22.Note 19. Subsequent Events
We have executed a non-binding Commitment Letter with DHCD to borrow $5.0 million, conditioned on the Company matching the $5.0 million loan with at least $5.0 million of new equity capital. We are targeting December 1, 2022 as the tentative closing date for the Loan Facility.
On April 19, 2021,October 3, 2022, we sold 318,769 shares of Common Stock under the ATM Facility and generated gross proceeds of $0.6 million. Commissions paid to the Placement Agents were insignificant. The issuance and sale of Common Stock under the ATM Facility was made pursuant to our registration statement on Form S-1 referred to in Note 1S-3 (file number 333-264299), which was declared effective by the SEC on April 26, 2022, and our prospectus supplement relating to the offering.
On October 31, 2022, we entered into securities purchase agreement with certain of our existing stockholders, including stockholders affiliated with our directors, pursuant to which we agreed to issue and sell, in a registered direct offering (the “Offering”), 10,723,926 shares of Common Stock at a sale price of $1.01 per share. The Offering was made without a placement agent, underwriter, broker or dealer and the Company is not paying underwriting discounts or commissions. The aggregate gross proceeds, before expenses, were $10.8 million. The estimated total expenses of the Offering were approximately $20,000. The Offering was made pursuant to our registration statement on Form S-3 (file number 333-253173).333-264299), which was declared effective by the SEC on April 26, 2022, and our prospectus supplement relating to the Offering.
Item 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONSManagement’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our condensed consolidated financial statements and other parts ofrelated notes included elsewhere in this Quarterly Report containon Form 10-Q. This discussion contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 (the “Securities Act”), as amended, and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). The forward-looking statements include, but are not limited to,reflecting our or management team’s expectations, hopes, beliefs, intentions, or strategies estimates and assumptions concerning events and financial trends that may affectregarding our future results of operations or financial condition.operations. In addition, any statements that refer to projections, forecasts or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking statements. The words “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “intends,” “may,” “might,” “plan,” “possible,” “potential,” “predict,” “project,” “should,” “will,” “would”“would,” and similar expressions may identify forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. TheseActual results and the timing of events may differ materially from those contained in these forward-looking statements are based on information available as of the date of this Quarterly Report and our management’s current expectations, forecasts and assumptions, and involvedue to a number of judgments, risks and uncertainties. Accordingly, forward-looking statements should not be relied upon as representingfactors, including those discussed in the section titled “Risk Factors” in our views as of any subsequent date.Annual Report on Form 10-K. We disclaim anyundertake no obligation to update forward-looking statements to reflect events or circumstances after the date they were made, whether as a result of new information, future events, or otherwise, except as specificallymay be required under applicable securities laws.
As a result of a number of known and unknown risks and uncertainties, our actual results and Unless the timing of events may differ materially from those expressed or implied by these forward-looking statements due to a number of factors, including those discussed in the section titled “Risk Factors” appearing elsewhere in this Quarterly Report.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our condensed consolidated financial statements and the related notes appearing in Part I, Item Icontext otherwise requires, for purposes of this Quarterly Report on Form 10-Qsection, the terms the “Company,” “we,” “us,” or “our” are intended to mean the business and with our Annual Report on Form 10-K for the year ended December 31, 2020 (the “2020 Annual Report”) which was filed with the SEC on March 29, 2021, pursuant to Rule 424(b)(4) under the Securities Act, as amended.operations of Clene Inc. and its consolidated subsidiaries.
Business Overview
We are a clinical-stage pharmaceutical company pioneering the discovery, development, and commercialization of novel clean-surfaced nano (CSN)nanotechnology (“CSN®”) therapeutics. CSN® therapeutics are comprised of atoms of transition elements that, when assembled in nanocrystallinenanocrystal form, possess unusually high, unique catalytic activities not present in those same elements in bulk form. These nanocatalyticcatalytic activities drive, support, and maintain beneficial metabolic and energetic intercellularcellular reactions within diseased, stressed, and damaged cells.
Our patent-protected, proprietary position affords us the potential to develop a broad and deep pipeline of novel CSN therapeutics to address a range of diseases with high impact on human health. We began in 2013 by innovating an electrochemistryelectro-crystal-chemistry drug development platform that draws from advances in nanotechnology, plasma and quantum physics, material science, and biochemistry. Our platform process results in nanocrystals with faceted structures and surfaces that are free of the chemical surface modifications that accompany other production methods. Many traditional methods of nanoparticle synthesis involve the unavoidable deposition of potentially toxic organic residues and stabilizing surfactants on the particle surfaces. Synthesizing stable nanocrystals that are both nontoxic and highly catalytic has overcome this significant hurdle in harnessing transition metal catalytic activity for human therapeutic use.
Our clean-surfaced nanocrystals exhibit catalytic activities many foldmany-fold higher than multiple other commercially available nanoparticles, produced using various techniques, that we have comparatively evaluated. We now have multiple drug assets currently in development and/or clinical trials for applications in neurology, infectious disease, and oncology. Our development and clinical efforts are currently focused on addressing the high unmet medical needs in two areas: first, those related to central nervous system disorders including Multiple Sclerosis (“MS”), Parkinson’s Disease (“PD”) and Amyotrophic Lateral Sclerosis (“ALS”ALS”), Multiple Sclerosis (“MS”), and Parkinson’s Disease (“PD”); and second, those related to the pandemic caused by COVID-19, a highly infectious viral respiratory disease with serious and sometimes fatal co-morbidities.
On December 30, 2020, Chelsea Worldwide, Inc., our predecessor company, consummated the previously announced business combination (referred to as the “Reverse Recapitalization”) pursuant to a merger agreement, dated as of September 1, 2020 (the “Merger Agreement”), by and among Clene Nanomedicine, Inc. (“Clene Nanomedicine”), Tottenham Acquisition I Limited (“Tottenham” or “TOTA”), the public entity prior to the Reverse Recapitalization, Chelsea Worldwide Inc., a Delaware corporation and wholly-owned subsidiary of Tottenham (“PubCo”), Creative Worldwide Inc., a Delaware corporation and wholly-owned subsidiary of PubCo (“Merger Sub”), and Fortis Advisors LLC, a Delaware limited liability company as the representative of our shareholders (“Shareholders’ Representative”). Prior to the Reincorporation Merger discussed below, Tottenham was incorporated in the British Virgin Islands as a blank check company for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities.
The Reverse Recapitalization was effected in two steps: (i) Tottenham was reincorporated to the state of Delaware by merging with and into PubCo (the “Reincorporation Merger”); (ii) promptly following the Reincorporation Merger, Merger Sub was merged with and into Clene Nanomedicine, resulting in Clene Nanomedicine being a wholly-owned subsidiary of PubCo (the “Acquisition Merger”). On the Closing Date, PubCo changed its name from Chelsea Worldwide Inc. to Clene Inc. and listed its shares of common stock, par value $0.0001 per share (“Common Stock”) on the Nasdaq Stock Exchange (the “Nasdaq”) under the symbol “CLNN.” As a result of the Reverse Recapitalization, Clene Nanomedicine became a wholly-owned direct subsidiary of Clene Inc. For periods prior to the closing of the Reverse Recapitalization on December 30, 2020, the disclosure in Management’s Discussion and Analysis of Financial Condition and Results of Operations has been updated to give effect to the Reverse Recapitalization.
We filed a registration statement on Form S-1 to register 4,541,481 shares of Common Stock underlying outstanding warrants that we had previously issued, which the SEC declared to be effective on April 19, 2021 (file number 333-253173). We will receive aggregate gross proceeds of $30.7 million if all of these warrants are exercised. In conjunction with the preparation of the registration statement on Form S-1, we incurred offering costs of $27 thousand, which were recognized as an expense within general and administrative expenses in the condensed consolidated statement of operations for the three months ended March 31, 2021.
We currently have no drugs approved by the US Food and Drug Administration (”FDA”) for commercial sale and have not generated any revenue from drug sales. We have never been profitable and have incurred operating losses in each year since inception. We began supplying low-dosegenerate revenue from sales of dietary supplements tothrough our wholly owned subsidiary, dOrbital, Inc., or through an exclusive license with 4Life Research LLC one of(“4Life”), a stockholder and related party. We anticipate these revenues to be small compared to our shareholders,operating expenses and had minimal directto the revenue we expect to generate from potential future sales of our rMetx™ ZnAg Immune Boost dietary supplement product. Our totaldrug candidates, for which we are currently conducting clinical trials. We incurred a loss from operations was $11.7of $9.8 million and $4.0$10.5 million for the three months ended March 31,September 30, 2022 and 2021, respectively; and 2020,$36.7 million and $35.9 million for the nine months ended September 30, 2022 and 2021, respectively. Substantially all of our losses from operations resulted from research and development expenses and administrative expenses. As of March 31, 2021September 30, 2022 and December 31, 2020,2021, we had an accumulated deficit of $193.3$192.2 million and $153.6$163.3 million, respectively.
We expect to continue investing in product development sales and marketing and customer support for our products andwe expect to incur additional losses in the future to fund our operations and conduct product research and development. We also recognize the need to raise additional capital to fully implement our business plan. The long-term continuation of our business plan is dependent upon the generation of sufficient revenues from our products to offset expenses and capital expenditures. In the event that we do not generate sufficient revenues and are unable to obtain funding, we will be forced to delay, reduce, or eliminate some or all of our research and development programs, product portfolio expansion, commercialization efforts, or capital expenditures, which could adversely affect our business prospects, ability to meet long-term liquidity needs, or we may be unable to continue operations.
26
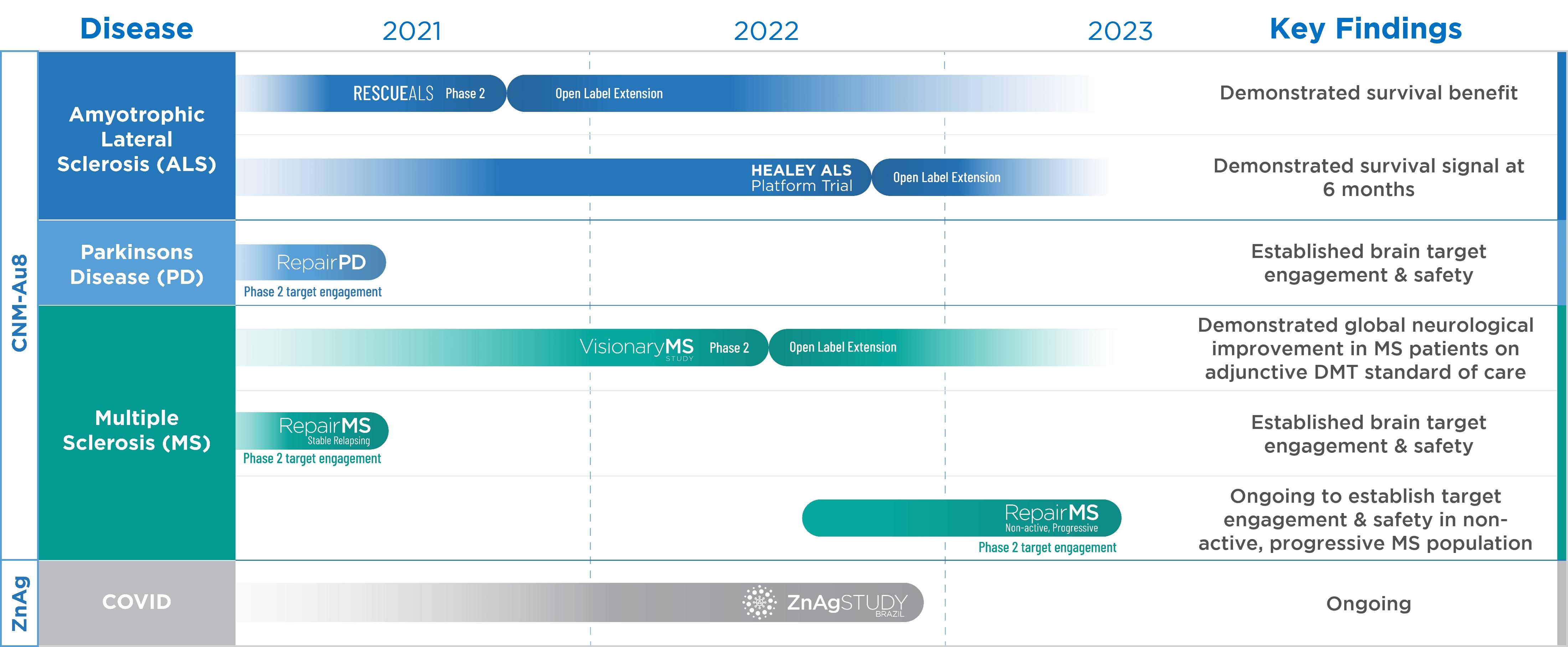
Recent Developments of Our Clinical Programs
CNM-Au8®: We recently reported topline data from the Phase 2/3 Healey ALS Platform Trial, to establish the safety and efficacy of CNM-Au8® in patients with ALS. The primary endpoint of slope of change in ALS Functional Rating Scale Revised (“ALSFRS-R”) scores adjusted for mortality was not significant (2% slowing, 95% CI: -20% to +19%) at 24 weeks. Secondary endpoints of Combined Assessment of Function and Survival (“CAFS”) and Slow Vital Capacity (“SVC”) were also not met at 24 weeks across the combined 30 mg and 60 mg CNM-Au8 doses.
