection is obtained, with term adjustments or extensions possible in certain cases based on patent office delays or pursuant to certain administrative and legislative exceptions. The actual protection afforded by a patent, which can vary from country to country, depends on the type of patent, the scope of its coverage and the availability of legal remedies in the country.
We continue to invest significant time, effort, and resources into the development and maintenance of our patent portfolio. As of December 31, 2020, we owned 16 issued U.S. patents and 36 issued non-U.S. patents, and had numerous patent applications pending worldwide including issued patents and applications in major markets such as the U.S., E.U., China, Japan, and India. The normal life (i.e. with no adjustments or extensions) of our key issued patents related to the composition of matter of TSC extends to 2026, with potential patent term extensions to 2031, and the normal life of our patents related to an oral formulation of TSC extends to 2031, with potential patent term extensions to 2036. Additional information regarding patent term extensions can be found under the heading, see — Government Regulation — The Hatch-Waxman Amendments — Patent Term Restoration and Marketing Exclusivity. In addition, TSC has been granted Orphan Drug designation by the FDA for the treatment of both GBM and metastatic brain cancer, which may provide us with certain rights of exclusivity under certain FDA regulations. Additional information regarding orphan and ultra-orphan can be found under the heading, see — Government Regulation — Certain Other FDA Regulations — The Orphan Drug Act of 1983.
Materials and Manufacturing
We do not currently own or operate anymanufacturing facilities, suitable fornor do we have plans to develop our own manufacturing TSC or any of our other product candidates on a scale required to support either clinical development or commercialization. We currently use third-party CMOs to manufacture API, other starting materials, and finished drug product for our preclinical studies and clinical trials and we intend to continue doing so foroperations in the foreseeable future. Our lead product candidate, neflamapimod, is a small molecule drug that is manufactured using commercially available technologies.
Our RewinD-LB Trial is being conducted with drug substance (or API) that has already been manufactured. This drug substance was manufactured at an established commercial CMO, that is approved for and manufactures drug both for investigational use and marketed products. We would anticipate utilizing this strategy willCMO for clinical trials beyond the Phase 3 clinical trial in DLB, as well as potentially for commercial use if neflamapimod is approved. However, supplies of our neflamapimod drug substance could be scalable ininterrupted from time to time, and we cannot be certain that alternative supplies could be obtained within a manner sufficient to support the production capacity required to support continued clinical development and ultimate commercialization. However, we do not have any formal agreementsreasonable timeframe, at this time with any CMO to cover commercial productionan acceptable cost, or at all. For a further description of TSC or any other product candidate. Although the use of third party CMOs to manufacture pharmaceutical products is common within the industry, this dependence on third-party CMOs exposes our business to certain risks including those described in related to our manufacturing, see “Part I –Item 1A. Risk Factors – Risks Related to the Company’s Clinical Development and Regulatory Approval – The Company’s reliance on third parties for the production of neflamapimod may result in delays in the Company’s clinical trials or regulatory approvals and Commercializationmay impair the development and ultimate commercialization of TSCneflamapimod, which would adversely impact the Company’s business and Our Other Product Candidatesfinancial position..
Our Competition”
Currently, medical optionsWe also currently rely on a third-party CMO (different than that for drug substance) for the manufacture of our neflamapimod drug product. We have used the same manufacturer for our neflamapimod drug product in all our clinical trials to improve oxygenation without riskdate. If neflamapimod is ultimately approved for commercial sale, we expect to continue to rely on third-party contractors for manufacturing the drug product. Although we intend to do so prior to any commercial launch, we have not yet entered into long-term agreements for the commercial supply of hyper-oxygenationeither drug substance or drug product with our current manufacturing providers, or with any alternate manufacturers.
Competition
Given the potential market opportunity for the treatment of DLB and other neurodegenerative diseases, an increasing number of established pharmaceutical firms and smaller biotechnology/biopharmaceutical companies are limited andpursuing a range of potential therapies for these diseases in various stages of clinical development.
While there are numerous companies pursuing AD disease modifying approaches, we believe that TSC's diffusion enhancing mechanism of action make it a first-in-class, novel, small molecule. However, there are severala limited number of companies currently developing or marketing products, therapeutics, or devicesand disease modifying approaches for DLB. With regard to public biopharmaceutical companies that may nevertheless bewe would consider competitive with TSC, if approved, including NuvOx Pharma LLC, Hemoglobin Oxygen Therapeutics LLC,our approach, and Cardax, Inc. In addition,actively evaluating treatments in the first quarter of 2021,DLB, we becameare aware of a third party affiliated with a former outside consultantEisai Co. Ltd., Cognition Therapeutics, Inc. and Athira Pharma, Inc., all of the Company which claims to bewhom remain in early-stageclinical stage development of their potential DLB treatments. None of these companies, however, are developing a product candidate that purportedly may operate through a similar mechanismtreatment specifically targeting patients with pure DLB, the target patient population of action to TSC. It is unclear if this particular product candidate would, if developed and approved, actually be competitive with TSC.our ongoing RewinD-LB Trial.
Our industry is highly competitive and subject to rapid and significant change. Potential competitors in the United States are numerous and include major pharmaceutical and specialty pharmaceutical companies, smaller biopharmaceutical companies, research universities, and others. The biopharmaceuticalbiotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on developing proprietary therapeutics. Numerousproducts. We face potential competition from many different sources, including pharmaceutical and biotechnology companies, are engagedacademic institutions and governmental agencies and public and private research institutions. Any product candidates that we successfully develop and commercialize, including neflamapimod, may compete with existing therapies and new therapies that may become available in the development, patenting, manufacturing, and marketing of health care products competitive with those that we are developing. Many of ourfuture.
Our competitors may have longer operating histories, greater name recognition, substantiallysignificantly greater financial resources, an established presence in the market, and largersignificantly greater expertise in research and development, staffs than we do, as well as substantially greater experience than us in developing products,manufacturing, preclinical and clinical testing, obtaining regulatory approvals and manufacturingreimbursement and marketing pharmaceutical products.approved products than we do. These competitors also compete with us in recruiting and retaining qualified scientific, sales, marketing and management personnel, establishing clinical trial sites and subject registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
The key competitive factors affecting the success of neflamapimod, and any other product candidates that we develop to address DLB and other CNS diseases, if approved, are likely to be their efficacy, safety, convenience, price, the level of competition, and the availability of reimbursement from government and other third-party payors. Our potential commercial opportunity could also be reduced or eliminated if our competitors develop and commercialize products that are more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours. In addition, a significant amount of research is carried out at academic and government institutions. These institutions are aware ofour ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the commercial value of their findings and are aggressive in pursuing patent protection and negotiating licensing arrangements to collect royalties for use of technology that they have developed. One or more of these companies or other entities may have one or more products under development that would be competitive with TSC.generic products.
Sales and Marketing
Intellectual Property
We currentlystrive to protect and enhance the proprietary technologies, inventions and improvements that we believe are important to our business, including seeking, maintaining and defending patent rights, whether developed internally or licensed from third parties. Our policy is to seek to protect our proprietary position by, among other methods, pursuing and obtaining patent protection in the United States and in jurisdictions outside of the United States related to our proprietary technology, inventions, improvements and our product candidates that are important to the development and implementation of our business.
We have no marketed productsmade a number of discoveries related to our lead product candidate, neflamapimod, which are reflected in ten main patent families, each of which we wholly own (dates below are without consideration of potential patent term extension, see section titled “—Patent Term Restoration” below):
| ● | The first patent family relates to methods of treating patients suffering from AD, as well as methods of reducing amyloid plaque burden. In this family, we hold issued patents in the United States, Europe, Japan, China, Canada, Australia, and Hong Kong. These patents are set to expire in 2032. |
| ● | The second patent family relates to the use of neflamapimod for improving cognition. In this family, we hold issued patents in the United States, Europe, Japan, and a pending application in China. These patents are set to expire in 2035. |
| ● | The third patent family relates to co-crystals of neflamapimod in this family, we hold an issued patent in the United States. This patent is set to expire in 2038. |
| ● | The fourth patent family relates to methods for promoting recovery of function in patients who have suffered acute neurologic injuries, including those resulting from various forms of stroke. In this family, we hold an issued patent in the United States, Europe, and Japan, and pending applications in Korea and China. These patents are set to expire in 2035-2036. |
| ● | The fifth patent family relates to methods of treating patients suffering from dementia. In this family, we have an issued patent the United States for the treatment to patients with MCI to improve episodic memory and a pending application in Europe. Patents that issue in this family, if any, are expected to expire in 2037. |
| ● | The sixth patent family relates to formulations of neflamapimod, including pharmaceutical compositions for oral administration exhibiting desirable pharmacokinetics and processes for the manufacture thereof. In this family, we have an issued patent in the United States that is set to expire in 2039. |
| ● | The seventh patent family relates to the treatment of DLB. In this family we have pending applications in the United States, Europe, Japan, China, Canada, and Hong Kong. Patents that issue in this family, if any, are expected to expire in 2040. |
| ● | The eighth patent family is co-owned by Boston University and relates to methods of treating prion disease. In this family, we have a pending application in the United States. Patents that issue in this family, if any, are expected to expire in 2040. |
| ● | The ninth patent family relates to treatment of gait dysfunction related to neurodegenerative disease. An International Application is pending. Patents that issue in this family, if any, are expected to expire in 2041. |
| ● | The tenth patent family relates to treatment of a subpopulation of patients having DLB but no substantial Alzheimer’s like tau pathology. Patents that issue in this family, if any, are expected to expire in 2042. |
Pursuant to the terms and accordingly, currentlyconditions of the Vertex Agreement, Vertex has granted us an exclusive license under specified Vertex patent rights, including U.S patent No. 5,945,418, which relates to the composition of matter for neflamapimod. This patent expired in 2017.
Individual patents extend for varying periods depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, patents issued for regularly filed applications in the United States are granted a term of 20 years from the earliest effective non-provisional filing date. In addition, in certain instances, a patent term can be extended to recapture a portion of the USPTO delay in issuing the patent as well as a portion of the term effectively lost as a result of the FDA regulatory review period. However, as to the FDA component, the restoration period cannot be longer than five years and the total patent term including the restoration period must not exceed 14 years following FDA approval. The duration of foreign patents varies in accordance with provisions of applicable local law, but typically is also 20 years from the earliest effective filing date. However, the actual protection afforded by a patent varies on a product-by-product basis, from country to country and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent.
We also rely upon trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality agreements and invention assignment agreements with our collaborators, employees and consultants, as we determine necessary. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. These agreements may be breached, and we may not have no salesadequate remedies for any breach. In addition, our trade secrets may otherwise become known or marketing personnel.be independently discovered by competitors. To the extent that our collaborators, employees and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Our commercial success will also depend in part on not infringing upon the proprietary rights of third parties. It is uncertain whether the issuance of any third-party patent would require us to alter our development or commercial strategies, or our drugs or processes, obtain licenses from third parties or cease certain activities.
From time to time, we may find it necessary or prudent to obtain licenses from third party patent owners. Where licenses are available at reasonable cost, such licenses are considered a normal cost of doing business. In other instances, we may use the results of freedom-to-operate studies to guide our early-stage research away from areas where we are likely to encounter obstacles in the form of third-party intellectual property. We strive to identify potential third-party intellectual property issues in the early stages of research in our programs in order to minimize the cost and disruption of resolving such issues.
Our breach of any license agreements or failure to obtain a license to proprietary rights that we may require to develop or commercialize our future drugs may have an adverse impact on us.
For more information, please see “Item 1A. Risk Factors—Risks Related to the Company’s Intellectual Property.”
Government Regulation
Pharmaceuticals like TSC, DFN-529,The FDA and other product candidates we may develop are highly regulated by governmentalcomparable regulatory authorities in other countries impose requirements upon companies involved in the U.S.clinical development, manufacture, marketing and distribution of drugs, such as those we are developing. These requirements can, in some instances, be substantial and burdensome. These agencies and other countries at the federal, state and local levels. These regulations are numerous and extensive in their scope, relating to,entities regulate, among other things, the research and development, testing, manufacture, storage, quality control, safety, effectiveness, labeling, storage, record keeping, approval, advertising and testing, approval, labeling and packaging, promotion, marketing, and advertising, distribution, post-approval monitoring and reporting, sampling and export and import and record keeping of pharmaceutical products.
The FDA Drug Approval Process
Generally, before a new pharmaceutical product can be marketed, considerable data demonstrating its quality, safety and efficacy must be obtained, organized into a format specific for each applicable regulatory authority, submitted for review, and approved by the competent regulatory authority. In the United States, the competent regulatory authority is the FDA, which, pursuant to the FDC Act, is responsible for the review and approval of all data required to support a license to commercially market pharmaceuticals such as TSC and DFN-529.
The process of obtaining regulatory approvals and the subsequent compliance with FDAapplicable federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resourcesresources.
U.S. Government Regulation of Drug Products
In the United States, the FDA regulates drugs under the FDCA and failureits implementing regulations. Failure to comply with the applicable U.S. requirements at any time during the product development process,and approval process or if approved, followingafter approval may subject an applicant to a variety of administrative or judicial sanctions. These sanctions could include, among other actions, the FDA’s refusal to approve a pending applications,NDA, withdrawal of an approval, license revocation,imposition of a clinical hold, untitled orissuance of warning letters voluntary or mandatoryother notices of violation, product recalls or market withdrawals, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement andor civil or criminal penalties, any of whichpenalties. Any agency or judicial enforcement action could have a material adverse effect on our business financial position, orand results of operations.
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
| ● | Completion of nonclinical laboratory tests, potentially animal studies and formulation studies in compliance with the FDA’s GLP regulations; |
| ● | Submission to the FDA of an IND, which must become effective before human clinical trials may begin; |
| ● | Approval by an IRB covering each clinical trial site before each trial may be initiated at that site; |
| ● | Performance of adequate and well-controlled human clinical trials in accordance with GCP regulations and other clinical trial-related requirements to establish the safety and efficacy of the proposed drug product for each indication; |
| ● | Submission to the FDA of an NDA seeking marketing approval; |
| ● | A determination by the FDA within 60 days of its receipt of an NDA that the NDA is sufficiently complete to permit a substantial review, in which case the NDA is filed; |
| ● | Satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP requirements and to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity; |
| ● | Satisfactory completion of FDA audits of clinical trial sites that generated data in support of the NDA to assure compliance with GCP regulations and the integrity of the clinical data and/or FDA audits of the nonclinical studies submitted as part of the NDA; and |
| ● | FDA review and approval of the NDA, including consideration of the views of an FDA advisory committee, if one was involved, prior to any commercial marketing or sale of the drug in the United States. |
Preclinical Studies and IND
Preclinical, or nonclinical studies generally include laboratory evaluation of product chemistry, toxicity and formulation, as well as in vitro and animal studies to assess the potential for adverse events and in some cases to establish a rationale for the investigational product’s therapeutic use. The Consolidated Appropriations Act for 2023, signed into law on December 29, 2022, (P.L. 117-328) amended the FDCA to specify that nonclinical testing for drugs may, but is not required to, include in vivo animal testing. According to the amended language, a sponsor may fulfill nonclinical testing requirements by completing various in vitro assays (e.g., cell-based assays, organ chips, or microphysiological systems), in silico studies (i.e., computer modeling), other human or non-human biology-based tests (e.g., bioprinting), or in vivo animal tests. The conduct of nonclinical studies is subject to federal regulations and requirements, including GLP regulations.
Process Overview
The FDA drug approval process generally involvesAn IND sponsor must submit the following steps:
• completion of extensive preclinical laboratory studies , including studies conducted in accordance with GLP requirements;
• submission to the FDA of an IND application, which must become effective before clinical trials involving human subjects or patients may begin;
• performance of adequate and well-controlled human clinical trials in accordance with applicable IND regulations, GCP requirements, and other clinical trial-related regulations to establish the safety and efficacy of the investigational product for each proposed indication, including approval by an IRB or independent ethics committee before each trial may be initiated;
• submission to the FDA of an NDA;
• a determination by the FDA within 60 days of its receipt of an NDA as to whether it will accept the filing for review;
• satisfactory completion of one or more FDA pre-approval inspections of the manufacturing facility or facilities where the drug will be produced to assess compliance with cGMP requirements and to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity;
• a potential FDA audit of the clinical trial sites that generated the data in support of the NDA;
• payment of user fees for FDA review of the NDA; and
• FDA review and approval of the NDA, including consideration of the views of any FDA advisory committee, prior to any commercial marketing or sale of the biologic or drug in the United States.
The preclinical and clinical testing and approval process requires substantial time, effort, and financial resources and satisfaction of FDA pre-market approval requirements typically takes many years, though the actual time required may vary substantially based upon the type, complexity, and novelty of the applicable product or indication to be treated. We cannot be certain that any approvals for TSC, DFN-529, or any product candidates we attempt to develop in the future will be granted on a timely basis or at all.
Preclinical Studies
Preclinical studies include laboratory evaluation of product chemistry, formulation, and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the drug. The results of preclinical testing are submittedtests, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. An IND along with other information relatedis a request for authorization from the FDA to theadminister an investigational new drug including information regarding its chemical make-up, manufacturing process,to humans, and quality controls, as well as a proposedit must become effective before human clinical trial protocol. Long-term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity,trials may begin. Some long-term nonclinical testing may continue even after the IND is submitted.submitted and clinical trials have been initiated. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA issues a notice expressly authorizing the proposed trial to proceed or raises concerns or questions related to one or more proposed clinical trials and places the clinical trial on a clinical hold. If the agency imposes a hold, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to initiate. Clinical holds also may be imposed by the FDA at any time before or during clinical trials due to safety concerns or non-compliance. A separate submission to an existing IND must also be made for each successive clinical trial conducted during product development.
Clinical Trials
Following completion of preclinical studies and the submission on an IND to the FDA, a 30-day waiting period is required. If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical trials involve the administration of anthe investigational new drug to healthy volunteers or patientshuman subjects under the supervision of a qualified investigator. These trials must be conductedinvestigators (generally physicians not employed by or under the trial sponsor’s control) in complianceaccordance with federal regulationsGCP requirements, which include the requirement that all research subjects provide their informed consent in writing for their participation in any clinical trial, as well as GCP,review and approval of the trial by an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors. Each trial isIRB for each participating site. Clinical trials are conducted under a protocolprotocols detailing, among other things, the objectives of the trial, the trial procedures, subject selection and exclusion criteria, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. EachA protocol involving testing on U.S. patientsfor each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. In addition, an IRB acting on behalf of each institution participating in the clinical trial must review and approve the trial plan, informed consent forms, and communications to trial subjects before the trial commences at that institution. An IRB considers, among other things, whether the risks to individuals participating in the trials are minimized and are reasonable in relation to anticipated benefits, and whether the planned human subject protections are adequate. The IRB must continue to oversee the clinical trial while it is being conducted and must operate in compliance with FDA regulations.
Sponsors of certain clinical trials generally must register such trials and disclose certain trial information within specific timeframes to the NIH for public dissemination on the ClinicalTrials.gov data registry. Information related to the investigational product, patient population, phase of investigation, trial sites and investigators and other aspects of the clinical trial is made public as part of the registration of the clinical trial. Sponsors are also obligated to disclose the results of their clinical trials after completion, but such disclosures may be delayed in some cases for up to two years after the date of completion of the trial. Failure to timely register a covered clinical study or to submit study results as provided for in the law can give rise to civil monetary penalties and also prevent the non-compliant party from receiving future grant funds from the federal government. The U.S. Department of Health and Human Services’ Final Rule and NIH’s complementary policy on ClinicalTrials.gov registration and reporting requirements became effective in 2017, and the government has brought enforcement actions against non-compliant clinical trial sponsors. Competitors may use the publicly available information about clinical trials to gain knowledge regarding the progress of development programs. Sponsors or distributors of investigational products for the diagnosis, monitoring, or treatment of one or more serious diseases or conditions must also have a publicly available policy on evaluating and responding to requests for expanded access requests.
Human clinical trials are typically conducted in three sequential phases, which may overlap or be combined:
| ● | Phase 1: The drug candidate is initially administered to healthy human volunteers and tested for safety, dosage tolerance, structure-activity relationships, mechanism of action, absorption, metabolism, distribution, and excretion. In the case of some products for severe or life-threatening diseases, such as cancer, especially when the product may be too inherently toxic to administer ethically to healthy volunteers, the initial human testing is often conducted in patients with the target disease or condition. If possible, Phase 1 trials may also be used to gain an initial indication of product effectiveness. |
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap, especially in certain indications such as cancer.
| ● | Phase 2: The drug candidate is administered to a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance and optimal dosage. Multiple Phase 2 clinical trials may be conducted by the sponsor to obtain information prior to beginning larger and more extensive clinical trials. |
• Phase 1 - In Phase 1 trials, an investigational new drug is introduced into healthy human subjects and is evaluated to assess pharmacological actions, side effects associated with increasing doses and, in certain cases, early evidence on efficacy.
| ● | Phase 3: The drug is administered to an expanded patient population, generally at geographically dispersed clinical trial sites, in well-controlled clinical trials to generate enough data to evaluate the efficacy and safety of the product for its intended use, to establish the overall risk-benefit profile of the product, and to provide adequate information for the labeling of the product as well as an adequate basis for marketing approval. Typically, two adequate, well-controlled multicenter trials are required by the FDA for drug product approval. Under some limited circumstances, however, the FDA may approve an NDA based upon a single Phase 3 clinical trial plus confirmatory evidence from a post-market trial or, alternatively, a single large, robust, well-controlled multicenter trial without confirmatory evidence. |
• Phase 2 - In Phase 2 trials, the drug is introduced to a limited patient population in a particular indication to determine metabolism, pharmacokinetics, the effectiveness of the drug for the indication, dosage tolerance and optimum dosage, and to identify potential adverse effects and safety risks.
• Phase 3 - In Phase 3 trials, if a drug has demonstrated evidence of effectiveness and an acceptable safety profile in prior Phase 2 trials, the drug is introduced to a larger patient population in the relevant indication to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug, and to provide adequate information for the labeling of the drug, if approved.
Not all drug development programs are required to follow the order and content of all three phases. For example, in August 2018, the FDA released a draft guidance entitled, “Expansion Cohorts: Use in First-In-Human Clinical Trials to Expedite Development of Oncology Drugs and Biologics,” which outlines how drug developers can utilize an adaptive trial design commonly referred to as a seamless trial design in early stages of oncology drug development, i.e., the first-in-human clinical trial, to compress the traditional three phases of trials into one continuous trial called an expansion cohort trial. Information to support the design of individual expansion cohorts is included in IND applications and assessed by FDA. Expansion cohort trials can potentially bring efficiency to drug development and reduce developmental costs and time.
Post-approval trials, sometimes referred to as Phase 4 clinical trials, may be conducted after initial marketing approval. These trials are used to gain additional experience from the treatment of patients in the intended therapeutic indication and are commonly intended to generate additional safety data regarding use of the product in a clinical setting.
| ● | Post-approval Trials: Sometimes referred to as “Phase 4” clinical trials, these trials may be conducted after initial marketing approval and are used to gain additional experience from the treatment of patients in the intended therapeutic indication. In certain instances, the FDA may mandate the performance of Phase 4 clinical trials as a condition of approval of an NDA. |
Progress reports detailing the results of the clinical trials among other information, must be submitted at least annually to the FDA and written IND safety reports must be submitted to the FDA and the investigators 15 calendar days after the trial sponsor determines the information qualifies for reporting for serious and unexpected suspected adverse events, findings from other studies orsuggesting a significant risk to humans exposed to the investigational drug, findings from animal or in vitro testing that suggest a significant risk for human subjects and any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator brochure. The sponsor also must notify the FDA of any unexpected fatal or life-threatening suspected adverse reaction as soon asIt is possible but in no case later than seven calendar days after the sponsor’s initial receipt of the information.
that Phase 1, Phase 2 or Phase 3 and other types of clinical trials may not be completed successfully within any specified period, ifor at all. The FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the patientsresearch subjects are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients. Sponsors may also choose to discontinue clinical trials as a result of risks to subjects, a lack of favorable results, or changing business priorities. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group provides authorization for whether a trial may move forward at designated checkpoints based on access to certain data from the trial.
Congress also recently amended the FDCA, as part of the Consolidated Appropriations Act for 2023, in order to require each sponsor of a Phase 3 clinical trial, or other “pivotal study” of a new drug to support marketing authorization, to design and submit a diversity action plan for such clinical trial. The action plan must include the sponsor’s diversity goals for enrollment, as well as a rationale for the goals and a description of how the sponsor will meet them. A sponsor must submit a diversity action plan to the FDA by the time the sponsor submits the relevant clinical trial protocol to the agency for review. The FDA may grant a waiver for some or all of the requirements for a diversity action plan. It is unknown at this time how the diversity action plan may affect Phase 3 trial planning and timing or what specific information FDA will expect in such plans, but if the FDA objects to a sponsor’s diversity action plan or otherwise requires significant changes to be made, it could delay initiation of the relevant clinical trial.
Concurrent with clinical trials, companies may perform additional nonclinical studies and develop additional information about a drug candidate’s chemistry and physical characteristics as well as finalize a process for its manufacturing in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the drug candidate and, among other things, the manufacturer must develop methods for testing the identity, strength, quality and purity of the final drug product. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that a drug candidate does not undergo unacceptable deterioration over its proposed labeled shelf life.
New DrugMarketing Application Submission, Review by the FDA, and FDA Review ProcessMarketing Approval
AfterAssuming successful completion of all required testing in accordance with all applicable regulatory requirements, the requiredresults of product development, preclinical studies and clinical testing, an NDA is prepared andtrials are submitted to the FDA containing data intendedas part of an NDA requesting approval to provide substantial evidence thatmarket the drug is safe and effective in the relevant indication, and FDA approval of theproduct for one or more indications. The NDA is required before commercial marketingmust contain proof of the product may begincandidate’s safety and substantial evidence of effectiveness for its proposed indication or indications in the United States. The NDA must include theform of relevant data available from pertinent preclinical and clinical studies, including negative or ambiguous results of all preclinical, clinical, and other testing and a compilation of dataas well as positive findings, together with detailed information relating to the product’s pharmacology, chemistry, manufacturing, controls, and controls. The costproposed labeling, among other things. In particular, a marketing application must demonstrate that the manufacturing methods and quality controls used to produce the drug product are adequate to preserve the drug’s identity, strength, quality, and purity. Data can come from company-sponsored clinical trials intended to test the safety and effectiveness of preparing and submittinga use of the product, or from a number of alternative sources, including studies initiated by investigators. FDA approval of an NDA must be obtained before the corresponding drug may be marketed in the United States.
Under PDUFA, each NDA submission is subject to a substantial application user fee, and the submissionsponsor of most NDAsan approved NDA is also subject to substantial initial and ongoing fees.an annual program fee. The FDA adjusts the PDUFA user fees on an annual basis. The application user fee must be paid at the time of the first submission of the application, even if the application is being submitted on a rolling basis. Fee waivers or reductions are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business.
The FDA hasreviews all NDAs submitted to determine if they are substantially complete before it accepts them for filing and may request additional information rather than accepting a submission for filing. The FDA must make a decision on accepting an NDA for filing within 60 days from itsof receipt and must inform the sponsor by the 74th day after the FDA’s receipt of an NDA to determinethe submission whether the NDA will be accepted for filing based on the agency’s threshold determination that itapplication is sufficiently complete to permit substantive review. The FDA may refuse to file any submission that it deems incomplete or not properly reviewable at the time of submission and may request additional information. In this event, the marketing application must be resubmitted with the additional information requested by the agency. The resubmitted application is also subject to review before the FDA accepts it for filing.
Once the submissionan NDA is accepted for filing, the FDA begins an in-depthFDA’s goal is to review subject to certain performance goals agreed upon by the FDA. Priorityapplication within 10 months after it accepts the application for filing, or, if the application meets the criteria for “priority review, can be applied in certain instances, including with respect to drugs that” six months after the FDA determines offer major advances in treatment or provide a treatment where no adequate therapy exists.accepts the application for filing. The review process whether standardis often significantly extended by FDA requests for additional information or priority,clarification after the NDA has been accepted for filing. The review process may be extended by the FDA for three additional months to consider certain late-submittednew information or information intendedin the case of a clarification provided by the applicant to clarify information already provided inaddress an outstanding deficiency identified by the FDA following the original submission.
During the review process, the FDA reviews the NDA to determine, among other things, whether the product is safe and effective and whether the facility in which it is manufactured, processed, packed, or held meets standards designed to assure the product’s continued strength, quality, and purity. The FDA may refer any NDA, including applications for novel drug candidates which present difficult questions of safety or efficacy to an advisory committee to provide clinical insight on application review questions. Typically, an advisory committee is a panel of independent experts, including clinicians and other scientific experts that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it considers such recommendations carefully when making final decisions on approval.
Before approving an NDA, the FDA will typically inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent manufacture of the product within required specifications. Additionally, before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally,If the FDA determines that the application, manufacturing process or manufacturing facilities are not acceptable, it will inspectoutline the facilitydeficiencies as part of the review process and often will request additional testing or facilities atinformation. Notwithstanding the submission of any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
Under the PREA, amendments to the FDCA, an NDA or supplement to an NDA must contain data that are adequate to assess the safety and efficacy of the product candidate for the claimed indications in all relevant pediatric populations and to support dosing and administration for each pediatric population for which the drugproduct is manufactured to confirm compliance with cGMP.safe and effective. The FDA may also refer applicationsgrant deferrals for novel drug products,submission of pediatric data or drug productsfull or partial waivers. The PREA requires a sponsor that present difficult questionsis planning to submit a marketing application for a product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of safetyadministration to submit an initial Pediatric Study Plan, or efficacy,PSP, within sixty days of an end-of-Phase 2 meeting or, if there is no such meeting, as early as practicable before the initiation of the Phase 3 or Phase 2/3 clinical trial. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including trial objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. The FDA and the sponsor must reach an agreement on the PSP. A sponsor can submit amendments to an advisory committee. This advisory committee is typically a panel that includes clinicians and other experts in the relevant indication or subject matter who review and evaluate the NDA and provide a recommendationagreed upon initial PSP at any time if changes to the FDA aspediatric plan need to whether the application should be approved.considered based on data collected from pre-clinical studies, early-phase clinical trials or other clinical development programs.
The testing and approval process requires substantial time, effort and financial resources, and each may take several years to complete. The FDA ismay not bound by the recommendation of an advisory committee, but it generally follows such recommendations.
grant approval on a timely basis, or at all, and we may encounter difficulties or unanticipated costs in our efforts to secure necessary governmental approvals, which could delay or preclude us from marketing its products. After the FDA evaluates thean NDA the clinical sites,and conducts inspections of the manufacturing facilities and, as needed, receives a recommendation fromwhere the advisory committee, it issues eitherinvestigational product and/or its drug substance will be produced, the FDA may issue an approval letter or a CRL. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. A CRL indicates that the review cycle of the application is complete response letter.and the application will not be approved in its present form. A complete response letterCRL generally outlines the deficiencies in the submission and may require substantial additional testing, information or information, in orderclarification for the FDA to reconsider the application. The FDA may delay or refuse approval of an NDA if applicable regulatory criteria are not satisfied, require additional testing or information and/or require post-marketing testing and surveillance to monitor safety or efficacy of a product. If a CRL is issued, the applicant may either resubmit the NDA, addressing all of the deficiencies identified in the letter, or withdraw the application. If and when the deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the marketing application, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in response to an issued CRL in either two toor six months depending on the type of new information included. Even if such data and information are submitted, the FDA may ultimately decide that the NDA does not satisfy the criteria for approval.
If regulatory approval of a product is granted, such approval is limited to the conditions of use (e.g., patient population, indication) described in the application and may entail further limitations on the indicated uses for which such product may be marketed. For example, the FDA Approval Letter
Anmay approve the NDA with a REMS plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA approval letter authorizes commercial marketingdetermines the requirement for a REMS, as well as the specific REMS provisions, on a case-by-case basis. If the FDA concludes a REMS plan is needed, the sponsor of the drug with specific prescribing informationNDA must submit a proposed REMS to obtain approval for specific indications. Moreover,the product. The FDA also may condition approval on, among other things, changes to proposed labeling (e.g., adding contraindications, warnings or precautions) or the development of adequate controls and specifications. Once approved, the FDA may withdraw the product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy including, in certain cases, REMS as described in more detail under the heading, —Certain Other FDA Regulations – Risk Evaluation and Mitigation Strategies and Other Post-Approval Requirements. Once granted, product approvals may be withdrawn if compliance with pre- and post-marketing regulatory standards is not maintained or if problems occur after the product reaches the marketplace. The FDA may require one or more Phase 4 post-market studies and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization and may limit further marketing of the product based on the results of these post-marketing studies. Some types of changes to an approved product, such as adding new indications, manufacturing changes and additional labeling claims, are identified following initial marketing.subject to further testing requirements and separate FDA review and approval. In addition, new government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or prevent regulatory approval of our products under development.
Fast Track, Priority Review, and Breakthrough Therapy Designations
Further, changesA sponsor may seek approval of its product candidate under programs designed to someaccelerate FDA’s review and approval of new drugs that meet certain criteria. Specifically, new drugs are eligible for fast track designation if they are intended to treat a serious or life-threatening condition and demonstrate the potential to address unmet medical needs for the condition. Fast track designation provides increased opportunities for sponsor interactions with the FDA during preclinical and clinical development, in addition to the potential for rolling review once a marketing application is filed, meaning that the FDA may consider for review sections of the NDA on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the application, the FDA agrees to accept the sections and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application. A fast track designated product candidate may also qualify for accelerated approval (described below) or priority review, under which the FDA sets the target date for FDA action on the NDA or biologics license application at six months after the FDA accepts the application for filing.
Priority review is granted when there is evidence that the proposed product would be a significant improvement in the safety or effectiveness of the treatment, diagnosis, or prevention of a serious condition. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting drug reaction, documented enhancement of patient compliance that may lead to improvement in serious outcomes, or evidence of safety and effectiveness in a new subpopulation. If criteria are not met for priority review, the application is subject to the standard FDA review period of 10 months after FDA accepts the application for filing.
In addition, a sponsor may seek FDA designation of its product candidate as a breakthrough therapy if the product candidate is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the therapy may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Breakthrough therapy designation provides all the features of fast track designation in addition to intensive guidance on an efficient development program beginning as early as Phase 1, and FDA organizational commitment to expedited development, including involvement of senior managers and experienced review and regulatory staff in a proactive, collaborative, cross-disciplinary review, where appropriate. A drug designated as breakthrough therapy is also eligible for accelerated approval if the relevant criteria are met.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions establishedfor qualification or decide that the time period for FDA review or approval will not be shortened. Fast track, priority review and breakthrough therapy designations do not change the scientific or medical standards for approval or the quality of evidence necessary to support approval but may expedite the development or approval process.
Accelerated Approval
In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval from the FDA and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The FDA may also grant accelerated approval for such a drug or biologic when it has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on IMM, and that is reasonably likely to predict an effect on IMM or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require that a sponsor of a drug receiving accelerated approval perform post-marketing clinical trials to verify and describe the predicted effect on IMM or other clinical endpoint, and the product may be subject to expedited withdrawal procedures. Drugs granted accelerated approval must meet the same statutory standards for safety and effectiveness as those granted traditional approval.
For the purposes of accelerated approval, a surrogate endpoint is a marker, such as a laboratory measurement, radiographic image, physical sign, or other measure that is thought to predict clinical benefit, but is not itself a measure of clinical benefit. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. An intermediate clinical endpoint is a measurement of a therapeutic effect that is considered reasonably likely to predict the clinical benefit of a drug or biologic, such as an effect on IMM. The FDA has limited experience with accelerated approvals based on intermediate clinical endpoints, but has indicated that such endpoints generally may support accelerated approval when the therapeutic effect measured by the endpoint is not itself a clinical benefit and basis for traditional approval, if there is a basis for concluding that the therapeutic effect is reasonably likely to predict the ultimate long-term clinical benefit of a drug.
The accelerated approval pathway is most often used in settings in which the course of a disease is long and an extended period of time is required to measure the intended clinical benefit of a drug, even if the effect on the surrogate or intermediate clinical endpoint occurs rapidly. For example, accelerated approval has been used extensively in the development and approval of drugs for treatment of a variety of cancers in which the goal of therapy is generally to improve survival or decrease morbidity and the duration of the typical disease course requires lengthy and sometimes large clinical trials to demonstrate a clinical or survival benefit.
The accelerated approval pathway is usually contingent on a sponsor’s agreement to conduct, in a diligent manner, additional post-approval confirmatory studies to verify and describe the product candidate’s clinical benefit. As a result, a product candidate approved application,on this basis is subject to rigorous post-marketing compliance requirements, including changes in indications, labeling,the completion of Phase 4 or manufacturing processespost-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or facilities,to confirm the predicted clinical benefit of the product during post-marketing studies, would allow the FDA to withdraw approval of the product. As part of the Consolidated Appropriations Act for 2023, Congress provided FDA additional statutory authority to mitigate potential risks to patients from continued marketing of ineffective drugs or biologics previously granted accelerated approval. Under the act’s amendments to the FDCA, FDA may require submissionthe sponsor of a product granted accelerated approval to have a confirmatory trial underway prior to approval. The sponsor must also submit progress reports on a confirmatory trial every six months until the trial is complete, and such reports are published on FDA’s website. The amendments also give FDA the option of using expedited procedures to withdraw product approval if the sponsor’s confirmatory trial fails to verify the claimed clinical benefits of the product.
All promotional materials for product candidates being considered and approved under the accelerated approval program are subject to prior review by the FDA.
Patent Term Restoration
Depending upon the timing, duration and specifics of FDA approval of a new NDA or NDA supplement beforeour product candidates, some of our United States patents may be eligible for limited patent term extension under the change can be implemented. An NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses similar procedures and actions in reviewing NDA supplements as it does in reviewing NDAs.
The Hatch-Waxman Amendments
The Drug Price Competition and Patent Term Restoration Act, commonly referred toinformally known as the Hatch-Waxman Amendments, is a 1984 U.S. federal law which established the modern system of generic drug regulation in the U.S.Act. The Hatch-Waxman Amendments were enacted to encourage the manufacture of generic drugs by outlining the process for generic pharmaceutical manufacturers to file an abbreviated new drug application and to provide certain related protections to drug development innovators, namely a new kind of market exclusivity period and the ability to potentially extend patent life by a portion of the time a drug is under regulatory review by the FDA.
Orange Book Listing and Abbreviated New Drug Applications
In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent whose claims cover the applicant’s product. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential generic competitors in support of approval of an ANDA.
An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the listed drug and has been shown through bioequivalence testing to be therapeutically equivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, preclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug and can often be substituted by pharmacists under prescriptions written for the original listed drug.
An ANDA applicant is required to make certain certifications to the FDA concerning any such patents listed in the Orange Book for the approved reference drug intended to confirm that the proposed generic equivalent will not infringe on any intellectual property related to the reference drug, commonly referred to as a Paragraph IV certification. The ANDA process gives the owner of the reference drug an opportunity to assertAct permits a patent infringement claim if it believes its intellectual property rights are being infringed upon following the submission of a Paragraph IV certification.
An ANDA will not be approved until all patents and non-patent exclusivity periods listed in the Orange Book for the reference drug have expired.
Patent Term Restoration and Marketing Exclusivity
Certain of our current and future product candidates, including TSC, may be eligible for patent term restoration and marketing exclusivity under the Hatch-Waxman Amendment.
The Hatch-Waxman Amendments permit restoration of the patent term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. PatentHowever, patent term restoration however, cannot extend the remaining term of a patent beyond a total of 14 years from the product’sproduct candidate’s approval date. The patent term restoration period is generally 50.0%one half of the amount of time between the effective date of an IND and the submission date of an NDA, plus the time between the submission date of anthe NDA and the approval of that application, except that the review period is reduced by any time during which the applicant failed to exercise due diligence. Only one patent applicable to an approved drugproduct candidate is eligible for such anthe extension and the application for the extension must be submittedmade prior to the expiration of the patent. The USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we intend to apply for restorations of patent term for some of our currently owned or licensed patents to add patent life beyond their current expiration date, depending on the expected length of clinical trials and other factors involved in the submission of the relevant NDA.
MarketingPediatric Exclusivity
Pediatric exclusivity is a type of non-patent marketing exclusivity available in the United States and, if granted, it provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity or listed patents. This six-month exclusivity may be granted if an NDA sponsor submits pediatric data that fairly respond to a written request from the FDA for such data. The data do not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the FDA’s request, the additional protection is granted. If reports of requested pediatric studies are submitted to and accepted by the FDA within the statutory time limits, whatever statutory or regulatory periods of exclusivity or patent protection cover the product are extended by six months. This is not a patent term extension, but it effectively extends the regulatory period during which the FDA cannot approve another application. The issuance of a written request does not require the sponsor to undertake the described studies.
Abbreviated NDAs for Generic Drugs
In 1984, with passage of the Hatch-Waxman Act, which established an abbreviated regulatory scheme authorizing the FDA to approve generic drugs based on an innovator or “reference” product, Congress also enacted Section 505(b)(2) of the FDCA, which provides a hybrid pathway combining features of a traditional NDA and a generic drug application. To obtain approval of a generic drug, an applicant must submit an ANDA to the agency. In support of such applications, a generic manufacturer may rely on the preclinical and clinical testing previously conducted for a drug product previously approved under an NDA, known as the RLD.
Specifically, in order for an ANDA to be approved, the FDA must find that the generic version is identical to the RLD with respect to the active ingredients, the route of administration, the dosage form, and the strength of the drug. At the same time, the FDA must also determine that the generic drug is “bioequivalent” to the innovator drug. Under the statute, a generic drug is bioequivalent to an RLD if “the rate and extent of absorption of the drug do not show a significant difference from the rate and extent of absorption of the listed drug.”
Upon approval of an ANDA, the FDA indicates whether the generic product is “therapeutically equivalent” to the RLD in its publication Approved Drug Products with Therapeutic Equivalence Evaluations, also referred to as the Orange Book. Clinicians and pharmacists consider a therapeutic equivalent generic drug to be fully substitutable for the RLD. In addition, by operation of certain state laws and numerous health insurance programs, the FDA’s designation of therapeutic equivalence often results in substitution of the generic drug without the knowledge or consent of either the prescribing clinicians or patient.
In contrast, Section 505(b)(2) permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference. Section 505(b)(2) NDAs may provide an alternate path to FDA approval for new or improved formulations or new uses of previously approved products; for example, an applicant may be seeking approval to market a previously approved drug for new indications or for a new patient population that would require new clinical data to demonstrate safety or effectiveness. A Section 505(b)(2) applicant may eliminate the need to conduct certain preclinical or clinical studies, if it can establish that reliance on studies conducted for a previously-approved product is scientifically appropriate. Unlike the ANDA pathway used by developers of bioequivalent versions of innovator drugs, which does not allow applicants to submit new clinical data other than bioavailability or bioequivalence data, the 505(b)(2) regulatory pathway does not preclude the possibility that a follow-on applicant would need to conduct additional clinical trials or nonclinical studies. The FDA may then approve the new product for all or some of the label indications for which the RLD has been approved, or for any new indication sought by the Section 505(b)(2) applicant, as applicable.
In addition, under the Hatch-Waxman Amendments, the FDA may not approve an ANDA or 505(b)(2) NDA until any applicable period of non-patent exclusivity for the RLD has expired. These market exclusivity provisions under the FDC ActFDCA also can also delay the submission or the approval of certain marketing applications. The FDC ActFDCA provides a period of five years of non-patent data exclusivity for a new drug containing an NCE. For the purposes of this provision, an NCE, is a drug that contains no active moiety that has previously been approved by the FDA in any other NDA. An active moiety is the molecule or ion responsible for the physiological or pharmacological action of the drug substance. In cases where such NCE exclusivity has been granted, an ANDA or 505(b)(2) NDA may not be filed with the FDA until the expiration of five years unless the submission is accompanied by a Paragraph IV certification (described below), in which case the applicant may submit its application four years following the original product approval.
The FDCA also provides for a period of three years of marketing exclusivity for an NDA, 505(b)(2) NDA or supplement to an existing NDA,thereto if one or more new clinical investigations, other than bioavailability or bioequivalence studies, that were conducted by or sponsored byfor the applicant are deemed by the FDA to be essential to the approval of the application, for example investigations relatedapplication. This three-year exclusivity period often protects changes to a previously approved drug product, such as a new indications, dosages,dosage form, route of administration, combination or strengths of an existing drug. Thisindication. The three-year exclusivity covers only the modification for which the drug received approval on the basisconditions of use associated with the new clinical investigations and does not prohibit the FDA from approving ANDAsfollow-on applications for drugs containing the original active agent for the original indication or condition of use. The FDC Actagent. Five-year and three-year exclusivity also provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to obtain approval of an NDA for a new chemical entity, meaning the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule or ion responsible for the action of the drug substance.
During the exclusivity period, the FDA may not accept for review an ANDA or a 505(b)(2) NDA submitted by another company for another drug based on the same active moiety, regardless of whether the drug is intended for the same indication as the original innovator drug or for another indication, where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement to one of the patents listed with the FDA by the innovator NDA holder. Three-year and five-year exclusivity will not delay the submission or approval of a full NDA.traditional NDA filed under Section 505(b)(1) of the FDCA. However, an applicant submitting a fulltraditional NDA would be required to either conduct or obtain a right of reference to all of the nonclinicalpreclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and efficacy.
Certain Other FDA Regulationseffectiveness.
The Orphan Drug Act of 1983Hatch-Waxman Patent Certification and the 30-Month Stay
Under the Orphan Drug Act,Upon approval of an NDA or a supplement thereto, NDA sponsors are required to list with the FDA may grant orphan designation to a drugeach patent with claims that cover the applicant’s product intended to treat a rare disease or condition, whichan approved method of using the product. Each of the patents listed by the NDA sponsor is generally a disease or condition that affects fewer than 200,000 individualspublished in the United States, or more than 200,000 individualsOrange Book. When an ANDA applicant files its application with the FDA, the applicant is required to certify to the FDA concerning any patents listed for the reference product in the United States andOrange Book, except for patents covering methods of use for which therethe ANDA applicant is no reasonable expectationnot seeking approval. To the extent that the cost of developing and makingSection 505(b)(2) NDA applicant is relying on studies conducted for an already approved product, the applicant is required to certify to the FDA concerning any patents listed for the approved product available in the United States for this type of disease or condition will be recovered from sales ofOrange Book to the product. Our lead product candidate, TSC, has been granted Orphan Drug designations bysame extent that an ANDA applicant would.
Specifically, the FDAapplicant must certify with respect to the teach patent that:
Orphan drug designation must be requested before submitting an NDA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. For example, we previously announced that TSC was granted orphan drug designation by the FDA for the treatment of GBM and metastatic brain cancer in July 2011 and in December 2012, respectively. However, orphan drug designation on its own does not convey any advantage in or shorten the duration of the regulatory review and approval process but may result in certain financial and marketing incentives if approved.
| ● | the required patent information has not been filed by the original applicant; |
| ● | the listed patent has expired; |
| ● | the listed patent has not expired, but will expire on a particular date and approval is sought after patent expiration; or |
| ● | the listed patent is invalid, unenforceable or will not be infringed by the manufacture, use or sale of the new product. |
If a product that has orphan designation subsequently receives the first FDA approval for the diseaseParagraph I or condition for which it has such designation, the productII certification is entitled to orphan drug exclusivity, which means thatfiled, the FDA may not approve any other applications to marketmake approval of the same drug forapplication effective immediately upon completion of its review. If a Paragraph III certification is filed, the same indication for seven years fromapproval may be made effective on the patent expiration date specified in the application, although a tentative approval may be issued before that time. If an application contains a Paragraph IV certification, a series of events will be triggered, the outcome of which will determine the effective date of such approval except in limited circumstances, such asof the ANDA or 505(b)(2) application.
If the follow-on applicant has provided a showing of clinical superiorityParagraph IV certification to the product with orphan exclusivityFDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the follow-on application in question has been accepted for filing by meansthe FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of greater effectiveness, greater safety,the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days after the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA or providing505(b)(2) NDA until the earlier of 30 months after the receipt of the Paragraph IV notice, expiration of the patent, or a major contributiondecision in the infringement case that is favorable to patient care,the ANDA or in instances of drug supply issues. Competitors, however, may receive505(b)(2) applicant. Alternatively, if the listed patent holder does not file a patent infringement lawsuit within the required 45-day period, the follow-on applicant’s ANDA or 505(b)(2) NDA will not be subject to the 30-month stay.
Post-Approval Requirements
Following approval of either a differentnew product, the manufacturer and the approved product are subject to pervasive and continuing regulation by the FDA, including, among other things, monitoring and recordkeeping activities, reporting of adverse experiences with the product, product sampling and distribution restrictions, complying with promotion and advertising requirements, which include restrictions on promoting drugs for unapproved uses or patient populations (i.e., “off-label use”) and limitations on industry-sponsored scientific and educational activities. The manufacturer and its products are also subject to similar post-approval requirements by regulatory authorities comparable to FDA in jurisdictions outside of the same indication orUnited States where the same product for a different indication. In the latter case, because health care professionalsproducts are free toapproved. Although physicians may prescribe legally available products for off-label uses, manufacturers may not market or promote such uses. The FDA and other agencies actively enforce the competitor’slaws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. If there are any modifications to the product, couldincluding changes in indications, labeling or manufacturing processes or facilities, the applicant may be used for the orphan indication despite our orphan exclusivity. Orphan drug exclusivity could also block therequired to submit and obtain FDA approval of onea new NDA or a supplement to an NDA, which may require the applicant to develop additional data or conduct additional nonclinical studies and clinical trials. The FDA may also place other conditions on approvals including the requirement for a REMS to assure the safe use of our products for seven years if a competitor obtains approval before we do for the same drugproduct. A REMS could include medication guides, physician communication plans or elements to assure safe use, such as restricted distribution methods, patient registries and same indication, as defined by the FDA, for which we are seekingother risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of products. Product approvals may be withdrawn for non-compliance with regulatory standards or if our product is determined to be contained within the scope of the competitor’s product for the same indication or disease. If we pursue marketing approval for an indication broader than the orphan drug designation we have received, we may not be entitled to orphan drug exclusivity. Orphan drug status in the E.U. has similar, but not identical, requirements and benefits.problems occur following initial marketing.
Expedited DevelopmentFDA regulations require that products be manufactured in specific approved facilities and Review Programs
in accordance with cGMPs. The FDA has a fast track program thatcGMP regulations include requirements relating to organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports and returned or salvaged products. The manufacturing facilities for our product candidates must meet applicable cGMP requirements to the FDA's or comparable foreign regulatory authorities' satisfaction before any product is intendedapproved and our commercial products can be manufactured. We rely, and expect to expedite or facilitate the process for reviewing new drugs that meet certain criteria. Specifically, new drugs are eligible for fast track designation if they are intendedcontinue to treat a serious or life-threatening condition and preclinical or clinical data demonstrate the potential to address unmet medical needsrely, on third parties for the condition. Fast track designation applies to bothproduction of clinical and commercial quantities of our products in accordance with cGMP regulations. These manufacturers must comply with cGMP regulations that require, among other things, quality control and quality assurance, the productmaintenance of records and documentation and the specific indication for which it is being studied. The sponsor can request the FDAobligation to designate the product for fast track statusinvestigate and correct any time before receiving NDA approval, but ideally no later than the pre-NDA meeting. Any product submitted to the FDA for marketing, including under a fast track program, may be eligible fordeviations from cGMP. Manufacturers and other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. Any product is eligible for priority review if it treats a serious or life-threatening condition and, if approved, would provide a significant improvement in safety and effectiveness compared to available therapies. The FDA will attempt to direct additional resources to the evaluation of an application for a new drug designated for priority review in an effort to facilitate the review.
A product may also be eligible for accelerated approval, if it treats a serious or life-threatening condition and generally provides a meaningful advantage over available therapies. In addition, it must demonstrate an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit or on a clinical endpoint that can be measured earlier than IMM that is reasonably likely to predict an effect on IMM or other clinical benefit. As a condition of approval, the FDA may require that a sponsor of a drug receiving accelerated approval perform adequate and well-controlled post-marketing clinical trials. If the FDA concludes that a drug shown to be effective can be safely used only if distribution or use is restricted, it will require such post-marketing restrictions, as it deems necessary to assure safe use of the product. If the FDA determines that the conditions of approval are not being met, the FDA can withdraw its accelerated approval for such drug.
Additionally, a drug may be eligible for designation as a breakthrough therapy if the product is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over currently approved therapies on one or more clinically significant endpoints. The benefits of breakthrough therapy designation include the same benefits as fast track designation, plus intensive guidance from the FDA to ensure an efficient drug development program.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or the time period for FDA review or approval may not be shortened. Furthermore, fast track designation, priority review, accelerated approval, and breakthrough therapy designation do not change the standards for approval, but may expedite the development or approval process.
Rare Pediatric Disease Priority Review Voucher Program
In 2012, Congress authorized the FDA to award priority review vouchers to sponsors of certain rare pediatric disease product applications. This program is designed to encourage development of new drug and biological products for prevention and treatment of certain rare pediatric diseases. Specifically, under this program, a sponsor who receives an approval for a drug or biologic for a “rare pediatric disease” may qualify for a voucher that can be redeemed to receive a priority review of a subsequent marketing application for a different product. The sponsor of a rare pediatric disease drug product receiving a priority review voucher may transfer (including by sale) the voucher to another sponsor. The voucher may be further transferred any number of times before the voucher is used, as long as the sponsor making the transfer has not yet submitted the application. The FDA may also revoke any priority review voucher if the rare pediatric disease drug for which the voucher was awarded is not marketedentities involved in the U.S. within one year following the datemanufacture and distribution of approval.
For purposes of this program, a “rare pediatric disease” is a (a) serious or life-threatening disease in which the serious or life-threatening manifestations primarily affect individuals aged from birth to 18 years, including age groups often called neonates, infants, children, and adolescents; and (b) rare diseases or conditions within the meaning of the Orphan Drug Act. On December 27, 2020, the Rare Pediatric Disease Priority Review Voucher Program was extended. Under the current statutory sunset provisions, after September 30, 2024, FDA may only award a voucher for an approved rare pediatric disease product application if the sponsor has rare pediatric disease designation for the drug, and that designation was granted by September 30, 2024. After September 30, 2026, FDA may not award any Rare Pediatric Disease Priority Review Voucher.
Disclosure of Clinical Trial Information
Sponsors of clinical trials of FDA-regulated productsdrugs are required to register their establishments with the FDA and disclosecertain state agencies and are subject to periodic prescheduled or unannounced inspections by the publicFDA and certain clinical trial information, including information related to the product, patient population, phase of investigation, study sites, investigators,state agencies for compliance with cGMP and other aspectslaws. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain cGMP compliance. Future inspections by the trial design. Sponsors are also obligatedFDA and other regulatory agencies may identify compliance issues at the facilities of our contract manufacturing organizations that may disrupt production or distribution or require substantial resources to discusscorrect. In addition, the resultsdiscovery of their clinical trialsconditions that violate these rules, including failure to conform to cGMPs, could result in enforcement actions, and the discovery of problems with a product after completion. However, disclosureapproval may result in restrictions on a product, manufacturer or holder of the results of these trials can be delayed until the new product or new indication being studied has beenan approved NDA, including voluntary recall and regulatory sanctions as competitors may otherwise use this or other publicly-available information to gain knowledge regarding the progress of development programs and gain a competitive advantage.described below.
Risk Evaluation and Mitigation Strategies and Other Post-Approval Requirements
AsOnce an approval or clearance of a condition of NDA approval,drug is granted, the FDA may requirewithdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS to help ensure that the benefits of the drug’s continued approval outweigh theprogram.
Other potential risks. In determining whether a REMS is necessary, the FDA must consider the size of the population likely to use the drug, the seriousness of the disease or condition to be treated, the expected benefit of the drug, the duration of treatment, the seriousness of known or potential adverse events, and whether the drug is a new molecular entity. If the FDA determines a REMS is necessary, the drug sponsor must agree to the REMS plan at the time of approval. REMS canconsequences include, medication guides, communication plans for healthcare professionals, and ETASU, or elements to assure safe use. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring requirements, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of a drug product.among other things:
| ● | Restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| ● | Fines, warning letters or other enforcement-related letters, or clinical holds on post-approval clinical trials; |
| ● | Refusal of the FDA to approve pending marketing applications or supplements to approved marketing authorizations, or suspension or revocation of product approvals; |
| ● | Product seizure or detention, or refusal to permit the import or export of products; |
| ● | Injunctions or the imposition of civil or criminal penalties; |
| ● | Consent decrees, corporate integrity agreements, debarment, or exclusion from federal health care programs; and/or |
| ● | Mandated modification of promotional materials and labeling and the issuance of corrective information. |
Even if the FDA does not require a REMS, once an NDA is approved, a product will be subject to certain post-approval regulations. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities, and promotional activities involving the internet. Adverse event reporting and the submission of periodic reports are also required following FDA approval of an NDA.
Drug Approval Process and Other Regulations Outside of the U.S.
