| ● | our ability to successfully commercialize the Nucynta Products will depend on many factors including, but not limited to, our ability to: | ·
| | develop and execute our sales and marketing strategies for the Nucynta Products;
|
| ·
| | achieve, maintain and grow market acceptance of, and demand for, the Nucynta Products;
|
| ·
| | obtain and maintain adequate coverage, reimbursement and pricing from managed care, government and other third-party payers;
|
| ·
| | maintain and manage the necessary sales, marketing, manufacturing, managed markets, and other capabilities and infrastructure that are required to successfully integrate and commercialize the Nucynta Products;
|
| ·
| | obtain adequate supply of Nucynta ER and Nucynta IR; and
|
| ·
| | comply with applicable legal and regulatory requirements. requirements, including any additional manufacturing or packaging requirements that may become applicable to certain opioid products. |
The success of our efforts to commercialize the Nucynta Products may also depend on additional factors, including the market acceptance of the Nucynta Products, and the outcome of a pending appellate decision in litigation between Depomed and ANDA filers who are seeking to market a generic version of the Nucynta Products in the U.S.
Many of these matters are beyond our control and are subject to other risks described elsewhere in this “Risk Factors” section. Accordingly, we cannot assure you that we will be able to continue successfully commercializecommercializing or to generate sufficient revenue from Xtampza, and/or the Nucynta Products.our products. If we cannot do so, or are significantly delayed in doing so, our business will be materially harmed. Despite receiving approval by the FDA, additional data may emerge that could change the FDA’s position on the product labeling of any of our products, including our abuse-deterrent claims with respect to Xtampza ER, and our ability to successfully market Xtampza or the Nucynta Productsour products successfully may be adversely affected. It is estimated that the U.S. market includes approximately 11 million patients
Xtampza ER was approved with chronic pain with dysphagia. Our Xtampza microspheres are designed to be removed from the capsule and sprinkled on food or into a cup, and then directly into the mouth, or in feeding tubes, without compromising their extended-release properties. On April 26, 2016, the FDA granted approval for the Xtampza NDA, including an approved product label. The FDA could change the product labeling. If the product label for Xtampza is modified in the future so as to exclude the flexible dose administration options, or the FDA requires us to have a boxed warning similar to competitor product labeling stating that “crushing, dissolving or chewing can cause rapid release and absorption of a potentially fatal doselanguage describing abuse-deterrent properties of the active drug,” it will limit our ability to differentiate Xtampza from other abuse-deterrent opioid formulations on the basis of flexible dosing options, and we may not be able to market Xtampza for use by patients with chronic pain with dysphagia. As a result, this may have an adverse effect on our business and our prospects for future growth. If the FDA does not conclude that our product candidates in development are sufficiently bioequivalent, or demonstrate comparable bioavailability to their respective listed drugs, or if the FDA otherwise does not conclude that our product candidates satisfy the requirements for the Section 505(b)(2) approval pathway, the approval pathway for those product candidates will likely take significantly longer, cost significantly more and entail significantly greater complications and risks than anticipated, and the FDA may not approve those product candidates.
A key element of our strategy is to seek FDA approval for our product candidates through the Section 505(b)(2) regulatory pathway. Section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act, or FD&C Act, permits the filing of an NDA that contains full safety and efficacy reports but where at least some of the information required for approval comes from studies not conducted by or for the applicant, such as the FDA’s findings of safety and efficacy in the approval of a similar drug, and for which the applicant has not obtained a right of reference and/or published literature. Such reliance is typically predicated on a showing of bioequivalence or comparable bioavailability to an approved drug.
If the FDA does not allow us to pursue the Section 505(b)(2) approval pathway for our product candidates, or if we cannot demonstrate bioequivalence or comparable bioavailability of our product candidates to approved products, we may need to conduct additional clinical trials, provide additional data and information, and meet additional standards for regulatory approval. If this were to occur, the time and financial resources required to obtain FDA approval for these product candidates would increase. Moreover, our inability to pursue the Section 505(b)(2) approval pathway could result in new competitive products reaching the market sooner than our product candidates, which could have a material adverse effect on our competitive position and our business prospects. Even if we are allowed to pursue the Section 505(b)(2) approval pathway, we cannot assure you that our product candidates will receive the requisite approvals for commercialization on a timely basis, if at all.
In addition, notwithstanding the approval of a number of products by the FDA under Section 505(b)(2) over the last few years, pharmaceutical companies and others have objected to the FDA’s interpretation of Section 505(b)(2). If the FDA’s interpretation of Section 505(b)(2) is successfully challenged, the FDA may change its policies and practicesformulation with respect to Section 505(b)(2) regulatory approvals, which could delay or even preventthe nasal and IV routes of abuse, consistent with Guidance for Industry, “Abuse-Deterrent Opioids- Evaluation and Labeling.” In November 2017, the FDA from approving any NDA that we submit under Section 505(b)(2).
Even if our product candidates are approved under Section 505(b)(2), the approval will likely be subject to limitations on the indicated uses for which the products may be marketed or to other conditions of approval, or may contain requirements for costly post-marketing testing and surveillance to monitor the safety or efficacy of the products, including additional preclinical studies and clinical trials.
Our decision to seek approval of our product candidates under Section 505(b)(2) increases the risk that a patent infringement suit may be filed against us, which would delay the FDA’s final regulatory approval of such product candidates.
In connection with any NDA that we file under Section 505(b)(2), we are required to notify the patent holders of the reference listed drug that we have certified to the FDA that any patents listed for the reference listed drug in the FDA’s Orange Book publication are invalid, unenforceable or will not be infringed by the manufacture, use or sale of our drug. If the patent holder files a patent infringement lawsuit against us within 45 days of its receipt of notice of our certification, the FDA is automatically prevented from approving our Section 505(b)(2) NDA until the earliest of 30 months, expiration of the patents, settlement of the lawsuit or a court decision in the infringement case that is favorable to us. Accordingly, we may invest significant time and expense in the development of our product candidates only to be subject to significant delay and expensive and time-consuming patent litigation before our product candidates may be commercialized.
Even if we are found not to infringe any potential plaintiff’s patent claims or the claims are found invalid or unenforceable, defending any such infringement claim could be expensive and time-consuming, and could delay the launch of our product candidates and distract management from their normal responsibilities. The Court could decline to hear our summary judgment motion, could decline to act expeditiously to issue a decision or hold a trial, or could decline to find that all of the listed patents are invalid or non-infringed. If we are unsuccessful in our defense of non-infringement and unable to prove invalidity of the listed patents, the court could issue an injunction prohibiting the launch of our product candidates. If we were to receive final regulatory approval by the FDA and launch any of our product candidates, prior to a full and final determination that the patents are invalid or non-infringed, we could be
subject to substantial liability for damages if we do not ultimately prevail on our defenses to a claim of patent infringement.
The regulatory approval processes of the FDA and foreign regulatory authorities are lengthy, time-consuming and unpredictable, and if we are ultimately unable to obtain regulatory approval for our product candidates, our business will be substantially harmed.
The time required to obtain approval by the FDA and foreign regulatory authorities is unpredictable, but typically takes many years following the commencement of preclinical studies and clinical trials and depends upon numerous factors, including the substantial discretion of the regulatory authorities. In addition, approval policies, regulations or the type and amount of clinical data necessary to gain approval varies among jurisdictions and may change during the course of a product candidate’s clinical development. Although the FDA has approved Xtampza, it is possible that none of our product candidates or any future product candidates that we may in-license, acquire or develop will ever obtain final regulatory approval from the FDA or any foreign regulatory authority. Moreover, even after any product candidate receives final regulatory approval, the FDA may require, as it hassNDA for Xtampza costly post-marketing requirements. SuccessfulER to include comparative oral pharmacokinetic data from a clinical study evaluating the effect of physical manipulation by crushing Xtampza ER compared with OxyContin and timely satisfactiona control (oxycodone hydrochloride immediate-release), results from an oral human abuse potential study and the addition of these post-marketing requirements will be necessary for us to maintain regulatory approval.an oral abuse deterrent claim.
Our product candidates could fail to receive regulatory approval from the FDA or a foreign regulatory authority, or we may be required to conduct more extensive studies and clinical trials in order to receive such approval, for many reasons, including, but not limited to:
| ·
| | the FDA and/or foreign regulatory authorities may disagree with or disapprove of the design or implementation of our clinical trials;
|
| ·
| | failure to demonstrate that a product candidate is safe and effective for its proposed indication;
|
| ·
| | failure to demonstrate that a product candidate is bioequivalent to its listed drug;
|
| ·
| | failure of clinical trials to meet criteria required for approval;
|
| ·
| | failure to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks;
|
| ·
| | the FDA or foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials;
|
| ·
| | deficiencies in the manufacturing processes or failure of third-party manufacturing facilities with whom we contract for clinical and commercial supplies to pass inspection;
|
| ·
| | the FDA or foreign regulatory authorities may not approve the manufacturing processes or facilities of third party manufacturers with which we contract for clinical and commercial supplies; or
|
| ·
| | insufficient data collected from clinical trials of our product candidates or changes in the approval policies or regulations that render our preclinical and clinical data insufficient to support the submission and filing of an NDA or to obtain regulatory approval.
|
The lengthy approval process, as well as the unpredictability of future clinical trial results, may result in our failing to obtain regulatory approval to market our product candidates, which would harm our business, results of operations and prospects significantly.
In addition, even if we obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited indications than we request, may not approve, with respect to certain foreign regulatory authorities, the price we intend to charge for our products, may grant approval contingent on the performance of costly post-marketing
requirements, or may approve a product label that does not include the labeling claims necessary or desirable for the successful commercialization of that product. Any of the foregoing scenarios could have a material adverse effect on our business.
The FDA or a foreign regulatory authority maycan require more information, including additional preclinical or clinical datachanges to support approval, which may delay or prevent approval and our commercialization plans, or cause us to abandon the development program. Even if we obtain regulatory approval, our product candidates may be approved for fewer or more limited indications than we request, such approval may be contingent on the performance of costly post-marketing requirements, or we may not be allowed to include the labeling claims necessary or desirable for the successful commercialization of such product candidate. In order to market and sell our products outside the United States, we will need to obtain separate marketing approvals and comply with numerous and varied regulatory requirements and regimes, which can involve additional testing, may take substantially longer than the FDA approval process, and still generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States, it is required that the product be approved for reimbursement before the product can be approved for sale in that country. FDA approval does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority outside the United States does not ensure approval by the FDA or regulatory authorities in other countries or jurisdictions. We may not obtain any regulatory approvals on a timely basis, if at all. We may not be able to file for marketing approvals and may not receive necessary approvals to commercialize our products in any market. If we are unable to obtain approval of any of our product candidates by regulatory authorities in countries outside the United States, the commercial prospects of that product candidate may be significantly diminished and our business prospects could decline.
Development of our product candidates is not complete, and we cannot be certain that our product candidates will be commercialized.
To commercialize our product candidates, in addition to commercializing Xtampza, we must successfully research, develop, obtain regulatory approval for, manufacture, launch, market and distribute product candidates under development. For each product candidate that we intend to commercialize, we must successfully meet a number of critical developmental milestones, including:
| ·
| | selecting and developing a drug delivery technology to deliver the proper dose of drug over the desired period of time;
|
| ·
| | determining the appropriate drug dosage that will be tolerated, safe and effective;
|
| ·
| | demonstrating the drug formulation will be stable for commercially reasonable time periods;
|
| ·
| | demonstrating that the drug is safe and effective in patients for the intended indication; and
|
| ·
| | completing the manufacturing development and scale-up to permit manufacture of our product candidates in commercial quantities and at acceptable prices.
|
The time necessary to achieve these developmental milestones for any individual product candidate is long and uncertain, and we may not successfully complete these milestoneslabeling for any of our product candidates in development. We may not be able to finalize the design or formulation of any product candidate. In addition, we may select components, solvents, excipients or other ingredients to include in our product candidates that have not been previously approved for use in pharmaceutical products which may require us to perform additional studies and may delay clinical testing and regulatory approval of our product candidates. Even after we complete the design of a product candidate, the product candidate must still be shown to be bioequivalent to an approved drug or safe and effective in required clinical trials before approval for commercialization.
We are continuing to test and develop our product candidates and may explore possible design or formulation changes to
address bioavailability, safety, efficacy, manufacturing efficiency and performance issues. We may not be able to complete development of any product candidates that will be safe and effective and that will have a commercially reasonable treatment and storage period. If we are unable to complete development of our product candidates, we will not be able to earn revenue from them.
Xtampza and the Nucynta Products are subject to mandatory REMS programs, which could increase the cost, burden and liability associated with the commercialization of these products. We anticipate that our product candidates, if approved, will also be subject to mandatory REMS programs.
The FDA has approved a REMS for extended release, or ER, and long acting, or LA, opioid drugs formulated with the active ingredients fentanyl, hydromorphone, methadone, morphine, oxycodone, oxymorphone, and others as part of a federal initiative to address prescription drug abuse and misuse, or the ER/LA opioid REMS. In September 2017, the FDA announced that immediate-release, or IR, opioid drugs will be subject to the same REMS as ER/LA opioids. One of the primary goals of the REMS is to ensure that the benefits of these drugs continue to outweigh the risks.
The REMS introduces new safety measures designed to reduce risks and improve the safe use of opioids, while continuing to provide access to these medications for patients in pain. The REMS applies to more than 20 companies that manufacture opioid analgesics. Under the REMS, companies are required to make education programs available to prescribers based on the FDA Blueprint for Prescriber Education for Extended Release and Long Acting Opioid Analgesics. It is expected that companies will meet this obligation by providing educational grants to continuing education providers, who will develop and deliver the training. The REMS also requires companies to distribute FDA-approved educational materials to prescribers and patients on the safe use of these drugs. The companies must perform periodic assessments of the implementation of the REMS and the success of the program in meeting its goals. The FDA will review these assessments and may require additional elements to achieve the goals of the program.
If the FDA determines that a REMS is necessary during review of an application, the drug sponsor must agree to the REMS plan at the time of approval. Xtampza and the Nucynta Products have been subject to the REMS requirement since their approval. REMS includes a Medication Guide that is dispensed with each prescription, physician training based on FDA-identified learning objectives, audits to ensure that the FDA’s learning objectives are addressed in the physician trainings, letters to prescribing physicians, professional organizations and state licensing entities alerting each to the REMS, and the establishment of a call center to provide more information about the REMS. We anticipate that our future product candidates will also be subject to these REMS requirements. There may be increased cost, administrative burden and potential liability associated with the marketing and sale of these types of product candidates subject to the REMS requirements, which could reduce the commercial benefits to us from the sale of these product candidates.
If we fail to comply with our obligations in the agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our business relationships with Depomed or other licensors, we could lose license rights that are important to our business.
We are, or may become, a party to certain intellectual property license agreements, including the Commercialization Agreement, that are important to our business and may enter into additional license agreements in the future. Our existing license agreements impose, and we expect that future license agreements will impose, various diligence, milestone, royalty and other obligations on us. If we fail to comply with the obligations under the Commercialization Agreement or other such agreements, Depomed or another such licensor may have the right to terminate the license, in which event we would not be able to market products covered by the license.
In addition, Depomed may terminate the Commercialization Agreement under certain circumstances, regardless of whether we are compliant with the terms of such agreement. If annual net sales of the Nucynta Products are less than $180,000,000 through January 1, 2022, or if they are less than $140,000,000 per year in any 12-month period commencing on January 1, 2022, then Depomed will have the right to terminate the Commercialization Agreement without penalty. Depomed may also terminate the Commercialization Agreement for convenience at any time prior to December 31, 2018, provided it will be required to pay a termination fee to us.
In some cases, patent prosecution of our licenses is controlled solely by the licensor. If our licensors fail to obtain and maintain patent or other protection for the proprietary intellectual property we license from them, we could lose our
rights to the intellectual property or our exclusivity with respect to those rights, and our competitors could market competing products using the intellectual property. In certain cases, we control the prosecution of patents resulting from licenses. In the event we breach any of our obligations related to such prosecution, we may incur significant liability to our licensing partners. Licensing of intellectual property is of critical importance to our business and involves complex legal, business and scientific issues and is complicated by the rapid pace of scientific discovery in our industry. Disputes may arise regarding intellectual property subject to a licensing agreement, including:
| ·
| | the scope of rights granted under the license agreement and other interpretation-related issues;
|
| ·
| | the extent to which our technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement;
|
| ·
| | the sublicensing of patent and other rights under our collaborative development relationships;
|
| ·
| | our diligence obligations under the license agreement and what activities satisfy those diligence obligations;
|
| ·
| | the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners; and
|
| ·
| | the priority of invention of patented technology.
|
If disputes over intellectual property that we have licensed prevent or impairwhich can impact our ability to maintain our current licensing arrangements on acceptable terms, we may be unable to successfully develop and commercialize the affected products or product candidates.
If we fail to obtain the necessary final regulatory approvals, or if such approvals are limited, we will not be able to commercialize our product candidates, and we will not generate product revenues.
Even if we comply with all FDA pre-approval regulatory requirements, the FDA may determine that our product candidates are not safe or effective, and we may never obtain final regulatory approval for such product candidates. If we fail to obtain final regulatory approval for some or all of our product candidates, we will have fewer commercial products and correspondingly lower product revenues. Even if our product candidates receive final regulatory approval, such final regulatory approval may involve limitations on the indications and conditions of use or marketing claims for our products, or may not include certain abuse-deterrence claims or clinical trial data that we have sought, and will seek, to include in the product label. If we do not receive regulatory approval to include certain abuse-deterrence claims, or certain clinical data, in our product labels, our ability to successfully commercialize our products may be limited and our financial results may be adversely impacted. Further, later discovery of previously unknown problems or adverse events could result in additional regulatory restrictions, including withdrawal of products and addition of warnings or other statements on the product label. The FDA may require us to perform lengthy Phase 4 post-approval clinical efficacy or safety trials. Post approval, the FDA may require us to study, as it has with respect to Xtampza, the serious risks of misuse, abuse, addiction, overdose, and death associated with long-term use of our medications for the management of chronic pain, as well as other risks. The FDA may also impose additional post-marketing requirements, which will be very expensive to satisfy.
sales. In jurisdictions outside the United States, we must receive marketing authorizations from the appropriate regulatory authorities before commercializing our product candidates. Regulatory approval processes outside the United States generally include requirements and risks similar to, and in many cases in excess of, those associated with FDA approval. The FDA may not approve product labeling for our product candidates that would permit us to market and promote our products in the United States by describing their abuse-deterrent features.
We invest substantial time and money conducting Category 1, Category 2 and Category 3 abuse deterrent studies to ensure that our product candidates developed with our DETERx technology comply with the FDA’s April 2015 guidance regarding opioid abuse deterrence. Our failure to achieve FDA approval of product labeling containing such
information will prevent or substantiality limit our promotion of the abuse deterrent features of our product candidates in order to differentiate them from other opioid products containing the same active ingredients. This would make our products less competitive in the market. There can be no assurance that any of our product candidates will receive final FDA-approved product labeling that describes the abuse deterrent features of such products. Furthermore, the FDA’s April 2015 final guidance on abuse deterrent opioids makes clear that the FDA expects sponsors to compare their formulations against approved abuse deterrent versions of the same opioid based on the relevant categories of testing. If a proposed product is less resistant to manipulation than an approved product, the FDA has stated that the proposed product may not be eligible for product labeling regarding abuse deterrent properties. If the FDA does not approve product labeling containing abuse deterrence claims, we will not be able to promote such products based on their abuse deterrent features, may not be able to differentiate such products from other opioid products containing the same active ingredients, and may need to lower the price of our products to the extent that there are competing products with abuse deterrent claims on their product labels.
Because the FDA closely regulates promotional materials and other promotional activities, even if the FDA initially approves product labeling that includes a description of the abuse deterrent characteristics of our product, the FDA may object to our marketing claims and product advertising campaigns. This could lead to the issuance of warning letters or untitled letters, suspension or withdrawal of our products from the market, recalls, fines, disgorgement of money, operating restrictions, injunctions, and civil or criminal prosecution. Any of these consequences would harm the commercial success of our products.
Even if any of our product candidates are approved for marketing with certain abuse-deterrence claims, the April 2015 final FDA guidance on abuse-deterrent opioids is not binding law and may be superseded or modified at any time. Also,particular, if the FDA determines that our post-marketing data dofor Xtampza ER does not demonstrate that the abuse-deterrent properties result in reduction of abuse, or demonstratedemonstrates a shift to routes of abuse that present a greater risk, the FDA may find that product labeling revisions are needed, and potentially require the removal of our abuse-deterrence claims.
Even ifclaims, which would have a material adverse effect on our product candidates receive regulatory approval, they will beability to continue successfully commercializing Xtampza ER.
Our opioid products are subject to ongoing regulatory requirements,mandatory REMS programs, which could increase the cost, burden and we may face regulatory enforcement action if we do not complyliability associated with the requirements.commercialization of these products. Even after
The FDA has imposed a productclass-wide REMS on all IR, ER and long-acting opioid drug products (known as the Opioid Analgesic REMS). The FDA continually evaluates whether the REMS program is approved, we will remain subjectmeeting its goal of ensuring that the benefit of these drugs continue to ongoing FDAoutweigh their risks, and other regulatory requirements governingwhether the product labeling, packaging, storage, distribution, safety surveillance, advertising, promotion, import, export, record-keeping and reportinggoals or elements of safety and other post-market information. If we experience delays in obtaining FDA approval of our advertising and promotional materials forthe program should be modified. As opioids, Xtampza the Nucynta Products, or any product candidate that receives marketing approval, or if FDA approval of such materials is contingent upon substantial modifications, our promotional efforts relating to Xtampza,ER, the Nucynta Products and any approved product candidate may be impaired, and sales of such products may suffer. The holder of an approved NDA is obligated to monitor and report adverse events, or AEs, and any failure of a product to meet the specifications in the NDA. In addition, manufacturers of drug products and their facilitiesBelbuca are subject to paymentthe Opioid Analgesic REMS.
Any modification of user fees and continual review and periodic inspectionsthe Opioid Analgesic REMS by the FDA and other regulatory authorities for complianceto impose additional or more burdensome requirements could increase the costs associated with current good manufacturing practices, marketing these products and/or cGMP, and other regulations. If we orreduce the willingness of healthcare providers to prescribe these products, which would have a regulatory agency discover problems with a product which were previously unknown, such asmaterial adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictionseffect on that product, the manufacturing facility or us, including requiring product recall, notice to physicians, withdrawal of the product from the market or suspension of manufacturing, among other things. If we, our product candidates or the manufacturing facilities for our product candidates fail to comply with applicable regulatory requirements, a regulatory agency may: | ·
| | issue warning letters or untitled letters;
|
| ·
| | mandate modifications to promotional materials or require us to provide corrective information to healthcare practitioners;
|
| ·
| | require us to enter into a consent decree, which can include the imposition of various fines, reimbursements for inspection costs and penalties for noncompliance, and require due dates for specific actions;
|
| ·
| | seek an injunction or impose civil, criminal and/or administrative penalties, damages, monetary fines, require disgorgement, consider exclusion from participation in Medicare, Medicaid and other federal healthcare programs and require curtailment or restructuring of our operations;
|
| ·
| | suspend or withdraw regulatory approval;
|
| ·
| | suspend any ongoing clinical trials;
|
| ·
| | refuse to approve pending applications or supplements to applications filed by us;
|
| ·
| | suspend or impose restrictions on operations, including costly new manufacturing requirements;
|
| ·
| | seize or detain products, refuse to permit the import or export of products, or require us to initiate a product recall; or
|
| ·
| | refuse to allow us to enter into government contracts.
|
Similar post-market requirements may apply in foreign jurisdictions in which we may seek approval of our products. Any government investigation of alleged violations of law could require us to expend significant time and resources in response and could generate negative publicity. The occurrence of any event or penalty described above may inhibit our ability to continue to successfully commercialize our products and generate sufficient revenue and may cause a material adverse impact on our financial condition and cash flows.from these products.
In addition, the FDA’s regulations, policies or guidance may change and new or additional statutes or government regulations in the United States and other jurisdictions may be enacted that could further restrict or regulate post-approval activities. We cannot predict the likelihood, nature or extent of adverse government regulation that may arise from pending or future legislation or administrative action, either in the United States or abroad. If we are not able to achieve and maintain regulatory compliance, we may not be permitted to market our products and/or product candidates, which would adversely affect our ability to generate revenue and achieve or maintain profitability.
Failure to comply with ongoing governmental regulations for marketing our products, and in particular any product, includingfailure to promote Xtampza and the Nucynta Products,ER’s abuse deterrent labeling in compliance with FDA regulations, could delay or inhibit our ability to generate revenues from their sale and could also expose us to claims or other sanctions. Advertising
In addition to scrutiny by the FDA, advertising and promotion of any pharmaceutical product that obtains approvalmarketed in the United States including Xtampza and the Nucynta Products, will beis heavily scrutinized by, among others, the FDA, the Department of Justice, or the DOJ, the Office of Inspector General offor the U.S. Department of Health and Human Services, or HHS, state attorneys general, members of Congress and the public. Violations, including promotion of Xtampza or the Nucynta Products, and any product for which we receive final regulatory approval,our products for unapproved or off-label uses, are subject to enforcement letters, inquiries and investigations, and civil and criminal sanctions by government agencies. In particular, Xtampza ER has FDA-approved product labeling that describes its abuse deterrent features, which allows us to promote those features and differentiate Xtampza ER from other opioid products containing the same active pharmaceutical ingredients. Because the FDA orclosely regulates promotional materials and other government agencies. Additionally,promotional activities, even though the FDA-approved product labeling includes a description of the abuse deterrent characteristics of Xtampza ER, the FDA may object to our marketing claims and product advertising and promotion of any product that obtains approval outside the United States will be heavily scrutinized by foreign regulatory authorities.campaigns. In the United States, engaging
Engaging in off-label promotion of our products, including Xtampza or the Nucynta Products, or any products, can alsoER, could subject us to false claims litigationliability under federal and state statutes, and other litigation and/or investigation, which caninvestigations, and could lead to civil and criminal penalties and fines and agreements that materially restrict the manner in which we promoteissuance of warning letters or distribute our drug products. These false claims statutes include the federal False Claims Act, which allows any individual to bring a lawsuit against a pharmaceutical company on behalf of the federal government alleging submission of falseuntitled letters, suspension or fraudulent claims, or causing to present such false or fraudulent claims, for payment by a federal program such as Medicare or Medicaid. If the government prevails in the lawsuit, the individual will share in any fines or settlement funds. False Claims Act lawsuits against pharmaceutical companies have increased significantly in volume and breadth in recent years, leading to several substantial civil and criminal settlements based on certain sales practices promoting off-label drug uses. This increased focus and scrutiny has increased the risk that a pharmaceutical company will have to defend a false claim action, pay settlement fines or restitution, agree to comply with burdensome reporting and compliance obligations, and be excluded from the Medicare, Medicaid and other federal and state healthcare programs.
If we are found to have promoted such off-label uses, we may become subject to significant liability. The federal government has levied large civil and criminal fines against companies for alleged off-label use and has enjoined several companies from engaging in off-label promotion. The FDA has also requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed. If we cannot successfully manage the promotionwithdrawal of our products we could become subject to significant liability, which could materially adversely affectfrom the market, recalls, fines, disgorgement money, operating restrictions, injunctions, and civil or criminal prosecution. Any of these consequences would harm the commercial success of our business and financial condition.products, including Xtampza ER.
In addition, later
Further, discovery of previously unknownserious and unanticipated adverse events associated with the product; the emergence of other problems with athe product, manufacturer or facility,facility; or our failure to updatemake required regulatory files,submissions may result in restrictions,adverse regulatory actions, including withdrawal of the product from the market. Anymarket or the requirement to add or strengthen label warnings about the product. The failure to obtain or maintain requisite governmental approvals or the imposition of the followingadditional or other similar events, if they were to occur,stronger warnings could delay or preclude us from further developing, marketing or realizing the full commercial potential of Xtampza, the Nucynta Products and our product candidates: | ·
| | failure to obtain or maintain requisite governmental approvals;
|
| ·
| | failure to obtain approvals of product labeling with abuse-deterrent claims; or
|
| ·
| | FDA required product withdrawals or warnings arising from identification of serious and unanticipated adverse side effects in our product candidates.
|
Xtampza, the Nucynta Products and our product candidates contain controlled substances, the manufacture, use, sale, importation, exportation and distribution of which are subject to regulation by state, federal and foreign law enforcement and other regulatory agencies.
Xtampza, the Nucynta Products and our product candidates contain, and our future product candidates will likely contain, controlled substances that are subject to state, federal and foreign laws and regulations regarding their manufacture, use, sale, importation, exportation and distribution. Xtampza’s active ingredient, oxycodone, and the Nucynta Products’ active ingredient, tapentadol, are both classified as controlled substances under the Controlled Substances Act of 1970, or CSA, and regulations of the U.S. Drug Enforcement Administration, or DEA. A number of states also independently regulate these drugs, including oxycodone and tapentadol, as controlled substances. Controlled substances are classified by the DEA as Schedule I, II, III, IV or V substances, with Schedule I substances considered to present the highest risk of substance abuse and Schedule V substances the lowest risk. Oxycodone and tapentadol are both listed by the DEA as Schedule II controlled substances under the CSA. For our product candidates containing controlled substances, we and our suppliers, manufacturers, contractors, customers and distributors are required to obtain and maintain applicable registrations from state, federal and foreign law enforcement and regulatory agencies and comply with state, federal and foreign laws and regulations regarding the manufacture, use, sale, importation, exportation and distribution of controlled substances. For example, all Schedule II drug prescriptions must be signed by a physician, physically presented to a pharmacist and may not be refilled without a new prescription.
Furthermore, the amount of Schedule II substances that can be obtained for clinical trials and commercial distribution is limited by the CSA and DEA regulations. We may not be able to obtain sufficient quantities of these controlled substances in order to complete our clinical trials or meet commercial demand. If commercial demand for Xtampza, or any of our other approved products, increases and we cannot meet such demand in a timely fashion because of our limited supply of its active ingredient(in the case of Xtampza, oxycodone) then physicians may perceive such product as unavailable and may be less likely to prescribe it in the future.
In addition, controlled substances are also subject to regulations governing manufacturing, labeling, packaging, testing, dispensing, production and procurement quotas, recordkeeping, reporting, handling, shipment and disposal. These regulations increase the personnel needs and the expense associated with development and commercialization of Xtampza, the Nucynta Products, and product candidates that include controlled substances. The DEA and some states conduct periodic inspections of registered establishments that handle controlled substances.
Failure to obtain and maintain required registrations or to comply with any applicable regulations could delay or
preclude us from developing and commercializing Xtampza, the Nucynta Products, and product candidates that contain controlled substances and subject us to enforcement action. The DEA may seek civil penalties, refuse to renew necessary registrations or initiate proceedings to revoke those registrations. In some circumstances, violations could lead to criminal proceedings. Because of their restrictive nature, these regulations could limit commercialization of products containing controlled substances.
Clinical development is a lengthy and expensive process with an uncertain outcome, and failure can occur at any stage of clinical development. If we are unable to design, conduct and complete clinical trials successfully, our product candidates will not be able to receive regulatory approval.
In order to obtain FDA approval for any of our product candidates, we must submit to the FDA an NDA with substantial evidence that demonstrates that the product candidate is both safe and effective in humans for its intended use. This demonstration requires significant research, preclinical studies and clinical trials.
