UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
____________________________
FORM 10-Q
_____________________________
(Mark One)
| | | | | |
| x | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended September 30, 2023March 31, 2024
Or
| | | | | |
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 001-37524
_____________________________
vTv Therapeutics Inc.
(Exact name of registrant as specified in its charter)
_____________________________
| | | | | |
| Delaware | 47-3916571 |
(State or other jurisdiction of
incorporation or organization) | (I.R.S. Employer
Identification No.) |
| |
3980 Premier Dr, Suite 310 High Point, NC | 27265 |
| (Address of principal executive offices) | (Zip Code) |
(336) 841-0300
(Registrant’s telephone number, including area code)
(Former name, former address and former fiscal year, if changed since last report)
_____________________________
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Class A common stock, par value $0.01 per share | VTVT | NASDAQ Capital Market |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | | | | |
| Large accelerated filer | o | | Accelerated filer | o |
| Non-accelerated filer | o | | Smaller reporting company | x |
| Emerging growth company | o | | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes o No x
| | | | | | | | |
| Class of Stock | | Shares Outstanding as of NovemberMay 9, 20232024 |
| Class A common stock, par value $0.01 per share | | 81,483,6002,432,857 | |
| Class B common stock, par value $0.01 per share | | 23,093,860577,349 | |
vTv THERAPEUTICS INC. AND SUBSIDIARIES
INDEX TO FORM 10-Q
FOR THE QUARTER ENDED SEPTEMBER 30, 2023MARCH 31, 2024
PART I – FINANCIAL INFORMATION
The financial statements and other disclosures contained in this report include those of vTv Therapeutics Inc. (“we”, the “Company” or the “Registrant”), which is the registrant, and those of vTv Therapeutics LLC (“vTv LLC”), which is the principal operating subsidiary of the Registrant. Unless the context suggests otherwise, references in this Quarterly Report on Form 10-Q to the “Company”, “we”, “us” and “our” refer to vTv Therapeutics Inc. and its consolidated subsidiaries.
vTv Therapeutics Inc.
Condensed Consolidated Balance Sheets
(in thousands, except number of shares and per share data)
| | September 30,
2023 | | December 31,
2022 |
| (Unaudited) | | |
| March 31,
2024 | | | March 31,
2024 | | December 31,
2023 |
| (Unaudited) | |
| Assets | |
| Assets | |
| Assets | Assets | |
| Current assets: | Current assets: | |
| Current assets: | |
| Current assets: | |
| Cash and cash equivalents | |
| Cash and cash equivalents | |
| Cash and cash equivalents | Cash and cash equivalents | $ | 8,238 | | | $ | 12,126 | |
| Accounts receivable | Accounts receivable | — | | | 173 | |
| G42 Promissory Note receivable | — | | | 12,243 | |
| | Prepaid expenses and other current assets | |
| Prepaid expenses and other current assets | |
| Prepaid expenses and other current assets | Prepaid expenses and other current assets | 1,872 | | | 2,537 | |
| Current deposits | Current deposits | 15 | | | 15 | |
| Total current assets | Total current assets | 10,125 | | | 27,094 | |
| Property and equipment, net | Property and equipment, net | 140 | | | 207 | |
| Operating lease right-of-use assets | Operating lease right-of-use assets | 272 | | | 349 | |
| Long-term investments | 4,387 | | | 5,588 | |
| | Total assets | Total assets | $ | 14,924 | | | $ | 33,238 | |
| Liabilities, Redeemable Noncontrolling Interest and Stockholders’ Deficit | | | |
| Total assets | |
| Total assets | |
| Liabilities, Redeemable Noncontrolling Interest and Stockholders’ Equity (Deficit) | |
| Current liabilities: | Current liabilities: | |
| Current liabilities: | |
| Current liabilities: | |
| Accounts payable and accrued expenses | |
| Accounts payable and accrued expenses | |
| Accounts payable and accrued expenses | Accounts payable and accrued expenses | $ | 9,620 | | | $ | 7,313 | |
| Current portion of operating lease liabilities | Current portion of operating lease liabilities | 165 | | | 154 | |
| Current portion of contract liabilities | Current portion of contract liabilities | 17 | | | 17 | |
| Current portion of notes payable | Current portion of notes payable | 473 | | | 224 | |
| Total current liabilities | Total current liabilities | 10,275 | | | 7,708 | |
| Contract liabilities, net of current portion | Contract liabilities, net of current portion | 18,669 | | | 18,669 | |
| Operating lease liabilities, net of current portion | Operating lease liabilities, net of current portion | 213 | | | 338 | |
| Warrant liability, related party | Warrant liability, related party | 278 | | | 684 | |
| Total liabilities | Total liabilities | 29,435 | | | 27,399 | |
| Commitments and contingencies | Commitments and contingencies | | Commitments and contingencies | | | |
| Redeemable noncontrolling interest | Redeemable noncontrolling interest | 10,722 | | | 16,579 | |
| Stockholders’ deficit: | |
| Class A common stock, $0.01 par value; 200,000,000 shares authorized, 81,483,600 shares outstanding as of September 30, 2023 and December 31, 2022 | 815 | | | 815 | |
| Class B common stock, $0.01 par value; 100,000,000 shares authorized, and 23,093,860 outstanding as of September 30, 2023 and December 31, 2022 | 232 | | | 232 | |
| Stockholders’ equity (deficit): | |
| Class A common stock, $0.01 par value; 200,000,000 shares authorized, 2,432,857 and 2,084,973 shares outstanding as of March 31, 2024 and December 31, 2023, respectively | |
| Class A common stock, $0.01 par value; 200,000,000 shares authorized, 2,432,857 and 2,084,973 shares outstanding as of March 31, 2024 and December 31, 2023, respectively | |
| Class A common stock, $0.01 par value; 200,000,000 shares authorized, 2,432,857 and 2,084,973 shares outstanding as of March 31, 2024 and December 31, 2023, respectively | |
| Class B common stock, $0.01 par value; 100,000,000 shares authorized, and 577,349 outstanding as of March 31, 2024 and December 31, 2023 | |
| | Additional paid-in capital | |
| Additional paid-in capital | |
| Additional paid-in capital | Additional paid-in capital | 254,912 | | | 253,737 | |
| Accumulated deficit | Accumulated deficit | (281,192) | | | (265,524) | |
| Total stockholders’ deficit attributable to vTv Therapeutics Inc. | (25,233) | | | (10,740) | |
| Total liabilities, redeemable noncontrolling interest and stockholders’ deficit | $ | 14,924 | | | $ | 33,238 | |
| Total stockholders’ equity (deficit) attributable to vTv Therapeutics Inc. | |
| Noncontrolling interest | |
| Total stockholders’ equity (deficit) | |
| Total liabilities, redeemable noncontrolling interest and stockholders’ equity (deficit) | |
The accompanying notes are an integral part of the unaudited condensed consolidated financial statements.
vTv Therapeutics Inc.
Condensed Consolidated Statements of Operations - Unaudited
(in thousands, except number of shares and per share data)
| | Three Months Ended
September 30, | | Nine Months Ended
September 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| Three Months Ended
March 31, | |
| Three Months Ended
March 31, | |
| Three Months Ended
March 31, | |
| 2024 | |
| 2024 | |
| 2024 | |
| Revenue | |
| Revenue | |
| Revenue | Revenue | $ | — | | | $ | — | | | $ | — | | | $ | 2,009 | |
| Operating expenses: | Operating expenses: | |
| Operating expenses: | |
| Operating expenses: | |
| Research and development | |
| Research and development | |
| Research and development | Research and development | 2,824 | | | 3,055 | | | 11,457 | | | 8,393 | |
| General and administrative | General and administrative | 2,544 | | | 2,634 | | | 9,338 | | | 9,813 | |
| General and administrative | |
| General and administrative | |
| Total operating expenses | |
| Total operating expenses | |
| Total operating expenses | Total operating expenses | 5,368 | | | 5,689 | | | 20,795 | | | 18,206 | |
| Operating loss | Operating loss | (5,368) | | | (5,689) | | | (20,795) | | | (16,197) | |
| Other (expense) income, net | (3,640) | | | 403 | | | (1,514) | | | (2,998) | |
| Other income (expense) – related party | 341 | | | (324) | | | 406 | | | 221 | |
| Operating loss | |
| Operating loss | |
| Other income, net | |
| Other income, net | |
| Other income, net | |
| Other expense– related party | |
| Other expense– related party | |
| Other expense– related party | |
| Interest income | Interest income | 131 | | | 150 | | | 384 | | | 200 | |
| Interest expense | (4) | | | (8) | | | (6) | | | (9) | |
| Interest income | |
| Interest income | |
| | Loss before income taxes and noncontrolling interest | |
| | Loss before income taxes and noncontrolling interest | |
| | Loss before income taxes and noncontrolling interest | Loss before income taxes and noncontrolling interest | (8,540) | | | (5,468) | | | (21,525) | | | (18,783) | |
| Income tax provision | Income tax provision | — | | | — | | | — | | | 200 | |
| Income tax provision | |
| Income tax provision | |
| Net loss before noncontrolling interest | |
| Net loss before noncontrolling interest | |
| Net loss before noncontrolling interest | Net loss before noncontrolling interest | (8,540) | | | (5,468) | | | (21,525) | | | (18,983) | |
| Less: net loss attributable to noncontrolling interest | Less: net loss attributable to noncontrolling interest | (1,886) | | | (1,207) | | | (4,753) | | | (4,564) | |
| Less: net loss attributable to noncontrolling interest | |
| Less: net loss attributable to noncontrolling interest | |
| Net loss attributable to vTv Therapeutics Inc. | |
| Net loss attributable to vTv Therapeutics Inc. | |
| Net loss attributable to vTv Therapeutics Inc. | Net loss attributable to vTv Therapeutics Inc. | $ | (6,654) | | | $ | (4,261) | | | $ | (16,772) | | | $ | (14,419) | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | Net loss attributable to vTv Therapeutics Inc. common shareholders | $ | (6,654) | | | $ | (4,261) | | | $ | (16,772) | | | $ | (14,419) | |
| Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted | $ | (0.08) | | | $ | (0.05) | | | $ | (0.21) | | | $ | (0.20) | |
| Weighted average number of vTv Therapeutics Inc. Class A common stock, basic and diluted | 81,483,600 | | | 80,490,121 | | | 81,483,600 | | | 72,649,531 | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Weighted average number of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Weighted average number of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Weighted average number of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
| (*) Adjusted retroactively for reverse stock split | |
| (*) Adjusted retroactively for reverse stock split | |
| (*) Adjusted retroactively for reverse stock split | |
The accompanying notes are an integral part of the unaudited condensed consolidated financial statements.
vTv Therapeutics Inc.
Condensed Consolidated Statement of Changes in Redeemable Noncontrolling Interest and Stockholders’ DeficitEquity (Deficit) - Unaudited
(in thousands, except number of shares)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the three months ended September 30, 2023 |
| | | | Class A Common Stock | | Class B Common Stock | | | | | | |
| Redeemable
Noncontrolling
Interest | | | Shares | | Amount | | Shares | | Amount | | Additional
Paid-in
Capital | | Accumulated Deficit | | Total Stockholders' Deficit |
| Balances at June 30, 2023 | $ | 18,879 | | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | 254,479 | | | $ | (280,809) | | | $ | (25,283) | |
| Net loss | (1,886) | | | | — | | | — | | | — | | | — | | | — | | | (6,654) | | | (6,654) | |
| Share-based compensation | — | | | | — | | | — | | | — | | | — | | | 433 | | | — | | | 433 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| Change in redemption value of noncontrolling interest | (6,271) | | | | — | | | — | | | — | | | — | | | — | | | 6,271 | | | 6,271 | |
| Balances at September 30, 2023 | $ | 10,722 | | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | 254,912 | | | $ | (281,192) | | | $ | (25,233) | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the three months ended March 31, 2024 |
| | | | Class A Common Stock | | Class B Common Stock | | | | | | | | | | |
| Redeemable
Noncontrolling
Interest | | | Shares | | Amount | | Shares | | Amount | | Additional
Paid-in
Capital | | Accumulated Deficit | | Total vTv Therapeutics Inc Stockholders’ Equity (Deficit) | | Noncontrolling
Interest | | Total Stockholders'
Equity (Deficit) |
| Balances at December 31, 2023 | $ | 6,131 | | | | 2,084,973 | | | $ | 21 | | | 577,349 | | | $ | 6 | | | $ | 256,335 | | | $ | (281,042) | | | $ | (24,680) | | | $ | — | | | $ | (24,680) | |
| Net loss attributable to vTv Therapeutics Inc. | — | | | | — | | | — | | | — | | | — | | | — | | | (4,865) | | | (4,865) | | | — | | | (4,865) | |
Net loss attributable to redeemable noncontrolling interest(*) | (1,085) | | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | |
| Change in redemption value of redeemable noncontrolling interest | 214 | | | | — | | | — | | | — | | | — | | | — | | | (214) | | | (214) | | | — | | | (214) | |
| Reclassification of redeemable noncontrolling interest to permanent equity (See Note 7) | (5,260) | | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 5,260 | | | 5,260 | |
| Share-based compensation | — | | | | — | | | — | | | — | | | — | | | 220 | | | — | | | 220 | | | — | | | 220 | |
| Issuance of Class A common stock and pre-funded warrants, net offering costs | — | | | | 347,884 | | | 3 | | | — | | | — | | | 50,332 | | | — | | | 50,335 | | | — | | | 50,335 | |
| Net loss attributable to noncontrolling interest | — | | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (69) | | | (69) | |
| Balances at March 31, 2024 | $ | — | | | | 2,432,857 | | | $ | 24 | | | 577,349 | | | $ | 6 | | | $ | 306,887 | | | $ | (286,121) | | | $ | 20,796 | | | $ | 5,191 | | | $ | 25,987 | |
| (*) Allocation of NCI net loss was a result from the reclassification to permanent equity on February 27, 2024 (See Note 7) | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the three months ended September 30, 2022 |
| | | | Class A Common Stock | | Class B Common Stock | | | | | | | | |
| Redeemable
Noncontrolling
Interest | | | Shares | | Amount | | Shares | | Amount | | Note Receivable for Common Stock | | Additional
Paid-in
Capital | | Accumulated Deficit | | Total Stockholders' Deficit |
| Balances at June 30, 2022 | $ | 15,916 | | | | 77,329,051 | | | $ | 773 | | | 23,093,860 | | | $ | 232 | | | $ | — | | | $ | 243,772 | | | $ | (253,303) | | | $ | (8,526) | |
| Net loss | (1,207) | | | | — | | | — | | | — | | | — | | | — | | | — | | | (4,261) | | | (4,261) | |
| Share-based compensation | — | | | | — | | | — | | | — | | | — | | | — | | | 338 | | | — | | | 338 | |
| Issuance of Class A common stock under CinRx purchase agreement, net of offering costs | — | | | | 4,154,549 | | | 42 | | | — | | | — | | | (4,000) | | | 9,336 | | | — | | | 5,378 | |
| Change in redemption value of noncontrolling interest | 9,498 | | | | — | | | — | | | — | | | — | | | — | | | — | | | (9,498) | | | (9,498) | |
| Balances at September 30, 2022 | $ | 24,207 | | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | (4,000) | | | $ | 253,446 | | | $ | (267,062) | | | $ | (16,569) | |
The accompanying notes are an integral part of the unaudited condensed consolidated financial statements.
vTv Therapeutics Inc.
Condensed Consolidated Statement of Changes in Redeemable Noncontrolling Interest and Stockholders’ Deficit - Unaudited
(in thousands, except number of shares)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the nine months ended September 30, 2023 |
| | | | Class A Common Stock | | Class B Common Stock | | | | | | |
| Redeemable
Noncontrolling
Interest | | | Shares | | Amount | | Shares | | Amount | | Additional
Paid-in
Capital | | Accumulated Deficit | | Total Stockholders' Deficit |
| Balances at December 31, 2022 | $ | 16,579 | | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | 253,737 | | | $ | (265,524) | | | $ | (10,740) | |
| Net loss | (4,753) | | | | — | | | — | | | — | | | — | | | — | | | (16,772) | | | (16,772) | |
| Share-based compensation | — | | | | — | | | — | | | — | | | — | | | 1,175 | | | — | | | 1,175 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| Change in redemption value of noncontrolling interest | (1,104) | | | | — | | | — | | | — | | | — | | | — | | | 1,104 | | | 1,104 | |
| Balances at September 30, 2023 | $ | 10,722 | | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | 254,912 | | | $ | (281,192) | | | $ | (25,233) | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the nine months ended September 30, 2022 |
| | | Class A Common Stock | | Class B Common Stock | | | | | | | | |
| Redeemable
Noncontrolling
Interest | | Shares | | Amount | | Shares | | Amount | | Note Receivable for Common Stock | | Additional
Paid-in
Capital | | Accumulated Deficit | | Total Stockholders' Deficit |
| Balances at December 31, 2021 | $ | 24,962 | | | 66,942,777 | | | $ | 669 | | | 23,093,860 | | | $ | 232 | | | $ | — | | | $ | 238,193 | | | $ | (248,834) | | | $ | (9,740) | |
| Net loss | (4,564) | | | — | | | — | | | — | | | — | | | — | | | — | | | (14,419) | | | (14,419) | |
| Share-based compensation | — | | | — | | | — | | | — | | | — | | | — | | | 981 | | | — | | | 981 | |
| Issuance of Class A common stock to collaboration partner, net of offering costs | — | | | 10,386,274 | | | 104 | | | — | | | — | | | — | | | 4,936 | | | — | | | 5,040 | |
| Issuance of Class A common stock under CinRx purchase agreement, net of offering costs | — | | | 4,154,549 | | | 42 | | | — | | | — | | | (4,000) | | | 9,336 | | | — | | | 5,378 | |
| Change in redemption value of noncontrolling interest | 3,809 | | | — | | | — | | | — | | | — | | | — | | | — | | | (3,809) | | | (3,809) | |
| Balances at September 30, 2022 | $ | 24,207 | | | 81,483,600 | | | $ | 815 | | | 23,093,860 | | | $ | 232 | | | $ | (4,000) | | | $ | 253,446 | | | $ | (267,062) | | | $ | (16,569) | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| For the three months ended March 31, 2023 |
| | | | Class A Common Stock | | Class B Common Stock | | | | | | |
| Redeemable
Noncontrolling
Interest | | | Shares(*) | | Amount(*) | | Shares(*) | | Amount(*) | | Additional
Paid-in
Capital(*) | | Accumulated Deficit | | Total Stockholders' Deficit |
| Balances at December 31, 2022 | $ | 16,579 | | | | 2,084,973 | | | $ | 21 | | | 577,349 | | | $ | 6 | | | $ | 254,757 | | | $ | (265,524) | | | $ | (10,740) | |
| Net loss | (1,275) | | | | — | | | — | | | — | | | — | | | — | | | (4,499) | | | (4,499) | |
| Share-based compensation | — | | | | — | | | — | | | — | | | — | | | 343 | | | — | | | 343 | |
| Change in redemption value of noncontrolling interest | 4,296 | | | | — | | | — | | | — | | | — | | | — | | | (4,296) | | | (4,296) | |
| Balances at March 31, 2023 | $ | 19,600 | | | | 2,084,973 | | | $ | 21 | | | 577,349 | | | $ | 6 | | | $ | 255,100 | | | $ | (274,319) | | | $ | (19,192) | |
| (*) Adjusted retroactively for reverse stock split | | | | | | | | | | | | | | | | |
The accompanying notes are an integral part of the unaudited condensed consolidated financial statements.
vTv Therapeutics Inc.