In addition, to regulations in the U.S., we are and will bedistribution of prescription pharmaceutical products is subject to the regulations of other countries inPDMA, which we conduct any of our clinical trials or engage in commercial sales or otherregulates the distribution of our products, if approved. Whether or not we obtain FDA approvaldrugs and drug samples at the federal level and sets minimum standards for conductthe registration and regulation of a clinical trial ordrug distributors by the states. Both the PDMA and state laws limit the distribution of aprescription pharmaceutical product we must obtain approval fromsamples and impose requirements to ensure accountability in distribution. Most recently, the competent regulatory authorityDSCSA was enacted with the aim of any country or economic areabuilding an electronic system to identify and trace certain prescription drugs distributed in which we would seek to commence a clinical trial or market products. For example, conduct of the TSC COVID Trial at the NIID in Bucharest, Romania, required certain approvals from regulatory authorities in Romania and the E.U. Certain countries outside of the United States haveStates. The DSCSA mandates phased-in and resource-intensive obligations for pharmaceutical manufacturers, wholesale distributors, and dispensers over a process similar10-year period, which culminated in November 2023. Most recently, the FDA announced a one-year stabilization period to November 2024, giving entities subject to the FDA’s IND process which requiresDSCSA additional time to finalize interoperable tracking systems and to ensure supply chain continuity. From time to time, new legislation and regulations may be implemented that could significantly change the submission of a CTA prior to the commencement of human clinical trials. In the E.U., for example, a CTA must be submitted to each country’s national health authority and an independent ethics committee, which operates similar to an IRB under U.S. regulations. Once the CTA is approved in accordance with a country’s requirements, the clinical trial may proceed in the applicable country. The approval process and requirementsstatutory provisions governing the conductapproval, manufacturing and marketing of clinical trials, product licensing, pricing, and reimbursement vary greatly from placeproducts regulated by the FDA. It is impossible to place, andpredict whether further legislative or regulatory changes will be enacted, whether FDA regulations, guidance or interpretations will be changed or what the timeimpact of such changes, if any, may be longer or shorter than that required for FDA approval.
In particular, in the E.U. a company may submit marketing authorization applications (comparable to an NDA submission in the US to the FDA) under either a centralized or decentralized procedure. The centralized procedure, which is compulsory for medicines produced by biotechnology or those medicines intended to treat AIDS, cancer, neurodegenerative disorders, or diabetes and is optional for medicines which are highly innovative, provides for the grant of a single marketing authorization that is valid for all E.U. member states. The decentralized procedure provides for mutual recognition of national approval decisions. Under this decentralized procedure, the holder of a national marketing authorization in any E.U. member state may submit an application to the remaining member states. Within ninety days of receiving the applications and assessments report, each member state must decide whether to recognize approval. If a member state does not recognize the marketing authorization, the disputed points are eventually referred to the European Commission, whose decision is binding on all member states. The E.U. also has procedures similar to those of the FDA pursuant to which a company may obtain marketing exclusivity for a product for up to 11 years and/or orphan drug designation and related exclusivity for up to ten years, as well as other expedited approval pathways available to certain drugs.
In addition, in some non-U.S. jurisdictions, the proposed pricing for a product candidate must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the E.U. provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our product candidates. Historically, product candidates launched in the E.U. do not follow price structures of the U.S. and generally tend to be significantly lower.
Certain Other Legislation and Regulationsbe.
Current HealthcareOther U.S. Health Care Laws and Regulations
Healthcare providers, physicians, and third party payors, including governmental payors such as Medicare and Medicaid, will play a significant roleIf our product candidates are approved in the recommendationUnited States, we will have to comply with various U.S. federal and prescription of any products for which we obtain marketing approval. Our future arrangements with third party payors, healthcare providers,state laws, rules and physicians may expose usregulations pertaining to broadly applicablehealth care fraud and abuse, and other healthcareincluding anti-kickback laws and regulations that may constrainphysician self-referral laws, rules and regulations. Violations of the business or financial arrangementsfraud and relationships through which we market, sell,abuse laws are punishable by criminal and distribute any drugs for which we obtain marketing approval.
civil sanctions, including, in some instances, exclusion from participation in federal and state health care programs, including Medicare and Medicaid. These laws include, without limitation, state and federal anti-kickback, false claims, physician transparency, and patient data privacy and security laws and regulations, and other federal, state, and local regulations and legislation impacting the pharmaceutical and biopharmaceutical industries, including but not limited to those described below.include:
| | •●
| Health Insurance Portability and Accountability Act - HIPAA imposes criminal and civil liability for,The federal AKS prohibits, among other things, persons from knowingly and willfully executingsoliciting, offering, receiving or paying remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made, in whole or in part, under a scheme, or attempting to execute a scheme, to defraud any healthcare benefitfederal health care program including private payors, or falsifying, concealing, or covering up a material fact or making any materially false statements in connection with the delivery of or payment for healthcare benefits, items or services. Similar to the federal Anti-Kickback Statute, asuch as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statuteAKS or specific intent to violate it in order to have committed a violation. HIPAA, as amended by HITECH and their respective implementing regulations, imposes, among other things, specified requirements on covered entities and their business associates relating toIn addition, the privacy and securitygovernment may assert that a claim including items or services resulting from a violation of individually identifiable health information, including mandatory contractual terms and required implementationthe AKS constitutes a false or fraudulent claim for purposes of technical safeguards of such information. HITECH also created new tiers ofthe FCA or federal civil monetarymoney penalties amended HIPAA to makestatute;
|
| ● | The federal civil and criminal penalties directly applicable to business associates, and gave state Attorneys General new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAAfalse claims laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. |
| •
| Affordable Care Act - The Affordable Care Act was enacted in March 2010 and included measures intended to significantly change the way healthcare is financed in the U.S. by both governmental and private insurers which have and may continue to impact the pharmaceutical and biopharmaceutical industries, including expanded Medicare and Medicaid benefits, expansion of healthcare fraud and abusemonetary penalty laws, establishment of the Centers for Medicare & Medicaid Services, annual reporting requirements for manufacturers and distributors. Since its enactment, there have been numerous judicial, administrative, executive, and legislative challenges to certain aspects of the Affordable Care Act, and we expect there will be additional challenges and amendments to the Affordable Care Act in the future. In addition, subsequent legislation, including the Budget Control Act of 2011, American Taxpayer Relief Act of 2012, and Coronavirus Aid, Relief, and Economic Security Act of 2020, has limited and supplemented various provisions of the Affordable Care Act. While we cannot predict what effect further changes to the Affordable Care Act would have on our business, the Affordable Care Act is likely to continue to impact the regulatory regime to which we are subject for the foreseeable future, and we cannot predict the ultimate content, timing, or effect of any healthcare reform legislation or the impact of potential legislation on us.
|
| •
| 21st Century Cures Act - The 21st Century Cures Act, which the U.S. House of Representatives passed in July 2015 and President Obama signed into law in December 2016, provides for a wide range of reforms that may impact our business, such as broadening the types of data required to support drug approval, extending protections from genetic competition, accelerating approval of breakthrough therapies, expanding the orphan drug product and compassionate use programs, and clarifying how manufacturers communicate about their products.
|
| •
| Anti-Kickback Laws - The federal Anti-Kickback Statute makes it illegal for any person, including a prescription drug manufacturer (or a party acting on its behalf), to knowingly and willfully solicit, receive, offer, or pay any remuneration, directly or indirectly, in cash or in kind, that is intended to induce or reward referrals, including the purchase, recommendation, order or prescription of a particular drug or any other good or service, for which payment may be made under a federal healthcare program, such as Medicare or Medicaid. Violations of this law are punishable by up to five years in prison, criminal fines, administrative civil money penalties, and exclusion from participation in federal healthcare programs. In addition, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it.
|
| •
| False Claims Laws - The federal False Claims Act, imposes civil penalties, including through civil whistleblower or qui tam actions, againstwhich prohibit, among other things, individuals or entities (including manufacturers) for, among other things,from knowingly presenting, or causing to be presented, false or fraudulent claims for payment to, or approval by aMedicare, Medicaid, or other federal healthcare programprograms, knowingly making, using or makingcausing to be made or used a false statementrecord or recordstatement material to payment of a false or fraudulent claim or an obligation to pay or transmit money to the federal government, or knowingly concealing or knowingly and improperly avoiding or decreasing or concealing an obligation to pay money to the federal government. TheManufacturers can be held liable under the FCA even when they do not submit claims directly to government may deem manufacturerspayers if they are deemed to have “caused”“cause” the submission of false or fraudulent claims by, for example, providing inaccurate billing or coding informationclaims. The FCA also permits a private individual acting as a “whistleblower” to customers or promoting a product off-label.bring actions on behalf of the federal government alleging violations of the FCA and to share in any monetary recovery;
|
| | •●
| Medicare Prescription Drug, Improvement,HIPAA imposes criminal and Modernization Act - The Medicare Prescription Drug, Improvement, and Modernization Act of 2003 imposes requirements on the distribution and pricing of prescription drugscivil liability for Medicare beneficiaries. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities which will provide coverage of outpatient prescription drugs. Part D plans include both stand-alone prescription drugexecuting a scheme to defraud any health care benefit plans and prescription drug coverage as a supplementprogram or making false statements relating to Medicare Advantage plans, but plan sponsors are not required to pay for all covered Part D drugs, and each drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any reduction in payment that results from this or any similar legislation in the future may result in a similar reduction in payments from non-governmental payors.health care matters;
|
| | •●
| HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, and its implementing regulations, also imposes obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| ● | The federal transparency requirements under the Physician Payments Sunshine Act - The Physician Payments Sunshine Act, enacted as part require manufacturers of the Affordable Care Act,, imposed new annual reporting requirements for certain manufacturers ofFDA-approved drugs, devices, biologics and medical supplies for which payment is available undercovered by Medicare or Medicaid orto report, on an annual basis, to the Children’s Health Insurance Program, for certainCMS information related to payments and “transfersother transfers of value” providedvalue to physicians, (definedcertain advanced non-physician health care practitioners, and teaching hospitals or to include doctors, dentists, optometrists, podiatrists and chiropractors)entities or individuals at the request of, or designated on behalf of, such physicians, non-physician health care practitioners, and teaching hospitals as well as certain ownership and investment interests held by physicians and their immediate family members. Effective January 1, 2022, these reporting obligations will extend to include transfers of value made to certain non-physician providers such as physician assistantsmembers; and nurse practitioners. |
| | •●
| State, Local,Analogous state and Non-U.S. Legislation and Regulations - In addition, to the legislation summarized above, we may also be subject now or in the future to analogous state, local, and non-U.S.foreign laws and regulations, such as state anti-kickback and false claims laws, which may be broader in scopeapply to sales or marketing arrangements and apply regardless of payor. Such laws are enforcedclaims involving health care items or services reimbursed by various state agencies and throughnongovernmental third-party payors, including private actions. For example, some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant federal government compliance guidance, require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers, and restrict marketing practices or require disclosure of marketing expenditures. Certain state and foreign laws also govern the privacy and security of health information in some circumstances and these data privacy and security laws may differ from both HIPAA and each other in significant ways, which would potentially increase our compliance burden.insurers.
|
The majority of states also have statutes or regulations similar to the aforementioned federal laws, some of which are broader in scope and apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines, or the relevant compliance guidance promulgated by the federal government, in addition to requiring drug manufacturers to report information related to payments to physicians and other health care providers or marketing expenditures to the extent that those laws impose requirements that are more stringent than the Physician Payments Sunshine Act. State and foreign laws also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
Due to the breadth of these laws and the narrowness of their exceptions and safe harbors, it is possible that business activities can be subject to challenge under one or more of such laws. The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of healthcare reform.reform, especially in light of the lack of applicable precedent and regulations. Federal and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry.
Ensuring that business arrangements with third parties comply with applicable healthcare laws and regulations is costly and time consuming. If ourbusiness operations are found to be in violation of any of thesethe laws described above or any other relatedapplicable governmental regulations that may apply to us, wea pharmaceutical manufacturer may be subject to significantpenalties, including civil, criminal and administrative penalties, damages, fines, disgorgement, individual imprisonment, disgorgement, exclusion of drugs from governmentgovernmental funded healthcare programs, such as Medicare and Medicaid, contractual damages, reputational harm, diminished profits and future earnings, additional oversight, and reporting obligations and oversight if we become subject to a corporate integrity agreement or similar settlementother agreement to resolve allegations of non-compliance with these laws, and the curtailment or restructuring of ouroperations, any of which could adversely affect a pharmaceutical manufacturer’s ability to operate its business and the results of its operations.
Future Pharmaceutical Coverage, Pricing, and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of products approved by the FDA and other government authorities. Sales of our products, when and if approved for marketing in the United States, will depend, in part, on the extent to which our products will be covered by third-party payors, such as federal, state, and foreign government healthcare programs, commercial insurance and managed healthcare organizations. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the price or reimbursement rate that the payor will pay for the product once coverage is approved. Third-party payors may limit coverage to specific products on an approved list, or formulary, which might not include all of the approved products for a particular indication. In addition, these third-party payors are increasingly reducing reimbursements for medical products, drugs and services. Furthermore, the U.S. government, state legislatures and foreign governments have continued implementing cost containment programs, including price controls, restrictions on coverage and reimbursement and requirements for substitution of generic products. Adoption of price controls and cost containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our net revenue and results. Limited third-party reimbursement for our product candidates or a decision by a third-party payor not to cover our product candidates could reduce physician usage of our products once approved and have a material adverse effect on our sales, results of operations and financial condition.
Healthcare Laws and RegulationsReform
In the United States and some foreign jurisdictions, there have been, a number ofand continue to be, several legislative and regulatory changes and proposed changes regarding the healthcare system and its regulation that could prevent or delay marketing approval of our product and therapeutic candidates, restrict or regulate post-approval activities, and affect ourthe ability to profitably sell any product and therapeutic candidates for which wethat obtain marketing approval. We expectThe FDA’s and other regulatory authorities’ policies may change and additional government regulations may be enacted that further implementationcould prevent, limit or delay regulatory approval of current laws, as well asour product and therapeutic candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we otherwise may have obtained and we may not achieve or sustain profitability, which would adversely affect our business, prospects, financial condition and results of operations.
As previously mentioned, the primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medical products and services, implementing reductions in Medicare and other healthcare reform measuresfunding and applying new payment methodologies. In recent years, the U.S. Congress has considered reductions in Medicare reimbursement levels for medicines and biologics administered by physicians. CMS, the agency that administers the Medicare and Medicaid programs, also has authority to revise reimbursement rates and to implement coverage restrictions for most drugs and biologics. Cost reduction initiatives and changes in coverage implemented through legislation or regulation could decrease utilization of and reimbursement for any approved products we may be adoptedmarket in the future,future. While Medicare regulations apply only to pharmaceutical benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own reimbursement rates. Therefore, any reduction in reimbursement that results from federal legislation or regulation may result in more rigorous coverage criteria anda similar reduction in additional downward pressure on the price that we, or any strategic collaborators, may receive for any approved products. Further,payments from private payors.
In recent years, there has recently been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which havehas resulted in several recent Congressional inquiries and proposed billsand enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products. Notably, the CREATES Act, which became effective on December 20, 2019, addresses concerns articulated by both the FDA and others in the industry that some brand manufacturers have improperly restricted the distribution of their products, including by invoking the existence of a REMS for certain products, to deny generic and biosimilar product developers access to samples of brand products. Because generic and biosimilar product developers need samples to conduct certain comparative testing required by the FDA, some have attributed the inability to timely obtain samples as a cause of delay in the entry of generic and biosimilar products. To remedy this concern, the CREATES Act establishes a private cause of action that permits a generic or biosimilar product developer to sue the brand manufacturer to compel it to furnish the necessary samples on “commercially reasonable, market-based terms.” Whether and how generic and biosimilar product developments will use this new pathway, as well as the likely outcome of any legal challenges to provisions of the CREATES Act, remain highly uncertain and its potential effects on our future commercial products are unknown.
U.S. Environmental, Health,More recently, in August 2022, President Biden signed into the law the IRA. Among other things, the IRA has multiple provisions that may impact the prices of drug products that are both sold into the Medicare program and Safety Laws
We are subjectthroughout the United States. Starting in 2023, a manufacturer of a drug or biological product covered by Medicare Parts B or D must pay a rebate to numerous environmental, health,the federal government if the drug product’s price increases faster than the rate of inflation. This calculation is made on a drug product by drug product basis and safety lawsthe amount of the rebate owed to the federal government is directly dependent on the volume of a drug product that is paid for by Medicare Parts B or D. Additionally, starting in payment year 2026, CMS will negotiate drug prices annually for a select number of single-source Part D drugs without generic or biosimilar competition. CMS will also negotiate drug prices for a select number of Part B drugs starting for payment year 2028. If a drug product is selected by CMS for negotiation, it is expected that the revenue generated from such drug will decrease. CMS has begun to implement these new authorities and regulations. From timeentered into the first set of agreements with pharmaceutical manufacturers to time andconduct price negotiations in October 2023. However, the IRA’s impact on the pharmaceutical industry in the future, our operations may involveUnited States remains uncertain, in part because multiple large pharmaceutical companies and other stakeholders (e.g., the useU.S. Chamber of hazardous and flammable materials, including chemicals and biological materials, and may also produce hazardous waste products. Even if we contract with third partiesCommerce) have initiated federal lawsuits against CMS arguing the program is unconstitutional for the disposala variety of these materials and waste products, we cannot completely eliminate the risk of contamination or injury resulting from these materials. In the event of contamination or injury resulting from the use or disposal of our hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties for failure to comply with such laws and regulations.
In addition, we may incur substantial costs in order to comply with current or future environmental, health, and safety laws and regulations. Current or future environmental laws and regulations may impair our research, development or production efforts. In addition, failure to comply with these laws and regulations may result in substantial fines, penalties orreasons, among other sanctions.complaints. Those lawsuits are currently ongoing.
In addition to the IRA’s drug price negotiation provisions, President Biden’s Executive Order 14087, issued in October 2022, called for the CMS innovation center to prepare and submit a report to the White House on potential payment and delivery modes that would complement to IRA, lower drug costs, and promote access to innovative drugs. In February 2023, CMS published its report which described three potential models focusing on affordability, accessibility and feasibility of implementation for further testing by the CMS Innovation Center. As of February 2024, the CMS Innovation Center continues to test the proposed models and has started to roll out plans for access model testing of certain product types (e.g., cell and gene therapies) by states and manufacturers.
At the state level, individual states are increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. In December 2020, the U.S. Supreme Court held unanimously that federal law does not preempt the states’ ability to regulate PBMs and other members of the healthcare and pharmaceutical supply chain, an important decision that may lead to further and more aggressive efforts by states in this area. The FTC in mid-2022 also launched sweeping investigations into the practices of the PBM industry that could lead to additional federal and state legislative or regulatory proposals targeting such entities’ operations, pharmacy networks, or financial arrangements. Significant efforts to change the PBM industry as it currently exists in the United States may affect the entire pharmaceutical supply chain and the business of other stakeholders, including pharmaceutical developers like us. In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. These measures could reduce the ultimate demand for our products, once approved, or put pressure on our product pricing.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. We expect that additional federal, state, and foreign healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in limited coverage and reimbursement and reduced demand for our products, once approved, or additional pricing pressures.
Public Company StatusRegulation Outside the United States
For countries outside of the United States, the requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country. In all cases, clinical trials must be conducted in accordance with GCP and the other applicable regulatory requirements. To the extent that any of our product candidates, once approved, are sold in a foreign country, we and our collaborators will be subject to applicable foreign laws and regulations, which may include, for instance, post-marketing requirements, including safety surveillance, anti-fraud and abuse laws and implementation of corporate compliance programs and reporting of payments or other transfers of value to healthcare professionals. If we or our collaborators fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension of clinical trials, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions, and criminal prosecution.
For example, to market our future products in the EEA (which is comprised of the 28 Member States of the European Union plus Norway, Iceland and Liechtenstein) and many other foreign jurisdictions, we must obtain separate regulatory approvals. More concretely, in the EEA, medicinal products can only be commercialized after obtaining an MA. There are two types of MAs:
| ● | The Community MA, which is issued by the European Commission through the Centralized Procedure, is based on the opinion of the Committee for Medicinal Products for Human Use of the EMA, and which is valid throughout the entire territory of the EEA. The Centralized Procedure is mandatory for certain types of products, including medicines containing novel active substances to treat neurodegenerative disorders. The Centralized Procedure is optional for products containing a new active substance not yet authorized in the EEA (other than those intended for the treatment of HIV/AIDS, cancer, diabetes, neurodegenerative diseases, auto-immune and other immune dysfunctions, or viral diseases, which must be authorized through the Centralized Procedure), or for products that constitute a significant therapeutic, scientific or technical innovation or which are in the interest of public health in the EU; and |
| ● | National MAs, which are issued by the competent authorities of the Member States of the EEA and only cover their respective territory, are available for products not falling within the mandatory scope of the Centralized Procedure. Where a product has already been authorized for marketing in a Member State of the EEA, this National MA can be recognized in another Member State through the Mutual Recognition Procedure. If the product has not received a National MA in any Member State at the time of application, it can be approved simultaneously in various Member States through the Decentralized Procedure. |
Under the procedures described above, before granting the MA the EMA or the competent authorities of the Member States of the EEA assess the risk-benefit balance of the product on the basis of scientific criteria concerning its quality, safety and efficacy.
In April 2023 the European Commission issued a proposal that will revise and replace the existing general pharmaceutical legislation. If adopted and implemented as currently proposed, these revisions will significantly change several aspects of drug development and approval in the European Union.
Data and Marketing Exclusivity
As a public company, we incur significant legal, accountingIn the EEA, new products authorized for marketing, or reference products, qualify for eight years of data exclusivity and other expenses to comply withan additional two years of market exclusivity upon marketing authorization. The data exclusivity period prevents generic applicants from relying on the reporting requirementsnonclinical and clinical trial data contained in the dossier of the Exchange Act and applicable requirementsreference product when applying for a generic marketing authorization in the European Union during a period of SOX andeight years from the Dodd-Frank Act, as well as rules and regulations subsequently implemented bydate on which the SEC and Nasdaq, includingreference product was first authorized in the establishment and maintenanceEuropean Union. The market exclusivity period prevents a successful generic applicant from commercializing its product in the European Union until 10 years have elapsed from the initial authorization of effective disclosure and financial controls, changesthe reference product in corporate governance practices and required filingthe European Union. The 10-year market exclusivity period can be extended to a maximum of annual, quarterly and current reportseleven years if, during the first eight years of those 10 years, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with respect to our business and operating results. These requirements increase our legal and financial compliance costs and make some activities more time-consuming and costly. In addition, our management and other personnel devote significant time and attention to these public company requirements.existing therapies.
Our People and Human Capital Resources
EmployeesOverview
As of December 31, 2020,2023, we had 12eight employees, all of whom we classify as full-time employees, and one part-time employee, up from 12four employees as of December 31, 2019, and as of March 15, 2021, we have added one additional full-time employee in 2021.2022. We consider the relationship with our employees to be good. We also regularly work with several independentengage outside consultants and other contract organizations to support our businesscontractors with unique expertise and we regularly evaluate additional talent to help support our product manufacturing, development, financial, and other capabilities.skills for specific purposes.
Our success depends upon our ability to attract and retain highly qualified management and technical employees. Talent management is critical to our ability to execute our long-term growth strategy, including providing career growth, on-the-job learning opportunities and competitive compensation. We are committed to an inclusive culture which values equality, opportunity and respect. We are focused on the engagement and empowerment of our employees through the demonstration of these foundational values.
None of our employees are represented by labor unions or covered by collective bargaining agreements.
Company Culture; Diversity and Inclusion
We believe that an inclusive culture is required to understand and develop products that benefit all patients. By embracing differences, we aim to foster an environment of respect and trust in an effort to facilitate creativity, spark passion and help us achieve better outcomes for all those who work at and with Diffusion.CervoMed. We are committed to creating and maintaining a workplace free from discrimination or harassment, including on the basis of any class protected by applicable law, and our recruitment, hiring, development, training, compensation, and advancement practices are based on qualifications, performance, skills, and experience without regard to gender, race, ethnicity or ethnicity. other demographics.
Our management team and employees are expected to exhibit and promote honest, ethical, and respectful conduct in the workplace, including adhering to the standards for appropriate behavior set forth in our code of conduct. An “open door” policy is maintained at all levels of the organization and any form of retaliation against an employee reporting or registering complaints in the event of any violation of our policies is strictly prohibited.
Compensation and Benefits
We operate in a highly competitive environment for human capital, particularly as we seek to attract and retain talent with relevant experience in the biotechnology and pharmaceutical sectors. Therefore, we strive to provide a total rewards package to our employees that is competitive with our peer companies includingand helps meet the needs of our employees. This package currently includes competitive pay,salaries, a cash bonus plan, a comprehensive healthcare benefits package (including an 80.0%a 90% employer contribution to family medical coverage), healthunlimited paid time off, a company-sponsored 401(k) savings accounts with a company contribution, 25 days of paid leave, the 401(k) plan, short-term and long-term disability, and other benefits, as well as remote working and flexible work schedules, and other benefits.
schedules. We also offer every full-time employee the benefit of equity ownership in DiffusionCervoMed through stock option grants.grants with vesting conditions designed to facilitate retention through the opportunity to benefit financially from our growth and profitability, as they generally vest over a three- or four-year period. We believe these grants bothalso help promote alignment between our employees and our stockholders and provide retention benefits, as the awards generally vest over a three-year period.stockholders.
We do not have any employees that are represented by a labor union or that have entered into a collective bargaining agreement with the Company.
Employee Engagement, Safety Wellness, and Our Response to COVID-19Wellness
At Diffusion,CervoMed, we believe that health matters to everyone and that the success of our business is fundamentally connected to the physical and mental well-being of our people. Accordingly, the safety health, and wellness of our employees is one of our top priorities. We are committed to developing and fostering a work environment that is safe, professional, and promotes teamwork, diversity, and trust in order to afford all of our employees the opportunity to contribute to the best of their abilities.
During 2020, in response In addition to the COVID-19 pandemic,benefits package described above, in recent years, we tookhave taken certain measures and responded to changes in our operational needs, including actions designed to providefurther promote a safe work environment for our employees. These actions includedemployees, such as investing in technology solutions to support increased work-from-home capabilities shifting work schedulesand moving to reduce the number of people present in our offices, requiring mask wearing and social distancing, making hand sanitizer readily available, and other measures intended to comply with health and safety protocols as required by federal, state, and local governmental agencies, as well as guidance from the U.S. Centers for Disease Control and Prevention and similar public health authorities.an unlimited paid leave policy.
Employee Development and Training
We believe our people are among our most important assets. We focus on attracting, retaining, and cultivating talented individuals and believe in investing in our people to help them grow. EmployeesOur employees are encouraged to attend scientific, clinical, technological, and other relevant meetings and conferences and we strive to provide employees access to a broad set of internal resources intended to help them be successful, including a variety of training and educational materials. During 2021, we intend to implementWe have also implemented a new, comprehensive employee evaluation program tied to the achievement of individual, team, and company goals to help further support, retain, and develop our people and further promote alignment of interests between our employees future target customers, and our stockholders.
Our Directors and Executive Officers
The table below sets forth, as of March 28, 2024, certain information set forth in concerning our current directors.
Name | | Age | | Director Since |
Joshua S. Boger, Ph.D. (Chair) | | 72 | | 2024 |
John Alam, M.D. | | 62 | | 2023 |
Robert J. Cobuzzi, Jr., Ph.D. | | 58 | | 2023 |
Sylvie Grégoire, PharmD. | | 62 | | 2023 |
Jane Hollingsworth, J.D. | | 65 | | 2020 |
Jeff Poulton | | 56 | | 2023 |
Marwan Sabbagh, M.D. | | 58 | | 2023 |
Frank Zavrl | | 58 | | 2023 |
Part III Our Executive Officers— Item 10 — Directors, Officers, and Corporate Governance of this Annual Report is incorporated herein by reference.
The table below sets forth, as of March 28, 2024, certain information concerning our current executive officers.
Name | | Age | | Position |
John Alam, M.D. | | 62 | | President and Chief Executive Officer (Principal Executive Officer) |
William Tanner, Ph.D. | | 65 | | Chief Financial Officer |
Robert J. Cobuzzi, Jr., Ph.D. | | 59 | | Chief Operating Officer |
Kelly Blackburn, M.H.A. | | 60 | | Senior Vice President, Clinical Development |
William Elder | | 41 | | General Counsel and Corporate Secretary (Acting Principal Financial Officer) |
Our CompanyScientific Advisory Board
We have assembled a highly qualified scientific advisory board comprised of thought leaders in the fields of cell biology, intracellular signal transduction, neurotherapeutics, and translational neuroscience.
Name | | Affiliated Entity |
Ole Isacson, Dr.Med.Sci. | | Professor of Neurology at Harvard Medical School, Founding Director of the Neuroregeneration Research Institute at McLean Hospital |
Lewis Cantley, Ph.D. | | Professor of Cell Biology at Harvard Medical School. Prior to this appointment, he was the Margaret and Herman Sokol Professor and Meyer Director of the Sandra and Edward Meyer Cancer Center at Weill Cornell Medical College/Ronald P. Stanton Clinical Cancer Program at New York Presbyterian Hospital (2012-22) |
Jeffrey Cummings, M.D., Sc.D. | | Joy Chambers-Grundy Professor of Brain Science and the Director of the Chambers-Grundy Center for Transformative Neuroscience at the UNLV School of Integrated Health Sciences. Prior to UNLV, Dr. Cummings served as founding director of the Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas, and as director of the Mary S. Easton Center for Alzheimer’s Disease Research, and director of the Deane F. Johnson Center for Neurotherapeutics, both at UCLA |
Heidi McBride, Ph.D. | | Canada Research Chair in Mitochondrial Cell Biology, Professor in the Department of Neurology and Neurosurgery at McGill University |
Corporate Information
Corporate Information andOur History
We were originally incorporated under the laws of the State of Nevada on January 10, 1995 and reincorporated under the laws of the State of Delaware on June 18, 2015 under the name, “RestorGenex Corporation.”2015. On January 8, 2016,August 16, 2023, we completed the merger of our wholly owned subsidiaryMerger Sub with and into Diffusion LLC,EIP, which was treated as a "reverse acquisition"recapitalization" under U.S. GAAP pursuant to which Diffusion LLC'sEIP’s historical results of operations replaced the Company's for all periods prior to the merger. Immediately following the closing of the merger, we changed our name from "RestorGenex Corporation" to "Diffusion Pharmaceuticals Inc." to "CervoMed Inc."
Where to Find Us
Our principal corporate office is located at 1317 Carlton Avenue,20 Park Plaza, Suite 200, Charlottesville, Virginia 22902,424, Boston, Massachusetts 02116, and our telephone number is (434) 220-0718.
(617) 744-4400. Our website, www.diffusionpharma.com,www.cervomed.com, including the Investor Relations section, investors.diffusionpharma.com, and our social media channels – Facebook (www.facebook.com/diffusionpharmaceuticalsinc/), Twitter (www.twitter.com/diffusionpharma) and LinkedIn (https://www.linkedin.com/company/diffusion-pharmaceuticals/) -- containir.cervomed.com, contains a significant amount of information about the Company. We encourage investors to visit these websites and social media channels as information is frequently updated and new information is shared.
However, the information included on our website and available through our social media channels is not incorporated by reference into, and should not be considered part of, this Annual Report or any other filings we make with the SEC.
Other Available Information
We make available on or through our website certain reports that we file with or furnish to the SEC in accordance with Exchange Act. These include our Annual Reports on Form 10-K, our Quarterly Reports on Form 10-Q, and our Current Reports on Form 8-K, as well as any amendments to those reports, filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act. We make this information available free of charge as soon as reasonably practicable after we electronically file the information with, or furnish it to, the SEC. The SEC also maintains a website, www.sec.gov, that contains reports, proxy and information statements, and other information regarding the Company and other issuers that file electronically with the SEC. We also make available, free of charge and through our website, the charters of the committees of the Board, our Corporate Governance Guidelines, and our Code of Business Conduct and Ethics.
Investing in our common stocksecurities involves a high degree of risk. Set forth below are certain material risks and uncertainties known to us that could adversely effectaffect our business, financial condition, or results of operations or could cause our actual results to differ materially from our expectations expressed elsewhere in this Annual Report.our filings with the SEC and other public statements. The occurrence of the events contemplated by one or more of the factors we describe below could cause the market price of our common stocksecurities to decline, resulting in the loss of all or part of any investment in our common stock. Furthermore, other risks that are currently unknown to us or that we currently believe to be immaterial may also, nevertheless, adversely effectaffect our business, financial condition, or results of operations in a way that is material.
Before investing in our common stock, youYou should carefully consider these risks and uncertainties,the risk factors set forth below as may updated by our subsequent filings under the Exchange Act together with all the other information in this Annual Report, including our consolidated financial statements and the related notes included in Part II, Item 8 – Financial Statements and Supplementary Data of this Annual Report and the information includedset forth in Part II, —Item 7 —7A -- Management’s Discussion and Analysis of Financial Condition and Results of Operations, of this Annual Report, and the information incorporated herein by reference.
Risks Related to the Development, Regulatory Approval, and Commercialization of TSC and Our Other Product Candidates
The success of Diffusion is dependent on the successful development, regulatory approval, and, ultimately, commercialization of TSC andas well as in our other product candidates. However,filings with the drug development process is expensive, time-consuming,SEC, before making any investment decisions. Furthermore, the risks and uncertain. Our efforts to develop, obtain regulatory approval for, and commercialize TSC or any other product candidate could fail at any stage of the development process for a variety of reasons, including the possibility that TSC fails to adequately demonstrate efficacy or proof of concept in the TSC Oxygenation Trials. Furthermore, because the results of preclinical studies and early-stage clinical trials are not necessarily predictive of future results, even if we are able to advance TSC or another product candidate into additional clinical trials – for example, our Planned Phase 2 Hypoxia-related Indication Trial we intend to commence assuming success in the TSC Oxygenation Trials – we may not continue to experience favorable results.
The success of Diffusion, including our ability to finance our operations and generate revenue in the future, will depend primarily on the successful development, regulatory approval, and, ultimately, commercialization of our product candidates. Currently, the majority of our product development resources are dedicated to our lead product candidate, TSC. We are also in early-stage development of our second product candidate, DFN-529,uncertainties described below and in the future, we may also seekother information mentioned above are not the only ones the Company faces. Additional risks and uncertainties not presently known to developthe Company or commercialize additional product candidates, including product candidates that we may in-licensecurrently believe to be immaterial could, nevertheless, adversely affect the Company’s business, operating results and financial condition, as well as adversely affect the value of an investment in the Company’s securities, and the occurrence of any of these risks might cause you to lose all or acquire to supplement our internally developed portfolio.part of your investment.
The drug development process is very expensive, time-consuming, difficult to design and implement, and its outcome is inherently uncertain. Most product candidates that commence clinical trials are never approved by regulatory authorities for commercialization and success in early-stage clinical trials does not ensure that later clinical trials will demonstrate the efficacy and safetySummary of an investigational drug in a manner adequate to support regulatory approval. Countless other companies, including many with greater resources and experience, have failed or suffered significant setbacks attempting to navigate the drug development process, including failed attempts to develop treatments for hypoxia and indications on the hypoxia-continuum that we may ultimately choose to target with TSC, and there can be no assurance that we will have success where others have failed.Risk Factors
Our product candidates, including TSC, remain in early stages of the development process and we expect that the additional clinical trials necessary to support an NDA will take several years to complete. We do not know whether the clinical trials we may conduct will demonstrate adequate efficacy and safety or otherwise provide adequate information to result in regulatory approval to market any of our product candidates in any particular jurisdiction. Furthermore, the timeline for our clinical trials may be delayed in the future for a variety of reason, including delays related to regulatory and IRB review and approval, slower than anticipated rates of enrollment in or early withdrawals from the trial, third party performance issues beyond our control including any CRO engaged in the conduct of the trial, discovery of series or unexpected toxicities or side effects, or a lack of effectiveness.
Whether we are able to successfully develop TSC or any of our other product candidates will depend on a large number of factors, including the following:
our ability to complete our planned and future clinical trials in a timely manner and, with respect to any future trials beyond the TSC Oxygenation Trials and our Planned Phase 2 Hypoxia-related Indication Trial we intend to commence assuming success in the TSC Oxygenation Trials, our ability to fund them;
| ● | The Company is a clinical stage biopharmaceutical company and has incurred significant losses since its inception. The Company expects its net losses to continue for the foreseeable future. The Company is not currently profitable and may never achieve or sustain profitability. The Company is unable to predict the extent of future losses or when it might become profitable, if ever. |
| ● | The Company will require additional capital to fund its operations. If the Company fails to obtain necessary financing on acceptable terms, or at all, it may not be able to complete the development and commercialization of neflamapimod. |
| ● | The Company currently does not have, and may never have, any products that generate significant revenues. |
| ● | The Company is heavily dependent on the success of its lead product candidate, neflamapimod, which is still under clinical development. If neflamapimod does not receive regulatory approval or is not successfully commercialized, the Company’s business will be materially harmed. |
| ● | The development and commercialization of drug products is subject to extensive regulation, and the regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time-consuming, and inherently unpredictable. There is no guarantee that the Company’s planned clinical trials for neflamapimod to treat patients with DLB, or in any other indications that the Company may pursue, will be successful. If the Company is ultimately unable to obtain regulatory approval for neflamapimod on a timely basis, or at all, its business will be substantially harmed. |
| ● | Clinical drug development involves a lengthy and expensive process, with an uncertain outcome. The Company may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of neflamapimod or any other product candidates the Company may develop or acquire. |
| ● | The Company has concentrated its research and development efforts on the treatment of DLB, a disease that has seen limited success in drug development. The ability to successfully develop drugs for DLB and other age-related neurologic disorders is extremely difficult and is subject to a number of unique challenges. In addition, its rationale for neflamapimod in the treatment of DLB is based on a scientific understanding of the disease that may be wrong. |
| ● | Enrollment and retention of participants in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible by multiple factors outside the Company’s control. |
| ● | Results of preclinical studies and early clinical trials may not be indicative of results obtained in later trials. In addition, preliminary, topline and interim data from the Company’s clinical trials that the Company may announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data. |
| ● | If the Company does not adequately protect its proprietary rights, the Company may not be able to compete effectively. |
| ● | The Company has no history of commercializing pharmaceutical products, which may make it difficult to evaluate the prospects for its future viability. |
| ● | Even if neflamapimod or any other product candidate the Company develops receives marketing approval, it may fail to achieve the level of acceptance necessary for commercial success. |
| ● | The Company’s future success depends in large part on the Company’s ability to retain its key employees, as well as its ability to attract, train and motivate additional qualified personnel. The Company may also encounter difficulties in managing its growth, which could disrupt its operations. |
| ● | The Company has identified material weaknesses in its internal control over financial reporting which, if not corrected, could affect the reliability of the Company’s financial statements and have other adverse consequences. The Company may identify additional material weaknesses in its internal controls over financial reporting which it may not be able to remedy in a timely manner. If the Company fails to maintain proper and effective internal controls, its ability to produce accurate financial statements on a timely basis could be impaired. |
our abilityRisks Related to the Company’s Limited Operating History, Financial Condition and Need for Additional Capital
The Company is a clinical stage biopharmaceutical company and has incurred significant losses since its inception. The Company expects its net losses to continue for the foreseeable future. The Company is not currently profitable and may never achieve or sustain profitability. The Company is unable to predict the extent of future losses or when it might become profitable, if ever.
Investment in pharmaceutical product development is highly speculative because it entails substantial upfront capital expenditures and significant risk that any potential product candidate will fail to demonstrate safety and efficacy to the satisfaction of the FDA and similar foreign regulatory authorities, and whether we are required by any such body to conduct additional clinical trials to support approval;
the receipt of necessary regulatory approvals, including acceptance of our proposed indications and primary endpoint assessments, marketing approvals, and labeling claims;
a continuedadequate effect or an acceptable safety profile, duringgain regulatory approval, and become commercially viable. The Company has incurred net losses since its inception, and as of December 31, 2023, it had an accumulated deficit of approximately $54.4 million. The Company expects to incur net losses for the foreseeable future as it incurs significant clinical development costs related to the advancement of neflamapimod. The Company has not commercialized any products and has never generated revenue from neflamapimod or any other product. In order to obtain revenues from any product candidate, the Company must succeed, either alone or in collaboration with others, in developing, obtaining regulatory approval for, and manufacturing and marketing drugs with significant market potential. The Company may never succeed in these activities and may never generate revenues that are significant enough to achieve profitability.
The Company expects to incur significant additional operating losses for at least the next several years as it advances neflamapimod through clinical development, conducts clinical trials, seeks regulatory approval and commercializes neflamapimod, if it is ultimately approved for marketing. The costs of advancing product candidates into each successive clinical phase of the clinical development process tend to increase substantially. Therefore, the total costs to advance neflamapimod to marketing approval in even a single jurisdiction will be substantial. Due to the numerous risks and uncertainties associated with pharmaceutical product development, the Company is unable to accurately predict the timing or amount of increased expenses, or when or if it will be able to begin generating revenue from the commercialization of neflamapimod, let alone achieve or maintain profitability.
The amount of the Company’s future net losses will depend, in part, on the rate of future growth of its expenses, if and when neflamapimod is approved for marketing in various jurisdictions and its ability to generate revenues from any drug candidate that may ultimately be approved. If the Company is unable to develop and commercialize one or more product candidates, either alone or through collaborations, or if revenues from any product that receives marketing approval are insufficient, it will not achieve profitability. Even if the Company does achieve profitability, it may not be able to sustain it, which could materially and adversely affect its business.
The Company will require additional capital to fund its operations. If the Company fails to obtain necessary financing on acceptable terms, or at all, it may not be able to complete the development and following approval, including the prevalence, duration and severitycommercialization of potential side effects experienced; andneflamapimod.
our ability
The Company expects to commercialize successfully, including scaling our manufacturing capabilities,spend substantial amounts to complete the development of, sales and marketing capabilities internally or through a third party, acceptance by physicians and patients of the benefits, safety and efficacy of our treatments.
Any of these factors, many of which are beyond our control, could result in significant delays or an inability to develop, obtainseek regulatory approvals for, and commercialize neflamapimod, if it is ultimately approved for marketing. These expenditures will include costs related to the RewinD-LB Trial and costs associated with its license agreement with Vertex, under which the Company is obligated to make certain payments in connection with the achievement of specified events.
Until such time, if ever, that the Company can generate sufficient product revenue and achieve profitability, it expects to seek to finance future cash needs through equity or debt financings and/or corporate collaboration, licensing arrangements and grants. Based upon the Company’s current operating plan, the Company believes that the Company’s cash and cash equivalents as of December 31, 2023, will not be sufficient to enable the Company to fund its operating expenses and capital expenditure requirements for a period of at least 12 months following the issuance of the financial statements included elsewhere in this Annual Report without an additional equity or debt financing. On March 28, 2024, the Company entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The aggregate upfront gross proceeds from the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and additional gross proceeds of up to approximately $99.4 million may be received if the Series A Warrants are exercised in full for cash. The foregoing estimate does not reflect the Company’s expected receipt of proceeds from the 2024 Private Placement.
The Company’s estimates and expectations regarding its cash runway are based on assumptions that may prove to be incorrect, and changing circumstances could cause it to consume capital faster or in different ways than the Company currently expects. For example, the RewinD-LB Trial may be more expensive, time-consuming, or difficult to implement than the Company currently anticipates. Because the length of time and activities associated with the successful development of neflamapimod are highly uncertain, the Company is unable to estimate the actual funds it will require to complete research and development and ultimately commercialize ourits drug candidate for one or more indications.
The Company’s future capital requirements will depend on, and could increase significantly as a result of, many factors, including:
| ● | the enrollment, progress, timing, costs and results of the RewinD-LB Trial and any future phase 3 trial evaluating neflamapimod in DLB, as well as if and when it pursues additional development plans for neflamapimod in other disease indications, such as recovery after anterior circulation ischemic stroke or EOAD; |
| ● | the outcome, timing and cost of meeting regulatory requirements established by the FDA and other comparable foreign regulatory authorities; |
| ● | its ability to reach certain milestone events set forth in its collaboration agreements and the timing of such achievements, triggering obligations to make applicable payments; |
| ● | the hiring of additional clinical, scientific and commercial personnel to pursue the Company’s development plans, as well the increased costs of internal and external resources as to support the Company’s operations as a public reporting company; |
| ● | the cost and timing of securing manufacturing arrangements for clinical or commercial production; |
| ● | the cost of establishing, either internally or in collaboration with others, sales, marketing and distribution capabilities to commercialize neflamapimod, if approved; |
| ● | the cost of filing, prosecuting, enforcing, and defending its patent claims and other intellectual property rights, including defending against any patent infringement actions brought by third parties against the Company; |
| ● | the ability to receive additional non-dilutive funding, including the Company’s pending request for additional funding under the NIA Grant and other grants from organizations and foundations; |
| ● | the Company’s ability to establish strategic collaborations, licensing or other arrangements with other parties on favorable terms, if at all; and |
| ● | the extent to which the Company may in-license or acquire other product candidates or technologies. |
The Company may raise additional capital in the future through a variety of sources, including public or private equity offerings, debt financings, grant funding, or strategic collaborations and licensing arrangements. However, adequate additional financing may not be available to the Company on acceptable terms, or at all. The Company’s failure to raise capital as and when needed would have a negative effect on its financial condition and its ability to pursue its business strategy. If the Company is unable to secure additional capital in sufficient amounts or on terms acceptable to the Company, it may have to delay, scale back or discontinue its development or commercialization activities for neflamapimod.
Further, to the extent that the Company raises additional capital through the sale of common stock or securities convertible or exchangeable into common stock, current stockholder’s ownership interest in the Company will be diluted. In addition, any debt financing may subject the Company to fixed payment obligations and covenants limiting or restricting its ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If the Company raises additional capital through collaborations, strategic alliances or licensing arrangements with third parties, the Company may have to relinquish certain valuable intellectual property or other rights to its product candidates, technologies, future revenue streams or research programs or grant licenses on terms that may not be favorable to it. Even if the Company were to obtain sufficient funding, there can be no assurance that it will be available on terms acceptable to the Company or its stockholders.
The Company currently does not have, and we may ultimatelynever have, any products that generate significant revenues.
The Company is a clinical-stage biopharmaceutical company focused on developing treatments for age-related neurologic disorders, currently has no products that are approved for commercial sale, and it is possible it may never be able to receive regulatory approvaldevelop a marketable product. To date, the Company has not generated any revenues from its lead product candidate, neflamapimod, or generate revenue from the sale of TSC or any other product candidate.
A number of companies in the pharmaceutical and biopharmaceutical industry have suffered significant setbacks in later-stage Phase 3 clinical development even after promising results in earlier preclinical studies or clinical trials. If later-stage clinical trials do not produce favorable results for TSC The Company cannot guarantee that neflamapimod, or any other product candidate that it may develop or we are unable to completeacquire in the necessary clinical trials for any reason (includingfuture, will ever become a lack of funding), our ability to achieve regulatory approval or successfully commercialize may be compromised. At any time, we may decide or be forced by circumstance to delay or discontinue the development or commercialization of TSC or any of our other product candidates, including as a result of unfavorable results in later-stage clinical trials, changes in our internal product, technology or indication focus, the appearance of new technologies that make our product candidate obsolete, competition from a competing product, or changes in (or failure to comply with) applicable regulatory requirements. If we decide or are forced to terminate the TSC development program or any future program in which we have invested significant resources, we will not receive any return on our investment despite the allocation of significant resources, we may not be able to execute on our business plan effectively, and our business, financial condition, or results of operations may be materially and adversely effected.marketable product.
Even if we are able to successfully complete the clinical trials and over development activities necessary to submit an NDA to the FDA or an application for marketing approval to an equivalent non-U.S. regulatory authority, we may be unable to obtain regulatory approval for TSC, DFN-529, or any other product candidates we may attempt to develop in future, for the indications for which we initially seek approval or at all. The FDA and similar non-U.S. regulatory authorities have significant discretion in the approval process, including the ability to delay, limit, or deny approval of product candidates. The delay, limitation, or denial of regulatory approval for any of our product candidates, and TSC in particular, would limit or restrict altogether our ability to commercialize the product and generate revenue, which could materially and adversely impact our business, financial condition, or results of operations.
We currently have no products approved for sale, and we may never obtain regulatory approval to commercialize TSC, DFN-529, or any other product candidates we may attempt to develop in the future. The research, testing, manufacturing, safety surveillance, efficacy, quality control, recordkeeping, labeling, packaging, storage, approval, sale, marketing and distribution import, export, and reporting of safety and other post-market information related to drug products are subject to extensive regulation byin the U.S. and in other countries. Before the FDA and other regulatory authorities in the United StatesEuropean Union and abroad, which often differ from countryelsewhere will approve neflamapimod (or any other drug candidate) for commercialization, the Company must demonstrate that it satisfies rigorous standards of safety and efficacy for each of its intended uses. If approved, in order to country. We willcompete effectively in the commercial marketplace, drugs must be easy to administer, cost-effective and economical to manufacture on a commercial scale. The Company may not achieve any of these objectives.
The Company initiated its RewinD-LB Trial in the second quarter of 2023 and anticipates completing enrollment in the study in the second quarter of 2024. The Company cannot be permitted to market TSCcertain that the RewinD-LB Trial or any future clinical development of our other current product candidatesneflamapimod will be successful, or that it will receive the regulatory approvals required to commercialize neflamapimod for any intended use, or that any future research and drug discovery programs undertaken by the Company will yield a drug candidate suitable for investigation through clinical trials. Even if the Company is able to successfully develop neflamapimod through approval and commercialization, any revenues from sales of the drug may not materialize for several years, if at all.
The Company has a history of operating losses and expect to continue to incur losses in the foreseeable future, which raises substantial doubt about its ability to continue as a going concern.
As discussed further in Note 2 to the Company’s consolidated financial statements included elsewhere in this Annual Report, the Company has a history of operating losses and expects to continue to incur losses in the foreseeable future, which raises substantial doubt regarding the Company’s ability to continue as a going concern. As described in further detail above, the Company currently has no sources of revenue and its ability to continue as a going concern is dependent on its ability to raise capital to fund operations and future business plans. Additionally, volatility in the capital markets and general economic and geopolitical conditions in the U.S. until we receive approval of an NDA or other applicable regulatory filing fromand globally may be a significant obstacle to raising the FDA,required funds as and we willwhen needed. The Company’s consolidated financial statements do not include any adjustments that might be permittednecessary should the Company be unable to market TSC or any of our other current product candidates in any non-U.S. countries until we receivecontinue as a going concern. If the requisite approval from the applicable regulatory authorities.
To gain approval to market a new drug, such as TSC, the FDA and similar non-U.S. regulatory authorities require the submission of an NDA (or similar application) that contains preclinical and clinical data adequately demonstrating the safety, purity, potency, efficacy, and compliant manufacturing of the productgoing concern basis were not appropriate for the intended indication. The FDA and their non-U.S. counterparts have substantial discretionthese financial statements, adjustments would be necessary in the drug approval process, includingcarrying value of assets and liabilities, the abilityreported expenses and the balance sheet classifications used. If the Company is unable to delay, limit,continue as a going concern, its stockholders could suffer the loss of all or deny approvala substantial portion of applications for many reasons, including:
deemed issues with the design or execution of one or more clinical trials;
deemed deficienciestheir investment in the formulation, quality control, labeling, or specifications of the product candidate;us.
deemed issues in our manufacturing processes or in the controls or facilities of third-party manufacturers or testing labs with which we contract;
The RewinD-LB Trial is funded by a determinationnon-dilutive grant that is subject to certain conditions for funding in subsequent years.
The Company’s RewinD-LB Trial is funded by a grant from the NIA, the funds from which will be disbursed over the course of the study as costs are incurred. The Company’s receipt of the funds awarded to support future year costs are subject to both the availability of funds (i.e., the NIA is funded by Congress in subsequent fiscal years) and the Company’s demonstration of progress in the project that is in line with the timelines provided in the grant. If such funds are no longer available, including due to a government shutdown that prohibits the disbursal of such funds, or the Company fails to demonstrate such progress, the Company’s ability to continue its clinical programs may be impaired and delayed, and the Company may otherwise need to seek additional financing. For example, the Company was granted access to $7.3 million under the NIA Grant in February 2024, 90% of the full amount of the second year of funding provided for in the NIA Grant, due to current NIA policy as a result of the U.S. government currently being funded on the basis of a continuing resolution. The timing of the Company’s receipt of the remaining 10% of the grant, or $0.8 million, of current year funding is dependent upon and subject to U.S. congressional approval of a final appropriations bill.
In addition, in December 2023, we submitted a request for supplemental funds in the amount of $4.0 million, of which, if approved, $3.9 million would be received in the current year and the remainder would be received in next the funding year. The request for supplemental funds was initially reviewed by the NIA in January 2024 but, due to the NIA currently working under the Continuing Resolution, completion of the review was delayed and the request is currently scheduled to be reviewed for approval in May 2024. We currently expect to receive the remaining 10%, or $0.8 million, of the previously approved year 2 funding upon U.S. congressional approval of a final appropriations bill, the supplemental amount of $4.0 million following NIA review of our supplement request, and the year 3 funding of $6.2 million in February 2025. However, there can be no guarantee that the dataNIA will approve this supplement request and that any such amounts will be received. If the Company is unable to secure additional capital through approval of the supplemental request or other means, it may have to delay, scale back or discontinue its development or commercialization activities for neflamapimod.
The Company could be subject to audit and repayment of the NIA Grant.
In connection with the NIA Grant, the Company may be subject to routine audits by certain government agencies. As part of an audit, these agencies may review the Company’s performance, cost structures and compliance with applicable laws, regulations, policies and standards and the terms and conditions of the applicable NIA Grant. If any of the Company’s expenditures are found to be unallowable or allocated improperly or if the Company has otherwise violated terms of the NIA Grant, the expenditures may not be reimbursed and/or it may be required to repay funds already disbursed. Any such audit may result in a material adjustment to the Company’s results of operations and financial condition and harm the Company’s ability to operate in accordance with its business plan.
The Company may be required to make significant payments to Vertex in connection with the Company’s license agreement.
Pursuant to the Vertex Agreement, the Company previously acquired an exclusive license to develop and commercialize neflamapimod for the diagnosis, treatment, and prevention of AD and other CNS disorders. Under the Vertex Agreement, the Company is subject to significant potential future obligations, including payment of development milestones and royalties on net product sales, as well as other material obligations. The Vertex Agreement sets forth specific regulatory and product approval events and the related payments that the Company would be obligated to make to Vertex, if and when such events occur.
Among other obligations, the Vertex Agreement provides that the Company will make royalty payments to Vertex in the event aggregate net sales for a commercialized licensed product meet specified thresholds, subject to adjustment in the event of certain events, such as the absence of a valid patent claim or if fees are due to a third party for a license necessary for the development, manufacture, sale or use of a licensed product. Such royalties will be on a sliding scale as a percentage of net sales, depending on the amount of net sales in the applicable years. The Company is also obligated to make a milestone payment to Vertex upon net sales reaching a certain specified amount in any 12-month period.
The first expected milestone events concern filing of an NDA with the FDA for marketing approval of a licensed product in the U.S., or a similar filing for a non-U.S. major market. Thus, although the Company does not expect any milestone or royalty payments to be due until such time, these potential obligations represent significant cash amounts that it may ultimately be obligated to pay. The Company cannot guarantee that it will have sufficient funds available to meet its obligations if and when these payments become due. The obligation to pay some or all of these milestone and royalty amounts may materially harm the Company’s development efforts, as well as its overall financial condition.
The Company may expend its limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
The Company intends to focus its limited financial and other resources on developing neflamapimod and future product candidates for specific indications that the Company identifies as most likely to succeed, in terms of both regulatory approval and commercialization. As a result, the Company may forego or delay pursuit of opportunities with other product candidates or for other indications that may prove to have greater commercial potential. The Company’s resource allocation decisions may cause the Company to fail to capitalize on viable commercial products or profitable market opportunities. Spending on current and future research and development programs and on product candidates for specific indications may not yield any commercially viable products. If the Company does not accurately evaluate the commercial potential or target market for a particular product candidate, it may relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for the Company to retain sole development and commercialization rights to such product candidate.
Risks Related to the Company’s Product Development and Regulatory Approval
The Company is heavily dependent on the success of its lead product candidate, neflamapimod, which is still under clinical development. If neflamapimod does not receive regulatory approval or is not successfully commercialized, the Company’s business will be materially harmed.
The Company has invested almost all of its efforts and financial resources to date in the development of neflamapimod. To date, the Company has not initiated or completed a pivotal clinical trial, obtained marketing approval for any product candidate, manufactured a commercial scale product or arranged for a third party to do so on its behalf, or conducted sales and marketing activities necessary for successful product commercialization. The Company’s future success is substantially dependent on its ability to successfully complete clinical development of, obtain regulatory approval for, and successfully commercialize neflamapimod as a treatment for DLB and additional indications, which may never occur.
The Company expects a substantial portion of its efforts and expenditures over the next few years will be devoted to the advancement of neflamapimod’s clinical development. In order to be successful, the Company will need to successfully manage clinical and manufacturing activities, the pursuit of regulatory approval in multiple jurisdictions, securing manufacturing supply, building a commercial organization, and significant marketing efforts, among other requirements, before it can generate any revenues from commercial sales. The Company cannot be certain that it will be able to successfully complete any or all of these activities.
Furthermore, the Company has not submitted an NDA to the FDA or comparable applications to other regulatory authorities for neflamapimod, and it does not expect to be in a position to do so in the near future, if ever. Significant additional clinical testing and research will be required before it can file an NDA or any other application seeking approval of neflamapimod for the treatment of DLB, or any other indication. If the Company is unable to obtain the necessary regulatory approvals for and commercialize neflamapimod, it would materially adversely affect the Company’s financial position, and the Company may not be able to generate sufficient revenue to continue its business.
The development and commercialization of drug products is subject to extensive regulation, and the regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time-consuming, and inherently unpredictable. There is no guarantee that the Company’s planned clinical trials for neflamapimod to treat patients with DLB, or in any other indications that the Company may pursue, will be successful. If the Company is ultimately unable to obtain regulatory approval for neflamapimod on a timely basis, or at all, its business will be substantially harmed.
Clinical trials are expensive and can be difficult to design and implement. Such trials can take many years to complete, and their outcomes are inherently uncertain. Failure can occur at any stage during the clinical development process. The Company may experience difficulties in initiating and completing the clinical trials that it intends to conduct, and the Company does not know whether such trials will enroll patients on time, need to be redesigned, or be completed on schedule, if at all. In connection with designing and conducting its clinical trials, the Company faces significant risks, including that its product candidate may not prove to be efficacious, patients may suffer adverse effects for reasons that may or may not be related to the product candidate being tested, the results may not confirm the positive results of its earlier preclinical studies and clinical trials, includedand the results may not meet the level of statistical significance required by the FDA or other regulatory agencies to support approval.
