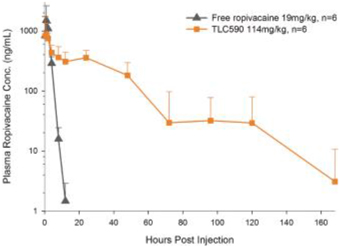
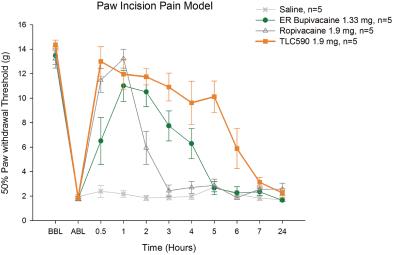
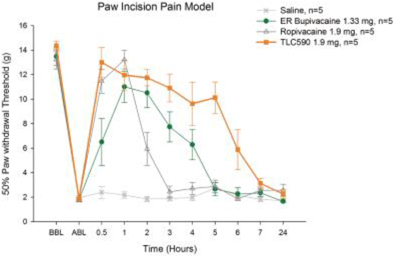
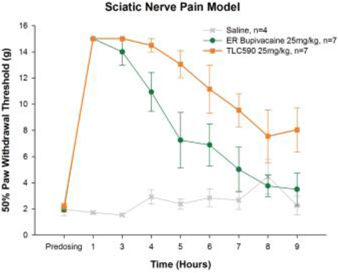
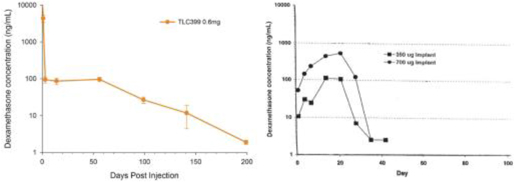
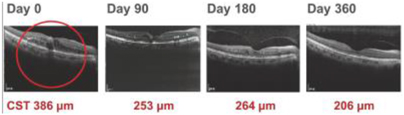
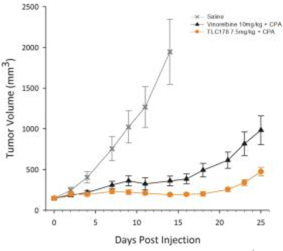
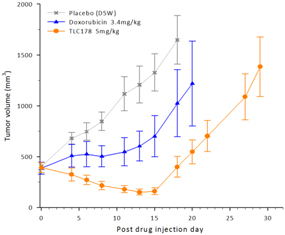
| (b) | The Group has elected to apply IFRS 16 by not restating the comparative information (referred herein as the “modified retrospective approach”) when applying IFRS 16 effective in 2019. Lease agreements classified as operating lease under IAS 17, except for short-term leases or leases oflow-value assets, are measured at the present value of the remaining lease payments, discounted using the lessee’s incremental borrowing interest rate as of January 1, 2019. Theright-of-use assets are measured at an amount equal to the lease liabilities adjusted by the amount of any prepaid or accrued lease payments. The incremental borrowing interest rate applied to the Group’s lease liabilities on January 1, 2019 ranged from 1.85% to 4.10%. In determining a lessee’s incremental borrowing interest rate used in discounting lease payments, a risk-free interest rate for relevant duration and the same currency is selected as a reference rate. The lessee’s credit spread adjustments and lease specific adjustments are also taken into account. For leases previously classified as finance leases under IAS 17, the Group recognized the carrying amount of the lease asset and lease liability immediately before transition as the carrying amount of theright-of-use asset and the lease liability at the date of initial application. (b) The cumulative effects of applying the new standards as of January 1, 2018 are summarized below:
| | Book value | | | Adjustment for | | | Adjusted amount | | | | under previous | | | initial application | | | after IFRS 15 | | | | revenue standard | | | of IFRS | | | adoption | | | | NT$000 | | | NT$000 | | | NT$000 | | Affected items | | | | | | | | | | | | | January 1, 2018 | | | | | | | | | | | | | Contract liabilities | | $ | — | | | $ | 7,941 | | | $ | 7,941 | | Total affected liabilities | | | — | | | | 7,941 | | | | 7,941 | | Accumulated deficit | | | (874,086 | ) | | | (7,941 | ) | | | (882,027 | ) | Total affected equity | | | (874,086 | ) | | | (7,941 | ) | | | (882,027 | ) | Total affected liabilities and equity | | ($ | 874,086 | ) | | $ | — | | | ($ | 874,086 | ) |
| | Book value | | | Adjustment for | | | Adjusted amount | | | | under previous | | | initial application | | | after IFRS 15 | | | | revenue standard | | | of IFRS | | | adoption | | | | US$000 | | | US$000 | | | US$000 | | Affected items | | | | | | | | | | | | | January 1, 2018 | | | | | | | | | | | | | Contract liabilities | | $ | — | | | $ | 259 | | | $ | 259 | | Total affected liabilities | | | — | | | | 259 | | | | 259 | | Accumulated deficit | | | (28,556 | ) | | | (259 | ) | | | (28,815 | ) | Total affected equity | | | (28,556 | ) | | | (259 | ) | | | (28,815 | ) | Total affected liabilities and equity | | ($ | 28,556 | ) | | $ | — | | | ($ | 28,556 | ) |
F-9
Table of Content
The Group’s authorization collaboration and development transactions generally authorizes intellectual property rights of the drug products to pharmaceutical companies. Though the Group will continuously provide research and development services on the drug products, pharmaceutical companies could make use of the research and development outcome at any time. Pharmaceutical companies pay a non-refundable up-front payment upon the signing of the contracts, and make milestone payments upon each milestone achieved. Pharmaceutical companies would have difficulty finding another service provider who offers the same services in terms of continuing research and development on the authorized drug products. Under the previous accounting policy, the Group recognized milestone payment revenue upon each milestone achieved, without distinguishing between authorization and continuing research and development services. After adopting IFRS 15, due to the authorization and follow-up milestones of research and development not meeting the criteria of distinction, the Group concluded that it will account for all the authorization and continuing research and development services as a single performance obligation and recognizes the transaction price according to the progress of performance obligations satisfied.
(c) Please refer to Note 12(5) for other disclosures in relation to the first adoption of IFRS 15.