All of our product candidates are in preclinical and clinical development. Clinical trials are time-consuming, expensive and difficult to design and implement, in part because they are subject to rigorous requirements and their outcomes are inherently uncertain. Clinical testing may take many years to complete, and failure can occur at any time during the clinical trial process, even with active ingredients that have previously been approved by the FDA as being safe and effective. We could encounter problems that halt our clinical trials or require us to repeat such clinical trials. If patients participating in clinical trials suffer drug-related adverse reactions during the clinical trials, or if we or the FDA believe that patients are being exposed to unacceptable health risks, such clinical trials may be suspended or terminated. Suspensions, termination or the need to repeat a clinical trial can occur at any stage.
The clinical trial success of each of our product candidates depends on reaching statistically significant changes in patients’ symptoms based on clinician-rated scales. There is a lack of consensus regarding standardized processes for assessing clinical outcomes based on clinician-rated scales. Accordingly, the scores from our clinical trials may not be reliable, useful or acceptable to the FDA or other regulatory agencies.
Changes in standards related to clinical trial design could have a material adverse effect on our ability to design and conduct clinical trials as planned. For example, we have conducted or will conduct clinical trials comparing our product candidates to both placebo and other approved drugs, but regulatory authorities may not allow us to compare our product candidates to a placebo in a particular clinical indication where approved products are available. In that case, both the cost and the amount of time required to conduct a clinical trial could increase. The FDA may disagree with our trial design and our interpretation of data from clinical trials, or may change the requirements for approval even after it has reviewed and commented on the design for our clinical trials. The FDA may also approve a product candidate for fewer or more limited indications than we request, or may grant approval contingent on the performance of costly post-approval clinical trials. In addition, the FDA may not approve the product labeling claims or removal of certain warnings that we believe are necessary or desirable for the successful commercialization of our product candidates.
Approval may be contingent on a REMS, which could have a material adverse effect on the product labeling, distribution or promotion of a drug product.
Any of these delays or additional requirements could cause our product candidates to not be approved, or if approved, significantly impact the timing of commercialization and significantly increase our overall costs of drug development.
Because the results of preclinical studies and early-stage clinical trials are not necessarily predictive of future results, any product candidate we advance into additional clinical trials may not continue to have favorable results or receive regulatory approval.
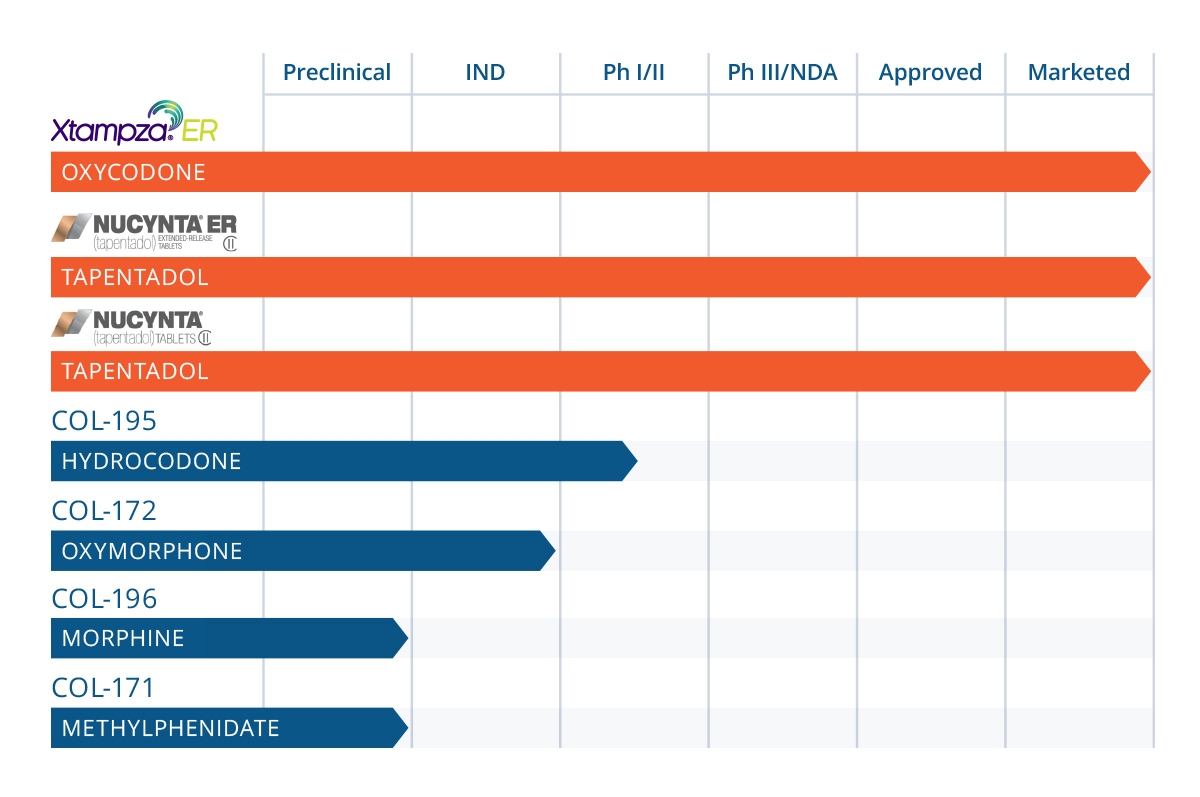
All of our product candidates are in preclinical or early-stage clinical development. Success in preclinical studies and early clinical trials does not ensure that later clinical trials will generate adequate data to demonstrate the efficacy and safety of an investigational drug. Many companies in the pharmaceutical and biotechnology industries, including those with greater resources and experience, have suffered significant setbacks in clinical trials, even after positive results in earlier clinical trials. Despite preliminary preclinical studies for our other extended-release, abuse deterrent product candidates, including hydrocodone and oxymorphone for pain, and methylphenidate for the treatment of ADHD, we do
not know whether the clinical trials we may conduct will demonstrate adequate efficacy and safety or otherwise provide adequate information to result in regulatory approval to market any of our product candidates in any particular jurisdiction. If later-stage clinical trials do not produce favorable results, our ability to achieve regulatory approval for any of our product candidates may be compromised.
Conducting clinical trials of Xtampza and our product candidates and any commercial sales of Xtampza, the Nucynta Products, and/or product candidates may expose us to expensive product liability claims, and we may not be able to maintain product liability insurance on reasonable terms or at all.
We currently carry product liability insurance with coverage up to approximately $10.0 million. Product liability claims may be brought against us by patients enrolled in our clinical trials, patients, healthcare providers or others using, administering or selling our products. If we cannot successfully defend ourselves against claims that our products or product candidates caused injuries, we could incur substantial liabilities. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. Regardless of merit or eventual outcome, liability claims may result in:
| ·
| | decreased demand for any product or product candidates that we may develop;
|
| ·
| | termination of clinical trial sites or entire trial programs;
|
| ·
| | injury to our reputation and significant negative media attention;
|
| ·
| | withdrawal of clinical trial participants;
|
| ·
| | significant costs to defend the related litigation;
|
| ·
| | substantial monetary awards to patients;
|
| ·
| | diversion of management and scientific resources from our business operations;
|
| ·
| | the inability to commercialize any products that we may develop; and
|
| ·
| | an increase in product liability insurance premiums or an inability to maintain product liability insurance coverage.
|
Our inability to maintain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of Xtampza, the Nucynta Products, and our product candidates. Any agreements we may enter into in the future with collaborators in connection with the development or commercialization of Xtampza and our product candidates may entitle us to indemnification against product liability losses, but such indemnification may not be available or adequate should any claim arise. In addition, many of our agreements require us to indemnify third parties and these indemnifications obligations may exceed the coverage under our product liability insurance policy.
Xtampza, the Nucynta Products, and our product candidates may be associated with undesirable adverse reactions or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of their approved product label, or result in significant negative consequences following any marketing approval.
Undesirable adverse reactions associated with Xtampza, the Nucynta Products, and our product candidates could cause us, our IRBs, clinical trial sites or regulatory authorities to interrupt, delay or halt clinical trials and could result in a restrictive product label or the delay, denial or withdrawal of regulatory approval by the FDA or foreign regulatory authorities. For example, even though Xtampza was generally well tolerated by patients in our clinical trials, in some
cases there were adverse reactions, one of which was a serious adverse event, moderate in severity, of gastroesophageal reflux.
If we or others identify undesirable adverse events associated with Xtampza, the Nucynta Products, or any product candidate for which we receive final regulatory approval, a number of potentially significant negative consequences could result, including:
| ·
| | we may be forced to suspend marketing of the product;
|
| ·
| | regulatory authorities may withdraw their approvals of the product or impose restrictions on its distribution;
|
| ·
| | regulatory authorities may require additional warnings or contradictions in the product label that could diminish the usage or otherwise limit the commercial success of the product;
|
| ·
| | we may be required to conduct additional post-marketing studies;
|
| ·
| | we could be sued and held liable for harm caused to patients; and
|
| ·
| | our reputation may suffer.
|
Any of these events could prevent us from achieving or maintaining market acceptance of Xtampza or any of our product candidates, if approved.
Risks Related to Intellectual Property Unfavorable outcomes in intellectual property litigation could result inbe costly litigation and potentially limit our ability to commercialize our products. Our commercial success depends upon our ability to develop product candidates and commercialize products without infringing the intellectual property rights of others. Our current or future product candidates or products, or any uses of them, may now or in the future infringe third-party patents or other intellectual property rights. This is due in part to the considerable uncertainty within the pharmaceutical industry about the validity, scope and enforceability of many issued patents in the United States and elsewhere in the world and, to date, there is no consistency regarding the breadth of claims allowed in pharmaceutical patents. We cannot currently determine the ultimate scope and validity of patents which may be granted to third parties in the future or which patents might be asserted to be infringed by the manufacture, use and sale of our products. In part as a result of this uncertainty, there has been, and we expect that there will continue to be, significant litigation in the pharmaceutical industry regarding patents and other intellectual property rights. Third parties may assert infringement claims against us, or other parties we have agreed to indemnify, based on existing patents or patents that may be granted in the future. We are aware of third-party patents and patent applications related to oxycodone, oxymorphone, hydrocodone, morphine, and methylphenidate drugs and formulations, including those listed in the FDA’s Orange Book for oxycodone products. Because of the delay between filing and publication of patent applications, and because applications can take several years to issue, there may be currently pending third-party patent applications that are unknown to us, which may later result in issued patents. Because of the uncertainty inherent in intellectual property litigation, we could lose, even if the case against us was weak or flawed.
If we are found to infringe a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue developing or commercializing Xtampza or our product candidates, products and technology. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even if we are able to obtain a license, it may be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. We could be forced, including by court order, to cease commercializing the infringing technology or product. In addition, in any such proceeding or litigation, we could be found liable for monetary damages, including treble damages and attorneys’ fees, if we are found to have willfully infringed a patent. A finding of infringement could prevent us from commercializing Xtampza or our product candidatesproducts or force us to cease some of our business operations. In connection with any NDA that we file under Section 505(b)(2), including the NDA for Xtampza, we are required to notify the patent holder of the reference listed drug that we identify in our NDA, that we have certified to the FDA that any patents listed for the listed drug in the FDA’s Orange Book publication are invalid, unenforceable or will not be infringed by the manufacture, use or sale of our drug. If the patent holder files a patent infringement lawsuit against us within 45 days of its receipt of notice of our certification, the FDA is automatically prevented from approving our Section 505(b)(2) NDA until the earliest of 30 months after the lawsuit is filed, expiration of the patents, settlement of the lawsuit and a court decision in the infringement case that is favorable to us. Accordingly, we may invest significant time and expense in the development of our product candidates only to be subject to significant delay and patent litigation before our product candidates may be commercialized.
If we are found by the court to have infringed a valid patent claim, we could be prevented from using the patented technology or be required to pay the patent holder for the right to license the patented technology. If we decide to pursue a license to use one or more of these patents, we may not be able to obtain a license on commercially reasonable terms, if at all, or the license we obtain may require us to pay substantial royalties or grant cross licenses to our patent rights. For example, if the relevant patent is owned by a competitor, such as Purdue, that competitor may choose not to license patent rights to us. If we decide to develop alternative technology, we may not be able to do so in a timely or cost-effective manner, if at all.
Even if we are found not to infringe or patent claims are found invalid or unenforceable, defending any such infringement claim would be expensive and time consuming, and could delay the approval or commercialization of our product candidates and distract management from their normal responsibilities.
Competitors may sue us as a way of delaying the introduction of our products. Any litigation, including any interference or derivation proceedings to determine priority of inventions, oppositions or other post-grant review proceedings to patents in the United States or in countries outside the United States, or litigation against our collaborators may be costly and time consuming and could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition. We expect that litigation may be necessary in some instances to determine the validity and scope of our proprietary rights. Litigation may be necessary in other instances to determine the validity, scope or non-infringement of certain patent rights claimed by third parties to be pertinent to the manufacture, use or sale of our products. Ultimately, the outcome of such litigation, including our pending litigation with Purdue, could compromise the validity and scope of our patents or other proprietary rights or hinder our ability to manufacture and market our products.
If we are unable to obtain or maintain intellectual property rights for our technology,technologies, products and product candidates,or any products we may acquire, we may lose valuable assets or experience reduced market share.be unable to compete effectively in our market. We depend on our ability to protect our proprietary technology. We rely on patent and trademark laws, unpatented trade secrets and know-how, and confidentiality, licensing and other agreements with employees and third parties, all of which offer only limited protection. Our success depends in large part on our ability to obtain and maintain patent protection in the United States and other countries with respect to our proprietary technology and product candidates.products. The steps we have taken to protect our proprietary rights may not be adequate to preclude misappropriation of our proprietary information or infringement of our intellectual property rights both inside and outsidein the United States. The rights already granted under any of our currently issued patents and those that may be granted under future issued patents may not provide us with the proprietary protection or competitive advantages we are seeking. The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of inventions made in the course of our development and commercialization activities before it is too late to obtain patent protection on them.
Given the amount of time required for the development, testing and regulatory review of product candidates, patents protecting such product candidates might expire before or shortly after such product candidates are commercialized. If
we are unable to obtain and maintain patent protection for our technology and products, or if the scope of the patent protection obtained is not sufficient, our competitors could develop and commercialize technology and products identical, similar or superior to ours, and our ability to successfully commercialize our technology and products may be adversely affected.
With respect to patent rights, our patent applications may not issue into patents, and any issued patents may not provide protection against competitive technologies, may be held invalid or unenforceable if challenged or may be interpreted in a manner that does not adequately protect our technology, product candidates or future product candidates. Even if our patent applications issue into patents, they may not issue in a form that will provide us with any meaningful protection, prevent competitors from competing with us, or otherwise provide us with any competitive advantage. The examination process may require us to narrow the claims in our patents, which may limit the scope of patent protection that may be obtained. Our competitors may design around or otherwise circumvent patents issued to us or licensed by us.
The scope of patent protection in the United States and in foreign jurisdictions is highly uncertain, and changes in U.S. and foreign patent lawWe have increased that uncertainty and could diminish the value of patents in general, thereby impairing our ability to protect our product candidates and any future products.
The patent position of pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been, the subject of much litigation. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection. The laws of foreign countries may not protect our rights to the same extent as the laws of the United States, and these foreign laws may also be subject to change.
Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions typically are not published until 18 months after filing or, in some cases, not at all. Therefore, we cannot be certain that we were the first to make the inventions claimed in our owned or licensed patents or pending patent applications, or that we were the first to file for patent protection of such inventions. As a result, the issuance, scope, validity, enforceability and commercial value of our patent rights, both in the United States and abroad, are highly uncertain.
Recent patent reform legislation could increase the uncertainties and costs associated with the prosecution of our patent applications and the enforcement or defense of our issued patents. The Leahy-Smith America Invents Act, or the Leahy-Smith Act, which was signed into law on September 16, 2011, made significant changes to U.S. patent law, including provisions that affect the way patent applications are prosecuted and litigated. Many of the substantive changes to patent law associated with the Leahy-Smith Act and, in particular, the “first to file” provisions described below, only became effective on March 16, 2013. The Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Pursuant to the Leahy-Smith Act, the United States transitioned to a “first to file” system in which the first inventor to file a patent application will be entitled to the patent. In addition, third parties are allowed to submit prior art before the issuance of a patent by the U.S. Patent and Trademark Office, or USPTO, and may become involved in opposition, derivation, reexamination, or inter partes review challenging our patent rights or the patent rights of others. Grounds for a validity challenge could be an alleged failurecontinue to meet any of several statutory requirements, including novelty, nonobviousness and enablement. It is possible that prior art of which both we and the patent examiner were unaware during prosecution exists, which could render our patents invalid. Moreover, there may exist prior art of which we were or are aware, and which we did not or do not consider relevant to our patents, but which could nevertheless be determined to render our patents invalid. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights, which could have a material adverse effect on our competitive position with respect to third parties.
Because the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, issued patents that we own or license from third parties may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in the loss of patent protection, the narrowing of claims in such patents, or the invalidity or unenforceability of such patents, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection for our technology and products. Protecting against the unauthorized use of our patented technology, trademarks and other intellectual property
rights is expensive, difficult and, may in some cases not be possible. In some cases, it may be difficult or impossible to detect third party infringement or misappropriation of our intellectual property rights, even in relation to issued patent claims, and proving any such infringement may be even more difficult.
We may be, forced to litigate to enforce or defend our intellectual property, which could be expensive, time consuming and unsuccessful, and result in the loss of valuable assets.
We have been, and may continue to be, forced to litigate to enforce or defend our intellectual property rights against infringement and unauthorized use by competitors, and to protect our trade secrets. To counter infringement or unauthorized use,secrets, including in connection with our pending litigation may be necessary in the futureagainst generic competitors that have filed Paragraph IV Certifications relating to enforce or defend our intellectual property rights, to protect our trade secrets or to determine the validity and scopecertain of our own intellectual property rights.products. In so doing, we may place our intellectual property at risk of being invalidated, rendered unenforceable or limited or narrowed in scope. Further, this can be This litigation is expensive and time consuming. Many of our current and potential competitors have the ability to dedicate substantially greater resources to defend their intellectual property rights than we can.
Accordingly, despite our efforts, we may not be able to prevent third parties from infringing upon or misappropriating our intellectual property. Litigation could result in substantial costs and diversion of management resources, which could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition. In addition, an adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated, held unenforceable or interpreted narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of shares of our common stock. We may be subject to claims by third parties of ownership of what we regard as our own intellectual property or obligations to make compensatory payments to employees or others.
While it is our policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing or obtaining such an agreement with each party who, in fact, develops intellectual property that we regard as our own. In addition, they may breach the assignment agreements or such agreements may not be self-executing, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property. If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to commercialize our technology or products. Such a license may not be available on commercially reasonable terms or at all. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed. In addition to seeking patents for some of our technology products and product candidates,products, we rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts both within and outsidein the United States may be less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent such competitor, or those towith whom they communicate, with, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed or independently developed, our competitive position would be harmed. We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on all of our product candidates throughout the world would be prohibitively expensive. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop and sell their own products and, further, may export otherwise infringing products to territories where we have patent protection but enforcement is not as strong as that in the United States. These products may compete with our products in jurisdictions where we do not have any issued patents or our patent claims or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to pharmaceuticals, which could make it difficult for us to stop the infringement of our patents or the marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business.
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
Many of our employees, including our senior management, were previously employed at other biotechnology or pharmaceutical companies, including potential competitors. These employees typically executed proprietary rights, non-disclosure and non-competition agreements in connection with their previous employment. Although we try to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or these employees have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer. We are not aware of any threatened or pending claims related to these matters, but in the future litigation may be necessary to defend against such claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs, damage our reputation and be a distraction to management.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submissions, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements. The USPTOUnited States Patent and various foreign governmental patent agencies requireTrademark Office (“USPTO”) requires compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. In addition, periodic maintenance fees on issued patents are required to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patents. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees, and failure to properly legalize and submit formal documents. If we fail to maintain the patents and patent applications covering our product candidates,products, our competitive position would be adversely affected. Risks Related to the Commercialization of Our Product CandidatesProducts If we are unable to successfully develop and utilize our own sales and marketing capabilities successfully or enter into strategic alliances with marketing collaborators, we may not continue to be successful in commercializing Xtampza, the Nucynta Products and our product candidatesproducts and may be unable to generate sufficient product revenue. Our commercial organization continues to grow and evolve and in light of its short history and limited track record, we cannot guarantee that we will continue to be successful in marketing Xtampza, the Nucynta Products or any of our product candidates that may be approved for marketing.products. In addition, we will have to compete with other pharmaceutical and biotechnology companies with extensive and well-funded sales and marketing operations to recruit, hire, train and retain sales and marketing personnel. If we are unable to continue to grow and maintain adequate sales, marketing and distribution capabilities, whether independently or with third parties, including with respect to our recent acquisition of Belbuca and Symproic, we may not be able to generate sufficient product revenue and may not becomeremain profitable. Factors that may inhibit our efforts to commercializecontinue successfully commercializing our product candidatesproducts in the United States include: | ● | ·
| | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| ● | ·
| | the inability of sales personnel to obtain access toreach adequate numbers of physicians who may prescribe Xtampza, the Nucynta Productsour products; and our product candidates; |
| ● | ·
| | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and
|
| ·
| | unforeseen costs and expenses associated with creating and maintaining an independent sales and marketing organization. |
If we are not successful in recruiting and retaining sales and marketing personnel or in building amaintaining our sales and marketing infrastructure or if we do not successfully enter into appropriatepreserve strategic alliances with marketing collaborators, agreements with contract sales organizations or collaboration arrangements, we will have difficulty commercializing Xtampza, the Nucynta Products orin continuing to commercialize our product candidates. To the extent we commercialize Xtampza or our product candidates by entering into agreements with third-party collaborators, we may have limited or no control over the sales, marketing and distribution activities of these third parties, in which case our future revenues would depend heavily on the success of the efforts of these third parties.products. If physicians, patients, healthcare payors and the medical community, patients, and healthcare payors do not accept and use Xtampza, the Nucynta Products or our product candidates, if approved,products, we will not achieve sufficient product revenues and our business will suffer. Physicians patients, healthcare payors and others in the medical community, patients, and healthcare payors may not continue to accept and use Xtampza, the Nucynta Productsour products, or accept and use any of our product candidates(if regulatory approval is obtained), for whichnew products that we receive final regulatory approval.may acquire. Acceptance and use of Xtampza, the Nucynta Products and any product candidates for which we receive final regulatory approvalour products will depend on a number of factors including: | ● | ·
| | the timing of market introduction of our products and product candidates as well as competitive products;
|
| ·
| | approved indications, warnings and precautions language that may be less desirable than anticipated; competitive products; |
| ● | ·
| | perceptions by members of thephysicians and other healthcare community including physicians, aboutmembers of the safety and efficacy of Xtampza, the Nucynta Products and our product candidates; products; |
| ● | ·
| | perceptions by members of the healthcare community, including physicians, about the relevance and efficacy of our abuse deterrent technology in reducing potential riskstechnology; |
| ● | the availability of unintended use;competitive products; |
| ● | ·
| | published studies demonstrating the pricing and cost-effectiveness of Xtampza, the Nucynta Products and our product candidatesproducts relative to competing products;
|
| ● | ·
| | the potential and perceived advantages of Xtampza, the Nucynta Products and our product candidatesproducts over alternative treatments; |
| ● | ·
| | the convenience and ease of administration to patients of Xtampza, the Nucynta Products and our product candidates; products; |
| ● | ·
| | actual and perceived availability and quality of coverage and reimbursement for Xtampza, the Nucynta Products and our product candidatesproducts from government or other third-party payors; |
| ● | ·
| | any negative publicity related to our products or negative or positive publicity related to our competitors’ products that include the same active ingredient as Xtampza, the Nucynta Products and our product candidates;
products; |
| ● | ·
| | the prevalence and severity of adverse side effects, including limitationseffects; |
| ● | policy initiatives by FDA, HHS, DEA, or warnings contained in a product’s FDA approved product labeling;other federal or state agencies regarding opioids; |
| ● | ·
| | our ability to implement acomply with the Opioid Analgesic REMS; and |
| ● | ·
| | the effectiveness of marketing and distribution efforts by us and any licensees and distributors. |
If Xtampza, the Nucynta Products, or our product candidates for which we receive final regulatory approval,products fail to achievehave an adequate level of acceptance by physicians, healthcare payors, patients or the medical community, patients, or healthcare payors, we will not be able to generate significantsufficient revenue and we may not become orto remain profitable. Since we expect to rely on sales generated by Xtampza andER, the Nucynta Products, Belbuca, and Symproic for substantially all of our revenues for the foreseeable future, the failure of Xtampza or the Nucynta Productsthese products to findmaintain market acceptance would harm our business prospects. Recently enacted
Some of our products contain controlled substances, and the manufacture, use, sale, importation, exportation and distribution of which are subject to regulation by state and federal law enforcement and other regulatory agencies. Some of our products contain controlled substances that are subject to state and federal laws and regulations regarding their manufacture, use, sale, importation, exportation and distribution. Xtampza ER’s active ingredient, oxycodone, and the Nucynta Products’ active ingredient, tapentadol, are both classified as Schedule II controlled substances under the CSA and regulations of the DEA and the active ingredient in Belbuca, buprenorphine, is classified as a Schedule III controlled substance. A number of states also independently regulate these drugs, including oxycodone, tapentadol and buprenorphine, as controlled substances. We and our suppliers, manufacturers, contractors, customers and distributors are required to obtain and maintain applicable registrations from state and federal law enforcement and regulatory agencies and comply with state and federal laws and regulations regarding the manufacture, use, sale, importation, exportation and distribution of controlled substances. Furthermore, the amount of Schedule II substances that can be obtained for clinical trials and commercial distribution is limited by the CSA and DEA regulations. For more information, refer to the section in our Annual Report entitled “Business — Government Regulation — DEA and Opioid Regulation.” We may not be able to obtain sufficient quantities of these controlled substances in order to meet commercial demand. If commercial demand for Xtampza ER, or any of our other approved products, increases and we cannot meet such demand in a timely fashion because of our limited supply of its active pharmaceutical ingredient (in the case of Xtampza ER, oxycodone) then physicians may perceive such product as unavailable and may be less likely to prescribe it in the future. In addition, controlled substances are also subject to regulations governing manufacturing, labeling, packaging, testing, dispensing, production and procurement quotas (for Schedule I and II substances), recordkeeping, reporting, handling, shipment and disposal. These regulations increase the personnel needs and the expense associated with commercialization of our products that include controlled substances. The DEA and some states conduct periodic inspections of registered establishments that handle controlled substances. Failure to obtain and maintain required registrations or to comply with any applicable regulations could delay or preclude us from manufacturing and commercializing our products that contain controlled substances and subject us to enforcement action. The DEA may seek civil penalties, refuse to renew necessary registrations or initiate proceedings to revoke those registrations. In some circumstances, violations could lead to criminal proceedings. Because of their restrictive nature, these regulations could limit commercialization of our products containing controlled substances. Current and future legislation may increase the difficulty and cost for us to continue to commercialize Xtampza, the Nucynta Products, and our product candidatesproducts and may reduce the prices we are able to obtain for our products. In the United States, and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system generally, and the manufacturing, distribution, and marketing of opioids in particular, that could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities or affect our ability to profitably sell Xtampza,commercialize our products. For example, several states, including New York, have imposed taxes or fees on the Nucynta Products,sale of opioids. Other states, and even the federal government, could impose similar taxes or any product candidates for which we obtain marketing approval. Cost reduction legislation could decrease the coveragefees, and price that we receive for any approved products, including for reimbursement through Medicaresuch laws and private payors.
The pricing of pharmaceutical products, in general, and specialty drugs, in particular, has also been a topic of concernproposals can vary in the U.S. government. There can be no assurance astax and fee amounts imposed and the means of calculation. Liabilities for taxes or assessments under any such laws could have an adverse impact on our results of operations.
California and several other states have enacted legislation related to howprescription drug pricing transparency and it is unclear the effect this scrutinylegislation will have on pricing of pharmaceutical products will impact future pricing of our products or pharmaceutical products generally. The current administration has indicated that reducing the price of prescription drugs will be a priority of the administration. The implementation of any price controls on prescription drugs, whether at the federal or state level, may adversely affect our business, operating results and financial condition. business. Laws intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for health care and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms may continue the downward pressure on pharmaceutical pricing, especially under the Medicare program, and may also increase our regulatory burdens and operating costs. The Affordable Care Act, as well as other healthcare reform measures that have been and may be adopted in the future, may result in more rigorous coverage criteria, new payment methodologies and in additional downward pressure on the price that we receive for any approved product, and could seriously harm our future revenues. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may compromise our ability to generate revenue, attain profitability or commercialize our products. At the same time, there have been significant ongoing efforts to modify or eliminate the Affordable Care Act. For example, the TCJA, enacted on December 22, 2017, repealed the shared responsibility payment for individuals who fail to maintain minimum essential coverage under section 5000A of the Internal Revenue Code, commonly referred to as the individual mandate, beginning in 2019. The Joint Committee on Taxation estimates that the repeal will result in over 13 million Americans losing their health insurance coverage over the next ten years and is likely to lead to increases in insurance premiums. Further legislative changes to and regulatory changes under the Affordable Care Act remain possible. It is unknown what form any such changes or any law proposed to replace the Affordable Care Act would take, and how or whether it may affect our business in the future.
Newly enacted FDA regulations may require us to expend additional resources to obtain or maintain regulatory approval. For example, in August 2017 President Trump signed into law the Food & Drug Administration Reauthorization Act (FDARA). This legislation imposes significant new requirements for clinical trial sponsors which will affect, among other things, the development of drugs and biological products for pediatric use. This legislation may result in new regulations, which may affect future options or timelines for regulatory approval.
Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. We cannot be sure whether additional legislative changes will be enacted, or whether the FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals of our product candidates, if any,products may be. In addition, increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post-marketing testing and other requirements. On February 27, 2018, a bipartisan group of senators introduced Senate Bill 2456 (S.2456). S.2456 is characterized as “CARA 2.0,” in reference to the Comprehensive Addiction and Recovery Act of 2016. CARA 2.0 would limit initial prescriptions for opioids to 3 days, while exempting initial prescriptions for chronic care, cancer care, hospice or end of life care, and palliative care. CARA 2.0 would also increase civil and criminal penalties for opioid manufacturers that fail to report suspicious orders for opioids or fail to maintain effective controls against diversion of opioids. The bill would increase civil fines from $10,000 to $100,000, and if a manufacturer fails to maintain effective controls or report suspicious orders with knowledge or willful disregard, the bill would double criminal penalties from $250,000 to $500,000. If this bill were signed into law, it could adversely affect our ability to successfully commercialize Xtampza, the Nucynta Products, and our product candidates if approved. In addition, in 2017 several states, including Indiana, Louisiana, and Utah, enacted laws that further limit or restrict opioid prescriptions.
In addition, state pharmacy laws may permit pharmacists to substitute generic products for branded products if the products are therapeutic equivalents, or may permit pharmacists and pharmacy benefit managers to seek prescriber authorization to substitute generics in place of Xtampza or our product candidates, which could significantly diminish demand for them and significantly impact our ability to successfully commercialize our products and generate revenues.
