UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
|
| | | | |
☒☒
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 20192022
OR
|
| | | | |
☐☐
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 FOR THE TRANSITION PERIOD FROM TO |
Commission File Number 001-37449
ALPINE IMMUNE SCIENCES, INC.
(Exact name of Registrant as specified in its Charter)
|
| | | | | | | |
Delaware | 20-8969493 |
(State or other jurisdiction of
incorporation or organization)
| (I.R.S. Employer
Identification No.)
|
| | |
|
188 East Blaine Street | Suite 200 | 98102 |
Seattle, | WA |
(Address of principal executive offices) | (Zip Code) |
Registrant’sRegistrant’s telephone number, including area code: (206) (206) 788-4545
Securities registered pursuant to Section 12(b) of the Act:
|
| | | | | | | |
Title of each class | Trading Symbol | Name of each exchange on which registered |
Common Stock, par value $0.001 per share | ALPN | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐☐ No No ☒☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes ☐☐ No No ☒☒
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrantregistrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes Yes☒ No ☒ NO ☐☐
Indicate by check mark whether the Registrantregistrant has submitted electronically every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§(§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes Yes☒ No ☒ NO ☐☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large“large accelerated filer,” “accelerated” “accelerated filer,” “smaller” “smaller reporting company,”” and “emerging“emerging growth company”company” in Rule 12b-2 of the Exchange Act.
|
| | | | | | | | | | | | | | | | | | | |
Large accelerated filerAccelerated Filer | | ☐☐
| | Accelerated Filer | | Accelerated filer☐
|
| ☐
|
| | | | | |
|
|
|
Non-accelerated filerFiler | | ☒
☒ | | Smaller Reporting Company | | Smaller reporting company
|
| ☒
☒ |
| | | | | | |
|
Emerging growth companyGrowth Company | | ☒
☐ | | | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐☐ No ☒☒
The aggregate market value of the voting and non-voting common equity held by non-affiliates of the Registrant,registrant, based on the closing price of the shares of common stock on the Nasdaq Stock Market on June 28, 2019,30, 2022, was approximately $21.4 million.$132.3 million. Shares of common stock held by each executive officer and director and by each other person who may be deemed to be an affiliate of the Registrant,registrant, have been excluded from this computation. The determination of affiliate status for this purpose is not necessarily a conclusive determination for other purposes.
The number of shares of registrant’sthe registrant’s common stock outstanding as of February 29, 2020March 14, 2023 was 47,756,907.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant’s Definitive Proxy Statement relating to the 2023 Annual Meeting of Stockholders, which will be filed with the Securities and Exchange Commission subsequent to the date hereof, are incorporated by reference into Part III of this Report. Such Proxy Statement will be filed with the Securities and Exchange Commission not later than 120 days following the end of the Registrant’s fiscal year ended December 31, 2022.
Table of Contents
In this report, unless otherwise stated or as the context otherwise requires, references to “Alpine,” “the Company,” “we,” “us,” “our” and similar references refer to Alpine Immune Sciences, Inc. “VIGD,”“SIP,” and “TIP” are registered trademarks and “NEON-1,” “NEON-2,” “SYNERGY,” “RUBY” and the Company logo are trademarks of Alpine Immune Sciences, Inc. in various jurisdictions. All rights reserved. This report also contains registered marks, trademarks, and trade names of other companies. All other trademarks, registered marks, and trade names appearing in this report are the property of their respective holders.
Forward-Looking StatementsOur business is subject to numerous risks and uncertainties, including those highlighted in the section of this report captioned “Risk Factors.” The following is a summary of the principal risks we face:•Our approach to the discovery and development of innovative therapeutic treatments based on our technology is unproven and may not result in marketable products.
•Our therapeutic candidates are in early stages of development and may fail in development or suffer delays that materially and adversely affect their commercial viability.
•Product development involves a lengthy and expensive process with an uncertain outcome, and results of earlier preclinical and clinical trials may not be predictive of future clinical trial results.
•We face competition from entities that have developed or may develop therapeutic candidates for our target disease indications, including companies developing novel treatments and technology platforms based on modalities and technology similar to us.
•To date, our revenue has been primarily derived from our collaboration agreements, and our success will be dependent, in part, on our collaborators’ efforts to develop our therapeutic candidates.
•If third parties on which we depend to conduct our clinical or preclinical studies, or any future clinical trials, do not perform as expected, fail to satisfy regulatory or legal requirements, or miss expected deadlines, our development program could be delayed, which may result in materially adverse effects on our business, financial condition, results of operations, and prospects.
•We may not successfully engage in strategic transactions, including any additional collaborations we seek, which could adversely affect our ability to develop and commercialize therapeutic candidates, impact our cash position, increase our expenses, and present significant distractions to our management.
•If any of our therapeutic candidates are approved for marketing and commercialization and we are unable to develop sales, marketing and distribution capabilities on our own or enter into agreements with third parties to perform these functions on acceptable terms, we may be unable to successfully commercialize any such future products.
•We will need to raise substantial additional funds to advance development of our therapeutic candidates, and we cannot guarantee we will have sufficient funds available in the future to develop and commercialize our current or future therapeutic candidates.
•We are an early-stage biopharmaceutical company with a history of losses, we expect to continue to incur significant loses for the foreseeable future, we may never achieve or maintain profitability, and we have a limited operating history that may make it difficult for investors to evaluate the potential success of our business.
•Our computer systems, or those of any of our CROs, manufacturers, other contractors or consultants or potential future collaborators, may fail or suffer security or data privacy breaches or incidents or other unauthorized or improper access to, use of, or destruction of our proprietary or confidential data, employee data, or personal data, which could result in additional costs, loss of revenue, significant liabilities, harm to our brand and material disruption of our operations.
•If we are not able to obtain and enforce patent protection for our technology, including therapeutic candidates, therapeutic products, and platform technology, development of our therapeutic candidates and platform, and commercialization of our therapeutic products may be materially and adversely affected.
•We may license patent rights from third-party owners or licensees. If such owners or licensees do not properly or successfully obtain, maintain or enforce the patents underlying such licenses, or if they retain or license to others any competing rights, our competitive position and business prospects may be materially and adversely affected.
•We or our licensors, collaborators, or any future strategic partners may become subject to third-party claims or litigation alleging infringement of patents or other proprietary rights or seeking to invalidate patents or other proprietary rights, and we may need to resort to litigation to protect or enforce our patents or other proprietary rights, all of which could be costly, time consuming, delay or prevent the development of our therapeutic candidates and commercialization of our therapeutic products, or put our patents and other proprietary rights at risk.
•If we fail to comply with our obligations under any license, collaboration, or other agreements, we may be required to pay damages and could lose intellectual property rights necessary for developing and protecting our
technology, including our platform technology, therapeutic candidates, and therapeutic products, or we could lose certain rights to grant sublicenses, either of which could have a material adverse effect on our results of operations and business prospects.
•We may be unable to obtain U.S. or foreign regulatory approval and, as a result, may be unable to commercialize our therapeutic candidates.
•The healthcare industry is heavily regulated in the U.S. at the federal, state, and local levels and in other jurisdictions in which we may conduct trials or other activities, and our failure to comply with applicable requirements may subject us to penalties and negatively affect our financial condition.
•Our stock price may be volatile, and an active, liquid, and orderly trading market may not develop for our common stock. As a result, stockholders may not be able to resell shares at or above their purchase price.
•Our officers and directors, and their respective affiliates, have a controlling influence over our business affairs and may make business decisions with which stockholders disagree and which may adversely affect the value of their investment.
Our Risk Factors are not guarantees that no such conditions exist as of the date of this report and should not be interpreted as an affirmative statement that such risks or conditions have not materialized, in whole or in part.
Forward-Looking Statements
This Annual Report on Form 10-K contains forward-looking statements and information within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, which are subject to the “safe harbor”“safe harbor” created by those sections. In some cases you can identify these statements by forward-looking words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “could,” “would,” “project,” “plan,” “expect,”“believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “could,” “would,” “project,” “plan,” “expect,” or similar expressions, or the negative or plural of these words or expressions. You should read these statements carefully because they discuss future expectations, contain projections of future results of operations or financial condition, or state other “forward-looking”“forward-looking” information. These statements relate to our future plans, objectives, expectations, intentions and financial performance and the assumptions that underlie these statements. These forward-looking statements include, but are not limited to:
•our ability to identify, develop and commercialize additional products or product candidates;
| |
••our estimates regarding our expenses, revenues, anticipated capital requirements and our needs for additional financing; •our ability to obtain funding for our operations; •the implementation of our business model and strategic plans for our business and technology; •the timing of the commencement, progress and receipt of data from any of our preclinical and clinical trials; •the expected results of any preclinical or clinical trial and the impact on the likelihood or timing of any regulatory approval; •the scope of protection we are able to establish and maintain for intellectual property rights covering our technology and product candidates; •the anticipated impact of the COVID-19 pandemic on our business, research and clinical development plans and timelines and results of operations; •the timing or likelihood of regulatory filings and approvals; •the therapeutic benefits, effectiveness and safety of our product candidates; •the rate and degree of market acceptance and clinical utility of any future products; •our ability to maintain and establish collaborations; •our ability to achieve milestones in our current and any future collaborations; •our expectations regarding market risk, including interest rate changes and general macroeconomic conditions; •our expectations regarding the sufficiency of our cash and cash equivalents to fund operations for at least the next 12 months; •developments relating to our competitors and our industry; and • | our ability to identify, develop and commercialize additional products or product candidates;
|
| |
•
| our estimates regarding our expenses, revenues, anticipated capital requirements and our needs for additional financing;
|
| |
•
| our ability to obtain funding for our operations;
|
| |
•
| the implementation of our business model and strategic plans for our business and technology;
|
| |
•
| the timing of the commencement, progress and receipt of data from any of our preclinical and clinical trials;
|
| |
•
| the expected results of any preclinical or clinical trial and the impact on the likelihood or timing of any regulatory approval;
|
| |
•
| the scope of protection we are able to establish and maintain for intellectual property rights covering our technology and product candidates;
|
| |
•
| the anticipated impact of the COVID-19 pandemic on our business, research and clinical development plans and timelines and results of operations;
|
| |
•
| the timing or likelihood of regulatory filings and approvals;
|
| |
•
| the therapeutic benefits, effectiveness and safety of our product candidates;
|
| |
•
| the rate and degree of market acceptance and clinical utility of any future products;
|
| |
•
| our ability to maintain and establish collaborations;
|
| |
•
| our ability to achieve milestones in our current and any future collaborations;
|
| |
•
| our expectations regarding market risk, including interest rate changes;
|
| |
•
| our expectations regarding the sufficiency of our cash and cash equivalents to fund operations for at least the next 12 months;
|
| |
•
| developments relating to our competitors and our industry; and
|
| |
•
| our expectations regarding licensing, acquisitions and strategic operations. |
These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from those anticipated in the forward-looking statements. Factors that might cause such a difference include, but are not limited to, those discussed in this report in Part I, Item 1A. Risk Factors, and elsewhere in this report. Forward-looking statements are based on our management’smanagement’s beliefs and assumptions and on information currently available to our management. These statements, like all statements in this report, speak only as of their date, and we undertake no obligation to update or revise these statements in light of future developments, except as required by law. In addition, statements that “we believe”“we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements.
PART I
Item 1. Business
Overview
We are a clinical-stage biopharmaceutical company dedicated to discovering and developing innovative, protein-based immunotherapies targeting the immune synapse to treat cancer and autoimmune and inflammatory diseases. Our approach includes a proprietary scientific platform that converts native immune system proteins into differentiated, multi-targeted therapeutics potentially capable of modulating the human immune system and significantly improvingtherapeutics. We are seeking to create first- or best-in-class multifunctional immunotherapies via our unique protein engineering technologies to improve outcomes in patients with serious diseases.
Our lead programAutoimmune and Inflammatory Diseases
ALPN-303, or povetacicept, is ALPN-101, a dual antagonist of the B cell activating factor, or BAFF, and a proliferation inducing ligand, or APRIL, cytokines, which play key roles in the activation, development, and survival of B cells. Based upon an engineered transmembrane activator and CAML interactor, or TACI, domain, povetacicept has exhibited greater potency in preclinical studies versus wild-type TACI-based comparators, as well as other inhibitors of BAFF and/or APRIL alone. In addition, povetacicept has been well-tolerated in preclinical models and exhibited superior pharmacokinetics and pharmacodynamics over wild-type TACI-Fc counterparts, including superior serum exposure, suppression of T-dependent antibody production, and/or serum immunoglobulins in mice and/or cynomolgus monkeys. In a randomized, placebo-controlled, first-in-human, Phase 1 study in adult healthy volunteers (NCT05034484), povetacicept has been well tolerated to date and has demonstrated encouraging dose-related pharmacokinetic and on-target pharmacodynamic effects, which we believe supports the use of a once every four-week dose regimen for subsequent studies and enables a broad development plan in multiple indications. Povetacicept is in development for multiple B cell and/or autoantibody-related diseases, including systemic lupus erythematosus, or SLE, glomerulonephritides, and autoimmune cytopenias.
ALPN-101, or acazicolcept, is a dual Inducible T cell Costimulator, or ICOS, and CD28 antagonist intended for the treatment of autoimmune and inflammatory diseases. Preclinical studies with acazicolcept have demonstrated efficacy in models of graft versus host disease,SLE, Sjögren’s syndrome, or GVHD,SjS, arthritis, inflammatory bowel disease, multiple sclerosis, type 1 diabetes, systemic lupus erythematosus, or SLE,uveitis, and Sjögren’s syndrome. In an oral presentation at the 2019 American Society of Hematology Annual Meeting, we discussed data from ourgraft versus host disease. We have evaluated acazicolcept in a Phase 1 healthy volunteer study demonstrating that ALPN-101 was well-tolerated as single intravenous or subcutaneous doses, without cytokine release, infusion-related reactions, hypersensitivity, or other signs of agonist activity. Based on ourand are currently evaluating acazicolcept in Synergy, a global, randomized, double-blind, placebo-controlled Phase 1 data, we opened BALANCE for enrollment, an open-label, dose escalation, and expansion Phase 1b/2 study of acazicolcept in patientsadults with steroid-resistantmoderate-to-severe SLE. In June 2020, we entered into an Option and License Agreement with AbbVie Ireland Unlimited Company, or steroid-refractory active acute GVHD. We intendAbbVie, which grants AbbVie an exclusive option to enroll patients throughout 2020take an exclusive license to acazicolcept. Through December 31, 2022, we have received $105.0 million in upfront and pre-option exercise development milestones as part of the Option and License Agreement with AbbVie, or the AbbVie Agreement.
In December 2021, we entered into a license and collaboration agreement, or the Horizon Agreement, with Horizon Therapeutics Ireland DAC, or Horizon, which grants Horizon an exclusive license for the development, manufacture and commercialization of one of our existing preclinical biologic therapeutic programs, or the Existing Program, and up to three additional autoimmune and inflammatory disease programs for other designated biological targets, or the Research Programs, generated from our libraries of proteins and molecules for research, discovery and identification of additional compounds. Under the terms of the Horizon Agreement, Horizon made an upfront payment to us of $25.0 million as well as an equity investment for which they paid $15.0 million, a 25% premium to the 30-day volume-weighted average share price as of December 9, 2021. Beyond acute GVHD,In addition, we believe that ALPN-101 has the potentialare eligible to be effectivereceive up to $381.0 million per program, or approximately $1.5 billion in inflammatory diseases like rheumatoid arthritis, SLE,total, in future success-based payments related to development, regulatory and Sjögren’s syndrome.commercial milestones as well as tiered royalties on global net sales.
Our lead oncology programImmuno-oncology
ALPN-202, or davoceticept, is ALPN-202, a conditional CD28 costimulator and dual checkpoint inhibitor intended for the treatment of cancer. Preclinical In October 2022, we announced the voluntary termination of enrollment of davoceticept clinical studies, including the NEON-1 study of davoceticept as monotherapy and the NEON-2 study of davoceticept in vivo data have demonstrated monotherapy efficacycombination with pembrolizumab. The decision to terminate enrollment in tumor models superior to approved therapies. In addition, ALPN-202 hasthe davoceticept studies was made following notification of a unique immuno-modulatory profile and has demonstrated evidence of anti-tumor immunitysecond Grade 5 serious adverse event (death) in preclinical models. Based on ALPN-202’s efficacy in preclinical models and favorable nonclinical safety and development profile, we have opened NEON-1 for enrollment, a Phase 1 dose escalation and expansion study in patients with advanced malignancies. We intend to enroll patients throughout 2020 and into 2021.the NEON-2 study.
Scientific Platform
Our scientific platform has also generated immune modulatory proteins with the potential of improving engineered cellularcell therapies or ECT, such as chimeric antigen receptor T cells, or CAR-T, T cell receptor-engineered T cells, or TCR-T, and tumor infiltrating lymphocytes, or TILs.lymphocytes. In
May 2019, we signed a collaboration and license agreement with Adaptimmune Therapeutics plc, or Adaptimmune, to develop next-generation SPEARTM T-cellSPEAR™ T cell products which incorporate our secreted and transmembrane immunomodulatory protein, (termed SIP™termed SIP™ and TIP™)TIP™, technology. We intend to continue to leverage our existing pipeline and platform to actively explore and evaluate potential value-creating partnering opportunities.
Our Strategy
Our goal is to discover and develop modern therapies to treat patients with serious conditions such as cancerautoimmune and inflammatory diseases. To achieve our goals, we intend to:
Aggressively•aggressively move our lead inflammation/autoimmunewholly-owned program, ALPN-101 throughpovetacicept, into clinical developmentstudies for the treatment of inflammatory diseases.multiple B cell and/or autoantibody-related diseases;
| |
•
| We intend to enroll patients in BALANCE, an open-label, dose escalation, and expansion Phase 1b/2 study in patients with active acute GVHD throughout 2020 and into 2021.
|
Aggressively•aggressively move our lead oncologymost advanced autoimmune and inflammatory program ALPN-202acazicolcept through clinical development as part of our collaboration with AbbVie, including conducting Synergy, our Phase 2 study for the treatment of cancer.SLE; and
| |
•
| We intend to enroll patients in NEON-1, a Phase 1 dose escalation and expansion clinical study in patients with advanced malignancies throughout 2020 and into 2021.
|
Maximize•maximize the value of our pipeline and platform via potential partnering activities.
| |
•
| We intend to continue to leverage our existing pipeline and platform to actively explore and evaluate potential value-creating partnering opportunities.
|
Product Pipeline
We have a diverse pipeline of novel therapies, as shown in Figure 1 below. In 2019, we completed enrollment in our Phase 1 healthy volunteer study of ALPN-101. We are open for enrollment in our study of ALPN-101 in acute GVHD and are considering additional indications, such as rheumatoid arthritis, SLE or Sjögren’s syndrome. We also are open for enrollment in our Phase 1 dose escalation and expansion study of ALPN-202 in advanced malignancies.
Figure 1
Our Scientific Platform
The human immune system is a complex network of biological processes and structures evolved to protect humans from external infections and harmful changes of internal cells. Within the immune system, proteins play a key role in a variety of essential functions, including recognition of foreign and self-antigens, cell adhesion and trafficking, and modulation of cellular activity through costimulatory or inhibitory signaling. Our scientific platform seeks to develop novel therapeutics by engineering native, or so-called “wild-type,”“wild-type,” proteins with unique properties that may benefit patients with cancer orautoimmune and inflammatory diseases. We have focused our efforts to-date on two major protein superfamilies that play critical roles in the regulation of immune cell signaling and activity: the immunoglobulin superfamily, (IgSF)or IgSF, and the tumor necrosis factor (receptor) superfamily, (TNFSF/TNFRSF). or TNFSF/TNFRSF.
The IgSF is the largest family of adhesion, costimulatory (activating), and inhibitory (blocking) proteins found on the surface of immunological, neurological, and other human cell types. These cell surface and soluble molecules are broadly involved with recognition of antigens, assisting in the formation of the immune synapse, and performing costimulatory, coinhibitory, and cytokine receptor signaling functions. This family includes many well-known targets, such as those seen in
Figure 2. We believe the IgSF protein family members may be particularly valuable because many IgSF proteins naturally bind multiple binding partners, also referred to as “counterstructures.” ALPN-101“counterstructures.” Acazicolcept and ALPN-202davoceticept are both derived from members of the IgSF.
Figure 2
TNFSF/TNFRSF proteins are expressed broadly in the immune system and play a critical role in immune cell signaling and proliferation. TNFSF/TNFRSF members are composed of 48 unique proteins that are structurally similar and are characterized by their ability to bind to trimeric tumor necrosis factors.factors (Figure 3). Members of the TNFSF/TNFRSF include many clinically relevant targets with applications in both autoimmune disease and immuno-oncology (e.g., CD40, TACI, BCMA, 4-1BB, TNFαTNFα). Several candidates developed as part of our ALPN-303 program were derived from the TNFSF/TNFRSF.
The TNF Superfamily
Figure 3
Our scientists create engineered proteins from IgSF members (variant immunoglobulin domains, or vIgDs) and TNFSF/TNFRSF members (variant TNF domains, or vTDs). We use directed evolution, which is an iterative scientific engineering process purposefully conducted to modify an IgSF andor TNFSF/TNFRSF protein for a desired therapeutic function. The potential to create therapies capable of working within a formed immune synapse, forcing a synapse to occur, or preventing a synapse from occurring are important, novel attributes of our scientific platform.
Figure 34 illustrates the process of directed evolution in our scientific platform. Our scientists utilize yeast display protein library strategies to identify variants of wild-type proteins with desired binding characteristics. We start with a wild-type IgSF or TNFSF/TNFRSF protein and then enter a cycle of library generation and yeast display. Flow cytometry or other methods are used to sort for yeast clones displaying variants with desired binding characteristics. Biologic and biophysical assays of purified proteins assess biological function and manufacturing characteristics. The end product is an optimized variant Ig domain, vIgD, or variant TNF domain, or vTD. Additional cycles can be carried out by building next generation libraries from the output of prior libraries to result in further optimization.
Figure 34
Our scientific platform is generally able to improve upon native IgSF or TNFSF/TNFRSF activity regardless of whether natural binding affinity is weak or strong. When starting affinity is very weak, techniques employed by our scientists have accomplished several thousand-fold increases in binding affinity with sometimes as few as two library generation cycles. Even when starting affinity is very high, our scientific platform can still improve binding affinities. The same general strategies can be used when the desired therapeutic profile requires reduced affinity compared to the wild-type protein. We have applied our scientific platform to several IgSF and TNFSF/TNFRSF protein targets, including the disclosed programs in Figure 4.
Figure 4
We believe our vIgDs and vTDs are highly flexible. In many cases, a single affinity-maturation campaign can result in multiple domains suitable for use in the formats such as those illustrated in Figure 5 and further described below.
Figure 5
vIgD-Fc or vTD-Fc
A vIgD- or vTD-Fc fusion protein is the simplest format. Our lead autoimmune/inflammation program, ALPN-101,Acazicolcept and lead oncology program, ALPN‑202,povetacicept are both examples of vIgD-Fc formats.and vTD-Fc formats, respectively. The engineered vIgD or vTD protein is fused to an Fc backbone. Combining vIgDs or vTDs with antibody Fc domains to make Fc fusion proteins potentially allows for better expression, facilitates purification, and improves pharmacokinetic (dosing) properties. Fc fusion proteins are a standard format in the industry, with examples such as etanercept, abatacept, and belatacept. A vIgD- or vTD-Fc could potentially be administered intravenously, subcutaneously, topically, or via other methods of delivery.
Cell Therapy Enhancement
Ourscientific platform has also generated immune modulatory proteins with the potential of improving engineered cellular therapies, such as CAR-Ts, TCR-Ts, or TILs.
Povetacicept, a Dual B Cell Cytokine Antagonist for B Cell-Mediated Autoimmune and Inflammatory Diseases
Overview
Povetacicept is an Fc fusion of an engineered variant TACI domain, structurally distinct from wild-type TACI-Ig and designed to more potently inhibit the pleiotropic B cell cytokines B cell activating factor, or BAFF, BLyS, and a proliferation inducing ligand, or APRIL (Figure 6).
Figure 6
BAFF and APRIL play complementary and partially overlapping roles in B cell development, activation, and survival. Early in B cell development, BAFF plays a dominant role related to BAFF-R expression, while later stages of B cell development are APRIL and/or BAFF-dependent, related to TACI and BCMA expression (Figure 7). Importantly, plasmablasts and plasma cells, which are a dominant source of the pathogenic autoantibodies in lupus and other related diseases, retain responsiveness to APRIL and BAFF. Co-neutralization of APRIL and BAFF dramatically reduces B cell survival and function, including antibody production, whereas inhibition of either BAFF or APRIL alone mediates only modest effects. Povetacicept, with enhanced inhibitory activity against BAFF & APRIL, could further improve clinical outcomes in B cell mediated autoimmune and inflammatory diseases.
Figure 7
Povetacicept Nonclinical Development
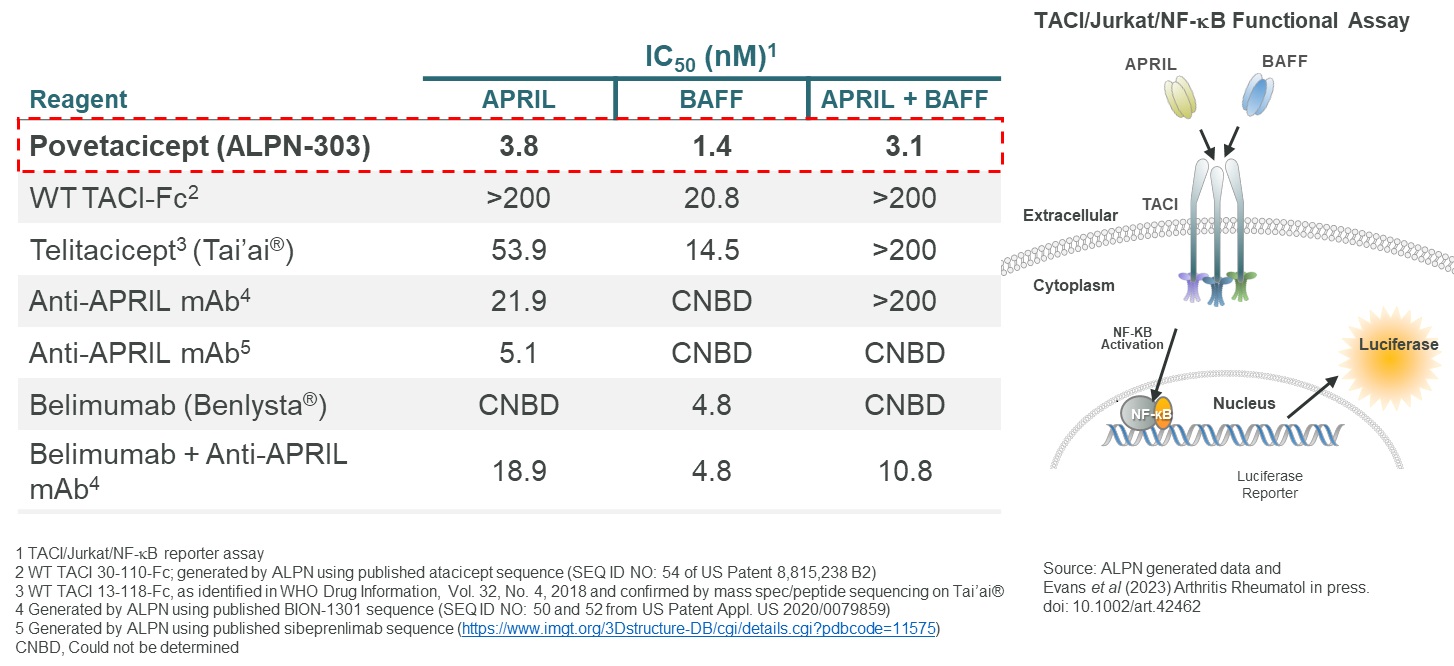
The engineering of the active domain used in povetacicept was focused on increasing the affinity of wild-type TACI against both BAFF and APRIL, with APRIL of particular interest given that wild-type TACI appears to be a relatively weak inhibitor of APRIL. Data presented at the 2022 EULAR Annual European Congress demonstrated that relative to wild-type TACI, povetacicept had improved affinity against BAFF 8 to 10-fold, and dramatically improved affinity against APRIL suggesting that povetacicept may be the first truly dual, potent inhibitor of these two cytokines. In Figure 8, WT TACI-Fc binds APRIL poorly by surface plasmon resonance, or SPR, with APRIL binding significantly improved by povetacicept.
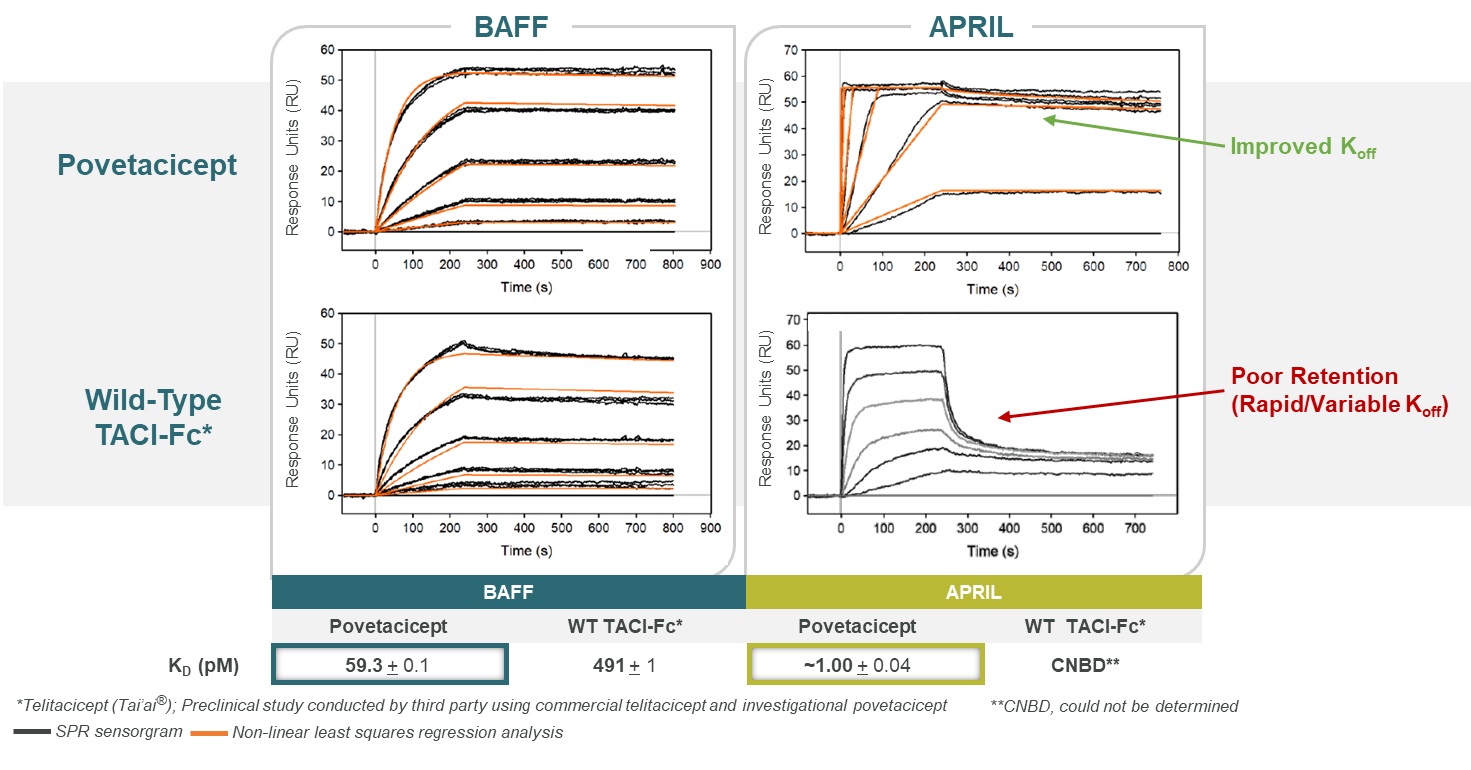 Figure 8
Figure 8CollaborationPovetacicept’s ability to inhibit the activity of APRIL and BAFF alone or in combination was evaluated in a TACI/Jurkat/NF-kB in vitro reporter assay, in comparison to wild-type TACI-Fc’s, belimumab, anti-APRIL mAbs, or the combination of anti-APRIL mAb plus belimumab. Based upon IC50, povetacicept more effectively inhibited APRIL activity than either of the two anti-APRIL mAbs evaluated, and inhibited BAFF activity > 3-fold more potently than belimumab (Figure 9). In addition, povetacicept most potently inhibited the combined activity of APRIL and BAFF, with Adaptimmune Therapeuticsa >50-fold better IC50 relative to telitacicept, and a > 3-fold better IC50 relative to the combination of anti-APRIL mAb plus belimumab.
Figure 9
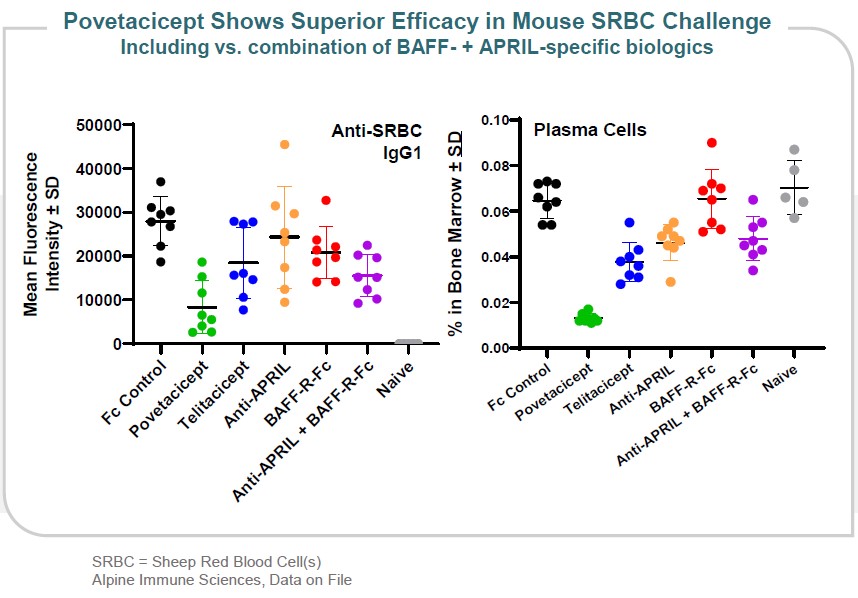
The improved affinity of povetacicept for both BAFF and APRIL correlates with significantly improved efficacy in multiple preclinical models. For example, in a sheep red blood cell challenge model, povetacicept resulted in the most potent reduction of antibody responses and plasma cell formation – compared to wild-type TACI, single inhibitors of BAFF or APRIL, or the combination of the two BAFF- and APRIL-specific biologics (Figure 10).
Figure 10
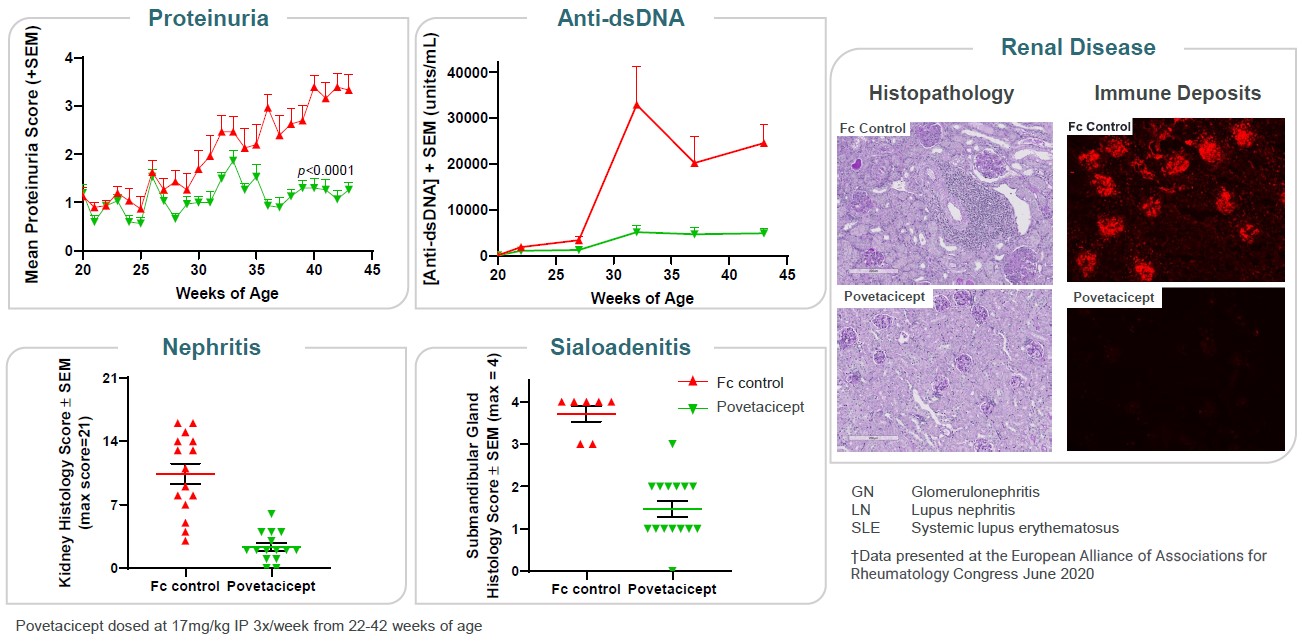
Furthermore, treatment with povetacicept potently suppressed anti-dsDNA auto antibodies, proteinuria, sialoadenitis, kidney lesions, and renal immune complex deposition in the (NZBxNZW)F1 lupus model (Figure 11).
Figure 11
In addition to advancing programs internally, we continue to seek partners who can bring therapeutic area experience,potential development expertise, commercialization capabilities,of povetacicept in SLE and funding allowing us to maximize the potential of vIgDsglomerulonephritis (e.g., IgA nephropathy, membranous nephropathy, and our scientific platform. In May 2019, we entered into a collaboration and license agreement with Adaptimmune Therapeutics to develop next-generation SPEAR™ T-cell products which incorporate our secreted and transmembrane immunomodulatory protein (termed SIP™ and TIP™) technology. We and Adaptimmune will collaborate on a specified number of programs to develop SIP and TIP candidates with tailored affinities and modulatory activities that may enhance the anti-tumor responses seen with Adaptimmune’s SPEAR™ T-cells. For each program, Adaptimmune has an option to take a worldwide exclusive licenselupus nephritis), there is strong rationale for development in cyotpenias including autoimmune hemolytic anemia, or AIHA, and commercializationimmune thrombocytopenic purpura, or ITP, which are diseases characterized by autoantibodies directed against red blood cells and platelets, respectively. AIHA includes both warm AIHA, the most common form of SPEAR™ T-cell products incorporatingAIHA, and cold AIHA, often referred to as cold agglutinin disease, or CAD. Significantly higher levels of both BAFF and APRIL have been observed in the developed SIPserum of patients with AIHA and ITP compared to healthy subjects, and polymorphisms
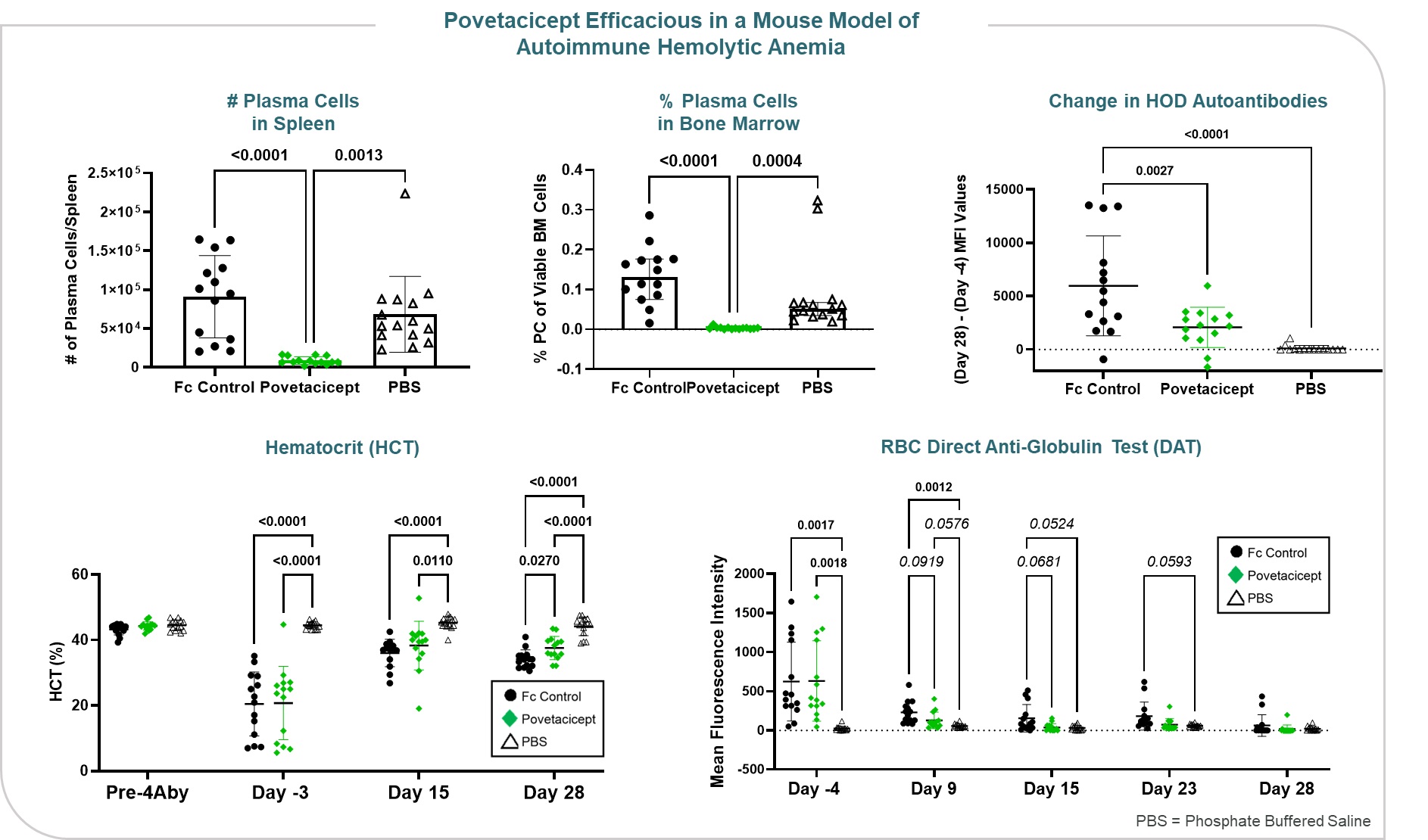
in BAFF and TACI have been associated with ITP. Data presented at ASH 2022 demonstrated that povetacicept was efficacious in a mouse model of AIHA, significantly increasing hematocrit while reducing pathogenic anti-RBC autoantibodies (Figure 12).
Figure 12
In cynomolgus monkeys, povetacicept exhibited pharmacokinetics and pharmacodynamics superior to wild-type TACI-Fc counterparts, including superior serum exposure and pharmacodynamic reduction of serum immunoglobulins (Figure 13). It has been well-tolerated in definitive (GLP) nonclinical toxicology studies, including up to six months in cynomolgus monkeys, without evidence of cytokine release or TIPlocal intolerance. Based on these nonclinical data, we believe that povetacicept represents an attractive development candidate for the treatment of cancer. Under the terms of the collaboration agreement, Adaptimmune provided an upfront paymentmultiple B cell mediated autoimmune and will provide research funding for ongoing programs. In addition, we may be eligible for downstream development and commercialization milestones up to $288 million, if all pre-specified milestones for each program are achieved. In addition, we are eligible to receive low-single digit royalties on worldwide net sales of the applicable products.inflammatory diseases.
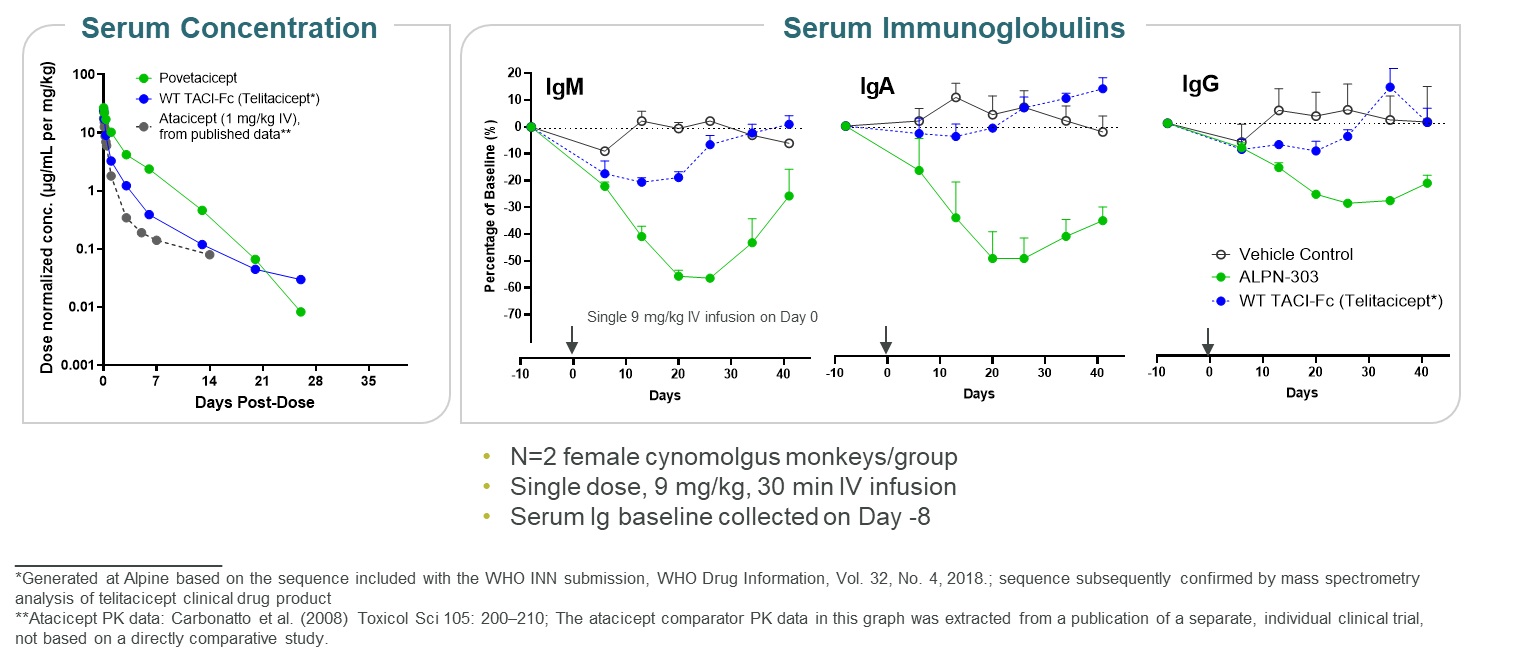
Figure 13
Our scientists and collaborators have demonstrated within in vivo preclinical studies that povetacicept:
ALPN-101,•showed the most potent reduction of antibody responses and plasma cell formation in a sheep red blood cell challenge model compared to wild-type TACI, single inhibitors of BAFF or APRIL, or the combination of the two BAFF- and APRIL-specific biologics;
•potently suppressed anti-dsDNA autoantibodies, sialoadenitis and glomerulonephritis (including proteinuria and renal immune complex deposition) in the (NZBxNZW)F1 lupus model relative to an Fc control;
•significantly increased hematocrit while reducing pathogenic anti-RBC autoantibodies in a mouse model of autoimmune hemolytic anemia (AIHA), relative to an Fc control; and
•is well tolerated in definitive repeat-dose nonclinical toxicology studies, including up to 6 months in cynomolgus monkeys.
Povetacicept Clinical Development
Preliminary findings from RUBY-1, a randomized, placebo-controlled, first-in-human, Phase 1 study of povetacicept in adult healthy volunteers (NCT05034484) were presented at our inaugural R&D Day in September 2022 and at subsequent scientific conferences, including the American Society of Nephrology Kidney Week 2022 Meeting, the ACR Convergence Meeting 2022, and the 2022 American Society of Hematology Annual Meeting.
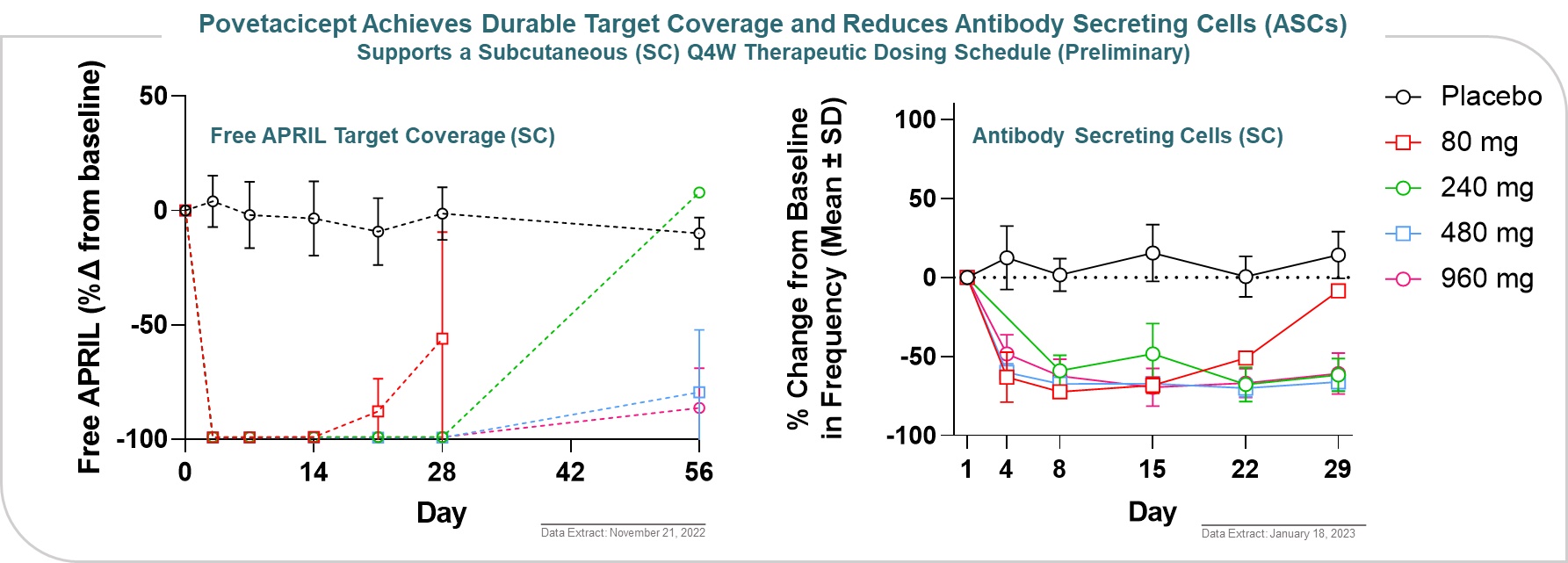
Overall, povetacicept was well tolerated as single intravenous, or IV, or subcutaneous, or SC, doses of up to 960 mg in adult healthy volunteers (Figure 14).
Figure 14
Povetacicept exhibited dose-related pharmacokinetic and on-target pharmacodynamic effects, including target coverage of free APRIL for 2-3 and ≥4 weeks with 80 and 240 mg, respectively and dose-dependent reduction in antibody-secreting cells (ASCs) (Figure 15).
Figure 15
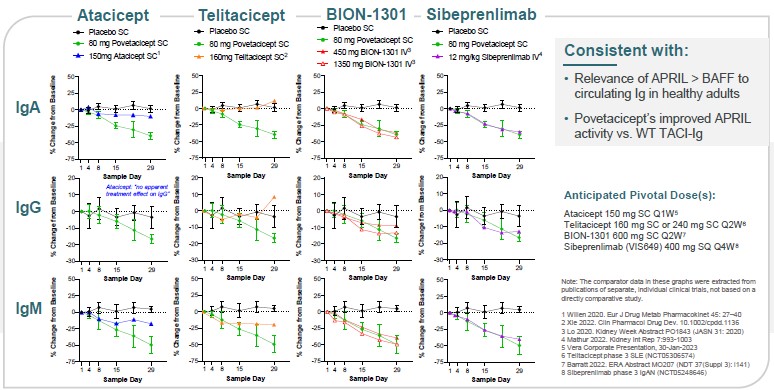
In addition, based on data extracted from publications for other drug products, not based on head-to-head studies, povetacicept reduced serum Ig levels favorably relative to comparator molecules in development. Povetacicept appears to result in greater Ig reductions than wild-type TACI-Fc (atacicept or telitacicept), and comparable Ig reductions versus anti-APRIL antibodies (Figure 16). Overall, these findings suggest that: (1) APRIL appears to play a more dominant role than BAFF in circulating Ig levels, at least in healthy adults; and (2) the observations with povetacicept are consistent with its improved APRIL activity compared to wild-type TACI.
Figure 16
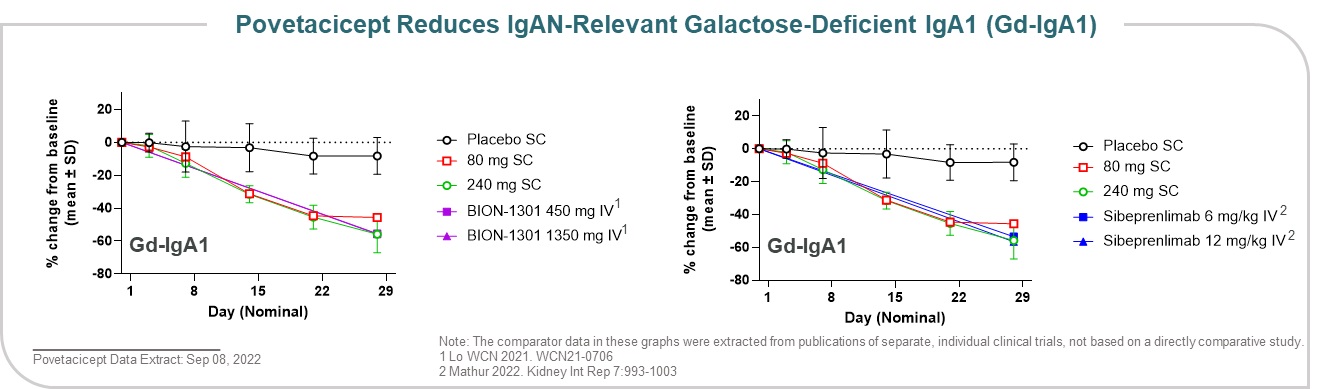
Data presented at the American Society of Nephrology Kidney Week 2022 Meeting demonstrated that povetacicept dose-dependently reduced Gd-IgA1, an important effector molecule and/or biomarker in IgA nephropathy, to a similar degree as comparator molecules when comparing historical data with single dose administration to healthy volunteers (Figure 17). Overall, the encouraging pharmacodynamic effects, including dose-dependent reductions in free cytokine, circulating
immunoglobulins, antibody-secreting cells, and Gd-IgA1, support therapeutic doses to be in the 80-240 mg dose range, administered subcutaneously every 4 weeks.
Figure 17
We believe povetacicept has the potential to become a development pipeline within a single product candidate, with significant market opportunity across multiple therapeutic areas including, but not limited to, nephrology, hematology, and connective tissue diseases such lupus. Based on our encouraging clinical and nonclinical data, we are initiating a broad development plan with multiple planned clinical studies, including RUBY-3, an open-label, dose-ranging basket study in autoimmune glomerulonephritis including IgA nephropathy, lupus nephritis, and primary membranous nephropathy; and RUBY-4, an open-label basket study in autoimmune cytopenias including immune thrombocytopenia, warm autoimmune hemolytic anemia, and cold agglutinin disease. Each of the indications in these basket studies has strong scientific rationale for povetacicept based on the importance of specific autoantibodies in disease pathogenesis, as well as high medical need. We initiated enrollment of RUBY-3 during the first quarter of 2023, and are targeting initiation of RUBY-4 in the second quarter of 2023. We also plan to initiate RUBY-2, a placebo-controlled, dose-ranging, randomized phase 2 study in systemic lupus erythematosus in mid-2024 based on enabling data from RUBY-3 and RUBY-4 (Figure 18). In addition to the initial indications outlined in Figure 18, BAFF and APRIL may also play a role in B-cell mediated diseases within other therapeutic areas such as dermatology (e.g., pemphigus) and neurology (e.g., myasthenia gravis) where B cell-targeting agents have demonstrated activity.
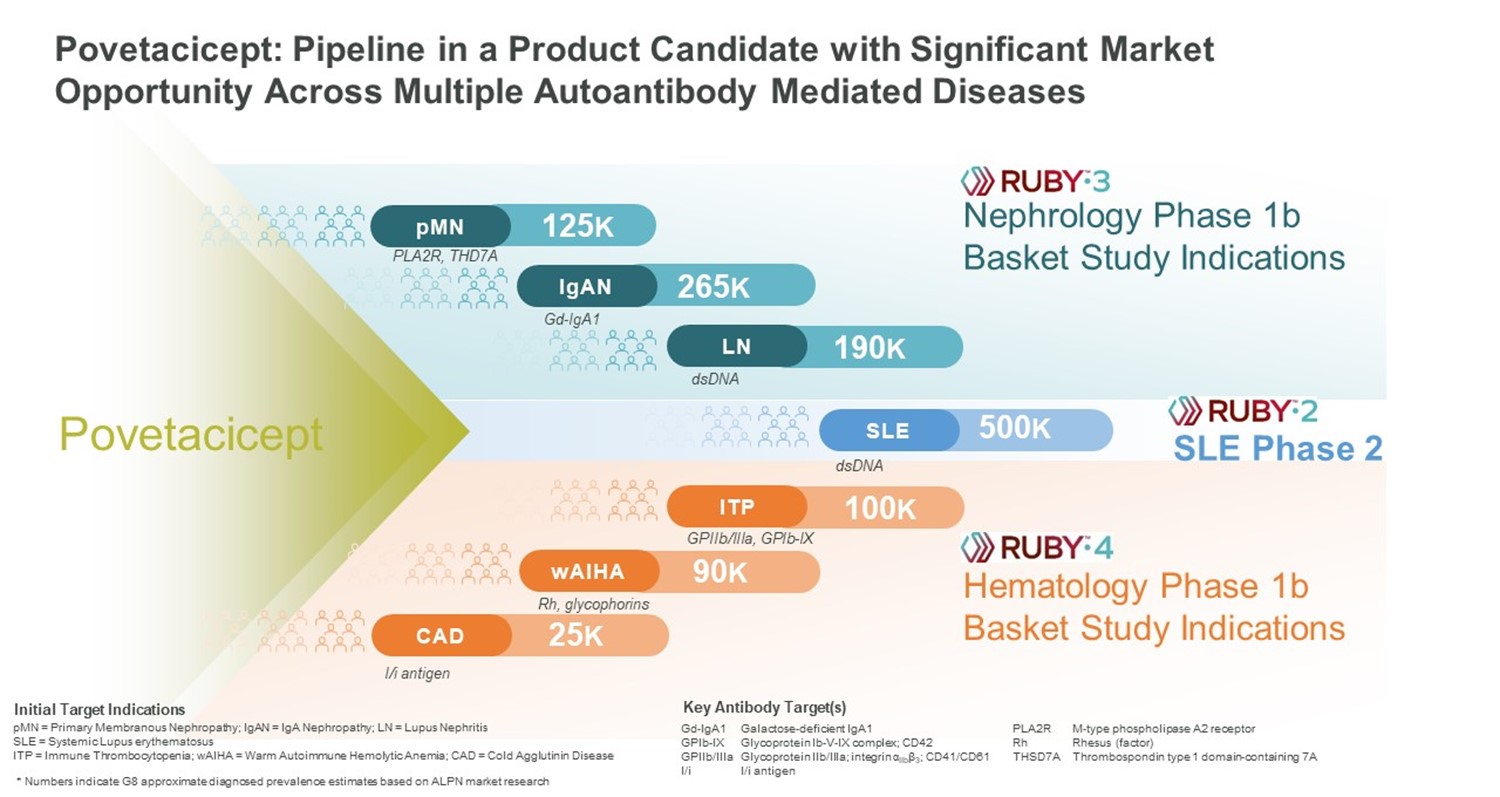
Figure 18
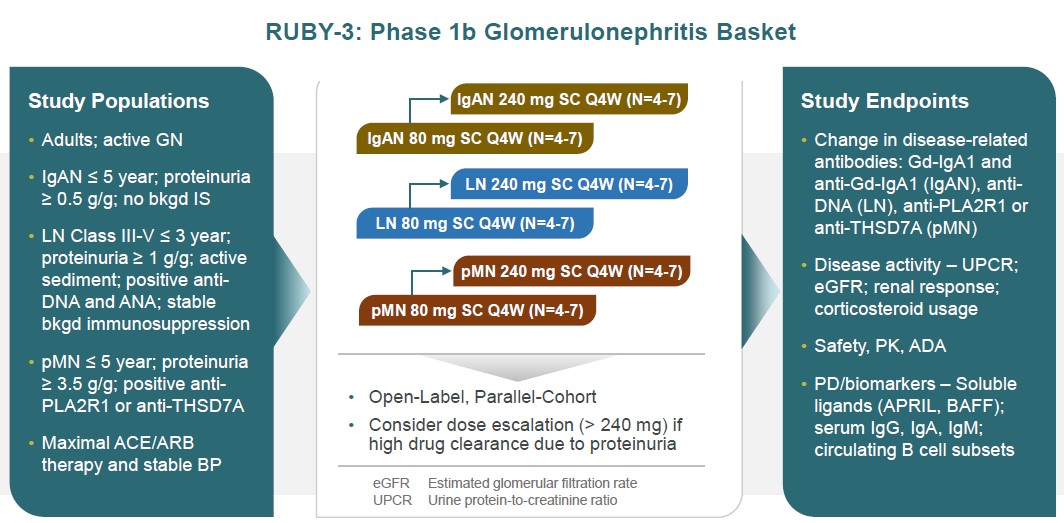
RUBY-3, our Phase 1b glomerulonephritis basket study, is designed as a multi-ascending dose, multi-cohort open label study in subjects with proteinuric IgA nephropathy, lupus nephritis or primary membranous nephropathy in small disease- and dose-specific cohorts for up to 48 weeks. Endpoints will include changes in disease-specific autoantibodies as well as proteinuria, eGFR and various definitions of renal response (Figure 19).
Figure 19
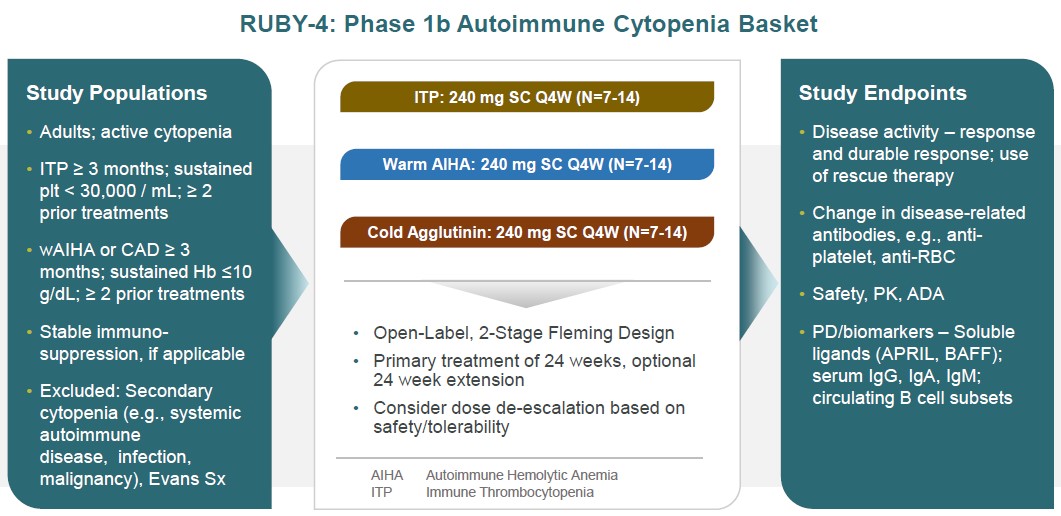
RUBY-4, our autoimmune cytopenia basket study, is designed as a parallel-arm, multi-cohort open label study that seeks to enroll subjects with immune thrombocytopenia, warm autoimmune hemolytic anemia or cold agglutinin disease in disease-specific cohorts, using a 2-stage Fleming design for up to 48 weeks. Subjects must have active cytopenia and endpoints will include standard definitions of response, such as durable responses. Changes in anti-platelet or anti-red blood cell autoantibodies will also be followed (Figure 20).
Figure 20
Acazicolcept, a Dual ICOS/CD28 Antagonist for Autoimmune and Inflammatory Diseases
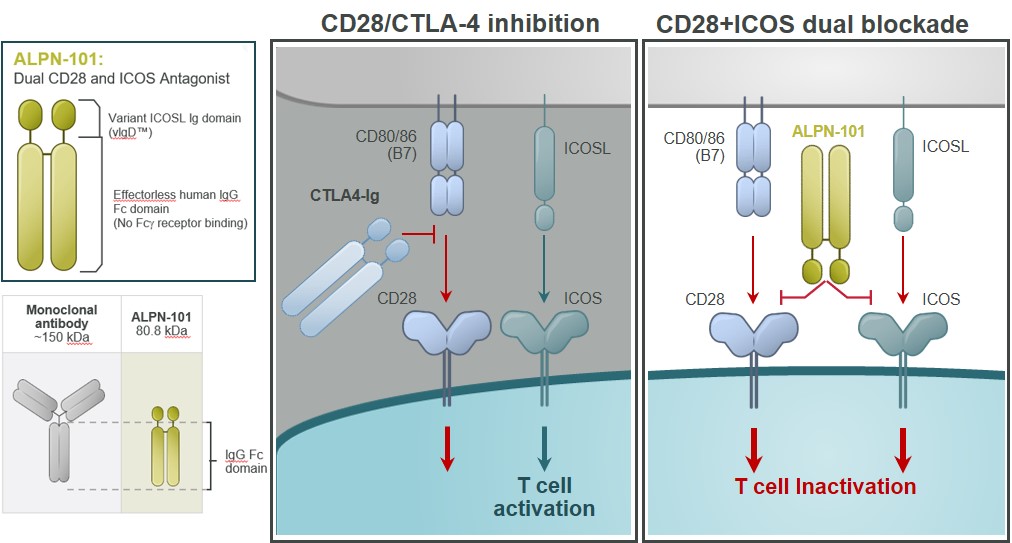
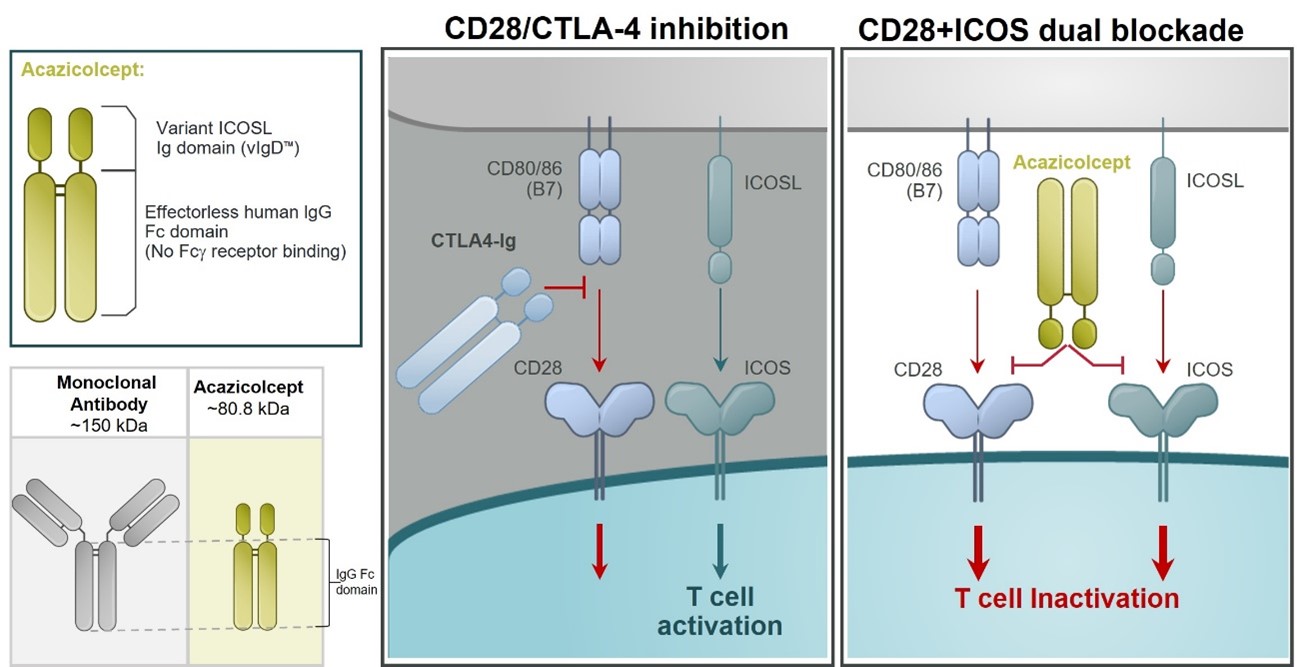
Our lead inflammatory disease program, ALPN-101,Acazicolcept is an Fc fusion protein of a human inducible T cell costimulator ligand, or ICOSL, vIgD designed to inhibit simultaneously the CD28 and ICOS T cell costimulatory pathways (Figure 621). This vIgD is fused to an “effectorless”“effectorless” Fc backbone and is intended for the potential treatment of certainautoimmune and inflammatory diseases. Notably, ALPN-101acazicolcept is not a bispecific antibody construct. A traditional bispecific might be constructed of one domain binding ICOS and one domain binding CD28. Instead, ALPN-101acazicolcept makes use of a novel single vIgD domain capable of binding both ICOS and CD28 engineered by our scientists using our proprietary scientific platform.
Figure 621
CD28 has been long recognized to be required for naïnaïve T cell activation. The therapeutic inhibitors of the CD28 pathway (e.g., abatacept, CTLA4-Ig; and belatacept, a second generation CTLA4-Ig) have proven valuable for the treatment of some inflammatory arthritis conditions (rheumatoid(e.g., rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis) and for the prevention of renal allograft rejection.rejection or graft versus host disease. However, therapeutic blockade of the CD28 pathway, primarily as studied with abatacept, has not been successful in several other inflammatory diseases (e.g., Crohn’sCrohn’s disease, lupus nephritis, multiple sclerosis) despite extensive evidence implicating T cells in disease pathogenesis and evidence of clinical biological activity. This suggests an additional pathogenic costimulatory pathway(s) remains unaddressed.
Inducible T cell Costimulator, or ICOS is part of the CD28 costimulatory family of molecules, including PD-1, CD28, and CTLA-4. ICOS is related to CD28, but, in contrast, is poorly expressed in naïnaïve T cells. ICOS is, however, rapidly induced upon T cell activation. It appears to be a dominant costimulatory pathway in at least some effector or pathogenic T cells, such as potentially in the absence of CD28. Elevated levels of ICOS‑ICOS‑expressing T cells have been described in an increasing number of autoimmune and inflammatory diseases, correlating with disease activity. At the same time, inhibition of ICOS is effective in several preclinical inflammatory disease models. The ICOS pathway may therefore represent a major costimulatory pathway, nonredundant with CD28, and highly relevant to autoimmune and inflammatory diseases.
We have performed a number of pre-clinicalpreclinical experiments demonstrating ALPN‑101acazicolcept is active in both in vitro and in vivo models, several of which are described below.
A potent immunomodulator of diseased cells
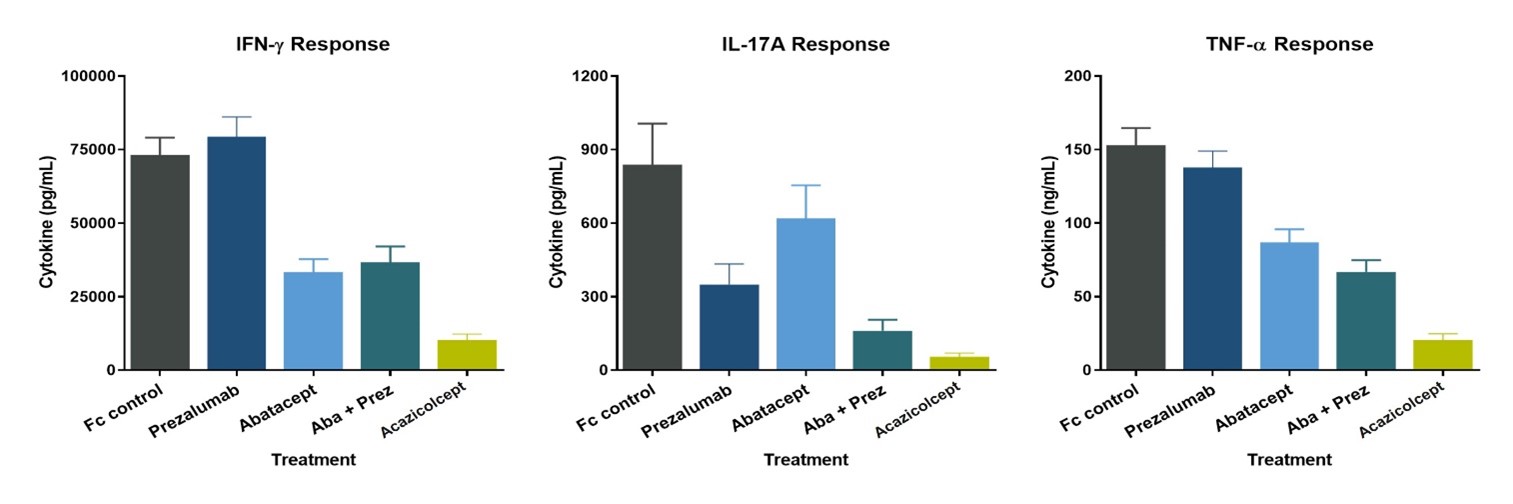
ALPN-101Acazicolcept inhibits cytokine production from human peripheral blood mononuclear cells in vitro more potently than single CD28 (abatacept, or CTLAr-Ig)CTLA4-Ig) or ICOS (prezalumab, or anti-ICOSL mAB)mAb) pathway inhibitors alone or in combination (Figure 722).
Figure 22
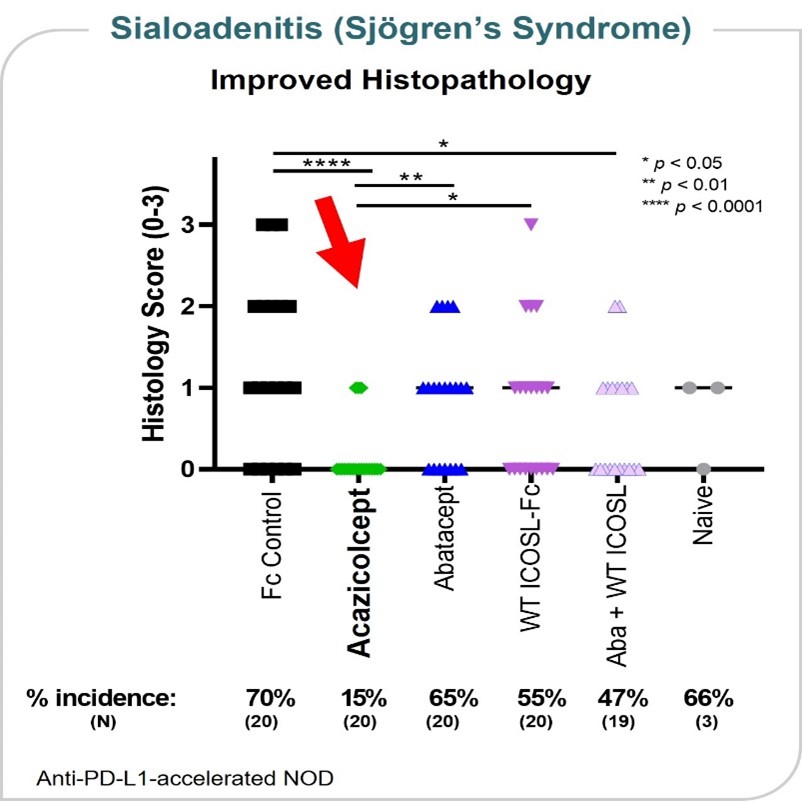
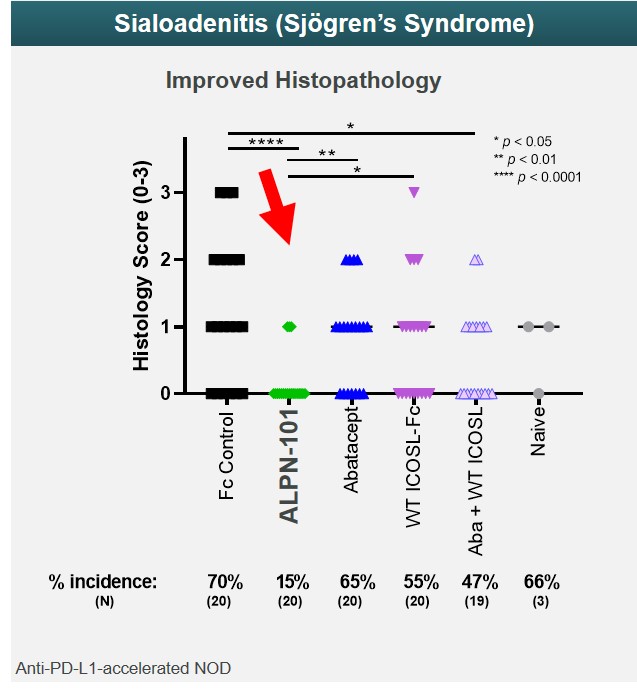
Sjögren’s Syndrome Model
Sjögren’s syndrome is an autoimmune disease in which immune cells attack the glands that produce saliva and tears, as well as other internal organs. In an animal model of salivary gland inflammation (sialoadenitis), a key organ manifestation of Sjögren’s syndrome, acazicolcept appeared more efficacious in reducing the incidence and severity of sialadenitis as compared to abatacept or wild-type ICOSL-Fc alone or in combination. These data were presented at the 2019 annual meeting of the ACR. (Figure 23)
Figure 23
Systemic Lupus Erythematosus Model
Acazicolcept has demonstratedefficacyin a preclinical model of SLE, a multiorgan autoimmune disease that that can lead to serious organ complications and death. In Figure 24, which evaluated acazicolcept in a bm12 inducible model of SLE, treatment with acazicolcept reduced serum titers of anti-dsDNA autoantibodies throughout the study compared to Fc control treatment. These data were presented at the 2019 annual meeting of the ACR. We evaluated acazicolcept in a Phase 1 healthy volunteer study and in the second quarter of 2021 initiated patient dosing in Synergy, a global, randomized, double-blind, placebo-controlled Phase 2 study of acazicolcept in adults with moderate-to-severe SLE.