The prespecified exploratory analyses of the secondary survival endpoint demonstrated a greater than 90% reduction in risk of death alone or in risk of death/permanently assisted ventilation at 24 weeks, when adjusted for baseline imbalances in risk (p=0.028 to p=0.075, unadjusted for multiple comparisons) with the CNM-Au8 30 mg dose. These survival results were statistically consistent for the 30 mg dose between the regimen only and full analysis sets, which included shared placebo from other regimens participating in the Healey ALS Platform Trial (Regimens A, B, and D). This survival signal is consistent with results previously reported by us in the Phase 2 RESCUE-ALS clinical trial with CNM-Au8.
The full analyses, including data on a subject level basis and exploratory efficacy parameters, are expected to be received from the Sean M. Healey & AMG Center for ALS at Massachusetts General Hospital (the “Healey Center”) by the end of 2022 and we expect to announce the results in the first quarter of 2023. Additionally, we expect data on biomarkers of neurodegeneration in the first quarter of 2023. The open-label extension will continue to follow participants for an additional 52-week treatment period and we expect matured survival data in the second quarter of 2023. We are in discussions with the Healey Center to offer a broader Expanded Access Program of CNM-Au8 30 mg for eligible participants of closed regimens and others.
Based on these topline findings, we have selected the CNM-Au8 30 mg dose for continued development in ALS. The CNM-Au8 60 mg dose did not demonstrate a significant survival benefit. CNM-Au8 was well tolerated, and there were no drug-related serious adverse events or significant safety findings reported. We are presently discussing the design of an international Phase 3 study with expert ALS clinical advisors with the 30 mg dose, RESTORE-ALS.
We recently presented updated interim data from the RESCUE-ALS clinical trial long-term open-label extension at the American Association of Neuromuscular & Electrodiagnostic Medicine (“AANEM”) Annual Meeting. The updated interim data demonstrated treatment with CNM-Au8 significantly improved long-term survival versus original placebo randomization, and compared to the European Network to Cure ALS (“ENCALS”) predicted median survival.
We plan to work closely with regulatory health authorities from the U.S. Food and Drug Administration (“FDA”) and European Medicines Agency, ALS experts, and patient representatives to determine the proper path to support potential approval. We do not know when or if we will be able to file a New Drug Application (“NDA”) with the FDA based on our accumulation of clinical evidence until we meet with the FDA in an end of Phase 2 meeting which is expected in mid-2023 after we receive the biomarker data and efficacy parameters that is forthcoming from the Healey ALS Platform Trial. We have paused our commercial expansion project at our Elkton, Maryland facility until we receive further clarity from the FDA on the path forward for CNM-Au8. The expansion of our North East, Maryland facility is on schedule; the North East, Maryland facility can meet current and future clinical development demand.
We recently reported positive topline data from our Phase 2 VISIONARY-MS clinical trial which evaluated the efficacy and safety of CNM-Au8 in stable relapsing remitting MS patients. The trial was stopped prematurely due to COVID-19 pandemic operational challenges, limiting enrollment to 73 out of the 150 planned participants. Due to the limited enrollment, the threshold for significance was pre-specified at p=0.10 prior to database lock. The primary analysis was conducted in a modified intent to treat (“mITT”) population, which censored invalid data. The mITT population excluded data from a single site (n=9) with Low Contrast Letter Acuity (“LCLA”) testing execution errors and the timed 25-foot walk data from one subject with a change in mobility assist device. The ITT results were directionally consistent with the mITT results, although the ITT results were not significant. The trial met the primary endpoint of change from baseline in LCLA at 48 weeks compared to placebo. The trial also met the secondary endpoints of mean standardized change from baseline in the modified MS Functional Composite (“mMSFC”) and mMSFC average rank score.
The primary and secondary results from baseline to week 48 were:
27
Consistent improvements favoring CNM-Au8 were observed across multiple paraclinical biomarkers, including multifocal visual evoked potentials amplitude and latency, optical coherence tomography, and MRI endpoints, including magnetization transfer ratio and diffusion tensor imaging metrics. Placebo treated patients, in contrast, generally worsened as expected across these measures during the 48-week period. These data provide independently assessed quantitative physiological evidence that supports the potential neuroprotective and remyelinating effects of CNM-Au8. CNM-Au8 was well-tolerated, and there were no significant safety findings reported. The open-label extension of VISIONARY-MS is ongoing.
We also completed the first dosing cohort of REPAIR-MS, an open-label, investigator blinded Phase 2 clinical trial, and have initiated a second dosing cohort in non-active progressive MS patients which is expected to be complete in the second half of 2023.
CNM-ZnAg: We have one Phase 2 clinical trial that recently concluded the blinded treatment period. The objective of this study is to investigate the efficacy and safety of CNM-ZnAg for the treatment of COVID-19. As pre-specified in the protocol, due to insufficient hospitalization events in the randomized study population, the primary and secondary endpoints were interchanged. The primary endpoint is now time to substantial alleviation of COVID-19 symptoms up to 28-days, over a continuous period greater than or equal to 48 hours. The key secondary endpoints include (i) time to complete alleviation of COVID-19 symptoms up to 28-days, over a continuous period greater than or equal to 48 hours; and (ii) the proportion of participants who are hospitalized, require hospitalization, or are deceased from baseline to day 28 (the original primary endpoint). Topline results are anticipated in the fourth quarter of 2022.
The chart below reflects the growing body of evidence for CSN therapeutics from our completed and ongoing clinical programs.
Recent Competition Update
Despite the great need for an effective disease-modifying treatment for ALS and significant research efforts by the pharmaceutical industry to meet this need, there have been limited clinical successes and no curative therapies approved to date. In May 2022, the FDA approved an orally administered version of edaravone, which has been available since 2017 as an intravenous infusion for the treatment of ALS. In July 2022, the FDA accepted an NDA for tofersen, an investigational drug from Biogen Inc., for the treatment of superoxide dismutase 1 ALS. The NDA has been granted priority review with a Prescription Drug User Fee Act goal date of April 25, 2023. Additionally, in September 2022, the FDA approved AMX0035, now branded as Relyvrio, a drug from Amylyx Pharmaceuticals, Inc. for the treatment of ALS. AMX0035 previously received a conditional approval by Health Canada in June 2022. In September 2022, Biohaven Pharmaceutical Holding Company Ltd. announced its drug candidate, verdiperstat, did not demonstrate efficacy for the treatment of ALS in the Healey ALS Platform Trial.
Impact of the COVID-19 Coronavirus Pandemic
The COVID-19 pandemic, which began in December 2019 and has spread worldwide, has caused many governments to implement measures to slow the spread of the COVID-19 outbreak. The outbreakCOVID-19 pandemic and government measures taken in response have
28
had a significant impact, both direct and indirect, on businesses and commerce, as worker shortages have occurred, supply chains have been disrupted, and facilities and production have been suspended. The future progression of the COVID-19 pandemic and its effects on our business and operations remain uncertain. The COVID-19 pandemic may affect our ability to initiate and complete preclinical studies and clinical trials, delay the initiation of future clinical trials, disrupt regulatory activities, or have other adverse effects on our business and operations. In particular, we and our clinicalthird-party contract research organizations (“CROs”CROs”) may facehave faced disruptions that may affecthave affected our ability to initiate and complete preclinical studies, causecaused manufacturing disruptions, or createand created delays at clinical trial sites.site initiation and clinical trial enrollment, leading to the early conclusion of an ongoing clinical trial. The COVID-19 pandemic has already caused significant disruptions in the financial markets, and may continue to cause such disruptions, which could impact our ability to raise additional funds to support our operations. Moreover, the COVID-19 pandemic has significantly impacted economies worldwide and could result in adverse effects on our business and operations.
We are monitoring the potential impact of the COVID-19 pandemic on our business, financial condition, results of operations, and financial statements.cash flows. While the COVID-19 pandemic has led to various research restrictions and pausedled to pauses and early conclusion of certain of our clinical trials, these impacts have been temporary and to date we have not experienced material business disruptions or incurred impairment losses in the carrying values of our assets as a result of the COVID-19 pandemic. We are not aware of any specific related event or circumstance that would require us to revise the estimates reflected in our condensed consolidated financial statements. The extent to which the COVID-19 pandemic will directly or indirectly impact our business, financial condition, results of operations, and financial condition,cash flows, including planned and future clinical trials and research and development costs, will depend on future developments that are highly uncertain, including as a result of new information that may emerge concerning COVID-19, the actions taken to contain or treat it, and the duration and intensity of the related effects.
Reverse Recapitalization with Tottenham and Clene Nanomedicine
On December 30, 2020 we completed(the “Closing Date”), Chelsea Worldwide Inc., our predecessor, consummated a business combination (the “Reverse Recapitalization”) by and among Clene Nanomedicine, Inc. (“Clene Nanomedicine”), Tottenham Acquisition I Limited (“Tottenham”), Chelsea Worldwide Inc. (“PubCo”), a Delaware corporation and wholly-owned subsidiary of Tottenham, Creative Worldwide Inc. (“Merger Sub”), a Delaware corporation and wholly-owned subsidiary of PubCo, and Fortis Advisors LLC, a Delaware limited liability company as the previously announced Reverse Recapitalization (see “Business Overview” above).
At the closingrepresentative of our stockholders. Prior to the Reverse Recapitalization, Clene Inc. acquired 100%Tottenham was incorporated in the British Virgin Islands as a blank check company for the purpose of the issued and outstanding Clene Nanomedicine commonentering into a merger, share exchange, asset acquisition, stock in exchange for 54,339,012 shares of Clene Inc. Common Stock issuedpurchase, recapitalization, reorganization, or other similar business combination with one or more businesses or entities. Prior to the Clene Nanomedicine common shareholders, of which 2,716,958 shares of the Clene Inc. Common Stock are to be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement.
At the closing of the Reverse Recapitalization, each stock optionthere was not a public market for the shares of Clene Nanomedicine common stock was cancelled and the holders thereof in exchange received 0.1320 newly-issued stock options of our Common Stock, which is 95% of the exchange ratio determined in the Merger Agreement. Pursuant to the Merger Agreement, we issued 370,101 of restricted stock units (“RSUs”) to the option holders which complements the 5% closing payment shares held in escrow for Clene Nanomedicine common shareholders discussed above. In addition, we issued 1,136,961 RSUs to option holders to complement the earn-out payments that would contingently be issued to certain current Clene Nanomedicine’s shareholders upon the achievement of milestones. See “Earn-out Shares” for the milestones detail. stock.
Immediately after giving effect to the Reverse Recapitalization and the PIPE offering discussed in below, there were 59,526,171 shares of Common Stock issued and outstanding and warrants to purchase 5,566,363 shares of Common Stock issued and outstanding.
The transaction was accounted for as a “reverse recapitalization” and Tottenham was treated as the “acquired” company for accounting purposes. Accordingly, for accounting purposes, the Reverse Recapitalization was treated aseffected in two steps: (i) Tottenham was reincorporated to the equivalentstate of Delaware by merging with and into PubCo; and (ii) Merger Sub was merged with and into Clene Nanomedicine, issuing shares forresulting in Clene Nanomedicine becoming a wholly-owned subsidiary of PubCo. On the net assets of Tottenham, accompanied by a recapitalization. The net assets of Tottenham were recorded at historical costs, with no goodwill or other intangible assets recorded. Reported amountsClosing Date, PubCo changed its name from operations included herein priorChelsea Worldwide Inc. to the Reverse Recapitalization are those of Clene Nanomedicine.
The PIPE Offering
Prior to the completion of the Reverse Recapitalization on December 30, 2020, we entered into subscription agreements on December 28, 2020, with various investors (the “PIPE”). Pursuant to the subscription agreements, we issued 2,239,500Inc. and listed its shares of our Common Stock (the “PIPE Shares”) at a price of $10.00common stock, par value $0.0001 per share with net proceeds of $22.2 million. The purpose of(“Common Stock”) on the PIPE is to fund general corporate expenses. In addition, investors inNasdaq Capital Market (“Nasdaq”) under the PIPE offering will also receive warrants to purchase a number of shares equal to one-half (1/2) of the number of PIPEsymbol “CLNN.”
Earn-Out Shares for an aggregate total of 1,119,750 shares of our Common Stock, at an exercise price of $0.01 per share (the “PIPE Warrants”), subject to a 180-day holding period.
Key Factors Affecting Our Results of Operations
Our results of operations, financial condition and the period-to-period comparability of our financial results are principally affected by the following factors:
Earn-out Shares
In connection with the Reverse Recapitalization, certain of Clene Nanomedicine’s current shareholderscommon stockholders are entitled to receive earn-out payments (the “Clene Nanomedicine Contingent Earn-out”), and Tottenham’s former officers and directors and the SponsorNorwich Investment Limited (collectively, the “Initial Shareholders”“Initial Stockholders”) are entitled to receive earn-out payments (the ““Initial Stockholders Contingent Earn-outsEarn-out,” and both collectively the “Contingent Earn-outs”) based on achieving milestones discussed below.certain milestones. The Contingent Earn-outs have been classified as liabilities in the condensed consolidated balance sheets and were initially measured at fair value on the date of the Reverse Recapitalization and will beare subsequently remeasured to fair value at each reporting date. The change in fair value of the Contingent Earn-outs has been recorded in the condensed consolidated statements of operations and comprehensive lossincome (loss) for the three and nine months ended March 31,September 30, 2022 and 2021.
