UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_______________________
FORM 10-Q
_______________________
|
|
☒ | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended SeptemberJune 30, 20162017
or
|
|
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number 001-35952
_______________________
ARATANA THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
_______________________
|
|
|
|
|
|
Delaware |
| 38-3826477 |
(State or other jurisdiction of incorporation or organization) |
| (I.R.S. Employer Identification Number) |
11400 Tomahawk Creek Parkway
Suite 340
Leawood, KS 66211
(913) 353-1000
(Address of principal executive offices, zip code and telephone number, including area code)
_______________________
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes: ☒ No: ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes: ☒ No: ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting company”, and “smaller reporting“emerging growth company” in Rule 12b-2 of the Exchange Act.
|
|
|
|
|
|
|
Large accelerated filer |
| ☐ |
| Accelerated filer |
| ☒ |
Non-accelerated filer |
| ☐ |
| Smaller reporting company |
| ☐ |
Emerging growth company | ☒ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes: ☐ No: ☒
As of October 31, 2016,August 1, 2017, there were 37,044,32243,001,799 shares of common stock outstanding.
ARATANA THERAPEUTICS, INC.
TABLE OF CONTENTSTable of Contents
|
|
| |
|
| Page | |
| |||
Item 1. | 3 | ||
| Consolidated Balance Sheets as of | 3 | |
| 4 | ||
| 5 | ||
| 6 | ||
| 7 | ||
Item 2. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | ||
Item 3. | |||
Item 4. | |||
| |||
Item 1. | |||
Item 1A. | |||
Item 2. | |||
Item 3. | |||
Item 4. | |||
Item 5. | |||
Item 6. | |||
2
Consolidated Balance Sheets (Unaudited)
(Amounts in thousands, except share and per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| September 30, 2016 |
| December 31, 2015 |
| June 30, 2017 |
| December 31, 2016 | ||||
Assets |
|
|
|
|
|
|
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
| $ | 100,847 |
| $ | 26,755 |
| $ | 78,823 |
| $ | 87,307 |
Short-term investments |
|
| 1,494 |
|
| 59,447 |
|
| 1,494 |
|
| 996 |
Accounts receivable, net |
|
| 15 |
|
| 60 |
|
| 4,893 |
|
| 87 |
Inventories |
|
| 5,575 |
|
| 1,306 |
|
| 6,961 |
|
| 11,130 |
Prepaid expenses and other current assets |
|
| 3,014 |
|
| 1,451 |
|
| 6,605 |
|
| 2,022 |
Total current assets |
|
| 110,945 |
|
| 89,019 |
|
| 98,776 |
|
| 101,542 |
Property and equipment, net |
|
| 2,122 |
|
| 2,555 |
|
| 1,503 |
|
| 1,948 |
Goodwill |
|
| 40,275 |
|
| 39,781 |
|
| 40,500 |
|
| 39,382 |
Intangible assets, net |
|
| 13,333 |
|
| 15,067 |
|
| 11,033 |
|
| 7,639 |
Restricted cash |
|
| 350 |
|
| 350 |
|
| 350 |
|
| 350 |
Other long-term assets |
|
| 289 |
|
| 294 |
|
| 555 |
|
| 545 |
Total assets |
| $ | 167,314 |
| $ | 147,066 |
| $ | 152,717 |
| $ | 151,406 |
Liabilities and Stockholders’ Equity |
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
| $ | 5,113 |
| $ | 1,400 |
| $ | 1,902 |
| $ | 7,436 |
Accrued expenses |
|
| 3,454 |
|
| 4,247 |
|
| 5,602 |
|
| 5,827 |
Licensing and collaboration commitment |
|
| 7,000 |
|
| — |
|
| 7,000 |
|
| 7,000 |
Current portion – loans payable |
|
| 5,833 |
|
| — |
|
| 8,797 |
|
| 14,413 |
Other current liabilities |
|
| 8 |
|
| 37 |
|
| — |
|
| 12 |
Total current liabilities |
|
| 21,408 |
|
| 5,684 |
|
| 23,301 |
|
| 34,688 |
Loans payable, net |
|
| 34,235 |
|
| 39,710 |
|
| 29,296 |
|
| 25,775 |
Other long-term liabilities |
|
| 440 |
|
| 122 |
|
| 72 |
|
| 540 |
Total liabilities |
|
| 56,083 |
|
| 45,516 |
|
| 52,669 |
|
| 61,003 |
Commitments and contingencies (Notes 5, 8 and 10) |
|
|
|
|
|
| ||||||
Commitments and contingencies (Notes 4 and 15) |
|
|
|
|
|
| ||||||
Stockholders’ equity: |
|
|
|
|
|
|
|
|
|
|
|
|
Common stock, $0.001 par value; 100,000,000 shares authorized at September 30, 2016 and December 31, 2015, 36,318,541 and 34,563,816 issued and outstanding at September 30, 2016 and December 31, 2015, respectively |
|
| 36 |
|
| 35 | ||||||
Common stock, $0.001 par value; 100,000,000 shares authorized at June 30, 2017 and December 31, 2016, 42,402,677 and 36,607,922 issued and outstanding at June 30, 2017 and December 31, 2016, respectively |
|
| 42 |
|
| 37 | ||||||
Treasury stock |
|
| (1,088) |
|
| (1,088) |
|
| (1,099) |
|
| (1,088) |
Additional paid-in capital |
|
| 282,972 |
|
| 263,941 |
|
| 318,044 |
|
| 286,909 |
Accumulated deficit |
|
| (162,260) |
|
| (152,018) |
|
| (208,798) |
|
| (185,593) |
Accumulated other comprehensive loss |
|
| (8,429) |
|
| (9,320) |
|
| (8,141) |
|
| (9,862) |
Total stockholders’ equity |
|
| 111,231 |
|
| 101,550 |
|
| 100,048 |
|
| 90,403 |
Total liabilities and stockholders’ equity |
| $ | 167,314 |
| $ | 147,066 |
| $ | 152,717 |
| $ | 151,406 |
The accompanying notes are an integral part of these unaudited interim consolidated financial statements.
3
Consolidated Statements of Operations (Unaudited)
(Amounts in thousands, except share and per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
| Nine Months Ended |
| Three Months Ended |
| Six Months Ended | ||||||||||||||||
|
| September 30, |
| September 30, |
| June 30, |
| June 30, | ||||||||||||||||
|
| 2016 |
| 2015 |
| 2016 |
| 2015 |
| 2017 |
| 2016 |
| 2017 |
| 2016 | ||||||||
Revenues |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Licensing and collaboration revenue |
| $ | — |
| $ | — |
| $ | 38,151 |
| $ | — |
| $ | 804 |
| $ | 38,000 |
| $ | 1,707 |
| $ | 38,151 |
Product sales |
|
| 40 |
|
| 229 |
|
| 108 |
|
| 615 |
|
| 4,354 |
|
| 47 |
|
| 7,246 |
|
| 68 |
Total revenues |
|
| 40 |
|
| 229 |
|
| 38,259 |
|
| 615 |
|
| 5,158 |
|
| 38,047 |
|
| 8,953 |
|
| 38,219 |
Costs and expenses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of product sales |
|
| 286 |
|
| 138 |
|
| 2,046 |
|
| 357 |
|
| 3,691 |
|
| 1,741 |
|
| 6,785 |
|
| 1,760 |
Royalty expense |
|
| 19 |
|
| 23 |
|
| 57 |
|
| 66 |
|
| 353 |
|
| 20 |
|
| 676 |
|
| 38 |
Research and development |
|
| 5,334 |
|
| 6,197 |
|
| 21,386 |
|
| 18,499 |
|
| 3,700 |
|
| 5,303 |
|
| 8,354 |
|
| 16,052 |
Selling, general and administrative |
|
| 6,924 |
|
| 4,997 |
|
| 19,623 |
|
| 14,061 |
|
| 6,918 |
|
| 6,148 |
|
| 14,413 |
|
| 12,699 |
Amortization of intangible assets |
|
| 91 |
|
| 483 |
|
| 281 |
|
| 1,449 |
|
| 86 |
|
| 95 |
|
| 150 |
|
| 190 |
Impairment of intangible assets |
|
| — |
|
| 43,398 |
|
| 2,780 |
|
| 43,398 |
|
| — |
|
| 2,780 |
|
| — |
|
| 2,780 |
Total costs and expenses |
|
| 12,654 |
|
| 55,236 |
|
| 46,173 |
|
| 77,830 |
|
| 14,748 |
|
| 16,087 |
|
| 30,378 |
|
| 33,519 |
Loss from operations |
|
| (12,614) |
|
| (55,007) |
|
| (7,914) |
|
| (77,215) | ||||||||||||
Income (loss) from operations |
|
| (9,590) |
|
| 21,960 |
|
| (21,425) |
|
| 4,700 | ||||||||||||
Other income (expense) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
| 116 |
|
| 33 |
|
| 276 |
|
| 147 |
|
| 88 |
|
| 83 |
|
| 173 |
|
| 160 |
Interest expense |
|
| (859) |
|
| (226) |
|
| (2,554) |
|
| (661) |
|
| (871) |
|
| (846) |
|
| (1,731) |
|
| (1,695) |
Other income (expense), net |
|
| (14) |
|
| — |
|
| (50) |
|
| 5,141 | ||||||||||||
Total other income (expense) |
|
| (757) |
|
| (193) |
|
| (2,328) |
|
| 4,627 | ||||||||||||
Loss before income taxes |
|
| (13,371) |
|
| (55,200) |
|
| (10,242) |
|
| (72,588) | ||||||||||||
Income tax benefit |
|
| — |
|
| 758 |
|
| — |
|
| 1,389 | ||||||||||||
Net loss |
| $ | (13,371) |
| $ | (54,442) |
| $ | (10,242) |
| $ | (71,199) | ||||||||||||
Net loss per share, basic and diluted |
| $ | (0.38) |
| $ | (1.58) |
| $ | (0.29) |
| $ | (2.08) | ||||||||||||
Weighted average shares outstanding, basic and diluted |
|
| 35,092,686 |
|
| 34,405,646 |
|
| 34,837,169 |
|
| 34,293,357 | ||||||||||||
Other expense, net |
|
| (7) |
|
| (1) |
|
| (9) |
|
| (36) | ||||||||||||
Total other expense |
|
| (790) |
|
| (764) |
|
| (1,567) |
|
| (1,571) | ||||||||||||
Net income (loss) |
| $ | (10,380) |
| $ | 21,196 |
| $ | (22,992) |
| $ | 3,129 | ||||||||||||
Net income attributable to participating securities |
|
| — |
|
| (20) |
|
| — |
|
| (3) | ||||||||||||
Net income (loss) attributable to common stockholders |
| $ | (10,380) |
| $ | 21,176 |
| $ | (22,992) |
| $ | 3,126 | ||||||||||||
Net income (loss) per share attributable to common stockholders: |
|
|
|
|
|
|
|
|
|
|
|
| ||||||||||||
Net income (loss) per share, basic and diluted |
| $ | (0.26) |
| $ | 0.61 |
| $ | (0.60) |
| $ | 0.09 | ||||||||||||
Weighted average shares outstanding, basic |
|
| 40,206,042 |
|
| 34,762,533 |
|
| 38,486,329 |
|
| 34,708,006 | ||||||||||||
Weighted average shares outstanding, diluted |
|
| 40,206,042 |
|
| 34,938,455 |
|
| 38,486,329 |
|
| 34,779,786 | ||||||||||||
The accompanying notes are an integral part of these unaudited interim consolidated financial statements.
4
Consolidated Statements of Comprehensive LossIncome (Loss) (Unaudited)
(Amounts in thousands)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
| Nine Months Ended | ||||||||
|
| September 30, |
| September 30, | ||||||||
|
| 2016 |
| 2015 |
| 2016 |
| 2015 | ||||
Net loss |
| $ | (13,371) |
| $ | (54,442) |
| $ | (10,242) |
| $ | (71,199) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Other comprehensive income (loss): |
|
|
|
|
|
|
|
|
|
|
|
|
Foreign currency translation adjustment |
|
| 418 |
|
| (60) |
|
| 891 |
|
| (3,251) |
Unrealized gain on available-for-sale securities |
|
| — |
|
| — |
|
| — |
|
| 2,622 |
Net gain reclassified into income on sale of |
|
| — |
|
| — |
|
| — |
|
| (3,874) |
Other comprehensive income (loss) |
|
| 418 |
|
| (60) |
|
| 891 |
|
| (4,503) |
Comprehensive loss |
| $ | (12,953) |
| $ | (54,502) |
| $ | (9,351) |
| $ | (75,702) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
| Six Months Ended | ||||||||
|
| June 30, |
| June 30, | ||||||||
|
| 2017 |
| 2016 |
| 2017 |
| 2016 | ||||
Net income (loss) |
| $ | (10,380) |
| $ | 21,196 |
| $ | (22,992) |
| $ | 3,129 |
Other comprehensive income (loss): |
|
|
|
|
|
|
|
|
|
|
|
|
Foreign currency translation adjustment |
|
| 1,422 |
|
| (704) |
|
| 1,721 |
|
| 473 |
Other comprehensive income (loss) |
|
| 1,422 |
|
| (704) |
|
| 1,721 |
|
| 473 |
Comprehensive income (loss) |
| $ | (8,958) |
| $ | 20,492 |
| $ | (21,271) |
| $ | 3,602 |
The accompanying notes are an integral part of these unaudited interim consolidated financial statements.
5
Consolidated Statements of Cash Flows (Unaudited)
(Amounts in thousands)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Nine Months Ended | Six Months Ended | ||||||||
| September 30, | June 30, | ||||||||
| 2016 |
| 2015 | 2017 |
| 2016 | ||||
Cash flows from operating activities |
|
|
|
|
|
|
|
|
|
|
Net loss | $ | (10,242) |
| $ | (71,199) | |||||
Net income (loss) | $ | (22,992) |
| $ | 3,129 | |||||
Adjustments to reconcile net loss to net cash provided by (used in) operating activities: |
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense |
| 6,536 |
|
| 6,489 |
| 3,658 |
|
| 4,449 |
Depreciation and amortization expense |
| 738 |
|
| 1,596 |
| 605 |
|
| 493 |
Impairment of intangible assets |
| 2,780 |
|
| 43,398 |
| — |
|
| 2,780 |
Gain on sale of marketable securities |
| — |
|
| (3,874) | |||||
Non-cash interest expense |
| 358 |
|
| 30 |
| 237 |
|
| 239 |
Market value adjustments to inventories |
| 1,552 |
|
| — |
| — |
|
| 1,552 |
Change in fair value of contingent consideration |
| — |
|
| (1,248) | |||||
Change in fair value of derivative instruments |
| — |
|
| (1,274) | |||||
Loss on disposition of property and equipment |
| 5 |
|
| — | |||||
Deferred tax benefit |
| — |
|
| (1,389) | |||||
Changes in operating assets and liabilities: |
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net |
| 45 |
|
| 208 |
| (4,806) |
|
| 43 |
Inventories |
| (5,821) |
|
| (746) |
| 4,169 |
|
| (2,812) |
Prepaid expenses and other current assets |
| (941) |
|
| (236) |
| (4,582) |
|
| (101) |
Other assets |
| 13 |
|
| (114) |
| 33 |
|
| 17 |
Accounts payable |
| 3,712 |
|
| (274) |
| (5,535) |
|
| 5,874 |
Accrued expenses and other liabilities |
| (487) |
|
| 529 |
| (709) |
|
| 624 |
Licensing and collaboration commitment |
| 7,000 |
|
| — |
| — |
|
| 7,000 |
Net cash provided by (used in) operating activities |
| 5,248 |
|
| (28,104) |
| (29,922) |
|
| 23,287 |
Cash flows from investing activities |
|
|
|
|
|
|
|
|
|
|
Milestone payments for intangible assets |
| (1,000) |
|
| — |
| (3,000) |
|
| — |
Purchases of property and equipment, net |
| (25) |
|
| (1,856) |
| (11) |
|
| (19) |
Proceeds from sales of marketable securities |
| — |
|
| 7,456 | |||||
Purchase of investments |
| (228,840) |
|
| (1,592,747) |
| (1,988) |
|
| (227,346) |
Proceeds from maturities of investments |
| 286,793 |
|
| 1,621,749 |
| 1,490 |
|
| 286,046 |
Net cash provided by investing activities |
| 56,928 |
|
| 34,602 | |||||
Net cash provided by (used in) investing activities |
| (3,509) |
|
| 58,681 | |||||
Cash flows from financing activities |
|
|
|
|
|
|
|
|
|
|
Taxes paid for awards vested under equity incentive plans |
| (11) |
|
| — | |||||
Proceeds from stock option exercises |
| 54 |
|
| 311 |
| 152 |
|
| 1 |
Proceeds from issuance of common stock under sales agreement, net of commission |
| 12,072 |
|
| — | |||||
Payments for sales agreement costs |
| (262) |
|
| — | |||||
Cash paid for contingent consideration |
| — |
|
| (3,000) | |||||
Net cash provided by (used in) financing activities |
| 11,864 |
|
| (2,689) | |||||
Proceeds from issuance of common stock, net of commission |
| 27,462 |
|
| — | |||||
Payments for common stock issuance costs |
| (345) |
|
| — | |||||
Payments on loans payable |
| (2,332) |
|
| — | |||||
Net cash provided by financing activities |
| 24,926 |
|
| 1 | |||||
Effect of exchange rate on cash |
| 52 |
|
| (111) |
| 21 |
|
| 35 |
Net increase in cash and cash equivalents |
| 74,092 |
|
| 3,698 | |||||
Cash and cash equivalents, beginning of period |
| 26,755 |
|
| 9,823 | |||||
Cash and cash equivalents, end of period | $ | 100,847 |
| $ | 13,521 | |||||
Net increase (decrease) in cash and cash equivalents |
| (8,484) |
|
| 82,004 | |||||
Cash, cash equivalents and restricted cash, beginning of period |
| 87,657 |
|
| 27,105 | |||||
Cash, cash equivalents and restricted cash, end of period | $ | 79,173 |
| $ | 109,109 | |||||
Supplemental disclosure of cash flow information |
|
|
|
|
|
|
|
|
|
|
Cash paid for interest | $ | 2,179 |
| $ | 628 | $ | 1,499 |
| $ | 1,447 |
Supplemental disclosure of noncash investing and financing activities: |
|
|
|
|
| |||||
Non-cash exercise of warrant | $ | — |
| $ | 750 | |||||
Receivable from broker for issuance of common stock | $ | 605 |
| $ | — | |||||
The accompanying notes are an integral part of these unaudited interim consolidated financial statements.
6
ARATANA THERAPEUTICS, INC.
Notes to Consolidated Financial Statements (Unaudited)
(Amounts in thousands, except share and per share data)
1. Summary of Significant Accounting Policies
Business Overview
Aratana Therapeutics, Inc., including its subsidiaries (the “Company” or “Aratana”),was incorporated on December 1, 2010 under the laws of the State of Delaware. The Company is a pet therapeutics company focused on licensing, developing and commercializing innovative biopharmaceutical productstherapeutics for companion animals.dogs and cats. The Company has one operating segment: pet therapeutics.
Basis of Presentation
The accompanying unaudited consolidated financial statements have been prepared in accordance with U.S.United States generally accepted accounting principles (“GAAP”) for interim financial information. Accordingly, they do not include all of the information and footnotes required by GAAP for complete financial statements. These unaudited consolidated financial statements should be read in conjunction with the audited consolidated financial statements of the Company for the year ended December 31, 20152016 and the notes thereto in the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 15, 2016.14, 2017. In the opinion of management, all adjustments, consisting of a normal and recurring nature, considered necessary for a fair presentation, have been included.
The Company has incurred recurring losses and negative cash flows from operations and has an accumulated deficit of $162,260$208,798 as of SeptemberJune 30, 2016.2017. The Company expects to continue to generate operating losses for the foreseeable future. The Company believes that its cash, cash equivalents and short-term investments on hand will be sufficient to fund operations and debt obligations for at least one year from the issuance of these consolidated financial statements.
The Company expects to continue to incur operating losses for the next several years as it works to develop and commercialize its therapeutics and therapeutic candidates. If the Company cannot generate sufficient cash from operations in the future, it may seek to fund its operations through September 30, 2017.collaborations and licensing arrangements, as well as public or private equity offerings or further debt (re)financings. If the Company is not able to raise additional capital on terms acceptable to it, or at all, as and when needed, it may be required to curtail its operations which could include delaying the commercial launch of its therapeutics, discontinuing therapeutic development programs, or granting rights to develop and market therapeutics or therapeutic candidates that it would otherwise prefer to develop and market itself. As disclosed in Note 87 to the consolidated financial statements, the Company has a term loan and a revolving credit facility with an aggregate principal balance of $40,000$37,667 as of SeptemberJune 30, 2016.2017. The terms of this agreement require the Company to receive unrestricted net cash proceeds of at least $45,000 from partnering transactions and/or the issuance of equity securities from October 16, 2015 to October 16, 2016. The loan agreements also require that the Company have at least three products fully United States Department of Agriculture (“USDA”) or U.S. Food and Drug Administration (“FDA”) approved for commercialization by December 31, 2016. With the FDA approval of GALLIPRANT in March 2016 and the receipt of the upfront payment of $45,000 under the Elanco Animal Health, Inc. (“Elanco”) agreement (Note 10) entered into in April 2016, the Company has met both conditions. Additionally, the loan agreement requires that the Company maintain certain minimum liquidity at all times (the greater of cash equal to fifty percent (50%) of outstanding balance or remaining months’ liquidity, which is calculated on an average trailing three (3) month basis, equal to six (6) months or greater), which as of SeptemberJune 30, 2016,2017, was approximately $38,600.$22,523. If the minimum liquidity covenant is not met, the Company may be required to repay the loans prior to their scheduled maturity dates. At June 30, 2017, the Company was in compliance with all financial covenants.
