• | The successful commercialization of our product candidates will depend in part on the extent to which third-party payors, including governmental authorities and private health insurers, provide coverage and adequate reimbursement levels, as well as implement pricing policies favorable for our product candidates. Failure to obtain or maintain coverage and adequate reimbursement for our product candidates, if approved, could limit
2
3
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS This Annual Report on Form 10-K In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “would,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” or “continue” or the negative of these terms or other similar expressions. Forward-looking statements contained in this report include, but are not limited to, statements about:
4
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
• | our plans relating to the further development and manufacturing of our product candidates, including additional indications that we may pursue; |
our plans relating to commercializing our product candidates, if approved, including the geographic areas of focus and sales strategy;
• | existing regulations and regulatory developments in the United States and other jurisdictions; |
the expected potential benefits of strategic collaborations with third parties and our ability to attract collaborators with development,
• | our continued reliance on third parties to conduct additional clinical trials of our product candidates, and for the manufacture of our product candidates for preclinical studies and clinical trials; |
regulatory and commercialization expertise;
• | our plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and the outcome of our ongoing arbitration proceedings; |
our estimates of the number of patients in the United States who suffer from the diseases we are targeting and the number of patients that will enroll in our clinical trials;
• | the need to hire additional personnel and our ability to attract and retain such personnel; |
the size of the market opportunity for our product candidates in each of the diseases we are targeting;
• | the accuracy of our estimates regarding expenses, future revenue, capital requirements, and needs for additional financing; |
our ability to expand our product candidates into additional indications and patient populations;
• | our financial performance; |
the success of competing therapies that are or may become available;
• | the sufficiency of our existing cash, cash equivalents and marketable securities to fund our future operating expenses and capital expenditure requirements; and |
the beneficial characteristics, safety, efficacy, and therapeutic effects of our product candidates;
• | our expectations regarding the period during which we will qualify as an emerging growth company under the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”). |
the timing or likelihood of regulatory filings and approvals, including our expectation to seek special designations, such as orphan drug designation, for our product candidates for various diseases;
our ability to obtain and maintain regulatory approval of our product candidates;
our plans relating to the further development and manufacturing of our product candidates, including additional indications that we may pursue;
existing regulations and regulatory developments in the United States and other jurisdictions;
our continued reliance on third parties to conduct additional clinical trials of our product candidates, and for the manufacture of our product candidates for preclinical studies and clinical trials;
our plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and the outcome of our ongoing arbitration proceedings;
the need to hire additional personnel and our ability to attract and retain such personnel;
the accuracy of our estimates regarding expenses, future revenue, capital requirements, and needs for additional financing;
our financial performance;
the sufficiency of our existing cash and cash equivalents to fund our future operating expenses and capital expenditure requirements; and
our expectations regarding the period during which we will qualify as an emerging growth company under the Jumpstart Our Business Startups Act of 2012 (JOBS Act).
We have based these forward-looking statements largely on our current expectations and projections about our business, the industry in which we operate and financial trends that we believe may affect our business, financial condition, results of operations, and prospects, and these forward-looking statements are not guarantees of future performance or development. These forward-looking statements speak only as of the date of this report and are subject to a number of risks, uncertainties, and assumptions described in the section titled “Risk Factors” and elsewhere in this report. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified, you should
4
not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. All forward-looking statements are made as of the date of this report and we do not undertake any obligation to update our forward-looking statements, except as required by applicable law.
In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain, and you are cautioned not to unduly rely upon these statements.
Investors and others should note that we may announce material business and financial information to our investors using our investor relations website (https://investors.compasstherapeutics.com), Securities and Exchange Commission or SEC,("SEC"), filings, webcasts, press releases, and conference calls. We use these mediums, including our website, to communicate with our members and public about our company, our products, and other issues. It is possible that the information that we make available may be deemed to be material information. We therefore encourage investors and others interested in our company to review the information that we make available on our website.
5
Overview
We are a clinical-stage, oncology-focused biopharmaceutical company developing proprietary antibodyantibody-based therapeutics intended to engagetreat multiple human diseases. Our scientific focus is on the relationship between angiogenesis, the immune system, to treat both solid tumors and hematological malignancies.tumor growth. Our immuno-oncologypipeline of novel product candidates include a clinical-stage monoclonal antibody targeting solid tumors, a novel bispecific targeting PD-1 and PD-L1, and a portfolio of bispecific and monoclonal antibodies at various stages of pre-clinical development. These product candidates areis designed to address threetarget multiple critical componentsbiological pathways required for an effective immune response to cancer:anti-tumor response. These include modulation of the microvasculature via angiogenesis-targeted agents, induction of a potent innate immune response; activation ofresponse via activators on effector cells in the adaptive immune system;tumor microenvironment, and alleviation of immunosuppressive mechanisms used by tumors to evade immune surveillance and activation.surveillance. We plan to advance our product candidates through clinical development either as both standalone therapies orand in combination with existing therapies as long as their continued development is supported byproprietary pipeline antibodies based on supportive clinical and nonclinical data.
Our pipeline is comprised of three product candidates. Our lead product candidate, CTX-009, is a bispecific antibody targeting Delta-like ligand 4 (“DLL4”), a ligand of Notch-1, and vascular endothelial growth factor A (“VEGF-A”). Simultaneous blockade of the VEGF-A and the Notch pathways is known to turn productive angiogenesis into non-productive angiogenesis, which leads to tumor shrinkage and apoptosis. Our second program CTX-471, is an agonistic antibody targeting a member of the tumor necrosis factor receptor superfamily member 9 (TNFRSF9), also known as CD-137, a co-stimulatory receptor which is mostly expressed on activated, but not on resting T-cells and NK cells. Our third program, CTX-8371, is a bispecific antibody targeting the programmed cell death protein-1 (“PD-1”), an inhibitory immune checkpoint receptor and its ligand PD-L1, two validated immune-oncology targets.
CTX-009, our bispecific antibody targeting DLL4 and VEGF-A, is currently undergoing clinical studies as a monotherapy and in combination with chemotherapy in the United States. We currently have two open U.S. clinical trials with CTX-009: a Phase 2 trial of CTX-009 as monotherapy in patients with metastatic colorectal cancer (“CRC”) who received two or three prior treatment regimens and a randomized Phase 2/3 trial of CTX-009 in combination with paclitaxel in patients with biliary tract cancer (“BTC”) who received one prior treatment regimen.
The two initial indications of BTC and CRC for CTX-009 were selected based on the results of the Phase 1,1b and 2 clinical trials and the significant unmet need for effective therapeutic regimens in these patient populations. There are an estimated 18,600 patients diagnosed with BTC in the U.S. each year and over 200,000 patients worldwide. Patients with BTC have a poor prognosis despite first line treatment with chemotherapy and immunotherapy, and no accepted standard of care in later treatment lines. For CRC, there are approximately 153,000 new patients diagnosed each year in the United States and more than 1.9 million patients worldwide. Despite advances in treatment of CRC in recent years with angiogenesis inhibitors and targeted therapies, there are few safe and effective treatment options for patients with CRC, and the majority of the patients are refractory to existing therapeutic regimens or relapse quickly and advance to later lines of therapy where treatment options are even more limited. We intend to expand the development of CTX-009 to additional tumor types with significant unmet need such as ovarian cancer and gastric cancer, among others. Following the generation of positive clinical data in later lines of therapy, we plan on studying CTX-009 in earlier settings in all indications where the data support it.
CTX-471, our CD137 agonistic antibody, is undergoing a two-part clinical trial. A Phase 1a trial of CTX-471 as monotherapy in patients with solid tumors who were previously treated with at least one checkpoint blocker and showed benefit for 3 months or more but ultimately progressed is fully enrolled and nearing completion. As of January 31, 2024, there is one patient remaining on treatment. In addition, a Phase 1b trial of CTX-471 in combination with the PD-1 inhibitor KEYTRUDA® in patients with selected solid tumors began in November 2022. The dose-escalation part of the study has been fully enrolled and enrollment in the dose expansion cohorts has begun. In the expansion cohort part of the study, we plan to enroll 60 patients with melanoma, non-small cell lung cancer (“NSCLC”) and small cell lung cancer (“SCLC”), who will randomly receive one of two doses.
CTX-471 targets a key node of the immune system with the goal of identifying a next generation immune-oncology treatment for the majority of patients who do not have a sustained response to current therapies across a variety of cancers. During its early clinical development, CTX-471 demonstrated monotherapy activity in the post PD-1/PD-L1 patient population across three solid tumor indications: melanoma, small cell lung cancer and mesothelioma. In the ongoing Phase 1b combination trial of CTX-471 with KEYTRUDA®we are looking to restore responses in patients who were previously treated with a checkpoint inhibitor, initially responded, but then progressed. The hypothesis we are testing is whether the addition of CTX-471 to KEYTRUDA®will restore responses in those patients. This trial is being conducted in patients with one of the following solid tumors: melanoma, non-small cell lung cancer and small cell lung cancer. Restoration of meaningful responses in one or more of those indications can help shape the regulatory path for CTX-471.
Our third program, CTX-8371, a bispecific antibody targeting PD-1 and PD-L1, is currently in a first-in-human Phase 1 clinical trial. We filed an investigational new drug application (“IND”) for CTX-8371 in the third quarter of 2023 and the IND was cleared by the FDA in the fourth quarter of 2023. The study is now open in the U.S. with the first patient expected to be dosed in the clinical trial by early second quarter 2024.
CTX-8371 emerged from an unbiased screen conducted with our StitchMabsTM platform. We have subsequently tested CTX-8371 in several in vitro and in vivo models where it demonstrated enhanced activation of immune responses when compared with commercially available checkpoint blockers. We believe that CTX-8371 has a potential to become a next generation checkpoint inhibitor with improved activity across various solid tumors relative to approved checkpoint blockers. Additionally, we are in the process of preclinically evaluating proprietary combination regimens of CTX-8371 with our other product candidates, CTX-009 and CTX-471.
In the fourth quarter of 2023 we announced a CEO succession plan; Vered Bisker-Leib, who was previously Compass President and COO, transitioned to Compass Chief Executive Officer and joined Compass board of directors and Thomas Schuetz, MD, PhD, Compass’ Scientific Founder and previously Chief Executive Officer, transitioned to President of Research and Development and appointed Vice Chair of the Compass board of directors. The transition took place on January 9, 2024.
Our Team
Our management team has a successful track record of building and growing biotechnology companies.
Our Chief Executive Officer, Vered Bisker-Leib, Ph.D., M.B.A., has over 20 years of experience in strategy, finance, business development, and operations of biotechnology and pharmaceutical companies. Under her financial leadership, Compass completed public and private financing transactions that generated more than $290 million in total proceeds since 2020. In 2021, Dr. Bisker-Leib identified and led the acquisition of TRIGR Therapeutics in a stock-for-stock transaction, which resulted in the addition of CTX-009 to the Compass’ pipeline. Dr. Bisker-Leib is also a board member of Ayala Pharmaceuticals. Prior to joining Compass Therapeutics, she served as an entrepreneur-in-residence with Atlas Venture. Previously, Dr. Bisker-Leib was chief business officer of Cydan (a biotech accelerator) and served as an executive director and global head of business development for the cardiovascular and metabolic franchises of Bristol-Myers Squibb.
Our President of Research and Development and Co-Founder, Thomas J. Schuetz, M.D., Ph.D. has over 30 years of experience in oncology, biopharmaceutical drug development and life science venture investing. Prior to co-founding Compass Therapeutics, Dr. Schuetz was a venture partner with OrbiMed Advisors LLC where he participated in OrbiMed’s investments in Enobia Pharma (sold to Alexion), Relypsa (sold to Galenica), Arteaus Therapeutics (sold to Eli Lilly), and Audentes (sold to Astellas) and served on the board of each of these companies. Dr. Schuetz was also the chief medical officer of Therion Biologics Corporation and was vice president of clinical affairs at Transkaryotic Therapies, a company acquired by Shire.
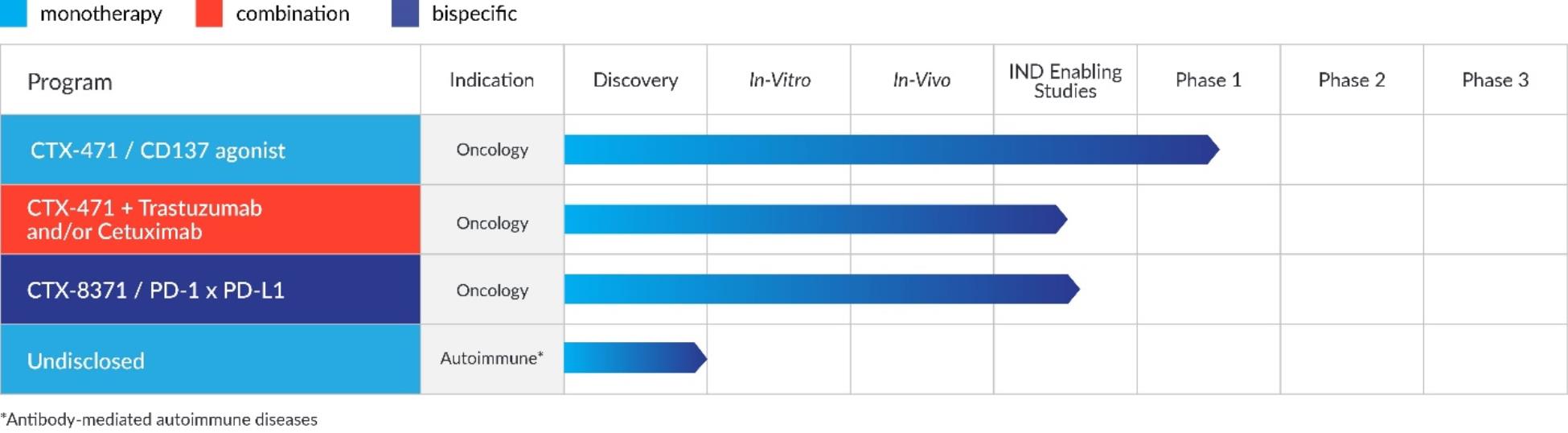
Pipeline
The figure below details our pipeline of product candidates: CTX-009, CTX-471 and CTX-8371.

Our Strategy
Our scientific focus is on the relationship between angiogenesis, the immune system, and tumor growth. Our pipeline of novel product candidates is designed to target multiple critical biological pathways required for an effective anti-tumor response. Our strategy to achieve this goal includes:
● | Advance our product candidate, CTX-009 (DLL4 x VEGF-A bispecific antibody), through clinical development to drug approval in multiple indications, either as a monotherapy or in combination with other therapies. CTX-009 is an investigational bispecific antibody that simultaneously blocks the DLL4 and VEGF-A signaling pathways, which are critical to angiogenesis and tumor vascularization. We chose BTC and CRC as our first indications based on a number of factors, including CTX-009 activity observed in the Phase 1, 1b and 2 clinical trials and lack of effective therapies for these patient populations. We have initiated a Phase 2 trial of CTX-009 in patients with advanced colorectal cancer and a randomized Phase 2/3 trial of CTX-009 in combination with paclitaxel in patients with BTC. Additionally, we intend to explore the potential of CTX-009 in other indications where the data support its potential therapeutic benefit such as ovarian cancer, gastric cancer, pancreatic cancer, renal cell cancer, liver cancer, neuroendocrine cancer and more. We are also developing a plan to study the combination of CTX-009 with our other product candidates, CTX-8371 and CTX-471. |
● | Advance our product candidate, CTX-471 (CD137 agonist antibody), through clinical development to evaluate its therapeutic potential alone and in combination with other therapies, and define a regulatory pathway for approval. We seek to translate the antitumor activity of CTX-471 observed in preclinical testing and in our Phase 1 trial into meaningful clinical results in patients with solid tumors, such as SCLC, NSCLC and melanoma. Our first Phase 1a clinical trial was conducted in patients who relapsed or progressed after at least three months of stable disease on prior checkpoint therapies. As of January 31, 2024, one patient remains on trial. Additionally, in November 2022, we dosed the first patient in a Phase 1b trial of CTX-471 in combination with KEYTRUDA®. Patients enrolled in the trial will be treated with CTX-471 in combination with KEYTRUDA® with the goal of restoring response. The dose-escalation portion of the study has been fully enrolled and enrollment in the dose expansion cohorts has begun. In the dose expansion cohorts, we plan on enrolling 60 patients with melanoma, NSCLC and SCLC, who will randomly receive one of two doses. This trial is enrolling patients with metastatic or locally advanced NSCLC, melanoma, and SCLC who have progressed after treatment with a checkpoint inhibitor. |
● | Advance CTX-8371 (PD-1 x PD-L1 bispecific antibody) through clinical development as a next generation checkpoint inhibitor. CTX-8371, our bispecific inhibitor that targets PD-1 and PD-L1, has demonstrated better antitumor activity in preclinical experiments than a single PD-1, a single PD-L1, or combinations of PD-1 and PD-L1 inhibitors. We submitted an IND to the FDA in the third quarter of 2023 and the FDA cleared the IND for CTX-8371 in the fourth quarter of 2023 allowing us to initiate a first-in-human Phase 1 clinical trial. The study is now open in the U.S. and we expect to dose the first patient in this study by early second quarter 2024. |
● | Seek strategic partnerships for select product candidates. Our technology platform is designed to generate a broad pipeline of product candidates with high potential for clinical application. We intend to assess on a case-by-case basis the opportunities for accelerating the preclinical and clinical development of these candidates in a capital-efficient manner, including selectively pursuing strategic partnerships with leading biopharmaceutical companies with domain-specific clinical development expertise to maximize the value of our pipeline. |
Product Candidates
We currently have three product candidates in the clinical stage of development: CTX-009, CTX-471 and CTX-8371.
CTX-009 (DLL4 X VEGF-A bispecific antibody)
CTX-009 (a.k.a. ABL001) is an investigational bispecific antibody that is designed to simultaneously block the DLL4 and VEGF-A signaling pathways, which are critical to angiogenesis and tumor vascularization. Preclinical and early clinical data of CTX-009 as a monotherapy and in combination with chemotherapy suggest that blockade of both pathways provides robust anti-tumor activity across several solid tumor indications, including colorectal, gastric, cholangiocarcinoma, pancreatic and non-small cell lung cancer.
CTX-009 is undergoing clinical development in patients with advanced solid tumors in the United States. A Phase 1 dose escalation and dose expansion monotherapy trial in patients with solid tumors, a Phase 1b trial of CTX-009 in combination with chemotherapy and a Phase 2 trial of CTX-009 in combination with chemotherapy in patients with advanced biliary tract cancer were completed in South Korea. Data from the Phase 2 trial were presented at ASCO GI in January 2023.
We currently have two open clinical trials in the United States: a Phase 2 trial of CTX-009 in patients with advanced colorectal cancer and a randomized Phase 2/3 trial of CTX-009 in combination with paclitaxel in patients with advanced biliary tract cancer.
We have licensed exclusive global rights to CTX-009, outside of South Korea, from ABL Bio, Inc. (“ABL Bio”), a South Korea-based clinical-stage company focused on developing antibody therapeutics. South Korean rights are held by Handok Pharmaceuticals, Inc. (“Handok”) and China rights were out-licensed from the Company to Elpiscience Biopharmaceuticals Co., Limited (“Elpiscience”).
Phase 1: Monotherapy Clinical Trial of CTX-009
An open-label, Phase 1 dose escalation and expansion trial designed to identify the optimal dose and to evaluate the safety, tolerability, pharmacokinetics (“PK”), pharmacodynamics and the anti-tumor activity of CTX-009 in patients with advanced solid tumors after failure of standard of care treatment was conducted by ABL Bio in South Korea. This trial consisted of a Phase 1a monotherapy dose escalation arm and a Phase 1b dose expansion arm. The trial was initiated in September 2017 and enrollment was completed in February 2021.
The dose escalation portion of the trial followed a traditional 3+3 dosing scheme where CTX-009 was administered by intravenous infusion across nine dose cohorts ranging from 0.3 to 17.5 mg/kg biweekly. Patients were enrolled in two arms: a Phase 1a dose escalation arm and a Phase 1b dose expansion arm. The expansion cohorts were 7.5, 10, 12.5 and 15 mg/kg. Patient tumor volumes were measured using CT scans at baseline and then every eight weeks.
Patient Demographics. A total of 45 patients were enrolled in the trial with advanced solid tumors that had been heavily pretreated, with a median of four prior lines of therapy. The median age of the patients was 53 years old and 54% of the patients were male and 46% female. Importantly, 75% of the patients had been previously dosed with anti-VEGF therapy. Most patients enrolled in the trial had either advanced colorectal cancer or advanced gastric cancer.
Safety Data Summary. A total of 45 patients were enrolled in the trial and received at least one dose of CTX-009. The dose escalation portion of the trial took place without interruption, and the highest dose arm was 17.5 mg/kg. Importantly, the maximal tolerated dose was not determined in this trial. CTX-009 was observed to be generally well-tolerated.
There were 44 Treatment Related Adverse Events (“TRAEs”) observed in more than 5% of the 45 patients enrolled. The most prominent TRAE was hypertension, which was observed in 37.8% of the patients. Grade 3 or higher TRAEs observed in over 5% of the patients include a total of 11 Grade 3 events, the most frequent being hypertension observed in 15.6% of the patients, followed by gastrointestinal disorders, observed in 4.4% of the patients, followed by general disorders and nervous system disorders, observed in 2.2% of the patients each. A summary of the TRAEs observed in over 5% of the patients is depicted in the table below.
Treatment-related adverse events observed in > 5% of patients | Total (n) | Total (%) | Grade 3 (n) | Grade 3 (%) | ||||||||||||
Hypertension* | 17 | 37.8 | 7 | 15.6 | ||||||||||||
General disorders (fatigue, fever, asthenia, edema, etc.) | 7 | 15.6 | 1 | 2.2 | ||||||||||||
Nervous system disorders (headache, dizziness) | 7 | 15.6 | 1 | 2.2 | ||||||||||||
Gastrointestinal disorders (nausea, vomiting, etc.) | 6 | 13.3 | 2 | 4.4 | ||||||||||||
Pulmonary hypertension | 4 | 8.9 | 0 | 0 | ||||||||||||
Proteinuria | 3 | 6.7 | 0 | 0 | ||||||||||||
* Hypertension is a well-known side effect of anti-VEGF blockers. In clinical trials of bevacizumab, incidence of Grade 3-4 hypertension ranged between 5%-18% (as indicated in the label). Hypertension is typically managed by anti-hypertensive drugs.
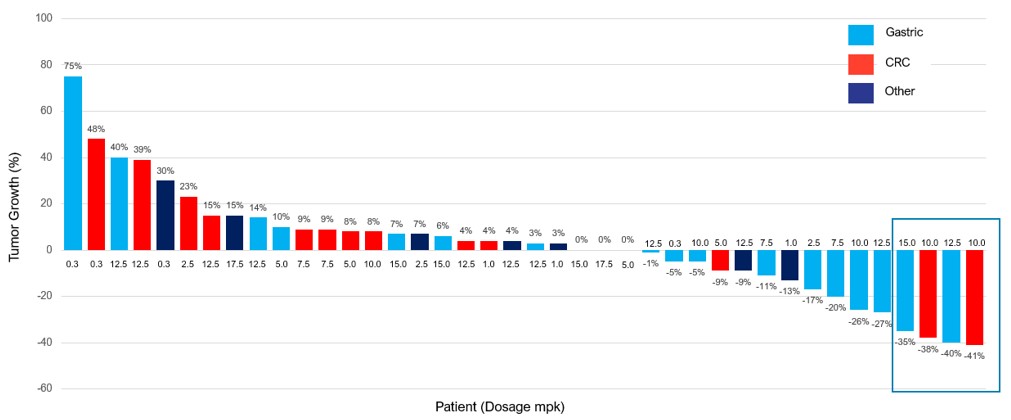
Activity Data Summary. A total of 40 out of the 45 patients enrolled in the trial were evaluable for the purpose of determination of anti-tumor activity of CTX-009 since five patients did not reach their first scan at week eight due to progressive disease ("PD") or for other reasons. Responses started to emerge at the 10 mg/kg dose level. Sixteen of the 40 evaluable patients were dosed at the 10 or 12.5 mg/kg dose levels, which represent what we project to be the efficacious dose levels. Among those 16 patients, there were three partial responses ("PRs") confirmed by RECIST 1.1 with an overall response rate ("ORR") of 18.8% and eight patients with stable disease (“SD”), with a clinical benefit rate ("CBR") of 68.8%. Two of the three PRs were in patients with advanced colorectal cancer and one of the three PRs was in a patient with advanced gastric cancer. In addition, one of the patients with gastric cancer had a 35% decline in tumor mass relative to baseline. However, that regression was not confirmed upon a second CT scan, and hence not included in the ORR, and the best response of this patient included in the data set is stable disease. The average time to progression (“TTP”) of the advanced colorectal cancer patients treated at the 10 or 12.5 mg/kg was 6.7 months, and the average TTP of the advanced gastric cancer patients treated at the 10 or 12.5 mg/kg was 3.9 months
.
Phase 1 monotherapy trial: expansion cohorts (10 and 12.5 mg/kg)
Monotherapy expansion cohorts (10 and 12.5 mg/kg) |
Patients | Prior VEGF target therapy | PR | SD | CBR | Median time to progression (“TTP”) | ||||||||||||||||||
All patients | 16 | 75 | % | 19 | % | 50 | % | 69 | % | 3.9 months | ||||||||||||||
Colorectal cancer | 6 | 100 | % | 33 | % | 33 | % | 67 | % | 6.7 months | ||||||||||||||
Gastric cancer | 8 | 63 | % | 13 | % | 63 | % | 75 | % | 3.9 months | ||||||||||||||
A waterfall plot depicting the best responses for each of the 40 evaluable patients is presented below.
Phase 1b: Combination Clinical Trial of CTX-009
An open-label, combination Phase 1b clinical trial to evaluate the safety, PK, anti-tumor activity and the recommended Phase 2 dose (“RP2D”) of CTX-009 in combination with paclitaxel or irinotecan chemotherapy was conducted by ABL Bio and Handok in South Korea. This trial was initiated in June 2020, enrollment was completed in December 2020, and the trial was completed in November 2021 (clinicaltrials.gov identifier NCT04492033).
The trial includes two cohorts, each of which is divided into two groups. The first cohort was administered 10 or 12.5 mg/kg of CTX-009 on a biweekly basis, in combination with 80 mg/m2 paclitaxel administered weekly. The second cohort was administered 10 or 12.5 mg/kg of CTX-009 in combination with 150 mg/m2 irinotecan on a biweekly basis.
Patient Demographics. A total of 17 patients were enrolled in the trial. Patients enrolled in the trial were heavily pretreated, and the trial included patients with advanced cholangiocarcinoma, colorectal, pancreatic, gastric and other cancers with a median of three prior lines of therapy.
Safety Data Summary. In general, CTX-009 was observed to be well-tolerated. Adverse Events (“AEs”) that were determined to be probably or possibly related to CTX-009 treatment included Grade 3 hypertension observed in four patients (24%). Other AEs observed were Grade 3 neutropenia (12%), Grade 3 anemia (18%) and Grade 3 thrombocytopenia (12%), which were all attributed to the concomitant chemotherapy agent (paclitaxel or irinotecan). Pulmonary hypertension was monitored carefully in the trial via measurement of BNP levels and echocardiograms, and there were five Grade 1 pulmonary hypertension events, all of which resolved.
A summary of the TRAEs is depicted in the table below:
Drug-related adverse events | Total | Total | Grade 3 | Grade 3 |
observed in > 1 patient | (n) | (%) | (n) | (%) |
Hypertension | 8 | 47 | 4 | 24 |
Nausea | 8 | 47 | 1 | 6 |
Fatigue | 6 | 35 | 1 | 6 |
Neutropenia* | 6 | 35 | 2 | 12 |
Anemia* | 4 | 24 | 3 | 18 |
Thrombocytopenia* | 2 | 12 | 2 | 12 |
Diarrhea | 5 | 29 | 0 | 0 |
Anorexia | 5 | 29 | 0 | 0 |
Proteinuria | 5 | 29 | 0 | 0 |
Pulmonary hypertension (all grade 1) | 5 | 29 | 0 | 0 |
Dyspnea | 4 | 24 | 0 | 0 |
Gingival edema (mucositis) | 2 | 12 | 0 | 0 |
Anal hemorrhage | 2 | 12 | 0 | 0 |
*Labeled Grade 3/4 cytopenia events for concomitant chemotherapy agent: | ||||
Irinotecan: 31.4% neutropenia, 4.5% anemia, 1.7% thrombocytopenia | ||||
Paclitaxel: 52% neutropenia, 16% anemia, 7% thrombocytopenia | ||||
Activity data summary. Of the 17 patients enrolled, there were four PRs, including three PRs that were confirmed by RECIST 1.1 and one PR which was unconfirmed, representing a 23.5% ORR and nine patients with SD, representing a CBR of 76.5%. The unconfirmed PR was in a patient with NSCLC who was on the trial for over a year with a measurable tumor decline. Of the four patients with advanced cholangiocarcinoma enrolled in the trial, there were two PRs confirmed by RECIST 1.1 with 41% and 62% declines in tumor burden, respectively, representing an ORR in cholangiocarcinoma of 50%. A third patient with cholangiocarcinoma had stable disease with 28% decline in the patient’s tumor burden, and therefore the CBR observed in cholangiocarcinoma is three out of four, or 75%. The responses in cholangiocarcinoma were particularly durable with a median duration of response, or DOR, of 9.7 months.
A waterfall plot depicting the best responses for each of the 17 evaluable patients in the Phase 1b trial is presented below.
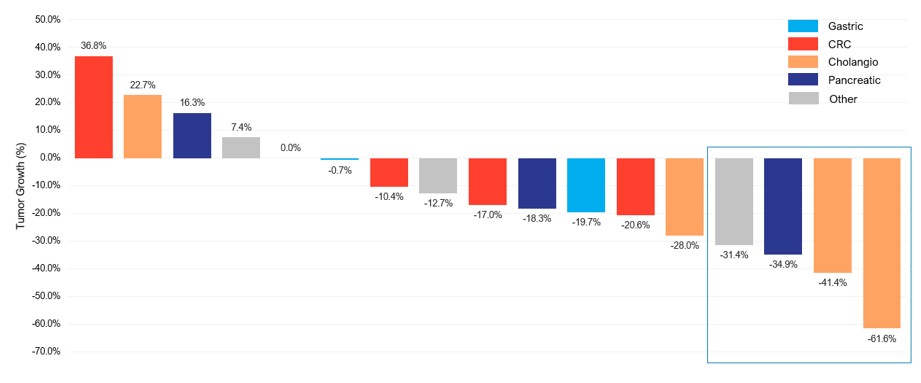
Phase 1: Summary of Clinical Activity of CTX-009 at the RP2D
The observed ORR of CTX-009 at the 10 and 12.5 mg/kg doses were 18.8% (3/16) as a monotherapy and 23.5% (4/17) in combination with chemotherapy. The CBR of CTX-009 at the 10 and 12.5 mg/kg were 68.8% (11/16) as a monotherapy and 76.5% (13/17) in combination with chemotherapy
On October 8, 2021, we, along with ABL Bio, presented clinical trial data from the CTX-009 Phase 1a/1b dose escalation and dose expansion trial at a plenary oral session during the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics (Abstract Number: 4749; Session title: Plenary Session 2: New Drugs on the Horizon I). The significant findings presented were as follows:
● | CTX-009 was generally well-tolerated and demonstrated single agent activity in heavily pre-treated patients with solid tumors who were resistant to anti-VEGF therapies, mostly of colorectal and gastric origins |
● | The maximum tolerated dose (“MTD”) was not reached, and the RP2D of CTX-009 were determined to be 10.0 and 12.5 mg/kg biweekly |
● | ORR of CTX-009 as a monotherapy across all doses tested (0.3 – 17.5 mg/kg) was 8% and the CBR was 62% in patients treated at the 3rd and 4th line settings |
● | Treatment with CTX-009 as a monotherapy at the RP2D (10.0 mg/kg and 12.5 mg/kg) led to 18.8% (n=3/16) ORR, not including an additional unconfirmed PR, and a 68.5% CBR (n=11/16) |
Phase 2: Data from Combination Clinical Trial of CTX-009 in BTC in South Korea
A Phase 2 trial of CTX-009 in combination with paclitaxel was initiated by Handok in the first quarter of 2021 in patients with BTCs. The study has been completed and final data analysis has begun. The trial enrolled patients with unresectable advanced, metastatic, or relapsed BTCs who had received one or two prior systemic therapies.
This Phase 2 trial utilizes a Simon Two-Stage adaptive design where the criteria to advance to the second stage of the trial was three PRs observed in 21 evaluable patients. As of November 9, 2022, in the preliminary analysis of all 24 patients participating in the study, CTX-009 with paclitaxel demonstrated a 37.5% ORR based on 9 patients with PRs that were confirmed by RECIST 1.1. The trial has met the criteria to advance to Part 2 and Part 1 of the study is now complete.
The results of Part 1 of the Phase 2 trial were presented at the 2023 American Society of Clinical Oncology Gastrointestinal Cancers Symposium (“ASCO GI”) in January 2023.
Safety Data Summary
CTX-009 safety data has been analyzed and was observed to be generally well-tolerated. The Phase 2 safety data are generally consistent with the safety data of the Phase 1 trials. Of the 24 patients enrolled in the first stage of the trial, all patients had at least one treatment emergent adverse event (“TEAE”). Grade 3 or greater TEAEs were reported in 95.8% of patients regardless of the relationship to CTX-009 or paclitaxel, including decreased neutrophil count (83.3%), hypertension (16.7%), anemia (20.8%), and decreased platelet count (12.5%). Grade 3 or greater adverse events that were designated to be of special interest (“AESIs”) by the trial investigators were hemoptysis or hemorrhage (12.5%) and GI or tumor perforation (8.3%), with 0% for pulmonary hypertension, wound healing complication and cardiac failure.
The table below depicts a summary of the TEAEs in 24 patients as of November 9, 2022.
Total | Total | Grade 3 | Grade 3 | |||||||||||||
Treatment Emergent Adverse Events observed in > 1 patient | (n) | (%) | (n) | (%) | ||||||||||||
Neutrophil count decreased | 22 | 92 | 20 | 83 | ||||||||||||
Hypertension | 12 | 50 | 4 | 17 | ||||||||||||
Platelet count decreased | 9 | 38 | 3 | 13 | ||||||||||||
Anaemia | 5 | 21 | 5 | 21 | ||||||||||||
Ascites | 4 | 17 | 2 | 8 | ||||||||||||
Decreased appetite | 4 | 17 | 2 | 8 | ||||||||||||
Neutropenia | 2 | 8 | 2 | 8 | ||||||||||||
Liver abscess | 2 | 8 | 2 | 8 | ||||||||||||
Hepatic infection | 2 | 8 | 2 | 8 | ||||||||||||
Cholangitis | 2 | 8 | 2 | 8 | ||||||||||||
Embolism | 2 | 8 | 2 | 8 | ||||||||||||
Activity Data Summary
The first stage of the trial enrolled 24 patients and 22 of those patients are considered evaluable.
