UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 10-K
|
| | | | |
| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2019
OR
|
| | | | |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from
____ to ____
Commission File Number: 001-37798
Selecta Biosciences, Inc.Inc.
(Exact name of registrant as specified in its charter)
|
| | | | | | | | | | |
| Delaware | 26-1622110 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
| 65 Grove Street, Watertown, MA | 02472 | |
| 480 Arsenal Way | Watertown | MA | 02472 | |
| (Address of principal executive offices) | (Zip Code) | |
(617)
(617) 923-1400
Registrant’s telephone number, including area code
|
| | | | | | | |
| Securities registered pursuant to Section 12(b) of the Act: |
| | |
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Common Stock, $0.0001 par value per share | SELB | The Nasdaq GlobalStock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No ý
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes o No ý
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports); and (2) has been subject to such filing requirements for the past 90 days. Yes ý☒ No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files).
Yes ý☒ No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and "emerging“emerging growth company"company” in Rule 12b-2 of the Exchange Act.
|
| | | | | | | | | | | | | | | | | | | |
| Large accelerated filer | ☐ | | Accelerated filer | ☐ | |
| Non-accelerated filer | ☒ | |
| Non-accelerated filer | ☐ | | Smaller reporting company | ☒ | |
| | | | Emerging growth company | ☒☐ | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to section 13(a) of the Exchange Act. ☒☐
Indicate by check mark whether the registrant has filed a report on and attested to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ý☒
The aggregate market value of the voting and non-voting common equitystock held by non-affiliates of the registrant on June 28, 2019, based on the closing price of $1.79 per share of the registrant'sregistrant’s common stock as reported byon the Nasdaq GlobalStock Market on June 30, 2021, the last business day of the registrant’s most recently completed second quarter, was approximately $58.5 million.$366,839,496.
As of March 6, 20204, 2022 the registrant had 86,420,195124,288,850 shares of common stock, par value $0.0001 per share, outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive Proxy Statement relating to its 20202022 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission are incorporated by reference into Part III of this Annual Report on Form 10-K.
| | | | | | | | |
| TABLE OF CONTENTS |
| | |
| | |
TABLE OF CONTENTS |
| | |
| | |
| | |
| | |
| | |
| | |
| Properties | | |
| | |
| | |
| | |
| | |
| | |
| | |
| Selected Consolidated Financial Data | | |
| | |
| | |
| | |
| | |
| Item 9A. | Controls and Procedures | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| Signatures | |
| | |
| | |
| | |
| | |
FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K, or the Annual Report, contains forward-looking statements. We intend such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act of 1933, as amended, (the “Securities Act”),or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, (the “Exchange Act”).or the Exchange Act. All statements other than statements of historical facts contained in this Annual Report, including statements regarding our future results of operations and financial position, business strategy, prospective products, product approvals, research and development costs, timing and likelihood of success, the plans and objectives of management for future operations and future results of anticipated products,the impact of the COVID-19 pandemic on our business and operations and our future financial results, and the period over which we estimate our existing cash and cash equivalents will be sufficient to fund our future operating expenses and capital expenditure requirements are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements.
In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential”, or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this Annual Report are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this Annual Report and are subject to a number of important factors that could cause actual results to differ materially from those in the forward-looking statements, including the factors described under the sections in this Annual Report titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” as well as the following:
| |
- | our status as a development-stage company and our expectation to incur losses in the future; |
| |
- | our ability to continue as a going concern, our future capital needs and our need to raise additional funds; |
| |
- | our ability to build a pipeline of product candidates and develop and commercialize drugs; |
| |
- | our unproven approach to therapeutic intervention; |
| |
- | our ability to enroll patients in clinical trials, timely and successfully complete those trials and receive necessary regulatory approvals; |
| |
- | our ability to have continued access to manufacturing facilities and to receive or manufacture sufficient quantities of our product candidates; |
| |
- | our ability to maintain our existing or future collaborations or licenses, including our ability to reach an agreement regarding an acceptable amendment of our exclusive patent license agreement with the Massachusetts Institute of Technology; |
| |
- | our ability to protect and enforce our intellectual property rights; |
| |
- | federal, state, and foreign regulatory requirements, including FDA regulation of our product candidates; |
| |
- | our ability to obtain and retain key executives and attract and retain qualified personnel; |
| |
- | developments relating to our competitors and our industry, including the impact of government regulation; and |
| |
- | our ability to successfully manage our growth. |
•our status as a development-stage company and our expectation to incur losses in the future;
•our future capital needs and our need to raise additional funds;
•our ability to build a pipeline of product candidates and develop and commercialize such pipeline;
•our unproven approach to therapeutic intervention;
•our ability to enroll patients in clinical trials, timely and successfully complete those trials and receive necessary regulatory approvals;
•our ability to access manufacturing facilities and to receive or manufacture sufficient quantities of our product candidates;
•our ability to maintain our existing or future collaborations or licenses;
•the continuing impact of the COVID-19 pandemic on our operations, the continuity of our business, including our preclinical studies and clinical trials, and general economic conditions;
•our ability to protect and enforce our intellectual property rights;
•federal, state, and foreign regulatory requirements, including FDA regulation of our product candidates;
•our ability to obtain and retain key executives and attract and retain qualified personnel;
•developments relating to our competitors and our industry, including the impact of government regulation; and
•our ability to successfully manage our growth.
Moreover, we operate in an evolving environment. New risk factorsrisks and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties.
You should read this Annual Report and the documents that we reference in this Annual Report completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
PART I
Item 1. Business
OVERVIEWOverview
We are a clinical-stage biopharmaceutical company using our ImmTOR™company. Our ImmTOR® platform encapsulates rapamycin, also known as sirolimus, an FDA approved immunomodulator, in biodegradable nanoparticles ImmTOR is designed to induce antigen-specific immune tolerance.
By combining ImmTOR with antigens of interest, our precision immune tolerance platform has the potential to novel biologic therapies that would otherwise be limited by immunogenicity withrestore self-tolerance to auto-antigens in autoimmune diseases, amplify the goalefficacy of effectivelybiologics (including gene therapies) and safely treating rare and serious diseases. Many such diseases are treated with biologic therapies that are foreign tomitigate the patient’s immune system and therefore elicit an undesired immune response. Of particular concern areformation of anti-drug antibodies, or ADAs, which are often produced by the immune system in response to biologic therapy and can adversely affect the efficacy and safety of treatment. Our proprietary tolerogenic ImmTOR platform encapsulates an immunomodulator in biodegradable nanoparticles and is designed to mitigate the formation of ADAs by inducing antigen-specific immune tolerance toagainst biologic drugs. ADAs can start developing in the body with the first dose of a biologic therapy and can render subsequent doses ineffective or unsafe, potentially depriving patients of life-saving therapeutic options and limiting the likelihood of success for many otherwise promising novel biologic drugs and technologies.
We continually seek to enhance ImmTOR. In recent preclinical studies we have conducted, we have observed that ImmTOR may have synergistic activity with interleukin-2, or IL-2, molecules that have been engineered to be selective for regulatory T cells, or Tregs. Treg-selective IL-2 mutant molecules, or IL-2 muteins, have been shown to transiently expand all pre-existing Tregs in preclinical studies conducted by others. We have observed in preclinical studies that the combination of ImmTOR, a Treg-selective IL-2 mutein and an antigen elicited an approximately three-fold increase in antigen-specific Tregs beyond ImmTOR alone with evidence of enhanced durability of immune tolerance and the potential for ImmTOR dose sparing. This combination of ImmTOR with a Treg selective IL-2 molecule represents an evolution of the ImmTOR platform, which we call ImmTOR-IL™. We believe this combination has the potential to be a best-in-class therapy in diseases where expansion of total Tregs may prove beneficial.
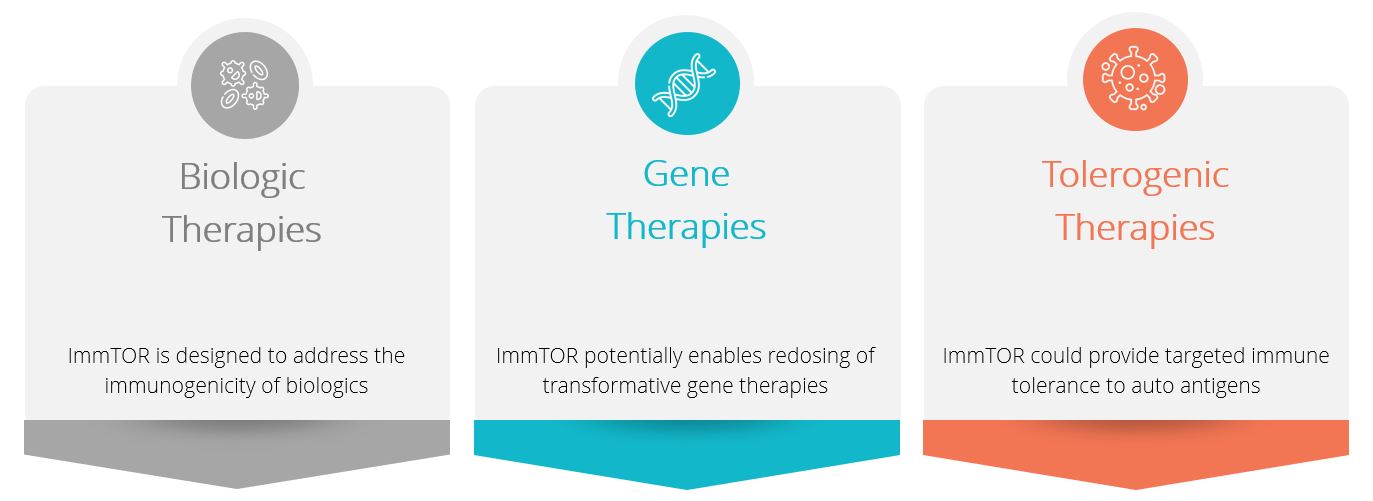
We believe ImmTOR hasand ImmTOR-IL have the potential to enhance both the efficacy without compromising theand safety of existing approved biologic drugs,therapies (including gene therapies), improve product candidates under development, and enable novel therapeutic modalities such as re-administration of systemic gene therapy. We intendin autoimmune disease. In clinical trials, ImmTOR has been observed to build a product pipeline by combining our ImmTOR platform with a range of biologics. Additionally, we plan to seek collaborations and license agreements to broaden our applications for our ImmTOR platform.
Our lead product candidate, SEL-212, is a proprietary combination of a therapeutic uricase enzyme (pegadricase) and our ImmTOR platform that is designed to durably control serum uric acid, or SUA, levels in patients with chronic refractory gout through monthly administration. Our preclinical data indicated that ImmTOR, when co-administered with pegadricase, induced antigen-specific immune tolerance to pegadricase and substantially reducedinhibit the formation of associated ADAs. Our Phase 1neutralizing antibodies to adeno-associated virus (AAV) capsids, potentially enabling re-dosing of gene therapies. Additionally, based on preclinical data showedin AAV gene therapies, we believe that ImmTOR mitigated the formation of ADAs against pegadricase after a single dose of SEL-212. Clinical data from our Phase 2 dose-finding study suggested the potential of monthly doses of SEL-212 to durably control serum uric acid levels by mitigating the formation of anti-pegadricase antibodies. Initial tissue imaging studies by dual energy computerized tomography (DECT) scans also provided evidence regarding the potential of SEL-212 to eliminate tissue deposits of uric acid crystals. This data also showed a low rate of gout flares. Based on our Phase 2 clinical data and feedback received during our end-of-Phase 2 meeting from the U.S. Food and Drug Administration, or FDA, we commenced enrollment in our Phase 2 head-to-head (COMPARE) clinical trial of SEL-212, utilizing revised stopping rules, compared to the current FDA-approved uricase therapy, KRYSTEXXA® (pegloticase) in March 2019. We initiated dosing in May 2019. In December 2019, we completed enrollment in this study. In the first quarter of 2020, we received feedback from the FDA on our planned Phase 3 program for SEL-212, which will consist of two placebo-controlled trials, each with a six month primary endpoint, one of which will have a six-month placebo-controlled extension. We plan to commence the Phase 3 clinical program in SEL-212 in the second half of 2020. We will require additional resources to complete the planned Phase 3 clinical program for SEL-212. We expect our clinical and, if approved, marketing strategy for SEL-212 to initially focus on the estimated 160,000 patients in the United States with chronic refractory gout, and to focus on those patients that are being treated by rheumatologists.
We are also applying our ImmTOR platform in an effort to mitigate the formation of ADAs when dosing gene therapy. Most gene therapies use a viral vector, such as an adeno-associated virus, or AAV, vector to place corrective genetic material into cells to treat genetic diseases. One of the key hurdles for the gene therapy field is to overcome immunogenicity against the viral vector. ADAs form in response to the first administration of a gene therapy vector and prevent effective subsequent doses of gene therapy. The ability to provide repeat doses may be particularly important for pediatric patients to receive continued treatment benefit later in life as gene expression wanes due to cellular turnover as patients grow. The ability to re-administer gene therapies also could provide the potential for dose titration and could be advantageous for diseases where the goal is to transfect a high number of cells.
Our investigational gene therapy program targets the treatment of rare genetic diseases, including methylmalonic acidemia, or MMA. We are collaborating with Asklepios BioPharmaceutical, Inc., or AskBio to develop a treatment for MMA, which, if successfully developed and approved, could havehas the potential to preventimprove efficacy and safety by increasing transgene expression, reducing hepatic inflammation and inhibiting undesired immune responses to both the vectorAAV capsid and the transgene product that can occur with the first dose of gene therapy by usingtherapy. In biologic therapies, clinical activity of ImmTOR in humans has been observed with pegadricase, a highly immunogenic pegylated uricase enzyme being developed for the treatment of patients with chronic refractory gout to conventional therapy. The combination of ImmTOR and pegadricase is currently being evaluated in a Phase 3 clinical trial that we are conducting on behalf of our partner Swedish Orphan Biovitrum AB, or Sobi. We intend to pursue development of therapies for autoimmune diseases where expansion of either all Tregs or antigen-specific Tregs has been shown to, or we believe is, likely to have a beneficial effect. We believe that ImmTOR and ImmTOR-IL have the potential to unlock antigen-specific therapies for autoimmune diseases and that ImmTOR-IL can further improve the efficacy and safety profile of biologic therapies beyond ImmTOR alone.
We have developed a portfolio of wholly owned and partnered candidates across gene therapies, biologic therapies and tolerogenic therapies for autoimmune diseases that leverage our ImmTOR platform. We plan to enter the clinic under this collaboration in 2020. For more information, see "Licenses and Collaborations - AskBio” below.
In additioncontinue to developing this non-immunogenic therapeutic product candidate, we intend to continue expanding ourdevelop proprietary immune tolerance pipelinecompounds and pursue collaboration-driven development in certain disease areas, which could include strategic collaborations, out-licensing, opportunities for select applicationsand in-licensing transactions.
Our ImmTOR Platform
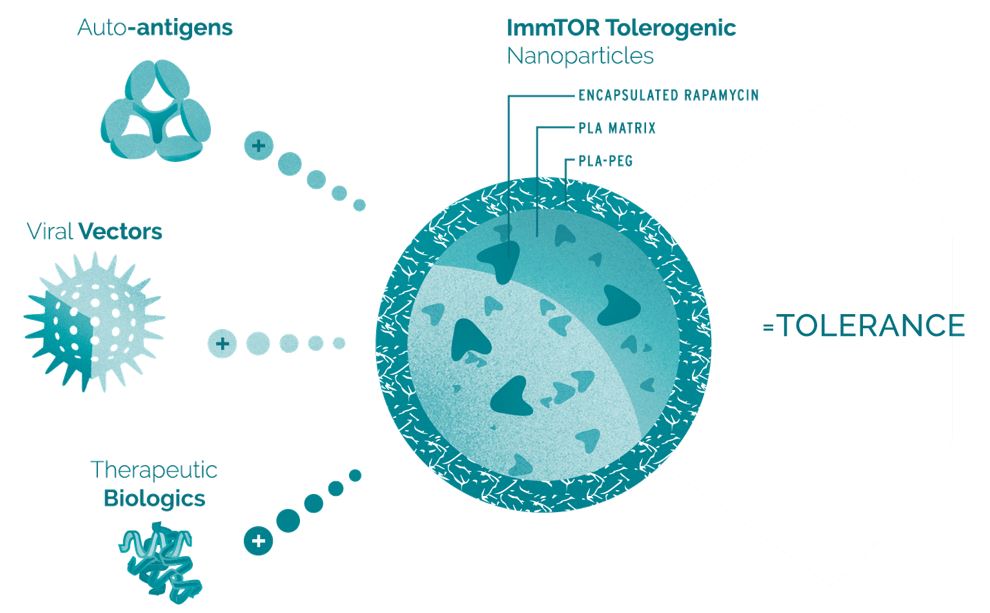
ImmTOR consists of ourbiodegradable nanoparticles encapsulating the immunomodulator rapamycin. Rapamycin is the active ingredient of Rapamune, an immunosuppressant that has been used extensively in humans and is currently FDA-approved as a prophylaxis of organ rejection in kidney transplant patients aged 13 or older. The rapamycin component of ImmTOR platform. In December 2016, we entered intois embedded in a license agreement, or the Spark License Agreement, with Spark Therapeutics, Inc.matrix of synthetic polymers called poly(D,L-lactide), or Spark, that provides Spark with exclusive worldwide rights to our ImmTOR platform to research, developPLA, and commercialize gene therapies for Factor VIII, an essential blood clotting protein relevant to the treatment of hemophilia A. For more information,
see “Licenses and Collaborations - Spark Therapeutics" below. Additionally, in December 2019 we entered into a license agreement,poly(D,L-lactide)-block-poly(ethylene-glycol), or the AskBio License Agreement, with AskBio, that provides AskBio with exclusive worldwide rights to our ImmTOR platform to research, develop and commercialize certain adeno-associated virus, or AAV gene therapy products targeting the glucosidase alpha, acid, or GAA gene, or derivatives thereof, to treat Pompe Disease. For more information, see "Licenses and Collaborations - AskBio” below.
In September 2018, we announced a collaboration with the European consortium, CureCN, for an ImmTOR+AAV gene therapy combination product candidate in Crigler-Najjar syndrome. We expect the CureCN consortium to obtain scientific advice from the German drug regulatory authority in 2020.
OUR STRATEGY
Our goalPLA-PEG. PLA is to become the first biopharmaceutical company to develop and commercialize targeted therapies that are designed to more effectively and safely treat rare and serious diseases by inducing immune tolerance to potent but immunogenic biologic therapies. In addition, we intend to maximize the value of our ImmTOR platform by collaborating with or licensing our ImmTOR platform out to biopharmaceutical companies on programs that can benefit from our technology but that are outside our area of focus. The key elements of our strategy include the following.
| |
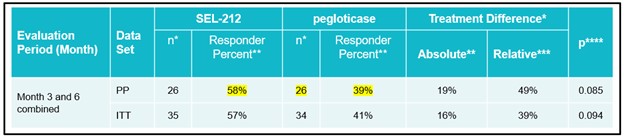
• | Advance the development of SEL-212 for the treatment of chronic refractory gout. We believe SEL-212 has the potential, if successfully developed and approved, to become the first monthly biologic treatment that durably controls uric acid while also dissolving harmful deposits of uric acid crystals for patients with chronic refractory gout. We have completed Phase 1 and Phase 2 dose-finding clinical programs, comprised of two Phase 1 clinical studies and an open-label, multiple ascending dose Phase 2 clinical program. We completed enrollment of our Phase 2 head-to-head (COMPARE) clinical study against KRYSTEXXA in December 2019 and expect to report top-line data in the third quarter of 2020. The two-armed, open label trial has enrolled approximately 150 patients, randomized 1:1, with one arm receiving KRYSTEXXA (as set forth in the prescribing information) and the other arm receiving six monthly doses of SEL-212. The primary endpoint in the study is the percentage of patients in each arm that maintain SUA control below 6.0 mg/dL, for at least 80% of the time during months three and six. In the first quarter of 2020, we received feedback from the FDA on our planned Phase 3 clinical program for SEL-212, which will consist of two placebo-controlled trials, each with a six month primary endpoint, one of which will have a six-month placebo-controlled extension. We plan to commence the Phase 3 clinical program in SEL-212 in the second half of 2020. We will require additional resources to complete the planned Phase 3 clinical program for SEL-212.
|
| |
• | Transform gene therapy by enabling repeated administration of systemically-administered AAV gene therapy vectors. One of the key challenges for the gene therapy field is the inability to administer more than one dose of AAV systemically due to the formation of neutralizing anti-AAV antibodies. We believe redosing has the potential to enable dose titration of gene therapy to achieve therapeutic expression of the transgene, to restore therapeutic expression in pediatric patients that have lost expression of the transgene due to target organ growth. A 2018 publication in Nature Communications provided peer-reviewed preclinical data supporting ImmTOR’s potential to mitigate the formation of neutralizing anti-AAV antibodies and enable productive AAV vector redosing in mice and nonhuman primates. In August 2019, we entered into a feasibility study and license agreement with AskBio, or the AskBio Collaboration Agreement, pursuant to which we and AskBio will conduct proof of concept studies to potentially validate the use of our ImmTOR platform in conjunction with an AAV gene therapy to mitigate the formation of neutralizing anti-AAV capsid antibodies, which currently precludes redosing.
|
| |
• | Leverage ImmTOR to develop additional novel uses and classes of non-immunogenic biologics. We intend to use our ImmTOR platform to develop a range of proprietary, non-immunogenic biologic therapeutic candidates. In each case, ImmTOR would be evaluated for its potential to mitigate undesired immune response to the biologic therapy with the goal of improving efficacy and/or safety. Our strategy is to develop proprietary treatments for rare and serious diseases by combining ImmTOR with biologics that have been in-licensed or provided through research collaborations.
|
| |
• | Establish infrastructure and capabilities to develop and commercialize our products in rare and orphan diseases. While we believe our ImmTOR platform may be broadly applicable across disease areas, we intend to focus our proprietary efforts on developing and commercializing proprietary ImmTOR-enabled products for rare and serious diseases where there is high unmet medical need. Therapies for treating these diseases require focused commercial efforts and coordination with patient groups and investigators.
|
| |
• | Selectively pursue licensing opportunities and maximize the value of our ImmTOR programs for immune tolerance. In addition to our own product development efforts and those on which we are collaborating, we are in discussions with potential licensees to pursue novel therapies that would utilize our ImmTOR platform. For example,
|
in December 2016, we entered into the Spark License Agreement to develop gene therapies for certain targets utilizing our ImmTOR platform. Additionally, in December 2019, we entered into the AskBio License Agreement to develop a gene therapy product for the treatment of Pompe Disease utilizing our ImmTOR platform.
OVERVIEW OF THE HUMAN IMMUNE SYSTEM
The human immune system is an integrated system of specialized immune cells, cell products and tissues that protect against infectious disease and cancer. The immune system recognizes antigens, which are substances, such as proteins, enzymes or complex sugars. These antigens can be endogenous, or self-antigens, which are produced by the body, or exogenous antigens derived from foreign sources, such as viruses, fungi or bacteria. The human immune system has evolved to recognize and destroy potentially harmful substances. To function effectively, the immune system must discern between harmful antigens and innocuous antigens. The immune system maintains a delicate balance between effector cells, which mount immune responses to antigens that represent potential threats, and regulatory cells, which mitigate undesired and potentially harmful immune responses through immune tolerance. Depending upon the characteristicspart of the antigenbroader poly(lactic-co-glycolic acid), or PLGA, family of biodegradable polymers that have more than 30 years of commercial use and the contextare formulation components in which the antigen is encountered, the immune system must determine whether to mount a defensive (effector) or regulatory (tolerogenic) immune response.
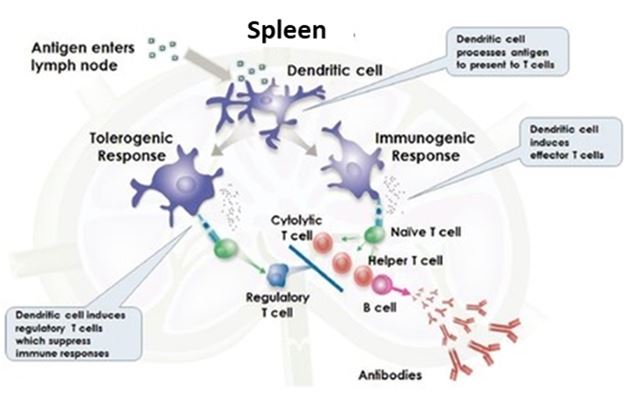
Antigens are processed in lymphoid organs, such as lymph nodes and the spleen, where the immune system determines whether to mount a defensive or regulatory response through a process called “antigen presentation.” In connection with antigen presentation, dendritic cells process the antigens and present them to T cells. When presented, antigens perceived as harmful induce a stimulatory response that can result in the activation of cytolytic T cells or helper T cells, the latter of which help to induce B cells to produce antibodies. The role of cytolytic T cells is to kill cells that harbor intracellular antigens, such as viruses. The role of antibodies is to neutralize or eliminate extracellular antigens on cell surfaces or in interstitial fluids, such as plasma. Figure 1 below depicts both antigen presentation and the related immune responses.
Figure 1. Antigen Presentation and Related Tolerogenic or Stimulatory Immune Response
There are a number of adverse outcomes that can occur when the immune system mounts an undesired response to an innocuous foreign antigenapproved products. Polyethylene glycol, or PEG, has been widely studied in clinical trials and is also a self-antigen. For example, food allergy occurs when the immune system mounts an immune response to innocuous food antigens. Another example of undesired immunogenicity occurs when the immune system is exposed to aformulation component in many approved biologic treatment, recognizes it as a foreign antigen and instructs the body to mount a defense by forming ADAs to the biologic, which can compromise a therapy’s desired beneficial effect. Undesired immunogenicity is common with biologic therapies, such as in enzyme and protein replacement therapies, and in novel technologies, such as gene therapy and antibody-drug conjugates.
OUR ImmTOR PLATFORM
Our ImmTOR platform is based in part on the pioneering research performed by our co-founders at Harvard University, Massachusetts Institute of Technology, or MIT, and Brigham and Women’s Hospital, or Brigham. In connection with our company’s founding, we licensed multiple patent families related to aspects of our ImmTOR platform pursuant to an agreement
with MIT. We believe one of the key insights from this research is that nanoparticles are uniquely suited to deliver precise instructions to the immune system as a result of the natural predisposition of the immune system to interrogate nanoparticles, such as viruses. We have aggressively sought to extend and protect the proprietary intellectual property underlying the composition and use of ImmTOR for antigen-specific immunotolerance. For more information, see "Intellectual Property" below.
We are currently developing our ImmTOR platform for:
combination with a uricase enzyme for the treatment of chronic refractory gout;
combination with gene therapies for the treatment of rare diseases;
potential application with other marketed products, product candidates in development and novel biologic drugs that would otherwise be too immunogenic to developproducts.
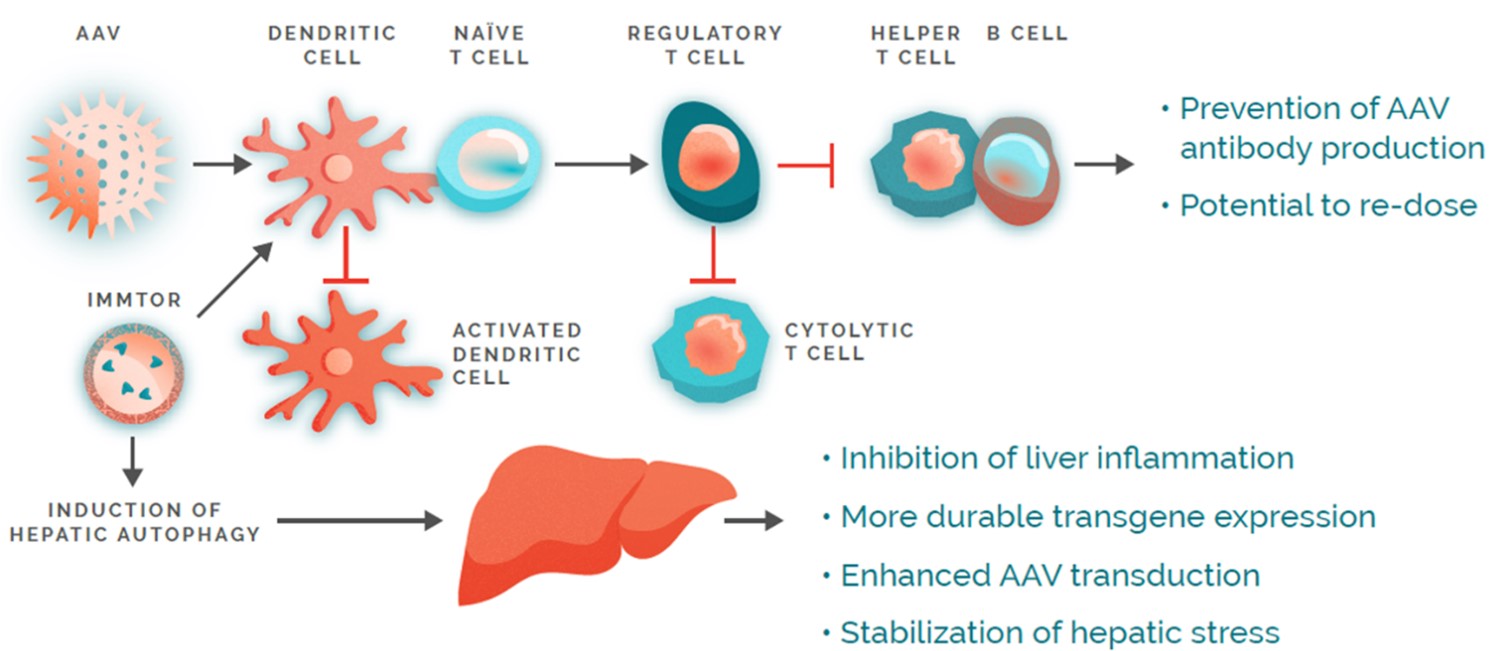
Our nanoparticles are designed to remain intact after injection into the body and accumulate selectivelypredominantly in lymphoid organs, which include lymph nodes, the spleen, and the spleen,liver, where the immune response isresponses are coordinated. The nanoparticles are designed to be processed by specialized immune cells, such as dendritic cells and other antigen-presenting cells, that initiate and regulate immune responses, where they deliver the immunomodulator in a coordinated and targeted manner. Depending on the type of immunomodulator encapsulated in the nanoparticles, our technology can be usedresponses. ImmTOR is intended to induce a tolerogenic response to mitigatephenotype in these antigen-presenting cells, which then process and present co-administered antigens in a manner that results in the formationinduction of ADAs against a biologic drug or to treat allergies and autoimmune diseases.
OUR ANTIGEN-SPECIFIC IMMUNE TOLERANCE PROGRAM
Our antigen-specific tolerance programs utilize ImmTOR, our biodegradable nanoparticle encapsulating the immunomodulator rapamycin. Rapamycin is a small molecule approved for the prevention of organ rejection in kidney transplant patients.regulatory T cells. To mitigate the formation of ADAs and induceunwanted immune responses by inducing precision immune tolerance in the body, we co-administeradminister our ImmTOR with a freethe desired antigen, such as an auto-antigen in the case of an autoimmune disease, a viral vector in the case of our gene therapy program, or a therapeutic enzyme, as depicted in the figure below.
In the case of a biologic drug, which is depicted in Figure 2 below.
Figure 2. Co-Administration of ImmTOR with a Biologic Drug
ImmTOR is designed to be co-administered (prior to theadministered in conjunction with such biologic drug) at the beginning of therapy with a biologic drugdrugs to mitigate the formation of ADAs without requiring the alteration of the drug or its dose regimen. As a result, we believe our ImmTOR may provide us with significant opportunities in the areasto co-administer ImmTOR with a variety of immune tolerancebiologic drugs, including therapeutic enzymes and ADA prevention because ImmTOR is designed to be co-administered at the beginning of therapy with many different biologic drugs. Importantly, wegene therapies. We believe each pairing of ImmTOR with a biologic drug also offers us the opportunity to pursue a distinct proprietary product candidate,candidates, which we believe hashave the potential to be separately patented, approved and marketed. ImmTOR is manufactured in facilities subject to current good manufacturing practice, or cGMP, requirements using well-defined commercial operations, which, we believe, further enhances the scalability of our tolerance programs.
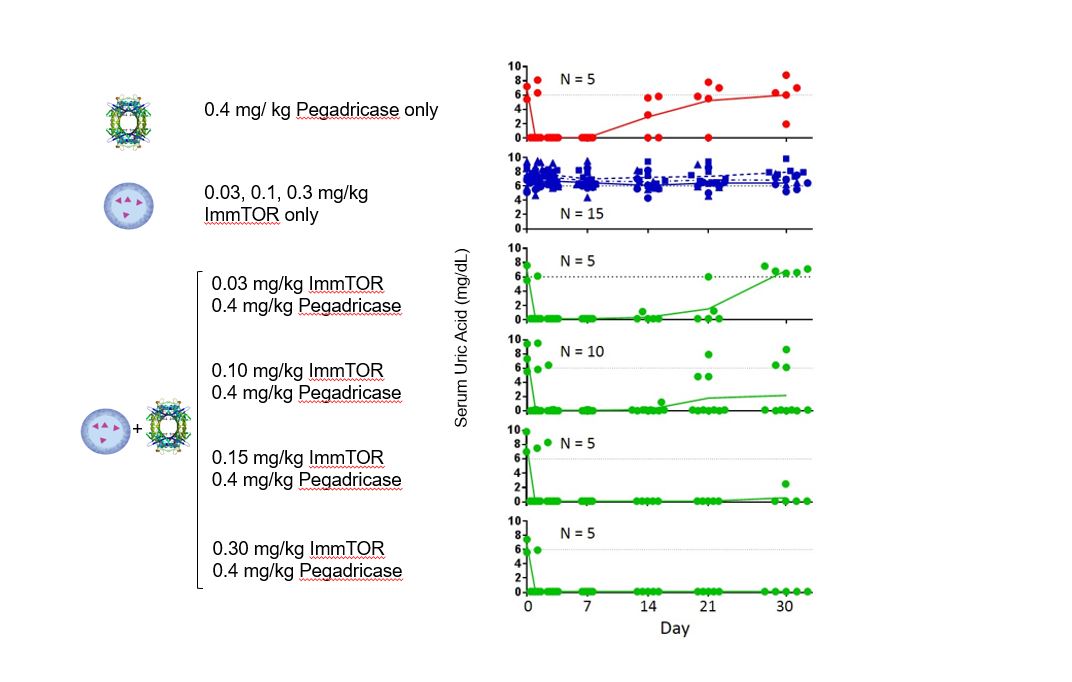
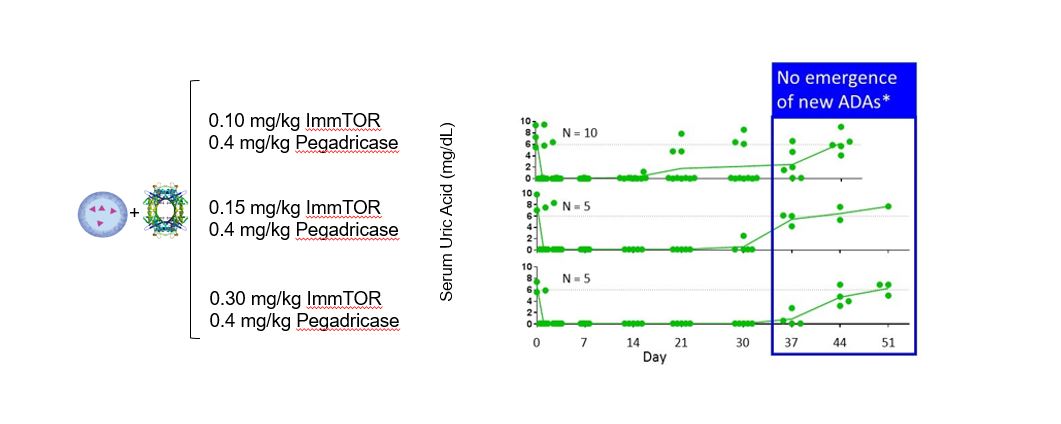
DuringIn preclinical studies, we observed that delivering an antigen together with ImmTOR provided the appropriate signals in vivo to induce regulatory T cells, which, in turn, inhibited effector immune responses, such as the formation of ADAs. In ouradditional preclinical studies, we observed that ImmTOR labeled with a fluorescent dye selectively accumulated in lymphoid organsthe spleen and the liver following intravenous dosing, where it was processed by antigen-presenting cells, such as dendritic cells. Figure 3The figure below depicts a model of how we believe ImmTOR would be taken up by a dendritic cell in the spleen. We believe that when delivered in the context of our ImmTOR, both the biologic drug and ImmTOR are taken up and processed by dendritic cells and other antigen presenting cells in a manner that may induceresult in the activation of antigen-specific regulatory T cells, which can potentially block the activation of helper T cells, thus mitigating the formation of ADAs.
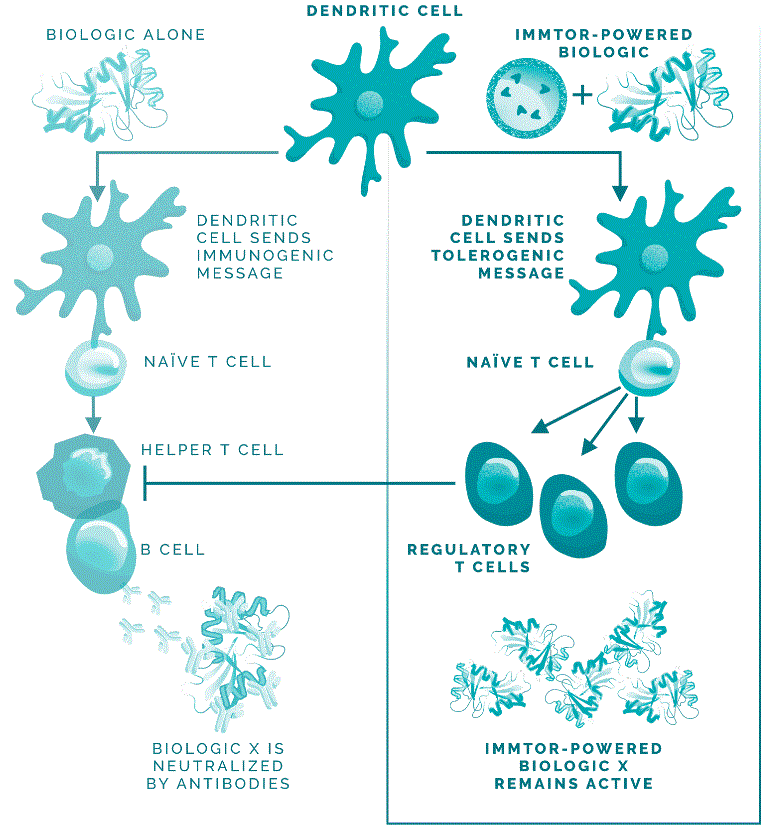
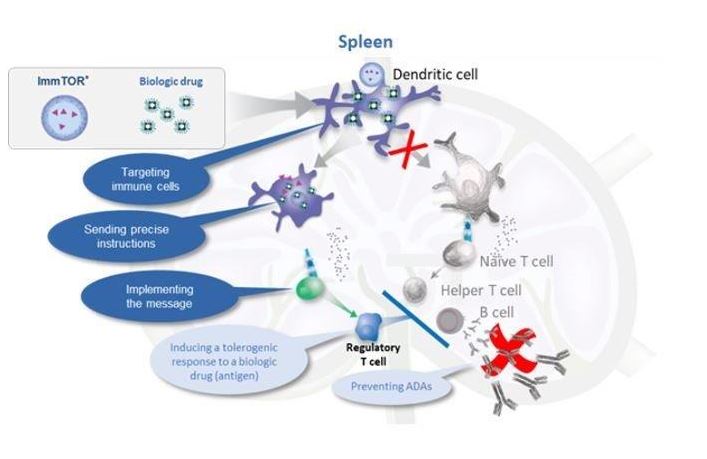
Figure 3.We look for ways to enhance our ImmTOR platform. Recent preclinical data generated by our scientific teams suggest that ImmTOR may have profound synergistic activity with engineered IL-2 molecules that are selective for Tregs. The IL-2 pathway influences critical aspects of both immune stimulation and immune regulation. Tregs express a high affinity form of the IL-2 receptor. Low doses of IL-2 have been shown by others to selectively activate Tregs resulting in expansion of pre-existing Tregs. Clinical trials of low dose IL-2 have generated evidence of efficacy in autoimmune diseases, such as systemic lupus erythematosus. Other investigators have shown that IL-2 can be engineered to selectively bind to the high affinity IL-2 receptor and expand pre-existing Tregs.
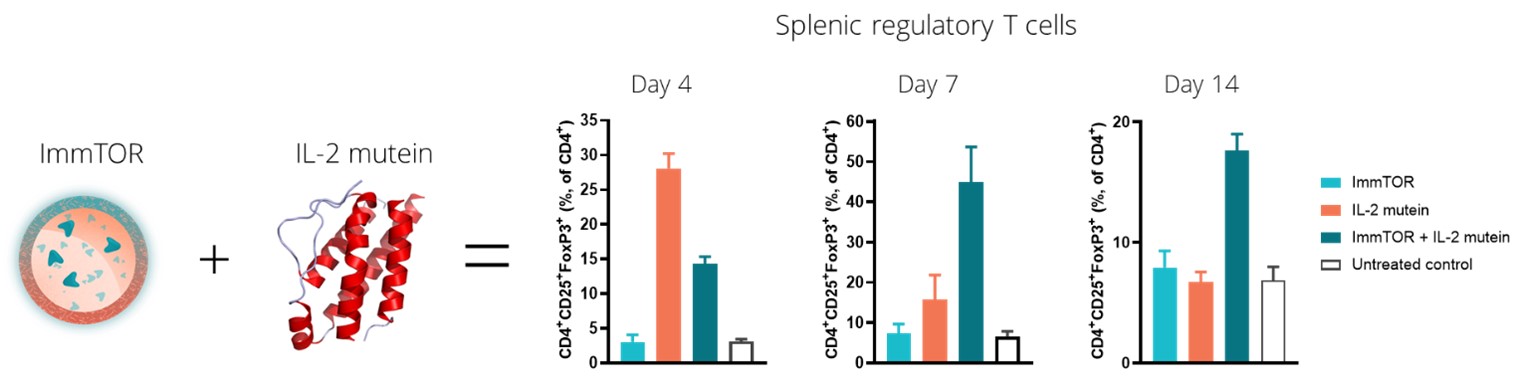
In our preclinical studies, we observed that ImmTOR combined with a Treg-selective IL-2 mutant protein, or IL-2 mutein, exhibited substantial synergistic activity in increasing the percentage and durability of total Treg expansion in the spleen. We believe that this combination has the potential to be a best-in-class therapy in diseases where expansion of total Treg may prove beneficial. The tables below show the synergistic expansion of total Treg we observed with treatment with ImmTOR and Antigen/Biologic Drug Presentationa Treg-selective IL-2 mutein. In the study, seven C57BL/6 mice per group were untreated, treated with ImmTOR alone, treated with IL-2 mutein alone or treated with a combination of ImmTOR and Related Immune Tolerance InductionIL-2 mutein. Expansion of CD4+, CD25+ and FoxP3+ T regulatory cells in the spleen was assessed at four, seven and 14 days after treatment.
Limitations
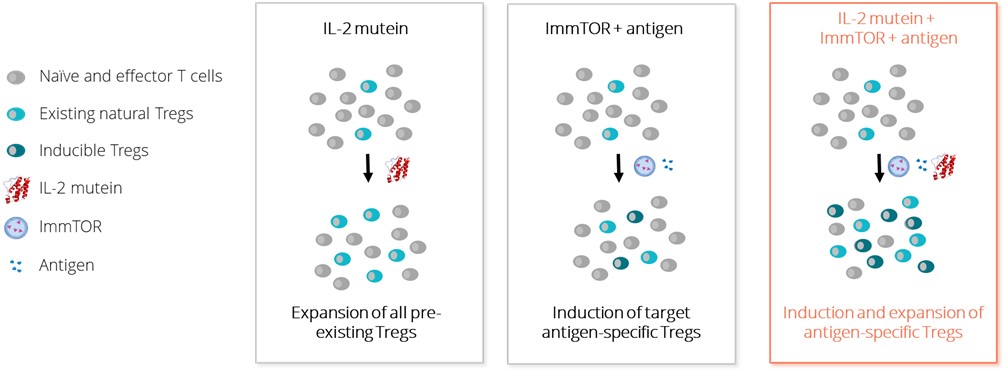
We believe that the power of existing therapies
All biologics, even those comprisedImmTOR is the ability to induce antigen-specific Treg to co-administered antigens. The ability to induce antigen-specific Treg may be beneficial in autoimmune diseases where the auto-antigens are well characterized. We believe the combination of human protein sequences, havea Treg-selective IL-2 with ImmTOR and an antigen has the potential to induce ADAs. Whetherand/or expand antigen specific Treg. The image below illustrates the possibility that the combination of a biologic drug elicitsTreg-selective IL-2 with ImmTOR plus an ADAantigen could give rise to the induction and/or expansion of an antigen-specific Treg.
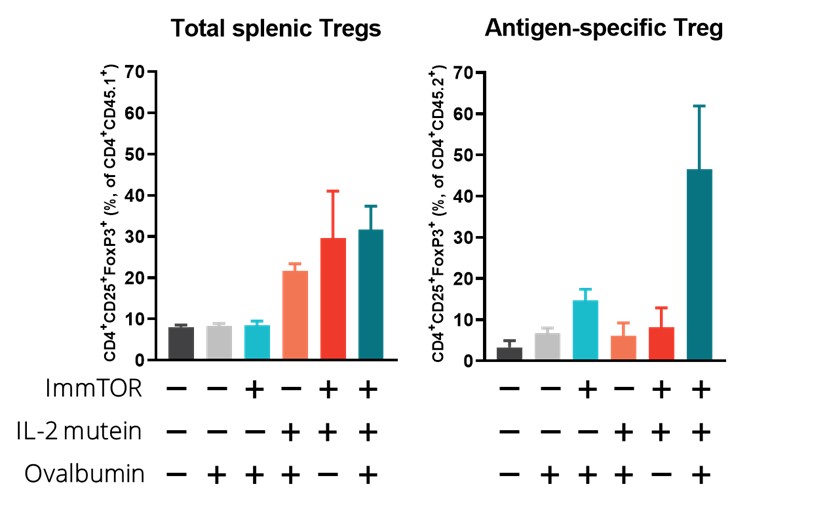
In a preclinical study, we evaluated the potential of a Treg-selective IL-2 to further expand antigen-specific Treg when combined with ImmTOR and an antigen. In this preclinical study, transgenic T cells expressing a T cell receptor specific for the antigen ovalbumin were adoptively transferred into wildtype mice. The next day, the mice were treated, left untreated or treated with ovalbumin or various combinations of ovalbumin with ImmTOR and/or IL-2 mutein. When total Treg were evaluated, we observed no increase an expansion of total Treg by IL-2 mutein + ovalbumin with a further increase in animals treated with ovalbumin + IL-2 mutein + ImmTOR. As we had expected, ImmTOR + ovalbumin alone did not increase total Treg. However, when ovalbumin-specific Treg were evaluated, we observed a significant increase in antigen-specific Treg in animals treated with the combination of ovalbumin + ImmTOR + IL-2 mutein. Animals treated with only ImmTOR + ovalbumin also were observed to have an increase in ovalbumin-specific Tregs but at levels that were approximately three-fold lower than those in the former group. The following graphs show expansion of antigen-specific Treg in mice treated with ImmTOR plus IL-2 mutein plus ovalbumin antigen.
Immune homeostasis is a dynamic process balancing immune stimulatory and immune tolerizing signals. This balance is thought to be mediated in part by the ratio of antigen-specific Treg to antigen-specific effector T cells. The expansion of antigen-specific Tregs has the potential to better withstand potent immune stimulatory signals and provide better durability of immune tolerance. In a preclinical study, we evaluated the ability of ImmTOR and IL-2 mutein to mitigate the immunogenicity to a co-administered AAV vector. Mice were injected with a dose of 2.7E12 vg/kg AAV8 on Day 0 and 5.0E12 vg/kg on Day 56 with or without IL-2 mutein and/or ImmTOR on the same days. Mice treated with AAV alone showed a robust antibody response dependsto the AAV capsid, as we had expected. Co-treatment with IL-2 mutein on both product-specific factors, such as propensityDays 0 and 56 showed a modest attenuation of the antibody response. Co-treatment with ImmTOR on Days 0 and 56 showed a dose-responsive effect on the antibody response, with a therapeutic dose of 200 µg ImmTOR providing inhibition of antibodies against AAV through at least Day 75, or 19 days after the second dose. At Day 91, some animals showed breakthrough of the antibody response. ImmTOR doses of 50 and 100 µg alone were sub-optimal. However, when ImmTOR was combined with IL-2 mutein, we observed inhibition of antibody formation through Day 117, 61 days after the second dose and the last time point measured. Importantly full inhibition of anti-AAV immunoglobulin G, or IgG, antibodies was observed even when IL-2 mutein was combined with 50 and 100 µg doses of ImmTOR. We believe these results suggest that the addition of a Treg-selective IL-2 to form aggregates,ImmTOR has the routepotential to increase the durability of administrationtolerance allow for dose-sparing of ImmTOR and mechanismthus, potentially enabling chronic dosing of action, as well as patient-specific factors, such as genetics, underlying disease and medications. For instance, many enzyme and protein replacement therapies usedImmTOR-IL in the treatment of rareautoimmune disease. The following graphs illustrate the combination of ImmTOR and serious diseases have a particularly high rate of immunogenicity because patients are genetically deficient in the target protein and, as a result, the therapeutic protein can be recognized as foreign by the patient’s immune system.
The formation of ADAs can lead to neutralization of efficacy, modification of pharmacokinetics and pharmacodynamics as well as allergic responses. Immunogenicity remains a significant hurdle for the development of safe and effective biologic treatments and has become a key concern for regulators, as evidenced by over 100 approved biologics that describe immunogenicity in their labels or clinical literature. According to product package inserts, approximately 50 currently marketed biologics report an incidence of ADAs affecting over 20% of patients. Among these biologics are Factor VIII products, such as Advate® (antihemophilic factor) for the treatment of hemophilia A and antibody therapeutics with fully human sequences, such as Humira® (adalimumab). We believe that immunogenicity also is a leading cause of product development failure for biopharmaceutical companies and that this issue is hampering the development of novel platform technologies, such as gene therapy and gene editing.
Undesired immunogenicity represents a significant hurdle that can hamper the clinical development of new biologic platforms. For example, in gene therapy, viral vectors are required to transport the genetic material into cells. The viral origin of these vectors explains their immunogenicity, which has led drug developers to limit applications to situations where the required frequency and site of administration are conducive to manageable immune responses.
Treatment and product development failure resulting from undesired immunogenicity has been recognized by regulators and patient advocacy organizations. In June 2014, the FDA and the National Organization for Rare Diseases, or NORD, co-sponsored a workshop on undesired immune responses to enzyme replacement therapies and called on the biopharmaceutical industry to take a more proactive approach to addressing immunogenicity to biologics.
Currently, we believe there are no comprehensive solutions to the complications of immunogenicity. Drug developers often stop the development of biologics that show an undesired immune response during preclinical and/or clinical development. In some cases, biopharmaceutical companies may attempt to reduce undesired immune responses by re-engineering the biologic through protein pegylation, removal or modification of immunogenic epitopes, or other mechanisms. However, these approaches are limited in their effectiveness. Physicians may try to address the issue of undesired immune responses by increasing the dose of the biologic, which can be prohibitively expensive and may present greater safety concerns, or in life-threatening situations, by using global immunosuppressive combination therapies. We believe that our tolerogenic ImmTOR platform could offer an
entirely new and effective treatment alternative for undesired immune responses, including with respect toIL-2 mutein mitigated the formation of ADAs, but potentially also for autoimmune diseases and allergies.anti-AAV antibodies.
OUR ImmTOR PROGRAMS DESIGNED TO INDUCE ANTIGEN-SPECIFIC IMMUNE TOLERANCE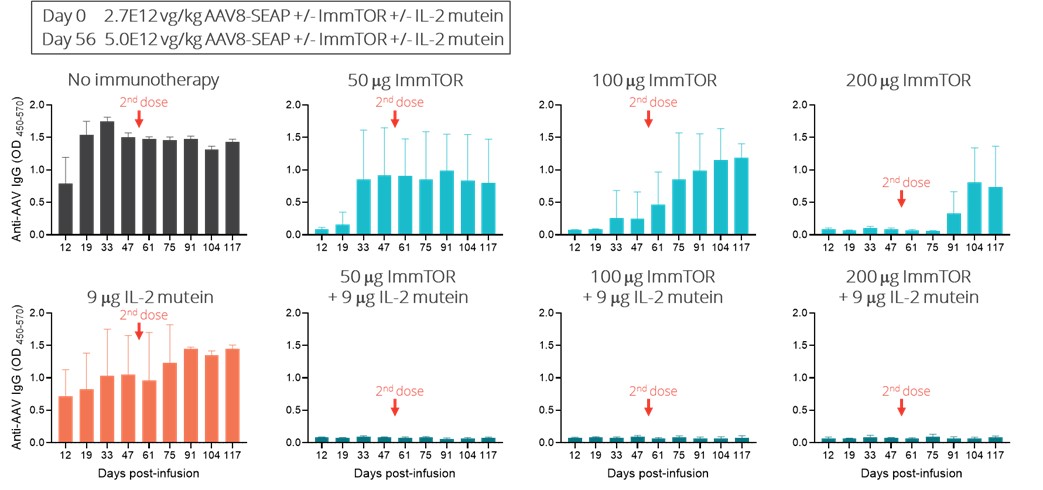
We believe our
Product Candidates
Our ImmTOR platform designed to induce antigen-specific immune tolerance has a broad range of potential applications. We are currently pursuing targetedOur product development strategies for multiple applicationsstrategy is built on the following three distinct pillars.
Biologic therapies. Biologic therapies are a class of biologic drugs frequently used to treat rare diseases. Through our analysis of biologic drugs, including in whichour preclinical studies, we believe ImmTOR products could be highly differentiated.
| |
• | Therapeutic enzymes. Therapeutic enzymes are a frequently used class of biologic drugshave observed that enzymes foreign to treat rare diseases. Through our analysis of biologic drugs, including our preclinical studies, we have observed that enzymes are especially prone to undesired immune responses. Our lead product candidate, SEL-212, includes pegadricase, a pegylated uricase enzyme, which is an example of an immunogenic enzyme for which we are applying ImmTOR with the intention of improving the enzyme’s efficacy and safety. We intend to seek opportunities to secure supply of and, if appropriate, licenses to these or other enzymes that we would evaluate in combination with ImmTOR.
|
| |
• | Gene therapies. We believe gene therapies have the potential to address key unmet medical needs for many rare genetic diseases, but undesired immune responses to the viral vectors used for gene replacement, augmentation and editing may be restricting their broader use. Through our analysis of genetic diseases, we have identified applications and patient segments that we believe would benefit from our ImmTOR platform. We intend to develop ImmTOR -enabled non-immunogenic gene therapy candidates which are designed to be utilized with AAV vectors (e.g., AAV8, AAV5). We believe our product candidates have the potential to mitigate the problem of pre-existing immunogenicity to the gene therapy vector and to prevent undesired immune responses to the vector and transgene that can occur with the first dose of gene therapy by using our ImmTOR platform. Our initial area of focus is on genetic metabolic diseases, but may also include lysosomal storage diseases and genetic muscular diseases. We believe we are the first company to systematically pursue the development of gene therapy product candidates with the goal of enabling repeat administration. We have engaged third parties with experience in gene therapy and rare diseases to support the development of our proprietary products. We also have licensed our ImmTOR platform to Spark Therapeutics and AskBio for the development of gene therapies for certain pre-specified targets. For more information, see "Licenses and Collaborations - Spark Therapeutics" and “Licenses and Collaborations - AskBio.”
|
| |
• | Other products and product candidates affected by undesired immune responses. We have generated preclinical data which we believe suggests a broad potential benefit of ImmTOR for immune tolerance. For many biologic drugs, undesired immune responses limit efficacy and cause safety concerns. We intend to strategically out-license ImmTOR platform for use with other products that are outside our focus to larger biopharmaceutical companies. We believe our ImmTOR platform may also be of interest to biopharmaceutical companies with novel biologic development concepts or product candidates in clinical development that have demonstrated initial efficacy but are experiencing issues with safety or sustained efficacy due to inhibitory ADAs.
|
SEL-212 for the treatmenthuman body, such as enzymes derived from microbes or replacement enzymes in the case of chronic refractory gout
Overview
SEL-212 is our proprietarypatients that are deficient in the specific enzyme, are especially prone to causing undesired immune responses. Our partnered product candidate, underSEL-212, which is currently in Phase 3 clinical development, for the treatment of chronic refractory gout. SEL-212 consists of ImmTOR co-administered with pegadricase, a pegylated uricase.uricase enzyme of fungal origin. This is an example of an immunogenic enzyme that we are combining with ImmTOR with the intention of improving the enzyme’s efficacy and safety. We believe that our SEL-212ImmTOR has the potential to enable and expand the use of enzymes derived from microbial sources, such as bacterial immunoglobulin A, or IgA, protease for the treatment of IgA nephropathy and bacterial IgG protease, or Xork, for the treatment of IgG-mediated autoimmune disease flares.
Additionally, we believe advances in protein engineering may lead to innovative therapeutic enzymes with improved pharmaceutical properties or entirely novel specificity and/or activity, but which may be recognized as foreign by the immune system. We are partnering with Ginkgo Bioworks, or Ginkgo, to design novel enzymes and proteins with transformative therapeutic potential which can be paired with ImmTOR to advance treatments for orphan and rare diseases. We intend to seek, if successfully developedappropriate, licenses to other enzymes to evaluate in combination with ImmTOR.
Gene therapies. We believe gene therapies have the potential to address key unmet medical needs for many rare genetic diseases, but undesired immune responses to the viral vectors used for gene replacement, augmentation and approved,editing may be restricting their broader use. AAV immunogenicity and AAV toxicity represent two major challenges for the gene therapy field; in many cases these two issues are inextricably linked. Immunogenicity of AAV vectors is thought to offer a uniquely effective treatment for patients with chronic refractory gout, while also providing evidence regarding the utility of our ImmTOR platform platform. Pegylated uricase, in the formcause or exacerbate many of the approved drug KRYSTEXXA,adverse events associated with AAV gene therapy. Induction of acute inflammation and capsid-specific CD8 T cells by AAV gene therapy is thought to contribute to observations of hepatotoxicity, which has demonstratedbeen associated with loss of transgene expression. The formation of neutralizing antibodies against AAV after initial treatment with AAV mediated gene therapies effectively prevents the possibility of re-dosing in patients who may benefit from additional doses due to either the failure to achieve therapeutic benefit or loss of transgene expression over time.
We have observed that ImmTOR, when used in combination with AAV gene therapy vectors, inhibited the immune response to the viral vector and enabled successful re-dosing in both mice and non-human primates. Currently, the ability to significantly reduce uric acidre-administer systemic AAV gene therapy is limited by the development of neutralizing antibodies. The ability to safely re-dose AAV may help achieve therapeutic benefit in patients who are under-dosed; it may also help restore transgene expression in patients, particularly pediatric patients, who may lose expression over time as they grow. In a study conducted in non-human primates, or NHP, we observed that co-administration of AAV vector and ImmTOR resulted in higher and more durable transgene expression after the first dose of gene therapy as well as robust inhibition of anti-AAV8 immunoglobulin G, or IgG and neutralizing antibodies. We believe the observation that co-administration of AAV vector and ImmTOR can lead to higher transgene expression illustrates the potential for dosing lower levels of AAV gene therapies when combined with ImmTOR. Integrating ImmTOR into a gene therapy protocol has the potential to provide a first dose benefit by enhancing liver-directed transgene expression and dissolvedurability, as well as the harmful uric acid crystalspotential to enable re-dosing to restore or augment transgene expression.
We have observed in preclinical studies that ImmTOR may also have hepatoprotective effects in mouse models of inflammation. As hepatotoxicity is associated with higher vector doses, one potential strategy that we are pursuing is to use ImmTOR to enable multiple lower doses of AAV vectors to mitigate toxicity risks associated with high vector
doses. We are concurrently working with our partner Ginkgo to develop a proprietary AAV capsid with improved transduction for liver-directed gene therapy. The below illustration depicts the manifestationspotential benefits of gout upon initial treatmentImmTOR in patients. However, we believe broad commercial adoption has not been achieved primarily duesystemic AAV gene therapy.
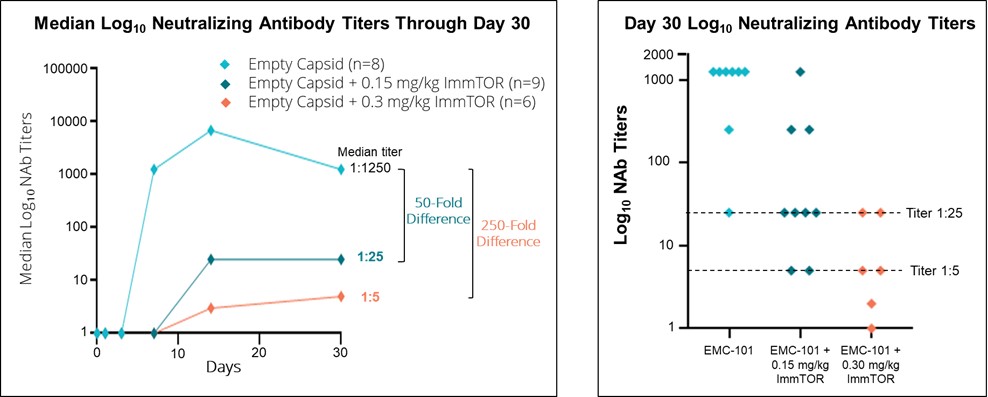
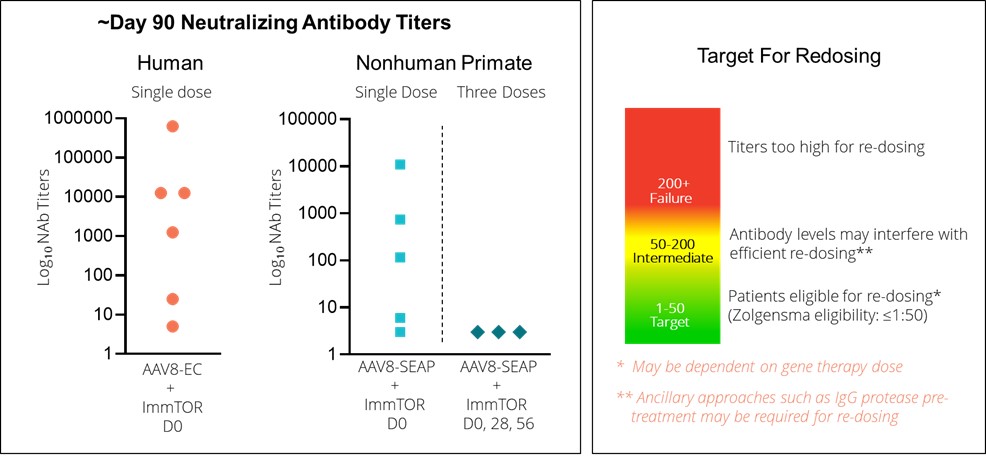
We recently completed a human proof-of-concept trial (SEL-399) in healthy volunteers who were treated with an empty AAV8 capsid (EMC-101), which is an AAV capsid containing no transgene, alone or in combination with ImmTOR. This clinical trial was conducted in Belgium in partnership with Asklepios BioPharmaceutical, Inc., or AskBio (a Bayer AG subsidiary). The goal of the SEL-399 clinical trial was to undesired immune responses. Based onevaluate the data received from our preclinical, Phase 1, Phase 2 dose-finding clinical studies, and certain data disclosed from the ongoing Phase 2 head-to-head (COMPARE) clinical study, we believe that by leveraging ourappropriate dose of ImmTOR platformin humans to mitigate the formation of ADAs followingantibodies to AAV capsids used in gene therapies. Top-line results indicated that AAV8 empty capsids elicited peak median anti-AA8 neutralizing antibody, or NAb, titers of 1:6875. Median day 30 NAb titers were reduced to titers of 1:25 and 1:5 in the dosing0.15 mg/kg and 0.3 mg/kg ImmTOR cohorts, respectively, representing a 50-fold and 250-fold difference, respectively, compared to the median of our pegylated uricase, pegadricase, SEL-212, if successfully developedcontrol subjects dosed with AAV8 empty capsid alone, as depicted in the figure below. Further, we observed that at Day 30, six of six, or 100%, of subjects that received 0.3 mg/kg exhibited NAb titers of 1:25 or less, and approved, could enablefour of six, or 67%, of those subjects at this dose exhibited NAb titers of 1:5 or less. We observed at Day 30 that six of nine, or 67%, of subjects that received 0.15 mg/kg of ImmTOR exhibited NAb titers of 1:25 or less, and two of nine, or 22%, of subjects at this dose had a titer of 1:5 or less. At Day 90, two of six subjects in the removal0.3 mg/kg cohort were observed to have sustained control of uric acid crystals.
The market for gout therapy
Gout is a painful and potentially disabling formneutralizing antibodies with titers of arthritis resulting from excess accumulation1:25 or less. Consistent with preclinical data, we observed that the single dose ImmTOR cohorts showed delayed formation of uric acid and deposition of uric acid crystals in joints and soft tissues, including those of the kidney and heart, causing harmful inflammation. Gout is caused by an overproduction of uric acid, a natural byproduct of purine metabolism that is produced after consumption of food with highneutralizing antibodies which eventually reached similar median levels of purines such as seafood, meat, yeastneutralizing antibodies to the control group by Day 90. Similar data were observed in NHP receiving a single dose of ImmTOR with an AAV gene therapy vector in a preclinical study. Importantly, we observed that two additional doses of ImmTOR, or a total of three monthly doses, provided durable inhibition of neutralizing antibodies in NHP. Although we could not evaluate three-monthly doses of ImmTOR in healthy volunteers, we intend to employ a three-monthly dose regimen in clinical trials performed in patients. ImmTOR showed safety results consistent with prior human studies and certain vegetables, and/or an inabilitywas generally well tolerated. No serious adverse events were reported. The most common treatment-related adverse events included mild-to-moderate stomatitis and rash. We believe this promising trial in healthy volunteers provides support for the potential use of ImmTOR for the kidneysinhibition of neutralizing antibodies to excrete
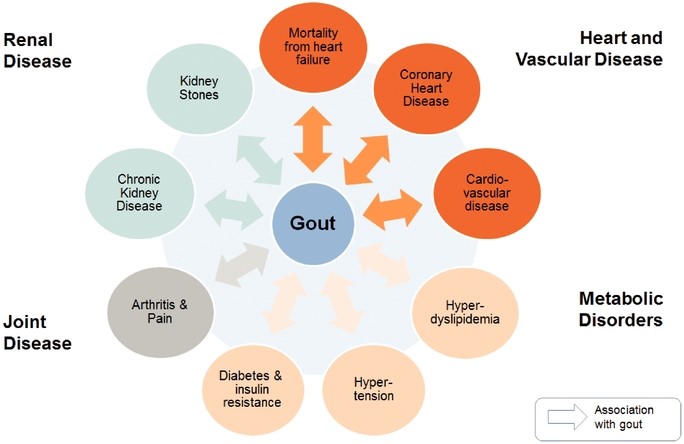
adequate amountsAAV8 in gene therapy clinical trials. The following graphs depict the effect of uric acid from the body. High concentrations of serum uric acid lead to formation of uric acid crystals in joints and tissues, causing pain, inflammation and joint damage, and increase the risk for other conditions, including cardiovascular, cardiometabolic, joint and kidney disease.
There are approximately 8.3 million and 10.0 million gout sufferers in the United States and the European Union, respectively. The first line of treatments for gout are allopurinol and febuxostat. Both drugs are xanthine oxidase inhibitors, oral drugs that reduce the synthesis of uric acid. Lesinurad and probenecid are oral gout drugs that increase the rate of excretion of uric acid through the kidneys, and are used almost exclusively in combination with these first line treatments. While these oral treatments are designed to preventImmTOR on the formation of uric acid deposits, theyneutralizing antibodies to AAV8 capsid in humans and NHP.
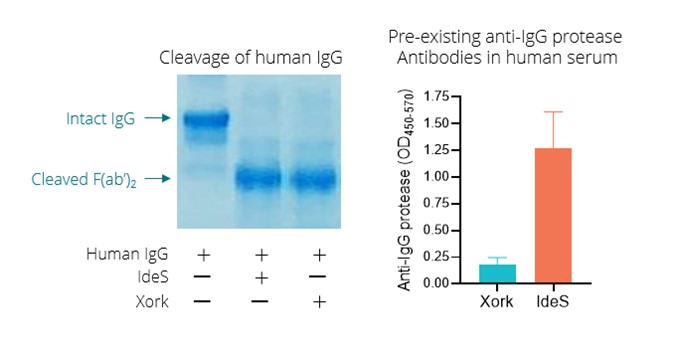
Finally, pre-existing neutralizing antibodies, which develop as a result of prior infection with wildtype AAV, are not well suiteda major exclusion factor in clinical trials causing many potential patients to reduce existing uric acid depositsbe ineligible for gene therapy. We have licensed a bacterial IgG protease, or Xork, from Genovis AB (publ.), or Genovis. IgG proteases have been shown by others to transiently cleave IgG and enable dosing of AAV vectors in jointsthe presence of pre-existing antibodies in NHP. However, IgG proteases, being of bacterial origin, are themselves immunogenic. The most commonly studied IgG protease, called IdeS or imlifidase, is derived from Streptococcus pyogenes, a common human pathogen. Most healthy individuals have been exposed to S. pyogenes and tissues.
Gouthave pre-existing antibodies against IdeS. We believe that Xork is a spectrum of disease thatdifferentiated product candidate, as it is typically diagnosed through the measurement of uric acid levels in the blood and/or the identification of uric acid crystalsderived from a visible tophus. High concentrationsStreptoccocal species that does not infect humans and so the enzyme shows very low cross reactivity to naturally existing antibodies in most human serum. We plan to develop Xork with the intention of serum uric acid increase the risk of co-morbidities, including cardiovascular, cardiometabolic, joint and kidney disease. Patientsenabling access to AAV gene therapy for those patients who are unablecurrently excluded due to reduce their SUA levels below 6.0 mg/dL with oral drugs are diagnosed with refractory gout. Patients whopre-existing anti-AAV antibodies.
Through our analysis of genetic diseases, we have uric acid deposits, or tophi, in soft tissues, joints, the urinary tract, the digestive tract or the heartidentified applications and a persistently elevated uric acid level when left untreated are diagnosed with chronic refractory gout. Tophi are a source of inflammation and pain. Chronic refractory gout constitutes a subset of gout patients exhibiting chronic high serum uric acid levels and painful and damaging uric acid deposits. In total,patient segments that we estimate that there are approximately 160,000 chronic refractory gout patients in the U.S.
Figure 4 below illustrates the association between gout and diseases of the heart, vascular system, metabolic process, kidney and joints.
Figure 4. Co-Morbidities Associated with Gout
Based on our preclinical studies, clinical databelieve would benefit from our Phase 1b, Phase 2 dose-finding, ongoing Phase 2 head-to-head (COMPARE) clinical studies,ImmTOR platform. Our initial area of focus is on genetic metabolic diseases but may also include lysosomal storage diseases and market research,genetic muscular diseases. We believe we believe that SEL-212 may potentially address several keyare the first company to systematically pursue the development of AAV gene therapy product candidates with the goal of enabling repeat administration. We have engaged third parties with experience in gene therapy and rare diseases to support the development of our proprietary products. We also have licensed our ImmTOR platform to AskBio, Sarepta Therapeutics, Inc., or Sarepta, and Takeda Pharmaceuticals USA, Inc., or Takeda, for certain pre-specified targets.
Tolerogenic Therapies for Autoimmune Disease: Autoimmune diseases are caused by a breakdown in natural tolerance to our own self-antigens. With over 24 million Americans afflicted with autoimmune diseases, there is a large unmet needs inmedical need. As the treatment of chronic refractory gout: the durable control of serum uric acid levels, the elimination of painful and damaging uric acid deposits, reduction in incidence and severity of flares, and once-monthly dosing.
Our product development strategyImmTOR platform is designed to address these unmet medical needs while improvinginduce or expand antigen specific T regulatory cells, we believe the dosing regimen to a once-monthly treatment. We completed enrollment for our Phase 2 head-to-head (COMPARE) clinical study against KRYSTEXXA, which is intended to evaluate the differences between KRYSTEXXA and SEL-212 with respect to SUA control, flare rates and dosing convenience, in December 2019. We expect to report top-line data from the COMPARE trial in the third quarter of 2020. In the first quarter of 2020, we received feedback from the FDA on our planned Phase 3 clinical program for SEL-212, which will consist of two placebo-controlled trials, each with a six month primary endpoint, one of which will have a six-month placebo-controlled extension. We plan to commence the Phase 3 clinical program in SEL-212 in the second half of 2020, following which, if successful, we plan to seek regulatory approval for the treatment of refractory gout based on an endpoint of reduction of SUA levels below 6.0 mg/dL for 80% of the time during month six. We intend for the Phase 3 protocols to include patients with chronic refractory gout. During our market research, physicians expressed their preference for a product that could be dosed monthly. If approved, we plan to position SEL-212 as a debulking therapy for gout
thatImmTOR platform has the potential to remove harmful uric acid deposits overtreat autoimmune diseases by restoring self-tolerance to auto-antigens.
Recent preclinical data generated by our scientific team suggest that ImmTOR may have profound synergistic activity with engineered IL-2 molecules that are selective for Tregs. The IL-2 pathway influences critical aspects of both immune stimulation and immune regulation. Tregs express a limited numberhigh affinity form of once-monthlythe IL-2 receptor. Low doses of IL-2 have been shown by others to selectively activate Tregs resulting in expansion of pre-existing Tregs. Clinical trials of
low dose IL-2 have shown evidence of efficacy in small clinical trials of autoimmune diseases, such as systemic lupus erythematosus. Other investigators have shown that IL-2 can be engineered to selectively bind the high affinity IL-2 receptor and subsequently allow patients to switch to oral gout maintenance therapy unless and until such patients experience a subsequent manifestation of uric acid deposits. We do not believeexpand pre-existing Tregs.
In our preclinical studies, we observed that oral therapy would completely prevent the build-up over time of uric acid crystals in patientsImmTOR combined with a historyTreg-selective IL-2 mutant protein, or IL-2 mutein, exhibited substantial synergistic activity in increasing the percentage and durability of chronic refractory gout and, as a result, we anticipate that treatment with SEL-212, if approved, would be required intermittentlytotal Treg expansion in such patients.the spleen. We believe that SEL-212, if approved, may be effective in removing harmful uric acid deposits in most patients with chronic refractory gout over multiple courses of treatment in their lifetimes. Figure 5 below depicts this positioning strategy as a sample diagram illustrating what we believecombination has the potential to be a shiftbest-in-class therapy in the treatment paradigm for chronic refractory gout.diseases where expansion of total Treg may prove beneficial. Furthermore, in our preclinical studies, when we combined ImmTOR-IL with an antigen, we measured an approximately three-fold increase in antigen-specific T regulatory cells vs ImmTOR alone.
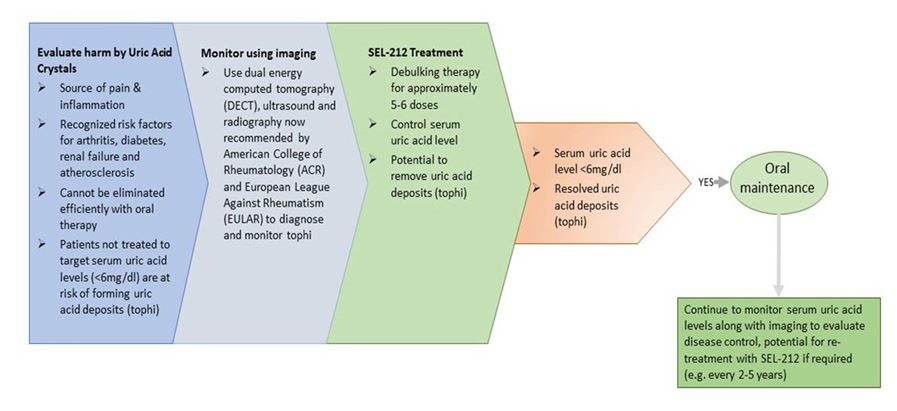
Figure 5. Sample Treatment Course
Our Product Candidates
Below is a summary of our ongoing discovery, research, and development programs:
| | | | | | | | | | | |
| Program | Phase of Development | Anticipated Next Steps | Commercial Rights |
| Biologic Therapies | | | |
SEL-212
(Chronic Refractory Gout) | Phase 3 clinical trials
(DISSOLVE I / DISSOLVE II) | Top-line data Q4 2022 | Sobi |
| IgA nephropathy | Preclinical | IND enabling studies, 2022 | Selecta |
| Gene Therapies | | | |
| Methylmalonic acidemia (MMA) | IND filed / Phase 1 | IND approval and study commencement, 2022 | Selecta |
| Ornithine Transcarbamylase (OTC) Deficiency | IND-enabling | Currently paused | Selecta |
| IgG protease (Xork) | Preclinical | IND enabling studies, 2022 | Selecta |
| Pompe disease | Preclinical | Plans to be announced
by our collaborator | AskBio |
| Duchenne muscular dystrophy (DMD) | Preclinical | Plans to be announced
by our collaborator | Sarepta |
| Limb-girdle muscular dystrophy (LGMD) | Preclinical | Plans to be announced
by our collaborator | Sarepta |
| Two indications for lysosomal storage disorders | Preclinical | Plans to be announced
by our collaborator | Takeda |
| Tolerogenic Therapies for Autoimmune Disease |
Proprietary IL-2
receptor agonist | Preclinical | | Selecta |
| Primary biliary cholangitis (PBC) | Preclinical | | Selecta |
Biologic Therapies – Chronic Refractory Gout.Gout
We expect our clinical and marketing strategy for SEL-212 to initially focus on the estimated 160,000 patients in the United States with chronic refractory gout, and to focus on those who are being treated by rheumatologists. If SEL-212 is approved, we expect our strategy for marketing SEL-212 to rheumatologists will be to promote a switch from oral therapies to SEL-212 for patients with SUA levels chronically above 6.0 mg/dL or those diagnosed with chronic refractory gout. Some rheumatologists have begun to utilize imaging technologies recommended by the guideline writing associations for rheumatology, including the American College of Rheumatology and the European League Against Rheumatism. In particular, DECT, can enable the visualization and quantification of uric acid deposits in joints and in tissues as depicted in Figure 6 below. Figure 6 below depicts a patient, using DECT imaging, with heavy urate burden at onset of study and almost a complete resolution of this after five months.
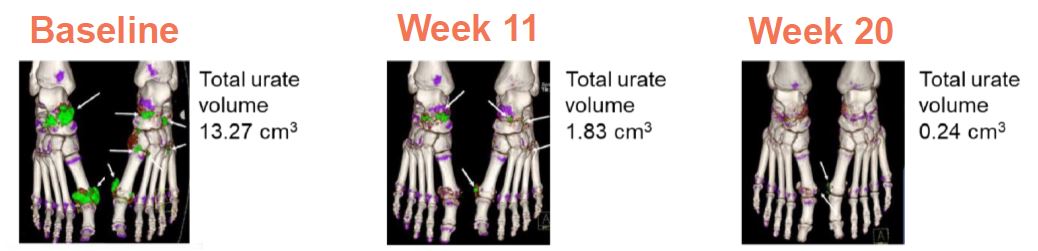
Figure 6. Tophi / Uric Acid Deposits (shown in green) Visualized Using Dual Energy Computed Tomography Imaging
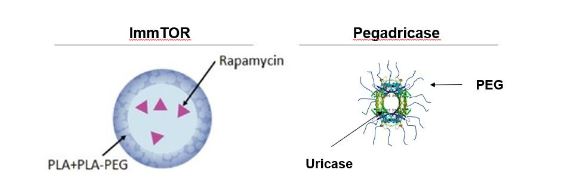
SEL-212 components
Our SEL-212 consists of ImmTOR co-administered with pegadricase. Our ImmTOR consists of nanoparticles composed of poly(D,L-lactide), or PLA, and poly(D,L-lactide)-block-poly(ethylene-glycol), or PLA-PEG, encapsulating rapamycin. Our
pegadricase consists of a yeast-derived uricase modified with poly(ethylene-glycol), or PEG. The components of SEL-212 are depicted in Figure 7 below.
Figure 7. Components of SEL-212
Our pegadricase is a pegylated version of the therapeutic enzyme uricase, which we have licensed from Shenyang Sunshine Pharmaceutical Co., Ltd., or 3SBio, exclusively for all markets, except Japan and Greater China, and exclusively for Japan only in combination with our ImmTOR platform. Uricase is an enzyme endogenous to all mammals, except for humans and certain primates, which converts uric acid to the more soluble metabolite, allantoin. There is a natural limit to the amount of uric acid that can be excreted by the kidneys, which decreases with age and can be reduced by some medications. By converting uric acid to allantoin, uricase provides an additional way for the body to reduce uric acid.
Pegadricase is designed for the treatment of patients with chronic gout, refractory to standard uric acid lowering treatment, by breaking down the excess serum uric acid, or sUA, to the more soluble allantoin. However, the immune response to
pegadricase limits administration to a single dose which is effective for less than one month. The addition of ImmTOR isto pegadricase (SEL-212) allows multiple monthly doses to be administered, thus reducing uric acid for a prolonged time.
