
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington,WASHINGTON, D.C. 20549
FORM 10-K
(Mark One)
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31 2017, 2023
or
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission File Number 001-37428
RITTER PHARMACEUTICALS, INC.
Qualigen Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 26-3474527 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
| ||
5857 Owens Avenue, Suite 300, Carlsbad, California92008
(Address of principal executive offices) (Zip Code)
(760)452-8111
Registrant’s telephone number, including area code: (310) 203-1000code
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol | Name of | ||
| Common Stock, par value $0.001 per share | The Nasdaq Capital Market |
Securities registered pursuant to Section 12(g) of the Act: None
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes [ ] ☐ No [X] ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes [ ] ☐ No [X] ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes [X] ☒ No [ ]☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes [X] ☒ No [ ]☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. [X]
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | Accelerated filer | |||
| Non-accelerated filer | Smaller reporting company | |||
| Emerging growth company |
If an emerging growth company, indicate by check mark if the Registrantregistrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. [ ]☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes [ ]☐ No [X]☒
As of June 30, 2017 (the last business day of the registrant’s most recently completed second fiscal quarter),2023, the aggregate market value of the registrant’s common stock held by non-affiliates was approximately $8.1 million$4,570,535 based uponon the closing price for the common stock of $0.91 on that date. Shares of common stock held by the registrant’s executive officers and directors have been excluded from this calculation, as such persons may be deemed to be affiliates of the registrant. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of March 25, 2024, there were shares of the registrant’s common stock of $0.55 as reported by the NASDAQ Capital Market on that date. For purposes of this calculation, the registrant has assumed that its directors, executive officers and holders of 5% or more of the outstanding common stock are affiliates.outstanding.
As of March 19, 2018, there were 49,406,521 shares outstanding of the registrant’s common stock, par value $0.001 per share.
DOCUMENTS INCORPORATED BY REFERENCE
None.
RITTER PHARMACEUTICALS, INC.TABLE OF CONTENTS
ANNUAL REPORT ON FORM 10-K
For the Year Ended December 31, 2017
Table of Contents
| 2 |
SPECIALCAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS AND INDUSTRY DATA
This Annual Report on Form 10-K (“Annual Report”) contains forward-looking statements by Qualigen Therapeutics, Inc. that involve substantial risks and uncertainties. Alluncertainties and reflect our judgment as of the date of this Report. These statements other than statements of historical facts contained in this Annual Report, including statements regardinggenerally relate to future events or our strategy, future operations, future financial position, future revenue, projected costs, prospects, plans, objectives of management and expected market growth are forward-looking statements. The words “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended toor operating performance. In some cases, you can identify forward-looking statements although not allbecause they contain words such as “may,” “will,” “should,” “expects,” “plans,” “anticipates,” “could,” “intends,” “target,” or “continue” or the negative of these words or other similar terms or expressions that concern our expectations, strategy, plans or intentions. Such forward-looking statements contain these identifying words. These statements are subjectmay relate to, knownamong other things, potential future development, testing and unknown risks, uncertaintieslaunch of products and other important factors thatproduct candidates. Actual events or results may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements.
Some of the factors that we believe could cause actual results to differ from those anticipated or predicted include:our expectations due to a number of factors.
These forward-looking statements include, but are not limited to, statements about:
| ● | our ability to procure sufficient working capital to continue and complete the development, testing and launch of our prospective drug products; | |
| ● | our ability to successfully develop any drugs; | |
| ● | our ability to progress our drug candidates through preclinical and clinical development; | |
| ● | our ability to obtain | |
| ● | ||
| our ability to | ||
| ● | ||
| ● | ||
| our ability to | ||
| ● | the likelihood that patents will issue on our in-licensed patent applications; | |
| ● | our ability to | |
| ● | ||
By their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics, and healthcare, regulatory and scientific developments and depend on the economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated. In light of the significant uncertainties in these forward-looking statements, you should not rely upon forward-looking statements as predictions of future events. Although we believe that we have a reasonable basis for each forward-looking statement contained in this Annual Report, we caution you that forward-looking statements are not guarantees of future performance and that our actual results of operations, financial condition and liquidity, and the development of the industry in which we operate may differ materially from the forward-looking statements contained in this Annual Report.
In addition, even if our results of operations, financial condition and liquidity, and the development of the industry in which we operate, are consistent in some future periods with the forward-looking statements contained in this Annual Report, they may not be predictive of results or developments in other future periods. Any forward-looking statement that we make in this Annual Report speaks only as of the date of this Annual Report, and we disclaim any intent or obligation to update these forward-looking statements beyond the date of this Annual Report, except as required by law. This caution is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995.
Future filings with the Securities and Exchange Commission (the “SEC”), future press releases and future oral or written statements made by us or with our approval, which are not statements of historical fact, may also contain forward-looking statements. Because such statements include risks and uncertainties, many of which are beyond our control, actual results may differ materially from those expressed or implied by such forward-looking statements. The forward-looking statements speak only as of the date on which they are made, and we undertake no obligation to update such statements to reflect events that occur or circumstances that exist after the date of this Annual Report. You should also read carefully the factors described in the “Risk Factors” section of this Annual Report to better understand the risks and uncertainties inherent in our business and underlying any forward-looking statements.on which they are made.
This Annual Report includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third-parties. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. While we believe these industry publications and third-party research, surveys and studies are reliable, we have not independently verified such data.
| 3 |
As used in this Annual Report, unless the context suggests otherwise, “we,” “us,” “our,”or “the Company” refer to Qualigen Therapeutics, Inc.
Item 1. Business
Overview
We are an early-clinical-stage therapeutics company focused on developing treatments for adult and pediatric cancer. Our business now consists of one early-clinical-stage therapeutic program (QN-302) and one preclinical therapeutic program (Pan-RAS).
Our lead program, QN-302, is an investigational small molecule G-quadruplexes (G4)-selective transcription inhibitor with strong binding affinity to G4s prevalent in cancer cells (such as pancreatic cancer). Such binding could, by stabilizing the G4s against DNA “unwinding,” help inhibit cancer cell proliferation. QN-302 is currently undergoing a Phase 1a clinical trial at START Midwest in Grand Rapids, Michigan, and HonorHealth in Scottsdale, Arizona.
Our Pan-RAS program, which is currently at the preclinical stage, consists of a family of RAS oncogene protein-protein interaction inhibitor small molecules believed to inhibit or block mutated RAS genes’ proteins from binding to their effector proteins thereby leaving the proteins from the mutated RAS unable to cause further harm. In theory, such mechanism of action may be effective in the treatment of about one quarter of all cancers, including certain forms of pancreatic, colorectal, and lung cancers. The investigational compounds within our Pan-RAS portfolio are designed to suppress the interaction of endogenous RAS with c-RAF, upstream of the KRAS, HRAS and NRAS effector pathways.
On May 22, 2020, we completed a “reverse recapitalization” transaction with Qualigen, Inc. (not to be confused with the Company); pursuant to which our merger subsidiary merged with and into Qualigen, Inc. with Qualigen, Inc. surviving as a wholly owned subsidiary of the Company. The Company, which had previously been known as Ritter Pharmaceuticals, Inc., was renamed Qualigen Therapeutics, Inc., and the former stockholders of Qualigen, Inc. acquired, via the recapitalization, a substantial majority of the shares of the Company. Ritter/Qualigen Therapeutics common stock, which was previously traded on the Nasdaq Capital Market under the ticker symbol “RTTR,” commenced trading on Nasdaq, on a post-reverse-stock-split adjusted basis, under the ticker symbol “QLGN” on May 26, 2020. We are no longer pursuing the gastrointestinal disease treatment business on which Ritter Pharmaceuticals, Inc. develops novel therapeutic productshad focused before the reverse recapitalization transaction. On July 20, 2023, we sold our Qualigen, Inc. subsidiary, which contained our former FastPack® diagnostics business to Chembio Diagnostics, Inc., an American subsidiary of French diagnostics provider Biosynex, S.A. Accordingly, our former FastPack® diagnostics business is reported as Discontinued Operations in this Annual Report.
The aggregate net purchase price for Qualigen, Inc. was $5.4 million in cash, of which $450,000 is being held in escrow to satisfy certain Company indemnification obligations. Any amounts remaining in the escrow that modulatehave not been offset or reserved for claims will be released to us within five business days following January 20, 2025.
We own a minority interest in NanoSynex, Ltd. (“NanoSynex”), a privately-held microbiologics diagnostic company domiciled in Israel. NanoSynex’s technology is for Antimicrobial Susceptibility Testing that aims to enable better targeting of antibiotics for their most suitable uses to ultimately result in faster and more efficacious treatment, hence reducing hospitals’ mortality and morbidity rates. On May 26, 2022, we acquired a 52.8% interest in NanoSynex from our related party Alpha Capital Anstalt (“Alpha”) and NanoSynex, and entered into a Master Agreement for the gut microbiomeOperational and Technological Funding of NanoSynex with NanoSynex (the “NanoSynex Funding Agreement”). On July 20, 2023, we entered into an Amendment and Settlement Agreement with NanoSynex (the “NanoSynex Amendment”), pursuant to treat gastrointestinal diseases. which we agreed to, in exchange for eliminating all future Funding Agreement obligations for us to invest further cash in NanoSynex (except for obligations to lend NanoSynex $560,000 on or before November 30, 2023, and $670,000 on or before March 31, 2024), surrender 281,000 Series B Preferred Shares of NanoSynex held by us, resulting in our ownership in NanoSynex being reduced from approximately 52.8% to approximately 49.97% of the voting equity of NanoSynex; in addition, we agreed to surrender approximately $3.0 million of promissory notes which NanoSynex had issued to us under the Funding Agreement. On November 22, 2023 we further agreed to eliminate our obligations to lend NanoSynex $560,000 on or before November 30, 2023, and $670,000 on or before March 31, 2024, by instead surrendering shares of Series A-1 Preferred Stock of NanoSynex in an amount that reduced our ownership in NanoSynex voting equity from approximately 49.97% to 39.90%. NanoSynex was deconsolidated from our financial statements as of July 20, 2023, and is reported as Discontinued Operations in this Annual Report. Our investment in NanoSynex will be accounted for in the future as an equity method investment.
| 4 |
Product Pipeline
QN-302
We exclusively in-licensed the global rights to the G-Quadruplex (“G4”) selective transcription inhibitor platform from University College London (“UCL”) in January 2022. The licensed technology comprises lead compound QN-302 (formerly known as SOP1812) and back-up compounds that target regulatory regions of cancer genes that down-regulate gene expression in multiple cancer pathways. Developed by Dr. Stephen Neidle and his group at UCL, the G4 binding concept is derived from nucleic acid research conducted over more than over 30 years, including research on G4s, which are advancing gut health researchhigher order DNA and RNA structures formed by exploringsequences containing guanine-rich repeats. G4s are overrepresented in telomeres (a region of repetitive DNA sequences at the gut microbiotaend of a chromosome) as well as promoter sequences and translatinguntranslated regions of many oncogenes. Their prevalence is therefore significantly greater in cancer cells compared to normal human cells.
G4-selective small molecules such as QN-302 and backup compounds target the functionalityregulatory regions of prebiotic-based therapeutics into applications intended tocancer genes, which have a meaningful impact onhigh prevalence of enriched G4s. Stable G4-QN-302 complexes can be impediments to replication, transcription or translation of those cancer genes containing G4s, and the drugs’ binding to G4s are believed to stabilize the G4s against possible “unwinding.” G4 binders like QN-302 could be efficacious in a patient’s health.variety of cancer types with a high prevalence of G4s.
Our first novel microbiome modulator, RP-G28, an orally administered, high purity galacto-oligosaccharide, is currently under development for the treatment of lactose intolerance. RP-G28 is designed to selectively stimulate the growth of lactose-metabolizing bacteria in the colon, thereby effectively adapting the gut microbiome to assist in digesting lactose (the sugar found in milk)We believe that reaches the large intestine. RP-G28QN-302 has the potential to becomedemonstrate superior efficacy and activity against pancreatic ductal adenocarcinoma (“PDAC”), which represents 98% of pancreatic cancers. Pancreatic cancer is the first drug approvedtenth most common cancer in men and the seventh most common in women, but it is the fourth leading cause of cancer deaths in men and the third leading cause in women; it accounts for about 3% of all cancers in the United States but is responsible for about 8% of all cancer-related deaths. It has one of the lowest rates of survival of all cancer types.
In-vitro and in-vivo studies have shown that G4 stabilization by QN-302 resulted in inhibition of target gene expression and cessation of cell growth in various cancers, including PDAC. In in-vitro studies, QN-302 was potent in inhibiting the growth of several PDAC cell lines at low nanomolar concentrations. Similarly, in in-vivo studies, QN-302 showed a longer survival duration in a KPC genetic mouse model for pancreatic cancer than gemcitabine (the current standard of care for PDAC) has historically shown. Additional preclinical in-vivo studies suggest activity in gemcitabine-resistant PDAC. Data further demonstrated that QN-302 had significant anti-tumor activity in three patient-derived PDAC xenograft models. Early safety indicators in pancreatic cancer mouse in-vivo models suggest no significant adverse toxic effects at proposed therapeutic doses.
On January 9, 2023, the U.S. Food and Drug Administration (“FDA”) granted Orphan Drug Designation (“ODD”) to QN-302 for the treatmentindication of lactose intolerance. RP-G28 has been studiedpancreatic cancer. ODD provides advantages to pharmaceutical companies that are developing investigational drugs or biological products that show promise in Phase 2a and Phase 2b clinical trials and is a first-in-class compound.
On March 28, 2017, we announced top-line results from our Phase 2b clinical trial of RP-G28 for the treatment of lactose intolerance. The Phase 2b trial was a double-blind, placebo-controlled, three-arm, multi-center study evaluating safety, efficacy and tolerability of two dosing regimens of RP-G28 in patients with lactose intolerance. Enrollment was initiated in March 2016 and the last patient completed dosing in October 2016. The study aimed to evaluate a patient’s ability to consume dairy foods post-treatment with improved tolerance and reduced digestive symptoms. A total of 368 subjects were randomized in the trial with 18 clinical sites participating throughout the United States. Patients underwent a screening period and a 30-day treatment period, followed by a 30-day post-treatment “real world” observation of milk and dairy product consumption period.
A subset of subjects enrolled into a 12-month extension study to evaluate long-term durability of treatment. The extension study also evaluated each participant’s microbiome, expanding knowledge of the effectstreating rare diseases or conditions that RP-G28 may have on adapting the gut microbiota in a beneficial manner. We completed this study in the fourth quarter of 2017.
We held a Type C meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products in March 2017, prior to the unblinding of our Phase 2b data, to discuss our development plans and Phase 2b clinical trial. The focus of the meeting was to obtain the FDA’s feedback on our Phase 2b clinical trial, including our statistical analysis plan (“SAP”), prior to unblinding any data.
We held an End-of-Phase 2 meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products in August 2017. The purpose of the meeting was to obtain the FDA’s feedback on our Phase 3 program. We reached general consensus with the FDA on certain elements of our current Phase 3 program and have received clear guidance and recommendations on many necessary components of our Phase 3 program; including the clinical, non-clinical, and chemistry, manufacturing and controls (CMC) requirements needed to support an NDA submission.
We have incorporated much of this guidance into our Phase 3 program. Our current Phase 3 clinical program will consist of two confirmatory clinical trials of similar trial design as our Phase 2b clinical trial and will include additional components that may allow for claims for durability of effect. These additional trials may be run in parallel. We anticipate that the first Phase 3 clinical trial will begin in the second quarter of 2018.
The Gut Microbiome
The human gut is a relatively under-explored ecosystem but provides a great opportunity for using dietary intervention strategies to reduce the impact of gastrointestinal disease. The human body carries about 100 trillion microorganisms in the intestines, which is 10 times greateraffect fewer than the number of cells in the human body. This microbial population is responsible for a number of beneficial activities such as fermentation, strengthening the immune system, preventing growth of pathogenic bacteria, providing nutrients, and providing hormones. The increasing knowledge of how these microbial populations impact human health provides opportunities for novel therapies to treat an assortment of diseases such as neurological disease, cardiovascular disease, obesity, irritable bowel syndrome, inflammatory bowel disease, colon cancer, allergies, autism and depression.
Lactose Intolerance
Lactose intolerance is a common condition attributed to the absence or insufficient levels of the enzyme lactase, which is needed to properly digest lactose, a complex sugar found in milk and milk-containing foods.
Studies have suggested that lactose intolerance is a widespread condition affecting over one billion people worldwide and over 40 million200,000 people in the United States, (or 15%including seven-year marketing exclusivity and eligibility to receive regulatory support and guidance from the FDA in the design of an overall drug development plan.
There are also economic advantages to receiving ODD, including a 25% federal tax credit for expenses incurred in conducting clinical research on the orphan designated product within the United States. Tax credits may be applied to the prior year or applied to up to 20 years of future taxes. ODD recipients may also have their Prescription Drug User Fee Act (PDUFA) application fees waived, a potential savings of around $3.2 million (as of fiscal year 2023) for applications requiring covered clinical data, and may qualify to compete for research grants from the Office of Orphan Products Development that support clinical studies.
On August 1, 2023 we announced that the FDA had cleared our investigational new drug (“IND”) application for QN-302, and on November 1, 2023 the first patient in our Phase 1a clinical trial for QN-302 was dosed at START Midwest in Grand Rapids, Michigan.
We will require additional cash resources to be able to continue and complete this Phase 1a clinical trial.
| 5 |
Pan-RAS (formerly referred to as RAS or RAS-F)
In July 2020 we entered into an exclusive worldwide in-license agreement with the University of Louisville’s Research Foundation (“UofL” or “ULRF”) for the intellectual property covering the “RAS” family of pan-RAS inhibitor small molecule drug candidates, which are believed to work by blocking RAS mutations directly, thereby inhibiting tumor formation (especially in pancreatic, colorectal and lung cancers). Pursuant to the license agreement, we will seek to identify and develop a lead drug candidate from the compound family and, upon commercialization, will pay UofL royalties in the low-to-mid-single-digit percentages on net sales of Pan-RAS inhibitor licensed products. The license agreement with UofL for Pan-RAS was amended in March 2021 and June 2023.
RAS is the most common oncogene in human cancer. Activating mutations in one of the U.S. population),three human RAS gene isoforms (KRAS, HRAS or NRAS) are present in about one-fourth to one-third of all cancers. For example, mutant KRAS is found in 98% of pancreatic ductal adenocarcinomas, 52% of colon cancers, and 32% of lung adenocarcinomas. For these three cancer types, cancers with an estimated nine million of those individuals demonstrating moderate to severe symptoms.
Current annual spending on over-the-counter lactose intolerance aidsmutant KRAS are diagnosed in more than 170,000 people each year in the United States has been estimated at approximately $2.45 billionand cause more than 120,000 deaths. Drugs that target signaling downstream of RAS are available; however, such drugs have shown disappointing clinical durability because RAS is a “hub” that activates multiple effectors, so drugs that block a single pathway downstream may not account for the many other activated pathways.
We also had a sponsored research agreement with UofL for Pan-RAS research; that agreement expired in December 2023.
We currently do not have the resources to advance our Pan-RAS program, and so we are seeking to out-license it.
On February 15, 2024, we entered into a License and Sublicense Agreement with Pan-RAS Holdings, Inc., a New York corporation (“Pan-RAS Holdings”), which contemplated an exclusive out-license of our Pan-RAS drug development program, including our rights under the ULRF license agreement, Pan-RAS Holdings.
Although the License and Sublicense Agreement called for a closing by March 16, 2024, the License and Sublicense Agreement was in essence structured as a 30-day option in favor of Pan-RAS Holdings.
At the contemplated closing, Pan-RAS Holdings would have paid us an upfront fee of $1,000,000 in cash. In addition, Pan-RAS Holdings would have become responsible to pay on our behalf our in-license royalty obligations to ULRF, as and when required.
Finally, if the contemplated closing had occurred, Pan-RAS Holdings would have required to pay to us for our own account, on a semiannual basis, royalties equal to 1.0% of net sales of any RAS products.
We would have owed certain amounts to ULRF under our in-license agreement from them, if, as and when we received any Non-Royalty Sublicensing Income from Pan-RAS Holdings.
Pan-RAS Holdings did not effectuate the closing by March 16, 2024, and we and they voluntarily terminated the License and Sublicense Agreement effective as of March 16, 2024.
.Previous ProgramsHowever,
We have discontinued all of our efforts the following programs, and we do not plan to resume them:
| 1. | QN-247(formerly referred to as ALAN or AS1411-GNP) – an oligonucleotide aptamer-based, nucleolin-inhibiting anticancer drug candidate, consisting of QN-165 conjugated with gold nanoparticles. | |
| 2. | QN-165 (formerly referred to as AS1411) – an oligonucleotide aptamer-based drug candidate for the potential broad-spectrum treatment of infectious diseases such as COVID-19. | |
| 3. | Selective Target Antigen Removal System (STARS) – a therapeutic blood-filtering device product concept, which would be designed to remove circulating tumor cells, viruses, inflammation factors and immune checkpoints. |
Research and Development
For research and development of our drug candidates, we have historically leveraged the scientific and technical resources and laboratory facilities of UofL and UCL, through technology licensing, sponsored research, and other consulting agreements. We have engaged contract research organizations (“CROs”) and clinical sites for the Phase 1a clinical trial of QN-302. We intend to focus our internal research and development on oversight of these optionsCROs. We currently have no internal research and development facilities.
Regulatory Matters
We have obtained FDA clearance/approval for our QN-302 Phase 1a clinical trial. We have not obtained FDA or other regulatory approval for any other drug candidate.
| 6 |
United States—FDA Drug Approval Process
The research, development, testing, and manufacture of product candidates are limitedextensively regulated by governmental authorities in the United States and there is no long-term treatment available.other countries. In the United States, the FDA regulates drugs under the Food, Drug and Cosmetics Act and its implementing regulations.
Unlike many common gastrointestinal conditions,The steps required to be completed before a drug may be marketed in the United States include, among others:
| ● | preclinical laboratory tests, animal studies, and formulation studies, all performed in accordance with the FDA’s Good Laboratory Practice (“GLP”) regulations; | |
| ● | submission to the FDA of an IND application for human clinical testing, which must become effective before human clinical trials may begin and for which progress reports must be submitted annually to the FDA; | |
| ● | approval by an independent institutional review board (“IRB”) or Ethics Committee (“EC”) at each clinical trial site before each trial may be initiated; | |
| ● | adequate and well-controlled human clinical trials, conducted in accordance with applicable IND regulations, Good Clinical Practices (“GCP”), and other clinical trial related regulations, to establish the safety and efficacy of the drug for each proposed indication to the FDA’s satisfaction; | |
| ● | submission to the FDA of a New Drug Application (“NDA”) and payment of user fees for FDA review of the NDA (unless a fee waiver applies); | |
| ● | satisfactory completion of an FDA pre-approval inspection of one or more clinical trial site(s) at which the drug was studied in a clinical trial(s) and/or of us as a clinical trial sponsor to assess compliance with GCP regulations; | |
| ● | satisfactory completion of an FDA pre-approval inspection of the manufacturing facility or facilities at which the drug is produced to assess compliance with current GMPs regulations; | |
| ● | agreement with the FDA on the final labeling for the product and the design and implementation of any required Risk Evaluation and Mitigation Strategy; and | |
| ● | FDA review and approval of the NDA, including satisfactory completion of an FDA advisory committee review, if applicable, based on a determination that the drug is safe and effective for the proposed indication(s). |
Preclinical tests include laboratory evaluation of product chemistry, toxicity, and formulation, as well as animal studies. The conduct of the preclinical tests and formulation of the compounds for testing must comply with federal regulations and requirements, including GLP regulations. The results of the preclinical tests, together with manufacturing information and analytical data, are submitted to the FDA as part of an IND application, which must become effective before human clinical trials may begin. An IND application will automatically become effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions about issues such as irritable bowel syndrome, inflammatory bowel diseases, gastroesophageal reflux disease, or dyspepsia (among many others), lactose intolerance symptoms can be completely abated by avoiding dietary lactose.the conduct of the trials as outlined in the IND application, and places the clinical trial(s) on a clinical hold. In this regard, lactose intolerance is an avoidance condition, similar to celiac sprue, food intolerances, or various environmental allergies. However, dairy avoidance may lead to inadequate calcium and vitamin D intake, which can predispose individuals to decreased bone accrual, osteoporosis, hypertension, rickets, osteomalacia, and possibly certain cancers. Although supplements and calcium-rich foods are available, several studies have shown that lactose intolerance patients had an average calcium intake of only 300-388 mg/day, significantly less thansuch a case, the 1000-1200 mg/day adult dietary recommended levels. The 2010 National Institutes of Health conference on lactose intolerance highlighted the long-term consequences of dairy avoidance demonstrating both the importance of treating the conditionIND application sponsor and the needFDA must resolve any outstanding FDA concerns or questions before clinical trials can proceed. We cannot be certain that submission of an IND application will result in the FDA allowing clinical trials to find improved solutionsbegin.
| 7 |
Clinical trials necessary for patients.
Diagnosis
Lactose intolerance is often diagnosed by evaluating an individual’sproduct approval are typically conducted in three sequential phases, but the phases may overlap or be combined. The study protocol and informed consent information for study subjects in clinical history, which reveals a relationship between lactose ingestion and onset of symptoms. Hydrogen breath tests maytrials must also be utilizedapproved by an IRB for each institution where the trials will be conducted, and each IRB must monitor the study until completion. Study subjects must provide informed consent and sign an informed consent form before participating in a clinical trial. Clinical testing also must satisfy the extensive GCP regulations for, among other things, informed consent and privacy of individually identifiable information.
| ● | Phase 1—Phase 1 clinical trials involve initial introduction of the study drug in a limited population of healthy human volunteers or patients with the target disease or condition. These studies are typically designed to test the safety, dosage tolerance, absorption, metabolism and distribution of the study drug in humans, evaluate the side effects associated with increasing doses, and, if possible, to gain early evidence of effectiveness. | |
| ● | Phase 2—Phase 2 clinical trials typically involve administration of the study drug to a limited patient population with a specified disease or condition to evaluate the preliminary efficacy, optimal dosages and dosing schedule and to identify possible adverse side effects and safety risks. Multiple Phase 2 clinical trials may be conducted to obtain information prior to beginning larger and more expensive Phase 3 clinical trials. | |
| ● | Phase 3—Phase 3 clinical trials typically involve administration of the study drug to an expanded patient population to further evaluate dosage, to provide substantial evidence of clinical efficacy and to further test for safety, generally at multiple geographically dispersed clinical trial sites. These clinical trials are intended to establish the overall risk/benefit ratio of the study drug and to provide an adequate basis for product approval. Generally, adequate and well-controlled Phase 3 clinical trials are required by the FDA for approval of an NDA. |
Post-approval trials, sometimes referred to diagnose lactose malabsorption and a milk challengeas Phase 4 clinical trials, may be conducted after receiving initial marketing approval. These trials are used to differentiate between lactose malabsorptiongain additional experience from the treatment of patients in the intended therapeutic indication and lactose intolerance. Further tests canare commonly intended to generate additional safety data regarding use of the product in a clinical setting. In certain instances, the FDA may mandate the performance of Phase 4 clinical trials as a condition of approval of an NDA or, in certain circumstances, post-approval.
The FDA has various programs, including fast track designation, breakthrough therapy designation, priority review and accelerated approval, which are intended to expedite or simplify the process for the development, and the FDA’s review of drugs (e.g., approving an NDA on the basis of surrogate endpoints subject to post-approval trials). Generally, drugs that may be conductedeligible for one or more of these programs are those intended to rule out other digestivetreat serious or life-threatening diseases or conditions, including: stool examinationthose with the potential to document the presenceaddress unmet medical needs for those disease or conditions, and/or those that provide a meaningful benefit over existing treatments. For example, a sponsor may be granted FDA designation of a parasite, blood testsdrug candidate as a “breakthrough therapy” if the drug candidate is intended, alone or in combination with one or more other drugs, to determine the presence of celiac disease, and intestinal biopsies to determine mucosal problems leading to malabsorption, such as inflammatory boweltreat a serious or life-threatening disease or ulcerative colitis.
Health Consequences
Substantialcondition and preliminary clinical evidence indicates that lactose intolerancethe drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. If a drug is designated as breakthrough therapy, the FDA will take actions to help expedite the development and review of such drug. Moreover, if a major factor in limiting calcium intakesponsor submits an NDA for a product intended to treat certain rare pediatric or tropical diseases or for use as a medical countermeasure for a material threat, and that meets other eligibility criteria, upon approval such sponsor may be granted a priority review voucher that can be used for a subsequent NDA. From time to time, we anticipate applying for such programs where we believe we meet the applicable FDA criteria. A company cannot be sure that any of its drugs will qualify for any of these programs, or even if a drug does qualify, that the review time will be reduced.
The results of the preclinical studies and of the clinical studies, together with other detailed information, including information on the manufacture and composition of the drug, are submitted to the FDA in the dietform of individuals who are lactose intolerant. Studies suggest that these individuals avoid milkan NDA requesting approval to market the product for one or more proposed indications. The testing and dairy products, resultingapproval process requires substantial time, effort and financial resources. Unless the applicant qualifies for an exemption, the filing of an NDA typically must be accompanied by a substantial “user fee” payment to the FDA. To support marketing approval, the data submitted must be sufficient in an inadequate intakequality and quantity to establish the safety and efficacy of calcium and significant nutritional and health risks.
At the 2010 National Institute of Health (“NIH”) Consensus Development Conference: Lactose Intolerance and Health, the NIH highlighted numerous health risks tied to lactose intolerance such as: osteoporosis; hypertension; and low bone density.There is substantial evidence indicating that lactose intolerance is a major factor in limiting calcium and nutrient intakeproduct in the dietproposed patient population to the satisfaction of people whothe FDA. After an NDA is accepted for filing, the FDA substantively reviews the application and may deem it to be inadequate, and companies cannot be sure that any approval will be granted on a timely basis, if at all. The FDA may also refer the application to an appropriate advisory committee, typically a panel of clinicians, for review, evaluation and a recommendation as to whether the application should be approved, but is not bound by the recommendations of the advisory committee.
| 8 |
Before approving an NDA, the FDA usually will inspect the facility or the facilities at which the drug is manufactured and determine whether the manufacturing and production and testing facilities are lactose intolerant. Adequate calcium intakein compliance with cGMP regulations. The FDA also may audit the clinical trial sponsor and one or more sites at which clinical trials have been conducted to determine compliance with GCPs and data integrity. If the NDA and the manufacturing facilities are deemed acceptable by the FDA, it may issue an approval letter, and, if not, the Agency may issue a Complete Response Letter (“CRL”). An approval letter authorizes commercial marketing of the drug with specific prescribing information for a specific indication(s). A CRL indicates that the review cycle of the application is essentialcomplete and the application is not ready for approval. A CRL may require additional clinical data and/or an additional pivotal Phase 3 clinical trial(s), and/or other significant, expensive and time-consuming requirements related to reducingclinical trials, preclinical studies or manufacturing. Even if such additional information is submitted, the risksFDA may ultimately decide that the NDA does not satisfy the criteria for approval. The FDA could also require, as a condition of osteoporosisNDA approval, post-marketing testing and hypertension.In addition, chronic calcium depletion has been linkedsurveillance to increased arterial blood pressure, thereby establishingmonitor the drug’s safety or efficacy or impose other conditions, or a relationship between hypertensionRisk Evaluation and low calcium intake.Moreover, there is evidenceMitigation Strategy that may include both special labeling and controls, known as Elements to Assure Safe Use, on the distribution, prescribing, dispensing and use of a correlation between calcium intakedrug product. Once issued, the FDA may withdraw product approval if, among other things, ongoing regulatory requirements are not met, certain defects exist in the NDA, or safety or efficacy problems occur after the product reaches the market.
Intellectual Property
Information regarding our (in-licensed) issued patents and both colonpending patent applications, as of December 31, 2023, is as follows (excluding patents and breast cancerpending patent applications which pertain to programs which we have discontinued). As of that date we did not have any directly-owned issued patents and pending patent applications.
| Subject Matter | Issued | Pending | Geographic Scope | Patent Term | ||||
| In-Licensed Patents | ||||||||
| University College London (UCL) | ||||||||
| QN-302 | 3 | 10 | U.S., Europe, Australia, Canada, China, Hong Kong, India, Japan, Korea, Russia | 2030-2040 | ||||
| University of Louisville (ULRF) | ||||||||
| Pan-RAS | 0 | 12 | U.S., Europe, Australia, Canada, China, Hong Kong, India, Israel, Japan, Korea, Mexico, Russia, South Africa | 2039* | ||||
| TOTAL | 3 | 22 |
*Decreased Calcium Intake Increases the Risk for HypertensionAnticipated patent term
Numerous published reports show that chronic calcium depletion may leadHuman Capital Management
As of March 25, 2024, we had 4 employees, all of whom were full-time. None of our employees is represented by a labor union or covered by a collective bargaining agreement.
Diversity & Inclusion. With respect to increased arterial blood pressure. Many additional papers have corroborated this relationship between hypertensionour employees overall, fifty percent (50%) are women and a low calcium intake.0% are people of color.
A growing body of evidence indicates that a nutritionally sound diet rich in fruits, vegetables and a generous component of low-fat dairy foods (sometimes referred to as the DASH diet) is optimal for reducing the risk of hypertension. Several reports have confirmed this finding in middle-aged and elderly women. Further, it appears that the DASH diet with generous low-fat dairy is associated with low prevalence of metabolic syndrome. Studies have suggested that the levels of dairy foods (three to four servings per day) required to achieve these effects are well above current U.S. averages and even further above those of lactose intolerant individuals who are avoiding dairy due to symptoms.Additional Information
Our History
We wereRitter Pharmaceuticals, Inc. (our predecessor) was formed as a Nevada limited liability company on March 29, 2004 under the name Ritter Natural Sciences, LLC. Our first prototype, Lactagen™, was an alternative lactose intolerance treatment method. In 2004, clinical testing was conducted, which included a 61-subject double-blind placebo controlled clinical trial. The results were published in the Federation of American Societies for Experimental Biology in May 2005 and demonstrated Lactagen™ to be an effective and safe product for reducing symptoms for nearly 80% of the clinical participants who were on Lactagen™.
InSeptember 2008, we expanded our focus by developing a prescription drug development program. We initiated the program by developing RP-G28, a second generation edition of Lactagen™. We believe that RP-G28 enables us to state stronger claims, garner more medical community support and reach a wider market in the effort to treat lactose intolerance.
To help fund the development of RP-G28, we were awarded a grant from the United States government’s Health Care Bill program, the Qualifying Therapeutic Discovery Project, in 2008. The grant program provides support for innovative projects that are determined by the U.S. Department of Health and Human Services to have reasonable potential to result in new therapies that treat areas of unmet medical need and/or prevent, detect or treat chronic or acute diseases and conditions.
On September 16, 2008, wethis company converted into a Delaware corporation under the name Ritter Pharmaceuticals, Inc.
We completed our initial public offering in June 2015. Our common stock is traded on On May 22, 2020, upon completing the Nasdaq Capital Market under the trading symbol “RTTR”.
Our Leading Product Candidate — RP-G28
Overview
RP-G28 is“reverse recapitalization” transaction with Qualigen, Inc., Ritter Pharmaceuticals, Inc. was renamed Qualigen Therapeutics, Inc. and Qualigen, Inc. became a novel highly purified GOS, which is synthesized enzymatically. The product is being developed for the treatment of lactose intolerance. The therapeutic is taken orally (a powder solution mixed in water) for 30 consecutive days. The proposed mechanism of action of RP-G28 is to increase the intestinal growth and colonization of bacteria that can metabolize lactose to compensate for a patient’s intrinsic inability to digest lactose. Once colonization of bacteria has occurred, it is hypothesized that patients will continue to tolerate lactose as long as they maintain their microflora balance. RP-G28 has the potential to become the first FDA-approved drug for the reduction of symptoms associated with lactose intolerance.
Galacto-oligosaccharides (GOS)
RP-G28 is a >95% purified GOS product derived from a commercially available GOS food ingredient, which is designated as generally recognized as safe (GRAS) by the FDA. GOS refers to a group of compounds containing β-linkages of 1 to 6 galactose units with a single glucose on the terminal end and are found at low levels in human milk. GOS is purified to a pharmaceutical grade by minimizing residual glucose, lactose, galactose and other impurities. Further processing includes ultra-filtration, nano-filtration, decolorization, deionization, and concentration to yield GOS 95 syrup, which is the starting material for RP-G28.
GOS products resist hydrolysis by salivary and intestinal enzymes becausewholly-owned subsidiary of the configurationCompany. On July 20, 2023 we sold Qualigen Inc. to ChemBio Diagnostics, Inc., an American subsidiary of their glycosidic bonds and reach the colon virtually intact. The undigested GOS enhances the growth of beneficial, lactose metabolizing, colonic bacteria that already exist in the subject’s digestive track, including multiple species and strains of bifidobacteria and lactobacilli. Once colonies of these bacteria have increased, continued lactose exposure should maintain tolerability of lactose without further exposureFrench diagnostics provider Biosynex S.A.
Our website address is www.qlgntx.com. We post links to RP-G28.
While formal nonclinical studies evaluating the safety of RP-G28 have not been performed, other commercially available GOS products have been evaluated in acute and repeat-dose general toxicology studies, reproductive toxicology studies, juvenile toxicology studies, genetic toxicology studies, and in long-term safety studies.
Clinical studies of GOS products were reviewed as part of the safety evaluation to support the Investigational New Drug Application (“IND”) for RP-G28. These include studies in adults (including pregnant women and geriatrics), children, infants and newborns (preterm and full term). The safety of GOS products in humans has been evaluated in 1316 adults at doses of 2.5 to 20 g/day for up to 12 months, and in 1125 children > 1 year of age at doses of 2.0 to 12 g/day for up to 1 year. Overall, no safety concerns attributableour website to the consumption of GOS were reported. Where side effects were observed,following filings as soon as reasonably practicable after they were typically mild and limited to increased flatulence, abdominal discomfort, and changes in stool consistency and frequency; however, effects were not consistently observed in all studies. Similar observations of increased flatulence have been reported following the consumption fructo-oligosaccharies (FOS) (15 g/day) over a 7-day period (Alles, 1996), and this symptom represents a localized effect that is expected in associationare electronically filed with the consumption of indigestible fiber in large quantities. There were no reports of events in other System Organ Class (SOC) suggestive of systemic toxicity.
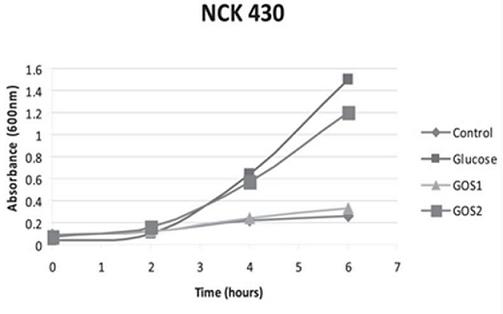
The significance of a higher purity GOS, namely RP-G28, was highlighted in a 2010 study by Klaenhammer. The in vitro study concluded that RP-G28 promoted growth of lactobacilli and bifidobacteria, but did not promote multiple strains of E. coli. In contrast, lower purity GOS stimulated both bifidobacteria as well as the strains of E. coli evaluated. (As seen below in Figure 1, NCK 430 (e. coli) grew in the presence of low purity GOS (GOS 2). Alternatively, the higher purity GOS (RP-G28/GOS 1) did not promote the growth of E. coli.).
Figure 1
Mechanism of Action
RP-G28 is understood to resist hydrolysis by salivary and intestinal enzymes dueor furnished to the configuration of their glycosidic bonds and consequently reach the colon virtually intact. The product is then broken down intracellularly by galactosidases, and eventually β-galactosidase hydrolyzes the terminal lactose. This leads to selective alterations in the composition and activity of the microbiome in which RP-G28 enhances the growth of lactose-metabolizing bacteria, including species of Bifidobacteria and Lactobacilli (30). In our Phase 2a Clinical Trial (G28-001), shifts in the fecal microbiome in 82% of participants on treatment and increases in relative abundance of both Bifidobacteria and Lactobacilli were reported. RP-G28 had a bifidogenic effect in 90% of responders, which included species Bifidobacterium longum, Bifidobacterium adolenscentis, Bifidobacterium catenulatum, Bifidobacterium breve, and Bifidobacterium dentium (30). The understood mechanism of action is that by increasing lactose-metabolizing bacteria, less undigested lactose is fermented, and thus reduces gas production and related LI symptoms. Data correlating bacterial taxa and symptom metadata support this proposed hypothesis. In the G28-001 study, microbiome changes correlated with clinical outcomes of improved lactose tolerance in which an increase in Bifidobacterium was associated with decreased pain and cramping outcomes.
Our Market Opportunity
Unmet Medical Needs
Lactose intolerance is a challenging condition to manage. According to a market research study conducted by Objective Insights in April 2012, approximately 60% of lactose intolerant sufferers reported experiencing symptoms daily, or bi-weekly. Not only can symptoms be painful and embarrassing, they can also dramatically affect one’s quality of life, social activities, and health. Currently there are few reliable, or effective, treatments available that provide consistent or satisfactory relief.
Currently, there is no approved prescription treatment for lactose intolerance. Most persons with lactose intolerance avoid ingestion of milk and dairy products while others substitute non-lactose-containing foods in their diet. However, complete avoidance of lactose-containing foods is difficult to achieve (especially for those with moderate to severe symptoms) and can lead to significant long-term morbidity (i.e., dietary deficiencies of calcium and vitamin D).
Treatment Options
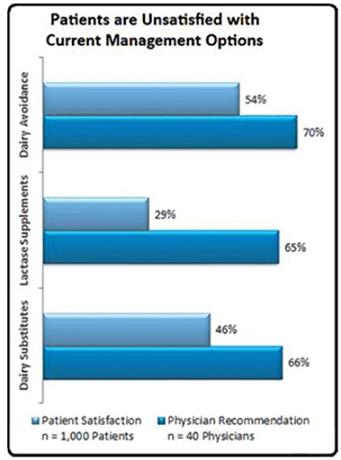
Doctors generally recommend the following treatments for the management of lactose intolerance: (1) dairy avoidance; (2) lactase supplements; (3) probiotics/dietary supplements; and (4) dairy substitutes/lactose free products. Despite educating their patients on all viable treatment options, physicians tend to advise their patients to refrain from consuming any dairy products whatsoever. However, in a 2008 survey conducted by Engage Health, 47% of lactose intolerance sufferers reported that this method was not effective (largely due to hidden dairy products in ingredients), and only 30% of lactose intolerance sufferers reported lactase supplements as being effective in managing their lactose intolerance.Further, while probiotics/dietary supplements have been demonstrated to aid and support one’s digestive system, helping break down general foods consumed, they don’t directly help with lactose intolerance. The 2008 survey by Engage Health suggests that the majority of lactose intolerance patients are dissatisfied with current treatment options.
Patients Unsatisfied with Current Management Options
Growing Awareness
Lactose intolerance is a condition that continues to expand as society advances and evolves. It has been estimated that gastroenterologists see approximately 15 new patients with lactose intolerance each month. Education and awareness have increased, and the American diet has greatly changed over the past decade to include more dairy-based goods. As the populace is growing older, the prevalence of lactose intolerance is increasing because more people tend to develop lactose intolerance later in life. Increased education and diagnosis is making more people aware of their allergies and digestive conditions. Physicians may compound the growth of lactose intolerance prevalence and its associated disorders by recommending individuals to avoid dairy products, a practice which in and of itself may increase severity of the intolerance.
Our Competitive Strengths
Market Opportunity
RP-G28 has the potential to become the first approved drug in the United States and Europe for the treatment of lactose intolerance.
Renowned Scientific Team and Management Team
Our leadership team has extensive biotechnology/pharmaceutical expertise in discovering, developing, licensing and commercializing therapeutic products. We have attracted a scientific team comprised of innovative researchers who are renowned in their knowledge and understanding of the host-microbiome in the field of lactose intolerance and gastroenterology.
Patent Portfolio
We have issued patents in the United States, in select countries in Europe (Germany, the United Kingdom, France, Spain, the Netherlands, Spain), and in other jurisdictions, directed to pharmaceutical compositions, methods of making such compositions, and methods of using such compositions for the treatment of lactose intolerance and certain of its symptoms. Additional worldwide patent applications are pending. The patent applications include claims covering pharmaceutical compositions, methods of making, methods of use, formulations and packaging.
In addition, in July 2015 we acquired the rights, title and interest to certain patents and related patent applications with claims covering a process for producing ultra-high purity galacto-oligosaccharide active pharmaceutical ingredients, including RP-G28 from our supplier. See “Manufacturing” for additional details regarding the second amendment to the exclusive supply agreement and our exercise of the exclusive option.
See “Intellectual Property” for additional information regarding our patent portfolio.
Our Growth Strategy
In order to achieve our objective of developing safe and effective applications to treat conditions associated with microbiome disfunctions, our near-term and long-term strategies include the following:
Clinical and Regulatory
IND Application/Phase 1
The IND for RP-G28 was activated initially to support a Phase 2a safety, tolerability and efficacy study in lactose intolerant patients. Standard Phase 1 single and repeat dose safety and tolerability studies in healthy volunteers were not needed because other GOS products that contain similar GOS constituents are generally regarded as safe (GRAS) and therefore supported the safety of RP-G28 in humans.
In 2018, a Phase 1 study will be conducted to understand the potential for systemic absorption of RP-G28 and any impact the presence of food may have on the pharmacokinetic profile of RP-G28. A Phase 1 QT/QTc study may be needed if there is measurable systemic exposure of RP-G28. Phase 1 systemic drug-drug-interaction (DDI) studies may also be needed if there is measurable systemic exposure of RP-G28, or if DDI potential within the gut is suggested by the results of in vitro DDI studies.
Phase 2a Study
We completed a double-blinded, randomized, multi-center, placebo-controlled Phase 2a clinical trial to validate the efficacy, safety and tolerance of RP-G28 compared to a placebo. We evaluated RP-G28 in 62 patients with lactose intolerance over a treatment period of 35 consecutive days. Post-treatment, subjects reintroduced dairy into their diets and were followed for an additional 30 days to evaluate lactose digestion, as measured by hydrogen production and symptom improvements. In order to confirm lactose intolerance and study participation, subjects underwent a 25-gram lactose challenge in the clinic. Lactose intolerance symptoms and hydrogen production via hydrogen breath test were assessed for six hours post-lactose dose. Eligible subjects were required to demonstrate a minimum symptom score and a “positive” hydrogen breath test in order to be eligible for randomization. A “positive” breath test was defined as a hydrogen gas elevation of 20 parts per million (ppm) at two time-points within the six hours following a lactose-loading dose. The primary endpoints included tracking patients’ gastrointestinal symptoms via a patient-reported symptom assessment instrument (a Likert Scale, measuring individual symptoms of flatulence, bloating, cramping, abdominal pain and diarrhea, on a scale of 0 (none) to 10 (worst)) at baseline, day 36 and day 66; as well as the measurement of hydrogen gas levels in their breath following a 25-gram lactose challenge.
Positive trends were seen when the entire per protocol study population was analyzed, including some statistically significant subgroup analysis, suggesting a therapeutically positive effect. Although there were few primary and secondary efficacy endpoints with statistically significant results, the combined data suggest that RP-G28 is exerting a positive therapeutic effect
Key findings of the Phase 2a clinical trial included:
Figure 2
The clinical results of our Phase 2a study were published in Nutrition Journal in a manuscript entitled “Improving lactose digestion and symptoms of lactose intolerance with a novel galacto-oligosaccharide (RP-G28): a randomized, double-blind clinical trial.” The microbiome results were published in the Proceedings of the National Academy of Science in a manuscript entitled“Impact of short-chain galacto-oligosaccharides on the gut microbiome of lactose-intolerant individuals.”
Type C Meeting with the FDA
We held a Type C meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products on February 2013. The purpose of the meeting was to obtain the FDA’s feedback on the planned Phase 2 program and Phase 3 programs, inform the FDA of our ongoing development plans, gain feedback on relevant clinical trial design and end points related to patient meaningful benefits, and to inform the FDA of the status of our product characterization.
Phase 2b Clinical Trial
Enrollment in our Phase 2b clinical trial of RP-G28 was initiated in March 2016 and completed in August 2016. The final patient completed dosing and all monitoring visits in October 2016.
The Phase 2b trial was a multi-center, randomized, double-blind, placebo-controlled, parallel-group trial of 368 subjects designed to determine the efficacy, safety, and tolerability of two dosing regimens of RP-G28 in subjects with moderate to severe lactose intolerance. Two hundred and forty-seven (247) subjects received RP-G28 while 121 subjects received placebo. Twenty-four (24) subjects were discontinued prematurely from the study and 344 (91.2%) completed the study.
The trial assessed patients with lactose intolerance symptoms as measured on a Likert scale after a lactose challenge. Entry criteria in the Phase 2b trial included a hydrogen breath test to validate lactase deficiency. The Phase 2b trial design included a screening period, a 30-day course treatment period, and a 30-day post-treatment “real world” observation period during which subjects were followed while lactose containing food products were re-introduced into their diets. The study was designed to escalate the dose beyond the 15 gm/day dose level evaluated in the Phase 2a study. Study subjects abstained from lactose containing food products and were then randomized evenly (1:1:1) to receive one of two doses of RP-G28 or placebo for 30 days.
The primary endpoint for the Phase 2b clinical trial was a LI symptom composite score response at day 31. A response was based on change from baseline (Day -7, visit 1) to end of treatment period at day 31 (visit 5), combined average of four maximum symptom scores taken over 0.5, 1, 2, 3, 4, and 5 hours for each symptom (abdominal pain, cramping, bloating, and gas movement) after a lactose challenge test. A response was defined as a 4-point or greater decrease from baseline or a composite score of zero at day 31. The Phase 2b trial further required the collection of fecal samples from patients enrolled to evaluate the baseline and changes to the patient’s microbiome that correlate to symptom reduction and lactose tolerance.
We held a Type C meeting with the FDA in March 2017, to discuss our development plans and Phase 2b clinical trial. The focus of the meeting was to obtain the FDA’s feedback on our Phase 2b clinical trial, including our SAP, prior to unblinding any data.
In order to gather long-term data on subjects exposed to RP-G28, we also offered enrollment in an observational 12-month extension study, G28-003XA, to subjects who completed the Phase 2b protocol. As RP-G28 is expected to provide extended relief from lactose intolerance symptoms beyond the initial 30-day treatment phase, this extension study for the Phase 2b program will assess the long-term treatment effect. The study is also evaluating each participant’s microbiome, expanding our knowledge of the effects that RP-G28 may have on adapting the gut microbiota in a beneficial manner. We completed this study in the fourth quarter of 2017. We intend for the results from this study to support durability of treatment and guide the need to evaluate an additional 30-day course of treatment in subjects who experience the return of lactose intolerance symptoms after an initial course of RP-G28.
Topline results of the Phase 2b clinical trial were announced in March 2017. Due to inconsistent data results from one study site, the data from this site was excluded from the primary analysis population (Efficacy Subset mITT). After excluding the data from the one anomalous study site, results showed a clinically meaningful benefit to subjects in the reduction of lactose intolerance symptoms across a variety of outcome measures. The majority of analyses showed positive outcome measures and the robustness of the data point to a clear drug effect. Treatment patients not only reported meaningful reduced symptoms, but also 30-days after taking the treatment, patients reported adequate relief from lactose intolerance symptoms and satisfaction with the results of the treatment, with RP-G28 preventing or treating their lactose intolerance symptoms. Greater milk and dairy product consumption was also reported by patients.
Because the efficacy data from one study site was found to be significantly different from that of the other study sites, the data from this site was excluded from the primary analysis population (Efficacy Subset mITT. n=296). It was decided that, in addition to the efficacy analysis for the mITT Population, the Efficacy Subset mITT population would be used to perform all efficacy analyses.
In the Efficacy Subset mITT Analysis group, the primary endpoint met statistical significance, (39.7% of the pooled dosing group compared to 25.8% of the placebo group responded (p=0.0159)). Because the primary analysis was statistically significant, the primary endpoint comparison between the high dose group and the placebo group was then tested and also met statistical significance (38.1% of the high dose group, compared to 25.8% of the placebo group responded (p=0.0294)). The comparison between the low dose group and the placebo group further met statistical significance (p=0.0434).
In the entire study population (mITT population), including patients from the excluded study site, taking at least one dose of drug (n=368), the comparison between the pooled treatment groups and the placebo group narrowly missed statistical significance (p=0.0618), (40.1% of the pooled treatment group responded compared to 31.4% of the placebo group). Both low dose and high dose group arms demonstrated a higher proportion of responders than the placebo group.
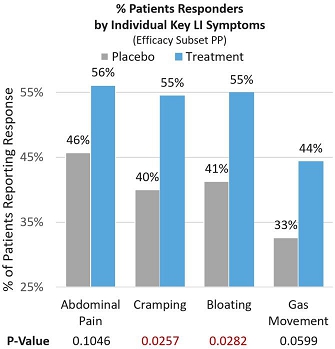
In the Efficacy Subset Per-protocol population (Efficacy Subset PP), significant and meaningful symptom improvement was consistently seen across key individual lactose intolerance symptoms by patients reporting a ≥4-point improvement from baseline (proportion of subjects on treatment that reported improvement in severity of each symptom). Of the treatment patients, 56.1% reported significant improvement in abdominal pain compared to 45.7% in the placebo group (p=0.1046). Of the treatment patients, 54.5% reported statistically significant improvement in cramping compared to 40.2% in the placebo group (p=0.0257). Of the treatment patients, 55% reported statistically significant improvement in bloating compared to 41.3% in the placebo group (p=0.0282). Finally, 44.4% of treatment patients reported significant improvement in gas movement compared to 32.6% in the placebo group (p=0.0599). See Figure 4 below.
Figure 4
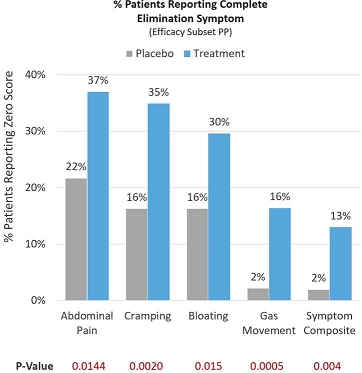
In a more stringent assessment, many patients reported that they experienced complete elimination of lactose intolerance symptoms, scoring a 0 out of 10 on a Likert pain scale post-treatment (Efficacy Subset PP). Of the treatment patients, 37.0% reported complete elimination of abdominal pain compared to 21.7% in the placebo group (p=0.0144). Of the treatment patients, 34.9% reported complete elimination of cramping compared to 16.3% in the placebo group (p=0.0020). Of the treatment patients, 29.6% reported complete elimination of bloating compared to 16.3% in the placebo group (p=0.015). Of the treatment patients, 16.4% reported complete elimination of gas movement compared to 2.2% in the placebo group (p=0.0005). Symptoms of abdominal pain, cramping, bloating and gas movement were then combined into a composite endpoint representing the key symptoms of lactose intolerance. Of the treatment patients, 13% experienced complete elimination of lactose intolerance symptoms compared to 2% in the placebo group (p=0.004). See Figure 5 below.
Figure 5
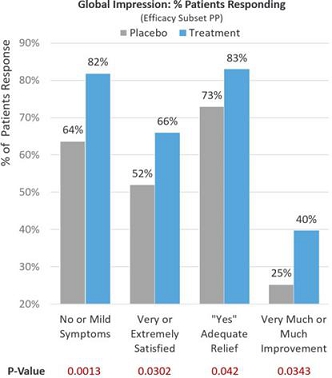
Observing global patient-reported assessments (Efficacy Subset PP) on multiple aspects of their symptom severity and treatment benefit experience 30 days after treatment and adding dairy and milk products back into their diets, 81.9% of treatment patients reported no or mild lactose intolerance symptoms compared to 63.7% in the placebo group (p=0.0013). Of the treatment patients, 66.3% reported being very or extremely satisfied with RP-G28 preventing or treating their lactose intolerance symptoms compared to 51.6% in the placebo group (p=0.0302). Of the treatment patients, 83.2% reported adequate relief from lactose intolerance symptoms compared to 72.5% in the placebo group (p=0.042). Of the treatment patients, 39.7% reported much or very much improvement in their lactose intolerance symptoms compared to 25.3% in the placebo group (p=0.0343). See Figure 6 below.
Figure 6
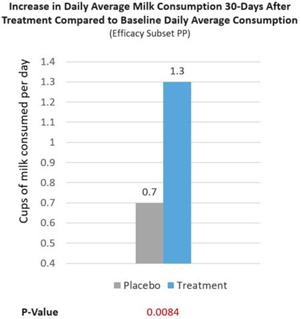
Further, a real-world milk intake assessment was conducted on treatment and placebo group patients (Efficacy Subset PP). At baseline, lactose intolerance patients reported consuming 0.2 cups/d of milk. After RP-G28, treatment patients increased their milk consumption to 1.5 cups/d of milk, consuming 1.3 cups/d more of milk (p=0.0084), 39% more milk consumed per day than placebo patients reported consuming (See Figure 7 below). We believe this is significant because the USDA recommends healthy individuals to consume 1.5 cups/d of milk. Overall, 62% of treatment patients consumed ≥1 cups/d of milk after being treated (p=0.0095). The increase in milk consumption is meaningful for dairy avoiders because it reflects increased lactose tolerance and may lead to more dietary calcium intake post-treatment as milk contains a higher percentage of one’s daily intake of calcium.
Figure 7
No serious adverse events related to treatment were reported and the number of adverse events reported was similar between treatment and placebo groups.
End-of-Phase 2 Meeting with the FDA
We held an End-of-Phase 2 meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products in August 2017. The purpose of the meeting was to obtain the FDA’s feedback on our planned Phase 3 program. We reached general consensus with the FDA on certain elements of our Phase 3 program and have received clear guidance and recommendations on many necessary components of our Phase 3 program; including the clinical, non-clinical, and chemistry, manufacturing and controls (CMC) requirements needed to support an NDA. We have incorporated much of this guidance into our Phase 3 program.
Elements of our Phase 3 program are expected to include the following:
| ||
In preparation for Phase 3, we have had regular communications with the FDA and have received feedback from the FDA on the Phase 3 protocols, the statistical analysis plan (SAP), non-clinical matters, chemistry and controls, as well as other items.
The FDA has provided the following recommendations with respect to our revised Phase 3 protocols, SAP and other items (all of which we intend to implement):
Nonclinical Safety Plans
Given the established safety profile of GOS in humans and the lack of significant safety concerns with RP-G28 administered to subjects in the Phase 2a and Phase 2b clinical trials, it was agreed with the FDA (August 2017 End-of-Phase 2 meeting) that no additional non-clinical safety studies are required to support continued evaluation of RP-G28 in the Phase 3 program. The FDA also agreed that no rat fertility, rat peri-post natal reproductive toxicity, genotoxicity or, importantly, rodent carcinogenicity studies are needed for the NDA submission.
As recommended by the FDA, we will continue to evaluate females of child-bearing potential who are willing to use appropriate contraception throughout the duration of any study. ICH-compliant embryo-fetal development toxicology studies of RP-G28 in the rat and rabbit will be conducted to support the NDA submission. Additional general toxicity studies may also need to be conducted for the NDA submission.
Manufacturing
We do not own or operate manufacturing facilities, nor do we have plans to develop our own manufacturing operations in the foreseeable future. We have an exclusive worldwide agreement (the “Supply Agreement”) to manufacture a higher purity form of GOS (referred to as “Improved GOS”) with Ricerche Sperimentali Montaleor (“RSM”) in connection with our clinical and nonclinical studies we will need to conduct prior to receiving regulatory approval for RPG-28. RSM has also agreed that it will not, except as necessary for RSM to perform its obligations under the Supply Agreement, market or sell Improved GOS, or any galacto-oligosaccharides that are of greater purity to any third party.
Pursuant to the terms of the Supply Agreement, as amended on July 24, 2015, we purchased the exclusive worldwide assignment of all right, title and interest to the Improved GOS (the “Improved GOS IP”) on July 30, 2015 for $800,000. We also issued 100,000 shares of our common stock to RSM pursuant to a stock purchase agreement.
Under the terms of the Supply Agreement, as amended, if we fail to make any future option payment required under the terms of the Supply Agreement, we may be required to return the Improved GOS IP to RSM. The terms of the Supply Agreement, as amended, require us to pay RSM $400,000 within 10 days following FDA approval of a new drug application for the first product owned or controlled by us using Improved GOS as its active pharmaceutical ingredient.
Commercialization
Given our stage of development, we have not yet established a commercial organization or distribution capabilities. RP-G28, if approved, is intended to be prescribed to patients suffering from lactose intolerance. These patients are normally under the care of a gastroenterologist and/or a primary care physician. Our current plan is to evaluate a possible partnership to commercialize RP-G28 for the treatment of lactose intolerance in patients in the United States and Europe if it is approved. We may also build our own commercial infrastructure or utilize contract reimbursement specialists, sales people and medical education specialists, and take other steps to establish the necessary commercial infrastructure at such time as we believe that RP-G28 is approaching marketing approval. Outside of the United States and Europe, subject to obtaining necessary marketing approvals, we will likely seek to commercialize RP-G28 through distribution or other collaboration arrangements for patients suffering from lactose intolerance.
Competition
The biopharmaceutical industry is characterized by intense competition and rapid innovation. Although we know of no drug candidate, other than RP-G28, in advanced clinical trials for treating lactose intolerance, other biopharmaceutical companies may be able to develop compounds or drugs that are able to achieve similar or better results. Our potential competitors include major multinational pharmaceutical companies, established biotechnology companies, specialty pharmaceutical companies and universities and other research institutions. Some of the pharmaceutical and biotechnology companies we expect to compete with include microbiome-based development companies such as Second Genome, Inc., Seres Health, Inc., Enterome SA, Vedanta Biosciences, Inc., and Rebiotix, Inc. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. We will also compete with providers of a wide variety of lactase supplements (the most widely used supplement in the United States being Lactaid®), probiotic/dietary supplements, and lactose-free and dairy-free products. We believe the key competitive factors that will affect the development and commercial success of our product candidates are efficacy, safety and tolerability profile, reliability, convenience of dosing, price and reimbursement.
Intellectual Property
The proprietary nature of, and protection for, our product candidates and our discovery programs, processes and know-how are important to our business. We have sought patent protection in the United States and internationally for uses of RP-G28 and our discovery programs, and any other inventions to which we have rights, where available and when appropriate. Our policy is to pursue, maintain and defend patent rights, whether developed internally or licensed from third parties, and to protect the technology, inventions and improvements that are commercially important to the development of our business. We also rely on trade secrets that may be important to the development of our business. We do not have composition of matter patent protection in the United States for RP-G28, which may result in competitors being able to offer and sell products so long as these competitors do not infringe any other patents that we hold, including patents directed to methods of manufacturing and purified RP-G28 or directed to methods of using RP-G28.
Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection of RP-G28 and any future product candidates and the methods used to develop and manufacture them, as well as successfully defending these patents against third-party challenges. Our ability to stop third parties from making, using, selling, offering to sell or importing our products depends on the extent to which we have rights under valid and enforceable patents or trade secrets that cover these activities. We cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents that may be granted to us in the future will be commercially useful in protecting our product candidates, discovery programs and processes from commercial competition. Furthermore, we cannot be sure that issued patents will not be challenged in court as invalid or in the Patent Office as unpatentable. For this and more comprehensive risks related to our intellectual property, please see “Risk Factors — Risks Relating to Our Intellectual Property.”
Patents and Proprietary Rights Covering Our Drug Candidates
We strive to protect our product candidates and exclusivity rights, as well as both maintain and fortify our position in the field of reduction of symptoms associated with lactose intolerance. We believe our intellectual property portfolio consists of early and broad filings in the area. We have focused on patents and patent applications directed to use of our products in disease treatment. We have sought and continue to seek the strongest possible intellectual property protection available to us in order to prevent others from directly competing with us, as well as to exclude competition around our products, their manufacture, and methods for use of the products in disease treatment. Our intellectual property portfolio directed to RP-G28 contains four issued patents relating to RP-G28 and its uses. That portfolio also includes at least 15 other related, pending patent applications in the United States and worldwide. We also own a patent family, including claims generally directed to processes for producing an improved form of galacto-oligosaccharides (GOS) mixtures (higher purity); this family includes issued patents in United States (not expiring until 2030), Italy (not expiring until 2029), and China, Germany, and the Netherlands (not expiring until 2030), as well as applications pending in the United States, Japan, India, and other jurisdictions that, if issued, will not expire until 2030.