For example, in the applicationCompany’s AscenD-LB Trial, neflamapimod demonstrated improvement versus placebo in dementia severity and motor function. Although the Company’s ongoing RewinD-LB Trial was designed as a confirmatory, hypothesis-testing, randomized, double-blind placebo-controlled clinical study of neflamapimod in subjects with DLB, the RewinD-LB Trial may not be successful, or the FDA may disagree with the Company’s interpretation of the clinical trial data or how those data inform the design of a potentially pivotal Phase 3 clinical trial for the Company’s lead indication. In addition, even if the AscenD-LB Trial results are confirmed in the RewinD-LB Trial, the Company will still need to successfully complete additional clinical trials, including a Phase 3 trial, before it is notprepared to submit an NDA for regulatory approval of neflamapimod in patients with DLB, assuming that the data collected from the Company’s clinical trials are deemed sufficient to support the submission of an NDA. The Company cannot predict with any certainty if or when it might complete its development efforts and submit an NDA for regulatory approval of neflamapimod, or dowhether any such NDA will be approved by the FDA. An NDA or comparable foreign submission seeking marketing approval for neflamapimod also may not meet a required levelbe accepted by FDA or foreign regulatory authorities due to, among other reasons, the content or formatting of statistical orthe submission.
This lengthy approval process, as well as the unpredictability of future clinical significance, includingtrial results, may result in the Company’s failure to obtain regulatory approval to market neflamapimod as a resulttreatment for DLB or any other indication, which would significantly harm the Company’s business, results of a differing interpretation of the data than that presented by the Companyoperations, and prospects. The FDA and comparable foreign regulatory authorities have substantial discretion in our application;
a determination that the perceived risks of approving the product candidate outweigh the clinical and other benefits of approval;
a determination that additional preclinical studies or clinical trials are required, either prior to or as a contingency to approval, and, for certain target indications such as pediatric populations, in the targeted sub-population;
a determination that a product candidate may only be approved on a contingent basis or for a more limited indication or patient population than we request;
a determination that labeling we believe is necessary or desirable for successful commercialization cannot be approved; or
unanticipated future changes to the approval process, and related regulations.
Historically, of the large number of drugs in development at any given time, only a small percentage successfully complete thedetermining when or whether regulatory approval processes andwill be obtained for any new product candidate. Accordingly, even if the Company believes the data collected from its clinical trials are ultimately commercialized. Our product candidatespromising, such data may not be approved for sale and marketingsufficient to support approval by the FDA or any other governmental authority, even if they meet specified endpoints in our clinical trials. The FDA or applicablecomparable foreign regulatory agenciesauthority. As a result, the Company may ask usbe required to conduct additional costly and time-consumingnonclinical studies, alter its proposed clinical trial designs, or conduct additional clinical trials to satisfy the regulatory authorities in ordereach of the jurisdictions in which it hopes to conduct clinical trials and develop and market neflamapimod or any of other product candidates, if approved.
The Company is also generally required to register certain clinical trials and post the results of completed clinical trials on a government-sponsored database, such as ClinicalTrials.gov in the U.S., within certain timeframes. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions.
Clinical drug development involves a lengthy and expensive process, with an uncertain outcome. The Company may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of neflamapimod or any other product candidates the Company may develop or acquire.
The risk of failure in drug development is high. Before obtaining marketing approval from regulatory authorities for the sale of any product candidate, a company must complete nonclinical development and conduct extensive clinical trials to demonstrate the safety and efficacy of its product candidates in humans. Clinical trials are expensive, difficult to design and implement and can take several years to complete, and their outcomes are inherently uncertain with the potential for failure at any time during the clinical development process. Preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and early-stage clinical trials have nonetheless failed to obtain marketing approval of their products. It is impossible to predict when or if neflamapimod will receive marketing approval.
The Company may experience numerous unforeseen events during, or as a result of, its clinical trials that could delay or prevent its ability to receive marketing approval or commercialize neflamapimod for DLB or any other indication. Clinical trials may be delayed, suspended or prematurely terminated because costs are greater than the Company anticipates or for a variety of other reasons, such as:
| ● | delay or failure in reaching agreement with the FDA or a comparable foreign regulatory authority on a trial design that the Company is able to execute; |
| ● | delay or failure in obtaining authorization to commence a trial, including approval from the appropriate IRB or ethics committee at each clinical site to conduct testing of a candidate on human subjects, or inability to comply with conditions imposed by a regulatory authority regarding the scope or design of a clinical trial; |
| ● | delays in reaching, or failure to reach, agreement on acceptable terms with prospective trial sites and prospective CROs, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| ● | inability, delay or failure in identifying and maintaining a sufficient number of trial sites, many of which may already be engaged in other clinical programs; |
| ● | inability, delay or failure in identifying, recruiting, and training suitable clinical investigators; |
| ● | delay or failure in recruiting, screening, and enrolling suitable subjects to participate in a trial; |
| ● | delay or failure in having subjects complete a trial or return for post-treatment follow-up; |
| ● | delays caused by operational issues at clinical trial sites, including insufficient staffing; |
| ● | changes to the clinical trial protocols and/or changes in regulatory requirements and guidance that require amending or submitting new clinical protocols; |
| ● | clinical sites and investigators deviating from the clinical protocol, failing to conduct the trial in accordance with Good Clinical Practices or other regulatory requirements, or dropping out of a trial; |
| ● | failure to initiate or delay of or inability to complete a clinical trial as a result of the authorizing IND or foreign clinical trial application being placed on temporary or permanent clinical hold by the FDA or comparable foreign regulatory authority; |
| ● | lack of adequate funding to continue a clinical trial, including as a result of unforeseen costs due to enrollment delays, requirements to conduct additional clinical trials and increased expenses associated with the services of the Company’s CROs and other third parties, or the cost of clinical trials being greater than the Company anticipated; |
| ● | delays in manufacturing, testing, releasing, validating or importing/exporting sufficient stable quantities of drug product for use in clinical trials or the inability to do any of the foregoing; |
| ● | developments on trials conducted by competitors for related technology that raise FDA or foreign regulatory authority concerns about risk to patients of a technology or in any indication more broadly; |
| ● | clinical trials of the Company’s product candidates may produce negative or inconclusive results, and the Company may decide, or regulators may require the Company, to conduct additional nonclinical studies, clinical trials or abandon product development programs; |
| ● | the number of patients required for clinical trials of the Company’s product candidates may be larger than the Company anticipates, enrollment in these clinical trials may be slower than it anticipates or participants may drop out of these clinical trials at a higher rate than it anticipates; |
| ● | the Company’s third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to the Company in a timely manner, or at all; |
| ● | regulators, the IRB or a Data Safety Monitoring Board if one is used for the Company’s clinical trials, may require that the Company suspend or terminate its clinical trials for various reasons, including noncompliance with regulatory requirements, unforeseen safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, or a finding that the participants are being exposed to unacceptable health risks; |
| ● | the supply or quality of the Company’s product candidates or other materials necessary to conduct clinical trials of the Company’s product candidates may be insufficient or inadequate; |
| ● | transfer of manufacturing processes to larger-scale facilities operated by a CMO, and delays or failure by the Company’s CMOs or the Company to make any necessary changes to such manufacturing process; |
| ● | the FDA or comparable foreign regulatory authorities may require the Company to submit additional data or impose other requirements before permitting it to initiate a clinical trial; or |
| ● | changes in governmental regulations or administrative actions. |
Many of the factors that cause, or lead to, enter into a further phasedelay in the commencement or completion of clinical development,trials may also ultimately lead to the denial of marketing approval for neflamapimod or any other future product candidates. Further, the FDA or comparable foreign regulatory authorities may disagree with the Company’s clinical trial design and the Company’s interpretation of data from clinical trials or may change the requirements for approval even after such agencythe FDA has reviewed and commented on the design for the Company’s clinical trials.
If the Company is required to conduct additional clinical trials or other preclinical studies of neflamapimod in various disease conditions beyond those that the Company currently contemplates, if it is unable to successfully complete clinical trials of the Company’s product candidates or other studies, if the results of these trials or tests are not positive or are only modestly positive or if there are safety concerns, the Company may:
| ● | be delayed in obtaining marketing approval for its product candidates; |
| ● | not obtain marketing approval for its product candidates at all; |
| ● | obtain approval for indications or patient populations that are not as broad as intended or desired; |
| ● | obtain approval with labeling that includes significant use or distribution restrictions or safety warnings that would reduce the potential market for its products or inhibit its ability to successfully commercialize the Company’s products; |
| ● | be subject to additional post-marketing restrictions or requirements, including post-marketing testing; or |
| ● | have the product removed from the market after obtaining marketing approval. |
Any failure or delay in obtaining,commencing or inability to obtain, thecompleting clinical trials or obtaining regulatory approvals necessary to market and sell our product candidatesfor neflamapimod would delay or preventthe Company’s commercialization prospects, substantially increase the costs of commercializing neflamapimod, and would materiallyseverely harm the Company’s business and adversely effect our business, financial condition, or result of operations. Furthermore, if we determine in the future that the development, approval, or commercial prospects of any product candidate are insufficient to justify our continued expenditure of the associated development and other costs, we may choose to delay, suspend, or abandon our development or commercialization efforts with respect thereto, which would reduce or eliminate our potential return on investment for those product candidates.condition.
Our ability to develop our product candidates depends,The Company has concentrated its research and if TSC or anydevelopment efforts on the treatment of our other product candidates are approved, ourDLB, a disease that has seen limited success in drug development. The ability to successfully commercialize our products will depend,develop drugs for DLB and other age-related neurologic disorders is extremely difficult and is subject to a number of unique challenges. In addition, its rationale for neflamapimod in partthe treatment of DLB is based on our ability to successfully obtain sufficient quantitiesa scientific understanding of the necessary APIs, other component substances and materials, and finished drug product for our product candidates. We are currently entirely dependent on third parties for the manufacture and supply of our product candidates and their component parts, including, with respect to TSC, a sole supplier. Wedisease that may be unable to continue to develop or commercialize our product candidates or face significant delays in that process if we are unable to successfully obtain these materials or manufacture drug product in sufficient quantities.wrong.
Maintaining an adequate supplyDrug development in the field of TSCbrain diseases, including age-related neurologic disorders and our other product candidates to meet our needs is critical to theneurodegenerative diseases in particular, has seen very limited success of our business. However, manufacturing and supply of APIs, other substances and materials and finished drug products is a complex and technically challenging process, and changes beyond our direct control can impact the quality, volume, price, and successful delivery of our product candidates or impede, delay, limit or prevent the successful development and commercialization of our product candidates. Mistakes and mishandling are not uncommon in thehistorically. There have been limited efforts by biopharmaceutical and pharmaceutical industrycompanies to develop treatments for DLB and can affect successful production and supply significantly.
As of the date of this Annual Report, wethere are no therapies available for patients that have no internal manufacturing capabilities and therefore we do not have direct control over our abilitybeen approved with a specific indication to maintain drug supply sufficient to serve our needs for our ongoing and planned clinical trials or, if any of our product candidatestreat DLB. Only symptomatic therapies that are approved commercialization. Although wefor other diseases, generally either AD or Parkinson’s disease, are ultimately responsiblecurrently utilized to manage patients with DLB. In addition, many potential disease-modifying therapies have been evaluated in other neurodegenerative diseases, particularly in AD, and these have encountered challenges in their development and, as a result, only recently two disease-modifying treatments to treat AD have been approved in the U.S. Developing a product candidate for ensuring compliance withtreatment of these brain diseases is extremely difficult and subjects the Company to a number of challenges, including obtaining regulatory requirements such as cGMPs, we are dependent on our third party CMOs and other contract suppliers and manufacturers for the manufacture of our drug product, including both APIs and finished products, as well as day-to-day compliance with cGMPs and certain other manufacturing-related regulatory requirements. Facilities used by our contract suppliers and manufacturers to produce the APIs and other substances and materials or finished products for commercial sale must pass inspection, provide regulators with certain technical information, and be approved byapproval from the FDA and other relevant regulatory authorities who have only a limited set of precedents to confirm compliance with cGMP requirementsrely on.
The Company’s approach to the treatment of DLB focuses in large part on neflamapimod’s ability to inhibit the intra-cellular enzyme p38α. The expression of p38α is considered to be a critical contributor in the toxicity of inflammation, alpha-synuclein, amyloid-beta and tau to neurons and synapses, which the Company and other regulatory requirements. If the safetyscientific experts believe leads to synaptic dysfunction. Synaptic dysfunction, specifically impaired synaptic plasticity, leads to disruption of TSC or any of our other product candidates (or any component thereof)episodic memory and is founda significant event in the future to be compromised, we may not be able to successfully commercialize or obtain regulatory approval for the product candidate,development and we may be held liable for injuries sustained as a result.symptomatology of DLB.
Any disruptionHowever, the Company cannot be certain that its approach will lead to the development of approvable or marketable products. To date the only drugs approved by the FDA to treat DLB have addressed the disease’s symptoms. In addition, there has never been an approval of a drug in our relationshipDLB and therefore, there are no regulatory precedents for endpoints in that indication. Consequently, the FDA has a limited set of products to rely upon in evaluating neflamapimod. This could result in a longer than expected regulatory review process, increased expected development costs or the delay or prevention of commercialization of neflamapimod for the treatment of DLB.
Moreover, given the history of clinical failures in this field, future clinical or regulatory failures by the Company or others may result in further negative perception of the likelihood of success in this field, which may significantly and adversely affect the Company’s business and the market price of its common stock.
Enrollment and retention of participants in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible by multiple factors outside the Company’s control.
The timely completion of clinical trials in accordance with these third parties or their protocols depends on, among other things, the Company’s ability to manufactureenroll a sufficient number of research participants who remain in the APIs and finished drug product we need for ourstudy until its conclusion. The Company may encounter delays in enrolling, or be unable to enroll, a sufficient number of individuals to complete any of its clinical trials, and other development activities could result in significant delays in our anticipated development timelines and/or significant additional supply costs. Sucheven once enrolled the Company may be unable to retain a disruption could be the result of anysufficient number of reasons, including contractual disputes with our partners, regulatory issues at our partner or at their facilities (whether or not relatedparticipants to Diffusion or our drug product), financial issues faced by our partners (including bankruptcy or insolvency), damages to our partners facility or equipment, communication breakdowns, or actscomplete any of God. For example, we are currently facing certain delaysits trials. Subject enrollment and retention in the manufacturing process for planned, new batches of TSC drug product due to the fact that, in connectionclinical trials depends on many factors, including:
| ● | the eligibility criteria defined in the protocol; |
| ● | the size of the patient population required for analysis of the trial’s primary endpoints; |
| ● | the nature of the trial protocol; |
| ● | the proximity of potential subjects to clinical sites; |
| ● | the existing body of safety and efficacy data with respect to the product candidate; |
| ● | the Company’s ability to recruit clinical trial investigators with the appropriate competencies and experience; |
| ● | clinicians’ and patients’ perceptions as to the potential advantages of the product candidate being studied in relation to other available therapies; |
| ● | competing clinical trials being conducted by other companies or institutions; |
| ● | the risk that participants enrolled in clinical trials will drop out of the trials before completion; and |
| ● | the operational efficiency of trial sites, including sufficient staffing. |
In addition, the U.S. federal government’s Operation Warp Speed initiative in responseCongress recently amended the FDCA to the COVID-19 pandemic, the facility at which our primary CMO partner conducts significant portionsrequire sponsors of the TSC manufacturing process has been mandateda Phase 3 clinical trial, or other “pivotal study” of a new drug or biologic to devote the majority of the facility’s available resourcessupport marketing authorization, to the manufacture of components of the COVID-19 vaccine. If our CMO is unable to manufacture sufficient quantities of drug supply to meet our needs indesign and submit a timely fashion or in accordance with the specifications contracteddiversity action plan for we may be forced to delay our development planssuch clinical trial. The action plan must describe appropriate diversity goals for TSC, find alternative suppliers, or pursue other available remedies, which could result in additional costs and the diversion of management’s time.
Amplifying this risk is the fact that we currently depend upon a sole source for manufacture of API for TSC and other aspects of our manufacturing process, limiting our available options to troubleshoot these issues. Although we are currently in the process of identifying and developing alternative manufacturing and supply alternatives, this process remains ongoing, will take time, and will involve significant costs. Furthermore, due to the significant regulatory oversight of the pharmaceutical manufacturing process, any changes in the identity of our third-party partners or in our manufacturing processes – even if in the best interests of the Company and successful – could result in regulatory and other delays,enrollment, as well as significant additional costs. In addition, if our current supplier terminated our arrangement or failed to meet our supply needs for any reason prior to the time we are able to identify sufficient alternative manufacturing capacity, we may be forced to delay our development plans significantly.
Our CMO and other manufacturing and supply partners are also engaged to supply and manufacture materials or products for other biopharmaceutical and pharmaceutical companies, exposing them to regulatory risks unrelated to the work they are doing for Diffusion but which may nevertheless impact their ability to meet their contractual requirements to us or otherwise impede their ability to supply us with sufficient quantities of drug product. Failure to meet the regulatory requirementsa rationale for the productiongoals and a description of those materials and products may also affecthow the regulatory clearance of a contract supplier’s or manufacturer’s facility. If the FDA or a comparable foreign regulatory agency does not approve these facilities for the supply or manufacturesponsor will meet them. Although none of our product candidates has reached Phase 3 of clinical development, we or if it withdraws its approval inour licensing partners must submit a diversity action plan to the FDA by the time a Phase 3 trial, or pivotal study, protocol is submitted to the agency for review, unless we or our licensing partners are able to obtain a waiver for some or all of the requirements for a diversity action plan. It is unknown at this time how the diversity action plan may affect the planning and timing of any future even if such lack of approval is unrelated to Diffusion orPhase 3 trial for our product candidates weor what specific information FDA will expect in such plans. However, initiation of such trials may needbe delayed if the FDA objects to find alternative supply or manufacturing facilities.
In addition, to date we have only manufactured TSC and our other product candidates in relatively small quantities for preclinical studies and clinical trials. As we prepare for additional, later-stage clinical trials and potential commercialization, we will need to take steps to substantially increase the scale at which we are able to product TSC, its API, and its other component parts. In order to meet these needs, our CMOs and suppliers will need to be able to produce our API, other components, and finished product in larger quantities, more cost effectively and, in certain cases, at higher yields than they currently achieve. These third-party contractors may not be able to successfully increase the manufacturing capacitya proposed diversity action plans for any of such drug substance and product candidates in a timely or cost-effective manner or at all. Even if such a scale up is possible, it may require additional processes, technologies, and validation studies, which are costly, may not be successful, and which the FDA and foreign regulatory authorities would need to review and approve prior to any commercial sale of TSC or any other product candidate. In addition, quality issues may arise during those scale-up activities because of the inherent properties of a product candidate itself or in combination with other components added during the process of manufacturing, packaging, shipping, or storage.
Our reliance on contract manufacturers and suppliers further exposes us to the possibility that they, or third parties with access to their facilities, will have access to and may misappropriate our trade secrets or other proprietary information. In addition, the manufacturing facilities of certain of our suppliers are located outside of the United States. This may give rise to difficulties in importing our products or product candidates or their components into the United States or other countries as a result of, among other things, regulatory agency approval requirements or import inspections, incomplete or inaccurate import documentation or defective packaging.
Any of these factors could cause a delay or termination of preclinical studies, clinical trials, other development activities, regulatory submissions or approvalsfuture Phase 3 trial of our product candidates, and we or ifour licensing partners may experience difficulties recruiting a diverse population of patients in attempting to fulfill the requirements of any approved commercial supply,diversity action plan.
Furthermore, any negative results the Company may report in clinical trials may make it difficult or impossible to recruit and couldretain subjects in other clinical trials of that same product candidate. Delays or failures in planned enrollment or retention of clinical trial subjects, including in the Company’s ongoing RewinD-LB Trial, may result in significant, unanticipatedincreased costs or anprogram delays, which could have a harmful effect on the Company’s ability to effectively develop our products candidatesa product candidate or commercialize our approved products on a timely basis, or at all, which could materially and adversely effect our business, financial condition, or results of operations.
We expect to rely on third-party CROs and other third parties to conduct and oversee our clinical trials and other aspects of ourrender further development process for TSC and our other product candidates. If these third parties do not meet our requirements or otherwise conduct the trials or perform the other services for which they are engaged, we may not be able to successfully develop, obtain regulatory approval for, or commercialize our product candidates when expected or at all. Furthermore, if we are not able to establish and maintain the necessary collaborative relationships with our CROs and other third party partners, we may have to alter our development and commercialization plans.impossible.
Conducting ourResults of preclinical studies and early clinical trials in a safe, compliant, and timely manner is critical to our success. We currently rely on third-party CROs to conduct and oversee our clinical trials and other aspects of our product development, as well as various medical institutions, clinical investigators, contract laboratories, consultants, and other third parties to design and conduct our trials, to analyze the results therefrom, and to ensure that the trials are conducted in accordance with our clinical protocols and all applicable regulatory requirements, including the FDA’s regulations and GCPs. These CROs and other third parties play a significant role in the conduct of these trials and the subsequent collection and analysis of data therefrom, as we control only certain aspects of their activities and rely heavily on them to execute our trials in a safe, compliant, and timely manner. Although we intend to internalize portions of some of these functions during 2021 and beyond as our organization grows, we expect to continue to rely on these third parties to a significant degree in the future.
If any of our CROs, clinical trial sites, or other third party partners terminates their involvement in one of our clinical trials (or with Diffusion entirely) for any reason, we may not be ableindicative of results obtained in later trials. In addition, preliminary, topline and interim data from the Company’s clinical trials that the Company may announce or publish from time to enter into alternative arrangementstime may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
The results of preclinical studies and early clinical trials of a product candidate, including neflamapimod, may not be predictive of the results of later-stage clinical trials. Product candidates in later stages of clinical trials may fail to show the desired safety and efficacy traits despite having progressed through preclinical studies and initial clinical trials. A number of companies in the biopharmaceutical industry, both generally and in the DLB treatment space in particular, have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety profiles, notwithstanding promising results in earlier trials. Even if the Company’s clinical trials for neflamapimod are completed as planned, including a future Phase 3 trial, the Company cannot be certain that their results will support the safety and efficacy sufficient to meet our needs, on a timely basis, on commercially reasonable terms,obtain regulatory approval, and the Company may decide, or at all. In addition, if our relationship withregulators may require it, to conduct additional clinical trial sites is terminated, we may incur significant additional costs or experience the loss of follow-up information on patients enrolled in our ongoing clinical trials, unless we are able to transfer the care of those patients to another qualified clinical trial site.trials.
We, as well asIn addition, from time-to-time, the CROs and other third-party contractors acting on our behalf, are required to comply with GCP and GLP requirements in all of ourCompany may announce or publish preliminary, topline, or interim data from its clinical trials, which are enforced through periodic inspectionsbased on a preliminary analysis of trial sponsors, principal investigators,then-available data. Such results and trial sites. If werelated findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study or anytrial. The Company also makes assumptions, estimations, calculations and conclusions as part of these third parties failits analyses of data, which may prove to complybe incomplete or flawed, and it may not have received or had the opportunity to fully and carefully evaluate all data. Preliminary and interim data are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Preliminary or interim data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data the Company previously published. As a result, preliminary and interim data are not necessarily predictive of final results and should be viewed with applicable GCP, GLP,caution until the final data are available. Adverse differences between preliminary or interim data and final data could significantly harm the Company’s business prospects.
Moreover, clinical data are often susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless failed to obtain approval from the FDA, the EMA or other regulatory requirements,agencies for their products. Others, including regulatory agencies, may not accept or agree with the clinicalCompany’s assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data generated in our clinical trials may be deemed unreliable and we may be required to perform additional clinical trials to supplementdifferently, which could impact the value of the particular program, the approvability or replace such data before receiving approvalcommercialization of athe particular product candidate fromand the FDA or a non-U.S. regulatory authority. Our clinical trials must also generally be conducted with product produced under cGMP regulations. Our and our partners’ compliance with these various regulations may be reviewed by regulatory inspections at any time, processes over which we will have very little control or immediate visibility, and a failure to comply with these regulations and policies by us, our CROs, or any of our other third party partners may resultCompany in significant delays in our development programs. In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and could receive cash or equity compensation in connection with such services. If these relationships and any related compensation result in perceived or actual conflicts of interest, the integrity of the data generated at the applicable clinical trial site may be questioned by the FDA.general.
In addition, in orderthe information the Company chooses to fundpublicly disclose regarding a particular study or clinical trial is typically selected from a more extensive amount of available information. Others may not agree with what the Company determines is the material or otherwise further development of our current or future product candidates, weappropriate information to include in its disclosure, and any information the Company determines not to disclose may collaborate with other pharmaceutical and biotechnology companies on their development and potential commercialization of those product candidates. We would faceultimately be deemed significant competition in seeking appropriate partners and whether we reach a definitive agreement for a collaboration will depend on many factors, including, our assessment of the partner’s resources and experience, the terms and conditions of the proposed collaboration, the likelihood of approval by the FDA or other regulatory authorities; the potential market for the subject product candidate; uncertainty with respect to our ownership of our intellectual property; and industry and market conditions generally. These types of collaborations are complex and time-consuming to negotiate and document and could ultimately result in lower returns on investment for our stockholders than would have been achieved developing thefuture decisions, conclusions, views, activities or otherwise regarding neflamapimod, a future product candidate, without a partner. Further,or the Company’s business. If the interim, preliminary, or topline data that the Company reports differ from later, final or actual results, or if we wereothers, including the FDA and comparable foreign regulatory authorities, disagree with the conclusions reached, the Company’s ability to breach our obligations under the agreements governing any such future collaboration, we may face substantial consequences, including potential termination of the collaboration,obtain approval for and, our rights to our partners’if approved, commercialize its product candidates inmay be harmed, which we have invested substantial time and money, would be lost.
Any failure to successfully enter into and maintain the necessary relationships with CROs and our other current and future third party partners and collaborators could materially and adversely effect ourharm its business, financial condition, or results of operations.operations and prospects.
General Risks Related toRegulatory authorities, including the Development, Regulatory Approval, and CommercializationFDA, may not accept data from clinical trials conducted outside of TSC and Our Other Product Candidatestheir jurisdiction.
Our business, financial condition, or results of operations may also be materially adversely effected by a number of general risks related toThe Company has in the developmentpast and regulatory approval of our product candidates that are not specific to our Company, including:
• Our TSC COVID Trial, which we completed in February 2021, was conducted at the NIID in Bucharest, Romania and we may in the future conduct additional clinical trials for TSC or our otherevaluating its product candidates, including neflamapimod, outside the U.S. In connection with an application for marketing approval, the FDA may determine not to acceptThe acceptance of trial data from clinical trials conducted outside of the U.S. if they determineby the FDA may be subject to certain conditions or may not be accepted at all, and other comparable non-U.S. regulatory authorities may have similar restrictions and conditions with respect to clinical trials conducted outside of their jurisdiction. In cases where data from non-U.S. clinical trials are intended to serve as the basis for marketing approval in the U.S., the FDA will generally not accept such foreign trial data unless: (i) the data presented therefrom cannotare determined to be considered valid without further inspection of the clinical trial site, are not applicable to the U.S. population and U.S. medical practice, or as a resultpractice; (ii) the trials were performed by clinical investigators of certain other factors. recognized competence and pursuant to GCP regulations; and (iii) the FDA is able to validate the data through an onsite inspection, if necessary. Additionally, the FDA’s clinical trial requirements, including sufficient size of patient populations and statistical powering, must be met. Many comparable non-U.S. regulatory authorities have similar approval requirements.
There can be no assurance that the FDA will accept data from trials conducted outside of the U.S. or that any comparable non-U.S. regulatory authority will accept data we obtain fromform trials conducted outside of the TSC COVID Trialapplicable jurisdiction. If the FDA or otherany comparable non-U.S. regulatory authority does not accept such data or believes that additional data is necessary to supplement such data, it would result in the need for additional trials, wewhich would be costly and time-consuming, could delay a product candidate’s development plan, and which may conductresult in product candidates not receiving approval for commercialization in the applicable jurisdiction.
Conducting clinical trials outside the U.S. may also expose the Company to additional risks, including risks associated with the following, among other things: additional foreign regulatory requirements; foreign exchange fluctuations; compliance with foreign manufacturing, customs, shipment and storage requirements; the failure of enrolled subjects in foreign countries to adhere to clinical protocol as a result of differences in standard-of-care; cultural differences in medical practice and clinical research; diminished protection of intellectual property rights; and compliance with general local legal requirements.
Safety issues with neflamapimod or with any other product candidate the Company may develop or acquire in the future.future, or with product candidates or approved products of third parties that are similar to the Company’s product candidates, could give rise to delays in the regulatory approval process, restrictions on labeling or product withdrawal after approval, if any, or may otherwise cause the Company to modify or supplement its clinical development program.
Results of any clinical trial the Company conducts could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics. Serious adverse events or undesirable side effects caused by neflamapimod, or any other product candidates the Company may develop or acquire, could cause it or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or other comparable foreign authorities. Many compounds that have initially showed promise in clinical or earlier stage testing are later found to cause undesirable or unexpected side effects that prevented further development of the compound. Further, problems with product candidates or approved products marketed by third parties that utilize the same therapeutic target or that belong to the same therapeutic class as neflamapimod or any future product candidates of the Company could adversely affect the development, regulatory approval and commercialization of the Company’s product candidates.
For example, to date, neflamapimod has been evaluated in over 200 patients, at doses up to 750 mg twice a day, and up to 24 weeks of treatment. The adverse effects (side effects) seen in more than 5% of neflamapimod-treated patients include headache (10% in neflamapimod-treated patients vs. 5% in placebo recipients), diarrhea (10% vs. 5%), abdominal pain (6% vs. 5%), respiratory infection (5% vs. 5%), and falls (5% vs. 5%). In each case, these events were generally mild and in all but one case (a case of diarrhea and abdominal pain) did not lead to treatment discontinuation. In addition, increased levels of certain “liver enzymes” in the blood are a well-known dose-dependent side effect of p38 MAPK inhibitors. These liver enzymes, aspartate aminotransferase and alanine aminotransferase, are proteins are commonly produced in the liver, the measurements of which can help doctors evaluate liver function. In an early 2000s study of neflamapimod conducted by Vertex, during 12 weeks of dosing at 250mg BID (i.e., four-fold higher daily dosing than the dose in the RewinD-LB Trial) in 44 subjects with rheumatoid arthritis, elevations in such liver enzymes levels were noted in six subjects (14%).
After the Company acquired an exclusive license from Vertex to develop and commercialize neflamapimod for the treatment of AD and other CNS disorders, the Company submitted an IND application to the DNP in February 2015. The DNP cleared the Company’s clinical trial application in March 2015. However, in August 2015, following a standard review of the long-term animal toxicity studies, the DNP placed a partial clinical hold on the Company’s then ongoing Phase 2a study in AD and any subsequent studies proposed under the IND. A partial clinical hold means that the FDA suspends part of the clinical work requested under the IND (e.g., a specific protocol or part of a protocol is not allowed to proceed); however, all other protocols and/or remaining parts of the protocol are allowed to proceed under the IND. Under DNP’s partial clinical hold that remains in effect for the neflamapimod IND, the agency limited administration of neflamapimod to doses that lead to plasma drug levels that provide a ten-fold safety margin to human subjects, based on the plasma drug levels in animals that had previously led to minimal or equivocal toxicity findings. The Company’s current understanding of plasma drug levels achieved with neflamapimod in humans means that its investigational dosing in the U.S. is limited by this partial clinical hold to no more than 40 mg three times daily in patients weighing 50 kg (110 lbs.) or more.
• We face a number of risks relatedThe Company’s ongoing RewinD-LB Trial is being conducted at 40mg three times daily (limited to patients weighing 50 kg (110 pounds) or more within the U.S., and not so limited outside the U.S.) and the Company does not expect this partial clinical hold to impact its ongoing and planned clinical trials or its current development plan for neflamapimod. With respect to the adverse effects discussed above, the patients were asymptomatic, there were no associated increases in bilirubin, and the elevations resolved with treatment discontinuation. Furthermore, no liver enzyme abnormalities were observed in the AscenD-LB Trial. However, as the Company continues the development and clinical trials of neflamapimod, treatment-related SAEs may arise in the future. Side effects that are deemed to be drug-related could affect patient recruitment or the ability of enrolled subjects to complete the trial or result in potential product liability claims. Undesirable side effects in one of the Company’s clinical trials for neflamapimod in one indication could adversely affect enrollment in clinical trials, regulatory approval and commercialization of the Company’s product candidate in other indications. These side effects may not be appropriately recognized or moremanaged by the treating medical staff. In addition, discovery of ourpreviously unknown class effect problems may prevent or delay clinical development and commercial approval of product candidates to causeor result in restrictions on permissible uses after their approval. If the Company or others identify undesirable side effects have other unexpected properties, contain manufacturing defects, or be subject to misuse or abuse. The occurrencecaused by the mechanisms of one or moreaction of these events with respect to a product candidate or a class of product could delaycandidates, the FDA may require the Company to conduct additional clinical trials, or preventto implement a REMS program prior to commercial approval. Alternatively, regulatory authorities may not approve the product candidate or, as a condition of approval, may require specific warnings and contraindications or place certain limitations on how the Company can promote the drug. Following a potential future drug product approval, regulatory authorities might also withdraw such approval due to the discovery of previously unknown safety issues relating to the product and require the Company to take its regulatory approval, limit its commercial potential, result in additional pre- or post-approval regulatory requirements, or subject us to product liability exposure to consumers, health care providers, or others. Product liability claims could be brought indrug off the future even if a product is approved for commercial salemarket. Any of these occurrences may harm the Company’s business, financial condition and manufactured in facilities licensed and regulated by the appropriate governmental authorities, and if product liability claims brought against us in the future were to be successful, we could incur substantial liability if our insurance coverage for those claims proved to be inadequate.prospects significantly.
• Our employees, independent contractors, principal investigators, consultants, vendors, CROs,Further, clinical trials, by their nature, utilize a sample of the potential patient population. With a limited number of patients, rare and other third parties we worksevere side effects of neflamapimod or future product candidates may only be uncovered with ina significantly larger number of patients exposed to the course of our development activities may engage in misconductproduct candidate. If neflamapimod, or other improper activities, including noncompliance with regulatory standards and requirements, during the course of their employment or other engagement with us. Any such misconduct or improper activities, whether intentional or negligent, could result in regulatory sanctions or other penalties against the Company, exclusion from federal healthcare programs such as Medicare and Medicaid, the incurrence of substantial defense costs, and serious harm to our reputation.
In addition, although we currently have no marketed products, in the event TSC or any of our other product candidates are approved forthe Company may develop or acquire, receives marketing approval and commercial salethe Company or others identify undesirable side effects caused by the FDA orsuch product candidates (or any other regulatory authority, our business, financial condition, or results of operations may be materially adversely effected bysimilar products) after such approval, a number of general risks related to the commercialization of such products that are not specific to our Company,potentially significant negative consequences could result, including:
• Even if our product candidates obtain regulatory approval, they may fail to achieve the broad degree of physician and patient adoption and use necessary for commercial success. The degree and rate of physician and patient adoption will depend on a number of factors, including the clinical indications for which a product candidate is approved and its effectiveness compared to other therapies, cost and the availability of reimbursement and other coverage from third party payors, our ability to educate patients and healthcare providers regarding a new therapy, and the effectiveness of our sales and marketing efforts. Furthermore, we will face significant competition, often from products sold and marketed by companies with far greater resources than Diffusion, and our failure to effectively compete may prevent us from achieving significant market penetration.
• With respect to any such future products available only by prescription, if we are unable to achieve and maintain coverage and adequate levels of reimbursement from third party payors – including governmental health programs such as Medicare and Medicaid and private insurance companies – and access to such third party payors’ drug formularies, the commercial success of those products may be severely hindered. If any such products do not demonstrate attractive efficacy profiles, they may not qualify for coverage and reimbursement and, even if we obtain coverage for a given product, the resulting reimbursement payment rates might not be adequate, may require co-payments that patients find unacceptably high, and may vary from payor to payor, and there is no assurance that coverage and reimbursement levels necessary to achieve commercial success will be obtained..
• Any such future products candidates that we commercialize will be subject to ongoing and continued regulatory review, including rules and regulations of the FDA and similar non-U.S. governmental authorities relating to advertising, marketing and labeling (including restrictions on the promotion of off-label use), potential REMS requirements, routine manufacturing and other review, and required compliance with GLP. If we or a regulatory agency discovers previously unknown problems with any such product, or any facility at or process by which it is manufactured, we may face restrictions on the sale or distribution of such product or on our Company as a whole, including regulatory actions requiring us to modify marketing or sales materials, suspend manufacturing or ongoing trials, initiate a recall or withdrawal the product from the market entirely, enter into a consent decree, or submit to other civil or criminal investigations and penalties. If we are not able to achieve and maintain regulatory compliance, we may not be permitted to market our product candidates, which would adversely affect our ability to generate revenue and achieve or maintain profitability.
| ● | regulatory authorities may withdraw or limit their approval of such product candidates; |
| ● | regulatory authorities may require the addition of labeling statements, such as a “Boxed” Warning or a contraindication; |
| ● | the Company may be required to change the way such product candidates are distributed or administered, conduct additional clinical trials or change the labeling of the product candidates; |
| ● | the FDA may require a REMS plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools, and regulatory authorities in other jurisdictions may require comparable risk mitigation plans; |
| ● | the Company may be subject to regulatory investigations and government enforcement actions; |
| ● | the FDA or a comparable foreign regulatory authority may require the Company to conduct additional clinical trials or costly post-marketing testing and surveillance to monitor the safety and efficacy of the product; |
| ● | the Company may decide to recall such product candidates from the marketplace after they are approved; |
| ● | the Company could be sued and held liable for injury caused to individuals exposed to or taking its product candidates; and |
| ● | the Company’s reputation may suffer. |
• The biopharmaceutical and pharmaceutical industries are highly regulated and the potential for future legislative reform provides uncertainty and potential threatsCompany may be unable to our business and our potential future revenue and profitability of any such future products. Inobtain regulatory approval in the U.S., there have been, or foreign jurisdictions and, as a result, be unable to commercialize its product candidates and its ability to generate revenue will be materially impaired.
The time required to obtain FDA and other approvals is unpredictable but typically takes many years following the commencement of clinical trials, depending upon the type, complexity and novelty of the product candidate. The standards that the FDA and its foreign counterparts use when regulating companies such as ours are not always applied predictably or uniformly and can change. Any analysis we expect there will continueperform of data from chemistry, manufacturing and controls, preclinical and clinical activities is subject to be, a number of legislativeconfirmation and interpretation by regulatory changes to the healthcare system intended to contain or reduce the costs of medical products and medical services including those described in Part I – Item 1. Business – Certain Other Legislation and Regulations – Current Healthcare Laws and Regulations of this Annual Report. Additional state and federal healthcare reform measures may be adopted in the future, any ofauthorities, which could delay, limit the amounts that federal and state governments will payor prevent regulatory approval. We may also encounter unexpected delays or increased costs due to new government regulations, for healthcare products and services, which could result in reduced demand for our products once approved or additional pricing pressures. We cannot predict the likelihood, nature, or extent of government regulation that may ariseexample, from future legislation or administrative action, or executive action,from changes in FDA policy during the period of product development, clinical trials and FDA regulatory review. It is impossible to predict whether legislative changes will be enacted, or whether FDA or foreign regulations, guidance or interpretations will be changed, or what the impact of such changes, if any, may be. Any delay or failure in obtaining required approvals could adversely affect our ability to generate revenues from the particular product candidate for which we are seeking approval.
Furthermore, obtaining and maintaining regulatory approval of our product candidates in one jurisdiction does not guarantee that we will be able to obtain or maintain regulatory approval in any other jurisdiction, while a failure or delay in obtaining regulatory approval in one jurisdiction may have a negative effect on the regulatory approval process in others. For example, even if the FDA grants marketing approval of a product candidate, similar foreign regulatory authorities must also approve the manufacturing, marketing and promotion of the product candidate in those countries. Approval and licensure procedures vary among jurisdictions and can involve requirements and administrative review periods different from, and greater than, those in the U.S.United States, including additional nonclinical studies or clinical trials as clinical trials conducted in one jurisdiction may not be accepted by regulatory authorities in other jurisdictions. In many jurisdictions outside the United States, a product candidate must be approved for reimbursement before it can be approved for sale in that jurisdiction. In some cases, the price that we intend to charge for our products is also subject to approval. If we fail to comply with the regulatory requirements in international markets and/or receive applicable marketing approvals, our target market territories we may pursue.
Risks Relatedwill be reduced and our ability to Our Intellectual Propertyrealize the full market potential of our product candidates will be harmed.
SinceIf the foundingCompany seeks to enter into collaborative arrangements or strategic alliances for its drug candidates, but fails to enter into and maintain successful relationships, it may have to reduce or delay its drug development activities or increase its expenditures.
An important element of Diffusion LLCa biotechnology company’s strategy for developing, manufacturing and commercializing its drug candidates may be to enter into strategic alliances with pharmaceutical companies or other industry participants to advance its programs and enable it to maintain its financial and operational capacity. Biotechnology companies at the Company’s stage of development sometimes rely upon collaborative arrangements or strategic alliances to complete the development and commercialization of drug candidates, particularly after the Phase 2 stage of clinical testing.
To date, the Company has not entered into any collaborative arrangements or strategic alliances, and it may face significant competition in 2001, we have been principally focused onseeking such relationships. In addition, such arrangements may place the development of TSCthe Company’s drug candidates outside its control, require the Company to relinquish important rights, or may otherwise be on terms unfavorable to the Company. The Company may not be able to negotiate collaborations and alliances on acceptable terms, if at all. If the Company enters a collaborative arrangement and it proves to be unsuccessful, the Company may have to delay, or limit the size or scope of, certain of its drug development activities.
Alternatively, if the Company elects to fund drug development or research programs on its own, it will have to increase its expenditures and will need to obtain additional funding, which may not be available to the Company on acceptable terms, if at all.
If the Company is unable to take full advantage of regulatory programs designed to expedite drug development or provide other incentives, its development programs may be adversely impacted.
There are a number of programs administered by the FDA and other regulatory bodies to facilitate and expedite development of drugs in areas of unmet medical need. For example, neflamapimod received a fast track designation in October 2019 from the FDA for theinvestigation as a treatment of hypoxiaDLB. Fast track designation is granted by FDA, in response to a sponsor’s request, upon a determination that the product candidate is intended to treat a serious or life-threatening disease or condition and ashas the potential to address an unmet medical need, meaning it could provide a platformtherapeutic option for patients where none exists or a therapy that may be potentially superior to enhance standard-of-care treatmentexisting therapy based on efficacy or safety factors.
Fast track designation does not ensure that neflamapimod will receive marketing approval or that approval will be granted within any particular timeframe. Although fast track designation and other available FDA programs may expedite the development or approval process for conditions complicatedcertain drug candidates, such programs do not change the standards for approval, and the Company may not experience a faster development or regulatory review or approval process with fast track designation compared to conventional FDA procedures. In addition, the FDA may withdraw fast track designation for neflamapimod if it believes that the designation is no longer supported by hypoxia. Manydata from the Company’s clinical development program.
Neflamapimod may not qualify for or maintain designations under this or other programs under any of our issued patents were issued earlythe FDA’s existing or future programs to expedite drug development in ourareas of unmet medical need. The Company’s inability to fully take advantage of these programs may require the Company to run larger trials, incur delays, lose opportunities that may not otherwise be available to it, and incur greater expense in the development of its product candidates.
The Company relies on third parties to conduct, supervise and monitor its clinical trials. If those third parties do not successfully carry out their contractual duties, or if they perform in an unsatisfactory manner, the Company’s history. While we are regularly engaged in further research and development activities with respect to TSC and our other product candidates that may result in novel developments and findings that ultimately lead to further intellectual property opportunities, if the terms of any of our key patents expire before or soon after our therapeutic candidates are approved, or if manufacturers of biosimilar drugs successfully challenge our patents, our business financial condition, or results of operations maywill be materially adversely effected.harmed.
PatentsAlthough the Company designs and manages its nonclinical studies and clinical trials, it does not currently have athe ability to conduct clinical trials for neflamapimod on its own. The Company has relied, and will continue to rely, on third parties such as CROs, medical institutions, and clinical investigators to ensure the proper and timely conduct of its clinical trials. The Company’s reliance on CROs for clinical development activities limits its control over these activities, but the Company remains responsible for ensuring that each of its trials is conducted in accordance with the applicable protocol, as well as legal and regulatory and scientific standards. The Company has limited duration. Incontrol over these third parties, and they may not devote sufficient time and resources to the United States, if all maintenance fees are timely paid, the natural expirationCompany’s projects, or their performance may be substandard, resulting in clinical trial delays or suspensions, delays in submission of marketing applications or failure of a patentregulatory authority to accept the Company’s applications for filing. There is generally 20 years fromno assurance that the third parties the Company engages will be able to provide the functions, tests, activities or services as agreed upon, or provide them at the agreed upon price and timeline or to the Company’s requisite quality standards, including due to geopolitical events, natural disasters, public health emergencies or pandemics, or poor workforce relations or human capital management.
The Company and its earliest non-provisional filing dateCROs are required to comply with GLP requirements for preclinical studies and GCP requirements for clinical trials, which are regulations and guidelines enforced by the FDA and required by comparable foreign regulatory authorities. If the Company or its CROs fail to comply with GCP requirements, the clinical data generated in the U.S. Various extensionsCompany’s clinical trials may be available, including under the Hatch-Waxman Amendments as described elsewhere in this Annual Report, but the life of a patent,deemed unreliable, and the protection it affords,FDA or comparable foreign regulatory authorities may require the Company to perform additional clinical trials before approving marketing applications for the Company’s product candidates. There is limited. Evenalso no assurance these third parties will not make errors in the design, management or retention of the Company’s data or data systems. Any failures by such third parties could lead to a loss of data, which in turn could lead to delays in clinical development and obtaining regulatory approval. Third parties may not pass FDA or other regulatory audits, which could also delay or prohibit regulatory approval. In addition, the cost of such services could significantly increase over time. If these third parties do not successfully carry out their contractual duties or obligations, fail to meet expected deadlines, or if patents covering TSC and our product candidates, including with respectthe quality or accuracy of the clinical data they obtain is compromised due to their manufacturethe failure to adhere to the Company’s clinical protocols or use, are obtained, onceregulatory requirements or for any other reason, the patent life has expired, weCompany’s clinical trials may be open to additional competition from competitive medications, including biosimilar medications. In the case of Diffusion, the normal lives (in other words, prior to the application of any patent term restoration of marketing exclusivity periods thatextended, delayed or terminated, and it may not be available) of our issued U.S. patents related to TSC begin to expire in 2023, which may be prior to the time that we are able to obtain regulatory approval for, or successfully commercialize any product candidate that it develops. As a result, the Company’s financial results and if approved, commercialize TSC.the commercial prospects for neflamapimod would be harmed, its costs could increase, and its ability to generate revenue could be delayed, all of which could have a material adverse effect on the Company’s business, financial condition, results of operations and prospects.
The Company has employed several different CROs for clinical trial services. Although the Company believes there are numerous alternatives to provide these services, in the event that it seeks a new CRO, the Company may not be able to enter into replacement arrangements without delays or incurring additional expenses. Switching or adding additional CROs involves substantial cost and requires management’s time and focus. In addition, there is a natural transition period when a new CRO commences work. Though the Company intends to carefully manage its relationships with its CROs, there can be no assurance that the Company will not encounter challenges or delays in the future or that these delays or challenges will not have an adverse impact on its business, financial condition and prospects.
Even if patent term extensionThe Company’s reliance on third parties for the production of neflamapimod may result in delays in the Company’s clinical trials or regulatory approvals and may impair the development and ultimate commercialization of neflamapimod, which would adversely impact the Company’s business and financial position.
The Company has no manufacturing facilities and does not have extensive experience in the manufacturing of drugs or in designing drug-manufacturing processes. The Company currently relies on third parties for the manufacture of drug substance, the manufacture of drug product, and the packaging of drug product for clinical use. This reliance on contract manufacturers and suppliers subjects the Company to inherent uncertainties related to product safety, availability, security and cost. Holders of NDAs, or other protectionsforms of FDA approvals, or those distributing a regulated product under their own name, are ultimately responsible for compliance with manufacturing obligations even if the manufacturing is conducted by a third party.
The Company further intends to rely on third-party CMOs for the production of commercial supply of neflamapimod if it is ultimately approved. If CMOs cannot successfully manufacture drug substance and drug product for the Company’s neflamapimod program, or any other product candidate that the Company may develop or acquire in the future, in conformity to its specifications and the applicable regulatory requirements, the Company will not be able to secure or maintain regulatory approval for the use of that product candidate in clinical trials, or for commercial distribution of that product candidate, if approved. Additionally, any problems the Company experiences with any such CMOs could delay the manufacturing of its product candidates, which could harm its results of operations. All drug manufacturers and packagers are required to operate in accordance with FDA-mandated cGMPs. A failure of any of the Company’s current or future CMOs to establish and follow cGMPs and to document their adherence to such practices may lead to significant delays in obtaining regulatory approval of product candidates or the ultimate launch of products based on the Company’s product candidates into the market. In the event of such failure, the Company could also face fines, injunctions, civil penalties, and other sanctions. Further, if the FDA or a comparable foreign regulatory authority finds deficiencies with or does not approve a CMO’s facilities for the future commercial manufacture of neflamapimod, or if it withdraws any such approval or finds deficiencies in the future, the Company may need to find alternative manufacturing facilities, which would delay its development program and significantly impact its ability to obtain regulatory approval for or commercialize neflamapimod.
In addition, if any facility of the Company’s third-party drug manufacturers or suppliers were to suffer an accident or a force majeure event such as marketing exclusivity are available uponwar, missile or terrorist attack, earthquake, major fire or explosion, major equipment failure or power failure lasting beyond the regulatory approval (if any)capabilities of TSCits backup generators or similar event, the Company could be materially adversely affected and any of ourits clinical trials could be materially delayed. An extended shut down may force the Company to procure a new research and development facility or another manufacturer or supplier, which could be time-consuming.
The Company’s ongoing RewinD-LB Trial is being conducted with a drug substance (the API) previously manufactured at a third-party CMO. Future supplies of the neflamapimod drug substance could be interrupted from time to time, and the Company cannot be certain that alternative supplies could be obtained within a reasonable timeframe, at an acceptable cost, or at all. During this period, the Company may be unable to receive investigational neflamapimod supplies or any other product candidates it may develop or acquire. In addition, a disruption in the benefits provided by these regulatory and legislative mechanisms is limited and typically less robust than full patent protection. For example,supply of drug substance could delay the five-year extensioncommercial launch of patent life potentially available under the Hatch-Waxman Amendments cannot extend the remaining term ofCompany’s product candidates, if approved, or result in a patent beyond a total of 14 yearsshortage in supply, which would impair its ability to generate revenues from the datesale of its product candidates. Growth in the costs and expenses of raw materials may also impair the Company’s ability to cost effectively manufacture its product candidates.
The Company also currently relies on a third-party CMO (different than that for the API) for the manufacture of neflamapimod drug product. The Company has used the same manufacturer for its neflamapimod drug product in all its clinical trials to date. If neflamapimod is ultimately approved for commercial sale, the Company expects to continue to rely on third-party contractors for manufacturing the drug product. Although the Company intends to do so prior to any commercial launch, it has not yet entered into long-term agreements for the commercial supply of either drug substance or drug product with its current manufacturing providers, or with any alternate manufacturers.
While the Company believes that there are multiple alternative sources available for manufacturing of both drug substance and drug product in its neflamapimod program, the Company may not be able to enter into replacement arrangements, on acceptable terms or at all, without delays or additional expenditures. It cannot estimate these delays or costs with certainty but, if they were to occur, they could cause a delay in the Company’s development and commercialization efforts.
Although the Company generally has not, and does not intend to, begin a clinical trial unless it believes it has on hand, or will be able to obtain, a sufficient supply of neflamapimod to complete the clinical trial, any significant delay in the supply of neflamapimod drug substance or drug product could considerably delay conducting the Company’s clinical trials and potential regulatory approval and only one patent applicableof its product candidates.
Further, third-party suppliers, manufacturers, or distributors may not perform as agreed or may terminate their agreements with the Company, including due to an approved drug may be extended. Receivingthe effects related to geopolitical events, natural disasters, public health emergencies or pandemics, such an extension is also subjectas the COVID-19 pandemic, or force majeure events that affect their facilities or ability to compliance with applicable deadlines for application and certain otherperform. Any significant problem that the Company’s suppliers, manufacturers, distributors or regulatory requirements. Moreover,service providers experience could delay or interrupt supply of materials necessary to produce the lengthCompany’s product candidates. Failure to obtain the needed quantities of the extension could be less than we request. If we are unable to obtain patent term extension or the term of any such extension is less than we request, the period during which we can enforce our patent rights for thatCompany’s product will be shortened and our competitors may obtain approval to market competing products sooner than we expect. Also, the scope of our right to exclude during any patent term extension period may be limited or may not cover a competitor’s product or product use.
Given the amount of time required for the development, testing, and regulatory review of new drug candidates patents protecting any drug candidate might expire before or shortly after such drug candidate is approved or commercialized. Similarly, our patents and patent applications may not provide us with sufficient time or rights to commercialize our product candidates or exclude others from commercializing products similar ours. If our patent protections for our product candidates have expired, this could include manufacturers of identical, biosimilar drugs.
If we are unable to obtain new, patent claims with respect to our existing product candidates and other technologies, in particular those related to our lead product candidate TSC, that both adequately protect our intellectual property position for commercial purposes and extend the term of overall protection as compared to our existing issued patents, we may be reliant on these types of patent term extensions and similar exceptions for such protection during the ultimate commercialization of TSC. This limited protection could have a material and adverse effect on ourits business, financial condition, or results of operations.operations and prospects.
We may not be able to obtain or enforce patent rights or other intellectual property rights that cover ourIf the Company changes the manufacturers of its product candidates, and technologies that areit may be required to conduct comparability studies evaluating the manufacturing processes of sufficient breadth to prevent third parties from competing against us.the product candidates.
The pharmaceutical industry is characterized by rapidly advancing technologies, intense competition,FDA and other regulatory agencies maintain strict requirements governing the manufacturing process for prescription drug products that would apply to the Company’s product candidates, if approved. For example, when a strong emphasismanufacturer seeks to make any change to the manufacturing process, the FDA typically requires the applicant to conduct nonclinical and, depending on developing proprietary therapeutics. Numerous companies are engagedthe magnitude of the changes, potentially clinical comparability studies that evaluate the potential differences in the product candidates resulting from the change in the manufacturing process. If the Company were to change manufacturers of its drug substance or drug product during or after the clinical trials and regulatory approval process for neflamapimod or any of its other product candidates, the Company will be required to conduct comparability studies assessing product candidates manufactured at the new manufacturing facility. Further, manufacturing changes are generally categorized as having either a substantial, moderate, or minimal potential to adversely affect the identity, strength or quality of the drug product as they may relate to the safety or effectiveness of the product, and if a change has a substantial potential to have an adverse effect on the drug product, an applicant must submit and receive FDA approval of a prior approval supplemental application before the product made with the manufacturing change is distributed. Other forms of notice to the FDA are also required for manufacturing changes that have a moderate or minimal potential to have an adverse effect on the drug product’s safety or effectiveness. Regardless of the type of manufacturing change, the methods used and the facilities and controls used for the manufacture, processing, packaging, or holding of human drugs must comply with applicable cGMP regulations.
Delays in designing and completing a comparability study to the satisfaction of the FDA or other regulatory agencies could delay or preclude the Company’s development patenting, manufacturing,plans and, marketing of health care products competitive with those that we are developing, and we face competition from a number of sources, such as pharmaceutical companies, generic drug companies, biotechnology companies and academic and research institutions. Accordingly, ourthereby, delay the Company’s ability to obtainreceive marketing approval or limit its revenue and maintain patent protectiongrowth, once approved. In addition, in both the U.S. and non-U.S. jurisdictions will be criticalevent that the FDA or other regulatory agencies do not accept nonclinical comparability data, the Company may need to ability to successfully develop, obtain regulatoryconduct a study involving dosing of patients comparing the two products. That study may result in a delay in the approval for, and, in particular, commercialize TSC and our other product candidates. These protections are and will be essential to preserving and protecting our novel inventions, proprietary developments, and trade secrets and to preventing third parties from infringing upon them. In particular, our ability to protector launch of any of our product candidates from unauthorized or infringing use by third parties depends in substantial part on our ability to obtain and maintain valid and enforceable patents in the U.S. and worldwide.
Our patent portfolio includes patents and patent applications in the U.S. and other major markets covering our technology with varying scope, including issued U.S. patents related to composition of matter, formulation, methods of delivery, and methods of use and the scope of coverage vary from country to country. Although we believe that our intellectual property position is strong and are currently assessing our operations and existing portfolio for additional intellectual property opportunities, we do not have – and may be unable to obtain – patent protection for every aspect of our technology. For aspects of our technology for which we do not have patent coverage, or in countries where we do not have granted patents, we may not have any ability to prevent the unauthorized use of our technologies or technologies substantially similar to ours, and any patents that we may obtain in the future may be narrow in scope and thus easily circumvented by competitors. Further, in countries where we do not have granted patents, third parties may be able to make, use or sell products identical to or substantially similar to, ourits product candidates.
DueRisks Related to legal standards relatingthe Company’s Intellectual Property
If the Company does not adequately protect its proprietary rights, the Company may not be able to patentability,compete effectively.