Figure 24
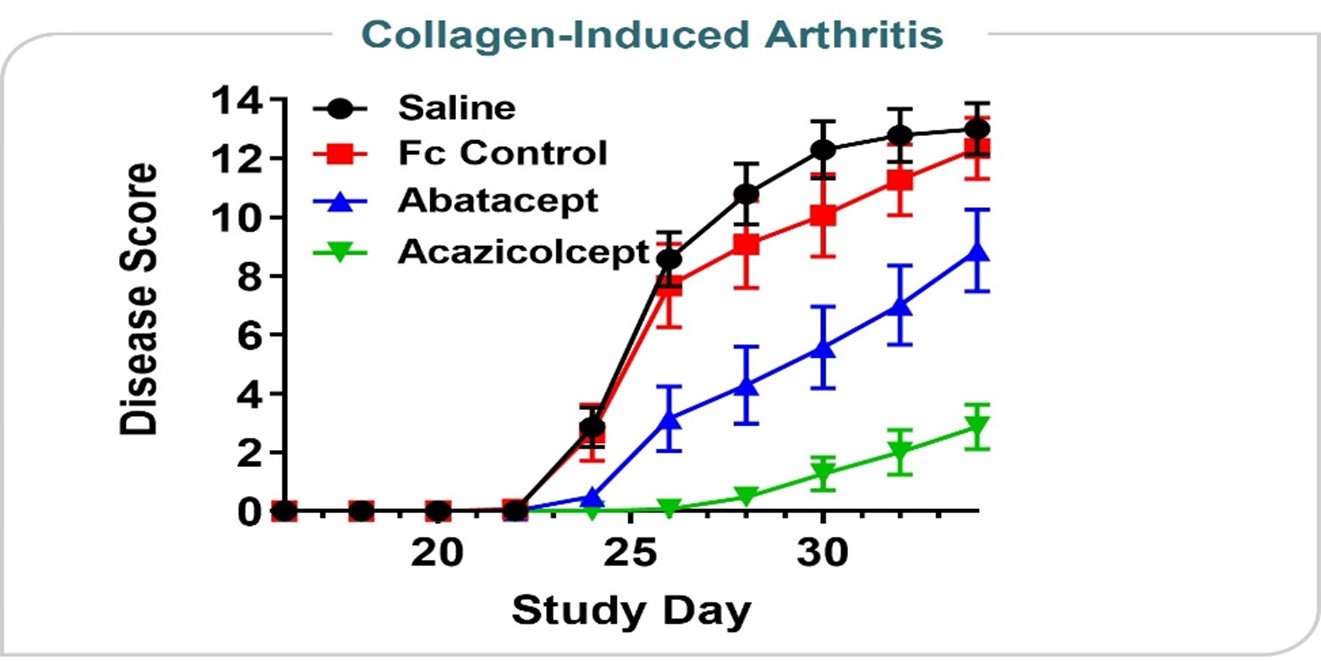
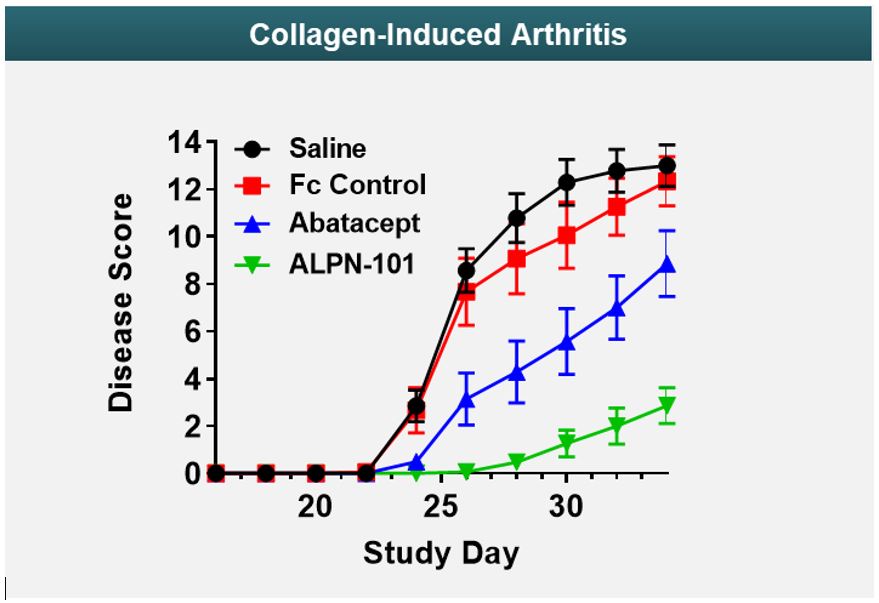
Arthritis Model
Figure 725 shows data from an in vivo collagen-induced arthritis model. This model is designed to test a drug’s ability to reduce inflammatory signals thought to play a role in rheumatoid arthritis, psoriatic arthritis, and other types of inflammatory arthritis conditions. In the data presented at the 2019 annual meeting of the American College of Rheumatology, acazicolcept was superior to abatacept, a drug approved by the FDA to treat rheumatoid, psoriatic, and juvenile idiopathic arthritis.
Figure 25
Human Xenograft GVHD Model
ALPN-101Acazicolcept has been studied in an in vivo mouse model of GVHD, a damaging and potentially fatal inflammatory disease frequently observed following allogeneic stem cell and/or bone marrow transplant treatments for cancer or other serious diseases. The results represented in Figure 826 shows ALPN-101show acazicolcept had superior survival when dosed three times per week for four weeks compared to belatacept (an FDA-approved drug for prevention of renal allograft rejection - a type of inflammation-related rejection process analogous to GVHD. Belatacept is a more potent variant of abatacept, which is FDA-approved for the prevention of GVHD). In fact, 100% of ALPN-101acazicolcept multi-dose treated animals across three different dose levels survived. Animals given only a single dose of ALPN-101acazicolcept performed comparably to animals treated with belatacept dosed 3x/week for four weeks, demonstrating the potency and efficacy of ALPN-101acazicolcept in this disease model.
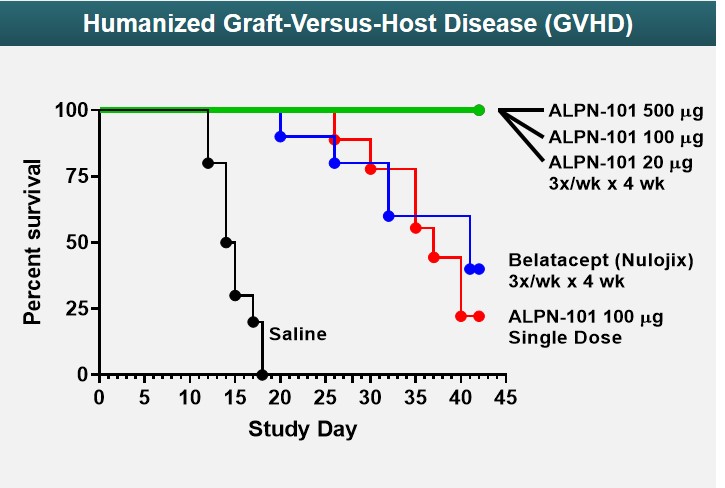
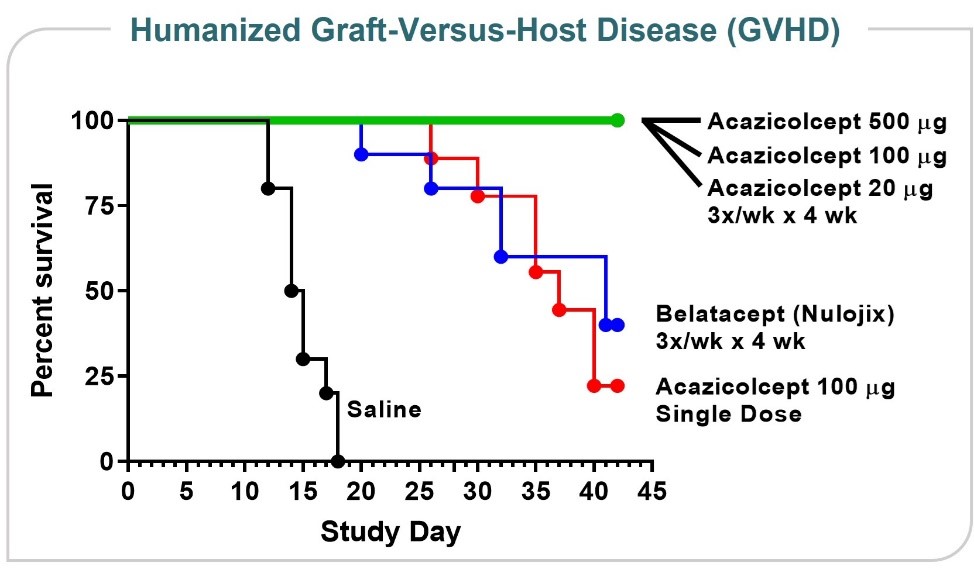
Figure 826
Arthritis Model
Figure 9 shows data from an in vivo collagen-induced arthritis model. This model is designed to test a drug’s ability to reduce inflammatory signals thought to play a role in rheumatoid arthritis, psoriatic arthritis, and other types of inflammatory arthritis conditions. In the data presented at the 2019 annual meeting of the American College of Rheumatology, ALPN-101 was superior to abatacept, a drug approved by the FDA to treat rheumatoid, psoriatic, and juvenile idiopathic arthritis.
Figure 9
Sjögren’s Syndrome Model
Sjögren’s syndrome is an autoimmune disease in which immune cells attack the glands that produce saliva and tears. In an animal model of salivary gland inflammation (sialoadenitis), a key organ manifestation of Sjögren’s syndrome, ALPN-101 appeared more efficacious in reducing the incidence and severity of sialadenitis as compared to abatacept or wild-type ICOSL-Fc alone or in combination. These data were presented at the 2019 annual meeting of the American College of Rheumatology. (Figure 10)
Figure 10
Summary of ALPN-101Acazicolcept Program Preclinical Data
Our scientists and collaborators have demonstrated in in vivo preclinical studies that ALPN-101:acazicolcept:
| |
•
| improves survival compared to belatacept in an in vivo animal GVHD model;
|
| |
•
| reduces disease severity and delays disease onset time relative to control in an in vivo arthritis model with activity superior to abatacept, an FDA-approved drug for rheumatoid, psoriatic, and juvenile idiopathic arthritis;
|
| |
•
| demonstrates control of colitis in an animal model of inflammatory bowel disease, or IBD;
|
| |
•
| shows improved disease scores in an animal model of multiple sclerosis, or MS, compared to abatacept; and
|
| |
•
| demonstrates a lower incidence and severity of sialadenitis, a model of Sjögren’s syndrome, as compared to abatacept or wild-type ICOSL-Fc alone or in combination.
|
ALPN-101•demonstrates a lower incidence and severity of sialadenitis, a model of Sjögren’s syndrome, as compared to abatacept or wild-type ICOSL-Fc alone or in combination;
•reduces levels of pathogenic anti-dsDNA autoantibodies compared to an Fc control in an animal model of SLE;
•reduces disease severity and delays disease onset time relative to control in an in vivo arthritis model with activity superior to abatacept, an FDA-approved drug for rheumatoid, psoriatic, and juvenile idiopathic arthritis;
•improves survival compared to belatacept in a humanized in vivo mouse GVHD model;
•demonstrates control of colitis in an animal model of inflammatory bowel disease, or IBD; and
•shows improved disease scores in an animal model of multiple sclerosis, or MS, compared to abatacept.
Acazicolcept Clinical Development
We recentlyhave completed a Phase 1 study of ALPN-101acazicolcept in healthy volunteers (NCT03748836). This study was designed to evaluate the safety and tolerability of single and multiple ascending intravenous and/or subcutaneous doses of ALPN-101.acazicolcept. In addition, pharmacokinetics, pharmacodynamics and exploratory biomarkers were evaluated to help determine ALPN-101’sacazicolcept’s potential for the treatment of autoimmune and inflammatory diseases. Results from the single ascending dose, or SAD, portion of the study were discussed at an oral presentationpresented at the 2019 American Society2020 European Alliance of Hematology Annual Meeting. In summary, ALPN-101Associations for Rheumatology (EULAR) E-Congress and published in the peer-reviewed journal Clinical Translational Science (doi:10.1111/cts.12983). The first-in-human study randomized 96 healthy adults to receive single or multiple, intravenous or subcutaneous, placebo or acazicolcept at doses ranging from 1 μg/kg to 10 mg/kg. At all dose levels, acazicolcept was generally well-tolerated, withoutwith no severe adverse events, clinically-significant immunogenicity events, or evidence of cytokine storm or release,release. Pharmacokinetics and without clinical signs or symptoms of immunogenicity. In addition, pharmacokinetic and pharmacodynamic datapharmacodynamics (Figure 1127) support developmentexhibited desirable dose dependence, with increasing doses corresponding to increasing duration of ALPN-101 in acute GVHD and additional indications, such as rheumatoid arthritis, systemic lupus erythematosus,complete, or SLE, and Sjögren’s syndrome. We anticipate additional results from the ALPN-101 Phase 1 healthy volunteer trial will be presented at an upcoming scientific conference, investor meeting, or other forum in 2020.
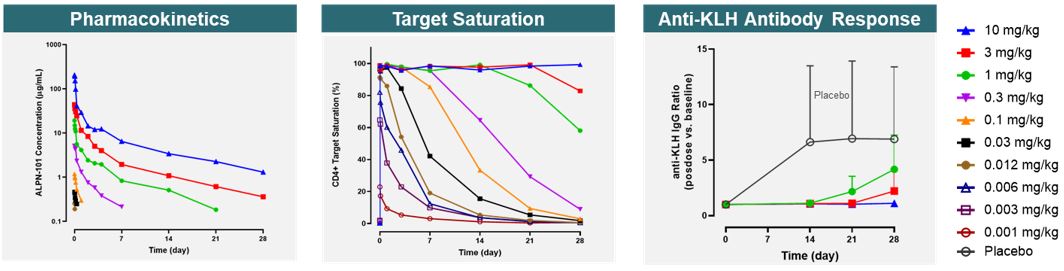
Figure 11
Graft versus host disease
We have opened BALANCE, a Phase 1b/2, open-label, dose escalation and expansion trial of ALPN-101 in patients with steroid-resistant or steroid-refractory active acute GVHD. GVHD is the most common life-threatening complication of allogeneic transplantation of hematopoietic cells and occurs when T cells in the donated tissue (graft) recognize the recipient (host) as foreign, resulting in donor T cell attack upon recipient cells and tissues. It is estimated by the Center for International Blood & Marrow Transplant Research that there are over 8,500 allogeneic hematopoietic cell transplants in the United States each year. Approximately 30-50% of all patients undergoing transplant develop acute GVHD, which classically begins before 100 days after transplantation. GVHD patients resistant or refractory to typical first-line agents including steroids (approximately 30-50% of acute GVHD patients), have a poor long-term prognosis, with a mortality rate of 75% or higher. Additional therapeutic options therefore remain critically needed. Based on our preclinical data demonstrating robust efficacy in GVHD modelsnear-complete target saturation, as well as literature showing superiorityinhibition of combined blockadeantibody responses to keyhole limpet hemocyanin, or KLH, immunization.
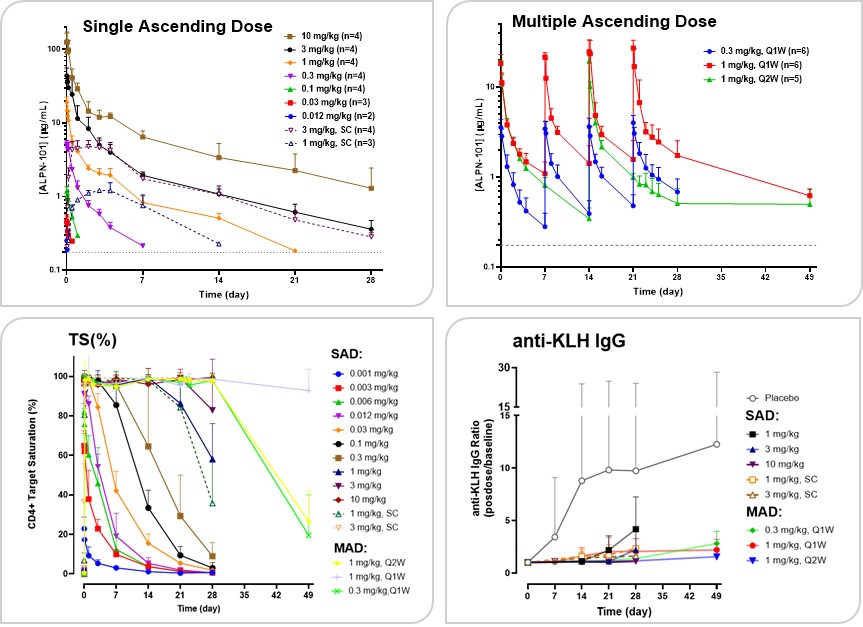
Figure 27
Supported by these results, and as part of CD28 and ICOS compared to isolated blockade of either pathway alone suggests that ALPN-101 has the potential to be beneficial for treating GVHD.
Additional indications
In addition to steroid-resistant or steroid-refractory acute GVHD,AbbVie Agreement, we believe that ALPN-101 has the potential to be effective in inflammatory disease like rheumatoid arthritis, SLE, and Sjögren’s based on:
| |
•
| Clinical experience with therapies targeting the CD28 and/or ICOS pathways;
|
| |
•
| Translational disease tissue expression of CD28 and/or ICOS pathway targets; and
|
| |
•
| Preclinical disease model efficacy (literature and/or ALPN unpublished)
|
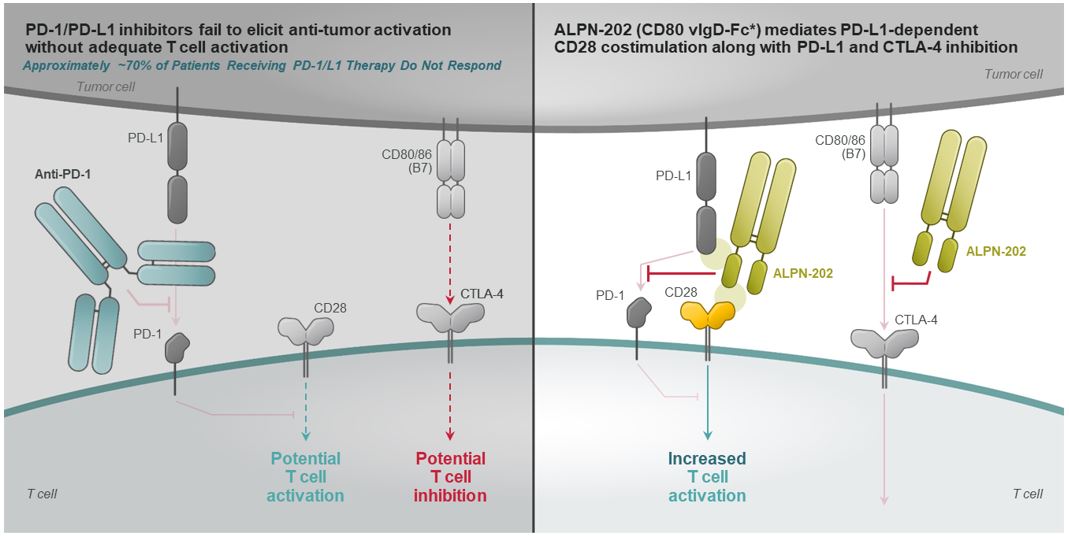
ALPN-202, a Conditional CD28 Costimulator and Dual PD-L1/CTLA-4 Inhibitor for Oncology
Immune checkpoint blockade using inhibitorsplacebo-controlled Phase 2 study of the cytotoxic T-lymphocyte antigen 4, or CTLA-4, or programmed death 1, or PD-1, pathways have been therapeutically successful for a wide variety of malignancies, dramatically altering the treatment paradigmacazicolcept in oncology. However, the majority of patients treated with an inhibitor of CTLA-4, PD-1, or programmed death-ligand 1, or PD-L1, fail to respond or develop resistance, indicating that additional strategies to improve anti-tumor immunity and responses remain needed to improve outcomes. Because immune checkpoints like PD-1 and CTLA-4 appear to suppress anti-tumor immune responsesSLE in part by inhibiting the activating signals mediated by cluster of differentiation 28, or CD28mid-2021 (Figure 12a), next generation immunotherapeutic strategies may substantially improve anti-tumor responses by activating a T cell (co-)stimulatory pathway(s) while also inhibiting a checkpoint pathway(s). Preferably, such activity might be focused primarily within the tumor microenvironment to limit potential systemic immune activation and toxicity.
ALPN-202 is an Fc fusion protein of a modified human cluster of differentiation 80, or CD80, vIgD designed to block the inhibitory immune checkpoints PD-L1 and CTLA-4, and to provide PD-L1-dependent T cell activation via CD28 costimulatory receptor. As illustrated in Figure 12b, ALPN-202 binds PD-L1 expressed on the tumor, blocking PD-L1/PD-1 interactions. Localized to the tumor, ALPN-202 is able to provide a CD28 signal to T cells (PD-L1-dependent CD28 costimulation). Additionally, ALPN-202 binds CTLA-4 expressed on T cells and blocks CTLA4-CD80/CD86 interactions.
Figure 12a Figure 12b
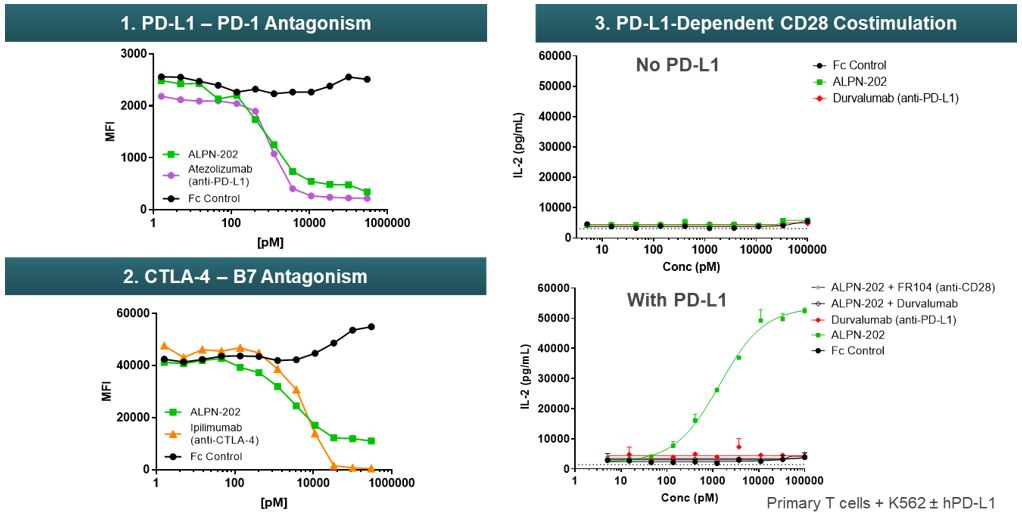
Using in vitro assays, we have characterized and validated the three primary mechanisms of action of ALPN-202: conditional CD28 costimulation and dual PD-L1/CTLA-4 inhibition (Figure 13). Importantly, CD28 costimulation with ALPN-202 requires both T-cell receptor, or TCR, signaling and expression of PD-L1 on the antigen presenting cell, or APC. PD-L1 dependent T-cell costimulation is a unique and key attribute of ALPN-202 which we believe may limit the risk for systemic toxicity while providing potent anti-tumor activity.
Figure 13
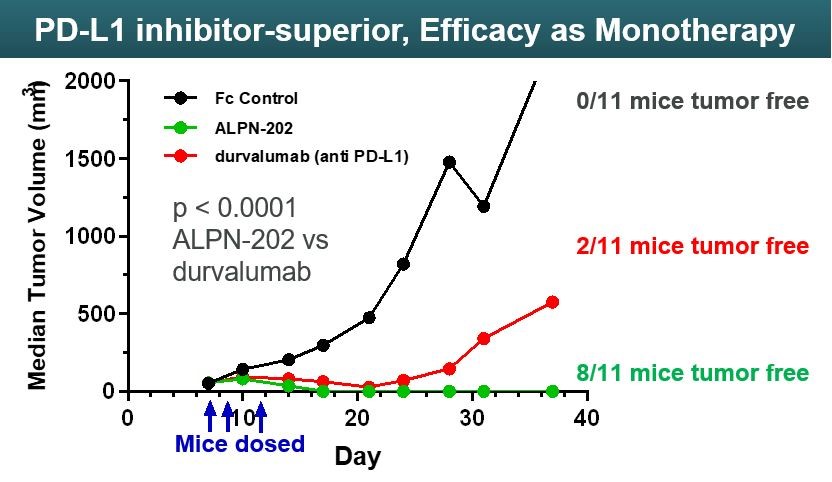
ALPN-202 has also been validated in in vivo tumor models. In a mouse model where mice were implanted with MC38 mouse colon cancer tumors transfected with human PD-L1, ALPN-202 as a monotherapy demonstrated superior tumor control compared to durvalumab, an FDA-approved anti-PD-L1 monoclonal antibody (Figure 1428).
Figure 14 Figure 1528
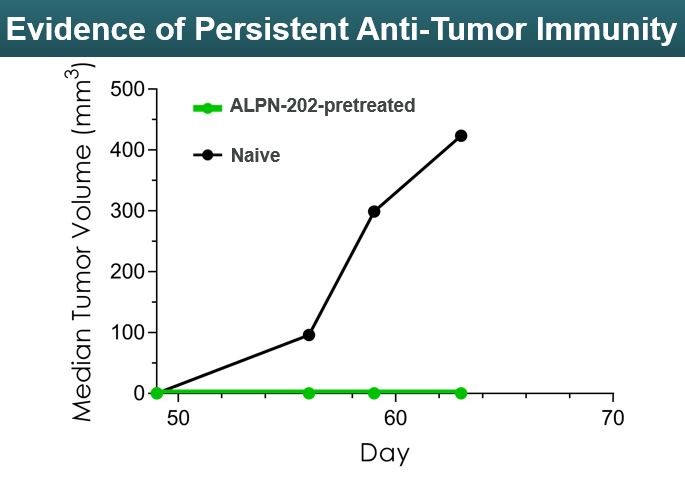
When tumor-free mice in the ALPN-202 arm were re-challenged with additional tumors, they continued to be tumor free despite no additional doses of ALPN-202. (Figure 15)
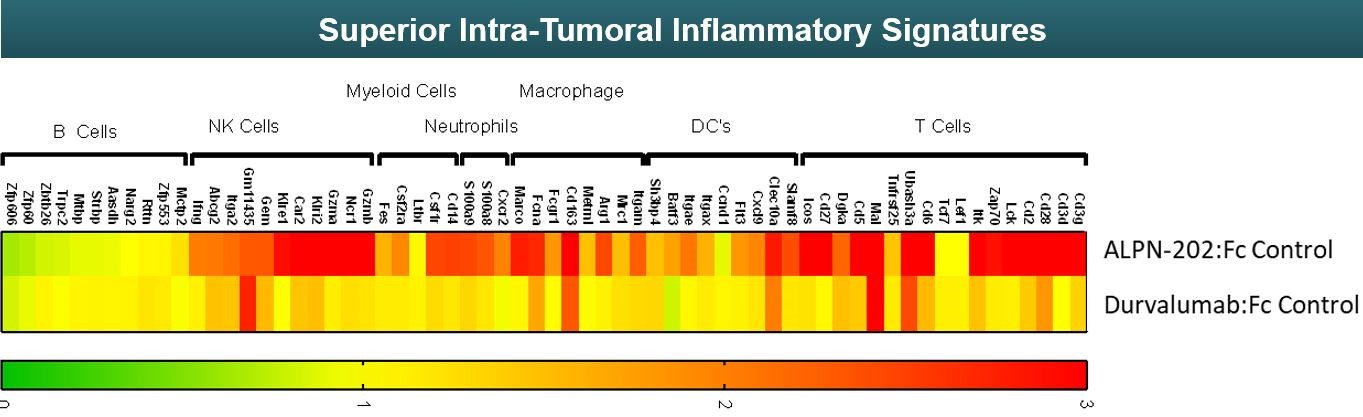
The effects of ALPN-202 or durvalumab on various components of the immune system were compared using a technique called RNA sequencing in the tumor model. This technique generates a display of inflammatory gene signatures where green represents lower or no upregulation of inflammatory genes and red represents higher upregulation of inflammatory genes. For the treatment of cancer, it is thought the more upregulation of the immune system - indicated by higher upregulation of inflammatory genes - potentially results in better outcomes for patients. As seen in Figure 16 below, ALPN-202 upregulates a broader variety of different inflammatory genes connected with several different types of immune cells compared to durvalumab.
Figure 16
Summary of ALPN-202 Preclinical Data
We have demonstrated in preclinical studies that ALPN-202:
| |
•
| exhibits three primary mechanisms of action: conditional CD28 costimulation and dual PD-L1/CTLA-4 inhibition;
|
| |
•
| improves tumor control in a human PD-L1 transduced mouse model of colon cancer compared to the FDA approved anti PD-L1 therapeutic durvalumab;
|
| |
•
| demonstrates a more robust intra-tumor inflammatory signature in the mouse colon cancer model than durvalumab, potentially indicating superior immune system upregulation to fight cancer; and
|
| |
•
| has the potential to be used as a monotherapy or in combination with standard of care chemotherapy or checkpoint only inhibition.
|
ALPN-202 Clinical Development
We have opened NEON-1, our phase 1 study in patients with advanced malignancies and intend to continue to enroll patients throughout 2020 and into 2021. Expansion cohorts in selected tumor types may be informed by a biomarker strategy which is actively under development.
Other Research Programs
In addition to our ALPN-101acazicolcept and ALPN-202povetacicept programs, we have a number of other research efforts underway to address cancerautoimmune and inflammatory diseases that we intend to continue to develop either internally or together with a partner. We plan
Partnerships
In addition to discloseadvancing programs internally, we continue to seek partners who can bring therapeutic area experience, development expertise, commercialization capabilities, and funding to maximize the potential of our existing programs and scientific platform.
Collaboration with Horizon (December 2021)
In December 2021, we entered into the Horizon Agreement, pursuant to which we granted to Horizon rights to our Existing Program and agreed to collaborate with Horizon in the discovery, research and preclinical data on our ALPN-303 programdevelopment of engineered B cell modulation for autoimmune disease at a scientific meeting in 2020. up to three Research Programs, and products arising from the Existing Program and the Research Programs, or the Agreement Products.
Existing Program
We granted to Horizon an exclusive, worldwide, royalty-bearing, sublicensable license under our intellectual property rights for the development, manufacture and commercialization of Agreement Products associated with the Existing Program, or the Existing Program Products, for all applications; however, Horizon has agreed not to pursue the Existing Program Products for oncology. In addition, we granted to Horizon a non-exclusive license to access our libraries of proteins and molecules for research, discovery and identification of additional compounds meeting the agreed criteria (subject to certain exceptions, including compounds reserved by us from the libraries, as set forth in the Horizon Agreement) which would be included in Horizon’s license.
We will conduct our activities under the Existing Program and deliver compounds meeting the agreed criteria pursuant to one or more deliverables plans as mutually agreed by the parties. In addition, Horizon will pay us for the costs and expenses of conducting such activities under the deliverables plans.
Research Programs
Under the Horizon Agreement, during a limited research term, we will conduct, together with Horizon, up to three Research Programs for specified biological targets selected by Horizon (subject to certain exceptions, including targets reserved
by us) to discover proteins and molecules with specified attributes and biological functions, pursuant to a mutually agreed research plan for each such Research Program. Each party has the right to reserve certain targets and to replace such reserved targets or reserve more targets (up to a fixed number) through a gatekeeper mechanism. We will deliver certain compounds meeting the agreed criteria for each Research Program as mutually agreed upon by the parties. Horizon will pay us for the costs and expenses for conducting its activities under the Research Programs. As agreed between the parties, Horizon will own the inventions and intellectual property developed from the Research Programs or otherwise associated with the Agreement Products arising therefrom.
Governance
The parties will establish a joint research committee composed of an equal number of representatives from each of Alpine and Horizon, which will, among other responsibilities, oversee and govern the Existing Program and the Research Programs and review and approve research plans. The parties will also form a joint patent committee comprised of one or two representatives of each party to discuss strategy and facilitate communication and coordination for prosecution and maintenance of the relevant patents under the Horizon Agreement.
Development, Manufacture, Regulatory and Commercialization.
Horizon will have the sole right and responsibility for development, manufacturing, regulatory activities and commercialization of all the Agreement Products. We will provide Horizon reasonable assistance and cooperation. In addition, we have agreed during the term of the Horizon Agreement not to conduct or enable any third party to conduct the development, manufacture or commercialization of certain competing products as set forth therein.
Financial Terms
In connection with the transaction, Horizon made an upfront payment to us of $25.0 million as well as an equity investment for which they paid $15.0 million, a 25% premium to the 30-day volume-weighted average share price as of December 9, 2021.
In addition to the upfront payment and equity consideration, Horizon has agreed to make milestone payments to us upon our achievement of certain preclinical, clinical and regulatory and commercialization milestones, up to an aggregate amount of $381.0 million per program, or approximately $1.5 billion in total, if all milestones are met.
Horizon has further agreed to pay us royalties based on future net sales of the Agreement Products. For the Existing Program, such royalty percentages range from a mid-single digit percentage to a low double-digit percentage of net sales, with the specific royalty rate depending on the aggregate net sales. For each Research Program, such royalty percentages are in a range of mid-single digit percentages of net sales, with the specific royalty rate depending on the aggregate net sales. Horizon’s obligations to pay royalties with respect to an Agreement Product and country will expire after specific criteria for such Agreement Product in such country including it no longer being covered by valid claims of applicable patent rights in such country, or the Royalty Term. Royalty payments are subject to reduction in specified circumstances, including expiration of patent rights, biosimilar competition, or Horizon is required to make payments to a third party with respect to an Agreement Product.
Term and Termination
Unless earlier terminated, the Horizon Agreement remains in effect until the expiration of the Royalty Term for all Agreement Products. The Horizon Agreement is subject to customary termination provisions including termination by a party for the other party’s uncured, material breach. Additionally, Horizon may terminate the Horizon Agreement with specified prior notice in its entirety or on a Program-by-Program basis, for any or no reason.
In the event of certain terminations of the Horizon Agreement, at our request, the parties will negotiate one or more licenses for us to develop, manufacture and commercialize terminated Agreement Products.
The Horizon Agreement includes certain other customary terms and conditions, including mutual representations and warranties, indemnification and confidentiality provisions.
Collaboration with AbbVie (June 2020)
In June 2020, we entered into the AbbVie Agreement, which grants to AbbVie an exclusive option to take an exclusive license to acazicolcept, or the License Option.
Under the terms of the AbbVie Agreement we granted to AbbVie an exclusive option to obtain an exclusive, royalty-bearing, sublicensable license to certain intellectual property rights for the research, development and commercialization of acazicolcept and any other molecule owned or controlled by us that binds to or directly modulates or targets ICOS at certain agreed-upon levels, or the Compounds, on a worldwide basis for all human and non-human diagnostic, prophylactic and therapeutic uses, subject to certain exceptions set forth in the AbbVie Agreement. The License Option is immediately exercisable and will expire 90 days following our delivery of the data package described below to AbbVie, subject to certain exceptions, including clearance under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, or the HSR Act, if required.
Financial Terms
In connection with the execution of the AbbVie Agreement, AbbVie paid us a nonrefundable upfront payment of $60.0 million in cash. If AbbVie exercises its License Option, they will pay a one-time cash payment of $75.0 million.
In addition to the upfront payment and License Option payment, AbbVie has agreed to make cash payments upon our achievement of certain development milestones, the Alpine Development Milestones, prior to the exercise of the License Option as set forth in a written development plan, up to an aggregate amount of $75.0 million. In the second quarter of 2021, we achieved $45.0 million of the Alpine Development Milestones. Following the exercise of the License Option, AbbVie has agreed to make cash payments of up to $205.0 million upon AbbVie’s achievement of certain development and commercial milestones and additional cash payments of up to $450.0 million upon AbbVie’s achievement of certain sales-based cash milestones. AbbVie has further agreed to pay royalties from a high-single digit percentage to a low double-digit percentage of net sales of any pharmaceutical product that contains a Compound, or a Licensed Product, with the specific royalty rate depending on the aggregate net sales. AbbVie’s obligations to pay royalties with respect to a Licensed Product and country will expire upon the latest of the expiration of the last to expire valid patent claim applicable to such Licensed Product in such country, 10 years from the first commercial sale of the Licensed Product in such country, and the expiration of regulatory exclusivity for such Licensed Product in such country. Royalty payments are subject to reduction in specified circumstances, including expiration of patent rights, if average net sales decrease by a certain percentage after the introduction of a generic product, or if AbbVie is required to pay amounts to a third party in order commercialize a Licensed Product in a particular country.
Development Activities
Prior to the exercise of the License Option, we will conduct research and development services, including conducting our Phase 2 study in SLE, based on an agreed-upon development plan, or the Development Plan, which provides for, among other things, the generation of a data package in order for AbbVie to evaluate exercising the License Option and an itemized budget for such activities, including all activities reasonably necessary to conduct a Phase 2 clinical study of acazicolcept for the treatment of SLE; all non-clinical activities; and all CMC activities agreed to under the Development Plan. We will be fully responsible for all costs incurred to conduct our activities, provided that, AbbVie may be responsible for increased costs under the Development Plan in connection with certain material amendments agreed upon with AbbVie.
Prior to the exercise of the License Option, we will be solely responsible, at our sole cost and expense, for preparing, filing and maintaining regulatory documentation, which AbbVie will be entitled to access and review. We will also be responsible for any and all correspondence with the applicable regulatory authorities and for maintaining all data related to acazicolcept. We will be solely responsible, at our sole cost and expense, for manufacturing the compounds necessary to complete the development activities consistent with the Development Plan.
Governance
The parties will establish a joint governance committee, or JGC, composed of an equal number of representatives from each of Alpine and AbbVie, which will, among other responsibilities, coordinate and oversee the development activities, approve amendments to the Development Plan and discuss interactions with regulatory authorities. The chairperson of the JGC will be appointed by AbbVie. AbbVie may disband the JGC, at its sole discretion, following the exercise of the License Option.
Commercialization
Upon AbbVie’s exercise of the License Option, AbbVie and its affiliates will be solely responsible, at AbbVie’s sole cost and expense, for the development, manufacture, commercialization, and regulatory compliance of any Licensed Product. Following exercise of the License Option, AbbVie shall use commercially reasonable efforts to develop and seek regulatory approval for one of the compounds in one indication in each of the United States and one of the United Kingdom, Germany,
France, Spain, or Italy, or the Major Markets, and, following receipt of any such regulatory approval, commercialize the compound in such country.
Changes in Control
We will notify AbbVie immediately upon the closing of any change in control (as defined in the AbbVie Agreement) during the term of the AbbVie Agreement. Following the delivery of such notice, AbbVie may, in its sole and absolute discretion, elect to continue the AbbVie Agreement subject to certain modifications as set forth in the AbbVie Agreement, including the assumption by AbbVie of responsibility to perform certain activities previously assigned to us.
Term and Termination
Unless earlier terminated, the AbbVie Agreement shall terminate either: (i) in the event that the License Option is not exercised by AbbVie, the first day following the last day of the License Option exercise period; or (ii) in the event that the License Option is exercised by AbbVie, the date of the expiration of the last Royalty Term for the last Licensed Product.
Both us and AbbVie may terminate the AbbVie Agreement upon written notice in the event of a material breach by the other party that has not been cured within a 90-day cure period. However, if the uncured material breach is with respect to AbbVie’s obligation to use commercially reasonable efforts to obtain regulatory approval for and commercialize a Licensed Product with respect to any Major Market (but not all Major Markets), then we will only be entitled to terminate the AbbVie Agreement with respect to such Major Market(s). Both AbbVie and us may also terminate the AbbVie Agreement upon written notice if the other party voluntarily or involuntarily files for bankruptcy or insolvency, makes an assignment for the benefit of creditors, has a receiver or trustee appointed over substantially all of such other party’s property, proposes or is party to any dissolution or liquidation, or admits in writing its inability generally to meet such other party’s obligations as they fall due in the general course.
AbbVie may terminate the AbbVie Agreement in its entirety or on a country-by-country basis, for any or no reason, by providing at least 90 days’ prior written notice to us. AbbVie may also terminate the AbbVie Agreement upon notice to us if (i) either we or AbbVie receives a second request for additional information under the HSR Act, provided AbbVie’s notice of termination is delivered within ten business days after AbbVie becomes aware of such request or receives notice from us regarding such request or (ii) the License Option has not been exercised or clearance under the HSR Act, if required, has not occurred within 180 days of submission of the parties’ request for such clearance, provided AbbVie’s notice of termination is delivered within ten business days after the end of such 180-day period.
Upon the termination of the AbbVie Agreement in its entirety for any reason, all licenses and other rights granted (i) to AbbVie by us and (ii) to us by AbbVie shall terminate. Upon termination in certain circumstances, AbbVie has agreed to grant to us licenses to certain intellectual property that is reasonably necessary, and that was actually used by AbbVie for the development, manufacturing or commercialization of the terminated products, to research, develop and commercialize the terminated products in the terminated countries.
In lieu of terminating the AbbVie Agreement in connection with an uncured material breach or the bankruptcy or insolvency of the Company, AbbVie may alternatively elect to continue the AbbVie Agreement subject to certain modifications, including that AbbVie will be entitled to conduct activities allocated to us under the Development Plan, subject to reimbursement by us for AbbVie’s out-of-pocket expenses in connection with such activities. If AbbVie’s right to terminate in connection with an uncured material breach or the bankruptcy or insolvency of the Company arises before exercise of the License Option, then the License Option exercise payment amount will be reduced by half and the amount of any then-unearned milestone payments will be reduced by half. If AbbVie’s right to terminate arises after exercise of the License Option, then the amount of any then-unearned milestone payments will be reduced by 25%.
The AbbVie Agreement includes certain other customary terms and conditions, including mutual representations and warranties, indemnification and confidentiality provisions.
Collaboration with Adaptimmune Therapeutics (May 2019)
In May 2019, we entered into a collaboration and license agreement with Adaptimmune, or the Adaptimmune Agreement, to develop next-generation SPEAR™ T cell products which incorporate our SIP and TIP technology. We and Adaptimmune will collaborate on a specified number of programs to develop SIP and TIP candidates with tailored affinities and modulatory activities that may enhance the anti-tumor responses seen with Adaptimmune’s SPEAR™ T cells. For each program, Adaptimmune has an option to take a worldwide exclusive license for development and commercialization of SPEAR™ T cell products incorporating the developed SIP or TIP candidate for the treatment of cancer. Under the terms of the collaboration agreement, Adaptimmune provided an upfront payment and will provide research funding for ongoing programs.
In addition, we may be eligible for downstream development and commercialization milestones up to $288.0 million, if all pre-specified milestones for each program are achieved. In addition, we are eligible to receive low-single digit royalties on worldwide net sales of the applicable products. In February 2022, Adaptimmune selected an additional research program, triggering a $1.0 million upfront payment, which was recorded as deferred revenue upon receipt and is recognized to revenue based on employee hours contributed.
Manufacturing
We have established in-house non-cGMPnon-current good manufacturing practices, recombinant protein generation capabilities enabling our scientific platform, including validation of new scientific discoveries in vitro and in vivo.vivo. Having protein production capabilities in-house allows more rapid progression for vIgDs and vTDs generated by our scientific platform.
We have chosen U.S.-based contract drug substance and U.S. and Australian drug product manufacturers for our initial current good manufacturing practices, or cGMP, clinical trial supplies of ALPN-101acazicolcept and ALPN-202.povetacicept. We believe these contract manufacturers’manufacturers’ particular expertise in protein production, analytical development and fill/finish provide us with the capability to meet rapid timelines encompassing the development of production cell-lines to manufacturing of clinical trial quantities of the biopharmaceutical product.
We have successfully completed two cGMP manufacturing drug substance campaigns for ALPN-101acazicolcept and onetwo cGMP campaigndrug substance campaigns for ALPN-202povetacicept and believe we have produced sufficient quantities of drug product necessary to execute our stated BALANCESynergy and NEON-1RUBY clinical trials. We have not yet manufactured any of our proteins at commercial scale.
Competition
We participate in the highly competitive sector of biotechnology and pharmaceuticals and in the subsector of immune modulation. This subsector has undergone tremendous technological advancement over the last decade due to advancements in understanding the role of the immune system across multiple therapeutic areas, including oncologyautoimmune and inflammatory disease. While we believe our novel technology platform, discovery programs, knowledge, experience, and scientific resources offer competitive advantages, we face competition from major pharmaceutical and biotechnology companies, academic institutions, governmental agencies, public and private research institutions, and others.
Any products we successfully develop and commercialize will face competition from currently approved therapies and new therapies potentially available in the future.
The availability of reimbursement from government and other third-party payors will also significantly affect the pricing and competitiveness of our products. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for our products, which could result in our competitors establishing a strong market position before we are able to enter the market.
Many of the companies we compete against may have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals, and marketing approved products. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These competitors also compete with us in recruitmentrecruiting and retention ofretaining qualified scientific and management personnel; recruitment of investigativepersonnel and establishing clinical trial sites and participantspatient registration for clinical trials; and the acquisition oftrials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Specifically, our competitors include companies developing therapies with the same target(s) as ALPN-101 in inflammatory diseasesacazicolcept and ALPN-202 in oncologypovetacicept, as well as companies building novel platforms to generate immunomodulatory multi-specific antibody or non-antibody-based targeting proteins.proteins, particularly in the area of autoimmunity.
See the risk factor ““We face competition from entities that have developed or may develop therapeutic candidates for our target disease indications, including companies developing novel treatments and technology platforms based on modalities and technology similar to us. If these companies develop technologies or therapeutic candidates more rapidly than we do, or their technologies, including delivery technologies, are more effective, our ability to develop and successfully commercialize therapeutic candidates may be adversely affected.”” in Part I, Item 1A of this report for more discussion of the effects of competition and competitors on our business.
ALPN-101Acazicolcept Program ICOSL/CD28 Competitors (ICOSL/CD28)
The competitors listed below have programs targeting either ICOS or CD28 (or one of their ligands). for autoimmune and inflammatory diseases. To our knowledge, there are currently no competitors with a single molecule targeting ICOS and CD28 simultaneously.
| |
•
| an anti-BAFF, anti-ICOSL bispecific antibody being developed by Amgen, Inc (rozibafusp alfa (AMG570/MEDI0700));
|
| |
•
| an anti-CD28 monoclonal antibody fragment being developed by OSE ImmunoTherapeutics SA (FR104); and
|
| |
•
| an anti-CD28 peptide being developed by AtoxBio, Inc (reltecimod (AB-103)).
|
ALPN-202 Program Competitors
There are hundreds of clinical trials for immuno-oncology products used as a single agent or in combination. One of the potentially novel attributes of the ALPN-202 program is it combines inhibitory receptor antagonism and activating costimulation with a single molecule interacting with multiple immune targets.
Examples•an anti-CD28 monoclonal antibody fragment being developed by OSE ImmunoTherapeutics SA and Veloxis Pharmaceuticals Inc., a subsidiary of additional multi-target compoundsAsahi Kasei (FR104);
•an anti-CD28 peptide being developed by AtoxBio, Inc. (reltecimod (AB-103));
•an anti-CD28 monoclonal antibody being development by TheraMAB (TAB08); and
•CTLA-4-Fc fusion proteins targeting CD80 and CD86 being marketed Bristol Myers Squibb (abatacept and belatacept).
Povetacicept Program Competitors
The competitors listed below have programs targeting either the TACI, BCMA, or BAFF pathway for immuno-oncology are highlighted below. To our knowledge, there are currently no competitors withautoimmune disease.
•Anti-BAFF antibody marketed by GSK plc (belimumab);
•TACI-Fc being developed by Vera Therapeutics (atacicept);
•TACI-Fc being developed by RemeGen Ltd. (telitacicept (RC18));
•Anti-BAFF-R IgG1 being developed by Novartis AG (Ianalumab (VAY736));
•Anti-APRIL antibody being developed by Visterra, Inc., a single molecule capablesubsidiary of dual PD-L1/CTLA-4 antagonismOtsuka Pharmaceutical Co., Ltd. (sibeprenlimab (VIS649));
•Anti-BAFF, anti-ICOSL bispecific antibody being developed by Amgen Inc. (rozibafusp alfa (AMG570/MEDI0700));
•Anti-APRIL antibody being developed by Chinook Therapeutics, Inc. (BION-1301); and PD-L1 dependent CD28 agonism.
| |
•
| wild-type CD80-Fc being developed by Five Prime Therapeutics, Inc. (FPT155);
|
| |
•
| bispecific antibodies being developed by Regeneron targeting tumor specific antigens and CD28 (REGN5678 anti-PSMAxCD28);
|
| |
•
| trispecific antibodies being developed by Sanofi (CD3xCD38xCD28);
|
| |
•
| bifunctional fusion protein composed of monoclonal antibody against PD-L1 fused to the extracellular domain of human transforming growth factor-ß, or TGF-ß, receptor II being developed by EMD Serono, Inc and GlaxoSmithKline plc (bintrafusp alfa, or M7824);
|
| |
•
| bifunctional fusion protein composed of PD-1 and OX40L developed by Shattuck Labs, Inc. (SL-279252);
|
| |
•
| bispecific fusion protein targeting 4-1BB and PD-L1 being developed by Pieris Pharmaceuticals, Inc. (PRS-344);
|
| |
•
| bispecific monoclonal antibodies being developed by Xencor, Inc. including XmAb20717 targeting CTLA-4 and PD-1, XmAb22841 targeting CTLA-4 and LAG-3, and XmAb23104 targeting PD-1 and ICOS;
|
| |
•
| bispecific constructs called “DARTs” being developed by Macrogenics Inc., including MGD013 targeting PD-1 and LAG-3 and MGD019 targeting PD-1 and CTLA-4;
|
| |
•
| bispecific monoclonal antibody being developed by Tesaro, Inc., which was purchased by GlaxoSmithKline plc, targeting PD-1 and LAG-3;
|
| |
•
| small molecule antagonists being developed by Aurigene Ltd and Curis, Inc., including CA-170 targeting PD-L1 and VISTA and CA-327 targeting PD-L1 and TIM-3;
|
| |
•
| FS118, a bispecific monoclonal antibody targeting PD-L1 and LAG-3 being developed by F-star Biotechnology, Ltd.;
|
| |
•
| various combinations of separate anti PD-1/L1 and anti-CTLA-4 monoclonal antibodies; and
|
| |
•
| various combinations of separate anti PD-1/L1 and costimulatory monoclonal antibodies such as OX-40, 4-1BB, and others.
|
•a recombinant Fc fusion protein designed to block BAFF and APRIL cytokines being developed by Aurinia Pharmaceuticals Inc. (AUR200).
Novel Platform Competitors
Multifunctional therapeutic protein platforms potentially competitive with our platform include:
•Amgen Inc. (BiTE®): fusion proteins consisting of two single-chain variable fragments to link T cells to tumors;
| |
•
| Amgen, Inc. (BiTE®): fusion proteins consisting of two single-chain variable fragments to link T-cells to tumors;
|
| |
•
| Macrogenics Inc. (DART®): Dual-Affinity Re-Targeting and Trident technology platforms bind multiple targets with a single molecule;
|
| |
•
| Xencor Inc. (XmAb Bispecific): Optimized Fc domains for improved potency, half-life and stability;
|
| |
•
| Zymeworks Inc. (Azymetric™): Proprietary amino acid modifications to facilitate interaction of distinct heavy chains;
|
| |
•
| Pieris Pharmaceuticals, Inc. (Anticalin®): Engineered proteins derived from natural lipocalins found in blood plasma;
|
| |
•
| Compass Therapeutics LLC (Targeted Immunomodulation™, StitchMabs™): Antibody discovery targeting the tumor-immune synapse;
|
| |
•
| Harpoon Therapeutics Inc.: TriTac™ (Tri-specific T cell Activating Construct) contain CD3 binding domain, half-life extension domain and antigen-binding domain;
|
| |
•
| Shattuck Labs, Inc.: Agonist Redirected Antibody platform claimed to bind tumor-necrosis factor, or TNF, and checkpoint targets;
|
| |
•
| Ablynx NV (Nanobody®•Macrogenics, Inc. (DART®): Dual-Affinity Re-Targeting and Trident technology platforms bind multiple targets with a single molecule; •Xencor, Inc. (XmAb Bispecific): Optimized Fc domains for improved potency, half-life and stability; •Zymeworks, Inc. (Azymetric™): Proprietary amino acid modifications to facilitate interaction of distinct heavy chains; •Pieris Pharmaceuticals, Inc. (Anticalin®): Engineered proteins derived from natural lipocalins found in blood plasma; •Compass Therapeutics, LLC (Targeted Immunomodulation™, StitchMabs™): Antibody discovery targeting the tumor-immune synapse; •Harpoon Therapeutics, Inc.: TriTAC™ (Tri-specific T cell Activating Construct) contain CD3 binding domain, half-life extension domain, and antigen-binding domain; •Shattuck Labs, Inc.: Agonist Redirected Antibody platform claimed to bind tumor-necrosis factor (“TNF”) and checkpoint targets; •Ablynx NV (Nanobody®), purchased by Sanofi Pharma, Inc.: Platform technology of single-domain, heavy-chain antibody fragments derived from camelidae (e.g., camels and llamas); |
| |
•
| Regeneron, Inc.: VEGF Trap and VelociSuite® antibody technology platforms; and
|
| |
•
| Five Prime Therapeutics, Inc: Proprietary protein library and rapid protein production and testing platform.
|
Intellectual Property•Regeneron, Inc.: VEGF Trap and VelociSuite® antibody technology platforms;
•Five Prime Therapeutics, Inc., purchased by Amgen Inc.: Proprietary protein library and rapid protein production and testing platform; and
•Merus N.V. (Merus Multiclonics® including Biclonics® and Triclonics®): Discovery, screening, and identification platform for bispecific and trispecific antibodies.
Intellectual Property
Our scientific platform and substantially all our intellectual property have been developed internally. As of December 31, 2019,2022, our patent portfolioconsistsof 54 granted patents and over100 200 pendingpatentapplications. Our existing patents and any patents that may issue from the pending applications are expected to expire between 2036 and 2043, not including any patent term extensions that may be afforded under the Federal Food, Drug, and Cosmetic Act, or FFDCA, (and the equivalent provisions in foreign jurisdictions) for any delays incurred during the regulatory approval process relating to human drug products (or processed for making or using human drug products) or any other type of patent term extensions that may be available. Several of our initial patent application isapplications are directed to our scientific platform itself. Our second patent application is directed to the TIP program. We filed subsequentOther patent applications directed toin our SIP program as well asportfolio are directed to various target domains and research programs under development. To date, some of these applications have published but none have yet matured into granted patents. Each of these patent applications is solely owned by us. As we continue the development of our scientific platform and target vIgDs, we intend to continue pursuing intellectual property protection for these technologies.
We have in-licensed some intellectual property and trade secret materials on a non-exclusive basis. To date, such non-exclusive in-licenses are solely related to commercially-available cell lines involved in the manufacture of our vIgD programs. To date, no other intellectual property related to our scientific platform has been in-licensed. We have out-licensed two programs under our TIP/TIP and SIP technology to Adaptimmune Therapeutics on an exclusive basis. Additionally, pursuant to the AbbVie Agreement, we have granted AbbVie an exclusive option to purchase an exclusive worldwide license to acazicolcept. If AbbVie exercises the License Option, AbbVie will take over the future development and commercialization. Finally, pursuant to the Horizon Agreement, we have granted Horizon an exclusive license of certain intellectual property and have entered into a collaboration for the development and commercialization of up to four preclinical candidates generated from Alpine’s unique discovery platform. These candidates include previously undisclosed multi-specific fusion protein-based therapeutic candidates for autoimmune and inflammatory diseases. No other out-licenses have been made.
Although we do not believe our technology infringes any intellectual property rights owned by third parties, we are aware of one or more patents and patent applications that may relate to our technology. Third parties may assert claims against us alleging infringement of their intellectual property rights regardless of whether their allegations have merit. Allegations of infringement could harm our reputation, may result in the expenditure of significant resources to defend and resolve such allegations, and could require us to pay monetary damages if we are found to have infringed any third-party intellectual property rights.
Government Regulation
The FDA and comparable regulatory authorities in state and local jurisdictions and in other countries impose substantial and burdensome requirements on the clinical development, manufacture, marketing, and distribution of therapeutic candidates. These agencies and other federal, state, and local entities regulate research and development activities and the testing, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, approval, advertising and promotion, and export and import of therapeutic candidates and products.
In the U.S., the FDA regulates drugs, medical devices, and biologic products under the Federal Food, Drug,FFDCA, which is the implementing regulation of the FDA, and Cosmetic Act, or FFDCA, its implementing regulations andunder other laws, including, in the case of biologics, the Public Health Service Act.Act, or PHSA. Our potential therapeutic candidates and products will be subject to regulation by the FDA as biologics. Biologics require the submission of a Biologics License Application, or BLA, and approval by the FDA before being marketed in the U.S. None of our therapeutic candidates have been approved by the FDA for marketing in the U.S., and we currently have no BLAs pending. If we fail to comply with applicable FDA or other requirements at any time during the product development process, clinical testing, the approval process, or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include the FDA’sFDA’s refusal to approve pending applications, license suspension or revocation, withdrawal of an approval, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, or criminal prosecution. Any FDA enforcement action could have a material adverse effect on us. The process required by the FDA before biologic therapeutic candidates may be marketed in the U.S. generally involves the following:
| |
•
| completion of extensive preclinical laboratory tests, preclinical animal studies, and formulation studies all performed in accordance with the FDA’s current good laboratory practice, or cGLP, regulations;
|
| |
•
| submission to the FDA of an IND application which must become effective before human clinical trials in the U.S. may begin;
|
| |
•
| performance of adequate and well-controlled human clinical trials to establish the safety and efficacy of the drug candidate for each proposed indication;
|
| |
•
| submission to the FDA of a BLA;
|
| |
•
| satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP regulations; and
|
| |
•
| FDA review and approval of the BLA prior to any commercial marketing, sale, or shipment of the therapeutic product.
|
•submission to the FDA of an Investigational New Drug, or IND, application which must become effective before human clinical trials in the U.S. may begin;
•performance of adequate and well-controlled human clinical trials to establish the safety and efficacy of the drug candidate for each proposed indication;
•submission to the FDA of a BLA;
•satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP regulations; and
•FDA review and approval of the BLA prior to any commercial marketing, sale, or shipment of the therapeutic product.
The testing and approval process requires substantial time, effort, and financial resources, and we cannot be certain any approvals for our therapeutic candidates will be granted on a timely basis, if at all.
Once a therapeutic candidate is identified for development, it enters the preclinical testing stage. Preclinical studies include laboratory evaluations of protein chemistry, formulation, and stability, as well as studies to evaluate toxicity in animals. The results of the preclinical studies, together with manufacturing information and analytical data, are submitted to the FDA as part of an IND application. Currently, the IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day period, raises concerns or questions about the conduct of the clinical trial, including concerns that human research subjects will be exposed to unreasonable health risks. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. Submission of an IND may result in the FDA not allowing the clinical trials to commence or not allowing the clinical trials to commence on the terms originally specified in the IND. A separate submission to an existing IND must also be made for each successive clinical trial conducted during drug development, and the FDA must grant permission, either explicitly or implicitly by not objecting, before each clinical trial can begin. We have completed dosing for only one clinical trial for any of our current therapeutic candidates, the AIS-A01 dosing study of ALPN-101 in healthy volunteers beginning in January 2019.
Clinical trials involve the administration of the therapeutic candidate to human subjects under the supervision of qualified investigators. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be used. Each protocol must be submitted to the FDA as part of the IND. For each medical center proposing to conduct a clinical trial, an institutional review board, or IRB, must also review and approve a plan for any clinical trial before it can begin at that center and the IRB must monitor the clinical trial until it is completed. The FDA, an IRB, or the sponsor may suspend or discontinue a clinical trial at any time on various grounds, including a finding that the subjects are being exposed to an unacceptable health risk. Clinical testing also must satisfy extensive Good Clinical Practice, or GCP, requirements, including the requirements for informed consent.
All clinical research performed in the U.S. in support of a BLA must be authorized in advance by the FDA under the IND regulations and procedures described above. However, a sponsor who wishes to conduct a clinical trial outside the U.S. may, but need not, obtain FDA authorization to conduct the clinical trial under an IND. If a foreign clinical trial is not conducted under an IND, the sponsor may submit data from the clinical trial to the FDA in support of a BLA so long as the clinical trial is conducted in compliance with an international guideline for the ethical conduct of clinical research known as the Declaration of Helsinki and/or the laws and regulations of the country or countries in which the clinical trial is performed, whichever provides the greater protection to the participants in the clinical trial.
Clinical Trials
For purposes of BLA submission and approval, clinical trials are typically conducted in three sequential phases, which may overlap or be combined.
•Phase 1 clinical trials are initially conducted in a limited population of subjects to test the therapeutic candidate for safety, dose tolerance, absorption, metabolism, distribution, and excretion in healthy humans or, on occasion, in patients with severe problems or life-threatening diseases to gain an early indication of its effectiveness.
| |
•
| Phase 1 clinical trials are initially conducted in a limited population of subjects to test the therapeutic candidate for safety, dose tolerance, absorption, metabolism, distribution, and excretion in healthy humans or, on occasion, in patients with severe problems or life-threatening diseases to gain an early indication of its effectiveness.
|
| |
•
| Phase 2 clinical trials are generally conducted in a limited patient population to evaluate preliminary efficacy of the therapeutic candidate for specific targeted indications in patients with the disease or condition under study, evaluate dosage tolerance and appropriate dosage, determine a dosage schedule, and identify possible adverse effects and safety risks.
|
| |
•
| Phase 3 clinical trials are commonly definitive efficacy studies of the experimental medication. Phase 3 trials are typically conducted when Phase 2 clinical trials demonstrate a dose range of the therapeutic candidate is effective and has an acceptable safety profile. Phase 3 clinical trials are generally undertaken with large numbers of patients, such as groups of several hundred to several thousand, to provide substantial evidence of clinical efficacy and to further test for safety in an expanded patient population at multiple, geographically-dispersed clinical trial sites.
|
•Phase 2 clinical trials are generally conducted in a limited patient population to evaluate preliminary efficacy of the therapeutic candidate for specific targeted indications in patients with the disease or condition under study, evaluate dosage tolerance and appropriate dosage, determine a dosage schedule, and identify possible adverse effects and safety risks.
•Phase 3 clinical trials are commonly definitive efficacy studies of the experimental medication. Phase 3 trials are typically conducted when Phase 2 clinical trials demonstrate a dose range of the therapeutic candidate is effective and has an acceptable safety profile. Phase 3 clinical trials are generally undertaken with large numbers of patients, such as groups of several hundred to several thousand, to provide substantial evidence of clinical efficacy and to further test for safety in an expanded patient population at multiple, geographically-dispersed clinical trial sites.
In some cases, the FDA may condition approval of a BLA on the sponsor’ssponsor’s agreement to conduct additional post-approval clinical trials to further assess the biologic’sbiologic’s safety and effectiveness after BLA approval. Such post-approval clinical trials are typically referred to as Phase 4 clinical trials.
Concurrent with clinical trials, companies usually complete additional animal trials and must also develop additional information about the chemistry and physical characteristics of the biologic and finalize a process for manufacturing the
biologic in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the therapeutic candidate and, among other things, the manufacturer must develop methods for testing the identity, strength, quality, and purity of the final biologic product. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate the therapeutic candidate does not undergo unacceptable deterioration over its shelf life.
Further, as a result of the COVID-19 pandemic, we may be required to develop and implement additional clinical trial policies and procedures designed to help protect subjects from the COVID-19 virus. For example, the FDA has issued guidance on conducting clinical trials during the pandemic, which describes a number of considerations for sponsors of clinical trials impacted by the pandemic, including certain reporting requirements, and additional guidance on the good manufacturing practice considerations for responding to COVID-19 infection and other topics. We may be required to make further adjustments to our clinical trials or business operations based on current or future guidance and regulatory requirements as a result of the COVID-19 pandemic.
Biologics License Applications
The results of preclinical studies and of the clinical trials, together with other detailed information, including extensive manufacturing information and information on the chemistry, pharmacology, clinical pharmacology, and the clinical effects of the biologic, are submitted to the FDA in the form of a BLA requesting approval to market the biologic for one or more specified indications. The FDA reviews a BLA to determine, among other things, whether a biologic is safe, pure, and potent and whether the facility in which the biological product is manufactured, processed, packed, or held meets standards designed to assure the biological product continues to be safe, pure, and potent.
Once a BLA has been accepted for filing, by law the FDA will review the application and respond to the applicant, but the review process may be significantly delayed by FDA’sFDA’s requests for additional information or clarification. Under the Prescription Drug User Fee Act, the FDA evaluates a standard original BLA submission within the first 60 days of its receipt to determine if it is sufficiently complete to conduct a full review, and the FDA has a goal of responding to the submission within ten months of the 60-day filing date, but this timeframe is often extended. The FDA may refer the application to an advisory committee for review, evaluation, and/or recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. The FDA may deny approval of a BLA if the applicable statutory and regulatory criteria are not satisfied, or for any reason, or it may require additional clinical data. Even if such data are submitted, the FDA may ultimately decide the BLA does not satisfy the criteria for approval. Data from clinical trials are not always conclusive and the FDA may interpret data differently than we interpret data. Once the FDA approves a BLA, or supplement thereto, the FDA may withdraw the approval if ongoing regulatory requirements are not met or if safety problems are identified after the biologic reaches the market. Where a withdrawal may not be appropriate, the FDA still may seize existing inventory of such biologic or require a recall of any biologic already on the market. In addition, the FDA may require testing, including Phase 4 clinical trials and surveillance programs to monitor the effect of approved biologics which have been commercialized. The FDA has the authority to prevent or limit further marketing of a biologic based on the results of these post-marketing programs.
A sponsor may also seek approval of its therapeutic candidates under programs designed to accelerate FDA review and approval of BLAs. For instance, a sponsor may seek FDA designation of a therapeutic candidate as a “fast“fast track product.”” Fast track products are those products intended for the treatment of a serious or life-threatening disease or condition and which demonstrate the potential to address unmet medical needs for such diseases or conditions. If fast track designation is obtained, the FDA may initiate review of sections of a BLA before the application is complete. This “rolling review”“rolling review” is available if the applicant provides, and the FDA approves, a schedule for the remaining information. In some cases, a fast track product may be approved on the basis of either a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint
that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments, under the FDA’sFDA’s accelerated approval program. Approvals of this kind typically include requirements for appropriate post-approval confirmatory clinical trials to validate the surrogate endpoint or otherwise confirm the effect of the clinical endpoint. On December 29, 2022, the Consolidated Appropriations Act, 2023, including the Food and Drug Omnibus Reform Act (FDORA), was signed into law. FDORA made several changes to the FDA’s authorities and its regulatory framework, including, among other changes, reforms to the accelerated approval pathway, such as requiring the FDA to specify conditions for post-approval study requirements and setting forth procedures for the FDA to withdraw a product on an expedited basis for non-compliance with post-approval requirements.
In addition, the Food and Drug Administration Safety and Innovation Act, or FDASIA, which was enacted and signed into law in 2012, established a new category of drugs referred to as “breakthrough therapies”“breakthrough therapies” that may be subject to accelerated approval. A sponsor may seek FDA designation of a drug candidate as a “breakthrough therapy”“breakthrough therapy” if the drug is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development.
Therapeutic candidates may also be eligible for “priority“priority review,”” or review within a six-month timeframe from the 60-day filing date, if a sponsor provides sufficient clinical data demonstrating its therapeutic candidate provides a significant improvement compared to marketed products. Even if a therapeutic candidate qualifies for one or more of these programs, the FDA may later decide the therapeutic candidate no longer meets the conditions for qualification or that the period for FDA review or approval will be lengthened. When appropriate, we intend to seek fast track designation and/or accelerated approval for our biologics. We cannot predict whether any of our therapeutic candidates will obtain a fast track and/or accelerated approval designation and, if so, whether such designation will be maintained or rescinded by FDA, or the ultimate impact, if
any, of the fast track or the accelerated approval process on the timing or likelihood of FDA approval of any of our proposed biologics.
Biologics may be marketed only for the FDA approvedFDA-approved indications and in accordance with the provisions of the approved labeling. Further, if there are any modifications to the biologic, including changes in indications, labeling, or manufacturing processes, equipment, or facilities, the applicant may be required to submit and obtain FDA approval of a new BLA or BLA supplement, which may require us to develop additional data or conduct additional preclinical studies and clinical trials.
Before approving an application, the FDA will inspect the facility or the facilities at which the biologic product is manufactured and will not approve the product unless cGMP compliance is satisfactory. The FDA may also inspect the sites at which the clinical trials were conducted to assess their compliance and will not approve the biologic unless compliance with Good Clinical PracticeGCP requirements is satisfactory.
The testing and approval processes require substantial time, effort, and financial resources, and each may take several years to complete. The FDA may not grant approval on a timely basis, or at all. Even if we believe a clinical trial has demonstrated safety and efficacy of one of our therapeutic candidates for the treatment of a disease, the results may not be satisfactory to the FDA. Preclinical and clinical data may be interpreted by the FDA in different ways, which could delay, limit, or prevent regulatory approval. We may encounter difficulties or unanticipated costs in our efforts to secure necessary governmental approvals which could delay or preclude us from marketing our therapeutic candidates. The FDA may limit the indications for use or place other conditions on any approvals restricting the commercial application of the products. After approval, certain changes to the approved biologic, such as adding new indications, change in personnel, manufacturing changes, or additional labeling claims, are subject to further FDA review and approval. Depending on the nature of the change proposed, a BLA supplement which may require additional studies to evaluate the effect of such change on the identity, strength, quality, purity, or potency of the product as they may relate to the safety or effectiveness of the product—product—must be filed and approved before the change may be implemented. As with new BLAs, the review process for BLA supplements may be delayed by the FDA through requests for additional information or clarification.
We believe any of our therapeutic products approved as a biological product under a BLA might qualify for a 12-year period of exclusivity currently permitted by the Biologics Price Competition and Innovation Act, or BPCIA. Specifically, the BPCIA established an abbreviated pathway for the approval of biosimilar and interchangeable biological products. The new abbreviated regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable”“interchangeable” based on its similarity to an existing brand product. Under the BPCIA, an application for a biosimilar product cannot be submitted by an applicant until four years after the date the reference product was first licensed and cannot be approved by the FDA until 12 years after the original branded product was first licensed under a BLA. There is a risk the U.S. Congress could amend the BPCIA to significantly shorten this exclusivity period
or the FDA will not consider our therapeutic candidates to be reference products for competing products, potentially creating the opportunity for competition sooner than anticipated. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of our reference products in a way similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing. The BPCIA is complex and is only beginning to be interpreted and implemented by the FDA and the courts. As a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when any such processes may be fully adopted by the FDA, any such processes operating to limit the scope or length of exclusivity afforded by the BPCIA could have a material adverse effect on the future commercial prospects for our biological products. In addition, foreign regulatory authorities may also provide for exclusivity periods for approved biological products or for abbreviated pathways for follow on biological products. For example, biological products in Europe may be eligible for a 10-year period of exclusivity.
Under the Orphan Drug Act, the FDA may grant orphan drug designation to therapeutic candidates intended to treat a rare disease or condition, which is generally a disease or condition affecting fewer than 200,000 individuals in the U.S. or more than 200,000 individuals in the U.S. and for which there is no reasonable expectation the cost of developing and making available in the U.S. a therapeutic candidate for this type of disease or condition will be recovered from sales in the U.S. for that therapeutic candidate. Orphan drug designation must be requested before submitting a marketing application for the therapeutic for that particular rare disease or condition. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The FDA may revoke orphan drug designation, and if it does, it will publicize the drug is no longer designated as an orphan drug. If a therapeutic candidate with orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the therapeutic candidate is entitled to orphan product exclusivity, which means the FDA may not approve any other applications to market the same therapeutic candidate for the same indication, except in very limited circumstances, for seven years. Orphan
drug exclusivity, however, could also block the approval of one of our therapeutic candidates for seven years if a competitor obtains approval of the same therapeutic candidate as defined by the FDA or if our therapeutic candidate is determined to be contained within the competitor’scompetitor’s therapeutic candidate for the same indication or disease.
In Catalyst Pharms., Inc. v. Becerra, 14 F.4th 1299 (11th Cir. 2021), the court disagreed with the FDA’s longstanding position that the orphan drug exclusivity only applies to the approved use or indication within an eligible disease, and not to all uses or indications within the entire disease or condition. In particular, the circuit court held that the orphan-drug exclusivity for Catalyst’s drug blocked FDA’s approval of another drug for all uses or indications within the same orphan-designated disease, or Lambert-Eaton myasthenic syndrome, or LEMS, even though Catalyst’s drug was approved at that time only for use in the treatment of LEMS in adults. Accordingly, the court ordered the FDA to set aside the approval of a drug indicated for LEMS in children. This decision created uncertainty in the application of the orphan drug exclusivity. On January 24, 2023, the FDA published a notice in the Federal Register to clarify that while the agency complies with the court’s order in Catalyst, FDA intends to continue to apply its longstanding interpretation of the regulations to matters outside of the scope of the Catalyst order – that is, the agency will continue tying the scope of orphan-drug exclusivity to the uses or indications for which a drug is approved, which permits other sponsors to obtain approval of a drug for new uses or indications within the same orphan designated disease or condition that have not yet been approved. It is unclear how future litigation, legislation, agency decisions, and administrative actions will impact the scope of the orphan drug exclusivity.
Under the Best Pharmaceuticals for Children Act, certain therapeutic candidates may obtain an additional six months of exclusivity if the sponsor submits information requested in writing by the FDA, referred to as a “Written“Written Request,”” relating to the use of the active moiety of the therapeutic candidate in children. The FDA may not issue a Written Request for studies on unapproved or approved indications where it determines information relating to the use of a therapeutic candidate in a pediatric population, or part of the pediatric population, may not produce health benefits in that population. In addition, the Pediatric Research Equity Act, or PREA, requires a sponsor to conduct pediatric studies for most therapeutic candidates and biologics, for a new active ingredient, new indication, new dosage form, new dosing regimen, or new route of administration. Under PREA, original or New Drug Applications, or NDAs, BLAs and supplements thereto must contain a pediatric assessment unless the sponsor has received a deferral or waiver. The required assessment must assess the safety and effectiveness of the therapeutic candidate for the claimed indications in all relevant pediatric subpopulations and support dosing and administration for each pediatric subpopulation for which the therapeutic candidate is safe and effective. The sponsor or the FDA may request a deferral of pediatric studies for some or all of the pediatric subpopulations. A deferral may be granted for several reasons, including a finding that the drug or biologic is ready for approval for use in adults before pediatric studies are complete or additional safety or effectiveness data needs to be collected before the pediatric studies begin. The FDA must send a noncompliance letter to any sponsor that fails to submit the required assessment, keep a deferral current, or fails to submit a request for approval of a pediatric formulation.
Other Regulatory Requirements
Any biologics manufactured or distributed by us or our collaborators pursuant to FDA approvals would be subject to continuing regulation by the FDA, including recordkeeping requirements and reporting of adverse experiences associated with the product. Manufacturers and their subcontractors are required to register their establishments with the FDA and certain state agencies and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with ongoing regulatory requirements, including cGMPs, which impose certain procedural and documentation requirements upon us and third-party manufacturers. Failure to comply with the statutory and regulatory requirements can subject a manufacturer to possible legal or regulatory action, such as warning letters, suspension of manufacturing, seizure of product, injunctive action or possible civil penalties. Our company cannot be certain it or its present or future third-party manufacturers or suppliers will be able to comply with the cGMP regulations and other ongoing FDA regulatory requirements. If our company or its present or future third-party manufacturers or suppliers are not able to comply with these requirements, the FDA may halt our clinical trials, require us to recall a drug from distribution, or withdraw approval of the BLA for the therapeutic product.
The FDA closely regulates the post-approval marketing and promotion of biologics, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities, and promotional activities involving the Internet. A company can make only those claims relating to safety and efficacy approved by the FDA. Failure to comply with these requirements can result in adverse publicity, warning letters, corrective advertising, and potential civil and criminal penalties. Physicians may prescribe legally available biologics for uses not described in the product’sproduct’s labeling and different from those tested by us and approved by the FDA. Such off-label uses are common across medical specialties. Physicians may believe such off-label uses are the best treatment for many patients in varied circumstances. The FDA does not regulate the behavior of physicians in their choice of treatments. The FDA does, however, impose stringent restrictions on manufacturers’manufacturers’ communications regarding off-label use.
Healthcare Reform
In 2010, Congress passed the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, which we refer to as the ACA, a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of health spending, enhance remedies against fraud and abuse, add new transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry, and impose additional policy reforms. The ACA contains a number of provisions, including those governing enrollment in federal healthcare programs, reimbursement changes, and fraud and abuse, impacting existing government healthcare programs and resulting in the development of new programs, including Medicare payment for performance initiatives, and improvements to the physician quality reporting system and feedback program. The Affordable Care ActACA also does, among other things, the following:
•Increases pharmaceutical manufacturer rebate liability under the Medicaid Drug Rebate Program due to an increase in the minimum basic Medicaid rebate on most branded prescription drugs, and the application of Medicaid rebate liability to drugs used in risk-based Medicaid managed care plans.
| |
••Expands the 340B Drug Pricing Program to require discounts for “covered outpatient drugs” sold to certain children’s hospitals, critical access hospitals, freestanding cancer hospitals, rural referral centers, and sole community hospital. | Increases pharmaceutical manufacturer rebate liability under the Medicaid Drug Rebate Program due to an increase in the minimum basic Medicaid rebate on most branded prescription drugs, and the application of Medicaid rebate liability to drugs used in risk-based Medicaid managed care plans.
|
•Requires pharmaceutical companies to offer discounts on brand-name drugs to patients who fall within the Medicare Part D coverage gap, commonly referred to as the “Donut Hole.”
20•Requires pharmaceutical companies to pay an annual non-tax-deductible fee to the federal government based on each company’s market share of prior year total sales of branded drugs to certain federal healthcare programs, such as Medicare, Medicaid, Department of Veterans Affairs, and Department of Defense.
•Establishes the Center for Medicare and Medicaid Innovation within the Centers for Medicare and Medicaid Services, or CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription drug spending.
| |
•
| Expands the 340B Drug Pricing Program to require discounts for “covered outpatient drugs” sold to certain children’s hospitals, critical access hospitals, freestanding cancer hospitals, rural referral centers, and sole community hospital.
|
| |
•
| Requires pharmaceutical companies to offer discounts on brand-name drugs to patients who fall within the Medicare Part D coverage gap, commonly referred to as the “Donut Hole.”
|
| |
•
| Requires pharmaceutical companies to pay an annual non-tax-deductible fee to the federal government based on each company’s market share of prior year total sales of branded drugs to certain federal healthcare programs, such as Medicare, Medicaid, Department of Veterans Affairs, and Department of Defense.
|
| |
•
| Establishes the Patient-Centered Outcomes Research Institute to identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. The research conducted by the Patient-Centered Outcomes Research Institute may affect the market for certain pharmaceutical products.
|
| |
•
| Establishes The Center for Medicare and Medicaid Innovation within the Centers for Medicare and Medicaid Services, or CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription drug spending.
|
From time to time, legislation is drafted, introduced, and passed in Congress that could significantly change the statutory provisions governing the sale, marketing, coverage, and reimbursement of products regulated by the CMS or other
government agencies. In addition to new legislation, CMS regulations and policies are often revised or interpreted by the agency in ways significantly affecting our business and our products.
In particular, we expect the current U.S. presidential administrationSince its enactment, there have been judicial and Congress will continueCongressional challenges to seek to modify, repeal, or otherwise invalidate all, or certain provisions of, the U.S. healthcare reform legislation. Since taking office, President Trump has continued to support the repeal of all or portions of the ACA. President Trump has also issued an executive order in which he stated it is his administration’s policy to seek the repealaspects of the ACA, and directed executive departmentswe expect there will be additional challenges and federal agenciesamendments to waive, defer, grant exemptions from, or delay the implementationACA in the future. For example, in June 2021, the United States Supreme Court held that Texas and other challengers had no legal standing to challenge the ACA, dismissing the case without specifically ruling on the constitutionality of the provisions ofACA. Accordingly, the ACA toremains in effect in its current form. It is unclear how this Supreme Court decision, future litigation, or healthcare measures promulgated by the maximum extent permitted by law. There is still uncertaintyBiden administration will impact our business, financial condition and results of operations. Complying with respect to the impact the current U.S. presidential administrationany new legislation or changes in healthcare regulation could be time-intensive and Congress may have, if any, and any changes will likely take time to unfold. Such reforms could have anexpensive, resulting in material adverse effect on anticipated revenuesour business.
Additionally, under the American Rescue Plan Act of 2021, effective January 1, 2024, the statutory cap on Medicaid Drug Rebate Program rebates that manufacturers pay to state Medicaid programs will be eliminated. Elimination of this cap may require pharmaceutical manufacturers to pay more in rebates than it receives on the sale of products, which could have a material impact on our business. Additionally, in July 2021, the Biden administration released an executive order, “Promoting Competition in the American Economy,” with multiple provisions aimed at prescription drugs. In response to Biden’s executive order, on September 9, 2021, HHS released a Comprehensive Plan for Addressing High Drug Prices that outlines principles for drug pricing reform and sets out a variety of potential legislative policies that Congress could pursue as well as potential administrative actions HHS can take to advance these principles. In August 2022, Congress passed the Inflation Reduction Act of 2022, which includes prescription drug provisions that have significant implications for the pharmaceutical industry and Medicare beneficiaries, including allowing the federal government to negotiate a maximum fair price for certain high-priced single source Medicare drugs, imposing penalties and excise tax for manufacturers that fail to comply with the drug price negotiation requirements, requiring inflation rebates for all Medicare Part B and Part D drugs, with limited exceptions, if their drug prices increase faster than inflation, and redesigning Medicare Part D to reduce out-of-pocket prescription drug costs for beneficiaries, among other changes. The impact of these legislative, executive, and administrative actions and any future healthcare measures and agency rules implemented by the Biden administration on us and the pharmaceutical industry as a whole is unclear. The implementation of cost containment measures or other healthcare reforms may prevent us from therapeuticbeing able to generate revenue, attain profitability, or commercialize our product candidates if approved. Complying with any new legislation and regulatory changes could be time-intensive and expensive, resulting in a material adverse effect on our business.
Further, many states have proposed or enacted legislation that seeks to indirectly or directly regulate pharmaceutical drug pricing, such as by requiring biopharmaceutical manufacturers to publicly report proprietary pricing information or to place a maximum price ceiling on pharmaceutical products purchased by state agencies. For example, a number of states are considering or have recently enacted state drug price transparency and reporting laws that could substantially increase our compliance burdens and expose us to greater liability under such state laws once we begin commercialization after obtaining regulatory approval for any of our products candidates. We cannot be sure to what extent these and future legislative and regulatory efforts, whether FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals of our product candidates, if any, may be. In addition, increased scrutiny by Congress of the FDA’s approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post-marketing testing and other requirements. These measures could reduce the ultimate demand for our products, once approved, or put pressure on our product pricing. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could affect the prices we may successfully develop andobtain for any of our product candidates for which we may obtain regulatory approval and may affect our overall financial condition and ability to develop therapeutic candidates. However, we cannot predict the ultimate content, timing, or effect of any healthcare reform legislation or the impact of potential legislation on our company.frequency with which any such product candidate, if approved, is prescribed or used.
Furthermore, political, economic, and regulatory influences are subjecting the health care industry in the U.S. to fundamental change. Initiatives to reduce the federal budget and debt and to reform health care coverage are increasing cost-containment efforts. We anticipate federal agencies, Congress, state legislatures, and the private sector will continue to review and assess alternative health care benefits, controls on health care spending, and other fundamental changes to the healthcare delivery system. Any proposed or actual changes could limit coverage for, or the amounts federal and state governments will pay for, health care products and services, which could also result in reduced demand for our products or additional pricing pressures, and limit or eliminate our spending on development projects and affect our ultimate profitability.