Even if we are able to commercialize Xtampza,the Nucynta Products, and any of our product candidates, ourOur products may become subject to unfavorable pricing regulations or third-party coverage and reimbursement policies, which could have a material adverse effect on our business. Such pricing regulations may address the rebates that manufacturers offer to pharmaceutical benefit managers, or the discounts that manufacturers provide others within the pharmaceutical distribution chain.
The regulations that govern marketing approvals, pricing and reimbursement for new drug products can vary widely from country to country.widely. Current and future legislation may significantly change the approval requirements in ways that could involve additional costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we might obtain marketing approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product, possibly for lengthy time periods, which could negatively impact the revenues we are able to generate from the sale of the product in that particular country. Pricing limitations may hinder our ability to recoup our investment in Xtampza, the Nucynta Products, and our product candidates even if our product candidates obtain marketing approval.products. Our ability to commercialize any product successfully will also depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third-party payors such as private health insurers and health maintenance organizations, determine which medications they will cover and establish reimbursement levels.levels and tiers of preference based on the perceived value and innovation of a given product. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications.medications and establishing administrative hurdles that incentivize use of generic and/or lower cost products first. Increasingly, third-party payors are requiring that drug companies provide them with discounts and rebates from list prices and are challenging the prices charged for medical products. We have agreed to provide such discounts and rebates to certain third-party payors. We expect increasing pressure to offer larger discounts and rebates. Additionally, a greater number of third-party payors may seek discounts and rebates in order to offer or maintain access for Xtampza, the Nucynta Products and our product candidates, if approved.products. We cannot be sure that high-quality coverage and reimbursement will be available for any product that we commercialize and, if reimbursement is available, what the level of reimbursement will be and whether it will be satisfactory. Coverage and reimbursement may impact the demand for, or the price of, any product candidate for which we obtain marketing approval. If coverage and reimbursement are not available or reimbursement is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval. There may be significant delays in obtaining coverage and reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA or foreign regulatory authorities. Moreover, eligibility for coverage and reimbursement does not imply that any drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient to cover our costs and may only be temporary. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost drugs and may be incorporated into existing payments for other services. Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from policy and payment limitations in setting their own reimbursement policies.
In August 2022, the Inflation Reduction Act of 2022 was signed into law. This legislation contains substantial drug pricing reforms, including the establishment of a drug price negotiation program within the U.S. Department of Health and Human Services that would subject manufacturers of some brand-name medications without generic or biosimilar competition to a price negotiation program that results in a negotiated “maximum fair price” (or pay an excise tax for noncompliance), the establishment of rebate payment requirements on manufacturers of drugs payable under Medicare Parts B and D to penalize price increases that outpace inflation, and revises the way manufacturers provide discounts on Part D drugs. The IRA also caps Medicare beneficiaries’ annual out-of-pocket drug expenses at $2,000 per year, thereby eliminating the Medicare Part D coverage gap or “donut hole.” Substantial penalties can be assessed for noncompliance with the drug pricing provisions in the IRA. The IRA could have the effect of reducing the prices we can charge and reimbursement we receive for our products, thereby reducing our profitability, and could have a material adverse effect on our financial condition, results of operations and growth prospects. The effect of the IRA on our business and the pharmaceutical industry in general is not yet known. Our inability to promptly obtainexpand and maintain coverage and profitable reimbursement rates from both government-funded and private payors for any approvedour products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to continue to commercialize our products and our overall financial condition. The Affordable Care Act and any changes in healthcare law may increase the difficulty and cost for us to continue to commercialize our products and affect the prices we may obtain. The United States and many foreign jurisdictions have enacted or proposed legislative and regulatory changes affecting the healthcare system that may affect our ability to profitably sell our products, including implementing cost-containment programs to limit the growth of government-paid healthcare costs, including price controls, restrictions on reimbursement and requirements for substitution of generic products for branded prescription drugs. The Affordable Care Act was intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. There have been significant ongoing judicial, administrative, executive and legislative efforts to modify or eliminate the Affordable Care Act, and the Affordable Care Act has also been subject to challenges in the courts. Refer to the section in our Annual Report entitled “Business — Government Regulation — Healthcare Reform.” Further changes to and under the Affordable Care Act remain possible. It is unknown what form any such changes or any law proposed to replace the Affordable Care Act would take, and how or whether it may affect our business in the future. We expect that changes to the Affordable Care Act, the Medicare and Medicaid programs, changes allowing the federal government to directly negotiate drug prices and changes stemming from other healthcare reform measures, especially with regard to healthcare access, financing or other legislation in individual states, could have a material adverse effect on the healthcare industry. Any reduction in reimbursement from Medicare, Medicaid, or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue and maintain profitability. Social issues around the abuse of opioids, including law enforcement concerns over diversion of opioids and regulatory and enforcement efforts to combat abuse, could decrease the potential market for Xtampza, the Nucynta Products,our products and may adversely impact external investor perceptions of our product candidates.business. Media stories regarding prescription drug abuse and the diversion of opioids and other controlled substances are commonplace.
Law enforcement and regulatory agencies may apply policies and guidelines that seek to limit the availability or use of opioids. Such efforts may inhibit our ability to continue to commercialize Xtampza, the Nucynta Products, and our product candidates.products. Aggressive enforcement and unfavorable publicity regarding, for example, the use or misuse of oxycodone or other opioid drugs; the limitations of abuse-resistant formulations; the ability of drug abuserspeople who abuse drugs to discover previously unknown ways to abuse opioid drugs, including Xtampza andER, the Nucynta Products;Products and Belbuca; public inquiries and investigations into prescription drug abuse; litigation; or regulatory activity regarding sales, marketing, distribution or storage of opioid drugs could have a material adverse effect on our reputation. Such negative publicity could reduce the potential size of the market for Xtampza, the Nucynta Products, and our product candidates andproducts, decrease the revenues we are able to generate from their sale.sale and adversely impact external investor perceptions of our business. Similarly, to the extent opioid abuse becomes less prevalent or less urgent of a public health issue, regulators and third party payersthird-party payors may not be willing to pay a premium for abuse-deterrent formulations of opioids. Efforts by the FDA and other regulatory bodies to combat abuse of opioids may negatively impact the market for our product and product candidates. In February 2016, the FDA released an action plan
Federal laws have been enacted to address the national epidemics of prescription opioid abuse epidemic and reassess the FDA’s approach to opioid medications. The plan identifies the FDA’s focus on implementing policies to reverse the opioid abuse epidemic, while maintaining access to effective treatments. The actions set forth in the FDA’s plan include strengthening post marketing study requirements to evaluate the benefit of long-termillicit opioid use, changingincluding the REMS requirements to provide additional fundingComprehensive Addiction and Recovery Act and the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for physician education courses, releasing a draft guidance setting forth approval standards for generic-abuse deterrent opioid formulations,Patients and seeking input fromCommunities Act. These laws are described in more detail in our Annual Report under the FDA’s Science Board to broaden the understanding of the public risks of opioid abuse. The FDA’s Science Board met to address these issues on March 1, 2016. The FDA’s plan is part of a broader initiative led by the HHS to address opioid-related overdose, deathcaption “Business — Government Regulation — DEA and dependence. The HHS initiative’s focus is on improving physician’s use of opioids through education and resources to address opioid over-prescribing, increasing use and development of improved delivery systems for naloxone, which can reverse overdose from both prescription opioids and heroin, to reduce overdose-related deaths, and expanding the use of Medication-Assisted Treatment, which couples counseling and behavioral therapies with medication to address substance abuse. Also as part of this initiative, the CDC has launched a state grant program to offer state health departments resources to assist with abuse prevention efforts, including efforts to track opioid prescribing through state-run electronic databases. In March 2016, as part of the HHS initiative, the CDC released a Guideline for Prescribing Opioids for Chronic Pain. The guideline is intended to assist primary care providers treating adults for chronic pain in outpatient settings. The guideline provides recommendations to improve communications between doctors and patients about the risks and benefits of opioid therapy for chronic pain, improve the safety and effectiveness of pain treatment, and reduce the risks associated with long-term opioid therapy. The guideline states that no treatment recommendations about the use of abuse-deterrent opioids can be made at this time.Opioid Regulation.”
The FDA continues to evaluate extended release and abuse-deterrent opioids in the postmarket setting. In March 2017, the FDA’s Advisory Committee met to discuss OPANA ER (oxymorphone hydrochloride) extended release tablets. A majority of the Advisory Committee voted that the benefits do not outweigh the risks of OPANA ER. Upon the FDA’s subsequent request in June 2017, OPANA ER was removed from the market. Also, in July 2017, the FDA held a public workshop to discuss available data and methods to assess the impact of opioid formulations with abuse-deterrent properties on misuse, abuse, addiction, overdose, and death in the postmarket context. The FDA will continue to scrutinize the impact of abuse-deterrent opioids and in the future could impose further restrictions to products currently on the market, which may include changing labeling, imposing additional prescribing restrictions, or seeking a product’s removal from the market.
The DEA continues to increase its efforts to hold manufacturers, distributors, prescribers and pharmacies accountable through various enforcement actions as well as the implementation of compliance practices for controlled substances. In addition, many state legislatures are considering various bills intended to reduce opioid abuse, for example by establishing prescription drug monitoring programs and mandating prescriber education. Further, the FDA is requiring “black-box” warnings on immediate release opioids highlighting the risk of misuse, abuse, addiction, overdose and death. In addition, during the 2016 presidential campaign, as well as implementing a REMS for immediate release opioids, many elected officials, including President Trump, called for the DEA to restrict the amount of opioids that can be manufactured in the U.S. The DEA recently proposed reducing the quota for controlled substances to be manufactured in the U.S. in 2018. In March 2017, President Trump announced the creation of a commission, through ONDCP, to make recommendations to the president on how to best combat opioid addiction and abuse. In August 2017, the commission issued a preliminary report calling on President Trump to officially declare the crisis of opioid abuse a national emergency. On October 26, 2017, President Trump declared the opioid crisis a “national public health emergency”. The commission’s final report was released in early November 2017.
Recently, CVS Pharmacy announced it would only fill first-time opioid prescriptions for acute pain for a seven day supply. In July 2017, the Pharmaceutical Care Management Association, a trade association representing pharmacy benefit managers, wrote a letter to the commissioner of FDA in which it expressed support for, among other things, the CDC guidelines and a seven-day limit on the supply of opioids for acute pain. In addition, states, including the Commonwealths of Massachusetts and Virginia and the States of New York, Ohio, Arizona, Maine, New Hampshire, Vermont, Rhode Island, Colorado, Wisconsin, Alabama, South Carolina, Washington and New Jersey, have either recently enacted, intend to enact, or have pending legislation or regulations designed to, among other things, limit the duration and quantity of initial prescriptions of immediate release forms of opiates and, mandate the use by prescribers of prescription drug databases and mandate prescriber education. Also, at the state and local level, a number of states and cities have brought separate lawsuits against various pharmaceutical companies marketing and selling opioid pain medications, alleging misleading or otherwise improper promotion of opioid drugs to physicians and consumers. In addition, the attorneys general from several states have announced the launch of a joint investigation into the marketing and sales practices of drug companies that market opioid pain medications. Many of these changes and others could cause us to expend additional resources in developing and commercializing Xtampza, the Nucynta Products, and our product candidates to meet additional requirements. Advancements in development and approval of generic abuse-deterrent opioids could also compete with and potentially impact physician use of our product candidates and cause our product candidates to be less commercially successful.
If the FDA or other applicable regulatory authorities approve generic products with abuse deterrent claims that compete with Xtampza, the Nucynta Products, or any of our product candidates, itproducts, our sales could reduce our sales.decline. Once an NDA, including a Section 505(b)(2) application, is approved, the product covered thereby becomes a “listed drug” which can, in turn, be cited by potential competitors in support of approval of an abbreviated NDA, or ANDA. The FD&CFederal Food, Drug, and Cosmetic Act, FDA regulations and other applicable regulations and policies provide incentives to manufacturers to create modified, non-infringing versions of a drug to facilitate the approval of an ANDA or other application for generic substitutes. These manufacturers might only be required to conduct a relatively inexpensive study to show that their product has the same active ingredients, dosage form, strength, route of administration, and conditions of use, or product labeling, as our product and that the generic product is absorbed in the body at the same rate and to the same extent as, or is bioequivalent to, our product. These generic equivalents would be significantly less costly than ours to bring to market and companies that produce generic equivalents are generally able to offer their products at lower prices. Additionally, under FDORA, FDA will assign therapeutic equivalence ratings for certain prescription drugs approved via the Section 505(b)(2) NDA pathway with respect to other approved drug products and it is unclear how assignment of these ratings will impact the market opportunity for our products. Thus, after the introduction of a generic competitor, a significant percentage of the sales of any branded product are typically lost to the generic product. Accordingly, competition from generic equivalents to our products would substantially limit our ability to generate revenues and therefore, to obtain a return on the investments we have made in our productproducts. In the past, we have initiated litigation with generic competitors that have filed Paragraph IV Certifications challenging certain of our patents. While we have entered into settlement agreements with certain competitors, we are currently pursuing litigation to defend against Paragraph IV Certifications related to Belbuca. Refer to Note 13, Commitments and product candidates.Contingencies, to our consolidated financial statements included in Part IV of this Annual Report on Form 10-K. We believe that we will continue to be subject to ANDA-related litigation, which can be costly and distracting and has the potential to impact the long-term value of our products. Guidelines
We may seek FDA pediatric exclusivity for some of our products. Pediatric exclusivity, if granted, adds six months of patent term and recommendations publishedmarketing exclusivity to existing exclusivity periods for all formulations, dosage forms, and indications for the active moiety, provided that at the time pediatric exclusivity is granted there is not less than nine months of term remaining. The regulatory exclusivity period for Nucynta IR in the United States has been extended through July 3, 2026, following the grant of New Patient Population exclusivity in pediatrics by various organizations can reducethe FDA in August 2023 based on data from pediatric trials which were submitted in response to the FDA's Pediatric Written Request to evaluate the use of our products,Nucynta as a treatment for pain in pediatric patients aged 6 years and older. If FDA deems these data to be responsive to its Written Request, the exclusivity of the entire Nucynta franchise could be extended an additional six months, to December 2025 for Nucynta ER and January 2027 for Nucynta IR. However, there is no guarantee that FDA will agree that the Written Request has been satisfied and that we will receive this additional exclusivity, or that we will maintain such exclusivity, if approved.granted. Government agencies promulgate regulations and guidelines directly applicable
In November 2017, the FDA issued a final guidance to us andassist industry in the development of generic versions of approved opioids with abuse-deterrent formulations, including recommendations about the types of studies that companies should conduct to demonstrate that the generic drug is no less abuse-deterrent than its brand-name counterpart. In the second half of 2018, the FDA posted three revised product-specific guidances related to generic abuse-deterrent opioid formulations, including one guidance specifically relating to Xtampza ER, which recommend specific in vivo studies and in vitro study considerations for abuse deterrence evaluations. These guidances are part of the Nucynta Products, andFDA’s wider focus on assisting developers of generic abuse-deterrent formulations in navigating the regulatory path to market more quickly. Earlier market entry of generic abuse-deterrent formulations could have a material adverse effect on our product candidates. In addition, professional societies, practice management groups, private health and science foundations and organizations involved in various diseases from time to time may also publish guidelines or recommendations to the healthcare and patient communities. Recommendations of government agencies or these other groups or organizations may relate to such matters as usage, dosage, route of administration and use of concomitant therapies. Recommendations or guidelines suggesting the reduced use of our products or the use of competitive or alternative products as the standard of care to be followed by patients and healthcare providers could result in decreased use of our products.business. Risks Related to Our Dependence on Third Parties If the third-party manufacturermanufacturers of Xtampza failsour products fail to devote sufficient time and resources to Xtampza,these products, or itstheir performance is substandard, and/or we encounter challenges with our dedicated manufacturing suite at our third-party manufacturer’s site for the manufacturing of Xtampza ER, our costs may be higher than expected and could have a material adverse effect on our business. Our commercialization partner also relies on a sole supplier to manufacture Nucynta ER, which presents a similar risk. We do not own any manufacturing facilities and have limited experience in drug development and commercial manufacturing. We currently have no plans to build our own clinical or commercial scale manufacturing facility. We lackfacility and do not have the resources and expertise to manufacture and test, on a commercial scale, the technical performance of Xtampza and our product candidates.products. We currently rely, and expect to continue to rely, on a limited number of experienced personnel and one contract manufacturermanufacturers for Xtampza and each product candidate,our products, as well as other vendors to formulate, test, supply, store and distribute Xtampza and our product candidates for our clinical trials and FDA registration,products, and we control only certain aspects of their activities. In 2020, we completed the build-out of a dedicated manufacturing suite for Xtampza ER at a site operated by our contract manufacturing organization, Patheon, part of Thermo Fisher Scientific. This facility requires the maintenance of regulatory approvals and other costs, all of which we absorb. We cannot guarantee that we will be able to continue to leverage the dedicated manufacturing suite in a profitable manner. If the demand for Xtampza ER and any future related products never meets our expectations and forecasts, or if we do not produce the output we plan, we may not be able to realize the return on investment we anticipated, which would have a negative impact on our financial condition and results of operations. We have also transitioned commercial manufacturing for Nucynta ER from Janssen to Patheon. While we were successful in our regulatory approval and validation activities, we could encounter issues in obtaining commercial supply from Patheon's facility due to technical problems or challenges obtaining adequate and/or timely DEA procurement quota. Although we have identified alternate sources for these services, it would be time-consuming, and require us to incur additional cost,costs, to qualify these sources. Our reliance on a limited number of vendors and, in particular, Patheon as our single manufacturer for Xtampza ER and Nucynta ER, exposes us to the following risks, any of which could delay FDA approval of our product candidates andimpact commercialization of our products, result in higher costs, or deprive us of potential product revenues: | ● | ·
| | Our contract manufacturer, or other third parties we rely on, may encounter difficulties in achieving the volume of production needed to satisfy commercial demand, may experience technical issues that impact quality or compliance with applicable and strictly enforced regulations governing the manufacture of pharmaceutical products, may be affected by natural disasters that interrupt or prevent manufacturing of our products, may experience shortages of qualified personnel to adequately staff production operations, may experience shortages of raw materials and may have difficulties finding replacement parts or equipment. equipment; |
| ● | ·
| | Our contract manufacturersmanufacturer could default on their agreementsagreement with us to meet our requirements for commercial supplies of Xtampza. our products and/or we could experience technical problems in the operation of our dedicated manufacturing suite; |
| ● | ·
| | The use of alternate manufacturers may be difficult because the number of potential manufacturers that have the necessary governmental licenses to produce narcotic products is limited. Additionally, the FDA and the DEA must approve any alternative manufacturer of Xtampza or any product candidate for which we receive regulatory approval,our products, before we may use the alternative manufacturer to produce commercial supplies. supplies; |
| ● | ·
| | It may be difficult or impossible for us to find a replacement manufacturer on acceptable terms quickly, or at all. Our contract manufacturer and vendors may not perform as agreed or may not remain in the contract manufacturing business for the time required to successfully produce, store and distribute our products. products successfully; and |
| ● | ·
| | If our contract manufacturer were to terminate our arrangement or fail to meet our commercial manufacturing demands, we may be forced to delay our development and commercial programs. |
Failure to obtain the necessary active pharmaceutical ingredients, excipients or components necessary to manufacture our products could adversely affect our ability to continue to commercialize our products, which could in turn adversely affect our results of operations and financial condition. Likewise, the inability of any of our sole or limited suppliers to provide components that meet our specifications and requirements could adversely impact our ability to manufacture our products. In addition, DEA regulations, through the quota procurement process, limit the amount of DEA-controlled active pharmaceutical ingredient we have available for manufacture. Consequently, we are limited in our ability to maintain an appreciable safety stock of finished drug product. Our reliance on third parties reduces our control over our developmentmanufacturing and commercialization activities but does not relieve us of our responsibility to ensure compliance with all required legal, regulatory and scientific standards. The FDA and other regulatory authorities require that Xtampza and our product candidates that we may eventually commercializeproducts to be manufactured according to cGMP and similar foreign standards.cGMP. Any failure by our third-party manufacturer to comply with cGMP or failure to scale up manufacturing processes, including any failure to deliver sufficient quantities of product candidatesproducts in a timely manner, could lead to inspection deficiencies, a delay in,shortage of commercial product, or failure to obtain, regulatory approval ofpotential products liability exposure for any of our product candidates. In addition, suchnoncompliant distributed products. Such failure could also be the basis for the FDA to issue a warning or untitled letter, withdraw approvals for products previously granted to us, or take other regulatory or legal action, including recall or seizure, total or partial suspension of production, suspension of ongoing clinical trials, refusal to approve pending applications or supplemental applications, detention orof product, refusal to permit the import or export of products, injunction, imposing civil penalties or pursuing criminal prosecution. Our commercialization partner currently relies on a single supplier to manufacture each of the Nucynta Products.
Any stock out, or failure to obtain sufficient supplies of eachany of the Nucynta Products,our products, or the necessary active pharmaceutical ingredients, excipients or components necessary to manufacture each of the Nucynta Products,our products, could adversely affect our ability to commercialize the Nucynta Products,such products, which could in turn adversely affect our results of operations and financial condition. Our commercialization partner, Depomed, experienced delays in the manufacture, packaging and delivery of certain dosage strengths of Nucynta ER in the third and fourth quarters of 2017 and the first quarter of 2018 following Hurricanes Irma and Maria in Puerto Rico. We and our commercialization partner may continue to experience further outages in the future. Because we currently rely on a sole supplier or limited number of suppliers to manufacture the active pharmaceutical ingredient of Xtampza,our products, any production problems with our supplierany of these suppliers could have a material adverse effect on us. We presently depend uponcurrently rely on a singlesole supplier foror limited number of suppliers to manufacture the active ingredient for Xtampza — oxycodone base — and we intend topharmaceutical ingredients of our products. We contract with this supplier, as necessary,these suppliers for commercial supply ofto manufacture our products. Although we have identified anFurther, our suppliers for Xtampza ER and the Nucynta Products active pharmaceutical ingredients also supply our primary competitor in the extended-release oxycodone space, Purdue. Identifying alternate sourcesources of active pharmaceutical ingredients for oxycodone base, it would beour products is generally time-consuming and costlycostly. Any changes that our suppliers make to qualify this source. Since we currently obtainthe respective drug substance raw materials, intermediates, or manufacturing processes would introduce technical and regulatory risks to our active ingredient from this manufacturer on a purchase-order basis, either we or our supplier may terminate our arrangement, without cause, at any time without notice.downstream drug product supply. If our suppliersuppliers were to terminate ouran arrangement for an active pharmaceutical ingredient, or fail to meet our supply needs (including as a result of any disruptions in personnel or the global supply chain), we might incur substantial costs and be forced to delay our development or commercialization programs. Any such delay could have a material adverse effect on our business. We rely on third parties to conduct
Global supply chain disruptions and shortages may limit manufacturing and commercial supply of our clinical trials. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, or if they terminate their agreement with us, we may not be able to obtain regulatory approval for or commercialize our product candidatesproducts and our business could sufferhave a material adverse effect.impact on our business. We have relied upon
There are currently global supply chain disruptions and plan to continue to rely upon contract research organizations, or CROs, to monitor and manage data for our ongoing preclinical and clinical programs. We rely on these parties for executionshortages caused by a variety of our clinical trials, and control only certain aspects of their activities. Nevertheless, we are responsible for ensuring that each of our studies and clinical trials are conducted in accordance withfactors, including geopolitical turmoil, such as the applicable protocol, legal, regulatory and scientific standards, and our reliance on the CROs does not relieve us of our regulatory responsibilities. We and our CROs are required to comply with federal regulationsUkrainian War and current Good Clinical Practices, or GCP, which are international standards meant to protect the rightsconflict in Israel and health of patients and to define the roles of clinical trial sponsors, advisors and monitors, enforced by the FDA, the Competent Authorities of the Member States of the European Economic Area, or EEA, and foreign regulatory authorities in the form of International Conference on Harmonization, or ICH, guidelines for all of our product candidates in clinical development. Regulatory authorities enforce these GCP through periodic inspections of trial sponsors, principal investigators and trial sites. In addition,Gaza. While we and our CROssuppliers are requiredstill able to comply with special regulations regarding the enrollment of recreational drug abusers in clinical trials. If we or any of our CROs fail to comply with applicable GCP and other regulations, including as a result of any recent changes in such regulations, the clinical data generated in our clinical trials may be deemed unreliable and the FDA or foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical trials comply with GCP requirements. In addition, our clinical trials must be conducted with product produced under cGMP requirements. While we have agreements governing activities of our CROs, we have limited influence over their actual performance. Failure to comply with applicable regulations in the conductreceive sufficient inventory of the clinical trials forkey materials and components needed, we could experience pressure on our product candidates may require us to repeat preclinical studiessupply chain, including shipping delays, higher prices from suppliers, and clinical trials, which would delay the regulatory approval process.
Our CROs arereduced availability of materials, including excipients and packaging components. To date, supply chain pressure has not had a material impact on our employees,results of operations. However, if these disruptions and except for remedies available to us under our agreements with such CROs, we cannot control whether or not they devote sufficient time and resources to our ongoing clinical and preclinical programs. If CROs do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they need to be replaced or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated andshortages continue, we may not be able to obtain regulatory approval for or successfully commercialize our product candidates. As a result, the commercial prospects for our product candidates would be harmed, our costs could increase substantially and our ability to generate revenue could be delayed.
Switching or adding additional CROs involves additional cost and requires management time and focus, and there is a limited number of CROs that are equipped and willing to manage clinical trials that involve recreational drug abusers. Our CROs have the right to terminate their agreements with us in the event of an uncured material breach. In addition, some of our CROs have an ability to terminate their respective agreements with us if it can be reasonably demonstrated that the safety of the patients participating in our clinical trials warrants such termination, if we make a general assignment for the benefit of our creditors or if we are liquidated. Identifying, qualifying and managing performance of third-party service providers can be difficult, time-consuming and cause delays in our development programs. In addition, there is a natural transition period when a new CRO commences work and the new CRO may not provide the same type or level of services as the original provider. Though we carefully manage our relationships with our CROs, there can be no assurance that we will not encounter challenges or delays in the future or that these delays or challenges will notexperience a material interruption to our supply chain. Such an interruption could have a material adverse impact on our business, financial condition and prospects. If any of our relationships with our CROs terminate, we mayincluding but not be ablelimited to, enter into arrangements with alternative CROs or to do so on commercially reasonable terms. As a result, delays may occur, which can materially impact our ability to meet our desired clinical development timelines.
Our internal capacity to perform these functions is limited. Outsourcing these functions involves risks that third parties may not perform to our standards, may not produce results in a timely manner or may fail to perform at all. In addition, the use of third-party service providers requires us to disclose our proprietary information to these parties, which could increase the risk that this information will be misappropriated. We currently have a small number of employees, which limits the internal resources we have available to identifymanufacture and monitor our third-party providers. To the extent, we are unable to identify and successfully manage the performance of third-party service providers in the future, our ability to advance our product candidates through clinical trials will be compromised. There can be no assurance that we will not encounter similar challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition and prospects.
In the future, we may depend on collaborations with third parties for the development and commercialization of Xtampza, the Nucynta Products and our product candidates. If those collaborations are not successful, we may not be able to capitalize on the market potential of these product candidates.
We may not be successful in establishing development and commercialization collaborations which could adversely affect, and potentially prohibit, our ability to develop or commercialize Xtampza, our products and our product candidates. These collaborations, including the Commercialization Agreement for Nucynta ER and Nucynta IR, pose the following risks to us:
| ·
| | Collaborators may have significant discretion in determining the efforts and resources that they will apply to these collaborations.
|
| ·
| | Collaborators may not pursue development and commercialization of our product or product candidates or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator’s strategic focus or available funding or external factors such as an acquisition that diverts resources or creates competing priorities.
|
| ·
| | Collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon our product or product candidate, repeat or conduct new clinical trials or require a new formulation of our product or product candidate for clinical testing.
|
| ·
| | Collaborators may conduct clinical trials inappropriately, or may obtain unfavorable results in their clinical trials, which may have an adverse effect on the development or commercialization of our product or product candidates.
|
| ·
| | Collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product or product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours.
|
| ·
| | A collaborator with marketing and distribution rights to one or more products may not commit sufficient resources to the marketing and distribution of such products.
|
| ·
| | Collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation.
|
| ·
| | Disputes may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization of our product and product candidates or that result in costly litigation or arbitration that diverts management attention and resources.
|
| ·
| | We may lose certain valuable rights under circumstances specified in our collaborations.
|
| ·
| | Collaborations may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product or product candidates.
|
| ·
| | Collaboration agreements may not lead to development or commercialization of products or product candidates in the most efficient manner or at all. If a future collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product development or commercialization program under such collaboration could be delayed, diminished or terminated.
|
| ·
| | Our ability to successfully commercialize product candidates pursuant to collaboration agreements may be adversely affected by disputes or delays arising from supply and/or manufacturing agreements between such collaborators and third parties—agreements to which we may not be a party.
|
We may rely on collaborators to market and commercialize our products, and, if approved, our product candidates, who may fail to effectively commercializedistribute our products.
We may utilize strategic collaborators or contract sales forces, where appropriate, to assist in the commercialization of Xtampza, the Nucynta Products and our product candidates, if approved by the FDA. We currently possess limited resources and may not be successful in establishing collaborations or co-promotion arrangements on acceptable terms, if at all. We also face competition in our search for collaborators and co-promoters. If we enter into strategic collaborations or similar arrangements, we will rely on third parties for financial resources and for development, commercialization, sales and marketing and regulatory expertise. Our collaborators, if any, may fail to develop or effectively commercialize our products and product candidates because they cannot obtain the necessary regulatory approvals, they lack adequate financial or other resources or they decide to focus on other initiatives. Any failure of our third-party collaborators to successfully market and commercialize our product and product candidates would diminish our revenues.