The Company relies upon a combination of patents, trade secret protection and confidentiality agreements to protect the intellectual property related to neflamapimod. The Company’s commercial success depends in part on obtaining and maintaining proprietary rights in the U.S. and in international jurisdictions, and successfully defending these rights against third-party challenges if and as they occur. The Company seeks to protect its proprietary position by filing patent applications related to neflamapimod in the U.S. and in other countries.
Although the Company has already obtained several issued patents and is working to expand its estate with additional patent applications, third parties may challenge the validity, enforceability, and claimor scope of the Company’s patents, covering pharmaceutical inventions, our abilitywhich may result in such patents being narrowed, invalidated, or held unenforceable. Any successful opposition to obtain, maintainthese patents or any other patents owned by or licensed to the Company could deprive it of rights necessary for the successful commercialization of neflamapimod, or any other product candidates it may develop. Further, if the Company encounters delays in regulatory approvals due to patent-related issues, the period of time during which it could market a product candidate under patent protection could be reduced.
The Company’s issued patents and enforcepatent applications also remain subject to uncertainty and continued monitoring. The Company’s patent applications may fail to result in issued patents is uncertain and involves complex legal and factual questions.with claims that provide further coverage for neflamapimod in the U.S. or in foreign countries. The patent applicationprosecution process also known as patent prosecution, is expensive and time-consuming, and wethe Company may not be able to prepare, file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It isThe Company may also possible that we will fail to identify further patentable aspects of inventions made in the course ofits research and development and commercialization activitiesoutput before it is too late to obtain patent protection, on them. Therefore, these and anyincluding as a result of ourthe publication of prior art. There is also no assurance that all potentially relevant prior art relating to the Company’s patents and applications may not be prosecuted and enforced in an optimal manner. It is also possible that defects of form in the preparation or filing of our patents or patent applications may exist,has been found, which can invalidate a patent or may arise in the future, such as with respect to inadvertent prior public disclosures, proper priority claims, inventorship, claim scope, orprevent a patent term adjustments. If our current or future third party development partners are not fully cooperative or disagree with us as to the prosecution, maintenance, or enforcement of any patent rights, those patent rights could be compromised and we might not be able to prevent third parties from making, using and selling competing products. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how. Accordingly, we cannot guarantee that any patents will issueissuing from any of our currentlya pending patent applications, which could impair our ability to prevent competition from third parties.application.
Even for aspectsThe patent position of our technology for which we have obtained, or obtain in the future, patent protection, the complexity oflife sciences companies can often involve complex legal and factual questions underlyingand in recent years has been the subject of significant litigation. Publications of discoveries in scientific literature often lag behind the actual discoveries, and patent applications in the U.S. and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore, the Company cannot know with certainty whether it was the first to make the inventions claimed in its owned or licensed patents or pending patent applications, or that it was the first to file for patent protection of such inventions. Further, the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and the Company’s patents may be challenged in the courts or patent offices in the U.S. or other jurisdictions. Such challenges may result in patent claims means theybeing narrowed, invalidated, held unenforceable, in whole or in part, or reduced in term. Such a result could limit the Company’s ability to prevent others from using or commercializing similar or identical technology and products.
Furthermore, generic drug manufacturers or other competitors may challenge the scope, validity or enforceability of the Company’s patents, requiring it to engage in complex, lengthy and costly litigation or other proceedings. Generic drug manufacturers may also develop, seek approval for and launch generic versions of the Company’s products.
Without patent protection for the Company’s current or future product candidates, these candidates may be open to competition from other products. As a result, the Company’s patent portfolio may not provide usthe Company with sufficient rights to exclude others from commercializing products similar or identical to the Company’s.
The Company may also seek to rely on regulatory exclusivity for protection for ourof its product candidates, if approved for commercial sale. Implementation and enforcement of regulatory exclusivity, which may consist of regulatory data protection and market protection, varies widely from country to affordcountry. Failure to qualify for regulatory exclusivity, or failure to obtain or to maintain the extent or duration of such protections that the Company expects for its product candidates, if approved, could affect the Company’s decision on whether to market the products in a commercial advantage against competitive productsparticular country or processes,countries or could otherwise have an adverse impact on its revenue or results of operations.
There is currently no composition of matter patent protection that covers neflamapimod.
EIP acquired an exclusive license from Vertex in 2014 to develop and commercialize neflamapimod for the treatment of AD and other CNS disorders. This license covers know-how, preclinical and clinical data, and certain specified Vertex patent rights, including a composition of matter patent for neflamapimod that expired in 2017. EIP has thus focused its efforts on discoveries related to neflamapimod that are reflected in issued patents and patent applications covering a range of subjects, including: methods of treating patients suffering from DLB or AD, as well as methods of reducing amyloid plaque burden; methods of improving cognition and treating neurologic disorders; methods for promoting recovery of function in patients who have suffered acute neurologic injuries, including those resulting from brandedvarious forms of stroke; and generic pharmaceutical companies. We cannot guarantee that the claimsmethods of these patents are or will be held valid or enforceable by the courts or will provide us with any significant protection against competitive products or otherwise be commercially valuable to us. Third parties may design around or challenge the validity, enforceability or scope of such issued patents or any other issued patents we own or license, which may result in such patents being narrowed, invalidated or held unenforceable. If the breadth or strength of protection provided by the patents we hold or pursue with respect to our product candidates is challenged, it could dissuade companiestreating patients suffering from collaborating with us to develop, or threaten our ability to commercialize, our product candidates. Changes in either the patent laws or in the interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property.
dementia. In addition, EIP has filed patents have a limited lifespan, presenting further challenges in effectively protecting our technologiesrelated to formulations of neflamapimod, including pharmaceutical compositions for oral administration exhibiting desirable pharmacokinetics and associated commercial position.processes for the manufacture thereof. In the U.S., the natural expiration of a patent is generally 20 years after it is filed. VariousAlthough various extensions may be available, under a variety of legislative and regulatory avenues but often the life afforded by these extensions and the protections they afford are often limited relative to fullof a patent protection. The extensive period of time between patent filing and regulatory approval for a product candidate limits the time during which we can market a product candidate underis limited.
Accordingly, there is currently no composition matter patent protection which may particularly affectthat covers neflamapimod. Rather, the profitabilityCompany’s patents provide protection around either the use of our early-stage product candidates. In particular, we have been developing TSC sinceneflamapimod for specific or medical indication (so called “use patents”) or the foundingadministration of Diffusion LLCneflamapimod in the early 2000s and, asspecific manner (e.g., at a result, portions of our patent portfolio, including certain patents related to TSC’sspecific dose or in a specific formulation). Patents that are not around composition of matter will expire beginningare narrower in 2023. Ifscope (i.e., they do not protect against development of neflamapimod in an indication other than that the otherpatent defines), may be more difficult to defend against challenges against validity, and may be more difficult to enforce against infringement. For these reasons, some pharmaceutical companies choose not to develop and/or license compounds that are not covered by a composition of matter patent. The Company owns a patent that is issued patents in our portfolio currently or that may issue in the future are not adequate to protect our technologies, our competitive position mayU.S. around co-crystals of neflamapimod, any of which if they were successfully developed would be harmed and we may face generic or other competition sooner than we otherwise would.afforded composition of matter patent protection under this patent.
Furthermore,Accordingly, the lawslack of some foreign jurisdictions do not provide intellectual property rightscomposition of matter patent protection that covers neflamapimod may subject the Company to the same extent asincreased risk of third-party litigation and/or reduce third party collaborators’ interest in the United States and many companies have encountered significant difficulties in protecting and defending such rights in foreign jurisdictions. The legal systemsor valuation of certain countries, particularly certain developing countries, do not favor the enforcementneflamapimod, any of patents and other intellectual property protection, particularly those relating to pharmaceuticals, which could make it difficult for us to stophave an adverse effect on the infringement of our patents or marketing of competing products in violation of our proprietary rights generally in those countries. If we encounter such difficulties in protecting or are otherwise precluded from effectively protecting our intellectual property in foreign jurisdictions, our business prospects could be substantially harmed.
Proprietary trade secrets and unpatented know-how are also very important to our business. Although we have taken steps to protect our trade secrets and unpatented know-how by entering into confidentiality agreements with third parties, and intellectual property protection agreements with certain employees, consultants and advisors, third parties may still obtain this information or we may be unable to protect our rights. We also have limited control over the protection of trade secrets used by our suppliers, manufacturers and other third parties. There can be no assurance that binding agreements will not be breached, that we would have adequate remedies for any breach or that our trade secrets and unpatented know-how will not otherwise become known or be independently discovered by our competitors. If trade secrets are independently discovered, we would not be able to prevent their use. Enforcing a claim that a third party illegally obtained and is using our trade secrets or unpatented know-how is expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the U.S. may be less willing to protect trade secret information.
If we are unable to adequately obtain or enforce our patent and other intellectual property rights for any reason, it could materially and adversely effect ourCompany’s business, financial condition or results of operations. For more information about our intellectual property and our competition, see the information included in Part I – Item 1. Business – Products, Product Development, and Our Competition of this Annual Report.
If wethe Company fails to comply with its obligations under its existing license agreement with Vertex, or with any future intellectual property licenses with third parties, the Company could lose license rights that are important to its business.
The Company is party to the Vertex Agreement pursuant to which it acquired an exclusive license to develop and commercialize neflamapimod for the diagnosis, treatment, and prevention of AD and other CNS disorders. Under the terms of the Vertex Agreement, the Company must use commercially reasonable efforts during the license term to develop and obtain regulatory approval for a licensed product in specified major markets, and to promptly and effectively commercialize the licensed product once such approval is obtained. The Vertex Agreement also contains certain specified minimum diligence requirements, including making annual expenditures set forth in a development plan, and commencing a Phase 2 clinical trial of the licensed product within a specified time period.
The Vertex Agreement provides that either party may terminate the agreement if the other party is in material breach of its obligations thereunder, following a 60-day notice and cure period, or if the other party files for bankruptcy, reorganization, liquidation, receivership, or an assignment of a substantial portion of assets to creditors. The Vertex Agreement also provides that in the event the Company materially breaches any of certain specified diligence obligations as to a specific major market, Vertex’s sole remedy for such breach, following the applicable notice and cure period, would be to terminate the license as to such specific major market country.
Accordingly, any uncured, material breach under the Vertex Agreement could result in the loss of certain of its rights to neflamapimod and could compromise the Company’s development and commercialization efforts. This in turn would have an adverse effect on the Company’s business, which could be material.
The Company may become subject to third parties’ claims alleging infringement of their patents and proprietary rights, or the Company may need to become involved in lawsuits to protect or enforce ourits patents, or other intellectual property, or if we are sued for infringing intellectual property rightseither of third parties, it willwhich could be costly and time-consuming,time consuming, potentially delay or prevent the development and an unfavorable outcome in that litigation could have a material adverse effect on our business, financial condition,commercialization of the Company’s product candidates, or results of operations.put its patents and other proprietary rights at risk.
Our ultimateThe Company’s commercial success depends, in part, upon ourthe Company’s ability to develop, manufacture, market and sell ourits lead product candidate, neflamapimod, without alleged or actual infringement, misappropriation or other violation of the patents and proprietary rights of third parties. While the Company is not currently subject to any pending intellectual property litigation, and is not aware of any such threatened litigation, the Company may be exposed to future litigation by third parties based on claims that its product candidates, and use our proprietary technologies in the U.S. and non-U.S. markets. In order to do so, it is critical that we prevent third parties from infringing on our intellectual property rights and that we operate our business without infringing onor activities infringe the intellectual property rights of others.
However, numerous U.S. and non-U.S. issued patents and pending patent applications owned by third parties exist in fields relating to TSC and our other product candidates, their potential methods of delivery, potential indications they Some claimants may be used to treat, and their other features, and, as more patents are issued over time, the risk increases that others may assert that our product candidates, technologies, or methods of delivery or use infringe their patent or other intellectual property rights, or that we discover a third party infringing on our rights. Moreover, it is not always clear to industry participants, including us, which patents cover various drugs, biologics, drug delivery systems, or their methods of use, which of these patents may be valid and enforceable, and what inventions or technologies may be claimed by non-public patent applications. Patent applications in the U.S. and many foreign jurisdictions are typically not published until 18 months after their first non-provisional filing and publications in the scientific literature often lag behind actual discoveries, meaning we cannot be certain whether others, including our competitors, have filed patent applications for technology covered by patents or our pending applications and whether any such filing has priority over our own applications or patents.
In the biopharmaceutical and pharmaceutical industries in particular, there is a substantial amount of litigation involving patent and other intellectual property rights. This type of litigation may occur unexpectedly but may also be prompted by specific events, such as a patent application being made public by the USPTO or a non-U.S. governmental authority or under Paragraph IV of the Hatch-Waxman Amendments. For more information regarding the Hatch-Waxman Amendments and Paragraph IV thereunder, see the information in Part I – Item 1. Business – Government Regulation – The Hatch-Waxman Amendments of this Annual Report.
As of the date of this Annual Report, no litigation asserting infringement claims has been brought against us, nor have we filed such a claim against any third party. However, we cannot assure you that the development or future commercialization of any of our product candidates or other technologies will not result in claims that our activities infringe on the existing or future intellectual property rights of third parties. Furthermore, potential competitors may infringe our intellectual property, including our patents. For example, in the first quarter of 2021, we became aware of a third party affiliated with a former outside consultant ofsubstantially greater resources than the Company which claims to be in early-stage development of a product candidate that purportedly may operate through a similar mechanism of action to TSC.
We may be required to file infringement claims to stop third-party infringement or unauthorized use or, if a third party claims we are infringing on their rights, respond to such claims. This process can be expensivedoes and time consuming, and could result in a court deciding that a patent of ours is not valid or is unenforceable, that a third party is not required to stop using a technology we believe infringe on our rights, significant costs, or the diversion of management’s time. An adverse determination in any litigation or other proceedings could put one or more of our patents at risk of being invalidated, interpreted narrowly, or amended such that they do not cover our product candidates in a manner sufficient to support our development and commercialization needs or that such product candidate needs to be significantly redesigned, or put our pending patent applications at risk of not issuing, or issuing with limited and potentially inadequate scope. Further, some of our competitors may be able to sustain the costs of complex intellectual property litigation to a greater degree and for longer periods of time than the Company. In addition, patent litigationholding companies that focus solely on extracting royalties and settlements by enforcing patent rights may target the Company in the future. As the biotechnology and pharmaceutical industries expand and more effectively than we can because they have substantially greater resources.patents are issued, the risk increases that the Company’s product candidates may be subject to claims of infringement of the intellectual property rights of third parties.
The Company may be subject to third-party claims including infringement, interference or derivation proceedings, reexamination proceedings, post-grant review and inter partes review before the USPTO or similar adversarial proceedings or litigation in other jurisdictions. Even if the Company believes such claims are without merit, a court of competent jurisdiction could hold that these third-party patents are valid, enforceable and infringed, and the holders of any such patents may be able to block the Company’s ability to commercialize its applicable product candidate unless the Company obtained a license under the applicable patents, or until such patents expire or are finally determined to be invalid or unenforceable. These proceedings may also result in the Company’s patent claims being invalidated or narrowed in scope. In addition, interference, derivation,a court may hold that a third-party is entitled to certain patent ownership rights instead of the Company.
As a result of patent infringement claims, or in order to avoid potential infringement claims, the Company may choose to seek, or be required to seek, a license from the third party, which may require it to pay license fees or royalties or both. These licenses may not be available on acceptable terms, or at all. Even if a license can be obtained on acceptable terms, the rights may be nonexclusive, which could give the Company’s competitors access to the same intellectual property rights. If the Company is unable to enter into a license on acceptable terms, it could be prevented from commercializing one or more of its product candidates, forced to modify such product candidates, or to cease some aspect of the Company’s business operations, which could harm the Company’s business significantly. In addition, if the breadth or strength of protection provided by the Company’s patents and patent applications is threatened, it could dissuade companies from collaborating with the Company to license, develop or commercialize current or future product candidates.
If the Company were to initiate legal proceedings against a third party to enforce a patent covering one of its product candidates, the defendant could counterclaim that the Company’s patent is invalid or unenforceable. The outcome of proceedings involving assertions of invalidity and unenforceability during patent litigation is unpredictable. With respect to the validity of patents, for example, the Company cannot be certain that there is no invalidating prior art of which the Company and the patent examiner were unaware during prosecution. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, the Company would lose at least part, and perhaps all, of the corresponding patent protection on its product candidates. Furthermore, the Company’s patents and other intellectual property rights also will not protect its technology if competitors design around the Company’s protected technology without infringing its patents or other intellectual property rights.
Finally, even if resolved in the Company’s favor, litigation or other legal proceedings relating to intellectual property claims may cause the Company to incur significant expenses and could distract its technical and management personnel from their normal responsibilities. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments, which could damage the Company’s reputation, harm its business, and the price of its common stock could be adversely affected.
The Company may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent, which might adversely affect the Company’s ability to develop, manufacture and market its product candidates.
From time to time, the Company may identify patents or applications in the same general area as its products and product candidates. The Company may determine these third-party patents are irrelevant to its business based on various factors including its interpretation of the scope of the patent claims and its interpretation of when the patent expires. If the patents are asserted against the Company, however, a court may disagree with the Company’s determinations. Further, while the Company may determine that the scope of claims that will issue from a patent application does not present a risk, it is difficult to accurately predict the scope of claims that will issue from a patent application, the Company’s determination may be incorrect, and the issuing patent may be asserted against the Company. The Company cannot guarantee that it will be able to successfully settle or otherwise resolve such infringement claims. If the Company fails in any such dispute, in addition to being forced to pay monetary damages, it may be temporarily or permanently prohibited from commercializing certain product candidates. The Company might also be forced to redesign its product candidates so that it no longer infringes the third-party intellectual property rights, if such redesign is even possible. Any of these events, even if the Company were ultimately to prevail, could require it to divert substantial financial and management resources that it would otherwise be able to devote to its business.
The Company may be involved in lawsuits to protect or enforce its patents or other intellectual property or the intellectual property of its licensors, which could be expensive, time-consuming, and unsuccessful.
Competitors may infringe the Company’s patents or other intellectual property or the intellectual property of its licensors. To cease such infringement or unauthorized use, the Company may be required to file patent infringement claims, which can be expensive and time-consuming and divert the time and attention of the Company’s management and scientific personnel. The Company’s pending patent applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues therefrom. In addition, in an infringement proceeding or a declaratory judgment action, a court may decide that one or more of the Company’s patents is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that the Company’s patents do not cover the technology in question. An adverse result in any litigation or defense proceeding could put one or more of the Company’s patents at risk of being invalidated, held unenforceable, or interpreted narrowly and could put the Company’s patent applications at risk of not issuing. Defense of these claims, regardless of their merit, may involve substantial litigation expense and may be a substantial diversion of employee resources from the Company’s business.
Interference or derivation proceedings provoked by third parties or brought atby the USPTO may be necessary to determine the priority or patentability of inventions with respect to, ouror the correct inventorship of, the Company’s patents or patent applications. An unfavorable outcome could result in a loss of the Company’s current patent rights and could require the Company to cease using the related technology or to attempt to license rights to it from the prevailing party. The Company’s business could be harmed if the prevailing party does not offer it a license on commercially reasonable terms. Litigation, interference, derivation or USPTOother proceedings brought by us may fail or may be invoked against us by third parties. Evenresult in a decision adverse to the Company’s interests and, even if we arethe Company is successful, in these proceedings, domestic or foreign litigation or USPTO or foreign patent office proceedings may result in substantial costs and distract the diversion of management’s time. WeCompany’s management and other employees.
Even if the Company establishes infringement, a court may decide not to grant an injunction against further infringing activity and instead award only monetary damages, which may or may not be able to prevent all misappropriation of our proprietary rights, particularly in countries with a legal framework that offers limited intellectual property protections or where the costs of enforcement outweigh the commercial and other benefits of maintaining intellectual property protections.
an adequate remedy. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, or other proceedings, there is a risk that some of ourthe Company’s confidential information could be compromised by disclosure during this type of litigation or other proceedings, including as a result oflitigation. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments,developments. If securities analysts or public accessinvestors perceive these results to related documents. This type of disclosurebe negative, it could put us athave a significant competitive disadvantage by disclosing important trade secrets or other proprietary information to our competitors and other third parties.
Any litigation or other challenge related to our intellectual property could materially and adverselysubstantial adverse effect our business, financial condition, or results of operations.
General Risks Related to Our Intellectual Property
Our business, financial condition, or results of operations may also be materially adversely effected by a number of general risks related to our intellectual property that are not specific to our Company, including:
• As is common inon the biopharmaceutical and pharmaceutical industries, some of our employees were formerly employed by companies in the industry, including our competitors or potential competitors, and some of our consultants actively work for other companies in the industry. As a result, although we have in place policies which prohibit the use of third-party confidential information in violation of any obligation to a former employer or otherwise, we may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed to us alleged trade secrets of their former employers or their former or current customers. In addition, if any of our current employees or consultants are engaged by a competitor in the future, it is possible that they may appropriate or otherwise improperly use our proprietary and confidential information. Anyprice of the foregoing events could result in significant costs and the diversion of time and resources.Company’s common stock.
• Changes in patent laws or patent jurisprudence could diminish the value of patents in general, thereby impairing the Company’s ability to protect its product candidates.
The Company’s success is heavily dependent on intellectual property, particularly patents, and obtaining and enforcing patents in its industry involves both technological and legal complexity. Changes in either the patent laws or interpretation of the patent laws in the U.S. and other countries may diminish the value of the Company’s patents or narrow the scope of its patent protection.
For example, the AIA, which was passed in September 2011, resulted in significant changes to the U.S. patent system. Pursuant to the AIA, the U.S. transitioned to a “first-to-file” system for deciding which party should be granted a patent when two or more patent applications are filed by different parties claiming the same invention. A third party that files a patent application in the USPTO after that date but before the Company could therefore be awarded a patent covering an invention of the Company’s even if the Company made the invention before it was made by the third party. This requires the Company to be cognizant going forward of the time from invention to filing of a patent application.
The AIA also introduced changes that provide opportunities for third parties to challenge any issued patent with the USPTO. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Such changes could increase the uncertainties and costs surrounding the prosecution of the Company’s patent applications and the enforcement or defense of its issued patents.
In addition, the laws of other countries may not protect the Company’s rights to the same extent as the laws of the U.S. The complexity and uncertainty of European patent laws has increased in recent years, and the European patent system is relatively stringent in the type of amendments that are allowed during prosecution. Complying with these laws and regulations could limit the Company’s ability to obtain new patents in the future that may be important for its business.
The Company enjoys only limited geographical protection with respect to certain patents, and it may not be able to protect its intellectual property rights throughout the world.
Filing, prosecuting and defending patents covering the Company’s product candidates in all countries throughout the world would be prohibitively expensive and time-consuming with diminishing marginal returns. Competitors may use the Company’s technologies in jurisdictions where it has not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where the Company has patent protection, but enforcement is not as strong as that in the U.S. or the European Union. These products may compete with the Company’s product candidates, and its patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Although the Company intends to seek protection of its intellectual property rights in its expected significant markets, the Company cannot ensure that it will be able to initiate or maintain similar efforts in all jurisdictions in which the Company may wish to market its product candidates. The Company may also decide to abandon national and regional patent applications before grant. The grant proceeding of each national or regional patent is an independent proceeding, which may lead to situations in which applications might in some jurisdictions be refused by the relevant patent offices, while granted by others.
The legal systems of certain countries do not favor the enforcement of patents, trade secrets and other intellectual property protection, which could make it difficult for the Company to stop the infringement of its patents or marketing of competing products in violation of the Company’s proprietary rights generally. Proceedings to enforce its patent rights in other jurisdictions, whether or not successful, could result in substantial costs and divert its efforts and attention from other aspects of the Company’s business, could put the Company’s patents at risk of being invalidated or interpreted narrowly and its patent applications at risk of not issuing, and could provoke third parties to assert claims against the Company. The Company may not prevail in any lawsuits that it initiates, and the damages or other remedies awarded, if any, may not be commercially meaningful.
Some countries also have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In addition, some countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. If the Company is forced to grant a license to any third parties with respect to any patents relevant to the Company’s business, its competitive position may be impaired.
The lives of the Company’s patents may not be sufficient to effectively protect the Company’s products and business.
Patents have a limited lifespan. For example, in the U.S., if all maintenance fees are paid timely, the natural expiration of a patent is generally 20 years after its first effective filing date. Although various extensions may be available, the life of a patent, and the protection it affords, is limited. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such product candidates are commercialized. Even if patents covering the Company’s product candidates are obtained, once the patent life has expired for a product, the Company may be open to competition from biosimilar or generic medications. The launch of a generic version of one of the Company’s products, in particular, would be likely to result in an immediate and substantial reduction in the demand for that product, which could have a material adverse effect on the Company’s business, financial condition, results of operations and prospects. As a result, the Company’s patent portfolio may not provide it with sufficient rights to exclude others from commercializing product candidates similar or identical to the Company’s product candidates. In addition, although upon issuance in the U.S. a patent’s life can be increased based on certain delays caused by the USPTO, this increase can be reduced or eliminated based on certain delays caused by the patent applicant during patent prosecution. A patent term extension based on regulatory delay may be available in the U.S. However, only a single patent can be extended for each marketing approval, and any patent can be extended only once, for a single product. Moreover, the scope of protection during the period of the patent term extension does not extend to the full scope of the claim, but instead only to the scope of the product as approved. Laws governing analogous patent term extensions in foreign jurisdictions vary widely, as do laws governing the ability to obtain multiple patents from a single patent family. Additionally, the Company may not receive an extension if the Company fail to exercise due diligence during the testing phase or regulatory review process, apply within applicable deadlines, fail to apply prior to expiration of relevant patents or otherwise fail to satisfy applicable requirements. If the Company is unable to obtain patent term extension or restoration, or the term of any such extension is less than the Company requests, the period during which the Company will have the right to exclusively market the Company’s product will be shortened and the Company’s competitors may obtain approval of competing products following the Company’s patent expiration and may take advantage of the Company’s investment in development and clinical trials by referencing the Company’s clinical and preclinical data to launch their product earlier than might otherwise be the case, and the Company’s revenue could be reduced, possibly materially. If the Company does not have sufficient patent life to protect the Company’s products, the Company’s business and results of operations will be adversely affected.
Intellectual property discovered or developed through government funded programs may be subject to federal regulations such as “march-in” rights, certain reporting requirements and a manufacturing preference for U.S.-based companies. Compliance with such regulations may limit the Company’s exclusive rights and limit its ability to contract with non-U.S. manufacturers.
The Company received the NIA Grant to support its ongoing RewinD-LB Trial. Pursuant to the Bayh-Dole Act, the U.S. government may have certain rights in any invention developed or reduced to practice with this funding. In addition, in the future the Company may discover, develop, acquire, or license intellectual property that has been generated through the use of U.S. government funding or grants in which the U.S. government may have certain rights pursuant to the Bayh-Dole Act. These U.S. government rights include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any governmental purpose. In addition, the U.S. government has the right, under certain limited circumstances, to require the Company to grant exclusive, partially exclusive, or non-exclusive licenses to any of these inventions to a third party if it determines that: (1) adequate steps have not been taken to commercialize the invention; (2) government action is necessary to meet public health or safety needs; or (3) government action is necessary to meet requirements for public use under federal regulations (also referred to as “march-in rights”). Such “march-in” rights would apply to new subject matter arising from the use of such government funding or grants and would not extend to pre-existing subject matter or subject matter arising from funds unrelated to the government funding or grants. If the U.S. government exercises its march-in rights in the Company’s intellectual property rights that are generated through the use of U.S. government funding or grants, the Company could be required to license or sublicense intellectual property discovered or developed by it or that it licenses on terms unfavorable to the Company, and there can be no assurance that the Company would receive compensation from the U.S. government for the exercise of such rights. The U.S. government also has the right to take title to these inventions if the grant recipient fails to disclose the invention to the government or fails to file an application to register the intellectual property within specified time limits. Intellectual property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require the Company to expend substantial resources. Should any of these events occur, it could significantly harm the Company’s business, results of operations and prospects. In addition, the U.S. government requires that, in certain circumstances, any products embodying any of these inventions or produced through the use of any of these inventions be manufactured substantially in the U.S. This preference for U.S. industry may be waived by the federal agency that provided the funding if the owner or assignee of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the U.S. or that under the circumstances domestic manufacture is not commercially feasible. This preference for U.S. industry may limit the Company’s ability to contract with non-U.S. product manufacturers for products covered by such intellectual property.
The Company’s reliance on third parties requires the Company to share its trade secrets, which increases the possibility that its trade secrets will be misappropriated or disclosed, and confidentiality agreements with employees and third parties may not adequately prevent disclosure of trade secrets and protect other proprietary information.
The Company may rely on trade secrets or confidential know-how to protect various aspects of its business, especially where patent protection is believed by the Company to be of limited value. Due to its reliance on third parties in various aspects of its business, including CMC, R&D and collaborations, the Company must, at times, share trade secrets with such parties. The Company may also conduct joint research and development programs that require it to share trade secrets under the terms of the Company’s research and development partnerships or similar agreements. Such trade secrets or confidential know-how can be difficult to protect as confidential.
To protect this type of information against disclosure or appropriation by competitors, the Company’s policy is to require its employees, consultants, contractors and advisors to enter into confidentiality agreements and, if applicable, material transfer agreements, consulting agreements or other similar agreements with the Company prior to beginning research or disclosing proprietary information. However, current or former employees, consultants, contractors and advisers may unintentionally or willfully disclose the Company’s confidential information to competitors, and confidentiality agreements may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. Enforcing a claim that a third party obtained illegally and is using trade secrets or confidential know-how is expensive, time-consuming and unpredictable. In addition, the enforceability of confidentiality agreements may vary from jurisdiction to jurisdiction.
Despite the Company’s efforts to protect its trade secrets, the Company’s competitors may discover the Company’s trade secrets, either through breach of the Company’s agreements with third parties, independent development or publication of information by any of its third-party collaborators. A competitor’s discovery of the Company’s trade secrets could impair its competitive position and have an adverse impact on its business.
Intellectual property rights do not necessarily address all potential threats to the Company’s competitive advantage.
The degree of future protection afforded by the Company’s intellectual property rights is uncertain because intellectual property rights have limitations and may not adequately protect the Company’s business or permit the Company to maintain its competitive advantage. For example:
| ● | others may be able to make product candidates that are similar to ours but that are not covered by the claims of the patents that the Company owns or has exclusively licensed; |
| ● | others may independently develop similar or alternative technologies or duplicate any of the Company’s technologies without infringing the Company’s intellectual property rights; |
| ● | it is possible that the Company’s pending patent applications will not lead to issued patents; |
| ● | the Company may not develop additional proprietary technologies that are patentable; |
| ● | the Company may choose not to file a patent in order to maintain certain trade secrets or know-how, and a third party may subsequently file a patent covering such intellectual property; |
| ● | the Company may fail to adequately protect and police the Company’s trademarks and trade secrets; and |
| ● | the patents of others may have an adverse effect on the Company’s business, including if others obtain patents claiming subject matter similar to or improving that covered by the Company’s patents and patent applications. |
Should any of these events occur, they could significantly harm the Company’s business, results of operations and prospects.
Obtaining and maintaining ourthe Company’s patent protection depends on compliance with various procedural, document submission, fee payment, and other requirements imposed by governmental patent agencies, and ourthe Company’s patent protection could be reduced or eliminated as a result of anyfor non-compliance with these requirements. We may also abandon certain intellectual property protections that we would otherwise maintain if we determine such protections
Periodic maintenance fees on any issued patent are not expecteddue to provide sufficient value relativebe paid to the costUSPTO and foreign patent agencies in several stages over the lifetime of ongoing maintenance.
• Patent lawsthe patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment, and other intellectual property protections availablesimilar provisions during the patent application process. Although an inadvertent lapse can, in many cases, be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the U.S., E.U.,relevant jurisdiction. Noncompliance events that could result in abandonment or other jurisdictions are subjectlapse of a patent or patent application include failure to change. These changes mayrespond to official actions within prescribed time limits, non-payment of fees, and failure to properly legalize and submit formal documents. In any such event, the Company’s competitors might be unpredictable, weaken our overall intellectual property position, increase our costs relatedable to maintenance and enforcement, or otherwise diminishenter the value of patents in general, thereby impairing our ability to protect our product candidates and maximize our returnmarket, which would have a material adverse effect on investment thereon.the Company’s business.
Risks Related to Our Business, Financial Position, Results of Operation, and Organizational StructureCommercialization
We will require additional capitalThe Company has no history of commercializing pharmaceutical products, which may make it difficult to fund our operations whichevaluate the prospects for its future viability.
The Company has not yet demonstrated, either on its own or through collaboration with third parties, an ability to successfully complete a large-scale, pivotal clinical trial, obtain marketing approval, manufacture a commercial product, or conduct sales and marketing activities necessary for successful product commercialization. Consequently, predictions about its future success or viability may not be available on acceptable terms or at all. If we fail to obtain necessary financing, weas accurate as they may be forced to delayif the Company had a longer operating history or curtail our clinical trialsa history of successfully developing and commercializing pharmaceutical products.
In addition, as a business with a limited operating history, the Company may encounter unforeseen expenses, complications, delays and other development activities orknown and unknown factors. If it is able to successfully develop neflamapimod, the Company may eventually need to transition from a company with a research focus to a company capable of supporting commercial activities. The Company may not be unablesuccessful in such a transition and, as a result, its business may be adversely affected.
As the Company continues to completebuild its business, the developmentCompany expects that its financial condition and commercialization of our product candidatesoperating results may fluctuate significantly from quarter to quarter and year to year due to a lackvariety of sufficient resources.
Although we expect that our existing cash resources will enable us to fund our operating expenses and capital expenditure requirements, including expected costs related tofactors, many of which are beyond its control. Accordingly, investors should not rely upon the planned TSC Oxygenation Trials and our Planned Phase 2 Hypoxia-related Indication Trial, through 2023, we expect to continue to spend substantial amountsresults of any particular quarterly or annual period as we continue to develop TSC and our other product candidates, we will need to obtain additional financing in the future in order to complete the development of TSC and fund our other development and operational activities.
We cannot be certain that the additional funding we will require will be available on acceptable terms or at all. Investors may demand significant discounts to market prices or that we agree to restrictive covenants or other limitations on our ability to operate our business, and conditions in the capital markets may make equity and debt financing more difficult to obtain or negatively impact our ability to complete a financing transaction at all. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us we may have to significantly delay, scale back, discontinue the development or commercialization of one or more of our product candidates, or seek alternative financing opportunities such as collaborations or licensing opportunities. For example, prior to completing the May 2020 Offering and February 2021 Offering, we determined that we did not have adequate resources to fully support the randomized portionindications of the TSC GBM Trial and commencement of enrollment in the trial was suspended following the run-in portion.
Furthermore, our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement and involves the associated risks and uncertainties. Although we have based this estimate on assumptions that we believe to be reasonable, they may prove to be wrong, we could utilize our available capital resources sooner than we currently expect, and actual results could vary greatly from our expectations expressed in this Annual Report as a result. The magnitude and timing of ourCompany’s future funding requirements, both near and long-term, will depend on many factors, including, but not limited to:operating performance.
the number, development stage, and other characteristics of product candidates that we choose to develop, including any product candidates that we may in-license or otherwise acquire in the future;
the clinical development plans we establish for these product candidates;
the magnitude of costs associated with filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights;
the initiation, progress, timing, costs, and results of clinical trials for such product candidates;
the outcome, timing and cost of regulatory approvals by the FDA and comparable foreign regulatory authorities, including the potential for the FDA or comparable foreign regulatory authorities to require that we perform more studies than those that we currently expect;
the cost and timing of completion of becoming a commercial organization; and
the effect of competing technological and market developments.
We currently generate no revenue fromThe Company’s business operations are subject to applicable healthcare laws and regulations. If neflamapimod is approved, the sale of products, have incurred significant losses since our inception, have a history of net lossesCompany will also be subject to stringent regulation and negative cash flow from operations, expectongoing regulatory obligations and restrictions, which could delay its marketing and commercialization activities and also expose it to incur losses forpenalties if the foreseeable future, and may never become profitable. In addition, our operating results may fluctuate significantly, which makes our future operating results difficultCompany fails to predict and could cause our operating results to fall below expectations. As a result, any investment in our common stock is speculative and risky.comply with applicable regulations.
We areAlthough the Company does not currently have any products on the market, once it begins commercializing neflamapimod or any other future product candidates, it will be subject to additional healthcare statutory and regulatory requirements and oversight by federal and state governments as well as foreign governments in the jurisdictions in which the Company conducts its business. Physicians, other healthcare providers and third party payors will play a clinical stage biotechnology companyprimary role in the recommendation, prescription and as a result, we have a limited operating history fromuse of any product candidates for which the Company obtains marketing approval. The Company’s future arrangements with such third parties may expose the Company to assess how we will respond to competitive, economic,broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or other challenges to our business,financial arrangements and our businessrelationships through which it markets, sells and prospects must be considered in light ofdistributes any products for which the risksCompany obtains marketing approval. Among others, restrictions under applicable domestic and uncertainties frequently encountered by similarly situated companies.foreign healthcare laws and regulations include:
We have limited cash resources, have generated substantial net losses and negative cash flow from operations since our inception, and we continue to incur significant research, development, and other expenses related to our ongoing operations, including the development of TSC and our other product candidates. To date, we have not yet obtained regulatory approvals for any of our product candidates and, accordingly, have not generated any revenues from the sale of products. We expect to continue to incur losses and negative cash flow for the foreseeable future. Furthermore, our future operating results may fluctuate due to a variety of other factors, many of which are outside of our control and may be difficult to predict, including the delays in our product development programs including as a result of regulatory review, increased expenditures related to manufacturing or the enforcement of intellectual property rights, other litigation costs, changes in accounting policies, or other unanticipated events.
Our ability to generate sufficient revenues from TSC or any of our other product candidates, if approved, will depend on numerous factors described throughout this Annual Report. Even if we are able to successfully develop and receive regulatory approval for TSC or any of our other product candidates, we do not know if or when any such product will achieve commercial success or generate revenue for us, and we will incur significant costs associated with the commercialization that will need to be offset by revenue before achieving a profit. We may also in the future enter into collaboration agreements and license agreements with other companies that include milestone expenditures and payments, in which case our ability to generate revenue or achieve profitability may be dependent on the achievement of those milestones. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods, and our prior losses and expected future losses have had and will continue to have an adverse effect on our stockholders’ equity. Furthermore, due to the uncertainty of the drug development process, we are often unable to predict the timing or amount of increased expenses, or when we will be able to achieve or maintain profitability, if at all.
We will need to further increase the size and complexity of our organization in the future including, if TSC or any of our other product candidates are approved for commercial sale, establishing sales and marketing capabilities. We may experience difficulties in executing our growth strategy or managing any growth that we do experience if we are unable to recruit and retain talented individuals in key positions.
Our ability to succeed in the highly competitive pharmaceuticals industry depends upon our ability to attract and retain highly qualified personnel. As of the date of this Annual Report, we have 13 full-time employees and one part-time employee, including our new Senior Director of Quality Assurance hired during the first quarter of 2021. Particularly given our near-term plans for the TSC development program and our compliance requirements with the FDA, SEC, and other regulatory bodies, we believe that our current staffing levels are inadequate to support our future needs. We anticipate adding additional personnel to our team throughout the organization during 2021 in an effort to support the development of TSC, grow our business more generally, and optimize the size of our organization. In addition, assuming success in our planned clinical trials, we expect to further supplement and grow our scientific, clinical, regulatory, financial, and other human resources to support our planned research, development, and commercialization plans for TSC and our other product candidates..
Our ability to effectively manage our anticipated growth will depend on multiple factors, including, among others, our ability to:
• effectively retain current talent and effectively recruit sufficient numbers of new talented employees;
| ● | the U.S. federal AKS, which prohibits, among other things, persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under a federal healthcare program such as Medicare and Medicaid; |
| ● | U.S. federal false claims, false statements and civil monetary penalties laws, including the U.S. federal False Claims Act, which impose criminal and civil penalties against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| ● | HIPAA, which imposes (i) criminal and civil liability for executing a scheme to defraud any healthcare benefit program, or knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services and (ii) obligations on certain covered entity healthcare providers, health plans, and healthcare clearinghouses as well as their business associates that perform certain services involving the use or disclosure of individually identifiable health information, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| ● | analogous state and foreign laws and regulations relating to healthcare fraud and abuse, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers; |
| ● | the U.S. federal “Physician Payments Sunshine Act”, which requires manufacturers of drugs, devices, biologics and medical supplies that are reimbursable under Medicare, Medicaid, or the Children’s Health Insurance Program to report to CMS information related to physician payments and other transfers of value to physicians, certain advanced non-physician health care practitioners, and teaching hospitals, as well as the ownership and investment interests of physicians and their immediate family members; |
| ● | analogous state and foreign laws that require pharmaceutical companies to track, report and disclose to the government or the public information related to payments, gifts, and other transfers of value or remuneration to physicians and other healthcare providers, marketing activities or expenditures, or product pricing or transparency information, or that require pharmaceutical companies to implement compliance programs that meet certain standards or to restrict or limit interactions between pharmaceutical manufacturers and members of the healthcare industry; |
| ● | U.S. federal laws that require pharmaceutical manufacturers to report certain calculated product prices to the government or provide certain discounts or rebates to government authorities or private entities, often as a condition of reimbursement under federal healthcare programs; and |
| ● | state and foreign laws that govern the privacy and security of health information in certain circumstances, including state security breach notification laws, state health information privacy laws and federal and state consumer protection laws, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
• manage our third-party supplyEnsuring business arrangements comply with applicable healthcare laws, as well as responding to possible investigations by government authorities, can be time- and manufacturing operations effectivelyresource-consuming and can divert a company’s attention from the business. Efforts to ensure that the Company’s business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of health care reform, including due to lack of applicable precedent and regulations. Any action against the Company for violation of these laws, even if the Company successfully defends against it, could cause the Company to incur significant legal expenses and divert its management’s attention from the operation of its business. The shifting compliance environment and the need to build and maintain robust and expandable systems to comply with multiple jurisdictions with different compliance or reporting requirements increases the possibility that a cost-effective manner,
• while increasing production capabilities for our currenthealth care company may run afoul of one or more of the requirements. If the FDA or a comparable foreign regulatory authority approves any of the Company’s product candidates, the Company will be subject to commercial levels;
• establishan expanded number of these laws and maintain relationshipsregulations and will need to expend resources to develop and implement policies and processes to promote ongoing compliance. It is possible that governmental authorities will conclude that the Company’s business practices may not comply with developmentcurrent or future statutes, regulations or case law involving applicable fraud and commercialization partners
• manage our developmentabuse or other healthcare laws and commercialization efforts effectively andregulations, resulting in a cost-effective manner; and
• continue to improve our operational, clinical, financial, management and regulatory compliance controls and reporting systems and procedures;government enforcement actions.
WeIf the Company’s operations are highly dependent on our management and scientific personnel, including our executive officers, certain other key employees and consultants, and the members of our Board. The loss of the servicesfound to be in violation of any of these individuals could impede, delay, or prevent completion of our planned clinical trials, regulatory approval, or commercialization of TSClaws or any other governmental regulations that may apply to the Company, it may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion of ourproducts from federal healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of the Company’s operations. If any of the physicians or other healthcare providers or entities with whom the Company expects to do business is found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from federal healthcare programs.
Even if neflamapimod or any other product candidates. If we losecandidate the servicesCompany develops receives marketing approval, it may fail to achieve the level of any of these individuals, we might not be able to find suitable replacements on a timely basis or at all, and our business could be harmed as a result. All of our employees, including our executive officers with whom we have employment agreements, are employed on an at-will basis and their employment can be terminated by us or them at any time. As part of our efforts to retain our valuable employees, we, among other things, provide a generous salary and benefits package, as described in more detail in Part I acceptance necessary for commercial success.— Item 1. Business – Our People and Human Capital Resources of this Annual Report
Nevertheless, weIf neflamapimod, or any other product candidate the Company may be unable to attractdevelop or retain qualified management and other key personnelacquire in the future, duereceives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, health care professionals, patients, third-party payors and others in the intense competition among biotechnology, pharmaceutical, and other businesses. Theremedical community. If the Company’s drug does not achieve an adequate level of acceptance, the Company may benot generate significant product revenues or become profitable. The degree of market acceptance will depend on a limited number of personsfactors, including but not limited to:
| ● | the ability to provide acceptable evidence of efficacy and potential advantages compared to alternative treatments; |
| ● | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| ● | the Company’s ability to offer its drug for sale at competitive prices, which may be subject to regulatory control; |
| ● | the availability of third-party insurance coverage and adequate reimbursement; |
| ● | the availability of alternative treatments and the cost of a new treatment in relation to those alternatives, including any similar generic treatments; |
| ● | the relative convenience and ease of administration of a new treatment compared to alternatives, and the prevalence and severity of any side effects of a new treatment; |
| ● | the strength and effectiveness of the Company’s sales, marketing and distribution capabilities, either internally or in collaboration with others; |
| ● | any restrictions on the use of the Company’s product together with other medications; and |
| ● | any restrictions on the distribution of the Company’s product such as those imposed under a mandatory REMS program. |
If neflamapimod or any other product candidate that the requisite skills to serveCompany may develop in these positions, and we cannot assure youthe future does not provide a treatment regimen that weis at least as beneficial as the current standard of care or otherwise does not provide some additional patient benefit over the current standard of care, that product will be able to identify or employ qualified personnel for any such position on acceptable terms, if at all,not achieve market acceptance, and the high levelsCompany will not generate sufficient revenues to achieve profitability. Because the Company expects sales of competition within the industry may mean that we will be required to expend significant financial resources in our employee recruitment and retention efforts. Many of the other pharmaceutical companies with whom we compete for qualified personnel have greater financial and other resources, different risk profiles and longer histories in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. If we are not able to attract and retain the necessary personnel to accomplish our business objectives, we may experience constraints that will harm our ability to implement our business strategy and achieve our business objectives. Furthermore, as we currently have no marketed products, we currently have no sales or marketing personnel or capabilities. To commercialize TSC or any of our otherits product candidates, if approved, into generate substantially all of its revenues for the we will need to build our marketing, sales, distribution, and other related capabilities or arrange with third parties to perform these services, and we may not be successful in doing so.
In addition, we have historically utilizedforeseeable future, the servicesfailure of certain outside independent contractors to perform a number of critical functions for our company, including with respect to clinical development, regulatory matters, accounting, and human resources, a practice we expect to continue and may choose to expand in the future. We rely on these independent contractors and effectively managing our relationships with them is and will remain a priority. However, there can be no assurance that we will be able to manage these relationships effectively, that such contractors will be able or choose to continue working with us in the future, or that we will be able to find additional or replacement services if and as needed, on economically reasonable terms or at all.
If we are not able to effectively manage our growth and expand our organization through a combination of effectively retaining our existing employees and third-party contractors and successfully recruiting new employees and contractors, we may be unable to effectively execute on our product development and other strategic plans, which my adversely effect our business, financial condition, or results of operations.
If we decide to in-license or acquire one or more additional product candidates or otherwise enter into a strategic transaction, it could impact our liquidity, increase our expenses, and present significant distractions to our management team.
We currently only have two product candidates under active development, our lead product candidate, TSC, and our second product candidates, DFN-529. We may in the future implement a strategy to in-license or acquire one or more additionalCompany’s product candidates to supplement our pipeline. We may also consider a variety of other strategic transactions, including spin-offs, partnerships, joint ventures, restructurings, divestitures, business combinations, and minority investments. Any such transactionfind market acceptance would expose us to a number of risks and uncertainties, including the potential incurrence of recurring, non-recurring (including unknown liabilities), or other charges (including amortization expenses, write-downs, or other impairment charges), increase of short- and long-term expenditures, or dilution our stockholders, as well as posing significant integration, implementation, or retention challenges and diverting our management team’s focus on other priorities, including the TSC development program. Any of the foregoing could have a material adverse effect on our business, financial condition, or results of operation. which could adversely affect our operations and financial results. There can be no assurance that we will undertake such a transaction or, if we do, that we will successfully complete the transaction or that the transaction will be additive to our business, financial condition, or results of operations.materially harm its business.
Our ability to utilize our NOL carryforwards and other deferred tax assetsIf the market opportunity for any product candidate that the Company develops is smaller than it believes, its revenue may be limited as a result of pastadversely affected and future issuances of our common stock.its business may suffer.
AsThe Company intends to initially focus its product candidate development on treatments for various CNS and neurodegenerative indications, in particular DLB. The addressable patient populations that may benefit from treatment with the Company’s product candidates, if approved, are based on its estimates. These estimates, which have been derived from a variety of December 31, 2020, we had $11.7 million in federalsources, including scientific literature, surveys of clinics, patient foundations and state NOL carryforwards availablemarket research, may prove to reduce future taxable income, if any, for income tax purposes. If not utilized,be incorrect. Further, new studies may change the NOL carryforwards will begin expiring during the year ending December 31, 2034. Under Section 382estimated incidence or prevalence of these CNS and neurodegenerative diseases. Any regulatory approval of the Tax Code,Company’s product candidates would be limited to the therapeutic indications examined in the Company’s clinical trials and as determined by the FDA, which would not permit the Company to market its products for any other therapeutic indications not expressly reviewed and approved as safe and effective. Additionally, the potentially addressable patient population for the Company’s product candidates may not ultimately be amenable to treatment with the Company’s product candidates. Even if a corporation undergoes an “ownership change,” generally defined as a greater than 50.0% change, measuredthe Company receives regulatory approval for any of its product candidates, such approval could be conditioned upon label restrictions that materially limit the addressable patient population. The Company’s market opportunity may also be limited by value, in its equity ownership over a three-year period,future competitor treatments that enter the corporation’s ability to use its pre-ownership change NOL carryforwards and other pre-ownership change tax attributes – such as research tax credits – to offset its post-ownership change income may be limited. In the year ended December 31, 2019, as a resultmarket. If any of the issuance of a significant number of shares our common stock pursuantCompany’s estimates prove to be inaccurate, the May 2019 Offering, November 2019 Offering, and December 2019 Offering, we performed an analysis under Section 382 ofmarket opportunity for any product candidate that the Tax Code and, as a result of such analysis and a determination that such an ownership change had taken place, we reduced our NOL carryforwards. In the event we issue additional shares of common stock to fund our future development efforts and those issuances result in additional ownership changes for purposes of Section 382 of the Tax Code, as we did in the May 2020 Offering and the February 2021 Offering, our NOL carryforwardsCompany or its strategic partners develop could be further reduced.significantly diminished and have an adverse material impact on its business.
General Risks RelatedThe Company faces substantial competition from other biotechnology and pharmaceutical companies, and its operating results will suffer if it fails to Our Business, Financial Condition, Results of Operations, and Organizational Structurecompete effectively.
Our business,The biotechnology and pharmaceutical industries are highly competitive and subject to significant and rapid technological change. If neflamapimod is approved, it will face intense competition from a variety of businesses, including large, fully integrated pharmaceutical companies, specialty pharmaceutical companies, biopharmaceutical companies in the U.S. and other jurisdictions, academic institutions and governmental agencies and public and private research institutions. These organizations may have significantly greater resources than the Company does. They may also conduct similar research, seek patent protection, and establish collaborative arrangements for research, development, manufacturing and marketing of products that may compete with neflamapimod.
Currently, there are a limited number of companies and disease modifying approaches for DLB. However, given the potential market opportunity for the treatment of DLB and other neurodegenerative diseases, an increasing number of established pharmaceutical firms and smaller biotechnology/biopharmaceutical companies are pursuing a range of potential therapies for these diseases in various stages of clinical development. In addition to these current and potential competitors, the Company anticipates that more companies will enter the DLB market in the future. The Company’s potential competitors could have significantly greater financial condition, or results of operationsresources, as well as drug development, manufacturing, marketing, and sales expertise. They may also be materially adversely affected by a number of general risks related theretoable to develop and to our organizational structurecommercialize products that are not specificsafer, more effective, less expensive, more convenient, easier to ouradminister, or have fewer severe effects, than existing treatments or, if it is ultimately approved, neflamapimod. Competitors may also obtain FDA or other regulatory approval for their product candidates more rapidly than the Company including:
• If we fail to maintain an effective system of internal control over financial reporting, we may not beobtain approval for neflamapimod, which could result in their establishing or strengthening a commercial position before the Company is able to accurately report our financial results or prevent fraud. Furthermore, our disclosure controlsenter the market. The highly competitive nature of the biotechnology and procedures are subjectpharmaceutical industries, as well as the rapid technological changes in those fields, could limit The Company’s ability to inherent limitations, human error,advance neflamapimod commercially. If the Company is unable to compete effectively, this could have a material adverse effect on its business and other systematic breakdowns, and therefore may not prevent or detect all errors or acts of fraud. As a result, stockholders could lose confidence in our financial and other public reporting, which could harm our business, financial condition, or results of operations.
• As our Company, the industry in which we operate, and the world-at-large become increasingly virtual, our acquisition and implementation of additional information technology solutions and our compliance with global privacy and data security requirements could result in additional costs and liabilities or inhibit our ability to collect and process data globally. Furthermore, any failure to comply with applicable requirements or best practices – as well as other events outside of our control – could result in a security breach or other disruption to our information technology systems, limit our capacity to effectively monitor and control our operations, compromise our or third parties’ confidential information, or otherwise adversely affect our business, financial condition, or results of operations.
• We incur significant costs as a result of our public company status and devote substantial management time to operating as a public company, including complying with the applicable requirements of the Securities Act, the Exchange Act, the Dodd-Frank Act, SOX, and the rules and regulations of Nasdaq. If, in the future, we are required to include in our annual report an attestation of our independent registered public accounting firm regarding internal control over financial reporting, the amount of these compliance costs would increase significantly.
• Although we have in place business continuity and disaster recovery plans, our business, financial condition, or results of operations could be negatively affected by volatility, disruptions, or other uncertainty caused by market fluctuations, economic downturns or unfavorable global economic conditions, pandemics, natural disasters or other catastrophic events, events of war, terrorism, or other man-made problems, or other geopolitical events outside of our control, including the COVID-19 pandemic and Brexit.
• If we fail to comply with applicable laws and regulations, including the healthcare laws and regulations described in Part I – Item 1. Business – Certain Other Legislation and Regulations – Current Healthcare Laws and Regulations of this Annual Reportand applicable environmental, health, and safety laws and regulations, we could become subject to fines, penalties, or other consequences.
The successful commercialization of neflamapimod, or any other product candidate the Company may develop or acquire, will depend in part on the extent to which governmental authorities and health insurers establish adequate coverage, reimbursement levels, and pricing policies. Enacted and future healthcare legislation may increase the difficulty and cost for the Company to obtain marketing approval of and commercialize its product candidates, if approved, and also affect the prices it may set. Failure to obtain or maintain coverage and adequate reimbursement for the Company’s product candidates, if approved, could limit its ability to market those products and decrease its ability to generate revenue.
There have been, and the Company expects will continue to be, a number of legislative and regulatory proposals and changes to the healthcare systems in the U.S. and other jurisdictions that could affect the Company’s future results of operations. In particular, a number of initiatives at the U.S. federal and state levels have aimed to reduce healthcare costs and improve the quality of healthcare. Existing regulatory policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of neflamapimod or any future product candidates the Company may develop or acquire. The Company cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the U.S. or abroad. If the Company is slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if it is not able to maintain regulatory compliance, the Company may lose any marketing approval that it may have obtained, and it may not achieve or sustain profitability.
In the U.S., the availability and adequacy of coverage and reimbursement by governmental healthcare programs such as Medicare and Medicaid, private health insurers, and other third-party payors are essential for most patients to be able to afford prescription medications such as neflamapimod, if it is approved. The Company’s ability to achieve acceptable levels of coverage, payment, and reimbursement for products by governmental authorities, private health insurers and other organizations will have an effect on the Company’s ability to successfully commercialize neflamapimod and any other potential future product candidates. Assuming the Company obtains coverage for neflamapimod by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. the Company cannot be sure that coverage, payment, and reimbursement in the U.S. or elsewhere will be available for or any drug product that the Company may develop, and any reimbursement that may become available may be decreased or eliminated in the future.
There have recently been and may continue to be a number of significant legislative initiatives in the U.S. to contain healthcare costs. Federal and state governments continue to propose and pass legislation designed to reform delivery of, or payment for, healthcare, which include initiatives to reduce the cost of healthcare. For example, in March 2010, the U.S. Congress enacted the ACA, which substantially changed the way healthcare is financed by both the government and private insurers, and significantly impacts the U.S. pharmaceutical industry. We expect that future changes or additions to the ACA, the Medicare and Medicaid programs, and changes stemming from other healthcare reform measures, especially with regard to healthcare access, financing or other legislation in individual states, could have a material adverse effect on the healthcare industry in the United States.
In August 2022, President Biden signed into law the IRA, which, among other things, requires manufacturers of certain drugs to engage in price negotiations with Medicare (beginning in 2026), with prices that can be negotiated subject to a cap; imposes rebates under Medicare Part B and Medicare Part D to penalize price increases that outpace inflation (first due in 2023); and replaces the Part D coverage gap discount program with a new discounting program (beginning in 2025). The IRA permits the Secretary of the Department of Health and Human Services to implement many of these provisions through guidance, as opposed to regulation, for the initial years. In addition, multiple large pharmaceutical companies and other stakeholders (e.g., the U.S. Chamber of Commerce) have initiated federal lawsuits against CMS arguing the program is unconstitutional for a variety of reasons, among other complaints. For these and other reasons, the implementation of the IRA and its impact on the Company’s business is currently unclear.