Financial Overview
The Contingent Earn-out provision for Clene Nanomedicine’s common shareholders (the “Clene Nanomedicine Contingent Earn-out”) includes (i) Milestone 1 that is based on achieving a certain volume-weighted average priceOur financial condition, results of operations, and the sharesperiod-to-period comparability of our Common Stock within three years afterfinancial results are principally affected by the closing of the Reverse Recapitalization or the change of control price equaling or exceeding a certain price if a change of control transaction occurs within the three years following the closing of the Reverse Recapitalization, (ii) Milestone 2 that is based on achieving a certain volume-weighted average price of the shares of our Common Stock within five years after the closing of the Reverse Recapitalization or the change of control price equaling or exceeding a certain price if a change of control transaction occurs within the five years following the closing of the Reverse Recapitalization, and (iii) Milestone 3 that is based on completing by December 30, 2021 a randomized placebo-controlled study for treatment of COVID-19 coronavirus.factors:
The Contingent Earn-out provision for the Initial Shareholders (the “Initial Shareholders Contingent Earn-out”) includes Milestone 1 and Milestone 2 listed above. Upon the consummation of the Reverse Recapitalization, Clene Nanomedicine and the Initial Shareholders are entitled to receive up to 8,346,185 and 750,000 shares of our Common Stock, respectively.
The estimated fair values of the contingent consideration were determined using Monte Carlo simulations that simulated the future path of our Common Stock price over the earn-out periods. The assumptions utilized in the calculations are based on the achievement of certain stock price milestones including projected stock price, volatility, and risk-free rate. For potential payments related to a product development milestone, the fair value was determined based on our expectations of achieving such a milestone and the simulated estimated stock price on the expected date of achievement.
Contingent Earn-out payments involve certain assumptions requiring significant judgment and actual results may differ from assumed and estimated amounts.
Research and Development ExpensesExpense
The discovery and development of novel drug candidates require a significant investment of resources over a prolonged period of time, and a core part of our strategy is to continue making sustained investments in this area. As a result of this commitment, our pipeline of drug candidates has been advancing and expanding, with two clinical-stage drug candidates currently being investigated.
29
We anticipate thatHistorically, substantially all of our research and development expenses will increase significantly duerelate to the increase in clinical trial expenses incurred to developCNM-Au8, our drug candidates, expenses incurred for payments to CROs, principal investigators and clinical trial sites, costs of materials to support our clinical trials and preclinical studies, costs associated with preclinical activities, share awards granted to our research and development personnel and salaries for our expanding research and development personnel headcount.lead asset. Our research and development expenses are affected by the timing and advancement of our existing product pipeline as well as the timing and quantity of new drug programs commenced.
Funding for Our Operations
Since our inception, we have dedicated substantially all of our resources to the development of our drug candidates. We have financed our operations principally through proceeds from the issuance of preferred stock, issuance of common stock upon exercise of common stock options, convertible promissory notes, issuances of notes payable, and the consummation of the Reverse Recapitalization.
Since our inception and through the date of this Quarterly Report, we have funded our operations primarily with proceeds from the following sources:
We have also been awarded grants from various other organizations, including the U.S. Congressionally Directed Medical Research Program administered by the Department of Defense, the National Multiple Sclerosis Society, and FightMND, a not-for-profit registered charity in Australia, who together have issued us grants totaling approximately $2.9 million. We also receive indirect financial support for one of the clinical studies in which we participate, the Healey ALS Platform Trial, administered by the Massachusetts General Hospital, which is conducting a study of our CNM-Au8 drug candidate along with other drugs in a platform trial, at significantly lower costs to us than we would otherwise incur if we were to conduct a comparably designed study on our own at reasonable market rates.
The net cash used in our operating activities was $9.2 million and $3.3 million for the three months ended March 31, 2021 and 2020, respectively. As of March 31, 2021, we had cash of $48.0 million. We expect that the cash on hand as of March 31, 2021 will be sufficient to fund our operations for a period extending beyond twelve months from the date the condensed consolidated financial statements are issued. We have based this estimate on assumptions that may prove to be wrong, and we may exhaust our available capital resources sooner than we anticipate. See “— Liquidity and Capital Resources.” We expect our expenses to increase significantly in connection with our ongoing activities, particularly as we advance the clinical development of our clinical-stage drug products and continue research and development of our preclinical drug products and initiate additional clinical trials of, and seek regulatory approval for, these and other future drug products. As we continue to grow and expand, we will incur more expenses relating to regulatory compliance and sales and marketing personnel as we prepare to commence commercialization once we obtain regulatory approval of our drug products.
General and Administrative Expenses
Our general and administrative expenses consist primarily of staff costs, agency and consulting fees, utilities, rent and general office expenses, share grants, and RSUs grants. We anticipate that our general and administrative expenses will increase in future periods to support increases in our research and development activities and as we continue to rapidly advance the clinical programs of our drug products and expect to commercialize our products once we receive regulatory approval. These increases will likely include increased headcount, increased share compensation charges, expanded infrastructure and increased insurance expenses. We also anticipate increasing legal, compliance, accounting and investor and public relations expenses associated with being a public company.
Grants and Government Tax Incentives
We received grants issued by non-government entities related to income which have future related costs expected to be incurred and require us to comply with conditions attached to the grants. These non-government grants related to income are recognized in profit or loss as an offset to research and development expenses when funding has been received and related costs have been incurred. We received tax incentives from the Australian government in the form of cash subsidies for research and development activities related to clinical trial activities conducted by our Australian subsidiary, which are recognized as other income upon compliance with certain conditions. We did not recognize grant funding against research and development expenses for the three months ended March 31, 2021. We recognized $0.2 million of grant funding against research and development expenses for the three months ended March 31, 2020. We recognized $0.3 million of other income for the three months ended March 31, 2021 that we classified as Australia research and development credit. We did not recognize other income for the three months ended March 31, 2020.
Commercialization of Our Drug Candidates
Our business and results of operations depend on our ability to commercialize our drug candidates, if approved for marketing. Our pipeline is comprised of four drug candidates ranging from pre-clinical to late-stage clinical programs, including two drug candidates at the clinical stage or IND stage. Although we currently do not have any drug candidates approved for commercial sale and have not generated any revenue from drug product sales, we expect to commercialize one or more of our drug products in the coming years as they move toward the final stages of development. While we began selling our ZnAg Immune Boost product online in May 2020, we anticipate revenue generated from sales of this dietary supplement will be small compared to our operating expenses as well as the revenue we expect to generate from future sales of our drug candidates for which we are currently conducting clinical trials.
Components of Results of Operations
Comparison of the three months ended March 31, 2021 and 2020
The following table summarizes our results of operations for the three months ended March 31, 2021 and 2020:
| Three Months ended | ||||||||
| March 31, | ||||||||
| 2021 | 2020 | |||||||
| (in thousands) | ||||||||
| Product revenue | $ | 199 | $ | 70 | ||||
| Royalty revenue | 14 | - | ||||||
| Total revenue | 213 | 70 | ||||||
| Operating expenses: | ||||||||
| Cost of revenue | 243 | 58 | ||||||
| Research and development | 6,275 | 3,202 | ||||||
| General and administrative | 5,390 | 812 | ||||||
| Total operating expenses | 11,908 | 4,072 | ||||||
| Loss from operations | (11,695 | ) | (4,002 | ) | ||||
| Other income (expenses): | ||||||||
| Interest expense | (551 | ) | (51 | ) | ||||
| Gain on extinguishment of debt | 647 | - | ||||||
| Change in fair value of preferred stock warrant liability | - | 112 | ||||||
| Change in fair value of derivative liability | - | 4 | ||||||
| Change in fair value of Clene Nanomedicine contingent earn-out | (25,610 | ) | - | |||||
| Change in fair value of Initial Shareholders contingent earn-out | (2,961 | ) | - | |||||
| Australia research and development credit | 339 | - | ||||||
| Other income, net | 3 | (4 | ) | |||||
| Total other income (expense), net | (28,133 | ) | 61 | |||||
| Net loss before income taxes | (39,828 | ) | (3,941 | ) | ||||
| Income tax benefit | 72 | - | ||||||
| Net loss | (39,756 | ) | (3,941 | ) | ||||
| Other comprehensive income (loss): | ||||||||
| Foreign currency translation adjustments | 24 | 6 | ||||||
| Total other comprehensive income (loss) | 24 | 6 | ||||||
| Comprehensive loss | $ | (39,732 | ) | $ | (3,935 | ) | ||
Revenue
We generated revenue of $0.2 million and $70 thousand for the three months ended March 31, 2021 and 2020, respectively, which we separate as product revenue and royalty revenue. Product revenue of $0.2 million and $70 thousand was recognized in our dietary supplement segment under a supply agreement with 4Life Research, LLC, a related party, for KHC46 and a low dose zinc-silver solution, two dietary (mineral) supplements that we began supplying during those periods. We also generated minimal product revenue from sales of rMetx™ ZnAg Immune Boost during those periods. In addition, $14 thousand of our revenue during the three months ended March 31, 2021 was paid to us by 4Life Research, LLC under an exclusive and royalty-bearing license agreement relating to sales of KHC46. We did not generate royalty revenue during the three months ended March 31, 2020. For more details on the license agreement, see Note 20 to our condensed consolidated financial statements included in Part I, Item 1 of this Quarterly Report on Form 10-Q.
Operating Expenses
Cost of Sales
We incurred cost of sales of $0.2 million and $58 thousand for the three months ended March 31, 2021 and 2020, respectively, relating to production and distribution costs for the sales of our KHC46 and low dose zinc-silver solution dietary supplement products through supply agreements we have entered into with a related party.
Research and Development Expenses
Research and development expenses were $6.3 million and $3.2 million, representing 52.7%, and 78.6% of our total operating expenses for the three months ended March 31, 2021 and 2020, respectively. During these periods, substantially all of our research and development expenses were related to the development and clinical trials of our lead drug candidate, CNM-Au8. This increase of $3.1 million, or 96.0%, was primarily due to the progression of our drug candidates through the clinical development process, including increased enrollment into the REPAIR-PD and the REPAIR-MS studies, and calendar payments due for our participation in the Healey-ALS Platform Trial. These efforts resulted in greater associated costs and manufacturing expenses in support of these trials. Also, during the three months ended March 31, 2021, Research and Development expenses included $1.3 million of share-based compensation expense related to RSUs.
Historically, substantially all of our research and development expenses relate to CNM-Au8, our lead asset. Drug candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to per patient clinical trial site fees for larger studies,clinical trials, the costs of opening and monitoring clinical sites, CRO activity, and manufacturing expenses. We expectanticipate that our research and development expenses will increase significantly due to the increase in connection withclinical trial expenses incurred to develop our clinical development activities in the near term and in the future.drug candidates.
Research and development costs are charged to operations as incurred. Research and development costs include payroll and personnel expenses, including salaries and related benefits and stock-based compensation expense for employees engaged in research and development functions,functions; clinical trial supplies fees forand materials to support our clinical trials; payments to CROs, principal investigators, and clinical trial services,sites; costs associated with preclinical activities; consulting costs,costs; and allocated overhead, including rent, equipment, utilities, depreciation, insurance, and facilities maintenance costs. We account for nonrefundable advance payments for goods and services that will be used in future research and development activities initially as an asset and then as expenses when the goods have been received or when the service has been performed rather than when the payment is made.
Our clinical trial accrual process seeks to account for expenses resulting from obligations under contracts with CROs, consultants, and under clinical site agreements in connection with conducting clinical trials. The financial terms of these contracts are subject to negotiations, which vary from contract to contract and may result in payment flows that do not match the periods over which materials or services are provided to us under such contracts. We reflect the appropriate trial expenses in the condensed consolidated financial statements by matching the appropriate expenses with the period in which services and efforts are expended. In the event advance payments are made to a CRO, the payments will be recorded as a prepaid asset, which will be expensed over the period of time the contracted services are performed.
General and Administrative ExpensesExpense
General and administrative expenses consist primarily of employee salarypayroll and personnel expenses, including salaries and related benefits share-basedand stock-based compensation expenses,expense; professional fees for legal, consultingaccounting, tax, and audit servicesinformation technology services; fees for directors’ and officers’ insurance; expenses for business development activities and investor and public relations; utilities and facility expenses; travel expenses,expenses; rental feesfees; consulting fees; and other administrative expenses. We
Our expectation for our general and administrative expenses in future periods is contingent on the outcome of our end of Phase 2 meeting with the FDA, which is expected in mid-2023 after we receive the biomarker data and efficacy parameters that is forthcoming from the Healey ALS Platform Trial, and our discussions with regulatory health authorities, ALS experts, and patient representatives to determine the proper path to support potential approval.
If we are able to file an NDA with the FDA based on our accumulation of clinical evidence, we would expect our general and administrative expenses to increase in future periods to support increases in our drug development activities and as we build out our commercial capabilities in advance of receiving regulatory approval. This potential increase will likely include increased headcount, increased stock compensation expenses, expanded infrastructure including certain sales and marketing activities performed ahead of regulatory approval, and increased insurance expenses.
If we are not able to file an NDA based on our accumulation of clinical evidence, we would need to continue to growinvesting in clinical research activities and expand. Generalwe would expect our general and administrative expenses to decrease in future periods as we decrease commercial expansion projects, including at our Elkton, Maryland facility, and as we implement cost-saving initiatives, including potentially delaying or reducing launch plus commercialization efforts, reduction in executive compensation, a hiring freeze, and elimination of certain staff positions.