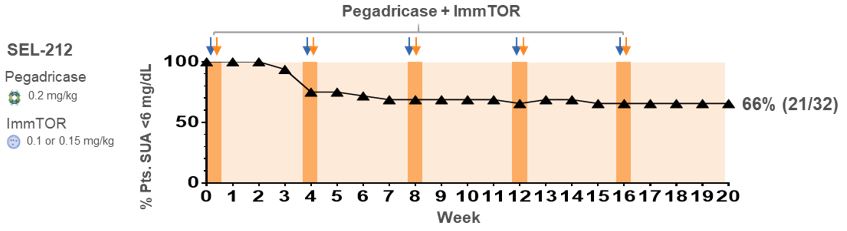
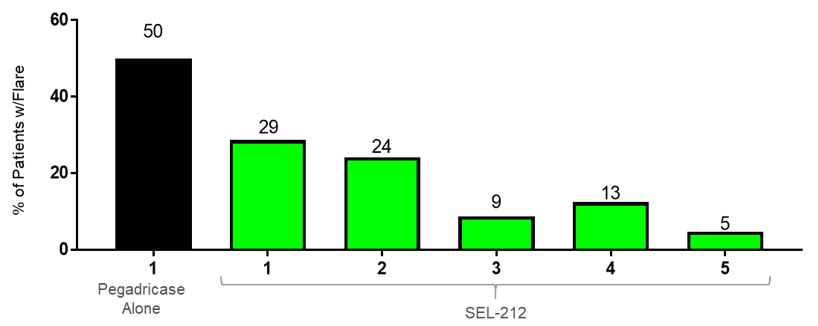
In preclinical studies and in our biodegradable nanoparticle that encapsulates rapamycin, also referred to as sirolimus. Rapamycin is the active ingredient of Rapamune, an immunosuppressant which has extensive prior use in humansPhase 1b and is currently FDA-approved as a prophylaxis of organ rejection in kidney transplant patients aged 13 or older. PLA is part of the broader poly(lactic-co-glycolic acid), or PLGA, family of biodegradable polymers that have more than 30 years of commercial use and are formulation components in a number of approved products. Polyethylene glycol, or PEG, has been widely studied inPhase 2 clinical trials, and is also a formulation component in many approved biologic products. In our preclinical studies,we observed that ImmTOR, when co-administered at the initiation of treatment with a biologic drugpegadricase, SEL-212 substantially reduced the formation of associated ADAsADAs. We believe that SEL-212 serves as proof of concept for the ImmTOR platform in ameliorating the unwanted immune response to an immunogenic biologic. SEL-212 is in two pivotal Phase 3 studies versus placebo, which we refer to as DISSOLVE I and induced antigen-specific immune toleranceDISSOLVE II, with top-line data expected in the fourth quarter of 2022. SEL-212 has been licensed (except as to Greater China) to Sobi, pursuant to our license and development agreement dated June 11, 2020, with Sobi, or the Sobi License.
Enrollment into the DISSOLVE II trial is ongoing and the study is being conducted in the United States and four countries across eastern Europe. As a result of the ongoing and rapidly evolving geopolitical situation in Ukraine and Russia, we have proactively undertaken mitigation steps to prioritize the safety of our patients and investigators, as well as address any potential disruptions. While we have temporarily closed screening and randomization at sites in both Russia and Ukraine, we have added 11 sites in the United States to further enrollment in DISSOLVE II. Out of these additional enrollment sites, nine have been activated and two are pending initiation, with activation expected imminently. Subject to ongoing geopolitical developments, we believe we remain on track to report data in the fourth quarter of 2022, and we will continue to work closely with our partner Sobi, clinical trial providers and regulatory authorities to evaluate any potential delays or adjustments to the biologic drug.timeline of the trial as a result of the circumstances.
Clinical developmentThe market for gout therapy
For chronic refractory gout, we are executingGout is a clinical development program that seeks to evaluate SEL-212’s ability to durably reduce SUApainful and potentially disabling form of arthritis associated with elevation of sUA levels below 6.0 mg/dL. The saturation pointcaused by an overproduction of uric acid and/or an inability of the maximum level at whichkidneys to excrete adequate amounts of uric acid will remain soluble in plasma, is 6.8 mg/dL. Whenfrom the body. High concentrations of sUA lead to formation of uric acid rises above 6.8 mg/dL, uric acid microcrystals begin to form and settlecrystals in joints and tissues, causing pain, inflammation and joint damage, and increase the risk for other tissues, creating an inflammatory response that is diagnosed as gout. This inflammatory response is the source of highly debilitating gout flares experienced by patients. When serum uric acid levels drop below the level of 6.0 mg/dL, microcrystals will beginconditions, including cardiovascular, cardiometabolic, joint and kidney disease.
Patients who are unable to dissolve and, over time, gout symptoms will resolve. Therefore, a serum uric acid level of less than 6.0 mg/dL is defined as the target for uric acid-lowering treatments by organizations such as the American College of Rheumatology and the European League Against Rheumatism. The FDA and EMA have also considered the maintenance of serum uric acidreduce their sUA levels below 6.0 mg/dL with oral drugs are defined as having refractory gout. Chronic refractory gout constitutes a subset of gout patients exhibiting chronic high sUA levels and painful and damaging uric acid deposits. We estimate that there are approximately 160,000 chronic refractory gout patients in the U.S.
We believe SEL-212 may potentially address several key unmet needs in the treatment of chronic refractory gout: the durable control of sUA levels, the elimination of painful and damaging uric acid deposits, reduction in incidence and severity of flares, based on our preclinical studies, clinical trials, and market research. SEL-212 is designed to be an acceptable primary endpoint inaddress these unmet medical needs while improving the dosing regimen to a once-monthly treatment.
SEL-212 Clinical Development
We conducted Phase 1a, Phase 1b, and Phase 2 clinical trials for urate lowering therapies. Available oral gout therapies are designed to prevent the formation of uric acid microcrystals but are not effective in rapidly removing existing crystalized deposits. In severe gout patients who have significant deposits of uric acid crystals, known as tophi, it is of clinical benefit to significantly reduce serum uric acid levels (i.e. below 0.1 mg/dL) for a period of time in order to rapidly dissolve the uric acid crystals.
As of March 8, 2020, we had dosed approximately 300 patients in multiple clinical studies in patients with gout or elevated levels of serum uric acid. We initiated our clinical program in the U.S. in the second quarter of 2015 with a multicenter Phase 1a trial of pegadricase in patients with serum uric acid levels greater than 6.0 mg/dL. We completed the patient treatment portion of our Phase 1a trial in November 2015, initiated a Phase 1b trial in December 2015 and reported data from both Phase 1 clinical trials in December 2016. In our Phase 1b trial, we demonstrated that a single dose of SEL-212 was capable of lowering serum uric acid levels below 0.1 mg/dL for at least 30 days, at therapeutic doses which is correlated with the inhibition of uricase-specific ADAs. We initiated patient recruitment in an open label multi-dose Phase 2 clinical trial of SEL-212 in patients with symptomatic gout and elevated uric acid levels in October 2016. This study was conducted at multiple clinical sites in the U.S. In May 2019, we initiated patient recruitment for our Phase 2 head-to-head (COMPARE) clinical study
against KRYSTEXXA, which is intended to evaluate the differences between KRYSTEXXA and SEL-212 with respect to SUA control. We have completed our initial end-of-Phase 2 meeting with the FDA and believe we have established a path forward for potential BLA submission. Additionally, in the first quarter of 2020, we received feedback from the FDA on our planned Phase 3 clinical program for SEL-212, which will consist of two placebo-controlled trials, each with a six month primary endpoint, one of which will have a six-month placebo-controlled extension.
Phase 1, Phase 2 dose-finding, and Phase 2 head-to-head (COMPARE) clinical trials
SEL-212 was evaluated in a Phase 1 and Phase 2 dose-finding clinical program that included a Phase 1a and Phase 1b clinical trial in patients with high uric acid levels as well as a Phase 2 clinical trial in patients with symptomatic gout and high uric acid levels. Each Phase 1 clinical trial was designed with the primary objective to evaluate the safety and tolerability of SEL-212 and its individual components. Additional objectives of the Phase 1 clinical trials included identifying a pegadricase dose that was capable of lowering serum uric acid levels over a period of one month, evaluating the immunogenicity of pegadricase after a single dose and demonstrating that ImmTOR co-administered with pegadricase reduced uric acid levels and mitigated the formation of uricase-specific ADAs. The Phase 2 clinical trial evaluated the effect of multiple doses over an extended period of time on serum uric acid and the formation of ADAs in patients. We commenced patient recruitment in the multiple ascending dose Phase 2 clinical trial in October 2016 and received final data from this trial in December 2018. We completed enrollment of our Phase 2 head-to-head (COMPARE) clinical study against KRYSTEXXA in December 2019 and expect to report top-line data in the third quarter of 2020. The two-armed, open label trial has enrolled approximately 150 patients, randomized 1:1, with one arm receiving KRYSTEXXA (as set forth in the prescribing information) and the other arm receiving six monthly doses of SEL-212. The primary endpoint in the study is the percentage of patients in each arm that maintain SUA control below 6.0 mg/dL, for at least 80% of the time during months three and six. We plan to commence the Phase 3 clinical program in SEL-212 in the second half of 2020. We will require additional resources to complete the planned Phase 3 clinical program for SEL-212.
Phase 1a clinical trial
The Phase 1a clinical trial for SEL-212 was conducted at multiple sites in the United StatesStates. These trials were designed to demonstrate the induction of an immune response to pegadricase and was athe ability of ImmTOR to mitigate that immune response to both single ascending dose trial of pegadricase alone in 22 patients with elevated serum uric acid levels greater than 6.0 mg/dL. At the outset ofand multiple doses. The table below summarizes the trial eachdesigns and objectives of five cohorts received a single intravenous infusion of pegadricase at ascending dose levels. We monitoredthese clinical trials.
| | | | | | | | |
| Design | Objective |
Phase 1a
SEL-212/101 | •Subjects with hyperuricemia •Single dose of pegadricase •n=22 | •Evaluate safety and tolerability •Define dose of pegadricase that would support monthly dosing •Demonstrate formation of ADAs |
Phase 1b
SEL-212/101 | •Subjects with hyperuricemia •Single dose of SEL-212 •n=53 | •Evaluate safety and tolerability •Proof-of-biological activity: •Mitigation of ADAs after single dose •Select dose to take forward into Phase 2 |
Phase 2
SEL-212/201 | •Subjects with symptomatic gout and hyperuricemia •Repeated monthly dosing of SEL ‑212 •n=152 | •Evaluate safety and tolerability •Clinical proof-of-concept: •Demonstrate sustained reduction of SUA with repeat dosing •Select dose to take into Phase 3 |
In the patients during a 30-day period post-infusion. We commenced enrollment of the clinicalPhase 1a trial, in the second quarter of 2015 and completed the treatment portion of the trial in November 2015. We observed that pegadricase showed no serious adverse events, or SAEs, and was well tolerated at the five dose levels tested. Additionally, we observed that a single dose of pegadricase rapidly reduced and sustained average serum uric acidSUA levels below 6.0 mg/dL for each cohort for 14dose level, although SUA levels returned close to 30 days, depending on the dose level.baseline by day 30. Consistent with our preclinical studies in animals, pegadricase induced uricase-specific ADAs in all patients with varying levels in this Phase 1a trial. The following table depicts the observations from the single ascending dose trial of pegadricase in patients with hyperuricemia. In this trial, patients with SUA > 6 mg/dL at screening were assigned to one of five cohorts receiving a single IV infusion of pegadricase (0.1, 0.2, 0.4, 0.8 or 1.2 mg/kg). Each cohort consisted of five patients, except for cohort 5 (1.2 mg/kg) which enrolled two patients.
Figure 8 below indicates the serum uric acid and uricase-specific ADA levels for each patient in Cohort #3
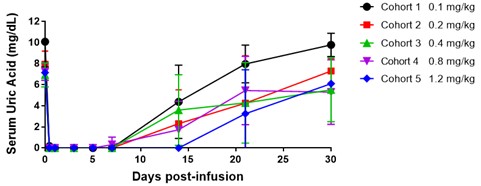
In the Phase 1a clinical trial, which received a single dose of 0.4 mg/kg of pegadricase. The serum uric acid levels were measured at baseline and days seven, 14, 21 and 30 and uricase-specific ADA levels at baseline and days seven, 14 and 30 following the single intravenous injection of pegadricase. We did not measure uricase-specific ADA levels at day 21 in the Phase 1a clinical trial. Patient number two in this cohort developed a relatively low level uricase-specific ADA titer of 40 and maintained uric acid levels below 0.5 mg/dL through the thirtieth day after dosing. By contrast, the remaining four patients in the cohort developed levels of uricase-specific ADAs greater than 1,000 titer and uric acid levels above 5 mg/dL by the thirtieth day after dosing. Based on the results from our Phase 1a1b clinical trial, we observed thatthe potential for increasing doses of ImmTOR to mitigate the formation of pegadricase atADAs and sustain the 0.4 mg/kg dose is capableanti-uricase enzyme activity through 30 days. The cohort of achieving and maintaining a reduction of serum uric acid below the target of 6.0 mg/dL for a 30-day period in the absence of inhibitory uricase-specific ADAs.
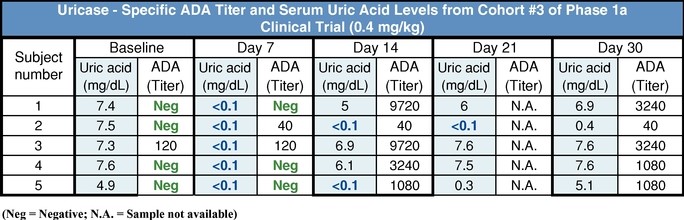
Figure 8. Phase 1a Clinical Trial: Serum Uric Acid and Uricase-Specific ADA Levels of the Third Cohort
Based on our analysis of the Phase 1a clinical trial data, we selected the pegadricase dose of 0.4 mg/kg from Cohort #3 of the Phase 1a clinical trial for further study in the Phase 1b clinical trial.
Phase 1b clinical trial
In December 2015, we initiated our Phase 1b clinical trial at multiple sites in the United States. The clinical trial enrolled 63 patients with serum uric acid levels greater than 6.0 mg/dL. One group of five patientssubjects who received a single 0.4 mg/kg dose of pegadricase alone which we refer to as the Pegadricase Cohort. Four groupsshowed development of patients, each containing two placebo-control patientsuricase ADAs, a short pegadricase half-life, and five test article patients,poor control of SUA at 30 days. Three cohorts who received a placebo or a single intravenous infusion of ImmTOR alone at the following ascending dose levels: 0.03 mg/kg, 0.1 mg/kg, 0.3 mg/kg and 0.5 mg/kg, which we refer to collectively as the ImmTOR Cohorts. Four groups of patients received a single intravenous infusion of ImmTOR at ascending dose levels of 0.03 mg/kg (5 patients), 0.1 mg/kg (10 patients), 0.15 mg/kg (5 patients) or 0.3 mg/kg (5 patients) with a fixed dose of pegadricase of 0.4 mg/kg, which we collectively call the SEL-212 cohorts. All patients were followed for at least 30 days after their initial dose.
Figure 9 below depicts the serum uric acid levels of the Pegadricase Cohort (depicted in red symbols), the ImmTOR Cohorts (depicted in blue symbols), and the SEL‑212 Cohorts (depicted in green symbols) from the Phase 1b clinical trial. Serum uric acid levels were measured at baseline and days seven, 14, 21, 30 and longer for some patients that maintained serum uric acid control at day 21. As expected, ImmTOR alone had no relevant effect on reducing serum uric acid levels across the ImmTOR Cohorts, as such levels remained relatively constant during the 30‑day period. Patients in the Pegadricase Cohort showed an immediate drop in uric acid levels after dosing, with uric acid levels returning close to or above baseline levels by day 21 in four of the five patients, similar to what was observed in Cohort #3 in the Phase 1a study. In contrast, patients in the SEL-212 Cohort receiving the lowest dose of ImmTOR (0.03 mg/kg) co-administered with pegadricase (0.4 mg/kg), showed four out of five patients maintaining serum uric acid levels below 6.0 mg/dL through day 21 after dosing. SEL-212 patients receiving ImmTOR at 0.1 mg/kg co-administered with 0.4 mg/kg of pegadricase plus ascending doses of ImmTOR (0.1, 0.15 and 0.3 mg/kg) showed seven outa dose related reduction in uricase ADAs, an increase in pegadricase half-life, and substantial reduction in SUA. This graphs below depict the observed correlation of 10SUA with anti-uricase IgG and serum pegadricase activity. In these graphs, anti-uricase IgG titers, serum pegadricase activity, and SUA are plotted against time for individual patients maintainedin cohorts A, G, H, and I. Each line represents an individual patient.
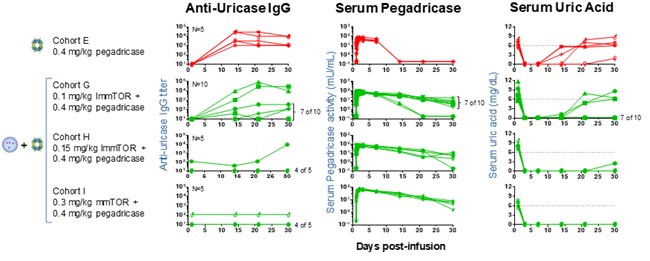
The Phase 2 trial included patients with symptomatic gout and elevated SUA levels in an open-label, multiple ascending-dose clinical trial of serum uric acidSEL-212 to demonstrate sustained reduction of less than 0.1 mg/dL through day 30.SUA and identify the best dose for Phase 3. Five monthly doses of SEL-212 patients receiving ImmTOR at 0.15 mg/kg co-administered with 0.4 mg/kg of pegadricase, showed all five patients maintaining levels of serum uric acid ofreduced SUA to less than 6.0 mg/dL through day 30. Finally, SEL-212in 21 of 32 patients, receiving ImmTOR at 0.3 mg/kg c- administered with 0.4 mg/kg ofor 66%. In distinct contrast, pegadricase showed all five patients maintaining levels of serum uric acid ofalone reduced SUA to less than 0.16.0 mg/dL through day 30. These results indicated that SEL-212 showed a dose-dependent reduction in serum uric acid levels.
Figure 9. Phase 1b Clinical Trial: Uric Acid Levels Across All Phase 1b Cohorts
Figure 10 below depicts the serum uric acid levels at day 30 next to the corresponding day 30 uricase-specific ADA titer for allonly three of 19 patients in the Pegadricase Cohort and in the SEL-212 Cohorts at the three higher dose levels. Four of the five patients treated with 0.4 mg/kg pegadricase alone showed their serum uric return to baseline by day 30 and all five showed uricase specific ADA titers greater than 1000. Seven of 10 SEL-212 patients treated with 0.1 mg/kg ImmTOR and 0.4 mg/kg pegadricase showed serum uric acid levels below 0.1 mg/dL with corresponding low or negative uricase specific ADA titers at day 30. All five SEL-212 patients treated with 0.15 mg/kg ImmTOR and 0.4 mg/kg pegadricase showed serum uric acid below 6 mg/dL at day 30. One patient developed uricase specific ADA titers greater than 1000 but still maintained his serum uric acid below the target threshold of 6 mg/dL at day 30. The other four patients showed no ADA titers and had serum uric acid levels below 0.1 mg/dL. Finally, all five SEL-212 patients treated with 0.3 mg/kg ImmTOR and 0.4 mg/kg pegadricase showed serum uric acid below 0.1 mg/dL at day 30 with corresponding low or negative ADA titers. The one patient with an ADA titer of 120 at baseline showed the same titer at baseline and at each time point tested, indicating that this patient had pre-existing low titer of antibody that was cross-reactive with pegadricase at baseline. These results indicated that SEL-212 mitigated the formation of uricase-specific ADAs which correlated with sustained control of serum uric acid levels through at least day 30month after a single dose.
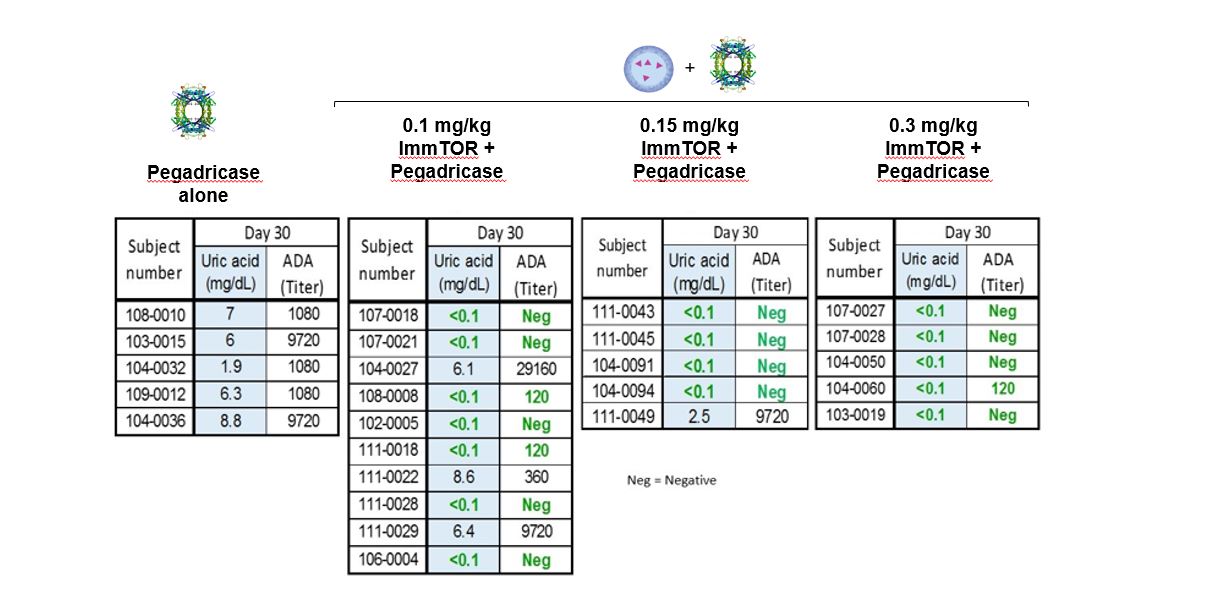
Figure 10. Comparisoninjection (data from pegadricase alone cohorts from the SEL-037/101, SEL-212/101, and SEL-212/201 trials). We believe these results support our view that the addition of Phase 1b Uric Acid and Uricase‑Specific ADA Levels
Certain patientsImmTOR to pegadricase is able dramatically enhance the efficacy of pegadricase in the SEL-212 Cohort were requested to return for additional visits on days 37, 44, and/or 51 because their serum uric acid levels were still under control (<6 mg/dL) at day 21. Not all patients were able to return for the additional visits.long-term reduction of SUA in gout patients. The data for those who could return are in included in Figure 11 below. The data show that uric acid levels eventually returned to baseline, as expectedchart below depicts SUA after a single dose of pegadricase. Importantly, no emergence of ADAs was detected in those patients who exhibited low or no ADA titers at day 30. These data suggest that the treatment with ImmTOR controlled immune responses to pegadricase for up to 51 days after dosing. The data also supportfive monthly dosing in the Phase 2 multiple dose trialdoses of SEL-212.
Figure 11. Comparison of Phase 1b Uric Acid and Uricase‑Specific ADA Levels
At the completion of our Phase 1b program for SEL-212 we had dosed a total of 64 patients in the trial with either SEL-212 (ImmTOR and pegadricase), ImmTOR alone, pegadricase alone or placebo. The median serum acid level of patients enrolled was 7.3 mg/dL, with a mean of 7.4 mg/dL at baseline. Patients presented with an average of 5.3 co-morbidities, such as hypertension and diabetes.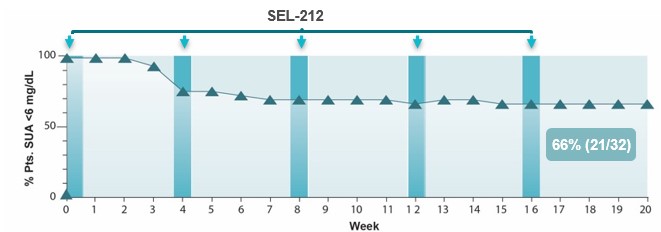
We have generally observed that SEL‑212 and its components, ImmTOR and pegadricase, have beenwere generally well-tolerated in the Phase 1a, 1b, and 2 clinical trials. In the Phase 1a trial we observed no serious adverse events, or SAEs, and that pegadricase was well tolerated in this patient population. There were a total ofat the five dose levels tested. Of the six SAEs in the Phase 1b trial. Of the six SAEs,trial, three were determined to not to be related to study drug by investigators. Of the remaining three SAEs that were determined to possibly or likely be related to study drug, two were cases of stomatitis that occurred at the highest dose of ImmTOR tested (0.5 mg/kg), leading us to define 0.3 mg/kg as the maximum tolerated dose of ImmTOR in this patient population. The remaining SAE was a case of drug hypersensitivity that occurred at a dose of 0.1 mg/kg of ImmTOR in combination with 0.4 mg/kg of pegadricase. In all cases, the patient fully recovered from the SAE without residual effects.
Phase 2 dose-finding clinical trial
In the fourth quarter of 2016, we began enrolling patients with symptomatic gout and elevated serum uric acid levels in an open-label, multiple ascending dose Phase 2 clinical trial of SEL-212. The primary and secondary endpoints for this trial included safety, tolerability, pharmacokinetics, and reduction of serum uric acid and ADA levels. We have also collected data regarding flares and other patient-related observations. We enrolled patients in multiple ascending dose cohorts with the primary goal of identifying the dose regimens to advance into our ongoing Phase 2 head-to-head (COMPARE) clinical study and planned Phase 3 clinical program.
A total of 152 patients were dosed in the Phase 2 trial, at 15 active U.S. clinical sites and the treatment phase of all SEL-212 cohorts had been completed. The following is a summary of clinical activity from the trial as defined by the primary clinical endpoint (i.e., serum uric acid levels below 6 mg/dL):
| |
• | Control and 0.05 mg/kg ImmTOR Dose Cohorts (cohorts receiving five monthly doses of pegadricase alone or three monthly doses of 0.2 mg/kg or 0.4 mg/kg of pegadricase + 0.05 mg/kg of ImmTOR followed by two monthly doses of pegadricase alone): Dosing of patients in the control cohorts receiving pegadricase alone was stopped early due to a loss of clinical activity caused by the immunogenicity of the enzyme. Clinical activity was lost by week 12 in the majority of patients receiving pegadricase in combination with the 0.05 mg/kg dose of ImmTOR.
|
| |
• | 0.08 and 0.1 mg/kg ImmTOR Dose Cohorts (cohorts receiving three monthly doses of 0.2 mg/kg or 0.4 mg/kg of pegadricase + 0.08 or 0.10 mg/kg of ImmTOR followed by two monthly doses of pegadricase alone): A majority of patients in these cohorts maintained clinical activity while receiving the combination therapy through week 12. These results were consistent with the level of clinical activity observed through day 30 at a similar SEL-212 dose level in our Phase 1b trial. At the 0.1 mg/kg dose level, half of the patients that maintained clinical activity through week 12 also maintained clinical activity through week 20.
|
| |
• | 0.125 and 0.15 mg/kg ImmTOR Dose Cohorts (cohorts receiving three monthly doses of pegadricase +0.125 or 0.15 mg/kg of ImmTOR followed by two monthly doses of pegadricase alone): A majority of patients in these cohorts maintained clinical activity while receiving the combination therapy through week 12. These results were consistent with the level of clinical activity observed through day 30 at a similar SEL-212 dose level in our Phase 1b trial.
|
Approximately 81% of evaluable patients (N=27) receiving up to 0.15 mg/kg ImmTOR in combination with 0.2 or 0.4 mg/kg of pegadricase followed by 2 monthly doses of pegadricase alone had serum uric acid control below 6 mg/dL at week 12.
| |
• | 0.10 and 0.15 mg/kg ImmTOR 5 Combination Dose Cohorts (cohorts receiving five monthly doses of pegadricase +0.10 or 0.15 mg/kg of ImmTOR ): Five monthly doses of SEL-212 resulted in sustained serum uric acid control and was well tolerated over the entire treatment period. 100% of the patients that had serum uric acid levels below 6 mg/dL at 12 weeks maintained control through 20 weeks. Approximately 66% of evaluable patients (21/32) completed week 20 with serum uric acid levels below 6 mg/dL. (Figure 12).
|
Figure 12. Five Combination Doses Controlled Serum Uric Acid <6 mg/dL at Week 20 in 66% of Patients
Data from the trial suggested that ADA levels were strongly correlated with serum uric acid levels. We have observed that ImmTOR reduced the formation of ADAs in a dose-dependent manner, which we believe enabled pegadricase to maintain clinical activity for extended durations as compared to pegadricase administered without ImmTOR.
Data from the trial suggest that sustained control of serum uric acid near 0 mg/dL led to reduction in uric acid deposits as measured by DECT imaging.
Approximately 29% of patients who received SEL-212 reported a gout flare during their first month of the trial. This was followed by a decline in flare rates during the remainder of the therapy (Figure 13). By comparison, 50% of patients reported a flare during the first month in the control cohorts receiving pegadricase alone before treatment was stopped due to loss of efficacy and safety.
Figure 13. Percentage of Patients Reporting a Gout Flare per Month
SEL-212 was generally well tolerated at clinically active doses following repeated administrations in the trial. Twenty patientssubjects reported a total of 23 SAEs, in the Phase 2 clinical trial. Nine SAEs were reportednine in the five dose combination cohorts, seven of which were reported to be not related or unlikely related to study drug, and two of which were infusion reactions. All SAEs were successfully treated without further issues.
Phase 2 head-to-head (COMPARE) clinical trial
In March 2019, we initiated a non-registrational Phase 2 head-to-head clinical trial of SEL-212, (COMPARE),which we refer to as COMPARE, in which SEL-212 is beingwas compared against the current FDA-approved therapy for chronic refractory gout, KRYSTEXXA,pegloticase, in multiple clinical sites in the United States.
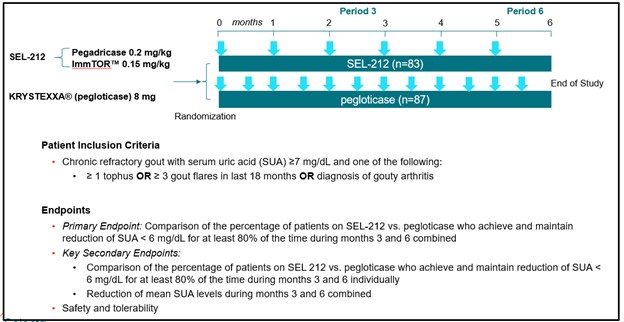
In September 2020, we announced top-line results from the COMPARE trial. We completedobserved a numerically higher response rate to pegloticase on the primary endpoint during months three and six combined but did not meet the primary endpoint of statistical superiority. SEL-212 did generate a statistically significant higher response rate of SEL-212 during the third month of treatment, as well as a statistically significant greater overall reduction in mean SUA levels in SEL-212 versus pegloticase in months three and six combined. We observed a numerically higher response rate of SEL-212 during the sixth month of treatment. In patients with tophi at baseline, we observed substantially higher responder rates for SEL-212 compared to pegloticase on the primary endpoint, and a statistically significant reduction in mean SUA levels when compared to pegloticase.
Approximately 41% of patients in the Phase 2 COMPARE trial had visible tophi at baseline, which is lower than we expected for the general refractory gout population. However, in these most severe patients with tophi, SEL-212 was superior to
pegloticase with a 58% responder rate for SEL-212 versus a 39 % responder rate for pegloticase. Response rates observed in patients with tophi at baseline are depicted below.
SEL-212 showed a favorable safety profile and was well-tolerated. There were no deaths during the study. There were no notable differences in serious TEAEs, treatment-related serious TEAEs, or infusion reactions between the two groups. A full analysis of safety signals, including gout flare incidence and severity, requires evaluation of the full data set and will be reported together with the full efficacy analysis in a manuscript in a medical journal.
Phase 3 DISSOLVE clinical program
In September 2020, we commenced the Phase 3 clinical program of SEL-212, which we refer to as DISSOLVE. We are responsible for the execution of the DISSOLVE program and are being reimbursed by Sobi on a quarterly basis for expenses incurred in connection with these activities. The Phase 3 clinical program consists of two double-blinded, placebo-controlled trials of SEL-212. We refer to these trials as DISSOLVE I and DISSOLVE II. Each trial is expected to enroll up to 120 patients, with up to 40 patients receiving 0.1 mg/kg of ImmTOR and 0.2 mg/kg of pegadricase, up to 40 patients receiving 0.15 mg/kg of ImmTOR and 0.2 mg/kg of pegadricase, and up to 40 patients receiving placebo. An additional 15 patients were added to the study sample size to account for potential treatment discontinuations that may occur due to the ongoing COVID-19 pandemic as a result of the emergence of COVID-19 variants which were not accounted for in the sample size calculations.
We commenced enrollment of this studyDISSOLVE I in September 2020 and DISSOLVE II in December 20192020. In December 2021, we announced the completion of enrollment of the DISSOLVE I trial. DISSOLVE I has a six-month primary endpoint followed by a six-month safety extension. DISSOLVE II will have a six-month primary endpoint with no extension. The primary endpoint of the DISSOLVE program is the maintenance of sUA levels below 6 mg/dL at six months. We currently expect that topline data from the Phase 3 DISSOLVE clinical program is expected in the fourth quarter of 2022.
Enrollment into the DISSOLVE II trial is ongoing and expectthe study is being conducted in the United States and four countries across eastern Europe. As a result of the ongoing and rapidly evolving geopolitical situation in Ukraine and Russia, we have proactively undertaken mitigation steps to prioritize the safety of our patients and investigators, as well as address any potential disruptions. While we have temporarily closed screening and randomization at sites in both Russia and Ukraine, we have added 11 sites in the United States to further enrollment in DISSOLVE II. Out of these additional enrollment sites, nine have been activated and two are pending initiation, with activation expected imminently. Subject to ongoing geopolitical developments, we currently believe we remain on track to report top-line data in the fourth quarter of 2022, and we will continue to work closely with our partner Sobi, clinical trial providers and regulatory authorities to evaluate any potential delays or adjustments to the timeline of the trial as a result of the circumstances.
Biologic Therapies – IgA Nephropathy
The second indication in our biologic therapies program is IgA nephropathy, an autoimmune kidney disease that occurs when immune complexes of a subclass of antibodies called immunoglobulin A1, or IgA1, accumulates in the kidneys. Previous studies in animal models conducted at independent laboratories demonstrated that bacterial IgA protease has the potential to remove injurious IgA immune complexes from kidneys and reduce inflammation, fibrosis, and hematuria. We believe these results suggest that IgA protease can potentially decrease the rate of disease progression and possibly even reverse the disease. The barrier to IgA protease commercialization has been the bacterial origin of the protease, which makes it highly immunogenic.
In clinical trials, we have observed ImmTOR mitigating the formation of ADAs to immunogenic enzymes. We intend to combine an IgA protease with our ImmTOR platform to develop a novel combination product candidate for the treatment of IgA nephropathy and IgA-mediated diseases.
In October 2020, we entered into an Option and License Agreement, or the IGAN Agreement, with IGAN Biosciences, Inc., or IGAN. Pursuant to the IGAN Agreement, IGAN granted us an exclusive license to research, evaluate, and conduct pre-clinical development activities on IGAN’s proprietary IgA proteases. We have an option term of 24 months, during which we can elect to obtain an exclusive license to further develop and commercialize the product candidate to treat all IgA-mediated diseases, including IgA nephropathy, Linear IgA bullous dermatitis, IgA pemphigus, and Henoch-Schonlein purpura (also known as IgA vasculitis).
In October of 2021 we entered into a Collaboration and License Agreement, or the First Ginkgo Agreement, with Ginkgo. Pursuant to the First Ginkgo Agreement, Ginkgo will leverage their high throughput enzyme discovery, design and screening capabilities to identify and further optimize a proprietary, next generation IgA protease. We have an exclusive license to further develop and commercialize the discovered IgA proteases to treat all IgA-mediated diseases, including IgA nephropathy, Linear IgA bullous dermatitis, IgA pemphigus, and Henoch-Schonlein purpura.
These collaborations build on extensive preclinical as well as strong clinical data from our Phase 2 COMPARE trial for the treatment of chronic refractory gout that we believe further supports ImmTOR’s potential for sustained therapeutic benefit when combined with immunogenic enzymatic therapies.
We are continuing to perform pre-clinical IND enabling studies.
Gene Therapies – Methylmalonic Acidemia
Our lead therapeutic gene therapy program, SEL-302, is intended to use ImmTOR to enhance the treatment of methylmalonic acidemia, or MMA, an inherited disorder in which the body is unable to process certain proteins and fats (lipids) properly. This program was previously being conducted under our collaboration with AskBio. In October and November 2020, we received rare pediatric disease designation and orphan drug designation, respectively, from the FDA for MMA-101, for the treatment of MMA due to methylmalonyl-CoA mutase, or MMUT gene mutations. See “⸺Licenses and Collaborations⸺AskBio” for more information. In April 2021, we were notified by AskBio that it intended to opt-out of development of the MMA indication. The feasibility study and license agreement with AskBio, or AskBio Collaboration Agreement, otherwise remains in effect. Manufacturing of a new lot has been completed and is currently undergoing final release testing. We filed an IND to conduct a Phase 1/2 clinical trial of SEL-302 in pediatric patients with methylmalonic acidemia in the third quarter of 2020. The two-armed, open label2021. On November 23, 2021, this trial has enrolled approximately 150 patients, randomized 1:1,was placed on clinical hold by the FDA, with one arm receiving KRYSTEXXA (as set forthquestions specifically relating to chemistry, manufacturing and controls, or CMC, of the AAV vector. On February 9, 2022, we submitted a written response to the FDA to answer its questions. On March 9, 2022, we received a letter from the FDA indicating the clinical hold was removed and the trial may proceed.
Gene Therapies – OTC Deficiency
Our second proprietary gene therapy product candidate, SEL-313, is being developed to treat ornithine transcarbamylase, or OTC deficiency, and is currently in preclinical development. OTC deficiency is a rare genetic disorder that causes ammonia to accumulate in the prescribing information) and the other arm receiving six monthly doses of SEL-212. The primary endpointblood due to mutations in the studyOTC gene, which is the percentage of patients in each arm that maintain SUA control below 6.0 mg/dL,critical for at least 80%proper function of the time during months threeurea cycle. The most severe form of the disorder presents within the first few days of life. Severe symptoms include inability to control body temperature and six.breathing rate, seizures, coma, developmental delays and intellectual disability. Less severe forms of the disorder are characterized by delirium, erratic behavior, aversion to high protein foods, vomiting and seizures. The development of this program is currently paused.
OurWe have exclusively licensed Xork, an IgG-specific protease from Genovis, an enzyme technology company. We plan to develop Xork, either alone or in combination with our ImmTOR programsplatform, with the goal of enabling the dosing of transformative gene therapies in patients with pre-existing AAV immunity due to natural exposures to AAV viruses. Currently, significant proportions of the potential patient populations for immune tolerance in gene therapy
Overview
Althoughmany gene therapy has made significant progress over the last several years, it faces certain limitationstrials are ineligible for treatment by AAV mediated gene therapies due to undesired immunogenicity to eitherpre-existing antibodies which limits transduction efficiency of the AAV vector or the encoded transgene.
Gene vector-specific ADAs frequently occur for patients who receive gene therapy and have been found to prevent the AAV vector from reaching its target cell. It is unknown exactly how long these neutralizing ADAs prevent redosing of gene therapy. However, it has been observed in studies with animals and humans that high titer AAV-specific ADAs develop and persist for more than 10 years, preventing vector readministration. In addition, cellularcould trigger potentially dangerous immune responses have been found to correlate with liver inflammation and loss of gene expression.responses.
Because of this, the inductioncommercial potential of lasting antibodies against AAV, we believe many gene therapy companies have focused ontherapies may be significantly limited due to the single localized dose in diseasesineligibility of large segments of the eyetarget patient population. This is particularly important for gene therapies given the low incidence and central nervous systemsmall prevalent populations of rare monogenic diseases which many gene therapies target. Patients that are ineligible for which immunogenicity is perceivedgene therapy due to be lesspre-existing immunity may have few or no alternative therapies available to them.
Xork, as a pre-treatment in advance of an issue.AAV gene therapy, can potentially open a treatment window by cleaving the IgG antibodies and transiently reducing their concentration.
IgG proteases are bacterial proteins, which are themselves immunogenic. We believe that manyXork is differentiated from other IgG proteases since it is not derived from a human pathogen and thus, exhibits low cross-reactivity with human sera. Additionally, to further reduce any potential immunogenicity of the Xork enzyme, we plan to explore the combination of Xork and ImmTOR. The following images depict how Xork cleaves human IgG specifically and efficiently, but shows low cross reactivity to human sera compared to IdeS, an IgG protease derived from the common human pathogen Streptococcus pyogenes.
We believe that the combination of Xork and ImmTOR has the potential to address two of the major challenges to AAV gene therapy, applications may require or would benefit from multiple doses, especiallypre-existing immunity that limits eligibility and de novo immunogenicity that can adversely affect safety and durability and which prevents the ability to re-dose AAV. We plan to explore the application of Xork in combination with our wholly owned gene therapies, designedas well as explore partnering opportunities for the enzyme with other gene therapy companies.
Tolerogenic Therapies for Autoimmune Disease – ImmTOR & ImmTOR-IL
We intend to apply our ImmTOR platform to treat autoimmune diseases. In preclinical studies, we have observed ImmTOR’s ability to induce antigen-specific T regulatory cells. We believe that ImmTOR, in combination with an autoantigen of interest, could create self-tolerance to auto-antigens and thus be a novel approach to the treatment of autoimmune diseases.
Additionally, in preclinical studies we have observed ImmTOR, in combination with IL-2 muteins, expanding T-regulatory cells beyond IL-2 alone and we intend to pursue a combination of ImmTOR and IL-2 (ImmTOR-IL) in diseases via intravenous administration. Somewhere general T cell expansion has shown a therapeutic benefit. Additionally, in our preclinical data we have observed a three-fold increase in antigen specific regulatory T cells when ImmTOR and IL-2 is combined with an antigen of these rare genetic deficienciesinterest. We intend to pursue and develop treatments for autoimmune diseases with well-defined antigens using either ImmTOR or ImmTOR-IL.
Cyrus Biotechnology, Inc., a collaboration partner, is engineering a proprietary IL-2 protein to combine with the ImmTOR platform to potentially mitigate unwanted immune responses by reducing the inherent immunogenicity of the protein while also promoting immune tolerance. The IL-2 pathway influences critical aspects of both immune stimulation and immune regulation, through the development and expansion of Treg cells. These Treg cells are best treated when patientsa specialized subpopulation of T cells involved in suppressing certain immune responses and maintaining the body’s self-tolerance. In preclinical studies investigating the effects of ImmTOR in combination with a Treg-selective IL-2 mutant protein, or IL-2 mutein, we have observed a substantial synergistic activity in increasing the percentage and durability of Treg expansion in the spleen.
Our lead autoimmune diseases indication is PBC, a T cell driven autoimmune disease that causes progressive destruction of the bile ducts. Patients with PBC are infantsin need of a highly targeted, liver-directed approach to treating the root cause of the disorder. We believe PBC has a well-defined target antigen, significant unmet medical need, and is well suited to the application of our ImmTOR immune tolerance platform, as preclinical data suggest that ImmTOR has the potential to enhance the tolerogenic environment in the liver and provide a hepatoprotective benefit.
Licenses and Collaborations
We intend to both partner and strategically out-license ImmTOR, ImmTOR-IL and Xork for use with other products that are outside our focus to larger biopharmaceutical companies. We believe our ImmTOR platform may also be of interest to biopharmaceutical companies with novel biologic development concepts or small childrenproduct candidates in clinical development that have demonstrated initial efficacy but are experiencing issues with safety or sustained efficacy due to inhibitory ADAs. Additionally, we may strategically in-license products to combine with ImmTOR and develop novel therapeutic products independently. Our key partnerships are listed below.
In-licenses
Ginkgo Bioworks Holdings, Inc.
On October 25, 2021, we entered into the First Ginkgo Agreement with Ginkgo. Under the First Ginkgo Agreement, Ginkgo will design next generation IgA proteases with potentially transformative therapeutic potential. In return, Ginkgo is eligible to earn both upfront research and development fees and milestone payments, including certain milestone payments in the form of our common stock, clinical and commercial milestone payments of up to $85 million in cash, as well as downstream value in the form of royalties on sales.
On January 3, 2022, we entered into a Collaboration and License Agreement, or the Second Ginkgo Agreement, with Ginkgo. Under the Second Ginkgo Agreement, we will engage with Ginkgo to design novel AAV capsids with potentially improved transduction, enhanced tissue tropism and reduced immunogenicity. In return, Ginkgo is eligible to earn both upfront research and development fees and milestone payments, including certain milestone payments in the form of our common stock, clinical and commercial milestone payments of up to $207 million in cash for each of a specified number of products which have the potential to total, in the aggregate, up to $1.1 billion. Ginkgo is also entitled to potential further downstream value in the form of royalties on sales.
Genovis
On October 21, 2021, we entered into a strategic licensing agreement with Genovis, or the Genovis Agreement. Under the Genovis Agreement, we paid to Genovis an upfront payment in exchange for an exclusive license to Genovis’ Xork enzyme technology for all therapeutic uses in humans, excluding research, preclinical, diagnostic, and other potential non-therapeutic applications of the enzyme. Genovis is eligible to earn development and sales-based milestones, as well as tiered royalties on worldwide sales in the low double digits.
Cyrus Biotechnology, Inc.
On September 7, 2021, we entered into a Collaboration and License Agreement with Cyrus, or the Cyrus Agreement, pursuant to which Cyrus agreed to grant us an exclusive, worldwide license to certain intellectual property in order to prevent developmental defects. However, pediatric patientsform a protein engineering collaboration combining the ImmTOR platform with Cyrus’ engineered protein therapeutics. We expect that novel engineered protein therapeutic candidates from the partnership will be used to expand our proprietary pipeline and further bolster the ImmTOR platform. In return for the licensed intellectual property, we made an upfront payment and will pay certain discovery, development, and sales-based milestones which could potentially total up to approximately $1.5 billion across multiple programs.
IGAN Biosciences
In October 2020, we entered into the IGAN Agreement. Pursuant to the IGAN Agreement, IGAN granted us an exclusive license to research, evaluate, and conduct preclinical development activities on IGAN’s proprietary IgA proteases. We have an option term of 24 months, or the Option Term, during which we can elect to obtain an exclusive license to further develop and commercialize the product to treat all IgA-mediated diseases, including IgA nephropathy, Linear IgA bullous dermatitis, IgA pemphigus, and Henoch-Schonlein purpura.
Upon execution of the IGAN Agreement, we paid IGAN a one-time upfront payment of $0.5 million and we would owe additional payments to IGAN if we were to opt-in to an exclusive license agreement, as well as upon the achievement of certain development and sales milestones. During the Option Term, we may haveterminate the IGAN Agreement immediately for any reason upon written notice to IGAN. If we opt-in to an exclusive license agreement, we may terminate the IGAN Agreement upon 120 days’ written notice.
Out-licenses
Takeda Pharmaceuticals USA, Inc.
On October 1, 2021, we entered into a higher needstrategic licensing agreement with Takeda, or the Takeda Agreement. Under the Takeda Agreement, we granted Takeda an exclusive license to our ImmTOR technology initially for repeat dosing duetwo specified disease indications within the field of lysosomal storage disorders. Under the terms of the Takeda Agreement, we received an upfront payment and are entitled to higher cell turnoverreceive up to $1.124 billion in future additional payments over the course of the partnership that are contingent on the achievement of development or commercial milestones or Takeda’s election to continue its activities at specified development stages. We are also eligible for tiered royalties on future commercial sales of any licensed products.
Swedish Orphan Biovitrum
In June 2020, we announced that we had entered into the Sobi License, pursuant to which we agreed to grant Sobi an exclusive, worldwide (except as to Greater China) license to develop, manufacture and commercialize SEL-212, which is currently in development for the treatment of chronic refractory gout. In September 2020, pursuant to the Sobi License, Sobi paid us a one-time, upfront payment of $75 million. Sobi has also agreed to make milestone payments totaling up to $630
million to us upon the achievement of various development and regulatory milestones and sales thresholds for annual net sales of SEL-212, and tiered royalty payments ranging from the low double digits on the lowest sales tier to the high teens on the highest sales tier.
Additionally, Sobi purchased an aggregate of 5,416,390 shares of our common stock at a purchase price of $4.6156 per share for aggregate gross proceeds of $25 million, which we refer to as the patient grows. Accordingly,Sobi Private Placement. The closing of the Sobi Private Placement occurred on July 31, 2020.
Under the Sobi License, we believewill have operational oversight of the Phase 3 DISSOLVE clinical program of SEL-212 (DISSOLVE I and DISSOLVE II) that commenced in September 2020, at Sobi’s expense.
Sarepta Therapeutics
In June 2020, we entered into a solution enabling repeat dosingresearch license and option agreement with Sarepta, or the Sarepta Agreement. Pursuant to the agreement, we granted Sarepta a license to research and evaluate ImmTOR in combination with Sarepta’s AAV gene therapy or gene editing technology, using viral or non-viral delivery, or the Sarepta Product, to treat Duchenne Muscular Dystrophy and certain Limb-Girdle Muscular Dystrophy subtypes, or the Sarepta Indications. Sarepta will have an option term of 24 months during which it can opt-in to obtain an exclusive license to further develop and commercialize the Sarepta Product to treat at least one Sarepta Indication, with a potential to extend the option term if Sarepta pays an additional fee to us. Sarepta made an upfront payment to us upon signing of the agreement, and we are eligible to receive additional payments under the option term. If Sarepta opts-in to an exclusive license agreement, we could receive option exercise payments per indication and we would significantly expandbe entitled to significant development and commercial milestone payments and tiered royalties ranging from the numbermid-to-high single digits based on net sales. In June 2021, we received a payment of diseases and patients that could be treated with gene therapies.
SEL-302$3.0 million for the Treatmentachievement ofMethylmalonic Acidemia certain pre-clinical milestones.
AskBio
Feasibility Study and License Agreement
In August 2019, we entered into a feasibility study and license agreement with AskBio, or the AskBio Collaboration Agreement, pursuant to which we and AskBio will conduct proof of concept studies to potentially validate the use of our ImmTOR platform in conjunction with an AAV gene therapy to mitigate the formation of neutralizing anti-AAV capsid antibodies, which currently precludes redosing. One of theAgreement. The initial product candidatescandidate being developed under this collaboration is a gene therapy candidate for MMA which can cause severe developmental defects and premature death as a result of an accumulation of toxic metabolites. We previously conducted preclinical studies for this product candidate based on SEL-302 and will leverage that previous work within the collaboration.
SEL-313 for the Treatment In April 2021, we were notified by AskBio that it intended to opt-out of OTC Deficiency
For our second gene therapy program, we plan to develop another product candidate for the treatment of ornithine transcarbamylase, or OTC, deficiency, which is another metabolic disorder. We are pursuing this second potential indication through collaborations with third parties with preclinical and clinical experience in this area.
Hemophilia A
On December 2, 2016, or the Spark Signing Date, we entered into the Spark License Agreement to collaborate on the development of gene therapies for certain targets utilizingthe MMA indication. The feasibility study and license agreement with AskBio, or AskBio Collaboration Agreement, otherwise remains in effect. We filed an IND to conduct a Phase 1/2 clinical trial of our ImmTOR platform. Under the terms of the Spark License Agreement, we granted Spark certain exclusive, worldwide, royalty-bearing licenses to our intellectual property and know-how relating to our ImmTOR platform to research, develop and commercialize gene therapies for hemophilia A, which is the initial target under the Spark License Agreement.
For more information about the Spark License Agreement, see the discussion under “Licenses and Collaborations - Spark” below.
Crigler Najjar Syndrome
In September 2018, we announced a collaboration with the European consortium, CureCN, for an ImmTOR+AAV gene therapy combinationSEL-302 product candidate in Crigler-Najjar syndrome,pediatric patients with methylmalonic acidemia in the third quarter of 2021. On November 23, 2021, this trial was placed on clinical hold by the FDA, with questions specifically relating to CMC of the AAV vector. On February 9, 2022, we submitted a rare genetic disorder characterized by an inabilitywritten response to properly convert and clear bilirubinthe FDA to answer its questions. On March 9, 2022, we received a letter from the body. The consortium plansFDA indicating the clinical hold was removed and the trial may proceed. ImmTOR manufacturing, controlled by us, continues to utilizeproceed in accordance with our expectations and we have not observed any impact to any of our ImmTOR platform in combination with their AAV gene therapy with the potential to re-dose patients who are not producing enough enzyme post their first dose of AAV gene therapy. We expect the CureCN consortium to obtain scientific adviceprograms. In October and November 2020, we received rare pediatric disease designation and orphan drug designation, respectively, from the German drug regulatory authority in 2020.
MANUFACTURING
We manufacture ImmTOR using a scalable, self-assembly nanoemulsion process with well-defined, pharmaceutical unit operations. This proprietary, highly specialized and precisely controlled manufacturing process enables us to reproducibly manufacture ImmTOR across many production scales, from milligram-scale at the laboratory bench to hundreds of grams to multi-kilogram scaleFDA for commercial production. This well-defined process has been produced at multiple scales. We have also developed and executed the required detailed analytic characterization of our products.
For the SEL-212 program, we have increased ImmTOR production to a 50-gram scale and have developed an approximately 400-gram scale process, which, at the current projected clinical dose, we believe would be suitable for commercialization. The process is designed such that this same equipment is capable of potentially producing up to a one kilogram batch size scale. As our nanoparticle manufacturing process is compact, and therefore also portable, our strategy is to transfer our custom designed process skids to a contract manufacturing organization, or CMO, and have the CMO produce the nanoparticles, under our direction. This is the strategy we use for production of clinical supplies for clinical trials and would be the expected strategy for commercial production.
The pegadricase enzyme for SEL-212 is produced by fermentation in E. Coli and is sourced from 3SBio in China. 3SBio is a Chinese pharmaceutical company that produces multiple approved products in China and also has product sales in other countries around the world. 3SBio supplies the pegadricase used in the current clinical trials of SEL-212 in the United States. Through a licensing arrangement, we own exclusive worldwide rights to pegadricase outside of China, with co-ownership of rights in Japan and with 3SBio owning all rights in China. Under this arrangement, 3SBio has agreed to supply us with pegadricase. We also have selected a back-up supplier for pegadricase in the United States.
LICENSES AND COLLABORATIONS
AskBio
Feasibility Study and License Agreement
In August 2019, we entered into the AskBio Collaboration Agreement. Pursuant to the AskBio Collaboration Agreement, we and AskBio agreed to license intellectual property rights to each other as part of a collaboration to research, develop, and commercialize certain AAV gene therapy products utilizing our ImmTOR platform to enable re-dosing of such AAV gene therapy products to treat serious rare and orphan genetic diseases for which there is a significant unmet medical need. Pursuant to the AskBio Collaboration Agreement, we and AskBio will conduct proof of concept studies, or POC Studies, to validate the use of ImmTOR in conjunction with AAVMMA-101, for the treatment of MMA based on our product candidate, SEL-302,due to mitigate the formation of neutralizing anti-AAV capsid antibodies. If the POC Studies are successful,methylmalonyl-CoA mutase, or the parties otherwise elect to do so, the parties will proceed with a collaboration to pursue the development and commercialization of AAVMMUT gene therapy products utilizing ImmTOR for the treatment of certain agreed serious rare and orphan genetic diseases. If the POC Studies fail to demonstrate a proof of concept, and the parties do not mutually agree in writing to proceed with the collaboration, the Agreement will expire. We plan to enter the clinic under this collaboration in 2020.
We and AskBio will share responsibility for the research, development and commercialization of products developed under this collaboration. The parties will also share research, development and commercialization costs equally for all collaboration products, but with a right of either party to opt out of certain products, and thereby no longer be required to share costs for such products. Each party will receive a percentage of net profits for each product sold under the collaboration equal to the percentage of shared costs borne by such party in the development of such product. Pursuant to the AskBio Collaboration Agreement, AskBio is responsible for manufacturing the AAV capsids and AAV vectors and we are responsible for manufacturing ImmTOR.mutations.
License Agreement for Pompe Disease
In December 2019, we entered into the AskBio License Agreement. PursuantAgreement which provides AskBio with exclusive worldwide rights to the AskBio License Agreement, AskBio exercised its option to exclusively license our intellectual property rights covering ImmTOR platform to research, develop and commercialize certain AAV geneAAV-gene therapy products utilizing ImmTOR, and targeting the GAA gene, or derivatives thereof, to treat Pompe Disease, which we refer to as the Licensed Products.
Disease. Pursuant to the AskBio License Agreement, AskBio agreed to pay topaid us upfront fees of an aggregate of $7.0 million. Also pursuant to the AskBio License Agreement, AskBio agreed to make additional payments to us based on the achievement of certain development and commercial milestones of up to an aggregate of $237.0 million. AskBio will also be obligated to make tiered royalty payments to us at percentages in the mid-to-high single digits to us based on achievement of certain sales milestones.
We will supply AskBio with our ImmTOR platform and AskBio will be responsible for all preclinical, clinical and commercial manufacture and supply of Licensed Productsproducts licensed under the AskBio License Agreement (other than ImmTOR) and carry out all other activities related to the research, development, and commercialization of Licensed Productssuch products at its sole expense, including all regulatory activities related thereto.
The AskBio License Agreement contains other customary terms and conditions, including representations and warranties, covenants, termination, and indemnification obligations in favor of each party.
Spark Therapeutics
In December 2016, we entered into the Spark License Agreement to develop gene therapies for certain targets utilizing our ImmTOR platform.
Under the terms of the Spark License Agreement, we granted Spark certain exclusive, worldwide, royalty-bearing licenses to our intellectual property and know-how relating to our ImmTOR platform to research, develop and commercialize gene therapies for Factor VIII, an essential blood clotting protein relevant to the treatment of hemophilia A, which is the initial target under the license agreement.
Pursuant to the Spark License Agreement, Spark made an upfront payment to us of $15.0 million. Additionally, Spark purchased an aggregate of $15.0 million of our common stock in connection with the Spark License Agreement. We will be eligible to receive up to an aggregate of $430.0 million in milestone payments, with up to $65.0 million being based on Spark’s achievement of specified development and regulatory milestones and up to $365.0 million for commercial milestones, as well as tiered royalties on global net sales at percentages ranging from mid-single to low-double digits. Until December 2019, Spark had the right to fund up to 50.0% of any development or regulatory milestone payable to us by issuing us shares of Spark’s common stock having a fair market value equal to the percentage of such development or regulatory milestone, as applicable.
In addition, until December 2019, Spark was eligible to exercise options to research, develop and commercialize gene therapies utilizing our ImmTOR platform for up to four additional targets. Spark did not exercise any of these options prior to the termination of the election period.
Each party is responsible for its own costs and expenses incurred in connection with its respective activities under the Spark License, except that Spark has agreed to reimburse us for FTE and out-of-pocket costs incurred in performing certain tasks or assistance specifically requested by Spark. We retain the responsibility to manufacture Spark’s preclinical, clinical and commercial requirements for the ImmTOR platform, subject to the terms of the Spark License.
The Spark License Agreement will continue on a country-by-country and product-by-product basis until the expiration of Spark’s royalty payment obligations with respect to such product in such country unless earlier terminated by the parties. The Spark License Agreement may be terminated by Spark for convenience upon ninety days’ notice. Either party may terminate the Spark License Agreement on a target-by-target basis for material breach with respect to such target.
In connection with the Spark License Agreement, we also entered into a stock purchase agreement with Spark, pursuant to which Spark has purchased in the aggregate $15.0 million of our common stock including (i) 197,238 shares for an aggregate purchase price of $5.0 million on the Spark Signing Date, (ii) 324,362 shares for an aggregate purchase price of $5.0 million on June 8, 2017, and (iii) 205,254 shares for an aggregate purchase price of $5.0 million on October 31, 2017.
Massachusetts Institute of Technology
In November 2008, we entered into a license agreement with the Massachusetts Institute of Technology, or MIT, which we refer to in its amended form as the MIT License. We amended the MIT License in January 2010, November 2012, August 2013, November 2016, December 2019 and December 2019.June 2020. Under the MIT License, we acquired an exclusive worldwide license, with the right to grant
sublicenses, to develop, make, sell, use and import certain licensed products that are therapeutic or prophylactic vaccines and use certain licensed processes in the exercise of rights to the licensed products, the manufacture, sale and practice of which are covered by patent rights owned or controlled by MIT, including patents jointly owned with Brigham and Women’s Hospital, or Brigham, the President and Fellows of Harvard College, the Immune Disease Institute and the Children’s Medical Center Corporation. Our exclusivity is subject to certain retained rights of these institutions and other third parties.
Upon our entry into the MIT License, we paid MIT a non-refundable license issue fee, reimbursed certain of MIT’s costs and issued shares of our common stock to MIT and the other institutional patent owners which were subject to certain anti-dilution, registration and other protective rights. We are obligated to pay MIT creditable annual maintenance fees, low-single-digit running royalty on annual net sales, developmental milestones up to an aggregate of $1.5 million, a mid-single digit percentage of certain payments we receive from corporate partners and a specified percentage of certain income received from sublicensees after 2009 between 10% and 30%. We may terminate the MIT License at any time upon six monthsmonths’ written notice. MIT has the right to terminate the MIT License immediately upon written notice to us if we cease to carry on our business related to the MIT License, fail to maintain insurance as required under the MIT License, file for bankruptcy, fail to pay amounts due under the MIT License, challenge or assist others in bringing a challenge to MIT’s patents or fail to cure material breach within 60
days’ written notice thereof. Absent early termination, the MIT License will continue until the expiration or abandonment of the last to expire patent right subject to the MIT License.
MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. In December 2019,June 2020, we entered into the Fourtha Fifth Amendment to the MIT License, which we refer to asor the MIT Amendment.Amendment, which was effective as of May 15, 2020. Pursuant to the MIT Amendment, certain of our diligence obligations were extended to the provisionsecond quarter of the MIT License under which we were obligated2021, including a diligence obligation to initiatecommence a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specifiedspecific date in the second quarter of 2020 or (ii) the effective date2021. Additionally, certain of a written amendment to the MIT License. Further, pursuant to the MIT Amendment, the parties agreed to negotiate in good faith to enter into a future amendment to the MIT License subject to our providing MIT with an amended diligence plan.development and regulatory milestones and payments upon achievement of such milestones were adjusted.
Shenyang Sunshine Pharmaceutical Co., Ltd.
In May 2014, we entered into a license agreement with 3SBio, as amended in May 2017, which we refer to as the 3SBio License. Pursuant to the 3SBio License, we were granted an exclusive license to certain pegadricase-related patents and related “know-how”know-how owned or in-licensed by 3SBio for the worldwide (except for Greater China and Japan) development and commercialization of products based thereupon for human therapeutic, diagnostic and prophylactic use. We are also granted a worldwide (except for Greater China) exclusive license to develop, commercialize and manufacture or have manufactured products combining our proprietary ImmTOR platform with pegadricase or related compounds supplied by 3SBio (or otherwise supplied if our rights to manufacture are in effect) for human therapeutic, diagnostic and prophylactic use. We were also granted a co-exclusive license to manufacture and have manufactured pegadricase and related compounds for our preclinical and clinical use or, if the 3SBio License is terminated for 3SBio’s material breach, for any use under the 3SBio License. In addition, the 3SBio License, as amended, permits us to utilize one or more third parties to provide up to 20% of our commercial supply of pegadricase. Otherwise, except in the case of a supply shortage on the part of 3SBio, we are obligated to obtain at least 80% of our supply of such compounds for Phase 3 clinical trials and commercial use from 3SBio under the terms of supply agreements to be negotiated.our separate Commercial Supply Agreement with 3SBio dated August 1, 2019.
Under the 3SBio License we have paid to 3SBio an aggregate of $3.0$7.0 million in upfront and milestone-based payments. We are required to make future payments to 3SBio contingent upon the occurrence of events related to the achievement of clinical and regulatory approval milestones of up to an aggregate of $21.0$15.0 million for products containing our ImmTOR platform, and up to an aggregate of $41.5 million for products without our ImmTOR platform. We are also required to pay 3SBio tiered royalties on annual worldwide net sales related to the pegadricase component of products at percentages ranging from the low-to-mid single digits for products containing our ImmTOR platform, and from the mid-single digits to low or low-to-mid teens for products without our ImmTOR platform, subject to specified reductions. These royalties are payable, on a country-by-country and product-by-product basis until the later of (i) the date that all of the patent rights for that product have expired in that country or (ii) a specified number of years from the first commercial sale of such product in such country.
The 3SBio License expires on the date of expiration of all of our royalty payment obligations unless earlier terminated by either party for an uncured material default of the other party or for the other party’s bankruptcy. We may also terminate the 3SBio License on a country-by-country or product-by-product basis for any reason effective upon 60 days’ prior written notice to 3SBio or, with respect to a given product, immediately upon written notice to 3SBio if we identify a safety or efficacy concern related to such product.
BINDManufacturing
In December 2008,We manufacture ImmTOR using a scalable, self-assembly nanoemulsion process with well-defined, pharmaceutical unit operations. This proprietary, highly specialized and precisely controlled manufacturing process enables us to reproducibly manufacture ImmTOR across many production scales, from milligram-scale at the laboratory bench to hundreds of grams to
multi-kilogram scale for commercial production. We have also developed and executed the required detailed analytic characterization of our products.
For our most advanced product candidate, SEL-212, we entered intoare producing ImmTOR at an approximately 400-gram scale process, which, at the doses being tested in the Phase 3 DISSOLVE program, we believe will be suitable for commercialization. The process is designed such that this same equipment is capable of potentially producing up to a cross-license agreementone-kilogram batch size scale. As our nanoparticle manufacturing process is compact, and therefore also portable, our strategy is to transfer our custom designed process skids to a contract manufacturing organization, or CMO, and have the CMO produce the nanoparticles under our direction. This is the strategy we use for production of clinical supplies for clinical trials and would be the expected strategy for commercial production.
The pegadricase enzyme for SEL-212 is produced by fermentation in E. coli and is sourced from 3SBio in China. Through a licensing arrangement, we own exclusive worldwide rights to pegadricase outside of China, with BIND Therapeutics, Inc. (formerly BIND Biosciences, Inc.), or BIND, whichco-ownership of rights in Japan and with 3SBio owning all rights in China. Under this arrangement, 3SBio has agreed to supply pegadricase for the SEL-212 program. There is also a second supplier for pegadricase in the United States.
Our AAV vector product candidates are produced using the established triple transfection production process at CMOs. We work closely with our CMOs who have platform AAV production processes that we referare able to as the BIND Agreement. Pursuantleverage for our specific AAV product candidates.
Intellectual Property
Our ImmTOR platform is designed to deliver precise instructions to the BIND Agreement, BIND granted usimmune system as a perpetual, irrevocable, royalty-free worldwide non-exclusive license under certain of BIND’s existing and future patent rights to make, have made, use, sell, offer for sale and import products and services covered by such patents and patent applications in the field of certain prophylactic and therapeutic vaccines. The time period for adding new patent rights, not included in the families of previously licensed patent rights, to our license grant from BIND has expired. Pursuant to the BIND Agreement, we granted BIND a perpetual, irrevocable, royalty-free, worldwide non-exclusive license under certain of our current and future patent rights to make, have made, use, sell, offer for sale and import products and services covered by such patents and patent applications in all other fields, in each case, excluding certain future patent rights of each party related to novel targeting agents. The time period for adding new patent rights, not included in the families of previously licensed patent rights, to BIND’s license grant from us has expired.
We have paid BIND an upfront license issuance fee and reimbursed certain of BIND’s fees in connection with the entry into the BIND Agreement. No royalties or other payments are due to or by either party. The BIND Agreement expires upon expirationresult of the last patent right covered bynatural predisposition of the BIND Agreement. Neither party may unilaterally terminate the BIND Agreement for any reason. If either party materially breaches the BIND Agreement, failsimmune system to expend a specified amount in research and development activities related to the BIND Agreement, undergoes bankruptcy or insolvency, or undergoes a change of control,
the future patent rights to be included in the license grant to the party breaching, failing to expendinterrogate nanoparticles such amounts or undergoing such event under the BIND Agreement will no longer be granted to the breaching party.
as viruses. In connection with BIND’s entry into bankruptcy proceedingour founding, we licensed multiple patent families, including a patent family based in 2016, Pfizer purchased substantially all ofpart on the assets of BIND, including those pertainingpioneering research performed by our co-founders at Harvard University, MIT and Brigham. We have also conducted extensive research in-house to further research this area, leading to key discoveries regarding and uses for our ImmTOR technology. We have aggressively sought to extend and protect the BIND Agreement.
Massachusetts Eyeproprietary intellectual property underlying the composition and Ear Infirmary
In May 2016, we entered into a license agreement with the Massachusetts Eye and Ear Infirmary and The Schepens Eye Research Institute, Inc., or, collectively, MEE, which we refer to as the MEE License. Under the MEE License, we were granted an exclusive commercial worldwide license, with the right to grant sublicenses through multiple tiers, to make, have made, use, offer to sell, sell and import certain products and to practice certain processes, the sale, use or practice of which are covered by patents and proprietary know-how owned or controlled by MEE, for use of Anc80 gene therapy vectorsImmTOR, such as for gene augmentation therapies expressing certain target sequences. On September 16, 2019, in accordance with the terms of the MEE License, we notified MEE of our intention to terminate the MEE License, effective December 15, 2019.
INTELLECTUAL PROPERTYantigen-specific immunotolerance.
We endeavor to protect our nanoparticle technology, which we consider fundamental to our business, by seeking, maintaining and defending patent and other intellectual property rights, whether developed internally or licensed from third parties, relating to our program, product candidates, their methods of use and the processes for their manufacture. Our practice is to strive to protect our intellectual property by, among other methods, pursuing and obtaining patent protection in the United States and in jurisdictions outside of the United States related to our proprietary technology, inventions, improvements, programs and product candidates that are commercially important to the operation and growth of our business. We also rely on trade secrets and know-how relating to our proprietary technology, programs and product candidates, continuing innovation and continue innovating and seeking in-licensing opportunities to maintain, advance and fortify our proprietary position in our nanoparticle-based immunotherapy program and product candidates as well as to develop new programs and other product candidates. Our commercial success will depend in part on our ability to obtain and maintain patent and other proprietary protection for our program technology programs, inventions and improvements; to preserve the confidentiality of our trade secrets; to maintain our licenses to use intellectual property owned or controlled by third parties; to defend and enforce our proprietary rights, including our patents; and to operate without infringing the patents and proprietary rights of third parties.
We have developed and in-licensed numerous patents and patent applications and possess substantial know-how and trade secrets relating to our nanoparticle-based immunotherapy technology programprograms and product candidates. Our patent portfolio contains a number of issued patents in the United States and certain foreign jurisdictions. We also own a number of pending patent applications in the United States and certain foreign jurisdictions. These patents and patent applications include claims directedrelated to:
•tolerance immunotherapy programs;
immune stimulation programs;
•methods and compositions incorporatingrelated to our proprietary nanoparticles in a variety of applications, including tolerance applications, including:such as:
◦mitigating or treating anti-drug antibodies and/or their effects associated with protein drugs, such as for chronic refractory gout, coveringincluding coverage for ImmTOR co-administered with pegadricase, which related patents are expected to expire between 2032 and 2038,2041, and
◦genetic therapies (such as viral delivery of genes)vector gene therapy), coveringincluding coverage for ImmTOR co-administered with a viral vector, which related patents are expected to expire between 2032 and 2039;2042; and
•development and commercialization of SEL-212, including both composition of matter and method of treatment claims (there are multiple patent families with claims that cover the SEL-212 product, one of which is a licensed, issued U.S. patent that covers the SEL-212 product, which expiresexpired in August 2021).
In addition, we have exclusively or non-exclusively licensed intellectual property, including U.S. issued patents, foreign issued patents, and pending applications in both the U.S. and foreign jurisdictions. The licensed patents and patent applications cover various aspects of the technology being developed by us, including claims directed to compositions of matter and methods of use, and have been filed in various countries worldwide including in North America, Europe and Asia, with material expiration dates varying from 2021 to, if claims are issued, 2028.2042. In addition to filing and prosecuting patent applications in the United States, we often file analogous patent applications in the European Union and in additional foreign countries where we believe such filing is likely to be beneficial, including but not limited to, Australia, Brazil, Canada, China, Europe, South Korea, Mexico, India, Israel, and Japan.
Each patent’s term depends upon the laws of the countries in which they are obtained. The patent term in most countries in which we file is 20 years from the earliest date of filing of a non-provisional patent application. Notably, the term of U.S. patents may be extended due to delays incurred due to compliance with FDA or by delays encountered during prosecution that are caused by the USPTO. For example, the Hatch-Waxman Act permits a patent term extension for FDA-approved drugs of up to five years beyond the expiration of the patent, depending upon the length of time the drug is under regulatory review. There is a limit to the amount of time a patent may be extended in the United States; no patent extension can extend the remaining term of a patent beyond a total of 14 years from the date of product approval and only one patent applicable to an approved drug may be extended. Similar patent term extensions are available in Europe and other jurisdictions for patents that cover regulatory-approved drugs. Currently, we own or license patents and patent applications with expected material expiration dates ranging from 20212032 to 2039.2042. However, the actual patent protection period varies on a product-by-product basis, from country-to-country, and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent.
COMPETITIONCompetition
The biotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We face potential competition from many different sources, including pharmaceutical and biotechnology companies, academic institutions and governmental agencies and public and private research institutions. Product candidates that we successfully develop and commercialize may compete with existing therapies and new therapies that may become available in the future.
Our competitors may have significantly greater financial resources, established presence in the market, expertise in research and development, manufacturing, preclinical and clinical testing, obtaining regulatory approvals and reimbursement and marketing approved products than we do. These competitors also compete with us in recruiting and retaining qualified scientific, sales, marketing and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
The key competitive factors affecting the success of SEL-212, and any other tolerance or immune stimulationmodulation product candidates that we develop, if approved, are likely to be their efficacy, safety, convenience, price, the level of generic competition and the availability of reimbursement from government and other third-party payors.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours. In addition, our ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic or biosimilar products.
SEL-212 may compete with others in the gout market, including KRYSTEXXA,pegloticase, which contains a pegylated uricase similar to the pegadricase component of SEL-212 and is indicated for the treatment of refractory gout. Horizon Pharma plc, whose affiliates own KRYSTEXXA,pegloticase, may find other approaches to eliminate undesired immunogenicity to KRYSTEXXA.pegloticase. Long-term treatment with global immunosuppressive products may increase the susceptibility to contract infections, tumors and may lead to organ failure.
Large companies with active research to prevent the formation of ADAs and treat allergies or autoimmune diseases include Eli Lilly, Roche Holding AG, Sanofi S.A., Pfizer Inc., or Pfizer, and Merck & Co., Inc., or Merck. Small, early-stage biopharmaceutical companies active in the research for new technologies to induce antigen-specific immune tolerance include Anokion SA, AnTolRx Inc., Apitope International NV, Caladrius Biosciences, Cour Pharmaceutical Development Company, Inc., Cue Biopharma, Dendright International, Inc., Parvus Therapeutics, REGiMMUNE Corporation, Rubius Therapeutics, Inc., Tolerion, Inc., Topas Therapeutics GmbH, SQZ Biotechnologies and Txcell SA. However, we believe that mostBiopharmaceutical companies active in the research for MMA include LogicBio, Poseida, and Moderna. Biopharmaceutical companies active in the research for Ornithine transcarbamylase include Ultragenyx, Poseida, TranslateBio, Arcturus, and Kaleido. Biopharmaceutical companies active in the research for IgA nephropathy include Omeros, Travere, EMD Serono, Novartis, Ionis, Visterra, Reata, and Alnylam. Biopharmaceutical companies active in the PBC research include Intercept Pharmaceuticals, Genfit, GenKyoTex, GSK, Eli Lilly, and CymaBay. Biopharmaceutical companies active in the IL-2 research include Roche Holding AG, Amgen, Bristol Myers Squibb, Eli Lilly, and Moderna.
GOVERNMENT REGULATIONGovernment Regulation
Government authorities in the United States, at the federal, state and local level, and in other countries extensively regulate, among other things, the research, development, testing, manufacturing, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, post-approval monitoring and reporting, marketing and export and import of products such as those we are developing.
Our most advanced product candidate, SEL-212 is subject to regulation in the United States as a combination product. If marketed individually, each component would be subject to different regulatory pathways and would require approval of independent marketing applications by the FDA. A combination product, however, is assigned to a Center that will have primary jurisdiction over its regulation based on a determination of the combination product’s primary mode of action, which is the single mode of action that provides the most important therapeutic action. In the case of our SEL-212, we believe that the
primary mode of action is attributable to the biologic component of the product. In the case of SEL-212, which we believe will be regulated as a therapeutic biologic, the FDA’s Center for Drug Evaluation and Research, or CDER, will have primary jurisdiction over premarket development. We expect to seek approval of SEL-212 through a single Biologics License Application, or BLA reviewed by CDER, and we do not expect that the FDA will require a separate marketing authorization for each constituent of SEL-212.
Biological products are subject to regulation under the Federal Food, Drug, and Cosmetic Act, or FD&C Act, and the Public Health Service Act, or PHS Act, and other federal, state, local and foreign statutes and regulations. SEL-212 and any other product candidates that we develop must be approved by the FDA before they may be legally marketed in the United States and by the appropriate foreign regulatory agency before they may be legally marketed in foreign countries.
U.S. biological products development process
The process required by the FDA before a biologic, including a gene therapy, may be marketed in the United States generally involves the following:is summarized below.
completion of extensive nonclinicalBiological product candidates are preclinically tested before any testing sometimes referred to as preclinical testing, including laboratoryis done in humans. These tests, animal trials and formulation studies in accordance with applicable regulations, including good laboratory practices, or GLPs, and applicable requirements for humane use of laboratory animals;
submission to the FDA of an IND, which must become effective before human clinical trials may begin;
performance of adequate and well-controlled human clinical trials according to good clinical practice, or GCP, regulations and any additional requirements for the protection of human research subjects and their health information, to establish the safety, purity and potency of the proposed biological product for its intended use;
submission to the FDA of a BLA for marketing approval that includes substantive evidence of safety, purity, and potency from results of nonclinical testing and clinical trials;
satisfactory completion of an FDA inspection of the manufacturing facility or facilities where the biological product is produced to assess compliance with cGMP requirements to assure that the facilities, methods and controls are adequate to preserve the biological product’s identity, strength, quality and purity;
potential FDA audit of the nonclinical and clinical study sites that generated the data in support of the BLA; and
FDA review and approval, or licensure, of the BLA.
Before testing any biological product candidate in humans, the product candidate enters the preclinical testing stage. Preclinical tests, also referred to as nonclinicalnon-clinical studies, include laboratory evaluations of product chemistry, toxicity and formulation, as well as animal studies to assess the potential safety and activity of the product candidate. The conduct of the preclinical tests must comply with federal regulations and requirements including GLPs.
The clinical study sponsor must submit the results of the preclinical tests, together with manufacturing information, analytical data, any available clinical data or literature and a proposed clinical protocol, to the FDA as part of the IND. Some preclinical testingIND which must become effective before human clinical trials may continue even after the IND is submitted.begin. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA places the clinical study on a clinical hold within that 30-day time period. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical study can begin. The FDA may also impose clinical holds on a biological product candidate at any time before or during clinical trials due to safety concerns or non-compliance. If the FDA imposes a clinical hold, trials may not recommence without FDA authorization and then only under terms authorized by the FDA.
In addition to the submission of an IND to the FDA, supervision of certain human gene transfer trialsthese requirements, biological product candidates may also require evaluation and assessment by an institutional biosafety committee, or IBC, a local institutional committee that reviews and oversees research utilizing recombinant or synthetic nucleic acid molecules at that institution. The IBC assesses the safety of the research and identifies any potential risk to public health or the environment, and such review may result in some delay before initiation of a clinical trial.
Clinical trials involve the administration of the biological product candidate to healthy volunteers or patients under the supervision of qualified investigators, generally physicians not employed by or under the study sponsor’s control. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical study, dosing procedures, patient selection and exclusion criteria, and the parameters to be used to monitor patient safety, including stopping rules that assure a clinical study will be stopped if certain adverse events should occur.safety. Each protocol and any amendments to the protocol must be submitted to the FDA as part of the IND. Clinical trials must be conducted and monitored in accordance with the FDA’s regulations comprising the GCP requirements, including the requirement that all research patientssubjects provide informed consent. Further, each clinical study must be reviewed and approved by an independent institutional review board, or IRB, at or
servicing each institution at which the clinical study will be conducted. Human clinical trials are typically conducted in three sequential phases that may overlap or be combined:
| |
• | Phase I. •Phase 1.The biological product candidate is initially introduced into healthy human patients and tested for safety. •Phase 2.The biological product candidate is evaluated in a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance, optimal dosage and dosing schedule. •Phase 3.The biological product candidate is initially introduced into healthy human patients and tested for safety. In the case of some products for severe or life-threatening diseases, especially when the product may be too inherently toxic to ethically administer to healthy volunteers, the initial human testing is often conducted in patients. |
| |
• | Phase II. The biological product candidate is evaluated in a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance, optimal dosage and dosing schedule.
|
| |
• | Phase III. Clinical trials are undertaken to further evaluate dosage, clinical efficacy, potency, and safety in an expanded patient population at geographically dispersed clinical study sites. These clinical trials are intended to establish the overall risk/benefit ratio of the product and provide an adequate basis for product labeling.
|
Post-approval clinical trials, sometimes referred to as Phase IV4 clinical trials, may be conducted after initial marketing approval. These clinical trials are usedapproval to gain additional experience from the treatment of patients in the intended therapeutic indication, particularly for long-term safety follow-up.