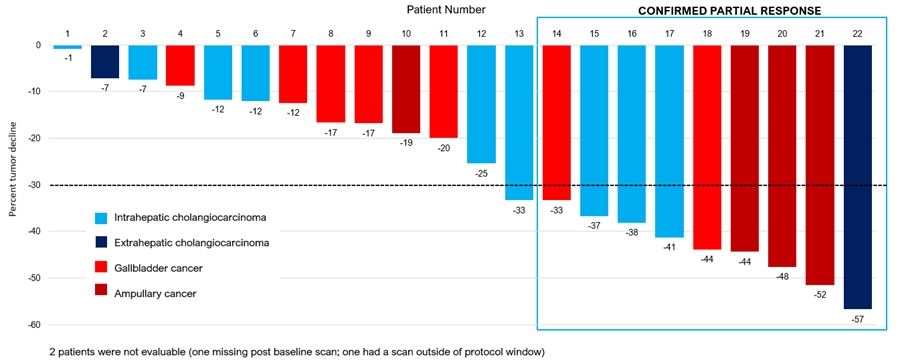
Nine PRs confirmed by RECIST 1.1 were observed leading to an ORR of 37.5%, and 22 of the 24 patients evaluated have had stable disease or better with a decline in tumor burden leading to a CBR of 92%. PRs were observed in all four tumor sub-types types (intrahepatic cholangiocarcinoma, extrahepatic cholangiocarcinoma, gallbladder cancer, and ampullary carcinoma). Of the 24 patients, 11 were treated in the second line with 7 PRs observed, leading to an ORR of 63.6% in this sub-group. In the third line, 13 patients were treated with 2 PRs observed, leading to an ORR of 15.4% in the second sub-group.
After a median follow up of approximately 12 months, the median progression free survival (“PFS”) was 9.4 months, median duration of response (“DOR”) was 6.9 months and median overall survival (“OS”) was 12.5 months.
For reference, one regimen that has been studied in patients with advanced BTC is FOLFOX, the regimen recommended in the guidelines of the National Comprehensive Cancer Network (NCCN) for patients with BTC treated in the second-line setting. FOLFOX demonstrated an ORR of 5%, a median PFS of 4.0 months, and a median OS of 6.2 months in a randomized study against best supportive care.
The waterfall plot below depicts the best response for the 22 patients evaluated in the trial as of November 9, 2022.
Phase 2 Data
CTX-009 + paclitaxel in BTC
Best Response by Patient
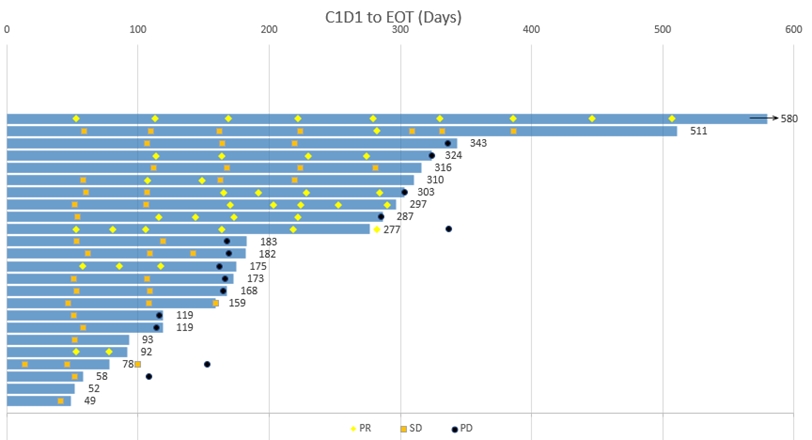
The swimmer plot below depicts the duration that each patient has been on treatment as of November 9, 2022 (N=24):
Phase 2 Data
CTX-009 + paclitaxel in BTC
Duration of Treatment by Patient
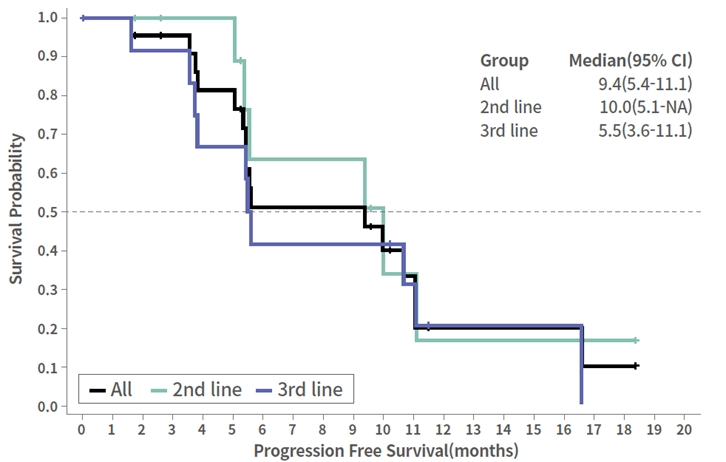
The median PFS of all patients in the study is 9.4 months. Patients treated in the second line setting had a median PFS of 10.0 months and patients treated in the third line setting had a median PFS of 5.5 months.
Phase 2 Data
CTX-009 + paclitaxel in BTC
Progression Free Survival
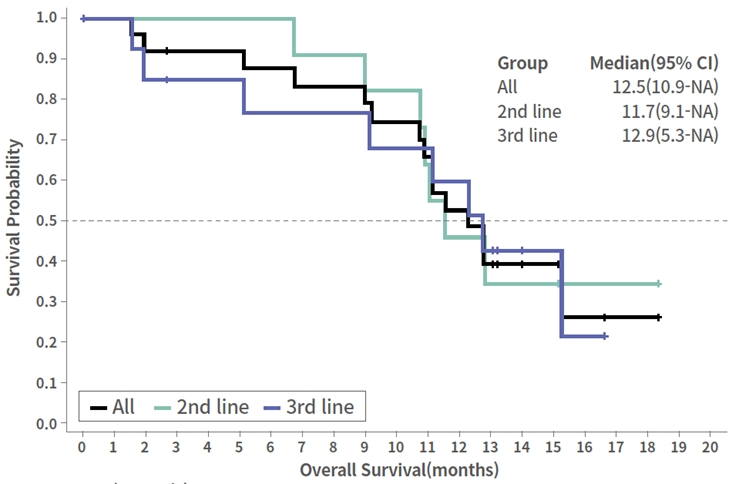
The median OS of all 24 patients enrolled and dosed in the study is 12.5 months. Patients treated in the second line setting had a median OS of 11.7 months and patients treated in the third line setting has a median OS of 12.9 months.
Phase 2 Data
CTX-009 + paclitaxel in BTC
Overall Survival
Status of CTX-009 Development
Our strategy is to develop CTX-009 in all of the indications in which patients have a need for effective and novel therapeutic agents and data support the potential therapeutic benefit of CTX-009. We chose BTC and CRC as our lead indications based on a number of factors, including CTX-009 activity observed in the Phase 1, 1b and 2 clinical trials, lack of effective therapies for these patient populations and the potential for a straight-forward regulatory route to approval.
We submitted an IND to the FDA in December 2021 for CTX-009 and the FDA cleared our IND application in January 2022. The following trials are being conducted in the United States under this IND.
Biliary Tract Cancers – BTC
Following conversations with the FDA, we submitted a protocol for a randomized Phase 2/3 trial for CTX-009 in combination with paclitaxel in adult patients with unresectable, advanced, metastatic or recurrent biliary tract cancers who have received one prior systemic chemotherapy regimen. The trial is designed to assess the safety and efficacy of the combination of CTX-009 and paclitaxel versus paclitaxel alone. A schema of the trial design is provided below:
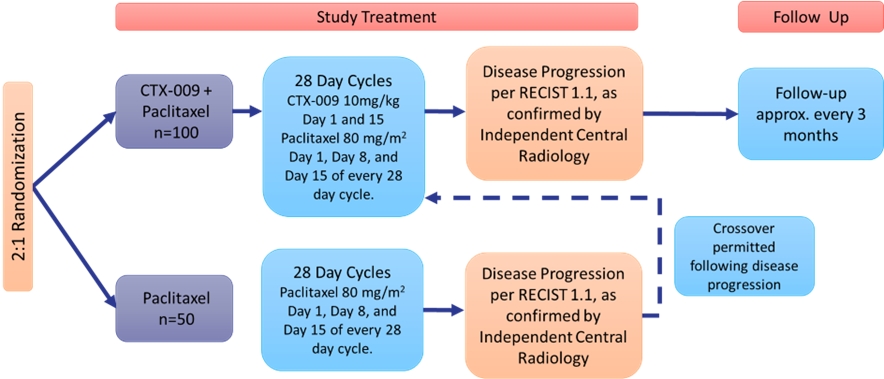
The trial will enroll 150 patients, who will be randomized in a 2:1 ratio to receive CTX-009 plus paclitaxel (n=100) or paclitaxel alone (n=50). The primary endpoint of the trial is ORR and the secondary endpoints include PFS, DCR, DOR and OS among other. Patients who were randomized to receive paclitaxel and have progressed on their regimen may cross over to the CTX-009 plus paclitaxel arm after progression on paclitaxel if they still meet the enrollment criteria for the study. A detailed description of the trial can be found on www.clinicaltrials.gov (Identifier NCT 05506943).
We are currently enrolling patients for this trial. Depending on the trial results, this trial could serve as a registrational trial to support a biologics license application ("BLA") submission for CTX-009 in combination with paclitaxel as a treatment of BTC patients in the second line setting. Enrollment is expected to be completed by mid-year 2024, with top line data expected in the second half of 2024. Based on the trial results, a BLA could be filed as early as the second half of 2025.
Colorectal Cancer – CRC
We are also conducting a Phase 2 monotherapy clinical trial of CTX-009 in patients with metastatic colorectal cancer who have received two or three prior systemic therapies. This trial is designed to assess the safety and efficacy of CTX-009 as a monotherapy in patients with advanced colorectal cancer. A schema of the trial design is provided below:
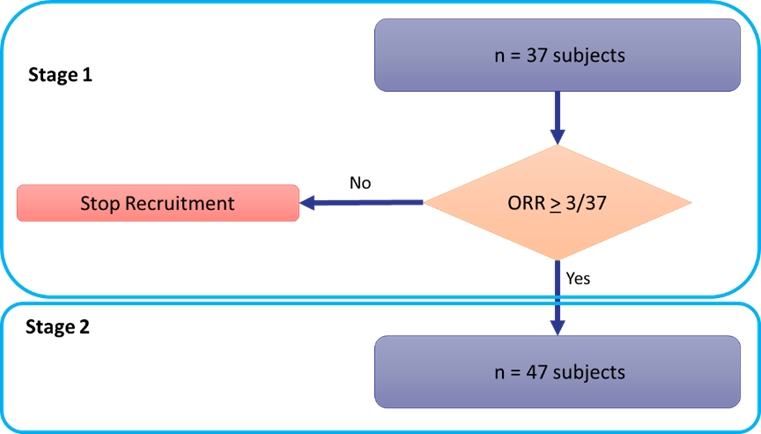
The trial utilizes a Simon Two-Stage adaptive design where the criteria to advance to the second stage of the trial is three PRs observed in 37 patients enrolled in Stage 1 of the trial. Based on the Simon Two-Stage design, when the criteria for the first stage is met, the trial may progress to the second stage, at which time 47 additional patients could be enrolled. The trial can be found on www.clinicaltrials.gov (identifier NCT 05513742).
In January 2023, the first patient was dosed in this trial and enrollment in Stage 1 of the trial was complete in the first quarter of 2024. Initial results from Stage 1 of this trial are expected by mid-year 2024.
Additional Plans for CTX-009
We intend to explore the potential of CTX-009 in additional indications, based on data from pre-clinical models, potential biomarkers such as DLL4, and clinical data from CTX-009 trials providing signs of potential activity of CTX-009 in additional indications such as ovarian cancer, gastric cancer, pancreatic cancer, renal cell cancer, liver cancer, neuroendocrine cancer and others.
In addition, we are developing a plan to study the combination of CTX-009 with our novel bispecific checkpoint blocker, CTX-8371, and with other checkpoint blockers, such as pembrolizumab and atezolizumab. Additionally, we are considering the combination of CTX-009 with our novel CD137 agonistic antibody, CTX-471, which is currently in a Phase 1b clinical trial in patients with advanced solid tumors.
CTX-471 (CD137agonist antibody)
CTX-471, our monoclonal antibody product candidate, is a fully human, IgG4 monoclonal antibody that is an agonist of CD137, a key co-stimulatory receptor on immune cells. In preclinical studies, weBinding of CTX-471 to CD137 has been observed that CTX-471 led to complete eradicationlead to ligand-stimulated activation of large tumors in mice when dosed as a monotherapy.T-cells and NK cells. In treated mice, dosing with CTX-471 was associated withled to extensive reprogramming of the tumor microenvironment, resultingincluding increased recruitment of immune cells, reversal of exhausted cytotoxic CD8+ T-cells, reductions in increased survivalimmunosuppressive regulatory T-cells and long-term immune protection.reductions in immunosuppressive tumor-associated macrophages. Long after therapy had been completed, after more thanthe completion of the treatment with CTX-471, a period equal to eight half-lives of the antibody, treated mice exhibited immune memory that prevented reestablishment of the same tumor. Based
The CD137 antigenic site recognized by CTX-471 does not block the binding of CD137 ligand and is differentiated from the site recognized by CD137 antibodies from competitors. We designed the antibody using different backbones and chose to use a human IgG4 backbone for CTX-471 to enable engagement of Fc receptors FcɣRI and FcɣRIIb to facilitate CD137 cross-linking while avoiding binding to FcɣRIIIa and depletion of immune effector cells through ADCC.
Immune cell depletion experiments showed that the activity of CTX-471 required the presence of CD4+ T-cells, CD8+ T-cells, and NK cells, indicating a coordinated involvement of both innate and adaptive immune cells. Encouragingly, treatment of tumors in mice with CTX-471 led to a marked reprogramming of the immune component of the tumor microenvironment. We also observed that tumors treated with CTX-471 had an approximate two-fold reduction in the number of immunosuppressive tumor-associated macrophages.
In addition, we have observed potent activity in other syngeneic tumor models including tumor eradication in the A20 model of lymphoma, the MC38 model of colon carcinoma and in the EMT6 model of breast cancer.
We believe that the ability of CTX-471 to transform the tumor microenvironment through the combined action of immune cell recruitment, alleviation of T-cell exhaustion, suppression of Tregs, and reduction of tumor suppressing macrophages leads to CTX-471’s antitumor activity in mouse models.
Dosing Strategy
In contrast to dosing strategies for other immuno-oncology antibodies, such as checkpoint inhibitors where the goal is often to deliver a dose that is capable of fully inhibiting the receptor at all times, our dose selection for CTX-471 studies was aimed at binding to only a fraction of the available CD137 receptors. Dosing of an agonist antibody, such as CTX-471, at levels capable of binding to the majority of receptors can lead to inappropriate cell activation and downregulation of the receptor and overall weaker activity.
Agonist antibodies typically trigger their activity through independent binding of each of their two antigen-binding domains to individual receptors on thesea cell surface. This binding to both receptors at once forces the receptors into close physical proximity. This grouping of receptors that drives receptor activation, especially when the ratio of antibody molecules to receptor molecules is relatively low. As the ratio of antibody to receptor increases, the level of receptor activation increases up to a point above which activation may decrease due to down-regulation of the receptors. This results in a bell-shaped activation curve in which maximal activation occurs at intermediate antibody concentrations.
We observed evidence of the importance of lower receptor occupancy while screening candidate antibodies against CD137. The antibodies with the greatest tumor-killing activity were the ones with intermediate potency. Very high-potency antibodies had weaker antitumor activity.
Consistent with the finding of lower activity at high antibody to receptor levels, we observed that the antitumor activity of CTX-471 appeared to peak at doses between 50 ug and other preclinical results,100 ug in the mouse CT26 tumor model. At the higher dose of 200 ug, the number of complete responses, four out of eight mice, was less than that observed at 100 ug, seven out of eight mice, suggesting that the optimal receptor occupancy had been exceeded. This is also consistent with our observation that intermediate affinity antibodies exhibited greater antitumor activity compared to high affinity antibodies.
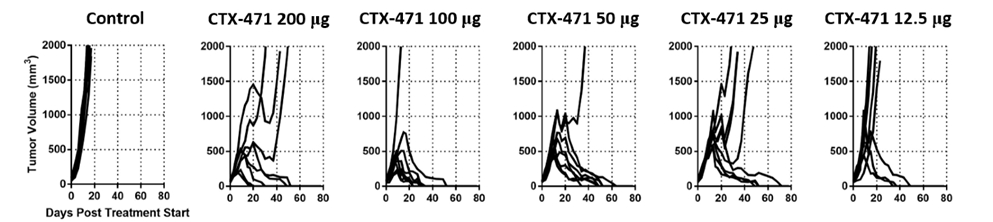
Antitumor activity of CTX-471 is optimized at intermediate dose and decreased at the highest dose level
Our findings are consistent with those reported for an agonist antibody against OX40, another immune target in oncology. Thus, for many agonist antibodies, it is likely that both intermediate affinities and intermediate doses will deliver optimal activity.
Phase 1 Clinical Trial of CTX-471
We are conducting a Phase 1, open-label, first-in-human trial of CTX-471 administered as a monotherapy or in combination with pembrolizumab (KEYTRUDA®) in patients with metastatic or locally advanced malignancies that have progressed while receiving an approved PD-1 or PD-L1 inhibitor. We selected this population of patients for this trial because multiple clinical trials and meta-analyses have shown that not all patients respond to checkpoint inhibitor therapy due to many possible factors. By focusing on those that did previously respond to checkpoint inhibitor therapy, we believe that this trial design enriches for patients who have tumors that are capable of being recognized and killed by their immune systems. The trial is being conducted with two treatment arms, the Monotherapy Arm and the Combination Arm. Each arm will have two parts: a dose escalation cohort and a dose expansion cohort. The Monotherapy Arm is nearing completion and enrollment began in the Combination Arm in the fourth quarter of 2022.
Phase 1 Clinical Trial Data
In July 2019, we initiated a Phase 1 trial evaluating the safety and tolerability of CTX-471 as a monotherapy in oncology patients with solid tumors who were previously treated with PD-1 or PD-L1 immune checkpoint inhibitors and subsequently relapsed or progressed after a period of stable disease. The design of this trial includes a dose escalation stage (Phase 1a) followed by a dose expansion stage (Phase 1b).stage. The dose escalationdose-escalation stage of the Phase 1 trialmonotherapy (Phase 1a monotherapy) has been completed and CTX-471 was observed to be generally well-tolerated. The dose expansion stage of the monotherapy trial (Phase 1b monotherapy) is currently ongoing and asnearing completion.
Phase 1: Dose Escalation – Monotherapy Arm
In the Phase 1a dose escalation stage, 19 patients received CTX-471 in the four dosing cohorts set forth in Figure 8 below.
Cohort 1 | Cohort 2 | Cohort 3 | Cohort 4 | |||||||||||||||||
Cohort | 0.1 mg/kg | 0.3 mg/kg | 0.6 mg/kg | 1.2 mg/kg | Total | |||||||||||||||
Enrolled | 3 | 3 | 6 | 7 | 19 | |||||||||||||||
The number of February 28, 2021, 11 patients have received at least onedosed with CTX-471 in the Phase 1a dose escalation stage
CTX-471 was observed to be generally well-tolerated in the Phase 1a stage of CTX-471. Of the 11 patients treated so far, six patients have reached their first tumor evaluation at week 9, of which five had stable disease. Subsequently, one of those patients who has advanced small cell lung cancer had a partial response at Week 17. As of February 28, 2021, there have been no treatment-relatedtrial. There were two serious adverse events or SAEs,(“SAEs”) determined to be treatment-related, which included one hypoxia event that resolved with approximately one day of supplemental oxygen therapy and one immune thrombocytic purpura event that also resolved. The dose-limiting toxicities were two events of thrombocytopenia in Cohort 4, which was expanded from three to six patients to collect additional safety data. Based on these results, 0.6 mg/kg was determined to be the maximum tolerated dose.
While the goal of the Phase 1a stage of the trial was to evaluate the safety and tolerability of CTX-471, we also collected data from these patients to evaluate the PK of CTX-471, the potential development of anti-drug antibodies of CTX-471, and to obtain certain efficacy data, based on the ORR, as per Response Evaluation Criteria in Solid Tumors, RECIST. Patients who were enrolled in the Phase 1a stage of the trial have been evaluated every eight weeks by imaging techniques, such as MRI or CT, until disease progression or withdrawal from the trial, in order to collect such data.
The Phase 1a stage of the trial is now complete. None of the patients enrolled in the Phase 1a stage of the trial had a complete response (“CR”) or a PR by RECIST. The best overall response has been stable disease. Two patients with NSCLC had stable disease until progression at Weeks 25 and 41, respectively. In addition, one patient with metastatic melanoma of mucosal origin had an approximately 24% decline in the total size of his measured metastatic tumors (target lesions) at week 33. This patient remained on the trial and received CTX-471 for 49 weeks.
We have analyzed preliminary PK data from the trial and these data have confirmed our receptor occupancy modeling. Based on this modeling and the correlation of the observed PK with our predictions, we selected 0.3 mg/kg and 0.6 mg/kg as the doses of the cohorts of our Phase 1b stage of the trial. We estimate that a dose of 0.3 mg/kg would lead to a peak receptor occupancy of approximately 50% and a dose of 0.6 mg/kg would lead to a peak receptor occupancy of approximately 70%.
Phase 1: Dose Expansion – Monotherapy Arm
The dose expansion stage of the trial is fully enrolled, and as of January 31, 2024, there is one ongoing patient in the trial and the study is nearing completion. 60 patients with 17 different cancers have been enrolled in the trial. One patient (noted below) had a CR and four additional patients had a PR. Of the four partial responses observed in this dose expansion cohort, three PRs have been confirmed by RECIST 1.1 and the fourth PR is unconfirmed and will remain unconfirmed. There have been nine SAEs related to CTX-471 in the dose expansion stage of the trial. We expect to complete the Phase 1b stageAll nine events resolved .
The responses observed as described below:
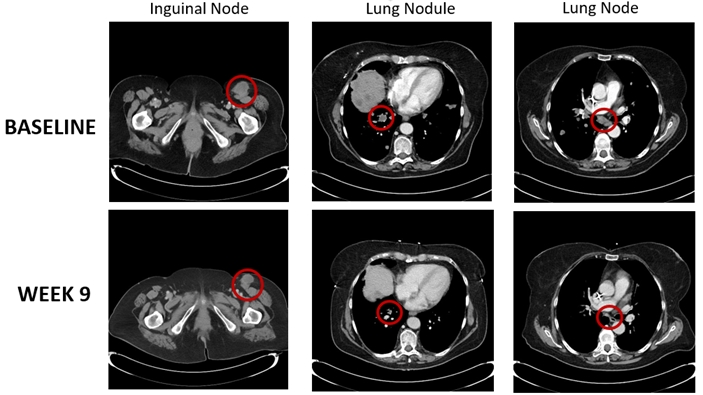
● | A PR was observed in a patient with metastatic melanoma who was previously treated with nivolumab and progressed on nivolumab. Below is a series of CT scan images from this patient. The patient had multiple metastases at baseline and reached a PR based on a 38% decline in linear tumor burden at week nine. This PR was confirmed at week 17. |
● | A PR was observed in a patient with metastatic melanoma of mucosal origin who was previously treated with first-line regimen of ipilimumab plus nivolumab followed by second-line nivolumab as a monotherapy. After progressing on the prior regimens, the patient had joined the trial with multiple metastases at baseline. This patient reached a PR based on a 58% decline in linear tumor burden at week 17. This PR was not confirmed. |
● | A PR was observed in a patient with mesothelioma. This response was confirmed by RECIST 1.1. |
● | A PR was observed in a patient with melanoma treated in the third line setting. This response was confirmed by RECIST 1.1. This patient has been on the study for more than 2 years and is still ongoing. |
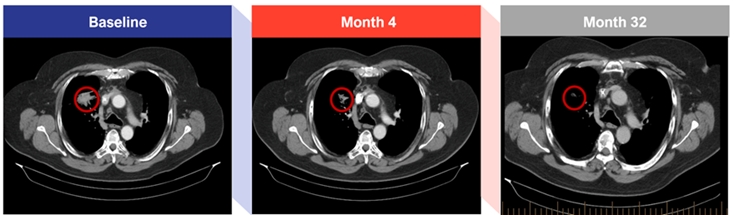
● | The CR was observed in a patient with advanced small cell lung cancer who was previously treated with the carboplatin/etoposide and atezolizumab (a PD-L1 blocker) regimen in the first line followed by a treatment with nivolumab (a PD-1 blocker) in the second line. After progression on prior regimens this patient joined the trial and had a PR at week 17 which was confirmed at week 25. This patient was dosed with CTX-471 for more than three years with a durable PR, and in Q4 2023, following tumor regression observed by CT-Scan, the patient was tested by PET scan and a CR was determined. Below is a series of CT scan images from this patient of the largest mass (RUL Lung) which was ~4 cm at baseline. |
Of note, out of the five responses observed in this trial, duringthree responses were in patients with melanoma, leading to a response rate of 27% in this subgroup of melanoma patients (3 out of 11). The other responses were in mesothelioma (1 out of 4) and SCLC (1 out of 3).
Phase 1: Dosing - Combination Arm
In October 2022, we announced a clinical collaboration with Merck (known as MSD outside the second half of 2021United States and Canada) to initiate a Phase 2/3 trial of CTX-471 in the second half of 2022.
Pending the results of our Phase 1 monotherapy trial of CTX-471, we plan to initiate a second Phase 1 trial ofevaluate CTX-471 in combination with trastuzumab, marketed as Herceptin® by Genentech, in selected human epidermal growth factor receptor 2, or Her2, positive tumorsKEYTRUDA® (pembrolizumab). Compass is the study sponsor and Merck provides the clinical supply of KEYTRUDA®. Additionally, we formed a joint development committee (JDC) with cetuximab, marketed as Erbitux® by Eli Lilly, in epidermal growth factor receptor, or EGFR, positive tumors. We could submit an IND application forMerck to review the results of this combinationclinical trial.
In November 2022, we announced the first patient was dosed in the second halfcombination arm of 2021.the Phase 1 trial. Patients enrolled in the combination arm will be treated with CTX-471 by intravenous infusion every two weeks in combination with a fixed dose of pembrolizumab (400 mg) by intravenous infusion every six weeks with the goal of restoring response. The dose-escalation portion of the study enrolled patients with metastatic or locally advanced non-small cell lung cancer, melanoma, small cell lung cancer, mesothelioma and head and neck cancer that have progressed after treatment with a PD-1 or PD-L1 checkpoint inhibitor. At the end of 2023, the dose-escalation portion of the study was fully enrolled and this portion of the study is near completion. No dose-limiting toxicities were observed. As of January 30, 2024, there have been no responses observed in the dose escalation portion of the Phase 1 combination study.
Our second product candidate,
In the expansion cohort, we plan on enrolling 60 patients with melanoma, NSCLC and SCLC, who will be randomly receive one of two doses of CTX-471: 0.3 (mg/Kg) every two weeks or 0.6 (mg/Kg) every two weeks in combination with a fixed dose of pembrolizumab (400 mg) every six weeks. Enrollment in the dose expansion cohorts began in Q4 2023.
Development Plans for CTX-471
The results of the monotherapy and combination arms of the Phase 1 trial will inform us on the next development steps.
CTX-8371 (PD-1 x PDL-1 Bispecific Antibody)
CTX-8371 is a bispecific antibody that simultaneously targetsbinds to both PD-1 and PD-L1, the targets of well-known and widely used checkpoint inhibitor antibodies. SinglePreclinical studies demonstrate that CTX-8371 has the ability to outperform PD-1, PD-L1, and combinations of the two to activate T-cells in in vitro assays. In mouse xenografts, treatment with CTX-8371 led to significantly greater tumor growth control and longer survival than treatment with a PD-1 inhibitor alone, a PD-L1 inhibitor alone or the combination of PD-1 and PD-L1 inhibitors. IND-enabling studies on CTX-8371, including GLP toxicology studies in non-human primates (“NHPs”) were completed in the first quarter of 2023. The FDA cleared the IND for CTX-8371 in the fourth quarter of 2023 and the first patient in the Phase 1 clinical trial is expected to be dosed by early second quarter 2024.
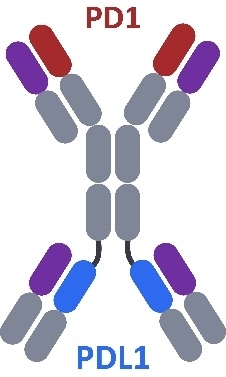
CTX-8371 is a PD-1 x PD-L1 bispecific antibody
Overview of PD-1 and PD-L1 Checkpoint Inhibitors
PD-L1 is a surface protein that is overexpressed by over 35% of certain types of cancer, such as melanoma, hepatocellular carcinoma, colorectal cancer, and NSCLC. Binding of PD-L1 to its receptor, PD-1, on immune T-cells leads to suppression of cytotoxic CD8+ T-cells preventing immune attack of the tumor. Multiple inhibitors of PD-1 orand PD-L1 include some of the highest-revenue-generating therapeutics in history and have been approved as therapies for the treatment of a widebroad range of tumors.tumors including melanoma, NSCLC, small cell lung cancer, head and neck squamous cell cancer, renal cell carcinoma, bladder cancer, gastric cancer, cervical cancer and other cancers with microsatellite instability or mismatch repair deficiency. While PD-1/PD-L1 checkpoint therapies have resulted in remarkable clinical efficacy across multiple cancer types, their efficacy, even in tumors with high immunogenicity, is limited to approximately 20% of patients. Nevertheless, sales of checkpoint therapies in 2022 were estimated to be $34 billion, and grew to $39.8 billion in 2023 at a compounded annual growth rate (CAGR) of 16.4%. The PD-1 and PD-L1 inhibitors market is expected to reach $70 billion in 2027 at a CAGR of 15.1%. There is no marketedapproved therapy that combines inhibition of both PD-1 and PD-L1 in the same moleculemolecule.
Discovery and Preclinical Activity for CTX-8371
The desire to improve the efficacy of PD-1/PD-L1 inhibitors has sparked multiple attempts to create bispecific antibodies in CTX-8371,which one antigen binding site targets PD-1 or PD-L1 and the other targets immuno-oncology receptors such as CTLA-4, LAG-3 or CD137. In contrast to those bispecific efforts described by others that have focused on a single pair of antigen-binding domains at a time, we are developing one. We discovered CTX-8371 usinghave applied our StitchMabsTM technology whento broadly screen for pairs of bispecific antigen-binding domains with the highest potential to generate antitumor activity. Our efforts were enabled not only by the StitchMabsTM technology, but also by our investment in generating a broad portfolio of selective antibodies to approximately 40 potential immune targets across the innate and adaptive immune system.
We designed our combinatorial screen such that one antigen-binding domain was directed against PD-1, and the other selected from our library of candidate antibodies. We screened these StitchMabsTM bispecific constructs in T-cell activation assays in the presence of PD-L1 expressing cells. Our unbiased screening led us to an antibody that pairs a PD-1 binding domain and a PD-L1 binding domain. This novel bispecific antibody contributed to T-cell activation that outperformed the activation observed in response to treatment with only PD-1 antibodies. We designated CTX-8371 as the bispecific antibody we screened forconstructed using our common light chain antibodies having a PD-1 and PD-L1 antigen binding domain.
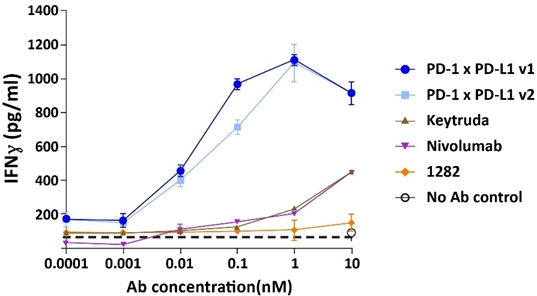
A PD-1 x PD-L1 bispecific antibody outperformed single PD-1 antibodies in a T-cell activation assay
The observation that the bestcombination of a PD-1 and PD-L1 antibody into a bispecific antibody would be hundreds to pair with our proprietarythousands fold more potent in a T-cell activation assay than a PD-1 blocker. Additional studies demonstratedantibody alone was unexpected. A simple model would suggest that inhibiting either PD-1 or PD-L1 should have approximately equal effects in this assay and there would be no advantage to inhibiting both. Further investigation into the mechanism of CTX-8371 works via a novelfound that it led to T-cell activation through four mechanisms:
● | Dual checkpoint blocker: preventing PD-L1 to PD-1 binding, thus relieving the immunosuppressive PD-1 signal; |
● | Cell engager: bridging the connection between the PD-L1 expressing tumor cell and the PD-1 expressing T-cell, potentially facilitating T-cell engagement and enhancement of effector function; |
● | Downregulation of PD-1: triggering the shedding of the extracellular domain of PD-1 receptors from the surface of T-cells resulting in a reduction in the levels of PD-1 on T-cells; and |
● | Indirect CD28 agonist: increasing the pool of free CD80 on tumor cells making it available to bind and activate the CD28 T-cell co-stimulatory receptor, thereby, sending a positive signal to the T-cell, which enhances its activation. |
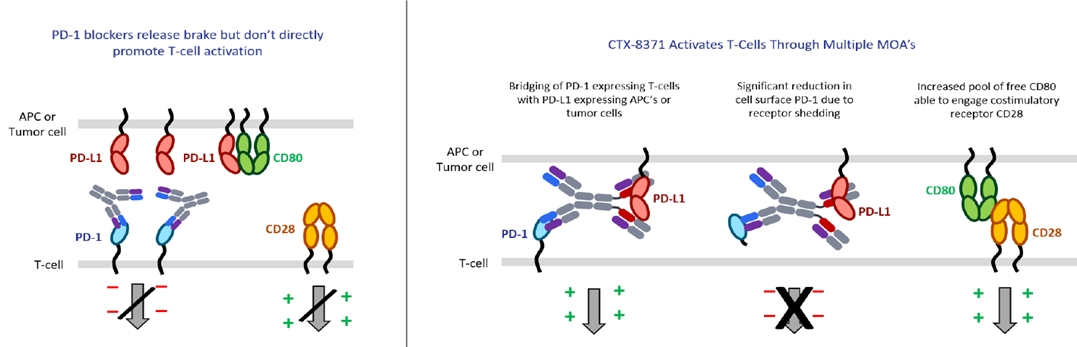
Differentiated mechanism of action not shared by singleof CTX-8371 drives enhanced T-cell activation
We also found that the greater activity of CTX-8371 in our T-cell activation assay compared to PD-1 or singleinhibition also extended to PD-L1 blockers. We have shown in animal models thatinhibition. Furthermore, CTX-8371 was associated with greatersignificantly more antitumor activity in a murine B16F10 melanoma model than was monotherapy with either a single PD-1 inhibitor or a single PD-L1 inhibitor or a combination of both. Tumor growth in monotherapy-treated mice and in the two. IND-enabling studiescombination PD-1 and PD-L1-treated mice was slowed to approximately half that observed with tumors in untreated mice. In contrast, tumor growth was essentially stopped by the CTX-8371 bispecific antibody. Treatment with CTX-8371 resulted in improved overall survival in this model and cured three of eight mice, such that their tumors were initiatedcompletely eradicated.