Total Other Income (Expense), Net
Total other income (expense), net, consists primarily of (i) changes in the fair value of our (a) common stock warrant liability and (b) Contingent Earn-outs, (ii) interest income and interest expense, (iii) interest income and expense resulting from changes in fair value of our notes payable, (iv) gains and losses on extinguishment of notes payable, (v) gains and losses on termination of leases, and (vi) the Australia research and development credit.
We also received grants issued by non-government entities which require us to comply with conditions attached to the grants. Income from grants is recognized in the period during which the related qualifying expenses are incurred, provided that the conditions under which the grants were $5.4provided have been met. We receive tax incentives from the Australian government in the form of cash subsidies for research and development activities related to clinical trial activities conducted by our Australian subsidiary, which are recognized as other income upon compliance with certain conditions.
30
Results of Operations
Our results of operations for the three and nine months ended September 30, 2022 and 2021 were as follows:
|
| Three Months Ended September 30, |
|
| Change |
|
| Nine Months Ended September 30, |
|
| Change |
| ||||||||||||||||||||
(in thousands) |
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
|
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
| ||||||||
Product revenue |
| $ | 130 |
|
| $ | 63 |
|
| $ | 67 |
|
|
| 106 | % |
| $ | 139 |
|
| $ | 400 |
|
| $ | (261 | ) |
|
| (65 | )% |
Royalty revenue |
|
| 44 |
|
|
| 47 |
|
|
| (3 | ) |
|
| (6 | )% |
|
| 100 |
|
|
| 124 |
|
|
| (24 | ) |
|
| (19 | )% |
Total revenue |
|
| 174 |
|
|
| 110 |
|
|
| 64 |
|
|
| 58 | % |
|
| 239 |
|
|
| 524 |
|
|
| (285 | ) |
|
| (54 | )% |
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||||
Cost of revenue |
|
| 19 |
|
|
| 14 |
|
|
| 5 |
|
|
| 36 | % |
|
| 19 |
|
|
| 812 |
|
|
| (793 | ) |
|
| (98 | )% |
Research and development |
|
| 6,403 |
|
|
| 6,146 |
|
|
| 257 |
|
|
| 4 | % |
|
| 24,149 |
|
|
| 18,893 |
|
|
| 5,256 |
|
|
| 28 | % |
General and administrative |
|
| 3,557 |
|
|
| 4,400 |
|
|
| (843 | ) |
|
| (19 | )% |
|
| 12,807 |
|
|
| 16,739 |
|
|
| (3,932 | ) |
|
| (23 | )% |
Total operating expenses |
|
| 9,979 |
|
|
| 10,560 |
|
|
| (581 | ) |
|
| (6 | )% |
|
| 36,975 |
|
|
| 36,444 |
|
|
| 531 |
|
|
| 1 | % |
Loss from operations |
|
| (9,805 | ) |
|
| (10,450 | ) |
|
| 645 |
|
|
| 6 | % |
|
| (36,736 | ) |
|
| (35,920 | ) |
|
| (816 | ) |
|
| (2 | )% |
Total other income (expense), net |
|
| (1,171 | ) |
|
| 39,325 |
|
|
| (40,496 | ) |
|
| (103 | )% |
|
| 7,872 |
|
|
| 21,544 |
|
|
| (13,672 | ) |
|
| (63 | )% |
Net income (loss) before income taxes |
|
| (10,976 | ) |
|
| 28,875 |
|
|
| (39,851 | ) |
|
| (138 | )% |
|
| (28,864 | ) |
|
| (14,376 | ) |
|
| (14,488 | ) |
|
| (101 | )% |
Income tax benefit |
|
| — |
|
|
| 69 |
|
|
| (69 | ) |
|
| (100 | )% |
|
| — |
|
|
| 213 |
|
|
| (213 | ) |
|
| (100 | )% |
Net income (loss) |
| $ | (10,976 | ) |
| $ | 28,944 |
|
| $ | (39,920 | ) |
|
| (138 | )% |
| $ | (28,864 | ) |
| $ | (14,163 | ) |
| $ | (14,701 | ) |
|
| (104 | )% |
Revenue
Product revenue totaled $0.1 million and $0.8$0.1 million for the three months ended March 31,September 30, 2022 and 2021, respectively; and 2020, respectively. This increase$0.1 million and $0.4 million for the nine months ended September 30, 2022 and 2021, respectively, in our Supplements segment related to (i) sales of $4.6an aqueous zinc-silver ion dietary (mineral) supplement sold by our wholly-owned subsidiary, dOrbital, Inc., under the trade name “rMetx™ ZnAg Immune Boost,” or under a supply agreement with 4Life under the trade name “Zinc Factor,” and (ii) sales of KHC46, an aqueous gold dietary (mineral) supplement of very low-concentration, sold under a supply agreement with 4Life under the trade name “Gold Factor.” During the three and nine months ended September 30, 2022, changes in product revenue were due to the timing of purchases of Zinc Factor and Gold Factor by 4Life under the supply agreement.
Royalty revenue totaled $44,000 and $47,000 for the three months ended September 30, 2022 and 2021, respectively; and $0.1 million or 563.8%and $0.1 million for the nine months ended September 30, 2022 and 2021, respectively, under an exclusive and royalty-bearing license agreement with 4Life relating to the sale of Gold Factor. For more details on the supply and license agreements, see Note 17 to our condensed consolidated financial statements.
Cost of Revenue
Cost of revenue totaled $19,000 and $14,000 for the three months ended September 30, 2022 and 2021, respectively; and $19,000 and $0.8 million for the nine months ended September 30, 2022 and 2021, respectively, relating to production and distribution costs for the sales of Gold Factor, Zinc Factor, and rMetx™ dietary supplements.
Research and Development Expense
Research and development expense for the three and nine months ended September 30, 2022 and 2021 was as follows:
|
| Three Months Ended September 30, |
|
| Change |
|
| Nine Months Ended September 30, |
|
| Change |
| ||||||||||||||||||||
(in thousands) |
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
|
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
| ||||||||
CNM-Au8 |
| $ | 2,083 |
|
| $ | 2,402 |
|
| $ | (319 | ) |
|
| (13 | )% |
| $ | 7,916 |
|
| $ | 6,908 |
|
| $ | 1,008 |
|
|
| 15 | % |
CNM-ZnAg |
|
| 638 |
|
|
| 153 |
|
|
| 485 |
|
|
| 317 | % |
|
| 2,531 |
|
|
| 670 |
|
|
| 1,861 |
|
|
| 278 | % |
Unallocated |
|
| 711 |
|
|
| 794 |
|
|
| (83 | ) |
|
| (10 | )% |
|
| 3,894 |
|
|
| 2,081 |
|
|
| 1,813 |
|
|
| 87 | % |
Personnel |
|
| 2,120 |
|
|
| 1,939 |
|
|
| 181 |
|
|
| 9 | % |
|
| 7,380 |
|
|
| 5,373 |
|
|
| 2,007 |
|
|
| 37 | % |
Stock-based compensation |
|
| 851 |
|
|
| 858 |
|
|
| (7 | ) |
|
| (1 | )% |
|
| 2,428 |
|
|
| 3,861 |
|
|
| (1,433 | ) |
|
| (37 | )% |
Total research and development |
| $ | 6,403 |
|
| $ | 6,146 |
|
| $ | 257 |
|
|
| 4 | % |
| $ | 24,149 |
|
| $ | 18,893 |
|
| $ | 5,256 |
|
|
| 28 | % |
The change in research and development expenses was primarily due to the following:
31
General and Administrative Expense
General and administrative expense for the three and nine months ended September 30, 2022 and 2021 was as follows:
|
| Three Months Ended September 30, |
|
| Change |
|
| Nine Months Ended September 30, |
|
| Change |
| ||||||||||||||||||||
(in thousands) |
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
|
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
| ||||||||
Directors’ and officersʼ insurance |
| $ | 849 |
|
| $ | 929 |
|
| $ | (80 | ) |
|
| (9 | )% |
| $ | 2,547 |
|
| $ | 2,832 |
|
| $ | (285 | ) |
|
| (10 | )% |
Legal |
|
| 87 |
|
|
| 389 |
|
|
| (302 | ) |
|
| (78 | )% |
|
| 414 |
|
|
| 1,034 |
|
|
| (620 | ) |
|
| (60 | )% |
Finance and accounting |
|
| 217 |
|
|
| 227 |
|
|
| (10 | ) |
|
| (4 | )% |
|
| 901 |
|
|
| 2,568 |
|
|
| (1,667 | ) |
|
| (65 | )% |
Public and investor relations |
|
| 124 |
|
|
| 264 |
|
|
| (140 | ) |
|
| (53 | )% |
|
| 593 |
|
|
| 1,141 |
|
|
| (548 | ) |
|
| (48 | )% |
Personnel |
|
| 706 |
|
|
| 787 |
|
|
| (81 | ) |
|
| (10 | )% |
|
| 3,147 |
|
|
| 2,416 |
|
|
| 731 |
|
|
| 30 | % |
Stock-based compensation |
|
| 1,247 |
|
|
| 1,567 |
|
|
| (320 | ) |
|
| (20 | )% |
|
| 4,056 |
|
|
| 6,084 |
|
|
| (2,028 | ) |
|
| (33 | )% |
Other |
|
| 327 |
|
|
| 237 |
|
|
| 90 |
|
|
| 38 | % |
|
| 1,149 |
|
|
| 664 |
|
|
| 485 |
|
|
| 73 | % |
Total general and administrative |
| $ | 3,557 |
|
| $ | 4,400 |
|
| $ | (843 | ) |
|
| (19 | )% |
| $ | 12,807 |
|
| $ | 16,739 |
|
| $ | (3,932 | ) |
|
| (23 | )% |
The change in general and administrative expense was primarily due to the following:
32
Total Other Income (Expenses)(Expense), Net
OtherTotal other income (expenses) consists of interest expenses, interest income, changes in fair value of preferred stock warrant liability, changes in fair value of derivative liability, change in fair value of contingent earn-out, a research and development credit received from the Australian government, foreign exchange gain, gain on disposal of assets, and loss on extinguishment of notes payable. Other income (expenses)(expense), net, for the three and nine months ended March 31,September 30, 2022 and 2021 was as follows:
|
| Three Months Ended September 30, |
|
| Change |
|
| Nine Months Ended September 30, |
|
| Change |
| ||||||||||||||||||||
(in thousands) |
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
|
| 2022 |
|
| 2021 |
|
| Dollars |
|
| % |
| ||||||||
Interest expense |
| $ | (857 | ) |
| $ | 80 |
|
| $ | (937 | ) |
|
| (1,171 | )% |
| $ | (2,390 | ) |
| $ | (497 | ) |
| $ | (1,893 | ) |
|
| (381 | )% |
Gain on extinguishment of notes payable |
|
| — |
|
|
| — |
|
|
| — |
|
|
| 0 | % |
|
| — |
|
|
| 647 |
|
|
| (647 | ) |
|
| (100 | )% |
Gain on termination of lease |
|
| — |
|
|
| — |
|
|
| — |
|
|
| 0 | % |
|
| 420 |
|
|
| — |
|
|
| 420 |
|
|
| 100 | % |
Change in fair value of common stock warrant liability |
|
| 149 |
|
|
| 414 |
|
|
| (265 | ) |
|
| (64 | )% |
|
| 151 |
|
|
| 547 |
|
|
| (396 | ) |
|
| (72 | )% |
Change in fair value of Clene Nanomedicine contingent earn-out liability |
|
| (1,591 | ) |
|
| 35,042 |
|
|
| (36,633 | ) |
|
| (105 | )% |
|
| 6,662 |
|
|
| 18,072 |
|
|
| (11,410 | ) |
|
| (63 | )% |
Change in fair value of Initial Stockholders contingent earn-out liability |
|
| (205 | ) |
|
| 3,439 |
|
|
| (3,644 | ) |
|
| (106 | )% |
|
| 849 |
|
|
| 1,710 |
|
|
| (861 | ) |
|
| (50 | )% |
Australia research and development credit |
|
| 1,346 |
|
|
| 364 |
|
|
| 982 |
|
|
| 270 | % |
|
| 2,001 |
|
|
| 1,078 |
|
|
| 923 |
|
|
| 86 | % |
Other income (expense), net |
|
| (13 | ) |
|
| (14 | ) |
|
| 1 |
|
|
| 7 | % |
|
| 179 |
|
|
| (13 | ) |
|
| 192 |
|
|
| 1,477 | % |
Total other income (expense), net |
| $ | (1,171 | ) |
| $ | 39,325 |
|
| $ | (40,496 | ) |
|
| (103 | )% |
| $ | 7,872 |
|
| $ | 21,544 |
|
| $ | (13,672 | ) |
|
| (63 | )% |
The change in total other income (expense), net, was primarily due to the following:
(i) recognized interest expense of $0.6 millionthree months ended September 30, 2021; and $51 thousand, respectively, due toa loss from an increase in the fair value of our notes payable. As of March 31, 2021, the fair value of our notes payable is determined based on the closing price of CLNN shares listed on the Nasdaq of $12.78 per share.