Manufacturing issues may arise that could increase product and regulatory approval costs, delay commercialization or limit commercial supply. As
In our current commercial manufacturing operations, and as we scale up manufacturing of our products and product candidates and conduct required stability testing, we may encounter product, packaging, equipment and process-related issues that may require refinement or resolution in order to proceed withsuccessfully commercialize our planned clinical trials, obtain regulatory approval for commercial marketing and build commercial supplies.products. In the future, we may identify impurities, which could result in increased scrutiny by regulatory authorities, delays in our clinical programs and regulatory approval, increases in our operating expenses, failure to obtain or maintain approval or limitations in our commercial supply. Our customer concentration may materially adversely affect
We depend on wholesale pharmaceutical distributors for retail distribution of our products; if we lose any of our significant wholesale pharmaceutical distributors or their distribution network is disrupted, our financial condition and results of operations.operations may be adversely affected. A significant percentage of our product shipments are to a limited number of independent wholesale drugpharmaceutical distributors. Three of our wholesale pharmaceutical distributors represented 39%, 29% and 23%greater than 90% of our product shipments for the year ended December 31, 2017. If we were to lose the business2023. Our loss of one or more of these distributors, or if any of these distributors failedwholesale pharmaceutical distributors’ accounts, or a material reduction in their purchases or a significant disruption to fulfill their obligationstransportation infrastructure or refused or experienced difficulty in paying us on a timely basis, or negotiated larger discounts, it wouldother means of distribution of our products, could have a material adverse effect on our business, results of operations, financial condition and prospects. The significance of each wholesale pharmaceutical distributor account to our business adversely impacts our ability to negotiate favorable commercial terms with each such distributor, and as a result, we may be forced to accept terms that adversely impact our results of operations. In addition, these wholesaler customers comprise a significant part of the distribution network for pharmaceutical products in the United States. This distribution network has undergone, and may continue to undergo, significant consolidation marked by mergers and acquisitions. As a result, a small number of large wholesale distributors control a significant share of the market. Consolidation of drug wholesalers has increased, and may continue to increase, competitive and pricing pressures on pharmaceutical products. We cannot guarantee that we can manage these pricing pressures or that wholesaler purchases will not fluctuate unexpectedly from period to period. Certain of our opioid products are subject to post-marketing requirements or commitments, which may, in some cases, not be capable of timely or satisfactory completion without participation in consortia over which we have limited control. For certain of our products, we are subject to post-marketing requirements to conduct epidemiological studies and clinical trials, or, in some cases, to conduct post-marketing surveillance or observational studies to gather additional information about our products. For our opioid products, we generally intend to fulfill our PMRs by virtue of our participation in the Opioid PMR Consortium (“OPC”). Although we retain discretion in how to discharge such PMRs, the scale and scope of the studies required by the FDA make it cost prohibitive to discharge these requirements other than by joining the OPC that was formed to conduct them. We are a member of the OPC and engage in decision-making as a member of that organization, but do not have a majority. If the OPC fails to conduct sufficiently rigorous studies or is unable to achieve the patient enrollment or other requirements established by the FDA, we may be unable to satisfy our PMRs and the FDA may choose to withdraw or otherwise restrict its approval of our opioid products. Additionally, there may be certain PMRs or post-marketing commitments that we fulfill on our own for our products, including via the conduct of post-marketing surveillance or observational studies. If such studies lead to the discovery of adverse findings regarding the safety or benefit profiles of our products, then the FDA may choose to withdraw or otherwise restrict the approval of our products or the FDA or we may determine that labeling changes are warranted based on their finding. Such withdrawal or restriction or labeling changes for our products would have an adverse impact on our business and financial condition. Risks Related to Our Business and Strategy We may not realize all the anticipated benefits from our future acquisitions, and we may be unable to successfully integrate future acquisitions. Our growth strategy will, in part, rely on acquisitions. We must plan and manage acquisitions effectively to achieve revenue growth and maintain profitability in our evolving market. We may not realize all the anticipated benefits from our future acquisitions, such as increased earnings, cost savings and revenue enhancements, for various reasons, including difficulties integrating operations and personnel, higher than expected acquisition and operating costs or other difficulties, inexperience with operating in new geographic regions, unknown liabilities, inaccurate reserve estimates and fluctuations in market prices. In addition, integrating acquired businesses and properties involves a number of special risks and unforeseen difficulties can arise in integrating operations and systems and in retaining and assimilating employees. These difficulties include, among other things: | ● | operating a larger organization; |
| ● | coordinating geographically disparate organizations, systems, and facilities; |
| ● | integrating corporate, technological, and administrative functions; |
| ● | diverting management’s attention from regular business concerns; |
| ● | diverting financial resources away from existing operations; |
| ● | increasing our indebtedness; and |
| ● | incurring potential environmental or regulatory liabilities and title problems. |
Any of these or other similar risks could lead to potential adverse short-term or long-term effects on our operating results. The process of integrating our operations could cause an interruption of, or loss of momentum in, the activities of our business. Members of our management may be required to devote considerable amounts of time to this integration process, which decreases the time they have to manage our business. If our management is not able to effectively manage the integration process, or if any business activities are interrupted as a result of the integration process, our business could suffer. Our business may be adversely affected by certain events or circumstances outside our control, including macroeconomic conditions and geopolitical turmoil. Events or circumstances outside of our control, including macroeconomic conditions such as recession or depression, inflation, and declines in consumer-spending could result in reduced demand for our products. An economic downturn could result in business closures, higher levels of unemployment, or declines in consumer disposable income which could have an impact on the number of patients seeking and receiving treatment for conditions that might otherwise result in the prescription of our products, as patients may make efforts to avoid or postpone seeking non-essential medical care to allocate their resources to other priorities or essential items. These circumstances, in addition to the impact of geopolitical turmoil, such as the ongoing Ukrainian War and current conflict in Israel and Gaza (including any escalation or expansion), social unrest, political instability in the United States and elsewhere, terrorism, cyberwarfare or other acts of war, may result in reduced demand for our products and negatively impact our sales, results of operations, and liquidity. Security breaches and other disruptions to our, or our vendors’, information technology systems may compromise our information and expose us to liability that could adversely impact our financial condition, operations, and reputation. We, our collaborators, third-party providers, distributors, customers and other contractors utilize information technology systems and networks (“Systems”) to transmit, store and otherwise process electronic data in connection with our business activities, including our supply chain processes, operations and communications including, in some cases, our business proprietary information, and Electronic Data Interchange (“EDI”) on purchase orders, invoices, chargebacks, among other things. Our Systems, along with those of the third parties whom we rely on to process confidential and sensitive data in a variety of contexts, are potentially vulnerable to a variety of evolving threats that may expose this data to unauthorized persons or otherwise compromise its integrity. These threats may include, but are not limited to, social-engineering attacks (including through phishing attacks), business email compromise, online and offline fraud, malicious code (such as viruses and worms), malware (including as a result of advanced persistent threat intrusions), denial-of-service attacks, access attacks (such as credential stuffing), personnel misconduct or error, ransomware attacks, supply-chain attacks, software bugs, server malfunctions, software or hardware failures, loss of data or other information technology assets, adware, telecommunications failures, earthquakes, fires, floods, and other similar threats. We may expend significant resources to try to protect against these threats to our Systems. Certain data privacy and security laws, as well as industry best practice standards, may require us to implement and maintain security measures. While we have implemented security measures designed to protect our Systems and confidential and sensitive data, there can be no assurance that these measures will be effective. Threat actors and their techniques change frequently, are often sophisticated in nature, and may not be detected until after a security incident has occurred. If we, or a third party upon whom we rely, experience a security incident or are perceived to have experienced a security incident, we may experience adverse consequences. These consequences may include: government enforcement actions (for example, investigations, fines, penalties, audits, and inspections); additional reporting requirements and/or oversight; restrictions on processing sensitive data (including personal data); litigation (including class claims); indemnification obligations; negative publicity; reputational harm; monetary fund diversions; interruptions in our operations (including availability of data); financial loss; and other similar harms. Further, our insurance coverage may not be adequate or sufficient in type or amount to protect us from or to mitigate liabilities arising out of our privacy and security practices. Litigation or regulatory action regarding opioid medications could negatively affect our business. Beginning in 2018, lawsuits alleging damages related to opioids have been filed naming us as a defendant along with other manufacturers of prescription opioid medications. These lawsuits, filed in multiple jurisdictions, are brought by various local governments as well as private claimants, against various manufacturers, distributors and retail pharmacies. These lawsuits generally allege that we had engaged in improper marketing practices related to Xtampza ER and the Nucynta Products. In March 2022, we entered into a Master Settlement Agreement resolving 27 pending opioid-related lawsuits brought against us by cities, counties, and other subdivisions in the United States. As part of the Master Settlement Agreement, we paid $2.75 million to the plaintiffs and the cases were dismissed, with prejudice. In late March 2023, three new cases were filed in three federal courts, naming us as one of numerous defendants, from which we have been dismissed. Certain governmental and regulatory agencies are focused on the abuse of opioid medications, a concern we share, and we have received Civil Investigative Demands or subpoenas from four state attorneys general investigating our sales and marketing of opioids and seeking documents relating to the manufacture, marketing and sale of opioid medications. In December 2021, we entered into an Assurance of Discontinuance with the Massachusetts Attorney General pursuant to which we provided certain assurances and agreed to pay certain of the Massachusetts Attorney General’s costs of investigation, in exchange for closure of the investigation and a release of claims pertaining to the subject matter of the investigation. We are cooperating fully in the open investigations. Managing litigation and responding to governmental investigations is costly and may involve a significant diversion of management attention. Such proceedings are unpredictable and may develop over lengthy periods of time. An adverse resolution of any of these lawsuits or investigations may involve injunctive relief or substantial monetary penalties, either or both of which could have a material adverse effect on our reputation, business, results of operations and cash flows. Risks Related to Our Business and Strategy
We face substantial competition from other biotechnology and pharmaceutical companies, which may result in others discovering, developing or commercializing products before or more successfully than we do. The biopharmaceutical industry is intensely competitive and subject to rapid and significant technological change. In addition, the competition
Competition in the pain and opioid market is intense. We haveOur competitors both in the United States and internationally, includinginclude major multinational pharmaceutical companies, biotechnology companies and universities and other research institutions. We face and will continue to face competition from other companies in the pharmaceutical and medical device industries. Xtampza, the Nucynta Products, and our product candidates, if approved, will Our products compete with currently marketed oral opioids, transdermal opioids, local anesthetic patches, stimulants and implantable and external infusion pumps that can be used for infusion of opioids and local anesthetics. Products of these types are marketed by Actavis, Depomed, Egalet, Endo, Mallinckrodt, Pernix, Pfizer, Purdue, Teva, and others. Some of these current and potential future competitors may be addressing the same therapeutic areas or indications as we are. Many of our current and potential future competitors have significantly greater research and development capabilities than we do, have substantially more marketing, manufacturing, financial, technical, human and managerial resources than we do, and have more institutional experience than we do. Mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated in our competitors.
As a result of these factors, our competitors may obtain regulatory approval of their products more rapidly than we are able to or may obtain patent protection or other intellectual property rights that allow them to develop and commercialize
their products before us and limit our ability to develop or commercialize our product and product candidates. Our competitors may also develop drugs that are safer, more effective, more widely used and less costly than ours, and they may also be more successful than us in manufacturing and marketing their products.
Furthermore, if the FDA approves a competitor’s 505(b)(2) application for a drug candidate before our application for a similar drug candidate and grants the competitor a period of exclusivity, the FDA may take the position that it cannot approve our NDA for a similar drug candidate. For example, several competitors have developed extended-release hydrocodone products, and if the FDA grants exclusivity, we could be subject to a delay that would dramatically reduce the expected market penetration for our hydrocodone product candidate. Additionally, even if our 505(b)(2) application is approved for marketing, we may still be subject to competition from other hydrocodone products, including approved products or other approved 505(b)(2) NDAs for different conditions of use that would not be restricted by any grant of exclusivity to us.
In addition, competitors have developed or are in the process of developingmay develop technologies that are, or in the future may be, the basis for competitive products. Some of these products may have an entirely different approach or means of accomplishing similar therapeutic effects than our product candidates. Our competitors may develop products that are safer, more effective or less costly than our product candidates and, therefore, present a serious competitive threat to our product offerings.
The widespread acceptance of currently available therapies with which our product and product candidates, if approved, compete may limit market acceptance of our product and product candidates even if commercialized. Oralproducts. Moreover, oral medications, transdermal drug delivery systems, such as drug patches, injectable products and implantable drug delivery devices are currently available treatments for chronic pain, are widely accepted in the medical community and have a long history of use. These treatments will compete with our product and product candidates, if approved,products and the established use of these competitive products may limit the potential for our product and product candidatesproducts to receive widespread acceptance if commercialized.acceptance.
The use of legal and regulatory strategies by competitors with innovator products, including the filing of citizen petitions, may delay or prevent the introduction or approval of our product candidates, increase our costs associated with the introduction or marketing
Commercial sales of our products or significantly reduce the profit potential of ourand any products we acquire, may expose us to expensive product candidates. Companies with innovator drugs often pursue strategies that may serve to prevent or delay competition from alternatives to their innovator products. These strategies include, but are not limited to:
| ·
| | filing “citizen petitions” with the FDA that may delay competition by causing delays of our product approvals;
|
| ·
| | seeking to establish regulatoryliability claims, and legal obstacles that would make it more difficult to demonstrate a product’s bioequivalence or “sameness” to the related innovator product;
|
| ·
| | filing suits for patent infringement that automatically delay FDA approval of products seeking approval based on the Section 505(b)(2) pathway;
|
| ·
| | obtaining extensions of market exclusivity by conducting clinical trials of innovator drugs in pediatric populations or by other methods;
|
| ·
| | persuading the FDA to withdraw the approval of innovator drugs for which the patents are about to expire, thus allowing the innovator company to develop and launch new patented products serving as substitutes for the withdrawn products;
|
| ·
| | seeking to obtain new patents on drugs for which patent protection is about to expire; and
|
| ·
| | initiating legislative and administrative efforts in various states to limit the substitution of innovator products by pharmacies.
|
These strategies could delay, reduce or eliminate our entry into the market and our ability to generate revenues from our product and product candidates.
Our future success depends on our ability to retain our key personnel.
We are highly dependent upon the services of our key personnel, including our President and Chief Executive Officer, Michael T. Heffernan, our Chief Financial Officer, Paul Brannelly, our Chief Operating Officer, Joseph Ciaffoni and our Chief Technology Officer, Alison Fleming, PhD. Each employee is employed by us at will and is permitted to terminate his or her employment with us at any time pursuant to the terms of his employment agreement. We do not maintain “key person” insurance for any of our executives or other employees. The loss of the services of Mr. Heffernan, Mr. Brannelly, Mr. Ciaffoni or Dr. Fleming could impede the achievement of our development and commercialization objectives.
If we are unable to attract and retain highly qualified scientific and technical employees, we may not be able to grow effectively.maintain product liability insurance on reasonable terms or at all.
Our future growth and success depend on our ability to recruit, retain, manage and motivate our scientific, clinical, manufacturing and commercial employees. The loss of any member of our senior management team or the inability to hire or retain experienced management personnel could compromise our ability to execute our business plan and harm our operating results. Because of the specialized scientific nature of our business, we rely heavily on our ability to attract and retain qualified personnel. The competition for qualified personnel in the pharmaceutical field is intense, and as a result, we
We currently carry product liability insurance. Product liability claims may be unable to continue to attract and retain qualified personnel necessary for the development ofbrought against us by patients; healthcare providers; or others using, administering or selling our business or to recruit suitable replacement personnel. We will need to grow the size ofproducts. If we cannot successfully defend ourselves against claims that our organization, andproducts caused injuries, we may experience difficulties in managing this growth.
We have experienced a period of rapid growth. Our management, personnel and systems may not be adequate to support this and future growth.could incur substantial liabilities. We may not be able to effectively manage the expansionmaintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. Regardless of our operations, whichmerit or eventual outcome, liability claims may result in weaknesses in our infrastructure, give rise to operational mistakes, loss of business opportunities, loss of employees and reduced productivity among remaining employees. Future growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of our existing or future product candidates. Future growth would impose significant added responsibilities on members of management, including:
| ·
| | managing the commercialization of any FDA-approved products;
|
| ·
| | overseeing clinical trials effectively;
|
| ·
| | identifying, recruiting, maintaining, motivating and integrating additional employees, including any sales and marketing personnel engaged in connection with the commercialization of any approved product;
|
| ·
| | managing our internal development efforts effectively while complying with our contractual obligations to licensors, licensees, contractors and other third parties;
|
| ·
| | improving our managerial, development, operational and financial systems and procedures; and
|
| ·
| | developing our compliance infrastructure and processes to ensure compliance with regulations applicable to public companies.
|
As our operations expand, we will need to manage additional relationships with various strategic collaborators, suppliers and other third parties. Our future financial performance and our ability to commercialize our product and product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To that end, we must be able to manage our development efforts and clinical trials effectively and hire, train and integrate additional management, administrative and sales and marketing personnel. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing our company.
We may acquire other assets or businesses, or form collaborations or make investments in other companies or technologies, that could have a material adverse effect on our operating results, dilute our shareholders’ ownership, increase our debt or cause us to incur significant expense.costs to defend the litigation.
As part of our business strategy, we may pursue acquisitions of assets, including preclinical, clinical or commercial stage products or product candidates, or businesses, in-licensing or out-licensing of products, product candidates or technologies, or other strategic alliances and collaborations, to expand our existing technologies and operations. We may not identify or complete these transactions in a timely manner, on a cost-effective basis, or at all, and we may not realize the anticipated benefits of any such transaction, any of which could have a material adverse effect on our financial condition, results of operations and cash flows. We have limited experience with acquiring other companies, products or product candidates, and limited experience with licensing and forming strategic alliances and collaborations. We may not find suitable acquisition candidates, and if we make an acquisition, we may not integrate the acquisition successfully into our existing business and we may incur additional debt or assume unknown or contingent liabilities in connection therewith. Integration of an acquired company or assets may also disrupt ongoing operations, require the hiring of additional personnel and the implementation of additional internal systems and infrastructure, especially the acquisition of commercial assets, and require management resources that would otherwise focus on developing our existing business. We may not be able to find suitable strategic alliance or collaborators or identify other investment opportunities, and we may experience losses related to any such investments.
To finance any acquisitions, licenses or collaborations, we may incur significant transaction expenses and we may choose to issue debt or shares of our common or preferred stock as consideration. Any such issuance of shares would dilute the ownership of our shareholders. If the price of our common stock is low or volatile, we may not be able to acquire, license, or otherwise obtain rights to other assets or companies or fund a transaction using our stock as consideration. Alternatively, it may be necessary for us to raise additional funds for acquisitions through public or private financings. Additional funds may not be available on terms that are favorable to us, or at all.
Our employees, independent contractors, principal investigators, CROs, consultants and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could cause significant liability for us and harm our reputation.
We are exposed to the risk that our employees, independent contractors, principal investigators, CROs, consultants and vendors may engage in fraudulent conduct or other illegal activity. Misconduct by these parties could include intentional, reckless and/or negligent conduct or disclosure of unauthorized activities to us that violates:
| ·
| | FDA, DEA or similar regulations of foreign regulatory authorities, including those laws requiring the reporting of true, complete and accurate information to such authorities;
|
| ·
| | manufacturing standards;
|
| ·
| | federal and state healthcare fraud and abuse laws and regulations and similar laws and regulations established and enforced by foreign regulatory authorities; or
|
| ·
| | laws that require the reporting of financial information or data accurately.
|
In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Activities subject to these laws also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. We have adopted a Code of Ethics, but it is not always possible to identify and deter misconduct by employees and other third parties, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a material adverse effect on our business and results of operations, including the imposition of civil, criminal and administrative penalties,
damages, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could have a material adverse effect on our ability to operate our business and our results of operations.
Our relationships with customers and payors are subject to applicable anti-kickback, fraud and abuse, transparency, and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, exclusion from government healthcare programs, contractual damages, reputational harm, administrative burdens, and diminished profits and future earnings. Healthcare providers, physicians and payors play a primary role in the recommendation and prescription of Xtampza, the Nucynta Products, and any product candidates for which we may obtain marketing approval.our products. Our future arrangements with payors and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we market, sell and distribute Xtampza, the Nucynta Products, and any product candidates for which we may obtain marketing approval.our products. Even though we do not and will not control referrals of healthcare services or bill directly to Medicare, Medicaid or other third-party payors federaldirectly, we may provide reimbursement guidance and support regarding our products to our customers and patients. Federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business. Restrictions under applicable federal, state and foreign healthcare laws and regulations may affect our ability to operate and expose us to areas of risk, including: | ·
| | the federal Anti-Kickback Statute, which prohibits, among other things, knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under federal and state healthcare programs such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
|
| ·
| | the federal False Claims Act, which imposes criminal and civil penalties, including through civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government. In addition, the government may assert that a claim including items and services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act;
|
| ·
| | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which imposes criminal and civil liability for executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute to defraud any healthcare benefit program or specific intent to violate it in order to have committed a violation;
|
| ·
| | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and its implementing regulations, which also imposes obligations on certain covered entity healthcare providers, health plans, and healthcare clearinghouses as well as their business associates that perform certain services involving the use or disclosure of individually identifiable health information, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information;
|
| ·
| | federal laws requiring drug manufacturers to report annually information related to certain payments and other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as ownership or investment interests held by physicians and their immediate family members, including under the federal Open Payments program, commonly known as the Sunshine Act, as well as other state and foreign laws regulating marketing activities and requiring manufacturers to report marketing expenditures, payments and other transfers of value to physicians and other healthcare providers;
|
| ·
| | federal government price reporting laws, which require us to calculate and report complex pricing metrics to government programs, where such reported prices may be used in the calculation of reimbursement and/or discounts on our marketed drugs. Participation in these programs and compliance with the applicable requirements may subject us to potentially significant discounts on our products, increased infrastructure costs, potential liability for the failure to report such prices in an accurate and timely manner, and potentially limit our ability to offer certain marketplace discounts; and
|
| ·
| | state and foreign equivalents of each of the above laws, including state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental payors, including private insurers; state laws which require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government or otherwise restricting payments that may be made to healthcare providers; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
|
While we do not submit claims and our customers will make the ultimate decision on how to submit claims, we may provide reimbursement guidance and support regarding our products to our customers and patients. If a government authority were to conclude that we provided improper advice to our customers and/or encouraged the submission of false claims for reimbursement, we could face action by government authorities. Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. Nonetheless, it is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion from participation in government funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations.
If
We or the third parties upon whom we fail to comply with environmental,depend may be adversely affected by natural disasters and/or health and safety laws and regulations, we could become subject to fines or penalties or incur significant costs. In connection with our research and development activitiesepidemics, and our manufacturebusiness continuity and disaster recovery plans may not adequately protect us from a serious disaster.
Natural disasters could severely disrupt our operations, and have a material adverse effect on our business, results of materialsoperations, financial condition and productsprospects. If a natural disaster, power outage, health epidemic or other event occurred that prevented us from using all or a significant portion of our facilities, that damaged critical infrastructure, such as the manufacturing facilities of our third-party contract manufacturers, or that otherwise disrupted operations, it might become difficult or, in certain cases, impossible for us to continue our business, and product candidates,any disruption could last for a substantial period of time. The disaster recovery and business continuity plans we are subject to federal, statehave in place, and local laws, rules, regulations and policies governing the use, generation, manufacture, storage, air emission, effluent discharge, handling and disposal of certain materials, biological specimens and wastes. Although we believetechnology that we have complied with the applicable laws, regulations and policies in all material respects and have not been requiredmay rely upon to correct any material noncompliance, weimplement such plans, may be required to incur significant costs to comply with environmental and health and safety regulations in the future. Current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions. Our research and development involves the use, generation and disposal of hazardous materials, including chemicals, solvents, agents and biohazardous materials. Although we believe that our safety procedures for storing, handling and disposing of such materials comply with the standards prescribed by state and federal regulations, we cannot completely eliminate the risk of accidental contamination or injury from these materials. We currently contract with third parties to dispose of these substances that we generate, and we rely on these third parties to properly dispose of these substances in compliance with applicable laws and regulations. We cannot eliminate the risk of contamination or injury from these materials. If these third parties do not properly dispose of these substances in compliance with applicable laws and regulations, we may be subject to legal action by governmental agencies or private parties for improper disposal of these substances. The costs of defending such actions and the potential liability resulting from such actions are often very large. In the event we are subject to such legal action or we otherwise fail to comply with applicable laws and regulations governing the use, generation and disposal of hazardous materials and chemicals, we could be held liable for any damages that result, and any such liability could exceed our resources.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees, this insurance may not provide adequate coverage against potential liabilities. We maintain insurance for environmental liability or toxic tort claims, but we may not continue to maintain such insurance in the future, and
such insurance, to the extent maintained, may not be adequate to cover liabilities that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
Our business and operations would sufferprove inadequate in the event of computer system failures, accidentsa serious disaster or security breaches.
Despitesimilar event. We may incur substantial expenses as a result of the implementation of security measures, our internal computer systems, and thoselimited nature of our CROs, contract manufacturing organization, or CMO,disaster recovery and other third parties onbusiness continuity plans, which we rely, are vulnerable to damage from computer viruses, unauthorized access, cyber attacks and other malfeasance, natural disasters, terrorism, war and telecommunication and electrical failures. System failures, accidents or security breaches could cause interruptions in our operations, and could result inhave a material disruptionadverse effect on our business, financial condition and results of our commercial and clinical activities and business operations, in addition to possibly requiring substantial expenditures of resources to remedy. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our commercialization and drug development programs. For example, the loss of clinical trial data from completed or ongoing clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach was to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further commercialization of our products and development of our product candidates could be delayed.operation.
Risks Related to Our Common Stock The price of our common stock may be volatile and you may lose all or part of your investment. The market price of our common stock is highly volatile and may be subject to wide fluctuations in response to numerous factors described in these “Risk Factors,” some of which are beyond our control. In addition to the factors discussed in these Risk Factors, these factors include: | ·
| | the success of competitive products or technologies;
|
| ·
| | regulatory actions with respect to our product and product candidates or our competitors’ products or product candidates;
|
| ·
| | actual or anticipated changes in our growth rate relative to our competitors;
|
| ·
| | the outcome of any patent infringement or other litigation that may be brought against us, including the ongoing Purdue litigation;
|
| ·
| | announcements by us or our competitors of significant acquisitions, strategic collaborations, joint ventures, collaborations or capital commitments;
|
| ·
| | results of clinical trials of our product and product candidates or those of our competitors;
|
| ·
| | regulatory or legal developments in the United States and other countries;
|
| ·
| | developments or disputes concerning patent applications, issued patents or other proprietary rights;
|
| ·
| | the recruitment or departure of key personnel;
|
| ·
| | the level of expenses related to our product and product candidates or clinical development programs;
|
| ·
| | actual or anticipated variations in our quarterly operating results;
|
| ·
| | the number and characteristics of our efforts to in-license or acquire additional product candidates or products;
|
| ·
| | introduction of new products or services by us or our competitors;
|
| ·
| | failure to meet the estimates and projections of the investment community or that we may otherwise provide to the public;
|
| ·
| | actual or anticipated changes in estimates as to financial results, development timelines or recommendations by securities analysts;
|
| ·
| | variations in our financial results or those of companies that are perceived to be similar to us;
|
| ·
| | fluctuations in the valuation of companies perceived by investors to be comparable to us;
|
| ·
| | share price and volume fluctuations attributable to inconsistent trading volume levels of our shares;
|
| ·
| | announcement or expectation of additional financing efforts;
|
| ·
| | sales of our common stock by us, our insiders or our other shareholders;
|
| ·
| | changes in accounting practices;
|
| ·
| | significant lawsuits, including patent or shareholder litigation;
|
| ·
| | changes in the structure of healthcare payment systems;
|
| ·
| | market conditions in the pharmaceutical and biotechnology sectors;
|
| ·
| | general economic, industry and market conditions;
|
| ·
| | publication of research reports about us, our competitors or our industry, or positive or negative recommendations or withdrawal of research coverage by securities or industry analysts; and
|
| ·
| | other events or factors, many of which are beyond our control.
|
In addition, theThe stock market in general, and pharmaceutical and biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies.fluctuations. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our business model, prospects or actual operating performance. The realization of any of the abovethese risks, or any of a broad range of other risks stated abovediscussed in this report, could have a material adverse effect on the market price of our common stock.
As we operate in the pharmaceutical and biotechnology industry, we are especially vulnerable to these factors to the extent that they affect our industry or our products. In the past, securities class action litigation has often been initiated against companies following periods of volatility in their stock price. This type of litigation could result in substantial costs and divert our management’s attention and resources, and could also require us to make substantial payments to satisfy judgments or to settle litigation.
Sales of a substantial number of shares of our common stock in the public market could cause our stock price to fall.
Sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock. As of December 31, 2017, holders of an aggregate of approximately 4.5 million shares of our common stock have rights, subject to certain conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other shareholders. Once we register these shares, they can be freely sold in the public market, subject to volume limitations applicable to affiliates.
Actual or potential sales of our common stock by our directors or employees, including our executive officers, pursuant to pre-arranged stock trading plans or otherwise could cause our stock price to fall or prevent it from increasing for numerous reasons, and actual or potential sales by such persons could be viewed negatively by investors.
In accordance with the guidelines specified under Rule 10b5-1 of the Exchange Act and our policies regarding stock transactions, our directors and employees, including our executive officers, could adopt stock trading plans pursuant to which they may sell shares of our common stock from time to time in the future. Generally, sales under such plans by our executive officers and directors require public filings. Actual or potential sales of our common stock by such persons could cause our common stock to fall or prevent it from increasing for numerous reasons. For example, a substantial number of shares of our common stock becoming available (or being perceived to become available) for sale in the public market could cause the market price of our common stock to fall or prevent it from increasing. Also, actual or
potential sales by such persons could be viewed negatively by investors.
Future issuances of our common stock or rights to purchase common stock, including pursuant to our equity incentive plans, could result in additional dilution of the percentage ownership of our shareholders and could cause our stock price to fall.
We expect that significant additional capital will be needed in the future to continue our planned operations. To raise capital, we may sell substantial amounts of common stock or securities convertible into or exchangeable for common stock. These future issuances of common stock or common stock-related securities, together with the exercise of outstanding options and any additional shares issued in connection with acquisitions, if any, may result in material dilution to our investors. Such sales may also result in material dilution to our existing shareholders, and new investors could gain rights, preferences and privileges senior to those of holders of our common stock.
Our principal shareholders and management own a majority of our stock and have the ability to exert significant control over matters subject to shareholder approval.
As of December 31, 2017, our executive officers, directors, holders of 5% or more of our capital stock and their respective affiliates beneficially owned a majority of our voting stock, including shares subject to outstanding options and warrants. As a result, if these shareholders were to choose to act together, they would be able to significantly influence the outcome of all matters requiring shareholder approval, including the election of directors, amendments of our organizational documents, or approval of any merger, sale of assets or other major corporate transaction. This may prevent or discourage unsolicited acquisition proposals or offers for our common stock that you may feel are in your best interest. The interests of this group of shareholders may not always coincide with your interests or the interests of other shareholders and they may act in a manner that advances their best interests and not necessarily those of other shareholders, including seeking a premium value for their common stock, and might affect the prevailing market price for our common stock. Such concentration of ownership control may:
| ·
| | delay, defer or prevent a change in control;
|
| ·
| | entrench our management and/or the board of directors; or
|
| ·
| | impede a merger, consolidation, takeover or other business combination involving us that other shareholders may desire.
|
In addition, persons associated with Longitude Capital Partners, LLC and Skyline Venture Partners V, L.P. currently serve on our board of directors. The interests of Longitude Capital Partners, LLC and Skyline Venture Partners V, L.P. may not always coincide with the interests of the other shareholders, and the concentration of control in Longitude Capital Partners, LLC and Skyline Venture Partners V, L.P. limits other shareholders’ ability to influence corporate matters. We may also take actions that our other shareholders do not view as beneficial, which may adversely affect our results of operations and financial condition and cause a decline in our stock price.