| (2)
| New standards and interpretations not yet adopted
|
Amendments to IFRSs which have been published but are not mandatory for the financial period ended December 31, 2018 are listed below:
IFRS 16 “Leases” is effective from January 1, 2019, with early application permitted. The new standard requires
| (c) | The Group has used the following practical expedients permitted by the standard at the date of initial application of IFRS 16: |
| i. | Elected not to reassess whether a contract is, or contains, a lease, instead, for contracts entered into before the transition date the Group relied on its assessment made applying IAS 17 and IFRIC 4. |
| ii. | The use of a single discount rate to a portfolio of leases with reasonably similar characteristics. |
| iii. | The exclusion of initial direct costs for the measurement of‘right-of-use asset’. |
| (d) | The reconciliation between operating lease commitments for the remaining lease payments under IAS 17 and lease liabilities recognized as of January 1, 2019 measured at the present value of the remaining lease payments, discounted using the lessee’s incremental borrowing rate is as follows: |
| | | | | | | | | | | | NT$000 | | | US$000 | | Operating lease commitments disclosed by applying IAS 17 as at December 31, 2018 | | $ | 78,102 | | | $ | 2,611 | | Add: Lease payable recognized under finance lease by applying IAS 17 as of December 31, 2018 | | | 48,781 | | | | 1,631 | | Less: Short-term andlow-value leases | | | (1,624) | | | | (54) | | | | | | | | | | | Total lease contracts amount recognized as lease liabilities by applying IFRS 16 on January 1, 2019 | | $ | 125,259 | | | $ | 4,188 | | | | | | | | | | | Incremental borrowing interest rate at the date of initial application | | | 1.85%~4.10 | % | | | 1.85%~4.10 | % | Lease liabilities recognized as at January 1, 2019 by applying IFRS 16 | | $ | 121,021 | | | $ | 4,046 | | | | | | | | | | | Of which are: | | | | | | | | | Current lease liabilities | | | 54,718 | | | | 1,829 | | Non-current lease liabilities | | | 66,303 | | | | 2,217 | | | | | | | | | | | | | | 121,021 | | | | 4,046 | | | | | | | | | | |
| (e) | The impact on assets and liabilities as of January 1, 2019 from the initial application of IFRS 16 is set out as follows: |
| | | | | | | | | | | | | | | | Carrying amount
as of December
31, 2018 | | | Adjustments
arising from
initial
application | | | Adjusted
carrying amount
as of January 1,
2019 | | | | | NT$000 | | | NT$000 | | | NT$000 | | Property, plant and equipment | | $ | 158,245 | | | ($ | 46,970 | ) | | $ | 111,275 | | Right-of-use assets | | | — | | | | 119,991 | | | | 119,991 | | | | | | | | | | | | | | | Total effect on assets | | | | | | $ | 73,021 | | | | | | | | | | | | | | | | | | | Total effect on assets (US$000) | | | | | | $ | 2,441 | | | | | | | | | | | | | | | | | | | Finance lease liabilities | | $ | 48,000 | | | ($ | 48,000 | ) | | $ | — | | Lease liabilities | | | — | | | | 121,021 | | | | 121,021 | | | | | | | | | | | | | | | Total effect on liabilities | | | | | | $ | 73,021 | | | | | | | | | | | | | | | | | | | Total effect on liabilities (US$000) | | | | | | $ | 2,441 | | | | | | | | | | | | | | | | | | |
In addition, operating leases to be accounted for through the recognition of a “right of use asset” and a corresponding lease liability. Interest-bearing borrowings and non-current assets will increase on implementation of this standard. Operating lease costs will no longer be classified within the income statement based on amounts paid, but via a “right of use asset” depreciation charge recognized within operating profit and a lease interest expense within finance costs, subject to the exemptions on amount and duration. As at the reporting date, the Group has non-cancellable operating lease commitments of NT$78,102 thousand (US$2,551 thousand), see Note 9(2). As of January 1, 2019, the Group expects to increase both “right-of-use asset” and “lease liability” by approximately NT$73,021 thousand (US$2,386 thousand) and no significant impact to retained earnings. Operating cash flows will increase and financing cash flows decrease by approximately NT$32,09965,455 thousand (US$1,0492,188 thousand) asgiven that repayment of the principal portion of thenon-finance lease liabilities will be classified as cash flows from financing activities, where previously it was classified as operating cash outflows.
F-10
| (2) | New standards and interpretations not yet adopted |
Table of ContentAmendments to IFRSs which have been published but are not mandatory for the financial period ended December 31, 2019 are listed below:
| | | | | | New Standards, Interpretations and Amendments | | Effective Date by International Accounting Standards Board (“IASB”) | | | | Amendments to IAS 1 and IAS 8, ‘Disclosure Initiative-Definition of Material’ | | January 1, 2020 | | | | Amendments to IFRS 3, ‘Definition Of A Business’ | | January 1, 2020 | | | Amendments to IFRS 9, IAS 39 and IFRS 7, ‘Interest Rate Benchmark Reform’ | | January 1, 2020 | | | | Amendments to IFRS 10 and IAS 28, ‘Sale Or Contribution Of Assets Between An Investor And Its Associate Or Joint Venture’ | | To be determined by IASB | | | | IFRS 17, ‘Insurance Contracts’ | | January 1, 2021 |
In addition, the following newThe above standards and amended standardsinterpretations have been issued but not effective, which are not expected to have ano significant impact onto the Group’s financial condition and financial performance based on the Group’s assessment.
New Standards, Interpretations and Amendments
4. | | Effective Date by
International
Accounting
Standards Board
(“IASB”)
| Amendments to IFRS 9, ‘Prepayment Features with Negative
Compensation and Modifications of Financial Liabilities’
| | January 1, 2019
| Amendments to IFRS 10 and IAS 28, ‘Sale or contribution of
assets between an investor and its associate or joint venture’
| | To be determined
by IASB
| IFRS 17, ‘Insurance Contracts’
| | January 1, 2021
| Amendments to IAS 19, ‘Plan Amendment, Curtailment or
Settlement’
| | January 1, 2019
| Amendments to IAS 28, ‘Long-term Interests in Associates and
Joint Ventures’
| | January 1, 2019
| IFRIC 23, ‘Uncertainty over Income Tax Treatments’
| | January 1, 2019
| Annual improvements 2015–2017 cycle
| | January 1, 2019
| Amendments to IFRS 3, ‘Definition of a business’
| | January 1, 2020
| Amendment to IAS 1 and IAS 8, ‘Disclosure Initiative-Definition
of Material’
| | January 1, 2020
|
4.
| SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES |
The principal accounting policies applied in the preparation of these consolidated financial statements are set out below. These policies have been consistently applied to all the periods presented, unless otherwise stated. | (1) | (1)
| Basis of preparation |
These consolidated financial statements have been prepared in accordance with International Financial Reporting Standards (“IFRSs”), which collective term includes all applicable individual IFRSs, International Accounting Standards (“IASs”) issued by the International Accounting Standards Board (“IASB”)IASB and Interpretations issued by the International Financial Reporting Interpretations Committee (“IFRIC”) of the IASB. | B. | Historical cost convention |
Except for defined benefit liabilities recognized based on the net amount of pension fund assets less present value of defined benefit obligation, these consolidated financial statements have been prepared under the historical cost convention. The preparation requires the use of certain critical accounting estimates and also requires management to exercise its judgement in the process of applying the accounting policies. The areas involving a higher degree of judgement or complexity, or areas where assumptions and estimates are significant to the consolidated financial statements are disclosed in Note 5. | (2) | C.
| In adopting IFRS 9 and IFRS 15 effective January 1, 2018, the Group elected to apply the modified retrospective approach whereby the cumulative impact of the adoption was recognized as retained earnings or other equity as of January 1, 2018 and the financial statements for each of the two years in the period ended December 31, 2017 were not restated. The financial statements for the years ended December 31, 2016 and 2017 were prepared in compliance with International Accounting Standard 39 (‘IAS 39’), International Accounting Standard 11 (‘IAS 11’), International Accounting Standard 18 (‘IAS 18’) and related financial reporting interpretations. Please refer to Notes 12(4) and (5) for details of significant accounting policies.