(ii) recognized gain on extinguishment of notes payable of $0.6 million, due to the forgiveness of the PPP Loan by the U.S. Small Business Administration. There was no gain on extinguishment of notes payableliabilities for the three months ended March 31, 2020.
(iii) recognized expense of $112 thousand relating to theSeptember 30, 2022. The changes in fair value of preferred stock warrant liability forwere due to changes in the three months ended March 31, 2020. There was no preferred stock warrant liability as a result of the Reverse Recapitalization on December 30, 2020. Upon the consummation of the Reverse Recapitalization, we determined that the warrants qualify for classification as permanent equity and we reclassified the resulting warrant liability to additional paid-in capital. No change in fair value of preferred stock warrant liability is recorded going forward.
(iv) recognized the change in fair valueprice of our Clene Nanomedicine contingent earn-out liability of $25.6 million forCommon Stock on Nasdaq and updates in the three months ended March 31, 2021. The change in fair value was primarily a result ofvaluation model assumptions (see “Critical Accounting Policies and Estimates”);
(v) recognized the change in fair value of our Initial Shareholders contingent earn-out liability of $3.0 million for the three months ended March 31, 2021. The change in fair value was primarily a result of the increase of the closing price of CLNN shares listed on the Nasdaq for $12.78 per share on March 31, 2021 from $9.01 per share on December 31, 2020 when we remeasured the Initial Shareholders contingent earn-out liability at December 31, 2020.
(vi) recognized income of $0.3 million relating to aAustralia research and development credit received from the Australian government for the three months ended March 31, 2021. We did not recognize income relating to research and development credit received from the Australian government for the three months ended March 31, 2020.credit. We recognized AustralianAustralia research and development credit in an amount equal to the qualifying expenses incurred in each period multiplied by the applicable reimbursement percentage. The increase in researchpercentage; and development credit is the result of increased research
Taxation
Comprehensive Loss
As a result of the foregoing, we incurred a comprehensive loss of $39.7 million and $3.9 million for the three months ended March 31, 2021 and 2020, respectively.
Taxation
United States
We are incorporated in Delaware in the U.S.state of Delaware and subject to statutory U.S. federal corporate income tax at a rate of 21% for the three and nine months ended March 31, 2021September 30, 2022 and 2020.2021. We are also subject to state income tax in Utah and Maryland, at a rate of 4.95%4.85% and 8.25%, respectively, for the three and nine months ended MarchSeptember 30, 2022 and 2021. As of September 30, 2022 and December 31, 2021, and 2020. Wewe recorded a full valuation allowance against our net deferred tax assets due to the uncertainty as to whether such assets will be realized resulting from our three-year cumulative loss position and the uncertainty surrounding our ability to generate pre-tax income in the foreseeable future.
33
Australia
Australia
Our wholly-owned subsidiary, Clene Australia Pty Ltd (“Clene Australia”), was established in Australia on March 5, 2018 and is subject to corporate income tax at a rate of 27.5%.25% for the three and nine months ended September 30, 2022 and 2021, respectively. Clene Australia total income tax benefit was $72 thousandtotaled $0 and $0.1 million for the three months ended March 31, 2021. DuringSeptember 30, 2022 and 2021, respectively; and $0 and $0.2 million for the nine months ended September 30, 2022 and 2021, respectively. We recorded other income of $1.3 million and $0.4 million for the three months ended March 31, 2020, Clene Australia had no taxable incomeSeptember 30, 2022 and therefore, no provision2021, respectively; and $2.0 million and $1.1 million for income taxes was required. We recorded $0.3 million as other income during the threenine months ended March 31,September 30, 2022 and 2021, respectively, for a refund of research and development credits pertaining to Clene Australia for the 2022 and 2021 tax year. We did not record any other income during the three months ended March 31, 2020 for a refund of research and development credits pertaining to Clene Australia for the 2020 tax year.years, respectively.
Netherlands
Netherlands
Our wholly-owned subsidiary, Clene Netherlands B.V., was established in the Netherlands on April 21, 2021 and will beis subject to corporate income tax at a rate of 15% up to €245,000€395,000 of taxable income and 25%25.8% for taxable income in excess of €245,000. As€395,000. Clene Netherlands was established subsequent to the quarter ended March 31, 2021, itB.V. had no taxable income and therefore, noor provision for income taxes was required.for the three and nine months ended September 30, 2022 and 2021.
JOBS Act
The JOBS Act permits an emerging growth company (“EGC”) such as us to take advantage of an extended transition period to comply with new or revised accounting standards applicable to public companies. We have elected to use the extended transition period under the JOBS Act until the earlier of the date we (1) are no longer an emerging growth company or (2) affirmatively and irrevocably opt out of the extended transition period provided in the JOBS Act. As a result, our condensed consolidated financial statements may not be comparable to companies that comply with new or revised accounting pronouncements as of public company effective dates.
We will remain an emerging growth company until the earliest to occur of: (1) the last day of the fiscal year in which we have more than $1.07 billion in annual revenue; (2) the date on which we are deemed to be a “large accelerated filer,” which would occur if the market value of our equity securities held by non-affiliates exceeds US$700 million as of the last business day of our most recently completed second fiscal quarter; (3) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period; and (4) the last day of the fiscal year ending after the fifth anniversary of Tottenham’s initial public offering, or August 6, 2023.
We may choose to early adopt any new or revised accounting standards whenever such early adoption is permitted for public companies.
Smaller Reporting Company Status
We are a Smaller Reporting Company (“SRC”) as defined in Item 10(f)(1) of Regulation S-K. Smaller reporting companies may take advantage of certain reduced disclosure obligations, including, among other things, providing only two years of audited financial statements. We will remain a smaller reporting company until (1) the market value of our Common Stock held by non-affiliates exceeds $250 million as of the end of the second fiscal quarter and our annual revenues exceed $100 million during the previous fiscal year, or (2) the market value of our Common Stock held by non-affiliates exceeds $700 million as of the end of the second fiscal quarter.
Liquidity and Capital Resources
Sources of Capital
Since inception, weWe have incurred annual netsignificant losses and negative cash flows from operations since our inception. We expect to incur additional losses in the future to fund our operations and conduct product research and development. We recognize the need to raise additional capital to fully implement our business plan. The long-term continuation of our business plan is dependent upon the generation of sufficient revenues from our operations. Substantiallyproducts to offset expenses and capital expenditures. In the event that we do not generate sufficient revenues and are unable to obtain funding, we will be forced to delay, reduce, or eliminate some or all of our losses have resulted from the funding of our research and development programs, and general and administrative expenses associated withproduct portfolio expansion, commercialization efforts, or capital expenditures, which could adversely affect our business prospects, ability to meet long-term liquidity needs, or we may be unable to continue operations. We incurred net losses
Since our inception, we have dedicated substantially all of $39.8 million and $3.9 million forour resources to the three months ended March 31, 2021 and 2020, respectively. Our loss from operations was $11.7 million and $4.0 million for the three months ended March 31, 2021 and 2020, respectively.development of our drug candidates. We have financed our operations principally through the following sources:
We also received indirect financial support for the Healey ALS Platform Trial, administered by Massachusetts General Hospital, which conducted a platform trial for the treatment of ALS with certain drug candidates, including CNM-Au8, at significantly lower costs than we would have otherwise incurred if allwe had conducted a comparably designed clinical trial at reasonable market rates.
Going Concern
We incurred a loss from operations of these warrants are exercised.
The net cash used in our operating activities was $9.2$9.8 million and $3.3$10.5 million for the three months ended MarchSeptember 30, 2022 and 2021, respectively; and $36.7 million and $35.9 million for the nine months ended September 30, 2022 and 2021, respectively. Our accumulated deficit was $192.2 million and $163.3 million as of September 30, 2022 and December 31, 2021, respectively. Our cash, cash equivalents, and 2020, respectively. Asmarketable securities totaled $16.2 million and $50.3 million as of MarchSeptember 30, 2022 and December 31, 2021, respectively, and net cash used in operating activities was $31.3 million and $25.0 million for the nine months ended September 30, 2022 and 2021, respectively.
We have incurred significant losses and negative cash flows from operations since our inception. We have not generated significant revenues since our inception, and we had cashdo not anticipate generating significant revenues unless we successfully complete development and obtain regulatory approval for commercialization of $48.0 million.a drug candidate. We expect thatto incur additional losses in the cash on hand as of March 31, 2021 will be sufficient to fund our operations for a period extending beyond twelve months from the date the condensed consolidated financial statements are issued. We have based this estimate on assumptions that may prove to be wrong, and we may exhaust our available capital resources sooner than we anticipate. We expect our expenses to increase significantly in connection with our ongoing activities,
34
future, particularly as we advance the clinical development of our clinical-stage drug products andcandidates, continue research and development of our preclinical drug productscandidates, and initiate additional clinical trials of, and seek regulatory approval for, these and other future drug products. Ascandidates.
Our management performs strategic reviews of our operating plans and budgets, considering the status of our product development programs, human capital, capital needs and resources, and current capital market conditions. Based on these reviews, our Board of Directors and management make adjustments to our operating plans and budgets to allocate our projected cash expenditures. Notwithstanding these ongoing adjustments, we continue to grow and expand,project that within the next twelve months, we will incur more expenses relatingnot have sufficient cash and other resources on hand to regulatory compliancesustain our current operations or meet our obligations as they become due, and saleswe must obtain additional financing. Additionally, pursuant to our term loan with Avenue Venture Opportunities Fund, L.P. (“Avenue”), we must maintain unrestricted cash and marketing personnelcash equivalents of at least $5.0 million to avoid acceleration of the full balance of the loan. These conditions raise substantial doubt about the Company’s ability to continue as a going concern.
To mitigate our funding needs, we prepareintend to commence commercialization once we obtain regulatory approvalimplement plans to raise additional funding, including exploring equity financing and offerings, debt financing, licensing or collaboration arrangements with third parties, as well as potentially utilizing additional funds available under our term loan with Avenue, subject to certain contingent conditions (see Note 9), as well as our existing at-the-market facility. These plans are subject to market conditions and reliance on third parties, and there is no assurance that effective implementation of our drug products.
Ourplans will result in the necessary funding to continue current operations. In October 2022, we announced an equity offering which provided net cash proceeds of $10.8 million. We are also in the process of implementing cost-saving initiatives, including potentially delaying or reducing research and development programs and commercialization efforts, reduction in executive compensation, a hiring freeze, and elimination of certain staff positions. We have concluded that our plans do not alleviate the substantial doubt about our ability to continue as a going concern may require obtaining additional funding to finance operations. As part of our ongoing business plans,beyond one year from the date the condensed consolidated financial statements are issued.
The accompanying condensed consolidated financial statements have been prepared assuming we will continue seeking fundingas a going concern, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. As a result, the accompanying condensed consolidated financial statements do not include any adjustments relating to the recoverability and classification of assets and their carrying amounts, or the amounts and classification of liabilities that may result should we be unable to continue as a going concern.
Short-Term Material Cash Requirements
For at least the next twelve months, our primary capital requirements are to fund our operations, including research and development, personnel, regulatory, and other clinical trial costs related to development of our lead drug candidate, CNM-Au8; and general and administrative costs to support our drug development and pre-commercial activities in advance of receiving regulatory approval for our drug candidates.
Firm commitments for funds include approximately $0.1 million and $1.0 million of payments under finance and operating lease obligations, respectively; payment of principal and interest on notes payable totaling $5.9 million; and commitments under various agreements for capital expenditures totaling $1.7 million related to the construction of our manufacturing facilities. We expect to meet our short-term liquidity requirements primarily through cash on hand. Additional sources of funds include equity financing, and may seek debt financing, or other capital sources. We may not be able to obtain financing on acceptable terms, or at all. The terms of any financing may adversely affect the holdings or the rights of our shareholders. If we are unable to raise capital when needed or on acceptable terms, we would be forced to delay, reduce or eliminate research and development programs and commercialization efforts.
The following table provides information regarding our cash flows for relevant periods:
| Three months ended | ||||||||
| March 31, | ||||||||
| 2021 | 2020 | |||||||
| (in thousands) | ||||||||
| Net cash used in operating activities | $ | (9,161 | ) | $ | (3,251 | ) | ||
| Net cash used in investing activities | (203 | ) | (23 | ) | ||||
| Net cash provided by (used in) financing activities | (1,896 | ) | 1,547 | |||||
| Net effect of foreign exchange rate changes | 26 | 55 | ||||||
| Net decrease in cash | (11,234 | ) | (1,672 | ) | ||||
Use of Funds
Our primary use of cash in all periods presented was to fund our research and development, regulatory and other clinical trial costs, and related supporting administration. Our prepaid expenses and other current assets, accounts payable and accrued expense balances in all periods presented were affected by the timing of vendor invoicing and payments, and impacted the cash provided by, or used in, operations. We have no commitments for capital expenditures as of the end of the latest fiscal period.
Operating Activities
Net cash used in operating activities was $9.2 million of cash for the three months ended March 31, 2021, which resulted from a net loss of $39.8 million, adjusted for (i) non-cash items of $32.0 million, which primarily consisted of depreciation expense of $0.2 million, stock-based compensation expenses of $3.3 million, changes in fair value of the Clene Nanomedicine contingent earn-out of $25.6 million, changes in fair value of the Initial Shareholders contingent earn-out of $3.0 million, gain on extinguishment of debt of $0.6 million and increase in interest accrued on notes payable and accretion of debt discount of $0.5 million, and (ii) a net decrease in operating assets and liabilities of $1. million. The net decrease in operating assets and liabilities was primarily attributable to an increase in inventory of $0.2 million, an increase in accounts receivable of $0.1 million, an increase in prepaid expenses and other current assets of $1.3 million due to the increase in Australia research and development credit receivable and prepayments to CROs and other vendors, $0.2 million increase in accounts payable, $0.1 million decrease in operating lease obligations, and $0.1 million increase in accrued liabilities due to the timing of vendor invoicing and payments.