We are subject to anti-takeover provisions in our second amended and restated articles of incorporation and amended and restated bylaws and under Virginia law that could delay or prevent an acquisition of our company, even if the acquisition would be beneficial to our shareholders. Certain provisions of Virginia law, the state in which we are incorporated, and our second amended and restated articles of incorporation and amended and restated bylaws could hamper a third party’s acquisition of us, or discourage a third party from attempting to acquire control of us. These provisions include: | ·
| | a provision allowing our board of directors to set the terms of and issue preferred stock with rights senior to those of the common stock without any vote or action by the holders of our common stock. The issuance of preferred stock could adversely affect the rights and powers, including voting rights, of the holders of common stock;
|
| ·
| | advance written notice procedures and notice requirements with respect to shareholder proposals and shareholder nomination of candidates for election as directors;
|
| ·
| | a provision that only the board of directors, the chairman of the board of directors or the president may call a special meeting of the shareholders;
|
| ·
| | the application of Virginia law prohibiting us from entering into certain transactions with the beneficial owner of more than 10 percent of our outstanding voting stock for a period of three years after such person first reached that level of stock ownership, unless certain conditions are met;
|
| ·
| | a provision dividing our board of directors into three classes, each serving three-year terms;
|
| ·
| | the requirement that the authorized number of our directors be changed only by resolution of our board of directors;
|
| ·
| | a provision that our board of directors shall fill any vacancies on our board of directors, including vacancies resulting from a board of directors’ resolution to increase the number of directors;
|
| ·
| | limitations on the manner in which shareholders can remove directors from the board of directors;
|
| ·
| | the lack of cumulative voting in the election of directors; and
|
| ·
| | the prohibition on shareholders acting by less-than-unanimous written consent.
|
These provisions also could limit the price that certain investors might be willing to pay in the future for shares of our common stock. In addition, these provisions make it more difficult for our shareholders to remove our boardBoard of directorsDirectors or management or elect new directors to our boardBoard of directors.Directors.
We may fail to qualify for continued listing on The NASDAQ Global Select Market which could make it more difficult for investors to sell their shares.
Our common stock is listed on The NASDAQ Global Select Market (NASDAQ). As a NASDAQ listed company, we are required to satisfy the continued listing requirements of NASDAQ for inclusion in the Global Select Market to maintain such listing, including, among other things, the maintenance of a minimum closing bid price of $1.00 per share and shareholders’ equity of at least $10.0 million. There can be no assurance that we will be able to maintain compliance with the continued listing requirements or that our common stock will not be delisted from NASDAQ in the future. If our common stock is delisted by NASDAQ, we could face significant material adverse consequences, including:
| ·
| | a limited availability of market quotations for our securities;
|
| ·
| | reduced liquidity with respect to our securities;
|
| ·
| | a determination that our shares are a “penny stock,” which will require brokers trading in our shares to adhere to more stringent rules, possibly resulting in a reduced level of trading activity in the secondary trading market for our shares;
|
| ·
| | a limited amount of news and analyst coverage for our company; and
|
| ·
| | a decreased ability to issue additional securities or obtain additional financing in the future.
|
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock depends in part on the research and reports that securities or industry analysts publish about us or our business. If one or more of the analysts who cover us downgrade our stock or publish inaccurate
or unfavorable research about our business, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our stock could decrease, which might cause our stock price and trading volume to decline.
We are an “emerging growth company” and we intend to take advantage of reduced disclosure and governance requirements applicable to emerging growth companies, which could result in our common stock being less attractive to investors and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies will make our shares of common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act, and may remain an emerging growth company for up to five years. For so long as we remain an emerging growth company, we are permitted and intend to rely on certain exemptions from various reporting requirements applicable to other public companies, but not to emerging growth companies, including, but not limited to, an exemption from the auditor attestation requirement of Section 404 of the Sarbanes-Oxley Act, reduced disclosure about executive compensation arrangements pursuant to the rules applicable to smaller reporting companies and no requirement to seek non-binding advisory votes on executive compensation or golden parachute arrangements. We will remain an emerging growth company until the earliest of (i) December 31, 2020, (ii) the first fiscal year after our annual gross revenue are $1.07 billion or more, (iii) the date on which we have, during the previous three-year period, issued more than $1.0 billion in non-convertible debt securities or (iv) the end of any fiscal year in which the market value of our common stock held by non-affiliates exceeded $700 million as of the end of the second quarter of that fiscal year.
In addition, Section 107 of the JOBS Act also provides that an emerging growth company can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. An emerging growth company can therefore delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. However, we are choosing to “opt out” of such extended transition period and, as a result, we will comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for non-emerging growth companies. Section 107 of the JOBS Act provides that our decision to opt out of the extended transition period for complying with new or revised accounting standards is irrevocable.
We cannot predict if investors will find our common stock less attractive as a result of our taking advantage of these exemptions. If some investors find our common stock less attractive as a result of our choices, there may be a less active trading market for our common stock and our stock price may be more volatile.
If investors find our common stock less attractive as a result of our reduced reporting requirements, there may be a less active trading market for our common stock and our stock price may be more volatile. We may also be unable to raise additional capital as and when we need it.
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial condition, results of operations or cash flows accurately, which may adversely affect investor confidence in us and, as a result, the value of our common stock. The Sarbanes-Oxley Act requires, among other things, that we maintain effective internal control over financial reporting. Commencing with our annual report on Form 10-K for the year ended December 31, 2016, weWe are required, under Section 404 of the Sarbanes-Oxley Act, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting. This assessment must include disclosure of any material weaknesses identified by our management in our internal control over financial reporting. A material weakness is a control deficiency, or combination of control deficiencies, in internal control over financial reporting that results in more than a reasonable possibility that a material misstatement of annual or interim financial statements will not be prevented or detected on a timely basis. Section 404 of the Sarbanes-Oxley Act also generally requires an attestation from our independent registered public accounting firm on the effectiveness of our internal control over financial reporting. However, for as long as we remain an emerging growth company as defined in the JOBS Act, we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not limited to, not being required to comply with the independent registered public accounting firm attestation requirement. Our compliance with Section 404 will require that we incur substantial accounting expense and expend significant management efforts. We currently do not have an internal audit group, and we will need to hire additional accounting and financial staff with appropriate public company experience and technical accounting knowledge, and compile the system and process documentation necessary to perform the evaluation needed to comply with Section 404. We may not be able to complete our evaluation, testing and any required remediation in a timely fashion, which could potentially subject us to sanctions or investigations by the SEC or other regulatory authorities. During the evaluation and testing process, ifIf we identify one or more material weaknesses in our internal control over financial reporting, we will be unable to assert that our internal control over financial reporting is effective. We cannot assure you that there will not be material weaknesses or significant deficiencies in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to accurately report our financial condition, results of operations or cash flows. If we are unable to conclude that our internal control over financial reporting is effective, or if our independent registered public accounting firm determines we have a material weakness or significant deficiency in our internal control over financial reporting once that firm begin its reviews,Further, we could lose investor confidence in the accuracy and completeness of our financial reports, the market price of our common stock could decline, and we could be subject to sanctions or investigations by NASDAQ, the SEC or other regulatory authorities. Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
Our disclosure controls and procedures may not prevent or detect all errors or acts
Sales of fraud. We are subject toour common stock in the periodic reporting requirements of the Exchange Act. Our disclosure controls and procedures are designed to reasonably assure that information required to be disclosedpublic market, either by us in reports we file or submit underby our current shareholders, or the Exchange Act is accumulated and communicated to management, recorded, processed, summarized and reported within the time periods specifiedperception that these sales could occur, could cause a decline in the rules and formsmarket price of the SEC. We believe that any disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.
These inherent limitations reflect the reality that judgments can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements due to error or fraud may occur and not be detected.
Thesecurities. Moreover, the exercise of options and warrants and other issuances of shares of common stock or securities convertible into or exercisable for shares of common stock will dilute your ownership interests and may adversely affect the future market price of our common stock.
Sales of our common stock in the public market, either by us or by our current shareholders, or the perception that these sales could occur, could cause a decline in the market price of our securities. All of the shares of our common stock held by those of our current shareholders may be immediately eligible for resale in the open market either in compliance with an exemption under Rule 144 promulgated under the Securities Act, or pursuant to an effective resale registration statement that we have previously filed with the SEC. Such sales, along with any other market transactions, could adversely affect the market price of our common stock. In addition, as As of December 31, 2017,2023, there were (a) outstanding options to purchase an
aggregate of 3,037,6901,176,750 shares of our common stock at a weighted average exercise price of $13.00$19.48 per share, of which options to purchase 1,025,2521,158,209 shares of our common stock were then exercisable, and (b) 2,445 shares of common stock issuable upon the exercise of warrants to purchase common stock at a weighted-average exercise price of $12.27 per share.exercisable. The exercise of options and warrants at prices below the market price of our common stock could adversely affect the price of shares of our common stock. Additional dilution may result from the issuance of shares of our common stock in connection with collaborations or manufacturing arrangements or in connection with other financing efforts. Any issuance
There can be no assurance that we will repurchase additional shares of our common stock that is not made solelyat all or at favorable prices. In August 2021, our Board of Directors authorized a repurchase program for the repurchase of up to then-existing shareholders proportionate to their interests, such as in the case$100 million of a stock dividend or stock split, will result in dilution to each shareholder by reducing his, her or its percentage ownershipshares of the total outstanding shares. Moreover, if we issue options or warrants to purchase our common stock in the future and those optionsat any time or warrants are exercised you may experience further dilution. Holderstimes through December 31, 2022 (the “Prior Repurchase Program”). We repurchased $61.9 million of shares of Directors authorized a share repurchase program for the repurchase of up to $100.0 million of shares of our common stock have no preemptive rights that entitle themthrough December 31, 2023 (the “2023 Repurchase Program”). We repurchased $75.0 million of shares pursuant to purchase their pro ratathe 2023 Repurchase Program prior to its expiration on December 31, 2023. In January 2024, our Board of Directors authorized a new share repurchase program for the repurchase of any offeringup to $150.0 million of shares of any classour common stock through June 30, 2025 (the “2024-2025 Repurchase Program”). The 2024-2025 Repurchase Program permits us to effect repurchases through a variety of methods, including open-market purchases (including pursuant to a trading plan adopted in accordance with Rule 10b5-1 of the Exchange Act), privately negotiated transactions, or series. We have broad discretionotherwise in compliance with Rule 10b-18 of the use ofExchange Act. Share repurchases under the 2024-2025 Repurchase Program will depend upon, among other factors, our cash balances and cash equivalents,potential future capital requirements, our results of operations and despite our efforts, we may use them in a manner that does not increase the value of your investment.
We have broad discretion in the use of our cash and cash equivalents, and investors must rely on the judgment of our management regarding the use of our cash and cash equivalents. Our management may not use cash and cash equivalents in ways that ultimately increase the value of our common stock. Our failure to use our cash and cash equivalents effectively could result in financial losses that could have a material adverse effect on our business, causecondition, the price of our common stock to declineon the NASDAQ Global Select Market, and delay the commercialization or development of our product and product candidates. We may invest our cash and cash equivalents in short-term or long-term, investment-grade, interest-bearing securities. These investments may not yield favorable returns. If we do not invest or apply our cash and cash equivalents in waysother factors that enhance shareholder value, we may faildeem relevant. We can provide no assurance that we will continue to achieve expected financial results, which could cause the pricerepurchase shares of our common stock to decline.at favorable prices, if at all.
Item 1B. Unresolved Staff Comments Because we do not anticipate paying any cash dividends on our capital stock in the foreseeable future, capital appreciation, if any, will be your sole source of gain.
Not applicable. Item 1C. Cybersecurity Risk Management and Strategy We have never declared or paid cash dividends onmaintain a cybersecurity program designed to assess, identify, and mitigate risks from cybersecurity threats. This program is informed by the five elements of the National Institute of Standards and Technology framework: identify, protect, detect, respond, and recover. We utilize various methods to achieve these objectives including but not limited to company-wide policies and operating procedures, periodic testing, systems monitoring, patch management, and mandatory ongoing employee trainings. Additionally, we partner with third-party experts to conduct periodic penetration tests and to evaluate our common stock.information technology infrastructure for vulnerabilities. We currently intendalso evaluate cybersecurity risks associated with third-party vendors that provide the hosted applications we use in our financial close process through review of their System and Organization Controls (“SOC”) 1 reports at least annually. We continue to retain allinvest in our information technology infrastructure and cybersecurity program to strengthen our ability to protect the confidentiality, integrity, and availability of our future earnings, if any,data and the security of our information systems. In addition to financeour cybersecurity program, we assess cybersecurity risks as part of our overall risk management processes, primarily through our annual Enterprise Risk Assessment. Our Enterprise Risk Assessment surveys various employees and leaders throughout our organization with the growth and developmentgoal of evaluating our risk landscape, enhancing our overall understanding of risks to our business, and doultimately managing and/or mitigating identified risks. We assess various risks, including cybersecurity related risks, based on the likelihood of an incident occurring, impact to our organization if an incident occurred, and the level of internal control we currently have over the risk. The results are analyzed to identify vulnerabilities and then risk management/mitigation plans are designed, implemented, and evaluated for effectiveness. If a cybersecurity incident were to occur, we would implement our incident response plan in an effort to contain and mitigate the threat. As part of our incident response plan, our Cybersecurity Incident Response Team (a cross-functional taskforce comprised of senior representatives), would convene to assess the potential impact to our business, including financial reporting requirements and legal implications. We, like other companies in our industry, face a number of cybersecurity risks in connection with our business. Although such risks have not anticipate declaringmaterially affected us, including our business strategy, results of operations or paying any cash dividendsfinancial condition, to date, we and/or our vendors have, from time to time, experienced threats to, or security incidents, related to our data and systems or that had the potential to otherwise impact our business. For more information about the cybersecurity risks we face, refer to “Item 1A. Risk Factors.” Governance One of the key functions of our Board is informed oversight of our risk management process. Our Board administers this oversight function directly through our Board as a whole, as well as through various standing committees of our Board that address risks inherent in their respective areas of oversight. Our Audit Committee, a subcommittee of our Board, is responsible for the foreseeable future. In addition, the termsoversight of any future debt agreements may preclude usrisks from paying dividends. As a result, capital appreciation,cybersecurity threats. The Audit Committee receives updates at least quarterly from our Head of Information Technology regarding developments in our information technology infrastructure and cybersecurity program. This includes updates, as appropriate, on key information technology initiatives, new and existing cybersecurity risks, how management is managing those risks, and, if any, material cybersecurity incidents and the impact to our business and performance. At the management level, our Head of Information Technology is responsible for assessing and managing risks from cybersecurity threats through oversight of our capital stock will be your sole sourceinformation technology infrastructure and cybersecurity program. The individual occupying this role has over 20 years of gainexperience in information technology and cybersecurity and has served in senior cybersecurity leadership positions for over 10 years. Thus, we believe our Head of Information Technology is well-qualified to serve in this role. Our Head of Information Technology conducts bi-weekly meetings with our information technology department to remain apprised of cybersecurity matters. If a cybersecurity incident were to occur, our Head of Information Technology may inform our Executive Vice President and Head of Technological Operations and/or Audit Committee, depending on the foreseeable future.severity of the incident in accordance with the established severity and response matrix as defined in our incident response plan. Item 1B. Unresolved Staff Comments
Not applicable.
Item 2. Properties Our corporate headquarters are located in Canton,Stoughton, Massachusetts, where we lease 19,33550,678 square feet of office space (including chemistry and pilot/formulation laboratories) under alaboratory space. We use this facility for commercial and general and administrative purposes. The corporate headquarters lease agreement that was amendedexpires in March 2015. The lease term terminates in August 2020, five years followingJuly 2029 and the date that the landlord delivered the expansion space with certain improvements substantially completed. The lease term may be extended for antwo additional five yearsfive-year terms at our election. We believe that our existing facility isfacilities are adequate for our current and expected future needs. We may seek to negotiate new leases or evaluate additional or alternate space for our operations. We believe that appropriate alternative space is readily available on commercially reasonable terms. Item 3. Legal Proceedings
Xtampza Litigation
We filed the NDA for Xtampza as a 505(b)(2) application, which allows us toDiscussion of legal matters is incorporated by reference data from an approved drug listed in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the Orange Book)Note 13, Commitments and Contingencies, in this case OxyContin OP. The 505(b)(2) process requires that we certify to the FDA and notify Purdue, as the holderConsolidated Financial Statements included in Item 8 of the NDA and any other Orange Book-listed patent owners, that we do not infringe anyPart II of the patents listed for OxyContin OP in the Orange Book, or that the patents are invalid. We made such certification and provided such noticethis Annual Report on February 11, 2015 and such certification documented why Xtampza does not infringe any of the 11 Orange Book listed patents for OxyContin OP, five of which have been invalidated in court proceedings. Under the Hatch-Waxman Act of 1984, Purdue had the option to sue us for infringement and receive a stay of up to 30 months before the FDA could issue a final approval for Xtampza ER, unless the stay was earlier terminated.Form 10-K.
Purdue exercised its option and elected to sue us for infringement in the District of Delaware on March 24, 2015 asserting infringement of three of Purdue’s Orange Book-listed patents (Patent Nos. 7,674,799, 7,674,800, and 7,683,072) and a non-Orange Book-listed patent (Patent No. 8,652,497), and accordingly, received a 30-month stay of FDA approval.
The Delaware court transferred the case to the District of Massachusetts. After we filed a partial motion for judgment on the pleadings relating to the Orange Book-listed patents, the District Court of Massachusetts ordered judgment in our favor on those three patents, and dismissed the claims asserting infringement of those patents with prejudice. Upon dismissal of those claims, the 30-month stay of FDA approval was lifted. As a result, we were able to obtain final approval for Xtampza ER and launch the product commercially.
In November 2015, Purdue filed a follow-on suit asserting infringement of another patent, Patent No. 9,073,933, which was late-listed in the Orange Book and therefore could not trigger any stay of FDA approval. In June 2016, Purdue filed another follow-on suit asserting infringement of another non-Orange Book listed patent, Patent No. 9,155,717. In April 2017, Purdue filed another follow-on suit asserting infringement of another patent, Patent No. 9,522,919, which was late-listed in the Orange Book and therefore could not trigger any stay of FDA approval. Then, in September 2017, Purdue filed another follow-on suit asserting infringement of another non-Orange Book listed patent, Patent No. 9,693,961.
In October 2017, and in response to the filing of our Supplemental NDA (“sNDA”) seeking to update the drug abuse and dependence section of the Xtampza label, Purdue filed another suit asserting infringement of the ʼ933 and ʼ919 patent. We filed a motion to dismiss that action, and the Court granted our motion on January 16, 2018.
The current suits have been consolidated by the District of Massachusetts, where Purdue continues to assert infringement of five patents: the ʼ497 patent, the ʼ933 patent, the ʼ717 patent, the ʼ919 patent, and the ʼ961 patent. None of these suits are associated with any stay of FDA approval for the Xtampza drug product. Purdue has made a demand for monetary relief but has not quantified their alleged damages. Purdue has also requested a judgment of infringement and an injunction on the sale of our products accused of infringement. We have denied all claims and seek a judgment that the patents are invalid and/or not infringed by us; we are also seeking a judgment that the case is exceptional, with an award to us of our fees for defending the case.
The parties are in the early stages of fact discovery. Written discovery has commenced with depositions expected to commence during the second half of 2018. A claim construction and summary judgment hearing was held on June 1, 2017. On November 21, 2017, the Court issued its claim construction ruling, construing certain claims of the ʼ933, ʼ497, and ʼ717 patents. At this time, the Motion for Summary Judgment, which asserted that claims of the ’933, ’497, and ’717 patents are invalid and not infringed, remains pending. We are not able to predict with certainty when the Court will decide our motion. The Scheduling Order has been amended to stay the close of fact discovery until after the Court decides our Motion for Summary Judgment. No trial date has been scheduled.
We are, and plan to continue, defending this case vigorously. At this stage, we are unable to evaluate the likelihood of an unfavorable outcome or estimate the amount or range of potential loss, if any.
Nucynta Litigation
On February 7, 2018, Purdue filed a patent infringement suit against Collegium NF, LLC and Collegium Pharmaceutical, Inc. in the District of Delaware. Specifically, Purdue argues that our sale of immediate release and extended release Nucynta infringes U.S. Patent Nos. 9,861,583, 9,867,784, and 9,872,836. Purdue has made a demand for monetary relief in the Complaint but has not quantified its alleged damages. Our response to the Complaint is currently due April 9, 2018.
We plan to defend this case vigorously. At this stage, we are unable to evaluate the likelihood of an unfavorable outcome or estimate the amount or range of potential loss, if any.
Teva Litigation
We filed the NDA for Xtampza as a 505(b)(2) application, which allows us to reference data from an approved drug listed in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the
Orange Book), in this case OxyContin OP.We have twelve patents listed in the FDA Orange Book as covering our abuse-deterrent product and methods of using it to treat patients: Patents Nos. 7,399,488; 7,771,707; 8,449,909; 8,557,291; 8,758,813; 8,840,928; 9,044,398; 9,248,195; 9,592,200; 9,682,075; 9,737,530 and 9,763,883.
Teva Pharmaceuticals USA filed a Notice Letter of Patent Certification against all twelve listed patents, alleging that they were invalid and/or not infringed by the proposed oxycodone product that is the subject of Teva’s ANDA. On February 22, 2018—within the 45-day period that gives us a 30-month stay on FDA approval of Teva’s ANDA while the parties have an opportunity to litigate—we sued Teva in the District of Delaware on eleven of the patents listed in the Orange Book. The case was assigned to the Hon. Judge Stark.
We plan to assert and defend our intellectual property vigorously in this case. At this stage, we are unable to evaluate the likelihood of an unfavorable outcome or estimate the amount or range of potential loss, if any.
Opioid-Related Request and Subpoenas
We, like a number of other pharmaceutical companies, have received subpoenas or civil investigative demands related to opioid sales and marketing. We have received such subpoenas or civil investigative demands from the Offices of the Attorney General of each of Washington, New Hampshire, and Massachusetts. We are currently cooperating with the each of the foregoing states in their respective investigations.
Item 4. Mine Safety Disclosures
Not applicable. PART II Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities Market Information Our common stock ishas been publicly traded on the NASDAQ Global Select Market under the symbol “COLL” since May 7, 2015. Prior to May 7, 2015, there was no public trading market for our common stock. The following table sets forth, for the periods indicated, the high and low sales prices for our common stock as reported on NASDAQ: | | | | | | | Year Ended December 31, 2017 | | High | | Low | First quarter | | $ | 17.60 | | $ | 9.88 | Second quarter | | $ | 13.20 | | $ | 7.37 | Third quarter | | $ | 13.47 | | $ | 9.03 | Fourth quarter | | $ | 20.92 | | $ | 9.01 | | | | | | | | Year Ended December 31, 2016 | | High | | Low | First quarter | | $ | 28.47 | | $ | 13.80 | Second quarter | | $ | 20.03 | | $ | 11.55 | Third quarter | | $ | 20.25 | | $ | 8.24 | Fourth quarter | | $ | 20.55 | | $ | 13.81 |
Holders As of February 28, 2018,January 31, 2024, there were 3612 holders of record of our common stock. The number of holders of record does not include beneficial owners whose shares are held by nominees in street name. Dividends We have never declared or paid cash dividends on our common stock, and we do not expect to pay any cash dividends on our common stock in the foreseeable future. Stock Performance Graph The following graph shows a comparison from May 7, 2015,sets forth the date on which our common stock first began trading on the NASDAQ Global Select Market, of theCompany’s total cumulative shareholder return as compared to the NASDAQ Composite Index and the NASDAQ Biotechnology Index for the 5-year period beginning on December 31, 2018 through December 31, 2023. Total cumulative shareholder return assumes an assumed investment of $100.00 in cash in our common stock asat the beginning of the 5-year period compared to the same investment in the NASDAQ Composite Index and the NASDAQ Biotechnology Index, all through December 31, 2017.Index. Such returns are based on historical results and are not intended to suggest future performance. Data for the NASDAQ Composite Index and NASDAQ Biotechnology Index assume reinvestment of dividends, however no dividends have been declared on our common stock to date. 

| | | | | | | | | | | | | | December 31, | December 31, | $100 investment in stock or index | | May 7, 2015 | | 2016 | 2017 | Collegium Pharmaceutical, Inc. (COLL) | | $ | 100.00 | | $ | 126.69 | $ | 150.20 | NASDAQ Composite Index (IXIC) | | $ | 100.00 | | $ | 109.84 | $ | 139.59 | NASDAQ Biotechnology Index (NBI) | | $ | 100.00 | | $ | 79.90 | $ | 93.20 |
| | | | | | | | | December 31, | | December 31, | $100 investment in stock or index | | 2018 | | 2023 | Collegium Pharmaceutical, Inc. (COLL) | | $ | 100.00 | | $ | 179.27 | NASDAQ Composite Index (IXIC) | | $ | 100.00 | | $ | 226.24 | NASDAQ Biotechnology Index (NBI) | | $ | 100.00 | | $ | 143.60 |
The performance graph and related information shall not be deemed to be “soliciting material” or to be “filed” with the SEC, nor shall such information be incorporated by reference into any future filing under the Securities Act, except to the extent that we specifically incorporate it by reference into such filing.
Recent Sales of Unregistered Securities There were no unregistered sales of equity securities during the period covered by this Annual Report on Form 10-K. Use of Proceeds
In March 2017, we commenced an “at-the-market” offering of our common stock and entered into a Controlled Equity
Offering Sales Agreement (the “ATM Sales Agreement”) with Cantor Fitzgerald, as agent, pursuant to which we may issue and sell, from time to time, shares of our common stock having an aggregate offering price of up to $60.0 million. As of December 31, 2017, we had sold an aggregate of 3,126,998 shares of common stock under the ATM Sales Agreement at an average gross sales price of $11.36 per share, generating net proceeds of $34.3 million after deduction of underwriting discounts and commissions and expenses payable by us. The proceeds from the sales were used to fund
the continued commercialization of Xtampza, research and development efforts of our other product candidates, working capital and other general corporate purposes.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers We did notThe following table sets forth shares of common stock repurchased under our repurchase anyprogram authorized by our Board of our equity securitiesDirectors in January 2023 (the “2023 Repurchase Program”), as well as shares transferred to us from employees in satisfaction of minimum tax withholding obligations associated with the vesting of performance share units and restricted stock units during the fourth quarter of the fiscal yearthree months ended December 31, 2017. 2023:
| | | | | | | | | | Period | Total number of shares purchased | | Average Price Paid per Share | | Total number of shares purchased as part of publicly announced plans or programs (1) | | Maximum approximate dollar value of Shares that may yet be purchased under the plans or programs
(in thousands) | October 1, 2023 through October 31, 2023 | 467,247 | | $ | 23.10 | | 462,442 | | $ | 50,000 | November 1, 2023 through November 30, 2023 | 867,532 | | | 27.09 | | 865,426 | | | 30,000 | December 1, 2023 through December 31, 2023 | 59,791 | | | 27.12 | | 57,349 | | | 25,000 | Total | 1,394,570 | (2) | $ | 25.75 | | 1,385,217 | (2) | $ | 25,000 |
| (1) | The 2023 Repurchase Program was announced on January 4, 2023. The 2023 Repurchase Program provided for the repurchase of up to $100.0 million of outstanding shares of our common stock at any time or times through December 31, 2023. The 2023 Repurchase Program expired on December 31, 2023 with approximately $25.0 million available for repurchase at the time of expiration. |
| (2) | The difference, if any, between the total number of shares purchased and the total number of shares purchased as part of a publicly announced program relates to common stock withheld by us for employees to satisfy their tax withholding obligations arising upon the vesting of performance share units and restricted stock units granted under our Amended and Restated 2014 Stock Incentive Plan. |
Item 6. Selected Financial Data[Reserved] You should read the following selected financial data together with our consolidated financial statements and the related notes appearing elsewhere in this Form 10-K and “Management’s
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” section of this Form 10-K. The selected historical financial information in this section is not intended to replace our financial statements and the related notes thereto. Our historical results are not necessarily indicative of results to be expected in any period in the future. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Years ended December 31, | | 2017 | | 2016 | | 2015 | | 2014 | | 2013 | | (in thousands, except share and per share amounts) | Statement of Operations Data: | | | | | | | | | | | | | | | Product revenues, net | $ | 28,476 | | $ | 1,711 | | $ | — | | $ | — | | $ | — | Costs and expenses | | | | | | | | | | | | | | | Cost of product revenues | | 2,595 | | | 213 | | | — | | | — | | | — | Research and development | | 8,572 | | | 14,948 | | | 7,975 | | | 14,959 | | | 14,157 | Selling, general and administrative | | 92,756 | | | 80,632 | | | 18,932 | | | 2,706 | | | 1,885 | Total costs and expenses | | 103,923 | | | 95,793 | | | 26,907 | | | 17,665 | | | 16,042 | Loss from operations | | (75,447) | | | (94,082) | | | (26,907) | | | (17,665) | | | (16,042) | Interest income (expense), net | | 582 | | | (94) | | | (439) | | | (252) | | | (76) | Other income (expense), net | | — | | | — | | | 91 | | | — | | | (79) | Net loss | $ | (74,865) | | $ | (94,176) | | $ | (27,255) | | $ | (17,917) | | $ | (16,197) | Basic and diluted net loss per common share(1): | $ | (2.47) | | $ | (3.88) | | $ | (1.48) | | $ | (22.72) | | $ | (4.06) | Weighted-average shares used to compute loss per common share(1): | | 30,265,262 | | | 24,262,945 | | | 13,542,282 | | | 933,997 | | | 1,697,044 |
(1)See Note 3 to our consolidated financial statements included elsewhere in this Form 10-K for an explanation of the method used to calculate net loss per common share attributable to common shareholders, including the method used to calculate the number of shares used in the computation of the per share amount.
| | | | | | | | | | | | | | | | | | As of December 31, | | | 2017 | | 2016 | | 2015 | | 2014 | | 2013 | Balance Sheet Data: | | | | | | | | | | | | | | | | Cash and cash equivalents | | $ | 118,697 | | $ | 153,225 | | $ | 95,697 | | $ | 1,634 | | $ | 7,551 | Working capital(1) | | | 101,996 | | | 132,979 | | | 88,451 | | | (5,921) | | | 5,643 | Total assets | | | 135,568 | | | 162,017 | | | 97,718 | | | 5,090 | | | 9,034 | Other long-term liabilities | | | — | | | 1,513 | | | 4,214 | | | 6,914 | | | 834 | Total shareholders’ equity (deficit) | | | 104,080 | | | 134,908 | | | 85,072 | | | (89,348) | | | (68,225) |
| (1)
| | Working capital is calculated as current assets minus current liabilities.
|
Operations Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and related notes appearing elsewhere in this Annual Report on Form 10‑-K. The following discussion contains forward‑lookingforward-looking statements that involve risks, uncertainties and uncertainties.assumptions. Our actual results and the timing of certain events could differ materially from those anticipated in these forward‑lookingforward-looking statements as a result of certainmany factors. We discuss factors including those discussedthat we believe could cause or contribute to these differences below and aselsewhere in this Form 10-K, including those set forth under “Forward-looking Statements” and “Risk Factors.Factors,” Please also referas revised and supplemented by those risks described from time to time in other reports which we file with the section under heading “Forward‑Looking Statements.”SEC. Overview We are building a leading, diversified specialty pharmaceutical company focused on becomingcommitted to improving the leaderlives of people living with serious medical conditions. We commercialize our pain portfolio, consisting of Xtampza ER, Nucynta ER and Nucynta IR (collectively the “Nucynta Products”), Belbuca, and Symproic, in responsible pain management by developing and commercializing innovative, differentiated products for patients suffering from pain. Our first product, the United States. Xtampza isER, an abuse-deterrent, extended-release, oral formulation of oxycodone, a widely prescribed opioid medication. In April 2016,was approved by the U.S.United States Food and Drug Administration or FDA, approved our new drug application, or NDA, filing for Xtampza(“FDA”) in April 2016 for the management of severe and persistent pain severe enough to requirethat requires an extended treatment period with a daily around-the-clock, long-term opioid treatmentanalgesic and for which alternative treatment options are inadequate. Certain human abuse potential studies are includedWe commercially launched Xtampza ER in the approved label, as well as data supporting the administration of the product as a sprinkle or administered through feeding tubes. In June 2016, we announced the commercial launch of Xtampza. In October 2016, we announced the submission of a New Drug Submission to Health Canada seeking marketing approval of Xtampza for the same indication for which we obtained approval from the FDA.2016. Xtampza has the same active ingredient as OxyContin OP, which is the largest selling abuse-deterrent, extended-release opioid in the United States by dollars, with $1.7 billion in U.S. sales in 2017. We conducted a comprehensive preclinical and clinical program for Xtampza consistent with FDA guidance on abuse-deterrence. These studies and clinical trials demonstrated that chewing, crushing and/or dissolving Xtampza, and then taking it orally or smoking, snorting, or injecting it did not meaningfully change its drug release profile or safety characteristics. By contrast, clinical trials performed by us and others — including head-to-head clinical trials comparing Xtampza with OxyContin OP — have shown that drug abusers can achieve rapid release and absorption of the active ingredient by manipulating OxyContin OP using common household tools and methods commonly available on the Internet. In November 2017, we announced the approval of a Supplemental New Drug Application to the FDA for Xtampza to include comparative oral pharmacokinetic data from a clinical study evaluating the effect of physical manipulation by crushing Xtampza compared with OxyContin OP and a control (oxycodone hydrochloride immediate-release), results from an oral human abuse potential study and the addition of an oral abuse deterrent claim.