Condensed Consolidated Statements of Cash Flows - Unaudited
(in thousands)
| | Three Months Ended March 31, | | | Three Months Ended March 31, |
| 2024 | | | 2024 | | 2023 |
| Cash flows from operating activities: | |
| Net loss before noncontrolling interest | |
| Net loss before noncontrolling interest | |
| Net loss before noncontrolling interest | |
| Adjustments to reconcile net loss before noncontrolling interest to net cash used in operating activities: | |
| Depreciation expense | |
| Depreciation expense | |
| Depreciation expense | |
| Loss from promissory note early redemption | |
| Non-cash interest income | |
| Share-based compensation expense | |
| Change in fair value of investments | |
| Change in fair value of warrants, related party | |
| Changes in assets and liabilities: | |
| Accounts receivable | |
| Accounts receivable | |
| Accounts receivable | |
| Prepaid expenses and other current assets | |
| Other assets | |
| Accounts payable and accrued expenses | |
| | Other liabilities | |
| Other liabilities | |
| Other liabilities | |
| Net cash used in operating activities | |
| | | Nine Months Ended September 30, |
| | 2023 | | 2022 |
| Cash flows from operating activities: | | | |
| Net loss before noncontrolling interest | $ | (21,525) | | | $ | (18,983) | |
| Adjustments to reconcile net loss before noncontrolling interest to net cash used in operating activities: | |
| Depreciation expense | 67 | | | 69 | |
| Loss from G42 Promissory Note early redemption | 313 | | | — | |
| Non-cash interest income | (100) | | | (200) | |
| Interest expense | — | | | 9 | |
| Share-based compensation expense | 1,175 | | | 981 | |
| Change in fair value of investments | (3,044) | | | 2,998 | |
| Impairment of investments in Anteris Bio, Inc. | 4,245 | | | — | |
| Change in fair value of warrants, related party | (406) | | | (221) | |
| Changes in assets and liabilities: | |
| Accounts receivable | 173 | | | — | |
| Prepaid expenses and other assets | 665 | | | 853 | |
| Accounts payable and accrued expenses | 2,270 | | | (1,417) | |
| Contract liabilities | — | | | 6,769 | |
| Net cash used in operating activities | (16,167) | | | (9,142) | |
| Cash flows from investing activities: | |
| Purchases of property and equipment | — | | | (21) | |
| Net cash used in investing activities | — | | | (21) | |
| Cash flows from financing activities: | Cash flows from financing activities: | |
| Proceeds from G42 Promissory Note early redemption related to sale of Class A common stock to collaboration partner | 12,030 | | | — | |
| Proceeds from sale of Class A common stock to collaboration partner, net of offering costs | — | | | 5,040 | |
| Proceeds from sale of Class A common stock and warrants, net of offering costs | — | | | 5,746 | |
| | Proceeds from debt issuance | 566 | | | 776 | |
| Cash flows from financing activities: | |
| | Cash flows from financing activities: | |
| Proceeds from sale of Class A common stock and pre-funded warrants, net of offering costs | |
| Proceeds from sale of Class A common stock and pre-funded warrants, net of offering costs | |
| Proceeds from sale of Class A common stock and pre-funded warrants, net of offering costs | |
| Proceeds from promissory note early redemption related to sale of Class A common stock to collaboration partner | |
| | Repayment of notes payable | |
| Repayment of notes payable | |
| Repayment of notes payable | Repayment of notes payable | (317) | | | (475) | |
| Net cash provided by financing activities | Net cash provided by financing activities | 12,279 | | | 11,087 | |
| Net (decrease) increase in cash and cash equivalents | (3,888) | | | 1,924 | |
| Net increase in cash and cash equivalents | |
| Total cash and cash equivalents, beginning of period | Total cash and cash equivalents, beginning of period | 12,126 | | | 13,415 | |
| Total cash and cash equivalents, end of period | Total cash and cash equivalents, end of period | $ | 8,238 | | | $ | 15,339 | |
| | Non-cash activities: | Non-cash activities: | |
| Non-cash activities: | |
| Non-cash activities: | |
| Change in redemption value of noncontrolling interest | Change in redemption value of noncontrolling interest | $ | (1,104) | | | $ | 3,809 | |
| Notes receivable recorded at fair value from collaboration partner | — | | | 11,891 | |
| Notes receivable for common stock from CinRx purchase agreement | $ | — | | | $ | 4,000 | |
| Change in redemption value of noncontrolling interest | |
| Change in redemption value of noncontrolling interest | |
| Reclassification of noncontrolling interest to additional paid-in capital | |
|
The accompanying notes are an integral part of the unaudited condensed consolidated financial statements.
vTv Therapeutics Inc.
Notes to Condensed Consolidated Financial Statements – Unaudited
(dollar amounts are in thousands, unless otherwise noted)
Note 1: Description of Business and Basis of Presentation and Going Concern
Description of Business
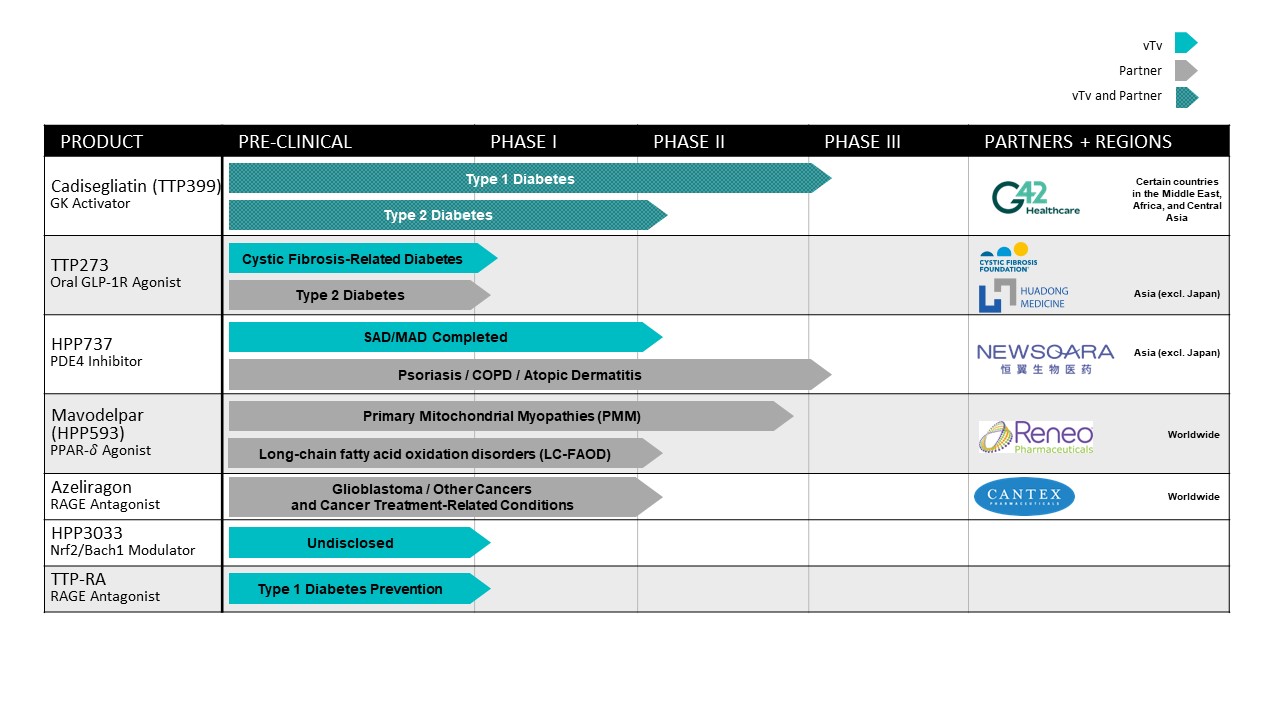
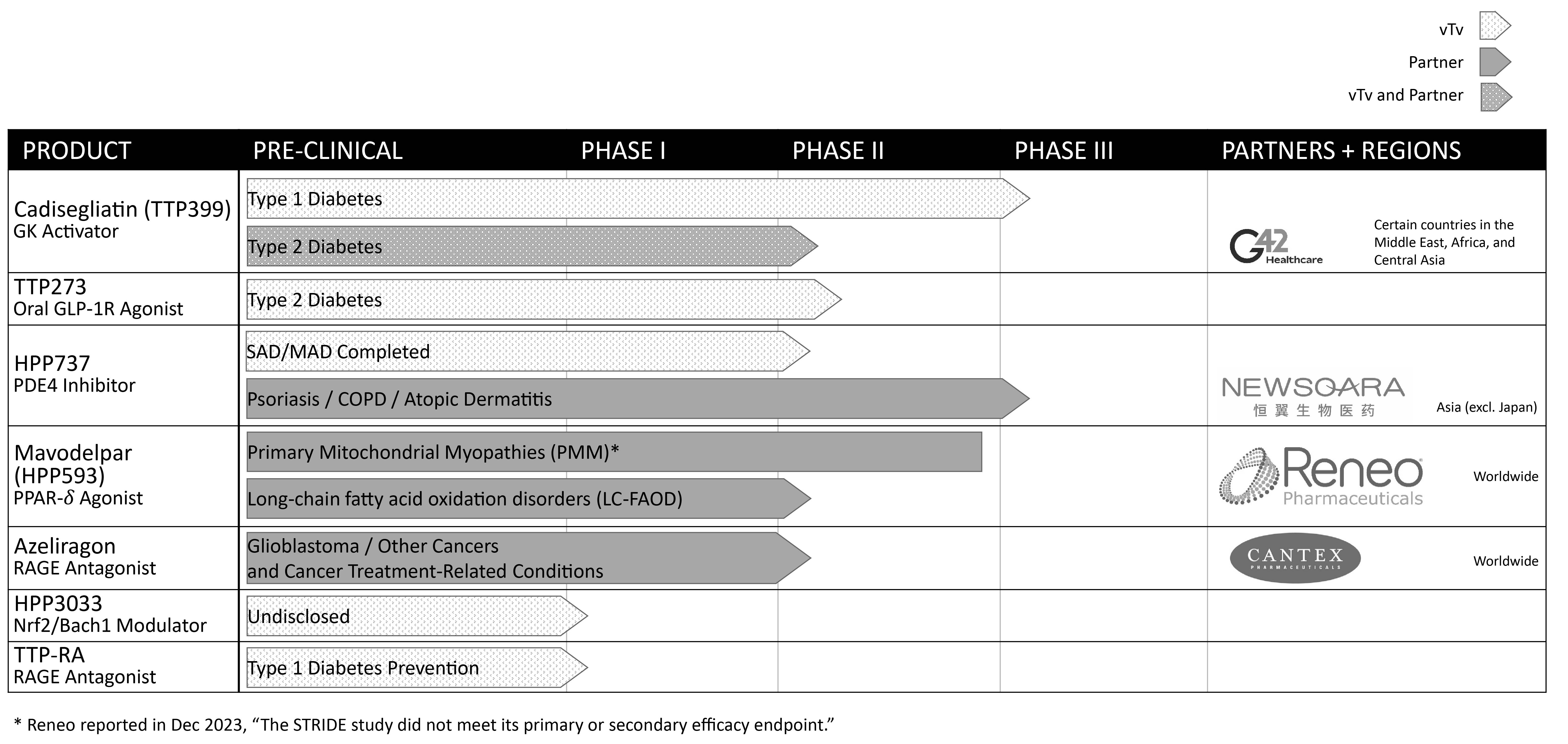
vTv Therapeutics Inc. (the “Company,” the “Registrant,” “we” or “us”) was incorporated in the state of Delaware in April 2015. The Company is a clinical stage pharmaceutical company focused on treating metabolic and inflammatory diseases to minimize their long-term complications and improve the lives of patients.through end-organ protection.
Principles of Consolidation
vTv Therapeutics Inc. is a holding company, and its principal asset is a controlling equity interest in vTv Therapeutics LLC (“vTv LLC”), the Company’s principal operating subsidiary, which is a clinical stage pharmaceutical company engaged in the discovery and development of orally administered small molecule drug candidates to fill significant unmet medical needs.
The Company has determined that vTv LLC is a variable-interest entity (“VIE”) for accounting purposes and that vTv Therapeutics Inc. is the primary beneficiary of vTv LLC because (through its managing member interest in vTv LLC and the fact that the senior management of vTv Therapeutics Inc. is also the senior management of vTv LLC) it has the power and benefits to direct all of the activities of vTv LLC, which include those that most significantly impact vTv LLC’s economic performance. vTv Therapeutics Inc. has therefore consolidated vTv LLC’s results pursuant to Accounting Standards Codification Topic 810, “Consolidation” in its Condensed Consolidated Financial Statements. The assets and liabilities of vTv LLC represent substantially all of the Company's consolidated assets and liabilities with the exception of the Warrants.
Various holders own nonvotingnon-voting interests in vTv LLC, representing a 22.1%19.2% economic interest in vTv LLC, effectively restricting vTv Therapeutics Inc.’s interest to 77.9%80.8% of vTv LLC’s economic results, subject to increase in the future, should vTv Therapeutics Inc. purchase additional nonvotingnon-voting common units (“vTv Units”) of vTv LLC, or should the holders of vTv Units decide to exchange such units (together with shares of the Company’s Class B common stock, par value $0.01 (“Class B common stock”)) for shares of Class A common stock (or cash) pursuant to the Exchange Agreement (as defined in Note 9)8). vTv Therapeutics Inc. has provided financial and other support to vTv LLC in the form of its purchase of vTv Units with the net proceeds of the Company’s initial public offering (“IPO”) in 2015, its registered direct offering in March 2019, and its agreeing to be a co-borrower under the Venture Loan and Security Agreement (the “Loan Agreement”) with Horizon Technology Finance Corporation and Silicon Valley Bank (together, the “Lenders”) which was entered into in 2016. vTv Therapeutics Inc. entered into the letter agreements with MacAndrews and Forbes Group LLC (“M&F Group”), a related party and an affiliate of MacAndrews & Forbes Incorporated (together with its affiliates “MacAndrews”) in December 2017, July 2018, December 2018, March 2019, September 2019, and December 2019 (each a “Letter Agreement” and collectively, the “Letter Agreements”). In addition, vTv Therapeutics Inc. also entered into the Controlled Equity OfferingSM Sales Agreement (the “Sales Agreement”) with Cantor Fitzgerald & Co. (“Cantor Fitzgerald”) (the “ATM Offering”), the purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln Park”) (the “LPC Purchase Agreement”), thea common stock purchase agreement with G42 Investments AI Holding RSC Ltd (“G42 Investments”) (the “G42 Purchase Agreement”) and, the common stock and warrant purchase agreement with CinPax, LLC and CinRx, LLC, respectively (the “CinRx Purchase Agreement”). In addition vTv Therapeutics Inc also entered into a Securities Purchase Agreement with Private Placement Investors and the sales agreement with Cowen and Company, LLC (“TD Cowen”) (“TD Cowen Sales Agreement”). vTv Therapeutics Inc. will not be required to provide financial or other support for vTv LLC. However, vTv Therapeutics Inc. will control its business and other activities through its managing member interest in vTv LLC, and its management is the management of vTv LLC. Nevertheless, because vTv Therapeutics Inc. will have no material assets other than its interests in vTv LLC, any financial difficulties at vTv LLC could result in vTv Therapeutics Inc. recognizing a loss.
Going Concern and Liquidity
To date, the Company has not generated any product revenue and has not achieved profitable operations. The continuing development of our drug candidates will require additional financing. From its inception through September 30, 2023,March 31, 2024, the Company has funded its operations primarily through a combination of private placements of common and preferred equity, research collaboration agreements, upfront and milestone payments for license agreements, debt and equity financings and the completion of its IPO in August 2015. As of September 30, 2023,March 31, 2024, the Company had an accumulated deficit of $281.2$286.1 million and has generated net losses in each year of its existence.
As of September 30, 2023,March 31, 2024, the Company’s liquidity sources included cash and cash equivalents of $8.2$52.3 million.
Based on our current operating plan, we believe that our current cash and cash equivalents will allow us to meet our liquidity requirements for at least the next twelve months.
On February 27, 2024, we entered into a securities purchase agreement (the “Securities Purchase Agreement”) with certain institutional accredited investors (the “Private Placement Investors”), pursuant to which we agreed to issue and sell to the first quarterPrivate Placement Investors in a private placement (the “Private Placement”) (i) an aggregate of 2024. To meet464,377 shares (the “Private Placement Shares”) of our future funding requirementsClass A common stock, at a purchase price of $11.81 per share, and (ii) pre-funded warrants (the “Private Placement Pre-Funded Warrants”) to purchase up to an aggregate of 3,853,997 shares of our Class A common stock (the “Private Placement Warrant Shares”) at a purchase price of $11.80 per Private Placement Pre-Funded Warrant (representing the $11.81 per Private Placement Share purchase price less the exercise price of $0.01 per Private Placement Warrant Share). We received aggregate gross proceeds from the Private Placement of approximately $51.0 million, before deducting offering expenses payable by us. The Private Placement Pre-Funded Warrants are exercisable at any time after their original issuance and will not expire.
On March 5, 2024, the Company entered into a letter agreement with the third quarter of 2024, including
funding the ongoing and future clinical trials of cadisegliatin (TTP399), we are evaluating several financing strategies, including direct equity investments and the potential licensing and monetization of other Company programs.
The Company may also use its remaining availability of $37.3 million under its Sales Agreement with Cantor FitzgeraldPrivate Placement Investors pursuant to which the Private Placement Investors agreed to exchange an aggregate of 116,493 Private Placement Shares for an aggregate of 116,590 Private Placement Pre-Funded Warrants.