Net cash used in operating activities was $3.3 million of cash for the three months ended March 31, 2020, which resulted from a net loss of $3.9 million, adjusted for (i) non-cash items of $0.3 million, which primarily consisted of depreciation expense of $0.2 million, stock-based compensation expenses of $0.2 million, and changes in the fair value of preferred stock warrant liability of $0.1 million , and (ii) a net decrease in operating assets and liabilities of $0.4 million. The net decrease in operating assets and liabilities was primarily attributable to an increase in prepaid expenses and other current assets of $0.1 million due to the increase in other vendors, $0.6 million increase in accounts payable, and $0.1 million decrease in accrued liabilities due to the timing of vendor invoicing and payments.
Investing Activities
Net cash used in investing activities was $0.2 million and $23 thousand for the three months ended March 31, 2021 and 2020, respectively, which in each instance was related to purchases of property and equipment.
Financing Activities
Net cash used in financing activities was $1.9 million for the three months ended March 31, 2021, which primarily resulted from payments of deferred offering costs of $1.9 million and payments on our finance lease obligations of $45 thousand, partially offset by proceeds from issuance of common stock upon exercise of common stock options of $50 thousand.
Net cash provided by financing activities was $1.5 million for the three months ended March 31, 2020, which primarily resulted from proceeds from the issuance of notes payable of $1.6 million partially offset by payments on our finance lease obligations of $0.1 million.
Debt Obligations
In February 2019, we entered into a loan agreement (the “2019 MD Loan”) with the Department of Housing and Community Development, a principal department of the State of Maryland (“Maryland”). Pursuant to the 2019 MD Loan, Maryland agreed to provide a $0.5 million term loan. Amounts outstanding under the 2019 MD Loan bear simple interest at an annual rate of 8.00%. Under the 2019 MD Loan, we agreed to affirmative and negative covenants to which we will remain subject until maturity. These covenants include providing information about the Company and our operations; limitations on our ability to retire, repurchase, or redeem our common or preferred stock, options, and warrants other than per the terms of the securities; and limitations on our ability to pay dividends of cash or property. There are no financial covenants associated with the Loan Agreement. Events of default under the Loan Agreement include failure to make payments when due, insolvency events, failure to comply with covenants, and material adverse effects with respect to the Company. We are not in violation of any affirmative or negative covenants. Repayment of the full balance outstanding is due on February 22, 2034. The 2019 MD Loan establishes “Phantom Shares,” based on 119,906 shares of our Common Stock (based on 863,110 Series C Preferred Shares prior to the Reverse Recapitalization), determined at issuance. The Loan Agreement states the repayment amount is to be the greater of the balance of principal and accrued interest or the Phantom Shares value. We determined that the note should be accounted for at fair value. We record the fair value of the debt at the end of each reporting period. In order to value the note, we consider the amount of the simple interest expense that would be due and the value of Phantom Shares. Upon the closing of the Reverse Recapitalization and as of December 31, 2020, the fair value of the 2019 MD Loan is determined based on the closing price of CLNN shares listed on the Nasdaq. Expense of $0.5 million and $10 thousand was recognized during the three months ended March 31, 2021 and 2020, respectively. The fair value of $1.5 million and $1.1 million of principal and accrued interest is included in long-term notes payable as of March 31, 2021 and December 31, 2020, respectively.
In April 2019, we entered into a loan agreement (the “2019 Cecil Loan”) with Cecil County, Maryland (“Cecil”). Pursuant to the 2019 Cecil Loan, Cecil agreed to provide a $0.1 million term loan. Amounts outstanding under the 2019 Cecil Loan bear simple interest at an annual rate of 8.00%. Under the 2019 Cecil Loan, we agreed to affirmative and negative covenants to which we will remain subject until maturity. These covenants include providing information about the Company and our operations; limitations on our ability to retire, repurchase, or redeem our common or preferred stock, options, and warrants other than per the terms of the securities; and limitations on our ability to pay dividends of cash or property. There are no financial covenants associated with the Loan Agreement. Events of default under the Loan Agreement include failure to make payments when due, insolvency events, failure to comply with covenants, and material adverse effects with respect to the Company. We are not in violation of any affirmative or negative covenants. Repayment of the full balance outstanding is due on April 30, 2034. The 2019 Cecil Loan establishes “Phantom Shares,” based on 23,981 shares of our Common Stock (based on 172,622 Series C Preferred Shares prior to the Reverse Recapitalization), determined at issuance. The 2019 Cecil Loan states the repayment amount is to be the greater of the balance of principal and accrued interest or the Phantom Share value. We determined that the note should be accounted for at fair value. We record the fair value of the debt at the end of each reporting period. In order to value the note, we consider the amount of the simple interest expense that would be due and the value of Phantom Shares. Upon the closing of the Reverse Recapitalization and as of December 31, 2020, the fair value of the 2019 Cecil Loan is determined based on the closing price of CLNN shares listed on the Nasdaq. Expense of $0.1 million and $2 thousand was recognized during the three months ended March 31, 2021 and 2020, respectively. The fair value of $0.3 million and $0.2 million of principal and accrued interest is included in long-term notes payable as of March 31, 2020 and December 31, 2020, respectively.
In February through July 2020, we issued convertible promissory notes (the “2020 Convertible Notes”) in an aggregate principal amount of $6.1 million, bearing interest at an annual rate of 5%. The 2020 Convertible Notes were convertible at the earlier of (i) one year, at which point the notes would be convertible into Series C preferred shares at the Series C preferred share issuance price, and (ii) next equity financing of no less than $10.0 million, at which point the notes would be convertible into shares issued in the next equity financing at 90% of the per share issuance price of the next equity financing. The redemption feature at the next equity financing met the requirements of an embedded derivative to be bifurcated and recorded at fair value. We bifurcated the embedded feature at issuance and recorded a derivative liability of $0.7 million at inception in conjunction with a discount on debt to be amortized over the life of the note. We recognized interest expense of $8 thousand, including amortization of debt discount of $20 thousand during the three months ended March 31, 2020, in connection with the 2020 Convertible Notes. We also identified two other embedded features in these convertible promissory notes that were not bifurcated, which were the conversion into Series C preferred shares upon maturity and the redemption upon a liquidation event. On August 11, 2020, in connection with our issuance and sale of Series D Preferred Stock prior to the Reverse Recapitalization, all of the outstanding principal and accrued interest under the 2020 Convertible Notes, totaling $6.9 million, was automatically converted into 1,497,135 shares of Series D Preferred Stock at a price equal to 90% of $4.60 per share, the per share price paid in cash by investors in the Series D preferred stock financing. We accounted for the conversion of the 2020 Convertible Notes as a debt extinguishment and recognized a loss on extinguishment of debt of $0.5 million within other income (expense), net in the consolidated statement of operations and comprehensive loss for the year ended December 31, 2020. As of the date of conversion, the unamortized discount on the 2020 Convertible Notes was $0.5 million. The loss on extinguishment was calculated as the difference between (i) the fair value of the 1,497,135 shares of Series D Preferred Stock issued to settle the 2020 Convertible Notes of $6.9 million and (ii) the carrying value of the 2020 Convertible Notes, including the principal balance of the 2020 Convertible Notes of $6.1 million and accrued but unpaid interest of $76 thousand, net of the unamortized debt discount of $5.7 million, plus the then-current fair value of derivative liability associated with the 2020 Convertible Notes at the time of the extinguishment of $0.7 million.
In May 2020, we entered into a note payable in the amount of $0.6 million (the “PPP Note”) under the Paycheck Protection Program of the CARES Act (the “PPP”). As amended, the PPP permits forgiveness of amounts loaned for payments of payroll and other qualifying expenses within 24 weeks of receipt of loaned funds, given that at least 60% of the total loan is used for payroll. Amounts not forgiven have a repayment period of five years. In January 2021, the full $0.6 million balance of the PPP Note was forgiven and has been recorded as a gain on extinguishment of debt during the three months ended March 31, 2021
Contractual Obligations and Commitments
There have been no material changes to our contractual obligations and other commitments as of March 31, 2021, as compared to those disclosed in Part II, Item 7 “Management’s Discussion and Analysis of Financial Condition and Results of Operations” of our 2020 Annual Report which was filed with the SEC on March 29, 2021.
We enter into agreements in the normal course of business with CROs for clinical trials and with vendors for preclinical studies and other services and products for operating purposes, which are cancelable at any time by us, subject to payment of our remaining obligations under binding purchase orders and, in certain cases, nominal early termination fees. These commitments are not deemed significant.
Long-Term Material Cash Requirements
Off-Balance Sheet ArrangementsBeyond the next twelve months, our primary capital requirements are to fund our operations, including research and development, personnel, regulatory, and other clinical trial costs related to development of our lead drug candidate, CNM-Au8; and general and administrative costs to support our drug development activities in advance of receiving regulatory approval for our drug candidates. Additional funds may be spent to initiate new clinical trials, at our discretion. Known obligations beyond the next twelve months include $0.1 million and $7.8 million of payments under finance and operating lease obligations, respectively; and interest and principal repayment of notes payable of $20.3 million. We expect to meet our long-term liquidity requirements primarily through equity financing, debt financing, or other capital sources.
35
DuringUse of Funds
Our cash flows for the nine months ended September 30, 2022 and 2021 were as follows:
|
| Nine Months Ended September 30, |
| |||||
(in thousands) |
| 2022 |
|
| 2021 |
| ||
Net cash used in operating activities |
| $ | (31,295 | ) |
| $ | (25,018 | ) |
Net cash used in investing activities |
|
| (12,446 | ) |
|
| (661 | ) |
Net cash provided by financing activities |
|
| 875 |
|
|
| 27,130 |
|
Effect of foreign exchange rate changes on cash |
|
| (155 | ) |
|
| (116 | ) |
Net increase (decrease) in cash |
| $ | (43,021 | ) |
| $ | 1,335 |
|
Our primary use of cash in all periods presented was to fund our research and development, regulatory and other clinical trial costs, and general corporate expenditures.
Operating Activities
Net cash used in operating activities was $31.3 million for the nine months ended September 30, 2022, which resulted from a net loss of $28.9 million, adjusted for non-cash items totaling $0.1 million and a net change in operating assets and liabilities of $2.6 million. Significant non-cash items included (i) depreciation expense of $0.7 million relating to laboratory and office equipment and leasehold improvements; (ii) non-cash lease expense of $0.3 million; (iii) stock-based compensation expense of $6.5 million; (iv) gain on termination of lease of $0.4 million; (v) accretion of debt discount of $0.7 million; (vi) non-cash interest expense of $0.1 million; and (vii) the changes in fair value of the Clene Nanomedicine and Initial Stockholders Contingent Earn-outs of $6.7 million and $0.8 million, respectively, and the change in fair value of common stock warrant liability of $0.2 million. The changes in fair value of these instruments were primarily driven by the decrease of the closing price of our Common Stock on Nasdaq. The net change in operating assets and liabilities was primarily attributable to the following: (a) an increase in accounts receivable of $0.1 million and an increase in accounts payable of $0.2 million due to the timing of vendor invoicing and payments; (b) an increase in prepaid expenses and other current assets of $0.9 million due to the timing of vendor invoicing and payments, the timing of receipt of metals to be used in research and development, and an increase in Australia research and development credit receivable; (c) a decrease in accrued liabilities of $1.5 million primarily due to decreased accrued compensation and benefits; and (d) a decrease in operating lease obligations of $0.4 million.
Net cash used in operating activities was $25.0 million for the nine months ended September 30, 2021, which resulted from a net loss of $14.2 million, adjusted for non-cash items totaling $10.4 million and a net change in operating assets and liabilities of $0.5 million. Non-cash items primarily consisted of the following: (i) depreciation expense of $0.7 million, (ii) stock-based compensation expense of $9.9 million, (iii) change in fair value of Clene Nanomedicine Contingent Earn-out of $18.1 million, (iv) change in fair value of Initial Stockholders Contingent Earn-out of $1.7 million, (iv) gain on extinguishment of debt of $0.6 million and increase in interest accrued on notes payable and accretion of debt discount of $0.1 million. The net change in operating assets and liabilities was primarily attributable to the following: (a) a decrease in inventory of $0.2 million, (b) an increase in accounts receivable of $48,000, (c) an increase in prepaid expenses and other current assets of $1.2 million due to the increase in Australia research and development credit receivable, metal to be used in research and development, and directors and officers insurance; partially offset by a decrease in CRO prepayments, (d) an increase in accounts payable of $0.4 million, (e) a decrease in operating lease obligations of $0.1 million, (f) an increase in accrued liabilities of $0.5 million due to the timing of vendor invoicing and payments, and (g) a decrease in deferred income tax of $0.2 million.
Investing Activities
Net cash used in investing activities was $12.4 million for the nine months ended September 30, 2022, which consisted of (i) purchases of marketable securities of $24.6 million and (ii) purchases of property and equipment of $3.5 million, offset primarily by (iii) proceeds from maturity of marketable securities of $8.0 million and (iv) proceeds from sale of marketable securities of $7.6 million. Net cash used in investing activities was $0.7 million for the nine months ended September 30, 2021, which consisted of purchases of property and equipment.