|
F-11
Table of Content
| (2)
| Basis of consolidation |
| A. | Basis for preparation of consolidated financial statements: |
| (a) | All subsidiaries are included in the Group’s consolidated financial statements. Subsidiaries are all entities (including structured entities) controlled by the Group. The Group controls an entity when the Group is exposed, or has rights, to variable returns from its involvement with the entity and has the ability to affect those returns through its power over the entity. Consolidation of subsidiaries begins from the date the Group obtains control of the subsidiaries and ceases when the Group loses control of the subsidiaries. |
| (b) | Inter-company transactions, balances and unrealized gains or losses on transactions between companies within the Group are eliminated. Accounting policies of subsidiaries have been adjusted where necessary to ensure consistency with the policies adopted by the Group. |
| B. | Subsidiaries included in the consolidated financial statements: |
| | | | | | | Ownership (%) | | | Ownership (%) | | Name of Investor | | Name of Subsidiary | | Main Business Activities | | December 31, 2017 | | | December 31, 2018 | | | Name of Subsidiary | | Main Business Activities | | December 31,
2018 | | December 31,
2019 | | Taiwan Liposome Company, Ltd. | | TLC Biopharmaceuticals, Inc. | | Research on new anti-cancer drugs and biotechnology services | | | 100 | | | | 100 | | | TLC Biopharmaceuticals, Inc. | | Research on new anti-cancer drugs and biotechnology services | | 100 | | | 100 | | Taiwan Liposome Company, Ltd. | | TLC Biopharmaceuticals B.V. | | Technical authorization and product development | | | 100 | | | | 100 | | | TLC Biopharmaceuticals B.V. | | Technical authorization and product development | | 100 | | | 100 | | Taiwan Liposome Company, Ltd. | | TLC Biopharmaceuticals, (H.K.) Limited | | Biotechnology services and reinvestment | | | 100 | | | | 100 | | | TLC Biopharmaceuticals, (H.K.) Limited | | Biotechnology services and reinvestment | | 100 | | | 100 | | Taiwan Liposome Company, Ltd. | | TLC Biopharmaceuticals Pty Ltd. | | Technical authorization and product development | | | 100 | | | | 100 | | | TLC Biopharmaceuticals Pty Ltd. | | Technical authorization and product development | | 100 | | | 100 | | Taiwan Liposome Company, Ltd. | | TLC Biopharmaceuticals, Japan Co., Ltd. | | Technical authorization and product development | | | 100 | | | | 100 | | | TLC Biopharmaceuticals, Japan Co., Ltd. | | Technical authorization and product development | | 100 | | | 100 | | TLC Biopharmaceuticals, (H.K.) Limited | | TLC Biopharmaceuticals, (Shanghai) Limited | | Consulting and technical service of medication | | | 100 | | | | 100 | | | TLC Biopharmaceuticals, (Shanghai) Limited | | Consulting and technical service of medication | | 100 | | | 100 | |
| C. | Subsidiaries not included in the consolidated financial statements: None. |
| D. | Adjustments for subsidiaries with different balance sheet dates: None. |
| E. | Significant restrictions: None. |
| F. | Subsidiaries that havenon-controlling interests that are material to the Group: None. |
F-12
Table of Content
| (3) | (3)
| Foreign currency translation |
Items included in the financial statements of each of the Group’s entities are measured using the currency of the primary economic environment in which the entity operates (the “functional currency”). The consolidated financial statements are presented in New Taiwan dollars, which is the Company’s functional and the Group’s presentation currency. | A. | Foreign currency transactions and balances |
| (a) | Foreign currency transactions are translated into the functional currency using the exchange rates prevailing at the dates of the transactions or valuation where items are remeasured. Foreign exchange gains and losses resulting from the settlement of such transactions are recognized in profit or loss in the period in which they arise. |
| (b) | Monetary assets and liabilities denominated in foreign currencies at the period end are re-translated at the exchange rates prevailing at the balance sheet date. Exchange differences arising uponre-translation at the balance sheet date are recognized in profit or loss. |
| (c) | All other foreign exchange gains and losses are presented in the statement of comprehensive income within other gains and losses. |
| B. | Translation of foreign operations |
The operating results and financial position of all the group entities that have a functional currency different from the presentation currency are translated into the presentation currency as follows: | (a) | Assets and liabilities for each balance sheet presented are translated at the closing exchange rate at the date of that balance sheet; |
| (b) | Income and expenses for each statement of comprehensive income are translated at average exchange rates of that period; and |
| (c) | All resulting exchange differences are recognized in other comprehensive income. |
| (4) | (4)
| Classification of current andnon-current items |
| A. | Assets that meet one of the following criteria are classified as current assets: |
| (a) | Assets arising from operating activities that are expected to be realized, or are intended to be sold or consumed within the normal operating cycle; |
| (b) | Assets held mainly for trading purposes; |
| (c) | Assets that are expected to be realized within twelve months from the balance sheet date; |
| (d) | Cash and cash equivalents, excluding restricted cash and cash equivalents and those that are to be exchanged or used to pay off liabilities more than twelve months after the balance sheet date. |
| B. | Liabilities that meet one of the following criteria are classified as current liabilities: |
| (a) | Liabilities that are expected to be paid off within the normal operating cycle; |
| (b) | Liabilities arising mainly from trading activities; |
| (c) | Liabilities that are to be paid off within twelve months from the balance sheet date; |
| (d) | Liabilities for which the repayment date cannot be extended unconditionally to more than twelve months after the balance sheet date. Terms of a liability that could, at the option of the counterparty, result in its settlement by the issue of equity instruments do not affect its classification. |
Assets and liabilities that are not classified as current are non-current assets and liabilities, respectively. Cash equivalents refer to short-term, highly liquid investments that are readily convertible to known amounts of cash, and are subject to an insignificant risk of changes in value, and have a short maturity of generally within three months when acquired..acquired. Time deposits that meet the definition above and are held for the purpose of meeting short-term cash commitments in operations are classified as cash equivalents. F-13
Table of Content
| (6) | (6)
| Accounts receivable |
| A. | In accordance with contracts, accounts receivable entitle the Group a legal right to receive consideration in exchange for transferred goods or rendered services. |
| B. | The short-term accounts receivable not bearing interest are measured at initial invoice amount as the effect of discounting is immaterial. |
| (7) | (7)
| Impairment of financial assets |
For financial assets at amortized cost including accounts receivable or contract assets that have a significant financing component, at each reporting date, the Group recognizes the impairment provision for 12 months expected credit losses if there has not been a significant increase in credit risk since initial recognition or recognizes the impairment provision for the lifetime expected credit losses (“ECLs”) if such credit risk has increased since initial recognition after taking into consideration all reasonable and verifiable information that includes forecasts. On the other hand, for accounts receivable or contract assets that do not contain a significant financing component, the Group recognizes the impairment provision for lifetime ECLs. | (8) | (8)
| Derecognition of financial assets |
The Group derecognizes a financial asset when one of the following conditions is met: | A. | The contractual rights to receive the cash flows from the financial asset expire. |
| B. | The contractual rights to receive cash flows of the financial asset have been transferred and the Group has transferred substantially all risks and rewards of ownership of the financial asset. |
| C. | The contractual rights to receive cash flows of the financial asset have been transferred and the Group has not retained control of the financial asset. |
| (9) | (9)
| Property, plant and equipment |
| A. | Property, plant and equipment are initially recorded at cost. Borrowing costs incurred during the construction period are capitalized. |
| B. | Subsequent costs are included in the asset’s carrying amount or recognized as a separate asset, as appropriate, only when it is probable that future economic benefits associated with the item will flow to the Group and the cost of the item can be reliably measured. The carrying amount of the replaced component is derecognized. All other repairs and maintenance are charged to profit or loss as incurred. |
| C. | Land is not depreciated. The cost model is applied to other property, plant and equipment which is depreciated on a straight-line basis. Each component of an item of property, plant, and equipment with a cost that is significant in relation to the total cost of the item is depreciated separately. |
| D. | The assets’ residual values, useful lives and depreciation methods are reviewed, and adjusted if appropriate, at each financialyear-end or earlier if events and circumstances warrant. If expectations for the assets’ residual values and useful lives differ from previous estimates or the patterns of consumption of the assets’ future economic benefits embodied in the assets have changed significantly, any change is accounted for as a change in estimate under IAS 8, ‘Accounting Policies, Changes in Accounting Estimates and Errors’, from the date of the change. The estimated useful lives of property, plant and equipment are as follows: |
The estimated useful lives of property, plant and equipment are as follows: | | | | | Buildings | | | 44 years | | Testing equipment | | | 3 years ~ 8 years | | Office equipment | | | 3 years ~ 5 years | | Transportation equipment | | | 3 years | | Leasehold assets | | | 5 years ~ 10 years | | Leasehold improvements | | | 1 years ~ 5 years | |