At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures. In December 2020, the U.S. Supreme Court held unanimously that federal law does not preempt the states’ ability to regulate PBMs and other members of the health care and pharmaceutical supply chain, an important decision that may lead to appears to be leading to further and more aggressive efforts by states in this area. The Federal Trade Commission in mid-2022 also launched sweeping investigations into the practices of the PBM industry, and members of Congress continue to propose reforms for the PBM industry, all or each of which could lead to additional federal and state legislative or regulatory proposals targeting such entities’ operations, pharmacy networks, or financial arrangements. In addition, in the last few years, several states have formed PDABs with the authority to implement UPLs on drugs sold in their respective jurisdictions. There are several pending federal lawsuits challenging the authority of states to impose UPLs, however.
Further, if neflamapimod is approved in any jurisdictions outside of the U.S., the Company may also be subject to extensive governmental price controls and other market regulations in those countries. Governments outside of the U.S., particularly the countries of the European Union, tend to impose strict price controls on prescription pharmaceutical products. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a product. To obtain reimbursement or pricing approval in some countries, the Company may be required to conduct a clinical trial that compares the cost-effectiveness of its product candidate to other available therapies. If reimbursement of the Company’s products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, the Company’s business could be harmed, possibly materially. As a result, the Company might obtain regulatory approval for a product in a particular country, but then be subject to price regulations that delay its commercial launch of the product and negatively impact the revenue the Company is able to generate from the sale of the product in that country. Adverse pricing limitations may hinder the Company’s ability to recoup its investment in its product candidates, even after obtaining regulatory approval.
The Company cannot predict the likelihood, nature, or extent of government regulation that may arise from future legislation or administrative action in the U.S. or any other jurisdiction. In the U.S., future laws and regulation may result in more rigorous coverage criteria and increased downward pressure on the price pharmaceutical companies may receive for any approved product. Reductions in reimbursement from Medicare or other government programs may result in similar reductions in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent the Company from being able to generate revenue, attain profitability or commercialize its product candidates. Further, if the Company or any third parties with whom it engages in the future are slow or unable to adapt to changes in existing requirements or policies, or if the Company is not able to maintain regulatory compliance, its ability to generate revenue, attain profitability, or commercialize neflamapimod or any other products for which it receives regulatory approval may be materially and adversely affected.
If the Company is unable to obtain adequate coverage and payment levels for its products from third-party payors, physicians may limit how much or under what circumstances they will prescribe or administer them, and patients may decline to purchase them. This in turn would affect the Company’s ability to successfully commercialize any approved products and thereby adversely impact its profitability, results of operations, and financial condition.
If the Company is unable to establish sales, marketing and distribution capabilities either on its own or in collaboration with third parties, it may not be successful in commercializing neflamapimod, if approved.
The Company does not currently have any infrastructure for the sales, marketing or distribution of an approved drug product, and the cost of establishing and maintaining such an organization may exceed the cost-effectiveness of doing so. In order to market and successfully commercialize neflamapimod, if approved, the Company must build its sales, distribution, marketing, managerial and other non-technical capabilities, or make arrangements with third parties to perform these services.
There are significant expenses and risks involved in establishing the Company’s own sales, marketing and distribution functions, including the Company’s ability to hire, retain and appropriately incentivize qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel, and effectively manage a geographically dispersed sales and marketing team. Alternatively, to the extent that the Company depends on third parties for such services, any revenues it receives will depend upon the efforts of those third parties, and there can be no assurance that such efforts will be successful.
If the Company is unable to establish adequate sales, marketing and distribution capabilities, either on its own or in collaboration with others, the Company will not be successful in commercializing neflamapimod, if it is ultimately approved, and it may never become profitable. The Company will be competing with companies that currently have extensive and well-funded marketing and sales operations. Without an internal team or the support of a third party to perform marketing and sales functions, the Company may be unable to compete successfully against these more established companies.
Consumers may sue the Company for product liability, which could result in substantial liabilities that exceed its available resources and damage its reputation.
Researching, developing, and commercializing drug products entail significant product liability risks. The use of neflamapimod or any other product candidates the Company may develop in clinical trials and the sale of any products for which it obtains marketing approval exposes it to the risk of product liability claims. Product liability claims might be brought against the Company by clinical trial participants, patients, healthcare providers, pharmaceutical distributors or others selling or otherwise coming into contact with its product candidates or future commercial products. The Company has obtained limited product liability insurance coverage for its clinical trials, which the Company believes to be reasonable given its current operations. However, the Company’s insurance coverage may not reimburse the Company or may not be sufficient to reimburse it for any expenses or losses it may suffer.
Although the Company currently has limited product liability insurance that covers its clinical trials, it will need to increase and expand this coverage as it commences larger scale trials, as well as if neflamapimod is ultimately approved for commercial sale. This insurance may be extremely expensive or may not fully cover the Company’s potential liabilities. Inability to obtain sufficient insurance coverage at an acceptable cost or otherwise to protect against potential product liability claims could prevent or inhibit the commercialization of neflamapimod, if it is approved. Product liability claims could have a material adverse effect on the Company’s business and results of operations.
Any product candidate for which the Company obtains marketing approval will be subject to extensive post-marketing regulatory requirements and could be subject to post-marketing restrictions or withdrawal from the market, and the Company may be subject to penalties if it fails to comply with regulatory requirements or if it experiences unanticipated problems with its products, when and if any of them are approved.
If the FDA or a comparable foreign regulatory authority approves neflamapimod or any of the Company’s future product candidates for marketing, activities such as the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for the product will be subject to extensive and ongoing regulatory requirements. The FDA or a comparable foreign regulatory authority may also impose requirements for costly post-marketing nonclinical studies or clinical trials (often called “Phase 4 trials”) and post-marketing surveillance to monitor the safety or efficacy of the product. If the Company or a regulatory authority discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, production problems or issues with the facility where the product is manufactured or processed, such as product contamination or significant not-compliance with applicable cGMPs, a regulator may impose restrictions on that product, the manufacturing facility or the Company. If the Company or its third party providers, including the Company’s CMOs, fail to comply fully with applicable regulations, then the Company may be required to initiate a recall or withdrawal of its products.
The Company must also comply with requirements concerning advertising and promotion for any of its product candidates for which it obtains marketing approval. Promotional communications with respect to prescription drugs are subject to a variety of legal and regulatory restrictions and must be consistent with the information in the product’s approved labeling. Thus, the Company will not be able to promote any products it develops for indications or uses for which they are not approved. The FDA and other agencies closely oversee the post-approval marketing and promotion of drugs to ensure drugs are marketed only for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on manufacturers’ communications regarding use of their products, and if the Company promotes its products beyond their approved indications, it may be subject to enforcement actions or prosecution arising from that off-label promotion. Violations of the FDCA relating to the promotion of prescription drugs may lead to investigations alleging violations of federal and state healthcare fraud and abuse laws, as well as state consumer protection laws. Accordingly, to the extent the Company receives marketing approval for neflamapimod, the Company and its CMOs and other third-party partners will continue to expend time, money and effort in all areas of regulatory compliance, including promotional and labeling compliance, manufacturing, production, product surveillance, and quality control. If the Company is not able to comply with post-approval regulatory requirements, it could have marketing approval for any of its products withdrawn by regulatory authorities and its ability to market any future products could be limited, which could adversely affect its ability to achieve or sustain profitability. Thus, the cost of compliance with post-approval regulations may have a negative effect on the Company’s operating results and financial condition.
The FDA’s policies may change and additional government regulations may be enacted that could prevent, limit or delay marketing approval of the Company’s product candidates. If the Company is slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if it is not able to maintain regulatory compliance, it may lose any marketing approval that it may have obtained, which would adversely affect the Company’s business, prospects and ability to achieve or sustain profitability.
Risks Related to Ownership of Our Common Stockthe Company’s Securities
OurThe Company’s stock price ismay be volatile, there may be limited liquidity in the trading market for the Company’s common stock, and any investmentthe market price of its common stock may drop in our securities may suffer a decline in value. In addition, this volatility may subject our business to additional risks, such as an increased risk of securities litigation or inability to meet the continued listing requirements of Nasdaq.future.
During the year ended December 31, 2020, the closing market price for our common stock varied between a high of $1.50 on July 21, 2020 and a low of $0.26 on March 16, 2020. As a result of fluctuations in the price of our common stock, you may be unable to sell shares or our common stock at or above the price you paid for them, even if your holding period is relatively short. The market price of ourthe Company’s common stock is likely to continue tomay be volatile and subject to significant price and volume fluctuations in response to market, industryfluctuations. Market prices for securities of early-stage pharmaceutical, biotechnology and other life sciences companies have historically been volatile. Some of the factors including the degree of analyst coverage of our stock, their valuations and recommendations, and whether any such analysts publish inaccurate or unfavorable research about our business. If the results of our business do not meet these analysts’ forecasts, the expectations of investors or the financial guidance we provide to investors in any period,that may cause the market price of ourthe Company’s common stock could decline. Furthermore, despite this volatility, due to fluctuate include among others:
| ● | the ability of the Company or its partners to develop product candidates and conduct clinical trials that demonstrate such product candidates are safe and effective; |
| ● | the ability of the Company or its partners to obtain regulatory approvals for product candidates, and delays or failures to obtain such approvals; |
| ● | failure of any of the Company’s product candidates to demonstrate safety and efficacy, receive regulatory approval and achieve commercial success; |
| ● | failure by the Company to maintain its existing third-party license, manufacturing and supply agreements; |
| ● | failure by the Company or its licensors to prosecute, maintain, or enforce its intellectual property rights; |
| ● | changes in laws or regulations applicable to the Company’s product candidates; |
| ● | any inability to obtain adequate supply of product candidates or the inability to do so at acceptable prices; |
| ● | adverse regulatory authority decisions; |
| ● | introduction of new or competing products by the Company’s competitors; |
| ● | failure to meet or exceed financial and development projections the Company may provide to the public; |
| ● | the perception of the pharmaceutical industry by the public, legislatures, regulators and the investment community; |
| ● | announcements of significant acquisitions, strategic partnerships, joint ventures, or capital commitments by the Company or its competitors; |
| ● | disputes or other developments relating to proprietary rights, including patents, litigation matters, and the Company’s ability to obtain intellectual property protection for its technologies; |
| ● | additions or departures of key personnel; |
| ● | significant lawsuits, including intellectual property or stockholder litigation; |
| ● | if securities or industry analysts do not publish research or reports about the Company, or if they issue an adverse or misleading opinions regarding its business and stock; |
| ● | changes in the market valuations of similar companies; |
| ● | general market or macroeconomic conditions; |
| ● | sales of its common stock by the Company or its stockholders in the future; |
| ● | the trading volume of the Company’s common stock; |
| ● | the limited percentage of the Company’s outstanding shares that are currently freely tradeable as a result of the significant holdings of the Company’s directors and officers; |
| ● | adverse publicity relating to the Company’s markets generally, including with respect to other products and potential products in such markets; |
| ● | changes in the structure of health care payment systems; and |
| ● | period-to-period fluctuations in the Company’s financial results. |
Accordingly, the fact that we have never declared or paid cash dividends on ourmarket price of Company’s common stock may be highly volatile and do not currently anticipate declaringcould fluctuate widely in price as a result of these or paying any cash dividendsother factors. In particular, the Company has relatively few shares of common stock outstanding in the foreseeable future, we expect that only appreciation“public float” as a higher percentage of the Company’s outstanding shares are held by a small number of shareholders. In addition, the shares of common stock may be sporadically or thinly traded. As a consequence of this lack of liquidity, the trading of relatively small quantities of shares by shareholders may disproportionately influence the price of ourthose shares in either direction, particularly over short periods of time. The price for such shares could, for example, decline precipitously in the event that a large number of the shares are sold on the market without commensurate demand, as compared to a seasoned issuer which could better absorb those sales without a material reduction in share price. An active trading market for the Company’s shares of common stock if any, will provide a returnmay never develop or be sustained. If an active market for its common stock does not develop or is not sustained, it may be difficult for its stockholders to our stockholders for the foreseeable future.sell their shares at an attractive price or at all.
Historically,Additionally, in the stock markets in general, and the markets for biotechnology stocks in particular,past, plaintiffs have experienced significant volatility that has at times been unrelated to the business, financial condition, or results of operations of particular companies. These broad market fluctuations may adversely affect the trading price of our common stock and, consequently, adversely affect the price at which you are able to sell any shares of our common stock that you own. In the past,often initiated securities class action litigation against a company following periods of volatility in the market or significant price declinesof its securities. The Company may in individual securities or the market as a whole, securities class-action litigation has often been instituted against companies. Suchfuture be the target of similar litigation if instituted against us,its stock continues to experience price volatility. Securities litigation could result in substantial costs and diversion ofliabilities and could divert management’s attention and resources, which could materially and adversely affect our business, financial condition, or results of operations.
In addition, our common stock is currently listed on the Nasdaq Capital Market. To maintain the listing of our common stock on the Nasdaq Capital Market, we are required to meet certain listing requirements, including, among others, maintaining a minimum closing bid price for our common stock. Particularly given the historical volatility experienced by our common stock, there is no assurance that we will continue to meet the minimum closing price requirement and other listing requirements of Nasdaq.resources.
We haveThe Company has funded ourits operations to date through the issuance of securities, including common stock, warrants to purchase common stock (including pre-funded warrants), convertible preferred stock, and convertible debt securities, and we expectexpects that in the future weit will need to raise additional capital through similar means to fund ourits continued development efforts for TSCoperations and our other liquidity needs. Assuming funding is available on acceptable terms, any future issuance of common stock or securities convertible for or exchangeable into common stock will result in dilution to ourthe Company’s existing stockholders and could depress the market price of ourits common stock. Furthermore, the terms of future financing transactiontransactions may contain provisions that restrict our operations or require us to relinquish certain rights to our product candidates or other technologies.
Although we believe we have sufficient cash resources to fund the TSC Oxygenation Trials, our Planned Phase 2 Hypoxia-related Indication Trial, and our other operating expenses and capital expenditure requirements through 2023, weThe Company will in the futurelikely need to raise additional funds in the future to continue ourits operations, fund additional clinical trials evaluating TSCresearch and our other product candidates,development, and, if approved, commercializing TSC. We plancommercialize its product candidates. The Company currently plans to continue to finance our operations with a combination of equity issuances, debt arrangements, and, potentially, licensing, or other partnering relationships. In addition, ourThe Board may determine at any time to raise additional capital if it believes the terms are in the best interests of the Company’s stockholders. In addition, the Company may also issue securities to counterparties as part of an acquisition, merger, or similar transaction, including as part of our stockholders.strategic review process.
Accordingly, new issuances of a substantial number of shares of our common stock could occur at any time. For example, in February 2021, we issued 33,658,538 shares of our common stock in connection with the completion of the February 2021 Offering. Any issuance or sale of shares, or the perception in the market of an intent to issue or sell shares in the near-term, by the Company or holders of a large number of shares could reduce the market price of ourthe Company’s common stock. Westock, including in connection with the 2024 Private Placement which is expected to close on or about April 1, 2024, subject to customary closing conditions. The Company also cannot assure you that any such sale of common stock or other securities will be at a price per share that is equal to or greater than the price per share paid by you for ourthe Company’s common stock. Furthermore, a depressed stock price could limit ourthe Company’s ability to raise necessary capital through the sale of additional equity securities on terms that are acceptable.
In addition, in connection with the 2024 Private Placement, the Company issued the Series A Warrants pursuant to which the holders thereof are entitled to purchase an aggregate of 2,532,285 shares of common stock at an exercise price equal to $39.24 per share. The Series A Warrants are exercisable immediately and will expire at the earlier of (i) April 1, 2027 or (ii) 180 days after the date that the Company makes a public announcement of positive top-line data from the RewinD-LB Trial, subject to certain beneficial ownership limitations and other conditions set forth therein. Any future issuance of common stock upon exercise of the Series A Warrants will result in dilution to the Company’s existing stockholders and could depress the market price of its common stock.
The Company may become obligated to pay liquidated damages if we fail to file, obtain effectiveness and maintain effectiveness of a registration statement in accordance with the terms of the securities purchase agreement related to its 2024 Private Placement.
In connection with the 2024 Private Placement, the Company granted the purchasers of securities in the offering certain resale registration rights pursuant to the terms of the securities purchase agreement. In addition to the registration rights, the purchaser may be entitled to receive liquidated damages upon the occurrence, or failure to occur, of a number of events relating to the filing, effectiveness and maintenance of effectiveness of a registration statement related to the common stock sold in the 2024 Private Placement. The liquidated damages will be payable upon the occurrence, or failure to occur, of each of those events and each monthly anniversary thereof until cured. The amount of liquidated damages payable per monthly period would be equal to 1% of the aggregate purchase price paid by the purchaser, provided, however, the maximum aggregate liquidated damages payable to the purchaser would be 5% of the aggregate amount paid by such purchaser for the purchaser of such securities in the 2024 Private Placement.
Ownership of the Company’s common stock is highly concentrated among its officers and directors, which may prevent the Company’s stockholders from influencing significant corporate decisions and may result in perceived conflicts of interest that could cause the Company stock price to decline.
As of March 26, 2024, executive officers and directors of the Company owned, directly or indirectly, approximately 47.4% of the outstanding shares of the Company common stock. Accordingly, these stockholders, in the aggregate, may exercise substantial influence over the outcome of corporate actions requiring stockholder approval, including the election of directors, any merger, consolidation or sale of all or substantially all of the Company assets or any other significant corporate transactions. These stockholders may also delay or prevent a change of control of the Company, even if such a change of control would benefit the other stockholders of the Company. The significant concentration of stock ownership may adversely affect the trading price of the Company’s common stock due to investors’ perception that conflicts of interest may exist or arise.
Future sales of shares by existing stockholders could cause the Company’s stock price to decline.
If existing stockholders of the Company sell, or indicate an intention to sell, substantial amounts of the Company’s common stock in the public market after certain legal and contractual restrictions on resale lapse, the trading price of the common stock of the Company could decline.
If equity research analysts do not publish research or reports, or publish unfavorable research or reports, about the Company, its business, or its market, its stock price and trading volume could decline.
The trading market for the Company’s common stock will be influenced by the research and reports that equity research analysts may publish about it and its business from time to time. Equity research analysts may elect not to provide or continue research coverage of the Company’s common stock, which may adversely affect the market price of the stock. In the event the Company does have equity research analyst coverage at any given time, the Company will not have any control over the analysts, or the content and opinions included in their reports. The price of the Company’s common stock could decline if one or more equity research analysts downgrade its stock or issue other unfavorable commentary or research. If one or more equity research analysts cease coverage of the Company or fails to publish reports on the Company regularly, demand for the Company’s common stock could decrease, which in turn could cause its stock price or trading volume to decline.
We may also seek additional capital throughIf the Company cannot continue to satisfy the Nasdaq Capital Market continued listing standards and other methods, either alone or in combination with the issuance of additional securities, including debt financings, receivables or royalty financings, strategic partnerships and alliances, and licensing arrangements, any ofNasdaq rules, its common stock could be delisted, which could be coupled with an equity component, such as warrants to purchase stock. The incurrence of indebtedness could result in increased fixed payment obligations, liens and other security interests being placed on certain of our assets, and certain restrictive covenants being imposed onharm the operation of ourCompany’s business, such as limitations on our ability to incur additional debt or acquire intellectual property rights. We may also in the future raise additional funds through strategic partnerships, alliances, and licensing arrangements with third parties, any of which could require us to relinquish valuable rights to TSC or our other product candidates. The restrictions imposed by any of these arrangement could materially decrease any potential returns on our investment in TSC or our other product candidates, or otherwise materially and adversely effect our business, financial condition, or results of operations.
Our organizational documents impose certain anti-takeover provisions and make the Delaware Chancery Court the exclusive forum for certain stockholder actions, which could depress the trading price of ourits common stock, the Company’s ability to raise additional capital and the liquidity of the market for its common stock.
OurThe Company’s common stock is currently listed on the Nasdaq Capital Market. To maintain this listing, the Company is required to meet certain listing requirements related to, among other things, the trading price of the Company’s common stock, the Company’s market capitalization and certain corporate governance-related requirements. In the event the Company’s common stock is delisted from Nasdaq for a failure to meet such requirements and is not eligible for quotation or listing on another market or exchange, trading of the Company’s common stock could be conducted only in the over-the-counter market or on an electronic bulletin board established for unlisted securities such as the Pink Sheets or the OTC Bulletin Board. In such event, it could become more difficult for the Company to raise capital and for the Company’s stockholders to dispose of, or obtain accurate price quotations for, the Company’s common stock. There would likely also be a decline in the liquidity of the trading market for the Company’s common stock and a reduction in the Company’s coverage by securities analysts and the news media, which could cause the price of the Company’s common stock to decline further.
Provisions in the Company’s corporate charter documents and under Delaware law could make an acquisition of the Company, which may be beneficial to the Company’s stockholders, more difficult and may prevent attempts by the Company’s stockholders to replace or remove its current directors and members of management.
Provisions in the Company’s certificate of incorporation, as amended, and our Bylaws contain provisions thatits amended and restated bylaws may make thediscourage, delay or prevent a merger, acquisition of our company, a proxy contest, or the nomination of a director candidate by a stockholder more difficult than such actions would beother change in the absence of such provisions, including that:
only our Board has the right to fill a vacancy on the Board created by an expansion or by the resignation, death, or removal of a director, which prevents stockholders from being able to fill vacancies on our Board;
only our Chairmancontrol of the Board, our Chief Executive Officer, orCompany that stockholders may consider favorable, including transactions in which the Company’s stockholders might otherwise receive a majority of our directorspremium for their shares. These provisions could also limit the price that investors are authorizedwilling to call a special meeting of stockholders;
we may issue undesignated preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval (notwithstanding any requirements imposed by the SEC or any exchange on which our common stock may now orpay in the future trade), and which may include rights superior to the rightsfor shares of the holdersCompany’s common stock, thereby depressing the market price of its common stock;
ourstock. In addition, because the Board is expressly authorizedresponsible for appointing the members of its management team, these provisions may frustrate or prevent any attempts by the Company’s stockholders to amend, restate,replace or repeal our Bylaws; and
advance notice is required with respectremove its current management by making it more difficult for stockholders to any nominations for election to our Board or for proposing matters that can be acted upon by stockholders at any meetingreplace members of stockholder s.the Board. Among other things, these provisions:
| ● | allow the authorized number of the Company’s directors to be changed only by resolution of the Board; |
| ● | limit the manner in which stockholders can remove directors from the Board; |
| ● | establish advance notice requirements for stockholder proposals that can be acted on at stockholder meetings and nominations to the Board; |
| ● | limit who may call stockholder meetings and the Company stockholders’ ability to act by written consent; |
| ● | authorize the Board to issue preferred stock without stockholder approval, which could be used to institute a “poison pill” that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by the Board; and |
| ● | require the approval of the holders of at least 2/3 of the votes that all the Company’s stockholders would be entitled to cast to amend or repeal specified provisions of the Company’s certificate of incorporation, as amended, or for stockholders to amend or repeal the Company’s amended and restated bylaws. |
In addition our Bylaws provideMoreover, because the Company is incorporated in Delaware, it is governed by the provisions of Section 203 of the DGCL, which generally prohibits a person who, together with their affiliates and associates, owns 15% or more of a company’s outstanding voting stock from, among other things, merging or combining with the company for a period of three years after the date of the transaction in which the person acquired ownership of 15% or more of the company’s outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
The Company’s certificate of incorporation designates the state courts in the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by its stockholders, which could discourage lawsuits against the company and its directors, officers and employees.
The Company’s certificate of incorporation provides that, unless we consentthe Company consents in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware is(or, if the Court of Chancery of the State of Delaware does not have jurisdiction, the federal district court for the District of Delaware) will be the sole and exclusive forum for certain actions, includingproceedings, including: (1) any derivative actionsaction or proceeding brought on the Company'sCompany’s behalf, stockholder actions claiming breaches(2) any action asserting a claim of breach of a fiduciary duty owed by any of our directors or officers, and claims arising under our organizational documents, in each case, subject to said Court of Chancery having personal jurisdiction over the indispensable parties named as defendants therein. Although this provision would not apply to any stockholder claims under the Exchange Act, there is uncertainty regarding whether a court would enforce such a forum selection provision as written would apply to stockholder claims under the Securities Act. Nevertheless, this forum selection provision may limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or ourCompany’s directors, officers, employees or agents,stockholders to the company or its stockholders, (3) any action asserting a claim arising pursuant to any provision of the DGCL or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware or (4) any action asserting a claim arising pursuant to any provision of the Company’s certificate of incorporation or amended and restated bylaws (in each case, as they may discourage lawsuits against us and such persons.
The limitations on certain stockholder rights imposedbe amended from time to time) or governed by thesethe internal affairs doctrine. These choice of forum provisions could also depresswill not apply to suits brought to enforce a duty or liability created by the trading price of our common stock.Securities Act, the Exchange Act or any other claim for which federal courts have exclusive jurisdiction.
These exclusive-forum provisions may make it more expensive for Company stockholders to bring a claim than if the stockholders were permitted to select another jurisdiction, and may limit the ability of the Company’s stockholders to bring a claim in a judicial forum that such stockholders find favorable for disputes with the Company or its directors, officers or employees, which may discourage such lawsuits against the Company and its directors, officers and employees. Alternatively, if a court were to find the choice of forum provisions contained in the Company’s certificate of incorporation to be inapplicable or unenforceable in an action, the Company may incur additional costs associated with resolving such action in other jurisdictions, which could materially adversely affect its business, financial condition and operating results.
The Company does not anticipate that it will pay any cash dividends in the foreseeable future.
The Company’s current expectation is that it will retain future earnings, if any, to fund the development and growth of the Company’s business. As a result, capital appreciation, if any, will be your sole source of potential gain on an investment in the Company’s common stock for the foreseeable future.
General Risks Related to the Company’s Business and Operations
The Company’s future success depends in large part on the Company’s ability to retain its key employees, as well as its ability to attract, train and motivate additional qualified personnel. The Company may also encounter difficulties in managing its growth, which could disrupt its operations.
The Company has a small number of employees, and it is highly dependent on the principal members of its management team, including its President and Chief Executive Officer, John Alam, M.D. Although the Company has employment agreements or offer letters with its executive officers and certain key employees, these agreements do not prevent them from terminating their services at any time.
Competition in the biotechnology industry for skilled and experienced employees is intense, particularly in the greater Boston, Massachusetts area, where the Company’s headquarters is located, and the Philadelphia, Pennsylvania area, where approximately 50% of the employee’s workforce is located. The Company also faces competition for the hiring of scientific and clinical personnel from universities and research institutions, many of which are near the Company’s headquarters. The loss of the services of any member of the Company’s senior management, clinical development or scientific staff, or any other key employee, may significantly delay or prevent the achievement of drug development and other business objectives and could have a material adverse effect on the Company’s business, operating results and financial condition.
The Company also relies on consultants and advisors to assist it in formulating and executing its business strategy. Many of the Company’s consultants and advisors are either self-employed or employed by other organizations, and they may have conflicts of interest or other commitments, such as consulting or advisory contracts with other organizations, which may affect their ability to contribute to the Company.
As the Company continues to develop neflamapimod for the treatment of DLB, and also to expand into clinical trials for other CNS disorders, the Company expects to experience significant growth in the number of employees and the scope of its operations. This strategy will require it to recruit additional clinical development, regulatory, scientific, and technical personnel, as well as sales and marketing personnel if neflamapimod is approved. If the Company is unable to attract, retain and motivate a sufficient number of highly qualified personnel to match such growth, its ability to further develop and commercialize neflamapimod, or any future product candidates the Company may develop or acquire, will be limited.
The Company may also be required to implement and improve managerial, operational and financial systems to manage its potential growth. Due to its limited financial and personnel resources, the Company may not be able to effectively manage the expansion of its operations or recruit and train a sufficient number of additional qualified personnel. The expansion of the Company’s operations may lead to significant costs and may divert its management and business development resources. Any inability to manage growth could delay the execution of the Company’s business plans or disrupt its operations.
The Company has identified material weaknesses in its internal control over financial reporting which, if not corrected, could affect the reliability of the Company’s financial statements and have other adverse consequences. The Company may identify additional material weaknesses in its internal controls over financial reporting which it may not be able to remedy in a timely manner. If the Company fails to maintain proper and effective internal controls, its ability to produce accurate financial statements on a timely basis could be impaired.
The Company is subject to the reporting requirements of the Exchange Act, the Sarbanes-Oxley Act, and the rules and regulations of Nasdaq. The Sarbanes-Oxley Act requires, among other things, that the Company maintain effective disclosure controls and procedures and internal control over financial reporting. The Company must perform system and process evaluation and testing of its internal control over financial reporting to allow management to report on the effectiveness of its internal controls over financial reporting in its Annual Report on Form 10-K filing for that year, as required by Section 404 of the Sarbanes-Oxley Act. This requires that the Company incur substantial professional fees and internal costs to expand its accounting and finance functions and that it expends significant management efforts. The Company may experience difficulty in meeting these reporting requirements in a timely manner.
A material weakness is a deficiency or combination of deficiencies in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of the Company’s consolidated financial statements would not be prevented or detected on a timely basis. The identified material weaknesses, if not corrected, could result in a material misstatement to the Company’s consolidated financial statements that may not be prevented or detected. The Company may discover weaknesses in its system of internal financial and accounting controls and procedures that could result in a material misstatement of its financial statements.
For example, in connection with the audit of the Company’s financial statements for the years ended December 31, 2023 and 2022, material weaknesses in the Company’s internal control over financial reporting were identified related to (i) the Company’s recording of significant complex transactions, and (ii) the absence of effective controls regarding the accurate identification, evaluation and proper recording of various expense accounts. The Company may identify additional material weaknesses in its internal controls over financing reporting in the future which it may not be able to remedy in a timely manner. Any material weaknesses will not be considered remediated until a remediation plan has been fully implemented, the applicable controls operate for a sufficient period of time, and it has been concluded, through testing, that the newly implemented and enhanced controls are operating effectively.
If the Company is not able to comply with the requirements of Section 404 of the Sarbanes-Oxley Act, or if it is unable to maintain proper and effective internal controls, the Company may not be able to produce timely and accurate financial statements. If that were to happen, the market price of its common stock could decline and it could be subject to sanctions or investigations by Nasdaq, the SEC, or other regulatory authorities. More generally, any failure by the Company to implement and maintain effective internal control over financial reporting could result in errors in the Company’s financial statements that could result in a restatement of the Company’s financial statements and could cause the Company to fail to meet its reporting obligations, any of which could diminish investor confidence in the Company and cause a decline in the price of the Company’s common stock.
The Company’s disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
The Company is subject to the periodic reporting requirements of the Exchange Act and its disclosure controls and procedures are designed to reasonably assure that information required to be disclosed by the Company in reports it files or submits under the Exchange Act is accumulated and communicated to management, recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. The Company believes that any disclosure controls and procedures or internal controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.
These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements or insufficient disclosures due to error or fraud may occur and not be detected.
The Company’s information technology systems, or those of its vendors, collaborators or other contractors or consultants, may fail or suffer security incidents, loss of data and other disruptions, which could result in a material disruption of its product development programs, compromise sensitive information related to its business or prevent it from accessing critical information, potentially exposing it to liability or otherwise adversely affecting its business.
In the ordinary course of the Company’s business, the Company collects and stores sensitive data, intellectual property, and proprietary business information. This data encompasses a wide variety of business-critical information including research and development information, clinical trial information, commercial information, and business and financial information. The Company faces risks relative to protecting this critical information including loss of access, unauthorized disclosure, unauthorized modification, and inadequate monitoring of its controls over these risks.
Despite the implementation of security measures, the Company’s internal IT systems and those of its current and any future third-party vendors, collaborators and other contractors or consultants are vulnerable to system failures, accidents, security incidents, damage, interruption or data theft from computer viruses, computer hackers, malicious code, employee theft or misuse, ransomware, social engineering (including phishing attacks), denial-of-service attacks, sophisticated nation-state and nation-state-supported actors, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. As use of digital technologies has increased, cyber incidents, including deliberate attacks and attempts to gain unauthorized access to computer systems and networks, have increased in frequency and sophistication. These threats pose a risk to the security of the Company’s IT systems and networks and the confidentiality, availability and integrity of the Company’s data. There can be no assurance that the Company will be successful in preventing cybersecurity incidents or successfully mitigating their effects.
Any such disruption or security incident could cause interruptions to its operations and result in disruption of the Company’s development programs and business operations. For example, the loss of clinical trial data from future clinical trials could result in delays in the Company’s regulatory approval efforts and significantly increase its costs to recover or reproduce the data. If the Company were to experience a significant cybersecurity incident that impacts its information systems or data, the costs associated with the investigation, remediation, and potential notification of the cybersecurity incident to counterparties, regulatory authorities, and data subjects could be material. In addition, the Company’s remediation efforts may not be successful. Cybersecurity incidents could also lead to significant business disruption, including transaction errors, supply chain or manufacturing interruptions, processing inefficiencies, data loss or the loss of or damage to intellectual property or other proprietary information. In addition, the Company’s recently-increased remote workforce could increase the Company’s cybersecurity risk, create data accessibility concerns, and make the Company more susceptible to communication disruption.
To the extent that any disruption or cybersecurity incident were to result in a loss of, or damage to, the Company’s or its third-party vendors’, collaborators’ or other contractors’ or consultants’ data or applications, or inappropriate disclosure of confidential or proprietary information, the Company could incur liability including litigation exposure, penalties and fines, the Company could become the subject of regulatory actions or investigations, its competitive position could be harmed and the further development and commercialization of its product candidates could be delayed. Any of the above could have a material adverse effect on the Company’s business, financial condition, reputation, competitive advantage, results of operations or prospects. While the Company maintains cyber-liability insurance, such insurance may not be adequate to cover any losses experienced as a result of a cybersecurity incident.
The Company’s business may be affected from time-to-time by government investigations and litigation with third parties, including its ongoing matter with Paul Feller.
The Company may from time to time receive inquiries and subpoenas and other types of information requests from government authorities and other third parties and may become subject to claims and other actions related to its business activities. While the ultimate outcome of investigations, inquiries, information requests and legal proceedings is difficult to predict, defense of litigation claims (even if ultimately successful) can be expensive, time-consuming and distracting, and adverse resolutions or settlements of those matters may result in, among other things, modifications to business practices, costs and significant payments, any of which could have a material adverse effect on the Company’s business, financial condition, results of operations and prospects.
For example, in August 2014, Paul Feller, the former Chief Executive Officer of the Company’s legal predecessor, filed a complaint asserting various causes of action related to his past affiliations with the Company’s legal predecessor. While the Company believes it has meritorious defense to the claims alleged in this matter and is defending itself vigorously, the Company is unable to predict the outcome and possible loss or range of loss, if any, associated with its resolution or any potential effect the matter may have on the Company’s financial position. Depending on the outcome or resolution of this matter, it could have a material effect on the Company’s consolidated financial position, results of operations and cash flows.
Now that the Merger has closed, there can be no further recourse by either party or its stockholders for a breach of representation or warranty by any of the parties to the Merger Agreement.
The representations and warranties of Diffusion, EIP and Merger Sub contained in the Merger Agreement or any certificate or instrument delivered pursuant to the Merger Agreement terminated at the Effective Time. To the extent that any such party’s breach of any representations and warranties is discovered or occurs in the future, there is no mechanism pursuant to which the other parties can pursue recourse or remedy.
The Company’s business is, or may in the future become, subject to complex and evolving U.S. and foreign laws and regulations relating to privacy and data protection. These laws and regulations are subject to change and uncertain interpretation, andthe Company’s actual or perceived failure to comply with such obligations could result in liability or reputational harm and could harm its business.
A wide variety of provincial, state, national, and international laws and regulations apply to the collection, use, retention, protection, disclosure, transfer and other processing of personal data. These data protection and privacy-related laws and regulations are evolving and may result in increased regulatory and public scrutiny and escalating levels of enforcement and sanctions. In the U.S., numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy laws and federal and state consumer protection laws govern the collection, use, disclosure and protection of health-related and other personal information. Failure to comply with data protection laws and regulations, where applicable, could result in government enforcement actions, which could include civil or criminal penalties, private litigation and/or adverse publicity and could negatively affect our operating results and business. For example, California has enacted the CCPA, which went into effect in January of 2020. The CCPA gives California residents expanded rights to access and require deletion of their personal information, opt out of certain personal information sharing, and receive detailed information about how their personal information is used. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches that may increase data breach litigation. Although the CCPA includes exemptions for certain clinical trials data, and HIPAA protected health information, the law may increase the Company’s compliance costs and potential liability with respect to other personal information the Company collects and processes about California residents. Additionally in 2020, California voters passed the CPRA, which went into full effect on January 1, 2023. The CPRA significantly amends the CCPA, potentially resulting in further uncertainty, additional costs related to our compliance efforts and additional potential for harm and liability if we fail to comply. Among other things, the CPRA established a new regulatory authority, the California Privacy Protection Agency, which is tasked with enacting new regulations under the CPRA and will have expanded enforcement authority. In addition to California, more U.S. states are enacting similar legislation, increasing compliance complexity and increasing risks of failures to comply. In 2023, comprehensive privacy laws in Virginia, Colorado, Connecticut, and Utah all took effect, and laws in Montana, Oregon, and Texas will take effect in 2024. In addition, laws in other U.S. states are set to take effect beyond 2024, and additional U.S. states have proposals under consideration, all of which could increase the Company’s regulatory compliance costs and risks, exposure to regulatory enforcement action and other liabilities.
Numerous other countries have, or are developing, laws governing the collection, use and transmission of personal information as well. For example, the European Parliament and the Council of the European Union adopted a comprehensive general data privacy framework called the GDPR which became fully effective in May 2018 and governs the collection and use of personal data in the European Union, including by companies outside of the European Union., The GDPR also imposes strict rules on the transfer of personal data out of the European Union to the United States. The GDPR imposes stringent data protection requirements and provides for penalties for noncompliance of up to the greater of €20 million or four percent of worldwide annual revenues. The GDPR and many other laws and regulations relating to privacy and data protection are still being tested in courts, and they are subject to new and differing interpretations by courts and regulatory officials. The Company may be required to devote significant additional resources to complying with these laws and regulations, and it is possible that the GDPR or other laws and regulations relating to privacy and data protection may be interpreted and applied in a manner that is inconsistent from jurisdiction to jurisdiction or inconsistent with the Company’s current policies and practices.
Applicable data privacy and data protection laws may conflict with each other, and by complying with the laws or regulations of one jurisdiction, the Company may find that it is violating the laws or regulations of another jurisdiction. Despite the Company’s efforts, the Company may not have fully complied in the past and may not in the future. That could require the Company to incur significant expenses, which could significantly affect its business. Failure to comply with data protection laws or to protect personal data or other data the Company processes or maintains may expose the Company to risk of enforcement actions taken by data protection authorities or other regulatory agencies, private rights of action in some jurisdictions, potential significant fines, penalties and other liabilities if it is found to be non-compliant, and damage to the Company’s reputation, any of which could materially affect its business, financial condition, results of operations and prospects. Furthermore, the number of government investigations related to data security incidents and privacy violations continue to increase and government investigations typically require significant resources and generate negative publicity, which could harm the Company’s business and reputation.
Past or future transactions resulting in an ownership change under Section 382 of the Code may subject the Company’s NOL carryforwards and certain other tax attributes to limitation.
As of December 31, 2023, the Company had U.S. federal NOL carryforwards of approximately $38.9 million. Under Sections 382 and 383 of the Code and corresponding provisions of state law, if a corporation undergoes an “ownership change” (within the meaning of Section 382), the corporation’s NOL carryforwards and certain other tax attributes (such as research tax credits) arising before the ownership change are subject to limitation on use after the ownership change. In general, an ownership change occurs if there is a cumulative change in the corporation’s equity ownership by certain stockholders that exceeds fifty percentage points (by value) over a rolling three-year period. Similar rules may apply under state tax laws. Past or future transactions to which the Company is a party may, alone or in the aggregate, result in such an ownership change and, accordingly, the Company’s NOL carryforwards and certain other tax attributes may be subject to limitations (or disallowance) on their use in the future. Consequently, even if the Company achieves profitability, it may not be able to utilize a material portion of its NOL carryforwards and other tax attributes, which could have a material adverse effect on cash flow and results of operations. There is also a risk that due to regulatory changes, such as suspensions on the use of NOLs or other unforeseen reasons, the Company’s existing NOLs could expire or otherwise be unavailable to offset future income tax liabilities.
The Company incurs costs and demands upon management as a result of complying with the laws, rules and regulations affecting public companies.
The Company incurs significant legal, accounting and other expenses associated with public company reporting requirements. The Company also incurs costs associated with corporate governance requirements, including requirements under the laws, rules and regulations of the SEC, as well as the rules and regulations of Nasdaq. These laws, rules and regulations also may make it difficult and expensive for the Company to obtain directors’ and officers’ liability insurance. As a result, it may be more difficult for the Company to attract and retain qualified individuals to serve on the Company’s Board or as executive officers of the Company, which may adversely affect investor confidence in the Company and could cause the Company’s business or stock price to suffer.
The Company may fail to comply with evolving privacy and data protection laws, which could adversely affect the Company’s business, results of operations and financial condition.
In California, the CCPA, which became effective in 2020, broadly defines personal information, gives California residents expanded individual privacy rights and protections and provides for civil penalties for violations and a private right of action for data breaches. Further, the CPRA, which became effective in 2023 and amends the CCPA, creates additional obligations with respect to processing and storing personal information. While there is limited exception for protected health information that is subject to HIPAA and clinical trial regulations, the CCPA may regulate or impact our processing of personal information depending on the context. Unlike other state privacy laws, the CCPA also regulates personal information collected in a business to business and in human resources contexts. Further, there continues to be some uncertainly about how provisions of the CCPA and the new regulations will be interpreted and how the law will be enforced. In addition to the CCPA, broad consumer privacy laws recently went into effect in Virginia on January 1, 2023, in Colorado and Connecticut on July 1, 2023, and in Utah on December 31, 2023. New privacy laws will also become effective in Florida, Montana and Texas in 2024, in Tennessee and Iowa in 2025, and in Indiana in 2026 and numerous other states are considering new privacy laws. Furthermore, other U.S. states, such as New York, Massachusetts, and Utah have enacted stringent data security laws and numerous other states have proposed similar privacy laws. The existence of differing comprehensive privacy laws in different states in the country will make the Company’s compliance obligations more complex and costly and may require us to modify the Company’s data processing practices and policies and to incur substantial costs and potential liability in an effort to comply with such legislation.
In the European Union and the United Kingdom, the Company may also face particular privacy, data security, and data protection risks in connection with requirements of the GDPR. The GDPR applies to any company established in the European Union as well as to those outside the European Union if they collect and use personal data in connection with the offering of goods or services to individuals in the European Union or the monitoring of their behavior. The GDPR imposes a broad range of data protection obligations on companies subject to the GDPR, including, for example, imposing obligations on companies around how they process personal data, stricter requirements relating to processing health and other sensitive data, ensuring there is a legal basis to justify the processing of personal data, stricter requirements relating to obtaining consent of individuals, expanded disclosures about how personal information is to be used, limitations on retention of information, mandatory data breach notification requirements, implementing safeguards to protect the security and confidentiality of personal data, taking certain measures on engagement with third parties, restrictions on transfers outside of the European Union to third countries deemed to lack adequate privacy protections, and has created onerous new obligations and liabilities on services providers or data processors. Non-compliance with the GDPR may result in monetary penalties of up to €20 million or 4% of worldwide revenue, whichever is higher. Moreover, data subjects can claim damages resulting from infringement of the GDPR. The GDPR further grants non-profit organizations the right to bring claims on behalf of data subjects. The GDPR and other changes in laws or regulations associated with the enhanced protection of certain types of personal data, such as healthcare data or other sensitive information, could greatly increase the Company’s cost of providing the Company’s products and services or even prevent us from offering certain services in jurisdictions that the Company may operate in. The GDPR may increase the Company’s responsibility and liability in relation to personal data that the Company processes where such processing is subject to the GDPR, and the Company may be required to put in place additional mechanisms to ensure compliance with the GDPR, including as implemented by individual countries. Ensuring the Company’s continued compliance with the GDPR is a rigorous and time-intensive process that may increase the Company’s cost of doing business or require us to change the Company’s business practices, and despite those efforts, there is a risk that the Company may be subject to fines and penalties, litigation, and reputational harm in connection with the Company’s European activities. Many jurisdictions outside of U.S. and Europe are also considering and/or enacting comprehensive data protection legislation that could have an impact on market expansion and clinical trials as well.
On July 10, 2023, the European Commission adopted an adequacy decision for a new mechanism for transferring data from the European Union to the United States – the EU-U.S. Data Privacy Framework, which provides European Union individuals with several new rights, including the right to obtain access to their data, or obtain correction or deletion of incorrect or unlawfully handled data. The adequacy decision followed the signing of an executive order introducing new binding safeguards to address the perceived deficiencies in the protection of EU-U.S. data transfers raised in the Maximilian Schrems vs. Facebook (Case C-311/18) decision by the Court of Justice of the European Union. The European Commission will continually review developments in the United States along with its adequacy decision. Adequacy decisions can be adapted or even withdrawn in the event of developments affecting the level of protection in the applicable jurisdiction. Future actions of EU data protection authorities are difficult to predict. Some customers or other service providers may respond to these evolving laws and regulations by asking us to make certain privacy or data-related contractual commitments that we are unable or unwilling to make. This could lead to the loss of current or prospective customers or other business relationships.
Because the interpretation and application of many privacy and data protection laws (including those state laws in the U.S. and the GDPR), commercial frameworks, and standards are uncertain, it is possible that these laws, frameworks, and standards may be interpreted and applied in a manner that is inconsistent with the Company’s existing data management practices and policies. If so, in addition to the possibility of fines, lawsuits, breach of contract claims, and other claims and penalties, the Company could be required to fundamentally change the Company’s business activities and practices or modify the Company’s solutions, which could have an adverse effect on the Company’s business. Any inability to adequately address privacy and security concerns, even if unfounded, or comply with applicable privacy and security or data security laws, regulations, and policies, could result in additional cost and liability to us, damage the Company’s reputation, inhibit the Company’s ability to conduct trials, and adversely affect the Company’s business.
The Company’s business and operations could suffer in the event of system failures, cyberattacks, or deficiency in the Company’s cyber security.
The Company relies on information technology systems and networks, including third-party "cloud-based" service providers, and the Company’s third-party CROs, to process, transmit and store electronic information in connection with the Company’s business activities. This includes crucial systems such as email, other communication tools, electronic document repositories, and archives. As use of digital technologies has increased, cyber incidents, including deliberate attacks and attempts to gain unauthorized access to computer systems and networks, have increased in frequency and sophistication. These threats pose a risk to the security of the Company’s systems and networks and the confidentiality, availability and integrity of the Company’s data. Cyberattacks could include wrongful conduct by hostile foreign governments, industrial espionage, wire fraud and other forms of cyber fraud, the deployment of harmful malware, denial-of-service, social engineering fraud or other means to threaten data security, confidentiality, integrity and availability. Furthermore, because the techniques used to obtain unauthorized access to, or to sabotage, systems change frequently and often are not recognized until launched against a target, the Company may be unable to anticipate these techniques or implement adequate preventative measures. The Company may also experience security breaches that may remain undetected for an extended period. A successful cyberattack could cause serious negative consequences for us, including, without limitation, the disruption of operations, the misappropriation of confidential business information, including financial information, trade secrets, financial loss and the disclosure of corporate strategic plans. As of the date of this Annual Report, there have been no cybersecurity incidents that have materially affected or are reasonably likely to materially affect the Company’s business strategy, results of operations, or financial condition. However, there can be no assurance that the Company will be successful in preventing cyber-attacks or successfully mitigating their effects.
The Company’s business activities may be subject to the FCPA and similar anti-bribery and anti-corruption laws.
The Company’s business activities may be subject to the FCPA and similar anti-bribery or anti-corruption laws, regulations or rules of other countries in which the Company operates, including the U.K. Bribery Act. The FCPA generally prohibits offering, promising, giving, or authorizing others to give anything of value, either directly or indirectly, to a non-U.S. government official in order to influence official action, or otherwise obtain or retain business. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect the transactions of the corporation and to devise and maintain an adequate system of internal accounting controls. The Company’s business is heavily regulated and therefore involves significant interaction with public officials, including officials of non-U.S. governments. Additionally, in many other countries, the health care providers who prescribe pharmaceuticals are employed by their government, and the purchasers of pharmaceuticals are government entities; therefore, any Company dealings with these prescribers and purchasers are subject to regulation under the FCPA. The SEC and U.S. Department of Justice have recently increased their FCPA enforcement activities with respect to biotechnology and pharmaceutical companies. There is no certainty that all the Company’s employees, agents, contractors, or collaborators, or those of the Company’s affiliates, will comply with all applicable laws and regulations, particularly given the high level of complexity of these laws. Violations of these laws and regulations could result in fines, criminal sanctions against the Company, its officers, or its employees, the closing down of facilities, requirements to obtain export licenses, cessation of business activities in sanctioned countries, implementation of compliance programs, and prohibitions on the conduct of its business. Any such violations could include prohibitions on the Company’s ability to offer its products in one or more countries and could materially damage the Company’s reputation, its brand, future international expansion efforts, its ability to attract and retain employees, and its business, prospects, operating results, and financial condition.
The Company’s employees, independent contractors, consultants, vendors and future commercial partners, if any, may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
The Company is exposed to the risk of fraud, misconduct or other illegal activity by its employees, independent contractors, consultants, vendors and other third parties. Misconduct by these parties could include intentional, reckless and negligent conduct that may fail to, among other things: comply with the rules and regulations of the FDA, EMA and other comparable foreign regulatory authorities; provide true, complete and accurate information to such authorities; comply with manufacturing standards the Company has established; comply with healthcare fraud and abuse laws; or report financial information or data accurately or to disclose unauthorized activities to the Company. If the Company obtains FDA approval of any of its product candidates and begins commercializing those products, its potential exposure under such laws will increase significantly, and its costs associated with compliance with such laws are also likely to increase. In particular, research, sales, marketing, education and other business arrangements in the healthcare industry are subject to extensive legal and regulatory requirements designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, educating, marketing and promotion, sales and commission, certain customer incentive programs and other business arrangements generally. Activities subject to these laws also involve the improper use of information obtained in the course of subject recruitment for clinical trials, which could result in regulatory sanctions and cause serious harm to the Company’s reputation. The Company has adopted a code of business conduct and ethics, but it is not always possible to identify and deter misconduct by employees and third parties, and the precautions the Company takes to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting it from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws. If any such actions are instituted against the Company, and the Company is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business, including the imposition of significant fines or other sanctions.
Inadequate funding for the FDA, the SEC and other government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner, or otherwise prevent those agencies from performing normal business functions on which the operation of the Company’s business may rely, which could negatively impact the Company’s business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, its ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of the FDA, the NIA, the SEC and other government agencies on which the Company’s operations may rely, including those that fund research and development activities, is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for clinical trial applications and/or marketing applications for new drugs to be reviewed or approved, which would adversely affect the Company’s business. Over the last several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA and the SEC, have had to furlough critical staff and stop critical activities. If a prolonged government or slowdown shutdown occurs, it could significantly impact the ability of the NIA to disburse funds for the Company’s clinical trial and for the FDA to timely review and process the Company’s regulatory submissions, which could have a material adverse effect on the Company’s business.
For example, the Company received access to $7.3 million under the NIA Grant in February 2024, 90% of the full amount of current year funding provided for in the NIA Grant, due to current NIA policy as a result of the U.S. government currently being funded on the basis of a continuing resolution. The timing of the Company’s receipt of the remaining 10%, or $0.8 million, of current year funding is dependent upon and subject to U.S. congressional approval of a final appropriations bill.
Future government shutdowns or slowdowns could also result in delays in the Company’s interactions with the SEC and other government agencies, which could impact the Company’s ability to access the public markets and obtain necessary capital in order to properly capitalize and continue its operations.
U.S. federal income tax reform or other changes in applicable tax law could adversely affect the Company’s business and financial condition.
The rules dealing with U.S. federal, state, and local income taxation are constantly under review by persons involved in the legislative process and by the Internal Revenue Service, the U.S. Treasury Department and other governmental bodies. In recent years, many such changes have been made and may continue to occur in the future. For example, in March 2020, the CARES Act was signed into law, which included certain changes in tax law intended to stimulate the U.S. economy in response to the COVID-19 coronavirus outbreak, including temporary beneficial changes to the treatment of net operating losses, interest deductibility limitations and payroll tax matters. Additionally, in December 2017, the TCJA was signed into law, which significantly reformed the Code. The TCJA included significant changes to corporate and individual taxation, some of which could adversely impact an investment in the Company’s common stock. For example, under the TCJA, in general, NOLs generated in taxable years beginning after December 31, 2017 may offset no more than 80 percent of such year’s taxable income and there is no ability for such NOLs to be carried back to a prior taxable year. The CARES Act modified the TCJA with respect to the TCJA’s limitation on the deduction of NOLs and provided that NOLs arising in taxable years beginning after December 31, 2017 and before January 1, 2021 may be carried back to each of the five taxable years preceding the tax year of such loss, but NOLs arising in taxable years beginning after December 31, 2020 may not be carried back. In addition, the CARES Act eliminated the limitation on the deduction of NOLs to 80 percent of current year taxable income for taxable years beginning before January 1, 2021 (but reinstated the limitation for taxable years beginning after December 31, 2020). As a result of such limitations, the Company may be required to pay federal income tax in some future year notwithstanding that it had a net loss for all years in the aggregate.
More generally, recent and future changes in tax laws could have a material adverse effect on the Company’s business, cash flow, financial condition or results of operations.
The Company faces risks associated with increased geopolitical uncertainty.
Ongoing and potential military actions across the globe, including the ongoing conflicts in Ukraine and the Middle East, as well as the sanctions, bans and other measures taken by governments, organizations and companies against the involved countries and certain citizens of those countries in response thereto, has increased the global political uncertainty and has strained the relations between a significant number of governments, including the U.S. The duration and outcome of these conflicts, any retaliatory actions or escalation, and the impact on regional or global economies is unknown but could have a material adverse effect on the Company’s business, financial condition and results of its operations.
Unfavorable global economic conditions could adversely affect the Company’s business, financial condition or results of operations.
The Company’s results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. For example, in 2008, the global financial crisis caused extreme volatility and disruptions in the capital and credit markets and, more recently, the COVID-19 pandemic caused significant volatility and uncertainty in U.S. and international markets. A severe or prolonged economic downturn, or additional global financial crises, could result in a variety of risks to the Company’s business, including weakened demand for its product candidates, if approved, or its ability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy could also strain the Company’s suppliers, possibly resulting in supply disruption. Any of the foregoing could harm the Company’s business and it cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact its business.
Epidemics, pandemics or other public health crises, including COVID-19, could adversely affect the Company’s business.
The Company’s operations could be significantly adversely affected by the effects of a widespread outbreak of epidemics, pandemics or other health crises, including COVID-19. The Company cannot accurately predict the impact of epidemics and pandemics would have on our operations and the ability of third parties to meet their obligations under contracts or arrangements with the Company, including uncertainties relating to the ultimate geographic spread of epidemics and pandemics, the severity of the underlying diseases, the duration of outbreaks, and the length of travel and quarantine restrictions imposed by governments of affected countries. In addition, a significant outbreak of contagious diseases in the human population could result in a widespread health crisis that could adversely affect the economies and financial markets of many countries, resulting in an economic downturn that could further affect the Company’s operations and ability to finance the Company’s operations.
Political uncertainty may have an adverse impact on the Company’s operating performance and results of operations.
General political uncertainty may have an adverse impact on the Company’s operating performance and results of operations. In particular, the United States continues to experience significant political events that cast uncertainty on global financial and economic markets, especially in light of the upcoming presidential election. It is presently unclear exactly what actions a new administration in the United States would implement, and if implemented, how these actions may impact the pharmaceutical industry in the United States.
The Company holds its cash and cash equivalents that it uses to meet its working capital needs in deposit accounts that could be adversely affected if the financial institutions holding such funds fail.
The Company holds its cash and cash equivalents that it uses to meet working capital needs in deposit accounts at certain third party financial institutions. The balances held in these accounts may exceed the FDIC, standard deposit insurance limit or similar government guarantee schemes. If a financial institution in which the Company holds such funds fails or is subject to significant adverse conditions in the financial or credit markets, the Company could be subject to a risk of loss of all or a portion of such uninsured funds or be subject to a delay in accessing all or a portion of such uninsured funds. Any such loss or lack of access to these funds could adversely impact the Company’s short-term liquidity and ability to meet its obligations.
For example, on March 10, 2023, Silicon Valley Bank, and on March 12, 2023, Signature Bank, were closed by state regulators and the FDIC was appointed receiver for each bank. The FDIC created successor bridge banks and all deposits of Silicon Valley Bank and Signature Bank were transferred to the bridge banks under a systemic risk exception approved by the U.S. Department of the Treasury, the Federal Reserve and the FDIC. While the Company did not hold any of its funds in accounts with either of these institutions, if financial institutions in which the Company holds funds for working capital were to fail, the Company cannot provide any assurances that such governmental agencies would take action to protect its uninsured deposits in a similar manner.
The Company may also, from time to time, maintain investment accounts with other financial institutions in which it holds its investments and, if access to the funds the Company uses for working capital is impaired, the Company may not be able to sell investments or transfer funds from its investment accounts to new accounts on a timely basis sufficient, or without incurring a loss or penalty as a result of such sale, to meet its working capital needs.
Certain stockholders could attempt to influence changes within the Company which could adversely affect the Company’s operations, financial condition and the value of its common stock.
One or more of the Company’s stockholders may from time to time seek to acquire a significant or controlling stake in the Company, engage in proxy solicitations, advance stockholder proposals or otherwise attempt to effect changes to the Company’s Board or corporate governance policies. Campaigns by stockholders to effect changes at publicly traded companies are sometimes led by investors seeking to increase short-term stockholder value through actions such as financial restructuring, increased debt, special dividends, stock repurchases or sales of assets or the entire company. Responding to proxy contests and other actions by activist stockholders can be costly and time-consuming, could disrupt the Company’s operations and divert the attention of the Company Board and senior management, and could adversely affect the Company’s operations, financial condition, and the value of its common stock.