Third-Party Payor Coverage and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any products for which we may obtain regulatory approval. In the U.S., sales of any products for which we may receive regulatory approval for commercial sale will depend in part on the availability of coverage and reimbursement from third-party payors. Third-party payors include government authorities such as Medicare, Medicaid, TRICARE, and the Veterans Administration, managed care providers, private health insurers and other organizations.
The Medicaid Drug Rebate Program, which is part of the federal Medicaid program, a program for financially needy patients, among others, requires pharmaceutical manufacturers to enter into and have in effect a national rebate agreement with the Secretary of the Department of Health and Human Services as a condition for states to receive federal matching funds for the manufacturer’smanufacturer’s outpatient drugs furnished to Medicaid patients.
In order for a pharmaceutical product to receive federal reimbursement under Medicare Part B, part of the federal Medicare program covering outpatient items and services for the aged and disabled, and Medicaid programs or to be sold directly to U.S. government agencies, the manufacturer must extend discounts to entities eligible to participate in the 340B drug pricing program, a federal program requiring manufacturers to provide discounts to certain safety-net providers. The required
340B discount on a given product is calculated based upon certain Medicaid Drug Rebate Program metrics reported by the manufacturer.
The process for determining whether a payor will provide coverage for a product is typically separate from the process for setting the reimbursement rate a payor will pay for the product. Third-party payors may limit coverage to specific products on an approved list or formulary which might not include all of the FDA-approved products for a particular indication. Also, third-party payors may refuse to include a particular branded product on their formularies or otherwise restrict patient access to a branded drug when a less costly generic equivalent or other alternative is available. However, under Medicare Part D - Medicare’sMedicare’s outpatient prescription drug benefit - there are protections in place to ensure coverage and reimbursement for oncology products and all Part D prescription drug plans are required to cover substantially all anti-cancer agents. Furthermore, a payor’spayor’s decision to provide coverage for a product does not imply an adequate reimbursement rate will be available. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
Third-party payors are increasingly challenging the price and examining the medical necessity and cost-effectiveness of medical products and services, in addition to their safety and efficacy. In order to obtain coverage and reimbursement for any product approved for sale, we may need to pursue compendia listings or conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of any products, in addition to the costs required to obtain regulatory approvals. Our drug candidates may not be considered medically necessary or cost-effective. If third-party payors do not consider a product to be cost-effective compared to other available therapies, they may not cover an approved product as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow a company to sell its products at a profit.
Other Healthcare Laws and Regulations
In the United States, the research, manufacturing, distribution, sale and promotion of drug products are subject to regulation by various federal, state and local authorities in addition to the FDA, including the Centers for Medicare & Medicaid Services, other divisions of the U.S. Department of Health and Human Services (e.g., the Office of Inspector General), the U.S. Department of Justice, state attorneys general, and other state and local government agencies.
If we obtain regulatory approval of our products, we may be subject to various federal and state laws targeting fraud and abuse in the healthcare industry. These laws may impact, among other things, our proposed sales and marketing strategies. In addition, we may be subject to patient privacy regulation by both the federal government and the states in which we conduct our business. The laws affecting our ability to operate include, but are not limited to:
•the federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, receiving, offering, or paying remuneration (a term interpreted broadly to include anything of value, including, for example, gifts, discounts, and credits), directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order, or recommendation of, an item or service reimbursable under a federal health care program, such as the Medicare and Medicaid programs;
| |
••federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment to Medicare, Medicaid, or other third-party payors that are false or fraudulent, or making a false statement or record material to payment of a false claim or avoiding, decreasing, or concealing an obligation to pay money owed to the federal government; • | the federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, receiving, offering, or paying remuneration (a term interpreted broadly to include anything of value, including, for example, gifts, discounts, and credits), directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order, or recommendation of, an item or service reimbursable under a federal health care program, such as the Medicare and Medicaid programs;
|
| |
•
| federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment to Medicare, Medicaid, or other third-party payors that are false or fraudulent, or making a false statement or record material to payment of a false claim or avoiding, decreasing, or concealing an obligation to pay money owed to the federal government;
|
| |
•
| provisions of HIPAA, prohibiting knowingly and willfully executing a scheme to defraud any health care benefit program and making false statements relating to health care matters; |
| |
•
| provisions of HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act and its implementing regulations, which imposes certain requirements relating to the privacy, security, and transmission of individually identifiable health information;
|
| |
•
| the federal transparency laws, including the federal Physician Payment Sunshine Act, which was part of the Affordable Care Act, requiring manufacturers of certain drugs and biologics to track and disclose payments and other transfers of value they make to U.S. physicians and teaching hospitals, as well as physician ownership and investment interests in the manufacturer, which information is subsequently made publicly available in a searchable format on a CMS website; and
|
| |
•
| state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers, state transparency reporting and compliance laws, and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
|
•the federal transparency laws, including the federal Physician Payment Sunshine Act, which was part of the ACA, requiring applicable manufacturers of certain drugs and biologics, among other covered medical products for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with certain exceptions, to track and report annually certain payments and other transfers of value they make to covered recipients, including U.S. physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors, other health care professionals (such as nurse practitioners and physician assistants, among others) and teaching hospitals, as defined by law, as well as physicians’ and physicians’ immediate family members’ ownership and investment interests in the applicable manufacturer, which are subsequently made publicly available in a searchable format on the CMS Open Payments website; and
•state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers, state transparency reporting and compliance laws, and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
The ACA broadened the reach of the fraud and abuse laws by, among other things, amending the intent requirement of the federal Anti-Kickback Statute and the applicable criminal healthcare fraud statutes contained within 42 U.S.C. §§ 1320a-7b. Pursuant to the statutory amendment, a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation. In addition, the ACA provides the government may assert a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act or the civil monetary penalties statute. Many states have adopted laws similar to the federal Anti-Kickback Statute, some of which apply to the referral of patients for healthcare items or services reimbursed by any source, not only the Medicare and Medicaid programs.
EmployeesIf our operations are found to be in violation of any of the U.S. federal and state laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including criminal and significant civil monetary penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, injunctions, recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals, private qui tam actions brought by individual whistleblowers in the name of the government or refusal to allow us to enter into supply contracts, including government contracts, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations. We may also be subject to additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement with a governmental entity to resolve allegations that we have violated these laws. To the extent that any of our product candidates, once approved, are sold in a foreign country, we may be subject to similar foreign laws and regulations, which may include, for instance, applicable post-approval requirements, including safety surveillance, anti-fraud and abuse laws, and implementation of corporate compliance programs and reporting of payments or transfers of value to healthcare professionals.
Human Capital
Our employees share a passion for meaningful work and are committed to solving the most complex problems in creating immunotherapies to treat autoimmune and inflammatory diseases. Our culture is guided by our core values of innovative thinking, collaboration, flexibility, bias for action, and healthy debate. As of December 31, 2019,2022, we had 53126 employees, of which 41104 are engaged in research and development activities.activities and 22 in general and administrative. None of our employees are represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be good.
Facilities
We leased a facility containing our research and development, laboratory, and office space located at 201 Elliott Avenue West, Seattle, Washington. The lease expired on December 31, 2019. In March 2019, we entered into a lease for 27,164 square feet of office and laboratory space located at 188 East Blaine Street, Seattle, Washington which we have occupied since November 2019. See Part I, Item 2of this report for more details on the terms of the lease. We believe that we provide competitive compensation and benefits for our existing facility is adequate to meet our business requirements for the near-termpersonnel and that additional space will be available on commercially reasonable terms, if required.our compensation and benefit packages are designed to attract and retain highly qualified personnel essential to our business. In addition to salary compensation, our compensation includes new equity grants, a 401(k) retirement plan, healthcare and insurance benefits and a flexible paid time off policy.We are committed to diversity, equity and inclusion. We recruit the best qualified employees regardless of gender, ethnicity or other protected traits and it is our policy to comply with all applicable laws related to discrimination in the workplace.
Corporate Information
On July 24, 2017, Alpine Immune Sciences, Inc., or Private Alpine, completed its business combination with Nivalis Therapeutics, Inc., or Nivalis, a publicly held company. In connection with the merger, Nivalis Therapeutics, Inc. changed its name to Alpine Immune Sciences, Inc. For additional information regarding this business combination, see Note 3 to our consolidated financial statements contained elsewhere in this Annual Report on Form 10-K. Nivalis Therapeutics, Inc. was incorporated in Delaware in March 2007. Alpine Immune Sciences, Inc. (prior to its business combination with Nivalis Therapeutics, Inc.)Nivalis) was incorporated in Delaware on December 30, 2014. Our principal executive office is located at 188 East Blaine Street, Suite 200, Seattle WA, 98102. Our telephone number is (206) 788-4545. Our website is www.alpineimmunesciences.com. Information contained in, or that can be accessed through, our website is not a part of, and is not incorporated into, this report.
This Annual Report on Form 10-K includes our trademarks and registered trademarks, including “vIgD”, “vTD”, “Transmembrane Immunomodulatory Protein” (“TIP”) and “Secreted Immunomodulatory Protein” (“SIP”).
Item 1A. Risk Factors
You should carefully consider the following risk factors, in addition to the other information contained in this Annual Report on Form 10-K, including Management’sManagement’s Discussion and Analysis of Financial Condition and Results of Operations included in Part II, Item 7, and our consolidated financial statements and related notes. If any of the events described in the following risk factors and the risks described elsewhere in this report occurs, our business, operating results and financial condition could be seriously harmed. Our Risk Factors are not guarantees that no such conditions exist as of the date of this report and should not be interpreted as an affirmative statement that such risks or conditions have not materialized, in whole or in part. This report on Form 10-K also contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those anticipated in the forward-looking statements as a result of factors that are described below and elsewhere in this report. Risks Related to Our Financial Position, Capital NeedsPipeline and BusinessProduct Development
We will need to raise substantial additional funds to advance development of our therapeutic candidates, and we cannot guarantee we will have sufficient funds available in the future to develop and commercialize our current or future therapeutic candidates.
We will need to raise substantial additional funds to expand our development, regulatory, manufacturing, marketing, and sales capabilities or contract with other organizations to provide these capabilities to us. We have used substantial funds to develop our therapeutic candidates and will require significant funds to conduct further research and development, preclinical testing, and clinical trials of our therapeutic candidates, to seek regulatory approvals for our therapeutic candidates, and to manufacture and market products, if any are approved for commercial sale. As of December 31, 2019, we had $40.9 million in cash and cash equivalents, restricted cash, and short-term investments. We have developed a plan to implement cost cutting measures to reduce our working capital requirements assuming no additional planned financing. The plan includes a delay in hiring and additional reductions in personnel-related costs and other discretionary expenditures that are within our control and do not effect the anticipated timing of our Phase 1 clinical trials. Based on this plan, we believe our available cash and cash equivalents, will be sufficient to fund our planned level of operations for at least the next 12 months. Our future capital requirements and the period for which we expect our existing resources to support our operations may vary significantly from what we expect. Our monthly spending levels vary based on new and ongoing development and corporate activities. Because the length of time and activities associated with successful development of our therapeutic candidates are highly uncertain, we are unable to estimate the actual funds we will require for development and any approved marketing and commercialization activities. To execute our business plan, we will need, among other things:
| |
•
| to obtain the human and financial resources necessary to develop, test, obtain regulatory approval for, manufacture, and market our therapeutic candidates;
|
| |
•
| to build and maintain a strong intellectual property portfolio and avoid infringing intellectual property of third parties;
|
| |
•
| to establish and maintain successful licenses, collaborations, and alliances;
|
| |
•
| to satisfy the requirements of clinical trial protocols, including patient enrollment;
|
| |
•
| to establish and demonstrate the clinical efficacy and safety of our therapeutic candidates;
|
| |
•
| to obtain regulatory approvals;
|
| |
•
| to manage our spending as costs and expenses increase due to preclinical studies, clinical trials, regulatory approvals, manufacturing scale-up, and commercialization;
|
| |
•
| to obtain additional capital to support and expand our operations; and
|
| |
•
| to market our products to achieveacceptanceand use by themedicalcommunityin general.
|
If we are unable to obtain necessary funding on a timely basis or on acceptable terms, we may have to delay, reduce, or terminate our research and development programs, preclinical studies, or clinical trials, if any, limit strategic opportunities, or undergo reductions in our workforce or other corporate restructuring activities. We also could be required to seek funds through arrangements with collaborators or others requiring us to relinquish rights to some of our technologies or therapeutic candidates we would otherwise pursue on our own. We do not expect to realize revenue from product sales, or royalties in the foreseeable future, if at all. Our revenue sources are, and will remain, extremely limited unless and until our therapeutic candidates are clinically tested, approved for commercialization, and successfully marketed.
To date, we have financed our operations primarily through the sale of equity securities and payments received under our collaboration agreements. We will be required to seek additional funding in the future and intend to do so through a combination of public or private equity offerings, debt financings, credit and loan facilities, research collaborations, and license
agreements. Our ability to raise additional funds from these or other sources will depend on financial, economic, and other factors, many of which are beyond our control. Additional funds may not be available to us on acceptable terms or at all.
If we raise additional funds by issuing equity securities, our stockholders will suffer dilution, and the terms of any financing may adversely affect the rights of our stockholders. For example, in January 2019, we issued in a private placement 4,706,700 shares of common stock and warrants to purchase an additional 1,835,610 shares of common stock for gross proceeds of approximately $25.3 million. We also have an Equity Distribution Agreement in place with Piper Jaffray to sell up to $50.0 million of our common stock, from time to time, through an “at the market” equity offering program under which Piper Jaffray acts as sales agent; however, in July 2019, our Registration Statement on Form S-3 expired pursuant to Rule 415(a)(5), and until a new Registration Statement on Form S-3 is filed and declared effective by the SEC and a prospectus supplement relating to any sales under the Equity Distribution Agreement is filed with the SEC, the equity offering program will not be available to us.
In addition, as a condition to providing additional funds to us, future investors may demand, and may be granted, rights superior to those of existing stockholders. Debt financing, if available, may involve restrictive covenants limiting our flexibility in conducting future business activities, and, in the event of a liquidation or insolvency, debt holders would be repaid before holders of equity securities receive any distribution of corporate assets. Our failure to raise capital or enter into such other arrangements within a reasonable timeframe would have a negative impact on our financial condition, and we may have to delay, reduce, or terminate our research and development programs, preclinical or clinical trials, or undergo reductions in our workforce or other corporate restructuring activities.
We are an early stage biopharmaceutical company with a history of losses, we expect to continue to incur significant losses for the foreseeable future, we may never achieve or maintain profitability, and we have a limited operating history that may make it difficult for investors to evaluate the potential success of our business.
We are a clinical-stage immunotherapy company, with a limited operating history, focused on developing treatments for autoimmune/inflammatory diseases and cancer. Since inception, we have devoted our resources to developing novel protein-based immunotherapies using our proprietary scientific platform technology, which produces variant Ig domains or vIgDs. We have had significant operating losses since inception. For 2019, our net loss was $41.9 million. Substantially all of our losses have resulted from expenses incurred in connection with our research programs and from general and administrative costs associated with our operations. Our technologies and therapeutic candidates are in early stages of development, and we are subject to the risks of failure inherent in the development of therapeutic candidates based on novel technologies.
We have historically generated revenue primarily from the receipt of research funding and upfront payments under our collaboration agreements. We have not generated, and do not expect to generate, any revenue from product sales for the foreseeable future, and we expect to continue to incur significant operating losses for the foreseeable future due to the cost of research and development, preclinical studies, clinical trials, and the regulatory approval process for therapeutic candidates. The amount of future losses is uncertain. Our ability to achieve profitability, if ever, will depend on, among other things, our or our existing collaborators, or any future collaborators, successfully developing therapeutic candidates, obtaining regulatory approvals to market and commercialize therapeutic candidates, manufacturing any approved products on commercially reasonable terms, establishing a sales and marketing organization or suitable third party alternatives for any approved product, and raising sufficient funds to finance business activities. If we or our existing collaborators, or any future collaborators, are unable to develop and commercialize one or more of our therapeutic candidates or if sales revenue from any therapeutic candidate receiving approval is insufficient, we will not achieve profitability, which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
Our approach to the discovery and development of innovative therapeutic treatments based on our technology is unproven and may not result in marketable products.
We plan to develop novel protein-based immunotherapies usingin part via our proprietary vIgD technologydirected evolution platform for the treatment of cancerautoimmune and autoimmune/inflammatory diseases. The potential to create therapies capable of working within and/or modulating an immune synapse, forcing a synapse to occur, or preventing a synapse from occurring is an important, novel attribute of the majority of our vIgDs.approaches. However, the scientific research forming the basis of our efforts to develop therapeutic candidates based on our platform is relatively new. Further, the scientific evidence to support the feasibility of developing therapeutic treatments based on vIgDsour platform is both preliminary and limited.
Relatively few therapeutic candidates based on immunoglobulin superfamily,IgSF domains, or IgSF,tumor necrosis factor receptor super family, or TNFRSF, domains, have been tested in animals or humans, and a number of clinical trials conducted by other companies using IgSF domains technologies have not been successful.humans. We may discover the therapeutic candidates developed using our scientific platform do not possess certain
properties required for the therapeutic candidate to be effective, such as the ability to remain stable or active in the human body for the period of time required for the therapeutic candidate to reach the target tissue and/or cell.effective. We currently have only limited data and no conclusive evidence, to suggest we can introduce these necessary therapeutic properties into vIgD-basedvariant Ig domain, or vIgD or variant TNF(R) domain, or vTD, based therapeutic candidates. We may spend substantial funds attempting to introduce these properties and may never succeed in doing so. In addition, vIgDs or vTDs may demonstrate different chemical and pharmacological properties in human subjects or patients than they do in laboratory studies. Even if our programs have successful results in animal studies, they may not demonstrate the same chemical and pharmacological properties in humans and may interact with human biological systems in unforeseen, ineffective, or harmful ways. For example, in the context of immunotherapies, in a Phase 1 clinical trial of TeGenero AG’s product candidate TGN1412, healthy volunteer subjects receiving the product candidate experienced a systemic inflammatory response resulting in renal and pulmonary failure requiring interventions such as dialysis and critical care support. Following this experience, regulatory agencies now ask for evaluation of immunomodulatory antibodies with a number of in vitro assays with human cells. While we continue to evaluate our vIgDs and vTDs preclinically and clinically, the risk profile in humans is still being fully assessed. Undesirable side effects that may be caused by our therapeutic candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or other comparable foreign authorities. Such side effects could also affect patient recruitment or the ability of enrolled patients to complete clinical trials or result in potential product liability claims. AnyFor example, we recently voluntarily terminated enrollment in both clinical studies involving davoceticept, including the NEON-1 study of davoceticept as monotherapy and the NEON-2 study of davoceticept in combination with pembrolizumab. The decision to terminate enrollment in the davoceticept studies was made following notification of a second Grade 5 serious adverse event (death) in the NEON-2 study. Occurrences like these occurrences may harm our business, financial condition and prospects significantly. As a result, we may never succeed in developing a marketable therapeutic, we may not become profitable, and the value of our common stock willmay decline.
Further, we believe that the FDA has nolittle prior experience with vIgDs and no regulatory authority has granted approval to any person or entity, including our company, to market and commercialize therapeutics using vIgDs,vTDs, which may increase the complexity, uncertainty, and length of the regulatory approval process for our therapeutic candidates. Our company and our current collaborators, or any future collaborators, may never receive approval to market and commercialize any therapeutic candidate. Even if our company or a collaborator obtains regulatory approval, the approval may be for disease indications or patient populations not as broad as we intended or desired or may require labeling, including significant use or distribution restrictions or safety warnings. Our company or a collaborator may be required to perform additional or unanticipated clinical trials to obtain approval or be subject to post-marketing testing requirements to maintain regulatory approval. If therapeutic candidates we develop using our scientific platform prove to be ineffective, unsafe, or commercially unviable, our entire platform and pipeline would have little, if any, value, which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
The market may not be receptive to our therapeutic products based on a novel therapeutic modality, and we may not generate any future revenue from the sale or licensing of therapeutic products.
Even if approval is obtained for a therapeutic candidate, we may not generate or sustain revenue from sales of the therapeutic product due to factors such as whether the therapeutic product can be sold at a competitive price and otherwise accepted in the market. Therefore, any revenue from sales of the therapeutic product may not offset the costs of development. The therapeutic candidates we are developing are based on new technologies and therapeutic approaches. Market participants with significant influence over acceptance of new treatments, such as physicians and third-party payors, may not adopt a treatment based on our therapeutic products, and we may not be able to convince the medical community and third-party payors to accept and use, or to provide favorable coverage or reimbursement for, any therapeutic products developed by our company, our existing collaborator, or any future collaborators. Market acceptance of our therapeutic products will depend on, among other factors:
•the timing of our receipt of any marketing and commercialization approvals;
| |
| the timing of our receipt of any marketing and commercialization approvals;
|
| |
•
| the terms and scope of any approvals and the countries in which approvals are obtained; |
| |
•
| the safety and efficacy of our therapeutic products;
|
| |
•
| the prevalence and severity of any adverse side effects associated with our therapeutic products;
|
| |
•
| the prevalence and severity of any adverse side effects associated with therapeutics of the same type or class as our therapeutic products;
|
| |
•
| limitations or warnings contained in any labeling approved by the FDA or other regulatory authority;
|
| |
•
| relative convenience and ease of administration of our therapeutic products;
|
| |
•
| the willingness of patients to accept any new methods of administration;
|
| |
•
| the success of our physician education programs;
|
| |
•
| the availability of adequate government and third-party payor coverage and reimbursement;
|
•the safety and efficacy of our therapeutic products;
26•the prevalence and severity of any adverse side effects associated with our therapeutic products;
•limitations or warnings contained in any labeling approved by the FDA or other regulatory authority;
| |
••relative convenience and ease of administration of our therapeutic products;
| the pricing of our products, particularly as compared to alternative treatments;
|
| |
•
| our ability to compliantly market and sell our products; and
|
| |
•
| availability of alternative effective treatments for the disease indications our therapeutic products are intended to treat and the relative risks, benefits, and costs of those treatments.
|
•the willingness of patients to accept, and the willingness of physicians to prescribe, any new methods of administration;
•the success of our physician education programs;
•the availability of adequate government and third-party payor coverage and reimbursement;
•the willingness of patients to pay out-of-pocket in the absence of coverage by government and third-party payors;
•the pricing of our products, particularly as compared to alternative treatments;
•our ability to compliantly and effectively market and sell our products;
•the timing of market introduction of our therapeutic products as well as alternative treatments; and
•availability of alternative effective treatments for the disease indications our therapeutic products are intended to treat and the relative risks, benefits, and costs of those treatments.
With our development focus, on engineering wild-type IgSFs proteins, these risks may increase to the extent this field becomes more competitive or less favored in the commercial marketplace. Additional risks apply in relation to any disease indications we pursue which are classified as rare diseases and allow for orphan drug designation by regulatory agencies in major commercial markets, such as the United States, European Union, and Japan. Because of the small patient population for a rare disease, if pricing is not approved or accepted in the market at an appropriate level for an approved therapeutic product with orphan drug designation, such drug may not generate enough revenue to offset costs of development, manufacturing, marketing, and commercialization despite any benefits received from the orphan drug designation, such as market exclusivity, assistance in clinical trial design, or a reduction in user fees or tax credits related to development expense. Market size is also a variable in disease indications classified as rare. Our estimates regarding potential market size for any rare indication may be materially different from what we discover to exist at the time we commence commercialization, if any, for a therapeutic product, which could result in significant changes in our business plan and have a material adverse effect on our business, financial condition, results of operations, and prospects.
If a therapeutic product with orphan drug designation subsequently receives the first FDA approval for the indication for which it has such designation, the therapeutic product is entitled to orphan product exclusivity, which means the FDA may not approve any other applications to market the same therapeutic product for the same indication, except in very limited circumstances, for seven years. Orphan drug exclusivity, however, could also block the approval of one of our therapeutic products for seven years if a competitor obtains approval of the same therapeutic product as defined by the FDA or if our therapeutic product is determined to be within the same class as the competitor’scompetitor’s therapeutic product for the same indication or disease.
In Catalyst Pharms., Inc. v. Becerra, 14 F.4th 1299 (11th Cir. 2021), the court disagreed with the FDA’s longstanding position that the orphan drug exclusivity only applies to the approved use or indication within an eligible disease, and not to all uses or indications within the entire disease or condition. In particular, the circuit court held that the orphan-drug exclusivity for Catalyst’s drug blocked FDA’s approval of another drug for all uses or indications within the same orphan-designated disease, or Lambert-Eaton myasthenic syndrome, or LEMS, even though Catalyst’s drug was approved at that time only for use in the treatment of LEMS in adults. Accordingly, the court ordered the FDA to set aside the approval of a drug indicated for LEMS in children. This decision created uncertainty in the application of the orphan drug exclusivity. On January 24, 2023, the FDA published a notice in the Federal Register to clarify that while the agency complies with the court’s order in Catalyst, FDA intends to continue to apply its longstanding interpretation of the regulations to matters outside of the scope of the Catalyst order – that is, the agency will continue tying the scope of orphan-drug exclusivity to the uses or indications for which a drug is approved, which permits other sponsors to obtain approval of a drug for new uses or indications within the same orphan designated disease or condition that have not yet been approved. It is unclear how future litigation, legislation, agency decisions, and administrative actions will impact the scope of the orphan drug exclusivity.
As in the United States, we may apply for designation of a therapeutic product as an orphan drug for the treatment of a specific indication in the European Union before the application for marketing authorization is made. Sponsors of orphan drugs in the European Union can enjoy economic and marketing benefits, including up to ten years of market exclusivity for the approved indication unless another applicant can show its therapeutic product is safer, more effective, or otherwise clinically superior to the orphan-designated therapeutic product. The respective orphan designation and exclusivity frameworks in the United States and in the European Union are subject to change, and any such changes may affect our ability to obtain EU or U.S. orphan designations in the future.
Our therapeutic candidates are in early stages of development and may fail in development or suffer delays that materially and adversely affect their commercial viability.
We have no products on the market and all of our therapeutic candidates are in early stages of development. Our ability to achieve and sustain profitability depends on obtaining regulatory approval and Institutional Review Board, or IRB approval to conduct clinical trials at particular sites, obtaining regulatory approvals to market our therapeutic candidates and successfully commercializing our therapeutic candidates, either alone or with third parties, such as our collaborators. Before obtaining regulatory approval for the commercial distribution of our therapeutic candidates, we or a collaborator must conduct extensive preclinical tests and clinical trials to demonstrate the safety and efficacy in humans of our therapeutic candidates. Preclinical testing and clinical trials are expensive, difficult to design and implement, can take many years to complete, and are uncertain as to outcome. For example, in October 2022, we announced the termination of enrollment of davoceticept clinical studies (NEON-1 and NEON-2) after we were notified of a second death in the NEON-2 study, which investigated davoceticept in combination with pembrolizumab. While we continue to monitor all NEON study participants previously enrolled in the NEON studies, we are currently focused on advancing the development of both ALPN-101acazicolcept and ALPN-202;povetacicept; however, even with the significant investment of time and funding to advance ALPN-101 and ALPN-202,these product candidates, we cannot guarantee that our clinical and pre-clinicalpreclinical development efforts will be successful or that any of our product candidates will advance to human clinical trials.successful. The start or end of a clinical study is often delayed or halted due to delays in or failure to obtain regulatory approval to commence the study, delays in or failure to reach agreement on acceptable terms with prospective contract research organizations, or CROs, or clinical trial sites, delays in or failure to obtain IRB approval at each site, changing regulatory requirements, manufacturing challenges, clinical sites or contract research organizationsCROs deviating from the trial protocol or failing to comply with regulatory requirements or meet contractual obligations, slower than anticipated patient enrollment, changing standards of care, availability or prevalence of use of a comparative therapeutic or required prior therapy, clinical outcomes, failure of patients to complete the trial or return for post-treatment follow-up, or financial constraints. For instance, delays or difficulties in patient enrollment or difficulties in retaining trial participants can result in increased costs, longer development times, or termination of a clinical trial. Clinical trials of a new therapeutic candidate require the enrollment of a sufficient number of patients, which may include patients who are suffering from the disease the therapeutic candidate is intended to treat and who meet other eligibility criteria. Rates of patient enrollment are affected by many factors, including the size of the patient
population, the eligibility criteria for the clinical trial, the age and condition of the patients, the stage and severity of disease, the nature of the protocol, the proximity of patients to clinical sites, and the availability of effective treatments or competing academic and other clinical trials for the relevant disease.
A therapeutic candidate can unexpectedly fail at any stage of preclinical and clinical development. The historical failure rate for therapeutic candidates is high due to scientific feasibility, safety, efficacy, changing standards of medical care, and other variables. The novelty of our platform may mean our failure rates are higher than historical norms. The results from preclinical testing or early clinical trials of a therapeutic candidate may not predict the outcome of later phase clinical trials of the therapeutic candidate, particularly in immuno-oncologyautoimmune and autoimmune/inflammatory disorders. To date, we have not conducted any clinical trials of our therapeutic candidates in patients with active disease. However, weWe will have to conduct additional trials in our proposed indications to verify the results obtained to date in our preclinical and clinical studies and to support any future regulatory submissions for further clinical development.submissions. A number of companies in the biopharmaceutical industry have suffered significant setbacks in
advanced clinical trials due to lack of efficacy or adverse safety profiles despite promising results in earlier, smaller clinical trials. Moreover, clinical data are often susceptible to varying interpretations and analyses. We do not know whether Phase 1, Phase 2, Phase 3, or other clinical trials we may conduct will demonstrate consistent or adequate efficacy and safety with respect to the proposed indication for use sufficient to receive regulatory approval or market our therapeutic candidates. To the extent that the results of the clinical trials are not satisfactory to the FDA or foreign regulatory authorities for supporting a marketing application, we may be required to expend significant resources, which may not be available to us, to conduct additional clinical trials in support of potential approval of our product candidates.
We, the FDA, an IRB, an independent ethics committee, or other applicable regulatory authorities may suspend clinical trials of a therapeutic candidate at any time for various reasons, including a belief that subjects participating in such trials are being exposed to unacceptable health risks or adverse side effects. Similarly, an IRB or ethics committee may suspend a clinical trial at a particular trial site. We may not have the financial resources to continue development of, or to enter into collaborations for, a therapeutic candidate if we experience any problems or other unforeseen events delaying or preventing clinical development or regulatory approval of, or our ability to commercialize, therapeutic candidates, including:
•negative or inconclusive results from our clinical trials, or the clinical trials of others for therapeutic candidates similar to ours, leading to a decision or requirement to conduct additional preclinical testing or clinical trials or abandon a program;
| |
•
| negative or inconclusive results from our clinical trials, or the clinical trials of others for therapeutic candidates similar to ours, leading to a decision or requirement to conduct additional preclinical testing or clinical trials or abandon a program;
|
| |
•
| serious and unexpected drug-related side effects experienced by participants in our clinical trials or by individuals using therapeutics similar to our therapeutic candidates;
|
| |
•
| serious drug-related side effects experienced in the past by individuals using therapeutics similar to our therapeutic candidates;
|
| |
•
| delays in submitting Investigational New Drug, or IND, applications or clinical trial applications, or comparable foreign applications, or delays or failure in obtaining the necessary approvals from regulators or IRBs to commence a clinical trial, or a suspension or termination of a clinical trial once commenced;
|
| |
•
| conditions imposed by the FDA or comparable foreign authorities, such as the European Medicines Agency, or EMA, regarding the scope or design of our clinical trials;
|
| |
•
| delays in enrolling research subjects in clinical trials;
|
| |
•
| high drop-out rates of research subjects;
|
| |
•
| inadequate supply or quality of therapeutic candidate or therapeutic candidate components, or materials or other supplies necessary for the conduct of our clinical trials, including those owned, manufactured, or provided by companies other than ours;
|
| |
•
| greater than anticipated clinical trial costs, including the cost of any approved drugs used in combination with our therapeutic candidates;
|
| |
•
| poor effectiveness of our therapeutic candidates during clinical trials;
|
| |
•
| unfavorable FDA or other regulatory agency inspection and review of a clinical trial site;
|
| |
•
| failure of our third-party contractors or investigators to comply with regulatory requirements or otherwise meet their contractual obligations in a timely manner, or at all;
|
| |
•
| delays and changes in regulatory requirements, policies, and guidelines, including the imposition of additional regulatory oversight around clinical testing generally or with respect to our technology in particular; or
|
| |
•
| varying interpretations of data by the FDA and similar foreign regulatory agencies.
|
•serious drug-related side effects experienced in the past by individuals using therapeutics similar to our therapeutic candidates;
•delays in submitting IND applications or clinical trial applications, or comparable foreign applications, or delays or failure in obtaining the necessary approvals from regulators or IRBs to commence a clinical trial, or a suspension or termination of a clinical trial once commenced;
•conditions imposed by the FDA or comparable foreign authorities, such as the European Medicines Agency, or EMA, regarding the scope or design of our clinical trials;
•delays in enrolling research subjects in clinical trials;
•high drop-out rates of research subjects;
•inadequate supply or quality of therapeutic candidate or therapeutic candidate components, or materials or other supplies necessary for the conduct of our clinical trials, including those owned, manufactured, or provided by companies other than ours;
•greater than anticipated clinical trial costs, including the cost of any approved drugs used in combination with our therapeutic candidates;
•poor effectiveness of our therapeutic candidates during clinical trials;
•unfavorable FDA or other regulatory agency inspection and review of a clinical trial site;
•failure of our third-party contractors or investigators to comply with regulatory requirements or otherwise meet their contractual obligations in a timely manner, or at all;
•delays and changes in regulatory requirements, policies, and guidelines, including the imposition of additional regulatory oversight around clinical testing generally or with respect to our technology in particular; or
•varying interpretations of data by the FDA and similar foreign regulatory agencies.
Because we have limited financial and operational resources, we must prioritize our research programs and will need to focus our discovery and development on select product candidates and indications. Correctly prioritizing our research and development activities is particularly important for us due to the breadth of potential product candidates and indications that we intend to utilize with our clinical development strategy. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on current and future research and development programs and product candidates for specific indications may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may also relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate.
Product development involves a lengthy and expensive process with an uncertain outcome, and results of earlier pre-clinicalpreclinical and clinical trials may not be predictive of future clinical trial results.
Clinical testing is expensive and generally takes many years to complete, and the outcome is inherently uncertain. Failure can occur at any time during the clinical trial process. The results of pre-clinicalpreclinical trials and early clinical trials of our product candidates may not be predictive of the results of larger, later-stage controlled clinical trials. Product candidates showing promising results in early-stage clinical trials may still suffer significant setbacks in subsequent clinical trials. We have evaluated ALPN-101acazicolcept in a phasePhase 1 healthy volunteer trial but we have conducted no clinical trials to date with ALPN-202 and have not dosed ALPN-101previously initiated a Phase 1b/2 study of acazicolcept in patients with steroid-resistant or steroid-refractory active inflammatory disease.acute graft-versus-host disease, or SR-aGVHD. We terminated this Phase 1b/2 SR-aGVHD study in June 2020. Our Phase 2 study in SLE has materially increased our research and development spending and we expect this increased spend will continue. SLE is a challenging indication and a number of trials conducted by other companies have failed after significant investment of time and funding. We cannot predict whether our efforts in this indication will be successful. If we are unsuccessful, it is unlikely that AbbVie would exercise its option for acazicolcept pursuant to our option and license agreement and, as a result, we would not receive the option payment pursuant to this agreement and we would not be eligible for future milestones and royalties. In addition, we had initiated our Phase 1 studies of davoceticept, but terminated enrollment in these studies in October 2022 following notification of a second Grade 5 serious adverse event (death) in the NEON-2 study. We will have to conduct additional preclinical studies and human trials in our proposed indications of acazicolcept and povetacicept to verify the results obtained to date and to support any regulatory submissions for further clinical development. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety profiles despite promising results in earlier, smaller clinical trials. Moreover, clinical data are often susceptible to varying interpretations and analyses. We do not know whether Phase 1, Phase 2, Phase 3, or other clinical trials we may conduct will demonstrate consistent or adequate efficacy and safety with respect to the proposed indication for use sufficient to receive regulatory approval or market our therapeutic candidates.
Additionally, disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed by necessary government agencies, which would adversely affect our business. For example, over the last several years, the U.S. government has shut down multiple times and certain regulatory agencies, such as the FDA, have had to furlough critical FDA and other government employees. In response to the COVID-19 public health emergency, the FDA has postponed some inspections and continues to conduct “mission-critical” inspections on a case-by-case basis, or, where possible to do so safely, has resumed prioritized domestic inspections, such as pre-approval and surveillance inspections. In 2020 and 2021, a number of companies announced receipt of complete response letters due to the FDA’s inability to complete required inspections for their applications. While the FDA has largely caught up with domestic preapproval inspections, it continues to work through its backlog of foreign inspections, the FDA may not be able to continue its current inspection pace or be unable to complete required inspections during the review period, or the review timelines could be extended, including delays or disruptions due to the COVID-19 pandemic, travel restrictions, and staffing shortages. Regulatory authorities outside the U.S. may adopt similar restrictions or other policy measures in response to the COVID-19 pandemic and may experience delays in their regulatory activities. If a prolonged government shutdown or other disruption occurs, or if global health or other concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities in a timely manner, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
If we encounter delays or difficulties enrolling patients in our clinical trials and/or retention of patients in clinical trials, our clinical development activities could be delayed or otherwise adversely affected.
We may experience difficulties in patient enrollment in our clinical trials for a variety of reasons, including supply chain disruptions, staffing shortages and other business and economic disruptions resulting from geopolitical actions, including war and terrorism, natural disasters, including earthquakes, typhoons, floods and fires, as well as other disruptions resulting from the impact of public health factors, including the COVID-19 pandemic, business disruptions of our strategic partners, third-party manufacturers, suppliers and other third parties upon which we rely. The timely completion of clinical trials in accordance with their protocols depends, among other things, on our ability to enroll a sufficient number of patients who remain in the trial until completion of treatment and adequate follow-up. The enrollment of patients depends on many factors, including:
•Inability to enroll, or delay in enrollment of, patients due to outbreaks and public health crises, such as the COVID-19 global pandemic;
•The patient eligibility criteria defined in the protocol;
•The perceived risks and benefits of the product candidate being studied;
•The size of the patient population required for analysis of the trial’s primary endpoints;
•The proximity of patients to trial sites;
•The design of the trial;
•Our ability to recruit clinical trial investigators with the appropriate competencies and experience;
•Our ability to obtain and maintain patient consent;
•Geopolitical events in countries where we have or seek to have clinical trial sites;
•Reporting of the preliminary results of any of our clinical trials; and
•The risk that patients enrolled in clinical trials will drop out of the trials before completion of treatment and adequate follow-up.
In addition, our clinical trials will compete with other clinical trials for product candidates that are in the same therapeutic areas as our product candidates, and this competition will reduce the number and types of patients available to us because some patients who might have opted to enroll in our trials may instead opt to enroll in a trial being conducted by one of our competitors. Since the number of qualified clinical investigation sites is limited, we expect to conduct some of our clinical trials at the same clinical trial sites that some of our competitors use, which will reduce the number of patients who are available for our clinical trials at such clinical trial sites. Geopolitical events in countries where we have or seek to have clinical trial sites can also negatively impact our ability to enroll patients in our trials. For example, we had intended to open trial sites in Russia for both our ongoing Synergy trial and our planned Phase 2 trial in SLE with povetacicept. Following the start of the Russia-Ukraine conflict, we abandoned our plans to open these sites. Although no sites in Russia had been opened, we had to revise our plans and locate alternative trial sites in order to help achieve targeted enrollment numbers and enrollment rates for these trials. While we have implemented various contingency plans to increase enrollment following the start of the Russia-Ukraine conflict, we cannot be certain that our contingency plans will be successful in replacing these anticipated sites or increasing enrollment rates generally. Any resulting delays in patient enrollment may increase our costs or may affect the timing or outcome of our ongoing and planned clinical trials, which could prevent completion or commencement of these trials and adversely affect our ability to advance the development of our product candidates.
Our product candidates may cause undesirable side effects or have other properties that could halt their clinical development, prevent their regulatory approval, require expansion of the trial size, limit their commercial potential, or result in significant negative consequences.
Clinical trials that involve patients with significant co-morbidities are associated with increased risks as such participants may be particularly susceptible to safety and toxicity risks. In addition, these side effects may not be appropriately recognized or managed by the treating medical staff, as safety and toxicity monitoring may be complicated and difficult to manage, which could result in patient death or other significant issues. Additionally, it can be difficult to determine if the serious adverse or unexpected side effects were caused by the product candidate or another factor, especially in subjects who may suffer from other medical conditions and take other medications. Such risks are increased in clinical trials that allow combinations of therapies.
If serious adverse events or undesirable side effects arise, we could be required to suspend, delay, or halt our clinical trials. For example, in October 2022, we voluntarily terminated enrollment in both clinical studies involving davoceticept following a second Grade 5 serious adverse event (patient death) in the NEON-2 trial. Additionally, serious adverse events or undesirable side effects could cause regulatory authorities to deny approval or require us to limit development of that product candidate to certain uses or subpopulations in which the undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk-benefit perspective. Undesirable side effects could also result in an expansion in the size of our clinical trials, increasing the expected costs and timeline of our clinical trials. Side effects that are observed during the trial, whether treatment related or not, could also affect patient recruitment for future trials or the ability of enrolled patients to complete the trial or result in potential product liability claims.
Further, if serious adverse events or undesirable side effects are identified during development or after approval and are determined to be attributed to any of our product candidates, we may be required to develop Risk Evaluation and Mitigation Strategies, or REMS, to ensure that the benefits of treatment with such product candidate outweigh the risks for each potential patient, which may include, among other things, a communication plan to health care practitioners, patient education, extensive patient monitoring or distribution systems and processes that are highly controlled, restrictive and more costly than what is typical for the industry.
Any of these occurrences may harm our business, financial condition and prospects significantly.
We face competition from entities that have developed or may develop therapeutic candidates for our target disease indications, including companies developing novel treatments and technology platforms based on modalities and technology similar to us.
We participate in the highly competitive sector of biotechnology and pharmaceuticals and in the subsector of immune modulation. This subsector has undergone tremendous technological advancement over the last decade due to advancements in understanding the role of the immune system across multiple therapeutic areas, including autoimmune and inflammatory disease. While we believe our novel technology platform, discovery programs, knowledge, experience, and scientific resources offer competitive advantages, we face competition from major pharmaceutical and biotechnology companies, academic institutions, governmental agencies, public and private research institutions, and others.
Any products we successfully develop and commercialize will face competition from currently approved therapies and new therapies potentially available in the future.
The availability of reimbursement from government and other third-party payors will also significantly affect the pricing and competitiveness of our products. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for our products, which could result in our competitors establishing a strong market position before we are able to enter the market.
Many of the companies we compete against may have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals, and marketing approved products. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs. If these companies develop technologies or therapeutic candidates more rapidly than we do, or their technologies, including delivery technologies, are more effective, our ability to develop and successfully commercialize therapeutic candidates may be adversely affected. For additional information regarding our competitors and the competitive landscape, please refer to the section of this report titled “Business — Competition.”
Many of our competitors have significantly greater financial, technical, manufacturing, marketing, sales, and supply resources or experience than we have. If we successfully obtain approval for any therapeutic candidate, we will face competition based on many different factors, including safety and effectiveness, ease with which our products can be administered and the extent to which patients accept relatively new routes of administration, timing and scope of regulatory approvals, availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage, and patent position of our products. Competing products could present superior treatment alternatives, including by being more effective, safer, less expensive, or marketed and sold more effectively than any products we may develop. Competitive products may make any products we develop obsolete or noncompetitive before we recover the expense of developing and commercializing our therapeutic candidates. Competitors could also recruit our employees, which could negatively impact our ability to execute our business plan.
We face risks related to COVID-19 and other health epidemics and outbreaks, which could significantly disrupt our operations and/or business, including our clinical trials.
The COVID-19 pandemic has had a broad adverse impact on the global economy across many industries and has resulted in significant government measures being implemented to control the spread of the virus, including quarantines, travel restrictions and business shutdowns, as well as significant volatility in global financial markets. Our business could be adversely impacted by the effects of the COVID-19 coronavirus outbreak, or by other epidemics or outbreaks.
For example, we have experienced and may continue to experience disruptions due to COVID-19 that could severely impact our business and clinical trials, including:
•delays or difficulties in enrolling patients in our clinical trials;
•delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff;
•diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals serving as our clinical trial sites and hospital staff supporting the conduct of our clinical trials;
•interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by federal or state governments, employers and others;
•limitations in employee resources that would otherwise be focused on the conduct of our clinical trials, including because of sickness of employees or their families or the desire of employees to avoid contact with large groups of people;
•delays in receiving approval from local regulatory authorities and ethics committees to initiate our planned clinical trials;
•delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials;
•interruption in global shipping that may affect the transport of clinical trial materials, such as investigational drug product used in our clinical trials;
•changes in local regulations as part of a response to the COVID-19 coronavirus outbreak which may require us to change the ways in which our clinical trials are conducted, which may result in unexpected costs, or to discontinue the clinical trials altogether;
•delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations in employee resources or forced furlough of government employees; and
•refusal of the FDA to accept data from clinical trials whose conduct has been affected by the COVID-19 outbreak, such as due to missing data.
Further, we may be required to develop and implement additional clinical trial policies and procedures designed to help protect subjects from the COVID-19 virus or any future health epidemic or outbreak. For example, in March 2020, the FDA issued guidance, which the FDA subsequently updated, on conducting clinical trials during the COVID-19 pandemic, which describes a number of considerations for sponsors of clinical trials impacted by the pandemic. The FDA has also published other COVID-19-related industry guidance, including updates to previous guidance documents, regarding Good Manufacturing Practices, or GMP, remote interactive evaluations of drug manufacturing and bioresearch monitoring facilities, and drug product manufacturing and supply chain inspections, among others.
Additionally, certain of our research and development efforts are also conducted globally. For example, the povetacicept healthy volunteer study includes investigative sites in Australia, and our Synergy trial includes investigative sites in Korea and Poland. A health epidemic or other outbreak, including the current COVID-19 outbreak, may materially and adversely affect our business, financial condition and results of operations. Our supply chain for raw materials, drug substance or drug product is also worldwide and, accordingly, could be subject to disruption. There may be restrictions on the export or shipment of raw materials, drug substance or drug product that could materially delay our business or clinical trials.
The extent to which the COVID-19 coronavirus or any other health epidemic or outbreak may impact our business and clinical trials will depend on future developments, which are highly uncertain and cannot be predicted with confidence.In addition, COVID-19, another pandemic or epidemic, or other infectious diseases could disrupt the global financial markets, reducing our ability to access capital, which could negatively affect our liquidity. If a resurgence of COVID-19, the emergence of another pandemic or epidemic, or the emergence of other infectious diseases were to occur, the volatility of the financial market may be heightened, which could adversely impact the value of our common stock.
We believe our development programs and platform have a particular mechanism of action, but this mechanism of action has not been proven conclusively.
Our scientific platform is novel, and the underlying science is not exhaustively understood nor conclusively proven. In particular, the interaction of vIgDs with the immune synapse, the ability of vIgDs to slow, stop, restart, or accelerate immune responses, and the ability of vIgD domains to interact with multiple counter structures is still largely theoretical. Graphical representations of proposed mechanisms of action of our therapies, the size, actual or relative, of our therapeutics, and how our therapeutics might interface with other cells within the human body, inside the immune synapse, or inside the disease and/or the tumor microenvironment are similarly theoretical and not yet conclusively proven. The lack of a proven mechanism of action may adversely affect our ability to raise sufficient capital, complete preclinical studies, adequately manufacture drug product, obtain regulatory clearance for clinical trials, gain marketing approval, or conclude collaborations, or interfere with our ability to market our product to patients and physicians or achieve reimbursement from payors.
Development of product candidates in combination with other therapies could expose us to additional risks.
Development of any of our product candidates in combination with one or more other therapies that have either been approved or not yet been approved for marketing by the FDA, EMA or comparable foreign regulatory authorities could expose
us to additional risks, as combination therapies may increase the rate of serious or unexpected adverse events, which could result in a clinical hold as well as pre-approval and post-approval restrictions by the FDA or other regulatory authorities on the proposed combination therapy, including narrowing of the indication, warnings, additional safety data collection and monitoring procedures, and REMS, even if the cause of such serious or unexpected adverse events are not directly attributed to our product candidate. Any of these events or restrictions could have a material adverse effect on our business, development of our product candidates, delay our regulatory approval, and decrease the market acceptance and profitability of our product candidate if approved for a combination therapy. For example, as discussed in the risk factor above, our NEON-2 trial evaluating davoceticept in combination with Merck’s pembrolizumab in adults with advanced malignancies was placed on partial clinical hold by the FDA between March 2022 and May 2022, during which time we were unable to enroll any additional participants in the clinical trial, and was later voluntarily terminated in October 2022 due to safety concerns.
We will not be able to market and sell any product candidate in combination with any unapproved therapies that do not ultimately obtain marketing approval. If the FDA, EMA or other comparable foreign regulatory authorities do not approve or revoke their approval of other therapies used in combination therapies, or if safety, efficacy, commercial adoption, manufacturing or supply issues arise with the therapies we choose to evaluate in combination with any of our product candidate, we may be unable to obtain approval of or successfully market any one or all of the product candidates we develop.
Even if any of our product candidates were to receive marketing approval or be commercialized for use in combination with other existing approved therapies, we would continue to be subject to the risks that the FDA, EMA or other comparable foreign regulatory authorities could revoke approval of the other therapy used in combination with any of our product candidates, or safety, efficacy, manufacturing or supply issues could arise with these existing therapies. In addition, it is possible that existing therapies with which our product candidates are approved for use could themselves fall out of favor or be relegated to later lines of treatment. This could result in the need to identify other combination therapies for our product candidates or our own products being removed from the market or being less successful commercially.
Additionally, if the third-party providers of therapies or therapies in development used in combination with our product candidates are unable to produce sufficient quantities for clinical trials or for commercialization of our product candidates, or if the cost of combination therapies is prohibitive, our development and commercialization efforts would be impaired, which would have an adverse effect on our business, financial condition, results of operations and growth prospects.
Any inability to present our data in scientific journals or at scientific conferences could adversely impact our business and stock price.
We may from time to time submit data related to our research and development activities in peer-reviewed scientific publications or apply to present data related to our research and development activities at scientific or other conferences. We have no control over whether these submissions or applications are accepted. Even if accepted for a conference, we have no control over whether presentations at scientific conferences will be accepted for oral presentation, poster presentation, or abstract publication only. Even when accepted for publication, we have no control over the timing of the release of the publication. Rejection by publications, delays in publication, rejection for presentation, or a less-preferred format for a presentation may adversely impact our stock price, ability to raise capital, and business.
Our business may be affected by adverse scientific publications or editorial or discussant opinions.
We may from time to time publish data related to our research and development activities in peer-reviewed scientific publications or present data related to our research and development activities at scientific or other conferences. Editorials or discussants unrelated to us may provide opinions on our presented data unfavorable to us. In addition, scientific publications or presentations may be made which are critical of our science or research or the field of immunotherapy in general. This may adversely affect our ability to raise necessary capital, complete clinical and preclinical studies, adequately manufacture drug product, obtain regulatory clearance for clinical trials, or approval for marketing, or interfere with our ability to market our product to patients and physicians or achieve reimbursement from payors.
Risks Related to Our Relationships with Third Parties
To date, our revenue has been primarily derived from our collaboration agreements, and our success iswill be dependent, in part, on our collaborators’collaborators’ efforts to develop our therapeutic candidates.
Our success is dependent, in part, on our collaborators’collaborators’ efforts to develop our therapeutic candidates and, historically, our revenue has been primarily derived from our agreements with collaborators. For example, in October 2015,June 2020, we entered into an exclusive, worldwide license and research agreement with Kite Pharma, Inc., or Kite, to research, develop, and commercialize engineered autologous T cell therapies incorporating two targets from our technology. In October 2017, Kite was acquired by Gilead Pharma, Inc.the AbbVie Agreement for the development of acazicolcept and, in May 2019, Kite provided notice to us of termination of the research and license agreement, which was effective in June 2019. Also, in May 2019,December 2021, we entered into the Horizon Agreement
pursuant to which we granted to Horizon rights to one of our existing preclinical biologic therapeutic programs and we and Horizon agreed to collaborate in the discovery, research and preclinical development of up to three additional autoimmune and inflammatory disease programs for other designated biological targets.
Pursuant to the terms of the AbbVie Agreement, we received an upfront payment of $60.0 million in cash and are eligible to receive up to $75.0 million in development milestones (of which $45.0 million was achieved in the second quarter of 2021), an additional $75.0 million if AbbVie exercises its option with respect to acazicolcept following our completion of certain development activities, additional development, commercial and sales-based milestones up to an aggregate of 655.0 million and royalties on any future net sales. Through December 31, 2022, we have received $105.0 million in upfront and pre-option exercise development milestones as part of the AbbVie Agreement.
Pursuant to the AbbVie Agreement, we will conduct certain development activities under a collaboration agreement with Adaptimmunedevelopment plan that provides for, among other things, the generation of a data package in order for AbbVie to evaluate exercising its exclusive option, including all activities reasonably necessary to complete our Phase 2 study of acazicolcept in SLE. Even if we successfully complete these activities, AbbVie may not exercise its option, which would make achievement of future milestones and receipt of future royalties unattainable. If AbbVie exercises its option, our realization of additional milestones and royalty payments will depend upon the efforts of AbbVie. If AbbVie fails to develop, next-generation SPEAR T-cell products.obtain regulatory approval for, or ultimately commercialize acazicolcept or if AbbVie terminates the collaboration, our business, financial condition, results of operations, and prospects could be materially and adversely affected. For additional information regarding the AbbVie Agreement, please refer to the section of this report titled “Business — Competition.”
Pursuant to the terms of the Horizon Agreement, we received an upfront payment of $25.0 million as well as an equity investment for which they paid $15.0 million. We are also eligible to receive milestone payments upon our achievement of certain preclinical, clinical and regulatory and commercialization milestones, up to an aggregate amount of $381.0 million per program, or approximately $1.5 billion in total, if all milestones are met, as well as royalties on future product sales. Pursuant to the Horizon Agreement, we will conduct certain research activities under a research program; however, even if we successfully perform our obligations under the Horizon Agreement, there is no certainty that Horizon will continue the development of any of the programs, or, if development is continued, if such programs will ultimately succeed and receive regulatory approval. Horizon will have discretion in determining and directing its efforts and resources for future development activities and, if approval is obtained, commercialization and marketing of the approved drug. As a result, there can be no assurances that we will achieve additional milestones pursuant to the Horizon Agreement. For additional information regarding the Horizon Agreement, please refer to the section of this report titled “Business — Competition.”
Our collaborations may also result in reduced royalty revenues if we are unable to obtain and maintain patent protection, as well as if we are unable to obtain patent term extension, for therapeutic candidates or products developed under our agreements with collaborators. In the event of expiration or invalidation of patents covering a therapeutic candidate or product, for example, our collaborators may be entitled to a significant decrease in royalty revenues owed to us under the agreements. Invalidation of patents and failure to obtain patent term extension for one or more patents in our portfolio may occur as a result of factors beyond our control due to the complex legal and factual questions surrounding pharmaceutical and biotechnology patents. If we are unable to obtain and maintain patent protection, or if we unable to obtain patent term extension for therapeutic candidates or products developed under our agreements with collaborators, our revenue derived from our collaborators may be less than the full amount anticipated, and our business, financial condition, results of operations, and growth prospects could be materially harmed.
Continued advancement of our other product candidates and other development efforts depends, in part, upon the efforts of AbbVie, Horizon, and our other current or future collaborators. If our collaborators do not dedicate sufficient resources to the development of product candidates that are the subject of our agreements, such product candidates may never be successful and we may be ineligible to receive additional milestone payments or royalties pursuant to the terms of our arrangements, which could have a material adverse impact on our financial results and operations. Even if we and our collaborators dedicate sufficient resources to our collaboration agreements, neither we nor our collaborators may be effective in obtaining approvals for any therapeutic candidates or, if approved, the successful commercialization of any approved products. Collaborators may change their strategic focus or pursue alternative technologies after entering into a collaboration agreement with us, which could result in reduced, delayed or no revenue to us. Disputes regarding collaboration agreements, including disputes pertaining to ownership of intellectual property, may also arise and if we and our collaborators are unable to resolve such disputes, litigation proceedings may occur, which could further delay development programs, create uncertainty as to ownership of intellectual property rights, distract management from other business activities and generate substantial expenses, any of which could materially and negatively impact our business.
If third parties on which we depend to conduct our clinical or preclinical studies, or any future clinical trials, do not perform as expected, fail to satisfy regulatory or legal requirements, or miss expected deadlines, our development program could be delayed, which may result in materially adverse effects on our business, financial condition, results of operations, and prospects.
We rely, in part, on third partythird-party clinical investigators, contract research organizations, or CROs, clinical data management organizations, and consultants to design, conduct, supervise, and monitor clinical trials and preclinical studies of our therapeutic candidates and may do the same for future clinical trials. Because we rely on third parties to conduct preclinical studies or clinical trials, we have less control over the timing, quality, compliance, and other aspects of preclinical studies and clinical trials than we would if we conducted all preclinical studies and clinical trials on our own. These investigators, CROs, and consultants are not our employees and we have limited control over the amount of time and resources they dedicate to our programs. These third parties may have contractual relationships with other entities, some of which may be our competitors, which may draw their time and resources away from our programs. The third parties with which we contract might not be
diligent, careful, compliant, or timely in conducting our preclinical studies or clinical trials, resulting in the preclinical studies or clinical trials being delayed or unsuccessful. Further, if any of our relationships with third-party CROs terminate, we may not be able to enter into arrangements with alternative CROs or to do so on commercially reasonable terms.
If we cannot contract with acceptable third parties on commercially reasonable terms, or at all, or if these third parties do not carry out their expected duties, satisfy legal and regulatory requirements for the conduct of preclinical studies or clinical trials, or meet expected deadlines, our clinical development programs could be delayed and otherwise adversely affected. In all events, we are responsible for ensuring each of our preclinical studies and clinical trials is conducted in accordance with the general investigational plan and protocols for the trial and with legal, regulatory and scientific standards. The FDA and certain foreign regulatory authorities, such as the EMA, require preclinical studies to be conducted in accordance with applicable Good Laboratory Practices, or GLPs, and clinical trials to be conducted in accordance with applicable FDA regulations and Good Clinical Practices, or GCPs, including requirements for conducting, recording, and reporting the results of preclinical studies and clinical trials to assure data and reported results are credible and accurate and the rights, integrity, and confidentiality of clinical trial participants are protected. Our reliance on third parties we do not control does not relieve us of these responsibilities and requirements. If we or any of our CROs fail to comply with applicable GCPs, the clinical data generated in our clinical trials may be deemed unreliable and the FDA, the EMA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical trials comply with GCP regulations. In addition, our clinical trials must be conducted with product produced under current good manufacturing practice, or cGMP regulations. Our failure to comply with these regulations may require us to repeat clinical trials, which would delay the regulatory approval process. Any such event could have a material adverse effect on our business, financial condition, results of operations, and prospects.
In addition, switching or adding additional CROs involves additional cost and requires management time and focus. There is also a natural transition period when a new CRO commences work. As a result, delays may occur, which could materially impact our ability to meet our desired clinical development timelines. There can be no assurance that we will not encounter such challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition and prospects.
The COVID-19 coronavirus could adversely impact our business, including our clinical trials.
In December 2019, a novel strain of coronavirus, SARS-CoV-2, the causative agent of coronavirus disease 2019, or COVID-19, was first reported. Since then, SARS-CoV-2 has spread to multiple countries, including the United States and European and Asia-Pacific countries, including countries in which we have planned or active clinical trial sites. As it continues to spread around the globe, we will likely experience disruptions that could severely impact our business and clinical trials, including:
| |
•
| delays or difficulties in enrolling patients in our clinical trials;
|
| |
•
| delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff;
|
| |
•
| diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals serving as our clinical trial sites and hospital staff supporting the conduct of our clinical trials;
|
| |
•
| interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by federal or state governments, employers and others;
|
| |
•
| limitations in employee resources that would otherwise be focused on the conduct of our clinical trials, including because of sickness of employees or their families or the desire of employees to avoid contact with large groups of people;
|
| |
•
| delays in receiving approval from local regulatory authorities and ethics committees to initiate our planned clinical trials;
|
| |
•
| delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials;
|
| |
•
| interruption in global shipping that may affect the transport of clinical trial materials, such as investigational drug product used in our clinical trials;
|
| |
•
| changes in local regulations as part of a response to the COVID-19 coronavirus outbreak which may require us to change the ways in which our clinical trials are conducted, which may result in unexpected costs, or to discontinue the clinical trials altogether;
|
| |
•
| delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations in employee resources or forced furlough of government employees; and
|
| |
•
| refusal of the FDA to accept data from clinical trials whose conduct has been affected by the COVID-19 outbreak, such as due to missing data.
|
The global outbreak of the COVID-19 coronavirus continues to rapidly evolve. The extent to which the COVID-19 coronavirus may impact our business and clinical trials will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the ultimate geographic spread of the disease, the duration of the outbreak, travel restrictions and social distancing in the United States and other countries, business closures or business disruptions and the effectiveness of actions taken in the United States and other countries to contain and treat the disease.
We face risks related to health epidemics and other outbreaks, which could significantly disrupt our operations and/or business.
Our business could be adversely impacted by the effects of the COVID-19 outbreak originating in China, or by other epidemics. Our supply chain for raw materials, drug substance or drug product is worldwide, including China, and accordingly could be subject to disruption. There may be restrictions on the export or shipment of raw materials, drug substance or drug product that could materially delay our business or clinical trials.
Certain of our research and development efforts are also conducted globally, for example the NEON-1 clinical trial includes investigative sites in Australia. A health epidemic or other outbreak, including the current COVID-19 outbreak, may materially and adversely affect our business, financial condition and results of operations. The extent to which the outbreak impacts our results will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of the outbreak and the actions to contain the outbreak or treat its impact, among others.
Because we rely on third partythird-party manufacturing and supply partners, our supply of clinical trial materials may become limited or interrupted or may not be of satisfactory quantity or quality, and our dependence on these third parties may impair the advancement of our research and development programs.
We have established in-house recombinant protein generation capabilities for producing sufficient protein materials to enable a portion of our current preclinical studies. We rely on third partythird-party supply and manufacturing partners to supply the materials, components, and manufacturing services for a portion of preclinical studies and also rely on such third parties for all our clinical trial drug supplies. We do not own manufacturing facilities or supply sources for such components and materials for clinical trial supplies and our current manufacturing facilities are insufficient to supply such components and materials for all of our preclinical studies. Certain raw materials necessary for the manufacture of our therapeutic products, such as cell lines, are available from a single or limited number of source suppliers on a purchase order basis. There can be no assurance our supply of research and development, preclinical study, and clinical trial drugs and other materials will not be limited, interrupted, restricted in certain geographic regions, of satisfactory quality or quantity, or continue to be available at acceptable prices. In particular, any replacement of our therapeutic substance manufacturer could require significant effort and expertise and could result in significant delay of our preclinical or clinical activities because there may be a limited number of qualified replacements. Even if we have sufficient quantities of drug product for planned or ongoing clinical trials, delays in trial initiation, patient enrollment or other extensions of trial timelines may result in the expiration of such drug product prior to its use in such trials and necessitate additional production runs and/or fill/finish work, which in turn could extend trial timelines. In
addition, disruptions to ports and other shipping infrastructure, due in part to the impacts of the COVID-19 pandemic, may result in shortages or delays impacting the availability of materials and other supplies, which could negatively impact our manufacturers, suppliers and other third parties on whom we rely. While we have not yet suffered any direct, material negative impacts from these ongoing supply chain disruptions, we cannot be certain that we will not be impacted, which could increase our costs or negatively impact our development timelines.
The manufacturing process for a therapeutic candidate is subject to FDA and foreign regulatory authority review, and the facilities used by our contract manufacturers to manufacture our therapeutic candidates must be approved by the FDA pursuant to inspections that will be conducted after we submit our marketing application(s) to the FDA. Suppliers and manufacturers must meet applicable manufacturing requirements and undergo rigorous facility and process validation tests required by regulatory authorities in order to comply with cGMP regulations or other regulatory standards. In the event any of our suppliers or manufacturers fails to comply with such requirements or to perform its obligations to us in relation to quality, timing, or otherwise, or if our supply of components or other materials becomes limited or interrupted for other reasons, we may experience shortages resulting in delayed shipments, supply constraints, and/or stock-outs of our products, be forced to manufacture the materials alone, for which we currently do not have the capabilities or resources, or enter into an agreement with another third party, which we may not be able to do on reasonable terms, if at all. In some cases, the technical skills or
technology required to manufacture our therapeutic candidates may be unique or proprietary to the original manufacturer and we may have difficulty, or there may be contractual and intellectual property restrictions prohibiting us from, transferring such skills or technology to another third party and a feasible alternative may not exist. These factors may increase our reliance on such manufacturer or require us to obtain a license from such manufacturer in order to have another third party manufacture our therapeutic candidates. If we are required to change manufacturers for any reason, we will be required to verify the new manufacturer maintains facilities and procedures complying with quality standards and with all applicable regulations. The delays associated with the verification of a new manufacturer could negatively affect our ability to develop therapeutic candidates in a timely manner, within budget, or at all.
We expect to continue to rely on third partythird-party manufacturers if we receive regulatory approval for any therapeutic candidate. To the extent we have existing, or enter into future, manufacturing arrangements with third parties, we will depend on these third parties to perform their obligations in a timely manner consistent with contractual and regulatory requirements, including those related to quality control and assurance. If we are unable to obtain or maintain third-party manufacturing for therapeutic candidates, or to do so on commercially reasonable terms, we may not be able to develop and commercialize our therapeutic candidates successfully. Our, or a third party’s,party’s, failure to execute on our manufacturing requirements could adversely affect our business in a number of ways, including as a result of:
•an inability to initiate or continue preclinical studies or clinical trials of therapeutic candidates under development;
| |
•
| an inability to initiate or continue preclinical studies or clinical trials of therapeutic candidates under development;
|
| |
•
| delay in submitting regulatory applications, or receiving regulatory approvals, for therapeutic candidates;
|
| |
•
| the loss of the cooperation of a collaborator;
|
| |
•
| subjecting manufacturing facilities of our therapeutic candidates to additional inspections by regulatory authorities;
|
| |
•
| requirements to cease distribution or to recall batches of our therapeutic candidates; and
|
| |
•
| in the event of approval to market and commercialize a therapeutic candidate, an inability to meet commercial demands for our products.
|
•delay in submitting regulatory applications, or receiving regulatory approvals, for therapeutic candidates;
•the loss of the cooperation of a collaborator;
•subjecting manufacturing facilities of our therapeutic candidates to additional inspections by regulatory authorities;
•requirements to cease distribution or to recall batches of our therapeutic candidates; and
•in the event of approval to market and commercialize a therapeutic candidate, an inability to meet commercial demands for our products.
As product candidates progress from preclinical studies to late-stage clinical trials to marketing approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods, materials and processes, are altered along the way in an effort to optimize yield, manufacturing batch size, minimize costs and achieve consistent purity, identity, potency, quality and results. Such changes carry the risk that they will not achieve these intended objectives. Any of these changes could cause our product candidates to perform differently and could affect planned or other clinical trials conducted with product candidates produced using the modified manufacturing methods, materials, and processes. This could delay completion of clinical trials and could require non-clinical or clinical bridging and comparability studies, which could increase costs, delay approval of our product candidates and jeopardize our ability to commercialize our product candidates, if approved.
We may not successfully engage in strategic transactions, including any additional collaborations we seek, which could adversely affect our ability to develop and commercialize therapeutic candidates, impact our cash position, increase our expenses, and present significant distractions to our management.
From time to time, we may consider strategic transactions, such as collaborations, acquisitions of companies, asset purchases or divestitures, and out- or in-licensing of therapeutic candidates or technologies. In particular, we intend to evaluate and, if strategically attractive, seek to enter into additional collaborations, including with major biotechnology or pharmaceutical companies. The competition for collaborative partners is intense, and the negotiation process is time-consuming and complex. Any new collaboration may be on suboptimal terms for us, and ultimately may not maximize value for our stockholders. In addition, we may be unable to maintain any new or existing collaboration if, for example, development or approval of a therapeutic candidate is delayed, sales of an approved therapeutic product do not meet expectations, or the collaborator terminates the collaboration. Any such collaboration, or other strategic transaction, may require us to incur non-recurring or other charges, increase our near- and long-term expenditures and pose significant integration or implementation challenges or disrupt our management or business.
These transactions would entail numerous operational and financial risks, including:
•exposure to unknown liabilities;
| |
•
| exposure to unknown liabilities;
|
| |
•
| disruption of our business and diversion of our management’s time and attention in order to manage a collaboration or develop acquired therapeutic candidates, or technologies;
|
| |
•
| incurrence of substantial debt or dilutive issuances of equity securities to pay transaction consideration or costs;
|
| |
•
| higher than expected collaboration, acquisition, or integration costs;
|
| |
•
| write-downs of assets or goodwill, or incurring impairment charges or increased amortization expenses; and
|
| |
•
| difficulty and cost in facilitating the collaboration or combining the operations and personnel of any acquired business or impairment of relationships with key suppliers, manufacturers, or customers of any acquired business due to changes in management and ownership and the inability to retain key employees of any acquired business.
|
•disruption of our business and diversion of our management’s time and attention in order to manage a collaboration or develop acquired therapeutic candidates, or technologies;
•incurrence of substantial debt or dilutive issuances of equity securities to pay transaction consideration or costs;
•higher than expected collaboration, acquisition, or integration costs;
•write-downs of assets, or incurring impairment charges or increased amortization expenses; and
•difficulty and cost in facilitating the collaboration or combining the operations and personnel of any acquired business or impairment of relationships with key suppliers, manufacturers, or customers of any acquired business due to changes in management and ownership and the inability to retain key employees of any acquired business.
Accordingly, although there can be no assurance we will undertake or successfully complete any transactions of the nature described above, any transactions we do complete may be subject to the foregoing or other risks and have a material
adverse effect on our business, results of operations, financial condition, and prospects. Conversely, any failure to enter any collaboration or other strategic transaction beneficial to us could delay the development and potential commercialization of our therapeutic candidates and have a negative impact on the competitiveness of any therapeutic candidate reaching market.
We face competition from entities that have developed or may develop therapeutic candidates for our target disease indications, including companies developing novel treatments and technology platforms based on modalities and technology similar
Risks Related to us. If these companies develop technologies or therapeutic candidates more rapidly than we do, or their technologies, including delivery technologies, are more effective, our abilityOur Ability to develop and successfully commercialize therapeutic candidates may be adversely affected.Commercialize Product Candidates
We participate in the highly competitive sector of biotechnology and pharmaceuticals and in the subsector of immune modulation. This subsector has undergone tremendous technological advancement over the last decade due to advancements in understanding the role of the immune system across multiple therapeutic areas, including oncology and autoimmune/inflammatory disease. While we believe our novel technology platform, discovery programs, knowledge, experience, and scientific resources offer competitive advantages, we face competition from major pharmaceutical and biotechnology companies, academic institutions, governmental agencies, public and private research institutions, and others.
Any products we successfully develop and commercialize will face competition from currently approved therapies and new therapies potentially available in the future.
The availability of reimbursement from government and other third-party payors will also significantly affect the pricing and competitiveness of our products. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for our products, which could result in our competitors establishing a strong market position before we are able to enter the market.
Many of the companies we compete against may have significantly greater financial resources and expertise in research and development, manufacturing, pre-clinical testing, conducting clinical trials, obtaining regulatory approvals, and marketing approved products. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Specifically, our competitors include companies developing therapies with the same target(s) as ALPN-101 and ALPN-202 as well as companies building novel platforms to generate multi-specific antibody or non-antibody-based targeting proteins.
ICOSL/CD28 Competitors
The competitors listed below have programs targeting either ICOS or CD28 (or one of their ligands). To our knowledge, there are currently no competitors with a single molecule targeting ICOS and CD28 simultaneously.
| |
•
| an anti-BAFF, anti-ICOSL bispecific antibody being developed by Amgen, Inc. (AMG570/MEDI0700);
|
| |
•
| an anti-CD28 monoclonal antibody fragment being developed by OSE ImmunoTherapeutics SA (FR104); and
|
| |
•
| a CD28-modulatory peptide being developed by AtoxBio, Inc. (reltecimod (AB-103));
|
ALPN-202 Program Competitors
There are numerous clinical trials for immuno-oncology products used as a single agent or in combination. One of the potentially novel attributes of the ALPN-202 program is it combines inhibitory receptor antagonism and activating costimulation with a single molecule interacting with multiple immune targets.
Examples of additional multi-target compounds for immuno-oncology are highlighted below. To our knowledge, there are currently no competitors with a single molecule capable of dual PD-L1/CTLA-4 antagonism and PD-L1-dependent CD28 agonism.
| |
•
| wild-type CD80-Fc being developed by Five Prime Therapeutics, Inc. (FPT155);
|
| |
•
| bispecific antibodies being developed by Regeneron targeting tumor specific antigens and CD28 (REGN5678 anti-PSMAxCD28);
|
| |
•
| trispecific antibodies being developed by Sanofi (CD3xCD38xCD28);
|
| |
•
| bifunctional fusion protein composed of monoclonal antibody against programmed death ligand 1 (“PD-L1”) fused to the extracellular domain of human transforming growth factor–β (“TGF-β”) receptor II being developed by EMD Serono, Inc and GlaxoSmithKline plc (bintrafusp alfa, or M7824);
|
| |
•
| bifunctional fusion protein composed of PD-1 and OX40L developed by Shattuck Labs, Inc. (SL-279252);
|
| |
•
| bispecific fusion protein targeting 4-1BB and PD-L1 being developed by Pieris Pharmaceuticals, Inc. (PRS-344);
|
| |
•
| bispecific monoclonal antibodies being developed by Xencor, Inc. including XmAb20717 targeting CTLA-4 and PD-1, XmAb22841 targeting CTLA-4 and LAG-3, and XmAb23104 targeting PD-1 and ICOS;
|
| |
•
| bispecific constructs called “DARTs” being developed by Macrogenics, Inc., including MGD013 targeting PD-1 and LAG-3 and MGD019 targeting PD-1 and CTLA-4;
|
| |
•
| bispecific monoclonal antibody being developed by Tesaro, Inc., which was purchased by GlaxoSmithKline plc, targeting PD-1 and LAG-3;
|
| |
•
| small molecule antagonists being developed by Aurigene Ltd and Curis, Inc., including CA-170 targeting PD-L1 and VISTA and CA-327 targeting PD-L1 and TIM-3;
|
| |
•
| FS118, a bispecific monoclonal antibody targeting PD-L1 and LAG-3 being developed by F-star Biotechnology, Ltd.;
|
| |
•
| various combinations of separate anti PD-1/L1 and anti-CTLA-4 monoclonal antibodies; and
|
| |
•
| various combinations of separate anti PD-1/L1 and costimulatory monoclonal antibodies such as OX-40, 4-1BB, and others.
|
Novel Platform Competitors
Multifunctional therapeutic protein platforms potentially competitive with our platform include:
| |
•
| Amgen, Inc. (BiTE®): fusion proteins consisting of two single-chain variable fragments to link T-cells to tumors;
|
| |
•
| Macrogenics, Inc. (DART®): Dual-Affinity Re-Targeting and Trident technology platforms bind multiple targets with a single molecule;
|
| |
•
| Xencor, Inc. (XmAb Bispecific): Optimized Fc domains for improved potency, half-life and stability;
|
| |
•
| Zymeworks, Inc. (Azymetric™): Proprietary amino acid modifications to facilitate interaction of distinct heavy chains;
|
| |
•
| Pieris Pharmaceuticals, Inc. (Anticalin®): Engineered proteins derived from natural lipocalins found in blood plasma;
|
| |
•
| Compass Therapeutics, LLC (Targeted Immunomodulation™, StitchMabs™): Antibody discovery targeting the tumor-immune synapse;
|
| |
•
| Harpoon Therapeutics, Inc.: TriTAC™ (Tri-specific T cell Activating Construct) contain CD3 binding domain, half-life extension domain, and antigen-binding domain;
|
| |
•
| Shattuck Labs, Inc.: Agonist Redirected Antibody platform claimed to bind tumor-necrosis factor (“TNF”) and checkpoint targets;
|
| |
•
| Ablynx NV (Nanobody®), purchased by Sanofi Pharma, Inc.: Platform technology of single-domain, heavy-chain antibody fragments derived from camelidae (e.g., camels and llamas);
|
| |
•
| Regeneron, Inc.: VEGF Trap and VelociSuite® antibody technology platforms; and
|
| |
•
| Five Prime Therapeutics, Inc.: Proprietary protein library and rapid protein production and testing platform.
|
Additionally, there are a number of other therapies for autoimmune/inflammatory diseases or cancer approved or in development that are also competitive with our lead program and other programs in development. Many of the other therapies include other types of immunotherapies with different targets than our programs. Other potentially competitive therapies work in ways distinct from our development programs.
Many of our competitors have significantly greater financial, technical, manufacturing, marketing, sales, and supply resources or experience than we have. If we successfully obtain approval for any therapeutic candidate, we will face competition based on many different factors, including safety and effectiveness, ease with which our products can be administered and the extent to which patients accept relatively new routes of administration, timing and scope of regulatory
approvals, availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage, and patent position of our products. Competing products could present superior treatment alternatives, including by being more effective, safer, less expensive, or marketed and sold more effectively than any products we may develop. Competitive products may make any products we develop obsolete or noncompetitive before we recover the expense of developing and commercializing our therapeutic candidates. Competitors could also recruit our employees, which could negatively impact our ability to execute our business plan.
Any inability to attract and retain qualified key management and technical personnel would impair our ability to implement our business plan.
Our success largely depends on the continued service of key management and other specialized personnel, including Mitchell H. Gold, M.D., our Executive Chairman and Chief Executive Officer, Stanford Peng, M.D., Ph.D., our President and Head of Research and Development, and Paul Rickey, our Senior Vice President and Chief Financial Officer.
The loss of one or more members of our management team or other key employees or advisors could delay our research and development programs and materially harm our business, financial condition, results of operations, and prospects. The relationships our key managers have cultivated within our industry make us particularly dependent upon their continued employment with us. We are dependent on the continued service of our technical personnel because of the highly technical nature of our therapeutic candidates and technologies, and the specialized nature of the regulatory approval process. Because our management team and key employees are not obligated to provide us with continued service, they could terminate their employment with us at any time without penalty. We do not maintain key person life insurance policies on any of our management team members or key employees. Our future success will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical, and management personnel, as well as personnel with expertise in clinical testing, manufacturing, governmental regulation, and commercialization. We face competition for personnel from other companies, universities, public and private research institutions, government entities, and other organizations, including significant competition in the Seattle employment market.
As our therapeutic candidates advance into clinical trials, we may experience difficulties in managing our growth and expanding our operations.
We have limited experience in therapeutic development and very limited experience with clinical trials of therapeutic candidates. As our therapeutic candidates enter and advance through preclinical studies and clinical trials, we will need to expand our development, regulatory, and manufacturing capabilities or contract with other organizations to provide these capabilities for us. In the future, we expect to have to manage additional relationships with collaborators or partners, suppliers, and other organizations. Our ability to manage our operations and future growth will require us to continue to improve our operational, financial, and management controls, reporting systems, and procedures. We may not be able to implement improvements to our management information and control systems in an efficient or timely manner and may discover deficiencies in existing systems and controls.
If any of our therapeutic candidates are approved for marketing and commercialization and we are unable to develop sales, marketing and distribution capabilities on our own or enter into agreements with third parties to perform these functions on acceptable terms, we may be unable to successfully commercialize any such future products.
We currently have no sales, marketing, or distribution capabilities or experience. If any of our therapeutic candidates are approved, we will need to develop internal sales, marketing, and distribution capabilities to commercialize such products, which may be expensive and time-consuming, or enter into collaborations with third parties to perform these services. If we decide to market our products directly, we will need to commit significant financial, legal, and managerial resources to develop a marketing and sales force with technical expertise and supporting distribution, administration, and compliance capabilities. If we rely on third parties with such capabilities to market our approved products, or decide to co-promote products with collaborators, we will need to establish and maintain marketing and distribution arrangements with third parties, and there can be no assurance we will be able to enter into such arrangements on acceptable, compliant terms or at all. In entering into third-party marketing or distribution arrangements, any revenue we receive will depend upon the efforts of the third parties and there can be no assurance such third parties will establish adequate sales and distribution capabilities or be successful in gaining market acceptance of any approved therapeutic. If we are not successful in commercializing any therapeutic approved in the future, either on our own or through third parties, our business, financial condition, results of operations, and prospects could be materially and adversely affected.
If we fail to comply with U.S. and foreign regulatory requirements, regulatory authorities could limit or withdraw any marketing or commercialization approvals we may receive and subject us to other penalties that could materially harm our business.
Our company, our therapeutic candidates, our suppliers, and our contract manufacturers, distributors, and contract testing laboratories are subject to extensive regulation by governmental authorities in the European Union, the United States, and other countries, with regulations differing from country to country.