This portfolio includes patents and proprietary rights related to:
We also are pursuing patent applications. These applications are pending in the United States, Europe, Japan and other jurisdictions, and, if they issue as patents, will not expire until at least 2030, and include claims generally directed to (i) oral dosage forms of a higher purity galacto-ologosaccharides (GOS), (ii) use of galacto-ologosaccharides (GOS) for treating lactose intolerance, and (iii) methods of preventing or reducing certain symptoms of lactose intolerance using galacto-ologosaccharides (GOS) dosage forms. For example, we have paid issue fees (and expect patents to issue in due course) in two United States patent applications: one includes claims directed to an oral dosage forms comprising a prebiotic composition comprising 95% or more galacto-oligosaccharides (GOS) by weight and less than 5% digestible saccharides by weight, in which the GOS comprises at least 45% by weight trisaccharides, and the other includes claims directed to oral dosage forms of GOS, comprising 0.1 to 10 g of a liquid GOS composition encapsulated in a gelatin capsule, in which the GOS composition comprises at least about 95% galacto-oligosaccharides (GOS) by weight, less than about 5% digestible saccharides by weight, and at least 45% by weight trisaccharides.
Trade Secrets
In addition to patents, we rely on trade secrets and know-how to develop and maintain our competitive position. Trade secrets and know-how can be difficult to protect. We seek to protect our proprietary processes, in part, by confidentiality agreements and invention assignment agreements with our employees, consultants, scientific advisors, contractors and commercial partners. These agreements are designed to protect our proprietary information. We also seek to preserve the integrity and confidentiality of our data, trade secrets and know-how by maintaining physical security of our premises and physical and electronic security of our information technology systems.
Government Regulation and Product Approval
Governmental authorities in the United States, at the federal, state and local level, and other countries extensively regulate, among other things, the research, development, testing, manufacture, labeling, packaging, promotion, storage, advertising, distribution, marketing and export and import of products such as those we are developing. Our product candidates must be approved by the FDA through the NDA process before they may be legally marketed in the United States and by the European Medicines Agency (the “EMA”) through the Marketing Authorization Application (“MAA”) process before they may be legally marketed in Europe. Our product candidates will be subject to similar requirements in other countries prior to marketing in those countries. The process of obtaining regulatory approvals and the subsequent compliance with applicable federal, state, local and foreign statutes and regulations require the expenditure of substantial time and financial resources.
United States Government Regulation
NDA Approval Processes
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act (the “FDCA”) and implemented regulations. Failure to comply with the applicable FDA requirements at any time during the product development process or approval process, or after approval, may subject an applicant to administrative or judicial sanctions, any of which could have a material adverse effect on us. These sanctions could include:
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
Once a pharmaceutical candidate is identified for development, it enters the preclinical or nonclinical testing stage. Nonclinical tests include laboratory evaluations of product chemistry, toxicity and formulation, as well as animal studies. A sponsor of an IND must submit the results of the nonclinical tests, together with manufacturing information and analytical data, to the FDA as part of the IND. Some nonclinical testing may continue even after the IND is submitted. In addition to including the results of the nonclinical studies, the IND will also include a clinical protocol detailing, among other things, the objectives of the clinical trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated if the first phase lends itself to an efficacy determination. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, notifies the sponsor of a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before clinical trials can begin. A clinical hold may occur at any time during the life of an IND, and may affect one or more specific studies or all studies conducted under the IND.
All clinical trials must be conducted under the supervision of one or more qualified investigators in accordance with GCPs. They must be conducted under protocols detailing the objectives of the trial, dosing procedures, research subject selection and exclusion criteria and the safety and effectiveness criteria to be evaluated. Each protocol must be submitted to the FDA as an amendment to the IND, and progress reports detailing the status of the clinical trials must be submitted to the FDA annually in the IND Annual Report. Sponsors must also report to the FDA, within required timelines, serious and unexpected adverse reactions, any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigation brochure, or any findings from other studies or animal or in vitro testing that suggest a significant risk in humans exposed to the drug. An institutional review board (“IRB”) at each institution participating in the clinical trial must review and approve the protocol before a clinical trial commences at that institution and must also approve the information regarding the trial and the consent form that must be provided to each research subject or the subject’s legal representative, monitor the study until completed and otherwise comply with IRB regulations.
Human clinical trials are typically conducted in three sequential phases that may overlap or be combined:
Human clinical trials are inherently uncertain and Phase 1, Phase 2 and Phase 3 testing may not be successfully completed. The FDA or the sponsor may suspend a clinical trial at any time for a variety of reasons, including a finding that the research subjects or patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients.
During the development of a new drug, sponsors are given opportunities to meet with the FDA at certain points. These points may be prior to the submission of an IND, at the end of Phase 2 and before an NDA is submitted. Meetings with the FDA may be granted at other times during the development program when requested. For instance, we held a Type C meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products on February 20, 2013. The purpose of the meeting was to obtain the FDA’s feedback on the planned clinical development program and future necessary clinical studies, inform the FDA of our ongoing development plans, gain feedback on relevant clinical trial design and end points related to patient meaningful benefits, and to inform the FDA of the status of our product characterization. Following analysis of the Phase 2a clinical trial, discussions with the FDA during the Type C Meeting in early 2013 about our clinical development plan, and further discussions with our regulatory consultants, we initiated the Phase 2b clinical trial of RPG-28 in March 2016 and completed final enrollment and dosing in October 2016.
FDA meetings can provide an opportunity for the sponsor to share information about the data gathered to date and for the FDA to provide advice on the next phase of development. Sponsors typically use the meeting at the end of Phase 2 to discuss their Phase 2 clinical results and present their plans for the pivotal Phase 3 clinical trial that they believe will support the approval of the new drug. Sponsors may request a Special Protocol Assessment (“SPA”), only if the sponsor has already had an end-of-phase 2/pre-phase 3 meeting or end-of-phase 1 meeting as appropriate. The purpose of the SPA is to reach agreement with the FDA on clinical trial protocol design and analysis that will form the primary basis of an efficacy claim.
According to published guidance on the SPA process, a sponsor which meets the prerequisites may make a specific request for a SPA and ask specific questions regarding the design and size of the proposed clinical trial. The FDA has a goal to evaluate the protocol within 45 days of the request to assess whether the proposed trial is adequate, and that evaluation may result in discussions and a request for additional information. A SPA request must be made before the proposed trial begins, and all open issues must be resolved before the trial begins. If a written agreement is reached, it will be documented and made part of the record. The agreement will be binding on the FDA and may not be changed by the sponsor or the FDA after the trial begins except with the written agreement of the sponsor and the FDA or if the FDA determines that a substantial scientific issue essential to determining the safety or efficacy of the drug was identified after the testing began.
Concurrent with clinical trials, sponsors usually complete additional animal safety studies and also develop additional information about the chemistry and physical characteristics of the drug and finalize a process for manufacturing commercial quantities of the product in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the drug and the manufacturer must develop methods for testing the identity, strength, quality, purity and potency of the drug. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the drug candidate does not undergo unacceptable deterioration over its proposed shelf-life.
The results of product development, nonclinical studies and clinical trials, along with descriptions of the manufacturing process, analytical tests and other control mechanisms, proposed labeling and other relevant information are submitted to the FDA as part of an NDA requesting approval to market the product. The submission of an NDA is subject to the payment of user fees, but a waiver of such fees may be obtained under specified circumstances. The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s threshold determination of whether it is sufficiently complete to permit substantive review. It may request additional information rather than accept an NDA for filing. In this event, the NDA must be resubmitted with the additional information. The resubmitted application also is subject to review before the FDA accepts it for filing.
Once the submission is accepted for filing, the FDA begins an in-depth review. NDAs receive either standard or priority review. A drug representing a significant improvement in treatment, prevention or diagnosis of disease may receive priority review. The FDA may refuse to approve an NDA if the applicable regulatory criteria are not satisfied or may require additional clinical or other data. Even if such data are submitted, the FDA may ultimately decide that the NDA does not satisfy the criteria for approval. The FDA reviews an NDA to determine, among other things, whether a product is safe and effective for its intended use and whether its manufacturing is cGMP-compliant. The FDA may refer the NDA to an advisory committee for review and recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will inspect the facility or facilities where the product is manufactured and tested.
Patent Term Restoration and Marketing Exclusivity
Depending upon the timing, duration and specifics of FDA marketing approval of RP-G28, one of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Act. The Hatch-Waxman Act permits a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND, and the submission date of an NDA, plus the time between the submission date of an NDA and the approval of that application. Only one patent applicable to an approved drug is eligible for the extension and the application for extension must be made prior to expiration of the patent. The United States Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we intend to apply for restorations of patent term for some of our currently owned or licensed patents to add patent life beyond their current expiration date, depending on the expected length of clinical trials and other factors involved in the submission of the relevant NDA.
Market exclusivity provisions under the FDCA also can delay the submission or the approval of certain applications. The FDCA provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to gain approval of an NDA for a new chemical entity. A drug is a new chemical entity if the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule or ion responsible for the action of the drug substance. During the exclusivity period, the FDA may not accept for review an abbreviated new drug application (“ANDA”), or a 505(b)(2) NDA submitted by another company for another version of such drug where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement. The FDCA also provides three years of marketing exclusivity for an NDA, 505(b)(2) NDA or supplement to an approved NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, for new indications, dosages or strengths of an existing drug. This three-year exclusivity covers only the conditions associated with the new clinical investigations and does not prohibit the FDA from approving ANDAs for drugs containing the original active agent. Five-year and three-year exclusivity will not delay the submission or approval of a full NDA; however, an applicant submitting a full NDA would be required to conduct or obtain a right of reference to all of the preclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
Pediatric Exclusivity and Pediatric Use
Under the Best Pharmaceuticals for Children Act (“BPCA”), certain drugs may obtain an additional six months of exclusivity, if the sponsor submits information requested in writing by the FDA (a Written Request) relating to the use of the active moiety of the drug in children. The FDA may not issue a Written Request for studies on unapproved or approved indications or where it determines that information relating to the use of a drug in a pediatric population, or part of the pediatric population, may not produce health benefits in that population.
We have not received a Written Request for such pediatric studies, although we may ask the FDA to issue a Written Request for such studies in the future. To receive the six-month pediatric market exclusivity, we would have to receive a Written Request from the FDA, conduct the requested studies in accordance with a written agreement with the FDA or, if there is no written agreement, in accordance with commonly accepted scientific principles, and submit reports of the studies. A Written Request may include studies for indications that are not currently in the labeling if the FDA determines that such information will benefit the public health. The FDA will accept the reports upon its determination that the studies were conducted in accordance with and are responsive to the original Written Request or commonly accepted scientific principles, as appropriate, and that the reports comply with the FDA’s filing requirements.
In addition, the Pediatric Research Equity Act (“PREA”) requires all applications (or supplements to an application) submitted under section 505 of the FDCA (21 U.S.C. §355) for a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration to contain a pediatric assessment unless the applicant has obtained a waiver or deferral. It also authorizes the FDA to require holders of approved NDAs for marketed drugs to conduct pediatric studies under certain circumstances. In general, PREA applies only to those drugs developed for diseases and/or conditions that occur in both the adult and pediatric populations. Products intended for pediatric-specific indications will be subject to the requirements of PREA only if they are initially developed for a subset of the relevant pediatric population.
As part of the Food and Drug Administration Safety and Innovation Act, Congress reauthorized both BPCA and PREA, which were slated to expire on September 30, 2012, and made both laws permanent.
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant orphan designation to a drug or biologic intended to treat a rare disease or condition, defined as a disease or condition with a patient population of fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 individuals in the United States and when there is no reasonable expectation that the cost of developing and making available the drug or biologic in the United States will be recovered from sales in the United States for that drug or biologic.
If a product that has orphan drug designation subsequently receives the first FDA approval for a particular active ingredient for the disease for which it has such designation, the product is entitled to orphan product exclusivity, which means that the FDA may not approve any other applications, including a full NDA, to market the same product for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity or if the FDA finds that the holder of the orphan drug exclusivity has not shown that it can assure the availability of sufficient quantities of the orphan drug to meet the needs of patients with the disease or condition for which the drug was designated. Orphan drug exclusivity does not prevent the FDA from approving a different drug or biologic for the same disease or condition, or the same drug or biologic for a different disease or condition. Among the other benefits of orphan drug designation are tax credits for certain research and a waiver of the NDA application user fee.
A designated orphan drug may not receive orphan drug exclusivity if it is approved for a use that is broader than the indication for which it received orphan designation. Orphan drug exclusive marketing rights in the United States also may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition.
We intend to explore orphan drug designation for RP-G28 for any orphan indication in which there is a medically plausible basis for treatment of the indication through colonic adaptation of gut bacteria.
Post-approval Requirements
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product may result in restrictions on the product or even complete withdrawal of the product from the market. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further FDA review and approval. In addition, the FDA may require testing and surveillance programs to monitor the effect of approved products that have been commercialized, and the FDA has the power to prevent or limit further marketing of a product based on the results of these post-marketing programs.
Any drug products manufactured or distributed by us pursuant to FDA approvals are subject to continuing regulation by the FDA, including, among other things:
Drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and some state agencies for compliance with cGMP and other laws.
We rely, and expect to continue to rely, on third parties for the production of clinical and commercial quantities of our products. Future FDA and state inspections may identify compliance issues at the facilities of our contract manufacturers that may disrupt production or distribution, or require substantial resources to correct.
From time to time, legislation is drafted, introduced and passed in Congress that could significantly change the statutory provisions governing the approval, manufacturing and marketing of products regulated by the FDA. In addition, FDA regulations and guidance are often revised or reinterpreted by the agency in ways that may significantly affect our business and our products. It is impossible to predict whether legislative changes will be enacted, or FDA regulations, guidance or interpretations changed or what the impact of such changes, if any, may be.
Regulation Outside of the United States
In addition to regulations in the United States, we will be subject to regulations of other countries governing clinical trials and commercial sales and distribution of our products. Whether or not we obtain FDA approval for a product, we must obtain approval by the comparable regulatory authorities of countries outside of the United States before we can commence clinical trials in such countries and approval of the regulators of such countries or economic areas, such as the European Union, before we may market products in those countries or areas. The approval process and requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from place to place, and the time may be longer or shorter than that required for FDA approval.
Under European Union regulatory systems, a company may submit marketing authorization applications either under a centralized or decentralized procedure. The centralized procedure, which is compulsory for medicines produced by biotechnology or those medicines intended to treat AIDS, cancer, neurodegenerative disorders or diabetes and optional for those medicines which are highly innovative, provides for the grant of a single marketing authorization that is valid for all European Union member states. The decentralized procedure provides for mutual recognition of national approval decisions. Under this procedure, the holder of a national marketing authorization may submit an application to the remaining member states. Within 90 days of receiving the applications and assessments report, each member state must decide whether to recognize approval. If a member state does not recognize the marketing authorization, the disputed points are eventually referred to the European Commission, whose decision is binding on all member states.
Reimbursement
Sales of our products will depend, in part, on the extent to which the costs of our products will be covered by third-party payors, such as government health programs, commercial insurance and managed healthcare organizations. These third-party payors are increasingly challenging the prices charged for medical products and services. Additionally, the containment of healthcare costs has become a priority of federal and state governments and the prices of drugs have been a focus in this effort. The U.S. government, state legislatures and foreign governments have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our net revenue and results. If these third-party payors do not consider our products to be cost-effective compared to other therapies, they may not cover our products after approved as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products on a profitable basis.
The Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (the “MMA”) imposed new requirements for the distribution and pricing of prescription drugs for Medicare beneficiaries. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities which will provide coverage of outpatient prescription drugs. Part D plans include both stand-alone prescription drug benefit plans and prescription drug coverage as a supplement to Medicare Advantage plans. Unlike Medicare Part A and B, Part D coverage is not standardized. Part D prescription drug plan sponsors are not required to pay for all covered Part D drugs, and each drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any formulary used by a Part D prescription drug plan must be developed and reviewed by a pharmacy and therapeutic committee. Government payment for some of the costs of prescription drugs may increase demand for our products for which we receive marketing approval. However, any negotiated prices for our products covered by a Part D prescription drug plan will likely be lower than the prices we might otherwise obtain. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payors.
The American Recovery and Reinvestment Act of 2009 provides funding for the federal government to compare the effectiveness of different treatments for the same illness. A plan for the research will be developed by the Department of Health and Human Services, the Agency for Healthcare Research and Quality and the National Institutes for Health, and periodic reports on the status of the research and related expenditures will be made to Congress. Although the results of the comparative effectiveness studies are not intended to mandate coverage policies for public or private payors, it is not clear what effect, if any, the research will have on the sales of any product, if any such product or the condition that it is intended to treat is the subject of a study. It is also possible that comparative effectiveness research demonstrating benefits in a competitor’s product could adversely affect the sales of our product candidates. If third-party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products on a profitable basis.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010 (collectively, the “ACA”), enacted in March 2010, is expected to have a significant impact on the health care industry. The ACA is expected to expand coverage for the uninsured while at the same time containing overall healthcare costs. With regard to pharmaceutical products, among other things, the ACA is expected to expand and increase industry rebates for drugs covered under Medicaid programs and make changes to the coverage requirements under the Medicare Part D program. We cannot predict the impact of the ACA on pharmaceutical companies, as many of the ACA reforms require the promulgation of detailed regulations implementing the statutory provisions which has not yet occurred. In addition, some members of the U.S. Congress have been seeking to repeal at least portions of the legislation and we expect they will continue to review and assess this legislation and alternative health care reform proposals. Any legal challenges to the ACA, as well as Congressional efforts to repeal the ACA, add to the uncertainty of the legislative changes enacted as part of the ACA.
In addition, in some non-U.S. jurisdictions, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the European Union provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European Union do not follow price structures of the United States and generally tend to be significantly lower.
Employees
As of the date of this Annual Report, we had nine employees, all of whom were full time employees. None of our employees are represented by a labor union, and we consider our relationship with our employees to be good.
Available Information
We file with the Securities and Exchange Commission (“SEC”)SEC: annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy andstatements, information statements, beneficial ownership reports and any amendments to those reports or statements filed or furnished pursuant to Sections 13(a), 14 andor 15(d) of the Securities Exchange Act of 1934, as amended. The public may obtain theseamended (the “Exchange Act”). All such filings at the SEC’s Public Reference Room at 100 F Street, NE, Washington, DC 20549 or by calling the SEC at 1-800-SEC-0330. The SEC also maintains aare available through our website at http://www.sec.gov that contains reports, proxy and information statements and other information regarding Ritter Pharmaceuticals, Inc. and other companies that file materials with the SEC electronically. As soon as practicable after filing with the SEC, we make copies of our reports on Form 10-K, Forms 10-Q and Forms 8-K available to the public, free of charge,charge. However, the information contained on or accessed through the investor relations tab on our web site,http://www.ritterpharmaceuticals.com/investors.
We operate in a dynamic and rapidly changing business environment that involves multiple risks and substantial uncertainty. The following discussion addresses risks and uncertainties that could cause, or contribute to causing, actual results to differ from expectations in material ways. In evaluating our business, investors should pay particular attention to the risks and uncertainties described below and in other sectionswebsite does not constitute part of this Annual Report, and references to our website address in this Annual Report are inactive textual references only. All such reports are also available free of charge via EDGAR through the SEC website at www.sec.gov.
| 9 |
Item 1A. Risk Factors
An investment in our subsequentcommon stock involves risks. You should carefully consider the risks described below, together with all of the other information included in this Annual Report, as well as in our other filings with the SEC. TheseSEC, in evaluating our business. If any of the following risks actually occur, our business, financial condition, operating results and uncertainties, or other eventsfuture prospects could be materially and adversely affected. In that we do not currently anticipate or that we currently deem immaterial also may affect our results of operations, cash flows and financial condition. Thecase, the trading price of our common stock could alsomay decline due to any of these risks, and you couldmight lose all or part of your investment. The followingrisks described below, which are the risks we judge (rightly or wrongly) to be the most significant to investors, are not the only ones we face. Additional risks that we currently do not judge to be among the “most significant” may also impair our business, financial condition, operating results and prospects.
Certain statements below are forward-looking statements. For additional information, should be read in conjunction with Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” andsee the financial statements and related notes included in Part II, Item 8, “Financial Statements and Supplementary Data”section of this Annual Report.Report under the caption “Cautionary Note Regarding Forward-Looking Statements.”
Risks RelatingRelated to Our Financial PositionBusiness Generally
Our business strategy is high-risk
We are focusing our resources and Needefforts on development of drug product candidates, which requires extensive cash needs for Additional Capitalresearch and development activities. This is a high-risk strategy because there is no assurance that that our cash resources will be adequate to develop our product candidates, that our product candidates will ever be proven to be safe and effective or that any products will ever become commercially viable. This makes our stock an unsuitable investment for many investors.
We do not currently have incurred netenough working capital to execute our strategic plan.
We have suffered recurring losses in each year since our inception. Currently, we have no products approved for commercial sale. As a result, our ability to reduce our losses and reach profitability is unknown,from operations, and we may never achieve or sustain profitability.
are now essentially a non-revenue company. We have incurred net losseswill need capital to maintain our operations and to support our intended development of our therapeutics business. Future financings will be necessary in each year since our inception. The accompanying financial statements have been prepared assuming that we will continueorder for us to survive as a going concern which contemplates, among other things, the realization of assets and satisfaction of liabilities in the normal course of business.
To date, we have devoted most ofto properly execute our financial resources to our corporate overhead and research and development, including our drug discovery research, preclinical development activities and clinical trials. We currently havestrategic plan. However, there can be no productsassurance that are approved for commercial sale. We expect to continue to incur net losses and negative operating cash flow for the foreseeablesuch future and we expect these losses to increase as we continue our development of, and seek regulatory approvals for, RP-G28, prepare for and begin the commercialization of RP-G28, and add infrastructure and personnel to support our product development efforts and operations. We anticipate that any such losses could be significant for the next several years as we begin our Phase 3 clinical trials for RP-G28 and related activities required for regulatory approval of RP-G28. If RP-G28 does not gain regulatory approval, or does not achieve market acceptance, we may never become profitable, unless we are able to develop and market some other product. Our net losses and negative cash flows have had, and will continue to have, an adverse effect on our stockholders’ equity and working capital.
Because of the numerous risks and uncertainties associated with pharmaceutical product development, we are unable to accurately predict the timing or amount of increased expenses or when, or if, we will be able to achieve profitability. In addition, our expenses could increase if we are required by the FDA or the EMA, to perform studies or trials in addition to those currently expected, or if there are any delays in completing our clinical trials or the development of RP-G28, or any other product candidates we may develop in the future. The amount of future net losses will depend, in part, on the rate of future growth of our expenses and our ability to generate revenues.
We will require substantial additional funding, which may not be available to us on acceptable terms, or at all, and, if not so available, may require us to delay, limit, reduce or cease our operations.
Developing pharmaceutical products, including conducting preclinical studies and clinical trials, is expensive. To complete the work necessary to file an NDA in the United States and a MAA in the European Union for RP-G28, which is currently anticipated to occur in 2019, we will require substantial additional funding. If the FDA or EMA requires that we perform additional nonclinical studies or clinical trials, our expenses would further increase beyond what we currently expect and the anticipated timing of any potential NDA or MAA would likely be delayed.
We will need to secure additional financing in order to complete clinical development of and commercialize RP-G28, if approved, and generally fund our operations. We can provide no assurances that any additional sources of financingfinancings will be available to us (or, if they are, that they can be consummated on favorable terms,desirable terms).
We may, in the short and long-term, seek to raise capital through the issuance of equity securities or through other financing sources. To the extent that we seek to raise additional funds by issuing equity or equity-linked securities, our stockholders may (as has already occurred several times) experience significant dilution. Any debt financing, if at all. If we are unable to obtain fundingavailable, may include financial and other covenants that could restrict our use of the proceeds from such financing or impose other business and financial restrictions on a timely basis,us. In addition, we may be requiredconsider alternative approaches such as licensing, joint venture, or partnership arrangements to significantly curtail our research and development program. We also could be required to seek funds through arrangements with collaborative partnersprovide short term or otherwise that may require us to relinquish rights to RP-G28 or otherwise agree to terms unfavorable to us.
We may sell additional equity or debt securities to fund our operations, which would result in dilution to our stockholders and imposed restrictions on our business.
We may seek additional funding through a combination of equity offerings, debt financings, government or other third-party funding, commercialization, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements.long term capital. Additional funding may not be available to us on acceptable terms, or at all. ToIn addition, any future financing (depending on the extent that we raise additional fundsterms and conditions) may be subject to the approval of Alpha, a related party and the holder of our 8% Senior Convertible Debenture and of our 8% Convertible Debenture (together, the “Debentures”), and/or trigger certain adjustments to the conversion prices of the Debentures or to the exercise prices of warrants held by issuing equity securities (including any common stock issued to Aspire Capital Fund, LLC (“Aspire Capital”) pursuant to our financing arrangement with Aspire Capital), our stockholders may experience significant dilution.Alpha and/or by other persons. See “Management’sPart II, Item 7 “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Aspire Capital Financing Arrangement.” Anyfor additional details regarding the Debentures.
Servicing our debt financing, if available, may involve restrictive covenants that impactwill require a significant amount of cash, and we do not expect to have sufficient cash flow from our business to pay this debt.
Our ability to conduct business.make payments to Alpha of principal or interest on the Debentures or to make any potential prepayments for the Debentures, to the extent applicable, depends on our future performance, which is subject to economic, financial, competitive and other factors beyond our control. Our cash resources currently on hand, plus any anticipated near-term cash flow from operations or dispositions, would not be sufficient to service our indebtedness and/or to make necessary expenditures.
In December 2022, we entered into a Securities Purchase Agreement with Alpha and in exchange for $3,000,000 in cash (less $50,000 for expense reimbursement) issued to Alpha our 8% Senior Convertible Debenture with an original face amount of $3,300,000 due on December 22, 2025 (the “2022 Debenture”), plus 2,500,000 common stock warrants exercisable (from June 22, 2023 through June 22, 2028) at $1.65 per share. Commencing June 1, 2023 and continuing on the first day of each month thereafter until the earlier of (i) December 22, 2025 and (ii) the full redemption of the 2022 Debenture (each such date, a “Monthly Redemption Date”), we must redeem $110,000 plus accrued but unpaid interest, liquidated damages and any amounts then owing under the 2022 Debenture (the “Monthly Redemption Amount”). The Monthly Redemption Amount must be paid in cash; provided that after the first two monthly redemptions, we may (if the Equity Conditions, as defined in the 2022 Debenture, are then satisfied or have been waived) elect to pay all or a portion of a Monthly Redemption Amount in shares of our common stock, based on a conversion price equal to the lesser of (i) the then applicable conversion price of the 2022 Debenture and (ii) 85% of the average of the VWAPs (as defined in the 2022 Debenture) for the five consecutive trading days ending on the trading day that is immediately prior to the applicable Monthly Redemption Date.
| 10 |
The 2022 Debenture accrues interest at the rate of 8% per annum, which began accruing on December 1, 2023, and will be payable on a quarterly basis. Interest may be paid in cash or shares of common stock or a combination thereof at our option; provided that the Equity Conditions have been satisfied.
Alpha has waived the Equity Conditions for certain Monthly Redemption Amounts, but Alpha is not required to continue such waivers beyond May 2024. For the foreseeable future, we do not expect to be able to satisfy the Equity Conditions; as a result, where there is no waiver of the Equity Conditions we would not have the opportunity to make 2022 Debenture payments in the form of stock rather than in the form of cash, even for types of payments for which payment in the form of stock would have been allowed.
The 2022 Debenture is convertible into our common stock at any time at the holder’s option; the conversion price was originally $1.32 but pursuant to a Securities Purchase Agreement amendment in December 2023 it was reduced to $0.73 and then in February 2024 it was adjusted downward to $0.26 per share by virtue of the operation of a “ratchet” antidilution provision. (The exercise price of the warrants issued with the 2022 Debenture was originally $1.65 but pursuant to a Securities Purchase Agreement amendment in December 2023 it was reduced to $0.73 and then in February 2024 it was adjusted downward to $0.26 per share by virtue of the operation of a “ratchet” antidilution provision.)
Other than the Monthly Redemption Amounts, the 2022 Debenture does not call for scheduled payments of principal before the scheduled maturity date.
Both the 2022 Debenture and the accompanying warrants provide for “ratchet” antidilution adjustments to their conversion price and exercise price.
Both the 2022 Debenture and the accompanying warrants include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
We granted Alpha resale registration rights for the common shares underlying the 2022 Debenture and the accompanying warrants.
The December 2023 amendment of the 2022 Debenture conversion price (and the accompanying warrants’ exercise price) to be $0.73 per share resulted in the 2022 Debenture’s then current $1,528,922 principal amount thereof becoming convertible into 2,094,414 shares of Company common stock (as opposed to the 1,158,274 shares into which such outstanding principal amount was convertible pre-adjustment). Also, the December 2023 amendment triggered a “ratchet” antidilution adjustment in the Company’s outstanding “exploding” “Series C Warrants,” resulting in such Series C Warrants becoming exercisable for 455,623 common shares (at an exercise price of $0.73 per share), as opposed to the 251,971 common shares into which such outstanding Series C Warrants would have been exercisable (at $1.32 per share) pre-adjustment. Finally, the $0.73 price triggered a “ratchet” antidilution adjustment in the exercise price of other outstanding Company common stock warrants, including 7,048 warrants held by Alpha and 67,620 warrants held by other persons, which were previously exercisable at $1.32 per share.
In February 2024, we entered into a Securities Purchase Agreement with Alpha and in exchange for $500,000 in cash (less $25,000 for expense reimbursement) issued to Alpha an 8% Convertible Debenture with a face amount of $550,000 due on December 31, 2024 (the “2024 Debenture”), plus 900,016 5-year common stock warrants exercisable at $0.26 per share. In addition, per this Securities Purchase Agreement Alpha obtained an option to purchase additional 8% Convertible Debentures, of like tenor, with face amounts of up to an aggregate of $1,100,000 (and with a proportional number of accompanying common stock warrants of like tenor, up to a total of 1,800,032 additional warrants), which would (if and when Alpha exercises such option) provide us up to an additional $1.0 million in cash proceeds (less expense reimbursement, and not including any possible cash proceeds from any future exercise of the additional warrants). This option is valid through July 1, 2024.
The 2024 Debenture has a maturity date of December 31, 2024 and is convertible, at any time, and from time to time, at Alpha’s option, into shares of our common stock, at $0.6111 per share. The 2024 Debenture does not call for scheduled payments of principal or interest before the scheduled maturity date. Interest on the 2024 Debenture accrues on its outstanding principal balance at the rate of 8% per annum.
Both the 2024 Debenture and the accompanying warrants provide for “ratchet” antidilution adjustments to the Conversion Price and Exercise Price.
Both the 2024 Debenture and the accompanying warrants include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
We granted Alpha “piggyback” registration rights for the common shares underlying the 2024 Debenture and the accompanying warrants.
| 11 |
The $0.26 exercise price of the warrants issued with the 2024 Debenture triggered a “ratchet” antidilution adjustment in the 2022 Debenture, resulting in the then current $1,198,922 principal amount thereof becoming convertible into 4,611,238 shares of Company common stock (as opposed to the 1,642,359 shares into which such outstanding principal amount were convertible pre-adjustment). Also, the $0.26 exercise price of the warrants issued with the 2024 Debenture triggered a “ratchet” antidilution adjustment in the Company’s outstanding “exploding” “Series C Warrants,” resulting in such Series C Warrants becoming exercisable for 1,279,261 common shares (at an exercise price of $0.26 per share), as opposed to the 455,623 common shares into which such outstanding Series C Warrants would have been exercisable (at $0.73 per share) pre-adjustment. Finally, the $0.26 exercise price of the Warrant would trigger a “ratchet” antidilution adjustment in the exercise price of other outstanding Company common stock warrants, including 2,507,048 warrants held by Alpha and 67,620 warrants held by other persons, all of which were previously exercisable at $0.73 per share.
If we continue to lack cash resources sufficient to service our indebtedness, we may be required to adopt one or more alternatives, such as selling assets, restructuring debt or issuing additional equity, equity-linked or debt instruments on terms that may be onerous or highly dilutive. Our ability to refinance our indebtedness will depend on the capital markets and our financial condition at such time. If we are not ableunable to raise additional capital when requiredengage in any of these activities or engage in these activities on acceptabledesirable terms, we may havebe unable to (i) significantly delay, scale backmeet our debt obligations, which would materially and adversely impact our business, financial condition and operating results or discontinue the development and/or commercializationeven put us out of RP-G28; (ii) seek collaborators for RP-G28 and possibly on terms thatbusiness.
Risks Related to Our Product Pipeline
Our product candidates are less favorable than might otherwise be available; or (iii) relinquish or otherwise dispose of rights related to RP-G28. In addition, the terms of any financing may adversely affect the holdings or the rights of our stockholders and the issuance of additional shares by us, or the possibility of such issuance, may cause the market price of our shares to decline.
Risks Relating to Regulatory Review and Approval of RP-G28
We cannot be certain that RP-G28 will receive regulatory approval, and without regulatory approval we will not be able to market RP-G28.
The development of a product candidate and issues relating to its approval and marketing are subject to extensive regulation by the FDAstill in the United States, the EMA in Europe, and regulatory authorities in other countries, with regulations differing from country to country. We are not permitted to market our product candidates in the United States or Europe untilearly stages of development. Although we receive approval of a NDA from the FDA or a MAA from the EMA, respectively. We have not submitted any marketing applications for RP-G28.
NDAs and MAAs must include extensive preclinical and clinical data and supporting information to establish the product candidate’s safety and effectiveness for each desired indication. NDAs and MAAs must also include significant information regarding the chemistry, manufacturing and controls for the product. Obtaining approval of a NDA or a MAA is a lengthy, expensive and uncertain process, and we may not be successful in obtaining approval. The FDA and the EMA review processes can take years to complete and approval is never guaranteed. If we submit a NDA to the FDA, the FDA must decide whether to accept or reject the submission for filing. We cannot be certain that any submissions will be accepted for filing and review by the FDA. Regulators of other jurisdictions, such as the EMA, have their own procedures for approval of product candidates. Even if a product is approved, the FDA or the EMA, as the case may be, may limit the indications for which the product may be marketed, require extensive warnings on the product labeling or require expensive and time-consumingbegun Phase 1a clinical trials or reporting as conditions of approval. Regulatory authorities in countries outside of the United States and Europe also have requirements for approval of drug candidates with whichQN-302, we must comply priormight be unable to marketing in those countries. Obtainingobtain further regulatory approval for marketing of a product candidate in one country does not ensure that we will be able to obtain regulatory approval inQN-302 or any other country. In addition, delays in approvals or rejections ofdrug candidate. We may never obtain marketing applications in the United States, Europe or other countries may be based upon many factors, including regulatory requests for additional analyses, reports, data, preclinical studies and clinical trials, regulatory questions regarding different interpretations of data and results, changes in regulatory policy during the period of product development and the emergence of new information regarding our product candidates or other products. Also, regulatory approval for any of our drug candidates.
We are still early in our Pan-RAS development efforts and have not yet sought approval for, or begun enrollment, in any clinical trials evaluating Pan-RAS. There can be no assurance that any of our drug product candidates will achieve success in their clinical trials or obtain regulatory approval.
Our ability to generate revenues from our drug product candidates will depend on the successful development and eventual commercialization of such drug candidates. The success of these products will depend on several factors, including the following:
| ● | successful completion of preclinical studies and clinical trials; | |
| ● | acceptance of an IND application by the FDA or other clinical trial or similar applications from foreign regulatory authorities for our future clinical trials for our pipeline; | |
| ● | timely and successful enrollment of patients in, and completion of, clinical trials with favorable results; | |
| ● | demonstration of safety, efficacy and acceptable risk-benefit profiles of our products to the satisfaction of the FDA and foreign regulatory agencies; | |
| ● | receipt and related terms of marketing approvals from applicable regulatory authorities, including the completion of any required post-marketing studies or trials; | |
| ● | obtaining and maintaining patent, trade secret and other intellectual property protection and regulatory exclusivity for our products; | |
| ● | developing and implementing marketing and reimbursement strategies; | |
| ● | establishing sales, marketing and distribution capabilities and launching commercial sales of our products, if and when approved, whether alone or in collaboration with others; | |
| ● | acceptance of our drugs, if and when approved, by patients, the medical community and third-party payors; | |
| ● | effectively competing with other therapies; | |
| ● | obtaining and maintaining third-party payor coverage and adequate reimbursement; and | |
| ● | maintaining a continued acceptable safety profile of the products following approval. |
| 12 |
Many of these factors are beyond our control, and it is possible that none of our drug candidates will ever obtain regulatory approval even if we expend substantial time and resources seeking such approval. If we do not achieve one or more of these factors in a timely manner or at all, we could experience significant delays or an inability to successfully commercialize our drug candidates. For example, our business could be harmed if results of the clinical trials of QN-302, Pan-RAS or any other drug candidates vary adversely from our expectations.
Drug development involves a lengthy and expensive process. We may incur additional costs or experience delays in completing, or ultimately be withdrawn.unable to complete, the development and commercialization of our drug product candidates.
Most drug candidates fail, and taking a drug candidate from concept through clinical trials and regulatory approval is not easy or guaranteed. We have completedare unable to predict when or if our drug candidates, will prove effective or safe in humans or will obtain marketing approval. Before obtaining marketing approval from regulatory authorities for the sale of these products, we must complete preclinical development and then conduct extensive clinical trials to demonstrate the safety and efficacy of these products for humans. Clinical testing is expensive, difficult to design and implement, can take many years to complete and is uncertain as to the outcome. A failure of one or more clinical trials can occur at any stage of testing. The outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim or preliminary results of a Phase 2a clinical trial and a Phase 2bdo not necessarily predict final results.
We may experience numerous unforeseen events that could delay or prevent our ability to obtain marketing approval or commercialize our drug candidates, including:
| ● | we may not be able to obtain enough capital to begin clinical trials ort to complete any already-begun clinical trials, or to complete any necessary preclinical studies; | |
| ● | regulators or IRBs or ECs may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site; | |
| ● | we may experience delays in reaching, or fail to reach, agreement on acceptable clinical trial contracts or clinical trial protocols with prospective trial sites; | |
| ● | clinical trials for our drug candidates may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical trials, delay clinical trials or abandon product development programs; | |
| ● | the number of patients required for clinical trials for our drug candidates may be larger than we anticipate, enrollment in these clinical trials may be slower than we anticipate, participants may drop out of these clinical trials at a higher rate than we anticipate or the duration of these clinical trials may be longer than we anticipate; | |
| ● | competition for clinical trial participants from investigational and approved therapies may make it more difficult to enroll patients in our clinical trials; | |
| ● | our third-party contractors may fail to meet their contractual obligations to us in a timely manner, or at all, or may fail to comply with regulatory requirements; | |
| ● | we may have to suspend or terminate clinical trials for our drug candidates for various reasons, including a finding that the participants are being exposed to unacceptable health risks; | |
| ● | our drug candidates may have undesirable or unexpected side effects or other unexpected characteristics, causing us or our investigators, regulators or IRBs/ECs to suspend or terminate the trials; | |
| ● | the cost of clinical trials for our drug candidates may be greater than we anticipate; and | |
| ● | the supply or quality of our drug candidates, or other materials necessary to conduct clinical trials may be insufficient or inadequate and result in delays or suspension of our clinical trials. |
Our product development costs will increase if we experience delays in preclinical studies or clinical trial for RP-G28.trials or in obtaining marketing approvals. We held an End-of-Phase 2 meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products in August 2017, regarding the path forward for RP-G28. We reached general consensus with the FDA on many elementsdo not know whether any of our Phase 3 program and received clear guidance and recommendationsplanned preclinical studies or clinical trials will begin on many other necessary components of our Phase 3 program; including the clinical, non-clinical, and chemistry, manufacturing and controls (CMC) requirements needed to support an NDA submission. However, nota timely basis or at all, clinical, non-clinical, and CMC items have been agreed to with the FDA, and remaining items will need to be reviewed by the agency and agreed to by us.
We will also need to conduct rat and embryo-fetal development toxicity studies.
Additional non-clinical development may be required to be conducted based on future FDA feedback and guidance. We cannot predict whether our future trials and studiesrestructured or will be successfulcompleted on schedule, or whether regulators will agree withat all. For example, the FDA may place a partial or full clinical hold on any of our conclusions regarding the preclinical studies and clinical trials we have conducted to date.for a variety of reasons.
If we are unable to obtain approval from the FDA, the EMA or other regulatory agencies for RP-G28, we will not be able to market RP-G28. If we are unable to market RP-G28, we may not be able to ever become profitable.
The FDA and other regulatory agencies outside the United States, such as the EMA, may not agree to our proposed endpoint for approval of RP-G28 for the treatment of lactose intolerance in patients, in which case we would need to complete additionalSignificant preclinical or clinical trials before seeking market approval.
We do not know if the FDA, the EMA or regulatory authorities in other countries will agree with our final primary endpoint for approval of RP-G28. The FDA, the EMA and regulatory authorities in other countries intrial delays also could shorten any periods during which we may seek approval forhave the exclusive right to commercialize our drug candidates or allow our competitors to bring products to market before we do and market RP-G28, may require additional nonclinical studies and/or clinical trials priorimpair our ability to granting approval, if at all. It may be expensive and time consuming to conduct and complete additional nonclinical studies and clinical trials that the EMA and other regulatory authorities may require us to perform. As such, any requirement by the EMA or other regulatory authorities that we conduct additional nonclinical studies or clinical trials could materially and adversely affectsuccessfully commercialize our business, financial condition and results of operations. Furthermore, even if we receive regulatory approval of RP-G28 for the treatment of lactose intolerance in patients, the labeling for RP-G28 in the United States, Europe or other countries in which we seek approval may include limitations that could impact the commercial success of RP-G28.drug candidates.
DelaysAny delays in the commencement enrollment andor completion, or termination or suspension, of our current clinical trial or our future clinical trials, if any, could result in increased costs to us, and delay or limit our ability to obtaingenerate revenue and adversely affect our commercial prospects.
Before we can initiate clinical trials of a drug candidate, we must submit the results of preclinical studies to the FDA along with other information as part of an IND application or similar regulatory filing, and the FDA (or corresponding foreign regulatory body) must approve the application.
Before obtaining marketing approval from the FDA for RP-G28.the sale of QN-302, Pan-RAS or any other future drug candidate, we must conduct extensive clinical studies to demonstrate safety and efficacy. Clinical testing is expensive, time consuming and uncertain as to outcome. The FDA may require us to conduct additional preclinical studies for any drug candidate before it allows us to initiate clinical trials under any IND application, which may lead to additional delays and increase the costs of our preclinical development programs.
DelaysAny delays in the commencement enrollmentor completion of our ongoing, planned or future clinical trials could significantly increase our costs, slow down our development and approval process and jeopardize our ability to commence product sales and generate revenues. We do not know whether our planned trials will begin on time or at all, or be completed on schedule, if at all. The commencement and completion of clinical trials could increase our product development costs or limit the regulatory approval of RP-G28. The commencement, enrollment and completion of clinical trials maycan be delayed or suspended for a varietynumber of reasons, including:including delays related to:
| ● | ||
| ● | ||
| ● | ||
| ● | obtaining approval from one or | |
| ● | IRBs/ECs refusing to approve, suspending or terminating the | |
| ● | changes to clinical trial protocol; | |
| ● | clinical sites deviating from trial protocol or dropping out of a trial; | |
| ● | failing to manufacture or obtain sufficient quantities of drug candidate, or, if applicable, combination therapies for use in clinical trials; | |
| ● | ||
| ● | ||
| ● | lack of adequate funding to continue the clinical trial; | |
| ● | patients experiencing severe or unexpected drug-related adverse | |
| ● | occurrence of serious adverse events in trials of the same class of agents conducted by other companies; | |
| ● | selecting or being required to use clinical end points that require prolonged periods of clinical observation or analysis of the resulting data; | |
| ● | a facility manufacturing our drug candidates, or any of their components, including without limitation, our own facilities being ordered by the FDA to temporarily or permanently shut down due to violations of cGMP, regulations or other applicable requirements, or infections or cross-contaminations in the manufacturing process; | |
| ● | lack of stability of our clinical trial material or any quality issues that arise with the clinical trial material; |
| 14 |
| ● | ||
| ● | ||
| ● | any third-party contractors becoming debarred or suspended or otherwise penalized by the FDA or other government or regulatory authorities for violations of regulatory requirements, in which case we may need to find a substitute contractor, and we may not be able to use some or all of the data produced by such contractors in support of our marketing applications. |
ChangesWe could also encounter delays if a clinical trial is suspended or terminated by us, by the IRBs/ECs of the institutions in which such trials are being conducted, by a Data Safety Monitoring Board for such trial or by the FDA. Such authorities may impose such a suspension or termination due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or trial site by the FDA resulting in the imposition of a clinical hold, unforeseen safety issues or adverse side effects, failure to demonstrate a benefit from using the product under investigation, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial. In addition, changes in regulatory requirements and guidancepolicies may also occur, and we may need to amend clinical trial protocols to reflectcomply with these changes with appropriate regulatory authorities.changes. Amendments may require us to resubmit our clinical trial protocols to IRBsIRBs/ECs for re-examination,reexamination, which may impact the costs, timing or successful completion of a clinical trial.
If we experience delays or difficulties enrolling patients in our ongoing or planned clinical trials, our receipt of necessary regulatory approval could be delayed or prevented.
We may not be able to initiate or continue our ongoing or planned clinical trials for our products if we are unable to identify and enroll a sufficient number of eligible patients to participate in these trials as required by the FDA. In addition, some of our competitors may have ongoing clinical trials for products that would treat the same patients as QN-302 or Pan-RAS, and patients who would otherwise be eligible for our clinical trials may instead enroll in clinical trials of our competitors’ products. In addition, introduction of new drugs or devices to the marketplace may have an effect on the number of patients available or timing of the availability of the patients.
Our inability to enroll a sufficient number of patients for our clinical trials would result in significant delays or may require us to abandon one or more clinical trials altogether.
Adverse side effects or other safety risks associated with our drug product candidates could delay or preclude approval, cause us to suspend or discontinue any clinical trials or abandon further development, limit the commercial profile of an approved label, or result in significant negative consequences following regulatory approval, if any.
Results of our current and planned clinical trials could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics. Undesirable side effects caused by our products could result in the delay, suspension or termination of clinical trials by us or the FDA for a number of reasons.
Moreover, if our products are associated with undesirable side effects in clinical trials or have characteristics that are unexpected, we may elect to abandon or limit their development to more narrow uses or subpopulations in which the undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk-benefit perspective, which may limit the commercial expectations for our products, if approved. We may also be required to modify our study plans based on findings in our clinical trials. Many drug candidates that initially showed promise in early stage testing have later been found to cause side effects that prevented further development. In addition, regulatory authorities may draw different conclusions or require additional testing to confirm these determinations.
It is possible that as we test our drug candidates in larger, longer and more extensive clinical trials, including with different dosing regimens, or as the use of our drug candidates becomes more widespread following any regulatory approval, illnesses, injuries, discomforts and other adverse events that were observed in earlier trials, as well as conditions that did not occur or went undetected in previous trials, will be reported by patients.
The development and commercialization of pharmaceutical and device products are subject to extensive regulation, and we may not obtain regulatory approvals for any product candidates, on a timely basis or at all.
The clinical development, manufacturing, labeling, packaging, storage, recordkeeping, advertising, promotion, export, import, marketing, distribution, adverse event reporting, including the submission of safety and other post-marketing information and reports, and other possible activities relating to drug product candidates such as ours are subject to extensive regulation.
| 15 |
We rely, and intend to continue to rely, on third parties to conduct our preclinical studies and clinical trials and perform some of our research and preclinical studies. If these third parties do not satisfactorily carry out their contractual duties, fail to comply with applicable regulatory requirements or do not meet expected deadlines, our development programs may be delayed or subject to increased costs or we may be unable to obtain regulatory approval.
We are dependent on third parties to conduct our planned preclinical studies and clinical trials of our drug product candidates. The timing of the initiation and completion of these trials will therefore be partially controlled by such third parties and may result in delays to our development programs. We have relied heavily on UofL for preclinical studies related to Pan-RAS, and we expect to rely heavily on CROs and sponsored academic researchers for any further preclinical studies. As to any clinical trials, we expect to rely on CROs, sponsored academic researchers, clinical investigators and/or consultants to play a significant role in the conduct of these trials and the subsequent collection and analysis of data. However, we will not be able to control all aspects of their activities. Nevertheless, we are responsible for ensuring that each clinical trial is conducted in accordance with the applicable protocol and legal, regulatory and scientific standards, including GCP, requirements, and our reliance on the CROs and other third parties does not relieve us of our regulatory responsibilities.
There is no guarantee that any such CROs, clinical trial investigators and/or other third parties on which we rely will devote adequate time and resources to our development activities or perform as contractually required. If any of these third parties fail to meet expected deadlines, adhere to our clinical protocols or meet regulatory requirements, otherwise perform in a substandard manner, or terminate their engagements with us, the timelines for our development programs may be extended or delayed or our development activities may be suspended or terminatedterminated. If one of our clinical trial site terminates for any reason, we may experience the loss of follow-up information on subjects enrolled in such clinical trial unless we are able to transfer those subjects to another qualified clinical trial site, which may be difficult or impossible.
If these third parties do not successfully carry out their contractual duties, meet expected deadlines or conduct clinical trials in accordance with regulatory requirements or our stated protocols, we will not be able to obtain, or may be delayed in obtaining, regulatory approvals for our drug product candidates and will not be able to, or may be delayed in our efforts to, successfully commercialize our products.
Manufacturing pharmaceutical products is complex and subject to product loss for a variety of reasons. We contract with third parties for the manufacture of our product candidates for preclinical testing and clinical trials and expect to continue to do so for commercialization. This reliance on third parties increases the risk that we will not have sufficient quantities of our product candidates or such quantities at an acceptable cost or quality, which could delay, prevent or impair our development or commercialization efforts.
We rely, and expect to continue to rely, on third parties for the manufacture of our products for preclinical and any time by us, ourclinical testing, as well as for future collaborators, the FDA or other regulatory authorities due to a number of factors, including:
In addition,commercial manufacture if we or any of our potential future collaborators are required to conduct additional clinical trials or other nonclinical studies of RP-G28 beyond those contemplated, our ability toproduct candidates obtain regulatory approval of RP-G28 and to generate revenue from its sales would be similarly harmed.
Clinical failure can occur at any stage of clinical development and may prevent us from receiving regulatory approval. The results of earlier clinical trials related to RP-G28 areThis reliance on third parties increases the risk that we will not necessarily predictive of future results.
Clinical failure can occur at any stagehave sufficient quantities of our clinical development. Clinical trials may produce negativeproduct candidates or inconclusive results, and wesuch quantities at an acceptable cost or quality, which could delay, prevent or impair our collaborators may decide,development or regulators may require us, to conduct additional clinical trials or nonclinical studies. In addition, data obtained from trials and studies are susceptible to varying interpretations, and regulators may not interpret our data as favorably as we do, which may delay, limit or prevent regulatory approval. Successful results from preclinical studies and early clinical trials do not ensure that subsequent clinical trials will generate the same or similar results or otherwise provide adequate data to demonstrate the efficacy and safety of a product candidate. A number of companies in the pharmaceutical industry, including those with greater resources and experience than us, have suffered significant setbacks in later clinical trials, even after seeing promising results in earlier clinical trials.commercialization efforts.
In addition, the design of a clinical trial can determine whether its results will support approval of a product and flaws in the design of a clinical trial may not become apparent until the clinical trial is well-advanced. We may be unable to design and execute a clinical trialestablish any agreements with third-party manufacturers or to support regulatory approval. Further, clinical trials of potential products often reveal that it is not practical or feasibledo so on favorable terms. Even if we are able to continue development efforts.establish agreements with third-party manufacturers, reliance on third-party manufacturers entails additional risks, including:
| ● | reliance on the third-party for regulatory, compliance and quality assurance; | |
| ● | operations of our third-party manufacturers or suppliers could be disrupted by conditions unrelated to our business or operations, including the bankruptcy of the manufacturer or supplier or the issuance of an FDA Form 483 notice or warning letter; | |
| ● | the possible breach of the manufacturing agreement by the third-party; and | |
| ● | the possible termination or nonrenewal of the agreement by the third-party at a time that is costly or inconvenient for us. |
In some instances, there can be significant variability in safety and/or efficacy results between different trials of the same product candidate due to numerous factors, including changes in trial protocols, differences in composition of the patient populations, adherence to the dosing regimen and other trial protocols and the rate of dropout among clinical trial participants. We do not know whether the clinical trials that we orhave manufacturing agreements in place for any of our potential future collaborators may conduct will demonstrate the consistent or adequate efficacycurrent drug candidates. We acquire many key materials on a purchase order basis. As a result, we do not have long-term committed arrangements with respect to our product candidates and safety that would be required toother materials. If we obtain regulatory approval and market RP-G28. Our Phase 3for any of our product candidates, we will need to establish an agreement for commercial manufacture with a third-party.
Any performance failure on the part of our existing or future manufacturers could delay clinical trialsdevelopment or regulatory approval. We do not currently have arrangements in place for RP-G28 may not provide sufficient support for NDA approval.
The FDA may require us to conduct one or more additional clinical trials, possibly involving a larger sample sizeredundant supply or a different clinical trial design,second source for bulk drug substance for QN-302 or may require longer follow-up periods. Pan-RAS.
| 16 |
We will need to seek and enter into out-licenses or collaborations with third parties for the development and commercialization of our products, resulting in a limitation of our upside potential.
We expect that we will need third-party out-licensees or collaborators for the development and commercialization of our products.
Our likely collaborators for any collaboration arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies. We face significant competition in seeking appropriate collaborators. Our ability to reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration and the proposed collaborator’s evaluation of a number of factors.
If we do enter into any such arrangements with any third parties, we will likely have limited control over the amount and timing of resources that such collaborators dedicate to the development or commercialization of our products. Our ability to generate revenues from these arrangements will depend on our collaborators’ abilities and efforts to successfully perform the functions assigned to them in these arrangements.
Any out-license or collaboration will necessarily result in a sharing of economics with the out licensee or collaborator, which might otherwise have been captured by us directly.
Risks Related to our Intellectual Property
If we are unable to bring RP-G28obtain and maintain sufficient patent protection for our therapeutic product candidates, or if the scope of the patent protection is not sufficiently broad, third parties, including our competitors, could develop and commercialize products similar or identical to market,ours, and our ability to create long-term stockholder value will be limited.
RP-G28 may have undesirable side effects which may delay or prevent marketing approval, or, if approval is received, require them to be taken off the market, require them to include safety warnings or otherwise limit their sales.
There were no notable differences observed between placebo-treated subjects and RP-G28-treated subjects in the Phase 2b trial. However, unforeseen side effects from RP-G28, could arise at any time during clinical development or, if approved, after the approved product has been marketed. Any undesirable or unacceptable side effects associated with RP-G28 could interrupt, delay or halt clinical trials, and results in delay of, or failure to obtain, marketing approval from the FDA and other regulatory authorities.
In addition:
Any of these events could substantially increase commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from the sale of our products.
Reimbursement decisions by third-party payors may have an adverse effect on pricing and market acceptance. If there is not sufficient reimbursement for our products, it is less likely that they will be widely used.
Market acceptance and sales of RP-G28, or any other product candidates we develop in the future, will depend on reimbursement policies andsuccessfully may be affected by, among other things, future healthcare reform measures. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drugs they will cover and establish payment levels. We cannot be certain that reimbursement will be available for RP-G28 or any other product candidates that we may develop in the future. Also, we cannot be certain that reimbursement policies will not reduce the demand for, or the price paid for, our products. If reimbursement is not available or is availableadversely affected.
Our commercial success depends significantly on a limited basis, we may not be able to successfully commercialize RP-G28, or other product candidates that we develop in the future.
In the United States, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (the “MMA”), changed the way Medicare covers and pays for pharmaceutical products. The legislation established Medicare Part D, which expanded Medicare coverage for outpatient prescription drug purchases by the elderly but provided authority for limiting the number of drugs that will be covered in any therapeutic class. The MMA also introduced a new reimbursement methodology based on average sales prices for physician-administered drugs. Any negotiated prices for our products covered by a Part D prescription drug plan will likely be lower than the prices we might otherwise obtain in the United States. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payors.
The United States and several other jurisdictions are considering, or have already enacted, a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sellprotect our products profitably. Among policy makersproprietary (and exclusively in-licensed) product candidates or technologies that we believe are important to our business, including pursuing, obtaining and payorsmaintaining patent protection in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems withother countries intended to cover the stated goalscomposition of containing healthcare costs, improving quality and/or expanding access to healthcare. In the United States, the pharmaceutical industry has been a particular focusmatter of these efforts and has been significantly affected by major legislative initiatives. We expect to experience pricing pressures in connection with the sale of RP-G28, and any otherour product candidates, the methods of use, related technologies, and other inventions that are important to our business. In addition to patent protection, we also rely on trade secrets to protect aspects of our business that are not amenable to, or that we develop in the future, due to the trend toward managed healthcare, the increasing influence of health maintenance organizations and additional legislative proposals.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010 (collectively, the “ACA”), enacted in March 2010, is a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirementsdo not consider appropriate for, healthcare and health insurance industries, impose new taxes and fees on health industry and impose additional health policy reforms. With regard to pharmaceutical products, among other things, the ACA is expected to expand and increase industry rebates for drugs covered under Medicaid programs and make changes to the coverage requirements under Medicare Part D program. Although it is too early to determine the full effect of the ACA, the law appears likely to continue the pressure on pharmaceutical pricing, especially under the Medicare program, and may also increase our regulatory burdens and operating costs. Since its enactment, there have been judicial and Congressional challenges to certain aspects of the ACA. Congress and President Trump have expressed their intentions to repeal and replace the ACA. President Trump issued an Executive Order and both chambers of Congress passed bills, all with the goal of fulfilling their intensions. However, to date, the Executive Order has had limited effect and the Congressional activities have not resulted in the passage of a law. If a law is enacted, many if not all of the provisions of the ACA may no longer apply to prescription drugs.
patent protection. If we do not adequately pursue, obtain, protection undermaintain, protect or enforce our intellectual property, third parties, including our competitors and/or collaborators, may be able to erode or negate any competitive advantage we may have, which could harm our business and ability to achieve profitability.
Moreover, depending on the Hatch-Waxman Actterms of any license agreements to which we may become a party, we may not have the right to control the preparation, filing, and similar legislation outsideprosecution of patent applications, or to maintain or defend the patents, covering technology licensed from third parties. Therefore, these patents and patent applications may not be prosecuted and enforced in a manner consistent with the best interests of our business.
We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patents, whether any issued patents will be found invalid and unenforceable or will be threatened by third parties or whether any issued patents will effectively prevent others from commercializing competing technologies and product candidates. Our licensors have not filed patent applications in every jurisdiction, and some filings are only pending in the United States.
Moreover, because the issuance of a patent, although presumptive, is not conclusive as to its inventorship, scope, validity or enforceability, our licensors’ patents or pending patent applications may be challenged in the courts or patent offices in the United States by extendingand abroad. Such challenges may result in loss of exclusivity or in the patent termsclaims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others from using or commercializing similar or identical products and obtaining data exclusivity fortechnologies or limit the duration of the patent protection of our products and technologies. Such challenges also may result in substantial cost and require significant time from our scientists and management, even if the eventual outcome is favorable to us.
Our licensors’ pending and future patent applications may not result in patents being issued that protect our product candidates and technologies, in whole or in part, or that effectively prevent others from commercializing competitive products and technologies. Even if the patent applications issue as patents, they may not issue in a form that will provide us with any meaningful protection, prevent competitors or other third parties from competing with us or otherwise provide us with any competitive advantage. Our competitors and other third parties may be able to circumvent our licensors’ patents by developing similar or alternative products or technologies in a non-infringing manner. Our competitors and other third parties may also seek approval to market their own products and technologies similar to or otherwise competitive with our products and technologies. Alternatively, our competitors or other third parties may seek to market generic versions of any approved products by submitting abbreviated NDAs to the FDA during which process they may claim that patents owned by us are invalid, unenforceable or not infringed. In these circumstances, we may need to defend or assert our licensors’ patents, or both, including by filing lawsuits alleging patent infringement. In any of these types of proceedings, a court or other agency with jurisdiction may find our licensors’ patents invalid or unenforceable, or that our competitors are competing in a non-infringing manner. Thus, even if we have in-licensed valid and enforceable patents, these patents still may not provide protection against competing products or processes sufficient to achieve our business may be materially harmed.objectives.
| 17 |
Depending upon the timing, duration and specifics of FDA marketing approval of RP-G28, oneThe term of our U.S.in-licensed patents may be eligibleinadequate to protect our competitive position on our products.
Given the amount of time required for a limited Patent Term Extension under the Drug Price Competitiondevelopment, testing and Patent Term Restoration Act of 1984, which is sometimes referred to as the Hatch-Waxman Act, provided our U.S. patent claims a method of treating lactose intolerance that is approved by the FDA. The Hatch-Waxman Act, 35 U.S.C. §156, permits a patent extension of up to five years as compensation for patent term lost during the FDA regulatory review process. The scope of protection afforded by the patent during the extended term is not commensurate with the scope of the unextended portion of the patent; for example, the “rights derived” from a method of use patent during the extended perioddrug candidates, our in-licensed patents protecting such candidates might expire before or shortly after such candidates are “limited to any use claimed by the patent and approved for the product.” 35 U.S.C. §156(b)(2). We may not be grantedcommercialized. In such an extension because of, for example, failing to apply for the extension within applicable deadlines, failing to apply prior to expiration of relevant patents or otherwise failing to satisfy applicable statutory and/or regulatory requirements including, for example, the requirement that the patent to be extended “claim” the approved product or a method of using the approved product. Moreover, the applicable period of extension could be less than we request. Ifevent (and if we are unable to obtain patent term extension or if the term of any such extension is shorterless than we request, the period during which we willrequest), our competitors and other third parties may be able to exclude others from marketing their versions of our product will be shortened and our competitors may obtain approval of genericcompeting products following our patent expiration and our revenue could be reduced, possibly materially. Similar concerns are associated with obtaining Supplemental Protection Certificates of certain patents issued in Europe and owned by Inalco SpA, to which we have an exclusive options of assignment, based upon patent terms lost during European regulatory review processes. In the event that we are unable to obtain any patent term extension, the issued patents for RP-G28 are expected to expire in 2030, assuming they withstand any challenge toothier validity and/or patentability.
If we market products in a manner that violates healthcare fraud and abuse laws, or if we violate government price reporting laws, we may be subject to civil or criminal penalties.
In addition to FDA restrictions on marketing of pharmaceutical products, several other types of state and federal healthcare laws, commonly referred to as “fraud and abuse” laws, have been applied in recent years to restrict certain marketing practices in the pharmaceutical industry. Other jurisdictions such as Europe have similar laws. These laws include false claims and anti-kickback statutes. If we market our products and our products are paid for by governmental programs, it is possible that sometake advantage of our business activities couldinvestment in development and clinical trials by referencing our clinical and preclinical data and launch their product earlier than might otherwise be subject to challenge under one or more of these laws.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government or knowingly making, or causing to be made, a false statement to get a false claim paid. The federal healthcare program anti-kickback statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce, orcase. Generic competition usually results in return for, purchasing, leasing, ordering or arrangingserious price erosion for the purchase, lease or order of any healthcare item or service covered by Medicare, Medicaid or other federally financed healthcare programs. This statute has been interpretedoriginal drug brand.