The FDA or the sponsor or its data safety monitoring board may suspend a clinical study at any time on various grounds, including a finding that the research patients or patients are being exposed to an unacceptable health risk.grounds. Similarly, an IRB can suspend or terminate approval of a clinical study at its institution if the clinical study is not being conducted in accordance with the IRB’s requirements or if the biological product candidate has been associated with unexpected serious harm to patients.
There are also requirements governing the reporting of ongoing clinical trials and completed clinical trial results to public registries. Sponsors of clinical trials of FDA-regulated products, including biologics, are also required to register and disclose certain clinical trial information, which is publicly available at www.clinicaltrials.gov. Concurrent with clinical trials, companies usually complete additional animal trials and must also develop additional information about the physical characteristics of the biological product candidate as well as finalize a process for manufacturing the product in commercial quantities in accordance with GMP requirements.
U.S. review and approval processes
After the completion of clinical trials of a biological product candidate, FDA approval of a BLA must be obtained before commercial marketing of the biological product. The BLA must include results of product development, laboratory and animal trials, human trials, information on the manufacture and composition of the product, proposed labeling and other relevant information. In addition, under the Pediatric Research Equity Act, or PREA, a BLA or supplement to a BLA must contain data to assess the safety and effectiveness of the biological product candidate for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The Food and Drug Administration Safety and Innovation Act, or FDASIA, requires that a sponsor who issponsors planning to submit a marketing applicationBLA for a drug or biological product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administrationin certain circumstances n submit an initial Pediatric Study Plan, or PSP, within sixty days after a Type C meeting or as may be agreed between the sponsor and FDA. Unless otherwise required by regulation, PREA does not apply to any biological product for an indication for which orphan designation has been granted.
Under the Prescription Drug Fee User Fee Act, or PDUFA, as amended, each BLA must be accompanied by a user fee. The FDA adjusts the PDUFA user fees on an annual basis. Fee waiverswaiver or reductions are available inunder certain circumstances, including a waiver of the application fee for the first application filed by a small business. Additionally,In addition, no user fees are assessed on BLAs foron products designated as orphan drugs unless the product also includes a non-orphan indication.
Within 60 days following submission of the application, the FDA reviews a BLA submitted to determine if it is substantially complete before the agency accepts it for filing.completeness. The FDA may refuse to file any BLA that it deems incomplete or otherwise not properly reviewable at the time of submission and may request additional information. In this event, the BLA must be resubmitted with the additional information. Once the submission is accepted for filing, the FDA begins an in-depth substantive review of the BLA. The FDAsubstantively reviews the BLA to determine, among other things, whether the proposed product is safe, pure and potent, for its intended use and whether the product is being manufactured in accordance with cGMP requirements to assure and preserve the product’s identity, safety, strength, quality, potency and purity.requirements. The FDA may refer applications for novel biological products or biological products that present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a non-binding recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.on approval. During the biological product approval process, the FDA also will determine whether a Risk Evaluation and Mitigation Strategy, or REMS, is necessary to assure the safe use of the biological
product candidate. If the FDA concludes a REMS is needed, the sponsor of the BLA must submit a proposed REMS; the FDA will not approve the BLA without a REMS, if required.
Before approving a BLA, the FDA will inspect the facilities atin which the product is manufactured. The FDA will not approve the product unless it determines thatmanufactured to determine whether the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production ofGMPs. The FDA may also audit the product within required specifications. Additionally, before approving a BLA, the FDA will typically inspect one or more clinical investigation sites to assuredetermine that thethey have complied with good clinical trials were conducted in compliance with IND study requirements and GCP requirements.practices.
Notwithstanding the submission of relevant data and information, the FDA may ultimately decide that the BLA does not satisfy its regulatory criteria for approval and deny approval. Data obtained from clinical trials are not always conclusive and the FDA may interpret data differently than the applicant interprets the same data. If the FDA decides not to approve thedenies approval of a BLA in its presentthen-current form, the FDA will issue a complete response letter that usually describes all of the specificdetailing deficiencies in the BLA identified by the FDA. Additionally, the complete response letter may include recommended actions that the applicant might take to place the application in a condition for approval.application. If a complete response letter is issued, the applicant may either resubmit the BLA, addressing all of the deficiencies identified in the letter, or withdraw the application.
If a product receives regulatory approval, the approval may be significantly limited to specific diseases and dosages or the indications for use may otherwise be limited, which could restrict the commercial value of the product. Further, the FDA may require that certain contraindications, warnings or precautions be included in the product labeling. The FDA may impose restrictions and conditions on product distribution, prescribing, or dispensing in the form of a REMS, or otherwise limit the scope of any approval. In addition, the FDA may require post marketing clinical trials, sometimes referred to as Phase IV clinical trials, designed to further assess a biological product’s safety and effectiveness, and testing and surveillance programs to monitor the safety of approved products that have been commercialized.
One of the performance goals agreed to by the FDA under the PDUFA is to review 90% of standard BLAs in 10 months from the filing date and 90% of priority BLAs in six months from the filing date, whereupon a review decision is to be made. The FDA does not always meet its PDUFA goal dates for standard and priority BLAs.
Orphan designationDesignation
ThePrior to the submission of a BLA, the FDA may grant orphan designation to drugs or biologics intended to treat a rare disease or condition that affects fewer than 200,000 individuals in the United States, or if it affects more than 200,000 individuals in the United States, there is no reasonable expectation that the cost of developing and marketing the product for this type of disease or condition will be recovered from sales in the United States. Orphan designation must be requested before submitting a BLA.States After the FDA grants orphan designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. Orphan designation does not convey any advantage in or shorten the duration of the regulatory review and approval process.
In the United States, orphan designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages and user-fee waivers. In addition, if a product receives the first FDA approval for the indication for which it has orphan designation, the product is entitled to orphan exclusivity, which means the FDA may not approve any other application to market the same product for the same indication for a period of seven years, except in limited circumstances, such as a showing of clinical superiority over the product with orphan exclusivity or where the manufacturer with orphan exclusivity is unable to assure sufficient quantities of the approvedapprove orphan designated product. Competitors, however, may
receive approval of different products for the indication for which the orphan product has exclusivity or obtain approval for the same product but for a different indication for which the orphan product has exclusivity. Orphan product exclusivity also could block the approval of one of our products for seven years if a competitor obtains approval of the same biological product as defined by the FDA or if our product candidate is determined to be contained within the competitor’s product for the same indication or disease. If a drug or biological product designated as an orphan product receives marketing approval for an indication broader than what is designated, it may not be entitled to orphan product exclusivity.
We have not requested orphanRare Pediatric Disease Designation
The Rare Pediatric Disease designation program allows for oura sponsor who receives an approval for a product candidates, but depending on the proposed indicationto potentially qualify for which we intenda voucher that can be redeemed to develop our future products, we may in the future request such designation.receive a priority review of a subsequent marketing application for a different product.
Expedited developmentDevelopment and review programsReview Programs
The FDA has a Fast Track program that is intended to expedite or facilitate the process for reviewing new biological products that meet certain criteria. Specifically, new biological products are eligible for Fast Track designation if they are intended to treat a serious or life-threatening disease or condition and demonstrate the potential to address unmet medical needs for the disease or condition. Fast Track designation applies to the combination of the product and the specific indication for which it is being studied. The sponsor of a new biologic may request that the FDA designate the biologic as a Fast Track product at any time during the clinical development of the product. The FDA may consider for review sections of the marketing application for a Fast Track product on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the
submission of the sections of the application, the FDA agrees to accept sections of the application and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application.
Any product submitted to the FDA for marketing, including under a Fast Track program, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. Any product is eligible for priority review if it treats a serious condition and, if approved, would provide a significant improvement in safety or effectiveness compared to marketed products. The FDA will attempt to direct additional resources to the evaluation of an application for a new biological product designated for priority review in an effort to facilitate the review. Additionally, a product may be eligible for accelerated approval. Biological products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may be eligible for accelerated approval, which means that they may be approved on the basis of adequate and well-controlled clinical studies establishing that the product has an effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit, or on the basis of an effect on a clinical endpoint other than survival or irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require that a sponsor of a biological product subject to accelerated approval perform adequate and well-controlled post-marketing clinical studies.studies to confirm such benefit. In addition, the FDA currently requires as a condition for accelerated approval pre-approval of promotional materials, which could adversely impact the timing of the commercial launch of the product. Fast Track designation, priority review and accelerated approval do not change the standards for approval but may expedite the development or approval process.
In addition, under the provisions of FDASIA, the FDA established a Breakthrough Therapy Designation which is intended to expedite the development and review of products that treat serious or life-threatening diseases or conditions. A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The designation includes all of the features of Fast Track designation, as well as more intensive FDA interaction and guidance. The Breakthrough Therapy Designation is a distinct status from both accelerated approval and priority review, but these can also be granted to the same product candidate if the relevant criteria are met. The FDA must take certain actions, such as holding timely meetings and providing advice, intended to expedite the development and review of an application for approval of a breakthrough therapy. Requests for breakthrough therapy designation will be reviewed within 60 days of receipt, and FDA will either grant or deny the request.
Fast Track designation, priority review, accelerated approval and breakthrough therapy designation do not change the standards for approval but may expedite the development or approval process. Even if we receive one of these designations for our product candidates, the FDA may later decide that our product candidates no longer meet the conditions for qualification. In addition, these designations may not provide us with a material commercial advantage.
Post-approval requirementsRequirements
Maintaining substantial compliance with applicable federal, state and local statutes and regulations requires the expenditure of substantial time and financial resources. Rigorous and extensive FDA regulation of biological products continues after approval, particularly with respect to cGMP requirements. Manufacturers of our products are required to comply with applicable requirements in the cGMP regulations, including quality control and quality assurance and maintenance of records and documentation. Other post-approval requirements applicable to biological products include record-keeping requirements, reporting of adverse effects and reporting updated safety and efficacy information.
We also must comply with the FDA’s advertising and promotion requirements, such as those related to direct-to-consumer advertising, the prohibition on promoting products for uses or in patient populations that are not described in the product’s approved labeling (known as “off-label use”), industry-sponsored scientific and educational activities and promotional activities involving the internet. Discovery of previously unknown problems or the failure to comply with the applicable regulatory requirements may result in restrictions on the marketing of a product or withdrawal of the product from the market as well as possible civil or criminal sanctions. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or after approval, may subject an applicant or manufacturer to administrative or judicial civil or criminal sanctions and adverse publicity. FDA sanctions could include refusal to approve pending applications, withdrawal of an approval, clinical hold, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, mandated corrective advertising or communications with doctors, debarment, restitution, disgorgement of profits, or civil or criminal penalties.
Biological product manufacturers and other entities involved in the manufacture and distribution of approved biological products are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP requirements and other laws. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to
maintain cGMP compliance. In addition, changes to the manufacturing process or facility generally require prior FDA approval before being implemented and other types of changes to the approved product, such as adding new indications and additional labeling claims, are also subject to further FDA review and approval.
Biosimilars and exclusivityExclusivity
The Patient Protection and Affordable Care Act, or ACA, signed into law on March 23, 2010, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, or BPCIA, which created an abbreviated approval pathway for biological products that are biosimilar to or interchangeable with an FDA-licensed reference biological product.
Biosimilarity, which requires that there be no clinically meaningful differences between the biological product and the reference product in terms of safety, purity and potency, can be shown through analytical studies, animal studies and a clinical study or studies. Interchangeability requires that a product is biosimilar to the reference product and the product must demonstrate that it can be expected to produce the same clinical results as the reference product in any given patient and, for products that are administered multiple times to an individual, the biologic and the reference biologic may be alternated or switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic. However, complexities associated with the larger, and often more complex, structures of biological products, as well as the processes by which such products are manufactured, pose significant hurdles to implementation of the abbreviated approval pathway that are still being worked out by the FDA.
Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date that the reference product was first licensed by the FDA. In addition, the approval of a biosimilar product may not be made effective by the FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product. The BPCIA also created certain exclusivity periods for certain biosimilars approved as interchangeable products. At this juncture, it is unclear whether products deemed “interchangeable” by the FDA will, in fact, be readily substituted by pharmacies, which are governed by state pharmacy law.
A biological product can also obtain pediatric market exclusivity in the United States. Pediatric exclusivity, if granted, adds six months to existing exclusivity periods and patent terms. This six-month exclusivity, which runs from the end of other exclusivity protection or patent term, may be granted based on the voluntary completion of a pediatric study in accordance with an FDA-issued "Written Request"“Written Request” for such a study.
The BPCIA is complex and continues to be interpreted and implemented by the FDA. As a result, the ultimate impact, implementation and meaning of the BPCIA is subject to significant uncertainty.
Government regulationRegulation outside of the United States
In addition to regulations in the United States, we will be subject to a variety of regulations in other jurisdictions governing, among other things, clinical studies and any commercial sales and distribution of our products. Because biologically sourced raw materials are subject to unique contamination risks, their use may be restricted in some countries.
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in foreign countries prior to the commencement of clinical studies or marketing of the product in those countries. Certain countries outside of the United States have a similar process that requires the submission of a clinical study application much like the IND prior to the commencement of human clinical studies. In the European Union, for example, a clinical trial authorization, or CTA, must be submitted to each country’s national health authority and an independent ethics committee, much like the FDA and the IRB, respectively. Once the CTA is approved in accordance with a country’s requirements, clinical study development may proceed.
The requirements and process governing the conduct of clinical studies, product licensing, pricing and reimbursement vary from country to country. In all cases, the clinical studies are conducted in accordance with GCP and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
In the European Economic Area, or EEA, which is composed of the 27 Member Statesmember states of the European Union plus Norway, Iceland and Liechtenstein and the United Kingdom (until the end of the transition period on 31 December 2020 provided for in Withdrawal Agreement between the EU and the UK) medicinal products can only be commercialized after obtaining a Marketing Authorization, or MA.
There are two types of MAs.
•The Community MA, which is issued by the European Commission through the Centralized Procedure, based on the opinion of the Committee for Medicinal Products for Human Use, or CHMP, of the European Medicines Agency, or EMA, and which is valid throughout the entire territory of the EEA. The Centralized Procedure is mandatory for
certain types of products, such as biotechnology medicinal products, orphan medicinal products, advanced therapy medicinal products (such as gene therapy, somatic cell therapy and tissue engineered products), and medicinal products that contain a new active substance indicated for the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, auto-immune and viral diseases.among others. The Centralized Procedure is optional for products containing a new active substance not yet authorized in the EEA, or for products that constitute a significant therapeutic, scientific or technical innovation or which are in the interest of public health in the EU. Under the Centralized Procedure the maximum timeframe for the evaluation of a marketing authorization application is 210 days (excluding clock stops, when additional written or oral information is to be provided by the applicant in response to questions asked by the CHMP). Accelerated evaluation might be granted by the CHMP in exceptional cases, when the authorization of a medicinal product is of major interest from the point of view of public health and in particular from the viewpoint of therapeutic innovation.cases. Under the accelerated procedure the standard 210 days review period is reduced to 150 days.
•National MAs, which are issued by the competent authorities of the Member States of the EEA and only cover their respective territory, are available for products not falling within the mandatory scope of the Centralized Procedure. Where a product has already been authorized for marketing in a Member State of the EEA, this National MA can be recognized in another Member States through the Mutual Recognition Procedure. If the product has not received a National MA in any Member State at the time of application, it can be approved simultaneously in various Member States through the Decentralized Procedure.
To obtain regulatory approval of an investigational biological product under European Union regulatory systems, we must submit a marketing authorization application, which is similar to the U.S. BLA. The European Union also provides opportunities for market exclusivity. For example, in the European Union, uponUpon receiving marketing authorization, new chemical entities generally receive eight years of data exclusivity, and an additional two years of market exclusivity. If granted, data exclusivitywhich prevents regulatory authorities in the European Union from referencing the innovator’s data to assess a generic or biosimilar application. During theapplication, and an additional two-year periodtwo years of market exclusivity, a generic marketing authorization can be submitted, and the innovator’s data may be referenced, butduring which no generic or biosimilar product can be marketed until the expiration of the market exclusivity.marketed. However, there is no guarantee that a product will be considered by the European Union’s regulatory authorities to be a new chemical entity, and products may not qualify for data exclusivity. Products receiving orphan designation in the European Union can receive ten years of market exclusivity, during which time no marketing authorization application shall be accepted, and no marketing authorization shall be granted for a similar medicinal product for the same indication. An orphan product can also obtain an additional two years of market exclusivity in the European Union for pediatric
studies. No extension to any supplementary protection certificate can be granted on the basis of pediatric studies for orphan indications.
The criteria for designating an “orphan medicinal product” in the European Union are similar in principle to those in the United States. Under Article 3 of Regulation (EC) 141/2000, a medicinal product may be designated as orphan if (1) it is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition; (2) either (a) such condition affects no more than five in 10,000 persons in the European Union when the application is made, or (b) the product, without the benefits derived from orphan status, would not generate sufficient return in the European Union to justify investment; and (3) there exists no satisfactory method of diagnosis, prevention or treatment of such condition authorized for marketing in the European Union, or if such a method exists, the product will be of significant benefit to those affected by the condition, as defined in Regulation (EC) 847/2000. Orphan medicinal products are eligible for certain financial incentives such as reduction of fees or fee waivers and are, upon grant of a marketing authorization, entitled to ten years of market exclusivity for the approved therapeutic indication. The application for orphan drug designation must be submitted before the application for marketing authorization. The applicant will receive a fee reduction for the marketing authorization application if the orphan drug designation has been granted, but not if the designation is still pending at the time the marketing authorization is submitted. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process.
The 10-year market exclusivity may be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation, for example, if the product is sufficiently profitable not to justify maintenance of market exclusivity. Additionally, marketing authorization may be granted to a similar product for the same indication at any time if the:
second applicant can establish that its product, although similar, is safer, more effective or otherwise clinically superior;
applicant consents to a second orphan medicinal product application; or
applicant cannot supply enough orphan medicinal product.incentives.
For other countries outside of the European Union, such as countries in Eastern Europe, Latin America or Asia, the requirements governing the conduct of clinical studies, product licensing, pricing and reimbursement vary from country to
country. In all cases, again, the clinical studies are conducted in accordance with GCP and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
When conducting clinical trials in the EU, we must adhere to the provisions of the European Union Clinical Trials Directive (Directive 2001/20/EC) and the laws and regulations of the EU Member States implementing them. These provisions require, among other things, that the prior authorization of an Ethics Committee and the competent Member State authority is obtained before commencing the clinical trial. In April 2014, the EU passed the Clinical Trials Regulation (Regulation 536/2014), which will replace the current Clinical Trials Directive. ToDirective, to ensure that the rules for clinical trials are identical throughout the European Union, the EU Clinical Trials Regulation was passed as a regulation that is directly applicable in all EU member states. All clinical trials performed in the European Union are required to be conducted in accordance with the Clinical Trials Directive until the Clinical Trials Regulation becomes applicable. According to the current plans of the EMA, the Clinical Trials Regulation is expected to become applicable in 2020.Union.
We are also subject to data privacy and security laws in the jurisdictions outside of the U.S. in which we are established, run clinical trials or in which we sell or market our products once approved. For example, in Europe we are subject to Regulation (EU) 2016/679 (General Data Protection Regulation or GDPR) in relation to our collection, control, processing and other use of personal data (i.e. data relating to an identifiable living individual). We process personal data in relation to participants in our clinical trials in the EEA., including the health and medical information of these participants. The GDPR is directly applicable in each E.U. Member State, however, it provides that E.U. Member States may introduce further conditions, including limitations which could limit our ability to collect, use and share personal data (including health and medical information), or could cause our compliance costs to increase, ultimately having an adverse impact on our business. The GDPR imposes onerous accountability and transparency obligations requiring data controllers and processors to maintain a record of their data processing and implement policies as part of its mandated privacy governance framework. It also requires data controllers to be transparent and disclose to data subjects (in a concise, intelligible and easily accessible form) how theirregarding personal information is to be used, imposes limitations on retention of personal data; defines for the first time pseudonymized (i.e., key-coded) data; introduces mandatory data breach notification requirements; and sets higher standards for data controllers to demonstrate that they have obtained valid consent for certain data processing activities.data. We are also subject to E.U. rules with respect to cross-border transfers of personal data out of the E.U. and EEA. We are subject to the supervision of local data protection authorities in those E.U. jurisdictions where we are established or otherwise subject to the GDPR. Fines for certain breaches of the GDPR are significant: up to the greater of EUR 20 million or 4% of total global annual turnover. In addition to the foregoing, aA breach of the GDPR could result in significant fines, regulatory investigations, reputational damage, orders to cease/ change our use of data, enforcement notices, as well potential civil claims including class action type litigation where individuals suffer harm. Moreover, the United Kingdom leaving the E.U. could also lead to further legislative and regulatory changes. It remains unclear how the United Kingdom data protection laws or regulations will develop in the medium to longer term and how data transfer to the United Kingdom from the E.U. will be regulated, especially following the United Kingdom's departure from the E.U. on January 31, 2020 without a deal.regulated. However, the United Kingdom has transposed the GDPR into domestic law with the Data Protection Act 2018, which remains in force following the United Kingdom'sKingdom’s departure from the EU.
If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Other healthcare laws
In addition to FDA restrictions on marketing of pharmaceutical and biological products, other U.S. federal and state healthcare regulatory laws restrict business practices in the biopharmaceutical industry, which include, but are not limited to, state and federal anti-kickback, false claims, data privacy and security, and physician payment and drug pricing transparency laws.Healthcare Laws
The federal Anti-Kickback Statute prohibits, among other things, any person or entity from knowingly (regardless of knowledge of this specific statute) and willfully offering, paying, soliciting, receiving or providing any remuneration, directly or indirectly, overtly or covertly, to induce or in return for purchasing, leasing, ordering, or arranging for or recommending the purchase, lease, or order of any item or service reimbursable, in whole or in part, under Medicare, Medicaid or other federal healthcare programs. The term “remuneration” has been broadly interpreted to include anything of value. The Anti-Kickback Statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers and formulary managers on the other. In addition, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. The majority of states also similar have anti-kickback laws, which establish similar prohibitions and in some cases may apply to items or services reimbursed by any third-party payor, including commercial insurers.are more restrictive.
The federal false claims and civil monetary penalties laws, including the civil False Claims Act, prohibit any person or entity from, among other things, knowingly presenting, or causing to be presented, a false, fictitious or fraudulent claim for payment to, or approval by, the federal government or knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or from knowingly making a false statement to
avoid, decrease or conceal an obligation. A claim includes “any request or demand” for money or property presented to the U.S. government. Violation of the federal Anti-Kickback Statute may also constitute a false or fraudulent claim for purposes of the federal civil False Claims Act. Actions under the civil False Claims Act may be brought by the Attorney GeneralDepartment of Justice or as a qui tam action by a private individual in the name of the government. Violations of the civil False Claims Act can result in very significant monetary penalties and treble damages. In addition, thedamages, and may be accompanied by additional civil monetary penalties statute imposes penalties against any person who is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent.
individuals. Many states also have similar fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Given the significant size of actual and potential settlements, it is expected that the government authorities will continue to devote substantial resources to investigating healthcare providers’ and manufacturers’ compliance with applicable fraud and abuse laws.
The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, prohibits, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
In addition, there has been a recent trend of increased federal and state regulation of payments made to physicians and certain other healthcare providers. The ACA imposed, among other things, new annual reporting requirements through the Physician Payments Sunshine Act for covered manufacturers for certain payments and “transfers of value” provided to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain other health care professionals beginning in 2022, and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Failure to submit timely, accurately and completely the required information for all payments, transfers of value and ownership or investment interests may result in civil monetary penalties. In addition, certain states require implementation of compliance programs and compliance with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, impose restrictions on marketing practices, and/or tracking and reporting of pricing information and marketing expenditure as well as gifts, compensation and other remuneration or items of value provided to physicians and other healthcare professionals and entities.
Violation of any of such laws or any other governmental regulations that may apply to us can result in penalties, including, without limitation, administrative, civil and criminal penalties, damages, fines, disgorgement, contractual damages, reputational harm, diminished profits and future earnings, the curtailment or restructuring of our operations, exclusion from participation in federal and state healthcare programs and individual imprisonment.
To the extent that any of our product candidates, once approved, are sold in a foreign country, we may be subject to similar foreign laws and regulations, which may include, for instance, applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws, and implementation of corporate compliance programs and reporting of payments or other transfers of value to healthcare professionals.
We may also be subject to data privacy and security regulation by both the federal government and the states in which we conduct our business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their respective implementing regulations, impose specified requirements relating to the privacy, security and transmission of individually identifiable health information held by covered entities and their business associates. Among other things, HITECH made HIPAA’s security standards directly applicable to, as well as imposed certain other privacy obligations on, “business associates,” defined as independent contractors or agents of covered entities that create, receive, maintain or transmit protected health information in connection with providing a service for or on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions. Even when HIPAA does not apply, according to the Federal Trade Commission or the FTC, failing to take appropriate steps to keep consumers’ personal information secure constitutes unfair acts or practices in or affecting commerce in violation of Section 5(a) of the Federal Trade Commission Act, or the FTCA, 15 U.S.C § 45(a). The FTC expects a company’s data security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities. Individually identifiable health information is considered sensitive data that merits stronger safeguards. The FTC’s guidance for appropriately securing consumers’ personal information is similar to what is required by the HIPAA Security Rule.
In addition, certain state laws govern the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same requirements, thus complicating compliance efforts. By
way of example, the California Consumer Privacy Act, or CCPA, which went into effect January 1, 2020, among other things, creates new data privacy obligations for covered companies and provides new privacy rights to California residents, including the right to opt out of certain disclosures of their information. The CCPA also creates a private right of action with statutory damages for certain data breaches, thereby potentially increasing risks associated with a data breach. Although the law includes limited exceptions, including for “protected health information” maintained by a covered entity or business associate, it may regulate or impact our processing of personal information depending on the context.
Coverage and reimbursementReimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any pharmaceutical or biological products for which we obtain regulatory approval. In the United States and markets in other countries, patients who are prescribed treatments for their conditions and providers performing the prescribed services generally rely on third-party payors to reimburse all or part of the associated healthcare costs. Patients are unlikely to use our products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of our products. Moreover, for drugs and biologics administered under the supervision of a physician, obtaining coverage and adequate reimbursement may be particularly difficult because of the higher prices often associated with such products. Sales of any products for which we receive regulatory approval for commercial sale will therefore depend, in part, on the availability of coverage and adequate reimbursement from third-party payors. Third-party payors include government authorities, managed care plans, private health insurers and other organizations.
The process for determining whether a third-party payor will provide coverage for a pharmaceutical or biological product typically is separate from the process for setting the price of such product or for establishing the reimbursement rate that the payor will pay for the product once coverage is approved. Third-party payors may limit coverage to specific products on an approved list, also known as a formulary, which might not include all of the FDA-approved products for a particular indication. A decision by a third-party payor not to cover our product candidates could reduce physician utilization of our products once approved and have a material adverse effect on our sales, results of operations and financial condition. Moreover, a third-party payor’s decision to provide coverage for a pharmaceutical or biological product does not imply that an adequate reimbursement rate will be approved. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development. Additionally, coverage and reimbursement for products can differ significantly from payor to payor. One third-party payor’s decision to cover a particular medical product or service does not ensure that other payors will also provide coverage for the medical product or service, or will provide coverage at an adequate reimbursement rate.
The containment of healthcare costs is a priority of federal, state and foreign governments, and the prices of pharmaceutical or biological products have been a focus in this effort. Third-party payors are increasingly challenging the prices charged for medical products and services, examining the medical necessity and reviewing the cost-effectiveness of pharmaceutical or biological products, medical devices and medical services, in addition to questioning safety and efficacy. If these third-party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products after FDA approval or, if they do, the level of payment may not be sufficient to allow us to sell our products at a profit.
Healthcare reformReform
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medical products. For example, the ACA, among other things, increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program; introduced a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care plans; subjected drug manufacturers to new annual fees based on pharmaceutical companies’ share of sales to federal healthcare programs; and created a new Patient Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research.
Since its enactment, there have been judicial and Congressional challenges to certain aspects of the ACA. By way of example, the Tax Cuts and Jobs Acts was enacted, which, among other things, removes penalties for not complying with the individual mandate to carry health insurance. Further, on December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, ruled that the individual mandate is a critical and inseverable feature of the ACA, and therefore, because it was repealed as part of the Tax Cuts and Jobs Acts, the remaining provisions of the ACA are invalid as well. On December 18, 2019, the U.S. Court of Appeals for the 5th Circuit ruled that the individual mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining provisions of the ACA are invalid as well. It is unclear how these decisions, subsequent appeals, if any, and other efforts to challenge, repeal or replace the ACA will impact the law.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA was enacted. This included aggregate reductions of Medicare payments to providers of 2% per fiscal year, which went into effect on April 1, 2013,
and, due to subsequent legislative amendments, will stay in effect through 2027 unless additional Congressional action is taken. On January 2, 2013, the American Taxpayer Relief Act was signed into law, which, among other things, further reduced Medicare payments to several providers, including hospitals and imaging centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. Recently there has also been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several Congressional inquiries and proposed and enacted legislation designed to, among other things, bring more transparency
to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies.
We expect that additional stateHuman Capital Resources
At Selecta Biosciences, we consider human capital to be an essential driver of our business and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federalsuccessful strategy creation and state governments will payexecution. Our people, driven by our Collaborative, Pioneering, and Patient Focused culture, propel our business forward, strengthening us for healthcare products and services, which could result in reduced demand for our products once approved or additional pricing pressures. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our drugs.
Employeeslong-term success.
As of JanuaryDecember 31, 2020,2021, we had 39 full-time58 employees, 2744 of whom wereare primarily engaged in research and development activities. A total of 12 employees have either one or both an M.D. or Ph.D. degree. Noneactivities and 14 in corporate functions. 66% of our employees have at least one of a Masters, PhD, or MD degree. Our 2021 annualized voluntary turnover rate was 12.87%. All employees reside and work in the United States and are not represented by a labor union and weunion. We consider our employee relations to be good.strong and in good standing.
Corporate InformationOur goal is to continually engage our talented and diverse workforce to drive value creation both for our business and ultimately our patient populations. We believe in a proactive approach to talent management focusing on retention of key talent, critical role successor identification, and impactful employment development. Additional priority areas intended to drive engagement include successful recruitment of diverse talent, continual promotion of professional development at all levels, introduction, and evolution of business-friendly HR solutions, coupled with an intentional culture dialog aimed to drive a high engagement, high performance, patient centric culture.
We were incorporated in Delaware in 2007. Our principal executive offices are located at 480 Arsenal Way, Watertown, MA 02472To further drive attraction and our telephone number is (617) 923-1400. Our website address is www.selectabio.com. Information contained on or accessible through our website is not a part of this Annual Report on Form 10-K, and the inclusionretention of our website addresshigh-quality, experienced, and diverse workforce, we invest in this Annual Report on Form 10-K isthe physical, emotional, and financial well-being of our employees. These investments include a competitive mix of compensation and generous insurance benefits. To assist employees with the rising cost of healthcare, we pay 100% of an inactive textual reference only.employee’s deductible and co-insurance payments. All employees are eligible to participate in our equity compensation programs. All employees are awarded new hire equity and annual equity as well as the opportunity to participate in our Employee Stock Purchase Plan. Employees are also eligible to receive an annual cash bonus and to participate in a 401(k)-retirement plan with an industry competitive company match.
In response to the COVID-19 pandemic, we initially implemented changes to our business practices in March 2020, taking precautions to protect the health and safety of our employees by instituting robust hygiene practices, installing temporary safety structures, increasing our cleaning protocols, implementing weekly COVID-19 testing, and limiting regular access to our facilities. As the health environment continues to evolve, we remain committed to actively monitor and evolve our protocols to align with the Center for Disease Control’s best practices, including tracking vaccination and booster status and providing regular communication related to community resources. Additionally, we introduced wellness initiatives as well as a flexible work environment policy and enhanced remote technology solutions, providing remote work opportunities for all employees both now, and well into the future.
Available Information
We file electronically with the SEC our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements and other information. Our SEC filings are available to the public over the Internet at the SEC's website at http://www.sec.gov. We make available on our website at www.selectabio.com, under “Investors,” free of charge, copies of these reports as soon as reasonably practicable after filing or furnishing these reports with the SEC. The hyperlink to our website is included as an inactive textual reference only, and the information on our website is not incorporated by reference in this Annual Report on Form 10-K or in any other filings we make with the SEC.
RISK FACTORS SUMMARY
Investing in our common stock involves various risks. You should carefully read and consider the matters discussed in this Annual Report under the heading “Risk Factors,” which include the following risks:
•We are a clinical-stage company and have incurred significant losses since our inception. We expect to incur losses for the foreseeable future and may never achieve or maintain profitability.
•We will need substantial additional funding in order to complete development of our product candidates and commercialize our products, if approved.
•The terms of our credit facility place restrictions on our operating and financial flexibility.
•Our product candidates are based on our ImmTOR platform, which is an unproven approach designed to induce antigen-specific immune tolerance to biologic drugs.
•Regulatory authorities in the United States and European Union have limited experience in reviewing and approving gene therapy products.
•Clinical drug development involves a lengthy and expensive process, with an uncertain outcome.
•The COVID-19 pandemic may continue to adversely impact our business, including sourcing raw materials and supplies to produce our product candidates, our preclinical studies and clinical trials.
•Geopolitical events, instability and wars can adversely affect both our clinical operations and supply chains that we rely on to advance our drug candidates.
•We rely on 3SBio in China as our primary supplier of pegadricase and on other third parties for the manufacture of our product candidates for preclinical and clinical testing, and expect to continue to do so for the foreseeable future.
•We rely, and expect to continue to rely, on third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including by failing to meet deadlines for the completion of such trials.
•If we or our licensors are unable to adequately protect our proprietary technology, or obtain and maintain issued patents that are sufficient to protect our product candidates, others could compete against us more directly, which would negatively impact our business.
•We may suffer from effects of macroeconomic instability and inflation.
•We would be reliant on Sobi to execute the marketing and sale of SEL-212, if SEL-212 were approved.
•We may not have the funds necessary to fulfill our obligation to repurchase certain warrants.
•We are involved in two securities class action lawsuits.
Item 1A. Risk Factors
Investing in our common stock involves a high degree of risk. You should consider carefully the risks described below, together with the other information included or incorporated by reference in this Annual Report on Form 10-K.Report. If any of the following risks occur, our business, financial condition, results of operations and future growth prospects could be materially and adversely affected. In these circumstances, the market price of our common stock could decline. Other events that we do not currently anticipate or that we currently deem immaterial may also affect our business, prospects, financial condition and results of operations.
RISKS RELATED TO OUR FINANCIAL POSITION AND NEED FOR ADDITIONAL CAPITAL
Risks Related to our Financial Position and Need for Additional Capital
We are a development‑stagedevelopment-stage company and have incurred significant losses since our inception. We expect to incur losses for the foreseeable future and may never achieve or maintain profitability.
Since inception, we have incurred significant operating losses in every year. Our net loss was $55.4losses were $25.7 million for the year ended December 31, 20192021, and $65.3$68.9 million and $55.4 million for each of the years ended December 31, 20182020 and 2017,2019, respectively. As of December 31, 2019,2021, we had an accumulated deficit of $335.8$430.3 million. To date, we have financed our operations primarily through the public offeringofferings and private placements of our securities, funding received from research grantscollaboration and collaborationlicense arrangements and our credit facility. We currently have no source of product revenue, and we do not expect to generate product revenue for the foreseeable future. All of our revenue to date has been collaboration and grant revenue. We have devoted substantially all of our financial resources and efforts to developing our ImmTOR platform, identifying potential product candidates and conducting preclinical studies and our clinical
trials. We are in the early stages of development of most of our product candidates, and we have not completed development of any ImmTOR-enabled therapies. We expect to continue to incur significant expenses and operating losses for the foreseeable future. We expect that our expenses will increase substantially as we:
| |
- | conduct additional clinical trials of SEL‑212, our lead product candidate; |
| |
- | continue the research and development of our other product candidates; |
| |
- | seek to enhance our ImmTOR platform and discover and develop additional product candidates; |
| |
- | seek to maintain and enter into collaboration, licensing and other agreements, including, but not limited to research and development, and/or commercialization agreements; |
| |
- | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
| |
- | potentially establish a sales, marketing and distribution infrastructure and scale up external manufacturing capabilities to commercialize any products for which we may obtain regulatory approval; |
| |
- | maintain, expand and protect our intellectual property portfolio, including through licensing arrangements; |
| |
- | add clinical, scientific, operational, financial and management information systems and personnel, including personnel to support our product development and potential future commercialization efforts and to support our operations as a public company; and |
| |
- | experience any delays or encounter any issues with any of the above, including, but not limited to, failed studies, complex results, safety issues or other regulatory, manufacturing or scale-up challenges. |
•continue the research and development of our product candidates;
•seek to enhance and evolve our ImmTOR platform and discover and develop additional product candidates;
•seek to maintain and enter into collaboration, licensing and other agreements, including, but not limited to research and development, and/or commercialization agreements;
•seek regulatory approvals for any product candidates that successfully complete clinical trials;
•potentially establish a sales, marketing and distribution infrastructure and scale up external manufacturing capabilities to commercialize any products for which we may obtain regulatory approval;
•maintain, expand and protect our intellectual property portfolio, including through licensing arrangements;
•add clinical, scientific, operational, financial and management information systems and personnel, including personnel to support our product development and potential future commercialization efforts;
•experience any delays or encounter any issues with any of the above, including, but not limited to, failed studies, complex results, safety issues or other regulatory, manufacturing or scale-up challenges; and
•are exposed to broad macroeconomic conditions including inflation and supply chain tightness which could result in us paying more, or being unable, to access goods and services.
To become and remain profitable, we must succeed in developing and eventually commercializing products that generate significant revenue. This will require us to be successful in a range of challenging activities, including completing preclinical testing and clinical trials of our product candidates, discovering additional product candidates, obtaining regulatory approval and securing reimbursement for these product candidates, manufacturing, marketing and selling any products for which we may obtain regulatory approval, and establishing and managing our collaborations at various stages of a product candidate’s development. We are only in the preliminary stages of most of these activities. We may never succeed in these activities and, even if we do, may never generate revenues that are significant enough to achieve profitability.
Because of the numerous risks and uncertainties associated with pharmaceutical and biological product development, we are unable to accurately predict the timing or amount of increased expenses or when, or if, we will be able to achieve profitability. If we are required by the FDA or other regulatory authorities to perform studies in addition to those currently expected, or if there are any delays in completing our clinical trials or the development of any of our product candidates, our expenses could increase and product revenue could be further delayed.
Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain profitable would depress our value and could impair our ability to raise capital, expand our business, maintain our research and development efforts, diversify our product offerings or continue our operations.
We will need substantial additional funding in order to complete development of our product candidates and commercialize our products, if approved. If we are unable to raise capital when needed and on terms favorable to us, we could be forced to delay, reduce or eliminate our product development programs or commercialization efforts.
We expect our expenses to increase in connection with our ongoing activities, particularly as we conduct our clinical trials of SEL‑212, continue to develop our gene therapy program,pipeline, including our collaboration with AskBio, research and develop our autoimmune programs and advance the evolution of our ImmTOR platform in combination with IL-2, and continue research and development for our other product candidates. In addition,Additionally, if we obtain regulatory approval for any of our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution. Accordingly, we will need to obtain substantial additional funding to continue operations. If we are unable to raise capital when needed or on attractive terms, we could be forced to delay, reduce or eliminate our clinical trials, our other research and development programs or any future commercialization efforts.
We believe that our existing cash, cash equivalents investments, and restricted cash as of December 31, 20192021 will enable us to fund our operating expenses and capital expenditure requirementscurrent planned operations into the firstthird quarter of 2021. We plan2023, though we may realize additional cash resources upon the achievement of certain contingent collaboration milestones or it may pursue additional cash resources through public or private equity or debt financings or by establishing collaborations with other companies. Management’s expectations with respect to commence our Phase 3 clinical program in SEL-212 in the second halfability to fund current and long-term planned operations are based on estimates that are subject to risks and uncertainties. If actual results are different from management’s estimates, we may need to seek additional strategic or financing opportunities sooner than would otherwise be expected. However, there is no guarantee that any collaboration milestones will be achieved or that any of 2020. Becausethese strategic or financing opportunities will be executed on favorable terms, and some could be dilutive to existing stockholders. If we are unable to obtain additional funding on a timely basis, we may be forced to significantly curtail, delay, or
discontinue one or more of its planned research or development programs or be unable to expand our current operating plan does not contain sufficient resources, we will require additional external sourcesoperations, meet long-term obligations or otherwise capitalize on our commercialization of capital to complete the planned Phase 3 clinical program for SEL-212.our product candidates. We have based this estimate on assumptions that may prove to be wrong, and we could use our capital resources sooner than we currently expect. Because of the uncertainty in securing additional capital, we have concluded that substantial doubt exists with respect to our ability to continue as a going concern within one year after the date of the filing of this Annual Report on Form 10-K.
Moreover, under the terms of the MIT License, MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. If we are unable to reach an agreement with MIT regarding an acceptable amendment of the MIT License and if we are unable to cure the breach, which is currently tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT Agreement, there could be a material adverse effect on our business. See “Business - Licenses and Collaborations – Massachusetts Institute of Technology.”
Our future capital requirements will depend on many factors, including:
| |
- | the scope, progress, results and costs of our clinical trials of SEL‑212; |
| |
- | the number of product candidates that we pursue; |
| |
- | our collaboration agreements remaining in effect, our entering into additional collaboration agreements and our ability to achieve milestones under these agreements; |
| |
- | the cost of manufacturing clinical supplies of our product candidates; |
| |
- | our headcount growth and associated costs; |
| |
- | the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials for our other product candidates; |
| |
- | the costs, timing and outcome of regulatory review of our product candidates; |
| |
- | the costs and timing of future commercialization activities, including manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval; |
| |
- | the revenue, if any, received from commercial sales of our product candidates for which we receive marketing approval; |
| |
- | the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property‑related claims; |
| |
- | the effect of competing technological and market developments; and |
| |
- | the extent to which we acquire or invest in businesses, products and technologies, including entering into licensing or collaboration arrangements for product candidates. |
•the scope, progress, results and costs of our clinical trials, preclinical development, and laboratory testing;
•the number of product candidates that we pursue and the speed with which we pursue development;
•our collaboration agreements remaining in effect, our entering into additional collaboration agreements and our ability to achieve milestones under these agreements;
•our headcount growth and associated costs;
•the costs, timing and outcome of regulatory review of our product candidates;
•the costs and timing of future commercialization activities, including manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval;
•the revenue, if any, from commercial sales of our product candidates for which we receive marketing approval;
•the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property-related claims;
•the effect of competing technological and market developments; and
•the extent to which we acquire or invest in businesses, products and technologies, including entering into licensing or collaboration arrangements for product candidates.
Any additional fundraising efforts may divert our management from their day‑to‑dayday-to-day activities, which may adversely affect our ability to develop and commercialize our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Market volatility resulting from the COVID-19 pandemic or other factors could also adversely impact our ability to access capital as and when needed. Moreover, the terms of any financing may adversely affect the holdings or the rights of our stockholders, and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our shares to decline. The sale of additional equity or convertible securities would dilute all of our stockholders. The incurrence of indebtedness could result in increased fixed payment obligations and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions
that could adversely impact our ability to conduct our business. We could also be required to seek funds through arrangements with collaborators or others at an earlier stage than otherwise would be desirable and we may be required to relinquish rights to some of our technologies or product candidates or otherwise agree to terms unfavorable to us, any of which may have a material adverse effect on our business, operating results and prospects.
If we are unable to obtain funding on a timely basis, we may be required to significantly curtail, delay or discontinue one or more of our research or development programs, including our clinical trial programs, or the commercialization of any product candidates, or be unable to sustain or expand our operations or otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results of operations.
Our recurring losses from operations and negative cash flows from operations raise substantial doubt regarding our ability to continue as a going concern.
As of December 31, 2019 and December 31, 2018, we had an accumulated deficit of $335.8 million and $280.4 million, respectively. We anticipate operating losses to continue for the foreseeable future due to, among other things, costs related to research, development of our product candidates, conducting preclinical studies and clinical trials, and our administrative organization. We will require substantial additional financing to fund our operations and to continue to execute our strategy, and we will pursue a range of options to secure additional capital. These conditions raise substantial doubt about our ability to continue as a going concern within one year after the date of filing this Annual Report on Form 10-K.
We are exploring various sources of funding such as strategic collaborations and the issuance of equity to fund our operations. If we raise additional funds through strategic collaborations and alliances, which may include existing collaboration partners, we may have to relinquish valuable rights to our technologies or product candidates, or, grant licenses on terms that are not favorable to us. To the extent that we raise additional capital through the sale of equity, the ownership interest of our existing shareholders will be diluted and other preferences may be necessary that adversely affect the rights of existing shareholders. Because our current operating plan does not contain sufficient resources, we will require additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212. If we are unable to raise sufficient capital, we intend to curtail expenses contemplated by the current operating plan, and we may be required to delay, limit, reduce or terminate our product development efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves. If the foregoing plans are unsuccessful and we are unable to continue as a going concern, you could lose all or part of your investment in the company.
Our limited operating history may make it difficult for you to evaluate the success of our business to date and to assess our future viability.
We commenced active operations in 2007, and our operations to date have been limited to developing and researching our ImmTOR platform and related products and programs, building our intellectual property portfolio, developing our supply chain, planning our business, raising capital and providing general and administrative support for these operations. Other than SEL-212, our lead product candidate, our other product candidates are still in preclinical development. While we have completed our early development clinical trials and a Phase 2 clinical trial for SEL‑212, we have not completed a clinical trial for any other product candidate, nor have we demonstrated our ability to successfully complete any Phase 3 or other pivotal clinical trials, obtain regulatory approvals, manufacture a commercial scale product, or arrange for a third party to do so on our behalf, or conduct sales and marketing activities necessary for successful product commercialization. Additionally, we expect our financial condition and operating results to continue to fluctuate significantly from quarter‑to‑quarter and year‑to‑year due to a variety of factors, many of which are beyond our control. Consequently, any predictions you make about our future success or viability may not be as accurate as they could be if we had a longer operating history.
The terms of our credit facility place restrictions on our operating and financial flexibility. If we raise additional capital through debt financing, the terms of any new debt could further restrict our ability to operate our business.
On September 12, 2017,August 31, 2020, we entered into a term loan, facilityor the 2020 Term Loan, of up to $21.0$35.0 million, with Silicon Valley Bank,consisting of term loans in an aggregate amount of $25.0 million, or SVB. Thethe Term A Loan, and term loan facility isloans in an aggregate amount of $10.0 million, or the Term B Loan, governed by a loan and security agreement dated September 12, 2017, betweenamong us and Oxford Finance LLC, or Oxford, as collateral agent and a lender, and Silicon Valley Bank, or SVB, whichas a lender. The Term A Loan was funded in full on September 13, 2017. TheAugust 31, 2020, the proceeds of which were used to repay our previously existing 2017 term loan facility with SVBand for general corporate and working capital purposes, and the draw period relating to the Term B Loan expired on September 30, 2021.
The 2020 Term Loan is secured by a lien on substantially all of our assets, other than intellectual property, provided that such lien on substantially all assets includes any rights to payments and proceeds from the sale, licensing or disposition of intellectual property. We also granted SVBOxford a negative pledge with respect to our intellectual property.
The term loan facility contains customaryFailure to satisfy our current and future debt obligations, including covenants and representations, including but not limited to financial reporting obligations and limitations on dividends, indebtedness, collateral, investments, distributions, transfers, mergerstake or acquisitions, taxes, corporate changes, deposit accounts, and subsidiaries. The term loan facility also contains other customary provisions, such as expense reimbursement, non-disclosure obligations as well as indemnification rights for the benefit of SVB. The events of defaultavoid specific actions, under the term loan facility include, but are not limited to, our failure to make any payments of principal or interest under the term loan facility or other transaction documents, our breach or default2020 Term Loan could result in the performance of any covenant under the
term loan facility or other transaction documents, the occurrence of a material adverse effect, making a false or misleading representation or warranty in any material respect under the term loan facility, our insolvency or bankruptcy, any attachment or judgment on our assets of at least approximately $0.3 million, or the occurrence of any default under any of our agreements or obligations involving indebtedness in excess of approximately $0.3 million. If an event of default, occurs, SVB is entitled to take enforcement action, including accelerationour lenders could accelerate all of the amounts due under the term loan facility.due. If we raise any additional debt financing, the terms of such additional debt could further restrict our operating and financial flexibility.
Changes in U.S. tax law may materially adversely affect our financial condition, results of operations and cash flows.
The Tax Cuts and Jobs Act of 2017, or TCJA, has significantly changed the U.S. federal income taxation of U.S. corporations. The TCJA remains unclear in many respects and has been, and may continue to be, the subject of amendments and technical corrections, as well as interpretations and implementing regulations by the Treasury and Internal Revenue Service, which have lessened or increased certain adverse impacts of the TCJA and may do so in the future. We continue to work with our tax advisors and auditors to determine the full impact that the TCJA will have on us. We urge our investors to consult with their legal and tax advisors with respect to the TCJA.
Our ability to use our net operating loss and research and development tax credit carryforwards to offset future taxable income may be subject to certain limitations.
We have net operating loss carryforwards, or NOLs, for federal and state income tax purposes that may be available to offset our future taxable income, if any. In general, under Sections 382 and 383 of the U.S. Internal Revenue Code of 1986, as amended, or the Code, a corporation that undergoes an “ownership change” is subject to limitations on its ability to use its pre‑changepre-change NOLs to offset future taxable income. If the U.S. Internal Revenue Service, or IRS, challenges our analysis that existing NOLs will not expire before utilization due to previous ownership changes, or if we undergo an ownership change in connection with or after a public offering, our ability to use our NOLs could be limited by Section 382 of the Code. Future changes in our stock ownership, some of which are outside of our control, could result in an ownership change under Sections 382 and 383 of the Code. Furthermore, our ability to use NOLs of companies that we may acquire in the future may be subject to limitations. As a result, we may not be able to use a material portion of the NOLs reflected on our balance sheet, even if we attain profitability. The reduction of the corporate tax rate under the TCJA may cause a reduction in the economic benefit of our NOLs and other deferred tax assets available to us. Under the TCJA, although the treatment ofcurrent law, NOLs arising on orthat arose before December 31, 2017 has generally not changed, NOLs arising on or after January 1, 2018, will generally onlybegin to expire in 2041. NOLs that arose after 2017 may be ableused to offset at most 80% of our taxable income. This changeincome to the extent not offset by pre-2018 NOLs. As a result, we may require usbecome required to pay federal income taxes in future years despite generating a losshaving generated losses for federal income tax purposes in prior years.
RISKS RELATED TO THE DISCOVERY, DEVELOPMENT AND REGULATORY APPROVAL OF OUR PRODUCT CANDIDATESRisks Related to the Development of our Product Candidates
Our product candidates are based on our ImmTOR platform, which is an unproven approach designed to induce antigen‑specificantigen-specific immune tolerance to biologic drugs. We are very early in most of our clinical development efforts and may not be successful in our efforts to use our ImmTOR platform to build a pipeline of product candidates and develop marketable drugs.
All of our product candidates are derived from our ImmTOR platform, which is an unproven approach to induce antigen‑specificantigen-specific immune tolerance and to mitigate the immunogenicity of biologic therapies currently being implemented to treat patients. We are primarily developing our ImmTOR platform to restore self-tolerance to autoantigens and potentially treat autoimmune diseases, to be co-administered with AAV gene therapies to potentially enable redosing of said gene therapies and improve and enable activity in biologics that are designed to treat rare(including therapeutic enzymes and serious diseases, with an initial focus on developing SEL‑212other immunogenic drugs).
While we have completed our early development clinical trials and a Phase 2 clinical trial for the treatment of chronic refractory gout. We are also leveraging our ImmTOR platform to pursue programs in additional therapeutic areas withSEL-212, we have not completed a focus on gene therapy.
We are developing two gene therapy product candidatesclinical trial for rare inborn errors of metabolism. Our lead gene therapy program, known as SEL-302, is a potential gene therapyany other product candidate, nor have we demonstrated our ability to successfully complete any Phase 3 or other pivotal clinical trials, obtain regulatory approvals, manufacture a commercial product, or arrange for MMA. In August 2019, we entered into a feasibility studythird party to do so on our behalf, or conduct other sales and license agreement with AskBio, the AskBio Collaboration Agreement, pursuant to which we and AskBio agreed to conduct proof of concept studies to potentially validate the use of our ImmTOR platform in conjunction with an AAV gene therapymarketing activities necessary for the treatment of MMA, based on SEL-302, to mitigate the formation of neutralizing anti-AAV capsid antibodies. If the proof of concept studies are successful we will proceed with a collaboration to pursue the development and commercialization of AAV gene therapy product candidates utilizing ImmTOR for the treatment of certain agreed serious rare and orphan genetic diseases. Our second gene therapy product candidate, known as SEL-313, is being developed to treat ornithine transcarbamylase deficiency. In September 2018, we announced a collaboration with the European consortium, CureCN, for an ImmTOR+AAV gene therapy combination product candidate in Crigler-Najjar syndrome, a rare genetic disorder characterized by an inability to properly convert and clear bilirubin from the body. We expect the CureCN consortium to obtain scientific advice from the German drug regulatory authority in 2020.
We are at an early stage of development of most of our product candidates and our technology has not yet led to, and may never lead to, approvable or marketable drugs.commercialization. We may have problems identifying new product candidates and applying our
technologies to these other areas. Even if we are successful in identifying new product candidates, they may not be suitable for clinical development, including as a result of harmful side effects, limited efficacy or other characteristics that indicate that they are unlikely to be products that will receive marketing approval and achieve market acceptance. The success of our product candidates will depend on several factors, including the following:
| |
- | design, initiation and completion of preclinical studies and clinical trials with positive results; |
| |
- | reliance on third parties (including but not limited to collaborators, licensees, clinical research organizations and contract manufacturing organizations); |
| |
- | receipt of marketing approvals from applicable regulatory authorities; |
| |
- | obtaining and maintaining patent and trade secret protection and regulatory exclusivity for our product candidates; |
| |
- | making arrangements with third-party manufacturers for, or establishing, commercial manufacturing capabilities, or establishing such capabilities ourselves; |
| |
- | launching commercial sales of our products, if and when approved, whether alone or in collaboration with others; |
| |
- | our existing collaboration agreements remaining in effect and our ability to enter into new collaborations throughout the development process as appropriate, from preclinical studies through to commercialization; |
| |
- | acceptance of our products, if and when approved, by patients and the medical community; |
| |
- | effectively competing with other therapies; |
| |
- | obtaining and maintaining coverage and adequate reimbursement by third‑party payors, including government payors, for our products, if approved; |
| |
- | protecting our rights in our intellectual property portfolio; |
| |
- | operating without infringing or violating the valid and enforceable patents or other intellectual property of third parties; |
| |
- | maintaining an acceptable safety profile of our products following approval; and |
| |
- | •design, initiation and completion of preclinical studies and clinical trials with positive results; •reliance on third parties, including but not limited to collaborators, licensees, clinical research organizations and contract manufacturing organizations; •receipt of marketing approvals from applicable regulatory authorities; •obtaining and maintaining patent and trade secret protection and regulatory exclusivity for our product candidates and not infringing or violating patents or other intellectual property of third parties; •making arrangements with third-party manufacturers for, or establishing, commercial manufacturing capabilities, or establishing such capabilities ourselves; •launching commercial sales of our products, if and when approved, whether alone or in collaboration with others; •acceptance of our products, if and when approved, by patients and the medical community; •effectively competing with other therapies; •obtaining and maintaining coverage and adequate reimbursement by third-party payors, including government payors, for our products, if approved; •maintaining an acceptable safety profile of our products following approval; and •maintaining and growing an organization of scientists and business people who can develop and commercialize our product candidates and technology. |
If we do not successfully develop and commercialize our product candidates based upon our technological approach, we will not be able to obtain future revenues, which would result in significant harm to our financial position and adversely affect our stock price.technology.
As a result, we cannot be certain that our approach, or our development of SEL-212, will lead to the development or approval of marketable products. In addition:
| |
- | due to the unproven nature of our ImmTOR therapeutics, there may be different efficacy and safety rates in various indications; |
| |
- | the FDA or other regulatory agencies may lack experience in evaluating the efficacy and safety of products based on ImmTOR or a biologic sourced from China or other jurisdictions, which could result in a longer‑than‑expected regulatory review process, increase our expected development costs or delay or prevent commercialization of our product candidates; and |
| |
- | in the event of a biologics license application, or BLA, for SEL‑212 or another product and a pre‑approval inspection by the FDA of the facilities of Shenyang Sunshine Pharmaceutical Co., Ltd., or 3SBio, or any other third party manufacturers we may use, the FDA may not approve the facility for production or may make observations that will take significant time for 3SBio or such other manufacturer to address. |
The occurrence of any of the foregoing would effectively prevent or delay approval of our lead and other product candidates. The outbreak of COVID-19 may continue to adversely impact our business, including our preclinical studies and clinical trials.
The ongoing COVID-19 pandemic has impacted our business and we expect it to continue to do so. In response to the spread of COVID-19, we closed our principal executive office with our administrative employees continuing their work outside of our office and limited the number of staff in any given research and development laboratory. As COVID-19 continues to spread in the United States and elsewhere, we may experience disruptions that could severely impact our business, preclinical studies and clinical trials, including:
•delays in receiving approval from local regulatory authorities to initiate our planned clinical trials;
•delays or difficulties in enrolling or sustaining patients in our clinical trials;
•delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff;
•delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials, such as ImmTOR including interruption in global shipping that may affect the transport of clinical trial materials;
•changes in local regulations as part of a response to the COVID-19 pandemic which may require us to change the ways in which our clinical trials are conducted, which may result in unexpected costs, or to discontinue the clinical trials altogether;
•diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals and clinics serving as our clinical trial sites and hospital and clinic staff supporting the conduct of our clinical trials;
•interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by federal or state governments, employers and others, or interruption of clinical trial subject visits and study procedures, or the closing of clinical trial sites due to the virus, the occurrence of which could affect the integrity of clinical trial data;
•risk that participants enrolled in our clinical trials will contract COVID-19 while the clinical trial is ongoing, or will withdraw from the clinical trial due to concerns over COVID-19, which could impact the results of the clinical trial, including by increasing the number of observed adverse events, or reducing the statistical power of the clinical trials;
•interruptions or delays in preclinical studies due to restricted or limited operations at our research and development laboratory facility;
•delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations in employee resources or forced furlough of government employees;
•limitations in employee resources that would otherwise be focused on the conduct of our clinical trials, including because of sickness of employees or their families or the desire of employees to avoid contact with large groups of people;
•refusal of the FDA to accept data from clinical trials in affected geographies;
•changes to the clinical endpoints, statistical analysis plan, or enrollment plans for ongoing clinical trials due to limitations in patients, resources, or sites due to COVID-19;
•interruption or delays to our sourced discovery and clinical activities; and
•impacts from prolonged remote work arrangements, such as increased cybersecurity risks and strains on our business continuity plans.
The extent to which the COVID-19 pandemic impacts our business, preclinical studies and clinical trials will depend on future developments, which are highly uncertain and cannot be predicted with confidence.
We are applying our ImmTOR platform to antigen-specific immune tolerance for gene therapy involving gene augmentation, replacement or editing. Regulatory authorities in the United States and European Union have limited experience in reviewing and approving gene therapy products, which could affect the time and data required to obtain marketing authorization of any of our product candidates.
Our future success depends in part on our successful development of viable gene therapy product candidates utilizing our ImmTOR platform. We may experience problems or delays in developing such product candidates and any such problems or delays (i) may result in unanticipated costs and time to develop our product candidates and/or (ii) may not be resolved in a satisfactory manner.
The process of obtaining marketing approvals, both in the United States and abroad, is expensive and may take many years. If additional clinical trials are required for certain jurisdictions, these trials can vary substantially based upon a variety of factors, including the type, complexity and novelty of the product candidates involved, and may ultimately be unsuccessful. Changes in marketing approval policies during the development period, changes in or the enactment or promulgation of additional statutes or regulations, respectively, or changes in the regulatory review process for each submitted product application, may cause delays in the review and approval of an application.
The regulatory approval process and clinical trial requirements for novel product candidates can be more expensive and take longer than for other, better known or more extensively studied product candidates, and we cannot predict how long it will take or how much it will cost to complete clinical developments and obtain regulatory approvals for a gene therapy product candidate in either the United States or the European Union or how long it will take to commercialize a gene therapy product candidate, if and when approved. Regulatory requirements governing gene therapy products have changed frequently and may continue to change in the future. For example, the FDA established the Office of Tissues and Advanced Therapies within its Center for Biologics Evaluation and Research, or CBER, to consolidate the review of gene therapy and related products, and the Cellular, Tissue and Gene Therapies Advisory Committee to advise CBER on its review. These and other regulatory review agencies, committees and advisory groups and the requirements and guidelines they promulgate may lengthen the regulatory review process, require us to perform additional preclinical studies or clinical trials, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of these treatment candidates or lead to significant post-approval limitations or restrictions.
Additionally, under the National Institutes of Health, or NIH, Guidelines for Research Involving Recombinant DNA Molecules, or the NIH Guidelines, supervision of human gene transfer trials includes evaluation and assessment by an institutional biosafety committee, or IBC, a local institutional committee that reviews and oversees research utilizing recombinant or synthetic nucleic acid molecules at that institution. The IBC assesses the safety of the research and identifies any potential risk to public health or the environment, and such review may result in some delay before initiation of a clinical trial. While the NIH Guidelines are not mandatory unless the research in question is being conducted at or sponsored by institutions receiving NIH funding of recombinant or synthetic nucleic acid molecule research, many companies and other institutions not otherwise subject to the NIH Guidelines voluntarily follow them.
A similar framework is in place in the European Union, or the EU. The European Medicines Agency, or the EMA, has a Committee for Advanced Therapies, or CAT, that is responsible for assessing the quality, safety and efficacy of advanced-therapy medicinal products. Advanced-therapy medical products include gene therapy medicine, somatic-cell therapy medicines and tissue-engineered medicines. The role of the CAT is to prepare a draft opinion on an application for marketing authorization for a gene therapy medicinal candidate that is submitted to the EMA. In the EU, the development and evaluation of a gene therapy medicinal product must be considered in the context of the relevant EU guidelines. The EMA may issue new guidelines concerning the development and marketing authorization for gene therapy medicinal products and require that we comply with these new guidelines. Similarly, complex regulatory environments exist in other jurisdictions in which we might consider seeking regulatory approvals for our product candidates, further complicating the regulatory landscape. As a result, the procedures and standards applied to gene therapy products and cell therapy products may be applied to any of our gene therapy or genome editing product candidates, but that remains uncertain at this point.
The clinical trial requirements of the FDA, the EMA and other regulatory authorities and the criteria these regulators use to evaluate the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty and intended use and market of the potential products. The regulatory approval process for product candidates created with novel genome editing technology such as ours can be more lengthy, rigorous and expensive than the process for other better known or more extensively studied product candidates and technologies. Since we are developing novel treatments for diseases in which there is little clinical experience with new endpoints and methodologies, there is heightened risk that the FDA, the European Medicines Agency, or the EMA, or comparable regulatory bodies may not consider the clinical trial endpoints to provide clinically meaningful results, and the resulting clinical data and results may be more difficult to analyze. This may be a particularly significant risk for many of the genetically defined diseases for which we may develop
product candidates alone or with collaborators due to small patient populations for those diseases, and designing and executing a rigorous clinical trial with appropriate statistical power is more difficult than with diseases that have larger patient populations. Regulatory agencies administering existing or future regulations or legislation may not allow production and marketing of products utilizing genome editing technology in a timely manner or under technically or commercially feasible conditions. Even if our product candidates obtain required regulatory approvals, such approvals may later be withdrawn as a result of changes in regulations or the interpretation of regulations by applicable regulatory agencies.
Changes in applicable regulatory guidelines may lengthen the regulatory review process for our product candidates, require additional studies or trials, increase development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of such product candidates, or lead to significant post-approval limitations or restrictions. Additionally, adverse developments in clinical trials conducted by others of gene therapy products or products
created using genome editing technology, or adverse public perception of the field of genome editing, may cause the FDA, the EMA and other regulatory bodies to revise the requirements for approval of any product candidates we may develop or limit the use of products utilizing genome editing technologies, either of which could materially harm our business. Furthermore, regulatory action or private litigation could result in expenses, delays or other impediments to our research programs or the development or commercialization of current or future product candidates.
As we advance any gene therapy product candidates, we will be required to consult with various regulatory authorities, and we must comply with applicable laws, rules, and regulations, which may change from time to time including during the course of development of our product candidates. If we fail to do so, we may be required to delay or discontinue the clinical development of certain of our product candidates. These additional processes may result in a review and approval process that is longer than we otherwise would have expected. Even if we comply with applicable laws, rules, and regulations, and even if we maintain close coordination with the applicable regulatory authorities with oversight over our product candidates, our development programs may fail to succeed. Regulatory authorities have substantial discretion in the approval process and may refuse to accept a marketing application as deficient or may decide that our data is insufficient for approval and require additional preclinical, clinical or other studies. In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit or prevent marketing approval of a product candidate. Additionally, adverse developments in clinical trials conducted by others of gene therapy products or products created using genome editing technology, or adverse public perception of the field of genome editing, may cause the FDA and other regulatory bodies to revise the requirements for approval of any product candidates we may develop or limit the use of products utilizing genome editing technologies, either of which could materially harm our business. Delay or failure to obtain, or unexpected costs in obtaining, the regulatory approval necessary to bring a potential product to market would materially and adversely affect our business, financial condition, results of operations and prospects.
Clinical drug development involves a lengthy and expensive process, with an uncertain outcome. We may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates.
Our lead product candidate,Except for SEL-212 was evaluated in a Phase 2 clinical program that was initiated in October 2016 and the final patient's last visit occurred in January 2019. In March 2019, we initiated COMPARE, a Phase 2 clinical trial designed to directly compare the safety, efficacy and tolerability of SEL-212 to the currently FDA-approved uricase therapy, KRYSTEXXA, for the treatment of patients with chronic refractory gout, and completedSEL 302, our targeted enrollment of the COMPARE trial in December 2019. We are preparing for the start of a pivotal Phase 3 program for SEL-212.
In May 2017 we licensed LMB-100, a potent immunotoxin, from the National Cancer Institute (NCI) and a Phase 1 clinical trial of LMB-100 plus ImmTOR (SEL-403) initiated in March 2018 by NCI in patients with malignant pleural or peritoneal mesothelioma who had undergone at least one regimen of chemotherapy under a Cooperative Research and Development Agreement (CRADA) between the NCI and the Company. In October 2018, the NCI informed the Company of a Grade 5 Serious Adverse Event (patient death) in this clinical trial related to pneumonitis, which was deemed by the trial investigator to be probably related to SVP-Rapamycin and possibly related to the patient’s pleural mesothelioma condition. This patient had received previous therapies, including two courses of radiation therapy and three different immune check point inhibitors, which have been reported to be associated with pneumonitis. However, the possible relationship to SVP-Rapamycin could not be excluded. Pneumonitis has been reported in patients receiving daily oral rapamycin. In addition, a Serious Adverse Event (pericardial effusion) was seen in one of the other three patients dosed in the SEL-403 clinical trial. Pericardial effusion can also be a side effect of immunotoxin therapies targeting mesothelin. The FDA placed the IND for SEL-403 on full clinical hold in response to adverse events observed during the Phase 1 trial. Selecta has terminated the license of LMB-100 from NCI, effective April 9, 2019 and is no longer pursuing this product candidate.
Aside from these programs, our other product candidates are in preclinical development. It is impossible to predict when or if any of our product candidates will prove effective and safe in humans or will receive regulatory approval, and the risk of failure through the development process is high. Before obtaining marketing approval from regulatory authorities for the sale of any product candidate, we must complete preclinical development and then conduct extensive clinical trials to demonstrate the safety and efficacy of our product candidates in humans. Preclinical development is costly and inherently uncertain. For example, we have invested significant resources in our preclinical gene therapy program, which has demonstrated the potential for treatment of rare inborn errors of metabolism. Early preclinical results may not be predictive of future results, however, if our technology proves to be ineffective or unsafe as a result of, among other things, adverse side effects, pre-existing anti-drug antibodies that can neutralize the viral vector and block gene transfer, or cellular immune response to the transduced cells, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the clinical development and commercialization of our product candidates.
Before obtaining marketing approval from regulatory authorities for the sale of our product candidates, we must conduct extensive clinical trials to demonstrate the safety and efficacy of the product candidate for its intended indications. Clinical testing is expensive, difficult to design and implement, can take many years to complete and its outcome is inherently uncertain. A failed clinical trial can occur at any stage of testing. Moreover, the outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results. For example, the topline clinical trial results we reported from our Phase 2 head-to-head (COMPARE)COMPARE study of SEL-212 including
interim results,and the top-line data from our SEL-399 program may not be predictive of future results. Moreover, we may not be able to complete, or may be required to deviate from the current clinical trial protocol for a variety of reasons.
Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in clinical trials after achieving positive results in preclinical development or early-stage clinical trials, and we cannot be certain that we will not face similar setbacks. These setbacks have been caused by, among other things, preclinical findings made while clinical trials were underway or safety or efficacy observations made in clinical trials, including adverse events. SAEs caused by, or other unexpected properties of, any product candidates that we may choose to develop could cause us, an institutional review board or regulatory authority to interrupt, delay or halt clinical trials of one or more of such product candidates and could result in a more restrictive label or the delay or denial of marketing approval by the FDA or comparable non-U.S. regulatory authorities. If any product candidate that we may choose to develop is associated with SAEs or other unexpected properties, we may need to abandon development or limit development of that product candidate to certain uses or subpopulations in which those undesirable characteristics would be expected to be less prevalent, less severe or more tolerable from a risk-benefit perspective. For example, in the SEL-403 Phase 1 clinical trial, a Grade 5 SAE (patient death) occurred that was deemed by the trial investigator to be probably related to SVP-Rapamycin and possibly related to the patient's pleural mesothelioma condition which led the Company to abandon development of SEL-403. In the SEL-212 Phase 1/2 clinical program, we have observed multiple SAEs, have occurred, and future SAEs may occur causing the Companyus to incur additional costs or experience delays in completing, or causing the Companyus to ultimately be unable to complete, the development and commercialization of our product candidates, and delay or prevent our ability to
obtain FDA approval. Moreover, preclinical and clinical data is often susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless failed to obtain FDA or other regulatory authority approval. If we fail to produce positive results in clinical trials of our product candidates, the development timeline and regulatory approval and commercialization prospects for our product candidates, and, correspondingly, our business and financial prospects, would be negatively impacted.
In addition, we cannot be certain as to what type and how many clinical trials the FDA will require us to conduct before we may gain regulatory approval to market SEL-212 or any of our other product candidates in the United States or other countries, if any. Prior to approving a new therapeutic product, the FDA generally requires that safety and efficacy be demonstrated in two adequate and well-controlled clinical trials. We expect that we may need to conduct more than one Phase 3 trial for SEL-212 for a chronic refractory gout indication in order to gain approval from the FDA. Even if we conduct more than one Phase 3 trial for SEL-212, the FDA may not accept the data, and may delay, limit or deny approval of SEL-212. Additional clinical trials could cause us to incur significant development costs, delay or prevent the commercialization of SEL-212 or otherwise adversely affect our business.
We may experience numerous unforeseen events during, or as a result of, clinical trials that could delay or prevent our ability to receive marketing approval for, or commercialize, our product candidates, including:
| |
- | clinical trials of our product candidates may produce unfavorable, incomplete or inconclusive results; |
| |
- | regulators or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site; |
| |
- | we may experience delays in reaching, or fail to reach, agreement on acceptable terms with contract research organizations, or CROs, or clinical trial sites; |
| |
- | we may be unable to recruit suitable patients to participate in a clinical trial, the number of patients required for clinical trials of our product candidates may be larger than we expect, enrollment in these clinical trials may be slower than we expect or participants may drop out of these clinical trials at a higher rate than we expect; |
| |
- | the number of clinical trial sites required for clinical trials of our product candidates may be larger than we expect; |
| |
- | our third‑party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all; |
| |
- | we may have to suspend or terminate clinical trials of our product candidates for various reasons, including a finding that the participants are being exposed to unacceptable health risks; |
| |
- | investigators, regulators, data safety monitoring boards or institutional review boards may require that we or our investigators suspend or terminate clinical research, or we may decide to do so ourselves; |
| |
- | investigators may deviate from the trial protocol, fail to conduct the trial in accordance with regulatory requirements or misreport study data; |
| |
- | the cost of clinical trials of our product candidates may be greater than we expect or we may have insufficient resources to pursue or complete certain aspects of our clinical trial programs or to do so within the timeframe we planned; |
•clinical trials of our product candidates may produce unfavorable, incomplete or inconclusive results;
| |
- | the supply or quality of raw materials or manufactured product candidates (whether provided by us or third parties) or other materials necessary to conduct clinical trials of our product candidates may be insufficient, inadequate or not available at an acceptable cost, or in a timely manner, or we may experience interruptions in supply; |
| |
- | regulators may revise the requirements for approving our product candidates, or such requirements may not be as we expect; |
| |
- | the FDA or comparable foreign regulatory authorities may disagree with our clinical trial design or our interpretation of data from preclinical studies and clinical trials, or may change the requirements for approval even after it has reviewed and commented on the design of our clinical trials; and |
| |
- | regarding trials managed by our existing or any future collaborators, our collaborators may face any of the above issues, and may conduct clinical trials in ways they view as advantageous to them but potentially suboptimal for us. |
•we may experience delays in reaching, or fail to reach, agreement on acceptable terms with contract research organizations, or CROs, or clinical trial sites;
•we may be unable to recruit suitable patients to participate in a clinical trial, the number of patients required for clinical trials of our product candidates may be larger than we expect, enrollment in these clinical trials may be slower than we expect or participants may drop out of these clinical trials at a higher rate than we expect, or enrollment could be affected by the ongoing COVID-19 pandemic or the ongoing conflict in Ukraine;
•the number of clinical trial sites required for clinical trials of our product candidates may be larger than we expect;
•our third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all;
•we may have to suspend or terminate clinical trials of our product candidates for various reasons, including a finding that the participants are being exposed to unacceptable health risks;
•investigators, regulators, data safety monitoring boards or institutional review boards may require that we or our investigators suspend or terminate clinical research, or we may decide to do so ourselves;
•investigators may deviate from the trial protocol, fail to conduct the trial in accordance with regulatory requirements or misreport study data;
•the cost of clinical trials of our product candidates may be greater than we expect or we may have insufficient resources to pursue or complete certain aspects of our clinical trial programs or to do so within the timeframe we planned;
•the supply or quality of raw materials or manufactured product candidates (whether provided by us or third parties) or other materials necessary to conduct clinical trials of our product candidates may be insufficient, inadequate or not available at an acceptable cost, or in a timely manner, or we may experience interruptions in supply;
•regulators may revise the requirements for approving our product candidates, or such requirements may not be as we expect;
•the FDA or comparable foreign regulatory authorities may disagree with our clinical trial design or our interpretation of data from preclinical studies and clinical trials, or may change the requirements for approval even after it has reviewed and commented on the design of our clinical trials;
•regarding trials managed by our existing or any future collaborators, our collaborators may face any of the above issues, and may conduct clinical trials in ways they view as advantageous to them but potentially suboptimal for us; and
•geopolitical events may affect international and overseas trial sites in ways beyond our control.
If we are required to conduct additional clinical trials or other testing of our product candidates beyond those that we currently contemplate, if we are unable to successfully complete clinical trials of our product candidates or other testing, if the results of these trials or tests are not positive or are only modestly positive or if there are safety concerns, or if we are forced to delay or abandon certain clinical trials or other testing in order to conserve capital resources, we may:
| |
- | be delayed in obtaining marketing approval for our product candidates, if at all; |
| |
- | lose the support of collaborators, requiring us to bear more of the burden of research and development; |
| |
- | not obtain marketing approval at all; |
| |
- | obtain marketing approval in some countries and not in others; |
| |
- | obtain approval for indications or patient populations that are not as broad as intended or desired; |
| |
- | obtain approval with labeling that includes significant use or distribution restrictions or safety warnings; |
| |
- | be subject to additional post‑marketing testing requirements; or |
| |
- | have a product removed from the market after obtaining marketing approval. |
•be delayed in obtaining marketing approval for our product candidates, if at all;
•lose the support of collaborators, requiring us to bear more of the burden of research and development;
•obtain marketing approval in some countries and not in others;
•obtain approval for indications or patient populations that are not as broad as intended or desired;
•obtain approval with labeling that includes significant use or distribution restrictions or safety warnings;
•be subject to additional post-marketing testing requirements; or
•have a product removed from the market after obtaining marketing approval.
In addition, disruptions caused by the COVID-19 pandemic may increase the likelihood that we encounter such difficulties or delays in initiating, enrolling, conducting or completing our planned and ongoing clinical trials. We could also encounter delays if a clinical trial is suspended or terminated by us, by the IRBs of the institutions in which such trials are being conducted, by a Data Safety Monitoring Board for such trial or by the FDA or comparable foreign regulatory authorities. Such authoritiesterminated. Authorities may impose such a suspension or termination due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or trial site by the FDA or comparable foreign regulatory authorities resulting in the imposition of a clinical hold, unforeseen safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial. In addition, changes in regulatory requirements and policies may occur, and we may need to amend clinical trial protocols to comply with these changes. Amendments may require us to resubmit our clinical trial protocols to IRBs for reexamination, which may impact the costs, timing or successful completion of a clinical trial.
We filed an IND to conduct a Phase 1/2 clinical trial of our SEL-302 product candidate in pediatric patients with methylmalonic acidemia in the third quarter of 2021. On November 23, 2021, this trial was placed on clinical hold by the FDA, with questions specifically relating to CMC of the AAV vector. On February 9, 2022, we submitted a written response to the FDA to answer its questions. On March 9, 2022, we received a letter from the FDA indicating the clinical hold was removed and the trial may proceed. ImmTOR manufacturing continues to proceed in accordance with our expectations, and we have not observed any impact to any of our ImmTOR programs.
Our product development costs will increase if we experience delays in clinical testing or in obtaining marketing approvals. We do not know whether any of our preclinical studies or clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. Significant preclinical or clinical trial delays also could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do, potentially impairing our ability to successfully commercialize our product candidates and harming our business and results of operations.
If we experience delays or difficulties in the enrollment of patients in clinical trials, our receipt of necessary regulatory approvals could be delayed or prevented.
We may not be able to initiate or continue clinical trials for our product candidates if we are unable to locate and enroll a sufficient number of eligible patients to participate in these trials as required by the FDA or similar regulatory authorities outside the United States. In addition, from time to time our competitors have ongoing clinical trials for product candidates that treat the same indications as our product candidates, and patients who would otherwise be eligible for our clinical trials may instead enroll in clinical trials of our competitors’ product candidates.
We are initially developing our lead product candidate, SEL‑212, For example, SEL-212 is being developed for the treatment of chronic refractory gout, which affects approximately 160,000 patients in the United States.a small patient population. Accordingly, there is a limited number of patients who could enroll in our clinical studies.
In addition tostudies for SEL-212. Additionally, the sizeDISSOLVE II trial of SEL-212 has enrolled subjects at clinical sites located in Russia and Ukraine. As a result of the patient population, patientongoing and rapidly evolving situation in the region, we may remove those patients from the trial, which as a result could potentially result in delays in the full enrollment is alsoof the study and/or the release of top-line results. The safety of our patients and investigators continues to be our utmost priority. Additionally, the COVID-19 pandemic has affected by other factors including:
| |
- | the severity of the disease under investigation; |
| |
- | the patient eligibility criteria for the studyour ability to enroll and sustain patients in question; |
| |
- | the perceived risks and benefits of the product candidate under study; |
| |
- | the availability of other treatments for the disease under investigation; |
| |
- | the existence of competing clinical trials; |
| |
- | our efforts to facilitate timely enrollment in clinical trials; |
| |
- | investigators engagement with, or enthusiasm about, the trial; |
| |
- | our payments for participating in clinical trials; |
| |
- | the patient referral practices of physicians; |
| |
- | the design of the trial; |
| |
- | the ability to monitor patients adequately during and after treatment; and |
| |
- | the proximity and availability of clinical trial sites for prospective patients. |
In addition, our clinical trials, will compete with other clinical trials for product candidates that areand could result in the same therapeutic areas as our product candidates, and this competition will reduce the number and typesinability of patients available to us, because some patients who might have opted to enroll in our trials may instead opt to enroll in a trial being conducted by one of our competitors. Since the number of qualified clinical investigators is limited, we expect to conduct some ofcomplete our clinical trials at the same clinical trial sites that some of our competitors use,trials. Additionally, we have enrolled subjects in countries where patient populations have less or limited access to COVID-19 vaccines, which will reduce the number of patients who are available forcould result in further delays in enrollment or a patient’s inability to complete our clinical trials at such clinical trial site.trials. Our inability to enroll a sufficient number of patients for our clinical trials would result in significant delays and could require us to abandon one or more clinical trials altogether. Enrollment delays in our clinical trials may result in increased development costs for our product candidates, which could cause the value of our common stock to decline and limit our ability to obtain additional financing.
We may conduct clinical trials for product candidates at sites outside the United States, and the FDA may not accept data from trials conducted in such locations or the complexity of regulatory burdens may otherwise adversely impact us.