On February 28, 2024, we entered into a sales agreement (the “TD Cowen Sales Agreement”) with Cowen and Company, LLC (“TD Cowen”), pursuant to which we may offer and sell, from time to time, through or to TD Cowen, as sales agent or principal, shares of the Company’sour Class A common stock, and the abilityhaving an aggregate offering price of up to $50.0 million (the “TD Cowen ATM Offering”). Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell an additional 9,437,376 shares of Class A common stock under the LPC Purchase Agreement basedsecurities registered on the remaining numberregistration statement relating to the TD Cowen ATM Offering with a value exceeding more than one-third of registered shares. However,our public float in any 12-month period so long as our public float remains below $75.0 million. Under the ability to use these sourcesterms of capital is dependent onthe TD Cowen Sales Agreement, we will pay TD Cowen a numbercommission of factors, including3.0% of the prevailing market priceaggregate proceeds from the sale of shares and the volume of trading in the Company’s Class A common stock. See Note 9 for further details.
If we are unable to raise additional capital as and when needed,reimburse certain legal fees or upon acceptable terms, such failure would have a significant negative impact on our financial condition. As such, these conditions raise substantial doubt about the Company’s ability to continue as a going concern.
The Company’s financial statements have been prepared assuming the Company will continue as a going concern, which contemplates, among other things, the realization of assets and satisfaction of liabilities in the normal course of business. The Condensed Consolidated Financial Statements do not include adjustments to reflect the possible future effects on the recoverability and classification of recorded assets or the amounts of liabilities that might be necessary should the Company be unable to continue as a going concern.disbursements.
Note 2: Summary of Significant Accounting Policies
Unaudited Interim Financial Information
The accompanying financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“GAAP”). The accompanying Condensed Consolidated Balance Sheet as of September 30, 2023,March 31, 2024, Condensed Consolidated Statements of Operations for the three and nine months ended September 30,March 31, 2024 and 2023, and 2022, Condensed Consolidated Statement of Changes in Redeemable Noncontrolling Interest and Stockholders’ DeficitEquity (Deficit) for the three and nine months ended September 30,March 31, 2024 and 2023 and 2022 and Condensed Consolidated Statements of Cash Flows for the ninethree months ended September 30,March 31, 2024 and 2023 and 2022 are unaudited. These unaudited financial statements have been prepared in accordance with the rules and regulations of the United States Securities and Exchange Commission (“SEC”) for interim financial information. Accordingly, they do not include all of the information and footnotes required by GAAP for complete financial statements. These financial statements should be read in conjunction with the audited financial statements and the accompanying notes for the year ended December 31, 2022,2023, contained in the Company’s Annual Report on Form 10-K. The unaudited interim financial statements have been prepared on the same basis as the annual financial statements and, in the opinion of management, reflect all adjustments (consisting of normal recurring adjustments) necessary to state fairly the Company’s financial position as of September 30, 2023,March 31, 2024, the results of operations for the three and nine months ended September 30,March 31, 2024 and 2023 and 2022 and cash flows for the ninethree months ended September 30, 2023March 31, 2024 and 2022.2023. The December 31, 20222023 Condensed Consolidated Balance Sheet included herein was derived from the audited financial statements but does not include all disclosures or notes required by GAAP for complete financial statements.
The financial data and other information disclosed in these notes to the financial statements related to the three and nine months ended September 30,March 31, 2024 and 2023 and 2022 are unaudited. Interim results are not necessarily indicative of results for an entire year.
The Company does not have any components of other comprehensive income recorded within its Condensed Consolidated Financial Statements, and, therefore, does not separately present a statement of comprehensive income in its Condensed Consolidated Financial Statements.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires the Company to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities as of the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
On an ongoing basis, the Company evaluates its estimates, including those related to the grant date fair value of equity awards, the fair value of warrants to purchase shares of its Class A common stock, the fair value of its Class B common stock, the useful lives of property and equipment and the fair value of the Company’s debt, among others. The Company bases its estimates on historical experience and on various other assumptions that it believes to be reasonable, the results of which form the basis for making judgments about the carrying value of assets and liabilities.
Concentration of Credit Risk
Financial instruments that potentially expose the Company to concentrations of credit risk consist principally of cash on deposit with onemultiple financial institution.institutions. The balance of the cash account frequently exceedexceeds insured limits. The associated risk of concentration for cash and cash equivalents is mitigated by transferring a majority of our cash to a AAA rated money market account with a creditworthy institution.
One hundred percent (100%)customer represented 100% of the revenue earned during the ninethree months ended September 30, 2022 was attributable from the satisfaction of a development milestone from the First Huadong Amendment (as defined herein).March 31, 2024. The Company did not have any revenue during the three months ended September 30, 2022. The Company did not have any revenue during the three and nine months ended September 30,March 31, 2023.
Cash and Cash Equivalents
The Company considers any highly liquid investments with an original maturity of three months or less to be cash and cash equivalents.
Investments
Investments in entities in which the Company has no control or significant influence, is not the primary beneficiary, and have a readily determinable fair value are classified as equity investments with readily determinable fair value. The investments are measured at fair value based on a quoted market price per unit in active markets multiplied by the number of units held without consideration of transaction costs (Level 1). Gains and losses are recorded in other income (expense), net on the Condensed Consolidated Statements of Operations.
Equity investments without readily determinable fair value include ownership rights that do not provide the Company with control or significant influence and these investments do not have readily determinable fair values. The Company has elected to measure its equity investments without readily determinable fair values at cost minus impairment, if any, plus or minus changes resulting from observable price changes in orderly transactions for the identical or similar investment.
Revenue Recognition
The Company uses the revenue recognition guidance established by ASC 606, Revenue From Contracts With Customers (“ASC 606”). When an agreement falls under the scope of other standards, such as ASC 808, Collaborative Arrangements (“ASC 808”), the Company will apply the recognition, measurement, presentation, and disclosure guidance in ASC 606 to the performance obligations in the agreements if those performance obligations are with a customer. Revenue recognized by analogizing to ASC 606, is recorded as collaboration revenue on the statements of operations.
The majority of the Company’s revenue results from its license and collaboration agreements associated with the development of investigational drug products. The Company accounts for a contract when it has approval and commitment from both parties, the rights of the parties are identified, payment terms are identified, the contract has commercial substance and collectability of consideration is probable. For each contract meeting these criteria, the Company identifies the performance obligations included within the contract. A performance obligation is a promise in a contract to transfer a distinct good or service to the customer. The Company then recognizes revenue under each contract as the related performance obligations are satisfied. The Company will recognize collaboration revenue under ASC 808 as a stand-ready obligation under ASC 606 over time based on the estimated period of performance.
The transaction price under the contract is determined based on the value of the consideration expected to be received in exchange for the transferred assets or services. Development, regulatory and sales milestones included in the Company’s collaboration agreements are considered to be variable consideration. The amount of variable consideration expected to be received is included in the transaction price when it becomes probable that the milestone will be met. For contracts with multiple performance obligations, the contract’s transaction price is allocated to each performance obligation using the Company’s best estimate of the standalone selling price of each distinct good or service in the contract. The primary method used to estimate standalone selling price is the expected cost plus margin approach. Revenue is recognized over the related period over which the Company expects the services to be provided using a proportional performance model or a straight-line method of recognition if there is no discernible pattern over which the services will be provided.
Research and Development
Major components of research and development costs include cash compensation, depreciation expense on research and development property and equipment, costs of preclinical studies, clinical trials and related clinical manufacturing, costs
of drug development, costs of materials and supplies, facilities cost, overhead costs, regulatory and compliance costs, and fees paid to consultants and other entities that conduct certain research and development activities on the Company’s behalf. Research and development costs are expensed as incurred.
The Company records accruals based on estimates of the services received, efforts expended and amounts owed pursuant to contracts with numerous contract research and manufacturing organizations. In the normal course of business, the Company contracts with third parties to perform various clinical study activities in the ongoing development of potential products. The financial terms of these agreements are subject to negotiation and variation from contract to contract and may result in uneven payment flows. Payments under the contracts depend on factors such as the achievement of certain events and the completion of portions of the clinical study or similar conditions. The objective of the Company’s accrual policy is to match the recording of expenses in its financial statements to the actual services received and efforts expended. As such, expense accruals related to clinical studies are recognized based on the Company’s estimate of the degree of completion of the event or events specified in the specific clinical study.
The Company records nonrefundable advance payments it makes for future research and development activities as prepaid expenses. Prepaid expenses are recognized as expensesexpense in the Condensed Consolidated Statements of Operations as the Company receives the related goods or services.
Research and development costs that are reimbursed under a cost-sharing arrangement are reflected as a reduction of research and development expense.
Recently Issued Accounting Pronouncements Not Yet Adopted
Fair Value Measurements:Segment Reporting: In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): “Improvements to Reportable Segment Disclosures” In June 2022,(ASU 2023-07). The ASU expands public entities' segment disclosures by requiring disclosures of significant segment expenses that are regularly provided to the Financial Accounting Standards Board (FASB) issued Accounting Standards Update (ASU) No. 2022-03 Fair Value Measurements (Topic 820): Fair Value MeasurementCODM and included within each reported measure of Equity Securities Subject to Contractual Sale Restrictions. These amendments clarify thatsegment profit or loss, an amount and description of its composition for other segment items, and interim disclosures of a contractual restrictionreportable segment's profit or loss and assets. Early adoption is permitted. Since the Company only has one segment, the Company does not expect an impact when adopting this ASU on the saleCompany's Consolidated Financial Statements and disclosures.
Income Taxes: In December 2023, the FASB issued ASU 2023-09: “Improvements to Income Tax Disclosures” (“ASU 2023-09”). The ASU is intended to enhance the transparency and decision usefulness of an equity security is not considered part ofincome tax disclosures. The amendments in the unit of account ofASU address investor requests for enhanced income tax information primarily through changes to the equity securityrate reconciliation and therefore, is not considered in measuring fair value. This guidance isincome taxes paid information. ASU 2023-09 will be effective for public business entities for fiscal years, including interim periods within those fiscal years,us in the annual period beginning after December 15, 2023. EarlyJanuary 1, 2025, though early adoption is permitted. The Company has assessedis currently evaluating the presentational effect that ASU 2022-03 and early adopted the guidance during the second quarter of 2022. The adoption did not2023-09 will have a material impact on the Company's Condensed Consolidated Financial Statements.Statements and disclosures, and we expect considerable changes to our income tax disclosures.
Note 3: Collaboration Agreements
G42 Purchase Agreement and Cogna Collaborative and License Agreement
The Company and G42 Investments AI Holding RSC Ltd, a private limited company (“G42 Investments”), entered into a Common Stock Purchase Agreement (the “G42 Purchase Agreement”) on May 31, 2022,, pursuant to which the Company sold to G42 Investments 10,386,274259,657 shares of the Company’s Class A common stock at a price per share of approximately $2.41,$96.40, for an aggregate purchase price of $25.0 million, which was paid (i) $12.5 million in cash at the closing and (ii) $12.5 million in the form of a promissory note of G42 Investments to be paid at May 31, 2023 (the “G42 Promissory Note”). As part of the G42 Purchase Agreement, G42 Investments nominated a director as appointee and the Company’s board of directors approved appointing the new director to the Company’s board. On February 28, 2023, the Company and G42 Investments amended the G42 Purchase Agreement and modified the G42 Promissory Note to accelerate the payment due under the note. Pursuant to the amendment, on February 28, 2023, the Company received $12.0 million, which reflected the original amount due under the G42 Promissory Note less a 3.75% discount, in full satisfaction of the note, resulting in a loss of $0.3 million and was recognized as a component of other income/(expense)income, net in the Company’s Condensed Consolidated Statements of Operations.
G42 Investments has agreed to certain transfer restrictions (including restrictions on short sales or similar transactions) and restrictions on further acquisitions of shares, in each case subject to specified exceptions. Following the expiration of a lock up period, from the period May 31, 2022 until December 31, 2024 (or if earlier, the date of receipt of U.S. Food and Drug Administration (“FDA”) approval in the U.S. for cadisegliatin (TTP399), the Company has granted to G42 Investments
certain shelf and piggyback registration rights with respect to those shares of Class A common stock issued to G42 Investments pursuant to the G42 Purchase Agreement, including the ability to conduct an underwritten offering to resell such shares under certain circumstances. The registration rights include customary cooperation, cut-back, expense reimbursement, and indemnification provisions.
Contemporaneously with the G42 Purchase Agreement, effective on May 31, 2022, the Company entered into a collaboration and license agreement (the “Cogna Agreement”) with Cogna Technology Solutions LLC, an affiliate of G42 Investments (“Cogna”), which requires Cogna to work with the Company in performing clinical trials for cadisegliatin
(TTP399)TTP399) as well as jointly creating a global development plan to develop, market, and commercialize cadisegliatin (TTP399) in certain countries in the Middle East, Africa, and Central Asia (the “Partner Territory”). Under the terms of the Cogna Agreement, Cogna will obtain a license under certain intellectual property controlled by the Company to enable it to fulfill its obligations and exercise its rights under the Cogna agreement, including to develop and commercializcommercialize e cadisegliatin (TTP399) in the Partner Territory, but will not have access to the various intellectual property (“IP”) related to the license and cadisegliatin (TTP399). Specifically, the Company will share various protocols with Cogna related to conducting the clinical trials and will provide the patient dosages and placebo of cadisegliatin (TTP399) needed to conduct the trials.
Under the Cogna Agreement, Cogna has the right to develop and commercialize the cadisegliatin (TTP399) in the Partner Territory at its own cost once restrictions on the use of the IP have been lifted by the Company. The Cogna Agreement determined which specific countries in the Partner Territory that Cogna may pursue development and commercialization and provides the Company with the ability to determine when Cogna can benefit from this IP through the powers granted to the Company to approve the global development plan. Further, the Company may supply at cost, or Cogna may manufacture, cadisegliatin (TTP399) for commercial sale under terms to be agreed upon by the parties at a later date.
Separately, the Company will conduct its clinical trials for cadisegliatin (TTP399) outside of the Partner Territory at its own cost. The results of each party’s clinical trials will be combined by the Company to seek FDA approval in the United States for cadisegliatin (TTP399). On December 21, 2022, G42 Healthcare Technology Solutions LLC (formerly known as Cogna Technology Solutions LLC) novated its rights and obligation under the Cogna agreement to G42 Healthcare Research Technology Projects LLC (“G42 Healthcare”), an affiliate of G42 Investments. As a result of the novation, all reference to Cogna herein shall be deemed to refer to G42 Healthcare.
The G42 Purchase Agreement also provides for, following the receipt of FDA approval ofthe cadisegliatin (TTP399),FDA Approval, at the option of G42 Investments, either (a) the issuance of the Company’s Class A common stock (the “Milestone Shares”) having an aggregate value equal to $30.0 million or (b) the payment by the Company of $30.0 million in cash (the “Milestone Cash Payment”). The issuance of the Milestone Shares or the payment of the Milestone Cash Payment, as applicable, is conditioned upon receipt of the cadisegliatin FDA approvalApproval and subject to certain limitations and conditions set forth in the G42 Purchase Agreement. There can be no assurance that the cadisegliatinFDA approvalApproval will be granted or as to the timing thereof.
Once commercialization takes place in the Partner Territory, the Company will receive royalties in the single digits from Cogna on the net sales of cadisegliatin (TTP399) for a period of at least ten years after the first commercial sale of cadisegliatin (TTP399) in the Partner Territory.
Common stock is generally recorded at fair value at the date of issuance. In determining the fair value of the Class A common stock issued to G42 Investments, the Company considered the closing price of the common stock on the effective date. The Company did not make an adjustment to the fair value for sale restrictions on the stock in accordance with guidance recently adopted in ASU 2022-03. See the “Recently Issued Accounting Guidance” in this quarterly report on Form 10-Q for details of the ASU. Accordingly, the Company determined that cash consideration of $5.7 million should be recorded as fair value of the Class A common stock at the effective date, utilizing the Class A common stock closing price of $0.55$22.04 at the effective date.
A premium was paid on the Class A common stock by G42 Investments of $18.7 million, net of a note receivable discount of $0.6 million. This premium is determined to be the transaction price for all remaining obligations under the agreements, which will be accounted for under ASC 808 or ASC 606 based on determination of the unit of account.
The Company determined that certain commitments under the agreements are in the scope of ASC 808 as both the Company and Cogna are active participants in the clinical trials of cadisegliatin (TTP399), and both are exposed to significant risks and rewards based on the success of the clinical trials and subsequent FDA approval. Cogna is determined to be a vendor of the Company during the clinical trial phase, working on the Company’s behalf to complete R&Dresearch and development activities, and not in a customer capacity. The Company accounted for the commitments related to the clinical trials, which includes transfer of trial protocols, supply of clinical trial dosages, and collaboration on the joint development committee (“JDC”) as an ASC 808 unit of account, applying the recognition and measurement principles of ASC 606 by analogy. The Company
will recognize collaboration revenue for its development activities under ASC 808 over time based on the estimated period of performance.
By applying the principals in ASC 606 by analogy, the Company identified the performance obligation and considered the timing of satisfaction of the obligation to account for the pattern of revenue recognition. In order to recognize collaboration revenue, generally, the Company would begin satisfying its performance obligation and Cogna would need to be able to use and benefit from delivery of the assets or services. The performance obligation under the agreements that fall within the ASC 808 unit of account are concentrated in the clinical trials. As of September 30, 2023,March 31, 2024, the clinical trials had not
commenced. Accordingly, no collaboration revenue was recognized for the ASC 808 unit of account during the three and nine months ended September 30, 2023.March 31, 2024.
The Company identified certain commitments that are in the scope of ASC 606 as Cogna’s relationship is that of a customer for these commitments. The significant performance obligations that are in the scope of ASC 606 are (1) the development, commercialization and manufacturing license of the IP once restrictions on the use of the IP have been lifted by the Company and (2) a potential material right to a commercial supply agreement. The Company will recognize revenue from the development, commercial and manufacturing license at a point in time when the Company releases the restrictions on the use of the IP, which is expected to be after cadisegliatin (TTP399) is approved by the FDA. The Company will recognize revenue from the material right related to Cogna’s ability to purchase the commercial supply at cost as Cogna purchases the commercial supply from the Company, which will occur after the completion of the initial clinical trials (if Cogna decides to purchase the clinical supply from the Company). As a result, the Company has not recognized any revenue under the ASC 606 unit of account during the three and nine months ended September 30, 2023.March 31, 2024.
On February 28, 2023, the Company and G42 Investments amended the G42 Purchase Agreement and modified the G42 Promissory Note to accelerate the payment due under the note. Pursuant to the amendment, on February 28, 2023, the Company received $12.0 million, which reflected the original amount due under the G42 Promissory Note less a 3.75% discount, in full satisfaction of the note, resulting in a loss of $0.3 million and was recognized as a component of other income/(expense)income,net in the Company’s Condensed Consolidated Statements of Operations. The G42 Promissory Note receivable was classified and accounted for under ASC 310 “ReceivablesReceivables“ (“ASC 310”) and was initially measured at its fair value of $11.9 million. The Company also recorded the $18.7 million as deferred revenue in the Condensed Consolidated Balance Sheets, as none of the underlying performance obligations had been satisfied as of and for the three and nine months ended September 30, 2023.March 31, 2024.