Even if we receive marketing and commercialization approval of a therapeutic candidate, we and our third-party service providers will be subject to continuing regulatory requirements, including a broad array of regulations related to establishment registration and product listing, manufacturing processes, risk management measures, quality and pharmacovigilance systems, post-approval clinical studies, labeling and packaging, advertising and promotional activities, record keeping, distribution, adverse event reporting, import and export of pharmaceutical products, pricing, sales, and marketing, and fraud and abuse requirements.
Furthermore, the FDA strictly regulates manufacturers’ promotional claims of drug products. In particular, a drug product may not be promoted by manufacturers for uses that are not approved by the FDA, as reflected in the FDA-approved labeling, although healthcare professionals are permitted to use drug products for off-label uses. The FDA, the Department of Justice, the Inspector General of the Department of Health and Human Services, among other government agencies, actively enforce the laws and regulations prohibiting manufacturers’ promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability, including large civil and criminal fines, penalties, and enforcement actions. The FDA has also imposed consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed for companies that engaged in such prohibited activities. If we cannot successfully manage the promotion of our approved product candidates, we could become subject to significant liability, which would materially adversely affect our business and financial condition.
We are required to submit safety and other post market information and reports, and are subject to continuing regulatory review, including in relation to adverse patient experiences with the product and clinical results reported after a product is made commercially available, both in the United States and in any foreign jurisdiction in which we seek regulatory approval. The FDA and certain foreign regulatory authorities, such as the EMA, have significant post-market authority, including the authority to require labeling changes based on new safety information and to require post-market studies or clinical trials to evaluate safety risks related to the use of a product or to require withdrawal of the product from the market.
The FDA also has the authority to require a Risk Evaluation and Mitigation Strategies, or REMS plan either before or after approval, which may impose further requirements or restrictions on the distribution or use of an approved therapeutic. The EMA now routinely requires risk management plans, or RMPs, as part of the marketing authorization application process, and such plans must be continually modified and updated throughout the lifetime of the product as new information becomes available. In addition, the relevant governmental authority of any EU member state can request an RMP whenever there is a concern about the risk/benefit balance of the product.
The manufacturers and manufacturing facilities we use to make a future product, if any, will also be subject to periodic review and inspection by the FDA and other regulatory agencies, including for continued compliance with cGMP requirements. The discovery of any new or previously unknown problems with our third-party manufacturers, manufacturing processes or facilities may result in restrictions on the product, manufacturers or facilities, including withdrawal of the product from the market. If we rely on third-party manufacturers, we will have limited control over compliance with applicable rules and regulations by such manufacturers.
If we or our collaborators, manufacturers, or service providers fail to comply with applicable continuing regulatory requirements in the U.S. or foreign jurisdictions in which we seek to market our products, we may be subject to, among other things, fines, warning and untitled letters, clinical holds, a requirement to conduct additional clinical trials, delay or refusal by the FDA or foreign regulatory authorities to approve pending applications or supplements to approved applications, suspension, refusal to renew or withdrawal of regulatory approval, product recalls, seizures, or administrative detention of products, refusal to permit the import or export of products, operating restrictions, inability to participate in government programs including Medicare and Medicaid, and total or partial suspension of production or distribution, injunction, restitution, disgorgement, debarment, civil penalties, and criminal prosecution.
Imposed price controls may adversely affect our future profitability.
In most countries, the pricing of prescription drugs is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after receipt of marketing approval for a product. In
addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic, and regulatory developments may further complicate pricing and reimbursement negotiations, and pricing negotiations may continue after reimbursement has been obtained.
Reference pricing used by various EU member states and parallel distribution, or arbitrage between low-priced and high-priced member states, can further reduce prices. In some countries, we or our collaborators may be required to conduct a clinical trial or other studies comparing the cost-effectiveness of our therapeutic candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If
reimbursement of any product candidate approved for marketing is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business, financial condition, results of operations, or prospects could be adversely affected.
Our business may become subject to economic, political, regulatory and other risks associated with international operations.
Our business is subject to risks associated with conducting business internationally, including our use of foreign clinical trial sites. Some of our suppliers, collaborators and clinical trial relationships are located outside the United States. Accordingly, our future results could be harmed by a variety of factors, including:
•economic instability or weakness, including inflation, reduced growth, diminished credit availability, weakened consumer confidence or increased unemployment;
•sociopolitical instability in particular foreign economies and markets;
•difficulties in compliance with non-U.S. laws and regulations;
•changes in non-U.S. regulations and customs, tariffs and trade barriers, including any changes that China may impose as a result of political tensions between the United States and China;
•changes in non-U.S. currency exchange rates and currency controls;
•trade protection measures, import or export licensing requirements or other restrictive actions by U.S. or non-U.S. governments;
•workforce uncertainty in countries where labor unrest is more common than in the United States;
•production shortages resulting from any events affecting raw material supply or manufacturing capabilities outside the United States;
•ongoing ramifications of the United Kingdom’s withdrawal from the European Union;
•business interruptions resulting directly or indirectly from geopolitical actions, including the Russia-Ukraine conflict, other regional conflicts, war and terrorism, or natural disasters including earthquakes, typhoons, floods and fires; and
•changes in regulatory requirements and policies that apply to our business, including changes in the regulatory approval process, clinical trial requirements, data standards, and export regulations and controls.
Risks Related to Our Personnel and Operations
We will need to raise substantial additional funds to advance development of our therapeutic candidates, and we cannot guarantee we will have sufficient funds available in the future to develop and commercialize our current or future therapeutic candidates.
We will need to raise substantial additional funds to expand our development, regulatory, manufacturing, marketing, and sales capabilities or contract with other organizations to provide these capabilities to us. We have used substantial funds to develop our therapeutic candidates and will require significant funds to conduct further research and development, preclinical testing, and clinical trials of our therapeutic candidates, to seek regulatory approvals for our therapeutic candidates, and to manufacture and market products, if any are approved for commercial sale. As of December 31, 2022, we had $273.4 million in cash and cash equivalents, restricted cash, and investments. Based on our current operating plan, we believe our available cash and cash equivalents and investments will be sufficient to fund our planned level of operations, including anticipated capital expenditures, for at least the next 12 months. Our future capital requirements and the period for which we expect our existing resources to support our operations may vary significantly from what we expect. Our monthly spending levels vary based on new and ongoing development and corporate activities. Because the length of time and activities associated with successful
development of our therapeutic candidates are highly uncertain, we are unable to estimate the actual funds we will require for development and any approved marketing and commercialization activities. To execute our business plan, we will need, among other things:
•to obtain the human and financial resources necessary to develop, test, obtain regulatory approval for, manufacture, and market our therapeutic candidates;
•to build and maintain a strong intellectual property portfolio and avoid infringing intellectual property of third parties;
•to establish and maintain successful licenses, collaborations, and alliances;
•to satisfy the requirements of clinical trial protocols, including patient enrollment;
•to establish and demonstrate the clinical efficacy and safety of our therapeutic candidates;
•to obtain regulatory approvals;
•to manage our spending as costs and expenses increase due to preclinical studies, clinical trials, regulatory approvals, manufacturing scale-up, and commercialization;
•to obtain additional capital to support and expand our operations; and
•to market our products to achieve acceptance and use by the medical community in general.
If we are unable to obtain necessary funding on a timely basis or on acceptable terms, we may have to delay, reduce, or terminate our research and development programs, preclinical studies, or clinical trials, if any, limit strategic opportunities, or undergo reductions in our workforce or other corporate restructuring activities. We also could be required to seek funds through arrangements with collaborators or others requiring us to relinquish rights to some of our technologies or therapeutic candidates we would otherwise pursue on our own. We do not expect to realize revenue from product sales, or royalties in the foreseeable future, if at all. Our revenue sources are, and will remain, extremely limited unless and until our therapeutic candidates are clinically tested, approved for commercialization, and successfully marketed.
To date, we have financed our operations primarily through the sale of equity securities, debt, and payments received under our collaboration agreements. We will be required to seek additional funding in the future and intend to do so through a combination of public or private equity offerings, debt financings, credit and loan facilities, research collaborations, and license agreements. Our ability to raise additional funds from these or other sources will depend on financial, economic, and other factors, many of which are beyond our control. Additional funds may not be available to us on acceptable terms or at all.
If we raise additional funds by issuing equity securities, our stockholders will suffer dilution, and the terms of any financing may adversely affect the rights of our stockholders. For example, in September 2021, we issued in a private placement 6,489,357 shares of common stock and prefunded warrants to purchase an additional 3,191,487 shares of common stock for gross proceeds of approximately $91.0 million. In December 2021, in connection with the Horizon Agreement, we sold 951,980 shares of our common stock to Horizon for which they paid $15.0 million. In September 2022, we sold an aggregate of 15,509,282 shares of common stock in an underwritten public offering, including the partial exercise of the underwriters’ over-allotment option in October 2022, pursuant to our effective shelf registration statement on Form S-3 (File No. 333-256107). We received resulting net proceeds of $106.7 million after deducting underwriting discounts, commissions and offering costs. From time to time, we also have entered into “at the market” equity offering arrangements that enable us to sell shares of our common stock from time to time through an “at the market” equity offering. In September 2022, we terminated our “at the market” equity offering arrangement with Cowen and Company, LLC in connection with our September 2022 underwritten offering, but we may in the future implement similar “at the market” equity offering arrangements.
In addition, as a condition to providing additional funds to us, future investors may demand, and may be granted, rights superior to those of existing stockholders. Debt financing, if available, may involve restrictive covenants limiting our flexibility in conducting future business activities, and, in the event of a liquidation or insolvency, debt holders would be repaid before holders of equity securities receive any distribution of corporate assets. Our failure to raise capital or enter into such other arrangements within a reasonable timeframe would have a negative impact on our financial condition, and we may have to delay, reduce, or terminate our research and development programs, preclinical or clinical trials, or undergo reductions in our workforce or other corporate restructuring activities.
We are an early stage biopharmaceutical company with a history of losses, we expect to continue to incur significant losses for the foreseeable future, we may never achieve or maintain profitability, and we have a limited operating history that may make it difficult for investors to evaluate the potential success of our business.
We are a clinical-stage immunotherapy company, with a limited operating history, focused on developing treatments for autoimmune and inflammatory diseases. Since inception, we have devoted our resources to developing novel protein-based immunotherapies primarily using our proprietary directed evolution platform, which converts native immune system proteins into potential differentiated, multi-targeted therapeutics designed to modulate the immune system. We have had significant operating losses since inception. For the year ended December 31, 2022, our net loss was $57.8 million. Substantially all of our losses have resulted from expenses incurred in connection with our research programs and from general and administrative costs associated with our operations. In addition, inflationary pressure could adversely impact our financial results. Our operating costs have increased, and may continue to increase, due to the continued inflationary environment. Our technologies and therapeutic candidates are in early stages of development, and we are subject to the risks of failure inherent in the development of therapeutic candidates based on novel technologies.
We have historically generated revenue primarily from the receipt of research funding and upfront and other payments under our collaboration agreements. We have not generated, and do not expect to generate, any revenue from product sales for the foreseeable future, and we expect to continue to incur significant operating losses for the foreseeable future due to the cost of research and development, preclinical studies, clinical trials, and the regulatory approval process for therapeutic candidates. The amount of future losses is uncertain. Our ability to achieve profitability, if ever, will depend on, among other things, our or our existing collaborators, or any future collaborators, successfully developing therapeutic candidates, obtaining regulatory approvals to market and commercialize therapeutic candidates, manufacturing any approved products on commercially reasonable terms, establishing a sales and marketing organization or suitable third-party alternatives for any approved product, and raising sufficient funds to finance business activities. If we or our existing collaborators, or any future collaborators, are unable to develop and commercialize one or more of our therapeutic candidates or if sales revenue from any therapeutic candidate receiving approval is insufficient, we will not achieve profitability, which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
Interim, preliminary, or topline data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publish interim, preliminary or topline data from clinical trials. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Interim or preliminary data from clinical trials that we may conduct may not be indicative of the final results of the trial and are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data becomes available. Interim or preliminary data also remains subject to audit and verification procedures that may result in the final data being materially different from the interim or preliminary data. As a result, interim or preliminary data should be viewed with caution until the final data are available. Adverse differences between interim, preliminary or topline data and final data could significantly harm our reputation and business prospects. We do not know whether any clinical trials we may conduct will demonstrate consistent or adequate efficacy and safety sufficient to obtain marketing approval to market our product candidates.
Moreover, preliminary, interim and topline data are subject to the risk that one or more of the clinical outcomes may materially change as more patient data become available when patients mature on study, patient enrollment continues or as other ongoing or future clinical trials with a product candidate further develop. Past results of clinical trials may not be predictive of future results. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is based on what is typically more extensive information, and you or others may not agree with what we determine is the material or otherwise appropriate information to include in our disclosure. Any information we determine not to disclose may ultimately be deemed significant with respect to future decisions, conclusions, views, activities or otherwise regarding a particular product candidate or our business. Similarly, even if we are able to complete our planned and ongoing preclinical studies and clinical trials of our product candidates according to our current development timeline, the positive results from such preclinical studies and clinical trials of our product candidates may not be replicated in subsequent preclinical studies or clinical trial results. Moreover, preclinical, nonclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless failed to obtain FDA or other regulatory approval.
Any inability to attract and retain qualified key management and technical personnel would impair our ability to implement our business plan.
Our success largely depends on the continued service of key management and other specialized personnel, including Mitchell H. Gold, M.D., our Executive Chairman and Chief Executive Officer, Stanford Peng, M.D., Ph.D., our President and Head of Research and Development, and Paul Rickey, our Senior Vice President and Chief Financial Officer.
The loss of one or more members of our management team or other key employees or advisors could delay our research and development programs and materially harm our business, financial condition, results of operations, and prospects. The relationships our key managers have cultivated within our industry make us particularly dependent upon their continued employment with us. We are dependent on the continued service of our technical personnel because of the highly technical nature of our therapeutic candidates and technologies, and the specialized nature of the regulatory approval process. Because our management team and key employees are not obligated to provide us with continued service, they could terminate their employment with us at any time without penalty. We do not maintain key person life insurance policies on any of our management team members or key employees. Our future success will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical, and management personnel, as well as personnel with expertise in clinical testing, manufacturing, governmental regulation, and commercialization. We face competition for personnel from other companies, universities, public and private research institutions, government entities, and other organizations, including significant competition in the Seattle employment market.
As our therapeutic candidates advance into clinical trials, we may experience difficulties in managing our growth and expanding our operations.
We have limited experience in therapeutic development and very limited experience with clinical trials of therapeutic candidates. As our therapeutic candidates enter and advance through preclinical studies and clinical trials, we will need to expand our development, regulatory, and manufacturing capabilities or contract with other organizations to provide these capabilities for us. For example, as we continue enrollment in our Phase 2 study in SLE and continue with the development of our other product candidates, we will need to hire additional personnel in clinical operations. We also must manage relationships with collaborators or partners, suppliers, and other organizations. Our ability to manage our operations and future growth will require us to continue to improve our operational, financial, and management controls, reporting systems, and procedures. We may not be able to implement improvements to our management information and control systems in an efficient or timely manner and may discover deficiencies in existing systems and controls.
Our business entails a significant risk of product liability and our inability to obtain sufficient insurance coverage could harm our business, financial condition, results of operations, or prospects.
Our business exposes us to significant product liability risks inherent in the development, testing, manufacturing, and marketing of therapeutic treatments. Product liability claims could delay or prevent completion of our development programs. If we succeed in marketing products, such claims could result in an investigation by certain regulatory authorities, such as FDA or foreign regulatory authorities, of the safety and effectiveness of our products, our manufacturing processes and facilities, or our marketing programs and potentially a recall of our products or more serious enforcement action, limitations on the approved indications for which they may be used, or suspension or withdrawal of approvals. Regardless of the merits or eventual outcome, liability claims may also result in decreased demand for our products, injury to our reputation, costs to defend the related litigation, a diversion of management’smanagement’s time and our resources, substantial monetary awards to trial participants or patients, and a decline in our valuation. We currently have product liability insurance we believe is appropriate for our stage of development and may need to obtain higher levels of product liability insurance prior to marketing any therapeutic candidates. Any insurance we have or may obtain may not provide sufficient coverage against potential liabilities. Furthermore, clinical trial and product liability insurance is becoming increasingly expensive. As a result, we may be unable to obtain sufficient insurance at a reasonable cost to protect us against losses caused by product liability claims with a potentially material adverse effect on our business.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect on our business.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include, but is not limited to:
•intentional failures to comply with FDA or U.S. health care laws and regulations, or applicable laws, regulations, guidance, or codes of conduct set by foreign governmental authorities or self-regulatory industry organizations;
| |
•
| intentional failures to comply with FDA or U.S. health care laws and regulations, or applicable laws, regulations, guidance, or codes of conduct set by foreign governmental authorities or self-regulatory industry organizations;
|
| |
•
| a provision of inaccurate information to any governmental authorities such as FDA;
|
| |
•
| noncompliance with manufacturing standards we may establish;
|
| |
•
| noncompliance with federal and state healthcare fraud and abuse laws and regulations; and
|
| |
•
| a failure to report financial information or data accurately or a failure to disclose unauthorized activities to us.
|
•a provision of inaccurate information to any governmental authorities such as FDA;
•noncompliance with manufacturing standards we may establish;
•noncompliance with federal and state healthcare fraud and abuse laws and regulations;
•noncompliance with the U.S. Foreign Corrupt Practices Act (the FCPA) and similar anti-bribery or anti-corruption laws, regulations or rules of other countries in which we operate; and
•a failure to report financial information or data accurately or a failure to disclose unauthorized activities to us.
In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws, regulations, guidance and codes of conduct intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws, regulations, guidance statements, and codes of conduct may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive program, health care professional, and other business arrangements. As our business is heavily regulated, it involves significant interaction with government officials, including potentially officials of non-U.S. governments. Additionally, in many countries, healthcare providers are employed by the government, and the purchasers of biopharmaceuticals are government entities. As a result, our dealings with are subject to regulation and such healthcare providers and employees of such purchasers may be considered “foreign officials” as defined in the FCPA. Recently, the Securities and Exchange Commission, or SEC, and Department of Justice have increased their FCPA enforcement activities with respect to biotechnology companies. We also may be subject to U.S. and foreign export controls, trade sanctions and import laws and regulations and if we fail to comply with these laws and regulations, we may face significant penalties, finds and/or denial of certain export privileges.
Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions, including debarment or disqualification of those employees from participation in FDA regulated activities and serious harm to our reputation. This could include violations of provisions of the U.S. federal Health Insurance Portability and Accountability Act, or HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, other U.S. federal and state law, and requirements of non-U.S. jurisdictions, including the European Union General Data Protection Directive.Regulation.
It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws, regulations, guidance or codes of conduct. Furthermore, we may be held liable under the FCPA and similar laws in other jurisdictions for the corrupt or other illegal activities of our employees, our third-party business partners, representatives and agents, even if we do not explicitly authorize such activities. If any such governmental investigations or other actions or lawsuits are instituted against us, and we are not successful in defending such actions or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines, exclusion from government programs, or other sanctions.
Our business involves the use of hazardous materials and we and our third-party manufacturers must comply with environmental laws and regulations, which may be expensive and restrict how we conduct business.
Our third-party manufacturers’manufacturers’ activities and our own activities involve the controlled storage, use and disposal of hazardous and flammable materials, including the components of our pharmaceutical product candidates, test samples and reagents, biological materials and other hazardous compounds. We and our manufacturers are subject to federal, state, local, and foreign laws and regulations governing the use, generation, manufacture, storage, handling, and disposal of these hazardous materials. Although we believe our safety procedures for handling and disposing of these materials and waste products comply with the standards prescribed by these laws and regulations, we cannot eliminate the risk of accidental injury or contamination from the use, storage, handling, or disposal of hazardous materials. In the event of an accident, state, or federal or other applicable authorities may curtail our use of these materials and/or interrupt our business operations. In addition, if an accident or environmental discharge occurs, or if we discover contamination caused by prior operations, including by prior owners and operators of properties we acquire, we could be liable for cleanup obligations, damages, and fines. If such unexpected costs are substantial, this could significantly harm our financial condition and results of operations.
Compliance with governmental regulations regarding the treatment of animals used in research could increase our operating costs, which would adversely affect the commercialization of our technology.
The Animal Welfare Act, or AWA, is the federal law covering the treatment of certain animals used in research. Currently, the AWA imposes a wide variety of specific regulations governing the humane handling, care, treatment, and transportation of certain animals by producers and users of research animals, most notably relating to personnel, facilities,
sanitation, cage size and feeding, watering, and shipping conditions. Third parties with whom we contract are subject to registration, inspections, and reporting requirements under the AWA. Furthermore, some states have their own regulations, including general anti-cruelty legislation, which establish certain standards in handling animals. Comparable rules, regulations, and or obligations exist in many foreign jurisdictions. If we or our contractors fail to comply with regulations concerning the treatment of animals used in research, we may be subject to fines and penalties and adverse publicity, and our operations could be adversely affected.
Our business and operations could suffer in the event of system failures.
Computer system, network or telecommunications failures due to events such as damage from malware, unauthorized access, terrorism, war, or natural disasters could interrupt our internal or partner operations. For example, the loss of pre-clinical trial data, data from completed or ongoing clinical trials for our product candidates or other confidential information could result in delays in our regulatory filings and development efforts, significantly increase our costs and result in other adverse impacts to our business. To the extent that any disruption or cybersecurity breach was to result in a loss of or damage to our data, or inappropriate disclosure of confidential or proprietary information, we could incur liability and other remediation costs, and the development of our product candidates could be delayed. While we have implemented security measures, our internal computer systems and the external systems and services used by our third-party CMOs, third-party CROs, or other contractors, consultants, directors and partners remain potentially vulnerable to damage from these events.
Our information technology systems could face serious disruptions adversely affecting our business.
Our information technology and other internal infrastructure systems, including corporate firewalls, servers, leased lines, and connection to the Internet, face the risk of systemic failure potentially disruptive to our operations. A significant disruption in the availability of our information technology and other internal infrastructure systems could cause interruptions in our collaborations with our partners and delays in our research and development work.
Our current operations are concentrated in one location and any events affecting this location may have material adverse consequences.
Our current operations are located in facilities situated in Seattle.Seattle, Washington. Any unplanned event, such as flood, fire, explosion, earthquake, extreme weather condition, medical epidemics, power shortage, power outage, telecommunication failure, or other natural or manmademan-made accidents or incidents resulting in our company being unable to fully utilize the facilities, may have a material adverse effect on our ability to operate our business, particularly on a daily basis, and have significant negative consequences on our financial and operating conditions. Loss of access to these facilities may result in increased costs, delays in the development of our therapeutic candidates, or interruption of our business operations. As part of our risk management policy, we maintain insurance coverage at levels we believe are appropriate for our business. However, in the event of an accident or incident at these facilities, we cannot assure you the amounts of insurance will be sufficient to satisfy any damages and losses or that the insurance covers all risks. If our facilities are unable to operate because of an accident or incident or for
any other reason, even for a short period of time, any or all of our research and development programs may be harmed. Any business interruption may have a material adverse effect on our business, financial position, results of operations, and prospects.
Our business may be affected by litigation and government investigations.
From time to time, we receive inquiries and other types of information requests from government authorities as well as correspondence from other third parties regarding various disputes and allegations. We cannot predict whether any such inquiries or correspondence may ultimately subject us to claims and other actions related to our business activities. While the ultimate outcome of investigations, inquiries, information requests, disputes, and legal proceedings is difficult to predict, responding to such investigations, inquiries and information requests or defending such claims can be expensive, time-consuming and distracting, and adverse resolutions or settlements of those matters may result in, among other things, modification of our business practices, costs, and significant payments, any of which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
Risks Related to Our Financial Position and Capital Needs
The investment of our cash and cash equivalents andin fixed income inand other marketable securities is subject to risks which may cause losses and affect the liquidity of these investments.
As of December 31, 2019,2022, we had $40.9$273.4 million in cash and cash equivalents, restricted cash, and short-term investments.investments; our investments primarily include funds invested in highly liquid funds with a contractual maturity of each security of less than two years. We expect to continue to invest our excess cash in fixed income and other marketable securities. These investments are subject to general credit, liquidity, market and interest rate risks, including potential future impacts similar to the impact of U.S. sub-prime mortgage defaults previously affecting various sectors of the financial markets and which caused credit and liquidity issues.risks. We may realize losses in the fair value of these investments, an inability to access cash in these investments for a potentially meaningful period, or a complete loss of these investments, which would have a negative effect on our financial statements.
Adverse events or perceptions affecting the financial services industry could adversely affect our operating results, financial condition and prospects.
Limited liquidity, defaults, non-performance or other adverse developments affecting financial institutions or parties with which we do business, or perceptions regarding these or similar risks, have in the past and may in the future lead to market-wide liquidity problems. For example, on March 10, 2023, Silicon Valley Bank, or SVB, was closed and placed in receivership and, subsequently, additional financial institutions have been placed into receivership. We have a banking relationship with SVB and also are party to an Amended and Restated Loan and Security Agreement with SVB. While we do not believe our exposure to a potential loss of cash, cash equivalents and investments as a result of SVB’s receivership was material compared to our total cash, cash equivalents and investments, we faced:
•delayed access to deposits or other financial assets, and potential uninsured loss of deposits or other financial assets;
•the inability to access, roll over or extend the maturity of, or enter into new credit facilities or raise other working capital resources;
Changes in accounting rules•potential breach of obligations, including U.S. federal and regulations, or interpretations thereof, could result in unfavorable accounting charges orstate wage laws and contracts that require us to change our compensation policies.maintain letters of credit or other credit support arrangements; and
Accounting methods•termination of cash management arrangements or delays in accessing funds subject to cash management arrangements.
As a result of the U.S. government intervention, we subsequently regained access to our accounts, including the uninsured portion of our deposit accounts. However, there is no guarantee that the U.S. government will intervene to provide access to uninsured funds in the future in the event of the failure of other financial institutions, or that they would do so in a timely fashion. In such an event, we and policiesour counterparties to commercial agreements may be unable to satisfy our respective obligations or enter into new commercial arrangements and may face liquidity issues.
Concerns regarding the U.S. or international financial systems could impact the availability and cost of financing, thereby making it more difficult for biopharmaceutical companies, including policies governing revenue recognition,us to acquire financing on acceptable terms or at all.
Any of these risks could materially impact our results of operations, liquidity, financial condition and prospects.
Our business may be materially affected by changes to fiscal and tax policies. Negative or unexpected tax consequences could adversely affect our results of operations.
New tax laws, statutes, rules, regulations or ordinances could be enacted at any time, which could adversely affect our business operations, and our business and financial performance. Further, existing tax laws, statutes, rules, regulations or ordinances could be interpreted, changed, modified or applied adversely to us. For example, the Tax Cuts and Jobs Act of 2017, or TCJA, enacted in December 2017, as modified by the Coronavirus Aid, Relief, and Economic Security Act, or the CARES Act, enacted in April 2020, significantly changed the U.S. Internal Revenue Code, or IRC. Such changes include a reduction in the corporate tax rate and limitations on certain corporate deductions and credits, among other changes. In addition, beginning in 2022, the TCJA eliminated the option to deduct research and development expenditures currently and requires taxpayers to capitalize and amortize them over five or fifteen years pursuant to IRC Section 174. Although Congress is considering legislation that would repeal or defer this capitalization and amortization requirement, it is not certain that this provision will be repealed or otherwise modified. This has increased our effective tax rate and our cash tax payable in 2022, although the negative cash impact is expected to decline annually over the amortization period. If the requirement to capitalize Section 174 expenditures is not modified, it may impact our effective tax rate and our cash tax liability if and when we become profitable.
We have generally accounted for changes related expenses, and accounting for stock-based compensation, are subject to review, interpretation,the TCJA in accordance with our understanding of the legislation and guidance from our auditors and relevant accounting authorities, includingavailable as of the SEC. Changes to accounting methods or policies, or interpretations thereof, may require us to reclassify, restate, or otherwise change or revisedate of this filing as described in more detail in our financial statements.statements and will continue to monitor and assess the impact of the federal legislation on our business and the extent to which various states conform to federal tax law. As another example, on August 16, 2022, the Inflation Reduction Act of 2022, or IRA, was signed into law, with tax provisions primarily focused on implementing a 15% minimum tax on global adjusted financial statement income, effective for tax years beginning after December 31, 2022, and a 1% excise tax on share repurchases occurring after December 31, 2022. Given its recent pronouncement, it is unclear at this time what, if any, impact the IRA will have on our company's tax rate and financial results. We will continue to evaluate the IRA’s impact (if any) as further information becomes available. In addition, adverse changes in the financial outlook of our operations or further changes in tax laws or regulations could lead to changes in our valuation allowances against deferred tax assets on our accompanying Consolidated Balance Sheets, which could materially affect our results of operations. Nivalis’Our pre-merger net operating loss carryforwards and certain other tax attributes are likelymay be subject to limitations. The pre-merger
In general, under IRC Section 382 and 383, a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change net operating loss carryforwards, or NOL carryforwards, to offset future taxable income. In general, an ownership change occurs if the aggregate stock ownership of certain stockholders, generally stockholders beneficially owning five percent or more of a corporation’s common stock, applying certain look-through and aggregation rules, increases by more than 50 percentage points (by value) over such stockholders’ lowest percentage ownership during the testing period, generally three years. It is possible that our NOL carryforwards and certain other tax attributes of Alpine and of the combined organization may also be subject to limitationslimitation as a result of ownership changes resulting fromin the merger.
In general,a corporationthatundergoesan “past or in the future because of, among other things, shifts in our stock ownership,change”issubjectto limitationson itsability many of which are outside our control. Consequently, even if we achieve profitability, we may not be able to utilizeitspre-changenetoperatingloss carryforwards, or NOLs, to offsetfuturetaxableincome.In general,an ownershipchangeoccursiftheaggregatestockownership a material portion of certainstockholders,generallystockholders beneficiallyowning fivepercentor moreof a corporation’scommonstock,applyingcertainlook-throughand aggregationrules,increasesby morethan50 percentagepointsoversuch stockholders’lowestpercentage ownershipduringthetestingperiod,generallythreeyears.Nivalismayhave experiencedownershipchangesin thepastand mayexperienceownershipchangesin thefuture. In addition,theclosingof themerger in 2017 likelyresultedin an ownershipchangeforNivalis. It is likely that, due to the method by which limitations on the utilization ofour NOL carryforwards are calculated, wewillnot be ableto utilize any of Nivalis’ netoperatinglosscarryforwardsand certain othertaxattributes.ItisalsopossiblethatAlpine’snetoperatinglosscarryforwardsand certainother taxattributes,maybe subjectto limitationas a resultof ownershipchangesin thepastand/ortheclosingof themerger.Consequently,even ifweachieveprofitability,wemaynot be ableto utilizea materialportion ofAlpine’s,or any of Nivalis’,netoperatinglosscarryforwardsand certain othertaxattributes,which couldhave a materialadverseeffecton cashflow and resultsof operations.
For example, based on a preliminary Section 382 study that was commissioned during the fourth quarter of 2022, focused on our wholly owned operating company and subsidiary, AIS Operating Co., Inc., we believe we likely experienced ownership changes in 2016 and 2021. As a result of this determination, our ability to utilize research and development tax credits and NOL carryforwards created prior to September 17, 2021 has been limited. While we continue to record a full valuation allowance for our domestic tax assets and have not reduced our research and development tax credit carryforwards as of December 31, 2022, the limitations could impact our ability to use the NOL carryforwards and research and development tax credit carryforwards before expiration.
Provisions of our debt instruments may restrict our ability to pursue our business strategies.
Our term loan agreement requires us, and any debt financing we may obtain in the future may require us, to comply with various covenants that limit our ability to, among other things:
•dispose of assets;
| |
•
| complete mergers or acquisitions;
|
| |
•
| pay dividends or make other distributions to holders of our capital stock;
|
| |
•
| make specified investments;
|
| |
•
| engage in any new line or business; and
|
| |
•
| engagement in certain transactions•complete mergers or acquisitions; •incur indebtedness; •encumber assets; •pay dividends or make other distributions to holders of our capital stock; •use institutions other than our lender for certain banking services; •make specified investments; •engage in any new line or business; and •engagement in certain transactions with our affiliates. |
These restrictions could inhibit our ability to pursue our business strategies. In addition, if we are required to use our lender for certain banking services, we could be exposed to the risks discussed in “Adverse events or perceptions affecting the financial services industry could adversely affect our operating results, financial condition and prospects.” If we default under our term loan agreement, including a material adverse change in our business, operations or condition (financial or otherwise), and such event of default is not cured or waived, the lenders could terminate commitments to lend and cause all amounts outstanding with respect to the debt to be due and payable immediately, which in turn could result in cross defaults under other debt instruments.instruments, if any. Our assets and cash flow may not be sufficient to fully repay borrowings under ourany outstanding debt instruments if some or all of these instruments are accelerated upon a default. We may incur additional indebtedness in the future. The debt instruments governing such indebtedness could contain provisions that are as, or more, restrictive than our existing debt instruments. If we are unable to repay, refinance or restructure our indebtedness when payment is due, the lenders could proceed against the collateral granted to them to secure such indebtedness or force us into bankruptcy or liquidation.
Risks Related to Cybersecurity
Our computer systems, or those of any of our CROs, manufacturers, other contractors or consultants or potential future collaborators, may fail or suffer security or data privacy breaches or incidents or other unauthorized or improper access to, use of, or destruction of our proprietary or confidential data, employee data, or personal data, which could result in additional costs, loss of revenue, significant liabilities, harm to our brand and material disruption of our operations.
Despite the implementation of security measures in an effort to protect systems that store our information, given their size and complexity and the increasing amounts of information maintained on our internal information technology systems, and those of our third-party CROs, other contractors (including sites performing clinical trials) and consultants, these systems are potentially vulnerable to breakdown or other damage or interruption from service interruptions, system malfunction, natural disasters, terrorism, war and telecommunication and electrical failures, as well as security breaches and incidents from inadvertent or intentional actions by our employees, contractors, consultants, business partners, and/or other third parties, or from cyber-attacks by malicious third parties (including supply chain cyber-attacks or the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of information), which may compromise our system infrastructure or lead to the loss, destruction, alteration, prevention of access to, disclosure, or dissemination of, or damage or unauthorized access to, our data (including trade secrets or other confidential information, intellectual property, proprietary business information, and personal information) or data that is processed or maintained on our behalf, or other assets, which could result in financial, legal,
business and reputational harm to us. The increase in remote working in recent years has increased certain security threats, and phishing and social engineering attacks have increased in recent years. Additionally, cybersecurity researchers have warned of heightened risks of cyberattacks in connection with Russia’s activities in Ukraine. To the extent that any disruption or security incident were to result in any loss, destruction, unavailability, alteration, disclosure, or dissemination of, or damage or unauthorized access to, our applications, any other data processed or maintained on our behalf or other assets, or for it to be affected by litigationbelieved or reported that any of these occurred, we could incur liability, financial harm and government investigations.
reputational damage and the development and commercialization of our product candidates could be delayed. We maycannot assure you that our data protection efforts and our investment in information technology, or the efforts or investments of CROs, consultants or other third parties, will prevent significant breakdowns or breaches in systems or other cyber incidents that cause loss, destruction, unavailability, alteration or dissemination of, or damage or unauthorized access to, our data and other data processed or maintained on our behalf or other assets that could have a material adverse effect upon our reputation, business, operations or financial condition. We and certain of our contractors and consultants are, from time to time, receive inquiriessubject to cyberattacks and subpoenassecurity incidents. While we do not believe that we have experienced any significant system failure, accident or security breach to date, if any system failure, accident, or other disruption, or any security breach or incident, impacting us or any of our third-party CROs or other contractors or consultants, were to occur and cause interruptions in our operations, it could result in a material disruption of our programs and the development of our product candidates could be delayed. In addition, the loss of clinical trial data for our product candidates could result in delays in our marketing approval efforts and significantly increase our costs to recover or reproduce the data. Further, any such event that leads to loss, damage, or unauthorized access to, or use, alteration, or disclosure, dissemination or other typesprocessing of, personal information, requestsincluding personal information regarding our clinical trial subjects or employees, could harm our reputation directly, compel us to comply with federal and/or state breach notification laws and foreign law equivalents, subject us to mandatory corrective action, and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information, which could result in significant legal and financial exposure and reputational damages that could potentially have an adverse effect on our business.
Notifications and follow-up actions related to a security breach or incident could impact our reputation and cause us to incur significant costs, including legal expenses and remediation costs. For example, the loss of clinical trial data from government authoritiescompleted or future clinical trials could result in delays in our regulatory approval efforts and otherssignificantly increase our costs to recover or reproduce the lost data. We expect to incur significant costs in an effort to detect and prevent security breaches and incidents, and we may becomeface increased costs and requirements to expend substantial resources in the event of an actual or perceived security breach or incident. We also rely on third parties to manufacture our product candidates, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security incident were to result in any loss, destruction, or alteration of, unavailability of, or damage or unauthorized access to, our data or other information that is processed or maintained on our behalf, or inappropriate disclosure of or dissemination of any such information, or if any of these were perceived or reported to have occurred, we could be exposed to litigation and governmental investigations, the further development and commercialization of our product candidates could be delayed, and we could be subject to claimssignificant fines or penalties for any noncompliance with certain state, federal and/or international privacy and other actions relatedsecurity laws.
Our insurance policies may not be adequate to compensate us for the potential losses arising from any such disruption in or, failure or security breach or incident of or impacting our systems or third-party systems where information important to our business activities. Whileoperations or commercial development is stored. In addition, such insurance may not be available to us in the ultimate outcomefuture on economically reasonable terms, or at all. Further, our insurance may not cover all claims made against us and could have high deductibles in any event, and defending a suit, regardless of investigations, inquiries,its merit, could be costly and divert management attention.
Our information requests,technology systems could face serious disruptions adversely affecting our business.
Our information technology and legal proceedings is difficultother internal infrastructure systems, including corporate firewalls, servers, leased lines, and connection to predict, defensethe Internet, face the risk of litigation claims can be expensive, time-consuming and distracting, and adverse resolutions or settlements of those matters may resultsystemic failure potentially disruptive to our operations. A significant disruption in among other things, modificationthe availability of our information technology and other internal infrastructure systems, or those of third parties that perform services or supply materials to us, could cause interruptions in our collaborations with our partners and delays in our research and development work.
Our facility is located in Seattle, Washington. We have not undertaken a systematic analysis of the potential consequences to our business practices, costs, and significant payments, any offinancial results from a major flood, blizzard, fire, earthquake, power loss, terrorist activity, pandemics or other disasters and do not have a recovery plan for such disasters. Also, our contract development and manufacturing organizations’ and suppliers’ facilities are located in multiple locations where other natural disasters or similar events which could severely disrupt our operations, could expose us to liability and could have a material adverse effect on our business, financial condition, results of operations, and prospects.
We believe our development programs and platform have a particular mechanism of action, but this mechanism of action has not been proven conclusively.
Our scientific platform is novel, and the underlying science is not exhaustively understood nor conclusively proven. In particular, the interaction of vIgDs with the immune synapse, the ability of vIgDs to slow, stop, restart, or accelerate immune responses, and the ability of vIgD domains to interact with multiple counter structures is still largely theoretical. Graphical representations of proposed mechanisms of action of our therapies, the size, actual or relative, of our therapeutics, and how our therapeutics might interface with other cells within the human body, inside the immune synapse, or inside the disease and/or the tumor microenvironment are similarly theoretical and not yet conclusively proven. The lack of a proven mechanism of action may adversely affect our ability to raise sufficient capital, complete preclinical studies, adequately manufacture drug product, obtain regulatory clearance for clinical trials, gain marketing approval, or conclude collaborations, or interfere with our ability to market our product to patients and physicians or achieve reimbursement from payors.
Any inability to present our data in scientific journals or at scientific conferences could adversely impact our business and stock price.
We may from time to time submit data related to our research and development activities in peer-reviewed scientific publications or apply to present data related to our research and development activities at scientific or other conferences. We have no control over whether these submissions or applications are accepted. Even if accepted for a conference, we have no control over whether presentations at scientific conferences will be accepted for oral presentation, poster presentation, or abstract publication only. Even when accepted for publication, we have no control over the timing of the release of the publication. Rejection by publications, delays in publication, rejection for presentation, or a less-preferred format for a presentation may adversely impact our stock price, ability to raise capital, and business.
Our business may be affected by adverse scientific publications or editorial or discussant opinions.
We may from time to time publish data related to our research and development activities in peer-reviewed scientific publications or present data related to our research and development activities at scientific or other conferences. Editorials or discussants unrelated to us may provide opinions on our presented data unfavorable to us. In addition, scientific publications or presentations may be made which are critical of our science or research or the field of immunotherapy in general. This may adversely affect our ability to raise necessary capital, complete clinical and preclinical studies, adequately manufacture drug product, obtain regulatory clearance for clinical trials, or approval for marketing, or interfere with our ability to market our product to patients and physicians or achieve reimbursement from payors.
Risks Related to Our Intellectual Property
If we are not able to obtain and enforce patent protection for our technology, including therapeutic candidates, therapeutic products, and platform technology, development of our therapeutic candidates and platform, and commercialization of our therapeutic products may be materially and adversely affected.
Our success depends in part on our ability to obtain and maintain patents and other forms of intellectual property rights, including in-licenses of intellectual property rights of others, for our technology, including platform technology and therapeutic candidates and products, methods used to manufacture our therapeutic candidates and products, and methods for treating patients using our therapeutic candidates and products, as well as our ability to preserve our trade secrets, to prevent third parties from infringing upon our proprietary rights, and to operate without infringing upon the proprietary rights of others. Our scientific platform and substantially all of our intellectual property have been developed internally. As of December 31, 2019,2022, our patent portfolio consists of 54 granted patents and over 100200 pending patent applications. We may not be able to apply for patents on certain aspects of our technology, including therapeutic candidates and products, in a timely fashion or at all. Any future patents we obtain may not be sufficiently broad to prevent others from using our technology or from developing competing therapeutics and technology. There is no guarantee that any of our pending patent applications will result in issued or granted patents, any of our issued or granted patents will not later be found to be invalid or unenforceable, or any issued or granted patents will include claims sufficiently broad to cover our technology, including platform technology and therapeutic candidates and products, or to provide meaningful protection from our competitors. Moreover, the patent position of pharmaceutical and biotechnology companies can be highly uncertain because it involves complex legal and factual questions. We will be able to protect our proprietary rights from unauthorized use by third parties only to the extent our current and future technology, including platform technology and therapeutic candidates and products, are covered by valid and enforceable patents or are effectively maintained as trade secrets. If third parties disclose or misappropriate our proprietary rights, it may materially and adversely impact our competitive position in the market.
The U.S. Patent and Trademark Office, or USPTO, and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment, and other provisions during the patent process. There are situations in which noncompliance can result in abandonment or lapse of a patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Patent offices may be affected by COVID-19 or other health epidemic shut-downs, resulting in, for example, non-essential administrative tasks being delayed or eliminated. This could affect patent rights, including the partial or complete loss of patent rights in jurisdictions such as the USPTO and international patent offices. In such an event, competitors might be able to enter the market earlier than would otherwise have been the case. Further, the standards applied by the USPTO and foreign patent offices in granting patents are not always applied uniformly or predictably. For example, there is no uniform worldwide policy regarding patentable subject matter or the scope of claims allowable in biotechnology and pharmaceutical patents. As such, we do not know the degree of future protection we will have on our technology, including platform technology and therapeutic candidates and products. While we will endeavor to try to protect our technology, including platform technology and therapeutic candidates and products, with intellectual property rights such as patents, as appropriate, the process of obtaining patents is time-consuming, expensive, and sometimes unpredictable, and we can provide no assurances our technology, including our platform technology, therapeutic candidates and products, will be adequately protected in the future against unauthorized uses or competing claims by third parties.
In addition, recent and future changes to the patent laws and to the rules of the USPTO or otherand foreign patent offices may have a significant impact on our ability to protect our technology, including therapeutic candidates and products, and enforce our intellectual property rights. For example, the Leahy-Smith America Invents Act enacted in 2011 involves significant changes in patent legislation. In addition, we cannot assure that court rulings or interpretations of any court decision will not adversely impact our patents or patent applications. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, there also may be uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, the USPTO, or made in foreign jurisdictions, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents we might obtain in the future.
Moreover, no earlier than June 1, 2023, European applications will soon have the option, upon grant of a patent, of becoming a Unitary Patent which will be subject to the jurisdiction of the Unitary Patent Court, or UPC. This will be a significant change in European patent practice. As the UPC is a new court system, there is no precedent for the court, increasing the uncertainty of any patent litigation in Europe.
The issuance of a patent is not conclusive as to its inventorship, scope, validity, or enforceability. Once granted, patents may remain open to opposition, interference, re-examination, post-grant review, inter partes review, revocation, nullification, or derivation action in court or before patent offices or similar proceedings before or after allowance or grant, during which time third parties can raise objections against such initial grant. In the course of such proceedings, which may continue for a protracted period of time, the patent owner may be compelled to limit the scope of the pending, allowed or granted claims thus attacked or may lose the allowed or granted claims altogether. Our patent risks include that:
•others may, or may be able to, make, use, offer to sell, or sell compounds that are the same as or similar to our therapeutic candidates and products but that are not covered by the claims of the patents we own or license;
| |
| others may, or may be able to, make, use, offer to sell, or sell compounds that are the same as or similar to our therapeutic candidates and products but that are not covered by the claims of the patents we own or license;
|
| |
•
| we or our licensors, collaborators, or any future collaborators may not be the first to file patent applications covering certain aspects of our technology, including our platform technology, therapeutic candidates and products; |
| |
•
| others may independently develop similar or alternative technology or duplicate any of our technology without infringing our intellectual property rights;
|
•others may independently develop similar or alternative technology or duplicate any of our technology without infringing our intellectual property rights;
41•a third party may challenge our patents and, if challenged, a court may not hold that our patents are valid, enforceable, or that a third party is infringing;
•the data collected from clinical trials of our therapeutic candidates may not be sufficient to support the submission of a BLA or other submission or to obtain regulatory approval in the United States, the European Union or elsewhere;
| |
••the FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; and
| the FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; and
|
| |
•
| the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval.
|
•the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval.
Any delay or failure in obtaining required approvals could have a material adverse effect on our ability to generate revenues from the particular therapeutic candidate for which we are seeking approval. The FDA, the EMA and other regulatory authorities have substantial discretion in the approval process, and in determining when or whether regulatory approval will be obtained for any of our therapeutic candidates. Even if we believe the data collected from preclinical and clinical trials of our therapeutic candidates are promising, such data may not be sufficient to support approval by the FDA, the EMA or any other regulatory authority. Furthermore, any regulatory approval to market a product may be subject to limitations on the approved uses for which we may market the product or in the product labeling or be subject to other restrictions. Regulatory authorities also may impose requirements for costly post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of the therapeutic. In addition, the FDA has the authority to require a REMS plan as part of the approval of a BLA or New Drug Application, or NDA, or after approval, which may impose further requirements or restrictions on the distribution or use of an approved drug or
biologic, such as limiting prescribing to certain physicians or medical centers that have undergone specialized training, limiting treatment to patients who meet certain safe-use criteria, and requiring treated patients to enroll in a registry. These limitations and restrictions may limit the size of the market for the therapeutic and affect coverage and reimbursement by third-party payors.
We are also subject to numerous foreign regulatory requirements governing, among other things, the conduct of clinical trials, manufacturing, marketing authorization, pricing, and third-party reimbursement. The foreign regulatory approval process varies among countries and may include all of the risks associated with FDA approval described above as well as risks attributable to the satisfaction of local regulations in foreign jurisdictions. We are subject to regulation by foreign regulatory authorities, ethics committees, and other governmental entities with respect to the clinical trials we conduct or sponsor outside of the U.S. For example, the EU Clinical Trials Regulation, or CTR, became applicable on January 31, 2022, repealing the EU Clinical Trials Directive. The implementation of the CTR includes the implementation of the Clinical Trials Information System, a new clinical trial portal and database that will be maintained by the EMA in collaboration with the European Commission and the EU Member States. Complying with changes in regulatory requirements can incur additional costs, delay our clinical development plans, or expose us to greater liability if we are slow or unable to adapt to changes in existing requirements or new requirements or policies governing our business operations, including our clinical trials. Moreover, the time required to obtain approval may differ from that required to obtain FDA approval. Approval by the FDA does not ensure approval by regulatory authorities outside the U.S. and vice versa.
If we fail to obtain orphan drug designation or obtain or maintain orphan drug exclusivity for certain of our products, our competitors may sell products to treat the same conditions and our revenue may be reduced.
Under the Orphan Drug Act, the FDA may designate a product as an orphan drug if it is intended to treat a rare disease or condition, defined as a patient population of fewer than 200,000 in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the United States. In the United States, orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages and user-fee waivers. In addition, if a therapeutic product with orphan drug designation subsequently receives the first FDA approval for the indication for which it has such designation, the therapeutic product is entitled to orphan product exclusivity, which means the FDA may not approve any other applications to market the same therapeutic product for the same indication for seven years, except in limited circumstances such as a showing of clinical superiority over the product with orphan exclusivity or where the manufacturer is unable to assure sufficient product quantity.
As discussed above, in Catalyst Pharms., Inc. v. Becerra, 14 F.4th 1299 (11th Cir. 2021), the court disagreed with the FDA’s longstanding position that the orphan drug exclusivity only applies to the approved use or indication within an eligible disease, and not to all uses or indications within the entire disease or condition. On January 24, 2023, the FDA published a notice in the Federal Register to clarify that while the agency complies with the court’s order in Catalyst, FDA intends to continue to apply its longstanding interpretation of the regulations to matters outside of the scope of the Catalyst order – that is, the agency will continue tying the scope of orphan-drug exclusivity to the uses or indications for which a drug is approved, which permits other sponsors to obtain approval of a drug for new uses or indications within the same orphan designated disease or condition that have not yet been approved. It is unclear how future litigation, legislation, agency decisions, and administrative actions will impact the scope of the orphan drug exclusivity.
As in the United States, we may apply for designation of a therapeutic candidate as an orphan drug for the treatment of a specific indication in the European Union before the application for marketing authorization is made. In the European Union, the EMA’sEMA’s Committee for Orphan Medicinal Products, grants orphan drug designation to promote the development of products that are intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition affecting not more than five in 10,000 persons in the European Union. Additionally, designation is granted for products intended for the diagnosis, prevention or treatment of a life-threatening, seriously debilitating or serious and chronic condition when, without incentives, it is unlikely that sales of the drug in the European Union would be sufficient to justify the necessary investment in developing the drug or biological product or where there is no satisfactory method of diagnosis, prevention or treatment, or, if such a method exists, the medicine must be of significant benefit to those affected by the condition. Sponsors of orphan drugs in the European Union can enjoy economic and marketing benefits, including reduction of fees or fee waivers and up to ten years of market exclusivity for the approved indication unless another applicant can show its therapeutic product is safer, more effective, or otherwise clinically superior to the orphan-designated therapeutic product. This period may be reduced to six years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity.
We may seek orphan drug designation from the FDA and the EMA for certain of our product candidates. However, we may never receive such designation. Even if we are able to obtain orphan designation, we may not be the first to obtain
marketing approval for any particular orphan indication due to the uncertainties associated with developing pharmaceutical products. In addition, exclusive marketing rights in the United States may be limited if we seek approval for an indication broader than the orphan-designated indication or may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties can be approved for the same condition. Even after an orphan drug is approved, regulatory authorities may subsequently approve the same drug with the same active moiety for the same condition if they conclude that the later drug is safer, more effective, or makes a major contribution to patient care. Orphan drug designation neither shortens the development time or regulatory review time of a product candidate nor gives the product candidate any advantage in the regulatory review or approval process. In addition, orphan drug exclusivity could block the approval of one of our therapeutic candidates if a competitor obtains approval of the same therapeutic product as defined by the FDA before we do, or if our therapeutic candidate is determined to be within the same class as the competitor’scompetitor’s therapeutic product for the same indication or disease.
The respective orphan designation and exclusivity frameworks in the United States and in the European Union are subject to change, and any such changes may affect our ability to obtain EU or U.S. orphan designations in the future.
If we or our existing or future collaborators, manufacturers, or service providers fail to comply with healthcare laws and regulations, we or such other parties could be subject to enforcement actions, which could adversely affect our ability to develop, market, and sell our therapeutics and may harm our reputation.
Although we do not currently have any products on the market, once we begin commercializing our therapeutic candidates, if approved, we will be subject to additional healthcare statutory and regulatory requirements and enforcement by the federal, state, and foreign governments of the jurisdictions in which we conduct our business. Healthcare providers, physicians, and third-party payors play a primary role in the recommendation and prescription of any therapeutic candidates for which we obtain marketing approval. Our future arrangements with third-party payors and customers may expose us to broadly applicable fraud, abuse, and other healthcare laws and regulations constraining the business or financial arrangements and relationships through which we market, sell, and distribute the therapeutic candidates for which we obtain marketing approval. Restrictions under applicable federal and state healthcare laws and regulations include the following:
•the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons from soliciting, receiving, offering, or providing remuneration, directly or indirectly, to induce either the referral of an individual for a healthcare item or service, or the purchasing or ordering of an item or service, for which payment may be made, in whole or in part, under a federal healthcare program such as Medicare or Medicaid;
| |
••the U.S. federal False Claims Act, which imposes criminal and civil penalties, including through civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, false or fraudulent claims for payment or making a false statement to avoid, decrease, or conceal an obligation to pay money to the federal government. In addition, the government may assert a claim including items and services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act; •state all-payor fraud laws, which impose criminal and civil liability for executing a scheme to defraud any healthcare benefit program, or knowingly and willfully falsifying, concealing, or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services; similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation; •HIPAA, HITECH, and their implementing regulations, which impose obligations on certain covered entity healthcare providers, health plans, and healthcare clearinghouses as well as their business associates performing certain services involving the use or disclosure of individually identifiable health information, including mandatory contractual terms, with respect to safeguarding the privacy, security, and transmission of individually identifiable health information, and require notification to affected individuals and regulatory authorities of certain breaches of security of individually identifiable health information; •the federal Physician Payment Sunshine Act and its implementing regulations, also referred to as “Open Payments,” require applicable manufacturers of pharmaceutical and biological drugs, among other covered medical products, reimbursable under Medicare, Medicaid, or Children’s Health Insurance Programs to track and report to the CMS certain payments and transfers of value made in the previous year, including but not limited to, | the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons from soliciting, receiving, offering, or providing remuneration, directly or indirectly, to induce either the referral of an individual for a healthcare item or service, or the purchasing or ordering of an item or service, for which payment may be made, in whole or in part, under a federal healthcare program such as Medicare or Medicaid;
|
| |
•
| the U.S. federal False Claims Act, which imposes criminal and civil penalties, including through civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, false or fraudulent claims for payment or making a false statement to avoid, decrease, or conceal an obligation to pay money to the federal government. In addition, the government may assert a claim including items and services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act;
|
| |
•
| state all-payor fraud laws, which impose criminal and civil liability for executing a scheme to defraud any healthcare benefit program, or knowingly and willfully falsifying, concealing, or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services; similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
|
| |
•
| HIPAA, HITECH, and their implementing regulations, which impose obligations on certain covered entity healthcare providers, health plans, and healthcare clearinghouses as well as their business associates performing certain services involving the use or disclosure of individually identifiable health information, including mandatory contractual terms, with respect to safeguarding the privacy, security, and transmission of individually identifiable health information, and require notification to affected individuals and regulatory authorities of certain breaches of security of individually identifiable health information;
|
| |
•
| the federal Physician Payment Sunshine Act and its implementing regulations, also referred to as “Open Payments,” issued under the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, or ACA, and any subsequent amending legislation or executive actions, which require manufacturers of pharmaceutical and biological drugs reimbursable under Medicare, Medicaid, and Children’s Health Insurance Programs to report to the Department of Health and Human Services
|
all consulting fees, travel reimbursements, and research grants made to cover recipients, including physicians (defined to include doctors, dentists, optometrists, podiatrists and otherchiropractors), certain non-physician healthcare professionals (such as physician assistants and nurse practitioners, among others), and teaching hospitals, as well as information regarding physicians’ and their immediate family members’ ownership and investment interests in the applicable manufacturer, with limited exceptions; and
•analogous and similar state and foreign laws and regulations, such as, state anti-kickback and false claims laws potentially applicable to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers; and some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring drug manufacturers to report information related to payments transfers of value or gifts made to physicians and teaching hospitals with limited exceptions; andother healthcare providers or marketing expenditures.
| |
•
| analogous state laws and regulations, such as, state anti-kickback and false claims laws potentially applicable to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers; and some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring drug manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures, and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
|
Ensuring our future business arrangements with third-partiesthird parties comply with applicable healthcare laws and regulations could involve substantial costs. If our operations are found to be in violation of any such requirements, we may be subject to penalties, including civil or criminal penalties, monetary damages, the curtailment or restructuring of our operations, or exclusion from participation in government contracting, healthcare reimbursement, or other government programs, including Medicare and Medicaid, any of which could adversely affect our financial results. Although effective compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, these risks cannot be entirely eliminated. Any action against us for an alleged or suspected violation could cause our company to incur significant legal expenses and could divert our management’smanagement’s attention from the operation of our business, even if our defense is successful. In addition, achieving and sustaining compliance with applicable laws and regulations may be costly to us in terms of money, time, and resources.
If we or our current or future collaborators, manufacturers, or service providers fail to comply with applicable federal, state, or foreign laws or regulations, we could be subject to enforcement actions, which could affect our ability to develop, market, and sell our therapeutics successfully and could harm our reputation and lead to reduced acceptance of our therapeutics by the market. These enforcement actions include, among others:
•adverse regulatory inspection findings;
| |
•
| adverse regulatory inspection findings;
|
| |
•
| warning or untitled letters;
|
| |
•
| voluntary product recalls with public notification or medical product safety alerts to healthcare professionals;
|
| |
•
| restrictions on, or prohibitions against, marketing our therapeutics;
|
| |
•
| restrictions on, or prohibitions against, importation or exportation of our therapeutics;
|
| |
•
| suspension of review or refusal to approve pending applications or supplements to approved applications;
|
| |
•
| exclusion from participation in government-funded healthcare programs;
|
| |
•
| exclusion from eligibility for the award of government contracts for our therapeutics;
|
| |
•
| suspension or withdrawal of therapeutic approvals;
|
| |
•
| seizures or administrative detention of therapeutics;
|
| |
•
| restitution, disgorgement of profits, or civil and criminal penalties and fines.
|
•warning or untitled letters;
•voluntary product recalls with public notification or medical product safety alerts to healthcare professionals;
•restrictions on, or prohibitions against, marketing our therapeutics;
•restrictions on, or prohibitions against, importation or exportation of our therapeutics;
•suspension of review or refusal to approve pending applications or supplements to approved applications;
•exclusion from participation in government-funded healthcare programs;
•exclusion from eligibility for the award of government contracts for our therapeutics;
•FDA debarment;
•suspension or withdrawal of therapeutic approvals;
•seizures or administrative detention of therapeutics;
•injunctions; and
•restitution, disgorgement of profits, or civil and criminal penalties and fines.
Enacted and future legislation may increase the difficulty and cost for us to obtain marketing approval of our therapeutic candidates.
The policies of the FDA or similar regulatory authorities may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. For example, in 2016, the 21st Century Cures Act, or Cures Act, was signed into law. The Cures Act, among other things, is intended to modernize the regulation of drugs and biologics and spur innovation, but it is still being implemented and its ultimate implementation is unclear. If we or our collaborators are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or our collaborators are not able to maintain regulatory compliance, our therapeutic candidates may not obtain or maintain regulatory approval, and we may not achieve or sustain profitability, which would adversely affect our business.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or executive or administrative action, either in the United States or abroad. The FDA’s and other regulatory authorities’ policies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, and we may not achieve or sustain profitability. For example, certain policies of the Trump administration may
impact our business and industry. Namely, the Trump administration has taken several executive actions,Consolidated Appropriations Act, 2023, including the issuance of a number of Executive Orders, that could impose significant burdens on,Food and Drug Omnibus Reform Act, or otherwise materially delay, FDA’s abilityFDORA, was signed into law in December 2022. FDORA made several changes to engage in routinethe FDA’s authorities and its regulatory and oversight activitiesframework, including, among other changes, reforms to the accelerated approval pathway, such as implementing statutes through rulemaking, issuance of guidance,requiring the FDA to specify conditions for post-approval study requirements and review and approval of marketing applications.setting forth procedures for the FDA to withdraw a product on an expedited basis for non-compliance with post-approval requirements. It is difficult to predict how these requirementscurrent and future legislation, executive actions, and litigation, including the executive orders, will be implemented, and the extent to which they will impact our business, our clinical development, and the FDA’sFDA’s and other agencies’ ability to exercise itstheir regulatory authority. If theseauthority, including FDA’s pre-approval inspections and timely review of any regulatory filings or applications we submit to the FDA. To the extent any legislative or executive actions impose constraints on FDA’sFDA’s ability to engage in oversight and implementation activities in the normal course, our business may be negatively impacted.
Any therapeutics we develop may become subject to unfavorable pricing regulations, third-party coverage and reimbursement practices, or healthcare reform initiatives, thereby harming our business.
The regulations governing marketing approvals, pricing, coverage, and reimbursement for new drugs and biologics vary widely from country to country. Many countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. Although we intend to monitor these regulations, our programs are currently in the early stages of development and we will not be able to assess the impact of price regulations for a number of years. As a result, we might obtain regulatory approval for a product in a particular country but then be subject to price regulations delaying our commercial launch of the product and negatively impacting the revenues we are able to generate from the sale of the product in that country.
Our ability to commercialize any therapeutics successfully also will depend in part on the extent to which coverage and reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers, and other organizations. However, there may be significant delays in obtaining coverage for newly-approved therapeutics. Moreover, eligibility for coverage does not necessarily signify a therapeutic will be reimbursed in all cases or at a rate covering our costs, including research, development, manufacture, sale, and distribution costs. Also, interim payments for new therapeutics, if applicable, may be insufficient to cover our costs and may not be made permanent. Thus, even if we succeed in bringing one or more therapeutics to the market, these products may not be considered cost-effective, and the amount reimbursed for any of them may be insufficient to allow us to sell them on a competitive basis. Because our programs are in the early stages of development, we are unable at this time to determine their cost effectiveness, coverage prospects, potential compendia listings, or the likely level or method of reimbursement, if covered. It is equally difficult for us to predict how Medicare coverage and reimbursement policies will be applied to our products in the future, and coverage and reimbursement under different federal healthcare programs are not always consistent. Medicare reimbursement rates may also reflect budgetary constraints placed on the Medicare program.
Third-party payors often rely upon Medicare coverage policies and payment limitations in setting their own reimbursement rates. These coverage policies and limitations may rely, in part, on compendia listings for approved therapeutics. Our inability to promptly obtain relevant compendia listings, coverage, and adequate reimbursement from both government-funded and private payors for new therapeutics we develop and for which we obtain regulatory approval could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products, and our financial condition.
We believe the efforts of governments and third-party payors to contain or reduce the cost of healthcare, and legislative and regulatory proposals to broaden the availability of healthcare, will continue to affect the business and financial condition of pharmaceutical and biopharmaceutical companies. A number of legislative and regulatory changes in the healthcare system in the United States and other major healthcare markets have been proposed or enacted, and such efforts have expanded substantially in recent years. These developments could, directly or indirectly, affect our ability to sell our products, if approved, at a favorable price. In addition, third-party payors who reimburse patients or healthcare providers, such as government and private insurance plans, are seeking greater upfront discounts, additional rebates, and other concessions to reduce the prices for therapeutics. If the price we are able to charge for any therapeutics we develop, or the reimbursement provided for such products, is inadequate, our return on investment could be adversely affected.
Pursuant to health reform legislation and related initiatives, the Centers for Medicare and Medicaid Services, or CMS are working with various healthcare providers to develop, refine, and implement Accountable Care Organizations, or ACOs, and other innovative models of care for Medicare and Medicaid beneficiaries, including the Bundled Payments for Care Improvement Initiative, the Comprehensive Primary Care Initiative, the Duals Demonstration, and other models. The continued development and expansion of ACOs and other innovative models of care will have an uncertain impact on any future reimbursement we may receive for approved therapeutics administered by such organizations.
In addition, in recent years, the U.S. Congress has enacted various laws seeking to reduce the federal debt level and contain healthcare expenditures. For example, as a result of the Budget Control Act of 2011 and the Bipartisan Budget Act of 2015, an annual 2% reduction to Medicare payments that took effect in 2013 has been extendedand will remain in effect through 2025.2031. Under current legislation, the actual reduction in Medicare payments will vary from 1% in 2022 to up to 4% in the final fiscal year of this sequester. These across-the-board spending cuts could adversely affect our future revenues, earnings, and cash flows.
From timeThere has been increasing legislative and enforcement interest in the United States with respect to time,specialty drug pricing practices. Specifically, there have been several recent U.S. Congressional inquiries and proposed federal and state legislation is drafted, introduced,designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and passed in Congress that could significantly changemanufacturer patient programs, and reform government program reimbursement methodologies for drugs. For example, under the American Rescue Plan Act of 2021, effective January 1, 2024, the statutory provisions governing coverage, reimbursement, and marketingcap on Medicaid Drug Rebate Program rebates that manufacturers pay to state Medicaid programs will be eliminated. Elimination of this cap may require pharmaceutical manufacturers to pay more in rebates than it receives on the sale of products, regulatedwhich could have a material impact on our business. In July 2021, the Biden administration released an executive order, “Promoting Competition in the American Economy,” with multiple provisions aimed at increasing competition for prescription drugs. In August 2022, Congress passed the Inflation Reduction Act of 2022, which includes prescription drug provisions that have significant implications for the pharmaceutical industry and Medicare beneficiaries, including allowing the federal government to negotiate a maximum fair price for certain high-priced single source Medicare drugs, imposing penalties and excise tax for manufacturers that fail to comply with the drug price negotiation requirements, requiring inflation rebates for all Medicare Part B and Part D drugs, with limited exceptions, if their drug prices increase faster than inflation, and redesigning Medicare Part D to reduce out-of-pocket prescription drug costs for beneficiaries, among other changes. The impact of these legislative, executive, and administrative actions and any future healthcare measures and agency rules implemented by CMSthe Biden administration on us and the pharmaceutical industry as a whole is unclear. Any reduction in reimbursement from Medicare or other government agencies. In addition to new legislation, CMS coverage and reimbursement policies are often revised or interpretedprograms may result in ways that may significantly affect our business and our products. In particular, we expect the current U.S. presidential administration and Congress will seek to modify, repeal, or otherwise invalidate all, or certain provisions of, U.S. healthcare legislation. Aa reduction in payments from private payors. Additionally, a number of additionalstates are considering or have recently enacted state drug price transparency and reporting laws that could substantially increase our compliance burdens and expose us to greater liability under such state laws once we begin commercialization after obtaining regulatory approval for any of our products.
The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our product candidates. The impact of legislative, executive, orders have been issued affecting, or potentially affecting, the ACA and other aspectsadministrative actions of the Biden administration on us and the biopharmaceutical industry as a whole is unclear. We expect that additional state and federal healthcare marketreform measures will be adopted in the United States. There is a high degreefuture, any of uncertainty with respect towhich could limit the impact President Trump’s Administrationamounts that federal and Congress may have,state governments will pay for healthcare products and any changes will likely take time to unfold. Such reformsservices, which could have an adverse effect on anticipated revenues from therapeuticresult in reduced demand for our product candidates we may successfully develop and for which we may obtain regulatory approval and may affect our overall financial condition and ability to develop therapeutic candidates. However, we cannot predict the ultimate content, timing, or effect of any healthcare reform legislation or executive orders or the impact of potential legislation and executive orders on us.additional pricing pressures.
The healthcare industry is heavily regulated in the U.S. at the federal, state, and local levels and in other jurisdictions in which we may conduct trials or other activities, and our failure to comply with applicable requirements may subject us to penalties and negatively affect our financial condition.
As a healthcare company, our operations, clinical trial activities, and interactions with healthcare providers will be subject to extensive regulation in the United States, particularly if we receive FDA approval for any of our products in the future.future, and we may be subject to laws and regulations in other jurisdictions as we conduct clinical trials or engage in other activities in foreign jurisdictions. For example, if we receive FDA approval for a therapeutic for which reimbursement is available under a federal healthcare program, it would be subject to a variety of federal laws and regulations, including those prohibiting the filing of false or improper claims for payment by federal healthcare programs, prohibiting unlawful inducements for the referral of business reimbursable by federal healthcare programs, and requiring disclosure of certain payments orand other transfers of value made to covered recipients in the previous year, including U.S.-licensed physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician healthcare professionals (such as physician assistants and nurse practitioners, among others), and teaching hospitals. We are not able to predict how government authorities will interpret these laws. They may challenge our practiceshospitals, as well as information regarding certain physicians’ and activities under one or more of these laws.their immediate family members’ ownership and investment interests in the applicable manufacturer with limited exceptions. If our past or present operations, or those of our contractors or agents who conduct business on our behalf, are found to be in violation of any of these laws, we could be subject to enforcement action, government investigation, civil and
criminal penalties, which could hurt our business, operations, and financial condition. It is not always possible to identify and deter misconduct by parties we may contract with, including employees, contractors, collaborators, CROs, and suppliers, and the precautions we take to detect and prevent misconduct may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant penalties, including civil, criminal and administrative penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government funded healthcare programs, such as Medicare and Medicaid, integrity oversight and reporting obligations, contractual damages, reputational harm, diminished profits and future earnings and the curtailment or restructuring of our operations.
Similarly, some state laws prohibit, among other offenses, knowingly and willfully executing a scheme to defraud any health care benefit program, including private payors, or falsifying, concealing, or covering up a material fact or making any materially false, fictitious, or fraudulent statement in connection with the delivery of or payment for items or services under a health care benefit program. We may also be subject to the privacy and security provisions of HIPAA, as amended by HITECH, which restricts the use and disclosure of patient-identifiable health information, mandates the adoption of standards relating to the privacy and security of patient-identifiable health information, and requires the reporting of certain security breaches to healthcare provider customers with respect to such information. Additionally, many states have enacted similar laws imposing more stringent requirements on entities like us. Failure to comply with applicable laws and regulations could result in substantial penalties and adversely affect our financial condition and results of operations. Complying with new regulatory requirements and changes in the laws and regulations will increase our compliance cost and exposure to potential liability.
Additionally, the collection and use of health data in the EU is governed by the General Data Protection Regulation, or GDPR, which extends the geographical scope of EU data protection law to non-EU entities under certain conditions and imposes substantial obligations upon companies and new rights for individuals. Further, state and foreign laws may apply generally to the privacy and security of information we maintain, and may differ from each other in significant ways, thus complicating compliance efforts, as discussed in the risk factor below titled, “Data collection is governed by restrictive regulations governing the use, processing and cross-border transfer of personal information, and actual or perceived failures to comply with applicable data protection, privacy and security laws, regulations, standards and other requirements could adversely affect our business, results of operations and financial condition.”
Data collection is governed by restrictive regulations governing the use, processing and cross-border transfer of personal information, and actual or perceived failures to comply with applicable data protection, privacy and security laws, regulations, standards and other requirements could adversely affect our business, results of operations and financial condition.
The global data protection landscape is rapidly evolving, and we are or may become subject to numerous state, federal and foreign laws, regulations, and other actual and asserted obligations governing the collection, use, disclosure, retention, and security of personal information, such as information collected or otherwise processed in connection with clinical trials in the U.S. and abroad. Implementation standards and enforcement practices are likely to remain uncertain for the foreseeable future, and we cannot yet determine the impact future laws, regulations, standards, or other actual or asserted obligations, or any perception of their requirements may have on our business. This evolution may create uncertainty in our business, affect our ability to operate in certain jurisdictions or to collect, store, transfer use and share personal information, necessitate the acceptance of more onerous obligations in our contracts, result in liability or impose additional costs on us. The cost of compliance with these laws, regulations, standards, and obligations is high and is likely to increase in the future. Any failure or perceived failure by us to comply with federal, state or foreign laws or regulations, our internal policies and procedures, our contractual obligations governing our processing of personal information, or any other standards or other actual or asserted obligations, could result in negative publicity, government investigations and enforcement actions, claims by third parties and damage to our reputation, any of which could have a material adverse effect on our business, results of operation, and financial condition.
In conducting and/or enrolling patients in our current or future clinical trials, we are subject to restrictions relating to privacy, data protection and data security and may be subject to additional restrictions as our clinical operations expand. For example, the collection, use, storage, disclosure, transfer, or other processing of personal data regarding individuals in the European Economic Area, or EEA, including personal health data, is subject to the GDPR. The GDPR is wide-ranging in scope and imposes numerous requirements on companies that process personal data, including requirements relating to processing health and other sensitive data, obtaining consent of the individuals to whom the personal data relates, providing information to individuals regarding data processing activities, implementing safeguards to protect the security and confidentiality of personal data, providing notification of data breaches (initially to supervisory authorities and, if the breach is serious enough, to
individuals), and taking certain measures when engaging third-party processors. The GDPR also imposes strict rules on the transfer of personal data to countries outside the EEA, including the United States, and permits data protection authorities to impose large penalties for violations of the GDPR, including potential fines of up to €20 million or 4% of annual global revenues, whichever is greater, for the most serious of violations. The GDPR also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory authorities, seek judicial remedies, and obtain compensation for damages resulting from violations of the GDPR. In addition, the GDPR includes restrictions on cross-border data transfers. Certain aspects of cross-border data transfers under the GDPR are uncertain as the result of legal proceedings in the EU, including a July 2020 decision by the Court of Justice for the European Union, or CJEU, that invalidated the EU-U.S. Privacy Shield and, to some extent, called into question the efficacy and legality of using standard contractual clauses approved by the European Commission, or SCCs. To address certain concerns of the CJEU, the European Commission issued revised SCCs in June 2021 that are required to be implemented. Regulatory guidance and other developments relating to cross-border personal data transfers, including the necessity of putting in place those revised SCCs and UK SCCs, as discussed below, may increase the complexity of transferring personal data across borders and may require us to engage in additional contractual negotiations and to modify our policies and practices relating to the transfer and other processing of personal data. The GDPR increased our responsibility and liability in relation to personal data that we process where such processing is subject to the GDPR, and we may be required to put in place additional mechanisms to ensure compliance with the GDPR, including as implemented by individual countries.
In the United Kingdom, or UK, the Data Protection Act of 2018 is effective along with a version of the GDPR referred to as the UK GDPR. Collectively, the Data Protection Act of 2018 and the UK GDPR authorize significant fines, up to the greater of £17.5 million or 4% of global turnover, and expose us to two parallel regimes and other potentially divergent enforcement actions for certain violations. Further, aspects of data protection in the UK remain uncertain. On June 28, 2021, the European Commission issued an adequacy decision under the GDPR and the Law Enforcement Directive, pursuant to which personal data generally may be transferred from the EU to the UK without restriction; however, this adequacy decision is subject to a four-year “sunset” period, after which the European Commission’s adequacy decision may be renewed, and this decision may be revoked or modified in the interim. Additionally, on February 2, 2022, the UK’s Information Commissioner’s Office issued new standard contractual clauses to support personal data transfers out of the UK, or the UK SCCs. The UK SCCs became effective March 21, 2022 and, similar to the EU SCCs, are required to be implemented. We may, in addition to other impacts, experience additional costs associated with increased compliance burdens and be required to engage in new contract negotiations with third parties that aid in processing personal data on our behalf or localize certain personal data.
Other jurisdictions also increasingly maintain laws and regulations addressing privacy, data protection, and information security. We may incur liabilities, expenses, costs, and other operational losses under GDPR and local laws of applicable EU member states, Switzerland, the UK, and other regions in connection with any measures we take to comply with them. Working to comply with the GDPR and other laws and regulations to which we are subject in Europe and other regions outside the United States relating to privacy, data protection, and information security will be a rigorous and time-intensive process that may increase our cost of doing business or require us to change our business practices, and despite those efforts, there is a risk that we may be subject to fines and penalties, litigation, and reputational harm in connection with our activities in those regions.
In addition, in California, the California Consumer Privacy Act, or CCPA creates new individual privacy rights for California consumers (as defined in the law) and places increased privacy and security obligations on entities handling personal data of consumers or households. The CCPA requires covered companies to provide new disclosure to consumers about such companies’ data collection, use and sharing practices, provide such consumers new ways to opt-out of certain sales or transfers of personal information, and provide consumers with additional causes of action in data breach situations. While it exempts some data regulated by HIPAA and certain clinical trial data, the CCPA, to the extent applicable to our business and operations, may increase our compliance costs and potential liability with respect to other personal information we collect about California residents. The CCPA went into effect on January 1, 2020, and the California Attorney General commenced enforcement actions for violations on July 1, 2020. Moreover, the California Privacy Rights Act, or CPRA was approved by California voters in the November 3, 2020 election. The CPRA significantly modified the CCPA, creating obligations beginning on January 1, 2022, with enforcement anticipated to commence July 1, 2023. The CPRA creates further uncertainty and may require us to incur additional costs and expenses. The CCPA and CPRA could mark the beginning of a trend toward more stringent privacy legislation in the United States. The CCPA has prompted a number of proposals for federal and state privacy legislation. For example, in March 2021, Virginia enacted the Virginia Consumer Data Protection Act, or CDPA, a comprehensive privacy statute that became effective on January 1, 2023. In addition, on July 7, 2021, Colorado enacted the Colorado Privacy Act, or CPA, which becomes effective July 1, 2023, on March 24, 2022, Utah enacted the Utah Consumer Privacy Act, or UCPA, which becomes effective December 31, 2023, and on May 10, 2022, Connecticut enacted the Act Concerning Personal Data Privacy and Online Monitoring, also known as the Connecticut Data Privacy Act or CTDPA, which becomes effective July 1, 2023. The CDPA, CPA, UCPA, and CTDPA are comprehensive privacy statutes that share similarities with the CCPA and
CPRA. The U.S. federal government also is contemplating federal privacy legislation. These and other new laws that may be proposed or enacted could increase our potential liability and adversely affect our business.
Compliance with U.S. and international data protection laws and regulations could require us to take on more onerous obligations in our contracts, restrict our ability to collect, use and disclose data, or in some cases, impact our ability to operate in certain jurisdictions. Any actual or alleged failure to comply with U.S. or international laws and regulations relating to privacy, data protection, and data security could result in governmental investigations, proceedings and enforcement actions (which could include civil or criminal penalties), private litigation or adverse publicity, harm to our reputation, and could negatively affect our operating results and business. Moreover, clinical trial subjects about whom we or our potential collaborators obtain information, as well as the providers who share this information with us, may contractually limit our ability to use and disclose the information or impose other obligations or restrictions in connection with our use, retention and other processing of information, and we may otherwise face contractual restrictions applicable to our use, retention, and other processing of information. Claims that we have violated individuals’ privacy rights, failed to comply with data protection laws, or breached our contractual obligations, even if we are not found liable, could be expensive and time-consuming to defend and could result in adverse publicity that could harm our business.
Our ability to obtain services, reimbursement, or funding from the federal government may be impacted by possible reductions in federal spending.
The U.S. federal budget remains in flux and could, among other things, cut Medicare payments to providers. The Medicare program is frequently mentioned as a target for spending cuts. The full impact on our business of any future cuts in Medicare or other programs is uncertain. In addition, weWe cannot predict any impact President Trump’sthe extent of legislative, executive, and administrative actions of the Biden administration and Congress maywill have on us and the federal budget.biopharmaceutical industry as a whole. If federal spending is reduced, anticipated budgetary shortfalls may also impact the ability of relevant agencies such as the FDA or the National Institutes of Health to continue to function at current levels. Amounts allocated to federal grants and contracts may be reduced or eliminated. These reductions may also impact the ability of relevant agencies to timely review and approve drug research and development, manufacturing, and marketing activities, which may delay our ability to develop, market, and sell any therapeutics we may develop.
If any of our therapeutic candidates receives marketing approval and we or others later identify undesirable side effects caused by the therapeutic product, our ability to market and derive revenue from the therapeutic products could be compromised.
In the event any of our therapeutic candidates receive regulatory approval and we or others identify undesirable side effects, adverse events, or other problems caused by one of our therapeutics, any of the following adverse events could occur, which could result in the loss of significant revenue to us and materially and adversely affect our results of operations and business:
•regulatory authorities may withdraw their approval of the product and require us to take the product off the market or seize the product;
| |
•
| regulatory authorities may withdraw their approval of the product and require us to take the product off the market or seize the product;
|
| |
•
| we may need to recall the therapeutic or change the way the therapeutic is administered to patients;
|
| |
•
| additional restrictions may be imposed on the marketing and promotion of the particular therapeutic or the manufacturing processes for the therapeutic or any component thereof;
|
| |
•
| we may not be able to secure or maintain adequate coverage and reimbursement for our proprietary therapeutic products from government (including U.S. federal health care programs) and private payors;
|
| |
•
| we may lose or see adverse alterations to compendia listings or treatment protocols specified by accountable care organizations;
|
| |
•
| we may be subject to fines, restitution, or disgorgement of profits or revenues, injunctions, or the imposition of civil penalties or criminal prosecution;
|
| |
•
| regulatory authorities may require the addition of labeling statements, such as a “black box” warning, or equivalent, or a contraindication;
|
| |
•
| regulatory authorities may require us to implement a REMS plan, or to conduct post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of the product;
|
| |
•
| we may be required to create a Medication Guide outlining the risks of such side effects for distribution to patients;
|
| |
•
| we could be sued and held liable for harm caused to patients;
|
| |
•
| the therapeutic may become less competitive; and
|
| |
•
| our reputation may suffer.
|
•we may need to recall the therapeutic or change the way the therapeutic is administered to patients;
•additional restrictions may be imposed on the marketing and promotion of the particular therapeutic or the manufacturing processes for the therapeutic or any component thereof;
•we may not be able to secure or maintain adequate coverage and reimbursement for our proprietary therapeutic products from government (including U.S. federal health care programs) and private payors;
•we may lose or see adverse alterations to compendia listings or treatment protocols specified by ACOs;
•we may be subject to fines, restitution, or disgorgement of profits or revenues, injunctions, or the imposition of civil penalties or criminal prosecution;
•regulatory authorities may require the addition of labeling statements, such as a “black box” warning, or equivalent, or a contraindication;
•regulatory authorities may require us to implement a REMS plan, or to conduct post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of the product;
•we may be required to create a Medication Guide outlining the risks of such side effects for distribution to patients;
•we could be sued and held liable for harm caused to patients;
•the therapeutic may become less competitive; and
•our reputation may suffer.