Opening trial sites outside the United States may involve additional regulatory, administrative and financial burdens, including compliance with foreign and local requirements relating to regulatory submission and clinical trial practices. Although the FDA may accept data from clinical trials conducted outside the United States, acceptance of these data is subject to certain conditions imposed by the FDA. For example, the clinical trial must be well designed and conducted and performed by qualified investigators in accordance with good clinical practices, or GCPs, and the FDA must be able to validate the data from the trial through an onsite inspection, if necessary. Generally, the patient population for any clinical trials conducted
outside the United States must be representative of the population for which we intend to seek approval in the United States. In addition, while these clinical trials are subject to the applicable local laws, FDA acceptance of the data will be dependent upon its determination that the trials also complied with all applicable U.S. laws and regulations. Nonetheless, there can be no assurance that the FDA will accept data from trials conducted outside the United States. If the FDA does not accept the data from any trial that we conduct outside the United States, it would likely result in the need for additional clinical trials, which would be costly and time-consuming and delay or permanently halt our development of any applicable product candidates.
In addition, the conduct of clinical trials outside the United States could have a significant impact on us. RisksAdditional risks inherent in conducting international clinical trials include:
| |
- | foreign regulatory requirements that could burden or limit our ability to conduct our clinical trials; |
| |
- | increased costs and heightened supply constraints associated with the acquisition of standard of care drugs and/or combination or comparator agents for which we may bear responsibility in certain jurisdictions; |
| |
- | administrative burdens of conducting clinical trials under multiple foreign regulatory schema; |
| |
- | foreign exchange fluctuations; |
| |
- | more burdensome manufacturing, customs, shipment and storage requirements; |
| |
- | cultural differences in medical practice and clinical research; |
| |
- | lack of consistency in standard of care from country to country; |
| |
- | diminished protection of intellectual property in some countries; and |
| |
- | changes in country or regional regulatory requirements. |
•foreign regulatory requirements that could burden or limit our ability to conduct our clinical trials;
•administrative burdens of conducting clinical trials under multiple foreign regulatory schema;
•foreign exchange fluctuations;
•more burdensome manufacturing, customs, shipment and storage requirements;
•cultural differences in medical practice and clinical research;
•lack of consistency in standard of care from country to country;
•diminished protection of intellectual property in some countries; and
•changes in country or regional regulatory requirements; and
•geopolitical instability or wars in regions outside of the United States where we conduct clinical trials may impact ongoing clinical trials.
We may not be able to qualify for or obtain orphan drug designation forvarious designations from regulators that would have the potential to expedite the review process of one or more of our product candidates and even if we do receive one or more such designations there is no guarantee that they will ultimately expedite the process, or aid in our obtaining marketing approval or provide market exclusivity.
There exist several designations that we may be unablecan apply for from the FDA and other regulators that would provide us with various combinations of the potential for expedited regulatory review, certain financial incentives as well as the potential for post-approval exclusivity for a period of time. These designations include but are not limited to maintain the benefits associated with orphan drug designation, including the potentialbreakthrough therapy designation, accelerated approval, fast track status and priority review for market exclusivity.
Regulatory authoritiesour product candidates. For example, we and AskBio received orphan drug designation for SEL-302 in some jurisdictions, including the United States and Europe, may designate drugs for relatively small patient populations as orphan drugs.November 2020. We expect to seek orphan drug designationone or more of these designations for severalour current and future product candidates. There can be no assurance that any of our other product candidates will qualify for any of these designations. There can also be no assurance that any of our product candidates. Under the Orphan Drug Act of 1983,candidates that do qualify for these designations will be granted such designations or that the FDA may designatewill not revoke a designation it grants at a later date. Further, there can be no assurance that any of our product as an orphan product if it is intended to treat a rare diseasecandidates that are granted such designations will ever benefit from such designations or condition, which is generally defined as a patient population of fewer than 200,000 individuals in the United States, or a patient population of greater than 200,000 individuals in the United States, but for which there is no reasonable expectation that the costFDA would not withdraw such designations once granted. Were we to receive a designation that promised a period of developing the drug or biologic will be recovered from sales in the United States.
In the United States, orphan designation entitles a party to financial incentivesmarket exclusivity, such as opportunities for grant funding towards clinical trial costs, tax advantages and user‑fee waivers. In addition, if a product candidate that has orphan designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications, including a full BLA or full new drug application, or NDA, to market the same biologic or drug for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity or where the manufacturer is unable to assure sufficient product quantity.
The applicable exclusivity period is ten years in the European Union, but such exclusivity period can be reduced to six years if a product no longer meets the criteria for orphan designation or if the product is sufficiently profitable so that market exclusivity is no longer justified.
Even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs can be approved for the same condition. EvenFurther with respect to orphan drug status, even after an orphan drug is approved, the FDA can subsequently approve the same drug for the same condition if the FDA concludes that the later drug is clinically superior if it is shown to be safer, more effective or makes a major contribution to patient care. Orphan drug designation neither shortens the development time or regulatory review time of a drug or biologic nor gives the drug or biologic any advantage in the regulatory review or approval process.
Interim, top-line and preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publish interim, top-line or preliminary data from our clinical studies, which is based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a full analyses of all data related to the particular trial. For example, we and AskBio announced top-line data from the Phase 1 trial for SEL-399 in November 2021. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the top-line results that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Preliminary or “top-line”top-line data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, preliminary and top-line data should be viewed with caution until the final data are available. We may also disclose interim data from our clinical trials. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Adverse differences betweenInterim, top-line or preliminary or interim data and
may not be representative of final data. If final data could significantly harmis not as positive as earlier interim, top-line or preliminary we have released, our business prospects.prospects would be significantly harmed.
Further, others, including regulatory agencies, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently, which could impact the value of the particular program, the approvability or commercialization of the particular product candidate or product and our company in general. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is based on what is typically extensive information, and you or others may not agree with what we determine is the material or otherwise appropriate information to include in our disclosure. Any information we determine not to disclose may ultimately be deemed significant by you or others with respect to future decisions, conclusions, views, activities or otherwise regarding a particular product candidate or our business. If the interim,As a result, preliminary and top-line or preliminary data that we report differ from final results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain approval for, and commercialize, product candidates may be harmed, which could seriously harm our business.
The regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time consuming and inherently unpredictable, and if we are ultimately unable to obtain regulatory approval for our product candidates, our business will be substantially harmed.
The time required to obtain approval by the FDA and comparable foreign authorities is unpredictable but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion of
the regulatory authorities. In addition, approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions. We have not obtained regulatory approval for any product candidate and it is possible that SEL-212 or any other product candidates we may seek to develop in the future will ever obtain regulatory approval. Neither we nor any future collaborator is permitted to market any of our product candidates in the United States until we receive regulatory approval of a Biologics License Application, or BLA, from the FDA.
Prior to obtaining approval to commercialize a product candidate in the United States or abroad, we or our collaborators must demonstrate with substantial evidence from well-controlled clinical trials, and to the satisfaction of the FDA or foreign regulatory agencies, that such product candidates are safe and effective, or in the case of biologics, safe, pure, and potent, for their intended uses. Results from nonclinical studies and clinical trials can be interpreted in different ways. Even if we believe the nonclinical or clinical data for our product candidates are promising, such data mayshould not be sufficient to support approval by the FDA and other regulatory authorities. The FDA may also require us to conduct additional preclinical studies or clinical trials for our product candidates either prior to or post-approval, or it may object to elements of our clinical development program.
The FDA or any foreign regulatory bodies can delay, limit or deny approval of our drug or device product candidates or require us to conduct additional nonclinical or clinical testing or abandon a program for, including the following:
the FDA or comparable foreign regulatory authorities may disagree with the design or implementation of our clinical trials;
we may be unable to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that a product candidate is safe and effective for its proposed indication;
the results of clinical trials may not meet the level of statistical significance required by the FDA or comparable foreign regulatory authorities for approval;
serious and unexpected drug-related side effects experienced by participantsrelied upon in making an investment decision in our clinical trials or by individuals using drugs similar to our product candidates;
we may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks;
the FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials;
the data collected from clinical trials of our product candidates may not be acceptable or sufficient to support the submission of a BLA or other submission or to obtain regulatory approval in the United States or elsewhere, and we may be required to conduct additional clinical studies;
the FDA’s or the applicable foreign regulatory agency may disagree regarding the formulation, labeling and/or the specifications of our product candidates;
the FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; and
the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval.
Of the large number of drugs in development, only a small percentage successfully complete the FDA or foreign regulatory approval processes and are commercialized. The lengthy approval process as well as the unpredictability of future clinical trial results may result in our failing to obtain regulatory approval to market our product candidates, which would significantly harm our business, results of operations and prospects.
In addition, even if we were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited indications than we request, may not approve the price we intend to charge for our products, may grant approval contingent on the performance of costly post-marketing clinical trials, including Phase 4 clinical trials, and/or the implementation of a Risk Evaluation and Mitigation Strategy, or REMS, which may be required to assure safe use of the drug after approval. The FDA or the applicable foreign regulatory agency also may approve a product candidate for a more limited indication or patient population than we originally requested, or may approve a product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate. Any of the foregoing scenarios could materially harm the commercial prospects for our product candidates.
Any breakthrough therapy designation that we may receive from the FDA for our product candidates may not lead to a faster development or regulatory review or approval process, and it does not increase the likelihood that our product candidates will receive marketing approval.
We may in the future seek breakthrough therapy designation for some of our product candidates. A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life‑threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. For drugs that have been designated as breakthrough therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Drugs designated as breakthrough therapies by the FDA are also eligible for accelerated approval.
Designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if we believe one of our product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. The availability of breakthrough therapy designation was established with the passage of the Food and Drug Administration Safety and Innovation Act of 2012. We cannot be sure that any evaluation we may make of our product candidates as qualifying for breakthrough therapy designation will meet the FDA’s expectations. In any event, the receipt of a breakthrough therapy designation for a product candidate may not result in a faster development process, review or approval compared to drugs considered for approval under conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, even if one or more of our product candidates qualify as breakthrough therapies, the FDA may later decide that such product candidates no longer meet the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.securities.
Negative public opinion and increased regulatory scrutiny of gene therapy and genetic research may damage public perception of our product candidates or compromise our ability to conduct our business or obtain regulatory approvals for our product candidates.
Gene therapy remains a novel technology. Public perception may be influenced by claims that gene therapy is unsafe, and gene therapy may not gain the acceptance of the public or the medical community. In particular, our success will depend upon physicians specializing in the treatment of those diseases that our product candidates target and prescribing treatments that involve the use of our product candidates in lieu of, or in addition to, existing treatments they are already familiar with and for which greater clinical data may be available. More restrictive government regulations or negative public opinion would have a negative effect on our business or financial condition and may delay or impair the development and commercialization of our product candidates or demand for any products we may develop. Our product candidates, including our products that utilize viral delivery systems, could produce adverse events. Adverse events in our clinical trials or following approval of any of our product candidates, even if not ultimately attributable to our product candidates, could result in increased governmental regulation, unfavorable public perception, potential regulatory delays in the testing or approval of our product candidates, stricter labeling requirements for those product candidates that are approved and a decrease in demand for any such product candidates.
Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following marketing approval, if any.
Undesirable side effects caused by our product candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials, and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or other comparable foreign authorities.authorities and could result in decreased market acceptance of any of our product candidates, if approved. Further, therapies such as those we are developing involve unique side effects that could be exacerbated compared tomore significant than side effects from other types of therapies with singular components. Results of our clinical trials could reveal a high and unacceptable severity and prevalence of side effects. In such an event, our trials could be suspended or terminated, and the FDA or comparable foreign regulatory authorities could order us to cease further development of or deny approval of our product candidates for any or all targeted indications. For example, a patient in the Phase 1 trial of SEL-403 experienced a Grade 5 SAE (patient death) related to pneumonitis, which was deemed by the trial investigator to be probably related to ImmTOR and possibly related to the patient’s pleural mesothelioma condition, and in November 2018, the FDA placed the IND for SEL-403 on full clinical hold due to adverse events observed in the Phase 1 trial. Selecta has terminated the license of LMB-100 from NCI, effective April 9, 2019 and is no longer pursuing this product candidate.
Further, the SEL-212 multi-year clinical development program requiringof SEL-212 over many years has required multiple clinical trials and resulted in the use of different formulations of ImmTOR. While we do not believe that such differences in formulation will affect the safety or the efficacy of SEL-212, we cannot guarantee that any such formulation changes will not negatively impact the results of any clinical trials related to SEL-212, or result in a significant difference in the safety and efficacy of SEL-212.
The drug‑relateddrug-related side effects observed in our clinical trials could also affect patient enrollment in our clinical trials or the ability of any enrolled patients to complete such trials or result in potential product liability claims. Any of these occurrences may harm our business, financial condition and prospects significantly.
Additionally, if one or more of our product candidates receives marketing approval, and we or others later identify undesirable side effects caused by such products, a number of potentially significant negative consequences could result, including:
| |
- | regulatory authorities may withdraw approvals of such product; |
| |
- | regulatory authorities may require the addition of labeling statements, such as a “black box” warning or a contraindication; |
| |
- | regulatory authorities may impose additional restrictions on the marketing of, or the manufacturing processes for, the particular product; |
| |
- | we may be required to create a medication guide outlining the risks of such side effects for distribution to patients; |
| |
- | we could be sued and held liable for harm caused to patients, or become subject to fines, injunctions or the imposition of civil or criminal penalties; |
| |
- | our reputation may suffer; and |
| |
- | we could be required to develop a risk evaluation and mitigation strategy (REMS) plan to prevent, monitor and/or manage a specific serious risk by informing, educating and/or reinforcing actions to reduce the frequency and/or severity of the event. |
•regulatory authorities may withdraw approvals of such product;
•regulatory authorities may require the addition of labeling statements, such as a “black box” warning or a contraindication;
•regulatory authorities may impose additional restrictions on the marketing of, or the manufacturing processes for, the particular product;
•we may be required to create a medication guide outlining the risks of such side effects for distribution to patients;
•we could be sued and held liable for harm caused to patients, or become subject to fines, injunctions or the imposition of civil or criminal penalties;
•our reputation may suffer; and
•we could be required to develop a REMS plan to prevent, monitor and/or manage a specific serious risk by informing, educating and/or reinforcing actions to reduce the frequency and/or severity of the event.
Any of these events could prevent us from achieving or maintaining market acceptance of a particular product candidate, if approved, and could significantly harm our business, results of operations and prospects.
Risks Related to our product candidates are associated with undesirable side effects in certain patient populations, such as pediatric patients or the elderly, we may need to abandon their development or limit development to more narrow uses or subpopulations in which the undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk‑benefit perspective, any of which would harm our business.
RISKS RELATED TO OUR DEPENDENCE ON THIRD PARTIES AND MANUFACTURINGDependence on Third Parties and Manufacturing
We rely on 3SBio in China as our primary supplier of pegadricase and on other third parties for the manufacture of our product candidates for preclinical and clinical testing, and expect to continue to do so for the foreseeable future. Our reliance on third parties increases the risk that we will not have sufficient quantities of our product candidates or that such quantities may not be available at an acceptable cost, or in compliance with regulatory requirements, which could delay, prevent or impair our development or commercialization efforts.
We obtain the biologic pegadricase, a component of SEL‑212,SEL-212, primarily from 3SBio in China. Under our license agreement withthe 3SBio License, we have limited rights to manufacture pegadricase and while we have entered into a contract with a back-up supplier located outside of China, we expect to continue to rely on 3SBio as the primary supplier of pegadricase for the foreseeable future.
Any disruption in production or inability of 3SBio in China to produce adequate quantities of pegadricase to meet our needs whether as a result of a natural disaster, public health emergency, such as the novel coronavirus, failure to comply with regulatory requirements or other causes, could impair our ability to operate our business on a day‑to‑dayday-to-day basis and to continue our research and development of our future product candidates. Furthermore, since 3SBio is located in China, we are exposed to the possibility of product supply disruption and increased costs in the event of changes in the policies, laws, rules and regulations of the United States or Chinese governments, political unrest or unstable economic conditions in China. For example, trade tensions between the United States and China have been escalating in recent years. Most notably, several rounds of U.S. tariffs have been placed on Chinese goods being exported to the United States. Each of these U.S. tariff impositions against Chinese exports were followed by a round of retaliatory Chinese tariffs on U.S. exports to China. Pegadricase is subject to, and any other components we purchase from China may be subject to, these tariffs, which could increase our manufacturing costs and could make our products, if successfully developed and approved, less competitive than those of our competitors whose inputs are not subject to these tariffs.
Moreover, as a result of the COVID-19 pandemic, certain of our suppliers and CMOs in the United States, China and other countries may be affected, which could disrupt their activities. We could face difficulty sourcing key components necessary to produce supply of SEL-212, which may negatively affect our clinical development activities and our agreement with Sobi. If COVID-19 continues to impact U.S. business operations, including those of our CMOs and suppliers, we could face additional disruptions to our supply chain that could affect the supply of drug product for our preclinical studies and clinical trials. Additionally, as our CMOs are producers of drug substances and drug products, including vaccines and therapeutics, they could be compelled by a national government, or choose themselves, to shift their resources to the production of a COVID-19 vaccine and/or therapeutics for COVID-19, which could disrupt any scheduled drug substance or drug product batches we may have and may prevent us from obtaining supplies for our programs in a timely manner to meet our development timelines.
Any of these matters could materially and adversely affect our business and results of operations. Any issues related to the manufacturing lots or similar action regarding pegadricase used in preclinical studies or clinical trials could delay the studies or trials or detract from the integrity of the trial data and its potential use in future regulatory filings. In addition, manufacturing interruptions or failure to comply or maintain compliance with regulatory requirements by 3SBio could significantly delay our clinical development of potential products and reduce third‑partythird-party or clinical researcher interest and support of our proposed trials. These interruptions or failures could also impede commercialization of our future product candidates and impair our competitive position. Further, we may be exposed to fluctuations in the value of the local currency in China. Future appreciation of the local currency could increase our costs. In addition, labor costs could continue to rise asThese factors and increasing wage rates increase due to increased demand for skilled laborers and the declining availability of skilled labor declines in China.China could cause our labor costs to rise.
In addition to 3SBio, weWe rely, and expect to continue to rely, in addition to 3SBio, on other third parties for the manufacture of our product candidates for supply in preclinical studies and clinical testing,trials, as well as for commercial manufacture if any of our product candidates receive marketing approval. Our reliance on such third parties increases the risk that we will not have sufficient quantities of our product candidates on a timely basis or at all, or that such quantities will be available at an acceptable cost or quality, which could delay, prevent or impair our development or commercialization efforts. For example, we rely on third parties for the manufacture of our gene therapy preclinical materials. Gene therapy is a relatively new area for commercial biopharmaceutical development and there are a limited number of CMOs with adequate facilities and expertise in this area. As a result, we may be unable to successfully manufacture our gene therapy preclinical materials through a third party or scale up the manufacture of our gene therapy product candidates for clinical testing or commercialization, if at all.
We may be unable to establish any agreements with third‑partythird-party manufacturers on acceptable terms or at all. Evenall, and even if we are able to establish agreements with third‑party manufacturers, reliancedo rely on third‑partythird-party manufacturers entails additional risks, including the:
| |
- | •inability, failure or unwillingness of third‑party manufacturers to comply with regulatory requirements, maintain quality assurance, meet our needs, specifications or schedules or continue to supply products to us; |
| |
- | reduced control we have over product development, including with respect to our lead product candidate, due to our reliance on such third‑party manufacturers, |
| |
- | breach of manufacturing agreements by the third‑party manufacturers; |
| |
- | misappropriation or disclosure of our proprietary information, including our trade secrets and know‑how; |
| |
- | relationships that the third-party manufacturer may have with others, some of which may be our competitors, and, if it does not successfully carry out its contractual duties, does not meet expectations, experiences work stoppages, or needs to be replaced, we may need to enter into alternative arrangements, which may not be available, desirable or cost‑effective; and |
| |
- | termination or nonrenewal of agreements by third‑party manufacturers at times that are costly or inconvenient for us. |
The facilities used by our contract manufacturers to manufacturecomply with regulatory requirements, maintain quality assurance, meet our needs, specifications or schedules or continue to supply products to us;
•reduced control we have over product candidates must be approveddevelopment, including with respect to our lead product candidate, due to our reliance on such third-party manufacturers,
•breach of manufacturing agreements by the FDA pursuant to inspections that will be conducted after we submit our marketing application to the FDA. We do not control the manufacturing processthird-party manufacturers;
•misappropriation or cGMPs, for manufacturedisclosure of our product candidates. Ifproprietary information, including our trade secrets and know-how;
•relationships that the third-party manufacturer may have with others, some of which may be our competitors, and, if it does not successfully carry out its contractual duties, does not meet expectations, experiences work stoppages, or needs to be replaced, we may need to enter into alternative arrangements, which may not be available, desirable or cost-effective; and
•termination or nonrenewal of agreements by third-party manufacturers at times that are costly or inconvenient for us.
Additionally, if our contract manufacturers cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA or others, they will not be able to secure and/or maintain regulatory approval for their manufacturing facilities. In addition, we have no control over the ability of our contract manufacturers or suppliers to maintain adequate quality control, quality assurance and qualified personnel. If the FDA or a comparable foreign regulatory authority does not approve these facilities for the manufacture of our product candidates or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved. Our failure, or the failure of our third‑partythird-party manufacturers, to comply with applicable regulations could result in sanctions being imposed on us, including clinical holds, fines, injunctions, civil penalties, delays, suspension or withdrawal of approvals, license revocations, seizures or recalls of product candidates or products, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect supplies of our products. In addition, there are a limited number of manufacturers that operate under cGMP regulations that might be capable of manufacturing our products. Therefore, our product candidates and any future products that we may develop may compete with other products for access to manufacturing facilities. Any failure to gain access to these limited manufacturing facilities could severely impact the clinical development, marketing approval and commercialization of our product candidates.
Any performance failure on the part of our existing or future manufacturers could delay clinical development or marketing approval. We do not currently have arrangements in place for redundant supply or a second source for required raw materials used in the manufacture of our product candidates or for the manufacture of finished product. Moreover, we often rely on one CMO to produce multiple product components. For instance, one of our CMOs produces several polymers used in our ImmTOR platform. If our current CMOs cannot perform as agreed, we may be required to replace such manufacturers and we may be unable to replace them on a timely basis or at all. Our current and expected future dependence upon others for the manufacture of our product candidates or products could delay, prevent or impair our development and commercialization efforts.
The novel coronavirus outbreak could adversely impact our business, financial condition and results of operations.
In December 2019, a strain of novel coronavirus surfaced in Wuhan, China. In January 2020, the World Health Organization declared the novel coronavirus outbreak a “Public Health Emergency of International Concern” and the U.S. Department of State instructed travelers to avoid all nonessential travel to China. The coronavirus has impacted the global economy and may
impact our operations, including the potential interruption of our clinical trial activities and our supply chain. As a result of the outbreak, certain of our suppliers and CMOs in China may be affected, which could disrupt their activities. We could therefore face difficulty sourcing key components necessary to produce supply of SEL-212, which may negatively affect our clinical development activities. While we have a back-up supplier for SEL-037 in the United States, they may not have capacity in a timely manner for our projects and they may cost more, which could affect our results of operations and financial position. If the coronavirus further impacts US business operations, including our CMOs and suppliers, we could face additional disruption to our supply chain that could affect the supply of drug product for the clinical trials. In addition, the coronavirus outbreak could delay enrollment in our clinical trials due to prioritization of hospital resources toward the outbreak, and some patients may not be able to comply with clinical trial protocols if quarantines impede patient movement or interrupt healthcare services.
At this point in time, there is significant uncertainty relating to the potential effect of the novel coronavirus on our business. Infections may become more widespread, including in countries where we are conducting clinical trials, and manufacturing closures and travel restrictions may remain or worsen, all of which would have a negative impact on our business, financial condition and results of operations.
Our existing collaborations are important to our business, and future licenses may also be important to us. If we are unable to maintain any of theseour existing collaborations, or if these arrangements are not successful, or we are unable to enter into future licenses, our business could be adversely affected.
We have entered into collaborations with other parties, including pharmaceutical and biotechnology companies and universities, to develop products based on our ImmTOR platform, and such collaborations and licensing arrangements currently represent a significant portion of our product pipeline and are expected to represent a larger portion of our pipeline in the future. Certain of our collaborations have provided us with important funding for some of our development programs and we expect to receive additional funding under collaborations in the future although not all of our collaborations may result in funding to the Company,us, and certain collaborations, licenses and agreements may result in increased expenditures by the Company.us. Our existing collaborations, and any future collaborations, we enter into, may pose a number of risks, including the following:
| |
- | collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| |
- | collaborators may not perform their obligations as expected; |
| |
- | •collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; •collaborators may not perform their obligations as expected; •collaborators may not pursue development and commercialization of any product candidates that achieve regulatory approval or may elect not to continue or renew development or commercialization programs based on preclinical or clinical trial results, changes in the collaborators’ strategic focus or available funding, or external factors, such as an acquisition, that divert resources or create competing priorities; |
| |
- | collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
| |
- | collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours, which may cause collaborators to cease to devote resources to the commercialization of our product candidates; |
| |
- | a collaborator with marketing and distribution rights to one or more of our product candidates that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of such product or products; |
| |
- | disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development, might cause delays or termination of the research, development or commercialization of product candidates, might lead to additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration, any of which would be time‑consuming and expensive; |
| |
- | collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| |
- | collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability; |
| |
- | collaborations may be terminated for the convenience of the collaborator or for our failure to comply with our obligations under existing or future collaborations and, if terminated, we would potentially lose the right to pursue further development or commercialization of the applicable product candidates; |
| |
- | collaborators may learn about our technology and use this knowledge to compete with us in the future; |
| |
- | there may be conflicts between different collaborators that could negatively affect those collaborations and potentially others; and |
| |
- | the number and type of our collaborations could adversely affect our attractiveness to future collaborators or acquirers. |
If our collaborations do not result in the successful development and commercialization of any product candidates that achieve regulatory approval or may elect not to continue or renew development or commercialization programs based on preclinical or clinical trial results, changes in the collaborators’ strategic focus or available funding, or external factors, such as an acquisition, that divert resources or create competing priorities;
•collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing;
•collaborators could independently develop, or develop with third parties, products that compete directly or if oneindirectly with our product candidates, which may cause collaborators to cease to devote resources to the commercialization of our collaborators terminates its agreementproduct candidates;
•a collaborator with us, wemarketing and distribution rights to one or more of our product candidates that achieve regulatory approval may not receive any futurecommit sufficient resources to the marketing and distribution of such product or products;
•disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development, might cause delays or termination of the research, and development funding or milestone or royalty payments under such collaborations. If we do not receive the funding we expect under these agreements, our continued developmentcommercialization
of product candidates, might lead to additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration, any of which would be time-consuming and expensive;
•collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation;
•collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability;
•collaborations may be delayedterminated and we may need additional resourceswould potentially lose the right to develop additional product candidates. All of the risks relating to product development, regulatory approval and commercialization described in this Annual Report on Form 10-K also apply to the activities of our therapeutic program collaborators and there can be no assurance that our collaborations will produce positive results or successful products on a timely basis or at all.
Additionally, subject to its contractual obligations to us, if one of our collaborators is involved in a business combination or otherwise changes its business priorities, the collaborator might deemphasize or terminate thepursue further development or commercialization of anythe applicable product candidate licensedcandidates as well as have difficulty entering into a similar collaboration where the potential collaborator is aware of the prior termination;
•collaborators may learn about our technology and use this knowledge to it by us. If onecompete with us in the future;
•there may be conflicts between different collaborators that could negatively affect those collaborations and potentially others; and
•the number and type of our collaborations could adversely affect our attractiveness to future collaborators terminates its agreement with us, we may find it more difficult to attract new collaborators and the perception of our business in the business and financial communities, and our stock price, could be adversely affected. In addition, we have a limited number of collaborations and if our relationship with any one or more of such collaborators were to cease, our business would be harmed as a result. For example, MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. In December 2019, we entered into the MIT Amendment. Pursuant to the MIT Amendment, the provision of the MIT License under which we were obligated to initiate a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT Agreement. The parties agreed to negotiate in good faith to enter into a future amendment to the MIT License after we provide MIT with an amended diligence plan. However, if we are unable to reach an agreement with MIT regarding an acceptable amendment of the MIT License and if we are unable to cure the breach, there could be a material adverse effect on our business. See “Business - Licenses and Collaborations - Massachusetts Institute of Technology.”acquirers.
We are actively exploring licenses and other strategic collaborations with additional pharmaceutical and biotechnology companies for development and potential commercialization of therapeutic products. However, we face significant competition in seeking appropriate collaborators. If we are unable to reach agreements with suitable collaborators on a timely basis, on acceptable terms, or at all, we may not be able to access specific antigens that would be suitable to development with our technology, have to curtail the development of a product candidate, reduce or delay its development program or one or more of our other development programs, delay its potential commercialization or reduce the scope of any sales or marketing activities, or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to fund and undertake development or commercialization activities on our own, we may need to obtain additional expertise and additional capital, which may not be available to us on acceptable terms or at all. If we fail to enter into collaborations and do not have sufficient funds or expertise to undertake the necessary development and commercialization activities, we may not be able to further develop our product candidates or bring them to market or continue to develop our programs, and our business may be materially and adversely affected.
We rely, and expect to continue to rely, on third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including by failing to meet deadlines for the completion of such trials.
We rely, and expect to continue to rely, on third parties, such as CROs, clinical data management organizations, medical institutions and clinical investigators, to conduct and manage our clinical trials, including our Phase 2 andongoing Phase 3 DISSOLVE clinical program for SEL-212, consisting of the DISSOLVE I and DISSOLVE II trials, which we have agreed to continue to run on behalf of SEL‑212Sobi, and for our other product candidates. We also expect to rely on other third parties to store and distribute drug supplies for our clinical trials.
Our relianceWhile we rely on these third parties for research and development activities, will reduce our control over these activities but does not relieve us of our responsibilities. For example, we remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the trial. Moreover, the FDA requires us to comply with GCP regulations, for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, safety and welfare of trial participants are protected. Other countries’ regulatory agencies also have requirements for clinical trials. Regulatory authorities enforce these GCPs through periodic inspections of trial sponsors, principal investigators and trial sites. If we or any of our CROs or third-party contractors fail to comply with applicable GCPs, the data generated in our clinical trials may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical trials complies with GCP regulations. In addition, our clinical trials must be conducted with
product produced under cGMP regulations. Our failure to comply with these regulations may require us to repeat clinical trials, which would delay the regulatory approval process. We also are required to register ongoing clinical trials and post the results of completed clinical trials on a government‑sponsoredgovernment-sponsored database, ClinicalTrials.govwww.ClinicalTrials.gov, within specified timeframes. Failure to do so can result in fines, adverse publicity, and civil and criminal sanctions.
Furthermore, these third parties may also have relationships with other entities, some of which may be our competitors. If these third parties do not successfully carry out their contractual duties, do not comply with confidentiality obligations, do not meet expected deadlines, experience work stoppages, terminate their agreements with us or need to be replaced, or do not conduct our clinical trials in accordance with regulatory requirements or our stated protocols, we may need to enter into new arrangements with alternative third parties, which could be difficult, costly or impossible, and our clinical trials may be extended, delayed or terminated, or may need to be repeated. If any of the foregoing occur, we may not be able to obtain, or may be delayed in obtaining, marketing approvals for our product candidates or in commercializing our product candidates.
We have no experience manufacturing our product candidates atfor commercial scale,use, and if we decide to establish our own commercial manufacturing facility, we cannot assure you that we can manufacture our product candidates in compliance with regulations at a cost or in quantities necessary to make them commercially viable.
We have a pilot manufacturing facility at our Watertown, Massachusetts location where we conduct process development, scale‑upscale-up activities and the manufacture of ImmTOR product candidates for preclinical use. We rely on our scaled equipment installed at our CMOs for the manufacture of the clinical supply of all of our product candidates. If our facility, or our CMOs’ facilities, were damaged or destroyed, or otherwise subject to disruption, it would require substantial lead‑time to replace our manufacturing capabilities. In such event, we would be forced to identify and rely entirely on alternative third‑party contract manufacturers for an indefinite period of time. Any disruptions or delays at our facility or its failure to meet regulatory compliance would impair our ability to develop and commercialize our product candidates, which would adversely affect our business and results of operations.
In addition, the FDA and other comparable foreign regulatory agencies must, pursuant to inspections that are conducted after submitting a BLA or relevant foreign marketing submission, confirm that the manufacturing processes for the product candidate meet cGMP regulations. We do not currently have any of our own manufacturing facilities that meet the FDA’s cGMP requirements for the production of any product candidates used in humans, and rely on our CMOs for clinical production.
We may choosecurrently have no plans to establish aour own commercial manufacturing facilityfacilities and we will continue to rely on our partnership with CMOs who are currently producing ImmTOR at the commercial scale for SEL-212 using our product candidates for production at a commercial scale. However, we have no experience in commercial‑scale manufacturingproprietary process and equipment.
Risks Related to Commercialization of our product candidatesProduct Candidates and this activity will require substantial additional funds and additional qualified employees. We may not be able to develop commercial‑scale manufacturing facilities that are adequate to produce materials for additional later‑stage clinical trials or commercial use.
The equipment and facilities employed in the manufacture of pharmaceuticals are subject to stringent qualification requirements by regulatory agencies, including validation of such facilities, equipment, systems, processes and analytics. We may be subject to lengthy delays and expense in conducting validation studies, if we can meet the requirements at all.
RISKS RELATED TO COMMERCIALIZATION OF OUR PRODUCT CANDIDATES AND OTHER LEGAL COMPLIANCE MATTERSLegal Compliance Matters
Even if any of our product candidates receives marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third‑partythird-party payors and others in the medical community necessary for commercial success.
If any of our product candidates receives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, patients, third‑partythird-party payors and others in the medical community. If our product candidates do not achieve an adequate level of acceptance, we may not generate significant product revenues and we may not become profitable. For example, even if the statistical results from our Phase 2 head-to-head (COMPARE) trial vs. KRYSTEXXA favor SEL-212 and SEL-212 receives marketing approval, the drug may fail to gain market acceptance from physicians, patients, third-party payors and others in the medical community who may continue to favor KRYSTEXXA. The degree of market acceptance of our product candidates, if any,approved for commercial sale, will depend on a number ofseveral factors, including:
| |
- | their efficacy, safety and other potential advantages compared to alternative treatments; |
| |
- | the clinical indications for which our product candidates are approved; |
| |
- | our ability to offer them for sale at competitive prices; |
| |
- | their convenience and ease of administration compared to alternative treatments; |
| |
- | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| |
- | the strength of marketing and distribution support; |
•the efficacy, safety and potential advantages compared to alternative treatments;
| |
- | the availability of third‑party coverage and adequate reimbursement for our product candidates; |
| |
- | the prevalence and severity of their side effects and their overall safety profiles; |
| |
- | any restrictions on the use of our product candidates together with other medications; |
| |
- | interactions of our product candidates with other medicines patients are taking; |
| |
- | our ability to create awareness with patients and physicians about the harmful effects of uric acid deposits; |
| |
- | the timing of market introduction of any approved product candidates as well as competitive products and other therapies; |
| |
- | inability of certain types of patients, particularly with respect to certain rare diseases or conditions, to take our product candidates; |
| |
- | their ability to remain attractive in the event of changing treatment guidelines; |
| |
- | adverse publicity about the product or favorable publicity about competitive products; and |
| |
- | potential product liability claims. |
•the convenience and ease of administration compared to alternative treatments;
•product labeling or product insert requirements of the FDA or foreign regulatory authorities, including any limitations or warnings contained in a product’s approved labeling, including any black box warning or REMS;
•the willingness of the target patient population to try new treatments and of physicians to prescribe these treatments;
•our ability to hire and retain a sales force in the United States;
•the strength of marketing and distribution support;
•the availability of third-party coverage and adequate reimbursement for our product candidates, once approved;
•the prevalence and severity of any side effects; and
•any restrictions on the use of our products together with other medications.
We currently have no sales organization.organization and expect to rely on Sobi for the marketing and sale of SEL-212, if approved. If we are unable to establish effective sales, marketing and distribution capabilities, or enter into agreements with third parties with such capabilities, we may not be successful in commercializing our product candidates if and when they are approved.
We do not have a sales or marketing infrastructure and have no experience in the sale, marketing or distribution of pharmaceutical products. To achieve commercial success for any product candidate for which we obtain marketing approval, we will need to establish a sales and marketing organization or make arrangements with third parties to perform sales and marketing functions and we may not be successful in doing so.
In We expect to rely on Sobi for the future,marketing and sale of SEL-212, if approved. For our other product candidates, we expect to build a focused sales and marketing infrastructure to market or co‑promoteco-promote our product candidates in the United States and potentially elsewhere, if and when they are approved. There are risks involved with establishing our own sales, marketing and distribution capabilities. For example, recruiting and training a sales force is expensive and time‑consumingtime-consuming and could delay any product launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Outside the United States, we may rely on third parties to sell, market and distribute our product candidates. We may not be successful in entering into arrangements with such third parties or may be unable to do so on terms that are favorable to us. In addition, our product revenue and our profitability, if any, may be lower if we rely on third parties for these functions than if we were to market, sell and distribute any products that we develop ourselves. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales, marketing and distribution capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
We face substantial competition, including from biosimilars, which may result in others discovering, developing or commercializing competing products before or more successfully than we do.
The development and commercialization of new drug and biologic products and technologies is highly competitive and is characterized by rapid and substantial technological development and product innovations. We are aware that pharmaceutical and biotechnology companies, including Horizon Pharma plc, offer or are pursuing the development of pharmaceutical products or technologies that may address one or more indications that our product candidates target, as well as smaller, early-stage companies, that offer or are pursuing the development of pharmaceutical products or technologies that may address one or more indications that our product candidates target. We face competition with respect to our current product candidates, and will face competition with respect to any product candidates that we may seek to develop or commercialize in the future, from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide.
Many of the companies against which we are competing or against which we may compete in the future have significantly greater financial resources, established presence in the market and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and reimbursement for product candidates and in marketing approved products than we do. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors.
These third parties compete with us in recruiting and retaining qualified scientific, sales and marketing and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market, especially for any competitor developing a competing immunomodulating therapeutic that will likely share our same regulatory approval requirements. In addition, our ability to compete may be affected in many cases by insurers or other third‑partythird-party payors seeking to encourage the use of generic or biosimilar products.
We expect the product candidates we develop will be regulated as biological products, or biologics, and therefore they may be subject to competition sooner than anticipated.
The Biologics Price Competition and Innovation Act of 2009 (BPCIA)BPCIA was enacted as part of the Affordable Care Act to establish an abbreviated pathway for the approval of biosimilar and interchangeable biological products. The regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable” based on its similarity to an approved biologic. Under the BPCIA, an application for a biosimilar product cannot be approved by the FDA until 12 years after the reference product was approved under a BLA. The law is complex and is still being interpreted and implemented by the FDA. AsFDA, and as a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when such processes intended to implement the BPCIA may be fully adopted by the FDA,However, any such processes could have a material adverse effect on the future commercial prospects for our biological products.
We believe that any product candidate approved in the United States as a biological product under a BLA should qualify for the 12-year period of exclusivity. However, there is a risk that this exclusivity could be shortened due to congressionalCongressional action or otherwise, or that the FDA will not consider the subject product candidates to be reference products for competing products, potentially creating the opportunity for generic competition sooner than anticipated. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of the reference products in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing.
Even if we are able to commercialize any product candidates, the products may become subject to unfavorable pricing regulations or third‑partythird-party coverage or reimbursement policies, any of which would have a material adverse effect on our business.
Significant uncertainty exists as to the coverage and reimbursement status of any product candidates for which we obtain regulatory approval, especially novel products like our gene therapy product candidates, and may be particularly difficult because of the higher prices associated with gene therapy product candidates. Our ability to commercialize any product candidates successfully will depend, in part, on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third‑partythird-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels.
Obtaining and maintaining adequate reimbursement for our products may be difficult. We cannot be certain if and when we will obtain an adequate level of reimbursement for our products by third‑partythird-party payors. Even if we do obtain adequate levels of reimbursement, third‑partythird-party payors, such as government or private healthcare insurers, carefully review and increasingly question the coverage of, and challenge the prices charged for, products. Government authorities and third‑partythird-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly, third‑partyThird-party payors are requiringoften require that pharmaceutical companies provide them with predetermined discounts from list prices and are challenging the prices charged for products. We may also be required to conduct expensive pharmacoeconomic studies to justify coverage and reimbursement or the level of reimbursement relative to other therapies. Some third-party payors may require pre-approval of coverage for new and innovative therapies, such as our product candidates, before they will provide reimbursement. If coverage and reimbursement are not available or reimbursement is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval.
There may be significant delays in obtaining reimbursement for newly approved products, and coverage may be more limited than the purposes for which the product is approved by the FDA or similar regulatory authorities outside of the United States. Moreover, eligibility for reimbursement does not imply that a product will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for new products,
if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the product and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost products and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of products from countries where they may be sold at lower prices than in the
United States. Our inability to promptly obtain coverage and adequate reimbursement rates from both government‑fundedgovernment-funded and private payors for any approved products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
The regulations that govern marketing approvals, pricing, coverage and reimbursement for new products vary widely from country to country. Current and future legislation may significantly change the approval requirements in ways that could involve additional costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a product before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control, including possible price reductions, even after initial approval is granted. As a result, we might obtain marketing approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product, possibly for lengthy time periods, and negatively impact the revenues we are able to generate from the sale of the product in that country. Adverse pricing limitations may hinder our ability to recoup our investment in one or more product candidates, even if our product candidates obtain marketing approval. There can be no assurance that our product candidates, if they are approved for sale in the United States or in other countries, will be considered medically necessary for a specific indication or cost‑effective,cost-effective, or that coverage or an adequate level of reimbursement will be available.
Moreover, there has recently beenis heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several Congressional inquiries and proposed bills designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products. There can be no assurance that our product candidates, if approved for sale in the United States or in other countries, will not be subject to heightened governmental scrutiny, unfavorable regulatory inquiry or action, or congressionalCongressional inquiry.
Product liability lawsuits against us could cause us to incur substantial liabilities and limit commercialization of any products that we may develop.
We face an inherent risk of product liability exposure related to the testing of our product candidates in clinical trials and will face an even greater risk if we commercially sell any products that we may develop. If we cannot successfully defend ourselves against claims that our product candidates or products caused injuries, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
| |
- | regulatory investigations, product recalls or withdrawals, or labeling, marketing or promotional restrictions; |
| |
- | decreased demand for any product candidates or products that we may develop; |
| |
- | injury to our reputation and significant negative media attention; |
| |
- | loss of clinical trial participants or increased difficulty in enrolling future participants; |
| |
- | significant costs to defend the related litigation or to reach a settlement; |
| |
- | substantial payments to trial participants or patients; |
| |
- | reduced resources of our management to pursue our business strategy; and |
| |
- | the inability to commercialize any products that we may develop. |
| |
- | distraction of management’s attention from our primary business; |
| |
- | substantial monetary awards to patients or other claimants; |
•regulatory investigations, product recalls or withdrawals, or labeling, marketing or promotional restrictions;
•decreased demand for any product candidates or products that we may develop;
•injury to our reputation and significant negative media attention;
•loss of clinical trial participants or increased difficulty in enrolling future participants;
•significant costs to defend the related litigation or to reach a settlement;
•substantial payments to trial participants or patients;
•loss of revenue;
•reduced resources of our management to pursue our business strategy;
•the inability to commercialize any products that we may develop;
•distraction of management’s attention from our primary business; and
•substantial monetary awards to patients or other claimants;
We maintain general liability, product liability and umbrella liability insurance. Our existing insurance coverage may not fully cover potential liabilities that we may incur. We may need to increase our insurance coverage as we expand our clinical trials or if we commence commercialization of our product candidates. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. A successful product liability claim or series of claims brought against us could cause our share price to decline and, if judgments exceed our insurance coverage, could adversely affect our results of operations and business, including preventing or limiting the commercialization of any product candidates we develop.
Failure to obtain marketing approval in international jurisdictions would prevent our product candidates from being marketed abroad.
Although we do not have any current plans to market and sell our products in other jurisdictions outside of the United States, we may decide to do so in the future and either we or our collaborators would need to obtain separate marketing approvals and comply with numerous and varying regulatory requirements. The approval procedure varies among countries and can involve
additional testing. The time required to obtain approval in foreign countries may differ substantially from that required to obtain FDA approval. Clinical trials conducted in one country may not be accepted by regulatory authorities in other countries. The regulatory approval process outside the United States generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States, it is required that the product candidate be approved for reimbursement before the product candidate can be approved for sale in that country. We or our collaborators may not obtain approvals for our product candidates from regulatory authorities outside the United States on a timely basis, if at all. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions, or by the FDA. However, a failure or delay in obtaining regulatory approval in one country may have a negative effect on the regulatory process in others. We may not be able to file for marketing approvals and may not receive necessary approvals to commercialize our product candidates in any market.
Our relationships with healthcare providers, customers and third‑partythird-party payors will be subject to applicable anti‑kickback,anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties,
exclusion from government healthcare programs, contractual damages, reputational harm and diminished profits and future earnings.
Arrangements with physicians, others who may be in a position to generate business for us, and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that constrain the business or financial arrangements and relationships through which we market, sell and distribute any products for which we obtain marketing approval. Restrictions under applicable federal and state healthcare laws and regulations include the following:
| |
- | the federal Anti‑Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service for which payment may be made under a federal healthcare program such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation; |
| |
- | the federal False Claims Act, which impose criminal and civil penalties against individuals or entities for knowingly presenting, or causing to be presented, to the federal government claims for payment that are false or fraudulent. Private individuals (e.g., whistleblowers) can bring these actions on behalf of the government; in addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti‑Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act; |
| |
- | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which imposes criminal and civil liability for, among other things, executing or attempting to execute a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation; |
| |
- | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and its implementing regulations, which also imposes obligations, including mandatory contractual terms, on certain types of people and entities with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| |
- | the federal Physician Payments Sunshine Act, which requires applicable manufacturers of certain products for which payment is available under a federal healthcare program to report annually to the government information related to certain payments or other “transfers of value” made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain other health care professionals beginning in 2022, and teaching hospitals, as well as ownership and investment interests held by the physicians and their immediate family members; |
| |
- | analogous state laws and regulations, such as state anti‑kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by third‑party payors, including private insurers; and requirements to comply with federal and pharmaceutical industry compliance guidelines; |
| |
- | state data privacy and price transparency laws, many of which differ from each other in significant ways and often are broader than and not preempted by HIPAA or the Sunshine Act, thus complicating compliance efforts; by way of example, the California Consumer Privacy Act, or CCPA, which went into effect January 1, 2020, among other things, creates new data privacy obligations for covered companies and provides new privacy rights to California residents, including the right to opt out of certain disclosures of their information. The CCPA also creates a private right of action with statutory damages for certain data breaches, thereby potentially increasing risks associated with a data breach. Although the law includes limited exceptions, including for “protected health information” maintained by a covered entity or business associate, it may regulate or impact our processing of personal information depending on the context; and |
•the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service for which payment may be made under a federal healthcare program such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation;
| |
- | similar healthcare laws and regulations in the European Union and other jurisdictions, including reporting requirements detailing interactions with and payments to healthcare providers and laws governing the privacy and security of certain protected information, such as the General Data Protection Regulation, or GDPR, which imposes obligations and restrictions on the collection and use of personal data relating to individuals located in the European Union (including health data); in addition, the United Kingdom leaving the E.U. could also lead to further legislative and regulatory changes. It remains unclear how the United Kingdom data protection laws or regulations will develop in the medium to longer term and how data transfer to the United Kingdom from the E.U. will be regulated, especially following the United Kingdom's departure from the E.U. on January 31, 2020 without a deal. However, the United Kingdom has transposed the GDPR into domestic law with the Data Protection Act 2018, which remains in force following the United Kingdom's departure from the E.U. |
•HIPAA which imposes criminal and civil liability for, among other things, executing or attempting to execute a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation;
•HIPAA, as amended by HITECH and its implementing regulations, which also imposes obligations, including mandatory contractual terms, on certain types of people and entities with respect to safeguarding the privacy, security and transmission of individually identifiable health information;
•the federal Physician Payments Sunshine Act, which requires applicable manufacturers of certain products for which payment is available under a federal healthcare program to report annually to the government information related to certain payments or other “transfers of value” made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain other health care professionals beginning in 2022, and teaching hospitals, as well as ownership and investment interests held by the physicians and their immediate family members;
•analogous state laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by third-party payors, including private insurers; and requirements to comply with federal and pharmaceutical industry compliance guidelines;
•state data privacy and price transparency laws, many of which differ from each other in significant ways and often are broader than and not preempted by HIPAA or the Sunshine Act, thus complicating compliance efforts; by way of example, the California Consumer Privacy Act, or CCPA, which went into effect January 1, 2020, among other things, creates new data privacy obligations for covered companies and provides new privacy rights to California residents, including the right to opt out of certain disclosures of their information. The CCPA also creates a private right of action with statutory damages for certain data breaches, thereby potentially increasing risks associated with a data breach. Although the law includes limited exceptions, including for “protected health information” maintained by a covered entity or business associate, it may regulate or impact our processing of personal information depending on the context; and
•similar healthcare laws and regulations in the European Union and other jurisdictions, including reporting requirements detailing interactions with and payments to healthcare providers and laws governing the privacy and security of certain protected information, such as the General Data Protection Regulation, or GDPR, which imposes obligations and restrictions on the collection and use of personal data relating to individuals located in the European Union (including health data); in addition, the United Kingdom leaving the EU could also lead to further legislative and regulatory changes. It remains unclear how the United Kingdom data protection laws or regulations will develop in the medium to longer term and how data transfer to the United Kingdom from the E.U. will be regulated. However, the United Kingdom has transposed the GDPR into domestic law with the Data Protection Act 2018, which remains in force following the United Kingdom’s departure from the EU
Efforts to ensure that our business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices, including our relationships with physicians and other healthcare providers, some of whom may recommend, purchase and/or prescribe our product candidates, if approved, may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or
any other governmental laws and regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion of products from government funded healthcare programs, such as Medicare and Medicaid, disgorgement, contractual damages, reputational harm, diminished profits and the curtailment or restructuring of our operations. If any of the physicians or other healthcare providers or entities with whom we expect to do business is found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs. The risk of our being found in violation of these laws is increased by the fact that many of them have not been fully interpreted by the regulatory authorities or the courts, and their provisions are open to a variety of interpretations.
Recently enacted and future legislation may increase the difficulty and cost for us to obtain marketing approval of and commercialize our product candidates and affect the prices we may obtain.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of our product candidates, restrict or regulate post‑approvalpost-approval activities and affect our ability to profitably sell any product candidates for which we obtain marketing approval.
For example, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or collectively the ACA, is a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms.
Among the provisions of the ACA of importance to our potential product candidates are the following:
| |
- | an annual, nondeductible fee payable by any entity that manufactures or imports specified branded prescription drugs and biologic agents; |
| |
- | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program; |
| |
- | a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; |
| |
- | extension of manufacturers’ Medicaid rebate liability to individuals enrolled in Medicaid managed care organizations; |
| |
- | expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program; |
| |
- | a new requirement to annually report drug samples that manufacturers and distributors provide to physicians; and |
| |
- | a new Patient‑Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. |
Since its enactment, there have been judicial and Congressional challenges to certain aspects of the ACA. By way of example, the Tax Cuts and Jobs Acts, or the Tax Act, was enacted, which, among other things, removes penalties for not complying with the individual mandate to carry health insurance. On December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, ruled that the individual mandate is a critical and inseverable feature of the ACA, and therefore, because it was repealed as part of the Tax Act, the remaining provisions of the ACA are invalid as well. On December 18, 2019, the U.S. Court of Appeals for the 5th Circuit ruled that the individual mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining provisions of the ACA are invalid as well. It is unclear how these decisions, subsequent appeals, if any, and other efforts to challenge, repeal or replace the ACA will impact the ACA or our business. We
cannot predict the ultimate content, timing or effect of any healthcare reform legislation or the impact of potential legislation on us.
We expect that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in additional reductions in Medicare and other healthcare funding, more rigorous coverage criteria, new payment methodologies and additional downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our products.
Even if we receive regulatory approval for any of our product candidates, we will be subject to ongoing obligations and continued regulatory review, which may result in significant additional expense. Additionally, any of our product candidates, if approved, could be subject to post‑marketing restrictions or withdrawal from the market, and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unexpected problems with our products.
Any product candidate for which we obtain marketing approval, along with the manufacturing processes, post‑approval clinical data, labeling, advertising and promotional activities for such product, will be subject to the continual requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post‑marketing information and reports, registration and listing requirements, cGMP requirements relating to manufacturing, quality control, quality assurance and corresponding maintenance of records and documents, requirements regarding the distribution of samples to physicians and recordkeeping. We and our contract manufacturers will also be subject to continual review and periodic inspections to assess compliance with cGMP. Accordingly, we and others with whom we work must continue to expend time, money and effort in all areas of regulatory compliance, including manufacturing, production and quality control.
Even if marketing approval of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the product may be marketed or to the conditions of approval, including the requirement to implement a risk evaluation and mitigation strategy, or REMS, which could include requirements for a medication guide, physician communication plans or additional elements to assure safe use, such as restricted distribution methods, patient registries and other risk mitigation tools. If any of our product candidates receives marketing approval, the accompanying label may limit the approved use of our product, which could limit sales of the product.
The FDA may also impose requirements for costly post‑marketing studies or clinical trials and surveillance to monitor the safety or efficacy of our approved products. The FDA closely regulates the post‑approval marketing and promotion of drugs and biologics to ensure they are marketed only for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on manufacturers’ communications regarding off‑label use, and if we market our products outside of their approved indications, we may be subject to enforcement action for off‑label marketing. Violations of the FDA’s restrictions relating to the promotion of prescription products may also lead to investigations alleging violations of federal and state health care fraud and abuse laws, as well as state consumer protection laws.
In addition, if a regulatory agency or we later discover previously unknown problems with our products, such as adverse events of unexpected severity or frequency or problems with manufacturers or manufacturing processes, the regulatory agency may impose restrictions on the products or us, including requiring withdrawal of the product from the market. Any failure to comply with applicable regulatory requirements may yield various results, including:
| |
- | litigation involving patients taking our products; |
| |
- | restrictions on such products, manufacturers or manufacturing processes; |
| |
- | restrictions on the labeling or marketing of a product; |
| |
- | restrictions on product distribution or use; |
| |
- | requirements to conduct post‑marketing studies or clinical trials; |
| |
- | withdrawal of products from the market; |
| |
- | suspension or termination of ongoing clinical trials; |
| |
- | refusal to approve pending applications or supplements to approved applications that we submit; |
| |
- | fines, restitution or disgorgement of profits or revenues; |
| |
- | suspension or withdrawal of marketing approvals; |
| |
- | damage to relationships with existing and potential collaborators; |
| |
- | unfavorable press coverage and damage to our reputation; |
| |
- | refusal to permit the import or export of our products; |
| |
- | product seizure or detention; |
| |
- | imposition of civil or criminal penalties. |
Noncompliance with other requirements in foreign jurisdictions regarding safety monitoring or pharmacovigilance can also result in significant financial penalties. Similarly, failure to comply with U.S. and foreign regulatory requirements regarding the development of products for pediatric populations and the protection of personal health information can also lead to significant penalties and sanctions.
Any government investigation of alleged violations of law could require us to expend significant time and resources in response, and could generate negative publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to commercialize and generate revenues. If regulatory sanctions are applied or if regulatory approval is withheld or withdrawn, the value of our company and our operating results will be adversely affected.
The FDA’s and other regulatory authorities’ policies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability, which would adversely affect our business, prospects, financial condition and results of operations.
We also cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. For example, certain policies of the current administration may impact our business and industry. Namely, the Trump administration has taken several executive actions, including the issuance of a number of Executive Orders, that could impose significant burdens on, or otherwise materially delay, FDA’s ability to engage in routine regulatory and oversight activities such as implementing statutes through rulemaking, issuance of guidance, and review and approval of marketing applications. It is difficult to predict how these Executive Orders will be implemented, and the extent to which they will impact the FDA’s ability to exercise its regulatory authority. If these executive actions impose constraints on FDA’s ability to engage in oversight and implementation activities in the normal course, our business may be negatively impacted.
Changes in funding forDisruptions at the FDA and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire, and retain or deploy key leadership and other personnel, or otherwise prevent new or modified products and services from being developed, approved developed or commercialized in a timely manner or at all, which could negatively impact our business.
The ability of the FDA to review and or approve new products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy changes, the FDA’s ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes.other events that may otherwise affect the FDA’s ability to perform routine functions. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs and biologics to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, overSeparately, in response to the last several years, including for 35 days beginning on December 22, 2018, the U.S. government has shut down several times and certain regulatory agencies, such asCOVID-19 pandemic, the FDA have hadhas periodically modified its regular practices with respect to furlough critical FDA employees and stop critical activities.inspection of manufacturing facilities. If a prolonged government shutdown occurs, or if global health concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA or other regulatory authorities to timely review and process our regulatory submissions, which could have a material adverse effect on our businessbusiness.
We are subject to U.S. and certain foreign export and import controls, sanctions, embargoes, anti‑corruptionanti-corruption laws, and anti‑moneyanti-money laundering laws and regulations. Compliance with these legal standards could impair our ability to compete in domestic and international markets. We can face criminal liability and other serious consequences for violations, which can have a material adverse effect on our business.
We are subject to export control and import laws and regulations, including the U.S. Export Administration Regulations administered by the U.S. Commerce Department’s Bureau of Industry and Security, U.S. Customscustoms regulations, various economic and trade sanctions regulations including those administered or enforced by relevant government authorities, such as by the U.S. Treasury Department’s Office of Foreign Assets Control or the U.S. Department of State, the U.S. Foreign Corrupt Practices Act of 1977, as amended, or FCPA, the U.S. domestic bribery statute contained in 18 U.S.C. § 201, the U.S. Travel Act, the USA PATRIOT Act, and other state and national anti‑briberyanti-bribery and anti‑moneyanti-money laundering laws in the countries in which
we conduct activities. U.S. sanctions laws and regulations may govern or restrict our business and activities in certain countries and with certain persons. Anti‑corruptionAnti-corruption laws are interpreted broadly and prohibit companies and their employees, agents, contractors and other partners from authorizing, promising, offering or providing, directly or indirectly, improper payments or anything else of value to recipients in the public or private sector. We may engage third parties for clinical trials outside of the United States, to sell our product candidates abroad once we enter a commercialization phase, and/or to obtain necessary
permits, licenses, patent registrations, and other regulatory approvals. We have direct or indirect interactions with officials and employees of government agencies or government‑affiliatedgovernment-affiliated hospitals, universities and other organizations. We can be held liable for the corrupt or other illegal activities of our employees, agents, contractors and other partners, even if we do not explicitly authorize or have actual knowledge of such activities. Our violations of the laws and regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the loss of export or import privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm and other consequences.
Governments outside the United States tend to impose strict price controls, which may adversely affect our revenues, if any.
In some countries, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a product candidate. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after coverage and reimbursement have been obtained. Reference pricing used by various countries and parallel distribution or arbitrage between low‑priced and high‑priced countries, can further reduce prices. To obtain reimbursement or pricing approval in some countries, we may be required to conduct a clinical trial that compares the cost‑effectiveness of our product candidate to other available therapies, which is time‑consuming and costly. If coverage and reimbursement of our product candidates are unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be materially adversely affected.
If we or our contract manufacturers or other third parties fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on our business.
We and our contract manufacturers and other third parties with whom we do business are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including biological materials and chemicals. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from our use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties for failure to comply with such laws and regulations.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. The failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions.
RISKS RELATED TO OUR INTELLECTUAL PROPERTYRisks Related to our Intellectual Property
If we or our licensors are unable to adequately protect our proprietary technology, or obtain and maintain issued patents that are sufficient to protect our product candidates, others could compete against us more directly, which would negatively impact our business.
Our success depends in large part on our ability to obtain and maintain patent and other intellectual property protection in the United States and other countries with respect to our proprietary technology and products. We seek to protect our proprietary position by filing patent applications in the United States and abroad related to our novel technologies and product candidates. We also rely on trade secrets to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection.
The patent prosecution process is expensive and time‑consuming,time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost, in a timely manner or in all jurisdictions. As we reach the statutory deadlines for deciding whether and where to initiate prosecution in specific foreign jurisdictions by filing national stage applications based on our Patent Cooperation Treaty, or PCT, applications, we will have to decide whether and where to pursue patent
protection for the various inventions claimed in our patent portfolio, and we will only have the opportunity to obtain patents in those jurisdictions where we pursue protection. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. It is possible that defects of form in the preparation or filing of our patents or patent applications may exist, or may arise in the future, such as, with respect to proper priority claims, inventorship, claim scope or patent term adjustments. If there are material defects in the form or preparation of our patents or patent applications, such patents or applications may be invalid and unenforceable. Moreover, our competitors may independently develop equivalent knowledge, methods and know‑how.know-how. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business. We also cannot guarantee that any of our patent searches or analyses, including but not limited to the identification of relevant patents, the scope of patent claims or the expiration of relevant patents, are complete and thorough, nor can we be certain that we have identified each and every patent and pending application in the United States and abroad that is relevant to or necessary for the commercialization of our product candidates in any jurisdiction.
In some circumstances, we may not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the patents covering technology that we license from third parties. We may also require the cooperation of our licensors to enforce any licensed patent rights, and such cooperation may not be provided. Therefore, these patents and applications may not be prosecuted and enforced in a manner consistent with the best interests of our business. Moreover, we have obligations under our licenses, and any failure to satisfy those obligations could give our licensor the right to terminate the license. Termination of a necessary license could have a material adverse impact on our business.
We cannot provide any assurances that the issued patents we currently own, or any future patents, include claims with a scope sufficient to protect our product candidates or otherwise provide any competitive advantage. Further, it is possible that a patent claim may provide coverage for some but not all parts of a product candidate or third‑partythird-party product. These and other factors may provide opportunities for our competitors to design around our patents.
Moreover, other parties may have developed technologies that may be related or competitive to our approach, and may have filed or may file patent applications, and may have received or may receive patents that may overlap or conflict with our patent applications, either by claiming similar methods or by claiming subject matter that could dominate our patent position. In addition, it may be some time before we understand how the patent office reacts to our patent claims and whether they identify prior art of relevance that we have not already considered.
Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore, we cannot know with certainty whether we were the first to make the inventions claimed in any owned patents or pending patent applications, or that we were the first to file for patent protection of such inventions, nor can we know whether those from whom we may license patents were the first to make the inventions claimed or were the first to file. For these and other reasons, the issuance, scope, validity, enforceability and commercial value of our patent rights are subject to a level of uncertainty. Our pending and future patent applications may not result in patents being issued that protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive technologies and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection.
We may be subject to a third‑partythird-party preissuance submission of prior art to the U.S. Patent and Trademark Office, or USPTO, or become involved in opposition, derivation, reexamination, inter partes review, post‑grantpost-grant review or interference proceedings challenging our patent rights or the patent rights of others. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights, allow third parties to commercialize our technology or products and compete directly with us, without payment to us, or result in our inability to manufacture or commercialize product candidates without infringing third‑partythird-party patent rights. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize current or future product candidates. Furthermore, an adverse decision in an interference proceeding can result in a third party receiving the patent right sought by us, which in turn could affect our ability to develop, market or otherwise commercialize our product candidates. The issuance, scope, validity, enforceability and commercial value of our patents are subject to a level of uncertainty.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. Due to legal standards relating to patentability, validity, enforceability and claim scope of patents covering biotechnological and pharmaceutical inventions, our ability to obtain, maintain and enforce patents is uncertain and involves complex legal and factual questions. Even if issued, a patent’s validity, inventorship, ownership or enforceability is not conclusive. Accordingly, rights under any existing patent or any patents we might obtain or license may not cover our product candidates, or may not provide us with sufficient protection for our product candidates to afford a commercial advantage against competitive products or processes, including those from branded and generic pharmaceutical companies.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know‑how,know-how, information, or technology that is not covered by our patents. Although our agreements require all of our employees to assign their inventions to us, and we require all of our employees, consultants, advisors and any other third parties who have access to our trade secrets, proprietary know‑howknow-how and other confidential information and technology to enter into appropriate confidentiality agreements, we cannot be certain that our trade secrets, proprietary know‑how,know-how, and other confidential information and technology will not be subject to unauthorized disclosure or that our competitors will not otherwise gain access to or independently develop substantially equivalent trade secrets, proprietary know‑how,know-how, and other information and technology. Furthermore, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property globally. If we are unable to prevent unauthorized disclosure of our intellectual property related to our product candidates and technology to third parties, we may not be able to establish or maintain a competitive advantage in our market, which could adversely affect our business and operations.
Any litigation to enforce or defend our patent rights, even if we were to prevail, could be costly and time‑consumingtime-consuming and would divert the attention of our management and key personnel from our business operations. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded if we were to prevail may not be commercially meaningful. Even if we are successful, domestic or foreign litigation, or USPTO or foreign patent office proceedings, may result in substantial costs and distraction to our management. We may not be able, alone or with our licensors or potential collaborators, to prevent misappropriation of our proprietary rights, particularly in countries where the laws may not protect such rights as fully as in the United States. Furthermore, because of the substantial amount of discovery required in connection with
intellectual property litigation or other proceedings, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation or other proceedings. In addition, during the course of this kind of litigation or proceedings, there could be public announcements of the results of hearings, motions or other interim proceedings or developments or public access to related documents. If investors perceive these results to be negative, the market price for our common stock could be adversely affected.
If we are unable to protect the confidentiality of our trade secrets and know‑how,know-how, our business and competitive position would be harmed.
In addition to seeking patents for some of our technology and product candidates, we also rely on trade secrets, including unpatented know‑how,know-how, technology and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non‑disclosurenon-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. We also seek to enter into confidentiality and invention or patent assignment agreements with our employees, advisors and consultants. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Our trade secrets may also be obtained by third parties by other means, such as breaches of our physical or computer security systems. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time‑consuming,time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. Moreover, if any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to, or independently developed by, a competitor, our competitive position would be harmed.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other biotechnology companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biotechnology industry involves both technological and legal complexity, and is therefore costly, time‑consumingtime-consuming and inherently uncertain. In addition, recent patent reform legislation could further increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. On September 16, 2011, the Leahy‑SmithThe Leahy-Smith Act America Invents Act, or the Leahy‑SmithLeahy-Smith Act, was signed into law. The Leahy‑Smith Act includes a number of significant changes to U.S. patent law. These includeincluded provisions that affect the way patent applications are prosecuted and may also affect patent litigation. The USPTO recently developed new regulations and procedures to govern administration of the Leahy‑Smith Act, and many of the substantive changes to patent law associated with the Leahy‑Smith Act, in particular the first to file provisions, became effective on March 16, 2013.litigation, including first-to-file provisions. A third party that files a patent application in the USPTO after that date but before us could therefore be awarded a patent covering an invention of ours even if we had made the invention before it was made by the third party. This requires us to be cognizant of the time from invention to filing of a patent application. Thus, for our U.S. patent applications containing a priority claim after March 16, 2013, the date such provisions became effective, there is a greater level of uncertainty in the patent law. Moreover, some of the patent applications in our portfolio will be subject to examination under the pre‑Leahy‑Smithpre-Leahy-Smith Act law and regulations, while other patents applications in our portfolio will
be subject to examination under the law and regulations, as amended by the Leahy‑SmithLeahy-Smith Act. This introduces additional complexities into the prosecution and management of our portfolio.
In addition, the Leahy‑SmithLeahy-Smith Act limits where a patentee may file a patent infringement suit and provides opportunities for third parties to challenge any issued patent in the USPTO. These provisions apply to all of our U.S. patents, even those issued before March 16, 2013. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal court necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a federal court action.
Accordingly, a third party may attempt to use the USPTO procedures to invalidate our patent claims because it may be easier for them to do so relative to challenging the patent in a federal court action. It is not clear what, if any, impact the Leahy‑SmithLeahy-Smith Act will have on the operation of our business. However, the Leahy‑SmithLeahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
In addition, recent U.S. Supreme Court rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. From time to time, the U.S. Supreme Court, other federal courts, the U.S. Congress or the USPTO may change the standards of patentability, and any such changes could have a negative impact on our business.
Depending on these and other decisions by the U.S. Congress, the federal courts and the USPTO, the laws and regulations governing patents could change or be interpreted in unpredictable ways that would weaken our ability to obtain new patents or to enforce any patents that may issue to us in the future. In addition, these events may adversely affect our ability to defend any patents that may issue in procedures in the USPTO or in courts.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could have a material adverse effect on the success of our business.
Our commercial success depends upon our ability, and the ability of our collaborators, to develop, manufacture, market and sell our product candidates and use our proprietary technologies without infringing the proprietary rights of third parties. There is considerable intellectual property litigation in the biotechnology and pharmaceutical industries. While no such litigation has been brought against us and we have not been held by any court to have infringed a third party’s intellectual property rights, we cannot guarantee that our technology, product candidates or use of our product candidates do not infringe third‑partythird-party patents.
We are aware of numerous patents and pending applications owned by third parties, and we monitor patents and patent applications in the fields in which we are developing product candidates, both in the United States and elsewhere. However, we may have failed to identify relevant third‑partythird-party patents or applications. For example, applications filed before November 29, 2000 and certain applications filed after that date that will not be filed outside the United States remain confidential until patents issue. Moreover, it is difficult for industry participants, including us, to identify all third‑partythird-party patent rights that may be relevant to our product candidates and technologies because patent searching is imperfect due to differences in terminology among patents, incomplete databases and the difficulty in assessing the meaning of patent claims. We may fail to identify relevant patents or patent applications or may identify pending patent applications of potential interest but incorrectly predict the likelihood that such patent applications may issue with claims of relevance to our technology. In addition, we may be unaware of one or more issued patents that would be infringed by the manufacture, sale or use of a current or future product candidate, or we may incorrectly conclude that a third‑partythird-party patent is invalid, unenforceable or not infringed by our activities. Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover our technologies, our product candidates or the use of our product candidates.
The biotechnology and pharmaceutical industries are characterized by extensive litigation regarding patents and other intellectual property rights. Other parties may allege that our product candidates or the use of our technologies infringes patent claims or other intellectual property rights held by them or that we are employing their proprietary technology without authorization. We may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect to our product candidates and technology, including interference or derivation proceedings before the USPTO and similar bodies in other countries. Third parties may assert infringement claims against us based on existing intellectual property rights and intellectual property rights that may be granted in the future.
Patent and other types of intellectual property litigation can involve complex factual and legal questions, and their outcome is uncertain. If we are found, or believe there is a risk we may be found, to infringe a third party’s intellectual property rights, we could be required or may choose to obtain a license from such third party to continue developing and marketing our product candidates and technology. However, we may not be able to obtain any such license on commercially reasonable terms or at all. Even if we were able to obtain a license, it could be non‑exclusive,non-exclusive, thereby giving our competitors access to the same technologies licensed to us. We could be forced, including by court order, to cease commercializing the infringing technology
or product. In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have willfully infringed a patent. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business.
Even if we are successful in such proceedings, we may incur substantial costs and divert management time and attention in pursuing these proceedings, which could have a material adverse effect on us. Patent litigation is costly and time‑consuming.time-consuming. We may not have sufficient resources to bring these actions to a successful conclusion. There could be public announcements of the results of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our common stock.
Any of these risks coming to fruition could have a material adverse impact on our business.
We may become involved in lawsuits to protect or enforce our patents or other intellectual property, and our issued patents covering our product candidates could be found invalid or unenforceable or could be interpreted narrowly if challenged in court.
Competitors may infringe our intellectual property, including our patents or the patents of our licensors. As a result, we may be required to file infringement claims to stop third‑partythird-party infringement or unauthorized use. This can be expensive, particularly for a company of our size, and time‑consuming.time-consuming. If we initiated legal proceedings against a third party to enforce a patent, if and when issued, covering one of our product candidates, the defendant could counterclaim that the patent covering our product candidate is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge include alleged failures to meet any of several statutory requirements, including lack of novelty, obviousness or non‑enablement,non-enablement, or failure to claim patent‑eligiblepatent-eligible subject matter. Grounds for unenforceability assertions include allegations that someone connected with the prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. Third parties may
also raise similar claims before administrative bodies in the United States or abroad, even outside the context of litigation. Such mechanisms include re‑examination, post‑grantre-examination, post-grant review, inter partes review, interference proceedings and equivalent proceedings in foreign jurisdictions, such as opposition proceedings. Such proceedings could result in revocation or amendment of our patents in such a way that they no longer cover our product candidates or competitive products. The outcome following legal assertions of invalidity and unenforceability is unpredictable. With respect to validity, for example, we cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. If a defendant were to prevail on a legal assertion of invalidity and/or unenforceability, we would lose at least part, and perhaps all, of the patent protection on our product candidates. Moreover, even if not found invalid or unenforceable, the claims of our patents could be construed narrowly or in a manner that does not cover the allegedly infringing technology in question. Such a loss of patent protection would have a material adverse impact on our business.
The lives of our patents may not be sufficient to effectively protect our products and business.
Patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years after its first effective non-provisional filing date. Although various extensions may be available, the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates, proprietary technologies and their uses are obtained, once the patent life has expired, we may be open to competition. In addition, although upon issuance in the United States a patent’s life can be increased based on certain delays caused by the USPTO, this increase can be reduced or eliminated based on certain delays caused by the patent applicant during patent prosecution. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. If we do not have sufficient patent life to protect our product candidates, proprietary technologies and their uses, our business and results of operations will be adversely affected.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent and, in some jurisdictions, during the pendency of a patent application. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Noncompliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non‑paymentnon-payment of fees and failure to properly
legalize and submit formal documents. In such an event, our competitors might be able to enter the market, which would have an adverse effect on our business.
If we fail to comply with our obligations in our intellectual property licenses and funding arrangements with third parties, we could lose rights that are important to our business.
We are party to multiple license agreements that impose, and we may enter into additional licensing and funding arrangements with third parties that may impose, diligence, development and commercialization timelines, milestone payment, royalty, insurance and other obligations on us. Under our existing licensing agreements, we are obligated to pay royalties on net product sales of product candidates or related technologies to the extent they are covered by the agreement. Our results of operations will be affected by the level of royalty payments that we are required to pay to third parties. We cannot precisely predict the amount, if any, of royalties that we will be required to pay to third parties in the future. Any disagreements with the counterparty over the amount of royalties owed could lead to litigation, which is costly. In addition, if we fail to comply with our obligations under current or future license agreements, our counterparties may have the right to terminate these agreements, in which event we might not be able to develop, manufacture or market any product candidate that is covered by these agreements, or may face other penalties under the agreements. Such an occurrence could materially adversely affect the value of product candidates being developed using rights licensed to us under any such agreement. Termination of these agreements or reduction or elimination of our rights under these agreements may result in our having to negotiate new or reinstated agreements with less favorable terms, or cause us to lose our rights under these agreements, including our rights to important intellectual property or technology. Furthermore, our counterparties may allege that we are operating outside the scope of the licenses granted and terminate our license or otherwise require us to alter development, manufacturing or marketing activities.
We may not be successful in obtaining or maintaining necessary rights to our product candidates through acquisitions and in-licenses.
We currently have rights to certain intellectual property, through licenses from third parties and under patents and patent applications that we own, to develop our product candidates. Because we may find that our programs require the use of proprietary rights held by third parties, the growth of our business may depend in part on our ability to acquire, in‑licensein-license or use these proprietary rights. We may be unable to acquire or in‑licensein-license compositions, methods of use, processes or other third‑party third-party
intellectual property rights from third parties that we identify as necessary for our product candidates. The licensing and acquisition of third‑partythird-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to license or acquire third‑partythird-party intellectual property rights that we may consider attractive. These established companies may have a competitive advantage over us due to their size, financial resources and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third‑partythird-party intellectual property rights on terms that would allow us to make an appropriate return on our investment.
We may also be unable to maintain third‑party intellectual property rights. For example, MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. In December 2019, we entered into the MIT Amendment. Pursuant to the MIT Amendment, the provision of the MIT License under which we were obligated to initiate a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT License. The parties agreed to negotiate in good faith to enter into a future amendment to the MIT License after we provide MIT with an amended diligence plan. However, if we are unable to reach an agreement with MIT regarding an acceptable amendment of the MIT License and if we are unable to cure the breach, there could be a material adverse effect on our business. See “Business - Licenses and Collaborations - Massachusetts Institute of Technology.”
If we are unable to successfully obtain rights to required third‑partythird-party intellectual property rights or maintain the existing intellectual property rights we have, we may have to abandon development of that program and our business and financial condition could suffer.
We may be subject to claims by third parties asserting that our employees or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Many of our employees were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may also engage advisors and consultants who are concurrently employed at universities or other organizations or who perform services for other entities. Although we try to ensure that our employees, advisors and consultants do not use the proprietary information or know‑howknow-how of others in their work for us, we may be subject to claims that we or our employees, advisors or consultants have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such party’s former or current employer or in violation of an agreement with another party. Although we have no knowledge of any such claims being alleged to date, if such claims were to arise, litigation may be necessary to defend against any such claims.
In addition, while it is our policy to require our employees, consultants, advisors and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. Our and their assignment agreements may not be self‑executingself-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property. Similarly, we may be subject to claims that an employee, advisor or consultant performed work for us that conflicts with that person’s obligations to a third party, such as an employer, and thus, that the third party has an ownership interest in the intellectual property arising out of work performed for us. Litigation may be necessary to defend against these claims. Although we have no knowledge of any such claims being alleged to date, if such claims were to arise, litigation may be necessary to defend against any such claims.
If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to management.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition among potential collaborators or customers in our markets of interest. At times, competitors may adopt trade names or trademarks similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected. Our efforts to enforce or protect our proprietary rights related to trademarks, trade secrets, domain names, copyrights or other intellectual property may be ineffective and could result in substantial costs and diversion of resources and could adversely impact our financial condition or results of operations.
We will not seek to protect our intellectual property rights in all jurisdictions throughout the world and we may not be able to adequately enforce our intellectual property rights even in the jurisdictions where we seek protection.
Filing, prosecuting and defending patents on product candidates in all countries and jurisdictions throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States could be less extensive than in the United States, assuming that rights are obtained in the United States and assuming that rights are pursued outside the United States. In this regard, in addition to the United States, we also seek to protect our intellectual property rights in other countries. The statutory deadlines for pursuing patent protection in individual foreign jurisdictions are based on the priority date of each of our patent applications. For all of the patent families in our portfolio, including the families that may provide coverage for our lead product candidate, the relevant statutory deadlines have not yet expired. Therefore, for each of the patent families that we believe provide coverage for our lead product candidate, we will need to decide whether and where to pursue additional protection outside the United States. In addition, the laws of some foreign countries, do not protect intellectual property rights to the same extent as federal and state laws in the United States. Consequently, for our existing patent rights outside the United States and any foreign patent rights we may decide to pursue in the future, we may not be able to obtain relevant claims and/or we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions.
Competitors may use our technologies in jurisdictions where we do not pursue and obtain patent protection to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as in the United States. These products may compete with our product candidates and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Even if we pursue and obtain issued patents in particular jurisdictions, our patent claims or other intellectual property rights may not be effective or sufficient to prevent third parties from so competing.
If we do not obtain additional protection under the Hatch‑WaxmanHatch-Waxman Act and similar foreign legislation extending the terms of our patents for our product candidates, our business may be harmed.
Depending upon the timing, duration and specifics of FDA regulatory approval for our product candidates, one or more of our U.S. patents may be eligible for limited patent term restoration under the Hatch‑WaxmanHatch-Waxman Act. The Hatch‑WaxmanHatch-Waxman Act permits a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. Patent term restorations, however, are limited to a maximum of five years and cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval by the FDA.
The application for patent term extension is subject to approval by the USPTO, in conjunction with the FDA. It takes at least six months to obtain approval of the application for patent term extension. We may not be granted an extension because of, for example, failing to apply within applicable deadlines, failing to apply prior to expiration of relevant patents or otherwise failing to satisfy applicable requirements. Moreover, the applicable time period or the scope of patent protection afforded could be less than we request. If we are unable to obtain patent term extension or restoration or the term of any such extension is less than we request, the period during which we will have the right to exclusively market our product will be shortened, our competitors may obtain earlier approval of competing products and our ability to generate revenues could be materially adversely affected.
RISKS RELATED TO OUR OPERATIONS
Our new corporate strategy may not be successful.
On January 3, 2019, following a strategic business review, we announcedRisks Related to our new strategy to focus on the development of SEL-212 for the treatment of chronic refractory gout and advancement of our ImmTOR platform in the area of gene therapy, specifically ImmTOR in combination with AAV gene therapy for the treatment of CN and MMA, as well as the deprioritization of our oncology development program. The success of this strategic shift will depend on our ability to successfully develop our product candidates, hire and retain senior management or other highly qualified personnel, prioritize competing projects and efforts and obtain sufficient resources, including additional capital, as well as our ability to enter into collaborations with third parties. The early stage development of novel product candidates is highly unpredictable due to the lengthy and expensive process of clinical drug development, potential for safety, efficacy or tolerability problems with such product candidates, unexpected expenses or inaccurate financial assumptions or forecasts, potential delays or unfavorable decisions of regulatory agencies and competition for targeted indications or within targeted markets. Accordingly, there are no assurances our change in strategic focus will be successful, which may have an adverse effect on our results of operations or financial condition.Operations
Our future success depends on our ability to retain key executives and to attract, retain and motivate qualified personnel.
We are highly dependent on Carsten Brunn, Ph.D., our President and Chief Executive Officer, as well as the other principal members of our management, scientific and clinical team. Although we have entered into employment agreements or offer letters with Dr. Brunn and other executive officers, each of them may terminate their employment with us at any time. We do not maintain “key person” insurance for any of our executives or other employees.
Recruiting and retaining qualified scientific, clinical, manufacturing, technology and sales and marketing personnel will also be critical to our success. The loss of the services of our executive officers or other key employees could impede the achievement of our research, development and commercialization objectives and seriously harm our ability to successfully implement our business strategy. Furthermore, replacing executive officers and key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop, gain regulatory approval of and commercialize product candidates. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate these key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. In addition, we rely on consultants and advisors, including scientific and clinical advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities that may limit their availability to us. If we are unable to continue to attract and retain high quality personnel, our ability to pursue our growth strategy will be limited.