Financing Activities
Net cash provided by financing activities was $0.9 million for the nine months ended September 30, 2022, which primarily consisted of (i) proceeds from exercise of stock options of $0.3 million, (ii) at-the-market offering proceeds of $0.1 million net of $4,000 placement agent commissions, and (iii) proceeds from the issuance of notes payable of $0.7 million, offset primarily by (iv) payments of finance lease obligations of $0.1 million, (v) payment of debt issuance costs of $30,000, and (vi) payment of deferred offering costs of $0.1 million. Net cash provided by financing activities was $27.1 million for the nine months ended September 30, 2021, which primarily consisted of (i) proceeds from exercise of stock options of $0.4 million, (ii) proceeds from the issuance of notes payable of $20.0 million offset by payments of debt issuance costs of $0.5 million, and (iii) proceeds from the May 2021 private placement of
36
common stock of $9.3 million, offset primarily by (iv) payments of finance lease obligations of $0.1 million and (v) payments of deferred offering costs of $1.9 million.
Maryland DHCD Loans
In February 2019, we entered into a loan agreement (the “2019 MD Loan”) with the Department of Housing and Community Development (“DHCD”), a principal department of the State of Maryland. The agreement provides for a term loan of $0.5 million bearing simple interest at an annual rate of 8.00%. We are subject to affirmative and negative covenants until maturity, including providing information about the Company and our operations; limitations on our ability to retire, repurchase, or redeem our common or preferred stock, options, and warrants other than per the terms of the securities; and limitations on our ability to pay dividends of cash or property. There are no financial covenants associated with the 2019 MD Loan. We are not in violation of any covenants. The 2019 MD Loan established “Phantom Shares” at issuance based on 119,907 shares of Common Stock. Repayment of the full balance is due on February 22, 2034, with the repayment amount and carrying value equal to the greater of the balance of principal plus accrued interest or the value of the Phantom Shares. The value of the Phantom Shares is based on the closing price of our Common Stock on Nasdaq at the end of each reporting period. As of September 30, 2022 and December 31, 2021, the note was recorded at principal plus accrued interest in the condensed consolidated balance sheets.
In May 2022, we entered into a loan agreement (the “2022 MD Loan”) with DHCD which provides for a term loan of up to $3.0 million bearing simple interest at an annual rate of 6.00% for the purchase of certain personal property (the “Assets”) related to the production of pharmaceutical drugs. As of September 30, 2022, we had drawn $0.7 million under the term loan, with the remainder available upon our submission of disbursement requests to purchase the Assets. The 2022 MD Loan matures on July 1, 2027 (the “Maturity Date”). The first twelve payments, commencing on July 1, 2022, are deferred. Immediately thereafter, there shall be eighteen monthly installments of interest-only based on the actual amount advanced under the loan, each up to a maximum amount of $15,000; followed by thirty monthly installments of principal and interest, each in the amount of $33,306, which is due and payable even if the entire loan has not been advanced prior to the date such monthly payment is due and payable, with a balloon payment of all accrued and unpaid interest and principal due on the Maturity Date. We recorded $31,000 of debt issuance costs that are being amortized over the contractual term using the effective interest method. Pursuant to the 2022 MD Loan, DHCD was granted a continuing security interest in the Assets as collateral. Under a priority of liens agreement by and between DHCD and Avenue Venture Opportunities Fund, L.P. (“Avenue”), an existing secured creditor of the Company, DHCD’s continuing security interest in the Assets shall be a first priority lien.
Advance Cecil Inc. Loan
In April 2019, we entered into a loan agreement (the “2019 Cecil Loan”) with Advance Cecil Inc., a non-stock corporation formed under the laws of the state of Maryland. The agreement provides for a term loan of $0.1 million bearing simple interest at an annual rate of 8.00%. We are subject to affirmative covenants until maturity, including providing information about the Company and our operations. There are no financial covenants associated with the 2019 Cecil Loan. We are not in violation of any covenants. The 2019 Cecil Loan established “Phantom Shares” at issuance based on 23,981 shares of Common Stock. Repayment of the full balance is due on April 30, 2034, with the repayment amount and carrying value equal to the greater of the balance of principal plus accrued interest or the value of the Phantom Shares. The value of the Phantom Shares is based on the closing price of our Common Stock on Nasdaq at the end of each reporting period. As of September 30, 2022 and December 31, 2021, the note was recorded at principal plus accrued interest in the condensed consolidated balance sheets.
Avenue Loan
In May 2021, we entered into a loan agreement (the “2021 Avenue Loan”) with Avenue. The agreement provides for a 42-month term loan of up to $30.0 million. The first tranche is $20.0 million (“Tranche 1”), of which $15.0 million was funded at close and $5.0 million was funded in September 2021. We incurred issuance costs of $0.6 million of which $46,951 was expensed immediately. The remaining unfunded tranche of $10.0 million (“Tranche 2”) is available until December 31, 2022. Funding of Tranche 2 is subject to (a) our receipt of $5.0 million financing through the state of Maryland; (b) our achievement of a statistically significant result in certain clinical trials (“Performance Milestone 1”); (c) our receipt of net proceeds of at least $30.0 million from the sale and issuance of our equity securities between May 2, 2021 and December 31, 2022; and (d) mutual agreement of us and Avenue. The 2021 Avenue Loan bears interest at a variable rate equal to the sum of (i) the greater of (a) the prime rate or (b) 3.25%, plus (ii) 6.60%. As of September 30, 2022 and December 31, 2021, the interest rate was 12.85% and 9.85%, respectively. Payments are interest-only for the first 12 months and have been extended an additional 12 months (the “First Interest-only Period Extension”) based on our achievement of Performance Milestone 1. Payments may be extended up to 36 months if we (i) achieve the First Interest-only Period Extension and (b) draw from Tranche 2. The loan principal will amortize equally from the end of the interest period presented,to the expiration of the 42-month term on December 1, 2024. On the maturity date, an additional payment equal to 4.25% of the funded loans, currently equal to $0.9 million (the “Final Payment”), is due in addition to the remaining unpaid principal and accrued interest. The Final Payment was recorded as a debt premium and is being amortized over the contractual term using the effective interest method. The Final Payment is related to the loan
37
host and is not bifurcated pursuant to Accounting Standards Codification (“ASC”) 815, Derivatives and Hedging (“ASC 815”). We are subject to affirmative and negative covenants until maturity in the absence of prepayments, including providing information about the Company and our operations; limitation on our ability to retire, repurchase, or redeem our Common Stock, options, and warrants other than per the terms of the securities; and limitations on our ability to pay dividends of cash or property. Also pursuant to the 2021 Avenue Loan, we are required to maintain unrestricted cash and cash equivalents of at least $5.0 million, provided that upon our (i) achievement of Performance Milestone 1, and (ii) receiving of net proceeds of at least $30.0 million from the sale and issuance of our equity securities, we shall no longer be subject to financial covenants. We are not in violation of any covenants. Avenue also has the ability to make all obligations under the 2021 Avenue Loan immediately due and payable upon occurrence of certain events of default or material adverse effects, as outlined in the loan agreement. The 2021 Avenue Loan is collateralized by substantially all of our assets other than intellectual property, including our capital stock and the capital stock of our subsidiaries, in which Avenue is granted a continuing security interest.
Pursuant to the agreement, we granted Avenue a warrant to purchase 115,851 shares of Common Stock (the “Avenue Warrant”) at an exercise price of $8.63 per share. Upon the funding of Tranche 2, the Avenue Warrant shall be adjusted to include an additional estimated 184,133 shares of Common Stock, which is equal to 5% of the principal amount of Tranche 2, divided by the five (5)-day VWAP per share as of the end of trading on the last trading day before the issuance of Tranche 2. We accounted for the Tranche 2 contingently-issuable warrant at inception of the 2021 Avenue Loan in accordance with ASC 815 and the fair value and issuable shares are remeasured at each reporting period.
Avenue has the right, in its discretion, but not the obligation, at any time between May 21, 2022 through May 21, 2024, while the loan is outstanding, to convert up to $5.0 million of principal into Common Stock (the “Conversion Feature”) at a price per share equal to 120% of the Avenue Warrant exercise price. The Conversion Feature is subject to certain minimum price and volume conditions of our Common Stock on Nasdaq. The Conversion Feature did not have,meet the requirements for separate accounting and is not accounted for as a derivative instrument. The number of shares of Common Stock contingently issuable upon conversion is 482,703 shares. We classified $5.0 million of the 2021 Avenue Loan as convertible notes payable as of September 30, 2022 and December 31, 2021, with unamortized debt discount and issuance costs of $0.2 million and $0.4 million, respectively.
At-the-Market Facility
On April 14, 2022, we currently do not have,entered into an Equity Distribution Agreement (the “ATM Facility”) with Canaccord Genuity LLC and Oppenheimer & Co. Inc., as placement agents (the “Placement Agents”). In accordance with the terms of the ATM Facility, we may offer and sell shares of Common Stock having an aggregate offering price of up to $50.0 million from time to time through the Placement Agents. The issuance and sale of Common Stock, if any, off-balance sheet arrangements, such as relationships with unconsolidated entities or financial partnerships,by us under the ATM Facility will be made pursuant to our registration statement on Form S-3 (file number 333-264299), which are often referredwas declared effective by the Securities and Exchange Commission on April 26, 2022 , and our prospectus supplement relating to as structured finance or special purpose entities, established for the purposeoffering.
Subject to terms of facilitating financing transactions thatthe ATM Facility, the Placement Agents are not required to be reflectedsell any specific number or dollar amount of Common Stock but will act as our placement agents, using commercially reasonable efforts to sell, on our balance sheets.behalf, all of the Common Stock requested by us to be sold, consistent with the Placement Agents’ normal trading and sales practices, on terms mutually agreed between the Placement Agents and us. The Placement Agents will be entitled to compensation under the terms of the ATM Facility at a fixed commission rate of 3.0% of the gross proceeds from each issuance and sale of Common Stock, if any.
During the three and nine months ended September 30, 2022, we sold 40,000 shares of Common Stock under the ATM Facility and generated gross proceeds of $0.1 million. Commissions paid to the Placement Agents were insignificant.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations is based on our condensed consolidated financial statements, which have been prepared in accordance with GAAP.U.S. Generally Accepted Accounting Principles. The preparation of these condensed consolidated financial statements requires us to make estimates, assumptions, and judgments that affect the reported amounts of assets, liabilities, revenues, costs, and expenses. We evaluate our estimates and judgments on an ongoing basis, and our actual results may differ from these estimates. We base our estimates on historical experience, known trends and events, contractual milestones, and other various factors that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources.
Our mostWe consider the following estimates to be critical accounting policiesas they involve a significant level of estimation uncertainty and have had or are described under the heading “Management’s Discussionreasonably likely to have a material impact on our financial condition and Analysisresults of Financial Condition and Results of Operations–Critical Accounting Policies” in Part II, Item 7 of our 2020 Annual Report which was filed with the SEC on March 29, 2021. There were no material changes to our critical accounting policies through March 31, 2021 from those discussed in our 2020 Annual Report.
Recent Accounting Pronouncements
operations. See Note 2 to our condensed consolidated financial statements included in Part I, Item 1, “Notes to Condensed Consolidated Financial Statements” of this Quarterly Report on Form 10-Q for a description of recentother significant accounting pronouncementspolicies.
38
Contingent Earn-Out Liabilities
In connection with the Reverse Recapitalization, certain stockholders are entitled to the Contingent Earn-outs payments based on achievement of certain milestones. In accordance with ASC 815, we classified the Contingent Earn-outs as liabilities and measured them at fair value on the date of the Reverse Recapitalization. We remeasure the liabilities at each reporting date and record the change in fair value as a component of other income (expense), net, in the condensed consolidated statements of operations and comprehensive income (loss). We estimate the fair value using a Monte Carlo valuation model, which requires significant judgment. The unobservable inputs include the expected stock price volatility, the risk-free interest rate, and the expected term.
As of September 30, 2022 and December 31, 2021, the unobservable inputs were as follows:
|
| September 30, |
|
| December 31, |
| ||
|
| 2022 |
|
| 2021 |
| ||
Expected stock price volatility |
|
| 115.00 | % |
|
| 105.00 | % |
Risk-free interest rate |
|
| 4.20 | % |
|
| 1.10 | % |
Expected dividend yield |
|
| 0.00 | % |
|
| 0.00 | % |
Expected term |
| 3.25 years |
|
| 4.00 years |
| ||
The change in fair value of the Clene Nanomedicine Contingent Earn-out resulted in a loss of $1.6 million and gain of $35.0 million for the three months ended September 30, 2022 and 2021, respectively; and gains of $6.7 million and $18.1 million for the nine months ended September 30, 2022 and 2021, respectively. The change in fair value of the Initial Stockholders Contingent Earn-out resulted in a loss of $0.2 million and gain of $3.4 million for the three months ended September 30, 2022 and 2021, respectively; and gains of $0.8 million and $1.7 million for the nine months ended September 30, 2022 and 2021, respectively.