Consolidation
The Company’s consolidated financial statements include its financial statements and those of its wholly-owned subsidiaries and a consolidated variable interest entity.entity (“VIE”) through the deconsolidation date in December 2016. Intercompany balances and transactions are eliminated in consolidation.
To determine if the Company holds a controlling financial interest in an entity, the Company first evaluates if it is required to apply the variable interest entity (“VIE”)VIE model to the entity. Where the Company holds current or potential rights that give it the power to direct the activities of a VIE that most significantly impact the VIE’s economic performance combined with a variable interest that gives it the right to receive potentially significant benefits or the obligation to absorb potentially significant losses, the Company is the primary beneficiary of that VIE. When changes occur to the design of an entity, the Company reconsiders whether it is subject to the VIE model. The Company continuously evaluates whether it is the primary beneficiary of a consolidated VIE.VIE and upon determination that the Company no longer remains the primary beneficiary, the Company deconsolidates the entity and a gain or loss is recognized upon deconsolidation.
Use of Estimates
The preparation of consolidated financial statements in conformity with GAAP requires management to make estimates, judgments and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. Estimates are periodically reviewed in light of changes in circumstances, facts and experience. Actual results could differ from those estimates.
Revenue Recognition
The Company recognizes revenue when all of the following conditions are met:
|
|
|
|
|
|
|
|
7
The Company’s principal revenue streams and their respective accounting treatments are discussed below:
(i) Product sales - Revenue for the sale of products is recognized when delivery has occurred and substantially all the risks and rewards of ownership have been transferred to the customer. Revenue for the sale of products are recorded net of sales returns, allowances and discounts.
(ii) Royalty revenue - Royalty revenue relating to the Company’s out-licensed technology is recognized when reasonably estimable. The revenues are recorded based on estimates of the licensee’s sales that occurred during the relevant period. Differences between actual and estimated royalty revenues are adjusted for in the period in which they become known, typically in the following quarter. If the Company is unable to reasonably estimate royalty revenue or does not have access to the information, then the Company records royalty revenue on a cash basis.
(iii) Licensing and collaboration revenues - Revenues derived from product out-licensing arrangements typically consist of an initial up-front payment at inception of the license and subsequent milestone payments contingent on the achievement of certain regulatory, development and commercial milestones.
Product out-licensing arrangements with multiple elements are divided into separate units of accounting if certain criteria are met. The up-front payment received is allocated among the separate units of accounting based on their respective fair values, and the applicable revenue recognition criteria are applied to each of the separate units of accounting. The application of the multiple element guidance requires subjective determinations, and requires the Company to make judgments about the individual deliverables and whether such deliverables are separable from the other aspects of the contractual relationship. Deliverables are considered separate units of accounting provided that:
(1) the delivered item(s) has value to the customer on a stand-alone basis and
(2) if the arrangement includes a general right of return relative to the delivered item(s), delivery or performance of the undelivered item(s) is considered probable and substantially in the Company's control.
In determining the units of accounting, the Company evaluates certain criteria, including whether the deliverables have stand-alone value, based on the consideration of the relevant facts and circumstances for each arrangement. In addition, the Company considers whether the buyer can use the other deliverable(s) for their intended purpose without the receipt of the remaining element(s), whether the value of the deliverable is dependent on the undelivered item(s), and whether there are other vendors that can provide the undelivered element(s).
Arrangement consideration that is fixed or determinable is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria. The Company determines the estimated selling price for deliverables within each agreement using vendor-specific objective evidence (“VSOE”) of selling price, if available, third-party evidence (“TPE”) of selling price if VSOE is not available, or management's best estimate of the selling price (“BESP”) if neither VSOE nor TPE is available. Determining the BESP for a unit of accounting requires significant judgment. In developing the BESP for a unit of accounting, the Company considers applicable market conditions and relevant entity-specific factors, including factors that were contemplated in negotiating the agreement with the customer and estimated costs.
If there are deliverables in an arrangement that are not separable from other aspects of the contractual relationship, they are treated as a combined unit of accounting, with the allocated revenue for the combined unit recognized in a manner consistent with the revenue recognition applicable to the final deliverable in the combined unit.
Amounts received prior to satisfying all relevant revenue recognition criteria are recorded as deferred revenue in the consolidated balance sheets and recognized as revenue when the related revenue recognition criteria are met. Amounts not expected to be recognized as revenue within the next twelve months of the consolidated balance sheet date are classified as long-term deferred revenue.
The Company recognizes revenue contingent upon the achievement of a milestone in its entirety in the period in which the milestone is achieved only if the milestone meets all the criteria to be considered substantive. At the inception of each arrangement that includes milestone payments, the Company evaluates each contingent payment on an individual basis to determine whether they are considered substantive milestones, specifically reviewing factors such as the degree of certainty in achieving the milestone, the research and development risk and other risks that must be overcome to achieve the milestone, as well as the level of effort and investment required and whether the milestone consideration is reasonable relative to all deliverables and payment terms in the arrangement. This evaluation includes an assessment of whether (a) the consideration is commensurate with either (1) the entity’s performance to achieve the milestone, or (2) the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting from the entity’s performance to achieve the milestone, (b) the consideration relates solely to past performance and (c) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement.
8
Milestone payments which are non-refundable and deemed substantive, non-creditable and contingent on achieving certain development, regulatory, or commercial milestones are typically recognized as revenues either on achievement of such milestones or over the period the Company has continuing substantive performance obligations. The Company recognizes revenue associated with the non-substantive milestones upon achievement of the milestone if there are no undelivered elements and the Company has no remaining performance obligations. Revenues from commercial milestone payments are recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met.
In the event that an agreement is terminated and the Company then has no further performance obligations, the Company recognizes as revenue any amounts that had not previously been recorded as revenue but were classified as deferred revenue at the date of such termination.
Cash considerations (including a sales incentive) given by the Company to a licensee/collaborator/customer is presumed to be a reduction of the selling prices of the Company’s products or services and is recognized as a reduction of revenue unless both of the following conditions are met:
a. The Company receives, or will receive, an identifiable benefit (goods or services) in exchange for the consideration. In order to meet this condition, the identified benefit must be sufficiently separable from the recipient’s purchase of the Company’s products such that the Company could have entered into an exchange transaction with a party other than a purchaser of its products or services in order to receive that benefit.
b. The Company can reasonably estimate the fair value of the benefit identified under the preceding condition. If the amount of consideration paid by the Company exceeds the estimated fair value of the benefit received, that excess amount shall be characterized as a reduction of revenue when recognized in the Company’s income statement.
If both conditions are met, the cash consideration is recognized as a cost incurred.
Pre-Launch Inventory
The Company may scale-up and make commercial quantities of certain of its product candidates prior to the date it anticipates that such products will receive final FDA/USDA approval. The scale-up and commercial production of pre-launch inventories involves the risk that such products may not be approved for marketing by the FDA/USDA on a timely basis, or ever. Inventory costs associated with product candidates that have not yet received regulatory approval are capitalized if the Company believes there is probable future commercial use and future economic benefit. If the probability of future commercial use and future economic benefit cannot be reasonably determined, then pre-launch inventory costs associated with such product candidates are expensed as research and development expense during the period the costs are incurred. Specifically, the Company has determined that for FDA-regulated product candidates there is a probable future commercial use and future economic benefit upon the receipt of the three major technical section complete letters from the FDA’s Center for Veterinary Medicine (“CVM”). For USDA product candidates, the Company has determined there is a probable future commercial use and future economic benefit upon the receipt of a conditional license from the USDA’s Center for Veterinary Biologics. The Company makes at least quarterly reassessments of the probability of regulatory approval and useful life of the pre-launch inventory, and determines whether such inventory continues to have a probable future economic benefit.
Property and Equipment, net
Property and equipment is recorded at historical cost, net of accumulated depreciation and amortization of $885$1,378 and $430$920 as of SeptemberJune 30, 2016,2017 and December 31, 2015,2016, respectively.
Intangible Assets, net
Beginning in the third quarter of 2016, the Company’s amortized intangible assets of intellectual property rights for currently marketed products include approval/post-approval milestone payments made under the Company’s license agreements. Amortization of intangible assets with finite lives is recognized over the time the intangible assets are estimated to contribute to future cash flows. The Company amortizes finite-lived intangible assets using the straight-line method as revenues cannot be reasonably estimated.
97
New Accounting Standards
Revenue from Contracts with Customers
In May 2014, the Financial Accounting Standards Board (“FASB”) issued guidance on recognizing revenue in contracts with customers. The guidance affects any entity that either enters into contracts with customers to transfer goods or services or enters into contracts for the transfer of nonfinancial assets unless those contracts are within the scope of other standards (e.g., insurance contracts or lease contracts). This guidance will supersede the revenue recognition requirements in topic,Revenue Recognition, and most industry-specific guidance. This guidance also supersedes certain cost guidance included in subtopic, Revenue Recognition – Construction-Type and Production-Type Contracts. In addition, the existing requirements for the recognition of a gain or loss on the transfer of nonfinancial assets that are not in a contract with a customer (e.g., assets within the scope of topic, Property, Plant, and Equipment, and tangible assets within the scope of topic, Intangibles – Goodwill and Other) are amended to be consistent with the guidance on recognition and measurement (including the constraint on revenue) in this guidance.
The core principle of the guidance is that an entity should recognize revenue to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services.
In July 2015, the FASB approved a one-year delay in the effective date of the new revenue standard. These changes become effective for the Company on January 1, 2018. Early adoption is permitted but not before the original effective date of January 1, 2017. The standard permits the use of either the retrospective or cumulative effect transition method. The Company is currently assessing the method of adoption and the impact this new guidance will have on its consolidated financial statements.
Presentation The timing of Financial Statements – Going Concern
In August 2014,revenue recognition for variable consideration under the FASB issued guidance on management’s responsibility to evaluate, at each annualCompany’s licensing and interim reporting period, whether there are conditions or events that raise substantial doubt about the entity’s ability to continuecollaboration agreements may be different as a going concern within one year after the date the financial statements are issued and to provide related disclosures. The guidance requires management to assess an entity’s ability to continue as a going concern by incorporating and expanding upon certain principles that are currently in U.S. auditing standards. This guidance is effective for the annual period ending after December 15, 2016, and for annual periods and interim periods thereafter. Early adoption is permitted.result of this new guidance. The Company does not expect that this new guidance will have a material impact onis reviewing its consolidated financial statements.licensing and collaboration agreements for variable consideration, and if any such consideration exists, whether it should be estimated and recognized earlier than under the current revenue guidance.
Inventory
In July 2015, the FASB issued guidance that requires entities to measure most inventory “at lower of cost and net realizable value” thereby simplifying the current guidance under which an entity must measure inventory at the lower of cost or market. This guidance is effective for financial statements issued for fiscal years beginning after December 15, 2016, and interim periods within those fiscal years. Early adoption is permitted and is to be applied using a prospective basis. The Company doesadopted this guidance on January 1, 2017, and the adoption did not expect that this new guidance will have a material impact on its consolidated financial statements.
Leases
In February 2016, the FASB issued guidance that requires, for operating leases, a lessee to recognize a right-of-use asset and a lease liability, initially measured at the present value of the lease payments, in its balance sheet. The standard also requires a lessee to recognize a single lease cost, calculated so that the cost of the lease is allocated over the lease term, on a generally straight-line basis. This guidance is effective for financial statements issued for fiscal years beginning after December 15, 2018, and interim periods within those fiscal years. Early adoption is permitted and is to be applied using a modified retrospective transition method. The Company is currently assessing the effect that adoption of this guidance will have on its consolidated financial statements.
Compensation – Stock Compensation
In March 2016, the FASB issued guidance that simplifies several aspects of the accounting for employee share-based payment transactions including accounting for income taxes, forfeitures and statutory tax withholding requirements, as well as classification in the statement of cash flows. This guidance is effective for financial statements issued for fiscal years beginning after December 15, 2016, and interim periods within those fiscal years. Early adoption is permitted. The Company is currently assessing the effect that adoption ofadopted this guidance willon January 1, 2017, and the adoption did not have a material impact on its consolidated financial statements.
Statement of Cash Flows
In August 2016, the FASB issued guidance on how certain cash receipts and cash payments are presented and classified in the statement of cash flows. This guidance addresses eight specific cash flow issues with the objective of reducing the existing diversity in practice. This guidance is effective for financial statements issued for fiscal years beginning after December 15, 2017, and interim periods within those fiscal years. Early adoption is permitted, provided that all of the amendments are adopted in the same period. The guidance requires application using a retrospective transition method. The Company doesadopted this guidance on January 1, 2017, and the adoption did not expect that this new guidance will have a material impact on its consolidated financial statements.
Intangibles—Goodwill and Other
In January 2017, the FASB issued guidance on simplifying the subsequent measurement of goodwill by eliminating Step 2 (measuring a goodwill impairment loss by comparing the implied fair value of a reporting unit’s goodwill with the carrying amount of that goodwill) from the goodwill impairment test. Under the amendments in this guidance, an entity should perform its annual, or interim, goodwill impairment test by comparing the fair value of a reporting unit with its carrying amount. An entity should recognize an impairment charge for the amount by which the carrying amount exceeds the reporting unit’s fair value; however, the loss recognized should not exceed the total amount of goodwill allocated to that reporting unit. Additionally, an entity should consider income tax effects from any tax deductible goodwill on the carrying amount of the reporting unit when measuring the goodwill impairment loss, if applicable. This guidance is effective for annual or interim goodwill impairment tests in fiscal years beginning after December 15, 2019. Early adoption is permitted for interim or annual goodwill impairment tests performed on testing dates after January 1, 2017. The guidance requires application using a prospective method. The Company adopted this guidance on January 1, 2017, and the adoption did not have a material impact on its consolidated financial statements.
108
Compensation – Stock Compensation
In May 2017, the FASB issued guidance on determining which changes to the terms or conditions of share-based payment awards require an entity to apply modification accounting. This guidance is effective for financial statements issued for fiscal years beginning after December 15, 2017, and interim periods within those fiscal years. Early adoption is permitted, and is applied prospectively to changes in terms or conditions of awards occurring on or after the adoption date. The Company is currently assessing the effect that adoption of this guidance will have on its consolidated financial statements.
2. Fair Value of Financial Assets and Liabilities
Financial Assets and Liabilities Measured at Fair Value on a Recurring Basis
The following financial assets are measured at fair value on a recurring basis using quoted prices in active markets for identical assets (Level 1); significant other observable inputs (Level 2); and significant unobservable inputs (Level 3).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fair Value Measurements as of |
|
|
|
| Fair Value Measurements as of | ||||||||||||||||||||
|
| Carrying |
| September 30, 2016 Using: |
| Carrying |
| June 30, 2017 Using: | ||||||||||||||||||||||
|
| Value |
| Level 1 |
| Level 2 |
| Level 3 |
| Total |
| Value |
| Level 1 |
| Level 2 |
| Level 3 |
| Total | ||||||||||
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash equivalents: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Certificates of deposit |
| $ | 7,221 |
| $ | — |
| $ | 7,221 |
| $ | — |
| $ | 7,221 |
| $ | 7,221 |
| $ | — |
| $ | 7,221 |
| $ | — |
| $ | 7,221 |
Short-term investments: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Short-term marketable securities - certificates of deposit |
|
| 1,494 |
|
| — |
|
| 1,494 |
|
| — |
|
| 1,494 |
|
| 1,494 |
|
| — |
|
| 1,494 |
|
| — |
|
| 1,494 |
|
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fair Value Measurements as of |
|
|
|
| Fair Value Measurements as of | ||||||||||||||||||||
|
| Carrying |
| December 31, 2015 Using: |
| Carrying |
| December 31, 2016 Using: | ||||||||||||||||||||||
|
| Value |
| Level 1 |
| Level 2 |
| Level 3 |
| Total |
| Value |
| Level 1 |
| Level 2 |
| Level 3 |
| Total | ||||||||||
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash equivalents: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Certificates of deposit |
| $ | 6,972 |
| $ | — |
| $ | 6,972 |
| $ | — |
| $ | 6,972 |
| $ | 7,719 |
| $ | — |
| $ | 7,719 |
| $ | — |
| $ | 7,719 |
Money market fund |
|
| 35 |
|
| 35 |
|
| — |
|
| — |
|
| 35 | |||||||||||||||
Short-term investments: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Short-term marketable securities - certificates of deposit |
|
| 747 |
|
| — |
|
| 747 |
|
| — |
|
| 747 |
|
| 996 |
|
| — |
|
| 996 |
|
| — |
|
| 996 |
Reverse repurchase agreements |
|
| 58,700 |
|
| — |
|
| 58,700 |
|
| — |
|
| 58,700 | |||||||||||||||
|
| $ | 66,454 |
| $ | 35 |
| $ | 66,419 |
| $ | — |
| $ | 66,454 |
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
| $ | — |
| $ | 8,715 |
Certain estimates and judgments are required to develop the fair value amounts shown above. The fair value amounts shown above are not necessarily indicative of the amounts that the Company would realize upon disposition, nor do they indicate the Company’s intent or ability to dispose of the financial instrument.
The following methods and assumptions were used to estimate the fair value of each material class of financial instrument:
| ||||
| Cash equivalents – the fair value of the cash equivalents has been determined to be amortized cost given the short duration of the securities. | |||
| Marketable securities (short-term) – the fair value of marketable securities has been determined to be amortized cost given the short duration of the securities. | |||
|
|
|
|
Financial Assets and Liabilities that are not Measured at Fair Value on a Recurring Basis
The carrying amounts and estimated fair value of the Company’s financial liabilities which are not measured at fair value on a recurring basis was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| September 30, 2016 |
| June 30, 2017 | ||||||||
|
| Carrying Value |
| Fair Value |
| Carrying Value |
| Fair Value | ||||
Liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Loans payable (Level 2) |
| $ | 40,068 |
| $ | 39,968 |
| $ | 38,093 |
| $ | 38,172 |
119
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| December 31, 2015 |
| December 31, 2016 | ||||||||
|
| Carrying Value |
| Fair Value |
| Carrying Value |
| Fair Value | ||||
Liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Loans payable (Level 2) |
| $ | 39,710 |
| $ | 40,569 |
| $ | 40,188 |
| $ | 40,709 |
Loans payable values above include both the current and the long-term loans balances as of June 30, 2017 and December 31, 2016.
Certain estimates and judgments were required to develop the fair value amounts. The fair value amount shown above is not necessarily indicative of the amounts that the Company would realize upon disposition, nor does it indicate the Company’s intent or ability to dispose of the financial instrument.
The fair value of loans payable was estimated using discounted cash flow analysis discounted at current rates.
Fair value information about the intangible assets that were fully impaired during the nine months ended September 30, 2016 (Note 7) was as follows:
| |||||||||||||||
|
| ||||||||||||||
|
|
|
|
| |||||||||||
|
|
|
|
|
|
|
|
|
| ||||||
|
|
|
|
| |||||||||||
|
|
|
|
|
|
|
|
| |||||||
The fair value amount is presented as of the date of impairment, as these assets are not measured at fair value on a recurring basis (Note 7). The fair value reflects intangible assets written down to fair value during the nine months ended September 30, 2016. Fair value was determined using the income approach, specifically, the multi-period excess earnings method, a form of a discounted cash flow method. The Company started with a forecast of all the expected net cash flows associated with the asset and then it applied an asset-specific discount rate to arrive at a net present value amount. Some of the more significant estimates and assumptions inherent in this approach include: the amount and timing of the projected net cash flows, which includes the expected impact of competitive legal and/or regulatory forces on the product and the impact of technological risk associated with IPR&D assets; the discount rate, which seeks to reflect the various risks inherent in the projected cash flows; and the tax rate, which seeks to incorporate the geographic diversity of the projected cash flows.
3. Investments
Marketable Securities
Marketable securities consisted of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| June 30, 2017 | ||||||||||
|
|
|
|
| Gross |
| Gross |
|
|
| ||
|
| Amortized |
| Unrealized |
| Unrealized |
| Fair | ||||
|
| Cost |
| Losses |
| Losses |
| Value | ||||
Short-term marketable securities: |
|
|
|
|
|
|
|
|
|
|
|
|
Certificates of deposit |
| $ | 1,494 |
| $ | — |
| $ | — |
| $ | 1,494 |
Total |
| $ | 1,494 |
| $ | — |
| $ | — |
| $ | 1,494 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| September 30, 2016 |
| December 31, 2016 | ||||||||||||||||||||
|
|
|
|
| Gross |
| Gross |
|
|
|
|
|
|
| Gross |
| Gross |
|
|
| ||||
|
| Amortized |
| Unrealized |
| Unrealized |
| Fair |
| Amortized |
| Unrealized |
| Unrealized |
| Fair | ||||||||
|
| Cost |
| Losses |
| Losses |
| Value |
| Cost |
| Losses |
| Losses |
| Value | ||||||||
Short-term marketable securities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Certificates of deposit |
| $ | 1,494 |
| $ | — |
| $ | — |
| $ | 1,494 |
| $ | 996 |
| $ | — |
| $ | — |
| $ | 996 |
Total |
| $ | 1,494 |
| $ | — |
| $ | — |
| $ | 1,494 |
| $ | 996 |
| $ | — |
| $ | — |
| $ | 996 |
At SeptemberJune 30, 2017 and December 31, 2016, short-term marketable securities consisted of investments that mature within one year. Short-term marketable securities are recorded as short-term investments in the consolidated balance sheets.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| December 31, 2015 | ||||||||||
|
|
|
|
| Gross |
| Gross |
|
|
| ||
|
| Amortized |
| Unrealized |
| Unrealized |
| Fair | ||||
|
| Cost |
| Losses |
| Losses |
| Value | ||||
Short-term marketable securities: |
|
|
|
|
|
|
|
|
|
|
|
|
Certificates of deposit |
| $ | 747 |
| $ | — |
| $ | — |
| $ | 747 |
Total |
| $ | 747 |
| $ | — |
| $ | — |
| $ | 747 |
12
At December 31, 2015, short-term marketable securities consisted of investments that mature within one year. Short-term marketable securities are recorded as short-term investments in the consolidated balance sheets.