Risks Related to applyEmployee Matters, Potential Dilution, Stock Price Variability and Other Risks Related to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers or formulary managers on the other. Although there are several statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exemption or safe harbor. Most states also have statutes or regulations similar to the federal anti-kickback law and federal false claims laws, which apply to items and services covered by Medicaid and other state programs, or, in several states, apply regardless of the payor. Administrative, civil and criminal sanctions may be imposed under these federal and state laws.Our Business
Over the past few years, a number of pharmaceutical and other healthcare companies have been prosecuted under these laws for a variety of promotional and marketing activities, such as: providing free trips, free goods, sham consulting fees and grants and other monetary benefits to prescribers; reporting inflated average wholesale prices that were then used by federal programs to set reimbursement rates; engaging in off-label promotion; and submitting inflated best price information to the Medicaid Rebate Program to reduce liability for Medicaid rebates.
Any delay or disruption in the manufacture and supply of RP-G28 may negatively impact our operations.
We do not intend to manufacture RP-G28. We have an agreement with RSM, our contract manufacturer, for the production of Improved GOS, the active pharmaceutical ingredient in RP-G28, and the formulation of sufficient quantities of Improved GOS for the clinical and nonclinical studies that we believe we will need to conduct prior to seeking regulatory approval for RP-G28. However, we do not have agreements for commercial supplies of RP-G28 and we may not be able to reach agreement with RSM or any other contract manufacturer for sufficient supplies to commercialize RP-G28 if it is approved.
Reliance on third-party manufacturers entails risks, to which we would not be subject if we manufactured the products ourselves, including:
Any of these factors could cause the delay of approval or commercialization of RP-G28, or cause us to incur higher costs. Furthermore, if RP-G28 is approved and contract manufacturers fail to deliver the required commercial quantities of finished product on a timely basis and at commercially reasonable prices and we are unable to find one or more replacement manufacturers capable of production at a substantially equivalent cost, in substantially equivalent volumes and quality and on a timely basis, we would likely be unable to meet demand for our product and could lose potential revenue. It may take several years to establish an alternative source of supply for RP-G28 and to have any such new source approved by the government agencies that regulate our products. In the event we do need to identify alternative manufacturing partners, we may have to secure licenses to manufacturing and/or purification technologies, including third-party patent licenses, to allow us to manufacture RP-G28 that is suitable for the late-stage regulatory review process and/or adequate to manufacture commercial quantities of RP-G28.
If the FDA and EMA and other regulatory agencies do not approve the manufacturing facilities of our future contract manufacturers for commercial production, we may not be able to commercialize RP-G28.
The facilities used by any contract manufacturer to manufacture RP-G28 must be the subject of a satisfactory inspection before the FDA or the regulators in other jurisdictions approve the product candidate manufactured at that facility. We are completely dependent on these third-party manufacturers for compliance with the requirements of U.S. and non-U.S. regulators for the manufacture of our finished products. If our manufacturers cannot successfully manufacture material that conform to our specifications and cGMP requirements of any governmental agency whose jurisdiction to which we are subject, our product candidates will not be approved or, if already approved, may be subject to recalls.
Even if RP-G28 receives regulatory approval, we may still face future development and regulatory difficulties.
RP-G28, and any other product candidates we develop in the future, if approved, will be subject to ongoing regulatory requirements for labeling, packaging, storage, advertising, promotion, record-keeping and submission of safety and other post-market information. In addition, approved products, manufacturers and manufacturers’ facilities are required to comply with extensive FDA and EMA requirements and requirements of other similar agencies, including ensuring that quality control and manufacturing procedures conform to cGMPs. Accordingly, we and others with whom we work must continue to expend time, money and effort in all areas of regulatory compliance, including manufacturing, production and quality control. We will also be required to report certain adverse reactions and production problems, if any, to the FDA and EMA and other similar agencies and to comply with certain requirements concerning advertising and promotion for our products. Promotional communications with respect to prescription drugs are subject to a variety of legal and regulatory restrictions and must be consistent with the information in the product’s approved label. Accordingly, we may not promote our approved products, if any, for indications or uses for which they are not approved.
If a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, or disagrees with the promotion, marketing or labeling of a product, it may impose restrictions on that product or us, including requiring withdrawal of the product from the market. If RP-G28 fails to comply with applicable regulatory requirements, a regulatory agency may:
Risks Relating to the Potential Commercialization of RP-G28
Even if approved, RP-G28 may not achieve broad market acceptance among physicians, patients and healthcare payors, and as a result our revenues generated from its sales may be limited.
The commercial success of RP-G28, if approved, will depend upon its acceptance among the medical community, including physicians, health care payors and patients. The degree of market acceptance of RP-G28 will depend on a number of factors, including:
If RP-G28 is approved, but does not achieve an adequate level of acceptance by physicians, patients, the medical community and healthcare payors, sufficient revenue may not be generated from its sales and we may not become or remain profitable. In addition, efforts to educate the medical community and third-party payors on the benefits of RP-G28 may require significant resources and may never be successful.
We have no internal sales, distribution and/or marketing capabilities at this time and we will have to invest significant resources to develop those capabilities or enter into acceptable third-party sales and marketing arrangements.
We have no internal sales, distribution and/or marketing capabilities at this time. To develop these capabilities, we will have to invest significant amounts of financial and management resources, some of which will be committed prior to any confirmation that RP-G28 will be approved. We could also face a number of additional risks, including:
We may have limited or no control over the sales, marketing and distribution activities of third parties. Our future revenues could depend heavilysuccess depends on the success of the efforts of third parties.
We may not be successful in establishing and maintaining development and commercialization collaborations, which could adversely affect our ability to develop RP-G28.retain key employees and to attract, retain and motivate qualified personnel.
Because developing pharmaceutical products, conducting clinical trials, obtaining regulatory approval, establishing manufacturing capabilitiesWe are highly dependent on Michael Poirier, our Chief Executive Officer and marketing approved products are expensive, we may seek to enter into collaborations with companies that have more experience. Additionally, if RP-G28 receives marketing approval, we may enter into salesChairman, and marketing arrangements with third parties with respect toChristopher Lotz, our unlicensed territories. If we are unable to enter into arrangements on acceptable terms, we may be unable to effectively marketVice President and sell RP-G28 in our target markets. We expect to face competition in seeking appropriate collaborators. Moreover, collaboration arrangements are complex and time consuming to negotiate, document and implement and they may require substantial resources to maintain. We may not be successful in our efforts to establish and implement collaborations or other alternative arrangements for the development of RP-G28.
If we collaborate with a third party for the development and commercialization of RP-G28, we can expect to relinquish some or all of the control over the future success of RP-G28. For example, we may relinquish rights to RP-G28 in jurisdictions outside of the United States. Our collaboration partner may not devote sufficient resources to the commercialization of RP-G28 or may otherwise fail in its commercialization. The terms of any collaboration or other arrangement that we establish may not be favorable to us.Chief Financial Officer. In addition, any collaboration that we enter into may be unsuccessful in the development and commercializationrest of RP-G28. In some cases, we may be responsible for continuing preclinical and initial clinical development of RP-G28 or research programs under a collaboration arrangement,our team has been sharply reduced due to rightsizing, voluntary departures and the paymentdisposition of our Qualigen, Inc. diagnostics-products subsidiary – we receive from our collaboration partner may be insufficient to cover the cost of this development. If we are unable to reach agreements with suitable collaborators, we would face increased costs, we may be forced to limit the territories in which we commercialize RP-G28. If we fail to achieve successful collaborations, our operating and financial condition will be materially and adversely affected.currently have only two other employees.
Risks Relating to Our Business and Strategy
We may face competition from other biotechnology and pharmaceutical companies and our operating results will suffer if we failability to compete effectively.
Although we know of no other drug candidates in advanced clinical trials for treating lactose intolerance, the biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. We have potential competitors in the United States, Europe and other jurisdictions, including major multinational pharmaceutical companies, established biotechnology companies, specialty pharmaceutical and generic drug companies and universities and other research institutions. Many of these potential competitors have greater financial and other resources, such as larger research and development staff and more experienced marketing and manufacturing organizations. Large pharmaceutical companies, in particular, have extensive experience in clinical testing, obtaining regulatory approvals, recruiting patients and manufacturing pharmaceutical products. These companies also have significantly greater research, sales and marketing capabilities and collaborative arrangements in our target markets with leading companies and research institutions. Established pharmaceutical companies may also invest heavily to accelerate discovery and development of novel compounds or to in-license novel compounds that could make the product candidates that we develop obsolete. As a result of all of these factors, these potential competitors may succeed in obtaining patent protection and/or FDA approval or discovering, developing and commercializing drugs for the diseases that we are targeting before we do. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. Some of the pharmaceutical and biotechnology companies we expect to compete with include microbiome based development companies: Second Genome, Inc., Seres Health, Inc., Enterome SA, Vedanta Biosciences, Inc., and Rebiotix Inc. In addition, many universities and private and public research institutes may become active in our target disease areas. These potential competitors may succeed in developing, acquiring or licensing on an exclusive basis, technologies and drug products that are more effective or less costly than RP-G28, which could render RP-G28 obsolete and noncompetitive.
We believe thatdepends upon our ability to successfully compete will depend on, among other things:
If our competitors market products that are more effective, safer or less expensive than RP-G28, or that reach the market sooner than RP-G28, we may not achieve commercial success. In addition, the biopharmaceutical industry is characterized by rapid technological change. Because our research approach integrates many technologies, it may be difficult for us to stay abreast of the rapid changes in each technology. If we fail to stay at the forefront of technological change, we may be unable to compete effectively. Technological advances or products developed by our competitors may render our technologies or product candidates obsolete, less competitive or not economical.
We depend on third-party contractors for a substantial portion of our operations and may not be able to control their work as effectively as if we performed these functions ourselves.
We outsource substantial portions of our operations to third-party service providers, including the conduct of preclinical studies and clinical trials, collection and analysis of data, and manufacturing. Our agreements with third-party service providers and CROs are on a study-by-study and project-by-project basis. Typically, we may terminate the agreements with notice and are responsible for the supplier’s previously incurred costs. In addition, any CRO that we retain will be subject to the FDA’s and EMA’s regulatory requirements and similar standards outside of the United States and Europe and we do not have control over compliance with these regulations by these providers. Consequently, if these providers do not adhere to applicable governing practices and standards, the development and commercialization of our product candidates could be delayed or stopped, which could severely harm our business and financial condition.
Because we have relied on third parties, our internal capacity to perform these functions is limited to management oversight. Outsourcing these functions involves the risk that third parties may not perform to our standards, may not produce results in a timely manner or may fail to perform at all. Although we have not experienced any significant difficulties with our third-party contractors, it is possible that we could experience difficulties in the future. In addition, the use of third-party service providers requires us to disclose our proprietary information to these parties, which could increase the risk that this information will be misappropriated. There are a limited number of third-party service providers that specialize or have the expertise required to achieve our business objectives. Identifying, qualifying and managing performance of third-party service providers can be difficult, time consuming and cause delays in our development programs. We currently have a small number of employees, which limits the internal resources we have available to identify and monitor third-party service providers. To the extent we are unable to identify,attract, retain and successfully manage the performance of third-party service providers in the future, our business may be adversely affected,motivate highly skilled and we may be subject to the imposition of civil or criminal penalties if their conduct ofexperienced personnel with scientific, clinical, trials violates applicable law.
A variety of risks associated with our possible international business relationships could materially adversely affect our business.
We may enter into agreements with other third parties for the developmentregulatory, manufacturing and commercialization of RP-G28, or other product candidates we develop in the future, in international markets. International business relationships subject us to additional risks that may materially adversely affect our ability to attain or sustain profitable operations, including:
We will need to expand our operationsmanagement skills and increase the size of our company, and we may experience difficulties in managing growth.
As we advance RP-G28 through clinical trials to commercialization and increase the number of ongoing product development programs, we will need to increase our product development, scientific and administrative headcount to manage these programs. In addition, to meet our obligations as a public company, we will need to increase our general and administrative capabilities. Our management, personnel and systems currently in place may not be adequate to support this future growth. Our need to effectively manage our operations, growth and various projects requires that we:
If we are unable to successfully manage this growth and increased complexity of operations, our business may be adversely affected.
We may not be able to manage our business effectively if we are unable to attract and retain key personnel and consultants.
experience. We may not be able to attract or retain qualified management, finance, scientific and clinical personnel and consultants due toin the intense competition for qualified personnel and consultants among biotechnology, pharmaceuticalfuture. Many of the companies against which we compete have greater financial and other businesses. Ifresources, different risk profiles and a longer history in the industry than we are not abledo. Our competitors may provide higher compensation, more diverse opportunities and/or better opportunities for career advancement. Any or all of these competing factors (as well as our own limited resources) may limit our ability to attract and retain necessaryhigh quality personnel, and consultants to accomplish our business objectives, we may experience constraints that will significantly impede the achievement of our development objectives,which could negatively affect our ability to raise additional capitalsuccessfully develop and commercialize our product candidates and to grow our business and operations as currently contemplated.
We will need to rebuild our development and regulatory teams.
Due to rightsizing, voluntary departures and the disposition of Qualigen, Inc. and our abilityformer FastPack®products business, we currently have only four employees. Although we outsource many drug development functions and may choose to implement our business strategy.
We are highly dependent on the development, regulatory, commercialization and business development expertise of Michael D. Step, our Chief Executive Officer, Andrew J. Ritter, our Founder and President, and Ira E. Ritter, our Executive Chairman and Chief Strategic Officer. If we werecontinue to lose one or more of these key employees, our ability to implement our business strategy successfully could be seriously harmed. Any of our executive officers may terminate their employment at any time. Replacing any of these persons would be difficult and could take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to develop, gain regulatory approval of and commercialize products successfully.
There is also a risk that other obligations could distract our officers and employees from our business, which could have negative impact on our ability to effectuate our business plans.
In addition, we have scientific and clinical advisors and consultants who assist us in formulating our research, development and clinical strategies. Competition to hire and retain consultants from a limited pool is intense. Further, because these advisors are not our employees, they may have commitments to, or consulting or advisory contracts with, other entities that may limit their availability to us, and typically they will not enter into non-compete agreements with us. If a conflict of interest arises between their work for us and their work for another entity, we may lose their services. In addition, our advisors may have arrangements with other companies to assist those companies in developing products or technologies that may compete with ours.
If we fail to maintain an effective system of internal control over financial reportingdo so in the future, we expect that (resources allowing) to recruit and retain more employees in all areas, and particularly in the areas of clinical development, clinical operations, and regulatory affairs (and maybe, longer-term, in areas such as manufacturing, sales, marketing and distribution). We will also need to implement and improve our managerial, operational and financial systems, and obtain stage-appropriate facilities. We do not currently have the cash resources needed for any of the above.
We currently rely, and for the foreseeable future will continue to rely, in substantial part, on certain third-party contract research organizations and consultants to provide certain services, including assuming substantial responsibilities for the conduct of our clinical trials. We cannot assure that the services of such third-party contract research organizations and consultants will continue to be available to us on a timely basis when needed, or that we can find qualified replacements. In addition, if we are unable to effectively manage our outsourced activities or if the quality or accuracy of the services provided by our vendors or consultants is compromised for any reason, our clinical trials may notbe extended, delayed or terminated. We cannot assure that we will be able to accurately reportproperly manage our existing vendors or consultants or find other competent outside vendors and consultants on economically reasonable terms, or at all.
We may engage in strategic transactions that could impact liquidity, increase expenses and present significant distractions to management.
From time to time, we may consider strategic transactions, such as acquisitions of companies, businesses or assets and out-licensing or in-licensing of products, drug candidates or technologies. Potential transactions that we may consider include a variety of different business arrangements, including spin-offs, in-licensing, strategic partnerships, joint ventures, restructurings, divestitures, business combinations and investments. Any such transaction may require us to incur non-recurring or other charges, may increase near term or long-term expenditures and may pose significant integration challenges or disrupt management or business, which could adversely affect our operations and financial results. These transactions may entail numerous operational and financial risks, including:
| ● | exposure to unknown liabilities; | |
| ● | disruption of business and diversion of management’s time and attention in order to develop acquired products, drug candidates or technologies; |
| 18 |
| ● | incurrence of substantial debt or dilutive issuances of equity securities to pay for acquisitions; | |
| ● | higher than expected acquisition and integration costs; | |
| ● | write-downs of assets or impairment charges; | |
| ● | increased amortization expenses; | |
| ● | difficulty and cost in combining the operations, systems and personnel of any acquired businesses with our operations, systems and personnel; | |
| ● | impairment of relationships with key suppliers or customers of any acquired businesses due to changes in management and ownership; and | |
| ● | inability to retain key employees of any acquired businesses. |
Our minority-interest investment in NanoSynex is illiquid and has many risks associated with it.
Our investment in NanoSynex has been reduced to a 39% equity interest and has a number of risks associated with it, including, among others, the following:
| ● | a history of operating losses, with no assurance of future revenues or operating profits; | |
| ● | uncertainty as to the availability to NanoSynex of the cash resources it needs to execute its plans; | |
| ● | technological risk; | |
| ● | our inability, now that we are no longer a majority shareholder of NanoSynex, to control or veto NanoSynex’s decisions; | |
| ● | risks associated with the development of medical devices and NanoSynex’s ability to obtain the necessary regulatory approvals for the development and commercialization of its antimicrobial susceptibility test platform; | |
| ● | very limited manufacturing, marketing, distribution and sales capabilities; | |
| ● | competition from both public and private companies and academic collaborators, many of which have significantly greater experience and financial resources; | |
| ● | acceptance by life sciences research and diagnostic communities is not assured; | |
| ● | commercial development of its antimicrobial susceptibility test platform is not assured; | |
| ● | an inability to manufacture, market or sell its proposed products if it is unsuccessful in entering into strategic alliances or joint ventures with third parties; and | |
| ● | risks related to the political, economic and military conditions in Israel. |
In addition, NanoSynex is a privately-held company and its shares are illiquid, which means that we could not readily obtain cash in exchange for some or all of our equity interest. We no longer hold any NanoSynex debt instruments.
Our reported financial condition may fluctuate significantly from quarter to quarter and year to year, which makes them difficult to predict or understand.
We expect our financial condition and results of operations to fluctuate from quarter to quarter and year to year due to a variety of factors, many of which are beyond our control. Accordingly, you should not blindly rely upon the results of any quarterly or cash flows,annual periods as indications of future financial status or operating performance. Other investors may, however, attach undue significance to reported results which may adversely affect investor confidenceare heavily influenced by such distortions and variability, which in usturn could cause our stock price to rise or fall despite there being no corresponding change in our prospects or position as a practical matter.
We have a substantial amount of derivative securities outstanding.
As of December 31, 2023 there were 398,924 stock options outstanding under our equity incentive plans, 3,081,717 outstanding warrants, and 1,943,729 shares issuable upon voluntary conversion of principal amount of the 2022 Debenture issued to Alpha. (At December 31, 2023, such principal amount was $1,418,922) Due to antidilution adjustments occurring as a result of the value2024 Debenture transaction in February 2024, the outstanding principal balance of the 2022 Debenture (which at March 25, 2024 was $1,088,922) is now convertible upon voluntary conversions into 4,188,162 shares; and in addition the 2024 Debenture’s principal amount is convertible upon voluntary conversions into 900,016 shares, and the warrants issued with the 2024 Debenture are exercisable for 900,016 shares.
| 19 |
The 2022 and 2024 Debentures issued to Alpha are convertible, at any time, and from time to time, at Alpha’s option, into shares of our common stock, subject to the terms and conditions described in the Debentures. Currently the conversion price for such optional conversions is $0.26 per share for the 2022 Debenture and $0.6111 per share for the 2024 Debenture. Furthermore, subject to certain terms and conditions described in the 2022 Debenture, we may elect to pay all or a portion of the Monthly Redemption Amount and/or interest required by the 2022 Debenture in shares of our common stock.
The issuance of shares upon the exercise or conversion of outstanding stock options, warrants and the Debentures (or our election to pay amounts owed under the Debentures in shares of our common stock) could result in significant dilution to the holders of our existing outstanding common stock.
The Sarbanes-Oxley Act requires,We rely significantly upon information technology, and any failure, inadequacy, interruption or security lapse of that technology, including any cyber security incidents, could harm our ability to operate our business effectively and result in a material disruption of our product development programs.
We utilize information technology systems to transmit and store information, including sensitive personal information and proprietary or confidential information, and otherwise to support our business and process. In the future, our systems may prove inadequate to our business needs and necessary upgrades may not operate as designed, which could result in excessive costs or disruptions in portions of our business. In particular, any disruptions, delays or deficiencies from our enterprise resource planning systems could adversely affect our ability to, among other things,matters, process orders, procure supplies, manufacture and ship products, send invoices and track payments, fulfill contractual obligations or otherwise operate our business.
We could also be subject to risks caused by misappropriation, misuse, leakage, falsification or intentional or accidental release or loss of information maintained in the information systems and networks of our company. Outside parties may attempt to penetrate our systems or those of our partners or fraudulently induce our employees or employees of our partners to disclose sensitive information to gain access to our data. Like other companies, we may experience threats to our data and systems, including malicious codes and computer viruses, cyber-attacks or other system failures. Furthermore, a security breach could be facilitated by ineffective protection measures, employee errors or omissions, and malfeasance. Despite our efforts to protect against cyber-attacks and security breaches, hackers and other cyber criminals are using increasingly sophisticated and constantly evolving techniques, and we may need to expend substantial additional resources to continue to protect against potential security breaches or to remediate problems caused by such attacks or any breach of our safeguards. Any system failure, accident or security breach that causes interruptions in our operations, for us or our partners, could result in a material disruption of our product development programs and business operations, in addition to possibly requiring substantial expenditures of resources to remedy. For example, the loss of clinical trial data from completed clinical trials could result in delays in our regulatory approval efforts and we could incur significant increases in costs to recover or reproduce the data. The risk of cyber incidents could also be increased by cyberwarfare in connection with the ongoing war in Ukraine, including potential proliferation of malware from the conflict into systems unrelated to the conflict. To the extent that any disruption or security breach results in a loss of, or damage to, our data or applications, or inappropriate public disclosure of confidential or proprietary information, we may incur liabilities and the further development of our product candidates may be delayed.
We or the third parties upon whom we depend may be adversely affected by natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
We are located in southern California and are subject to risks posed by natural disasters, including wildfires, earthquakes and severe weather that may interfere with our operations. Extreme weather events and other natural disasters could severely disrupt our operations, and have a material adverse effect on our business, results of operations, financial condition and prospects. If a natural disaster, power outage or other event occurred that damaged critical infrastructure, such as the facilities of our third-party clinical sites or contract manufacturers, or that otherwise disrupted operations, it may be difficult or, in certain cases, impossible for us to continue our business for a substantial period of time. Any disaster recovery and business continuity plans we have in place may prove inadequate in the event of a serious disaster or similar event.
Any failure to develop or maintain effective internal controls for financial reporting and disclosure controls and procedures. We are required, under Section 404 of the Sarbanes-Oxley Act, to furnish with our annual report on Form 10-K a report by management on, among other things, the effectiveness of our internal control over financial reporting. This assessment must include disclosure of any material weaknesses identified by our management in our internal control over financial reporting. A material weakness is a control deficiency, or combination of control deficiencies, in internal control over financial reporting that resultsor difficulties encountered in more than a reasonable possibility that a material misstatement of annualimplementing or interim financial statements will not be prevented or detected on a timely basis. Section 404 of the Sarbanes-Oxley Act also generally requires an attestation from our independent registered public accounting firm on the effectiveness of our internal control over financial reporting. However, for as long as we remain an emerging growth company, as defined in the JOBS Act, we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not limited to, not being required to comply with the independent registered public accounting firm’s requirement to attest to the effectiveness ofimproving our internal controls over financial reporting.reporting could harm our operating results and prevent us from meeting our reporting obligations.
Our compliance with Section 404 requires thatEffective internal controls, particularly those related to financial reporting, are necessary for us to produce reliable financial reports. If we incur substantial accounting expensecannot provide reliable financial reports, our business and expend significant management efforts. We may notoperating results could be able to complete our evaluation, testing and any required remediation in a timely fashion. During the evaluation and testing process, if we identify one or more material weaknessesharmed, investors could lose confidence in our internal control overreported financial reporting, we will be unable to assert that our internal control over financial reporting is effective. We cannot assure you that there will not be material weaknesses or significant deficiencies in our internal control over financial reporting ininformation, and the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to accurately report our financial condition, results of operations or cash flows. If we are unable to conclude that our internal control over financial reporting is effective, or if our independent registered public accounting firm determines we have a material weakness or significant deficiency in our internal control over financial reporting once that firm begin its Section 404 reviews, we could lose investor confidence in the accuracy and completeness of our financial reports, the markettrading price of our common stock could decline,drop significantly. In addition, investors relying upon this misinformation could make an uninformed investment decision, and we could be subject to sanctions or investigations by the NASDAQ stock market, the SEC or other regulatory authorities. Failureauthorities or to remedy anystockholder class action securities litigation.
| 20 |
As previously described in our annual report on Form 10-K for the year ended December 31, 2021, in connection with the audit of our financial statements as of and for the year ended December 31, 2021 (the “2021 audit”), our management identified a material weakness in our internal control over financial reporting or to implement or maintain other effective control systems required of public companies, could also restrict our future accessrelated to the capital markets.lack of accounting department resources and/or policies and procedures to ensure recording and disclosure of items in compliance with U.S. GAAP. This material weakness resulted in adjustments to our warrant valuations in connection with the 2021 audit. In response to the material weakness, we took a number of remediation steps to enhance our internal controls, including implementing additional procedures and utilizing external consulting resources with experience and expertise in U.S. GAAP and public company accounting and reporting requirements to assist management with its accounting and reporting of complex and/or non-recurring transactions and related disclosures.
In connection with the audit of our financial statements as of and for the year ended December 31, 2022 (the “2022 audit”), our management determined that the material weakness identified in connection with the 2021 audit had not been fully remediated and resulted in adjustments to the accounting treatment related to convertible debt, the business combination and goodwill impairment during the 2022 audit, which resulted in the late filing of the 2022 Annual Report.
Our disclosure controlsIn connection with the audit of our financial statements as of and procedures may not prevent or detect all errors or actsfor the year ended December 31, 2023, our management identified material weaknesses in our internal control over financial reporting related to limited accounting personnel and resources resulting in lack of fraud.
We are subjectsegregation of duties, and to the periodic reporting requirements of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Our disclosurefact that we have not designed and implemented effective Information Technology General Controls related to access controls and procedures are designed to reasonably assure that information required to be disclosed by us in reports we file or submit under the Exchange Act is accumulated and communicated to management, recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls and procedures or internal controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.financing accounting systems.
These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements or insufficient disclosure due to error or fraud may occur and not be detected.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements and insider trading, which could significantly harm our business.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to comply with the regulations of the FDA and non-U.S. regulators, provide accurate information to the FDA and non-U.S. regulators, comply with health care fraud and abuse laws and regulations in the United States and abroad, report financial information or data accurately or disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the health care industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. We have adopted an employee handbook, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines or other sanctions.
We face potential product liability exposure, and if successful claims are brought against us, we may incur substantial liability for a product candidate and may have to limit its commercialization.
The use of our product candidates in clinical trials and the sale of any products for which we may obtain marketing approval expose us to the risk of product liability claims. Product liability claims may be brought against us or our potential future collaborators by participants enrolled in our clinical trials, patients, health care providers or others using, administering or selling our products. If we cannot successfully defend ourselves against any such claims, we would incur substantial liabilities. Regardless of merit or eventual outcome, product liability claims may result in:
We have product liability insurance coverage for our clinical trials in the United States and in selected other jurisdictions where we intend to conduct clinical trials at levels we believe are sufficient and consistent with industry standards for companies at our stage of development. However, our insurance coverage may not reimburse us or may not be sufficient to reimburse us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive, and, in the future, we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to product liability. We intend to expand our insurance coverage for products to include the sale of any commercial products for which we obtain marketing approval, but we may be unable to obtain commercially reasonable product liability insurance for any products approved for marketing. Large judgments have been awarded in class action lawsuits based on drugs that had unanticipated side effects. A successful product liability claim or series of claims brought against us, particularly if judgments exceed our insurance coverage, could decrease our cash resources and adversely affect our business.
Our insurance policies are expensive and only protect us from some business risks, which will leave us exposed to significant uninsured liabilities.
We do not carry insurance for all categories of risk that our business may encounter. Some of the policies we currently maintain include general liability ($2.0 million coverage), employment practices liability, workers’ compensation, and directors’ and officers’ insurance at levels we believe are typical for a company in our industry and at our stage of development. We currently carry clinical trial liability insurance for our clinical trials at levels we believe are sufficient and consistent with industry standards for companies at our stage of development. We do not know, however, if we will be able to maintain insurance with adequate levels of coverage. Any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our financial position and results of operations.
If we engage in an acquisition, reorganization or business combination, we will incur a variety of risks that could adversely affect our business operations or our stockholders.
From time to time we have considered, and we will continue to considertake steps to enhance our internal controls, including implementing additional internal procedures and utilizing well-established external consulting resources with experience and expertise in the future, strategic business initiatives intended to further the expansionU.S. GAAP and development of our business. These initiatives may include acquiring businesses, technologies or products or entering into a business combination with another company. If we pursue such a strategy, we could, among other things:public company accounting and reporting requirements.
Although we intend to evaluate and consider acquisitions, reorganizations and business combinations in the future, we have no agreements or understandings with respect to any acquisition, reorganization or business combination at this time.
Risks Relating to Our Intellectual Property
It is difficult and costly to protect our proprietary rights, and we may not be able to ensure their protection. If our patent position does not adequately protect our product candidates, others could compete against us more directly, which would harm our business, possibly materially.
Our commercial success will depend in part on obtaining, maintaining and enforcing patent protection and on developing, preserving and enforcing current trade secret protection. In particular, it will depend in part on our ability to obtain, maintain and enforce patents, especially those directed to methods of using our current product, RP-G28, and other future drug candidates, and those directed to the methods used to develop and manufacture our products, as well as successfully defending these patents against third-party challenges. Our ability to stop third parties from making, using, selling, offering to sell or importing our products depends on the extent to which we have rights under valid and enforceable patents (and/or trade secrets) that cover these activities. We cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents that may be granted to us in the future will withstand subsequent challenges to their validity, enforceability, and/or patentability, or if they will be commercially useful in protecting our product candidates, discovery programs and processes. Furthermore, we cannot be sure that our existing patents and patent applications will embrace (or “claim”) the particular uses for RP-G28 that will be approved by the FDA.
The patent positions of biotechnology and pharmaceutical companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved.
No consistent policy regarding the patentability and/or validity of patent claims related to pharmaceutical patents has emerged, to date, in the United States or in most jurisdictions outside of the United States. Changes in either the patent laws (be they substantive or procedural) or in the interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of any claims that will issue or will be enforceable in the patents that have or may be issued from the patents and applications we currently own or may in the future own or license from third parties. Further, if any patents we obtain, or to which we obtain licenses, are deemed invalid, unpatentable and unenforceable, our ability to commercialize or license our technology could be adversely affected.
In the future others may file patent applications directed to products, uses for products, and manufacturing techniques and related technologies that are similar, identical or competitive to ours or important to our business. We cannot be certain that any patent or patent application owned by a third party will not have priority over patent applications filed or in-licensed by us in the future, or that we or our licensors will not be involved in interference, opposition, inter partes review or invalidity proceedings before U.S. or non-U.S. patent offices or courts.
The degree of future protection for our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
Patents directed to pharmaceutical compositions containing RP-G28 or methods of using RP-G28 expire in 2030 if the appropriate maintenance fee renewal, annuity, or other government fees are paid, unless a patent term extension based on regulatory delay is obtained. We expect that expiration in 2030 of some of our pharmaceutical composition and method-of-use patents directed to RP-G28 and its use in treating lactose intolerance will have a limited impact on our ability to protect our intellectual property in the United States, where we have additional issued patents directed to such compositions and uses that extend until 2030. In other countries, our issued patents and pending patent applications directed to compositions containing or methods of using RP-G28 for treating other indications, if issued, would expire in 2030. We will attempt to mitigate the effect of patent expiration by seeking data exclusivity, or the foreign equivalent thereof, in conjunction with product approval, as well as by filing additional patent applications covering improvements in our intellectual property.
We expect that the other patent applications for the RP-G28 portfolio, if issued, and if the appropriate maintenance, renewal, annuity or other governmental fees are paid, would expire in 2030. We own pending applications in the United States, Europe, and certain other countries directed to uses of RP-G28 to treat a variety of disorders, including lactose intolerance. Patent protection, to the extent these patents issue, would be expected to extend to 2030, unless a patent term extension based on regulatory delay is obtained.
Due to the patent laws of a country, or the decisions of a patent examiner in a country, or our own filing strategies, we may not obtain patents directed to all of our product candidates or methods involving these candidates in the parent patent application. We plan to pursue divisional patent applications or continuation patent applications in the United States and other countries to obtain claims directed to inventions that were disclosed but not claimed in the parent patent application.
We also may rely on trade secrets to protect our technology, especially where we do not believe patent protection is appropriate or feasible. However, trade secrets are difficult to protect. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, outside scientific collaborators and other advisors may unintentionally or willfully disclose our information to competitors. Enforcing a claim that a third party illegally obtained and is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States are sometimes less willing to protect trade secrets. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
Our patents are not directed to RP-G28 as a composition of matter.
Although we own certain patents and patent applications with claims directed to specific pharmaceutical compositions and methods of using RP-G28 to treat lactose intolerance, we do not have patents directed to RP-G28 as a composition of matter in the United States or elsewhere. As a result, we may be limited in our ability to list our patents in the FDA’s Orange Book if our product or the use of our product, consistent with its FDA-approved label, would not fall within the scope of our patent claims. Also, our competitors may be able to offer and sell products so long as these competitors do not infringe any other patents that we (or third parties) hold, including patents with claims directed to the manufacture of RP-G28, pharmaceutical compositions containing RP-G28, and/or method of using RP-G28. In general, pharmaceutical composition patents and method of use patents are more difficult to enforce than composition of matter patents because, for example, of the risks that FDA may approve alternative uses of the subject compounds not covered by the method of use patents, and others may engage in off-label sale or use of the subject compounds. Physicians are permitted to prescribe an approved product for uses that are not described in the product’s labeling. Although off-label prescriptions may infringe our method of use patents, the practice is common across medical specialties and such infringement is difficult to prevent or prosecute. FDA approval of uses that are not covered by our patents would limit our ability to generate revenue from the sale of RP-G28, if approved for commercial sale. Off-label sales would limit our ability to generate revenue from the sale of RP-G28, if approved for commercial sale.
We may incur substantial costs as a result of litigation or other proceedings relating to patent and other intellectual property rights.
If we choose to go to court to stop another party from using the inventions claimed in any patents we obtain, that individual or company may seek a post grant review (including inter partes review) of our patents, and has the right to ask the court to rule that such patents are invalid or should not be enforced against that third party. These lawsuits and administrative proceedings are expensive and would consume time and resources and divert the attention of managerial and scientific personnel even if we were successful in stopping the infringement of such patents. In addition, there is a risk that the court or administrative body will decide that such patents are not valid or unpatentable and that we do not have the right to stop the other party from using the inventions. There is also the risk that, even if the validity/patentability of such patents is upheld, the court will refuse to stop the other party on the ground that such other party’s activities do not infringe our patents. In addition, the U.S. Supreme Court and the Court of Appeals for the Federal Circuit have articulated and/or modified certain tests used by the U.S. Patent and Trademark Office (the “USPTO”), in assessing patentability and by the courts in assessing validity and claim scope, which may decrease the likelihood that we will be able to obtain patents and increase the likelihood that others may succeed in challenging any patents we obtain or license.
We may infringe the intellectual property rights of others, which may prevent or delay our product development efforts and stop us from commercializing or increase the costs of commercializing our product candidates.
Our success will depend in part on our ability to operate without infringing the proprietary rights of third parties. We cannot guarantee that our products, our methods of manufacture, or our uses of RP-G28 (or our other product candidates), will not infringe third-party patents. Furthermore, a third party may claim that we or our manufacturing or commercialization collaborators are using inventions covered by the third party’s patent rights and may go to court to stop us from engaging in our normal operations and activities, including making or selling our product candidates. These lawsuits are costly and could affect our results of operations and divert the attention of managerial and scientific personnel. There is a risk that a court would decide that we or our commercialization collaborators are infringing the third party’s patents and would order us or our collaborators to stop the activities covered by the patents. In that event, we or our commercialization collaborators may not have a viable way around the patent and may need to halt commercialization of the relevant product. In addition, there is a risk that a court will order us or our collaborators to pay the other party damages for having violated the other party’s patents. In the future, we may agree to indemnify our commercial collaborators against certain intellectual property infringement claims brought by third parties. The pharmaceutical and biotechnology industries have produced a proliferation of patents, and it is not always clear to industry participants, including us, which patents cover various types of products or methods of use. The scope of coverage of a patent is subject to interpretation by the courts, and the interpretation is not always uniform. If we are sued for patent infringement, the patentee would need to demonstrate, by a preponderance of the evidence that our products or methods infringe the patent claims of the relevant patent, and we would need to demonstrate either that we do not infringe or, by clear and convincing evidence, that the patent claims are invalid; we may not be able to do this. Proving invalidity is difficult. For example, in the United States, proving invalidity requires a showing of clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents. Even if we are successful in these proceedings, we may incur substantial costs and divert management’s time and attention in pursuing these proceedings, which could have a material adverse effect on us. If we are unable to avoid infringingremediate the patent rights of others, we maymaterial weaknesses and achieve and maintain effective internal control over financial reporting and effective disclosure controls, our business could be requiredadversely affected.
Our right to seek a license, which may not be available, defend an infringement action or challenge the validity or enforceability of the patents in court. Patent litigationuse our “shelf” Form S-3 registration statement is costly and time consuming. We may not have sufficient resources to bring these actions to a successful conclusion. In addition, if we do not obtain a license, develop or obtain non-infringing technology, otherwise fail to defend an infringement action successfully or have a court hold that any patent we infringe is invalid or unenforceable, we may incur substantial monetary damages, encounter significant delays in bringing our product candidates to market and we may be precluded from manufacturing or selling our product candidates.sharply limited.
We cannot be certain that others have not filed patent applications for technology claimed in our pending applications, or that we were the first to invent the technology, because:
Our competitors may have filed, and may in the future file, patent applications directed to technology similar to ours. Any such patent application may have priority over our patent applications, which could further require us to obtain rights to issued patents directed to such technologies. If another party has filed a U.S. patent application on inventions similar to ours, we may have to participate in an interference proceeding declared by the USPTO to determine priority of invention in the United States. The costs of these proceedings could be substantial, and it is possible that such efforts would be unsuccessful if, unbeknownst to us, the other party had independently arrived at the same or similar invention prior to our own invention, resulting in a loss of our U.S. patent position with respect to such inventions. Other countries have similar laws that permit secrecy of patent applications, and other parties may be entitled to priority over our applications in such jurisdictions.
Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. In addition, any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise the funds necessary to continue our operations.
Obtaining and maintaining our patent portfolio depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patents could be deemed abandoned or eliminated for non-compliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and/or applications will be due to be paid to the USPTO and various governmental patent agencies outside of the United States in several stages over the lifetime of the patents and/or applications. We employ an outside firm to pay fees due to non-U.S. patent agencies and this outside firm has systems in place to ensure compliance on payment of fees. The USPTO and various non-U.S. governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. We employ reputable law firms and other professionals to help us comply, and in many cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordanceForm S-3 “shelf” registration statement with the applicable rules. However, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, our competitors might be able to enter the market and this circumstance would have a material adverse effect on our business.
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers. If we are not able to adequately prevent disclosure of trade secrets and other proprietary information, the value of our technology and products could be significantly diminished.
As is common in the biotechnology and pharmaceutical industries, we employ individuals who were previously employed at other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may be subject to claims that these employees, or we, have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
We rely on trade secrets to protect our proprietary technologies, especially where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. We rely in part on confidentiality agreements with our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to protect our trade secrets and other proprietary information. These agreements may not effectively prevent disclosure of confidential information and may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. In addition, others may independently discover our trade secrets and proprietary information. For example, the FDA, as part of its Transparency Initiative, is currently considering whether to make additional information publicly available on a routine basis, including information that we may consider to be trade secrets or other proprietary information, and it is not clear at the present time how the FDA’s disclosure policies may change in the future, if at all. Costly and time-consuming litigation could be necessary to enforce and determine the scope of our proprietary rights, and failure to obtain or maintain trade secret protection could adversely affect our competitive business position.
Failure to secure trademark registrations could adversely affect our business.
We have not developed a trademarkSEC for our RP-G28 product. Hence, we do not currently own any actual or potential trademark rights associated with our RP-G28 product. If we seek to register additional trademarks, including trademarks associated with our RP-G28 product, our trademark applications may not be allowed for registration or our registered trademarks may not be maintained or enforced. During trademark registration proceedings, we may receive rejections. Although we are given an opportunity to respond to those rejections, we may be unable to overcome such rejections. In addition, in the USPTO and in comparable agencies in many other jurisdictions, third parties are given an opportunity to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition or cancellation proceedings may be filed against our trademarks, and our trademarks may not survive such proceedings. If we do not secure registrations for our trademarks, we may encounter more difficulty in enforcing them against third parties than we otherwise would.
Risks Relating to Our Capital Stock
An active trading market for our common stock may not develop or be sustained.
Prior to our initial public offering, there was no public market for our common stock. Since our initial public offering in June 2015, there has been, and we expect that there will continue to be, only a limited volume of trading in our common stock. An active trading market in our common stock may not develop or, if developed, may not be sustained. The lack of an active market may impair your ability to sell your shares at the time you wish to sell them or at a price that you consider reasonable. The lack of an active market may also reduce the fair market value of your shares. An inactive market may also impair our ability to raise capital to continue to fund operations by selling shares and may impair our ability to acquire other companies or technologies by using our shares as consideration.
Our share price may be volatile, which could subject us to securities class action litigation and prevent you from being able to sell your shares at or above your purchase price.
The market price of shares of our common stock could be subject to wide fluctuations in response to many risk factors listed in this section, and others beyond our control, including:
Furthermore, the stock markets have experienced extreme price and volume fluctuations that have affected and continue to affect the market prices of equity securities of many companies. These fluctuations often have been unrelated or disproportionate to the operating performance of those companies. These broad market and industry fluctuations, as well as general economic, political and market conditions such as recessions, interest rate changes or international currency fluctuations, may negatively impact the market price of shares of our common stock. In addition, such fluctuations could subject us to securities class action litigation, which could result in substantial costs and divert our management’s attention from other business concerns, which could seriously harm our business.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our share price and trading volume could decline.
The trading market for our common stock will depend on the research and reports that securities or industry analysts publish about us or our business. We do not have any control over these analysts. There can be no assurance that analysts will cover us or provide favorable coverage. If one or more of the analysts who cover us downgrade our stock or change their opinion of our stock, our share price would likely decline. If one or more of these analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in the financial markets, which could cause our share price or trading volume to decline.
There is no active, public market for our Series A Preferred stock.
There is no established public trading market for our Series A Preferred stock. We do not intend to apply to list the Series A Preferred stock on a securities exchange. Without an active trading market, the liquidity of the Series A Preferred stock will be limited.
Holders of Series A Preferred have limited voting rights.
Except with respect to certain material changes in the terms of the Series A Preferred stock and certain other matters and except as may be required by Delaware law, holders of Series A Preferred stock have no voting rights.
Future sales of our common stock, or the perception that future sales may occur, may cause the market price of our common stock to decline, even if our business is doing well.
Sales by our stockholders of a substantial number of shares of our common stock in the public market could occur in the future. These sales, or the perception in the market that the holders of a large number of shares of common stock intend to sell shares, could reduce the market price of our common stock.
We may sell up to $6.5 million of our shares of common stock to Aspire Capital pursuant to financing arrangement with Aspire Capital. The sale of a substantial number of shares of our common stock by Aspire Capital, or anticipation of such sales, could cause the trading price of our common stock to decline or make it more difficult for us to sell equity or equity-related securities in the future at a time and at a price that we might otherwise desire. However, we have the right to control the timing and amount of sales of our shares to Aspire Capital, and we may terminate the financing arrangement at any time at our discretion without any penalty or cost to us.
Exercise of options or warrants or conversion of convertible securities may have a dilutive effect on your percentage ownership and may result in a dilution of your voting power and an increase in the number of shares of common stock eligible for future resale in the public market, which may negatively impact the trading price of our shares of common stock.
The exercise or conversion of some or all of our outstanding options, warrants, or convertible securities could result in significant dilution in the percentage ownership interest of investors in this offering and in the percentage ownership interest of our existing common stockholders and in a significant dilution of voting rights and earnings per share.
Additionally, the issuance of sharesup to $150,000,000 of securities, and the SEC declared the registration statement effective on August 5, 2022. However, due to the “baby shelf” rules adopted by the SEC, the maximum amount of securities we can sell under this registration is now limited to one-third of our common stock upon exercise of stock options outstandingpublic float. Because our public float is very modest (e.g., $2.9 million at December 31, 2023), the maximum amount we could sell using this registration statement was under our stock incentive plans will further dilute our stockholders’ voting interests. To$1.0 million at that time. Therefore, the extent options and/or warrants and/or conversion rights are exercised, additional shares of common stock will be issued, and such issuance will dilute stockholders.
Our executive officers, directors and principal stockholders maintain the ability to exert substantial influence over all matters submitted to stockholders for approval.
Our executive officers, directors and principal stockholders beneficially own shares representing approximately 28.5% of our outstanding capital stock. As a result, if these stockholders were to choose to act together, they would be able to exert substantial influence over all matters submitted to our stockholders for approval, as well as our management and affairs. For example, these persons, if they choose to act together, would exert substantial influence over the election of directors and approval of any merger, consolidation or sale of all or substantially all of our assets. This concentration of voting power could delay or prevent an acquisition of our Company on terms that other stockholders may desire.
We are an “emerging growth company” and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, which could make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act and we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies” including not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
We may take advantage of these reporting exemptions until we areregistration statement no longer constitutes an “emerging growth company.” We will remain an emerging growth company untilimportant tool for accessing the earlier of (i) the last day of the fiscal year (a) following the fifth anniversary of the date we completedpublic markets to satisfy our initial public offering, which was June 29, 2015, (b) in which we have total annual gross revenue of at least $1.07 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeded $700.0 million as of the prior June 30th, or (ii) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.needs for capital.
Our failure to meet the continued listing requirements of NASDAQNasdaq could result in a de-listingdelisting of our common stock.
If we fail to satisfy the continued listing requirements of NASDAQ, such as the corporate governance requirements or the minimum closing bid price requirement, NASDAQNasdaq, Nasdaq may take steps to de-listdelist our common stock. Such a de-listingdelisting would likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so.
On April 20, 2023, we received a notification letter from the Listing Qualifications Department of Nasdaq indicating that, as a result of our delay in filing the 2022 Annual Report, we were not in compliance with the timely filing requirements for continued listing under Nasdaq Listing Rule 5250(c)(1). The notification letter had no immediate effect on the listing or trading of our common stock on the Nasdaq Capital Market. On May 2, 2023, the Company filed the Form 10-K with the SEC and was subsequently notified by Nasdaq on May 4, 2023 that it had regained compliance with Nasdaq’s listing rule 5250(c)(1) as a result thereof and that the matter was closed.
On November 20, 2023, we received a letter (the “Bid Price Deficiency Notice”) from The Nasdaq Stock Market (“Nasdaq”) notifying the Company that, because the closing bid price for its common stock has been below $1.00 per share for 30 consecutive business days, it no longer complies with the minimum bid price requirement for continued listing on The Nasdaq Capital Market. Nasdaq Listing Rule 5550(a)(2) requires listed securities to maintain a minimum bid price of $1.00 per share (the “Minimum Bid Price Requirement”), and Listing Rule 5810(c)(3)(A) provides that a failure to meet the Minimum Bid Price Requirement exists if the deficiency continues for a period of 30 consecutive business days.
The Bid Price Deficiency Notice has no immediate effect on the listing of the Company’s common stock on The Nasdaq Capital Market. Pursuant to Nasdaq Marketplace Rule 5810(c)(3)(A), the Company has been provided an initial compliance period of 180 calendar days, or until May 20, 2024 to regain compliance with the Minimum Bid Price Requirement. During the compliance period, the Company’s shares of common stock will continue to be listed and traded on The Nasdaq Capital Market. To regain compliance, the closing bid price of the Company’s common stock must meet or exceed $1.00 per share for a minimum of 10 consecutive business days during the 180 calendar day grace period.
| 21 |
In the event the Company is not in compliance with the Minimum Bid Price Requirement by May 20, 2024, the Company may be afforded a second 180 calendar day grace period.
The Company intends to actively monitor the bid price for its common stock between now and May 20, 2024 and will consider available options to regain compliance with the Minimum Bid Price Requirement.
On November 21, 2023, the Company also received a letter (the “Equity Deficiency Letter”) from Nasdaq notifying the Company that, based on the Company’s stockholders’ deficit of ($1,640,552) as of September 30, 2023, as reported in the Company’s Quarterly Report on Form 10-Q for the quarterly period ended September 30, 2023, it is no longer in compliance with the minimum stockholders’ equity requirement for continued listing on The Nasdaq Capital Market under Nasdaq Listing Rule 5550(b)(1), which requires listed companies to maintain stockholders’ equity of at least $2.5 million (the “Minimum Stockholders’ Equity Requirement”), or the alternative criteria of $35 million market value of listed securities or $500,000 in net income from continuing operations in the most recent fiscal year or two or the last three fiscal years—which alternatives, as noted in the Equity Deficiency Letter, the Company does not meet. The Company was given until January 5, 2024 to provide Nasdaq with a specific plan (the “Compliance Plan”) to achieve and sustain compliance with the Minimum Stockholders’ Equity Requirement or its alternatives. If the Company’s Compliance Plan is accepted, Nasdaq may grant an extension of up to 180 calendar days from the date of the Equity Deficiency Letter for the Company to evidence compliance.
The Company submitted a Compliance Plan to Nasdaq on January 5, 2024 to regain compliance with the Nasdaq Listing Rules. The Compliance Plan was accepted by Nasdaq and the Company was granted an extension of up to 180 calendar days from the date of the Equity Deficiency Letter (i.e., until May 20, 2024) for the Company to evidence compliance. If the Company does not regain compliance within the requisite time period, or if the Company fails to satisfy another Nasdaq requirement for continued listing, Nasdaq could provide notice that the Company’s securities will become subject to delisting, which delisting determination the Company has the right to appeal.
If we are unable to maintain compliance with Nasdaq’s continued listing requirements, and in the event of a de-listing,delisting, we would take actionsaction to restore our compliance with NASDAQ’sNasdaq’s listing requirements, but we can provide no assurance that any such action taken by us would allow our common stock to become listed again, stabilize the market price or improve the liquidity of our common stock, prevent our common stock from dropping below the NASDAQNasdaq minimum bid price requirement or prevent future non-compliance with NASDAQ’sNasdaq’s other listing requirements.
On June 7, 2017, we received a notice from NASDAQ that, because the closing bid price ofLosing our common stock has been below $1.00 per share for 30 consecutive business days, it no longer complies with the minimum bid price requirement for continuedNasdaq other listing on The NASDAQ Capital Market. NASDAQ Listing Rule 550(a)(2) requires listed securities to maintain a minimum bid price of $1.00 per share, and Listing Rule 5810(c)(3)(A) provides that a failure to meet the minimum bid price requirement exists if the deficiency continues for a period of 30 consecutive business days.
On November 2, 2017,would seriously harm us, by undermining our board of directors approved a reverse stock split of our outstanding shares of common stock at a ratio within a range of 1-for-8 to 1-for-15, to be determined by the board of directors at a later date, and subject to stockholder approval. At a special meeting of stockholders held on December 20, 2017, our stockholders approved the reverse stock split within a range of 1-for-8 to 1-for-15. On March 1, 2018, our board of directors approved a 1-for-10 reverse stock split, with an anticipated effective date of on or before March 23, 2018.
We have until June 4, 2018 to regain compliance with the minimum bid price requirement. During the compliance period, our shares of common stock will continue to be listed and traded on The Nasdaq Capital Market. To regain compliance, the closing bid price of our common stock must meet or exceed $1.00 per share for a minimum of 10 consecutive business days during the 180 calendar day grace period.
Provisions in our corporate charter documents and under Delaware law could make an acquisition of our company, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our amended and restated certificate of incorporation and our amended and restated bylaws may discourage, delay or prevent a merger, acquisition or other change in control of our company that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock, thereby depressing the market price of our common stock. In addition, because our board of directors is responsible for appointing the members of our management team, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors. Among other things, these provisions provide that:
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law (the “DGCL”), which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
Claims for indemnification by our directors and officers may reduce our available funds to satisfy successful stockholder claims against us and may reduce the amount of money available to us.
As permitted by Section 102(b)(7) of the DGCL, our amended and restated certificate of incorporation limits the liability of our directors to the fullest extent permitted by law. In addition, as permitted by Section 145 of the DGCL, our amended and restated certificate of incorporation and our amended and restated bylaws provide that we shall indemnify, to the fullest extent authorized by the DGCL, each person who is involved in any litigation or other proceeding because such person is or was a director or officer of our company or is or was serving as an officer or director of another entity at our request, against all expense, loss or liability reasonably incurred or suffered in connection therewith. Our amended and restated certificate of incorporation provides that the right to indemnification includes the right to be paid expenses incurred in defending any proceeding in advance of its final disposition, provided, however, that such advance payment will only be made upon delivery to us of an undertaking, by or on behalf of the director or officer, to repay all amounts so advanced if it is ultimately determined that such director is not entitled to indemnification. If we do not pay a proper claim for indemnification in full within 60 days after we receive a written claim for such indemnification, except in the case of a claim for an advancement of expenses, in which case such period is 20 days, our restated certificate of incorporation and our restated bylaws authorize the claimant to bring an action against us and prescribe what constitutes a defense to such action.
Section 145 of the DGCL permits a corporation to indemnify any director or officer of the corporation against expenses (including attorney’s fees), judgments, fines and amounts paid in settlement actually and reasonably incurred in connection with any action, suit or proceeding brought by reason of the fact that such person is or was a director or officer of the corporation, if such person acted in good faith and in a manner that he reasonably believed to be in, or not opposed to, the best interests of the corporation, and, with respect to any criminal action or proceeding, if he or she had no reason to believe his or her conduct was unlawful. In a derivative action, (i.e., one brought by or on behalf of the corporation), indemnification may be provided only for expenses actually and reasonably incurred by any director or officer in connection with the defense or settlement of such an action or suit if such person acted in good faith and in a manner that he or she reasonably believed to be in, or not opposed to, the best interests of the corporation, except that no indemnification shall be provided if such person shall have been adjudged to be liable to the corporation, unless and only to the extent that the court in which the action or suit was brought shall determine that the defendant is fairly and reasonably entitled to indemnity for such expenses despite such adjudication of liability.
The rights conferred in the restated certificate of incorporation and the restated bylaws are not exclusive, and we are authorized to enter into indemnification agreements with our directors, officers, employees and agents and to obtain insurance to indemnify such persons. We have entered into indemnification agreements with each of our officers and directors.
The above limitations on liability and our indemnification obligations limit the personal liability of our directors and officers for monetary damages for breach of their fiduciary duty as directors by shifting the burden of such losses and expenses to us. Although we plan to increase the coverage under our directors’ and officers’ liability insurance, certain liabilities or expenses covered by our indemnification obligations may not be covered by such insurance or the coverage limitation amounts may be exceeded. As a result, we may need to use a significant amount of our funds to satisfy our indemnification obligations, which could severely harm our business and financial condition and limit the funds available to stockholders who may choose to bring a claim against our company.
We have never paid dividends on our common stock and do not anticipate paying dividends for the foreseeable future, and accordingly, stockholders must rely on stock appreciation for any return on their investment.
We have never paid dividends on our common stock and we do not anticipate paying dividends on our common stock for the foreseeable future. Accordingly, any return on an investment in our common stock will be realized, if at all, only when stockholders sell their shares. In addition, our failure to pay dividends may make our stock less attractive to investors, adversely impacting trading volume and price.
Our ability to useraise capital and decreasing our net operating loss carryforwards and certain other tax attributes may be limited.attractiveness to possible merger partners.
As of December 31, 2017, we had federal net operating loss carryforwards (“NOLs”) of approximately $39.0 million which begin to expire in 2028. Our ability to utilize our NOLs may be limited under Section 382 and 383 of the Internal Revenue Code. The limitations apply if an ownership change, as defined by Section 382, occurs. Generally, an ownership change occurs when certain shareholders increase their aggregate ownership by more than 50 percentage points over their lowest ownership percentage in a testing period (typically three years). Although we have not undergone a Section 382 analysis, it is possible that the utilization of the NOLs, could be substantially limited. Additionally, U.S. tax laws limit the time during which these carryforwards may be utilized against future taxes. As a result, we may not be able to take full advantage of these carryforwards for federal and state tax purposes. Future changes in stock ownership may also trigger an ownership change and, consequently, a Section 382 limitation.
Item 1B. Unresolved Staff CommentsComments.
None.Not applicable.
Our processes for assessing, identifying, and managing material risks from cybersecurity threats are not advanced. None of our personnel are trained specialists in cybersecurity; we rely on off-the-shelf” security software. We have not conducted employee training regarding cybersecurity or conducted internal tests or simulations. Nonetheless, we believe that, due to the nature and status of our business, our cybersecurity position is currently satisfactory for our situation. We do not believe that cybersecurity risks have materially affected (or are reasonably likely to materially affect) us.
We have only four employees and so disclosure of cybersecurity events to management would occur organically, or would in the first instance be observed by management.
For information of ours which resides on our CROs’ computer systems, we rely on the CROs’ own cybersecurity processes and systems.
Item 2. PropertiesProperties.
On July 9, 2015, we entered into a newNone. The lease with Century Park, pursuant to which we are leasing approximately 2,780 square feet of office space in Los Angeles, California for our headquarters. The lease providesprevious operating facilities at 2042 Corte Del Nogal, Carlsbad, California had been in the name of our subsidiary Qualigen, Inc. We sold Qualigen, Inc. in 2023 and the Company has no responsibility for a term of sixty-one (61) months, which commenced October 1, 2015. We pay $9,733 per month in base rent with normal escalations until November 2018, after which our monthly payment will increase ratably per year pursuant to the terms of the lease agreement. We have the optiongoing forward. The Company had also utilized such facility (in addition to extend the term of the lease for one five-year term, provided that the rent would be subject to market adjustment at the beginning of the renewal term. We believe that our facilityQualigen, Inc.’s use), but now has removed from it. The Company is suitable and adequate for our current needs.currently essentially “virtual.”
Item 3. Legal ProceedingsProceedings.
From time to time, we have been and may again become involved in legal proceedings arising in the ordinary course of our business. We are not presently a party to any material litigation.None.
| 22 |
Item 4. Mine Safety DisclosuresDisclosures.
Not Applicable.applicable.
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity SecuritiesSecurities.
Our shares of common stock havehas been listed and traded on The NASDAQthe Nasdaq Capital Market under the symbol “RTTR”“QLGN” since June 24, 2015. Prior to that date, there was no public market for our common stock.May 26, 2020.
The following table sets forth, for the periods indicated, the high and low intra-day sale prices in dollars on The NASDAQ Capital Market for our common stock.Holders
| 2017 | ||||||||
| High | Low | |||||||
| First Quarter | $ | 3.75 | $ | 1.29 | ||||
| Second Quarter | $ | 1.46 | $ | 0.52 | ||||
| Third Quarter | $ | 0.68 | $ | 0.34 | ||||
| Fourth Quarter | $ | 0.42 | $ | 0.28 | ||||
| 2016 | ||||||||
| High | Low | |||||||
| First Quarter | $ | 1.79 | $ | 0.98 | ||||
| Second Quarter | $ | 1.89 | $ | 1.10 | ||||
| Third Quarter | $ | 2.47 | $ | 1.20 | ||||
| Fourth Quarter | $ | 3.26 | $ | 1.62 | ||||
Holders
As of March16,2018 25, 2024, there were approximately 36287 registered holders of record of our common stock. These figures doThis figure does not reflect the beneficial ownership orof shares held in nominee name.
Dividend Policy
We have never paid or declared any cash dividends on our common stock, and we do not anticipate paying any cash dividends on our common stock in the foreseeable future. We intend to retain all available funds and any future earnings to fund the development and expansion of our business. Any future determination to pay dividends will be at the discretion of our board of directors and will depend upon a number of factors, including our results of operations, financial condition, future prospects, contractual restrictions, restrictions imposed by applicable law and other factors our board of directors deems relevant. See “Risk Factors — Risks Relating to Our Common Stock— We have never paid dividends on our common stock and do not anticipate paying dividends for the foreseeable future, and accordingly, stockholders must rely on stock appreciation for any return on their investment.”
Securities Authorized for Issuance Under Equity Compensation Plans
See Part III, Item 12.12 “Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters” for information relating to our equity compensation plans.
Recent Sales of Unregistered Securities
On May 4, 2017, we entered into a common stock purchase agreement with Aspire Capital (the “2017 Aspire Purchase Agreement”), which provides that upon the terms and conditions set forth therein, Aspire CapitalNo further disclosure is committedrequired in response to purchase up to an aggregate of $6.5 million of shares of our common stock over the 30-month term of the 2017 Aspire Purchase Agreement. On any trading day on which the closing sale price of our common stock exceeds $0.25, we have the right, in our sole discretion, to present Aspire Capital with a purchase notice, directing Aspire Capital (as principal) to purchase up to 100,000 shares of our common stock per trading day, for up to $6.5 million of our common stock in the aggregate at a per share price, calculated by reference to the prevailing market price of our common stock (as provided in the 2017 Aspire Purchase Agreement). As of the date of this Annual Report, no shares of common stock have been sold to Aspire Capital under the 2017 Aspire Purchase Agreement.Item.
As a condition to the 2017 Aspire Purchase Agreement, we issued 137,324 shares of our common stock to Aspire Capital as a commitment fee (the “Commitment Shares”). The issuance of the Commitment Shares were exempt from registration under the Securities Act of 1933, as amended (the “Securities Act”), pursuant to the exemption for transactions by an issuer not involving any public offering under Section 4(a)(2) of the Securities Act.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Item 6. Selected Financial Data[Reserved]
Not applicable.
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of OperationsOperations.
TheYou should read the following discussion and analysis should be readof our financial condition and results of operations together with ourthe consolidated financial statements and the related notes appearingthat are included elsewhere in this Annual Report. This discussion contains forward-looking statements reflecting ourbased upon current expectations that involve risks and uncertainties. See “Special Note Regarding Forward-Looking Statements and Industry Data” for a discussion of the uncertainties, risks and assumptions associated with these statements. ActualOur actual results and the timing of events couldmay differ materially from those discussedanticipated in ourthese forward-looking statements as a result of manyvarious factors, including those set forth under “Risk Factors” and elsewhereor in other parts of this Annual Report. See “Cautionary Note Regarding Forward-Looking Statements” for additional information. Unless otherwise indicated, all information in this Annual Report.Report on Form 10-K gives effect to a 1-for-10 reverse stock split of our common stock that became effective on November 23, 2022, and all references to shares of common stock outstanding and per share amounts give effect to the reverse stock split.
Overview
Ritter Pharmaceuticals, Inc. develops novel therapeutic products that modulate the gut microbiome to treat gastrointestinal diseases. We are advancing gut health researchan early-clinical-stage therapeutics company focused on developing treatments for adult and pediatric cancer. Our business now consists of one early-clinical-stage therapeutic program (QN-302) and one preclinical therapeutic program (Pan-RAS).
Our lead program, QN-302, is an investigational small molecule G-quadruplexes (G4)-selective transcription inhibitor with strong binding affinity to G4s prevalent in cancer cells (such as pancreatic cancer). Such binding could, by exploringstabilizing the metabolic capacityG4s against DNA “unwinding,” help inhibit cancer cell proliferation. QN-302 is currently undergoing a Phase 1a clinical trial at START Midwest in Grand Rapids, Michigan, and HonorHealth in Scottsdale, Arizona.
Our Pan-RAS program, which is currently at the preclinical stage, consists of a family of RAS oncogene protein-protein interaction inhibitor small molecules believed to inhibit or block mutated RAS genes’ proteins from binding to their effector proteins thereby leaving the proteins from the mutated RAS unable to cause further harm. In theory, such mechanism of action may be effective in the treatment of about one quarter of all cancers, including certain forms of pancreatic, colorectal, and lung cancers. The investigational compounds within our Pan-RAS portfolio are designed to suppress the interaction of endogenous RAS with c-RAF, upstream of the gut microbiotaKRAS, HRAS and translating the functionality of prebiotic-based therapeutics into applications intended to have a meaningful impact on a patient’s health.NRAS effector pathways.