| (10) | Leasing agreement(lessee)-right-of-use assets/ lease liabilities |
Effective beginning from 2019 | A. | Leases are recognized as aright-of-use asset and a corresponding lease liability at the date at which the leased asset is available for use by the Group. For short-term leases or leases oflow-value assets, lease payments are recognized as an expense on a straight-line basis over the lease term. |
| B. | Lease liabilities include the net present value of the remaining lease payments at the commencement date, discounted using the incremental borrowing interest rate. Lease payments are comprised of fixed payments, less any lease incentives receivable. The Group subsequently measures the lease liability at amortized cost using the interest method and recognizes interest expense over the lease term. |
At the commencement date, the Group assesses whether the lessee is reasonably certain to exercise an option to extend the lease or to purchase the underlying asset, or not to exercise an option to terminate the lease. The Group considers all relevant facts and circumstances that create an economic incentive for the lessee to exercise, or not to exercise, the option, including any expected changes in facts and circumstances from the commencement date until the exercise date of the option. Main factors considered include contractual terms and conditions for the optional periods, the importance of the underlying asset to the lessee’s operations, etc. The lease term is reassessed if a significant change in circumstances that are within the control of the Group occurs. The lease liability is remeasured and the amount of remeasurement is recognized as an adjustment to theright-of-use asset when there are changes in the lease term or lease payments and such changes do not arise from contract modifications. | C. | At the commencement date, theright-of-use asset is stated at cost comprising the following: |
| (a) | The amount of the initial measurement of lease liability; and |
| (b) | Any lease payments made at or before the commencement date. |
Theright-of-use asset is measured subsequently using the cost model and is depreciated from the commencement date to the earlier of the end of the asset’s useful life or the end of the lease term. When the lease liability is remeasured, the amount of remeasurement is recognized as an adjustment to theright-of-use asset. | (11) | (10)
| Leased assets/ lessee |
Prior to 2019 | A. | Based on the terms of a lease contract, a lease is classified as a finance lease if the Group assumes substantially all the risks and rewards incidental to ownership of the leased asset. |
F-14
Table of Content
| (a) | (a)
| A finance lease is recognized as an asset and a liability at the lease’s commencement at the lower of the fair value of the leased asset or the present value of the minimum lease payments. |
| (b) | The minimum lease payments are apportioned between the finance charges and the reduction of the outstanding liability. The finance charges are allocated to each period over the lease term so as to produce a constant periodic rate of interest on the remaining balance of the liability. |
| (c) | Property, plant and equipment held under finance leases are depreciated over their estimated useful lives. If there is no reasonable certainty that the Group will obtain ownership at the end of the lease, the asset shall be depreciated over the shorter of the lease term and its useful life. |
| B. | Payments made under an operating lease (net of any incentives received from the lessor) are recognized in profit or loss on a straight-line basis over the lease term. |
| (12) | (11)
| Intangible assets |
| A. | Professional technology, mainly patents and technology knowledge, which the Group acquired from third parties, is stated at cost and amortized on a straight-line basis over 10 years. |
| B. | Computer software is stated at cost and amortized on a straight-line basis over its contract terms of 1 to 4 years. |
| (13) | (12)
| Impairment ofnon-financial assets |
The Group assesses at each balance sheet date the recoverable amounts of those assets where there is an indication of impairment. An impairment loss is recognized for the amount by which the asset’s carrying amount exceeds its recoverable amount. The recoverable amount is the higher of an asset’s fair value less costs to sell or value in use. When the circumstances or reasons for recognizing impairment loss for an asset in prior years no longer exist or diminish, the impairment loss is reversed. The increased carrying amount due to reversal should not be more than what the depreciated or amortized historical cost would have been if the impairment had not been recognized. Borrowings are recognized initially at fair value, net of transaction costs incurred. Borrowings are subsequently stated at amortized cost; any difference between the proceeds (net of transaction costs) and the redemption value is recognized in profit or loss over the period of the borrowings using the effective interest method. | (15) | (14)
| Derecognition of financial liabilities |
A financial liability (or a part of a financial liability) is derecognized when the obligation specified in the contract is either discharged or cancelled or expires. Decommissioning provisions are recognized when the Group has a present legal or constructive obligation as a result of past events, and it is probable that an outflow of economic resources will be required to settle the obligation and the amount of the obligation can be reliably estimated. Provisions are measured at the present value of the expenditures expected to be required to settle the obligation on the balance sheet date, which is discounted using apre-tax discount rate that reflects the current market assessments of the time value of money and the risks specific to the obligation. When discounting is used, the increase in the provision due to passage of time is recognized as interest expense. Provisions are not recognized for future operating losses. | (17) | (16)
| Employee benefits |
| A. | Short-term employee benefits |
Short-term employee benefits are measured at the undiscounted amount of the benefits expected to be paid in respect of service rendered by employees in a period and should be recognized as expenses in that period when the employees render service. F-15
Table of Content
| (a) | Defined contribution plans |
For defined contribution plans, the contributions are recognized as pension expensenexpenses on an accrual basis. Prepaid contributions are recognized as an asset to the extent of a cash refund or a reduction in the future payments. | i. | Net obligation under a defined benefit plan is defined as the present value of an amount of pension benefits that employees will receive on retirement for their services with the Group in current period or prior periods. The liability recognized is the present value of the defined benefit obligation at the balance sheet date less the fair value of plan assets. The net defined benefit net obligation is calculated annually by independent actuaries using the projected unit credit method. The rate used to discount is determined by using interest rates of government bonds (at the balance sheet date) of a currency and term consistent with those of the employment benefit obligations. |
| ii. | Remeasurements arising on defined benefit plans are recognized in other comprehensive income in the period in which they arise and are recorded as retained earnings. |
| (18) | (17)
| Employee share-based payment |
| A. | A. (a)
| For the equity-settled share-based payment arrangements, the employee services received are measured at the fair value of the equity instruments granted at the grant date, and are recognized as compensation cost over the vesting period, with a corresponding adjustment to equity. The fair value of the equity instruments granted shall reflect the impact of market vesting conditions. Compensation cost is subject to adjustment based on the service conditions that are expected to be satisfied and the estimates of the number of equity instruments that are expected to vest at each balance sheet date. Ultimately, the amount of compensation cost recognized is based on the number of equity instruments that eventually vest. |
| (b) | Whenever share options expire, the previous compensation costs recognized in “Capital surplus – Share options” are reclassified as “Capital surplus – Additionalpaid-in capital”. |
| (a) | Restricted stocks issued to employees are measured at the fair value of the equity instruments granted at the grant date subtracting the subscription price of NT$10 (in dollars), and are recognized as compensation cost over the vesting period. The Group has set the date when employees signed the agreement as the grant date of restricted stocks. |
| (b) | For restricted stocks where those stocks do not restrict distribution of dividends to employees and employees are not required to return the dividends received if they resign during the vesting period, the Group recognizes the fair value of the dividends received by the employees who are expected to resign during the vesting period as compensation cost at the date of dividend declaration. |
| (c) | For restricted stocks where employees have to pay to acquire those stocks, if employees resign during the vesting period, they must return the stocks to the Group and the Group must refund their payments on the stocks based on the original subscription price, the Group recognizes the payments from the employees who are expected to resign during the vesting period as liabilities at the grant date, and recognizes the payments from the employees who are expected to be eventually vested with the stocks in “Capital surplus – Restricted stocks”. |
| A. | The tax expense for the period comprises current and deferred tax. Tax is recognized in profit or loss, except to the extent that it relates to items recognized in other comprehensive income or items recognized directly in equity, in which cases the tax is recognized in other comprehensive income or equity, respectively. |
F-16
Table of Content
| B. | B.