In addition, our preclinical studies and clinical trials have shown that the contents of the Xtampza capsule can be removed from the capsule and sprinkled on food or into a cup, and then directly into the mouth, or administered through feeding tubes, without compromising their drug release profile, safety or abuse-deterrent characteristics. By contrast, OxyContin OP, which is formulated in hard tablets, has a black box warning label stating that crushing, dissolving, or chewing can cause rapid release and absorption of a potentially fatal dose of the active ingredient. We believe that Xtampza can address the pain management needs of patients in the United States who suffer from chronic pain and have difficulty swallowing.
In December 2017, we entered into a Commercialization Agreement with Depomed, Inc., or Depomed, pursuant to which Depomed agreed to grant us a sublicense of certain of its intellectual property related to Nucynta ER and Nucynta IR, or theThe Nucynta Products for commercializationare extended-release (“ER”) and immediate-release (“IR”) formulations of such products in the United States, the District of Columbia and Puerto Rico.tapentadol. Nucynta ER is an extended release formulation of tapentadol that is indicated for the management of severe and persistent pain severe enough to requirethat requires an extended treatment period with a daily around‑the‑clock, long term opioid treatment,analgesic, including neuropathic pain associated with diabetic peripheral neuropathy in adults, and for which alternate treatment options are inadequate. Nucynta IR is an immediate release formulation of tapentadol that is indicated for the management of moderate to severe acute pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate in adults.
Since 2010, we have devoted substantially all of our resources to the development of our patented DETERx platform technology, the preclinicaladults and clinical advancement of our product candidates, pre-commercialization activitiespediatric patients aged 6 years and the creation and protection of related intellectual property. Since 2011, we have generated limited revenue from product sales and we continue to incur significant research, development and other expenses related to our ongoing operations. Prior to our initial public offering of common stock, or IPO, in May 2015, we funded our operations primarily through the private placement of preferred stock, convertible notes and commercial bank debt. Since our IPO, we have funded our operations primarily through the proceeds of public offerings and sale of our equity securities.
Outlook
We expect to continue to incur significant commercialization expenses related to marketing, manufacturing, distribution, selling and reimbursement activities. We are detailing Xtampza to approximately 11,000 physicians who write approximately 58% of the branded extended-release oral opioid prescriptions in the United Statesolder with a sales teambody weight of approximately 131 sales representatives. In addition, we deploy a separate, hospital focused sales team.
at least 40 kg. We began shipping and recognizing product sales on the Nucynta Products onin January 9, 2018 and we began commercial promotion ofmarketing the Nucynta Products in February 2018. We are detailingIn August 2023, the FDA granted New Patient Population exclusivity in pediatrics for Nucynta ProductsIR. This grant extended the period of U.S. exclusivity for Nucynta IR from June 27, 2025 to substantiallyJuly 3, 2026. On March 22, 2022, we acquired BioDelivery Sciences International, Inc. (“BDSI”), a specialty pharmaceutical company working to deliver innovative therapies for individuals living with serious and debilitating chronic conditions (the “BDSI Acquisition”). Upon closing of the same physicians to whomBDSI Acquisition, we detail Xtampza, leveraging our existing sales organization. We will payacquired Belbuca and Symproic. Belbuca is a royalty to Depomed on all revenues frombuccal film that contains buprenorphine, a Schedule III opioid, and was approved by the sale of Nucynta Products based on certain net sales thresholds,FDA in October 2015 for severe and persistent pain that requires an extended treatment period with a minimum royaltydaily opioid analgesic and for which alternative options are inadequate. Symproic was approved by the FDA in March 2017 for the treatment of $135.0 million per year during the first four years of the Commercialization Agreement, subjectopioid-induced constipation (“OIC”) in adult patients with chronic non-cancer pain, including patients with chronic pain related to certain conditions. If Depomedprior cancer or its contract manufacturers are unabletreatment who do not require frequent (e.g., weekly) opioid dosage escalation. We began shipping and recognizing product sales related to deliver a certain percentage of ordered quantities of the Nucynta Products for a period of two months or longerBelbuca and Symproic in calendar year 2018, then Depomed may be required to make a payment (or offset the minimum royalties) to ensure that we receive a minimum level of gross profit for 2018.March 2022. We have never been profitable and have incurred net losses in each year since inception. We incurred net losses of $74.9 million, $94.2 million and $27.3 million for the years ended December 31, 2017, 2016 and 2015, respectively. As of December 31, 2017, we had an accumulated deficit of $298.0 million. Substantially all of our net losses resulted from costs incurred in connection with our research and development programs and from selling, general and administrative costs associated with our operations. We expect to continue to incur net losses as we continue to commercialize Xtampza and the Nucynta Products. Our net losses may fluctuate significantly from quarter to quarter and year to year. We expect our expenses will increase in connection with our ongoing activities as we:
| ·
| | expand our sales and marketing efforts for Xtampza and the Nucynta Products, including hiring additional personnel to expand our commercial organization;
|
| ·
| | expand our regulatory and compliance functions;
|
| ·
| | conduct clinical trials of our product candidates;
|
| ·
| | continue scale-up and improvement of our manufacturing processes;
|
| ·
| | continue our research and development efforts;
|
| ·
| | manufacture preclinical study and clinical trial materials;
|
| ·
| | maintain, expand and protect our intellectual property portfolio;
|
| ·
| | seek regulatory approvals for our product candidates that successfully complete clinical trials;
|
| ·
| | hire additional clinical, quality control and technical personnel to conduct our clinical trials;
|
| ·
| | hire additional scientific personnel to support our product development efforts;
|
| ·
| | expand operational, financial and management systems; and
|
| ·
| | hire additional selling, general and administrative personnel to operate as a commercial stage public company.
|
We believe that our cash and cash equivalents at December 31, 2017, together with expected cash inflows from the commercialization of Xtampza and the Nucynta Products, will enable us to fund our operating expenses, debt service and capital expenditure requirements into 2020. In addition, we may in the future seek to fund our operations through additional public or private equity or debt financings or other sources. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. If we are unable to obtain financing or increase profitability, the related lack of liquidity will have a material adverse effect on our operations and future prospects.
Financial Operations Overview Product Revenues Product Revenues Product revenuerevenues through the year ended December 31, 2017 has been2023 were primarily generated from product sales of Xtampza. Product sales of Xtampza ER, the Nucynta Products, Belbuca, and Symproic. In accordance with Accounting Standards Codification Topic 606, Revenue from Contracts with Customers, (“ASC 606”)product sales are recorded upon delivery of products to customers (upon the transfer of control of the product to the customer), net of a provision for estimated chargebacks, rebates, sales incentives and allowance,allowances, distribution service fees, as well as estimated productand returns.
Cost of Product Revenues Cost of product revenues include amortization and impairment expense for the intangible assets acquired in connection with business combinations and asset acquisitions, royalty expenses, the cost of active pharmaceutical ingredient, (“API”), the cost of producing finished goods that correspond with revenue for the reporting period, as well as certain period costs related to freight, packaging, stability and quality testing. Refer to Note 5, License Agreements, and Note 11, Goodwill andIntangible Assets, for further detail around the intangible assets acquired from the BDSI Acquisition, the Nucynta Intangible Asset and royalty expenses. Research and Development Expenses Research and development expenses consisthave historically consisted of product development expenses incurred in identifying, developing, and testing product candidates including stock-based compensation; costs associated with conducting our DETERx platform technologyclinical and product candidates programs.non-clinical activities, including clinical and non-clinical trials that we conduct for post-marketing requirements; and costs for laboratory supplies, depreciation of lab equipment, and other expenses including allocated expenses for rent and maintenance of facilities. These costs arehave historically been expensed as incurred and include: | ·
| | compensation and employee‑related costs, including stock‑based compensation;
|
| ·
| | costs associated with conducting our preclinical, clinical and regulatory activities, including fees paid to third‑party professional consultants and service providers;
|
| ·
| | costs incurred under clinical trial agreements;
|
| ·
| | costs for laboratory supplies;
|
| ·
| | costs to acquire, develop and manufacture preclinical study and clinical trial materials; and
|
| ·
| | facilities, depreciation and other expenses including allocated expenses for rent and maintenance of facilities.
|
incurred. We cannot determine with certainty the timingAs of initiation, the duration or the completion costs of current or future preclinical studies and clinical trials of our product candidates. At this time, due to the inherently unpredictable nature of preclinical and clinical development, and given the early stage of our product candidates,April 1, 2022, we are unable to estimate with any certainty the costs we will incur and the timelines required for the development of our product candidates. Clinical and preclinical development timelines, the probability of success and development costs can differ materially from expectations. In addition, we cannot forecast which product candidates may be subject to future collaborations, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
Ourfocused entirely on commercial products rather than research and development has been focused primarily on developing our DETERx platform technology and Xtampza. Accordingly, historically we have not trackedredirected resources from research and development costs by project. In addition, we use our employee and infrastructure resources across multipleactivities. As such, there were no expenses incurred in research and development projects. We expect to track specific project costs when additional product candidates enter clinical trials in humans.after the three months ended March 31, 2022.
Selling, General and Administrative Expenses Selling, general and administrative expenses consist primarily of salaries and employee‑relatedemployee-related costs, including stock‑basedstock-based compensation and travel expenses for our employees in executive, finance, sales and marketing and administrative functions.employees. Other selling, general and administrative expenses include facility‑relatedfacility-related costs and professional fees for directors, accounting and legal services, and expenses associated with obtaining and maintaining patents. We anticipate that As we continue to invest in the commercialization of our products, we expect our selling, general and administrative expenses will increaseto continue to be substantial for the foreseeable future.
Interest Expense Interest expense consists primarily of cash and non-cash interest costs related to our debt, including the term loan issued in March 2022 in connection with the future as we increase our administrative headcount to support our continued researchBDSI Acquisition (the “2022 Term Loan”), the term notes (the “2020 Term Loan”) and developmentconvertible notes (the “2026 Convertible Notes”) issued in February 2020 in connection with the Nucynta Acquisition, and potential commercialization of our product candidates,convertible notes issued in additionFebruary 2023 (the “2029 Convertible Notes”). On March 22, 2022 the outstanding balance related to the continued expansion2020 Term Loan was fully paid in connection with the closing of our commercialization efforts for Xtampzathe BDSI Acquisition and establishment of the Nucynta Products. We also anticipate increased expenses related to audit, legal, regulatory and tax‑related services associated with maintaining compliance with exchange listing and SEC requirements, director and officer insurance premiums, and investor relations costs associated with being a public company.2022 Term Loan. Other Expense, NetInterest Income
Other expense, netInterest income consists of interest earned on our cash, cash equivalents, and marketable securities.
Provision for Income Taxes The provision for income taxes reflects expense or tax benefit for federal and interest expense. Emerging Growth Company Status
Section 107state income taxes, as well as the impact of non-deductible expenses. During the JOBS Act provides that an emerging growth company can take advantage ofyear ended December 31, 2021, we removed the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. Thus, an emerging growth company can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standardsvaluation allowance on the relevant dates on which adoptionsubstantial majority of such standards is required for companies that are not emerging growth companies.our deferred tax assets, resulting in the recognition of a discrete deferred tax benefit of $78.0 million in 2021.
Critical Accounting Policies and Significant Judgments and Estimates Our management’s discussion“Management’s Discussion and analysisAnalysis of our financial conditionFinancial Condition and resultsResults of operationsOperations” are based on our consolidated financial statements, which have been prepared in accordance with generally accepted accounting principles in the United States (GAAP)of America (“GAAP”). The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenue and expenses and the disclosure of contingent assets and liabilities in our consolidated financial statements. We evaluate our estimates and judgments on an ongoing basis. Estimates include revenue recognition, including the estimates of product returns, units prescribed, discounts and allowances related to commercial sales of Xtampza,our products, estimates related to the fair value of assets acquired and liabilities assumed, including acquired intangible assets and the fair value of inventory acquired, estimates utilized in the ongoing valuation of inventory related to potential unsalable product, estimates of useful lives with respect to intangible assets, accounting for stock-based compensation, contingencies, intangible assets and deferred tax valuation reserves.allowances. We base our estimates and assumptions on historical experience when available and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Actual results may differ from these estimates under different assumptions or conditions. We believe that several accounting policies are important to understanding our historical and future performance. We refer to these policies as “critical” because these specific areas generally require us to make judgments and estimates about matters that are uncertain at the time we make the estimate, and different estimates—which also would have been reasonable—could have been used, which would have resulted in different financial results. While our significant accounting policies are described in more detail in the notes Note 2, Summary of Significant Accounting Policies, to our consolidated financial statements appearing elsewhere in this Form 10-K,, we believe the following accounting policies to be most critical to the significant judgments and estimates used in the preparation of our consolidated financial statements. Revenue Recognition Our accounting policy for revenue recognition will have a substantial impact on reported results and relies on certain estimates. Estimates are based on historical experience, current conditions and on various other assumptions that we believe are reasonable, under the circumstances, the results of which form the basis for making judgments about the carrying values of assets, liabilities and equity and the amounts of revenues and expenses. Actual results may differ from these estimates under different assumptions or conditions. Product Revenue Our only source of revenue to date has been generated by sales of our products, which are primarily sold to distributors (“customers”), which in turn sell the product to pharmacies and others for the treatment of patients. Revenue for product sales is recognized when there is persuasive evidencea customer obtains control of promised goods or services, in an arrangement; title and risk of loss have passedamount that reflects the consideration which the entity expects to the customer, whichreceive in exchange for those goods or services. This generally occurs upon delivery;delivery to our customers when estimated provisions for chargebacks, rebates, sales incentives and allowances, distribution service fees, and returns are reasonably determinable; and when collectability is reasonably assured. Productdeterminable. Therefore, product sales are recorded upon delivery to our customers net of estimated chargebacks, rebates, sales incentives and allowances, distribution service fees, as well as estimated product returns. Beginning in the third quarter of 2017, we determined that we have sufficient experience with sales of Xtampza to estimate our returns at time of shipment. We sell our products primarily to distributors and retailers (“customers”), which in turn sell the product to pharmacies for the treatment of patients. We provide the right of return to our customers for a limited time before and after the product expiration date. As a result of our experience to date with Xtampza sales, we determined that we can reasonably estimate the amount of future product returns. This determination has enabled us to recognize revenue earlier on the sell-in method, net of a provision for estimated returns, because we can record revenue once sold to the customer rather than waiting until the product is sold to the end user on a sell-through method. We recorded a one-time $4.4 million increase to revenues during the three months ended September 30, 2017 as a result of our change to the sell-in method in the third quarter of 2017.
Sales Deductions Sales deductions consist primarily of provisions for: (i) rebates and incentives, including managed care rebates;rebates, government rebates;rebates, co-pay program incentives;incentives, and sales incentives and allowances; provisions(ii) product returns, including return estimates for product returns;our products; and (iii) trade allowances and chargebacks, including fees for distribution service fees;fees, prompt pay discounts;discounts, and chargebacks. We estimate the amount of variable consideration that should be included in revenue under the expected value method for all sales deductions other than trade allowances, which are estimated under the most likely amount method. These deductionsprovisions reflect our best estimates of the amount of revenue to which we are recorded as reductions to revenue inentitled based on the same period as the related sales are recognized. Reservesterms of our contracts. Provisions for rebates and incentives are based on estimatesthe estimated amount of the amounts earned orrebates and incentives to be claimed on the related sales. Estimatessales from the period. As our rebates and incentives are based on ourproducts dispensed to patients, we are required to estimate the expected value of claims at the time of product delivery to distributors. Given that distributors sell the product to pharmacies, which in turn dispense the product to patients, claims can be submitted significantly after the related sales are recognized. Our estimates of these claims are based on the historical experience of existing or similar programs, including current contractual and statutory requirements, specific known market events and trends, industry data, and forecasted customer buying and payment patterns. As a result, we estimate the accrualsestimated distribution channel inventory levels. Accruals and related reserves required for amounts payable under these programs. rebates and incentives are adjusted as new information becomes available, including actual claims. If actual results vary, we may need to adjust these estimates, which could have an effect on earnings in the period of the adjustment. Inventory
Upon approval of Xtampza by the FDA in April 2016, we began capitalizing inventory costsProvisions for product returns, including returns for Xtampza, the Nucynta Products, Belbuca and Symproic, are based on product-level returns rates, including processed as well as unprocessed return claims, in preparationaddition to relevant market events and other factors. Estimates of the future product returns are made at the time of revenue recognition to determine the amount of consideration to which we expect to be entitled (that is, excluding the products expected to be returned).
At the end of each reporting period, we analyze trends in returns rates and update our assessment of variable consideration for returns. To the extent we receive amounts in excess of what we expect to be entitled to receive due to a product launch. Priorreturn, we do not recognize revenue when we transfer products to April 2016, we expensed costs associatedcustomers but instead recognize those excess amounts received as a refund liability. We update the measurement of the refund liability at the end of each reporting period for changes in expectations about the amount of refunds with Xtampza, including raw materials, work in process and finished goods,the corresponding adjustments recognized as research and development expense. revenue (or reductions of revenue). We have not capitalized inventory costs relatedprovide the right of return to our other drug development programs.customers for an 18-month window beginning six months prior to expiration and up until twelve months after expiration. Our customers short-pay an existing invoice upon notice of a product return claim. Adjustments to the preliminary short-paid claims are processed when the return claim is validated and finalized. Our return policy requires that product is returned and that the return is claimed within the 18-month window. Refer to Note 3, Revenue from Contracts with Customers, for more information. Provisions for trade allowances and chargebacks are primarily based on customer-level contractual terms. Accruals and related reserves are adjusted as new information becomes available, which generally consists of actual trade allowances and chargebacks processed. Actual results may differ from these estimates under different assumptions or conditions. Business Combination Accounting and Valuation of Acquired Assets We have capitalized $1.8 millioncompleted the BDSI Acquisition on March 22, 2022, which was accounted for as a business combination. To determine whether the acquisition should be accounted for as a business combination or as an asset acquisition, we made certain judgments regarding whether the acquired set of inventoryactivities and assets met the definition of a business. Judgment is required in assessing whether the acquired processes or activities, along with their inputs, would be substantive to constitute a business, as defined by U.S. GAAP. The acquisition method of accounting requires that we recognize the assets acquired and liabilities assumed at their acquisition date fair values. Goodwill is measured as the excess of consideration transferred over the acquisition date net fair values of the assets acquired and the liabilities assumed. The determination of the fair value of the acquired assets and liabilities assumed is a critical accounting estimate because the estimation of fair values requires significant management judgment and requires various assumptions based on non-observable inputs that are included in valuation models. An income approach, which generally relies upon projected cash flow models, is used in estimating the fair value of the acquired intangible assets and the fair value of acquired inventory. These cash flow projections are based on management’s estimates of economic and market conditions including the estimated future cash flows from revenues of acquired assets, the timing and projection of costs and expenses and the related profit margins, tax rates, and an appropriate discount rate. During the measurement period, which occurs before finalization of the purchase price allocation, changes in assumptions and estimates that result in adjustments to the fair values of assets acquired and liabilities assumed, if based on facts and circumstances existing at the acquisition date, are recorded on a retroactive basis as of December 31, 2017. We expect salesthe acquisition date, with the corresponding offset to goodwill. Any adjustments not based on facts and circumstances existing at the acquisition date, or if subsequent to the conclusion of the capitalized unitsmeasurement period, will be recorded to occur duringour consolidated statements of operations. Intangible Assets We record the next twelve months. We expect costsfair value of product revenues to increase due to the expected increases in net product sales of Xtampza and the fact that we had expensed all manufacturing costs as research and development expense in periods prior to FDA approval of Xtampza. The impact on cost of product revenues as a result of inventory not capitalized prior to FDA approval is immaterial. Impairment of Long‑Lived Assets
Long‑lived assets consist primarily ofacquired finite-lived intangible assets as of the transaction date. Intangible assets are then amortized over their estimated useful lives using either the straight-line method, or if reliably determinable, based on the pattern in which the economic benefit of the asset is expected to be utilized, which is generally based on our cash flow projections. Future events, such as competition, technological advances, or other changes, are subject to uncertainty and property and equipment.could cause subsequent evaluations to cash flow projections. We test long‑livedintangible assets for potential impairment whenever triggering events or circumstances present an indication of impairment. If the sum of expected undiscounted future cash flows of the long‑livedintangible assets (or asset group) is less than the carrying amount of such assets,
the long‑livedintangible assets would be written down to the estimated fair value, calculated based on the present value of expected future cash flows. As of December 31, 2017,2023, our only intangible asset wasassets included those acquired in connection with the licensed right to developBDSI Acquisition and commercialize Onsolis from BioDelivery Sciences International, Inc. (“BDSI”). Onsolis is a Transmucosal Immediate-Release Fentanyl film indicated for the management of breakthrough pain in cancer patients 18 years of age and older, who are already receiving and who are tolerant to opioid therapy for their underlying persistent cancer pain. On December 8, 2017, after a review of our product portfolio, we provided written notice to BDSI of the termination of the License and Development Agreement dated May 11, 2016, or the License Agreement, which termination will be effective pursuant to the terms of such agreement on March 8, 2018. Upon such termination of the License Agreement, our rights to develop and commercialize Onsolis will revert to BDSI. As such, we considered this notice a triggering event warranting an assessment of impairment indicators and test for recoverability. Based on the results of the impairment assessment and recoverability test, we recognized a full impairment of $1.8 million related to the license of Onsolis in December 2017.Nucynta Intangible Asset. Income Taxes We have not recognized any impairment losses on propertyutilize the asset and equipmentliability method of accounting for income taxes. Under this method, deferred tax assets for the years ended December 31, 2017, 2016 and 2015. Stock‑Based Compensation
We account for grants of stock options and restricted stock to employees based on their grant date fair value and recognize compensation expense over the vesting periods. We estimate the fair value of stock options as of the date of grant using the Black‑Scholes option pricing model, and we estimate the fair value of restricted stock awards and restricted stock unitsliabilities are determined based on the fair valuedifference between the financial statement carrying amounts and tax basis of assets and liabilities using enacted tax rates in effect for years in which the underlying common stock as determined by our board of directors or the value of the services provided, whichever is more readily determinable. We account for stock options, restricted stock awards and restricted stock units to non‑employees using the fair value approach. Stock options and restricted stock awards to non‑employees are subject to periodic revaluation over their vesting terms.
Stock‑based compensation expense represents the cost of the grant date fair value of employee stock option grants recognized over the requisite service period of the awards (usually the vesting period) on a straight‑line basis, net of estimated forfeitures. We estimate the fair value of stock option grants using the Black‑Scholes option pricing model, which requires the input of subjective assumptions, including (i) the risk‑free interest rate, (ii) the expected volatility of our stock, (iii) the expected term of the award and (iv) the expected dividend yield. The risk‑free interest rates for periods within the expected life of the option are based on the yields of zero‑coupon U.S. Treasury securities. Prior to our IPO, there was no public market for the trading of our common stock. Due to the lack of a public market for the trading of our common stock prior to our recent IPO and a lack of Company‑specific historical and implied volatility data, we have based our estimate of expected volatility on the historical volatility of a group of similar companies that are publicly traded. For these analyses, we have selected companies with comparable characteristics to ours, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected life of the stock‑based awards. We compute the historical volatility data using the daily closing prices for the selected companies’ shares during the equivalent period of the calculated expected term of our stock‑based awards. We will continue to apply this process until a sufficient amount of historical information regarding the volatility of our own stock price becomes available. The expected term represents the period of time that optionstemporary differences are expected to be outstanding. Because there was not enough historical exercise behavior through December 31, 2017, we determined the expected life assumption using the simplified method, which is an average of the contractual term of the option and the vesting period.reverse.
Fair Value of Common Stock. After our stock began trading on NASDAQ on May 7, 2015, the fair value of common stock underlying our options was determined by the closing price of our common stock on the date of the grant. Prior to the IPO, the fair value of the shares of our common stock underlying our stock options was determined by our board of directors. Because there was no public market for our common stock, our board of directors determined the fair value of our common stock at the time of grant of the option by consideringWe provide a number of objective and subjective factors, including valuations of comparable companies, sales of our convertible preferred stock to unrelated third parties, our operating and financial performance and general and industry specific economic outlook.
Net Operating Loss Carryforwards
As of December 31, 2017, we had a federal net operating loss, or NOL, carryforward of approximately $249.5 million
and state net operating loss carryovers of approximately $205.1 million, which are available to offset future taxable income. The U.S. federal NOL carryforwards begin to expire in 2022, and the state net operating losses begin to expire in 2030. We also had U.S. federal tax credits of approximately $3.4 million, and state tax credits of approximately $589,000. These tax attributes are generally subject to a limited carryover/carryback period, and are also subject to the annual limitations that may be imposed under Section 382 of the Code, or Section 382.
The TCJA generally will allow losses incurred after 2017 to be carried over indefinitely, but will limit the NOL deduction to the lesser of the NOL carryover or 80% of a corporation’s taxable income (subject to Code Sections 382/383 and other limitations). Also, there will be no carryback for losses incurred after 2017. Losses incurred prior to 2018 generally will be deductible to the extent of the lesser of a corporation’s NOL carryover or 100% of a corporation’s taxable income, and be available for twenty years from the period the loss was generated. We have not finalized our review of the impact of TCJA on the NOL rules, and the impact, if any, to our ability to utilize and carryover net operating losses.
The federal R&D credit generally has a twenty year carryover term, and our state R&D credit is generally available for a fifteen year carryover.
Under Section 382, if a corporation undergoes an “ownership change,” generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period, the corporation’s ability to use its pre-change NOLs and other pre-change tax attributes (such as research and development tax credits) to offset its post-change income may be limited. We may experience ownership changes in the future as a result of shifts in our stock ownership some of which are outside our control. We have not completed a current study to assess whether an ownership change has occurred or whether there have been multiple ownership changes since our formation. As a result, if we earn net taxable income, our ability to use our pre-change NOL carryforwards to offset U.S. federal taxable income may be subject to limitations, which could potentially result in increased future tax liability to us. In addition, at the state level, there may be periods during which the use of NOLs is suspended or otherwise limited, which could accelerate or permanently increase state taxes owed.
As of December 31, 2017 and 2016, we have provided a full valuation allowance for deferred tax assets including NOL and tax credit carryovers.
Uncertain Tax Positions and Income Taxes
We record uncertain tax positions on the basis of a two‑step process whereby (i) we determine whetherwhen it is more likely than not that deferred tax assets will not be realized. In determining the extent to which a valuation allowance for deferred tax positions will beassets is required, we evaluate all available evidence including projections of future taxable income, carryback opportunities, reversal of certain deferred tax liabilities, and other tax planning strategies, all of which are subject to uncertainty. Certain deferred tax assets, such as net operating losses and tax credits, expire at varying dates and are generally subject to annual limitations under Section 382 of the Internal Revenue Code of 1986, as amended (“IRC 382”). Significant judgment is required in making these evaluations, including comparing future annual income projections to the expiration dates and annual limitations of such assets. To the extent our future expectations change, we would have to assess the recoverability of these deferred tax assets at that time.
As a result of sustained positive earnings history as demonstrated through cumulative earnings, we are using projections of future taxable income as a source of realizing our deferred tax assets. We have maintained a valuation allowance on the basisportion of the technical merits of the positions and (ii) for thoseour deferred tax positionsassets that meet the more‑likely‑than‑are not recognition threshold, we recognize the largest amount of tax benefit that is more likely than 50% likelynot to be realized upon ultimate settlement with the relateddue to tax authority. We recognize interest and penalties related to unrecognized tax benefits within income tax expense. Any accrued interest and penalties are included within the related tax liability. During 2017, the Company identified unrecognized tax benefits associated with the IRS auditlimitation or other conditions of its 2015 corporate income tax return, and the risk$5.8 million as of claiming R&D credits without a formal R&D study. The gross unrecognized tax benefits associated with these two items at December 31, 2017 is $1.4 million.2023.