We completed a Phase 2a clinical trial of our leading product candidate, RP-G28, an orally administered, high purity oligosaccharide in November 2011.
We completed a Phase 2b multi-center, randomized, double-blind, placebo-controlled, parallel group trial of RP-G28 in October 2016. Topline results of the trial were announced in March 2017. Due to inconsistent data results from one study site, the data from this site was excluded from the primary analysis population (Efficacy Subset mITT). After excluding the data from the one anomalous study site, results showed a clinically meaningful benefit to subjects in the reduction of lactose intolerance symptoms across a variety of outcome measures. The majority of analyses showed positive outcome measures and the robustness of the data point to a clear drug effect. Treatment patients not only reported meaningful reduced symptoms, but also 30 days after taking the treatment, patients reported adequate relief from lactose intolerance symptoms and satisfaction with the results of the treatment, with RP-G28 preventing or treating their lactose intolerance symptoms. Greater milk and dairy product consumption was also reported by patients.
A subset of subjects from our Phase 2b clinical trial has been rolled intoOn November 23, 2022, we effected a 12-month extension study to evaluate long-term durability of treatment. The study is also evaluating each participant’s microbiome, expanding our knowledge of the effects that RP-G28 may have on adapting the gut microbiota in a beneficial manner. We completed this study in the fourth quarter of 2017.
We held an End-of-Phase 2 meeting with the FDA’s Division of Gastroenterology and Inborn Errors Products in August 2017. The purpose of the meeting was to obtain the FDA’s feedback on our Phase 3 program. We reached general consensus with the FDA on certain elements1-for-10, reverse stock split of our current Phase 3 programoutstanding shares of common stock (the “Reverse Stock Split”). The Reverse Stock Split reduced our shares of outstanding common stock, stock options, and have received clear guidance and recommendations on many necessary componentswarrants to purchase shares of our Phase 3 program; including the clinical, non-clinical, and chemistry, manufacturing and controls (CMC) requirements needed to support an NDA submission.
Wecommon stock. Fractional shares of common stock that would have incorporated much of this guidance into our Phase 3 program. Our current Phase 3 clinical program will consist of two confirmatory clinical trials of similar trial design and size as our Phase 2b clinical trial and will include additional components that may allow for claims for durability of effect. These additional trials may be run in parallel.
We have devoted substantially all of our resources to development efforts relating to RP-G28, including conducting clinical trials of RP-G28, providing general and administrative support for these operations and protecting our intellectual property. We currently do not have any products approved for sale and we have not generated any revenue from product sales since our inception.
We have incurred net losses in each year since our inception, including net losses of approximately $7.9 million for the year ended December 31, 2017. We had an accumulated deficit of approximately $53.3 million as of December 31, 2017. Substantially all our net lossesotherwise resulted from costs incurredthe Reverse Stock Split were rounded down to the nearest whole share and cash in connection with our researchlieu of fractional shares was paid to stockholders. All share and development programs, stock-based compensation,per share data for all periods presented in this Annual Report on Form 10-K have been adjusted retrospectively to reflect the Reverse Stock Split. The number of authorized shares of common stock and from general and administrative costs associated with our operations.the par value per share remains unchanged.
We expect to continue to incur significant expenses and increasing operating losses for at least the next several years. We anticipate that our expenses will increase substantially as we:
We do not expect to generate revenuebe profitable before products from product sales unlessour therapeutics pipeline are commercialized. To experience losses while therapeutic products are still under development is, of course, typical for biotechnology companies.
Recent Developments
Phase 1 Clinical Trial of QN-302
On August 1, 2023, we announced that the FDA has cleared our IND application for QN-302. Based on this clearance, we chose Translational Drug Development, LLC (“TD2”) to serve as our contract research organization to conduct a Phase 1 clinical trial in patients with advanced or metastatic solid tumors. The Phase 1 trial (NCT06086522) is designed as a multicenter, open-label, dose escalation, safety, pharmacokinetic, and untilpharmacodynamic study with dose expansion to evaluate safety, tolerability, and antitumor activity of QN-302 in patients with advanced solid tumors that have not responded to or that have recurred following treatment with available therapies. On November 7, 2023, we successfully complete developmentannounced that the first patient had been enrolled and obtain marketing approval for one or moredosed in the dose escalation (Phase 1a) portion of our product candidates, whichthe study. Subject to available funding (which is, however, not all currently in hand), we expect will take aanticipate that Phase 1a of the trial can be completed by the end of 2024. The exact number of yearspatients to be enrolled in the trial will depend on the observed safety profile, which will determine the number of patients per dose level, as well as the number of dose escalations required to meet the Maximum Tolerated Dose (“MTD”). Once the MTD has been established in dose escalation, dose expansion will begin.
Sale of Diagnostics Business
On July 20, 2023, we sold all of the issued and outstanding shares of common stock of Qualigen, Inc., a wholly-owned subsidiary and the legal entity operating our FastPack™ diagnostic business, to Chembio Diagnostics, Inc. (“Chembio”), a subsidiary of Biosynex, S.A. As consideration for the shares of Qualigen, Inc., we received cash payments of approximately $4.9 million, which payment is subject to significant uncertainty.post-closing adjustments. An additional $450,000 was delivered by Chembio to an escrow account to satisfy our indemnification obligations. Any amounts remaining in the escrow account that have not been offset or reserved for claims will be released to us within five business days following January 20, 2025. Following the consummation of the transaction, Qualigen, Inc. became a wholly-owned subsidiary of Chembio.
Amendment and Settlement Agreement with NanoSynex Ltd.
On July 20, 2023, we entered into and effectuated the NanoSynex Amendment, by which we agreed to, among other things, forfeit 281,000 Series B Preferred Shares of NanoSynex held by us, resulting in our ownership in NanoSynex being reduced from approximately 52.8% to approximately 49.97% of the voting equity of NanoSynex. In addition, we agreed to cancel approximately $3.0 million of promissory notes which NanoSynex had issued to us under the NanoSynex Funding Agreement, relieving NanoSynex of any repayment obligations to us with respect to such notes. The NanoSynex Amendment superseded any NanoSynex Funding Agreement obligations to provide funding to NanoSynex, except we agreed to provide future loans as follows: (i) $560,000 on or before November 30, 2023, and (ii) $670,000 on or before March 31, 2024. However, on November 22, 2023, in full settlement of any additional funding obligations to NanoSynex, we forfeited certain of our shares of Series A-1 Preferred Stock of NanoSynex in an amount that reduced our ownership in NanoSynex from approximately 49.97% to 39.90%. Accordingly, we anticipate that we will need to raise additional capital prior to the commercialization of RP-G28. Until such time, if ever, as we can generate substantial revenueNanoSynex was deconsolidated from product sales, we expect to finance our operating activities through a combination of equity offerings (including shares sold to Aspire Capital pursuant to the 2017 Aspire Purchase Agreement), debt financings, government or other third-party funding, commercialization, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Our failure to raise capital or enter into such other arrangements as and when needed would have a negative impact on our financial conditionstatements as of July 20, 2023, and our ability to develop our product candidates.
Financial Overview
Revenue
We have not generated any revenue since our inception.is reported as Discontinued Operations in this Annual Report. Our ability to generate revenueinvestment in NanoSynex will be accounted for in the future will depend almost entirely on our ability to successfully develop, obtain regulatory approval for and then successfully commercialize RP-G28 in the United States. In the event we choose to pursue a partnering arrangement to commercialize RP-G28 or other products outside the United States, we would expect to initiate additional research and development and clinical trial activities in the future.as an equity method investment.
Research and Development Expenses
Since our inception, we have focused our resources on our research and development activities, including conducting nonclinical studies and clinical trials, manufacturing development efforts and activities related to regulatory filings for RP-G28. Our research and development expenses consist primarily of:
From inception through December 31, 2017, we have incurred approximately $22.1 million in research and development expenses. We plan to increase our research and development expenses for the foreseeable future as we continue the development of RP-G28 for the reduction of symptoms associated with lactose intolerance in patients and other indications, subject to the availability of additional funding.
The successful development of RP-G28 is highly uncertain. At this time, we cannot reasonably estimate the nature, timing or costs of the efforts that will be necessary to complete the development of RP-G28 or when, if ever, net cash inflows from RP-G28 may commence. This is due to the numerous risks and uncertainties associated with developing drugs, including the uncertainty of:
For example, if the FDA or another regulatory authority were to require us to conduct clinical trials beyond those that we currently anticipate will be required for the completion of the clinical development of RP-G28 or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development.
Patent Costs
Patent costs consist primarily of professional fees for legal services to prosecute patents and maintain patent rights.
General and Administrative Expenses
General and administrative expenses include allocation of facilities costs, salaries, benefits, and stock-based compensation for employees, professional fees for directors, fees for independent contractors and accounting and legal services.
We expect that our general and administrative expenses will increase if RP-G28 is approved for commercialization. We believe that these increases will likely include increased costs for director and officer liability insurance, and increased fees for outside consultants, lawyers and accountants, among other expenses.
Interest Income and Interest Expense
Interest income consists of interest earned on our cash.
Critical Accounting Policies and Estimates
Our consolidated financial statements historically have not separated our diagnostics-related activities from our therapeutics-related activities. All of our historically reported revenue was diagnostics-related. Before the third quarter of 2023, our reported expenses represented the total of our diagnostics-related and therapeutics-related expenses. In this Annual Report, all diagnostics-related revenues and expenses have been reclassified to discontinued operations (See Note 5 - Discontinued Operations).
| 24 |
This discussion and analysis is based on our consolidated financial statements, which have been prepared in accordance with generally accepted accounting principles in the United States (“GAAP”).U.S. GAAP. The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses and the disclosure of contingent assets and liabilities in our consolidated financial statements. On an ongoing basis, we evaluate our estimates and judgments, including those related to impairment of goodwill and other intangible assets, fair value of financial instruments, research and development costs, accrued expenseswarrant liabilities, and stock-based compensation. We base our estimates on historical experience, known trends and events and various other factors we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in Note 31 to our consolidated financial statements appearing in “Item 8. Financial Statements and Supplementary Data,” we believe that the following accounting policies are the most critical to aid you in fully understanding and evaluating our financial condition and results of operations.operations:
| ● | Research and development | |
| ● | Discontinued operations | |
| ● | Impairment of long-lived assets | |
| ● | Business combinations | |
| ● | Derivative financial instruments and warrant liabilities | |
| ● | Stock-based compensation | |
| ● | Income taxes |
Fair ValueWarrant Liabilities
In 2004, Qualigen, Inc. issued Series C preferred stock warrants to investors and brokers in connection with a private placement. These warrants were subsequently extended and survived the May 2020 Ritter reverse recapitalization transaction and are now exercisable for Qualigen Therapeutics common stock. These warrants contain a provision that if the Company issues shares (except in certain defined scenarios) at a price below the warrants’ exercise price, the exercise price will be re-set to such new price and the number of Financial Instruments
Fair value measurement guidelines are prescribed by GAAPshares underlying the warrants will be increased in the same proportion as the exercise price decrease. For accounting purposes, such warrants give rise to value financial instruments. The guidance includes a definitionwarrant liabilities. Accounting principles generally accepted in the United States of fair value, prescribes methods for measuring fair value, establishes a fair value hierarchy based on the inputs usedAmerica (“U.S. GAAP”) require us to measure fair value and expands disclosures about the use of fair value measurements.
The valuation techniques utilized are based upon observable and unobservable inputs. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect internal market assumptions. Assets are classified in their entirety based on the lowest level of input that is significant torecognize the fair value measurement.
These two types of inputs create the following fair value hierarchy:
Level 1 — Quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. Level 1 primarily consists of financial instruments whose value is based on quoted market prices such as exchange-traded instruments and listed equities.
Level 2 — Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly or indirectly. Level 2 includes financial instruments that are valued using models or other valuation methodologies. These models consider various assumptions, including volatility factors, current market prices and contractual prices for the underlying financial instruments. Substantially all of these assumptions are observablewarrants as warrant liabilities on our Consolidated Balance Sheets and to reflect period-to-period changes in the marketplace, can be derived from observable data or are supported by observable levels at which transactions are executed in the marketplace.
Level 3 — Unobservable inputs for the asset or liability. Financial instruments are considered Level 3 when their fair values are determined using pricing models, discounted cash flows or similar techniques and at least one significant model assumption or input is unobservable
The carrying amounts reported in the balance sheet for cash and cash equivalents, prepaid expenses, accounts payable, accrued expenses, and the notes payable approximate the fair values due to the short-term nature of the instruments.
Research and Development Costs
We expense the cost of research and development as incurred. Research and development expenses comprise costs incurred in performing research and development activities, including clinical study costs, contracted services, and other external costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity is performed or when the goods have been received, rather than when payment is made, in accordance with ASC 730,Research and Development.
Accrued Expenses
As part of the process of preparing our financial statements, we are required to estimate our accrued expenses. This process involves reviewing quotations and contracts, identifying services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of the actual cost. The majority of our service providers invoice us monthly in arrears for services performed or when contractual milestones are met. We make estimates of our accrued expenses as of each balance sheet date in our financial statements based on facts and circumstances known to us at that time. We periodically confirm the accuracy of our estimates with the service providers and make adjustments if necessary. The significant estimates in our accrued research and development expenses include fees due to service providers.
We base our expenses on our estimates of the services received and efforts expended pursuant to quotes and contracts with our service providers that conduct research and development on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. There may be instances in which payments made to our vendors will exceed the level of services provided and result in a prepayment of the research and development expense. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from our estimate, we adjust the accrual accordingly. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and could result in us reporting amounts that are too high or too low in any particular period.
Stock-Based Compensation
Stock-based compensation cost for equity awards granted to employees and nonemployees is measured at the grant date based on the calculated fair value of the award using the Black-Scholes option-pricing model,warrant liabilities on our Consolidated Statements of Operations. The estimated fair value of these warrant liabilities was approximately $0.1 million and is recognized as an expense, under the straight-line method, over the requisite service period (generally the vesting period$3.6 million at December 31, 2023 and 2022, respectively. There were 455,623 of the equity grant). If we determine that other methods are more reasonable, or other methods for calculating these assumptions are prescribed by regulators,warrants outstanding at December 31, 2023 and 1,349,571 of these warrants outstanding at December 31, 2022.
Because the fair value calculated forof the above liability classified warrants will be determined each quarter on a “mark-to-market” basis , significant variability in our future quarterly and annual Consolidated Statement of Operations and Consolidated Balance Sheets could occur based on changes in our public market common stock price. Pursuant to U.S. GAAP, a quarter-to-quarter increase in our stock options could change significantly. Higher volatility and longer expected livesprice would result in an increase to stock-based compensation expense to non-employees determined at the date of grant.
In addition to the assumptions used in the Black-Scholes option-pricing model, we also estimate a forfeiture rate to calculate the stock-based compensation for our equity awards. We will continue to use judgment in evaluating the expected volatility, expected terms and forfeiture rates utilized for our stock-based compensation calculations on a prospective basis.
Emerging Growth Company Status
On April 5, 2012, the Jumpstart Our Business Startups Act of 2012 (“JOBS Act”) was enacted. Section 107fair value of the JOBS Act provides that an “emerging growth company” can take advantagewarrant liabilities and a quarter-to-quarter decrease in our stock price would result in a decrease in the fair value of the extended transition period provided in Section 7(a)(2)(B)warrant liabilities.
On December 22, 2022, as part of the Securities Act of 1933, as amended (the Securities Act), for complying with new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting standards until those standards would otherwise apply2022 Debenture financing, we issued to private companies. We have electedAlpha a common stock warrant (exercisable from June 22, 2023 through June 22, 2028) to use the extended transition period for complying with new or revised accounting standards under Section 102(b)(1) of the JOBS Act. This election allows us to delay the adoption of new or revised accounting standards that have different effective dates for public and private companies until those standards apply to private companies. As a result of this election, our financial statements may not be comparable to companies that comply with public company effective dates.
We are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements provided by the JOBS Act. Subject to certain conditions set forth in the JOBS Act, as an “emerging growth company,” we intend to rely on certain of these exemptions, including without limitation, (i) providing an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and (ii) complying with any requirement that may be adopted by the PCAOB regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an emerging growth company until the earlier of (i) the last day of the fiscal year (a) following the fifth anniversary of the date we completed our initial public offering, which was June 29, 2015, (b) in which we have total annual gross revenue of at least $1.07 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market valuepurchase 2,500,000 shares of our common stock that is held by non-affiliates exceeded $700.0 million asstock. The exercise price of the priorwarrant was modified from $1.65 to $0.73 on December 5, 2023, and was further modified to $0.26 on February 27, 2024. The warrant may be exercised by Alpha, in whole or in part before June 30th,22, 2028. The warrant was originally liability classified, but was modified on December 5, 2023 to allow for equity classification. The estimated fair value of this warrant upon reclassification from warrant liabilities to equity was approximately $1.6 million and (ii) the dateestimated fair value of this warrant which was included in warrant liabilities-related party on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.December 31, 2022 was approximately $2.8 million.
Results of Operations
Comparison of the years endedYears Ended December 31, 20172023 and 20162022
For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| EXPENSES | ||||||||
| General and administrative | $ | 6,095,607 | 10,274,600 | |||||
| Research and development | 5,209,250 | 4,486,120 | ||||||
| Total expenses | 11,304,857 | 14,760,720 | ||||||
| LOSS FROM OPERATIONS | (11,304,857 | ) | (14,760,720 | ) | ||||
| OTHER EXPENSE (INCOME), NET | ||||||||
| Gain on change in fair value of warrant liabilities | (2,035,469 | ) | (907,203 | ) | ||||
| Interest expense, net | 1,524,722 | 34,397 | ||||||
| Loss on voluntary conversion of convertible debt | 1,077,287 | — | ||||||
| Loss on debt extinguishment | 625,653 | — | ||||||
| Loss on fixed asset disposal | 21,747 | — | ||||||
Other income, net | (38,994 | ) | ||||||
| Total other expense (income), net | 1,174,946 | (872,806 | ) | |||||
| LOSS BEFORE (BENEFIT) PROVISION FOR INCOME TAXES | (12,479,803 | ) | (13,887,914 | ) | ||||
| (BENEFIT) PROVISION FOR INCOME TAXES | (4,793 | ) | 6,548 | |||||
| NET LOSS FROM CONTINUING OPERATIONS | (12,475,010 | ) | (13,894,462 | ) | ||||
| DISCONTINUED OPERATIONS | ||||||||
| Loss from discontinued operations, net of tax | (683,008 | ) | (7,140,181 | ) | ||||
| Loss on disposal of discontinued operations, net of tax | (602,232 | ) | — | |||||
| LOSS FROM DISCONTINUED OPERATIONS | (1,285,240 | ) | (7,140,181 | ) | ||||
| NET LOSS | (13,760,250 | ) | (21,034,643 | ) | ||||
| Net loss attributable to non-controlling interest from discontinued operations | (343,038 | ) | (2,394,100 | ) | ||||
| Net loss attributable to Qualigen Therapeutics, Inc. | $ | (13,417,212 | ) | $ | (18,640,543 | ) | ||
| Net loss per common share, basic and diluted - continuing operations | $ | (2.46 | ) | $ | (3.62 | ) | ||
| Net loss per common share, basic and diluted - discontinued operations | $ | (0.19 | ) | $ | (1.24 | ) | ||
| Weighted—average number of shares outstanding, basic and diluted | 5,072,709 | 3,840,340 | ||||||
| Other comprehensive loss, net of tax | ||||||||
| Net loss | $ | (13,760,250 | ) | $ | (21,034,643 | ) | ||
| Foreign currency translation adjustment from discontinued operations | 119,473 | 50,721 | ||||||
| Other comprehensive loss | (13,640,777 | ) | (20,983,922 | ) | ||||
| Comprehensive loss attributable to noncontrolling interest from discontinued operations | (304,735 | ) | (2,394,100 | ) | ||||
| Comprehensive loss attributable to Qualigen Therapeutics, Inc. | $ | (13,336,042 | ) | $ | (18,589,822 | ) | ||
| 26 |
The following table summarizes the results of our operations for the years ended December 31, 2017 and 2016, together with the changes in those items in dollars and as a percentage:
| For the Years Ended December 31, | Dollar | Percentage | ||||||||||||||
| 2017 | 2016 | Change | Change | |||||||||||||
| Statement of Operations Data: | ||||||||||||||||
| Operating costs and expenses | ||||||||||||||||
| Research and development | $ | 2,874,184 | $ | 13,292,488 | $ | (10,418,304 | ) | (78 | )% | |||||||
| Patent costs | 250,372 | 272,514 | (22,142 | ) | (8 | )% | ||||||||||
| General and administrative | 4,777,902 | 4,881,725 | (103,823 | ) | (2 | )% | ||||||||||
| Total operating costs and expenses | 7,902,458 | 18,446,727 | (10,544,269 | ) | (57 | )% | ||||||||||
| Loss from operations | (7,902,458 | ) | (18,446,727 | ) | (10,544,269 | ) | (57 | )% | ||||||||
| Other income | ||||||||||||||||
| Interest income | 40,227 | 60,879 | (20,652 | ) | (34 | )% | ||||||||||
| Other (expense) income | (1,627 | ) | 1,214 | (2,841 | ) | (134 | )% | |||||||||
| Total other income | 38,600 | 62,093 | (23,493 | ) | (38 | )% | ||||||||||
| Net loss applicable to common stockholders | $ | (7,863,858 | ) | $ | (18,384,634 | ) | $ | (10,520,776 | ) | (57 | )% | |||||
Research and Development Expenses
ResearchGeneral and developmentAdministrative Expenses
General and administrative expenses decreased by approximately $10.4from $10.3 million or (78%), for the year ended December 31, 2017 as compared2022 to the year ended December 31, 2016. The primary reason for the decrease is that our Phase 2b clinical trial, which was initiated in March 2016, was completed during the fourth quarter of 2016. A decrease of approximately ($11.5)$6.1 million attributable to fees paid to our third-party CRO for the year ended December 31, 20162023. This decrease was due to a $3.8 million decrease in stock-based compensation expense, a $0.4 million decrease in payroll related expenses, a $0.3 million decrease in insurance expenses, and a $0.2 million decrease in license fees, offset by an increase of $0.5 million in professional fees. (The foregoing comparison, and all other comparisons presented in this Item, exclude Qualigen, Inc. and NanoSynex, Ltd. results for both years.)
Research and Development Costs
Research and development expenses increased from $4.5 million for the year ended December 31, 2022 to approximately $5.2 million for year ended December 31, 2023. This increase in research and development expenses for the year ended December 31, 2023 compared to for the year ended December 31, 2022 was primarily due to a $2.1 million increase in pre-clinical and clinical research costs for QN-302, offset by a $1.0 million decrease in pre-clinical research costs for QN-247 a $0.3 million decrease in preclinical research costs for Pan-RAS, and a $0.1 million decrease in preclinical research costs for QN-165.
Other Expense (Income)
Change in Fair Value of Warrant Liabilities
During the year ended December 31, 2023 we experienced a $2.0 million gain in other income because of the change in fair value of the warrant liabilities arising from our liability classified warrants described above. The estimated fair value of these warrant liabilities decreased to $0.1 million as partof December 31, 2023 from $3.6 million as of December 31, 2022 due to a reduction in fair value of the warrant liabilities resulting from an associated decrease in the market price of our Phase 2b clinical trialcommon stock, and the reclassification at fair value of a liability classified warrant to equity of $1.6 million. For the year ended December 31, 2022, the gain on change in fair value of warrant liabilities was offset by approximately $0.7$0.9 million spentdue to an associated decrease in the market price of our common stock. Typically, a decline in our stock price would result in a decline in the fair value of our warrant liabilities, generating a gain, while an increase in our stock price would result in an increase in the fair value of our warrant liabilities, generating a loss.
The remaining liability classified warrants expire on regulatory consultingJune 26, 2024. Because the fair value of the warrant liabilities will be determined each quarter on a “mark-to-market” basis, this item is likely to, until then, continue to result in variability in our future quarterly Consolidated Statements of Operations based on unpredictable changes in our public market common stock price and the number of warrants outstanding at the end of each quarter.
Interest (Income) Expense, Net
There was $1.5 million in net interest expense during the year ended December 31, 2017 in preparation for our Phase 3 clinical study as well as $0.4 million in fees associated with our extension study to evaluate the long-term durability of treatment.
Patent Costs
Patent costs were approximately in $250,000 and $273,000 for the years ended December 31, 2017 and 2016, respectively, representing a decrease of approximately $22,000, or (8%). The primary reason for the decrease is that our costs and expenses related to the maintenance of patent rights, the prosecution of patents, the application for the issuance of patents, as well as the preparation to file national phase applications in certain foreign countries was higher in 2016 than 2017.
General and Administrative Expenses
General and administrative expenses decreased by approximately $104,000, or (2%), for the year ended December 31, 2017 as2023 compared to the year ended December 31, 2016. The decrease in general and administrative expenses was mainly due to a decrease in stock-based compensation expensenet interest income of approximately $0.5 million$34,000 during the year ended December 31, 2017 partially offset by an2022. The increase in professional feeswas due to the interest on the 2022 Debenture.
Loss on Voluntary Conversion of Convertible Debt
During the year ended December 31, 2023 we issued 841,726 shares of common stock upon Alpha’s partial voluntary conversion of the 2022 Debenture at $1.32 per share for a total of $1,111,078 principal converted. Upon conversion, we recognized a loss on voluntary conversion of convertible debt of approximately $0.3$1.1 million.
Other IncomeLoss on Debt Extinguishment
Interest income decreased by $21,000, or (34%)
During the year ended December 31, 2023, we issued 309,665 shares of common stock in lieu of cash for the October and December 2023 monthly redemptions, for a total of $220,000 principal redeemed, pursuant to the terms of the 2022 Debenture at a weighted average share price of $0.71. Upon redemption in shares, we recognized a loss on partial debt extinguishment of $34,315. The modification of the 2022 Debenture during the year ended December 31, 20172023 met the criteria to be accounted for as compared toa debt extinguishment in the amount of $591,338. Accordingly, we recognized an additional loss on partial debt extinguishment of that amount.
Loss on Fixed Asset Disposal
During the year ended December 31, 2016, primarily2023, we incurred a $21,747 loss on fixed asset disposal due to usedisposal of cash to complete the Phase 2b study in 2016. This was partially offset by the interest earned in the fourth quarter of 2017 from the proceeds of our follow-on offering (described below).research and development equipment previously used for QN-165.
Liquidity and Capital ResourcesGoing Concern
SourcesOur financial position is weak. As of Liquidity
Since our inception,December 31, 2023, we had approximately $0.4 million in cash and net accounts payable of over $2.2 million. We are in arrears on accounts payable to important partners. We have incurred netrecurring losses and negative cash flows from operations and as of December 31, 2017, we hadhave an accumulated deficit of approximately $53.3 million. Substantially all our net losses resulted from costs incurred in connection with our research and development programs, stock-based compensation, and from general and administrative costs associated with our operations.
At$116.8 million at December 31, 2017,2023. We expect to continue to incur losses subsequent to the consolidated balance sheet date of December 31, 2023. For the years ended December 31, 2023 and 2022, we had working capitalused cash of $20.1$10.3 million and cash of $22.6 million.$13.2 million, respectively, in operations. We have not generated any product revenues and have not achieved profitable operations.sold our Qualigen, Inc. FastPack® diagnostics products business in 2023.
Aspire Capital Financing Arrangement
On May 4, 2017,February 26, 2024, we entered into a Securities Purchase Agreement (“Agreement”) with Alpha. The transactions contemplated by the Agreement closed on February 27, 2024, at which time we delivered to Alpha a new Debenture and warrant, as described in this paragraph, and Alpha paid the Company a cash purchase price of $500,000 (less expenses). Pursuant to the Agreement, we issued to Alpha an 8% Convertible Debenture (the “2024 Debenture”) in the principal amount of $550,000. The 2024 Debenture has a maturity date of December 31, 2024 and is convertible, at any time, and from time to time, at Alpha’s option, into shares of common stock of the Company, at $0.6111 per share, subject to adjustment as described in the 2024 Debenture. The 2024 Debenture accrues interest on its outstanding principal balance at the rate of 8% per annum, payable at maturity. Pursuant to the terms of the Agreement, we also issued to Alpha a 5-year common stock purchase agreementwarrant to purchase (at $0.26 per share) 900,016 shares of common stock of the Company. We also granted to Alpha an option, exercisable until July 1, 2024, to purchase from us additional 8% Convertible Debentures, of like tenor, with Aspire Capital. The 2017 Aspire Purchase Agreement provides access to usface amounts of up to an aggregate of $6.5$1,100,000 (and with a proportional number of accompanying common stock warrants of like tenor, up to a total of 1,800,032 additional warrants), which would (if and when Alpha exercises such option) provide us up to an additional $1.0 million throughin cash proceeds (less expense reimbursement, and not including any possible cash proceeds from any future exercise of the additional warrants).
We currently expect our cash balances to fund operations only into the second quarter of 2024. We expect to continue to have net losses and negative cash flow from operations, which will challenge our liquidity. These factors raise substantial doubt regarding our ability to continue as a going concern for the one-year period following the date that the financial statements in this Annual Report were issued.
There is no assurance that we will ever achieve profitable operations, or, if achieved, could be sustained on a continuing basis. In order to fully execute our business plan, we will require significant additional financing for planned research and development activities, capital expenditures, QN-302 clinical trials, and preclinical development of Pan-RAS, as well as commercialization activities.
Historically, our principal sources of cash have, in addition to revenue from FastPack product sales and license revenues (see Note 5 - Discontinued Operations), included proceeds from the issuance of common and preferred equity and proceeds from the issuance of debt. In December 2022 and February 2024 we raised approximately $3.0 million and $0.5 million, respectively from the sale of shares of our common stock, over a 30-month period. In consideration for entering into the 2017 Aspire Purchase Agreement,convertible debentures to Alpha. There can be no assurance that further financing can be obtained on favorable terms, or at all. If we issuedare unable to Aspire Capital 137,324 shares of our common stock with an aggregate dollar value equalobtain funding, we could be required to $97,500.
Under the 2017 Aspire Purchase Agreement, on any trading day we select, we have the right, in our sole discretion, to present Aspire Capital with a purchase notice (each, a “Purchase Notice”), directing Aspire Capital (as principal) to purchase up to 100,000 shares of our common stock per trading day (which may be increased by as much as an additional 2,000,000 shares per trading day by mutual agreement), up to an aggregate of $6.5 million of our common stock, at a per share price (the “Purchase Price”) equal to the lesser of:
The aggregate purchase price payable by Aspire Capital on any one purchase date may not exceed $500,000, unless otherwise mutually agreed.
In addition, on any date on which we submit a Purchase Notice to Aspire Capital in an amount equal to 100,000 sharesdelay, reduce or eliminate research and our stock price is not less than $0.25 per share, we may also, in our sole discretion, present Aspire Capital with a volume-weighted average price purchase notice (each, a “VWAP Purchase Notice”) directing Aspire Capital to purchase an amount of our common stock equal to up to 30% of the aggregate shares of our common stock traded on its principal market on the next trading day (the “VWAP Purchase Date”), as determined by us. The purchase price per share pursuant to such VWAP Purchase Notice is generally 97% of the volume-weighted average price for our common stock traded on its principal market on the VWAP Purchase Date.
We may deliver multiple Purchase Notices and VWAP Purchase Notices to Aspire Capital from time to time during the term of the 2017 Aspire Purchase Agreement, so long as the most recent purchase has been completed.
The Purchase Agreement provides that we may not effect any sales under the 2017 Aspire Purchase Agreement on any date where the closing sale price of our common stock is less than $0.25. There are no trading volume requirementsdevelopment programs, product portfolio expansion or restrictions under the 2017 Aspire Purchase Agreement,future commercialization efforts, and we control the timing and amount of sales of our common stock to Aspire Capital.
The 2017 Aspire Purchase Agreement provides that the number of shares that maycould be sold pursuant to Aspire Capital will be limited to 2,842,417 (the “Exchange Cap”), which represents 19.99% of our outstanding shares of common stock as of May 2, 2017, unless stockholder approval or an exception pursuant to the rules of the NASDAQ Capital Market is obtained to issue more than 19.99%. This limitation will not apply if, at any time the Exchange Cap is reached and at all times thereafter, the average price paid for all shares issued under the 2017 Aspire Purchase Agreement is equal to or greater than $0.68, which was the consolidated closing bid price of our common stock on May 4, 2017. We are not required or permitted to issue any shares of common stock under the 2017 Aspire Purchase Agreement if such issuance would breach our obligations under the rules or regulations of the NASDAQ Capital Market.
We expect to use the Aspire facility to complement, rather than replace, other financing that may be required during the next twelve monthsunable to continue our operations and support our capital needs.operations.
October 2016 Public Offering
On October 31, 2016, we closed a public offering of 2,127,660 shares of our common stock at a price to the public of $2.35 per share, for net proceeds of approximately $4.4 million, after deducting underwriting discounts and commissions and offering expenses payable by us. The offering was made pursuant to a shelf registration statement on Form S-3 (Registration Number 333-213087).
October 2017 Public Offering
On October 3, 2017, we closed a public offering of (i) 34,550,000 Class A Units consisting of 34,550,000 shares of our common stock and warrants to purchase 34,550,000 shares of our common stock, at a public offering price of $0.40 per unit, and (ii) 9,180 Class B Units consisting of 9,180 shares of our Series A Convertible Preferred stock, with a stated value of $1,000, and convertible into an aggregate of 22,950,000 shares of our common stock, and warrants to purchase 22,950,000 shares of our common stock, at a public offering price of $1,000 per unit. We received approximately $21.0 million in net proceeds from this offering, after deducting the underwriting discounts and commissions and estimated offering expenses payable by us. The offering was made pursuant to a shelf registration statement on Form S-3 (Registration Number 333-219147).
Cash Flows
The following table sets forth the significant sources and uses of cash for the periods set forth below:
| For the Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Net cash (used in) provided by: | ||||||||
| Operating activities | $ | (7,397,912 | ) | $ | (15,208,718 | ) | ||
| Investing activities | (7,678 | ) | (8,063 | ) | ||||
| Financing activities | 22,991,279 | 6,443,497 | ||||||
| Net (decrease) increase in cash | $ | 15,585,689 | $ | (8,773,284 | ) | |||
Operating Activities
The use of cash in both periods resulted primarily from our net losses adjusted for non-cash charges and changes in components of working capital. Net cash used in operating activities was approximately $7.4 million during the year ended December 31, 2017 compared to $15.2 million during the year ended December 31, 2016. The decrease in cash used in operating activities was driven primarily by the completion of the Phase 2b clinical trial in October 2016 with 368 patients at 18 clinical sites.
Investing Activities
Net cash used in investing activities of was approximately $8,000 during the years ended December 31, 2017 and 2016. These expenditures were for purchases of furniture and equipment used in the business.
Financing Activities
Net cash provided by financing activities was approximately $23.0 million during the year ended December 31, 2017 compared to $6.4 million during the year ended December 31, 2016. During the 2017 year, we received proceeds from the October 2017 public offering. The cash provided by financing activities in 2016 was from the October 2016 public offering and the issuance of shares to Aspire Capital.
Future Funding Requirements
To date, we have not generated any revenue. We do not know when, or if, we will generate any revenue from product sales. We do not expect to generate significant revenue from product sales unless and until we obtain regulatory approval of and commercialize RP-G28 or any of our other product candidates. At the same time, we expect our expenses to increase in connection with our ongoing development activities, particularly as we continue the research, development and clinical trials of, and seek regulatory approval for, RP-G28. In addition, subject to obtaining regulatory approval of RP-G28, we expect to incur significant commercialization expenses for product sales, marketing, manufacturing and distribution. We anticipate that we will need substantial additional funding in connection with our continuing operations.
Based upon our current operating plan, we believe that our existing cash and cash equivalents, together with interest and any proceeds received from our sale of shares of common stock to Aspire Capital in the future pursuant to the 2017 Aspire Purchase Agreement, will enable us to fund our operating expenses and capital expenditure requirements through 2018.
Our future capital requirements will depend on many factors, including:
Until such time, if ever, as we can generate substantial revenue from product sales, we expect to finance our cash needs through a combination of equity offerings, debt financings, government or other third-party funding, commercialization, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interests of our common stockholders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our common stockholders. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through government or other third-party funding, commercialization, marketing and distribution arrangements or other collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us. In addition, any future financing (depending on the terms and conditions) may be subject to the approval of Alpha under the terms of the Debentures and/or trigger certain adjustments to the Debentures or warrants held by Alpha.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The financial statements do not include any adjustments that would be necessary should the Company be unable to continue as a going concern, and therefore, be required to liquidate its assets and discharge its liabilities in other than the normal course of business and at amounts that may differ from those reflected in the accompanying financial statements.
Our current liabilities at December 31, 2023 include $2.2 million of accounts payable, $0.6 million of accrued expenses and other current liabilities, $0.1 million in warrant liabilities, and $1.3 million of convertible debt to a related party.
Contractual Obligations and Commitments
We have no material contractual obligations that are not fully recorded on our consolidated balance sheets or fully disclosed in the notes to the financial statements.
License and Sponsored Research Agreements
We have obligations under various license and sponsored research agreements to make future payments to third parties that become due and payable on the achievement of certain development, regulatory and commercial milestones (such as the start of a clinical trial, filing for product approval with the FDA or other regulatory agencies, product approval by the FDA or other regulatory agencies, product launch or product sales) or on the sublicense of our rights to another party. We have not included these commitments on our balance sheet because the achievement and timing of these events is not determinable. Certain milestones are in advance of receipt of revenue from the sale of products and, therefore, we may require additional debt or equity capital to make such payments.
| 28 |
We have multiple license and sponsored research agreements with ULRF. Under these agreements, we have taken over development, regulatory approval and commercialization of various drug compounds from ULRF and are responsible for maintenance of the related intellectual property portfolio. We agreed to reimburse ULRF for sponsored research expenses of up to $2.7 million and prior patent costs of up to $112,000 for Pan-RAS. As of December 31, 2023, there were no remaining un-expensed amounts under this sponsored research agreement for Pan-RAS. Under the terms of these agreements, we are required to make patent maintenance payments and payments based upon development, regulatory and commercial milestones for any products covered by the in-licensed intellectual property. The maximum aggregate milestone payments we may be obligated to make per product are $5 million. We will also be required to pay a royalty on net sales of products covered by the in-licensed intellectual property in the low single digits. The royalty is subject to reduction for any third-party payments required to be made, with a minimum floor in the low single digits. We have the right to sublicense our rights under these agreements, but we will be required to pay ULRF a percentage of any sublicense income.
We previously had sponsored research agreements with ULRF for QN-247 and QN-165. As of December 31, 2023, there were no remaining un-expensed amounts under these sponsored research agreements and the agreements were terminated effective August 31, 2022, and November 30, 2021 respectively.
On January 13, 2022, we entered into a License Agreement with UCL Business Limited to obtain an exclusive worldwide in-license of a genomic quadruplex (G4)-selective transcription inhibitor drug development program which had been developed at University College London, including lead and back-up compounds, preclinical data and a patent estate. (UCL Business Limited is the commercialization company for University College London.) We are further developing the program’s lead compound under the name QN-302. The License Agreement requires (if and when applicable) tiered royalty payments in the low to mid-single digits, clinical/regulatory/sales milestone payments, and sharing of a percentage of any non-royalty sublicensing consideration paid to the Company. In November 2023, we became obligated to pay $100,000 to UCL Business Limited upon the first patient dosing of QN-302, which is included in accounts payable in our consolidated balance sheet.
Master ServicesAlpha Convertible Debt
On December 22, 2022, pursuant to the terms of a Securities Purchase Agreement, dated December 21, 2022 (the “Alpha Purchase Agreement”), we issued to Alpha, in exchange for $3,000,000 in cash (less $50,000 for expense reimbursement), the 2022 Debenture with an original face amount of $3,300,000 due on December 22, 2025, plus 2,500,000 common stock warrants exercisable (from June 22, 2023 through June 22, 2028) at $1.65 per share.
Commencing June 1, 2023 and continuing on the first day of each month thereafter until the earlier of (i) December 22, 2025 and (ii) the full redemption of the 2022 Debenture, we must redeem $110,000 plus accrued but unpaid interest, liquidated damages and any amounts then owing under the 2022 Debenture. The Monthly Redemption Amount must be paid in cash; provided that after the first two monthly redemptions, we may (if the Equity Conditions, as defined in the 2022 Debenture, are then satisfied or have been waived) elect to pay all or a portion of a Monthly Redemption Amount in shares of our common stock, based on a conversion price equal to the lesser of (i) the then applicable conversion price of the 2022 Debenture and (ii) 85% of the average of the VWAPs (as defined in the 2022 Debenture) for the five consecutive trading days ending on the trading day that is immediately prior to the applicable Monthly Redemption Date.
The 2022 Debenture accrues interest at the rate of 8% per annum, which began accruing on December 1, 2023, and will be payable on a quarterly basis. Interest may be paid in cash or shares of common stock or a combination thereof at our option; provided that the Equity Conditions have been satisfied.
Alpha has waived the Equity Conditions for certain Monthly Redemption Amounts, but Alpha is not required to continue such waivers beyond May 2024. For the foreseeable future, we do not expect to be able to satisfy the Equity Conditions; as a result, where there is no waiver of the Equity Conditions we would not have the opportunity to make 2022 Debenture payments in the form of stock rather than in the form of cash, even for types of payments for which payment in the form of stock would have been allowed.
The 2022 Debenture is convertible into our common stock at any time at the holder’s option; the conversion price was originally $1.32 but pursuant to a Securities Purchase Agreement amendment it was reduced to $0.73 on December 5, 2023 and then on February 27, 2024 it was adjusted downward to $0.26 per share by virtue of the operation of a “ratchet” antidilution provision. (The exercise price of the warrants issued with the 2022 Debenture was originally $1.65 but pursuant to the Securities Purchase Agreement amendment it was reduced to $0.73 on December 5, 2023 and then on February 27, 2024 it was adjusted downward to $0.26 per share by virtue of the operation of a “ratchet” antidilution provision.)
Both the 2022 Debenture and the accompanying warrants provide for “ratchet” antidilution adjustments to their conversion price and exercise price.
Both the 2022 Debenture and the accompanying warrants include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
| 29 |
We granted Alpha resale registration rights for the common shares underlying the 2022 Debenture and the accompanying warrants.
On December 5, 2023, we entered into an Amendment No. 1 with regard to Securities Purchase Agreement, with Alpha, which, among other things, revised certain provisions of the 2,500,000 warrants to clarify the intention that such 2,500,000 warrants would not be liability-classified for GAAP purposes.
During the year ended December 31, 2023, we recognized an extinguishment loss on voluntary conversion of convertible debt of approximately $1.1 million, an extinguishment loss of $0.6 million upon October and December 2023 share redemptions and the modification of the 2022 Debenture in December 2023, and recorded accrued interest of approximately $1.5 million, in other expenses in the consolidated statements of operations. During the year ended December 31, 2023 we paid Monthly Redemption Amounts of $550,000 in cash and $220,000 in common stock, and as of December 31, 2023 the remaining 2022 Debenture principal balance was approximately $1.4 million, the remaining discount was approximately $0.1 million, and the fair value of the suite of bifurcated embedded derivative features was $0.
Reference is also made to the 2024 Debenture, which was issued to Alpha after the end of the 2023 fiscal year and is described above.
NanoSynex Funding Agreement
On December 30, 2015,As a condition to our acquisition of a majority voting equity interest in NanoSynex from Alpha and NanoSynex, we entered into a Master Service Agreement with Covance, with an effective date of December 29, 2015. Pursuant to the terms of the Master Service Agreement, Covance (or one or more of its affiliates) will provide Phase 1, 2, 3, and 4 clinical services for a clinical study or studies to us, and, at our request, assist us with the design of such studies, in accordance with the terms of separate individual project agreements to be entered into by the parties. The term of the agreement is for three years and will renew automatically for successive one year periods unless Covance is no longer providing services under the agreement or either party has terminated the agreement upon written notice. We may terminate the Master Service Agreement or any individual project agreement entered into under the Master Service Agreement prior to the applicable study’s completion at any time for any reason upon 30 days written notice to Covance, except when the reason for termination is the safety of subjects, in which case it may be terminated immediately. Covance may not terminate any individual project agreement without cause, except when the reason for the termination is the safetyOperational and Technological Funding of subjects, in which case it may be terminated immediately. In the event of a termination of the Master Service Agreement, Covance will be entitled to full payment for (i) work performedNanoSynex (the “Funding Agreement”), on the applicable project upon through the date work on such project is concluded and (ii) reimbursement for all non-cancellable and non-refundable expenses and financial obligations which Covance (or an affiliate) has incurred or undertaken on our behalf.
Clinical Supply and Cooperation Agreement with Ricerche Sperimentali Montale (“Ricerche”) and Inalco SpA (“Inalco”)
Under the terms of the Supply Agreement with RSM, we are required to pay RSM $400,000 within 10 days following FDA approval of a NDA for the first product owned or controlled by us using Improved GOS as its active pharmaceutical ingredient.
Lease Agreement
We lease office space for our headquarters in California. The lease provides for a term of sixty-one (61) months, commencing on October 1, 2015. We pay $9,733 per month in base rent until November 2018, after which our monthly payment will increase ratably per yearMay 26, 2022, pursuant to the termswhich we agreed to fund NanoSynex up to an aggregate of the lease agreement. The base rent payments do not include our proportionate share of any operating expenses, including real estate taxes. We have the option to extend the term of the lease for one five-year term, provided that the rent would beapproximately $10.4 million, subject to market adjustment atNanoSynex’s achievement of certain performance milestones specified in the beginningFunding Agreement and the satisfaction of other terms and conditions described in the renewal term. We will recognize rent expense on a straight-line basis overFunding Agreement.
During the lease term.
Rent expense, recognized on a straight-line basis, was approximately $115,000 for the yearsyear ended December 31, 20172022, we funded a total of approximately $2.4 million and 2016, respectively,in February 2023 we funded an additional $0.5 million to NanoSynex under the Funding Agreement.
On July 20, 2023, we entered into the NanoSynex Amendment, which amended the Funding Agreement, pursuant to which the Company agreed to, among other things, forfeit 281,000 Series B Preferred Shares of NanoSynex held by the Company, resulting in our ownership in NanoSynex being reduced from approximately 52.8% to approximately 49.97% of the voting equity of NanoSynex. In addition, we agreed to cancel approximately $3.0 million of promissory notes which NanoSynex had issued to us under the NanoSynex Funding Agreement, relieving NanoSynex of any repayment obligations to us with respect to such notes. The surrender of shares reducing our interest in NanoSynex from approximately 52.8% to approximately 49.97% occurred on July 20, 2023. Accordingly, NanoSynex was deconsolidated from our financial statements as of July 20, 2023, and is recordedreported as Discontinued Operations in generalthis Annual Report.
The NanoSynex Amendment superseded any payment obligations contemplated by the original Funding Agreement and administrative expensesamended our obligations to provide funding to NanoSynex, except we agreed to provide future funding as follows: (i) $560,000 on or before November 30, 2023, and (ii) $670,000 on or before March 31, 2024, in each case issued in the accompanying statementform of operations.
Off-Balance Sheet Arrangements
We did not have duringa promissory note to the periods presented, and we do not currently have, any off-balance sheet arrangements as defined under SEC rules.
Recent Accounting Pronouncements
In February 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update 2016-02,Leases (Topic 842)(“ASU 2016-02”). The provisions of ASU 2016-02 set out the principles for the recognition, measurement, presentation and disclosure of leases for both parties to a contract (i.e., lessees and lessors). The new standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification will determine whether the lease expense is recognized based on an effective interest method or on a straight line basis over the term of the lease. A lessee is also required to record a right-of-use asset and a lease liability for all leasesCompany with a termface value in the amount of greater than 12 months regardlesssuch funding. However, on November 22, 2023, in full settlement of their classification. Leases with a termany additional funding obligations to NanoSynex, we forfeited certain of 12 months or lessour shares of Series A-1 Preferred Stock of NanoSynex in an amount that reduced our ownership in NanoSynex from approximately 49.97% to 39.90%. Our investment in NanoSynex will be accounted for underas an equity method investment prospectively from the existing guidance for operating leases today. Topic 842 supersedesJuly 20, 2023 deconsolidation date.
Other Service Agreements
We enter into contracts in the previous lease standard, Topic 840Leases. The guidance is effective for annual periodsnormal course of business, including with clinical sites, contract research organizations, and interim periods within those annual periods beginning after December 15, 2018, and is effectiveother professional service providers for the Companyconduct of clinical trials, contract manufacturers for the production of our product candidates, contract research service providers for preclinical research studies, professional consultants for expert advice and vendors for the sourcing of clinical and laboratory supplies and materials. These contracts generally provide for termination on notice, and therefore are cancelable contracts.
| 30 |
Cash Flows
The following table sets forth the significant sources and uses of cash for the periods set forth below:
| For the Twelve Months Ended | ||||||||
| December 31, | ||||||||
| 2023 | 2022 | |||||||
| Net cash (used in) provided by: | ||||||||
| Operating activities | $ | (10,304,263 | ) | $ | (13,247,541 | ) | ||
| Investing activities | 4,215,943 | (183,763 | ) | |||||
| Financing activities | (550,000 | ) | 2,910,515 | |||||
| Effect of exchange rate on cash | — | 22,639 | ||||||
| Net decrease in cash and restricted cash | $ | (6,638,320 | ) | $ | (10,498,150 | ) | ||
Net Cash Used in Operating Activities
During the year ended December 31, 2023, operating activities used $10.3 million of cash, primarily resulting from a loss from continuing operations of $12.5 million. Cash flows from operating activities for the year endingended December 31, 2019. The Company is currently evaluating the impact that the implementation2023 were positively impacted by adjustments for a $1.1 million non cash loss on voluntary conversion of this standard will haveconvertible debt, a $0.6 million non cash loss on the Company’s financial statements.
On March 30, 2016, the FASB issued Accounting Standards Update No. 2016-09,Compensation - Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting (“ASU 2016-09”). Among other things, ASU 2016-09 requires that entities recognize excess tax benefitsconvertible debt extinguishment, accretion of discount of $1.5 million on convertible debt, a $1.6 million increase in accounts payable, and deficiencies related to employee share-based payment transactions as income tax expense or benefit. ASU 2016-09 also eliminates the requirement to reclassify excess tax benefits and deficiencies$1.1 million in non cash stock-based compensation expense. Cash flows from operating activities for the year ended December 31, 2023 were negatively impacted by adjustments for a $2.0 million decrease in fair value of warrant liabilities, a $0.3 million increase in prepaid expenses and other assets, a $0.2 million decrease in accrued expenses and other current liabilities, and cash used in discontinued operations of $1.2 million.
During the year ended December 31, 2022, operating activities used $13.2 million of cash, primarily resulting from a loss from continuing operations of $13.9 million. Cash flows from operating activities for the year ended December 31, 2022 were positively impacted by an adjustment for $4.8 million in non cash stock-based compensation expense. Cash flows from operating activities for the year ended December 31, 2022 were negatively impacted by cash used in discontinued operations of $2.6 million, a $0.9 million decrease in fair value of warrant liabilities, a $0.5 million decrease in accrued expenses and other current liabilities, and a $0.1 million increase in prepaid expenses.
Net Cash Provided By Investing Activities
During the year ended December 31, 2023, net cash provided by investing activities was approximately $4.2 million resulting from discontinued operations due to $4.9 million in proceeds received from the sale of Qualigen, Inc., offset by $0.5 million advanced to NanoSynex, and $0.2 million in purchases of property and equipment prior to deconsolidation.
During the year ended December 31, 2022, net cash used in investing activities was approximately $0.2 million, due to capital expenditures offset by cash acquired in the NanoSynex acquisition.
Net Cash Provided by Financing Activities
Net cash provided by financing activities in the statement of cash flows. The guidance is effective for the annual periods and interim periods within those annual periods beginning after December 15, 2016. The adoption of this standard did not have a material impact on the Company’s financial statements.
On August 26, 2016, the FASB issued Accounting Standards Update No. 2016-15,Statement of Cash Flows (Topic 230),a consensus of the FASB’s Emerging Issues Task Force (“ASU 2016-15”). The new guidance amends Accounting Standards Codification No. 230 (“ASC 230”) to add or clarify guidance on the classification of certain cash receipts and payments in the statement of cash flows. ASC 230 lacks consistent principles for evaluating the classification of cash payments and receipts in the statement of cash flows. This has led to diversity in practice and, in certain circumstances, financial statement restatements. Therefore, the FASB issued the ASU 2016-15 with the intent of reducing diversity in practice with respect to eight types of cash flows. ASU 2016-15 is effective for annual and interim periods in fiscal years beginning after December 15, 2017, and is effective for the Company for the yearending ended December 31, 2018. The Company is currently evaluating2023, was approximately $0.6 million, due to monthly redemption payments which we made in the impact thatform of stock (rather than in the implementationform of this standard will havecash) on the Company’s financial statements.2022 Debenture.
In May 2017,
Net cash provided by financing activities for the FASB issued Accounting Standards Update No. 2017-09,Compensation - Stock Compensation (Topic 718): Scope of Modification Accounting(“ASU 2017-09”). ASU 2017-09 provides guidance on the types of changesyear ended December 31, 2022, was approximately $2.9 million, due to the terms or conditionsissuance of share-based payment awardsconvertible debt to which an entity would be required to apply modification accounting. An entity would not apply modification accounting if the fair value, vesting conditions, and classification of the awards are the same immediately before and after the modification. The amendments are effective for the Company’s interim and annual reporting periods beginning January 1, 2018. The Company does not expect the adoption of ASU 2017-09 to have a material impact on its financial statements.Alpha.
In July 2017, the FASB issued ASU 2017-11,Earnings Per Share (Topic 260); Distinguishing Liabilities from Equity (Topic 480); Derivatives and Hedging (Topic 815): (Part I) Accounting for Certain Financial Instruments with Down Round Features, (Part II) Replacement of the Indefinite Deferral for Mandatorily Redeemable Financial Instruments of Certain Nonpublic Entities and Certain Mandatorily Redeemable Noncontrolling Interests with a Scope Exception.The ASU allows companies to exclude a down round feature when determining whether a financial instrument (or embedded conversion feature) is considered indexed to the entity’s own stock. As a result, financial instruments (or embedded conversion features) with down round features may no longer be required to be accounted classified as liabilities. A company will recognize the value of a down round feature only when it is triggered and the strike price has been adjusted downward. For equity-classified freestanding financial instruments, such as warrants, an entity will treat the value of the effect of the down round, when triggered, as a dividend and a reduction of income available to common shareholders in computing basic earnings per share. For convertible instruments with embedded conversion features containing down round provisions, entities will recognize the value of the down round as a beneficial conversion discount to be amortized to earnings. The guidance in ASU 2017-11 is effective for fiscal years beginning after December 15, 2018, and interim periods within those fiscal years. Early adoption is permitted, and the guidance is to be applied using a full or modified retrospective approach. The Company adopted this guidance in the current quarter, effective October 1, 2017. As a result, the warrants issued on October 3, in connection with the October 2017 Public Offering, were equity-classified.
Item 7A. Quantitative and Qualitative Disclosures about Market RiskRisk.
Not applicable.
Item 8. Financial Statements and Supplementary Data.
The financial statements and the reports of our independent registered accounting firm required pursuant to this itemWe are included in Item 15 of this report and are presented beginning on page F-1.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
There has been no change of accountants nor any disagreements with accountants on any matter of accounting principles or practices or financial disclosure required to be reported under this Item.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our chief executive officer (our principal executive officer) and vice president finance (our principal financial officer), evaluated the effectiveness of our disclosures controls and procedures,a smaller reporting company as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act of 1934, as amended (the “Exchange Act”), as of December 31, 2017. The term “disclosure controls and procedures,” as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, means controls and other procedures of a company that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company’s management, including its principal executive and principal financial officers, as appropriate, to allow timely decisions regarding required disclosure.
Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2017, our principal executive officer and principal financial officer concluded that, as of such date, our disclosure controls and procedures were effective at a reasonable assurance level.
Management’s Annual Report on Internal Control Over Financial Reporting
Internal control over financial reporting refers to the process designed by, or under the supervision of, our chief executive officer and vice president finance, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Internal control over financial reporting includes those policies and procedures that: (1) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; (2) provide a reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made in accordance with authorizations of our management and directors; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitation. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgement and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management override. Because of such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk.
Management is responsible for establishing and maintaining adequate internal control over our financial reporting, as such term is defined in Rule 13a-15(f) under the Exchange Act. Under the supervision and with the participation of our management, including our chief executive officer and vice president finance, we conducted an evaluation of the effectiveness of our internal control over financial reporting. Management has used the framework set forth in the report entitled “Internal Control – Integrated Framework” published by the Committee of Sponsoring Organizations of the Treadway Commission to evaluate the effectiveness of our internal control over financial reporting. Based on its evaluation, management has concluded that our internal control over financial reporting was effective as of December 31, 2017, the end of our most recent fiscal year.
Changes in Internal Control over Financial Reporting
We regularly review our system of internal control over financial reporting and make changes to our processes and systems to improve controls and increase efficiency, while ensuring that we maintain an effective internal control environment. Changes may include such activities as implementing new, more efficient systems, consolidating activities, and migrating processes. There were no changes in our internal control over financial reporting that occurred during our last fiscal quarter that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
None.
Item 10. Directors, Executive Officers, and Corporate Governance
The Board of Directors
Our board of directors currently consists of eight members. Biographical information with respect to our directors is provided below.
Our directors hold office for one year or until their successors have been duly elected and qualified or until the earlier of their death, resignation or removal. Our amended and restated bylaws provide that the authorized number of directors comprising our board of directors will be fixed, from time to time, by a majority of the total number of directors.
There are no family relationships among any of our directors or executive officers, other than Ira and Andrew Ritter, who are father and son, respectively.
| Name | Position with the Company | Age as of the Annual Meeting | Director Since | ||||
| Michael D. Step | Chief Executive Officer and Director | 58 | 2012 | ||||
| Andrew J. Ritter | President and Director | 35 | 2008 | ||||
| Ira E. Ritter | Executive Chairman, Chief Strategic Officer and Director | 69 | 2008 | ||||
| Noah J. Doyle | Director | 50 | 2008 | ||||
| Matthew W. Foehr | Director | 45 | 2015 | ||||
| Paul V. Maier | Director | 70 | 2015 | ||||
| Dr. William M. Merino | Director | 75 | 2017 | ||||
| Gerald T. Proehl | Director | 59 | 2015 |
Michael D. Stepbecame our Chief Executive Officer on October 1, 2014. He has served as a director of the Company since 2012. Mr. Step has over 20 years of business development and corporate development experience in the pharmaceutical industry. Prior to joining the Company as its Chief Executive Officer, Mr. Step served as Senior Vice President of Corporate Development at Santarus, Inc. (“Santarus”), and a member of its executive committee, from 2005 to January 2014, when Santarus was sold to Salix Pharmaceuticals, Ltd. At Santarus, Mr. Step was responsible for corporate development activities. Prior to joining Santarus, he served as Vice President, Corporate Development for Amylin Pharmaceuticals, Inc. (“Amylin”) from 2000 to 2005. In this capacity, he was responsible for leading corporate development activities, including product licensing, strategic planning, and mergers and acquisitions evaluations. Before joining Amylin, Mr. Step served as Senior Director, Business Development at Dura Pharmaceuticals, Inc. (“Dura Pharmaceuticals”) from 1997 to 2000. In this position, his duties included licensing of marketed pharmaceutical products. Prior to joining Dura Pharmaceuticals, he served in corporate development and strategic planning at Hoffmann-La Roche, from 1996 to 1997, and held various sales and management roles at Roche Labs, from 1994 to 1996, and Syntex Labs, from 1992 to 1994. Mr. Step holds a B.A. in political science from Vanderbilt University and a M.B.A. from the University of Southern California.
Qualifications: We believe that Mr. Step is well qualified to serve on our board of directors and as Chief Executive Officer of the Company due to his over 20 years’ experience in the pharmaceutical industry, serving in senior leadership roles within public pharmaceutical companies including in the gastrointestinal disease segment. Mr. Step has served in various executive management positions in sales and sales management, and has had experience with many aspects of pharmaceutical commercialization, strategic planning, business development and licensing providing both strategic and operational vision and guidance. His extensive experience gives him valuable insight into our industry as well as seasoned business judgment.
Andrew J. Ritterserved as Co-Founder, President and Chief Executive Officer of the Company’s predecessor in interest from its inception in 2004 until relinquishing the role of Chief Executive Officer to Mr. Step in October 2014. Mr. Ritter was a member of the board of directors of the Company’s predecessor since its inception in 2004 and has been a member of our board of directors since 2008 when the Company was formed. Mr. Ritter has been actively studying the field of lactose intolerance for over 15 years and currently holds over a dozen patents and over twenty pending international patent applications. In addition, he has co-published articles, given presentations at major healthcare and medical conferences, and has been a guest lecturer of entrepreneurship at various graduate and undergraduate schools throughout Los Angeles including: University of Southern California Marshall School of Business, University of California at Los Angeles Anderson School of Business and Pepperdine University Graziadio School of Business and Management. Mr. Ritter served as a Los Angeles City Commissioner on the Commission for Children, Youth and Their Families from 2000 to 2002. He holds a B.A. in Political Science and a minor in Business from the University of Southern California. Mr. Ritter received a Master of Business Administration from the Wharton School of Business.
Qualifications: We believe that Mr. Ritter is well qualified to serve on our board of directors due to his over 15 years of research experience working in lactose intolerance and digestive diseases. Having founded the Company and invented Lactagen™, Mr. Ritter has an in depth knowledge of the Company, and provides senior leadership on the clinical and product development matters facing the Company. Mr. Ritter also brings to the board of directors an extensive scientific and operational background gained previously at Ritter Natural Sciences and over the years at Ritter.
Ira E. Ritter served as Co-Founder, Chief Strategic Officer and Executive Chairman of the Company’s predecessor in interest from its inception in 2004 through the formation of the Company in 2008 and has served in those positions with the Company since 2008. Mr. Ritter has extensive experience creating and building diverse business enterprises and has provided corporate management, strategic planning and financial consulting for a wide range of market segments. Since 2010, Mr. Ritter has also acted as a managing partner of Stonehenge Partners. Mr. Ritter served as President and Vice Chairman of Quality King, Inc., a national wholesale distributor of healthcare products, from 1992 to 2000. From 1998 to 2001, he served as President and Chairman of Rockwood Investments Inc., a business he developed which produced private label health and beauty products for major national retailers, including GNC and K-Mart. He also served as Chairman of ON-TV, a division of Oak Industries, Inc., from 1982 to 1985, where he managed the television division initiating exclusive broadcasts of Los Angeles, Chicago, and New York professional baseball, basketball, and hockey games. During this tenure, he produced the first televised home shopping program and directed development of the largest “pay-per-view” channel system for its time. Mr. Ritter served on the board of directors for Martin Lawrence Art Galleries from 1980 to 1985 helping take it public on The New York Stock Exchange. During his 20 years as a publisher, he produced monthly national consumer magazines focused on health & fitness, women’s issues and the environment. Mr. Ritter also has a long history of public service that includes appointments by three Governors to several State of California Commissions including eight years served as Commissioner on the California Prison Industry Authority. He has guest lectured at University of Southern California Marshall School of Business and Pepperdine University Graziadio School of Business where he also serves as an advisory board member to Pepperdine’s Graduate School of Education and Psychology, Social Entrepreneurship and Change Program. Presently he serves on the board of directors for Vitavis Laboratories. In 1981, Mr. Ritter was honored with the City of Hope’s Man of the Year award.
Qualifications: We believe that Mr. Ritter is well suited to serve on our board of directors due to his over 40 years’ experience overseeing daily operations of diverse business enterprises, and his managing public as well as private companies. Mr. Ritter provides our board of directors with extensive background in operational and strategic planning, as well as general executive and leadership expertise. Mr. Ritter has served on the boards of several companies during his career.