HuadongNewsoara License Agreement
The Company is party to a license agreement with Hangzhou Zhongmei Huadong PharmaceuticalNewsoara Biopharma Co., Ltd., (“Huadong”Newsoara”) (the “Huadong“Newsoara License Agreement”), under which HuadongNewsoara obtained an exclusive and sublicensable license to develop and commercialize the Company’s glucagon-like peptide-1 receptor agonistphosphodiesterase type 4 inhibitors (“GLP-1r”PDE4”) program, including the compound TTP273HPP737, for therapeutic uses in humans or animals, in China, Hong Kong, Macau, Taiwan and certain other Pacific Rimpacific rim countries including Australia and South Korea (collectively, the “Huadong“Newsoara License Territory”). Additionally, under the HuadongNewsoara License Agreement, the Company obtained a nonexclusive,non-exclusive, sublicensable, royalty-free license to develop and commercialize certain HuadongNewsoara patent rights and know-how related to the Company’s GLP-1rPDE4 program for therapeutic uses in humans or animals outside of the HuadongNewsoara License Territory.
On January 14, 2021, theThe Company entered into the first amendment to the Huadong License Agreement ( the “First Huadong Amendment”) which eliminated the Company’s obligation to sponsor a multi-region clinical trial (the “Phase 2 MRCT”), and corresponding obligation to contribute up to $3.0 million in support of such trial. The amendment also reduced the total potential development and regulatory milestone payments by $3.0 million.
Prior to the First Amendment, the Company hadhas fully allocated a portion of the transaction price to the obligation to sponsor and conduct a portion of the Phase 2 MRCT. Upon the removal of this performance obligation, the Company evaluated the impact of the modification under the provisions of ASC 606 and performed a reallocation of the transaction price among the remaining performance obligations. The majority of the transaction price originally allocated to the Phase 2 MRCT performance obligation was reallocated to the license and technology transfer services combined performance obligation discussed below, which had already been completed. The reallocation of the purchase price in connection with the First Huadong Amendment was made based on the relative estimated selling prices of the remaining performance obligations.
The significant performance obligations under the Huadong License Agreement, as amended, were determined to be (i) the exclusive license to develop and commercialize the Company’s GLP-1r program, (ii) technology transfer services related to the chemistry and manufacturing know-how for a defined period after the effective date, (iii) the Company’s obligation to participate on a joint development committee (the “JDC”), and (iv) other obligations considered to be immaterial in nature.
The Company has determined that the license and technology transfer services related to the chemistry and manufacturing know-how represent a combined performance obligation because they were not capable of being distinct on their own. The Company also determined that there was no discernible pattern in which the technology transfer services would be provided during the transfer service period. As such, the Company recognized the revenue related to this combined performance obligation using the straight-line method over the transfer service period. In the first quarter of 2022, the transaction price for this performance obligation was increased by $2.0 million due to the satisfaction of a development milestone under the license agreement. This amount was fully recognized as revenue during the nine months ended
September 30, 2022 as the related performance obligation was fully satisfied. No revenue related to this combined performance obligation was recognized during the nine months ended September 30, 2023.
A portion of the transaction price allocated to the obligation to participate in the JDC to oversee the development of products and the Phase 2 MRCT in accordance with the development plan remained deferred as of September 30, 2023, and revenue will be recognized using the proportional performance model over the period of the Company’s participation on the JDC. The unrecognized amount of the transaction price allocated to this performance obligation as of September 30, 2023 was immaterial. An immaterial amount of revenue for this performance obligation was recognized during the nine months ended September 30, 2022. No revenue for this performance obligation has been recognized during the nine months ended September 30, 2023.
Anteris License Agreement
On December 11, 2020, the Company entered into a license agreement with Anteris Bio, Inc. (“Anteris”) (the “Anteris License Agreement”), under which Anteris obtained a worldwide, exclusive and sublicensable license to develop and commercialize the Company’s Nrf2 activator, HPP971.
Under the terms of the Anteris License Agreement, Anteris paid the Company an initial license fee of $2.0 million. The Company is eligible to receive additional potential development, regulatory, and sales-based milestone payments totaling up to $151.0 million. Anteris is also obligated to pay vTv royalty payments at a double-digit rate based on annual net sales of licensed products. Such royalties will be payable on a licensed product-by-licensed product basis until the latest of expiration of the licensed patents covering a licensed product in a country, expiration of data exclusivity rights for a licensed product in a country, or a specified number of years after the first commercial sale of a licensed product in a country. As additional consideration, the Company received preferred stock representing a minority ownership interest in Anteris.
Pursuant to the terms of the Anteris License Agreement, the Company was required to provide technology transfer services for a 30 day period after the effective date. In accordance with ASC 606, the Company identified all of the performance obligations at the inception of the Anteris License Agreement. The significant obligations were determined to be the license and the technology transfer services. The Company has determined that the license and technology transfer services representwhich represents a single performance obligation because they were not capable of being distinct on their own. The transaction priceCompany recognized revenue for this performance obligation using the straight-line method over the transfer service period. The revenue for this performance obligation has been fully allocated torecognized as of March 31, 2024. In the first quarter of 2024, the transaction price for this combined performance obligation and consistedwas increased by $1.0 million due to the satisfaction of a development milestone under the $2.0 million initial license paymentNewsoara License Agreement. This amount was fully recognized as wellrevenue during the three months ended March 31, 2024, as the fair value of the equity interest received in Anteris of $4.2 million. Therelated performance obligation was fully satisfied. No revenue related to this performance obligation was fully recognized during the year ended December 31, 2020, as the technology transfer servicesand there were considered complete as of that date. No revenue was recognized relatedno changes to the Anteris License Agreementtransaction price during the three and nine months ended September 30, 2023 and 2022.
On September 25, 2023, Anteris notified the Company that it was terminating the Anteris License Agreement. Under the terms of the Anteris License Agreement, the termination would become effective ninety days following the receipt of notice, or December 24,March 31, 2023. The Company has begun the process of winding up the license, including obtaining all regulatory, manufacturing and other records generated by Anteris during the term of the Anteris License Agreement.
On October 16, 2023, the Company received a Section228(e) notice from Anteris. that the stockholders voted in favor of the dissolution of the Company and the adoption of a plan of Liquidation and Dissolution (“Plan of Dissolution”). (See Note 14).
Contract Liabilities
Contract liabilities related to the Company’s collaboration agreements consisted of the following (in thousands):
| | | | | | | | | | | |
| September 30, 2023 | | December 31, 2022 |
| Current portion of contract liabilities | $ | 17 | | | $ | 17 | |
| Contract liabilities, net of current portion | 18,669 | | | 18,669 | |
| Total contract liabilities | $ | 18,686 | | | $ | 18,686 | |
Note 4: Share-Based Compensation
The Company has issued nonqualifiednon-qualified stock option awards to management, other key employees, consultants, and nonemployeenon-employee directors. These option awards generally vest ratably over a three-year period and the option awards expire after a term of ten years from the date of grant. As of September 30, 2023,March 31, 2024, the Company had total unrecognized stock-based compensation expense for its outstanding stock option awards of approximately $1.8$1.3 million, which is expected to be
recognized over a weighted average period of 2.22.1 years. The weighted average grant date fair value of options granted during the ninethree months ended September 30,March 31, 2023 and 2022 was $0.76 and $0.68$0.80 per option, respectively.option. There were no options granted during the three months ended March 31, 2024. The aggregate intrinsic value of the in-the-money awards outstanding at September 30, 2023March 31, 2024 was immaterial.de minimis.
On February 23, 2024, in connection with the Private Placement, several directors resigned as members of the Company’s Board of Directors, effective on the closing of the Private Placement. As a result of their resignations, 14,340 stock options to purchase shares of common stock were modified to increase the time period to exercise the options and 7,590 stock options to purchase shares of common stock were modified to accelerate vesting at the termination date. All the unvested options were modified to be fully vested as of the posting of the Private Placement which resulted in a reduction in their fair value. The Company incurred $0.1 million reduction in stock compensation expense for the modifications for the three months ended March 31, 2024.
The following table summarizes the activity related to the stock option awards for the ninethree months ended September 30, 2023:March 31, 2024:
| | | | | | | | | | | |
| Number of Shares | | Weighted
Average Exercise Price |
| Awards outstanding at December 31, 2022 | 8,418,571 | | | $ | 2.17 | |
| Granted | 1,176,868 | | | 0.87 | |
| Forfeited | (50,000) | | | 1.29 | |
| Awards outstanding at September 30, 2023 | 9,545,439 | | | $ | 2.01 | |
| Options exercisable at September 30, 2023 | 5,018,072 | | | $ | 3.04 | |
| Weighted average remaining contractual term | 6.6 Years | | |
| Options vested and expected to vest at September 30, 2023 | 8,188,444 | | | $ | 2.21 | |
| Weighted average remaining contractual term | 7.5 Years | | |
| | | | | | | | | | | |
| Number of Shares | | Weighted
Average Exercise Price |
| Awards outstanding at December 31, 2023 | 249,247 | | | $ | 77.53 | |
| | | |
| Forfeited | (625) | | | 28.80 | |
| Awards outstanding at March 31, 2024 | 248,622 | | | $ | 77.65 | |
| Options exercisable at March 31, 2024 | 163,336 | | | $ | 102.02 | |
| Weighted average remaining contractual term | 6.4 Years | | |
| Options vested and expected to vest at March 31, 2024 | 220,524 | | | $ | 83.88 | |
| Weighted average remaining contractual term | 7.0 Years | | |
Compensation expense related to the grants of stock options is included in research and development and general and administrative expense as follows (in thousands):
| | Three Months Ended September 30, | | Nine Months Ended September 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| Three Months Ended March 31, | |
| Three Months Ended March 31, | |
| Three Months Ended March 31, | |
| 2024 | |
| 2024 | |
| 2024 | |
| Research and development | |
| Research and development | |
| Research and development | Research and development | $ | 113 | | | $ | 109 | | | $ | 316 | | | $ | 289 | |
| General and administrative | General and administrative | 320 | | | 229 | | | 859 | | | 692 | |
| General and administrative | |
| General and administrative | |
| Total share-based compensation expense | Total share-based compensation expense | $ | 433 | | | $ | 338 | | | $ | 1,175 | | | $ | 981 | |
| Total share-based compensation expense | |
| Total share-based compensation expense | |
Note 5: Investments
In connection with the Reneo Pharmaceuticals, Inc. (“Reneo”) and Anteris License Agreements, the Company received equity ownership interests of less than 20% of the voting equity of the investees. Further, the Company does not have the ability to exercise significant influence over the investees. The investments are classified as long-term investments in the Company’s Condensed Consolidated Balance Sheets.
Reneo completed its initial public offering in April 2021. Prior to Reneo becoming a publicly traded company, the Company’s investment in Reneo did not have a readily determinable fair value and was measured at cost less impairment, adjusted for any changes in observable prices, under the measurement alternative. Subsequent to Reneo’s initial public offering, the Company’s investment in Reneo is considered to have a readily determinable fair value and, as such, is adjusted to its fair value each period with changes in fair value recognized as a component of net loss.
The Company’s investment in Anteris does not have a readily determinable fair value and is measured at cost less impairment, adjusted for any changes in observable prices.
The Company’s investments consist of the following:
| | | | | | | | | | | |
| September 30, 2023 | | December 31, 2022 |
| Equity investment with readily determinable fair value: | | | |
| Reneo common stock | $ | 4,387 | | | $ | 1,343 | |
| | | |
| Equity investment without readily determinable fair values assessed under the measurement alternative: | | | |
| Anteris preferred stock | — | | | 4,245 | |
| Total | $ | 4,387 | | | $ | 5,588 | |
The Company recognized an unrealized gain on its investment in Reneo of $0.6 million and $3.0 million for the three and nine months ended September 30, 2023, respectively. The Company recognized an unrealized gain on its investment in Reneo of $0.4 million and an unrealized loss of $3.0 million for the three and nine months ended September 30, 2022,
respectively. These adjustments were recognized as a component of other income/(expense) in the Company’s Condensed Consolidated Statements of Operations.
On October 16, 2023, the Company received a Section228(e) notice from Anteris that the stockholders voted in favor of the dissolution of Anteris and the adoption of a plan of Liquidation and Dissolution (“Plan of Dissolution”). The Company determined the carrying value of this investment was not expected to be recoverable due to the Plan of Dissolution. As a result, the Company recorded a $4.2 million impairment charge during the three and nine months ended September 30, 2023.
On October 30, 2023, the Company entered into a Common Stock Repurchase Agreement (the “Repurchase Agreement”) with Reneo to purchase all of its common stock held by vTv LLC for total proceeds to vTv LLC of approximately $4.4 million.
Note 6:5: Commitments and Contingencies
Legal Matters
From time to time, the Company is involved in various legal proceedings arising in the normal course of business. If a specific contingent liability is determined to be probable and can be reasonably estimated, the Company accrues and discloses the amount. The Company is not currently a party to any material legal proceedings.
Novo Nordisk
In February 2007, the Company entered into an Agreement (the “Novo License Agreement”) Concerning Glucokinase Activator Project with Novo Nordisk A/S (the “Novo Nordisk”) whereby the Company obtained an exclusive, worldwide, sublicensable license under certain Novo Nordisk intellectual property rights to discover, develop, manufacture, have manufactured, use and commercialize products for the prevention, treatment, control, mitigation or palliation of human or animal diseases or conditions. As part of this license grant, the Company obtained certain worldwide rights to Novo Nordisk’s GKA program, including rights to preclinical and clinical compounds such as cadisegliatin (TTP399)TTP399. This agreement was amended in May 2019 to create milestone payments applicable to certain specific and nonspecificnon-specific areas of therapeutic use. Under the terms of the amended Novo License Agreement, the Company has potential developmental and regulatory milestone payments payable totaling up to $9.0 million for approval of a product for the treatment of type 1 diabetes, $50.5 million for approval of a product for the treatment of type 2 diabetes, or $115.0 million for approval of a product in any other indication. The Company may also be obligated to pay an additional $75.0 million in potential sales-based milestones, as well as royalty payments, at mid-single digit royalty rates, based on tiered sales of commercialized licensed products. During the fourth quarter of 2021, the Company made a payment of $2.0 million related to the satisfaction of the milestone to complete the phase 2 trials for cadisegliatin (TTP399) under this agreement.
Note 7:6: Leases
In August 2019, the Company leased office space for its headquarters location under an operating lease. This lease commenced in November 2019 after the completion of certain tenant improvements made by the lessor. The lease includes an
option to renew for a five-year term as well as an option to terminate after three years, neither of which have been recognized as part of its related right of use assets or lease liabilities as their election is not considered reasonably certain. In November 2022, the Company entered into a second amendment to the lease, (i) to reduce the square footage and (ii) to extend the lease term, which constituted a modification event under ASC 842 and, the lease classification for the asset remains as an operating lease. As a result of the remeasurement of the associated lease liabilities, the Company recognized additional right of use assets and corresponding lease liabilities of $0.1 million. Further, the second amendment to the lease does not include any material residual value guarantee or restrictive covenants.
At each of September 30, 2023March 31, 2024 and December 31, 2022,2023, the weighted average incremental borrowing rate for the operating leases held by the Company was 9.5%. At September 30, 2023March 31, 2024 and December 31, 2022,2023, the weighted average remaining lease terms for the operating leases held by the Company were 2.21.7 years and 2.91.9 years, respectively.
Maturities of lease liabilities for the Company’s operating leases as of September 30, 2023March 31, 2024 were as follows (in thousands):
| | 2023 (remaining three months) | $ | 48 | |
| 2024 | 194 | |
| 2024 (remaining nine months) | |
| 2025 | 2025 | 177 | |
| 2026 | 2026 | — | |
| 2027 | 2027 | — | |
| 2028 | |
| Thereafter | Thereafter | — | |
| Total lease payments | Total lease payments | 419 | |
| Less: imputed interest | Less: imputed interest | (41) | |
| Present value of lease liabilities | Present value of lease liabilities | $ | 378 | |
Operating lease cost and the related operating cash flows for the ninethree months ended September 30, 2023 and 2022March 31, 2024 was $0.2$0.1 million and $0.1 million, respectively.was immaterial for the three months ended March 31, 2023.
Note 8: Redeemable7: Noncontrolling Interest
The Company is subject to the Exchange Agreement with respect to the vTv Units representing the 22.1%19.2% noncontrolling interest in vTv LLC outstanding as of September 30, 2023March 31, 2024 (see Note 9). The Exchange Agreement requires the surrender of an equal number of vTv Units and Class B common stock for (i) shares of Class A common stock on a one-for-one basis or (ii) cash (based on the fair market value of the Class A common stock as determined pursuant to the Exchange Agreement), at the Company’s option (as the managing member of vTv LLC), subject to customary conversion rate adjustments for stock splits, stock dividends and reclassifications. The exchange value is determined based on a 20-day volume weighted average price of the Class A common stock as defined in the Exchange Agreement, subject to customary conversion rate adjustments for stock splits, stock dividends and reclassifications.
On February 27, 2024, in connection with the Private Placement financing, the Investor Rights Agreement altered M&F TTP Holdings Two LLC (“M&F”) governance rights such that directors designated by M&F no longer comprised a majority of the Company’s Board of Directors (see Note 9). The redeemable noncontrolling interest redemption feature to exchange vTv Units for cash rather than shares of Class A common stock is recognizeda contingent event that is now within control of the Company through the Company’s independent Board of Directors. As a result, $5.3 million representing the fair value of redeemable noncontrolling interest on February 27, 2024, was reclassified from temporary equity in the mezzanine section of the Condensed Consolidated Balance Sheets to noncontrolling interest as a component of permanent equity.
Prior to February 27, 2024, the Company recorded redeemable noncontrolling interest at the higher of (1) its initial fair value plus accumulated earnings/losses associated with the noncontrolling interest or (2) the redemption value as of the balance sheet date. At September 30, 2023 and December 31, 2022,2023, the redeemable noncontrolling interest was recorded based on the redemption value as of the balance sheet date of $10.7 million and $16.6 million, respectively.$6.1 million.