Reverse Repurchase Agreements
The Company, as part of its cash management strategy, may invest excess cash in reverse repurchase agreements. All reverse repurchase agreements are tri-party and have maturities of three months or less at the time of investment. The underlying collateral is U.S. government securities including U.S. treasuries, agency debt and agency mortgage securities. The underlying collateral posted by each counterparty is required to cover 102% of the principal amount and accrued interest after the application of a discount to fair value. The Company was not invested in any reverse repurchase agreements as of September 30, 2016.
4. Derivative Financial Instruments
In 2015, the Company’s derivative financial instrument, the warrant the Company received in connection with the license agreement with Advaxis, Inc. (“Advaxis”), was not designated as a hedging instrument and was adjusted to fair value through earnings in other income (expense). During the year ended December 31, 2015, the Company exercised the Advaxis warrant and subsequently sold the shares of common stock received upon exercise.
The following table shows the Company’s gain recognized in other income (expense) for the three and nine months ended:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gain Recognized in |
| Gain Recognized in | ||||||||
|
| Other Income (expense) |
| Other Income (expense) | ||||||||
|
| Three Months Ended |
| Nine Months Ended | ||||||||
|
| September 30, |
| September 30, | ||||||||
|
| 2016 |
| 2015 |
| 2016 |
| 2015 | ||||
Derivative assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Warrant |
| $ | — |
| $ | — |
| $ | — |
| $ | 1,274 |
As the Company exercised the warrant and subsequently sold the shares of common stock received upon exercise during the second quarter of 2015, no gain was recorded during the three and nine months ended September 30, 2016.
Inventories are stated at the lower of cost or marketnet realizable value and consisted of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| September 30, 2016 |
| December 31, 2015 |
| June 30, 2017 |
| December 31, 2016 | ||||
Raw materials |
| $ | 88 |
| $ | 120 |
| $ | 1,491 |
| $ | 1,441 |
Work-in-process |
|
| 5,270 |
|
| 441 |
|
| 5,199 |
|
| 8,153 |
Finished goods |
|
| 217 |
|
| 745 |
|
| 271 |
|
| 1,536 |
|
| $ | 5,575 |
| $ | 1,306 |
| $ | 6,961 |
| $ | 11,130 |
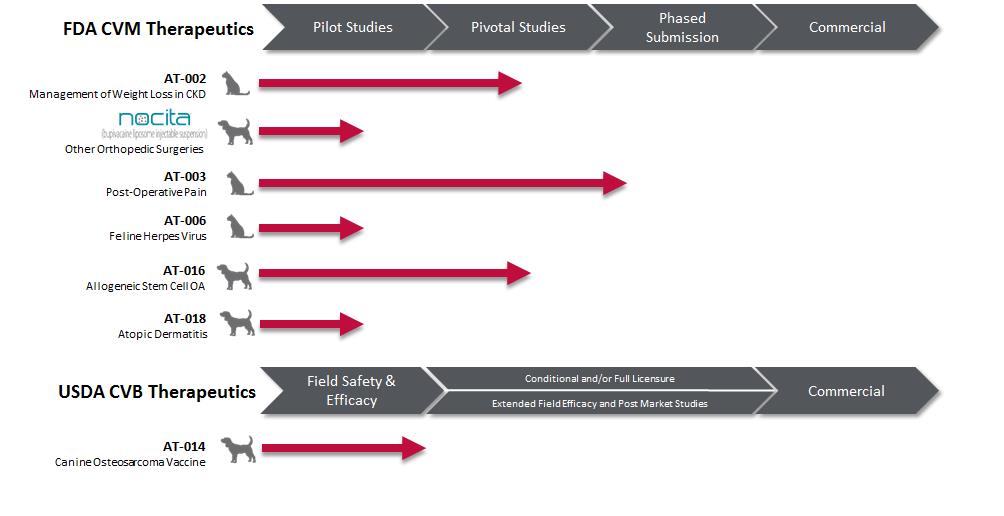
Work-in-process inventories at September 30, 2016, included $4,774 of pre-launch product costs of GALLIPRANT® (grapiprant tablets), ENTYCE® (capromorelin oral solution) and NOCITA® (bupivacaine liposome injectable suspension) capitalized due to anticipated benefit from future commercialization of these products. GALLIPRANT was approved by the CVM for the control of pain and inflammation associated with osteoarthritis in dogs in the first quarter of 2016, ENTYCE was approved by the CVM for appetite stimulation in dogs in the second quarter of 2016, and NOCITA was approved by the CVM as a local post-operative analgesia for cranial cruciate ligament surgery in dogs in the third quarter of 2016 and was made commercially available to veterinarians in the United States in October 2016. As of SeptemberJune 30, 2017and December 31, 2016, the Company had non-cancellable open orders for the purchase of inventories of approximately $21,800.$27,667 and $17,800, respectively.
During the threeAs of June 30, 2017 and nine months ended September 30,December 31, 2016, the Company recognized inventory valuation adjustment losseshad deposits for inventories of $5,313 and $0, respectively, recorded as prepaid expenses and other current assets in the amount of $111 and $1,664, respectively, from application of lower of cost or market, in cost of product sales. The losses related to TACTRESS™ inventories that were written off and pre-launch GALLIPRANT inventories marked to market due to terms agreed upon in the Elanco collaboration agreement (Note 10).
consolidated balance sheets.
Goodwill is recorded as an indefinite-lived asset and is not amortized for financial reporting purposes but is tested for impairment on an annual basis or when indications of impairment exist. No goodwill impairment losses have been recognized to date. Goodwill is not expected to be deductible for income tax purposes. The Company performs its annual impairment test of the carrying value of the Company’s goodwill during the third quarter of each year.
1310
6. Goodwill
The Company completed its annual goodwill impairment testing during the third quarter of 2016. The Company elected to bypass the qualitative assessment. The Company determined as of the testing date that it consisted of one operating segment, which is comprised of one reporting unit. In performing step one of the assessment, the Company determined that its fair value, determined to be its market capitalization, was greater than its carrying value, determined to be stockholders’ equity. Based on this result, step two of the assessment was not required to be performed, and the Company determined there was no impairment of goodwill during the third quarter of 2016.
Goodwill as of SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gross |
| Impairment |
| Net | |||
|
| Carrying Amount |
| Losses |
| Carrying Value | |||
Goodwill |
| $ | 40,275 |
| $ | — |
| $ | 40,275 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gross |
| Impairment |
| Net | |||
|
| Carrying Value |
| Losses |
| Carrying Value | |||
Goodwill |
| $ | 40,500 |
| $ | — |
| $ | 40,500 |
The change in the net book value of goodwill for the ninesix months ended SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
| |
As of January 1, |
| $ | |
Effect of foreign currency exchange |
|
| |
As of the end of the period, |
| $ | |
7.6. Intangible Assets, Net
The change in the net book value of intangible assets for the ninesix months ended SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
| |
As of January 1, |
| $ | |
Additions (Note 9) |
|
| |
Amortization expense |
|
| |
Effect of foreign currency exchange |
|
| |
| |||
As of the end of the period, |
| $ | |
The estimated useful lives of the individual categories of intangible assets were based on the nature of the applicable intangible asset and the expected future cash flows to be derived from the intangible asset. Amortization of intangible assets with finite lives is recognized over the shorter of the respective term of the agreement or the period of time the intangible assets are expected to contribute to future cash flows. The Company amortizes finite-lived intangible assets using the straight-line method. The Company recognized amortization expense of $91$86 and $281$150 for the three and ninesix months ended SeptemberJune 30, 2016,2017, respectively, and $483$95 and $1,449$190 for the three and ninesix months ended SeptemberJune 30, 2015,2016, respectively. Indefinite-lived in-process research and development (“IPR&D”) intangible assets are not amortized until a product reaches its first conditional license or approval, and then they are amortized over their estimated useful lives.
Unamortized Intangible Assets
Unamortized intangible assets as of SeptemberJune 30, 2016,2017, were as follows:
|
|
|
|
|
|
|
|
|
| Net | |
|
| Carrying | |
|
| Value | |
| |||
Intellectual property rights acquired for in-process research and development |
| $ | |
The net carrying value above includes asset impairment charges to date of $16,765.
14
Return of AT-006 Global Rights
On May 11, 2016, the Company and Elanco agreed to terminate the Exclusive License, Development, and Commercialization Agreement with Elanco (the “Elanco AT-006 Agreement”) (Note 10) that granted Elanco global rights for development and commercialization of licensed animal health products for an anti-viral for the treatment of feline herpes virus induced ophthalmic conditions. As a result of the termination of the Elanco AT-006 Agreement, the Company conducted an impairment assessment of the AT-006 intangible asset to assess the impact of the termination agreement. As part of the assessment, the Company considered development timing and expenses, manufacturing expenses, technology royalties and royalties due to Elanco, anticipated timing of commercial availability, as well as marketing, and selling expenses to commercialize the product. The Company concluded that the AT-006 intangible asset was not impaired due to the termination of the Elanco AT-006 Agreement.
Impairment of UnamortizedAmortized Intangible Assets
AT-007 (Feline immunodeficiency virus)
The Company has been considering out-licensing or internally advancing the AT-007 program for feline immunodeficiency virus since an impairment expense was recorded in the third quarterAmortized intangible assets as of 2015. Due to the return of the AT-006 global rights from Elanco in May 2016 (Note 10) and ensuing development program portfolio prioritization, including consideration of the Company’s focus on commercial launch activities to support its recently approved products, the Company decided to discontinue the development of AT-007 during the second quarter of 2016. This resulted in an impairment charge of $2,229, which was recorded during the three months ended June 30, 2016, reducing the carrying value of AT-0072017, were as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gross |
|
|
|
| Net |
| Weighted | |||
|
| Carrying |
| Accumulated |
| Carrying |
| Average | ||||
|
| Value |
| Amortization |
| Value |
| Useful Life | ||||
Intellectual property rights for currently marketed products |
| $ | 42,652 |
| $ | 38,836 |
| $ | 3,816 |
| 11.7 | Years |
Accumulated amortization above includes both amortization expense and asset impairment charges. Asset impairment charges to $0.
date are $34,575. Unfavorable outcomes of the Company’s development activities or the Company’s estimates of the market opportunities for the producttherapeutic candidates could result in additional impairment charges in future periods.
Amortized Intangible Assets
Amortized intangible assets as of September 30, 2016, were as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gross |
|
|
|
| Net |
| Weighted | |||
|
| Carrying |
| Accumulated |
| Carrying |
| Average | ||||
|
| Value |
| Amortization |
| Value |
| Useful Life | ||||
Intellectual property rights for currently marketed products |
| $ | 29,572 |
| $ | 23,348 |
| $ | 6,224 |
| 19.73 | Years |
Accumulated amortization includes both amortization expense and asset impairment charges. Asset impairment charges to date are $20,228.
Impairment of Amortized Intangible Assets
TACTRESS
Since the acquisition of Vet Therapeutics, Inc. (October 2013), the Company has been performing various scientific and clinical activities to gain further knowledge around the science and efficacy of TACTRESS. In the third quarter of 2015, the Company’s interim analysis of the clinical results indicated that TACTRESS did not seem to be adding significant progression free survival in canine T-cell lymphoma; those results were confirmed in the final study results in July 2016. In addition, scientific studies suggested that TACTRESS was not as specific to the target as expected. Given those clinical and scientific results, the Company no longer believed that TACTRESS would capture the desired T-cell lymphoma market opportunity and recorded an impairment expense.
While TACTRESS remains commercially available, the use by oncologists has been more limited than the Company anticipated, resulting in sales during the quarter ended June 30, 2016, being significantly lower than forecasted. The Company deemed the events and market projections described above to be indicators of potential impairment of its finite-lived intangible asset TACTRESS during the second quarter of 2016. The Company performed impairment testing for the intangible asset TACTRESS as of June 30, 2016, and recorded an impairment expense of $551 during the second quarter of 2016, resulting in a net carrying value of $0 for TACTRESS.
Unfavorable outcomes of the Company’s development activities or the Company’s estimates of the market opportunities for the product candidates could result in additional impairment charges in future periods. For example, we anticipate the results of the BLONTRESS post-approval study, Mini B-CHOMP, in late 2016.
15
8.7. Debt
Loan and Security Agreements
Effective as of October 16, 2015, the Company and Vet Therapeutics, Inc., (the “borrowers”“Borrowers”), entered into a Loan and Security Agreement, as amended on February 24, 2017 (“Loan Agreement”), with Pacific Western Bank, or Pacific Western, as a collateral agent and Oxford Finance, LLC (the “Lenders”). The loan is secured by substantially all of the borrowers’Borrowers’ personal property other than intellectual property.property and certain other customary exclusions. Subject to customary exceptions, the Company is not permitted to encumber its intellectual property. The outstanding principal balance under the Loan Agreement was $35,000$32,667 under the term loan facility and $5,000 under the revolving credit facility at SeptemberJune 30, 2016.2017. Under the Loan Agreement, the Company was required to make interest-only payments on the term loan for 18 months, and beginning on May 1, 2017, began to make payments of principal and accrued interest on the term loan in equal monthly installments over a term of 30 months. The Company was required to make interest-only payments on the revolving credit facility until October 16, 2017, when all principal and accrued interest rate, which iswere due. The
11
term loan and revolving credit facility bear interest per annum at the greater of (i) 6.91% or (ii) 3.66% plus the prime rate, onwhich is customarily defined. As of June 30, 2017, the interest rate for the term loan and the revolving credit facility as of September 30, 2016, was 7.16%7.91%. DuringDuring the three and ninesix months ended SeptemberJune 30, 2017, the Company recognized interest expense of $871 and $1,731, respectively, and during the three and six months ended June 30, 2016, the Company recognized interest expense of $851$843 and $2,538, respectively, and during the three and nine months ended September 30, 2015, the Company recognized interest expense of $221 and $655,$1,687, respectively.
The Loan Agreement contains customary representations and warranties and customary affirmative and negative covenants, including, among others, limits or restrictions on the borrowers’Borrowers’ ability to incur liens, incur indebtedness, make certain restricted payments, make certain investments, merge, consolidate, make an acquisition, enter into certain licensing arrangements and dispose of certain assets. In addition, the Loan Agreement contains customary events of default that entitle the Lenders to cause the borrowers’Borrowers’ indebtedness under the Loan Agreement to become immediately due and payable. The events of default, some of which are subject to cure periods, include, among others, a non-payment default, a covenant default, the occurrence of a material adverse change, the occurrence of an insolvency, a material judgment default, defaults regarding other indebtedness and certain actions by governmental authorities. Upon the occurrence and for the duration of an event of default, an additional default interest rate equal to 4% per annum will apply to all obligations owed under the Loan Agreement.
The Loan Agreement requires that the Company receive unrestricted net cash proceeds of at least $45,000 from partnering transactions and/or the issuance of equity securities from October 16, 2015 to October 16, 2016. The Loan Agreement also requires that the Company has at least three products fully USDA- or FDA-approved for commercialization by December 31, 2016. With the FDA approval of GALLIPRANT in March 2016 and the receipt of the upfront payment of $45,000 under the Elanco collaboration agreement (Note 10) entered into in April 2016, the Company has met both conditions. Additionally, the Loan Agreement requires that the Company maintain certain minimum liquidity at all times (the greater of cash equal to fifty percent (50%) of outstanding credit extensions or remaining months’ liquidity, which is calculated on an average trailing three (3) month basis, equal to six (6) months or greater), which as of SeptemberJune 30, 2016,2017, was approximately $38,600. At September 30, 2016, the Company was in compliance with all financial covenants.$22,523. If the minimum liquidity covenant is not met, the Company may be required to repay the term loan and the revolving credit facility prior to their scheduled maturity date.dates. At June 30, 2017, the Company was in compliance with all financial covenants.
LoansThe Company’s loans payable balance as of SeptemberJune 30, 2016, were2017, was as follows:
|
|
|
|
Principal amounts |
|
|
|
Term loan, |
| $ | |
Revolving |
|
| 5,000 |
Add: accretion of final payment and termination fees |
|
| |
Less: unamortized debt issuance costs |
|
| |
As of the end of the period |
| $ |
As of SeptemberJune 30, 2016, $5,8332017, $8,667 and $130 related to the term loan was reclassifiedand the revolving credit facility, respectively, were classified as Currentcurrent portion – loans payable. Portions of the term loan and the revolving credit facility have been reclassified from current portion – loans payable to loans payable as the Company refinanced its debt after the balance sheet date.
Effective as of July 31, 2017, the Borrowers and Lenders entered into a second amendment to the Loan Agreement (“the Second Amendment”). The terms of the Second Amendment, among other things, extend the maturity date of the existing revolving credit facility to October 16, 2019 (the “Revolving Line Maturity Date”), with amortized equal repayments of the principal outstanding under the revolving credit facility beginning November 1, 2018, and provide a six-month interest only period for the term loans, starting on the date of the Second Amendment. The Company is not subject to any new financial covenants as a result of the Second Amendment. At the closing of the Second Amendment, the Company paid the Lenders an amendment fee of $150 and a facility fee of $60. The Company is also obligated to pay a new termination fee equal to $165 upon the earliest to occur of the Revolving Line Maturity Date, the acceleration of the revolving credit facility or the termination of the revolving credit facility. The existing termination fee of $165 will continue to be payable upon the earliest to occur of the original revolving maturity date (October 16, 2017), the acceleration of the revolving credit facility or the termination of the revolving credit facility.
9.8. Accrued Expenses
Accrued expenses consisted of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| September 30, 2016 |
| December 31, 2015 |
| June 30, 2017 |
| December 31, 2016 | ||||
Accrued expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
Payroll and related expenses |
| $ | 1,944 |
| $ | 1,922 |
| $ | 1,489 |
| $ | 2,321 |
Professional fees |
|
| 226 |
|
| 388 |
|
| 236 |
|
| 219 |
Royalty expense |
|
| 53 |
|
| 1 |
|
| 370 |
|
| 71 |
Interest expense |
|
| 239 |
|
| 238 |
|
| 240 |
|
| 247 |
Research and development costs |
|
| 594 |
|
| 1,111 |
|
| 511 |
|
| 364 |
Unbilled inventories |
|
| — |
|
| 465 | ||||||
Accrued loss on a firm purchase commitment |
|
| 1,983 |
|
| 1,983 | ||||||
Milestone |
|
| 176 |
|
| 500 |
|
| 481 |
|
| 17 |
Other |
|
| 222 |
|
| 87 |
|
| 292 |
|
| 140 |
Total accrued expenses |
| $ | 3,454 |
| $ | 4,247 | ||||||
Total |
| $ | 5,602 |
| $ | 5,827 | ||||||
1612
10.9. Agreements
RaQualia Pharma Inc. (“RaQualia”)
On December 27, 2010, the Company entered into two Exclusive License Agreements with RaQualia (the(as amended, the “RaQualia Agreements”) that granted the Company global rights, subject to certain exceptions for injectables in Japan, Korea, China and Taiwan for development and commercialization of licensed animal health products for compounds RQ-00000005 (ENTYCE®, also known as AT-002) and RQ-00000007 (GALLIPRANT®, also known as AT-001). The Company will be required to pay RaQualia remaining milestone payments associated with GALLIPRANT and ENTYCE of up to $7,000$4,000 and $6,500,$6,000, respectively, upon the Company’s achievement of certain development, regulatory and commercial milestones, as well as mid-single digit royalties on the Company’s or the Company’s sublicensee’s product sales, if any.sales.
The Company achieved milestones totaling $0 and $5,000 which were expensed within research and development expensesa $3,000 milestone during the three and ninesix months ended SeptemberJune 30, 2016, respectively.2017, which was paid and capitalized as an intangible asset in the first quarter of 2017. As of SeptemberJune 30, 2016,2017, the Company had paid $5,000$8,500 in milestone payments and no royalty payments since execution of the RaQualia Agreements.
It is possible that multiple additional milestones related to the RaQualia Agreements are achieved within the next 12 months totaling $6,500.$3,000.
Elanco
BLONTRESS
On January 2, 2015, the Company was granted a full product license from the USDA for BLONTRESS. The approval resulted in a $3,000 milestone payment being earned and due to the Company per the terms of the Exclusive Commercial License Agreement with Elanco (formerly Novartis Animal Health, Inc.) (the “Elanco Agreement”). During the first quarter of 2015, the Company recognized $3,000 of licensing revenue related to the milestone payment.
On February 24, 2015, the Company and Elanco agreed to terminate the Elanco Agreement. In consideration for the return of the commercial license granted to Elanco, the Company paid Elanco $2,500 in March 2015, and will be required to pay an additional $500 upon the first commercial sale by the Company. At that time the Company determined that it was probable that the $500 payment will be paid, and recorded the $500 as a current liability in the first quarter of 2015. The first commercial sale occurred in March 2016. The Company recorded the $3,000 owed to Elanco as a reduction in revenues received from Elanco as the payment was to re-acquire rights that the Company had previously licensed to Elanco.
On February 25, 2016, the Company and Elanco agreed to amend the terms related to the $500 payment due upon the first commercial sale by the Company. Under the amended terms, upon the first commercial sale in March 2016, the Company is required to pay quarterly a royalty per vial sold until $500 in royalties are paid or the end of two years. After two years, the Company will be required to pay Elanco $500 plus 10% interest, compounded annually against any unpaid balance, less any royalties paid during the two years. If during the two years following the first commercial sale the Company withdraws BLONTRESS from the market and ceases all commercialization, the remaining royalty and related interest are no longer payable. As of September 30, 2016, $313 of the remaining $489 accrued milestone was included in other long-term liabilities in the consolidated balance sheets.