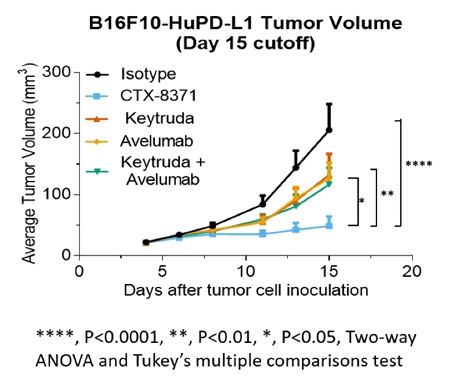
Tumor growth inhibition was improved when treating mice with CTX-8371 compared to treating them with monoclonal antibodies that inhibited either PD-1, PD-L1, or the combination of PD-1 and PD-L1
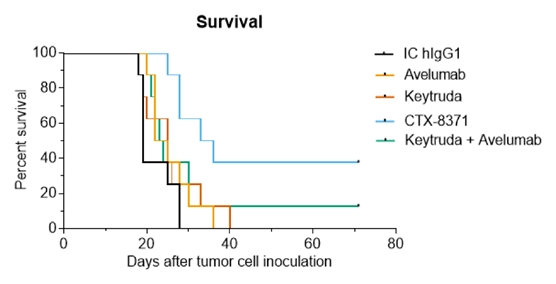
Dosing with CTX-8371 led to improved overall survival in August 2020. Wea B16F10 melanoma model compared to either PD-1 or PD-L1 checkpoint inhibitors or to the combination of both
CTX-8371 also reduced tumor growth in the syngeneic MB49 bladder cancer model and in the syngeneic EMT-6 breast cancer models which are targeting an IND submissionknown to be non-responsive to checkpoint blocker treatments.
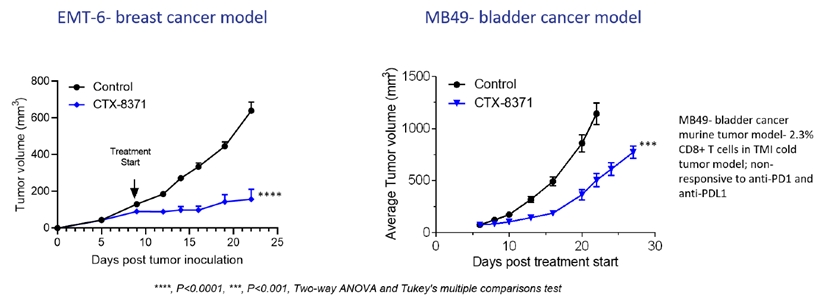
Dosing with CTX-8371 led to tumor growth inhibition in the syngeneic EMT-6 breast cancer model and in the syngeneic MB49 bladder cancer model
In April 2022, we presented preclinical data on CTX-8371’s potential unique mechanism of action ("MOA") that involves cleavage of cell surface PD-1, at the 2022 American Association for Cancer Research ("AACR") annual meeting. A summary of the results are as follows:
● | Treatment with CTX-8371 led to PD-1 loss from the surface of intra-tumoral T cells in tumor-bearing transgenic hPD-1/h-PD-L1 mice, and on peripheral blood T cells in cynomolgus monkeys; this unique MOA differentiates CTX-8371 from marketed inhibitors targeting either PD-1 or PD-L1 |
● | Clearance and half-life of CTX-8371 were within the expected ranges for a human IgG1 antibody in NHPs with a linear PK |
● | Treatment with CTX-8371 in the aggressive MC38-hPD-L1 colorectal mouse model led to a dose-proportional reduction in tumor volume and a complete eradication of tumors at the highest dose |
● | Taken together, the murine and cynomolgus monkey PK data, receptor occupancy data, and in vivo efficacy data in murine models was used to calculate the predicted human efficacious dose range for CTX-8371 |
Development Plans for CTX-8371
An IND was submitted to the FDA in the third quarter of 2023. This IND was cleared by the FDA in October 2023. In the first quarter of 2024 we initiated a first-in-human Phase 1 study of CTX-8371 in patients with metastatic or locally advanced malignancies. This Phase 1 trial is a multiple ascending, dose escalation and there are five doses planned in this study: 0.1, 0.3, 1.0, 3.0 and 10 mg/Kg. Patients eligible to participate in the trial are patients who progressed while receiving an approved PD-1 or PD-L1 inhibitor. Other eligibility criteria include patients with metastatic of locally advanced melanoma, non-small cell lung cancer, head and neck cancer, Hodgkin’s Lymphoma and triple negative breast cancer. The first patient in this Phase 1 clinical trial is expected to be dosed by early 2022 and we could deliver early safety and top-line data later in 2022.second quarter 2024.
6
In addition
Early-Stage Discovery
Traditionally, our approach to CTX-471 and CTX-8371, we are also developing a portfolio of bispecific and monoclonal antibody product candidates, which are currently in preclinical development. These programs all derive from our in-house antibodyearly-stage discovery and development platforms.
Our approach ishas been based on the observation that traditional methods of antibody discovery are slow, inefficient, and are limited by lack of diversity of antigenic sites, or epitopes, that are recognized using these methods. We believe these limitations impair drug developers’ ability to identify the best product candidates. We have created several technological solutions that are designed to address the key challenges in antibody development with the goal of incorporating our solutions into bispecific product candidates. First, we developed and acquired several complementary platforms that enable us to generate antibodies with a high level of epitope diversity and excellent physical and biochemical properties. Second, we have developed sophisticated technologies to screen our antibody sets in functional biological assays designed to prioritize antibodies with desirable biological activities. Third, we have developed our proprietary StitchMabsTM technology that allows us to rapidly evaluate the potential of the antibodies we discover in a bispecific antibody format.
We have also developed a proprietary transgenic mouse line that produces antibodies with the differentiated property that they all share a human common light chain. We imposed this restriction at the earliest stage of our bispecific antibody discovery process in anticipation of the need to simplify the manufacturing of our bispecific product candidates. Sharing a common light chain enables our bispecific antibodies to be manufactured using a well-established process that has been successfully used by the biopharmaceutical industry to produce monoclonal antibodies at commercial scale, thereby avoiding the complexities associated with the manufacture of bispecific products that lack this property. We found that imposing this restriction on the construction of the antibody pool did not hinder our ability to obtain highly potent and selective antibodies.
In addition to our development of antibody product candidates for immuno-oncology, we have also leveraged our proprietary platform technologies to generate and identify monoclonal antibodies and bispecifics that suppress immune response, which we are evaluating as therapies for autoimmune indications. Several of these programs are currently undergoing characterization and in vitro and in vivo testing.
We have recently conducted a review of our pipeline and have made the strategic decision to deprioritize the development of our NKp30 innate cell engager platform. Along with this decision, we have discontinued the efforts to advance CTX-8573 to IND-enabling studies.
Our management team has a successful record of building and growing biotechnology companies. Our Chief Executive Officer and co-founder, Thomas J. Schuetz, M.D., Ph.D. has over 20 years of experience in oncology, biopharmaceutical drug development and life science venture investing. Prior to co-founding Compass Therapeutics, Dr. Schuetz was a venture partner with OrbiMed Advisors LLC where he participated in OrbiMed’s investments in Enobia Pharma (sold to Alexion), Relypsa (sold to Galenica), Arteaus Therapeutics (sold to Eli Lilly), and Audentes (sold to Astellas) and served on the board of each of these companies. Dr. Schuetz was also the chief medical officer of Therion Biologic Corporation and was vice president of clinical affairs at Transkaryotic Therapies, a company acquired by Shire.
Our President and Chief Operating Officer, Vered Bisker-Leib, Ph.D., M.B.A., has over 18 years of experience in strategy, business development, finance and operations of biotechnology and pharmaceutical companies. Prior to joining Compass Therapeutics, she served as an entrepreneur-in-residence with Atlas Venture. Previously, Dr. Bisker-Leib was chief business officer of Cydan, a biotech accelerator, where she co-founded IMARA, Inc. and other biotech companies focused on therapies addressing rare diseases and served as an executive director and global head of business development for the cardiovascular and metabolic franchises of Bristol-Myers Squibb.
Our investors include leading life science venture and institutional funds led by OrbiMed, F-Prime Capital, Cowen Healthcare Investments, Biomatics Capital, Consonance Capital and Borealis Ventures.
7
Pipeline
The figure below details our pipeline of product candidates, including our lead product candidate, CTX-471, our bispecific product candidate, CTX-8371, and our other discovery stage programs.
Our Strategy
Our goal is to expand and extend the role of the immune system in fighting cancer with antibody-based therapies. We believe our approach can be applied both to solid tumors as well as to hematologic malignancies. Our strategy to achieve this goal includes:
Advance our lead product candidate, CTX-471, through clinical development to evaluate its therapeutic potential alone and in combination with other therapies. We seek to translate the antitumor activity of CTX-471 observed in preclinical testing into meaningful clinical results in patients with immunogenic tumors, such as non-small cell lung cancer, or NSCLC, and melanoma. Our ongoing Phase 1 clinical trial is being conducted in patients who relapse or progress after at least 3 months of stable disease on prior checkpoint therapies. We expect to complete the Phase 1b dose expansion stage of the Phase 1 trial during the second half of 2021 and to initiate a Phase 2/3 trial of CTX-471 in the second half of 2022. Pending the results of our Phase 1 monotherapy trial of CTX-471, we plan to initiate a second Phase 1 trial of CTX-471 in combination with trastuzumab or cetuximab, and we could submit an IND application for this combination in the second half of 2021.
Advance CTX-8371 into clinical development as a next generation checkpoint inhibitor. Our bispecific inhibitor that targets PD-1 and PD-L1 has demonstrated higher antitumor activity in preclinical experiments than single PD-1, a single PD-L1, or combinations of PD-1 and PD-L1 inhibitors. IND-enabling studies with CTX-8371 were initiated in August 2020 with the goal of submitting an IND in early 2022 and we could deliver early safety and top-line data later in 2022.
Advance our pre-clinical programs for autoimmune indications. In addition to our development of antibody product candidates for immuno-oncology, we have also leveraged our proprietary platform technologies to generate and identify monoclonal antibodies and bispecifics that suppress immune response. We are in the process of advancing our proprietary monoclonal antibodies and bispecifics through in vitro and in vivo testing and characterization with the goal of identifying at least one clinical candidate that can be advanced into IND-enabling studies in the first half of 2022.
|
|
8
Seek strategic partnerships for select product candidates. Our technology platform is designed to generate a broad pipeline of product candidates with high potential for clinical application. We intend to assess on a case-by-case basis the opportunities for accelerating the preclinical and clinical development of these candidates in a capital-efficient manner, including selectively pursuing strategic partnerships with leading biopharmaceutical companies with domain-specific clinical development expertise to maximize the value of our pipeline.
Our approach
We are focused exclusively on modulation of the immune system through the development of novel antibody therapeutics. Antibodies are structurally distinct Y-shaped proteins formed through the pairing of two long proteins, called heavy chains, and two short proteins, called light chains. Each heavy and light chain pair forms a binding site where the antibody specifically binds its target, which is also known as an antigen.
The immune system is capable of not only fighting foreign invaders, but also of recognizing and eliminating a human body’s own cells that have become pathogenic after transformation, such as in cancer. There are two broad classes of antibodies used in cancer therapy. The majority of antibodies directly target the tumor or its surroundings. The more recent class consists of antibodies that modulate the immune system leading to immune- mediatedimmune-mediated killing of tumors. These antibody drugs mainly exert this effect via a single modulation of the immune system. We believe that modulation of more than one function of the immune system simultaneously has the potential to improve the therapeutic benefit and utility of immuno-oncology therapies.
Antibodies can be generated in many ways, and multiple companies claim to possess proprietary antibody discovery platforms, each with specific advantages. Our antibody platform was designed with a broad set of capabilities and resources that we can leverage with the goal of generating a portfolio of highly distinct bispecific products.
Our approach to bispecific antibody discovery encompasses four principles:
● | antibody diversity is required to generate a representative sample of possible therapies; |
● | functional screening is critical to identifying optimal solutions; |
antibody diversity is required to generate a representative sample of possible therapies;
functional screening is critical to identifying optimal solutions;
a combinatorial approach enables parallel assessment of many potential bispecific antibodies; and
decisions made at the start of the discovery process have a major impact on successful clinical and commercial-scale manufacturing.
● | a combinatorial approach enables parallel assessment of many potential bispecific antibodies; and |
● | decisions made at the start of the discovery process have a major impact on successful clinical and commercial-scale manufacturing. |
Antibody diversity
We obtainobtained our initial pools of antibodies from multiple internally-developed platforms, including our custom phage display library and our transgenic mouse line. We constructed our phage display library based on the peripheral B cell diversity of 70 healthy human donors. This system allows us to generate large and highly diverse sets of antibodies that are fully human; target multiple epitopes on a target of interest; and possess excellent physical and biochemical properties. We describe these antibodies as having good ‘drug-like’ properties. To generate additional antibody candidates, we can also immunize a proprietary line of humanized transgenic mice with antigens of interest to isolate a diverse set of fully human antibodies that share a common human light chain, but distinct native mouse heavy chains. We estimate that the pool of antibodies from these two platforms represents over 1010 unique sequences.
We expresshave expressed libraries of antibodies against any particular target using our Human Display technology which streamlines the expression of functional antibodies such that each cell expresses only one antibody clone. We then further screen our diverse sets of antibodies expressed with our Human Display
9
technology to fine-tune for specificity. Sequence changes can be readily introduced to further optimize leads from our screens.
Our ability to generate viable antibody candidates, with good drug-like properties and high manufacturability potential in a high-throughput manner has enabled us to rapidly assemble a portfolio of proprietary antibodies to over 40 key innate and adaptive immune targets and tumor antigens. This portfolio of antibodies is designed to provide us with a set of well-characterized antibodies that can be incorporated into our combinatorial bispecific antibody screening platform.

Figure 1. We have discovered proprietary antibodies that modulate two key components of the immune system: innate immunity and adaptive immunity as well as antibodies to selected antigens on tumor cells. This expansive collection of antibodies allows us to use our proprietary technologies to uncover unexpected synergies.
Functional screeningScreening
A critical part of our antibody discovery process is our ability to produce sufficient quantities of purified antibodies to assess their biological activities both in cells and, in some cases, animal models. Our human display technology allows us to efficiently express full-length antibodies on cell surface, thereby facilitating the high throughput screening of our antibodies across multiple functional screens. While we do assess standard biochemical parameters such as binding affinity and specificity as part of the initial screening of our candidate sets, we note that activity in a complex biological system cannot be predicted based on physical and biochemical parameters alone. We have shown with CTX-471, for example, that activity in a complex biological system cannot necessarily be predicted on strictly biochemical parameters.
StitchMabsTM technology Technology
A natural antibody recognizes a single target antigen and is therefore monospecific. Because a natural antibody features two identical binding sites, it is considered bivalent for that target. Although natural antibodies recognize a single target antigen, it is possible to engineer antibodies so that their two binding sites bind two different targets. The construction of a bispecific antibody typically requires a significant investment in cloning, construct optimization, protein expression, and protein purification before the therapeutic potential of any particular bispecific antibody can be assessed. In practice, these requirements mean that the diversity of antigen pairs targeted by bispecific antibodies is limited, and development is oftentimes prioritized for antigen pairs suggested by existing scientific literature.
10
Our proprietary StitchMabsTM technology is a novel screening approach which we developed to assess the potential of any pair of antigen-binding sites in a bispecific antibody format. This combinatorial antibody-linking technology stably and irreversibly attaches a second pair of antigen-binding domains to a standard antibody during a 15-minute incubation at room temperature. The resulting stitched antibody acts structurally and functionally like a bispecific antibody.
StitchMabsTM allows us to assess our large library of antigen-binding domains in combinatorial fashion. Once we have generated and purified large numbers of bispecific candidates, we then assess the potential of these candidates in functional assays and determine whether these bispecifics have additive, reductive or synergistic activity. Screening of these bispecific molecules in functional assays has led us to discover novel product candidates with unexpected synergistic activity in cellular and animal models.
Our common light chain platform greatly simplifies manufacturing
Common Light Chain Platform
The embedded common light chain feature in our antibodies greatly simplifies the manufacture of our bispecific product candidates. Most antigen-binding domains of antibodies are composed of a heavy chain and a light chain that have been optimized together to recognize a specific antigen. If these two chains are expressed independently, as is the case with most antibody manufacturing processes, they are often reassembled in various ways, leading to heterogenous mixture of the desired product along with peptide segments corresponding to two heavy chains and two light chains. Separation of the desired product from the mixture is a technically challenging and expensive process. We address this challenge by including only common light chain compatible antibodies as part of our antibody discovery process for potential incorporation into bispecifics. The variability in the antigen-binding domain of our antibodies in the heavy chain is sufficient to generate a diverse, potent, selective, and functionally active set of antibodies. We further simplified the manufacturing of our bispecific antibodies by assembling a single heavy chain construct that encodes both antigen-binding domains. As a result, the manufacturing of our bispecifics closely resembles that of standard monoclonal antibodies, which include—include one heavy chain and one light chain. Our focus on common light chain antibodies simplifies the process of converting our StitchMabsTM screening candidates into bispecific antibody product candidates.
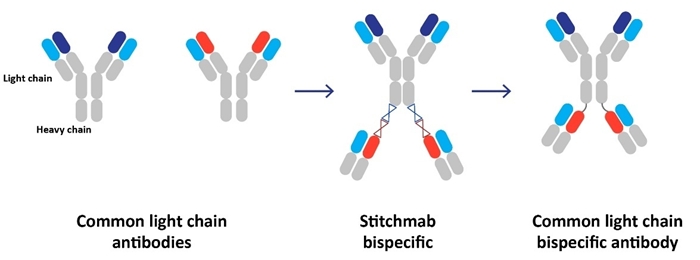
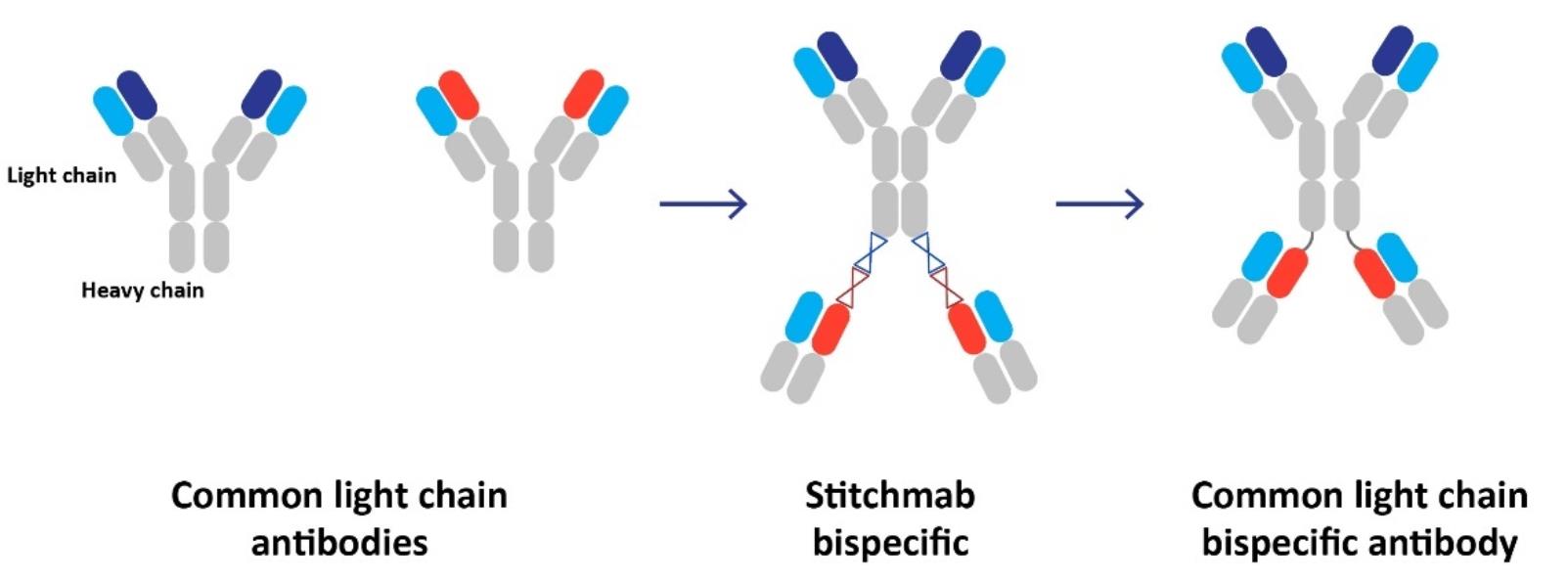
Figure 2. A common light chain simplifies the production of bispecific antibodies
CTX-471,
License Agreements
CTX-009 (DLL4 X VEGF-A bispecific antibody)
Our wholly owned subsidiary Trigr Therapeutics, Inc. (“TRIGR”) and ABL Bio, a monoclonalSouth Korean biotechnology company, entered into an exclusive global (excluding South Korea) license agreement (the “TRIGR License Agreement”) which granted TRIGR a license to ABL001, ABL Bio’s bispecific antibody targeting CD137
CTX-471, our monoclonal antibody product candidate, is an agonist of CD137, a key co-stimulatory receptor on immune cells. Binding of CTX-471 to CD137 leads to ligand-stimulated activation of T-cellsDLL4 and NK cells. In tumor models, treatment with CTX-471 as a monotherapy led to recruitment and activation of immune cells inVEGF-A (renamed CTX-009). Under the tumor microenvironment. In the treated mice, dosing with CTX-471 led to extensive reprogrammingterms of the tumor microenvironment, including increased recruitmentagreement, ABL Bio and TRIGR would jointly develop CTX-009, with ABL Bio responsible for development of immune cells, reversionCTX-009 throughout the end of exhausted cytotoxic CD8+ T-cells, reductions in immunosuppressive regulatory T-cells, and reductions in immunosuppressive tumor- associated macrophages. Long after the completion of the
11
treatment with CTX-471, a period described as eight half-lives of the antibody, treated mice exhibited immune memory that prevented reestablishment of the same tumor.
In July 2019, we initiated a Phase 1 trial evaluating CTX-471 as a monotherapy in oncology patients who were previously treated with a PD-1 or PD-L1 immune checkpoint inhibitorclinical trials and subsequently relapsed or progressed after a period of stable disease. The design of this trial includes a Phase 1a dose escalation stage followed by a Phase1b dose expansion stage. The Phase 1a dose escalation stage has been completed and the Phase 1b dose expansion stage is ongoing.
Overview of NSCLC
According to the World Health Organization, an estimated 1.8 million people worldwide die of lung cancer each year, which accountsTRIGR responsible for approximately 18% of all cancer deaths, making lung cancer the leading cause of cancer-related death. In the United States, there are an estimated 228,000 newly diagnosed cases of lung cancer and approximately 137,000 deaths annually. NSCLC accounts for approximately 80%-85% of lung cancer cases. The treatment paradigm for NSCLC has significantly changed over the past few years. Previously, patients were primarily treated with radiation therapy or combinations of cytotoxic drugs. Recent advancements have led to the development of targeted therapies basedCTX-009 from Phase 2 and onward.
ABL Bio received a $5 million upfront payment and a $6 million milestone payment. In addition, ABL Bio is eligible to receive up to $96 million of development and regulatory milestone payments, up to $303 million of commercial milestone payments and tiered single-digit royalties on blockadenet sales of alterationCTX-009 in mutated genes, such as EGFR, anaplastic lymphoma kinase gene, or ALK, ROS1 or BRAF. UpOncology. ABL Bio is also eligible to two thirdsreceive up to $75 million in development and regulatory milestone payments and up to $110 million in commercial milestone payments and tiered, single-digit royalties on net sales of advanced or metastatic NSCLC patients who are ineligible for or resistantCTX-009 in Ophthalmology. The financial terms of the agreement were amended in May 2021 but remain substantially similar to treatment with targeted therapies have tumors that express PD-L1 and are candidates for checkpoint inhibitor therapies, which lead to significant improvements in progression free survival and overall survival compared to standard chemotherapy. Despite the availability of these therapies, the prognosis in NSCLC remains poor, with an overall five-year survival for all patients diagnosed with NSCLC of 19%. In the KEYNOTE-042 trial in treatment naïve metastatic NSCLC patients, conducted by Merck from December 2014 to March 2017, treatment with pembrolizumab as monotherapy led to partial responses in 27% of patients and complete responses in 0.5%. The duration of responseterms in the majorityTRIGR License Agreement. As a result of the patients was less than one year. We believe there remains significant unmet medical need in this patient population that could be addressed with novel antibody therapeutics.
Role of CD137 in immunology
CD137, also known as 4-1BBTRIGR acquisition, we have assumed all the rights and TNFRSF9, is an inducible co-stimulatory receptor expressed on T-cells and NK cells. Activation of CD137 triggers a signaling cascade that results in upregulation of antiapoptotic molecules, cytokine secretion and enhanced cell killing function. On NK cells, CD137 signaling can increase antibody-dependent cell-mediated cytotoxicity, or ADCC.
When antigen-presenting cells, such as dendritic cells, express CD137L or 4-1BBL, the natural ligand for CD137, they induce increases in the levels of CD137 on T-cells. Tumors with a high tumor mutation burden are enriched in these antigen-presenting cells and such tumors represent promising opportunities to improve on standard of care checkpoint inhibitors by adding antibody therapies directed against CD137.
Historically, across preclinical cancer models, agonist antibodies targeting CD137 have been immunotherapeutic agents that showed great promise. In the clinic, however, these agents have been hampered, in part by dose-limiting toxicities, as seen with urelumab, and, in part by weak agonist activity, as seen with utomilumab.
Our product candidate, CTX-471
CTX-471 is a fully human, IgG4 monoclonal antibody that is an agonistobligations of the CD137 receptor. We selected CTX-471 from among a panel of CD137 antibodies based on multiple preclinical parameters. The CD137 antigenic site recognized by CTX-471 does not block the binding of CD137 ligand and is differentiated from the site recognized by CD137 antibodies from competitors. We designed and made the antibody using different backbones and chose to use a human IgG4 backbone for CTX-471 to enable
12
engagement of Fc receptors FcgRI and FcgRIIb to facilitate CD137 cross-linking while avoiding binding to FcgRIIIa and depletion of immune effector cells through ADCC.
Identification through functional screening
We evaluated a panel of anti-CD137 antibodies as potential candidates for CTX-471 and used a series of in vitro and in vivo functional assays to screen for the best candidate. One of the most stringent assays was antitumor activity in a CT26 mouse colon carcinoma model in which tumors were allowed to grow to 500 mm3 before CTX-471 candidates were administered. Tumors of this size are generally considered futile to treat and are highly resistant to monotherapy with other immuno-oncology therapies such as checkpoint inhibitors.TRIGR License Agreement.
Figure 3. Preclinical antitumor activity evaluation of CTX-471 was conducted in syngeneic mice with 500 mm3 CT26 tumors
We observed that multiple CTX-471 candidates exhibited activity treatments in this model, leading to the complete eradication of these large tumors when dosed as monotherapy. Certain antibody candidates exhibited greater activity than others and there was not a strict correlation between potency for the CD137 antigen and antitumor activity. We selected the antibody candidate that became CTX-471 based on a combination of in vivo and in vitro properties. We also tested antibodies that target PD-1, PD-
13
L1, CTLA-4 and OX-40 in the CT26 model alongside CTX-471 and observed that these antibodies failed to generate similar responses in this model.
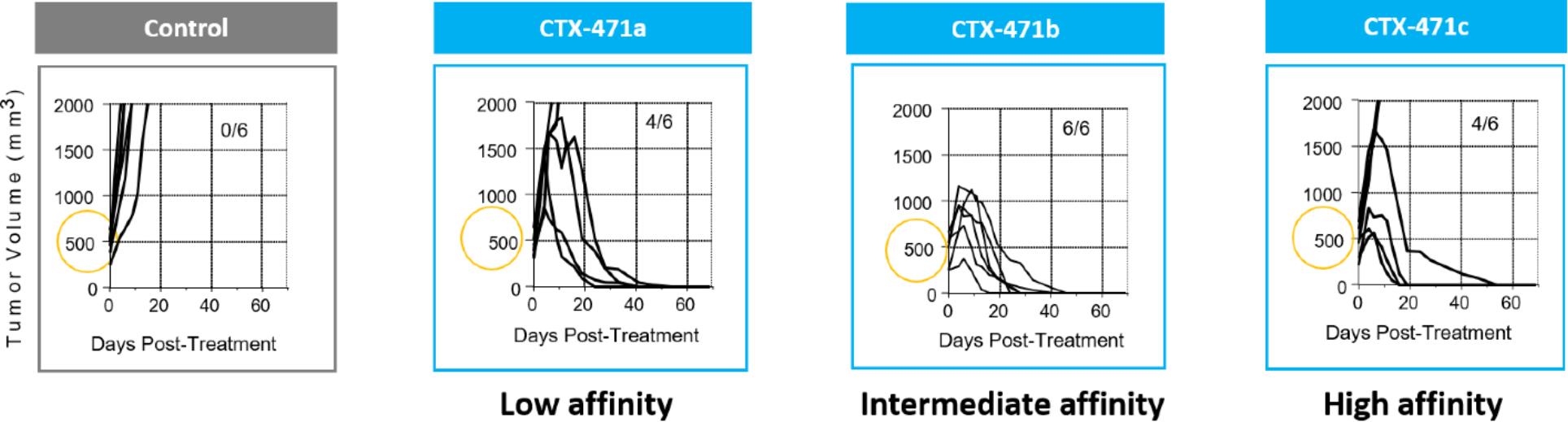
Figure 4. CTX-471a, CTX-471b and CTX-471c are three CD137 agonistic antibodies with low, intermediate and high affinity to CD137 respectively. Four groups of CT26 syngeneic mice models were dosed with control antibody, CTX-471a, CTX-471b and CTX-471c. Treatment initiated when tumors reached 500 mm3. CTX-471b exhibited the most activity, eradicating tumors in 6/6 mice, followed by CTX-471a and CTX-471c, which eradicated tumors in 4/6 mice each, and none in the control group.
Immunoregulatory role
Treatment of mice with CTX-471 stimulated long-term immunological memory. In order to assess the long-term immunological memory, we tested tens of mice cured of their initial tumors by CTX-471 monotherapy to a re-challenge with the same tumor. Upon a re-challenge, these mice have all demonstrated resistance to establishment of new tumors. To investigate whether this observed effect may be explained by residual CTX-471, we have conducted some of these re-challenge experiments 88 days after dosing, or greater than eight half-lives of CTX-471. We believe that, in mice previously cured of CT26 tumors by CTX-471, the inability to establish CT26 tumors is consistent with the ability of CTX-471 to induce long-term immune memory capable of rejecting the reintroduced tumor cells.
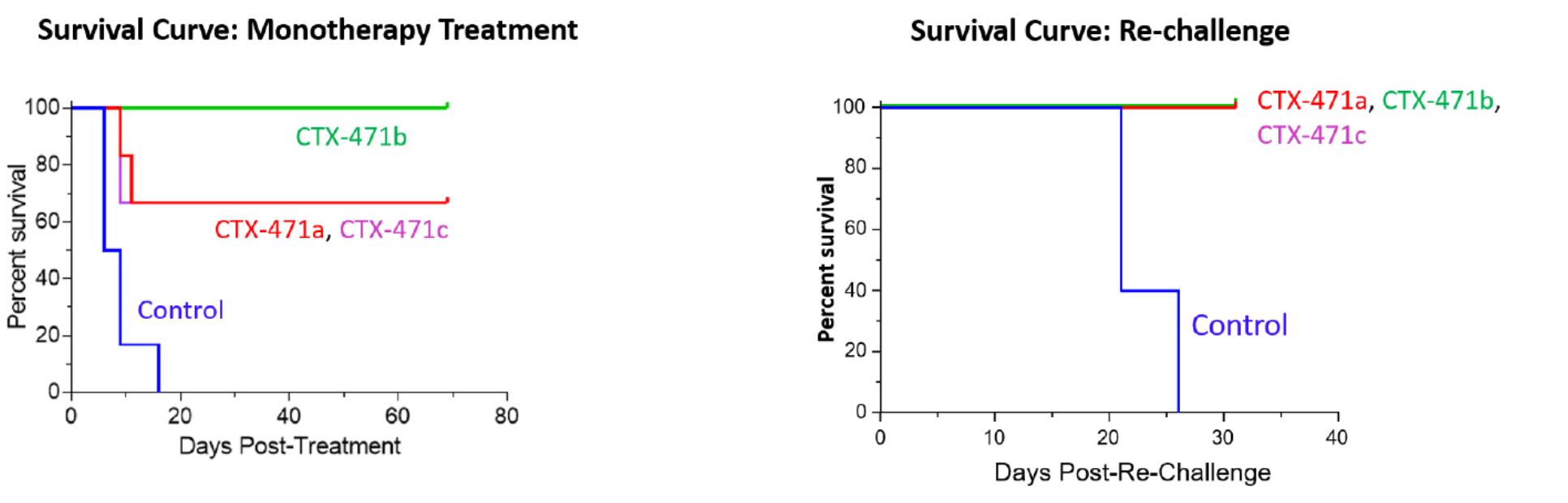
Figure 5. All mice cured by CTX-471 treatment were resistant to re-challenge with the same tumor
Immune cell depletion experiments showed that the activity of CTX-471 required the presence of CD4+ T-cells, CD8+ T-cells, and NK cells, indicating a coordinated involvement of both innate and adaptive immune cells. Encouragingly, treatment of tumors in mice with CTX-471 led to a marked reprogramming of the immune component of the tumor microenvironment. CTX-471-treated mice had over three times more tumor infiltrating immune cells than control mice. Of the CD8+ T-cells in these tumors, there was a reduction of exhausted T-cells, determined by the reduction of CD8+ T-cells that express both PD-1 and TIGIT, from 43% to 8%. Similarly, treatment with CTX-471 led to a sharp decline
14
in immunosuppressive regulatory T-cells, or Tregs, from 31% to 7%. We also observed that tumors treated with CTX-471 had an approximate two-fold reduction in the number of immunosuppressive tumor-associated macrophages.