We expect to expand our development and regulatory capabilities and potentially implement sales, marketing and distribution capabilities, and as a result, we may encounter difficulties in managing our growth, which could disrupt our operations.
We expect to experience significant growth in the number of our employees and the scope of our operations, particularly in the areas of lead discovery and product development, regulatory affairs, clinical affairs and manufacturing and, if any of our product candidates receives marketing approval, sales, marketing and distribution. To manage our expected future growth, we must continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified personnel. Due to our limited financial resources and the limited experience of our management team in managing a company with such expected growth, we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel in a timely manner, if at all. The expansion of our operations may lead to significant costs and may divert our management and business development resources. Any inability to manage or financially support growth could delay the execution of our business plans or disrupt our operations.
We have incurred increased costs as a result of operating as a public company, and our management will be required to devote substantial time to compliance initiatives and corporate governance practices.
As a public company, and particularly after we are no longer an emerging growth company, we have incurred and expect to continue to incur significant legal, accounting and other expenses thatexpenses. In particular, beginning with this Annual Report, we did not incur as a private company. The Sarbanes‑Oxley Act of 2002, the Dodd‑Frank Wall Street Reform and Consumer Protection Act, the listing requirements of The Nasdaq Global Market and other applicable securities rules and regulations impose various requirements on public companies, including establishment and maintenance of effective disclosure and financial controls and corporate governance practices. Our management and other personnel need to devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations have increased our legal and financial compliance costs and made some activities more time‑consuming and costly.
Pursuant to Section 404 of the Sarbanes-Oxley Act of 2002, or Section 404, we are required to furnish a report by our management on our internal control over financial reporting. However, while we remain an emerging growth company, we will not be required to include an attestation report on internal control over financial reporting issued by our independent registered public accounting firm. If we are unable to maintain effective internal control over financial reporting, we may not have adequate, accurate or timely financial information, and we may be unable to meet our reporting obligations as a public company or comply with the requirements of the SEC or Section 404. This could result in a restatement of our financial statements, the imposition of sanctions, including the inability of registered broker dealers to make a market in our common stock, or investigation by regulatory authorities. Any such action or other negative results caused by our inability to meet our reporting requirements or comply with legal and regulatory requirements or by disclosure of an accounting, reporting or control issue could adversely affect the trading price of our securities and our business. Material weaknesses in our internal control over financial reporting could also reduce our ability to obtain financing or could increase the cost of any financing we obtain. This could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial statements.
A variety of risks associated with maintaining our subsidiary in Russia or expanding operations internationally could adversely affect our business.
In addition to our U.S. operations, we maintain a wholly owned subsidiary in Russia, Selecta RUS.(RUS). However, we plan to wind down these operations. We may face risks associated with maintaining our subsidiary in Russia, or with any international operations, including possible unfavorable regulatory, pricing and reimbursement, legal, political, tax and labor conditions, and risks associated with our compliance with evolving international sanctions, which could harm our business. We may also rely
on collaborators to commercialize any approved product candidates outside of the United States. Doing business internationally involves a number of risks, including but not limited to:
| |
- | multiple, conflicting and changing laws and regulations, such as privacy regulations, tax laws, export and import restrictions, employment laws, regulatory requirements and other governmental approvals, permits and licenses; |
| |
- | failure by us to obtain and maintain regulatory approvals for the use of our product candidates in various countries; |
| |
- | additional potentially relevant third‑party patent rights; |
| |
- | complexities and difficulties in obtaining protection of and enforcing our intellectual property rights; |
| |
- | difficulties in staffing and managing foreign operations; |
| |
- | complexities associated with managing multiple‑payor reimbursement regimes, government payors or patient self‑pay systems; |
| |
- | limits on our ability to penetrate international markets; |
| |
- | financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for our product candidates and exposure to foreign currency exchange rate fluctuations, which could result in increased operating expenses and reduced revenues; |
| |
- | natural disasters, political and economic instability, including wars, events of terrorism and political unrest, outbreak of disease, including the novel coronavirus boycotts, curtailment of trade and other business restrictions and economic weakness, including inflation; |
| |
- | changes in diplomatic and trade relationships; |
| |
- | challenges in enforcing our contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual property rights to the same extent as the United States; |
| |
- | restriction on cross-border investment, including enhanced oversight by the Committee on Foreign Investment in the United States (CFIUS) and substantial restrictions on investment from China; |
| |
- | certain expenses including, among others, expenses for travel, translation and insurance; |
•multiple, conflicting and changing laws and regulations, such as privacy regulations, tax laws, export and import restrictions, employment laws, regulatory requirements and other governmental approvals, permits and licenses;
| |
- | legal risks, including use of the legal system by the government to benefit itself or affiliated entities at our expense, including expropriation of property; and |
| |
- | regulatory and compliance risks that relate to maintaining accurate information and control over sales and activities that may fall within the purview of the FCPA its books and records provisions, or its anti‑bribery provisions. |
•additional potentially relevant third-party patent rights;
•complexities and difficulties in obtaining protection of and enforcing our intellectual property rights;
•difficulties in staffing and managing foreign operations;
•complexities associated with managing multiple-payor reimbursement regimes, government payors or patient self-pay systems;
•limits on our ability to penetrate international markets;
•financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for our product candidates and exposure to foreign currency exchange rate fluctuations, which could result in increased operating expenses and reduced revenues;
•natural disasters, political and economic instability, including wars, events of terrorism and political unrest, outbreak of disease, including the COVID-19 pandemic, boycotts, curtailment of trade and other business restrictions, economic sanctions, and economic weakness, including inflation;
•changes in diplomatic and trade relationships;
•challenges in enforcing our contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual property rights to the same extent as the United States;
•restriction on cross-border investment, including enhanced oversight by the Committee on Foreign Investment in the United States and substantial restrictions on investment from China;
•certain expenses including, among others, expenses for travel, translation and insurance;
•legal risks, including use of the legal system by the government to benefit itself or affiliated entities at our expense, including expropriation of property;
•regulatory and compliance risks that relate to maintaining accurate information and control over sales and activities that may fall within the purview of the FCPA its books and records provisions, or its anti-bribery provisions; and
•risks that we may suffer reputational harm as a result of our operations in Russia.
Any of these factors could significantly harm our future international expansion and operations and, consequently, our results of operations.
Our business and operations would suffer in the event of system failures or unauthorized or inappropriate use of or access to our systems.
Despite the implementation of security measures, our internal computer systems and those of our current and future contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we are not aware of any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our product candidates and conduct clinical trials, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our product candidates could be delayed.
Acquisitions or joint ventures could disrupt our business, cause dilution to our stockholders and otherwise harm our business.
We may acquire other businesses, product candidates or technologies as well as pursue strategic alliances, joint ventures, technology licenses or investments in complementary businesses. We have not made any acquisitions to date, and our ability to do so successfully is unproven. Any of these transactions could be material to our financial condition and operating results and expose us to many risks, including:
| |
- | disruption in our relationships with future customers or with current or future distributors or suppliers as a result of such a transaction; |
| |
- | unexpected liabilities related to acquired companies; |
| |
- | difficulties integrating acquired personnel, technologies and operations into our existing business; |
| |
- | diversion of management time and focus from operating our business to acquisition integration challenges; |
| |
- | increases in our expenses and reductions in our cash available for operations and other uses; |
| |
- | possible write‑offs or impairment charges relating to acquired businesses; and |
| |
- | inability to develop a sales force for any additional product candidates. |
•disruption in our relationships with future customers or with current or future distributors or suppliers as a result of such a transaction;
•unexpected liabilities related to acquired companies;
•difficulties integrating acquired personnel, technologies and operations into our existing business;
•diversion of management time and focus from operating our business to acquisition integration challenges;
•increases in our expenses and reductions in our cash available for operations and other uses;
•possible write-offs or impairment charges relating to acquired businesses; and
•inability to develop a sales force for any additional product candidates.
Foreign acquisitions involve unique risks in addition to those mentioned above, including those related to integration of operations across different cultures and languages, currency risks and the particular economic, political and regulatory risks associated with specific countries.
Also, the expected benefit of any acquisition may not materialize. Future acquisitions or dispositions could result in potentially dilutive issuances of our equity securities, the incurrence of debt, contingent liabilities or amortization expenses or write‑offswrite-offs of goodwill, any of which could harm our financial condition. We cannot predict the number, timing or size of future joint ventures or acquisitions, or the effect that any such transactions might have on our operating results.
RISKS RELATED TO OUR COMMON STOCKRisks Related to our Common Stock
The market price of our common stock may be volatile and fluctuate substantially, which could result in substantial losses for purchasers of our common stock.
The trading price of our common stock is likely to be volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control. The stock market in general and the market for smaller biopharmaceutical companies in particular have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. As a result of this volatility, you may not be able to sell your common stock at or above the price at which you purchased. The market price for our common stock may be influenced by many factors, including:
| |
- | the success of competitive products or technologies; |
•the success of competitive products or technologies;
| |
- | results or progress, or changes in approach or timelines, of clinical trials of our product candidates or those of our competitors; |
| |
- | failure or discontinuation of any of our development programs; |
| |
- | commencement of, termination of, or any development related to any collaboration or licensing arrangement; |
| |
- | regulatory or legal developments in the United States and other countries; |
| |
- | development of new product candidates that may address our markets and make our product candidates less attractive; |
| |
- | changes in physician, hospital or healthcare provider practices that may make our product candidates less useful; |
| |
- | announcements by us, our partners or our competitors of significant acquisitions, strategic partnerships, joint ventures, collaborations or capital commitments; |
| |
- | announcement or market expectation of additional financing efforts; |
| |
- | developments or disputes concerning patent applications, issued patents or other proprietary rights; |
| |
- | the recruitment or departure of key personnel; |
| |
- | the level of expenses related to any of our product candidates or clinical development programs; |
| |
- | failure to meet or exceed financial estimates, projections or development timelines of the investment community or that we provide to the public; |
| |
- | the results of our efforts to discover, develop, acquire or in‑license additional product candidates or products; |
| |
- | actual or expected changes in estimates as to financial results, development timelines or recommendations by securities analysts; |
| |
- | variations in our financial results or those of companies that are perceived to be similar to us; |
| |
- | changes in the structure of healthcare payment systems; |
| |
- | sale of common stock by us or our stockholders in the future as well as the overall trading volume of our common stock; |
| |
- | market conditions in the pharmaceutical and biotechnology sectors; |
| |
- | general economic, industry and market conditions; and |
| |
- | the other factors described in this “Risk factors” section. |
•failure or discontinuation of any of our development programs;
•commencement of, termination of, or any development related to any collaboration or licensing arrangement;
•regulatory or legal developments in the United States and other countries;
•development of new product candidates that may address our markets and make our product candidates less attractive;
•changes in physician, hospital or healthcare provider practices that may make our product candidates less useful;
•announcements by us, our collaborators or our competitors of significant acquisitions, strategic partnerships, joint ventures, collaborations or capital commitments;
•announcement or market expectation of additional financing efforts;
•developments or disputes concerning patent applications, issued patents or other proprietary rights;
•the recruitment or departure of key personnel;
•the level of expenses related to any of our product candidates or clinical development programs;
•failure to meet or exceed financial estimates, projections or development timelines of the investment community or that we provide to the public;
•the results of our efforts to discover, develop, acquire or in-license additional product candidates or products;
•actual or expected changes in estimates as to financial results, development timelines or recommendations by securities analysts;
•variations in our financial results or those of companies that are perceived to be similar to us;
•changes in the structure of healthcare payment systems;
•sale of common stock by us or our stockholders in the future as well as the overall trading volume of our common stock;
•changes in the composition of our stockholder base;
•activity in the options market for shares of our common stock;
•market conditions in the pharmaceutical and biotechnology sectors;
•general economic, industry and market conditions; and
•the other factors described in this “Risk Factors” section.
Our executive officers, directors, and principal stockholders, if they choose to act together, will continue to have the ability to control or significantly influence all matters submitted to stockholders for approval.
Our executive officers, directors and stockholders who own more than 5% of our outstanding common stock and their respective affiliates, in the aggregate, hold shares representing approximately 45.9%31.1% of our outstanding voting stock as of December 31, 2019.2021. As a result, if these stockholders choose to act together, they would be able to control or significantly influence all matters submitted to our stockholders for approval, as well as our management and affairs. For example, these persons, if they choose to act together, would control or significantly influence the election of directors, the composition of our management and approval of any merger, consolidation or sale of all or substantially all of our assets.
A significant portion of our total outstanding shares are eligible to be sold into the market, which could cause the market price of our common stock to drop significantly, even if our business is doing well.
Sales of a substantial number of shares of our common stock in the public market, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock. Holders
Sales of an aggregatea substantial number of approximately 0.34 million shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock reduce. As of December 31, 2021, we had 123,622,965 shares of common stock outstanding. Also, as of December 31, 2019 have rights, subject to specified conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other stockholders, until such shares can otherwise be sold without restriction under Rule 144 or until the rights terminate pursuant to the terms of the investors’ rights agreement between us2021, 11,039,873 and such holders. We have also registered and intend to continue to register all394,450 shares of common stock that we may issueare subject to outstanding options or restricted stock unit awards, respectively, under our outstanding equity compensation plans. Once we registerplans are eligible for sale in the public market to the extent permitted by the provisions of various vesting schedules, and Rule 144 under the Securities Act. Additionally, as of December 31, 2021, up to 10,735,980 shares of common stock are issuable upon exercise of outstanding warrants. If these additional shares of common stock are sold, or it is perceived that they canwill be freely sold, in the public market, upon issuance, subject to volume limitations applicable to affiliates.
In addition, on June 27, 2017, we issued and sold in a private placement 3,088,791 sharesthe trading price of our common stock (of which 338,791 shares have continuing registration rights) and a warrant to purchase 79,130 shares of our common stock, including to certain of our affiliates. Pursuant to a registration rights agreement entered into with the investors in the private placement, on July 13, 2017, we filed a Registration Statement on Form S-3 to register the shares of common stock sold in the private
placement and the shares of common stock issuable upon exercise of the warrant. Similarly, on December 23, 2019, we issued and sold in a private placement 37,634,883 shares of our common stock and warrants to purchase 31,330,629 shares of our common stock, including to certain of our affiliates. Pursuant to a registration rights agreement entered into with the investors in the private placement, on January 29, 2019, we filed a Registration Statement on Form S-3 to register the shares of common stock sold in the private placement and the shares of common stock issuable upon exercise of the warrant. As a result, these shares can be freely sold in the public market.could decline.
We may not have the funds necessary to fulfill our obligation to repurchase certain warrants.
Under certain circumstances, holders of certain warrants issued in December 2019 may require us to repurchase the remaining unexercised portion of such warrants for an amount of cash equal to the value of the warrant as determined in accordance with the Black ScholesBlack-Scholes option pricing model and the terms of the warrants. Our ability to repurchase the warrants depends on our ability to generate cash flow in the future. To some extent, this is subject to general economic, financial, competitive, legislative and regulatory factors and other factors that are beyond our control. We cannot be certain that we will maintain sufficient cash reserves or that our business will generate cash flow from operations at levels sufficient to permit us to repurchase the warrants.
We are an “emerging growth company,” and the reduced disclosure requirements applicable to emerging growth companies may make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and may remain an emerging growth company until the last day of the fiscal year following the fifth anniversary of the closing of the initial public offering of our common stock. However, if certain events occur prior to the end of such five‑year period, including if we become a “large accelerated filer,” our annual gross revenues exceed $1.07 billion or we issue more than $1.07 billion of non‑convertible debt in any three‑year period, we will cease to be an emerging growth company prior to the end of such five‑year period. For so long as we remain an emerging growth company, we are permitted and intend to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include:
| |
- | not being required to comply with the auditor attestation requirements in the assessment of our internal control over financial reporting; |
| |
- | not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements; |
| |
- | reduced disclosure obligations regarding executive compensation; and |
| |
- | exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. |
We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. We have, historically, relied on these exemptions, and we may continue to do so until they are no longer available to us. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be reduced or more volatile. In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of these accounting standards until they would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
If securities or industry analysts do not publish research or reports about our business, or if they issue an adverse or misleading opinion regarding our stock, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that industry or securities analysts publish about us or our business. If any of the analysts who cover us issue an adverse or misleading opinion regarding us, our business model, our intellectual property or our stock performance, or if our preclinical studies, clinical trial programs and operating results fail to meet the expectations of analysts, our stock price would likely decline. If one or more of these analysts ceases coverage of us or fails to publish reports on us regularly, we could lose visibility in the financial markets, which in turn could cause our stock price or trading volume to decline.
Provisions in our restated certificate of incorporation and restated bylaws and under Delaware law could make an acquisition of our company, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our restated certificate of incorporation and our restated bylaws which became effective upon the closing of the initial public offering of our common stock may discourage, delay or prevent a merger, acquisition or other change in control of
our company that stockholders may consider favorable, including transactions in which youa stockholder might otherwise receive a premium for yourits shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock, thereby depressing the market price of our common stock. In addition, because our board of directors is responsible for appointing the members of our management team, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors.
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the General Corporation Law of the State of Delaware, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
Furthermore, our restated certificate of incorporation specifies that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for most legal actions involving claims brought against us by stockholders. We believe this provision benefits us by providing increased consistency in the application of Delaware law by chancellors particularly experienced in resolving corporate disputes, efficient administration of cases on a more expedited schedule relative to other forums and protection against the burdens of multi‑forummulti-forum litigation. However, the provision may have the effect of discouraging lawsuits against our directors, officers, employees and agents as it may limit any stockholder’s ability to bring a claim in a judicial forum that such stockholder finds favorable for disputes with us or our directors, officers, employees or agents. The enforceability
We could be subject toare involved in two securities class action litigation.lawsuits.
In the past, securities class action litigation has often been brought against a company following a decline in the market price of its securities. This risk is especially relevant for us because biopharmaceutical companies have experienced significant stock price volatility in recent years. If we faceInvolvement in such litigation, it could result in substantial costs and a diversion of management’s attention and resources, which could harm our business.
On August 3, 2020, a stockholder of Selecta filed a stockholder derivative action, purportedly on behalf of Selecta and against certain current and former members of the Company’s Board of Directors, as well as one affiliated company owned by a current board member, in the Court of Chancery of the State of Delaware, namely Franchi v. Barabe, et al. The complaint alleges that the individual defendants breached their fiduciary duties and committed corporate waste when they authorized a private placement transaction, announced on December 19, 2019, at a price allegedly below fair value. The complaint further alleges that the four defendant directors who participated in the private placement were unjustly enriched in connection with the transaction. On September 25, 2020, the defendants filed a motion to dismiss the lawsuit. On November 6, 2020, the plaintiff filed an amended complaint, and the defendants filed a second motion to dismiss on January 8, 2021. On December 31, 2020, we received a litigation demand letter from two other putative stockholders relating to the same private placement transaction. On April 12, 2021, the Court of Chancery in the State of Delaware granted a motion to stay the litigation pending a review by a Special Committee appointed by the Company’s Board of Directors. While the litigation was stayed, the parties reached an agreement in principle to settle the matter, and they expect to submit documentation to the Court for its approval of the settlement in the near future. We could receive other demands or be subject to other litigation. While we intend to vigorously defend against any demands which we believe to be without merit, there can be no assurance as to the outcome of any stockholder litigation. Unfavorable outcomes in securities class action litigation could require us to pay extensive damages, which could delay or prevent our ability to develop our product candidates and harm our operations. Item 1B. Unresolved Staff Comments
Not applicable.
Item 2. Properties
Our corporate headquarters are currently located at 480 Arsenal Way,65 Grove Street, Watertown, Massachusetts and consist of 32,93325,078 total square feet of leased office and laboratory space under a lease that expires in March 2020.
In July 2019, we entered into a lease for 25,078 square feet of laboratory and office space located at 65 Grove Street, Watertown, Massachusetts, commencing in March 2020, which will serve as our new corporate headquarters.May 2028.
We also lease approximately 2,500 square feet of office and laboratory space in Moscow, Russia on a month to monthmonth-to-month basis.
Item 3. Legal Proceedings
We are not partyOn August 3, 2020, a stockholder of Selecta filed a stockholder derivative action, purportedly on behalf of Selecta and against certain current and former members of the Company’s Board of Directors, as well as one affiliated company owned by a current board member, in the Court of Chancery of the State of Delaware, namely Franchi v. Barabe, et al. The complaint alleges that the individual defendants breached their fiduciary duties and committed corporate waste when they authorized a private placement transaction, announced on December 19, 2019, at a price allegedly below fair value. The complaint further alleges that the four defendant directors who participated in the private placement were unjustly enriched in connection with the transaction. On September 25, 2020, the defendants filed a motion to any material legal proceedings.dismiss the lawsuit. On November 6, 2020, the plaintiff filed an amended complaint, and the defendants filed a second motion to dismiss on January 8, 2021. On December 31, 2020, we received a litigation demand letter from two other putative stockholders relating to the same private placement transaction. On April 12, 2021, the Court of Chancery in the State of Delaware granted a motion to stay the litigation pending a review by a Special Committee appointed by the Company’s Board of Directors. While the litigation was stayed, the parties reached an agreement in principle to settle the matter, and they expect to submit documentation to the Court for its approval of the settlement in the near future.
Item 4. Mine Safety Disclosures
Not applicable.
Information about our Directors
Carsten Brunn, Ph.D. has served as our President, Chief Executive Officer and member of our board of directors since December 2018. Prior to joining Selecta Biosciences, Inc., Dr. Brunn was the President of Pharmaceuticals for the Americas Region at Bayer and a member of the Global Pharmaceutical Executive Committee. Prior to being appointed to that role in 2017, he was the President of Bayer Pharmaceuticals in Japan, a role he held since 2013. He also served as the Chairman of the European Federation of Pharmaceutical Industries and Associations (EFPIA) Japan, an organization representing innovative pharmaceutical companies in Japan. Over the last 25 years, Dr. Brunn has held a number of senior leadership positions in Europe, Asia and the United States at leading companies such as Eli Lilly, Novartis, Basilea and Bausch and Lomb. Dr. Brunn
graduated from the University of Freiburg in Germany with a Master of Science in Pharmaceutical Sciences. In addition, he studied at the University of Washington under a research scholarship. He received his Ph.D. in Chemistry from the University of Hamburg, Germany and completed his executive education at London Business School. Dr. Brunn's experience as a senior executive of life sciences companies and knowledge of the pharmaceutical and biotechnology industries contributed to our board of directors' conclusion that he should serve as a director of our company.
Carrie S. Cox has served as a member of our board of directors and as chairman of the board of directors since November 2019. Ms. Cox most recently served as the Chief Executive Officer of Humacyte, Inc., a regenerative medicine company based in Durham, North Carolina, from 2010 to June 2018, and has served as a member of its board of directors since 2010, serving as chairman from 2011 to June 2019. Ms. Cox has served on the boards of directors of Texas Instruments Incorporated since 2004 and Cardinal Health, Inc. since 2009, and as the chairman of the board of directors of electroCore, Inc. since July 2018. Ms. Cox previously served on the board of directors of Celgene Corporation from 2009 to November 2019 and as chairman of the board of directors of Array BioPharma, Inc. from August 2018 to July 2019.Ms. Cox received a B.S. from the Massachusetts College of Pharmacy, and was a registered pharmacist. Ms. Cox’s vast experience as a pharmaceutical executive and member of multiple boards of directors in the biotechnology industry as well as her knowledge of corporate strategy qualifies her to serve as a director of our company.
Timothy C. Barabe has served as a member of our board of directors since July 2016. Mr. Barabe also serves on the boards of Veeva Systems Inc. and Vigilant Biosciences, Inc., a private company. From 2001 to January 2020, Mr. Barabe served on the board of ArQule, Inc., and from 2014 to 2017, Mr. Barabe served on the board of directors of Opexa Therapeutics, Inc. Mr. Barabe retired in June 2013 from his position as Executive Vice President and Chief Financial Officer of Affymetrix, Inc. Previously, from July 2006 until March 2010, he was Senior Vice President and Chief Financial Officer of Human Genome Sciences, Inc. From 2004 to 2006, he served as Chief Financial Officer of Regent Medical Limited, a U.K.-based, privately owned, surgical supply company. Mr. Barabe served with Novartis AG from 1982 through August 2004 in a succession of senior executive positions in finance and general management, most recently as the Chief Financial Officer of Sandoz GmbH, the generic pharmaceutical subsidiary of Novartis. Mr. Barabe received his B.B.A. degree from the University of Massachusetts (Amherst) and his M.B.A. degree from the University of Chicago. Mr. Barabe’s experience as a senior financial executive of life sciences companies and knowledge of the pharmaceutical and biotech industries contributed to our board of directors’ conclusion that he should serve as a director of our company.
Scott D. Myers has served as a member of our board of directors since June 2019. Mr. Myers most recently served as Chief Executive Officer, President, and Chairman of the Board of Rainier Therapeutics, a clinical-stage biotechnology company focused on metastatic bladder cancer, from June 2018 to January 2020. Prior to Rainier Therapeutics, Mr. Myers served as Chief Executive Officer and as a member of the board of directors of Cascadian Therapeutics, an oncology company, from April 2016 until it was acquired by Seattle Genetics in March 2018. Prior to Cascadian, Mr. Myers served as Chief Executive Officer of Aerocrine AB, a medical device company, from 2011 through its acquisition by Circassia Pharmaceuticals plc in July 2015. Mr. Myers held senior commercial operations, general management, and information management positions for UCB SA, a Belgium-based biopharmaceutical company, and Johnson & Johnson. He currently serves on the board of directors of Harpoon Therapeutics. Mr. Myers earned his B.A. in biology from Northwestern University and his M.B.A. from the University of Chicago Booth School of Business. Mr. Myers’ experience as a senior executive of life sciences companies and knowledge of the pharmaceutical and biotech industries contributed to our board of directors’ conclusion that he should serve as a director of our company.
Amir Nashat, Ph.D. has served as a member of our board of directors since 2008. Dr. Nashat has been a Partner at Polaris Partners, a venture capital firm, since 2009 and focuses on investments in the life sciences. He currently serves on the board of directors of Fate Therapeutics, Inc., Syros Pharmaceuticals, Scholar Rock Inc., and several private companies. Dr. Nashat has also served as a director of Receptos, Inc., BIND Therapeutics, Inc., aTyr Pharma, as well as Adnexus Therapeutics, Inc. (acquired by Bristol-Myers Squibb Company) and other private companies. Dr. Nashat completed his Ph.D. as a Hertz Fellow in Chemical Engineering at MIT with a minor in biology. Dr. Nashat earned both his M.S. and B.S. in materials science and mechanical engineering at the University of California, Berkeley. Dr. Nashat’s extensive experience as a venture capitalist and board member to numerous companies in the biotechnology industry contributed to our board of directors’ conclusion that he should serve as a director of our company.
Aymeric Sallin, M.S. has served as a member of our board of directors since 2008. Mr. Sallin has served as the Chief Executive Officer of NanoDimension, a venture capital firm, since 2002 and is the founder of that firm. Since 2014, Mr. Sallin has served as a strategic advisory board member of the École Polytechnique Fédérale de Lausanne, or EPFL. Since 2002, Mr. Sallin has worked to promote nanotechnology around the world, and has received the NSTI Fellow Award and 2012 EPFL Alumni award for his contribution to the field of nanotechnology. Mr. Sallin has worked to generate and close investments of hundreds of millions of dollars into several of NanoDimension’s portfolio companies. He currently serves as a board member of H55, Inc., View, Inc., CROCUS Technology and Tarveda Therapeutics. Mr. Sallin is also a member of the Swiss Academy of Engineering Science. Mr. Sallin received his Masters in Physical Engineering from EPFL in Lausanne, Switzerland. Mr. Sallins' extensive
knowledge of our business and the nanomedicine field contributed to our board of directors’ conclusion that he should serve as a director of our company.
Timothy A. Springer, Ph.D. has served as a member of our board of directors since June 2016 and as a scientific advisor to us since December 2008. Since 1989, Dr. Springer has served as the Latham Family Professor at Harvard Medical School. He has also served as Senior Investigator in the Program in Cellular and Molecular Medicine at Boston Children’s Hospital since 2012, and as Professor of Biological Chemistry and Molecular Pharmacology at Harvard Medical School and Professor of Medicine at Boston Children’s Hospital since 2011. Dr. Springer was the Founder of LeukoSite, a biotechnology company acquired by Millennium Pharmaceuticals in 1999. Additionally, he is a founder, investor and board member of Scholar Rock and Morphic Therapeutic. Dr. Springer is a member of the National Academy of Sciences and his honors include the Crafoord Prize, the American Association of Immunologists Meritorious Career Award, the Stratton Medal from the American Society of Hematology, and the Basic Research Prize from the American Heart Association. Dr. Springer received a B.A. from the University of California, Berkeley, and a Ph.D. from Harvard University. Dr. Springer’s extensive knowledge of our business and the nanomedicine field contributed to our board of directors’ conclusion that he should serve as a director of our company.
Patrick Zenner has served as a member of our board of directors since June 2017. Mr. Zenner retired in 2001 from the position of President and Chief Executive Officer of Hoffmann-La Roche Inc., North America, based in Nutley, N.J. Mr. Zenner held various executive positions during his 32-year career with the company. Mr. Zenner is currently a member of the board of trustees of Creighton University and is Chairman of the board of trustees of Fairleigh Dickinson University. In addition, Mr. Zenner is Chairman of the board and a director of West Pharmaceutical Services, Inc. From 2002 until January 2020, Mr. Zenner served as Chairman of the board and a director of ArQule, Inc. Until its sale in 2012, Mr. Zenner was a director of Par Pharmaceuticals, Inc. In 2010, he resigned from the boards of Geron Corporation, Xoma Ltd. and Exact Sciences, Inc. Until its sale in September 2009, Mr. Zenner was a director of CuraGen Corporation. Mr. Zenner received a B.S./B.A. from Creighton University and an M.B.A. from Fairleigh Dickinson University. Mr. Zenner’s extensive experience as a senior pharmaceutical executive and board member to numerous companies in the biotechnology industry contributed to our board of directors’ conclusion that he should serve as a director of our company.
Information about our Executive Officers
Bradford D. Dahms has served as our Chief Financial Officer since September 2019. Prior to joining Selecta Biosciences, Mr. Dahms was a Senior Vice President in the Healthcare Group at Cantor Fitzgerald & Co. from April 2014 to August 2019, focused on providing strategic and financial advice to life sciences companies. Prior to joining Cantor, Mr. Dahms worked as an investment banker at RBC Capital Markets from 2012 to 2014, covering the healthcare sector. Previously, he worked in the Corporate and Investment Banking Group at J.P. Morgan. Mr. Dahms received his a B.S. in Economics, with honors, from the Ohio State University.
Lloyd Johnston, Ph.D. has served as our Chief Operating Officer and Senior Vice President, Research and Development since January 2014. Dr. Johnston served as our Senior Vice President of Pharmaceutical Research, Development and Operations from January 2011 to December 2013 and Vice President of Pharmaceutical Research from July 2008 to January 2011. Prior to joining Selecta, Dr. Johnston was Vice President of Operations for Alkermes, Inc. from 2004 to 2008, and served in several roles, including Director of Manufacturing, from 1999 to 2004, with responsibility for process development, scale-up, and clinical manufacturing for pulmonary and sustained release injectable products, as well as leadership of Alkermes’ manufacturing facility in Chelsea, MA. At Alkermes, Dr. Johnston was also a project leader and member of Steering Committees for numerous products through various stages of development from Phase 1 through registration. Dr. Johnston was an original member of Advanced Inhalation Research Inc., or AIR, a private company formed in 1998 and acquired by Alkermes in 1999. Prior to joining AIR, Dr. Johnston was a lecturer in the Department of Chemical Engineering at the University of New South Wales in Sydney, Australia. He received his B.Sc. in Chemical Engineering from Queen’s University in Ontario, Canada, and his M.S. and Ph.D. in Chemical Engineering from MIT.
Takashi Kei Kishimoto, Ph.D. has served as our Chief Scientific Officer since June 2011. Prior to joining Selecta, Dr. Kishimoto was Vice President of Discovery Research at Momenta Pharmaceuticals, Inc., where he served in several leadership positions from March 2006 to June 2011 and led a multidisciplinary team in advancing both novel and complex generic products for inflammation, oncology, and cardiovascular disease. He served as Senior Director of Inflammation Research at Millennium Pharmaceuticals, Inc. from 1999 to 2006, where he provided the scientific leadership for four programs in clinical development, and as an Associate Director of Research at Boehringer Ingelheim Pharmaceuticals. Dr. Kishimoto has published over 60 peer-reviewed articles in scientific journals, including Nature, Science, Cell and the New England Journal of Medicine. Dr. Kishimoto received his B.A. from New College of the University of South Florida and his Ph.D. in Immunology from Harvard University.
Elona Kogan, J.D. has served as our General Counsel and Secretary since March 2019. Ms. Kogan most recently served as General Counsel and head of Government Relations at ARIAD Pharmaceuticals, a rare disease oncology company, from July 2016 to April 2017, where she was a key executive through the acquisition of the company by Takeda Pharmaceuticals Company Ltd. Prior to joining ARIAD, from May 2011 to August 2015, Ms. Kogan led the legal and government affairs functions of a publicly traded pharmaceutical company dedicated to developing treatments for central nervous system disorders, where she played a central role in the strategic acquisition of the company by Otsuka Pharmaceutical Co. Ltd. Prior roles included positions of increasing responsibility at King Pharmaceuticals, Bristol-Myers Squibb, and Bergen Brunswig Corporation. Ms. Kogan is also a member of the board of directors of Cardax, Inc., a biotechnology company, and serves as the Chairperson of the Compensation Committee, and a member of the Audit Committee. Ms. Kogan is a graduate of the SCALE program at Southwestern University School of Law. Ms. Kogan graduated cum laude from Columbia University, Barnard College, with a degree in economics.
Alison D. Schecter, M.D. has served as our Chief Medical Officer since July 2019. Prior to joining Selecta, Dr. Schecter served as Global Project Head (GPH), Rare Diseases at Sanofi, a pharmaceutical company, from January 2017 to July 2019. She also was the primary BD strategy liaison in Rare Disease. Prior to Sanofi, Dr. Schecter was GPH at Baxalta, a pharmaceutical company, from September 2016 to December 2017. Prior to Baxalta, she was VP of Cardiovascular and Metabolism (CVM) at the J&J Innovation Center, a pharmaceutical company, from January 2015 to September 2016. Previously, she led translational medicine in CVM at Novartis from April 2008 to December 2014. Earlier, Dr. Schecter was Associate Professor in Immunology and Medicine at Mount Sinai. Her innovative academic translational research led to a successful career in biotechnology. She completed her internal medicine residency at Johns Hopkins Hospital, cardiology fellowship at Massachusetts General Hospital, and a research fellowship at Mount Sinai. She earned her M.D. from SUNY Downstate.
Stephen Smolinski has served as our Chief Commercial Officer since October 2017. Mr. Smolinski previously served as the Vice President and Head of the North American Rheumatology Business Unit of Sanofi/Genzyme, a pharmaceutical company from June 2015 to August 2017, where he led the development of the company’s commercialization plans for the rheumatoid arthritis medicine KEVZARA®. Prior to this, he served as Group Vice President of Immunology & Inflammation, Global Strategic Unit at Sanofi a pharmaceutical company, from July 2013 to June 2015. Mr. Smolinski also previously held senior commercial roles at Roche-Genentech, Bristol-Myers Squibb, Johnson & Johnson and Savient Pharmaceuticals. Mr. Smolinski received a B.S. in health care administration from Oregon State University.
PART II
Item 5. Market for Registrant'sRegistrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
Our common stock is publicly traded on The Nasdaq GlobalStock Market under the symbol “SELB.”
Holders
As of March 6, 2020,4, 2022, there were approximately 86,420,195124,288,850 shares of our common stock outstanding held by approximately 6030 holders of record. The actual number of stockholders is greater than this number of record holders, and includes stockholders who are beneficial owners, but whose shares are held in street name by brokers and other nominees.
Dividends
We have never declared or paid any cash dividends on our capital stock. We intend to retain future earnings, if any, to finance the operation and expansion of our business and do not expect to pay any cash dividends in the foreseeable future. Any future determination related to our dividend policy will be made at the discretion of our board of directors after considering our financial condition, results of operations, capital requirements, business prospects and other factors the board of directors deems relevant, and subject to the restrictions contained in any future financing instruments. In addition, our loan and security agreement with Oxford and SVB currently prohibits us from paying dividends on our equity securities, and any future debt agreements may likewise preclude us from paying dividends. See “Management’s Discussion and Analysis of Financial Condition and Results of Operations-LiquidityOperations⸻Liquidity and Capital Resources.”
Stock Performance Graph
The graph set forth below compares the cumulative total stockholder return on our common stock between June 21, 2016 (the date of our initial public offering) and December 31, 2019,2021, with the cumulative total return of (a) the Nasdaq Composite Index and (b) the Nasdaq Biotechnology Index, over the same period. This graph assumes the investment of $100 at the market close on June 21, 2016 in our common stock, the Nasdaq Composite Index and the Nasdaq Biotechnology Index and assumes the reinvestment of dividends, if any. The stock price performance of the following graph is not necessarily indicative of future stock price performance.
COMPARISON OF CUMULATIVE TOTAL RETURN SELECTA BIOSCIENCES, INC.Comparison Of Cumulative Total Return Selecta Biosciences, Inc.,
NASDAQ COMPOSITE INDEX AND NASDAQ BIOTECHNOLOGY INDEX
This performance graph shall not be deemed “soliciting material” or to be “filed” with the SEC for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities under that Section, and shall not be deemed to be incorporated by reference into any of our filings under the Securities or the Exchange Act.
Purchases of Equity Securities by the Issuer or Affiliated Purchasers
We did not repurchase any of our equity securities during the quarter ended December 31, 2019.2021.
Recent Sales of Unregistered Securities and Use of Proceeds from Registered Securities
None.
On December 18, 2019, we entered into a Securities Purchase Agreement (the “2019 Purchase Agreement”) with the purchasers named therein (each, an “Investor” and, collectively, the “Investors”), including certain members of our board of directors. Pursuant to the 2019 Purchase Agreement, we sold (i) an aggregate of 37,634,883 shares of our common stock at a purchase price of $1.46 per share, which was equal to the most recent consolidated closing bid price on the Nasdaq Global Market on December 18, 2019, (ii) warrants to purchase an aggregate of 22,988,501 shares of common stock at a purchase price equal to $0.125 per share underlying each warrant, and (iii) pre-funded warrants to purchase an aggregate of 8,342,128 shares of common stock at a purchase price equal to $1.46 per share underlying each pre-funded warrant, for aggregate net proceeds of approximately $65.6 million, after deducting approximately $4.4 million in placement agent commissions and other transaction costs. Each warrant has an exercise price per share of common stock equal to $1.46 per share. Each pre-funded warrant has an exercise price per share of common stock equal to $0.0001 per share. The closing of the offering occurred on December 23, 2019.
This transaction was exempt from registration pursuant to Section 4(a)(2) of the Securities Act and Rule 506 of Regulation D promulgated thereunder, as a transaction by an issuer not involving a public offering. Each Investor has represented that it is an accredited investor, as defined in Regulation D, and has acquired the securities for investment only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends have been affixed to the securities issued in this transaction.
Item 6. Selected Consolidated Financial Data
You should read the following selected consolidated financial data together with our consolidated financial statements and the related notes and with the “Management’s Discussion and Analysis of Financial Condition and Results of Operations” section of this Annual Report on Form 10-K. The selected consolidated financial data in this section are not intended to replace the consolidated financial statements and are qualified in their entirety by the consolidated financial statements and related notes included elsewhere in this Annual Report on Form 10-K.[Reserved]
We have derived the consolidated statement
Table of operations and comprehensive loss data for the years ended December 31, 2019, 2018, and 2017, and the consolidated balance sheet data as of December 31, 2019, and 2018, from our audited consolidated financial statements included elsewhere in this Annual Report on Form 10-K. The consolidated statement of operations and comprehensive loss for the years ended December 31, 2016 and 2015 the consolidated balance sheet data as of December 31, 2017, 2016 and 2015 are derived from our audited consolidated financial statements that are not included in this Annual Report. Our historical results are not necessarily indicative of the results that should be expected in the future.Contents |
| | | | | | | | | | | | | | | | | | | |
| | Year Ended December 31, |
| (In thousands, except share and per share data) | 2019 | | 2018 | | 2017 | | 2016 | | 2015 |
| Consolidated Statement of Operations Data | | | | | | | | �� | |
| Grant and collaboration revenue | $ | 6,677 |
| | $ | 903 |
| | $ | 207 |
| | $ | 8,083 |
| | $ | 6,011 |
|
| Operating expenses | 59,132 |
| | 65,925 |
| | 63,991 |
| | 42,753 |
| | 31,315 |
|
| Loss from operations | (52,455 | ) | | (65,022 | ) | | (63,784 | ) | | (34,670 | ) | | (25,304 | ) |
| Loss on extinguishment of debt | — |
| | — |
| | (673 | ) | | — |
| | — |
|
| Change in fair value of warrant liabilities | (857 | ) | | — |
| | — |
| | — |
| | — |
|
| Other income (expense), net | (2,038 | ) | | (314 | ) | | (864 | ) | | (1,540 | ) | | 130 |
|
| Net loss | $ | (55,350 | ) | | $ | (65,336 | ) | | $ | (65,321 | ) | | $ | (36,210 | ) | | $ | (25,174 | ) |
| Net loss per share: | | | | | | | | | |
| Basic and diluted | $ | (1.22 | ) | | $ | (2.92 | ) | | $ | (3.20 | ) | | $ | (3.89 | ) | | $ | (15.13 | ) |
| Weighted average common shares outstanding: | | | | | | | | | |
| Basic and diluted | 45,548,511 |
| | 22,389,286 |
| | 20,425,050 |
| | 10,493,939 |
| | 2,150,422 |
|
| | | | | | | | | | |
| | As of December 31, |
| (In thousands) | 2019 | | 2018 | | 2017 | | 2016 | | 2015 |
| Consolidated Balance Sheet Data | | | | | | | | | |
| Cash, cash equivalents, short-term deposits and investments | $ | 89,893 |
| | $ | 37,403 |
| | $ | 96,562 |
| | $ | 84,141 |
| | $ | 36,462 |
|
| Total assets | $ | 99,569 |
| | $ | 44,482 |
| | $ | 101,100 |
| | $ | 89,301 |
| | $ | 42,824 |
|
| Loans payable | $ | 18,905 |
| | $ | 21,385 |
| | $ | 21,042 |
| | $ | 12,044 |
| | $ | 11,855 |
|
| Warrant liabilities | $ | 41,549 |
| | $ | — |
| | $ | — |
| | $ | — |
| | $ | — |
|
| Redeemable convertible preferred stock | $ | — |
| | $ | — |
| | $ | — |
| | $ | — |
| | $ | 137,482 |
|
| Total stockholders' equity (deficit) | $ | 8,397 |
| | $ | (5,418 | ) | | $ | 51,814 |
| | $ | 54,957 |
| | $ | (116,493 | ) |
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with “Item 6. Selected Consolidated Financial Data” and our consolidated financial statements and related notes appearing elsewhere in this Annual Report on Form 10-K. This discussion and other parts of this Annual Report on Form 10-K contain forward-looking statements that involve risks and uncertainties, such as statements regarding our plans, objectives, expectations, intentions and projections. Our actual results could differ materially from those discussed in these forward-looking statements. Important factors that could cause or contribute to such differences include, but are not limited to, those discussed in Item 1A1A. “Risk Factors.” A discussion of the year ended December 31, 20182020 compared to the year ended December 31, 20172019 has been reported previously in our Annual Report on Form 10-K for the year ended December 31, 2018,2020, filed with the SEC on March 15, 2019,12, 2021, under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
OVERVIEWOverview
We are a clinical-stage biopharmaceutical company using our ImmTOR platform with the goal to effectively and safely treat rare and serious diseases by enabling the development of novel biologic therapies that would otherwise be limited by their immunogenicity. Many such diseases are treated with biologic therapies that are foreign to the patient’s immune system and therefore elicit an undesired immune response.
company. Our proprietary tolerogenic ImmTORImmTOR® platform encapsulates rapamycin, also known as sirolimus, an FDA approved immunomodulator, in biodegradable nanoparticles andImmTOR is designed to mitigate the formation of ADAs by inducinginduce antigen-specific immune tolerance.
We continually seek to enhance ImmTOR. In recent preclinical studies we have conducted, we have observed that ImmTOR may have synergistic activity with interleukin-2, or IL-2, molecules that have been engineered to be selective for regulatory T cells, or Tregs. Treg-selective IL-2 mutant molecules, or IL-2 muteins, have been shown to transiently expand all pre-existing Tregs in preclinical studies conducted by others. We have observed in preclinical studies that the combination of ImmTOR, a Treg-selective IL-2 mutein and an antigen elicited an approximately three-fold increase in antigen-specific Tregs beyond ImmTOR alone with evidence of enhanced durability of immune tolerance and the potential for ImmTOR dose sparing. This combination of ImmTOR with a Treg selective IL-2 molecule represents an evolution of the ImmTOR platform, which we call ImmTOR-IL™. We believe this combination has the potential to biologic drugs. be a best-in-class therapy in diseases where expansion of total Tregs may prove beneficial.
We believe ImmTOR hasand ImmTOR-IL have the potential to enhance both the efficacy without compromising theand safety of existing approved biologic drugs,therapies (including gene therapies), improve product candidates under development, and enable novel therapeutic modalities such as re-administration of systemic gene therapy.
Our Current Programs
Chronic Refractory Gout
Our lead product candidate, SEL-212, is designedin autoimmune disease. In clinical trials, ImmTOR has been observed to be a monthly treatment for chronic refractory gout, a debilitating rare disease with an unmet medical need. SEL-212 consists of a combination of our ImmTOR platform co-administered with pegadricase. Pegadricase is an investigational recombinant pegylated uricase (urate oxidase), an enzyme not naturally found in humans, and is therefore highly immunogenic. This enzyme is designed to treat patients with symptomatic gout, refractory to standard uric acid lowering treatment, by breaking down the excess uric acid to the more soluble allantoin. In preclinical studies, we observed that ImmTOR, when co-administered with pegadricase, induced antigen-specific immune tolerance to pegadricase and substantially reducedinhibit the formation of associated ADAs. Basedneutralizing antibodies to adeno-associated virus (AAV) capsids, potentially enabling re-dosing of gene therapies. Additionally, based on our Phase 1/2 clinicalpreclinical data in AAV gene therapies, we believe that SEL-212ImmTOR has the potential to control SUA levelsimprove efficacy and mitigatesafety by increasing transgene expression, reducing hepatic inflammation and inhibiting undesired immune responses to both the formation of ADAs in response toAAV capsid and the therapeutic enzyme.
Our Phase 1 data provided evidencetransgene product that ImmTOR mitigatedcan occur with the formation of ADAs against pegadricase in a dose-dependent manner after a singlefirst dose of SEL-212.gene therapy. In our Phase 2 trial,biologic therapies, clinical activity of ImmTOR inhibitedin humans has been observed with pegadricase, a highly immunogenic pegylated uricase enzyme being developed for the formationtreatment of ADAs in patients with up to five monthly doses, resulting in sustained reduction of SUA levels. We also observed a lower-than-expected rate of gout flares in the first months after initiation of SEL-212 treatment, with further reductions observed in months three to five. SEL-212, if successfully developed and approved, has the potential to offer a unique treatment for patients with chronic refractory gout including reduced immunogenicity, improvedto conventional therapy. The combination of ImmTOR and pegadricase is currently being evaluated in a Phase 3 clinical trial that we are conducting on behalf of our partner Swedish Orphan Biovitrum AB, or Sobi. We intend to pursue development of therapies for autoimmune diseases where expansion of either all Tregs or antigen-specific Tregs has been shown to, or we believe is, likely to have a beneficial effect. We believe that ImmTOR and ImmTOR-IL have the potential to unlock antigen-specific therapies for autoimmune diseases and that ImmTOR-IL can further improve the efficacy and monthly dosing comparedsafety profile of biologic therapies beyond ImmTOR alone.
Impact of COVID-19
We are closely monitoring how the COVID-19 pandemic is affecting our employees, business, preclinical studies and clinical trials. In response to other FDA-approved treatments, and provide clinical evidence supporting the utilityspread of COVID-19, we have continued to have our administrative employees work outside of our ImmTOR platformoffices and limited the number of staff in providing patients with antigenic specific tolerance.
In March 2019, we initiatedany given research and development laboratory. Disruptions caused by the COVID-19 pandemic may result in difficulties or delays in initiating, enrolling, conducting or completing our planned and ongoing clinical trials, and the incurrence of unforeseen costs as a Phase 2 head-to-headresult of preclinical study or clinical trial of SEL-212 (COMPARE), in which SEL-212 is being compared againstdelays.
While the current FDA-approved therapy for chronic refractory gout, KRYSTEXXA, in multipleCOVID-19 pandemic has not had a material impact on our clinical sites in the United States. We completed enrollment of our Phase 2 head-to-head (COMPARE) clinical study against KRYSTEXXA in December 2019 and expect to report top-line data in the third quarter of 2020. The two-armed, open label trial has enrolled approximately 150 patients, randomized 1:1, with one arm receiving KRYSTEXXA (as set forth in the prescribing information) and the other arm receiving six monthly doses of SEL-212. The primary endpoint in the study is the percentage of patients in each arm that maintain SUA control below 6.0 mg/dL, for at least 80%programs as of the time during months three and six. We plandate of this Annual Report, it could have an impact on our ability to commencecomplete the Phase 3 DISSOLVE clinical program of SEL-212, as the pandemic presents the potential to experience delays in SEL-212 inenrollment as well as the second halfinability of 2020. We will require additional resourcescertain patients to complete the planned Phase 3trial due to suffering from COVID-19, our ability to commence preclinical studies and clinical program for SEL-212.
We expecttrials of our clinicalIgA nephropathy, gene therapy, and if approved, marketing strategyautoimmune disease programs, and our ability to obtain supply of both active drug substances and finished drug product as well as efficient execution of the overall supply chain for SEL-212 to initially focusand our other programs.
At this time, any impact of COVID-19 on our business, revenues, results of operations and financial condition will largely depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the estimated 160,000 patientsduration of the pandemic, the emergence of new virus variants, travel restrictions and social distancing in the United States with chronic refractory gout, and other countries, business closures or disruptions, supply chain disruptions, the ultimate impact on financial markets and the global economy, and the effectiveness of actions taken in the United States and other countries to focus on those patients that are being treated by rheumatologists.contain and treat the disease.
Gene Therapy
In August 2019, we entered into a feasibility study and license agreement with AskBio, the AskBio Collaboration Agreement, pursuant to which we and AskBio will conduct proofAdditionally, in December 2019 we entered into the AskBio License Agreement which provides AskBio with exclusive worldwide rights to our ImmTOR platform to research, develop and commercialize certain AAV-gene therapy products targeting the GAA gene, or derivatives thereof, to treat Pompe Disease.
In September 2018, we announced a collaboration with the European consortium, CureCN, for an ImmTOR+AAV gene therapy combination product candidate in Crigler-Najjar syndrome, a rare genetic disorder characterized by an inability to properly convert and clear bilirubin from the body. We expect the CureCN consortium to obtain scientific advice from the German drug regulatory authority in 2020.
In December 2016, we entered into the Spark License Agreement which provides Spark with exclusive worldwide rights to our ImmTOR platform to research, develop and commercialize gene therapies for Factor VIII, an essential blood clotting protein relevant to the treatment of hemophilia A.
Our proprietary gene therapy product candidate, SEL-313, is being developed to treat OTC deficiency and is currently in preclinical development.
FINANCIAL OPERATIONS OVERVIEW
Financial Operations
To date, we have financed our operations primarily through the public offeringofferings and private placements of our securities, funding received from research grants, collaboration and collaborationlicense arrangements and our credit facility. We do not have any products approved for sale and have not generated any product sales. All of our revenue to date has been collaboration and grant revenue.
Since inception, we have incurred significant operating losses. We incurred net losses of $55.4$25.7 million and $65.3$68.9 million for the years ended December 31, 20192021 and 2018,2020, respectively. As of December 31, 2019,2021, we had an accumulated deficit of $335.8$430.3 million. We expect to continue to incur significant expenses and operating losses for at least the next several years as we:
| |
- | conduct additional clinical trials for SEL‑212; |
| |
- | continue the research and development of our other product candidates as well as product candidates that we may be developing jointly with collaboration partners; |
| |
- | seek to enhance our ImmTOR platform and discover and develop additional product candidates; |
| |
- | seek to enter into collaboration, licensing and other agreements, including, but not limited to research and development, and/or commercialization agreements; |
| |
- | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
| |
- | potentially establish a sales, marketing and distribution infrastructure and scales‑up external manufacturing capabilities to commercialize any products for which we may obtain regulatory approval; |
| |
- | maintain, expand and protect our intellectual property portfolio, including through licensing arrangements; and |
| |
- | add clinical, scientific, operational, financial and management information systems and personnel, including personnel to support our product development and potential future commercialization efforts and to support our operations as a public company. |
•continue the research and development of our other product candidates as well as product candidates that we may be developing jointly with collaboration partners;
•seek to enhance our ImmTOR platform and discover and develop additional product candidates;
•seek to enter into collaboration, licensing and other agreements, including, but not limited to research and development, and/or commercialization agreements;
•seek regulatory approvals for any product candidates that successfully complete clinical trials;
•potentially establish a sales, marketing and distribution infrastructure and scales-up external manufacturing capabilities to commercialize any products for which we may obtain regulatory approval;
•maintain, expand and protect our intellectual property portfolio, including through licensing arrangements; and
•add clinical, scientific, operational, financial and management information systems and personnel, including personnel to support our product development and potential future commercialization efforts and to support our operations as a public company.
Until such time, if ever, as we can generate substantial product revenues, if ever, we expect to finance our cash needs through a combination of equity offerings, debt financings, and license and collaboration agreements, and research grants.agreements. We may be unable to raise capital when needed or on reasonable terms, if at all, which would force us to delay, limit, reduce or terminate our product development or future commercialization efforts. We will need to generate significant revenues to achieve profitability, and we may never do so.
We will require additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212. Under the terms of our exclusive patent license agreement with the Massachusetts Institute of Technology, or the MIT License, MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. On December 13, 2019, we entered into the Fourth Amendment, which we refer to as the MIT Amendment, to the Exclusive Patent License Agreement by and between us and the Massachusetts Institute of Technology, or the MIT Agreement. Pursuant to the MIT Amendment, a provision of the MIT Agreement under which we were obligated to initiate a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT Agreement. Further, pursuant to the MIT Amendment, the parties agreed to negotiate in good faith to enter into a future amendment to the MIT Agreement after we provide MIT with an amended diligence plan. If we are unable to reach an agreement with MIT regarding an acceptable amendment of the MIT License and if we are unable to cure the breach, there could be a material adverse effect on our business.
We believe that our existing cash, cash equivalents, investments,restricted cash, and restricted cashmarketable securities as of December 31, 20192021 will enable us to fund our operating expenses and capital expenditure requirements into the firstthird quarter of 2021. Because our current operating plan does not contain sufficient resources, we will require additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212.2023. We have based this estimate on assumptions that may prove to be wrong, and we could use our capital resources sooner than we currently expect. Because of the uncertainty in securing additional capital, we have concluded that substantial doubt exists with respect to our ability to continue as a going concern within one year after the date of the filing of this Annual Report on Form 10-K. For additional information, see “Liquidity and Capital Resources.”
The consolidated financial information presented below includes the accounts of Selecta Biosciences, Inc. and our wholly owned subsidiaries, Selecta (RUS) LLC, a Russian limited liability company, or Selecta RUS,(RUS), and Selecta Biosciences Security Corporation, a Massachusetts securities corporation. All intercompany accounts and transactions have been eliminated.
GrantCollaboration and collaborationlicense revenue
To date, we have not generated any revenue from product sales. Our revenue consists primarily of grantcollaboration and collaborationlicense revenue, which includes amounts recognized related to upfront and milestone payments for research and development funding under collaboration and license agreements. In addition, we earn revenue under the terms of government contracts or grants, which require the performance of certain research and development activities. We expect that any revenue we generate will fluctuate from quarter to quarter because of the timing and amountamounts of fees, research and development reimbursements and other payments from collaborators. We do not expect to generate revenue from product sales for at least the next several years. If we or our collaborators fail to complete the development of our product candidates in a timely manner or fail to obtain regulatory approval as needed, our ability to generate future revenue will be harmed, and will affect the results of our operations and financial position. For a further description of the agreements underlying our collaboration and grant‑basedlicense revenue, see Notes 2 and 12 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.Report.
Research and development
Our research and development expenses consist of external research and development costs, which we track on a program‑by‑programprogram-by-program basis and primarily include CMO relatedCMO-related costs, fees paid to CROs and internal research and development costs, which are primarily compensation expenses for our research and development employees, lab supplies, analytical testing, allocated overhead costs and other related expenses. Our internal research and development costs are often devoted to expanding our programs and are not necessarily allocable to a specific target.
We have incurred a total of $240.8$364.0 million in research and development expenses from inception through December 31, 2019,2021, with a majority of the expenses being spent on the development of SEL‑212 and a prior nicotine vaccine candidate,SEL-212 and the remainder being spent on our various discovery and preclinical stage product candidate programs and the general expansion of our technology.
In connection with our intention to focus on advancing our ImmTOR platform, as stated in January 2019, we have ceased ongoing work on our immune stimulation programs SELA-070 and SEL-701, and currently do not have plans to move these programs forward or to perform any additional work on either
As we expand the clinical development of SEL‑212 and our gene therapy programs, we expect our research and development expenses to increase.
We expense research and development costs as incurred. Conducting a significant amount of research and development is central to our business model. Product candidates in clinical development generally have higher development costs than those in earlier stages of development, primarily due to the size, duration and cost of clinical trials. We plan to increase our research and development expenses for the foreseeable future as we seek to complete development of SEL‑212, and to further advance our preclinical and earlier stage research and development projects. The successful development of our clinical and preclinical product candidates is highly uncertain. At this time, we cannot reasonably estimate the nature, timing or costs of the efforts that
will be necessary to complete the development of SEL‑212 or any of our preclinical programs or the period, if any, in which material net cash inflows from these product candidates may commence. Clinical development timelines, the probability of success and development costs can differ materially from our expectations. For example, if the FDA or another regulatory authority were to require us to conduct clinical trials beyond those which we currently expect will be required for the completion of clinical development of a product candidate, or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time to complete any clinical development.
The following table sets forth the components of our research and development expenses during the periods indicated (in thousands):
| | | | | | | | | | | Year Ended December 31, | |
| | Year ended December 31, | | | 2021 | | 2020 | | 2019 | |
| SEL-212 | | SEL-212 | | $ | 31,446 | | | $ | 32,288 | | | $ | 25,489 | | |
| AskBio collaboration | | AskBio collaboration | | 3,888 | | | 2,807 | | | — | | |
| | 2019 | | 2018 | | 2017 | |
| Research and development expenses (key projects and initiatives): | | | | | | |
| SEL-212 | $ | 25,489 |
| | $ | 22,770 |
| | $ | 19,593 |
| |
| SELA-070 | 46 |
| | 1,602 |
| | 1,227 |
| |
| Discovery and preclinical stage product candidate programs, collectively | 1,614 |
| | 2,623 |
| | 6,933 |
| |
| Preclinical stage product candidate programs | | Preclinical stage product candidate programs | | 11,080 | | | 1,717 | | | 1,660 | | |
| Other internal research and development expenses | 15,594 |
| | 20,692 |
| | 17,412 |
| Other internal research and development expenses | | 22,322 | | | 17,693 | | | 15,594 | | |
| Total research and development expenses | $ | 42,743 |
| | $ | 47,687 |
| | $ | 45,165 |
| Total research and development expenses | | $ | 68,736 | | | $ | 54,505 | | | $ | 42,743 | | |
In June 2020, we and Sobi entered into the Sobi License. Pursuant to the Sobi License, clinical trial costs incurred to complete development of SEL-212, including but not limited to costs incurred while conducting and completing the Phase 3 DISSOLVE trials, will be reimbursed by Sobi. These costs, when reimbursed, will be recognized as revenue consistent with the revenue recognition methodology disclosed in Note 12 to our consolidated financial statements included elsewhere in this Annual Report. The reimbursable costs exclude any costs of additional development activities required that are related to ImmTOR and that are unrelated to SEL-212.
General and administrative
General and administrative expenses consist primarily of salaries and related benefits, including stock-based compensation, related to our executive, finance, business development and support functions. Other general and administrative expenses include facility-related costs not otherwise allocated to research and development expenses, travel expenses for our general and administrative personnel and professional fees for auditing, tax and corporate legal services, including intellectual property-related legal services.
Investment income
Investment income consists primarily of interest income earned on our cash, and cash equivalents and short-term investments.marketable securities.
Interest expense
Interest expense consists of interest expense on amounts borrowed under our credit facilities.
Other income (expense)
Other income (expense)was de minimis during the years ended December 31, 2021 and 2020, and for the year ended December 31, 2019 it consists primarily of issuance fees associated with warrant liabilities for the year ended December 31, 2019, and was de minimis for each of the years ended December 31, 2018 and 2017.liabilities.
Change in fair value of warrant liabilities
Common warrants classified as liabilities are remeasured at fair value, utilizing a Black-Scholes valuation methodology, quarterly with the change in fair value recognized as a component of earnings.
Foreign currency transaction gain (loss)
The functional currency of our Russian subsidiary is the Russian ruble. In addition to holding cash denominated in Russian rubles, our Russian bank accounts also hold cash balances denominated in U.S. dollars to facilitate payments to be settled in U.S. dollars or other currencies. At eachAs of December 31, 20192021 and December 31, 2018,2020, we maintained cash of $0.4$0.3 million in Russian banks, all of which was denominated in U.S. dollars. The amounts denominated in U.S. dollars and used in transacting the day-to-day operations of our Russian subsidiary are subject to transaction gains and losses, which are reported as incurred.
CRITICAL ACCOUNTING POLICIES AND USE OF ESTIMATES
Results of Operations
Comparison of the Years Ended December 31, 2021 and 2020
Collaboration and license revenue
The following is a comparison of collaboration and license revenue for the years ended December 31, 2021 and 2020 (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | |
| | Year Ended
December 31, | | Increase |
| | 2021 | | 2020 | | (decrease) |
| | | | | |
| Collaboration and license revenue | $ | 85,077 | | | $ | 16,597 | | | $ | 68,480 | | | 413 | % |
| | | | | | | |
During the years ended December 31, 2021 and 2020, we recognized $83.5 million and $16.6 million, respectively, under the license agreement with Sobi resulting from both the shipment of clinical supply and the reimbursement of costs incurred for the Phase 3 DISSOLVE clinical program. The significant revenue increase is the result of the continued enrollment of the Phase 3 DISSOLVE clinical program that was initiated in the third quarter of 2020. Additionally, during the year ended December 31, 2021, we recognized $1.0 million under the license agreement with Takeda, $0.4 million for shipments under the license agreement with Sarepta, and $0.1 million resulting from the expiration of the contractual audit term under the Skolkovo Foundation grant.
Research and development
The following is a comparison of research and development expenses for the years ended December 31, 2021 and 2020 (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Year Ended
December 31, | | | | Increase |
| | 2021 | | 2020 | | | | | | | | (decrease) |
| SEL-212 | $ | 31,446 | | | $ | 32,288 | | | | | | | | | $ | (842) | | | (3) | % |
| AskBio collaboration | 3,888 | | | 2,807 | | | | | | | | | 1,081 | | | 39 | % |
| Preclinical stage product candidate programs | 11,080 | | | 1,717 | | | | | | | | | 9,363 | | | 545 | % |
| Other internal research and development expenses | 22,322 | | | 17,693 | | | | | | | | | 4,629 | | | 26 | % |
| Total research and development expenses | $ | 68,736 | | | $ | 54,505 | | | | | | | | | $ | 14,231 | | | 26 | % |
During the year ended December 31, 2021, our research and development expenses increased by $14.2 million, or 26%, as compared to 2020. The increase in cost was primarily the result of expenses incurred for the preclinical programs, salaries for increases in headcount and AskBio collaboration costs. Additionally, we paid Genovis and Ginkgo, $4.0 million and $0.5 million, respectively, for in-license agreements.
General and administrative
The following is a comparison of general and administrative expenses for the years ended December 31, 2021 and 2020 (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | |
| | Year Ended
December 31, | | Increase |
| | 2021 | | 2020 | | (decrease) |
| | | | | |
| General and administrative | $ | 20,938 | | | $ | 18,913 | | | $ | 2,025 | | | 11 | % |
During the year ended December 31, 2021, our general and administrative expenses increased by $2.0 million, or 11%, as compared to 2020. The increase in costs was primarily the result of stock compensation and consulting fees, offset primarily by reduction in professional fees.
Investment income
Investment income was de minimis for the years ended December 31, 2021 and 2020, respectively.
Loss on extinguishment of debt
For the year ended December 31, 2020, we recognized a $0.5 million loss on extinguishment of the 2017 Term Loan (see Note 9).
Foreign currency transaction gain (loss)
We recognized de minimis foreign currency gains during each of the years ended December 31, 2021 and 2020.
Interest expense
Interest expense was $2.8 million and $1.6 million for the years ended December 31, 2021 and 2020, respectively, representing interest expense and amortization of the carrying costs of our credit facilities.
Change in fair value of warrant liabilities
For the year ended December 31, 2021, we recognized $2.3 million of loss from the change in the fair value of warrant liabilities utilizing the Black-Scholes valuation methodology. The increase in value was primarily driven by a slight increase in the Company’s share price, offset by the decrease of outstanding warrants. For the year ended December 31, 2020, we recognized $10.4 million change for the increase in the fair value of warrant liabilities primarily driven by an increase in the share price and volatility.
Other income (expense)
Other income (expense) was de minimis for each of the years ended December 31, 2021 and 2020.
Income taxes
For the year ended December 31, 2021, we recognized $16.0 million of expense for the income taxes primarily related to the license agreement with Sobi upon the Company’s election to opt out of the installment sale method of taxation. As a result of this election, the Company has prepaid all taxes related to future Sobi License revenue streams.
Net loss
Net loss for the year ended December 31, 2021 decreased to $25.7 million as compared to a net loss of $68.9 million in 2020 primarily due to increased revenue of $66.9 million under the license agreement with Sobi resulting from both the shipment of clinical supply and the reimbursement of costs incurred for the Phase 3 DISSOLVE clinical program, partially offset by increased research and development expenses on preclinical programs.
Liquidity and Capital Resources
Since our inception, we have incurred recurring net losses. We expect that we will continue to incur losses and that such losses will increase for the foreseeable future. We expect that our research and development and general and administrative expenses will continue to increase and, as a result, we will need additional capital to fund our operations, which we may raise through a combination of equity offerings, debt financings, third-party funding and other collaborations and strategic alliances.
From our inception through December 31, 2021, we have raised an aggregate of $637.5 million to fund our operations, which includes $118.5 million from the sale of preferred stock, $11.1 million in government grant funding, $36.7 million from borrowings under our credit facilities past and present, $202.4 million from our collaborations and license agreements, $64.5 million in combined net proceeds from our initial public offering, $149.3 million in combined net proceeds from private placements and follow-on offerings of our common stock, and, through December 31, 2021, $55.0 million in aggregate net proceeds from “at-the-market” offerings of our common stock.
As of December 31, 2021, our cash, cash equivalents, restricted cash, and marketable securities were $129.4 million, of which $1.4 million was restricted cash related to lease commitments and $0.3 million was held by our Russian subsidiary designated solely for use in its operations. Our Russian subsidiary cash is consolidated for financial reporting purposes.
In addition to our existing cash equivalents, we receive research and development funding pursuant to our collaboration and license agreements. Currently, funding from payments under our collaboration and license agreements represent our only source of committed external funds.
Collaboration and License Agreements
In-licenses
In October 2021, we entered into the Ginkgo Agreement, and paid Ginkgo a $0.5 million one-time upfront payment and we entered into the Genovis Agreement, and paid Genovis a $4.0 million one-time upfront payment.
On September 7, 2021, we entered into the Cyrus Agreement, and purchased 2,326,934 shares of Cyrus’ Series B Preferred Stock, par value $0.0001 per share at a purchase price of $0.8595 per share for $2.0 million.
Out-licenses
On October 1, 2021, we entered into the Takeda Agreement. We received a $3.0 million upfront payment and are entitled to receive up to $1.124 billion in future additional payments over the course of the partnership that are contingent on the achievement of development or commercial milestones or Takeda’s election to continue its activities at specified development stages.
In June 2020, we entered into the Sobi License. Sobi paid us a one-time, upfront payment of $75 million, and upon the closing of the Sobi Private Placement, we received an additional $25 million from Sobi in consideration for Sobi’s purchase of our common stock at $4.6156 per share. We are eligible to receive $630 million in milestone payments upon the achievement of various development and regulatory milestones and sales thresholds for annual net sales of SEL-212, and tiered royalty payments ranging from the low double digits on the lowest sales tier to the high teens on the highest sales tier. Sobi has agreed to fund the Phase 3 clinical program of SEL-212, which commenced in September 2020. We expect this to substantially reduce our annual operating expenses. Additionally, in June 2020, we entered into the Sarepta Agreement. Sarepta paid us a $2.0 million upfront payment upon closing, and a $3.0 million for the achievement of certain pre-clinical milestones in June 2021.
In December 2019, we entered into the AskBio License Agreement. Pursuant to the AskBio License Agreement, AskBio has exercised its option to exclusively license intellectual property rights covering ImmTOR to research, develop, and commercialize certain AAV gene therapy products utilizing ImmTOR, and targeting the GAA gene, or derivatives thereof, to treat Pompe Disease. We received $7.0 million of upfront fees pursuant to the AskBio License Agreement and are eligible to receive $237 million in milestone payments, and royalties on net sales ranging from the mid-to-high single digits.
Financings
In August 2017, we entered into a sales agreement, or the 2017 Sales Agreement, with Jefferies LLC, as sales agent, to sell shares of our common stock with an aggregate value of up to $50.0 million in an “at-the-market” offering. In August 2020, concurrent with the filing of a new shelf registration statement, we entered into a new sales agreement, or the 2020 Sales Agreement, with Jefferies LLC, as sales agent, pursuant to which we may, from time to time, issue and sell common stock with an aggregate value of up to $50.0 million in an “at-the-market” offering. The 2017 Sales Agreement terminated pursuant to its terms in August 2020. On October 8, 2021, we delivered notice to Jefferies LLC that we were terminating the 2020 Sales Agreement, with effect as of October 19, 2021.
On October 25, 2021, we entered into a Sales Agreement, or the 2021 Sales Agreement, with SVB Leerink LLC to sell shares of our common stock, from time to time, through an “at the market” equity offering program under which SVB Leerink will act as sales agent. The shares of common stock sold pursuant to the 2021 Sales Agreement will be issued pursuant to our shelf registration statement on Form S-3 (File No. 333-241692), filed on August 6, 2020 with the Securities and Exchange Commission and related prospectus supplement, filed on October 25, 2021 with the Securities and Exchange Commission, for aggregate gross sales proceeds of up to $75.0 million.
During the year ended December 31, 2021, we sold 13,767,511 shares of our common stock pursuant to the 2021 and 2020 Sales Agreements, as applicable, for aggregate net proceeds of $51.9 million, after deducting commissions and other transaction costs. During the year ended December 31, 2020, we sold 1,069,486 shares of our common stock pursuant to the 2020 and 2017 Sales Agreements, as applicable, for aggregate net proceeds of $2.1 million, after deducting commissions and other transaction costs.
Indebtedness
On August 31, 2020, we entered into a term loan of up to $35.0 million, consisting of term loans in an aggregate amount of $25.0 million, or the Term A Loan, and term loans in an aggregate amount of $10.0 million, or the Term B Loan, governed by a loan and security agreement among us and Oxford Finance LLC, or Oxford, as collateral agent and a lender, and Silicon Valley Bank, or SVB, as a lender. The Term A Loan was funded in full on August 31, 2020, the proceeds of which were used to repay our previously existing 2017 Term Loan and for general corporate and working capital purposes. The second draw period expired on September 30, 2021 and the Term B Loan is no longer available to be drawn in the future.
The 2020 Term Loan is secured by a lien on substantially all of our assets, other than intellectual property, provided that such lien on substantially all assets includes any rights to payments and proceeds from the sale, licensing or disposition of intellectual property. We also granted Oxford a negative pledge with respect to our intellectual property.
The 2020 Term Loan contains customary covenants and representations, including but not limited to financial reporting obligations and limitations on dividends, indebtedness, collateral, investments, distributions, transfers, mergers or acquisitions, taxes, corporate changes, deposit accounts, and subsidiaries. The 2020 Term Loan also contains other customary provisions, such as expense reimbursement, non-disclosure obligations as well as indemnification rights.
The events of default under the 2020 Term Loan include, but are not limited to, our failure to make any payments of principal or interest under the 2020 Term Loan or other transaction documents, our breach or default in the performance of any covenant under the 2020 Term Loan or other transaction documents, the occurrence of a material adverse event, making a false or misleading representation or warranty in any material respect under the 2020 Term Loan, our insolvency or bankruptcy, any attachment or judgment on our assets of at least approximately $0.5 million, or the occurrence of any default under any of our
agreements or obligations involving indebtedness in excess of approximately $0.5 million. If an event of default occurs, Oxford and SVB are entitled to take enforcement action, including acceleration of amounts due under the 2020 Term Loan. If we raise any additional debt financing, the terms of such additional debt could further restrict our operating and financial flexibility.
For a further description of the 2020 Term Loan, see Note 9 to our consolidated financial statements included elsewhere in this Annual Report.
Future funding requirements
As of the date of this Annual Report, we have not generated any revenue from product sales. We do not know when, or if, we will generate revenue from product sales. We will not generate significant revenue from product sales unless and until we obtain regulatory approval and commercialize one of our current or future product candidates. Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, third-party clinical research and development services, laboratory and related supplies, clinical costs, legal and other regulatory expenses, and general overhead costs. We expect that we will continue to generate losses for the foreseeable future, and we expect the losses to increase as we continue the development of, and seek regulatory approvals for, our product candidates, and begin to commercialize any approved products. We are subject to risks in the development of our products, and we may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may adversely affect our business. We expect that we will need substantial additional funding to support our continuing operations.
As of December 31, 2021, we had an accumulated deficit of $430.3 million. We anticipate operating losses to continue for the foreseeable future due to, among other things, costs related to research, development of our product candidates, conducting preclinical studies and clinical trials, and our administrative organization. We will require substantial additional financing to fund our operations and to continue to execute our strategy, and we will pursue a range of options to secure additional capital.
We are continually evaluating various potential sources of additional funding such as strategic collaborations, license agreements and the issuance of equity to fund our operations. If we raise additional funds through strategic collaborations and alliances, which may include existing collaboration partners, we may have to relinquish valuable rights to our technologies or product candidates, or grant licenses on terms that are not favorable to us. To the extent that we raise additional capital through the sale of equity, the ownership interest of our existing shareholders will be diluted, and other preferences may be necessary that adversely affect the rights of existing shareholders.
We believe that our existing cash, cash equivalents, restricted cash, and marketable securities as of December 31, 2021 will enable us to fund our current planned operations into the third quarter of 2023, though we may realize additional cash resources upon the achievement of certain contingent collaboration milestones or it may pursue additional cash resources through public or private equity or debt financings or by establishing collaborations with other companies. Management’s expectations with respect to our ability to fund current and long-term planned operations are based on estimates that are subject to risks and uncertainties. If actual results are different from management’s estimates, we may need to seek additional strategic or financing opportunities sooner than would otherwise be expected. However, there is no guarantee that any collaboration milestones will be achieved or that any of these strategic or financing opportunities will be executed on favorable terms, and some could be dilutive to existing stockholders. If we are unable to obtain additional funding on a timely basis, we may be forced to significantly curtail, delay, or discontinue one or more of its planned research or development programs or be unable to expand our operations, meet long-term obligations or otherwise capitalize on our commercialization of our product candidates.
Additionally, while the potential economic impact brought by and the duration of the COVID-19 pandemic may be difficult to assess or predict, the widespread pandemic has resulted in, and may continue to result in, significant disruption of global financial markets, reducing our ability to access capital as and when needed. We have based this estimate on assumptions that may prove to be wrong, and we could use our capital resources sooner than we currently expect.
Our future capital requirements will depend on many factors, including:
•the number of product candidates that we pursue;
•our collaboration agreements remaining in effect, our entering into additional collaboration agreements and our ability to achieve milestones under these agreements;
•the cost of manufacturing clinical supplies of our product candidates;
•our headcount growth and associated costs;
•the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials for our other product candidates;
•the costs, timing and outcome of regulatory review of our product candidates;
•the costs and timing of future commercialization activities, including manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval;
•the revenue, if any, received from commercial sales of our product candidates for which we receive marketing approval;
•the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property-related claims;
•the effect of competing technological and market developments; and
•the extent to which we acquire or invest in businesses, products and technologies, including entering into licensing or collaboration arrangements for product candidates.
Summary of Cash Flows
| | | | | | | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| (In thousands) | | | | 2021 | | 2020 | | 2019 |
| Cash (used in) and provided by: | | | | | | | | |
| Operating activities | | | | $ | (60,382) | | | $ | 34,881 | | | $ | (51,435) | |
| Investing activities | | | | (17,140) | | | (741) | | | 229 | |
| Financing activities | | | | 52,897 | | | 14,431 | | | 105,041 | |
| Effect of exchange rate changes on cash | | | | (3) | | | (58) | | | 34 | |
| Net change in cash, cash equivalents, and restricted cash | | | | $ | (24,628) | | | $ | 48,513 | | | $ | 53,869 | |
Operating activities
Net cash used in operating activities for the year ended December 31, 2021 was $60.4 million compared to $34.9 million provided in the same period in 2020. The decrease in net cash used in operating activities was primarily due to $12.2 million of net losses, adjusted for non-cash items, and uses of cash of approximately $48.2 million for changes in operating assets and liabilities.
Investing activities
Net cash used in investing activities for the year ended December 31, 2021 was $17.1 million compared to net cash used in investing activities of $0.7 million in the same period in 2020. The net cash used in investing activities in 2021 was primarily to purchase marketable securities and to invest in Cyrus Biotechnology, offset by proceeds from the maturities of marketable securities. The net cash used in investing activities in 2020 was to purchase property and equipment.
Financing activities
Net cash provided by financing activities for the year ended December 31, 2021 was $52.9 million compared to net cash provided by financing activities of $14.4 million in the same period in 2020. The net cash provided by financing activities in 2021 was primarily the result of net proceeds from “at-the-market” offerings and from the exercise of stock options.
The net cash provided by financing activities in 2020 was the result of $10.3 million from the Sobi Private Placement, $24.7 million from the Term A Loan, $2.1 million net proceeds from sales of common stock in “at-the-market” offerings, and $1.0 million proceeds from warrant exercises, offset by $4.4 million of issuance costs paid for December 2019 financing and $19.3 million principal payment on outstanding debt.
Research and development contract obligations
Under our license agreement with MIT, milestone payments are due upon the occurrence of certain events and royalty payments commence upon our commercialization of a product. As of December 31, 2021, contractual obligations were $0.4 million. We have assumed license payments are fully offset by royalty payments in 2028.
Recent Accounting Pronouncements
For a discussion of recently adopted or issued accounting pronouncements please see Note 2 to our consolidated financial statements included elsewhere in this Annual Report.
Off-Balance Sheet Arrangements
As of December 31, 2021, we did not have any off-balance sheet arrangements as defined in the rules and regulations of the Securities and Exchange Commission.
Critical Accounting Policies and Use of Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States, orU.S. GAAP. The preparation of these consolidated financial statements requires us to make estimates and judgmentsjudgements that affect the reported amounts of assets, liabilities, revenues and liabilitiesexpenses, and the disclosure of contingent assets and liabilities in our consolidated financial statements, as well as the reported revenues and expenses during the reporting periods. These items are monitored and analyzed by us for changes in facts and circumstances, and material changes in these estimates could occur in the future. We base our estimates on historical experience, known trends and events, and on various other factors that we believe are reasonable under
the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Changes in estimates are reflected in reported results for the period in which they become known.statements. Actual results may differ materially from these estimates under different assumptions or conditions.