The Company may not be able to enter into a transaction with a suitable acquiror or licensee for its product candidate TSC or any transaction entered into may not be on terms that are favorable to the Company.
As previously announced, in connection with Diffusion’s strategic review process during 2022-23, Diffusion made the decision to voluntarily pause the development program for TSC, Diffusion's lead drug candidate prior to the Merger. Currently, the Company does not intend to pursue the development of TSC and believes the primary path available to derive value from its TSC-related assets would be to find a suitable acquiror or licensee. Although the Company’s management has contacted numerous parties to assess their potential interest in such a transaction, to date, the Company has been unable to identify an interested counterparty. Furthermore, even if the Company is able to identify such a counterparty, supporting diligence activities conducted by potential acquirors or licensees and negotiating the financial and other terms of an agreement or license are typically long and complex processes, and the results of such processes cannot be predicted. There can be no assurance that the Company will enter into any transaction as a result of these effort or that any transaction involving the Company’s TSC-related assets will be entered into or, if entered into, will be on terms that are favorable to the Company. Furthermore, the Company cannot predict the impact that such a transaction or, alternatively, a failure to monetize the TSC assets in any material way, might have on its stock price.
Artificial intelligence presents risks and challenges that can impact the Company’s business including by posing security risks to confidential information, proprietary information, and personal data.
Issues in the development and use of artificial intelligence, combined with an uncertain regulatory environment, may result in reputational harm, liability, or other adverse consequences to the Company’s business operations. The Company may adopt and integrate generative artificial intelligence tools into our systems for specific use cases reviewed by legal and information security. The Company’s vendors may incorporate generative artificial intelligence tools into their offerings, and the providers of these generative artificial intelligence tools may not meet existing or rapidly evolving regulatory or industry standards with respect to privacy and data protection and may inhibit the Company’s or its vendors’ ability to maintain an adequate level of service and experience. If the Company, its vendors, or its third-party partners experience an actual or perceived violation of applicable privacy or data protection laws or regulations, or a cybersecurity incident due to the use of generative artificial intelligence, the Company could be subject to regulatory fines, investigations, enforcement actions, penalties and other liabilities, claims for damages from affected individuals, and the Company may lose valuable intellectual property and confidential information and its reputation and the public perception of the effectiveness of its privacy or cybersecurity measures could be harmed. Any of these outcomes could damage the Company’s reputation, result in the loss of valuable property and information, and adversely impact its business.
ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
Cybersecurity Risk Management Program Overview
We recognize the critical role that properly managing cybersecurity risk plays in maintaining the trust and confidence of our stockholders, the patients in our clinical trials, our employees, our business partners and our other stakeholders. Accordingly, our cybersecurity program is designed to identify, assess, manage and mitigate material risks from cybersecurity threats through a variety measures, including risk assessments, implementation of security measures, and ongoing monitoring of systems and networks. In collaboration with our third-party information technology service providers, a cross-functional team comprised of representatives from our administrative, finance and legal functions actively monitor the current threat landscape in an effort to identify material risks arising from new and evolving cybersecurity threats. We also engage external experts, including information technology experts, other consultants, and auditors to evaluate our cybersecurity measures and risk management processes.
We also identify our cybersecurity threat risks by comparing our processes to industry standards and best practices as well as by engaging experts to manage our information systems. To provide for the availability of critical data and systems, maintain regulatory compliance, manage our material risks from cybersecurity threats, and protect against and respond to cybersecurity incidents, we undertake the following activities:
| ● | monitor emerging data protection laws and implement changes to our processes that are designed to comply with such laws; |
| ● | through our policies, practices and contracts (as applicable), require employees, as well as third parties that provide services on our behalf, to treat confidential information and data with care; |
| ● | employ technical safeguards that are designed to protect our information systems from cybersecurity threats, which are evaluated and improved through vulnerability assessments and other evaluations on a routine basis; |
| ● | provide training for our employees regarding cybersecurity threats as a means to equip them with effective tools to address cybersecurity threats, and to communicate our evolving information security policies, standards, processes and practices; |
| ● | leverage threat intelligence available to us and our third party IT service provider to help us identify, protect, detect, respond and recover when there is an actual or potential cybersecurity incident; and |
| ● | carry information security risk insurance that provides protection against the potential losses arising from a cybersecurity incident. |
Board Oversight of Cybersecurity Risk Management and Governance
Our Board is responsible for general oversight of our risk environment and associated management policies and practices and has delegated to its Audit Committee the responsibility for oversight of our certain major risk categories and exposures, including with respect to cybersecurity and management’s processes to monitor and control them. The Audit Committee meets regularly throughout the year and, on no less than a quarterly basis, receives and reviews a report from management, including the Company’s General Counsel, regarding the Company’s IT, cybersecurity, data security, and physical security risk, including any suspected material or immaterial cybersecurity incidents during the preceding quarter, if any, and discusses such matters with appropriate management and other personnel. In addition, on a semi-annual basis, the Audit Committee receives a report from the Company’s primary third-party information technology and cybersecurity regarding the Company’s IT environment, overall cybersecurity risk management program and strategy and education regarding emerging trends and threats.
Management's Role in Cybersecurity Risk Management and Governance
Our executive management team is responsible for assessing and managing material risks from cybersecurity threats and possess relevant experience and expertise in various disciplines that are key to effectively managing such risks. The experience and expertise of our executive management team is also supplemented by our external IT service providers that collectively have extensive, broad experience and expertise in these areas. Our executive management team reports information about such risks to the Audit Committee of the Board on at least a quarterly basis.
We depend on and engage various other third parties, including suppliers, vendors, and service providers, to support key elements of our business including our information technology infrastructure. Our processes address cybersecurity threat risks associated with our use of such third-party service providers. In addition, cybersecurity considerations affect the selection and oversight of our third-party service providers. We perform diligence on third parties that have access to our systems, data or facilities that house such systems or data, and continually monitor cybersecurity threat risks identified through such diligence. Our management is informed about and monitors the prevention, detection, mitigation, and remediation of cybersecurity incidents, including through the receipt of notifications from service providers and reliance on communications with risk management, legal, information technology, and/or compliance personnel.
In response to an identified cybersecurity incident, a group comprised of appropriate management personnel, our third-party information technology service provider and, depending on the scope and severity of the incident, additional third-party subject matters experts, will be assembled to develop and implement a response strategy to contain, control, and remediate the cybersecurity incident, including securing our affected systems and/or information, mitigating harmful effects of the incident, preventing further compromises, and communicating information to affected parties, regulatory agencies and law enforcement, as necessary. This group will also report any such cybersecurity incident to the Audit Committee of the Board.
Assessment of Cybersecurity Risk
The potential impact of risks from cybersecurity threats are assessed on an ongoing basis by both management and the Board, including how such risks could materially affect our business strategy, operational results, and financial condition.
As of the date of this Annual Report, we have not experienced a cybersecurity incident that results in a material effect on our business strategy, results of operations or financial condition, but we cannot provide assurance that we will not be materially affected in the future by such an incident or risks related thereto.
We describe whether and how risks from identified cybersecurity threats, including as a result of any previous cybersecurity incidents, have materially affected or are reasonably likely to materially affect us, including our business strategy, results of operations, or financial condition, under the heading Item 1A. Risk Factors – General Risks Related to the Company’s Business and Operations,” which disclosures are incorporated by reference herein.
Our principal executiveWe currently have a short-term lease for office is locatedspace in Boston, Massachusetts and, in addition, previously had a leased facilityshort-term agreement to utilize membership-based co-working space in Charlottesville, Virginia, where we leasewhich was terminated in the first quarter of 2024. Rent expense related to the Company’s short-term agreements was approximately 8,000 square feet of office space$34,000 and $45,000 for approximately $10,000 per month. The current term of our Charlottesville lease will expire in April 2022.the years ended December 31, 2023 and 2022, respectively.
We believe the space is adequate to meet our near-term needs.
The information in Note 6,10, Commitments and Contingencies — Legal Proceedings to our consolidated financial statements set forthincluded in, Part II — Item 8 — Financial Statements and Supplementary Data of this Annual Report is incorporated herein by reference.
In addition, from time to time, we are subject to various pending or threatened legal actions and proceedings, including those that arise in the ordinary course of its business, which may include employment matters, breach of contract disputes and stockholder litigation. Such actions and proceedings are subject to many uncertainties and to outcomes that are not predictable with assurance and that may not be known for extended periods of time. We record a liability in our consolidated financial statements for costs related to claims, including future legal costs, settlements and judgments, when we have assessed that a loss is probable and an amount can be reasonably estimated. If the reasonable estimate of a probable loss is a range, we record the most probable estimate of the loss or the minimum amount when no amount within the range is a better estimate than any other amount. We disclose a contingent liability even if the liability is not probable or the amount is not estimable, or both, if there is a reasonable possibility that a material loss may have been incurred. In the opinion of management, as of the date hereof, the amount of liability, if any, with respect to these matters, individually or in the aggregate, will not materially affect our consolidated results of operations, financial position or cash flows.
ITEM 4. | MINE SAFETY DISCLOSURES |
None.
PART II
ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock trades publicly on the Nasdaq Capital Market under the symbol “DFFN.“CRVO.”
Holders
As of March 15, 2021,26, 2024, there were 353115 record holders of our common stock. This does not include beneficial owners of our common stock whose stock is held in nominee or “street name”.
Dividends
To date, we have not declared or paid any cash dividends on our common stock and do not intend to do so in the near future.
Securities Authorized for Issuance Under Equity Compensation Plans
The information set forth in, Part III — Item 12 – Security(Security Ownership of Certain Beneficial Owners and Management and Related Stockholder MattersMatters) of this Annual Report is incorporated herein by reference to the extent required by Item 201(d) of Regulation S-K.
Recent Unregistered Sales of Equity Securities and Use of Proceeds
NoneOn March 28, 2024, we entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The aggregate upfront gross proceeds from the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and additional gross proceeds of up to approximately $99.4 million may be received if the Series A Warrants are exercised in full for cash.
The 2024 Private Placement is exempt from the registration requirements of the Securities Act pursuant to the exemption for transactions by an issuer not involving any public offering under Section 4(a)(2) of the Securities Act and in reliance on similar exemptions under applicable state laws, as well as in accordance with applicable Nasdaq rules. The purchasers in the 2024 Private Placement represented that they were institutional accredited investors within the meaning of rules promulgated under the Securities Act and were acquiring the securities for investment only and with no present intention of distributing any of such securities or any arrangement or understanding regarding the distribution thereof. The securities were offered without any general solicitation by us or our representatives. The securities sold and issued in the 2024 Private Placement will not be registered under the Securities Act or any state securities laws and may not be offered or sold in the U.S. absent registration with the SEC or an applicable exemption from the registration requirements.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
ITEM 6. | SELECTED FINANCIAL DATA[RESERVED] |
The information required by Item 6 of Form 10-K has been omitted from this Annual Report pursuant to the amendments to Regulation S-K adopted by the SEC on November 19, 2020.
ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
Introduction
This discussion and analysis contains information related to historical and prospective events intended to enable you to assess our business, financial condition and results of operations. The information contained in this discussion and analysis should be read in conjunction with our consolidated financial statements and the related notes contained elsewhere in this Annual Report, as well as the risks and uncertainties discussed in Part Iunder the headings, "Item 1A –— Item 1A. Risk FactorsFactors" and of this Annual Report.“Note Regarding Forward-Looking Statements.”
Diffusion Pharmaceuticals: Enhancing Oxygen, Fueling Life
Overview
We are an innovative biopharmaceuticala clinical-stage biotechnology company focused on developing novel therapies that enhance the body’s ability to deliver oxygen to the areas where it is needed most. Our lead product candidate, TSC, is being developed to enhance the diffusion of oxygen to tissues with low oxygen levels, also known as hypoxia, a serious complication of many of medicine’s most intractable and difficult-to-treat conditions.
In addition to TSC, our product candidate DFN-529, a novel, allosteric PI3K/Akt/mTOR pathway inhibitor, is in early-stage development.treatments for age-related neurologic disorders. We previously completed two Phase 1 clinical trials evaluating DFN-529 in age-related macular degeneration. DFN-529 was also previously in preclinical development in oncology, specifically GBM.
Highlights from 2020 and Early 2021
| ●
| Board & Management Additions and Changes - During the second half of 2020, our leadership team changed significantly. We appointed our new chief executive officer, Robert J. Cobuzzi, Jr., Ph.D., and our new general counsel, William Elder, in September 2020, followed by our new chief medical officer, Christopher Galloway, M.D., in October 2020. We also welcomed a new director, Jane Hollingsworth, to our Board in August 2020. Dr. Cobuzzi joined the Board in January 2020.
|
| ●
| Initiation and Completion of TSC COVID Trial - In September 2020, we announced the dosing of the first two patients in our TSC COVID Trial evaluating TSC in hospitalized COVID-19 patients at the NIID in Bucharest, Romania. On February 9, 2021, we completed dosing of the twenty-fourth and final patient in the TSC COVID Trial. No dose-limiting toxicities or serious adverse events were observed among any patients in the study, including those who received the highest dose of 1.5 mg/kg every 6 hours. Evaluation of secondary endpoint data is ongoing and we anticipate this data will be available early in the second quarter of 2021.
|
| ●
| February 2021 Equity Offering - During the first quarter of 2021, we completed the February 2021 Offering resulting in aggregate gross net proceeds to the Company of approximately $34.5 million, before deducting underwriting discounts and commissions and offering expenses payable by us. In addition, during the period from January 1, 2021 through March l5, 2021, certain holders of warrants to purchase common stock cash exercised such warrants resulting in aggregate net proceeds to the Company of approximately $2.2 million. As a result, combined with our cash and cash equivalents as of December 31, 2020 of $18.5 million, we expect that our existing cash and cash equivalents will enable us to fund our operating expenses and capital expenditures (including our planned clinical trials) through 2023.
|
Our Strategic Priorities for 2021
Since the founding of Diffusion LLC, we have beenare currently focused on the development of TSC forour lead drug candidate, neflamapimod, an investigational, orally administered, small molecule brain penetrant that inhibits p38α in the treatmentneurons (nerve cells) within the brains of hypoxiapeople with neurodegenerative diseases. Neflamapimod has the potential to treat and asimprove synaptic dysfunction, the reversible aspect of the underlying disease processes in DLB and certain other major neurological disorders, and is currently being evaluated in our ongoing RewinD-LB Trial, a platformPhase 2b study in patients with DLB funded by a $21.0 million grant from the NIA. We expect to enhance standard-of-carecomplete enrollment in the RewinD-LB Trial during the second quarter of 2024 and to report initial results from the placebo-controlled portion of the study during the fourth quarter of 2024.
Our novel approach focuses on reducing the impact of inflammation in the brain, or neuroinflammation, which we believe is a key factor in the manifestation of degenerative diseases of the brain, including DLB. Chronic activation of the enzyme p38α in the neurons (nerve cells) within the brains of people with neurodegenerative diseases is believed to impair how neurons communicate through synapses (the connections between neurons). This impairment, termed synaptic dysfunction, leads to deterioration of cognitive and motor abilities. Left untreated, synaptic dysfunction can result in neuronal loss that leads to devastating disabilities, significant reliance on a caretaker, long term care living, and, ultimately, death. However, before neuronal loss commences, disease progression in major neurodegenerative disorders, including DLB, initially involves a protracted period of functional loss, particularly with respect to the synapses. We believe that inhibiting p38α activity in the brain, by interfering with key pathogenic drivers of disease, has the potential to reverse the clinical progression observed in early-stage neurodegenerative diseases, and that it is possible to slow further progression by delaying permanent synaptic dysfunction and neuron death.
We believe we are a leader in the industry in developing a treatment for conditions complicated by hypoxia. PriorDLB, as we are the only company of which we are aware with an asset that has shown statistically significant improvements compared to 2020, these efforts generated a substantial amount of data related to TSC, including data related to chemistry, manufacturing, and controls, preclinical safety and/or efficacy dataplacebo in a wide arrayPhase 2a clinical trial (our AscenD-LB Trial) and has initiated a Phase 2b clinical evaluation (our ongoing RewinD-LB Trial), from which we expect initial results before the end of experimental models, single dose safety, tolerability, and pharmacokinetic data,2024. The clinical symptoms in DLB are most directly linked to synaptic dysfunction in cholinergic neurons (neurons producing the neurotransmitter acetylcholine) in a part of the brain named the basal forebrain. Based on available preclinical and clinical data, and other information related to TSC's potential as a supplement to standard-of-care treatment in GBM, peripheral artery disease, and stroke.
In March 2020, the COVID-19 pandemic acceleratedwe believe if neflamapimod is given in the U.S. resultingearly stages of certain degenerative diseases of the brain, it may reverse synaptic dysfunction and improve neuron health and function. In preclinical studies, neflamapimod has been shown to reverse the neurodegenerative process in numerous delays and other challenges for biopharmaceutical companies conductingthe BFC system. Following earlier clinical studies in indications not related to COVID-19, including our TSC Stroke Trial. In early April 2020,demonstrating blood-brain-barrier penetration, target (p38α) engagement, and identification of dose-response, we announced plans to initiate aobtained positive Phase 2a clinical research program to evaluate TSCdata in patients with COVID-19 dueDLB in our AscenD-LB Trial. Specifically, statistically significant improvement was observed in patients treated with neflamapimod compared to patients treated with placebo on measures of dementia severity (as measured by CDR-SB) and functional mobility (i.e., walking ability, as measured by the anticipated persistenceTUG test) in the primary (intention-to-treat) analysis that includes all patients randomized into the study that had at least one measurement of the COVID-19 pandemic coupledendpoint analyzed. In addition, in a secondary analysis, neflamapimod demonstrated statistically significant improvement compared to placebo in a battery of cognitive tests, particularly with our belief in the potential of TSCrespect to improve low tissue oxygen levels, a common symptom and complicating factor in the treatment of COVID-19. We believe this decision was both opportunistic and a natural extension of the previous development work conducted with TSC, and in September 2020 we commenced the TSC COVID Trial. tests that measured attention.
In November 2020,October 2023, the major clinical neurology journal, Neurology, published additional analyses of the AscenD-LB Trial data that further strengthened these conclusions regarding neflamapimod’s potential efficacy and identified the DLB patient population most responsive to neflamapimod treatment. In these analyses, the results were stratified by pre-treatment levels of plasma ptau181, which recent scientific literature has identified as a biomarker to differentiate DLB patients with AD-associated co-pathology – a form of mixed dementia which we announced that, following changessometimes refer to our management teamas “DLB+AD” – from DLB patients without AD-associated co-pathology – which we sometimes refer to as “pure DLB.” In pure DLB patients, who generally represent early-stage patients with limited neurodegeneration in the hippocampus, the treatment response to neflamapimod in the AscenD-LB Trial was substantial (Cohen’s d effect size ≥ 0.7 and statistically significant vs. placebo on the CDR-SB, TUG, cognitive tests of attention and working memory) and greater than the overall patient population. In a February 2024 publication in the Journal of Prevention of Alzheimer’s Disease, results from our prior two months, we had initiated a thorough reviewclinical trials of our then-existing development program for TSCneflamapimod in AD and commenced plansDLB were integrated to modifyshow not only the program withdemonstrated effects of neflamapimod on cognition and function, but on other biomarkers such as EEG and brain volume and functional connectivity in the intent of accomplishing two principal strategic objectives:basal forebrain.
• ToOur ongoing RewinD-LB Trial is a double-blind, placebo-controlled, 16-week Phase 2b study in 160 patients with pure DLB funded by a $21.0 million grant from the NIA. The trial is intended to confirm the efficacy findings from the AscenD-LB Trial and definitively demonstrate proof-of-concept. We have utilized our subsequent analyses of the AscenD-LB data and the other information described above to optimize the clinical doseRewinD-LB Trial’s design and bolster the trial’s statistical power. Critically, the RewinD-LB Trial will exclude patients with Alzheimer’s disease related co-pathology as evaluated by plasma ptau181 levels (i.e. the study will only enroll patients with pure DLB) and, to enrich for such patients, the global CDR-SB score at entry will be limited to 0.5 or 1.0. Together with additional modifications to the Phase 2a design related to dosing frequency for TSC.regimen and primary endpoint, sample size calculations indicate that the RewinD-LB Phase Trial has greater than 95% statistical power (approaching 100%) to meet its primary objective of demonstrating improvement relative to placebo on change in CDR-SB over the course of the study.
• To evaluate TSC in clinical models designed to establish proof of concept for improvement in oxygenation.
The completion of the TSC COVID Trial in February 2021 provided the first multiple daily dose safety and tolerability data for TSC. We expect corresponding pharmacokinetic and other secondary endpoint data from this trial to be available earlycomplete enrollment in the RewinD-LB Trial during the second quarter of 2021. Obtaining these data represents2024 and to report initial results from the first, major step in our ongoing efforts to implement our modified development strategy, intended to strategically gap-fill, supplement, and expand on our past development work in TSC, with the ultimate goal of maximizing the probability of regulatory approval and, if TSC is approved, commercial success.
During the next twelve months, we intend to continue to execute our strategy and accomplish the following principal, strategic objectives, all of which we expect to be able to fund with cash-on-hand:
• Complete the TSC Oxygenation Trials – As described in more detail under the heading — Our Anticipated Next Steps in the TSC Development Program: The TSC Oxygenation Trials, the next phase in our development plan is to evaluate TSC's effects in clinical models of oxygenation in an effort to establish a dose-response relationship. We expect to commence the TSC TCOM Trial before the end of March 2021 with data expected in the second quarter of 2021, and we intend to commence both the TSC DLCO Trial and TSC Induced Hypoxia Trial in the third quarter of 2021 with data expected from each study expected within two months of their respective completion.
• Initiate Phase 2 Trial of TSC in Hypoxia-related Indication – We believe positive data from one or moreplacebo-controlled portion of the three TSC Oxygenation Trials would provide definitive evidence of TSC's enhancement of oxygenation, whether through increased uptake in the lungs, enhanced delivery, increased utilization at the tissue level, or some combination thereof. Positive data from any of these studies, if obtained, will guide the subsequent steps of our development strategy focused on demonstrating the clinical and therapeutic benefits of TSC in relevant patient populations on the hypoxia continuum. In particular, if successful in one or more of the three TSC Oxygenation Trials, we intend to initiate a Phase 2, controlled, clinical outcome study evaluating TSC in one or more appropriate hypoxia-related indications, which we refer to as our Planned Phase 2 Hypoxia-related Indication Trial, by the first quarter of 2022.
• Continue to Improve Our Efficiency and Effectiveness – This will include evaluating, refining, and improving our policies, procedures, and processes related to chemistry, manufacturing, and controls, quality assurance, information technology, intellectual property, human capital, and other areas, including as described in more detail under the headings, — Products, Product Development, and Our Competition and — Our People and Human Capital Resources in Part I – Item 1. Business of this Annual Report.
Our Anticipated Next Steps in the TSC Development Program: The TSC Oxygenation Trials
Following completion of the TSC COVID Trial in February 2021, the next step we have planned in the development of TSC is the design and execution of a trilogy of clinical studies using short-term, experimental models to evaluate the clinical effects of TSC on oxygenation. As of March 15, 2021, TSC has been administered to more than 180 subjects in our clinical trials. Data from these clinical trials have contributed significantly to our understanding of the safety, tolerability, and pharmacokinetics of TSC. In addition, post hoc analyses of two of our prior studies involving patients with peripheral artery disease with claudication and unresected GBM tumors have provided preliminary signals suggesting TSC's effect on oxygenation. However, neither of these studies was statistically powered to formally evaluate efficacy, and we therefore believe that further, robust clinical development of TSC requires a prospective exploration of the relationship between the level of exposure (dose) and response (change in oxygenation).
To this end, we plan to execute the three, short-term TSC Oxygenation Trials during 2021, each of which we expect to conduct in the U.S. and fund with cash-on-hand. We believe positive data from any or more of the three TSC Oxygenation Trials would provide evidence of a definitive effect of TSC on oxygenation, whether through increased uptake in the lungs, enhanced delivery, increased utilization at the tissue level, or some combination thereof. Positive data from any of these studies, if obtained, will also guide the subsequent steps of our development strategy focused on demonstrating the clinical and therapeutic benefits of TSC in relevant patient populations across the hypoxia continuum.
Assuming success in one or more of the three TSC Oxygenation Trials, we expect to identify and announce the specific, hypoxia-related indications we will target in advancing TSC's development in the fourth quarter of 2021 and intend2024. The results of the RewinD-LB Trial are intended to initiateprovide the data necessary to finalize our Planneddesign of a Phase 2 Hypoxia-related Indication Trial3 clinical trial, the general framework of which, including a 24-week treatment duration, has been agreed upon with the FDA.
In addition to neflamapimod’s potential to treat DLB, we believe the benefit of targeting neuroinflammation-induced synaptic dysfunction in the first quarterBFC system can be applied to other neurologic indications in which treatment of 2022.BFC dysfunction and degeneration would be expected to be clinically beneficial, including as treatment promoting recovery in the three months after ischemic stroke, as a disease-modifying treatment for early-stage Alzheimer’s disease, and as a treatment for certain forms of frontotemporal dementia.
Financial Summary
As of December 31, 2020,2023, we had a cash and cash equivalents balance of $18.5approximately $7.8 million. WeTo date, we have incurred operating losses since inception,not had any products approved for sale and have not generated any revenue from product sales revenue, and haveour ability to do so in the future will depend on the successful development and eventual commercialization of neflamapimod (or another product candidate that we could acquire or develop in the future). We do not achieved profitable operations. We incurred net losses of $14.2 million and $11.8 million for the years ended December 31, 2020 and 2019, respectively. To date, we have funded our operations and short-term liquidity needs primarily through the issuance and sale of common stock, warrants to purchase common stock, convertible debt, and convertible preferred stock. We expect to continue funding our operations through similar means for the foreseeable future, assuming the availability of additional capital, though we may enter into strategic partnerships or other alternative transactions in order to fund our ongoing capital requirements.generate revenue from product sales until such time, if ever.
Our accumulated deficit as of December 31, 2020,2023 was $105.9 million$54.4 million. We have never been profitable, and we expect towill continue to incur substantial losses in future periodsrequire additional capital to develop neflamapimod and fund operations for the foreseeable future. We also anticipatehave historically incurred net losses in each year since inception. Our net loss was $2.2 million and $5.8 million in the years ended December 31, 2023 and December 31, 2022, respectively. We expect our expenses will increase in connection with our ongoing activities, as we:
| ● | advance neflamapimod through clinical trials, including our ongoing Phase 2b trial for DLB, through to initiation of a Phase 3 trial in DLB; |
| ● | manufacture supplies for our nonclinical studies and clinical trials; |
| ● | obtain, maintain, expand, and protect our intellectual property portfolio; |
| ● | hire additional personnel to support our operations and growth; and |
| ● | continue to operate as a public company. |
Based on our current operating plan, we believe that our cash and cash equivalents on hand as of December 31, 2023, along with the remaining funds expected to be received from the NIA Grant, will not be sufficient to allow us to fund our current operations and continue as a going concern through at least one year from the date of the issuance of our consolidated financial statements. We expect to incur substantial expenditures for the foreseeable future for the development of neflamapimod and will require additional financing to continue this development.
On March 28, 2024, we entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The aggregate upfront gross proceeds for the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and up to an additional approximately $99.4 million in gross proceeds if the Series A Warrants are fully exercised for cash. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The information contained in this Management’s Discussion and Analysis of Financial Condition and Results of Operations, including information regarding our liquidity, capital resources and cash runway, does not reflect the anticipated consummation of, or our anticipated receipt of proceeds from, the 2024 Private Placement. For additional information regarding the 2024 Private Placement, the terms thereof (including the conditions to closing), and our expected use of the net proceeds therefrom, refer to our Current Report on Form 8-K filed with the SEC on March 28, 2024.
Financial Operations Overview
Revenue
To date, we have not generated any revenue from product sales and we do not expect to do so in the near future. In January 2023, we were awarded our $21.0 million NIA Grant. Funding from the NIA Grant is recognized as grant revenue as the qualifying expenses related thereto are incurred. As of December 31, 2023, $7.1 million of grant funding was recognized as revenue, of which $6.2 million has been received and the remaining $0.9 million has been recorded as grant receivable. As the NIA Grant was initially awarded in January 2023, there was no grant revenue in the year ended December 31, 2022.
Research and Development Expenses
Research and development expenses account for a significant portion of our operating expenses and primarily consist of costs incurred for the discovery and development of our product candidates, including:
| ● | expenses incurred under agreements with CROs, preclinical testing organizations, consultants, and other third-party vendors, collaborators and service providers; |
| ● | costs related to production of clinical materials, including fees paid to CMOs; |
| ● | vendor expenses related to the execution of preclinical studies and clinical trials; |
| ● | personnel-related expenses, including salaries, benefits, and stock-based compensation for personnel engaged in research and development functions; |
| ● | costs related to the preparation of regulatory submissions; |
| ● | third-party license fees; and |
| ● | expenses for rent and other supplies. |
We recognize research and development expenses as incurred. Costs for certain development activities are recognized based on an evaluation of the progress to completion of specific tasks using information and data provided to us by our vendors, collaborators, and third-party service providers. Non-refundable advance payments made by us for future research and development activities are capitalized and expensed as the related goods are delivered and as services are performed.
Specific program expenses include expenses associated with the development of our lead product candidate, neflamapimod, which recently initiated a Phase 2b clinical trial for treatment of subjects with DLB. Personnel or other operating expenses incurred for our research and development programs primarily relate to salaries and benefits, stock-based compensation, and facility expenses.
At this time, we cannot reasonably estimate or know the nature, timing, and estimated costs of the efforts that will be necessary to complete the development of, and obtain regulatory approval for, neflamapimod, or for any other product candidates that we may develop or acquire. We expect our research and development expenses to increase substantially for the foreseeable future as we continue to advanceinvest in R&D activities related to developing neflamapimod such as conducting larger clinical trials, seeking regulatory approval and incurring expenses associated with hiring personnel to support other R&D efforts. The process of conducting the necessary clinical research to obtain regulatory approval is costly and time-consuming, and the successful development of TSC,product candidates, including any costs related to:neflamapimod, is highly uncertain.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel-related costs, including stock-based compensation for our ongoingpersonnel in executive, finance and planned clinical trials, including the ongoing and planned TSC Oxygenation Trials and our Planned Phase 2 Hypoxia-related Indication Trial;
any additional studies we may undertake, including other preclinical and clinical studies to support the filing of any NDA with the FDA;
other research, development, and manufacturing activities designed to develop and optimize formulation, manufacturing processes, dosage, dose forms,accounting, and other characteristics prioradministrative functions. General and administrative expenses also include legal fees relating to
the maintenance, expansion, and protection our global intellectual property portfolio;
the hiring of additional clinical, manufacturing, scientific, sales, or other personnel; and
investments in operational, financial, and management information systems.corporate matters, professional fees paid for accounting, auditing, consulting, and tax services, insurance costs, and facility costs.
We intendanticipate that our general and administrative expenses will increase in the future as we increase our headcount to usesupport our existingcontinued research and development activities and as we continue development activities pursuant to the NIA Grant. We also anticipate that we will incur increased expenses as a result of operating as a public company, including expenses related to compliance with the rules and regulations of the SEC and those of any national securities exchange on which our securities are traded, legal, auditing, additional insurance expenses, investor relations activities, and other administrative and professional services.
Other Income (Expense)
Other income (expense) consists of interest earned on our cash and cash equivalents and the change in fair value of the previously outstanding EIP Convertible Notes.
Results of Operations
Comparison of the Years Ended December 31, 2023 and 2022
The following table summarizes our results of operations:
| | | December 31, | | | | | | | | | |
| | | 2023 | | | 2022 | | | $ Change | | | % Change | |
Grant revenue | | $ | 7,144,872 | | | $ | - | | | $ | 7,144,872 | | | | 100 | % |
Operating expenses: | | | | | | | | | | | | | | | | |
Research and development | | | 8,438,499 | | | | 1,336,469 | | | | 7,102,030 | | | | 531 | % |
General and administrative | | | 6,519,268 | | | | 2,139,065 | | | | 4,380,203 | | | | 205 | % |
Loss from operations | | | (7,812,895 | ) | | | (3,475,534 | ) | | | (4,337,361 | ) | | | 125 | % |
Other income (expense): | | | | | | | | | | | | | | | | |
Other income (expense) | | | 5,421,592 | | | | (2,389,152 | ) | | | 7,810,744 | | | | -327 | % |
Interest income | | | 219,430 | | | | 62,226 | | | | 157,204 | | | | 253 | % |
Interest expense | | | - | | | | (587 | ) | | | 587 | | | | -100 | % |
Total other income (expense) | | | 5,641,022 | | | | (2,327,513 | ) | | | 7,968,535 | | | | -342 | % |
Net loss | | $ | (2,171,873 | ) | | $ | (5,803,047 | ) | | $ | 3,631,174 | | | | -63 | % |
Grant Revenue
Grant revenue was $7.1 million for working capitalthe year ended December 31, 2023 which was a result of services performed during the year ended December 31, 2023 related to the $21.0 million grant awarded to us by the NIA in January 2023 to support a Phase 2b study of neflamapimod in DLB. At December 31, 2023, we had a receivable of $0.9 million for expenses incurred but not yet refunded by the NIA. As the NIA Grant was initially awarded in January 2023, there was no grant revenue in 2022.
Research and Development Expenses
Research and development expenses were $8.4 million for the year ended December 31, 2023, compared to $1.3 million for the year ended December 31, 2022. The increase of $7.1 million was primarily due to our DLB Phase 2b trial beginning in the first quarter of 2023 resulting in an increase in outsourced CRO and related site expenses.
General and Administrative Expenses
General and administrative expenses were $6.5 million for the year ended December 31, 2023, compared to $2.1 million for the year ended December 31, 2022. The increase of $4.4 million was primarily due to the Merger and public company related costs. The drivers of the increase were outsourced accounting/audit fees of $1.3 million, insurance costs of $1.0 million (including a one-time cost related to the merger of $0.8 million), headcount costs of $0.8 million, and investor/public relations costs of $0.7 million.
Other Income (Expense)
Other income (expense) was $5.4 million for the year ended December 31, 2023, compared to $(2.4) million for the year ended December 31, 2022. The amount for the year ended December 31, 2022 was driven by an increase in the estimated fair value of the Convertible Notes while the increase in the year ended December 31, 2023 was driven by the stock price on the date of conversion as a result of the Merger.
Interest income
Interest income was $0.2 million and $0.1 million for the years ended December 31, 2023 and 2022, respectively. The increase was primarily due to higher interest earned as a result of an increased cash equivalents balance.
Liquidity and Capital Resources
Capital Requirements
From the date of our inception through December 31, 2023, our operations had primarily been financed through the issuance of common stock, convertible preferred stock and convertible debt financings. As of December 31, 2023, we had approximately $7.8 million of cash and cash equivalents. We have not generated positive cash flows from operations and as of December 31, 2023, we had an accumulated deficit of approximately $54.4 million. In January 2023, we were awarded a $21.0 million grant from the NIA to support the Phase 2b study of neflamapimod in DLB, which is expected to be received over a three-year period. As of December 31, 2023, total cash funding of $6.2 million had been received from the NIA Grant.
In addition, we are party to our 2022 Sales Agreement with BTIG LLC (“BTIG”). The 2022 Sales Agreement is an "at-the-market" sales agreement pursuant to which we may, from time to time and through BTIG as our agent, sell up to an aggregate of $20.0 million in shares of common stock by any permissible method deemed an “at the market offering” as defined in Rule 415(a)(4) under the Securities Act. As of the date of this Annual Report, however, we have not sold any shares pursuant to the 2022 Sales Agreement.
On March 28, 2024, we entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The aggregate upfront gross proceeds from the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and additional gross proceeds of up to approximately $99.4 million may be received if the Series A Warrants are exercised in full for cash.
Our primary uses of cash are to fund theour operations, which consist primarily of research and development expenditures related to our programs and, to a lesser extent, general and administrative expenditures. Cash used to fund operating expenses is impacted by the timing of TSC, includingwhen we pay these expenses, as reflected in the TSC TCOM Trial,change in our outstanding accounts payable and accrued expenses.
Our losses from operations, negative operating cash flows and accumulated deficit, as well as the TSC VO2 Trial,additional capital needed to fund operations within one year of the issuance date of our financial statements for the period ended December 31, 2023, raise substantial doubt about our ability to continue as a going concern.
Any product candidates we may develop may never achieve commercialization, and we anticipate that we will continue to incur losses for the TSC DLCO Trial.foreseeable future. We expect that our research and development expenses, general and administrative expenses, and capital expenditures will continue to increase. In addition, we expect to incur costs associated with operating as a public company. As a result, until such time, if ever, as we can generate substantial product revenue, we expect to finance our cash needs through a combination of equity offerings, debt financings or other capital sources, including potential collaborations, licenses and other similar arrangements. Our primary uses of capital are, and we expect will continue to be, costs related to clinical research, manufacturing and development services; compensation and related expenses; costs relating to the build-out of our headquarters, other offices and laboratories; license payments or milestone obligations that may arise; laboratory expenses and costs for related supplies; manufacturing costs; legal and other regulatory expenses and general overhead costs.
Based on our current operating plan, we believe that our existing cash and cash equivalents on hand as of December 31, 2020, combined2023, along with the $36.7 million of aggregate gross proceedsremaining funds expected to be received byfrom the Company in connection with the completion of the February 2021 Offering (before deducting underwriting discounts and commissions and offering expenses payable by us) and the exercise of certain warrants to purchase common stock during the first quarter of 2021 priorNIA Grant (but without giving effect to the date of this Annual Report, willexpected proceeds from the 2024 Private Placement), are not sufficient to enable us to fund our operating expenses and capital expenditure requirements including expected costs related tofor at least one year from the planned TSC Oxygenation Trials and our Planned Phase 2 Hypoxia-related Indication Trial through 2023.
Financial Operations Overview
Revenues
date of the issuance of this Annual Report. We have not yet generated any revenue from product sales.based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect. We do not expect to generate revenue from product sales for the foreseeable future.
Research and Development Expense
R&D expenses include, but are not limited to, third-party CRO arrangements and employee-related expenses, including salaries, benefits, stock-based compensation, and travel expense reimbursement. R&D activities are central to our business model. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical studies. As we advance our product candidates, we expect the amount of R&D costs will continue to increase for the foreseeable future. R&D costs are chargedrequire additional financing to expense as incurred.
Generaladvance our current product candidates through clinical development, to develop, acquire or in-license other potential product candidates and Administrative Expense
G&A expenses consist principally of salaries and related costs for executive and other personnel, including stock-based compensation, other employee benefit costs, expenses associated with investment bank and other financial advisory services, and travel expenses. Other G&A expenses include, facility-related costs, communication expenses and professional fees for legal, patent prosecution and maintenance, consulting, accounting, and other professional services.
Interest Income
Interest income consists of interest earned from our cash and cash equivalents.
Income Tax Benefit (Expense)
We recognize income tax benefit to utilize indefinite deferred tax liabilities as a source of income against indefinite lived portions of our deferred tax assets. Our NOLs and tax credit carryforwards are subject to review and possible adjustment by the Internal Revenue Service and state tax authorities. NOL and tax credit carryforwards may become subject to an annual limitation in the event of a greater than 50.0% cumulative change in the ownership interest of significant stockholders over a three year period, as defined under Sections 382 and 383 of the Internal Revenue Code as well as similar state provisions. These limitations may, in certain cases, limit the amount of income tax benefit that can be utilized annually to offset taxable income or tax liabilities in future periods. The amount of the annual limitation is determined based on the Company’s value immediately prior to the ownership change, and subsequent ownership changes may further affect the limitation in future years. In 2019, due to the significant changes to our stockholder base as a result of the equity financing we completed during that year, we performed an analysis under Section 382 of the Internal Revenue Code and, as a result, reduced the magnitude of our NOL carryforwards to account for the ownership changes. In addition, the cumulative benefit of our NOLs was remeasured, resulting in tax expense recognized during the year ended December 31, 2019. We have not yet performed an analysis to determine whether or not ownership changes that have occurred in year ended December 31, 2020 give rise to any further limitations.
Critical Accounting Policies
Certain of our critical accounting policies require estimates that involve the application of significant judgment by management in selecting the appropriate assumptions in determining the estimate. By their nature, these judgments are subject to an inherent degree of uncertainty. We develop these judgments based on our historical experience, terms of existing contracts, our observance of trends in the industry, and information available from other outside sources, as appropriate. Actual results may differ from these judgments under different assumptions or conditions. Different, reasonable estimates could have been used for the current period. Additionally, changes in accounting estimates are reasonably likely to occur from period to period. Both of these factors could have a material impact on the presentation of our financial condition, changes in financial condition, or results of operations. We believe the accounting policies described below are among the most critical to aid in fully understanding and evaluating our financial statements, as they require estimates which involve our most subjective or complex judgments.
Intangible Assets
Our sole intangible asset as of December 31, 2020 consisted of DFN-529, which was acquired in 2016 pursuant to our merger with RestorGenex Corporation and is accounted for as an IPR&D intangible asset. The fair value of the IPR&D asset was determined as of the acquisition date using the cost approach, often referred to as current replacement cost, which establishes a value based on the cost of reproducing or replacing the asset. The cost approach was chosen as we were not able to estimate an income stream attributable to the IPR&D asset, given the fact that the related products had only completed limited preclinical and clinical trials and the timeline to commercial viability, if the FDA approval process were to be successful, is uncertain, would take a number of years, and the costs would be significant. As the development efforts for DFN-529 continue, based on the facts and circumstances at the time of a future valuation for the purposes of assessing impairment, it is possible that the value for the IPR&D asset could be substantially reduced or eliminated, which could result in a maximum pre-tax charge tofund operations equal to the current carrying value of the asset, or $8.6 million as of December 31, 2020. We most recently tested the IPR&D intangible asset for impairment on October 1, 2020, which is our annual impairment testing date. In addition to our annual impairment testing, upon the occurrence of certain triggering events, we may determine that an interim IPR&D impairment analysis is warranted. There was no impairment to the IPR&D asset during the years ended December 31, 2020 and 2019.
Results of Operations for Year Ended December 31, 2020 Compared to Year Ended December 31, 2019
The following table summarizes our results of operations for the years ended December 31, 2020 and 2019:
| | | Year ended December 31, | | | | | |
| | | 2020 | | | 2019 | | | Change | |
Operating expenses: | | | | | | | | | | | | |
Research and development | | $ | 9,427,667 | | | $ | 6,619,597 | | | $ | 2,808,070 | |
General and administrative | | | 6,444,109 | | | | 4,834,284 | | | | 1,609,825 | |
Depreciation | | | 103,168 | | | | 97,915 | | | | 5,253 | |
Loss from operations | | | (15,974,944 | ) | | | (11,551,796 | ) | | | 4,423,148 | |
Interest income | | | 114,257 | | | | 85,302 | | | | 28,955 | |
Loss from operations before income taxes | | | (15,860,687 | ) | | | (11,466,494 | ) | | | (4,394,193 | ) |
Income tax benefit (expense) | | | 1,675,381 | | | | (332,885 | ) | | | 2,008,266 | |
Net loss | | $ | (14,185,306 | ) | | $ | (11,799,379 | ) | | $ | (2,385,927 | ) |
Research and development expenses were $9.4 million during the year ended December 31, 2020 compared to $6.6 million during the year ended December 31, 2019, an increase of 42.4%. A significant portion of this increase was attributable to the TSC COVID Trial, which was initiated in September 2020, and our related ramp-up, including a $1.1 million increase in manufacturing costs and a $2.2 million increase in clinical trial and other related R&D expenses. Other R&D costs, including share based compensation, also increased by $0.3 million. These increases were slightly offset by decreases of $0.3 million and $0.2 million related to the wind-down of our TSC GBM Trial and our TSC Stroke Trial, respectively and a decrease in salaries and wages expenses of $0.3 million.
General and administrative expenses were $6.4 million during the year ended December 31, 2020 compared to $4.8 million during the year ended December 31, 2019, an increase of 33.3%. The increase in G&A expense was primarily due to a $0.7 million increase in professional fees and a $0.9 million increase in salaries, wages, and stock-based compensation expense, including non-recurring expenses related to the retirement, resignation, and separation of our former Chief Executive Officer in the third quarter of 2020.
Interest income increased slightly for the year ended December 31, 2020 compared to the year ended December 31, 2019 on an absolute basis due to our having a larger cash and cash equivalents balance and therefore earning more interest during the year ended December 31, 2020 compared to the year ended December 31, 2019.
For the year ended December 31, 2020, we recognized an income tax benefit of $1.7 million to reflect the utilization of indefinite deferred tax liabilities as a source of income against indefinite lived portions of the our deferred tax assets. As a result of a deemed change in ownership under the provisions of Section 382 of the Tax Code, the cumulative benefit of our NOLs was remeasured during 2019 resulting in the reversal of an income tax benefit of $0.4 million recorded in 2018. Accordingly, net of a $0.1 million benefit related to losses incurred during the year, we recognized an income tax expense of $0.3 million for the year ended December 31, 2019.
Liquidity and Capital Resources
Working Capital
The following table summarizes our working capital as of December 31, 2020 and 2019:
| | | December 31, | |
| | | 2020 | | | 2019 | |
Cash and cash equivalents | | $ | 18,515,595 | | | $ | 14,177,349 | |
Prepaid expenses, deposits and other assets | | | 260,825 | | | | 472,464 | |
Total current liabilities | | | 2,435,783 | | | | 1,721,421 | |
Working capital | | $ | 16,340,637 | | | $ | 12,928,392 | |
We expect to continue to incur net losses for the foreseeable future. We intendwill continue to use our existing cash and cash equivalents for workingseek funds through equity offerings, debt financings or other capital and to fund the research and development of TSC and our other product candidates, included the TSC Oxygenation Studies and our Planned Phase 2 Hypoxia-related Indication Trial, through 2023.
Cash Flows
The following table sets forth our cash flows for the years ended December 31, 2020 and 2019:
| | | December 31, | |
Net cash (used in) provided by: | | 2020 | | | 2019 | |
Operating activities | | $ | (13,552,628 | ) | | $ | (9,858,375 | ) |
Financing activities | | | 17,890,875 | | | | 16,044,552 | |
Net increase in cash and cash equivalents | | $ | 4,338,247 | | | $ | 6,186,177 | |
Operating Activities
For the year ended December 31, 2020, net cash used in operating activities increased $3.7 million, or 37.5%, compared to the year ended December 31, 2019.
Net cash used in operating activities of $13.6 million during the year ended December 31, 2020 was primarily attributable to our net loss of $14.2 million and a $1.7 million change in deferred income taxes. This amount was offset by our net change in operating assets and liabilities of $1.5 million, and non-cash charges comprised of $0.7 million of stock-based compensation expense and depreciation expense of $0.1 million.
Net cash used in operating activities of $9.9 million during the year ended December 31, 2019 was primarily attributable to our net loss of $11.8 million and a $0.3 million change in deferred income taxes. This amount was offset by our net change in operating assets and liabilities of $1.0 million, and non-cash charges comprised of $0.5 million of stock-based compensation expense and depreciation expense of $0.1 million.
Investing Activities
Cash flows from investing activities represent our sources, and uses of cash from our investments, including purchases and sales of equipmentpotential collaborations, licenses and other assets. During each of the years ended December 31, 2020 and 2019, we had no such cash flows.
Financing Activities
For the year ended December 31, 2020, net cash provided by financing activities increased $1.8 million, or 11.5%, compared to the year ended December 31, 2019.
Net cash provided by financing activities of $17.9 million during the year ended December 31, 2020 was primarily attributable to the $10.8 million in proceeds (net of underwriting discounts and commissions payable by us) received in connection with the May 2020 Offering and $8.0 million in proceeds received in connection with the exercise of common stock warrants stock during the year. These cash inflows were offset in part by the payment of $1.0 million in additional financing costs.
Net cash provided by financing activities of $16.0 million during the during the year ended December 31, 2019 was primarily attributable to the $12.3 million in aggregate proceeds (net of underwriting discounts and commissions payable by us) received in connection with the May 2019 Offering, the November 2019 Offering, and the December 2019 Offering and $3.9 million in proceeds received in connection with the exercise of common stock warrants during the year. These cash inflows were offset in part by the payment of $0.2 million in additional financing costs.
Capital Requirements
We expect to continue to incur substantial expenses and generate significant operating losses as we continue to pursue our business strategy of developing TSC. Our operations have consumed substantial amounts of cash since inception and we expect to continue to spend substantial amounts of cash to advance the clinical development of TSC, DFN-529, and our other product candidates. As of the date of this Annual Report, most our cash resources for clinical development are dedicated to our ongoing and planned TSC Oxygenation Trials and our Planned Phase 2 Hypoxia-related Indication Trial. While we believe we have adequate cash resources to continue operations through 2023, we anticipate that we will need additional funding in order to complete development of TSC which, if available, could be obtained through additional capital raising transactions, entry into strategic partnerships or collaborations, or alternative financingsimilar arrangements.
As of December 31, 2020, we did not have any credit facilities in place under which we could borrow funds or any other sources of committed capital. In the future, However, we may seekbe unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. If we do raise additional capital through various sources. However, we can give no assurances that wepublic or private equity offerings, the ownership interest of our existing stockholders will be able to secure additional sourcesdiluted, and the terms of funds to supportthese securities may include liquidation or other preferences that adversely affect our operations, or if such funds are available to us, that such additional financing will be sufficient to meet our needs or be on terms acceptable to us. This risk may increase if economic and market conditions deteriorate.stockholders’ rights. If we are unable to obtain additional financing when needed, we may need to terminate, significantly modify, or delay the development of TSC or our product candidates, or we may need to obtain funds through collaborations or otherwise on terms that may require us to relinquish rights to our technologies or product candidates that we might otherwise seek to develop or commercialize independently. If we are unable to raise adequate additional capital as and when required in the future, we could be forced to cease development activities and terminate our operations, and you could experience a complete loss of your investment.
To the extent that we raise additional capital in the future through the sale of our common stock or securities convertible or exchangeable for common stock such as common stock warrants, convertible preferred stock, or convertiblea debt instruments, the interests of our current stockholdersfinancing, we may be diluted or otherwise impacted. In particular, specific rights grantedsubject to future holders of preferred stock or convertible debt securities may include voting rights, preferences as to dividends and liquidation, conversion and redemption rights, sinking fund provisions, and restrictions on our ability to merge with or sell our assets to a third party. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any failure to raise capital as and when needed could have a negative impact on our financial condition and on our ability to pursue our business plans and strategies. If we are unable to raise capital, we will need to delay, reduce or terminate planned activities to reduce costs, including our development or commercialization activities for neflamapimod. We might also be required to seek funds through arrangements with third parties that require us to relinquish certain of our rights to neflamapimod or otherwise agree to terms unfavorable to us.
Because of the numerous risks and uncertainties associated with research, development and commercialization of product candidates, we are unable to estimate the exact amount of our operating capital requirements. Our future capital requirements will depend on, and could increase significantly as a result of, many factors, including:
| ● | the enrollment, progress, timing, costs and results of the RewinD-LB Trial, as well as additional development plans for neflamapimod in other disease indications, such as Recovery after Anterior Circulation Ischemic Stroke and FTD; |
| ● | the outcome, timing and cost of meeting regulatory requirements established by the FDA and other comparable foreign regulatory authorities; |
| ● | our ability to reach certain milestone events set forth in our collaboration agreements and the timing of such achievements, triggering our obligation to make applicable payments; |
| ● | the hiring of additional clinical, scientific and commercial personnel to pursue our development plans, as well the increased costs of internal and external resources as to support our operations as a public reporting company; |
| ● | the cost and timing of securing manufacturing arrangements for clinical or commercial production; |
| ● | the cost of establishing, either internally or in collaboration with others, sales, marketing and distribution capabilities to commercialize neflamapimod, if approved; |
| ● | the cost of filing, prosecuting, enforcing, and defending our patent claims and other intellectual property rights, including defending against any patent infringement actions brought by third parties against us; |
| ● | the ability to receive additional non-dilutive funding, including grants from organizations and foundations; |
| ● | our ability to establish strategic collaborations, licensing or other arrangements with other parties on favorable terms, if at all; and |
| ● | the extent to which we may in-license or acquire other product candidates or technologies. |
A change in the outcome of any of these or other variables could significantly alter the costs and timing associated with the development of neflamapimod. Furthermore, our operating plans may change in the future, and we may need additional funds to meet operational needs and capital requirements associated with such operating plans.
Cash Flows
| | | December 31, | |
| | | 2023 | | | 2022 | |
| | | | | | | | | |
Net cash used in operating activities | | $ | (7,449,847 | ) | | $ | (2,572,759 | ) |
Net cash provided by financing activities | | | 11,149,114 | | | | - | |
Net increase (decrease) in cash and cash equivalents | | $ | 3,699,267 | | | $ | (2,572,759 | ) |
Operating Activities
For the year ended December 31, 2023, cash used in operating activities was $7.4 million. The net cash outflow from operations primarily resulted from net loss of $2.2 million which included a $5.4 million non-cash gain due to a change in fair value of convertible debt and changes in operating assets and liabilities of $0.3 million, offset by a non-cash charge of $0.4 million for stock-based compensation.
For the year ended December 31, 2022, cash used in operating activities was $2.6 million. The net cash outflow from operations primarily resulted from net loss of $5.8 million and change in fair value of convertible debt of $2.4 million, offset by a non-cash charge of $0.3 million for stock-based compensation, $0.1 million of capital in lieu of executive compensation and changes in operating assets and liabilities of $0.4 million.
Financing Activities
For the year ended December 31, 2023, net cash provided by financing activities was $11.1 million. The net cash provided by financing activities primarily resulted from the net assets assumed in connection with the reverse recapitalization and sale of common stock offset by the payment of offering costs.
Investing Activities
We did not have any cash provided by or used in investing activities for the years ended December 31, 2023 or 2022.
Contractual Obligations and Other Commitments
We enter into contracts in the normal course of business with third-party contract organizations for clinical trials, nonclinical studies and manufacturing, and other services for operating purposes. The amount and timing of contractual obligations may vary based on the timing of services. We can generally elect to discontinue the work under these agreements at any time. In the future, we could also enter into additional collaborative research, contract research, manufacturing and supplier agreements which may require upfront payments or long-term commitments of cash.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements, as defined by the rules and regulations of the SEC that have or are reasonably likely to have a material effect on our financial condition, changes in financial condition, revenue or expenses, results of operations, liquidity, capital expenditures or capital resources. As a result, we are not materially exposed to any financing, liquidity, market or credit risk that could arise if we had engaged in these arrangements.
Critical Accounting Polices and Estimates
Management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported revenue generated and expenses incurred during the reporting periods. Our estimates are based on our historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions, and any such differences may be material. We believe that the accounting policies discussed below are critical to understanding our historical and future performance, as these policies relate to the more significant areas involving management’s judgments and estimates. We believe the following are our more significant estimates and judgments used in the preparation of our financial statements.
Research and Development Costs
Research and development costs are expensed as incurred and consist primarily of new product development. Research and development costs include salaries and benefits, consultants’ fees, process development costs and stock-based compensation, as well as fees paid to third parties that conduct certain research and development activities on our behalf.
A substantial portion of our ongoing research and development activities are conducted by third-party service providers. We record accrued expenses for estimated preclinical study and clinical trial expenses. Estimates are based on the services performed pursuant to contracts with research institutions, contract research organizations in connection with clinical studies, investigative sites in connection with clinical studies, vendors in connection with preclinical development activities, and contract manufacturing organizations in connection with the production of materials for clinical trials. Further, we accrue expenses related to clinical trials based on the level of subject enrollment and activity according to the related agreement. We monitor subject enrollment levels and related activity to the extent reasonably possible and make judgments and estimates in determining the accrued balance in each reporting period. Payments for these activities are based on the terms of the individual arrangements, which may differ from the pattern of costs incurred, and are reflected in the financial statements as prepaid or accrued research and development.
If we underestimate or overestimate the level of services performed or the costs of these services, actual expenses could differ from estimates. To date, we have not experienced significant changes in our estimates of preclinical studies and clinical trial accruals.
Stock-based Compensation
Stock-based compensation for employee and non-employee awards is measured on the grant date based on the fair value of the award and recognized on a straight-line basis over the requisite service period. The fair value of stock options to purchase common stock are measured using the Black-Scholes option pricing model. We account for forfeitures as they occur. The fair value of stock options is determined by us using the methods and assumptions discussed below. Each of these inputs is subjective and generally requires significant judgment and estimation by management.
Expected Term. The expected term represents the period that stock-based awards are expected to be outstanding. We use the “simplified method” to estimate the expected term of stock option grants. Under this approach, the weighted-average expected life is presumed to be the average of the contractual term of ten years and the weighted-average vesting term of our stock options, taking into consideration multiple vesting tranches. We utilize this method due to lack of historical data and the plain-vanilla nature of our stock-based awards.
Expected Volatility. We have limited information on the volatility of common stock as the shares were not actively traded on any public markets until recently. As such, expected volatility is derived from the historical stock volatilities of comparable peer public companies within our industry. These companies are considered to be comparable to our business over a period equivalent to the expected term of the stock-based awards.
Risk-Free Interest Rate. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the date of grant for zero-coupon U.S. Treasury notes with maturities approximately equal to the stock options expected term.
Expected Dividend Rate. The expected dividend is zero as we have not paid, nor do we anticipate paying, any dividends on our stock options in the foreseeable future.
In periods prior to the Merger, the grant date fair value of EIP Common Stock was typically determined by EIP’s Board of Directors with the assistance of management and a third party valuation specialist.
For additional information regarding stock-based compensation in periods following the Merger, see Note 12 to the consolidated financial statements included elsewhere in this Annual Report.