GALLIPRANT
On April 22, 2016, the Company entered into a Collaboration, License, Development and Commercialization Agreement (the “Collaboration Agreement”) with Eli Lilly and Company, acting on behalf of its Elanco Animal Health Division (“Elanco”) pursuant to which the Company granted Elanco rights to develop, manufacture, market and commercialize the Company’s products based on licensed grapiprant rights and technology (the “Product”), including GALLIPRANT.GALLIPRANT (collectively, “Grapiprant Products”). Pursuant to the Collaboration Agreement, Elanco will have exclusive rights globally outside the United States and co-exclusive rights with the Company in the United States during the term of the Collaboration Agreement.
Under the terms of the Collaboration Agreement, the Company received a non-refundable, non-creditable upfront payment of $45,000. The Company is entitled to a $4,000 milestone payment upon European approval of GALLIPRANTa Grapiprant Product for the treatment of pain and inflammation, another $4,000 payment upon achievement of a development milestone related to the manufacturing of GALLIPRANT,a Grapiprant Product, and payments up to $75,000 upon the achievement of certain sales milestones. The sales milestone payments are subject to a one-third reduction for each year the occurrence of the milestone is not achieved beyond December 31, 2021, with any non-occurrence beyond December 31, 2023, cancelling out the applicable milestone payment obligation entirely.
The Collaboration Agreement also provides that Elanco will pay the Company royalty payments on a percentage of net sales in the mid-single to low-double digits. The Company is responsible for all development activities required to obtain the first registration or regulatory approval for thea Grapiprant Product for use in dogs in each of the European Union (“the EU Product Registration”) and the United States, and Elanco is responsible for all other development activities. First registration for thea Grapiprant Product in the U.S.United States was achieved before the completion of the Collaboration Agreement. In addition, the Company and Elanco have agreed to pay 25% and 75%, respectively, of all third-party development fees and expenses through December 31, 2018, in connection with preclinical and clinical trials necessary for any additional registration or regulatory approval of the Grapiprant Products, provided that the Company’s contribution to such development fees and expenses is capped at $7,000 (“R&D Cap”). Commencing on the effective date of the Collaboration Agreement,
17
the Company is responsible for the manufacture and supply of all of Elanco’s reasonable requirements of GALLIPRANT under the supply terms agreed upon pursuant to the Collaboration Agreement. However, Elanco retains the ability to assume all or a portion of the manufacturing responsibility during the term of the Collaboration Agreement. The parties have agreed under the Collaboration Agreement to negotiate and enter into a supply agreement formalizing the terms of supply of active product ingredients and/or finished GALLIPRANT by the Company to Elanco.
On April 22, 2016, in connection with the Collaboration Agreement, the Company entered into a Co-Promotion Agreement (the “Co-Promotion Agreement”) with Elanco to co-promote the Product in the United States.
Under the terms of the Co-Promotion Agreement, Elanco has agreed to pay the Company, as a fee for promotional services performed and expenses incurred by the Company under the Co-Promotion Agreement, (i) 25% of the gross margin on net sales of Product sold in the United States under the Collaboration Agreement prior to December 31, 2018 (unless extended by mutual agreement), and (ii) a mid-single digit percentage of net sales of the Product in the United States after December 31, 2018 through 2028 (unless extended by mutual agreement).
The Company concluded that the Collaboration Agreement and Co-Promotion Agreement represent a multiple-element arrangement, and evaluated if deliverables in the arrangement represent separate units of accounting. The Company identified the following deliverables under the agreement: (i) a royalty-bearing, sub-licensable, development, manufacturing and commercialization license; (ii) manufacturing and supply services; (iii) participation in a joint manufacturing subcommittee; and (iv) services associated with obtaining the EU Product Registration. The Company performed an assessment and concluded that the license had stand-alone value from the other undelivered elements in the arrangement. The Company’s best estimate of the selling price for the manufacturing subcommittee and the EU Product Registration services were immaterial and, therefore, no consideration was allocated to these deliverables. Under the manufacturing and supply services terms, Elanco will be obligated to pay for any future orders at a price per unit representative of market value, and, therefore, no upfront consideration was allocated to this deliverable. The Company allocated $38,000 of the $45,000 upfront payment to the license, and recognized $38,000 of licensing and collaboration revenue during the quarter ended June 30, 2016. The Company allocated $7,000 of upfront consideration to the R&D Cap, which was recorded as licensing and collaboration commitment liability in the consolidated balance sheetsheets at SeptemberJune 30, 2017 and December 31, 2016. The Company classified the licensing and collaboration commitment liability as a current liability due to the Company having no control over when R&D Cap expenses will be incurred and the expected timing of R&D Cap expenses being unknown as of SeptemberJune 30, 2016.2017. The licensing and collaboration commitment liability will be reduced in future periods as the related expenses are incurred by Elanco and paid for by the Company. Any remaining balance not paid to Elanco will be recognized as licensing and collaboration revenue on December 31, 2018, when the Company’s obligation to fund 25% of Elanco’s development efforts expires.
The Company evaluated ifCommencing on the sales and other milestones ineffective date of the Collaboration Agreement, are substantive. Thethe Company determined thatis responsible for the milestones are non-substantive,manufacture and therefore, these milestones will be allocated amongstsupply of all of Elanco’s reasonable requirements of active pharmaceutical ingredient (“API”) and/or finished Grapiprant Products under the delivered, and any undelivered elements atsupply terms agreed upon pursuant to the timeCollaboration Agreement. However, Elanco retains the milestones are earned. If there are no undelivered elements,ability to assume all or a portion of the milestone payments will be recognized as revenue in their entirety upon achievementmanufacturing responsibility during the term of each milestone. For the three and the nine months ended September 30, 2016, no milestones were achieved, and accordingly, no revenues were recognized from the milestones.
AT-006
Collaboration Agreement. On May 11, 2016,April 28, 2017, the Company and Elanco agreed to terminate the Elanco AT-006 Agreement that granted Elanco global rights for development and commercialization of licensed animal health products for an anti-viral for the treatment of feline herpes virus induced ophthalmic conditions. In consideration for the return of the Elanco AT-006 Agreement global rights, the Company is required to pay Elanco a low single-digit royalty on any product sales, if any, up to an amount in the low-single digit millions.
Pacira Pharmaceuticals, Inc. (“Pacira”)
On December 5, 2012, the Company entered into an Exclusive License, Development, and Commercialization Agreement with Paciraamendment (the “Pacira Agreement”“Amendment”) that grantedto the Collaboration Agreement. Under the Amendment, Elanco has agreed to submit binding purchase orders to the Company, global rightswithin 15 days of the effective date of the Amendment, for developmentcertain finished Grapiprant Products to be produced from certain batches of API the Company has agreed to purchase from its third-party manufacturer (the “API Batches”). In addition, Elanco has agreed to pay the Company for the API Batches within 30 days after the Company provides Elanco with proof of payment to the manufacturer for such API Batches. The Amendment provides that, in the event Elanco provides notice of its intent to assume responsibility for manufacturing, Elanco would assume all responsibilities of the Company with respect to any undelivered API, including paying the third-party manufacturer for such undelivered API. In July 2017, pursuant to Sections 8.2.2 and commercialization10.1(c) of licensed animal health productsthe Collaboration Agreement, as amended, Elanco provided the Company notice of its intent to assume responsibility for NOCITA® (also known as AT-003).manufacturing of the Grapiprant Products and its intent to assume the applicable regulatory approvals. The Company is required to pay Pacira milestone paymentsbelieves the transfers of up to $40,000 upon the Company’s achievement of certain commercial milestones, as well as tiered royalties on the Company’s product sales, if any. The commercial milestones owed to Pacira under the Pacira Agreement begin to be triggered once NOCITA annual net sales reach $100,000 with the final tier being owed to Pacira once NOCITA annual net sales reach $500,000.
The Company achieved milestones totaling $1,000manufacturing responsibility and $2,000 during the three and nine months ended September 30, 2016, respectively. The $1,000 of milestones achieved during the third quarter of 2016 was capitalized as intangible assets and the other $1,000 was expensed within research and development expensessuch regulatory approvals in the second quarter of 2016. As of September 30, 2016, the Company had paid $2,500 in milestone payments and no royalty payments since execution of the Pacira Agreement.
United States will be completed by December 31, 2017.
1813
11.
On April 22, 2016, in connection with the Collaboration Agreement, the Company entered into a Co-Promotion Agreement (the “Co-Promotion Agreement”) with Elanco to co-promote Grapiprant Products in the United States.
Under the terms of the Co-Promotion Agreement, Elanco has agreed to pay the Company, as a fee for promotional services performed and expenses incurred by the Company under the Co-Promotion Agreement, (i) 25% of the gross margin on net sales of Grapiprant Product sold in the United States under the Collaboration Agreement prior to December 31, 2018 (unless extended by mutual agreement), and (ii) a mid-single digit percentage of net sales of the Grapiprant Product in the United States after December 31, 2018 through 2028 (unless extended by mutual agreement).
As of SeptemberJune 30, 2016,2017, there were 36,318,54142,402,677 shares of the Company’s common stock outstanding, net of 504,812584,591 shares of unvested restricted common stock.
At-the-Market Offering
On October 16, 2015, the Company entered into a Sales Agreement (“Sales Agreement”) with Barclays Capital, Inc. (“Barclays”) pursuant to which the Company maycould sell from time to time, at its option, up to an aggregate of $52,000 of shares of its common stock (the “Shares”) through Barclays, as sales agent.agent (“ATM Program”). Sales of the Shares if any, will bewere made under the Company’s previously filed and currentlythen effective Registration Statementregistration statement on Form S-3 (Reg. No. 333-197414), by means of ordinary brokers’ transactions on the NASDAQ Global Market or otherwise. Additionally, under the terms of the Sales Agreement, the Shares maycould be sold at market prices, at negotiated prices or at prices related to the prevailing market price. The Company will paypaid Barclays a commission of 2.75% of the gross proceeds from the sale of the Shares.
During the quarterthree and six months ended SeptemberJune 30, 2016,2017, the Company agreed to sell 1,629,408sold 305,372 and 546,926 Shares of which 1,416,052 Shares settled during the third quarter of 2016 for aggregate net proceeds of $12,676, of which $605 was received by$1,565 and $2,788, respectively.
On April 28, 2017, the Company after September 30, 2016. Subsequentterminated its Sales Agreement. Prior to September 30, 2016,termination, the remaining 213,356 Shares settled for aggregate net proceeds of $1,911. AsCompany sold approximately $18,000 of the date of this filing, approximately $37,000 of Shares remained$52,000 available for saleto be sold under the Sales Agreement. The Company hasterminated the Sales Agreement because it did not intend to raise additional capital through the ATM Program, and no additional shares of the Company’s common stock were sold pursuant to the Sales Agreement. The Company did not incur any termination penalties as a result of its termination of the Sales Agreement.
Registered Direct Offering
On May 3, 2017, the Company entered into a Placement Agency Agreement (“PAA”) with Barclays, pursuant to which Barclays agreed to sell any additional Shares since Septemberserve as placement agent for an offering of shares of common stock. In conjunction with the PAA, on May 3, 2017, the Company also entered into a Securities Purchase Agreement with certain investors for the sale by the Company of 5,000,000 shares of common stock at a purchase price of $5.25 per share (the “Offering”). The shares of common stock were offered and sold pursuant to the Company’s previously filed and then effective registration statement on Form S-3 (File No. 333-197414) and a related prospectus supplement. The Company agreed to pay Barclays an aggregate fee equal to 6.0% of the gross proceeds received by the Company from the Offering. The Offering closed on May 9, 2017. During the three months ended June 30, 2016.2017, the Company received aggregate net proceeds from the Offering of approximately $24,400, after deducting placement agent fees of $1,575 and offering expenses of $273.
12.11. Stock-Based Awards
2010 Equity Incentive Plan
Activity related to stock options under the 2010 Equity Incentive Plan (the “2010 Plan”) for the ninesix months ended SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighted |
|
|
|
|
|
|
|
| Weighted |
|
| ||
|
| Shares |
| Weighted |
| Average |
|
|
| Shares |
| Weighted |
| Average |
|
| ||||
|
| Issuable |
| Average |
| Remaining |
| Aggregate |
| Issuable |
| Average |
| Remaining |
| Aggregate | ||||
|
| Under |
| Exercise |
| Contractual |
| Intrinsic |
| Under |
| Exercise |
| Contractual |
| Intrinsic | ||||
|
| Options |
| Price |
| Term |
| Value |
| Options |
| Price |
| Term |
| Value | ||||
|
|
|
|
|
|
| (In Years) |
|
|
|
|
|
|
|
|
| (In Years) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Outstanding as of December 31, 2015 |
| 86,490 |
| $ | 2.95 |
| 7.09 |
| $ | 228 | ||||||||||
Outstanding as of December 31, 2016 |
| 65,931 |
| $ | 3.73 |
| 6.09 |
| $ | 228 | ||||||||||
Granted |
| — |
|
| — |
|
|
|
|
|
| — |
|
| — |
|
|
|
|
|
Exercised |
| (20,559) |
|
| 0.44 |
|
|
|
|
|
| (8,537) |
|
| 0.40 |
|
|
|
|
|
Forfeited |
| — |
|
| — |
|
|
|
|
|
| — |
|
| — |
|
|
|
|
|
Expired |
| — |
|
| — |
|
|
|
|
|
| — |
|
| — |
|
|
|
|
|
Outstanding as of September 30, 2016 |
| 65,931 |
| $ | 3.73 |
| 6.35 |
| $ | 371 | ||||||||||
Outstanding as of June 30, 2017 |
| 57,394 |
| $ | 4.22 |
| 5.62 |
| $ | 173 | ||||||||||
AsNo stock options have been granted under the 2010 Plan since the effective date of September 30, 2016, 5,516 shares of common stock granted from early exercised options are unvested and subject to repurchase. the 2013 Incentive Award Plan (the “2013 Plan”). For the ninesix months ended SeptemberJune 30, 2016,2017, the total intrinsic value of options exercised was $180$53 and the total received from stock option exercises was $9.
Activity related to restricted stock under the 2010 Plan for the nine months ended September 30, 2016, was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighted | |
|
|
|
| Average Grant | |
|
| Shares |
| Date Fair Value | |
Unvested restricted common stock as of December 31, 2015 |
| 37,078 |
| $ | 0.36 |
Issued |
| — |
|
| — |
Vested |
| (37,078) |
|
| 0.36 |
Forfeited |
| — |
|
| — |
Unvested restricted common stock as of September 30, 2016 |
| — |
| $ | — |
For the nine months ended September 30, 2016, the total fair value of restricted common stock vested was $212.
1914
exercises was $3.
2013 Incentive Award Plan
On January 1, 2017, the annual increase in the number of shares available for issuance under the 2013 Plan was determined to be 1,203,369 shares in accordance with the automatic annual increase provisions of the 2013 Plan. As of June 30, 2017, there were 1,609,078 shares available for future grant under the 2013 Plan.
Activity related to stock options under the 2013 Incentive Award Plan (the “2013 Plan”) for the ninesix months ended SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighed |
|
|
|
|
|
|
|
|
| Weighed |
|
|
|
|
| Shares |
| Weighted |
| Average |
|
|
|
| Shares |
| Weighted |
| Average |
|
|
| ||
|
| Issuable |
| Average |
| Remaining |
| Aggregate |
| Issuable |
| Average |
| Remaining |
| Aggregate | ||||
|
| Under |
| Exercise |
| Contractual |
| Intrinsic |
| Under |
| Exercise |
| Contractual |
| Intrinsic | ||||
|
| Options |
| Price |
| Term |
| Value |
| Options |
| Price |
| Term |
| Value | ||||
|
|
|
|
|
|
| (in years) |
|
|
|
|
|
|
|
|
| (in years) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Outstanding as of December 31, 2015 |
| 1,728,199 |
| $ | 16.57 |
| 8.31 |
| $ | — | ||||||||||
Outstanding as of December 31, 2016 |
| 2,251,518 |
| $ | 12.43 |
| 7.78 |
| $ | 2,261 | ||||||||||
Granted |
| 822,433 |
|
| 4.35 |
|
|
|
|
|
| 503,400 |
|
| 7.82 |
|
|
|
|
|
Exercised |
| (7,458) |
|
| 6.00 |
|
|
|
|
|
| (47,416) |
|
| 3.14 |
|
|
|
|
|
Forfeited |
| (153,891) |
|
| 10.39 |
|
|
|
|
|
| (28,351) |
|
| 10.91 |
|
|
|
|
|
Expired |
| (42,976) |
|
| 16.72 |
|
|
|
|
|
| (83,753) |
|
| 19.15 |
|
|
|
|
|
Outstanding as of September 30, 2016 |
| 2,346,307 |
| $ | 12.72 |
| 7.95 |
| $ | 4,361 | ||||||||||
Outstanding as of June 30, 2017 |
| 2,595,398 |
| $ | 11.50 |
| 7.85 |
| $ | 2,115 | ||||||||||
For the ninesix months ended SeptemberJune 30, 2016,2017, the weighted average grant date fair value of stock options granted was $2.92.$5.18. For the ninesix months ended SeptemberJune 30, 2016,2017, the total intrinsic value of options exercised was $11$114 and the total received from stock option exercises was $45.$149.
Activity related to restricted stock under the 2013 Plan for the ninesix months ended SeptemberJune 30, 2016,2017, was as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
| Weighted | ||
|
|
|
| Weighted |
|
|
| Average Grant | ||
|
|
|
| Average Grant |
| Shares |
| Date Fair Value | ||
|
| Shares |
| Date Fair Value | ||||||
Unvested restricted common stock as of December 31, 2015 |
| 333,263 |
| $ | 17.77 | |||||
Unvested restricted common stock as of December 31, 2016 |
| 461,463 |
| $ | 8.30 | |||||
Issued |
| 427,317 |
|
| 3.72 |
| 326,700 |
|
| 7.89 |
Vested |
| (207,635) |
|
| 13.00 |
| (192,780) |
|
| 10.12 |
Forfeited |
| (53,649) |
|
| 7.44 |
| (10,792) |
|
| 7.32 |
Unvested restricted common stock as of September 30, 2016 |
| 499,296 |
| $ | 8.84 | |||||
Unvested restricted common stock as of June 30, 2017 |
| 584,591 |
| $ | 7.49 | |||||
For the ninesix months ended SeptemberJune 30, 2016,2017, the total fair value of restricted common stock vested was $1,088.$1,418. The Company did not receive cash proceeds for any of the restricted common stock issued during the ninesix months ended SeptemberJune 30, 2016.2017.
Stock-Based Compensation
The Company recognizes compensation expense for only the portionUpon adoption of awards that are expected to vest. In developing a forfeiture rate estimate,ASU 2016-09 (Compensation – Stock Compensation) on January 1, 2017, the Company has consideredelected to change its historical experienceaccounting policy to estimate pre-vestingaccount for forfeitures for service-based awards.as they occur. The impactchange was applied on a modified retrospective basis with a cumulative-effect adjustment to accumulated deficit of a forfeiture rate adjustment will be recognized in full in$213 (which increased the periodaccumulated deficit) as of adjustment, and if the actual forfeiture rate is materially different from the Company’s estimate,January 1, 2017. Prior to adoption of this guidance the Company may be required to record adjustments to stock-based compensation expense in future periods.estimated forfeitures.
The Company recorded stock-based compensation expense related to stock options and restricted stock as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||||
|
| Three Months Ended |
| Nine Months Ended |
| Three Months Ended |
| Six Months Ended | ||||||||||||||||
|
| September 30, |
| September 30, |
| June 30, |
| June 30, | ||||||||||||||||
|
| 2016 |
| 2015 |
| 2016 |
| 2015 |
| 2017 |
| 2016 |
| 2017 |
| 2016 | ||||||||
Cost of product sales and inventories |
| $ | 30 |
| $ | 31 |
| $ | 79 |
| $ | 98 |
| $ | 44 |
| $ | 19 |
| $ | 84 |
| $ | 50 |
Research and development |
|
| 227 |
|
| 364 |
|
| 856 |
|
| 1,363 |
|
| 238 |
|
| 264 |
|
| 488 |
|
| 629 |
Selling, general and administrative |
|
| 1,831 |
|
| 1,705 |
|
| 5,601 |
|
| 5,028 |
|
| 1,562 |
|
| 1,913 |
|
| 3,086 |
|
| 3,770 |
|
| $ | 2,088 |
| $ | 2,100 |
| $ | 6,536 |
| $ | 6,489 |
| $ | 1,844 |
| $ | 2,196 |
| $ | 3,658 |
| $ | 4,449 |
TheAs of June 30, 2017, the Company had an aggregate of $7,915$6,634 and $3,145$3,582 of unrecognized stock-based compensation expense for options outstanding and restricted stock awards, respectively, as of September 30, 2016, which is expected to be recognized over a weighted average period of 2.172.32 years and 1.501.95 years, respectively.