Our therapeutic candidates for which we intend to seek approval as biologic products may face competition sooner than anticipated.
The Patient Protection and Affordable Care Act,ACA, signed into law in 2010, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, or BPCIA, which created an abbreviated approval pathway for biological products that are biosimilar to or interchangeable with an FDA-licensed reference biological product. Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date that the reference product was first licensed by the FDA. In addition, the approval of a biosimilar product may not be made effective by the FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing the sponsor’ssponsor’s own pre-clinicalpreclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product. The law is complex and is still being interpreted and implemented by the FDA. As a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when such processes intended to implement BPCIA may be fully adopted by the FDA, any such processes could have a material adverse effect on the future commercial prospects for our biological products.
We believe that any of our therapeutic candidates approved as a biological product under a BLA should qualify for the 12-year period of exclusivity. However, there is a risk that this exclusivity could be shortened due to congressional action or otherwise, or that the FDA will not consider our therapeutic candidates to be reference products for competing products, potentially creating the opportunity for generic competition sooner than anticipated. Other aspects of the BPCIA, some of which may impact the BPCIA exclusivity provisions, have also been the subject of recent litigation. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of our reference products in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing.
Significant developments stemming from the United Kingdom’s recent withdrawal from the European Union could have a material adverse effect on us.
In June 2016, the United Kingdom held a referendum and voted in favor of leaving the European Union, and in January 2020, the United Kingdom officially left the European Union, with a transitional period scheduled to end on December 31, 2020. The United Kingdom’s withdrawal from the European Union and ongoing negotiations related to the United Kingdom’s future trade and other relationships with the European Union have created political and economic uncertainty, particularly in the United Kingdom and the European Union. Any business we conduct, now and in the future, in the United Kingdom, the European Union, and worldwide could be affected during this period of uncertainty, and perhaps longer, by the impact of the United Kingdom’s decision to withdraw from the European Union. There are many ways in which our business could be affected, only some of which we can identify as of the date of this filing.
The decision of the United Kingdom to withdraw from the European Union has caused and, along with events that could occur in the future as a consequence of the United Kingdom’s withdrawal, may continue to cause significant volatility in global financial markets, including in global currency and debt markets. This volatility could cause a slowdown in economic activity in the United Kingdom, Europe, or globally, which could adversely affect our operating results and growth prospects. In addition, our business could be negatively affected by new trade agreements or data transfer agreements between the United Kingdom and other countries, including the United States, and by the possible imposition of trade or other regulatory barriers in the United Kingdom.
It is currently unknown how regulations affecting clinical trials, the approval of our future products, and the sale of these products in the United Kingdom or elsewhere in Europe will be affected by the United Kingdom’s withdrawal from the European Union.
These possible negative impacts, and others resulting from the United Kingdom’s withdrawal from the European Union, may adversely affect our operating results and growth prospects.
Risks Related to Ownership of Our Common Stock
Our stock price may be volatile, and an active, liquid, and orderly trading market may not develop for our common stock. As a result, stockholders may not be able to resell shares at or above their purchase price.
Although our common stock is listed on the Nasdaq Global Market, an active trading market for our common stock may not develop or, if it develops, may not be sustained. The lack of an active market may impair the ability of our stockholders to sell their shares at the time they wish to sell them or at a price that they consider reasonable, which may reduce the fair market value of their shares. Further, an inactive market may also impair our ability to raise capital by selling our common stock should we determine additional funding is required.
The market price of our common stock could be subject to significant fluctuations. Market prices for securities of early-stage pharmaceutical, biotechnology, and other life sciences companies have historically been particularly volatile. Some of the factors that may cause the market price of our common stock to fluctuate include:
•our and our collaborators’ ability to obtain regulatory approvals for product candidates, and delays or failures to obtain such approvals;
| |
| our ability to obtain regulatory approvals for product candidates, and delays or failures to obtain such approvals;
|
| |
•
| the failure of any of our product candidates, if approved, to achieve commercial success; •issues in manufacturing our approved products, if any, or product candidates; •the results of current, and any future, preclinical or clinical trials of our product candidates; •our ability to achieve development milestones and receive associated milestone payments pursuant to the terms of our collaboration agreements; •the entry into, or termination of, key agreements, including key licensing, collaboration or acquisition agreements; •the initiation of, any of our product candidates, if approved, to achieve commercial success; |
| |
•
| issues in manufacturing our approved products, if any, or product candidates;
|
| |
•
| the results of current, and any future, preclinical or clinical trials of our product candidates;
|
| |
•
| the entry into, or termination of, key agreements, including key licensing, collaboration or acquisition agreements;
|
| |
•
| the initiation or material developments in, or conclusion of, litigation to enforce or defend any of our intellectual property rights or defend against the intellectual property rights of others;
|
| |
•
| announcements by commercial partners or competitors of new commercial products, clinical progress (or the lack thereof), significant contracts, commercial relationships, or capital commitments;
|
| |
•
| adverse publicity relating to our markets, including with respect to other products and potential products in such markets;
|
| |
•
| adverse publicity about our company, employees, therapeutic candidates, and/or therapeutic products in the media or on social media;
|
| |
•
| the impact of COVID-19 on our company or the economy generally;
|
| |
••announcements by commercial partners or competitors of new commercial products, clinical progress (or the lack thereof), significant contracts, commercial relationships, or capital commitments;
| the introduction of technological innovations or new therapies competing with our potential products;
|
| |
•
| the loss of key employees;
|
| |
•
| changes in estimates or recommendations by securities analysts, if any, who cover our common stock;
|
| |
•
| general and industry-specific economic conditions potentially affecting our research and development expenditures;
|
| |
•
| changes in the structure of health care payment systems;
|
| |
•
| unanticipated serious safety concerns related to the use of any of our product candidates;
|
| |
•
| failure to meet or exceed financial and development projections we may provide to the public;
|
| |
•
| failure to meet or exceed the financial and development projections of the investment community;
|
| |
•
| the perception of the pharmaceutical industry by the public, legislators, regulators, and the investment community;
|
| |
•
| adverse regulatory decisions;
|
| |
•
| disputes or other developments relating to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies;
|
| |
•
| commencement of, or our involvement in, litigation;
|
| |
•
| sales of our common stock by us or our stockholders in the future;
|
| |
•
| trading volume of our common stock;
|
| |
•
| period-to-period fluctuations in our financial results; and
|
| |
•
| the other factors described in this “Risk Factors” section.
|
•adverse publicity relating to our markets, including with respect to other products and potential products in such markets;
•adverse publicity about our company, employees, therapeutic candidates, and/or therapeutic products in the media or on social media;
•the impact of COVID-19 on our company or the economy generally;
•the introduction of technological innovations or new therapies competing with our potential products;
•the loss of key employees;
•changes in estimates or recommendations by securities analysts, if any, who cover our common stock;
•general and industry-specific economic conditions potentially affecting our research and development expenditures;
•changes in the structure of health care payment systems;
•unanticipated serious safety concerns related to the use of any of our product candidates;
•failure to meet or exceed financial and development projections we may provide to the public;
•failure to meet or exceed the financial and development projections of the investment community;
•the perception of the pharmaceutical industry by the public, legislators, regulators, and the investment community;
•adverse regulatory decisions;
•disputes or other developments relating to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies;
•threats of, commencement of, or our involvement in, litigation;
•sales of our common stock by us or our stockholders in the future;
•trading volume of our common stock;
•additional instability in the domestic or global banking system;
•period-to-period fluctuations in our financial results; and
•the other factors described in this “Risk Factors” section. Moreover, the stock markets in general have experienced substantial volatility that has often been unrelated to the operating performance of individual companies or the biotechnology sector. These broad market fluctuations may also adversely affect the trading price of our common stock.
In the past, following periods of volatility in the market price of a company’scompany’s securities, stockholders have often instituted class action securities litigation against those companies. Such litigation, if instituted, could result in substantial costs and diversion of management attention and resources, which could significantly harm our business and reputation.
Our officers and directors, and their respective affiliates, have a controllingsignificant influence over our business affairs and may make business decisions with which stockholders disagree and which may adversely affect the value of their investment.
Our executive officers and directors together with their respective affiliates, beneficially own approximately 76%43% of our common stock as of December 31, 2019.2022. As a result, if some of these persons or entities act together, they will have the ability to exercise significant influence over matters submitted to the stockholders for approval, including the election of directors, amendments to the certificate of incorporation and bylaws and the approval of any strategic transaction requiring the approval of the stockholders. These actions may be taken even if they are opposed by other stockholders. This concentration of ownership may also have the effect of delaying or preventing a change of control of our company or discouraging others from making tender offers for our shares, which could prevent our stockholders from receiving a premium for their shares. Some of these persons or entities who make up our principal stockholders may have interests different from other stockholders. The significant concentration of stock ownership may adversely affect the trading price of our common stock due to investors’investors’ perception that conflicts of interest may exist or arise.
Future sales, or the perception of future sales, of a substantial amount of our common stock could depress the trading price of our common stock.
Our stock price could decline as a result of sales of a large number of shares of our common stock or the perception that these sales could occur. These sales, or the possibility that these sales may occur, also might make it more difficult for us to sell equity securities in the future at a time and at a price that we deem appropriate.
For example, in connection with our January 2019July 2020 private placement, we entered into a registration rights agreement with the private placement investors that required us to prepare and file a registration statement on March 18, 2019. The resale registration statement, which was declared effective by the SEC on April 4, 2019August 18, 2020 and permits the resale by the private placement
investors of approximately 4.75.1 million shares of our common stock as well as approximately 1.82.6 million shares of common stock issuable upon the exercise of prefunded warrants and warrants issued in the July 2020 private placement. Additionally, in connection with our September 2021 private placement, we entered into a registration rights agreement with the private placement investors that required us to prepare and file a resale registration statement, which was declared effective by the SEC on November 19, 2021 and permits the resale by the private placement investors of approximately 6.5 million shares of our common stock as well as approximately 3.2 million shares of common stock issuable upon the exercise of prefunded warrants issued in the September 2021 private placement. We have in the past and may again in the future sell shares of our common stock to strategic partners in connection with collaboration agreements, as we did in December 2021 in connection with our agreement with Horizon. The shares subject to outstanding options and warrants, of which options and warrants (including prefunded warrants) to purchase 1.54.0 million shares and 1.98.8 million shares, respectively, were exercisable as of December 31, 2019,2022, and the shares reserved for future issuance under our equity incentive plans will become available for sale immediately upon the exercise of such options.
We also register the offer and sale of all shares of common stock that we may issue under our equity incentive plans. Once we register the offer and sale of shares for the holders of registration rights and option holders, they can be freely sold in the public market upon issuance, subject to any related lock-up agreements or applicable securities laws.
In addition, in the future, we may issue additional shares of common stock or other equity or debt securities convertible into common stock in connection with a financing, acquisition, litigation settlement, employee arrangements or otherwise. Any such future issuance including any issuances pursuant to our “at the market” equity offering program, could result in substantial dilution to our existing stockholders and could cause our stock price to decline.
We have broad discretion over the use of the proceeds to us from our financing activities and may apply the proceeds to uses that do not improve our operating results or the value of your securities.
We have broad discretion over the use of proceeds to us from our financing activities and our stockholders rely solely on the judgment of our board of directors and management regarding the application of these proceeds. Our use of proceeds may not improve our operating results or increase the value of our common stock. Any failure to apply these proceeds effectively could have a material adverse effect on our business, delay the development of our product candidates and cause the market price of our common stock to decline.
Anti-takeover provisions in our charter documents and under Delaware or Washington law could discourage, delay or prevent a change in control of our company, limit attempts by our stockholders to replace or remove our current management and may affect the trading price of our common stock.
Our corporate documents contain provisions that may delay or discourage transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders might otherwise receive a premium for their shares, or transactions that our stockholders might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our stock. Among other things, our certificate of incorporation and bylaws:
•stagger the terms of our board of directors and require 66 and 2/3% stockholder voting to remove directors, who may only be removed for cause;
The JOBS•provide that the authorized number of directors may be changed only by resolution of the board of directors;
•provide that all vacancies, including newly-created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum;
•authorize our board of directors to issue “blank check” preferred stock and to determine the rights and preferences of those shares, which may be senior to our common stock, without prior stockholder approval;
•establish advance notice requirements for nominating directors and proposing matters to be voted on by stockholders at stockholders’ meetings;
•prohibit our stockholders from calling a special meeting and prohibit stockholders from acting by written consent;
•require 66 and 2/3% stockholder voting to effect certain amendments to our certificate of incorporation and bylaws; and
•prohibit cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates.
In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, or DGCL, which generally prohibits a Delaware corporation from engaging in any of a broad range of business combinations with any “interested” stockholder for a period of three years following the date on which the stockholder became an “interested” stockholder. Likewise, because our principal executive offices are located in Washington, the anti-takeover provisions of the Washington Business Corporation Act allowsmay apply to us under certain circumstances now or in the future. These provisions prohibit a “target corporation” from engaging in any of a broad range of business combinations with any stockholder constituting an “acquiring person” for a period of five years following the date on which the stockholder became an “acquiring person.” These provisions could discourage, delay or prevent a transaction involving a change in control of our company. These provisions could also discourage proxy contests and make it more difficult for stockholders to elect directors of their choosing and cause us to postpone the datetake other corporate actions our stockholders desire.
Claims for indemnification by which we must comply with certain lawsour directors and regulations intendedofficers may reduce our available funds to protect investorssatisfy successful third-party claims against us and tomay reduce the amount of informationavailable cash.
Our amended and restated certificate of incorporation provides that we will indemnify our directors to the fullest extent permitted by Delaware law.
In addition, as permitted by Section 145 of the DGCL, our amended and restated bylaws and our indemnification agreements that we have entered into with our directors and officers provide that:
•We will indemnify our directors and officers for serving us in those capacities or for serving other business enterprises at our request, to the fullest extent permitted by Delaware law. Delaware law provides that a corporation may indemnify such person if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of the corporation and, with respect to any criminal proceeding, had no reasonable cause to believe such person’s conduct was unlawful.
•We may, in our reports fileddiscretion, indemnify other employees and agents in those circumstances where indemnification is permitted by applicable law.
•We are required to advance expenses, as incurred, to our directors and officers in connection with defending a proceeding, unless the SEC. proceeding is excluded pursuant to the amended and restated bylaws, except that such directors or officers shall undertake to repay such advances if it is ultimately determined that such person is not entitled to indemnification.
•We cannotwill not be obligated pursuant to our amended and restated bylaws to indemnify any director or officer in connection with any proceeding (or part thereof) (a) for which payment has actually been made to such person, (b) for an accounting or disgorgement of profits pursuant to Section 16(b) of the Exchange Act, (c) for any reimbursement of the Company by such person of any bonus or other incentive-based or equity-based compensation, (d) initiated by such person unless the proceeding was authorized in the specific case by our board of directors or such indemnification is required to be made pursuant to our amended and restated bylaws or applicable law, nor (e) if prohibited by applicable law.
•The rights conferred in our amended and restated bylaws are not exclusive, and we are authorized to enter into indemnification agreements with our directors, officers, employees and agents and to obtain insurance to indemnify such persons.
•We may not retroactively amend our amended and restated bylaw provisions to reduce our indemnification obligations to our directors or officers.
As a result, if we are required to indemnify one or more of our directors or officers, it may reduce our available funds to satisfy successful third-party claims against us, may reduce the amount of available cash and may have a material adverse effect on our business and financial condition.
Our amended and restated certificate of incorporation and our amended and restated bylaws designate the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be
initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers, employees or agents.
Our amended and restated certificate of incorporation and/or our amended and restated bylaws provide that, unless we consent in writing to an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for any derivative action or proceeding brought on our behalf, any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers, stockholders, employees or agents to us or our stockholders, any action asserting a claim arising pursuant to any provision of the DGCL, our amended and restated certificate of incorporation or our amended and restated bylaws or any action asserting a claim that is governed by the internal affairs doctrine, in each case subject to the Court of Chancery having personal jurisdiction over the indispensable parties named as defendants therein and the claim not being one which is vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery or for which the Court of Chancery does not have subject matter jurisdiction. Any person purchasing or otherwise acquiring any interest in any shares of our common stock shall be deemed to have notice of and to have consented to this provision of our amended and restated certificate of incorporation. In addition, our amended and restated bylaws provide that the U.S. federal district courts will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act.
These choice of forum provision may limit our stockholders’ ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, employees or agents, which may discourage such lawsuits against us and our directors, officers, employees and agents even though an action, if thissuccessful, might benefit our stockholders. Stockholders who do bring a claim in the Court of Chancery could face additional litigation costs in pursuing any such claim, particularly if they do not reside in or near Delaware. The Court of Chancery may also reach different judgments or results than would other courts, including courts where a stockholder considering an action may be located or would otherwise choose to bring the action, and such judgments or results may be more favorable to us than to our stockholders. Alternatively, if a court were to find these choice of forum provisions in our amended and restated certificate of incorporation and amended and restated bylaws inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could have a material adverse effect on our business, financial condition or results of operations.
We do not expect to pay any dividends on our common stock for the foreseeable future.
We currently expect to retain all future earnings, if any, for future operations and expansion, and have no current plans to pay any cash dividends to holders of our common stock for the foreseeable future. Any decision to declare and pay dividends in the future will be made at the discretion of our board of directors and will depend on, among other things, our results of operations, financial condition, cash requirements, contractual restrictions and other factors that our board of directors may deem relevant. As a result, stockholders may not receive any return on an investment in our common stock unless stockholders sell our common stock for a price greater than that which they paid for it.
The Nasdaq Global Market may delist our common stock from its exchange, which could limit investors’ ability to make transactions in our securities and subject us to additional trading restrictions.
Our shares of common stock are listed on the Nasdaq Global Market under the trading symbol “ALPN.” Our securities may fail to meet the continued listing requirements to be listed on the Nasdaq Global Market. If Nasdaq delists our shares of common stock from trading on its exchange, we could face significant material adverse consequences, including:
•significant impairment of the liquidity for our common stock, which may substantially decrease the market price of our common stock;
•a limited availability of market quotations for our securities;
•a determination that our common stock qualifies as a “penny stock” which will require brokers trading in our common stock to adhere to more stringent rules and possibly resulting in a reduced level of trading activity in the secondary trading market for our common stock;
•a limited amount of news and analyst coverage for our company; and
•a decreased ability to issue additional securities or obtain additional financing in the future.
Risks Related to Our Financial Reporting and Disclosure
We are a smaller reporting company, and any decision on our part to comply only with reduced reporting and disclosure willrequirements applicable to such companies could make our common stock less attractive to investors.
The JOBS Act is intended to reduce the regulatory burden on “emerging growth companies.” AsWe are a “smaller reporting company,” as defined in the JOBSSecurities Exchange Act of 1934, as amended, or the Exchange Act. For as long as we qualify as an “emerging growthcontinue to be a smaller reporting company,” we may choose to take advantage of exemptions from various reporting requirements applicable to other public companies that are not smaller reporting companies, including, but not limited to, reduced disclosure obligations regarding executive compensation in our periodic reports and couldproxy statements.
We will remain an “emerging growtha smaller reporting company” until as late as December 31, 2020. For so long as, as of June 30 of the preceding year, (i) the market value of our shares of common stock held by non-affiliates, or our public float, is less than $250 million; or (ii) we are an “emerging growth company,”have annual revenues less than $100 million and either we will, among other things:
| |
•
| not be required to comply with the auditor attestation requirements of Section 404(b) of Sarbanes-Oxley;
|
| |
•
| not be required to hold a nonbinding advisory stockholder vote on executive compensation pursuant to Section 14A of the Securities Exchange Act of 1934, as amended, or the Exchange Act;
|
| |
•
| not be required to seek stockholder approval of any golden parachute payments not previously approved pursuant to Section 14A of the Exchange Act;
|
| |
•
| be exempt from any rule adopted by the Public Company Accounting Oversight Board, requiring mandatory audit firm rotation or a supplemental auditor discussion and analysis; and
|
| |
•
| be subject to reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements.
|
We have previously decided to opt out of an extended transition period under the JOBS Act that permits an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. Our decisionno public float or our public float is irrevocable. As a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other companies.less than $700 million.
Furthermore, ifIf we take advantage of some or all of the reduced disclosure requirements above,available to smaller reporting companies, investors may find our common stock less attractive, which may result in a less active trading market for our common stock and greater stock price volatility. For so long as we are a smaller reporting company and not classified as an “accelerated filer” or “large accelerated filer” pursuant to SEC rules, we will continue to be exempt from the auditor attestation requirements of Section 404(b) of Sarbanes-Oxley.
If we fail to maintain proper and effective internal controls, our ability to produce accurate financial statements on a timely basis could be impaired.
We are subject to the reporting requirements of the Exchange Act, the Sarbanes-Oxley Act and the rules and regulations of The Nasdaq Stock Market LLC. The Sarbanes-Oxley Act requires, among other things, that we maintain effective disclosure controls and procedures and internal control over financial reporting. We must perform system and process evaluation and testing of our internal control over financial reporting to allow management to report on the effectiveness of our internal controls over financial reporting in our Annual Report on Form 10-K filing for that year, as required by Section 404 of the Sarbanes-Oxley Act.
We may discover weaknesses in our system of internal financial and accounting controls and procedures that could result in a material misstatement of our financial statements. Our internal control over financial reporting will not prevent or detect all errors and all fraud. An internal control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the internal control system’ssystem’s objectives will be met. Because of the inherent limitations in all internal control systems, no evaluation of internal controls can provide absolute assurance that misstatements due to error or fraud will not occur or that all internal control issues and instances of fraud will be detected.
If we are not able to comply with the requirements of Section 404 of the Sarbanes-Oxley Act, or if we are unable to maintain proper and effective internal controls, we may not be able to produce timely and accurate financial statements. If that were to happen, the market price of our common stock could decline and we could be subject to sanctions or investigations by The Nasdaq Stock Market LLC, the SEC, or other regulatory authorities.
Our disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
Our disclosure controls and procedures are designed to reasonably ensure that information required to be disclosed by us in reports we file or submits under the Exchange Act is accumulated and communicated to management and recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls and procedures as well as internal controls and procedures, no matter how well-conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are and will be met. These inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements due to error or fraud may occur and not be detected.
We will continue to incur costs and demands upon management as a result of complying with the laws and regulations affecting public companies.
We will incur significant legal, accounting, and other expenses associated with public company reporting requirements. We will also incur costs associated with corporate governance requirements, including requirements under the Sarbanes-Oxley Act, as well as new rules implemented by the SEC and The Nasdaq Stock Market LLC. Although the JOBS Actour status as a smaller reporting company may for a limited period of time somewhat lessen the cost of complying with these additional regulatory and other requirements, we nonetheless expect that these rules and regulations will increase our legal and financial compliance costs and to make some activities more time-consuming and costlier. For example, our management team consists in part of the executive officers of Alpine prior to the merger, some of whom may not have previously managed and operated a public company. TheseOur executive officers and other personnel will need to devote substantial time to gaining expertise regardingoversee our operations as a public company and compliance with applicable laws and regulations. These rules and regulations may also make it difficult and expensive for us to obtain directors and officer’sofficer’s liability insurance. As a result, it may be more difficult for us to attract and retain qualified individuals to serve on our board of directors or as executive officers of our company, which may adversely affect investor confidence in us and could cause our business or stock price to suffer.
Anti-takeover provisions
General Risk Factors
Changes in accounting rules and regulations, or interpretations thereof, could result in unfavorable accounting charges or require us to change our charter documentscompensation policies.
Accounting methods and under Delawarepolicies for biopharmaceutical companies, including policies governing revenue recognition, research and development and related expenses, and accounting for stock-based compensation, are subject to review, interpretation, and guidance from our auditors and relevant accounting authorities, including the SEC. Changes to accounting methods or Washington law could discourage, delaypolicies, or preventinterpretations thereof, may require us to reclassify, restate, or otherwise change or revise our financial statements.
Environmental, social and governance matters may impact our business and reputation.
Companies are increasingly being judged by their performance on a change in controlvariety of our company, limit attempts by our stockholdersenvironmental, social and governance, or ESG, matters, which are considered to replace or remove our current managementcontribute to the long-term sustainability of companies’ performance.
A variety of organizations measure the performance of companies on such ESG topics, and may affect the trading priceresults of our common stock.
Our corporate documents contain provisions that may delay or discourage transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders might otherwise receive a premium for their shares, or transactions that our stockholders might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our stock. Among other things, our certificate of incorporation and bylaws:
| |
•
| stagger the terms of our board of directors and require 66 and 2/3% stockholder voting to remove directors, who may only be removed for cause;
|
| |
•
| provide that the authorized number of directors may be changed only by resolution of the board of directors;
|
| |
•
| provide that all vacancies, including newly-created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum;
|
| |
•
| authorize our board of directors to issue “blank check” preferred stock and to determine the rights and preferences of those shares, which may be senior to our common stock, without prior stockholder approval;
|
| |
•
| establish advance notice requirements for nominating directors and proposing matters to be voted on by stockholders at stockholders’ meetings;
|
| |
•
| prohibit our stockholders from calling a special meeting and prohibit stockholders from acting by written consent;
|
| |
•
| require 66 and 2/3% stockholder voting to effect certain amendments to our certificate of incorporation and bylaws; and
|
| |
•
| prohibit cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates.
|
assessments are widely publicized. In addition, because weinvestment in funds that specialize in companies that perform well in such assessments are incorporatedincreasingly popular, and major institutional investors have publicly emphasized the importance of such ESG measures to their investment decisions. Topics considered in Delaware, we are governed bysuch assessments include, among others, the provisions of Section 203role of the Delaware General Corporation Law, which generally prohibits a Delaware corporation from engagingcompany’s board of directors in anysupervising various ESG issues and board diversity.
In light of a broad range of business combinations with any “interested” stockholder for a period of three years following the dateinvestors’ increased focus on which the stockholder became an “interested” stockholder. Likewise, because our principal executive offices are located in Washington, the anti-takeover provisions of the Washington Business Corporation Act may apply to us under certain circumstances now or in the future. These provisions prohibit a “target corporation” from engaging in any of a broad range of business combinations with any stockholder constituting an “acquiring person” for a period of five years following the date on which the stockholder became an “acquiring person.” These provisions could discourage, delay or prevent a transaction involving a change in control of our company. These provisions could also discourage proxy contests and make it more difficult for stockholders to elect directors of their choosing and cause us to take other corporate actions our stockholders desire.
Claims for indemnification by our directors and officers may reduce our available funds to satisfy successful third-party claims against us and may reduce the amount of available cash.
Our amended and restated certificate of incorporation providesESG matters, there can be no certainty that we will indemnify our directors to the fullest extent permitted by Delaware law.
In addition, as permitted by Section 145 of the DGCL, our amended and restated bylaws and our indemnification agreementsmanage such issues successfully, or that we have entered into with our directors and officers provide that:
| |
•
| we will indemnify our directors and officers for serving us in those capacities or for serving other business enterprises at our request, to the fullest extent permitted by Delaware law. Delaware law provides that a corporation may indemnify such person if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of the corporation and, with respect to any criminal proceeding, had no reasonable cause to believe such person’s conduct was unlawful.
|
| |
•
| we may, in our discretion, indemnify other employees and agents in those circumstances where indemnification is permitted by applicable law.
|
| |
•
| we are required to advance expenses,will successfully meet expectations as incurred, to our directors and officers in connection with defending a proceeding, except that such directors or officers shall undertake to repay such advances if it is ultimately determined that such person is not entitled to indemnification.
|
| |
•
| we will not be obligated pursuant to our amended and restated bylaws to indemnify any director or officer in connection with any proceeding (or part thereof) initiated by such person unless the proceeding was authorized in the specific case by our board of directors or such indemnification is required to be made pursuant to our amended and restated bylaws.
|
| |
•
| the rights conferred in our amended and restated bylaws are not exclusive, and we are authorized to enter into indemnification agreements with our directors, officers, employees and agents and to obtain insurance to indemnify such persons.
|
| |
•
| we may not retroactively amend our amended and restated bylaw provisions to reduce our indemnification obligations to our directors or officers.
|
As a result, if we are required to indemnify one or more of our directors or officers, it may reduce our available funds to satisfy successful third-party claims against us, may reduce the amount of available cash and may have a material adverse effect on our business and financial condition.
Our amended and restated certificate of incorporation designates the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers, employees or agents.
Our amended and restated certificate of incorporation provides that, unless we consent in writing to an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for any derivative action or proceeding brought on our behalf, any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers, employees or agents to us or our stockholders, any action asserting a claim arising pursuant to any provision of the DGCL, our amended and restated certificate of incorporation or our amended and restated bylaws or any action asserting a claim that is governed by the internal affairs doctrine, in each case subject to the Court of Chancery having personal jurisdiction over the indispensable parties named as defendants therein and the claim not being one which is vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery or for which the Court of Chancery does not have subject matter jurisdiction. Any person purchasing or otherwise acquiring any interest in any shares of our common stock shall be deemed to have notice of and to have consented to this provision of our amended and restated certificate of incorporation. This choice of forum provision may limit our stockholders’ ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, employees or agents, which may discourage such lawsuits against us and our directors, officers, employees and agents even though an action, if successful, might benefit our stockholders. Stockholders who do bring a claim in the Court of Chancery could face additional litigation costs in pursuing any such claim, particularly if they do not reside in or near Delaware. The Court of Chancery may also reach different judgments or results than would other courts, including courts where a stockholder considering an action may be located or would otherwise choose to bring the action, and such judgments or results may be more favorable to us than to our stockholders. Alternatively, if a court were to findproper role. Any failure or perceived failure by us in this provision of our amended and restated certificate of incorporation inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, whichregard could have a material adverse effect on our reputation and on our business, share price, financial condition, or results of operations.
We do not expect to pay any dividends on our common stock foroperations, including the foreseeable future.
We currently expect to retain all future earnings, if any, for future operations and expansion, and have no current plans to pay any cash dividends to holderssustainability of our common stock for the foreseeable future. Any decision to declare and pay dividends in the future will be made at the discretion of our board of directors and will depend on, among other things, our results of operations, financial condition, cash requirements, contractual restrictions and other factors that our board of directors may deem relevant. As a result, stockholders may not receive any return on an investment in our common stock unless stockholders sell our common stock for a price greater than that which they paid for it.business over time.
If equity research analysts do not publish research or reports, or publish unfavorable research or reports, about us, our business or our market, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that equity research analysts publish about us and our business. Equity research analysts may elect not to provide research coverage of our common stock or discontinue existing research coverage, and such lack of research coverage may adversely affect the market price of our common stock. We do not have any control over the analysts, or the content and opinions included in their reports. The price of our common stock could decline if one or more equity research analysts downgrade our stock or issue other unfavorable commentary or research. If one or more equity research analysts ceases coverage of us or fails to publish reports regularly, demand for our common stock could decrease, which in turn could cause our stock price or trading volume to decline.
Nasdaq may delist our common stock from its exchange, which could limit investors’ ability to make transactions in our securities and subject us to additional trading restrictions.
Our common shares are listed on Nasdaq under the trading symbol “ALPN.” Our securities may fail to meet the continued listing requirements to be listed on Nasdaq. If Nasdaq delists our common shares from trading on its exchange, we could face significant material adverse consequences, including:
| |
•
| significant impairment of the liquidity for our common stock, which may substantially decrease the market price of our common stock;
|
| |
•
| a limited availability of market quotations for our securities;
|
| |
•
| a determination that our common stock qualifies as a “penny stock” which will require brokers trading in our common stock to adhere to more stringent rules and possibly resulting in a reduced level of trading activity in the secondary trading market for our common stock;
|
| |
•
| a limited amount of news and analyst coverage for our company; and
|
| |
•
| a decreased ability to issue additional securities or obtain additional financing in the future.
|
Item 1B. Unresolved Staff Comments.
None.
None.
Item 2. Properties.
We leased a facility containing our research and development, laboratory, and office space located at 201 Elliott Avenue West, Seattle, Washington. The lease expired on December 31, 2019.
In March 2019, we entered into a lease for 27,164 square feet of office and laboratory space located at 188 East Blaine Street, Seattle, Washington. The term of the lease is 10.8 years with one option to extend the term by 5.0 years. The lease term commenced in June 2019. We believe that our existing facility is adequate for our current needs as the facility has sufficient space to house additional personnel as we expand.
Item 3. Legal Proceedings.
We are not engaged in any material legal proceedings. From time to time, we may become involved in litigation relating to claims arising from the ordinary course of business. We believe that there are no claims or actions pending against us currently, the ultimate disposition of which would have a material adverse effect on our consolidated results of operation, financial condition or cash flows.flows.
Item 4. Mine Safety Disclosures.
Not applicable.applicable.
PART II
Item 5. Market for Registrant’sRegistrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market Information
From June 17, 2015 through July 24, 2017, our common stock was traded under the symbol “NVLS.”“NVLS.” On July 24, 2017, in connection with the business combination of Nivalis Therapeutics, Inc. and Alpine Immune Sciences, Inc., we completed a 1-for-4 reverse stock split. Commencing on July 25, 2017, our common stock began trading on The Nasdaq Global Market under the symbol “ALPN.” “ALPN.” As of February 29, 2020,March 14, 2023, we have approximately 2225 stockholders of record for our common stock, which excludes stockholders whose shares were held in nominee or street name accounts through brokers.
Dividends
We have never declared or paid cash dividends on our capital stock. We currently intend to retain any future earnings for use in the operation of our business and do not intend to declare or pay any cash dividends in the foreseeable future. Any further determination to pay dividends on our capital stock will be at the discretion of our board of directors, subject to applicable laws, and will depend on our financial condition, results of operations, capital requirements, general business conditions and other factors that our board of directors considers relevant.
Stock Performance Graph
As a “smaller “smaller reporting company,”” as defined by Rule 12b-2 of the Exchange Act, and pursuant to Instruction 6 to Item 201(e) of Regulation S-K we are not required to provide the stock performance graph.
Recent Sales of Unregistered Securities
Item 6. Selected Financial Data.
Warrant Exercises
AsOn January 10, 2023, we issued 1,641,544 shares of our common stock, $0.001 par value per share, or the First Warrant Net Exercise Shares, to warrant holders upon their exercise of outstanding pre-funded warrants to purchase an aggregate of 1,641,773 shares of our common stock, $0.001 par value per share, pursuant to a “smaller reporting company,” as defined by Rule 12b-2net exercise mechanism under the warrants. Each pre-funded warrant had an exercise price of $0.001 per share. The issuances of the ExchangeFirst Warrant Net Exercise Shares were exempt from registration under the Securities Act andof 1933, as amended, pursuant to Item 301(c) of Regulation S-K we are not required to provide selected financial data.Section 3(a)(9) thereof as an exchange with an existing security holder where no commission or other remuneration is paid or given for soliciting such exchange.
On January 25, 2023, we issued 66,991 shares of our common stock, $0.001 par value per share, or the Second Warrant Net Exercise Shares, to warrant holders upon their exercise of outstanding pre-funded warrants to purchase an aggregate of 67,000 shares of our common stock, $0.001 par value per share, pursuant to a net exercise mechanism under the warrants. Each pre-funded warrant had an exercise price of $0.001 per share. The issuances of the Second Warrant Net Exercise Shares were exempt from registration under the Securities Act of 1933, as amended, pursuant to Section 3(a)(9) thereof as an exchange with an existing security holder where no commission or other remuneration is paid or given for soliciting such exchange.
Item 6. [Reserved]
Item 7. Management’sManagement’s Discussion and Analysis of Financial Condition and Results of Operations.
You should read the following discussionManagement’s Discussion and analysisAnalysis of our financial conditionFinancial Condition and resultsResults of operationsOperations together with our consolidated financial statements and the related notes appearing elsewhere in this Annual Report on Form 10-K. In addition to historical information, some of the information contained in this discussion and analysis or set forth elsewhere in this report, including information with respect to our plans and strategy for our business, future financial performance, expense levels and liquidity sources, includes forward-looking statements that involve risks and uncertainties. You should read Risk Factors in Part I, Item 1A of this report for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.The discussion regarding our financial condition and results of operations for fiscal 2021 as compared to fiscal 2020 has been omitted from this Annual Report on Form 10-K and is incorporated by reference from our Annual Report on Form 10-K for the fiscal year ended December 31, 2021, filed with the SEC on March 17, 2022, under the section titled “Part II, Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations.”
Overview
We are a clinical-stage biopharmaceutical company dedicated to discovering and developing innovative, protein-based immunotherapies targeting the immune synapse to treat cancer and autoimmune and inflammatory diseases. Our approach includes a proprietary scientific platform that converts native immune system proteins into differentiated, multi-targeted therapeutics potentially capable of modulating the human immune system and significantly improvingtherapeutics. We are seeking to create first- or best-in-class multifunctional immunotherapies via our unique protein engineering technologies to improve outcomes in patients with serious diseases.
Our lead programAutoimmune and Inflammatory Diseases
Povetacicept is ALPN-101,a dual antagonist of BAFF and APRIL cytokines, which play key roles in the activation, development, and survival, of B cells. Based upon an engineered TACI domain, povetacicept has exhibited greater potency in preclinical studies versus wild-type TACI-based comparators, as well as other inhibitors of BAFF and/or APRIL alone. In addition, povetacicept has been well-tolerated in preclinical models and exhibited superior pharmacokinetics and pharmacodynamics over wild-type TACI-Fc counterparts, including superior serum exposure, suppression of T-dependent antibody production, and/or serum immunoglobulins in mice and/or cynomolgus monkeys. In a randomized, placebo-controlled, first-in-human, Phase 1 study in adult healthy volunteers (NCT05034484), povetacicept has been well tolerated to date and has demonstrated encouraging dose-related pharmacokinetic and on-target pharmacodynamic effects, which we believe supports the use of a once every four-week dose regimen for subsequent studies and enables a broad development plan in multiple indications. Povetacicept is in development for multiple B cell and/or autoantibody-related diseases, including SLE, glomerulonephritides, and autoimmune cytopenias.
Acazicolcept is a dual ICOS and CD28 antagonist intended for the treatment of autoimmune and inflammatory diseases. Preclinical studies with acazicolcept have demonstrated efficacy in models of graft versus host disease, or GVHD,SLE, SjS, arthritis, inflammatory bowel disease, multiple sclerosis, type 1 diabetes, systemic lupus erythematosus, or SLE,uveitis, and Sjögren’s syndrome. In an oral presentation at the 2019 American Society of Hematology Annual Meeting, we discussed data from ourgraft versus host disease. We have evaluated acazicolcept in a Phase 1 healthy volunteer study demonstrating that ALPN-101 was well-tolerated as single intravenous or subcutaneous doses, without cytokine release, infusion-related reactions, hypersensitivity, or other signsand are currently evaluating acazicolcept in Synergy, a global, randomized, double-blind, placebo-controlled Phase 2 study of agonist activity. Based on our Phase 1 data,acazicolcept in adults with moderate-to-severe SLE. In June 2020, we entered into the AbbVie Agreement, which grants AbbVie an exclusive option to take an exclusive license to acazicolcept. Through December 31, 2022, we have opened BALANCE,received $105.0 million in upfront and pre-option exercise development milestones as part of the AbbVie Agreement.
In December 2021, we entered into an open-label, dose escalation,exclusive license and expansion Phase 1b/2 studycollaboration agreement with Horizon which grants Horizon an exclusive license for the development, manufacture and commercialization of one Existing Program and up to three additional Research Programs generated from our libraries of proteins and molecules for research, discovery, and identification of additional compounds. Under the terms of the Horizon Agreement, Horizon made an upfront payment to us of $25.0 million as well as an equity investment for which they paid $15.0 million, a 25% premium to the 30-day volume-weighted average share price as of December 9, 2021. In addition, we are eligible to receive up to $381.0 million per program, or approximately $1.5 billion in patients with steroid-resistant or steroid-refractory active acute GVHD. We intendtotal, in future success-based payments related to enroll patients throughout 2020development, regulatory and into 2021. Beyond acute GVHD, we believe that ALPN-101 has the potential to be effective in inflammatory diseases like rheumatoid arthritis, SLE, and Sjögren’s syndrome.commercial milestones as well as tiered royalties on global net sales.
Immuno-oncology
Our lead oncology programDavoceticept is ALPN-202, a conditional CD28 costimulator and dual checkpoint inhibitor intended for the treatment of cancer. Preclinical cancer. In October 2022, we announced the voluntary termination of enrollment of davoceticept clinical studies, including the NEON-1 study of davoceticept as monotherapy and the NEON-2 study of davoceticept in vivo data have demonstrated monotherapy efficacycombination with pembrolizumab. The decision to terminate enrollment in tumor models superior to approved therapies. In addition, ALPN-202 hasthe davoceticept studies was made following notification of a unique immuno-modulatory profile and has demonstrated evidence of anti-tumor immunitysecond Grade 5 serious adverse event (death) in preclinical models. Based on ALPN-202’s efficacy in preclinical models and favorable nonclinical safety and development profile, we have opened NEON-1, a Phase 1 dose escalation and expansion study in patients with advanced malignancies. We intend to enroll patients throughout 2020 and into 2021. the NEON-2 study.
Scientific Platform
Our scientific platform has also generated immune modulatory proteins with the potential of improving engineered cellularcell therapies or ECT, such as chimeric antigen receptor T cells, or CAR-T, T cell receptor-engineered T cells, or TCR-T, and tumor infiltrating lymphocytes, or TILs.lymphocytes. In May 2019, we signed a collaboration and license agreement with Adaptimmune Therapeutics plc, or Adaptimmune, to develop next-generation SPEARTM T-cellSPEAR™ T cell products which incorporate our secretedSIP and transmembrane immunomodulatory protein (termed SIP™ and TIP™) technology. We intend to continue to leverage our existing pipeline and platform to actively explore and evaluate potential value-creating partnering opportunities.
Our Strategy
Our goal is to discover and develop modern therapies to treat patients with serious conditions such as cancerautoimmune and inflammatory diseases. To achieve our goals, we intend to:
| |
•
| aggressively move our lead inflammation/autoimmune program ALPN-101 through clinical development for the treatment of inflammatory diseases, such as GVHD;
|
| |
•
| aggressively move our lead oncology program ALPN-202 through clinical development for the treatment of cancer; and
|
| |
•
| maximize the value of our pipeline and platform via potential partnering activities.
|
•aggressively move our lead wholly-owned program povetacicept into clinical studies for the treatment of multiple B cell and/or autoantibody-related diseases;
•aggressively move our most advanced autoimmune and inflammatory program acazicolcept through clinical development as part of our collaboration with AbbVie including conducting Synergy, our Phase 2 study for the treatment of SLE; and
•maximize the value of our pipeline and platform via potential partnering activities.
Our operations to date have been limited to business planning, raising capital, developing our platform technology, identifying potential immunotherapy candidates, clinical studies, and other research and development activities. To date, we have financed operations primarily through public offerings of common stock and warrants, private placements of common stock, warrants and convertible preferred stock, funds received from license and research agreements, debt financing and assets acquired upon the close of our merger with Nivalis Therapeutics Inc., or Nivalis. We do not have any products approved for sale and have not generated any product sales. Since inception and through December 31, 2019,2022, excluding amounts borrowed through debt financing, we have raised an aggregate of $125.2approximately $526.6 million to fund operations, of which $23.6$292.3 million was from the sale of common stock $49.2and warrants, $141.0 million was through our license and collaboration agreements, $49.2 million was from the sale of convertible preferred stock, $8.3and $44.1 million was through our license and collaboration agreements, and $44.1 million in cash, cash equivalents,
and marketable securities acquired through the merger with Nivalis. As of December 31, 2019,2022, we had cash, cash equivalents, restricted cash, and short-term investments totaling $40.9 million.$273.4 million.
Our net loss was $41.9$57.8 million,, $36.5 and $50.3 million,, and $7.8 million for the years ended December 31, 2019, 2018,2022 and 2017,2021, respectively. We expect to continue incurring significant expenses and operating losses for at least the next several years as we:
•initiate and complete nonclinical studies and clinical trials for our product candidates, including acazicolcept, a dual ICOS/CD28 antagonist program targeting autoimmune and inflammatory disorders, and povetacicept, a dual B cell cytokine antagonist for B cell-mediated autoimmune and inflammatory diseases;
| |
•
| initiate and complete clinical trials for product candidates, including ALPN-101, a dual ICOS/CD28 antagonist program targeting autoimmune/inflammatory disorders and ALPN-202, a CD80 vIgD-Fc that mediates PD-L1-dependent CD28 costimulation and inhibits the PD-L1 and CTLA-4 checkpoints targeting cancer;
|
| |
•
| contract to manufacture and perform additional process development for our product candidates;
|
| |
•
| continue research and development efforts to build our pipeline beyond the current product candidates;
|
| |
•
| maintain, expand, and protect our intellectual property portfolio;
|
| |
•
| hire additional clinical, quality control, scientific, and management personnel; and
|
| |
•
| add operational and financial personnel to support our product development efforts and operational capabilities applicable to operating as a public company.
|
•contract to manufacture and perform additional process development for our product candidates;
•continue research and development efforts to build our pipeline beyond the current product candidates;
•maintain, expand, and protect our intellectual property portfolio;
•hire additional clinical, quality control, scientific, and management personnel; and
•add operational and financial personnel to support our product development efforts and operational capabilities applicable to operating as a public company.
We do not expect to generate product revenue unless and until we successfully complete development of, obtain marketing approval for, and commercialize our product candidates, either alone or in collaboration with third parties. We expect these activities will take a number of years and our success in these efforts is subject to significant uncertainty. Accordingly, we will need to raise additional capital prior to the regulatory approval and commercialization of any of our product candidates. Until such time, if ever, as we can generate substantial product revenues, we expect to finance our operating activities through equity or debt financings, collaborations or licenses, capital lease transactions, or other available financing transactions. However, additional capital may not be available on reasonable terms, if at all, and if we raise additional funds through the issuance of additional equity or debt securities, it could result in dilution to our existing stockholders and increased fixed payment obligations.
Financial Overview
Collaboration Revenue
We derive our collaboration revenue primarily from our collaboration and licensing agreements. We may generate revenue in the future from research support or milestone payments received pursuant to our collaboration agreementand licensing agreements with AbbVie, Horizon, Adaptimmune, or the Adaptimmune Collaboration Agreement, or from payments from future license or collaboration agreements, product sales, or government contracts and grants. We expect any revenue we generate, if any, will fluctuate from quarter to quarter.
AbbVie
In June 2020, we entered into the AbbVie Agreement for the development of acazicolcept. The License Option available under the AbbVie Agreement is exercisable by AbbVie at any time and will expire 90 days from the achievement of certain development milestones. If AbbVie exercises the License Option, AbbVie will take over the future development and commercialization. Prior to the exercise of the License Option, we will perform research and development services, including conducting our Phase 2 study in SLE, based on the Development Plan. We adopted Accounting Standards Codification,will be fully responsible for all costs incurred to conduct the activities under the Development Plan, provided that, AbbVie may be responsible for increased costs under the Development Plan in connection with certain material amendments proposed by AbbVie. We will also be solely responsible, at our sole cost and expense, for manufacturing and regulatory filings for acazicolcept necessary to complete activities under the Development Plan.
In June 2020, in connection with the execution of the AbbVie Agreement, AbbVie paid us a nonrefundable upfront payment of $60.0 million. Prior to the exercise of the License Option, AbbVie has agreed to make cash payments upon our achievement of the Alpine Development Milestones, up to an aggregate amount of $75.0 million. In 2021, we achieved $45.0 million of the Alpine Development Milestones. If AbbVie exercises the License Option, they will pay a one-time cash payment of $75.0 million. Following the exercise of the License Option, AbbVie has also agreed to make aggregate cash payments of up to $205.0 million upon AbbVie’s achievement of certain development and commercial milestones and additional aggregate cash payments of up to $450.0 million upon AbbVie’s achievement of certain sales-based cash milestones, collectively referred to as the AbbVie Milestones. Subsequent to commercialization, we are also eligible to receive high single-digit to low double-digit percentage royalties on worldwide net sales of licensed products.
For revenue recognition purposes, we determined that our contractual promises in the AbbVie Agreement are not distinct and are interdependent with our performance obligation to provide research and development services under the Development Plan. Thus, all contractual promises related to the upfront payment and Alpine’s Development Milestones were combined into a single performance obligation. We determined the Alpine Development Milestone payments are probable of significant revenue reversal as the achievement is highly dependent on factors outside our control. Therefore, these milestone payments were fully constrained and were not initially included in the transaction price. In June 2021, we re-evaluated and updated the transaction price to include the achieved portion of the Alpine Development Milestones. We will continue to re-evaluate the transaction price each reporting period and update as uncertain events are resolved or ASC, Topic 606,other changes in circumstances occur.
The License Option and the AbbVie Milestones were determined not to be performance obligations at the inception of the contract as they did not represent material rights. If exercised, the License Option and AbbVie Milestones will be accounted for as a separate contract and will be recognized as revenue if and when triggered. Any consideration related to sales-based royalties and profit-sharing payments will be recognized when the related sales occur.
We use a cost-based input method to measure progress toward completion of the performance obligation and to calculate the corresponding revenue to recognize each period. In applying the cost-based input, we use actual costs incurred relative to budgeted costs for the combined performance obligation. These costs consist primarily of internal personnel efforts and third-party contract costs relative to the level of patient enrollment in the study. Revenue from Contracts with Customers , as amended, orwill be recognized based on the newlevel of costs incurred relative to the total budgeted costs for the performance obligation. A cost-based input method of revenue standard or ASC 606, on January 1, 2018, resulting in a changerecognition requires management to make estimates of costs to complete our accounting policy for revenue recognition. We used the modified retrospective method transition option and recognized theperformance obligation. In making such estimates, significant judgment is required to evaluate assumptions related to cost estimates. The cumulative effect of adoptingrevisions to estimated costs to complete our performance obligation will be recorded in the newperiod in which changes are identified and amounts can be reasonably estimated. A significant change in these assumptions and estimates could have a material impact on the timing and amount of revenue standardrecognized in future periods.
We recognized revenue from the AbbVie Agreement of $17.1 million and $23.4 million for the years ended December 31, 2022 and 2021, respectively. We expect to recognize the remaining deferred revenue over the remainder of our Development Plan, which began in June 2020 and will end upon the later of the exercise or expiration of the option.
Horizon
In December 2021, we entered into the Horizon Agreement, which grants Horizon an exclusive license for the development, manufacture and commercialization of one Existing Program and up to three additional Research Programs generated from our libraries of proteins and molecules for research, discovery and identification of additional compounds.
Under the terms of the agreement, Horizon made an upfront payment to us of $25.0 million as well as an adjustmentequity investment for which they paid $15.0 million, a 25% premium to the opening accumulated deficit at January 1, 2018. Accordingly, 2017 comparative information has30-day volume-weighted average share price as of December 9, 2021. In addition, we are eligible to receive up to $381.0 million per program, or approximately $1.5 billion in total, in future success-based payments related to development, regulatory and commercial milestones as well as tiered royalties on global net sales. We have completed our activities under the Existing Program and will conduct additional activities for up to three Research Programs to deliver compounds meeting agreed criteria. In addition, Horizon will pay us for the costs and expenses of conducting such activities under the deliverables plans. Horizon will then assume responsibility for development and commercialization activities and costs.
For revenue recognition purposes, we determined that the Existing Program and each Research Program are distinct performance obligations. We allocated revenue to each performance obligation based on its relative stand-alone selling price. The future success-based payments related to development and regulatory milestones are probable of significant revenue reversal as the achievement is highly dependent on factors outside our control. Therefore, these milestone payments are fully constrained and are not
been adjustedinitially included in the transaction price. We will continue to re-evaluate the transaction price each reporting period and
continuesupdate as uncertain events are resolved or other changes in circumstances occur. Any consideration related to
commercial milestones and royalties will be
reported under previous accounting standards. See Note 12recognized when the related sales occur. We recognized revenue from the Horizon Agreement of $12.0 million for additional information.the year ended December 31, 2022, and no revenue for the year ended December 31, 2021.
Adaptimmune Therapeutics plc
In May 2019, we entered into a collaboration and licensing agreement, or the Adaptimmune Collaboration Agreement. with Adaptimmune Therapeutics plc, or Adaptimmune, a clinical-stage biopharmaceutical company primarily focused on providing novel cell therapies to patients, particularly for the treatment of solid tumors,Agreement to develop next-generation SPEAR T-cell products which incorporate our secreted and transmembrane immunomodulatory protein (termed SIP™ and TIP™) technology.T cell products. Under the Adaptimmune Collaboration Agreement, we are to perform certain research services and grant Adaptimmune an exclusive license to programs from our SIP and TIP technologies. In June 2019,
Through December 31, 2022, we have recorded a total of $3.0 million in license payments under the terms of the Adaptimmune Collaboration Agreement we received anconsisting of a $2.0 million upfront license payment received in June 2019 and an additional $1.0 million license fee upon Adaptimmune’s selection of $2.0 million and as of December 31, 2019an additional research program in June 2022. Furthermore, we have received an additional $750,000recorded $2.0 million in research support payments to fund ongoing programs. These payments were recorded as deferred revenue and will be recognized to revenue based on employee hours contributed to each performance obligation. We have recognized a total of $1.3 million in revenue for the year ended programs through December 31, 2019 related to our collaboration agreement with Adaptimmune. In addition,2022. If respective pre-specified milestones for each program are achieved, we are eligible for additional research support payments, one-time payments and downstream development and commercialization milestones of up to $288.0$288.0 million,, if all pre-specified
milestones for each program are achieved. We and we are also eligible to receive low-single digit percentage royalties on worldwide net sales of the applicable products.
Kite Pharma, a Gilead company
In October 2015, we entered into the Kite Collaboration Agreement, providing Kite with access to two transmembrane immunomodulatory protein, or TIP, programs for use in Kite’s engineered cellular therapy programs. In May 2019, Kite provided us notice of termination of the Kite Collaboration Agreement following the expiration of theFor revenue recognition purposes, licensing and research term. Upon termination, the confidentiality and indemnity obligations of the parties survived and the licenses granted to Kitesupport fees billed under the Kite Collaboration Agreement terminated. Pursuantagreement are being recorded as deferred revenue and recognized to the terms of the Kite Collaboration Agreement, the termination was effective in June 2019, thirty days after the effectiveness of Kite’s notice.revenue based on employee hours contributed to each performance obligation. We recognized a totalrevenue from the Adaptimmune Agreement of $5.6$1.0 million infor the year ended December 31, 2022 and no revenue related tofor the Kite Collaboration Agreement over the life of the agreement.year ended December 31, 2021.
Research and Development Expenses
We focus our resources on research and development activities, including the conduct of preclinical studies, product development, regulatory support, and clinical studiestrials for our product candidates. We recognize research and product development and expense such costsexpenses as they are incurred. Our research and development expenses consist of:
•
| |
•
| employee-related expenses, including salaries, benefits, taxes, travel, and stock-based compensation expense for personnel in research and development functions; |
| |
•
| expenses related to process development and production of product candidates paid to contract manufacturing organizations;
|
| |
•
| costs associated with preclinical activities and regulatory operations, including the cost of acquiring, developing, and manufacturing research material;
|
| |
•
| clinical trials and activities related to regulatory filings for our product candidates; and
|
| |
•
| allocation of facilities, overhead, depreciation, and amortization of laboratory equipment and other expenses.
|
We incurred $35.8 million, $29.0 million, and $10.6 million in research and development functions;
•expenses related to process development and production of product candidates paid to contract manufacturing organizations;
•costs associated with preclinical activities and regulatory operations, including the cost of acquiring, developing, and manufacturing research material;
•clinical trials and activities related to regulatory filings for the years ended our product candidates; and
•December 31, 2019, 2018,allocation of facilities, overhead, depreciation, and amortization of laboratory equipment and other expenses.2017, respectively. Our
We expect our direct and indirect research and development expenses couldto increase for the foreseeable future as we continue to develop our platform and product candidates. We remain focused on using our resources to further advance povetacicept’s broad development plan. We expect the future increase in investment in this program, including a phase 2 study in SLE, and open label basket studies in glomerulonephritis and autoimmune cytopenias, to more than offset the expected significant decrease in costs to support davoceticept as a result of the voluntary termination of enrollment of davoceticept clinical studies as announced by us in October 2022.
The successful development of our platform and product candidates is highly uncertain. At this time, we cannot reasonably estimate the nature, timing, or costs of the efforts necessary to finish developing any of our product candidates or the period in which material net cash, if any, from these product candidates may commence. This is due to the numerous risks and uncertainties associated with developing therapeutics, including the uncertainty of:
•the scope, rate of progress, expense, and results of clinical trials;
| |
•
| the scope, rate of progress, expense, and results of clinical trials;
|
| |
•
| the scope, rate of progress, and expense of process development and manufacturing;
|
| |
•
| preclinical and other research activities; and
|
| |
•
| the timing of regulatory approvals.
|
•the scope, rate of progress, and expense of process development and manufacturing;
•preclinical and other research activities; and
•the timing of regulatory approvals.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related costs for employees in executive, business development, finance, legal, and administrative functions. Other significant general and administrative expenses include professional fees for accounting and legal services, expenses associated with obtaining and maintaining patents and other intellectual property, and allocation of facility and overhead costs.
We expect general and administrative expenses couldto increase as we expand infrastructure, headcount, and continue to prosecute our patents and other intellectual property. Other increases could potentially include increased costs for director and officer liability insurance, costs related to the hiring of additional personnel, and increased fees for directors, outside consultants, lawyers, and accountants. We expect to incur significant costs to comply with corporate governance, internal controls, and similar requirements applicable to public companies.
Loss on Sale of Intangible Asset
Loss on sale of intangible asset relates solely to the sale of the GSNOR asset to Laurel Venture Capital Ltd., or Laurel, in June 2018. For additional information regarding the sale of the GSNOR asset, please see Note 8 to our consolidated financial statements contained elsewhere in this Annual Report on Form 10-K. Bargain Purchase Gain
As Alpine was the accounting acquirer in the merger agreement with Nivalis Therapeutics, Inc., we allocated the purchase price to the acquired tangible and intangible assets and assumed liabilities of Nivalis based on their estimated fair values as of the acquisition date. The excess of the estimated fair values of net assets acquired over the acquisition consideration paid was recorded as a bargain purchase gain in the consolidated statements of operations and comprehensive income (loss). The determination of the fair values of the assets acquired and liabilities assumed requires significant judgment, including third party valuation estimates relating to the value of the acquired in-process research and development asset, or IPR&D.
Interest and Other Expense
Interest and other expense consists primarily of interest associated with our term loan with Silicon Valley Bank and the amortization of the related debt discount.
Interest Income
Interest income consists of interest earned on our cash, cash equivalents, and short-term investments.
Interest Expense
Interest expense consists primarily of interest associated with our term loans with Silicon Valley Bank, or SVB, and the amortization of the related debt discount.
Other, Incomenet
Other incomeFor the years ended December 31, 2022 and 2021, other expense consists of a refundable Australian tax credit from our wholly-owned Australian subsidiary.the loss on the sale of equipment.
Income Tax Benefit (Expense)
Income tax expense for 2022 primarily relates to a cumulative change to our foreign income tax provision and domestic foreign and state income taxes recognized as the result of capitalization of research and development expenses under the TCJA. The income tax benefit for 20182021 relates to the saleremoval of the valuation allowance against our in-process research and development, or IPR&D. Income tax benefit for 2017 relates to theforeign deferred tax liability recorded in connection with the acquisition of our IPR&D, whichassets as we had a financial reporting basis of $1.5 million and a tax basis of zero. generated sufficient foreign taxable income to utilize all historical operating losses.
Results of Operations
Comparison of Years Ended December 31, 20192022 and 20182021
The following table summarizes our results of operations for the years ended December 31, 2019 and 2018(in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Years Ended December 31, | | | | |
| | | 2022 | | 2021 | | $ Change | | % Change |
| Collaboration revenue | | $ | 30,064 | | | $ | 23,443 | | | $ | 6,621 | | | 28 | % |
| Operating expenses: | | | | | | | | |
| Research and development | | 70,243 | | | 58,742 | | | 11,501 | | | 20 | % |
| General and administrative | | 17,968 | | | 14,560 | | | 3,408 | | | 23 | % |
| Total operating expenses | | 88,211 | | | 73,302 | | | 14,909 | | | 20 | % |
| Loss from operations | | (58,147) | | | (49,859) | | | (8,288) | | | 17 | % |
| Other income (expense): | | | | | | | | |
| Interest income | | 3,288 | | | 259 | | | 3,029 | | | 1,169 | % |
| Interest expense | | (476) | | | (816) | | | 340 | | | (42) | % |
| Other, net | | (97) | | | (4) | | | (93) | | | 2,325 | % |
| Loss before taxes | | (55,432) | | | (50,420) | | | (5,012) | | | 10 | % |
| Income tax benefit (expense) | | (2,330) | | | 87 | | | (2,417) | | | (2,778) | % |
| Net loss | | $ | (57,762) | | | $ | (50,333) | | | $ | (7,429) | | | 15 | % |
|
| | | | | | | | | | | | |
| | Years Ended December 31, | | Increase/ |
| | 2019 | | 2018 | | (Decrease) |
Collaboration revenue | | $ | 1,740 |
| | $ | 705 |
| | $ | 1,035 |
|
Operating expenses: | | | | | | |
Research and development | | 35,847 |
| | 28,970 |
| | 6,877 |
|
General and administrative | | 9,467 |
| | 8,362 |
| | 1,105 |
|
Loss on sale of intangible asset | | — |
| | 1,203 |
| | (1,203 | ) |
Total operating expenses | | 45,314 |
| | 38,535 |
| | 6,779 |
|
Loss from operations | | (43,574 | ) | | (37,830 | ) | | (5,744 | ) |
Other income (expense): | | | | | | |
Interest and other expense | | (338 | ) | | (319 | ) | | (19 | ) |
Interest income | | 1,248 |
| | 1,296 |
| | (48 | ) |
Other income | | 812 |
| | — |
| | 812 |
|
Loss before taxes | | (41,852 | ) | | (36,853 | ) | | (4,999 | ) |
Income tax benefit | | — |
| | 366 |
| | (366 | ) |
Net loss | | $ | (41,852 | ) | | $ | (36,487 | ) | | $ | (5,365 | ) |
Collaboration Revenue
Revenue for the year ended December 31, 2019 consists of $1.3The following table summarizes our collaboration revenue by partner (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | | | |
| 2022 | | 2021 | | $ Change | | % Change |
| AbbVie | $ | 17,064 | | | $ | 23,443 | | | $ | (6,379) | | | (27) | % |
| Horizon | 11,964 | | | — | | | 11,964 | | | NM |
| Adaptimmune | 1,036 | | | — | | | 1,036 | | | NM |
| Total collaboration revenue | $ | 30,064 | | | $ | 23,443 | | | $ | 6,621 | | | 28 | % |
| NM: No amount in comparable period or not a meaningful comparison. |
The $6.6 million, related to the Adaptimmune Collaboration Agreement and $0.4 million related to the milestone payment from Laurel from the sale of our GSNOR assets. Revenue for the year ended December 31, 2018or 28%, increase in collaboration revenue relates primarily to upfront research support payments of $5.5$12.0 million underin revenue recognized for services performed in connection with the Kite CollaborationHorizon Agreement, which were initially recorded as deferred revenuewas executed in late 2021, and recognized over the perioda $1.0 million increase related to Adaptimmune’s selection of thean additional research term completed during the first half of 2018. The adoption of ASC 606 resultedprogram in $0.2 million in higher revenue for the 2018 period, as compared to what would have been recorded under previous accounting guidance.
Research and Development Expenses
The $6.9 millionincrease in research and development expenses was primarily attributable to an increase of $4.9 million in clinical trial activity, an increase of $0.3 million in direct research activities, an increase of $2.3 million in personnel-related expenses as a result of growth in headcount to support ongoing discovery and development programs, an increase of $0.8 million in stock-based compensation, and an increase of $1.4 million in allocated overhead and facilities.June 2022. These increases were partially offset by a $6.4 million decrease in revenue recognized from our AbbVie Agreement as result of $2.8upward revisions to the projected program duration and corresponding increase in estimated total direct and indirect development costs to complete the program.
Research and Development Expenses
Our direct research and development expenses consist primarily of expenses incurred pursuant to agreements with third-party manufacturing organizations for our product candidates, CROs, clinical trial sites, collaborators, and consultants. Other direct costs included direct research and development costs incurred before a selected product candidate begins clinical trials.
We use our employee and infrastructure resources across multiple research and development programs that we are advancing in parallel, and therefore do not allocate salaries, stock-based compensation, employee benefit expenses or other indirect costs related to our research and development to specific product candidates. These expenses are included in indirect research and development expense by type in the table below. Our research and development expenses are summarized as
follows (in thousands): | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Years Ended December 31, | | | | |
| | | 2022 | | 2021 | | | | $ Change | | % Change |
| Direct research and development expense by program: | | | | | | | | | | |
| Acazicolcept | | $ | 12,282 | | | $ | 10,923 | | | | | $ | 1,359 | | | 12 | % |
| Davoceticept | | 10,035 | | | 10,640 | | | | | (605) | | | (6) | % |
| Povetacicept | | 12,561 | | | 13,311 | | | | | (750) | | | (6) | % |
| Other | | 1,070 | | | 515 | | | | | 555 | | | 108 | % |
| Total direct research and development expense | | 35,948 | | | 35,389 | | | | | 559 | | | 2 | % |
| Indirect research and development expense by type: | | | | | | | | | | |
| Personnel-related costs | | 28,239 | | | 17,456 | | | | | 10,783 | | | 62 | % |
| Research and development supplies and services | | 2,825 | | | 2,833 | | | | | (8) | | | — | % |
| Allocated facility, equipment and other expenses | | 3,231 | | | 3,064 | | | | | 167 | | | 5 | % |
| Total indirect research and development expense | | 34,295 | | | 23,353 | | | | | 10,942 | | | 47 | % |
| Total research and development expense | | $ | 70,243 | | | $ | 58,742 | | | | | $ | 11,501 | | | 20 | % |
Total research and development expenses increased by $11.5 million, or 20%, in 2022. The increase was primarily attributable to a $10.9 million, or 47%, increase in indirect research and development expenses, which largely related to a $10.8 million increase in personnel-related expenses. The rise in personnel costs, which includes an increase of $2.4 million in contractnon-cash stock-based compensation expense, was primarily due to increased headcount to support increased clinical trial activities.
Direct research and development expenses increased by $0.6 million, or 2%, in 2022, due largely to a $1.4 million increase in costs related to our acazicolcept program, which is primarily attributable to increased spending on our Phase 2 SLE study, partially offset by decreased manufacturing costs, and processa $0.6 million increase in other direct research and development expense, incurred in support of our product candidates.pipeline and ongoing collaborations. Povetacicept program costs decreased $0.8 million, primarily related to decreased manufacturing costs, partially offset by increased clinical trial costs as we move the program into clinical studies for the treatment of multiple B cell and/or autoantibody-related diseases. Davoceticept program costs decreased $0.6 million, primarily related to the termination of NEON-1 and NEON-2 clinical study enrollment.
General and Administrative Expenses
The $1.1$3.4 million, or 23%, increase in general and administrative expenses was primarily attributable to a $0.7increases of $2.4 million in personnel related expenses, which includes an increase of $1.0 million in personnel-related expenses relatednon-cash stock-based compensation expense, due primarily to an increase in administrative headcount, a $0.3headcount; $0.8 million increase inrelated to professional and legal services,services; and an increase of $0.1$0.2 million in insurance, facility and other costs to support the growth and expansion of our business.
Loss on Sale of Intangible AssetInterest Income
Loss on sale of intangible asset relates solely to the sale of the GSNOR asset to Laurel in June 2018.
Other Income
The $0.8 millionincrease in other income relates to income from the Australian tax credit from our wholly-owned Australian subsidiary.
Comparison of Years Ended December 31, 2018 and 2017
The following table summarizes our results of operations for the years ended December 31, 2018 and 2017 (in thousands):
|
| | | | | | | | | | | | |
| | Years Ended December 31, | | Increase/ |
| | 2018 | | 2017 | | (Decrease) |
Collaboration revenue | | $ | 705 |
| | $ | 1,731 |
| | $ | (1,026 | ) |
Operating expenses: | | | | | | |
Research and development | | 28,970 |
| | 10,626 |
| | 18,344 |
|
General and administrative | | 8,362 |
| | 6,079 |
| | 2,283 |
|
Loss on sale of intangible asset | | 1,203 |
| | — |
| | 1,203 |
|
Total operating expenses | | 38,535 |
| | 16,705 |
| | 21,830 |
|
Loss from operations | | (37,830 | ) | | (14,974 | ) | | (22,856 | ) |
Other income (expense): | | | | | | |
Bargain purchase gain | | — |
| | 6,601 |
| | (6,601 | ) |
Interest and other expense | | (319 | ) | | (152 | ) | | (167 | ) |
Interest income | | 1,296 |
| | 542 |
| | 754 |
|
Loss before taxes | | (36,853 | ) | | (7,983 | ) | | (28,870 | ) |
Income tax benefit | | 366 |
| | 200 |
| | 166 |
|
Net loss | | $ | (36,487 | ) | | $ | (7,783 | ) | | $ | (28,704 | ) |
Collaboration Revenue
The $1.0 milliondecrease in revenue was primarily attributable to the timing of revenue recognized under the Kite Collaboration Agreement, the recognition of the first research support payment under the amendment to the Kite Collaboration Agreement dated as of October 20, 2017, or the Kite Amendment, and the adoption of ASC 606. Under the terms of the Kite Collaboration Agreement, we received upfront payments of $5.5 million, which were initially recorded as deferred revenue and recognized over the period of the research term, which was completed during the first half of 2018. During 2018, the expected research term was extended pursuant to the Kite Amendment and we recognized the first research support payment under the amended agreement. The adoption of ASC 606 resulted in $0.2 million in higher revenue for the 2018 period, as compared to what would have been recorded under previous accounting guidance.
Research and Development Expenses
The $18.3 millionincrease in research and development expenses was primarily attributable to an increase of $9.2 million in contract manufacturing and process development of our product candidates, an increase of $5.3 million in direct research activities, an increase of $2.7 million in personnel-related expenses as a result of growth in headcount to support ongoing discovery and development programs, an increase of $0.6 million in stock-based compensation, and an increase of $0.5 million in allocated overhead and facilities.
General and Administrative Expenses
The $2.3 millionincrease in general and administrative expenses was primarily attributable to a $1.6$3.0 million increase in personnel-related expenses relatedinterest income was attributable to an increase in administrative headcount, an increase of $0.9 million in stock-based compensation,higher investment balances and a $0.5 million increase in insurance and facility costs to support the growth and expansion of our business. Offsetting this increase was a $0.7rising interest rates.
Interest Expense
The $0.3 million decrease in professionalinterest expense was attributable to less interest expense and legal services, which relates primarily to lower merger-related costs, partially offset by higher costs to support operating as a public company for a full year in 2018.
Loss on Sale of Intangible Asset
Loss on sale of intangible asset relates solely to the saleamortization of the GSNOR assetrelated debt discount due to Laurel in June 2018.
Bargain Purchase Gain
The bargain purchase gain relates solely to the excess of the estimated fair values of net assets acquired over the acquisition consideration paid for Nivalis.
Interest and Other Expense
Interest expense relates primarily to interest paida lower principle balance on our term loan with Silicon Valley Bank, whichSVB.
Income Tax Benefit (Expense)
The $2.4 million change in income tax benefit (expense) primarily relates to $1.3 million in tax expense resulting from a cumulative change in our foreign income tax provision as the result of a foreign income tax return filed under a revised transfer pricing model for the activities of our wholly owned subsidiary, Alpine Immune Sciences Australia PTY LTD. In addition, in 2022 we drew downrecognized $1.0 million in June 2017, and the related non-cash interestincome tax expense associated with the amortizationas a result of the debt discount. mandatory capitalization of research and development expenses as required under IRC Section 174, pursuant to the TCJA, which took effect in 2022.
Interest Income
The increase in interest income relates primarily to more interest earned on our short-term investments as we maintained a higher average investment balance during the 2018 period.
Liquidity and Capital Resources
Sources of Liquidity
To date, we have financed our operations primarily through the sale of equity securities, payments received under our collaboration agreements, debt, and funds acquired upon the close of our merger with Nivalis. As of December 31, 2019,2022, we had cash, cash equivalents, restricted cash, and short-term investments totaling $273.4 million. Except for any obligations of our collaborators to make milestone payments under our agreements with them, we do not have any committed external sources of capital. Until such time as we can generate substantial product revenue, if ever, we expect to finance our cash needs through a combination of collaboration agreements and equity or debt financings.
Equity Financing Agreements
In September 2022, we entered into an underwriting agreement, the Underwriting Agreement, with Morgan Stanley & Co. LLC, SVB Securities LLC and Cowen and Company, LLC, acting as representatives of the several underwriters named therein, or, collectively, the Underwriters, pursuant to which we sold 15,509,282 shares of our common stock, or the Firm Shares, in an underwritten public offering, including the subsequent partial exercise of the underwriters’ over-allotment option in October 2022, pursuant to our effective shelf registration statement on Form S-3 (File No. 333-256107). The net proceeds of the public offering were approximately $106.7 million, after deducting underwriting discounts, commissions and offering expenses.
In December 2021, in connection with the execution of the Horizon Agreement, we entered into a Stock Purchase Agreement with Horizon, or the Purchase Agreement, in which Horizon made an equity investment in Alpine of 951,980 shares of our common stock for approximately $15.76 per share, for an aggregate purchase price of $15.0 million. The purchase price represents a 25% premium to the volume-weighted average share price of our common stock for the 30-day period ended December 9, 2021. The shares were recorded at the fair value of our common stock on the effective date of the Horizon Agreement, with the excess of the proceeds recorded to deferred revenue.
In September 2021, we entered into a securities purchase agreement, or the 2021 Securities Purchase Agreement, for a private placement with a select group of institutional investors, pursuant to which we sold 6,489,357 shares of our common stock, or the Shares, and prefunded warrants to purchase 3,191,487 Shares, or the Prefunded Warrants. The purchase price for each Share and for each Prefunded Warrant was $9.40 per share, for an aggregate purchase price of approximately $91.0 million. The Prefunded Warrants became fully exercisable upon the closing date and have an exercise price of $0.001 per share. In connection with the 2021 Securities Purchase Agreement, approximately 3.7 million of the Shares issued and approximately 2.3 million of the Prefunded Warrants issued, for gross proceeds of approximately $57.0 million, were issued to certain stockholders whose beneficial ownership exceeded 5% prior to completion of the 2021 Securities Purchases Agreement.
In July 2021, we entered into a sales agreement, or the Sales Agreement, with Cowen and Company, LLC, or Cowen, pursuant to which we may sell shares of our common stock from time to time through an “at the market” equity offering for up to $75.0 million in gross cash proceeds. Cowen will act as the sales agent and will be entitled to compensation for services of up to 3.0% of the gross sales price per share of all shares sold through Cowen under the Sales Agreement. The shares would be issued pursuant to our effective shelf registration statement on Form S-3 (File No. 333-256107). We filed a prospectus supplement, dated July 2, 2021, with the SEC in connection with the offer and sale of the shares pursuant to the Sales Agreement. In connection with our September 2022 public offering, we and Cowen mutually terminated the Sales Agreement, and, as a result, we wrote off $0.5 million in deferred financing costs to expense, which were previously included within prepaid expenses and other current assets on our accompanying $40.9 millionConsolidated Balance Sheets. Excluding amounts borrowed through debt financing, we have raised an aggregate of $125.2 million to fund operations, of which $23.6 million was from the saleNo shares of common stock were sold under the Sales Agreement prior to its termination. In July 2020, we entered into a securities purchase agreement, or the 2020 Securities Purchase Agreement, for a private placement with a select group of institutional investors, pursuant to which we sold 5,139,610 units, or the Common Units, and 790,710 units, or the Prefunded Warrant Units, for an aggregate purchase price of $60.0 million. Each Common Unit consists of one share of our common stock plus a warrant to purchase 0.3 shares of common stock, or the Common Stock Warrants, and each Prefunded Warrant Unit consists of one prefunded warrant to purchase one share of common stock, or the Prefunded Warrants, plus one Common Stock Warrant to purchase 0.3 shares of common stock. The Prefunded Warrant Units and the Common Units are collectively referred to as the Units and each Unit has a purchase price of $10.1175. The Common Stock Warrants have an exercise price of $12.74 and a term of 3.5 years. The Prefunded Warrants became fully exercisable upon the closing date and have an exercise price of $0.001 per share.
Debt Financing Agreements
As of December 31, 2022, we had $3.4 million in remaining principal balance and accrued final payment fees outstanding under our 2019 term loans agreement with SVB and were in compliance with our respective loan covenants. See $49.2 millionNote 8 for further discussion of our term loans. Cash Flows
The following is a summary of our cash flows (in thousands):
| | | | | | | | | | | | | | | | |
| | | Years Ended December 31, |
| | | 2022 | | 2021 | | |
| Net cash used in operating activities | | $ | (43,754) | | | $ | (15,248) | | | |
| Net cash used in investing activities | | (113,348) | | | (52,480) | | | |
| Net cash provided by financing activities | | 102,689 | | | 100,764 | | | |
Net cash used in operating activities:
Net cash used in operating activities of $43.8 million for the year ended December 31, 2022 was primarily attributable to our net loss of $57.8 million, partially reduced by $4.1 million in net changes in our operating assets and liabilities. Additionally, we had $9.9 million in net non-cash adjustments, which primarily relate to stock-based compensation, depreciation and amortization.
Net cash used in operating activities of $15.2 million for the year ended December 31, 2021 was primarily attributable to our net loss of $50.3 million, partially reduced by $27.3 million in net changes in our operating assets and liabilities. Additionally, we had $7.8 million in net non-cash adjustments, which primarily relate to stock-based compensation, depreciation and amortization.
Net cash used in investing activities:
Cash flows from investing activities primarily reflect cash used to purchase investments and proceeds from the salematurities and sales of convertible preferred stock, $8.3 million throughinvestments, thus causing a shift between our licensecash and collaboration agreements,cash equivalents and $44.1 million ininvestment balances. We manage our cash usage with respect to our total cash, cash equivalents and marketable securities acquired throughinvestments.
Net cash used in investing activities was $113.3 million and $52.5 million for the merger with Nivalis. In June 2017, August 2019,years ended December 31, 2022 and March 2020, we drew down term loans from Silicon Valley Bank, or SVB, as discussed below. In addition2021, respectively, and consisted primarily of our purchases and maturities of investments, and purchases of property and equipment, primarily lab equipment, to support our existing cash, cash equivalents, and marketable securities, we are eligible to receive research and development funding and to earn milestone and other contingent paymentsefforts.
Net cash provided by financing activities:
Net cash provided by financing activities was $102.7 million for the achievementyear ended December 31, 2022 and consisted of defined collaboration objectivesthe net proceeds of $106.7 million related to the sale of approximately 15.5 million shares of common stock in our underwritten public offering, and certain development$0.8 million in proceeds from the exercise of stock options. The net proceeds received from issuances of common stock were partially offset by $4.8 million in principal payments on our debt.
Net cash provided by financing activities was $100.8 million for the year ended December 31, 2021 and regulatory milestonesconsisted primarily of the net proceeds of $90.7 million related to the sale of approximately 6.5 million shares of common stock and royalty payments3.2 million prefunded warrants under our collaboration with Adaptimmune; however,2021 Securities Purchase Agreement, $11.9 million in proceeds from issuance of common stock to Horizon in December 2021, and $0.5 million related to stock option exercises. These net proceeds from issuances of common stock were partially offset by $2.4 million in principal payments on our ability to earn these milestone and contingent payments and the timing of achieving these milestones is uncertain.debt.
Funding Requirements
We have incurred operating losses since inception. We expect to continue to incur significant expenses and operating losses for the foreseeable future as we continue our research and preclinical and clinical development of our product candidates; expand the scope of our current studies for our product candidates; initiate additional preclinical, clinical or other studies for our product candidates, including under any collaboration agreements; change or add additional manufacturers or suppliers; seek regulatory and marketing approvals for any of our product candidates that successfully complete clinical
studies; seek to identify, evaluate and validate additional product candidates; acquire or in-license other product candidates and technologies; maintain, protect and expand our intellectual property portfolio; attract and retain skilled personnel; and experience any delays or encounter issues with any of the above. Additionally, we have ongoing obligations with respect to our term loans with SVB, and our operating lease and certain contingencies, as described below.