Valuation of Convertible Notes
The fair value of the Convertible Notes as of December 31, 2022 were estimated as the combination of a zero-coupon bond and a call option. The combined values for each of the Convertible Notes as of December 31, 2022 were then weighted by the probability of completing a financing or reverse merger. This approach resulted in the classification of the Convertible Notes as of December 31, 2022 as Level 3 of the fair value hierarchy (see Note 9 to the consolidated financial statements included elsewhere in this Annual Report). The assumptions utilized to value the 2020 Notes and the 2021 Notes as of December 31, 2022 were an estimated term of 0.94 years, volatility of 80.0% and a market yield of 55.2%.
In connection with the closing of the Merger, all outstanding EIP Convertible Notes converted into shares of EIP Common Stock at the fixed conversion price of $1.47 per share of EIP Common Stock, which shares of EIP Common Stock were subsequently converted into the right to receive shares of our CervoMed common stock (or pre-funded warrants in lieu thereof) upon closing of the Merger.
Recently Issued Accounting Pronouncements
The information in Note 3, Basis of Presentation and Summary of Significant Accounting Policies to our consolidated financial statements set forth in, “Part II — Item 8 — Financial Statements” of this Annual Report is incorporated herein by reference.
ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURE ABOUT MARKET RISK |
As a "smaller“smaller reporting company"company” (as such term is defined in Rule 12b-2 of the Exchange Act), we are not required to provide the information described in Item 305 of Regulation S-K and, accordingly, the information required by Item 67A of Form 10-K has been omitted from this Annual Report.
ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
Description | Page |
| | |
Report of Independent Registered Public Accounting Firm (PCAOB ID: 49) | 47103
|
| | |
Consolidated Balance Sheets as of December 31, 20202023 and 20192022 | 48104
|
| | |
Consolidated Statements of Operations for the years ended December 31, 20202023 and 20192022 | 49105
|
| | |
Consolidated Statements of Changes in Convertible Preferred Stock and Stockholders’ Equity (Deficit) for the years ended December 31, 20202023 and 20192022 | 50106
|
| | |
Consolidated Statements of Cash Flows for the years ended December 31, 20202023 and 20192022 | 51107
|
| | |
Notes to the Consolidated Financial Statements for the years ended December 31, 2020 and 2019 | 52108
|
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors
Diffusion Pharmaceuticals of CervoMed Inc.:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Diffusion PharmaceuticalsCervoMed Inc. and its subsidiaries (the Company) as of December 31, 20202023 and 2019,2022, the related consolidated statements of operations, changes in convertible preferred stock and stockholders’ equity (deficit) and cash flows for the years then ended, and the related notes to the consolidated financial statements (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 20202023 and 2019,2022, and the results of its operations and its cash flows for each of the years then ended, in conformity with U.S.accounting principles generally accepted accounting principles.in the United States of America.
The Company’s Ability to Continue as a Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Company has suffered recurring losses from operations since inception and will be required to raise additional capital to fund operations. This raises substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters also are described in Note 2. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidatedthe Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidatedfinancial statements that was communicated or required to be communicated to the audit committee and that: (1) relatesrelate to accounts or disclosures that are material to the consolidatedfinancial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of a critical audit mattermatters does not alter in any way our opinion on the consolidatedfinancial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinionopinions on the critical audit matter or on the accounts or disclosures to which it relates.they relate.
Deemed dividendAccounting for Reverse Recapitalization
As discussed in Note 7 to4 of the consolidatedaccompanying financial statements, on August 16, 2023, EIP Pharma, Inc. (“EIP”) merged with Merger Sub, a wholly-owned subsidiary of Diffusion Pharmaceuticals Inc. (“Diffusion”), whereby EIP became the surviving entity and a wholly-owned subsidiary of Diffusion (“the Merger”). Immediately following the Merger, the Company entered intochanged its name to CervoMed Inc. (“the Company”). The Merger was accounted for as a warrant exercise agreement with an investor who held a previously outstanding warrant. The Company recognized a deemed dividend of $2.0 millionreverse recapitalization under the United States generally accepted accounting principles (“U.S. GAAP”), in which EIP was determined to reflectbe the fair valueaccounting acquirer based upon the terms of the consideration given as an inducement for the investor to exercise the warrants.Merger and other certain factors.
We identified the evaluation ofaccounting for the deemed dividendreverse recapitalization as a critical audit matter. A higher degree of auditor judgment was required to evaluate the applicationmatter because of the accounting guidance due tocomplexity in the unique naturedetermination of the proper treatment of the transaction in accordance with U.S. GAAP, including judgments made by management to arrive at the investor.proper conclusion. This required subjective auditor judgment and increased level of effort when performing audit procedures, including the involvement of professionals with specialized skills and knowledge.
The following are the
Our audit procedures we performedrelated to address this critical audit matter. We read and evaluated the Company’s accounting memorandum that documentedfor the business purpose,reverse recapitalization included the terms of the agreements underlying the transaction, and the factors the Company considered in determining the applicable accounting treatment. We compared the facts and circumstances in the Company’s accounting memorandum to the agreements underlying the transaction.following, among others:
| ● | Obtained the relevant agreements and compared the underlying terms and conditions to management’s analysis and supporting documentation. |
| ● | With assistance from professionals with specialized skills and knowledge, obtained and evaluated management’s analysis and conclusions regarding the accounting treatment of the transaction, including determination of the accounting acquirer and whether the acquiree was considered to be a business. |
| ● | Tested the conversion of shares in common stock of the Company, including the conversion of EIP convertible notes and preferred stock and the recalculation of shares issued to Diffusion shareholders. |
/s/ KPMGRSM US LLP
We have served as the Company’s auditor since 2015.2017.
McLean, Virginia
Boston, Massachusetts
March 16, 202129, 2024
DIFFUSION PHARMACEUTICALS INC.
CONSOLIDATED BALANCE SHEETS
CervoMed Inc. |
Consolidated Balance Sheets |
| | | December 31, | |
| | | 2020 | | | 2019 | |
Assets | | | | | | | | |
Current assets: | | | | | | | | |
Cash and cash equivalents | | $ | 18,515,595 | | | $ | 14,177,349 | |
Prepaid expenses, deposits and other current assets | | | 260,825 | | | | 472,464 | |
Total current assets | | | 18,776,420 | | | | 14,649,813 | |
Property and equipment, net | | | 149,198 | | | | 252,366 | |
Intangible asset | | | 8,639,000 | | | | 8,639,000 | |
Right of use asset | | | 149,162 | | | | 247,043 | |
Other assets | | | 15,771 | | | | 322,301 | |
Total assets | | $ | 27,729,551 | | | $ | 24,110,523 | |
Liabilities and Stockholders’ Equity | | | | | | | | |
Current liabilities: | | | | | | | | |
Accounts payable | | $ | 545,844 | | | $ | 1,251,412 | |
Accrued expenses and other current liabilities | | | 1,776,470 | | | | 358,532 | |
Current operating lease liability | | | 113,469 | | | | 111,477 | |
Total current liabilities | | | 2,435,783 | | | | 1,721,421 | |
Deferred income taxes | | | 443,893 | | | | 2,119,274 | |
Noncurrent operating lease liability | | | 35,693 | | | | 135,566 | |
Total liabilities | | | 2,915,369 | | | | 3,976,261 | |
Commitments and Contingencies (Note 6) | | | | | | | | |
Stockholders’ Equity: | | | | | | | | |
Common stock, $0.001 par value: 1,000,000,000 shares authorized; 64,015,441 and 33,480,365 shares issued and outstanding at December 31, 2020 and 2019, respectively | | | 64,016 | | | | 33,481 | |
Additional paid-in capital | | | 130,659,550 | | | | 111,824,859 | |
Accumulated deficit | | | (105,909,384 | ) | | | (91,724,078 | ) |
Total stockholders' equity | | | 24,814,182 | | | | 20,134,262 | |
Total liabilities and stockholders' equity | | $ | 27,729,551 | | | $ | 24,110,523 | |
| | | December 31, | |
| | | 2023 | | | 2022 | |
Assets | | | | | | | | |
Current assets: | | | | | | | | |
Cash and cash equivalents | | $ | 7,792,846 | | | $ | 4,093,579 | |
Prepaid expenses | | | 1,256,501 | | | | 64,127 | |
Grant receivable | | | 915,404 | | | | - | |
Total current assets | | | 9,964,751 | | | | 4,157,706 | |
Other assets | | | 7,770 | | | | - | |
Total assets | | $ | 9,972,521 | | | $ | 4,157,706 | |
Liabilities, Convertible Preferred Stock and Stockholders’ Equity (Deficit) | | | | | | | | |
Current liabilities: | | | | | | | | |
Accounts payable | | $ | 662,471 | | | $ | 97,302 | |
Accrued expenses and other current liabilities | | | 1,933,276 | | | | 644,252 | |
Convertible notes | | | - | | | | 12,414,000 | |
Total liabilities | | | 2,595,747 | | | | 13,155,554 | |
Commitments and Contingencies (Note 10) | | | | | | | | |
Convertible preferred stock: | | | | | | | | |
| Series A preferred stock $0.001 par value; 30,000,000 and 0 shares authorized at December 31, 2023 and 2022, respectively, 0 shares issued and outstanding at December 31, 2023 and December 31, 2022 | | | - | | | | - | |
Series A-1 preferred stock, $0.001 par value; 0 and 1,960,600 shares authorized at December 31, 2023 and 2022, respectively; 0 and 1,960,600 shares issued and outstanding at December 31, 2023 and December 31, 2022, respectively | | | - | | | | 246,849 | |
Series A-2 preferred stock, $0.001 par value; 0 and 335,711 shares authorized at December 31, 2023 and 2022, respectively; 0 and 335,711 shares issued and outstanding at December 31, 2023 and December 31, 2022, respectively | | | - | | | | 4,173,267 | |
Series B preferred stock, $0.001 par value; 0 and 1,034,890 shares authorized at December 31, 2023 and 2022, respectively; 0 and 1,034,890 shares issued and outstanding at December 31, 2023 and December 31, 2022, respectively | | | - | | | | 19,867,095 | |
Total convertible preferred stock | | | - | | | | 24,287,211 | |
Stockholders’ Equity (Deficit): | | | | | | | | |
Common stock, $0.001 par value: 1,000,000,000 and 4,163,600 shares authorized as of December 31, 2023 and 2022, respectively, 5,674,520 and 518,140 shares issued and outstanding at December 31, 2023 and December 31, 2022, respectively | | | 5,674 | | | | 518 | |
Additional paid-in capital | | | 61,811,889 | | | | 18,983,339 | |
Accumulated deficit | | | (54,440,789 | ) | | | (52,268,916 | ) |
Total stockholders' equity (deficit) | | $ | 7,376,774 | | | | (33,285,059 | ) |
Total liabilities, convertible preferred stock and stockholders' equity (deficit) | | $ | 9,972,521 | | | $ | 4,157,706 | |
See accompanying notes to the consolidated financial statements.statements
DIFFUSION PHARMACEUTICALS INC.CervoMed Inc.
CONSOLIDATED STATEMENTS OF OPERATIONSConsolidated Statements of Operations
| | | Year Ended December 31, | |
| | | 2020 | | | 2019 | |
Operating expenses: | | | | | | | | |
Research and development | | $ | 9,427,667 | | | $ | 6,619,597 | |
General and administrative | | | 6,444,109 | | | | 4,834,284 | |
Depreciation | | | 103,168 | | | | 97,915 | |
Loss from operations | | | (15,974,944 | ) | | | (11,551,796 | ) |
Other income: | | | | | | | | |
Interest income | | | 114,257 | | | | 85,302 | |
Loss from operations before income taxes | | | (15,860,687 | ) | | | (11,466,494 | ) |
Income tax benefit (expense) | | | 1,675,381 | | | | (332,885 | ) |
Net loss | | $ | (14,185,306 | ) | | $ | (11,799,379 | ) |
Deemed dividend arising from warrant exchange | | | (1,950,378 | ) | | | — | |
Net loss attributable to common stockholders | | $ | (16,135,684 | ) | | $ | (11,799,379 | ) |
| | | | | | | | | |
Share information: | | | | | | | | |
Net loss per share of common stock, basic and diluted | | $ | (0.30 | ) | | $ | (1.76 | ) |
Weighted average shares outstanding, basic and diluted | | | 53,831,973 | | | | 6,706,509 | |
| | | Years Ended December 31, | |
| | | 2023 | | | 2022 | |
Grant revenue | | $ | 7,144,872 | | | $ | - | |
Operating expenses: | | | | | | | | |
Research and development | | | 8,438,499 | | | | 1,336,469 | |
General and administrative | | | 6,519,268 | | | | 2,139,065 | |
Total operating expenses | | | 14,957,767 | | | | 3,475,534 | |
Loss from operations | | | (7,812,895 | ) | | | (3,475,534 | ) |
Other income (expense): | | | | | | | | |
Other income (expense) | | | 5,421,592 | | | | (2,389,152 | ) |
Interest income | | | 219,430 | | | | 62,226 | |
Interest expense | | | - | | | | (587 | ) |
Total other income (expense) | | | 5,641,022 | | | | (2,327,513 | ) |
Net loss | | $ | (2,171,873 | ) | | $ | (5,803,047 | ) |
Per share information: | | | | | | | | |
Net loss per share of common stock - basic and diluted | | $ | (0.82 | ) | | $ | (11.20 | ) |
Weighted average shares outstanding - basic and diluted | | | 2,661,416 | | | | 518,140 | |
See accompanying notes to the consolidated financial statements.statements
DIFFUSION PHARMACEUTICALS INC.
CervoMed Inc.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERSConsolidated Statements of Changes in Convertible Preferred Stock and Stockholders’ EQUITYEquity (Deficit)
| | | Stockholders' Equity | |
| | | Common Stock | | | | | | | | | | |
| | | | | | Additional
Paid-in | | | Accumulated | | | Total
Stockholders' | |
| | | Shares | | | Amount | | | Capital | | | Deficit | | | Equity | |
Balance at December 31, 2018 | | | 3,376,230 | | | $ | 3,377 | | | $ | 95,532,881 | | | $ | (79,924,699 | ) | | $ | 15,611,559 | |
Issuance of common stock, pre-funded warrants and warrants, net of issuance costs | | | 12,688,276 | | | | 12,688 | | | | 11,905,207 | | | | — | | | | 11,917,895 | |
Issuance of common stock upon exercise of warrants | | | 17,415,859 | | | | 17,416 | | | | 3,871,009 | | | | — | | | | 3,888,425 | |
Stock-based compensation expense | | | — | | | | — | | | | 515,762 | | | | — | | | | 515,762 | |
Net loss | | | — | | | | — | | | | — | | | | (11,799,379 | ) | | | (11,799,379 | ) |
Balance at December 31, 2019 | | | 33,480,365 | | | | 33,481 | | | | 111,824,859 | | | | (91,724,078 | ) | | | 20,134,262 | |
Issuance of common stock and warrants, net of issuance costs | | | 11,428,572 | | | | 11,429 | | | | 10,330,202 | | | | — | | | | 10,341,631 | |
Issuance of common stock upon exercise of warrants | | | 19,106,504 | | | | 19,106 | | | | 7,768,370 | | | | — | | | | 7,787,476 | |
Stock-based compensation expense | | | — | | | | — | | | | 736,119 | | | | — | | | | 736,119 | |
Net loss | | | — | | | | — | | | | — | | | | (14,185,306 | ) | | | (14,185,306 | ) |
Balance at December 31, 2020 | | | 64,015,441 | | | $ | 64,016 | | | $ | 130,659,550 | | | $ | (105,909,384 | ) | | $ | 24,814,182 | |
| | | Years Ended December 31, 2023 and 2022 | |
| | | Series A-1 Preferred Stock | | | Series A-2 Preferred Stock | | | Series B Preferred Stock | | | Common Stock | | | Additional | | | Accumulated | | | Total Stockholders' | |
| | | Shares | | | Amount | | | Shares | | | Amount | | | Shares | | | Amount | | | Shares | | | Amount | | | Paid in Capital | | | Deficit | | | Equity (Deficit) | |
Balance at January 1, 2022 | | | 1,960,600 | | | | 246,849 | | | | 335,711 | | | | 4,173,267 | | | | 1,034,890 | | | | 19,867,095 | | | | 518,140 | | | | 518 | | | | 18,521,988 | | | | (46,465,869 | ) | | | (27,943,363 | ) |
Stock-based compensation expense | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 333,835 | | | | - | | | | 333,835 | |
Contributed capital in lieu of executive compensation | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 127,516 | | | | - | | | | 127,516 | |
Net loss | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | (5,803,047 | ) | | | (5,803,047 | ) |
Balance at January 1, 2023 | | | 1,960,600 | | | $ | 246,849 | | | | 335,711 | | | $ | 4,173,267 | | | | 1,034,890 | | | $ | 19,867,095 | | | | 518,140 | | | $ | 518 | | | $ | 18,983,339 | | | $ | (52,268,916 | ) | | $ | (33,285,059 | ) |
Conversion of convertible preferred stock to common stock | | | (1,960,600 | ) | | | (246,849 | ) | | | (335,711 | ) | | | (4,173,267 | ) | | | (1,034,890 | ) | | | (19,867,095 | ) | | | 2,936,566 | | | | 2,937 | | | | 24,284,274 | | | | - | | | | 24,287,211 | |
Issuance of common stock upon settlement of Convertible Notes | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 795,905 | | | | 796 | | | | 6,988,953 | | | | - | | | | 6,989,749 | |
Issuance of common stock to Diffusion stockholders in reverse recapitalization, net of issuance costs | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 1,360,244 | | | | 1,360 | | | | 10,337,754 | | | | - | | | | 10,339,114 | |
Sale of common stock | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 63,422 | | | | 63 | | | | 809,937 | | | | - | | | | 810,000 | |
Stock-based compensation expense, including vesting of RSUs | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 77 | | | | - | | | | 407,632 | | | | - | | | | 407,632 | |
Issuance of common stock from exercises of stock options | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | 359 | | | | - | | | | - | | | | - | | | | - | |
Repurchase of common stock from net settled stock option | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | (193 | ) | | | - | | | | - | | | | - | | | | - | |
Net loss | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | - | | | | (2,171,873 | ) | | | (2,171,873 | ) |
Balance at December 31, 2023 | | | - | | | $ | - | | | | - | | | $ | - | | | | - | | | $ | - | | | | 5,674,520 | | | $ | 5,674 | | | $ | 61,811,889 | | | $ | (54,440,789 | ) | | $ | 7,376,774 | |
See accompanying notes to the consolidated financial statements.statements
DIFFUSION PHARMACEUTICALS INC.CervoMed Inc.
CONSOLIDATED STATEMENTS OF CASH FLOWSConsolidated Statements of Cash Flows
| | | Year Ended December 31, | |
| | | 2020 | | | 2019 | |
Operating activities: | | | | | | | | |
Net loss | | $ | (14,185,306 | ) | | $ | (11,799,379 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | | | |
Depreciation | | | 103,168 | | | | 97,915 | |
Stock-based compensation expense | | | 736,119 | | | | 515,762 | |
Change in deferred income taxes | | | (1,675,381 | ) | | | 332,885 | |
Changes in operating assets and liabilities: | | | | | | | | |
Prepaid expenses, deposits and other assets | | | 518,169 | | | | 426,774 | |
Accounts payable, accrued expenses and other liabilities | | | 950,602 | | | | 567,668 | |
Net cash used in operating activities | | | (13,552,629 | ) | | | (9,858,375 | ) |
| | | | | | | | | |
Cash flows provided by financing activities: | | | | | | | | |
Proceeds from the sale of common stock | | | 10,827,100 | | | | — | |
Proceeds from the sale of common stock, pre-funded warrants and warrants | | | — | | | | 12,318,956 | |
Proceeds from the exercise of common stock warrants and pre-funded warrants | | | 8,046,103 | | | | 3,888,425 | |
Payment of offering costs | | | (982,328 | ) | | | (162,829 | ) |
Net cash provided by financing activities | | | 17,890,875 | | | | 16,044,552 | |
| | | | | | | | | |
Net increase in cash and cash equivalents | | | 4,338,247 | | | | 6,186,177 | |
| | | | | | | | | |
Cash and cash equivalents at beginning of year | | | 14,177,349 | | | | 7,991,172 | |
Cash and cash equivalents at end of year | | $ | 18,515,595 | | | $ | 14,177,349 | |
| | | | | | | | | |
Supplemental disclosure of non-cash investing and financing activities: | | | | | | | | |
Offering costs in accounts payable and accrued expenses | | $ | — | | | $ | 238,232 | |
Operating lease right of use asset and liability | | $ | — | | | $ | 334,205 | |
| | | Years Ended December 31, | |
| | | 2023 | | | 2022 | |
Cash flows from operating activities: | | | | | | | | |
Net loss | | $ | (2,171,873 | ) | | $ | (5,803,047 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | | | |
Stock-based compensation expense | | | 407,632 | | | | 333,835 | |
Capital in lieu of executive compensation | | | - | | | | 127,516 | |
Change in fair value of convertible debt | | | (5,424,251 | ) | | | 2,389,000 | |
Changes in operating assets and liabilities: | | | | | | | | |
Prepaid expenses, deposits and other assets | | | (1,200,144 | ) | | | 85,104 | |
Grant receivable | | | (915,404 | ) | | | - | |
Accounts payable | | | 565,169 | | | | 79,154 | |
| Acccrued expenses and other liabilities | | | 1,289,024 | | | | 215,679 | |
Net cash used in operating activities | | | (7,449,847 | ) | | | (2,572,759 | ) |
Net assets assumed in connection with reverse recapitalization | | | 11,887,757 | | | | - | |
Proceeds from sale of common stock | | | 810,000 | | | | - | |
Payment of reverse recapitalization costs | | | (1,548,643 | ) | | | - | |
Net cash provided by financing activities | | | 11,149,114 | | | | - | |
| | | | | | | | | |
Net increase (decrease) in cash and cash equivalents | | | 3,699,267 | | | | (2,572,759 | ) |
Cash and cash equivalents at beginning of year | | | 4,093,579 | | | | 6,666,338 | |
| Cash and cash equivalents at end of year | | $ | 7,792,846 | | | $ | 4,093,579 | |
| | | | | | | | | |
Supplemental disclosure of non-cash financing activities: | | | | | | | | |
Conversion of Convertible Notes | | $ | 6,989,749 | | | $ | - | |
Conversion of convertible preferred stock | | $ | 24,287,211 | | | $ | - | |
See accompanying notes to the consolidated financial statements.statements
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTSCervoMed Inc.
Notes to the Consolidated Financial Statements
1. OrganizationThe Company and Description of Business
The Company is a corporation organized under the laws of the state of Delaware and headquartered in Boston, Massachusetts. The Company is a biotechnology company developing treatments for age-related neurologic disorders. The Company is currently developing its product candidate neflamapimod, an investigational orally administered small molecule brain penetrant that inhibits p38α. Neflamapimod has the potential to treat synaptic dysfunction, the reversible aspect of the underlying neurodegenerative processes that cause disease in DLB and certain other major neurological disorders and is currently being evaluated in a Phase 2b study in patients with DLB.
On March 30, 2023, Diffusion Pharmaceuticals Inc. (“Diffusion”), Dawn Merger Inc., a Delaware corporation, an innovative biopharmaceutical company developing novel therapies that enhancewholly-owned subsidiary of Diffusion (“Merger Sub”) and EIP Pharma, Inc. (“EIP”) entered into the body’s abilityMerger Agreement (Note 4), pursuant to deliver oxygenwhich, at the Effective Time, Merger Sub merged with and into EIP, with EIP surviving the Merger as a wholly-owned subsidiary of the Company. In connection with the Merger, on August 16, 2023, the Company changed its corporate name from “Diffusion Pharmaceuticals Inc.” to the areas where it is needed most. The Company’s lead product candidate, TSC, is being developed to enhance the diffusion of oxygen to tissues with low oxygen levels, also known as hypoxia, a serious complication of many of medicine’s most intractable and difficult-to-treat conditions.“CervoMed Inc.”
On August 16, 2023, Diffusion approved a one-for-1.5 reverse stock split which was consummated for historical Diffusion shares in connection with the Merger. In addition, upon consummation of the Merger, all historical EIP shares were adjusted using an exchange ratio of 0.1151. All information in the accompanying consolidated financial statements and notes thereto regarding share amounts of common stock, price per share of common stock and the conversion factor for preferred stock into common stock has been adjusted to TSC,reflect the Company's product candidate DFN-529,application of the reverse stock split and the exchange ratio on a novel, allosteric PI3K/Akt/mTOR pathway inhibitor, isretroactive basis.
All shares of EIP common stock outstanding immediately prior to the Effective Time, after giving effect to the conversion of EIP preferred stock and the Convertible Notes (and excluding shares held as treasury stock by EIP, shares held or owned by the Company and any dissenting shares), converted into the right to receive, in early-stage development. The Company previously completed two Phase 1 clinical trials evaluating DFN-529 in age-related macular degeneration. DFN-529 was also previously in preclinical development in oncology, specifically GBM.the aggregate, 4,314,033 shares of the Company’s common stock and prefunded warrants to purchase 495,995 shares of common stock, based on an exchange ratio of 0.1151.
2. Liquidity and Capital Resources
The Company has not generated any revenuesnegative cash flows from product salesoperations and, has funded operations primarilyas of December 31, 2023, had an accumulated deficit of approximately $54.4 million. In January 2023, the Company was awarded a $21.0 million grant from the NIA to support its ongoing RewinD-LB Trial, which is expected to be received over a three-year period. In July 2023, the Company sold common stock for proceeds of public$0.8 million. In addition, the Company received $12.7 million in cash and private offerings of equity, convertible debt and convertible preferred stock. Substantial additional financing will be required bycash equivalents through the reverse recapitalization. The Company expects to continue to fundgenerate operating losses for the foreseeable future. The Company’s future viability is dependent on its researchability to raise additional capital to finance its operations and development activities. No assurancepursue its business strategies. There can be givenno assurances that any such financingadditional funding will be available when needed,on terms acceptable to the Company, or at all, or thatall. These conditions cause substantial doubt regarding the Company’s research and development efforts will be successful.ability to continue as a going concern.
The Company regularly explores alternative means ofwill continue to require additional financing its operations and seeks fundingto advance current product candidates through various sources, including public and private securities offerings, collaborative arrangements with third parties and other strategic alliances and business transactions. The Company does not have any commitments to obtain additional funds and may be unable to obtain sufficient funding in the future on acceptable terms, if at all. If the Company cannot obtain the necessary funding, it will need to delay, scale back or eliminate some or all of its research andclinical development, programs or enter into collaborations with third parties to commercialize potential products or technologies that it might otherwise seek to develop, acquire or commercialize independently; considerin-license other various strategic alternatives, including a merger or sale of the Company; or cease operations. If the Company engages in collaborations, it may receive lower consideration upon commercialization of such products than if it had not entered such arrangements or if it entered into such arrangements at later stages in the product development process.
Operations of the Company are subject to certain risks and uncertainties including various internal and external factors that will affect whether and when the Company’spotential product candidates become approved drugs and how significant their market share will be, some of which are outside ofto fund operations for the Company’s control. The length of time and cost of developing and commercializing these product candidates and/or failure of them at any stage of the drug approval process will materially affect the Company’s financial condition and future operations.foreseeable future. The Company expects that its existing cash and cash equivalents as of December 31, 2020, combined2023, along with the proceedsremaining funds expected to be received byfrom the Company in connection with the completion of the February 2021 Offering and the exercise of certain warrantsNIA Grant, will not be sufficient to purchase common stock during the first quarter of 2021 prior to the date of this Annual Report, will enable it to fund its operating expenses and capital expenditure requirements, and continue as a going concern for at least twelve months following the issuance of these consolidated financial statements. The Company will continue to seek funds through equity offerings, debt financings or other capital sources, including expected costspotential collaborations, grants, licenses and other similar arrangements. However, the Company may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Without additional funding, the Company would be forced to delay, reduce or eliminate its research and development programs. Accordingly, since the financing discussed below has not been completed, substantial doubt exists about the Company’s ability to continue as a going concern within one year after the date these financial statements are issued. The accompanying consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
On March 28, 2024, the Company entered into a securities purchase agreement with certain purchasers named therein related to the planned TSC Oxygenation Trialsprivate placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The aggregate upfront gross proceeds for the Planned Phase 2 Hypoxia-related Indication Trial through 2023.2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and up to an additional approximately $99.4 million in gross proceeds if the Series A Warrants are fully exercised for cash. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The consolidated financial statements do not reflect and do not include any adjustments for the Company’s expected receipt of proceeds from the 2024 Private Placement.
3.Basis of Presentation and Summary of Significant Accounting Policies
Basis of Presentationpresentation
The accompanying consolidated financial statements of the Company have been prepared in accordanceconformity with GAAP. Any reference in these notes to applicable guidance is meant to refer toUS GAAP as found indefined by the Accounting Standards Codification and Accounting Standards UpdatesFASB.
Consolidation
The consolidated financial statements include the accounts of the Financial Accounting Standards Board.Company and its wholly-owned subsidiaries. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimatesestimates
The preparation of consolidated financial statements in conformity with US GAAP requires the Companymanagement to make estimates and assumptions that affect the reported amounts of assets, liabilities, grant revenue, expenses, and liabilitiesrelated disclosures. On an ongoing basis, the Company’s management evaluates its estimates, including estimates related to money market accounts, clinical trial accruals, stock-based compensation expense, grant revenue, Convertible Notes, and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period.
On an ongoing basis, the The Company evaluatesbases its estimates usingon historical experience and other factors, including the current economic environment. Significant items subject to such estimates aremarket-specific or relevant assumptions used for purposes of determining stock-based compensation and accounting for research and development activities. Managementthat it believes its estimates to be reasonable under the circumstances. Actual results couldmay differ significantly from those estimates.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Fair Value of Financial Instruments
The carrying amounts of the Company’s financial instruments, including cash equivalents and accounts payable approximate fair value due to the short-term nature of those instruments.estimates or assumptions.
Concentration of Credit Risk
Financial instruments that potentially exposesubject the Company to concentrations of credit risk consist principally ofare primarily cash on depositand cash equivalents. The Company maintains its cash and cash equivalent balances with multiple financial institutions the balancesthat management believes are creditworthy. The Company has no financial instruments with off-balance-sheet risk of which frequently exceed federally insured limits.loss. The Company has not experienced any losses in such accounts.
Cash and Cash Equivalents
The Company considers anyall highly liquid investments such as money market funds, with an original maturitymaturities of three months90 days or less at the date of purchase to be cash and cash equivalents.
Property and Equipment
The Company records property and equipment Cash equivalents, which consist of amounts invested in money market funds, are stated at cost less accumulated depreciation and amortization. Costs of renewals and improvements that extend the useful lives of the assetsfair value. There are capitalized. Maintenance and repairs are expensed as incurred. Depreciation is recognized on a straight-line basis over the estimated useful lives of the assets, which generally range from 2 to 15 years. The Company amortizes leasehold improvements over the shorter of the estimated useful life of the asset or the term of the related lease. Upon retirement or disposition of assets, the costs and related accumulated depreciation and amortization are removed from the accounts with the resultingno unrealized gains or losses if any, reflected in results of operations.
Long-Lived Assets
Long-lived assets are reviewedon the money market funds for potential impairment whenever events indicate that the carrying amount of such assets may not be recoverable. The Company does this by comparing the carrying value of the long-lived assets with the estimated future undiscounted cash flows expected to result from the use of the assets, including cash flows from disposition. If it is determined an impairment exists, the asset is written down to its estimated fair value. The Company has not recognized any impairment of long-lived assets during the years ended December 31, 20202023 and 2019.2022.
Intangible AssetFair Value of Financial Instruments
The Company’s financial instruments consist primarily of cash and cash equivalents, accounts payable, previously outstanding Convertible Notes and accrued liabilities. The Company’s cash and cash equivalents, accounts payable and accrued liabilities approximate fair value due to their relatively short maturities. The Company determined the fair value of the Convertible Notes as described in Note 9. In connection with the consummation of the Merger (Note 4) on August 16, 2023, the Convertible Notes were converted into EIP Common Stock which was subsequently converted into the right to exchange such shares of EIP Common Stock for shares of the Company’s common stock.
The Company utilizes valuation techniques that maximize the use of observable inputs and minimize the use of unobservable inputs to the extent possible. The Company determines the fair value of its financial instruments based on assumptions that market participants would use in pricing an asset or liability in the principal or most advantageous market. When considering market participant assumptions in fair value measurements, the following fair value hierarchy distinguishes between observable and unobservable inputs, which are categorized in one of the following levels:
Level 1 – Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date;
Level 2 – Inputs are observable, unadjusted quoted prices in active markets for similar assets or liabilities, unadjusted quoted prices for identical or similar assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the related assets or liabilities; and
Level 3 – Unobservable inputs that are significant to the measurement of the fair value of the assets or liabilities that are supported by little or no market data.
The following table presents the Company’s assets that are measured at fair value on a recurring basis:
| | | December 31, 2023 | |
| | | (Level 1) | | | (Level 2) | | | (Level 3) | |
Assets | | | | | | | | | | | | |
Cash equivalents (money market accounts) | | $ | 7,792,846 | | | $ | - | | | $ | - | |
Total assets measured at fair value | | $ | 7,792,846 | | | $ | - | | | $ | - | |
| | | December 31, 2022 | |
| | | (Level 1) | | | (Level 2) | | | (Level 3) | |
Assets | | | | | | | | | | | | |
Cash equivalents (money market accounts) | | $ | 4,093,579 | | | $ | - | | | $ | - | |
Total assets measured at fair value | | $ | 4,093,579 | | | $ | - | | | $ | - | |
| | | | | | | | | | | | | |
Liabilities | | | | | | | | | | | | |
Convertible Notes | | $ | - | | | $ | - | | | $ | 12,414,000 | |
Total liabilities measured at fair value | | $ | - | | | $ | - | | | $ | 12,414,000 | |
The following table presents a roll-forward of the fair value of the Convertible Notes (Note 9) for which fair value is determined by Level 3 inputs:
| | | Year Ended | |
| | | December 31, 2023 | | | December 31, 2022 | |
Beginning balance | | $ | 12,414,000 | | | $ | 10,025,000 | |
Fair value adjustment | | | (5,424,251 | ) | | | 2,389,000 | |
Reclassification to additional paid in capital upon conversion | | | (6,989,749 | ) | | | - | |
Ending balance | | $ | - | | | $ | 12,414,000 | |
Valuation techniques used to measure fair value maximize the use of relevant observable inputs and minimize the use of unobservable inputs (Note 9). The Company’s Convertible Notes are classified within Level 3 of the fair value hierarchy because the fair value measurement is based, in part, on significant inputs not observed in the market.
There were no transfers among Level 1, Level 2 or Level 3 categories in the years ended December 31, 2023 or 2022.
The fair value of the 2020 Notes and the 2021 Notes, and collectively the Convertible Notes (Note 9) as of December 31, 2022 were estimated as the combination of a zero-coupon bond and a call option. The combined values for each of the 2020 Notes and the 2021 Notes as of December 31, 2022 were then weighted by the probability of completing a financing or reverse merger. This approach resulted in the classification of the 2020 Notes and the 2021 Notes as of December 31, 2022 as Level 3 of the fair value hierarchy. The assumptions utilized to value the 2020 Notes and the 2021 Notes as of December 31, 2022 were an estimated term of 0.94 years, volatility of 80.0% and a market yield of 55.2%. The measurement of fair value incorporates expected future cash flows associated with interest payments; as such, there is no separate accrual for interest accrued but not yet paid.
Leases
In February 2016, the FASB issued ASU No. 2016-02, “Leases”, which establishes a ROU model. That requires a lessee to recognize an ROU asset and corresponding lease liability on the balance sheet for all leases with a term longer than 12 months. Leases will be classified as finance or operating, with classification affecting the pattern and classification of expense recognition in the statement of operations as well as the reduction of the ROU asset. The new standard provides a number of optional practical expedients in transition. The Company has elected to apply (i) the practical expedient, which allows us to not separate lease and non-lease components, for new leases and (ii) the short-term lease exemption for all leases with an original term of less than 12 months, for purposes of applying the recognition and measurements requirements in the new standard.
At the inception of an arrangement, the Company determines whether the arrangement is or contains a lease based on specific facts and circumstances, the existence of an identified asset(s), if any, and the Company’s control over the use of the identified asset(s), if applicable. Operating lease liabilities and their corresponding ROU assets are recorded based on the present value of future lease payments over the expected lease term. The interest rate implicit in lease contracts is typically not readily determinable. As such, the Company will utilize the incremental borrowing rate, which is the rate incurred to borrow on a collateralized basis over a similar term an amount equal to the lease payments in a similar economic environment.
The Company has an indefinite-lived IPR&D asset, DFN-529, which haselected to combine lease and non-lease components as a single component. Operating leases will be recognized on the consolidated balance of $8.6 million at both December 31, 2020sheet as ROU assets, lease liabilities current and December 31, 2019. DFN-529 is a PI3K/Akt/mTOR pathway inhibitorlease liabilities non-current. Fixed rent payments are included in preclinical development for oncology.
Intangible assets deemed to have indefinite lives are not amortized but rather are assessed for impairment annually on October 1the calculation of the Company’s fiscal year or more frequently if impairment indicators exist. There was no impairment tolease balances, while variable costs paid for certain operating and pass-through costs are excluded. Lease expense is recognized over the Company’s DFN-529 intangible asset recognized during the years ended December 31, 2020 and 2019.expected term on a straight-line basis.
Research and Development
Major componentsResearch and development costs are expensed as incurred and consist primarily of researchnew product development. Research and development costs include internalsalaries and benefits, consultants’ fees, process development costs and stock-based compensation, as well as fees paid to third parties that conduct certain research and development (such as salaries and related employee benefits, equity-based compensation, supplies and allocated facility costs) and contracted services (researchactivities on the Company’s behalf.
A substantial portion of the Company’s ongoing research and development activities performedare conducted by third-party service providers. The Company records accrued expenses for estimated preclinical study and clinical trial expenses. Estimates are based on the Company’s behalf). Costs incurredservices performed pursuant to contracts with research institutions, contract research organizations in connection with clinical studies, investigative sites in connection with clinical studies, vendors in connection with preclinical development activities, and contract manufacturing organizations in connection with the production of materials for researchclinical trials. Further, the Company accrues expenses related to clinical trials based on the level of subject enrollment and developmentactivity according to the related agreement. The Company monitors subject enrollment levels and related activity to the extent reasonably possible and makes judgments and estimates in determining the accrued balance in each reporting period. Payments for these activities are expensed as incurred.
Atbased on the endterms of the reporting period,individual arrangements, which may differ from the Company compares payments made to third-party service providers topattern of costs incurred, and are reflected in the estimated progress toward completion of the research or development objectives. Such estimates are subject to changefinancial statements as additional information becomes available. Depending on the timing of payments to the service providers and the progress that the Company estimates has been made as a result of the services provided, the Company may record net prepaid or accrued expenses relating to these costs.
Upfront payments made to third parties who perform research and development services on the Company’s behalf are expensed as services are rendered.development.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTSIf the Company underestimates or overestimates the level of services performed or the costs of these services, actual expenses could differ from estimates. To date, the Company has not experienced significant changes in its estimates of preclinical studies and clinical trial accruals.
Patent Costs
PatentAll patent-related costs including related legal costs,incurred in connection with filing and prosecuting patent applications are expensed as incurred anddue to the uncertainty about the recovery of the expenditure. Amounts incurred are recorded withinclassified as general and administrative expenses in the consolidated statements of operations.
Stock-based Compensation
Stock-based compensation for employee and non-employee awards is measured on the grant date based on the fair value of the award and recognized on a straight-line basis over the requisite service period. The fair value of stock options to purchase common stock are measured using the Black-Scholes option pricing model. The Company accounts for forfeitures as they occur.
The fair value of stock options is determined by the Company using the methods and assumptions discussed below. Each of these inputs is subjective and generally requires significant judgment and estimation by management.
Expected Term—The expected term represents the period that stock-based awards are expected to be outstanding. The Company uses the “simplified method” to estimate the expected term of stock option grants. Under this approach, the weighted-average expected life is presumed to be the average of the contractual term of ten years and the weighted-average vesting term of the Company stock options, taking into consideration multiple vesting tranches. The Company utilizes this method due to lack of historical data and the plain-vanilla nature of the Company’s stock-based awards.
Expected Volatility—The Company has limited information on the volatility of its common stock as the shares were not actively traded on any public markets until recently. The expected volatility was derived from the historical stock volatilities of comparable peer public companies within its industry. These companies are considered to be comparable to the Company’s business over a period equivalent to the expected term of the stock-based awards.
Risk-Free Interest Rate—The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the date of grant for zero-coupon U.S. Treasury notes with maturities approximately equal to the and stock options expected term.
Expected Dividend Rate—The expected dividend is zero as the Company has not paid, nor does it anticipate paying, any dividends on its stock options in the foreseeable future.
Grant Revenue Recognition
The Company generates revenue from government contracts that reimburse the Company for certain allowable costs for funded projects.
The Company recognizes funding received as grant revenue for the Company’s grant from the NIA, rather than as a reduction of research and development expenses, because the Company is the principal in conducting the research and development activities and these contracts are central to its ongoing operations. Revenue is recognized as the qualifying expenses related to the contracts are incurred. Revenue recognized upon incurring qualifying expenses in advance of receipt of funding is recorded in the Company’s consolidated balance sheets as accounts receivable. Funding received in advance of services rendered are recorded in the Company’s consolidated balance sheets as deferred grant revenue. The related costs incurred by the Company are included in research and development expense in the Company’s consolidated statements of operations.
Income Taxes
As a corporation, theThe Company usesaccounts for income taxes under the asset and liability method, which requires the recognition of accountingdeferred tax assets and liabilities for income taxes. Deferredthe expected future tax consequences of events that have been included in the consolidated financial statements. Under this method, deferred tax assets and liabilities are recognized fordetermined on the estimated future tax consequences attributable tobasis of the differences between the financial statement carrying amountsand tax basis of existing assets and liabilities and their respectiveby using enacted tax bases and operating loss and credit carryforwards. Deferredrates in effect for the year in which the differences are expected to recover or settle. The effect of a change in tax rates on deferred tax assets and liabilities is recognized in income for the period that includes the enactment date.
The deferred tax assets are measured using enacted tax rates expectedrecognized to apply to taxable income in the years in which those temporary differencesextent the Company believes that these assets are expectedmore likely than not to be recovered or settled.realized. A valuation allowance is provided when it is more likely than not that some portion or all of a deferred tax asset will not be realized. The Company recognizesDue to the benefit of an uncertainCompany’s historical operating performance and the recorded cumulative net losses in prior fiscal periods, the net deferred tax position that it has taken or expects to take on its income tax return it files, if suchassets have been fully offset by a position is more likely than not to be sustained.valuation allowance.
ASC 740-10 defines the criterion an individual tax position must meet for any part of the benefit of the tax position to be recognized in financial statements prepared in conformity with GAAP. The Company may recognize the tax benefit from anrecords uncertain tax position only ifpositions using a two-step process. First, the Company determines whether it is more likely than not suchthat the tax positionpositions will be sustained on examination by the taxing authorities, based solely onbasis of the technical merits of the respectiveposition. Second, for those tax position. The tax benefits recognized inpositions that meet the financial statements from such a tax position should be measured based onmore-likely-than-not recognition threshold, the Company recognizes the largest amount of tax benefit having a greaterthat is more than 50.0% likelihood of being50% likely to be realized upon ultimate settlement with the related tax authority. In accordance with the disclosure requirements of ASC 740-10, the Company’s policy on income statement classification of interest and penalties related to income tax obligations is to include such items as part of total interest expense and other expense, respectively.
Stock-based Compensation
The Company measures stock-based awards at grant-date fair valuerecognizes interest and records compensation expense on a straight-line basis over the vesting period of the award. The Company uses the Black-Scholes Modelpenalties, if any, related to value its stock option awards. Estimating the fair value of stock option awards requires management to apply judgment and make estimates, including the volatility of the Company’s common stock, the expected term of the Company’s stock options, the expected dividend yield and the fair value of the Company’s common stockunrecognized tax benefits on the measurement date. As a result, if factors changeinterest expense line and management uses different assumptions, stock-based compensationother expense could be materially different for future awards.
For certain stock option grants,line, respectively, in the expected term was estimated using the “simplified method” for employee options as the Company has limited historical information to develop reasonable expectations about future exercise patternsaccompanying statements of operations. Accrued interest and post vesting employment termination behavior for its stock option grants. The simplified method is basedpenalties are included on the average of the vesting tranches and the contractual life of each grant. During the year ended December 31, 2020, it became apparent that the expected term of the Company's stock options was commensurate with the contractual life (i.e. 10 years) of the stock option and therefore the Company began to use the contractual life as the expected term.
For stock price volatility, the Company uses a combination of their own historical stock price and comparable public companies as a basis for its expected volatility to calculate the fair value of option grants. The Company assumes no dividend yield because dividends are not expected to be paidrelated liability lines in the near future, which is consistent with the Company’s history of not paying dividends. The risk-free interest rate is based on U.S. Treasury notes with a term approximating the expected life of the option. The Company accounts for forfeitures in the periods they occur.consolidated balance sheets.
Net Loss Per Common ShareLoss Per Share
Basic net loss per share is computed by dividing net loss attributable to common stockholders by the weighted averageweighted-average number of shares of common stock outstanding during each period.period (and potential shares of common stock that are exercisable for little or no consideration). Diluted net loss per share includes the effect, if any, from the potential exercise or conversion of securities such as convertible debt, convertible preferred stock, common stock warrants and stock options and unvested restricted stock thatwhich would result in the issuance of incremental shares of common stock. In computing the basic andFor diluted net loss per share, applicable to common stockholders, the weighted averageweighted-average number of shares remainsof common stock is the same for both calculationsbasic net loss per share due to the fact that when a net loss exists, dilutive sharessecurities are not included in the calculation as the impact is anti-dilutive.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS The pre-funded warrants to purchase common stock issued in connection with the Merger are included in the calculation of basic and diluted net loss per share as the exercise price of $0.001 per share is non-substantive and is virtually assured. The pre-funded warrants are more fully described in Note 11.
The following potentially dilutive securities outstanding have been excluded from the computation of diluted weighted average shares outstanding, as they would be anti-dilutive:
| | | December 31, | |
| | | 2023 | | | 2022 | |
| | | | | | | | | |
Preferred Series A-1 | | | - | | | | 1,960,600 | |
Preferred Series A-2 | | | - | | | | 335,711 | |
Preferred Series B | | | - | | | | 1,034,890 | |
Warrants | | | 598,457 | | | | 43,618 | |
Stock options | | | 349,384 | | | | 114,516 | |
Total | | | 947,841 | | | | 3,489,335 | |
| | | December 31, | |
| | | 2020 | | | 2019 | |
Common stock warrants | | | 9,100,112 | | | | 22,385,141 | |
Stock options | | | 2,240,204 | | | | 309,276 | |
| | | | 11,340,316 | | | | 22,694,417 | |
Segments
The Company has one operating segment. The Company’s chief operating decision maker, its Chief Executive Officer, manages the Company’s operations on a consolidated basis for purposes of allocating resources.
Recently Issued But Not Yet Adopted Accounting Pronouncements
In December 2019,January 2021, the FASB issued ASU No. 2019-12, Income Taxes2021-01 “Reference Rate Reform (Topic 740)848): SimplifyingScope” (“ASU 2021-01”), which was effective immediately and permits entities to elect certain optional expedients and exceptions when accounting for derivatives and certain hedging relationships affected by changes in interest rates and the Accounting for Income Taxes. This guidance appliestransition. Additionally, ASU 2022-06 “Reference Rate Reform (Topic 848): Deferral of the Sunset Date of Topic 848” defers the sunset date of ASC 848 from December 31, 2022 to all entities and aims to reduce the complexity of tax accounting standards while enhancing reporting disclosures. ThisDecember 31, 2024. The new guidance is effective for fiscal years beginning after December 15, 2020 and interim periods therein. Early adoption is permitted for any annual periods for which financial statements have not been issued and interim periods therein.31, 2024. The Company isdoes not currently analyzing the impact of ASU No. 2019-12 on the consolidated financial statements.
Recently Adopted Accounting Pronouncements
In August 2018, the FASB issued ASU 2018-03, Disclosure Framework- Changes to the Disclosure Requirements for Fair Value Measurements, which changes the fair value measurement disclosure requirements of ASC 820. The goal of the ASU is to improve the effectiveness of ASC 820's disclosure requirements. The Company adoptedbelieve that this standard in January 2020 and the adoption did nottransition from LIBOR will have a material impact on its relatedfinancial statements.
In November 2023, the FASB issued ASU No. 2023-07 "Segment Reporting - Improvements to Reportable Segment Disclosures" (“ASU 2023-07”), which updates reportable segment disclosure requirements, primarily through enhanced disclosures about significant segment expenses and information used to assess segment performance. The guidance is effective for the Company beginning in the year ended December 31, 2025, with early adoption permitted. The Company expects the new guidance will have an immaterial impact on its consolidated financial statements.
In December 2023, the FASB issued ASU No. 2023-09, Income Taxes (Topic 740): “Improvements to Income Tax Disclosures” (“ASU 2023-09”). ASU No. 2023-09 is intended to improve income tax disclosure requirements by requiring (1) consistent categories and greater disaggregation of information in the rate reconciliation and (2) the disaggregation of income taxes paid by jurisdiction. The guidance makes several other changes to the income tax disclosure requirements. The guidance in ASU 2023-09 will be effective for annual reporting periods in fiscal years beginning after December 15, 2024. The Company is currently evaluating the impact that the adoption of ASU 2023-09 will have on its consolidated financial statements and disclosures.
4.Property and Equipment Merger
PropertyOn August 16, 2023, the Company completed the Merger of EIP and equipment consistsMerger Sub as discussed in Note 1. For financial reporting purposes, EIP was determined to be the accounting acquirer based upon the terms of the following: Merger and other factors, including: (i) EIP securityholders immediately prior to the Merger owning approximately 76% of the Company immediately following the Merger, (ii) EIP appointing the majority (five of seven) of the Company’s Board immediately following the Merger and (iii) former EIP management holding the majority of key positions of management, including the Chief Executive Officer and Chairman of the Board positions, immediately following the Merger. The Merger was also accounted for as a reverse recapitalization under US GAAP because the primary assets of the Company immediately prior to the Merger were cash and cash equivalents. Accordingly, (i) for all periods prior to the Merger, EIP’s historical financial statements and results of operations replace and are deemed to be the Company’s financial statements and results of operations for such periods, (ii) the Merger was treated as the equivalent of EIP issuing shares of common stock to the holders of the Company's common stock immediately prior to the Merger as consideration to acquire the net assets of the Company, and (iii) the net assets of the Company as of immediately prior to the Merger were recorded at their acquisition-date fair value in the consolidated financial statements of EIP. Immediately after the Merger, there were approximately 5,674,277 shares of the Company’s common stock outstanding.
| | | December 31, | |
| | | 2020 | | | 2019 | |
Laboratory equipment | | $ | 182,357 | | | $ | 182,357 | |
Furniture and office equipment | | | 151,442 | | | | 151,442 | |
Leasehold improvements | | | 430,000 | | | | 430,000 | |
Total property and equipment | | | 763,799 | | | | 763,799 | |
Less: accumulated depreciation and amortization | | | (614,601 | ) | | | (511,433 | ) |
Property and equipment, net | | $ | 149,198 | | | $ | 252,366 | |
DepreciationThe following table shows the net assets acquired in the Merger:
| | | August 16, | |
| | | 2023 | |
Cash and cash equivalents | | $ | 12,705,140 | |
Prepaid and other assets | | | 406,488 | |
Accounts payable and accrued expenses | | | (1,223,871 | ) |
Total net assets assumed | | | 11,887,757 | |
Minus: Transaction costs | | | (1,548,643 | ) |
Total net assets assumed minus transaction costs | | $ | 10,339,114 | |
5. Significant Agreements and amortization expenseContracts
Vertex Option and License Agreement
In August 2012, the Company entered the Vertex Agreement, as amended, to acquire an exclusive license to develop and commercialize a drug candidate “VX-745” from Vertex. In August 2014, the Company exercised its
option to acquire the license and paid an option fee of $100,000, which was expensed as incurred as a component of research and development expense.
The Vertex Agreement granted the Company the exclusive worldwide use of VX-745 in the field of diagnosis, treatment and prevention of Alzheimer’s disease and related central nervous system disorders in humans.
As part of the Vertex Agreement, the Company is obligated to make certain payments totaling up to approximately $0.1$117.0 million upon achievement of certain regulatory and sales milestones, and royalties on net sales of products on indications covered by the Vertex Agreement. The first expected milestone events concern filing of an NDA, with the FDA for bothmarketing approval of neflamapimod, in the U.S., or a similar filing for a non-U.S. major market, as specified in the Vertex Agreement, and such royalties will be on a sliding scale of percentages of net sales in the low- to mid-teens, depending on the amount of net sales in the applicable years. The Company is also obligated to make a milestone payment to Vertex upon net sales reaching a certain specified amount in any 12-month period. The Vertex Agreement states that royalties will be reduced by 50% during any portion of the royalty term when there is no valid claim of an issued patent within specified patent rights covering the licensed product. The Company also has the right to deduct, on a country by country basis, from royalties otherwise payable to Vertex under the terms of the Vertex Agreement, 50% of all royalties, upfront fees, milestones and other payments paid by the Company or any of the Company’s affiliates or sublicensees to third parties under licenses that are necessary for the development, manufacture, sale or use of a licensed product, provided that in no event will the royalty payable to Vertex be reduced to less than 50% of the rates specified in the Vertex Agreement, subject to certain adjustments specified therein. The Company has made a total of $100,000 in payments to Vertex related to the Vertex Agreement. No payments were made during the years ended December 31, 20202023 and 2019.2022.
National Institute of Aging Grant
In January 2023, the Company was awarded a $21.0 million grant from the NIA to support a Phase 2b study of neflamapimod in dementia with Lewy bodies. The grant monies are expected to be received over a period of three years including $6.7 million in 2023, $8.1 million in 2024 and $6.2 million in 2025.
The total revenue recognized from the NIA Grant was $7.1 million year ended December 31, 2023. As of December 31, 2023, total cash funding of $6.2 million has been received from the NIA Grant, resulting in approximately $15.8 million in funding remaining. In addition, $0.9 million has been recorded as a receivable in the consolidated balance sheet at December 31, 2023, which was received subsequent to December 31, 2023.
The Company received access to the current year 2 (i.e., the year ending December 31, 2024) funding in the amount of $7.3 million in February 2024. This amount was 90% of the full year 2 amount provided for in the NIA Grant due to current NIA policy as a result of the U.S. government currently being funded on the basis of a continuing resolution (the “Continuing Resolution”).
6. Prepaid Expenses
Prepaid expenses consisted of the following:
| | | December 31, | |
| | | 2023 | | | 2022 | |
Prepaid clinical expenses | | $ | 711,362 | | | $ | - | |
Insurance | | | 436,859 | | | | 9,937 | |
Rent | | | - | | | | 2,455 | |
Prepaid professional services | | | 37,917 | | | | - | |
Other | | | 70,363 | | | | 51,735 | |
Total | | $ | 1,256,501 | | | $ | 64,127 | |
5.7. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consistconsisted of the following:
| | | December 31, | |
| | | 2023 | | | 2022 | |
Employee compensation costs | | $ | 1,026,054 | | | $ | 364,070 | |
Clinical development costs | | | 389,045 | | | | 23,185 | |
Professional fees | | | 309,062 | | | | 206,675 | |
State franchise and excise tax | | | 120,456 | | | | - | |
Other | | | 88,659 | | | | 50,322 | |
Total | | $ | 1,933,276 | | | $ | 644,252 | |
| | | December 31, | |
| | | 2020 | | | 2019 | |
Accrued payroll and payroll related expenses | | $ | 653,899 | | | $ | 182,708 | |
Accrued professional fees | | | 31,809 | | | | 48,338 | |
Accrued clinical studies expenses | | | 1,055,398 | | | | 57,378 | |
Other | | | 35,364 | | | | 70,108 | |
Total | | $ | 1,776,470 | | | $ | 358,532 | |
8. Line of Credit
The Company established a line of credit with a lender during the year ended December 31, 2020 in the amount of $2.5 million with a variable interest rate of 1.75% over the 30-day LIBOR (7.22% and 6.08% at December 31, 2023 and December 31, 2022, respectively). The line is secured by the personal assets of the Company’s Chief Executive Officer and the former Chair of the Board.
No drawdowns were made, and no costs incurred related to the line of credit during the year ended December 31, 2023 or 2022. The line of credit was terminated on February 14, 2024.
9. Convertible Notes
In December 2020, EIP issued the 2020 Notes to predominantly related party investors for proceeds of $5.1 million. In December 2021, EIP issued the 2021 Notes to predominantly related party investors for proceeds of $6.0 million. Upon issuance, the Company elected the fair value option for the Convertible Notes in accordance with ASC 825, “Financial Instruments,” pursuant to which the entire instrument, including interest expense, is measured at fair value with the initial change in fair value deemed to be a capital contribution and any subsequent changes in fair value being recorded to other income (expense) on the consolidated statement of operations. The fair value adjustments recognized in other income (expense) were $5.4million and $(2.4)million for the years ended December 31, 2023 and 2022, respectively.
In April 2022, the Company entered into the 2022 Notes Amendment with the noteholders for the 2020 Notes (the “Amendment”). In accordance with the Amendment, the maturity of the 2020 Notes was extended from June 2022 to December 2023, the interest rate was modified so interest accrued at 5% through the original maturity of June 2022 and at 0% thereafter, the conversion discount was increased from 20% to 30%, and a conversion price limit of $3.00 was established, as discussed further below. Expenses associated with the amendment were de minimis.