Noah J. Doyle has served as a director of the Company since September 2008. He has been an entrepreneur and investor for over 20 years. Mr. Doyle is the managing director of Javelin GP, LLC, the general partner of Javelin GP, LP, which is the general partner of Javelin and the manager of Javelin SPV. Prior to forming the first Javelin entities in 2008, Mr. Doyle supported over a dozen start-ups as an angel investor, including Keyhole, Inc. (“Keyhole”) (acquired by Google Inc. in 2004), Cantametrix, Inc. (acquired by Gracenote, Inc. in 2002), Amae Software (acquired by Verint Systems, Inc. in 2006), Nuvon, Inc., Aquea Scientific Corporation, Emdigo Inc., Magnacash Inc. (acquired by Yaga, Inc. in 2001), and i-mint India. Mr. Doyle most recently directed the enterprise product line for Google’s geospatial products, Google Earth and Google Maps, from 2004 to 2007. From 2002 to 2004 he managed the Sales and Corporate Development functions at Keyhole, which created the first Web hosted digital earth model. Prior to Keyhole, Mr. Doyle helped establish the Internet loyalty rewards marketplace as a co-founder of MyPoints.com (“MyPoints”), the largest Internet loyalty program with over 6 million active members, where he led product management and business development functions from the company’s inception in 1996 through its initial public offering and subsequent acquisition by United Airlines in 2002. Prior to joining MyPoints, Mr. Doyle was based in Tokyo where he managed overseas sales and marketing for the OEM channel of Matsushita’s (Panasonic) communications equipment subsidiary in Japan, from 1990 to 1994. Mr. Doyle served on the board of directors of MOL Global, Inc. from July 2014 to February 2016. He was also chairman of the management board of the University of California, Berkeley’s campus bookstore, a $17 million retail operation, and also held product management and operations management roles at IBM/Rational (Pure Atria) and Oracle, from 1989 to 1990. Mr. Doyle holds M.B.A. and B.A. Economics degrees, as well as certificates in Management of Technology and Global Management from University of California — Berkeley.
Qualifications: We believe that Mr. Doyle is well suited to serve on our board of directors due to his over 20 years of experience as an entrepreneur and investor. Mr. Doyle has experience as a venture capitalist building and serving on the boards of many public and private emerging companies in leadership roles providing guidance on finance, development and operational growth.
Matthew W. Foehr has served as a director of the Company since February 2015. He currently serves as President and Chief Operating Officer at Ligand Pharmaceuticals Incorporated (“Ligand”), a commercial stage biopharmaceutical company. Prior to joining Ligand in 2011, Mr. Foehr was Vice President and Head of Consumer Dermatology R&D, as well as Acting Chief Scientific Officer of Dermatology, in the Stiefel division of GlaxoSmithKline (“GSK”). Following GSK’s acquisition of Stiefel Laboratories, Inc. (“Stiefel”) in 2009, Mr. Foehr led the R&D integration of Stiefel into GSK. At Stiefel Laboratories, Inc., Mr. Foehr served as Senior Vice President of Global R&D Operations, Senior Vice President of Product Development& Support, and Vice President of Global Supply Chain Technical Services. Prior to joining Stiefel, Mr. Foehr held various executive roles at Connetics Corporation including Senior Vice President of Technical Operations and Vice President of Manufacturing. Currently, he is a member of the board of directors of Viking Therapeutics Inc. Mr. Foehr is the author of multiple scientific publications and is a named inventor on numerous U.S. patents. He received his Bachelor of Science degree in Biology from Santa Clara University.
Qualifications: We believe that Mr. Foehr is well suited to serve on our board of directors due to his more than 20 years of experience in the pharmaceutical industry and his experience managing global operations and research and development programs.
Paul V. Maier has served as a director of the Company since April 2015. From November 2009 through June 2014, Mr. Maier served as the Chief Financial Officer of Sequenom Inc., a publicly held company serving the discovery, clinical research, and diagnostics market. From February 2007 until November 2009, he served as an independent financial consultant. Previously, Mr. Maier was Senior Vice President and Chief Financial Officer of Ligand from 1992 through 2007. From 1990 to 1992, Mr. Maier served as Vice President, Finance of DFS West, a division of DFS Group LP, a private multinational retailer. From 1984 to 1990, Mr. Maier was employed by ICN Pharmaceuticals, a pharmaceutical and biotechnology research products company, where he held various executive positions in finance and general management in ICN as well as SPI Pharmaceuticals, a publicly held subsidiary. Mr. Maier currently serves on the board of directors of International Stem Cell Corporation, Apricus Biosciences, MabVax Therapeutics, and Biological Dynamics. Mr. Maier received an MBA from Harvard Business School and a BS from Pennsylvania State University.
Qualifications: We believe that Mr. Maier is well suited to serve on our board of directors due to his over 25 years of experience as a senior executive in biotechnology and pharmaceutical companies and his extensive experience in finance.
Dr. William M. Merino has served as a director of the Company since January 17, 2017. Dr. Merino served as the Senior Vice President of Worldwide Regulatory Affairs for Warner Lambert Pharmaceuticals from 1987 to 2000, where he was a member of the Office of the Chairman and responsible for the registration and approval of pharmaceuticals products with regulatory agencies around the world. He was also responsible for quality assurance, quality control and drug safety for the company, and led efforts to gain expedited registration of Lipitor in the United States and abroad in 20 other countries. He also has previous experience leading international regulatory affairs at Alcon Pharmaceuticals, G.D. Searle & Co., and Riker Laboratories. Dr. Merino has served as a senior clinical and regulatory advisory to the Company. Dr. Merino received his PhD in Pharmacology from Purdue University.
Qualifications: We believe that Dr. Merino’s deep global experience in drug and device registration and his extensive work with senior members of the FDA as well as several international regulatory authorities will bring important insight and acumen to our board of directors, as the Company continues its interactions with the FDA in an effort to bring RP-G28 to market.
Gerald T. Proehl has served as a director of the Company since April 2015. Currently, Mr. Proehl is President, CEO, Founder and Director of Dermata Therapeutics, LLC, a private biopharmaceutical company. From January 2002 to January 2014, he was the President, Chief Executive Officer and a Director of Santarus, a company that he helped to found in 1999 and sold to Salix Pharmaceuticals in January 2014 for $2.6 billion. From March 2000 through December 2001, Mr. Proehl was President and Chief Operating Officer of Santarus, and from April 1999 to March 2000, Mr. Proehl was Vice President, Marketing and Business Development of Santarus. Prior to joining Santarus, Mr. Proehl was with Hoechst Marion Roussel, Inc. (“Hoechst”), a global pharmaceutical company, for 14 years, where he served in various capacities, including Vice President of Global Marketing. During his career at Hoechst he worked across numerous therapeutic areas, including CNS, cardiovascular, and gastrointestinal. Mr. Proehl currently serves on the board of directors of two other public company boards, Sophiris Bio Inc. and Tenax Therapeutics, Inc. Mr. Proehl also serves on a number of private company boards including Kinetek Sports, Patara Pharma LLC, MDRejuvena, Inc. and Dermata Therapeutics, LLC. He also served on the board of directors of Auspex Pharmaceuticals, Inc. from January 2014 to May 2015. Mr. Proehl holds a B.S. in education from the State University of New York at Cortland, an M.A. in exercise physiology from Wake Forest University and an M.B.A. from Rockhurst College.
Qualifications: We believe that Mr. Proehl is well suited to serve on our board of directors due to his general business and commercial experience in the pharmaceutical industry, as well as his strong background in business operations developed through his leadership at other companies.
Executive Officers
Our Executive Officers as of the date of this Annual Report are as follows:
Officers serve at the discretion of the board of directors. There are no family relationships among any of our directors or executive officers, other than Ira and Andrew Ritter, who are father and son, respectively. There is no arrangement or understanding between any executive officer and any other person pursuant to which the executive officer was selected.
Michael D. Step. For Mr. Step’s biography, please see above under “Board of Directors.”
Andrew J. Ritter. For Mr. Ritter’s biography, please see above under “Board of Directors.”
Ira E. Ritter. For Mr. Ritter’s biography, please see above under “Board of Directors.”
Jeffrey Benjamin has served as our Vice President, Finance since October 2017. Mr. Benjamin previously served as Interim Corporate Controller for Unified Grocers (acquired by Supervalu Inc. in June 2017) since February 2017. Prior to that he served as a principal consultant for Tatum Consulting from January 2015 to January 2017, Chief Financial Officer of Communications Infrastructure Corporation from March 2013 to January 2015, and Corporate Controller of Liaison Technologies, Inc. from April 2012 to March 2013.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Securities Exchange Act, and the rules issued thereunder, requires our directors and executive officers and beneficial owners of more than 10% of the outstanding shares of our equity securities to file reports of ownership and changes in beneficial ownership of our equity securities with the SEC. Copies of these reports are furnished to us. We are required to identify any of those individuals who failed to file such reports on a timely basis. Based solely on our review of the copies of such reports furnished to us, and representations from the persons subject to Section 16(a) with respect to the Company, we believe that during 2017 all of our executive officers, directors and 10% stockholders complied with the Section 16(a) requirements.
Code of Business Conduct and Ethics
We adopted a code of business conduct and ethics that applies to all of our employees, officers and directors, including those officers responsible for financial reporting prior to the closing of our initial public offering. The code of business conduct and ethics is available on our website atwww.ritterpharmaceuticals.com. Any amendments to the code of business conduct and ethics, or any waivers of its requirements that apply to our principal executive officer, principal financial officer or principal accounting officer, will be disclosed on our website.
Audit Committee
The current members of our audit committee are Matthew W. Foehr, Paul V. Maier and Gerald T. Proehl, with Mr. Maier serving as chairman. Our board of directors has determined that each member of our Audit Committee is independent under Rule 10A-312b-2 of the Exchange Act and the continued listing requirements of NASDAQ, and that each member of our Audit Committee satisfies the other continued listing requirements of NASDAQ for audit committee membership. Our board of directors has also determined that Mr. Maier qualifies as an “audit committee financial expert,” as such term is defined by the SEC, and that he has the requisite level of financial sophistication required by the continued listing requirements of NASDAQ.
Item 11. Executive Compensation
Summary Compensation Table (2017 and 2016)
The following table sets forth the compensation paid or earned for the fiscal years ended December 31, 2017 and 2016 to our named executive officers for each of those years, who are comprised of (1) our principal executive officer for such year, and (2) our next two highest compensated executive officers other than the principal executive officer (whose compensation exceeded $100,000).
| Name and Principal Position | Year | Salary ($) | Bonus ($)(2) | Option Awards(1) ($) | Nonequity Incentive Compensation ($) | All Other Compensation ($) | Total ($) | |||||||||||||||||||||
| Michael D. Step | 2017 | $ | 411,875 | $ | — | $ | 88,425 | — | $ | — | $ | 500,300 | ||||||||||||||||
| Chief Executive Officer | 2016 | $ | 376,269 | $ | — | $ | 126,280 | — | $ | — | $ | 502,549 | ||||||||||||||||
| Andrew J. Ritter | 2017 | $ | 331,569 | $ | 121,320 | $ | — | — | $ | — | $ | 452,889 | ||||||||||||||||
| President | 2016 | $ | 324,010 | $ | 117,180 | $ | 490,394 | — | $ | — | $ | 931,584 | ||||||||||||||||
| Ira E. Ritter | 2017 | $ | 330,367 | $ | 101,115 | $ | — | — | $ | — | $ | 431,482 | ||||||||||||||||
| Executive Chairman and | 2016 | $ | 308,332 | $ | 97,571 | $ | 490,394 | — | $ | — | $ | 896,297 | ||||||||||||||||
| Chief Strategic Officer | ||||||||||||||||||||||||||||
In addition to the assumptions used in the Black-Scholes option-pricing model, we also estimate a forfeiture rate to calculate the stock-based compensation for our equity awards. We will continue to use judgment in evaluating the expected volatility, expected terms and forfeiture rates utilized for our stock-based compensation calculations on a prospective basis.
Narrative to Summary Compensation Table
Letter Agreement with Michael D. Step
On December 2, 2014, we entered into a letter agreement (the “Step Letter Agreement”), with Mr. Step, our current Chief Executive Officer, setting forth the terms of his employment. The Step Letter Agreement provides that Mr. Step will be entitled to an annual base salary of $360,000. Pursuant to the Step Letter Agreement, Mr. Step was also entitled to receive three stock options.
The first two options entitle Mr. Step to purchase 646,537 and 73,377 shares of Common Stock of the Company, respectively, for an exercise price of $5.86 per share. Each of these options was immediately exercisable in full as of the date of the grant, with 44/48ths of the total number of shares covered by each option subject to a right of repurchase by the Company upon termination of Mr. Step’s employment with us for any reason. This right of repurchase will lapse over a period of 44 months, with 1/44th of the total number of shares subject to the right of repurchase lapsing on January 1, 2015 and on the first day of each month thereafter. In addition, the right of repurchase will lapse in its entirety upon a termination of the employment of Mr. Step by us without Cause or by Mr. Step with Good Reason and upon a Termination upon a Change in Control.
The third option became exercisable upon the closing of our initial public offering on June 29, 2015. Pursuant to the terms of the agreement, the option is exercisable for a total of 163,799 shares of our Common Stock, which, together with the shares subject to the first option, represent 7.5% of the shares of Common Stock deemed to be outstanding at June 29, 2015 on a fully-diluted basis, after giving effect to the number of shares subject to the third option. Seventy-five percent of the shares subject to the third option are subject to a right of repurchase by us upon termination of Mr. Step’s employment for any reason. This right of repurchase will lapse with respect to 1/36th of the total number of shares subject to the right of repurchase on the first day of each month following the date on which the third option first becomes exercisable. In addition, the right of repurchase will lapse in its entirety upon Mr. Step’s termination of employment under certain circumstances.
For purposes of the Step Letter Agreement, the terms “Cause,” “Good Reason,” and “Termination upon a Change in Control” each have the meanings ascribed to such terms in the Executive Severance & Change in Control Agreement described below.
Offer Letters with Andrew Ritter and Ira Ritter
The compensation terms outlined in the offer letters, which became effective June 29, 2015, superseded and replaced those provided in the Executive Compensation Plan, which is described above, other than certain provisions related to the bonus opportunities. The offer letters provide that Andrew Ritter is entitled to an annual base salary of $310,000 and Ira Ritter is entitled to an annual base salary of $295,000. In accordance with his offer letter, Andrew Ritter also became entitled to receive up to $180,000 payable over a three-year period for tuition reimbursement.
Pursuant to their respective offer letters, Andrew Ritter and Ira Ritter each have the opportunity to earn an annual bonus based upon a percentage of their base salary and the achievement of specific performance as determined by the Company. The initial target bonus opportunities are 40% and 35% of the base salary for Andrew Ritter and Ira Ritter, respectively.
2015 Equity Incentive Plan
On June 15, 2015, our board of directors approved the 2015 Equity Incentive Plan, and on June 17, 2015, the 2015 Equity Incentive Plan was approved by our stockholders. The 2015 Equity Incentive Plan was subsequently amended by the stockholders of the Company on June 3, 2016 and June 2, 2017.
The purposes of the 2015 Equity Incentive Plan are to optimize the profitability and growth of the Company through long-term incentives that are consistent with the Company’s objectives and that link the interests of award recipients (“Grantees”), to those of the Company’s stockholders; to give award recipients an incentive for excellence in individual performance; to promote teamwork among Grantees; and to give the Company flexibility in attracting and retaining key employees, directors and consultants.
Selected employees, officers and directors of the Company or any subsidiary, and consultants, advisors and independent service providers to the Company and any subsidiary who qualify as a “consultant” under the applicable rules of the SEC for registration of shares on a Form S-8 registration statement, are eligible to receive awards under the 2015 Equity Incentive Plan. The plan administrator may also grant awards to individuals in connection with hiring, retention or otherwise before the date the individual first performs services for the Company or any subsidiary; provided, however, that those awards will not become vested or exercisable before the date the individual first performs services for the Company or any subsidiary.
The number of shares of Common Stock that we may issue pursuant to awards under the 2015 Plan is (i) 27,500,000 plus (ii) any shares which were available for grant under the 2008 Stock Plan or the 2009 Stock Plan (collectively, the “Prior Plans”), on the effective date of the 2015 Equity Incentive Plan or are subject to awards under the Prior Plans which, after the effective date of the 2015 Equity Incentive Plan, are forfeited or lapse unexercised or are settled in cash and are not issued under the Prior Plans. No more than 27,500,000 shares of Common Stock may be issued pursuant to incentive stock options intended to qualify under Section 422 of the Internal Revenue Code (the “Code”). No awards may be granted under any Prior Plan; however, any awards granted under any Prior Plan that were outstanding as of the effective date of the 2015 Plan continue to be subject to the terms and conditions of such Prior Plan. The maximum number of shares of Common Stock subject to awards of any combination that may be granted under the 2015 Equity Incentive Plan during any calendar year to any one individual is limited to 3,000,000 shares.
These limits will be appropriately adjusted to reflect any stock dividend, stock split, combination or exchange of shares, merger, consolidation or other distribution and/or similar transactions. To the extent that (i) an award terminates, expires, lapses or is forfeited for any reason, (ii) any award is settled in cash (in whole or in part) without the delivery of shares to the Grantee, or (iii) any shares subject to an award under any Prior Plan terminate, expire, lapse or are forfeited for any reason or an award under any Prior Plan is settled for cash (in whole or in part), then any shares subject to the award, to the extent of such termination, expiration, lapse, forfeiture or cash settlement, will again be available for the grant of an award pursuant to the 2015 Equity Incentive Plan. Any shares tendered or withheld to satisfy the grant or exercise prior or tax withholding obligation pursuant to any award will again be available for the grant of an award pursuant to the 2015 Equity Incentive Plan.
The 2015 Equity Incentive Plan may be administered by a committee or subcommittee of the board of directors as the board of directors may appoint from time to time, or by our full board of directors if no committee is designated or for other specific purposes. At present, the 2015 Equity Incentive Plan is administered by our compensation committee. The plan administrator has the full authority and discretion to administer the 2015 Equity Incentive Plan and to take any action that is necessary or advisable in connection with the administration of the 2015 Equity Incentive Plan, including without limitation the authority and discretion to interpret and administer the 2015 Equity Incentive Plan and any award agreement relating to the 2015 Equity Incentive Plan or any award made thereunder, the authority to designate Grantees to receive awards under the 2015 Equity Incentive Plan and to determine the type or types of awards to be granted to such Grantees, the authority to determine the terms and conditions of awards granted under the 2015 Equity Incentive Plan, and the authority to determine whether, to what extent, and pursuant to what circumstances and award may be settled in, or the exercise price of an award may be paid in, cash, shares, other awards, or other property, or an award may be canceled, forfeited or surrendered. The plan administrator’s determinations will be final and conclusive. The plan administrator may delegate certain of its authority to others as specified in the 2015 Equity Incentive Plan.
The 2015 Equity Incentive Plan provides for grants of stock options (including incentive stock options qualifying under Section 422 of the Code and nonstatutory stock options), restricted stock awards, stock appreciation rights, restricted stock units, performance awards, other stock-based awards or any combination of the foregoing.
Stock options. The 2015 Equity Incentive Plan allows the plan administrator to grant incentive stock options, as that term is defined in Section 422 of the Code, or nonqualified stock options. No incentive stock option award may be granted to any person who is not an employee of the Company or any subsidiary. Options must have an exercise price at least equal to the fair market value of the underlying shares on the date of grant. In addition, in the case of incentive stock options granted to a greater than 10% stockholder of the Company, such exercise price may not be less than 110% of the fair market value of the underlying shares on the date of grant. The option holder may pay the exercise price in cash or by check, by tendering shares of Common Stock (including shares issuable in settlement of the award), payment through a broker or by any other means that the plan administrator approves. Options granted under the 2015 Equity Incentive Plan will have a term of no more than 10 years, or five years in the case of incentive stock options granted to a greater than 10% stockholder of the Company; however, the options will expire earlier if the option holder’s service relationship with us terminates or as otherwise provided in an award agreement.
Restricted stock awards. The 2015 Equity Incentive Plan allows the plan administrator to grant restricted stock awards, which issue to the holder a certain number of shares of Common Stock that are subject to restrictions or conditions as the plan administrator deems appropriate, such as time-based or performance-based criteria, and which become vested upon the lapse or satisfaction of such conditions. The plan administrator may apply limitations to any restricted stock award and establish the purchase price (or provide for no purchase price), provided that if a purchase price is established, it may not be less than par value of the shares to be purchased.
Stock appreciation rights. The 2015 Equity Incentive Plan allows the plan administrator to grant awards of stock appreciation rights, which entitle the holder to receive a payment in cash, in shares of Common Stock, or in a combination of both, having an aggregate value equal to the spread on the date of exercise between the fair market value of the underlying shares on that date and the base price of the shares specified in the grant agreement, multiplied by the number of shares specified in the award being exercised and as otherwise provided in an award agreement. Stock appreciation rights may not have a base price of less than 100% of the fair market value of the underlying shares on the date of grant.
Restricted stock units. The 2015 Equity Incentive Plan allows the plan administrator to grant awards of restricted stock units (“RSUs”), which entitle the holder to a number of shares of Common Stock, a cash payment or some combination thereof, upon satisfaction of vesting and other criteria for issuance or upon such later date as specified in the award agreement, as established by the plan administrator in the award agreement.
Other stock-based awards. The 2015 Equity Incentive Plan allows the plan administrator to grant other stock-based stock awards to eligible participants, including dividend equivalent rights, stock payments and/or deferred stock. A dividend equivalent may be granted alone or in conjunction with another type of award, and generally provides for payment, in cash, Common Stock or some combination thereof, of an amount equal to the dividends that would have been payable with respect to a specified number of underlying shares. A stock payment is an award to a Grantee, only upon satisfaction of performance-based criteria or other criteria specified by the plan administrator, of a specified number of shares of Common Stock, or an option to purchase Common Stock, which may be (but is not required to be) in lieu of base salary, bonus, fees or other cash consideration toprovide the Grantee. A deferred stock award is a grant to a Grantee, only upon satisfaction of performance-based criteria or other criteria specified by the plan administrator, of a specified number of shares of Common Stock.information otherwise required under this Item.
Performance awards. The 2015 Equity Incentive Plan allows the plan administrator to grant performance awards which become payable in Common Stock, in cash or in a combination of Common StockItem 8. Consolidated Financial Statements and cash, on account of attainment of one or more performance goals established by the plan administrator on one or more specified dates or over a specified period or periods. The plan administrator may establish performance goals relating to any of the following: (i) gross or net earnings (either before or after one or more of the following: interest, taxes, depreciation and amortization); (ii) gross or net sales or revenue; (iii) gross or net income or adjusted income (either before or after taxes); (iv) operating earnings or profit; (v) cash flow (including, but not limited to, operating cash flow and free cash flow); (vi) return on assets; (vii) return on capital; (viii) return on stockholders’ equity; (ix) return on sales; (x) gross or net profit or operating margin; (xi) costs; (xii) funds from operations; (xiii) expenses; (xiv) working capital; (xv) earnings per share or adjusted earnings per share; (xvi) price per share of Common Stock; (xvii) regulatory body approval for commercialization of a product; (xviii) implementation or completion of critical projects; (xix) market share; or (xx) total stockholder return; any of which may be measured either in absolute terms or as compared to any incremental increase or decrease or as compared to results of a peer group or to market performance indicators or indices.Supplementary Data
The plan administrator may, in its sole discretion, provide that one or more objectively determinable adjustments will be made to one or more of the performance goals described above, such as adjustments to account for changes in the Company’s or segment’s business (e.g., restructuring, acquisition or disposal or discontinuance of a business segment), accounting or financial reporting (e.g., change in accounting principles, significant income or expense or amortization of assets) or for other unusual or non-recurring events, all as further detailed in the 2015 Equity Incentive Plan. For all awards intended to qualify as performance-based compensation, such determinations shall be made within the time periods prescribed by, and otherwise in compliance with, Section 162(m) of the Code.INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Amendment and termination. No award will be granted under the 2015 Equity Incentive Plan after the tenth anniversary of the effective date of the 2015 Equity Incentive Plan. Subject to applicable laws and exchange limitations, our board of directors or the plan administrator may terminate, amend or modify the 2015 Equity Incentive Plan, or any portion thereof, at any time. Stockholder approval will be required to (i) increase the limits imposed on the maximum number of shares which may be issued under the 2015 Equity Incentive Plan or as incentive stock options (other than an appropriate adjustment due to stock dividend, stock split, combination or exchange of shares, merger, consolidation or similar circumstance), (ii) reduce the price per share of any outstanding option or stock appreciation right or cancel any such award in exchange for cash when the exercise price per share exceeds the fair market value of the underlying shares, or (iii) materially change the class of persons who are eligible to participate in the 2015 Equity Incentive Plan; provided, however, that no amendment, suspension or termination of the 2015 Equity Incentive Plan may, without the consent of the Grantee, materially impair any rights or obligations under any award granted or awarded thereunder, unless the award itself otherwise expressly so provides.
Outstanding Equity Awards at 2017 Fiscal Year-End
The following table presents the outstanding equity awards held by each of the named executive officers as of December 31, 2017.
| Name | Number of Securities Underlying Unexercised Options Exercisable | Number of Securities Underlying Unexercised Options Unexercisable | Option Exercise Price ($) | Option Expiration Date | ||||||||||
| Michael D. Step | 26,163 | — | $ | 1.14 | 8/16/2022 | |||||||||
| 646,537 | (1) | — | $ | 5.86 | 12/2/2024 | |||||||||
| 73,377 | (2) | — | $ | 5.86 | 12/2/2024 | |||||||||
| 163,799 | (3) | — | $ | 5.86 | 12/2/2024 | |||||||||
| 29,042 | (4) | 52,958 | (4) | $ | 1.54 | 7/5/2026 | ||||||||
| 13,292 | (5) | 44,708 | (5) | $ | 2.89 | 1/25/2027 | ||||||||
| Andrew J. Ritter | 24,878 | (6) | 3,094 | (6) | $ | 1.27 | 9/25/2023 | |||||||
| 20,979 | — | $ | 5.86 | 12/2/2024 | ||||||||||
| 351,354 | (7) | 81,081 | (7) | (7) | 12/2/2024 | |||||||||
| 29,042 | (8) | 52,958 | (8) | $ | 1.54 | 7/5/2026 | ||||||||
| 23,341 | (9) | 116,703 | (9) | $ | 2.60 | 10/25/2026 | ||||||||
| Ira E. Ritter | 24,878 | (10) | 3,094 | (10) | $ | 1.27 | 9/25/2023 | |||||||
| 20,979 | — | $ | 5.86 | 12/2/2024 | ||||||||||
| 351,354 | (11) | 81,081 | (11) | (11) | 12/2/2024 | |||||||||
| 29,042 | (11) | 52,958 | (11) | $ | 1.54 | 7/5/2026 | ||||||||
| 23,341 | (12) | 116,703 | (12) | $ | 2.60 | 10/25/2026 | ||||||||
| Page | ||
|
|
Payments Due Upon Termination of Employment or a Change in Control
Executive Severance & Change in Control Agreements
We have entered into Executive Severance & Change in Control Agreements (the “Severance Agreements”), with each of our named executive officers. The Severance Agreements provide that if we terminate the executive’s employment without Cause, or the executive terminates his employment for Good Reason, the executive will be entitled to: (i) the Accrued Obligations; (ii) an amount equal to twelve (12) months of base salary, as in effect immediately prior to the termination date; (iii) medical, dental benefits provided by the Company to the executive and his spouse and dependents at least equal to the levels of benefits provided to other similarly situated active employees of the Company and its subsidiaries until the earlier of (a) the twelve (12) month anniversary of the date of termination or (b) the date that the executive becomes covered under a subsequent employer’s medical and dental plans; and (iv) acceleration of vesting of all equity and equity-based awards.
Pursuant to the terms of the Severance Agreements, in the event that within one (1) month prior to or the twelve (12) months following a Change in Control, the Company terminates the executive’s employment without Cause, or the executive terminates his employment for Good Reason, then, in lieu of the payments and benefits otherwise due to the executive in the preceding paragraph, the executive will be entitled to: (i) the Accrued Obligations; (ii) an amount equal to the sum of twelve (12) months of base salary, as in effect on the date of termination or the date of the Change in Control, whichever is greater; (iii) medical, dental benefits provided by the Company to the executive and his spouse and dependents at least equal to the level of benefits provided to other similarly situated active employees of the Company and its subsidiaries until the earlier of (a) the twelve (12) month anniversary of the date of termination or (b) the date that the executive becomes covered under a subsequent employer’s medical and dental plans; and (iv) acceleration of vesting of all equity and equity-based awards.
In the event the executive’s employment is terminated by him without Good Reason, by the Company for Cause or due to the executive’s death or disability, the executive and/or his estate or beneficiaries will be solely entitled to the Accrued Obligations.
The executive’s entitlement to the payments (other than the Accrued Obligations) and benefits described above is expressly contingent upon him providing the Company with a signed release satisfactory to the Company.
For purposes of the Severance Agreements:
“Accrued Obligations” means (i) earned but unpaid base salary through the date of termination; (ii) payment of any annual, long-term, or other incentive award which relates to a completed fiscal year or performance period, as applicable, and is payable (but not yet paid) on or before the date of termination; (iii) a lump-sum payment in respect of accrued but unused vacation days at the executive’s per-business-day base salary rate in effect as of the date of termination; and (iv) any unpaid expense or reimbursements due pursuant to Company expense reimbursement policy.
“Cause” means a finding by the Company that the executive has (i) been convicted of a felony or crime involving moral turpitude; (ii) disclosed trade secrets or confidential information of the Company (or any parent or subsidiary) to persons not entitled to receive such information; (iii) engaged in conduct in connection with the executive’s employment or service to the Company (or any parent or subsidiary), that has, or could reasonably be expected to result in, material injury to the business or reputation of the Company (or any parent or subsidiary), including, without limitation, act(s) of fraud, embezzlement, misappropriation and breach of fiduciary duty; (iv) violated the operating and ethics policies of the Company (or any parent or subsidiary) in any material way, including, but not limited to those relating to sexual harassment and the disclosure or misuse of confidential information; (v) engaged in willful and continued negligence in the performance of the duties assigned to the executive by the Company, after the executive has received notice of and failed to cure such negligence; or (vi) breached any material provision of any agreement between the executive and the Company (or any parent or subsidiary), including, without limitation, any confidentiality agreement.
“Change in Control” means the occurrence of any of the following events:
“Good Reason” means, without the executive’s express written consent, the occurrence of any one or more of the following: (i) a substantial and material diminution in the executive’s duties or responsibilities; (ii) a material reduction in the executive’s Base Salary; or (iii) the relocation of the executive’s principal place of employment to a location more than 50 miles from the executive’ principal work location to a location that is more than 50 miles from the prior location. Notwithstanding the foregoing, a relocation of Mr. Step’s principal place of employment to a location closer to Mr. Step’s principal residence in San Diego, California shall not constitute “Good Reason.” A termination of employment by the executive for Good Reason will be effectuated by giving the Company written notice, or Notice of Termination for Good Reason, not later than 90 days following the occurrence of the circumstance that constitutes Good Reason, setting forth in reasonable detail the specific conduct of the Company that constitutes Good Reason and the specific provision(s) of this Agreement on which the executive relied. The Company will be entitled, during the 30-day period following receipt of a Notice of Termination for Good Reason, to cure the circumstances that gave rise to Good Reason, provided that the Company shall be entitled to waive its right to cure or reduce the cure period by delivery of written notice to that effect to the executive (such 30-day or shorter period, the “Cure Period”). If, during the Cure Period, such circumstance is remedied, the executive will not be permitted to terminate his employment for Good Reason as a result of such circumstance. If, at the end of the Cure Period, the circumstance that constitutes Good Reason has not been remedied, the executive will terminate employment for Good Reason on the date of expiration of the Cure Period.
2009 Stock Plan
The 2009 Stock Plan provides that in the event we merge with or into another corporation, or a Change in Control (as defined below) occurs, each outstanding option and stock purchase right will be assumed or an equivalent option substituted by the successor corporation or a parent or subsidiary of the successor corporation. In the event that the successor corporation in a merger or Change in Control refuses to assume or substitute for the option or stock purchase right, then the optionee will fully vest in and have the right to exercise the option or stock purchase right as to all of the optioned stock, including shares as to which it would not otherwise be vested or exercisable; provided, however, that such exercise will only be permitted as and to the extent it complies with Code Section 409A or does not cause the option or stock purchase right to cease to be exempt from that statute.
For purposes of the 2009 Stock Plan, “Change in Control” means the occurrence of any of the following events:
2015 Equity Incentive Plan
The 2015 Equity Incentive Plan provides that notwithstanding any other provision of the 2015 Equity Incentive Plan, in the event of a Change in Control (as defined below), unless otherwise determined by the plan administrator, each outstanding award under the plan will be assumed or an equivalent award substituted by the successor corporation or a parent or subsidiary of the successor corporation. In the event that, or to the extent that, the successor corporation in a Change in Control refuses to assume or substitute for the award, or if the plan administrator determines that such assumption or substitution is not desirable or is only desirable for a portion of any outstanding award, then the plan administrator may take any or all of the following actions: (i) determine that an outstanding award will accelerate and become exercisable, or determine that the restrictions and conditions on an outstanding award will lapse, in whole or in part, as applicable, upon the Change of Control or upon such other event as the plan administrator determines; (ii) require that a Grantee surrender his or her outstanding award, or any portion of such outstanding award, in exchange for a payment by the Company, in cash or stock, as determined by the plan administrator, in an amount equal to the fair market value of the vested portion of the award (with respect to options or stock appreciation rights, or other similar appreciation value awards, such value shall be determined by the amount by which the then fair market value of the shares subject to the Grantee’s unexercised award exceeds the any applicable exercise price or other grant price or base value or the award); or (iii) after giving the Grantee an opportunity to exercise the vested portion of his or her outstanding award, terminate any or all unexercised portion of the award at such time as the plan administrator deems appropriate. Such surrender or termination will take place as of the date of the Change of Control or such other date as the plan administrator may specify.
For purposes of the 2015 Equity Incentive Plan, “Change in Control” means the occurrence of any of the following events:
Persons will be considered to be acting as a group if they are owners of a corporation that enters into a merger, consolidation, purchase or acquisition of stock, or similar business transaction with us.
Compensation of Directors
Non-Employee Director Compensation Program
Our non-employee directors received the following compensation for their services:
Beginning January 23, 2018, our non-employee directors are entitled to receive the following compensation for their services:
2017 Director Compensation
The following table sets forth the compensation paid or earned for the fiscal year ended December 31, 2017 to our non-employee directors. Compensation paid to Michael D. Step, Andrew Ritter, and Ira Ritter is presented as part of the “Summary Compensation Table (2017 and 2016)” above. Our employee directors do not receive compensation for their service as directors. Dr. Merino is not included in the table below, as he was not appointed to our board of directors until January 17, 2017.
| Name of Director | Fees Earned and Paid in Cash ($) | Option Awards(2) ($) | All other compensation ($) | Total ($) | ||||||||||||
| Noah Doyle(1) | $ | — | $ | — | — | $ | — | |||||||||
| Matthew W. Foehr | $ | 25,000 | $ | — | — | $ | 25,000 | |||||||||
| Paul V. Maier | $ | 27,500 | $ | — | — | $ | 27,500 | |||||||||
| William M. Merino | $ | 20,000 | 13,068 | 33,068 | ||||||||||||
| Gerald T. Proehl | $ | 23,500 | $ | — | — | $ | 23,500 | |||||||||
We utilize the Black-Scholes option-pricing model to value awards. Key valuation assumptions include:
In addition to the assumptions used in the Black-Scholes option-pricing model, we also estimate a forfeiture rate to calculate the stock-based compensation for our equity awards. We will continue to use judgment in evaluating the expected volatility, expected terms and forfeiture rates utilized for our stock-based compensation calculations on a prospective basis.
As of December 31, 2017, each of our non-employee directors (other than Mr. Doyle and Dr. Merino) held option awards to purchase an aggregate of 12,000 shares of our Common Stock and no stock awards. Mr. Doyle held no stock awards or option awards as of December 31, 2017. Dr. Merino held option awards to purchase an aggregate of 12,000 shares of our common stock and no stock awards.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The following table sets forth certain information regarding the beneficial ownership of our common stock as of March 1, 2018 by:
Beneficial ownership is determined in accordance with the rules of the SEC and includes voting or investment power with respect to the securities. Shares of common stock that may be acquired by an individual or group within 60 days of March 1, 2018, pursuant to the exercise of options or warrants are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be outstanding for the purpose of computing the percentage ownership of any other person shown in the table. The percentage of beneficial ownership of our common stock is calculated based on an aggregate of 49,406,521 shares outstanding as of March 1, 2018.
Except as indicated in footnotes to this table, we believe that the stockholders named in this table have sole voting and investment power with respect to all shares of common stock shown to be beneficially owned by them, based on information provided to us by such stockholders. Unless otherwise indicated, the address for each director and executive officer listed is: c/o Ritter Pharmaceuticals, Inc., 1880 Century Park East, #1000, Los Angeles, California 90067.
| Beneficial Owner | Number of Shares Beneficially Owned | Percentage of Common Stock Beneficially Owned | ||||||
| Five Percent Stockholders | ||||||||
| Javelin Entities(1) | 7,776,534 | 15.6 | % | |||||
| Aleyska Investment Group L.P.(2) | 4,902,285 | 9.9 | % | |||||
| Aspire Capital Fund LLC(3) | 4,900,000 | 9.9 | % | |||||
| Baker Bros. Funds (4) | 2,800,000 | 5.7 | % | |||||
| Executive Officers, Directors and Director Nominees | ||||||||
| Michael D. Step(5) | 1,044,818 | 2.1 | % | |||||
| Andrew J. Ritter(6) | 1,735,364 | 3.5 | % | |||||
| Ira E. Ritter(7) | 1,735,364 | 3.5 | % | |||||
| Noah J. Doyle(1)(8) | 7,806,761 | 15.7 | % | |||||
| Matthew W. Foehr(9) | 53,833 | * | ||||||
| Paul V. Maier(10) | 18,208 | * | ||||||
| Gerald T. Proehl(11) | 68,208 | * | ||||||
| Dr. William M. Merino(12) | 13,398 | * | ||||||
| All current executive officers and directors as a group (9 persons)(13) | 16,322,434 | 28.5 | % | |||||
* Represents beneficial ownership of less than 1% of the shares of common stock.
(1) According to a Schedule 13D/A filed with the SEC on November 28, 2017 by Javelin Venture Partners, L.P. (“Javelin”), Javelin Venture Partners I SPV I, LLC (“Javelin SPV���), Javelin Venture Partners GP, L.P. (“Javelin GP, LP”), Javelin Venture Partners GP, LLC (“Javelin GP, LLC”), Noah J. Doyle and Jed Katz, in which the reporting person reported shared voting and dispositive power with respect to 7,776,534 shares. According to the Schedule 13D/A, this number consists of (i) 7,047,804 shares of common stock held directly by Javelin and 83,224 shares of common stock that Javelin has the right to acquire upon exercise of warrants to purchase common stock that are currently exercisable and (ii) 322,753 shares of common stock held directly by Javelin SPV and 322,753 shares of common stock that Javelin SPV has the right to acquire upon exercise of a warrant to purchase common stock that is currently exercisable. Javelin GP, LP serves as the general partner for Javelin and Javelin SPV, Javelin GP, LLC serves as the general partner of Javelin GP, LP, and Noah Doyle and Jed Katz serve as the managers of Javelin GP, LLC. As a result of the application of the beneficial ownership limitation described in this footnote, this number does not include 5,000,000 shares of common stock issuable upon exercise of warrants to purchase common stock owned by Javelin. Under the terms of these warrants, Javelin is not permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Javelin (and its affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. This limitation is referred to in this footnote as the “beneficial ownership limitation”. Javelin has the right to increase the beneficial ownership limitation in its discretion on 61 days’ prior written notice to us, provided that in no event is Javelin permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Javelin (and its affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. The address of the Javelin Entities is One Rincon Center, 101 Spear Street, Suite 255, San Francisco, California 94105. As a Manager of Javelin GP, LLC, Noah Doyle may be deemed the beneficial owner of these shares. Mr. Doyle expressly disclaims beneficial ownership over these shares except to the extent of his pecuniary interest therein.
(2) According to a Schedule 13G/A filed with the SEC on February 14, 2018 by Aleyska Investment Group, L.P., Alyeska Fund GP, LLC and Anand Parekh, in which the reporting persons reported shared voting and dispositive power with respect to 4,999,371 shares. As a result of the application of the beneficial ownership limitation described in this footnote, this number does not include 4,250,000 shares of common stock issuable upon the exercise of warrants to purchase common stock owned by Aleyska Investment Group L.P. (“Aleyska”). Under the terms of these warrants, Aleyska is not permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Aleyska (and its affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. This limitation is referred to in this footnote as the “beneficial ownership limitation”. Aleyska has the right to increase the beneficial ownership limitation in its discretion on 61 days’ prior written notice to us, provided that in no event is Aleyska permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Aleyska (and its affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. The address of Aleyska is 77 West Wacker Drive, 7th Floor, Chicago, IL 60601.
(3) As a result of the application of the beneficial ownership limitation described in this footnote, this number does not include (i) 7,500,000 shares of common stock issuable upon the exercise of warrants to purchase common stock owned by Aspire Capital Fund, LLC (“Aspire”) or (ii) 2,600,000 shares of common stock issuable upon the conversion of 1,040 shares of Series A Convertible preferred stock owned by Aspire. Under the terms of the warrants and Series A Convertible Preferred Stock issued to Aspire, Aspire is not permitted to exercise such warrants to purchase common stock or convert such Series A Convertible Preferred Stock into common stock to the extent that such exercise or conversion would result in Aspire (and its affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock or conversion of Series A Convertible Preferred Stock. This limitation on the exercise of the warrants and conversion of the Series A Convertible Preferred Stock to purchase common stock issued to Aspire is referred to in this footnote as the “beneficial ownership limitation.” Aspire has the right to increase the beneficial ownership limitation in its discretion on 61 days’ prior written notice to us, provided that in no event is Aspire permitted to exercise such warrants to purchase common stock or convert Series A Convertible Preferred Stock to the extent that such exercise or conversion would result in Aspire (and its affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock or conversion of Series A Preferred Stock. The address for Aspire is 155 N. Wacker Drive, Suite 1600, Chicago, IL 60606.
(4) According to a Schedule 13G filed with the SEC on February 13, 2018 by Baker Bros. Advisors LP, Baker Bros. Advisors (GP) LLC, Felix J. Baker, and Julian C. Baker, in which the reporting persons reported sole voting and dispositive power with respect to 2,800,000 shares. According to the Schedule 13G, this number consists of (i) 264,902 shares of our common stock owned by 667, L.P. and (ii) 2,535,098 shares of our common stock owned by Baker Brothers Life Sciences, L.P. (collectively, the “Baker Bros. Funds”). As a result of the application of the beneficial ownership limitation described in this footnote, this number does not include the following: (a) 1,238,287 shares of common stock issuable upon exercise of warrants to purchase common stock owned by 667, L.P. and 11,261,713 shares of common stock issuable upon exercise of warrants to purchase common stock owned by Baker Brothers Life Sciences, L.P. and (b) 1,000,000 shares of common stock issuable upon conversion of 400 shares of Series A Convertible Preferred Stock owned by 667, L.P. and 9,100,000 shares of common stock issuable upon conversion of 3,640 shares of Series A Convertible Preferred Stock owned by Baker Brothers Life Sciences, L.P. The information in this footnote is provided to us by Baker Bros. Advisors LP. Baker Bros. Advisors LP serves as the investment advisor to the Baker Bros. Funds. Baker Bros. Advisors (GP) LLC is the sole general partner of Baker Bros. Advisors LP. Julian C. Baker and Felix J. Baker are principals of Baker Bros. Advisors (GP) LLC. Baker Bros. Advisors LP has complete and unlimited discretion and authority with respect to the investment and voting power of the securities held by the Baker Bros. Funds. Julian C. Baker, Felix J. Baker, Baker Bros. Advisors LP and Baker Bros. Advisors (GP) LLC disclaim beneficial ownership of all shares held by the Baker Bros. Funds, except to the extent of their indirect pecuniary interest therein. Under the terms of the warrants and Series A Convertible Preferred Stock issued to the Baker Bros. Funds, the funds are not permitted to exercise such warrants to purchase common stock or convert such Series A Convertible Preferred Stock into common stock to the extent that such exercise or conversion would result in the Baker Bros. Funds (and their affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock or conversion of Series A Convertible Preferred Stock. This limitation on the exercise of the warrants and conversion of the Series A Convertible Preferred Stock to purchase common stock issued to the Baker Bros. Funds is referred to in this footnote as the “Beneficial Ownership Limitation.” The Baker Bros. Funds have the right to increase the Beneficial Ownership Limitation in their discretion on 61 days’ prior written notice to us, provided that in no event are the Baker Bros. Funds permitted to exercise such warrants to purchase common stock or convert Series A Convertible Preferred Stock to the extent that such exercise or conversion would result in the Funds (and their affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock or conversion of Series A Preferred Stock. The address for the Baker Bros. Funds is 860 Washington Street, 3rd Floor, New York, NY 10014.
(5) Includes 994,818 shares underlying stock option awards held by Mr. Step that are currently exercisable or exercisable within 60 days of March 1, 2018. The number of shares issuable upon exercise of options includes 70,230 shares subject to options that are currently exercisable, but are not subject to vesting within 60 days of March 1, 2018 and accordingly, if exercised, are subject to a repurchase right until vested.
(6) Includes 6,250 shares owned directly, 536,843 shares underlying stock option awards that are currently exercisable or exercisable within 60 days of March 1, 2018 and 1,192,271 shares beneficially owned by Stonehenge Partners LLC (“Stonehenge”), including 187,500 shares that are issuable upon the exercise of warrants that are currently exercisable. Under the terms of the warrants, Stonehenge is not permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Stonehenge (and its affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. This limitation is referred to in this footnote as the “beneficial ownership limitation”. Stonehenge has the right to increase the beneficial ownership limitation in its discretion on 61 days’ prior written notice to us, provided that in no event is Stonehenge permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Stonehenge (and its affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. As a managing partner of Stonehenge, Andrew Ritter may be deemed the beneficial owner of these shares. Andrew Ritter expressly disclaims beneficial ownership of the shares held by Stonehenge.
(7) Includes 536,843 shares underlying stock option awards that are currently exercisable or exercisable within 60 days of March 1, 2018, 6,250 shares held in a retirement plan trust of which the reporting person and his spouse are trustees, and 1,192,271 shares beneficially owned by Stonehenge, including 187,500 shares that are issuable upon the exercise of warrants that are currently exercisable. Under the terms of the warrants, Stonehenge is not permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Stonehenge (and its affiliates) beneficially owning more than 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. This limitation is referred to in this footnote as the “beneficial ownership limitation”. Stonehenge has the right to increase the beneficial ownership limitation in its discretion on 61 days’ prior written notice to us, provided that in no event is Stonehenge permitted to exercise such warrants to purchase common stock to the extent that such exercise would result in Stonehenge (and its affiliates) beneficially owning in the aggregate more than 19.99% of the number of shares of our common stock outstanding or the combined voting power of our securities outstanding immediately after giving effect to the issuance of shares of common stock issuable upon exercise of such warrants to purchase common stock. As a managing partner of Stonehenge, Ira Ritter may be deemed the beneficial owner of these shares. Ira Ritter expressly disclaims beneficial ownership of the shares held by Stonehenge.
(8) Includes 22,727 shares owned directly by Mr. Doyle, 7,500 shares underlying a stock option award held by Mr. Doyle that are currently exercisable or exercisable within 60 days of March 1, 2018 and the shares beneficially owned by the Javelin Entities reflected in footnote (1) above. Mr. Doyle express disclaims beneficial ownership of the share held by the Javelin Entities.
(9) Includes 18,833 shares underlying a stock option award held by Mr. Foehr that are currently exercisable or exercisable within 60 days of March 1, 2018.
(10) Represents shares underlying a stock option award held by Mr. Maier that are currently exercisable or exercisable within 60 days of March 1, 2018.
(11) Includes 18,208 shares underlying a stock option award held by Mr. Proehl that are currently exercisable or exercisable within 60 days of March 1, 2018.
(12) Includes 12,000 shares underlying a stock option award held by Dr. Merino that are currently exercisable or exercisable within 60 days of March 1, 2018.
(13) Includes 7,775,481shares underlying stock options and warrants that are currently exercisable or exercisable within 60 days of March 1, 2018.
Equity Compensation Plan Information
The following table sets forth aggregate information for the fiscal year ended December 31, 2016, regarding the Company’s compensation plans, including individual compensation agreements, under which equity securities of the Company are authorized for issuance:
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights (#) | Weighted average exercise price of outstanding options, warrants and rights ($) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (#) | |||||||||
| (a) | (b) | (c) | ||||||||||
| Equity compensation plans approved by security holders | 2,541,800 | (1) | 5.93 | 26,720,193 | (2) | |||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 2,541,800 | (1) | 5.93 | 26,720,193 | (2) | |||||||
Item 13. Certain Relationships and Related Transactions and Director Independence
Certain Relationships and Related Party Transactions
Our Audit Committee is responsible for reviewing, approving and overseeing any transaction between the Company and its directors, director nominees, executive officers, greater than 5% beneficial owners, and each of their respective immediate family members, where the amount involved exceeds the lesser of (i) $120,000 and (ii) one percent (1%) of the average of our total assets at year-end for the prior two fiscal years. Since January 1, 2016, there have been no such transactions, except as described below.
October 2016 Public Offering
On October 31, 2016, we closed a public offering of 2,127,660 shares of our common stock at a price to the public of $2.35 per share. One of our existing stockholders holding in excess of 5% of our outstanding shares prior to the October 2016 Public Offering, Aspire, purchased sharesin the offeringfor $1,199,999.
October 2017 Public Offering
On October 3, 2017, we closed a public offering of (i) 34,550,000 Class A Units consisting of 34,550,000 shares of our common stock and warrants to purchase 34,550,000 shares of our common stock, at a public offering price of $0.40 per unit, and (ii) 9,180 Class B Units consisting of 9,180 shares of our Series A Convertible Preferred stock, with a stated value of $1,000, and convertible into an aggregate of 22,950,000 shares of our common stock, and warrants to purchase 22,950,000 shares of our common stock, at a public offering price of $1,000 per unit.Two of our existing stockholders holding in excess of 5% of our outstanding shares prior to the October 2017 Public Offering, Javelin and Aleyska, purchased Class A Units in the public offering for $2.0 million and $1.7 million, respectively.
Director Independence
Under NASDAQ’s continued listing requirements, a majority of a listed company’s board of directors must be comprised of independent directors, subject to certain exceptions and phase-in rules. In addition, NASDAQ’s continued listing requirements require that, subject to certain exceptions and phase-in rules, each member of a listed company’s audit, compensation and governance and nominating committees must be independent. Audit committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. Under NASDAQ’s continued listing requirements, a director will only qualify as an “independent director” if, in the opinion of that company’s board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
Based upon information requested from and provided by each director concerning their background, employment and affiliations, including family relationships, our board of directors has determined that each of Messrs. Doyle, Foehr, Maier, Proehl and Dr. Merino are independent under the applicable rules and regulations of NASDAQ. In making such determinations, the board of directors considered the relationships that each such non-employee director has with our company and all other facts and circumstances the board of directors deemed relevant in determining their independence.
Item 14. Principal Accountant Fees and Services
Fees and Services of Mayer Hoffman McCann P.C.
The following table sets forth the aggregate fees billed to the Company by MHM for the fiscal years ended December 31, 2017 and 2016:
| 2017 | 2016 | |||||||
| Audit Fees(1) | $ | 106,419 | $ | 167,049 | ||||
| Audit-Related Fees | ─ | ─ | ||||||
| Tax Fees | ─ | ─ | ||||||
| All Other Fees(2) | 77,900 | ─ | ||||||
| Total | $ | 184,319 | $ | 167,049 | ||||
The Audit Committee has adopted a formal policy on auditor independence requiring the advance approval by the Audit Committee of all audit and non-audit services provided by our independent registered public accounting firm. In determining whether to approve any services by our independent registered public accounting firm, the Audit Committee reviews the services and the estimated fees, and considers whether approval of the proposed services will have a detrimental impact on the auditor’s independence. On an annual basis, our management reports to the Audit Committee all audit services performed during the previous 12 months and all fees billed by our independent registered public accounting firm for such services.
In fiscal 2017 and 2016, all audit services and the corresponding fees were approved by our board of directors.
Item 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
The following financial statements of Ritter Pharmaceuticals, Inc., together with the report thereon of Mayer Hoffman McCann P.C., an independent registered public accounting firm, are included in this Annual Report on Form 10-K:
All schedules have been omitted because they are not required or because the required information is given in the Financial Statements or Notes thereto set forth under Item 8 above.
* Filed herewith.
+ Indicates management contract or compensatory plan or arrangement.
# XBRL (Extensible Business Reporting Language) information is furnished and not filed herewith, is not a part of a registration statement or Prospectus for purposes of sections 11 or 12 of the Securities Act of 1933, is deemed not filed for purposes of section 18 of the Securities Exchange Act of 1934, and otherwise is not subject to liability under these sections.
None.
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Date: March 19, 2018
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Michael D. Step, Andrew J. Ritter and Ira E. Ritter, jointly and severally, his attorneys-in-fact, each with the power of substitution, for him in any and all capacities, to sign any amendments to this Annual Report on Form 10-K, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, hereby ratifying and confirming all that each of said attorneys-in-fact, or his substitute or substitutes, may do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant on March 19, 2018 in the capacities indicated.
INDEX TO FINANCIAL STATEMENTS
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders
of Qualigen Therapeutics, Inc.
RITTER PHARMACEUTICALS, INC.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Ritter Pharmaceuticals (“Company”Qualigen Therapeutics, Inc. (the “Company”) as of December 31, 20172023 and 2016, andDecember 31, 2022, the related consolidated statements of operations and comprehensive loss, stockholders’ equity (deficit) and cash flows for each of the two years in the period ended December 31, 2017,2023, and the related notes (collectively referred to as the “financial“consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 20172023 and 2016,December 31, 2022, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2017,2023, in conformity with accounting principles generally accepted in the United States of America.
Going Concern Uncertainty
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 12 to the consolidated financial statements, the Company has incurred recurring operating lossesCompany’s current liquidity position and is dependent on additional financing to fund operations. These conditionsprojected cash needs raise substantial doubt about the Company’sits ability to continue as a going concern. Management’s plans in regard toregarding these matters are also described in Note 1 to the financial statements.2. The consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that maymight result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on thesethe Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing separate opinions on the critical audit matter or on the accounts or disclosures to which it relates.
| 33 |
Accounting for Financial Instruments – Modification of Convertible Debt with Warrants
Critical Audit Matter Description
As described in Note 8 to the consolidated financial statements, during the year ended December 31, 2023, the Company modified the conversion price and strike price of its convertible debenture and common stock purchase warrants, respectively. Further, the Company modified certain terms of its stock warrant agreements that were originally classified as liabilities enabling them to be classified as equity.
We identified the accounting for the modification of this complex financial instrument as a critical audit matter. This includes both the evaluation of the various features as potential embedded derivatives and the determination of the respective fair value of the instruments and the embedded features, as well as the determination of the appropriate classification of warrants between equity and liabilities. The application of the accounting guidance applicable to issuing and modifying a complex financial instrument requires significant judgment.
Determination of appropriate classification of warrants requires management’s judgments relating to the interpretations of relevant accounting guidance based on specific provisions of the warrant agreement. Accounting for the convertible notes and embedded conversion features requires management’s judgments related to initial and subsequent recognition of the debt and related features, use of a valuation model, and key inputs used in the selected valuation model.
How We Addressed the Matter in Our Audit
The primary procedures we performed to address this critical audit matter included:
| ● | Inspecting the agreements associated with the transactions and evaluating management’s technical accounting analysis, including the application of the relevant accounting literature. | |
| ● | Utilizing an auditor’s specialist to assist in assessing management’s analysis of the transaction, including (i) evaluating the contracts to identify relevant terms that affect the recognition of the financial instruments, (ii) assessing the appropriateness of conclusions reached by management, and (iii) reviewing the valuation model for derivatives, performing independent calculations, and examining the significant assumptions utilized in the valuation model. |
/s/ Baker Tilly US, LLP
We have served as the Company’s auditor since 2014.2018.