Until such time as we can generate substantial product revenue, if ever, we expect to finance our cash needs through a combination of equity or debt financings and collaboration agreements. Except for any obligations of our collaborator to make milestone payments under our agreement with them, we do not have any committed external sources of capital. To the extent that we raise additional capital through the future sale of equity or debt, the ownership interest of our stockholders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our existing common stockholders. If we raise additional funds through collaboration agreements in the future, we may have to relinquish valuable rights to our technologies, future revenue streams or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
Our future capital requirements are difficult to forecast and will depend on many factors, including:
•the number and characteristics of the future product candidates we pursue either from our internal research efforts or through acquiring or in-licensing other product candidates or technologies;
| |
•
| the number and characteristics of the future product candidates we pursue either from our internal research efforts or through acquiring or in-licensing other product candidates or technologies;
|
| |
•
| the scope, progress, results and costs of independently researching and developing any of our future product candidates, including conducting preclinical research and clinical trials;
|
| |
•
| whether our existing collaboration generates substantial milestone payments and, ultimately, royalties on future approved products for us;
|
| |
•
| the timing of, and the costs involved in, obtaining regulatory approvals for any future product candidates we develop independently;
|
| |
•
| the cost of future commercialization activities, if any;
|
| |
•
| the cost of manufacturing our future product candidates and products, if any;
|
| |
•
| our ability to maintain our existing collaboration and to establish new collaborations, licensing or other arrangements and the financial terms of such arrangements;
|
| |
•
| the costs of preparing, filing, prosecuting, maintaining, defending and enforcing patents, including litigation costs and the outcome of such litigation; and
|
| |
•
| the timing, receipt and amount of sales of, or royalties on, our current or future collaborators’ product candidates, and our future products, if any.
|
•the scope, progress, results and costs of independently researching and developing any of our future product candidates, including conducting preclinical research and clinical trials;
•whether our existing collaborations generate substantial milestone payments and, ultimately, royalties on future approved products for us;
•the timing of, and the costs involved in, obtaining regulatory approvals for any future product candidates we develop independently;
•the cost of future commercialization activities, if any;
•the cost of manufacturing our future product candidates and products, if any;
•our ability to maintain our existing collaborations and to establish new collaborations, licensing or other arrangements and the financial terms of such arrangements;
•the costs of preparing, filing, prosecuting, maintaining, defending and enforcing patents, including litigation costs and the outcome of such litigation; and
•the timing, receipt and amount of sales of, or royalties on, our current or future collaborators’ product candidates, and our future products, if any.
We have considered that our long-term operations anticipate continuing net losses and the need for potential equity or debt financing. We have also considered that new collaborations or selectively partnering our technology or programs may provide other sources of capital. However, there can be no assurances that additional funding or other sources of capital will be available on terms acceptable to us, or at all. Therefore, we developed a plan to implement cost cutting measures to reduce our working capital requirements assuming no additional planned financing. This plan includes a delay in hiring and additional reductions in personnel-related costs and other discretionary expenditures that are within our control and do not effect the anticipated timing of our Phase 1 clinical trials. Based on thisour current operating plan, we believe our available cash and cash equivalents and investments, will be sufficient to fund our planned level of operations for at least the next 12 months. We have based this estimate on assumptions that may prove to be wrong, and we could use our capital resources sooner than we expect. Additionally, the process of testing drug candidates in preclinical and clinical studies is costly, and the timing of progress in these studies remains uncertain.
Financing Agreements
In January 2019, we entered into a securities purchase agreement, or Further, inflation may affect our use of capital resources by increasing our cost of labor and clinical trial expenses. Our long-term funding requirements will consist of operational, capital, and manufacturing expenditures, including those contractual commitments described above. Because of the Purchase Agreement,inherent risks and uncertainties associated with a limited number of accredited investors, pursuant to which we sold approximately 4.7 million units, or the Units, for an aggregate purchase price of $25.3 million in a private placement, which we refer to as the Private Placement. Each Unit has a purchase price of $5.37 per Unitdevelopment and consists of one sharecommercialization of our common stock and a warrant to purchase 0.39 shares of common stock. Pursuant to the terms of the Purchase Agreement,product candidates, we issued approximately 4.7 million shares of common stock and warrants to purchase an aggregate of approximately 1.8 million shares of common stock. The warrants have an exercise price of $12.74 and have a term of five years.
Prior to execution and delivery of the merger agreement with Nivalis, certain holders of our Series A-1 convertible preferred stock purchased shares of our Series A-1 convertible preferred stock. In March 2017, we issued and sold 707,330 shares of Series A convertible preferred stock and received a total of $4.0 million. In April 2017, we issued and sold 2,947,211 shares of our Series A-1 convertible preferred stock for an aggregate of $16.7 million in net proceeds. In addition, contemporaneously with the close of the merger with Nivalis, certain existing stockholders of Alpine purchased 1,335,118 additional shares of Alpine’s capital stock for an aggregate of $17.0 million in net proceeds.
In June 2018, we entered into an equity distribution agreement, or the Equity Distribution Agreement, with Piper Jaffray & Co., or Piper Jaffray, pursuant to which we may sell shares of our common stock through an “at the market” equity offering program for up to $50.0 million in gross cash proceeds. Piper Jaffray will be entitled to compensation for its services of up to 3.0% of the gross sales price per share of all shares sold through Piper Jaffray under the Equity Distribution Agreement. The Equity Distribution Agreement may be terminated by us upon written notice to Piper Jaffray for any reason, or by Piper Jaffray upon written notice to us for any reason, or at any time under certain circumstances, including but not limited to if we experience a material adverse change.
Under the Equity Distribution Agreement, we will set the parameters for the sale of shares, including the number of shares to be issued, the time period during which sales are requested to be made, limitation on the number of shares that may be sold in any one trading day and any minimum price below which sales may not be made. We have no obligation to sell any shares under the Equity Distribution Agreement and may at any time suspend solicitation and offers under the Equity Distribution Agreement. As of December 31, 2019, we have made no sales under the Equity Distribution Agreement. In July 2019, our Registration Statement on Form S-3 (File No. 333-212404) expired pursuant to Rule 415(a)(5) under the Securities Act of 1933, as amended. We will be unable to sell shares underestimate the Equity Distribution Agreement until a new Registration Statement on Form S-3 is filedamounts of capital outflows and declared effective by the SEC and a prospectus supplement relating to any sales under the Equity Distribution Agreement is filed with the SEC.
Long-Term Financing
In December 2016, we entered into a Loan and Security Agreement, or the Original Agreement, with SVB under which we borrowed $5.0 million. The Original Agreement accrued interest at a floating per annum rate equal to the lender’s prime rate minus 1.75%. The Original Agreement had an interest-only period through July 2018.
In August 2019, we entered into an Amended and Restated Loan and Security Agreement, or the Loan Agreement, with SVB, pursuant to which SVB agreed to extend term loans to us with an aggregate principal amount of up to $15.0 million, or the Term Loans. Borrowings under the Loan Agreement consist of up to three separate tranches. The initial tranche of $5.0 million was funded in August 2019, $3.0 million of which was used to repay amounts owing under our Original Agreement. We intend to use the remaining proceeds for potential working capital and other general corporate purposes, including the advancement of our development programs. In March 2020, the second tranche of $5.0 million was funded to us. The third and final tranche of up to $5.0 million is available at our option at any time from the date on which SVB receives and approves evidence that we have initiated a Phase 2a trial of ALPN-101 in psoriatic arthritis through July 31, 2020. Each term loan advance, other than the final term loan advance, must be in an amount of not less than $0.5 million and, after repayment, no term loan advance may be re-borrowed.
The Term Loans accrue interest at a floating per annum rate of 0.25% above the prime rate, subject to a floor of 5.75%, which interest is payable monthly commencing in September 2019. The Term Loans are interest only until September 30, 2020, after which the Term Loans will be payable in 34 equal monthly installments of principal plus interest, with the final installment due and payable on July 1, 2023. A final payment in the amount of 5.5% of the Term Loans funded is payable to the SVB on the date on which the Term Loans are prepaid, paid or become due and payable in full.
We may prepay all, but not less than all, of the Term Loans subject to a prepayment fee equal to $75,000, which represents the deferred portion of the final payment due under the Original Agreement, plus the outstanding principal balance under the Term Loans at the time of such prepayment multiplied by (i) 2.0%, if the prepayment occurs on or prior to the first
anniversary of August 26, 2019, (ii) 1.0%, if the prepayment occurs after the first anniversary of August 26, 2019, but on or prior to the second anniversary of August 26, 2019 or (iii) 0%, if the prepayment occurs after the second anniversary of August 26, 2019, but prior to the maturity date for the Term Loan. A fee in the amount of 5.5% of the Term Loans funded is payable to the Bank on the date on which the Term Loans are prepaid, paid or become due and payable in full.
The Loan Agreement contains customary representations and warranties, events of default and affirmative and negative covenants, including, among others, covenants that limit or restrict our ability to, among other things, incur additional indebtedness, grant liens, merge or consolidate, make acquisitions, pay dividends or other distributions or repurchase equity, make investments, dispose of assets, engage in any new line of business, and enter into certain transactions with affiliates, in each case subject to certain exceptions. Among other events, a failure to make a required loan payment, an uncured covenant breach or a material adverse change in our business, operations or condition (financial or otherwise) could lead to an event of default, and in such case, all amounts then outstanding may become due and payable immediately. We were in complianceoperating expenditures associated with our covenants as of December 31, 2019. As security for its obligations under the Loan Agreement, we granted SVB a first priority security interest on substantially all of our assets, except intellectual property,long-term anticipated preclinical studies and subject to certain other exceptions. As of December 31, 2019, we had $5.0 million outstanding principal amount under our term loan agreement. See Note 10 for further discussion of our Term Loans.clinical trials.Operating Lease
In March 2019, we entered into a lease with ARE-Seattle No. 28, LLC, or the Landlord, for 27,164 square feet of office and laboratory space located at 188 East Blaine Street, Seattle, Washington. The term of the lease is 10.8 years with one option to extend the term by 5 years. The lease term commenced in June 2019. The “Rent“Rent Commencement Date” will beDate” began in March 2020, nine months after the commencement date. We arewere not required to pay base rent from the Rent Commencement
Date through November 2020, the last day of the ninth month following the Rent Commencement Date. The annual base rent under the lease is $1.7 million for the first year and will increase by 3.0% each year thereafter. We will receivereceived a maximum tenant improvement allowance of $5.4 million, which is included in our base rent, and a maximum additional tenant improvement allowance of $1.8 million, which will resultresulted in additional rent amortized over the term of the lease at an annual rate of 8.0%. The lease also requires us to pay additional amounts for operating and maintenance expenses. In March 2019, in connection with the lease, we provided a $254,000$254,000 letter of credit as a security deposit, which is recorded as restricted cash in our accompanying Consolidated Balance Sheets.Sheets Cash Flows
The following is a summary. See further discussion of our cash flows (in thousands): operating lease in Note 9. |
| | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2019 | | 2018 | | 2017 |
Net cash used in operating activities | | $ | (35,346 | ) | | $ | (28,416 | ) | | $ | (16,572 | ) |
Net cash provided by (used in) investing activities | | 16,763 |
| | 32,118 |
| | (29,803 | ) |
Net cash provided by (used in) financing activities | | 24,255 |
| | (991 | ) | | 42,688 |
|
Net cash used in operating activities:
Net cash used in operating activities was $35.3 million for the year ended December 31, 2019 and consisted primarily of our net loss of $41.9 million. This was offset by increases of $3.2 million in our net operating assets and liabilities and $3.3 million in our net non-cash adjustments, which primarily relatesCertain credits received related to stock-based compensation, depreciation and amortization.
Net cash used in operating activities was $28.4 million for the year ended December 31, 2018 and consisted primarily of our net loss of $36.5 million. This was offset by increases of $5.0 million in our net operating assets and liabilities and $3.1 million in our net non-cash adjustments, which primarily relates to the loss on the sale of our intangible asset, stock-based compensation, the write-off of our deferred tax liability, depreciation and amortization.
Net cash used in operating activities was $16.6 million for the year ended December 31, 2017 and consisted primarily of our net loss of $7.8 million and bargain purchase gain of $6.6 million. We also had a $3.2 million decrease in our net operating assets and liabilities, partially offset by a $1.0 million increase in our net non-cash adjustments.
Net cash provided by (used in) investing activities:
Cash flows from investing activities primarily reflect cash used to purchase short-term investments and proceeds from the maturities of short-term investments, thus causing a shift between our cash and cash equivalents and short-term investment balances. We manage our cash usage with respect to our total cash, cash equivalents and short-term investments.
Net cash provided by investing activities was $16.8 million for the year ended December 31, 2019 and consisted primarily of the net maturities, sales and purchases of short-term investments in U.S. Treasury securities, commercial paper, and corporate debt securities, partially offset by purchases of property and equipment, primarily lab equipment, to support our research and development efforts.expenditures, which were previously recorded within other income within our
Net cash providedConsolidated Statements of Operations and Comprehensive Income (Loss), were subject to review by investing activities was $32.1foreign taxing authorities. We reached constructive agreement with the Australian Taxation Office during 2022 and recorded an estimated current foreign income tax provision of $1.3 million for the year ended December 31, 2018expected repayments, of which we paid $0.6 million, and consisted primarily of the net maturities and purchases of short-term investments in U.S. Treasury securities, commercial paper, and corporate debt securities, partially offset by purchases of property and equipment, primarily lab equipment, to support our research and development efforts.Net cash used in investing activities was $29.8remaining $0.8 million during the year ended December 31, 2017 and consisted primarily of the net purchases and sales of short-term investments, and purchases of property and equipment to build out our laboratory, partially offset by consideration acquired in the merger.
Net cash provided by (used in) financing activities:
Net cash provided by financing activities was $24.3 million for the year ended December 31, 2019 and consisted primarily of the net proceeds of $23.6 million related to the sale of approximately 4.7 million Units under our Purchase Agreement and $2.0 million in net proceeds from our debt refinancing, partially offset by $1.3 million in principal payments is included within accrued liabilities on our debt. accompanying Consolidated Balance Sheets.Net cash used in financing activities was $1.0 million for the year ended December 31, 2018 and consisted of principal payments on our debt, partially offset by the proceeds from stock option exercises.
Net cash provided by financing activities was $42.7 million for the year ended December 31, 2017 and consisted primarily of $37.7 million in proceeds from the sale of preferred stock and $5.0 million from the advance of a long-term loan.
Contractual Obligations and Contingent Liabilities
As a “smaller reporting company,” as defined by Rule 12b-2 of the Exchange Act, we are not required to provide additional information on our contractual obligations and contingent liabilities pursuant to Item 303 of Regulation S-K.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined under SEC rules.
JOBS Act
On April 5, 2012, the JOBS Act was enacted. Section 107 of the JOBS Act provides that an emerging growth company can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act of 1933, as amended, or the Securities Act, for complying with new or revised accounting standards. Thus, an emerging growth company can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies. In addition, for so long as we are an “emerging growth company,” which is until as late as December 31, 2020, we will, among other things not be required to comply with the auditor attestation requirements of Section 404(b) of Sarbanes-Oxley.
Critical Accounting Policies and Estimates
Our management’smanagement’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which we have prepared in accordance with generally accepted accounting principles in the United States, or GAAP. The preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the consolidated financial statements, as well as the reported revenues and expenses during the reporting periods. We evaluate these estimates and judgments on an ongoing basis. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in Note 2 to our consolidated financial statements, we believe that the following accounting policies are the most critical to fully understanding and evaluating our financial condition and results of operations. Accrued Liabilities
As part of the process of preparing our consolidated financial statements, we are required to estimate accruals for professional services and research and development expenses. This process involves reviewing contracts and vendor agreements and communicating with applicable personnel to identify services that have been performed on our behalf. We estimate the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of the actual cost. We estimate accrued liabilities as of each balance sheet date based on known facts and circumstances.
Although we do not expect our estimates to be materially different from amounts actually incurred, if our estimates of the status and timing of services performed differ from the actual status and timing of services performed, we may report amounts that are too high or too low in any particular period. To date, we have not experienced any significant adjustments to our estimates.
Revenue Recognition (effective January 1, 2018)
We adopted ASC 606 on January 1, 2018, resulting inRevenue recognition is a changecritical accounting estimate due to the magnitude and nature of the upfront cash and milestone payments we receive. Our primary source of revenue is derived from our accounting policy for revenue recognition. We used the modified retrospective method transition optioncollaboration and recognized the cumulative effect of adopting the new revenue standard as an adjustment to increase the opening accumulated deficit at January 1, 2018. Accordingly, 2017 comparative information has not been adjusted and continues to be reported under previous accounting standards.licensing agreements.
Revenue is recognized when control of the promised goods or services is transferred to our customers, in an amount that reflects the consideration we expect to be entitled to in exchange for those goods or services. Our steps for recognizing revenue consist of;of: (1) identifying the contract,contract; (2) identifying the performance obligations as either distinct or bundled goods and services,services; (3) determining the transaction price associated with each performance obligation for which we expect to be entitled in exchange for transferring such goods and services,services; (4) allocating the transaction price to the performance obligations in the contractcontract; and (5) recognizing revenue upon satisfaction of performance obligations.
Our collaboration agreements principally contain multiple performance obligations, which may include (1) grants of, or options to obtain, intellectual property licenses; (2) research and development services; and/or (3) manufacturing or supply services. Payments typically received under these arrangements typically include one or more of the following: non-refundable upfront license fees, option exercise fees, payment for research and/or development efforts, amounts due upon the achievement of specified objectives, and/or royalties on future product sales. Our revenue is primarily derived from our collaboration agreements with Adaptimmune, AbbVie, and Kite.Horizon. See further discussion of our collaboration agreements in Note 1210. We allocate revenue to each performance obligation based on its relative stand-alone selling price. We generally determine stand-alone selling prices at the inception of the contract based on our best estimate of what the selling price would be if the deliverable was regularly sold by us on a stand-alone basis. Payments received prior to satisfying the relevant revenue recognition criteria are recorded as deferred revenue in the accompanying Consolidated Balance Sheets and recognized as revenue when the related revenue recognition criteria are met. We recognize revenue under our collaboration agreements based onby using a cost-based input method to measure progress toward completion of the performance obligation, including employee hours contributed to each performance obligation.obligation, and to calculate the corresponding revenue to recognize each period. Our collaboration agreements provide for non-refundable milestone payments. We recognize revenue that is contingent upon the achievement of a substantive milestone in its entirety in the period in which the milestone is achieved. A milestone is
considered substantive when the consideration payable to us for such milestone (1) is consistent with our performance necessary to achieve the milestone or the increase in value to the collaboration resulting from our performance; (2) relates solely to our past performance; and (3) is reasonable relative to all of the other deliverables and payments within the arrangement. In making this assessment, we consider all facts and circumstances relevant to the arrangement, including factors such as the scientific, regulatory, commercial, and other risks that must be overcome to achieve the milestone, the level of effort and investment required to achieve the milestone and whether any portion of the milestone consideration is related to future performance or deliverables.
We review the contributed employee hours and progress towards completion for each performance obligation under our collaboration agreements and adjust the revenue recognized to reflect changes in assumptions relating to the estimated satisfaction of the performance obligation. We could accelerate revenueRevenue recognition may be accelerated in the event of early termination of programs or if our expectations change. Alternatively, we could decelerate revenue recognition may be decelerated if programs are extended or delayed. While such changes to our estimates have no impact on our reported cash flows, the timing of revenue recorded in future periods could be materially impacted.
Stock-based Compensation
Stock-based compensation is recognized for all share-based payments based on the estimated fair value as of the date of grant. The fair value of our stock options is calculated using the Black-Scholes option pricing model, which requires us to apply our judgment regarding certain key assumptions including risk-free interest rate, expected term, volatility and volatility.dividend yield. For risk-free interest rate, we use the zero-coupon U.S. Treasury instruments security rate with a term equal to the expected life of the option. We use the “simplified method”“simplified method” for options to determine the expected term of stock option granted to employees.options. Under this approach, the weighted-average expected life is presumed to be the average of the vesting term and the contractual term of the option. For volatility, we analyzedanalyze the stock price volatility of companies at a similar stage of development to estimate expected volatility of our stock price. Our assumed dividend yield is zero as we have never paid cash dividends and have no present intention to pay cash dividends. The fair value of restricted stock units, or RSUs, is based on the closing price of our common stock on the award date. Stock-based compensation expense is recognized over the requisite service period of the awards, usually the vesting period, on a straight-line basis. For performance-based awards where the vesting of the options may be accelerated upon the achievement of certain milestones, vesting and the related stock-based compensation is recognized as an expense when it is probable the milestone will be met. We recognize forfeitureforfeitures of awards as they occur rather than estimating the expected forfeiture rate.
If factors change and we employ different assumptions for estimating stock-based compensation expense in future periods, or if we decide to use a different valuation model, the stock-based compensation expense we recognize in future periods may differ significantly from what we have recorded in the current period and could materially affect our financial statements.
Recently Issued Accounting Pronouncements
In December 2019, the Financial Accounting Standards Board, or FASB, issued Accounting Standards Update, or ASU, No. 2019-12, Simplifying the Accounting for Income Taxes. The objective of the standard is to improve areas of GAAP by removing certain exceptions permitted by ASC Topic 740, Income Taxes
Income taxes are accounted for using an asset and clarifying existing guidance to facilitate consistent application. The standard will become effectiveliability approach that requires the recognition of deferred tax assets and liabilities for us beginning on January 1, 2021. We are currently evaluating the new standard to determineexpected future tax consequences of temporary differences between the potential impact on our financial condition, results of operations, cash flows, andconsolidated financial statement disclosures.
In November 2018,and tax bases of assets and liabilities at the FASB issued ASU No. 2018-18, Collaborative Arrangements: Clarifying the Interaction between Topic 808 and Topic 606. This ASU clarifies that certain transactions between collaborative arrangement participants should be accountedapplicable enacted tax rates. We establish a valuation allowance for as revenue when the collaborative arrangement participant is a customer in the context of a unit of account and precludes recognizing as revenue consideration received from a collaborative arrangement participant if the participant is not a customer. This ASU is effective for public companies for annual reporting periods and interim periods within those annual periods beginning after December 15, 2019. We do not anticipate the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
In August 2018, the FASB issued ASU No. 2018-13, Fair Value Measurement. ASU 2018-13 modifies the disclosure requirements for fair value measurements by removing, modifying, or adding certain disclosures. This ASU is effective for fiscal years beginning after December 15, 2019, and interim periods within those fiscal years, with early adoption permitted for any eliminated or modified disclosures. We do not expect the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
In June 2016,We recognize the FASB issued ASU 2016-13, Financial Instruments: Credit Losses, as clarified in ASU 2019-04 and ASU 2019-05. The objectivetax benefit from uncertain tax positions only if it is more likely than not that the tax position will be sustained on examination by the tax authorities, based on the technical merits of the standardposition. The tax position is measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement. We recognize interest and penalties related to provide information about expected credit losses on financial instruments at each reporting date and to change how other-than-temporary impairments on investment securities are recorded. The standard will become effective for us beginning on January 1, 2020. We do not anticipate the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.income tax matters in income tax expense if incurred.
Recently Adopted Accounting Pronouncements
In June 2018, the FASB issued ASU No. 2018-07, which aligns the measurement and classification guidance for share-based payments to nonemployees with the guidance for share-based payments to employees, with certain exceptions. Under the guidance, the measurement of equity-classified nonemployee awards will be fixed at the grant date. Upon transition, nonemployee awards will be required to be measured at fair value as of the adoption date with a cumulative-effect adjustment recognized in retained earnings as of the beginning of the annual period of adoption. ASU 2018-07 is effective for fiscal years beginning after December 15, 2018, including interim periods within that fiscal year. We adopted this standard on January 1, 2019 and it did not have a material impact on our financial statements and related disclosures.
In February 2016, the FASB issued ASU No. 2016-02, Leases. ASU 2016-02 requires a lessee to separate the lease components from the non-lease components in a contract and recognize in the statement of financial position a liability to make lease payments (the lease liability) and a right-of-use asset representing its right to use the underlying asset for the lease term. It also aligns lease accounting for lessors with the revenue recognition guidance in ASU 2014-09. We adopted this ASU effective January 1, 2019 and elected the modified retrospective method transition option, which permitted us not to restate the comparative period presented. Upon adoption, we recorded an operating lease right-of-use asset of $797,000, a corresponding operating lease liability of $883,000, and reduced our deferred rent balance by $86,000 to $0 on our accompanying Consolidated Balance Sheets; there was no effect on opening retained earnings, and we continue to account for leases in the prior period financial statements under ASC Topic 840. In adopting the new standard, we elected to apply the practical expedients regarding the identification of leases, lease classification, indirect costs, and the combination of lease and non-lease components.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
As a “smaller“smaller reporting company,”” as defined by Rule 12b-2 of the Exchange Act, and pursuant to Item 305 of Regulation S-K, we are not required to provide quantitative and qualitative disclosures about market risk.
Item 8. Financial Statements and Supplementary Data.
As a “smaller“smaller reporting company,”” as defined by Rule 12b-2 of the Exchange Act and pursuant to Article 8, Regulation X and Item 302 of Regulation S-K, we are permitted to provide scaled Item 8 disclosure.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
None.
Item 9A. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2019.2022. The term “disclosure“disclosure controls and procedures,”” as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, or the Exchange Act, means controls and other procedures of a company that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the Securities and Exchange Commission’sCommission’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company’scompany’s management, including its principal executive and principal financial officers, as appropriate to allow timely decisions regarding required
disclosure. Our management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and our management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2019,2022, our Chief Executive Officer and Chief Financial Officer concluded that, as of such date, our disclosure controls and procedures were effective at the reasonable assurance level.
Management’sManagement’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rule 13a-15(f) and Rule 15d-15(f) of the Exchange Act. Our internal control over financial reporting is a process to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Our internal control over financial reporting includes those policies and procedures that:
(i)pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets;
| |
(i)
| pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets;
|
| |
(ii)
| provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and
|
| |
(iii)
| provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
|
(ii)provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and
(iii)provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
The effectiveness of any system of internal control over financial reporting, including ours, is subject to inherent limitations, including the exercise of judgment in designing, implementing, operating, and evaluating the controls and procedures, and the inability to eliminate misconduct completely. Accordingly, any system of internal control over financial reporting, including ours, no matter how well designed and operated, can only provide reasonable, not absolute, assurances. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate. Our management assessed the effectiveness of our internal control over financial reporting as of December 31, 2019.2022. In making this assessment, our management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission, or COSO, Internal Control-Integrated Framework (2013). Based on our assessment using those criteria, our management has concluded that, as of December 31, 2019,2022, our internal control over financial reporting was effective.effective at the reasonable assurance level.
Changes in Internal Control Over Financial Reporting
No significant changes in our internal control over financial reporting occurred during the fiscal quarter ended December 31, 2019,2022, that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
We ceased to be an “emerging growth company” under the JOBS Act effective December 31, 2020. However, for so long as we are not classified as an “accelerated filer” or “large accelerated filer” pursuant to SEC rules, we will continue to be exempt from the auditor attestation requirements of Section 404(b) of Sarbanes-Oxley.
Item 9B. Other Information.
None.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not applicable.
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Executive Officers and Directors
The following table sets forthinformation required by Item 10 of Form 10-K is incorporated by reference to our Proxy Statement for the names, ages and positions2023 Annual Meeting of our executive officers and directors as of March 30, 2020:
|
| | | | | |
Name
|
| Age
|
| Position
|
Executive Officers
|
|
|
|
|
Mitchell H. Gold, M.D.
|
| 52
|
|
| Executive Chairman and Chief Executive Officer
|
Stanford Peng, M.D., Ph.D.
|
| 49
|
|
| President and Head of Research and Development
|
Paul Rickey
|
| 41
|
|
| Senior Vice President and Chief Financial Officer
|
Non-Employee Directors
|
|
|
|
|
Robert Conway
|
| 66
|
|
|
|
Min Cui, Ph.D.
|
| 51
|
|
|
|
Paul Sekhri
|
| 61
|
|
|
|
Peter Thompson, M.D.
|
| 60
|
|
|
|
James Topper, M.D., Ph.D.
|
| 58
|
|
|
|
Jay Venkatesan, M.D.
|
| 48
|
|
|
|
Christopher Peetz
|
| 41
|
|
|
|
Executive Officers
Mitchell H. Gold, M.D. has served as our executive chairman, chief executive officer and a member of our board of directors sinceStockholders to be filed with the completionSEC within 120 days after the end of the merger of Nivalis and Alpine in July 2017 and prior to the merger served as Alpine’s chief executive officer since June 2016 and as Alpine’s executive chairman and member of Alpine’s board of directors since January 2015. Prior to co-founding Alpine, Dr. Gold was Chairman and Founder of Alpine Biosciences, a privately-held biotech company, from 2012 to 2014. From 2001 to 2012, Dr. Gold served in a variety of roles with Dendreon Corporation (which was acquired by Valeant Pharmaceuticals International, Inc. through an asset purchase agreement), including President, Chief Executive Officer, and Chairman of the board of directors. Earlier in his career, Dr. Gold served as Vice President of Business Development at Data Critical from 2000 to 2001. From 1995 to 2000, Dr. Gold was President and Chief Executive Officer of Elixis Corporation. Dr. Gold is currently a Managing Partner at Alpine BioVentures. Dr. Gold holds an M.D. from Rush Medical College of Rush University Medical Center and a B.S. in Biology from the University of Wisconsin. We believe that Dr. Gold possesses specific attributes that qualify him to serve as a member of our board of directors, including more than 20 years of experience in senior executive management roles with both early stage and public biopharmaceutical companies.
Stanford Peng has served as our president and head of research and development since April 2019, prior to which he served as our executive vice president of research and development and chief medical officer since the completion of the merger of Nivalis and Alpine in July 2017, as Alpine’s chief medical officer from September 2016 to February 2017 and as Alpine’s executive vice president of research and development and chief medical officer from February 2017 until completion of the merger. Prior to joining Alpine, Dr. Peng was chief medical officer and head of clinical development at Stemcentrx, providing strategic oversight of the company’s clinical and translational programs from 2015 to 2016. Previously, Dr. Peng was executive medical director at Seattle Genetics where he developed multiple programs for antibody-drug conjugates from 2014 to 2015. Earlier in his career, he directed translational research and auto-immune related clinical trials as head of the Rheumatology Clinical Research Unit at the Benaroya Research Institute from 2009 to 2014 and served as senior director, clinical research and exploratory development at Roche from 2005 to 2008. Between 2009 and 2014, Dr. Peng also served as member physician at Virginia Mason Medical Center. Dr. Peng served as an assistant professor at the Washington University School of Medicine from 2002 to 2005. From 2008 to 2009, Dr. Peng served as senior director at ARYx Therapeutics, Inc. (Nasdaq: ARYX). Dr. Peng received an M.D. and Ph.D. in biology from the Yale University School of Medicine and a B.A. in music and B.S. in biological sciences from Stanford University.
Paul Rickey has served as our senior vice president and chief financial officer since the completion of the merger of Nivalis and Alpine in July 2017 and prior to the merger served as Alpine’s senior vice president and chief financial officer since
April 2017. Mr. Rickey previously served as chief financial officer of Sound Pharmaceuticals, overseeing finance, accounting and human resources from March 2016 to March 2017. Before joining Sound Pharmaceuticals, Mr. Rickey was vice president of finance and administration of Immune Design Corp. from 2009 to May 2015, which was a publicly traded biotechnology company for a portion of his time there, where he helped complete the company’s private offerings, initial public offering, and follow-on financing, and also oversaw the corporate development, accounting and human resource functions. Before joining Immune Design in 2009, Mr. Rickey was corporate controller of Northstar Neuroscience, a publicly-traded medical device company, where he managed the company’s finance and accounting groups following Northstar’s initial public offering. Prior to his role at Northstar Neuroscience, Mr. Rickey was the accounting manager for Mobliss, Inc., a mobile technology company that was sold to Index Corp., of Japan. Mr. Rickey started his finance career at Ernst & Young LLP. Mr. Rickey graduated from the University of Washington with a B.A. and Masters in Professional Accounting and is a certified public accountant, inactive.
Our Directors
Robert Conway has served as a member of our board of directors since the completion of the merger of Nivalis and Alpine in July 2017 and previously served as a member of the board of directors of Nivalis since April 2015. From 1999 to 2012, Mr. Conway served as the chief executive officer and member of the board of directors of Array BioPharma (Nasdaq: ARRY), a publicly traded biopharmaceutical company. Prior to joining Array, Mr. Conway was the chief operating officer and executive vice president of Hill Top Research, from 1996 to 1999. From 1979 until 1996, Mr. Conway held various executive positions for Corning Inc. (NYSE: GLW), including corporate vice president and general manager of Corning Hazleton, a contract research organization. Since 2013, Mr. Conway has served on the board of directors of ARCA BioPharma (Nasdaq: ABIO), a publicly traded biopharmaceutical company, and was elected Chairman in June 2014. From 2004 to 2013, Mr. Conway served on the board of directors of PRA International (Nasdaq: PRAH), which was a public company for a portion of his tenure there. Mr. Conway is also a member of the board of directors of Signant Health and Advarra, Inc., private clinical technology companies. In addition, Mr. Conway is a member of the strategic advisory committee of Genstar Capital. Mr. Conway received a B.S. in accounting from Marquette University in 1976. We believe that Mr. Conway’s experience and expertise in the pharmaceutical industry, pharmaceutical development and clinical trials, and corporate finance, governance, accounting and public company compliance give him the qualifications and skills to serve on our board of directors.
Min Cui, Ph.D. has served as a member of our board of directors since January 2019. Dr. Cui has served as managing director of Decheng Capital, an investment firm focused on life sciences companies, since he founded the firm in 2011. Prior to founding Decheng, Dr. Cui was an investment partner at Bay City Capital, an international life science venture capital firm in San Francisco. Dr. Cui was previously director of strategic investment for the Southern Research Institute, a not-for-profit research organization. Prior to that, Dr. Cui co-founded Pan Pacific Pharmaceuticals and Hucon Biopharmaceuticals, where he led efforts in discovery and development of several key technologies in the fields of oncology, cardiology, infectious and inflammatory diseases. Dr. Cui has served as a member of the board of directors of ARMO BioSciences, Inc., a publicly-traded immuno-oncology company acquired by Eli Lilly and Company in May 2018, from August 2017 to May 2018, and also currently serves on the boards of directors of several private companies. Dr. Cui holds a Ph.D. in Cancer Biology from Stanford University and a BS and MS in Molecular Biology from Peking University. We believe that Dr. Cui’s venture capital and management experience in the pharmaceuticals industry provides him with the qualifications and skills necessary to serve as a member of our board of directors. Dr. Cui was appointed to the Board pursuant to the terms of the Purchase Agreement.
Christopher Peetz has served as a member of our board of directors since April 2018. Mr. Peetz has been the president and chief executive officer of Mirum Pharmaceuticals, Inc. since March 2019 and president since November 2018. He has served as an entrepreneur-in-residence at Frazier Healthcare Partners since May 2017. He served as the chief executive officer of Flashlight Therapeutics, Inc. from May 2017 untilfiscal year ended December 2019 and served as chief financial officer and head of corporate development at Tobira Therapeutics, Inc., a publicly-traded biotechnology company acquired by Allergan plc in November 2016, from May 2014 to December 2016. Prior to joining Tobira Therapeutics, Mr. Peetz served as vice president, finance & corporate development of Jennerex Biotherapeutics, a private biopharmaceutical company. Prior to Jennerex, Mr. Peetz held various positions at Onyx Pharmaceuticals, Inc. (now Amgen), including oversight of financial planning and analysis, corporate strategy, product lifecycle management and commercial roles. Prior to Onyx, Mr. Peetz provided merger and acquisition advisory services at LaSalle Corporate Finance, a part of ABN AMRO, and held positions at Abgenix Inc. and Solazyme Inc. Mr. Peetz received an MBA from Stanford Graduate School of Business and a B.S.B.A. in Finance, International Business and French from Washington University in St. Louis. We believe Mr. Peetz’ experience in senior management positions in both business and finance and his experience supporting various corporate and financing transactions provide him with the qualifications and skills to serve on our board of directors.
Paul Sekhri has served as a member of our board of directors since the completion of the merger of Nivalis and Private Alpine in July 2017 and previously served as a member of the board of directors of Nivalis since February 2016. Mr. Sekhri has
been the president and chief executive officer of eGenesis, Inc., a private life sciences company, since January 2019. Before Mr. Sekhri joined eGenesis, he was the president and chief executive officer of Lycera Corp., a private biopharmaceutical company from February 2015 through January 2019. Prior to this position, he served as senior vice president, integrated care for Sanofi from April 2014 through January 2015, and as group executive vice president, global business development and chief strategy officer for Teva Pharmaceutical Industries, Ltd. from March 2013 to March 2014. Prior to joining Teva, Mr. Sekhri spent five years from January 2009 to February 2013, as operating partner and head of the biotechnology operating group at TPG Biotech, the life sciences venture capital arm of TPG Capital. From 2004 to 2009 Mr. Sekhri was founder, president, and chief executive officer of Cerimon Pharmaceuticals, Inc. Prior to founding Cerimon, Mr. Sekhri was president and chief business officer of ARIAD Pharmaceuticals, Inc. Previously, Mr. Sekhri spent four years at Novartis, as senior vice president, and head of global search and evaluation, business development and licensing for Novartis Pharma AG.
Mr. Sekhri has been a director on more than 24 private and public company boards, and is currently a member of the board of directors of Veeva Systems Inc. Mr. Sekhri is also the chairman of the board of Pharming N.V., Petra Pharma, Inc., Topas Therapeutics GmbH, and Compugen Ltd. Additionally, he serves on several non-profit boards including the BioExec Institute, Inc., the TB Alliance, Young Concert Artists, Inc., The English Concert in America (TECA), and the Caramoor Center for Music and the Arts. Mr. Sekhri also served as a member of the board of trustees of Carnegie Hall from 2010 to 2012, where he is now an active member of their Patrons Council. We believe that Mr. Sekhri’s extensive experience in operational and strategic drug development and his outstanding reputation and expertise in the biomedical community give him the qualifications and skills to serve as a director on our board of directors.
Peter Thompson, M.D. has served as a member of our board of directors since the completion of the merger of Nivalis and Private Alpine in July 2017 and previously served as a member of the board of directors of Private Alpine since June 2016. Dr. Thompson currently serves as a private equity partner for OrbiMed Advisors LLC, an investment firm focused on the healthcare sector, where he has also served as venture partner since joining in September 2010. Dr. Thompson is a co-founder of and has served as a member of the board of directors of Corvus Pharmaceuticals, Inc. (Nasdaq: CRVS) since December 2014. Dr. Thompson also served as a director of Adaptimmune Therapeutics plc (Nasdaq: ADAP), a biopharmaceutical company, from 2014 until June 2018. Dr. Thompson has served as a member of the board of directors of Prevail Therapeutics Inc. (Nasdaq: PRVL) since October 2017 and also currently serves on the boards of directors of several private companies. Dr. Thompson is a board-certified internist and oncologist and has served as Affiliate Professor of Neurosurgery at the University of Washington since 2010. Dr. Thompson co-founded and served as the chief executive officer of Trubion Pharmaceuticals, Inc., a biopharmaceutical company, from 2002 to 2009. Dr. Thompson previously held executive positions at Chiron Corporation and Becton Dickinson and served on the faculty of the National Cancer Institute following his medical staff fellowship there. Dr. Thompson holds a Sc. B. in Molecular Biology and Mathematics from Brown University and an M.D. from Brown University Medical School. We believe that Dr. Thompson’s venture capital and management experience in the pharmaceuticals industry provides him with the qualifications and skills necessary to serve as a member of our board of directors.
James N. Topper, M.D., Ph.D. has served as a member of our board of directors since the completion of the merger of Nivalis and Alpine in July 2017 and prior to the merger served as a member of the board of directors of Alpine since June 2016. Dr. Topper has been a partner with Frazier Healthcare Partners since August 2003, serving as general partner since 2005. Before joining Frazier Healthcare Partners, Dr. Topper served as head of the cardiovascular research and development division of Millennium Pharmaceuticals, Inc. and ran Millennium San Francisco (formerly COR Therapeutics, Inc.) from 2002 to 2003. Before the merger of COR and Millennium in 2002, Dr. Topper served as the vice president of biology at COR from 1999 to 2002. Dr. Topper currently serves as a member of the board of directors of AnaptysBio, Inc. (Nasdaq: ANAB), Allena Pharmaceuticals (Nasdaq: ALNA), Aptinyx, Inc. (Nasdaq: APTX) and Entasis Therapeutics Holdings Inc. (Nasdaq: ETTX) and has served on numerous other boards of directors, including Sierra Oncology, Inc. (formerly ProNai) (Nasdaq: SRRA), Amicus Therapeutics, Inc. (Nasdaq: FOLD), Portola Pharmaceuticals, Inc. (Nasdaq: PTLA), and La Jolla Pharmaceutical Company (Nasdaq: LJPC). Dr. Topper received his M.D. and Ph.D. in biophysics from Stanford University and his B.S. in biology from the University of Michigan. We believe that Dr. Topper’s experience overseeing Frazier Healthcare Partners’ investments in biotechnology, his experience in senior management positions, and his significant knowledge of industry, medical and scientific matters, provide Dr. Topper with the qualifications and skills to serve on our board of directors.
Jay Venkatesan, M.D. has served as a member of our board of directors since the completion of the merger of Nivalis and Alpine in July 2017, served as our president from July 2017 to August 2018 and previously served as Alpine’s chief executive officer from November 2015 to June 2016 and Alpine’s president from June 2016 to July 2017. Dr. Venkatesan also served as a member of Alpine’s board of directors since November 2015. Since May 2018, Dr. Venkatesan has served as chief executive officer and a member of the board of directors of Angion Biomedica, Inc., a private pharmaceutical company. Prior to joining Alpine, Dr. Venkatesan was the executive vice president and general manager of Oncothyreon, Inc. (now Cascadian Therapeutics, which was acquired by Seattle Genetics, Inc. in March 2018) from August 2014 to May 2015 following
Oncothyreon’s acquisition of Alpine Biosciences, where he served as co-founder and chief executive officer. Previously, Dr. Venkatesan was the founder, portfolio manager, and managing director of Ayer Capital Management, a global healthcare equity fund from 2008 to 2013. Prior to that, he was a director at Brookside Capital Partners from 2002 to 2007. Earlier in his career, Dr. Venkatesan was involved in healthcare investing at Partricof & Co. Ventures from 1995 to 1996 and consulting at McKinsey & Company from 1993 to 1995. Dr. Venkatesan is currently a managing partner at Alpine BioVentures. In addition, Dr. Venkatesan currently serves on the board of directors of CellBioTherapy and Exicure Therapeutics (Nasdaq: XCUR). Dr. Venkatesan received an M.D. from the University of Pennsylvania School of Medicine, an M.B.A. from the Wharton School of the University of Pennsylvania, and a B.A. in Chemistry from Williams College. We believe that Dr. Venkatesan possesses specific attributes that qualify him to serve as a member of our board of directors, including his experience on the boards of and in management positions with biopharmaceutical companies, including publicly-traded companies.
Code of Business Conduct and Ethics31, 2022.
We are committed to the highest standardshave a written code of integrityconduct and ethics in the way we conduct our business. Our board of directors has adopted a Code of Business Conduct and Ethics, whichthat applies to all of our employees,directors, officers and directors, including our chief executive officer, chief financial officer, and other executive and senior financial officers. Our Codeemployees. A copy of Business Conduct and Ethics establishes our policies and expectations with respect to a wide rangethe most up-to-date version of business conduct, including preparation and maintenance of financial and accounting information, compliance with laws and conflicts of interest. In accordance with our code of conduct each ofis available on our employees, officers and directors is required to report suspected or actual violations to the extent permitted by law. In addition, our board of directors has adopted separate policies and procedures concerning the receipt and investigation of complaints relating to accounting, internal accounting controls or auditing matters, which are administered by our audit committee. Our Code of Business Conduct and Ethics is posted on the Corporate Governance portion of ourcompany website located at https://ir.alpineimmunesciences.com/governance. We will postintend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding amendments to our Code of Business Conduct and Ethics or waivers of our Codecode of Business Conductconduct and Ethics for directors andethics that apply to our principal executive officers on the same website.
Audit Committee
The responsibilities of the audit committee include, but are not limited to, the following:
| |
•
| meeting with our independent auditors, our management team and such other personnel as it deems appropriate to conduct and assist with certain audit committee functions;
|
| |
•
| overseeing our accounting and financial reporting processes and audits of its financial statements;
|
| |
•
| deciding whether to appoint, retain or terminate our independent auditors, including the sole authority to approve all audit engagement fees and terms and to pre-approve all audit and permitted non-audit and tax services to be provided by the independent auditors;
|
| |
•
| reviewing and discussing with management and our independent auditors the financial statements of Alpine Immune Sciences, Inc, including certain disclosures, addressing any issues encountered in the course of the audit work, and evaluating the performance of our independent auditors;
|
| |
•
| discussing with management our earnings press releases,officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions by posting that information and any earnings guidance provided to analysts and ratings agencies;
|
| |
•
| discussing with Alpine Immune Sciences and the internal auditors (if any) our disclosure controls, internal accounting and financial controls and accounting policies and practices;
|
| |
•
| discussing with management any outsourcing of the internal audit function (if any), including selection of vendor, fees paid and areas to be audited;
|
| |
•
| establishing procedures for the receipt, retention and treatment of complaints received by us regarding certain accounting or audit matters;
|
| |
•
| establishing policies governing the hiring by us of any current or former employee of our independent auditors;
|
| |
•
| reviewing our compliance with applicable laws and regulations and to review and oversee our policies and procedures designed to promote and monitor regulatory compliance;
|
| |
•
| obtaining assurance from the independent auditors that the audit of the financial statements was conducted in a manner consistent with Section 10A of the Exchange Act;
|
| |
•
| reviewing, approving and overseeing transactions between us and any related person and any other potential conflict of interest situation;
|
| |
•
| administering our Whistleblower and Non-Retaliation Policy and responding to and resolving related complaints or concerns;
|
| |
•
| overseeing portions of our Code of Business Conduct and Ethics as designated by our board of directors;
|
| |
•
| providing our board of directors with the results of its monitoring and recommendations derived from its responsibilities;
|
| |
•
| reviewing and approving our investment policy;
|
| |
•
| providing the independent and internal auditors with access to the board of directors; and
|
| |
•
| producing the report required to be prepared for inclusion in our annual proxy statement.
|
Since April 2018 the audit committee has been composed of three directors: Messrs. Conway (chairman), Peetz and Sekhri. Our board of directors has determined that Mr. Conway is an “audit committee financial expert” as defined in the SEC rules and made a qualitative assessment of Mr. Conway’s level of knowledge and experience based on several factors, including his prior experience, business acumen and independence. Our board of directors has concluded that the composition of the audit committee meets the requirements for independence under the rules and regulations of Nasdaq and the SEC.
The audit committee met four times during 2019. The audit committee also meets periodically with our outside auditors without management present, at such times as it deems appropriate. Our board of directors has adopted a written charter for the audit committee in compliance with the applicable rules of the SEC and the Nasdaq listing standards and which is available on our website at https://ir.alpineimmunesciences.com/governance.address specified above.
Item 11. Executive Compensation.
Summary Compensation Table
The following table provides information regarding the compensationrequired by Item 11 of our named executive officers: |
| | | | | | | | | | | | | | | | | | | | | |
Name and Principal Position | | Year | | Salary ($) | | | | Option Awards ($)(1) | | | | Nonequity Incentive Plan Compensation ($)(2) | | All Other Compensation ($)(3) | | Total ($) |
Mitchell H. Gold, M.D. | | 2019 | | 485,000 |
| | | | 852,681 |
| | | | 218,250 |
| | — |
| | 1,555,931 |
|
Executive Chairman and Chief Executive Officer | | 2018 | | 400,000 |
| | | | 512,630 |
| | | | 130,000 |
| | — |
| | 1,042,630 |
|
Paul Rickey | | 2019 | | 370,000 |
| | | | 319,757 |
| | | | 119,788 |
| | — |
| | 809,545 |
|
Senior Vice President and Chief Financial Officer | | 2018 | | 335,000 |
| | | | 329,764 |
| | | | 86,500 |
| | — |
| | 751,264 |
|
Stanford Peng, M.D., Ph.D. | | 2019 | | 442,708 |
| | | | 319,757 |
| | (4) | | 208,126 |
| | — |
| | 970,591 |
|
President and Head of Research and Development | | 2018 | | 400,000 |
| | | | 1,532,873 |
| | | | 103,300 |
| | — |
| | 2,036,173 |
|
| |
(1)
| Amounts shown in this column do not reflect dollar amounts actually received by our named executive officers. Instead, these amounts reflect the aggregate grant date fair value of the stock options granted, computed in accordance with the provisions of FASB ASC Topic 718. For additional details regarding the assumptions and methodologies used to calculate the amounts reported, please see the discussion of equity awards contained in Note 13 to our consolidated financial statements contained elsewhere in this Annual Report on Form 10-K. |
| |
(2)
| 2019 amounts represent cash bonuses earned by the named executive officers pursuant to our Executive Incentive Compensation Plan, or Incentive Plan, for 2019 performance, paid in 2020. In lieu of 100% of such cash bonus amount, Dr. Gold was granted a restricted stock unit, or RSU, award for 67,569 shares of our common stock, representing an aggregate grant date value of $218,250. In lieu of 50% of such cash bonus amounts, Dr. Peng and Mr. Rickey were each granted an RSU award for 32,217 shares of our common stock and 18,543 shares of our common stock, respectively, representing an aggregate grant date value of $104,063 and $59,894, respectively, with the remaining amounts of the cash bonuses earned paid in cash in 2020. 2018 amounts represent cash bonuses earned under our 2018 performance bonus plan, paid in 2019.
|
| |
(3)
| Perquisites and personal benefits are excluded as the total value of all perquisites and personal benefits for each named executive officer is less than $10,000.
|
| |
(4)
| Reflects the probable value of $0 for Dr. Peng’s April 22, 2019 performance-based option award. The maximum value of such award is $231,160.
|
Executive Employment Arrangements
We entered into amended and restated executive employment agreements with each of Drs. Gold and Peng and Mr. Rickey effective January 1, 2018. Pursuant to these agreements, the annual base salaries for Drs. Gold and Peng and Mr. Rickey were $400,000, $400,000, and $335,000 respectively. In February 2019, the compensation committee approved adjusted salaries for Drs. Gold and Peng and Mr. Rickey of $485,000, $425,000 and $370,000, respectively. Dr. Peng’s salary was increased to $450,000 effective April 16, 2019, in connection with his appointment as our President and Head of Research and Development. In January 2020, the compensation committee approved adjusted salaries for Drs. Gold and Peng and Mr. Rickey of $500,000, $464,000, and $382,000 respectively. Additionally, Drs. Gold and Peng and Mr. Rickey are eligible to earn cash bonuses of up to 50%, 50% and 35%, respectively, of their base salary. The actual amount of such bonusesForm 10-K is tied to the achievement of various objectives for each year. Dr. Gold’s bonus is based solely on achievement of corporate objectives. The bonuses for Dr. Peng and Mr. Rickey are based on 75% corporate objectives and 25% individual objectives. These agreements also provide for certain severance benefits upon the termination of employment or a change in control of the company pursuantincorporated by reference to our Change in Control and Severance Policy, orProxy Statement for the Severance Policy.
Pursuant2023 Annual Meeting of Stockholders to the Severance Policy, if we terminate the employment of any of Dr. Gold, Dr. Peng or Mr. Rickey, each an Eligible Employee, other than for cause, death or disability, or the Eligible Employee resigns for good reason on or within 12 months following a change of control, then, subject to the Eligible Employee signing and not revoking a separation agreement and release of claims and continuing to adhere to the Eligible Employee’s non-competition, non-disclosure and invention
assignment agreement, such Eligible Employee will be eligible to receive the following severance benefits, less applicable tax withholdings:
| |
•
| A lump-sum payment totaling 100% (or, in case of Dr. Gold, 150%) of the Eligible Employee’s applicable annual base salary.
|
| |
•
| A lump-sum payment equal to (1) 100% of the Eligible Employee’s applicable target annual bonus plus (2) a payment equal to the Eligible Employee’s pro-rated applicable target annual bonus.
|
| |
•
| 100% of the Eligible Employee’s then-outstanding and unvested time-based equity awards will become vested and exercisable.
|
| |
•
| Payment or reimbursement of continued health coverage for the Eligible Employee and the Eligible Employee’s dependents under COBRA for a period of up to 12 months (or, in Dr. Gold’s case, 18 months).
|
Further, under the Severance Policy, if the Company terminates an Eligible Employee’s employment other than for cause, death or disability or such Eligible Employee resigns for good reason at any time other than during the period lasting from the date of a change of control or within 12 months thereafter, then, subject to the Severance Conditions, such Eligible Employee will be eligible to receive the following severance benefits, less applicable tax withholdings:
| |
•
| Continued payments totaling 75% (or, in Dr. Gold’s case, 100%) of the Eligible Employee’s applicable annual base salary over a period of 9 months (or in Dr. Gold’s case, 12 months).
|
| |
•
| 100% of the Eligible Employee’s then-outstanding and unvested time-based equity awards granted prior to the closing of the merger by and between Alpine Immune Sciences, Inc. and Nivalis Therapeutics, Inc. that would have otherwise vested during the 12-month period following the date of the Eligible Employee’s termination, and 0% in all other cases.
|
| |
•
| Payment or reimbursement of continued health coverage for the Eligible Employee and the Eligible Employee’s dependents under COBRA for a period of up to 9 months (or, in Dr. Gold’s case, 12 months).
|
Outstanding Equity Awards at Year-End
The following table provides information regarding the equity awards outstanding at December 31, 2019 held by each of our named executive officers: |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | Option Awards |
Name | | Vesting Commencement Date | | Grant Date | | Number of Securities Underlying Unexercised Options (#) Exercisable | | Number of Securities Underlying Unexercised Options (#) Unexercisable | | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) | | | | Option Exercise Price ($) | | Option Expiration Date |
Mitchell H. Gold | | 01/16/2015 | | 12/16/2015 | | 51,242 |
| | — |
| | — |
| | | | 0.45 |
| | 12/15/2025 |
| | 01/20/2017 | | 03/14/2017 | | 219,209 |
| | 81,415 |
| | — |
| | (1) | | 0.65 |
| | 03/13/2027 |
| | 01/20/2017 | | 04/12/2017 | | 152,340 |
| | 56,576 |
| | — |
| | (1) | | 5.02 |
| | 04/11/2027 |
| | 01/02/2018 | | 01/02/2018 | | 33,541 |
| | 36,459 |
| | — |
| | (1) | | 11.31 |
| | 01/01/2028 |
| | 02/06/2019 | | 02/06/2019 | | — |
| | 200,000 |
| | — |
| | (1) | | 6.51 |
| | 02/05/2029 |
Paul Rickey | | 04/01/2017 | | 04/12/2017 | | 49,693 |
| | 24,842 |
| | — |
| | (1) | | 5.02 |
| | 04/11/2027 |
| | 01/02/2018 | | 01/02/2018 | | 21,562 |
| | 23,438 |
| | — |
| | (1) | | 11.31 |
| | 01/01/2028 |
| | 02/06/2019 | | 02/06/2019 | | — |
| | 75,000 |
| | — |
| | (1) | | 6.51 |
| | 02/05/2029 |
Stanford Peng | | 09/06/2016 | | 09/22/2016 | | 131,216 |
| | 30,276 |
| | — |
| | (1) | | 0.65 |
| | 09/21/2026 |
| | 09/06/2016 | | 03/14/2017 | | 30,283 |
| | 6,984 |
| | — |
| | (1) | | 0.65 |
| | 03/13/2027 |
| | 01/02/2018 | | 01/02/2018 | | 31,145 |
| | 33,855 |
| | — |
| | (1) | | 11.31 |
| | 01/01/2028 |
| | 09/28/2018 | | 09/28/2018 | | — |
| | 250,000 |
| | — |
| | (2) | | 6.33 |
| | 09/27/2028 |
| | 02/06/2019 | | 02/06/2019 | | — |
| | 75,000 |
| | — |
| | (1) | | 6.51 |
| | 02/05/2029 |
| | 04/22/2019 | | 04/22/2019 | | — |
| | — |
| | 50,000 |
| | (3) | | 7.20 |
| | 04/21/2029 |
| |
(1)
| 1/4th of the shares will vest on the one-year anniversary of the vesting commencement date, and 1/36th of the remaining shares shall vest on each monthly anniversary thereafter, such that 100% of the shares shall be vested and exercisable as of the four-year anniversary of the vesting commencement date.
|
| |
(2)
| 1/2 of the shares subject to the option become vested and exercisable on October 1, 2020 and the balance of the shares subject to the option become vested and exercisable on October 1, 2022, subject to continued service through each such date.
|
| |
(3)
| 100% of the shares underlying the option will vest upon the achievement of specified performance goals that are achieved on or prior to April 16, 2023, as determined by the board of directors or the compensation committee of the board of directors.
|
401(k) Plan
We have adopted the WTIA 401(k) Multiple Employer Plan, maintained by Washington Technology Industry Association, which is a defined contribution retirement plan, in which all Alpine employees providing at least 20 hours of service a week are eligible to participate. This plan provides our eligible employees with an opportunity to save for retirement on a tax advantaged basis, and participants are able to defer a portion of their eligible compensation. All participants’ interests in their deferrals are 100% vested when contributed. The 401(k) plan permits us to make matching contributions and profit-sharing contributions to eligible participants. We have not provided a discretionary company match to employee contributions during the periods presented. Pre-tax contributions are allocated to each participant’s individual account and are then invested in selected investment alternatives according to the participants’ directions. The 401(k) plan is intended to qualify under Sections 401(a) and 501(a) of the Internal Revenue Code. As a tax-qualified retirement plan, contributions to the 401(k) plan and earnings on those contributions are not taxable to the employees until distributed from the 401(k) plan and all contributions are deductible by us when made.
Director Compensation
Director Compensation Policy
In July 2015, our board of directors approved a director compensation policy for our non-employee directors that became effective following our initial public offering and which was most recently amended in March 2019. For purposes of the policy, the board of directors classified each director into one of the two following categories: (1) an “employee director,” is a director who is employed by us; and (2) a “non-employee director,” is a director who is not an employee director. Only non-employee directors will receive compensation under the director compensation policy. Non-employee directors will receive compensation in the form of equity and cash under the director compensation policy, as described below. We believe our non-employee director compensation program provides reasonable compensation to our non-employee directors that is appropriately aligned with our peers and is commensuratefiled with the services and contributionsSEC within 120 days after the end of our non-employee directors.
Non-employee directors receive an initial stock option grant to purchase shares of our common stock upon appointment or election to the board of directors. Pursuant to the March 2019 amendments to the policy, the size of the initial stock option grant is 15,000 shares. Non-employee directors also receive on an annual basis, an additional stock option grant to purchase 7,650 shares. These annual grants occur on the first trading day in January of each year. All options are expected to have an exercise price equal to the closing price of our common stock as reported by Nasdaq on the date of grant subject to vesting in 36 equal monthly installments over a three-year period from the grant date for initial option grants, or in 12 equal monthly installments over a 12-month period from the grant date for annual stock option grants, subject to further evaluation by the compensation committee. On a change in control, all outstanding, unvested options held by non-employee directors are expected to vest in full.
Each non-employee director is eligible to receive the following cash annual retainer, which will be paid quarterly in arrears on a prorated basis.
|
| | | |
| |
Annual retainer for board membership | $ | 40,000 |
|
Annual retainer for board chairperson | 25,000 |
|
Annual retainer for audit committee chairperson | 15,000 |
|
Annual retainer for audit committee member | 7,500 |
|
Annual retainer for compensation committee chairperson | 10,000 |
|
Annual retainer for compensation committee member | 5,000 |
|
Annual retainer for nominating and corporate governance committee chairperson | 7,500 |
|
Annual retainer for nominating and corporate governance committee member | 3,750 |
|
2019 Director Compensation Table
The following table shows for the fiscal year ended December 31, 2019 certain information with respect to the compensation of our non-employee directors who served on our board of directors during any part of 2019.2022.
|
| | | | | | | | | | | | |
Name | | Fees Earned or paid in Cash ($) | | Option Awards ($)(1) | | Total ($) |
Robert Conway(2) | | $ | 58,750 |
| | $ | 19,259 |
| | $ | 78,009 |
|
Min Cui, Ph.D.(3) | | 40,986 |
| | 28,217 |
| | 69,203 |
|
Peter Thompson, M.D.(4) | | 52,500 |
| | 19,259 |
| | 71,759 |
|
James N. Topper, M.D., Ph.D.(5) | | 50,000 |
| | 19,259 |
| | 69,259 |
|
Paul Sekhri(6) | | 53,385 |
| | 19,259 |
| | 72,644 |
|
Christopher Peetz(7) | | 47,500 |
| | 19,259 |
| | 66,759 |
|
Jay Venkatesan, M.D.(8) | | 40,000 |
| | 19,259 |
| | 59,259 |
|
| |
(1)
| Amounts shown in this column do not reflect dollar amounts actually received by our non-employee directors. Instead, these amounts reflect the aggregate grant date fair value of the stock options granted, computed in accordance with the provisions of FASB ASC Topic 718.
|
| |
(2)
| As of December 31, 2019, Mr. Conway held outstanding options to purchase 23,155 shares of common stock.
|
| |
(3)
| As of December 31, 2019, Dr. Cui held outstanding options to purchase 7,650 shares of common stock.
|
| |
(4)
| As of December 31, 2019, Dr. Thompson held outstanding options to purchase 15,300 shares of common stock.
|
| |
(5)
| As of December 31, 2019, Dr. Topper held outstanding options to purchase 15,300 shares of common stock.
|
| |
(6)
| As of December 31, 2019, Mr. Sekhri held outstanding options to purchase 21,243 shares of commons stock.
|
| |
(7)
| As of December 31, 2019, Mr. Peetz held outstanding options to purchase 15,300 shares of common stock.
|
| |
(8)
| As of December 31, 2019, Dr. Venkatesan held outstanding options to purchase 122,713 shares of common stock.
|
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
Securities AuthorizedThe information required by Item 12 of Form 10-K is incorporated by reference to our Proxy Statement for Issuance Under Equity Compensation Plans
The following table provides information asthe 2023 Annual Meeting of Stockholders to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2019, with respect to the shares of our common stock that may be issued under existing equity compensation plans: 2022.
|
| | | | | | | | | | |
| | A | | B | | C |
Plan Category | | Number of Securities to be Issued Upon Exercise of Outstanding Options and Rights | | Weighted- Average Exercise Price of Outstanding Options and Rights | | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (Excluding Securities Reflected in Column A) (1) |
Equity compensation plans approved by security holders | | | | | | |
Amended and Restated 2015 Stock Plan, as amended, or the 2015 Stock Plan | | 1,369,752 |
| | $ | 3.49 |
| | — |
|
2015 Equity Incentive Plan | | 495,548 |
| | $ | 11.30 |
| | — |
|
2018 Equity Incentive Plan | | 1,399,266 |
| | $ | 6.35 |
| | 269,959 |
|
Employee Stock Purchase Plan | | — |
| | N/A |
| | 45,211 |
|
Total | | 3,264,566 |
| | $ | 5.90 |
| | 315,170 |
|
| |
(1)
| Represents the number of securities remaining available for future issuance under the 2015 Equity Incentive Plan, the 2015 Stock Plan, the 2018 Equity Incentive Plan and the Employee Stock Purchase Plan. The number of shares available for issuance under the 2018 Equity Incentive Plan is subject to an annual increase on the first day of each year equal to the lesser of (a) 1,500,000 shares or (b) 5% of the total number of shares of common stock outstanding on December 31 of the preceding calendar year or (c) a lesser number of shares of common stock approved by the board of directors prior to January 1 of a given year.
|
Principal Stockholders
The following table sets forth certain information with respect to the beneficial ownership of our common stock as of February 29, 2020 for:
| |
•
| each person who we know beneficially owns more than 5% of our common stock;
|
| |
•
| each of our named executive officers; and
|
| |
•
| all of our directors and executive officers as a group.
|
The percentage of beneficial ownership shown in the table is based upon 18,587,892 shares outstanding as of February 29, 2020.
We have determined beneficial ownership in accordance with the rules of the SEC. Except as indicated by the footnotes below, we believe, based on the information furnished to us, that the persons and entities named in the table below have sole voting and investment power with respect to all shares of common stock that they beneficially own, subject to applicable community property laws.
In computing the number of shares beneficially owned by a person and the percentage ownership of that person, we take into account shares of common stock issuable pursuant to stock options, warrants and restricted stock units that may be exercised or that are scheduled to vest on or before the 60th day after February 29, 2020. These shares are deemed to be outstanding and beneficially owned by the person holding those options, warrants or restricted stock units for the purpose of computing the percentage ownership of that person, but they are not treated as outstanding for the purpose of computing the percentage ownership of any other person.
Except as otherwise noted below, the address for each person or entity listed in the table is c/o Alpine Immune Sciences, Inc., 188 East Blaine Street, Suite 200, Seattle, Washington 98102.
|
| | | | | | |
| | Common Stock Beneficially Owned |
Name of Beneficial Owner | | Shares | | Percentage |
5% Stockholders: | | | | |
Alpine Immunosciences, L.P.(1) | | 4,069,222 |
| | 21.8 | % |
OrbiMed Private Investments VI, LP(2) | | 3,816,206 |
| | 20.4 | % |
Decheng Capital China Life Sciences USD Fund III, L.P.(3) | | 4,400,371 |
| | 22.2 | % |
Frazier Life Sciences VIII, L.P.(4) | | 2,716,701 |
| | 14.5 | % |
Entities affiliated with BVF Partners L.P.(5) | | 1,137,764 |
| | 6.1 | % |
Directors and Executive Officers: | | | | |
Mitchell H. Gold(6) | | 4,633,474 |
| | 24.1 | % |
Paul Rickey(7) | | 113,092 |
| | * |
|
Stanford Peng(8) | | 236,496 |
| | 1.3% |
|
Jay Venkatesan(9) | | 4,241,914 |
| | 22.6 | % |
Peter Thompson(10) | | 3,833,418 |
| | 20.4 | % |
James N. Topper(11) | | 2,733,913 |
| | 14.6 | % |
Robert Conway(12) | | 40,067 |
| | * |
|
Paul Sekhri(13) | | 23,155 |
| | * |
|
Christopher Peetz (14) | | 14,662 |
| | * |
|
Min Cui(15) | | 4,405,470 |
| | 22.2 | % |
All current directors and executive officers as a group (10 persons)(16) | | 16,206,439 |
| | 76.1 | % |
|
| |
(*)
| Less than one percent.
|
| |
(1)
| According to a Schedule 13D filed on January 23, 2019 with the Securities and Exchange Commission, Alpine BioVentures, GP, LLC, Mitchell H. Gold and Jay Venkatesan may be deemed to beneficially own 4,069,222 shares which are held by Alpine Immunosciences, L.P., including 74,441 shares issuable upon the exercise of warrants, which are exercisable within 60 days of February 29, 2020. Alpine BioVentures GP, LLC is the general partner of Alpine Immunosciences, L.P. Dr. Gold and Dr. Venkatesan are the Managing Partners of Alpine BioVentures GP, LLC. Dr. Gold and Dr. Venkatesan are also limited partners of Alpine Immunosciences, L.P. By virtue of such relationships, Dr. Gold and Dr. Venkatesan may be deemed to have voting and investment power with respect to the shares held by Alpine Immunosciences, L.P. and as a result may be deemed to have beneficial ownership of such shares. Each of Dr. Gold and Dr. Venkatesan disclaims beneficial ownership of the shares held by Alpine Immunosciences, L.P., except to the extent of his pecuniary interest therein, if any. The address for Alpine Immunosciences, L.P. is 600 Stewart Street, Suite 1503, Seattle Washington 98101.
|
| |
(2)
| According to a Schedule 13D filed on January 23, 2019 with the Securities and Exchange Commission, OrbiMed Advisors LLC and OrbiMed Capital GP VI LLC may be deemed to beneficially own 3,816,206 shares which are held by OrbiMed Private Investments VI, LP, including 145,251 shares issuable upon the exercise of warrants, which are exercisable within 60 days of February 29, 2020. OrbiMed Capital GP VI LLC (“GP VI”) is the general partner of OrbiMed Private Investments VI, LP. OrbiMed Advisors LLC (“OrbiMed Advisors”) is the managing member of GP VI. Carl L. Gordon, Sven H. Borho and Jonathan T. Silverstein share voting and investment power over the shares held by OrbiMed Private Investments VI, LP and as a result may be deemed to have beneficial ownership of such shares. Dr. Thompson, an employee of OrbiMed Advisors, may be deemed to have beneficial ownership of such shares. Each of GP VI, OrbiMed Advisors, Carl L. Gordon, Sven H. Borho, Jonathan T. Silverstein and Dr. Thompson disclaims beneficial ownership of the shares held by OrbiMed Private Investments VI, LP, except to the extent of its or his pecuniary interest therein, if any. The address for OrbiMed Private Investments VI, LP is 601 Lexington Avenue, 54th Floor, New York, New York 10022.
|
| |
(3)
| Decheng Capital Management III (Cayman), LLC (“Decheng Capital Management”) and Min Cui may be deemed to beneficially own 4,400,371 shares which are held by Decheng Capital China Life Sciences USD Fund III, L.P. (“Decheng”), including 1,234,636 shares issuable upon the exercise of warrants that are exercisable within 60 days of February 29, 2020. Decheng Capital Management is the general partner of Decheng. Dr. Cui is the sole manager of Decheng Capital Management and may be deemed to have voting and investment power with respect to the shares held
|
by Decheng and as a result may be deemed to have beneficial ownership of such shares. The address for Decheng is 3000 Sand Hill Road, Building 2, Suite 110, Menlo Park, California 94025.
| |
(4)
| According to a Schedule 13D filed on January 23, 2019 with the Securities and Exchange Commission, FHM Life Sciences VIII, L.P., FHM Life Sciences VIII, L.L.C., James Topper and Patrick J. Heron may be deemed to beneficially own 2,716,701 shares which are held by Frazier Life Sciences VIII, L.P., including 145,251 shares issuable upon the exercise of warrants that are exercisable within 60 days of February 29, 2020. FHM Life Sciences VIII, LP is the general partner of Frazier Life Sciences VIII, L.P. and FHM Life Sciences VIII, LLC is the general partner of FHM Life Sciences VIII, LP. Dr. Topper and Patrick J. Heron are the sole members of FHM Life Sciences VIII, LLC and therefore share voting and investment power over the shares held by Frazier Life Sciences VIII, L.P. Dr. Topper and Mr. Heron disclaim beneficial ownership of the shares held by Frazier Life Sciences VIII, L.P. except to the extent of their pecuniary interests in such shares, if any. The address for Frazier Life Sciences VIII, L.P. is 601 Union Street, Suite 3200, Seattle, Washington 98101.
|
| |
(5)
| According to a Schedule 13G filed on February 14, 2020 with the Securities and Exchange Commission, (i) Biotechnology Value Fund, L.P. (“BVF”) may be deemed to beneficially own 571,646 shares, (ii) Biotechnology Value Fund II, L.P. (“BVF2”) may be deemed to beneficially own 460,853 shares, and (iii) Biotechnology Value Trading Fund OS LP (“Trading Fund OS”) may be deemed to beneficially own 83,002 shares. BVF Partners OS Ltd. (“Partners OS”), as the general partner of Trading Fund OS, may be deemed to beneficially own the 83,002 shares held by Trading Fund OS. BVF Partners L.P. (“BVF Partners”), as the general partner of BVF, BVF2, the investment manager of Trading Fund OS, and the sole member of Partners OS, may be deemed to beneficially own the 1,137,764 shares beneficially owned in the aggregate by BVF, BVF2, Trading Fund OS, and a certain BVF Partners managed account (“Partners Managed Account”), including 22,263 shares held in the Partners Managed Account. BVF Inc., as the general partner of BVF Partners, may be deemed to beneficially own the 1,137,764 shares beneficially owned by BVF Partners. Mr. Mark N. Lampert, as a director and officer of BVF Inc., may be deemed to beneficially own the 1,137,764 shares beneficially owned by BVF Inc. These shares include an aggregate of 217,875 shares subject to warrants held by BVF, BVF2 and Trading Fund OS that are exercisable within 60 days of February 29, 2020. The warrants are only exercisable to the extent that the holder, together with its affiliates and any other person or entity acting as a group, would not beneficially own more than 4.99% of the outstanding shares of common stock after giving effect to such exercise, as such percentage ownership is determined in accordance with the terms of the warrants (the “Beneficial Ownership Limitation”). As a result, the table above excludes the 217,875 shares subject to warrants exercisable within 60 days of February 29, 2020 due to the Beneficial Ownership Limitation, based on the number of shares outstanding as February 29, 2020. The address of each of BVF Inc., Partners, BVF, BVF2, Trading Fund OS, Partners OS and Mr. Lampert is 44 Montgomery Street, 40th Floor, San Francisco, California 94104.
|
| |
(6)
| Consists of (i) 1,292 shares of our common stock held directly by Dr. Gold, (ii) 562,960 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020, (iii) 3,994,781 shares of our common stock held directly by Alpine Immunosciences, L.P and (iv) 74,441 shares of our common stock issuable upon the exercise of warrants held by Alpine Immunosciences, L.P. which are exercisable within 60 days of February 29, 2020. Please see footnote 1 regarding Dr. Gold’s voting and investment power over the shares held by Alpine Immunosciences, L.P.
|
| |
(7)
| Consists of (i) 10,000 shares of our common stock held directly by Mr. Rickey and (ii) 103,092 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020.
|
| |
(8)
| Consist of 236,496 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020.
|
| |
(9)
| Consists of (i) 21,739 shares of our common stock held directly by Dr. Venkatesan, (ii) 37,266 shares of our common stock held in trust for the benefit of Dr. Venkatesan’s children, (iii) 113,687 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020, (iv) 3,994,781 shares of our common stock held directly by Alpine Immunosciences, L.P., and (v) 74,441 shares of our common stock issuable upon the exercise of warrants held by Alpine Immunosciences, L.P. which are exercisable within 60 days of February 29, 2020. Please see footnote 1 regarding Dr. Venkatesan’s voting and investment power over the shares held by Alpine Immunosciences, L.P.
|
| |
(10)
| Consists of (i) 17,212 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020, (ii) 3,670,955 shares of our common stock held directly by OrbiMed Private Investments VI, LP and (iii) 145,251 shares of our common stock issuable upon the exercise of warrants held by OrbiMed Private Investments VI, LP which are exercisable within 60 days of February 29, 2020. Please see footnote 2 regarding Dr. Thompson’s voting and investment power over the shares held by OrbiMed Private Investments VI, LP.
|
(11) Consists of (i) 17,212 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020, (ii) 2,571,450 shares of our common stock held directly by Frazier Life Sciences VIII, L.P. and (iii) 145,251 shares of our common stock issuable upon the exercise of warrants held by Frazier Life Sciences VIII, L.P. which are exercisable within 60 days of February 29, 2020. Please see footnote 4 regarding Dr. Topper’s voting and investment power over the shares held by Frazier Life Sciences VIII, L.P.
(12) Consists of 15,000 shares of our common stock held directly by Mr. Conway and 25,067 shares of our common stock issuable upon exercise of options within 60 days of February 29, 2020.
| |
(13)
| Consist of 23,155 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020.
|
| |
(14)
| Consist of 14,662 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020.
|
| |
(15)
| Consists of (i) 5,099 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020, (ii) 3,165,735 shares of our common stock held directly by Decheng Capital China Life Sciences USD Fund III, L.P. and (iii) 1,234,636 shares of our common stock issuable upon the exercise of warrants held by Decheng Capital China Life Sciences USD Fund III, L.P. which are exercisable within 60 days of February 29, 2020. Please see footnote 3 regarding Dr. Cui’s voting and investment power over the shares held by Decheng Capital China Life Sciences USD Fund III, L.P.
|
| |
(16)
| Includes only current directors and executive officers serving in such capacity as of February 29, 2020. Includes 1,118,642 shares of our common stock issuable upon the exercise of options within 60 days of February 29, 2020 and 1,599,579 shares of our common stock issuable upon the exercise of warrants within 60 days of February 29, 2020.
|
Item 13. Certain Relationships and Related Transactions, and Director Independence.
Related Party Transaction Policy
Our boardThe information required by Item 13 of directors has adopted a written policy governingForm 10-K is incorporated by reference to our Proxy Statement for the review and approval2023 Annual Meeting of related party transactions. The audit committee of our board of directors hasStockholders to be filed with the primary responsibility for reviewing and approving or disapproving related party transactions, as designated inSEC within 120 days after the audit committee charter. In addition, our Code of Business Conduct and Ethics requires that each of our employees and directors inform his or her superior or the chairmanend of the audit committee, respectively, of any material transaction or relationship that comes to his or her attention that could reasonably be expected to create a conflict of interest. Further, at least annually, each director and executive officer will complete a detailed questionnaire that asks questions about any business relationship that may give rise to a conflict of interest and all transactions in which we are involved and in which an executive officer, a director or a related person has a direct or indirect material interest.fiscal year ended December 31, 2022.
Affiliations with Principal Stockholders
Dr. Gold is an executive officer, and Dr. Venkatesan was an executive officer until August 2018. Each of Drs. Venkatesan and Gold is also a member of our board of directors and, in their individual capacities, a limited partner of Alpine Immunosciences, L.P., Delaware limited partnership, which is a holder of more than 5% of our outstanding capital stock. In addition, each of Drs. Venkatesan and Gold, in their individual capacities, is a Managing Partner of Alpine BioVentures, GP, LLC, a Delaware limited liability company, which is the general partner of Alpine Immunosciences, L.P.
Dr. James N. Topper is a member of our board of directors and, in his individual capacity, is a managing member of FHM Life Sciences VIII, LLC, a Delaware limited liability company. FHM Life Sciences VIII, LLC is the general partner of FHM Life Sciences VIII, LP, a Delaware limited partnership. FHM Life Sciences VIII, LP is the general partner of Frazier Life Sciences VIII, L.P., a Delaware limited partnership, which is a holder of more than 5% of our outstanding capital stock.
Mr. Peetz is a member of our board of directors and, in his individual capacity, is an Entrepreneur-in-Residence at Frazier Healthcare Partners, which is an affiliate of Frazier Life Sciences VIII, L.P.
Dr. Peter Thompson is a member of our board of directors and, in his individual capacity, is an employee of OrbiMed Advisors LLC. OrbiMed Advisors LLC is the managing member of OrbiMed Capital GP VI LLC. OrbiMed Capital GP VI LLC is the general partner of OrbiMed Private Investments VI, LP, which is a holder of more than 5% of our outstanding capital stock.
Dr. Min Cui is a member of our board of directors and, in his individual capacity, is the manager of Decheng Capital Management III (Cayman), LLC, which in turn is the general partner of Decheng Capital China Life Sciences USD Fund III, L.P. Decheng Capital China Life Sciences USD Fund III, L.P. is a holder of more than 5% of our outstanding capital stock.
Indemnification Agreements
We have entered into indemnification agreements with each of our directors and with each of our executive officers. Pursuant to the indemnification agreements, we have agreed to indemnify and hold harmless these directors and officers to the fullest extent permitted by applicable law. The agreements generally cover expenses that a director or officer incurs or amounts that a director or officer becomes obligated to pay because of any proceeding to which he or she is made or threatened to be made a party or participant by reason of his or her service as a current or former director, officer, employee or agent of Alpine Immune Sciences. The agreements also provide for the advancement of expenses to the directors and executive officers subject to specified conditions. There are certain exceptions to our obligation to indemnify the directors and officers, including any intentional misconduct or act where the director or officer did not in good faith believe he or she was acting in our best interests, with respect to “short-swing” profit claims under Section 16(b) of the 1934 Act and, with certain exceptions, with respect to proceedings that he or she initiates.
Private Placement
On January 15, 2019, we entered into a securities purchase agreement in connection with the sale and issuance of 4,706,700 units for $5.37 per unit representing (i) 4,706,700 shares of our common stock and (ii) warrants to purchase an additional 1,835,610 shares of common stock for $12.74 per share, with each unit consisting of one share of common stock and a warrant to purchase 0.39 of a share of common stock.
The following table summarizes the purchases on January 18, 2019 of our securities by our 5% stockholders:
|
| | | | | | | | | | |
Name of Purchaser | | Number of Shares of Common Stock Purchased | | Number of Shares of Common Stock Subject to Warrants | | Aggregate Purchase Price |
Alpine Immunosciences, L.P. | | 190,875 |
| | 74,441 |
| | $ | 1,024,998.75 |
|
OrbiMed Private Investments VI, LP | | 372,439 |
| | 145,251 |
| | $ | 1,999,997.43 |
|
Frazier Life Sciences VIII, L.P. | | 372,439 |
| | 145,251 |
| | $ | 1,999,997.43 |
|
We also entered into a registration rights agreement requiring us to register the resale of the shares of common stock issued and issuable upon the exercise of warrants. We filed a registration statement on Form S-1 on March 18, 2019 covering the resale of such shares, which registration statement was declared effective by the Securities and Exchange Commission on April 4, 2019.
Indebtedness of Directors and Officers
None of our current or former directors or executive officers is indebted to us, nor are any of these individuals indebted to another entity which indebtedness is the subject of a guarantee, support agreement, letter of credit or other similar arrangement or understanding provided by us.
Other Transactions
We have granted stock options and/or restricted stock units to our named executive officers, other executive officers and our directors.