2015
13.12. Net LossIncome (Loss) Per Share
Basic and diluted net lossincome (loss) per share was calculated as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
| Six Months Ended | ||||||||
|
| June 30, |
| June 30, | ||||||||
|
| 2017 |
| 2016 |
| 2017 |
| 2016 | ||||
Basic net income (loss) per share attributable to common stockholders: |
|
|
|
|
|
|
|
|
|
|
|
|
Numerator: |
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss) |
| $ | (10,380) |
| $ | 21,196 |
| $ | (22,992) |
| $ | 3,129 |
Net income attributable to participating securities |
|
| — |
|
| (20) |
|
| — |
|
| (3) |
Net income (loss) attributable to common stockholders |
| $ | (10,380) |
| $ | 21,176 |
| $ | (22,992) |
| $ | 3,126 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Denominator: |
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average shares outstanding – basic |
|
| 40,206,042 |
|
| 34,762,533 |
|
| 38,486,329 |
|
| 34,708,006 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss) per share attributable to common stockholders – basic |
| $ | (0.26) |
| $ | 0.61 |
| $ | (0.60) |
| $ | 0.09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Diluted net income (loss) per share attributable to common stockholders: |
|
|
|
|
|
|
|
|
|
|
|
|
Numerator: |
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss) |
| $ | (10,380) |
| $ | 21,196 |
| $ | (22,992) |
| $ | 3,129 |
Net income attributable to participating securities |
|
| — |
|
| (20) |
|
| — |
|
| (3) |
Net income (loss) attributable to common stockholders |
| $ | (10,380) |
| $ | 21,176 |
| $ | (22,992) |
| $ | 3,126 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Denominator: |
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average shares outstanding – basic |
|
| 40,206,042 |
|
| 34,762,533 |
|
| 38,486,329 |
|
| 34,708,006 |
Dilutive effect of outstanding stock awards |
|
| — |
|
| 175,922 |
|
| — |
|
| 71,780 |
Weighted average shares outstanding – diluted |
|
| 40,206,042 |
|
| 34,938,455 |
|
| 38,486,329 |
|
| 34,779,786 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss) per share attributable to common stockholders – diluted |
| $ | (0.26) |
| $ | 0.61 |
| $ | (0.60) |
| $ | 0.09 |
���
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
| Nine Months Ended | ||||||||
|
| September 30, |
| September 30, | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 2016 |
| 2015 |
| 2016 |
| 2015 | ||||
Basic and diluted net loss per share: |
|
|
|
|
|
|
|
|
|
|
|
|
Numerator: |
|
|
|
|
|
|
|
|
|
|
|
|
Loss before income taxes |
| $ | (13,371) |
| $ | (55,200) |
| $ | (10,242) |
| $ | (72,588) |
Income tax benefit |
|
| — |
|
| 758 |
|
| — |
|
| 1,389 |
Net loss |
| $ | (13,371) |
| $ | (54,442) |
| $ | (10,242) |
| $ | (71,199) |
Denominator: |
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average shares outstanding, basic and diluted |
|
| 35,092,686 |
|
| 34,405,646 |
|
| 34,837,169 |
|
| 34,293,357 |
Net loss per share, basic and diluted |
| $ | (0.38) |
| $ | (1.58) |
| $ | (0.29) |
| $ | (2.08) |
The Company’s participating securities consisted of unvested restricted common stock issued from early exercised stock options and restricted common stock awards granted under the 2010 Plan as these shares have non-forfeitable dividend rights.
Stock options for the purchase of 2,412,238 and 1,808,8592,652,792 shares of common stock were excluded from the computation of diluted net loss per share attributable to common stockholders for the three and ninesix months ended SeptemberJune 30, 2016 and 2015, respectively,2017, because those options had an anti-dilutive impact due to the net loss attributable to common stockholders incurred for the period.Stock options for the purchase of 1,705,396 and 1,941,521 weighted average shares of common stock and 551,709 and 557,149 of unvested restricted stock awards were excluded from the computation of diluted net income per share attributable to common stockholders for the three and six months ended June 30, 2016, respectively, because those awards had an anti-dilutive impact on the net income attributable to common stockholders.
14.13. Income Taxes
The Company recorded no income tax expense or benefit during the three and ninesix months ended SeptemberJune 30, 2016, compared to an income tax benefit of $7582017 and $1,389 during the three and nine months ended September 30, 2015, respectively, which was recognized for losses incurred that would reduce the amount of deferred tax liability related to intangible assets. The Company recognized no deferred tax benefit for losses incurred during the three and nine months ended September 30, 2016, due to a full valuation allowance recognized against its deferred tax assets.
16
14. Accumulated Other Comprehensive Loss
The changes in accumulated other comprehensive loss, net of their related tax effects, were as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Foreign |
| Accumulated | ||
|
| Currency |
| Other | ||
|
| Translation |
| Comprehensive | ||
|
| Adjustment |
| Loss | ||
As of December 31, 2015 |
| $ | (9,320) |
| $ | (9,320) |
Foreign currency translation adjustment |
|
| 891 |
|
| 891 |
As of September 30, 2016 |
| $ | (8,429) |
| $ | (8,429) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Foreign |
| Accumulated | ||
|
| Currency |
| Other | ||
|
| Translation |
| Comprehensive | ||
|
| Adjustment |
| Loss | ||
As of December 31, 2016 |
| $ | (9,862) |
| $ | (9,862) |
Foreign currency translation adjustment |
|
| 1,721 |
|
| 1,721 |
As of June 30, 2017 |
| $ | (8,141) |
| $ | (8,141) |
Amounts reclassified from accumulated other comprehensive loss were as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Amounts Reclassified from |
| Amounts Reclassified from | ||||||||
|
|
| Accumulated Other |
| Accumulated Other | ||||||||
|
|
| Comprehensive Loss |
| Comprehensive Loss | ||||||||
|
|
| Three Months Ended |
| Nine Months Ended | ||||||||
| Location in |
| September 30, |
| September 30, | ||||||||
| Statements of Operations |
| 2016 |
| 2015 |
| 2016 |
| 2015 | ||||
Gain on sale of securities available-for-sale | Other income (expense) |
| $ | — |
| $ | — |
| $ | — |
| $ | 3,874 |
|
|
| $ | — |
| $ | — |
| $ | — |
| $ | 3,874 |
15. Legal Contingencies
From time to time, the Company may become subject to legal proceedings, claims and litigation arising in the ordinary course of business, including those related to patents, product liability and government investigations. Except as described below, the Company is not presently a party to any litigation which it believes to be material, and is not aware of any pending or threatened litigation against the Company which it believes could have a material effect on its financial statements. The Company accrues contingent liabilities when it is probable that a future liability has been incurred and such liability can be reasonably estimated.
In February 2017, two purported class action lawsuits were filed in the United States District Court for the Southern District of New York against the Company and two of its current officers. Those cases have been consolidated into one purported class action lawsuit under the caption, In re Aratana Therapeutics, Inc. Securities Litigation, Case No. 1:17-cv-00880. The consolidated lawsuit asserts claims under Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, and is premised on allegedly false and/or misleading statements, and alleged non-disclosure of material facts, regarding the Company’s business, operations, prospects and performance during the proposed class period of March 16, 2015 to February 3, 2017. The Company intends to vigorously defend all claims asserted. Given the early stage of the litigation, at this time a loss is not probable or reasonably estimable.
21
16. Variable Interest Entity
ViroVet BVBA (“ViroVet”)
During the third quarter of 2015, the Company reviewed certain operations of its wholly owned subsidiary, Aratana Therapeutics NV (“Aratana NV”). As a result, the Company made a strategic decision to wind down pre-clinical discovery efforts being performed at Aratana NV and focus future efforts of Aratana NV on clinical assets, the development of core legacy programs, i.e. AT-001, AT-002 and AT-003 for EU approval and business development and monetization of production animal assets and know-how obtained in the acquisition of Okapi Sciences. To facilitate this reorganization, the Company, via Aratana NV, along with the former General Manager of Aratana NV, the current General Manager of Aratana NV and a consultant to the Company, formed ViroVet BVBA. During the third quarter of 2015 the Company began to transition some of its employees from Aratana NV to ViroVet. The Company plans to terminate additional Aratana NV employees, and transition selected Aratana NV assets and liabilities throughout 2016 to ViroVet to further pursue the research and development of production animal products.
Except for the financing matters described below, the Company will have little to no involvement in the operations of ViroVet.
Equity Investment
In July 2015 and August 2016, the Company paid $2 and $4, respectively, for 28% ownership interest in ViroVet. The Company has no further obligation to provide any further capital.
Convertible Loan Agreement
On September 11, 2015, Aratana NV and ViroVet executed a convertible loan agreement in which Aratana NV agreed to loan ViroVet $335 (€300) on September 15, 2015. The convertible loan agreement requires ViroVet to use the proceeds towards the development and operations of ViroVet in accordance with the budget prepared by ViroVet. The loan bears an annual interest rate of 7% and is unsecured. In September 2016, the Company agreed to extend the term of the convertible loan agreement up to March 31, 2017.
Primary Beneficiary
The Company determined ViroVet was a VIE and it had a controlling financial interest in ViroVet due to the Company having the power to direct the activities of ViroVet that most significantly impact ViroVet’s economic performance and having the obligation to absorb losses or receive benefits. The Company will continue to consolidate ViroVet unless a reconsideration event occurs, for example, an equity financing.
Total assets and liabilities of the Company’s consolidated VIE were not material as of September 30, 2016.
For the three and nine months ended September 30, 2016, ViroVet’s net loss and non-controlling interest were not material and are included in the Company’s consolidated statement of operations. Creditors in ViroVet only have recourse to the assets owned by the VIE and not to the Company’s general credit. The Company currently does not have implicit support arrangements with ViroVet.
2217
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our financial statements and the related notes and other financial information included elsewhere in this Quarterly Report on Form 10-Q. Some of the statements contained in this discussion and analysis or set forth elsewhere in this Quarterly Report on Form 10-Q that are not statements of historical fact are forward-looking statements within the meaning of the Private Securities Litigation Reform Act. In this Quarterly Report on Form 10-Q, the words “anticipates,” “believes,” “expects,” “intends,” “future,” “could,” “estimates,” “plans,” “would,” “should,” “potential,” “continues” and similar words or expressions (as well as other words or expressions referencing future events, conditions or circumstances) identify forward-looking statements. The forward-looking statements herein include without limitation, statements with respect to our plans and strategy for our business, anticipated timing of regulatory submissions and approvals, anticipated timing of availability and announcement of study results, anticipated timing of launch and commercialization of producttherapeutic candidates, ongoing efforts in preparation for commercialization of producttherapeutic candidates, beliefs regarding market opportunities for our products and potential success of our producttherapeutic candidates; Elanco’s intent to assume manufacturing responsibility for GALLIPRANT under the Collaboration Agreement; and anticipated milestone payments. These and other forward-looking statements included in this Quarterly Report on Form 10-Q involve risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to: our history of operating losses and our expectation that we will continue to incur losses for the foreseeable future; failure to obtain sufficient capital to fund our operations; risks relating to the impairment of intangible assets AT-004, AT-005,BLONTRESS, TACTRESS, AT-007 and AT-011;effects ofstockholder class action lawsuits; unstable market and economic conditions; restrictions on our financial flexibility due to the terms of our credit facility; our substantial dependence upon the commercial success of our product candidates;therapeutics GALLIPRANT, ENTYCE and NOCITA; development of our biologic producttherapeutic candidates is dependent upon relatively novel technologies and uncertain regulatory pathways, and biologics may not be commercially viable; denial or delay of regulatory approval for our existing or future producttherapeutic candidates; failure of our producttherapeutics, and our current or future therapeutic candidates that receivemay obtain regulatory approval to obtainachieve market approvalacceptance or achieve commercial success; effects of product liability lawsuits; failure to realize anticipated benefits of our acquisitions and difficulties associated with integrating the acquired businesses; development of pet therapeutics is a lengthy and expensive process with an uncertain outcome; competition in the pet therapeutics market, including from generic alternatives to our producttherapeutic candidates, and failure to compete effectively; failure to identify, license or acquire, develop and commercialize additional producttherapeutic candidates; failure to attract and retain senior management and key scientific personnel; our reliance on third-party manufacturers, suppliers and collaborators; regulatory restrictions on the marketing of our productapproved therapeutics and therapeutic candidates; our small commercial sales organization, and any failure to create a sales force or collaborate with third-parties to commercialize our productapproved therapeutics and therapeutic candidates; difficulties in managing the growth of our company; significant costs of being a public company; risks related to the restatement of our financial statements for the year ended December 31, 2013, and the identification of a material weakness in our internal control over financial reporting; changes in distribution channels for pet therapeutics; consolidation of our veterinarian customers; limitations on our ability to use our net operating loss carryforwards; impacts of generic products; safety or efficacy concerns with respect to our product candidates;therapeutics; effects of system failures or security breaches; failure to perform under our agreements with Elanco Animal Health, or termination thereof; failure to obtain ownership of issued patents covering our producttherapeutic candidates or failure to prosecute or enforce licensed patents; failure to comply with our obligations under our license agreements; effects of patent or other intellectual property lawsuits; failure to protect our intellectual property; changing patent laws and regulations; non-compliance with any legal or regulatory requirements; litigation resulting from the misuse of our confidential information; the uncertainty of the regulatory approval process and the costs associated with government regulation of our producttherapeutic candidates; failure to obtain regulatory approvals in foreign jurisdictions; effects of legislative or regulatory reform with respect to pet therapeutics; the volatility of the price of our common stock; our status as an emerging growth company, which could make our common stock less attractive to investors; dilution of our common stock as a result of future financings; the influence of certain significant stockholders over our business; and provisions in our charter documents and under Delaware law could delay or prevent a change in control. These and other important factors discussed under the caption “Risk Factors” in our Annual Report on Form 10-K for the fiscal year ended December31, 2015,2016, filed with the Securities and Exchange Commission (the “SEC”) on March15, 2016, 14, 2017, and the “Risk Factors” section of this Quarterly Report on Form 10-Q, could cause actual results to differ materially from those indicated by the forward-looking statements made in this Quarterly Report on Form 10-Q.
Overview
We are a pet therapeutics company focused on licensing, developing and commercializing innovative biopharmaceutical productstherapeutics for companion animals.dogs and cats. We operate in one business segment: pet therapeutics which sits at the intersection of the more than $60 billion annual U.S. pet market, and more than $23 billion annual worldwide animal health market. . Our current product portfolio includes multiple therapeutic productstherapeutics and producttherapeutic candidates in development consisting of both small molecule pharmaceuticals and large molecule biologics that target largebiologics. We intend for our portfolio to capture opportunities in seriousunmet or underserved medical conditions in pets.dogs and cats.
We have three United States Food and Drug Administration (“FDA”) approved therapeutics including, GALLIPRANT ® (grapiprant tablets) for the control of pain and inflammation associated with osteoarthritis in dogs; ENTYCE ® (capromorelin oral solution) for appetite stimulation in dogs; and NOCITA ® (bupivacaine liposome injectable suspension) as a local post-operative analgesia for cranial cruciate ligament surgery in dogs. BLONTRESS ® and TACTRESS® are our two canine-specific monoclonal antibody (MAb) therapies that are fully licensed by the United States Department of Agriculture (“USDA”) to aid in the treatment of dogs with B-cell
18
and T-cell lymphoma, respectively. Our lead products and productpipeline has multiple therapeutic candidates in development include small molecules directed at treating osteoarthritisfor the potential treatment of pain, management of weight loss, viral diseases, allergy and inflammation (GALLIPRANT®), appetite stimulation (ENTYCE®)cancer for dogs and post-operative pain (NOCITA®).cats.
We have incurred significant net losses since our inception. These losses have resulted principally from costs incurred in connection with in-licensing our producttherapeutic candidates, research and development activities, and selling, general and administrative costs associated with our operations. As of SeptemberJune 30, 2016,2017, we had a deficit accumulated since inception of $162.3$208.8 million and cash, cash equivalents, restricted cash and short-term investments of $102.3$80.7 million.
Business Updates
During the three months ended June 30, 2017, we continued to grow our commercial presence by gaining access to target clinics and driving re-orders for existing accounts. The second quarter of 2017 was the first full quarter of sales of GALLIPRANT, which we are selling in collaboration with Eli Lilly and Company, operating on behalf of its Elanco Animal Health division (“Elanco”), and total GALLIPRANT revenues which included product sales for supply sold to Elanco and GALLIPRANT licensing and collaboration revenue increased sequentially during the quarter. Additionally, we continued to leverage our relationships with veterinary surgeons to increase the number of new accounts and drive existing account re-orders of NOCITA. NOCITA sales also increased sequentially during the second quarter of 2017.
We continued to advance our late-stage pipeline of therapeutic candidates for dogs and cats. Specifically, in June 2017, we submitted final pivotal field safety study data from an evaluation of AT-014 to the USDA and we filed the target animal safety technical section for AT-003 in cats with the FDA’s Center for Veterinary Medicine (“CVM”). In July 2017, we demonstrated positive results from the AT-003 pivotal field effectiveness study in cats.
In July 2017, pursuant to the Collaboration, License, Development and Commercialization Agreement with Elanco (the “Collaboration Agreement”), Elanco provided us with notice of its intent to assume responsibility for the manufacturing of the Company’s products based on licensed grapiprant rights and technology, including GALLIPRANT (collectively, “Grapiprant Products”) and its intent to assume applicable regulatory approvals. We believe the transfers of manufacturing responsibility and such regulatory approvals in the United States to Elanco will be completed by December 31, 2017. Additionally, in late-June 2017, we re-submitted the prior-approval supplement (“PAS”) to CVM for the manufacturing transfer of ENTYCE.
Research and Development
The following table identifies the most advanced therapeutic candidates being developed under CVM or the USDA’s Center for Veterinary Biologics (“CVB”) regulations and their current regulatory status:

ENTYCE (capromorelin oral solution) for dogs
ENTYCE was approved by the FDA for appetite stimulation in dogs in 2016. We continue to anticipate that ENTYCE will be commercially available by the fall of 2017, depending on the timing of CVM’s approval, if any, of the PAS. See “Manufacturing and Supply Chain” below for additional information.
2319
Business Updates
AT-003 (bupivacaine liposome injectable suspension) for cats
DuringIn June 2017, we filed the nine months ended September 30,target animal safety technical section with CVM for review and in July 2017, we completed an FDA-concurred pivotal field effectiveness study evaluating bupivacaine liposome injectable suspension for post-operative pain management in cats.
The multi-center, placebo-controlled, randomized and masked pivotal field effectiveness study evaluated approximately 200 client-owned cats undergoing an elective onychectomy. Results from the study showed bupivacaine liposome injectable suspension met protocol-defined efficacy success criteria, which were statistically significant (p<0.05). The therapeutic candidate was well-tolerated. Data from the study will be used as part of the effectiveness technical section submission to CVM to support the intended NOCITA label expansion to include cats.
AT-016 (allogeneic adipose-derived stem cells) for dogs
VetStem BioPharma Inc., our exclusive license partner responsible for development of AT-016, an adipose-derived allogeneic stem cell therapeutic candidate for osteoarthritis pain in dogs, has on-going FDA-concurred target animal safety and target animal efficacy studies with results anticipated from both in 2017.
AT-014 (canine osteosarcoma vaccine) for dogs
In the first quarter of 2017, we completed enrollment in the pivotal field safety study evaluating AT-014 for the treatment of canine osteosarcoma in dogs and in June 2017, we submitted the safety data to the USDA. We continue to anticipate conditional licensure in the second half of 2017. We are finalizing our plans with respect to commercializing AT-014, assuming conditional licensure, which may include making AT-014 available to a group of veterinary oncologists as we complete the additional work required by the USDA for full licensure.
In addition to United States regulatory and development activities, we also have regulatory and development efforts outside the United States:
AT-001 (grapiprant) for dogs in Europe
Under the Collaboration Agreement, Elanco has exclusive rights to our products based on licensed grapiprant rights and technology, including GALLIPRANT (collectively, “Grapiprant Products”), globally outside the United States. We are responsible under the Collaboration Agreement for all development activities required to obtain the first regulatory approval for grapiprant for use in dogs in the European Union (“EU”) and Elanco is responsible for all other development activities. In February 2016, we continuedfiled a marketing authorization application with the European Medicines Agency (“EMA”) for grapiprant in dogs in the EU. In cooperation with Elanco, we have generated additional data and responses to questions which we plan to submit to the EMA. We believe that the filing and other interactions with the EMA will support a positive opinion in late-2017 and marketing authorization in the first half of 2018.
Other therapeutics for dogs in Europe
We continue to make progress towardswith regulatory authorities in Europe with respect to certain of our objective of becoming a fully integratedother therapeutic candidates.
Manufacturing and Supply Chain
We manage third-party manufacturers to supply active pharmaceutical ingredient (“API”), drug product and packaged product for the development and commercialization of our small molecule product candidates. We have chosen to rely on third-party contract manufacturer organizations (“CMO”) rather than devote resources toward developing or acquiring internal manufacturing facilities.
GALLIPRANT
GALLIPRANT has been available to customers since January 2017, and as part of the Collaboration Agreement, we have agreed to provide the initial commercial companysupply of GALLIPRANT. On April 28, 2017, we and Elanco entered into an amendment (the “Amendment”) to the Collaboration Agreement. Under the Amendment, Elanco has agreed to submit binding purchase orders to us, within 15 days of the effective date of the Amendment, for certain finished Grapiprant Products to be produced from certain batches of API we have agreed to purchase from our third-party manufacturer (the “API Batches”). In addition, Elanco has agreed to pay us for the API Batches within 30 days after we provide Elanco with proof of payment to the manufacturer for such API Batches. The Amendment provides that, in 2016. During the third quarterevent Elanco provides notice of 2016, we receivedits intent to assume responsibility for manufacturing, Elanco would assume all of our third U.S. Food and Drug Administration (“FDA”) approval, initiated two pivotal field studies, raised capital, continuedresponsibilities with respect to progress onany undelivered API, including paying the third-party manufacturer for such undelivered API. In July 2017, Elanco provided us with notice of its intent to assume responsibility for manufacturing of commercial supply for upcoming product launches,the Grapiprant Products and prepared ourselvesits intent to have a commercial presenceassume the regulatory approvals. We believe the transfers of manufacturing responsibility and the regulatory approvals in the pet therapeutic market. Shortly afterUnited States to Elanco will be completed by December 31, 2017. The companies are currently formulating a plan for the endtransfer of manufacturing responsibilities and the third quarterNew Animal Drug Application (“NADA”).