Results of Operations In this section, we discuss the results of our operations for the year ended December 31, 2023 compared to the year ended December 31, 2022. For a discussion of the year ended December 31, 2022 compared to the year ended December 31, 2021, refer to Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2022. Comparison of the Years Ended December 31, 2017, 20162023 and 20152022 The following table summarizes the results of our operations for the years ended December 31, 2017, 20162023 and 2015:2022: | | | | | | | Years Ended December 31, | | 2023 | | 2022 | | | (in thousands) | Product revenues, net | $ | 566,767 | | $ | 463,933 | Cost of product revenues | | | | | | Cost of product revenues (excluding intangible asset amortization) | | 94,838 | | | 118,190 | Intangible asset amortization and impairment | | 145,760 | | | 136,255 | Total cost of products revenues | | 240,598 | | | 254,445 | Gross profit | | 326,169 | | | 209,488 | Operating expenses | | | | | | Research and development | | — | | | 3,983 | Selling, general and administrative | | 159,208 | | | 172,186 | Total operating expenses | | 159,208 | | | 176,169 | Income from operations | | 166,961 | | | 33,319 | Interest expense | | (83,339) | | | (63,213) | Interest income | | 15,615 | | | 1,047 | Loss on extinguishment of debt | | (23,504) | | | — | Income (loss) before income taxes | | 75,733 | | | (28,847) | Provision for (benefit from) income taxes | | 27,578 | | | (3,845) | Net income (loss) | $ | 48,155 | | $ | (25,002) |
| | | | | | | | | | Years ended December 31, | | 2017 | | 2016 | | 2015 | | (in thousands) | Product revenues, net | $ | 28,476 | | $ | 1,711 | | $ | — | Cost of product revenues | | 2,595 | | | 213 | | | — | Research and development | | 8,572 | | | 14,948 | | | 7,975 | Selling, general and administrative | | 92,756 | | | 80,632 | | | 18,932 | Interest income (expense), net | | 582 | | | (94) | | | (348) | Net loss | $ | (74,865) | | $ | (94,176) | | $ | (27,255) |
Comparison of the Years Ended December 31, 2017 and 2016
Product revenues, net Product revenues, net were $28.5$566.8 million for the year ended December 31, 2017,2023 (“2023”), compared to $1.7$463.9 million for the year ended December 31, 2016.2022 (“2022”), representing a $102.9 million increase. The $26.8$102.9 million increase wasis primarily due to increases in revenue for Belbuca of $55.6 million, Xtampza ER of $38.6 million, the Nucynta Products of $6.3 million, and Symproic of $4.2 million, partially offset by decreases in other revenue of $1.8 million. The increase in revenue for Belbuca of $55.6 million and Symproic of $4.2 million is primarily due to a full year of revenue in 2023, compared to a partial year of revenue in 2022 due to the timing of the BDSI Acquisition. The increase in revenue for Xtampza ER of $38.6 million is primarily due to lower gross-to-net adjustments primarily related to an $18.6 millionprovisions for rebates and higher gross price, partially offset by lower sales volume and higher gross-to-net adjustments related to provisions for returns. The increase in sold-through unitsrevenue for the Nucynta Products of Xtampza, as well as a $3.8$6.3 million increase as a resultis primarily due to lower gross-to-net adjustments primarily related to provisions for rebates and returns and higher gross price, partially offset by decreased sales volume. Cost of changing to the sell-in method during the year ended December 31, 2017. In addition, a $4.4 million increase toproduct revenues was recorded in the third quarter of 2017 to recognize revenue from shipments from prior periods as a result of changing to the sell-in method in the third quarter of 2017. Cost of product revenues (excluding intangible asset amortization) was $2.6$94.8 million for the year ended December 31, 2017,2023, compared to $213,000$118.2 million for the year ended December 31, 2016.2022. The $2.4$23.4 million increasedecrease was primarily related to increased2022 including higher cost of product revenues related to the step-up basis in inventory acquired from BDSI as well as lower sales volume in 2023 for Xtampza and the Nucynta Products. Intangible asset amortization was $145.8 million for 2023, compared to $136.3 million for 2022. The $9.5 million increase in intangible asset amortization was due to a full year ended December 31, 2017.of amortization expense recognized in 2023 related to the intangible assets acquired from BDSI of which $435.0 million of consideration was allocated to our acquired intangible assets, compared to a partial year of amortization expense in 2022. This increase was partially offset by a decrease in amortization expense as a result of the FDA granting New Patient Population exclusivity in pediatrics for Nucynta IR to July 3, 2026, resulting in an extension of the estimated useful life of the underlying intangible asset. The intangible assets are amortized on a straight-line basis over the respective estimated useful lives. ResearchOperating expenses
We did not recognize research and development expenses in 2023, compared to $4.0 million recognized in 2022. The $4.0 million decrease was due to redirection of resources from research and development activities during 2022 as we shifted our focus to supporting our commercial products rather than research and development. Selling, general and administrative expenses were $8.6$159.2 million for the year ended December 31, 2017,2023, compared to $14.9$172.2 million for the year ended December 31, 2016.2022. The $6.3$13.0 million decrease was primarily related to: | ● | ·
| | a decrease in clinical trial costsacquisition related expenses classified as selling, general and administrative of $4.0 million due to the completion of certain clinical trials in 2016; $30.6 million; partially offset by |
| ● | ·
| | a decrease in research-related regulatory costs of $2.1 million following the commercial launch of Xtampza in 2016;
|
| ·
| | a decrease in Xtampza manufacturing costs of $1.9 million reflecting that, prior to April 2016, we expensed manufacturing costs associated with Xtampza as research and development expense; offset by
|
| ·
| | an increase in non-clinical trial costs of $932,000 relating to studies required to be conducted following FDA approval of Xtampza; and
|
| ·
| | an increase in salaries, wages, and benefits of $678,000$8.7 million, primarily due to increases in personnel costs for employees retained following the BDSI Acquisition and higher stock-based compensation expenses; |
| ● | an overall increase in researchaudit and development, including an increase in incentive compensation and stock-based compensation expense. |
Selling, general and administrativelegal expenses were $92.8 million for the year ended December 31, 2017, compared to $80.6 million for the year ended December 31, 2016. The $12.2 million increase was primarily related to:
| ·
| | an increase in salaries, wages and benefits of $15.8$5.0 million, primarily due to an increase from 234 to 250 employees, including the addition of a sales force of approximately 150 employees in the second quarter of 2016, and stock-based compensation expense;
$8.5 million litigation settlement; |
| ● | ·
| | an increase in legal fees of $2.1 million, primarily due to costs related to litigation;
|
| ·
| | an increase of $1.8 million due to the impairment charge relating to the termination of the License Agreement with BDSI; offset by
|
| ·
| | a decrease in PMR and other regulatory costs associated with FDA approval of Xtampza of $3.9 million, primarily due to higher one-time costs incurred upon the commercial launch of Xtampza in 2016;
|
| ·
| | a decrease in commercial, sales and marketing costs of $2.9 million, primarily due to higher costs incurred upon the commercial launch of Xtampza in 2016; and
|
| ·
| | a decrease in distribution and commercial manufacturing costs of $667,000.
|
Comparison of the Years Ended December 31, 2016 and 2015
Product revenues, net were $1.7 million for the year ended December 31, 2016, compared to zero for the year ended December 31, 2015. The $1.7 million increase was due to the commercial launch of Xtampza in June 2016.
Cost of product revenues was $213,000 for the year ended December 31, 2016, compared to zero for the year ended December 31, 2015. The $213,000 increase was due to the commercial launch of Xtampza in June 2016.
Research and development expenses were $14.9 million for the year ended December 31, 2016, compared to $8.0 million for the year ended December 31, 2015. The $6.9 million increase was primarily related to:
| ·
| | an increase in clinical trial costs of $5.2 million due to clinical trials with Xtampza and the commencement of clinical trials for our second product candidate;
|
| ·
| | an increase in salaries, wages and benefits of $1.6 million primarily due to headcount, bonuses and stock compensation expense; and
|
| ·
| | an increase in manufacturing and transfer costs of $1.2 million primarily related to the development of a manufacturing process for Onsolis; offset by
|
| ·
| | a decrease in consulting costs of $1.1 million primarily due to the completion of FDA advisory committee preparation in 2015.
|
Selling, general and administrative expenses were $80.6 million for the year ended December 31, 2016 compared to $18.9 million for the year ended December 31, 2015. The $61.7 million increase was primarily related to:
| ·
| | an increase in salaries, wages and benefits of $26.4 million primarily due to an increase from 35 to 206 employees,including the addition of a sales force of approximately 150 employees, and an increase in stock-based compensation expense;
|
| ·
| | an increase in sales and marketing costsexpenses of $15.9$3.1 million, primarily due to preparation forexpenses incurred to support the ongoing commercialization of products acquired from BDSI; and support of the commercial launch of Xtampza; |
| ● | ·
| | an increase in commercial costsregulatory expenses of $9.0$0.8 million, primarily due to consultant costsa full year of expenses related to analytics and strategies for the commercialization of Xtampza; products acquired from BDSI. |
Interest expense and Interest income Interest expense was $83.3 million for 2023, compared to $63.2 million for 2022. The $20.1 million increase was primarily due to the 2022 Term Loan that we entered into in connection with the BDSI Acquisition, higher interest rates impacting our variable rate term loan debt, and higher interest expense due to the issuance of the 2029 Convertible Notes (offset by the partial repurchase of the 2026 Convertible Notes). | ·
| | an increase in Post Marketing Requirement and PDUFA costs required for Xtampza of $7.5 million;
|
| ·
| | an increase in professional fees of $1.2 million primarily due to audit, insurance, accounting, recruiting and board of director fees;
|
| ·
| | an increase in distribution and commercial manufacturing costs of $1.0 million;
|
| ·
| | an increase in legal fees of $600,000 primarily due to costs related to litigation; and
|
| ·
| | an increase in amortization expense of $397,000 associated with the upfront fee for the Onsolis License Agreement.
|
Interest income was $15.6 million for 2023, compared to $1.0 million for 2022. The $14.6 million increase was primarily due to a higher overall balance invested in 2023 compared to 2022 and higher interest rates earned on cash equivalents and marketable securities. Loss on extinguishment of debt We recognized a $23.5 million loss on extinguishment of debt in 2023 in connection with the repurchase of a portion of our 2026 Convertible Notes. This transaction involved a contemporaneous exchange of cash between us and holders of the 2026 Convertible Notes participating in the issuance of the 2029 Convertible Notes. The repurchase of the 2026 Convertible Notes and issuance of the 2029 Convertible Notes were deemed to have substantially different terms based on the present value of the cash flows immediately prior to and after the exchange. Therefore, the repurchase of the 2026 Convertible Notes was accounted for as a debt extinguishment. Income Taxes The provision for income taxes was $27.6 million for 2023, compared to a $3.8 million benefit from income taxes for 2022. The tax provision for 2023 was impacted by non-deductible costs, including non-deductible costs associated with the debt extinguishment that occurred in the first quarter of 2023 as well as non-deductible costs related to stock-based compensation, including 162(m) limitations. In 2022, we recognized a deferred tax benefit, partially offset by state income tax expense. The provision for income taxes in 2022 primarily consisted of state income tax for states for which our state-level NOLs did not fully offset state-level taxable income. The effective tax rate was 36.4% and 13.3% for 2023 and 2022, respectively. Liquidity and Capital Resources Sources of liquidityLiquidity We have incurred net losses and negative cash flows from operations since inception. Since inception,
Historically, we have funded our operations primarily through the private placements and/or public offerings of our preferred stock, and convertible notes, public offerings of common stock, and convertible notes; commercial bank debt.debt; and cash inflows from sales of our products. We are primarily dependent on the commercial success of Belbuca, Xtampza, and the Nucynta Products. In March 2022, our debt balance increased significantly as we modified the 2020 Term Loan with Pharmakon to an increased principal balance of $650.0 million to fund a portion of the consideration paid to complete the BDSI Acquisition. We paid $100.0 million in principal payments during the first year of the 2022 Term Loan and the remaining $550.0 million balance will be paid in equal quarterly installments over the remaining three years of the term note. As of December 31, 2017,2023, the outstanding principal balance of the 2022 Term Loan was $412.5 million, of which $183.3 million in principal payments are due within the next 12 months. As of December 31, 2023, the outstanding principal balance of the Convertible Notes was $267.9 million, of which $26.4 million is due in 2026 and $241.5 million is due in 2029. As of December 31, 2023, and December 31, 2022, we had $118.7$238.9 million and $173.7 million in cash and cash equivalents.equivalents, respectively. Equity Financing
In January 2016, we issuedWe believe that our cash, cash equivalents, and sold in a public offering an aggregate of 2,750,000 shares of our common stock at $20.00 per share. We received proceeds from this public offering of approximately $51.2 million, after deduction of underwriting discounts and commissions and expenses payable by us.
In October 2016, we issued and sold in a public offering an aggregate of 5,750,000 shares of our common stock at $16.00 per share, including 750,000 shares of common stock upon the exercise by the underwriters of their option to purchase additional shares at the public offering price. We received net proceeds from this public offering of approximately $86.2 million, after deduction of underwriting discounts and commissions and estimated expenses payable by us.
In March 2017, we commenced an “at-the-market” offering of our common stock and entered into the ATM Sales Agreement with Cantor Fitzgerald,marketable securities as agent, pursuant to which we may issue and sell, from time to time, shares of our common stock having an aggregate offering price of up to $60.0 million. As of December 31, 2017,2023, together with expected cash inflows from operations, will enable us to fund our operating expenses, debt service and capital expenditure requirements under our current business plan for the foreseeable future.
Borrowing Arrangements and Equity Offerings The following transactions represent our material borrowing arrangements and equity offerings: the 2022 Term Loan, the 2026 Convertible Notes, and the 2029 Convertible Notes. Refer to Note 14, Debt, for more information. Cash flows In this section, we had sold an aggregate of 3,126,998 shares of common stock under the ATM Sales Agreement at an average gross sales price of $11.36 per share, generating net proceeds of $34.3 million after deduction of underwriting discounts and commissions and expenses payable by us, all of which were sold duringdiscuss cash flows for the year ended December 31, 2017. Silicon Valley Bank Term Loan Facility
Since August 2012, the Company has maintained a term loan facility with Silicon Valley Bank, which was amended and replaced in connection with, and as a condition2023 compared to consummation of the transactions contemplated by the Commercialization Agreement. Under the amended and replaced term loan, the Company now has a term loan facility in an amount of $11.5 million (the “New Term Loan”), which replaces the Company’s previously existing term loan facility. The proceeds of the New Term Loan were used by the Company to finance certain payment obligations under the Commercialization Agreement and to repay the balance of the previously existing term loan. The New Term Loan also provided SVB’s consent with respect to transactions contemplated by the Commercialization Agreement, including the delivery by SVB of a standby letter of credit in an aggregate amount of $33.75 million.
The New Term Loan bears interest at a rate per annum of 0.75% above the prime rate (as defined in the agreement governing the New Term Loan). The Company will repay the New Term Loan in equal consecutive monthly installments of principal plus monthly payments of accrued interest, commencing in July 2019, provided that, if the Company achieves EBITDA (as defined in the agreement governing the New Term Loan) in excess of $2.5 million for two (2) consecutive calendar quarters prior to June 2019, such payments will commence in January 2020. All outstanding principal and accrued and unpaid interest under the New Term Loan, and all other outstanding obligations with respect to the New Term Loan, are due and payable in full in December 2022. The Company may prepay the New Term Loan, in full but not in part, with a prepayment fee of (i) 3.0% of the outstanding principal balance prior to January 2019, (ii) 2.0% of the outstanding principal balance following January 2019 and prior to January 2020 and (iii) 1.0% of the outstanding principal balance following January 2020, plus, in each case, a final payment fee of $719.
Under the New Term Loan, the Company will be required to maintain a liquidity ratio of at least 2.0 to 1.0. Any amounts outstanding during the continuance of any event of default under the New Term Loan will bear additional interest at the per annum rate of 5.0%.
Cash flows
| | | | | | | | | | Years ended December 31, | | 2017 | | 2016 | | 2015 | Net cash used in operating activities | $ | (67,018) | | $ | (75,053) | | $ | (21,567) | Net cash used in investing activities | | (990) | | | (2,977) | | | (362) | Net cash provided by financing activities | | 33,480 | | | 135,558 | | | 115,992 |
Operating activities. Cash used in operating activities was $67.0 million in the year ended December 31, 2017 and $75.1 million in2022. For a discussion of the year ended December 31, 2016.2022 compared to the year ended December 31, 2021, refer to Part II, Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations" in our Annual Report on Form 10-K for the year ended December 31, 2022.
| | | | | | | Years Ended December 31, | | 2023 | | 2022 | | | (in thousands) | Net cash provided by operating activities | $ | 274,749 | | $ | 124,230 | Net cash used in investing activities | | (70,812) | | | (573,691) | Net cash (used in) provided by financing activities | | (140,178) | | | 436,723 | Net increase (decrease) in cash, cash equivalents and restricted cash | $ | 63,759 | | $ | (12,738) |
Operating activities. Cash provided by operating activities was $274.7 million in 2023, compared to $124.2 million in 2022. The $8.1$150.5 million decreaseincrease in cash used inprovided by operating activities was primarily due to the change in net loss, partially offset by changes in the working capital accounts, including significant changes in accounts receivable and accrued rebates, returns and discounts in the year ended December 31, 2017, and non-cash operating activities such as stock-based compensation expense, non-cash impairment charges and depreciation and amortization. We expect cash used in operating activities to increase in the foreseeable future as we continue to commercialize Xtampza and the Nucynta Products and fund research, development and clinical activities for additional product candidates. Cash used in operating activities was $75.1 million in the year ended December 31, 2016 and $21.6 million in the year ended December 31, 2015. The $53.5 million increase in cash used inflow from operating activities was due primarily to the changeresults, which reflects operating earnings, after adjustment for non-cash items that are included in net income, including higher intangible asset amortization as a result of the BDSI Acquisition and recognition of a loss partially offset byon extinguishment of debt in connection with the repurchase of a portion of our 2026 Convertible Notes, as well as changes in the working capital accounts.capital.
Investing activities. Cash used in investing activities was $990,000 in the year ended December 31, 2017 and $3.0$70.8 million in the year ended December 31, 2016.2023, compared to $573.7 million in 2022. The $502.9 million decrease in cash used in investing activities was primarily due to a one-time upfront fee paid to BDSIthe use of $572.1 million for the Onsolis License AgreementBDSI Acquisition in the year ended December 31, 2016.2022, net of cash acquired, partially offset by purchases and maturities of marketable securities in 2023. Financing activities.Cash used in investingfinancing activities was $3.0$140.2 million in the year ended December 31, 2016 and $362,000 in the year ended December 31, 2015. The increase in2023, compared to cash used in investing activities was primarily due to a one-time upfront fee paid to BDSI for the Onsolis License Agreement in the year ended December 31, 2016. Financing activities. Cash provided by financing activities of $436.7 million in 2022. The $576.9 million decrease was primarily due to:
| ● | the repayment of the outstanding balance of the 2020 Term Loan and establishment of the 2022 Term Loan in connection with the BDSI Acquisition, which was accounted for as a debt modification and resulted in $517.7 million in proceeds from the term note modification in 2022; |
| ● | an increase in repayments of term notes of $87.5 million; and |
| ● | an increase in repurchases of common stock of $60.9 million, which includes $75.0 million related to accelerated share repurchases executed in 2023; partially offset by |
| ● | the repurchase of a portion of our 2026 Convertibles Notes and issuance of our 2029 Convertible Notes which resulted in net proceeds of $96.6 million in 2023. |
Funding requirements We believe that our cash, cash equivalents, and marketable securities as of December 31, 2023, together with expected cash inflows from operations, will enable us to fund our operating expenses, debt service and capital expenditure requirements under our current business plan for the year ended December 31, 2017 primarily represents net proceeds of $34.3 million from the issuance of common stock, partially offset by the repayment of term notes of $2.7 million. Cash provided by financing activities for the year ended December 31, 2016 primarily represents net proceeds of $137.3 million from the issuance of common stock, partially offset by the repayment of term notes of $2.7 million.
Cash provided by financing activities for the year ended December 31, 2015 primarily represents net proceeds from the IPO and from the sale of Series D convertible preferred stock of $72.0 million and $44.8 million, respectively.
Funding requirements
Since 2011,foreseeable future. However, we have generated limited revenue from product sales and we continue to incur significant research, development and other expenses related to our ongoing operations. As we continue to commercialize Xtampza and add the Nucynta Products to our portfolio, we anticipate that we will continue to incur losses in the near future as we grow our commercial organization and continue the development of, and seek regulatory approvals for, other product candidates. We are subject to all of the risks common to the commercialization and development of new pharmaceutical products, and we may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may adversely affect our business.
We will also incur additional costs associated with operating as a commercial stage public company. We anticipate that we will need substantial additional funding in connection with our continuing operations. Until we can generate a sufficient amount of revenue from our pharmaceutical products, if ever, we expect to financehave significant future cash needs through public or private equity or debt offerings. Additional capital may not be available on reasonable terms, if at all. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue the development or commercialization of one or more of our products or product candidates. If we raise additional funds through the issuance of additional debt or equity securities, it could result in dilution to our existing shareholders, increased fixed payment obligations and the existence of securities with rights that may be senior to those of our common stock. If we incur indebtedness, we could become subject to covenants that would restrict our operations and potentially impair our competitiveness, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. Any of these events could significantly harm our business, financial condition and prospects.requirements, including:
| ● | expected operating expenses to manufacture and commercialize our products and to operate our organization; |
| ● | repayment of outstanding principal amounts and interest in connection with our 2022 Term Loan and Convertible Notes; |
| ● | royalties we pay on sales of certain products within our portfolio; |
| ● | operating lease obligations; |
| ● | minimum purchase obligations in connection with our contract manufacturer; and |
| ● | cash paid for income taxes. |
Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement and involves risks and uncertainties, and actual results could vary as a result of a number of factors. WeIn addition, we have based this estimate on assumptions that may prove to be wrong, and we could utilize our availablesignificant potential future capital resources sooner than we currently expect. The amount and timing of future funding requirements, both near- and long-term, will depend on many factors, including:
| ● | ·
| | the cost of growing and maintaining sales, marketing and distribution capabilities for Xtampza, the Nucynta Products and any other products for which we may receive regulatory approval;
enter into business development transactions, including acquisitions, collaborations, licensing arrangements and equity investments, that require additional capital; and |
| ● | ·
| | in January 2024, our Board of Directors authorized a new share repurchase program for the generationrepurchase of reasonable levelsup to $150.0 million of revenue from the saleshares of Xtampzaour common stock through June 30, 2025. Future share repurchases will depend upon, among other factors, our cash balances and the Nucynta Products; |
| ·
| | potential future capital requirements, our ability to pay license fees and royalties for our in-licensed products; |
| ·
| | the design, initiation, progress, size, timing, costs and results of preclinical studiesoperations and clinical trials forfinancial conditions, the price of our product candidates;
|
| ·
| | common stock on the outcome, timingNASDAQ Global Select Market, and cost of regulatory approvals by the FDA and comparable foreign regulatory authorities, including the potential for the FDA or comparable foreign regulatory authorities to requireother factors that we perform more studies than, or evaluate clinical endpoints other than those that we currently expect; may deem relevant. |
| ·
| | the timing and costs associated with manufacturing Xtampza and our product candidates for preclinical studies, clinical trials and, if approved, for commercial sale;
|
| ·
| | the number and characteristics of product candidates that we pursue;
|
| ·
| | the cost of patent infringement litigation, including our litigation with each of Purdue Pharma, L.P., or Purdue, and Teva Pharmaceuticals USA, Inc., or Teva, relating to Xtampza, the Nucynta Products or our product candidates, which may be expensive to defend and delay the commercialization of our product candidates;
|
| ·
| | our need to expand our research and development activities, including our need and ability to hire additional employees;
|
| ·
| | our need to implement additional infrastructure and internal systems and hire additional employees to operate as a public company;
|
| ·
| | our need to expand our regulatory and compliance functions; and
|
| ·
| | the effect of competing technological and market developments.
|
If we cannot expand our operations or otherwise capitalize on our business opportunities because we lack sufficient capital, our business, financial condition and results of operations could be materially adversely affected.
Contractual Obligations The following table summarizes ourOur contractual obligations as of December 31, 20172023 that will affect our future liquidity:
| | | | | | | | | | | | | | | | | | | | | | Less than | | | | | | | | More than | | | | Total | | 1 year | | 1 - 3 years | | 3 - 5 years | | 5 years | | | | (in thousands) | | Operating lease obligations (1) | | $ | 639 | | $ | 234 | | $ | 405 | | $ | — | | $ | — | | Long term debt (including interest) (2) | | | 1,479 | | | 1,479 | | | — | | | — | | | — | | Purchase obligations (3) | | | 9,000 | | | 3,000 | | | 6,000 | | | — | | | — | | Total | | $ | 11,118 | | $ | 4,713 | | $ | 6,405 | | $ | — | | $ | — | |
(1)Operatingliquidity include our term loan, including interest; convertible senior notes, including interest; operating lease obligations; and purchase obligations. For further detail regarding our term notes and convertible senior notes, refer to Note 14, Debt. For further detail regarding our operating lease obligations, represent future minimum lease payments under our non‑cancelable operating lease in effect as of December 31, 2017, reflecting remaining lease payments for our current facility in Canton, Massachusetts.refer to Note 15, Leases.
(2)Long‑term debt obligations represent future principal and interest payments under our Original Term Loan, as amended as of December 31, 2017.
(3)PurchaseOur purchase obligations represent the minimum purchase obligations of up to $3.0 million under a manufacturing agreementper year with our contract manufacturer which are in effect as of December 31, 2017. The disclosed amounts represent2023 and will remain in effect each year until the maximum amount that could be payable under the minimum purchase obligations.
We also have employment agreements with executive officers that would require us to make severance payments to them if we terminate their employment without cause or the executives resign for good cause. These payments are contingent upon the occurrence of various future events, and the amounts payable under these provisions depend upon the level of compensation at the time of termination of employment, and therefore, are therefore not calculable at this time, and, astime. Non-GAAP Financial Measures To supplement our financial results presented on a result,GAAP basis, we have not included any such amounts ininformation about certain non-GAAP financial measures. We believe the table above.presentation of these non-GAAP financial measures, when viewed with our results under GAAP and the accompanying reconciliations, provide analysts, investors, lenders, and other third parties with insights into how we evaluate normal operational activities, including our ability to generate cash from operations, on a comparable year-over-year basis and manage our budgeting and forecasting. In addition, certain non-GAAP financial measures, primarily Adjusted EBITDA, are used to measure performance when determining components of annual compensation for substantially all non-sales force employees, including senior management. Off‑Balance Sheet Arrangements
We didmay discuss the following financial measures that are not have duringcalculated in accordance with GAAP in our quarterly and annual reports, earnings press releases and conference calls. Adjusted EBITDA Adjusted EBITDA is a non-GAAP financial measure that represents GAAP net income or loss adjusted to exclude interest expense, interest income, the periods presented any off‑balance sheet arrangements,benefit from or provision for income taxes, depreciation, amortization, stock-based compensation, and other adjustments to reflect changes that occur in our business but do not represent ongoing operations. Adjusted EBITDA, as defined under SEC rules.used by us, may be calculated differently from, and therefore may not be comparable to, similarly titled measures used by other companies. There are several limitations related to the use of adjusted EBITDA rather than net income or loss, which is the nearest GAAP equivalent, such as: | ● | adjusted EBITDA excludes depreciation and amortization, and, although these are non-cash expenses, the assets being depreciated or amortized may have to be replaced in the future, the cash requirements for which are not reflected in adjusted EBITDA; |
| ● | we exclude stock-based compensation expense from adjusted EBITDA although: (i) it has been, and will continue to be for the foreseeable future, a significant recurring expense for our business and an important part of our compensation strategy; and (ii) if we did not pay out a portion of our compensation in the form of stock-based compensation, the cash salary expense included in operating expenses would be higher, which would affect our cash position; |
| ● | adjusted EBITDA does not reflect changes in, or cash requirements for, working capital needs; |
| ● | adjusted EBITDA does not reflect the benefit from or provision for income taxes or the cash requirements to pay taxes; |
| ● | adjusted EBITDA does not reflect historical cash expenditures or future requirements for capital expenditures or contractual commitments; |
| ● | we exclude impairment expenses from adjusted EBITDA and, although these are non-cash expenses, the asset(s) being impaired may have to be replaced in the future, the cash requirements for which are not reflected in adjusted EBITDA; |
| ● | we exclude restructuring expenses from adjusted EBITDA. Restructuring expenses primarily include employee severance and contract termination costs that are not related to acquisitions. The amount and/or frequency of these restructuring expenses are not part of our underlying business; |
| ● | we exclude litigation settlements from adjusted EBITDA, as well as any applicable income items or credit adjustments due to subsequent changes in estimates. This does not include our legal fees to defend claims, which are expensed as incurred; |
| ● | we exclude acquisition related expenses as the amount and/or frequency of these expenses are not part of our underlying business. Acquisition related expenses include transaction costs, which primarily consisted of financial advisory, banking, legal, and regulatory fees, and other consulting fees, incurred to complete the acquisition, employee-related expenses (severance cost and benefits) for terminated employees after the acquisition, and miscellaneous other acquisition related expenses incurred; |
| ● | we exclude recognition of the step-up basis in inventory from acquisitions (i.e., the adjustment to record inventory from historic cost to fair value at acquisition) as the adjustment does not reflect the ongoing expense associated with sale of our products as part of our underlying business; and |
| ● | we exclude losses on extinguishments of debt as these expenses are episodic in nature and do not directly correlate to the cost of operating our business on an ongoing basis. |
Adjusted EBITDA for the years ended December 31, 2023 and 2022 was as follows: | | | | | | | Years Ended December 31, | | 2023 | | 2022 | | | (in thousands) | GAAP net income (loss) | $ | 48,155 | | $ | (25,002) | Adjustments: | | | | | | Interest expense | | 83,339 | | | 63,213 | Interest income | | (15,615) | | | (1,047) | Loss on extinguishment of debt | | 23,504 | | | — | Provision for (benefit from) income taxes | | 27,578 | | | (3,845) | Depreciation | | 3,496 | | | 2,684 | Amortization | | 145,760 | | | 131,469 | Impairment expense | | — | | | 4,786 | Stock-based compensation | | 27,136 | | | 22,874 | Litigation settlements | | 8,500 | | | — | Acquisition related expenses | | — | | | 31,297 | Recognition of step-up basis in inventory | | 15,116 | | | 39,584 | Total adjustments | $ | 318,814 | | $ | 291,015 | Adjusted EBITDA | $ | 366,969 | | $ | 266,013 |
Adjusted EBITDA was $367.0 million for 2023 compared to $266.0 million for 2022. The $101.0 million increase was primarily due to higher revenues and gross profit before excluded costs, partially offset by higher adjusted operating expenses. The following is a summary of 2023 quarterly Adjusted EBITDA: | | | | | | | | | | | | | | | First Quarter | | Second Quarter | | Third Quarter | | Fourth Quarter | | | (in thousands) | GAAP net (loss) income | | $ | (17,426) | | $ | 13,007 | | $ | 20,634 | | $ | 31,940 | Adjustments: | | | | | | | | | | | | | Interest expense | | | 21,427 | | | 21,863 | | | 20,768 | | | 19,281 | Interest income | | | (2,747) | | | (4,027) | | | (4,538) | | | (4,303) | Loss on extinguishment of debt | | | 23,504 | | | — | | | — | | | — | (Benefit from) provision for income taxes | | | (131) | | | 4,790 | | | 8,149 | | | 14,770 | Depreciation | | | 817 | | | 895 | | | 835 | | | 949 | Amortization | | | 37,466 | | | 37,463 | | | 36,317 | | | 34,514 | Stock-based compensation | | | 6,035 | | | 7,072 | | | 7,027 | | | 7,002 | Litigation settlements | | | 8,500 | | | — | | | — | | | — | Recognition of step-up basis in inventory | | | 10,170 | | | 4,748 | | | 198 | | | — | Total adjustments | | $ | 105,041 | | $ | 72,804 | | $ | 68,756 | | $ | 72,213 | Adjusted EBITDA | | $ | 87,615 | | $ | 85,811 | | $ | 89,390 | | $ | 104,153 |
Adjusted Operating Expenses Adjusted operating expenses is a non-GAAP financial measure that represents GAAP operating expenses adjusted to exclude stock-based compensation expense, and other adjustments to reflect changes that occur in our business but do not represent ongoing operations. Adjusted operating expenses for the years ended December 31, 2023 and 2022 were as follows: | | | | | | | Years Ended December 31, | | 2023 | | 2022 | | | (in thousands) | GAAP operating expenses | $ | 159,208 | | $ | 176,169 | Adjustments: | | | | | | Stock-based compensation | | 27,136 | | | 22,874 | Litigation settlements | | 8,500 | | | — | Acquisition related expenses | | — | | | 31,297 | Total adjustments | $ | 35,636 | | $ | 54,171 | Adjusted operating expenses | $ | 123,572 | | $ | 121,998 |
Adjusted operating expenses were $123.6 million for 2023 compared to $122.0 million for 2022. The $1.6 million increase was primarily driven by: | ● | an increase in salaries, wages, and benefits (excluding stock-based compensation) of $1.8 million, primarily due to increases in personnel costs for employees retained following the BDSI Acquisition; |
| ● | an increase in sales and marketing expenses of $3.1 million, primarily due to expenses incurred to support the ongoing commercialization of products acquired from BDSI; |
| ● | an increase in regulatory expenses of $0.8 million, primarily due to a full year of expenses related to products acquired from BDSI; partially offset by |
| ● | a decrease in audit and legal expenses (excluding litigation settlements) of $3.5 million. |
The following is a summary of 2023 quarterly adjusted operating expenses: | | | | | | | | | | | | | First Quarter | | Second Quarter | | Third Quarter | | Fourth Quarter | | | (in thousands) | GAAP operating expenses | $ | 52,775 | | $ | 38,193 | | $ | 35,298 | | $ | 32,942 | Adjustments: | | | | | | | | | | | | Stock-based compensation | | 6,035 | | | 7,072 | | | 7,027 | | | 7,002 | Litigation settlements | | 8,500 | | | — | | | — | | | — | Total adjustments | | 14,535 | | | 7,072 | | | 7,027 | | | 7,002 | Adjusted operating expenses | $ | 38,240 | | $ | 31,121 | | $ | 28,271 | | $ | 25,940 |
Adjusted Net Income and Adjusted Earnings Per Share Adjusted net income is a non-GAAP financial measure that represents GAAP net income or loss adjusted to exclude significant income and expense items that are non-cash or not indicative of ongoing operations, including consideration of the tax effect of the adjustments. Adjusted earnings per share is a non-GAAP financial measure that represents adjusted net income per share. Adjusted weighted-average shares - diluted is calculated in accordance with the treasury stock, if-converted, or contingently issuable accounting methods, depending on the nature of the security. Adjusted net income and adjusted earnings per share for the years ended December 31, 2023 and 2022 were as follows: | | | | | | | Years Ended December 31, | | 2023 | | 2022 | | | (in thousands, except share and per share data) | GAAP net income (loss) | $ | 48,155 | | $ | (25,002) | Adjustments: | | | | | | Non-cash interest expense | | 8,635 | | | 8,285 | Loss on extinguishment of debt | | 23,504 | | | — | Amortization | | 145,760 | | | 131,469 | Impairment expense | | — | | | 4,786 | Stock-based compensation | | 27,136 | | | 22,874 | Litigation settlements | | 8,500 | | | — | Acquisition related expenses | | — | | | 31,297 | Recognition of step-up basis in inventory | | 15,116 | | | 39,584 | Income tax effect of above adjustments (1) | | (53,526) | | | (60,553) | Total adjustments | $ | 175,125 | | $ | 177,742 | Non-GAAP adjusted net income | $ | 223,280 | | $ | 152,740 | | | | | | | Adjusted weighted-average shares — diluted (2) | | 41,788,125 | | | 39,531,814 | Adjusted earnings per share (2) | $ | 5.47 | | $ | 3.96 |
| (1) | The income tax effect of the adjustments was calculated by applying our blended federal and state statutory rate to the adjustments that have a tax effect. The blended federal and state statutory rate for the years ended December 31, 2023 and 2022 were 25.9% and 26.0%, respectively. As such, the non-GAAP effective tax rates for the years ended December 31, 2023 and 2022 were 23.4% and 25.4%, respectively. |
| (2) | Adjusted weighted-average shares - diluted were calculated using the “if-converted” method for the convertibles notes in accordance with ASC 260, Earnings per Share. As such, adjusted weighted-average shares – diluted includes shares related to the assumed conversion of our convertible notes and the associated cash interest expense added-back to non-GAAP adjusted net income. For the years ended December 31, 2023 and 2022, adjusted weighted-average shares – diluted includes 6,793,421 and 4,925,134 shares, respectively, attributable to our convertible notes. In addition, adjusted earnings per share includes other potentially dilutive securities to the extent that they are not antidilutive. |
The following is a summary of 2023 quarterly adjusted net income and adjusted earnings per share: | | | | | | | | | | | | | First Quarter | | Second Quarter | | Third Quarter | | Fourth Quarter | | | (in thousands, except share and per share data) | GAAP net (loss) income | $ | (17,426) | | $ | 13,007 | | $ | 20,634 | | $ | 31,940 | Adjustments: | | | | | | | | | | | | Non-cash interest expense | | 2,287 | | | 2,261 | | | 2,124 | | | 1,963 | Loss on extinguishment of debt | | 23,504 | | | — | | | — | | | — | Amortization | | 37,466 | | | 37,463 | | | 36,317 | | | 34,514 | Stock-based compensation | | 6,035 | | | 7,072 | | | 7,027 | | | 7,002 | Litigation settlements | | 8,500 | | | — | | | — | | | — | Recognition of step-up basis in inventory | | 10,170 | | | 4,748 | | | 198 | | | — | Income tax effect of above adjustments (1) | | (18,874) | | | (12,100) | | | (11,300) | | | (11,252) | Total adjustments | $ | 69,088 | | $ | 39,444 | | $ | 34,366 | | $ | 32,227 | Non-GAAP adjusted net income | $ | 51,662 | | $ | 52,451 | | $ | 55,000 | | $ | 64,167 | | | | | | | | | | | | | Adjusted weighted-average shares — diluted (2) | | 40,196,015 | | | 42,849,952 | | | 42,058,820 | | | 41,279,982 | Adjusted earnings per share (2) | $ | 1.32 | | $ | 1.26 | | $ | 1.34 | | $ | 1.58 |
| (1) | The income tax effect of the adjustments was calculated by applying our blended federal and state statutory rate to the adjustments that have a tax effect. The blended federal and state statutory rate for the three months ended March 31, June 30, September 30, and December 31, 2023 were 26.8%, 24.0%, 25.6%, and 25.9%, respectively. As such, the non-GAAP effective tax rates for the three months ended March 31, June 30, September 30, and December 31, 2023 were 21.5%, 23.5%, 24.7%, and 25.9%, respectively. |
| (2) | Adjusted weighted-average shares - diluted were calculated using the “if-converted” method for the convertibles notes in accordance with ASC 260, Earnings per Share. As such, adjusted weighted-average shares – diluted includes shares related to the assumed conversion of our convertible notes and the associated cash interest expense added-back to non-GAAP adjusted net income. For the three months ended March 31, June 30, September 30, and December 31, 2023, adjusted weighted-average shares – diluted includes 4,646,372, 7,509,104, 7,509,104, and 7,509,104 shares, respectively, attributable to our convertible notes. In addition, adjusted earnings per share includes other potentially dilutive securities to the extent that they are not antidilutive. |
Item 7A. Quantitative and Qualitative Disclosures about Market Risks We are exposed to market risk related to changes in interest rates. As of December 31, 2017, we had cash and cash equivalents consisting of cash and money market funds of $118.7 million. Our primary exposure to market risk is interest rate sensitivity in connection with our investment portfolio and the 2022 Term Loan. None of these market risk sensitive instruments are held for trading purposes.