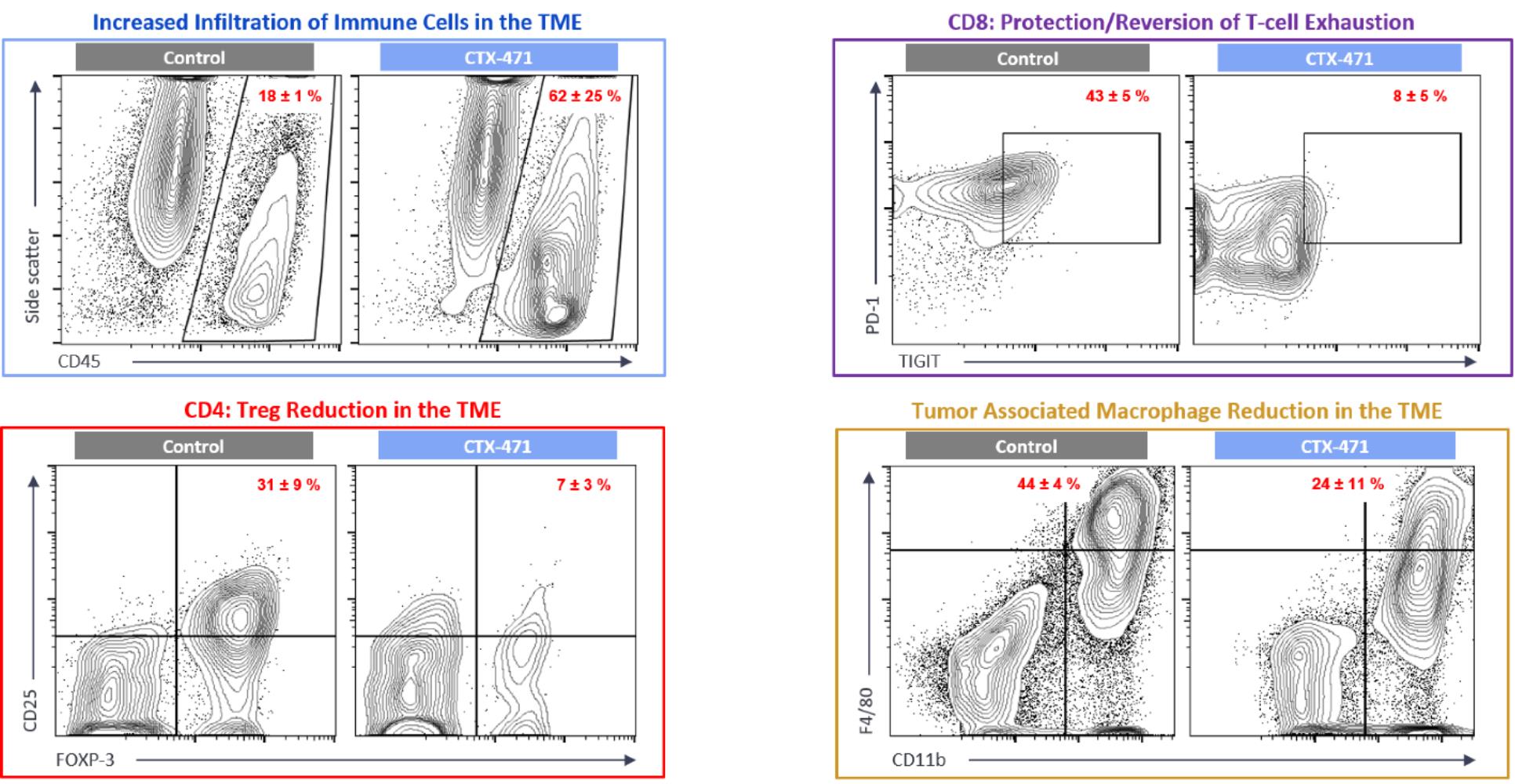
Figure 6. CTX-471 altered the immune composition in the tumor microenvironment
We believe that the ability of CTX-471 to transform the tumor microenvironment through the combined action of immune cell recruitment, alleviation of T-cell exhaustion, suppression of Tregs, and reduction of tumor suppressing macrophages is what drives CTX-471’s antitumor activity in mouse models. We believe that CTX-471 has the potential to affect the same aspects of the immune system in cancer patients in the clinic, which could lead to improved patient outcomes.
In addition to testing CTX-471 in the CT26 syngeneic mouse model described above, we have also tested CTX-471 as a monotherapy in multiple other syngeneic tumor models of different histologies and have observed potent activity, including various levels of tumor eradication. CTX-471 has demonstrated activity and led to tumor eradication in the A20 model of lymphoma, the MC38 model of colon carcinoma, and in the EMT6 model of breast cancer. We believe that this broad biological activity across multiple tumor models of different histologies suggests that CTX-471 might benefit patients with different tumor types.
Phase 1 clinical trial of CTX-471
We are currently conducting a Phase 1 clinical trial of CTX-471 in adult patients who have achieved three months of stable disease or better after treatment with PD-1/PD-L1 checkpoint therapy and who have subsequently relapsed or progressed. We selected this population of patients for this trial because multiple clinical trials and meta-analyses have shown that not all patients respond to checkpoint inhibitor therapy due to many possible reasons. By focusing on those that did previously respond to checkpoint inhibitor therapy, we believe that this trial design enriches for patients who have tumors that are capable of being recognized and killed by their immune systems. We believe that disease progression after the initial checkpoint inhibitor response is likely due to an increase in immunosuppressive activity that CTX-471 has the potential to overcome.
This Phase 1 trial is an open-label multiple ascending dose, dose-escalation trial. After a period of 28 days to allow checkpoint inhibitors and other drugs to be eliminated from the body, each patient
15
receives CTX-471 by intravenous infusion every two weeks as monotherapy. Disease progression is measured by CT scans every eight weeks. We collect blood samples to assess standard safety biomarkers as well as cytokines and potential pharmacodynamic biomarkers. Baseline tumor biopsies are also collected for retrospective analyses.
The primary objective of the Phase 1a dose escalation stage of the trial was to assess the safety and tolerability of CTX-471 monotherapy at various doses. The goal of the Phase 1b dose expansion stage is to determine an optimized dose for future clinical trials. Secondary endpoints include measures of overall response rate and progression-free survival, among others.
Dosing strategy
In contrast to dosing strategies for other immuno-oncology antibodies, such as checkpoint inhibitors where the goal is often to deliver a dose that is capable of fully inhibiting the receptor at all times, our dose selection for this trial is aimed at binding to only a fraction of the available CD137 receptors. Dosing of an agonist antibody, such as CTX-471, at levels capable of binding to the majority of receptors can lead to inappropriate cell activation and downregulation of the receptor and overall weaker activity.
Agonist antibodies typically trigger their activity through independent binding of each of their two antigen-binding domains to individual receptors on a cell surface. This binding to both receptors at once forces the receptorsentered into close physical proximity. This grouping of receptors that drives receptor activation, especially when the ratio of antibody molecules to receptor molecules is relatively low. As the ratio of antibody to receptor increases, the level of receptor activation increases up to a point above which activation may decrease due to down-regulation of the receptors. This results in a bell-shaped activation curve in which maximal activation occurs at intermediate antibody concentrations.
We observed evidence of the importance of lower receptor occupancy while screening candidate antibodies against CD137. The antibodies with the greatest tumor-killing activity were the ones with intermediate potency. Very high-potency antibodies had weaker antitumor activity.
Consistent with the finding of lower activity at high antibody to receptor levels, we observed that the antitumor activity of CTX-471 appeared to peak at doses between 50 ug and 100 ug in the mouse CT26 tumor model. At the higher dose of 200 ug, the number of complete responses, four out of eight mice, was less than that observed at 100 ug, seven out of eight mice, suggesting that the optimal receptor occupancy had been exceeded. This is also consistent with our observation that intermediate affinity antibodies exhibited greater antitumor activity compared to high affinity antibodies.
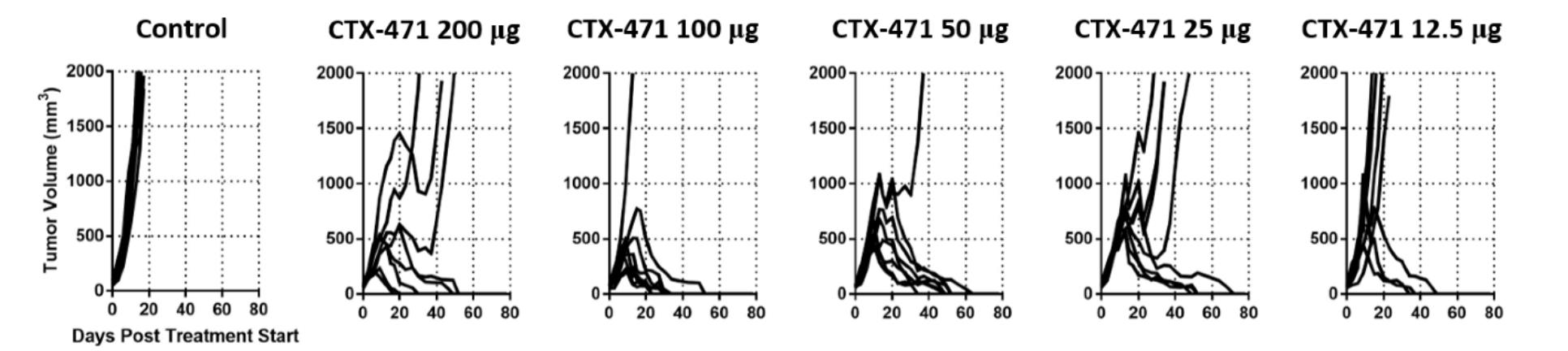
Figure 7. Antitumor activity of CTX-471 is optimized at intermediate dose and decreased at the highest dose level
Our findings are consistent with those reported for an agonist antibody against OX40, another immune target in oncology. Thus, for many agonist antibodies, it is likely that both intermediate affinities and intermediate doses will deliver optimal activity.
16
Phase 1 clinical trial data
We are conducting a Phase 1 trial of CTX-471 in patients with metastatic or locally advanced solid tumors whose tumors have progressed while receiving an approved PD-1 or PD-L1 inhibitor after a period of stable disease. The design of this trial includes a dose escalation stage (Phase 1a) followed by a dose expansion stage (Phase 1b). Our selection of doses in the Phase 1a stage of the trial was informed by multispecies pharmacokinetics and by the intent to select doses capable of maintaining receptor occupancy between 20% and 80% in tumors.
Phase 1a - Dose Escalation
In the Phase 1a dose escalation stage, 19 patients received CTX-471 in the four dosing cohorts set forth in Figure 8 below.
Cohort |
| Cohort 1 0.1 mg/kg |
|
| Cohort 2 0.3 mg/kg |
|
| Cohort 3 0.6 mg/kg |
|
| Cohort 4 1.2 mg/kg |
|
| Total |
| |||||
Enrolled |
|
| 3 |
|
|
| 3 |
|
|
| 6 |
|
|
| 7 |
|
|
| 19 |
|
Figure 8. The number of patients dosed with CTX-471 in the Phase 1a dose escalation stage
CTX-471 was observed to be generally well-tolerated in the Phase 1a stage of the trial. There were two SAEs determined to be treatment-related, which included one hypoxia event that resolved with approximately one day of supplemental oxygen therapy and one immune thrombocytic purpura event that also resolved. The dose-limiting toxicities were two events of thrombocytopenia in Cohort 4, which was expanded from three to six patients to collect additional safety data. Based on these results, 0.6 mg/kg was determined to be the maximum tolerated dose.
While the goal of the Phase 1a stage of the trial was to evaluate the safety and tolerability of CTX-471, we also collected data from these patients to evaluate the pharmacokinetics of CTX-471, the potential development of anti-drug antibodies of CTX-471, and to obtain certain efficacy data, based on the objective response rate, or ORR, as per Response Evaluation Criteria in Solid Tumors, RECIST. Patients who were enrolled in the Phase 1a stage of the trial have been evaluated every eight weeks by imaging techniques, such as MRI or CT, until disease progression or withdrawal from the trial, in order to collect such data.
The Phase 1a stage of the trial is now complete. None of the patients enrolled in the Phase 1a stage of the trial had a complete response or a partial response by RECIST. The best overall response has been stable disease. Two patients with NSCLC had stable disease until progression at Weeks 25 and 41, respectively. In addition, one patient with metastatic melanoma of mucosal origin had an approximately 24% decline in the total size of his measured metastatic tumors (target lesions) at week 33. This patient remained on study and received CTX-471 for 49 weeks.
We have analyzed preliminary pharmacokinetic data from the trial and these data have confirmed our receptor occupancy modeling. Based on this modeling and the correlation of the observed pharmacokinetics with our predictions, we selected 0.3 mg/Kg and 0.6 mg/Kg as the doses of the cohorts of our Phase 1b stage of the trial. We estimate that a dose of 0.3 mg/kg would lead to a peak receptor occupancy of approximately 50% and a dose of 0.6 mg/kg would lead to a peak receptor occupancy of approximately 70%.
Phase 1b - Dose Expansion
In September 2020, we enrolled the first patient in the Phase 1b dose expansion stage of the trial. As of February 28, 2021, 11 patients have received at least one dose of CTX-471. Of the 11 patients treated so far, six patients have reached their first tumor evaluation at week 9, of which five had stable disease. Subsequently, one of those patients who has advanced small cell lung cancer had a partial response at Week 17. As of February 28, 2021, there have been no treatment-related SAEs in the Phase
17
1b dose expansion stage of the trial. This Phase 1b dose expansion stage of the trial is intended to inform the Phase 2 recommended dose.
Subject to the results of our Phase 1 monotherapy trial of CTX-471 and the receipt of additional funding, we also plan to initiate a second Phase 1 trial of CTX-471 in combination with trastuzumab, marketed as Herceptin® by Genentech, in Her2 positive tumors, and with cetuximab, marketed as Erbitux® by Eli Lilly, in EGFR positive tumors.
Potential market opportunity for CTX-471
In preclinical studies, CTX-471 was associated with antitumor activity as a monotherapy in multiple syngeneic tumor models, including colon carcinoma, lymphoma, and breast cancer. This broad biological activity suggests that CTX-471 may have benefit as a therapy for patients with different tumor types.
We seek to maximize the potential value of each of our product candidates, if any, across all indications in which it may demonstrate promising clinical results and receives marketing approval. While we have not selected a specific target indication for CTX-471, as an example for the potential size of the market opportunity for CTX-471 in one of those potential target indications, we have modeled the positioning of CTX-471 as a second line therapy for advanced/metastatic NSCLC.
In the United States, there are 228,000 lung cancer patients each year, of those 80-85% have NSCLC. Patients with stage 0-2 NSCLC are treated with surgery or a combination of surgery and chemotherapy, which are generally effective. However, some patients will progress to the later stages of the disease, and other patients already have locally advanced or metastatic disease at the time of diagnosis. There are approximately 90,000 patients with advanced/metastatic NSCLC per year who are in great need of pharmacological treatment.
In the 1st line setting, the majority of the advanced/metastatic NSCLC patients without defined point mutations are treated by either PD-1 blocker alone or PD-1 blocker combination with chemotherapy, depending on PD-1 expression levels. Patients who do not respond to the 1st line settings have very limited therapeutic options, mostly comprising chemotherapy combinations, with or without checkpoint blockers. We estimate that there are approximately 36,000 patients in this category who will progress after 1st line treatment to 2nd line setting as depicted in the diagram below.

Figure 9. Estimate of the number of treatment-eligible advanced/metastatic NSCLC patients based on NCCN guidelines and other Compass analysis.
CTX-8371, a PD-1 x PD-L1 bispecific antibody
CTX-8371 is a bispecific antibody that binds to both PD-1 and PD-L1. Preclinical studies demonstrate that CTX-8371 has the ability to outperform PD-1, PD-L1, and combinations of the two to
18
activate T-cells in in vitro assays. In mouse xenografts, treatment with CTX-8371 led to significantly greater tumor growth control and longer survival than treatment with a PD-1 inhibitor alone, a PD-L1 inhibitor alone or the combination of PD-1 and PD-L1 inhibitors. IND-enabling studies on CTX-8371 were initiated in August 2020.
Figure 10. CTX-8371 is a PD-1 x PD-L1 bispecific antibody
Overview of PD-1 and PD-L1 checkpoint inhibitors
PD-L1 is a surface protein that is overexpressed by over 35% of certain types of cancer, such as melanoma, hepatocellular carcinoma, colorectal cancer, and NSCLC. Binding of PD-L1 to its receptor, PD-1, on immune T-cells leads to suppression of cytotoxic CD8+ T-cells preventing immune attack of the tumor. Multiple inhibitors of PD-1 and PD-L1 have been approved as therapies for a broad range of tumors including melanoma, NSCLC, small cell lung cancer, head and neck squamous cell cancer, renal cell carcinoma, bladder cancer; gastric cancer, cervical cancer; and other cancers with microsatellite instability or mismatch repair deficiency. While PD-1/PD-L1 checkpoint therapies have resulted in remarkable clinical efficacy across multiple cancer types, their efficacy, even in tumors with high immunogenicity, is limited to approximately 20% of patients. Nevertheless, sales of checkpoint therapies in 2019 were estimated to be $22 billion. There is no approved therapy that combines inhibition of both PD-1 and PD-L1 in the same molecule.
Discovery and preclinical activity of CTX-8371
The desire to improve the efficacy of PD-1/PD-L1 inhibitors has sparked multiple attempts to create bispecific antibodies in which one antigen binding site targets PD-1 or PD-L1 and the other targets immuno-oncology receptors such as CTLA-4 or LAG-3. In contrast to those bispecific efforts described by others that have focused on a single pair of antigen-binding domains at a time, we have applied our StitchMabsTM technology to broadly screen for pairs of bispecific antigen-binding domains with the highest potential to generate antitumor activity. Our efforts were enabled not only by the StitchMabsTM technology, but also by our investment in generating a broad portfolio of selective antibodies to 40 potential immune targets across the innate and adaptive immune system.
We designed our combinatorial screen such that one antigen-binding domain was directed against PD-1, and the other selected from our library of candidate antibodies. We screened these StitchMabsTM bispecific constructs in T-cell activation assays in the presence of PD-L1 expressing cells. Our unbiased screening led us to an antibody that pairs a PD-1 binding domain and a PD-L1 binding domain. This novel bispecific antibody contributed to T-cell activation that outperformed the activation observed in response
19
to treatment with PD-1-only antibodies. We designated CTX-8371 as the bispecific antibody we constructed using our common light chain antibodies having a PD-1 and PD-L1 antigen binding domains.
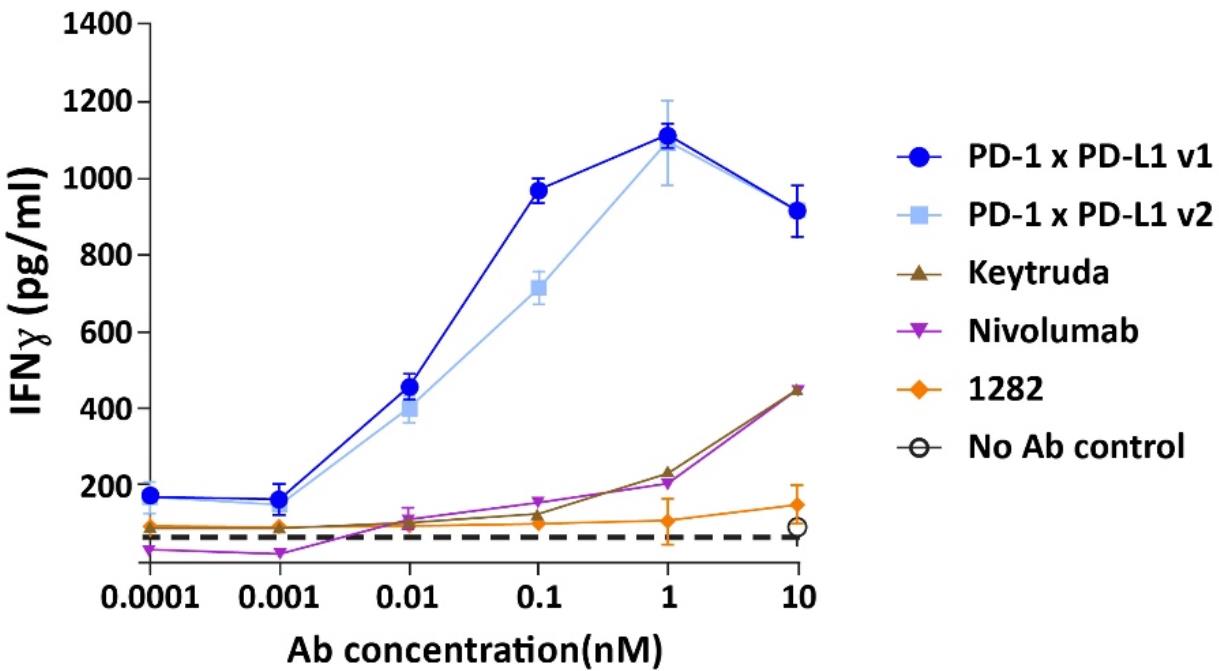
Figure 11. A PD-1 x PD-L1 bispecific antibody outperformed single PD-1 antibodies in a T-cell activation assay
The observation that the combination of a PD-1 and PD-L1 antibody into a bispecific antibody would be hundreds to thousands fold more potent in a T-cell activation assay than a PD-1 antibody alone was unexpected. A simple model would suggest that inhibiting either PD-1 or PD-L1 should have approximately equal effects in this assay and there would be no advantage to inhibiting both. Further investigation into the mechanism of CTX-8371 found that it led to T-cell activation through four mechanisms:
Dual checkpoint blocker: preventing PD-L1 to PD-1 binding, thus relieving the immunosuppressive PD-1 signal;
Cell engager: bridging the connection between the PD-L1 expressing tumor cell and the PD-1 expressing T-cell, potentially facilitating T-cell engagement and enhancement of effector function;
Downregulation of PD-1: triggering the shedding of the extracellular domain of PD-1 receptors from the surface of T-cells resulting in a reduction in the levels of PD-1 on T-cells; and
Indirect CD28 agonist: increasing the pool of free CD80 on tumor cells making it available to bind and activate the CD28 T-cell co-stimulatory receptor, thereby, sending a positive signal to the T-cell, which enhances its activation.
20
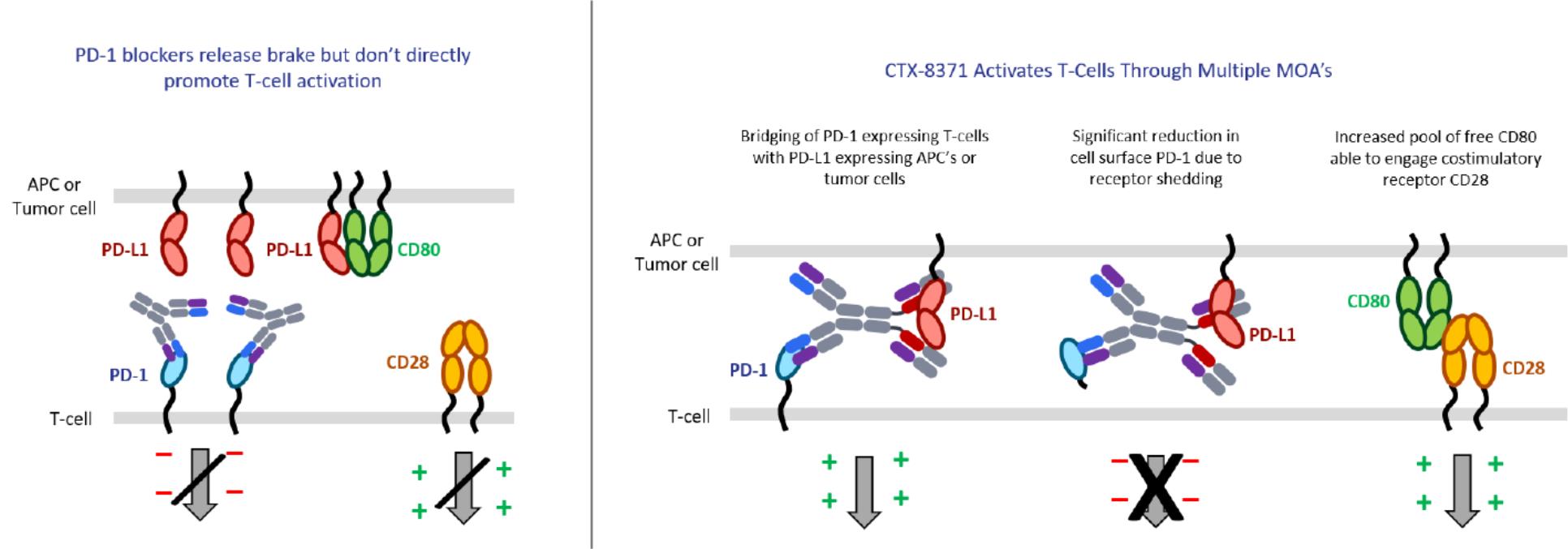
Figure 12. Differentiated mechanism of action of CTX-8371 drives enhanced T-cell activation
We also found that the greater activity of CTX-8371 in our T-cell activation assay compared to PD-1 inhibition also extended to PD-L1 inhibition. Furthermore, CTX-8371 was associated with significantly more antitumor activity in a murine B16F10 melanoma model than was monotherapy with either a PD-1 inhibitor or a PD-L1 inhibitor or combination of both. Tumor growth in monotherapy-treated mice and in the combination PD-1 and PD-L1-treated mice was slowed to approximately half that observed with tumors in untreated mice. In contrast, tumor growth was essentially stopped by the CTX-8371 bispecific antibody. Treatment with CTX-8371 resulted in improved overall survival in this model and cured three of eight mice, such that their tumors were completely eradicated.

Figure 13. Tumor growth inhibition was improved when treating mice with CTX-8371 compared to treating them with monoclonal antibodies that inhibited either PD-1, PD-L1, or the combination of PD-1 and PD-L1
21

Figure 14. Dosing with CTX-8371 led to improved overall survival in a B16F10 melanoma model compared to either PD-1- or PD-L1- checkpoint inhibitors or to the combination of both
CTX-8371 also reduced tumor growth in the syngeneic MB49 bladder cancer model and in the syngeneic EMT-6 breast cancer models which are known to be non-responsive to checkpoint blocker treatments.
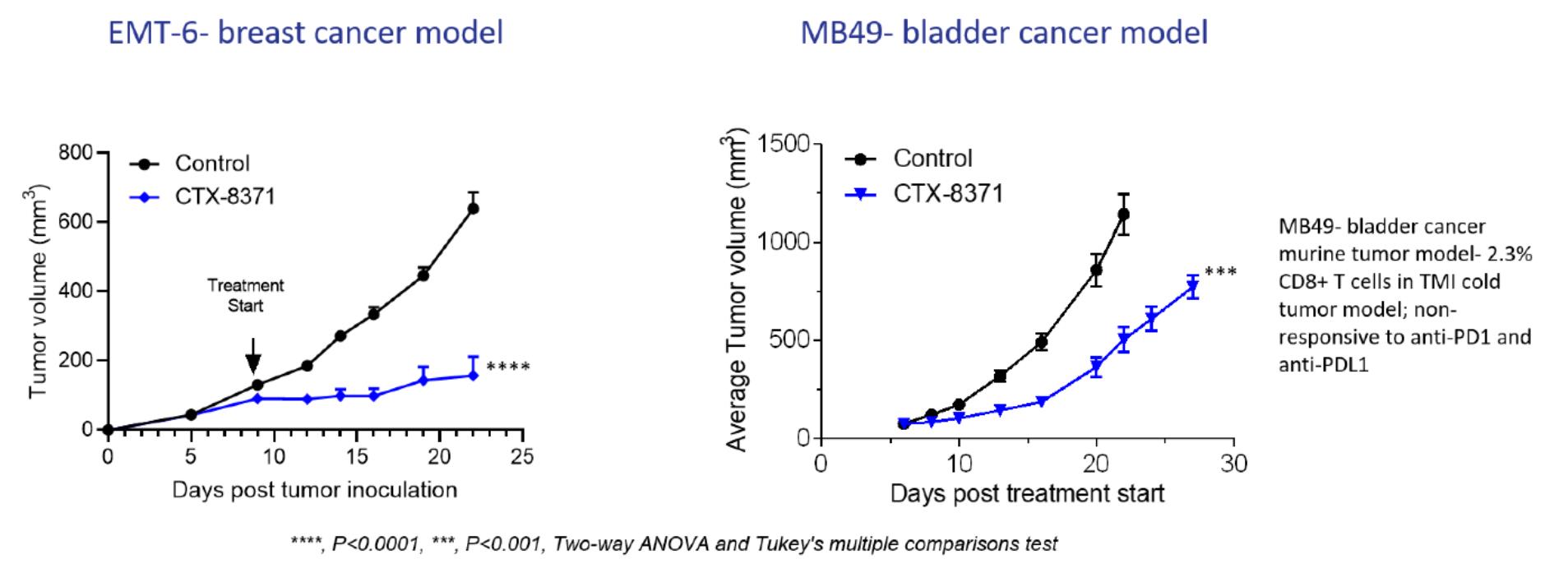
Figure 15. Dosing with CTX-8371 led to tumor growth inhibition in the syngeneic EMT-6 breast cancer model and in the syngeneic MB49 bladder cancer model
IND-enabling studies with CTX-8371 were initiated in August 2020 with the goal of initiating its clinical testing in the second half of 2021 following submission and acceptance of an IND.
License Agreement
We are successor to an amended and restated collaboration agreement with Adimab, LLC or Adimab,("Adimab"), dated February 11, 2015, as amended. This agreement relates to our license from Adimab for certain antibodies for development and commercialization as biopharmaceutical products, including our lead product candidate, CTX-471. We were granted an exclusive option to license antibodies under the agreement, which we exercised with respect to CTX-471, through which we gained an exclusive license to certain Adimab patents and know-how related to CTX-471. We are required to use commercially reasonable efforts to develop, seek marketing authorization for, launch and commercialize the licensed antibody. We are required to make payments upon achievement of development milestones that, as of December 31, 2020,2023, amounted to $3.5 million, of which we have already made $1.5 million in milestone payments, and we have additional potential payments due in the amount of $2.0 million. In addition, we
22
are required to pay royalties at rates ranging in the single digits as a percentage of future net sales within a specified term from the first commercial sale.
The agreement will expire on a country-by-country basis on the expiration of the last royalty term for a product in the particular country, which commences from the first commercial sale of such product in such country until the twelve-year anniversary of such sale, in which case the license for any licensed antibody will automatically convert to be perpetual, irrevocable, non-exclusive and fully-paid in such country. The agreement may also be terminated by the parties for uncured material breach by the other party, and we may also terminate the agreement upon three months prior written notice to Adimab.
Intellectual Property
Overview
We strive to protect the proprietary technology, inventions, and know-how to enhance improvements that are important to the development of our business, including seeking, maintaining, and defending patent rights. We also rely on trade secrets and know-how relating to our proprietary technology platform, on continuing technological innovation and on in-licensing opportunities to develop, strengthen and maintain the strength of our position in the field of antibody therapeutics that may be important for the development of our business. We additionally may rely on regulatory protection afforded through data exclusivity, market exclusivity and patent term extensions where available.
Our success depends in part on our ability to: obtain and maintain patent and other protections for commercially important technology, inventions and know-how related to our business; defend and enforce our patents; preserve the confidentiality of our trade secrets; and operate without infringing the valid enforceable patents and intellectual property rights of third parties.
Our ability to stop third parties from making, using, selling, offering to sell, or importing our products depends in large part on the extent to which we have rights under valid and enforceable licenses, patents, or trade secrets that cover these activities. In some cases, these rights may need to be enforced by third-party licensors. With respect to company-owned intellectual property, we cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents that may be granted to us in the future will be useful in protecting our commercial products and methods of manufacturing the same. For more information, please see “Risk Factors—Risks Related to Our Intellectual Property.”
We seek to protect our proprietary position by, among other things, filing patent applications in the United States and internationally in certain jurisdictions where it is available. For example, we file U.S. and selected foreign patent applications related to our proprietary technology, inventions, and improvements that are important to the development of our business. We also intend to seek patent protection, or rely upon trade secret rights, to protect other technologies that may be used to discover and validate targets and that may be used to identify and develop novel products or improvements thereof. We seek protection, in part, through confidentiality and proprietary information agreements.
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries in which we file, the patent term is 20 years from the earliest date of filing a non-provisional application which matures into a granted patent. A U.S. patent also may be accorded a patent term adjustment or PTA,("PTA"), under certain circumstances to compensate for delays in obtaining the patent caused by the U.S. Patent and Trademark Office. In some instances, such a PTA may result in a U.S. patent term extending beyond 20 years from the earliest date of filing a non-provisional patent application. In addition, in the U.S., the term of a U.S. patent that covers an FDA approved drug may also be eligible for patent term extension, which permits patent term restoration as compensation for the patent term lost during the FDA regulatory review process. The Hatch Waxman Act permits a patent term extension of up to five years beyond the expiration of the patent. The length of the patent term extension is related to the length of time the drug is under regulatory review. Patent term
23
extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval and only one patent applicable to an approved drug may be extended. Similar provisions are available in Europe and other foreign jurisdictions to extend the term of a patent that covers an approved drug. In the future, if and when our products receive FDA approval, we expect to apply for patent term extensions on patents covering those products. We plan to seek patent term extensions to any of our issued patents in any jurisdiction where these are available; however, there is no guarantee that the applicable authorities, including the FDA in the U.S., will agree with our assessment of whether such extensions should be granted, and if granted, the length of such extensions.
Patent Protection
For all patent applications, we determine strategy for claim scope on a case-by-case basis, taking into account advice of counsel and our business model and needs. We file patents containing claims for protection of all useful applications of our proprietary technologies and any products, as well as all new applications and/or uses we discover for existing technologies and products, based on our assessment of their strategic value. We continuously reassess the number and type of patent applications, as well as pending and issued patent claims to ensure that maximum coverage and value are obtained for our processes and compositions, given existing patent office rules and regulations. Further, claims may be modified during patent prosecution to meet our intellectual property and business needs.
Our patent estate includes patent applications with claims relating to our product candidates, methods of use and manufacturing processes, and claims for potential future products and developments. As of February 28, 2021,January 31, 2024, we have had 74greater than 80 issued patents and patent applications pending in the United States and foreign jurisdictions relating to CTX-009, CTX-471, CTX-8371 and other discovery and research programs. We have six7 patents which haveissued in the United States and 1 patent issued in Taiwan related to our CTX-471 program, and 1 patent issued in the United States related to our CTX-471CTX-8371 program. We also have access to patents issued in Australia, Europe and the United States related to our antibody and display programs, as well as 27 patents issued in Australia, Canada, China, Europe, Japan, Korea, Russia and 2 in United States related to our CTX-009 program.