GALLIPRANT is currently available in 20 mg and 60 mg tablets in a variety of 2016,packaging configurations, which we commercially launched our FDA-approved therapeutic NOCITA.
Research and Development
believe provide appropriate treatment options for the majority of dogs. The following tables identify the most advanced product candidates being developed under the FDA’s Center for Veterinary Medicine (“CVM”) regulations and their current regulatory status:
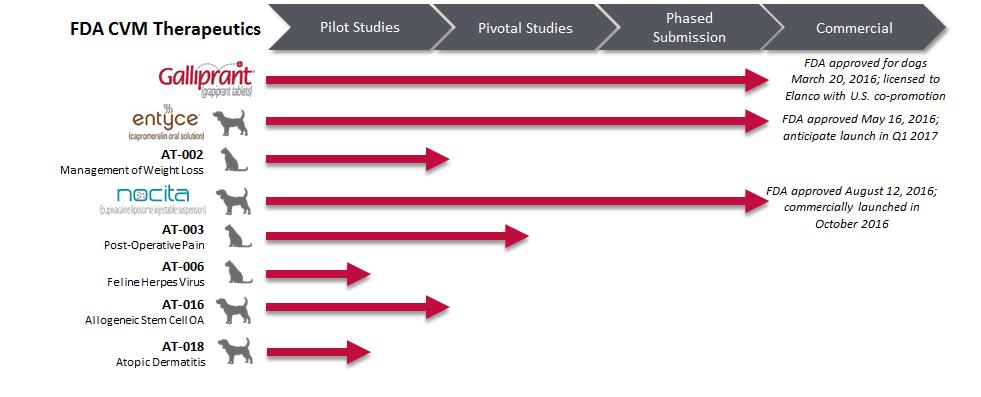
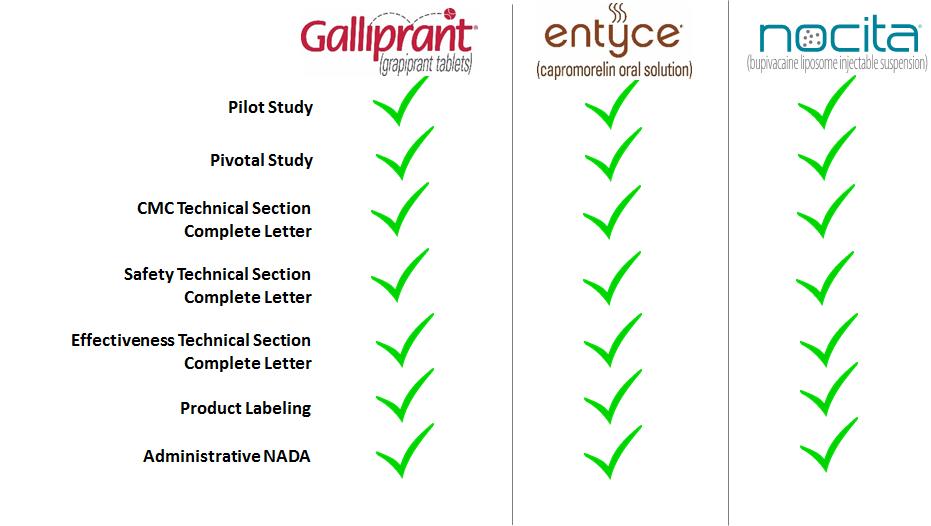
100 mg tablets of GALLIPRANT remain on backorder,
2420
NOCITA
On August 12, 2016, the FDA’s CVM approved NOCITA (bupivacaine liposome injectable suspension), a local post-operative analgesia for cranial cruciate ligament surgery in dogs. NOCITA was made commercially available to veterinarianswhich we anticipate resolving in the United Statescoming quarters.
ENTYCE
In late-June 2017, we re-submitted the PAS for the transfer and commercial scale-up of the API of ENTYCE to a different manufacturer from the manufacturer included in October 2016.
AT-003 for cats
Duringour NADA. As part of the third quarter of 2016,PAS re-submission, we began enrollmentprovided what we believe was the appropriate information related to a raw material and its vendor. However, the CVM may request additional information which could result in the pivotal field effectiveness studyneed for post-operative pain management in cats under an FDA-concurred protocol.
AT-016 for dogs
Duringus to amend our filing or make additional filings. If we are unable to resolve any questions from CVM, we may be required to switch commercial manufacturers, or our manufacturer may be required to source the third quarter of 2016, our exclusive license partner responsible for development, VetStem Biopharma, Inc. (“VetStem”), initiated the pivotal field effectiveness study for dogs with pain associated with osteoarthritis under an FDA-concurred protocol.
AT-018 for dogs
In October 2016, we and our license partner, Atopix Therapeutics, Ltd, reviewed the high-level results ofraw material from a multi-center, masked, placebo-controlled, randomized pilot field study in client-owned dogs with recurrent atopic dermatitis. The pilot study for AT-018 was designed to evaluate the product candidate’s ability to maintain the reduction of clinical signs in dogs with recurrent atopic dermatitis after receiving corticosteroids. The study design required that patients were pre-treated with prednisone, and the principle evaluation was based on subsequent differences in symptom scores in dogs that had responded to prednisone. Seventy-five dogs were enrolled in the study but only approximately 40 percent of the cases were evaluable because of the unexpectedly high failure rate of prednisone. We do not believe that AT-018 demonstrated sufficient activity to merit continued development in this severe disease, and based on our recent discussions with the FDA, we plan to pursuean alternative clinical setting. Based on the encouraging laboratory studies, discussions with the FDA, and the fact that AT-018 appears well-tolerated, we intend to investigate AT-018 to prevent clinical signs in at-risk dogs. We intend to initiate a pilot study in 2017 to probe this new indication and we remain enthusiastic about continued development of AT-018.
Products in our pipeline regulated by the United States Department of Agriculture (“USDA”) are:
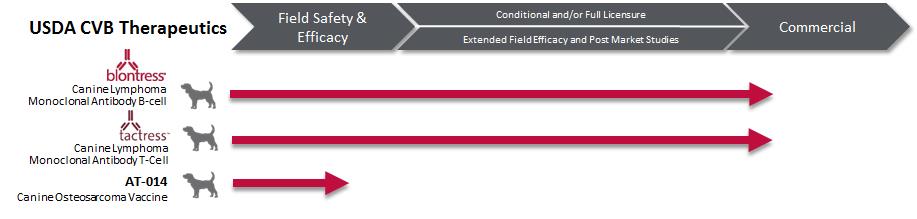
BLONTRESS and TACTRESS
We anticipate the results of the BLONTRESS post-approval study, Mini B-CHOMP, in late 2016. In the third quarter of 2015, our interim analysis of clinical results indicated that TACTRESS did not seem to be adding significant progression-free survival in canine T-cell lymphoma; those results were confirmed in the final study results in July 2016.
AT-014 for dogs
During the third quarter of 2016, we continued enrollment in the pivotal field safety study for AT-014, an investigational canine osteosarcoma vaccine that we licensed from Advaxis, Inc.different vendor. We continue to anticipate conditional licensure in the first half of 2017.
Manufacturing and Supply Chain
GALLIPRANT andthat ENTYCE
During the third quarter of 2016, we continued to transfer the manufacturing technology processes for GALLIPRANT and ENTYCE to our identified active pharmaceutical ingredient (“API”) and formulated product contract manufacturers to provide commercial supplies. We have completed the required manufacturing validation work in regards to API for both GALLIPRANT and ENTYCE. We made additional manufacturing filings with the FDA to obtain approvals that are required for commercial launch. Further, we continue to complete the required manufacturing validation work of formulated product and packaging to produce inventory for commercial availability.
25
NOCITA
We have initial NOCITA product supply to support commercial launch. While this initial supply is labeled with 24-month shelf life, to comply with recent FDA guidance affecting our product, subsequent supply will be labeled with 12-month shelf life. We anticipate that this labeling change will not impact our abilitycommercially available by the fall of 2017, depending on the timing of CVM’s approval, if any, of the PAS. Subsequent to market the product.
Sales and Marketing
We have built ourre-submission of the PAS, we resumed manufacturing additional commercial organization to target companion animal veterinarians ininventory of ENTYCE, including API from the United States who typically act as not only the prescriber of product, but as the pharmacist as well. During the third quarter of 2016,manufacturer for which we hired approximately two dozen Therapeutic Specialists in territories across the United States and we hosted our first national sales meeting in Kansas City the last week of September 2016. Regional Sales Leaders, National Account Managers and Medical Scientific Liaisons, as well as experienced sales and marketing leadership, round out the commercial organization.
In October 2016, in conjunction with our attendance at the American College of Veterinary Surgeons Surgery Summit in Seattle we commercially launched NOCITA. Our commercial focus for the rest of 2016 will be NOCITA.are seeking approval from CVM.In November 2016, Elanco Animal Health, Inc. (“Elanco”), a division of Eli Lilly & Co., communicated to us that they anticipate commercial launch of GALLIPRANT in early 2017, assuming Aratana’s supply of GALLIPRANT is approved and released.
Recent Developments
Properties
Effective as of July 1, 2016,31, 2017, we renewedamended our lease of approximately 6,600 square feet of office, laboratoryLoan Agreement. See“Financial Condition, Liquidity and manufacturing space in San Diego, California, that now expires on June 30, 2021. We did not renew our lease of corporate office space in Boston, Massachusetts, which expired pursuant to the lease terms on August 31, 2016.
Capital Resources - Indebtedness” below for additional information.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of our consolidated financial statements and related disclosures requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, and revenues, costs and expenses and related disclosures during the reporting periods. On an ongoing basis, we evaluate our estimates and judgments. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
There have been no material changes to our critical accounting policies through SeptemberJune 30, 2016,2017, from those discussed in our Annual Report on Form 10-K for the fiscal year ended December 31, 2015,2016, filed with the SEC on March 15, 2016. 14, 2017.
Results of Operations
Comparison of the Three and Six Months Ended June 30, 2017 and 2016
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
|
|
|
| Six Months Ended |
|
|
| ||||||||
|
| June 30, |
|
|
|
| June 30, |
|
|
| ||||||||
|
| 2017 |
| 2016 |
| % Change |
| 2017 |
| 2016 |
| % Change | ||||||
|
| (Dollars in thousands) |
|
|
|
| (Dollars in thousands) |
|
|
| ||||||||
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Licensing and collaboration revenue |
| $ | 804 |
| $ | 38,000 |
| (97.9) | % |
| $ | 1,707 |
| $ | 38,151 |
| (95.5) | % |
Product sales |
|
| 4,354 |
|
| 47 |
| >100.0 | % |
|
| 7,246 |
|
| 68 |
| >100.0 | % |
Total revenues |
|
| 5,158 |
|
| 38,047 |
| (86.4) | % |
|
| 8,953 |
|
| 38,219 |
| (76.6) | % |
Costs and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of product sales |
|
| 3,691 |
|
| 1,741 |
| >100.0 | % |
|
| 6,785 |
|
| 1,760 |
| >100.0 | % |
Royalty expense |
|
| 353 |
|
| 20 |
| >100.0 | % |
|
| 676 |
|
| 38 |
| >100.0 | % |
Research and development |
|
| 3,700 |
|
| 5,303 |
| (30.2) | % |
|
| 8,354 |
|
| 16,052 |
| (48.0) | % |
Selling, general and administrative |
|
| 6,918 |
|
| 6,148 |
| 12.5 | % |
|
| 14,413 |
|
| 12,699 |
| 13.5 | % |
Amortization of intangible assets |
|
| 86 |
|
| 95 |
| (9.5) | % |
|
| 150 |
|
| 190 |
| (21.1) | % |
Impairment of intangible assets |
|
| — |
|
| 2,780 |
| (100.0) | % |
|
| — |
|
| 2,780 |
| (100.0) | % |
Other income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
| 88 |
|
| 83 |
| 6.0 | % |
|
| 173 |
|
| 160 |
| 8.1 | % |
Interest expense |
|
| (871) |
|
| (846) |
| 3.0 | % |
|
| (1,731) |
|
| (1,695) |
| 2.1 | % |
Other expense, net |
|
| (7) |
|
| (1) |
| >100.0 | % |
|
| (9) |
|
| (36) |
| (75.0) | % |
Revenues
During the three and six months ended June 30, 2017, total revenues decreased by $32.9 million and $29.3 million, respectively, as compared to the corresponding 2016 periods. The decreases in total revenues during the three and six months ended June 30, 2017, were due to decreases of $37.2 million and $36.4 million, respectively, in licensing and collaboration revenue from the Collaboration Agreement and the Co-Promotion Agreement with Elanco (collectively, the “Elanco GALLIPRANT Agreements”), partially offset by increases of $4.3 million and $7.2 million in net product sales primarily due to net sales of GALLIPRANT and NOCITA. Total revenues for the three and six months ended June 30, 2016, included $38.0 million of licensing and collaboration revenue recognized
2621
Results of Operations
Comparison offrom the Three and Nine Months Ended September 30, 2016 and 2015
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
|
|
|
| Nine Months Ended |
|
|
| ||||||||
|
| September 30, |
|
|
|
| September 30, |
|
|
| ||||||||
|
| 2016 |
| 2015 |
| % Change |
| 2016 |
| 2015 |
| % Change | ||||||
|
| (Dollars in thousands) |
|
|
|
| (Dollars in thousands) |
|
|
| ||||||||
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Licensing and collaboration revenue |
| $ | — |
| $ | — |
| NA |
|
| $ | 38,151 |
| $ | — |
| NA |
|
Product sales |
|
| 40 |
|
| 229 |
| (82.5) | % |
|
| 108 |
|
| 615 |
| (82.4) | % |
Total revenues |
|
| 40 |
|
| 229 |
| (82.5) | % |
|
| 38,259 |
|
| 615 |
| >100.0 | % |
Costs and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of product sales |
|
| 286 |
|
| 138 |
| >100.0 | % |
|
| 2,046 |
|
| 357 |
| >100.0 | % |
Royalty expense |
|
| 19 |
|
| 23 |
| (17.4) | % |
|
| 57 |
|
| 66 |
| (13.6) | % |
Research and development |
|
| 5,334 |
|
| 6,197 |
| (13.9) | % |
|
| 21,386 |
|
| 18,499 |
| 15.6 | % |
Selling, general and administrative |
|
| 6,924 |
|
| 4,997 |
| 38.6 | % |
|
| 19,623 |
|
| 14,061 |
| 39.6 | % |
Amortization of intangible assets |
|
| 91 |
|
| 483 |
| (81.2) | % |
|
| 281 |
|
| 1,449 |
| (80.6) | % |
Impairment of intangible assets |
|
| — |
|
| 43,398 |
| (100.0) | % |
|
| 2,780 |
|
| 43,398 |
| (93.6) | % |
Other income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
| 116 |
|
| 33 |
| >100.0 | % |
|
| 276 |
|
| 147 |
| 87.8 | % |
Interest expense |
|
| (859) |
|
| (226) |
| >100.0 | % |
|
| (2,554) |
|
| (661) |
| >100.0 | % |
Other income (expense), net |
|
| (14) |
|
| — |
| NA |
|
|
| (50) |
|
| 5,141 |
| <(100.0) | % |
Income tax benefit |
| $ | — |
| $ | 758 |
| (100.0) | % |
| $ | — |
| $ | 1,389 |
| (100.0) | % |
Revenues
Elanco GALLIPRANT Agreements. During the three and ninesix months ended SeptemberJune 30, 2016, total revenues decreased by $0.22017, product sales consisted of net sales of GALLIPRANT, NOCITA, BLONTRESS and TACTRESS. GALLIPRANT product sales during the three and six months ended June 30, 2017, consisted of net sales of finished goods to Elanco under the supply terms of the Collaboration Agreement. NOCITA net sales were $0.6 million and increased by $37.6$1.0 million during the three and six months ended June 30, 2017, respectively, as compared to $0 for the corresponding 20152016 periods. The decreaseWe believe that the growth in NOCITA product sales is primarily a result of continued growth of new accounts, as well as strong re-order rates, which accounted for more than 75% of NOCITA revenue within the three month period wasmonths ended June 30, 2017.
We believe that product sales for 2017 will be composed primarily due to a decrease in theof product sales of TACTRESS. The increase during the nine month period was primarily due to an increase of $38.0 million in licensing and collaboration revenue related to the Collaboration, License, Development and Commercialization Agreement with Elanco entered into in April 2016 (the “Collaboration Agreement”), partially offset by a decrease of $0.5 million in product sales. During the three and nine months ended September 30, 2016, product sales consisted of sales of BLONTRESS and TACTRESS. We believe that BLONTRESS and TACTRESS product sales will remain modest for the rest of 2016, NOCITA, product sales began in the fourth quarter of 2016 and product sales from GALLIPRANT, and ENTYCE, for which sales are still anticipated to begin by the fall of 2017, depending on the timing of CVM’s approval, if any, of the PAS. We believe NOCITA future product sales in 2017 will be dependent on our continuing efforts to commercialize the product. With respect to GALLIPRANT, in July 2017, Elanco gave us notice of its intent to assume all manufacturing responsibilities for GALLIPRANT under the Collaboration Agreement, which we believe will be completed by December 31, 2017. AnySee “Manufacturing and Supply Chain” above for additional information. The amount of any future product sales under the supply terms of the Collaboration Agreement will depend on how long Elanco utilizes us to supply GALLIPRANT. At this time, we believe customer demand does not justify manufacturing additional BLONTRESS or TACTRESS. Hence, we anticipate both therapeutics will no longer be commercially available when the current inventories are exhausted by late-2017. We believe any future licensing and collaboration revenue in 2017 will be substantially dependent on Elanco’s ability to successfully commercialize GALLIPRANT in accordance with the Collaboration Agreement.Elanco GALLIPRANT Agreements.
Cost of product sales
During the three and the ninesix months ended SeptemberJune 30, 2016,2017, cost of product sales increased by $0.1$2.0 million and $5.0 million, respectively, as compared to the corresponding 2016 periods. These increases were primarily due to cost of product sales of GALLIPRANT and NOCITA. Cost of product sales is anticipated to increase in the second half of 2017 due to the anticipated full period sales of NOCITA and GALLIPRANT, and anticipated second half year sales of ENTYCE. We believe the cost of product sales as a percentage of overall product revenues will improve as we move GALLIPRANT, NOCITA and ENTYCE to full commercial manufacturing scale over time.
Royalty expense
During the three and six months ended June 30, 2017, royalty expense increased by $0.3 million and $0.6 million, respectively, as compared to the corresponding 2016 periods. The increases were primarily a result of sales of GALLIPRANT and NOCITA.
Research and development
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
|
|
|
| Six Months Ended |
|
|
| ||||||||
|
| June 30, |
|
|
|
| June 30, |
|
|
| ||||||||
|
| 2017 |
| 2016 |
| % Change |
| 2017 |
| 2016 |
| % Change | ||||||
|
| (Dollars in thousands) |
|
|
|
| (Dollars in thousands) |
|
| |||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contracted development costs |
| $ | 2,808 |
| $ | 2,735 |
| 2.7 | % |
| $ | 6,440 |
| $ | 6,788 |
| (5.1) | % |
Milestones |
|
| — |
|
| 1,200 |
| (100.0) | % |
|
| — |
|
| 6,200 |
| (100.0) | % |
Personnel costs |
|
| 781 |
|
| 1,181 |
| (33.9) | % |
|
| 1,692 |
|
| 2,677 |
| (36.8) | % |
Other costs |
|
| 111 |
|
| 187 |
| (40.6) | % |
|
| 222 |
|
| 387 |
| (42.6) | % |
Total research and development |
| $ | 3,700 |
| $ | 5,303 |
| (30.2) | % |
| $ | 8,354 |
| $ | 16,052 |
| (48.0) | % |
During the three and six months ended June 30, 2017, research and development expense decreased by $1.6 million and $7.7 million, respectively, as compared to the corresponding 2016 periods. The decrease during the three months ended June 30, 2017, was due primarily to a decrease of $1.2 million in milestone payments and a decrease of $0.4 million in personnel costs related to a lower headcount in 2017. The decrease during the six months ended June 30, 2017, was due primarily to a decrease of $6.2 million in milestone payments, a decrease of $0.3 million in contracted development costs due to the prioritization of spending for ongoing programs, a $1.0 million decrease in personnel costs related to a lower headcount in 2017, and a $0.2 million decrease in other costs.
We expect that, in 2017, our research and development expenses will be primarily related to expanding the label of our approved therapeutics for additional indications and/or species and advancing our AT-016 and AT-018 programs.
Selling, general and administrative
During the three and six months ended June 30, 2017, selling, general and administrative expense increased by $0.8 million and $1.7 million, respectively, as compared to the corresponding 20152016 periods. The increases were primarily due to inventory valuation adjustment losses in the amount of $0.1 million and $1.7 million for the three and nine months of 2016, respectively, from the write-off of TACTRESS inventories and pre-launch GALLIPRANT inventories marked to market due to terms agreed upon in the Collaboration Agreement.
Royalty expense
During the three and nine months ended September 30, 2016, royalty expense decreased by $4,000 and $9,000, respectively, as compared to the corresponding 2015 periods. The decreases in both the three and nine month periods were primarily a result of the decreased sales of TACTRESS.