Convertible Notes
Pursuant to the 2021 Avenue Loan, $5.0 million of the outstanding principal is subject to the Conversion Feature. In accordance with Accounting Standards Update (“ASU”) 2020-06, Debt—Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, we classified this portion as convertible notes payable in the condensed consolidated balance sheets and did not bifurcate the Conversion Feature from the host contract. Consequently, we account for the convertible note as a single liability measured at its amortized cost. As of September 30, 2022 and December 31, 2021, the convertible note was carried at $4.8 million and $4.6 million, respectively.
Common Stock Warrant Liability
Pursuant to the 2021 Avenue Loan, we issued the Avenue Warrant. In accordance with ASC 815, we recognized both the issued Tranche 1 warrant and the Tranche 2 warrant issuable pursuant to the potential draw of Tranche 2 as liabilities measured at fair value. As we did not complete a bona fide round of equity financing by March 31, 2022, the exercise price and underlying shares of the Tranche 1 warrant became fixed and therefore qualified for equity classification. We remeasured the Tranche 1 warrant liability to fair value as of March 31, 2022 and recognized the change in fair value in the condensed consolidated statements of operations and comprehensive income (loss) and the Tranche 1 warrant liability was reclassified to additional paid-in-capital. We remeasure the Tranche 2 warrant liability at each reporting date and record the change in fair value as a component of other income (expense), net, in the condensed consolidated statements of operations and comprehensive income (loss).
We estimate the fair value using a Black-Scholes option-pricing model, with a probability weight related to the potential draw of Tranche 2, which requires significant judgment. The unobservable inputs include the expected stock price volatility, risk-free interest rate, expected term, and the probability of drawing Tranche 2. As of September 30, 2022 and December 31, 2021, the unobservable inputs were as follows:
|
| September 30, |
|
| December 31, |
| ||
|
| 2022 |
|
| 2021 |
| ||
Expected stock price volatility |
|
| 115.00 | % |
|
| 105.00 | % |
Risk-free interest rate |
|
| 4.20 | % |
|
| 1.20 | % |
Expected dividend yield |
|
| 0.00 | % |
|
| 0.00 | % |
Expected term |
| 3.47 years |
|
| 3.89 – 4.39 years |
| ||
Probability of drawing Tranche 2 |
|
| 5.00 | % |
|
| 50.00 | % |
39
We recorded a change in fair value of the common stock warrant liability of $0.1 million and $0.4 million for the three months ended September 30, 2022 and 2021, respectively; and $0.2 million and $0.5 million for the nine months ended September 30, 2022 and 2021, respectively.
Income Taxes
We account for uncertainty in income taxes by applying a two-step process to determine the amount of tax benefit to be recognized in the condensed consolidated financial statements. First, the tax position is evaluated to determine the likelihood that it will be sustained upon external examination by the taxing authorities. If the tax position is deemed more-likely-than-not to be sustained, the tax position is then assessed to determine the amount of benefit to recognize. The amount of the benefit that may be recognized is the largest amount that has a greater than 50% likelihood of being realized upon ultimate settlement. Additionally, we assess the likelihood that our deferred tax assets will be recovered from future taxable income and, to the extent we believe, based upon the weight of available evidence, that it is more likely than not that all or a portion of the deferred tax assets will not be realized, a valuation allowance is established through a charge to income tax expense. Potential for recovery of deferred tax assets is evaluated by estimating the future taxable profits expected and considering prudent and feasible tax planning strategies. The estimation of these factors requires significant judgment. Based on our evaluation of these factors, we have not recorded income tax benefits for the net operating losses or for research and development tax credits or other deferred tax assets due to uncertainty of realizing benefits from these items.
Stock-Based Compensation
We account for stock-based compensation arrangements using a fair value-based method for costs related to all share-based payments including stock options and stock awards. The fair value is recognized over the period during which a grantee was required to provide services in exchange for the option award and service-based stock awards, known as the requisite service period (usually the vesting period), on a straight-line basis. For stock awards with market conditions, the fair value is recognized over the period based on the expected milestone achievement dates as the derived service period (usually the vesting period), on a straight-line basis. For stock awards with performance conditions, the grant-date fair value of these awards is the market price on the applicable grant date, and compensation expense will be recognized when the conditions become probable of being satisfied. We will recognize a cumulative true-up adjustment once the conditions become probable of being satisfied as the related service period had been completed in a prior period. We elect to our business.account for forfeitures as they occur, rather than estimating expected forfeitures.
We estimate the fair value of stock options using a Black-Scholes option-pricing model, which requires significant judgment. The unobservable inputs include the expected price volatility, risk-free interest rate, expected dividend yield, and expected term. For the nine months ended September 30, 2022 and 2021, the unobservable inputs were as follows:
|
| Nine Months Ended September 30, | ||
|
| 2022 |
| 2021 |
Expected stock price volatility |
| 89.57% – 98.13% |
| 84.80% – 87.40% |
Risk-free interest rate |
| 1.65% – 3.00% |
| 0.59% – 0.94% |
Expected dividend yield |
| 0.00% |
| 0.00% |
Expected term of options |
| 5.00 – 6.98 years |
| 6.00 years |
ITEMWe estimate the fair value of restricted stock awards using a Monte Carlo valuation model to simulate the achievement of certain stock price milestones. The unobservable inputs include the expected stock price volatility, risk-free interest rate, and expected term. No restricted stock awards were granted during the nine months ended September 30, 2022 and 2021.
Item 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISKQuantitative and Qualitative Disclosures About Market Risk
InformationAs a smaller reporting company, we are not required to provide information required by this Item.
Item is not applicable as we are electing scaled disclosure requirements available to Smaller Reporting Companies with respect to this Item.4. Controls and Procedures
ITEM 4. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our Chief Executive Officerprincipal executive officer and Chief Financial Officer,principal financial officer, we conducted an evaluation of the effectiveness of our disclosure controls and procedures as of March 31, 2021,September 30, 2022, as such term is defined in Rules 13a-15(e) and 15d-15(e) underof the Securities Exchange Act. Based onAct of 1934 (the “Exchange Act”). As a result of this evaluation, our Chief Executive Officerprincipal executive officer and Chief Financial Officerprincipal financial officer have concluded that, as of March 31, 2021,September 30, 2022, our disclosure controls and procedures were not effective due to the material weaknesses in internal control over financial reporting described below. Notwithstanding the identified material weaknesses, management, including our Chief Executive Officerprincipal executive officer and Chief Financial Officer,principal financial officer, believes the condensed consolidated financial statements included in this Quarterly Report on Form 10-Q fairly represent, in all
40
material respects, our financial condition, results of operations and cash flows atas of and for the periods presented in accordance with U.S. GAAP.United States Generally Accepted Accounting Principles.
Disclosure controls and procedures are designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’sSecurities and Exchange Commission (the “SEC”) rules and forms, and that such information is accumulated and communicated to our management, including our Chief Executive Officerprincipal executive officer and Chief Financial Officer,principal financial officer, as appropriate to allow timely decisions regarding required disclosure.
Material Weaknesses in Internal Control over Financial Reporting
In connection with the audit of our financial statements as of and for the years ended December 31, 20202021 and 2019,2020, our management identified material weaknesses in our internal control over financial reporting. A material weakness is a deficiency, or combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis. The material weaknesses identified relate to the fact that we did not design or maintain an effective control environment commensurate with our financial reporting requirements, including (a) lack of a sufficient number of trained professionals with an appropriate level of accounting knowledge, training and experience to appropriately analyze, record and disclose accounting matters timely and accurately, and (b) lack of structures, reporting lines and appropriate authorities and responsibilities to achieve financial reporting objectives.requirements. This deficiency in our control environment contributed to the following additional deficiencies (each of which individually represents a material weakness) inweaknesses related to control activities and information and communication within our internal control over financial reporting:
•
The control deficiencies described above resulted in the misstatement of our redeemable convertible preferred stock warrant liability, accrued liabilities, general and administrative expenses, Australian research and development credit, and amounts and classification within our statement of cash flows and related financial disclosures as of and for the year ended December 31, 2019 and in the misstatement of our prepaid expenses and other current assets, accrued liabilities, earn-out liabilities, redeemable convertible preferred stock warrant liability, general and administrative expenses, amounts and classification within our statement of equity, and amounts and classification within our statement of cash flows and related financial disclosures as of and for the year ended December 31, 2020. Additionally, eachEach of the control deficiencies described above could result in a misstatement of one or more account balances or disclosures that would result in a material misstatement to the annual or interim consolidated financial statements that would not be prevented or detected. Accordingly, our management has determined that each of the control deficiencies described above constitute material weaknesses.
Material Weakness Remediation
Management iscontinues to be actively engaged and committed to taking the steps necessary to remediate the control deficiencies that constituted the above material weakness. During 2020,2021, we made the following enhancements to our control environment:
Our remediation activities are continuing during 2021.2022. In addition to the above actions, we expect to engage in additional activities, or have completed additional activities, including, but not limited to:
41
We continue to enhance corporate oversight over process-level controls and structures to ensure that there is appropriate assignment of authority, responsibility, and accountability to enable remediation of our material weaknesses. We believe that our remediation plan will be sufficient to remediate the identified material weakness and strengthen our internal control over financial reporting. As we continue to evaluate, and work to improve, our internal control over financial reporting, management may determine that additional measures to address control deficiencies or modifications to the remediation plan are necessary.
Changes in Internal Control over Financial Reporting
We are engagedOther than changes described under “—Material Weakness Remediation,” there were no changes in the process of the design and implementation of our internal control over financial reporting in a manner commensurate with the scale of our operations following the Reverse Recapitalization. Duringduring the quarter ended March 31, 2021, we began implementing a new ERPSeptember 30, 2022, that have materially affected, or are reasonably likely to enhance the accuracy ofmaterially affect, our internal control over financial records, enable the enforcement of systematic segregation of duties, and to improve our information technology general controls environment.reporting.
42
PART II – II—OTHER INFORMATION
Item 1. Legal Proceedings
We may be subject to legal proceedings, investigations and claims incidental to the conduct of our business from time to time. We are not currently a party to any material litigation or otherpending legal proceedings. From time to time, we may, however, be involved in legal proceedings brought against us.in the ordinary course of business. We are also not awarecannot predict the outcome of any such legal proceeding, investigation or claim, orproceedings, and despite the potential outcomes, the existence thereof may have an adverse impact on us because of defense and settlement costs, diversion of management resources and other legal exposure that has a more than remote possibility of having a material adverse effect on our business, financial condition or results of operations.factors.
Item 1A. Risk Factors
Our business, financial condition, and results of operations can be affected by a number of factors, whether currently known or unknown, including but not limited to those described in Part I, Item 1A, “Risk Factors”Risk Factors of our 20202021 Annual Report on Form 10-K (“2020 Annual Report”) which was filed with the SEC on March 29, 2021.11, 2022. There have been no material changes to ourthe risk factors since previously disclosed in the 20202021 Annual Report.Report on Form 10-K. Any one or more of these factors could, directly or indirectly, cause our actual financial condition and results of operations to vary materially from past, or from anticipated future, financial condition and results of operations. Any of these factors, in whole or in part, could materially and adversely affect our business, financial condition, results of operations, and stock price.
ITEMItem 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDSUnregistered Sales of Equity Securities and Use of Proceeds
None. None. None. Item 3. Defaults Upon Senior Securities None. Item 4. Mine Safety Disclosures Not Applicable. Item 5. Other Information 43 Exhibits * Filed herewith. ** Furnished herewith. # Schedules and similar attachments to this exhibit have been omitted pursuant to SIGNATURES Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized. CLENE INC. Dated: By: /s/ Robert Etherington Name: Robert Etherington Title: President, Chief Executive Officer and Director Dated: By: /s/ Name: Morgan R. Brown Title: Chief Financial Officer 45None.We entered into subscription agreements with various investors for the private placement of Common Stock (the “Private Placement”), all of which closed shortly before the closing of the Reverse Recapitalization. Under the Private Placement, 2,239,500 shares of Common Stock (the “PIPE Shares”) were sold, resulting in net proceeds of $22.2 million. Pursuant to the subscription agreements, investors in the Private Placement also received warrants to purchase a number of shares equal to one-half (1/2) of the number of PIPE Shares, totaling 1,119,750 shares of PubCo Common Stock, at an exercise price of $0.01 per share for each of the PIPE Shares (the “PIPE Warrants”), subject to a 180-day holding period. We filed a registration statement on Form S-1 to register the PIPE Shares and the Common Stock underlying the PIPE Warrants, which the SEC declared to be effective on April 19, 2021 (file number 333-253173).We have been using and will continue to use these proceeds primarily (1) to fund our VISIONARY-MS, REPAIR-MS, REPAIR-PD, and RESCUE-ALS studies, and for our participation in the Healey-ALS Platform Trial, and our other clinical research and development activities, and (2) for general and administrative purposes.ITEM 3. DEFAULTS UPON SENIOR SECURITIESNone.ITEM 4. MINE SAFETY DISCLOSURESITEM 5. OTHER INFORMATIONNone.None.*Portions ofRule 601(b)(10)Item 601(a)(5) of Regulation S-K. TheWe agree to furnish supplementally a copy of such omitted information is not material and would likely cause competitive harmmaterials to the registrant if publicly disclosed.**Indicates a management contract or a compensatory plan or agreement.SEC upon request.CLENE INC.May 10, 2021November 7, 2022May 10, 2021November 7, 2022Ted (Tae Heum) JeongMorgan R. BrownTed (Tae Heum) Jeong49