More specifically, we have licensed 2 patent families with 2 issued patents in U.S. and 27 issued patents in foreign jurisdictions, related to our DLL4/VEGF antibody program including, but not limited to, our CTX-009 therapeutic candidate. Patents in these patent families are generally expected to start to expire in 2033, subject to possible extension.
We own five4 pending patent families with six7 issued U.S. patents fourand 1 issued patent in Taiwan, 3 U.S. Utility or provisional patent applications, one Patent Cooperation Treaty, or PCT, patent applications, and 2643 patent applications in foreign jurisdictions, related to our CD137 agonist antibody therapeutic platform including, but not limited to, our CTX-471 therapeutic candidate. Patents that grant from these patent families are generally expected to start to expire in 2038, subject to possible patent term extension.
We own two1 pending patent familiesfamily with three1 issued U.S. Utility or provisional patent, applications, one PCT1 U.S. patent application, and two18 patent applications in foreign jurisdictions, related to our PD-1/PD-L1 bispecific antibody therapeutic platform including, but not limited to, our CTX-8371 therapeutic candidate. Patents that grant from these patent families are generally expected to start to expire in 2039, subject to possible patent term extension.
We own, or have an ownership interest in, seven2 pending patent families with eight2 U.S. Utility or provisionalpending patent applications and two PCT patent applications and eight3 patent applications in foreign jurisdictions, related to our CD277 discovery and research programs. Patents that grant from these patent families are generally expected to start to expire in 2039, subject to possible patent term extension.
We own four1 pending patent familiesfamily with two1 U.S. Utility or provisionalpending patent applicationsapplication and two PCT1 patent applicationsapplication in foreign jurisdictions related to our antibody and display programs including, but not limited to, common light chains and mammalian display platforms. Patents that grant from thesethis patent familiesfamily are generally expected to start to expire in 2039, subject to possible patent term extension.
Trademark Protection
We have filed for and obtained trademark protection in multiple jurisdictions for the COMPASS THERAPEUTICS word mark and logo for goods and services. We have filed for and obtained trademark protection on the StitchMabsTM word mark in the U.S.United States for goods.services.
24
Trade Secret Protection
Finally, we may rely, in some circumstances, on trade secrets to protect our technology. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors and contractors. We also seek to preserve the integrity and confidentiality of our data and trade secrets by maintaining physical security of our premises and physical and electronic security of our information technology systems. While we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our consultants, contractors or collaborators use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions. For further information, please see “Risk Factors—Factors—Risks Related to Our Intellectual Property.”
Competition
The biotechnology and pharmaceutical industries, and the immuno-oncology subsector, are characterized by rapid evolution of technologies, fierce competition and strong defense of intellectual property. We believe that our programs, including CTX-009, CTX-471, CTX-8371 and our platform technologies, including our StitchMabsTM platform and our programs, technology, knowledge, experience and scientific resources provide us with competitive advantages, but we also face competition from pharmaceutical and biotechnology companies, academic institutions, governmental agencies and public and private research institutions, among others. Our competitors include larger and better funded biopharmaceutical, biotechnology and therapeutics companies, including companies focused on cancer immunotherapies, such as AbbVie, Amgen, Inc., AstraZeneca plc, Bristol-Myers Squibb Company, or BMS, Eli Lilly, Genentech, Inc., GlaxoSmithKline PLC, Johnson & Johnson, Merck & Co., Inc., Merck KGaA, Novartis AG, Pfizer Inc., Roche Holding Ltd and Sanofi S.A. Moreover, we may also compete with smaller or earlier-stage companies, universities and other research institutions that have developed, are developing or may be developing current and future cancer therapeutics.
Product candidates that we successfully develop will compete with a range of therapies that are currently approved and any new therapies that may become available in the future. Key product features that would affect our ability to effectively compete with other therapeutics include the efficacy, safety and convenience of our products. Currently marketed oncology drugs and therapeutics range from traditional cancer therapies, including chemotherapy, to antibody-drug conjugates, such as Genentech Inc.’s Kadcyla, to immune checkpoint inhibitors targeting CTLA-4, such as BMS’BMS’s Yervoy, andto PD-1/PD-L1, such as BMS’BMS’s Opdivo, Merck & Co.’s Keytruda and Genentech’s Tecentriq, to T cell-engager immunotherapies, such as Amgen’s Blincyto.Blincyto and VEGF targets, such as Genentech Inc.’s Avastin. In addition to these marketed therapies, numerous compounds are in clinical development for the potential treatment of cancer.
Biliary tract cancers are aggressive and rare gastrointestinal cancers that have a very poor prognosis. First line treatment of locally advanced or metastatic BTC includes the chemotherapy combination of gemcitabine and cisplatin, often with the addition of the PD-1 inhibitor Imfinzi® (durvalumab). In September of 2022, AstraZeneca received FDA approval of durvalumab in combination with gemcitabine/cisplatin for the first line treatment of BTC. The only FDA approved therapies for second line treatment of BTC are targeted therapies that address specific tumor mutations or solid tumors that are microsatellite instability high. There is also recently emerging clinical data that blockade of the human epidermal growth factor receptor 2 (“Her-2”) may be beneficial to the small subset of patients in whom HER-2 is amplified. We believe that these targeted therapies are appropriate for 10-15% of BTC patients.
Colorectal cancer is the second most common cause of cancer deaths in the United States and constitutes approximately 10% of all annually diagnosed cancer and cancer related deaths globally. Treatment of metastatic disease in the first line typically includes either the VEGF inhibitor Avastin® (bevacizumab) or an EGFR inhibitor such as Erbitux® (cetuximab), combined with chemotherapy. Treatment in the second line includes re-treatment with either a VEGR or EGFR inhibitor and a different chemotherapy regimen that is either oxaliplatin or irinotecan based. Approved therapies for advanced patients with metastatic disease are limited, with approved agents such as Lonsurf® (trifluridine and tipiracil) and Stivarga® (regorafenib) offering only 1-2% response rates and improvement in survival of 1-2 months.
If we are successful in advancing one or more of our product candidates toward registrational studiestrials and filing a BLA or BLAs, and if we are successful at obtaining approvals from the FDA or any other regulatory agency to market one or more of our product candidates, then the availability of reimbursement from government and other third-party payors will also significantly affect the pricing and competitiveness of our products. Our competitors, who may be successful at obtaining marketing approval from the FDA or other regulatory approval for their products prior to us obtaining marketing approval for our product candidates, could result in our competitors launching their products sooner and establishing a strong market position before we are even able to enter the market.
Sales and Marketing
We hold worldwide rights to all of our product candidates (with the exception of limited countries for CTX-009), which provide us the optionality to grow our internal pipeline independently or partner selected rights to our product candidates in different geographies throughout the world. Subject to receiving marketing approval, we intend to maximize the value of our product candidates by either independently planning to pursue the commercialization of our
25
products in one or more major geographies by building an internal sales and marketing organization, or by seeking collaborations with third parties with commercialization infrastructure.
At the appropriate time in the future, and if one or more of our product candidates continues to advance successfully in development and enter registrational studies,trials, we also plan to build a marketing and sales management organization to create and implement marketing strategies for any product candidates that we would potentially market through our own sales organization and to oversee and support our sales force. The responsibilities of such marketing organization would include developing educational initiatives with respect to approved products and establishing relationships with researchers and practitioners in relevant fields of medicine.
Manufacturing
We do not currently own or operate manufacturing facilities for the production of clinical quantities of our product candidates. We have relied on, and intend to continue to rely on, qualified third-party contract manufacturers to produce our product candidates, including clinical supplies to support our clinical trials. At the appropriate time in the product development process, we will determine whether to establish manufacturing facilities or continue to rely on third parties to manufacture clinical quantities of any products that we may successfully develop. We expect that commercial quantities of any compound and materials for our product candidates, if approved, will be manufactured in facilities and by processes that comply with FDA and other regulations, which may differ from our current clinical supply manufacturers.
Government Regulation
Government authorities in the United States, including federal, state, and local authorities, and in other countries, extensively regulate, among other things, the manufacturing, research and clinical development, marketing, labeling and packaging, storage, distribution, post-approval monitoring and reporting, advertising and promotion, and export and import of biological products, such as those we are developing. In addition, some government authorities regulate the pricing of such products. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources.
Review and Approval for Licensing Biologics in the United States
In the United States, the FDA regulates biological products under the Federal Food, Drug, and Cosmetic Act or FDCA,("FDCA"), and the Public Health Service Act or PHSA,("PHSA"), and their implementing regulations. FDA approval is required before any biological product can be marketed in the United States. Biological products are also subject to other federal, state, and local statutes and regulations. If we fail to comply with applicable FDA or other requirements at any time during the product development process, clinical testing, the approval process or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include the FDA’s refusal to approve pending applications, license suspension or revocation, withdrawal of an approval, untitled or warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties or criminal prosecution. Any FDA enforcement action could have a material adverse effect on us.
The process required by the FDA before product candidates may be marketed in the United States generally involves the following:
completion of extensive nonclinical laboratory tests and nonclinical animal studies, all performed in accordance with the Good Laboratory Practices, or GLP, regulations;
● | completion of extensive nonclinical laboratory tests and nonclinical animal studies, all performed in accordance with the Good Laboratory Practices ("GLP"), regulations; |
● | submission to the FDA of an |
26
● | approval by an independent institutional review board ("IRB"), or ethics committee representing each clinical site before each clinical trial may be initiated; |
approval by an independent institutional review board, or IRB, or ethics committee representing each clinical site before each clinical trial may be initiated;
● | performance of adequate and well-controlled human clinical trials in accordance with Good Clinical Practices ("GCPs"), to establish the safety and efficacy of the product candidate for each proposed indication; |
performance of adequate and well-controlled human clinical trials in accordance with Good Clinical Practices, or GCPs, to establish the safety and efficacy of the product candidate for each proposed indication;
● | preparation of and submission to the FDA of a BLA, after completion of all pivotal clinical trials; |
preparation of and submission to the FDA of a biologics license application, or BLA, after completion of all pivotal clinical trials;
● | review of the product application by an FDA advisory committee, where appropriate and if applicable; |
review of the product application by an FDA advisory committee, where appropriate and if applicable;
● | a determination by the FDA within 60 days of its receipt of a BLA to file the application for review; satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities where the proposed product is produced to assess compliance with current Good Manufacturing Practices ("cGMP"); |
a determination by the FDA within 60 days of its receipt of a BLA to file the application for review; satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities where the proposed product is produced to assess compliance with current Good Manufacturing Practices, or cGMP;
● | satisfactory completion of any FDA audits of the clinical trial sites to assure compliance with GCPs, and the integrity of clinical data in support of the BLA; and |
satisfactory completion of any FDA audits of the clinical study sites to assure compliance with GCPs, and the integrity of clinical data in support of the BLA; and
● | FDA review and approval of a BLA for a biological product candidate that is safe, pure, and potent prior to any commercial marketing or sale of the product in the United States. |
FDA review and approval of a BLA for a biological product candidate that is safe, pure, and potent prior to any commercial marketing or sale of the product in the United States.
The nonclinical and clinical testing and approval process requires substantial time, effort, and financial resources, and we cannot be certain that any approvals for our product candidates will be granted on a timely basis, if at all.
Before testing any drug or biologic in humans, a product candidate must undergo rigorous preclinical testing. Preclinical studies include laboratory evaluations of product chemistry, formulation, and stability, as well as in vitro and animal studies to assess safety and in some cases to establish the rationale for therapeutic use. The conduct of preclinical studies is subject to federal and state regulation and requirements, including GLP requirements for safety/toxicology studies. The results of the preclinical studies, together with manufacturing information and analytical data, must be submitted to the FDA as part of an IND.
An IND is a request for authorization from the FDA to administer an investigational biological product to humans in clinical trials. The central focus of an IND submission is on the general investigational plan, the protocol(s) for human trials and the safety of studytrial participants. The IND also includes results of animal and in vitro studies assessing the toxicology, pharmacokinetics,PK, pharmacology, and pharmacodynamic characteristics of the product; chemistry, manufacturing and controls information; and any available human data or literature to support the use of the investigational product. An IND must become effective before human clinical trials may begin. An IND will automatically become effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to the proposed clinical trials. In such a case, the IND may be placed on clinical hold and the IND sponsor and the FDA must resolve any outstanding concerns or questions before clinical trials can begin. Accordingly, submission of an IND may or may not result in the FDA allowing clinical trials to commence. The FDA may impose a clinical hold at any time during clinical trials and may impose a partial clinical hold that would limit trials, for example, to certain doses or for a certain length of time.
Clinical Trials
Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified investigators in accordance with GCPs, which include the requirement that all research subjects provide their informed consent for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study,trial, the inclusion and exclusion criteria, the parameters to be used in monitoring safety, and the efficacy criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. Additionally, approval must also be obtained from each clinical trial site’s IRB, before the trials may be initiated and the IRB must monitor the trial until completed. There are also requirements governing the reporting of ongoing clinical trials and clinical trial results to public registries.
27
The clinical investigation of a biological product is generally divided into three or four phases. Although the phases are usually conducted sequentially, they may overlap or be combined.
Phase 1. The investigational product is initially introduced into healthy human subjects or patients with the target disease or condition. These studiestrials are designed to evaluate the safety, dosage tolerance, metabolism and pharmacologic actions of the investigational product in humans, the side effects associated with increasing doses, and if possible, to gain early evidence on effectiveness.
Phase 2. The investigational product is administered to a limited patient population to evaluate dosage tolerance and optimal dosage, identify possible adverse side effects and safety risks, and preliminarily evaluate efficacy.
Phase 3. The investigational product is administered to an expanded patient population, generally at geographically dispersed clinical trial sites to generate enough data to statistically evaluate safety, purity and potency, to evaluate the overall benefit-risk profile of the investigational product, and to provide an adequate basis for physician labeling.
Phase 4. In some cases, the FDA may condition approval of a BLA for a product candidate on the sponsor’s agreement to conduct additional clinical trials after approval. In other cases, a sponsor may voluntarily conduct additional clinical trials after approval to gain more information about the biological product. Such post-approval studiestrials are typically referred to as Phase 4 clinical trials.
In March 2022, the FDA released a final guidance entitled “Expansion Cohorts: Use in First-In-Human Clinical Trials to Expedite Development of Oncology Drugs and Biologics,” which outlines how drug developers can utilize an adaptive trial design commonly referred to as a seamless trial design in early stages of oncology drug development (i.e., the first-in-human clinical trial) to compress the traditional three phases of trials into one continuous trial called an expansion cohort trial. Information to support the design of individual expansion cohorts is included in IND applications and assessed by FDA. Expansion cohort trials can potentially bring efficiency to drug development and reduce development costs and time.
Sponsors must also report to the FDA, within certain timeframes, serious and unexpected adverse reactions, any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator’s brochure, or any findings from other studies or animal or in vitro testing that suggest a significant risk in humans exposed to the product candidate. The FDA, the IRB, or the clinical trial sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data and safety monitoring board or committee. This group provides authorization for whether or not a trial may move forward at designated check points based on access to certain data from the trial. We may also suspend or terminate a clinical trial based on evolving business objectives or competitive climate.
A manufacturersponsor of an investigational biological product for a serious disease or condition is required to make available, such as by posting on its website, its policy on evaluating and responding to requests for individual patient access to such investigational biological product. This requirement applies on the earlier of the first initiation of a Phase 2 or Phase 3 trial of the investigational biological product or, as applicable, 15 days after the biological product receives a designation as a breakthrough therapy or fast track product.
Concurrent with clinical trials, sponsors usually complete additional animal studies and must also develop additional information about the chemistry and physical characteristics of the product candidate and finalize a process for manufacturing the drug product in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the product candidate and manufacturers must develop, among other things, methods for testing the identity, strength, quality, and purity of the final drug product. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate that the product candidate does not undergo unacceptable deterioration over its shelf life.
Submission of a BLA to the FDA
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, detailed investigational product information is submitted to the FDA in the form of a BLA requesting approval to market the product for one or more indications. Under federal law, the submission of most BLAs is subject to an application user fee. For fiscal year 2020,2024, the application user fee is $2,942,965,$4,048,695, and the sponsor of an approved BLA is also subject to an annual program fee of $325,424$416,734 for each approved biological product on the market. These fees are typically increased annually. Applications for orphan drug products are exempted from the BLA application fee and may be exempted from program fees, unless the application includes an indication for other than a rare disease or condition.
A BLA must include all relevant data available from pertinent nonclinical studies and clinical trials, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s chemistry, manufacturing, controls, and proposed labeling, among other things.
28
Data can come from company-sponsored clinical trials intended to test the safety and effectiveness of a use of a product, or from a number of alternative sources, including trials initiated by investigators. To support marketing approval, the data submitted must be sufficient in quality and quantity to establish the safety and effectiveness of the investigational product to the satisfaction of the FDA.
The FDA conducts a preliminary review of all BLAs within the first 60 days after submission before accepting them for filing to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an application for filing. Once a BLA has been submitted, the FDA’s goal for novel biological products generally is to review the application within ten months after it accepts the application for filing, or, if the application relates to an unmet medical need in a serious or life-threatening indication, six months after the FDA accepts the application for filing. The review process is often significantly extended by the FDA’s requests for additional information or clarification.
Before approving a BLA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving a BLA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP.
The FDA is required to refer an application for a novel biological product to an advisory committee or explain why such referral was not made. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
The FDA’sFDA’s Decision on a BLA
After the FDA evaluates the BLA and conducts relevant inspections, it may issue an approval letter or a Complete Response Letter. An approval letter authorizes commercial marketing of the biological product with specific prescribing information for specific indications. A Complete Response Letter indicates that the review cycle of the application is complete and the application is not ready for approval. A Complete Response Letter will identify the deficiencies that prevent the FDA from approving the application and may require additional clinical data or an additional Phase 3 clinical trial(s), or other significant, expensive and time-consuming requirements related to clinical trials, nonclinical studies or manufacturing. Even if such additional information is submitted, the FDA may ultimately decide that the BLA does not satisfy the criteria for approval and issue a denial.
The FDA could also approve the BLA with a Risk Evaluation and Mitigation Strategy or REMS,("REMS"), program to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA also may condition approval on, among other things, changes to proposed labeling, development of adequate controls and specifications, or a commitment to conduct one or more post-market studies or clinical trials. Such post-market testing may include Phase 4 clinical trials and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization.
New government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or prevent regulatory approval of our products under development.
Expedited Review and Accelerated Approval Programs
A sponsor may seek approval of its product candidate under programs designed to accelerate FDA’s review and approval of BLAs. For example, Fast Track Designationfast track designation may be granted to a biological
29
product intended for treatment of a serious or life-threatening disease or condition that has potential to address unmet medical needs. The key benefits of fast track designation are more frequent interactions with the FDA during development and testing, the eligibility for priority review, and rolling review, which is submission of portions of an application before the complete marketing application is submitted.
Based on results of the Phase 3 clinical trial(s) submitted in a BLA,
Additionally, the FDA may grant the BLA a product candidate priority review designation, which sets the target date for FDA action on the marketing application for a novel product at six months after the FDA accepts the application for filing. Priority review is granted where there is evidence that the proposed product is intended to treat a serious condition and, if approved, would beprovide a significant improvement in the safety or effectiveness of the treatment, diagnosis, or prevention of a serious condition.effectiveness. If criteria are not met for priority review, the application is subject to the standard FDA review period of ten months after FDA accepts the application for filing. Priority review designation does not change the scientific/medical standard for approval or the quality of evidence necessary to support approval.
Under the accelerated approval program, the FDA may approve a BLA onfor a product that is intended to treat a serious or life-threatening disease or condition upon the basis ofdetermination that the product generally provides meaningful therapeutic benefit to patients over available treatments and demonstrates an effect on either a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. Post-marketingAccelerated approval is usually contingent on a sponsor’s agreement to conduct post-approval confirmatory trials or completion ofcomplete ongoing trials after marketing approval are generally requiredin a diligent manner to verify the biological product’s clinical benefit in relationship to the surrogate endpoint or ultimate outcome in relationship to the clinical benefit. Under the Food and Drug Omnibus Reform Act of 2022 (“FDORA”), the FDA is now permitted to require, as appropriate, that post-approval confirmatory trials be underway prior to approval or within a specific time period after accelerated approval is granted and the FDA has increased authority for expedited procedures to withdraw approval of a product or an indication approved under accelerated approval if, for example, the confirmatory trial is not conducted with due diligence or fails to verify the predicted clinical benefit of the product. Additionally, the FDA generally requires, unless otherwise informed by the agency, that all advertising and promotional materials intended for dissemination or publication within 120 days of marketing approval be submitted to the agency for preapproval and pre-use review.
In addition, a sponsor may seek FDA designation of its product candidate as a breakthrough therapy if the biological product is intended, alone or in combination with one or more other drugs or biological products, to treat a serious or life- threatening disease or condition and preliminary clinical evidence indicates that the biological product candidate may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The benefits of breakthrough therapy designation include the same benefits as a Fast Trackfast-track designation, in addition to intensive guidance from FDA to ensure an efficient development program.
Finally, with respect to oncology products, the FDA may review applications under Real-Time Oncology Review (“RTOR”) established by the FDA’s Oncology Center of Excellence. RTOR allows an applicant to pre-submit components of the application to allow the FDA to review clinical data before the complete filing is submitted in order to create more efficient review process to ensure that safe and effective treatments are available to patients as early as possible, while maintaining and improving review quality. Drugs considered for review under RTOR must, among other things, be likely to demonstrate substantial improvements on a clinically relevant endpoint(s) over available therapy and must have easily interpreted endpoints. In addition, no aspect of the application should be likely to require a longer review time (for example, a Risk Evaluation and Mitigation Strategy). To determine eligibility for RTOR, the FDA requires top-line efficacy and safety results from an applicant.
Post-Approval Requirements
Biological products manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims, are subject to prior FDA review and approval. Biological product manufacturers are subject to periodic unannounced inspections by the FDA and state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated, and, depending on the significance of the change, may require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP and impose reporting and documentation requirements upon us and any third-party manufacturers that we may decide to use. Manufacturers and manufacturers’ facilities are also required to comply with applicable product tracking and tracing requirements and notify the FDA of counterfeit, diverted, stolen and intentionally adulterated products or products that are otherwise unfit for distribution in the United States. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain compliance with cGMP and other aspects of regulatory compliance.
We rely, and expect to continue to rely, on third parties for the production of clinical quantities of our product candidates and expect to rely in the future on third parties for the production of commercial quantities. Future FDA and state inspections may identify compliance issues at our facilities or at the facilities of our contract manufacturers that may disrupt production, or distribution, or may require substantial resources to correct. In addition, discovery of previously unknown problems with a product or the failure to comply with applicable requirements may result in restrictions on a product, manufacturer or holder of an approved BLA, including withdrawal or recall of the product from the market or other voluntary, FDA-initiated or judicial action that could delay or prohibit further marketing.
30
The FDA may suspend or revoke product license approvals if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution restrictions or other restrictions under a REMS program. FDA has authority to require post-market studies, in certain circumstances, on reduced effectiveness of a biological product and FDA may require labeling changes related to new reduced effectiveness information. Other potential consequences of a failure to maintain regulatory compliance include, among other things:
restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
fines, untitled or warning letters or holds on post-approval clinical trials;
refusal of the FDA to approve pending BLAs or supplements to approved BLAs;
product seizure or detention, or refusal to permit the import or export of products; or
● | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
injunctions or the imposition of civil or criminal penalties.
● | fines, untitled or warning letters or holds on post-approval clinical trials; |
● | refusal of the FDA to approve pending BLAs or supplements to approved BLAs; |
● | product seizure or detention, or refusal to permit the import or export of products; or |
● | injunctions or the imposition of civil or criminal penalties. |
The FDA strictly regulates marketing, labeling, advertising, and promotion of products that are placed on the market. Biological products may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability.
Pediatric Trials and Exclusivity
A sponsor who is planning to submit a marketing application for a biological product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration must submit an initial Pediatric Study Plan or PSP,("PSP"), within sixty days of an end of Phase 2 meeting or as may be agreed between the sponsor and FDA. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including study objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. Generally, development program candidates designated as orphan drugs are exempt from the above requirements. FDA and the sponsor must reach agreement on the PSP. A sponsor can submit amendments to an agreed upon initial PSP at any time if changes to the pediatric plan need to be considered based on data collected from nonclinical studies, early phase clinical trials, and/or other clinical development programs.
Pediatric exclusivity is another type of non-patent exclusivity in the United States and, if granted for a biologic, provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity for all formulations, dosage forms, and indications of the biologic, including the five-year and three-year non-patent and orphan exclusivity. This six-month exclusivity may be granted if a BLA sponsor submits pediatric data that fairly respond to a written request from the FDA for such data.data, provided that at the time pediatric exclusivity is granted there is not less than nine months of term remaining. The data do not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the FDA’s request, the additional protection is granted. If reports of FDA-requested pediatric trials are submitted to and accepted by the FDA within the statutory time limits, whatever statutory or regulatory periods of exclusivity or patent protection covering the product are extended by six months. This is not a patent term extension, but it effectively extends the regulatory period during which the FDA cannot accept or approve another application relying on the BLA sponsor’s data.
31
Patent Term Restoration
Depending upon the timing, duration, and specifics of the FDA approval of the use of our product candidates, some of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, commonly referred to as the Hatch-Waxman Amendments. The Hatch- Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND and the submission date of a BLA, plus the time between the submission date and the approval of that application. Only one patent applicable to an approved product is eligible for the extension and the application for the extension must be submitted prior to the expiration of the patent and within 60 days of the product’s approval. The U.S. Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we may apply for restoration of patent term for one of our currently owned or licensed patents to add patent life beyond its current expiration date, depending on the expected length of the clinical trials and other factors involved in the filing of the relevant BLA.
Biosimilars and Exclusivity
The Patient Protection and Affordable Care Act or ("Affordable Care Act,Act"), signed into law on March 23, 2010, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009 or BPCI Act,("BPCI"), which created an abbreviated approval pathway for biological products shown to be similar to, or interchangeable with, an FDA-licensed reference biological product. This amendment to the PHSA attempts to minimize duplicative testing. Biosimilarity, which requires that there be no clinically meaningful differences between the proposed biological product and the reference product in terms of safety, purity, and potency, can be shown through analytical studies, animal studies, and a clinical trial or trials. Interchangeability requires that a product is biosimilar to the reference product and the product must demonstrate that it can be expected to produce the same clinical results as the reference product and, for products administered multiple times, the biologic and the reference biologic may be switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic.
A
Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date that the reference biologic is granted twelveproduct was first licensed by the FDA. In addition, the approval of a biosimilar product may not be made effective by the FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, from the time of first licensureanother company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing that applicant’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity, and potency of its product. In addition, The BPCIA also created certain exclusivity periods for biosimilars approved as interchangeable products. The first biologic submitted under the abbreviated approval pathway that is determined to be interchangeable with the reference product is eligible for a period of exclusivity against other biologics submitted under the abbreviated approval pathway during which time the FDA may not determine that another product is interchangeable with the same reference product for any condition of use. The FDA may approve multiple “first” interchangeable products so long as they are all approved on the same first day of marketing. This exclusivity period, which may be shared amongst multiple first interchangeable products, lasts for the lesser of (i) one year after the first commercial marketing, (ii) 18 months after approval if there is no legal challenge, (iii) 18 months after the resolution in the applicant’s favor of a lawsuit challenging the biologic’s patents if an application has been submitted, or (iv) 42 months after the application has been approved if a lawsuit is ongoing within the 42-month period. Products deemed “interchangeable” by the FDA may be readily substituted by pharmacies and such substitution is which are governed by state pharmacy law.
European Union/Rest of World Government Regulation
In addition to regulations in the United States, we will be subject to a variety of regulations in other jurisdictions governing, among other things, clinical trials and any commercial sales and distribution of our products. The cost of establishing a regulatory compliance system for numerous varying jurisdictions can be very significant. Although many of the issues discussed above with respect to the United States apply similarly in the context of the European Union and in other jurisdictions, the approval process varies between countries and jurisdictions and can involve additional product testing and additional administrative review periods. The time required to obtain approval in other countries and jurisdictions might differ from and be longer than that required to obtain FDA approval. Regulatory approval in one country or jurisdiction does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country or jurisdiction may negatively impact the regulatory process in others.
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in foreign countries prior to the commencement of clinical trials or marketing of the product in those countries. Certain countries outside of the United States have a similar process that requires the submission of a clinical trial application much like the IND prior to the commencement of human clinical trials. In the European Union, for example, a clinical trial authorization application, or CTA, must be submitted for each clinical protocol to each country’s national health authority and an
32
independent ethics committee, much like the FDA and IRB, respectively. Once the CTA is accepted in accordance with a country’s requirements, the clinical trial may proceed.
The requirements and process governing the conduct of clinical trials vary from country to country. In all cases, the clinical trials are conducted in accordance with GCP, the applicable regulatory requirements, and the ethical principles that have their origin in the Declaration of Helsinki.
To obtain regulatory approval of an investigationala medicinal product under European Union regulatory systems, we must submit a marketing authorization application. The content of the BLA filed in the United States is similar to that required in the European Union, with the exception of, among other things, country-specific document requirements.
For other countries outside of the European Union, such as countries in Eastern Europe, Latin America or Asia, the requirements governing product licensing, pricing, and reimbursement vary from country to country.
Countries that are part of the European Union, as well as countries outside of the European Union, have their own governing bodies, requirements, and processes with respect to the approval of biological products. If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Clinical Trial Approval in the European Union
In April 2014, the European Union adopted the Clinical Trials Regulation (EU) No 536/2014, which replaced the Clinical Trials Directive 2001/20/EC on January 31, 2022. The Clinical Trials Regulation is directly applicable in all European Union Member States meaning no national implementing legislation in each European Union Member State is required. The Clinical Trials Regulation aims to simplify and streamline the approval of clinical trials in the European Union. Under the new coordinated procedure for the approval of clinical trials, the sponsor of a clinical trial is required to submit a single application for approval of a clinical trial to a reporting European Union Member State through a European Union Portal. The submission procedure is the same irrespective of whether the clinical trial is to be conducted in a single European Union Member State or in more than one European Union Member State.
Marketing Authorization Procedures in the European Union
Medicines can be authorized in the European Union by using either the centralized authorization procedure or national authorization procedures.
Centralized procedure. The European Medicines Agency or EMA,("EMA"), implemented the centralized procedure for the approval of human medicines to facilitate marketing authorizations that are valid throughout the European Economic Area or EEA,("EEA"), which is comprised of the 28 member statesMember States of the European Union plus Norway, Iceland, and Lichtenstein. This procedure results in a single marketing authorization issued by the EMA that is valid across the EEA. The centralized procedure is compulsory for human medicines that are: derived from biotechnology processes, such as genetic engineering, contain a new active substance indicated for the treatment of certain diseases, such as HIV/HIV, AIDS, cancer, diabetes, neurodegenerative disorders or autoimmune diseases and other immune dysfunctions, advanced therapy medicines (gene-therapy, somatic cell-therapy or tissue-engineered medicines) and officially designated orphan medicines.
For medicines that do not fall within these categories, an applicant has the option of submitting an application for a centralized marketing authorization to the European Commission following a favorable opinion by the EMA, as long as the medicine concerned is a significant therapeutic, scientific or technical innovation, or if its authorization would be in the interest of public health.health in the European Union.
Under the centralized procedure, the EMA’s Committee for Medicinal Products for Human Use ("CHMP") is responsible for conducting the initial assessment of a product and for several post-authorization and maintenance activities, such as the assessment of modifications or extensions to an existing marketing authorization. Under the centralized procedure in the European Union, the maximum timeframe for the evaluation of a marketing authorization application is 210 days (excluding clock stops, when additional written or oral information is to be provided by the applicant in response to questions asked by the CHMP). Clock stops may extend the timeframe of evaluation of a marketing authorization application considerably beyond 210 days. Where the CHMP gives a positive opinion, it provides the opinion together with supporting documentation to the European Commission, who makes the final decision to grant a marketing authorization, which is issued within 67 days of receipt of the EMA’s recommendation. Accelerated evaluation might be granted by the CHMP in exceptional cases, when a medicinal product is expected to be of a major public health interest, particularly from the point of view of therapeutic innovation. In this circumstance, the EMA ensures that the opinion of the CHMP is given within 150 days, excluding clock stops, but it is possible that the CHMP can revert to the standard time limit for the centralized procedure if it considers that it is no longer appropriate to conduct an accelerated assessment.
National authorization procedures. There are also two other possible routes to authorize medicinal products in several European Union countries, which are available for investigational medicinal products that fall outside the scope of the centralized procedure:
Decentralized procedure. Using the decentralized procedure, an applicant may apply for simultaneous authorization in more than one European Union countryMember State of medicinal products that have not yet been authorized in any European Union countryMember State and that do not fall within the mandatory scope of the centralized procedure.
Mutual recognition procedure. In the mutual recognition procedure, a medicine is first authorized in one European Union Member State, in accordance with the national procedures of that country.Member State. Following this, further marketing authorizations can be sought from other European Union countriesMember States in a procedure
33
whereby the countriesMember State concerned agree to recognize the validity of the original, national marketing authorization.
In some cases, a Pediatric Investigation Plan or PIP,("PIP"), or a request for waiver or deferral, is required for submission prior to submitting a marketing authorization application. A PIP describes, among other things, proposed pediatric trials and their timing relative to clinical trials in adults.
New Chemical Entity
Data and Market Exclusivity in the European Union
In the European Union, new chemical entities, sometimes referred to as new active substances,innovative medicinal products approved on the basis of a complete and independent data package qualify for eight years of data exclusivity upon marketing authorization and an additional two years of market exclusivity. This data exclusivity, if granted, prevents regulatory authorities in the European Union from referencing the innovator’s pre-clinical and clinical trial data to assesscontained in the dossier of the reference product when applying for a generic (abbreviated) application foror biosimilar marketing authorization in the European Union, during a period of eight years afterfrom the date on which the reference product was first authorized in the European Union. During an additional two-year period of market exclusivity, a generic marketing authorizationor biosimilar MAA can be submitted and authorized, and the innovator’s data may be referenced, but not approved for two years.no generic or biosimilar medicinal product can be placed on the European Union market until the expiration of the market exclusivity. The overall ten-year period will be extended to a maximum of eleven years if, during the first eight years of those ten years, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies. There is no guarantee that a product will be considered by the EMA to be an innovative medicinal product, and products may not qualify for data exclusivity. Even if a product is considered to be an innovative medicinal product so that the innovator gains the prescribed period of data exclusivity, another company nevertheless could also market another version of the product if such company obtained a marketing authorization based on a marketing authorization application with a complete and independent data package of pharmaceutical tests, preclinical tests and clinical trials.
Accelerated Review
UnderThe aforementioned European Union rules are generally applicable in the centralized procedureEEA.