Clinical Trial Costs
Clinical trial expensesconditions and could have a material impact on our reported results. While our significant accounting policies are a significant component of research and development expenses, andmore fully described in the Notes to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K, we outsource a significant portion of these costsbelieve the following accounting policies to third parties. Third party clinical trial expenses include patient costs, clinical research organization costs and costs for data management. The accrual for site and patient costs includes inputs such as estimates of patient enrollment, patient cycles incurred, clinical site activations, and other pass-through costs. Payments for these activities are based onbe the terms ofmost critical in understanding the individual arrangements, which may differ from the pattern of costs incurred, and are reflected on the consolidated balance sheets as a prepaid asset or accrued clinical trial cost. These third party agreements are generally cancelable, and related costs are recorded as research and development expenses as incurred. Non-refundable advance clinical payments for goods or services that will be used or rendered for future R&D activities are recorded as a prepaid asset and recognized as expense as the related goods are delivered or the related services are performed. We also record accruals for estimated ongoing clinical research and development costs. When evaluating the adequacy of the accrued liabilities, we analyze progress of the studies, including the phase or completion of events, invoices received and contracted costs. Significant judgments and estimates may be madewe use in determining the accrued balances at the end of any reporting period. Actual results could differ from the estimates made by the Company. The historical clinical accrual estimates made by the Company have not been materially different from the actual costs.preparing our consolidated financial statements:
Revenue Recognition
Revenue is recognized when a customer obtains control of promised goods or services, in an amount that reflects the consideration which the entity expects to receive in exchange for those goods or services. Pursuant to ASC Topic 606, Revenue from Contracts with Customers (ASC 606), a customer is a party that has contracted with an entity to obtain goods or services that are an output of the entity’s ordinary activities in exchange for consideration. To determine revenue recognition for arrangements that an entity determines are within the scope of ASC 606, we perform the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract, including whether they are distinct in the context of the contract; (iii) determine the transaction price, including the constraint on variable consideration; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy each performance obligation. We only apply the five-step model to contracts when it is probable that the entity will collect the consideration it is entitled to in exchange for the goods or services it transfers to the customer. At contract inception, once the contract is determined to be within the scope of ASC 606, we assess the goods or services promised within each contract and determinesdetermine those that are performance obligations, and assessesassess whether each promised good or service is distinct. If a promised good or service is not distinct, it is combined with other performance obligations. We then recognize as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied. For example, certain performance obligations associated with SparkSwedish Orphan Biovitrum, or Sobi, Asklepios Biopharmaceutical, Inc., or AskBio, Sarepta Therapeutics, Inc., or Sarepta, and Takeda Pharmaceuticals USA, Inc., or Takeda, (see Note 12 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K)12) will be satisfied over time, and revenue will be recognized using the output method, based on the proportion of actual deliveries to the total expected deliveries over the initial term.
Collaboration and GrantLicense Revenue: We currently generate our revenue through grants, collaboration and license agreements with strategic collaborators for the development and commercialization of product candidates. GrantsCollaboration and license agreements with customers are generally accounted for in accordance with ASC 606. We analyze collaboration arrangements by first assessing whether they are within the scope of ASC Topic 808, Collaborative Arrangements (ASC 808)808), and evaluate whether such arrangements involve joint operating activities performed by parties that are both active participants in the activities and exposed to significant risks and rewards that are dependent on the commercial success of such activities. Collaboration agreements with customers that are not within the scope of ASC 808 are accounted for in accordance with ASC 606. To the extent the collaboration agreement is within the scope of ASC 808, we also assess whether any aspects of the agreement are within the scope of other accounting literature (specifically ASC 606). We early adopted ASU No. 2018-18, Collaborative Arrangements (Topic 808): Clarifying the Interaction between Topic 808 and Topic 606, which provides guidance on evaluating certain transactions between collaborative arrangement participants. If we conclude that some or all aspects of the agreement are distinct and represent a transaction with a customer, we account for those aspects of the arrangement within the scope of ASC 606. We recognize the shared costs incurred that are not within the scope of other accounting literature as a component of the related expense in the period incurred by analogy to ASC Topic 730, Research and Development (ASC 730), and record reimbursements from counterparties as an offset to the related costs. In determining the appropriate amount of revenue to be recognized as it fulfillswe fulfill our obligations under the agreements in accordance with ASC 606, we perform the five steps above. As part of the accounting for the arrangement, we must develop assumptions that require judgment to determine the stand-alone selling price for each performance obligation identified in the contract. We use key assumptions to determine the stand-alone
selling price, which may include market conditions, reimbursement rates for personnel costs, development timelines and probabilities of regulatory success. The assumptions used to determine the stand-alone selling price and our satisfaction of performance obligations have a material effect on our collaboration and license revenue and may prove to be wrong.
The terms of our arrangements typically include one or more of the following: (i) up-frontupfront fees; (ii) milestone payments related to the achievement of development, regulatory, or commercial goals; (iii) royalties on net sales of licensed products; (iv) reimbursements or cost-sharing of R&Dresearch and development (R&D) expenses; and (v) profit/loss sharing arising from co-promotion arrangements.
Licenses of intellectual property:Intellectual Property: If the license to our intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, we recognize revenues from non-refundable, up-frontupfront fees allocated to the license when the license is transferred to the customer and the customer is able to use and benefit from the license. If not distinct, the license is combined with other performance obligations in the contract. For licenses that are combined with other performance obligations, we assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring
progress for purposes of recognizing revenue. We evaluate the measure of progress each reporting period and, if necessary, adjustsadjust the measure of performance and related revenue recognition. Optional licenses are evaluated to determine if they are issued at a discount, and therefore, represent material rights and should be accounted for as separate performance obligations.
Milestone Payments: At the inception of each arrangement that includes developmental and regulatory milestone payments, we evaluate whether the achievement of each milestone specifically relates to our efforts to satisfy a performance obligation or transfer a distinct good or service within a performance obligation. If the achievement of a milestone is considered a direct result of our efforts to satisfy a performance obligation or transfer a distinct good or service and the receipt of the payment is based upon the achievement of the milestone, the associated milestone value is allocated to that distinct good or service. If the milestone payment is not specifically related to our effortefforts to satisfy a performance obligation or transfer a distinct good or service, the amount is allocated to all performance obligations using the relative standalone selling price method. We also evaluate the milestonesmilestone to determine whether they are considered probable of being reached and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the associated milestone value is included in the transaction price to be allocated, otherwise, such amounts are constrained and excluded from the transaction price. At the end of each subsequent reporting period, we re-evaluate the probability of achievement of such development milestones and any related constraint, and if necessary, adjustsadjust our estimate of the transaction price. Any such adjustments to the transaction price are allocated to the performance obligations on the same basis as at contract inception. Amounts allocated to a satisfied performance obligation shall be recognized as revenue, or as a reduction of revenue, in the period in which the transaction price changes.
Manufacturing Supply Services: Arrangements that include a promise for future supply of drug substance or drug product for either clinical development or commercial supply at the customer’s discretion are evaluated to determine if they are distinct and optional. For optional services that are distinct, we assess if they are priced at a discount, and therefore, provide a material right to the licensee to be accounted for as separate performance obligations.
Royalties: For arrangements that include sales-based royalties, including milestone payments based on the level of sales, and the license is deemed to be the predominant item to which the royalties relate, we will recognize revenue at the later of (i) when the related sales occur, or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied (or partially satisfied) in accordance with the royalty recognition constraint.
Clinical Trial Costs
Clinical trial expenses are a significant component of research and development expenses, and we outsource a significant portion of these costs to third parties. Third party clinical trial expenses include patient costs, clinical research organization costs and costs for data management. The accrual for site and patient costs includes inputs such as estimates of patient enrollment, patient cycles incurred, clinical site activations, and other pass-through costs. Payments for these activities are based on the terms of the individual arrangements, which may differ from the pattern of costs incurred, and are reflected on the consolidated balance sheets as a prepaid asset or accrued clinical trial cost. These third party agreements are generally cancellable, and related costs are recorded as research and development expenses as incurred. Non-refundable advance clinical payments for goods or services that will be used or rendered for future R&D activities are recorded as a prepaid asset and recognized as expense as the related goods are delivered or the related services are performed. We also record accruals for estimated ongoing clinical research and development costs. When evaluating the adequacy of the accrued liabilities, we analyze progress of the studies, including the phase or completion of events, invoices received and contracted costs. Significant judgments and estimates may be made in determining the accrued balances at the end of any reporting period. Actual results could differ from the estimates made by us materially affecting our results of operations. The historical clinical accrual estimates made by us have not been materially different from the actual costs.
Warrant Liabilities
In December 2019, we issued common warrants in connection with the 2019 Purchase Agreement.a securities purchase agreement between us and a group of institutional investors and certain members of our board of directors. Pursuant to the terms of these common warrants, we could be required to settle the common warrants in cash in the event of certain acquisitions of the Companyus and, as a result, the common warrants are required to be measured at fair value and reported as a liability on the balance sheet. We recorded the fair value of the common warrants of $40.7 million upon issuance using the Black-Scholes valuation model, and are required to revalue the common warrants at each reporting date with any changes in fair value recorded on our statement of operations. Inputs used to determine estimated fair value of the common warrant liabilities include the estimated fair value of the underlying stock at the valuation date, the estimated term of the warrants, risk-free interest rates, expected dividends and the expected volatility of the underlying stock. As of December 31, 2019,The estimates used to determine the fair value of thethese common warrants of $41.5 million was recorded as a long-term liability onrepresent our balance sheet, which resulted in abest estimates, but may prove to be wrong. Therefore, the change in fair value of $0.9 million forwarrant liabilities could be materially different in the year ended December 31, 2019. Additionally, we allocated $1.2 millionfuture.
Stock-Based Compensation
Stock‑Based Compensation
We account for all stock‑basedstock-based compensation granted to employees and non‑employeesnon-employees using a fair value method. Stock‑basedStock-based compensation is measured at the grant date fair value using the Black‑Scholes option pricing model and is recognized over the requisite service period of the awards, usually the vesting period, on a straight‑linestraight-line basis, net of estimated forfeitures. We
reduce recorded stock‑based compensation for estimated forfeitures. To the extent that actual forfeitures differ from management’sour estimates, the differences are recorded as a cumulative adjustment in the period the estimates were adjusted. Stock‑basedStock-based compensation expense recognized in the consolidated financial statements is based on awards that are ultimately expected to vest.
Emerging Growth Company Status
The Jumpstart Our Business Startups Actassumptions used in determining the fair value of 2012,stock-based awards represent our best estimates, but the estimates involve inherent uncertainties and the application of our judgment. As a result, if factors change and we use significantly different assumptions or estimates, our stock-based compensation expense could be materially different in the JOBS Act, permits an ‘‘emerging growth company’’ such as us to take advantage of an extended transition period to comply with new or revised accounting standards applicable to public companies until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.future.
Smaller Reporting Company
We qualify as a “smaller reporting company” under the rules of the Securities Act and the Exchange Act. As a result, in addition to the exemptions available to us as an “emerging growth company,” we may choose to take advantage of certain scaled disclosure requirements available specifically to smaller reporting companies. Additionally, even if we cease to be an emerging growth company as noted above, as long as we continue to be a smaller reporting company, we may continue to rely on the reduced executive compensation disclosure obligations available to emerging growth companies. We will remain a smaller reporting company until the last day of the fiscal year in which the aggregate market value of our common stock held by non-affiliated persons and entities, or our public float, was lessis more than $250$700 million as of the last business day of our most recently completed second fiscal quarter, or the last day of the fiscal year in which we have at least $100 million in revenue and at least $700$250 million in public float as of the last business day of our most recently completed second fiscal quarter.
RESULTS OF OPERATIONS
Comparison of the Years Ended December 31, 2019 and 2018
Revenue
The following is a comparison of revenue for the years ended December 31, 2019 and 2018 (in thousands, except percentages):
|
| | | | | | | | | | | | | | |
| | Year Ended December 31, | | Increase |
| | 2019 | | 2018 | | (decrease) |
| Grant revenue | $ | — |
| | $ | 903 |
| | $ | (903 | ) | | — | % |
| Collaboration revenue | 6,677 |
| | — |
| | 6,677 |
| | — | % |
| Total revenue | $ | 6,677 |
| | $ | 903 |
| | $ | 5,774 |
| | 639 | % |
During the year ended December 31, 2019, we recognized $6.7 million in revenue upon expiration of the term for Spark to exercise additional target options that represented material rights and less than $0.1 million of revenue for two shipments to Spark under our collaboration agreement. During the year ended December 31, 2018, we recognized the remaining $0.9 million in grant revenues from NIDA after receiving final approval from NIDA.
Research and development
The following is a comparison of research and development expenses for the years ended December 31, 2019 and 2018 (in thousands, except percentages): |
| | | | | | | | | | | | | | |
| | Year Ended December 31, | | Increase |
| | 2019 | | 2018 | | (decrease) |
| Research and development | $ | 42,743 |
| | $ | 47,687 |
| | $ | (4,944 | ) | | (10 | )% |
During the year ended December 31, 2019, our research and development expenses decreased by $5.0 million, or 10%, as compared to 2018. The decrease reflects reduced costs in 2019 resulting from the completion of prior programs in 2018 combined with reduced salaries and benefits resulting from the headcount reduction in early 2019. The cost reductions were offset by an overall increase in costs incurred on our lead product candidate, SEL-212.
General and administrative
The following is a comparison of general and administrative expenses for the years ended December 31, 2019 and 2018 (in thousands, except percentages): |
| | | | | | | | | | | | | | |
| | Year Ended December 31, | | Increase |
| | 2019 | | 2018 | | (decrease) |
| General and administrative | $ | 16,389 |
| | $ | 18,238 |
| | $ | (1,849 | ) | | (10 | )% |
During the year ended December 31, 2019, our general and administrative expenses decreased by $1.8 million, or 10%, as compared to 2018. The decrease is the result of lower salary and stock compensation expense resulting from a reduction in headcount at the end of 2018, and lower patent and professional fees.
Investment income
Investment income remained relatively unchanged during the years ended December 31, 2019 as compared to 2018.
Foreign currency transaction gain (loss)
We recognized minimal foreign currency losses of less than $0.1 million and gains of $0.1 million during the year ended December 31, 2019 and 2018, respectively.
Interest expense
Interest expense was $1.5 million for each of the years ended December 31, 2019 and 2018, representing interest expense and amortization of the carrying costs of our credit facilities.
Change in fair value of warrant liabilities
We recognized $0.9 million as a change in the fair value of warrant liabilities utilizing a Black-Scholes valuation methodology, for the year ended December 31, 2019, primarily driven by an increase in the share price from issuance (see Note 5).
Other income (expense)
Other (expense) income was $1.3 million and less than $0.1 million for the years ended December 31, 2019 and 2018, respectively. The increase in the other expense during 2019 was the result of $1.2 million of fees incurred as a result of the issuance of liability classified common warrants in our December 2019 private placement (see Note 10).
Net Loss
Net loss for the year ended December 31, 2019 was $55.4 million compared to $65.3 million for the year ended December 31, 2018.
LIQUIDITY AND CAPITAL RESOURCES
Since our inception, we have incurred recurring net losses. We expect that we will continue to incur losses and that such losses will increase for the foreseeable future. We expect that our research and development and general and administrative expenses will continue to increase and, as a result, we will need additional capital to fund our operations, which we may raise through a combination of equity offerings, debt financings, third‑party funding and other collaborations and strategic alliances.
From our inception through December 31, 2019, we have raised an aggregate of $416.0 million to fund our operations, which includes $118.5 million from the sale of preferred stock, $11.1 million in government grant funding, $25.3 million from borrowings under our credit facility, $46.3 million from our collaborations and license agreements, $64.5 million in combined net proceeds from our initial public offering $118.4 million in combined net proceeds from private placements of our common stock in June 2017 and August and December 2019, $30.9 million from an underwritten follow-on offering of our common stock in January 2019, and $1.0 million in aggregate net proceeds from "at-the-market" offerings of our common stock in 2019.
Collaborations
On December 17, 2019, we entered into the AskBio License Agreement. Pursuant to the AskBio License Agreement, AskBio has exercised its option to exclusively license the Company’s intellectual property rights covering ImmTOR to research, develop, and commercialize certain AAV genetherapy products utilizing ImmTOR, and targeting the GAA gene, or derivatives thereof, to treat Pompe Disease. As of December 31, 2019, AskBio paid $2.0 million of the aggregate $7.0 million of upfront fees.
Financings
On December 23, 2019, we sold an aggregate of 37,634,883 shares of our common stock at a purchase price of $1.46 per share, warrants to purchase an aggregate of 22,988,501 shares of common stock at a purchase price of $0.125 per share underlying
each common warrant, and pre-funded warrants to purchase an aggregate of 8,342,128 shares of common stock at a purchase price of $1.46 per share for net proceeds of $65.6 million, after deducting commissions and other transaction costs.
On August 19, 2019, we sold 3,178,174 shares of its common stock pursuant to a Stock Purchase Agreement to individual investors, including certain of our executive officers and members of our board of directors for aggregate net proceeds of approximately $5.7 million, after deducting transaction costs.
On January 25, 2019, we completed a public offering of 20,000,000 shares of our common stock at a public offering price of $1.50 per share. On January 29, 2019, an additional 2,188,706 shares were sold at a public offering price of $1.50 per share. The total net proceeds from the offering were $30.9 million, after deducting underwriting discounts and commissions.
In August 2017, we entered into a sales agreement, or the Sales Agreement, with Jefferies LLC, as sales agent, pursuant to which we may, from time to time, issue and sell common stock with an aggregate value of up to $50 million in an "at-the-market" offering. Sales of common stock, if any, pursuant to the Sales Agreement, may be made in sales deemed to be an “at the market offering” as defined in Rule 415(a) of the Securities Act, including sales made directly through the Nasdaq Global Market or on any other existing trading market for our common stock. We intend to use the proceeds from the offering for working capital and other general corporate purposes. We may suspend or terminate the Sales Agreement at any time. During the year ended December 31, 2019, we sold 615,453 shares of our common stock pursuant to the Sales Agreement at an average price of approximately $1.84 per share for aggregate net proceeds of $1.0 million, after deducting commissions and other transaction costs.
As of December 31, 2019, our cash, cash equivalents, and restricted cash were $91.6 million, of which $1.7 million was restricted cash related to lease commitments and $0.4 million was held by our Russian subsidiary designated solely for use in its operations. Our Russian subsidiary cash is consolidated for financial reporting purposes.
In addition to our existing cash equivalents, we receive research and development funding pursuant to our research grants and collaboration agreements. Currently, funding from research grants and payments under our collaboration agreements represent our only source of committed external funds.
Indebtedness
On September 12, 2017, we entered into a term loan facility of up to $21.0 million with Silicon Valley Bank, a California corporation, or SVB, the proceeds of which were used to repay our previously existing term loan facility with Oxford Finance LLC and Pacific Western Bank, as successor in interest to Square 1 Bank, and for general corporate and working capital purposes. The term loan facility is governed by a loan and security agreement, dated September 12, 2017, between us and SVB, which was funded in full on September 13, 2017. The term loan facility with SVB is secured by a lien on substantially all assets, other than intellectual property, provided that such lien on assets includes any rights to payments and proceeds from the sale, licensing or disposition of intellectual property. We also granted SVB a negative pledge with respect to our intellectual property.
The term loan facility contains customary covenants and representations, including but not limited to financial reporting obligations and limitations on dividends, indebtedness, collateral, investments, distributions, transfers, mergers or acquisitions, taxes, corporate changes, deposit accounts, and subsidiaries. The term loan facility also contains other customary provisions, such as expense reimbursement, non-disclosure obligations as well as indemnification rights for the benefit of SVB.
The events of default under the term loan facility include, but are not limited to, our failure to make any payments of principal or interest under the term loan facility or other transaction documents, our breach or default in the performance of any covenant under the term loan facility or other transaction documents, the occurrence of a material adverse effect, making a false or misleading representation or warranty in any material respect under the term loan facility, our insolvency or bankruptcy, any attachment or judgment on our assets in excess of approximately $0.3 million, or the occurrence of any default under any of our agreements or obligations involving indebtedness in excess of approximately $0.3 million. If an event of default occurs, SVB is entitled to take enforcement action, including acceleration of amounts due under the term loan facility. If we raise any additional debt financing, the terms of such additional debt could further restrict our operating and financial flexibility.
Plan of operations and future funding requirements
As of the date of this Annual Report on Form 10-K, we have not generated any product sales. We do not know when, or if, we will generate revenue from product sales. We will not generate significant revenue from product sales unless and until we obtain regulatory approval and commercialize one of our current or future product candidates. Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, third-party clinical research and development services, laboratory and related supplies, clinical costs, legal and other regulatory expenses, and general overhead costs. We expect that we will continue to generate losses for the foreseeable future, and we expect the losses to increase as we continue the development of, and seek regulatory approvals for, our product candidates, and begin to commercialize any approved products. We are subject to risks in the development of our products, and we may encounter unforeseen expenses, difficulties,
complications, delays and other unknown factors that may adversely affect our business. We expect that we will need substantial additional funding to support our continuing operations.
As of December 31, 2019 and December 31, 2018, we had an accumulated deficit of $335.8 million and $280.4 million, respectively. We anticipate operating losses to continue for the foreseeable future due to, among other things, costs related to research, development of our product candidates, conducting preclinical studies and clinical trials, and our administrative organization. We will require substantial additional financing to fund our operations and to continue to execute our strategy, and we will pursue a range of options to secure additional capital.
Management is exploring various sources of funding such as strategic collaborations and the issuance of equity to fund our operations. If we raise additional funds through strategic collaborations and alliances, which may include existing collaboration partners, we may have to relinquish valuable rights to our technologies or product candidates, or grant licenses on terms that are not favorable to us. To the extent that we raise additional capital through the sale of equity, the ownership interest of our existing shareholders will be diluted and other preferences may be necessary that adversely affect the rights of existing shareholders.
We will require additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212. Under the terms of our exclusive patent license agreement with the Massachusetts Institute of Technology, or the MIT License, MIT may terminate the MIT License if we fail to meet a diligence obligation, including the initiation of a Phase 3 clinical trial by a specified date in the fourth quarter of 2019. On December 13, 2019, we entered into the Fourth Amendment, which we refer to as the MIT Amendment, to the Exclusive Patent License Agreement by and between us and the Massachusetts Institute of Technology, or the MIT Agreement. Pursuant to the MIT Amendment, a provision of the MIT Agreement under which we were obligated to initiate a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT Agreement. Further, pursuant to the MIT Amendment, the parties agreed to negotiate in good faith to enter into a future amendment to the MIT Agreement after we provide MIT with an amended diligence plan. If we are unable to reach an agreement with MIT regarding an acceptable amendment of the MIT License and if we are unable to cure the breach, there could be a material adverse effect on our business.
We believe that our existing cash, cash equivalents, investments, and restricted cash as of December 31, 2019 will enable us to fund our operating expenses and capital expenditure requirements into the first quarter of 2021. We plan to commence our Phase 3 clinical program in SEL-212 in the second half of 2020. Because our current operating plan does not contain sufficient resources, we will require additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212. We have based this estimate on assumptions that may prove to be wrong, and we could use our capital resources sooner than we currently expect. Because of the uncertainty in securing additional capital, we have concluded that substantial doubt exists with respect to our ability to continue as a going concern within one year after the date of the filing of this Annual Report on Form 10-K.
Our future capital requirements will depend on many factors, including:
| |
- | the scope, progress, results and costs of our clinical trials of SEL-212; |
| |
- | the number of product candidates that we pursue; |
| |
- | our collaboration agreements remaining in effect, our entering into additional collaboration agreements and our ability to achieve milestones under these agreements; |
| |
- | the cost of manufacturing clinical supplies of our product candidates; |
| |
- | our headcount growth and associated costs; |
| |
- | the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials for our other product candidates; |
| |
- | the costs, timing and outcome of regulatory review of our product candidates; |
| |
- | the costs and timing of future commercialization activities, including manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval; |
| |
- | the revenue, if any, received from commercial sales of our product candidates for which we receive marketing approval; |
| |
- | the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property-related claims; |
| |
- | the effect of competing technological and market developments; and |
| |
- | the extent to which we acquire or invest in businesses, products and technologies, including entering into licensing or collaboration arrangements for product candidates. |
Summary of Cash Flows
|
| | | | | | | | | | | |
| | Year Ended December 31, |
| (In thousands) | 2019 | | 2018 | | 2017 |
| Cash provided by (used in): | | | | | |
| Operating activities | $ | (51,435 | ) | | $ | (59,161 | ) | | $ | (52,026 | ) |
| Investing activities | 229 |
| | 25,272 |
| | (2,098 | ) |
| Financing activities | 105,041 |
| | 697 |
| | 66,023 |
|
| Effect of exchange rate changes on cash | 34 |
| | (153 | ) | | 78 |
|
| Net change in cash, cash equivalents, and restricted cash | $ | 53,869 |
| | $ | (33,345 | ) | | $ | 11,977 |
|
Operating activities
Net cash used in operating activities for the year ended December 31, 2019 was $51.4 million compared to $59.2 million in the same period in 2018, a decrease of $7.8 million. The decrease in net cash used in operating activities was primarily due to a $10.0 million decrease in recorded net loss, a decrease of $5.8 million in prepaid expenses, and a decrease of $1.4 million resulting from non-cash charges which were offset by an increase of $4.9 million in accrued expenses and other liabilities, and an increase of $5.0 million in accounts receivable. The change in deferred revenue is due to the receipt of $2.0 million and a receivable of $5.0 million in proceeds from AskBio offset by $6.7 million from the expiration of the term for Spark to exercise additional target options.
Investing activities
Net cash provided by investing activities for the year ended December 31, 2019 was $0.2 million compared to net cash provided from investing activities of $25.3 million in the same period in 2018. The net cash provided by investing activities in 2019 was the result of purchases of short-term investments of $18.2 million, offset by $16.4 million of maturities and $2.0 million of sales of short term investments.
The net cash provided by investing activities in 2018 was primarily due to receipts of $41.7 million from maturities of short-term investments, offset by $15.6 million of purchases of short-term investments.
Financing activities
Net cash provided by financing activities for the year ended December 31, 2019 was $105.0 million compared to $0.7 million in the same period in 2018. The net cash provided by financing activities in 2019 was the result of $30.9 million net proceeds from an underwritten follow-on offering of our common stock in January, $75.7 million net proceeds from private placement offerings in August and December and $1.0 million net proceeds from "at-the-market" offerings, offset by $2.8 million principal payment on outstanding debt.
The net cash provided by financing activities in 2018 was due to $0.2 million from the issuance of common stock under the ESPP and $0.5 million from the exercise of employee stock options.
Contractual Obligations
The following summarizes our principal contractual obligations as of December 31, 2019:
|
| | | | | | | | | | | | | | | | | | | | | | | |
| | Total | | 2020 | | 2021 | | 2022 | | 2023 | | Later |
| Operating leases obligations (1) | $ | 16,169 |
| | $ | 1,566 |
| | $ | 1,811 |
| | $ | 1,865 |
| | $ | 1,921 |
| | $ | 9,006 |
|
| Research and development contract obligations (2) | 180 |
| | 60 |
| | 60 |
| | 60 |
| | — |
| | — |
|
| Debt obligations (3) | 20,261 |
| | 9,084 |
| | 8,716 |
| | 2,461 |
| | — |
| | — |
|
| Total contractual obligations | $ | 36,610 |
| | $ | 10,710 |
| | $ | 10,587 |
| | $ | 4,386 |
| | $ | 1,921 |
| | $ | 9,006 |
|
(1) Operating lease obligations represent future minimum lease payments under noncancellable property leases in Watertown, Massachusetts. The minimum lease payments do not include common area maintenance charges, real estate taxes or any sublease income we may earn.
(2) Research and development contract obligations represent minimum annual license fees payable to universities or partners under our license agreements. Under our license agreement with the Massachusetts Institute of Technology, or MIT, milestone payments are due upon the occurrence of certain events and
royalty payments commence upon our commercialization of a product. For purposes of presenting our contractual obligations under the MIT agreement, we have assumed license payments are fully offset by royalty payments in 2023.
(3) Debt obligations payments are based on the contractual payment dates.
Recent Accounting Pronouncements
For a discussion of recently adopted or issued accounting pronouncements please refer to Part IV, Note 2 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
Off-Balance Sheet Arrangements
As of December 31, 2019, we did not have any off-balance sheet arrangements as defined in the rules and regulations of the Securities and Exchange Commission.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
The market risk inherent in our financial instruments and in our financial position represents the potential loss arising from adverse changes in interest rates. As of December 31, 20192021 and December 31, 2018,2020, we had cash, cash equivalents, restricted cash and investmentsmarketable securities of $91.6$129.4 million and $37.7$140.1 million, respectively, consisting of non‑interestnon-interest and interest‑bearinginterest-bearing money market accounts. Our primary exposure to market risk is interest rate sensitivity, which is affected by changes in the general level of U.S. interest rates. Due to the short‑termshort-term and the low risk profile of our money market accounts and investments,marketable securities, and our current plan to hold investmentsmarketable securities to maturity, an immediate 100 basis point change in interest rates would not have a material effect on the fair market value of our cash equivalents or short-term investments.marketable securities.
In addition, we are subject to currency risk for balances held in Russian rubles in our foreign subsidiary. We hold portions of our funds in both U.S. dollars and Russian rubles. The exchange rate between the U.S. dollar and Russian ruble changes from period to period. As of December 31, 2019, we held cash and cash equivalents totaling $0.4 million in Russian banks to support our Russian subsidiary, all of which were denominated in U.S. dollars. We do not hedge against foreign currency risks. We do not believe that inflation and changing prices had a significant impact on our results of operations for any periods presented herein.
Item 8. Financial Statements and Supplementary Data
The consolidated financial statements together with the report of our independent registered public company accounting firm, required to be filed pursuant to this Item 8 are appended to this Annual Report on Form 10-K.Report. An index of those consolidated financial statements is found in Item 15 of this Annual Report on Form 10-K.15.
Item 9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Limitations on effectiveness of controls and procedures
In designing and evaluating our disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints and that management is required to apply judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Evaluation of disclosure controlsDisclosure Controls and proceduresProcedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated, as of the end of the period covered by this Annual Report on Form 10-K, the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”))Act). Based on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of December 31, 2019.2021.
Management’s annual reportAnnual Report on internal controlInternal Control over financial reporting
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting, as such term is defined in Rule 13a-15(f) under the Exchange Act.
Our management conducted an assessment of the effectiveness of our internal control over financial reporting based on the criteria set forth in “Internal Control - Integrated Framework (2013)” issued by the Committee of Sponsoring Organizations of the Treadway Commission.
Based on this assessment, our management concluded that, as of December 31, 2019,2021, our internal control over financial reporting was effective.
Inherent Limitations of Internal Controls
While we believe we have a robust and efficient system of internal and disclosure controls and procedures, our management, including our Chief Executive Officer and Chief Financial Officer, recognize that it is impossible for our disclosure controls and procedures or our internal controls to prevent all errors and all fraud. A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all
control issues and instances of fraud, if any, within our company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of a simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the control. The design of any system of controls also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Over time, controls may become inadequate because of changes in conditions, or the degree of compliance with the policies or procedures may deteriorate. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Changes in Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act during the three months ended December 31, 2021 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Attestation Report of the Registered Public Accounting Firm
This Annual Report on Form 10-K does not include an attestation report of our registered public accounting firm due to an exemption established by the JOBS Act for "emerging growth“smaller reporting companies."”
Changes in internal control over financial reporting
There were no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the three months ended December 31, 2019 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information.Information
None.
PART III
Item 10. Directors, Executive Officers and Corporate Governance
Our board of directors has adopted a written Code of Business Conduct and Ethics applicable to all officers, directors and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. We have posted a current copy of our Code of Business Conduct and Ethics on our website at www.selectabio.com in the “Investors & Media” section under “Corporate Governance.” We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding amendment to, or waiver from, a provision of our Code of Business Conduct and Ethics, as well as Nasdaq’s requirement to disclose waivers with respect to directors and executive officers, by posting such information on our website at the address and location specified above. The information contained on our website is not incorporatedIncorporated by reference intofrom the information in our Proxy Statement for our 2022 Annual Meeting of Stockholders, which we will file with the SEC within 120 days of the end of the fiscal year to which this Annual Report on Form 10-K.10-K relates.
The information required by this Item is contained in part under the caption “Information about our Executive Officers” at the end of Part I of this Annual Report on Form 10-K. The remainder of the response to this item will be included in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders under the headings “Proposal 1-Election of Directors,” “Delinquent Section 16(a) Reports,” and “Committees of the Board” and is incorporated herein by reference.
Item 11. Executive Compensation
TheIncorporated by reference from the information required by this Item regarding executive compensation will be included in our definitive Proxy Statement for the 2020our 2022 Annual Meeting of Stockholders, underwhich we will file with the headings “Executive and Director Compensation” and “Compensation Committee Interlocks and Insider Participation” and is incorporated herein by reference, as applicable.SEC within 120 days of the end of the fiscal year to which this Annual Report on Form 10-K relates.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
TheIncorporated by reference from the information required by this Item will be included in our definitive Proxy Statement for the 2020our 2022 Annual Meeting of Stockholders, underwhich we will file with the headings “Security OwnershipSEC within 120 days of Certain Beneficial Owners and Management” and “Executive and Director Compensation-Equity Compensation Plan Information” and is incorporated herein by reference.the end of the fiscal year to which this Annual Report on Form 10-K relates.
Item 13. Certain Relationships and Related Transactions, and Director Independence
TheIncorporated by reference from the information required by this Item will be included in our definitive Proxy Statement for the 2020our 2022 Annual Meeting of Stockholders, underwhich we will file with the headings “Corporate Governance,” “CommitteesSEC within 120 days of the Board” and “Certain Relationships” and is incorporated herein by reference.end of the fiscal year to which this Annual Report on Form 10-K relates.
Item 14. Principal Accountant Fees and Services
TheIncorporated by reference from the information required by this Item regarding principal accountant fees and services will be included in our definitive Proxy Statement for the 2020our 2022 Annual Meeting of Stockholders, underwhich we will file with the heading “Independent Registered Public Accounting Firm Fees and Other Matters” and is incorporated herein by reference.
SEC within 120 days of the end of the fiscal year to which this Annual Report on Form 10-K relates.
PART IV
Item 15. Exhibits, Financial Statement Schedules
(a)(1) Financial Statements
TheSee the “Index to Consolidated Financial Statements” on page F-1 below for the list of financial statements listed below are filed as part of this Annual Report on Form 10-K.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
(a)(2) Financial Statement Schedules
All financial schedules have been omitted because the required information is either presented in the consolidated financial statements filed as part of this Annual Report on Form 10-K or the notes thereto or is not applicable or required.
(a)(3) Exhibits
The following is a list of exhibits filed as part of this Annual Report on Form 10-K.
|
| | | | | | | | | | | | |
| | | | | Incorporated by Reference | | |
Exhibit Number | | Exhibit Description | | Form | | File No. | | Exhibit | | Filing Date | | Filed Herewith |
| | | | 8-K | | 001-37798 | | 3.1 | | 6/29/2016 | | |
| | | | 8-K | | 001-37798 | | 3.2 | | 6/29/2016 | | |
| | | | S-1/A | | 333-211555 | | 4.1 | | 6/8/2016 | | |
| | | | S-1 | | 333-211555 | | 4.2 | | 5/24/2016 | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Incorporated by Reference |
Exhibit
Number | | Exhibit Description | | Form | | File No. | | Exhibit | | Filing
Date |
| | | | 8-K | | 001-37798 | | 3.1 | | 6/29/2016 |
| | | | 8-K | | 001-37798 | | 3.2 | | 9/30/2021 |
| | | | S-1 | | 333-211555 | | 4.2 | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 4.5 | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 4.6 | | 5/24/2016 |
| | | | 8-K | | 001-37798 | | 4.1 | | 6/28/2017 |
| | | | 8-K | | 001-37798 | | 10.2 | | 12/26/2019 |
| | | | 10-Q | | 001-37798 | | 4.1 | | 8/6/2020 |
| | | | 10-Q | | 001-37798 | | 4.2 | | 11/5/2020 |
| | | | 8-K | | 001-37798 | | 4.1 | | 12/26/2019 |
| | | | 8-K | | 001-37798 | | 4.1 | | 9/3/2020 |
|
| | | | | | | | | | | | |
| | | | S-1 | | 333-211555 | | 4.5 | | 5/24/2016 | | |
| | | | S-1 | | 333-211555 | | 4.6 | | 5/24/2016 | | |
| | | | 8-K | | 001-37798 | | 4.1 | | 6/28/2017 | | |
| | | | 8-K | | 001-37798 | | 10.3 | | 6/28/2017 | | |
| | | | 8-K | | 001-37798 | | 10.2 | | 12/26/2019 | | |
| | | | 8-K | | 001-37798 | | 4.1 | | 12/26/2019 | | |
| | | | 8-K | | 001-37798 | | 4.2 | | 12/26/2019 | | |
| | | | | | | | | | | | * |
| | | | S-1/A | | 333-211555 | | 10.2 | | 6/8/2016 | | |
| | | | S-1/A | | 333-211555 | | 10.3 | | 6/8/2016 | | |
| | | | S-8 | | 333-230501 | | 10.1 | | 3/25/2019 | | |
| | | | S-1/A | | 333-211555 | | 10.1 | | 6/20/2016 | | |
| | | | 10-Q | | 001-37798 | | 10.3 | | 8/8/2018 | | |
| | | | S-1 | | 333-211555 | | 10.5 | | 5/24/2016 | | |
| | | | S-1 | | 333-211555 | | 10.7(a) | | 5/24/2016 | | |
| | | | S-1 | | 333-211555 | | 10.7(b) | | 5/24/2016 | | |
| | | | S-1 | | 333-211555 | | 10.7(c) | | 5/24/2016 | | |
|
| | | | | | | | | | | | |
| | | | S-1 | | 333-211555 | | 10.7(d) | | 5/24/2016 | | |
| | | | S-1 | | 333-211555 | | 10.7(e) | | 5/24/2016 | | |
| | | | 8-K/A | | 001-37798 | | 10.3(a) | | 12/14/2016 | | |
| | | | 8-K/A | | 001-37798 | | 10.3(b) | | 12/14/2016 | | |
| | | | 8-K/A | | 001-37798 | | 10.3(c) | | 12/14/2016 | | |
| | | | | | | | | | | | * |
| | | | 10-Q | | 001-37798 | | 10.6 | | 8/11/2017 | | |
| | | | S-1 | | 333-211555 | | 10.10 | | 5/24/2016 | | |
| | Lease, dated as of September 30, 2008, as amended by the First Amendment, dated as of July 12, 2011, the Second Amendment, dated as of October 11, 2011 and the Third Amendment, dated as of April 6, 2015, by and between the Registrant and ARE-480 Arsenal Street, LLC | | S-1 | | 333-211555 | | 10.13 | | 5/24/2016 | | |
| | | | 8-K | | 001-37798 | | 10.1 | | 9/14/2016 | | |
| | | | 10-Q | | 001-37798 | | 10.3 | | 11/8/2019 | | |
| | | | 8-K | | 001-37798 | | 10.2 | | 9/27/2018 | | |
| | | | S-1/A | | 333-211555 | | 10.18 | | 6/8/2016 | | |
|
| | | | | | | | | | | | |
| | | | S-1/A | | 333-211555 | | 10.21 | | 6/8/2016 | | |
| | | | 10-Q | | 001-37798 | | 10.1 | | 11/8/2019 | | |
| | | | 10-Q | | 001-37798 | | 10.2 | | 8/8/2019 | | |
| | | | 10-Q | | 001-37798 | | 10.2 | | 5/8/2019 | | |
| | | | 10-Q | | 001-37798 | | 10.3 | | 11/7/2017 | | |
| | | | 8-K/A | | 001-37798 | | 10.1 | | 2/14/2017 | | |
| | | | 8-K/A | | 001-37798 | | 10.2 | | 12/14/2016 | | |
| | | | 10-Q | | 001-37798 | | 10.7 | | 8/11/2017 | | |
| | | | 10-Q | | 001-37798 | | 10.2 | | 11/8/2019 | | |
| |
| | 8-K | | 001-37798 | | 10.2 | | 6/28/2017 | | |
| | | | 8-K | | 001-37798 | | 10.1 | | 8/20/2019 | | |
| | | | 8-K | | 001-37798 | | 10.1 | | 12/26/2019 | | |
| | | | 8-K | | 001-37798 | | 10.1 | | 9/13/2017 | | |
| | | | S-1 | | 333-211555 | | 21.1 | | 5/24/2016 | | |
| | | | | | | | | | | | * |
| | | | | | | | | | | | * |
|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | — | | — | | — | | | | *Filed herewith |
| | | | S-1/A | | 333-211555 | | 10.2 | | | | **6/8/2016 |
| | | | S-1/A | | 333-211555 | | 10.3 | | | | **6/8/2016 |
101.INS | | Inline XBRL Instance Document - the Instance Document does not appear in the interactive data file because its XBRL tags are embedded within the Inline XBRL Document | | S-8 | | 333-230501 | | 10.1 | | | | ***3/25/2019 |
101.SCH | | | | S-1/A | | 333-211555 | | 10.1 | | 6/20/2016 |
| | | | 10-Q | | 001-37798 | | 10.2 | | 11/10/2021 |
| | | | S-1 | | 333-211555 | | 10.5 | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 10.7(a) | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 10.7(b) | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 10.7(c) | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 10.7(d) | | 5/24/2016 |
| | | | S-1 | | 333-211555 | | 10.7(e) | | 5/24/2016 |
| | | | 8-K/A | | 001-37798 | | 10.3(a) | | 12/14/2016 |
| | | | 8-K/A | | 001-37798 | | 10.3(b) | | 12/14/2016 |
| | | | 8-K/A | | 001-37798 | | 10.3(c) | | 12/14/2016 |
| | | | 10-K | | 001-37798 | | 10.7(i) | | 3/12/2020 |
| | | | 10-Q | | 001-37798 | | 10.1 | | 8/6/2020 |
| | | | 10-Q | | 001-37798 | | 10.6 | | 8/11/2017 |
| | | | S-1 | | 333-211555 | | 10.10 | | 5/24/2016 |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | 10-Q | | 001-37798 | | 10.3 | | 11/8/2019 |
| | | | 8-K | | 001-37798 | | 10.2 | | 9/27/2018 |
| | | | S-1/A | | 333-211555 | | 10.18 | | 6/8/2016 |
| | | | S-1/A | | 333-211555 | | 10.21 | | 6/8/2016 |
| | | | 10-Q | | 001-37798 | | 10.1 | | 11/5/2020 |
| | | | 10-Q | | 001-37798 | | 10.1 | | 11/10/2021 |
| | | | 8-K/A | | 001-37798 | | 10.2 | | 12/14/2016 |
| | | | 10-Q | | 001-37798 | | 10.2 | | 11/8/2019 |
| | | | 10-Q | | 001-37798 | | 10.2 | | 8/6/2020 |
| | | | 8-K | | 001-37798 | | 10.2 | | 6/28/2017 |
| | | | 8-K | | 001-37798 | | 10.1 | | 8/20/2019 |
| | | | 8-K | | 001-37798 | | 10.1.1 | | 9/3/2020 |
| | | | 10-Q | | 001-37798 | | 10.3 | | 11/9/2021 |
| | | | S-1 | | 333-211555 | | 21.1 | | 5/24/2016 |
| | | | — | | — | | — | | Filed herewith |
| | | | — | | — | | — | | Filed herewith |
| | | | — | | — | | — | | Filed herewith |
| | | | — | | — | | — | | Furnished herewith |
| 101.INS | | Inline XBRL Instance Document - the Instance Document does not appear in the interactive data file because its XBRL tags are embedded within the Inline XBRL Document | | — | | — | | — | | Filed herewith |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document | | — | | — | | — | | | | ***Filed herewith |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document | | — | | — | | — | | | | ***Filed herewith |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase Document | | — | | — | | — | | | | ***Filed herewith |
| 101.LAB | | Inline XBRL Taxonomy Extension Label Linkbase Document | | — | | — | | — | | | | ***Filed herewith |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document | | — | | — | | — | | | | ***Filed herewith |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | | — | | — | | — | | | | *** |
| Filed herewith |
| | |
| * | Filed herewith. |
| ** | Furnished herewith. |
| *** | Submitted electronically herewith. |
| # | Indicates management contract or compensatory plan. | | | | | | | | |
| | † | Portions of this exhibit (indicated by asterisks) have been omitted pursuant to a request for confidential treatment pursuant to Rule 24h-2 under the Securities Exchange Act of 1934. |
Item 16. Form 10-K Summary
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
| | | | | | | | | | |
| | SELECTA BIOSCIENCES, INC. |
| | | |
Date: March 12, 202010, 2022 | | By: | /s/ Carsten Brunn, Ph.D. |
| | | Carsten Brunn, Ph.D. |
| | | President and Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed by the following persons on behalf of the registrant in the capacities and on the dates indicated.
| | | | | | | | | | | | | | |
| Signature | | Title | | Date |
| | | |
Signature | | Title | Date |
| | | |
| /s/ Carsten Brunn, Ph.D. | | President and Chief Executive Officer, and Director | | March 12, 202010, 2022 |
| Carsten Brunn, Ph.D. | | (Principal Executive Officer) | | |
| | | | |
/s/ Bradford D. DahmsKevin Tan | | Chief Financial Officer | | March 12, 202010, 2022 |
Bradford D. DahmsKevin Tan | | (Principal Financial and Accounting Officer) | | |
| | | | |
| /s/ Carrie S. Cox | | Director | | March 12, 202010, 2022 |
| Carrie S. Cox | | | | |
| | | | |
| /s/ Göran A. Ando, M.D. | | Director | | March 10, 2022 |
| Göran A. Ando, M.D. | | | | |
| | | | |
| /s/ Timothy C. Barabe | | Director | | March 12, 202010, 2022 |
| Timothy C. Barabe | | | | |
| | | | |
| /s/ Nishan de Silva, M.D. | | Director | | March 10, 2022 |
| Nishan de Silva, M.D. | | | | |
| | | | |
| /s/ Scott D. Myers | | Director | | March 12, 202010, 2022 |
| Scott D. Myers | | | | |
| | | | |
/s/ Amir Nashat, Ph.D | | Director | March 12, 2020 |
Amir Nashat, Ph.D | | | |
| | | |
| /s/ Aymeric Sallin | | Director | | March 12, 202010, 2022 |
| Aymeric Sallin | | | | |
| | | | |
| /s/ Timothy Springer, Ph.D. | | Director | | March 12, 202010, 2022 |
| Timothy Springer, Ph.D. | | | | |
| | | | |
| /s/ Patrick Zenner | | Director | | March 12, 202010, 2022 |
| Patrick Zenner | | | | |
Selecta Biosciences, Inc. and Subsidiaries
| | | | | | | | | | | |
| Index to Consolidated Financial Statements | Pages | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Selecta Biosciences, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Selecta Biosciences, Inc. and subsidiaries (the Company) as of December 31, 20192021 and 2018,2020, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity (deficit), and cash flows for each of the three years in the period ended December 31, 2019,2021, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 20192021 and 2018,2020, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2019,2021, in conformity with U.S. generally accepted accounting principles.
The Company's Ability to Continue as a Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has recurring losses from operations and insufficient cash resources and has stated that substantial doubt exists about the Company’s ability to continue as a going concern. Management's evaluation of the events and conditions and management’s plans regarding these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Adoption of ASU No. 2014-09
As discussed in Note 12 to the consolidated financial statements, the Company changed its method of accounting for revenue in 2018 due to the adoption of ASU No. 2014-09, Revenue from Contracts with Customers (Topic 606) and the related amendments.
Adoption of ASU No. 2016-02
As discussed in Note 8 to the consolidated financial statements, the Company changed its method of accounting for leases in 2019 due to the adoption of ASU No. 2016-02, Leases (Topic 842) and the related amendments.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’sCompany's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
| | | | | | | | |
| | Revenue Recognition for License and Development Agreement with Swedish Orphan Biovitrum (“Sobi License Agreement”) |
| Description of the Matter | | As discussed in Note 12 to the consolidated financial statements, the Company recognized $83.5 million in revenue under the Sobi License Agreement during the year ended December 31, 2021. The Company recognizes revenue for the Combined License Obligation using the output method, based on the proportion of cumulative supply shipped for use in the clinical trials to the Company’s estimate of the total supply required during the clinical trial period.
Auditing management’s calculation of revenue recognized for the Combined License Obligation under the output method is especially challenging because the assessment of the proportion of cumulative supply shipped required a high degree of audit judgment due to the subjectivity in estimating the remaining supply necessary to satisfy the Combined License Obligation. |
| | | | | | | | |
| | |
| How We Addressed the Matter in Our Audit | | To audit the Company’s revenue recognition for the Combined License Obligation, we performed audit procedures that included, among others, testing the reasonableness of the Company’s estimate of the total supply required during the clinical trial period as well as testing the accuracy and completeness of the underlying data used in those estimates. We corroborated management estimates and judgments by reviewing the clinical plans and evaluating the accuracy of the prior period estimates and judgments. We also discussed the estimate of the total supply required during the clinical trial period with the Company’s research and development personnel that oversee the activity related to the Sobi License Agreement. Additionally, we performed an independent sensitivity analysis to evaluate the impact on revenues of changes in management’s estimate of remaining supply required to satisfy the Combined License Obligation. |
/s/ Ernst & Young LLP
We have served as the Company'sCompany’s auditor since 2009.
Boston, Massachusetts
March 12, 2020
Selecta Biosciences, Inc. and Subsidiaries
Consolidated Balance Sheets
(Amounts in thousands, except share data and par value)
| | | | December 31, | | December 31, | | December 31, |
| | 2019 | | 2018 | | 2021 | | 2020 |
| | | | | |
| Assets | | | | Assets | | | |
| Current assets: | | | | Current assets: | | | |
| Cash and cash equivalents | $ | 89,893 |
| | $ | 37,403 |
| Cash and cash equivalents | $ | 114,057 | | | $ | 138,685 | |
| Restricted cash | 279 |
| | — |
| |
| | Marketable securities | | Marketable securities | 13,998 | | | — | |
| Accounts receivable | 5,000 |
| | — |
| Accounts receivable | 9,914 | | | 7,224 | |
| Prepaid expenses and other current assets | 1,495 |
| | 4,673 |
| Prepaid expenses and other current assets | 6,474 | | | 5,434 | |
| Total current assets | 96,667 |
|
| 42,076 |
| Total current assets | 144,443 | | | 151,343 | |
| Non-current assets: | | Non-current assets: | |
| Property and equipment, net | 1,222 |
| | 2,127 |
| Property and equipment, net | 2,142 | | | 1,395 | |
| Right-of-use asset, net | 301 |
| | — |
| Right-of-use asset, net | 9,829 | | | 10,948 | |
| Long-term restricted cash | 1,379 |
| | 279 |
| Long-term restricted cash | 1,379 | | | 1,379 | |
| Investments | | Investments | 2,000 | | | — | |
| Other assets | | Other assets | 90 | | | 370 | |
| Total assets | $ | 99,569 |
|
| $ | 44,482 |
| Total assets | $ | 159,883 | | | $ | 165,435 | |
| Liabilities and stockholders’ equity (deficit) | | | | Liabilities and stockholders’ equity (deficit) | | | |
| Current liabilities: | | | | Current liabilities: | | | |
| Accounts payable | $ | 500 |
| | $ | 1,100 |
| Accounts payable | $ | 224 | | | $ | 443 | |
| Accrued expenses | 13,492 |
| | 11,700 |
| Accrued expenses | 10,533 | | | 8,146 | |
| Loan payable | 18,905 |
| | 21,385 |
| Loan payable | 5,961 | | | — | |
| Lease liability | 372 |
| | — |
| Lease liability | 1,049 | | | 908 | |
| Income taxes payable | | Income taxes payable | 601 | | | — | |
| Deferred revenue | 1,674 |
| | 959 |
| Deferred revenue | 53,883 | | | 72,050 | |
| | Total current liabilities | 34,943 |
|
| 35,144 |
| Total current liabilities | 72,251 | | | 81,547 | |
| Non‑current liabilities: | | | | |
| Non-current liabilities: | | Non-current liabilities: | |
| | Loan payable, net of current portion | | Loan payable, net of current portion | 19,673 | | | 24,793 | |
| Lease liability, net of current portion | | Lease liability, net of current portion | 8,598 | | | 9,647 | |
| Deferred revenue | 14,680 |
| | 13,818 |
| Deferred revenue | 11,417 | | | 38,746 | |
| Warrant liabilities | 41,549 |
| | — |
| Warrant liabilities | 25,423 | | | 28,708 | |
| Other long‑term liabilities | — |
| | 938 |
| |
| | Total liabilities | 91,172 |
|
| 49,900 |
| Total liabilities | 137,362 | | | 183,441 | |
| Commitments and contingencies (Note 17) |
| |
| Commitments and contingencies (Note 17) | 0 | | 0 |
| | Stockholders’ equity (deficit): | | | | Stockholders’ equity (deficit): | | | |
| Preferred stock, $0.0001 par value; 10,000,000 shares authorized; no shares issued and outstanding at December 31, 2019 and December 31, 2018, respectively | — |
| | — |
| |
| Common stock, $0.0001 par value; 200,000,000 shares authorized; 86,325,547 and 22,471,776 shares issued and outstanding as of December 31, 2019 and December 31, 2018, respectively | 9 |
| | 3 |
| |
| Preferred stock, $0.0001 par value; 10,000,000 shares authorized; no shares issued and outstanding at December 31, 2021 and December 31, 2020 | | Preferred stock, $0.0001 par value; 10,000,000 shares authorized; no shares issued and outstanding at December 31, 2021 and December 31, 2020 | — | | | — | |
| Common stock, $0.0001 par value; 200,000,000 shares authorized; 123,622,965 and 108,071,249 shares issued and outstanding as of December 31, 2021 and December 31, 2020, respectively | | Common stock, $0.0001 par value; 200,000,000 shares authorized; 123,622,965 and 108,071,249 shares issued and outstanding as of December 31, 2021 and December 31, 2020, respectively | 12 | | | 11 | |
| Additional paid-in capital | 348,664 |
| | 279,539 |
| Additional paid-in capital | 457,391 | | | 391,175 | |
| | Accumulated deficit | (335,753 | ) | | (280,403 | ) | Accumulated deficit | (430,316) | | | (404,629) | |
| Accumulated other comprehensive loss | (4,523 | ) | | (4,557 | ) | Accumulated other comprehensive loss | (4,566) | | | (4,563) | |
| Total stockholders’ equity (deficit) | 8,397 |
| | (5,418 | ) | Total stockholders’ equity (deficit) | 22,521 | | | (18,006) | |
| Total liabilities and stockholders’ equity (deficit) | $ | 99,569 |
|
| $ | 44,482 |
| Total liabilities and stockholders’ equity (deficit) | $ | 159,883 | | | $ | 165,435 | |
The accompanying notes are an integral part of these consolidated financial statements.
Selecta Biosciences, Inc. and Subsidiaries
Consolidated Statements of Operations and Comprehensive Loss
(Amounts in thousands, except share and per share data)
| | | | | | | | | | | Year Ended December 31, |
| | Year Ended December 31, | | | 2021 | | 2020 | | 2019 |
| | 2019 | | 2018 | | 2017 | |
| | | | | | | |
| Grant and collaboration revenue | $ | 6,677 |
| | $ | 903 |
| | $ | 207 |
| |
| Collaboration and license revenue | | Collaboration and license revenue | | $ | 85,077 | | | $ | 16,597 | | | $ | 6,677 | |
| Operating expenses: | | | | | | Operating expenses: | | |
| Research and development | 42,743 |
| | 47,687 |
| | 45,165 |
| Research and development | | 68,736 | | | 54,505 | | | 42,743 | |
| General and administrative | 16,389 |
| | 18,238 |
| | 18,826 |
| General and administrative | | 20,938 | | | 18,913 | | | 16,389 | |
| Total operating expenses | 59,132 |
|
| 65,925 |
| | 63,991 |
| Total operating expenses | | 89,674 | | | 73,418 | | | 59,132 | |
| Loss from operations | (52,455 | ) | | (65,022 | ) | | (63,784 | ) | |
| Operating loss | | Operating loss | | (4,597) | | | (56,821) | | | (52,455) | |
| Investment income | 834 |
| | 1,050 |
| | 617 |
| Investment income | | 44 | | | 260 | | | 834 | |
| Loss on extinguishment of debt | — |
| | — |
| | (673 | ) | Loss on extinguishment of debt | | — | | | (461) | | | — | |
| Foreign currency transaction (loss), net | (47 | ) | | 120 |
| | (123 | ) | |
| Foreign currency transaction gain (loss), net | | Foreign currency transaction gain (loss), net | | — | | | 56 | | | (47) | |
| Interest expense | (1,519 | ) | | (1,494 | ) | | (1,206 | ) | Interest expense | | (2,844) | | | (1,556) | | | (1,519) | |
| Change in fair value of warrant liabilities | (857 | ) | | — |
| | — |
| Change in fair value of warrant liabilities | | (2,339) | | | (10,443) | | | (857) | |
| Other (expense), net | (1,306 | ) | | 10 |
| | (152 | ) | |
| Other income (expense), net | | Other income (expense), net | | 15 | | | 89 | | | (1,306) | |
| Loss before income taxes | | Loss before income taxes | | (9,721) | | | (68,876) | | | (55,350) | |
| Income tax expense | | Income tax expense | | (15,966) | | | — | | | — | |
| Net loss | (55,350 | ) |
| (65,336 | ) | | (65,321 | ) | Net loss | | (25,687) | | | (68,876) | | | (55,350) | |
| Other comprehensive loss: | | | | | | |
| | Other comprehensive income (loss): | | Other comprehensive income (loss): | | |
| Foreign currency translation adjustment | 34 |
| | (153 | ) | | 78 |
| Foreign currency translation adjustment | | (2) | | | (40) | | | 34 | |
| Unrealized gain on securities | — |
| | 16 |
| | 20 |
| |
| Unrealized loss on marketable securities | | Unrealized loss on marketable securities | | (1) | | | — | | | — | |
| Total comprehensive loss | $ | (55,316 | ) |
| $ | (65,473 | ) | | $ | (65,223 | ) | Total comprehensive loss | | $ | (25,690) | | | $ | (68,916) | | | $ | (55,316) | |
| | | | | | | | | |
| Net loss per share: | | | | | | Net loss per share: | | |
| Basic and diluted | $ | (1.22 | ) | | $ | (2.92 | ) | | $ | (3.20 | ) | Basic and diluted | | $ | (0.22) | | | $ | (0.68) | | | $ | (1.22) | |
| | Weighted average common shares outstanding: | | | | | | Weighted average common shares outstanding: | | | | | | |
| Basic and diluted | 45,548,511 |
| | 22,389,286 |
| | 20,425,050 |
| Basic and diluted | | 114,328,798 | | | 101,202,176 | | | 45,548,511 | |
|
The accompanying notes are an integral part of these consolidated financial statements.
Selecta Biosciences, Inc. and Subsidiaries
Consolidated Statements of Changes in Stockholders’ Equity (Deficit)
(Amounts in thousands, except share data)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | Accumulated | | |
| | | | | Additional | | | | | | other | | Stockholders’ |
| | Common stock | | paid-in | | | | Accumulated | | comprehensive | | Equity |
| | Shares | Amount | | capital | | | | deficit | | loss | | (Deficit) |
| Balance at December 31, 2018 | 22,471,776 | | $ | 3 | | | $ | 279,539 | | | | | $ | (280,403) | | | $ | (4,557) | | | $ | (5,418) | |
| Issuance of common stock under Employee Stock Purchase Plan | 17,205 | | — | | | 28 | | | | | — | | | — | | | 28 | |
| Issuance of common stock upon exercise of options | 125,600 | | — | | | 150 | | | | | — | | | — | | | 150 | |
| Issuance of vested restricted stock units | 93,750 | | — | | | — | | | | | — | | | — | | | — | |
| Issuance of common stock, net | 22,188,706 | | 2 | | | 30,940 | | | | | — | | | — | | | 30,942 | |
| Issuance of common stock through at-the-market offering, net | 615,453 | | — | | | 1,006 | | | | | — | | | — | | | 1,006 | |
| Issuance of common stock through private placement | 3,178,174 | | — | | | 5,715 | | | | | — | | | — | | | 5,715 | |
| Issuance of common stock, pre-funded warrants and warrants through private placement | 37,634,883 | | 4 | | | 26,125 | | | | | — | | | — | | | 26,129 | |
| Stock-based compensation expense | — | | — | | | 5,161 | | | | | — | | | — | | | 5,161 | |
| Currency translation adjustment | — | | — | | | — | | | | | — | | | 34 | | | 34 | |
| Net loss | — | | — | | | — | | | | | (55,350) | | | — | | | (55,350) | |
| Balance at December 31, 2019 | 86,325,547 | | $ | 9 | | | $ | 348,664 | | | | | $ | (335,753) | | | $ | (4,523) | | | $ | 8,397 | |
| Issuance of common stock under Employee Stock Purchase Plan | 110,212 | | — | | | 184 | | | | | — | | | — | | | 184 | |
| Issuance of common stock upon exercise of options | 76,128 | | — | | | 193 | | | | | — | | | — | | | 193 | |
| Issuance of vested restricted stock units | 93,750 | | — | | | — | | | | | — | | | — | | | — | |
| Issuance of common stock through at-the-market offering, net | 1,069,486 | | — | | | 2,108 | | | | | — | | | — | | | 2,108 | |
| Issuance of common stock through private placement | 5,416,390 | | — | | | 10,268 | | | | | — | | | — | | | 10,268 | |
| Issuance of common stock upon exercise of pre-funded warrants | 8,342,128 | | 1 | | | — | | | | | — | | | — | | | 1 | |
| Issuance of common stock upon exercise of warrants | 6,637,608 | | 1 | | | 24,262 | | | | | — | | | — | | | 24,263 | |
| Other financing fees | — | | — | | | (370) | | | | | — | | | — | | | (370) | |
| Issuance of common warrants with long-term debt, net | — | | — | | | 444 | | | | | — | | | — | | | 444 | |
| Stock-based compensation expense | — | | — | | | 5,422 | | | | | — | | | — | | | 5,422 | |
| Currency translation adjustment | — | | — | | | — | | | | | — | | | (40) | | | (40) | |
| Net loss | — | | — | | | — | | | | | (68,876) | | | — | | | (68,876) | |
| Balance at December 31, 2020 | 108,071,249 | | $ | 11 | | | $ | 391,175 | | | | | $ | (404,629) | | | $ | (4,563) | | | $ | (18,006) | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| Issuance of common stock under Employee Stock Purchase Plan | 58,794 | | — | | | 161 | | | | | — | | | — | | | 161 | |
| Issuance of common stock upon exercise of options | 447,492 | | — | | | 778 | | | | | — | | | — | | | 778 | |
| Issuance of vested restricted stock units | 201,250 | | — | | | — | | | | | — | | | — | | | — | |
| Issuance of common stock through at-the-market offering, net | 13,767,511 | | 1 | | | 51,933 | | | | | — | | | — | | | 51,934 | |
| Issuance of common stock upon exercise of warrants | 1,076,669 | | — | | | 5,624 | | | | | — | | | — | | | 5,624 | |
| | | | | | | | | | | | |
| Stock-based compensation expense | — | | — | | | 7,720 | | | | | — | | | — | | | 7,720 | |
| Currency translation adjustment | — | | — | | | — | | | | | — | | | (2) | | | (2) | |
| Unrealized loss on marketable securities | — | | — | | | — | | | | | — | | | (1) | | | (1) | |
| Net loss | — | | — | | | — | | | | | (25,687) | | | — | | | (25,687) | |
| Balance at December 31, 2021 | 123,622,965 | | $ | 12 | | | $ | 457,391 | | | | | $ | (430,316) | | | $ | (4,566) | | | $ | 22,521 | |
The accompanying notes are an integral part of these consolidated financial statements.
|
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | Accumulated | | |
| | | | | Additional | | Stock | | | | other | | Stockholders’ |
| | Common stock | | paid‑in | | option | | Accumulated | | comprehensive | | Equity |
| | Shares | Amount | | capital | | receivable | | deficit | | loss | | (Deficit) |
| Balance at December 31, 2016 | 18,438,742 |
| $ | 1 |
| | $ | 211,125 |
| | $ | (75 | ) | | $ | (151,576 | ) | | $ | (4,518 | ) | | $ | 54,957 |
|
| Issuance of common stock under Employee Stock Purchase Plan | 16,263 |
| — |
| | 180 |
| | — |
| | — |
| | — |
| | 180 |
|
| Issuance of common stock upon exercise of options | 269,842 |
| — |
| | 630 |
| | 75 |
| | — |
| | — |
| | 705 |
|
| Stock‑based compensation expense | — |
| — |
| | 4,080 |
| | — |
| | — |
| | — |
| | 4,080 |
|
| Issuance of common stock, license agreement | 529,616 |
| — |
| | 10,000 |
| | — |
| | — |
| | — |
| | 10,000 |
|
| Issuance of common stock through private placement, net of issuance costs | 3,088,791 |
| 2 |
| | 47,113 |
| | — |
| | — |
| | — |
| | 47,115 |
|
| Currency translation adjustment | — |
| — |
| | — |
| | — |
| | — |
| | 78 |
| | 78 |
|
| Unrealized gains on securities | — |
| — |
| | — |
| | — |
| | — |
| | 20 |
| | 20 |
|
| Net loss | — |
| — |
| | — |
| | — |
| | (65,321 | ) | | — |
| | (65,321 | ) |
| Balance at December 31, 2017 | 22,343,254 |
| $ | 3 |
| | $ | 273,128 |
| | $ | — |
| | $ | (216,897 | ) | | $ | (4,420 | ) | | $ | 51,814 |
|
| Adoption of new accounting principle | — |
| — |
| | — |
| | — |
| | 1,830 |
| | — |
| | 1,830 |
|
| Issuance of common stock under Employee Stock Purchase Plan | 24,738 |
| — |
| | 196 |
| | — |
| | — |
| | — |
| | 196 |
|
| Issuance of common stock upon exercise of options | 103,784 |
| — |
| | 501 |
| | — |
| | — |
| | — |
| | 501 |
|
| Stock‑based compensation expense | — |
| — |
| | 5,714 |
| | — |
| | — |
| | — |
| | 5,714 |
|
| Currency translation adjustment | — |
| — |
| | — |
| | — |
| | — |
| | (153 | ) | | (153 | ) |
| Unrealized gains on securities | — |
| — |
| | — |
| | — |
| | — |
| | 16 |
| | 16 |
|
| Net loss | — |
| — |
| | — |
| | — |
| | (65,336 | ) | | — |
| | (65,336 | ) |
| Balance at December 31, 2018 | 22,471,776 |
| $ | 3 |
| | $ | 279,539 |
| | $ | — |
| | $ | (280,403 | ) | | $ | (4,557 | ) | | $ | (5,418 | ) |
| Issuance of common stock under Employee Stock Purchase Plan | 17,205 |
| — |
| | 28 |
| | — |
| | — |
| | — |
| | 28 |
|
| Issuance of common stock upon exercise of options | 125,600 |
| — |
| | 150 |
| | — |
| | — |
| | — |
| | 150 |
|
| Issuance of vested restricted stock units | 93,750 |
| — |
| | — |
| | — |
| | — |
| | — |
| | — |
|
| Issuance of common stock, net of issuance costs | 22,188,706 |
| 2 |
| | 30,940 |
| | — |
| | — |
| | — |
| | 30,942 |
|
| Issuance of common stock through at-the-market offering, net of issuance costs | 615,453 |
| — |
| | 1,006 |
| | — |
| | — |
| | — |
| | 1,006 |
|
| Issuance of common stock through private placement, net of issuance costs | 3,178,174 |
| — |
| | 5,715 |
| | — |
| | — |
| | — |
| | 5,715 |
|
| Issuance of common stock, pre-funded warrants and warrants through private placement, net of issuance costs | 37,634,883 |
| 4 |
| | 26,125 |
| | — |
| | — |
| | — |
| | 26,129 |
|
| Stock‑based compensation expense | — |
| — |
| | 5,161 |
| | — |
| | — |
| | — |
| | 5,161 |
|
| Currency translation adjustment | — |
| — |
| | — |
| | — |
| | — |
| | 34 |
| | 34 |
|
| Net loss | — |
| — |
| | — |
| | — |
| | (55,350 | ) | | — |
| | (55,350 | ) |
| Balance at December 31, 2019 | 86,325,547 |
| $ | 9 |
| | $ | 348,664 |
| | $ | — |
| | $ | (335,753 | ) | | $ | (4,523 | ) | | $ | 8,397 |
|
Selecta Biosciences, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
| | | | | | | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| | | | 2021 | | 2020 | | 2019 |
| | | | | | |
| Cash flows from operating activities | | | | (Amounts in thousands) |
| Net loss | | | | $ | (25,687) | | | $ | (68,876) | | | $ | (55,350) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | | | |
| Depreciation and amortization | | | | 1,252 | | | 734 | | | 726 | |
| Amortization of premiums and discounts on marketable securities | | | | 57 | | | — | | | (154) | |
| Non-cash lease expense | | | | 1,119 | | | 1,127 | | | 1,301 | |
| Loss on disposal of property and equipment | | | | — | | | (52) | | | 104 | |
| Stock-based compensation expense | | | | 7,720 | | | 5,422 | | | 5,161 | |
| Non-cash interest expense | | | | 1,012 | | | 620 | | | 402 | |
| Warrant liabilities revaluation | | | | 2,339 | | | 10,443 | | | 857 | |
| Loss on extinguishment of debt | | | | — | | | 461 | | | — | |
| Net realized losses on marketable securities | | | | — | | | — | | | (1) | |
| Changes in operating assets and liabilities: | | | | | | | | |
| Accounts receivable | | | | (2,690) | | | (2,224) | | | (5,000) | |
| Prepaid expenses, deposits and other assets | | | | (1,451) | | | (4,418) | | | 3,179 | |
| Accounts payable | | | | (219) | | | (57) | | | (600) | |
| Income taxes payable | | | | 601 | | | — | | | — | |
| Deferred revenue | | | | (45,496) | | | 94,462 | | | 337 | |
| | | | | | | | |
| Accrued expenses and other liabilities | | | | 1,061 | | | (2,761) | | | (2,397) | |
| Net cash (used in) provided by operating activities | | | | (60,382) | | | 34,881 | | | (51,435) | |
| Cash flows from investing activities | | | | | | | | |
| Proceeds from maturities of marketable securities | | | | 16,400 | | | — | | | 16,350 | |
| Payment made for investments | | | | (2,000) | | | — | | | — | |
| Purchases of marketable securities | | | | (30,455) | | | — | | | (18,188) | |
| Sales of marketable securities | | | | — | | | — | | | 1,992 | |
| Purchases of property and equipment | | | | (1,085) | | | (815) | | | (47) | |
| Proceeds from the sale of property and equipment | | | | — | | | 74 | | | 122 | |
| | | | | | | | |
| Net cash used in investing activities | | | | (17,140) | | | (741) | | | 229 | |
| Cash flows from financing activities | | | | | | | | |
| Proceeds from issuance of long-term debt, net of expenses | | | | — | | | 24,736 | | | — | |
| Repayments of principal on outstanding debt | | | | — | | | (19,313) | | | (2,800) | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
| Net proceeds from issuance of common stock | | | | — | | | — | | | 30,942 | |
| Net proceeds from issuance of common stock- at-the-market offering | | | | 51,958 | | | 2,108 | | | 1,006 | |
| Net proceeds from issuance of common stock- private placement | | | | — | | | 10,268 | | | 5,715 | |
| Issuance costs paid for December 2019 financing | | | | — | | | (4,381) | | | — | |
| Other financing fees | | | | — | | | (343) | | | — | |
| Proceeds from exercise of pre-funded and common warrants | | | | — | | | 979 | | | 70,000 | |
| Proceeds from exercise of stock options | | | | 778 | | | 193 | | | 150 | |
| Proceeds from issuance of common stock under Employee Stock Purchase Plan | | | | 161 | | | 184 | | | 28 | |
| Net cash provided by financing activities | | | | 52,897 | | | 14,431 | | | 105,041 | |
| Effect of exchange rate changes on cash | | | | (3) | | | (58) | | | 34 | |
| Net change in cash, cash equivalents, and restricted cash | | | | (24,628) | | | 48,513 | | | 53,869 | |
| Cash, cash equivalents, and restricted cash at beginning of period | | | | 140,064 | | | 91,551 | | | 37,682 | |
| Cash, cash equivalents, and restricted cash at end of period | | | | $ | 115,436 | | | $ | 140,064 | | | $ | 91,551 | |
| Supplement cash flow information | | | | | | | | |
| Cash paid for interest | | | | $ | 2,002 | | | $ | 1,018 | | | $ | 1,223 | |
| Noncash investing and financing activities | | | | | | | | |
| Cashless warrant exercise | | | | $ | 5,624 | | | $ | 21,790 | | | $ | — | |
| Reclassification of warrant liability to equity upon exercise of warrants | | | | $ | — | | | $ | 1,494 | | | $ | — | |
| Fair value of warrants issued in connection with issuance of long-term debt | | | | $ | — | | | $ | 444 | | | $ | — | |
| Purchase of property and equipment not yet paid | | | | $ | 224 | | | $ | 4 | | | $ | — | |
| Equity offering costs in accrued liabilities | | | | $ | 24 | | | $ | 27 | | | $ | 4,381 | |
| Unrealized loss on marketable securities | | | | $ | (1) | | | $ | — | | | $ | — | |
| | | | | | | | |
| | | | | | | | |
| Debt issuance costs in accrued liabilities | | | | $ | — | | | $ | 2 | | | $ | — | |
| | | | | | | | |
| | | | | | | | |
The accompanying notes are an integral part of these consolidated financial statements.
Selecta Biosciences, Inc. and Subsidiaries
(Amounts in thousands) |
| | | | | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| | | | | | |
| Cash flows from operating activities | | | | | |
| Net loss | $ | (55,350 | ) | | $ | (65,336 | ) | | $ | (65,321 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | | | |
| Depreciation | 726 |
| | 975 |
| | 750 |
|
| Amortization of premiums (accretion of discounts) on investments | (154 | ) | | (101 | ) | | 233 |
|
| Amortization of right-of-use assets | 1,301 |
| | — |
| | — |
|
| Loss (gain) on disposal of property and equipment | 104 |
| | (81 | ) | | 36 |
|
| Stock‑based compensation expense | 5,161 |
| | 5,714 |
| | 4,081 |
|
| Non‑cash interest expense | 402 |
| | 449 |
| | 390 |
|
| Warrant liabilities revaluation | 857 |
| | — |
| | — |
|
| Loss on extinguishment of debt | — |
| | — |
| | 673 |
|
| Net realized losses on investments | (1 | ) | | — |
| | — |
|
| Changes in operating assets and liabilities: | | | | | |
| Accounts receivable | (5,000 | ) | | — |
| | 152 |
|
| Prepaid expenses, deposits and other assets | 3,179 |
| | (2,631 | ) | | 1,225 |
|
| Accounts payable | (600 | ) | | (516 | ) | | (2,327 | ) |
| Deferred revenue | 337 |
| | (101 | ) | | 2,432 |
|
| Accrued expenses and other liabilities | (2,397 | ) | | 2,467 |
| | 5,650 |
|
| Net cash used in operating activities | (51,435 | ) | | (59,161 | ) | | (52,026 | ) |
| Cash flows from investing activities | | | | | |
| Receipts from the maturity of short-term investments | 16,350 |
| | 41,655 |
| | 60,158 |
|
| Purchases of short-term investments | (18,188 | ) | | (15,598 | ) | | (61,527 | ) |
| Sale of short term investments | 1,992 |
| | — |
| | — |
|
| Purchases of property and equipment | (47 | ) | | (884 | ) | | (733 | ) |
| Proceeds from the sale of property and equipment | 122 |
| | 99 |
| | 4 |
|
| Net cash provided by (used in) investing activities | 229 |
| | 25,272 |
| | (2,098 | ) |
| Cash flows from financing activities | | | | | |
| Proceeds from issuance of long-term debt, net of expenses | — |
| | — |
| | 20,957 |
|
| Repayments of principal on outstanding debt | (2,800 | ) | | — |
| | (12,934 | ) |
| Net proceeds from issuance of common stock | 30,942 |
| | — |
| | 10,000 |
|
| Net proceeds from issuance of common stock- at-the-market offering | 1,006 |
| | — |
| | — |
|
| Net proceeds from issuance of common stock- private placement | 5,715 |
| | — |
| | 47,114 |
|
| Net proceeds from issuance of common stock, common warrants, and pre-funded warrants | 70,000 |
| | — |
| | — |
|
| Proceeds from exercise of stock options | 150 |
| | 501 |
| | 706 |
|
| Proceeds from issuance of common stock under Employee Stock Purchase Plan | 28 |
| | 196 |
| | 180 |
|
| Net cash provided by financing activities | 105,041 |
| | 697 |
| | 66,023 |
|
| Effect of exchange rate changes on cash | 34 |
| | (153 | ) | | 78 |
|
| Net change in cash, cash equivalents, and restricted cash | 53,869 |
| | (33,345 | ) | | 11,977 |
|
| Cash, cash equivalents, and restricted cash at beginning of period | 37,682 |
| | 71,027 |
| | 59,050 |
|
| Cash, cash equivalents, and restricted cash at end of period | $ | 91,551 |
| | $ | 37,682 |
| | $ | 71,027 |
|
| Supplement cash flow information | | | | | |
| Cash paid for interest | $ | 1,223 |
| | $ | 1,134 |
| | $ | 905 |
|
| Noncash investing and financing activities | | | | | |
| Purchase of property and equipment not yet paid | $ | — |
| | $ | 145 |
| | $ | 103 |
|
| Issuance costs in connection with common stock, common warrants, and pre-funded warrants in accrued liabilities | $ | 4,381 |
| | $ | — |
| | $ | — |
|
| Unrealized gain on marketable securities | $ | — |
| | $ | 16 |
| | $ | 20 |
|
The accompanying notes are an integral part of these consolidated financial statements.
Selecta Biosciences, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
1. NatureDescription of the Business and Basis of Presentation
Selecta Biosciences, Inc. (the “Company”), or the Company, was incorporated in Delaware on December 10, 2007, and is based in Watertown, Massachusetts. The Company is a clinical-stage biotechnology company focused on unlockingbiopharmaceutical company. The Company’s ImmTOR® platform encapsulates rapamycin, also known as sirolimus, an FDA approved immunomodulator, in biodegradable nanoparticles ImmTOR is designed to induce antigen-specific immune tolerance. The Company believes, by combining ImmTOR with antigens of interest, the full potential of biologic therapies based on itsCompany’s precision immune tolerance technology (ImmTOR��) platform. The Company plansplatform has the potential to combine ImmTOR with a rangerestore self-tolerance to auto-antigens in autoimmune diseases, amplify the efficacy of biologics (including gene therapies) and mitigate the formation of anti-drug antibodies, or ADAs, against biologic therapies for rare and serious diseases that require new treatment options due to high immunogenicity of existing therapies. drugs.
Since inception, the Company has devoted its efforts principally to research and development of its technology and product candidates, recruiting management and technical staff, acquiring operating assets, and raising capital.
The Company is subject to risks common to companies in the biotechnology industry including, but not limited to, new technological innovations, protection of proprietary technology, dependence on key personnel, compliance with government regulations and the need to obtain additional financing. Product candidates currently under development will require significant additional research and development efforts, including extensive preclinical and clinical testing and regulatory approval, prior to commercialization. These efforts require significant amounts of additional capital, adequate personnel infrastructure and extensive compliance-reporting capabilities.
The Company’s product candidates are in development. There can be no assurance that the Company’s research and development will be successfully completed, that adequate protection for the Company’s intellectual property will be obtained, or maintained, that any products developed will obtain necessary government regulatory approval or that any approved products will be commercially viable. Even if the Company’s product development efforts are successful, it is uncertain when, if ever, the Company will generate significant revenue from product sales. The Company operates in an environment of rapid change in technology and substantial competition from pharmaceutical and biotechnology companies. In addition, the Company is dependent upon the services of its employees and consultants.
The accompanying financial statements have been prepared on a basis that assumes the Company is a going concern, and does not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classifications of liabilities that may result from any uncertainty related to its ability to continue as a going concern.
Liquidity and Management'sManagement’s Plan
The future success of the Company is dependent on its ability to develop its product candidates and ultimately upon its ability to attain and sustain profitable operations. The Company is subject to a number of risks similar to other early-stage life science companies, including, but not limited to, successful development of its product candidates, raising additional capital with favorable terms, protection of proprietary technology and market acceptance of any approved future products. The successful development of product candidates requires substantial working capital, which may not be available to the Company on favorable terms or at all.
To date, the Company has financed its operations primarily through the initial public offering of its common stock, a private placementplacements of its common stock, issuances of common and preferred stock, debt, research grants, research collaborations and research collaborations.licenses. The Company currently has no source of product revenue, and it does not expect to generate product revenue for the foreseeable future. To date, all of the Company'sCompany’s revenue has primarily been from collaboration and grant revenue.agreements. The Company has devoted substantially all of its financial resources and efforts to developing its ImmTOR platform, identifying potential product candidates and conducting preclinical studies and its clinical trials. The Company is in the early stages of development of its product candidates, and it has not completed development of any ImmTOR-enabled therapies.
As of December 31, 2019,2021, the Company’s cash, cash equivalents, and restricted cash and marketable securities were $91.6$129.4 million, of which $1.7$1.4 million was restricted cash related to lease commitments and $0.4$0.3 million was held by its Russian subsidiary designated solely for use in its operations. The Company has incurred lossesbelieves the cash, cash equivalents, restricted cash and negativemarketable securities as of December 31, 2021 will enable it to fund its current planned operations for at least the next twelve months from the date of issuance of these financial statements, though it may realize additional cash flowsresources upon the achievement of certain contingent collaboration milestones or it may pursue additional cash resources through public or private equity or debt financings or by establishing collaborations with other companies. Management’s expectations with respect to its ability to fund current and long term planned operations are based on estimates that are subject to risks and uncertainties. If actual results are different from operating activities since inception.management’s estimates, the Company may need to seek additional strategic or financing opportunities sooner than would otherwise be expected. However, there is no guarantee that any collaboration milestones will be achieved or that any of these strategic or financing opportunities will be executed on favorable terms, and some could be dilutive to existing stockholders. If the Company is unable to obtain additional funding on a timely basis, it may be forced to significantly curtail, delay, or discontinue one or more of its planned research or development programs or be unable to expand its operations or otherwise capitalize on its commercialization of its product candidates. As of December 31, 2019 and December 31, 2018,2021, the Company had an accumulated deficit of $335.8 million and $280.4 million, respectively.$430.3 million. The Company anticipates operating losses to continue for the
foreseeable future due to, among other things, costs related to research and development of its product candidates conducting preclinical studies and clinical trials, and its administrative organization. The Company will require substantial additional financing to fund its
At this time, any impact of COVID-19 on the Company’s business, revenues, results of operations and to continue to execute its strategy,financial condition will largely depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the duration of the pandemic, the emergence of new virus variants, travel restrictions and social distancing in the United States and other countries, business closures or disruptions, supply chain disruptions, the ultimate impact on financial markets and the Company will pursue a rangeglobal economy, and the effectiveness of options to secure additional capital. These conditions raise
substantial doubt aboutactions taken in the Company's ability to continue as a going concern within one year after the date that the financial statements are issued.