Orange County, CASan Diego, California
March 19, 2018April 5, 2024
RITTER PHARMACEUTICALS,QUALIGEN THERAPEUTICS, INC.
| December 31, | December 31, | |||||||||||||||
| December 31, 2017 | December 31, 2016 | 2023 | 2022 | |||||||||||||
| ASSETS | ||||||||||||||||
| Current assets | ||||||||||||||||
| Cash and cash equivalents | $ | 22,631,971 | $ | 7,046,282 | ||||||||||||
| Prepaid expenses | 167,400 | 156,752 | ||||||||||||||
| Cash | $ | 401,803 | $ | 3,165,985 | ||||||||||||
| Prepaid expenses and other current assets | 764,964 | 1,366,704 | ||||||||||||||
| Current assets of discontinued operations | — | 6,287,849 | ||||||||||||||
| Total current assets | 22,799,371 | 7,203,034 | 1,166,767 | 10,820,538 | ||||||||||||
| Property and equipment, net | — | 26,242 | ||||||||||||||
| Other assets | 10,326 | 10,326 | 866,481 | — | ||||||||||||
| Property and equipment, net | 23,873 | 23,542 | ||||||||||||||
| Non-current assets of discontinued operations | — | 8,236,711 | ||||||||||||||
| Total Assets | $ | 22,833,570 | $ | 7,236,902 | $ | 2,033,248 | $ | 19,083,491 | ||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY (DEFICIT) | ||||||||||||||||
| Current liabilities | ||||||||||||||||
| Accounts payable | $ | 2,237,579 | $ | 1,896,368 | $ | 2,222,983 | $ | 619,568 | ||||||||
| Accrued expenses | 454,252 | 1,222,735 | ||||||||||||||
| Other liablities | 15,757 | 14,736 | ||||||||||||||
| Accrued expenses and other current liabilities | 560,006 | 864,559 | ||||||||||||||
| Warrant liabilities | 54,600 | 788,100 | ||||||||||||||
| Warrant liabilities - related party | — | 2,834,547 | ||||||||||||||
| Convertible debt - related party | 1,299,216 | 60,197 | ||||||||||||||
| Current liabilities of discontinued operations | — | 3,441,198 | ||||||||||||||
| Total current liabilities | 2,707,588 | 3,133,839 | 4,136,805 | 8,608,169 | ||||||||||||
| Stockholders’ equity | ||||||||||||||||
| Series A preferred stock, $0.001 par value; 15,000,000 shares authorized; 9,140 and 0 shares issued and outstanding as of December 31, 2017 and 2016, respectively | 5,128,536 | - | ||||||||||||||
| Common stock, $0.001 par value; 225,000,000 and 25,000,000 shares authorized; 49,406,521 and 11,619,197 shares issued and outstanding as of December 31, 2017 and December 31, 2016, respectively | 49,407 | 11,619 | ||||||||||||||
| Non-current liabilities of discontinued operations | — | 1,708,732 | ||||||||||||||
| Total liabilities | 4,136,805 | 10,316,901 | ||||||||||||||
| Commitments and Contingencies (Note 10) | - | - | ||||||||||||||
| Stockholders’ equity (deficit) | ||||||||||||||||
| Qualigen Therapeutics, Inc. stockholders’ equity (deficit): | ||||||||||||||||
| Common stock, $ par value; shares authorized; and shares issued and outstanding as of December 31, 2023 and December 31, 2022, respectively | 43,262 | 42,110 | ||||||||||||||
| Additional paid-in capital | 68,279,473 | 49,559,020 | 114,655,565 | 110,528,050 | ||||||||||||
| Accumulated other comprehensive income | — | 50,721 | ||||||||||||||
| Accumulated deficit | (53,331,434 | ) | (45,467,576 | ) | (116,802,384 | ) | (103,385,172 | ) | ||||||||
| Total stockholders’ equity | 20,125,982 | 4,103,063 | ||||||||||||||
| Total Liabilities and Stockholders’ Equity | $ | 22,833,570 | $ | 7,236,902 | ||||||||||||
| Total Qualigen Therapeutics, Inc. stockholders’ equity (deficit) | (2,103,557 | ) | 7,235,709 | |||||||||||||
| Noncontrolling interest | — | 1,530,881 | ||||||||||||||
| Total Stockholders’ Equity (deficit) | (2,103,557 | ) | 8,766,590 | |||||||||||||
| Total Liabilities & Stockholders’ Equity (Deficit) | $ | 2,033,248 | $ | 19,083,491 | ||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
RITTER PHARMACEUTICALS,QUALIGEN THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
| 2023 | 2022 | |||||||
| For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| EXPENSES | ||||||||
| General and administrative | $ | 6,095,607 | 10,274,600 | |||||
| Research and development | 5,209,250 | 4,486,120 | ||||||
| Total expenses | 11,304,857 | 14,760,720 | ||||||
| LOSS FROM OPERATIONS | (11,304,857 | ) | (14,760,720 | ) | ||||
| OTHER EXPENSE (INCOME), NET | ||||||||
| Gain on change in fair value of warrant liabilities | (2,035,469 | ) | (907,203 | ) | ||||
| Interest expense, net | 1,524,722 | 34,397 | ||||||
| Loss on voluntary conversion of convertible debt | 1,077,287 | — | ||||||
| Loss on debt extinguishment | 625,653 | — | ||||||
| Loss on fixed asset disposal | 21,747 | — | ||||||
| Other income, net | (38,994 | ) | — | |||||
| Total other expense (income), net | 1,174,946 | (872,806 | ) | |||||
| LOSS BEFORE (BENEFIT) PROVISION FOR INCOME TAXES | (12,479,803 | ) | (13,887,914 | ) | ||||
| (BENEFIT) PROVISION FOR INCOME TAXES | (4,793 | ) | 6,548 | |||||
| NET LOSS FROM CONTINUING OPERATIONS | (12,475,010 | ) | (13,894,462 | ) | ||||
| DISCONTINUED OPERATIONS | ||||||||
| Loss from discontinued operations, net of tax | (683,008 | ) | (7,140,181 | ) | ||||
| Loss on disposal of discontinued operations, net of tax | (602,232 | ) | — | |||||
| LOSS FROM DISCONTINUED OPERATIONS | (1,285,240 | ) | (7,140,181 | ) | ||||
| NET LOSS | (13,760,250 | ) | (21,034,643 | ) | ||||
| Net loss attributable to non-controlling interest from discontinued operations | (343,038 | ) | (2,394,100 | ) | ||||
| Net loss attributable to Qualigen Therapeutics, Inc. | $ | (13,417,212 | ) | $ | (18,640,543 | ) | ||
| Net loss per common share, basic and diluted - continuing operations | $ | ) | $ | ) | ||||
| Net loss per common share, basic and diluted - discontinued operations | $ | ) | $ | ) | ||||
| Weighted-average number of shares outstanding, basic and diluted | ||||||||
| Other comprehensive loss, net of tax | ||||||||
| Net loss | $ | (13,760,250 | ) | $ | (21,034,643 | ) | ||
| Foreign currency translation adjustment from discontinued operations | 119,473 | 50,721 | ||||||
| Other comprehensive loss | (13,640,777 | ) | (20,983,922 | ) | ||||
| Comprehensive loss attributable to noncontrolling interest from discontinued operations | (304,735 | ) | (2,394,100 | ) | ||||
| Comprehensive loss attributable to Qualigen Therapeutics, Inc. | $ | (13,336,042 | ) | $ | (18,589,822 | ) | ||
| For the Year Ended | ||||||||
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Operating costs and expenses: | ||||||||
| Research and development | $ | 2,874,184 | $ | 13,292,488 | ||||
| Patent costs | 250,372 | 272,514 | ||||||
| General and administrative | 4,777,902 | 4,881,725 | ||||||
| Total operating costs and expenses | 7,902,458 | 18,446,727 | ||||||
| Operating loss | $ | (7,902,458 | ) | $ | (18,446,727 | ) | ||
| Other income: | ||||||||
| Interest income | 40,227 | 60,879 | ||||||
| Other (expense) income | (1,627 | ) | 1,214 | |||||
| Total other income | 38,600 | 62,093 | ||||||
| Net loss | $ | (7,863,858 | ) | $ | (18,384,634 | ) | ||
| Deemed dividend of Series A preferred stock | (3,111,020 | ) | ― | |||||
| Net loss applicable to common stockholders | $ | (10,974,878 | ) | $ | (18,384,634 | ) | ||
| Net loss per common share – basic and diluted | $ | (0.50 | ) | $ | (2.04 | ) | ||
| Weighted average common shares outstanding – basic and diluted | 22,149,510 | 8,993,317 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
RITTER PHARMACEUTICALS,QUALIGEN THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
| Stockholders’ Equity | Total | |||||||||||||||||||||||||||
| Series A Preferred Stock | Common Stock | Additional | Accumulated | Stockholders’ | ||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Paid-in Capital | Deficit | Equity | ||||||||||||||||||||||
| Balance at December 31, 2015 | - | - | 8,582,004 | 8,582 | 41,759,355 | (27,082,942 | ) | 14,684,995 | ||||||||||||||||||||
| Issuance of shares upon follow-on offering | 2,127,660 | 2,127 | 4,997,873 | 5,000,000 | ||||||||||||||||||||||||
| Commissions and offering costs of follow-on offering | (598,118 | ) | (598,118 | ) | ||||||||||||||||||||||||
| Issuance of shares under common stock purchase agreement | 888,835 | 889 | 2,025,173 | 2,026,062 | ||||||||||||||||||||||||
| Stock based compensation | 1,359,205 | 1,359,205 | ||||||||||||||||||||||||||
| Exercise of options on common stock | 20,698 | 21 | 15,532 | 15,553 | ||||||||||||||||||||||||
| Net loss | (18,384,634 | ) | (18,384,634 | ) | ||||||||||||||||||||||||
| - | $ | - | 11,619,197 | $ | 11,619 | $ | 49,559,020 | $ | (45,467,576 | ) | $ | 4,103,063 | ||||||||||||||||
| Issuance of shares upon follow-on offering | 9,180 | 5,150,980 | 34,550,000 | 34,550 | 17,844,220 | 23,029,750 | ||||||||||||||||||||||
| Commissions and offering costs of follow-on offering | (2,038,471 | ) | (2,038,471 | ) | ||||||||||||||||||||||||
| Issuance of shares under common stock purchase agreement | 3,137,324 | 3,138 | 1,996,862 | 2,000,000 | ||||||||||||||||||||||||
| Stock based compensation | 895,498 | 895,498 | ||||||||||||||||||||||||||
| Conversion of series A preferred shares | (40 | ) | (22,444 | ) | 100,000 | 100 | 22,344 | - | ||||||||||||||||||||
| Net loss | (7,863,858 | ) | (7,863,858 | ) | ||||||||||||||||||||||||
| Balance at December 31, 2017 | 9,140 | $ | 5,128,536 | 49,406,521 | $ | 49,407 | $ | 68,279,473 | $ | (53,331,434 | ) | $ | 20,125,982 | |||||||||||||||
| Shares | Amount | Paid-In Capital | Comprehensive Income | Accumulated Deficit | Equity (Deficit) | Noncontrolling Interest | Equity (Deficit) | |||||||||||||||||||||||||
| Common Stock | Additional | Accumulated Other Comprehensive | Total Qualigen Therapeutics, Inc. Stockholders’ | Total Stockholders’ | ||||||||||||||||||||||||||||
| Shares | Amount | Paid-In Capital | Income (Deficit) | Accumulated Deficit | Equity (Deficit) | Noncontrolling Interest | Equity (Deficit) | |||||||||||||||||||||||||
| Balance at December 31, 2022 | 4,210,737 | $ | 42,110 | $ | 110,528,050 | $ | 50,721 | $ | (103,385,172 | ) | $ | 7,235,709 | $ | 1,530,881 | $ | 8,766,590 | ||||||||||||||||
| Voluntary conversion of convertible debt into common stock | 841,726 | 842 | 1,111,740 | — | — | 1,112,582 | — | 1,112,582 | ||||||||||||||||||||||||
| Redemptions of convertible debt into common stock | 310 | 254,006 | — | — | 254,316 | — | 254,316 | |||||||||||||||||||||||||
| Fair value of warrant modification for professional services | — | — | 7,945 | — | — | 7,945 | — | 7,945 | ||||||||||||||||||||||||
| Fair value of warrant reclassified from liabilities to equity | — | — | 1,626,694 | — | — | 1,626,694 | — | 1,626,694 | ||||||||||||||||||||||||
| Stock-based compensation | — | — | 1,098,533 | — | — | 1,098,533 | 9,297 | 1,107,830 | ||||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | 28,597 | 81,170 | — | 109,767 | 38,303 | 148,070 | ||||||||||||||||||||||||
| Deconsolidation of discontinued operations | — | — | — | (131,891 | ) | — | (131,891 | ) | (1,235,443 | ) | (1,367,334 | ) | ||||||||||||||||||||
| Net loss | — | — | — | — | (13,417,212 | ) | (13,417,212 | ) | (343,038 | ) | (13,760,250 | ) | ||||||||||||||||||||
| Balance at December 31, 2023 | 5,362,128 | $ | 43,262 | $ | 114,655,565 | $ | — | $ | (116,802,384 | ) | $ | (2,103,557 | ) | $ | — | $ | (2,103,557 | ) | ||||||||||||||
| Common Stock | Additional | Accumulated Other | Total Qualigen Therapeutics, Inc. | Total | ||||||||||||||||||||||||||||
| Shares | Amount | Paid-In Capital | Comprehensive Income | Accumulated Deficit | Stockholders’ Equity | Noncontrolling Interest | Stockholders’ Equity | |||||||||||||||||||||||||
| Balance at December 31, 2021 | 3,529,018 | $ | 35,290 | $ | 101,274,073 | $ | — | $ | (84,744,629 | ) | $ | 16,564,734 | $ | — | $ | 16,564,734 | ||||||||||||||||
| Balance | 3,529,018 | $ | 35,290 | $ | 101,274,073 | $ | — | $ | (84,744,629 | ) | $ | 16,564,734 | $ | — | $ | 16,564,734 | ||||||||||||||||
| Stock issued upon exercise of warrants | 332,000 | 3,320 | 4,711 | — | — | 8,031 | — | 8,031 | ||||||||||||||||||||||||
| Stock-based compensation | — | — | 5,484,044 | — | — | 5,484,044 | — | 5,484,044 | ||||||||||||||||||||||||
| Common stock and prefunded warrants issued for business acquisition | 350,000 | 3,500 | 3,740,417 | — | — | 3,743,917 | 3,882,225 | 7,626,142 | ||||||||||||||||||||||||
| Noncontrolling interest adjustments relating to Stock-based compensation and other | — | — | (42,756 | ) | — | — | (42,756 | ) | 42,756 | — | ||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | — | 50,721 | — | 50,721 | — | 50,721 | ||||||||||||||||||||||||
| Fair value of warrant modification for professional services | — | — | 67,370 | — | — | 67,370 | — | 67,370 | ||||||||||||||||||||||||
| Fair value of warrant modification for business acquisition | — | — | 696 | — | — | 696 | — | 696 | ||||||||||||||||||||||||
| Issuance of rounded shares as a result of the reverse stock split | (281 | ) | — | (505 | ) | — | — | (505 | ) | — | (505 | ) | ||||||||||||||||||||
| Net loss | — | — | — | — | (18,640,543 | ) | (18,640,543 | ) | (2,394,100 | ) | (21,034,643 | ) | ||||||||||||||||||||
| Balance at December 31, 2022 | 4,210,737 | $ | 42,110 | $ | 110,528,050 | $ | 50,721 | $ | (103,385,172 | ) | $ | 7,235,709 | $ | 1,530,881 | $ | 8,766,590 | ||||||||||||||||
| Balance | 4,210,737 | $ | 42,110 | $ | 110,528,050 | $ | 50,721 | $ | (103,385,172 | ) | $ | 7,235,709 | $ | 1,530,881 | $ | 8,766,590 | ||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
RITTER PHARMACEUTICALS,QUALIGEN THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| For the Years Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Cash flows from operating activities | ||||||||
| Net loss | $ | (7,863,858 | ) | $ | (18,384,634 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | 5,720 | 5,209 | ||||||
| Stock based compensation | 895,498 | 1,359,205 | ||||||
| Loss on disposal of equipment | 1,627 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses and other assets | (10,648 | ) | 32,384 | |||||
| Accounts payable | 341,211 | 1,157,011 | ||||||
| Accrued expenses | (768,483 | ) | 608,594 | |||||
| Other liabilities | 1,021 | 13,513 | ||||||
| Net cash used in operating activities | (7,397,912 | ) | (15,208,718 | ) | ||||
| Cash flows from investing activities | ||||||||
| Purchase of property and equipment | (7,678 | ) | (8,063 | ) | ||||
| Net cash used in investing activities | (7,678 | ) | (8,063 | ) | ||||
| Cash flows from financing activities | ||||||||
| Proceeds from the issuance of shares upon closing of follow-on offering | 23,029,750 | 5,000,000 | ||||||
| Commission and issuance costs of follow-on offering | (2,038,471 | ) | (598,118 | ) | ||||
| Proceeds from the issuance of shares from common stock purchase agreement | 2,000,000 | 2,026,062 | ||||||
| Proceeds from exercising of options on common stock | - | 15,553 | ||||||
| Net cash provided by financing activities | 22,991,279 | 6,443,497 | ||||||
| Net (decrease) increase in cash and cash equivalents | 15,585,689 | (8,773,284 | ) | |||||
| Cash and cash equivalents at beginning of period | 7,046,282 | 15,819,566 | ||||||
| $ | 22,631,971 | $ | 7,046,282 | |||||
| Non-cash financing activities: | ||||||||
| Deemed dividend on Series A Preferred Stock | $ | 3,111,020 | $ | - | ||||
| Cash paid for taxes | $ | 800 | $ | 80,702 | ||||
| 2023 | 2022 | |||||||
| For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | ||||||||
| Net loss | $ | (13,760,250 | ) | $ | (21,034,643 | ) | ||
| Loss from discontinued operations, net of tax | (1,285,240 | ) | (7,140,181 | ) | ||||
| Loss from continuing operations | (12,475,010 | ) | (13,894,462 | ) | ||||
| Adjustments to reconcile loss from continuing operations to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 4,495 | 12,165 | ||||||
| Stock-based compensation | 1,098,533 | 4,765,276 | ||||||
| Change in fair value of warrant liabilities | (2,035,469 | ) | (906,345 | ) | ||||
| Loss on voluntary conversion of convertible debt | 1,077,287 | — | ||||||
| Accretion of discount on convertible debt | 1,469,640 | — | ||||||
| Loss on debt extinguishment | 625,653 | — | ||||||
| Loss on disposal of fixed assets | 21,747 | — | ||||||
| Fair value of warrant modification for professional services | 7,945 | 67,370 | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses and other assets | (264,741 | ) | (126,985 | ) | ||||
| Accounts payable | 1,603,422 | (43,440 | ) | |||||
| Accrued expenses and other current liabilities | (227,101 | ) | (472,441 | ) | ||||
| Net cash used in operating activities - continuing operations | (9,093,599 | ) | (10,598,862 | ) | ||||
| Net cash provided by (used in) operating activities - discontinued operations | (1,210,664 | ) | (2,648,679 | ) | ||||
| Net cash used in operating activities | (10,304,263 | ) | (13,247,541 | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Net cash provided by (used in) investing activities - discontinued operations | 4,215,943 | (183,763 | ) | |||||
| Net cash provided by (used in) investing activities | 4,215,943 | (183,763 | ) | |||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Net proceeds from warrant exercises | — | 7,173 | ||||||
| Proceeds from issuance of convertible debt | — | 2,903,847 | ||||||
| Payments on convertible notes payable | (550,000 | ) | — | |||||
| Fractional share payments related to the reverse stock split | — | (505 | ) | |||||
| Net cash (used in)/provided by financing activities - continuing operations | (550,000 | ) | 2,910,515 | |||||
| Net cash used in financing activities - discontinued operations | — | — | ||||||
| Net cash (used in) provided by financing activities | (550,000 | ) | 2,910,515 | |||||
| Net change in cash and restricted cash | (6,638,320 | ) | (10,520,789 | ) | ||||
| Effect of exchange rate changes on cash and restricted cash | — | 22,639 | ||||||
| Cash and restricted cash from continuing operations - beginning of period | 3,165,985 | 9,174,383 | ||||||
| Cash and restricted cash from discontinued operations - beginning of period | 3,874,138 | 8,363,890 | ||||||
| Less: cash and restricted cash from discontinued operations - end of period | — | (3,874,138 | ) | |||||
| Cash from continuing operations - end of period | $ | 401,803 | $ | 3,165,985 | ||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION | ||||||||
| Cash paid during the year for: | ||||||||
| Interest | $ | — | $ | — | ||||
| Taxes | $ | 5,571 | $ | 5,571 | ||||
| NONCASH FINANCING AND INVESTING ACTIVITIES: | ||||||||
| Net transfers to equipment held for lease from inventory | $ | 83,281 | $ | — | ||||
| Fair value of warrant liabilities on date of exercise | $ | — | $ | 858 | ||||
| Redemption of convertible debt into common stock | $ | 254,316 | $ | — | ||||
| Voluntary conversion of convertible debt into common stock | $ | 1,112,582 | $ | — | ||||
| Fair value of warrant modifications pursuant to Securities Purchase Agreement | $ | — | $ | 9,439 | ||||
| Fair value of warrant modifications for business acquisition | $ | — | $ | 33,543 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
RITTER PHARMACEUTICALS,QUALIGEN THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 — ORGANIZATION AND PRINCIPAL ACTIVITIESSUMMARY OF SIGNIFICANT ACCOUNTING POLICIES AND ESTIMATES
Organization
Ritter Pharmaceuticals, Inc. (“Ritter” or the “Company”) is a Delaware corporation headquartered in Los Angeles, California. The Company(the Company’s predecessor) was formed as a Nevada limited liability company on March 29, 2004 under the name Ritter Natural Sciences, LLC, andLLC. In September 2008, this company converted into a Delaware corporation on September 16, 2008.
under the name Ritter Pharmaceuticals, Inc. develops therapeutic products that modulate the human gut microbiomeOn May 22, 2020, upon completing a “reverse recapitalization” transaction with Qualigen, Inc., Ritter Pharmaceuticals, Inc. was renamed Qualigen Therapeutics, Inc. (the “Company”). Qualisys Diagnostics, Inc. was formed as a Minnesota corporation in 1996, reincorporated to treat gastrointestinal diseases. The Company conducts human gut health research by exploring metabolic capacitybecome a Delaware corporation in 1999, and then changed its name to Qualigen, Inc. in 2000. Qualigen, Inc. was a wholly-owned subsidiary of the gut microbiotaCompany. On July 20, 2023, the Company sold all of the issued and translatingoutstanding shares of common stock of Qualigen, Inc. to Chembio Diagnostics, Inc. (“Chembio”), a wholly-owned subsidiary of Biosynex, S.A. (“Biosynex”). Following the functionalityconsummation of prebiotic-based therapeutics. Thethis transaction, Qualigen, Inc. became a wholly-owned subsidiary of Chembio (see Note 5 – Discontinued Operations).
On May 26, 2022, the Company acquired shares of Series A-1 Preferred Stock of NanoSynex, Ltd. (“NanoSynex”) from Alpha Capital Anstalt (“Alpha”), a related party, in exchange for reverse split adjusted shares of the Company’s lead compound, RP-G28 is currently under developmentcommon stock and a prefunded warrant to purchase 331,464 reverse split adjusted shares of the Company’s common stock at an exercise price of $0.001 per share. These warrants were subsequently exercised on September 13, 2022. Concurrently with this transaction, the Company also entered into a Master Funding Agreement for the treatmentOperational and Technology Funding of lactose intolerance. There currently is no drug approved by the Food and Drug Administration (“FDA”NanoSynex Ltd., dated May 26, 2022, with NanoSynex (the “NanoSynex Funding Agreement”), to, among other things, provide for the treatmentfurther funding of lactose intolerance,NanoSynex, and purchased shares of Series B preferred stock from NanoSynex for a debilitating disease that affects over 1 billion people worldwide.
total purchase price of $600,000. The transactions resulted in the Company currently operatesacquiring a 52.8% interest in one business segment focusing onNanoSynex (the “NanoSynex Acquisition”). NanoSynex is a nanotechnology diagnostics company domiciled in Israel. On July 20, 2023, the developmentCompany entered into an Amendment and commercializationSettlement Agreement with NanoSynex (the “NanoSynex Amendment”), which amended the NanoSynex Funding Agreement, to, among other things, eliminate most of RP-G28. Thethe Company is not organized by market and is managed and operated as one business. A single management team reportsobligation for the further funding of NanoSynex. Pursuant to the chief operating decision maker,terms of the Chief Executive Officer. TheNanoSynex Amendment, the Company does not currently operate any separate lineslost its controlling interest in NanoSynex (see Note 5 -Discontinued Operations).
Basis of business or separate business entities.Presentation
NOTE 2 — BASIS OF PRESENTATION
The accompanying consolidated financial statements of the Company have been prepared in accordanceconformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”), Regulation S-X and rules and regulations of the Securities and Exchange Commission (“SEC”).
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company and its former wholly-owned and majority owned subsidiaries. All intercompany balances and transactions have been eliminated in consolidation. Any reference in these notes to applicable guidance is meant to refer to U.S. GAAP. The Company views its operations and manages its business in one operating segment. In general, the functional currency of the Company and its subsidiaries is the U.S. dollar. For NanoSynex, the functional currency was the local currency, New Israeli Shekels (NIS). As such, assets and liabilities for NanoSynex were translated into U.S. dollars with the effects of foreign currency translation adjustments reflected as a component of accumulated other comprehensive loss within the Company’s consolidated statements of changes in stockholders’ equity (deficit).
As of July 20, 2023, NanoSynex was deconsolidated from these financial statements as the transactions contemplated by the NanoSynex Amendment resulted in a loss of control of a subsidiary that constitutes a business under ASC 810. The retained investment in NanoSynex is accounted for prospectively as an equity method investment. See Note 5 – Discontinued Operations for further information.
Discontinued Operations
On July 20, 2023, the Company completed the sale of Qualigen, Inc. to Chembio Diagnostics, Inc. The sale of Qualigen Inc. constituted a significant disposition and as such, the Company concluded that the disposition of ownership in Qualigen, Inc. represented a strategic shift that had a major effect on its operations and financial results. Therefore, Qualigen, Inc. is classified as discontinued operations for all adjustments necessaryperiods presented herein.
| 39 |
On July 20, 2023, the Company entered into the NanoSynex Amendment, which amended the Master Funding Agreement for the fair presentationOperational and Technology Funding of NanoSynex Ltd., dated May 26, 2022, by and between the Company and NanoSynex (the “NanoSynex Funding Agreement”), a former majority owned subsidiary of the Company’s financial position for the periods presented.
Going Concern and Liquidity
The accompanying financial statements have been prepared assuming the Company, will continue as a going concern, which contemplates,to, among other things, forfeit 281,000 Series B Preferred Shares of NanoSynex held by the realization of assets and satisfaction of liabilitiesCompany, resulting in the normal coursedeconsolidation of business.NanoSynex. The disposition represents a strategic shift that will have a material effect on the Company’s operations and financial results. Accordingly, the business of NanoSynex is classified as discontinued operations for all periods presented herein.
See Note 5 - Discontinued Operations for further information.
Equity Method Investments
Following deconsolidation of NanoSynex on July 20, 2023, the Company accounts for its retained investment under the equity method of accounting as it retained the ability to exercise significant influence over the operating and financial policies of the investee. Under the equity method, the Company recognizes its proportionate share earnings or losses each reporting period with an adjustment to the carrying value of the investment. As of December 31, 2023, the carrying value of the retained investment was zero, and therefore the Company has not generated any product revenue and has not achieved profitable operations. The Company had net losses of approximately $7.9 million and $18.4 million for the years ended December 31, 2017 and 2016, respectively, and had net cash used in operating activities of approximately $7.4 million and $15.2 million, for the years ended December 31, 2017 and 2016, respectively. There is no assurance that profitable operations will ever be achieved, and, if achieved, could be sustained on a continuing basis. In addition, development activities, clinical and pre-clinical testing, and commercializationsuspended application of the Company’s products will require significant financing. These matters, among others, raise substantial doubt about the Company’s ability to continueequity method as a going concern.
Since inception, the operations of the Company have been funded through the sale of common shares, preferred shares and convertible debt. Management cannot be certain that additional funding will be available on acceptable terms, or at all. To the extent that the Company raises additional funds by issuing equity securities, the Company’s stockholders may experience significant dilution. Any debt financing, if available, may involve restrictive covenants that impact the Company’s ability to conduct business. If the Company is not ableliable for the obligations of the investee nor otherwise committed to raise additional capital when required or on acceptable terms,provide financial support. Future equity method earnings, if any, will not be recognized until the Company may have to (i) significantly delay, scale back or discontinueamount exceeds the development and/or commercialization of one or more product candidates; (ii) seeks collaboratorsunrecognized net losses in prior periods. See Note 5 – Discontinued Operations for product candidates at an earlier stage than otherwise would be desirablefurther information.
Accounting Estimates
Management uses estimates and on terms that are less favorable than might otherwise be available; (iii) relinquish or otherwise dispose of rights to technologies, product candidates or products that the Company would otherwise seek to develop or commercialize.
The financial statements do not include any adjustments that might be necessary if the Company is unable to continue as a going concern.
NOTE 3 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Use of Estimates
The preparation ofassumptions in preparing its consolidated financial statements in accordance with GAAP requires management to makeU.S. GAAP. Those estimates and assumptions that affect the reported amounts of assets and liabilities, andthe disclosure of contingent assets and liabilities, asand the reported revenues and expenses. The most significant estimates relate to the estimated fair value of in-process research and development, goodwill, warrant liabilities, and stock-based compensation. Actual results could vary from the dateestimates that were used.
Reverse Stock Split
On November 23, 2022, the Company effected a 1-for-10 reverse stock split of its outstanding shares of common stock (the “Reverse Stock Split”). The Reverse Stock Split reduced the Company’s shares of outstanding common stock, stock options, and warrants to purchase shares of common stock. Fractional shares of common stock that would have otherwise resulted from the Reverse Stock Split were rounded down to the nearest whole share and cash in lieu of fractional shares was paid to stockholders. All share and per share data for all periods presented in the accompanying financial statements and the reported amountsrelated disclosures have been adjusted retrospectively to reflect the Reverse Stock Split. The number of revenues and expenses during the reporting period. Actual results could differ from those estimates and such differences may be material to the financial statements. The more significant estimates and assumptions by management include among others; the valuation allowanceauthorized shares of deferred tax assets resulting from net operating lossescommon stock and the valuationpar value per share remains unchanged.
Cash
The Company considers all highly liquid investments purchased with an initial maturity of options on90 days or less and money market funds to be cash equivalents.
The Company maintains the Company’s common stock.
majority of its cash in government money market mutual funds and in accounts at banking institutions in the U.S. that are of high quality. Cash and Cash Equivalents
Cash consistsheld in these accounts often exceed the Federal Deposit Insurance Corporation (FDIC) insurance limits. If such banking institutions were to fail, the Company could lose all or a portion of amounts held in a financial institution and consists of immediately available fund balances. The funds were maintained at a stable financial institution, generally at amounts in excess of federally insured limits, which represents a concentrationsuch insurance limitations. In March 2023, Silicon Valley Bank and Signature Bank, and more recently in May 2023, First Republic Bank, were closed due to liquidity concerns and taken over by the FDIC. While the Company did not have an account at any of credit risk. Thethese banks, in the event of failure of any of the financial institutions where the Company has not experienced any losses on deposits ofmaintains its cash and cash equivalents, there can be no assurance that the Company would be able to date.access uninsured funds in a timely manner or at all. Any inability to access or delay in accessing these funds could adversely affect the Company’s business and financial position.
Property and Equipment
Property and equipment are recorded at cost and depreciated over their estimated useful lives using the straight-line method (see Note 4). Upon retirement or sale, the cost of assets disposed of and the related accumulated depreciation are removed from the accounts and any resulting gain or loss is credited or charged to income. Maintenance and repairs are charged to expense as incurred while expenditures for refurbishments and improvements that significantly add to the productive capacity or extend the useful life of an asset are capitalized.
Impairment of Long-Lived Assets
The Company periodically assesses potential impairments to its long-lived assets when there is evidence that events or changes in circumstances indicate that assets may not be recoverable. An impairment loss would be recognized when the impairment of long lived assets in accordance with Accounting Standards Codification (“ASC”) Topic 360,Property Plant and Equipment. When indicators of impairment are present, the Company evaluates the carrying value of these assets in relation to the operating performancesum of the business andexpected future undiscounted cash flows expected to result fromis less than the usecarrying amount of thesethe assets. No such impairments have been recognized duringThe amount of impairment loss, if any, will generally be measured as the difference between the net book value of the assets and their estimated fair values. During the years ended December 31, 2017 or 2016.2023 and 2022, no such impairment losses have been recorded.
Clinical TrialSegment Reporting
Operating segments are identified as components of an enterprise about which separate discrete financial information is available for evaluation by the chief operating decision-maker in making decisions regarding resource allocation and Pre-Clinical Study Accrualsassessing performance. To date, the Company has viewed its operations and managed its business as one segment operating primarily within the United States (and in Israel prior to the NanoSynex deconsolidation).
| 40 |
Research and Development
Except for acquired in process research and development (IPR&D), the Company expenses research and development costs as incurred including therapeutics license costs.
Patent Costs
The Company makes estimatesexpenses all costs as incurred in connection with patent applications (including direct application fees, and the legal and consulting expenses related to making such applications) and such costs are included in general and administrative expenses in the consolidated statement of accrued expensesoperations.
Business Combinations
The Company accounts for business combinations using the acquisition method pursuant to Financial Accounting Standards Board’s (“FASB”) ASC Topic 805. This method requires, among other things, that results of operations of acquired companies are included in the Company’s financial results beginning on the respective acquisition date, and that assets acquired and liabilities assumed are recognized at fair value as of each balance sheet datethe acquisition date. Intangible assets acquired in its financial statementsa business combination are recorded at fair value using a discounted cash flow model. The discounted cash flow model requires assumptions about the timing and amount of future net cash flows, the cost of capital and terminal values from the perspective of a market participant. Each of these factors can significantly affect the value of the intangible asset. Any excess of the fair value of consideration transferred (the “purchase price”) over the fair values of the net assets acquired is recognized as goodwill. The fair value of assets acquired and liabilities assumed in certain cases may be subject to revision based on the factsfinal determination of fair value during a period of time not to exceed 12 months from the acquisition date. Legal costs, due diligence costs, business valuation costs and circumstances known to it at that time. Accrued expenses for pre-clinical studies and clinical trials are based on estimates ofall other acquisition-related costs incurred and fees that may be associated with services provided by contract research organizations, clinical trial investigational sites, and other related vendors. Payments under certain contracts with such parties depend on factors such as successful enrollment of patients, site initiation and the completion of milestones. In accruing service fees, management estimates the time period over which services will be performed and the level of effort to be expended in each period. If possible, the Company obtains information regarding unbilled services directly from these service providers. However, the Company may be required to estimate these services based on other information available to it. If the Company underestimates or overestimates the activity or fees associated with a study or service at a given point in time, adjustments to research and development expenses may be necessary in future periods. Historically, estimated accrued liabilities have approximated actual expense incurred. Subsequent changes in estimates may result in a material change in the Company’s accruals.
Research and Development
The Company expenses the cost of research and development as incurred. Research and development expenses comprise costs incurred in performing research and development activities, including clinical trial costs, manufacturing costs for both clinical and pre-clinical materials as well as other contracted services, license fees, and other external costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when incurred.
Goodwill
Goodwill represents the activity is performed or whendifference between the goods have been received, rather than when payment is made, in accordance with ASC Topic 730,Researchpurchase price and Development.
Patent Costs
The Company has no historical data to support a probable future economic benefit for the arising patent applications, filing and prosecution costs. Therefore, patent costs are expensed as incurred. Should the Company experience a legal cost to defend a patent in the future, that cost would be capitalized only when it is part of the cost of retaining and obtaining the future economic benefit of the patent. Costs related to an unsuccessful outcome would be expensed.
Stock-based Compensation
Stock-based compensation cost for stock awards issued to employees, members of the Company’s board of directors and non-employees, is measured at the grant date based on the fair value of the awardidentifiable tangible and intangible net assets acquired, when accounted for using the purchase method of accounting. Goodwill has an indefinite useful life and is recognized as expense overnot amortized but is reviewed for impairment annually and whenever events or changes in circumstances indicate that the required service period, which is generally equal to the vesting period. Stock-based compensation is recognized only for those awards that are ultimately expected to vest. Common stock, stock options or warrants issued to non-employees, including consultants and memberscarrying value of the Company’s Scientific Advisory Board as considerationgoodwill may not be recoverable. In testing for goods or services received by the Company, are accounted for based onimpairment, the fair value of the equity instruments issued unlessreporting unit is compared to the carrying value. If the net assets assigned to the reporting unit exceed the fair value consideration received can be more reliably measured. The fair value of stock optionsthe reporting unit, an impairment loss equal to the difference is determined using the Black-Scholes option-pricing model. The fair value of any options issued to non-employees is recorded as expense over the vesting period. See Note 8 for further information.
Fair Value Measurements
The Company follows authoritative accounting guidance, which among other things, defines fair value, establishes a consistent framework for measuring fair value and expands disclosure for each major asset and liability category measured at fair value on either a recurring or nonrecurring basis. Fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability.
recorded. As a basis for considering such assumptions, a three-tier fair value hierarchy has been established, which prioritizes the inputs used in measuring fair value as follows:
As of December 31, 2017 and 2016, the carrying amounts of cash and cash equivalents, prepaid expenses, accounts payable and accrued expenses are generally considered to be representative of their fair value becauseresult of the short term natureannual goodwill impairment analysis, the Company recognized a $4,239,000 non-cash goodwill and fixed asset impairment charge in the valuation of these instruments. No transfers between levels have occurred duringits business acquisition of NanoSynex for the periods presented.year ended December 30, 2022.
Convertible Preferred Stock
The Company follows authoritative accounting guidance to distinguish liabilities from equity when assessing the classification and measurement of preferred stock. Preferred shares subject to mandatory redemptions are considered liabilities and measured at fair value. Conditionally redeemable preferred shares are considered temporary equity. All other preferred shares are considered as stockholders’ equity.
Derivative Financial Instruments and Warrant Liabilities
The Company does not use derivative instruments to hedge exposures to cash flow, market, or foreign currency risks. ManagementThe Company evaluates all of the Company’sits financial instruments, including issued stock purchase warrants, to determine if such instruments are derivatives or contain features that qualify as embedded derivatives. TheFor derivative financial instruments that are accounted for as liabilities, the derivative instrument is initially recorded at its fair value and is then re-valued at each reporting date, with changes in the fair value reported in the consolidated statements of operations and comprehensive loss. Depending on the features of the derivative financial instrument, the Company generally uses either the Black-Scholes option-pricing model or a Monte CarloMonte-Carlo simulation as applicable, to value the derivative instruments at inception and subsequent valuation dates when needed.dates. The classification of derivative instruments, including whether such instruments should be recorded as liabilities or as equity, is re-assessed at the end of each reporting period.period (See Note 7-Warrant Liabilities and Note 8- Convertible Debt - Related Party).
Accounting for Income TaxesFair Value Measurements
Deferred taxThe Company determines the fair value measurements of applicable assets and liabilities based on a three-tier fair value hierarchy established by accounting guidance and prioritizes the inputs used in measuring fair value. The Company discloses and recognizes the fair value of its assets and liabilities using a hierarchy that prioritizes the inputs to valuation techniques used to measure fair value. The hierarchy gives the highest priority to valuations based upon unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurements) and the lowest priority to valuations based upon unobservable inputs that are recognized forsignificant to the expected future consequencesvaluation (Level 3 measurements). The guidance establishes three levels of eventsthe fair value hierarchy as follows:
| ● | Level 1 - Inputs that reflect unadjusted quoted prices in active markets for identical assets or liabilities that the Company has the ability to access at the measurement date; |
| 41 |
| ● | Level 2 - Inputs other than quoted prices that are observable for the assets or liability either directly or indirectly, including inputs in markets that are not considered to be active; and | |
| ● | Level 3 - Inputs that are unobservable. |
Fair Value of Financial Instruments
Cash, accounts receivable, prepaids, accounts payable, and accrued liabilities are carried at cost, which management believes approximates fair value due to the short-term nature of these instruments.
Comprehensive Loss
Comprehensive loss consists of net income and foreign currency translation adjustments related to the discontinued operations of NanoSynex. Comprehensive gains (losses) have been reflected in the financial statements. Deferred tax assetsstatements of operations and liabilities are determinedcomprehensive loss and as a separate component in the statements of stockholders’ equity (deficit) for all periods presented.
Stock-Based Compensation
Stock-based compensation cost for equity awards granted to employees and non-employees is measured at the grant date based on the calculated fair value of the award using the Black-Scholes option-pricing model, and is recognized as an expense, under the straight-line method, over the requisite service period (generally the vesting period of the equity grant). If the Company determines that other methods are more reasonable, or other methods for calculating these assumptions are prescribed by regulators, the fair value calculated for the Company’s stock options could change significantly. Higher volatility, lower risk-free interest rates, and longer expected lives would result in an increase to stock-based compensation expense to employees and non-employees determined at the date of grant.
Income Taxes
Deferred income taxes are recognized for temporary differences betweenin the book and tax basis of assets and liabilities for financial statement and operating loss carryforwards, usingincome tax rates expectedreporting that arise due to be in effect for the years in which the differences are expected to reverse. Such differences arise primarily from stock-based compensation and net operating loss carryforwards. The Company recordscarry forwards, research and development credit carry forwards and from using different methods and periods to calculate depreciation and amortization, allowance for doubtful accounts, accrued vacation, research and development expenses, and state taxes. A provision has been made for income taxes due on taxable income and for the deferred taxes on the temporary differences.
Deferred tax assets are reduced by a valuation allowance to reduce deferred income tax assets when, in the opinion of management, it is more likely than not that some portion or all of the deferred tax assetassets will not be realized. Prior to September 15, 2008,Deferred tax assets and liabilities are adjusted for the Company was a limited liability companyeffects of changes in tax laws and the Company’s tax losses and credits generally flowed directly to the members.
Net Loss Per Share
Basic net loss per share is calculated by dividing net loss by the weighted-average common shares outstanding during the period. Diluted net loss per share is calculated by dividing net loss by the weighted-average common shares outstanding during the period using the treasury stock method or the two-class method, whichever is more dilutive. The potentially dilutive stock options issued under the 2015 Equity Incentive Plan (described in Note 8), Series A Convertible Preferred Stock (described in Note 6) and warrantsrates on the Company’s common stock (described in Notes 6 and 7) are not considered in the computationdate of diluted net loss per share because they would be anti-dilutive.
Comprehensive Income (Loss)
Comprehensive income (loss) is defined as the change in equity during a period from transactions and other events and circumstances from non-owner sources. The Company is required to record all components of comprehensive loss in the financial statements in the period in which they are recognized. Net income (loss) and other comprehensive loss, including foreign currency translation adjustments and unrealized gains and losses on investments are reported, net of their related tax effect, to arrive at a comprehensive loss. For the years ended December 31, 2017 and 2016, comprehensive loss was equal to the net loss.
Recent Accounting Pronouncements
In February 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update 2016-02,Leases (Topic 842)(“ASU 2016-02”). The provisions of ASU 2016-02 set out the principles for the recognition, measurement, presentation and disclosure of leases for both parties to a contract (i.e., lessees and lessors). The new standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification will determine whether the lease expense is recognized based on an effective interest method or on a straight line basis over the termenactment. Realization of the lease. A lesseedeferred income tax asset is also requireddependent on generating sufficient taxable income in future years. For more information, refer to record a right-of-use asset and a lease liability for all leases with a term of greater than 12 months regardless of their classification. Leases with a term of 12 months or less will be accounted for under the existing guidance for operating leases today. Topic 842 supersedes the previous lease standard, Topic 840Leases. The guidance is effective for annual periods and interim periods within those annual periods beginning afterNote 14 - Income Taxes.
In December 15, 2018, and is effective for the Company for the year ending December 31, 2019. The Company is currently evaluating the impact that the implementation of this standard will have on the Company’s financial statements.
On March 30, 2016,2023, the FASB issued Accounting Standards Update No. 2016-09,Compensation - Stock Compensation (Topic 718):2023-09, Improvements to Employee Share-Based Payment Accounting (“ASU 2016-09”). Among other things, ASU 2016-09Income Tax Disclosures, which requires that entities recognize excess tax benefits and deficiencies related to employee share-based payment transactions asmore detailed income tax expense or benefit. ASU 2016-09 also eliminates the requirement to reclassify excess tax benefits and deficiencies from operating activities to financing activities in the statement of cash flows.disclosures. The guidance isrequires entities to disclose disaggregated information about their effective for the annual periods and interim periods within those annual periods beginning after December 15, 2016.tax rate reconciliation as well as expanded information on income taxes paid by jurisdiction. The adoption of this standard did not havedisclosure requirements will be applied on a material impact on the Company’s financial statements.
On August 26, 2016, the FASB issued Accounting Standards Update No. 2016-15,Statement of Cash Flows (Topic 230),a consensus of the FASB’s Emerging Issues Task Force (“ASU 2016-15”). The new guidance amends Accounting Standards Codification No. 230 (“ASC 230”) to add or clarify guidance on the classification of certain cash receipts and payments in the statement of cash flows. ASC 230 lacks consistent principles for evaluating the classification of cash payments and receipts in the statement of cash flows. This has led to diversity in practice and, in certain circumstances, financial statement restatements. Therefore, the FASB issued ASU 2016-15prospective basis, with the intent of reducing diversity in practice with respect to eight types of cash flows. ASU 2016-15 is effective for annual and interim periods in fiscal years beginning after December 15, 2017, and is effective for the Company for the yearending December 31, 2018. The Company is currently evaluating the impact that the implementation of this standard will have on the Company’s financial statements.
In May 2017, the FASB issued Accounting Standards Update No. 2017-09,Compensation - Stock Compensation (Topic 718): Scope of Modification Accounting(“ASU 2017-09”). ASU 2017-09 provides guidance on the types of changes to the terms or conditions of share-based payment awards to which an entity would be requiredoption to apply modification accounting. An entity would not apply modification accounting if the fair value, vesting conditions, and classification of the awards are the same immediately before and after the modification.them retrospectively. The amendments are effective for the Company’s interim and annual reporting periods beginning January 1, 2018. The Company does not expect the adoption of ASU 2017-09 to have a material impact on its financial statements.
In July 2017, the FASB issued ASU 2017-11,Earnings Per Share (Topic 260); Distinguishing Liabilities from Equity (Topic 480); Derivatives and Hedging (Topic 815): (Part I) Accounting for Certain Financial Instruments with Down Round Features, (Part II) Replacement of the Indefinite Deferral for Mandatorily Redeemable Financial Instruments of Certain Nonpublic Entities and Certain Mandatorily Redeemable Noncontrolling Interests with a Scope Exception.The ASU allows companies to exclude a down round feature when determining whether a financial instrument (or embedded conversion feature) is considered indexed to the entity’s own stock. As a result, financial instruments (or embedded conversion features) with down round features may no longer be required to be classified as liabilities. A company will recognize the value of a down round feature only when it is triggered and the strike price has been adjusted downward. For equity-classified freestanding financial instruments, such as warrants, an entity will treat the value of the effect of the down round, when triggered, as a dividend and a reduction of income available to common shareholders in computing basic earnings per share. For convertible instruments with embedded conversion features containing down round provisions, entities will recognize the value of the down round as a beneficial conversion discount to be amortized to earnings. The guidance in ASU 2017-11standard is effective for fiscal years beginning after December 15, 2018,2024, with early adoption permitted. The Company is evaluating the disclosure requirements related to the new standard.
Foreign Currency Translation
The functional currency for the Company is the U.S. dollar. The functional currency for the discontinued operations of NanoSynex was the New Israeli Shekel (NIS). The financial statements of NanoSynex were translated into U.S. dollars using exchange rates in effect at each period end for assets and interim periods withinliabilities; using exchange rates in effect during the period for results of operations; and using historical exchange rates for certain equity accounts. The adjustment resulting from translating the financial statements of NanoSynex was reflected as a separate component of other comprehensive income (loss) (see Note 5 - Discontinued Operations).
Recent Accounting Pronouncements
The Company reviewed all other recently issued accounting pronouncements and concluded that they were either not applicable or not expected to have a significant impact to the financial statements.
| 42 |
Global Economic Conditions
Ongoing Wars in Ukraine and Israel
In February 2022, Russia invaded Ukraine. While the Company has no direct exposure in Russia and Ukraine, the Company continues to monitor any broader impact to the global economy, including with respect to inflation, supply chains and fuel prices. The full impact of the conflict on the Company’s business and financial results remains uncertain and will depend on the severity and duration of the conflict and its impact on regional and global economic conditions.
In October 2023, Hamas conducted terrorist attacks in Israel resulting in ongoing war. There continue to be hostilities between Israel and Hezbollah in Lebanon and Hamas in the Gaza Strip, both of which have resulted in rockets being fired into Israel, causing casualties and disruption of economic activities. In early 2023, there were a number of changes proposed to the political system in Israel by the current government which, if implemented as planned, could lead to large-scale protests and additional uncertainty, negatively impacting the operating environment in Israel. Popular uprisings in various countries in the Middle East over the last few years have also affected the political stability of those countries and have led to a decline in the regional security situation. Such instability may also lead to deterioration in the political and trade relationships that exist between Israel and these countries. Any armed conflicts, terrorist activities or political instability involving Israel or other countries in the region could adversely affect the Company’s minority interest in NanoSynex, its results of operations, financial condition, cash flows and prospects (see Note 5 – Discontinued Operations).
Inflation and Global Economic Conditions
During the year ended 2022 and continuing into the current fiscal years. Early adoption is permitted,year, global commodity and labor markets experienced significant inflationary pressures attributable to government stimulus and recovery programs, government deficit spending and supply chain issues. The Company cannot provide assurance that it will be successful in fully offsetting increased costs resulting from inflationary pressure. In addition, the global economy suffers from slowing growth and rising interest rates, and some economists believe that there may be a global recession in the near future. If the global economy slows, the Company’s business may be adversely affected.
Impact of COVID-19 Pandemic
The COVID-19 pandemic has had a dramatic impact on businesses globally and on the Company’s business as well. During the height of the pandemic, sales of diagnostic products decreased significantly and the guidance isCompany’s net loss increased significantly, as clinics and small hospitals’ demand for Qualigen, Inc.’s FastPack™ diagnostic test kits was reduced sharply, largely due to be applied usingdeferral of patients’ non-emergency visits to physician offices. In July 2023 the Company sold Qualigen, Inc., its wholly-owned subsidiary, to Chembio (see Note 5 - Discontinued Operations).
Other accounting standard updates are either not applicable to the Company or are not expected to have a full or modified retrospective approach.material impact on the Company’s consolidated financial statements.
NOTE 2 — LIQUIDITY AND GOING CONCERN
As of December 31, 2023, the Company had approximately $0.4million in cash and an accumulated deficit of $116.8 million. For the years ended December 31, 2023 and 2022, the Company used cash of $10.3million and $13.2 million, respectively, in operations.
The Company’s cash balances as of the date that these financial statements were issued along with the proceeds from the above sale to Chembio, without additional financing, are expected to fund operations only into the second quarter of 2024. The Company early adopted this guidanceexpects to continue to have net losses and negative cash flow from operations, which will challenge its liquidity. These factors raise substantial doubt about the Company’s ability to continue as a going concern for the one-year period following the date that these financial statements were issued.
| 43 |
There is no assurance that profitable operations will ever be achieved, or, if achieved, could be sustained on a continuing basis. In order to fully execute its business plan, the Company will require significant additional financing for planned research and development activities, capital expenditures, clinical testing for QN-302 and preclinical development of Pan-RAS, as well as commercialization activities.
Historically, the Company’s principal sources of cash have included proceeds from the issuance of common and preferred equity and proceeds from the issuance of debt. In December 2022 the Company raised $3.0 million from the sale of an 8% Senior Convertible Debenture (the “Debenture”) to Alpha (see Note 8 - Convertible Debt - Related Party). There can be no assurance that further financing can be obtained on favorable terms, or at all. If the Company is unable to obtain funding, the Company could be required to delay, reduce or eliminate research and development programs, product portfolio expansion or future commercialization efforts, which could adversely affect the Company’s business prospects.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The financial statements do not include any adjustments that would be necessary should the Company be unable to continue as a going concern, and therefore, be required to liquidate its assets and discharge its liabilities in other than the normal course of business and at amounts that may differ from those reflected in the accompanying financial statements
NOTE 3 — PREPAID EXPENSES AND OTHER CURRENT ASSETS
Prepaid expenses and other current quarter, effective October 1, 2017. As a result,assets consisted of the warrants issued in 2017, in connection with the October 2017 Public Offering,following at December 31, 2023 and December 31, 2022:
SCHEDULE OF PREPAID EXPENSES AND OTHER CURRENT ASSETS
| December 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Prepaid insurance | $ | 566,011 | $ | 1,329,034 | ||||
| Other prepaid expenses | 25,053 | 37,670 | ||||||
| Receivable from sale of Qualigen, Inc. | — | — | ||||||
| Prepaid research and development expenses | 173,900 | — | ||||||
| Prepaid expenses and other current assets | $ | 764,964 | $ | 1,366,704 | ||||
Prepaid expenses attributable to Qualigen, Inc. and NanoSynex were equity-classified.deemed disposed of as discontinued operations (see Note 5 - Discontinued Operations).
NOTE 4 — PROPERTYOTHER NON-CURRENT ASSETS
Other non-current assets consisted of the following at December 31, 2023:
SCHEDULE OF OTHER NON CURRENT ASSETS
| December 31, | ||||
| 2023 | ||||
| Funds held in escrow | $ | 450,000 | ||
| Long-term research and development deposits | 416,481 | |||
| Other non-current assets | $ | 866,481 | ||
| 44 |
NOTE 5 — DISCONTINUED OPERATIONS
The summary of assets and liabilities classified in discontinued operations as of December 31, 2022 are as follows:
SCHEDULE OF BALANCE SHEETS AND EQUIPMENTINCOME STATEMENT CLASSIFIED IN DISCONTINUED OPERATIONS
| Qualigen, Inc. | NanoSynex | Total | ||||||||||
| Current assets of discontinued operations | $ | 4,448,999 | $ | 1,838,850 | $ | 6,287,849 | ||||||
| Non-current assets of discontinued operations | 1,876,270 | 6,360,441 | 8,236,711 | |||||||||
| Total assets of discontinued operations | $ | 6,325,269 | $ | 8,199,291 | $ | 14,524,560 | ||||||
| Current liabilities of discontinued operations | $ | 1,299,948 | $ | 2,141,250 | $ | 3,441,198 | ||||||
| Non-current liabilities of discontinued operations | 1,350,975 | 357,757 | 1,708,732 | |||||||||
| Total liabilities of discontinued operations | $ | 2,650,923 | $ | 2,499,007 | $ | 5,149,930 | ||||||
The summary of gain (loss) from discontinued operations, net of tax, as of December 31, 2023 and 2022 are as follows:
| Year ended December 31, 2023 | Year ended December 31, 2022 | |||||||||||||||||||||||
| Qualigen, Inc. | NanoSynex | Total | Qualigen, Inc. | NanoSynex | Total | |||||||||||||||||||
| (Loss) from discontinued operations | $ | (171,701 | ) | $ | (511,307 | ) | $ | (683,008 | ) | $ | (2,142,763 | ) | $ | (4,997,418 | ) | $ | (7,140,181 | ) | ||||||
| Gain (loss) on disposal of discontinued operations | 3,876,778 | (4,479,010 | ) | (602,232 | ) | — | — | — | ||||||||||||||||
| Total gain (loss) from discontinued operations | $ | 3,705,077 | $ | (4,990,317 | ) | $ | (1,285,240 | ) | $ | (2,142,763 | ) | $ | (4,997,418 | ) | $ | (7,140,181 | ) | |||||||
Property
Sale of Qualigen, Inc.
On July 20, 2023, the Company completed the sale of Qualigen, Inc., its formerly wholly-owned subsidiary, to Chembio Diagnostics, Inc. for net cash consideration of $5.4 million, of which $4.9 million was received during the year ended December 31, 2023, and equipment consists$450,000 is being held in escrow until January 20, 2025 to satisfy certain Company indemnification obligations.
The assets and liabilities classified in discontinued operations for Qualigen, Inc. as of December 31, 2022 are as follows:
ASSETS AND LIABILITIES CLASSIFIED IN DISCONTINUED OPERATIONS
| December 31, | ||||
| 2022 | ||||
| Cash | $ | 2,246,482 | ||
| Accounts receivable, net | 512,088 | |||
| Inventory, net | 1,586,297 | |||
| Prepaid expenses and other current assets | 104,132 | |||
| Total current assets of discontinued operations | 4,448,999 | |||
| Right-of-use assets | 1,422,538 | |||
| Property and equipment, net | 289,696 | |||
| Intangible assets, net | 145,702 | |||
| Other assets | 18,334 | |||
| Total non-current assets of discontinued operations | 1,876,270 | |||
| Total assets of discontinued operations of Qualigen, Inc. | $ | 6,325,269 | ||
| Accounts payable | $ | 236,470 | ||
| Accrued vacation | 187,906 | |||
| Accrued expenses and other current liabilities | 518,766 | |||
| Deferred revenue, current portion | 116,161 | |||
| Operating lease liability, current portion | 240,645 | |||
| Total current liabilities of discontinued operations | 1,299,948 | |||
| Operating lease liability, net of current portion | 1,301,919 | |||
| Deferred revenue, net of current portion | 49,056 | |||
| Total non-current liabilities of discontinued operations | 1,350,975 | |||
| Total liabilities of discontinued operations of Qualigen, Inc. | $ | 2,650,923 | ||
| 45 |
The Company reclassified the following:
| Estimated Life | December 31, 2017 | December 31, 2016 | ||||||||
| Computer equipment | 5 years | $ | 13,582 | $ | 10,274 | |||||
| Furniture and fixtures | 7 years | 19,158 | 23,325 | |||||||
| Total property and equipment | 32,740 | 33,599 | ||||||||
| Accumulated depreciation | (8,867 | ) | (10,057 | ) | ||||||
| Property and equipment, net | $ | 23,873 | $ | 23,542 | ||||||
Depreciation expensefollowing statement of approximately $5,700 and $5,200 was recognizedoperations items to discontinued operations for the years ended December 31, 20172023 and 2016, respectively,2022, respectively:
| 2023 | 2022 | |||||||
For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| REVENUES | ||||||||
| Net product sales | $ | 3,661,121 | $ | 4,983,556 | ||||
| Total revenues | 3,661,121 | 4,983,556 | ||||||
| EXPENSES | ||||||||
| Cost of product sales | 2,551,114 | 4,302,755 | ||||||
| General and administrative | 610,559 | 561,047 | ||||||
| Research and development | 206,819 | 1,245,973 | ||||||
| Sales and marketing | 405,626 | 950,420 | ||||||
| Goodwill and fixed asset impairment | — | 75,000 | ||||||
| Total expenses | 3,774,118 | 7,135,195 | ||||||
| OTHER EXPENSE (INCOME), NET | ||||||||
| Loss on disposal of equipment held for lease | 63,302 | — | ||||||
| Interest (income) expense, net | — | (7,751 | ) | |||||
| Other expense (income), net | (4,898 | ) | (1,125 | ) | ||||
| Loss on fixed asset disposal | 300 | — | ||||||
| Total other expense (income), net | 58,704 | (8,876 | ) | |||||
| INCOME (LOSS) FROM DISCONTINUED OPERATIONS BEFORE DISPOSAL | (171,701 | ) | (2,142,763 | ) | ||||
| Gain on sale of Qualigen, Inc., net of tax | 3,876,778 | — | ||||||
| INCOME (LOSS) FROM DISCONTINUED OPERATIONS OF QUALIGEN, INC. | $ | 3,705,077 | $ | (2,142,763 | ) | |||
In connection with this transaction, the Company recorded a gain on the sale of Qualigen, Inc. in its consolidated financial statements for the years ended December 31, 2023:
Gain on sale of Qualigen, Inc. | ||||
| Fair value of consideration received | $ | 5,489,337 | ||
| Working capital adjustment | 235,402 | |||
| Total Assets of discontinued operations | (4,225,562 | ) | ||
| Total Liabilities of discontinued operations | 3,005,407 | |||
| Transaction expenses | (627,806 | ) | ||
| Gain on sale of Qualigen, Inc. | $ | 3,876,778 | ||
In the fourth quarter of 2023, the gain was adjusted upward by $17,000 upon final payment of transaction costs.
Amendment and Settlement Agreement with NanoSynex Ltd.
On July 20, 2023, the Company entered into and effectuated the NanoSynex Amendment, pursuant to which the Company agreed to, in exchange for eliminating all future NanoSynex Funding Agreement obligations for the Company to invest further cash in NanoSynex (except for obligations to lend NanoSynex $560,000 on or before November 30, 2023, and $670,000 on or before March 31, 2024), surrender Series B Preferred Shares of NanoSynex held by the Company, resulting in the Company’s ownership in NanoSynex being reduced from approximately 52.8% to approximately 49.97% of the voting equity of NanoSynex; in addition, the Company agreed to surrender approximately $ million of promissory notes which NanoSynex had issued to the Company under the NanoSynex Funding Agreement. On November 22, 2023, the Company further agreed to eliminate the Company’s obligations to lend NanoSynex $560,000 on or before November 30, 2023, and $670,000 on or before March 31, 2024, by instead surrendering shares of Series A-1 Preferred Stock of NanoSynex in an amount that reduced the Company’s ownership in NanoSynex voting equity from approximately 49.97% to 39.90%.
The surrender of Series B Preferred Shares of NanoSynex was accounted for as a loss of control of a subsidiary that constitutes a business under ASC 810. As a result, on July 20, 2023, the Company deconsolidated NanoSynex’s related assets, liabilities, accumulated other comprehensive income, and the noncontrolling interest. Subsequently, the retained investment in NanoSynex is accounted for as an equity method investment. On the date of deconsolidation, the Company recognized its retained investment at fair value, which during the preparation of these financial statements was determined to be de minimis based on various economic, industry, and other factors. As a result, the Company has discontinued recognition of its proportionate share of equity method losses following the date of initial recognition. Future equity method earnings, if any, will not be recognized until the amount exceeds the unrecognized net losses in prior periods.
| 46 |
Based upon the magnitude of the disposition and because the Company is exiting certain research and development operations, the disposition represents a strategic shift that will have a material effect on the Company’s operations and financial results. Accordingly, the business of NanoSynex is classified as discontinued operations for all periods presented herein.
The assets and liabilities classified in generaldiscontinued operations for NanoSynex as of December 31, 2022 are as follows:
ASSETS AND LIABILITIES CLASSIFIED IN DISCONTINUED OPERATIONS
| December 31, | ||||
| 2022 | ||||
| Cash | $ | 1,621,967 | ||
| Accounts receivable, net | 26,499 | |||
| Prepaid expenses and other current assets | 190,384 | |||
| Total current assets of discontinued operations | 1,838,850 | |||
| Restricted cash | 5,690 | |||
| Property and equipment, net | 29,149 | |||
| Intangible assets, net | 5,700,000 | |||
| Goodwill | 625,602 | |||
| Total non-current assets of discontinued operations | 6,360,441 | |||
| Total assets of discontinued operations of NanoSynex | $ | 8,199,291 | ||
| Accounts payable | $ | 1,273 | ||
| Accrued vacation | 115,002 | |||
| Accrued expenses and other current liabilities | 293,571 | |||
| R&D grant liability | 780,682 | |||
| Short term debt-related party | 950,722 | |||
| Total current liabilities of discontinued operations | 2,141,250 | |||
| Deferred tax liability | 357,757 | |||
| Total non-current liabilities of discontinued operations | 357,757 | |||
| Total liabilities of discontinued operations of NanoSynex | $ | 2,499,007 | ||
The Company reclassified the following statement of operations items to discontinued operations for the years ended December 31, 2023 and administrative expense2022, respectively:
| 2023 | 2022 | |||||||
| For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| EXPENSES | ||||||||
| Research and development | $ | 869,064 | $ | 1,105,040 | ||||
| Goodwill and fixed asset impairment | — | 4,164,000 | ||||||
| Total expenses | 869,064 | 5,269,040 | ||||||
| Loss on disposal of discontinued operations | 4,479,010 | — | ||||||
| (BENEFIT) PROVISION FOR INCOME TAXES | (357,757 | ) | (271,622 | ) | ||||
| LOSS FROM DISCONTINUED OPERATIONS | (4,990,317 | ) | (4,997,418 | ) | ||||
| Loss attributable to noncontrolling interest | (343,038 | ) | (2,394,100 | ) | ||||
| NET LOSS ATTRIBUTABLE TO STOCKHOLDERS | $ | (4,647,279 | ) | $ | (2,603,318 | ) | ||
| 47 |
In connection with this transaction, the Company recorded a loss on deconsolidation of NanoSynex in its consolidated financial statements for the years ended December 31, 2023:
| Loss on deconsolidation of NanoSynex | ||||
| Fair value of NanoSynex interest retained | $ | — | ||
| Net assets deconsolidated | (2,768,403 | ) | ||
| Non-controlling interest share | 1,235,443 | |||
| Accumulated other comprehensive income attributable to NanoSynex | 131,891 | |||
| Forgiveness of debt | (3,077,941 | ) | ||
| Loss from deconsolidation of NanoSynex | $ | (4,479,010 | ) | |
NOTE 6 — ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
Accrued expenses and other current liabilities consisted of the following at December 31, 2023 and December 31, 2022:
SCHEDULE OF ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
| December 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Board compensation | $ | 129,499 | 70,000 | |||||
| Interest (Convertible debt) | 10,004 | 2,829 | ||||||
| License fees | 32,975 | 150,130 | ||||||
| Payroll | 1,215 | 1,247 | ||||||
| Professional fees | 121,775 | 136,203 | ||||||
| Research and development | 104,402 | 329,412 | ||||||
| Vacation | 151,286 | 165,040 | ||||||
| Other | 8,850 | 9,698 | ||||||
| Accrued expenses and other current liabilities | $ | 560,006 | $ | 864,559 | ||||
Other accrued liabilities attributable to Qualigen Inc, and NanoSynex were deemed disposed of as discontinued operations (see Note 5 – Discontinued Operations).
NOTE 7 – WARRANT LIABILITIES
In 2004, the Company issued warrants to various investors and brokers for the purchase of Series C preferred stock in connection with a private placement (the “Series C Warrants”). The Series C Warrants were subsequently extended and, upon closing of the reverse recapitalization transaction with Ritter, exchanged for warrants to purchase common stock of the Company, at $7.195 per share, subject to adjustment. As of December 31, 2023, the Series C Warrants had a remaining term of 0.49 years. The Series C Warrants were determined to be liability-classified pursuant to the guidance in ASC 480 and ASC 815-40, based on the inclusion of a leveraged ratchet provision for subsequent dilutive issuances. On April 25, 2022, the Series C warrants were repriced from $7.195 per share exercise price to $6.00 per share exercise price with additional ratchet Warrants issued. On May 26, 2022, the Series C warrants were repriced from $6.00 per share exercise price to $5.136 per share exercise price with additional ratchet Warrants issued. As a result of these repricings, warrants were forfeited and warrants were reissued. On December 22, 2022, the Series C Warrants were repriced again from $5.136 per share exercise price to $1.32 per share exercise price with additional ratchet Warrants issued.
Additionally, on December 22, 2022, in conjunction with the issuance of the Debenture to Alpha (see Note 8 – Convertible Debt – Related Party), the Company issued to Alpha a warrant to purchase 2,500,000 shares of the Company’s common stock (the “Alpha Warrant”). The exercise price of the Alpha Warrant was $1.65 (equal to 125% of the conversion price of the Debenture on the closing date). The Alpha Warrant may be exercised by Alpha, in whole or in part, at any time before June 22, 2028, subject to certain terms and conditions described in the accompanying StatementsAlpha Warrant. The fair value of Operations.this warrant was included in Warrant liabilities-related party on the Company’s consolidated balance sheet as of December 31, 2022. On December 5, 2023, the Company entered into an Amendment No. 1 with regard to Securities Purchase Agreement, with Alpha. This Amendment amended two instruments which the Company issued under the Securities Purchase Agreement dated December 21, 2022: (a) the 8% Senior Convertible Debenture dated December 22, 2022 in favor of Alpha, and (b) the Common Stock Purchase Warrant dated December 22, 2022 in favor of Alpha. The Amendment reduced the conversion price of the Debenture from $1.32 per share to $0.73 per share (subject to possible future adjustment pursuant to the terms of the Debenture) and reduced the exercise price of the Warrant from $1.65 per share to $0.73 per share (subject to possible future adjustment pursuant to the terms of the Warrant). The Amendment also eliminated certain adjustment provisions of the Warrant. The Company determined the event resulted in equity classification for the Warrant and, accordingly, the Company remeasured the warrant liabilities to fair value, and reclassified.
As a result of the Alpha Warrant repricing, on December 5, 2023 the Series C Warrants were repriced again from $1.32 per share exercise price to $0.73 per share exercise price with 203,652 additional ratchet Series C Warrants issued, resulting in of these Series C Warrants outstanding and exercisable as of December 31, 2023.
| 48 |
The following table summarizes the activity in liability classified warrants for the year ended December 31, 2023:
SCHEDULE OF WARRANTS ACTIVITY
| Common Stock Warrants | ||||||||||||||||
| Shares | Weighted– Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2022 | 3,849,571 | $ | 1.53 | $1.32 - $1.65 | ||||||||||||
| Exercised | — | — | — | — | ||||||||||||
| Forfeited | (2,751,976 | ) | 1.53 | — | — | |||||||||||
| Expired | (1,097,595 | ) | 1.32 | 1.32 | — | |||||||||||
| Granted | 455,623 | 0.73 | 0.73 | |||||||||||||
| Total outstanding – December 31, 2023 | 455,623 | $ | 0.73 | $ | 0.73 | |||||||||||
| Exercisable | 455,623 | $ | 0.73 | $ | 0.73 | |||||||||||
The following table summarizes the activity in the Common Stock Warrants received in exchange for the Series C Warrants for the year ended December 31, 2022:
| Common Stock Warrants | ||||||||||||||||
| Shares | Weighted– Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding –December 31, 2021 | 248,162 | $ | 7.20 | $ | 7.20 | |||||||||||
| Exercised | (536 | ) | 7.20 | |||||||||||||
| Forfeited | (247,625 | ) | 7.20 | |||||||||||||
| Expired | — | — | ||||||||||||||
| Granted | 3,849,570 | 1.53 | ||||||||||||||
| Total outstanding – December 31, 2022 | 3,849,571 | $ | 1.53 | $ | 1.32 - 1.65 | |||||||||||
| Exercisable | 1,349,571 | $ | 1.32 | $ | 1.32 | |||||||||||
The following table presents the Company’s fair value hierarchy for its Common Stock Warrant liabilities measured at fair value on a recurring basis as of December 31, 2023:
SCHEDULE OF FAIR VALUE HIERARCHY FOR WARRANT LIABILITIES
| Quoted | ||||||||||||||||
| Market | Significant | |||||||||||||||
| Prices for | Other | Significant | ||||||||||||||
| Identical | Observable | Unobservable | ||||||||||||||
| Assets | Inputs | Inputs | ||||||||||||||
| Common Stock Warrant liabilities | (Level 1) | (Level 2) | (Level 3) | Total | ||||||||||||
| Balance as of December 31, 2022 | $ | — | $ | — | $ | 3,622,647 | $ | 3,622,647 | ||||||||
| Exercises | — | — | — | — | ||||||||||||
| Fair value of warrant reclassified from liabilities to equity | — | — | (1,626,694 | ) | (1,626,694 | ) | ||||||||||
| Loss on debt extinguishment | — | — | 94,116 | 94,116 | ||||||||||||
| Gain on change in fair value of warrant liabilities | — | — | (2,035,469 | ) | (2,035,469 | ) | ||||||||||
| Balance as of December 31, 2023 | $ | — | $ | — | $ | 54,600 | $ | 54,600 | ||||||||
There were no transfers of financial assets or liabilities between category levels for the year ended December 31, 2023.