Changes in the Company’s ownership interest in vTv LLC while the Company retains its controlling interest in vTv LLC are accounted for as equity transactions, and the Company is required to adjust noncontrolling interest and equity for
such changes. The following is a summary of net income attributable to vTv Therapeutics Inc. and transfers to noncontrolling interest:
| | For the Three Months Ended September 30, | | For the Nine Months Ended September 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| For the Three Months Ended March 31, | |
| For the Three Months Ended March 31, | |
| For the Three Months Ended March 31, | |
| 2024 | |
| 2024 | |
| 2024 | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | Net loss attributable to vTv Therapeutics Inc. common shareholders | $ | (6,654) | | | $ | (4,261) | | | $ | (16,772) | | | $ | (14,419) | |
| Decrease/(Increase) in vTv Therapeutics Inc. accumulated deficit for purchase of vTv LLC Units as a result of common stock issuances | 1,592 | | | (1,174) | | | 4,212 | | | (103) | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | |
| Net loss attributable to vTv Therapeutics Inc. common shareholders | |
| Increase in vTv Therapeutics Inc. stockholders' equity (deficit) for sale of vTv Units as a result of common stock issuances | |
| Increase in vTv Therapeutics Inc. stockholders' equity (deficit) for sale of vTv Units as a result of common stock issuances | |
| Increase in vTv Therapeutics Inc. stockholders' equity (deficit) for sale of vTv Units as a result of common stock issuances | |
| Change from net loss attributable to vTv Therapeutics Inc. common shareholders and transfers to noncontrolling interest | Change from net loss attributable to vTv Therapeutics Inc. common shareholders and transfers to noncontrolling interest | $ | (5,062) | | | $ | (5,435) | | | $ | (12,560) | | | $ | (14,522) | |
| Change from net loss attributable to vTv Therapeutics Inc. common shareholders and transfers to noncontrolling interest | |
| Change from net loss attributable to vTv Therapeutics Inc. common shareholders and transfers to noncontrolling interest | |
Note 9:8: Stockholders’ DeficitEquity (Deficit)
Amendment to Certificate of Incorporation
On May 4, 2021, the Company filed an amendment to its Amended and Restated Certificate of Incorporation (the “Charter Amendment”) to increase the number of shares of Class A common stock that the Company is authorized to issue from 100,000,000 shares of Class A common stock to 200,000,000 shares of Class A common stock, representing an increase of 100,000,000 shares of authorized Class A common stock, with a corresponding increase in the total authorized common stock, which includes Class A common stock and Class B common stock, from 200,000,000 to 300,000,000, and a corresponding increase in the total authorized capital stock, which includes common stock and preferred stock, from 250,000,000 shares to 350,000,000 shares.
On November 20, 2023, the Company filed an amendment to its Amended and Restated Certificate of Incorporation as amended, to effect a reverse stock split at a ratio of 1-for-40 (the "Reverse Stock Split"). Pursuant to the Reverse Stock Split, every 40 shares of the Company’s Class A common stock was combined into one issued and outstanding share of Class A Common Stock and every 40 shares of the Company’s Class B common stock was combined into one issued and outstanding share of Class B Common Stock. The Reverse Stock Split did not reduce the number of authorized shares of Class A and Class B common stock, which remained at 200,000,000 and 100,000,000 respectively and did not change the par value of the common stock, which remained at $0.01 per share. The Reverse Stock Split did not have any effect on the number of authorized shares of the Company’s preferred stock, par value of $0.01 per share, which would remain at 50,000,000 shares. Currently no shares of preferred stock are outstanding.
Common Stock and Pre-funded Warrants
In February 2024, the Company entered into a Securities Purchase Agreement with certain Private Placement Investors, pursuant to which we agreed to issue and sell to the Private Placement Investors in a private placement (i) an aggregate of 464,377 shares of our Class A common stock, at a purchase price of $11.81 per share and (ii) issued pre-funded warrants to purchase an aggregate of 3,853,997 shares of the Company’s Class A common stock at a price of $11.80 per pre-funded warrant. The pre-funded warrants were immediately exercisable, have an exercise price of $0.01 and may be exercised at any time after the date of issuance. A holder of pre-funded warrants may not exercise the warrant if the holder, together with its affiliates, would beneficially own more than 9.99% of the number of shares of the Company’s common stock outstanding immediately after giving effect to such exercise. A holder of the pre-funded warrants may increase or decrease this percentage not in excess of 19.99% by providing at least 61 days’ prior notice to the Company. As of March 31, 2024, there were pre-funded warrants to purchase an aggregate of 3,970,587 shares of the Company’s common stock that remained available for exercise.
The pre-funded warrants were classified as a component of permanent equity in the Company's Condensed Consolidated Balance Sheet as they are freestanding financial instruments that are immediately exercisable, do not embody an obligation for the Company to repurchase its own shares and permit the holders to receive a fixed number of shares of common stock upon exercise. All of the shares underlying the pre-funded warrants have been included in the weighted-average number of shares of common stock used to calculate net loss per share attributable to common stockholders because the shares may be issued for little or no consideration, are fully vested and are exercisable after the original issuance date of the pre-funded warrants.
On March 5, 2024, the Company entered into a letter agreement with the Private Placement Investors pursuant to which the Private Placement Investors agreed to exchange an aggregate of 116,493 Private Placement Shares for an aggregate of 116,590 Private Placement Pre-Funded Warrants.
G42 Investments Transaction
On May 31, 2022, the Company and G42 Investments entered in to the G42 Purchase Agreement (see Note 3), pursuant to which the Company agreed to sell to G42 Investments 10,386,274259,657 shares of the Company's Class A common stock at a price per share of approximately $2.41,$96.40, for an aggregate purchase price of $25.0 million, consisting of (i) $12.5 million in cash at the closing of the transaction and (ii) $12.5 million in the form of a promissory note of G42 Investments to be paid at the one-year anniversary of the execution of the G42 Purchase Agreement (the “G42 Promissory Note”). On February 28, 2023, the Company and G42 Investments amended the G42 Purchase Agreement and modified the G42 Promissory Note to accelerate the payment due under the note. Pursuant to the amendment, on February 28, 2023, the Company received $12.0 million, which reflected the original amount due under the G42 Promissory Note less a 3.75% discount, in full satisfaction of the note, resulting in a loss of $0.3 million and was recognized as a component of other income/(expense)income, net in the Company’s Condensed Consolidated Statements of Operations.
CinPax and CinRx Transaction
On July 22, 2022 (the “Transaction Date”), the Company entered into the CinRx Purchase Agreement with CinPax, LLC (“CinPax”) and CinRx Pharma,CinRx-Pharma, LLC (“CinRx”), pursuant to which the Company agreed to sell to CinPax 4,154,549103,864 shares of the Company’s Class A common stock at a price per share of approximately $2.41,$96.40, for an aggregate purchase price of $10.0 million, which was paid (i) $6.0 million in cash at the closing of the transaction and (ii) $4.0 million in the form of a non-interest-bearing promissory note with CinPax and was paid to the Company on November 22, 2022. The CinRx Purchase Agreement provides CinPax the right to nominate a director to be approved to sit on the Company’s Board of Directors and a board observer, which was subsequently approved by the Company’s board.
Common stock is generally recorded at fair value at the date of issuance. In determining the fair value of the Class A common stock issued to CinPax, the Company considered the closing price of the common stock on the Transaction Date. The Company did not make an adjustment to the fair value for sale restrictions on the stock in accordance with guidance recently adopted in ASU 2022-03. See the “Recently Issued Accounting Guidance” in this quarterly report on Form 10-Q for details of the ASU. Accordingly, the Company determined that cash consideration of $3.0 million should be recorded as fair value of the Class A common stock at the effective date, utilizing the Class A common stock closing price of $0.72$28.80 at the effective date.
The CinRx Purchase Agreement also provides CinRx warrants to purchase up to 1,200,00030,000 shares of common stock at an initial exercise of price of approximately $0.72$28.80 per share (the “CinRx Warrants”). The CinRx Warrants were initially measured at fair value of $0.4 million using the Black-Scholes option model at the time of issuance and will be recorded in Warrant liability related party in the Condensed Consolidated Balance Sheets and will be subsequently remeasured at fair value through earnings on a recurring basis. (see Note 13)12)
The CinRx Warrants will become exercisable by CinRx only if (i) the Company receives approval from the U.S. Food and Drug Administration (“FDA Approval”) to market and distribute the pharmaceutical product containing the Company’s proprietary candidate, cadisegliatin (TTP399) (the “Product”), or (ii) the Company is acquired by a third party, sells all or substantially all of its assets related to the Product to a third party or grants a third party an exclusive license to develop, commercialize and manufacture the Product in the United States. If neither of these events happen within five years of the date of the issuance of the CinRx Warrants, the CinRx Warrants will expire and not be exercisable by CinRx. The exercise price of the CinRx Warrants and the number of shares issuable upon exercise of the CinRx Warrants are subject to adjustments in accordance with the terms of the CinRx Warrants.
Additionally, in conjunction with the CinRx Purchase Agreement the Company and CinRx entered into a Master Service Agreement (“CinRx MSA”) whereby CinRx provides the Company with consulting, preclinical and clinical trial services, as enumerated in project proposals negotiated between the Company and CinRx from time to time. (see Note 10)9)
The Company did not identify any other promises in the CinRx Purchase Agreement (aside from the issuance of common shares and the CinRx Warrants) and determined since there is no value ascribed to the CinRx MSA, the right to appoint a member and observer to the board of directors, that the remaining unallocated amount meets the definition of contributed equity and represents the amount in excess of par.
ATM Offering
In April 2020, The Company, CinPax and CinRx subsequently amended the Company entered into the SalesCinRx Purchase Agreement with Cantor as the sales agent, pursuant to which the Company may offer and sell, from time to time, through Cantor, shares of its Class A common stock, par value $0.01 per share, having an aggregate offering price of up to $13.0 million by any method deemed to be an “at the market offering” as
defined in Rule 415(a)(4) under the Securities Act (the “ATM Offering”). The shares are offered and sold pursuant to the Company’s shelf registration statement on Form S-3.
On January 14, 2021 and June 25, 2021, the Company filed a prospectus supplementFebruary 27, 2024, in connection with the Private Placement. The CinRx Purchase Agreement provides CinPax the right for two years following the Closing to designate a board observer, which has been subsequently approved by the Company’s board.
ATM Offering to increase the size of the at-the-market offering
On February 28, 2024, we entered into a sales agreement (the “TD Cowen Sales Agreement”) with Cowen and Company, LLC (“TD Cowen”), pursuant to which the Companywe may offer and sell, from time to time, through or to Cantor,TD Cowen, as sales agent or principal, shares of the Company’s Class A common stock, by an aggregate offering price of $5.5 million and $50.0 million, respectively.
During the three and nine months ended September 30, 2023 and 2022, the Company did not sell any shares under the ATM Offering.
Lincoln Park Capital Transaction
On November 24, 2020, the Company entered into the LPC Purchase Agreement and a registration rights agreement (the “Registration Rights Agreement”), pursuant to which the Company has the right to sell to Lincoln Park shares of the Company’sour Class A common stock, having an aggregate valueoffering price of up to $47.0$50.0 million subject(the
“TD Cowen ATM Offering”). Pursuant to certain limitations and conditions set forthGeneral Instruction I.B.6 of Form S-3, in no event will we sell securities registered on the LPC Purchase Agreement. The Companyregistration statement relating to the TD Cowen ATM Offering with a value exceeding more than one-third of our public float in any 12-month period so long as our public float remains below $75.0 million. Under the terms of the TD Cowen Sales Agreement, we will controlpay TD Cowen a commission of 3.0% of the timing and amount of any salesaggregate proceeds from the sale of shares to Lincoln Park pursuant to the LPC Purchase Agreement. During the three and nine months ended September 30, 2023 and 2022, the Company did not sell any shares under the LPC Purchase Agreement.reimburse certain legal fees or other disbursements.
Note 10:9: Related-Party Transactions
MacAndrews & Forbes Incorporated
MacAndrews directly or indirectly controls 23,084,267577,108 shares of Class B common stock. Further, as of September 30, 2023,March 31, 2024, MacAndrews directly or indirectly holds 36,519,212912,982 shares of the Company’s Class A common stock. As a result, MacAndrews’ holdings represent approximately 57.0%49.5% of the combined voting power of the Company’s outstanding common stock.
The Company has entered into several agreements with MacAndrews or its affiliates as further detailed below:
Letter Agreements
The Company had previously entered into the Letter Agreements with MacAndrews. Under the terms of the Letter Agreements, during the one year commitment period beginning on the date of each Letter Agreement, the Company had the right to sell to MacAndrews shares of its Class A common stock at a specified price per share, and MacAndrews had the right (exercisable up to three times) to require the Company to sell to it shares of Class A common stock at the same price. The commitment period of each of the Letter Agreements has now expired. In addition, in connection with and as a commitment fee for the entrance into certain of these Letter Agreements, the Company also issued MacAndrews warrants (the “Letter Agreement Warrants”) to purchase additional shares of the Company’s Class A common stock.
The Letter Agreement Warrants have been recorded as warrant liability, related party within the Company’s Condensed Consolidated Balance Sheets based on their fair value. The issuance of the Letter Agreement Warrants was considered to be a cost of equity recorded as a reduction to additional paid-in capital.
Exchange Agreement
Pursuant to the terms of the Exchange Agreement, but subject to the Amended and Restated LLC Agreement of vTv Therapeutics LLC, the vTv Units (along with a corresponding number of shares of the Class B common stock) are exchangeable for (i) shares of the Company’s Class A common stock on a one-for-one basis or (ii) cash (based on the fair market value of the Company’s Class A common stock as determined pursuant to the Exchange Agreement), at the Company’s option (as the managing member of vTv Therapeutics LLC), subject to customary conversion rate adjustments for stock splits, stock dividends and reclassifications. Any decision to require an exchange for cash rather than shares of Class A common stock will ultimately be determined by the entire Board of Directors. As of September 30, 2023,March 31, 2024, MacAndrews had not exchanged any shares under the provisions of the Exchange Agreement.
Tax Receivable Agreement
The Company and MacAndrews are party to a tax receivable agreement (the “Tax Receivable Agreement”), which provides for the payment by the Company to M&F TTP Holdings Two LLC (“M&F”), as successor in interest to vTv Therapeutics Holdings, LLC (“vTv Therapeutics Holdings”), and M&F TTP Holdings LLC (or certain of its transferees or other assignees) of 85% of the amount of cash savings, if any, in U.S. federal, state and local income tax or franchise tax that
the Company actually realizes (or, in some circumstances, the Company is deemed to realize) as a result of (a) the exchange of Class B common stock, together with the corresponding number of vTv Units, for shares of the Company’s Class A common stock (or for cash), (b) tax benefits related to imputed interest deemed to be paid by the Company as a result of the Tax Receivable Agreement and (c) certain tax benefits attributable to payments under the Tax Receivable Agreement.
As no shares have been exchanged by MacAndrews pursuant to the Exchange Agreement (discussed above), the Company has not recognized any liability, nor has it made any payments pursuant to the Tax Receivable Agreement as of September 30, 2023.March 31, 2024.
Investor Rights Agreement
The Company is party to an investor rights agreement with M&F, as successor in interest to vTv Therapeutics Holdings (the “Investor Rights Agreement”). The Investor Rights Agreement provides M&F with certain demand, shelf, and piggyback registration rights with respect to its shares of Class A common stock and also provides M&F with certain governance rights, depending on the size of its holdings of Class A common stock. Under the Investor Rights Agreement,
M&F was initially entitled to nominate a majority of the members of the Board of Directors and designate the members of the committees of the Board of Directors.
G42 Investments
On May 31, 2022, The Investor Rights Agreement was amended on February 27, 2024 to alter M&F governance rights that now entitles M&F the Company entered into a common stock purchase agreement with G42 Investments pursuantright to which the Company sold to G42 Investments 10,386,274 sharesdesignate two members of the Company’s Class A common stock at a price per shareour Board of approximately $2.41, for an aggregate purchase price of $25.0 million, which was paid (i) $12.5 million in cash at the closingDirectors, and (ii) $12.5 million in the form of a promissory note of G42 Investments to be paid on May 31, 2023 (the “G42 Promissory Note”). Asas part of the G42 Purchase Agreement, G42 Investments put forwardPrivate Placement, the Private Placement Investors have rights to designate three members of our Board of Directors, making it more difficult for a director as appointee andthird party to acquire control of our Board. The agreement with the Company’s boardPrivate Placement Investors also provides that five of our directors appointed the new directormust approve certain actions including any acquisition by a third party, which makes it more difficult for our Board of Directors to the Company’s board on July 11, 2022. On February 28, 2023, the Company and G42 Investments entered into an amendment of the common stock purchase agreement pursuant to which G42 Investments agreed to accelerate payment of the amount due under the G42 Promissory Note. On February 28, 2023, the Company received $12.0 million from G42 Investments, which representedapprove such a 3.75% discount to the full amount due under the G42 Promissory Note, in full and final satisfaction of the promissory note, resulting in a loss of $0.3 million and was recognized as a component of other income/(expense) in the Company’s Condensed Consolidated Statements of Operations.
CinRx Pharma, LLC
Master Services Agreement
On July 22, 2022, the Company entered into the CinRx MSA with CinRx. Under the CinRx MSA, CinRx provides the Company with consulting and clinical trial services, as enumerated in project proposals negotiated between the Company and CinRx from time to time. As of October 10, 2022, the Company agreed to pay CinRx fees of up to $0.2 million per month until approximately December 2024 in respect of ongoing agreed project proposals under the CinRx MSA, plus out-of-pocket expenses incurred by CinRx on the Company’s behalf. Dr. Jonathan Isaacsohn, who was appointed as chair of the Company’s board of directors on August 9, 2022, is the President and Chief Executive Officer of CinRx. CinPax, a subsidiary of CinRx, currently holds 4,154,549 shares of the Company’s Class A common stock.transaction.
Note 11:10: Income Taxes
The Company is subject to U.S. federal income taxes as well as state taxes. The Company did not record an income tax provision for the three and nine months ended September 30, 2023. The Company’s income tax provision for the ninethree months ended September 30, 2022March 31, 2024 was $0.2$0.1 million representing foreign withholding taxes incurredaccrued in connection with payments receivedrevenue recorded under license agreements with foreign entities. The Company did not record an income tax provision for the three months ended September 30, 2022.March 31, 2023.
Management has evaluated the positive and negative evidence surrounding the realization of its deferred tax assets, including the Company’s history of losses, and under the applicable accounting standards determined that it is more likely than notmore-likely-than-not that the deferred tax assets will not be realized. The difference between the effective tax rate of the Company and the U.S. statutory tax rate of 21% on September 30, 2023March 31, 2024, is due to the valuation allowance against the Company’s expected net operating losses.
As discussed in Note 10,9, the Company is party to a tax receivable agreement with a related party which provides for the payment by the Company to M&F (or certain of its transferees or other assignees) of 85% of the amount of cash savings, if any, in U.S. federal, state and local income tax or franchise tax that the Company actually realizes (or, in some circumstances, the Company is deemed to realize) as a result of certain transactions. As no transactions have occurred which
would trigger a liability under this agreement, the Company has not recognized any liability related to this agreement as of September 30, 2023.March 31, 2024.
Note 12:11: Net Loss per Share
Basic loss per share is computed by dividing net loss attributable to vTv Therapeutics Inc. by the weighted average number of shares of Class A common stock outstanding during the period. Diluted loss per share is computed giving effect to all potentially dilutive shares. Diluted loss per share for all periods presented is the same as basic loss per share as the inclusion of potentially issuable shares would be antidilutive.