Management is actively exploring licensesUnited States and other strategic collaborations that havecountries to contain and treat the potential to provide non-dilutive capital and accelerate the development of new or existing product candidates incorporating the Company’s ImmTOR platform. Additionally, the Company may seek to fund its operations through issuances of equity and other securities. If the Company enters into strategic collaborations and alliances, which may include existing collaboration partners, the Company may have to relinquish valuable rights to its technologies or product candidates, or grant licenses on terms that are not favorable to the Company. To the extent that the Company raises additional capital through the sale of equity, the ownership interest of its existing shareholders will be diluted and other preferences may be necessary that adversely affect the rights of existing shareholders. The Company requires additional external sources of capital to complete the planned Phase 3 clinical program for SEL-212. If the Company is unable to raise sufficient capital through strategic collaborations and the sale of equity or other securities, it intends to curtail expenses contemplated by the current operating plan, and the Company may be required to delay, limit, reduce or terminate its product development efforts or grant rights to develop and market product candidates that it would otherwise prefer to develop and market itself. Because of the uncertainty in securing additional capital and the insufficient amount of capital resources at December 31, 2019, management has concluded that substantial doubt exists with respect to the Company's ability to continue as a going concern within one year after the date of the filing of this Annual Report on Form 10-K.
All amounts due under the 2017 Term Loan (see Note 9) have been classified as a current liability as of December 31, 2019 due to the considerations discussed above and the assessment that the material adverse change clause under the 2017 Term Loan is not within the Company's control. The Company has not been notified of an event of default by the Lender as of the date of the filing of this Annual Report on Form 10-K.
Guarantees and Indemnifications
As permitted under Delaware law, the Company indemnifies its officers, directors, consultants and employees for certain events or occurrences that happen by reason of the relationship with, or position held at, the Company. Through December 31, 2019,2021, the Company had not experienced any losses related to these indemnification obligations, and no claims were outstanding. The Company does not expect significant claims related to these indemnification obligations and, consequently, concluded that the fair value of these obligations is negligible, and no related reserves were established.
2. Summary of Significant Accounting Policies
Principles of Consolidation
The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries, Selecta RUS,(RUS), LLC, (“or Selecta (RUS)”), a Russian limited liability corporation, and Selecta Biosciences Security Corporation, a Massachusetts Security Corporation.securities corporation. All significant intercompany accounts and transactions have been eliminated.
Foreign Currency
The functional currency of Selecta (RUS) is the Russian ruble. Assets and liabilities of Selecta (RUS) are translated at period-end exchange rates, while revenues and expenses are translated at average exchange rates for the period. Translation gains and losses are reflected in accumulated other comprehensive loss within stockholders’ equity (deficit). Foreign currency transaction gains or losses are reflected in the consolidated statements of operations and comprehensive loss.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires the Company’s management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities atas of the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. The Company’s management considers many factors in selecting appropriate financial accounting policies and controls, and bases its estimates on historical experience and other market-specific or other relevant assumptions that it believes to be reasonable under the circumstances. In preparing these consolidated financial statements, management used significant estimates in the following areas, among others: revenue recognition, accounting for stock-based compensation, the valuation of its warrant liabilities and estimating accrued research and development expenses. The Company assesses the above estimates on an ongoing basis; however, actual results could materially differ from those estimates.
Segment Information
Operating segments are identified as components of an enterprise about which separate discrete financial information is available for evaluation by the chief operating decision maker, the Company’s Chief Executive Officer, in making decisions regarding resource allocation and assessing performance. The Company views its operations and manages its business in 1 operating segment, the research and development of nanoparticle immunomodulatory drugs for the treatment and prevention of human diseases.
Cash Equivalents, Short-term Investments and Restricted Cash, Marketable Securities and Investments
Cash equivalents include all highly liquid investments maturing within 90 days from the date of purchase. InvestmentsMarketable securities consist of securities with remaining maturities greater than 90 days when purchased. The Company classifies these marketable securities as available-for-sale and records them at fair value in the accompanying consolidated balance sheets. InvestmentsMarketable securities with less than one year until maturity are classified as short term, while investmentsmarketable securities with maturities greater than one year are classified as long term. Unrealized gains or losses are included in accumulated other comprehensive income (loss). Premiums or discounts from par value are amortized to investment income over the life of the underlying investment.
Although available to be sold to meet operating needs or otherwise, securities are generally held through maturity. The cost of securities sold is determined based on the specific identification method for purposes of recording realized gains and losses. During
We also invest in equity securities of companies whose securities are not publicly traded and where fair value is not readily available. These investments are recorded using cost minus impairment adjusted for changes in observable prices, depending on our ownership percentage and other factors that suggest we have significant influence. We monitor these investments to evaluate whether any increase or decline in their value has occurred, based on the year ended December 31, 2019, there were de minimis realized losses on salesimplied value of recent company financings, public market prices of comparable companies and general market conditions. These investments are included in investments and no investments were adjusted for other than temporary declinesassets in fair value.
As of December 31, 2019, the Company had restricted cash balances relating to secured letters of credit in connection with its current Headquarters Lease and New Headquarters Lease (as defined in Note 8). The following table provides a reconciliation of cash, cash equivalents, and restricted cash reported within theour consolidated balance sheet that sum to the total of the same such amounts shown in the consolidated statement of cash flows:
|
| | | | | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| Cash and cash equivalents | $ | 89,893 |
| | $ | 37,403 |
| | $ | 70,622 |
|
| Restricted cash | 279 |
| | — |
| | 76 |
|
| Long-term restricted cash | 1,379 |
| | 279 |
| | 329 |
|
| Total cash, cash equivalents, and restricted cash shown in the consolidated statement of cash flows | $ | 91,551 |
| | $ | 37,682 |
| | $ | 71,027 |
|
Concentrations of Credit Risk and Off‑BalanceOff-Balance Sheet Risk
Financial instruments that potentially subject the Company to concentration of credit risk consist primarily of cash, cash equivalents, short-term deposits and marketable securities, investments, and accounts receivable. Cash and cash equivalents are deposited with federally insured financial institutions in the United States and may, at times, exceed federally insured limits.
Management believes that the financial institutions that hold the Company’s deposits are financially creditworthy and, accordingly, minimal risk exists with respect to those balances. Generally, these deposits may be redeemed upon demand and therefore bear minimal interest rate risk. As an integral part of operating its Russian subsidiary, theThe Company also maintains cash in Russian bank accounts in denominations of both Russian rubles and U.S. dollars. As of December 31, 2019,2021, the Company maintained approximately $0.4$0.3 million in Russian bank accounts, all of which was held in U.S. dollars.
The Company did not have any off-balance sheet arrangements as of December 31, 2019 and December 31, 2018.
Fair Value of Financial Instruments
The Company’s financial instruments consist mainly of cash equivalents, restricted cash, accounts payable, loans payable, marketable securities, investments and common warrants. The carrying amounts of cash equivalents, restricted cash, accounts receivable, and accounts payable approximate their estimated fair value due to their short-term maturities. At December 31, 2019, the carrying amount of the Company's loan payable approximates its estimated fair value due to the short-term nature of the instrument.
Accounting standards define fair value as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. A three‑levelthree-level hierarchy is used to prioritize the inputs to valuation techniques used to measure fair value. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurements), and the lowest priority to unobservable inputs (Level 3 measurements). The three levels of the fair value hierarchy are described below:
Level 1—Level 1 inputs are quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date.
Level 2—Level 2 inputs are inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly or indirectly. If the asset or liability has a specified (contractual) term, a Level 2 input must be observable for substantially the full term of the asset or liability.
Level 3—Level 3 inputs are unobservable inputs for the asset or liability in which there is little, if any, market activity for the asset or liability at the measurement date.
To the extent that a valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. The fair value of warrant liabilities wereis determined using Level 3 inputs.
Fair value is a market‑basedmarket-based measure considered from the perspective of a market participant rather than an entity‑specificentity-specific measure. Therefore, even when market assumptions are not readily available, the Company’s own assumptions are set to reflect those that market participants would use in pricing the asset or liability at the measurement date. The Company uses prices and inputs that are current as of the measurement date, including during periods of market dislocation. In periods of market dislocation, the observability of prices and inputs may change for many instruments. This condition could cause an instrument to be reclassified within levels in the fair value hierarchy. There were no transfers within the
The carrying amounts reflected in our consolidated balance sheet for investments approximate fair value, hierarchy during the years ended December 31, 2019 or December 31, 2018.and are assessed for impairment quarterly.
Property and Equipment
Property and equipment are recorded at cost and depreciated using the straight‑linestraight-line method over the estimated useful lives of the respective assets, generally seven years for furniture and fixtures, five years for laboratory equipment, software and office equipment and three years for computer equipment. Leasehold improvements are amortized over their useful life or the life of the lease, whichever is shorter. Major additions and betterments are capitalized. Maintenance and repairs, which do not improve or extend the life of the respective assets, are charged to operations as incurred. Costs incurred for construction in progress are recorded as assets and are not amortized until the construction is substantially complete and the assets are ready for their intended use.
Impairment of Long‑LivedLong-Lived Assets
The Company reviews long‑livedlong-lived assets for impairment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. In order to determine if assets have been impaired, assets are tested at the lowest level for which identifiable independent cash flows are available, which is at the entity level ("(“asset group"group”). An impairment loss is recognized when the sum of projected undiscounted cash flows is less than the carrying value of the asset group. The measurement of the impairment loss to be recognized is based on the difference between the fair value and the carrying value of the asset group. Based on management'smanagement’s evaluation, the fair value of the asset group, measured as the market capitalization of the Company exceeds its carrying value, and for this reason the Company did 0t recognize any material impairment losses during the years ended December 31, 2019 and 2018.value.
Debt Issuance Costs
Debt issuance costs and fees paid to lenders are classified as a debt discount and are recorded as a direct deduction from the face amount of the related debt. Issuance costs paid to third parties that are the direct result of the debt issuance are capitalized as a direct deduction from the face amount of the related debt. Debt issuance costs are amortized over the term of the related debt using the effective interest method and recorded as interest expense. Costs and fees paid to third parties are expensed as incurred.
Accumulated Other Comprehensive Income (Loss)
Comprehensive income (loss) is defined as the change in the equity of a business entity during a period from transactions and other events and circumstances from non‑ownernon-owner sources. It includes all changes in equity during a period except those resulting from investments by owners and distributions to owners. Comprehensive income (loss) consists of: (i) all components of net loss and (ii) all components of comprehensive loss other than net loss, referred to as other comprehensive loss. Other comprehensive loss is comprised of foreign currency translation adjustments and the unrealized gains and losses recognized through net income.
The components of accumulated other comprehensive income (loss), net of tax, were as follows (in thousands): |
| | | | | | | | | | | |
| | Foreign currency translation adjustment | | Unrealized gains (losses) on marketable securities | | Accumulated other comprehensive income (loss) |
| Balance at December 31, 2016 | $ | (4,482 | ) | | $ | (36 | ) | | $ | (4,518 | ) |
| Other comprehensive income during the year | 78 |
| | 20 |
| | 98 |
|
| Balance at December 31, 2017 | $ | (4,404 | ) | | $ | (16 | ) | | $ | (4,420 | ) |
| Other comprehensive income (loss) during the year | (153 | ) | | 16 |
| | (137 | ) |
| Balance at December 31, 2018 | $ | (4,557 | ) | | $ | — |
| | $ | (4,557 | ) |
| Other comprehensive income during the year | $ | 34 |
| | $ | — |
| | $ | 34 |
|
| Balance at December 31, 2019 | $ | (4,523 | ) | | $ | — |
| | $ | (4,523 | ) |
Revenue Recognition
Revenue is recognized when a customer obtains control of promised goods or services, in an amount that reflects the consideration which the entity expects to receive in exchange for those goods or services. Pursuant to ASC Topic 606, Revenue from Contracts with Customers (ASC 606), a customer is a party that has contracted with an entity to obtain goods or services that are an output of the entity’s ordinary activities in exchange for consideration. To determine revenue recognition for arrangements that an entity determines are within the scope of ASC 606, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract, including whether they are distinct in the context of the contract; (iii) determine the transaction price, including the constraint on variable consideration; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the Company satisfies each performance obligation. The Company only applies the five-step model to contracts when it is probable that the entity will collect the consideration it is entitled to in exchange for the goods or services it transfers to the customer. At contract inception, once the contract is determined to be within the scope of ASC 606, the Company assesses the goods or services promised within each contract and determines those that are performance obligations, and assesses whether each promised good or service is distinct. If a promised good or service is not distinct, it is combined with other performance obligations. The Company then recognizes as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied. For example, certain performance obligations associated with SparkSwedish Orphan Biovitrum, or Sobi, Asklepios Biopharmaceutical, Inc., or AskBio, Sarepta Therapeutics, Inc., or Sarepta, and AskBioTakeda Pharmaceuticals USA, Inc., or Takeda, (see Note 12) will be satisfied over time, and revenue will be recognized using the output method, based on the proportion of actual deliveries to the total expected deliveries over the initial term.
Collaboration and GrantLicense Revenue: The Company currently generates its revenue through grants, collaboration and license agreements with strategic collaborators for the development and commercialization of product candidates. GrantsCollaboration and license agreements with customers are generally accounted for in accordance with ASC 606. The Company analyzes collaboration arrangements by first assessing whether they are within the scope of ASC Topic 808, Collaborative Arrangements (ASC 808), and evaluates whether such arrangements involve joint operating activities performed by parties that are both active participants in the activities and exposed to significant risks and rewards that are dependent on the commercial success of such activities. Collaboration agreements with customers that are not within the scope of ASC 808 are accounted for in accordance with ASC 606. To the extent the collaboration agreement is within the scope of ASC 808, the Company also assesses whether any aspects of the agreement are within the scope of other accounting literature (specifically ASC 606). The Company early adopted ASU No. 2018-18, Collaborative Arrangements (Topic 808): Clarifying the Interaction between Topic 808 and Topic 606, which provides guidance on evaluating certain transactions between collaborative arrangement participants. If the Company concludes that some or all aspects of the agreement are distinct and represent a transaction with a customer, the Company accounts for those aspects of the arrangement within the scope of ASC 606. The Company recognizes the shared costs incurred that are not within the scope of other accounting literature as a component of the related expense in the period incurred by analogy to ASC Topic 730, Research and Development (ASC 730), and records reimbursements from counterparties as an offset to the related costs. In determining the appropriate amount of revenue to be recognized as it fulfills its obligations under the agreements in accordance with ASC 606, the Company performs the five steps above. As part of the accounting for the arrangement, the Company must develop assumptions that require judgment to determine the stand-alone selling price for each performance obligation identified in the contract. The Company uses key assumptions to determine the stand-alone selling price, which may include market conditions, reimbursement rates for personnel costs, development timelines and probabilities of regulatory success.
The terms of the Company’s arrangements typically include one or more of the following: (i) up-frontupfront fees; (ii) milestone payments related to the achievement of development, regulatory, or commercial goals; (iii) royalties on net sales of licensed
products; (iv) reimbursements or cost-sharing of research and development (R&D) expenses; and (v) profit/loss sharing arising from co-promotion arrangements.
Licenses of Intellectual Property: If the license to the Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company recognizes revenues from non-refundable, up-frontupfront fees allocated to the license when the license is transferred to the customer and the customer is able to use and benefit from the license. If not distinct, the license is combined with other performance obligations in the contract. For licenses that are combined with other performance obligations, the Company assesses the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition. Optional
licenses are evaluated to determine if they are issued at a discount, and therefore, represent material rights and accounted for as separate performance obligations.
Milestone Payments: At the inception of each arrangement that includes developmental and regulatory milestone payments, the Company evaluates whether the achievement of each milestone specifically relates to the Company’s efforts to satisfy a performance obligation or transfer a distinct good or service within a performance obligation. If the achievement of a milestone is considered a direct result of the Company’s efforts to satisfy a performance obligation or transfer a distinct good or service and the receipt of the payment is based upon the achievement of the milestone, the associated milestone value is allocated to that distinct good or service. If the milestone payment is not specifically related to the Company’s effort to satisfy a performance obligation or transfer a distinct good or service, the amount is allocated to all performance obligations using the relative standalone selling price method. The Company also evaluates the milestone to determine whether they are considered probable of being reached and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the associated milestone value is included in the transaction price to be allocated, otherwise, such amounts are constrained and excluded from the transaction price. At the end of each subsequent reporting period, the Company re-evaluates the probability of achievement of such development milestones and any related constraint, and if necessary, adjusts its estimate of the transaction price. Any such adjustments to the transaction price are allocated to the performance obligations on the same basis as at contract inception. Amounts allocated to a satisfied performance obligation shall be recognized as revenue, or as a reduction of revenue, in the period in which the transaction price changes.
Manufacturing Supply Services: Arrangements that include a promise for future supply of drug substance or drug product for either clinical development or commercial supply at the customer’s discretion are evaluated to determine if they are distinct and optional. For optional services that are distinct, the Company assesses if they are priced at a discount, and therefore, provide a material right to the licensee to be accounted for as separate performance obligations.
Royalties: For arrangements that include sales-based royalties, including milestone payments based on the level of sales, and the license is deemed to be the predominant item to which the royalties relate, the Company will recognize revenue at the later of (i) when the related sales occur, or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied (or partially satisfied) in accordance with the royalty recognition constraint.
Research and Development Costs
Costs incurred in the research and development of the Company’s products are expensed as incurred. Research and development expenses include costs incurred in performing research and development activities, including salaries and benefits, stock-based compensation expenses, facilities cost, overhead costs, contract services, supplies and other outside costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity has been performed or when the goods have been received rather than when the payment is made.
Clinical Trial Costs
Clinical trial expenses are a significant component of research and development expenses, and the Company outsources a significant portion of these costs to third parties. Third party clinical trial expenses include patient costs, clinical research organization costs and costs for data management. The accrual for site and patient costs includes inputs such as estimates of patient enrollment, patient cycles incurred, clinical site activations, and other pass-through costs. Payments for these activities are based on the terms of the individual arrangements, which may differ from the pattern of costs incurred, and are reflected on the consolidated balance sheets as a prepaid asset or accrued clinical trial cost. These third party agreements are generally cancelable,cancellable, and related costs are recorded as research and development expenses as incurred. Non-refundable advance clinical payments for goods or services that will be used or rendered for future R&D activities are recorded as a prepaid asset and recognized as expense as the related goods are delivered or the related services are performed. The Company also records accruals for estimated ongoing clinical research and development costs. When evaluating the adequacy of the accrued liabilities, the Company analyzes progress of the studies, including the phase or completion of events, invoices received and contracted costs. Significant judgments and estimates may be made in determining the accrued balances at the end of any
reporting period. Actual results could differ from the estimates made by the Company. The historical clinical accrual estimates made by the Company have not been materially different from the actual costs.
In June 2020, the Company and Sobi entered into a license and development agreement, or the Sobi License. Pursuant to the Sobi License, clinical trial costs incurred to complete development of SEL-212, including but not limited to costs incurred while conducting and completing the Phase 3 DISSOLVE trials, will be reimbursed by Sobi. These costs, when reimbursed, will be recognized as revenue consistent with the revenue recognition methodology disclosed in Note 12. The reimbursable costs exclude any costs of additional development activities required that are related to ImmTOR and that are unrelated to SEL-212.
Income Taxes
The Company provides deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the Company’s financial statement carrying amounts and the tax basis of assets and liabilities using enacted tax rates expected to be in effect in the years in which the differences are expected to reverse. A valuation allowance is provided to reduce the deferred tax assets to the amount that will more-likely-than-not be realized.
The Company determines whether it is more likely than not that a tax position will be sustained upon examination. If it is not more-likely-than-not that a position will be sustained, none of the benefit attributable to the position is recognized. The tax benefit to be recognized for any tax position that meets the more‑likely‑than‑notmore-likely-than-not recognition threshold is calculated as the largest amount that is more than 50% likely of being realized upon resolution of the contingency. The Company accounts for interest and penalties related to uncertain tax positions as part of its provision for income taxes. To date, the Company has not incurred interest and penalties related to uncertain tax positions.
Warrants
The Company determines the accounting classification of warrants that are issued, as either liability or equity, by first assessing whether the warrants meet liability classification in accordance with ASC 480-10, Accounting for Certain Financial Instruments with Characteristics of both Liabilities and Equity, and then in accordance with ASC 815-40, Accounting for Derivative Financial Instruments Indexed to, and Potentially Settled in, a Company’s Own Stock. Under ASC 480, warrants are considered liability classified if the warrants are mandatorily redeemable, obligate the issuer to settle the warrants or the underlying shares by paying cash or other assets, or must or may require settlement by issuing variable number of shares.
If warrants do not meet liability classification under ASC 480-10, the Company assesses the requirements under ASC 815-40, which states that contracts that require or may require the issuer to settle the contract for cash are liabilities recorded at fair value, irrespective of the likelihood of the transaction occurring that triggers the net cash settlement feature. If the warrants do not require liability classification under ASC 815-40, in order to conclude equity classification, the Company assesses whether the warrants are indexed to its common stock and whether the warrants are classified as equity under ASC 815-40 or other applicable GAAP. After all relevant assessments are made, the Company concludes whether the warrants are classified as liability or equity. Liability classified warrants are required to be accounted for at fair value both on the date of issuance and on subsequent accounting period ending dates, with all changes in fair value after the issuance date recorded in the statements of operations as a gain or loss. Equity classified warrants are accounted for at fair value on the issuance date with no changes in fair value recognized after the issuance date.
Stock‑BasedStock-Based Compensation
The Company accounts for all stock‑basedstock-based compensation granted to employees and non‑employeesnon-employees using a fair value method. Stock‑basedStock-based compensation is measured at the grant date fair value and is recognized over the requisite service period of the awards, usually the vesting period, on a straight‑linestraight-line basis, net of estimated forfeitures. The Company reduces recorded stock‑based compensation for estimated forfeitures. To the extent that actual forfeitures differ from the Company’s estimates, the differences are recorded as a cumulative adjustment in the period the estimates were adjusted. Stock‑basedStock-based compensation expense recognized in the consolidated financial statements is based on awards that are ultimately expected to vest.
Net Loss Per Share
The Company has reported losses since inception and has computedcalculates basic net loss per share by dividing net loss attributable to common stockholders by the weighted average number of common shares and pre-funded warrantsoutstanding during the period. Diluted net income per share is calculated by dividing the net income attributable to common stockholders by the weighted-average number of common equivalent shares outstanding for the period. Theperiod, including any dilutive effect from outstanding stock options, restricted stock units, warrants to purchase common stock, and employee stock purchase plan stock using the treasury stock method. Given that the Company has computedrecorded a net loss for each of the periods presented, there is no difference between basic and diluted net loss per common share after considering all potentially dilutive common shares, including stock options, convertible preferred stock, and warrants outstanding during the period except wheresince the effect of including such securitiescommon stock equivalents would be antidilutive. Becauseanti-dilutive and are, therefore, excluded from the Company has reporteddiluted net losses since inception, these potential common shares have been anti‑dilutive and basic and diluted loss per share have been the same.calculation.
Contingent Liabilities
The Company accounts for its contingent liabilities in accordance with ASC No. 450, Contingencies. A provision is recorded when it is both probable that a liability has been incurred and the amount of the loss can be reasonably estimated. With respect to legal matters, provisions are reviewed and adjusted to reflect the impact of negotiations, estimated settlements, legal rulings, advice of legal counsel and other information and events pertaining to a particular matter. As of December 31, 2019 and December 31, 2018, the Company was not a party to any litigation that could have a material adverse effect on the Company’s business, financial position, results of operations or cash flows.
Leases
UnderThe Company accounts for its leases in accordance with ASC Topic 842, Leases (ASC 842), which was adopted January 1, 2019, the Companyand determines whether the arrangement is or contains a lease based on the unique facts and circumstances present. Most leases with a term greater than one year are recognized on the balance sheet as right-of-use assets, lease liabilities and, if applicable, long-term lease liabilities. The Company elected not to recognize leases with aan original term less than one year on its balance sheet. Operating lease right-of-useright-of-
use (ROU) assets and their corresponding lease liabilities are recorded based on the present value of lease payments over the expected remaining lease term. The interest rate implicit in lease contracts is typically not readily determinable. As a result, the Company utilizes its incremental borrowing rates, which are the rates incurred to borrow, on a collateralized basis over a similar term, an amount equal to the lease payments in a similar economic environment.
In accordance with the guidance in ASC 842, components of a lease should be split into three categories: lease components (e.g. land, building, etc.), non-lease components (e.g. common area maintenance, consumables, etc.), and non-components (e.g. property taxes, insurance, etc.). Then the fixed and in-substance fixed contract consideration (including any related to non-components) must be allocated based on fair values to the lease components and non-lease components. Although separation of lease and non-lease components is required, the Company elected the practical expedient to not separate lease and non-lease components. The lease component results in an operating right-of-use asset being recorded on the balance sheet and amortized on a straight-line basis as lease expense. Right-of-use assets and operating lease liabilities are remeasured upon certain modifications to leases using the present value of remaining lease payments and estimated incremental borrowing rate upon lease modification. See Note 8
The Company enters into lease agreements with terms generally ranging from 2-8 years. Some of the Company’s lease agreements include Company options to either extend and/or early terminate the lease, the costs of which are included in its operating lease liabilities to the extent that such options are reasonably certain of being exercised. Leases with renewal options allow the Company to extend the lease term typically between 1 and 5 years. When determining the lease term, renewal options reasonably certain of being exercised are included in the lease term. When determining if a renewal option is reasonably certain of being exercised, the Company considers several economic factors, including but not limited to, the significance of leasehold improvements incurred on the property, whether the asset is difficult to replace, underlying contractual obligations, or specific characteristics unique to that particular lease that would make it reasonably certain that the Company would exercise such option. Renewal and termination options were generally not included in the lease term for details.
Under prior guidance, rentthe Company’s existing operating leases. Leases with an initial term of 12 months or less are not recorded on the balance sheet; the Company recognizes lease expense and lease incentives from operatingfor these leases were recognized on a straight‑linestraight-line basis over the lease term. The difference between rent expense recognized and rental payments was recorded as deferred rent in the accompanying consolidated balance sheets.
Recent Accounting Pronouncements
Recently Adopted
In February 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update No. (“ASU”) 2016-02, Leases (Topic 842). ASU 2016-02 requires lessees to recognize most leases on their balance sheet as a right-of-use asset and a lease liability. Leases are classified as either operating or finance based on criteria similar to current lease accounting, with the classification affecting the pattern and classification of expense recognition in the statement of operations.
Subsequently, in July 2018,October 2020, the FASB issued ASU No. 2018-11,2020-10, Leases (Topic 842): TargetedCodification Improvements (ASU 2018-11), which includes certain amendmentsupdates various codification topics by clarifying or improving disclosure requirements to ASU 2016-02 intended to provide relief in implementing the new standard. Among these amendments is the option to not restate comparative periods presented in the financial statements. The Company has elected this transition approach, using a cumulative-effect adjustment on the effective date of the standard, with comparative periods presented in accordancealign with the existing guidance in ASC 840.
SEC’s regulations. The Company adopted the new standard as of the required effective date of January 1, 2019 resulting in the recognition of a net additional lease liability and right-of-use asset. The standard did not impact the Company's consolidated net loss. See Note 8 for details.
In November 2018, the FASB issued ASU No. 2018-18, Collaborative Arrangements (Topic 808): Clarifying the Interaction between Topic 808 and Topic 606 (ASU 2018-18), which provides guidance on whether certain transactions between collaborative arrangement participants should be accounted for with revenue under Topic 606. The new standard is effective for fiscal years beginning after December 15, 2019, including interim periods within those fiscal years with early adoption permitted. The Company early adopted the new standard effective September 30, 2019,2021, and there was no impact on its consolidated financial statements. See Note 14 for details.
Not Yet Adopted
In December 2019, the FASB issued ASU 2019-12, Income Taxes (Topic 740) - – Simplifying the Accounting for Income Taxes. ASU 2019-12 simplifies the accounting for income taxes by removing certain exceptions to the general principles in Topic 740. The Company adopted the new standard effective January 1, 2021, and there was no impact on its consolidated financial statements.
Not Yet Adopted
In May 2021, the FASB issued ASU 2021-04, Earnings Per Share (Topic 260), Debt – Modifications and Extinguishments (Subtopic 470-50), Compensation – Stock Compensation (Topic 718), and Derivatives and Hedging – Contracts in Entity’s Own Equity (Subtopic 815-40): Issuer's Accounting for Certain Modifications or Exchanges of Freestanding Equity-Classified Written Call Options. ASU 2021-04 provides guidance as to how entities should account for a modification of the terms or conditions or an exchange of a freestanding equity-classified written call option (i.e., a warrant) that remains equity-classified after modification or exchange as an exchange of the original instrument for a new instrument. An entity should apply the guidance provided in ASU 2021-04 prospectively to modifications or exchanges occurring on or after the effective date. This ASU isnew standard will be effective for public entitiesus for fiscal years beginning after December 15, 2020.2021 including interim periods within those fiscal years. The adoption of ASU 2021-04 is not expected to have an impact on the Company’s financial position or results of operations upon adoption.
In August 2020, the FASB issued ASU 2020-06, Debt – Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging – Contracts in Entity’s Own Equity (Subtopic 815-40). ASU 2020-06 simplifies the accounting for certain financial instruments with characteristics of liabilities and equity, including convertible instruments and contracts on an entity’s own equity. This new standard will be effective for smaller reporting companies for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. The Company is assessing the impact this standard will have on its consolidated financial statements and disclosures.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments-Credit Losses (Topic 326), Measurement of Credit Losses on Financial Instruments. Subsequently, in November 2018, the FASB issued ASU 2018-19, Codification Improvements to Topic 326, Financial Instruments-Credit Losses. ASU 2016-13 requires entities to measure all expected credit losses for most financial assets held at the reporting date based on an expected loss model which includes historical experience, current conditions, and reasonable and supportable forecasts. ASU 2016-13 also requires enhanced disclosures to help financial
statement users better understand significant estimates and judgments used in estimating credit losses. This ASU is effective for smaller reporting companies for fiscal years beginning after December 15, 2022, with early adoption permitted. The Companyadoption of ASU 2016-13 is assessingnot expected to have an impact on the impact this standard will have on its consolidatedCompany’s financial statements and disclosures.position or results of operations upon adoption.
In August 2018,The following table summarizes the FASB issued ASU No. 2018-13, Fair Value Measurement (Topic 820)marketable securities held as of December 31, 2021 (in thousands): Changes
| | | | | | | | | | | | | | | | | | | | | | | |
| Amortized
cost | | Unrealized gains | | Unrealized losses | | Fair
value |
| December 31, 2021 | | | | | | | |
| | | | | | | |
| Corporate bonds | $ | 2,007 | | | $ | — | | | $ | (1) | | | $ | 2,006 | |
| Commercial paper | 11,992 | | | — | | | — | | | 11,992 | |
| | | | | | | |
| Total | $ | 13,999 | | | $ | — | | | $ | (1) | | | $ | 13,998 | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
All marketable securities held at December 31, 2021 had maturities of less than 12 months when purchased and are classified as short-term marketable securities on the accompanying consolidated balance sheet. During the year ended December 31, 2021, there were no marketable securities adjusted for other than temporary declines in fair value. The Company does not intend to sell its investments and it is not more likely than not that the Disclosure Requirements for Fair Value Measurement (ASU 2018-13) which changes the fair value measurement disclosure requirements of ASC 820. EntitiesCompany will no longer be required to disclosesell the amountinvestments before recovery of and reasons for, transfers between Level 1 and Level 2their amortized cost bases, which may be maturity. As of December 31, 2021, the Company has a $2.0 million investment in Cyrus pursuant to the Cyrus Agreement. The Company’s maximum exposure to loss related to this variable interest entity is limited to the carrying value of the fair value hierarchy, the policy of timing of transfers between levels of the fair value hierarchy and the valuation processesinvestment. See Note 14 for Level 3 fair value measurements. This ASU is effective for public entities for fiscal years beginning after December 15, 2019, with early adoption permitted. The Company is assessing the impact this standard will have on its consolidated financial statements and disclosures.
3. Marketable Securitiesdetails.
As of December 31, 2019, and December 31, 2018,2020, the Company did not haveheld no marketable securities.securities or investments.
4. Net Loss Per Share
The Company has reported a net loss for the years ended December 31, 2019, 2018,2021, 2020 and 2017. For this reason basic and diluted net loss per share are2019. The Company used the same for all periods presented. Sincetreasury stock method to determine the shares underlying the 8,342,128 pre-funded warrants are issuable for little or no consideration, they are considered outstanding for both basic and diluted earnings per share.number of dilutive shares. The following table sets forth the computation of basic and diluted net loss per share (in thousands, except share and per‑shareper-share data):
|
| | | | | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| Numerator: | | | | | |
| Net loss attributable to common stockholders | $ | (55,350 | ) | | $ | (65,336 | ) |
| $ | (65,321 | ) |
| Denominator: | | | | | |
| Weighted‑average common shares and pre-funded warrants outstanding—basic and diluted | 45,548,511 |
| | 22,389,286 |
| | 20,425,050 |
|
| Net loss per share attributable to common stockholders —basic and diluted | $ | (1.22 | ) | | $ | (2.92 | ) | | $ | (3.20 | ) |
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Year Ended December 31, |
| | | | | | | | | 2021 | | 2020 | | 2019 |
| Numerator: | | | | | | | | | | | | |
| Net loss | | | | | | | | $ | (25,687) | | | $ | (68,876) | | | $ | (55,350) | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| Denominator: | | | | | | | | | | | | |
| Weighted-average common shares outstanding - basic and diluted | | | | | | | | 114,328,798 | | | 101,202,176 | | | 45,548,511 | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| Net loss per share: | | | | | | | | | | | | |
| Basic and diluted | | | | | | | | $ | (0.22) | | | $ | (0.68) | | | $ | (1.22) | |
| | | | | | | | | | | | |
AllThe following table represents the potential dilutive common shares have been excluded from the computation of the diluted net loss per share for all periods presented, as the effect would have been anti-dilutive. Potential dilutive common share equivalents consistanti-dilutive:
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Year Ended December 31, |
| | | | | | | | | 2021 | | 2020 | | 2019 |
| Options, RSUs and ESPP shares | | | | | | | | 11,492,002 | | | 7,909,583 | | | 7,002,527 | |
| Warrants to purchase common stock | | | | | | | | 10,735,980 | | | 12,378,016 | | | 23,084,120 | |
| Total | | | | | | | | 22,227,982 | | | 20,287,599 | | | 30,086,647 | |
Table of the following: |
| | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| Stock options to purchase common stock | 6,796,669 |
| | 4,093,979 |
| | 2,657,187 |
|
| Unvested restricted stock units | 181,250 |
| | 175,000 |
| | — |
|
| Stock warrants to purchase common stock | 23,084,120 |
| | 95,619 |
| | 176,432 |
|
| Total | 30,062,039 |
| | 4,364,598 |
| | 2,833,619 |
|
5. Fair Value Measurements
Assets and Liabilities Measured at Fair Value on a Recurring Basis
The following tables below present information about the Company’s financial assets and liabilities that are measured and carried at fair value as of December 31, 2019 and December 31, 2018, and indicate the level within the fair value hierarchy where each measurement is classified. Below is a summary of assets and liabilities measured at fair value on a recurring basis (in thousands): |
| | | | | | | | | | | | | | | |
| | December 31, 2019 |
| | Total | | (Level 1) | | (Level 2) | | (Level 3) |
| Assets: | | | | | | | |
| Money market funds | $ | 50,401 |
| | $ | 50,401 |
| | $ | — |
| | $ | — |
|
| Total | $ | 50,401 |
| | $ | 50,401 |
| | $ | — |
| | $ | — |
|
| | | | | | | | |
| Liabilities: | | | | | | | |
| Warrant liabilities | $ | 41,549 |
| | $ | — |
| | $ | — |
| | $ | 41,549 |
|
| Total | $ | 41,549 |
| | $ | — |
| | $ | — |
| | $ | 41,549 |
|
|
| | | | | | | | | | | | | | | |
| | December 31, 2018 |
| | Total | | (Level 1) | | (Level 2) | | (Level 3) |
| Assets: | | | | | | | |
| Money market funds | $ | 10,123 |
| | $ | 10,123 |
| | $ | — |
| | $ | — |
|
| Total | $ | 10,123 |
| | $ | 10,123 |
| | $ | — |
| | $ | — |
|
At eachas of December 31, 20192021 and 2020 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2021 |
| Total | | Level 1 | | Level 2 | | Level 3 |
| Assets: | | | | | | | |
| Money market funds (included in cash equivalents) | $ | 66,563 | | | $ | 66,563 | | | $ | — | | | $ | — | |
| Marketable securities: | | | | | | | |
| Corporate bonds | 2,006 | | | — | | | 2,006 | | | — | |
| Commercial paper | 11,992 | | | — | | | 11,992 | | | — | |
| | | | | | | |
| Total assets | $ | 80,561 | | | $ | 66,563 | | | $ | 13,998 | | | $ | — | |
| | | | | | | |
| Liabilities: | | | | | | | |
| Warrant liabilities | $ | 25,423 | | | $ | — | | | $ | — | | | $ | 25,423 | |
| Total liabilities | $ | 25,423 | | | $ | — | | | $ | — | | | $ | 25,423 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2020 |
| Total | | Level 1 | | Level 2 | | Level 3 |
| Assets: | | | | | | | |
| Money market funds (included in cash equivalents) | $ | 80,576 | | | $ | 80,576 | | | $ | — | | | $ | — | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| Total assets | $ | 80,576 | | | $ | 80,576 | | | $ | — | | | $ | — | |
| | | | | | | |
| Liabilities: | | | | | | | |
| Warrant liabilities | $ | 28,708 | | | $ | — | | | $ | — | | | $ | 28,708 | |
| Total liabilities | $ | 28,708 | | | $ | — | | | $ | — | | | $ | 28,708 | |
There were no transfers within the fair value hierarchy during the years ended December 31, 2018,2021 or 2020.
Cash, Cash Equivalents, and Restricted Cash
As of December 31, 2021 and 2020, the money market funds were classified as cash and cash equivalentequivalents on the accompanying consolidated balance sheetsheets as they mature within 90 days from the date of purchase.
Assumptions UsedAs of December 31, 2021, the Company had restricted cash balances relating to a secured letter of credit in Determining Fair Valueconnection with its lease for the Company’s headquarters (see Note 8 included elsewhere in this Annual Report). The Company’s consolidated statement of cash flows includes the following as of December 31, 2021, 2020 and 2019 (in thousands):
| | | | | | | | | | | | | | | | | | | | |
| | Year Ended December 31, |
| | | | 2021 | | 2020 | | 2019 |
| Cash and cash equivalents | | | | $ | 114,057 | | | $ | 138,685 | | | $ | 89,893 | |
| Short-term restricted cash | | | | — | | | — | | | 279 | |
| Long-term restricted cash | | | | 1,379 | | | 1,379 | | | 1,379 | |
| Total cash, cash equivalents, and restricted cash | | | | $ | 115,436 | | | $ | 140,064 | | | $ | 91,551 | |
Marketable Securities
As of December 31, 2021, marketable securities classified as Level 2 within the valuation hierarchy consist of corporate bonds and commercial paper. Marketable securities represent holdings of available-for-sale marketable debt securities in accordance with the Company’s investment policy. The Company estimates the fair value of these marketable securities by taking into consideration valuations that include market pricing based on real-time trade data for the same or similar securities, and other observable inputs. The amortized cost of available-for-sale debt securities is adjusted for amortization of premiums and accretion of discounts to the earliest call date for premiums or to maturity for discounts.
Loans Payable
At December 31, 2021, in light of the recent issuance of the Term A Loan under the 2020 Term Loan, the Company believes the carrying value approximates the fair value of the loan.
Common Warrants
In December 2019, wethe Company issued common warrants in connection with oura private placement of common shares. Pursuant to the terms of the common warrants, wethe Company could be required to settle the common warrants in cash in the event of certain acquisitions of the Company and, as a result, the common warrants are required to be measured at fair value and reported as a liability on the balance sheet. WeThe Company recorded the fair value of the common warrants upon issuance using the Black-Scholes valuation model and areis required to revalue the common warrants at each reporting date with any changes in fair value recorded on ourin the statement of operations.operations and comprehensive loss. The valuation of the common warrants is considered under Level 3 of the fair value hierarchy due to the need to use assumptions in the valuation that are both significant to the fair value measurement and unobservable. The significant unobservable inputs used in the fair value measurement of the warrant liabilities wereincluding the volatility rate and the estimated term of the warrants. Generally, increases (decreases) in the fair value of the underlying stock and estimated term would result in a directionally similar impact to the fair value measurement. The changechanges in the fair valuevalues of the Level 3 warrant liability isare reflected in the statement of operations and comprehensive loss for the yearyears ended December 31, 2021, 2020 and 2019.
The estimated fair value of warrants is determined using Level 3the following inputs inherent into the Black ScholesBlack-Scholes simulation valuation.valuation:
Estimated fair value of the underlying stock. The Company estimates the fair value of the common stock based on the closing stock price at the end of each reporting period.
Risk-free interest rate. The risk-free interest rate is based on the U.S. Treasury at the valuation date commensurate with the expected remaining life assumption.
Dividend rate. The dividend rate is based on the historical rate, which the Company anticipates will remain at zero.
Expected life. The expected life of the warrants is assumed to be equivalent to their remaining contractual term which expires on December 23, 2024.
Volatility. The Company estimates stock price volatility based on the Company’s historical volatility and the historical volatility of peer companies for a period of time commensurate with the expected remaining life of the warrants.
A summary of the Black ScholesBlack-Scholes pricing model assumptions used to record the fair value of the warrantswarrant liability is as follows:
|
| | | | | |
| | Issued on December 23, | | Year Ended December 31, |
| | 2019 | | 2019 |
| Risk-free interest rate | 1.75 | % | | 1.69 | % |
| Dividend yield | — |
| | — |
|
| Expected life (in years) | 5.00 |
| | 4.98 |
|
| Expected volatility | 87.29 | % | | 87.74 | % |
| | | | | | | | | | | |
| December 31, |
| | 2021 | | 2020 |
| Risk-free interest rate | 0.97 | % | | 0.36 | % |
| Dividend yield | — | | | — | |
| Expected life (in years) | 2.98 | | 3.98 |
| Expected volatility | 96.10 | % | | 98.63 | % |
Changes in Level 3 Liabilities Measured at Fair Value on a Recurring Basis
The following table reflects the change ina roll-forward of fair value for the Company’s Level 3 warrant liabilities (see Note 10), for the year ended December 31, 20192021 (in thousands):
| | | | | |
| Warrant liabilities |
| Fair value as of December 31, 2020 | $ | 28,708 | |
| Exercises | (5,624) | |
| Change in fair value | 2,339 | |
| Fair value as of December 31, 2021 | $ | 25,423 | |
| |
| |
| |
|
| | | |
| | Warrant liabilities |
| Fair value as of December 31, 2018 | $ | — |
|
| Warrants issued in connection with December 2019 private placement | 40,692 |
|
| Change in fair value | 857 |
|
| Fair value as of December 31, 2019 | $ | 41,549 |
|
6. Property and Equipment
Property and equipment consists of the following (in thousands):
| | | | December 31, | | December 31, | | December 31, |
| | 2019 | | 2018 | | 2021 | | 2020 |
| Laboratory equipment | $ | 4,836 |
| | $ | 5,379 |
| Laboratory equipment | $ | 5,134 | | | $ | 4,427 | |
| Computer equipment and software | 515 |
| | 561 |
| Computer equipment and software | 731 | | | 532 | |
| Leasehold improvements | 278 |
| | 278 |
| Leasehold improvements | 45 | | | 38 | |
| Furniture and fixtures | 237 |
| | 247 |
| Furniture and fixtures | 332 | | | 327 | |
| Office equipment | 135 |
| | 135 |
| Office equipment | 163 | | | 163 | |
| Construction in process | 2 |
| | 79 |
| Construction in process | 534 | | | 163 | |
| Total property and equipment | 6,003 |
|
| 6,679 |
| Total property and equipment | 6,939 | | | 5,650 | |
| Less accumulated depreciation | (4,781 | ) | | (4,552 | ) | Less accumulated depreciation | (4,797) | | | (4,255) | |
| Property and equipment, net | $ | 1,222 |
|
| $ | 2,127 |
| Property and equipment, net | $ | 2,142 | | | $ | 1,395 | |
Depreciation expense was $0.7$0.6 million, $1.0$0.6 million and $0.8$0.7 million for the years ended December 31, 2021, 2020 and
2019, 2018, and 2017, respectively. The Company recorded accelerated depreciation costs of $0.04 million in the reported property and equipment for the year ended December 31, 2019 relating to the upcoming new corporate headquarters move in 2020.
7. Accrued Expenses
Accrued expenses consist of the following (in thousands):
| | | | December 31, | | December 31, | | December 31, |
| | 2019 | | 2018 | | 2021 | | 2020 |
| Payroll and employee related expenses | $ | 2,235 |
| | $ | 2,497 |
| Payroll and employee related expenses | $ | 3,179 | | | $ | 3,049 | |
| Current portion of deferred rent and lease incentive | — |
| | 117 |
| |
| Collaboration and licensing | 1,050 |
| | 1,222 |
| Collaboration and licensing | — | | | 1,350 | |
| Accrued patent fees | 487 |
| | 736 |
| Accrued patent fees | 309 | | | 534 | |
| Accrued external research and development costs | 4,379 |
| | 5,344 |
| Accrued external research and development costs | 4,339 | | | 2,029 | |
| Accrued professional and consulting services | 446 |
| | 994 |
| Accrued professional and consulting services | 815 | | | 798 | |
| Accrued grant refund | — |
| | 175 |
| |
| Accrued interest | 82 |
| | 106 |
| Accrued interest | 170 | | | 170 | |
| Issuance costs, December financing | 4,381 |
| | — |
| |
| Other | 432 |
| | 509 |
| Other | 1,721 | | | 216 | |
| Accrued expenses | $ | 13,492 |
|
| $ | 11,700 |
| Accrued expenses | $ | 10,533 | | | $ | 8,146 | |
Other accrued expenses as of December 31, 2021 include a $0.9 million estimated liability for plaintiff’s litigation relating to the two lawsuits described further within Note 17.
8. Leases
On January 1, 2019, the Company adopted ASC 842 using the modified retrospective approach. The Company recorded operating lease assets (right-of-use assets) of $1.6 million and operating lease liabilities of $1.8 million and reversed a lease liability of $0.2 million related to straight-line rent and incentives. There was no impact to accumulated deficit upon adoption of ASC 842. The underlying assets of the Company’s leases are primarily office space. The Company determines if an arrangement qualifies as a lease at its inception.
As a practical expedient permitted under ASC 842, the Company has elected to account for the lease and non-lease components as a single lease component for all leases of which it is the lessee.65 Grove Street Lease payments, which may include lease and non-lease components, are included in the measurement of the Company’s lease liabilities to the extent that such payments are either fixed amounts or variable amounts that depend on a rate or index as stipulated in the lease contract.
When the Company cannot readily determine the rate implicit in the lease, the Company determines its incremental borrowing rate by using the rate of interest that it would have to pay to borrow on a collateralized basis over a similar term, an amount equal to the lease payments in a similar economic environment. On January 1, 2019, the discount rate used on existing operating leases at adoption, which had remaining lease terms of 15 months, was 10.0%. For new or renewed leases starting in 2020, the discount rate is determined based on the Company’s incremental borrowing rate adjusted for the lease term including any reasonably certain renewal periods.
The Company enters into lease agreements with terms generally ranging from 2-8 years. Some of the Company’s lease agreements include Company options to either extend and/or early terminate the lease, the costs of which are included in its operating lease liabilities to the extent that such options are reasonably certain of being exercised. Leases with renewal options allow the Company to extend the lease term typically between 1 and 5 years. When determining the lease term, renewal options
reasonably certain of being exercised are included in the lease term. When determining if a renewal option is reasonably certain of being exercised, the Company considers several economic factors, including but not limited to, the significance of leasehold improvements incurred on the property, whether the asset is difficult to replace, underlying contractual obligations, or specific characteristics unique to that particular lease that would make it reasonably certain that the Company would exercise such option. Renewal and termination options were generally not included in the lease term for the Company’s existing operating leases.
Leases with an initial term of 12 months or less are not recorded on the balance sheet; the Company recognizes lease expense for these leases on a straight-line basis over the lease term. For lease agreements entered into or reassessed after the adoption of ASC 842, lease and non-lease components are combined.
The Company has a non‑cancellable operating lease for its laboratory and office space located at 480 Arsenal Way, Watertown, Massachusetts ("Headquarters Lease"). As part of the Headquarters Lease agreement, the landlord provided the Company a tenant improvement allowance of up to $0.7 million, which the Company fully utilized during 2012. The leasehold improvements are capitalized as a component of property and equipment. In connection with the Headquarters Lease, the Company secured a letter of credit for $0.3 million which renews automatically each year and is classified in restricted cash. In August 2016, the Company signed an amendment to the Headquarters Lease, which extends the term through March 31, 2020.
In October 2017, the Company entered into a lease for approximately 5,100 square feet of additional office space located at 75 North Beacon Street, Watertown, Massachusetts (the “75 North Beacon Lease”). On January 11, 2019, the Company vacated 75 North Beacon Street, Watertown, MA and consolidated all employees at its corporate headquarters at 480 Arsenal Way, Watertown, MA. The right-of-use asset carrying amount of $0.2 million attributable to the 75 North Beacon Lease was written down to 0 during the first quarter of 2019.
The Company has a month‑to‑month facility agreement for its Moscow, Russia office. Rent expense is recognized as incurred.
As of December 31, 2018 and prior to the adoption of ASC 842, the aggregate future minimum lease payments related to leases are as follows (in thousands):
|
| | | |
| Year ending December 31, | |
| 2019 | 1,482 |
|
| 2020 | 375 |
|
| Total minimum lease payments | $ | 1,857 |
|
In July 2019, the Company entered into a lease for 25,078 square feet of laboratory and office space located at 65 Grove Street, Watertown, Massachusetts, (the “Newor the Headquarters Lease”). TheLease. As part of the Headquarters Lease, the Company estimates that it will incurincurred $0.8 million in non-reimbursable lessee-paid construction costs for lessor assets. None of these costs were incurred as of December 31, 2019.costs. The lease beginsbegan in March 2020, consistent with when the Company takestook control of the office space, and the expected lease term is 8 years, therefore the right-of-use asset and lease liability is not recordedyears. The discount rate of 8.9% was determined based on the balance sheet as of December 31, 2019. Rent payments are expected to occur in May 2020, and the base rentCompany’s incremental borrowing rate adjusted for the first year is $0.2 million per month. The total minimum rental commitments for the New Headquarters Lease are $15.8 million.lease term, including any reasonably certain renewal periods. In connection with the New Headquarters Lease, the Company secured a letter of credit from Silicon Valley Bank, or SVB, for $1.4 million, which renews automatically each year.
The Company's total minimum rental commitments for the New Headquarters Leaserecognized as long-term restricted cash, as of December 31, 2019 are as follows (in thousands):2021 and 2020, respectively, which automatically renews each year.
|
| | | |
| | December 31, |
| | 2019 |
| 2020 | $ | 1,191 |
|
| 2021 | 1,811 |
|
| 2022 | 1,865 |
|
| 2023 | 1,921 |
|
| 2024 | 1,979 |
|
| Thereafter | 7,027 |
|
| Total New Headquarters Lease commitment | $ | 15,794 |
|
The right-of-use asset and lease liabilityCompany has not been recordeda month-to-month facility agreement for its Moscow, Russia office. Rent expense is recognized as of December 31, 2019 as lease commencement will occur in 2020.
Rent expense for the years ended December 31, 2021, 2020 and 2019 2018 and 2017 was $2.1$2.9 million, $2.0$2.7 million, and $1.9$2.1 million, respectively.
For the yearyears ended December 31, 2021, 2020 and 2019, the components of lease costs were as follows (in thousands):
|
| | | |
| | Year Ended December 31, |
| | 2019 |
| Operating lease expense | $ | 1,365 |
|
| Variable lease expense | 828 |
|
| Short-term lease expense | 16 |
|
| Total lease expense | $ | 2,209 |
|
| | | | | | | | | | | | | | | | | |
| Year Ended
December 31, |
| 2021 | | 2020 | | 2019 |
| Operating lease cost | 2,023 | | | 2,096 | | | 1,365 | |
| Variable lease cost | 834 | | | 624 | | | 828 | |
| Short-term lease cost | 10 | | | 10 | | | 16 | |
| Total lease cost | $ | 2,867 | | | $ | 2,730 | | | $ | 2,209 | |
The maturity of the Company'sCompany’s operating lease liabilities as of December 31, 20192021 were as follows (in thousands):
|
| | | |
| | December 31, |
| Operating leases: | 2019 |
| 2020 | 375 |
|
| Total future minimum lease payments | $ | 375 |
|
| Less imputed interest | 3 |
|
| Total operating lease liabilities | $ | 372 |
|
| Included in the condensed consolidated balance sheet: | |
| Current operating lease liabilities | $ | 372 |
|
| Non-current operating lease liabilities | — |
|
| Total operating lease liabilities | $ | 372 |
|
| | | | | | | |
| December 31, | | |
| 2021 | | |
| 2022 | 1,866 | | | |
| 2023 | 1,922 | | | |
| 2024 | 1,980 | | | |
| 2025 | 2,039 | | | |
| 2026 | 2,101 | | | |
| Thereafter | 2,844 | | | |
| Total future minimum lease payments | 12,752 | | | |
| Less imputed interest | 3,105 | | | |
| Total operating lease liabilities | $ | 9,647 | | | |
The following information represents supplemental disclosure for the statement of cash flows related to operating leases were as follows (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2021 | | 2020 |
| Cash paid for amounts included in the measurement of lease liabilities: | $ | 1,812 | | | $ | 2,523 | |
| | | |
| | | |
| | | |
|
| | | |
| | Year Ended
December 31, |
| Operating leases: | 2019 |
| Cash paid for amounts included in the measurement of lease liabilities: | |
| Operating cash flows from operating leases | $ | 1,482 |
|
TheOther than the initial recording of the right-of-use asset and lease liability for the Headquarters Lease in 2020, which was non-cash, the changes in the Company’s right-of-use asset and lease liability for the yearyears ended December 31, 20192021 and 2020 are reflected in the changes in prepaid expenses, deposits and other assetsnon-cash lease expense and accrued expenses and other liabilities, respectively, in the consolidated statements of cash flows.
The following summarizes additional information related to operating leases:
| | | | | | | | | | | |
| December 31, |
| 2021 | | 2020 |
| Weighted-average remaining lease term | 6.4 years | | 7.4 years |
| Weighted-average discount rate | 8.9 | % | | 8.9 | % |
|
| | |
| December 31, |
Operating leases: | 2019 |
Weighted-average remaining lease term | 0.3 years |
|
Weighted-average discount rate | 10 | % |
9. Debt
20172020 Term Loan
On September 12, 2017,August 31, 2020, the Company entered into a term loan facility of up to $21.0$35.0 million, (the “2017 Term Loan”) with Silicon Valley Bank, a California corporation (“SVB”). The 2017or the 2020 Term Loan, isconsisting of term loans in an aggregate amount of $25.0 million, or the Term A Loan, and term loans in an aggregate amount of $10.0 million, or the Term B Loan, governed by a loan and security agreement, dated September 12, 2017,or the Loan Agreement, between the Company and Oxford Finance LLC, or Oxford, as Collateral Agent and a Lender, and SVB, (the “Loan Agreement”).as a Lender. The 2017 Term A Loan was funded in full on September 13, 2017 (the “Funding Date”).
OnAugust 31, 2020, or the Funding Date,Date. The second draw period expired on September 30, 2021 and the Term B Loan is no longer available to be drawn by the Company entered into a payoff letter with SVB, pursuant to which SVB utilized $10.0 million of the 2017 Term Loan to pay off all outstanding obligations under the 2015 Term Loan. The Company recognized a loss on extinguishment of debt in the amount of $0.7 million during the three months ended September 30, 2017.future.
The Company incurred less than $0.1 million in debt issuance costs in connection with the closing of the 2017 Term Loan. Debt issuance costs are presented in the consolidated balance sheet as a direct deduction from the associated liability and amortized to interest expense over the term of the related debt.
The 20172020 Term Loan will mature on FebruaryAugust 1, 2022.2025. Each advance under the 2017 Term Loan accrues interest at a floating per annum rate equal to one-halfthe greater of one percent above(a) 7.90%, and (b) the lesser of (x) the sum of (i) the prime rate (as publishedreported in the money rates section of The Wall Street Journal)Journal on the last business day of the month that immediately precedes the month in which the interest will accrue, and (ii) 4.65% and (y) 10.00%. The 2017 Term Loan providedprovides for interest-only payments on a monthly basis until August 31, 2019. On SeptemberApril 1, 2019, the Company began making2022. Thereafter, amortization payments on the Term Loan, which will continue to be payable monthly in equal installments of principal and variable interest to fully amortize the outstanding principal over the remaining term of the loan. The monthly interest isloan, subject to recalculation upon a change in the prime rate. The Company may prepay the 2017 Term Loan in full but not in part provided that the Company (i) provides five businessten days’ prior written notice to SVB,Collateral Agent, (ii) pays on the date of such prepayment for(A) all outstanding principal plus accrued and unpaid interest, 1% if prepaid afterand (B) a prepayment fee of between 3.0% and 1.0% of the second anniversary.aggregate original principal amount advanced by the lender depending on the
timing of the prepayment. Amounts outstanding during an event of default are payable upon SVB’s demand and shall accrue interest at an additional rate of 4.0%5.0% per annum of the past due amount outstanding. At the end of the loan term (whether at maturity, by prepayment in full or otherwise), the Company shall make a final payment to the lender in the amount of 9.0% of the aggregate original principal amount advanced by the lender. The final payment fee totaling $2.3 million is recorded as a loan discount.
The Term Loan is secured by a lien on substantially all of the assets of the Company, other than intellectual property, provided that such lien on substantially all assets includes any rights to payments and proceeds from the sale, licensing or disposition of intellectual property. The Company has also granted the Collateral Agent a negative pledge with respect to its intellectual property.
The Loan Agreement contains customary covenants and representations, including but not limited to financial reporting obligations and limitations on dividends, indebtedness, collateral, investments, distributions, transfers, mergers or acquisitions, taxes, corporate changes, deposit accounts, and subsidiaries. The Loan Agreement also contains other customary provisions, such as expense reimbursement, non-disclosure obligations as well as indemnification rights for the benefit of the Collateral Agent.
The events of default under the Loan Agreement include, but are not limited to, the Company’s failure to make any payments of principal or interest under the Loan Agreement or other transaction documents, the Company’s breach or default in the performance of any covenant under the Loan Agreement or other transaction documents, the occurrence of a material adverse effect,change, the Company making a false or misleading representation or warranty in any material respect under the Loan Agreement, the Company’s insolvency or bankruptcy, any attachment or judgment on the Company’s assets in excess of approximately $0.3at least $0.5 million, or the occurrence of any default under any agreement or obligation of the Company involving indebtedness in excess of approximately $0.3$0.5 million. If an event of default occurs, SVBthe Collateral Agent is entitled to take enforcement action, including acceleration of amounts due under the Loan Agreement.
The 2017 Term Loan is secured by a lien on substantially allCompany incurred $0.4 million in debt issuance costs in connection with the closing of the assets2020 Term Loan. Debt issuance costs are presented in the consolidated balance sheet as a direct deduction from the associated liability and amortized to interest expense over the term of the Company, other than intellectual property, provided that such lien on substantially all assets includes any rights to payments and proceeds from the sale, licensing or disposition of intellectual property. The Company has also granted SVB a negative pledge with respect to its intellectual property.
The 2017 Term Loan does not include any financial covenants. The 2017 Term Loan requires a final payment fee of 5% on the aggregate principal amounts borrowed upon repayment at maturity, on a prepayment date, or upon default. The final payment fee totaling $1.1 million is recorded as a loan discount. Under the 2017 Term Loan, the Company is not required to maintain a minimum cash balance. All deposits in operating, depository and securities accounts are required to be maintained with SVB in an amount equal to the lessor of (i) 100% of the Company's cash balance or (ii) 105% of the dollar amount of the then outstanding obligations. In addition, the 2017 Term Loan contains a subjective acceleration clause whereby in an event of default, an immediate acceleration of repayment occurs if there is a material impairment of the lenders’ lien or the value of the collateral, a material adverse change in the business condition or operations, or a material uncertainty exists that any portion of the loan may not be repaid.related debt.
The Company assessed all terms and features of the 20172020 Term Loan in order to identify any potential embedded features that would require bifurcation. As part of this analysis, the Company assessed the economic characteristics and risks of the 20172020 Term Loan, including any put, call, and callcontingent features. The Company determined that all featuresthe interest rate collar and prepayment call option did not require bifurcation; whereas the contingent put option and default (contingent) interest rate feature met bifurcation criteria resulting in immaterial amounts.
Warrants
On August 31, 2020, in connection with the 2020 Term A Loan, the Company issued warrants to the Lenders to purchase an aggregate of 196,850 shares of its common stock at an exercise price equal to $2.54 per share. In accordance with ASC 815-40, these warrants are classified as permanent equity in the accompanying consolidated balance sheets and will expire ten years from the date of issuance. The initial grant date fair value of the warrants was $0.4 million as determined by the Black-Scholes valuation model and recorded to stockholders' equity, with the SVB portion allocated to the reacquisition price of the 2017 Term Loan were clearly and closely associated with the debt host and did not require bifurcationOxford fair value portion as a derivative liability, orloan discount to the fair valueTerm A Loan.
Payoff
On the Funding Date, the Company entered into a payoff letter with SVB, pursuant to which the Company utilized $13.7 million of the embedded feature was immaterial2020 Term Loan to pay off all outstanding obligations under the Company's consolidated financial statements. Theprevious term loan, consisting of the principal payment, final prepayment and accrued interest. During the year ended December 31, 2020, the Company reassessesrecognized a loss on extinguishment of debt in the identified features on a quarterly basis to determine if they require bifurcation.amount of $0.5 million determined as the difference between the reacquisition price and carrying value at August 31, 2020.
As of December 31, 20192021 and December 31, 2018,2020, the outstanding principal balance under the 20172020 Term Loan was $18.2 million$25.0 million.
Total 2020 Term Loan and $21.0 million, respectively.unamortized debt discount balances as of December 31, 2021 are as follows (in thousands):
| | | | | |
| Face value | $ | 25,000 | |
| Venture debt termination fee | 2,250 | |
| Less: Debt discount | (1,616) | |
| Less: Current portion of loan payable | (5,961) | |
| Loan payable, net of current portion | $ | 19,673 | |
Future minimum principal
and interest payments on the
20172020 Term Loan as of December 31,
20192021 are as follows (in thousands):
|
| | | |
| 2020 | 9,084 |
|
| 2021 | 8,716 |
|
| 2022 | 2,461 |
|
| Total minimum debt payments | $ | 20,261 |
|
| Less: Amount representing interest | (1,011 | ) |
| Less: Debt discount and deferred charges | (345 | ) |
| Less: Current portion of loan payable | (18,905 | ) |
| Loan payable, net of current portion | $ | — |
|
All amounts due under the 2017 Term Loan have been classified as a current liability as of December 31, 2019 due to the considerations discussed in Note 1 and the assessment that the material adverse change clause under the 2017 Term Loan is not within the Company's control. The Company has not been notified of an event of default by SVB as of the date of the filing of this Annual Report on Form 10-K. | | | | | |
| Year ended: | |
| 2022 | $ | 5,488 | |
| 2023 | 7,317 | |
| 2024 | 7,317 | |
| 2025 | 4,878 | |
| Total minimum principal payments | $ | 25,000 | |
During the years ended December 31, 2019, 20182021, 2020 and 2017,2019, the Company recognized $1.5$2.8 million, $1.5$1.6 million and $1.2$1.5 million respectively of interest expense related to the 2020 and 2017 Term Loan.Loans.
10. Equity
Equity Financings
“At-the-Market” Offerings
2017 Sales Agreement and August 2020 Shelf Registration Statement
In August 2017, the Company entered into a sales agreement, or the 2017 Sales Agreement, with Jefferies LLC, as sales agent, to sell shares of its common stock with an aggregate value of up to $50.0 million in an “at the market offering.” On August 6, 2020, concurrent with the filing of the updated shelf registration statement, the Company entered into a sales agreement, or the 2020 Sales Agreement with Jefferies LLC, as sales agent, pursuant to which the Company may, from time to time, issue and sell common stock with an aggregate value of up to $50.0 million in an “at the market offering.” The 2017 Sales Agreement terminated pursuant to its terms in August 2020. On August 6, 2020, the Company filed an updated universal shelf registration statement on Form S-3 (Reg. No. 333-241692) with the SEC to sell an aggregate amount of up to $200.0 million of certain of its securities. The shelf registration statement was declared effective by the SEC on August 14, 2020. On October 8, 2021, the Company delivered notice to Jefferies LLC that the Company was terminating the 2020 Sales Agreement, with effect as of October 19, 2021.
2021 Sales Agreement
On October 25, 2021, the Company entered into a Sales Agreement, or the 2021 Sales Agreement, with SVB Leerink LLC to sell shares of the Company’s common stock, from time to time, through an “at the market” equity offering program under which SVB Leerink will act as sales agent. The shares of common stock sold pursuant to the 2021 Sales Agreement will be issued pursuant to the Company’s shelf registration statement on Form S-3 (File No. 333-241692), filed on August 6, 2020 with the Securities and Exchange Commission and related prospectus supplement, filed on October 25, 2021 with the SEC, for aggregate gross sales proceeds of up to $75.0 million.
During the year ended December 31, 2021, the Company sold 13,767,511 shares of its common stock pursuant to the 2021 and 2020 Sales Agreement for aggregate net proceeds of $51.9 million, after deducting commissions and other transaction costs. During the year ended December 31, 2020, the Company sold 1,069,486 shares of its common stock pursuant to the 2020 and 2017 Sales Agreements for aggregate net proceeds of $2.1 million, after deducting commissions and other transaction costs.
June 2020 Sobi Stock Purchase
On June 11, 2020, the Company entered into a stock purchase agreement with Sobi, pursuant to which the Company sold an aggregate of 5,416,390 shares of its common stock at a purchase price equal to $4.6156 per share, which represented 120% of the 10-day volume-weighted average price of the Company’s common stock prior to signing, for aggregate gross proceeds of $25.0 million, or the Sobi Private Placement. The closing of the Sobi Private Placement occurred on July 31, 2020.
In accordance with ASC 815, this forward sale treatment qualified as equity classification as the shares are not within the scope of ASC 480. The gross proceeds of $25.0 million were determined to include a premium to the fair value of the Company’s shares as of July 28, 2020 of approximately $14.5 million. As a result, such amount was included in the transaction price for revenue recognition of the Sobi License. See Note 12 for details.
Also on June 11, 2020, the Company entered into a registration rights agreement (as amended by that certain letter agreement, dated as of November 4, 2020, the “Sobi Registration Rights Agreement”) with Sobi, pursuant to which the Company agreed to prepare and file a registration statement with respect to the resale of the shares of common stock acquired in the Sobi Private Placement. The Company will be required to file this resale registration statement within 30 days following receipt by the Company of a written request from Sobi to file such resale registration statement, and to have the registration statement declared effective within ten (10) Business Days after the SEC informs the Company that no review of such resale registration statement will be made or that the SEC has no further comments on such resale registration statement.
December 2019 Financing
On December 18, 2019, the Company entered into a privatesecurities purchase agreement, (the "2019or the 2019 Purchase Agreement"),Agreement, with a group of institutional investors and closedcertain members of the Offering on December 23, 2019.Board of Directors. Pursuant to the 2019 Purchase Agreement, the Company sold an aggregate of 37,634,883 shares of its common stock at a purchase price of $1.46 per share, warrants to purchase an aggregate of 22,988,501 shares of common stock at a purchase price of $0.125 per share underlying each common warrant, and pre-funded warrants to purchase an aggregate of 8,342,128 shares of common stock at a purchase price of $1.46 per share, all with five year terms.terms, or the 2019 PIPE. The closing of the 2019 PIPE occurred on December 23, 2019. The exercise price of the pre-funded warrants wasis $0.0001 per share and the exercise price for the common warrants is $1.46 per share. In the event of a certain sale of the Company, the terms of the common warrants require us to make a payment to such common warrant holders based on a Black-Scholes valuation (using variables as specified in the warrants). This provision does not apply to the pre-funded warrants. Therefore, we arethe Company is required to account for the common warrants as liabilities and record them at fair value, while the pre-funded warrants met the criteria to be classified as permanent equity. We
The Company recorded the fair value of the common warrants of $40.7 million upon issuance using the Black-Scholes valuation model. The common warrants were revalued as of December 31, 2019 at $41.5 million; the change in fair value of $0.9 million was recorded in our statement of operations for the year ended December 31, 2019. Issuance costs were allocated between the equity component with an offset to additional paid-in capital and the liability component recorded as expense on a relative fair value basis. Total net proceeds from the equity offering was $65.6 million, after deducting transaction costs and commissions of $4.4 million thatmillion.
The common warrants were accrued atrevalued as of December 31, 2019.
Pursuant to2021 at $25.4 million. During the Registration Rights Agreement, the Company agreed to prepareyears ended December 31, 2021, 2020 and file a registration statement with the Securities and Exchange Commission (the “SEC”) within 45 days after the closing of the Offering for purposes of registering the resale of the Shares, shares of Common Stock issuable upon exercise of the Warrants, and any shares of Common Stock issued as a dividend or other distribution with respect to the Shares or shares of Common Stock issuable upon exercise of the Warrants. If the Company did not file such registration statement by the 45-day filing deadline, the Company would have been required to make pro-rata payments to each investor in an amount equal to 1% of the aggregate amount paid pursuant to the stock purchase agreement entered into by such investor for each 30-day period or pro-rata portion thereof following the filing deadline. The Company filed a registration statement on Form S-3 on January 29, 2020, which became effective on February 6, 2020, so no such payments were required.
The Company agreed, among other things, to indemnify the Investors, their officers, directors, members, employees and agents, successors and assigns under the registration statement from certain liabilities and to pay all fees and expenses (excluding any legal fees of the selling holder(s), and any underwriting discounts and selling commissions) incident to the Company’s obligations under the Registration Rights Agreement.
August 2019 Financing
On August 19, 2019, the Company sold 3,178,174 shares of its common stock pursuant torecorded a stock purchase agreement (the "2019 Private Placement") to individual investors, including certain executive officers and membersdecrease in the fair value of the boardwarrants of directors of the Company for aggregate net proceeds of approximately $5.7$2.3 million, after deducting transaction costs, at a purchase price
equal to $1.81 per share, which was equal to the most recent consolidated closing bid price on the Nasdaq Global Market on August 19, 2019. The shares$10.4 million, and $0.9 million, respectively, in the 2019 Private Placement were issued as “restricted securities” (as defined in Rule 144consolidated statements of the Securities Act)operations and carry no registration rights that require or permit the filing of any registration statement.
January 2019 Financing
On January 25, 2019, the Company completed an underwritten public offering (the “2019 Follow-On”) of 20,000,000 shares of its common stock at a public offering price of $1.50 per share. On January 29, 2019, an additional 2,188,706 shares were sold at a public offering price of $1.50 per share pursuant to the underwriters’ exercise of an over allotment option. The total net proceeds from the offering were $30.9 million, after deducting underwriting discounts, transaction costs and commissions.
2019 “At-the-Market” Offerings
Concurrent with the filing of the shelf registration statement, the Company entered into a sales agreement (the “Sales Agreement”) with Jefferies LLC, as sales agent, pursuant to which the Company may, from time to time, issue and sell common stock with an aggregate value of up to $50 million in an "at-the-market" offering.
Sales of common stock, if any, pursuant to the Sales Agreement, may be made in sales deemed to be an “at the market offering” as defined in Rule 415(a) of the Securities Act, including sales made directly through the Nasdaq Global Market or on any other existing trading market for the Company’s common stock. The Company intends to use the proceeds from the offering for working capital and other general corporate purposes. The Company may suspend or terminate the Sales Agreement at any time.
During the year ended December 31, 2019, the Company sold 615,453 shares of its common stock pursuant to the Sales Agreement at an average price of approximately $1.84 per share for aggregate net proceeds of $1.0 million, after deducting commissions and other transaction costs.
August 2017 Shelf Registration Statement
On August 10, 2017, the Company filed a universal shelf registration statement on Form S-3 with the SEC to sell an aggregate amount of up to $200.0 million of certain of our securities. The shelf registration statement was declared effective by the SEC on August 28, 2017.comprehensive loss.
June 2017 Financing
OnIn June 26, 2017, the Company entered into a securities purchase agreement, (the "Institutionalor the Institutional Purchase Agreement")Agreement with a select group of institutional investors, (the “Institutional Investors”)or the Institutional Investors and a securities purchase agreement with Timothy A. Springer, Ph.D., a member of the board of directors, (the "Springeror the Springer Purchase Agreement")Agreement, for a private placement of the Company'sCompany’s securities, (the "2017 PIPE"). The closing ofor the 2017 PIPE occurred on June 27, 2017.
PIPE. Pursuant to the Institutional Purchase Agreement, the Company sold an aggregate of 2,750,000 shares of its common stock at a purchase price equal to $16.00 per share. Pursuant to the Springer Purchase Agreement, the Company sold to Dr. Springer an aggregate of 338,791 shares of common stock at a purchase price equal to $17.71 per share, which was equal to the most recent consolidated closing bid price on the Nasdaq GlobalStock Market on June 23, 2017, and warrants to purchase up to 79,130 shares of common stock, (“or the Warrant Shares”),Shares, exercisable at $17.71 per Warrant Share, and with a term of five years. The purchase price for each warrant was equal to $0.125 for each Warrant Share, consistent with Nasdaq GlobalStock Market requirements for an “at the market” offering. Under the terms of the Common Stock Purchase Warrant, the warrants can be settled in unregistered shares. The Warrant Shares qualify for equity classification. The fair value of the allocated proceeds was determined on the relative fair value basis. After deducting for placement agent fees and offering expenses, the aggregate net proceeds from the 2017 PIPE were approximately $47.1 million.
On June 27, 2017, in connection withWarrants
During the 2017 PIPE, the Company entered intoyear ended December 31, 2021, warrant holders exercised 1,642,036 common warrants on a Registration Rights Agreement (the “Registration Rights Agreement”) with the Institutional Investorscashless basis and Dr. Springer. Pursuant to the Registration Rights Agreement, the Company agreed to prepare and file a registration statement with the SEC within 20 days after the closing of the 2017 PIPE for purposes of registering the resale of thereceived 1,076,669 shares of common stock issued and sold in the 2017 PIPE (the “Shares”), the Warrant Shares, and any shares of common stock issued as a dividend or other distribution with respect to the Shares or Warrant Shares. The 2017 PIPE registration statement was declared effective by the SEC on July 21, 2017.stock.
The Company agreed to indemnify the Institutional Investors and Dr. Springer, their officers, directors, members, employees and agents, successors and assigns under the registration statement from certain liabilities and to pay all fees and expenses (excluding any legal fees of the selling holder(s), and any underwriting discounts and selling commissions) incident to the Company’s obligations under the Registration Rights Agreement.
Warrants
The following table summarizes warrant activity for the years ended December 31, 2019, 2018 and 2017 as follows:
|
| | | | | | | | | | |
| | Number of Warrants | | |
| | Equity classified | Liability classified | Total | | Weighted average exercise price |
| Outstanding at December 31, 2017 | 176,432 |
| — |
| 176,432 |
| | $ | 17.32 |
|
| Issuance | — |
| — |
| — |
| | — |
|
| Exercises | — |
| — |
| — |
| | — |
|
| Expirations/ cancellations | (80,813 | ) | — |
| (80,813 | ) | | 17.55 |
|
| Outstanding at December 31, 2018 | 95,619 |
| — |
| 95,619 |
| | 17.12 |
|
| Issuance | 8,342,128 |
| 22,988,501 |
| 31,330,629 |
| | 1.07 |
|
| Exercises | — |
| — |
| — |
| | — |
|
| Expirations/ cancellations | — |
| — |
| — |
| | — |
|
| Outstanding at December 31, 2019 | 8,437,747 |
| 22,988,501 |
| 31,426,248 |
| | $ | 1.12 |
|
| | | | | | | | | | | | | | | | | | | | | | | |
| Number of Warrants | | |
| Equity
classified | | Liability classified | | Total | | Weighted average
exercise price |
| Outstanding at December 31, 2019 | 8,437,747 | | | 22,988,501 | | | 31,426,248 | | | $ | 1.12 | |
| Exercises | (8,342,128) | | | (10,902,954) | | | (19,245,082) | | | 0.83 | |
| Issuance | 196,850 | | | — | | | 196,850 | | | 2.54 | |
| Outstanding at December 31, 2020 | 292,469 | | | 12,085,547 | | | 12,378,016 | | | $ | 1.60 | |
| Exercises | — | | | (1,642,036) | | | (1,642,036) | | | 1.46 | |
| | | | | | | |
| Outstanding at December 31, 2021 | 292,469 | | | 10,443,511 | | | 10,735,980 | | | $ | 1.62 | |
Common Stock
As of December 31, 2019,2021, the Company had 200,000,000 shares of common stock authorized for issuance, $0.0001 par value per share, with 86,325,547123,622,965 shares issued and outstanding. The voting, dividend and liquidation rights of the common stockholders are subject to and qualified by the rights, powers and preferences of the preferred stock. The common stock has the following characteristics:
Voting
The common stockholders are entitled to 1 vote for each share of common stock held with respect to all matters voted on by the stockholders of the Company.
Dividends
The common stockholders are entitled to receive dividends, if and when declared by the Board of Directors. Through December 31, 2019, 02021, no dividends have been declared or paid on common stock.
Liquidation
Upon liquidation of the Company, the common stockholders are entitled to receive all assets of the Company available for distribution to such stockholders.
Reserved Shares
The Company has authorized shares of common stock for future issuance as follows: |
| | | | | |
| | Period ending |
| | December 31, 2019 | | December 31, 2018 |
| Exercise of common and pre-funded warrants | 31,426,248 |
| | 95,619 |
|
| Shares available for future stock incentive awards | 1,765,018 |
| | 1,586,925 |
|
| Unvested restricted stock units | 181,250 |
| | 175,000 |
|
| Outstanding common stock options | 6,796,669 |
| | 4,093,979 |
|
| Total | 40,169,185 |
|
| 5,951,523 |
|
| | | | | | | | | | | |
| | As of |
| | December 31, 2021 | | December 31, 2020 |
| Exercise of warrants | 10,735,980 | | | 12,378,016 | |
| Shares available for future stock incentive awards | 6,039,564 | | | 4,916,374 | |
| | | |
| Unvested restricted stock units | 394,450 | | | 87,500 | |
| Outstanding common stock options | 11,039,873 | | | 7,775,249 | |
| Total | 28,209,867 | | | 25,157,139 | |
11. Stock Incentive Plans
Stock Options
The Company maintains the 2008 Stock Incentive Plan, (the “2008 Plan”)or the 2008 Plan, for employees, consultants, advisors, and directors. The 2008 Plan provided for the granting of incentive and non‑qualifiednon-qualified stock option and restricted stock awards as determined by the Board. At inception of
In June 2016, the 2008Company’s stockholders approved the 2016 Incentive Award Plan, a total of 2,213,412or the 2016 Plan, which authorized 1,210,256 shares of common stock were authorized for grantsfuture issuance under the 2008 Plan. The2016 Plan and the Company ceased granting awards under the 2008 Plan uponPlan. Upon the effectivenesseffective date of the 2016 Plan, (as defined below); however, awards issued under the 2008 Plan remain subject to the terms of the 2008 Plan and the applicable 2008 Plan agreement. Shares subject to awards that werePlan. Awards granted under the 2008 Plan and that expire, lapse or terminate following the
effectiveness of the 2016 Plan become available under the 2016 Plan as shares available for future grants. All unvested stock options granted under
Additionally, pursuant to the 2008 Plan may be exercised into restricted stock subject to forfeiture upon termination prior to vesting.
On June 7, 2016, the Company’s stockholders approved the 2016 Incentive Award Plan (the “2016 Plan”), which became effective June 21, 2016. The 2016 Plan provides for the granting of incentive and non‑qualified stock option, restricted stock and other stock and cash-based awards as determined by the Board. Shares subject to awards that are granted under the 2016 Plan and that expire, lapse or terminate are available for future grants under the 2016 Plan. At inceptionterms of the 2016 Plan, a total of 1,210,256 shares ofthe Board is authorized to grant awards with respect to common stock, wereand may delegate to a committee of one or more members of the Board or executive officers of the Company the authority to grant options and restricted stock units. On December 9, 2020, the Board established a Stock Option Committee authorized for future issuance underto grant awards to certain employees and consultants subject to conditions and limitations within the 2016 Plan. The number of shares of common stock that may be issued under the 2016 Plan automatically increases on the first day of each calendar year, beginning in 2017In January 2021 and ending in and including 2026, by an amount equal to the lesser of: (i) 4% of the number of shares of the Company’s common stock outstanding on the last day of the applicable preceding calendar year and (ii) such smaller number of shares as is determined by the Board. During the year ended December 31, 2019 and 2018,2020, the number of shares of common stock that may be issued under the 2016 Plan was increased by 898,871 shares4,322,850 and 893,7303,453,022 shares, respectively. As of December 31, 2019, 267,6122021, 1,925,537 shares remain available for future issuance under the 2016 Plan.