Director Independence
Our board of directors has undertaken a review of its composition, the composition of its committees and the independence of current directors and considered whether any such director has a material relationship with us that could compromise his ability to exercise independent judgment in carrying out his responsibilities. Based upon information requested from and provided by each current director concerning his background, employment and affiliations, including family relationships, the board of directors has determined that (1) none of our current directors except for Drs. Gold and Venkatesan, has a relationship which would interfere with the exercise of independent judgment in carrying out the responsibilities of a director and that each of these directors is an “independent director” as defined under the rules of the Nasdaq. The board of directors also determined that Messrs. Conway (chairman), Sekhri and Peetz, who comprise our audit committee, Drs. Topper (chairman) and Thompson and Mr. Sekhri, who comprise our compensation committee, and Drs. Thompson (chairman) and Cui and Mr. Conway, who comprise our nominating and corporate governance committee, satisfy the independence standards for those committees established by applicable SEC rules and the rules of Nasdaq.
Item 14. Principal Accounting Fees and Services.
The following table presents fees for professional audit services and other services renderedinformation required by Item 14 of Form 10-K is incorporated by reference to our company by EYProxy Statement for the years2023 Annual Meeting of Stockholders to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2019 and 2018.2022.
|
| | | | | | | | |
| | Year Ended December 31, |
Fee Category | | 2019 | | 2018 |
Audit fees(1) | | $ | 393,778 |
| | $ | 490,171 |
|
Audit-related fees(2) | | — |
| | — |
|
Tax fees(3) | | — |
| | — |
|
All other fees(4) | | — |
| | — |
|
Total fees | | $ | 393,778 |
| | $ | 490,171 |
|
| |
(1)
| Audit fees consist of fees for professional services provided in connection with the audit of our annual consolidated financial statements, review of our quarterly consolidated financial statements, procedures for comfort letters, consents and assistance with and review of documents filed with the SEC.
|
| |
(2)
| Audit-related fees consist of assurance and related services that are reasonably related to the performance of the audit or review of our financial statements and are not reported under “Audit Fees”.
|
| |
(3)
| Tax fees consist of fees associated with tax compliance, tax advice and tax planning fees.
|
| |
(4)
| All other fees include any fees billed that are not audit fees, audit-related fees or tax fees.
|
All fees described above were pre-approved by the audit committee.
Audit Committee Policy on Pre-Approval of Audit and Permissible Non-Audit Services of Independent Registered Public Accounting Firm
Pursuant to its charter, the audit committee must review and approve, in advance, the scope and plans for the audits and the audit fees and approve in advance (or, where permitted under the rules and regulations of the SEC, subsequently) all non-audit services to be performed by the independent auditor that are not otherwise prohibited by law and any associated fees. All fees paid to EY for 2019 and 2018 were pre-approved by our audit committee. The audit committee may delegate to one or more members of the committee the authority to pre-approve audit and permissible non-audit services, as long as this pre-approval is presented to the full committee at scheduled meetings.
PART IV
Item 15. Exhibits, Financial Statement Schedules.
(a)The financial statements, schedules and exhibits filed as a part of this Annual Report on Form 10-K are as follows:
(a)Financial statements – The financial statements included in Item 8 are filed as part of this Annual Report on Form 10-K.
(b)Financial Statement Schedules – All schedules have been omitted because they are not applicable or required, or the information required to be set forth therein is included in the consolidated financial statements or notes thereto included in Item 8 of this Annual Report on Form 10-K. (c)Exhibits – The exhibits required to be filed as part of this report are listed in the Exhibit List attached hereto and are incorporated herein by reference.
| |
(a)
| The financial statements, schedules and exhibits filed as a part of this Annual Report on Form 10-K are as follows:
|
| |
(a)
| Financial statements – The financial statements included in Item 8 are filed as part of this Annual Report on Form 10-K.
|
| |
(b)
| Financial Statement Schedules – All schedules have been omitted because they are not applicable or required, or the information required to be set forth therein is included in the consolidated financial statements or notes thereto included in Item 8 of this Annual Report on Form 10-K. |
| |
(c)
| Exhibits – The exhibits required to be filed as part of this report are listed in the Exhibit List attached hereto and are incorporated herein by reference.
|
INDEX TO EXHIBITS
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Incorporated by Reference |
| Exhibit Number | | Description | | Form | | File No. | | Exhibit | | Filing Date |
| 3.1 | | | | 10-K | | 001-37449 | | 3.1 | | March 28, 2018 |
| 3.2 | | | | 8-K | | 001-37449 | | 3.1 | | January 26, 2023 |
| 4.1 | | | | 10-K | | 001-37449 | | 4.1 | | March 28, 2018 |
| 4.2 | | | | 10-K | | 001-37449 | | 4.5 | | March 28, 2018 |
| 4.3 | | | | 10-K | | 001-37449 | | 4.6 | | March 28, 2018 |
| 4.4 | | | | 8-K | | 001-37449 | | 10.3 | | January 16, 2019 |
| 4.5 | | | | 8-K | | 001-37449 | | 4.1 | | August 28, 2019 |
| 4.6 | | | | 8-K | | 001-37449 | | 10.3 | | July 24, 2020 |
| 4.7+ | | | | | | | | | | |
| 4.8 | | | | 8-K | | 001-37449 | | 10.3 | | September 14, 2021 |
| 10.1* | | | | S-8 | | 333-205220 | | 4.4 | | June 25, 2015 |
| 10.2* | | | | S-8 | | 333-205220 | | 4.5 | | June 25, 2015 |
| 10.3* | | | | S-8 | | 333-205220 | | 4.6 | | June 25, 2015 |
| 10.4* | | | | S-8 | | 333-205220 | | 4.7 | | June 25, 2015 |
| 10.5* | | | | S-1 | | 333-204127 | | 10.18 | | May 13, 2015 |
| 10.6* | | | | 10-K | | 001-37449 | | 10.6 | | March 18, 2021 |
| 10.7* | | | | 8-K | | 001-37449 | | 10.1 | | December 11, 2017 |
|
| | | | | | | | | | |
| | | | Incorporated by Reference |
Exhibit Number | | Description | | Form | | File No. | | Exhibit | | Filing Date |
2.1† | | | | 8-K | | 001-37449 | | 2.1 | | April 18, 2017 |
3.1 | | | | 10-K | | 001-37449 | | 3.1 | | March 28, 2018 |
3.2 | | | | S-1 | | 333-204127 | | 3.4 | | May 13, 2015 |
4.1 | | | | 10-K | | 001-37449 | | 4.1 | | March 28, 2018 |
4.2 | | | | S-1 | | 333-204127 | | 4.2 | | May 13, 2015 |
4.3 | | | | S-1 | | 333-204127 | | 4.3 | | May 13, 2015 |
4.4 | | | | 10-K | | 001-37449 | | 4.5 | | March 28, 2018 |
4.5 | | | | 10-K | | 001-37449 | | 4.6 | | March 28, 2018 |
4.6 | | | | 8-K | | 001-37449 | | 10.3 | | January 16, 2019 |
4.7 | | | | 8-K | | 001-37449 | | 4.1 | | August 28, 2019 |
4.8+ | | | | | | | | | | |
10.1* | | | | S-8 | | 333-205220 | | 4.4 | | June 25, 2015 |
10.2* | | | | S-8 | | 333-205220 | | 4.5 | | June 25, 2015 |
10.3* | | | | S-8 | | 333-205220 | | 4.6 | | June 25, 2015 |
10.4* | | | | S-8 | | 333-205220 | | 4.7 | | June 25, 2015 |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Incorporated by Reference |
| Exhibit Number | | Description | | Form | | File No. | | Exhibit | | Filing Date |
| 10.8* | | | | 10-K | | 001-37449 | | 10.33 | | March 28, 2018 |
| 10.9* | | | | 10-K | | 001-37449 | | 10.35 | | March 28, 2018 |
| 10.10* | | | | 10-K | | 001-37449 | | 10.37 | | March 28, 2018 |
| 10.11* | | | | 10-Q | | 001-37449 | | 10.1 | | August 11, 2022 |
| 10.12* | | | | 10-Q | | 001-37449 | | 10.1 | | November 14, 2022 |
| 10.13* | | | | S-8 POS | | 333-218134 | | 4.1 | | September 11, 2017 |
| 10.14* | | | | S-8 POS | | 333-218134 | | 4.2 | | September 11, 2017 |
| 10.15* | | | | 8-K | | 001-37449 | | 10.1 | | June 17, 2020 |
| 10.16* | | | | 8-K | | 001-37449 | | 10.2 | | June 14, 2018 |
| 10.17* | | | | 8-K | | 001-37449 | | 10.1 | | January 27, 2020 |
| 10.18 | | | | 8-K | | 001-37449 | | 10.1 | | January 16, 2019 |
| 10.19* | | | | 8-K | | 001-37449 | | 10.1 | | April 1, 2019 |
| 10.20 | | | | 10-Q | | 001-37449 | | 10.6 | | May 9, 2019 |
| 10.21 | | | | 8-K | | 001-37449 | | 10.1 | | August 28, 2019 |
| 10.22** | | | | 10-Q | | 001-37449 | | 10.2 | | August 11, 2020 |
| 10.23 | | | | 8-K | | 001-37449 | | 10.1 | | July 24, 2020 |
| 10.24 | | | | 8-K | | 001-37449 | | 10.2 | | July 24, 2020 |
| 10.25 | | | | 8-K | | 001-37449 | | 10.1 | | September 14, 2021 |
| 10.26 | | | | 8-K | | 001-37449 | | 10.2 | | September 14, 2021 |
| 10.27 | | | | 8-K | | 001-37449 | | 10.4 | | September 14, 2021 |
| 10.28** | | | | 10-K | | 001-37449 | | 10.30 | | March 17, 2022 |
|
| | | | | | | | | | |
| | | | Incorporated by Reference |
Exhibit Number | | Description | | Form | | File No. | | Exhibit | | Filing Date |
10.5* | | | | S-1 | | 333-204127 | | 10.18 | | May 13, 2015 |
10.6* | | | | 8-K | | 001-37449 | | 10.2 | | April 1, 2019 |
10.7* | | | | 8-K | | 001-37449 | | 10.1 | | December 11, 2017 |
10.8* | | | | 10-K | | 001-37449 | | 10.33 | | March 28, 2018 |
10.9* | | | | 10-K | | 001-37449 | | 10.35 | | March 28, 2018 |
10.10* | | | | 10-K | | 001-37449 | | 10.37 | | March 28, 2018 |
10.11* | | | | S-8 POS | | 333-218134 | | 4.1 | | September 11, 2017 |
10.12* | | | | S-8 POS | | 333-218134 | | 4.2 | | September 11, 2017 |
10.13 | | | | 8-K | | 001-37449 | | 1.1 | | June 11, 2018 |
10.14* | | | | 8-K | | 001-37449 | | 10.1 | | June 14, 2018 |
10.15* | | | | 8-K | | 001-37449 | | 10.2 | | June 14, 2018 |
10.16* | | | | 8-K | | 001-37449 | | 10.1 | | January 27, 2020 |
10.17 | | | | 8-K | | 001-37449 | | 10.1 | | January 16, 2019 |
10.18 | | | | 8-K | | 001-37449 | | 10.2 | | January 16, 2019 |
10.19* | | | | 8-K | | 001-37449 | | 10.1 | | April 1, 2019 |
10.20 | | | | 10-Q | | 001-37449 | | 10.6 | | May 9, 2019 |
10.21* | | | | 10-Q | | 001-37449 | | 10.1 | | August 13, 2019 |
10.22 | | | | 8-K | | 001-37449 | | 10.1 | | August 28, 2019 |
21.1 | | | | 10-K | | 001-37449 | | 21.1 | | March 28, 2018 |
23.1+ | | | | | | | | | | |
24.1+ | | | | | | | | | | |
31.1+ | | | | | | | | | | |
31.2+ | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Incorporated by Reference |
| Exhibit Number | | Description | | Form | | File No. | | Exhibit | | Filing Date |
| 10.29 | | | | 10-K | | 001-37449 | | 10.31 | | March 17, 2022 |
| 10.30 | | | | 10-Q | | 001-37449 | | 10.2 | | August 11, 2022 |
| 21.1+ | | | | | | | | | | |
| 23.1+ | | | | | | | | | | |
| 24.1+ | | | | | | | | | | |
| 31.1+ | | | | | | | | | | |
| 31.2+ | | | | | | | | | | |
| 32.1+ | | | | | | | | | | |
| 32.2+ | | | | | | | | | | |
| 101.INS+ | | Inline XBRL Instance Document | | | | | | | | |
| 101.SCH+ | | Inline XBRL Taxonomy Extension Schema Document | | | | | | | | |
| 101.CAL+ | | Inline XBRL Taxonomy Extension Calculation Linkbase Document | | | | | | | | |
| 101.LAB+ | | Inline XBRL Taxonomy Extension Label Linkbase Document | | | | | | | | |
| 101.PRE+ | | Inline XBRL Taxonomy Extension Presentation Linkbase Document | | | | | | | | |
| 101.DEF+ | | Inline XBRL Taxonomy Extension Definition Linkbase Document | | | | | | | | |
| 104 | | Cover page formatted as Inline XBRL and contained in Exhibit 101 | | | | | | | | |
|
| | | | | | | | | | |
|
|
|
| Incorporated by Reference
|
Exhibit Number
|
| Description
|
| Form
|
| File No.
|
| Exhibit
|
| Filing Date
|
32.1+
|
| |
|
|
|
|
|
|
|
|
32.2+
|
| |
|
|
|
|
|
|
|
|
101.INS+
|
| Inline XBRL Instance Document
|
|
|
|
|
|
|
|
|
101.SCH+
|
| Inline XBRL Taxonomy Extension Schema Document
|
|
|
|
|
|
|
|
|
101.CAL+
|
| Inline XBRL Taxonomy Extension Calculation Linkbase Document
|
|
|
|
|
|
|
|
|
101.LAB+
|
| Inline XBRL Taxonomy Extension Label Linkbase Document
|
|
|
|
|
|
|
|
|
101.PRE+
|
| Inline XBRL Taxonomy Extension Presentation Linkbase Document
|
|
|
|
|
|
|
|
|
101.DEF+
|
| Inline XBRL Taxonomy Extension Definition Linkbase Document
|
|
|
|
|
|
|
|
|
104
|
| Cover page formatted as Inline XBRL and contained in Exhibit 101
|
|
|
|
|
|
|
|
|
|
| |
* | Indicates a management contract or a compensatory plan, contract or arrangement. |
|
| | | | |
†+
| All schedules and exhibits to the Merger Agreement have been omitted pursuant to Item 601(b)(2) of Regulation S-K. A copy of any omitted schedule and/or exhibit will be furnished to the Securities and Exchange Commission upon request.Filed herewith.
|
|
| |
#
| Portions of this exhibit have been omitted pursuant to a request forin accordance with Item 601(b)(10) of Regulation S-K because they are private, confidential treatment and the omitted portions have been filed separately with the Securities and Exchange Commission. not material. |
Item 16. Form 10-K Summary.
Not Applicable
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
| | | | | | | | | | | | | |
| | | ALPINE IMMUNE SCIENCES, INC. |
| | | | |
Date: | March 30, 202023, 2023 | | By: | /s/ Mitchell H. Gold, M.D. |
| | | Name: | Mitchell H. Gold, M.D. |
| | | Title | Executive Chairman and Chief Executive Officer |
| | | |
| | | ALPINE IMMUNE SCIENCES, INC. |
| | | | |
Date: | March 30, 202023, 2023 | | By: | /s/ Paul Rickey |
| | | Name: | Paul Rickey |
| | | Title: | Senior Vice President and Chief Financial Officer |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Mitchell H. Gold, M.D. and Paul Rickey, and each of them, with full power of substitution and resubstitution and full power to act without the other, as his or her true and lawful attorney-in-fact and agent to act in his or her name, place and stead and to execute in the name and on behalf of each person, individually and in each capacity stated below, and to file, any and all documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing, ratifying and confirming all that said attorneys-in-fact and agents or any of them or their and his or her substitute or substitutes, may lawfully do or cause to be done by virtue thereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this Report has been signed below by the following persons on behalf of the Registrant in the capacities and on the dates indicated.
|
| | | | | | | | | | | | | |
Name | | Title | | Date |
| | | | |
/s/ Mitchell H. Gold, M.D. | | Chief Executive Officer and Executive Chairman of the Board of Directors (Principal Executive Officer) | | March 30, 202023, 2023 |
Mitchell H. Gold, M.D. | | | | |
| | | | |
/s/ Paul Rickey | | Senior Vice President and Chief Financial Officer (Principal Accounting and Financial Officer) | | March 30, 202023, 2023 |
Paul Rickey | | | | |
| | | | |
/s/ Peter Thomson, M.D. | | Director | | March 30, 202023, 2023 |
Peter Thompson, M.D. | | | | |
| | | | |
/s/ James N. Topper, M.D., Ph.D. | | Director | | March 30, 202023, 2023 |
James N. Topper, M.D., Ph.D. | | | | |
| | | | |
/s/ Jay Venkatesan, M.D.
|
| Director
|
| March 30, 2020
|
Jay Venkatesan, M.D.
|
|
|
|
|
|
|
|
|
|
/s/ Robert Conway | | Director | | March 30, 202023, 2023 |
Robert Conway | | | | |
| | | | |
/s/ Paul SekhriNatasha Hernday | | Director | | March 30, 202023, 2023 |
Paul SekhriNatasha Hernday
| | | | |
| | | | |
| /s/ Christopher Peetz | | Director | | March 23, 2023 |
| Christopher Peetz | | | | |
| | | | |
| | Director | | |
| Xiangmin Cui, Ph.D. | | | | |
| | | | |
/s/ Christopher Peetz |
| Director
|
| March 30, 2020
|
Christopher Peetz
|
|
|
|
|
|
|
|
|
|
/s/ Min Cui,Jörn Drappa, M.D., Ph.D.
| | Director | | March 30, 202023, 2023 |
Min Cui, Jörn Drappa, M.D., Ph.D.
| | | | |
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Report of Independent Registered Public Accounting Firm
To the ShareholdersStockholders and the Board of Directors of Alpine Immune Sciences, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Alpine Immune Sciences, Inc. (the Company) as of December 31, 20192022 and 2018,2021, the related consolidated statements of operations and comprehensive income (loss), convertible preferred stock and shareholders' stockholder’s equity (deficit) and cash flows for each of the three years in the period ended December 31, 2019,2022, and the related notes (collectively(collectively referred to as the “consolidated“consolidated financial statements”statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 20192022 and 2018,2021, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2019,2022, in conformity with U.S. generally accepted accounting principles.
Adoption of New Accounting Standard
As discussed in Note 2 to the consolidated financial statements, the Company has changed its method for accounting for leases in 2019 due to the adoption of ASU No. 2016-02, Leases (Topic 842).Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’sCompany’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the account or disclosure to which it relates.
| | | | | |
| Accounting for Revenue under the AbbVie and Horizon Collaboration Agreements |
| Description of the Matter | As discussed in Note 10 to the consolidated financial statements, the Company entered into license and collaboration agreements with AbbVie Ireland Unlimited Company and Horizon Therapeutics Ireland DAC in June 2020 and December 2021, respectively. Collaboration revenue recognized from the agreements was $29.0 million for the year ended December 31, 2022. The Company recognized revenue under both agreements by using a cost-based input method to measure progress toward completion of the performance obligation and to calculate the corresponding revenue to recognize each period.
Auditing the Company’s estimated measure of progress toward completion of the performance obligation is complex. A cost-based input method of revenue recognition requires management to make estimates of total expected costs to complete the performance obligation. In making such estimates, significant judgment is required to evaluate key assumptions related to cost estimates, including internal personnel efforts and third-party contract costs. |
| How We Addressed the Matter in Our Audit | To test revenue recognized, we performed audit procedures that included, among others, gaining an understanding and testing the Company’s estimates of total expected costs including testing the completeness and accuracy of the underlying data, inspecting evidence of actual costs incurred and comparing with previous estimates. We evaluated any changes in the total expected costs, inspected communications between the Company and the collaborators regarding updates to estimated budgeted costs as well as any significant changes in its development plans and compared the estimates to actual incurred costs for the same or similar activities. We discussed the basis for key assumptions with the Company's research and development personnel to assess management's key assumptions used in the Company's estimates of total expected costs. |
/s/ Ernst && Young LLP
We have served as the Company’sCompany’s auditor since 2015.
Seattle, WAWashington
March 30, 2020
ALPINE IMMUNE SCIENCES, INC.
CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share amounts)
| | | | | | | | | | | | | | |
| | | December 31, |
| | | 2022 | | 2021 |
| Assets | | | | |
| Current assets: | | | | |
| Cash and cash equivalents | | $ | 13,376 | | | $ | 67,907 | |
| Short-term investments | | 224,265 | | | 94,396 | |
| Accounts receivable | | 392 | | | 25,000 | |
| | | | |
| Prepaid expenses and other current assets | | 2,960 | | | 4,710 | |
| Total current assets | | 240,993 | | | 192,013 | |
| Restricted cash, noncurrent | | 254 | | | 254 | |
| Property and equipment, net | | 1,584 | | | 1,716 | |
| Operating lease, right-of-use asset | | 8,219 | | | 8,837 | |
| Long-term investments | | 35,481 | | | 52,866 | |
| Deferred tax asset | | 155 | | | 214 | |
| Total assets | | $ | 286,686 | | | $ | 255,900 | |
| Liabilities and stockholders’ equity | | | | |
| Current liabilities: | | | | |
| Accounts payable | | $ | 4,286 | | | $ | 3,349 | |
| Accrued liabilities | | 14,003 | | | 9,417 | |
| Deferred revenue, current | | 35,571 | | | 51,773 | |
| Operating lease liability, current | | 756 | | | 617 | |
| Current portion of long-term debt | | 3,380 | | | 4,622 | |
| Total current liabilities | | 57,996 | | | 69,778 | |
| Deferred revenue, noncurrent | | 39,185 | | | 50,830 | |
| Operating lease liability, noncurrent | | 10,085 | | | 11,009 | |
| Long-term debt | | — | | | 3,380 | |
| Total liabilities | | 107,266 | | | 134,997 | |
| Commitments and contingencies | | | | |
| Stockholders’ equity: | | | | |
| Preferred stock, $0.001 par value per share; 10,000,000 shares authorized at December 31, 2022 and 2021; zero shares issued and outstanding at December 31, 2022 and 2021 | | — | | | — | |
| Common stock, $0.001 par value per share; 200,000,000 shares authorized at December 31, 2022 and 2021; 47,234,900 shares issued and 45,984,433 shares outstanding at December 31, 2022; 31,444,746 shares issued and 30,194,279 shares outstanding at December 31, 2021 | | 46 | | | 30 | |
| Treasury stock, at cost; 1,250,467 shares at December 31, 2022 and 2021 | | — | | | — | |
| Additional paid-in capital | | 404,456 | | | 287,345 | |
| Accumulated other comprehensive loss | | (1,121) | | | (273) | |
| Accumulated deficit | | (223,961) | | | (166,199) | |
| Total stockholders’ equity | | 179,420 | | | 120,903 | |
| Total liabilities and stockholders’ equity | | $ | 286,686 | | | $ | 255,900 | |
|
| | | | | | | | |
| | December 31, |
| | 2019 | | 2018 |
Assets | | | | |
Current assets: | | | | |
Cash and cash equivalents | | $ | 16,123 |
| | $ | 10,711 |
|
Short-term investments | | 24,397 |
| | 41,592 |
|
Restricted cash, current | | 132 |
| | — |
|
Prepaid expenses and other current assets | | 1,650 |
| | 1,242 |
|
Total current assets | | 42,302 |
| | 53,545 |
|
Restricted cash, noncurrent | | 254 |
| | 132 |
|
Property and equipment, net | | 1,552 |
| | 1,196 |
|
Operating lease, right-of-use asset | | 9,985 |
| | — |
|
Total assets | | $ | 54,093 |
| | $ | 54,873 |
|
Liabilities and stockholders’ equity | | | | |
Current liabilities: | | | | |
Accounts payable | | $ | 1,543 |
| | $ | 1,716 |
|
Accrued liabilities | | 5,285 |
| | 4,363 |
|
Deferred revenue | | 1,435 |
| | — |
|
Current portion of long-term debt | | 418 |
| | 2,048 |
|
Total current liabilities | | 8,681 |
| | 8,127 |
|
Operating lease liability, noncurrent | | 11,429 |
| | — |
|
Long-term debt | | 4,509 |
| | 2,155 |
|
Total liabilities | | 24,619 |
| | 10,282 |
|
Commitments and contingencies | |
|
| |
|
|
Convertible preferred stock, $0.001 par value per share; 10,000,000 shares authorized at December 31, 2019 and 2018; zero shares issued and outstanding at December 31, 2019 and 2018 | | — |
| | — |
|
Stockholders’ equity: | | | | |
Common stock, $0.001 par value per share; 200,000,000 shares authorized at December 31, 2019 and 2018; 18,638,359 shares issued and 18,587,892 shares outstanding at December 31, 2019; 13,904,672 shares issued and 13,854,205 shares outstanding at December 31, 2018 | | 19 |
| | 14 |
|
Treasury stock, at cost; 50,467 shares at December 31, 2019 and 2018 | | — |
| | — |
|
Additional paid-in capital | | 117,371 |
| | 90,664 |
|
Accumulated other comprehensive gain (loss) | | 10 |
| | (13 | ) |
Accumulated deficit | | (87,926 | ) | | (46,074 | ) |
Total stockholders’ equity | | 29,474 |
| | 44,591 |
|
Total liabilities and stockholders’ equity | | $ | 54,093 |
| | $ | 54,873 |
|
The accompanying notes are an integral part of these consolidated financial statements.
ALPINE IMMUNE SCIENCES, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(in thousands, except share and per share amounts)
| | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Collaboration revenue | $ | 30,064 | | | $ | 23,443 | | | $ | 9,335 | |
| Operating expenses: | | | | | |
| Research and development | 70,243 | | | 58,742 | | | 27,185 | |
| General and administrative | 17,968 | | | 14,560 | | | 10,899 | |
| | | | | |
| Total operating expenses | 88,211 | | | 73,302 | | | 38,084 | |
| Loss from operations | (58,147) | | | (49,859) | | | (28,749) | |
| Other income (expense): | | | | | |
| Interest income | 3,288 | | | 259 | | | 245 | |
| Interest expense | (476) | | | (816) | | | (775) | |
| Other, net | (97) | | | (4) | | | 1,333 | |
| Loss before taxes | (55,432) | | | (50,420) | | | (27,946) | |
| Income tax benefit (expense) | (2,330) | | | 87 | | | 6 | |
| Net loss | $ | (57,762) | | | $ | (50,333) | | | $ | (27,940) | |
| Comprehensive income (loss): | | | | | |
| Unrealized loss on investments | (901) | | | (238) | | | (15) | |
| Unrealized gain (loss) on foreign currency translation | 53 | | | (88) | | | 58 | |
| Comprehensive loss | $ | (58,610) | | | $ | (50,659) | | | $ | (27,897) | |
| Weighted-average shares used to compute basic and diluted net loss per share | 33,435,280 | | 25,476,889 | | 20,826,466 |
| Basic and diluted net loss per share | $ | (1.73) | | | $ | (1.98) | | | $ | (1.34) | |
|
| | | | | | | | | | | |
| Years Ended December 31, |
| 2019 | | 2018 | | 2017 |
Collaboration revenue | $ | 1,740 |
| | $ | 705 |
| | $ | 1,731 |
|
Operating expenses: | | | | | |
Research and development | 35,847 |
| | 28,970 |
| | 10,626 |
|
General and administrative | 9,467 |
| | 8,362 |
| | 6,079 |
|
Loss on sale of intangible asset | — |
| | 1,203 |
| | — |
|
Total operating expenses | 45,314 |
| | 38,535 |
| | 16,705 |
|
Loss from operations | (43,574 | ) | | (37,830 | ) | | (14,974 | ) |
Other income (expense): | | | | | |
Bargain purchase gain | — |
| | — |
| | 6,601 |
|
Interest and other expense | (338 | ) | | (319 | ) | | (152 | ) |
Interest income | 1,248 |
| | 1,296 |
| | 542 |
|
Other income | 812 |
| | — |
| | — |
|
Loss before taxes | (41,852 | ) | | (36,853 | ) | | (7,983 | ) |
Income tax benefit | — |
| | 366 |
| | 200 |
|
Net loss | $ | (41,852 | ) | | $ | (36,487 | ) | | $ | (7,783 | ) |
Comprehensive income (loss): | | | | | |
Unrealized gain (loss) on investments | 29 |
| | 46 |
| | (59 | ) |
Unrealized loss on foreign currency translation | (6 | ) | | — |
| | — |
|
Comprehensive loss | $ | (41,829 | ) | | $ | (36,441 | ) | | $ | (7,842 | ) |
Weighted-average shares used to compute basic and diluted net loss per share | 18,358,864 |
| | 13,849,470 |
| | 6,481,665 |
|
Basic and diluted net loss per share | $ | (2.28 | ) | | $ | (2.63 | ) | | $ | (1.20 | ) |
The accompanying notes are an integral part of these consolidated financial statements.
ALPINE IMMUNE SCIENCES, INC.
CONSOLIDATED STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’STOCKHOLDERS’ EQUITY
(in thousands, except share and per share amounts)shares)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Shares | | Common Stock, Par Value | | | | Additional
Paid-in Capital | | Accumulated
Other Comprehensive (Loss) Gain | | Accumulated Deficit | | Total
Stockholders’ Equity |
| | | | | | Common | | Treasury | | | | | | |
| Balance, December 31, 2019 | | | | | 18,587,892 | | | 50,467 | | | $ | 19 | | | | | $ | 117,371 | | | $ | 10 | | | $ | (87,926) | | | $ | 29,474 | |
| Issuance of Units in private offering, net of offering costs | | | | | 5,139,610 | | | — | | | 5 | | | | | 56,253 | | | — | | | — | | | 56,258 | |
| Issuance of warrants | | | | | — | | | — | | | — | | | | | 60 | | | — | | | — | | | 60 | |
| Issuance of common stock under equity incentive plans | | | | | 75,681 | | | — | | | — | | | | | 123 | | | — | | | — | | | 123 | |
| Stock-based compensation | | | | | — | | | — | | | — | | | | | 4,140 | | | — | | | — | | | 4,140 | |
| Unrealized loss on investments | | | | | — | | | — | | | — | | | | | — | | | (15) | | | — | | | (15) | |
| Unrealized gain on foreign currency translation | | | | | — | | | — | | | — | | | | | — | | | 58 | | | — | | | 58 | |
| Net loss | | | | | — | | | — | | | — | | | | | — | | | — | | | (27,940) | | | (27,940) | |
| Balance, December 31, 2020 | | | | | 23,803,183 | | | 50,467 | | | 24 | | | | | 177,947 | | | 53 | | | $ | (115,866) | | | 62,158 | |
| Issuance of common stock in Private Placement, net of offering costs | | | | | 6,489,357 | | | — | | | 6 | | | | | 90,727 | | | — | | | — | | | 90,733 | |
| Issuance of common stock to Horizon | | | | | 951,980 | | | — | | | 1 | | | | | 11,928 | | | — | | | — | | | 11,929 | |
| Exchange of common stock for prefunded warrants | | | | | (1,200,000) | | | 1,200,000 | | | (1) | | | | | 1 | | | — | | | — | | | — | |
| Stock-based compensation | | | | | — | | | — | | | — | | | | | 6,240 | | | — | | | — | | | 6,240 | |
| Issuance of common stock under equity incentive plans | | | | | 149,759 | | | — | | | — | | | | | 502 | | | — | | | — | | | 502 | |
| Unrealized loss on investments | | | | | — | | | — | | | — | | | | | — | | | (238) | | | — | | | (238) | |
| Unrealized loss on foreign currency translation | | | | | — | | | — | | | — | | | | | — | | | (88) | | | — | | | (88) | |
| Net loss | | | | | — | | | — | | | — | | | | | — | | | — | | | (50,333) | | | (50,333) | |
| Balance, December 31, 2021 | | | | | 30,194,279 | | | 1,250,467 | | | 30 | | | | | 287,345 | | | (273) | | | (166,199) | | | 120,903 | |
| Issuance of common stock in public offering, net of financing costs | | | | | 15,509,282 | | | — | | | 16 | | | | | 106,690 | | | — | | | — | | | 106,706 | |
| Stock-based compensation | | | | | — | | | — | | | — | | | | | 9,648 | | | — | | | — | | | 9,648 | |
| Issuance of common stock under equity incentive plans | | | | | 280,872 | | | — | | | — | | | | | 773 | | | — | | | — | | | 773 | |
| Unrealized loss on investments | | | | | — | | | — | | | — | | | | | — | | | (901) | | | — | | | (901) | |
| Unrealized gain on foreign currency translation | | | | | — | | | — | | | — | | | | | — | | | 53 | | | — | | | 53 | |
| Net loss | | | | | — | | | — | | | — | | | | | — | | | — | | | (57,762) | | | (57,762) | |
| Balance, December 31, 2022 | | | | | 45,984,433 | | | 1,250,467 | | | $ | 46 | | | | | $ | 404,456 | | | $ | (1,121) | | | $ | (223,961) | | | $ | 179,420 | |
|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Convertible Preferred Stock | | | Common Stock | | Treasury | | Additional Paid-in Capital | | Accumulated Other Comprehensive Loss | | Accumulated Deficit | | Total Stockholders’ Equity |
| Shares | | Amount | | | Shares | | Amount | | Shares | | Amount | | | | |
Balance, December 31, 2016 | 4,311,770 |
| | $ | 11,535 |
| | | 608,701 |
| | $ | — |
| | — |
| | $ | — |
| | $ | 144 |
| | $ | — |
| | $ | (1,601 | ) | | $ | (1,457 | ) |
Issuance of Series A-1 convertible preferred stock | 4,989,663 |
| | 37,666 |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | — |
|
Conversion of convertible preferred stock to common stock | (9,301,433 | ) | | (49,201 | ) | | | 9,301,433 |
| | 1 |
| | — |
| | — |
| | 49,200 |
| | — |
| | — |
| | 49,201 |
|
Common stock acquired in business combination | — |
| | — |
| | | 3,914,058 |
| | — |
| | — |
| | — |
| | 38,103 |
| | — |
| | — |
| | 38,103 |
|
Adjustment of par value from $0.0001 per share to $0.001 per share | — |
| | — |
| | | — |
| | 13 |
| | — |
| | — |
| | (13 | ) | | — |
| | — |
| | — |
|
Conversion of warrant liability to equity | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | 52 |
| | — |
| | — |
| | 52 |
|
Exercise of stock options and common stock warrants | — |
| | — |
| | | 57,453 |
| | — |
| | — |
| | — |
| | 22 |
| | — |
| | — |
| | 22 |
|
Repurchase of common stock | — |
| | — |
| | | (50,467 | ) | | — |
| | 50,467 |
| | — |
| | — |
| | — |
| | — |
| | — |
|
Stock-based compensation | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | 838 |
| | — |
| | — |
| | 838 |
|
Unrealized loss on investments | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | (59 | ) | | — |
| | (59 | ) |
Net loss | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (7,783 | ) | | (7,783 | ) |
Balance, December 31, 2017 | — |
| | — |
| | | 13,831,178 |
| | 14 |
| | 50,467 |
| | — |
| | 88,346 |
| | (59 | ) | | (9,384 | ) | | 78,917 |
|
Cumulative effect of changes related to adoption of new revenue standard | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (203 | ) | | (203 | ) |
Exercise of stock options | — |
| | — |
| | | 23,027 |
| | — |
| | — |
| | — |
| | 9 |
| | — |
| | — |
| | 9 |
|
Stock-based compensation | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | 2,309 |
| | — |
| | — |
| | 2,309 |
|
Unrealized gain on investments | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | 46 |
| | — |
| | 46 |
|
Net loss | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (36,487 | ) | | (36,487 | ) |
Balance, December 31, 2018 | — |
| | — |
| | | 13,854,205 |
| | 14 |
| | 50,467 |
| | — |
| | 90,664 |
| | (13 | ) | | (46,074 | ) | | 44,591 |
|
Issuance of Units in Private Placement, net of offering costs | — |
| | — |
| | | 4,706,700 |
| | 5 |
| | — |
| | — |
| | 23,593 |
| | — |
| | — |
| | 23,598 |
|
Exercise of stock options | — |
| | — |
| | | 26,987 |
| | — |
| | — |
| | — |
| | 13 |
| | — |
| | — |
| | 13 |
|
Stock-based compensation | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | 3,041 |
| | — |
| | — |
| | 3,041 |
|
Issuance of warrants | | | | | | | | | | | | | | 60 |
| | | | | | 60 |
|
Unrealized gain on investments | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | 29 |
| | — |
| | 29 |
|
Unrealized loss on foreign currency translation | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | (6 | ) | | — |
| | (6 | ) |
Net loss | — |
| | — |
| | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (41,852 | ) | | (41,852 | ) |
Balance, December 31, 2019 | — |
|
| $ | — |
|
|
| 18,587,892 |
|
| $ | 19 |
|
| 50,467 |
|
| $ | — |
|
| $ | 117,371 |
|
| $ | 10 |
|
| $ | (87,926 | ) |
| $ | 29,474 |
|
The accompanying notes are an integral part of these consolidated financial statements
ALPINE IMMUNE SCIENCES, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands) |
| | | | | | | | | | | |
| Years Ended December 31, |
| 2019 | | 2018 | | 2017 |
Operating activities | | | | | |
Net loss | $ | (41,852 | ) | | $ | (36,487 | ) | | $ | (7,783 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | |
Loss on sale of property and equipment | 16 |
| | — |
| | — |
|
Loss on sale of intangible asset | — |
| | 1,203 |
| | — |
|
Bargain purchase gain | — |
| | — |
| | (6,601 | ) |
Depreciation expense | 468 |
| | 388 |
| | 241 |
|
Amortization of premium/discount on investments | (360 | ) | | (669 | ) | | — |
|
Non-cash interest expense | 140 |
| | 169 |
| | 87 |
|
Deferred income tax | — |
| | (305 | ) | | (204 | ) |
Stock-based compensation expense | 3,041 |
| | 2,309 |
| | 838 |
|
Changes in operating assets and liabilities: | | | | | |
Prepaid expenses and other current assets | (408 | ) | | 66 |
| | (1,193 | ) |
Right-of-use asset | 1,553 |
| | — |
| | — |
|
Accounts payable and accrued liabilities | 816 |
| | 5,390 |
| | (226 | ) |
Deferred revenue | 1,435 |
| | (480 | ) | | (1,731 | ) |
Lease liabilities | (195 | ) | | — |
| | — |
|
Net cash used in operating activities | (35,346 | ) | | (28,416 | ) | | (16,572 | ) |
Investing activities | | | | | |
Purchases of property and equipment | (821 | ) | | (495 | ) | | (586 | ) |
Proceeds from sale of intangible asset | — |
| | 250 |
| | — |
|
Purchase of short-term investments | (59,382 | ) | | (72,863 | ) | | (88,307 | ) |
Maturities of short-term investments | 75,575 |
| | 105,226 |
| | — |
|
Proceeds from the sale of short-term investments | 1,391 |
| | — |
| | 27,960 |
|
Cash and cash equivalents acquired in connection with merger | — |
| | — |
| | 31,130 |
|
Net cash provided by (used in) investing activities | 16,763 |
| | 32,118 |
| | (29,803 | ) |
Financing activities | | | | | |
Proceeds from sale of preferred stock | — |
| | — |
| | 37,666 |
|
Proceeds from sale of common stock, net of offering costs | 23,598 |
| | — |
| | — |
|
Proceeds from borrowings, net of issuance costs | 1,977 |
| | — |
| | 5,000 |
|
Repayment of debt | (1,333 | ) | | (1,000 | ) | | — |
|
Proceeds from exercise of stock options | 13 |
| | 9 |
| | 22 |
|
Net cash provided by (used in) financing activities | 24,255 |
| | (991 | ) | | 42,688 |
|
Effect of exchange rate on cash, cash equivalents and restricted cash | (6 | ) | | — |
| | — |
|
Net increase (decrease) in cash and cash equivalents and restricted cash | 5,666 |
| | 2,711 |
| | (3,687 | ) |
Cash and cash equivalents and restricted cash, beginning of period | 10,843 |
| | 8,132 |
| | 11,819 |
|
Cash and cash equivalents and restricted cash, end of period | $ | 16,509 |
| | $ | 10,843 |
| | $ | 8,132 |
|
Supplemental Information | | | | | |
Recognition of right-of-use asset | $ | 11,173 |
| | $ | — |
| | $ | — |
|
Cash paid for interest | $ | 170 |
| | $ | 149 |
| | $ | 53 |
|
Convertible preferred stock exchanged for common stock | $ | — |
| | $ | — |
| | $ | 49,201 |
|
Discount in connection with issuance of debt | $ | — |
| | $ | — |
| | $ | 428 |
|
Cash paid for income taxes | $ | — |
| | $ | — |
| | $ | 76 |
|
The accompanying notes are an integral part of these consolidated financial statements.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Operating activities | | | | | |
| Net loss | $ | (57,762) | | | $ | (50,333) | | | $ | (27,940) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | | | |
| Loss on sale of property and equipment | 93 | | | 4 | | | 5 | |
| Depreciation expense | 597 | | | 620 | | | 578 | |
| Amortization of premium/discount on investments | (700) | | | 906 | | | 103 | |
| Non-cash interest expense | 178 | | | 274 | | | 261 | |
| Deferred income tax | 46 | | | (214) | | | — | |
| Stock-based compensation expense | 9,648 | | | 6,240 | | | 4,140 | |
| Changes in operating assets and liabilities: | | | | | |
| Accounts receivable | 24,602 | | | — | | | — | |
| Prepaid expenses and other current assets | 2,001 | | | (3,063) | | | 255 | |
| Operating lease, right-of-use asset | 618 | | | 564 | | | 592 | |
| Accounts payable and accrued liabilities | 5,557 | | | 5,970 | | | (491) | |
| Deferred revenue | (27,847) | | | 24,628 | | | 51,540 | |
| Operating lease liability | (785) | | | (844) | | | 1,041 | |
| Net cash (used in) provided by operating activities | (43,754) | | | (15,248) | | | 30,084 | |
| Investing activities | | | | | |
| Purchases of property and equipment | (412) | | | (118) | | | (802) | |
| | | | | |
| Purchase of investments | (266,984) | | | (133,518) | | | (101,328) | |
| Maturities of investments | 154,048 | | | 81,156 | | | 29,311 | |
| | | | | |
| Net cash used in investing activities | (113,348) | | | (52,480) | | | (72,819) | |
| Financing activities | | | | | |
| Proceeds from sale of common stock and warrants, net of offering costs | 106,716 | | | 90,733 | | | 56,258 | |
| Proceeds from issuance of common stock to Horizon | — | | | 11,929 | | | — | |
| Proceeds from borrowings, net of issuance costs | — | | | — | | | 5,000 | |
| Repayment of debt | (4,800) | | | (2,400) | | | — | |
| Proceeds from exercise of stock options | 773 | | | 502 | | | 123 | |
| Net cash provided by financing activities | 102,689 | | | 100,764 | | | 61,381 | |
| Effect of exchange rate on cash and cash equivalents | (118) | | | (88) | | | 58 | |
| Net (decrease) increase in cash and cash equivalents and restricted cash | (54,531) | | | 32,948 | | | 18,704 | |
| Cash and cash equivalents and restricted cash, beginning of period | 68,161 | | | 35,213 | | | 16,509 | |
| Cash and cash equivalents and restricted cash, end of period | $ | 13,630 | | | $ | 68,161 | | | $ | 35,213 | |
| Supplemental Information | | | | | |
| | | | | |
| Cash paid for interest | $ | 322 | | | $ | 554 | | | $ | 490 | |
| Discount in connection with issuance of debt | $ | — | | | $ | — | | | $ | 334 | |
| Cash paid for income taxes | $ | 543 | | | $ | — | | | $ | — | |
The accompanying notes are an integral part of these consolidated financial statements.
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Description of Business
Alpine Immune Sciences, Inc. (the “Company”“Company”, “Alpine”“Alpine”, “we”“we”, “us”“us”, or “our”“our”), together with its consolidated subsidiaries, is a clinical-stage immunotherapybiopharmaceutical company committeddedicated to leading a new wave of immune therapeutics, creating potentially powerful multifunctionaldiscovering and developing innovative, protein-based immunotherapies to improve patients’ lives via unique protein engineering technologies. Alpine has two lead programs, ALPN-101 for autoimmune/treat autoimmune and inflammatory diseases, and ALPN-202 for the treatment of cancer.diseases. Our approach includes a proprietary scientific platform uses a process known as directed evolution to convertthat converts native immune system proteins from the Immunoglobulin Super Family,into differentiated, multi-targeted therapeutics. We are seeking to create first- or IgSF, into multi-targeted therapeutics potentially capable of modulating the human immune system.best-in-class multifunctional immunotherapies via our unique protein engineering technologies to improve outcomes in patients with serious diseases. We were incorporated under the laws of the State of Delaware and are headquartered in Seattle, Washington.
Liquidity
In accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Update (“ASU”) 2014-15, Presentation of Financial Statements - Going Concern (“Subtopic 205-40”), our management evaluates whether there are conditions or events, considered in aggregate, that raise substantial doubt about our ability to continue as a going concern within one year after the date that the financial statements are issued. We considered such conditions or events and we expect our existing cash and cash equivalents will be sufficient to fund our operations until at least March 30, 2021.
In performing the assessment under Accounting Standards Codification (“ASC”) Topic 205-40, we considered that our long-term operations anticipate continuing net losses and the need for potential equity or debt financing. We also considered that new collaborations or selectively partnering our technology or programs may provide other sources of capital. However, there can be no assurances that additional funding or other sources of capital will be available on terms acceptable to us, or at all. Therefore, we developed a plan to implement cost cutting measures to reduce our working capital requirements assuming no additional planned financing. The plan includes a delay in hiring and additional reductions in personnel-related costs and other discretionary expenditures that are within our control and do not effect the anticipated timing of our Phase 1 clinical trials. Based off of our operating plan, it is probable that the cost cutting measures described above can be effectively implemented to allow us to meet our obligations as they become due within one year after the date that the financial statements are issued.
Reverse Merger and Subscription Agreement
On April 18, 2017, we entered into a merger agreement with Nivalis Therapeutics, Inc. (“Nivalis”), a public biotechnology company, and one of its wholly-owned subsidiaries pursuant to which, the subsidiary merged with and into Alpine, with Alpine continuing as a wholly owned subsidiary of Nivalis and the surviving corporation of the merger (the “Merger Agreement”). Nivalis Therapeutics, Inc. was incorporated in Delaware in March 2007. Alpine Immune Sciences, Inc. (prior to its business combination with Nivalis Therapeutics, Inc.) was incorporated in Delaware on December 30, 2014.
The merger is intended to qualify for federal income tax purposes as a tax-free reorganization under the provisions of Section 368(a) of the Internal Revenue Code of 1986, as amended. At the closing of the merger, each outstanding share of our capital stock (common stock and preferred stock) was converted into the right to receive shares of Nivalis common stock (subject to the payment of cash in lieu of fractional shares and after giving effect to a 1:4 reverse stock split of Nivalis common stock) such that, immediately following the effective time of the merger, preexisting Nivalis stockholders, optionholders, and warrantholders owned, or held rights to acquire, approximately 26% of the fully-diluted common stock of Nivalis, which changed its name to “Alpine Immune Sciences, Inc.” following the completion of the merger and Alpine’s preexisting stockholders, optionholders, and warrantholders owned, or held rights to acquire approximately 74% of the fully-diluted common stock of Nivalis. The issuance of the shares to our pre-existing stockholders was registered under the Securities Act of 1933, as amended, pursuant to a registration statement on Form S-4 (No. 333-218134) (the “Registration Statement”) declared effective by the Securities and Exchange Commission (the “SEC”) on June 6, 2017.
Contemporaneously with the execution and delivery of the Merger Agreement, certain of our pre-existing stockholders entered into a subscription agreement with us pursuant to which such stockholders purchased, immediately prior to the closing of the merger, 1,335,118 shares of our capital stock at a purchase price of $12.74 per share for an aggregate purchase price of approximately $17.0 million.
The merger and the subscription described above were consummated on July 24, 2017.
ALPINE IMMUNE SCINECES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
2. Summary of Significant Accounting Policies
Basis of Presentation and Use of Estimates
The accompanying consolidated financial statements have been prepared in accordance with the rules and regulations of the SECSecurities and Exchange Commission (“SEC”) and generally accepted accounting principles in the United States of America (“GAAP”(“GAAP”). The preparation of financial statements in conformity with GAAP requires management to make judgments, estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. Significant estimates inherent in the preparation of the accompanying consolidated financial statements include those used for revenue recognition, accruals for clinical trial activities and other accruals, the potential outcome of uncertain tax positions that have been recognized in our consolidated financial statements or tax returns, and the estimated fair value of equity-based awards. We base our estimates and assumptions on historical experience when available and on various factors we believe to be reasonable under the circumstances. Actual results could differ materially from those estimates.
Principles of Consolidation
Our consolidated financial statements include the financial position and results of operations of Alpine Immune Sciences, Inc. and our wholly owned operating company and subsidiary, AIS Operating Co., Inc., and our wholly-ownedwholly owned subsidiary, Alpine Immune Sciences Australia PTY LTD. All inter-company balances and transactions have been eliminated in consolidation.
Segments
We operate as 1one operating segment and use cash flow as the primary financial measure to manage our business.
Cash and Cash Equivalents and Restricted cashCash
We consider all highly liquid investments with an original maturity of 90 days or less at the time of purchase to be cash equivalents. Cash and cash equivalents consist of deposits with commercial banks in checking and interest-bearing accounts, and highly liquid money market funds.
Restricted cash represents cash drawn on linesour line of credit used to establish collateral to support the security deposit on our operating leaseslease to rent office and laboratory space in Seattle, Washington.
Periodically, we maintain deposits in financial institutions in excess of government insured limits. We believe we are not exposed to significant credit risk as our deposits, which are held at financial institutions, are high credit quality securities such as money market funds, U.S. Treasury securities, and commercial paper. To date, we have not realized any losses on these deposits.
Short-Term Investments
Our short-term investments include funds invested in highly liquid money market funds, U.S. Treasury securities, commercial paper,U.S. agency securities, non-U.S. government securities, and corporate debt securities and commercial paper with a finalcontractual maturity of each security of less than one year. Alltwo years. These investments are classified as available-for-sale debt securities, andwhich are recorded at fair value based on quoted prices in active markets,markets. We classify our investments maturing within one year of the reporting date as short-term investments.
If the estimated fair value of a debt security is below its amortized cost basis, we evaluate whether it is more likely than not that we will sell the security before its anticipated recovery in market value and whether credit losses exist for the related securities. A credit loss exists if the present value of expected cash flows is less than the amortized cost basis of the security. Credit-related losses are recognized as an allowance for credit losses on the balance sheet with unrealizeda corresponding
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
adjustment to earnings. Unrealized gains and losses excluded from earnings andthat are unrelated to credit deterioration are reported in other comprehensive income (loss). Purchase premiums and discounts are recognized as interest income using the interest method over the terms of the securities. Realized gains and losses and declines in fair value deemed to be other than temporary are reflected in theour Consolidated Statements of Operations and Comprehensive Income (Loss) using the specific-identification method. Property and Equipment
Property and equipment are stated at cost, net of accumulated depreciation. Depreciation is recorded using the straight-line method over the estimated useful lives of the assets, generally three to five years, while leasehold improvements are amortized over the shorter of their estimated useful lives or the related lease term. Upon retirement or sale, the cost of assets disposed of and the related accumulated depreciation are removed from the accounts and any resulting gain or loss is credited or charged to operations. Maintenance and repairs are expensed as incurred. Major improvements are capitalized as additions to property and equipment.
Impairment of Long-lived Assets
We evaluate our long-lived tangible assets for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable.fully recoverable.When such events occur, we assess recoverability by comparing the asset's carrying value to the total undiscounted future cash flows the asset is expected to generate. If the carrying value exceeds the undiscounted future cash flows estimated to result from the use and eventual dispositionamount of the asset, we write down the asset to its estimated fair value. Impairment is assessed by comparing the undiscounted cash flows, expectedwe then measure impairment by reducing the carrying value to be generated by the asset to its carryingassets estimated fair value. We did not record any impairments in the years ended December 31, 2019, 2018 and 2017.2022, 2021 or 2020.
ALPINE IMMUNE SCINECES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
Accrued Liabilities
As part of the process of preparing our consolidated financial statements, we are required to estimate accruals for professional services and research and development expenses. This process involves reviewing contracts and vendor agreements and communicating with applicable personnel to identify services that have been performed on our behalf. We estimate the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of the actual cost. We estimate accrued liabilities as of each balance sheet date based on known facts and circumstances.
Although we do not expect our estimates to be materially different from amounts actually incurred, if our estimates of the status and timing of services performed differ from the actual status and timing of services performed, we may report amounts that are too high or too low in any particular period. To date, we have not experienced any significant adjustments to our estimates.
Leases (effective January 1, 2019)
We account for our leases under Accounting Standards Codification (“ASC”(“ASC”) 842, Leases. Under this guidance, arrangementswe applied the practical expedients regarding the identification of leases, lease classification, indirect costs, and the combination of lease and non-lease components. Arrangements meeting the definition of a lease are classified as operating or financing leases, and are recorded on the consolidated balance sheet as both a right-of-use asset and lease liability, calculated by discounting fixed lease payments over the lease term at the rate implicit in the lease or our incremental borrowing rate. As we do not know the lessor’slessor’s implicit rate, we use our incremental borrowing rate at the commencement date of the lease in determining the present value of lease payments. Lease liabilities are increased by interest and reduced by payments each period, and the right-of-use asset is amortized over the lease term. For operating leases, interest on the lease liability and the amortization of the right-of-use asset result in straight-line rent expense over the lease term. For finance leases, interest on the lease liability and the amortization of the right-of-use asset results is front-loaded expense over the lease term. Variable lease expenses are recorded when incurred.
In calculating the right-of-use asset and lease liability, we elected to combine lease and non-lease components. We exclude short-term leases having initial terms of 12 months or less from the new guidance as an accounting policy election, and recognize rent expense on a straight-line basis over the lease term. We continue to account for leases in the prior period financial statements under ASC Topic 840.
Common Stock Warrants
We grant common stock warrants to certain non-employee professional advisers from time to time. We account for our warrants at fair value, with changes in fair value recognized in operating expenses. Common stock warrants are initially recorded at their issuance date fair value and unvested warrants are subsequently remeasured at each balance sheet date. These warrants are valued using the Black-Scholes option pricing model based on the estimated market value of the underlying common stock at the valuation measurement dates, the remaining contractual term of the warrant, risk-free interest rates, expected dividends, and expected volatility of the price of the underlying common stock.
Derivative Financial Instruments
We evaluate all of our financial instruments, including issuedprefunded warrants and warrants to purchase common stock, purchase warrants, to determine if such instruments are derivatives or contain features qualifying as embedded derivatives. For derivative financial instruments accounted for as liabilities, the derivative instrument is initially recorded at its fair value and is then re-valued at each reporting date, with changes in the fair value reported in the Consolidated Statements of Operations and Comprehensive Income (Loss). We use the Black-Scholes option-pricing model to value the derivative instruments at inception and subsequent valuation dates. The classification of derivative instruments, including whether such instruments should be recorded as
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
liabilities or as equity, is re-assessed at the end of each reporting period. We do not use derivative instruments to hedge exposures to cash flow, market or foreign currency risks.
Revenue Recognition (effective January 1, 2018)
We adopted ASC Topic 606, Revenue from Contracts with Customers , as amended (“new revenue standard” or “ASC 606”) on January 1, 2018, resulting in a change to our accounting policy for revenue recognition. We used the modified retrospective method transition option and recognized the cumulative effect of adopting the new revenue standard as an adjustment to increase the opening accumulated deficit at January 1, 2018. Accordingly, 2017 comparative information has not been adjusted and continues to be reported under previous accounting standards.
Revenue is recognized when control of the promised goods or services is transferred to our customers, in an amount that reflects the consideration we expect to be entitled to in exchange for those goods or services. Our steps for recognizing revenue consist of;of (1) identifying the contract,contract; (2) identifying the performance obligations as either distinct or bundled goods and
ALPINE IMMUNE SCINECES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
services,services; (3) determining the transaction price associated with each performance obligation for which we expect to be entitled in exchange for transferring such goods and services,services; (4) allocating the transaction price to the performance obligations in the contractcontract; and (5) recognizing revenue upon satisfaction of performance obligations.
Our collaboration agreements principally contain multiple performance obligations, which may include (1) grants of, or options to obtain, intellectual property licenses; (2) research and development services; and/or (3) manufacturing or supply services. Payments typically received under these arrangements typically include one or more of the following: non-refundable upfront license fees, option exercise fees, payment for research and/or development efforts, amounts due upon the achievement of specified objectives, and/or royalties on future product sales. Our revenue is primarily derived from our collaboration agreements with Adaptimmune Therapeutics plc (“Adaptimmune)(“Adaptimmune”), AbbVie Ireland Unlimited Company (“AbbVie”), and Kite Pharma, a Gilead company (“Kite”Horizon Therapeutics Ireland DAC (“Horizon”). See further discussion of our collaboration agreements in Note 1210. We allocate revenue to each performance obligation based on its relative stand-alone selling price. We generally determine stand-alone selling prices at the inception of the contract based on our best estimate of what the selling price would be if the deliverable was regularly sold by us on a stand-alone basis. Payments received prior to satisfying the relevant revenue recognition criteria are recorded as deferred revenue in the accompanying Consolidated Balance Sheets and recognized as revenue when the related revenue recognition criteria are met. We recognize revenue under our collaboration agreements based onby using a cost-based input method to measure progress toward completion of the performance obligation, including employee hours contributed to each performance obligation.obligation, and to calculate the corresponding revenue to recognize each period. Our collaboration agreements provide for non-refundable milestone payments. We recognize revenue that is contingent upon the achievement of a substantive milestone in its entirety in the period in which the milestone is achieved. A milestone is considered substantive when the consideration payable to us for such milestone (1) is consistent with our performance necessary to achieve the milestone or the increase in value to the collaboration resulting from our performance; (2) relates solely to our past performance; and (3) is reasonable relative to all of the other deliverables and payments within the arrangement. In making this assessment, we consider all facts and circumstances relevant to the arrangement, including factors such as the scientific, regulatory, commercial, and other risks that must be overcome to achieve the milestone, the level of effort and investment required to achieve the milestone and whether any portion of the milestone consideration is related to future performance or deliverables.
We review the contributed employee hours and progress towards completion for each performance obligation under our collaboration agreements, and adjust the revenue recognized to reflect changes in assumptions relating to the estimated satisfaction of the performance obligation. We could accelerate revenueRevenue recognition may be accelerated in the event of early termination of programs or if our expectations change. Alternatively, we could decelerate revenue recognition may be decelerated if programs are extended or delayed. While such changes to our estimates have no impact on our reported cash flows, the timing of revenue recorded in future periods could be materially impacted.
Research and Development
Research and development costs are expensed as incurred. Research and development costs include personnel costs, clinical trials, external contract research and development expenses, raw materials, drug product manufacturing costs and allocated overhead –– including depreciation, rent and utilities. Research and development costs that are paid in advance of performance are capitalized as a prepaid expense and amortized over the service period as the services are provided.
Stock-based Compensation
Stock-based compensation is recognized for all share-based payments based on the estimated fair value as of the date of grant. The fair value of our stock options is calculated using the Black-Scholes option pricing model, which requires judgmental assumptions including volatility, risk-free interest rate, expected term and expected term.dividend yield. The fair value of restricted stock units (“RSUs”) is based on the closing price of our common stock on the award date. Stock-based compensation expense is recognized over the requisite service period of the awards, usually the vesting period, on a straight-line basis. For performance-based awards where the vesting of the options may be accelerated upon the achievement of certain milestones, vesting and the
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
related stock-based compensation is recognized as an expense when it is probable the milestone will be met. We recognize forfeiture of awards as they occur rather than estimating the expected forfeiture rate.
Income Taxes
Income taxes are accounted for using an asset and liability approach that requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the consolidated financial statement and tax bases of assets and liabilities at the applicable enacted tax rates. We will establish a valuation allowance for deferred tax assets if it is more likely than not that these items will expire before we are able to realize their benefits or that future deductibility is uncertain.
ALPINE IMMUNE SCINECES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
We recognize the tax benefit from uncertain tax positions only if it is more likely than not that the tax position will be sustained on examination by the tax authorities, based on the technical merits of the position. The tax position is measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement. We recognize interest and penalties related to income tax matters in income tax expense if incurred.
Comprehensive Income (Loss)
Comprehensive income (loss) is comprised of net loss and certain changes in equity excluded from net loss. For the years ended December 31, 2019, 2018,2022, 2021 and 2017,2020, other comprehensive loss consisted of unrealized gains and losses on our short-term investments and unrealized gains and losses on foreign currency translation.
Foreign Currency Translation
Our wholly-owned Australian subsidiary uses the Australian dollar as its functional currency. All assets and liabilities related to this subsidiary are translated using period-end exchange rates and revenues and expenses are translated at average exchange rates for the year. Translation adjustments are included as components of comprehensive gainincome (loss) in the Consolidated Statements of Operations and Comprehensive Income (Loss). Recently Issued Accounting Pronouncements
In December 2019, the FASB issued ASU No. 2019-12, Simplifying the Accounting for Income Taxes. The objective of the standard is to improve areas of GAAP by removing certain exceptions permitted by ASC Topic 740, Income Taxes, and clarifying existing guidance to facilitate consistent application. The standard will become effective for us beginning on January 1, 2021. We are currently evaluating the new standard to determine the potential impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
In November 2018, the FASB issued ASU No. 2018-18, Collaborative Arrangements: Clarifying the Interaction between Topic 808 and Topic 606. This ASU clarifies that certain transactions between collaborative arrangement participants should be accounted for as revenue when the collaborative arrangement participant is a customer in the context of a unit of account and precludes recognizing as revenue consideration received from a collaborative arrangement participant if the participant is not a customer. This ASU is effective for public companies for annual reporting periods and interim periods within those annual periods beginning after December 15, 2019. We do not anticipate the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
In August 2018, the FASB issued ASU No. 2018-13, Fair Value Measurement. ASU 2018-13 modifies the disclosure requirements for fair value measurements by removing, modifying, or adding certain disclosures. This ASU is effective for fiscal years beginning after December 15, 2019, and interim periods within those fiscal years, with early adoption permitted for any eliminated or modified disclosures. We do not expect the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments: Credit Losses, as clarified in ASU 2019-04 and ASU 2019-05. The objective of the standard is to provide information about expected credit losses on financial instruments at each reporting date and to change how other-than-temporary impairments on investment securities are recorded. The standard will become effective for us beginning on January 1, 2020. We do not anticipate the adoption of this ASU to have a material impact on our financial condition, results of operations, cash flows, and financial statement disclosures.
Recently Adopted Accounting Pronouncements
In June 2018, the FASB issued ASU No. 2018-07, which aligns the measurement and classification guidance for share-based payments to nonemployees with the guidance for share-based payments to employees, with certain exceptions. Under the guidance, the measurement of equity-classified nonemployee awards will be fixed at the grant date. Upon transition, nonemployee awards will be required to be measured at fair value as of the adoption date with a cumulative-effect adjustment recognized in retained earnings as of the beginning of the annual period of adoption. ASU 2018-07 is effective for fiscal years beginning after December 15, 2018, including interim periods within that fiscal year. We adopted this standard on January 1, 2019 and it did not have a material impact on our financial statements and related disclosures.
In February 2016, the FASB issued ASU No. 2016-02, Leases. ASU 2016-02 requires a lessee to separate the lease components from the non-lease components in a contract and recognize in the statement of financial position a liability to make lease payments (the lease liability) and a right-of-use asset representing its right to use the underlying asset for the lease term. It also aligns lease accounting for lessors with the revenue recognition guidance in ASU 2014-09. We adopted this ASU effective January 1, 2019 and elected the modified retrospective method transition option, which permitted us not to restate the comparative period presented. Upon adoption, we recorded an operating lease right-of-use asset of $797,000, a corresponding
ALPINE IMMUNE SCINECES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
operating lease liability of $883,000, and reduced our deferred rent balance by $86,000 to $0 on our accompanying Consolidated Balance Sheets; there was no effect on opening retained earnings, and we continue to account for leases in the prior period financial statements under ASC Topic 840. In adopting the new standard, we elected to apply the practical expedients regarding the identification of leases, lease classification, indirect costs, and the combination of lease and non-lease components.
3. Business Combination
On July 24, 2017, we closed the merger on the terms described in more detail in Note 1. In connection with the merger, Nivalis effected a 1:4 reverse stock split of its common stock. Upon the closing of the merger, (1) a wholly-owned subsidiary of Nivalis merged with and into Alpine, with Alpine (renamed as “AIS Operating Co., Inc.”) remaining as the surviving entity; and (2) Nivalis was renamed as “Alpine Immune Sciences, Inc.”Under the terms of the Merger Agreement, Nivalis issued shares of its common stock to Alpine’s stockholders, at an exchange rate of 0.4969 shares of Nivalis common stock, after taking into account the 1:4 reverse stock split, for each share of Alpine’s common stock and preferred stock outstanding immediately prior to the merger. The exchange rate was determined through arms-length negotiations between Nivalis and Alpine. Nivalis also assumed all of the stock options outstanding under Alpine’s Amended and Restated 2015 Stock Plan, as amended (the “Alpine Plan”), and stock warrants for Alpine’s capital stock outstanding immediately prior to the merger, with such stock options and warrants henceforth representing the right to purchase a number of shares of the Nivalis common stock equal to 0.4969 multiplied by the number of shares of Alpine’s common stock or preferred stock previously represented by such options and warrants. Nivalis also assumed the Alpine Plan. Immediately after the merger, there were 13,881,645 shares of common stock outstanding. Immediately after the merger, Alpine’s former stockholders, warrantholders, and optionholders owned, or held rights to acquire, approximately 74% of the fully-diluted common stock of Nivalis, which for these purposes is defined as the outstanding common stock of Nivalis, plus “in the money” options and warrants to purchase shares of Nivalis’ common stock, assuming all “in the money” options and warrants of Nivalis outstanding immediately prior to the merger are exercised on a cashless basis immediately prior to the closing of the merger, with Nivalis’ stockholders, optionholders, and warrantholders immediately prior to the merger owning, or holding rights to acquire, approximately 26% of the fully diluted common stock of Nivalis.
The issuance of shares of Nivalis’ common stock to our pre-existing stockholders was registered with the SEC pursuant to the Registration Statement. Immediately prior to the merger, we issued and sold an aggregate of approximately $17.0 million of shares of our capital stock to certain existing stockholders. For accounting purposes, our historical financial statements were not adjusted to reflect the merger, other than adjustments to the capital structure to reflect the historical capital structure of Nivalis. No other adjustments to our historical assets and liabilities were made as a result of the merger.
In addition to the operating assets and liabilities of Nivalis, we also acquired Nivalis’ tax attributes, which primarily consisted of net operating losses which begin to expire in 2032. Our ability to utilize the tax attributes of Nivalis may be limited under Section 382 of the U.S. Internal Revenue Service and as such, have been reserved. We recorded a deferred tax liability related to future tax benefits arising from IPR&D acquired in the Merger. The combined organization is focusing on the development and commercialization of our innovative immunotherapies. Following the merger, the increased cash resources and increased access to capital of the combined organization will help to support the clinical development of our products.
Consideration Transferred
The fair value of the consideration transferred was based on the most reliable measure, which was determined to be the market price of Nivalis shares of common stock as of the acquisition date. The fair value of the consideration transferred consisted of the following (in thousands except share and per share amounts): |
| | | |
Outstanding Nivalis common stock | 3,914,058 |
|
Per share fair value of Nivalis common stock | $ | 9.60 |
|
Outstanding Nivalis stock options | 421,992 |
|
Weighted average per share fair value of Nivalis stock options | $ | 1.25 |
|
Total fair value of consideration (in 000’s) | $ | 38,103 |
|
Pursuant to the Merger Agreement, unvested Nivalis stock options immediately vested as of the closing of the business combination and were adjusted to give effect to the recapitalization.
Purchase Price Allocation
As Alpine was the accounting acquirer in the merger, we allocated the purchase price to the acquired tangible and intangible assets and assumed liabilities of Nivalis based on their estimated fair values as of the acquisition date. The excess of the estimated fair values of net assets acquired over the acquisition consideration paid was recorded as a bargain purchase gain in the Consolidated Statements of Operations and Comprehensive Income (Loss). The determination of the fair values of the assets acquired and liabilities assumed requires significant judgment, including third party valuation estimates relating to the value of the acquired IPR&D. The allocation of the purchase consideration to the assets acquired and liabilities assumed in our financial statements was finalized as of December 31, 2017. The final allocation of the purchase consideration is as follows (in thousands): |
| | | |
Assets: | Fair Value |
Cash and cash equivalents | $ | 31,130 |
|
Marketable securities | 12,952 |
|
Other receivables | 79 |
|
IPR&D | 1,453 |
|
Total assets acquired | 45,614 |
|
Liabilities: | |
|
Accrued liabilities | (401 | ) |
Deferred tax liability | (509 | ) |
Total liabilities assumed | (910 | ) |
Bargain purchase gain | (6,601 | ) |
Total | $ | 38,103 |
|
We relied on significant Level 3 unobservable inputs to estimate the fair value of our acquired IPR&D using management’s estimate of future royalties and expected earnings of the assets after taking into account an estimate of future expenses necessary to bring the products to completion. These projected cash flows were then discounted to their present values using a discount rate of 17%, which was considered commensurate with the risks and stages of development of the IPR&D.
The bargain purchase gain resulted from expenses incurred by Nivalis between the time the purchase price was negotiated and the close of the transaction, and changes in the Nivalis stock price during that period as the exchange ratio was fixed when the purchase price was negotiated.
Pro Forma Financial Information
The following pro forma consolidated results of net loss for the year ended December 31, 2017 assume the business combination was completed as of January 1, 2017 (in thousands, except per share amounts): |
| | | |
| Year Ended December 31, |
| 2017 |
Pro forma revenues | $ | 1,731 |
|
Pro forma net loss | $ | (18,327 | ) |
Pro forma basic and diluted net loss per share | $ | (1.32 | ) |
For purposes of the pro forma disclosures above, the primary adjustments for the year ended December 31, 2017 includes the elimination of acquisition related costs and acceleration of stock compensation expense upon the change in control.
4.3. Net Loss Per Share
Basic net loss per share is computed by dividing net loss by the weighted-average number of common shares outstanding during the period.
The net loss per share for the year ended December 31, 20192022 reflects 4,706,700the impact to weighted-average shares from issuance of 15,509,282 shares of our common stock issued pursuant to a private placement financing completedthat were sold in January 2019.September 2022 in an underwritten public offering, including the subsequent partial exercise of the underwriters’ over-allotment option in October 2022. The net loss per share for the year ended December 31, 2017 includes the conversion of 9,301,4332021 reflects 6,489,357 shares of our convertible preferred stock into common stock pursuant to a private placement financing completed in September 2021 and 3,914,058951,980 shares acquiredof our common stock issued in connectionDecember 2021 pursuant to a private placement financing with Horizon. The net loss per share for the mergeryear ended December 31, 2020 reflects 5,139,610 shares of our common stock issued pursuant to the securities offering completed in July 2017. 2020. The significant number of shares issued has affectedduring the year-over-yearperiods presented impacts the comparability of our net loss per share calculations.for each period.
The common stock issuable upon the conversion or exercise of the following dilutive securities has been excluded from the diluted net loss per share calculation because their effect would have been antidilutive.anti-dilutive. Diluted net loss per share, therefore, does not differ fromis the same as basic net loss per share for the periods presented. |
| | | | | | | | |
| December 31, |
| 2019 | | 2018 | | 2017 |
Warrants to purchase common stock | 1,877,094 |
| | 24,123 |
| | 24,123 |
|
Options to purchase common stock | 3,252,144 |
| | 2,509,850 |
| | 1,611,996 |
|
Total | 5,129,238 |
| | 2,533,973 |
| | 1,636,119 |
|
| | | | | | | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 | | 2020 |
| | | | | |
| Common stock warrants | 3,656,497 | | | 3,666,435 | | | 3,673,551 | |
| Prefunded warrants to purchase common stock | 5,182,197 | | | 5,182,197 | | | 790,710 | |
| Stock options and RSUs outstanding | 7,236,758 | | | 5,877,309 | | | 4,175,345 | |
| Total | 16,075,452 | | | 14,725,941 | | | 8,639,606 | |
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
4. Cash Equivalents and Short-Term Investments
The amortized cost and fair value of our cash equivalents and short-term investments are as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2022 |
| Amortized Cost | | Gross unrealized gains | | Gross unrealized losses | | Fair market value |
| Money market funds | $ | 11,004 | | | $ | — | | | $ | — | | | $ | 11,004 | |
| U.S. treasury bills | 113,964 | | | 5 | | | (721) | | | 113,248 | |
| U.S. agency securities | 10,921 | | | 5 | | | (11) | | | 10,915 | |
| Non-U.S. government securities | 6,060 | | | — | | | (22) | | | 6,038 | |
| Corporate debt securities and commercial paper | 129,940 | | | 4 | | | (399) | | | 129,545 | |
| Total | $ | 271,889 | | | $ | 14 | | | $ | (1,153) | | | $ | 270,750 | |
| | | | | | | |
| Classified as: | | | | | | | |
| Cash equivalents | | | | | | | $ | 11,004 | |
| Short-term investments | | | | | | | 224,265 | |
| Long-term investments | | | | | | | 35,481 | |
| Total | | | | | | | $ | 270,750 | |
| | | December 31, 2019 | | December 31, 2021 |
| Amortized
Cost | | Gross
unrealized
gains | | Gross
unrealized
losses | | Fair market
value | | Amortized Cost | | Gross unrealized gains | | Gross unrealized losses | | Fair market value |
Money market funds | $ | 9,995 |
| | $ | — |
| | $ | — |
| | $ | 9,995 |
| Money market funds | $ | 50,277 | | | $ | — | | | $ | — | | | $ | 50,277 | |
U.S. treasury bills | 5,019 |
| | 2 |
| | — |
| | 5,021 |
| U.S. treasury bills | 30,006 | | | 1 | | | (100) | | | 29,907 | |
| Non-U.S. government securities | | Non-U.S. government securities | 3,090 | | | — | | | (10) | | | 3,080 | |
Corporate debt securities and commercial paper | 21,862 |
| | 14 |
| | — |
| | 21,876 |
| Corporate debt securities and commercial paper | 114,402 | | | 2 | | | (129) | | | 114,275 | |
Total | $ | 36,876 |
| | $ | 16 |
| | $ | — |
| | $ | 36,892 |
| Total | $ | 197,775 | | | $ | 3 | | | $ | (239) | | | $ | 197,539 | |
| | Classified as: | | Classified as: | |
| Cash equivalents | | Cash equivalents | | $ | 50,277 | |
| Short-term investments | | Short-term investments | | 94,396 | |
| Long-term investments | | Long-term investments | | 52,866 | |
| Total | | Total | | $ | 197,539 | |
|
| | | | | | | | | | | | | | | |
| December 31, 2018 |
| Amortized Cost | | Gross unrealized gains | | Gross unrealized losses | | Fair market value |
Money market funds | $ | 6,405 |
| | $ | — |
| | $ | — |
| | $ | 6,405 |
|
U.S. treasury bills | 13,966 |
| | — |
| | (2 | ) | | 13,964 |
|
Corporate debt securities and commercial paper | 31,331 |
| | — |
| | (11 | ) | | 31,320 |
|
Total | $ | 51,702 |
| | $ | — |
| | $ | (13 | ) | | $ | 51,689 |
|
All short-term investments held as of December 31, 20192022 and 20182021 were classified as available-for-sale debt securities and consist of highly liquid funds with high credit ratings that, on their date of purchase, had a contractual maturitiesmaturity of less than one year.two years or less. There were 0no realized gains andor losses on these securities for the periods presented. Unrealized gains and losses on these investments were primarily due to changes in interest rates. The aggregate fair value of available-for-sale debt securities in an unrealized loss position was $163.9 million and $83.0 million as of December 31, 2022 and 2021, respectively. Investments in a continuous unrealized loss position for more than twelve months related to U.S. treasury bills, non-U.S. government securities, and corporate debt securities and commercial paper, which had a carrying value of $42.8 million and gross unrealized losses of $0.6 million as of December 31, 2022; there were none as of December 31, 2021. We evaluated our investments that are in an unrealized loss position and believe it is more likely than not that we will hold these investments until maturity and will recover the amortized cost basis of these investments.
6.5. Fair Value Measurements
Cash and cash equivalents, restricted cash, receivables, accounts payable and accrued liabilities, which are recorded at invoiced amount or cost, approximate fair value based on the short-term nature of these financial instruments. Fair value is defined as the exchange price received for an asset or paid to transfer a liability, or an exit price, in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value must maximize the use of observable inputs and minimize the use of unobservable inputs. The three-tier fair value hierarchy, which prioritizes the inputs used in measuring fair value, is as follows:
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Observable inputs other than Level 1 prices, such as quoted prices for similar assets or liabilities, quoted prices in inactive markets, or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3: Unobservable inputs supported by little or no market activity and significant to the fair value of the assets or liabilities.
As of December 31, 20192022 and 2018,2021, cash of $3.6$2.4 million and $614,000,$17.6 million, respectively, is excluded from the fair value table below. The following tables summarize our financial assets and liabilities measured at fair value on a recurring basis (in thousands): | | Assets: | December 31, 2019 | Assets: | December 31, 2022 |
| Level 1 | | Level 2 | | Level 3 | | Total | | Level 1 | | Level 2 | | Level 3 | | Total |
Money market funds | $ | 9,995 |
| | $ | — |
| | $ | — |
| | $ | 9,995 |
| Money market funds | $ | 11,004 | | | $ | — | | | $ | — | | | $ | 11,004 | |
U.S. treasury bills | 5,021 |
| | — |
| | — |
| | 5,021 |
| U.S. treasury bills | 113,248 | | | — | | | — | | | 113,248 | |
| U.S. agency securities | | U.S. agency securities | — | | | 10,915 | | | — | | | 10,915 | |
| Non-U.S. government securities | | Non-U.S. government securities | — | | | 6,038 | | | — | | | 6,038 | |
Corporate debt securities and commercial paper | — |
| | 21,876 |
| | — |
| | 21,876 |
| Corporate debt securities and commercial paper | — | | | 129,545 | | | — | | | 129,545 | |
Total | $ | 15,016 |
| | $ | 21,876 |
| | $ | — |
| | $ | 36,892 |
| Total | $ | 124,252 | | | $ | 146,498 | | | $ | — | | | $ | 270,750 | |
|
| | | | | | | | | | | | | | | |
Assets: | December 31, 2018 |
| Level 1 | | Level 2 | | Level 3 | | Total |
Money market funds | $ | 6,405 |
| | $ | — |
| | $ | — |
| | $ | 6,405 |
|
U.S. treasury bills | 13,964 |
| | — |
| | — |
| | 13,964 |
|
Corporate debt securities and commercial paper | — |
| | 31,320 |
| | — |
| | 31,320 |
|
Total | $ | 20,369 |
| | $ | 31,320 |
| | $ | — |
| | $ | 51,689 |
|
| | | | | | | | | | | | | | | | | | | | | | | |
| Assets: | December 31, 2021 |
| Level 1 | | Level 2 | | Level 3 | | Total |
| Money market funds | $ | 50,277 | | | $ | — | | | $ | — | | | $ | 50,277 | |
| U.S. treasury bills | 29,907 | | | — | | | — | | | 29,907 | |
| Non-U.S. government securities | — | | | 3,080 | | | — | | | 3,080 | |
| Corporate debt securities and commercial paper | — | | | 114,275 | | | — | | | 114,275 | |
| Total | $ | 80,184 | | | $ | 117,355 | | | $ | — | | | $ | 197,539 | |
Our Level 2 assets consist of commercial paperU.S. agency securities, non-U.S government securities, and corporate debt securities.securities and commercial paper. We review trading activity and pricing for our available-for-sale securities as of the measurement date. When sufficient quoted pricing for identical securities is not available, we use market pricing and other observable market inputs for similar securities obtained from various third-party data providers. These inputs either represent quoted prices for similar assets in active markets or have been derived from observable market data.
7.6. Property and Equipment
Property and equipment, net, consistconsists of the following (in thousands):
|
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Laboratory equipment | $ | 1,838 |
| | $ | 1,506 |
|
General equipment and furniture | 486 |
| | 158 |
|
Computer equipment and software | 169 |
| | 103 |
|
Leasehold improvements | 85 |
| | 128 |
|
Property and equipment, at cost | 2,578 |
| | 1,895 |
|
Less accumulated depreciation and amortization | (1,026 | ) | | (699 | ) |
Property and equipment, net | $ | 1,552 |
| | $ | 1,196 |
|
| | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Laboratory equipment | $ | 3,191 | | | $ | 3,132 | |
| General equipment and furniture | 459 | | | 479 | |
| Computer equipment and software | 217 | | | 211 | |
| Leasehold improvements | 118 | | | 85 | |
| Property and equipment, at cost | 3,985 | | | 3,907 | |
| Less accumulated depreciation and amortization | (2,401) | | | (2,191) | |
| Property and equipment, net | $ | 1,584 | | | $ | 1,716 | |
Depreciation expense was $468,000, $388,000$597,000, $620,000 and $241,000$578,000 for the years ended December 31, 2019, 20182022, 2021 and 2017,2020, respectively.