Reform of the Regulatory Framework in the European Union
The European Commission introduced legislative proposals in April 2023 that, if implemented, will replace the maximum timeframecurrent regulatory framework in the European Union for all medicines (including those for rare diseases and for children). The European Commission has provided the evaluation of a marketing authorization application is 210 days (excluding clock stops, when additional written or oral information islegislative proposals to the European Parliament and the European Council for their review and approval. In October 2023, the European Parliament published draft reports proposing amendments to the legislative proposals, which will be provideddebated by the applicant in response to questions asked byEuropean Parliament. Once the EMA’s Committee for Medicinal Products for Human Use,European Commission’s legislative proposals are approved (with or CHMP). Accelerated evaluation mightwithout amendment), they will be granted by the CHMP in exceptional cases, when a medicinal product is expected to be of a major public health interest, particularly from the point of view of therapeutic innovation. In this circumstance, EMA ensures that the opinion of the CHMP is given within 150 days, excluding clock stops.adopted into European Union law.
Pharmaceutical Coverage, Pricing and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any products for which we may obtain regulatory approval. In the United States and markets in other countries, sales of any products for which we may receive regulatory approval for commercial sale will depend in part on the availability of coverage and reimbursement for our products from third-party payors, such as government healthcare programs (e.g., Medicare, Medicaid), managed care providers, private health insurers, health maintenance organizations, and other organizations. These third-party payors decide which medications they will pay for and will establish reimbursement levels. The availability of coverage and extent of reimbursement by governmental and other third-party payors is essential for most patients to be able to afford treatments such as antibody-based therapies.
In the United States, the principal decisions about reimbursement for new medicines are typically made by the Centers for Medicare & Medicaid Services or CMS,("CMS"), an agency within the U.S. Department of Health and Human Services or HHS.("HHS"). CMS decides whether and to what extent our products will be covered and reimbursed under Medicare and private payors tend to follow CMS to a substantial degree. Factors payors consider in determining reimbursement are based on whether the product is:
● | a covered benefit under its health plan; |
● | safe, effective and medically necessary; |
a covered benefit under its health plan;
safe, effective and medically necessary;
appropriate for the specific patient;
cost-effective; and
34
● | appropriate for the specific patient; |
● | cost-effective; and |
neither experimental nor investigational.
● | neither experimental nor investigational. |
Our ability to successfully commercialize our product candidates will depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from third-party payors.payors, including government healthcare programs (e.g., Medicare, Medicaid), managed care providers, private health insurers, health maintenance organizations, and other organizations. Moreover, a payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved.
No uniform policy of coverage and reimbursement for drug products exists among third-party payors. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the reimbursement rate that the payor will pay for the product. One payor’s determination to provide coverage for a product does not assure that other payors will also provide coverage and reimbursement for the product, and the level of coverage and reimbursement can differ significantly from payor to payor. Third-party payors may also limit coverage to specific products on an approved list, or formulary, which might not include all of the FDA-approved products for a particular indication.
Further, third-party payors are increasingly challenging the price and examining the medical necessity and cost- effectiveness of medical products and services, in addition to their safety and efficacy. In order to secure coverage and reimbursement for any product that might be approved for sale, we may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of our products, in addition to the costs required to obtain FDA or comparable regulatory approvals. Additionally, we may also need to provide discounts to purchasers, private health plans or government healthcare programs. Our product candidates may nonetheless not be considered medically necessary or cost-effective. If third-party payors do not consider a product to be cost-effective compared to other available therapies, they may not cover the product after approval as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow a company to sell its products at a profit. A decision by a third-party payor not to cover a product could reduce physician utilization once the product is approved and have a material adverse effect on sales, our operations and financial condition.
In some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, in the European Union Member States can restrict the range of medicinal products for which their national health insurance systems provide reimbursement and they can control the prices of medicinal products for human use. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost effectiveness of a particular product candidate to currently available therapies. A Member State may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. Approaches between Member States are diverging. For example, in France, effective market access will be supported by agreements with hospitals and products may be reimbursed by the Social Security Fund. The price of medicines is negotiated with the Economic Committee for Health Products or CEPS.("CEPS"). There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our product candidates. Historically, products launched in the European Union do not follow price structures of the United States and generally prices tend to be significantly lower.
Current and future healthcare reform legislation
In the United States and in some foreign jurisdictions, there have been, and likely will continue to be, a number of legislative and regulatory changes and proposed changes regarding the healthcare system directed at broadening the availability of healthcare, improving the quality of healthcare, and containing or lowering the cost of healthcare. For example, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, collectively, the Affordable Care Act or ACA,("ACA"), among other things, subjects biological products to potential competition by lower-cost
35
biosimilars, expands the types of entities eligible for the 340B drug discount program, addresses a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected, increases rebates for drugs sold to Medicaid programs owed by manufacturers under the Medicaid Drug Rebate Program and extends the rebate program to individuals enrolled in Medicaid managed care organizations, establishes annual fees and taxes on manufacturers of certain branded prescription drugs, and created a mandatory discount program for certain Medicare Part D beneficiaries in which manufacturers must agree to offer 50% (increased to 70% pursuant to the Bipartisan Budget Act of 2018, or BBA, effective as of January 2019) point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D.
Since its enactment, there have been numerous judicial, administrative, executive,
In addition, other legislative and legislative challenges to certain aspects of the ACA, and we expect there will be additional challenges and amendments to the ACA in the future. Various portions of the ACA are currently undergoing legal and constitutional challenges in the United States Supreme Court; the previous Trump administration issued various Executive Orders which eliminated cost sharing subsidies and various provisions that would impose a fiscal burden on states or a cost, fee, tax, penalty or regulatory burden on individuals, healthcare providers, health insurers, or manufacturers of pharmaceuticals or medical devices; and Congress has introduced several pieces of legislation aimed at significantly revising or repealing the ACA. It is unclear whether the ACA will be overturned, repealed, replaced, or further amended. We cannot predict what affect further changes to the ACA would have on our business.
Other legislative changes have been proposed and adopted in the United States since the ACA was enacted. For example, on August 2, 2011, the Budget Control Act of 2011 among other things, created measures for spending reductions by Congress. Specifically, the Joint Select Committee on Deficit Reduction was created to recommend to Congress proposals in spending reductions. The Joint Select Committee on Deficit Reduction did not achieve a targeted deficit reduction of at least $1.2 trillion for the years 2012 through 2021, thereby triggering the legislation’s automatic reduction to several government programs. This includes aggregate reductions to Medicare payments to providers of up to 2% per fiscal year, and due to subsequent legislative amendments, will stay in effect through 2030 unless additional Congressional action is taken. However, under the Coronavirus Aid, Relief and Economic Security Act, or CARES Act, the 2% Medicare sequestration reductions will be suspended from May 1, 2020 through December 31, 2020 due to the COVID-19 pandemic. The American Taxpayer Relief Act of 2012, or the ATRA, among other things, reduced Medicare payments to several providers and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. Additionally, in December 2018, CMS published a final rule permitting further collections and payments to and from certain ACA qualified health plans and health insurance issuers under the Affordable Care Act risk adjustment program in response to the outcome of a federal district court litigation regarding the method CMS uses to determine this risk adjustment. Since then, the ACA risk adjustment program payment parameters have been updated annually.enacted:
● | On August 2, 2011, the U.S. Budget Control Act of 2011, among other things, included aggregate reductions of Medicare payments to providers of 2% per fiscal year, which will remain in effect through 2031. |
● | On January 2, 2013, the U.S. American Taxpayer Relief Act of 2012 was signed into law, which, among other things, further reduced Medicare payments to several types of providers and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. |
● | On April 13, 2017, CMS published a final rule that gives states greater flexibility in setting benchmarks for insurers in the individual and small group marketplaces, which may have the effect of relaxing the essential health benefits required under the ACA for plans sold through such marketplaces. |
● | On May 23, 2019, CMS published a final rule to allow Medicare Advantage Plans the option of using step therapy for Part B drugs beginning January 1, 2020. |
● | On March 11, 2021, President Biden signed the American Rescue Plan Act of 2021 into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug’s average manufacturer price, for single source and innovator multiple source drugs, beginning January 1, 2024. Due to the Statutory Pay-As-You-Go Act of 2010, estimated budget deficit increases resulting from the American Rescue Plan Act of 2021, and subsequent legislation, Medicare payments to providers will be further reduced starting in 2025 absent further legislation. |
Moreover, payment methodologies may be subject to changes in healthcare legislation and regulatory initiatives. For example, CMS may develop new payment and delivery models, such as bundled payment models. Recently, there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products. Such scrutiny has resulted in several recent U.S. Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, reduce the cost of drugs under Medicare and reform government program reimbursement methodologies for drug products. For example, at the federal level, the previous Trump administration’s budgetPresident Biden has issued multiple executive orders that have sought to reduce prescription drug costs. In February 2023, HHS also issued a proposal for fiscal year 2021in response to an October 2022 executive order from President Biden that includes a $135 billion allowance to support legislative proposals seeking to reduce drug prices, increase competition, lower out-of-pocket drug costs for patients, and increase patient access to lower-cost generic and biosimilar drugs. On March 10, 2020, the previous Trump administration sent “principles” forproposed prescription drug pricing to Congress, calling for legislationmodel that would, among other things, capwill test whether targeted Medicare Part D beneficiary out-of-pocket pharmacy expenses, provide an option to cap Medicare Part D beneficiary monthly out-of-pocket expenses, and place limits on
36
pharmaceutical price increases. Additionally, the previous Trump administration previously released a “Blueprint” to lower drug prices and reduce out of pocket costs of drugs that contained proposals to increase manufacturer competition, increase the negotiating power of certain federal healthcare programs,payment adjustments will sufficiently incentivize manufacturers to lower the list price of their products and reduce the out-of-pocket costs of drug products paid by consumers. HHS has solicited feedback on some of these measures and has implemented others under its existing authority. For example, in May 2019, CMS issued a final rule to allow Medicare Advantage Plans the option of using step therapy, a type of prior authorization,complete confirmatory trials for Part B drugs beginning January 1, 2020. This final rule codified CMS’s policy change that became effective as of January 1, 2019.approved through FDA’s accelerated approval pathway. Although a number of these and other proposed measures may require authorization through additional authorizationlegislation to become effective, and the Biden administration may reverse or otherwise change these measures, both the Biden administration and Congress hashave indicated that they will continue to seek new legislative and/or administrative measures to control drug costs.
On July 24, 2020 and September 13, 2020, then-President Trump announced
In August 2022, the IRA was signed into law. The IRA includes several executive orders relatedprovisions that will impact our business to prescription drug pricingvarying degrees, including provisions that seekcreate a $2,000 out-of-pocket cap for Medicare Part D beneficiaries, impose new manufacturer financial liability on all drugs in Medicare Part D, allow the U.S. government to implement several of the administration's proposals. In response, the FDA released a final rule on September 24, 2020, which went into effect on November 30, 2020, providing guidance for states to build and submit importation plans for drugs from Canada. Further, on November 20, 2020, CMS issued an Interim Final Rule implementing the Most Favored Nation (“MFN”) Model under whichnegotiate Medicare Part B reimbursement rates will be calculatedand Part D pricing for certain high-cost drugs and biologicals based onbiologics without generic or biosimilar competition, require companies to pay rebates to Medicare for drug prices that increase faster than inflation, and delay the lowestrebate rule that would require pass through of pharmacy benefit manager rebates to beneficiaries. Further, under the IRA, orphan drugs are exempted from the Medicare drug price drug manufacturers receive in Organizationnegotiation program, but only if they have one orphan designation and for Economic Cooperation and Development countries withwhich the only approved indication is for that disease or condition. If a similar gross domestic product per capita. The MFN Model regulations mandate participation by identified Part B providers and will apply in all U.S. states and territories for a seven-year period beginning January 1, 2021 and ending December 31, 2027. On December 28, 2020, a judge in the U.S. District Courtreceives multiple orphan designations or has multiple approved indications, it may not qualify for the Northern Districtorphan drug exemption. The implementation of California granted a preliminary injunction prohibiting CMS from implementing the MFN rule.IRA is currently subject to ongoing litigation challenging the constitutionality of the IRA’s Medicare drug price negotiation program. The effect of IRA on our business and the healthcare industry in general is not yet known.
At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing.
On May 30, 2018, the Right to Try Act, was signed into law. The law, among other things, provides a federal framework for certain patients to access certain investigational new drug products that have completed a Phase 1 clinical trial and that are undergoing investigation for FDA approval. Under certain circumstances, eligible patients can seek treatment without enrolling in clinical trials and without obtaining FDA permission under the FDA expanded access program. There is no obligation for a drug manufacturer to make its drug products available to eligible patients as a result of the Right to Try Act.
Other Healthcare Laws and Compliance Requirements
Healthcare providers, physicians, and third-party payors will play a primary role in the recommendation and prescription of any products for which we obtain marketing approval. Our business operations and any current or future arrangements with third-party payors, healthcare providers and physicians may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we develop, market, sell and distribute any drugs for which we obtain marketing approval. In the United States, these laws include, among others:
the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, receiving, offering or paying remuneration (including any kickback, bribe, or certain rebates), directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or for the purchase, lease, order or recommendation of, or arranging for, an item, good, facility or service for which payment may be made under a federal healthcare program such as Medicare and Medicaid. The term remuneration has been interpreted broadly to include anything of value. The federal Anti-Kickback Statute has been interpreted to apply to arrangements between manufacturers
37
|
● | the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, receiving, offering or paying remuneration (including any kickback, bribe, or certain rebates), directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or for the purchase, lease, order or recommendation of, or arranging for, an item, good, facility or service for which payment may be made under a federal healthcare program such as Medicare and Medicaid. The term remuneration has been interpreted broadly to include anything of value. The federal Anti-Kickback Statute has been interpreted to apply to arrangements between manufacturers on one hand and prescribers, purchasers, and formulary managers on the other. A person or entity need not have actual knowledge of the federal Anti-Kickback Statute or specific intent to violate it in order to have committed a violation. Violations are subject to significant civil and criminal fines and penalties for each violation, plus up to three times the remuneration involved, imprisonment, and exclusion from government healthcare programs. In addition, a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act ("FCA"); |
● | federal civil and criminal false claims laws, including the FCA, and civil monetary penalty laws, which prohibit, among other things, individuals or |
federal civil and criminal false claims laws, including the FCA, and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment or approval from Medicare, Medicaid, or other third-party payors, that are false, fictitious or fraudulent; knowingly making, using, or causing to be made or used, a false statement or record material to a false or fraudulent claim or obligation to pay or transmit money or property to the federal government; or knowingly concealing or knowingly and improperly avoiding or decreasing an obligation to pay money to the federal government. Manufacturers can be held liable under the FCA even when they do not submit claims directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims. The FCA also permits a private individual acting as a “whistleblower” to bring qui tam actions on behalf of the federal government alleging violations of the FCA and to share in any monetary recovery. When an entity is determined to have violated the FCA, the government may impose civil fines and penalties for each false claim, plus treble damages, and exclude the entity from participation in Medicare, Medicaid and other federal healthcare programs;
the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created additional criminal and civil liability for knowingly and willfully executing, or attempting to execute, a scheme, to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity can be found guilty of violating HIPAA without actual knowledge of the statute or specific intent to violate it;
the federal Physician Payment Sunshine Act, created under the ACA and its implementing regulations, which requires drug, device, medical supply, and biologics manufacturers to disclose payments under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report annually to HHS information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Effective January 1, 2022, these reporting obligations will extend to include transfers of value made to certain non-physician providers such as physician assistants and nurse practitioners;
HIPAA, as amended by the Health Information Technology and Clinical Health Act of 2009, or HITECH, and its implementing regulations, which imposes, among other things, certain requirements relating to the privacy, security and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to “business associates,” those independent contractors or agents of covered entities that create, receive, maintain, transmit or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions; and
38
● | the federal Health Insurance Portability and Accountability Act of 1996 ("HIPAA"), which created additional criminal and civil liability for knowingly and willfully executing, or attempting to execute, a scheme, to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity can be found guilty of violating HIPAA without actual knowledge of the statute or specific intent to violate it; |
● | the federal Physician Payment Sunshine Act, created under the ACA and its implementing regulations, which requires drug, device, medical supply, and biologics manufacturers to disclose payments under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report annually to HHS information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain other licensed healthcare practitioners and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; |
● | HIPAA, as amended by the Health Information Technology and Clinical Health Act of 2009 ("HITECH"), and its implementing regulations, which imposes, among other things, certain requirements relating to the privacy, security and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to “business associates,” those independent contractors or agents of covered entities that create, receive, maintain, transmit or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions; |
● | federal government price reporting laws, which require us to calculate and report complex pricing metrics in an accurate and timely manner to government programs; and |
● | analogous state and foreign law equivalents of each of the above U.S. federal laws, such as anti- kickback and false claims laws, which may apply to items or services reimbursed by any third-party payor, including commercial insurers or patients; state and local marketing and/or transparency laws applicable to manufacturers that may be broader in scope than the federal requirements; state laws that require the reporting of information related to drug pricing; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures and pricing information; state and local laws that require the licensure of sales representatives; state laws that require biopharmaceutical companies to comply with the biopharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government; state and local laws that require the registration of pharmaceutical sales representatives; data privacy and security laws and regulations in foreign jurisdictions that may be more stringent than those in the United States (such as the European Union, which adopted the General Data Protection Regulation, which became effective in May 2018); and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not pre-empted by HIPAA, thus complicating compliance efforts. |
analogous state and foreign law equivalents of each of the above U.S. federal laws, such as anti- kickback and false claims laws, which may apply to items or services reimbursed by any third-party payor, including commercial insurers or patients; state and local marketing and/or transparency laws applicable to manufacturers that may be broader in scope than the federal requirements; state laws that require the reporting of information related to drug pricing; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures and pricing information; state and local laws that require the licensure of sales representatives; state laws that require biopharmaceutical companies to comply with the biopharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government; state and local laws that require the registration of pharmaceutical sales representatives; data privacy and security laws and regulations in foreign jurisdictions that may be more stringent than those in the United States (such as the European Union, which adopted the General Data Protection Regulation, which became effective in May 2018); and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not pre-empted by HIPAA, thus complicating compliance efforts.
The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of healthcare reform, especially in light of the lack of applicable precedent and regulations. Federal and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to administrative, civil, and criminal penalties, exclusion from participation in government healthcare programs, such as Medicare and Medicaid, imprisonment, damages, fines, disgorgement, reputational harm, the curtailment or restructuring of our operations, and additional oversight and reporting obligations if we become subject to a corporate integrity agreement or similar settlement to resolve allegations of non-compliance with these laws and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations. If any of the physicians or other healthcare providers or entities with whom we expect to do business is found to be not in compliance with applicable laws, they may be subject to similar actions, penalties and sanctions. Ensuring business arrangements comply with applicable healthcare laws, as well as responding to possible investigations by government authorities, can be time- and resource-consuming and can divert a company’s attention from its business.
We are also subject to the U.S. Foreign Corrupt Practices Act or FCPA,("FCPA"), which prohibits improper payments or offers of payments to foreign governments and their officials for the purpose of obtaining or retaining business and requires companies to maintain accurate books and records and a system of internal accounting controls. Safeguards we implement to discourage improper payments or offers of payments by our employees, consultants, and others may be ineffective, and violations of the FCPA and similar laws may result in severe criminal or civil sanctions, or other liabilities or proceedings against us, any of which would likely harm our reputation, business, financial condition and result of operations.
In the event we decide to conduct future clinical trials in the European Union, we may be subject to additional privacy restrictions. The collection, use, storage, disclosure, transfer, or other processing of personal data regarding individuals in the European Economic Area, or EEA, including personal health data, is subject to the EU General Data Protection Regulation, or GDPR, which became effective on May 25, 2018. The GDPR is wide-ranging in scope and imposes numerous requirements on companies that process personal data, including requirements relating to processing health and other sensitive data, obtaining consent of the individuals to whom the personal data relates, providing information to individuals regarding data processing activities, implementing safeguards to protect the security and confidentiality of personal data, providing notification of data breaches, and taking certain measures when engaging third-party processors. The GDPR also imposes strict rules on the transfer of personal data to
39
countries outside the EEA, including the United States, and permits data protection authorities to impose large penalties for violations of the GDPR, including potential fines of up to €20 million or 4% of annual global revenues, whichever is greater. The GDPR also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory authorities, seek judicial remedies, and obtain compensation for damages resulting from violations of the GDPR. In addition, the GDPR includes restrictions on cross-border data transfers. The GDPR may increase our responsibility and liability in relation to personal data that we process where such processing is subject to the GDPR, and we may be required to put in place additional mechanisms to ensure compliance with the GDPR, including as implemented by individual countries. Compliance with the GDPR will be a rigorous and time-intensive process that may increase our cost of doing business or require us to change our business practices, and despite those efforts, there is a risk that we may be subject to fines and penalties, litigation, and reputational harm in connection with our European activities. Further, the United Kingdom’s decision to leave the European Union, often referred to as Brexit, has created uncertainty with regard to data protection regulation in the United Kingdom. In particular, it is unclear how data transfers to and from the United Kingdom will be regulated now that the United Kingdom has left the European Union.
Employees and Human Capital
As of December 31, 2020,2023, we had 31 employees,32 employees,19 of which all were full-time employees, 19 were primarily engaged in research and development activities and 13 hold M.D. or Ph.D. degrees.clinical activities. None of our employees isare represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be good.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing and integrating our existing and new employees, advisors and consultants. The principal purposes of our equity and cash incentive plans are to attract, retain and reward personnel through the granting of stock-based and cash-based compensation awards, in order to increase shareholder value and the success of our company by motivating such individuals to perform to the best of their abilities and achieve our objectives.
As an emerging company operating in a competitive industry, much of our success is rooted in the diversity of our teams and our commitment to inclusion. We value diversity at all levels and continue to focus on extending our diversity and inclusion initiatives across our entire workforce, from working with managers to develop strategies for building diverse teams to promoting the advancement of leaders from different backgrounds.
Corporate Information
We were incorporated as Olivia Ventures, Inc. in the State of Delaware on March 20, 2018. On June 17, 2020, a wholly-owned subsidiary of ours merged with and into Compass Therapeutics, a private limited liability company formed in 2014. Following the Merger, Compass Therapeutics was the surviving entity and became our wholly-owned subsidiary, and all of the outstanding common and preferred membership interests of Compass Therapeutics were converted into shares of our common stock. On June 17, 2020, we changed our name to Compass Therapeutics, Inc. As a result of the Merger, we acquired the business of Compass Therapeutics and we will continue the existing business operations of Compass Therapeutics as a public reporting company under the name Compass Therapeutics, Inc.
Our principal executive offices are located at 80 Guest Street, Suite 601, Boston, Massachusetts 02135, and our telephone number is (617) 500-8099.
Available Information
Our website address is www.compasstherapeutics.com.www.compasstherapeutics.com. Our Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and any amendments to such reports are filed with the SEC. We are subject to the informational requirements of the Exchange Act and file or furnish reports, proxy
40
statements, and other information with the SEC. Such reports and other information filed by us with the SEC will be available free of charge on our website at www.compasstherapeutics.com when such reports are available on the SEC’s website. The SEC maintains a website that contains reports, proxy and information statements, and other information that issuers file electronically with the SEC at www.sec.gov.
The contents of the websites referred to above are not incorporated into this filing. Further, our references to the URLs for these websites are intended to be inactive textual references only.
41
Investing in our common stock involves a high degree of risk. You should carefully consider the risks described below together with all of the other information in this Annual Report on Form 10-K or ("Form 10-K,10-K"), including our financial statements and the related notes and the information described in the section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and in our other filings with the SEC. If any of the events described below actually occurs, our business, results of operations, financial conditions, cash flows or prospects could be harmed. If that were to happen, you could lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also impair our business.
Risks Related to Our Financial Position and Need for Additional Capital
We have a limited operating history and no products approved for commercial sale. We have a history of significant losses, expect to continue to incur significant losses for the foreseeable future and may never achieve or maintain profitability.
We are a clinical-stage biopharmaceutical company with a limited operating history. Since our founding in 2014, we have incurred significant net losses. Our net losses were $29.5with an accumulated deficit of $315 million and $34.7 million for the years ended December 31, 2020 and 2019, respectively, and as of December 31, 2020, we had an accumulated deficit of $151.4 million. In addition, as of December 31, 2020, we had stockholders’ equity of $39.9 million.2023. We have funded our operations to date primarily with proceeds from private placements of preferred and common equity, an underwritten public offering and borrowingsa PIPE offering, as well as sales under the 2018 loan and security agreementour at-the-market offering program pursuant to an Open Market Sale AgreementSM with Pacific Western Bank, or the 2018 Credit Facility.Jefferies LLC. Since commencing operations, we have devoted substantially all of our efforts and financial resources to organizing and staffing our company, identifying business development opportunities, raising capital, securing intellectual property rights related to our product candidates, conducting discovery, and research and development activities, including clinical development, for our product candidates.
We expect that it will be several years, if ever, before we have a commercialized product. We expect to continue to incur significant expenses and operating losses for the foreseeable future. The net losses we incur may fluctuate significantly from quarter to quarter. We anticipate that our expenses will increase substantially if, and as, we:
continue to advance the preclinical and clinical development of our existing product candidates and our research programs;
● |
|
● | leverage our research and development capabilities, including our proprietary StitchMabsTM technology, to advance additional product candidates into preclinical and clinical development; |
● | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
● | hire additional clinical, quality control, regulatory, scientific and administrative personnel; |
● | expand our operational, financial and management systems and increase personnel, including to support our clinical development and our operations as a public company; |
● | maintain, expand and protect our intellectual property portfolio; |
● | establish a marketing, sales, distribution and medical affairs infrastructure to commercialize any products for which we may obtain marketing approval and commercialize, whether on our own or jointly with a partner; |
● | acquire or in-license other technologies or engage in strategic partnerships; and |
seek regulatory approvals for any product candidates that successfully complete clinical trials;
hire additional clinical, quality control, regulatory, scientific and administrative personnel;
expand our operational, financial and management systems and increase personnel, including to support our clinical development and our operations as a public company;
maintain, expand and protect our intellectual property portfolio;
establish a marketing, sales, distribution and medical affairs infrastructure to commercialize any products for which we may obtain marketing approval and commercialize, whether on our own or jointly with a partner;
acquire or in-license other technologies or engage in strategic partnerships; and
42
● | incur additional legal, accounting or other expenses in operating our business, including the additional costs associated with operating as a public company. |
incur additional legal, accounting or other expenses in operating our business, including the additional costs associated with operating as a public company.
To become and remain profitable, we must develop and eventually commercialize products with significant market potential. This will require us to be successful in a range of challenging activities, including completing preclinical studies and clinical trials, obtaining marketing approval for product candidates, manufacturing, marketing and selling products and satisfying any post-marketing requirements. We may never succeed in any or all of these activities and, even if we do, we may never generate revenue that is significant or large enough to achieve profitability. If we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain profitable would decrease the value of our company and could impair our ability to raise capital, maintain our research and development efforts, expand our business or continue our operations. A decline in the value of our company could also cause you to lose all or part of your investment.
We have never generated revenue from product sales and may never be profitable.
Our ability to generate revenue from product sales and achieve profitability depends on our ability, alone or with our collaboration partners, to successfully complete the development of, and obtain the regulatory approvals necessary to commercialize, our product candidates. We do not anticipate generating revenue from product sales for the next several years, if ever. Our ability to generate future revenue from product sales depends heavily on our, or our existing or future collaborators’, success in:
● | completing preclinical studies and clinical trials of our product candidates; |
● | seeking and obtaining marketing approvals for any product candidates that we or our collaborators develop; |
● | identifying and developing new product candidates; |
● | launching and commercializing product candidates for which we obtain marketing approval by establishing a marketing, sales, distribution and medical affairs infrastructure or, alternatively, collaborating with a commercialization partner; |
● | achieving coverage and adequate reimbursement by hospitals and third-party payors, including governmental authorities, such as Medicare and Medicaid, private insurers and managed care organizations, for product candidates, if approved, that we or our collaborators develop; |
● | obtaining market acceptance of product candidates, if approved, that we develop as viable treatment options; |
● | addressing any competing technological and market developments; |
● | negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter and performing our obligations under such arrangements; |
● | maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how; |
● | defending against third-party interference or infringement claims, if any; and |
● | attracting, hiring and retaining qualified personnel. |
completing preclinical studies and clinical trials of our product candidates, including our ongoing Phase 1 clinical trial of CTX-471 as a monotherapy and other clinical trials for CTX-471 and CTX-8371;
seeking and obtaining marketing approvals for any product candidates that we or our collaborators develop;
identifying and developing new product candidates;
launching and commercializing product candidates for which we obtain marketing approval by establishing a marketing, sales, distribution and medical affairs infrastructure or, alternatively, collaborating with a commercialization partner;
achieving coverage and adequate reimbursement by hospitals and third-party payors, including governmental authorities, such as Medicare and Medicaid, private insurers and managed care organizations, for product candidates, if approved, that we or our collaborators develop;
obtaining market acceptance of product candidates, if approved, that we develop as viable treatment options;
addressing any competing technological and market developments;
negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter and performing our obligations under such arrangements;
maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how;
defending against third-party interference or infringement claims, if any; and
attracting, hiring and retaining qualified personnel.
43
We anticipate incurring significant costs associated with commercializing any product candidate that is approved for commercial sale. Our expenses could increase beyond expectations if we are required by the FDA or other regulatory agencies to perform clinical trials or studies in addition to those that we currently anticipate, or if there are any delays in establishing appropriate manufacturing arrangements for or in completing our clinical trials for the development of any of our product candidates, for example, as a result of any setbacks or delays due to the COVID-19 pandemic.candidates. Even if we are able to generate revenue from the sale of any approved products, we may not become profitable and may need to obtain additional funding to continue operations.
We will require substantial additional financing to pursue our business objectives, which may not be available on acceptable terms, or at all. A failure to obtain this necessary capital when needed could force us to delay, limit, reduce or terminate our product development, commercialization efforts or other operations.
Our operations have consumed substantial amounts of cash since inception. We expect to continue to spend substantial amounts to continue the preclinical and clinical development of our current and future programs. If we receive marketing approval for any product candidates, including CTX-471 and CTX-8371, we will require significant additional amounts of cash in order to launch and commercialize such product candidates. In addition, other unanticipated costs may arise. Because the designs and outcomes of our planned and anticipated clinical trials are highly uncertain, we cannot reasonably estimate the actual amounts necessary to successfully complete the development of and commercialize any product candidate we develop. Additionally, any COVID-19 related program setbacks or delays due to changes in federal, state, or local laws and regulations or clinical site policies could impact our programs and increase our expenditures.
Our future capital requirements depend on many factors, including:
the scope, progress, timing, results and costs of researching and developing CTX-471, CTX-8371 and our other product candidates, and of conducting preclinical studies and clinical trials;
the timing of, and the costs involved in, obtaining marketing approval for CTX-471, CTX-8371 and any future product candidates we develop, if clinical trials are successful;
the costs of manufacturing CTX-471, CTX-8371 and any future product candidates for preclinical studies and clinical trials and in preparation for marketing approval and commercialization;
the impact of COVID-19 on the initiation or completion of preclinical studies or clinical trials, the third-parties on whom we rely, and the supply of our product candidates;
the costs of commercialization activities, including marketing, sales and distribution costs, for CTX-471, CTX-8371 and any future product candidates we develop, whether alone or with a collaborator, if any of these product candidates are approved for sale;
our ability to establish and maintain strategic collaborations, licensing or other arrangements on favorable terms, if at all;
the costs involved in preparing, filing, prosecuting, maintaining, expanding, defending and enforcing patent claims, including litigation costs and the outcome of any such litigation;
our current collaboration and license agreements remaining in effect and our achievement of milestones and the timing and amount of milestone payments we are required to make, or that we may be eligible to receive, under those agreements;
the timing, receipt and amount of sales of, on our future products, if any; and
44
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
● | the scope, progress, timing, results and costs of researching and developing our product candidates, and of conducting preclinical studies and clinical trials; |
the emergence of competing therapies and other adverse developments in the oncology and immunology market.
● | the timing of, and the costs involved in, obtaining marketing approval for any of our current or future product candidates we develop, if clinical trials are successful; |
● | the costs of manufacturing our current and any future product candidates for preclinical studies and clinical trials and in preparation for marketing approval and commercialization; |
● | the costs of commercialization activities, including marketing, sales and distribution costs, for our products and any future product candidates we develop, whether alone or with a collaborator, if any of these product candidates are approved for sale; |
● | our ability to establish and maintain strategic collaborations, licensing or other arrangements on favorable terms, if at all; |
● | the costs involved in preparing, filing, prosecuting, maintaining, expanding, defending and enforcing patent claims, including litigation costs and the outcome of any such litigation; |
● | our current collaboration and license agreements remaining in effect and our achievement of milestones and the timing and amount of milestone payments we are required to make, or that we may be eligible to receive, under those agreements; |
● | the timing, receipt and amount of sales of, on our future products, if any; and |
● | the emergence of competing therapies and other adverse developments in the oncology and immunology market. |
Until we can generate sufficient product revenue to finance our cash requirements, which we may never do, we expect to finance our future cash needs through a combination of public or private equity and debt financings, marketing and distribution arrangements, other collaborations, strategic alliances and licensing arrangements. As of December 31, 2020,2023, we had $47.1$152.5 million in cash, cash equivalents and cash equivalents.marketable securities. Based on our research and development plans, we expect that these cash resources will enable us to fund our operating expenses and capital expenditure requirements into the second quarter of 2022.mid-2026. This estimate is based on assumptions that may prove to be wrong, and we could use our available capital resources sooner than we expect. Changes may occur beyond our control that would cause us to consume our available capital before that time, including changes in, and progress of, our development activities, acquisitions of additional product candidates and changes in regulation.
If we raise additional capital through marketing, sales and distribution arrangements or other collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish certain valuable rights to our product candidates, future revenue streams or research programs, technologies or grant licenses on terms that may not be favorable to us. If we raise additional capital through public or private equity offerings, the terms of these securities may include liquidation or other preferences that adversely affect our stockholders’ rights. Further, to the extent that we raise additional capital through additional sales of common stock or securities convertible or exchangeable into common stock, investors’ ownership interest will be diluted. If we raise additional capital through debt financing, we would be subject to fixed payment obligations and may be subject to covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
Adequate additional financing may not be available to us on acceptable terms, or at all. If we are unable to obtain additional financing on favorable terms when needed, we may be required to delay, limit, reduce or terminate preclinical studies, clinical trials, or other research and development activities or one or more of our development programs.
The current economic downturn and inflationary environment may harm our business and results of operations.
Our overall performance depends, in part, on worldwide economic conditions. In recent months, we have observed increased economic uncertainty in the United States and abroad. Impacts of such economic weakness include reduced credit availability, higher borrowing costs, reduced liquidity, volatility in credit, equity and foreign exchange markets, and bankruptcies. These developments could lead to supply chain disruption, inflation, higher interest rates, and uncertainty about business continuity, which may adversely affect our business.