27
Research and development
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Three Months Ended |
|
|
|
| Nine Months Ended |
|
|
| ||||||||
|
| September 30, |
|
|
|
| September 30, |
|
|
| ||||||||
|
| 2016 |
| 2015 |
| % Change |
| 2016 |
| 2015 |
| % Change | ||||||
|
| (Dollars in thousands) |
|
|
|
| (Dollars in thousands) |
|
| |||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contracted development costs |
| $ | 4,014 |
| $ | 4,018 |
| (0.1) | % |
| $ | 10,802 |
| $ | 12,020 |
| (10.1) | % |
Milestones |
|
| 250 |
|
| 500 |
| (50.0) | % |
|
| 6,450 |
|
| 600 |
| >100.0 | % |
Personnel costs |
|
| 900 |
|
| 1,081 |
| (16.7) | % |
|
| 3,577 |
|
| 4,497 |
| (20.5) | % |
Other costs |
|
| 170 |
|
| 598 |
| (71.6) | % |
|
| 557 |
|
| 1,382 |
| (59.7) | % |
Total research and development |
| $ | 5,334 |
| $ | 6,197 |
| (13.9) | % |
| $ | 21,386 |
| $ | 18,499 |
| 15.6 | % |
During the three and nine months ended September 30, 2016, research and development expense decreased by $0.9 million and increased by $2.9 million, respectively, as compared to the corresponding 2015 periods. The decrease forincrease during the three months ended SeptemberJune 30, 2016,2017, was due to a decrease of $0.3 million in milestone payments, $0.2 million decrease in personnel costs relating to a lower headcount in 2016 and $0.4 million decrease in other costs. The increase for the nine months ended September 30, 2016, was primarily due to an increase of $5.9$1.1 million in milestones relating to GALLIPRANT, ENTYCE, NOCITA, and AT-016. This increase was partially offset by a decrease of $0.9 million in personnel costs relating to a lower headcount in the 2016 nine month period, a decrease of $1.2 million in contracted development costs as a result of the completion of several clinical studies for our lead programs, and a $0.8 million decrease in other costs.
In addition, based on our development plans as of September 30, 2016, we have committed to make potential future milestone payments to third parties of up to approximately $112.1 million as part of our various collaborations, including licensing and development programs. Payments under these agreements generally become due and payable only upon achievement of certain development, regulatory or commercial milestones. The $112.1 million consists of $80.4 million of commercial milestones and $31.7 million of development and regulatory milestones. Of the $80.4 million, we anticipate paying $6.2 million in the next 12 months. In the next five years, we anticipate paying up to $10.0 million of first commercial sales milestones for AT-001 and AT-002 if those products are launched in the U.S. and Europe, and the remainder if certain net revenue thresholds are achieved thereafter. Of the $31.7 million of development and regulatory milestones, we anticipate paying $0.5 million during the next 12 months, provided various development and regulatory milestones are achieved. The $31.7 million in milestones is spread across AT-001, AT-002, AT-003, AT-014, AT-016, AT-017, AT-018, and other pre-development candidates. Amounts related to contingent milestone payments are not considered contractual obligations as they are contingent on the successful achievement of certain development, regulatory approval and commercial milestones.
In 2016, we expect to incur research and development expenses related to technology transfer to contract manufacturing suppliers for commercial supply for our lead programs and regulatory expenses related to product filings.
We expect to fund our research and development expenses from our cash, cash equivalents and short-term investments and any future collaboration or financing arrangements. We cannot forecast with any degree of certainty which product candidates may be subject to future collaborations or contracts, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
Selling, general and administrative
During the three and nine months ended September 30, 2016, selling, general and administrative expense increased by $1.9 million and $5.6 million, respectively, as compared to the corresponding 2015 periods. For the three months and nine months ended September 30, 2016, these increases were due to increases of $0.9 million and $1.9 million, respectively, in personnel expenses primarily as a result of higher sales and marketing headcount, and increasespartially offset by a decrease of $1.0$0.3 million and $2.5 million, respectively, in expenses incurred in preparation forother expenses. The increase during the commercialization of product candidates for which we have received regulatory approval in 2016. The increases in the commercialization expenses were primarilysix months ended June 30, 2017, was due to product branding, sponsorshipan increase of $2.4 million in personnel expenses primarily as a result of higher sales and marketing headcount, partially offset by a decrease of $0.7 million in conjunction with our participation in several key veterinary conferences to build awareness of Aratana and our product candidates, and expenses related to corporate technology and infrastructure. For the nine months ended September 30, 2016, the $5.6 million increase in selling, general and administrative expenses was also due to a credit of $1.2 million recorded in the first quarter of 2015 to reduce the fair value of the contingent consideration to zero, which had originally been due under the Vet Therapeutics, Inc. merger agreement.
other expenses. We expect that, in 2017, our selling, general and administrative expense to continue to increasewill remain relatively constant as we have substantially completed the build out of our sales organization and corporate infrastructure in the support of the expected commercialization of GALLIPRANT, ENTYCE, NOCITA, AT-014, and other development programs.
continued
2822
Amortizationcommercialization of intangible assets
During the threeNOCITA and nine months ended September 30, 2016, amortizationGALLIPRANT and expected commercialization of intangible assets decreased by $0.4 million and $1.2 million, respectively, ENTYCE.as compared to the corresponding 2015 periods. The decreases in both the three and nine month periods reflect the impact of the impairment of BLONTRESS and TACTRESS in 2015. The lower carrying value due to the impairment charge resulted in the lower expense being recognized. Lower carrying values due to the TACTRESS impairment charge in 2016 will also result in lower amortization of intangible assets for the impaired intangibles in future periods.
Impairment of intangible assets
During both the three and ninesix months ended SeptemberJune 30, 2016,2017, impairment of intangible assets decreased by $43.4$2.8 million, and $40.6 million, respectively, as compared to the corresponding 2015 periods.2016 periods as there were no intangible asset impairment charges recognized during the three and six months ended June 30, 2017. The impairment of intangible assets in 2016 was related to impairment charges of TACTRESS ($0.6 million) and AT-007 ($2.2 million). The impairment charge related to TACTRESS resulted from updated sales expectations and resulted in a carrying value of $0 for TACTRESS. The impairment charge related to AT-007 was the result of our decision to discontinue the development of AT-007 due to the return of global rights of AT-006 and ensuing development program portfolio prioritization, including consideration of our focus on commercial launch activities to support our recently approved products, and resulted in a carrying value of $0 for AT-007. For more information regarding the impairment charges, see Note 7 to the consolidated financial statements. Unfavorable outcomes of our development activities or our estimates of the market opportunities for the product candidates could result in additional impairment charges in future periods. For example, we anticipate the results of the BLONTRESS post-approval study, Mini B-CHOMP, in late 2016.
Interest income
During the three and nine months ended September 30, 2016, interest income increased by $83,000 and $129,000, respectively, as compared to the corresponding 2015 periods. The increases primarily related to interest earned at higher interest rates and on higher deposits held at Square 1 Bank and short-term investments in reverse repurchase agreements.
Interest expense
During the three and nine months ended September 30, 2016, interest expense increased by $0.6 million and $1.9 million, respectively, as compared to the corresponding 2015 periods. These increases were due to interest expense related to our Loan Agreement, as discussed below in “Financial Condition, Liquidity and Capital Resources – Indebtedness”, which was entered into during October 2015. During the three and nine months ended September 30, 2016, accretion of the debt discount and amortization of deferred financing costs totaled $0.1 million and $0.4 million, respectively, which is non-cash interest included in our interest expense above.
Other income (expense), net
During the three and nine months ended September 30, 2016, other income (expense), net decreased by $14,000 and $5.2 million, respectively, as compared to the corresponding 2015 periods. The decrease during the nine month period was primarily related to a $3.5 million gain on the sale of Advaxis stock, a $1.3 million increase in the fair value of the Advaxis warrant, and a $0.3 million gain on the sale of shares received from the exercise of the Advaxis warrant that occurred in the corresponding period in 2015.
Income tax benefit
During the three and nine months ended September 30, 2016, income tax benefit decreased by $0.8 million and $1.4 million, respectively, as compared to the corresponding 2015 periods. The income tax benefit in 2015 was recognized for losses incurred that would reduce the amount of deferred tax liability related to intangible assets. There was no deferred tax benefit recognized for losses incurred during the three and nine months ended September 30, 2016, due to a full valuation allowance recognized against our deferred tax assets.
29
Financial Condition, Liquidity and Capital Resources
Our financial condition is summarized as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
|
| September 30, 2016 |
| December 31, 2015 |
| Change % |
| June 30, 2017 |
| December 31, 2016 |
| Change % | ||||||
|
| (Dollars in thousands) |
|
|
|
| (Dollars in thousands) |
|
|
| ||||||||
Financial assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
| $ | 100,847 |
| $ | 26,755 |
| >100.0 | % |
| $ | 78,823 |
| $ | 87,307 |
| (9.7) | % |
Marketable securities - short-term |
|
| 1,494 |
|
| 747 |
| 100.0 | % |
|
| 1,494 |
|
| 996 |
| 50.0 | % |
Reverse repurchase agreements |
|
| — |
|
| 58,700 |
| (100.0) | % | |||||||||
Total cash, cash equivalents, marketable securities and reverse repurchase agreements |
| $ | 102,341 |
| $ | 86,202 |
| 18.7 | % | |||||||||
Total cash, cash equivalents and marketable securities |
| $ | 80,317 |
| $ | 88,303 |
| (9.0) | % | |||||||||
Borrowings: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loans payable, net |
| $ | 40,068 |
| $ | 39,710 |
| 0.9 | % |
| $ | 38,093 |
| $ | 40,188 |
| (5.2) | % |
Working capital: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets |
| $ | 110,945 |
| $ | 89,019 |
| 24.6 | % |
| $ | 98,776 |
| $ | 101,542 |
| (2.7) | % |
Current liabilities |
|
| 21,408 |
|
| 5,684 |
| >100.0 | % |
|
| 23,301 |
|
| 34,688 |
| (32.8) | % |
Total working capital |
| $ | 89,537 |
| $ | 83,335 |
| 7.4 | % |
| $ | 75,475 |
| $ | 66,854 |
| 12.9 | % |
We have incurred significant net losses since our inception. These losses have resulted principally from costs incurred in connection with in-licensing of our producttherapeutic candidates, research and development activities and selling, general and administrative costs associated with our operations. As of SeptemberJune 30, 2016,2017, we had an accumulated deficit of $162.3$208.8 million and cash, cash equivalents and short-term investments of $102.3$80.3 million.
We expect to continue to incur operating losses for the next several years as we work to develop and commercialize our producttherapeutics and therapeutic candidates. As a result,If we expect tocannot generate sufficient cash from operations in the future, we may seek to fund our operations through corporate collaborations and licensing arrangements, as well as public or private equity offerings or further debt (re)financings. If we are not able to raise additional capital on terms acceptable to us, or at all, as and when needed, we may be required to curtail our operations which could include delaying the commercial launch of our products,therapeutics, discontinuing producttherapeutic development programs, or granting rights to develop and market productstherapeutics or producttherapeutic candidates that we would otherwise prefer to develop and market ourselves. Our failure to raise capital as and when needed would have a negative impact on our financial condition and our ability to pursue our business strategies as this capital is necessary for us to perform the research and development and commercial activities required to generate future revenue streams. Arrangements with collaborators or others may require us to relinquish rights to certain of our technologies or product candidates. In addition, there is a risk that we may never successfully complete development of our product candidates, obtain adequate patent protection for our technology, obtain necessary regulatory approval for our product candidates or achieve commercial viability for any approved product candidates. As disclosed in Note 87 to theour consolidated financial statements, we have a term loan and a revolving credit facility with an aggregate principal balance of $40.0$37.7 million as of SeptemberJune 30, 2016.2017. The terms of the loan agreement require us to receive unrestricted net cash proceeds of at least $45.0 million from partnering transactions and/or the issuance of equity securities from October 16, 2015 to October 16, 2016. The loan agreement also requires that we have at least three products fully USDA-or FDA-approved for commercialization by December 31, 2016. With the FDA approval of GALLIPRANT in March 2016 and the receipt of the upfront payment of $45.0 million under the Collaboration Agreement with Elanco entered into in April 2016, we have met both conditions. Additionally, the loan agreement requires that we maintain certain minimum liquidity at all times (the greater of cash equal to fifty percent (50%) of outstanding credit extensionsbalance or remaining months’ liquidity, which is calculated on an average trailing three (3) month basis, equal to six (6) months or greater,greater), which as of SeptemberJune 30, 2016,2017, was approximately $38.6$22.5 million. Due to the increase in cash used as a result of milestone and inventory purchases for upcoming launches, the third quarter cash usage increased resulting in an increase inIf the minimum liquidity requirement. is not met, we may be required to repay the loans prior to their scheduled maturity dates. At June 30, 2017, we were in compliance with all financial covenants. As of the date of the filing of this Quarterly Report on Form 10-Q, we believe that our existing cash, and cash equivalents and short-term investments of $102.3$80.3 million as of June 30, 2017, will allow us to fund our operations and our debt obligations for at least one year from the issuance of our consolidated financial statements. Based on our current operating plan, which contemplates the launch of ENTYCE by the fall of 2017, we believe that our cash, cash equivalents and short-term investments will be sufficient to fund our operations and debt obligations through September 30, 2017.at least 2018. Our current operating plan also contemplates continued growth in sales of GALLIPRANT, which we believe will result in the achievement of certain milestones under the Collaboration Agreement.
Cash, Cash Equivalents and Investments
Until required for another use in our business, we typically invest our cash reserves in bank deposits, certificates of deposit, and other interest bearing debt instruments in accordance with our investment policy. It is our policy to mitigate credit risk in our cash reserves and investments by maintaining a well-diversified portfolio that limits the amount of exposure as to institution, maturity, and investment type. The value of our investments, however, may be adversely affected by increases in interest rates, instability in the global financial markets that reduces the liquidity of securities included in our portfolio, and by other factors which may result in declines in the value of the investments. Each of these events may cause us to record charges to reduce the carrying value of our investment portfolio if the declines are other-than-temporary or sell investments for less than our acquisition cost which could adversely impact our financial position and our overall liquidity.
3023
At-the-Market Offering
On October 16, 2015, we entered into athe Sales Agreement (“Sales Agreement”) with Barclays Capital, Inc. (“Barclays”), pursuant to which we maycould sell from time to time, at our option, up to an aggregate of $52.0 million of shares of ourits common stock (the “Shares”) through Barclays, as sales agent.agent (“ATM Program”). Sales of the Shares if any, will bewere made under our previously filed and currentlythen effective Registration Statementregistration statement on Form S-3 (Reg. No. 333-197414), by means of ordinary brokers’ transactions on the NASDAQ Global Market or otherwise. Additionally, under the terms of the Sales Agreement, the Shares maycould be sold at market prices, at negotiated prices or at prices related to the prevailing market price. We will paypaid Barclays a commission of 2.75% of the gross proceeds from the sale of the Shares.
During the quarterthree and six months ended SeptemberJune 30, 2016,2017, we agreed to sell 1,629,408sold 305,372 and 546,926 Shares of which 1,416,052 Shares settled during the third quarter of 2016 for aggregate net proceeds of $12.7$1.6 million and $2.8 million, respectively.
On April 28, 2017, we terminated our Sales Agreement. Prior to termination, we sold approximately $18.0 million of which $0.6the $52.0 million was received by us after September 30, 2016. Subsequentavailable to September 30, 2016, the remaining 213,356 Shares settled for aggregate net proceeds of $1.9 million. As of the date of this filing, approximately $37.0 million of Shares remained available for salebe sold under the Sales Agreement. We haveterminated the Sales Agreement because we did not intend to raise additional capital through the ATM Program, and no additional shares of our common stock were sold pursuant to the Sales Agreement. We did not incur any termination penalties as a result of our termination of the Sales Agreement.
Registered Direct Offering
On May 3, 2017, we entered into a Placement Agency Agreement (“PAA”) with Barclays, pursuant to which Barclays agreed to sell any additional Shares since September 30, 2016. serve as placement agent for an offering of shares of common stock. In conjunction with the PAA, on May 3, 2017, we also entered into a Securities Purchase Agreement with certain investors for the sale by us of 5,000,000 shares of common stock at a purchase price of $5.25 per share (the “Offering”). The shares of common stock were offered and sold pursuant to our previously filed and then effective registration statement on Form S-3 (File No. 333-197414) and a related prospectus supplement. We agreed to pay Barclays an aggregate fee equal to 6.0% of the gross proceeds received by us from the Offering. The Offering closed on May 9, 2017. We received aggregate net proceeds from the Offering of approximately $24.4 million, after deducting placement agent fees of $1.6 million and estimated offering expenses of $0.3 million.
Indebtedness
On October 16, 2015, we and Vet Therapeutics (together the “Borrowers”) entered into a Loan and Security Agreement, (“Loanas amended on February 24, 2017 (the “Loan Agreement”) with Pacific Western Bank (“Pacific Western Bank”Western”) as collateral agent (“Collateral Agent”) and a lender and Oxford Finance LLC as a lender (“Oxford” and together with Pacific Western, Bank, the “Lenders”), pursuant to which the Lenders agreed to make available to the Borrowers, a term loansloan in an aggregate principal amount up to $35.0 million (the “Term Loan”), and a revolving credit facility in an aggregate principal amount up to $5.0 million (the “Revolving Line” and together with the Term Loan, the “Credit Extensions”), subject to certain conditions to funding. A term loan was made on October 16, 2015 in an aggregate principal amount equal to $35.0 million, and an advance under the Revolving Line was made on October 16, 2015 in an aggregate principal amount equal to $5.0 million. The Borrowers arewere required to make interest-only payments on the Term Loan for 18 months, and beginning on May 1, 2017, are requiredbegan to make payments of principal and accrued interest on the Term Loan in equal monthly installments over a term of 30 months. The interest-only period can be extended by one year to May 1, 2018 ifTerm Loan and the Borrowers have at least four products fully USDA- or FDA-approved, plus another product conditionally- or fully-approved, in each case for commercialization by December 31, 2016, and agree to certain other financial covenants with the Lenders. The Credit ExtensionsRevolving Line bear interest per annum at the greater of (i) 6.91% or (ii) 3.66% plus the prime rate, which is customarily defined. AllUnder the Loan Agreement, all principal and accrued interest on the Term Loan arewere due on October 16, 2019 (the “Term Loan Maturity Date”), and all principal and accrued interest on the Revolving Line arewere due on October 16, 2017 (the “Revolving Maturity Date”).
The Borrowers used approximately $15.0 million of the proceeds from the Credit Extensions to repay all the amounts owed under their Loan and Security Agreement, dated as of March 4, 2013, as amended, with Pacific Western Bank (as successor in interest to Square 1 Bank) and the lenders party thereto.
As security for their obligations under the Loan Agreement, the Borrowers granted a security interest in substantially all of their existing and after-acquired assets except for their intellectual property and certain other customary exclusions. Subject to customary exceptions, the Borrowers are not permitted to encumber their intellectual property.
Upon execution of the Loan Agreement, the Borrowers were obligated to pay a facility fee to the Lenders of $0.2 million and an agency fee to the Collateral Agent of $0.1 million. In addition, the Borrowers are or will be obligated to pay a final payment fee equal to 3.30% of such Term Loan being prepaid or repaid with respect to the Term LoansLoan upon the earliest to occur of the Term Loan Maturity Date, the acceleration of any Term Loan or the prepayment of a Term Loan. The Borrowers will also be obligated to pay a termination fee equal to 3.30% of the highest outstanding amount of the Revolving Line with respect to the Revolving Line upon the earliest to occur of the Prior Revolving Maturity Date, the acceleration of the Revolving Line or the termination of the Revolving Line. The Borrowers will also be obligated to pay an unused-line fee equal to 0.25% per annum of the average unused portion of the Revolving Line.
The Loan Agreement contains customary representations and warranties and customary affirmative and negative covenants, including, among others, limits or restrictions on the Borrowers’ ability to incur liens, incur indebtedness, make certain restricted payments, make certain investments, merge, consolidate, make an acquisition, enter into certain licensing arrangements and dispose of certain assets. In addition, the Loan Agreement contains customary events of default that entitle the Lenders to cause the Borrowers’ indebtedness under the Loan Agreement to become immediately due and payable. The events of default, some of which are subject to cure periods, include, among others, a non-payment default, a covenant default, the occurrence of a material adverse change, the occurrence of an insolvency, a material judgment default, defaults regarding other indebtedness and certain actions by governmental authorities. Upon the occurrence and for the duration of an event of default, an additional default interest rate equal to 4% per annum will apply to all obligations owed under the Loan Agreement.
31
The Loan Agreement requires that the Borrowers receive unrestricted net cash proceeds of at least $45.0 million from partnering transactions and/or the issuance of equity securities from October 16, 2015 to October 16, 2016. The Loan Agreement also requires that the Borrowers have at least three products fully USDA- or FDA approved for commercialization by December 31, 2016. With the FDA approval of GALLIPRANT in March 2016 and the receipt of the upfront payment of $45.0 million under the Collaboration Agreement with Elanco entered into in April 2016, we have met both conditions. Additionally, the Loan Agreement requires that we maintain certain minimum liquidity at all times, which as of SeptemberJune 30, 2016,2017, was approximately $38.6$22.5 million. If the minimum liquidity requirement is not met, the Borrowers may be required to repay the loans prior to their
24
scheduled maturity dates. At SeptemberJune 30, 2016,2017, the Borrowers were in compliance with all financial covenants.covenants, including the minimum liquidity covenant.
Effective as of July 31, 2017, we amended the Loan Agreement (“Second Amendment”). The terms of the Second Amendment, among other things, extend the maturity of the Revolving Line to October 16, 2019 (the “Revolving Line Maturity Date”), with amortized equal repayments of the principal outstanding under the Revolving Line beginning November 1, 2018, and provide a six (6) month interest only period for the Term Loan, starting on the date of the Second Amendment.