8. Loss on Sale of Intangible Asset
In February 2018, we entered into an Option License Agreement (“Option Agreement”) with Laurel Venture Capital Ltd. (“Laurel”), which granted Laurel a limited license to evaluate the indefinite-life GSNOR inhibitor IPR&D asset acquired as part of the merger with Nivalis in 2017. The IPR&D represents the processes, expertise, and technology employed in the development of GSNOR inhibitors and Nivalis’ lead product candidate, cavosonstat. Under the Option Agreement, we received an upfront non-refundable payment of $75,000, which was recognized as revenue in our accompanying Consolidated Statements of Operations and Comprehensive Income (Loss).In June 2018, we entered into an Asset Purchase Agreement (“Purchase Agreement”) with Laurel and completed the sale of global rights to the GSNOR asset. As consideration under the Purchase Agreement, we received a non-refundable closing payment of $250,000, which was accounted for as a purchase of our intangible asset. In June 2019, we recognized as revenue an additional payment of $425,000, related to the asset purchase. In addition, we are eligible to receive milestone payments of up to $20.0 million, in the aggregate upon satisfaction by Laurel of certain regulatory approval milestones. We will also be eligible to receive royalty payments equal to a low single-digit percentage rate of worldwide net sales of any approved products.
ALPINE IMMUNE SCIENCES, INC.
9.7. Additional Balance Sheet Information
Prepaid expenses and other current assets consist of the following (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Tennant improvement allowance receivable | $ | 586 |
| | $ | — |
|
Prepaid research and development | 574 |
| | 83 |
|
Prepaid insurance | 301 |
| | 300 |
|
Deferred financing costs | 15 |
| | 477 |
|
Prepaid other | 74 |
| | 145 |
|
Other receivables | 100 |
| | 237 |
|
Prepaid expenses and other current assets | $ | 1,650 |
| | $ | 1,242 |
|
| | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Prepaid research and development | $ | 1,881 | | | $ | 3,315 | |
| Prepaid insurance | 368 | | | 454 | |
| Prepaid other | 394 | | | 266 | |
| Deferred financing | — | | | 352 | |
| Other receivables | 317 | | | 323 | |
| Prepaid expenses and other current assets | $ | 2,960 | | | $ | 4,710 | |
Accrued liabilities consist of the following (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Research and development services | $ | 2,543 |
| | $ | 2,457 |
|
Employee compensation | 1,761 |
| | 1,009 |
|
Legal and professional fees | 515 |
| | 646 |
|
Accrued other | 466 |
| | 251 |
|
Total | $ | 5,285 |
| | $ | 4,363 |
|
| | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Research and development services | $ | 7,657 | | | $ | 5,536 | |
| Employee compensation | 4,110 | | | 3,084 | |
| Accrued taxes | 1,847 | | | 127 | |
| Legal and professional fees | 334 | | | 394 | |
| Accrued other | 55 | | | 276 | |
| Accrued liabilities | $ | 14,003 | | | $ | 9,417 | |
10.8. Long-term Debt
In December 2016, we entered into a Loan and Security Agreement (the “Original Agreement”), with SVB, under which we borrowed $5.0 million. The Original Agreement accrued interest at a floating per annum rate equal to the lender’s prime rate minus 1.75%. The Original Agreement had an interest-only period through July 2018.
On August 26, 2019, (the “Effective Date”), we entered into an Amended and Restated Loan and Security Agreement (the “Loan Agreement”“Loan Agreement”) with SVB,Silicon Valley Bank (“SVB”), pursuant to which SVB agreed to extend term loans to us with an aggregate principal amount of up to $15.0$15.0 million (the “Term Loans”“Term Loans”). Borrowings under the Loan Agreement consistconsisted of up to 3three separate tranches. The initial tranche of $5.0$5.0 million was funded in August 2019, $3.0$3.0 million of which was used to repay amounts owing under our Original Agreement. We intend to use the remaining proceeds for potential working capitalprior loan and other general corporate purposes, including the advancement of our development programs.security agreement with SVB. In March 2020, the second tranche of $5.0$5.0 million was funded to us. The third andWe did not draw down the final tranche of up to $5.0$5.0 million, is available at our option at any time from the date on which SVB receives and approves evidence that we have initiated a phase 2a trial of ALPN-101expired in psoriatic arthritis through July 31, 2020.
The Term Loans accrue interest at a floating per annum rate of 0.25% above the prime rate, subject to a floor of 5.75%, which interest is payable monthly commencing in September 2019. Upon the occurrence and during the continuance of an event of default, a default interest rate will apply that is 4.0% above the otherwise applicable interest rate. The Term Loans arewere interest only until September 30, 2020, after whichhowever, under the Loan Agreement our interest only period automatically extended to June 30, 2021 if we received aggregate new capital of at least $40.0 million no later than June 30, 2020. We met this milestone in June 2020 in conjunction with the execution of the agreement with AbbVie Ireland Unlimited Company (“AbbVie”), discussed in detail in Note 10. As a result of the interest only extension, the Term Loans will beare payable in 3425 equal monthly installments of principal plus interest, with the final installment due and payable on July 1, 2023. We may prepay all, but not less than all, of the Term Loans subject to a prepayment fee equal to $75,000, which represents the deferred portion of the final payment due under the Original Agreement, plus the outstanding principal balance under the Term Loans at the time of such prepayment multiplied by a prepayment fee of 2.0% in the first year, 1.0% in the second year, and 0% in the third year and thereafter.$75,000. Additionally, a final payment in the amount of 5.5% of the funded Term Loans is payable to SVB on the date on which the Term Loans are prepaid, paid or become due and payable in full. The final payment fees are recorded in long-term debt with an offsetting reduction to debt discount onin our accompanying Consolidated Balance Sheets.
The Loan Agreement contains customary representations and warranties, events of default and affirmative and negative covenants, including, among others, covenants that limit or restrict our ability to, among other things, incur additional
indebtedness, grant liens, merge or consolidate, make acquisitions, pay dividends or other distributions or repurchase equity, make investments, dispose of assets, engage in any new lines of business, and enter into certain transactions with affiliates, in each case subject to certain exceptions. We assessed the likelihood of the lender accelerating payment of the loan due to a material adverse change in our business, operations, financial, or other condition as remote. We were in compliance with our covenants as of December 31, 2019. As such, as of December 31, 2019, the classification of the loan is split between current and noncurrent based on the timing of payment obligations.2022. As security for itsour obligations under the Loan Agreement, we granted SVB a first priority security interest on substantially all of our assets, except intellectual property, and subject to certain other exceptions.
In connection with the Loan Agreement, we issued a warrant to SVB to purchase up to 52,083 shares of our common stock at a price of $4.32$4.32 per share, 17,361 shares of which became exercisable in August 2019 after we drew down the initial tranche. In March 2020, after we drew down the second tranche of our Term Loan,Loans, an additional 17,361 shares became exercisable. If we draw downThe remaining warrants did not vest and expired upon the expiration of the third tranche additional shares will become exercisable.of our Term Loans. The fair value of the warrants on the date of issuance for the initial tranche and second tranche was $60,000 and $60,000,
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
$60,000,respectively, determined using the Black-Scholes option-pricing model, and was recorded as a component of equity and as a debt discount onin our accompanying Consolidated Balance Sheets. In connection with Original Agreement, SVB also holds 7,069 fully vested common stock warrants at an exercise price of $12.38 per share. The Term Loan was accounted for as a debt modification in a non-troubled debt restructuring, rather than a debt extinguishment, based on a comparison of the present value of the cash flows under the terms of the debt immediately before and after the Effective Date of the Term Loan, which resulted in a change of less than 10%. As a result, the remaining unamortized debt discount recorded in connection with the Original Agreement will be amortized to interest expense over the repayment term of Loan Agreement. In connection with the Loan Agreement, weWe recorded a total debt discount of $477,000,$812,000 in connection with the Loan Agreement, which is being amortized to interest expense using the effective interest method over the repayment term of the loan. Non-cash interestThe unamortized discount included in the current portion of long-term debt in our accompanying Consolidated Balance Sheets was $45,000 as of December 31, 2022. Interest expense associated with the amortization of the discount wasis recorded as non-cash interest expense on our accompanying $140,000Consolidated Statements of Cash Flows, $169,000, and $87,000, for years ended . As of December 31, 2019, 2018,2022, $2.8 million in remaining principle and 2017, respectively. The unamortized discount was $423,000 as of December 31, 2019.Scheduled principal payments on our outstanding debt as of December 31, 2019$625,000 in final payment fees due under our Loan Agreement excluding final fee amounts, are as follows (in thousands):classified within current portion of long-term debt in our accompanying |
| | | | |
Year Ending December 31, | | Total |
2020 | | $ | 441 |
|
2021 | | 1,765 |
|
2022 | | 1,765 |
|
2023 | | 1,029 |
|
2024 | | — |
|
Total future principal payments | | $ | 5,000 |
|
11.9. Commitments and Contingencies
Operating Leases
We leased office and laboratory space located at 201 Elliott Avenue West, in Seattle, Washington, under an agreement classified as an operating lease that expired on December 31, 2019. In May 2017, as required by the terms of the lease, we entered into a line of credit to establish collateral to support the security deposit in an amount of $132,000. This is recorded as current restricted cash in the accompanying Consolidated Balance Sheets. In March 2019, we entered into a lease for office and laboratory space located at 188 East Blaine Street, Seattle, Washington. The term of the lease is 10.8 years with 1one option to extend the term by 5.0 years.years. Our option to extend the rental term of our lease was not considered reasonably certain as of December 31, 2022. The lease term commenced in June 2019. The “Rent“Rent Commencement Date” will beDate” began in March 2020, nine months after the commencement date. The annual base rent under the lease is $1.7$1.7 million for the first year and will increase by 3.0% each year thereafter. We arewere not required to pay base rent from the Rent Commencement Date through November 2020, the last day of the ninth month following the Rent Commencement Date. We will receivereceived a maximum tenant improvement allowance of $5.4$5.4 million,, which is included in our base rent, and a maximum additional tenant improvement allowance of $1.8$1.8 million,, which will result in additional rent amortized over the term of the lease at an annual rate of 8.0%. The lease also requires us to pay additional amounts for operating and maintenance expenses. In March 2019, in connection with the lease, we provided a $254,000$254,000 letter of credit as a security deposit, which is recorded as noncurrent restricted cash in our accompanying Consolidated Balance Sheets. Upon adoption of ASC Topic 842, Leases, on January 1, 2019, we recognized As$883,000 of operating lease liabilities and $797,000 of operating lease right-of-use assets for our existing leases on our consolidated balance sheet. As of December 31, 20192022, our operating lease right-of-use assets and operating lease liability associated with our leases were $10.0
$8.2 million and $11.4$10.8 million,, respectively. The increases in our operating lease liabilities and operating lease right-of-use assets during 2019 reflect our new lease that commenced in June 2019. Our option to extend the rental term of our lease was not considered reasonably certain as of December 31, 2019.
Supplemental operating lease information for the year ended December 31, 2019 wasis as follows (in thousands): |
| | | |
| Twelve months ended December 31, 2019 |
Operating lease cost | $ | 1,905 |
|
Variable lease cost | 370 |
|
Total lease cost | $ | 2,275 |
|
Cash paid for amounts included in the measurement of lease liabilities | $ | 837 |
|
Right-of-use assets exchanged for new operating lease liabilities | $ | 11,173 |
|
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2022 | | 2021 | | 2020 |
| Operating lease cost | $ | 1,839 | | $ | 1,837 | | $ | 1,872 |
| Variable lease cost | 823 | | 603 | | 475 |
| Total lease cost | $ | 2,662 | | $ | 2,440 | | $ | 2,347 |
| Other information: | | | | | |
| Cash paid for amounts included in the measurement of lease liabilities | $ | 2,009 | | | $ | 2,117 | | $ | 213 |
| | | | | |
| Weighted-average remaining lease term (years) | 7.2 | | 8.2 | | 9.2 |
| Weighted-average discount rate | 10.7% | | 10.7% | | 10.7% |
Variable lease costs represent our share of the landlord’slandlord’s operating expenses. We do not act as a lessor or have any leases classified as financing leases. As of
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
December 31, 2019, the weighted average remaining lease term for our operating lease was 10.2 years and the weighted average discount for our operating leases was 10.7%. Maturities of our operating lease liabilities as of December 31, 20192022 are as follows (in thousands): | | | | | |
| | Minimum Lease Payments |
| 2023 | $ | 1,889 | |
| 2024 | 2,111 | |
| 2025 | 2,167 | |
| 2026 | 2,224 | |
| 2027 | 2,283 | |
| Thereafter | 5,155 | |
| Total future minimum lease payments | 15,829 | |
| Less: imputed interest | (4,988) | |
| Operating lease liabilities | $ | 10,841 | |
|
| | | |
| Minimum Lease Payments |
2020 | $ | 216 |
|
2021 | 1,961 |
|
2022 | 2,012 |
|
2023 | 2,065 |
|
2024 | 2,119 |
|
Thereafter | 11,870 |
|
Total future minimum lease payments | 20,243 |
|
Less: imputed interest | (8,814 | ) |
Operating lease liabilities | $ | 11,429 |
|
Rent expense, which isCertain credits received related to our research and development expenditures were previously recorded on a straight-line basis, waswithin other income within our $811,000Consolidated Statements of Operations and Comprehensive Income (Loss) were subject to review by foreign taxing authorities. We reached constructive agreement with the Australian Taxation Office during 2022 and $529,000recorded an estimated current foreign income tax provision of $1.3 million for the yearsexpected repayments, of which we paid $543,000. The remaining $849,000 is included as accrued taxes within accrued liabilities on our accompanying Consolidated Balance Sheets.10. License and Collaboration Agreements
AbbVie
In June 2020, we entered into an option and license agreement with AbbVie (the “AbbVie Agreement”) for the development of ALPN-101, (“acazicolcept”). The AbbVie Agreement grants AbbVie the exclusive option to purchase an exclusive worldwide license to acazicolcept (the “License Option”). The License Option is exercisable by AbbVie at any time and will expire 90 days from the achievement of certain development milestones. If AbbVie exercises the License Option, AbbVie will take over the future development and commercialization. Prior to the exercise of the License Option, we will perform research and development services, including conducting our Phase 2 study in systemic lupus erythematosus, based on an agreed-upon development plan (the “Development Plan”). We will be fully responsible for all costs incurred to conduct the activities under the Development Plan, provided that, AbbVie may be responsible for increased costs under the Development Plan in connection with certain material amendments proposed by AbbVie. We will also be solely responsible, at our sole cost and expense, for manufacturing and regulatory filings for acazicolcept necessary to complete activities under the Development Plan.
In June 2020, in connection with the execution of the AbbVie Agreement, AbbVie paid us a nonrefundable upfront payment of $60.0 million. Prior to the exercise of the License Option, AbbVie has agreed to make cash payments upon our achievement of certain predefined pre-option development milestones (the “Alpine Development Milestones”) up to an aggregate amount of $75.0 million. In 2021, we received $45.0 million of the Alpine Development Milestones. If AbbVie exercises the License Option, they will pay a one-time cash payment of $75.0 million. Following the exercise of the License Option, AbbVie has also agreed to make aggregate cash payments of up to $205.0 million upon AbbVie’s achievement of certain development and commercial milestones and additional aggregate cash payments of up to $450.0 million upon AbbVie’s achievement of certain sales-based cash milestones, collectively referred to as (the “AbbVie Milestones”). Subsequent to commercialization, we are also eligible to receive high single-digit to low double-digit percentage royalties on worldwide net sales of licensed products.
For revenue recognition purposes, we determined that our contractual promises in the AbbVie Agreement are not distinct and are interdependent with our performance obligation to provide research and development services under the Development Plan. Thus, all contractual promises related to the upfront payment and Alpine’s Development Milestones were combined into a single performance obligation. We determined the Alpine Development Milestone payments are probable of significant revenue reversal as the achievement is highly dependent on factors outside our control. Therefore, these milestone payments were fully constrained and were not initially included in the transaction price. In June 2021, we re-evaluated and updated the transaction price to include the achieved portion of the Alpine Development Milestones. We will continue to re-evaluate the transaction price each reporting period and update as uncertain events are resolved or other changes in circumstances occur. We expect to recognize the remaining deferred revenue over the remaining life of the Development Plan, which began in June 2020 and ends upon the later of the exercise or expiration of the option.
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
The License Option and the AbbVie Milestones were determined not to be performance obligations at the inception of the contract as they did not represent material rights. If exercised, the License Option and AbbVie Milestones will be accounted for as a separate contract and will be recognized as revenue if and when triggered. Any consideration related to sales-based royalties and profit-sharing payments will be recognized when the related sales occur.
Horizon
In December 2021, we entered into an exclusive license and collaboration agreement with Horizon (the “Horizon Agreement”) for the development and commercialization of up to four preclinical candidates generated from our unique discovery platform. The agreement includes licensing of one of our existing preclinical biologic therapeutic programs (the “Existing Program”) as well as a research partnership to jointly develop candidates for up to three additional autoimmune and inflammatory disease programs for other designated biological targets (the “Research Programs”). These candidates include previously undisclosed multi-specific fusion protein-based therapeutic candidates for autoimmune and inflammatory diseases. We will advance candidate molecules to predefined preclinical milestones while Horizon will be responsible for the respective costs and, ultimately, Horizon will assume responsibility for development and commercialization activities and costs.
In connection with the execution of the Horizon Agreement in December 2021, we entered into a stock purchase agreement under which Horizon purchased 951,980 shares of our common stock in a private placement for approximately $15.76 per share and aggregate proceeds of $15.0 million. The shares were sold at a 25% premium to the volume-weighted average share price of our common stock for a specified 30-day period prior to entering into the agreement. The fair value of the common stock issued to Horizon of $11.9 million was recorded to equity, based on the closing price of common stock on the effective date of the Horizon Agreement. For accounting purposes, the $3.1 million difference between the cash proceeds and the fair value of the common stock was treated as additional consideration attributable to the Horizon Agreement.
Under the terms of the agreements, Horizon also paid us a non-refundable upfront payment of $25.0 million in the first quarter of 2022. In addition, we are eligible to receive up to $381.0 million per program, or up to approximately $1.5 billion in total, in future success-based payments related to development, regulatory and commercial milestones. Furthermore, we are eligible to receive tiered royalties from a mid-single digit percentage to a low double-digit percentage on global net sales. In addition to proceeds from the non-refundable upfront payment, we have recognized $839,000 in research and development support provided by us during the year ended December 31, 20182022.
For revenue recognition purposes, we determined the transaction price at inception was $28.1 million, which consists of the upfront payment of $25.0 million and 2017, respectively.the $3.1 million premium on the stock purchase, and that the Existing Program and each Research Program are distinct performance obligations. We allocated revenue to each performance obligation based on its relative stand-alone selling price. The future success-based payments related to development and regulatory milestones are probable of significant revenue reversal as the achievement is highly dependent on factors outside our control. Therefore, these milestone payments are fully constrained and are not initially included in the transaction price. We will continue to re-evaluate the transaction price each reporting period and update as uncertain events are resolved or other changes in circumstances occur. Any consideration related to commercial milestones and royalties will be recognized when the related sales occur.
12. Collaboration and License Agreement
Adaptimmune
In May 2019, we entered into a collaboration and licensing agreement with Adaptimmune (the “Adaptimmune Collaboration Agreement”Therapeutics plc (“Adaptimmune”) to develop next-generation SPEAR T-cell products.T cell products (the “Adaptimmune Agreement”). Under the Adaptimmune Collaboration Agreement, we are to perform certain research services and grant Adaptimmune an exclusive license to programs from our secreted immunomodulatory protein (“SIP”(“SIP”) and transmembrane immunomodulatory protein (“TIP”(“TIP”) technologies. In June 2019,
Through December 31, 2022, we have recorded a total of $3.0 million in license payments under the terms of the Adaptimmune Collaboration Agreement we received anconsisting of a $2.0 million upfront license payment of $2.0 million,received in June 2019 and as of December 31, 2019 we have received an additional $750,000$1.0 million license fee upon Adaptimmune’s selection of an additional research program in June 2022. We have also recorded $2.0 million in research support payments to fund ongoing programs. These payments were recorded as deferred revenue and will be recognized to revenue based on employee hours contributed to each performance obligation. We have recognized a total of $1.3 million in revenue for the year ended programs through December 31, 2019 related to our collaboration agreement with Adaptimmune.2022. In addition, we are eligible for additional research support payments, one-time payments and downstream development and commercialization milestones of up to $288.0$288.0 million,, if allrespective pre-specified milestones for each program are achieved. We are also eligible to receive low-single digit percentage royalties on worldwide net sales of the applicable products.
Kite
In October 2015, we entered into a collaborationFor revenue recognition purposes, licensing and licensing agreement (the “Kite Collaboration Agreement”) with Kite, providing Kite with access to two TIP programs for use in Kite’s engineered cellular therapy program. In May 2019, Kite provided us notice of termination of the Kite Collaboration Agreement following the expiration of the research term. Upon termination, the confidentiality and indemnity obligations of the parties survived and the licenses granted to Kitesupport fees billed under the Kite Collaboration Agreement terminated. Pursuant to the terms of the Kite Collaboration Agreement, the termination was effective in June 2019, thirty days after the effectiveness of Kite’s notice.
Under the terms of the Kite Collaboration Agreement, in 2015, Kite made upfront payments to us of $5.5 million, which were initiallyagreement are being recorded as deferred revenue. Under the Kite Collaboration Agreement, we recorded revenue of $0, $630,000and $1.7 million for the years ended December 31, 2019, 2018 and 2017, respectively.
As discussed in Note 2, on January 1, 2018, we adopted the new revenue standard using the modified retrospective method. Our Kite Collaboration Agreement was the only contract that was impacted by the adoption of the new revenue standard. Priorrecognized to the adoption of the new revenue standard, we recognized revenue under the Collaboration Agreement based upon the estimated performance periods related to the non-refundable upfront payments we received from Kite. Under the new standard, we recognize revenue based on employee hours contributed to each performance obligation.Contract balances and revenue recognition
We report contract assets resulting from unconditional rights to consideration related to upfront payments, and for completed but unpaid research and development services within accounts receivable in our accompanying Consolidated
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
Balance Sheets. Contract liabilities, representing advance consideration for licensing rights bundled with research and development services and other promises for which the underlying performance obligations have not yet been satisfied, are reported as deferred revenue in our accompanying Consolidated Balance Sheets. Contract liabilities are presented as current and noncurrent based on estimated timing of when the underlying performance obligations will be met. Respective balances are as follows (in thousands): | | | | | | | | | | | |
| December 31, |
| 2022 | | 2021 |
| Contract Assets | $ | 392 | | | $ | 25,000 | |
| Contract Liabilities | $ | 74,756 | | | $ | 102,603 | |
We use cost-based input methods to measure progress towards completion of our performance obligations and to calculate the corresponding revenue to recognize under our contracts with customers each period. In applying the cost-based input, we use actual costs incurred relative to budgeted costs for each combined performance obligation. Actual costs consist primarily of labor related to internal personnel and third-party contracts. Revenue is recognized based on the proportion of costs incurred relative to the total estimated costs for the performance obligation. A cost-based input method of revenue recognition requires management to estimate the costs to complete our performance obligations. In making such estimates, significant judgment is required to evaluate assumptions related to cost estimates. The cumulative effect adjustmentof revisions to estimated costs to complete our performance obligations will be recorded uponin the adoptionperiod in which changes are identified and amounts can be reasonably estimated. A significant change in these assumptions and estimates could have a material impact on the timing and amount of revenue recognized in future periods.
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2022 | | 2021 | | 2020 |
| AbbVie | $ | 17,064 | | | $ | 23,443 | | | $ | 7,025 | |
| Horizon | 11,964 | | | — | | | — | |
| Adaptimmune | 1,036 | | | — | | | 2,310 | |
| Total collaboration revenue | $ | 30,064 | | | $ | 23,443 | | | $ | 9,335 | |
Revenue recognized during the new revenue standard resultedperiod that was included in a $203,000 decrease to the opening contract liability balance of retained earningswas $28.2 million, $23.4 million and a $203,000 increase to deferred revenue as of January 1, 2018. As a result, we recognized $203,000 in higher revenue$2.3 million for the yearyears ended December 31, 2018, as compared2022, 2021 and 2020, respectively. The change in the contract liability balance for AbbVie was reduced by a cumulative catch-up adjustment related to what would have been recorded under previous accounting guidance.upward revisions to the projected program duration and corresponding increase in estimated total direct and indirect development costs to complete the program.
13. Stockholders’ Deficit 11. Stockholders’ Equity
Common Stock
Shares of common stock reserved for future issuance were as follows: | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Shares to be issued upon exercise of outstanding stock options | 7,058,140 | | | 5,611,743 | |
| Shares to be issued upon release of RSUs | 178,618 | | | 265,566 | |
| Shares to be issued upon conversion of common stock warrants | 3,656,497 | | | 3,666,435 | |
| Shares to be issued upon conversion of prefunded warrants | 5,182,197 | | | 5,182,197 | |
| Shares available for future stock grants | 314,523 | | | 284,906 | |
| Shares to be issued under employee stock purchase plan | 45,211 | | | 45,211 | |
| Shares of common stock reserved for future issuance | 16,435,186 | | | 15,056,058 | |
|
| | | | | |
| December 31, |
| 2019 | | 2018 |
Shares to be issued upon exercise of outstanding stock options | 3,252,144 |
| | 2,509,850 |
|
Shares to be issued upon conversion of common stock warrants | 1,877,094 |
| | 24,123 |
|
Shares available for future stock grants | 269,959 |
| | 496,530 |
|
Shares to be issued under employee stock purchase plan | 45,211 |
| | 45,211 |
|
Shares of common stock reserved for future issuance | 5,444,408 |
| | 3,075,714 |
|
Securities Offerings
In September 2022, we sold an aggregate of 15,509,282 shares of our common stock in an underwritten public offering, including the subsequent partial exercise of the underwriters’ over-allotment option in October 2022, pursuant to our
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
Securities Offeringseffective shelf registration statement on Form S-3 (File No. 333-256107). We received resulting net proceeds of $106.7 million after deducting underwriting discounts, commissions and offering costs. Offering costs of $448,000 were netted against the proceeds within additional-paid-in-capital on our accompanying Consolidated Balance Sheets.In January 2019,September 2021, we entered into a securities purchase agreement (the ““2021 Securities Purchase Agreement”Agreement”) for a private placement with a limited numberselect group of accreditedinstitutional investors, pursuant to which we sold 4,706,700 units6,489,357 shares of our common stock (the “Units”“Shares”) and prefunded warrants to purchase 3,191,487 Shares (the “Prefunded Warrants”). The purchase price for each Share and for each Prefunded Warrant was $9.40 per share, for an aggregate purchase price of approximately $91.0 million. The Prefunded Warrants became fully exercisable upon the closing date and have an exercise price of $0.001 per share. We incurred $266,000 in financing costs associated with the 2021 Securities Purchase Agreement, which was netted against the proceeds within additional-paid-in-capital on our accompanying $25.3 millionConsolidated Balance Sheets in. In September 2021, we entered into an exchange agreement (the “Exchange Agreement”) with Frazier Life Sciences VIII, L.P. (the “Exchanging Stockholder”), which Exchanging Stockholder is affiliated with a member of our board of directors, pursuant to which we exchanged an aggregate of 1,200,000 shares of common stock held by the Exchanging Stockholder for Prefunded Warrants (the “Exchange Warrants”) to purchase an aggregate of 1,200,000 shares of common stock. Upon the closing of the exchange, we reclassified 1,200,000 shares of common stock into treasury stock on our accompanying Consolidated Balance Sheets. In July 2020, we entered into a securities purchase agreement (the “2020 Securities Purchase Agreement”) for a private placement with a select group of institutional investors, pursuant to which we sold 5,139,610 units (the “Private Placement”“Common Units”). Each Unit has a and 790,710 units (the “Prefunded Warrant Units”), for an aggregate purchase price of $5.37 and$60.0 million. Each Common Unit consists of one share of our common stock andplus a warrant to purchase 0.390.3 shares of common stock.stock (the “Common Stock Warrants”), and each Prefunded Warrant Unit consists of one prefunded warrant to purchase one share of common stock plus 0.3 Common Stock Warrants. The Prefunded Warrant Units and the Common Units are collectively referred to as the “Units” and each Unit has a purchase price of $10.1175. Pursuant to the terms of the 2020 Securities Purchase Agreement, we issued 4,706,700warrants to purchase 1,779,096 shares of common stock with an exercise price of $12.74 and a term of 3.5 years. Additionally, we issued 790,710 prefunded warrants, to purchase an aggregate of 1,835,610 shares of common stock. The warrantswhich became fully exercisable upon the closing date and have an exercise price of $0.001 per share. We incurred $3.7 million in financing costs associated with the 2020 Securities Purchase Agreement, which was netted against the proceeds within additional-paid-in-capital on our accompanying $12.74 and have a term of five yearsConsolidated Balance Sheets. The issuance of the securities sold inunder the Private Placement was2021 and 2020 Securities Purchase Agreements have not been registered under the Securities Act of 1933, as amended, or state securities laws and such securities couldmay not be offered or sold in the United States absent registration with the SEC or an applicable exemption from such registration requirements. In March 2019, weWe filed a registration statementstatements for the 2021 and 2020 Securities Purchase Agreements with the SEC, coveringwhich were declared effective by the SEC in November 2021 and August 2020, respectively, which cover the resale of the shares of common stock issuable in connection with the Private Placementprivate placements and upon exercise of the warrants,warrants. In May 2021, the registration statement for the 2019 Securities Purchase Agreement was deactivated following the expiration of our obligation to maintain its effectiveness under the related registration rights agreement.
Financing Agreements
In September 2022, we and Cowen and Company, LLC (“Cowen”) mutually terminated the sales agreement (the “Sales Agreement”) dated July 2, 2021. The Sales Agreement provided that we may sell shares of our common stock from time to time through an “at the market” equity offering for up to $75.0 million in gross cash proceeds, under which registration was declared effective byCowen acted as the SEC on April 4, 2019.
We have incurred legal, accounting and other directsales agent. No sales of our common stock were sold under the Sales Agreement. In connection with the termination of the Sales Agreement, we wrote off $488,000 of deferred financing costs related to our efforts to raise capital. These costs have been capitalized as deferred offering costs and areexpense, which were previously included within prepaid expenses and other current assets inon our accompanying Consolidated Balance Sheets. These were deferred until completion of the Private Placement, at which time Sheets$1.7 million were reclassified to additional paid-in capital as a reduction of the proceeds..In June 2018, we entered into an equity distribution agreement, (“Equity Distribution Agreement”), with Piper Jaffray & Co., (“Piper Jaffray”), pursuant to which we may sell shares of our common stock through an “at the market” equity offering program for up to $50.0 million, in gross cash proceeds. Piper Jaffray will be entitled to compensation for its services of up to 3.0% of the gross sales price per share of all shares sold through Piper Jaffray under the Equity Distribution Agreement. The Equity Distribution Agreement may be terminated by us upon written notice to Piper Jaffray for any reason or by Piper Jaffray upon written notice to us for any reason or at any time under certain circumstances, including but not limited to if we experience a material adverse change.
Under the Equity Distribution Agreement, we will set the parameters for the sale of shares, including the number of shares to be issued, the time period during which sales are requested to be made, limitation on the number of shares that may be sold in any one trading day and any minimum price below which sales may not be made. We have no obligation to sell any shares under the Equity Distribution Agreement and may at any time suspend solicitation and offers under the Equity
Distribution Agreement. In July 2019, our Registration Statement on Form S-3 (File No. 333-212404), expired pursuant to Rule 415(a)(5) under the Securities Act of 1933, as amended. We will be unable to sell shares under the Equity Distribution Agreement until a new Registration Statement on Form S-3 is filed and declared effective by the SEC and a prospectus supplement relating to any sales under the Equity Distribution Agreement is filed with the SEC. As of December 31, 2019, no sales under our Equity Distribution Agreement have occurred.
Common Stock Warrants
We have issued warrants in connection with our securities offerings, SVB loans, and to certain non-employee professional advisers, andadvisers. Excluding the prefunded warrants we issued in connection with our Private Placement. We also assumed warrants in connection with securities offerings discussed above,
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
the merger. No warrants were exercised or forfeited duringtable below summarizes our common stock warrant activity: | | | | | | | | | | | | | | | | | |
| | Warrants
Outstanding | | Weighted-
average
Exercise
Price | | Weighted-
average
Remaining
Contract
Term
(in years) |
| Outstanding at December 31, 2021 | 3,666,435 | | | $ | 12.64 | | | 2.12 |
| Exercised | (9,938) | | | 5.02 | | | |
| | | | | |
| Outstanding and Exercisable at December 31, 2022 | 3,656,497 | | | $ | 12.66 | | | 1.12 |
Equity Incentive Plans
Our board of directors approved an “inducement” grant, pursuant to Rule 5635(c)(4) of the
year ended December 31, 2019. A summaryNasdaq Listing Rules, to our Chief Medical Officer for options to purchase 160,000 shares of our warrant activitycommon stock at a per-share exercise price of $8.38, which was set based on our stock price as of the grant date in August 2022. The option grant is presented below: |
| | | | | | | | |
| Warrants Outstanding | | Weighted- average Exercise Price | | Weighted- average Remaining Contract Term (in years) |
Outstanding at December 31, 2018 | 24,123 |
| | $ | 24.86 |
| | 5.62 |
Granted | 1,852,971 |
| | $ | 12.66 |
| | 4.10 |
Outstanding at December 31, 2019 | 1,877,094 |
| | $ | 12.82 |
| | 4.11 |
Exercisable at December 31, 2019 | 1,873,225 |
| | $ | 12.83 |
| | 4.11 |
Equity Incentive Plans
In June 2018, our stockholders approved the 2018 Equity Incentive Plan (“(“2018 Plan”Plan”). Upon adoption, we ceased granting stock awards under the Nivalis Therapeutics, Inc. 2015 Equity Incentive Plan (the “2015 EIP”“2015 EIP”) and the Amended and Restated 2015 Stock Plan (the “2015 Plan”“2015 Plan”), collectively, the “Legacy Plans”“Legacy Plans”. All shares of common stock subject to awards under the Legacy Plans that expire or terminate without having been exercised in full, or are forfeited to or repurchased by the company, will be added to the 2018 Plan, up to a maximum of 1,972,784 shares. In June 2020, in conjunction with our annual meeting of stockholders, our stockholders approved an additional increase of 743,515 shares authorized under our 2018 Plan.
Under our 2018 Plan we may issue stock options, stock appreciation rights, restricted stock, restricted stock unitsRSUs or performance shares. As of December 31, 20192022, we have only issued stock options to purchase shares of common stock.and RSUs. Our 2018 Plan provides for an annual increase in the number of shares reserved for insurance equal to the lesser of (1) 5% of the number of shares of common stock outstanding as of the last day of the preceding calendar year or (2) 1,500,000.1,500,000. However, our board of directors may act prior to January 1 of a given year to provide that there will be no January 1 increase for such year or that the increase for such year will be a lesser number of shares. On January 1, 2020,2023, a total of 929,3941,500,000 additional shares were automatically added to the shares authorized under the 2018 Plan.
In July 2017, in connection with the merger, we assumed Nivalis’Nivalis’ Employee Stock Purchase Plan (the “ESPP”“ESPP”) and the 2015 EIP. Upon assumption of the ESPP, there were 45,211 shares available for issuance under the ESPP. As of December 31, 2019,2022, we have not activated the ESPP.
Stock options granted under our equity plans generally vest within four years and vested options are exercisable from the grant date until ten years after the date of grant. Vesting of certain employee options may be accelerated in the event of a change in control of the Company. We grant stock options to employees with exercise prices equal to the fair value of our common stock on the date of grant. The term of incentive stock options may not exceed ten years from the date of grant. We utilize newly issued shares to satisfy option exercises.
As of December 31, 2019,2022, a total of 3,753,7948,116,862 shares of common stock were authorized for issuance under our 2018 Plan, 2015 Plan and 2015 EIP.
A summary of stock option activity under our plans is presented below:
|
| | | | | | | | | | | | |
| Options Outstanding | | Weighted- average Exercise Price | | Weighted- average Remaining Contract Term (in years) | | Aggregate Intrinsic Value (in thousands) |
Outstanding at December 31, 2018 | 2,509,850 |
| | $ | 5.82 |
| |
| |
|
|
Granted | 1,156,350 |
| | $ | 6.36 |
| | | | |
Exercised | (26,987 | ) | | $ | 0.48 |
| | | | |
Forfeited/Expired | (387,069 | ) | | $ | 7.06 |
| | | | |
Outstanding at December 31, 2019 | 3,252,144 |
| | $ | 5.91 |
| | 7.67 | | $ | 2,569 |
|
Vested and expected to vest after December 31, 2019 | 3,202,144 |
| | $ | 5.89 |
| | 7.64 | | $ | 2,569 |
|
Exercisable at December 31, 2019 | 1,469,574 |
| | $ | 5.09 |
| | 6.50 | | $ | 2,121 |
|
As of December 31, 2019, there was $6.6 million of unrecognized stock-based compensation expense related to nonvested stock options that is expected to be recognized over a weighted-average period of 2.4 years. | | | | | | | | | | | | | | | | | | | | | | | |
| | Options
Outstanding | | Weighted-
average
Exercise
Price
(per share) | | Weighted-
average
Remaining
Contract
Term
(in years) | | Aggregate
Intrinsic
Value
(in thousands) |
| Outstanding at December 31, 2021 | 5,611,743 | | | $ | 7.41 | | | | | |
| Granted | 1,904,650 | | | $ | 10.95 | | | | | |
| Exercised | (226,480) | | | $ | 3.20 | | | | | |
| Forfeited/Expired | (231,773) | | | $ | 11.20 | | | | | |
| Outstanding at December 31, 2022 | 7,058,140 | | | $ | 8.37 | | | 7.12 | | $ | 9,833 | |
| Vested and expected to vest after December 31, 2022 | 6,918,140 | | | $ | 8.38 | | | 7.11 | | $ | 9,833 | |
| Exercisable at December 31, 2022 | 4,035,424 | | | $ | 6.75 | | | 5.97 | | $ | 8,865 | |
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
The aggregate intrinsic value of stock options exercised during the yearyears ended December 31, 2019, 20182022, 2021 and 20172020 was $155,000, $214,000$1.1 million, $546,000 and $104,000, respectively. The fair value of stock options vested during the years ended December 31, 2022, 2021 and 2020 was $8.6 million, $3.4 million and $4.2 million, respectively.
A summary of our RSU activity under our plans is presented below:$18,000. | | | | | | | | | | | | | | | | | | | | | | | |
| | Number of Shares | | Weighted-
Average
Grant Date Fair Value
(per share) | | Weighted-
average
Remaining
Contract
Life
(in years) | | Aggregate
Intrinsic
Value
(in thousands) |
| Non-vested at December 31, 2021 | 265,566 | | | $ | 12.00 | | | | | |
| Granted | 3,750 | | | $ | 9.00 | | | | | |
| Released | (61,227) | | | $ | 12.00 | | | | | |
| Forfeited | (29,471) | | | $ | 11.62 | | | | | |
| Non-vested at December 31, 2022 | 178,618 | | | $ | 12.00 | | | 8.90 | | $ | 1,313 | |
The aggregate intrinsic value of RSUs released during the years ended December 31, 2022, 2021 and 2020 was $527,000, $0 and $1.6 million, respectively. The fair value of RSUs vested during the years ended December 31, 2022, 2021 and 2020 was $735,000, $0 and $461,000, respectively.
We utilize newly issued shares to satisfy option exercises. The total fair valueexercises and RSU releases. As of shares vested during the years ended December 31, 2019, 20182022, there was $19.6 million of unrecognized stock-based compensation expense related to approximately 3.2 million nonvested stock options and 2017 was $3.0 million, $1.8 million and $139,000, respectively.RSU awards that are expected to be recognized over a weighted-average period of 2.5 years.
In January 2020, we issued 156,326 share of restricted stock units (“RSUs”) at a grant date fair value of $3.23 per share to certain employees in lieu of cash incentive compensation. Half of the shares underlying each RSU will vest on June 30, 2020, with the remaining half vesting on December 31, 2020, subject to each grantee’s continued employment or service to the Company on the applicable vesting dates. In addition, upon a change in control as defined in the 2018 Plan, any unvested shares underlying the RSU will immediately vest.
Stock-Based Compensation Expense
We use the Black-Scholes option pricing model to estimate the fair value of stock options at the grant date. The fair value of RSUs is equal to the closing stock price on the date of grant. The Black-Scholes option pricing model requires us to make certain estimates and assumptions, including assumptions related to the expected price volatility of our stock, the period during which the options will be outstanding, the rate of return on risk-free investments, and the expected dividend yield of our stock.
The fair values of stock options granted to employees were calculated using the following assumptions: | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Weighted-average estimated fair value at grant | $7.56 | | $8.44 | | $3.03 |
| Risk-free interest rate (1) | 1.39% - 4.04% | | 0.25% - 1.3% | | 0.38% - 1.68% |
| Expected term of options (in years) (2) | 5.27 - 6.08 | | 3.49 – 6.08 | | 5.27 – 6.90 |
| Expected stock price volatility (3) | 80% - 83% | | 79% - 83% | | 73% - 82% |
| Expected dividend yield (4) | —% | | —% | | —% |
|
| | | | | |
| Years Ended December 31, |
| 2019 | | 2018 | | 2017 |
Weighted-average estimated fair value at grant | $4.14 | | $5.43 | | $4.69 |
Risk-free interest rate (1) | 1.42% - 2.63% | | 2.27% - 3.07% | | 1.90% - 2.26% |
Expected term of options (in years) (2) | 5.27 – 6.08 | | 5.50 – 7.00 | | 5.69 – 6.32 |
Expected stock price volatility (3) | 70% - 77% | | 70% - 77% | | 72% - 83% |
Expected dividend yield (4) | —% | | —% | | —% |
| |
(1)
| The risk-free interest rate assumption was based on zero-coupon U.S. Treasury instruments that had terms consistent with the expected term of our stock option grants.
|
| |
(2)
| We used the “simplified method”(2)We used the “simplified method” for options to determine the expected term of stock options granted, to employees, since we do not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term due to the limited time our shares have been publicly traded. Under this approach, the weighted-average expected life is presumed to be the average of the vesting term and the contractual term of the option. |
| |
(3) | Volatility is a measure of the amount by which a financial variable, such as share price, has fluctuated or is expected to fluctuate during a period. We analyzed the historical stock price volatility of companies at a similar stage of development to estimate expected volatility of our stock price. |
| |
(4) | We have never declared or paid any cash dividends and do not presently plan to pay cash dividends in the foreseeable future. |
Stock-based compensation expense is classified in the Consolidated Statements of Operations and Comprehensive Income (Loss) as follows (in thousands): |
| | | | | | | | | | | |
| Years Ended December 31, |
| 2019 | | 2018 | | 2017 |
Employee: | |
| | |
| | |
|
Research and development | $ | 1,608 |
| | $ | 890 |
| | $ | 183 |
|
General and administrative | 1,359 |
| | 1,385 |
| | 588 |
|
Non-Employee: | | | |
| | |
|
Research and development | 68 |
| | 16 |
| | 52 |
|
General and administrative | 6 |
| | 18 |
| | 15 |
|
Total stock-based compensation expense | $ | 3,041 |
| | $ | 2,309 |
| | $ | 838 |
|
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
| | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Research and development | $ | 5,713 | | | $ | 3,347 | | | $ | 2,179 | |
| General and administrative | 3,935 | | | 2,893 | | | 1,961 | |
| Total stock-based compensation expense | $ | 9,648 | | | $ | 6,240 | | | $ | 4,140 | |
12. Income Taxes
14. Income TaxesOur loss before taxes is derived from domestic (United States) and foreign (Australian) sources as follows (in thousands): | | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2022 | | 2021 | | 2020 |
| Domestic | $ | (55,475) | | | $ | (48,932) | | | $ | (28,356) | |
| Foreign | 43 | | | (1,488) | | | 410 | |
| Total | $ | (55,432) | | | $ | (50,420) | | | $ | (27,946) | |
OnThe provision for income taxes is composed of the following (in thousands): | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Current: | | | | | |
| U.S. - Federal | $ | (981) | | | $ | — | | | $ | — | |
| U.S. - State | (16) | | | — | | | 6 | |
| Foreign | (1,285) | | | (129) | | | — | |
| Total current | (2,282) | | | (129) | | | 6 | |
| Deferred: | | | | | |
| U.S. - Federal | — | | | — | | | — | |
| U.S. - State | — | | | — | | | — | |
| Foreign | (48) | | | 216 | | | — | |
| Total deferred | (48) | | | 216 | | | — | |
| Total income tax benefit (expense) | $ | (2,330) | | | $ | 87 | | | $ | 6 | |
The effective tax rate of the provision for income taxes differs from the federal statutory rate as follows: | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| U.S. Statutory rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| Effect of: | | | | | |
| State taxes (net of federal benefit) | 0.1 | % | | — | % | | — | % |
| Permanent differences | — | % | | (0.4) | % | | 0.1 | % |
| Federal research and development credit | 5.0 | % | | 4.5 | % | | 2.6 | % |
| Change in valuation allowance | (28.6) | % | | (21.4) | % | | (20.7) | % |
| Global intangible low-taxed income recapture | (0.6) | % | | (1.4) | % | | — | % |
| Stock-based compensation | (1.9) | % | | (1.8) | % | | (2.8) | % |
| Foreign rate differential | — | % | | 0.2 | % | | (0.1) | % |
| Other | — | % | | (0.5) | % | | (0.1) | % |
| Voluntary disclosure net change in prior years’ tax liabilities | 0.8 | % | | — | % | | — | % |
| Effective income tax rate | (4.2) | % | | 0.2 | % | | — | % |
For the year ended December 22, 2017, H.R.1, commonly referred to as31, 2022, $1.0 million of the tax expense represents recognition of current domestic federal and state income tax liabilities, which was a result of the mandatory capitalization of research and development expenses under Internal Revenue Code (“IRC”) Section 174. As part of the Tax Cuts and Jobs Act of 2017 (TCJA) (“Tax Act”) was enacted into law, beginning with the 2022 tax year, expenses that are incurred for research and development in the United States of America. We continue to considerU.S. will be capitalized and amortized
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
over five years, and expenses that are incurred for research and experimentation outside the impactU.S. will be capitalized and amortized over 15 years.
For the year ended December 31, 2022, $1.3 million of the Base Erosiontax expense represents recognition of current tax liabilities, which were the result of a cumulative change to our foreign income tax provision for the activities of our wholly owned subsidiary, Alpine Immune Sciences Australia PTY LTD. In June 2022, Alpine Immune Sciences Australia PTY LTD’s was notified that the tax return filed under a revised transfer pricing model was accepted for the six-months period ended December 31, 2019. Following the acceptance notification, the tax provisions for each open tax year have been recalculated using the same transfer pricing methodology underlying the accepted return. Additionally, consistent with the change to our transfer pricing methodology, we recorded $48,000 of deferred tax expense resulting from a reduction to our deferred tax assets for the year ended December 31, 2022.
The $87,000 tax benefit for the year ended December 31, 2021 represents deferred tax benefits from true-ups and Anti-Abuse Tax ("BEAT"), Global Intangible Low-Taxed Income (“GILTI”), the deduction for foreign derived intangible income and other provisionsremoval of the Tax Act on an on-going basis.valuation allowance in place against our foreign deferred tax assets, partially offset by income tax expense for current year activity.
In addition to tax rate impacts attributable to our revised transfer pricing model and the mandatory capitalization of research and development expenses under IRC Section 174 for the year ended December 31, 2022, the difference between the U.S. federal statutory tax rates of 21% and our effective tax rate in all periods is primarily due to changes in our valuation allowance related to our deferred tax assets and the generation and consumption of federal research and development tax credits. We have elected to treat taxes due on future U.S. inclusions in taxable income under the GILTIglobal intangible low-taxed income (“GILTI”) provision as a current-period expense when incurred. As such, expected future GILTI inclusions have not been factored into the measurement of our deferred taxes.
Our income (loss) before taxes is derived from domestic (United States) and foreign (Australian) sources as follows: |
| | | | | | | |
| Years Ended December 31, |
| 2019 | | 2018 |
Domestic | $ | (38,234 | ) | | $ | (36,853 | ) |
Foreign | (3,618 | ) | | — |
|
Total | $ | (41,852 | ) | | $ | (36,853 | ) |
The provision for income taxes is composed of the following (in thousands): |
| | | | | | | |
| Years Ended December 31, |
| 2019 | | 2018 |
Current: | |
| | |
|
U.S. - Federal | $ | — |
| | $ | — |
|
U.S. - State | — |
| | (61 | ) |
Total current | — |
| | (61 | ) |
Deferred: | |
| | |
|
U.S. - Federal | — |
| | (305 | ) |
U.S. - State | — |
| | — |
|
Total deferred | — |
| | (305 | ) |
Total income tax benefit | $ | — |
| | $ | (366 | ) |
The effective tax rate of the provision for income taxes differs from the federal statutory rate as follows: |
| | | | | |
| Years Ended December 31, |
| 2019 | | 2018 |
U.S. Statutory rate | 21.0 | % | | 21.0 | % |
Effect of: | |
| | |
|
State taxes (net of federal benefit) | — | % | | 3.8 | % |
Permanent differences | (0.1 | )% | | — | % |
Federal research and development credit | 1.1 | % | | 4.2 | % |
Change in valuation allowance | (19.8 | )% | | (27.3 | )% |
Benefit of a lower tax rate | — | % | | (0.1 | )% |
Stock-based compensation | (0.5 | )% | | (0.6 | )% |
Foreign rate differential | 0.6 | % | | — | % |
Other | (2.3 | )% | | — | % |
Effective income tax rate | — | % | | 1.0 | % |
We recorded 0 tax expense for the year ended December 31, 2019 and a tax benefit of $366,000 for the year ended December 31, 2018 representing effective tax rates of 0.0% and 1.0% for the years ended December 31, 2019 and 2018, respectively. The difference between the U.S. federal statutory tax rates of 21% and our effective tax rate in all periods is primarily due to a full valuation allowance related to our deferred tax assets, the generation and consumption of federal R&D tax credits, as well as a refund of state taxes from a carryback of the 2018 net operating loss.
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. The following table represents the significant components of our deferred tax assets and liabilities for the periods presented (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Deferred tax assets: | |
| | |
|
Net operating loss | $ | 17,193 |
| | $ | 10,546 |
|
Research and development credits | 3,182 |
| | 1,958 |
|
Intangible asset basis | 35 |
| | 54 |
|
Lease liability | 2,400 |
| | — |
|
Deferred rent | — |
| | 21 |
|
Stock based compensation | 1,586 |
| | 1,382 |
|
Other | 1 |
| | 3 |
|
Gross deferred tax assets | 24,397 |
| | 13,964 |
|
Valuation allowance | (22,040 | ) | | (13,774 | ) |
Total deferred tax assets, net of valuation allowance | 2,357 |
| | 190 |
|
Deferred tax liabilities: | |
| | |
|
Prepaid expenses | (75 | ) | | (91 | ) |
Fixed asset basis | (185 | ) | | (99 | ) |
Right-of-use asset basis | (2,097 | ) | | — |
|
Total deferred tax liability | (2,357 | ) | | (190 | ) |
Net deferred tax assets and liabilities | $ | — |
| | $ | — |
|
| | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Deferred tax assets: | | | |
| Net operating loss | $ | 18,590 | | | $ | 22,054 | |
| | | |
| Research and development credits | 6,240 | | | 6,297 | |
| Intangible asset basis | 2 | | | 13 | |
| Lease liability | 2,315 | | | 2,476 | |
| Deferred revenue | 14,752 | | | 8,333 | |
| Stock based compensation | 2,343 | | | 1,672 | |
| Capitalized research and experimental costs | 12,681 | | | — | |
| Other | 156 | | | 217 | |
| Gross deferred tax assets | 57,079 | | | 41,062 | |
| Valuation allowance | (54,841) | | | (38,752) | |
| Total deferred tax assets, net of valuation allowance | 2,238 | | | 2,310 | |
| Deferred tax liabilities: | | | |
| Prepaid expenses | (173) | | | (147) | |
| Fixed asset basis | (182) | | | (93) | |
| Right-of-use asset basis | (1,728) | | | (1,856) | |
| | | |
| Total deferred tax liability | (2,083) | | | (2,096) | |
| Net deferred tax assets and liabilities | $ | 155 | | | $ | 214 | |
A valuation allowance is provided for deferred tax assets where the recoverability of the assets is uncertain. The determination to provide a valuation allowance is dependent upon the assessment of whether it is more likely than not that sufficient taxable income will be generated to utilize the deferred tax assets. BasedFor the year ended December 31, 2022, we determined that based on the weightan evaluation of theour sources of income and all available evidence, which includesboth positive and negative, including our historical operatinglatest forecasts and cumulative losses uncertaintyin recent years, it was more likely than not that none of future taxableour domestic deferred tax assets would be realized and therefore we continued to record a full valuation allowance. For the year ended December 31, 2022, we determined that it was more likely than not that our foreign deferred tax assets, with the exception of our foreign research and development income and the accumulated deficit,tax credit carryforward deferred tax assets, would be realized. As such, we providedhave
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
recorded a full valuation allowance against our foreign research and development income tax credit carryforward deferred tax assets. The valuation allowance increased by $8.3$16.1 million and $10.0$10.9 million during the yearyears ended December 31, 20192022 and 2018,2021, respectively.
We have net operating loss (“NOL”) carryforwards as follows (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Federal (before January 1, 2018) | $ | 11,094 |
| | $ | 11,094 |
|
Federal (after January 1, 2018) | $ | 67,500 |
| | $ | 33,417 |
|
State | $ | 6,433 |
| | $ | 16,756 |
|
Foreign | $ | 787 |
| | $ | — |
|
| | | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 | | |
| Federal (before January 1, 2018) | $ | — | | | $ | 11,094 | | | |
| Federal (after January 1, 2018) | $ | 85,012 | | | $ | 91,592 | | | |
| State | $ | 6,433 | | | $ | 6,433 | | | |
| Foreign | $ | — | | | $ | — | | | |
Federal net operating loss carryforwards created before January 1, 2018 begin to expire in 2037. Federal net operating lossNOL carryforwards created after January 1, 2018 carryforward indefinitely. State net operating losscarry forward indefinitely, and our state NOL carryforwards begin to expire in 2038. Foreign net operating losses carryforward indefinitely.
We have net research and development tax credit carryforwards as follows (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Federal | $ | 3,986 |
| | $ | 2,456 |
|
| | | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 | | |
| Federal | $ | 7,892 | | | $ | 7,880 | | | |
| Foreign | $ | 527 | | | $ | — | | | |
Federal research and development tax credit carryforwards begin to expire in 2035.2035, and our foreign research and development tax credits carry forward indefinitely.
Current tax laws impose substantial restrictions on the utilization of R&Dresearch and development credit and net operating lossNOL carryforwards in the event of an ownership change, as defined by the Internal Revenue CodeIRC Section 382 and 383. Such an event may limit our ability to utilize net operating lossesNOLs and R&Dresearch and development tax credit carryforwards. We are currently reviewing the preliminary results of a Section 382 study that was commissioned during the fourth quarter of 2022, focused on our wholly owned operating company and subsidiary, AIS Operating Co., Inc. It is likely we experienced ownership changes on June 10, 2016 and September 17, 2021, and as a result, our ability to utilize research and development tax credit and NOL carryforwards created prior to September 17, 2021 has been limited. We have incorporated the resulting limitations into our provision for income taxes for the year ended December 31, 2022 and will continue to evaluate the results throughout the first quarter of 2023. Though limited in the amount which can be utilized for the year ended December 31, 2022 and annually thereafter, the restrictions do not definitively prevent the impacted research and development tax credit carryforwards from utilization in future years and before expiration. As such, we have not reduced our research and development tax credit carryforwards as of December 31, 2022.
Under Internal Revenue CodeIRC Section 382 and 383, the 2017 merger with Nivalis Therapeutics Inc., or Nivalis, is likely considered an ownership change with respect to the potential limitation of the Nivalis federal tax credits and net operating losses.NOLs. As such, it is likely that any future utilization of Nivalis federal tax credits and net operating lossesNOLs is substantially limited. Therefore, as of December 31, 2018, all Nivalis tax credit and net operating lossNOL carryforwards have been reduced to 0.zero.
We account for uncertainty in income taxes in accordance with ASC 740.740, Income Taxes. Tax positions are evaluated in a two-step process, whereby we first determine whether it is more likely than not that a tax position will be sustained upon examination by the tax authority, including resolutions of any related appeals or litigation processes, based on technical merit. If a tax position meets the more-likely-than-not recognition threshold it is then measured to determine the amount of benefit to recognize in the financial statements. The tax position is measured as the largest amount of benefit that is greater than 50% likely of being realized upon ultimate settlement.
The following table summarizedsummarizes the activity related to unrecognized tax benefits (in thousands): |
| | | | | | | |
| December 31, |
| 2019 | | 2018 |
Unrecognized benefits – beginning of year | $ | 469 |
| | $ | 114 |
|
Gross increases (decreases) – prior year tax positions | — |
| | 59 |
|
Gross increases – current year tax positions | 306 |
| | 296 |
|
Unrecognized benefit – end of year | $ | 775 |
| | $ | 469 |
|
| | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2022 | | 2021 | | 2020 |
| Unrecognized benefits – beginning of year | $ | 1,553 | | | $ | 990 | | | $ | 775 | |
| Gross increases (decreases) – prior year tax positions | — | | | — | | | (7) | |
| Gross increases – current year tax positions | 596 | | | 563 | | | 222 | |
| Unrecognized benefit – end of year | $ | 2,149 | | | $ | 1,553 | | | $ | 990 | |
ALPINE IMMUNE SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (CONTINUED)
All of the unrecognized tax benefits as of December 31, 20192022 are accounted for as a reduction in our deferred tax assets. Due to our valuation allowance, none of the $775,000$2.1 million of unrecognized tax benefits would affect our effective tax rate, if recognized. We do not believe it is reasonably possible that our unrecognized tax benefits will significantly change in the next twelve months.
We recognize interest and penalties related to unrecognized tax benefits as income tax expense. There were 0 accrued interest or penalties related to unrecognized tax benefits for 2019 and 2018.
We do not expect any significant change in our unrecognized tax benefits during the next twelve months.
We recognize interest and penalties related to unrecognized tax benefits as income tax expense. There were no accrued interest or penalties related to unrecognized tax benefits for 2022, 2021 or 2020.
Our material income tax jurisdictions are the United States (federal), California (state), and Australia (foreign). We are subject to audit for tax years 2012 and forward for federal purposes, 20152017 and forward for California purposes, and 2019 and forward for foreign purposes.
15.13. Related Party Transactions
In January 2019,September 2021, in connection with our 2021 Securities Purchase Agreement, we sold an aggregate of 935,753issued 3,723,402 shares of common stock and issued2,340,424 prefunded warrants to purchase an aggregate of 364,943 shares of common stock for gross proceeds of approximately $5.0$57.0 million to certain of our stockholders whose beneficial ownership exceeded 5% stockholders. See Part III, Item 13 prior to the completion of this Form 10-Kthe 2021 Securities Purchases Agreement. Also in September 2021, we exchanged an aggregate of 1,200,000 shares of common stock held Frazier Life Sciences VIII, L.P. for additional information.Prefunded Warrants to purchase an aggregate of 1,200,000 shares of common stock. None of the purchasers in the July 2020 private placement was a greater than 5% holder of our outstanding capital stock prior to the July 2020 private placement. 16.
14. 401(k) Retirement Plan
We have adopted a 401(k) defined contribution retirement savings plan. All employees are eligible to participate, provided they meet the requirements of the plan. To date,Beginning in 2022, we havestarted offering employer 401(k) matching contributions at our discretion subject to annually determined limits. The related expense recognized was $287,000 for the year ended December 31, 2022. Prior to 2022, we did not matchedmatch employee contributions to the plan.
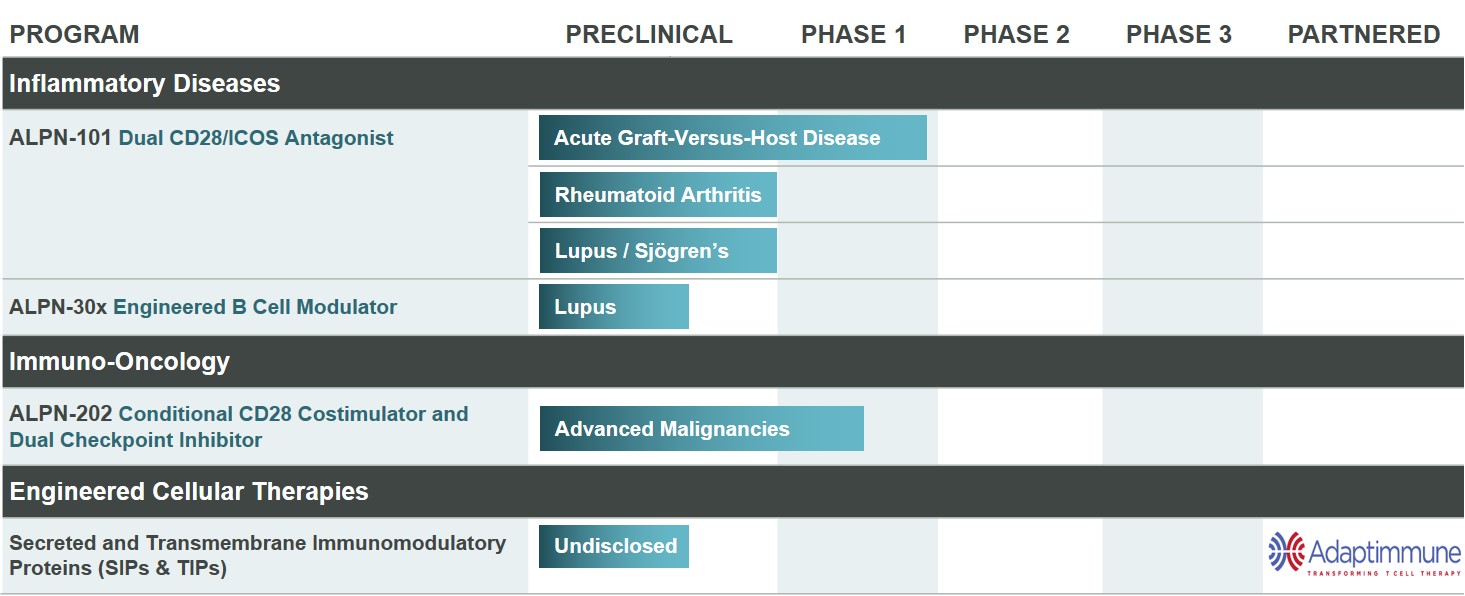
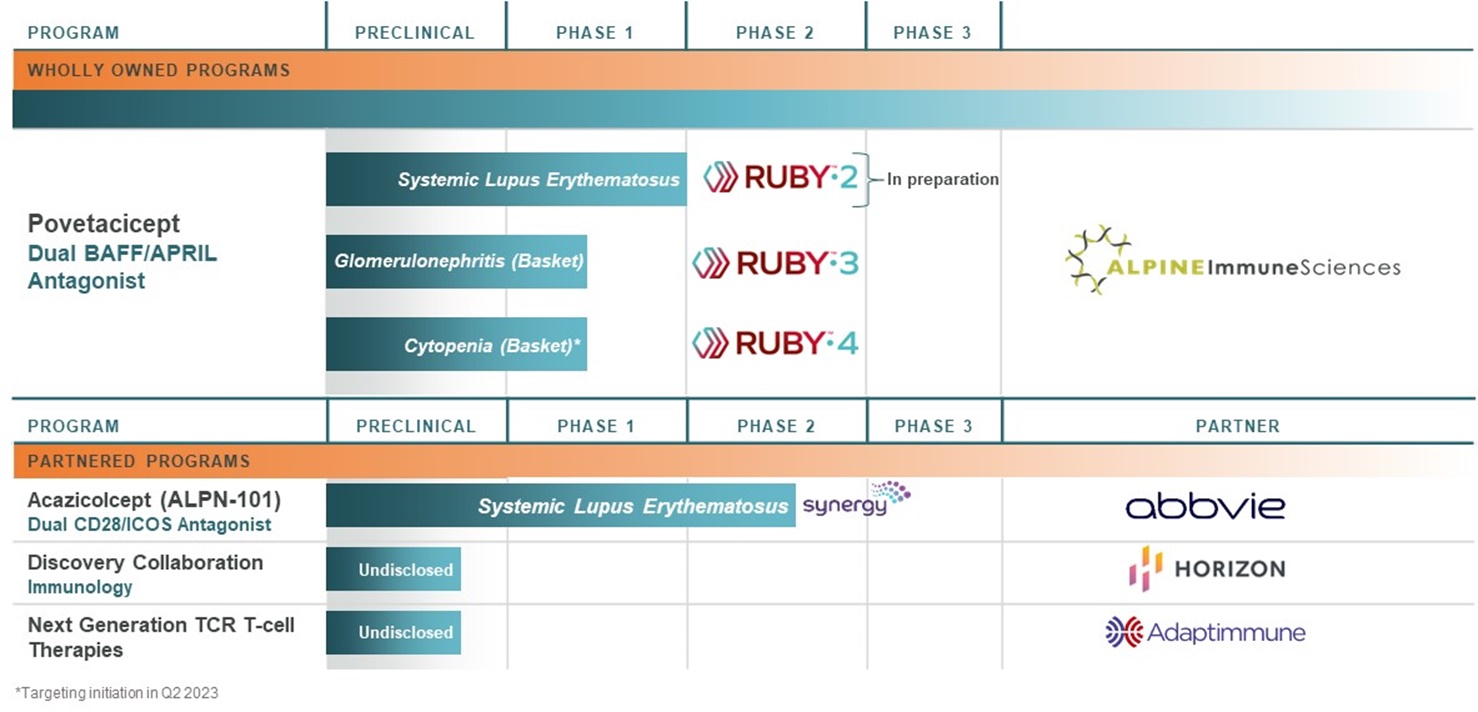
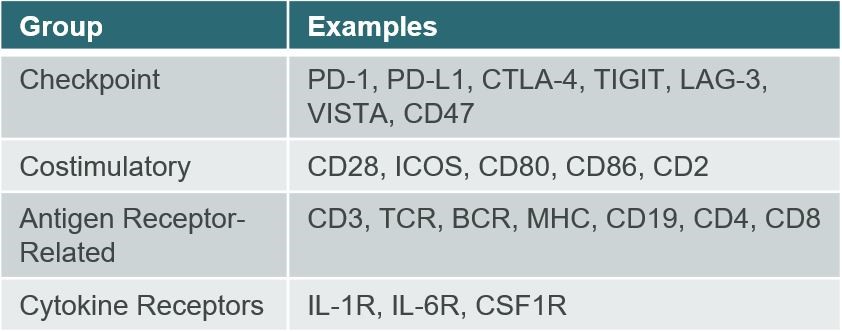
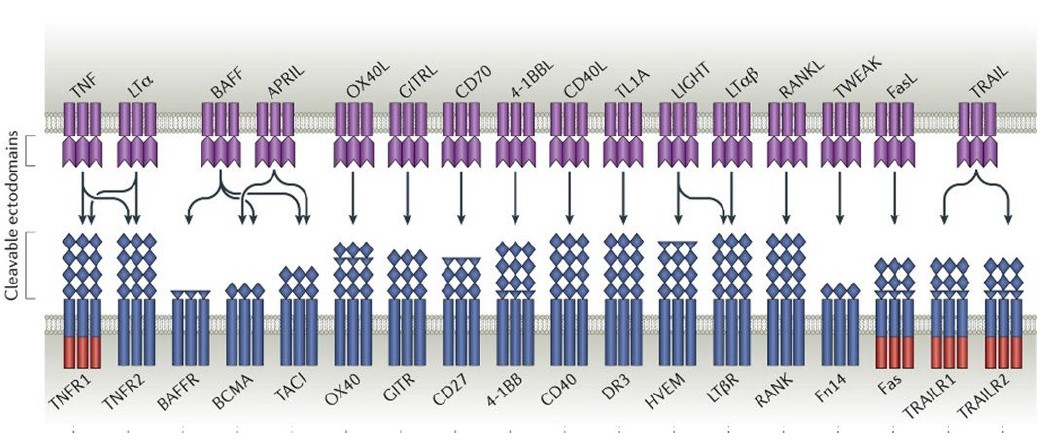

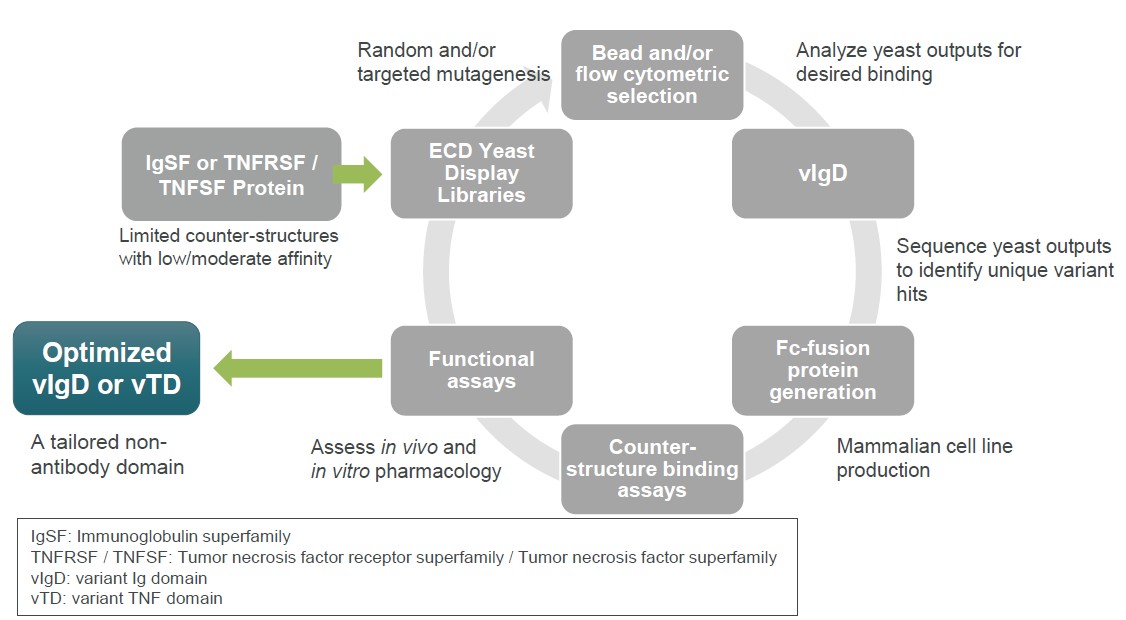
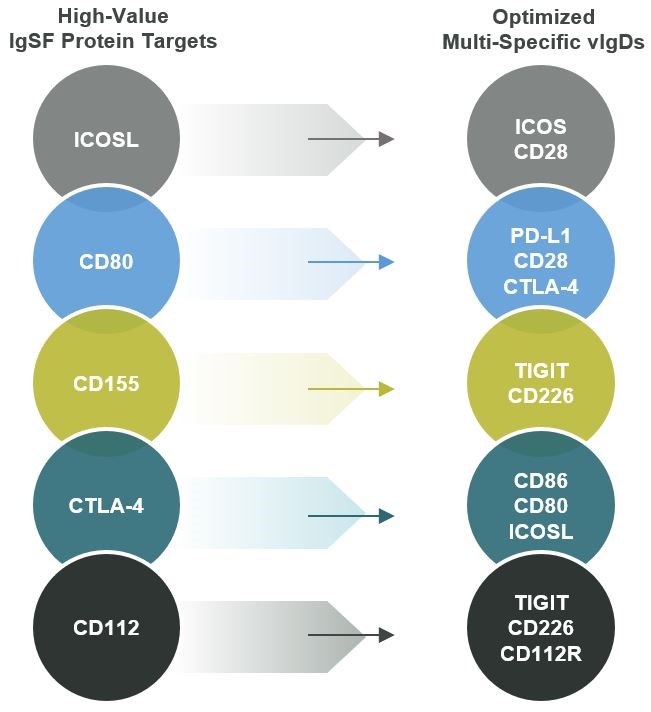
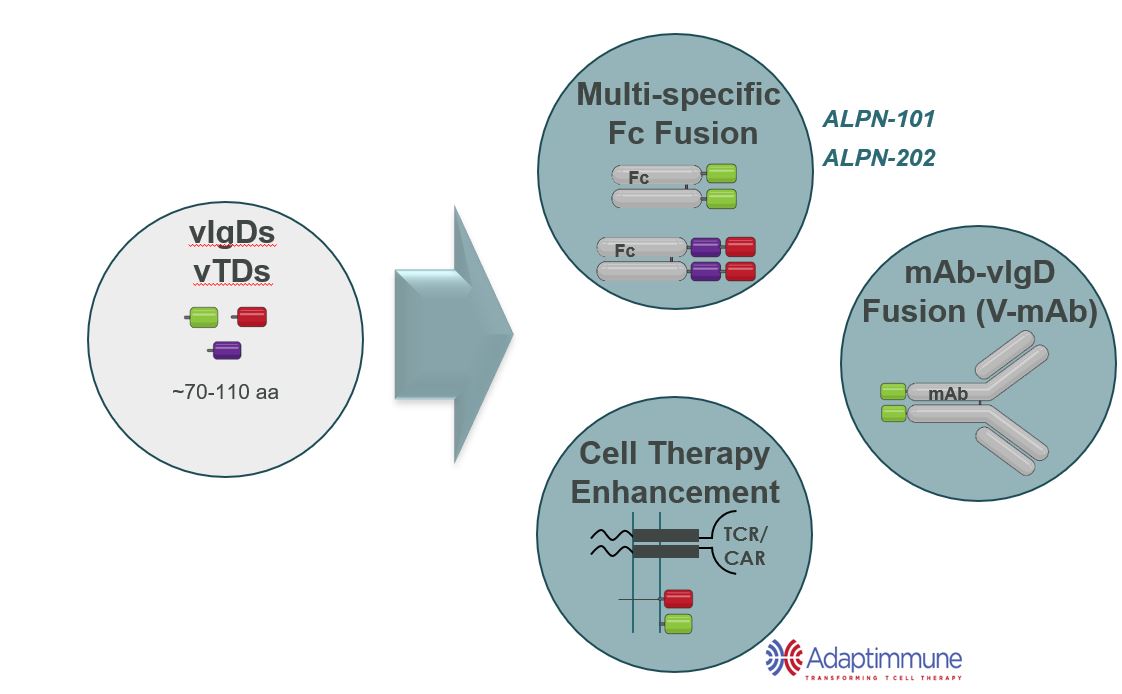
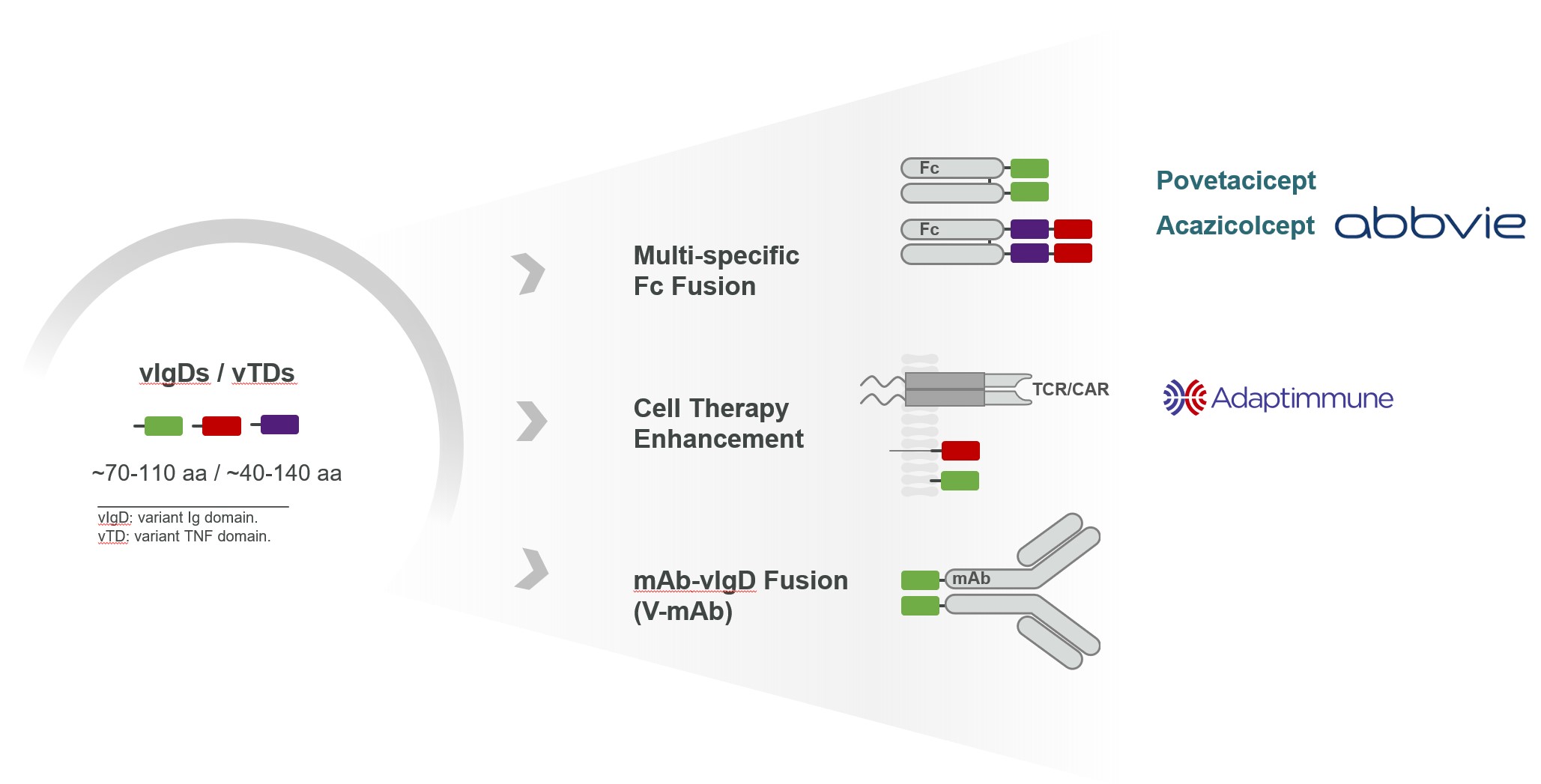
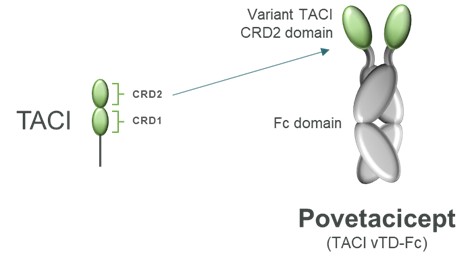
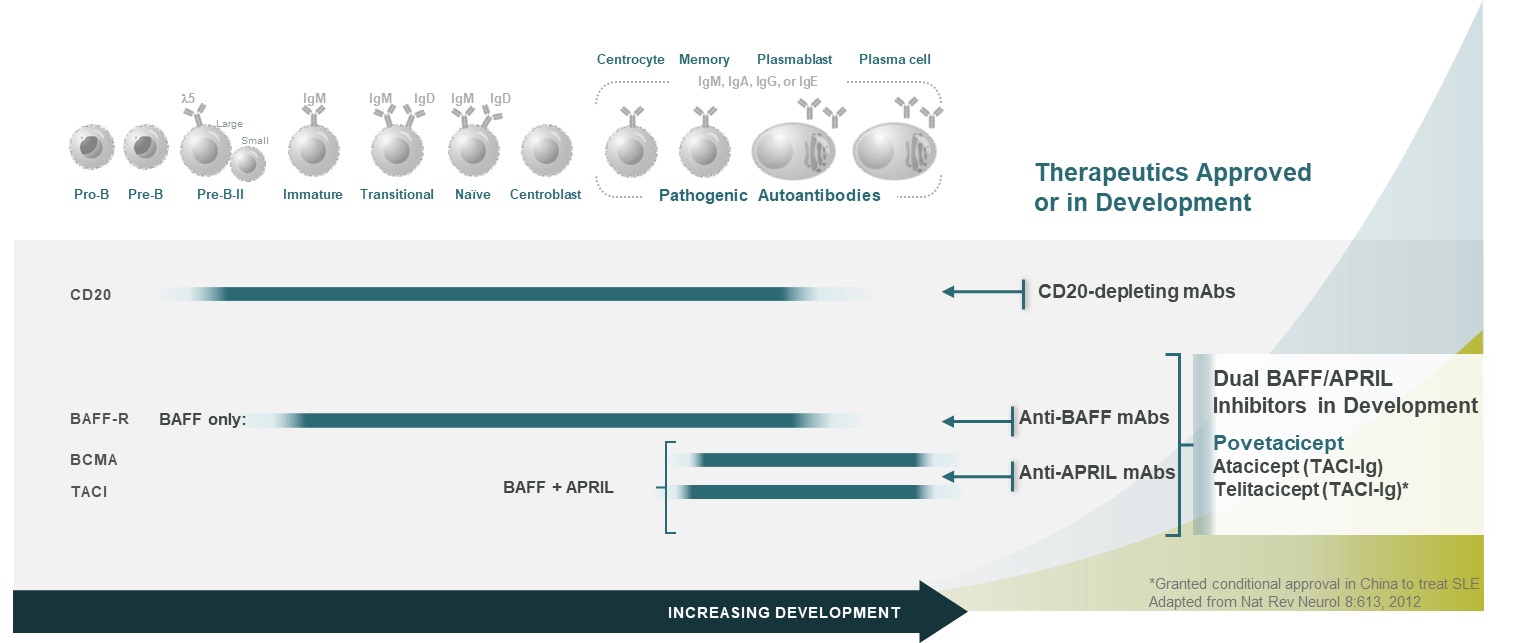
 Figure 8
Figure 8