Recent volatility in capital markets and lower market prices for our securities may affect our ability to access new capital through sales of shares of our common stock or issuance of indebtedness, which may harm our liquidity and limit our ability to grow our business, pursue acquisitions, or improve our operating infrastructure.
Our operations consume substantial amounts of cash, and we intend to continue to make significant investments to support our business growth, respond to business challenges or opportunities, develop our product candidates, retain or expand our current levels of personnel, enhance our operating infrastructure, and potentially acquire complementary businesses and technologies. Our future capital requirements may be significantly different from our current estimates and will depend on many factors, including the need to:
● | finance unanticipated working capital requirements; |
● | develop our product candidates; |
● | pursue acquisitions or other strategic relationships; and |
● | respond to competitive pressures. |
Accordingly, we may need to pursue equity or debt financings to meet our capital needs. With uncertainty in the capital markets and other factors, such financing may not be available on terms favorable to us or at all. If we raise additional funds through further issuances of equity or convertible debt securities, our existing stockholders could suffer significant dilution, and any new equity securities we issue could have rights, preferences, and privileges superior to those of holders of our common stock. Any debt financing secured by us in the future could involve additional restrictive covenants relating to our capital-raising activities and other financial and operational matters, which may make it more difficult for us to obtain additional capital and to pursue business opportunities, including potential acquisitions. If we are unable to obtain adequate financing or financing on terms satisfactory to us, we could face significant limitations on our ability to invest in our operations and otherwise suffer harm to our business.
Risks Related to the Discovery and Development of Our Product Candidates
Our business is dependent on our ability to advance our current and future product candidates through clinical trials, obtain marketing approval and ultimately commercialize them.
We are early in our development efforts. We are conducting our first clinical trial for CTX-471, our lead product candidate. Our ability to generate product revenues, which we do not expect will occur for several years, if ever, will depend heavily on the successful development and eventual commercialization of CTX-471, CTX-8371 and any otherour current products or future product candidates we develop, which may never occur. Our current product candidates including CTX-471, CTX-8371 and any future product candidates we develop will require additional preclinical or clinical development, management of clinical, preclinical and manufacturing activities, marketing approval in the United States and other jurisdictions, demonstration of effectiveness to pricing and reimbursement authorities, sufficient cGMP manufacturing supply for both preclinical and clinical development and commercial production, building of a commercial organization and substantial investment and significant marketing efforts before we generate any revenues from product sales.
The clinical and commercial success of our current and future product candidates will depend on several factors, including the following:
● | timely and successful completion of preclinical studies and our clinical trials; |
● | sufficiency of our financial and other resources to complete the necessary preclinical studies and clinical trials; |
● | our plans to successfully submit new INDs with the FDA for our current and future product candidates; |
● | our ability to complete preclinical studies for current or future product candidates; |
● | successful enrollment in, and completion of clinical trials; |
● | successful data from our clinical program that supports an acceptable risk-benefit profile of our product candidates in the intended patient populations; |
● | our ability to establish agreements with third-party manufacturers on a timely and cost-efficient manner; |
● | whether we are required by the FDA or comparable foreign regulatory authorities to conduct additional clinical trials or other studies beyond those planned or anticipated to support approval of our product candidates; |
● | acceptance of our proposed indications and the primary endpoint assessments evaluated in the clinical trials of our product candidates by the FDA and comparable foreign regulatory authorities; |
timely and successful completion of preclinical studies and our clinical trials;
sufficiency of our financial and other resources to complete the necessary preclinical studies and clinical trials;
45
our plans to successfully submit investigational new drug, or IND, applications with the FDA for CTX-471, CTX-8371 and/or other current and future product candidates;
our ability to complete preclinical studies for current or future product candidates;
successful enrollment in, including maintaining or reaching target enrollment levels during the COVID-19 pandemic, and completion of clinical trials;
successful data from our clinical program that supports an acceptable risk-benefit profile of our product candidates in the intended patient populations;
our ability to establish agreements with third-party manufacturers on a timely and cost-efficient manner;
whether we are required by the FDA or comparable foreign regulatory authorities to conduct additional clinical trials or other studies beyond those planned or anticipated to support approval of our product candidates;
acceptance of our proposed indications and the primary endpoint assessments evaluated in the clinical trials of our product candidates by the FDA and comparable foreign regulatory authorities;
receipt and maintenance of timely marketing approvals from applicable regulatory authorities;
successfully launching commercial sales of our product candidates, if approved;
the prevalence, duration and severity of potential side effects or other safety issues experienced with our product candidates, if approved;
entry into collaborations to further the development of our product candidates;
obtaining and maintaining patent and trade secret protection or regulatory exclusivity for our product candidates;
acceptance of the benefits and uses of our product candidates, if approved, by patients, the medical community and third-party payors;
maintaining a continued acceptable safety, tolerability and efficacy profile of the product candidates following approval;
our compliance with any post-approval requirements imposed on our products, such as post-marketing studies, a Risk Evaluation and Mitigation Strategy, or REMS, or additional requirements that might limit the promotion, advertising, distribution or sales of our products or make the products cost prohibitive;
competing effectively with other therapies;
obtaining and maintaining healthcare coverage and adequate reimbursement from third-party payors;
our ability to identify bispecifics; and
enforcing and defending intellectual property rights and claims.
46
● | receipt and maintenance of timely marketing approvals from applicable regulatory authorities; |
● | successfully launching commercial sales of our product candidates, if approved; |
● | the prevalence, duration and severity of potential side effects or other safety issues experienced with our product candidates, if approved; |
● | entry into collaborations to further the development of our product candidates; |
● | obtaining and maintaining patent and trade secret protection or regulatory exclusivity for our product candidates; |
● | acceptance of the benefits and uses of our product candidates, if approved, by patients, the medical community and third-party payors; |
● | maintaining a continued acceptable safety, tolerability and efficacy profile of the product candidates following approval; |
● | our compliance with any post-approval requirements imposed on our products, such as post-marketing studies, REMS or additional requirements that might limit the promotion, advertising, distribution or sales of our products or make the products cost prohibitive; |
● | competing effectively with other therapies; |
● | obtaining and maintaining healthcare coverage and adequate reimbursement from third-party payors; |
● | our ability to identify bispecifics; and |
● | enforcing and defending intellectual property rights and claims. |
These factors, many of which are beyond our control, could cause us to experience significant delays or an inability to obtain regulatory approvals or commercialize our current or future product candidates, and could otherwise materially harm our business. Successful completion of preclinical studies and clinical trials does not mean that CTX-471, CTX-8371 or any other current or future product candidates we develop will receive regulatory approval. Even if regulatory approvals are obtained, we could experience significant delays or an inability to successfully commercialize our current and any future product candidates we develop, which would materially harm our business. If we are not able to generate sufficient revenue through the sale of any current or future product candidate, we may not be able to continue our business operations or achieve profitability.
Clinical development involves a lengthy and expensive process with uncertain outcomes. We may incur additional costs and experience delays in developing and commercializing or be unable to develop or commercialize our current and future product candidates.
To obtain the requisite regulatory approvals to commercialize any of our product candidates, we must demonstrate through extensive preclinical studies and clinical trials that our product candidates are safe, pure and potent in humans. Clinical testing is expensive and can take many years to complete, and its outcome is highly uncertain. Failure can occur at any time during the clinical trial process and our future clinical trial results may not be successful. We may experience delays in completing our clinical trials or preclinical studies and initiating or completing additional clinical trials. Although we have completed the Phase 1a stage of our clinical trial of CTX-471 and are in the process of conducting the Phase 1b stage of the trial, we may experience delays in completing this trial or in initiating any planned clinical trials and development efforts. Additionally, weWe cannot be certain the ongoing and planned preclinical studies or clinical trials for CTX-471, CTX-8371our current or any other future product candidates will begin on time, not require redesign, enroll an adequate number of subjects on time or be completed on schedule, if at all. We may also experience numerous unforeseen events during our clinical trials that could delay or prevent our ability to receive marketing approval or commercialize the product candidates we develop, including:
results from preclinical studies or clinical trials may not be predictive of results from later clinical trials of any product candidate;
the FDA or other regulatory authorities, Institutional Review Boards, or IRBs, or independent ethics committees may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site;
the FDA or other regulatory authorities may require us to submit additional data such as long-term toxicology studies, or impose other requirements on us, before permitting us to initiate a clinical trial;
we may experience delays in reaching, or fail to reach, agreement on acceptable terms with prospective trial sites and prospective contract research organizations, or CROs, as the terms of these agreements can be subject to extensive negotiation and vary significantly among different CROs and trial sites;
● | results from preclinical studies or clinical trials may not be predictive of results from later clinical trials of any product candidate; |
clinical trials of any product candidate may fail to show safety, purity or potency, or may produce negative or inconclusive results, which may cause us to decide, or regulators to require us, to conduct additional nonclinical studies or clinical trials or which may cause us to decide to abandon product candidate development programs;
● | the FDA or other regulatory authorities, Institutional Review Boards ("IRBs"), or independent ethics committees may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site; |
● | the FDA or other regulatory authorities may require us to submit additional data such as long-term toxicology studies, or impose other requirements on us, before permitting us to initiate a clinical trial; |
● | we may experience delays in reaching, or fail to reach, agreement on acceptable terms with prospective trial sites and prospective contract research organizations ("CROs"), as the terms of these agreements can be subject to extensive negotiation and vary significantly among different CROs and trial sites; |
● | clinical trials of any product candidate may fail to show safety, purity or potency, or may produce negative or inconclusive results, which may cause us to decide, or regulators to require us, to conduct additional nonclinical studies or clinical trials or which may cause us to decide to abandon product candidate development programs; |
Critical Accounting Policies and Significant Judgments and Estimates
Our consolidated financial statements are prepared in accordance with generally accepted accounting principles in the United States or GAAP.("GAAP"). The preparation of our consolidated financial statements and related disclosures requires us to make estimates and judgments that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amount of revenue and expenses during the reporting period. We base our estimates on historical experience, known trends and events, and various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in Note 3 to our consolidated financial statements appearing in this Form 10-K, we believe that the following accounting policies are the most critical to the judgments and estimates used in the preparation of our consolidated financial statements.
Research and Development Expenses
Research and development expenses consist primarily of costs incurred in connection with the development of our product candidates. We expense research and development costs as incurred.
At the end of each reporting period, we compare payments made to third-party service providers to the estimated progress toward completion of the applicable research or development objectives. Such estimates are subject to change as additional information becomes available. Depending on the timing of payments to the service providers and the progress that we estimate has been made as a result of the service provided, we may record net prepaid or accrued expenses relating to these costs. As of December 31, 2020, we have not made any material adjustments to our prior estimates of accrued research and development expenses.
Valuation of Derivative Liability
Our derivative liability is comprised of the contingent interest rate reset features and a contingent feature to pay a success fee upon the occurrence of certain liquidity events under the 2018 Credit Facility. At issuance and at each reporting period, we are required to estimate the fair value of the derivative liability using a probability-weighted expected return method. This method requires judgment when estimating the timing and probability of future events, such as a change in control event, future liquidity events, and repayment of our debt obligation under the 2018 Credit Facility. We then apply a risk-adjusted discount rate reflecting the expected risk profile for each of the potential settlement scenarios and relating timing. Due to the nature of and inputs in the model used to assess the fair value of the future tranche rights, it is not abnormal to experience significant fluctuations during each remeasurement period. The contingent success fee derivative was settled at the time of the Merger in June 2020.
114
Stock Awards and Unit-Based Compensation
The following table summarizes stock awards and unit-based compensation expense:
|
| Year Ended December 31, |
| Year Ended | ||||||||||||
|
| 2020 |
|
| 2019 |
| 2023 | 2022 | ||||||||
|
| (000's) |
| (000's) | ||||||||||||
Research and development |
| $ | 605 |
|
| $ | 383 |
| $ | 1,998 | $ | 1,793 | ||||
General and administrative |
|
| 3,411 |
|
|
| 532 |
| 4,122 | 3,537 | ||||||
Total unit-based compensation |
| $ | 4,016 |
|
| $ | 915 |
| ||||||||
Total | $ | 6,120 | $ | 5,330 | ||||||||||||
See Notes 3 and 9 to our consolidated financial statements appearing in this Form 10-K for additional stock compensation information.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
Recently Issued and Adopted Accounting Pronouncements
A description of recently issued accounting pronouncements that may potentially impact our financial position and results of operations is disclosed in Note 3 to our consolidated financial statements appearing in this Annual Report.
JOBS Act
In April 2012, the Jumpstart Our Business Startups Act of 2012 or the JOBS Act,(the "JOBS Act") was enacted. Under Section 107(b) of the JOBS Act, an “emerging growth company” can delay the adoption of new or revised accounting standards until such time as those standards would apply to private companies. We have made the election to delay the adoption of such accounting standards as provided in the JOBS Act. There are other exemptions and reduced reporting requirements provided by the JOBS Act that we are currently evaluating. For example, as an “emerging growth company”, we are exempt from Sections 14A(a) and (b) of the Exchange Act that would otherwise require us to (i) submit certain executive compensation matters to stockholder advisory votes, such as “say-on-pay”, “say-on-frequency”, and “golden parachutes”; and (ii) disclose certain executive compensation related items such as the correlation between executive compensation and performance and comparisons of our Chief Executive Officer’s compensation to our median employee compensation. We also intend to rely on an exemption from the rule requiring us to provide an auditor’s attestation report on our internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act. We will continue to remain an “emerging growth company” until the earliest of the following: (i) the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement; (ii) the last day of the fiscal year in which our total annual gross revenue is equal to or more than $1.07$1.235 billion; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
115
Item 7A. Quantitative and QualitativeQualitative Disclosures About Market Risk.
Our cash is held on deposit in demand accounts at a large financial institution in amounts in excess of the Federal Deposit Insurance Corporation or FDIC,("FDIC") insurance coverage limit of $250,000 per depositor, per FDIC-insured bank, per ownership category. We have reviewed the consolidated financial statements of this institution and believe it has sufficient assets and liquidity to conduct its operations in the ordinary course of business with little or no credit risk to us. Financial instruments that potentially subject us to concentrations of credit risk principally consist of cash equivalents.equivalents and marketable securities including corporate bonds, commercial paper certificates of deposit, U.S. government treasuries and asset backed securities, all of which are subject to interest rate risk. We limit our credit risk associated with cash equivalents by placing investmentsinvesting in highly-rated money market funds.
As discussed above under “—Liquidity and Capital Resources—Indebtedness”,securities. Changes in the 2018 Credit Facility bears interest at a floating interest rate, which resets monthly and is equal to the greatergeneral level of 6.25% and 1.5%, plus the prime rate as published by The Wall Street Journal. As a result, we are exposed to risks from changes in interest rates. A 1.0% increase in interest rates would have resultedcan affect the fair value of our investment portfolio. If interest rates in the general economy were to change, our holdings could change as a $0.1 million increase to our interest expense for the year ended December 31, 2020.result.
116
Item 8. Financial Statements and Supplementary Data.
COMPASS THERAPEUTICS, INC. AND SUBSIDIARIES
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| Page(s) |
| |
Consolidated Financial Statements | |
| |
Consolidated Statements of Operations and Comprehensive Loss |
|
Consolidated Statements of Changes in |
|
| |
|
117
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders
Compass Therapeutics, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Compass Therapeutics, Inc. and subsidiaries (the “Company”) as of December 31, 20202023 and 2019, and2022, the related consolidated statements of operations and comprehensive loss, changes in convertible preferred stock and stockholders’ equity (deficit), and cash flows for each of the two years thenin the period ended December 31, 2023, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 20202023 and 2019,2022, and the results of its operations and its cash flows foreach of the two years thenin the period ended December 31, 2023 in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) ("PCAOB") and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’sentity's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ CohnReznick LLP
We have served as the Company’s auditor since March 2020.
Hartford, ConnecticutMelville, New York
March 5, 202121, 2024
118
Compass Therapeutics, Inc. and Subsidiaries
(In thousands, except par value per share data)
|
| December 31, |
| |||||
|
| 2020 |
|
| 2019 |
| ||
|
|
|
|
|
|
|
|
|
Assets |
|
|
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
| $ | 47,076 |
|
| $ | 25,303 |
|
Prepaid expenses and other current assets |
|
| 3,126 |
|
|
| 935 |
|
Total current assets |
|
| 50,202 |
|
|
| 26,238 |
|
Property and equipment, net |
|
| 1,126 |
|
|
| 3,751 |
|
Restricted cash |
|
| 263 |
|
|
| 263 |
|
Other assets |
|
| 320 |
|
|
| 129 |
|
Total assets |
| $ | 51,911 |
|
| $ | 30,381 |
|
Liabilities and Stockholders' Equity (Deficit) |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
| $ | 1,061 |
|
| $ | 629 |
|
Accrued expenses |
|
| 1,571 |
|
|
| 3,122 |
|
Current portion of long-term debt |
|
| 7,467 |
|
|
| 5,576 |
|
Derivative liability related to loan |
|
| — |
|
|
| 494 |
|
Total current liabilities |
|
| 10,099 |
|
|
| 9,821 |
|
Long-term debt, net of current portion |
|
| 1,867 |
|
|
| 9,293 |
|
Total liabilities |
|
| 11,966 |
|
|
| 19,114 |
|
Commitments and Contingencies (Note 11) |
|
|
|
|
|
|
|
|
Convertible preferred stock - 207,164 authorized, issued, and outstanding as of December 31, 2019. No shares authorized, issued, and outstanding as of December 31, 2020. |
|
| — |
|
|
| 129,870 |
|
Stockholders' equity (deficit): |
|
|
|
|
|
|
|
|
Preferred stock, $0.0001 par value: 10,000 shares authorized; no shares issued and outstanding as of December 31, 2020 and 2019. |
|
| — |
|
|
| — |
|
Common stock, $0.0001 par value: 300,000 shares authorized; 52,117 and 9,073 shares issued at December 31, 2020 and 2019, respectively; 51,221 and 7,034 shares outstanding at December 31, 2020 and 2019, respectively. |
| 5 |
|
| 1 |
| ||
Additional paid-in-capital |
|
| 191,348 |
|
|
| 3,304 |
|
Accumulated deficit |
|
| (151,408 | ) |
|
| (121,908 | ) |
Total stockholders' equity (deficit) |
|
| 39,945 |
|
|
| (118,603 | ) |
Total liabilities and stockholders' equity (deficit) |
| $ | 51,911 |
|
| $ | 30,381 |
|
The accompanying notes are an integral part of these consolidated financial statements.
119
Compass Therapeutics, Inc. and Subsidiaries
Consolidated Statements of Operations
(In thousands, except per share data)
|
| Year Ended December 31, |
| |||||
|
| 2020 |
|
| 2019 |
| ||
Operating expenses: |
|
|
|
|
|
|
|
|
Research and development |
| $ | 14,904 |
|
| $ | 22,449 |
|
General and administrative |
|
| 12,908 |
|
|
| 11,603 |
|
Total operating expenses |
|
| 27,812 |
|
|
| 34,052 |
|
Loss from operations |
|
| (27,812 | ) |
|
| (34,052 | ) |
Other income (expense): |
|
|
|
|
|
|
|
|
Interest expense |
|
| (908 | ) |
|
| (1,228 | ) |
Other income (expense), net |
|
| (748 | ) |
|
| 627 |
|
Total other expense |
|
| (1,656 | ) |
|
| (601 | ) |
Loss before income tax expense |
|
| (29,468 | ) |
|
| (34,653 | ) |
Income tax expense |
|
| (32 | ) |
|
| (91 | ) |
Net loss |
| $ | (29,500 | ) |
| $ | (34,744 | ) |
Net loss per share - basic and diluted |
| $ | (0.96 | ) |
| $ | (5.19 | ) |
Basic and diluted weighted average shares outstanding |
|
| 30,776 |
|
|
| 6,691 |
|
December 31, | ||||||||
2023 | 2022 | |||||||
Assets | ||||||||
Current assets: | ||||||||
Cash and cash equivalents | $ | 24,228 | $ | 34,946 | ||||
Marketable securities | 128,233 | 151,663 | ||||||
Prepaid expenses and other current assets | 1,420 | 8,182 | ||||||
Total current assets | 153,881 | 194,791 | ||||||
Property and equipment, net | 898 | 1,567 | ||||||
Operating lease, right-of-use ("ROU") asset | 1,776 | 2,967 | ||||||
Other assets | 320 | 320 | ||||||
Total assets | $ | 156,875 | $ | 199,645 | ||||
Liabilities and Stockholders' Equity | ||||||||
Current liabilities: | ||||||||
Accounts payable | $ | 4,090 | $ | 3,382 | ||||
Accrued expenses | 2,514 | 11,690 | ||||||
Operating lease obligations, current portion | 1,197 | 1,097 | ||||||
Total current liabilities | 7,801 | 16,169 | ||||||
Operating lease obligations, net of current portion | 536 | 1,838 | ||||||
Total liabilities | 8,337 | 18,007 | ||||||
Commitments and Contingencies (Note 11) | ||||||||
Stockholders' equity: | ||||||||
Common stock, $0.0001 par value: 300,000 shares authorized; 127,668 shares issued and outstanding at December 31, 2023; 126,495 shares issued and 126,302 shares outstanding at December 31, 2022 | 13 | 13 | ||||||
Additional paid-in-capital | 463,796 | 454,741 | ||||||
Accumulated other comprehensive income (loss) | 37 | (302 | ) | |||||
Accumulated deficit | (315,308 | ) | (272,814 | ) | ||||
Total stockholders' equity | 148,538 | 181,638 | ||||||
Total liabilities and stockholders' equity | $ | 156,875 | $ | 199,645 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
120
Compass Therapeutics, Inc. and Subsidiaries
Consolidated Statements of Changes in Convertible Preferred StockOperations and Stockholders’ Equity (Deficit)Comprehensive Loss
(In thousands)thousands, except per share data)
|
| Convertible Preferred Stock |
|
| Common Stock |
|
| Additional Paid-in |
|
| Accumulated |
|
| Total Stockholders' |
| |||||||||||||
|
| Shares |
|
| Amount |
|
| Shares |
|
| Amount |
|
| Capital |
|
| Deficit |
|
| Equity (Deficit) |
| |||||||
Balance at January 1, 2019 |
|
| 207,164 |
|
| $ | 129,870 |
|
|
| 6,355 |
|
| $ | 1 |
|
| $ | 2,389 |
|
| $ | (87,164 | ) |
| $ | (84,774 | ) |
Vesting of share-based awards |
|
| — |
|
|
| — |
|
|
| 679 |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
Share-based compensation expense |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| 915 |
|
|
| — |
|
|
| 915 |
|
Net loss |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| (34,744 | ) |
|
| (34,744 | ) |
Balance at December 31, 2019 |
|
| 207,164 |
|
| $ | 129,870 |
|
|
| 7,034 |
|
| $ | 1 |
|
| $ | 3,304 |
|
| $ | (121,908 | ) |
| $ | (118,603 | ) |
|
| Convertible Preferred Stock |
|
| Common Stock |
|
| Additional Paid-in |
|
| Accumulated |
|
| Total Stockholders' |
| |||||||||||||
|
| Shares |
|
| Amount |
|
| Shares |
|
| Amount |
|
| Capital |
|
| Deficit |
|
| Equity (Deficit) |
| |||||||
Balance at January 1, 2020 |
|
| 207,164 |
|
| $ | 129,870 |
|
|
| 7,034 |
|
| $ | 1 |
|
| $ | 3,304 |
|
| $ | (121,908 | ) |
| $ | (118,603 | ) |
Common shares issued to former shareholders of Olivia Ventures Inc. |
|
| — |
|
|
| — |
|
|
| 1,000 |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
Conversion of Compass Therapeutics LLC preferred shares into common shares upon consummation of the reverse merger |
|
| (207,164 | ) |
|
| (129,870 | ) |
|
| 30,630 |
|
|
| 3 |
|
|
| 129,867 |
|
|
| — |
|
|
| 129,870 |
|
Common shares issued in private placement, net of issuance costs of $6.3 million |
|
| — |
|
|
| — |
|
|
| 12,096 |
|
|
| 1 |
|
|
| 54,230 |
|
|
| — |
|
|
| 54,231 |
|
Payment to non-participating Compass Therapeutics LLC members upon consummation of Merger |
|
| — |
|
|
| — |
|
|
| (13 | ) |
|
| — |
|
|
| (69 | ) |
|
| — |
|
|
| (69 | ) |
Vesting of share-based awards |
|
| — |
|
|
| — |
|
|
| 474 |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
Stock-based compensation |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| 4,016 |
|
|
| — |
|
|
| 4,016 |
|
Net loss |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| (29,500 | ) |
|
| (29,500 | ) |
Balance at December 31, 2020 |
|
| — |
|
| $ | — |
|
|
| 51,221 |
|
| $ | 5 |
|
| $ | 191,348 |
|
| $ | (151,408 | ) |
| $ | 39,945 |
|
Year Ended December 31, | ||||||||
2023 | 2022 | |||||||
Operating expenses: | ||||||||
Research and development | $ | 38,120 | $ | 29,997 | ||||
General and administrative | 12,243 | 11,658 | ||||||
Total operating expenses | 50,363 | 41,655 | ||||||
Loss from operations | (50,363 | ) | (41,655 | ) | ||||
Other income | 7,869 | 2,430 | ||||||
Loss before income tax expense | (42,494 | ) | (39,225 | ) | ||||
Income tax expense | — | — | ||||||
Net loss | $ | (42,494 | ) | $ | (39,225 | ) | ||
Net loss per share - basic and diluted | $ | (0.33 | ) | $ | (0.37 | ) | ||
Basic and diluted weighted average shares outstanding | 127,027 | 105,186 | ||||||
Other comprehensive loss: | ||||||||
Net loss | $ | (42,494 | ) | $ | (39,225 | ) | ||
Unrealized gain (loss) on marketable securities | 339 | (302 | ) | |||||
Comprehensive loss | $ | (42,155 | ) | $ | (39,527 | ) | ||
The accompanying notes are an integral part of these consolidated financial statements.
121
Compass Therapeutics, Inc. and Subsidiaries
Consolidated Statements of Cash FlowsChanges in Stockholders’ Equity
(In thousands)
|
| Year Ended December 31, |
| |||||
|
| 2020 |
|
| 2019 |
| ||
Cash flows from operating activities: |
|
|
|
|
|
|
|
|
Net loss |
| $ | (29,500 | ) |
| $ | (34,744 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
Depreciation and amortization |
|
| 2,404 |
|
|
| 2,120 |
|
Loss on disposal of equipment |
|
| 281 |
|
|
| — |
|
Noncash interest expense |
|
| 90 |
|
|
| 116 |
|
Share-based compensation |
|
| 4,016 |
|
|
| 915 |
|
Change in fair value of derivative liability |
|
| 556 |
|
|
| 104 |
|
Changes in operating assets and liabilities: |
|
|
|
|
|
|
|
|
Prepaid expenses and other current assets |
|
| (2,191 | ) |
|
| 435 |
|
Other long-term assets |
|
| (290 | ) |
|
| (56 | ) |
Accounts payable |
|
| 432 |
|
|
| (1,101 | ) |
Accrued expenses |
|
| (1,551 | ) |
|
| 509 |
|
Deferred rent |
|
| — |
|
|
| (39 | ) |
Settlement of derivative liability |
|
| (1,050 | ) |
|
| — |
|
Net cash used in operating activities |
|
| (26,803 | ) |
|
| (31,741 | ) |
Cash flows from investing activities: |
|
|
|
|
|
|
|
|
Purchases of property and equipment |
|
| (106 | ) |
|
| (466 | ) |
Proceeds from sale of equipment |
|
| 144 |
|
|
| — |
|
Net cash provided by (used in) investing activities |
|
| 38 |
|
|
| (466 | ) |
Cash flows from financing activities: |
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock |
|
| 60,482 |
|
|
| — |
|
Issuance costs from issuance of common stock |
|
| (6,319 | ) |
|
| — |
|
Repayment of borrowings under loan |
|
| (5,625 | ) |
|
| — |
|
Net cash provided by financing activities |
|
| 48,538 |
|
|
| — |
|
Net change in cash, cash equivalents and restricted cash |
|
| 21,773 |
|
|
| (32,207 | ) |
Cash, cash equivalents and restricted cash at beginning of year |
|
| 25,566 |
|
|
| 57,773 |
|
Cash, cash equivalents and restricted cash at end of year |
| $ | 47,339 |
|
| $ | 25,566 |
|
Supplemental disclosure of cash flow information |
|
|
|
|
|
|
|
|
Cash paid for interest |
| $ | 854 |
|
| $ | 1,115 |
|
Supplemental disclosure of financing activities |
|
|
|
|
|
|
|
|
Conversion of preferred units |
| $ | 129,870 |
|
| $ | — |
|
Common Stock | Additional | Accumulated Other | Accumulated | Total | ||||||||||||||||||||
Shares | Amount | Capital | Income (Loss) | Deficit | Equity | |||||||||||||||||||
Balance at December 31, 2021 | 100,832 | $ | 10 | $ | 373,657 | $ | — | $ | (233,589 | ) | $ | 140,078 | ||||||||||||
Common shares issued in PIPE Offering, net | 25,000 | 3 | 75,739 | — | — | 75,742 | ||||||||||||||||||
Common stock issued upon exercise of options | 6 | 0 | 15 | — | — | 15 | ||||||||||||||||||
Share-based awards | 559 | — | — | — | — | — | ||||||||||||||||||
Stock-based compensation | — | 0 | 5,330 | — | — | 5,330 | ||||||||||||||||||
Cancelled shares | (95 | ) | — | — | — | — | — | |||||||||||||||||
Unrealized loss on marketable securities | — | — | — | (302 | ) | — | (302 | ) | ||||||||||||||||
Net loss | — | — | — | — | (39,225 | ) | (39,225 | ) | ||||||||||||||||
Balance at December 31, 2022 | 126,302 | $ | 13 | $ | 454,741 | $ | (302 | ) | $ | (272,814 | ) | $ | 181,638 | |||||||||||
Common shares issued in ATM Offering, net | 952 | $ | — | $ | 3,032 | $ | — | $ | — | $ | 3,032 | |||||||||||||
Common stock issued upon exercise of options | 29 | — | 62 | — | — | 62 | ||||||||||||||||||
Share-based awards, net of tax remittance | 385 | — | (159 | ) | — | — | (159 | ) | ||||||||||||||||
Stock-based compensation | — | — | 6,120 | — | — | 6,120 | ||||||||||||||||||
Unrealized gain on marketable securities | — | — | — | 339 | — | 339 | ||||||||||||||||||
Net loss | — | — | — | — | (42,494 | ) | (42,494 | ) | ||||||||||||||||
Balance at December 31, 2023 | 127,668 | $ | 13 | $ | 463,796 | $ | 37 | $ | (315,308 | ) | $ | 148,538 | ||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
122
Compass Therapeutics, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
(In thousands)
Year Ended December 31, | ||||||||
2023 | 2022 | |||||||
Cash flows from operating activities: | ||||||||
Net loss | $ | (42,494 | ) | $ | (39,225 | ) | ||
Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
Depreciation and amortization | 699 | 782 | ||||||
Gain on disposal of equipment | — | (70 | ) | |||||
Share-based compensation | 6,120 | 5,330 | ||||||
Amortization of premium and discount on marketable securities | (3,226 | ) | (801 | ) | ||||
ROU asset amortization | 1,191 | 1,122 | ||||||
Changes in operating assets and liabilities: | ||||||||
Prepaid expenses and other current assets | 6,762 | (5,591 | ) | |||||
Accounts payable | 708 | 2,515 | ||||||
Accrued expenses | (9,176 | ) | 2,915 | |||||
Operating lease liability | (1,202 | ) | (1,102 | ) | ||||
Net cash used in operating activities | (40,618 | ) | (34,125 | ) | ||||
Cash flows from investing activities: | ||||||||
Purchases of marketable securities | (155,418 | ) | (200,119 | ) | ||||
Proceeds from sale or maturities of marketable securities | 182,413 | 48,955 | ||||||
Purchases of property and equipment | (30 | ) | (212 | ) | ||||
Proceeds from sale of equipment | — | 176 | ||||||
Net cash provided by (used) in investing activities | 26,965 | (151,200 | ) | |||||
Cash flows from financing activities: | ||||||||
Proceeds from issuance of common stock | 3,126 | 76,237 | ||||||
Issuance costs from issuance of common stock | (94 | ) | (495 | ) | ||||
Proceeds from exercise of stock options | 62 | 15 | ||||||
Taxes related to the vesting of RSUs | (159 | ) | — | |||||
Net cash provided by financing activities | 2,935 | 75,757 | ||||||
Net change in cash and cash equivalents | (10,718 | ) | (109,568 | ) | ||||
Cash and cash equivalents at beginning of year | 34,946 | 144,514 | ||||||
Cash and cash equivalents at end of year | $ | 24,228 | $ | 34,946 | ||||
Supplemental disclosure of cash flow information | ||||||||
Unrealized (gain) loss on marketable securities | $ | (339 | ) | $ | 302 | |||
The accompanying notes are an integral part of these consolidated financial statements.
Compass Therapeutics, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
1. |
|
|
Compass Therapeutics, Inc. (“Compass” or the “Company”) is a clinical-stage, oncology-focused biopharmaceutical company developing proprietary antibodyantibody-based therapeutics intended to engagetreat multiple human diseases. Our scientific focus is on the relationship between angiogenesis and the immune system to treat both solid tumors and hematological malignancies. The Company’s immuno-oncologysystem. Our pipeline includes novel product candidates that leverage our understanding of the tumor microenvironment, including both angiogenesis-targeted agents and immune-oncology focused agents. These product candidates are designed to optimize critical components required for an effective anti-tumor response to cancer. These include modulation of the microvasculature via angiogenesis-targeted agents; induction of a clinical-stage monoclonal antibody and a portfolio of bispecific antibodies. The Company was incorporated as Olivia Ventures, Inc. (“Olivia”)potent immune response via activators on effector cells in the Statetumor microenvironment; and alleviation of Delaware on March 20, 2018. Priorimmunosuppressive mechanisms used by tumors to the Merger (as defined below), Olivia was a “shell company” (as definedevade immune surveillance. We plan to advance our product candidates through clinical development as both standalone therapies and in Rule 12b-2 of the Securities Exchange Act of 1934,combination with our proprietary drug candidates as amended).long as their continued development is supported by clinical and nonclinical data. References to Compass or the Company herein include Compass Therapeutics, Inc. and its wholly-owned subsidiaries.
On June 17, 2020, the Company’s Board of Directors and the Company’s pre-Merger (defined below) stockholders approved an amended and restated certificate of incorporation, which, among other things, increased authorized capital stock from 50,000,000 shares of common stock par value $0.0001 and 5,000,000 shares of preferred stock, par value $0.0001 per share, to 300,000,000 shares of common stock, par value $0.0001 per share, and 10,000,000 shares of preferred stock, par value $0.0001 per share.