DIFFUSION PHARMACEUTICALS INC.The Company concluded the 2022 Notes Amendment qualified as a troubled debt restructuring, in accordance with FASB ASC 470, Debt, as the noteholders for the 2020 Notes, for economic reasons related to the Company’s financial difficulties, granted concessions to the Company. The Company concluded no gain or loss, and no adjustment to, or reclassification of, the carrying value of the 2020 Notes were considered necessary as a result of the 2022 Notes Amendment. In addition, the Company concluded there was no other financial statement impact as a result of the 2022 Notes Amendment, as any prospective change would be related to interest and, as a result of the amendment, the interest rate decreased to 0% following the original maturity of June 2022.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
In June 2023, EIP entered into the 2023 Notes Amendment which amended the conversion price of the Convertible Notes to $1.47 per share of EIP Common Stock upon effectiveness of the Merger with the Diffusion or a 30% conversion discount upon the occurrence of any other reverse merger. Further, the 2023 Notes Amendment provided that if the Merger with the Company resulted in a holder of these notes beneficially owning more than 9.99% of the outstanding voting stock of the Company, then, the holder of these notes shall be granted pre-funded warrants in lieu of the Company’s common stock for the conversion of any principal and accrued but unpaid interest in excess of such threshold. The exercise price of one share of the Company’s common stock under this pre-funded warrant is equal to $0.001 (Note 11).
The 2023 Notes Amendment qualified as a modification in accordance with FASB ASC 470 Debt, since there were no concessions granted and no substantive change to the fair value of the conversion option before and after the 2023 Notes Amendment. There was no financial statement impact as a result of the 2023 Notes Amendment other than the change in fair value of the Convertible Notes during the year ended December 31, 2023 and debt issuance costs of approximately $50,000 that was recorded to general and administrative expenses in the statements of operations.
As a result of the Merger (Note 4), pursuant to the terms thereof, the Convertible Notes converted into shares of EIP Common Stock which were subsequently converted into the right to exchange such shares for 897,272 shares of the Company’s common stock and, in certain cases, pre-funded warrants to purchase the Company’s common stock. Accordingly, the Convertible Notes were adjusted to fair value prior to conversion by multiplying the trading price of the Company’s common stock at the date of the Effective Time and the 795,905 common shares and 101,367 pre-funded warrants issued upon conversion. The Company recorded a gain on the fair value adjustment of the Convertible Notes of $5.4 million for year ended December 31, 2023 and recorded $7.0 million to additional paid in capital for the issuance of common stock upon settlement of the Convertible Notes.
6.10. Commitments and Contingencies
Office Space Lease CommitmentOperating Leases
The Company has a non-cancelable operating lease for office and laboratoryshort-term agreement to utilize membership-based co-working space in Charlottesville, Virginia which beganand a short-term lease for office space in April 2017 and, as of December 31, 2020, has a remaining lease term of approximately 1.3 years. The discount rate used to account for the Company's operating lease is the Company’s estimated incremental borrowing rate of 10.0%. The original term of the lease ends in the second quarter of 2022 and the Company has an option to extend for another 5 years. This option to extend was not recognized as part of the Company's measurement of the right-of-use asset and operating lease liability as of December 31, 2020.
Boston, Massachusetts. Rent expense related to the Company's operating leasewas approximately $34,000 and $45,000 for both the years ended December 31, 20202023 and 2019 was approximately $0.1 million. Future minimum rental payments under the Company's non-cancelable operating lease at December 31, 2020 were as follows:2022, respectively.
| | | Rental Commitments | |
2021 | | $ | 118,519 | |
2022 | | | 39,735 | |
Total | | | 158,254 | |
Less: imputed interest | | | (9,092 | ) |
| | | $ | 149,162 | |
Research and Development Arrangements
In the course of normal business operations, the Company enterswould enter into agreements with universities and contract research organizations, or CROs to assist in the performance of research and development activities and contract manufacturers to assist with chemistry, manufacturing, and controls related expenses. Expenditures to CROs representrepresented a significant cost in clinical development for the Company. The Company could also enter into additional collaborative research, contract research, manufacturing, and supplier agreements in the future, which may require upfront payments and long-term commitments of cash.
Defined Contribution Retirement Plan
The Company has established its 401(k) Plan, which covers all employees who qualify under the terms of the plan. Eligible employees may elect to contribute to the 401(k) Plan up to 90.00%90% of their compensation, limited by the IRS-imposed maximum. The Company provides a safe harbor match with a maximum amount of 4.0%4% of the participant’s compensation. The Company made matching contributions under the 401(k) Plan of approximately $56,000 and $68,000de minimis amounts for the years ended December 31, 20202023 and 2019, respectively.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS2022.
Legal Proceedings
On August 7, 2014, a complaint was filed in the Superior Court of Los Angeles County, California by Paul Feller, the former Chief Executive Officer of the Company'sCompany’s legal predecessor under the caption Paul Feller v. RestorGenex Corporation, Pro Sports & Entertainment, Inc., ProElite, Inc. and Stratus Media Group, GmbH (Case No. BC553996). The complaint asserts various causes of action, including, among other things, promissory fraud, negligent misrepresentation, breach of contract, breach of employment agreement, breach of the covenant of good faith and fair dealing, violations of the California Labor Code and common counts. The plaintiff is seeking, among other things, compensatory damages in an undetermined amount, punitive damages, accrued interest and an award of attorneys’ fees and costs. On December 30, 2014, the Company filed a petition to compel arbitration and a motion to stay the action. On April 1, 2015, the plaintiff filed a petition in opposition to the Company’s petition to compel arbitration and a motion to stay the action. After a related hearing for the petition and motion on April 14, 2015, the Courtcourt granted the Company’s petition to compel arbitration and a motion to stay the action. On January 8, 2016, the plaintiff filed an arbitration demand with the American Arbitration Association. On November 19, 2018 at an Order to Show Cause Re Dismissal Hearing, the Courtcourt found sufficient grounds not to dismiss the case and an arbitration hearing was scheduled, originally for November 2020. In August 2020 but later postponed due to the ongoing COVID-19 pandemic and related restrictions on gatherings in the State of California,California. In addition, following the November 2018 hearing, an automatic stay was placed on the arbitration hearingin connection with the plaintiff filing for personal bankruptcy protection. On October 22, 2021, following a determination by the bankruptcy trustee not to pursue the claims and release them back to the plaintiff, the parties entered into a stipulation to abandon arbitration and return the matter to state court. A case management conference was postponedheld on February 23, 2022 at which an initial trial date of May 24, 2023 was set, and the parties have agreed to August 16, 2021. stipulate to mediation in advance of the trial. On October 20, 2022, the parties filed a joint stipulation to continue the trial and certain deadlines related to the mediation in order to allow plaintiff’s counsel to continue to seek treatment for an ongoing medical issue. On November 1, 2022, based on the parties joint stipulation, the court entered an order continuing the trial date to October 25, 2023, on October 6, 2023, the court entered an order further continuing the trial date to April 24, 2024 , and on March 3, 2024, based on an additional joint stipulation of the parties, the court entered an order continuing the trial date to October 23, 2024.
The Company believes that is has meritorious defenses to the claims alleged in this matter and is without merit and intends to defend the arbitrationdefending itself vigorously. However, at this stage, the Company is unable to predict itsthe outcome and the possible loss or range of loss, if any, associated with its resolution or any potential effect the matter may have on the Company’s financial position. Depending on the outcome or resolution of this matter, it could have a material effect on the Company’s financial position.position, results of operations and cash flows.
7.11. Stockholders’Stockholders' Equity (Deficit) and Common Stock Warrants
2020 Common Stock Offering
In May 2020,On August 16, 2023 in connection with the Company completedclosing of the May 2020 Offering, a public offeringMerger, the following is reflected on the consolidated financial statements of 11,428,572convertible preferred stock and stockholders’ equity (deficit) for the year ended December 31, 2023: (i) the issuance of 795,905 shares of common stock for a purchase price of $1.05 per share for net proceeds of $10.3 million after deducting commissions, discounts, and other offering costs. In addition, at101,367 pre-funded warrants upon the closingsettlement of the May 2020 Offering,Convertible Notes, (ii) the Company issuedconversion of 3,331,201 shares of convertible preferred stock into 2,936,566 shares of common stock and 394,628 prefunded warrants, to purchase up to 571,429and (iii) the issuance of 1,360,244 shares of common stock to designees of the placement agent for the May 2020 Offering. The placement agent's warrants have an exercise price of $1.3125 per share and a term of five years from the date of issuance.
Additionally, also in May 2020, the Company entered into a warrant exercise agreement with an investor who held a previously outstanding warrant to purchase up to an aggregate of 5,000,000 shares of our common stock at an exercise price of $0.35 per share(the Prior Warrant). InDiffusion stockholders as consideration for the exercise of the Prior Warrant for cash and an additional $0.125 per each share of common stock resulting from the exercise, the exercising investor received new unregistered warrants to purchase up to an aggregate of 5,000,000Merger.
In July 2023, EIP sold 63,422 shares of common stock in a private placement. The warrants are exercisable immediately at an exercise price of $0.5263$12.78 per share and exercisable until November 8, 2025. The Company recognized a deemed dividend of $2.0 million to reflect the consideration given as an inducement(as adjusted for the investor to exercise the warrants. This deemed dividend is recorded in the Company's consolidated statement of operations during the year ended December 31, 2020 as an increase to the net loss attributable to common stockholders for purposes of computing net loss per share, basic and diluted.
In connection with the warrants exercised in May 2020, the Company issued warrants to purchase up to 250,000 shares of common stock to the placement agent with an exercise price of $0.5938 per share and otherwise have identical terms to the warrants issued to the investor.
2019 Common Stock Offerings
In December 2019, Company completed the December 2019 Offering, an offering of 6,266,787 shares of its common stock and warrants to purchase 6,266,787 shares of common stock. The shares of common stock and warrants were sold for a combined purchase price of $0.5585 per shareExchange Ratio) for net proceeds of $3.0approximately $0.8 million. The December 2019 Offering warrants are exercisable beginning on the date of their issuance until June 13, 2025 at an initial exercise price equal to $0.4335 per share.
In addition, at the closing of the December 2019 Offering, the Company issued warrants to purchase up to 313,339 shares of common stock to designees of the placement agent. The placement agent's warrants have an exercise price of $0.6981 per share and a term of five years from the date of issuance.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
In November 2019, the Company completed the November 2019 Offering, a registered direct public offering of 5,104,429 shares of its common stock, and 6,324,143 pre-funded warrants each to purchase one share of common stock, together with warrants to purchase up to 22,857,144 shares of common stock at a combined public offering price of $0.35 per share and associated warrants for total net proceeds of $3.3 million. The warrants were issued with an exercise price of $0.35 per warrant and are exercisable beginning on their date of issuance. Of the warrants issued, 11,428,572 have a term of 18 months and 11,428,572 have a term of 5 years. During the year ended December 31, 2019, 11,091,716 of those warrants were exercised for additional proceeds of $3.9 million.
In addition, at the closing of the November 2019 Offering, the Company issued warrants to purchase up to 571,429 shares of common stock to designees of the placement agent. The placement agent's warrants have an exercise price of $0.4375 per share and a term of five years from the date of issuance.
In May 2019, the Company completed the May 2019 Offering, a registered direct public offering of 1,317,060 shares of common stock and a private placement of warrants to purchase 1,317,060 shares of common stock. The shares of common stock and warrants were sold for a combined purchase price of $4.895 for total net proceeds of $5.6 million. The warrants are exercisable beginning on the date of their issuance until November 29, 2024 at an initial exercise price equal to $5.00.
In addition, at the closing of the May 2019 Offering, the Company issued warrants to purchase up to 65,853 shares of common stock to designees of the placement agent. The placement agent's warrants have an exercise price of $6.11875 per share, a term of 5 years from the date of issuance and otherwise substantially similar terms to the form of the investor warrant.
Common Stock Warrants
During its evaluation of equity classification for the Company's common stock warrants issued in 2020 and 2019, the Company considered the conditions as prescribed within ASC 815-40, Derivatives and Hedging, Contracts in an Entity’s own Equity. The conditions within ASC 815-40 are not subject to a probability assessment. The warrants do not fall under the liability criteria within ASC 480, Distinguishing Liabilities from Equity as they are not puttable and do not represent an instrument that has a redeemable underlying security. The warrants do meet the definition of a derivative instrument under ASC 815, but are eligible for the scope exception as they are indexed to the Company’s own stock and would be classified in permanent equity if freestanding.
As of December 31, 2020,2023, the Company had the following warrants outstanding to acquire shares of its common stock:
| | | Outstanding | | | Range of exercise price per share | | Expiration dates |
Common stock warrants issued in 2017 related to Series A convertible preferred stock offering | | | 903,870 | | | | $33.30 | | | March 2022 |
Common stock warrants issued in 2018 related to the January 2018 common stock offering | | | 1,181,421 | | | $12.00 | - | $15.00 | | January 2023 |
Common stock warrants issued related to the May 2019 Offering | | | 1,382,913 | | | $5.00 | - | $6.11875 | | May and December 2024 |
Common stock warrants issued related to the November 2019 Offering | | | 497,140 | | | $0.35 | - | $0.4375 | | May 2024 |
Common stock warrants issued related to the December 2019 Offering | | | 313,339 | | | $0.4335 | - | $0.6981 | | December 2024 and June 2025 |
Common stock warrants issued related to the May 2020 Offering | | | 571,429 | | | | $1.31 | | | March 2025 |
Common stock warrants issued related to the May 2020 Investor Warrant Exercise | | | 4,250,000 | | | $0.5263 | - | $0.5938 | | November 2025 |
| | | | 9,100,112 | | | | | | | |
| | | Warrants | | | Exercise | | |
| | | Outstanding | | | Price | | Expiration Date |
Historical Diffusion common stock warrants | | | 58,844 | | | $26.27 | - | $459.06 | | May 2024 through February 2026 |
Historical EIP common stock warrants | | | 43,618 | | | $19.81 | | April 2028 |
Pre-funded warrants issued related to closing of reverse recapitalization | | | 495,995 | | | $0.001 | | None |
| | | | 598,457 | | | | | | |
DuringUpon completion of the year ended December 31, 2020, noMerger, the Convertible Notes and outstanding shares of EIP preferred stock converted into shares of EIP Common Stock which were subsequently converted into the right to exchange such shares for shares of the Company’s common stock or, in certain cases, pre-funded warrants expiredto purchase the Company’s common stock. All of the warrants are equity-classified because they are indexed to the Company’s own shares and 19,106,504 warrants were exercised for gross proceeds of $8.0 million. Duringmeet the year ended December 31, 2019, 1,767 warrants expired and 17,415,859 warrants were exercised for net proceeds of $3.9 million.criteria to be classified as an equity instrument.
The Company is party to the 2022 Sales Agreement with BTIG. The 2022 Sales Agreement is an "at-the-market" sales agreement pursuant to which the Company may, from time to time and through BTIG as the Company’s agent, sell up to an aggregate of $20.0 million in shares of common stock by any permissible method deemed an “at the market offering” as defined in Rule 415(a)(4) under the Securities Act. As of the date of this Annual Report, however, the Company has not sold any shares pursuant to the 2022 Sales Agreement.
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
8.12. Stock-Based Compensation
2015 Equity Plan
The 2015 Equity Plan provides for increases to the number of shares reserved for issuance thereunder each January 1 equal to 4.0% of the total shares of the Company’s common stock outstanding as of the immediately preceding December 31, unless a lesser amount is stipulated by the Compensation Committee of the Company's boardCompany’s Board of directors. Accordingly, 2,560,618 shares were added to the reserve as of January 1, 2021, which shares may be issued in connection with the grant of stock-based awards, including stock options, restricted stock, restricted stock units, stock appreciation rights and other types of awards as deemed appropriate, in each case, in accordance with the terms of the 2015 Equity Plan.Directors. As of December 31, 2020,2023, there were no12,580 shares of common stock available for future issuance under the 2015 Equity Plan. Further,On January 1, 2024, the Company granted options to purchase 270,000number of shares of common stock to employees during the year ended December 31, 2020 that were granted outside ofavailable for future issuance under the 2015 Equity Plan as an inducement materialincreased by 226,981.
2018 Employee, Director and Consultant Equity Incentive Plan
On March 28, 2018, EIP adopted the 2018 Plan, which was assumed by the Company pursuant to the employee’s acceptance of employment with the Companyand in accordance with the terms of the Merger Agreement. Under the 2018 Plan, the Company may issue incentive stock options, non-qualified stock options, stock grants, and other stock-based awards to employees, directors, and consultants, as specified in the 2018 Plan and subject to applicable SEC and Nasdaq Listing Rule 5635(c)(4). Generally,rules and regulations. The Board of Directors has the authority to determine to whom options or stock will be granted, the number of shares, the term, and the exercise price. Options granted under the 2018 Plan have a term of up to ten years and generally vest over a four-year period with 25% of the options have a ten (10) year contractual termvesting after one-year of service and vest in equalthe remainder vesting monthly installments over three (3) years.thereafter. As of December 31, 2023, there were no shares available for issuance.
The Company recorded stock-based compensation expense in the following expense categories of its consolidated statements of operations for the periods indicated:operations:
| | | Year Ended December 31, | |
| | | 2023 | | | 2022 | |
Research and development | | $ | 143,685 | | | $ | 174,710 | |
General and administrative | | | 263,947 | | | | 159,125 | |
Total stock-based compensation expense | | $ | 407,632 | | | $ | 333,835 | |
| | | Year ended December 31, | |
| | | 2020 | | | 2019 | |
Research and development | | $ | 164,791 | | | $ | 54,155 | |
General and administrative | | | 571,328 | | | | 461,607 | |
Total stock-based compensation expense | | $ | 736,119 | | | $ | 515,762 | |
The following table summarizes the activity related to all stock option:option grants for the year ended December 31, 2023:
| | | Number of
Options | | | Weighted
average
exercise price
per share | | | Weighted
average
remaining
contractual life
(in years) | | | Aggregate
Intrinsic
Value | |
Balance at January 1, 2019 | | | 203,736 | | | $ | 88.14 | | | | | | | | | |
Granted | | | 117,270 | | | | 2.62 | | | | | | | | | |
Forfeited | | | (11,583 | ) | | | 83.81 | | | | | | | | | |
Expired | | | (147 | ) | | | 276.00 | | | | | | | | | |
Balance at December 31, 2019 | | | 309,276 | | | $ | 55.78 | | | | 6.98 | | | | — | |
Granted | | | 1,931,100 | | | | 0.68 | | | | | | | | | |
Expired | | | (172 | ) | | | 142.50 | | | | | | | | | |
Outstanding at December 31, 2020 | | | 2,240,204 | | | $ | 8.28 | | | | 8.90 | | | $ | 289,067 | |
Exercisable at December 31, 2020 | | | 1,129,733 | | | $ | 15.64 | | | | 8.28 | | | $ | 219,010 | |
Vested and expected to vest at December 31, 2020 | | | 2,240,204 | | | $ | 8.28 | | | | 8.90 | | | $ | 289,067 | |
| | | Number of Options | | | Weighted average exercise price per share | | | Weighted Average Remaining Contractual Term | | | Aggregate Intrinsic Value | |
Balance at January 1, 2023 | | | 114,516 | | | $ | 25.98 | | | | 6.72 | | | | | |
Options assumed in the merger | | | 52,574 | | | $ | 313.67 | | | | | | | | | |
Granted | | | 198,600 | | | $ | 5.95 | | | | | | | | | |
Cancelled | | | (15,957 | ) | | $ | 180.80 | | | | | | | | | |
| Exercised | | | (359 | ) | | $ | 5.33 | | | | | | | | | |
Outstanding at December 31, 2023 | | | 349,374 | | | $ | 51.15 | | | | 8.12 | | | $ | 358,340 | |
Exercisable at December 31, 2023 | | | 157,301 | | | $ | 103.67 | | | | 6.35 | | | $ | 39,820 | |
The weighted averageBlack-Scholes option-pricing model was used to estimate the grant date fair value of each stock option awards granted was $0.64 and $2.16 duringgrant at the years ended December 31, 2020 and 2019, respectively. The total fair valuetime of options vested duringgrant using the years ended December 31, 2020 and 2019 were $0.7 million and $0.6 million, respectively. No options were exercised during any of the periods presented. At December 31, 2020, there was $0.8 million of unrecognized compensation cost related to unvested options that will be recognized as expense over afollowing weighted-average period of 2.2 years.assumptions:
DIFFUSION PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
The grant date fair value of employee stock options is determined using the Black-Scholes Model. The following assumptions were used during the years ended December 31, 2020 and 2019:
| | | 2020 | | 2019 |
Expected term (in years) | | 5.31 | — | 10.00 | | 5.25 | — | 5.77 |
Risk-free interest rate | | 0.4% | — | 1.7% | | 1.9% | — | 2.5% |
Expected volatility | | 113.4% | — | 124.8% | | 112.4% | — | 114.4% |
Dividend yield | | — | — | — | | — | — | — |
Restricted Stock Unit Awards
| | | December 31, | |
| | | 2023 | | | 2022 | |
Expected term (in years) | | 5.75 | | | | 6.00 | |
Risk-free interest rate | | 3.9% | - | 4.5% | | | | 1.9 | % |
Expected volatility | | 81.7% | - | 83.3% | | | | 80.3 | % |
Dividend yield | | 0.0% | | | | 0.0 | % |
During the year ended December 31, 2020,2023, 359 shares underlying options were exercised, of which 193 were withheld as consideration for the Company granted 153,000 restricted stock unitsexercise price of such shares pursuant to membersthe cashless exercise provision of the board of directors of the Company. The weighted average grant date fair value of the restricted stock units grantedrelated option award agreement. No options were exercised during the year ended December 31, 20202022. At December 31, 2023, there was $0.65.$1.1 million of unrecognized compensation expense that will be recognized over a weighted-average period of 2.1 years.
Contributed Capital in lieu of Executive Compensation
In 2022, the former Chair of the Board and the Chief Executive Officer offered to forego, without repayment, certain compensation to ensure the Company had enough resources to maintain operations until the financial funding was completed. The shares begin to vest 18 months afteramount of $0.1 million for the grant date. The Company recognized approximately $18,000year ended December 31, 2022, which is recorded as contributed capital in expense related to these unitslieu of executive compensation in additional paid-in capital, will not be paid in cash, debt or equity in the future. No such event occurred during the year ended December 31, 2020. At December 31, 2020, there was approximately $82,000 of unrecognized compensation cost that will be recognized over a weighted average period of 2.4 years.2023.
9.13. Income Taxes
Since inception, the Company has incurred net losses, and until 2018, had not recorded any U.S. federal or state income tax benefits for the losses. In 2018, as a resultA reconciliation of the change in net operating loss carryforward period associated with the 2017 Tax Act, the Company recognized an income tax benefit to reflectat the adjustment allowed bystatutory federal income tax rate and income taxes as reflected in the 2017 Tax Act to utilize indefinite deferred tax liabilitiesfinancial statements as a source of income against indefinite lived portions of the Company’s deferred tax assets. In 2019, as a result of a change in ownership under the provisions of Internal Revenue Code Section 382, the cumulative benefit of net operating losses was remeasured which resulted in tax expense to reverse the 2018 benefit recordedDecember 31, 2023 and record a benefit relative to losses incurred in 2019 after the date of the change in ownership.2022:
Rate reconciliation: | | 2023 | | | 2022 | |
Federal tax benefit at statutory rate | | | 21.0 | % | | | 21.0 | % |
State tax, net of federal benefit | | | 6.3 | % | | | 8.0 | % |
Change in convertible debt | | | 52.8 | % | | | -8.6 | % |
Research & Development credit | | | 17.1 | % | | | 1.2 | % |
Change in valuation allowance | | | -95.9 | % | | | -15.8 | % |
Share-based compensation | | | -1.0 | % | | | 0.0 | % |
Other | | | -0.3 | % | | | -5.8 | % |
Total provision | | | 0.0 | % | | | 0.0 | % |
Deferred tax assets and liabilities are determined based on the differences between the financial statement carrying amounts and tax bases of assets and liabilities using enacted tax rates in effect for years in which differences are expected to reverse.
Significant components of the Company's deferred tax assets for federal income taxes as of December 31, 2023 and December 31, 2022 consisted of the following:
Deferred tax assets | | December 31, 2020 | | | December 31, 2019 | |
Net operating loss carryforwards | | $ | 3,864,189 | | | $ | 1,504,496 | |
Stock-based compensation | | | 1,571,227 | | | | 1,381,750 | |
Orphan Drug credits | | | 541,384 | | | | 81,700 | |
Lease liability | | | 38,394 | | | | 63,589 | |
Capitalized start-up costs and other | | | 10,709,631 | | | | 9,187,898 | |
Valuation allowance | | | (14,906,646 | ) | | | (12,051,440 | ) |
Deferred tax assets | | | 1,818,179 | | | | 167,993 | |
| | | | | | | | | |
Deferred tax liabilities | | | | | | | | |
Intangible assets | | | (2,223,678 | ) | | | (2,223,678 | ) |
Right of use asset | | | (38,394 | ) | | | (63,589 | ) |
Deferred tax liabilities | | | (2,262,072 | ) | | | (2,287,267 | ) |
| | | | | | | | | |
Net deferred tax liability | | $ | (443,893 | ) | | $ | (2,119,274 | ) |
| | | 2023 | | | 2022 | |
Deferred tax assets: | | | | | | | | |
Net operating loss | | $ | 10,495,881 | | | $ | 10,977,455 | |
Research and development credits | | | 708,443 | | | | 354,283 | |
Capitalized research expenditures | | | 2,271,853 | | | | 259,749 | |
Stock-based compensation | | | 567,473 | | | | 514,654 | |
Reserves and accruals | | | - | | | | 105,399 | |
Intangibles | | | 223,548 | | | | 262,872 | |
Gross deferred tax assets | | | 14,267,198 | | | | 12,474,412 | |
Less valuation allowance | | | (14,100,543 | ) | | | (12,474,412 | ) |
Total deferred tax assets | | | 166,655 | | | | - | |
| | | | | | | | | |
Deferred tax liabilities: | | | | | | | | |
Prepaids | | $ | (166,655 | ) | | $ | - | |
Gross deferred tax liabilities | | | (166,655 | ) | | | - | |
Deferred tax assets, net | | $ | - | | | $ | - | |
The Company does not have unrecognized tax benefits as of December 31, 20202023 or December 31, 2019.2022. The Company recognizes interest and penalties accrued on any unrecognized tax benefits as a component of income tax expense.
In assessing the realization of deferred tax assets, management considers whether it is more likely than not that some portion or all of the deferred assets will be realized. The ultimate realization of the deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Evaluating the need for a valuation allowance for deferred tax assets often requires judgment and analysis of all the positive and negative evidence available, including cumulative losses in recent years and projected future taxable income, to determine whether all or some portion of the deferred tax assets will not be realized. As of December 31, 2023, the Company has utilized a full valuation allowance to offset the net deferred tax assets as the Company believes it is not more likely than not that the net deferred tax assets will be fully realizable. The valuation allowance increased by $1.6 million during the year ended December 31, 2023.
DIFFUSION PHARMACEUTICALS INC.As of December 31, 2023, the Company had NOL carryforwards of approximately $38.9 million and $37.0 million for federal and state tax purposes, respectively. Federal NOL carryforwards will not expire and state NOL carryforwards will begin to expire in 2038, if not utilized. The TCJA enacted on December 22, 2017 limits a taxpayer’s ability to utilize NOL deduction in a year to 80% taxable income for federal net operating losses arising in tax years beginning after 2017, however, federal NOLs post 2017, are now indefinite lived.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
As of December 31, 2023, the Company also had federal and state research credit carryforwards of $0.6 million and $0.1 million, respectively. The federal and state research credits will begin to expire in 2038 and 2034, respectively.
Generally, utilization of the NOL carryforwards and credits may be subject to an annual limitation due to the ownership change limitations provided by Section 382 of the Code, which provides for limitations on NOL carryforwards and certain built-in losses following ownership changes, and Section 383 of the Code, which provides for special limitations on certain excess credits, as well as similar state provisions. Accordingly, the Company’s ability to utilize NOL carryforwards may be limited as the result of such an “ownership change.” A formal Section 382 study was performed through December 31, 2023 which resulted concluded there have been no historical section 382 ownership changes, thus the NOL carryforwards are not be subject to an annual limitation. With respect to Diffusion, the Company deems the historical Diffusion tax attributes (NOLs/Credits) are unusable due to the IRC Section 382 limitation. ASC 740-10-25 states that a “write off might be appropriate if there is only a remote likelihood that the entity will utilize the carryforward (i.e. NOL), it is acceptable for the entity to write off the deferred tax assets against the valuation allowance, thereby eliminating the need to disclose the gross amounts. As such, the Company has written off these attributes.
The Company had NOL carryforwards forfiles federal and state income tax purposes at December 31, 2020returns in jurisdictions with varying statutes of limitations. Due to its NOL carryforwards, the Company’s income tax returns generally remain subject to examination by federal and 2019state tax authorities. The Company is currently not subject to any income tax audits by federal or state taxing authorities. The statute of approximately:limitations for tax liabilities for all years remains open.
Combined NOL Carryforwards: | | December 31, 2020 | | | December 31, 2019 | |
Federal | | $ | 15,013,388 | | | $ | 5,844,972 | |
State | | | 15,007,966 | | | | 5,844,972 | |
The pre-2018 net operating loss carryforwards begin expiring in 2020Company uses the “more likely than not” criterion for both federal and staterecognizing the income tax purposes. In November 2019, as a resultbenefit of a change of ownership, under the provisions of Internal Revenue Code Section 382uncertain income tax positions and similar state provisions, the Company’s ability to utilize their net operating loss carryforwards to offset futureestablishing measurement criteria for income was limited.tax benefits. The Company recorded a valuation allowance against a portionhas evaluated the impact of their deferredthese positions and believes that its income tax assetsfiling positions and deductions will be sustained upon examination. Accordingly, no reserves for uncertain income tax positions or related accruals for interest and penalties have been recorded as of December 31, 2020 because2023 and 2022.
14. Subsequent Events
2024 Private Placement
On March 28, 2024, the Company entered into a securities purchase agreement with certain purchasers named therein related to the private placement of an aggregate of 2,532,285 units, each comprised of (i) (A) one share of common stock or (B) one Pre-Funded Warrant and (ii) one Series A Warrant. The 2024 Private Placement is expected to close on or about April 1, 2024, subject to customary closing conditions. The aggregate upfront gross proceeds from the 2024 Private Placement are expected to be approximately $50 million, before deducting offering fees and expenses, and additional gross proceeds of up to approximately $99.4 million may be received if the Series A Warrants are exercised in full for cash.
Pre-Funded Warrant Amendment and Exercise
On February 26, 2024, following the effectiveness of an amendment eliminating certain beneficial ownership limitations set forth therein, the Company’s previously outstanding pre-funded warrant was exercised in full by the holder thereof pursuant to the cashless exercise provision in Section 2(c) of the uncertaintypre-funded warrant. Upon exercise, 36 shares were withheld in lieu of their realization.
A reconciliationa cash payment of income tax benefit at the statutory federal income tax rateexercise price and income taxes as reflected in the consolidated financial statements is as follows:holder was issued 495,959 shares of common stock.
Rate reconciliation: | | December 31, 2020 | | | December 31, 2019 | |
Federal tax benefit at statutory rate | | | (21.0 | )% | | | (21.0 | )% |
State tax, net of Federal benefit | | | (4.7 | )% | | | (2.5 | )% |
Orphan drug credit | | | (2.9 | )% | | | 25.3 | % |
Change in valuation allowance | | | 18.1 | % | | | (7.5 | )% |
Stock compensation | | | — | % | | | 8.6 | % |
Other | | | — | % | | | 0.1 | % |
Total provision | | | (10.5 | )% | | | 3.0 | % |
The Company files income tax returns in the U.S. federal jurisdiction and various state jurisdictions. The Company’s 2017 to 2019 tax years remain open and subject to examination.
10. Related Party Transactions
The Company’s Director of Information Technologies is the son of the Chairman of the Board of Directors and he has held that position since December 2014.
11.Subsequent Events
2021 Warrant Exercises
From January 1, 2021 through March 15, 2021, warrants were exercised by multiple holders to purchase a total of 4,230,000 shares of the Company's common stock for aggregate gross proceeds of approximately $2.2 million.
2021 Common Stock Offering
In February 2021, the Company completed the February 2021 Offering in which it offered and sold 33,658,538 shares of its common stock in an underwritten, public offering for a purchase price to the public of $1.025 per share, inclusive of shares offered and sold pursuant to the exercise-in-full by the underwriter of its 30-day option to purchase additional shares. The February 2021 Offering resulted in aggregate net proceeds to the Company of $31.2 million, after deducting underwriting commissions, discounts, and expenses but prior to deducting other offering costs. In addition, at the closings of the February 2021 Offering, the Company issued to designees of the underwriter of the transaction warrants to purchase up to an aggregate of 1,682,927 shares of common stock to designees. The underwriter warrants have an exercise price of $1.28125 per share and a term of five years from the date of issuance.
ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
ITEM 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
The term “disclosureWe maintain disclosure controls and procedures” means our controlsprocedures (as defined in Rules 13a-15I and other procedures15d-15(e) promulgated under the Exchange Act) that are designed to ensureprovide reasonable assurance that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms. Disclosure controlsforms and procedures include, without limitation, controls and procedures designed to ensure that such information required to be disclosed by us in the reports that we file or submit under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and principal financial officer,officers, or persons performing similar functions, as appropriate to allow timely decisions regarding required disclosure.
We carried out an evaluation, under In designing and evaluating our disclosure controls and procedures, we recognize that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the supervisiondesired control objectives, and we are required to apply our judgment in evaluating the cost-benefit relationship of possible internal controls. Our management evaluated, with the participation of our management, including our principal executive officer and principal financial officer, the effectiveness of the effectivenessdesign and operation of our disclosure controls and procedures (as definedas of the end of the period covered in Exchange Act Rules 13a – 15(e) and 15d – 15(e)).this report. Based uponon that evaluation, our principal executive officer and principal financial officer concluded that as of the end of the period covered in this report, our disclosure controls and procedures were effectiveare ineffective due to ensure that information required to be disclosedthe material weaknesses noted below in reports filed under the Exchange Act is recorded, processed, summarized and reported within the required time periods and is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate to allow timely decisions regarding required disclosure.
Our principal executive officer and principal financial officer do not expect that our disclosure controls or internal controls will prevent all error and all fraud. Although our disclosure controls and procedures were designed to provide reasonable assurance of achieving their objectives, a control system, no matter how well conceived and operated, can provide only reasonable, not absolute assurance that the objectives of the system are met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within our company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of a simple error or mistake. Additionally, controls can be circumvented if there exists in an individual a desire to do so. There can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.subsequent paragraph.
Management’s Report onMaterial Weaknesses in Internal Control Overover Financial Reporting
Our management is responsibleIn connection with the audit of the Company’s consolidated financial statements for establishingthe years ended December 31, 2023 and maintaining adequate2022, material weaknesses in the Company’s internal control over financial reporting aswere identified in relation to: (i) the recording of significant complex transactions and (ii) the absence of effective controls regarding the accurate identification, evaluation and proper recording of various expense accounts.
A material weakness is a deficiency or combination of deficiencies in internal control over financial reporting such termthat there is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Under the supervision and with the participationa reasonable possibility that a material misstatement of our Chief Executive Officercondensed consolidated interim financial statements would not be prevented or detected on a timely basis. The identified material weaknesses, if not remediated, could result in a material misstatement to the Company’s consolidated financial statements that may not be prevented or detected. A material weaknesses will not be considered remediated until a remediation plan has been fully implemented, the applicable controls operate for a sufficient period of time, and Senior Vice President, Finance, our management conducted an evaluationit has been concluded, through testing, that the newly implemented and enhanced controls are operating effectively.
On August 16, 2023, we completed the Merger. For financial reporting purposes, EIP was determined to be the accounting acquirer and, accordingly, for all periods prior to the Merger, EIP’s historical financial statements and results of operations replace and are deemed to be the Company’s financial statement and results of operations for such periods. While Diffusion was previously subject to the provisions of SOX, EIP, as a private, non-reporting operating company prior to the Merger, was not. Accordingly, upon consummation of the effectivenessMerger, we began the process of integrating the pre-Merger business of EIP into Diffusion’s pre-established public company, internal control framework, including internal controls and information systems and we continue to implement measures designed to improve our internal control over financial reporting based onto remediate the framework in Internal Control—Integrated Framework (2013) issued by the Committee of Sponsoring Organizationsmaterial weaknesses. As of the Treadway Commission. Based on that evaluation, our management concluded that ourdate of this Annual Report, we continue to be actively engaged in these efforts through, among other things, adding additional review procedures by qualified personnel over complex accounting matters, and we currently expect to complete the remediation plan during the year ending December 31, 2024. However, the Company cannot predict the success of such efforts or the outcome of its assessment of the remediation efforts and the Company’s efforts may not remediate this material weakness in its internal control over financial reporting, was effective as of December 31, 2020.or additional material weaknesses may be identified in the future.
Notwithstanding the material weaknesses in internal control over financial reporting described above, our management has concluded that our consolidated financial statements included in this Annual Report are fairly stated in all material respects in accordance with U.S. GAAP.
Attestation Report of the Independent Registered Public Accounting Firm
This report does not include an attestation report of our independent registered public accounting firm regarding internal control over financial reporting. As a "smaller reporting company" (as such term is defined in Rule 12b-2 of the Exchange Act), pursuant to Section 989G of the Dodd-Frank Act, we are exempt from the requirement subjecting management’s report to attestation by our independent registered public accounting firm.
Change in Internal Control Over Financial Reporting
ThereExcept as set forth above, there was no change in our internal control over financial reporting that occurred during our fourth quarter ended December 31, 20202023 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. | OTHER INFORMATION |
None.Acting Principal Financial Officer
On March 28, 2024, J. William Tanner, Ph.D., our Chief Financial Officer, commenced a temporary leave of absence from the Company in order to undergo treatment for a medical condition. In connection with Dr. Tanner’s temporary leave of absence, the Board appointed William Elder to temporarily serve as Acting Principal Financial Officer of the Company, effective as of March 28, 2024. Mr. Elder, age 41, has served as our General Counsel and Corporate Secretary since September 2020 and will continue to serve in those roles. Mr. Elder also previously served as our Principal Financial Officer from June 2023 to August 2023.
In connection with such appointment, Mr. Elder did not receive any consideration nor were any modifications made to Mr. Elder’s existing employment agreement or outstanding equity awards, which are described under the heading, “Diffusion Executive and Director Compensation,” in the Registration Statement and the Company’s other filings with the SEC. Except as previously disclosed in the Registration Statement and the Company’s other filings with the SEC, (i) there are no arrangements or understandings between Mr. Elder and any other person pursuant to which Mr. Elder was selected as the Acting Principal Financial Officer, (ii) Mr. Elder does not have any direct or indirect material interest in any transaction requiring the disclosure of the information required by Item 404(a) of Regulation S-K, (iii) there is no material plan, contract or arrangement to which Mr. Elder is a party or in which he participates that was entered into, or any grant or award to Mr. Elder or modification thereto, under any such plan, contract or arrangement in connection with his appointment as Acting Principal Financial Officer, and (iv) there are also no family relationships between Mr. Elder and any director or executive officer of the Company.
Adoption, Termination or Modification of Trading Arrangements
During the three months ended December 31, 2023, none of our directors or officers (as defined in Rule 16a-1(f) of the Exchange Act), adopted, terminated or modified a Rule 10b5-1 trading arrangement or non-Rule 10b5-1 trading arrangement (as such terms are defined in Item 408 of Regulation S-K).
ITEM 9C. | DISCLOSURES REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS |
Not applicable.
PART III
ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
The additional information required by Item 10 of Form 10-K will be set forth in the Proxy Statement and is incorporated herein by reference.reference from our Proxy Statement, to be filed with the SEC within 120 days after December 31, 2023, the end of the fiscal year to which this Annual Report relates, including the information set forth under the captions, “Corporate Governance,” “Election of Directors,” “Executive Officers,” and “Certain Relationships and Related Party Transactions.”
ITEM 11. | EXECUTIVE COMPENSATION |
The additional information required by Item 11 of Form 10-K will be set forth in the Proxy Statement and is incorporated herein by reference.reference from our Proxy Statement, to be filed with the SEC within 120 days after December 31, 2023, the end of the fiscal year to which this Annual Report relates, including the information set forth under the captions, “Executive Compensation,” “Director Compensation,” “Compensation Committee Report,” and “Corporate Governance – Compensation Committee Interlocks and Insider Participation.”
ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The additional information required by Item 12 of Form 10-K will be set forth in the Proxy Statement and is incorporated herein by reference.reference from our Proxy Statement, to be filed with the SEC within 120 days after December 31, 2023, the end of the fiscal year to which this Annual Report relates, including the information set forth under the caption, “Security Ownership of Certain Beneficial Owners and Management.”
ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The additional information required by Item 13 of Form 10-K will be set forth in the Proxy Statement and is incorporated herein by reference.reference from our Proxy Statement, to be filed with the SEC within 120 days after December 31, 2023, the end of the fiscal year to which this Annual Report relates, including the information set forth under the captions, “Corporate Governance” and “Certain Relationships and Related Party Transactions.”
ITEM 14. | PRINCIPAL ACCOUNTINGACCOUNTANT FEES AND SERVICES |
The additional information required by Item 14 of Form 10-K will be set forth in the Proxy Statement and is incorporated herein by reference.reference from our Proxy Statement, to be filed with the SEC within 120 days after December 31, 2023, the end of the fiscal year to which this Annual Report relates, including the information set forth under the caption, “Ratification of Selection of Independent Registered Public Accounting Firm.”.
PART IV
ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
1.(a) Our financial statements are included in Part II, –Item 8. Financial Statements and Supplementary Data8 of this Annual Report.
2.(b) All financial statement schedules have been omitted from this Item 15 as the required information is not applicable, is not present in amounts sufficient to require submission of such schedules, or because the information required is included in our financial statements or the related notes included in Part II, –Item 8. Financial Statements and Supplementary Data8 of this Annual Report.
3.(c) The exhibits set forth in the following "Index to Exhibits" are filed with, furnished with, and/or incorporated herein by reference into this Annual Report, as described thereon.set forth therein. A copy of any of such exhibit will be furnished at a reasonable cost, upon receipt from any person of a written request for any such exhibit. Such request should be sent to Diffusion PharmaceuticalsCervoMed Inc., 1317 Carlton Avenue,20 Park Plaza, Suite 200, Charlottesville, Virginia 22902,424, Boston, Massachusetts 02116, Attention: General Counsel.
INDEX TO EXHIBITS
Exhibit
No.
| Description | | Method of Filing |
3.12.1Δ
| CertificateAgreement and Plan of IncorporationMerger, dated as of March 30, 2023, by and among Diffusion Pharmaceuticals Inc., as amendedEIP Pharma, Inc. and Dawn Merger Sub Inc.
| | Incorporated by reference to Exhibit 3.12.1 to the registrant’s annual reportCompany’s Current Report on Form 10-K for the year ended December 31, 20198-K filed on March 30, 2023. |
| | | | |
3.23.1
| BylawsCertificate of Diffusion PharmaceuticalsIncorporation of CervoMed Inc., as amended
| | Incorporated by reference to Exhibit 3.1 to the Company’s Annual Report on Form 10-K filed on March 24, 2023. |
| | | |
| 3.2 | Certificate of Amendment, dated August 16, 2023, to the Certificate of Incorporation, as amended, of CervoMed Inc. (Reverse Stock Split) | | Incorporated by reference to Exhibit 3.3 to the Company’s Current Report on Form 8-K filed on August 17, 2023. |
| | | |
| 3.3 | Certificate of Amendment, dated August 16, 2023, to the Certificate of Incorporation, as amended, of CervoMed Inc. (Name Change) | | Incorporated by reference to Exhibit 3.4 to the registrant’s annual reportCompany’s Current Report on Form 10-K for8-K filed on August 17, 2023. |
| | | |
3.4 | Bylaws of CervoMed Inc., as amended | | Incorporated by reference to Exhibit 3.5 to the year ended December 31, 2015Company’s Current Report on Form 8-K filed on August 17, 2023. |
| | | | |
4.1 | Form of 2017 Private PlacementMay 2019 Investor Warrant | | Incorporated by reference to Exhibit 4.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on March 15, 2017May 28, 2019. |
| | | | |
4.2 | Form of 2018 Common StockMay 2019 Placement Agent Warrant | | Incorporated by reference to Exhibit 4.14.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on January 19, 2018May 28, 2019. |
| | | | |
4.3 | Form of 2018 Underwriter'sNovember 2019 Series I Investor Warrant | | Incorporated by reference to Exhibit 4.24.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on January 22, 2018November 13, 2019. |
| | | | |
4.4 | Form of MayNovember 2019 Common StockSeries II Investor Warrant | | Incorporated by reference to Exhibit 4.14.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on May 28, 2019November 13, 2019. |
| | | | |
4.5 | Form of MayDecember 2019 Placement Agent’sInvestor Warrant | | Incorporated by reference to Exhibit 4.24.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on May 28, 2019December 13, 2019. |
| | | | |
4.6 | Form of NovemberDecember 2019 Series I Common StockPlacement Agent Warrant | | Incorporated by reference to Exhibit 4.14.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on NovemberDecember 13, 20192019. |
| | | | |
4.7 | Form of November 2019 Series II Common StockMay 2020 Placement Agent Warrant (In Respect of Exercise Transaction) | | Incorporated by reference to Exhibit 4.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on November 13, 2019May 8, 2020. |
| | | | |
4.8 | Form of November 2019 Pre-Funded Common StockMay 2020 Placement Agent Warrant (In Respect of Offering Transaction) | | Incorporated by reference to Exhibit 4.34.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on November 13, 2019May 20, 2020. |
| | | | |
4.9 | Form of November 2019 Placement Agent’sFebruary 2021 Underwriter Warrant | | Incorporated by reference to Exhibit 4.4 to the registrant’s current report on Form 8-K filed on November 13, 2019
|
| | | |
4.10
| Form of December 2019 Common Stock Warrant
| | Incorporated by reference to Exhibit 4.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on December 13, 2019February 18, 2021. |
4.10 | Form of 2023 Pre-Funded Investor Warrant | | Incorporated by reference to Exhibit 4.3 to the Company’s Current Report on Form 8-K filed on August 17, 2023. |
| | | | |
4.11 | Form of December 2019 Placement Agent’sEIP 2018 Investor Warrant | | Incorporated by reference to Exhibit 4.24.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on December 13, 2019August 17, 2023. |
| | | | |
4.12 | Form of May 2020 Common StockEIP 2018 Investor Warrant (AI EIPP Holdings LLC) | | Incorporated by reference to Exhibit 4.14.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on May 8, 2020August 17, 2023. |
| | | | |
4.13 | Form of May 2020Series A Warrant expected to be issued in connection with the 2024 Private Placement Agent's Warrant (In Respect of Exercise Transaction) | | Incorporated by reference to Exhibit 4.2 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on May 8, 2020 March 28, 2024 |
| | | | |
4.14 | Form of May 2020Pre-Funded Warrant expected to be issued in connection with the 2024 Private Placement Agent's Warrant (In Respect of Offering Transaction) | | Incorporated by reference to Exhibit 4.34.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on May 20, 2020 March 28, 2024 |
| | | | |
4.15 | Form of February 2021 Underwriter's WarrantSpecimen Stock Certificate
| | Incorporated by reference to Exhibit 4.1 to the registrant’s current report on Form 8-K filed on February 18, 2021Filed herewith.
|
| | | | |
4.16 | Description of Securities of CervoMed Inc. | | Incorporated by reference to Exhibit 4.12 to the registrant's annual report on 10-K for the year ended December 31, 2019Filed herewith.
|
| | | | |
10.110.1#
| Amended & Restated Employment Agreement, dated as of September 8, 2020February 1, 2024, by and between John Alam, M.D. and CervoMed Inc. | | Incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed on February 2, 2024. |
| | | |
10.2# | Amended & Restated Employment Agreement, dated as of February 1, 2024, by and between Robert J. Cobuzzi, Jr., Ph.D. and Diffusion PharmaceuticalsCervoMed Inc.* | | Incorporated by reference to Exhibit 10.2 to the registrant’s current report on Form 8-K filed on September 9, 2020 |
10.2
| Amended and Restated Employment Agreement, dated as of September 21, 2018, by and between William Karl Hornung and Diffusion Pharmaceuticals Inc. *
| | Incorporated by reference to Exhibit 10.2 to the registrant'sCompany’s Current Report on Form 8-K filed September 27, 2018on February 2, 2024.
|
| | | | |
10.310.3#
| Employment Agreement, dated as of October 19, 2020,November 15, 2023, by and between Christopher D. Galloway, M.D.J. William Tanner, Ph.D. and Diffusion PharmaceuticalsCervoMed Inc. * | | Incorporated by reference to Exhibit 10.1 to the registrant'sCompany’s Current Report on Form 8-K filed October 20, 2020on November 17, 2023. |
| | | | |
10.410.4#
| Employment Agreement, dated as of February 1, 2024, by and between Kelly Blackburn and CervoMed Inc. | | Incorporated by reference to Exhibit 10.3 to the Company’s Current Report on Form 8-K filed on February 2, 2024. |
| | | |
10.5# | Employment Agreement, dated as of September 23, 2020, by and between William Elder and Diffusion PharmaceuticalsCervoMed Inc. * | | Incorporated by reference to Exhibit 10.1 to the registrant'sCompany’s Current Report on Form 8-K filed September 25, 20202020. |
| | | | |
10.510.6#
| Amendment to Employment Agreement, dated as of September 6, 2016,March 29, 2023, by and between David G. KalergisCervoMed Inc. and Diffusion Pharmaceuticals Inc.*William Elder | | Incorporated by reference to Exhibit 10.110.5 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on September 8, 2016March 30, 2023. |
| | | | |
10.610.7#
| Separation Agreement dated as of September 8, 2020 by and between David G. Kalergis and Diffusion PharmaceuticalsCervoMed Inc.* 2015 Equity Incentive Plan
| | Incorporated by reference to Exhibit 10.1Appendix C to the registrant’s current reportCompany’s definitive proxy statement on Form 8-KSchedule 14A filed on September 9, 2020June 10, 2016. |
10.8# | Amendment No. 1 to CervoMed Inc. 2015 Equity Incentive Plan | | Incorporated by reference to Appendix B to the Company’s definitive proxy statement on Schedule 14A filed on June 10, 2016. |
| | | | |
10.710.9#
| Employment Agreement, dated as of October 18, 2016, byEIP Pharma, Inc. 2018 Employee, Director and between John L. Gainer and Diffusion Pharmaceuticals Inc.*Consultant Equity Incentive Plan
| | Incorporated by reference to Exhibit 10.1810.31 to the registrant’s annual reportCompany’s Registration Statement on Form 10-K/S-4/A filed on April 28, 2017July 12, 2023. |
| | | | |
10.810.10#
| SeparationForm of Stock Option Award Agreement dated as of March 12, 2020, by and between John L. Gainer and Diffusion Pharmaceuticals Inc.*under 2015 Equity Incentive Plan
| | Incorporated by reference to Exhibit 10.110.7 to the registrant’s current reportCompany’s Annual Report on Form 8-K filed on March 18, 202010-K for the year ended December 31, 2021. |
| | | | |
10.910.11#
| ConsultingForm of Stock Option Award Agreement dated as of March 12, 2020, byunder 2018 Employee, Director and between John L. Gainer and Diffusion Pharmaceuticals Inc.*Consultant Equity Incentive Plan
| | Incorporated by reference to Exhibit 10.2 to the registrant’s current report on Form 8-K filed on March 18, 2020Filed herewith.
|
| | | | |
10.110.12#
| Diffusion PharmaceuticalsForm of Indemnification Agreement between CervoMed Inc. 2015 Equity Incentive Plan**and each of its directors and officers
| | Incorporated by reference to Appendix C to the registrants definitive proxy statement on Schedule 14A filed on June 10, 2016Filed herewith.
|
| | | | |
10.1110.13*
| Amendment No. 1 to DiffusionOption and License Agreement, dated as of August 27, 2012, by and between EIP Pharma LLC and Vertex Pharmaceuticals Inc. 2015 Equity Incentive PlanIncorporated
| | Incorporated by reference to Appendix BExhibit 10.23 to the registrants definitive proxy statementCompany’s Registration Statement on Schedule 14AForm S-4/A filed on June 10, 2016July 12, 2023. |
| | | | |
10.1210.14*
| FormAmendment No.1, dated as of StockApril 8, 2014, to Option Award Agreement*
| | Incorporatedand License Agreement, dated August 27, 2012, by reference to Exhibit 10.5 to the registrant’s annual report on Form 10-K for the year ended December 31, 2017 |
| | | |
10.13
| Form of Diffusionand between EIP Pharma LLC and Vertex Pharmaceuticals LLC Stock Option Award Agreement*Incorporated
| | Incorporated by reference to Exhibit 10.24 to the registrant’s annual reportCompany’s Registration Statement on Form 10-K for the year ended December 31, 2015S-4/A filed on July 12, 2023. |
| | | | |
10.1410.15*
| FormAmendment No.2, dated as of IndemnificationNovember 17, 2015, to Option and License Agreement, dated August 27, 2012, as amended April 18, 2014, by and between DiffusionEIP Pharma LLC and Vertex Pharmaceuticals Inc. and each of its Directors and Officers*Incorporated
| | Incorporated by reference to Exhibit 10.310.25 to the registrant’s annual reportCompany’s Registration Statement on Form 10-K for the year ended December 31, 2015 |
| | | |
10.15
| Form of Securities Purchase Agreement, dated as of May 18, 2020, by and among Diffusion Pharmaceuticals Inc. and the purchasers party thereto
| | Incorporated by reference to Exhibit 10.1 to the registrant’s current report on Form 8-KS-4/A filed on May 20, 2020July 12, 2023.
|
| | | | |
10.16 | LeaseAt-The-Market Sales Agreement, dated March 31, 2017,as of July 22, 2022, by and between Diffusion PharmaceuticalsCervoMed Inc. and One CarltonBTIG, LLC
| | Incorporated by reference to Exhibit 10.11.1 to the registrant’s current reportCompany’s Current Report on Form 8-K filed on March 15, 2017July 22, 2022. |
| | | | |
21.110.17
| SubsidiariesForm of Diffusion Pharmaceuticals Inc.EIP Subscription Agreement, dated as of July 10, 2023
| | Filed herewithIncorporated by reference to Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q filed on November 13, 2023.
|
| | | | |
23.110.18
| ConsentForm of KPMG LLP, independent registered public accounting firmLock-up Agreement, dated as of March 30, 2023, related to Merger Agreement
| | Filed herewithIncorporated by reference to Exhibit 10.2 to the Company’s Current Report on Form 8-K filed on March 30, 2023.
|
| | | | |
10.19 | Securities Purchase Agreement, dated March 28, 2024, by and between CervoMed Inc. and each of the purchasers party thereto, related to the 2024 Private Placement | | Incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed on March 28, 2024. |
*104
| Cover Page Interactive Data File (embedded within the Inline XBRL document and contained in Exhibit 101). | | |
# | Indicates a management contract or compensatory plan or arrangement. |
Δ | Schedules and exhibits have been omitted from this filing pursuant to Item 601(a)(5) of Regulation S-K. The Company agrees to furnish on a supplemental basis a copy of any omitted schedule or exhibit to the SEC upon its request; provided, however, that the Company may request confidential treatment pursuant to Rule 24b-2 of the Exchange Act for any schedule or exhibit so furnished. |
* | Portions of this exhibit (indicated by asterisks) have been omitted in accordance with the rules of the SEC. |
ITEM 16. | FORM 10-K SUMMARY |
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Dated: March 16, 202129, 2024 | DIFFUSION PHARMACEUTICALSCERVOMED INC.
| |
| | | |
| By: | /s/ Robert J. Cobuzzi, Jr., Ph.D.John Alam, M.D. | John Alam, M.D. |
| | Robert J. Cobuzzi, Jr., Ph.D.
| |
| | President, Chief Executive Officer and Director | |
| | (Principal Executive Officer) | |
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Name and Signature | | Title | | Date |
| | | | |
/s/ Robert J. Cobuzzi, Jr., Ph.D.John Alam, M.D. | | President, Chief Executive Officer and Director | | March 16, 202129, 2024 |
Robert J. Cobuzzi, Jr., Ph.D.John Alam, M.D.
| | (Principal Executive Officer) | | |
| | | | | |
/s/ William K. HornungElder | | Chief Financial OfficerGeneral Counsel and Corporate Secretary
| | March 16, 2021 29, 2024 |
William K. HornungElder | | (Acting Principal Financial and Accounting Officer) | | |
| | | | | |
/s/ David G. KalergisJoshua S. Boger, Ph.D. | | ChairmanChair of the Board
| | March 16, 2021 29, 2024 |
David G. KalergisJoshua S. Boger, Ph.D.
| | | | |
| | | | | |
/s/ Robert AdamsJ. Cobuzzi, Ph.D. | | Chief Operating Officer and Director | | March 29, 2024 |
Robert J. Cobuzzi, Ph.D. | | | | |
| | | | |
/s/ Sylvie Grégoire, PharmD. | | Director | | March 16, 2021 29, 2024 |
Robert AdamsSylvie Grégoire, PharmD.
| | | | |
| | | | | |
/s/ Jane H. Hollingsworth, J.D. | | Director | | March 16, 2021 29, 2024 |
Jane H. Hollingsworth, J.D. | | | | |
| | | | | |
/s/ John L. Gainer, Ph.D.Jeff Poulton | | Director | | March 16, 2021 29, 2024 |
John L. Gainer, Ph.D.Jeff Poulton
| | | | |
| | | | | |
/s/ Mark T. GilesMarwan Sabbagh, M.D. | | Director | | March 16, 2021 29, 2024 |
Mark T. GilesMarwan Sabbagh, M.D.
| | | | |
| | | | | |
/s/ Alan LevinFrank Zavrl | | Director | | March 16, 2021 29, 2024 |
Alan LevinFrank Zavrl
| | | | | |