The value of the warrant liabilities was based on a valuation received from an independent valuation firm determined using a Monte-Carlo simulation. For volatility, the Company considers comparable public companies as a basis for its expected volatility to calculate the fair value of common stock warrants and transitions to its own volatility as the Company develops sufficient appropriate history as a public company. The risk-free interest rate is based on U.S. Treasury notes with a term approximating the expected term of the common stock warrant. The Company uses an expected dividend yield of zero based on the fact that the Company has never paid cash dividends and does not expect to pay cash dividends in the foreseeable future. Any significant changes in the inputs may result in significantly higher or lower fair value measurements.
| 49 |
The following are the assumptions used in, and the weighted average and the range of assumptions used in estimating the fair value of warrant liabilities (weighted average calculated based on the number of outstanding warrants on each issuance) as of December 31, 2023 and December 31, 2022:
SCHEDULE OF ASSUMPTIONS OF WARRANT LIABILITIES
| December 31, 2023 | December 31, 2022 | |||||||||
| Actual | Range | Weighted Average | ||||||||
| Risk-free interest rate | 5.13 | % | 3.906% — 4.628% | 4.15 | % | |||||
| Expected volatility (peer group) | 68.9 | % | 88% — 103% | 98 | % | |||||
| Term of warrants (in years) | 0.49 | .90 — 5.48 | 3.9 | |||||||
| Expected dividend yield | 0.00 | % | 0.00% | 0.00 | % | |||||
NOTE 8 — CONVERTIBLE DEBT- RELATED PARTY
On December 22, 2022, the Company issued to Alpha an 8% Senior Convertible Debenture in the aggregate principal amount of $3,300,000 for a purchase price of $3,000,000 (less expenses) pursuant to the terms of a Securities Purchase Agreement, dated December 21, 2022. The Debenture has a maturity date of December 22, 2025 and was initially convertible, at any time, and from time to time, until the Debenture is no longer outstanding, at Alpha’s option, into shares of common stock of the Company (the “Conversion Shares”), at a price equal to $1.32 per share, subject to adjustment as described in the Debenture (the “Conversion Price”) and other terms and conditions described in the Debenture, including the necessary stockholder approvals, which the Company obtained at its 2023 annual meeting of stockholders on July 13, 2023. As a part of the same transaction, on December 22, 2022, the Company issued to Alpha a liability classified warrant (the “Alpha Warrant”) to purchase 2,500,000 shares of the Company’s common stock (see Note 7 - Warrant Liabilities). The exercise price of the Alpha Warrant was initially $1.65 (equal to 125% of the Conversion Price of the Debenture on the closing date) subject to adjustment as described in the Alpha Warrant. The Alpha Warrant may be exercised by Alpha, in whole or in part, at any time before June 22, 2028, subject to certain terms and conditions described in the Alpha Warrant, including the necessary stockholder approvals, which the Company obtained at its 2023 annual meeting of stockholders on July 13, 2023.
The proceeds from the transaction were used to advance the Company’s QN-302 Investigative New Drug candidate towards clinical trials and other working capital purposes.
Commencing June 1, 2023 and continuing on the first day of each month thereafter until the earlier of (i) December 22, 2025 and (ii) the full redemption of the Debenture (each such date, a “Monthly Redemption Date”), the Company must redeem $110,000 plus accrued but unpaid interest, liquidated damages and any amounts then owing under the Debenture (the “Monthly Redemption Amount”). The Monthly Redemption Amount must be paid in cash; provided that after the first two monthly redemptions, the Company may elect to pay all or a portion of a Monthly Redemption Amount in shares of common stock of the Company, based on a Conversion Price equal to the lesser of (i) the then Conversion Price of the Debenture and (ii) 85% of the average of the VWAPs (as defined in the Debenture) for the five consecutive trading days ending on the trading day that is immediately prior to the applicable Monthly Redemption Date. The Company may also redeem some or all of the then outstanding principal amount of the Debenture at any time for cash in an amount equal to 105% of the then outstanding principal amount of the Debenture being redeemed plus accrued but unpaid interest, liquidated damages and any amounts then owing under the Debenture. The Company’s election to pay monthly redemptions in Conversion Shares or to effect an optional redemption is subject to the satisfaction (or waiver) of the Equity Conditions (as defined in the Debenture), including the necessary stockholder approvals, which the Company obtained at its 2023 annual meeting of stockholders on July 13, 2023.
The Debenture accrues interest at the rate of 8% per annum, which did not begin accruing until December 1, 2023. Interest may be paid in cash or shares of common stock of the Company or a combination thereof at the option of the Company; provided that interest may only be paid in shares if the Equity Conditions have been satisfied (or waived).
Both the Debenture and the Alpha Warrant provide for adjustments to the Conversion Price and exercise price, respectively, in connection with stock dividends and splits, subsequent equity sales and rights offerings, pro rata distributions, and certain fundamental transactions. Both the Debenture and the Alpha Warrant include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
Pursuant to resale registration rights granted by the Company to Alpha in such Securities Purchase Agreement, the Company filed a resale registration statement on Form S-3 (File Number 333-269088) on December 30, 2022 registering the resale by Alpha of up to shares of common stock of the Company which could be issued to Alpha pursuant to the Debenture and the Alpha Warrant, which registration statement was declared effective by the SEC on January 5, 2023 (the “Original Registration Statement”). The Company later became ineligible to update the Original Registration Statement via incorporation by reference of its future SEC periodic and current reports as a result of its failure to timely file its annual report on Form 10-K for the fiscal year ended December 31, 2022. Therefore, the Company filed a Post-Effective Amendment No. 1 to Form S-3 on Form S-1 (No. 333-269088)(the “Post-Effective Amendment No. 1”) on September 1, 2023 in order to maintain the registration of the resale by Alpha of up to shares of common stock of the Company issuable under the Debenture and the Alpha Warrant., which Post-Effective Amendment No. 1 was declared effective by the SEC on September 7, 2023.
| 50 |
The Company evaluated the Debenture and the Alpha Warrant and determined that the Alpha Warrant is a freestanding financial instrument. Initially, the Alpha Warrant is not considered indexed to the Company’s own stock, because the settlement amount would not equal the difference between the fair value of a fixed number of the Company’s equity shares and a fixed strike price and all of the adjustment features in Section 3(b) of the Alpha Warrant are not down round provisions, as defined in ASU 2017-11. Accordingly, the Alpha Warrant was classified as a liability and recognized at fair value, with subsequent changes in fair value recognized in earnings.
The proceeds from the Debenture were allocated to the initial fair value of the Alpha Warrant, with the residual balance allocated to the initial carrying value of the Debenture. The Company has not elected the fair value option for the Debenture. The Debenture was recognized as proceeds received after allocating the proceeds to the Alpha Warrant, and then allocating remaining proceeds to a suite of bifurcated embedded derivative features (conversion option, contingent acceleration upon an Event of Default, and contingent interest upon an Event of Default), with the resulting difference, if any, allocated to the loan host instrument. The suite of derivative features was measured and determined to have no fair value.
The original issue discount of $0.3 million, the initial fair value of the Alpha Warrant of $2.8 million, the initial fair value of the suite of bifurcated embedded derivative features of $0, and the fees and costs paid to Alpha and other third parties of $0.1 million comprised the debt discount upon issuance. The debt discount is amortized to interest expense over the expected term of the Debenture using the effective interest method, in accordance with ASC 835-30. The debt host instrument of the Debenture will subsequently be measured at amortized cost using the effective interest method to accrete interest over its term to bring the Debenture’s initial carrying value to the principal balance at maturity.
Between January 9 and 12, 2023, the Company issued shares of common stock upon Alpha’s partial conversion of the Debenture at $1.32 per share for a total of $1,111,078 principal. Upon conversion, the Company recognized a loss on conversion of convertible debt of approximately $1.1 million, recorded to other expenses in the consolidated statements of operations.
In October and December 2023, the Company issued shares of common stock to Alpha in lieu of cash for monthly redemption payments on the Debenture at a weighted average price of $ per share. Upon redemption for shares, the Company recognized a loss on partial debt extinguishment of $34,315.
On September 22, 2023, the Company entered into a consent and waiver (the “Waiver”) with Alpha. Pursuant to the Waiver, Alpha consented to the Company’s election to pay all of the Monthly Redemption Amount for October 2023 in Conversion Shares (the “October Payment”) and waived the requirement of satisfaction of the Equity Conditions in relation to the October and December Payment. On October 3, 2023 the Company issued shares of common stock to Alpha in satisfaction of the October Payment. On December 8, 2023 the Company issued shares of common stock to Alpha in satisfaction of the December Payment.
On December 5, 2023, the Company and Alpha executed Amendment No. 1 with regard to Securities Purchase Agreement (the “SPA Amendment”), pursuant to which the Company and Alpha agreed to, among other things, reduce the Conversion Price of the Debenture from $1.32 per share to $0.73 per share and reduce the exercise price of the Alpha Warrant from $1.65 per share to $0.73 per share, in each case subject to certain adjustments. In addition, the SPA Amendment revised certain provisions of the Alpha Warrant to (i) limit the circumstances which would trigger a potential adjustment to the exercise price of the Alpha Warrant and (ii) clarify the treatment of the Alpha Warrant upon a Fundamental Transaction. The purpose of these revisions was to remove the terms that caused the Alpha Warrant to be liability-classified under U.S. GAAP. The Company performed an assessment and concluded that all remaining adjustment features in the revised language meet the FASB’s definition of a down-round feature. In addition, the Alpha Warrant was determined to meet all of the additional requirements for equity classification. Accordingly, as of December 5, 2023, the Company remeasured the Alpha Warrant to its fair value immediately prior to the modification and recognized the change in fair value in earnings. The incremental fair value impact from the Alpha Warrant modification of $0.09 million was included in the Company’s evaluation of the Debenture modification under ASC 470, discussed further below. The Company then reclassified the Alpha Warrant liability to equity at its post-modification fair value of $1.6 million.
In accordance with ASC 470-50, the Company determined that the modified terms of the Debenture were substantially different when compared to the original terms that existed prior to the SPA Amendment, and thus the event was required to be accounted for as a debt extinguishment. Accordingly, the Company derecognized the net carrying value of the original Debenture, and recorded the new debt instrument at its fair value of $1.4 million, and recorded a $0.6 million loss on debt extinguishment. The difference between the remaining Debenture principal and its fair value on December 5, 2023 was recorded as a debt discount and will be amortized to interest expense over the expected term of the Debenture using the effective interest method, in accordance with ASC 835-30.
During the year ended December 31, 2023, the Company recognized a loss on voluntary conversion of convertible debt of approximately $1.1 million, recognized an extinguishment loss of $0.6 million upon October and December 2023 share redemptions and December 2023 modification of the Debenture, and recorded accrued interest of approximately $1.5 million, in other expenses in the consolidated statements of operations. During the year ended December 31, 2023 the Company paid Monthly Redemption Amounts of $550,000 in cash and $220,000 in common stock, and as of December 31, 2023 the remaining Debenture principal balance was approximately $1.4 million, the remaining discount was approximately $0.1 million, and the fair value of the suite of bifurcated embedded derivative features was $0.
| 51 |
The senior convertible debt comprises the following:
SCHEDULE OF SENIOR SECURED CONVERTIBLE DEBT
December 31, 2023 | December 31, 2022 | |||||||
| Senior convertible debenture | $ | 1,418,922 | $ | 3,300,000 | ||||
| Discount on convertible debenture | (119,706 | ) | (3,239,803 | ) | ||||
| Total convertible debt-related party | $ | 1,299,216 | $ | 60,197 | ||||
As of December 31, 2023, there were no unwaived events of default or violation of any covenants under the Company’s financing obligations.
Basic loss per share (“EPS”) is computed by dividing net loss by the weighted-average number of common shares outstanding. Diluted EPS is computed based on the sum of the weighted-average number of common shares and potentially dilutive common shares outstanding during the period. Potentially dilutive common shares consist of shares issuable from convertible debt, stock options and warrants.
SCHEDULE OF DILUTIVE SECURITIES EXCLUDED FROM DILUTED NET LOSS PER SHARE
| 2023 | 2022 | |||||||
For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Net loss used for basic earnings per share | $ | (13,417,212 | ) | $ | (18,640,543 | ) | ||
| Basic weighted-average common shares outstanding | 5,072,709 | 3,840,340 | ||||||
| Dilutive potential shares issuable from convertible debt, stock options and warrants | — | — | ||||||
| Diluted weighted-average common shares outstanding | 5,072,709 | 3,840,340 | ||||||
NOTE 10 — COMMITMENTS AND CONTINGENCIES
Master ServicesLitigation and Other Legal Proceedings
On November 9, 2021, the Company was named as a defendant in an action brought by Mediant Communications Inc. (“Mediant”) in the U.S. District Court for the Southern District of New York. The complaint alleged that Qualigen entered into an implied contract with Mediant, whereby Qualigen retained Mediant to distribute proxy materials and subsequently conduct shareholder vote tabulations. The Company filed a Motion to Dismiss with the District Court and on March 14, 2022 a hearing was held during which the presiding judge ruled in favor of the Motion to Dismiss. The Company and Mediant settled the litigation on April 5, 2022 in the amount of $96,558, at which time the amount was paid.
NOTE 11 — RESEARCH AND LICENSE AGREEMENTS
UCL Business Limited
In January 2022, the Company entered into a License Agreement with UCL Business Limited to obtain an exclusive worldwide in-license of a genomic quadruplex (G4)-selective transcription inhibitor drug development program which had been developed at University College London, including lead and back-up compounds, preclinical data and a patent estate. (UCL Business Limited is the commercialization company for University College London.) The program’s lead compound is now being developed at the Company under the name QN-302 as a candidate for treatment for pancreatic ductal adenocarcinoma, which represents the vast majority of pancreatic cancers. The License Agreement required a $150,000 upfront payment, reimbursement of past patent prosecution expenses (approximately $160,000), and (if and when applicable) tiered royalty payments in the low to mid-single digits, clinical/regulatory/sales milestone payments and a percentage of any non-royalty sublicensing consideration paid to the Company.
| 52 |
For the years ended December 31, 2023 and 2022 there were license costs of approximately $128,000 and $338,000, respectively, related to this agreement which are included in research and development expenses in the consolidated statements of operations and other comprehensive loss.
QN-302 Phase 1 Study
On December 30, 2015, weIn June 2023, the Company entered into a Master ServiceClinical Research Services Agreement with Covance,Translational Drug Development, LLC (“TD2”) whereby TD2 agreed to perform certain clinical research and development services for the Company including but not limited to trial management, side identification and selection, site monitoring/management, medical monitoring, project management, data collection, statistical programming or analysis, quality assurance auditing, scientific and medical communications, regulatory affairs consulting and submissions, strategic consulting, and/or other related services. From time to time, the Company intends to enter into statements of work with TD2 for the performance of specific services under this Master Clinical Research Services Agreement.
In June 2023, the Company entered into a Master Laboratory Services Agreement with MLM Medical Labs, LLC (“MLM”) whereby MLM agreed to perform certain clinical research and development services for the Company including but not limited to laboratory, supply, testing, validation, data management, and storage services. From time to time, the Company intends to enter into work orders with MLM for the performance of specific services under this Master Laboratory Services Agreement.
In June 2023, the Company entered into a Master Services Agreement with Clinigen Clinical Supplies Management, Inc. (“Covance”Clinigen”) whereby Clinigen agreed to provide certain pharmaceutical products and/or services. From time to time, the Company intends to enter into statements of work with Clinigen for the performance of specific services under this Master Services Agreement.
In July 2023, pursuant to the above agreements, the Company entered into work orders and statements of work for clinical trial services for the conduct of the QN-302 Phase 1 study. The project timeline started in July 2023 and is expected to continue until approximately July 2026. The total amount to be paid under these work orders and statements of work is currently expected to be approximately $7.6 million over the term of the QN-302 Phase 1 study, subject to available funding.
University of Louisville Research Foundation
In March 2019, the Company entered into a sponsored research agreement and an option for a license agreement with University of Louisville Research Foundation, Inc. (“ULRF”) for development of several small-molecule RAS interaction inhibitor drug candidates. Under the terms of this agreement, the Company agreed to reimburse ULRF for sponsored research expenses of initially up to $693,000 for this program. This agreement was amended in February 2021, March 2022 and August 2023, with the current term of this agreement set to expire in December 2023 and the aggregate amount that the Company would reimburse ULRF for sponsored research expenses increased to approximately $2.9 million. In July 2020, the Company entered into an exclusive license agreement with ULRF for RAS interaction inhibitor drug candidates. Under the agreement, the Company took over development, regulatory approval and commercialization of the candidates from ULRF and is responsible for maintenance of the related intellectual property portfolio. In return, ULRF received approximately $112,000 for an upfront license fee and reimbursement of prior patent costs. In addition, the Company has agreed to pay ULRF (i) royalties, on patent-covered net sales associated with the commercialization, of 4% (on net sales up to a cumulative $250,000,000) or 5% (on net sales above a cumulative $250,000,000), until expiration of the licensed patent, and 2.5% (on net sales for any sales not covered by Licensed Patents), (ii) 30% to 50% of any non-royalty sublicensee income received (50% for sublicenses granted in the first two years of the ULRF license agreement, 40% for sublicenses granted in the third or fourth years of the ULRF license agreement, and 30% for sublicenses granted in the fifth year of the ULRF license agreement or thereafter), (iii) reimbursements for ongoing costs associated with the preparation, filing, prosecution and maintenance of licensed patents, incurred prior to July 2020, and (iv) payments ranging from $50,000 to $5,000,000 upon the achievement of certain regulatory and commercial milestones. Milestone payments for the first therapeutic indication would be $50,000 for first dosing in a Phase 1 clinical trial, $100,000 for first dosing in a Phase 2 clinical trial, $150,000 for first dosing in a Phase 3 clinical trial, $300,000 for regulatory marketing approval and $5,000,000 upon achieving a cumulative $500,000,000 of Licensed Product sales. The Company also must pay ULRF shortfall payments if the total amounts actually paid with respect to royalties and non-royalty sublicensee income for any year is less than the applicable annual minimum (ranging from $20,000 to $100,000) for such year.
Sponsored research expenses related to these RAS agreements for the years ended December 31, 2023 and December 31, 2022 were approximately $743,000 and $758,000, respectively, and are recorded in research and development expenses in the Consolidated Statements of Operations. License costs related to these agreements for the years ended December 31, 2023 and December 31, 2022 were approximately $133,000 and $40,000, respectively, and are included in research and development expenses in the Consolidated Statements of Operations.
| 53 |
Between June 2018 and April 2022, the Company entered into license and sponsored research agreements with ULRF for QN-247, a novel aptamer-based compound that has shown promise as an anticancer drug. Under the agreements, the Company took over development, regulatory approval and commercialization of the compound from ULRF and is responsible for maintenance of the related intellectual property portfolio. In return, ULRF received a $50,000 convertible promissory note in payment of an upfront license fee, which was subsequently converted into the Company’s common stock, and the Company agreed to reimburse ULRF for sponsored research expenses of up to approximately $805,000 and prior patent costs of up to $200,000. In addition, the Company agreed to pay ULRF (i) royalties, on patent-covered net sales associated with the commercialization of anti-nucleolin agent-conjugated nanoparticles, of 4% (on net sales up to a cumulative $250,000,000) or 5% (on net sales above a cumulative $250,000,000), until expiration of the last to expire of the licensed patents, (ii) 30% to 50% of any non-royalty sublicensee income received (50% for sublicenses granted in the first two years of the ULRF license agreement, 40% for sublicenses granted in the third or fourth years of the ULRF license agreement, and 30% for sublicenses granted in the fifth year of the ULRF license agreement or thereafter), (iii) reimbursements for ongoing costs associated with the preparation, filing, prosecution and maintenance of licensed patents, incurred prior to June 2018, and (iv) payments ranging from $100,000 to $5,000,000 upon the achievement of certain regulatory and commercial milestones. Milestone payments for the first therapeutic indication would be $100,000 for first dosing in a Phase 1 clinical trial, $200,000 for first dosing in a Phase 2 clinical trial, $350,000 for first dosing in a Phase 3 clinical trial, $500,000 for regulatory marketing approval and $5,000,000 upon achieving a cumulative $500,000,000 of Licensed Product sales. The Company also agreed to pay another $500,000 milestone payment for any additional regulatory marketing approval for each additional therapeutic (or diagnostic) indication. The Company must also pay ULRF shortfall payments if the total amounts actually paid with respect to royalties and non-royalty sublicensee income for any year is less than the applicable annual minimum (ranging from $10,000 to $50,000) for such year.
The sponsored research agreement for QN-247 expired in August 2022. The sponsored research expenses related to these QN-247 agreements for the years ended December 31, 2023 and December 31, 2022 were $0 and approximately $164,000, respectively, and these amounts are recorded in research and development expenses in the consolidated statements of operations and other comprehensive loss. License costs related to these agreements were approximately $23,000 and $94,000 for the years ended December 31, 2023 and December 31, 2022, respectively, and are included in research and development expenses in the consolidated statements of operations and other comprehensive loss.
In June 2020, the Company entered into an exclusive license agreement with ULRF for its intellectual property in the use of QN-165 as a treatment for COVID-19. Under the agreement, the Company took over development, regulatory approval and commercialization of the compound (for such use) from ULRF and is responsible for maintenance of the related intellectual property portfolio. In return, ULRF received approximately $24,000 for an upfront license fee and reimbursement of prior patent costs. In addition, the Company was required to enter into a separate sponsored research agreement with ULRF (for QN-165 as a treatment for COVID-19) for at least $250,000. In November 2020, the Company executed a sponsored research agreement with ULRF (for QN-165 as a treatment for COVID-19) supporting up to approximately $430,000 in research which satisfied this requirement. This sponsored research agreement expired in November 2021 and the exclusive license agreement was terminated on October 31, 2022. There were no sponsored research expenses or license costs related to these QN-165 agreements for the years ended December 31, 2023 and 2022.
NOTE 12 — STOCKHOLDERS’ EQUITY
As of December 31, 2023, and 2022 the Company had two classes of capital stock: common stock and preferred stock.
Common Stock
Holders of common stock generally vote as a class with the holders of the preferred stock and are entitled to one vote for each share held. Subject to the rights of the holders of the preferred stock to receive preferential dividends, the holders of common stock are entitled to receive dividends when and if declared by the Board of Directors. Following payment of the liquidation preference of the preferred stock, any remaining assets will be distributed ratably among the holders of the common stock and, on an as-if-converted basis, the holders of any preferred stock upon liquidation, dissolution or winding up of the affairs of the Company. The holders of common stock have no preemptive, subscription or conversion rights and there are no redemption or sinking fund provisions.
At December 31, 2023, the Company has reserved shares of authorized but unissued common stock for possible future issuance as follows:
| Exercise of issued and future grants of stock options | 755,715 | |||
| Conversion of convertible debt | 1,943,729 | |||
| Exercise of stock warrants | 3,081,717 | |||
| Total | 5,781,161 |
| 54 |
Preferred Stock
At December 31, 2023 and December 31, 2022, there were shares of preferred stock outstanding.
Stock Options and Equity Classified Warrants
Stock Options
The Company recognizes all compensatory stock-based payments as compensation expense over the service period, which is generally the vesting period.
In April 2020, the Company adopted the 2020 Stock Incentive Plan (the “2020 Plan”) which provides for the grant of incentive or non-statutory common stock options, restricted stock, stock bonus awards, stock appreciation rights, restricted stock units and performance awards to qualified employees, officers, directors, consultants and other service providers. At December 31, 2023 and December 31, 2022 there were and outstanding stock options, respectively, under the 2020 Plan and there were and Plan shares available, respectively, for future grant.
SCHEDULE OF STOCK OPTION ACTIVITY
| Shares | Weighted–Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2022 | 608,012 | $ | 35.02 | $ - $ | ||||||||||||
| Granted | — | — | — | — | ||||||||||||
| Expired | — | — | — | — | ||||||||||||
| Forfeited | (209,088 | ) | 34.71 | - | — | |||||||||||
| Total outstanding – December 31, 2023 | 398,924 | $ | 35.21 | $ — $ | ||||||||||||
| Exercisable (vested) | 320,918 | $ | 41.97 | $ — $ | ||||||||||||
| Non-Exercisable (non-vested) | 78,006 | $ | 7.36 | $ - $ | 8.36 | |||||||||||
The following represents a summary of the options granted (under the 2020 Plan and otherwise) to employees and non-employee service providers that were outstanding at December 31, 2022, and changes during the twelve months then ended:
| Shares | Weighted–Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2021 | 484,186 | $ | 60.70 | $ — $ | ||||||||||||
| Granted | 134,469 | 5.24 | — | |||||||||||||
| Expired | (9,379 | ) | 932.75 | - | — | |||||||||||
| Forfeited | (1,264 | ) | 22.64 | - | — | |||||||||||
| Total outstanding – December 31, 2022 | 608,012 | $ | 35.02 | $ — $ | ||||||||||||
| Exercisable (vested) | 288,704 | $ | 46.32 | $ — $ | ||||||||||||
| Non-Exercisable (non-vested) | 319,308 | $ | 24.80 | $ — $ | 8.59 | |||||||||||
There was approximately $ million and $ million of compensation costs related to outstanding options for the year ended December 31, 2023 and December 31, 2022, respectively. This cost is expected to be recognized over a weighted average period of years.
stock options were granted or exercised during the year ended December 31, 2023 or 2022.
The options awarded under the 2020 Plan will vest as determined by the Board of Directors but will not exceed a 10-year period.
| 55 |
There were options granted during the year ended December 31, 2023. The weighted average grant date fair value per share of the shares underlying options granted during the year ended December 31, 2022 was $.
Fair Value of Equity Awards
The Company utilizes the Black-Scholes option pricing model to value awards under the 2020 Plan, and for equity classified compensatory warrants. Key valuation assumptions include:
| ● | Expected dividend yield. The expected dividend is assumed to be zero, as the Company has never paid dividends and has no current plans to pay any dividends on the Company’s common stock. | |
| ● | Expected stock-price volatility. The Company’s expected volatility is derived from the average historical volatilities of publicly traded companies within the Company’s industry that the Company considers to be comparable to the Company’s business over a period approximately equal to the expected term. | |
| ● | Risk-free interest rate. The risk-free interest rate is based on the U.S. Treasury yield in effect at the time of grant for zero coupon U.S. Treasury notes with maturities approximately equal to the expected term. | |
| ● | Expected term. The expected term represents the period that the stock-based awards are expected to be outstanding. The Company’s historical share option exercise experience does not provide a reasonable basis upon which to estimate an expected term because of a lack of sufficient data. Therefore, the Company estimates the expected term by using the simplified method provided by the SEC. The simplified method calculates the expected term as the average of the time-to-vesting and the contractual life of the options. |
SCHEDULE OF ASSUMPTION USED IN BLACK-SCHOLES OPTION-PRICING METHOD
For the Years Ended December 31, | ||||||
| 2023 | 2022 | |||||
| Expected dividend yield | n/a | 0.00% | ||||
| Expected stock-price volatility | n/a | 103% | ||||
| Risk-free interest rate | n/a | % — % | ||||
| Expected average term of options (in years) | n/a | |||||
| Stock price | n/a | - | ||||
SCHEDULE OF SHARE-BASED COMPENSATION EXPENSE
| 2023 | 2022 | |||||||
| For the Years Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| General and administrative | $ | 939,228 | $ | 4,649,649 | ||||
| Research and development | 159,305 | 834,395 | ||||||
| Total | $ | 1,098,533 | $ | 5,484,044 | ||||
Equity Classified Compensatory Warrants
In connection with the $4.0 million equity capital raise as part of the May 2020 reverse recapitalization transaction, the Company issued common stock warrants to an advisor and its designees for the purchase of 81,143 reverse split adjusted shares of the Company’s common stock at a reverse split adjusted exercise price of $11.10 per share. The issuance cost of these warrants was charged to additional paid-in capital, and did not result in expense in the Company’s consolidated statements of operations and comprehensive loss.
In addition, various service providers hold equity classified compensatory warrants issued in 2017 and earlier (originally exercisable to purchase Series C convertible preferred stock, and now instead exercisable to purchase common stock) for the purchase of 66,802 reverse split adjusted shares of Company common stock at a weighted average exercise price of $23.40 per share. These are to be differentiated from the Series C Warrants described in Note 7- Warrant Liabilities.
| 56 |
On April 25, 2022, 60,000 warrants were repriced from $ per share exercise price to a reverse split adjusted exercise price of $6.00 per share exercise price and extended from June 3, 2023 to September 14, 2023. The increase in fair value of $67,370 for the modification of these warrants was charged to general and administrative expenses in the Company’s consolidated statements of operations and comprehensive loss. These warrants expired on September 14, 2023. On April 25, 2022 and May 26, 2022 an additional 67,620 reverse split adjusted warrants were repriced from reverse split adjusted $11.10 per share exercise price to $5.136 per share exercise price. The increase in fair value of $31,010 for the modification of these warrants was charged to additional paid-in capital and did not result in expense on the Company’s consolidated statements of operations and comprehensive loss. On December 22, 2022 67,620 warrants were repriced from $5.136 per share exercise price to $1.32 per share exercise price. The increase in fair value of $8,548 for the modification of these warrants was charged to additional paid-in capital and did not result in expense on the Company’s consolidated statements of operations and comprehensive loss. On December 5, 2023, 67,620 warrants were repriced from $1.32 per share exercise price to $0.73 per share exercise price. The increase in fair value of $7,945 for the modification of these warrants was charged to general and administrative expenses in the Company’s consolidated statements of operations and comprehensive loss.
No new compensatory warrants were issued during the year ended December 31, 2023 or 2022.
The following table summarizes the equity classified compensatory warrant activity for the year ended December 31, 2023:
SCHEDULE OF WARRANT ACTIVITY
| Common Stock | ||||||||||||||||
| Shares | Weighted– Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2022 | 179,046 | $ | 9.12 | $1.32 — $25.40 | ||||||||||||
| Granted to advisor and its designees | — | — | ||||||||||||||
| Exercised | — | — | ||||||||||||||
| Expired | (60,000 | ) | 6.00 | 6.00 | ||||||||||||
| Forfeited | — | — | ||||||||||||||
| Total outstanding – December 31, 2023 | 119,046 | $ | 10.69 | $0.73 — $25.40 | ||||||||||||
| Exercisable | 119,046 | $ | 10.69 | $0.73 — $25.40 | ||||||||||||
| Non-Exercisable | — | $ | — | $ | — | — | ||||||||||
The following table summarizes the equity classified compensatory warrant activity for the year ended December 31, 2022:
| Common Stock | ||||||||||||||||
| Shares | Weighted– Average Exercise Price | Range of Exercise Price | Weighted–Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2021 | 179,046 | $ | 15.20 | $11.10 — $25.40 | ||||||||||||
| Granted to advisor and its designees | — | — | ||||||||||||||
| Exercised | — | — | ||||||||||||||
| Expired | — | — | ||||||||||||||
| Forfeited | — | — | ||||||||||||||
| Total outstanding – December 31, 2022 | 179,046 | $ | 9.12 | $1.32 — $25.40 | ||||||||||||
| Exercisable | 179,046 | $ | 9.12 | $1.32- $25.40 | ||||||||||||
| Non-Exercisable | — | $ | — | $ | — | — | ||||||||||
There was $ in compensation costs related to outstanding warrants for the year ended December 31, 2023 and $ for the year ended December 31, 2022. As of December 31, 2023 and December 31, 2022, there was unrecognized compensation cost related to nonvested warrants.
| 57 |
Noncompensatory Equity Classified Warrants
In May 2020, as a commitment fee, the Company issued noncompensatory equity classified warrants to Alpha (a related party) for the purchase of 27,048 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $11.10 per share (of which warrants for 20,000 shares were subsequently exercised in December 2020). In July 2020 the Company issued noncompensatory equity classified warrants to Alpha for the purchase of 78,019 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $0.01 per share (which were subsequently exercised in July 2020), and 192,068 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $52.50 per share. In August 2020, the Company issued noncompensatory equity classified warrants to Alpha for the purchase of 128,783 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $60.00 per share. In December 2020, the Company issued noncompensatory equity classified warrants to Alpha for the purchase of 100,000 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $0.10 per share (which were exercised in February 2021) and 219,101 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $40.70 per share. In May 2022, the Company issued noncompensatory equity classified warrants to Alpha for the purchase of 331,464 reverse split adjusted shares of Company common stock at a reverse split adjusted exercise price of $0.01 per share.
On November 29, 2021, with the exception of the warrants to purchase 27,048 reverse split adjusted shares of the Company’s common stock at a reverse split adjusted exercise price of $11.10 per share, the exercise prices of all outstanding warrants to purchase a total of 539,951 reverse split adjusted shares of the Company’s common stock were modified to a reverse split adjusted exercise price of $20.00 per share and each of their remaining terms extended by six months. The fair value of the modification cost of these warrant modifications of approximately $2.3 million was charged to additional paid-in capital and did not result in expense on the Company’s consolidated statements of operations and comprehensive loss. In May 2022, pre-funded warrants to purchase 331,464 reverse split adjusted shares of the Company’s common stock at a reverse split adjusted exercise price of $0.01 per share with no expiration date were issued to Alpha. These warrants were subsequently exercised in 2022.
In conjunction with the NanoSynex Acquisition, on April 25, 2022 the exercise price of 7,048 reverse split adjusted outstanding warrants at $11.10 was modified to a reverse split adjusted exercise price of $6.00. The increase in fair value of $2,533, using a Monte Carlo pricing model for the modification of these warrants, was charged to additional paid-in capital and did not result in expense on the Company’s consolidated statements of operations and comprehensive loss. On May 26, 2022, the reverse split adjusted exercise price of these warrants was modified again to $5.136, and the increase in fair value of $696, using a Monte Carlo pricing model for the modification of these warrants, was included in consideration transferred in the NanoSynex Acquisition. On December 22, 2022 the exercise price of these warrants was modified again to $1.32. The increase in fair value of $891, using a Monte Carlo pricing model for the modification of those warrants, was charged to additional paid-in capital and did not result in expense on the Company’s consolidated statements of operations and comprehensive loss.
On December 5, 2023, the Company entered into an Amendment No. 1 with regard to Securities Purchase Agreement, with Alpha. This Amendment amended two instruments which the Company issued under the Securities Purchase Agreement dated December 21, 2022: (a) the 8% Senior Convertible Debenture dated December 22, 2022 in favor of Alpha, and (b) the Common Stock Purchase Warrant dated December 22, 2022 in favor of Alpha. The Amendment reduced the Conversion Price of the Debenture from $1.32 per share to $0.73 per share (subject to possible future adjustment pursuant to the terms of the Debenture) and reduced the Exercise Price of the Alpha Warrant from $1.65 per share to $0.73 per share (subject to possible future adjustment pursuant to the terms of the Alpha Warrant). The Amendment revised certain provisions of the Warrant which resulted in reclassification of the Warrant from liabilities to equity. For more details see Note 7 - Warrant Liabilities.
The following table summarizes the noncompensatory equity classified warrant activity for the year ended December 31, 2023:
SCHEDULE OF WARRANT ACTIVITY
| Common Stock | ||||||||||||||||
| Shares | Weighted–Average Exercise Price | Range of Exercise Price | Weighted– Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2022 | 547,003 | $ | 19.76 | $1.32 - $20.00 | ||||||||||||
| Legacy Ritter warrants | — | — | ||||||||||||||
| Reclassification of Alpha Warrant from warrant liabilities to equity | 2,500,000 | 0.73 | 0.73 | |||||||||||||
| Exercised | — | — | ||||||||||||||
| Expired | (539,953 | ) | 20.00 | |||||||||||||
| Forfeited | — | — | ||||||||||||||
| Total outstanding – December 31, 2023 | 2,507,050 | $ | 0.73 | |||||||||||||
| Exercisable | 2,507,050 | $ | 0.73 | 0.73 | ||||||||||||
| Non-Exercisable | — | $ | — | $ | — | — | ||||||||||
| 58 |
The following table summarizes the noncompensatory equity classified warrant activity for the year ended December 31, 2022:
| Common Stock | ||||||||||||||||
| Shares | Weighted– Average Exercise Price | Range of Exercise Price | Weighted– Average Remaining Life (Years) | |||||||||||||
| Total outstanding – December 31, 2021 | 554,914 | $ | 20.10 | 11.10— 37.78 | ||||||||||||
| Legacy Ritter warrants | — | — | ||||||||||||||
| Granted | 331,464 | 0.01 | 0.01 | |||||||||||||
| Exercised | (331,464 | ) | 0.01 | 0.01 | ||||||||||||
| Expired | (7,911 | ) | 37.78 | 37.78 | ||||||||||||
| Forfeited | — | — | 0 | |||||||||||||
| Total outstanding – December 31, 2022 | 547,003 | $ | 19.76 | 1.32 - 20.00 | ||||||||||||
| Exercisable | 547,003 | $ | 19.76 | 1.32 - 20.00 | ||||||||||||
| Non-Exercisable | — | $ | — | $ | — | — | ||||||||||
NOTE 13 — RELATED PARTY TRANSACTIONS
Convertible Debt
See Note 8 – Convertible Debt – Related Party for additional information concerning convertible debt – related party transactions. On December 22, 2022, the Company issued to Alpha, an 8% Senior Convertible Debenture in the aggregate principal amount of $3,300,000 for a purchase price of $3,000,000 pursuant to the terms of a Securities Purchase Agreement, dated December 21, 2022. As of December 31, 2023 the Debenture had a remaining principal balance of $1,418,922, and was convertible, at any time, and from time to time, at Alpha’s option, into shares of common stock of the Company, at a price equal to $0.73 per share, subject to adjustment as described in the Debenture and other terms and conditions described in the Debenture.
Warrants
Additionally, on December 22, 2022, in conjunction with the issuance of the Debenture to Alpha, the Company issued to Alpha the Alpha Warrant to purchase 2,500,000 shares of the Company’s common stock (the “Alpha Warrant”). As of December 31, 2023, the exercise price of the Alpha Warrant was $0.73. The Alpha Warrant may be exercised by Alpha, in whole or in part, at any time before June 22, 2028, subject to certain terms and conditions described in the Alpha Warrant. The Alpha Warrant is included in equity on the Company’s consolidated balance sheets (see Note 12 – Stockholders’ Equity).
NanoSynex
Pursuant to a Share Purchase Agreement dated April 29, 2022, the Company acquired shares of NanoSynex Series A-1 Preferred Stock from Alpha in exchange for reverse split adjusted shares of the Company’s common stock and a prefunded warrant to purchase 331,464 reverse split adjusted shares of the Company’s common stock at an exercise price of $0.001 per share.
NOTE 14 — INCOME TAXES
The following table presents domestic and foreign components of consolidated loss before income taxes from continuing operations for the periods presented:
SCHEDULE OF DOMESTIC AND FOREIGN COMPONENTS
December 31, 2023 | December 31, 2022 | |||||||
| Domestic | $ | (12,479,803 | ) | $ | (13,887,914 | ) | ||
| Foreign | — | — | ||||||
| Loss before provision for income taxes | $ | (12,479,803 | ) | $ | (13,887,914 | ) | ||
| 59 |
A reconciliation of the statutory income tax rates and the Company’s effective tax rate is as follows:
SCHEDULE OF RECONCILIATIONS OF STATUTORY INCOME TAX RATE
| December 31, 2023 | December 31, 2022 | |||||||
| Statutory federal income tax rate | 21.00 | % | 21.00 | % | ||||
| State taxes, net of federal tax benefit | 1.76 | % | 7.50 | % | ||||
| Non-deductible expenses | -0.64 | % | -2.09 | % | ||||
| NOL expiration | -5.26 | % | -19.88 | % | ||||
| Tax credit | 2.92 | % | 3.71 | % | ||||
| Goodwill impairment | 0.00 | % | 0.00 | % | ||||
| Foreign rate differential | 0.00 | % | 0.00 | % | ||||
| Change in fair value of warrant liability | 3.35 | % | 0.00 | % | ||||
| Tax impact of convertible debenture | -5.46 | % | 1.37 | % | ||||
| Tax impact of divestiture | -229.26 | % | 4.01 | % | ||||
| True-up | -10.65 | % | 2.24 | % | ||||
| Change in valuation allowance | 222.28 | % | -17.91 | % | ||||
| Income taxes provision (benefit) | 0.04 | % | -0.05 | % | ||||
Income tax expense for the year ended December 31, 2023 and 2022 consisted of the following:
SCHEDULE OF PROVISION FOR INCOME TAXES
| December 31, 2023 | December 31, 2022 | |||||||
| For the Years Ended | ||||||||
| December 31, 2023 | December 31, 2022 | |||||||
| Current | ||||||||
| U.S. Federal | $ | (10,000 | ) | $ | — | |||
| U.S. State | 5,000 | 7,000 | ||||||
| Total current provision | (5,000 | ) | 7,000 | |||||
| Deferred Benefit | ||||||||
| U.S. Federal | 23,128,000 | (236,000 | ) | |||||
| U.S. State | 4,697,000 | (2,252,000 | ) | |||||
| Total deferred benefit | 27,825,000 | (2,488,000 | ) | |||||
| Change in valuation allowance | (27,825,000 | ) | 2,488,000 | |||||
| Total provision (benefit) for income taxes | $ | (5,000 | ) | $ | 7,000 | |||
The components of deferred tax assets and liabilities are as follows:
SCHEDULE OF DEFERRED TAX ASSETS AND LIABILITIES
| December 31, 2023 | December 31, 2022 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss | $ | 8,644,000 | $ | 32,587,000 | ||||
| Research and development credits | 4,970,000 | 7,857,000 | ||||||
| Accrued expenses | 68,000 | 1,020,000 | ||||||
| Patent | — | — | ||||||
| Stock compensation | 3,004,000 | 3,069,000 | ||||||
| Research and development expenses | 1,102,000 | 1,196,000 | ||||||
| Fixed assets | — | 280,000 | ||||||
| Total deferred income tax assets | 17,788,000 | 46,009,000 | ||||||
| Deferred tax liabilities: | ||||||||
| Intangible assets | — | (13,000 | ) | |||||
| Right-of-use asset | — | (382,000 | ) | |||||
| Total deferred income tax liabilities | — | (395,000 | ) | |||||
| Net deferred income tax assets | 17,788,000 | 45,614,000 | ||||||
| Valuation allowance | (17,788,000 | ) | (45,614,000 | ) | ||||
| Deferred tax asset, net of allowance | $ | — | $ | — | ||||
| 60 |
Based on the available objective evidence, including the Company’s history of cumulative losses, management believes it is likely that the Company’s U.S. federal and state net deferred tax assets will not be realizable. Accordingly, the Company provided for a full valuation allowance against its U.S. federal and state net deferred tax assets at December 31, 2023 and December 31, 2022.
Due to the full valuation allowance already in place on the Company’s U.S. federal and state net deferred tax assets, the Company does not anticipate significant changes in the Company’s effective tax rate. However, there is no valuation allowance recorded against the Company’s foreign net operating loss deferred tax assets, as the Company’s foreign IPR&D deferred tax liabilities and foreign net operating loss deferred tax assets are both indefinite-lived and thus they may be netted to arrive at a net foreign deferred tax liability.
The Tax Cuts and Jobs Act resulted in significant changes to the treatment of research or experimental (“R&E”) expenditures under Section 174. For tax years beginning after December 31, 2021, taxpayers are required to capitalize and amortize all R&E expenditures that are paid or incurred in connection with their trade or business which represent costs in the experimental or laboratory sense. Specifically, costs for U.S. based R&E activities must be amortized over five years and costs for foreign R&E activities must be amortized over 15 years; both using a midyear convention. The Company has incorporated the impact of this new tax legislation into its 2023 and 2022 consolidated financial statements, noting that the impact on the Company’s consolidated financial statements was immaterial.
At December 31, 2023, the Company has U.S. federal and state net operating loss carryforwards of approximately $15,942,000 and $78,693,000, respectively, which are available to offset future taxable income. U.S. federal and state net operating loss carryovers began to expire in 2020. As a result of the May 2020 reverse recapitalization, an ownership change has occurred. The Company has not completed an Internal Revenue Code Section 382 analysis. As a result, there could be substantial limitations on the Company’s ability to utilize its pre-ownership change net operating loss and tax credit carryforwards. These substantial limitations may result in both a permanent loss of certain tax benefits related to net operating loss carryforwards and federal research and development credits, and an annual utilization limitation.
The Company also has research and development credit carryforwards for federal and state tax purposes of approximately $3,560,000 and $1,410,000, respectively. The research and development credit carryforwards began to expire in 2020 for federal tax purposes and have an indefinite life for state tax purposes.
U.S. income tax has not been recognized on the excess of the amount for financial reporting over the tax basis of investments in foreign subsidiaries that is indefinitely reinvested outside the United States. This amount becomes taxable upon a repatriation of assets from the subsidiary or a sale or liquidation of the subsidiary. Determination of the amount of any unrecognized deferred income tax liability on this temporary difference is not practicable because of the complexities of the hypothetical calculation.
The Company files income tax returns in the U.S. federal jurisdiction and in various states. The Company’s U.S. federal income tax returns remain subject to examination by the Internal Revenue Service. The Company’s California income tax returns remain subject to examination by the California Franchise Tax Board. Due to net operating losses, research and development credits and other tax credit carryforwards that may be utilized in future years, all U.S. federal and state tax years are open to examination.
Generally accepted accounting principles clarify the accounting for uncertainty in income taxes recognized in the Company’s financial statements and prescribe thresholds for financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return, and also provide guidance on de-recognition and measurement of a tax position taken or expected to be taken in a tax return. The Company adopted these provisions effective April 1, 2009.
The Company did not have any unrecognized tax benefits as of December 31, 2023 and December 31, 2022 and does not expect this to change significantly over the next 12 months. In accordance with generally accepted accounting principles, the Company will recognize interest and penalties accrued on any unrecognized tax benefits as a component of income tax expense. As of December 31, 2023, the Company has not accrued any interest or penalties related to uncertain tax positions.
NOTE 15 — SUBSEQUENT EVENTS
On February 26, 2024, the Company entered into a Securities Purchase Agreement (“Agreement”) with Alpha. The transactions contemplated by the Agreement closed on February 27, 2024, at which time the Company delivered to Alpha the 2024 Debenture and the 2024 Warrant, as described below, and Alpha paid the Company a cash purchase price of $500,000 (less $25,000 for expense reimbursement). Pursuant to the Agreement, the Company issued to Alpha an 8% Convertible Debenture (the “2024 Debenture”) in the principal amount of $550,000. The Debenture has a maturity date of December 31, 2024 and is convertible, at any time, and from time to time, at Alpha’s option, into shares of common stock of the Company, at $0.6111 per share, subject to adjustment as described in the 2024 Debenture (the “Conversion Price”). The Debenture accrues interest on its outstanding principal balance at the rate of 8% per annum. The 2024 Debenture does not call for scheduled payments of principal or interest before the scheduled maturity date of December 29, 2015.31, 2024. Pursuant to the terms of the Master Service Agreement, Covance (or onethe Company also issued to Alpha a 5-year common stock purchase warrant (the “2024 Warrant”) to purchase (at $0.26 per share) 900,016 shares of common stock of the Company. Under the Agreement, Alpha also has an option, exercisable until July 1, 2024, to purchase from the Company up to an additional $1,100,000 in principal amount of 2024 Debentures of like tenor, together with up to an additional 2024 Warrants of like tenor, which would (if and when Alpha exercises such option) provide us up to an additional $1.0 million in cash proceeds (less expense reimbursement, and not including any possible cash proceeds from any future exercise of the additional 2024 Warrants). We granted Alpha “piggyback” registration rights for the common shares underlying the 2024 Debenture and the 2024 Warrant.
| 61 |
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
Not applicable.
Item 9A. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our principal executive officer and principal financial officer, evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2023, the end of the year covered by this Annual Report. Based on this evaluation, our principal executive officer and principal financial officer concluded that, as of December 31, 2023, our disclosure controls and procedures were effective. We believe that a disclosure controls system, no matter how well designed and operated, cannot provide absolute assurance that the objectives of the disclosure controls system are met, and no evaluation of disclosure controls can provide absolute assurance that all disclosure control issues, if any, within a company have been detected.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act. Internal control over financial reporting is a process designed under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of consolidated financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
As of December 31, 2023, our management assessed the effectiveness of our internal control over financial reporting using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework. Based on the continuing material weakness described below, our management concluded that as of December 31, 2023, our internal control over financial reporting was not effective.
Description of Material Weaknesses
In connection with the audit of our financial statements as of and for the year ended December 31, 2023, we identified material weaknesses related to a lack of sufficient number of personnel within our accounting function to adequately segregate duties, and we have not designed and implemented effective Information Technology General Controls (“ITGC”) related to access controls to financial accounting systems.
We lack the resources to employ additional personnel to help mitigate these material weaknesses and we foresee that these material weaknesses will not be remediated until we receive additional funding to support our accounting department.
Remediation of Material Weakness
We cannot assure you that these or moreother measures will fully remediate the material weakness in a timely manner. Notwithstanding the identified material weakness, our management believes that the consolidated financial statements included in this report fairly represent in all material respects our financial condition, results of operations and cash flows at and for the periods presented in accordance with U.S. GAAP.
| 62 |
Changes in Internal Control over Financial Reporting
Other than as described above, there were no changes in our internal control over financial reporting identified in management’s evaluation pursuant to Rules 13a-15(d) or 15d-15(d) of the Exchange Act during the year ended December 31, 2023 that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Limitation on Effectiveness of Controls
In designing and evaluating our controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable and not absolute assurance of achieving the desired control objectives. No evaluation of internal control can provide absolute assurance that all internal control issues and instances of fraud, if any, within a company are detected. In reaching a reasonable level of assurance, management necessarily was required to apply its affiliates) will provide Phase 1, 2, 3,judgment in evaluating the cost-benefit relationship of possible controls and 4 clinical services for a clinical studyprocedures. There are inherent limitations to the effectiveness of any system of disclosure controls and procedures, including the possibility of human error and the circumvention or studies to us,overriding of the controls and at our request, assist us withprocedures. In addition, the design of such studies,any system of controls is based in accordancepart upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Over time, controls may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Item 9B. Other Information.
None.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
None.
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
The Board of Directors
Our board of directors currently consists of six members, each of whose current term of office as a director expires at the 2024 annual meeting of stockholders. Biographical information with respect to our directors is provided below.
Our directors hold office for one year or until their respective successors have been duly elected or until their death, resignation or removal. Our amended and restated bylaws provide that the authorized number of directors comprising our board of directors will be fixed, from time to time, by a majority of the total number of directors.
There are no family relationships among any of our directors or executive officers. There is no arrangement or understanding between any director and any other person pursuant to which the director was selected.
| Name | Position with the Company | Age | Director Since | |||||||
| Michael Poirier | Chairman and Chief Executive Officer | 68 | 2020 | |||||||
| Richard David | Director | 64 | 2020 | |||||||
| Sidney Emery, Jr. | Director | 77 | 2020 | |||||||
| Matthew Korenberg | Director | 49 | 2020 | |||||||
| Kurt Kruger | Director | 68 | 2020 | |||||||
| Ira Ritter | Director | 75 | 2008 | |||||||
Michael S. Poirier. Mr. Poirier founded the Qualigen business in 1996 and is its Chairman and Chief Executive Officer. Before founding Qualigen, Mr. Poirier had relevant operating, marketing and sales positions with Ashirus Technologies, Inc., EnSys, Inc., Sanofi Pasteur and Abbott Laboratories, Inc. Before working at Abbott, Mr. Poirier served as an officer in the United States Navy, assigned to the US Atlantic Fleet. Mr. Poirier holds a B.A. from Providence College and attended the University of Zürich, Switzerland, School of Law.
Mr. Poirier’s commitment to our strategic goals, his long experience leading our company and his deep knowledge of its technologies and business contributed to our board of directors’ conclusion that he should serve as a director of our company.
Richard A. David, MD FACS. Dr. David serves as Chief Medical Officer for the Los Angeles Division of Genesis Healthcare Partners, the largest urology group in Southern California. He also serves as medical director for Genesis’ Advanced Prostate Cancer Center of Excellence. In addition, Dr. David serves as Clinical Professor of Urology for the David Geffen School of Medicine at UCLA. Dr. David obtained his undergraduate education at Stanford University and his medical degree at Thomas Jefferson University in Philadelphia. He also holds a Master’s degree in Medical Management (MMM) from the Marshall School of Business at the University of Southern California. He trained in general surgery and completed his urology residency at UCLA Medical Center in Los Angeles. Dr. David is a fellow of the American College of Surgeons.
Dr. David’s experience as an executive of a large healthcare organization, including his background as a medical doctor, contributed to our board of directors’ conclusion that he should serve as a director of our company.
Sidney W. Emery, Jr. Mr. Emery is a seasoned executive in manufacturing, distribution and supply chain management. He served as Chairman and Chief Executive Officer of MTS Systems Corporation (Nasdaq-GS: MTSC), a leading global supplier of mechanical testing systems and high-performance industrial position sensors. Mr. Emery served on the Board of Directors of Allete, Inc. (NYSE: ALE), a Minnesota-based utilities and energy company, from 2006 to 2018. Mr. Emery chairs the University of St. Thomas College of Engineering Board of Governors. Mr. Emery holds a PhD in Industrial Engineering from Stanford University and a B.S. in Engineering from the US Naval Academy.
Mr. Emery’s extensive board service with and executive leadership of major companies contributed to our board of directors’ conclusion that he should serve as a director of our company.
Matthew E. Korenberg. Mr. Korenberg has served as President and Chief Operating Officer of Ligand Pharmaceuticals Incorporated (Nasdaq: LGND), a biopharmaceutical company focused on developing or acquiring technologies that help pharmaceutical companies discover and develop medicines, since November 2022, and before that as Executive Vice President, Finance and Chief Financial Officer of Ligand Pharmaceuticals Incorporated since August 2015. Before joining Ligand, commencing in September 2013, Mr. Korenberg was the founder, Chief Executive Officer and a director of NeuroCircuit Therapeutics, a company focused on developing drugs to treat genetic disorders of the brain with an initial focus on Down syndrome. Before founding NeuroCircuit Therapeutics, Mr. Korenberg was a Managing Director and member of the healthcare investment banking team at Goldman Sachs from July 1999 through August 2013. During his 14 year tenure at Goldman Sachs, Mr. Korenberg was focused on advising and financing companies in the biotechnology and pharmaceutical sectors and was based in New York, London and San Francisco. Before Goldman Sachs, Mr. Korenberg was a healthcare investment banker at Dillon, Read & Co. Inc. where he spent two years working with healthcare companies in the biotechnology and pharmaceutical sectors and industrial companies. Mr. Korenberg holds a B.B.A. in Finance and Accounting from the University of Michigan.
| 64 |
Mr. Korenberg’s financial and accounting expertise, his experience as chief financial officer of a large public biopharmaceutical company and his investment banking background contributed to our board of directors’ conclusion that he should serve as a director of our company.
Kurt H. Kruger. Mr. Kruger has enjoyed a 30-year career in medical technology. His deep involvement in the field has ranged from product design and development as a biomedical engineer to raising capital for, and following, publicly traded medical product companies as an equities research analyst. As a marketing manager at Guidant, now a part of Boston Scientific, he developed the launch plans for the first-ever implantable defibrillator. As a securities analyst he led Hambrecht & Quist’s efforts in providing venture funds for, and then taking public, Ventritex, which was later acquired by St. Jude Medical/Abbott. After H&Q, Mr. Kruger worked as an analyst for Montgomery Securities and Bank of America. Across 20 years of research work, Mr. Kruger has overseen the IPOs of over 30 medical products companies. Later he headed up the Life Sciences banking effort for WR Hambrecht & Co. Mr. Kruger received a Sc.B. degree in Biomedical Engineering from Brown University; a Master’s degree in Bioengineering from the University of Michigan; and a business degree (S.M.) from the Sloan School at the Massachusetts Institute of Technology (MIT). He also completed the premedical post-baccalaureate program at Columbia University.
Mr. Kruger’s long experience in investment banking and securities analysis with a life sciences focus contributed to our board of directors’ conclusion that he should serve as a director of our company.
Ira E. Ritter. Mr. Ritter served as Co-Founder, Chief Strategic Officer and Executive Chairman of the Company during its Ritter Pharmaceuticals, Inc. phase, from its inception in 2004 through the formation of the Ritter Pharmaceuticals, Inc. corporate entity in 2008 and served in those positions with Ritter Pharmaceuticals, Inc. from 2008 until the May 22, 2020 reverse recapitalization transaction (the “Reverse Recapitalization Transaction”) in which Ritter Pharmaceuticals, Inc. changed its name to Qualigen Therapeutics, Inc. Mr. Ritter has extensive experience creating and building diverse business enterprises and since 1987 through Andela Corporation, of which he is the CEO, has provided corporate management, strategic planning and financial consulting for a wide range of market segments including; health product related national distribution and private label production, television and publishing. He assisted taking Ritter Pharmaceuticals, Inc. public on Nasdaq and Martin Lawrence Art Galleries public on the New York Stock Exchange. Since 2010, Mr. Ritter has also acted as a managing partner of Stonehenge Partners, LLC. Mr. Ritter has a long history of public service that includes appointments by three Governors to several State of California Commissions including eight years as Commissioner on the California Prison Industry Authority.
Mr. Ritter’s experience as an entrepreneur and chairman of a publicly traded development-phase therapeutics company contributed to our board of directors’ conclusion that he should serve as a director of our company. Mr. Ritter continued his service on our board of directors, by agreement in connection with the termsReverse Recapitalization Transaction, as the designated legacy member from the pre-Reverse Recapitalization Transaction public-company board of separate individual project agreementsdirectors.
Committees of the Board of Directors
Our board of directors has established an Audit Committee, a Compensation Committee and a Nominating and Corporate Governance Committee. Each committee operates under a charter. Copies of each committee’s charter are posted on the Investor Relations section of our website, which is located at www.qlgntx.com.
Audit Committee. The current members of our Audit Committee are Mr. Kruger (Chair), Mr. Emery, and Mr. Korenberg, each of whom was determined by our board of directors to be entered intoindependent under Rule 10A-3 under the Exchange Act and the continued listing requirements of Nasdaq, and to satisfy the other continued listing requirements of Nasdaq for audit committee membership. The Company has identified Matthew Korenberg as an “audit committee financial expert” as such term is defined in Item 407(d)(5) of SEC Regulation S-K, and has determined that he has the requisite level of financial sophistication required by the parties. The termcontinued listing requirements of Nasdaq; this identification does not constitute a determination that other members of the agreementAudit Committee would not also be able to qualify as an “audit committee financial expert.”
| 65 |
EXECUTIVE OFFICERS
The following table sets forth information about our current executive officers.
| Name | Age | Position with the Company | ||||
| Michael Poirier | 68 | Chairman and Chief Executive Officer | ||||
| Christopher Lotz | 59 | Chief Financial Officer, Vice President of Finance | ||||
Officers serve at the discretion of the board of directors. There are no family relationships among any of our directors or executive officers. There is no arrangement or understanding between any executive officer and any other person pursuant to which the executive officer was selected.
For the biography of Mr. Poirier, please see “Board of Directors” above.
Christopher L. Lotz | Chief Financial Officer, Vice President of Finance. Mr. Lotz joined Qualigen, Inc. as Director of Finance in 2002 and was promoted to his current role of Chief Financial Officer, Vice President of Finance in 2003. He became an officer of the Company at the time of the Reverse Recapitalization Transaction in 2020. Before joining Qualigen, Inc. Mr. Lotz spent the previous 15 years serving in financial leadership positions with Bexcom, an Asian-based software developer, California Furniture Collections, Inc., a custom furniture manufacturer, and Group Publishing, Inc., an educational media publisher. Mr. Lotz holds a B.S. in Business Administration from Colorado State University.
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange Act requires the Company’s officers and directors, and persons who own more than 10% of our common stock, to file reports of securities ownership and changes in such ownership with the SEC. Officers, directors, and greater than 10% stockholders also are required by SEC rules to furnish the Company with copies of all Section 16(a) forms they file.
Based solely on the Company’s review of Forms 3, 4 and 5 filed by such persons and information provided by the Company’s directors and officers, the Company believes that during the year ended December 31, 2023, all Section 16(a) filing requirements applicable to such persons were met in a timely manner.
Item 11. Executive Compensation.
Summary Compensation Table (2023 and 2022)
The following table sets forth the compensation paid to or earned by our named executive officers for three yearsthe periods presented.
| Name and Principal Position | “Year” | Salary ($) | Bonus ($) | Option Awards(1) ($) | All Other Compensation(2) ($) | Total ($) | ||||||||||||||||||
| Michael Poirier, Chairman and Chief Executive Officer | 2023 | 512,635 | 118,174 | — | 1,889 | 632,698 | ||||||||||||||||||
| 2022 | 575,000 | — | 145,274 | 8,180 | 728,454 | |||||||||||||||||||
| Tariq Arshad, Former Chief Medical Officer and Senior Vice President (3) | 2023 | 356,615 | 101,231 | — | 5,326 | 463,172 | ||||||||||||||||||
| 2022 | 400,000 | — | 39,512 | 138 | 439,650 | |||||||||||||||||||
Amy Broidrick, Former President, Chief Strategy and Operating Officer(4) | 2023 | 194,017 | — | — | 371,454 | 565,470 | ||||||||||||||||||
| 2022 | 450,000 | — | 50,359 | 7,642 | 508,001 | |||||||||||||||||||
| (1) | The amounts reported in this column reflect the aggregate grant date fair value of the option awards granted during 2022, computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718 for stock-based compensation transactions (“ASC 718”). Such grant date fair values do not take into account any estimated forfeitures related to service-based vesting conditions. Assumptions used in the calculation of these amounts are included in the notes to our consolidated financial statements included in this Annual Report. These amounts do not reflect the actual economic value that may be realized by the executive officers upon the exercise of the stock options or the sale of the common stock underlying such stock options. There were no option awards granted during 2023. | |
| (2) | The amounts reported in this column represent 401(k) matching contributions and life insurance premiums paid by us for Mr. Poirier and Dr. Arshad, and $1,316 in 401(k) matching contributions and life insurance premiums paid by us for Ms. Broidrick, and $370,138 in severance compensation for Ms. Broidrick. | |
| (3) | Dr. Arshad resigned from his role as Chief Medical Officer and Senior Vice President effective February 25, 2024. | |
| (4) | Ms. Broidrick resigned from her role as President, Chief Strategy and Operating Officer and as a Director effective June 16, 2023. The 2023 “Salary” for Ms. Broidrick includes amounts we paid through June 16, 2023. The 2023 “All Other Compensation” for Ms. Broidrick includes $370,138 of severance compensation. |
| 66 |
Executive Employment Agreements
Employment Agreement with Michael Poirier
Mr. Poirier is party to an Executive Employment Agreement dated February 1, 2017, as amended January 9, 2018 (the “Poirier Employment Agreement”). The Poirier Employment Agreement had an initial three-year term and will renewis now automatically renewed for successive one yearone-year periods unless Covance is no longer providing services under the agreement or either party has terminatedgives notice of nonrenewal at least 90 days before the agreement upon written notice. We may terminate the Master Service Agreement or any individual project agreement entered into under the Master Service Agreement prior to the applicable study’s completion at any time for any reason upon 30 days written notice to Covance, except when the reason for termination is the safetyend of subjects, in which case it may be terminated immediately. Covance may not terminate any individual project agreement without cause, except when the reason for the termination is the safety of subjects, in which case it may be terminated immediately. In the event ofsuch a termination of the Master Service Agreement, Covance will be entitled to full payment for (i) work performed on the applicable project upon through the date work on such project is concluded and (ii) reimbursement for all non-cancellable and non-refundable expenses and financial obligations which Covance (or an affiliate) has incurred or undertaken on our behalf.one-year period.