A reconciliation of the numerator and denominator used in the calculation of basic and diluted net loss per share of Class A common stock is as follows (amounts in thousands, except per share amounts):
| | For the Three Months Ended September 30, | | For the Nine Months Ended September 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| For the Three Months Ended March 31, | |
| For the Three Months Ended March 31, | |
| For the Three Months Ended March 31, | |
| 2024 | |
| 2024 | |
| 2024 | |
| Numerator: | |
| Numerator: | |
| Numerator: | Numerator: | | | | | | | |
| Net loss | Net loss | $ | (8,540) | | | $ | (5,468) | | | $ | (21,525) | | | $ | (18,983) | |
| Net loss | |
| Net loss | |
| Less: Net loss attributable to noncontrolling interests | |
| Less: Net loss attributable to noncontrolling interests | |
| Less: Net loss attributable to noncontrolling interests | Less: Net loss attributable to noncontrolling interests | (1,886) | | | (1,207) | | | (4,753) | | | (4,564) | |
| Net loss attributable to common shareholders of vTv Therapeutics Inc., basic and diluted | Net loss attributable to common shareholders of vTv Therapeutics Inc., basic and diluted | (6,654) | | | (4,261) | | | (16,772) | | | (14,419) | |
| Net loss attributable to common shareholders of vTv Therapeutics Inc., basic and diluted | |
| Net loss attributable to common shareholders of vTv Therapeutics Inc., basic and diluted | |
| Denominator: | Denominator: | | | | | | | |
| Weighted average vTv Therapeutics Inc. Class A common stock, basic and diluted | 81,483,600 | | | 80,490,121 | | | 81,483,600 | | | 72,649,531 | |
| Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted | $ | (0.08) | | | $ | (0.05) | | | $ | (0.21) | | | $ | (0.20) | |
| Denominator: | |
| Denominator: | |
Weighted average vTv Therapeutics Inc. Class A common stock, basic and diluted (1)(*) | |
Weighted average vTv Therapeutics Inc. Class A common stock, basic and diluted (1)(*) | |
Weighted average vTv Therapeutics Inc. Class A common stock, basic and diluted (1)(*) | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
Net loss per share of vTv Therapeutics Inc. Class A common stock, basic and diluted (*) | |
| (*) Adjusted retroactively for reverse stock split | |
| (*) Adjusted retroactively for reverse stock split | |
| (*) Adjusted retroactively for reverse stock split | |
| | | | | | | | |
| (1) | The shares underlying the pre-funded warrants to purchase shares of the Company’s common stock have been included in the calculation of the weighted-average number of shares outstanding, basic and diluted, for the three months ended March 31, 2024. |
Potentially dilutive securities not included in the calculation of dilutive net loss per share are as follows:
| | | | | | | | | | | |
| September 30, 2023 | | September 30, 2022 |
Class B common stock (1) | 23,093,860 | | | 23,093,860 | |
| Common stock options granted under the Plan | 9,545,439 | | | 7,718,944 | |
| Common stock warrants | 3,214,503 | | | 3,214,503 | |
| Total | 35,853,802 | | | 34,027,307 | |
| | | | | | | | | | | |
| March 31, 2024 | | March 31, 2023 |
Class B common stock (1)(*) | 577,349 | | | 577,349 | |
Common stock options granted under the Plan (*) | 248,622 | | | 227,372 | |
Common stock warrants (*) | 75,595 | | | 80,359 | |
Total (*) | 901,566 | | | 885,080 | |
| (*) Adjusted retroactively for reverse stock split | | | |
_____________________________________________________
| | | | | | | | |
| (1) | Shares of Class B common stock do not share in the Company’s earnings and are not participating securities. Accordingly, separate presentation of loss per share of Class B common stock under the two-class method has not been provided. Each share of Class B common stock (together with a corresponding vTv Unit) is exchangeable for one share of Class A common stock. |
Note 13:12: Fair Value of Financial Instruments
The carrying amount of certain of the Company’s financial instruments, including cash and cash equivalents, net accounts receivable, note receivable, accounts payable and other accrued liabilities approximate fair value due to their short-term nature.
The Company measures the value of its equity investments without readily determinable fair values at cost minus impairment, if any, plus or minus changes resulting from observable price changes in orderly transactions for the identical or similar investment. During the year ended December 31, 2021, Reneo completed its initial public offering. As a result, the fair value of the Company’s investment in Reneo’s common stock now has a readily determinable market value and is no longer eligible for the practical expedient for investments without readily determinable fair market values.
Assets and Liabilities Measured at Fair Value on a Recurring Basis
The Company evaluates its financial assets and liabilities subject to fair value measurements on a recurring basis to determine the appropriate level in which to classify them for each reporting period. This determination requires significant
judgments. The Company determined that the G42 Promissory Note receivable was level 2 and the fair value measurement was based on the market yield curves. The following table summarizes the conclusions reached regarding fair value measurements as of September 30, 2023March 31, 2024 and December 31, 20222023 (in thousands):
| | Balance at March 31, 2024 | | | Balance at March 31, 2024 | | Quoted Prices in Active
Markets for Identical Assets
(Level 1) | | Significant Other Observable
Inputs
(Level 2) | | Significant Unobservable Inputs
(Level 3) |
| | | Balance at September 30, 2023 | | Quoted Prices in Active
Markets for Identical Assets
(Level 1) | | Significant Other Observable
Inputs
(Level 2) | | Significant Unobservable Inputs
(Level 3) |
| Assets: | | | | | | | |
| Equity securities with readily determinable fair value | $ | 4,387 | | | $ | 4,387 | | | $ | — | | | $ | — | |
| Total | $ | 4,387 | | | $ | 4,387 | | | $ | — | | | $ | — | |
| | Liabilities: | |
| | Liabilities: | |
| | | Liabilities: | Liabilities: | |
Warrant liability, related party (1) | Warrant liability, related party (1) | $ | 278 | | | $ | — | | | $ | — | | | $ | 278 | |
Warrant liability, related party (1) | |
Warrant liability, related party (1) | |
| Total | Total | $ | 278 | | | $ | — | | | $ | — | | | $ | 278 | |
| | Balance at December 31, 2023 | | | Balance at December 31, 2023 | | Quoted Prices in Active
Markets for Identical Assets
(Level 1) | | Significant Other Observable
Inputs
(Level 2) | | Significant Unobservable Inputs
(Level 3) |
| | | Balance at December 31, 2022 | | Quoted Prices in Active
Markets for Identical Assets
(Level 1) | | Significant Other Observable
Inputs
(Level 2) | | Significant Unobservable Inputs
(Level 3) |
| Assets: | | | | | | | |
| Equity securities with readily determinable fair value | $ | 1,343 | | | $ | 1,343 | | | $ | — | | | $ | — | |
| Total | $ | 1,343 | | | $ | 1,343 | | | $ | — | | | $ | — | |
| | Liabilities: | |
| | Liabilities: | |
| | | Liabilities: | Liabilities: | |
Warrant liability, related party (1) | Warrant liability, related party (1) | $ | 684 | | | $ | — | | | $ | — | | | $ | 684 | |
Warrant liability, related party (1) | |
Warrant liability, related party (1) | |
| Total | Total | $ | 684 | | | $ | — | | | $ | — | | | $ | 684 | |
_____________________________
| | | | | |
| (1) | Fair value determined using the Black-Scholes option pricing model. Expected volatility is based on the historical volatility of the Company’s common stock over the most recent period. The risk-free rate is based on the U.S. Treasury yield curve in effect at the time of valuation. |
| | Changes in Level 3 instruments for the three months ended September 30, |
| Balance at January 1 | | Net Change in
fair value included in
earnings | | Purchases /
Issuance | | Sales /
Repurchases | | Balance at September 30, |
| 2023 | | | | | | | | | |
| Changes in Level 3 instruments for the three months ended March 31, | | | Changes in Level 3 instruments for the three months ended March 31, |
| Balance at January 1 | | | Balance at January 1 | | Net Change in
fair value included in
earnings | | Purchases /
Issuance | | Sales /
Repurchases | | Balance at March 31, |
| 2024 | |
| Warrant liability, related party | |
| Warrant liability, related party | |
| Warrant liability, related party | Warrant liability, related party | $ | 684 | | | $ | (406) | | | $ | — | | | $ | — | | | $ | 278 | |
| Total | Total | $ | 684 | | | $ | (406) | | | $ | — | | | $ | — | | | $ | 278 | |
| | 2022 | |
| 2023 | |
| 2023 | |
| 2023 | |
| Warrant liability, related party | |
| Warrant liability, related party | |
| Warrant liability, related party | Warrant liability, related party | $ | 1,262 | | | $ | (221) | | | $ | 368 | | | $ | — | | | $ | 1,409 | |
| Total | Total | $ | 1,262 | | | $ | (221) | | | $ | 368 | | | $ | — | | | $ | 1,409 | |
There were no transfers into or out of level 3 instruments and/or between level 1 and level 2 instruments during the three and nine months ended September 30, 2023.March 31, 2024. Gains and losses recognized due to the change in fair value of the warrant liability, related party are recognized as a component of other income –expense, related party in the Company’s Condensed Consolidated Statements of Operations.
The fair value of the Letter Agreement Warrants was determined using the Black-Scholes option pricing model or option pricing models based on the Company’s current capitalization. Expected volatility is based on the historical volatility
of the Company’s common stock over the most recent period. The risk-free rate is based on the U.S. Treasury yield curve in effect at the time of valuation. Significant inputs utilized in the valuation of the Letter Agreement Warrants as of September 30, 2023March 31, 2024 and December 31, 20222023, were:
| | September 30, 2023 | | December 31, 2022 |
| Range | Weighted Average | | Range | Weighted Average |
| March 31, 2024 | | | March 31, 2024 | | December 31, 2023 |
| Range | | | Range | Weighted Average | | Range | Weighted Average |
| Expected volatility | Expected volatility | 68.37% - 77.01% | 76.06% | | 76.94% - 85.88% | 82.17% | Expected volatility | 93.31% - 142.10% | 101.45% | | 79.96% - 89.61% | 81.55% |
| Risk-free interest rate | Risk-free interest rate | 4.78% - 5.38% | 4.88% | | 4.11% - 4.43% | 4.19% | Risk-free interest rate | 4.45% - 5.25% | 4.64% | | 4.01% - 4.87% | 4.15% |
The fair value of the CinRx Warrants was determined using the Black-Scholes option pricing model. Expected volatility is based on the historical volatility of the Company’s common stock over the most recent period. The risk-free rate is based on the U.S. Treasury yield curve in effect at the time of valuation. Significant inputs utilized in the valuation of the CinRx Warrants as of September 30, 2023March 31, 2024, were:
| | | | | |
| Expected volatility | 82.3891.6 | % |
| Expected life of options in years | 3.753.3 |
| Risk-free interest rate | 4.734.4 | % |
| Expected dividend yield | — | % |
The weighted average expected volatility and risk-free interest rate was based on the relative fair values of the warrants.
Changes in the unobservable inputs noted above would impact the amount of the liability for the Letter Agreement Warrants and CinRx Warrants. Increases (decreases) in the estimates of the Company’s annual volatility would increase (decrease) the liability and an increase (decrease) in the annual risk-free rate would increase (decrease) the liability.
Note 14:13: Subsequent Events
On October 16, 2023,The Company evaluated subsequent events through May 9, 2024 and determined that there have been no events that have occurred that would require adjustments to our disclosures or the Company received a Section 228(e) notice from Anteris that the stockholders voted in favor of the dissolution of the Company and the adoption of a plan of Liquidation and Dissolution (“Plan of Dissolution”) (See Note 5).
On October 30, 2023, vTv LLC, entered into a Common Stock Repurchase Agreement (the “Repurchase Agreement”) with Reneo pursuant to which Reneo agreed to purchase all shares of common stock of Reneo that had been owned by vTv LLC. Pursuant to the Repurchase Agreement, Reneo agreed to pay vTv LLC gross proceeds of approximately $4.4 million, which was received by vTv LLC on October 30, 2023. vTv LLC had acquired the Reneo stock in connection with the December 21, 2017 License Agreement between vTv LLC and Reneo, which remains in effect.unaudited condensed consolidated financial statements.
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Cautionary Note Regarding Forward Looking Statements
As used in this Quarterly Report on Form 10-Q, the “Company”, the “Registrant”, “we” or “us” refer to vTv Therapeutics Inc. and “vTv LLC” refers to vTv Therapeutics LLC. The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our financial statements and related notes that appear elsewhere in this report. In addition to historical financial information, this Quarterly Report on Form 10-Qthe following discussion contains forward-looking statements within the meaning of the federal securities laws regarding, among other things,that reflect our intentions, plans, estimates, assumptions predictions and beliefs. Although we believe that these forward-looking statements are based upon reasonable assumptions that we have made in light of our experience in the industry, as well as our perceptions of historical trends, current conditions, expected future developments and other factors that we believe are appropriate under the circumstances, ourOur actual results could differ materially from those discussed in the forward-looking statements. Factors that could cause or contribute to these differences include those discussed below and elsewhere in this report under “Part II, Other Information—Item 1A, Risk Factors” and under the heading “Risk Factors” under Item 1A of Part I in our Annual Report on Form 10-K.Factors.” Forward-looking statements include information concerning our possible or assumed future results of operations, business strategies and operations, financing plans, potential growth opportunities, potential market opportunities, potential results of our drug development efforts or trials, and the effects of competition. Forward-looking statements include all statements that are not historical facts and can be identified by terms such as “anticipates,” “believes,” “could,” “seeks,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would” or similar expressions and the negatives of those terms. Given the numerousthese uncertainties, and factors to our operations and business environment, all of which are difficult to predict and many of which are beyond our control, you should not place undue reliance on these forward-looking statements and understand that these statements are not guarantees of performance or results.statements. Also, forward-looking statements represent our management’s plans, estimates, assumptions and beliefs only as of the date of this report. Except as required by law, we assume no obligation to update these forward-looking statements publicly or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future.
Company Overview
We are a clinical stage pharmaceutical company focused on treating metabolic and inflammatory diseases to minimize their long-term complications and improve the lives of patients. We have an innovative pipeline of first-in-class small molecule clinical and preclinical drug candidates. Our lead program is cadisegliatin (TTP399)(TTP399), an orally administered, small molecule, liver-selective glucokinase activator (“GKA”) as an adjunctive therapy to insulin for the treatment of type 1 diabetes (“T1D”).
Recent Developments
On September 6, 2023, the Board approved,February 27, 2024, we entered into a securities purchase agreement (the “Securities Purchase Agreement”) with certain institutional accredited investors (the “Private Placement Investors”), pursuant to which we agreed to issue and recommended that the stockholders of the Company entitled to vote thereon, approve, via written consent, the Amendmentsell to the CertificatePrivate Placement Investors in a private placement (the “Private Placement”) (i) an aggregate of Incorporation to effect a reverse stock split464,377 shares (the “Private Placement Shares”) of the issued and outstandingour Class A common stock, at a purchase price of $11.81 per share, and Class B common stock, by a ratio(ii) pre-funded warrants (the “Private Placement Pre-Funded Warrants”) to purchase up to an aggregate of any whole number in the range3,853,997 shares of 1-for-10 to 1-for-40 (the “Reverse Stock Split”), with such ratio to be determined at the discretion of the Board and with such action to be effected at such time and date, if at all, as determined by the Board on or prior to December 31, 2023. On September 13, 2023, the holders of a majority of the voting power of the Company’s issued and outstanding capital stock entitled to vote thereon (the “Consenting Stockholders”) approved the Reverse Stock Split by means of a written consent. The same ratio will be used to effect a reverse stock split of both theour Class A common stock and(the “Private Placement Warrant Shares”) at a purchase price of $11.80 per Private Placement Pre-Funded Warrant (representing the Class B common stock.$11.81 per Private Placement Share purchase price less the exercise price of $0.01 per Private Placement Warrant Share). We received aggregate gross proceeds from the Private Placement of approximately $51.0 million, before deducting offering expenses payable by us. The Reverse Stock SplitPrivate Placement Pre-Funded Warrants are exercisable at any time after their original issuance and will not changeexpire.
On March 5, 2024, the numberCompany entered into a letter agreement with the Private Placement Investors pursuant to which the Private Placement Investors agreed to exchange an aggregate of authorized116,493 Private Placement Shares for an aggregate of 116,590 Private Placement Pre-Funded Warrants.
ATM
On February 28, 2024, we entered into a sales agreement (the “TD Cowen Sales Agreement”) with Cowen and Company, LLC (“TD Cowen”), pursuant to which we may offer and sell, from time to time, through or to TD Cowen, as sales agent or principal, shares of our Class A common stock, having an aggregate offering price of up to $50.0 million (the “TD Cowen ATM Offering”). Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell securities registered on the registration statement relating to the TD Cowen ATM Offering with a value exceeding more than one-third of our public float in any 12-month period so long as our public float remains below $75.0 million. Under the terms of the TD Cowen Sales Agreement, we will pay TD Cowen a commission of 3% of the aggregate proceeds from the sale of shares and reimburse certain legal fees or Class B common stock.other disbursements.
The Board has not yet determined the date on which the reverse stock split will occur or the ratio.
Our Product Pipeline
The following table summarizes our drug candidates, their partnership status and their respective stages of development:
Our Type 1 Diabetes Program – Cadisegliatin (TTP399)
The Company has alignment with the FDA on trial designs that will support an efficient regulatory pathway to support registration ofU.S. Food and Drug Administration (FDA) granted Breakthrough Therapy designation in 2021 for cadisegliatin (TTP399) as an adjunctive therapy to insulin for the treatment of type 1 diabetes consistent with the breakthrough therapy designation granted by the FDA and expects to initiate studies as soon as possible.diabetes. The FDA and the Company have agreed on the primary endpoint for the studies as the reduction in the number of hypoglycemic events between placebo and cadisegliatin (TTP399) treated groups.
In October 2021, we announced positive results of a mechanistic study of cadisegliatin (TTP399) in patients with T1D. The study demonstrated that patients with T1D taking cadisegliatin (TTP399) experienced no increase in ketone levels relative to placebo during a period of acute insulin withdrawal, indicating no increased risk of ketoacidosis. Consistent with previous clinical studies, improved fasting plasma glucose levels and fewer hypoglycemic events were observed in the cadisegliatin (TTP399) treated group during the week of treatment prior to the insulin withdrawal test. The results of the mechanistic study provided additional evidence to support the idea that treatment with cadisegliatin (TTP399) will not increase the risk of diabetic ketoacidosis (“DKA”) in patients with T1D. The data demonstrate that in contrast to agents such as SGLT2 inhibitors and GLP-1RAs, cadisegliatin (TTP399) does not increase the risk of ketoacidosis when used as an adjunctive therapy to insulin in individuals with T1D. Moreover, these findings support prior studies that demonstrate that cadisegliatin (TTP399) improves glucose control and reduces hypoglycemia and suggests a protective effect of cadisegliatin (TTP399) against acidosis in people with T1D. Full study results were published in the Diabetes Obesity and Metabolism journal in conjunction with the 82nd American Diabetes Association Scientific Sessions on June 6, 2022.