| The current income tax expense is calculated on the basis of the tax laws enacted or substantively enacted at the balance sheet date in the countries where the Company and its subsidiaries operate and generate taxable income. Management periodically evaluates positions taken in tax returns with respect to situations in accordance with applicable tax regulations. It establishes provisions where appropriate based on the amounts expected to be paid to the tax authorities. |
| C. | Deferred tax is recognized, using the balance sheet liability method, on temporary differences arising between the tax bases of assets and liabilities and their carrying amounts in the consolidated balance sheet. However, the deferred tax is not accounted for if it arises from initial recognition of an asset or liability in a transaction other than a business combination that at the time of the transaction affects neither accounting nor taxable profit or loss. Deferred tax is provided on temporary differences arising on investments in subsidiaries, except where the timing of the reversal of the temporary difference is controlled by the Group and it is probable that the temporary difference will not reverse in the foreseeable future. Deferred tax is determined using tax rates (and laws) that have been enacted or substantially enacted by the balance sheet date and are expected to apply when the related deferred tax asset is realized or the deferred tax liability is settled. |
| D. | Deferred tax assets are recognized only to the extent that it is probable that future taxable profit will be available against which the temporary differences can be utilized. At each balance sheet date, unrecognized and recognized deferred tax assets are reassessed. |
| E. | Current income tax assets and liabilities are offset and the net amount reported in the balance sheet when there is a legally enforceable right to offset the recognized amounts and there is an intention to settle on a net basis or realize the asset and settle the liability simultaneously. Deferred tax assets and liabilities are offset on the balance sheet when the entity has the legally enforceable right to offset current tax assets against current tax liabilities and they are levied by the same taxation authority on either the same entity or different entities that intend to settle on a net basis or realize the asset and settle the liability simultaneously. |
| F. | A deferred tax asset shall be recognized for the carryforward of unused tax credits resulting from research and development expenditures and employees’ training costs to the extent that it is possible that future taxable profit will be available against which the unused tax credits can be utilized. |
| A. | Common shares are classified as equity. Incremental costs directly attributable to the issue of new shares or stock options are shown in equity as a deduction, net of tax, from the proceeds. |
| B. | Where the Company repurchases the Company’s equity share capital that has been issued, the consideration paid, including any directly attributable incremental costs (net of income taxes) is deducted from equity attributable to the Company’s equity holders. Where such shares are subsequently reissued, the difference between their book value and any consideration received, net of any directly attributable incremental transaction costs and the related income tax effects, is included in equity attributable to the Company’s equity holders. |
| (21) | (20)
| Revenue recognition |
The Group’s revenue is comprised ofup-front payments and milestone payments under authorization collaboration and development contracts, and sales-based royalty payment under royalty contracts. Authorization collaboration and development revenue The Group’s authorization collaboration and development transactions generally authorizes intellectual property rights of the drug products to pharmaceutical companies. Though the Group will continuously provide research and development services on the drug products, pharmaceutical companies could make use of the research and development outcome at any time. Pharmaceutical companies pay anon-refundableup-front payment upon signing of the contracts, and make milestone payments upon each milestone achieved. Based on the Group’s assessment, the Group uses its proprietary drug delivery technologies to continue the research and development related services, which are unique such that pharmaceutical companies would have difficulty finding another service provider who offers the same services in terms of continuing research and development on the authorized drug products. The authorization and subsequent research and development services provided by the Group are bonded and highly interrelated and therefore not distinct and as such are accounted for as one performance obligation to be delivered over time. At the inception of an agreement that includes research and development milestone payments, the Group evaluates each milestone to determine when and how much of the milestone to be included in the transaction price. The Group first estimates the amount of the milestone payment that the Group could receive using the most likely amount approach. The Group primarily uses the most likely amount approach as that approach is generally most predictive for milestone payments with a binary outcome. Then, the Group considers whether any portion of that estimated amount is subject to the variable consideration constraint (that is, whether it is probable that a significant reversal of cumulative revenue would not occur upon resolution of the uncertainty.) The Group updates the estimate of variable consideration included in the transaction price at each reporting date which includes updating the assessment of the likely amount of consideration and the application of the constraint to reflect current facts and circumstances. The revenue is recognized based on the transaction price, excluding variable considerations considered not achievable, and the stage of completion, which is measured by the proportion of contract costs incurred for research and development services as of the financial reporting date to the estimated total research and development costs for the authorization collaboration and development contracts. As the Group’s inputs, including costs of Contract Research Organizations, Contract Manufacture Organizations, and medicines, which have direct relationship with the transfer of control of services to customers, the Group uses the cost incurred method to measure progress towards complete satisfaction of a performance obligation. The Group evaluates the measure of progress each reporting period and, if necessary, adjust the measure of performance and related revenue recognition. The customer pays at the time specified in the payment schedule. If the services rendered exceed the payment, a contract asset is recognized. If the payments exceed the services rendered, a contract liability is recognized. A contract liability is recognized as revenue through the performance obligation is satisfied over time. If a license to the Group’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Group will recognize revenues when the license is transferred to the licensee and the licensee is able to use and benefit from the license. Given that the period between the transfer of promised services to customers and payment by customers exceed one year for authorization collaboration and development contracts, the transaction price is adjusted using the discount rate that would be reflected in a separate financing transaction between the Group and its customers at contract inception. The discount rate would reflect the credit characteristics of the Group receiving funding from financial institution. Royalty revenue The Group also entered into contracts with customers, where the Group is entitled to a sales-based royalty in exchange for a license of manufacturing and the right to sell pharmaceutical products. In accordance with the contracts, the Group will not undertake any activities that will significantly affect the intellectual property to which the customer has rights. The nature of the Group’s promise in granting a license is a promise to provide a right to use the Group’s intellectual property and therefore the revenue is recognized when transferring the license to a customer at a point in time. The Group recognizes revenue at the later of when the performance obligation has been satisfied and the subsequent sale occurs. | (22) | A.
| The Group’s authorization collaboration and development transactions generally authorizes intellectual property rights of the drug products to pharmaceutical companies. Though the Group will continuously provide research and development services on the drug products, pharmaceutical companies could make use of the research and development outcome at any time. Pharmaceutical companies pay a non-refundable up-front payment upon signing of the contracts, and make milestone payments upon each milestone achieved. Based on the Group’s assessment, the Group uses its proprietary drug delivery technologies to continue the research and development related services, which are unique such that pharmaceutical companies would have difficulty finding another service provider who offers the same services in terms of continuing research and development on the authorized drug products. The authorization and subsequent research and development services provided by the Group are bonded and highly interrelated and therefore not distinct and as such are accounted for as one performance obligation to be delivered over time. At the
|
F-17
Table of Content
| | inception of an agreement that includes research and development milestone payments, the Group evaluates each milestone to determine when and how much of the milestone to be included in the transaction price. The Group first estimates the amount of the milestone payment that the Group could receive using the most likely amount approach. The Group primarily uses the most likely amount approach as that approach is generally most predictive for milestone payments with a binary outcome. Then, the Group considers whether any portion of that estimated amount is subject to the variable consideration constraint (that is, whether it is probable that a significant reversal of cumulative revenue would not occur upon resolution of the uncertainty.) The Group updates the estimate of variable consideration included in the transaction price at each reporting date which includes updating the assessment of the likely amount of consideration and the application of the constraint to reflect current facts and circumstances. The revenue is recognized based on the transaction price, excluding variable considerations considered not achievable, and the stage of completion, which is measured by the proportion of contract costs incurred for research and development services as of the financial reporting date to the estimated total research and development costs for the authorization collaboration and development contracts. As the Group’s inputs, including costs of Contract Research Organizations, Contract Manufacture Organizations, and medicines, which have direct relationship with the transfer of control of services to customers, the Group uses the cost incurred method to measure progress towards complete satisfaction of a performance obligation. The Group evaluates the measure of progress each reporting period and, if necessary, adjust the measure of performance and related revenue recognition. The customer pays at the time specified in the payment schedule. If the services rendered exceed the payment, a contract asset is recognized. If the payments exceed the services rendered, a contract liability is recognized. A contract liability is recognized as revenue through the performance obligation is satisfied over time.
|
| B.
| The Group also entered into contracts with customers, where the Group is entitled to a sales-based royalty in exchange for a license of manufacturing and the right to sell pharmaceutical products. In accordance with the contracts, the Group will not undertake any activities that will significantly affect the intellectual property to which the customer has rights. The nature of the Group’s promise in granting a license is a promise to provide a right to use the Group’s intellectual property and therefore the revenue is recognized when transferring the license to a customer at a point in time. The Group recognizes revenue at the later of when the performance obligation has been satisfied and the subsequent sale occurs.
|
| C.
| Given that the period between the transfer of promised services to customers and payment by customers exceed one year for authorization collaboration and development contracts, the transaction price is adjusted using the discount rate that would be reflected in a separate financing transaction between the Group and its customers at contract inception. The discount rate would reflect the credit characteristics of the Group receiving funding from financial institution.