Clinical Supply and Cooperation Agreement with Ricerche Sperimentali Montale (“Ricerche”) and Inalco SpA (“Inalco”)
Effective July 24, 2015, we entered into an amended Clinical Supply and Cooperation Agreement (the “Amended Supply Agreement”) with Ricerche and Inalco (collectively, “RSM”). The Amended Supply Agreement amends certain terms of the Clinical Supply and Cooperation Agreement, dated December 16, 2009, amended on September 25, 2010 (the “Existing Supply Agreement”).
Under the Existing Supply Agreement, RSM granted us an exclusive worldwide option in a specified field and territory to assignment of all right, title and interest to a purified galacto-oligosaccharides product (“Improved GOS”), the composition of matter of the Improved GOS and any information relating to the Improved GOS, including certain specified technical information and other intellectual property rights (the “Improved GOS IP”). Pursuant to the amended terms, we could exercise the option by paying RSM $800,000 within ten days after the effective date of the Amended Supply Agreement. We exercised this option on July 30, 2015 and RSM transferred the Improved GOS IP to us. Under the terms of the Existing SupplyPoirier Employment Agreement, if a further option paymentMr. Poirier is entitled to an annual base salary of $1 million dueat least $315,000, is eligible to participate in the futureCompany’s bonus plans, benefit programs and medical benefits, is not made, we may be requiredeligible for certain event-based bonuses (including for “Liquidity Event” acquisition transactions), and is entitled to return the Improved GOS IP to RSM.
The Amended Supply Agreement also provides that we must pay RSM $400,000 within 10 days following FDA approvalfour weeks of a new drug applicationvacation per year. If Mr. Poirier’s employment is terminated without Cause or he resigns for the first product owned or controlled by us using Improved GOS as its active pharmaceutical ingredient.
In consideration for RSM entering into the Amended Supply Agreement, we issued 100,000 shares of our common stock, par value $0.001 per share (the “RSM Shares”), to RSM on November 30, 2015. Fair value of these shares totaling $416,000 was recognized in stockholders’ equityGood Reason (as such terms are defined in the balance sheetPoirier Employment Agreement), and he provides a general release to the Company, he will be entitled to one year of salary continuation plus the cost of COBRA coverage continuation for such one year period. In May 2021, our board of directors and its compensation committee increased Mr. Poirier’s annual base salary to $575,000. On January 13, 2023, the Company’s board of directors, as part of certain cost-cutting measures, approved a temporary 20% reduction to the base salaries of all executive officers of the Company. Accordingly, on January 16, 2023, Mr. Poirier’s base salary was reduced to $460,000; it was subsequently restored in August 2023.
Employment Agreement with Christopher Lotz
Mr. Lotz is party to an Executive Employment Agreement dated February 1, 2017, as amended January 9, 2018 (the “Lotz Employment Agreement”). The Lotz Employment Agreement had an initial three-year term and is now automatically renewed for successive one-year periods unless either party gives notice of nonrenewal at least 90 days before the end of such a one-year period.
Under the terms of the Lotz Employment Agreement, Mr. Lotz is entitled to an annual base salary of at least $225,000, is eligible to participate in the Company’s bonus plans, benefit programs and medical benefits, is eligible for certain event-based bonuses (including for “Liquidity Event” acquisition transactions), and is entitled to four weeks of vacation per year. If Mr. Lotz’s employment is terminated without Cause or he resigns for Good Reason (as such terms are defined in the Lotz Employment Agreement), and he provides a general release to the Company, he will be entitled to 180 days of salary continuation plus the cost of COBRA coverage continuation for such 180 day period. In May 2021, our board of directors and its compensation committee increased Mr. Lotz’s annual base salary to $300,000. On January 13, 2023, the Company’s board of directors, as part of certain cost-cutting measures, approved a temporary 20% reduction to the base salaries of all executive officers of the Company. Accordingly, on January 16, 2023, Mr. Lotz’s base salary was reduced to $240,000; it was subsequently restored in August 2023.
Offer Letter with Tariq Arshad
Under the terms of his hire offer letter with the Company, dated May 17, 2021, Dr. Arshad was entitled to an annual base salary of at least $400,000. He received a cash signing bonus of $25,000 when he joined the Company, was eligible to receive annual cash bonuses equal to an amount up to 40% of his annualized base salary, and is entitled to four weeks of vacation per year. Under the terms of his hire offer letter, if Dr. Arshad’s employment is terminated without Cause or he resigns for Good Reason, and he provides a general release to the Company, he will be entitled to 180 days of salary continuation plus the cost of COBRA coverage continuation for such 180 day period.
On February 25, 2024, Dr. Arshad resigned from his position as the Company’s Chief Medical Officer and Senior Vice President. He did not assert that the resignation was for Good Reason and he did not provide a general release to the Company.
Employment Agreement with Amy Broidrick
Ms. Broidrick was party to an Executive Employment Agreement with Qualigen, Inc., a former wholly-owned subsidiary of the Company, dated December 31, 2015.
Lease Agreement
10, 2021. On July 9, 2015, weMay 16, 2023, Ms. Broidrick resigned from all officer and director positions with the Company and its subsidiaries, which became effective June 16, 2023 (the “Separation Date”). Ms. Broidrick’s departure was not related to any disagreement with the Company on any matter relating to the Company’s operations, policies, or practices. In connection with her termination of employment, on June 20, 2023, Qualigen, Inc, entered into a leaseseparation agreement and general release with Century Park,Ms. Broidrick, which became effective after a California limited partnership, pursuant7 day revocation period following Ms. Broidrick’s signing of it, on June 24, 2023. Under the terms of the Separation Agreement, Qualigen, Inc. was obligated to which we are leasing approximately 2,780 square feetprovide Ms. Broidrick severance in the form of office spacecontinued salary pay at the rate then in Los Angeles, California for our headquarters. The lease provideseffect on the Separation Date ($360,000 per annum) for a termperiod of sixty-one (61)12 months commencing on October 1, 2015. We paid no rentfollowing the Separation Date, subject to applicable withholding, and payment or reimbursement for the first monthcost of COBRA continuation medical and dental insurance coverage for 12 months following the Separation Date, less any required taxes or withholdings. Upon the July 20, 2023 closing of our sale of Qualigen, Inc., Chembio (as the new parent company of Qualigen, Inc.) undertook the remaining severance liability to Ms. Broidrick.
Stock Incentive Plan
The material terms of our 2020 Stock Equity Incentive Plan (as amended, the “2020 Plan”) are outlined below. This summary is qualified in its entirety by reference to the complete text of the term and will pay base rent of $9,174 per month for months 2 through 13 of the term, with increasing base rent for each twelve month period thereafter under the term of the lease to a maximum of $10,325 per month for months 50 through 61. The base rent payments do not include our proportionate share of any operating expenses, including real estate taxes. We have the option to extend the term of the lease for one five-year term, provided that the rent would be subject to market adjustment at the beginning of the renewal term. We will recognize rent expense on a straight-line basis over the lease term.2020 Plan, which is incorporated herein by reference.
Rent expense, recognized on a straight-line basis, was approximately $115,000 and $115,000 for the years ended December 31, 2017 and 2016, respectively, and is recorded in general and administrative expenses in the accompanying statements of operations.
The following table summarizes our lease obligations at December 31, 2017:
| LEASE COMMITMENTS | ||||
| Years ended December 31, | Operating Lease | |||
| 2018 | $ | 117,376 | ||
| 2019 | 120,898 | |||
| 2020 | 103,254 | |||
| Total minimum lease payments | $ | 341,528 | ||
Legal
The Company is not currently involved in any legal matters arising in the normal course of business. From time to time, the Company could become involved in disputes and various litigation matters that arise in the normal course of business. These may include disputes and lawsuits related to intellectual property, licensing, contract law and employee relations matters. Periodically, the Company reviews the status of significant matters, if any exist, and assesses its potential financial exposure. If the potential loss from any claim or legal claim is considered probable and the amount can be estimated, the Company accrues a liability for the estimated loss. Legal proceedings are subject to uncertainties, and the outcomes are difficult to predict. Because of such uncertainties, accruals are based on the best information available at the time. As additional information becomes available, the Company reassesses the potential liability related to pending claims and litigation.
NOTE 6— STOCKHOLDERS’ EQUITY
Authorized Shares
On September 15, 2017, the Company amended its Amended and Restated Certificate. We have reserved an aggregate of Incorporation to authorize the issuance of up to 225,000,000755,702 shares of our common stock $0.001 par value per share, and 15,000,000for issuance under the 2020 Plan. The number of shares of preferred stock, $0.001 par value per share.
As of December 31, 2017, the Company had 49,406,521 shares of common stock and 9,140 shares of Series A convertible preferred stock issued and outstanding. Each share of the Company’s common stock is entitled to one vote, and all shares rank equally as to voting and other matters. Each share of Series A preferred stock is convertible by the holder at $0.40 per share; subject to adjustment forin the event of any recapitalization, stock splits,split, reclassification, stock dividends, subsequent rights offerings, pro rata distributions, and fundamental transactions. Holders are entitled to receive, anddividend or other change in our capitalization. In addition, the Company shall pay, dividends on outstandingfollowing shares of Series A preferredour common stock on an as-if-converted-to-common-stock basis, equalwill be available for grant and issuance under the 2020 Plan:
| ● | shares subject to stock options or stock appreciation rights (“SARs”), granted under the 2020 Plan that cease to be subject to the stock option or SAR for any reason other than exercise of the stock option or SAR; | |
| ● | shares subject to awards granted under the 2020 Plan that are subsequently forfeited or repurchased by us at the original issue price; |
| ● | shares subject to awards granted under the 2020 Plan that otherwise terminate without shares being issued; | |
| ● | shares surrendered, canceled, or exchanged for cash or a different award (or combination thereof); and | |
| ● | shares subject to awards under the 2020 Plan that are used to pay the exercise price of an award or withheld to satisfy the tax withholding obligations related to any award. |
Plan Administration. The 2020 Plan will be administered by our Compensation Committee or by our board of directors acting in place of our Compensation Committee. Our Compensation Committee will have the authority to construe and ininterpret the same form as dividends actually paid on outstanding common shares when, as2020 Plan, grant awards and if such dividends are paid on outstanding common shares. Upon any liquidation, dissolutionmake all other determinations necessary or winding-upadvisable for the administration of the Company, whether voluntary or involuntary,2020 Plan.
Awards and Eligible Participants. The 2020 Plan authorizes the holdersaward of Series A preferred stock shalloptions, stock appreciation rights, restricted stock unit, performance awards and stock bonuses. The 2020 Plan provides for the grant of awards to our employees, directors, consultants and independent contractor service providers, subject to certain exceptions. No non-employee director may be entitledgranted awards under the 2020 Plan in any calendar year that, taken together with any cash fees paid by us to receive outsuch non-employee director during such calendar year, exceed $5,000,000 (calculating the value of any award based on the assets, whether capital or surplus,grant date fair value determined in accordance with GAAP). No more than 98,000,000 shares of the Company the same amount that a holder ofour common stock would receive if the Series A preferred stock were fully converted to common stock, which amounts shallwill be paid pari passu with all common shareholders. Holders of Series A preferred stock have no voting rights. However, as long as any shares of Series A preferred stock are outstanding, the Company shall not, without the affirmative vote of the holders of a majority of the then outstanding shares of Series A preferred stock, (a) alter or change adversely the powers, preferences or rights given to the Series A preferred stock or alter or amend the applicable Certificate of Designation, (b) amend the Company’s certificate of incorporation or other charter documents in any manner that adversely affects any rights of the holders of Series A preferred stock, (c) increase the number of authorized shares of Series A preferred stock, or (d) enter into any agreement with respect to any of the foregoing.
Aspire Capital Financing Arrangement
On December 18, 2015, the Company entered into a common stock purchase agreement (the “2015 Aspire Purchase Agreement”) with Aspire Capital Fund, LLC (“Aspire Capital”), pursuant to which Aspire Capital was committed to purchase up to an aggregate of $10.0 million of the Company’s shares of common stock over the approximate 30-month term of the 2015 Aspire Purchase Agreement. As of December 31, 2017, the Company had issued an aggregate of 4,577,699 shares of its common stock to Aspire Capital under the 2015 Aspire Purchase Agreement for approximate proceeds of $5.0 million.
On May 4, 2017, the Company terminated the 2015 Aspire Purchase Agreement and entered into a new common stock purchase agreement with Aspire Capital (the “2017 Aspire Purchase Agreement”), which provides that upon the terms and conditions set forth therein, Aspire Capital is committed to purchase up to an aggregate of $6.5 million of shares of the Company’s common stock over the 30-month term of the 2017 Aspire Purchase Agreement. On any trading day on which the closing sale price of the Company’s common stock exceeds $0.25, the Company has the right, in its sole discretion, to present Aspire Capital with a purchase notice, directing Aspire Capital (as principal) to purchase up to 100,000 shares of the Company’s common stock per trading day, for up to $6.5 million of the Company’s common stock in the aggregate at a per share price, calculated by reference to the prevailing market price of the Company’s common stock (as provided in the 2017 Aspire Purchase Agreement).
As a condition to the 2017 Aspire Purchase Agreement, the Company issued 137,324 shares of its common stock to Aspire Capital as a commitment fee. As of the date of this Annual Report, no shares of common stock have been sold to Aspire Capital under the 2017 Aspire Purchase Agreement.
October 2016 Public Offering
On October 31, 2016, the Company closed a public offering, selling 2,127,660 shares of the Company’s common stock at a price to the public of $2.35 per share, for aggregate gross proceeds to the Company of approximately $5.0 million. The Company paid to the underwriters underwriting discounts and commissions of approximately $0.4 million in connection with the offering, and approximately $0.2 million of other expenses in connection with the offering.
This offering was made pursuant to a shelf registration statement on Form S-3, which was declared effective by the SEC on August 23, 2016. The shelf registration statement allows the Company to issue, from time to time at prices and on terms to be determined at or prior to the time of an offering, up to $150,000,000 of any combination of an indeterminate number of shares of common stock, an indeterminate number of shares of preferred stock, an indeterminate principal amount of debt securities, an indeterminate number of warrants, rights and purchase contracts to purchase common stock or debt securities, and an indeterminate number of units. If any debt securities are issued at an original issue discount, then the offering price of such debt securities shall be in such greater principal amount as shall result in an aggregate offering price not to exceed $150,000,000, less the aggregate dollar amount of all securities previously issued hereunder. The securities registered also include such indeterminate number of shares of common stock and preferred stock that may be issued upon conversion or exchange of convertible or exchangeable securities being registered or2020 Plan pursuant to the anti-dilution provisionsexercise of any such securities.incentive stock options.
October 2017 Public Offering
On October 3, 2017, the Company closed a public offering, selling an aggregate of (i) 34,550,000 Class A Units consisting of 34,550,000 shares of the Company’s common stock and warrants to purchase 34,550,000 shares of the Company’s common stock at a public offering price of $0.40 per unit, and (ii) 9,180 Class B Units consisting of 9,180 shares of Series A Convertible Preferred Stock with a stated value of $1,000 per unit, and convertible into an aggregate of 22,950,000 shares of the Company’s common stock, and warrants to purchase an aggregate of 22,950,000 shares of the Company’s common stock. The securities were offered by the Company pursuant to a registration statement filed with the SEC that was declared effective on September 28, 2017. The final prospectus relating to the offering was filed with the SEC on October 2, 2017.
The warrants have an exercise price of $0.44, are exercisable upon issuance and expire five years from the date of issuance. The warrant agreements provide for an adjustment to the number of common shares issuable under the warrants and/or adjustment to the exercise price, including but not limited to, if: (a) the Company issues shares of common stock as a dividend or distribution to holders of its common stock; (b) the Company subdivides or combines its common stock; or (c) adjustment of the exercise price upon subsequent equity sales or issuance of new securities by the Company at less than the exercise price.
The Company granted the underwriters a 45-day option to purchase an additional 8,625,000 shares of the Company’s common stock and/or warrants to purchase an additional 8,625,000 shares of the Company’s common stock. As of the closing of the offering, the underwriters have exercised their over-allotment option for warrants to purchase 2,975,000 shares of the Company’s common stock.
Aggregate gross proceeds to the Company from the public offering were approximately $23.0 million. The Company paid underwriting discounts and commissions of approximately $1.6 million in connection with the offering, and approximately $0.4 million of other expenses in connection with the offering.
The Company early adopted the provisions of ASU 2017-11 in recognizing the warrants. As a result, the exercise price reset provisions were excluded from the assessment of whether the warrants are considered indexed to the Company’s own stock. The warrants otherwise meet the requirements for equity classification, as such were initially classified in Stockholders’ Equity. The Company will recognize the value of the exercise price reset provision if and when it becomes triggered, by recognizing the value of the effect of the exercise price reset as a deemed dividend and a reduction of income available to common shareholders in computing basic earnings per share.
The proceeds received in the October 2017 Public Offering were allocated to each instrument on a relative fair value basis. Total proceeds of $23.0 million were allocated to warrants issued, $10.1 million, Common Stock, $7.8 million and Series A Preferred Stock, $5.1 million. The allocation resulted in an effective conversion price for the Series A preferred stock that was below the quoted market price of the Company’s common stock on the closing date. As such, the Company recognized a beneficial conversion feature equal to the intrinsic value of the conversion feature on the closing date, resulting in a deemed dividend for the Series A preferred stock of approximately $3.1 million recognized on the closing date.
NOTE 7 — WARRANTS
Warrants to purchase an aggregate of 61,053,323 shares of the Company’s common stock were outstanding at December 31, 2017. These warrants are all vested and exercisable, have exercise prices ranging from $0.44 to $9.30 per share, with a weighted average exercise price of $0.52, and expire at various dates through October 2022.
NOTE 8 — STOCK-BASED COMPENSATION
Equity Incentive PlansOptions
The Company has issued equity awards pursuant to its 2015 Equity Incentive Plan (the “2015 Plan”), 2009 Stock Plan and 2008 Stock Plan (collectively the “Plans”). The Plans permit the Company2020 Plan permits us to grant non-statutory stock options, incentive stock options and other equity awards to the Company’s employees, outside directors and consultants; however, incentivenon-qualified stock options may only be granted to the Company’s employees. Beginning June 29, 2015, no further awards may be granted under the 2009 Stock Plan or 2008 Stock Plan. However, to the extent awards under the 2008 Plan or 2009 Plan are forfeited or lapse unexercised or are settled in cash, the common stock subject to such awards will be available for future issuance under the 2015 Equity Incentive Plan.
On June 2, 2017, the stockholders of the Company approved an amendment to the 2015 Plan at the 2017 annual meeting of stockholders, which among other things, increased the number of shares that may be issued pursuant to awards under the 2015 Plan by 838,000 shares of common stock.
On September 15, 2017, the stockholders of the Company approved an amendment to the 2015 Plan at a special meeting of stockholders, which among other things, increased the number of shares that may be issued pursuant to awards under the 2015 Plan by 25,858,711 shares of common stock. As of December 31, 2017, the aggregate number of shares of common stock authorized for issuance under the 2015 Plan, as amended, was 27,500,000, and 26,720,193 shares were available for issuance as of December 31, 2017.
The following represents a summary of the options granted to employees and non-employees that are outstanding at December 31, 2017 and changes during the period then ended:
| Number of Shares | Weighted- Average Exercise Price | Aggregate Intrinsic Value | Weighted- Average Remaining Contractual Life (in years) | |||||||||||||
| Outstanding at December 31, 2016 | 2,476,924 | $ | 6.01 | $ | 497,351 | 8.3 | ||||||||||
| Options granted | 88,000 | 2.89 | ― | 7.3 | ||||||||||||
| Options forfeited | (23,124 | ) | 2.52 | ― | ― | |||||||||||
| Outstanding at December 31, 2017 | 2,541,800 | 5.93 | ― | 7.3 | ||||||||||||
| Exercisable at December 31, 2017 | 1,718,370 | $ | 6.02 | $ | ― | 7.0 | ||||||||||
options. The exercise price for an option issued under the Plans isof stock options will be determined by the Board of Directors, but willour Compensation Committee, and may not be (i) in the case of an incentive stock option (A) granted to an employee who, at the time of grant of such option, is a 10% stockholder, no less than 110% of the fair market value per share on the date of grant; or (B) granted to any other employee, no less than 100% of the fair market value per shareof our common stock on the date of grant; and (ii)grant. Our Compensation Committee has the authority to reprice any outstanding stock option (by reducing the exercise price, or canceling the stock option in exchange for cash or another equity award) under the 2020 Plan without the approval of our stockholders. Stock options may vest based on the passage of time or the achievement of performance conditions in the casediscretion of our compensation committee. Our Compensation Committee may provide for stock options to be exercised only as they vest or to be immediately exercisable with any shares issued on exercise being subject to our right of repurchase that lapses as the shares vest. The maximum term of stock options granted under the 2020 Plan is 10 years.
Stock Appreciation Rights. SARs provide for a non-statutorypayment to the holder, in cash or shares of our common stock, option,based upon the difference between the fair market value of our common stock on the date of exercise and the stated exercise price on the date of grant, up to a maximum amount of cash or number of shares. SARs may vest based on the passage of time or the achievement of performance conditions in the discretion of our Compensation Committee. Our Compensation Committee has the authority to reprice any outstanding SAR (by reducing the exercise price, or canceling the SAR in exchange for cash or another equity award) under the 2020 Plan without the approval of our stockholders.
Restricted Stock Awards. A restricted stock award represents the issuance to the holder of shares of our common stock, subject to the forfeiture of those shares in the event of failure to achieve certain performance conditions or termination of employment. The purchase price, if any, for the shares will be determined by our Compensation Committee. Unless otherwise determined by the administrator at the time of award, vesting will cease on the date the holder no longer provides services to us and unvested shares will be forfeited to us or can be repurchased by us.
Restricted Stock Units. Restricted stock units (“RSUs”) represent the right on the part of the holder to receive shares of our common stock at a specified date in the future, subject to forfeiture of that right in the event of failure to achieve certain performance conditions or termination of employment. If a RSU has not been forfeited, then, on the specified date, we will deliver to the holder of the RSU shares of our common stock, cash or a combination of cash and shares of our common stock, as previously determined by the Compensation Committee at the time of the award.
Performance Awards. Performance awards cover a number of shares of our common stock that may be settled upon achievement of performance conditions as provided in the 2020 Plan in cash or by issuance of the underlying common stock. These awards are subject to forfeiture before settlement in the event of failure to achieve certain performance conditions or termination of employment.
Stock Bonuses. Stock bonuses may be granted as additional compensation for past or future service or performance and, therefore, no payment will be required from a participant for any shares awarded under a stock bonus. Unless otherwise determined by our Compensation Committee at the time of award, vesting will cease on the date the holder no longer provides services to us and unvested shares will be forfeited to us.
| 68 |
Change-in-Control. If we are party to a merger or consolidation, sale of all or substantially all our assets or similar change-in-control transaction, outstanding awards, including any vesting provisions, may be assumed or substituted by the successor company. In the alternative, the successor company may issue, in place of outstanding shares held by a 2020 Plan participant, substantially similar shares or other property subject to repurchase obligations no less than 100%favorable to the participant. Outstanding awards that are not assumed, substituted or cashed out will accelerate in full and expire immediately before the transaction, and awards will be exercisable for a period of time determined by the administrator.
Amendment; Termination. The 2020 Plan will terminate 10 years from April 8, 2020, unless it is terminated earlier by our board of directors. Our board of directors may amend, suspend or terminate the 2020 Plan at any time, subject to compliance with applicable law.
Federal Income Tax Summary. The following is a brief summary of the principal federal income tax consequences to us and to an eligible person (who is a citizen or resident of the United States for U.S. federal income tax purposes) (a “Participant”) of awards that may be granted under the 2020 Plan. The summary is not intended to be exhaustive and, among other things, does not describe state, local or foreign tax consequences. The federal income tax consequences of an eligible person’s award under the 2020 Plan are complex, are subject to change and differ from person to person. Each person should consult with his or her own tax adviser as to his or her own particular situation.
This discussion is based on the Code, Treasury Regulations promulgated under the Code, Internal Revenue Service rulings, judicial decisions and administrative rulings as of the date of this proxy statement, all of which are subject to change or differing interpretations, including changes and interpretations with retroactive effect. No assurance can be given that the tax treatment described herein will remain unchanged at the time that awards under the 2020 Plan are made.
A Participant will not recognize income upon the grant of an option or at any time prior to the exercise of the option. At the time the participant exercises a non-qualified option, he or she will recognize compensation taxable as ordinary income in an amount equal to the excess of the fair market value per shareof the common stock on the date the option is exercised over the price paid for the common stock, and we will then be entitled to a corresponding deduction.
A Participant who exercises an incentive stock option will not be taxed at the time he or she exercises his or her options or a portion thereof. Instead, he or she will be taxed at the time he or she sells the common stock purchased pursuant to the option. The Participant will be taxed on the excess of the amount for which he or she sells the stock over the price he or she had paid for the stock. If the Participant does not sell the stock prior to two years from the date of grant of the option and one year from the date the stock is transferred to him or her upon exercise, the gain will be capital gain and we will not get a corresponding deduction. If the Participant sells the stock at a gain prior to that time, the difference between the amount the Participant paid for the stock and the lesser of the fair market value on the date of grant. The options awardedthe exercise or the amount for which the stock is sold, will be taxed as ordinary income and we will be entitled to a corresponding deduction. If the Participant sells the stock for less than the amount he or she paid for the stock prior to the one or two year periods indicated, no amount will be taxed as ordinary income and the loss will be taxed as a capital loss.
A Participant generally will not recognize income upon the grant of a stock appreciation right or a restricted stock unit. At the time a Participant receives shares or cash payment under any such award, he or she generally will recognize compensation taxable as ordinary income in an amount equal to the cash or the fair market value of the common stock received, less any amount paid for the stock, and we will then be entitled to a corresponding deduction. Upon a subsequent sale of the shares received under the Plansstock appreciation right or restricted stock unit, if any, the difference between the amount realized on the sale and the Participant’s tax basis (the amount previously included in income) is generally taxable as a capital gain or loss, which will vest as determinedbe short-term or long-term depending on the Participant’s holding time of such shares.
The taxation of restricted stock is dependent on the actions taken by the BoardParticipant. Generally, absent an election to be taxed currently under Section 83(b) of Directors butthe Code, or an 83(b) election, there will not exceedbe no federal income tax consequences to the Participant upon the grant of a ten-year period.restricted stock award. At the lapse of the restrictions or satisfaction of the conditions on the restricted stock, the Participant will recognize ordinary income equal to the fair market value of our common stock at that time. If the Participant makes an 83(b) election within 30 days of the date of grant, he or she will recognize ordinary income equal to the fair market value of our common stock at the time of grant, determined without regard to the applicable restrictions. If an 83(b) election is made, no additional income will be recognized by the Participant upon the lapse of the restrictions or satisfaction of the conditions on the restricted stock award. We generally should be entitled to a deduction equal to the amount of ordinary income recognized by the Participant, at the same time as the ordinary income is recognized by the Participant. Upon a subsequent sale of the formerly restricted stock, the difference between the amount realized on the sale and the Participant’s tax basis (the amount previously included in income) is generally taxable as a capital gain or loss, which will be short-term or long-term depending on the Participant’s holding time of such shares.
Fair ValueThe tax consequences to Participants who receive performance-based awards depend on the particular type of award issued. Our ability to take a deduction for such awards similarly depends on the terms of the awards and the limitations of Section 162(m) of the Code, if applicable. Section 162(m) of the Code currently imposes a $1 million limit on the amount that a public company may deduct for compensation paid to an employee who is chief executive officer, chief financial officer, or another “covered employee” (as defined by Section 162(m)), or was such an employee beginning in any year after 2017. The Compensation Committee retains the discretion to establish the compensation paid or intended to be paid or awarded to the executive officers as the Compensation Committee may determine is in the best interest of us and our stockholders, and without regard to any limitation provided in Section 162(m). This discretion is an important feature of the Compensation Committee’s compensation practices because it provides the Compensation Committee with sufficient flexibility to respond to specific circumstances facing us.
| 69 |
Outstanding Equity Awards at December 31, 2023
The following table presents the outstanding stock options and compensatory warrants held by each of the named executive officers as of December 31, 2023. There were no direct stock awards, restricted stock units or stock appreciation rights outstanding at December 31, 2023. All pre-2020 “option” awards shown were initially issued as Qualigen, Inc. Series C Warrants, and became warrants exercisable instead for our common stock (at an adjusted exercise price) upon the Reverse Recapitalization Transaction. The share numbers and exercise prices in the table below reflect the reverse stock split, which was effected by the Company on November 23, 2022 (the “Reverse Stock Split”).
| Equity Awards | ||||||||||||||||
| Name | Grant Date | Number of Securities Underlying Unexercised Awards (#) Exercisable | Number of Securities Underlying Unexercised Awards (#) Unexercisable | Exercise Price ($) | Expiration Date | |||||||||||
| Michael Poirier | 7/11/2022 | 12,500 | 25,000 | (1) | 5.14 | 7/11/2032 | ||||||||||
| 6/5/2020 | 100,000 | — | 51.30 | 6/5/2030 | ||||||||||||
| 9/22/2016 | 1,443 | — | 25.41 | 9/22/2026 | ||||||||||||
| 3/3/2015 | 2,214 | — | 25.41 | 3/2/2025 | ||||||||||||
| 8/2/2014 | 2,984 | — | 20.66 | 8/2/2024 | ||||||||||||
| 1/31/2014 | 2,214 | — | 20.66 | 1/31/2024 | ||||||||||||
| Tariq Arshad | 7/11/2022 | 3,400 | 6,800 | (1) | 5.14 | 7/11/2032 | ||||||||||
| 12/8/2021 | 20,000 | 10,000 | (2) | 12.40 | 5/17/2031 | |||||||||||
| 5/17/2021 | 6,667 | 3,333 | (1) | 18.00 | 5/17/2031 | |||||||||||
| Amy Broidrick | 7/11/2022 | — | 13,000 | (3) | 5.14 | 7/11/2032 | ||||||||||
| 12/8/2021 | — | 30,000 | (3) | 12.40 | 12/8/2031 | |||||||||||
| 12/7/2020 | — | 15,000 | (3) | 35.20 | 12/7/2030 | |||||||||||
| 8/27/2020 | — | 5,000 | (3) | 47.00 | 8/27/2030 | |||||||||||
| (1) | Shares underlying the stock option vest over three years in three equal annual installments from the date of grant. | |
| (2) | Shares underlying the stock option vest over three years in three equal annual installments from the vesting commencement date of May 17, 2021. | |
| (3) | Following Ms. Broidrick’s termination of employment on June 16, 2023, she did not exercise any vested stock options, and all of her equity awards were subsequently forfeited. |
Pay Versus Performance (PVP)
In accordance with the SEC’s disclosure requirements regarding pay versus performance, or PVP, this section presents the SEC-defined “Compensation Actually Paid,” or CAP of our principal executive officer (“PEO”) and named executive officers (“NEOs”) for each of the fiscal years ended December 31, 2023, 2022, and 2021, and our financial performance. Also as required by the SEC, this section compares CAP to various measures used to gauge performance at the Company for each such fiscal year.
Pay versus Performance Table - Compensation Definitions
Salary, Bonus, Stock Awards, and All Other Compensation are each calculated in the same manner for purposes of both CAP and Summary Compensation Table, or SCT values. The primary difference between the calculation of CAP and SCT total compensation is the calculation of the value of “Stock Awards,” with the table below describing the differences in how these awards are valued for purposes of SCT total and CAP:
| SCT Total | CAP | |||
| Stock Awards | Grant date fair value of stock awards granted during the year | Fair value of stock awards that are unvested as of the end of the year, or vested during the year |
Pay Versus Performance Table
In accordance with the SEC’s new PVP rules, the following table sets forth information concerning the compensation of our NEOs for each of the fiscal years ended December 31, 2023, 2022, and 2021, and our financial performance for each such fiscal year:
| Year | Summary Compensation Table Total for PEO | Compensation Actually Paid to PEO | Average Summary Compensation Table Total for non-PEO Named Executive Officers | Average Compensation Actually Paid to non-PEO Named Executive Officers | Value of Initial Fixed $100 Investment Based On Total Shareholder Return | Net Loss Attributable to Qualigen Therapeutics, Inc. (millions) | ||||||||||||||||||
| 2023 | $ | 632,698 | $ | 612,865 | $ | 514,321 | $ | 488,856 | $ | 1.83 | $ | (13.4 | ) | |||||||||||
| 2022 | 728,454 | 262,274 | 473,826 | 121,235 | 4.29 | (18.6 | ) | |||||||||||||||||
| 2021 | 742,279 | (753,431 | ) | 811,499 | 609,691 | 38.21 | (17.9 | ) | ||||||||||||||||
The Company utilizesprincipal executive officer (“PEO”) in 2023, 2022, and 2021 was Michael Poirier, our Chairman and Chief Executive Officer. The Non-PEO NEOs in 2023, 2022, and 2021 were Amy Broidrick, who was our President, Chief Strategy and Operating Officer, and Tariq Arshad, who was our Chief Medical Officer and Senior Vice President. The CAP was calculated beginning with the NEOs SCT total. The following amounts were deducted from and added to the applicable SCT total compensation:
| SCT Total | Stock awards deducted from SCT | Increase for fair value of awards granted during the year that remain unvested as of year end | Decrease in fair value from prior year-end to current year-end for awards granted in prior years and unvested as of year end | Decrease in fair value from prior year-end to current year vesting date for awards granted in prior years | Total CAP | |||||||||||||||||||
| (A) | (B) | (C ) | (D) | (E ) | A-B+C+D+E | |||||||||||||||||||
| PEO | ||||||||||||||||||||||||
| 2023 | $ | 632,698 | $ | - | $ | - | $ | (13,650 | ) | $ | (6,183 | ) | $ | 612,865 | ||||||||||
| 2022 | 728,454 | (145,274 | ) | 31,387 | (218,695 | ) | (133,598 | ) | 262,274 | |||||||||||||||
| 2021 | 742,279 | - | - | (1,236,534 | ) | (259,176 | ) | (753,431 | ) | |||||||||||||||
| Average Non-PEO NEO | ||||||||||||||||||||||||
| 2023 | $ | 514,321 | $ | - | $ | - | $ | (23,708 | ) | $ | (1,757 | ) | $ | 488,856 | ||||||||||
| 2022 | 473,826 | (44,936 | ) | 9,709 | (218,541 | ) | (98,823 | ) | 121,235 | |||||||||||||||
| 2021 | 811,499 | (363,370 | ) | 314,858 | (104,300 | ) | (48,997 | ) | 609,691 | |||||||||||||||
The fair value of stock options reported for CAP purposes in columns (B), (C), (D) and (E) above was estimated using a Black-Scholes option pricing model for the purposes of this PVP calculation in accordance with SEC rules. This model uses both historical data and current market data to estimate the fair value of options and requires several assumptions. The assumptions used in estimating fair value for awards under its Plans. Key valuationgranted during 2022 were as follows: volatility 102%, expected life 5.99 years, expected dividend yield 0%, risk-free rate 3.04%. The assumptions include:used in estimating fair value for awards granted during 2021 and prior were as follows: volatility 102%, expected life 5.99 years, expected dividend yield 0%, risk-free rate 0.42% - 1.43%. There were no awards granted in 2023.
Analysis of Information Presented in the Pay versus Performance Table
Our executive compensation program reflects a variable pay-for-performance philosophy. While we utilize several performance measures to align executive compensation with Company performance, all of those Company measures are not presented in the Pay versus Performance table. Moreover, we generally seek to incentivize long-term performance, and therefore we do not specifically align our performance measures with compensation that is actually paid (as computed in accordance with SEC rules) for a particular year. Further, we do not have the right to (without the executive’s consent) reduce an executive’s salary for a particular year to an amount lower than is provided for in any employment agreement with the executive which covers such year. In accordance with SEC rules, we provide the following narrative disclosure:
Compensation Actually Paid and Cumulative Total Stockholder Return
Compensation actually paid to our PEO increased from ($753,431) in 2021 to $262,274 in 2022, and further increased to $612,865 in 2023. Average compensation actually paid to our named executive officers other than our PEO decreased from $609,691 in 2021 to $121,235 in 2022, and increased to $488,856 in 2023. Over the same period, the value of an investment of $100 in our common stock on the last trading day of 2020 decreased by $61.79 to $38.21 during 2021, further decreased by $33.92 to $4.29 during 2022, and further decreased by $2.46 during 2023, for a total decrease over 2021, 2022, and 2023 of $98.17.
| 71 |
Compensation Actually Paid and Net Loss
Compensation actually paid to our PEO increased from ($753,431) in 2021 to $262,274 in 2022, and further increased to $612,865 in 2023. Average compensation actually paid to our named executive officers other than our PEO decreased from $609,691 in 2021 to $121,235 in 2022, and increased to $488,856 in 2023. Over the same period, our net loss increased by $0.7 million during 2022 (from a net loss in 2021 of $17.9 million to a net loss in 2022 of $18.6 million), and decreased by $5.2 million during 2023 (from a net loss in 2022 of $18.6 million to a net loss in 2023 of $13.4 million).
Compensation of Directors
For 2023, our non-employee directors were eligible to receive $35,000 in annual cash compensation. The Audit Committee chair was eligible to receive additional annual cash compensation of $15,000 and the other Board committee chairs were eligible to receive additional annual cash compensation of $10,000. Each non-chair member of each Board committee was eligible to receive additional annual cash compensation of $7,500 (Audit Committee) and $5,000 (other Committees). On January 13, 2023, the Company’s board of directors, as part of certain cost-cutting measures, approved a temporary 20% reduction to the compensation of all directors of the Company effective January 1, 2023. On August 1, 2023 the Company’s board of directors approved the reinstatement of the compensation of all directors of the Company to the above amounts effective August 1, 2023. Non-employee directors did not receive stock option grants during 2023. The amounts in the table below represent fees actually paid in cash during 2023 and include some fees earned in 2022.
Compensation paid to Mr. Poirier and Ms. Broidrick is presented as part of the “Summary Compensation Table” above, rather than here. Our employee directors do not receive compensation for their service as directors.
| Name of Director | Fees Paid in Cash ($) | Option Awards ($) | All other compensation ($) | Total ($) | ||||||||||||
| Richard David | 40,000 | — | — | 40,000 | ||||||||||||
| Sidney Emery, Jr. | 46,000 | — | — | 46,000 | ||||||||||||
| Matthew Korenberg | 42,000 | — | — | 42,000 | ||||||||||||
| Kurt Kruger | 40,000 | — | — | 40,000 | ||||||||||||
| Ira Ritter | 16,333 | — | — | 16,333 | ||||||||||||
As of December 31, 2023, all non-employee directors had been paid for compensation earned through July 31, 2023.
Hedging or Offsetting Against Compensatory Securities
We have adopted a policy that our employees (including officers) and directors shall not purchase securities or other financial instruments, or otherwise engage in transactions, that hedge or offset, or are designed to hedge or offset, any decrease in the market value of equity securities granted as compensation to, or held directly or indirectly by, those persons.
We have adopted a formal claw-back policy for the recovery of incentive-based executive compensation erroneously awarded to executive officers based on misstated financial reporting measures.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
The following table sets forth certain information regarding the beneficial ownership of our common stock as of March 25, 2024 by:
| our named executive officers; | ||
| our directors; | ||
| all of | ||
| each stockholder known by us to own beneficially more than 5% of our common stock. |
Beneficial ownership is determined in accordance with the rules of the SEC and includes voting or investment power with respect to the securities. Shares of common stock that may be acquired by an individual or group within 60 days after March 25, 2024, pursuant to the exercise of options or warrants, are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be outstanding for the purpose of computing the percentage ownership of any other person shown in the table. The percentage of beneficial ownership of our common stock is calculated based on an aggregate of 6,307,371 shares outstanding as of March 25, 2024.
| 72 |
Except as indicated in the footnotes to this table, we believe that the stockholders named in this table have sole voting and investment power with respect to all shares of common stock shown to be beneficially owned by them, based on information provided to us by such stockholders. Unless otherwise indicated, the address for each director and executive officer listed is: c/o Qualigen Therapeutics, Inc., 5857 Owens Avenue, Suite 300, Carlsbad, California 92008 USA.
| Beneficial Owner | Number of Shares Beneficially Owned | Percentage of Common Stock Beneficially Owned | ||||||
| Five Percent Stockholders | ||||||||
| Alpha Capital Anstalt (1) | 700,041 | 9.99 | % | |||||
| Executive Officers, Directors and Director Nominees | ||||||||
| Michael Poirier (2) | 142,376 | 2.2 | % | |||||
| Christopher Lotz (3) | 72,353 | 1.1 | % | |||||
| Richard David (4) | 7,219 | *% | ||||||
| Sidney Emery, Jr. (5) | 8,302 | *% | ||||||
| Matthew Korenberg (6) | 6,334 | *% | ||||||
| Kurt Kruger (7) | 9,019 | *% | ||||||
| Ira Ritter (8) | 6,738 | *% | ||||||
| All current executive officers and directors as a group (7 persons)(9) | 252,341 | 3.9 | % | |||||
* Represents beneficial ownership of less than 1% of the shares of common stock.
| (1) | Includes shares of common stock issuable upon the | |
| (2) | Includes 112,500 shares of common stock exercisable within 60 days under outstanding stock options and 8,855 shares of common stock exercisable within 60 days under outstanding warrants. | |
| (3) | Includes 63,333 shares of common stock exercisable within 60 days under outstanding stock options and 7,766 shares of common stock exercisable within 60 days under outstanding warrants. | |
| (4) | Includes 6,334 shares of common stock exercisable within 60 days under outstanding stock options and 885 shares of common stock exercisable within 60 days under outstanding warrants. | |
| (5) | Includes 6,334 shares of common stock exercisable within 60 days under outstanding stock options. | |
| (6) | Includes 6,334 shares of common stock exercisable within 60 days under outstanding stock options. | |
| (7) | Includes 6,334 shares of common stock exercisable within 60 days under outstanding stock options and 885 shares of common stock exercisable within 60 days under outstanding warrants. | |
| (8) | Includes 6,334 shares of common stock exercisable within 60 days under outstanding stock options. Also includes shares of common stock held in a retirement plan trust of which Ira Ritter and his spouse are trustees; and also includes shares beneficially owned by Stonehenge Partners. As a managing partner of Stonehenge Partners, Ira Ritter may be deemed the | |
| (9) | Includes 207,503 shares of common stock exercisable within 60 days under outstanding stock options and 18,391 shares of common stock exercisable within 60 days under outstanding warrants. |
| 73 |
Equity Compensation Plan Information
The following table presents information regarding securities authorized for issuance under equity compensation plans as of December 31, 2023:
| Plan Category | Number of Securities to be Issued upon Exercise of Outstanding Options, Warrants and Rights | Weighted-Average Exercise Price of Outstanding Options, Warrants and Rights | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (excluding securities reflected in column (a)) | |||||||||
| (a) | (b) | (c) | ||||||||||
| Equity compensation plans approved by stockholders | 398,924 | $ | 35.21 | 356,791 | ||||||||
| Equity compensation plans not approved by stockholders (1) | 119,046 | $ | 10.35 | — | ||||||||
| Total | 517,970 | $ | 29.50 | 356,791 | ||||||||
| (1) | Consists of shares of common stock issuable upon the exercise of compensatory warrants granted to service providers. |
Item 13. Certain Relationships and Related Transactions, and Director Independence.
Certain Relationships and Related Party Transactions
Our Audit Committee is responsible for reviewing, approving and overseeing any transaction between the Company electedand its directors, director nominees, executive officers, greater than 5% beneficial owners, and each of their respective immediate family members, where the amount involved exceeds the lesser of (i) $120,000 and (ii) 1% of the average of our total assets at year-end for the prior two fiscal years. Since January 1, 2021, there have been no such transactions except as described below.
On May 26, 2022, the Company acquired 2,232,861 shares of Series A-1 Preferred Stock of NanoSynex, Ltd. (“NanoSynex”) from Alpha a related party, in exchange for 350,000 reverse split adjusted shares of the Company’s common stock and a prefunded warrant to adoptpurchase 331,464 reverse split adjusted shares of the amendmentsCompany’s common stock at an exercise price of ASU 2016-09 (described$0.001 per share. These warrants were subsequently exercised on September 13, 2022.
On December 21 and 22, 2022, we entered into a Securities Purchase Agreement with Alpha and in Note 3) relatedexchange for $3,000,000 in cash (less $50,000 for expense reimbursement) issued to Alpha the 2022 Debenture, plus 2,500,000 common stock warrants exercisable (from June 22, 2023 through June 22, 2028) at $1.65 per share. Commencing June 1, 2023 and continuing on the first day of each month thereafter until the earlier of (i) December 22, 2025 and (ii) the full redemption of the 2022 Debenture (each such date, a “Monthly Redemption Date”), we must redeem $110,000 plus accrued but unpaid interest, liquidated damages and any amounts then owing under the 2022 Debenture (the “Monthly Redemption Amount”). The Monthly Redemption Amount must be paid in cash; provided that after the first two monthly redemptions, we may (if the Equity Conditions, as defined in the 2022 Debenture, are then satisfied or have been waived) elect to pay all or a portion of a Monthly Redemption Amount in shares of our common stock, based on a conversion price equal to the presentationlesser of excess tax benefits(i) the then applicable conversion price of the 2022 Debenture and (ii) 85% of the average of the VWAPs (as defined in the 2022 Debenture) for the five consecutive trading days ending on the statementtrading day that is immediately prior to the applicable Monthly Redemption Date.
The 2022 Debenture accrues interest at the rate of 8% per annum, which began accruing on December 1, 2023, and will be payable on a quarterly basis. Interest may be paid in cash flows usingor shares of common stock or a prospective transition methodcombination thereof at our option; provided that the Equity Conditions have been satisfied.
The 2022 Debenture is convertible into our common stock at any time at the holder’s option; the conversion price was originally $1.32.
Other than the Monthly Redemption Amounts, the 2022 Debenture does not call for scheduled payments of principal before the scheduled maturity date.
Both the 2022 Debenture and there was no impactthe accompanying warrants provide for “ratchet” antidilution adjustments to these financial statements.their conversion price and exercise price.
The material factors incorporated
| 74 |
Both the 2022 Debenture and the accompanying warrants include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
On December 5, 2023, we and Alpha entered into an Amendment No. 1 with regard to Securities Purchase Agreement, under which the conversion price of the 2022 Debenture and the exercise price of the associated warrants were reduced to $0.73 per share, in exchange for Alpha permitting us to make certain Monthly Redemption Amount payments in the Black-Scholes modelform of our stock rather than in estimatingcash even though the fair valueEquity Conditions were not satisfied (which would otherwise have prevented payment in the form of stock). In addition, such Amendment revised certain provisions of the options2,500,000 common stock warrants to (i) limit the circumstances which would trigger a potential adjustment to the exercise price of the 2,500,000 common stock warrants and (ii) clarify the treatment of the 2,500,000 common stock warrants upon a “Fundamental Transaction.” (The purpose of these revisions was to remove the terms that caused the 2,500,000 common stock warrants to be liability-classified under U.S. GAAP).
The new $0.73 per share conversion/exercise price triggered a “ratchet” antidilution adjustment in the Company’s outstanding “exploding” “Series C Warrants,” resulting in such Series C Warrants becoming exercisable for 455,623 common shares (at an exercise price of $0.73 per share), as opposed to the 251,971 common shares into which such outstanding Series C Warrants would have been exercisable (at $1.32 per share) pre-adjustment. Finally, the $0.73 per share price triggered a “ratchet” antidilution adjustment in the exercise price of other outstanding Company common stock warrants, including 7,084 warrants held by Alpha and 67,620 warrants held by other persons, all of which were previously exercisable at $1.32 per share.
In February 26 and 27, 2024, we entered into a Securities Purchase Agreement with Alpha and in exchange for $500,000 in cash (less $25,000 for expense reimbursement) issued to Alpha an 8% Convertible Debenture with a face amount of $550,000 due on December 31, 2024 (the “2024 Debenture”), plus 900,016 5-year common stock warrants exercisable at $0.26 per share. In addition, per this Securities Purchase Agreement Alpha obtained an option to purchase additional 8% Convertible Debentures, of like tenor, with face amounts of up to an aggregate of $1,100,000 (and with a proportional number of accompanying common stock warrants of like tenor, up to a total of 1,800,032 additional warrants), which would (if and when Alpha exercises such option) provide us up to an additional $1.0 million in cash proceeds (less expense reimbursement, and not including any possible cash proceeds from any future exercise of the additional warrants). This option is valid through July 1, 2024.
The 2024 Debenture has a maturity date of December 31, 2024 and is convertible, at any time, and from time to time, at Alpha’s option, into shares of our common stock, at $0.6111 per share. The 2024 Debenture does not call for scheduled payments of principal or interest before the scheduled maturity date. Interest on the 2024 Debenture accrues on its outstanding principal balance at the rate of 8% per annum.
Both the 2024 Debenture and the accompanying warrants provide for “ratchet” antidilution adjustments to their conversion price/exercise price.
Both the 2024 Debenture and the accompanying warrants include a beneficial ownership blocker of 9.99%, which may only be waived by Alpha upon 61 days’ notice to the Company.
We granted Alpha “piggyback” registration rights for the periods presented were as follows:common shares underlying the 2024 Debenture and the accompanying warrants.
| Years ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Expected dividend yield | 0.00 | % | 0.00 | % | ||||
| Expected stock-price volatility | 53.08% - 54.08 | % | 53.60% - 59.03 | % | ||||
| Risk-free interest rate | 1.89% - 2.58 | % | 1.29% - 1.78 | % | ||||
| Term of options | 10 | 10 | ||||||
| Stock price | $0.30 - $3.48 | $1.13 - $1.68 | ||||||
Stock-Based Compensation
The Company recognized stock-based compensation expense for services within general and administrative expense$0.26 exercise price of the warrants issued with the 2024 Debenture triggered a “ratchet” antidilution adjustment in the accompanying statements2022 Debenture, resulting in the then current $1,198,922 principal amount thereof becoming convertible into 4,611,238 shares of operationsCompany common stock (as opposed to the 1,642,359 shares into which such outstanding principal amount were convertible pre-adjustment). Also, the $0.26 exercise price of approximately $895,000the warrants issued with the 2024 Debenture triggered a “ratchet” antidilution adjustment in the Company’s outstanding “exploding” “Series C Warrants,” resulting in such Series C Warrants becoming exercisable for 1,279,261 common shares (at an exercise price of $0.26 per share), as opposed to the 455,623 common shares into which such outstanding Series C Warrants would have been exercisable (at $0.73 per share) pre-adjustment. Finally, the $0.26 exercise price of the warrant triggered a “ratchet” antidilution adjustment in the exercise price of other outstanding Company common stock warrants, including 2,507,048 warrants held by Alpha and $1,359,00067,620 warrants held by other persons, all of which were previously exercisable at $0.73 per share.
In connection with her termination of employment, on June 20, 2023, Qualigen, Inc., a former wholly-owned subsidiary of the Company, signed a separation agreement and general release (the “Separation Agreement”) with Amy Broidrick, which became effective on June 24, 2023.
Under the terms of the Separation Agreement, Qualigen, Inc. provided Ms. Broidrick with (i) $16,637 in cash compensation, less applicable withholdings for federal and state income and employment taxes, which represented Ms. Broidrick’s accrued but unpaid salary and vacation pay through the Separation Date, and reimbursement of certain expenses incurred by Ms. Broidrick, (ii) severance in the form of continued salary pay to Ms. Broidrick at the rate then in effect on the Separation Date ($360,000 per annum) for a period of 12 months following the Separation Date, subject to applicable withholding, and (iii) payment or reimbursement for the cost of COBRA continuation medical and dental insurance coverage for 12 months following the Separation Date, less any required taxes or withholdings. In addition, Ms. Broidrick is entitled to any rights or benefits under Qualigen Inc.’s employee benefit plans, to the extent earned and vested, and had three months from the Separation Date to exercise any vested stock options. Ms. Broidrick did not exercise any vested stock options.
| 75 |
Director Independence
Under Nasdaq’s continued listing requirements, a majority of a listed company’s board of directors must be comprised of independent directors, subject to certain exceptions. In addition, Nasdaq’s continued listing requirements require that, subject to certain exceptions, each member of a listed company’s audit, compensation and governance and nominating committees must be independent. Audit Committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. Under Nasdaq’s continued listing requirements, a director will only qualify as an “independent director” if, in the opinion of that company’s board of directors, such person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
Based upon information requested from and provided by each director concerning their background, employment and affiliations, including family relationships, our board of directors determined that each of Messrs. David, Emery, Korenberg, Kruger and Ritter are independent under the applicable rules and regulations of Nasdaq. In making such determinations, the board of directors considered the relationships that each such non-employee director has with our company and all other facts and circumstances the board of directors deemed relevant in determining their independence.
Item 14. Principal Accounting Fees and Services.
Baker Tilly US, LLP (“Baker Tilly”) serves as the Company’s independent registered public accounting firm and has served in that capacity since June 2018.
The Audit Committee considered the independence of Baker Tilly and whether the audit services Baker Tilly provided to the Company are compatible with maintaining that independence. The Audit Committee has adopted procedures by which the Audit Committee must approve in advance all services provided by and fees paid to the Company’s independent registered public accounting firm. The advance approval requirement was not waived in any instance during 2023 or 2022.
Fees and Services of Baker Tilly US, LLP
The following table sets forth the aggregate fees billed to the Company by Baker Tilly for the years ended December 31, 20172023 and 2016, respectively. As2022:
| 2023 | 2022 | |||||||
| Audit Fees(1) | $ | 495,955 | $ | 411,362 | ||||
| Audit-Related Fees | — | — | ||||||
| Tax Fees (2) | 18,789 | 35,050 | ||||||
| All Other Fees | — | — | ||||||
| Total | $ | 514,744 | $ | 446,412 | ||||
| (1) | Audit fees consisted of fees for audit work performed in the audit of financial statements, as well as fees for quarterly reviews and registration statements. | |
| (2) | These fees were incurred for professional services rendered in connection with tax compliance, tax advice, and tax planning. These services included income tax compliance and related tax services. |
The Audit Committee has adopted a formal policy on auditor independence requiring the advance approval by the Audit Committee of December 31, 2017, there was approximately $414,000all audit and non-audit services provided by our independent registered public accounting firm. In determining whether to approve any services by our independent registered public accounting firm, the Audit Committee reviews the services and the estimated fees, and considers whether approval of total unrecognized compensation cost relatedthe proposed services will have a detrimental impact on the auditor’s independence. On an annual basis, our management reports to unvested stock-based compensation arrangements. This cost is expected to be recognized over a weighted average period of 1.0 years.the Audit Committee all audit services performed during the previous 12 months and all fees billed by our independent registered public accounting firm for such services.
The aggregate intrinsic value of stock options exercised duringFor the years ended December 31, 20172023 and 2016 was approximately $02022, all audit services and $56,000, respectively.the corresponding fees were approved by our Audit Committee.
PART IV
Item 15. Exhibits and Financial Statement Schedules
(a) The cash received from exercised options was approximately $16,000following documents are filed as part of this Annual Report:
1. Financial Statements. The following documents are included in Part II, Item 8 of this Annual Report and are incorporated by reference herein:
2. Financial Statement Schedules. Financial statement schedules have been omitted because they are not required or are not applicable, or the required information is shown in the consolidated financial statements or notes thereto.
3. Exhibits. See EXHIBIT INDEX
EXHIBIT INDEX
| 4.7 | Form of liability classified Warrant to Purchase Common Stock | 10-K | 001-37428 | 4.13 | 3/31/2021 | |||||
| 4.8 | Form of “service provider” compensatory equity classified Warrant | 10-K | 001-37428 | 4.14 | 3/31/2021 | |||||
| 4.9 | Description of Common Stock | 10-K/A | 001-37428 | 4.9 | 7/7/2023 | |||||
| 4.10 | Amended and Restated Common Stock Purchase Warrant to GreenBlock Capital LLC, dated April 25, 2022 | 10-Q | 001-37428 | 4.15 | 5/13/2022 | |||||
| 4.11 | Amended and Restated Common Stock Purchase Warrant to Christopher Nelson, dated April 25, 2022 | 10-Q | 001-37428 | 4.16 | 5/13/2022 | |||||
| 4.12 | Common Stock Purchase Warrant for 2,500,000 shares in favor of Alpha Capital Anstalt, dated December 22, 2022 | 8-K | 001-37428 | 4.1 | 12/22/2022 |
| 10.17+ | Hire offer letter from the Company to Tariq Arshad, dated April 22, 2021 | 10-Q | 001-37428 | 10.1 | 8/16/2021 | |||||
| 10.20 | License Agreement with UCL Business Limited dated January 12, 2022 | 10-K | 001-37428 | 10.55 | 3/31/2022 | |||||
| 10.21 | First Deed of Variation to License Agreement with UCL Business Limited dated March 30, 2022 | 10-K | 001-37428 | 10.21 | 5/2/2023 | |||||
| 10.22 | Series B Preferred Share Purchase Agreement between the Company and NanoSynex Ltd. dated April 29, 2022 | 10-Q | 001-37428 | 10.1 | 5/13/2022 | |||||
| 10.23 | Share Purchase Agreement between the Company and Alpha Capital Anstalt dated April 29, 2022 | 10-Q | 001-37428 | 10.2 | 5/13/2022 |
| 31.1 | Certificate of principal executive officer pursuant to Rule 13a-14(a)/15d-14(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |||||||||
| 31.2 | Certificate of principal financial officer pursuant to Rule 13a-14(a)/15d-14(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |||||||||
| 32.1 | Certificate of principal executive officer and principal financial officer pursuant to 18 U.S.C. § 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | |||||||||
| 97.1* | Clawback Policy | |||||||||
| 101.INS# | Inline XBRL Instance Document. | |||||||||
| 101.SCH# | Inline XBRL Taxonomy Extension Schema Document. | |||||||||
| 101.CAL# | Inline XBRL Taxonomy Extension Calculation Linkbase Document. | |||||||||
| 101.DEF# | Inline XBRL Taxonomy Extension Definition Linkbase Document. | |||||||||
| 101.LAB# | Inline XBRL Taxonomy Extension Label Linkbase Document. | |||||||||
| 101.PRE# | Inline XBRL Taxonomy Extension Presentation Linkbase Document. | |||||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
* Filed or furnished herewith.
** Schedules have been omitted pursuant to Item 601(b)(2) of Regulation S-K. A copy of any omitted schedules will be furnished to the SEC upon request.
+ Indicates management contract or compensatory plan or arrangement.
# XBRL (Extensible Business Reporting Language) information is furnished and not filed herewith, is not a part of a registration statement or Prospectus for the year ended December 31, 2017.
NOTE 10 — RELATED PARTY TRANSACTIONS
A directorpurposes of sections 11 or 12 of the CompanySecurities Act of 1933, is a managing directordeemed not filed for purposes of Javelin Venture Partners GP, LLC,section 18 of the general partnerSecurities Exchange Act of Javelin Venture Partners GP, L.P., which holds a significant investment1934, and otherwise is not subject to liability under these sections.
Item 16. Form 10-K Summary
Not applicable.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Annual Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Qualigen Therapeutics, Inc. | ||
| By: | /s/ Michael S. Poirier | |
| Name: | Michael S. Poirier | |
| Title: | Chief Executive Officer (Principal Executive Officer) | |
| Date: April 5, 2024 | ||
| By: | /s/ Christopher L. Lotz | |
| Name: | Christopher L. Lotz | |
| Title: | Vice President of Finance, Chief Financial Officer (Principal Financial Officer and Chief Accounting Officer) | |
| Date: April 5, 2024 | ||
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below hereby constitutes and appoints Michael S. Poirier and Christopher L. Lotz, and each of them individually, his true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for him and in his name, place, and stead, in any and all capacities, to sign any and all amendments to this Annual Report, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or any of them, or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this Annual Report has been signed below by the following persons on behalf of the Registrant and in the Company’s common stockcapacities and warrants. Two directors of the Company have acted as a managing director of Stonehenge Partners, LLC, which holds significant investment in the Company’s common stock.
Other than disclosed, the Company has not entered into or been a participant in any transaction in which a related party had or will have a direct or indirect material interest.
NOTE 11 — INCOME TAXES
As of December 31, 2017, the Company has net operating loss carryforwards of approximately $39 million available to reduce future taxable income, if any, for Federal and state income tax purposes. The U.S. federal and state net operating loss carryforwards will begin to expire in 2028.
As of December 31, 2017, the Company has Federal and state research and development credit carryforwards of approximately $1.4 million and $800,000, respectively, available to reduce future taxable income, if any, for Federal and state income tax purposes. The Federal credit carryforwards begin to expire in 2029. California credits have no expiration date.
Under the Internal Revenue Code (“IRC”) Sections 382 and 383, annual use of the Company’s net operating loss and research tax credit carryforwards to offset taxable income may be limited based on cumulative changes in ownership. The Company has not completed an analysis to determine whether any such limitations have been triggered as of December 31, 2017. The Company has no income tax affect due to the recognition of a full valuation allowance on the expected tax benefits of future loss carry forwards based on uncertainty surrounding realization of such assets.dates indicated.
A reconciliation of the statutory income tax rates and the Company’s effective tax rate is as follows:
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Statutory U.S. federal rate | 34.0 | % | 34.0 | % | ||||
| State income tax, net of federal benefit | 5.8 | % | 5.8 | % | ||||
| Meals & entertainment | (0.2 | )% | (0.1 | )% | ||||
| Valuation allowance | (39.6 | )% | (39.7 | )% | ||||
| Provision for income taxes | 0.0 | % | 0.0 | % | ||||
The tax effects of the temporary differences and carry forwards that give rise to deferred tax assets consist of the following:
| As of December 31, | ||||||||
| 2017 | 2016 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carry forwards | $ | 10,906,689 | $ | 12,862,811 | ||||
| Patent costs | 325,615 | 363,775 | ||||||
| Accrued Vacation | 21,019 | 31,583 | ||||||
| Research and development credit | 1,932,804 | 1,951,539 | ||||||
| Stock-based compensation | 1,722,380 | 2,095,076 | ||||||
| Other | 8,766 | 7,535 | ||||||
| Gross deferred tax assets | 14,917,273 | 17,312,319 | ||||||
| Valuation allowance | (14,917,273 | ) | (17,312,319 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
The Company did not record any accruals for income tax accounting uncertainties for the years ended December 31, 2017 and 2016.
Authoritative guidance requires companies to accrue interest and related penalties, if applicable, on all tax positions for which reserves have been established consistent with jurisdictional tax laws. The Company’s policy is to recognize interest and penalties that would be assessed in relation to the settlement value of unrecognized tax benefits as a component of income tax expense. The Company did not accrue either interest or penalties from inception through December 31, 2017.
The Company does not have any unrecognized tax benefits that will significantly decrease or increase within 12 months of December 31, 2017.
The Company’s major tax jurisdictions are the United States and California. All of the Company’s tax years will remain open three and four years for examination by the Federal and state tax authorities, respectively, from the date of utilization of the net operating loss. The Company does not have any tax audits pending.
Regarding 2018 federal tax reform, the Company re-measured certain deferred tax assets and liabilities based on the rates at which they are expected to reverse in the future, which is generally 21%. For certain deferred tax assets, the Company has recorded a decrease of approximately $5.5 million, with a corresponding adjustment to valuation allowance for the year ended December 31, 2017. There is no impact on the tax expense.
| Signature | Title | Date | ||
| /s/ Michael S. Poirier | Chairman of the Board, Chief Executive Officer | April 5, 2024 | ||
| Michael S. Poirier | (Principal Executive Officer) | |||
| /s/ Christopher L. Lotz | Vice President of Finance, Chief Financial Officer | April 5, 2024 | ||
| Christopher L. Lotz | (Principal Financial and Accounting Officer) | |||
| /s/ Richard A. David | Director | April 5, 2024 | ||
| Richard A. David | ||||
| /s/ Sidney W. Emery, Jr. | Director | April 5, 2024 | ||
| Sidney W. Emery, Jr. | ||||
| /s/ Matthew E. Korenberg | Director | April 5, 2024 | ||
| Matthew E. Korenberg | ||||
| /s/ Kurt H. Kruger | Director | April 5, 2024 | ||
| Kurt H. Kruger | ||||
| /s/ Ira E. Ritter | Director | April 5, 2024 | ||
| Ira E. Ritter |