In April 2021, we announced that the FDA granted Breakthrough Therapy Designation (“BTD) for cadisegliatin (TTP399) as an adjunctive therapy to insulin for the treatment of T1D. This designation provides a sponsor with added support and the potential to expedite development and review timelines for a promising new investigational medicine. The Breakthrough Therapy designation for cadisegliatin in T1D was supported by the positive results from the Phase 2 SimpliciT-1 Study, a multi-center, randomized, double-blind, adaptive study assessing the safety and efficacy of cadisegliatin as an adjunct to insulin therapy in adults with T1D. In this trial, treatment with cadisegliatin resulted in a statistically significant improvement in HbA1c relative to placebo and a clinically meaningful decrease (40%) in the frequency of severe and symptomatic hypoglycemia. Cadisegliatin demonstrated a favorable safety profile, in which abnormal levels of serum or urine ketones were detected less frequently in patients taking cadisegliatin than those taking placebo.
In May of 2023, the FDA issued new draft guidance on "Diabetes Mellitus: Efficacy Endpoints for Clinical Trials Investigating Antidiabetic Drugs and Biological Products" which, for the first time, permitted the use of hypoglycemia as an endpoint to support a label claim. Consistent with this guidance and with input from the FDA, we have initiated a Phase 3 double-blind controlled trial to assess the effect of cadisegliatin on reducing the frequency of Level 2 hypoglycemia (blood glucose levels are less than 54 mg/dL or 3 mmol/L, regardless of symptoms) and Level 3 hypoglycemia ("severe" hypoglycemia e.g., requiring assistance of another person) in 150 patients with type 1 diabetes. Participants in the trial will be randomized to two doses of cadisegliatin or placebo. Reduction in glycated hemoglobin (HbA1c), a traditional efficacy endpoint in diabetes trials, is a key secondary endpoint to assess the potential of cadisegliatin to reduce hyperglycemia, Following the initial assessment of efficacy after six months of treatment, participants will remain on trial for another six months to assess the durability of potential beneficial effects and safety. The study protocol was submitted to the FDA on February 29, 2024. The trial will be conducted in the U.S. only and is expected to complete enrollment by the fourth quarter in 2024 providing top line 1-year data by the first quarter in 2026.
Concurrently, we will be working on the design for two international registrational studies for cadisegliatin in type 1 diabetes,which we expect to start in 2026.
In addition, we continue to work with our partner, G42 Investments AI Holding RSC Ltd. (“G42”), to initiate a double-blind, randomized, controlled Phase 2 trial in the Middle East region in 450 insulin-using patients with type 2 diabetes. We expect that trial to begin in 2024.
Holding Company Structure
vTv Therapeutics Inc. is a holding company and its principal asset is a controlling equity interest in vTv Therapeutics LLC (“vTv LLC”), the principal operating subsidiary. We have determined that vTv LLC is a variable-interest entity (“VIE”) for accounting purposes and that vTv Therapeutics Inc. is the primary beneficiary of vTv LLC because (through its managing
member interest in vTv LLC and the fact that the senior management of vTv Therapeutics Inc. is also the senior management of vTv LLC) it has the power to direct all of the activities of vTv LLC, which include those that most significantly impact vTv LLC’s economic performance. vTv Therapeutics Inc. has therefore consolidated vTv LLC’s results under the VIE accounting model in its consolidated financial statements.
Financial Overview
Revenue
To date, we have not generated any revenue from drug sales. Our revenue has been primarily derived from milestone payments, up-front proceeds and research fees under collaboration and license agreements.
In the future, we may generate revenue from a combination of product sales, license fees, milestone payments and royalties from the sales of products developed under licenses of our intellectual property. We expect that any revenue we generate will fluctuate from quarter to quarter as a result of the timing and amount of license fees, milestone and other payments, and the amount and timing of payments that we receive upon the sale of our products, to the extent any are successfully commercialized. If we fail to complete the development of our drug candidates in a timely manner or obtain regulatory approval for them, our ability to generate future revenue and our results of operations and financial position will be materially adversely affected.
Research and Development Expenses
Since our inception, we have focused our resources on our research and development activities, including conducting preclinical studies and clinical trials, manufacturing development efforts and activities related to regulatory filings for our drug candidates. We recognize research and development expenses as they are incurred. Our direct research and development expenses consist primarily of external costs such as fees paid to investigators, consultants, central laboratories and clinical research organizations in connection with our clinical trials, and costs related to acquiring and manufacturing clinical trial materials. Our indirect research and development costs consist primarily of cash and share-based compensation costs, the cost of employee benefits and related overhead expenses for personnel in research and development functions. Since we typically use our employee and infrastructure resources across multiple research and development programs such costs are not allocated to the individual projects.
From our inception, including our predecessor companies, through September 30, 2023, we have incurred approximately $623.9 million in research and development expenses.
Our research and development expenses by project for the three and nine months ended September 30,March 31, 2024 and 2023 and 2022 were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended September 30, | | Nine Months Ended September 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| Direct research and development expense: | | | | | | | |
| Cadisegliatin (TTP399) | $ | 1,854 | | | $ | 2,309 | | | $ | 8,943 | | | $ | 6,438 | |
| Other projects* | 163 | | | 173 | | | 475 | | | 417 | |
| Indirect research and development expense | 807 | | | 573 | | | 2,039 | | | 1,538 | |
| Total research and development expense | $ | 2,824 | | | $ | 3,055 | | | $ | 11,457 | | | $ | 8,393 | |
*Includes HPP737 and Azeliragon | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2024 | | 2023 | | | | |
| Direct research and development expense: | | | | | | | |
| Cadisegliatin | $ | 1,563 | | | $ | 3,056 | | | | | |
| HPP737 | 10 | | | 4 | | | | | |
| | | | | | | |
| Other projects | 213 | | | 249 | | | | | |
| Indirect research and development expense | 863 | | | 633 | | | | | |
| Total research and development expense | $ | 2,649 | | | $ | 3,942 | | | | | |
We plan to continue to incur significant research and development expenses for the foreseeable future as we continue the development of cadisegliatin (TTP399) and further advance the development of our other drug candidates, subject to the availability of additional funding.
The successful development of our clinical and preclinical drug candidates is highly uncertain. At this time, we cannot reasonably estimate the nature, timing or costs of the efforts that will be necessary to complete the remainder of the development of any of our clinical or preclinical drug candidates or the period, if any, in which material net cash inflows
from these drug candidates may commence. This is due to the numerous risks and uncertainties associated with the development of our drug candidates, including:
•the uncertainty of the scope, rate of progress and expense of our ongoing, as well as any additional, clinical trials and other research and development activities;
•the potential benefits of our candidates over other therapies;
•our ability to market, commercialize and achieve market acceptance for any of our drug candidates that we are developing or may develop in the future;
•future clinical trial results;
•our ability to enroll patients in our clinical trials;
•the timing and receipt of any regulatory approvalsapprovals;
•our ability to secure sufficient capital and cash resources, including access to available debt and equity financing and revenues from operations, to satisfy all of our short-term and longer-term cash requirements and other cash needs, at the times and in the amounts needed;
•legislation and regulatory actions and changes in laws or regulations; and
•the filing, prosecuting, defending and enforcing of patent claims and other intellectual property rights, and the expense of doing so.
A change in the outcome of any of these variables with respect to the development of a drug candidate could mean a significant change in the costs and timing associated with the development of that drug candidate. For example, if the FDA or another regulatory authority were to require us to conduct clinical trials beyond those that we currently anticipate will be required for the completion of clinical development of a drug candidate, or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time with respect to the development of that drug candidate.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries, benefits and related costs for employees in executive, finance, corporate development, human resources and administrative support functions. Other significant general and administrative expenses include accounting and legal services, expenses associated with obtaining and maintaining patents, cost of various consultants, occupancy costs and information systems.
Interest Income
Interest income represents noncash interest income related to the imputed interest from the G42 Promissory Note receivable using the effective interest method, which ended in 2023, and cash interest income representsfrom dividends and interest from our money market account, all of which are recognized in our Condensed Consolidated Statement of Operations.
Interest Expense
The Company’s interest expense is immaterial.Other (Expense)/Income, net
Other (Expense) Income, Net
Other income/expenseexpense/income primarily consists of unrealized gains or losses attributable to the changes in fair value of the equity investment held,investments in Reneo Pharmaceuticals, Inc ("Reneo"), the recognition of changes in fair value of the warrants to purchase shares of our Class A common stock held by related parties and the loss from the G42 Promissory Notepromissory note early redemption on February 28, 2023 and the impairment charge from Anteris Bio, Inc. (“Anteris”) liquidation and dissolution.2023.
Results of Operations
Comparison of the three months ended September 30,March 31, 2024 and 2023 and 2022
The following table sets forth certain information concerning our results of operations for the periods shown:
| | (dollars in thousands) | (dollars in thousands) | Three Months Ended September 30, | (dollars in thousands) | Three Months Ended March 31, |
| Statement of operations data: | Statement of operations data: | 2023 | | 2022 | | Change | Statement of operations data: | 2024 | | 2023 | | Change |
| Revenue | Revenue | $ | — | | $ | — | | $ | — | Revenue | $ | 1,000 | | $ | — | | $ | 1,000 |
| Operating expenses: | Operating expenses: | |
| Research and development | |
| Research and development | |
| Research and development | Research and development | 2,824 | | 3,055 | | (231) | 2,649 | | 3,942 | | (1,293) |
| General and administrative | General and administrative | 2,544 | | 2,634 | | (90) | General and administrative | 3,978 | | 3,485 | | 493 |
| Total operating expenses | Total operating expenses | 5,368 | | 5,689 | | (321) | Total operating expenses | 6,627 | | 7,427 | | (800) |
| Operating loss | Operating loss | (5,368) | | (5,689) | | 321 | Operating loss | (5,627) | | (7,427) | | 1,800 |
| Interest income | Interest income | 131 | | 150 | | (19) | Interest income | 79 | | 100 | | (21) |
| Interest expense | Interest expense | (4) | | (8) | | 4 | Interest expense | — | | — |
| Other (expense) income, net | Other (expense) income, net | (3,299) | | 79 | | (3,378) | Other (expense) income, net | (371) | | 1,553 | | (1,924) |
| Loss before income taxes and noncontrolling interest | Loss before income taxes and noncontrolling interest | (8,540) | | (5,468) | | (3,072) | Loss before income taxes and noncontrolling interest | (5,919) | | (5,774) | | (145) |
| Income tax provision | Income tax provision | — | | — | | — | Income tax provision | 100 | | — | | 100 |
| Net loss before noncontrolling interest | Net loss before noncontrolling interest | (8,540) | | (5,468) | | (3,072) | Net loss before noncontrolling interest | (6,019) | | (5,774) | | (245) |
| Less: net loss attributable to noncontrolling interest | Less: net loss attributable to noncontrolling interest | (1,886) | | (1,207) | | (679) | Less: net loss attributable to noncontrolling interest | (1,154) | | (1,275) | | 121 |
| Net loss attributable to vTv Therapeutics Inc. | Net loss attributable to vTv Therapeutics Inc. | $ | (6,654) | | $ | (4,261) | | $ | (2,393) | Net loss attributable to vTv Therapeutics Inc. | $ | (4,865) | | $ | (4,499) | | $ | (366) |
Revenue
Revenue for the three months ended March 31, 2024, includes a $1.0 million increase to the transaction price for the license performance obligation under the Newsoara License Agreement due to the satisfaction of a development milestones. There was no revenue for the three months ended September 30, 2023 and September 30, 2022.March 31, 2023.
Research and Development Expenses
Research and development expenses were $2.8$2.6 million and $3.1$3.9 million for the three months ended September 30,March 31, 2024 and 2023, and 2022, respectively. The decrease in research and development expenses during this period of $0.2$1.3 million or 7.6%32.8%, was primarily driven by i) lower spending on cadisegliatin (TTP399) of $0.4$1.5 million, due to decreases in toxicity study costs and drug productmanufacturing related costs as well as lower spending onpartially offset by increases in clinical trial preparationstart-up costs, offset byand ii) an increase in indirect costs and other projects of $0.2 million.
General and Administrative Expenses
General and administrative expenses were $2.5$4.0 million and $2.6$3.5 million for the three months ended September 30,March 31, 2024 and 2023, and 2022, respectively. The decreaseincrease in general and administrative expenses during this period of $0.1$0.5 million, or 3.4%14.1%, was primarily driven by i) decreasesincreases of $0.4$0.8 million in payroll related costs, ii) increases of $0.3 million in legal expense, ii)expenses, partially offset by iii) decreases of $0.3$0.5 million in other general and administrative costs, partially offset by iii) increases of $0.5 million in payroll costs and iv) increasesdecreases of $0.1 million in share-based expense.
Interest Income
Interest income for the three months ended September 30, 2023March 31, 2024, of $0.1 million, is related to dividend income from our money market account.account. Interest income for the three months ended September 30, 2022March 31, 2023, of $0.20.1 million, is related to imputed interest on the promissory notes.
Interest Expense
Interest expense for the three months ended September 30, 2023 and 2022 was insignificant.
Other (Expense)/Income, Net
Other expense was $3.3$0.4 million for the three months ended September 30, 2023March 31, 2024, and was driven by the recording of an impairment charge on a cost-method investment of $4.2 million and an unrealized gain related to our investment in Reneo, as well as gainslosses related to the change in the fair value of the outstanding warrants to purchase shares of our own stock issued to related parties (“Related Party Warrants”). Other income was $0.1$1.6 million for the three months ended September 30, 2022March 31, 2023,
and was driven by an unrealized gain related to our investment in Reneo, as well as thelosses related to the change in the fair value of the outstanding warrants to purchase shares of our own stock issued to related parties.
Comparison ofparties and the nine months ended September 30, 2023 and 2022
The following table sets forth certain information concerning our results of operations for the periods shown:
| | | | | | | | | | | | | | | | | |
| (dollars in thousands) | Nine Months Ended September 30, |
| Statement of operations data: | 2023 | | 2022 | | Change |
| Revenue | $ | — | | $ | 2,009 | | $ | (2,009) |
| Operating expenses: | | | | | |
| Research and development | 11,457 | | 8,393 | | 3,064 |
| General and administrative | 9,338 | | 9,813 | | (475) |
| Total operating expenses | 20,795 | | 18,206 | | 2,589 |
| Operating loss | (20,795) | | (16,197) | | (4,598) |
| Interest income | 384 | | 200 | | 184 |
| Interest expense | (6) | | (9) | | 3 |
| Other expense, net | (1,108) | | (2,777) | | 1,669 |
| Loss before income taxes and noncontrolling interest | (21,525) | | (18,783) | | (2,742) |
| Income tax provision | — | | 200 | | (200) |
| Net loss before noncontrolling interest | (21,525) | | (18,983) | | (2,542) |
| Less: net loss attributable to noncontrolling interest | (4,753) | | (4,564) | | (189) |
| Net loss attributable to vTv Therapeutics Inc. | $ | (16,772) | | $ | (14,419) | | $ | (2,353) |
Revenue
There was no revenue for the nine months ended September 30, 2023. Revenue for the nine months ended September 30, 2022 relates to the increase in the transaction price for the license performance obligations under the amended license agreement with Huadong due to the satisfaction of a development milestone.
Research and Development Expenses
Research and development expenses were $11.5 million and $8.4 million for the nine months ended September 30, 2023 and 2022, respectively. The increase in research and development expenses during the period of $3.1 million, or 36.5%, was primarily driven by i) higher spending on cadisegliatin (TTP399) of $2.5 million due to increases in drug product related costs as well as higher spending on trial preparation costs, and ii) an increase of $0.6 million in indirect costs and other projects.
General and Administrative Expenses
General and administrative expenses were $9.3 million and $9.8 million for the nine months ended September 30, 2023 and 2022, respectively. The decrease of $0.5 million has been primarily driven by i) decreases of $2.8 million in legal expense, ii) decreases of $0.8 million in severance costs, partially offset by iii) increases of $1.7 million in payroll costs, iv) increases of $1.2 million in other general and administrative costs, and v) increases of $0.2 million share-based expense.
Interest income
Interest income for the nine months ended September 30, 2023 of $0.4 million is related to imputed interest onloss from the G42 Promissory Note and dividend income from our money market account. Interest income for the nine months ended September 30, 2022 of $0.2 million is related to imputed interest on the promissory note.note early redemption.
Interest Expense
Interest expense for the nine months ended September 30, 2023 and 2022 was insignificant.
Other (Expense), Net
Other expense was $1.1 million for the nine months ended September 30, 2023, and is driven by the recording of a impairment charge on a cost-method investment of $4.2 million offset by an unrealized gain recognized related to the Company’s investment in Reneo as well as gains related to the change in the fair value of the outstanding warrants to
purchase shares of our own stock held by related parties. Other expense was $2.8 million for the nine months ended September 30, 2022, and is driven by an unrealized loss recognized related to the Company’s investment in Reneo as well as gains related to the change in the fair value of the outstanding warrants to purchase shares of our own stock issued to related parties.
Liquidity and Capital Resources
Liquidity and Going Concern
As of September 30, 2023,March 31, 2024, we had an accumulated deficit of $281.2$286.1 million. Since our inception, we have experienced a history of negative cash flows from operating activities. We anticipate that we will continue to incur losses and negative cash flow from operations for the foreseeable future as we continue our clinical trials. Further, we expect that we will need additional capital to continue to fund our operations. As of September 30, 2023,March 31, 2024, we had cash and cash equivalents of $8.2$52.3 million. The Company received proceeds of $4.4 million from the sale of Reneo Pharmaceuticals, Inc stock on October 30, 2023 (see Note 14).
Based on our current operating plan, we may rely onOn February 27, 2024, the remaining availabilityCompany closed a private placement financing of $37.3up to $51.0 million under our Controlled Equity OfferingSM Sales Agreement (the “Sales Agreement”) with Cantor Fitzgerald & Co. (“Cantor Fitzgerald”) pursuantand additionally granting investors the right to which we could offer and sell, from timepurchase up to time, sharesan additional $30.0 million of our Class A common stock (the “ATM Offering”) and our ability to sell approximately 9.4 million shares of Class A common stock to Lincoln Park Capital Fund, LLC (“Lincoln Park”) pursuant and subject to18 months following the limitationsclosing of the purchase agreement (the “LPC Purchase Agreement”). However,private placement financing. The financing raised will allow the abilityCompany to use these sources of capital is dependent on a number of factors, including the prevailing market price of and the volume of trading in our Class A common stock.further advance its lead program for cadisegliatin.