Investment Portfolio Our investment portfolio includes financial instruments that are sensitive to interest rate risks. Our investment portfolio is used to preserve capital, maintain liquidity sufficient to meet cash flow requirements, and maximize returns commensurate with our risk appetite. We invest in instruments that meet the credit quality, diversification, liquidity, and maturity standards outlined in our investment policy. As of December 31, 2023, our investment portfolio includes $82.0 million of cash equivalents and $71.6 million of marketable securities, which is affected by changes in the general levelare primarily comprised of money market funds, U.S. interest rates, particularly because ourTreasury securities, corporate debt, and government-sponsored securities. Our money market funds are short-term highly liquid investments, and our marketable securities have active secondary or resale markets to help ensure liquidity. We account for marketable securities as available-for-sale, thus, no gains or losses are realized due to changes in the fair value of our marketable securities unless we sell our investments prior to maturity or incur a credit loss. Furthermore, our investment policy includes guidelines limiting the term-to-maturity of our investments. Due to the short-term duration and the low risk profilenature of our investments, an immediate 10% changewe do not believe that the fair value of our investments has a material exposure to interest rate risk. 2022 Term Loan The 2022 Term Loan bears interest at a rate based upon the Secured Overnight Financing Rate (“SOFR”) plus a spread adjustment of 0.26% (subject to a floor of 1.20%), plus a margin of 7.5% per annum. Based on the outstanding principal amount of the 2022 Term Loan as of December 31, 2023 of $412.5 million and the applicable interest rate, a hypothetical 1% increase or decrease in interest rates would not have a material effect on the fair market value of our portfolio.increase or decrease future interest expense by approximately $4.1 million. Item 8. Consolidated Financial Statements and Supplementary Data
Our consolidated financial statements,Consolidated Financial Statements, together with the reports of our independent registered public accounting firms, begin on page F‑-1 of this Form 10‑-K. Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None. Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures The Company'sOur management, with the participation of the Chief Executive Officer and the Chief Financial Officer, has evaluated the effectiveness of the Company'sour disclosure controls and and procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, as of the end of the period covered by this report. The term “disclosure controls and procedures” as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, means controls and other procedures of a company that are designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective as of December 31, 2017.2023.
Management’s Report on Internal Control Over Financial Reporting Internal control over financial reporting refers to the process designed by, or under the supervision of, our Chief Executive Officer and Chief Financial Officer, and effected by our boardBoard of directors,Directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of consolidated financial statements for external purposes in accordance with generally accepted accounting principles, and includes those policies and procedures that: (1) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of consolidated financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’sour assets that could have a material effect on the consolidated financial statements. Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitations. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgment and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management override. Also, projections of any evaluation of effectiveness of internal control over financial reporting to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with policies or procedures may deteriorate. Because of such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk. Management is responsible for establishing and maintaining adequate internal control over our financial reporting, as such term is defined in Rules 13a 15(f) and 15d 15(f) under the Exchange Act. Under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting. Management has used the framework set forth in the report entitled “Internal Control—Integrated Framework (2013)” published by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) to evaluate the effectiveness of our internal control over financial reporting. Based on itsour evaluation, managementManagement has concluded that our internal control over financial reporting was effective at the reasonable assurance level as of December 31, 2017, the end of our most recent fiscal year.2023. Attestation Report of the Registered Public Accounting Firm
This Form 10-K does not include an attestation report of our registered public accounting firm due to an exemption established by the JOBS Act for “emerging growth companies.”
Changes in Internal Control Over Financial Reporting There has been noAs required by Rule 13a-15(d) of the Exchange Act, our management, including our Chief Executive Officer and our Chief Financial Officer, conducted an evaluation of the internal control over financial reporting to determine whether any changes occurred during the quarter ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting. Based on that evaluation, our Chief Executive Officer and Chief Financial Officer did not identify any change in our internal control over financial reporting during the fiscal quarter ended December 31, 20172023 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM To the shareholders and the Board of Directors of Collegium Pharmaceutical, Inc. Opinion on Internal Control over Financial Reporting We have audited the internal control over financial reporting of Collegium Pharmaceutical, Inc. and subsidiaries (the “Company”) as of December 31, 2023, based on criteria established in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). In our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control — Integrated Framework (2013) issued by COSO. We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated financial statements as of and for the year ended December 31, 2023, of the Company and our report dated February 22, 2024, expressed an unqualified opinion on those financial statements. Basis for Opinion The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB. We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion. Definition and Limitations of Internal Control over Financial Reporting A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. /s/ Deloitte & Touche LLP Boston, Massachusetts February 22, 2024 Item 9B. Other Information Insider Trading Arrangements During the three months ended December 31, 2023, none of our directors or officers adopted, amended, or terminated a “Rule 10b5-1 trading arrangement” or “non-Rule 10b5-1 trading arrangement,” as each term is defined in Item 408(a) of Regulation S-K. Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections Not applicable. PART IIIIII Item 10. Directors,Directors, Executive Officers, and Corporate Governance Other than the information regarding our executive officers provided in Part I of this report under the heading “Business—“Business — OurExecutive Officers of the Registrant,,” the information required to be furnished pursuant to this item is incorporated herein by reference to our definitive proxy statement for the 20182024 Annual Meeting of the Shareholders. Our boardBoard of directorsDirectors has adopted a Code of Ethics applicable to all of our employees, executive officers and directors. The Code of Ethics is available on our website at www.collegiumpharma.com. Our boardBoard of directorsDirectors is responsible for overseeing compliance with the Code of Ethics, and our boardBoard of directorsDirectors or an appropriate committee thereof must approve any waivers of the Code of Ethics for employees, executive officers or directors. Disclosure regarding any amendments to the Code of Ethics, or any waivers of its requirements, will be made on our website. Insider Trading Policies Our Board of Directors has adopted an Insider Trading Policy which governs the purchase, sales, and/or other dispositions of our securities by directors, officers, and employees. Our Insider Trading Policy is attached hereto as Exhibit 19 and incorporated herein. Item 11. Executive Compensation The information required by this Item 11 is incorporated herein by reference from our definitive proxy statement for the 20182024 Annual Meeting of Shareholders.Shareholders under the captions “Compensation Discussion and Analysis,” “Executive Compensation” (excluding the information under the heading “Pay Versus Performance”), “Director Compensation” and “Compensation Committee Report.” Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this Item 12 is incorporated herein by reference from our definitive proxy statement for the 20182024 Annual Meeting of Shareholders.Shareholders under the captions “Compensation Discussion and Analysis,” “Executive Compensation” (excluding the information under the heading “Pay Versus Performance”), “Director Compensation” and “Compensation Committee Report.” Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this Item 13 is incorporated herein by reference from our definitive proxy statement for the 20182024 Annual Meeting of Shareholders.Shareholders under the captions “Compensation Discussion and Analysis,” “Executive Compensation” (excluding the information under the heading “Pay Versus Performance”), “Director Compensation” and “Compensation Committee Report.” Item 14. PrincipalPrincipal Accountant Fees and Services
The information required by this Item 14 is incorporated herein by reference from our definitive proxy statement for the 20182024 Annual Meeting of Shareholders.Shareholders under the captions “Compensation Discussion and Analysis,” “Executive Compensation” (excluding the information under the heading “Pay Versus Performance”), “Director Compensation” and “Compensation Committee Report.” PART IV
PART IV Item 15. Exhibits and Financial Statement Schedules
Consolidated Financial Statements SeeRefer to Part II, Item 8 for the Consolidated Financial Statements required to be included in this Form 10-K.
Consolidated Financial Statement Schedules All financial statement schedules are omitted because they are not applicable or the required information is included in the consolidated financial statementsConsolidated Financial Statements or notes thereto. Exhibits Exhibits
| | | | Exhibit Number | | Exhibit Description | | 2.1 | | Exhibit Number
| Exhibit Description
| 2.1††
| Agreement and Plan of Merger, dated July 10, 2014,as of February 14, 2022, by and betweenamong Collegium Pharmaceutical, Inc., a Delaware corporation,Bristol Acquisition Company, Inc. and Collegium Pharmaceutical,BioDelivery Sciences International, Inc., a Virginia corporation. | (1) | 3.1†3.1
| † | SecondThird Amended and Restated Articles of Incorporation of Collegium Pharmaceutical, Inc.
| (2) | 3.2†3.2
| † | Amended and Restated Bylaws of Collegium Pharmaceutical, Inc.(8) | (3) | 4.1†4.1
| † | Eighth Amended and Restated Investor Rights Agreement,Indenture, dated March 6, 2015, by and among Collegium Pharmaceutical, Inc. and certainas of its shareholders.(1)
| 4.2†
| Warrant to Purchase Stock, dated October 28, 2010, issued by Collegium Pharmaceutical, Inc. to Comerica Bank.(1)
| 10.1†
| Office Lease Agreement, dated August 28, 2012, by and between 780 Dedham Street Holdings, LLC and Collegium Pharmaceutical, Inc.(1)
| 10.2†
| Loan and Security Agreement, dated August 28, 2012, by and between Silicon Valley Bank and Collegium Pharmaceutical, Inc.(1)
| 10.3†
| First Amendment to Loan and Security Agreement, dated January 31, 2014, by and between Silicon Valley Bank and Collegium Pharmaceutical, Inc.(1)
| 10.4†
| Assumption and Second Amendment to Loan and Security Agreement, dated August 12, 2014, by and between Silicon Valley Bank and Collegium Pharmaceutical, Inc.(1)
| 10.5†
| Third Amendment to Loan and Security Agreement, dated September 25, 2014, by and between Silicon Valley Bank and Collegium Pharmaceutical, Inc.(1)
| 10.6†
| Fourth Amendment to Loan and Security Agreement, dated October 31, 2014, by and between Silicon Valley Bank and Collegium Pharmaceutical, Inc.(1)
| 10.7†
| Consent and Sixth Amendment to Loan and Security Agreement, dated January 9, 2018, by andFebruary 13, 2020, between Collegium Pharmaceutical, Inc. and Silicon Valley Bank.(9)The Bank of New York Mellon Trust Company, N.A., as trustee.
| (4) | 10.8†4.2
| Subordination Agreement, dated November 14, 2014, by and among Collegium Pharmaceutical, Inc., Silicon Valley Bank and the creditors named therein.(1)
| 10.9†
| Subordination Agreement, dated December 2, 2014, by and among Collegium Pharmaceutical, Inc., Silicon Valley Bank and the creditors named therein.(1)
| 10.10+†
| Restricted Stock Award Agreement,First Supplemental Indenture, dated Juneas of February 13, 2012, by and2020, between Collegium Pharmaceutical, Inc. and Michael T. Heffernan.The Bank of New York Mellon Trust Company, N.A., as trustee.(1)
| (4) | 10.11+4.3
| † | Restricted Stock Award Agreement,Form of certificate representing the 2.625% Convertible Senior Notes due 2026 (included as Exhibit A to Exhibit 4.2).
| (4) | 4.4 | † | Indenture, dated July 18, 2012, by andas of February 10, 2023, between Collegium Pharmaceutical, Inc. and Gino Santini.The Bank of New York Mellon Trust Company, N.A., as trustee.(1) | (5) | 10.12+4.5
| † | Restricted Stock Award Agreement, dated March 5, 2014,Form of certificate representing the 2.875% Convertible Senior Notes due 2029 (included as Exhibit A to Exhibit 4.4).
| (5) | 4.6 | † | Description of the Registrant’s Securities Registered Pursuant to Section 12 of the Securities Exchange Act of 1934. | (6) | 10.1 | † | Office Lease agreement by and between Campanelli-Trigate 100 TCD Stoughton, LLC, and Collegium Pharmaceutical, Inc. and Gino Santini.Inc as of March 23, 2018.(1) | (7) | 10.13†10.2
| Form of Confidentiality and Inventions Agreement.(1)
|
10.14++†
| Offer Letter, dated January 29, 2015, by and between Collegium Pharmaceutical, Inc. and Garen Bohlin.(1)
| 10.15†
| Series D Convertible Preferred Stock Purchase Agreement, dated March 6, 2015, by and among Collegium Pharmaceutical, Inc. and the purchasers thereto.(1)
| 10.16†
| First Amendment to Lease, dated March 24, 2015, by and between Park at 95, LLC (as successor in interest to 780 Dedham Street Holdings, LLC) and Collegium Pharmaceutical, Inc.(1)
| 10.17+†
| 2015 Employee Stock Purchase Plan.(3) | (8) | 10.18+†10.3
| Performance Bonus Plan. (4)
| 10.19(a)+†
| Performance Bonus Plan. | (9) | 10.4(a) | +† | Amended and Restated 2014 Stock Incentive Plan. (3) | (8) | 10.19(b)10.4(b)
| +† | Form of Incentive Stock Option Agreement under the Amended and Restated 2014 Stock Incentive Plan. (3) | (8) | 10.19(c)10.4(c)
| +† | Form of Non-Qualified Stock Option Agreement under the Amended and Restated 2014 Stock Incentive Plan.(3) | (8) | 10.19(d)10.4(d)
| +† | Form of Restricted Stock AwardUnit Agreement under the Amended and Restated 2014 Stock Incentive Plan. (3) | | 10.20+10.4(e)
| +† | RestrictedForm of Performance Share Unit Agreement under the Amended and Restated 2014 Stock Award Agreement, dated April 2, 2015, by and between Collegium Pharmaceutical, Inc. and Michael T. Heffernan.Incentive Plan. (4)
| (10) | 10.21†10.5
| † | Form of Indemnification Agreement. (4) | (9) | 10.22+10.6
| +† | Amended & Restated Employment Agreement, dated August 4, 2015, by and between Michael Heffernan and Collegium Pharmaceutical, Inc.(5) | 10.23+†
| Employment Agreement, dated August 4, 2015, by and between Paul Brannelly and Collegium Pharmaceutical, Inc.(5)
| 10.24+†
| Employment Agreement, dated August 4, 2015, by and between Barry S. Duke and Collegium Pharmaceutical, Inc.(6)
| 10.25*†
| License and Development Agreement, dated as of May 11, 2016, by and between Collegium Pharmaceutical, Inc. and BioDelivery Systems International, Inc.(7)
| 10.26+†
| Employment Agreement, dated May 31, 2017,December 27, 2020, by and between Collegium Pharmaceutical, Inc. and Joseph Ciaffoni.(10)
| (11) |
10.7 | +† | Amended & Restated Employment Agreement, dated December 27, 2020, by and between Shirley Kuhlmann and Collegium Pharmaceutical, Inc. | (11) | 10.27*10.8
| +† | CommercializationAmended & Restated Employment Agreement, dated December 27, 2020, by and among, Depomed, Inc.,between Collegium Pharmaceutical, Inc. and Collegium NF, LLC,Scott Dreyer.
| (11) | 10.9 | † | License Agreement (U.S.), dated as of December 4, 2017. | 10.28 | Amendment dated January 9, 2018 to Commercialization Agreement13, 2015, by and among Depomed,Grünenthal GmbH, Janssen Research & Development, LLC, Assertio Therapeutics, Inc. and Collegium Pharmaceutical, Inc.
| (12) | 10.10* | † | Consent Agreement, dated January 30, 2020, by and among Grünenthal GmbH, Assertio Therapeutics, Inc. and Collegium NF, LLCPharmaceutical, Inc. | (12) | 10.11* | † | Settlement Agreement, dated September 29, 2020, by and among Collegium Pharmaceutical, Inc. and Teva Pharmaceuticals USA, Inc. | (13) | 10.12 | +† | Employment Agreement, dated May 24, 2021, by and between Colleen Tupper and Collegium Pharmaceutical, Inc. | (14) | 10.13* | † | Amended and Restated Loan Agreement, dated as of March 22, 2022, by and among Collegium Pharmaceutical, Inc., the guarantors party thereto, BioPharma Credit PLC, and BioPharma Credit Investments V (Master) LP, as lenders. | (15) | 10.14 | | First Amendment and Consent to Amended and Restated Loan Agreement, dated as of January 3, 2023, by and among Collegium Pharmaceutical, Inc., the guarantors party thereto, BioPharma Credit PLC, and BioPharma Credit Investments V (Master) LP, as lenders. | | 10.15 | † | Second Amendment to Loan Agreement, dated as of February 6, 2023, Amended and Restated Loan Agreement, dated as of March 22, 2022, by and among Collegium Pharmaceutical, Inc., the guarantors party thereto, BioPharma Credit PLC, and BioPharma Credit Investments V (Master) LP, as lenders. | (16) | 10.16 | † | Third Amendment to Loan Agreement, dated as of June 23, 2023, by and among Collegium Pharmaceutical, Inc., the guarantors party thereto, BioPharma Credit PLC, and Bio Pharma Credit Investments V (Master) LP, as lenders. | (17) | 10.17* | † | Exclusive License Agreement, dated April 4, 2019, between the Company and Shionogi, Inc. (incorporated by reference to Exhibit 10.19 to the Annual Report on Form 10-K filed by BDSI on March 9, 2022). | (18) | 10.18 | +† | Amendment to Employment Agreement, dated January 20, 2022 by and between Collegium Pharmaceutical, Inc. and Joseph Ciaffoni. | (18) | 10.19 | +† | Amendment to Employment Agreement, dated January 20, 2022 by and between Collegium Pharmaceutical, Inc. and Colleen Tupper. | (18) | 10.20 | +† | Amendment to Employment Agreement, dated January 20, 2022 by and between Collegium Pharmaceutical, Inc. and Shirley Kuhlmann. | (18) | 10.21 | +† | Amendment to Employment Agreement, dated January 20, 2022 by and between Collegium Pharmaceutical, Inc. and Scott Dreyer. | (18) | 10.22 | +† | Employment Agreement, dated March 23, 2022 by and between Collegium Pharmaceutical, Inc. and Thomas Smith. | (18) | 19 | | Insider Trading Policy | | 21.1 | | Subsidiaries of Collegium Pharmaceutical, Inc. | | 23.1 | | Consent of Deloitte & Touche LLP, Independent Registered Public Accounting Firm. | 23.2
| Consent of Grant Thornton LLP, Independent Registered Public Accounting Firm.
| 31.1 | | Certifying Statement of the Chief Executive Officer pursuant to Section 302 of the Sarbanes‑Sarbanes‑Oxley Act of 2002. | | 31.2 | | Certifying Statement of the Chief Financial Officer pursuant to Section 302 of the Sarbanes‑Sarbanes‑Oxley Act of 2002. | | 32.1 | | Certifying Statement of the Chief Executive Officer pursuant to Section 1350 of Title 18 of the United States Code. | | 32.2 | | Certifying Statement of the Chief Financial Officer pursuant to Section 1350 of Title 18 of the United States Code. | | 97 | | Compensation Recovery Policy | | 101 | | The following financial information from this Annual Report on Form 10-K for the year ended December 31, 2017,2023, formatted in Inline XBRL: (i) Consolidated Balance Sheets as of December 31, 2017, 2016,2023, and 2022, (ii) Consolidated Statements of Operations for the years ended December 31, 2017, 20162023, 2022 and 2015,2021, (iii) Consolidated Statements of Convertible Redeemable Preferred Stock and Shareholders' Equity (Deficit) for the Years Ended December 31, 2017, 20162023, 2022 and 2015,2021, (iv) Consolidated Statements of Cash Flows for the years ended December 31, 2017, 20162023, 2022 and 2015,2021, and (v) Notes to Consolidated Financial Statements, tagged as blocks of text. | |
104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | |
†Previously filed. +Indicates management contract or compensatory plan. * Certain portions of the exhibits that are not material and would be competitively harmful if publicly disclosed have been redacted pursuant to Item 601(b)(10)(iv) of Regulation S-K. Copies of the unredacted exhibits will be furnished to the Securities and Exchange Commission (“SEC”) upon request.
| (1) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on February 14, 2022. |
| (2) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2020 filed with the SEC on August 5, 2020. |
| (3) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on December 4, 2017. |
| (4) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on February 13, 2020. |
| (5) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on February 13, 2023. |
| (6) | Previously filed as an exhibit to the registrant’s Annual Report on Form 10-K for the year ended December 31, 2022 filed with the SEC on February 23, 2023. |
| (7) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2018 filed with the SEC on May 9, 2018. |
| (8) | Previously filed as an exhibit to the registrant’s Registration Statement on Form S-8 (File No. 333-207744) filed with the SEC on November 2, 2015. |
| (9) | Previously filed as an exhibit to the registrant’s Registration Statement on Form S-1/A (File No. 333-203208) filed with the SEC on April 27, 2015. |
| (10) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2019 filed with the SEC on May 8, 2019. |
| (11) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on December 30, 2020. |
| (12) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2020 filed with the SEC on May 7, 2020. |
| (13) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on September 30, 2020. |
| (14) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2021 filed with the SEC August 5, 2021. |
| (15) | Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the SEC on March 23, 2022. |
| (16) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2023 filed with the SEC May 4, 2023. |
* Subject to confidential treatment request.
(1)Previously filed as an exhibit to the registrant’s Registration Statement on Form S-1 (File No. 333-203208) filed with the Commission on April 2, 2015.
(2)Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Commission on May 12, 2015.
(3)Previously filed as an exhibit to the registrant’s Registration Statement on Form S-8 (File No. 333-207744) filed with the Commission on November 2, 2015.
(4)Previously filed as an exhibit to the registrant’s Registration Statement on Form S-1/A (File No. 333-203208) filed with the Commission on April 27, 2015.
(5)Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Commission on August 10, 2015.
(6)Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2015 filed with the Commission on August 12, 2015.
(7)Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2016 filed with the Commission on August 11, 2016.
(8)Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Commission on December 1, 2017.
(9)Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Commission on January 10, 2018.
(10)Previously filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Commission on May 31, 2017.
| (17) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2023 filed with the SEC August 3, 2023. |
| (18) | Previously filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2022 filed with the SEC May 10, 2022. |
Item 16. Form 10-K Summary None.
TableSIGNATURES Pursuant to the requirements of ContentsSection 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized. | | | COLLEGIUM PHARMACEUTICAL, INC. | | | | By: /s/ Joseph Ciaffoni | | Joseph Ciaffoni | | Chief Executive Officer |
| | | | | Signature | | Title | | Date | | | | | | /s/ Joseph Ciaffoni | | President and Chief Executive Officer (Principal Executive Officer) and Director | | February 22, 2024 | Joseph Ciaffoni | | | | | | | | | | | | | | | /s/ Colleen Tupper | | Executive Vice President and Chief Financial Officer (Principal Financial and Accounting Officer) | | February 22, 2024 | Colleen Tupper | | | | | | | | | | /s/ Michael T. Heffernan, R.Ph. | | Chairman of the Board | | February 22, 2024 | Michael T. Heffernan, R.Ph. | | | | | | | | | | /s/ Rita Balice-Gordon, Ph.D. | | Director | | February 22, 2024 | Rita Balice-Gordon, Ph.D. | | | | | | | | | | /s/ Garen G. Bohlin | | Director | | February 22, 2024 | Garen G. Bohlin | | | | | | | | | | /s/ John A. Fallon, M.D. | | Director | | February 22, 2024 | John A. Fallon, M.D. | | | | | | | | | | /s/ John G. Freund, M.D. | | Director | | February 22, 2024 | John G. Freund, M.D. | | | | | | | | | | /s/ Gwen Melincoff | | Director | | February 22, 2024 | Gwen Melincoff | | | | | | | | | | /s/ Gino Santini | | Director | | February 22, 2024 | Gino Santini | | | | | | | | | | /s/ Neil McFarlane | | Director | | February 22, 2024 | Neil McFarlane | | | | |
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this report has been signed by the following persons in the capacities and on the dates indicated. COLLEGIUM PHARMACEUTICAL, INC. Index to Consolidated Financial Statements | | | Audited Consolidated Financial Statements |
| | | | | Audited Financial Statements
| | Pages | ReportsReport of Independent Registered Public Accounting FirmsFirm (PCAOB ID 34)
| | F-2 | | | | Consolidated Balance Sheets as of December 31, 20172023 and 20162022 | | F-4 | | | | Consolidated Statements of Operations for the Years Ended December 31, 2017, 2016,2023, 2022, and 20152021 | | F-5 | | | | Consolidated Statements of Convertible Redeemable Preferred Stock and Shareholders' Equity (Deficit) for the Years Ended December 31, 2017, 20162023, 2022, and 20152021 | | F-6F-7
| | | | Consolidated Statements of Cash Flows for the Years Ended December 31, 2017, 20162023, 2022, and 20152021 | | F-7
| Notes to Consolidated Financial Statements
| | F-8 |
| | | Notes to Consolidated Financial Statements | | F-9 | |