|
| (21)
| Research and development costs |
Research and development costs that do not meet the criteria of internally generated intangible assets of IAS 38 “Intangible Assets” are expensed in the period in which it is incurred. | (23) | (22)
| Government grants |
Government grants are recognized at their fair value only when there is reasonable assurance that the Group will comply with any conditions attached to the grants and the grants will be received. Government grants are recognized in profit or loss on a systematic basis over the periods in which the Group recognizes expenses for the related costs for which the grants are intended to compensate. | (24) | (23)
| Operating segments |
Operating segments are reported in a manner consistent with the internal reporting provided to the chief operating decision-maker. The Group’s chief operating decision-maker is responsible for allocating resources and assessing performance of the operating segments. F-18
Table of Content
| (25) | (24)
| Convenience translation into U.S. dollar amounts |
The Group maintains its accounts and expresses its consolidated financial statements in New Taiwan dollars. For convenience purposes, U.S. dollar amounts presented in the accompanying consolidated financial statements have been translated from New Taiwan dollars to U.S. dollars at the noon buying rate in the City of New York for cable transfers as certified for customs purposes by the Federal Reserve Bank of New York as of December 31, 2018,2019, which was NT$30.6129.91 to US$1.00. These convenience translations should not be construed as representations that the New Taiwan dollar amounts have been, or could in the future be, converted into U.S. dollars at this or any other rate of exchange. 5. | CRITICAL ACCOUNTING ESTIMATES AND KEY SOURCES OF ASSUMPTION UNCERTAINTY |
The preparation of these consolidated financial statements requires management to make critical judgements in applying the Group’s accounting policies and make critical assumptions and estimates concerning future events. Assumptions and estimates may differ from the actual results and are continually evaluated and adjusted based on historical experience and other factors. Such assumptions and estimates have a significant risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next financial year; and the related information is addressed below: | (1) | Critical judgements in applying the Group’s accounting policies: |
Identification of performance obligation The Group provides authorization collaboration and development services to its customers, which generally includes authorization of intellectual property rights of drug products to pharmaceutical companies together with continuing research and development services, which pharmaceutical companies can access and make use of the research outcome at any time. In determining the performance obligations under authorization collaboration and development contracts, the Group takes into account the guidance IFRS 15 par. 29: Factors that indicate that an entity’s promise to transfer a good or service to a customer is separately identifiable (in accordance with paragraph 27(b)) include, but are not limited to, the following: a.The
| a. | the entity does not provide a significant service of integrating the good or service with other goods or services promised in the contract into a bundle of goods or services that represent the combined output for which the customer has contracted. In other words, the entity is not using the good or service as an input to produce or deliver the combined output specified by the customer. |
| b. | the good or service does not significantly modify or customize another good or service promised in the contract. |
| c. | the good or service with other goods or services promised in the contract into a bundle of goods or services that represent the combined output for which the customer has contracted. In other words, the entity is not using the good or service as an input to produce or deliver the combined output specified by the customer. b.The good or service does not significantly modify or customize another good or service promised in the contract.
c.The good or service is not highly dependent on, or highly interrelated with, other goods or services promised in the contract. For example, the fact that a customer could decide to not purchase the good or service without significantly affecting the other promised goods or services in the contract might indicate that the good or service is not highly dependent on, or highly interrelated with, those other promised goods or services.
|
Based on the Group’s assessment, the Group uses its proprietary drug delivery technologies to continue the research and development related services, which are unique, and based on the intellectual property rights authorized and therefore, pharmaceutical companies would have difficulty finding alternative service provider in offering the same services. In addition, the authorization and subsequent research and development services provided by the Group are bonded and highly interrelated and therefore not distinct and are identified as one performance obligation to be delivered over time. If the Group assesses that the technologies or intellectual property rights authorized are well developed and the licensee is able to benefit from the license, the authorization and subsequent research and development services provided by the Group are not combined and therefore distinct. | (2) | Critical accounting estimates and assumptions:assumptions |
Revenue recognition The Group accounts for all the authorization and subsequent research and development services as one performance obligation and recognizes revenue based on the transaction price and the stage of completion, which is measured by the proportion of contract costs incurred for research and development services as of the financial reporting date to the estimated total research and development costs for the authorization collaboration and development contracts. The estimated total research and development costs would be affected by the progress of research and development, development of pharmaceutical products, collaboration with pharmaceutical companies, clinical trials, etc. The Group reassesses the reasonableness of estimates periodicallyperiodically. F-19
Table of Content
6. | DETAILS OF SIGNIFICANT ACCOUNTS |
| (1) | (1)
| Cash and cash equivalents |
| | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Cash on hand | | $ | 81 | | | $ | 56 | | | $ | 2 | | Checking and demand deposits | | | 547,232 | | | | 807,428 | | | | 26,378 | | Time deposits | | | 404,400 | | | | — | | | | — | | | | $ | 951,713 | | | $ | 807,484 | | | $ | 26,380 | |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Cash on hand | | $ | 56 | | | $ | 50 | | | $ | 2 | | Checking and demand deposits | | | 807,428 | | | | 978,854 | | | | 32,727 | | Time deposits | | | — | | | | 44,970 | | | | 1,503 | | | | | | | | | | | | | | | | | $ | 807,484 | | | $ | 1,023,874 | | | $ | 34,232 | | | | | | | | | | | | | | |
The Group transacts with a variety of financial institutions with good credit quality to disperse credit risk, so it expects that the probability of counterparty default is remote. | (2) | (2)
| Financial assets at amortized cost |
| | December 31, | | Items | | 2017 | | | 2018 | | Current items: | | NT$000 | | | NT$000 | | | US$000 | | Time deposit with maturity over three months | | $ | — | | | $ | 307,150 | | | $ | 10,034 | |
| | | | | | | | | | | | | | | | December 31, | | Items | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US $000 | | Current items: | | | | | | | | | | | | | Time deposits with maturity over three months | | $ | 307,150 | | | $ | — | | | $ | — | | | | | | | | | | | | | | |
Amounts recognized in profit or loss in relation to financial assets at amortized cost are listed below: | | December 31, | | | | | | | | Items | | 2017 | | | 2018 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | US$000 | | Interest income | | $ | — | | | $ | 380 | | | $ | 12 | | | $ | 380 | | | $ | 787 | | | $ | 26 | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Accounts receivable | | $ | 27,475 | | | $ | 33,252 | | | $ | 1,112 | | Less: Allowance for doubtful accounts | | | (18,132 | ) | | | (18,132 | ) | | | (606 | ) | | | | | | | | | | | | | | | | $ | 9,343 | | | $ | 15,120 | | | $ | 506 | | | | | | | | | | | | | | |
| | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Accounts receivable | | $ | 26,754 | | | $ | 27,475 | | | $ | 898 | | Less: Allowance for doubtful accounts | | | (18,132 | ) | | | (18,132 | ) | | | (593 | ) | | | $ | 8,622 | | | $ | 9,343 | | | $ | 305 | |
| A. | The Group does not hold any collateral as securitysecurity. |
| B. | The ageing analysis of accounts receivable is as follows: |
| | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Not past due | | $ | 8,622 | | | $ | 9,343 | | | $ | 305 | | Up to 30 days | | | — | | | | — | | | | — | | 31 to 90 days | | | — | | | | — | | | | — | | 91 to 180 days | | | — | | | | — | | | | — | | Over 181 days | | | 18,132 | | | | 18,132 | | | | 593 | | | | $ | 26,754 | | | $ | 27,475 | | | $ | 898 | |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Not past due | | $ | 9,343 | | | $ | 15,120 | | | $ | 506 | | Up to 30 days | | | — | | | | — | | | | — | | 31 to 90 days | | | — | | | | — | | | | — | | 91 to 180 days | | | — | | | | — | | | | — | | Over 181 days | | | 18,132 | | | | 18,132 | | | | 606 | | | | | | | | | | | | | | | | | $ | 27,475 | | | $ | 33,252 | | | $ | 1,112 | | | | | | | | | | | | | | |
The above ageing analysis was based on past due date. F-20
Table of Content
| C. | C.