The 2008 Plan and 2016 Plan provide thatIn September 2018, the exercise price of incentive stock options cannot be less than 100% of the fair market value of the Company's common stock on the grant date for participants who own 10% or less of the total combined voting power of the Company, and not less than 110% for participants who own more than 10% of the Company’s voting power. Options and restricted stock awards granted under the 2008 Plan and 2016 Plan vest over periods as determined by the Board, which are generally four years and, for options, with terms that generally expire ten years from the grant date.
The Company’s 2018 Employment Inducement Incentive Award Plan, (the “Inducementor the 2018 Inducement Incentive Award Plan”), whichPlan was adopted by the Board on September 25, 2018 without stockholder approval pursuant to Rule 5635(c)(4) of the Nasdaq Stock Market LLC listing rules, ("Rule 5635(c)(4)"), provides for the grant of equity-based awards in the form of non-qualified stock options, stock appreciation rights, restricted stock awards, restricted stock unit awards and other stock or cash based awards. In accordance with Rule 5635(c)(4), awards under the Inducement Incentive Award Plan may only be made to a newly hired employee who has not previously been a member of the Board, or an employee who is being rehired following a bona fide period of non-employment by the Company, as a material inducement to the employee’s entering into employment with the Company. The Company reservedwhich authorized 1,175,000 shares of its common stock for issuance under the Inducement Incentive Award Plan. Onissuance. In March 25, 2019, the Board approved the amendment and restatement of the 2018 Inducement Incentive Award Plan to reserve an additional 2,000,000 shares of the Company’s common stock for issuance thereunder. As of December 31, 2019,2021, there are 750,0001,591,661 shares available for future grant under the 2018 Inducement Incentive Award Plan.
Stock-Based Compensation Expense
Stock-based compensation expense only on awards that are expected to vest.by classification included within the consolidated statements of operations and comprehensive loss was as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Year Ended December 31, |
| | | | | | | | | 2021 | | 2020 | | 2019 |
| Research and development | | | | | | | | $ | 3,204 | | | $ | 2,271 | | | $ | 2,079 | |
| General and administrative | | | | | | | | 4,516 | | | 3,151 | | | 3,082 | |
| Total stock-based compensation expense | | | | | | | | $ | 7,720 | | | $ | 5,422 | | | $ | 5,161 | |
Stock Options
The estimated grant date fair values of employee stock option awards granted under the 2016 Plan and the 2018 Inducement Incentive Award Plan were calculated using the Black-Scholes option pricing model, based on the following weighted-average assumptions:
| | | | Year Ended December 31, | | | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 | | | 2021 | | 2020 | | 2019 |
| Risk-free interest rate | 1.86 | % | | 2.85 | % | | 2.03 | % | Risk-free interest rate | | 0.79 | % | | 1.11 | % | | 1.86 | % |
| Dividend yield | — |
| | — |
| | — |
| Dividend yield | | — | | | — | | | — | |
| Expected term | 5.94 |
| | 6.06 |
| | 5.90 |
| Expected term | | 6.03 | | 6.04 | | 5.94 |
| Expected volatility | 87.66 | % | | 85.17 | % | | 84.52 | % | Expected volatility | | 95.04 | % | | 91.00 | % | | 87.66 | % |
| Weighted-average fair value of common stock | $ | 2.01 |
| | $ | 8.45 |
| | $ | 15.32 |
| Weighted-average fair value of common stock | | $ | 3.58 | | | $ | 2.49 | | | $ | 2.01 | |
The weighted average grant date fair value of stock options granted to employees during the years ended December 31, 2021, 2020 and 2019 2018was $2.73, $1.86, and 2017 was $1.47 $6.17, and $10.97 respectively. The aggregate intrinsic value of stock options exercised during the years ended December 31, 2019, 2018 and 2017 was $0.1 million, $0.6 million, and $2.6 million respectively.
As of December 31, 20192021 and December 31, 2018,2020, total unrecognized compensation expense related to unvested employee stock options was $9.8$11.5 million and $9.8$8.0 million, respectively, which is expected to be recognized over a weighted average period of 2.52.7 years and 3.1 years, respectively.
Effective November 20, 2019, the Company amended the terms of the options granted to a non-employee director in connection with his resignation from the board of directors, whereby he was entitled to receive immediate vesting of his outstanding, unvested stock options (141,328 shares), provided that the options will become exercisable in accordance with their original vesting schedules irrespective of his termination of service, and an extension of the right to exercise each of his vested stock options (234,772 shares) until the final expiration date of the applicable option, resulting in a modification. The subsequent stock-based compensation amount recognized as of December 31, 2019 was $0.1 million.
The estimated grant date fair values of non-employee stock option awards granted under the 2016 Plan were calculated using the Black-Scholes option pricing model, based on the following weighted-average assumptions: |
| | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| Risk-free interest rate | 1.92 | % | | 2.77 | % | | — | % |
| Dividend yield | — |
| | — |
| | — |
|
| Expected life (in years) | 5.33 |
| | 5.81 |
| | 0.00 |
|
| Expected volatility | 88.60 | % | | 85.86 | % | | — | % |
The weighted average grant date fair value of stock options granted to non-employees during the years ended December 31, 2019 and 2018 was $1.32 and $6.78, respectively.
As of December 31, 2019 and 2018 total unrecognized compensation expense related to unvested non‑employee stock options was less than $0.1 million and $1.1 million, respectively, which is expected to be recognized over a weighted average period of 0.4 years and 2.22.3 years, respectively.
The following table summarizes the stock option activity under the 2008 Plan, 2016 Plan, and 2018 Inducement Incentive Award Plan:
|
| | | | | | | | | | | | |
| | | | | | Weighted‑average | | |
| | | | | | remaining | | Aggregate |
| | Number of | | Weighted-average | | contractual term | | intrinsic value |
| | options | | exercise price ($) | | (in years) | | (in thousands) |
| Employee awards | | | | | | | |
| Outstanding at December 31, 2018 | 3,681,575 |
| | $ | 9.49 |
| | 7.77 | | $ | 300 |
|
| Granted | 3,876,255 |
| | $ | 2.01 |
| | | | |
| Exercised | (125,600 | ) | | $ | 1.20 |
| | | | |
| Forfeited | (1,108,634 | ) | | $ | 10.38 |
| | | | |
| Outstanding at December 31, 2019 | 6,323,596 |
| | $ | 4.91 |
| | 8.71 | | $ | 1,716 |
|
| | | | | | | | |
| Vested at December 31, 2019 | 1,485,228 |
| | $ | 10.02 |
| | 6.73 | | $ | 90 |
|
| Vested and expected to vest at December 31, 2019 | 5,843,053 |
| | $ | 5.10 |
| | 8.65 | | $ | 1,500 |
|
| | | | | | | | |
| Non‑employee awards | | | | | | | |
| Outstanding at December 31, 2018 | 412,404 |
| | $ | 6.44 |
| | 6.42 | | $ | 28 |
|
| Granted | 73,489 |
| | $ | 1.86 |
| | | | |
| Exercised | — |
| | $ | — |
| | | | |
| Forfeited | (12,820 | ) | | $ | 0.47 |
| | | | |
| Outstanding at December 31, 2019 | 473,073 |
| | $ | 5.89 |
| | 6.23 | | $ | 38 |
|
| | | | | | | | |
| Vested at December 31, 2019 | 330,528 |
| | $ | 5.61 |
| | 5.06 | | $ | 21 |
|
| Vested and expected to vest at December 31, 2019 | 473,073 |
| | $ | 5.89 |
| | 6.23 | | $ | 38 |
|
| | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Weighted-average | | |
| | | | | | remaining | | Aggregate |
| | Number of | | Weighted-average | | contractual term | | intrinsic value |
| | options | | exercise price ($) | | (in years) | | (in thousands) |
| Employees | | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| Outstanding at December 31, 2020 | 7,302,176 | | | $ | 3.98 | | | 8.43 | | $ | 4,456 | |
| Granted | 4,625,611 | | | $ | 3.58 | | | | | |
| Exercised | (398,754) | | | $ | 1.72 | | | | | |
| Forfeited | (912,233) | | | $ | 2.84 | | | | | |
| Outstanding at December 31, 2021 | 10,616,800 | | | $ | 3.99 | | | 8.19 | | $ | 4,982 | |
| | | | | | | |
| Vested at December 31, 2021 | 4,459,309 | | | $ | 4.72 | | | 7.23 | | $ | 3,149 | |
| Vested and expected to vest at December 31, 2021 | 9,917,248 | | | $ | 4.03 | | | 8.13 | | $ | 4,796 | |
| | | | | | | |
| Non-employee consultants | | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| Outstanding at December 31, 2020 | 473,073 | | | $ | 5.89 | | | 5.23 | | $ | 86 | |
| | | | | | | |
| Exercised | (50,000) | | | $ | 2.04 | | | | | |
| | | | | | | |
| Outstanding at December 31, 2021 | 423,073 | | | $ | 6.34 | | | 3.85 | | $ | 42 | |
| | | | | | | |
| Vested at December 31, 2021 | 423,073 | | | $ | 6.34 | | | 3.85 | | $ | 42 | |
| Vested and expected to vest at December 31, 2021 | 423,073 | | | $ | 6.34 | | | 3.85 | | $ | 42 | |
Restricted Stock Units
During the first quarter of 2019,year ended December 31, 2021, the Company awarded 100,000granted 407,700 restricted stock units under the Inducement Incentive Award Plan, of which 50,000 were determined to be granted and 50,000 were reserved for issuance consistentawards with ASC Topic 718, Compensation-Stock Compensation (ASC 718). The 50,000 granted restricted stock units had a weighted average fair value of $2.29$3.11 per share based on the closing price of the Company’s common stock on the date of grant.grant to employees under the 2016 Plan, which will vest over a four year term. Forfeitures are estimated at the time of grant and are adjusted, if necessary, in subsequent periods if actual forfeitures differ from those estimates. The Company has estimated a forfeiture rate of 10% for restricted stock awards to employees based on historical attrition trends. In addition, the Company awarded 197,500 restricted stock units to executives under the 2016 Plan. These restricted stock units were valued at approximately $0.1 million, and will vestvested in two equal installments on the date andates applicable performance condition isconditions were achieved on or prior to December
31, 2020. Ifduring the performance condition is not satisfied on or prior toyear ended December 31, 2020, the restricted stock units will be forfeited for no consideration.
The 50,000 reserved restricted stock units did not have defined performance criteria until August 6, 2019, at which time, they were deemed both granted and vested. These restricted stock units had a fair value of $1.65 per share. These restricted stock units were valued at approximately $0.1 million and fully expensed as of December 31, 2019.2021.
Unrecognized compensation expense for theall restricted stock units was $0.8$0.9 million as of December 31, 2019,2021, which is expected to be recognized over a weighted average period of 2.82.7 years.
The following table summarizes the status of the Company’s restricted stock units:units under the 2016 Plan and 2018 Inducement Incentive Award Plan:
| | | | | | | | | | | |
| | Number of shares | | Weighted average
grant date
fair value ($) |
| Unvested at December 31, 2020 | 87,500 | | | $ | 6.03 | |
| Granted | 605,200 | | | 2.10 | |
| Vested | (201,250) | | | 1.31 | |
| | | |
| Forfeited | (97,000) | | | 1.76 | |
| Unvested at December 31, 2021 | 394,450 | | | $ | 3.45 | |
|
| | | | | | |
| | Number of shares | | Weighted average fair value ($) |
| Unvested at December 31, 2018 | 175,000 |
| | $ | 6.03 |
|
| Granted | 100,000 |
| | 1.97 |
|
| Vested | 93,750 |
| | 1.65 |
|
| Forfeited | — |
| | — |
|
| Unvested at December 31, 2019 | 181,250 |
| | $ | 5.00 |
|
Employee Stock Purchase Plan
OnIn June 7, 2016, the Company’s stockholdersCompany approved the 2016 Employee Stock Purchase Plan, (the “ESPP”), which became effective June 21, 2016. The ESPP is intended to qualify as an "employee stock purchase plan" under Section 423 of the Internal Revenue Code of 1986 with the purpose of providing employees with an opportunity to purchase the Company's common stock through accumulated payroll deductions.
Underor the ESPP, the Company has set 2 six-month offering periods during each calendar year, one beginning March 1st and the other beginning September 1st of each calendar year, during which employees may elect to have up to 25% of their eligible compensation deducted on each payday on an after-tax basis for use in purchasing the Company's common stock on the last trading day of each offering period, subject to limits imposed by the Internal Revenue Code. The purchase price of the shares may not be less than 85% of the fair market value on the first or last trading day of the offering period, whichever is lower. The first ESPP offering period began on March 1, 2017.
At inception of the ESPP, a total ofauthorized 173,076 shares of common stock were authorized and reserved for future issuance under the ESPP. The number of shares of common stock that may be issued under the ESPP will automatically increase on the first day of each calendar year, beginning in 2017to participating employees. In January 2021 and ending in and including 2026, by an amount equal to the lesser of: (i) 1% of the number of shares of the Company’s common stock outstanding on the last day of the applicable preceding calendar year and (ii) such smaller number of shares as is determined by the Company’s Board of Directors. During the year ended December 31, 2019 and 2018,2020, the number of shares of common stock that may be issuedauthorized for issuance under the ESPP was increased by 224,7171,080,711 shares and 223,432863,254 shares, respectively. During the year ended December 31, 2019,2021, the Company issued 17,20558,794 shares of common stock under the ESPP. As of December 31, 2019, 747,4062021, 2,522,366 shares remain available for future issuance under the ESPP.
For each of the years ended December 31, 20192021 and 2018,2020, the Company recognized less than $0.1 million of stock-based compensation expense under the ESPP.ESPP, respectively.
The Company recorded stock-based compensation expense related to stock option awards, restricted stock units and the ESPP in the following expense categories of its consolidated statements of operations and comprehensive loss (in thousands): |
| | | | | | | | | | | |
| | Year Ended December 31, |
| | 2019 |
| 2018 |
| 2017 |
| Research and development | $ | 2,079 |
| | $ | 2,453 |
| | $ | 1,779 |
|
| General and administrative | 3,082 |
| | 3,261 |
| | 2,302 |
|
| Total stock-based compensation expense | $ | 5,161 |
| | $ | 5,714 |
| | $ | 4,081 |
|
12. Revenue Arrangements
Takeda Pharmaceuticals USA, Inc.
On October 1, 2021, the Company entered into a License Agreement, or the Takeda Agreement, with Takeda. Under the Takeda Agreement, the Company granted Takeda an exclusive license to the Company’s ImmTOR technology initially for two specified disease indications within the field of lysosomal storage disorders. Takeda paid a $3.0 million upfront payment to the Company upon signing of the Takeda Agreement, and the Company is entitled to receive up to $1.124 billion in future additional payments over the course of the partnership that are contingent on the achievement of development or commercial milestones or Takeda’s election to continue its activities at specified development stages. The Company is also eligible for tiered royalties on future commercial sales of any licensed products.
Pursuant to the Takeda Agreement, the Company determined the Takeda Agreement represents a service arrangement under the scope of ASC 606, and given the reversion of the rights under the Takeda Agreement represents a penalty in substance for a termination by Takeda, the contract term would remain the stated term of the Takeda Agreement. The Company determined that the research license, the licensed know-how, and the manufactured supply and delivery of materials represent a single promise and performance obligation to be transferred to Takeda over time due to the nature of the promises in the contract. The delivery of the manufactured supply is the predominant promise within the arrangement, as it is essential to the utility of the licensed intellectual property. The material to be supplied by the Company to Takeda is unique to the Company and cannot be obtained by other vendors. As such, consideration in the initial transaction price will be allocated to the single performance obligation and the recognition period would not extend beyond the initial contractual period. The Company will recognize the revenue associated with the upfront payment and combined single performance obligation utilizing the output method over the term that manufactured supply is delivered to Takeda.
In determining the transaction price, the Company concluded the payment associated with all the performance milestones will be fully constrained and only be included in the transaction price when the respective milestone is deemed probable of achievement. Each of these variable consideration items were evaluated under the most likely amount method to determine whether such amounts were probable of occurrence, or whether such amounts should be constrained until they become probable. As part of its evaluation of the constraint, the Company considered numerous factors, including that receipt and timing of such study milestones is outside the control of the Company and probability of success criteria is estimated. The Company will re-evaluate the transaction price in each reporting period, as uncertain events are resolved, or as other changes in circumstances occur. Takeda has the right to exercise covenant release rights on a field-by-field basis. If Takeda exercises its covenant release rights, we could receive exercise payments per indication and would be entitled to significant development and commercial milestone payments and tiered royalties on commercial sales. The Company determined that a significant financing component does not exist in its arrangement with Takeda. The Company also determined the options to negotiate additional fields, pursue other products, enter into a supply agreement explore additional fields, and pursue additional development under the initial fields do not represent material rights under the agreement. Takeda has the right to terminate the Takeda Agreement in its entirety or on a field-by-field basis, upon 90 days’ written notice to the Company.
As of December 31, 2021, the Company recorded $1.0 million and $1.0 million, as a short-term and long-term contract liabilities, respectively, representing deferred revenue associated with this agreement. Revenue of $1.0 million related to the Takeda Agreement was recognized during the year ended December 31, 2021.
Swedish Orphan Biovitrum
License and Development Agreement
On June 11, 2020, the Company and Sobi entered into the Sobi License. Pursuant to the Sobi License, the Company has agreed to grant Sobi an exclusive, worldwide (except as to Greater China) license to develop, manufacture and commercialize the Company’s SEL-212 drug candidate, which is currently in development for the treatment of chronic refractory gout. The SEL-212 drug candidate is a pharmaceutical composition containing a combination of SEL-037, or the Compound, and ImmTOR. Pursuant to the Sobi License, in consideration of the license, Sobi agreed to pay the Company a one-time, upfront payment of $75.0 million. Sobi has also agreed to make milestone payments totaling up to $630.0 million to the Company upon the achievement of various development and regulatory milestones and, if commercialized, sales thresholds for annual net sales of SEL-212, and tiered royalty payments ranging from the low double digits on the lowest sales tier to the high teens on the highest sales tier.
Pursuant to the Sobi License, the Company has agreed to supply (at cost) quantities of the Compound and ImmTOR as necessary for completion of the two Phase 3 clinical trials of SEL-212 (DISSOLVE I and DISSOLVE II) and a 6-month placebo extension. The Company is required to supply quantities of the Compound until all rights to the Compound and any materials needed to manufacture the Compound are transferred to Sobi. Sobi has agreed to reimburse the Company for all budgeted costs incurred to complete development of SEL-212, including but not limited to costs incurred while conducting and completing the Phase 3 DISSOLVE trials, except for any costs of additional development activities required that are related to ImmTOR and that are unrelated to SEL-212. Sobi will have control and responsibility over all regulatory filings, including any investigational drug applications (IND), biologics license applications (BLA), and marketing authorization applications (MAA) relating to the licensed product.
The transactions contemplated by the Sobi License were consummated on July 28, 2020 following the expiration or termination of the required waiting periods under the Hart-Scott-Rodino Antitrust Improvements Act of 1976. Sobi may terminate the Sobi License for any reason upon 180 days’ written notice to the Company, whereby all rights granted under the Sobi License would revert back to the Company. In addition, if Sobi were to terminate the Sobi License, the Company has the option to obtain a license to all patents and know-how necessary to exploit SEL-212 in existence as of the termination date from Sobi in return for making an equitable royalty payment to Sobi.
Additionally, on June 11, 2020, the Company entered into the Sobi Purchase Agreement in connection with the Sobi License. The closing of the Sobi Private Placement occurred on July 31, 2020, following the closing of the transactions contemplated under the Sobi License. See Note 10 for details.
The Company adopteddetermined that the Sobi License represents a service arrangement under the scope of ASC 606 on January 1, 2018, using606. In addition, given the modified retrospective methodSobi License and Sobi Purchase Agreement were executed contemporaneously and negotiated as a package with a single commercial objective, the Company will account for all contracts not completedthe two agreements as a single contract. The term of the Sobi License commenced upon the effective date of adoption.July 28, 2020 and will continue on a product-by-product basis until the royalty terms for each country have expired. The reported resultsroyalty term for 2018 reflecta given product begins upon the applicationfirst commercial sale of ASC 606 guidance while the reported resultsproduct in a country and ends at the later of ten years from the first commercial sale, expiration of the last valid patent claim covering the product and expiration of all regulatory exclusivity periods for 2017 were preparedthe product in a country. Given the reversion of the rights under the guidance of ASC 605, Revenue Recognition (ASC 605), which is also referred to herein as "legacy GAAP" or the "previous guidance". The adoption of ASC 606Sobi License represents a changepenalty in accounting principlesubstance for a termination by Sobi, the contract term would remain the stated term of the Sobi License.
The Company determined that will more closely align revenue recognition withthe Sobi License contains 3 distinct performance obligations due to the nature of the promises in the contract, which includes conducting the Phase 3 DISSOLVE trials, Sobi’s option to set-up a second source supplier, and a combined obligation comprised of the delivery of the Company's services and will provide financial statement readers with enhanced disclosures.
Financial Statement Impact of Adopting ASC 606
The cumulative effect of applying the new guidancelicense to all contracts with customers that were not completed as of January 1, 2018, was recorded as an adjustment to accumulated deficit asSEL-212, transfer of the adoption date.know-how and the manufacturing and delivery of SEL-212 supply for development, or the Combined License Obligation. As the set-up of a resultsecond source supplier is optional for Sobi and the Company will be reimbursed at cost for its efforts in the subsequent set-up and technology transfer, the option for this future service was determined to be at a significant and incremental discount to its standalone selling price and treated as a material right in the arrangement, namely a distinct performance obligation.
In determining the transaction price, the Company concluded the upfront payment of applying$75.0 million and the modified retrospective$5.0 million development milestone associated with the dosing of the first patient in the Phase 3 DISSOLVE trials will be included in the transaction price. All other development milestones will be fully constrained and only be included in the transaction price when the respective milestone is deemed probable of achievement. Each of these variable consideration items was evaluated under the most likely amount method to adoptdetermine whether such amounts were probable of occurrence, or whether such amounts should be constrained until they become probable. As part of the new revenue guidance,evaluation of the constraint, the Company recorded an adjustment to long term deferredconsidered numerous factors, including that receipt of such milestones is outside the control of the Company and probability of success criteria is estimated. The Company will re-evaluate the transaction price in each reporting period, as uncertain events are resolved. In accordance with ASC 606, the Company will only recognize revenue associated with sales-based milestones and accumulated deficit totaling $1.8 million.royalties when the subsequent sales thresholds are reached and underlying sales occur, respectively. In connection with the adoptionSobi Purchase Agreement, the Company determined that the gross proceeds of $25.0 million from the Sobi Private Placement included a premium to the fair value of the Company’s shares as of July 28, 2020 equal to approximately $14.5 million. The premium amount will be included in the transaction price for revenue recognition. The Company will estimate and include in the transaction price the total reimbursements to be received from Sobi for both the manufacturing and delivery of the Compound and ImmTOR as well as conducting the Phase 3 DISSOLVE trials. The Company determined that a significant financing component does not exist in its arrangement with Sobi.
The Company allocated the transaction price based on the relative standalone selling prices of the 3 distinct performance obligations. The Company estimated the standalone selling price of conducting the Phase 3 DISSOLVE trials by forecasting its anticipated costs and applying a margin reflective of the industry. The Company must determine the standalone selling price of the second source supplier option by determining the discount given to Sobi multiplied by the likelihood that Sobi will exercise the option in the future. Similar to the Phase 3 program estimate, the Company estimated the discount of the option by forecasting the set-up costs and applying a margin that is reflective of the industry. As the Company will be providing the set-up and technology transfer services and the future supply at cost, the discount of the option is equal to the margin amount. The Company considered discussions with Sobi as well as probability of regulatory success of SEL-212 in determining the likelihood of exercise. The Company estimated the standalone selling price of the Combined License Obligation by utilizing a discounted cash flow model.
The Company determined that the delivery of the supply to Sobi best represents the pattern of delivery of the Combined License Obligation as the supply is essential to the utility of the license and know-how. The Company will recognize the revenue allocated to the Combined License Obligation by utilizing the output method. The Company estimated the total supply of the Compound and ImmTOR to be required during the clinical trial period and will recognize revenue as this supply is shipped for use in the clinical trials. The Company will recognize the revenue allocated to the conducting of the Phase 3 DISSOLVE trials obligation by utilizing the input method. The Company estimated the total budgeted costs to be incurred over the Phase 3 DISSOLVE trials and will recognize revenue as these costs are incurred. The Company’s costs best represent the pattern of transfer as these will capture all performance of the trials completed to date and can be readily measured. The Company will recognize the revenue allocated to the second source supplier option when the future services and goods are transferred.
As of December 31, 2021 and 2020, the Company recorded $37.5 million and $68.3 million, respectively, as a short-term contract liability and $5.1 million and $24.2 million, respectively, as a long-term contract liability, representing deferred revenue associated with this agreement. In addition, as of December 31, 2021 the Company has recorded $0.7 million of contract assets related to incremental costs that would not have been incurred if the Sobi License had not been obtained, of which $0.6 million is presented in prepaid expenses and other current assets and less than $0.1 million is presented in other assets in the accompanying consolidated balance sheets. Amortization of contract assets was $0.7 million for the year ended December 31, 2021.
As of December 31, 2021 and 2020, the Company recorded a total outstanding receivable of $9.9 million and $6.9 million, respectively, representing billings for the Phase 3 DISSOLVE program that are subject to reimbursement by Sobi. Revenue of $83.5 million and $16.6 million related to the Sobi License was recognized during the years ended December 31, 2021 and 2020, respectively.
Sarepta Therapeutics, Inc.
Research License and Option Agreement
In June 2020, the Company and Sarepta entered into a Research License and Option Agreement, or the Sarepta Agreement. Pursuant to the Sarepta Agreement, the Company agreed to grant Sarepta a license under the Company’s intellectual property rights covering the Company’s antigen-specific biodegradable nanoparticle encapsulating ImmTOR to research and evaluate ImmTOR in combination with Sarepta’s adeno-associated virus gene therapy technology, or gene editing technology, using viral or non-viral delivery, to treat Duchenne Muscular Dystrophy and certain Limb-Girdle Muscular Dystrophy subtypes, or the Indications. Sarepta will have an option term of 24 months during which it can opt-in to obtain an exclusive license to further develop and commercialize the Product to treat at least one Indication, with a potential to extend the option term for an additional fee. The Company will supply ImmTOR to Sarepta for clinical supply on a cost-plus basis.
Sarepta paid a $2.0 million upfront payment to the Company upon signing of the Sarepta Agreement, and the Company is eligible to receive additional preclinical payments during the option term. If Sarepta opts-in to an exclusive license agreement, the Company could receive option exercise payments per Indication upon execution of the exclusive license, and the Company would be entitled to significant development and commercial milestone payments and tiered royalties ranging from the mid-to-high single digits based on net sales.
Pursuant to the Sarepta Agreement, the Company determined the Sarepta Agreement represents a service arrangement under the scope of ASC 606, with a 24 month contract duration. Given the reversion of the rights under the Sarepta Agreement represents a penalty in substance for a termination by Sarepta, the contract term would remain the stated term of the Sarepta Agreement.
The Company determined that the Sarepta Agreement and supply obligation including the delivery of the research license, the licensed know-how, the manufactured supply and delivery of materials represent a single promise and performance obligation to be transferred to Sarepta over time due to the nature of the promises in the contract. The delivery of the manufactured supply is the predominant promise within the arrangement, as it is essential to the utility of the licensed intellectual property. As such, consideration in the initial transaction price will be allocated to the single performance obligation based on the contractual price.
In determining the transaction price, the Company identifiedconcluded the payment associated with all the performance milestones will be fully constrained and only be included in the transaction price when the respective milestone is deemed probable of achievement. Each of these variable consideration items was evaluated under the most likely amount method to determine whether such amounts were probable of occurrence, or whether such amounts should be constrained until they become probable. As part of its evaluation of the constraint, the Company considered numerous factors, including that receipt of such study milestones is outside the control of the Company and probability of success criteria is estimated.
The Company also determined the option to enter into a future commercial license agreement and extend the term of the option does not represent a material right since it was not priced at an incremental discount. Sarepta may terminate the Sarepta Agreement for any reason upon 30 days’ written notice to the Company. The Sarepta Agreement contains other customary terms and conditions, including representations and warranties, covenants, termination, and indemnification obligations in favor of each party. During the year ended December 31, 2020, the Company and Sarepta entered into 2 amendments relating to an additional feasibility study. During the year ended December 31, 2021, the Company and Sarepta entered into a third amendment relating to the additional feasibility study.
On April 13, 2021, the Company was notified by Sarepta of the achievement of the milestone event related to the completion of a non-clinical study for Duchenne muscular dystrophy and certain limb-girdle muscular dystrophies under the Sarepta Agreement. Accordingly, the Company received a milestone payment of $3.0 million during the three collaboration/grant arrangements that required analysis to quantify the impactmonths ended June 30, 2021.
As of adoption to its opening accumulated deficit balanceDecember 31, 2021, two milestones remained constrained, and as of January 1, 2018: Spark Therapeutics, Inc., Skolkovo FoundationDecember 31, 2020, all milestones were constrained. The Company will re-evaluate the transaction price in each reporting period, as uncertain events are resolved. The Company will recognize the revenue associated with the upfront payment and National Institutescombined single performance obligation utilizing the output method, over the 24 month term as the manufactured supply is delivered to Sarepta.
As of Health.December 31, 2021 and 2020, the Company recorded $4.6 million and $2.0 million, respectively, as a short-term contract liability representing deferred revenue associated with this agreement. Revenue of $0.4 million related to the Sarepta Agreement was recognized during the year ended December 31, 2021. De minimis revenue related to the Sarepta License Agreement was recognized during the year ended December 31, 2020.
Asklepios Biopharmaceutical, Inc.
License Agreement for Pompe Disease
OnIn December 17, 2019, the Company and AskBio entered into a License Agreement, referred to aslicense agreement, or the AskBio License Agreement. Pursuant to the AskBio License Agreement, AskBio has exercised its option to exclusively license the Company’s intellectual property rights covering the Company’s ImmTOR platform to research, develop, and commercialize certain AAV gene therapy products utilizing ImmTOR, and targeting the GAA gene, or derivatives thereof, to treat Pompe Disease.
Pursuant to the AskBio License Agreement and ancillary documents, AskBio agreed to pay to the Company upfront fees of an aggregate of $7.0 million. Assuming successful development and commercialization, the Company could receive up to an additional $237.0 million in development, regulatory, and sales milestone payments. If commercialized, the Company would be eligible to receive tiered royalties on global net sales at percentages ranging from mid-to-high single digits. Under the terms of the agreement, the Company will be eligible to receive these royalties commencing on the first commercial sale of the licensed product until the expiration of the later of (i) ten years after the first commercial sale and (ii) expiration of the last to expire valid claim on patents covering the licensed product.
Pursuant to the AskBio License Agreement, the Company will supply AskBio with its ImmTOR platform, ("or the Supply Obligation")Obligation, and AskBio will be responsible for all preclinical, clinical and commercial manufacture and supply of Licensed Productslicensed products (other than ImmTOR) and carry out all other activities related to the research, development, and commercialization of Licensed Productslicensed products at its sole expense, including all regulatory activities related thereto.
The Company determined that the AskBio License was not capable of being distinct from theAgreement and Supply Obligation. The Company has concluded thatObligation represent a single promise and performance obligation. This is because AskBio cannot derive benefit from the license without the simultaneous transfer of the patent protected ImmTOR supply. Therefore, the License Obligation and Supply Obligation represent the only promise in the arrangement and are combined as a single performance obligation (the “AskBio License and Supply Obligation”).obligation.
In determining the transaction price, the Company concluded that the future development milestones, regulatory milestones, sales milestones, and sales royalties all represent variable consideration. Each of these variable consideration items was evaluated under the most likely amount method to determine whether such amounts were probable of occurrence, or whether such amounts should be constrained until they become probable. As part of its evaluation of the constraint, the Company considered numerous factors, including that receipt of such milestones is outside the control of the Company. Consideration related to sales-based milestones as well as royalties on net sales upon commercialization by AskBio, will be recognized when the related sales occur, as they were determined to relate predominantly to the intellectual property granted to AskBio and, therefore, have also been excluded from the transaction price in accordance with the royalty recognition constraint. As of December 31, 2019,2021 and 2020, all milestones were constrained. The Company will re-evaluate the transaction price in each reporting period, as uncertain events are resolved, or as other changes in circumstances occur.
The total initial transaction price of the contract on the effective date iswas $7.0 million, which is comprised of thea $2.0 million initial up-frontupfront payment upon agreement of terms, and a $5.0 million initial up-frontupfront execution fee. The $5.0 million execution fee became billable on the effective date and is recorded on the balance sheet as a receivable and a corresponding contract liability as
At each of December 31, 2019.
As of December 31, 2019,2021 and 2020, the Company recorded $1.7 million as a short-term contract liability and $5.3 million as a long-term contract liability, representing deferred revenue associated with this agreement. Revenue will be recognized over the period in which the particles are delivered. No revenue related to the AskBio License Agreement was recognized during the years ended December 31, 2021 and 2020 as no deliveries were made during these periods.
Spark Therapeutics, Inc.
Spark License Agreement
In December 2016, the Company entered into a Licenselicense and Option Agreement (“option agreement, or the Spark License Agreement”)Agreement, with Spark Therapeutics, Inc. (“Spark”) pursuant to which the Company and Spark agreed to collaborate on the development of gene therapies for certain targets utilizing the ImmTOR platform. The Spark License Agreement provides Spark with certain exclusive, worldwide, royalty bearing licenses to the Company’s intellectual property, allowing Spark to develop and commercialize gene therapies in combination with ImmTOR for Factor VIII, an essential blood clotting protein relevant to the treatment of hemophilia A, the initial identified target.
In additionPursuant to an upfront cash payment of $10.0 million under the Spark License Agreement, additional paymentsSpark made an upfront payment of an aggregate of $5.0 million$15.0 million. Additionally, in 2 payments of $2.5 million each were paid within twelve months of December 2, 2016 (“Contract Date”). The first of the two additional payments was scheduled to be made on or before May 31, 2017 (the “May 2017 License Payment”) (see “Spark Letter Agreement” below) and the second was made on October 31, 2017. Spark may also exercise options to research, develop and commercialize gene therapies utilizing the ImmTOR platform for up to 4 additional targets. The Company was eligible to receive a variable fee up to $2.0 million for each additional target option elected, dependent on the incidence of the applicable indication. The election period in which Spark could have exercised additional targets underconnection with the Spark License Agreement, was a term of three years from the Contract Date, which expired on December 1, 2019.
Assuming successful development and commercialization, the Company could receive up to an additional $65.0 million in development and regulatory milestone payments and $365.0 million in commercialization milestone payments for each indication. If commercialized, the Company would be eligible to receive tiered royalties on global net sales at percentages ranging from mid-single to low-double digits, all of which apply on a target-by-target basis. Under the terms of the agreement, the Company will be eligible to receive these royalties commencing on the first commercial sale of the licensed product and terminating upon the later of (i) ten years after the first commercial sale, (ii) expiration of the last to expire valid claim on patents covering the jointly invented field specific improvements, or (iii) the expiration of regulatory exclusivity in the applicable country for the licensed product.
The Spark License Agreement may be terminated by Spark for convenience upon ninety days’ notice. Either party may terminate the Spark License Agreement on a target-by-target basis for material breach with respect to such target.
In December 2016, the Company also entered into a ShareStock Purchase Agreement (the “Spark Purchase Agreement”) with Spark. Pursuant to the Spark Purchase Agreement, the Company sold 197,238 sharesSpark purchased a total of $15.0 million of the Company’s common stock to Sparkincluding 197,238 shares for gross proceedsan aggregate purchase price of $5.0 million or $25.35 per share of common stock, at an initial closing (the “Initial Closing”). The purchase price per share represents an amount equal to 115% of the average daily volume weighted average price (“VWAP”) of the common stock during the thirty consecutive calendar days leading up to and ending on the day prior to the Contract Date.
Beyond the Initial Closing, the Spark Purchase Agreement contemplated potential future sales of shares by the Company to Spark as follows:
First Acquisition Right. During the period beginning on May 1, 2017 and ending on June 1, 2017, Spark had the right (the “First Acquisition Right”) to purchase a number of324,362 shares of common stock equal tofor an aggregate purchase price of $5.0 million. See "Spark Letter Agreement" below.
Second Acquisition Right. During the period beginningmillion on October 1,June 8, 2017, and ending on November 1, 2017, Spark had the right (the “Second Acquisition Right”) to purchase a number of shares of common stock equal to an aggregate price of $5.0 million. On October 31, 2017 Spark exercised this right and purchased 205,254 shares of common stock from the Company for an aggregate purchase price of $5.0 million or $24.36 per share of common stock. The purchase price per share represents an amount equal to 115.0% of the average daily VWAP of the common stock during the thirty consecutive calendar days leading up to and ending on the day prior to the Second Acquisition Right notification date.
The First Acquisition Rights and Second Acquisition Rights are collectively referred to herein as the “Acquisition Rights”.
Under the Spark Purchase Agreement, Spark agreed not to dispose of any of the shares acquired at either the Initial Closing or the from the subsequent Acquisition Rights that it may acquire until January 1, 2018 and, thereafter, transfers are contractually subject to volume limitations applicable to an “affiliate” under Rule 144 of the Securities Act.October 31, 2017.
In connection with the Spark License Agreement and Spark Purchase Agreement, the Company has made contractual payments defined in the MIT license agreement (see Note 14) totaling $2.2 million for the MIT sub-license provided to Spark, and $0.4 million relative to the calculated premium paid by Spark for the equity investments made under the Spark Purchase Agreement.
The terms of the Spark Purchase Agreement and the Spark License Agreement were negotiated at the same time between the parties and the terms of the Spark Purchase Agreement are referenced in the Spark License Agreement in multiple sections. The pricing and terms of the agreements are unique and must be considered in contemplation with each other. There are provisions within the Spark License Agreement that link to the Spark Purchase Agreement related to provisions that constitute a material breach of the license agreement. Therefore, the Company concluded that the 2 agreements must be combined and evaluated as a single agreement. While the Spark Purchase Agreement and the Spark License agreement are considered to be a single
agreement, the Company determined that the purchase of common stock and future acquisition rights are not within the scope of ASC 606. The Company determined that the initial purchase of common stock combined with the embedded future stock Acquisition Rights had a fair value of $2.7 million and this amount was recorded in equity as of the effective date. The remaining $2.3 million of cash received in exchange for the stock and acquisition rights is included in allocable consideration, as this represents the premium paid by Spark on the purchase of common stock, and should be allocated to the remaining performance obligations.
The Company identified the following components of the agreement: (1) certain exclusive, worldwide, royalty bearing licenses to the Company’s intellectual property and a license to conduct certain research activities under the collaboration, (the "Spark License"), (2) options to research, develop and commercialize gene therapies utilizing the ImmTOR platform for up to 4 additional target therapy options, (the "Option Obligation"), (3) manufactured supply of ImmTOR, (the "Supply Obligation") at a discount. In exchange, the Company received an upfront payment of $15.0 million and is eligible to receive additional payments of up to $35.0 million based on the achievement by Spark of future specified development milestones, up to $30.0 million based on the achievement by Spark of future specified regulatory milestones, up to $110.0 million based on the achievement by Spark of future specified commercial milestones, and up to $255.0 million based on the achievement by Spark of future specified sales milestones. The Company will also be eligible to receive tiered royalty payments that reach low double-digits based on future net sales for the duration of the royalty term.
The Company determined that the Spark License and Supply Obligation represent a single promise and performance obligation (the “Combined License and Supply Obligation”). This is because Spark cannot derive benefit from the license without the simultaneous transfer of the patent protected ImmTOR supply. The Company also determined that the Target Options, which includes the related Supply Obligation, provides the customer with a material right and is considered a performance obligation in the arrangement since it was priced at an incremental discount. Therefore, the Company determined that the Spark agreement contains 5 distinct performance obligations: the Combined License and Supply Obligation, and the 4 separate Target Options.
In determining the transaction price, the Company considered the future development milestones, regulatory milestones, commercial milestones, sales milestone, and sales royalties all represent variable consideration. Each of these variable consideration items was evaluated under the most likely amount method to determine whether such amounts were probable of occurrence, or whether such amounts should be constrained until they become probable. As part of its evaluation of the constraint, the Company considered numerous factors, including that receipt of such milestones is outside the control of the Company. Separately, any consideration related to sales-based milestones as well as royalties on net sales upon commercialization by Spark, will be recognized when the related sales occur as they were determined to relate predominantly to the intellectual property granted to Spark and, therefore, have also been excluded from the transaction price in accordance with the royalty recognition constraint. As of December 31, 2019, all future milestones are constrained. The Company will re-evaluate the transaction price in each reporting period, as uncertain events are resolved, or as other changes in circumstances occur.
The Company determined that the up-front payment of $12.3 million ($15.0 million, less fair value of the equity totaling $2.7 million as discussed above) was included in the transaction price and was allocated to the performance obligations based on the Company’s best estimate of their relative stand-alone selling prices. The Company allocated $7.1 million to the Combined License and Supply Obligation and $5.2 million to the discount on the Target Options ($1.3 million for each option) using the relative standalone selling price method to each obligation. The standalone selling price for the Combined License and Supply Obligation was determined using a discounted cash flow model. The standalone selling price for the Target Options were determined based on the fair value of the license minus the strike price of the option (the probability of exercise was included in the valuation) as well as the estimated discount of the Supply Obligation.
The estimated proceeds expected to be received from the sale of the Supply Obligation were also included in the transaction price for the Combined License and Supply Obligation. The total consideration allocated to the Combined License and Supply Obligation will be recognized using the output method, based on the proportion of actual deliveries to the total expected deliveries over the initial term which was initially estimated to be approximately four years.
On December 1, 2019, the term for Spark to exercise additional target options expired; the Company recognized $6.7 million in revenue from deferred revenue as originally allocated. In addition, during the year ended December 31, 2019, there were two deliveries resulting in less than $0.1 million of revenue recognized. NaN revenue related to the Spark License Agreement was recognized during the year ended December 31, 2018.
As of December 31, 2019, there was a contract liability of $9.2 million representing deferred revenue presented as non-current associated with this agreement. As of December 31, 2018, there was $14.7 million of deferred revenue related to this agreement.
Spark Letter Agreement
On June 6, 2017, the Company and Spark entered into a letter agreement, (the “Letter Agreement”),or the Letter Agreement, pursuant to which the parties agreed that Spark would make the May 2017 License Payment by June 6, 2017. The May 2017 License Payment was received, and recorded as a liability as of June 30, 2017, of which some or all may potentially constitute the reimbursement described below. The parties also agreed that Spark would be deemed to have delivered notice on May 31, 2017 exercising its right to purchase the shares pursuant to the First Acquisition Right. The Letter Agreement further outlines a cost reimbursement arrangement, pursuant to which the Company agreed to reimburse Spark for all costs and expenses, including the cost of materials provided by the Company, associated with the preclinical research and toxicology studies being performed by Spark for any licensed products for a specified amount of time, (the “Reimbursement Period”),or the Reimbursement Period, in an amount not to exceed $2.5 million. In
The Company determined that the Letter Agreement resulted in a modification to the original agreement. The amount received totaling $2.5 million and the reimbursements pursuant to the Letter Agreement totaling $2.5 million were both included in the transaction price, and a liability was recorded for the amount expected to be repaid. As repayments were made, the underlying liability was reduced. To the extent that an amount was expected to be applied towards the clinical supply obligation, the analysis of variable consideration was updated accordingly.
On October 31, 2017, Spark paid the Company a $2.5 million milestone payment pursuant to the Spark License Agreement, which was included in the transaction price and allocated to the performance obligations using the relative standalone selling price. In addition, Spark exercised the Second Acquisition Right set forth in Section 2.4 of the Spark Purchase Agreement and purchased 205,254 shares of common stock from the Company for $5.0 million, or $24.36 per share of common stock. The purchase price per share represents an amount equal to 115.0% of the average daily VWAP of the common stock during the thirty consecutive calendar days leading up to and ending on the day prior to the Second Acquisition Right notification date.
On June 5, 2019, the term of the Reimbursement Period under the Letter Agreement expired. During the year ended December 31, 2019, the Company updated its estimate of variable consideration included in the transaction price to include $1.2 million of unpaid reimbursements to Spark.
In December 2019, the term for Spark to exercise additional target options expired. Therefore, during the year ended December 31, 2019, the Company recognized $6.7 million in revenue. Additionally, during the year ended December 31, 2019, there were two deliveries resulting in less than $0.1 million of revenue recognized. As of December 31, 2020, there was a contract liability of $9.2 million representing long-term deferred revenue to be recognized upon the occurrence of future deliveries under this agreement.
No revenue related to the Spark License Agreement was recognized during the years ended December 31, 2021 and 2020, as no deliveries were made during these periods.
On January 18, 2022, both parties agreed to mutually terminate the Spark License Agreement. Therefore, the contract liability of $9.2 million representing deferred revenue is presented as short-term on the accompanying consolidated balance sheet as of December 31, 2021.
Skolkovo Foundation
The Company has received grant funding fromDuring the year ended December 31, 2021, revenue of $0.1 million related to the remaining contract liability of the Russia-based Development Fund of New Technologies Development and Commercialization Center, ("Skolkovo"). From grant inception through December 31, 2019, the Company received $2.0 million from Skolkovo.
Based on the guidance in ASC 606, the Company concluded that the entire $2.0 million of grant funds received from Skolkovo is variable consideration. Although the Company believes it has an enforceable right to the amounts received, there is risk that an audit could result in the Company needing to refund certain amounts back to Skolkovo, resulting in variability in the transaction price. The Company utilized the “expected value” approach in determining the amount that can be recognized. The Company estimated that it will be entitled to revenue of $1.8 million from theor Skolkovo, grant and recorded this amount. The remainder of $0.2 millionfunding was recorded as a contract liability.
During the year ended December 31, 2018, the Company made a decision to cease work relating to the Skolkovo grant. As a result, Skolkovo performed a formal review of project expenses incurred by the Company. Skolkovo concluded that the Company should (i) return unused grant funds to Skolkovo in the amount of less than $0.1 million and (ii) reimburse $0.1 million of costs deemed to have been overspent relative to the cost share requirement stipulated in the grant.
As of December 31, 2019, a contract liability of $0.1 million remains on the balance sheet and will not be recognized as revenue untilat the expiration of the three-year audit period, expected April 2021, or sooner, if resolution is reached with Skolkovo or there is a change in the estimate.period.
Transaction Price Allocated to Future Performance Obligations
Remaining performance obligations represent the transaction price of contracts for which work has not been performed (or has been partially performed). As of December 31, 2019,2021, the aggregate amount of the transaction price allocated to remaining performance obligations was $9.2$65.3 million. The Company does not expect to recognize revenue on the remaining performance obligations over the next 12 months.
Contract Balances from Contracts with Customers (Takeda,Sobi, Sarepta, AskBio, Spark and Skolkovo Foundation)
The following table presents changes in the Company’s contract liabilities during the year ended December 31, 20192021 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Balance at | | | | | | Balance at |
| beginning of period | | Additions | | Deductions | | end of period |
| Contract liabilities: | | | | | | | |
| Deferred revenue | $ | 110,796 | | | $ | 6,000 | | | $ | (51,496) | | | $ | 65,300 | |
| Total contract liabilities | $ | 110,796 | | | $ | 6,000 | | | $ | (51,496) | | | $ | 65,300 | |
|
| | | | | | | | | | | | | | | | |
| | | Balance at | | | | | | Balance at |
| | | beginning of period | | Additions | | Deductions | | end of period |
| Year Ended December 31, 2019 | | | | | | | | |
| Contract liabilities: | | | | | | | | |
| Deferred revenue | | $ | 14,777 |
| | $ | 8,254 |
| | $ | (6,677 | ) | | $ | 16,354 |
|
| Other liabilities (1) | | 2,126 |
| | — |
| | (2,126 | ) | | — |
|
| Total contract liabilities | | $ | 16,903 |
| | $ | 8,254 |
| | $ | (8,803 | ) | | $ | 16,354 |
|
(1) On June 5, 2019, the term of the Reimbursement Period under the Letter Agreement expired. During the year ended December 31, 2019, the Company updated its estimate of variable consideration included in the transaction price to include $1.2 million of unpaid reimbursements to Spark.
13. Related‑Party Transactions
Financing
During the year ended December 31, 2019, the Company completed the 2019 Purchase Agreement, 2019 Private Placement, and the 2019 Follow-On. The following tables set forth the number of shares of common stock, common warrants, and pre-funded warrants purchased by executive officers and members of the board of directors of the Company (and related parties thereto), as described in Note 10.
|
| | | | | | | | | |
| | 2019 Purchase Agreement |
| Name | Shares of common
stock purchased | | Common warrants
to be purchased | | Total
aggregate purchase price |
| TAS Partners, LLC | 3,940,887 |
| | 1,970,443 |
| | $ | 6,000,000 |
|
| Carrie S. Cox | 65,681 |
| | 32,840 |
| | $ | 99,999 |
|
| Timothy Barabe | 65,681 |
| | 32,840 |
| | $ | 99,999 |
|
| Scott D. Myers | 39,409 |
| | 19,704 |
| | $ | 60,000 |
|
|
| | | | | | |
| | 2019 Private Placement |
| Name | Shares of common
stock purchased | | Total
purchase price |
| Timothy A. Springer, Ph.D. | 1,600,000 |
| | $ | 2,896,000 |
|
| TAS Partners, LLC (affiliate of Timothy A. Springer, Ph.D.) | 1,100,000 |
| | $ | 1,991,000 |
|
| Elona Kogan, J.D. | 82,872 |
| | $ | 149,998 |
|
| Patrick Zenner | 55,248 |
| | $ | 99,999 |
|
| Takashi Kei Kishimoto, Ph.D. | 50,000 |
| | $ | 90,500 |
|
| Carsten Brunn, Ph.D. | 41,436 |
| | $ | 74,999 |
|
| Scott D. Myers | 41,436 |
| | $ | 74,999 |
|
| Stephen Smolinski | 13,812 |
| | $ | 25,000 |
|
|
| | | | | | |
| | 2019 Follow-On |
| Name | Shares of common
stock purchased | | Total
purchase price |
| Timothy A. Springer, Ph.D. | 4,000,000 |
| | $ | 6,000,000 |
|
| Entities affiliated with Polaris | 666,666 |
| | $ | 999,999 |
|
| Chafen Lu (Timothy A. Springer’s wife) | 66,666 |
| | $ | 99,999 |
|
| Jed Springer (Timothy A. Springer’s brother) | 1,000 |
| | $ | 1,500 |
|
Consulting Services
During 2018, the Company entered into an amended consulting agreement, (the "Consulting Agreement") with Dr. Omid Farokhzad, a member of its Board of Directors. The term of the amendment to the Consulting Agreement was April 1, 2018 to December 31, 2019, which extended the original consulting term for an additional nine months from March 31, 2019. Compensation included a $85,000 payment for the period beginning January 1, 2019 and ending December 31, 2019. The $85,000 was paid quarterly across the contract term in arrears beginning March 31, 2019. Included within this agreement, a stock option award of 75,000 shares was granted, with a weighted average grant date fair value of $4.35. Effective November 20, 2019, Dr. Farokhzad resigned as a member of its Board of Directors.
In addition, the Company incurred expenses for consulting services provided by its founders totaling $0.5$0.1 million, $0.2$0.1 million and $0.2$0.5 million during the years ended December 31, 2021, 2020 and 2019, 2018 and 2017, respectively. The agreements associated with the above consulting services provided by its founders were terminated effective December 31, 2019. The Company entered into newconsulting agreements with its founders to serve roles on its Scientific Advisory Board, effective January 1, 2020 to December 31, 2021, under which they will bewere paid quarterly for their services.
14. Collaboration and License Agreements
Ginkgo Bioworks Holdings, Inc.
Collaboration and License Agreement
On October 25, 2021, the Company entered into a Collaboration and License Agreement, or the First Ginkgo Agreement, with Ginkgo. Under the First Ginkgo Agreement, Ginkgo will design next generation IgA proteases with potentially transformative therapeutic potential. In return, Ginkgo is eligible to earn both upfront research and development fees and milestone payments, including certain milestone payments for fixed fair values in the form of Selecta common stock, clinical and commercial milestone payments of up to $85.0 million in cash. The Ginkgo Agreement was assessed for collaboration components and was determined not to be within the scope of ASC 808 as the risk and rewards are not shared by both parties. The Company will expense costs related to the Ginkgo Agreement as incurred until regulatory approval is received in accordance with ASC 730. The Company is accounting for the contingently issuable shares to be issued in exchange for the license obtained from Ginkgo as a liability classified stock based compensation arrangement with a non-employee which will be recognized when achievement of the milestones is probable. The Company will assess the capitalization of costs incurred
after the receipt of regulatory approval and, if applicable, will amortize these payments based on the expected useful life of each asset, typically based on the expected commercial exclusivity period. The Company is also obligated to pay Ginkgo tiered royalties ranging from low-single digit to high-single digit percentages of annual net sales of collaboration products which will be expensed as the commercial sales occur.
Genovis AB (publ.)
License Agreement
On October 21, 2021, the Company entered into an Exclusive License Agreement, or the Genovis Agreement, with Genovis. Under the Genovis Agreement, the Company paid to Genovis an upfront payment in exchange for an exclusive license to Genovis’ IgG Protease, or Xork, enzyme technology across all therapeutic uses in humans, excluding research, preclinical, diagnostic and other potential non-therapeutic applications of the enzyme. Genovis is eligible to earn development and sales-based milestones. The Genovis Agreement was assessed for collaboration components and was determined not to be within the scope of ASC 808 as the risk and rewards are not shared by both parties. The Company will expense costs related to the Genovis Agreement as incurred until regulatory approval is received in accordance with ASC 730. The Company will assess the capitalization of costs incurred after the receipt of regulatory approval and, if applicable, will amortize these payments based on the expected useful life of each asset, typically based on the expected commercial exclusivity period. The Company is also obligated to pay Genovis tiered royalties of low double digit percentages of worldwide annual net sales of collaboration products which will be expensed as the commercial sales occur.
Cyrus Biotechnology, Inc.
Collaboration and License Agreement
On September 7, 2021, the Company and Cyrus Biotechnology, Inc., or Cyrus, entered into a collaboration and license agreement, or the Cyrus Agreement. Pursuant to the Cyrus Agreement, Cyrus agreed to grant the Company an exclusive, worldwide license to certain intellectual property to form a protein engineering collaboration combining the Company’s ImmTOR platform with Cyrus’ ability to redesign protein therapeutics. The lead program is a proprietary interleukin-2, or IL-2, protein agonist designed to selectively promote expansion of regulatory T cells for treatment of patients with autoimmune diseases and other deleterious immune conditions. In return for the licensed intellectual property, the Company made an upfront payment and is obligated to pay certain discovery, development, and sales-based milestones which could potentially total up to approximately $1.5 billion across multiple programs. The Cyrus Agreement was assessed for collaboration components and was determined not to be within the scope of ASC 808 as the risk and rewards are not shared by both parties. The Company will expense costs related to the Cyrus Agreement as incurred until regulatory approval is received in accordance with ASC 730. The Company will assess the capitalization of costs incurred after the receipt of regulatory approval and, if applicable, will amortize these payments based on the expected useful life of each asset, typically based on the expected commercial exclusivity period. The Company is also obligated to pay Cyrus tiered royalties ranging from mid-single digit to low-double digit percentages of annual net sales of collaboration products which will be expensed as the commercial sales occur.
Additionally, on September 7, 2021, the Company entered into a stock purchase agreement, or the Series B Preferred Stock Purchase Agreement, in connection with the Cyrus Agreement. Pursuant to the Series B Preferred Stock Purchase Agreement, the Company purchased 2,326,934 shares of Cyrus’ Series B Preferred Stock, par value $0.0001 per share, at a purchase price of $0.8595 per share for $2.0 million.
In accordance with ASC 810, the Company has a variable interest in Cyrus resulting from its equity investment. The Company will share in Cyrus’ expected losses or receive a portion of its expected returns and absorb the variability associated with changes in the entity’s net assets. However, the Company is not the primary beneficiary as it does not have the power to direct the activities most significant to Cyrus, and therefore it is not required to consolidate Cyrus. The Company determined its equity interest to be within the scope of ASC 321 and elected to record the $2.0 million investment of Cyrus’ Series B Preferred Stock at cost on the purchase date.
As of December 31, 2021, no impairment indicators are present and therefore the carrying value of the investment in Cyrus is $2.0 million on the accompanying consolidated balance sheet. The Company’s maximum exposure to loss related to this variable interest entity is limited to the carrying value of the investment. The Company has not provided financing to Cyrus other than the amount contractually required by the Series B Preferred Stock Purchase Agreement.
Asklepios Biopharmaceutical, Inc.
Feasibility Study and License Agreement
OnIn August 6, 2019, the Company entered into a Feasibility Studyfeasibility study and License Agreementlicense agreement with AskBio, which is referred to asor the AskBio Collaboration Agreement. Pursuant to the AskBio Collaboration Agreement, the Company and AskBio agreed to license intellectual property rights to each other as part of a collaboration to research, develop, and commercialize certain adeno-associated virus (“AAV”)AAV gene therapy products utilizing the Company’s ImmTOR platform to enable re-dosing of such AAV gene therapy products to treat serious rare and orphan genetic diseases for which there is a significant unmet medical need.
Pursuant to the AskBio Collaboration Agreement, the Company and AskBio agreed to conduct proof of concept studies to potentially validate the use of ImmTOR in conjunction with AskBio’s AAV gene therapy, or SEL-302, (previously disclosed as MMA-101, in combination with ImmTOR) for the treatment of methylmalonic acidemia (MMA), based on the Company’s product candidate SEL-302,MMA, to mitigate the formation of neutralizing anti-AAV capsid antibodies, (the POC Studies). Ifor the POC Studies are successful,Studies. On April 29, 2021, the Company was notified by AskBio that it intended to opt-out of development of the MMA indication. The feasibility study and license agreement with AskBio, or AskBio Collaboration Agreement, otherwise remains in effect. Consequently, the parties otherwise electCompany has assumed all rights to do so, the parties will proceedMMA program and intends to continue to progress the SEL-302 program through clinical development. The Company filed an IND to conduct a Phase 1/2 clinical trial of its SEL-302 product candidate in pediatric patients with methylmalonic acidemia in the third quarter of 2021. On November 23, 2021, this trial was placed on clinical hold by the FDA, with questions specifically relating to chemistry, manufacturing and controls, or CMC, of the AAV vector. On February 9, 2022, we submitted a collaborationwritten response to pursue the developmentFDA to answer its questions. On March 9, 2022, we received a letter from the FDA indicating the clinical hold was removed and commercializationthe trial may proceed.
The SEL-399 program combined an empty AAV capsid (EMC-101), which is an AAV capsid containing no transgene, with ImmTOR and is being conducted in partnership with AskBio. Building on the preclinical data the Company has generated showing ImmTOR’s effect on mitigating or reducing the formation of neutralizing antibodies to AAV gene therapy product candidates utilizingtherapies, the Company completed a clinical trial of SEL-399 in healthy adult volunteers in Belgium. The goal of the SEL-399 clinical trial was to demonstrate the appropriate dose of ImmTOR in humans to mitigate the formation of antibodies to AAV capsids used in gene therapies. This promising study in healthy volunteers provides support for the potential use of ImmTOR for the treatmentinhibition of certain agreed serious rare and orphan genetic diseases. If the POC Studies failneutralizing antibodies to demonstrate a proof of concept, and the parties do not mutually agreeAAV8 in writing to proceed with the collaboration, the AskBio Collaboration Agreement will expire.gene therapy clinical trials.
The Company and AskBio will share responsibility for the research, development and commercialization of products developed under thisthe SEL-399 program collaboration. The parties will also share research, development, and commercialization costs equally for all collaboration products, but with a right of either party to opt out of certain products, and thereby no longer be required to share costs for such products. Each party will receive a percentage of net profits for each product sold under the collaboration equal to the percentage of shared costs borne by such party in the development of such product. Pursuant to the AskBio Collaboration Agreement, AskBio is responsible for manufacturing the AAV capsids and AAV vectors and the Company is responsible for manufacturing ImmTOR.
The AskBio Collaboration Agreement is considered to be within the scope of ASC 808, as both parties are active participants and exposed to the risks and rewards of the collaborative activity. The Company evaluated the terms of the AskBio Collaboration Agreement and have identified the following promises in the arrangement (1) conducting research and development activities to develop and commercialize products under the collaboration, (the “Ror the R&D Services”),Services, (2) granting a non-exclusive, non-transferable, royalty-free, fully paid up, worldwide license to certain intellectual property of the Company, (the “IP Rights”)or the IP Rights, for the purpose of performing the POC Studies, (the “Research License”),or the Research License, (3) granting an exclusive, nontransferable, worldwide license to the IP Rights for use in certain indications, (the”or the Collaboration License”),License, (4) providing manufactured supply of preclinical and clinical ImmTOR, (the “Manufactured Supply”),or the Manufactured Supply, (5) participation on identified steering committees responsible for the oversight of the collaboration, (the “JSC Participation”),or the JSC Participation, and (6) granting an exclusive option to obtain a license under the IP Rights to research, develop and commercialize Licensed Products. The Company determined that the R&D Services, Research License, Collaboration License, Manufactured Supply, and JSC Participation were not capable of being distinct, and therefore must be combined into a single performance obligation. Therefore, promises (1) through (5) identified above were combined into a single performance obligation. Furthermore, the Company evaluated the Option Agreement and determined that it does not provide AskBio with a material right under ASC 606 as the option was not priced at a discount (see discussion of the Optionoption exercise in Note 12). The Company noted that AskBio did not meet the definition of a customer within the scope of ASC 606 for any distinct performance obligations as the Company concluded that such items were not an output of the Company’s ordinary activities. As such, the Company determined that the entire arrangement would be accounted for within the scope of ASC 808.
In accordance with ASC 808, collaboration expenses are recognized within R&D expense and selling, general and administrative expense on ourthe Company’s condensed consolidated statements of operations. For the year ended December 31, 2019, the
Company did not recognize any collaboration expense or record a receivable under the AskBio Collaboration Agreement in which actual costs incurred by both parties are an approximated 50% cost share.
Under certain collaborative arrangements, the Company is entitled to reimbursement of certain R&D expense. Activities under collaborative arrangements for which the Company is entitled to reimbursement are considered to be collaborative activities under the scope of ASC 808. For these units of account, the Company does not analogize to ASC 606 or recognize revenue. Rather, the Company analogizes to the guidance in ASC 730, which requires that reimbursements from counterparties be recognized as an offset to the related costs. In accordance with ASC 730, the Company records reimbursement payments received from collaboration partnerscollaborators as reductions to R&D expense.
For the years ended December 31, 2021 and 2020, the Company recognized $2.7 million and $3.8 million, respectively, of collaboration expense under the AskBio Collaboration Agreement in which actual costs incurred by both parties approximate a 50% cost share.
Massachusetts Institute of Technology
On December 13, 2019, the Company entered into the Fourth Amendment (the MIT Amendment) to the Exclusive Patent License Agreement by and between the Company and the Massachusetts Institute of Technology (MIT) (the MIT Agreement). Pursuant to the MIT Amendment, a provision of the MIT Agreement under which the Company was obligated to initiate a Phase 3 clinical trial for a licensed product by a specified date in the fourth quarter of 2019 is tolled until the earlier of (i) a specified date in the second quarter of 2020 or (ii) the effective date of a written amendment to the MIT Agreement. Further, pursuant to the MIT Amendment, the parties agreed to negotiate in good faith to enter into a future amendment to the MIT Agreement after the Company provides MIT with an amended diligence plan.
OnIn November 25, 2008, the Company entered into an Exclusive Patentexclusive patent license agreement, or the MIT License, agreement with the Massachusetts Institute of Technology, or MIT, under which is referred to as the Exclusive Patent License. The Company received an exclusive royalty‑bearingroyalty-bearing license to utilize patents held by MIT in exchange for upfront consideration and annual license maintenance fees. Such fees are expensed as incurred and have not been material to any period presented.
In June 2020, the Company entered into a Fifth Amendment, or the MIT Amendment, to the MIT License, which is effective as of May 15, 2020. Pursuant to the MIT Amendment, certain of the Company’s diligence obligations were extended. The extension included the obligation to commence a Phase 3 trial for a licensed product by the second quarter of 2021 or to file an IND (or equivalent) with the FDA or comparable European regulatory agency for a licensed product by the second quarter of 2023. Additionally, certain of the Company’s development and regulatory milestones and payments upon achievement of such milestones were adjusted.
As of December 31, 2019,2021, and in connection with the execution of the Spark License Agreement, the Company has made contractual payments pursuant to the Exclusive PatentMIT License totaling $2.2 million, for the sublicense granted to Spark, and $0.4 million relative to the calculated premium paid by Spark for the equity investments made under the Spark Purchase Agreement. The Company made 0no additional payments during the year ended December 31, 2019.2021.
Shenyang Sunshine Pharmaceutical Co., Ltd
In May 2014, the Company entered into a license agreement, or the 3SBio License, with Shenyang Sunshine Pharmaceutical Co., Ltd. (“3SBio”), which is referred to as the 3SBio License.or 3SBio. The Company has paid to 3SBio an aggregate of $3.0$7.0 million in upfront and milestone-based payments under the 3SBio License as of December 31, 2019. We are2021. The Company is required to make future payments to 3SBio contingent upon the occurrence of events related to the achievement of clinical and regulatory approval milestones of up to an aggregate of $21.0$15.0 million for products containing our ImmTOR platform, and up to an aggregate of $41.5 million for products without ourthe Company’s ImmTOR platform.
Massachusetts Eye and Ear Infirmary and The Schepens Eye Research Institute, Inc.
In May 2016, the Company entered into a license agreement with the Massachusetts Eye and Ear Infirmary and The Schepens Eye Research Institute, Inc. (collectively, “MEE”), which is referred to as the MEE License. On September 16, 2019, in accordance with the terms of the MEE License, the Company notified MEE of its intention to terminate the MEE License, effective December 15, 2019. In connection with the termination, the Company has accrued less than $0.1 million for final license fees as of December 31, 2019. Through December 31, 2019, the Company paid a total of $0.4 million in license fees due under the MEE License.
15. Income Taxes
The Company provides for income taxes under ASC 740. Under ASC 740, the Company provides deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the Company’s financial statement carrying amounts and the tax bases of assets and liabilities using enacted tax rates expected to be in effect in the years in which the differences are expected to reverse.
On June 11, 2020, the Company entered into the Sobi License (see note 12). In September 2020, Sobi paid the Company a one-time upfront payment of $75.0 million. Sobi has also agreed to make milestone payments totaling up to $630.0 million to the Company upon achievement of various development and regulatory milestones and sales thresholds for annual net sales of SEL-212, and tiered royalty payments ranging from low double digits on the lowest sales tier to high teens on the highest sales tier.
For income tax purposes, the transfer of trademark and product rights is treated as a sale and the net proceeds from the sale are taxed under the default installment method as cash is received by the Company. During the year ending December 31, 2021, the Company completed an analysis of future tax obligations under the default installment sale method versus making a timely filed election on its 2020 tax return due October 15, 2021 to elect out of the installment sale method for income tax purposes. As a result, the Company elected out of the default installment sale treatment with the filing of its tax return. In the elect out method, the Company was taxed based upon the estimated fair value of all present and future proceeds from the sale and the Company utilized all of its available net operating losses and income tax credits, which served to reduce the federal and state tax liability. As the Company recognizes future revenue under the Sobi license for US GAAP purposes, the Company will exclude that revenue from taxable income.
For the year ended December 31, 2021, the Company recognized a current tax expense of $16.0 million, inclusive of estimated penalties and interest of $1.3 million. For the years ended December 31, 2019, 20182020 and 2017,2019, the Company did not record a current or deferred income tax expense or benefit. The following table reconciles the federal statutory income rate to the Company'sCompany’s effective income tax rate:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Statutory U.S. federal rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| State income taxes - net of federal benefit | (166.0 | %) | | 5.8 | % | | 6.3 | % |
| Permanent items | 8.3 | % | | (2.9 | %) | | (2.1 | %) |
| Research tax credits | 55.0 | % | | 1.0 | % | | 1.1 | % |
| Deferred revenue | 156.5 | % | | — | % | | — | % |
| Other | (3.7 | %) | | — | % | | — | % |
| Valuation allowance, net | (230.1 | %) | | (23.6 | %) | | (26.3 | %) |
| Stock compensation | (5.2 | %) | | (1.3 | %) | | — | % |
| Effective income tax rate | (164.2 | %) | | — | % | | — | % |
|
| | | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 | | 2017 |
| Statutory U.S. federal rate | 21.0 | % | | 21.0 | % | | 34.0 | % |
| State income taxes - net of federal benefit | 6.3 | % | | 7.0 | % | | 5.8 | % |
| Permanent items | (2.1 | )% | | (0.5 | )% | | (0.1 | )% |
| Research tax credits/other | 1.1 | % | | 1.6 | % | | 0.5 | % |
| Change in enacted rates | — | % | | — | % | | (35.8 | )% |
| Valuation allowance, net | (26.3 | )% | | (29.1 | )% | | (6.7 | )% |
| Other | — | % | | — | % | | 2.3 | % |
| Effective income tax rate | — | % | | — | % | | — | % |
The tax effects of temporary differences that give rise to the Company'sCompany’s net deferred tax assets are as follows (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2021 | | 2020 |
| Deferred Tax Assets | | | |
| Net operating loss carryforwards | $ | 13,932 | | | $ | 63,678 | |
| Research and development credits | 1,426 | | | 7,632 | |
| Stock-based compensations expense | 3,955 | | | 2,919 | |
| Other expenses | 745 | | | 788 | |
| Deferred revenue | 102,583 | | | 26,699 | |
| Operating lease liability | 2,636 | | | 2,884 | |
| Patent costs/amortization | 6,843 | | | 5,379 | |
| | | |
| | | |
| Gross deferred tax assets | 132,120 | | | 109,979 | |
| | | |
| Deferred Tax Liabilities | | | |
| Depreciation | $ | (90) | | | $ | (11) | |
| Operating lease right-of-use asset | (2,685) | | | (2,991) | |
| | | |
| | | |
| Gross deferred tax liabilities | (2,775) | | | (3,002) | |
| Net deferred tax assets before valuation allowance | 129,345 | | | 106,977 | |
| Valuation allowance | (129,345) | | | (106,977) | |
| Net deferred tax assets | $ | — | | | $ | — | |
|
| | | | | | | |
| | Year Ended December 31, |
| | 2019 | | 2018 |
| Deferred Tax Assets | | | |
| Net operating loss carryforwards | $ | 71,270 |
| | $ | 59,126 |
|
| Research and development credits | 6,586 |
| | 5,780 |
|
| Stock-based compensations expense | 2,516 |
| | 1,946 |
|
| Deferred rent and other expenses | 812 |
| | 458 |
|
| Deferred revenue | 4,468 |
| | 4,037 |
|
| Operating lease liability | 102 |
| | — |
|
| Patent costs/amortization | 5,111 |
| | 4,881 |
|
| Gross deferred tax assets | 90,865 |
| | 76,228 |
|
| | | | |
| Deferred Tax Liabilities | | | |
| Depreciation | $ | (57 | ) | | $ | (66 | ) |
| Operating lease right-of-use asset | (82 | ) | | — |
|
| Gross deferred tax liabilities | (139 | ) | | (66 | ) |
| Net deferred tax assets before valuation allowance | 90,726 |
| | 76,162 |
|
| Valuation allowance | (90,726 | ) | | (76,162 | ) |
| Net deferred tax assets | $ | — |
| | $ | — |
|
The Company has provided a full valuation allowance against its net deferred tax assets, as the Company believes that it is more likely than not that the deferred tax assets will not be realized.
Realization of future tax benefits is dependent on many factors, including the Company’s ability to generate taxable income within the net operating loss carryforward period. The Company has evaluated the positive and negative evidence bearing upon the realizability of its deferred tax assets and concluded that it is more likely than not that the Company will not realize the benefit of its deferred tax assets. The valuation allowance increased by $14.6$22.4 million and $18.5$16.3 million for the years ended December 31, 20192021, and 2018,2020, respectively, primarily as a result of an increase in net operating loss.deferred revenue. In 2014, the Company'sCompany’s Russian subsidiary was granted a 10 year tax holiday in Russia. The Company'sCompany’s foreign operations continue to benefit from the tax holiday, which is set to expire on December 31, 2023, however the Company is in the process of closing down operations in Russia and does not expect any tax liability.
At December 31, 2019,2021, the Company hadhas federal and state net operating loss carryforwards of $262.2$51.1 million and $256.5$50.8 million, respectively, which will expire at various times through 2037.2041. Of the federal net operating loss carryforwards, $99.7losses, $51.1 million can be carried forward indefinitely.indefinitely but will be subject to an 80% limitation. The Company also has $1.2 million and $0.3 million, respectively, of federal and state research and development tax credit carryforwards, of $4.2 million and $3.0 million, respectively, available to reduce future tax liabilities, which will expire at various times through 2039.
2041. Utilization of the net operating lossNOL carryforwards and research and developmentorphan drug credit carryforwards may be subject to a substantial annual limitation under Section 382 and 383 of the Internal Revenue Code of 1986, as amended (the Code), and similar state law due to ownership change limitations that have occurred previously, orchanges that could occur in the future.
These ownership changes may limit the amount of net operating loss and research and development credit carryforwards that can be utilized annually to offset future taxable incomeincome. If the Company experiences a change of control, as defined by Section 382 of the Code and tax, respectively. Assimilar state law, utilization of the NOL carryforwards or research and orphan drug credit carryforwards may be subject to an annual limitation under Section 382 of the Code, which is determined by first multiplying the value of the Company’s stock at the time of the ownership change by the applicable long-term tax-exempt rate, and then could be subject to additional adjustments, as required.
Any limitation may result in expiration of a portion of the NOL carryforwards or research and orphan drug credit carryforwards before utilization. The Company performed an analysis of ownership changes through December 31, 2019,2021. Based on this analysis, the Company completed adoes not believe that any of its tax attributes through December 31, 2021 will expire unutilized due to Section 382 study, noting that an ownership change occurred during 2017. However, the Company has determined that all net operating losses would be available in the future. As a result, the deferred tax assets related to the federal and Massachusetts net operating losses and credit carryforwards are not currently limited.limitations.
The Company applies ASC 740, Income Taxes to uncertain tax positions. As of the adoption date on January 1, 2010 and through December 31, 2019,2021, the Company had 0no unrecognized tax benefits or related interest and penalties accrued.
TheDuring 2021, the Company has not, as of yet, conductedcompleted a detailed study of its research and development credit carryforwards. This study may result in an adjustment to the Company’s research and development credit carryforwards; however, until a study is completed and any adjustment is known, no amounts are being presented as an uncertain tax position. A full valuation allowance has been provided against the Company’s research and developmentorphan drug credits and, if an adjustment is required, this adjustment would be offset by an adjustment to the valuation allowance.through December 31, 2020. As a result, there would be no impact to the consolidated balance sheets, statements of operationsCompany adjusted its current tax payable and comprehensive loss, or cash flows if an adjustment was required.deferred tax asset balances and the impacts are included in the federal research and orphan drug credit and state income taxes lines in the effective rate reconciliation above.
Interest and penalty charges, if any, related to unrecognized tax benefits would be classified as income tax expense in the accompanying statement of operations. As of December 31, 2019,2021, the Company had 0no accrued interest related to uncertain tax positions.
The statute of limitations for assessment by the Internal Revenue Service and Massachusetts tax authorities is open for tax years since inception.inception as the Company claimed research tax credits on its 2020 tax return which remains open for examination for the 2020 year as well as for any year in which a credit has been claimed for. The Company files income tax returns in the United States and Massachusetts. There are currently no federal, state or foreign audits in progress.
16. Defined Contribution Plan
The Company maintains a defined contribution plan, or the 401(k) Plan, under Section 401(k) of the Internal Revenue Code (the “401(k) Plan”).Code. The 401(k) Plan covers all employees who meet defined minimum age and service requirements and allows participants to defer a portion of their annual compensation on a pretax basis. The 401(k) Plan provides for matching contributions on a portion of participant contributions pursuant to the 401(k) Plan’s matching formula. All matching contributions vest ratably over 4four years and participant contributions vest immediately. Contributions by the Company totaled $0.2 million, $0.1 million, $0.2 million and $0.2$0.1 million during each of the years ended December 31, 2021, 2020 and 2019, 2018 and 2017.respectively.
17. Commitments and Contingencies
As of December 31, 2019,2021, the Company had operating lease agreementswas not a party to any litigation that could have a material adverse effect on the Company’s business, financial position, results of operations or cash flows.
On August 3, 2020, a stockholder of Selecta filed a stockholder derivative action, purportedly on behalf of Selecta and against certain current and former members of the Company’s Board of Directors, as well as one affiliated company owned by a current board member, in the Court of Chancery of the State of Delaware, namely Franchi v. Barabe, et al. The complaint alleges that the individual defendants breached their fiduciary duties and committed corporate waste when they authorized a private placement transaction, announced on December 19, 2019, at a price allegedly below fair value. The complaint further alleges that the four defendant directors who participated in the private placement were unjustly enriched in connection with the transaction. On September 25, 2020, the defendants filed a motion to dismiss the lawsuit. On November 6, 2020, the plaintiff filed an amended complaint, and the defendants filed a second motion to dismiss on January 8, 2021. On December 31, 2020, we received a litigation demand letter from two other putative stockholders relating to the same private placement transaction. On April 12, 2021, the Court of Chancery in the State of Delaware granted a motion to stay the litigation pending a review by a Special Committee appointed by the Company’s Board of Directors. While the litigation was stayed, the parties reached an agreement in principle to settle the matter, and they expect to submit documentation to the Court for officesits approval of the settlement in Watertown, MA. See Note 8the near future. As of December 31, 2021, the Company accrued an estimated liability of $0.9 million for additional information regarding the Company's leases.plaintiff’s litigation, as the liability has been determined to be probable.
Other
As permitted under Delaware law, the Company indemnifies its directors for certain events or occurrences while the director is, or was, serving at the Company’s request in such capacity. The term of the indemnification is for the director’s lifetime. The maximum potential amount of future payments the Company could be required to make is unlimited; however, the Company has directors’ insurance coverage that limits its exposure and enables it to recover a portion of any future amounts paid. The Company also has indemnification arrangements under certain of its facility leases that require it to indemnify the landlord against certain costs, expenses, fines, suits, claims, demands, liabilities, and actions directly resulting from certain breaches, violations, or non‑performancenon-performance of any covenant or condition of the Company’s lease. The term of the indemnification is for the term of the related lease agreement. The maximum potential amount of future payments the Company could be
required to make under these indemnification agreements is unlimited. To date, the Company had not experienced any material losses related to any of its indemnification obligations, and no material claims with respect thereto were outstanding.
The Company is a party in various other contractual disputes and potential claims arising in the ordinary course of business. The Company does not believe that the resolution of these matters will have a material adverse effect the Company'sCompany’s business, financial position, results of operations or cash flows.
18. Selected Quarterly Financial Data (Unaudited)
The following table summarizes unaudited quarterly financial data for the years ended December 31, 2019 and 2018 (in thousands, except per share data).
|
| | | | | | | | | | | | | | | | |
| | | Three Months Ended (unaudited) |
| | | March 31, | | June 30, | | September 30, | | December 31, |
| | | 2019 | | 2019 | | 2019 | | 2019 |
| Grant and collaboration revenue | | $ | 10 |
| | $ | 13 |
| | $ | — |
| | $ | 6,654 |
|
| Operating expenses | | $ | 11,866 |
| | $ | 16,248 |
| | $ | 11,794 |
| | $ | 19,224 |
|
| Net loss | | $ | (12,074 | ) | | $ | (16,394 | ) | | $ | (11,994 | ) | | $ | (14,888 | ) |
| Net loss attributable to common stockholders | | $ | (12,074 | ) | | $ | (16,394 | ) | | $ | (11,994 | ) | | $ | (14,888 | ) |
| | | | | | | | | |
| Net loss per share attributable to common stockholders, basic and diluted | | $ | (0.31 | ) | | $ | (0.37 | ) | | $ | (0.26 | ) | | $ | (0.28 | ) |
| | | | | | | | | |
| | | Three Months Ended (unaudited) |
| | | March 31, | | June 30, | | September 30, | | December 31, |
| | | 2018 | | 2018 | | 2018 | | 2018 |
| Grant and collaboration revenue | | $ | — |
| | $ | — |
| | $ | — |
| | $ | 903 |
|
| Operating expenses | | $ | 15,813 |
| | $ | 18,769 |
| | $ | 15,941 |
| | $ | 15,402 |
|
| Net loss | | $ | (15,888 | ) | | $ | (18,796 | ) | | $ | (16,001 | ) | | $ | (14,651 | ) |
| Net loss attributable to common stockholders | | $ | (15,888 | ) | | $ | (18,796 | ) | | $ | (16,001 | ) | | $ | (14,651 | ) |
| | | | | | | | | |
| Net loss per share attributable to common stockholders, basic and diluted | | $ | (0.71 | ) | | $ | (0.84 | ) | | $ | (0.71 | ) | | $ | (0.65 | ) |
18. Subsequent Events
Collaboration and License Agreements
On January 3, 2022, the Company entered into a Collaboration and License Agreement with Ginkgo Bioworks, Inc, or the Second Ginkgo Agreement. Under this agreement, the Company will engage with Ginkgo to develop AAV capsids designed to enhance transduction efficiency and transgene expression. In return, Ginkgo is eligible to earn both upfront research and development fees and milestone payments, including certain milestone payments in the form of Selecta common stock, clinical and commercial milestone payments of up to $207 million in cash, as well as downstream value in the form of royalties on sales.
“At-the-Market” Offerings
Subsequent to December 31, 2021, the Company sold 576,418 shares of its common stock pursuant to the 2021 Sales Agreement for aggregate net proceeds of $1.7 million, after deducting commissions and other transaction costs.