We are not subject to any new financial covenants as a result of the Second Amendment. At the closing of the Second Amendment, we paid the Lenders an amendment fee of $0.2 million and a facility fee of $0.1 million. We are obligated to pay a new termination fee equal to $0.2 million upon the earliest to occur of the Revolving Line Maturity Date, the acceleration of the Revolving Line or the termination of the Revolving Line. The existing termination fee of $0.2 million will continue to be payable upon the earliest to occur of the original revolving maturity date (October 16, 2017), the acceleration of the Revolving Line or the termination of the Revolving Line.
Working Capital
We define working capital as current assets less current liabilities. The increase in working capital at June 30, 2017, from December 31, 2015,2016, reflects an increasea decrease in total current assets of $21.9$2.8 million and an increasea decrease in total current liabilities of $15.7$11.4 million. The increasedecrease in total current assets was primarily driven by an increasea decrease in cash and cash equivalents due to the receipt of the $45.0 million upfront payment under the Collaboration Agreementpayments for our research and receipt of $12.7 million from issuance of common stock in 2016.development activities related to our programs, payments for inventories, milestones and selling, general and administrative expenses. The increasedecrease in total current liabilities was primarily a result of increases inpayments for ENTYCEGALLIPRANT inventories and NOCITA inventories, an increasea decrease in the current portion of loans payable and an increase in the licensing and collaboration commitment.due to amending payment terms under our Loan Agreement on July 31, 2017.
Cash Flows
SummaryA summary of our cash flows for the ninesix months ended SeptemberJune 30, 2017 and 2016, and 2015, wasis as follows:
|
|
|
|
|
|
|
|
| Nine Months Ended | ||||
|
| September 30, | ||||
|
| 2016 |
| 2015 | ||
|
| (Dollars in thousands) | ||||
Net cash provided by (used in) operating activities |
| $ | 5,248 |
| $ | (28,104) |
Net cash provided by investing activities |
| $ | 56,928 |
| $ | 34,602 |
Net cash provided by (used in) financing activities |
| $ | 11,864 |
| $ | (2,689) |
|
|
|
|
|
|
|
|
| Six Months Ended | ||||
|
| June 30, | ||||
|
| 2017 |
| 2016 | ||
|
| (Dollars in thousands) | ||||
Net cash provided by (used in) operating activities |
| $ | (29,922) |
| $ | 23,287 |
Net cash provided by (used in) investing activities |
| $ | (3,509) |
| $ | 58,681 |
Net cash provided by financing activities |
| $ | 24,926 |
| $ | 1 |
Net cash provided by (used in) operating activities
During the ninesix months ended SeptemberJune 30, 2016,2017, net cash provided byused in operating activities was $5.2$29.9 million. We had a net loss of $10.2$23.0 million which includes an adjustment of a non-cash expense for stock-based compensation of $6.5$3.7 million, a non-cash depreciation and amortization expense of $0.7 million, a non-cash impairment of intangible assets of $2.8$0.6 million and a lowernon-cash interest expense of cost or market adjustment to inventories of $1.6$0.2 million. Our net loss was primarily attributed to our research and development activities related to our programs and our selling, general and administrative expenses, partially offset by licensing and collaboration revenues of $38.0$1.7 million from the Collaboration Agreement.Agreement and product sales of $7.2 million. Net cash provided by changesused in our working capital consistedoperating assets and liabilities was primarily of an increasedue to a decrease in accounts payable of $3.7$5.5 million, an increase of $7.0 million in licensing and collaboration commitment under the Collaboration Agreement, partially offset by a decrease in accrued expenses and other liabilities of $0.5$0.7 million, an increase in account receivable, net of $4.8 million, an increase in prepaid expenses and other current assets of $0.9$4.6 million and an increasea decrease in inventories of $5.8$4.2 million. The increase in inventories was primarily related to GALLIPRANT and ENTYCE pre-launch inventories. The increasedecrease in accounts payable was primarily related to ENTYCEpayments for GALLIPRANT inventories and trade payables. The increase in prepaid expensesaccounts receivable, net and other current assets wasdecrease in inventories were primarily related to GALLIPRANT sales. Also, accounts receivable, net increased due advance payments made for our outsourced research and development activities.to receivables from the Elanco GALLIPRANT Agreements.
During the ninesix months ended SeptemberJune 30, 2015,2016, net cash used inprovided by operating activities was $28.1$23.3 million. We had a pretax lossnet income of $72.6$3.1 million which includes a gain on sale of marketable securities of $3.9 million, an adjustment of a non-cash expense for stock-based compensation of $6.5$4.5 million, a non-cash depreciation and amortization expense of $1.6 million, and an impairment of intangible assets loss of $43.4 million, partially offset by a non-cash change in fair value of contingent consideration of $1.2$0.5 million, a non-cash change in fair valueimpairment of derivative instrumentsacquired intangible assets expense of $1.3$2.8 million, and a non-cash deferred income tax benefitlower of $0.8cost or market adjustment to inventories of $1.6 million. Our net losses wereincome was primarily attributed to impairmentlicensing and collaboration revenues of intangible assets, to$38.0 million from the Collaboration Agreement, research and development activities related to our programs and our selling, general and administrative expenses. Net cash usedprovided by changes in our working capitaloperating assets and liabilities consisted primarily of an increase in inventories of $0.7 million, a decrease in accounts payable of $0.3$5.9 million, an increase of $7.0 million in prepaid expenses of $0.2 million, partially offset bylicensing and collaboration commitment under the Collaboration Agreement and an increase in accrued expenses and other liabilities of $0.5$0.6 million, partially offset by an increase in prepaid expenses of $0.1 million and an increase in inventories of $2.8 million. The decreaseincrease in inventories was primarily related to GALLIPRANT pre-launch inventories. The increase in accounts payable was primarily related to the timing of payments made for our outsourced researchachieved milestones relating to GALLIPRANT, ENTYCE and development activities.NOCITA. The increase in accrued expenses and other current liabilities was primarily due to the licensing and collaboration commitment related to the Collaboration Agreement.
Net cash provided by (used in) investing activities
During the ninesix months ended SeptemberJune 30, 2017, net cash used in investing activities was $3.5 million, which primarily consisted of a $3.0 million milestone payment for intangible assets for currently marketed products and the purchases of investments of $2.0 million,
25
partially offset by proceeds from the maturities and sales of investments of $1.5 million.
During the six months ended June 30, 2016, net cash provided by investing activities was $56.9$58.7 million, which primarily consisted of the proceeds from the maturities and sales of investmentsmarketable securities of $286.8$286.0 million, partially offset by the purchases of investments of $228.8 million and $1.0 million milestone payments for intangible assets for currently marketed products.
32
During the nine months ended September 30, 2015, net cash provided by investing activities was $34.6 million, which related to the proceeds from the maturities and sales of marketable securities and investments of $1,629.2 million, partially offset by the purchases of investments of $1,592.7 million and $1.9 million for purchases of property and equipment.$227.3 million.
Net cash provided by (used in) financing activities
During the ninesix months ended SeptemberJune 30, 2016,2017, net cash provided by financing activities was $11.9$24.9 million. Net cash provided by financing activities consisted of the net proceeds from the issuance of common stock of $12.1$27.5 million, partially offset by $2.3 million of payments on loans payable and $0.3 million of payments for stock issuance costs, and the proceeds of stock option exercises of $0.1 million.costs.
During the ninesix months ended SeptemberJune 30, 2015,2016, net cash used inprovided by financing activities was $2.7 million. Net cash used in financing activities primarily resulted from cash paid for contingent considerationconsisted solely of $3.0 million to the former shareholders of Vet Therapeutics, Inc., partially offset by cash receivedproceeds from stock option exercises of $0.3 million.$1,000.
Contractual Obligations and Off-Balance Sheet Arrangements
Contractual Obligations
Our contractual obligations primarily consist of our obligations under our loans payable, non-cancellable operating leases, minimum royalties and other purchase obligations, excluding amounts related to other funding commitments, contingent development, regulatory and commercial milestone payments, contract manufacturer commitments and off-balance sheet arrangements as described below. As of SeptemberJune 30, 2016,2017, there were no material changes in our contractual obligations since December 31, 2015,2016, except for the contract manufacturer commitments described below.
Other Funding Commitments
As of SeptemberJune 30, 2016,2017, we have several on-goingongoing development programs in various stages of the regulatory process. Our most significant expenditures are to clinical research and contract manufacturing organizations. The contracts are generally cancellable, with notice, at our option.
Contingent Development, Regulatory and Commercial Milestone Payments
Based on our development plans as of SeptemberJune 30, 2016,2017, we have committed to make potential future milestone payments to third parties of up to approximately $112.1$108.6 million, of which $80.4$77.4 million are for commercial milestones, as part of our various collaborations, including licensing and development programs. Approximately $68.9 million of the commercial milestones relate to the achievement of various sales thresholds. During the first nine months of 2016, we achieved milestones in the amount of $0.3 million that as of September 30, 2016, had not been paid and are not included in the $112.1 million amount. Payments under these agreements generally become due and payable only upon achievement of certain development, regulatory or commercial milestones. Because the achievement of these milestones had not occurred or was not considered probable as of SeptemberJune 30, 2016,2017, such contingencies have not been recorded in our consolidated financial statements, except for $0.5 million due to our former commercial licensee of BLONTRESS.
Including the $0.3 million of unpaid milestones achieved year to date, weWe anticipate that we may pay approximately $0.8$0.6 million and $6.9$4.0 million of milestone payments during the remainder of 2016,2017, and the next 12 months, respectively, provided various development, regulatory or commercial milestones are achieved. Amounts related to contingent milestone payments are not considered contractual obligations as they are contingent on the successful achievement of certain development, regulatory approval and commercial milestones that may not be achieved.
Contract Manufacturer Commitments
Our independent contract manufacturersCMOs manufacture our products and product components based on our forecasts. These forecasts are based on estimates of future demand for our products, which are in turn based on available historical trends and an analysis from sales and product marketing organizations, adjusted for overall market conditions. In order to reduce manufacturing lead times and plan for adequate supply, we may issue forecasts and orders for components and products that are non-cancelable. As of SeptemberJune 30, 20162017 and December 31, 2015,2016, we had non-cancellable open orders for the purchase of inventories of $21.8$27.7 million and $0$17.8 million, respectively.
Off-Balance Sheet Arrangements
We have not engaged in the use of any off-balance sheet arrangements, such as structured finance entities or special purpose entities.
Recently Issued and Adopted Accounting Pronouncements
For a discussion of new accounting standards please read Note 1, “Summary of Significant Accounting Policies – New Accounting StandardsStandards” to our consolidated financial statements included within this report.
3326
Item 3. Quantitative and Qualitative Disclosures About Market Risk
Our market risks, and the ways we manage them are summarized in Part II, Item 7A, “Quantitative and Qualitative Disclosures About Market Risk” of our Annual Report on Form 10-K for the fiscal year ended December 31, 2015,2016, filed with the SEC on March 15, 2016.14, 2017. As of SeptemberJune 30, 20162017, there were no material changes to our market risks or management of such risks since December 31, 2015.2016.
Item 4. Controls and Procedures
Limitations on Effectiveness of Controls and Procedures
In designing and evaluating our disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints and that management is required to apply judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated, as of the end of the period covered by this Quarterly Report on Form 10-Q, the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”))amended). Based on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of SeptemberJune 30, 2016.2017.
Changes in Internal Control over Financial Reporting
There was no change in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Securities Exchange Act)Act of 1934, as amended) identified in connection with the evaluation of our internal control performed during the fiscal quarter ended SeptemberJune 30, 2016,2017, that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
3427
WeFrom time to time, we may become involved in legal proceedings arising in the ordinary course of our business. Except as described below, we are not currentlypresently a party to any litigation that we believe to be material legal proceedings.and we are not aware of any pending or threatened litigation against us that we believe could have a material adverse effect on our business, operating results, financial condition or cash flows.
In February 2017, two purported class action lawsuits were filed in the United States District Court for the Southern District of New York against the Company and two of its current officers. Those cases have been consolidated into one purported class action lawsuit under the caption, In re Aratana Therapeutics, Inc. Securities Litigation, Case No. 1:17-cv-00880. The consolidated lawsuit asserts claims under Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, and is premised on allegedly false and/or misleading statements, and alleged non-disclosure of material facts, regarding the Company’s business, operations, prospects and performance during the proposed class period of March 16, 2015 to February 3, 2017. The Company intends to vigorously defend all claims asserted. Given the early stage of the litigation, at this time a loss is not probable or reasonably estimable.
Our business faces significant risks and uncertainties, which may have a material adverse effect on our business prospects, financial condition and results of operations, and you should carefully consider them.
Apart from the below, thereThere have been no material changes in the six months ended June 30, 2017, to the risk factors described in Part I, Item 1A of our Annual Report on Form 10-K for the fiscal year ended December 31, 2015.
Our Collaboration Agreement and Co-Promotion Agreement with Elanco are important to our business. If we or Elanco fail to adequately perform under the Collaboration Agreement and/or the Co-Promotion Agreement, or if we or Elanco terminate the Collaboration Agreement and/or the Co-Promotion Agreement, the development and commercialization of GALLIPRANT and any other grapiprant product candidates would be delayed or terminated and our business would be adversely affected.2016.
The Collaboration Agreement and Co-Promotion Agreement are important to our business, and our ability to develop and commercialize GALLIPRANT and any grapiprant product candidates is dependent upon these agreements.
The Collaboration Agreement may be terminated by Elanco at any time upon 90 days’ written notice to us. The Collaboration Agreement may also be terminated by either party:
|
|
|
|
|
|
Upon Elanco’s voluntary termination or termination for Elanco’s breach, among other things, all licenses and rights granted to Elanco will terminate and revert to us, and Elanco has agreed to assign to us all registrations and trademarks obtained in connection with the products covered by the agreement. Upon termination for our breach, among other things, Elanco may elect to retain its rights to the licenses granted by us under the Collaboration Agreement subject to specified payment obligations.
Elanco may terminate the Co-Promotion Agreement in the event Elanco substantially stops marketing the products covered by the Collaboration Agreement, and either party may terminate the Co-Promotion Agreement upon the other party’s material breach, where such breach is not cured within the timeframe specified by the Co-Promotion Agreement. In addition, the Co-Promotion Agreement provides that it will automatically terminate if the Collaboration Agreement is terminated early.
Termination of the Collaboration Agreement and/or the Co-Promotion Agreement could cause significant delays in our product development and commercialization efforts that could prevent us from commercializing our grapiprant product candidates, including GALLIPRANT, without first expanding our internal capabilities, securing additional financing or entering into another agreement with a third party. Any alternative collaboration or license could also be on less favorable terms to us.
Under the Collaboration Agreement, among other things, we are responsible for the manufacture and supply of all of Elanco’s reasonable requirements of the products covered by the agreement. If we are unable to meet our manufacture and supply obligations, Elanco may claim that we have materially breached the Collaboration Agreement and terminate such agreement, which could adversely affect our business and our ability to successfully develop and commercialize any products covered by the agreement, including GALLIPRANT.
Under the Collaboration Agreement, Elanco has agreed to provide funding for certain clinical development activities. If the Collaboration Agreement were terminated, we may need to seek additional financing to support the research and development of any terminated products or discontinue any terminated products, which could adversely affect our business. In addition, under the Collaboration Agreement, Elanco is solely responsible for commercializing products outside the United States. We cannot directly control Elanco’s commercialization activities or the resources it allocates to our product candidates. Our interests and Elanco’s interests may differ or conflict from time to time, or we may disagree with Elanco’s level of effort or resource allocation. Elanco may internally prioritize our product candidates differently than we do or it may not allocate sufficient resources to effectively or optimally commercialize them. If these events were to occur, our business would be adversely affected.
35
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds
Unregistered Sales of Equity Securities
None.
Repurchases of Common Stock
There were no share repurchases during the three months ended September 30, 2016.None.
Item 3. Defaults Upon Senior Securities
None.
Item 4. Mine Safety Disclosures
Not applicable.
None.On July 31, 2017, we and Vet Therapeutics, Inc. (“the Borrowers”) entered into a Second Amendment (the “Second Amendment”) to the Loan and Security Agreement dated as of October 16, 2015, as previously amended (the “Loan Agreement”) with Pacific Western Bank, in its capacity as collateral agent (in such capacity, “Collateral Agent”), and the Lenders party thereto.
The terms of the Second Amendment, among other things, extend the maturity of the existing revolving credit facility (the “Revolving Line”) to October 16, 2019 (the “Revolving Line Maturity Date”), with amortized equal repayments of the principal outstanding under the revolving credit facility beginning November 1, 2018, and provide a six (6) month interest only period for the term loans, starting on the date of the Second Amendment.
We are not subject to any new financial covenants as a result of the Second Amendment. At the closing of the Second Amendment, we paid the Lenders an amendment fee of $150,000 and a facility fee of $60,000. We will also be obligated to pay a new termination fee equal to $165,000 upon the earliest to occur of the Revolving Line Maturity Date, the acceleration of the revolving credit facility or the termination of the revolving credit facility. The existing termination fee of $165,000 will continue to be payable upon the earliest to occur of the original revolving maturity date (October 16, 2017), the acceleration of the revolving credit facility or the termination of the revolving credit facility.
The foregoing description of the Second Amendment is a summary, and is qualified in its entirety by reference to the Second Amendment, which is filed herewith as Exhibit 10.4, and is incorporated herein by reference.
A list of exhibits is set forth on the Exhibit Index immediately following the signature page of this Quarterly Report on Form 10-Q, and is incorporated herein by reference.
3628
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ARATANA THERAPEUTICS, INC. | ||
|
|
|
| |||
Date: |
|
|
| By: |
| /s/ Steven St. Peter |
|
|
|
|
|
| Steven St. Peter, M.D. |
|
|
|
|
|
| President and Chief Executive Officer |
|
|
|
|
|
| (Principal Executive Officer) |
|
|
|
| |||
Date: |
|
|
| By: |
| /s/ Craig Tooman |
|
|
|
|
|
| Craig Tooman |
|
|
|
|
|
| Chief Financial Officer |
|
|
|
|
|
| (Principal Financial and Accounting Officer) |
3729
Exhibit Index
|
|
|
|
|
|
| |
|
| Incorporated by Reference |
| ||||
Exhibit Number | Exhibit Description | Form | File No. | Exhibit | Filing Date | Filed/ Furnished Herewith | |
3.1 | Restated Certificate of Incorporation | 8-K | 001-35952 | 3.1 | 7/3/13 |
| |
3.2 | Amended and Restated Bylaws | 8-K | 001-35952 | 3.2 | 7/3/13 |
| |
10.1 | Consulting and Separation Agreement, dated as of August 30, 2016 between Aratana Therapeutics, Inc. and Julia Stephanus | 8-K | 001-35952 | 10.1 | 9/1/16 |
| |
31.1 | Certification of Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| * | |
31.2 | Certification of Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| * | |
32.1 | Certification of Principal Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| ** | |
32.2 | Certification of Principal Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| ** | |
101.INS | XBRL Instance Document |
|
|
|
|
| |
101.SCH | XBRL Taxonomy Extension Schema Document |
|
|
|
|
| |
101.CAL | XBRL Taxonomy Extension Calculation Linkbase |
|
|
|
|
| |
101.DEF | XBRL Taxonomy Extension Definition Linkbase |
|
|
|
|
| |
101.LAB | XBRL Taxonomy Extension Label Linkbase |
|
|
|
|
| |
101.PRE | XBRL Taxonomy Extension Presentation Linkbase |
|
|
|
|
| |
|
|
|
|
|
|
| |||
|
|
|
|
|
|
| |||
|
| Incorporated by Reference |
| ||||||
Exhibit Number | Exhibit Description | Form | File No. | Exhibit | Filing Date | Filed/ Furnished Herewith | |||
3.1 | Restated Certificate of Incorporation | 8-K | 001-35952 | 3.1 | 7/3/13 |
| |||
3.2 | Amended and Restated Bylaws | 8-K | 001-35952 | 3.2 | 7/3/13 |
| |||
10.1 | Securities Purchase Agreement, dated May 3, 2017, by and among Aratana Therapeutics, Inc. and the investors party thereto | 8-K | 001-35952 | 10.1 | 5/4/17 |
| |||
10.2 | Placement Agency Agreement, dated May 3, 2017, by and between the Company and Barclays Capital, Inc. | 8-K | 001-35952 | 10.2 | 5/4/17 |
| |||
10.3+ | Amendment, effective as of April 28, 2017, to the Collaboration, License, Development and Commercialization Agreement, dated April 22, 2016, by and between the Company and Eli Lilly and Company, operating on behalf of its Elanco Animal Health Division |
|
|
|
| * | |||
10.4 | Second Amendment to Loan and Security Agreement, dated as of July 31, 2017, by and among Pacific Western Bank, in its capacity as collateral agent and the Lenders party thereto. |
|
|
|
| * | |||
31.1 | Certification of Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| * | |||
31.2 | Certification of Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| * | |||
32.1 | Certification of Principal Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| ** | |||
32.2 | Certification of Principal Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| ** | |||
101.INS | XBRL Instance Document |
|
|
|
|
| |||
101.SCH | XBRL Taxonomy Extension Schema Document |
|
|
|
|
| |||
101.CAL | XBRL Taxonomy Extension Calculation Linkbase |
|
|
|
|
| |||
101.DEF | XBRL Taxonomy Extension Definition Linkbase |
|
|
|
|
| |||
101.LAB | XBRL Taxonomy Extension Label Linkbase |
|
|
|
|
| |||
101.PRE | XBRL Taxonomy Extension Presentation Linkbase |
|
|
|
|
| |||
* Filed herewith.
** Furnished herewith and not “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended.
+ Confidential treatment has been requested with respect to certain portions of this exhibit, which portions have been filed separately with the Securities and Exchange Commission.
3830