InIn addition to available cash and cash equivalents and available funds discussed above, we are seeking possible additional partnering opportunities for our GKA, GLP-1r and other drug candidates which we believe may provide additional cash for use in our operations and the continuation of the clinical trials for our drug candidates. We are evaluating several financing strategies to fund our planned and ongoing clinical trials, including direct equity investments and future public offerings of our common stock. The timing and availability of such additional financing are not yet known. We are currently in active discussions with respect to financing, partnering and licensing transactions for the future development of cadisegliatin (TTP399), but we may not be successful in completing such transactions. These factors raise substantial doubt about our ability to continue as a going concern.
ATM Offering
We haveTD Cowen Sales Agreement
On February 28, 2024, we entered into thea sales agreement (the "TD Cowen Sales AgreementAgreement") with Cantor FitzgeraldCowen and Company, LLC (“TD Cowen”) pursuant to which we may offer and sell, from time to time, through or to Cantor Fitzgerald,TD Cowen, as sales agent or principal, shares of our Class A common stock having an aggregate offering price of up to $68.5 million.$50.0 million, although we may only offer and sell under the TD Cowen ATM Offering up to one-third of the aggregate market value of our Class A common stock held by non-affiliates during any 12 calendar month period pursuant to General Instruction I.B.6 of Form S-3. We are not obligated to sell any shares under the TD Cowen Sales Agreement. Under the terms of the TD Cowen Sales Agreement, we will pay Cantor FitzgeraldTD Cowen a commission of up to 3% of the aggregate proceeds from the sale of shares and reimburse certain legal fees or other disbursements. As of September 30, 2023, we have sold $31.2 million worth of Class A common stock under the ATM Offering for net proceeds of $30.3 million, leaving $37.3 million available to be sold.
Lincoln Park Purchase Agreement
We have entered into the LPC Purchase Agreement, pursuant to which we have the right to sell to Lincoln Park shares of the Company’s Class A common stock having an aggregate value of up to $47.0 million. As of September 30, 2023, we have issued 5,331,306 of these shares for gross proceeds of approximately $11.1 million, leaving $35.9 million available to be sold.
Over the 36-month term of the LPC Purchase Agreement, we have the right, but not the obligation, from time to time, in our sole discretion, to direct Lincoln Park to purchase up to 250,000 shares per day (the “Regular Purchase Share Limit”) of the Class A common stock (each such purchase, a “Regular Purchase”). The Regular Purchase Share Limit will increase to 275,000 shares per day if the closing price of the Class A common stock on the applicable purchase date is not below $4.00 per share and will further increase to 300,000 shares per day if the closing price of the Class A common stock on the applicable purchase date is not below $5.00 per share. In any case, Lincoln Park’s maximum obligation under any single Regular Purchase will not exceed $2,000,000. The purchase price for shares of Class A common stock to be purchased by Lincoln Park under a Regular Purchase will be equal to the lower of (in each case, subject to the adjustments described in the LPC Purchase Agreement): (i) the lowest sale price for the Class A common stock on the applicable purchase date and (ii) the arithmetic average of the three lowest closing sales prices for the Class A common stock during the 10 consecutive trading days prior to the purchase date.
If we direct Lincoln Park to purchase the maximum number of shares of Class A common stock that we may sell in a Regular Purchase, then in addition to such Regular Purchase, and subject to certain conditions and limitations in the LPC Purchase Agreement, we may direct Lincoln Park to make an “accelerated purchase” and an “additional accelerated purchase”, each of an additional number of shares of Class A common stock which may not exceed the lesser of: (i) 300% of the number of shares purchased pursuant to the corresponding Regular Purchase and (ii) 30% of the total number of shares of the common stock traded during a specified period on the applicable purchase date as set forth in the LPC Purchase Agreement. The purchase price for such shares will be the lesser of (i) 97% of the volume weighted average price of the Class A common stock over a certain portion of the date of sale as set forth in the LPC Purchase Agreement and (ii) the closing sale price of the Class A common stock on the date of sale (an “Accelerated Purchase”). Under certain circumstances and in accordance with the LPC Purchase Agreement, we may direct Lincoln Park to purchase shares in multiple Accelerated Purchases on the same trading day.
The LPC Purchase Agreement also prohibits us from directing Lincoln Park to purchase any shares of its Class A common stock if those shares, when aggregated with all other shares of Class A common stock then beneficially owned by Lincoln Park and its affiliates, would result in Lincoln Park and its affiliates having beneficial ownership, at any single point in time, of more than 9.99% of the then total outstanding shares of Class A common stock as calculated pursuant to Section 13(d) of the Securities Exchange Act of 1934, as amended, and Rule 13d-3 thereunder.
Cash Flows
| | Nine Months Ended September 30, |
| 2023 | | 2022 |
| Three Months Ended March 31, | | | Three Months Ended March 31, |
| 2024 | | | 2024 | | 2023 |
| (dollars in thousands) | (dollars in thousands) | | | |
| Net cash used in operating activities | Net cash used in operating activities | $ | (16,167) | | | $ | (9,142) | |
| Net cash used in investing activities | — | | | (21) | |
| Net cash used in operating activities | |
| Net cash used in operating activities | |
| | Net cash provided by financing activities | Net cash provided by financing activities | 12,279 | | | 11,087 | |
| Net (decrease) increase in cash and cash equivalents | $ | (3,888) | | | $ | 1,924 | |
| Net cash provided by financing activities | |
| Net cash provided by financing activities | |
| Net increase in cash and cash equivalents | |
Operating Activities
For the ninethree months ended September 30, 2023,March 31, 2024, our net cash used in operating activities increased by $7.0$2.2 million from the ninethree months ended September 30, 2022March 31, 2023. The significant contributor to the change in cash used during the year wasdriven by working capital changes, and offset by cash received of $6.8 million in excess of the fair value of the Class A common stock issued to G42 investments in 2022.changes.
Investing Activities
There were no cash flows from investing activities for the ninethree months ended September 30,March 31, 2024 and 2023. For the nine months ended September 30, 2022, net cash used in investing activities was insignificant.
Financing Activities
For the ninethree months ended September 30, 2023, net cash provided by financing activities driven by the receipt of proceeds of $12.0 million from the G42 Promissory Note early redemption during the nine months ended September 30, 2023. For the nine months ended September 30, 2022,March 31, 2024, net cash provided by financing activities was driven by sales of our Class A common stock to a collaboration partner and proceeds from pre-funded warrants of $51.0 million from the CinRx Purchase Agreement.Private Placement financing. For the three months ended March 31, 2023, net cash provided by financing activities was driven by the receipt of proceeds of $12.0 million from the G42 promissory note early redemption.
Future Funding Requirements
To date, we have not generated any revenue from drug product sales. We do not know when, or if, we will generate any revenue from drug product sales. We do not expect to generate revenue from drug sales unless and until we obtain regulatory approval of and commercialize any of our drug candidates. At the same time, we expect our expenses to continue or to increase in connection with our ongoing development activities, particularly as we continue the research, development and clinical trials of, and seek regulatory approval for, our drug candidates. In addition, subject to obtaining regulatory approval of any of our drug candidates, we expect to incur significant commercialization expenses for product sales, marketing, manufacturing and distribution. We anticipate that we will need substantial additional funding in connection with our continuing operations.
Based on our current operating plan, we believe that our current cash and cash equivalents will allow us to meet our liquidity requirements intofor at least the first quarter of 2024.next twelve months. We plan to finance our operations into the first quarter of 2024 2026 through the use of our cash and cash equivalents and based on current operating plans, we are evaluating several financing strategies
to fund the ongoing and future clinical trials of cadisegliatin (TTP399), including direct equity investments and the potential licensing and monetization of other Company programs. The timing of any such transactions is not certain, and we may not be able to complete such transactions on acceptable terms, or at all. Even if we are able to complete such transactions, they may contain restrictions on our operations or cause substantial dilution to our stockholders. We have based our estimates on assumptions that may prove to be wrong, and we may use our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with the development and commercialization of our drug candidates, we are unable to estimate the amounts of increased capital outlays and operating expenditures necessary to complete the development of our drug candidates. Additionally, we may rely on our ability to sell shares of our Class A common stock pursuant to the ATM Offering and LPC Purchase Agreement.Offering. However, the ability to use these sourcesthis source of capital is dependent on a number of factors, including the prevailing market price of and the volume of trading in the Company’s Class A common stock.
Our future capital requirements will depend on many factors, including:
•the progress, costs, results and timing of our planned trials to evaluate cadisegliatin (TTP399) as a potential adjunctive therapy for the treatment of type 1 diabetes;
•the willingness of the FDA to rely upon our completed and planned clinical and preclinical studies and other work, as the basis for review and approval of our drug candidates;
•our ability to maintain control over our costs in line with our budget to complete the Phase 3 clinical trial for our lead product candidate, cadisegliatin;
•the outcome, costs and timing of seeking and obtaining FDA and any other regulatory approvals;
•the number and characteristics of drug candidates that we pursue, including our drug candidates in preclinical development;
•the ability of our drug candidates to progress through clinical development successfully;
•our need to expand our research and development activities;
•the costs associated with securing, establishing and maintaining commercialization capabilities;
•the costs of acquiring, licensing or investing in businesses, products, drug candidates and technologies;
•our ability to maintain, expand and defend the scope of our intellectual property portfolio, including the amount and timing of any payments we may be required to make, or that we may receive, in connection with the licensing, filing, prosecution, defense and enforcement of any patents or other intellectual property rights;
•our need and ability to hire additional management, scientific, and medical personnel;
•the effect of competing technological and market developments;
•our need to implement additional internal systems and infrastructure, including financial and reporting systems;
•the economic and other terms, timing and success of our existing licensing arrangements and any collaboration, licensing or other arrangements into which we may enter in the future; and
•the amount of any payments we are required to make to M&F TTP Holdings Two LLC in the future under the Tax Receivable Agreement.
Until such time, if ever, as we can generate substantial revenue from drug sales, we expect to finance our cash needs through a combination of equity offerings, debt financings, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. We currently have potential external source of funds available through the ATM Offering and LPC Purchase Agreement. However, the ability to use these sources of capital is dependent on a number of factors, including the prevailing market price of and the volume of trading in the Company’s Class A common stock.
To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interests of our common stockholders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our common stockholders. Debt financing and preferred equity financing, if available, may involve agreements that include covenants that will further limit or restrict our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may be required to relinquish valuable rights to our technologies, future revenue streams or drug candidates or grant licenses on terms that may not be favorable to us.
Our recurring losses, accumulated deficit and our current levels
Off-Balance Sheet Arrangements
As of cash and cash equivalents raise substantial doubt about our ability to continueMarch 31, 2024, we did not have outstanding any off-balance sheet arrangements as a going concern as of the date of this Quarterly Report on Form 10-Q. If we are unable to
continue as a going concern, we may have to liquidate our assets and it is likely that investors will lose all or a significant part of their investments. If we seek additional financing to fund our business activities in the future and there remains substantial doubt about our ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding to us on commercially reasonable terms or at all, and such additional funding may cause substantial dilution to our existing investors. If we are unable to obtain additional funding, we could be forced to delay, reduce or eliminate our research and development programs or commercialization efforts, or pursue one or more alternative strategies, such as restructuring, any of which could adversely affect our business prospects.defined under SEC rules.
Discussion of Critical Accounting Policies and Estimates
For a discussion of our critical accounting policies and estimates, please refer to Part II, Item 7. “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2022.2023. There have been no material changes to our critical accounting policies and estimates in 2023.2024.
Forward-Looking Statements
This quarterly report includes certain forward-looking statements within the meaning of the federal securities laws regarding, among other things, our management’s intentions, plans, beliefs, expectations, or predictions of future events, which are considered forward-looking statements. You should not place undue reliance on those statements because they are subject to numerous uncertainties and factors relating to our operations and business environment, all of which are difficult to predict and many of which are beyond our control. Forward-looking statements include information concerning our possible or assumed future results of operations, including descriptions of our business strategy. These statements often include words such as “anticipates,” “believes,” “could,” “seeks,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would” or similar expressions and the negatives of those terms. These statements are based upon assumptions that we have made in light of our experience in the industry, as well as our perceptions of historical trends, current conditions, expected future developments and other factors that we believe are appropriate under the circumstances. As you read this quarterly report, you should understand that these statements are not guarantees of performance or results. They involve known and unknown risks, uncertainties, and assumptions, including those described under the heading “Risk Factors” under Item 1A of Part I in our Annual Report on Form 10-K and under Item 1A of Part II of this Quarterly Report on Form 10-Q. Although we believe that these forward-looking statements are based upon reasonable assumptions, you should be aware that many factors, including those described under the heading “Risk Factors” under Item 1A of Part I in our Annual Report on Form 10-K and under Item 1A of Part II of this Quarterly Report on Form 10-Q, could affect our actual financial results or results of operations and could cause actual results to differ materially from those in the forward-looking statements.
Our forward-looking statements made herein are made only as of the date of this quarterly report. We expressly disclaim any intent, obligation or undertaking to update or revise any forward-looking statements made herein to reflect any change in our expectations with regard thereto or any change in events, conditions, or circumstances on which any such statements are based. All subsequent written and oral forward-looking statements attributable to us or persons acting on our behalf are expressly qualified in their entirety by the cautionary statements contained in this quarterly report.
Effect of Recent Accounting Pronouncements
See discussion of recent accounting pronouncements in Note 2, “Summary of Significant Accounting Policies”, to the Condensed Consolidated Financial Statements in this Form 10-Q.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Interest Rate Risk
We do not currently have any material interest rate exposure.
Market Risk
Our exposure to market risk is limited to our cash and cash equivalents, all of which have maturities of one year or less. The goals of our investment strategy are preservation of capital, fulfillment of liquidity needs and fiduciary control of cash and investments. We also seek to maximize income from our investments without assuming significant risk. To achieve our goals, we maintain a portfolio ofcash and cash equivalents in awith multiple financial institutioninstitutions that management believes to be of high credit quality. Because of the short-term maturities of our investments, we do not believe that an increase in market rates would have a material negative impact on the value of our investment portfolio.
Foreign Currency Risk
We do not have any material foreign currency exposure.
ITEM 4. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Under the supervision and with the participation of our Chief Executive Officer (our principal executive officer)Principal Executive Officer) and Chief Financial Officer (our principal financial officer)Principal Financial Officer), management has evaluated the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) or 15d-15(e) of the Securities Exchange Act of 1934) as of September 30, 2023.March 31, 2024. Based upon that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that, as of September 30, 2023,March 31, 2024, our disclosure controls and procedures were effective in causing material information relating to us (including our consolidated subsidiaries) to be recorded, processed, summarized, and reported by management on a timely basis and to ensure the quality and timeliness of our public disclosures pursuant to SEC disclosure obligations.
Our management, including our Chief Executive Officer and Chief Financial Officer, does not expect that our disclosure controls and procedures will prevent all errors and all fraud. A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, with the Company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple error and mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by management override of controls.
The design of any system of controls also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future
conditions. Over time, a control may become inadequate because of changes in conditions or because the degree of compliance with the policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and may not be detected.
Changes to Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting during our most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Website Availability of Reports and other Corporate Governance Information
The Company maintains a comprehensive corporate governance program, including Corporate Governance Guidelines for its Board of Directors, Board Guidelines for Assessing Director Independence, and charters for its Audit Committee, Nominating and Corporate Governance Committee and Compensation Committee. The Company maintains a corporate investor relations website, www.vtvtherapeutics.com, where stockholders and other interested persons may review, without charge, among other things, corporate governance materials and certain SEC filings, which are generally available on the same business day as the filing date with the SEC on the SEC’s website http://www.sec.gov. The contents of our website are not made a part of this Quarterly Report on Form 10-Q.
PART II – OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
We are not currently a party to any material legal proceedings.
ITEM 1A. RISK FACTORS
Our risk factors are set forth under the heading “Risk Factors” under Item 1A of Part I in our Annual Report on Form 10-K for the year ended December 31, 2022.2023. There have been no material changes to our risk factors from those previously disclosed in our Annual Report on Form 10-K for the year ended December 31, 2022.2023.
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
ThereOn February 27, 2024, we entered into a securities purchase agreement (the “Securities Purchase Agreement”) with certain institutional accredited investors (the “Private Placement Investors”), pursuant to which we agreed to issue and sell to the Private Placement Investors in a private placement (the “Private Placement”) (i) an aggregate of 464,377 shares (the “Private Placement Shares”) of our Class A common stock, at a purchase price of $11.81 per share, and (ii) pre-funded warrants (the “Private Placement Pre-Funded Warrants”) to purchase up to an aggregate of 3,853,997 shares of our Class A common stock (the “Private Placement Warrant Shares”) at a purchase price of $11.80 per Private Placement Pre-Funded Warrant (representing the $11.81 per Private Placement Share purchase price less the exercise price of $0.01 per Private Placement Warrant Share). We received aggregate gross proceeds from the Private Placement of approximately $51.0 million, before deducting offering expenses payable by us. The Private Placement Pre-Funded Warrants are exercisable at any time after their original issuance and will not expire.
On March 5, 2024, the Company entered into a letter agreement with the Private Placement Investors pursuant to which the Private Placement Investors agreed to exchange an aggregate of 116,493 Private Placement Shares for an aggregate of 116,590 Private Placement Pre-Funded Warrants.
The Private Placement Shares and the Private Placement Pre-Funded Warrants were no salesissued pursuant to the exemption from registration contained in Section 4(a)(2) of unregistered equity securities during the three months ended September 30, 2023.Securities Act.
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
None.
ITEM 4. MINE SAFETY DISCLOSURES
None.
ITEM 5. OTHER INFORMATION
Rule 10b5-1 Trading Plans
During the first fiscal quarter ended September 30, 2023,March 31, 2024, none of our directors or executive officers adopted or terminated any contract, instruction or written plan for the purchase or sale of our securities to satisfy the affirmative defense conditions of Rule 10b5-1(c) or any “non-Rule 10b5-1 trading arrangement.”
ITEM 6. EXHIBITS
| | | | | | | | |
Exhibit
Number | | Description |
| | |
| 31.1* | | |
| | |
| 31.2* | | |
| | |
| 32.1* | | |
| | |
| 32.2* | | |
| | |
| 101.INS | | Inline XBRL Instance Document |
| | |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema |
| | |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase |
| | |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Document |
| | |
| 101.LAB | | Inline XBRL Taxonomy Extension Label Linkbase |
| | |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase |
| | |
| 104 | | Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibit 101) |
___________________________
* Filed herewith
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
Date: NovemberMay 9, 20232024
| | | | | | | | |
| | VTV THERAPEUTICS INC. |
| | (Registrant) |
| | |
| By: | /s/ Paul J. Sekhri |
| | Paul J. Sekhri |
| | President and Chief Executive Officer |
| | |
| By: | /s/ Steven Tuch |
| | Steven Tuch |
| | Chief Financial Officer |