On June 17, 2020, the Company completed a merger (the “Merger”) of its wholly-owned subsidiary, Compass Therapeutics LLC, pursuant to an Agreement and Plan of Merger and Reorganization (the “Merger Agreement”), by and among Olivia Ventures, Inc., Compass Acquisition LLC, Compass Therapeutics LLC, and as a result, Compass Therapeutics LLC became a wholly-owned subsidiary of the Company. Additionally, certain of the Company’s wholly-owned subsidiaries (each, a “Blocker Merger Sub”) merged with and into the applicable blocker entity (“Blockers”) in transactions that are referred to as “Blocker Mergers.”
At the effective time of the Merger and the applicable effective time of each Blocker Merger, (collectively, the “Effective Time”), an aggregate of 31,627,139 shares of the Company’s common stock were issued to holders of common membership interests of Compass Therapeutics LLC (including common membership interests issued upon the conversion of preferred membership interests) and 7,428,217 shares of its common stock were issued to the holders of equity interests of the Blockers. The issuances of shares of the Company’s common stock to the security holders of Compass Therapeutics LLC and the Blockers are collectively referred to as the Share Conversion.
In addition, 2,930,836 shares of the Company’s common stock were reserved for issuance under the Company’s 2020 Stock Option and Incentive Plan. Immediately prior to the Effective Time, an aggregate of 4,000,000 of the 5,000,000 shares of the Company’s common stock held by pre-Merger stockholders of Olivia Ventures, Inc. were forfeited and surrendered for cancellation. No fractional shares of the Company’s common stock were issued in connection with the Merger, and holders of membership interests of Compass Therapeutics LLC received cash in lieu thereof.
The Merger and the Blocker Mergers were treated as a recapitalization and reverse acquisition for financial reporting purposes. Compass Therapeutics LLC is considered the acquirer for accounting purposes, and the Company’s historical financial statements before the Merger have been replaced with the historical financial statements of Compass Therapeutics LLC in filings with the SEC subsequent to the Merger. As a result, the vested and outstanding common membership interests of Compass Therapeutics LLC have been presented as outstanding shares of the Company’s common stock for all periods presented. All outstanding preferred membership interests of Compass Therapeutics LLC are presented as convertible preferred stock for all periods and until such interests were converted into shares of the Company’s common stock at the time of the Merger.
123
On June 19, 2020, the Company completed a private placement (“Private Placement”) and sold 12,096,442 shares of its common stockat a purchase price of $5.00 per share and received net proceeds of $54.2 million, after associated offering costs.
The Company is subject to risks and uncertainties common to companies in the biotechnology and pharmaceutical industries. There can be no assurance that the Company’s research and development will be successfully completed, that adequate protection for the Company’s technology will be obtained, that any products developed will obtain necessary government regulatory approval or that any approved products will be commercially viable. The Company operates in an environment of rapid change in technology and substantial competition from pharmaceutical and biotechnology companies. In addition, the Company is dependent upon the services of its employees and consultants.
2. | Liquidity, Uncertainties and Going Concern |
|
The Company has evaluated whether there are conditions and events, considered in the aggregate, that raise substantial doubt about its ability to continue as a going concern within one year after the date that the consolidated financial statements are issued.
Since its inception, the Company has funded its operations primarily with proceeds from the sale of its equity securities and borrowings under the 2018 Credit Facility.securities. The Company has incurred recurring losses since its inception and had an accumulated deficit of $151.4$315.3 million on December 31, 2020.2023. The Company expects to continue too generate operating losses for the foreseeable future. The Company expects that its cash, and cash equivalents and marketable securities will be sufficient to fund its operating expenses and capital expenditure requirements into the second quarter of 2022.mid-2026. The future viability of the Company beyond that point is dependent on its ability to raise additional capital to finance its operations.
The Company is subject to risks common to early stage companies in the biotechnology industry including, but not limited to: having a limited operating history and no products approved for commercial sale; having a history of significant losses; its need to obtain additional financing; dependence on its ability to advance its current and future product candidates through clinical trials, marketing approval and commercialization; the lengthy and expensive nature and uncertain outcomes of the clinical development process; the lengthy, time consuming and unpredictable nature of the regulatory approval process; the results of preclinical studies and early stage clinical trials that may not be predictive of future results; dependence on its key personnel; risks related to patent protection and the Company’s pending patent applications; dependence on third party collaborators for the discovery, development and commercialization of current and future product candidates; and significant competition from other biotechnology and pharmaceutical companies. Even if the Company’s development efforts are successful, it is uncertain when, if ever, the Company will realize significant revenue from product sales.
Due to the evolving and uncertain global impacts of the COVID-19 pandemic, we cannot precisely determine or quantify the impact this pandemic will have on our business operations in the future. We have been carefully monitoring the COVID-19 pandemic and its potential impact on our business and have taken important steps to help ensure the safety of our employees and their families and to reduce the spread of COVID-19 community-wide. We are continuing to assess the potential impact of the COVID-19 pandemic on our current and future business and operations, including our expenses and clinical trials, as well as on our industry and the healthcare system. To date, we have been able to continue to pursue our Phase 1 clinical trial without significant delays or major difficulties despite the COVID-19 pandemic.
124
3. | Summary of Significant Accounting Policies |
Basis of Presentation
The accompanying consolidated financial statements are presented in U.S. dollars and have been prepared in conformity with accounting principles generally accepted in the United States of America (“GAAP”). Any reference in these notes to applicable guidance is meant to refer to the authoritative GAAP as found in the Accounting Standards Codification (“ASC”) and as amended by Accounting Standards Update (“ASU”) of the Financial Accounting Standards Board (“FASB”).
In June 2020, the Company completed the Merger, as discussed in Note 1. Upon the closing of the Merger, the common and convertible preferred units of Compass Therapeutics LLC were converted into the Company’s common stock. The Company effected a 0.15-for-one stock conversion ratio for its issued and outstanding convertible preferred units. Common units were converted using the same ratio after factoring in the relevant strike price of each grant. Subsequent to the Merger, there were no common or convertible preferred units outstanding. All of the share and per share information presented in the accompanying financial statements has been adjusted to reflect the stock split on a retroactive basis for all periods and as of all dates presented.
Upon the closing of the Merger, the Company’s certificate of incorporation was amended and restated to provide for 10 million authorized shares of preferred stock with a par value of $0.0001 per share and 300 million authorized shares of common stock with a par value of $0.0001 per share.
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of Compass Therapeutics, Inc., and its wholly-owned subsidiaries, including Compass Therapeutics LLC, and Compass Therapeutics Advisors Inc., Trigr Therapeutics, Inc. and Compass Therapeutics Securities Corporation. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period. Significant estimates and assumptions reflected in these financial statements include, but are not limited to, the accrual of research and development expenses, useful lives of equipment, interest rate and term operating lease ROU and liability, the valuationpercentage of the embedded derivative,completion of contractual arrangements, future cash expenditures for liquidity estimates, the valuation of common stock and estimates associated with stock-based awards. Estimates are periodically reviewed in light of changes in circumstances, facts and experience. Actual results could differ from those estimates. Changes in estimates are recorded prospectively in the period that they become known.
Segment Information
Operating segments are defined as components of an enterprise for which separate and discrete information is available for evaluation by the chief operating decision-maker in deciding how to allocate resources and assess performance. The Company has one operating segment. The Company’s chief operating decision-maker, its chief executive officer, manages the Company’s operations on a consolidated basis for the purpose of allocating resources. All of the Company’s long-lived assets are held in the United States.
125
Cash and Cash Equivalents
The Company considers all highly liquid investments that are readily convertible into cash with original maturities of three months or less from the date of purchase to be cash equivalents. Cash and cash equivalents include cash held in banks and amounts held in money market funds.funds or commercial paper. Cash equivalents are stated at cost, which approximates market value. CashCash and Cash equivalents consisted of money market funds of $43.6were $24.2 million and $22.8$34.9 million on December 31, 20202023 and 2019,2022, respectively.
Restricted Cash
AsMarketable Securities
All of December 31, 2020the Company’s investment securities are debt securities and 2019, thebank instruments. The Company was required to maintaincarries these investments at fair value. Unrealized gains and losses, if any, are reported as a separate cash balancecomponent of $0.2 millionstockholders’ equity. The cost of investment securities is adjusted for amortization of premiums and accretion of discounts to collateralize corporate credit cards with a bank, which was classified as restricted cashmaturity. Such amortization and accretion are included in interest income. Realized gains and losses, if any, are also included in interest income. The cost of securities sold is based on the consolidated balance sheets as a non-current asset.specific identification method.
In connection with the Company’s lease agreement entered into July 2016 (see Note 11), the Company is required to maintain a letter of credit of $0.1 million for the benefit of the landlord. As of December 31, 2020 and 2019, the underlying cash balance securing this letter of credit was classified as restricted cash on the consolidated balance sheets as a non-current asset.
Concentrations of Credit Risk
Financial instruments, thatwhich potentially exposesubject the Company to concentrations of credit risk, principally consist primarily of cash, cash equivalents and restricted cash.marketable securities. The Company maintainsinvests its excess cash cash equivalentsprimarily in money market funds, U.S. treasury notes, and restricted cashhigh quality, marketable debt instruments of corporations in accordance with financial institutions that management believesthe Company’s investment policy. The Company’s investment policy defines allowable investments and establishes guidelines relating to becredit quality, diversification, and maturities of high-credit quality. its investments to preserve principal and maintain liquidity. The Company has not experienced any realized losses related to its cash cash equivalents and restricted cash.marketable securities.
Property and Equipment
Property and equipment are stated at cost, net of accumulated depreciation and amortization. Depreciation and amortization are recorded using the straight-line method over the estimated useful lives of the related assets as follows:
Asset Classification | Estimated Useful Life | ||
Equipment | 5 years | ||
Furniture and fixtures | 7 years | ||
Software | 5 years | ||
Leasehold improvements | Lesser of estimated useful life or lease term |
| |
Estimated useful lives are periodically assessed to determine if changes are appropriate. Maintenance and repairs are charged to expense as incurred. When assets are retired or otherwise disposed of, the cost of these assets and related accumulated depreciation or amortization are eliminated from the consolidated balance sheet and any resulting gains or losses are included in the consolidated statement of operations and comprehensive loss in the period of disposal. Costs for capital assets not yet placed into service are capitalized as construction-in-progress and depreciated once placed into service. Assets held under capital leases are stated at the lesser of the present value of future minimum lease payments or the fair value of the leased asset at the inception of the lease. Amortization of assets held under capital leases is computed using the straight-line method over the shorter of the estimated useful life of the asset or the period of the related lease.
Impairment of Long-Lived Assets
Long-lived assets consist of property, equipment and equipment.right-of-use (“ROU”) assets. Long-lived assets to be held and used are tested for recoverability whenever events or changes in business circumstances indicate that the
126
carrying amount of the assets may not be fully recoverable. Factors that the Company considers in deciding when to perform an impairment review include significant underperformance of the business in relation to expectations, significant negative industry or economic trends and significant changes or planned changes in the use of the assets. If an impairment review is performed to evaluate a long-lived asset group for recoverability, the Company compares forecasts of undiscounted cash flows expected to result from the use and eventual disposition of the long-lived asset group to its carrying value. An impairment loss would be recognized in the consolidated statements of operations when estimated undiscounted future cash flows expected to result from the use of an asset group are less than its carrying amount. The impairment loss would be based on the excess of the carrying value of the impaired asset group over its fair value, determined based on discounted cash flows. The Company did not record any impairment losses on long-lived assets during the yearyears ended December 31, 20202023 and 2019.2022.
Fair Value Measurements
Certain assets and liabilities of the Company are carried at fair value under GAAP. Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value must maximize the use of observable inputs and minimize the use of unobservable inputs. Financial assets and liabilities carried at fair value are to be classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable:
Level 1 – Quoted prices in active markets for identical assets or liabilities.
● | Level 1 – Quoted prices in active markets for identical assets or liabilities. |
Level 2 – Observable inputs (other than Level 1 quoted prices), such as quoted prices in active markets for similar assets or liabilities, quoted prices in markets that are not active for identical or similar assets and liabilities, or other inputs that are observable or can be corroborated by observable market data.
● | Level 2 – Observable inputs (other than Level 1 quoted prices), such as quoted prices in active markets for similar assets or liabilities, quoted prices in markets that are not active for identical or similar assets and liabilities, or other inputs that are observable or can be corroborated by observable market data. |
Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to determining the fair value of the assets or liabilities, including pricing models, discounted cash flow methodologies and similar techniques.
● | Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to determining the fair value of the assets or liabilities, including pricing models, discounted cash flow methodologies and similar techniques. |
To the extent that the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
An entity may choose to measure many financial instruments and certain other items at fair value at specified election dates. Subsequent unrealized gains and losses on items for which the fair value option has been elected are reported in earnings.
The Company’s cash equivalents are carried at fair value according to the fair value hierarchy described above and were determined based on Level 1 measurements (see Note 4). The Company’s restricted cash is carried at fair value according to the fair value hierarchy described above and were determined based on Level 2 measurements (see Note 4). The carrying values of other current assets and accounts payable approximate their fair value due to the short‑term nature of these assets and liabilities. The carrying values of the Company’s loan approximated its fair value as of December 31, 2020 and 2019 due to its variable interest rate. The fair value of the loan related embedded derivative (see Note 4) was determined based on Level 3 measurements.
127
Research and Development Costs
Costs associated with internal research and development and external research and development services, including drug development and preclinical studies, are expensed as incurred. Research and development expenses include costs for salaries, employee benefits, subcontractors, facility-related expenses, depreciation and amortization, stock-based compensation, third-party license fees, laboratory supplies, and external costs of outside vendors engaged to conduct discovery, preclinical and clinical development activities and clinical trials as well as to manufacture clinical trial materials and other costs. The Company recognizes external research and development costs based on an evaluation of the progress to completion of specific tasks using information provided to the Company by its service providers.
Nonrefundable advance payments for goods or services to be received in the future for use in research and development activities are recorded as prepaid expenses. Such prepaid expenses are recognized as an expense when the goods have been delivered or the related services have been performed, or when it is no longer expected that the goods will be delivered, or the services rendered.
Costs associated with licenses of technology acquired as part of collaborative arrangements are expensed as incurred and are generally included in research and development expense in the consolidated statements of operations if it is determined the license has no alternative future use.
Accrued Research and Development Expenses
The Company has entered into various research and development and other agreements with commercial firms, researchers, universities and others for provisions of goods and services. These agreements are generally cancelable, and the related costs are recorded as research and development expenses as incurred. The Company records accruals for estimated ongoing research and development costs. When evaluating the adequacy of the accrued liabilities, the Company analyzes progress of the studies or clinical trials, including the phase or completion of events, invoices received and contracted costs. Significant judgments and estimates are made in determining the accrued balances at the end of any reporting period. Actual results could differ materially from the Company’s estimates. The Company’s historical accrual estimates have not been materially different from the actual costs.
Debt Issuance Costs
Debt issuance costs consist of payments made to secure commitments under certain debt financing arrangements. These amounts are recognized as interest expense over the period of the financing arrangement using the effective interest method. If the financing arrangement is canceled or forfeited, or if the utility of the arrangement to the Company is otherwise compromised, these costs are recognized as interest expense immediately.
The Company’s consolidated financial statements present debt issuance costs related to a recognized debt liability as a direct reduction from the carrying amount of that debt liability.
Patent Costs
All patent-related costs incurred in connection with filing and prosecuting patent applications are expensed as incurred due to the uncertainty about the recovery of the expenditure. Amounts incurred are classified as general and administrative expense in the consolidated statements of operations.
128
Stock-Based Compensation
The Company recognizes the grant‑date fair value of stock‑based awards issued to employees and nonemployee board members as compensation expense on a straight‑line basis over the service period of the award. The Company uses the Black‑Scholes option pricing model to determine the grant‑date fair value of stock options and adjusts expense for forfeitures in the periods they occur.
The fair value of each equity award was determined by the Company on the date of grant and by using the methods and assumptions discussed below. Certain of these inputs are subjective and generally require judgment to determine.
Stock price: See below.The stock price used to value equity awards is based on the closing price of the Company’s common stock as reported on the date of the grant. For equity awards issued after June 2020 until the Company started trading on a public market in the second quarter of 2021, the valuation of the Company’s common stock was $5.00 per share, which was the share price paid by outside investors in the Company’s Private Placement in June 2020.
Expected term: The expected term of the equity award represents the weighted average period the award is expected to be outstanding. The Company uses the simplified method for estimating the expected term as provided by the Securities and Exchange Commission. The simplified method calculates the expected term as the average time to vesting and the contractual life of the award.
Expected volatility – Due toThe expected volatility is calculated based on the Company’s limited operating history and lackhistorical volatility of Company-specific historical or implied volatility,our common stock over the expected volatility assumption was determined by examiningterm of the historical volatilities of a group of industry peers whose share prices are publicly available.option.
Risk-free interest rate – The risk-free rate assumption is based on U.S. Treasury instruments, the terms of which were consistent with the expected term of the Company’s equity award.
Expected dividend – The Company has not paid and does not intend to pay dividends.
Stock Price - Prior to the Merger
The Company issued Class A and Class C common units to various employees, directors and consultants. The units constituted “profits interests” for tax purposes and were accounted for as share-based payment arrangements. Upon consummation of the Merger, all outstanding vested units were converted into shares of common stock and all outstanding unvested units were converted into shares of restricted stock that continue to vest over the remaining term of the original award.
The estimated fair value was determined by our board of directors as of the date of each stock award, with input from management, considering our most recently available third-party valuation, and our board of directors’ assessment of additional objective and subjective factors that it believed were relevant and which may have changed from the date of the most recent valuation through the date of the grant. These third-party valuations were performed in accordance with the guidance outlined in the American Institute of Certified Public Accountants’ Accounting and Valuation Guide, Valuation of Privately-Held-Company Equity Securities Issued as Compensation. In addition to considering the results of these third-party valuations, our board of directors considered various objective and subjective factors to determine the fair value of our stock awards as of each grant date, including:
the prices at which we sold preferred membership interests and the superior rights and preferences of the preferred membership interests relative to our membership interests at the time of each grant;
the progress of our commercialization efforts;
the progress of our research and development programs, including the status and results of preclinical studies for our product candidates;
129
our stage of development and our business strategy;
external market conditions affecting the medical device industry and trends within the medical device industry;
our financial position, including cash on hand, and our historical and forecasted performance and operating results;
the lack of an active public market for our common and preferred membership interests;
the likelihood of achieving a liquidity event, such as an initial public offering, or IPO, reverse merger, or sale of our Company in light of prevailing market conditions; and
the analysis of IPOs and the market performance of similar companies in the biopharmaceutical industry.
In determining the estimated fair value, our board of directors considered the subjective factors discussed above in conjunction with the most recent valuations of our membership interests that were prepared by an independent third party.
Stock Price - Subsequent to the Merger
A public trading market for the Company’s common stock was not established between the closing of the Merger and December 31, 2020. For the valuation of the Company’s common stock at September 30, 2020 and December 31, 2020, the Company used $5.00 per share, which is the share price paid by outside investors in the Company’s Private Placement that closed in June 2020.
Upon establishing a public market for the Company’s securities, the stock price of the Company’s common stock used to value equity awards will be based on the closing price of the Company’s common stock as reported on the date of the grant.
Net Loss per Share
Basic loss per share is computed by dividing net loss by the weighted average number of shares of common stock outstanding during each period. Diluted loss per share includes the effect, if any, from the potential exercise or conversion of securities, such as convertible preferred stock, stock options, restricted stock units, unvested restricted stock awards and common stock warrants that would result in the issuance of incremental shares of common stock. In computing the basic and diluted net loss per share, the weighted average number of shares remains the same for both calculations due to the fact that when a net loss exists, dilutive shares are not included in the calculation as the impact is anti-dilutive.
The following potentially dilutive securities outstanding as of December 31, 20202023 and 20192022 have been excluded from the computation of diluted weighted average shares outstanding, as they would be anti-dilutive:
|
| December 31, |
| |||||
|
| 2020 |
|
| 2019 |
| ||
|
| (000's) |
| |||||
Convertible preferred stock |
|
| — |
|
|
| 30,630 |
|
Stock options |
|
| 2,159 |
|
|
| — |
|
Nonvested restricted stock |
|
| 896 |
|
|
| 1,917 |
|
Common unit warrants |
|
| — |
|
|
| 3,114 |
|
Total |
|
| 3,055 |
|
|
| 35,661 |
|
130
December 31, | ||||||||
2023 | 2022 | |||||||
(000's) | ||||||||
Restricted stock units | 1,500 | 900 | ||||||
Stock options | 7,876 | 5,378 | ||||||
Nonvested restricted stock | — | 193 | ||||||
Total | 9,376 | 6,471 | ||||||
Income Taxes
Income taxes are accounted for under the asset and liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to temporary differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, operating losses and tax credit carryforwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in operations in the period that includes the enactment date. Deferred income tax assets are reduced, as necessary, by a valuation allowance when management determines it is more likely than not that some or all of the deferred tax benefits will not be realized.
The Company files income tax returns in the U.S. Federal jurisdiction and in various states. The Company has tax net operating loss carryforwards that are subject to examination for a number of years beyond the year in which they were generated for tax purposes. Since a portion of these net operating loss carryforwards may be utilized in the future, many of these net operating loss carryforwards will remain subject to examination.
Prior
New Accounting Pronouncements
In December 2023, the FASB issued ASU No. 2023-09, Income Taxes (Topic 740): Improvements to tax disclosures. The purpose of ASU 2023-09 is to enhance the Merger, Compass Therapeutics LLC elected to be treated as a partnership fortransparency and decision usefulness of income tax reporting purposes and therefore, federal and Massachusetts and any other state income taxes are the responsibilitydisclosures. The ASU requires a significant expansion of the individual members. As such, no federal or stategranularity of the income taxes relatedtax rate reconciliation as well as an expansion of other income tax disclosures. The ASU is effective on a prospective basis for annual periods beginning after December 15, 2024. This will result in additional disclosures being included in our consolidated financial statements, once adopted. The Company is evaluating the impact of ASU 2023-09 and does not expect ASU-2023-09 to the LLC are recorded inhave a significant impact on the consolidated financial statements. The Company’s wholly-owned subsidiary, Compass Therapeutics Advisors Inc., is organized as a C-corporation and is subject to federal and state income taxes. All such taxes have been recorded in the consolidated financial statements.
Recently Adopted Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016 02, Leases, which requires a lessee to record a right of use asset and a corresponding lease liability on the balance sheet for all leases with terms longer than 12 months. The standard continues to have two types of leases for income statement recognition purposes: operating leases and finance leases. Operating leases result in the recognition of a single lease expense on a straight-line basis over the lease term, similar to the treatment for operating leases under the old standard. Finance leases result in an accelerated expense similar to the accounting for capital leases under the old standard. The new standard also contains amended guidance regarding the identification of embedded leases in service contracts and the identification of lease and non-lease components of an arrangement. The Company adopted the new standard on January 1, 2021, using a modified retrospective approach and as a result did not adjust prior periods. Adoption of the standard resulted in the recording of $5.1 million of operating lease ROU assets and operating lease liabilities, but did not have a material impact on the Company’s net income or cash flows.
Recently Issued Accounting Pronouncements
In August 2018, the FASB issued ASU No. 2018‑13, Disclosure Framework—Changes to the Disclosure Requirements for Fair Value Measurements, which changes the fair value measurement disclosure requirements of ASC Topic 820. The goal of the ASU is to improve the effectiveness of ASC Topic 820’s disclosure requirements. The Company adopted this guidance on January 1, 2020 and was not material to its consolidated financial statements and related disclosures.
131
In December 2019, the FASB issued ASU No. 2019-12, Income Taxes(Topic 740): Simplifying the Accounting for Income Taxes ("ASU 2019-12"), which is intended to simplify the accounting for income taxes. ASU 2019-12 removes certain exceptions to the general principles in Topic 740 and also clarifies and amends existing guidance to improve consistent application. The new standard will bewas effective beginning January 1, 2022. The Company does not expect the adoption of ASU 2019-12 todid not have a material impact on itsthe Company’s financial position and results of operations upon adoption.
The Company adopted Accounting Standards Update ASU No. 2016-13, Financial Instruments — Credit Losses (Topic 326):Measurement of Credit Losses of Financial Instruments on January 1, 2023. Marketable debt securities for which the estimated fair value is below amortized cost are evaluated for credit impairment. Credit impairment is recorded through the statements of operations via an allowance for credit losses and any remaining unrealized gains and losses are reported as a component of other comprehensive income (loss) within the statements of operations and comprehensive loss and as a separate component of stockholders’ equity. For all marketable securities which the estimated fair value was below amortized cost as of December 31, 2023 and 2022, the decline in fair value was not driven by credit impairment.
There are no other pending accounting pronouncements that are expected to have a material impact on the Company’s consolidated financial statements.
4. |
|
|
The following tables present information about the Company’s financial assets and liabilities that are measured at fair value on a recurring basis and indicate the level of the fair value hierarchy utilized to determine such fair values:
Fair Value Measurements as of December 31, 2023 (000's): | ||||||||||||||||
Quoted Prices | Significant | |||||||||||||||
in Active | Other | Significant | ||||||||||||||
Markets for | Observable | Unobservable | ||||||||||||||
Identical Assets | Inputs | Inputs | ||||||||||||||
(Level 1) | (Level 2) | (Level 3) | Fair Value | |||||||||||||
Assets | ||||||||||||||||
Corporate bonds | $ | — | $ | 54,281 | $ | — | $ | 54,281 | ||||||||
Commercial paper | 28,534 | — | — | 28,534 | ||||||||||||
Certificates of deposit | — | 18,866 | — | 18,866 | ||||||||||||
U.S. government treasuries | 16,080 | — | — | 16,080 | ||||||||||||
Asset-backed securities | — | 10,472 | — | 10,472 | ||||||||||||
Money market funds (cash equivalents) | 575 | — | — | 575 | ||||||||||||
Total assets | $ | 45,189 | $ | 83,619 | $ | — | $ | 128,808 | ||||||||
Fair Value Measurements as of December 31, 2022 (000's): | ||||||||||||||||
Quoted Prices | Significant | |||||||||||||||
in Active | Other | Significant | ||||||||||||||
Markets for | Observable | Unobservable | ||||||||||||||
Identical Assets | Inputs | Inputs | ||||||||||||||
(Level 1) | (Level 2) | (Level 3) | Fair Value | |||||||||||||
Assets | ||||||||||||||||
Corporate bonds | $ | — | $ | 87,760 | $ | — | $ | 87,760 | ||||||||
Commercial paper | 37,682 | — | — | 37,682 | ||||||||||||
Certificates of deposit | — | 19,667 | — | 19,667 | ||||||||||||
Asset-backed securities | — | 6,554 | — | 6,554 | ||||||||||||
Money market funds (cash equivalents) | 9,438 | — | — | 9,438 | ||||||||||||
Total assets | $ | 47,120 | $ | 113,981 | $ | — | $ | 161,101 | ||||||||
| Fair Value Measurements as of December 31, 2020 Using (000's): |
| ||||||||||
| Quoted |
|
|
|
|
|
|
|
|
|
| |
| Prices |
|
|
|
|
|
|
|
|
|
| |
| in Active |
| Significant |
|
|
|
|
|
|
| ||
| Markets for |
| Other |
| Significant |
|
|
|
| |||
| Identical |
| Observable |
| Unobservable |
|
|
|
| |||
| Assets |
| Inputs |
| Inputs |
|
|
|
| |||
| (Level 1) |
| (Level 2) |
| (Level 3) |
| Fair Value |
| ||||
Assets |
|
|
|
|
|
|
|
|
|
|
|
|
Cash equivalents - money market funds | $ | 43,631 |
| $ | — |
| $ | — |
| $ | 43,631 |
|
Total assets | $ | 43,631 |
| $ | — |
| $ | — |
| $ | 43,631 |
|
| Fair Value Measurements as of December 31, 2019 Using (000's): |
| ||||||||||
| Quoted |
|
|
|
|
|
|
|
|
|
| |
| Prices |
|
|
|
|
|
|
|
|
|
| |
| in Active |
| Significant |
|
|
|
|
|
|
| ||
| Markets for |
| Other |
| Significant |
|
|
|
| |||
| Identical |
| Observable |
| Unobservable |
|
|
|
| |||
| Assets |
| Inputs |
| Inputs |
|
|
|
| |||
| (Level 1) |
| (Level 2) |
| (Level 3) |
| Fair Value |
| ||||
Assets |
|
|
|
|
|
|
|
|
|
|
|
|
Cash equivalents - money market funds | $ | 22,784 |
| $ | — |
| $ | — |
| $ | 22,784 |
|
Total assets | $ | 22,784 |
| $ | — |
| $ | — |
| $ | 22,784 |
|
Liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
Derivative liability related to loan | $ | — |
| $ | — |
| $ | 494 |
| $ | 494 |
|
Total liabilities | $ | — |
| $ | — |
| $ | 494 |
| $ | 494 |
|
5. | Marketable Securities |
ValuationThe objectives of Derivative Liabilitythe Company’s investment policy are to ensure the safety and preservation of invested funds, as well as to maintain liquidity sufficient to meet cash flow requirements. The Company invests its excess cash in securities issued by financial institutions, commercial companies, and government agencies that management believes to be of high credit quality in order to limit the amount of its credit exposure. The Company has not realized any net losses from its investments.
Unrealized gains and losses on investments that are available for sale are recognized in accumulated comprehensive loss, unless an unrealized loss is considered to be other than temporary, in which case the unrealized loss is charged to operations. The Company periodically reviews its investments for other than temporary declines in fair value below cost basis and whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable.
The Company’s derivative liability was comprised ofCompany believes the contingentindividual unrealized losses represent temporary declines primarily resulting from interest rate reset featureschanges. Realized gains and a contingent feature to pay a success fee upon the occurrence of certain liquidity events, each of which met the definition of a derivative instrument, which termslosses are included in the loan and security agreement (see Note 7). The Company classified these instruments as a liability on the consolidated balance sheet because these features were not clearly and closely related to its host instrument and met the definition of a derivative. The derivative liability was initially recorded at fair value upon issuance of the loan and was being subsequently remeasured to fair value at each reporting date. Changes in the fair value of the derivative liability were recognized as a component of other income (expense), net in the consolidated statements of operations. The success fee was paid in full following the closing of the Merger in June 2020.
132
The fair value of the derivative liability recognized was determined based on significant inputs not observable in the market, which represents a Level 3 measurement within the fair value hierarchy. The fair value of the derivative liability wasoperations and comprehensive loss and are determined using the probability-weighted expected returnspecific identification method which consideredwith transactions recorded on a trade date basis. The Company classifies marketable securities that are available for use in current operations as inputscurrent assets on the type, timing and probability of occurrence of a change-of-control event, the future equity financing and cash settlement of the loans; the potential amount of the payment under each of these potential settlement scenarios; and the risk-adjusted discount rate reflecting the expected risk profile for each of the potential settlement scenarios.consolidated balance sheet.
The following table provides a roll forward of the aggregate fair values of the Company’s derivative liability:tables summarize marketable securities held at December 31, 2023 and 2022 (in thousands):
Fair Value Measurements as of December 31, 2023 Using: | ||||||||||||||||
Amortized Cost | Unrealized gains | Unrealized Losses | Fair Value | |||||||||||||
Assets | ||||||||||||||||
Corporate bonds | $ | 54,256 | $ | 74 | $ | (49 | ) | $ | 54,281 | |||||||
Commercial paper | 28,507 | 30 | (3 | ) | 28,534 | |||||||||||
Certificates of deposit | 18,850 | 17 | (1 | ) | 18,866 | |||||||||||
U.S. government treasuries | 16,127 | 3 | (50 | ) | 16,080 | |||||||||||
Asset-backed securities | 10,456 | 23 | (7 | ) | 10,472 | |||||||||||
Total assets | $ | 128,196 | $ | 147 | $ | (110 | ) | $ | 128,233 | |||||||
Fair Value Measurements as of December 31, 2022 Using: | ||||||||||||||||
Amortized Cost | Unrealized gains | Unrealized Losses | Fair Value | |||||||||||||
Assets | ||||||||||||||||
Corporate bonds | $ | 87,998 | $ | 12 | $ | (250 | ) | $ | 87,760 | |||||||
Commercial paper | 37,680 | 33 | (31 | ) | 37,682 | |||||||||||
Certificates of deposit | 19,689 | 16 | (38 | ) | 19,667 | |||||||||||
Asset-backed securities | 6,598 | 3 | (47 | ) | 6,554 | |||||||||||
Total assets | $ | 151,965 | $ | 64 | $ | (366 | ) | $ | 151,663 | |||||||
|
| Derivative Liability |
| |
|
| (000"s) |
| |
Balance at January 1, 2019 |
| $ | 390 |
|
Change in fair value |
| 104 |
| |
Balance at December 31, 2019 |
|
| 494 |
|
Change in fair value |
| 556 |
| |
Payment of success fee |
|
| (1,050 | ) |
Balance at December 31, 2020 |
| $ | — |
|
As of December 31, | ||||||||
2023 | 2022 | |||||||
Maturing in one year or less | 93,117 | 134,620 | ||||||
Maturing after one year through two years | 35,116 | 17,043 | ||||||
Total | $ | 128,233 | $ | 151,663 | ||||
| Property and Equipment |
|
Property and equipment consist of the following:
|
| December 31, |
| December 31, | ||||||||||||
|
| 2020 |
|
| 2019 |
| 2023 | 2022 | ||||||||
|
| (000's) |
| (000's) | ||||||||||||
Equipment |
| $ | 5,356 |
|
| $ | 7,230 |
| $ | 5,167 | $ | 5,137 | ||||
Furniture and fixtures |
|
| 629 |
|
|
| 629 |
| 22 | 22 | ||||||
Leasehold improvements |
|
| 896 |
|
|
| 896 |
| 1,612 | 1,612 | ||||||
Software |
|
| 180 |
|
|
| 669 |
| 364 | 364 | ||||||
Assets not yet placed in service |
|
| — |
|
|
| 230 |
| ||||||||
Total property and equipment–at cost |
|
| 7,061 |
|
|
| 9,654 |
| 7,165 | 7,135 | ||||||
Less: Accumulated depreciation |
|
| (5,935 | ) |
|
| (5,903 | ) | ||||||||
Less: Accumulated depreciation and amortization | (6,267 | ) | (5,568 | ) | ||||||||||||
Property and equipment, net |
| $ | 1,126 |
|
| $ | 3,751 |
| $ | 898 | $ | 1,567 | ||||
Total depreciation and amortization expense for yearyears ended December 31, 20202023 and 2019,2022, was $2.4$0.7 million and $2.1$0.8 million, respectively.
| Accrued Expenses |
|