| Information relating to credit risk of accounts receivable is provided in Note 12(2):. |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Net input VAT | | $ | 40,614 | | | $ | 34,591 | | | $ | 1,156 | | Prepaid insurance expense | | | 1,568 | | | | 3,190 | | | | 107 | | Prepaid handling charges | | | 1,294 | | | | 1,209 | | | | 40 | | Prepaid repair expense | | | 1,580 | | | | 975 | | | | 33 | | Others | | | 11,455 | | | | 11,019 | | | | 368 | | | | | | | | | | | | | | | | | $ | 56,511 | | | $ | 50,984 | | | $ | 1,704 | | | | | | | | | | | | | | |
| | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Net input VAT | | $ | 36,103 | | | $ | 40,614 | | | $ | 1,326 | | Prepaid repair expense | | | 7,042 | | | | 1,580 | | | | 52 | | Prepaid insurance expense | | | 1,068 | | | | 1,568 | | | | 51 | | Prepaid handling charges | | | 1,352 | | | | 1,294 | | | | 42 | | Prepaid rent | | | 301 | | | | 487 | | | | 16 | | Prepaid expense for medical research | | | 1,850 | | | | 3 | | | | 1 | | Prepaid service charges | | | 15,742 | | | | — | | | | — | | Others | | | 7,942 | | | | 10,965 | | | | 358 | | | | $ | 71,400 | | | $ | 56,511 | | | $ | 1,846 | |
| (5)
| Property, plant and equipment |
| A. | The details of property, plant and equipment are as follows: |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Land | | | Buildings | | | Testing
equipment | | | Office
equipment | | | Leasehold
assets | | | Leasehold
improvements | | | Total | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 40,708 | | | $ | 18,329 | | | $ | 98,170 | | | $ | 73,014 | | | $ | 274,715 | | Accumulated depreciation | | | — | | | | (5,360 | ) | | | (25,771 | ) | | | (12,777 | ) | | | (23,681 | ) | | | (53,791 | ) | | | (120,880 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Opening net book amount | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | | Additions | | | — | | | | — | | | | 34,802 | | | | 1,318 | | | | — | | | | 5,950 | | | | 42,070 | | Disposals | | | — | | | | — | | | | (9 | ) | | | — | | | | — | | | | — | | | | (9 | ) | Reclassification | | | — | | | | — | | | | 9,114 | | | | 1,847 | | | | (7,679 | ) | | | (1,847 | ) | | | 1,435 | | Transfers (Note 2) | | | — | | | | — | | | | 143 | | | | — | | | | — | | | | — | | | | 143 | | Depreciation charges | | | — | | | | (656 | ) | | | (6,143 | ) | | | (3,157 | ) | | | (19,840 | ) | | | (9,519 | ) | | | (39,315 | ) | Net exchange differences | | | — | | | | — | | | | 87 | | | | (4 | ) | | | — | | | | 3 | | | | 86 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | At December 31, 2018 | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 82,584 | | | $ | 19,878 | | | $ | 50,013 | | | $ | 77,208 | | | $ | 274,177 | | Accumulated depreciation | | | — | | | | (6,016 | ) | | | (29,653 | ) | | | (13,822 | ) | | | (3,043 | ) | | | (63,398 | ) | | | (115,932 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Land | | | Buildings | | | Testing
equipment | | | Office
equipment | | | Transportation
equipment | | | Leasehold
assets | | | Leasehold
improvements | | | Total | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2019 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 82,584 | | | $ | 19,878 | | | $ | — | | | $ | 50,013 | | | $ | 77,208 | | | $ | 274,177 | | Accumulated depreciation | | | — | | | | (6,016 | ) | | | (29,653 | ) | | | (13,822 | ) | | | — | | | | (3,043 | ) | | | (63,398 | ) | | | (115,932 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | �� | | | | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | — | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Opening net book amount | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | — | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | Additions | | | — | | | | — | | | | 4,379 | | | | 281 | | | | 120 | | | | — | | | | 486 | | | | 5,266 | | Disposals | | | — | | | | — | | | | (1,041 | ) | | | (28 | ) | | | — | | | | — | | | | (518 | ) | | | (1,587 | ) | Reclassification (Note 1) | | | — | | | | — | | | | (36,315 | ) | | | — | | | | — | | | | (46,970 | ) | | | — | | | | (83,285 | ) | Transfers (Note 2) | | | — | | | | — | | | | 71 | | | | — | | | | — | | | | — | | | | — | | | | 71 | | Depreciation charges | | | — | | | | (656 | ) | | | (4,139 | ) | | | (2,998 | ) | | | (23 | ) | | | — | | | | (9,049 | ) | | | (16,865 | ) | Net exchange differences | | | — | | | | — | | | | (40 | ) | | | (40 | ) | | | — | | | | — | | | | (82 | ) | | | (164 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 14,962 | | | $ | 22,860 | | | $ | 15,846 | | | $ | 3,271 | | | $ | 97 | | | $ | — | | | $ | 4,647 | | | $ | 61,683 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | At December 31, 2019 | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 40,307 | | | $ | 18,675 | | | $ | 120 | | | $ | — | | | $ | 63,767 | | | $ | 167,363 | | Accumulated depreciation | | | — | | | | (6,672 | ) | | | (24,461 | ) | | | (15,404 | ) | | | (23 | ) | | | — | | | | (59,120 | ) | | | (105,680 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 14,962 | | | $ | 22,860 | | | $ | 15,846 | | | $ | 3,271 | | | $ | 97 | | | $ | — | | | $ | 4,647 | | | $ | 61,683 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | At December 31, 2019(US$000) | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 500 | | | $ | 987 | | | $ | 1,348 | | | $ | 624 | | | $ | 4 | | | $ | — | | | $ | 2,132 | | | $ | 5,595 | | Accumulated depreciation | | | — | | | | (223 | ) | | | (818 | ) | | | (515 | ) | | | (1 | ) | | | — | | | | (1,976 | ) | | | (3,533 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 500 | | | $ | 764 | | | $ | 530 | | | $ | 109 | | | $ | 3 | | | $ | — | | | $ | 156 | | | $ | 2,062 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Land | | | Buildings | | | Testing equipment | | | Office equipment | | | Leasehold assets | | | Leasehold improvements | | | Total | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2017 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 66,266 | | | $ | 16,235 | | | $ | 100,070 | | | $ | 72,504 | | | $ | 299,569 | | Accumulated depreciation | | | — | | | | (4,703 | ) | | | (34,843 | ) | | | (9,458 | ) | | | (30,239 | ) | | | (41,383 | ) | | | (120,626 | ) | | | $ | 14,962 | | | $ | 24,829 | | | $ | 31,423 | | | $ | 6,777 | | | $ | 69,831 | | | $ | 31,121 | | | $ | 178,943 | | 2017 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Opening net book amount | | $ | 14,962 | | | $ | 24,829 | | | $ | 31,423 | | | $ | 6,777 | | | $ | 69,831 | | | $ | 31,121 | | | $ | 178,943 | | Additions | | | — | | | | — | | | | 12,069 | | | | 2,840 | | | | — | | | | 1,072 | | | | 15,981 | | Disposals | | | — | | | | — | | | | — | | | | (4 | ) | | | — | | | | — | | | | (4 | ) | Reclassifications | | | — | | | | — | | | | (25,390 | ) | | | — | | | | 25,642 | | | | (252 | ) | | | — | | Transfer (Note) | | | — | | | | — | | | | 1,256 | | | | — | | | | — | | | | — | | | | 1,256 | | Depreciation charges | | | — | | | | (657 | ) | | | (4,045 | ) | | | (3,581 | ) | | | (20,983 | ) | | | (12,660 | ) | | | (41,926 | ) | Net exchange differences | | | — | | | | — | | | | (376 | ) | | | 20 | | | | (1 | ) | | | (58 | ) | | | (415 | ) | | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | | At December 31, 2017 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 40,708 | | | $ | 18,329 | | | $ | 98,170 | | | $ | 73,014 | | | $ | 274,715 | | Accumulated depreciation | | | — | | | | (5,360 | ) | | | (25,771 | ) | | | (12,277 | ) | | | (23,681 | ) | | | (53,791 | ) | | | (120,880 | ) | | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | |
| Note 1: | Information about the reclassification from leasehold assets toright-of-use assets for the initial application of IFRS 16 at January 1, 2019 is provided in Note 6(6). |
F-21
Table of Content
| Note 2: | Transferred from prepayments for equipment (shown as “Othernon-current assets”). |
| | Land | | | Buildings | | | Testing equipment | | | Office equipment | | | Leasehold assets | | | Leasehold improvements | | | Total | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 40,708 | | | $ | 18,329 | | | $ | 98,170 | | | $ | 73,014 | | | $ | 274,715 | | Accumulated depreciation | | | — | | | | (5,360 | ) | | | (25,771 | ) | | | (12,277 | ) | | | (23,681 | ) | | | (53,791 | ) | | | (120,880 | ) | | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | | 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Opening net book amount | | $ | 14,962 | | | $ | 24,172 | | | $ | 14,937 | | | $ | 6,052 | | | $ | 74,489 | | | $ | 19,223 | | | $ | 153,835 | | Additions | | | — | | | | — | | | | 34,802 | | | | 1,318 | | | | — | | | | 5,950 | | | | 42,070 | | Disposals | | | — | | | | — | | | | (9 | ) | | | — | | | | — | | | | — | | | | (9 | ) | Reclassifications | | | — | | | | — | | | | 9,114 | | | | 1,847 | | | | (7,679 | ) | | | (1,847 | ) | | | 1,435 | | Transfer (Note) | | | — | | | | — | | | | 143 | | | | — | | | | — | | | | — | | | | 143 | | Depreciation charges | | | — | | | | (656 | ) | | | (6,143 | ) | | | (3,157 | ) | | | (19,840 | ) | | | (9,519 | ) | | | (39,315 | ) | Net exchange differences | | | — | | | | — | | | | 87 | | | | (4 | ) | | | — | | | | 3 | | | | 86 | | | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | At December 31, 2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 14,962 | | | $ | 29,532 | | | $ | 82,584 | | | $ | 19,878 | | | $ | 50,013 | | | $ | 77,208 | | | $ | 274,177 | | Accumulated depreciation | | | — | | | | (6,016 | ) | | | (29,653 | ) | | | (13,822 | ) | | | (3,043 | ) | | | (63,398 | ) | | | (115,932 | ) | | | $ | 14,962 | | | $ | 23,516 | | | $ | 52,931 | | | $ | 6,056 | | | $ | 46,970 | | | $ | 13,810 | | | $ | 158,245 | | At December 31, 2018 (US$000) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Cost | | $ | 489 | | | $ | 965 | | | $ | 2,698 | | | $ | 649 | | | $ | 1,634 | | | $ | 2,522 | | | $ | 8,957 | | Accumulated depreciation | | | — | | | | (197 | ) | | | (969 | ) | | | (451 | ) | | | (99 | ) | | | (2,071 | ) | | | (3,787 | ) | | | $ | 489 | | | $ | 768 | | | $ | 1,729 | | | $ | 198 | | | $ | 1,535 | | | $ | 451 | | | $ | 5,170 | |
Note: Transferred from prepayments for equipment (shown as “Other non-current assets”).
| B. | Information about the investing activities that were partially paid by cash is provided in Note 6(26)6(27). |
| C. | Information about the leasehold assets is provided in Note 6(11)6(12). |
| D. | Information about the property, plant and equipment that were pledged to others as collateral is provided in Note 8. |
F-22
| (6) | Leasing arrangement – lessee |
Table of ContentEffective from 2019
| A. | The Group leases various assets including buildings, testing equipment and transportation equipment. Lease agreements are typically made for periods of 1 to 6 years. Lease terms are negotiated on an individual basis and contain a wide range of different terms and conditions. The lease agreements do not impose covenants, but leased assets may not be used as security for borrowing purposes. |
| B. | (6)The carrying amount ofright-of-use assets and the depreciation charges are as follows:
|
| | | | | | | | | | | | | | | | December 31, 2019 | | | | | Cost | | | Accumulated
depreciation | | | Carrying
amount | | | | | NT$000 | | | NT$000 | | | NT$000 | | Buildings | | $ | 71,477 | | | ($ | 27,062 | ) | | $ | 44,415 | | Testing equipment | | | 86,328 | | | | (23,698 | ) | | | 62,630 | | Transportation equipment | | | 697 | | | | (131 | ) | | | 566 | | | | | | | | | | | | | | | | | $ | 158,502 | | | ($ | 50,891 | ) | | $ | 107,611 | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | December 31, 2019 | | | | | Cost | | | Accumulated
depreciation | | | Carrying
amount | | | | | US$000 | | | US$000 | | | US$000 | | Buildings | | $ | 2,390 | | | ($ | 905 | ) | | $ | 1,485 | | Testing equipment | | | 2,886 | | | | (792 | ) | | | 2,094 | | Transportation equipment | | | 23 | | | | (4 | ) | | | 19 | | | | | | | | | | | | | | | | | $ | 5,299 | | | ($ | 1,701 | ) | | $ | 3,598 | | | | | | | | | | | | | | |
| | | | | | | | | | | | 2019 | | | | | NT$000 | | | US$000 | | Buildings | | $ | 27,104 | | | $ | 906 | | Testing equipment | | | 20,654 | | | | 691 | | Transportation equipment | | | 131 | | | | 4 | | | | | | | | | | | | | $ | 47,889 | | | $ | 1,601 | | | | | | | | | | |
| C. | For 2019, the additions toIntangibleright-of-use assets amounted to NT$1,770 thousand (US$59 thousand). |
A.The details of intangible assets are as follows:
| | Professional technology | | | Computer software | | | Total | | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2017 | | | | | | | | | | | | | Cost | | $ | 49,239 | | | $ | 17,589 | | | $ | 66,828 | | Accumulated amortization | | | (41,299 | ) | | | (12,216 | ) | | | (53,515 | ) | | | $ | 7,940 | | | $ | 5,373 | | | $ | 13,313 | | 2017 | | | | | | | | | | | | | Opening net book amount | | $ | 7,940 | | | $ | 5,373 | | | $ | 13,313 | | Additions | | | — | | | | 5,706 | | | | 5,706 | | Transfers (Note) | | | — | | | | 227 | | | | 227 | | Amortization charges | | | (5,102 | ) | | | (5,468 | ) | | | (10,570 | ) | Net exchange differences | | | (39 | ) | | | — | | | | (39 | ) | Closing net book amount | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | | At December 31, 2017 | | | | | | | | | | | | | Cost | | $ | 49,114 | | | $ | 23,522 | | | $ | 72,636 | | Accumulated amortization | | | (46,315 | ) | | | (17,684 | ) | | | (63,999 | ) | | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | |
| | Professional technology | | | Computer software | | | Total | | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2018 | | | | | | | | | | | | | Cost | | $ | 49,114 | | | $ | 23,522 | | | $ | 72,636 | | Accumulated amortization | | | (46,315 | ) | | | (17,684 | ) | | | (63,999 | ) | | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | | 2018 | | | | | | | | | | | | | Opening net book amount | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | | Additions | | | — | | | | 3,537 | | | | 3,537 | | Amortization charges | | | (2,799 | ) | | | (5,345 | ) | | | (8,144 | ) | | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | At December 31, 2018 | | | | | | | | | | | | | Cost | | $ | 49,290 | | | $ | 27,058 | | | $ | 76,438 | | Accumulated amortization | | | (49,290 | ) | | | (23,028 | ) | | | (72,318 | ) | | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | At December 31, 2018 (US$000) | | | | | | | | | | | | | Cost | | $ | 1,610 | | | $ | 884 | | | $ | 2,494 | | Accumulated amortization | | | (1,610 | ) | | | (753 | ) | | | (2,363 | ) | | | $ | — | | | $ | 131 | | | $ | 131 | |
(Note): Transferred from prepayments for equipment (shown as “Other non-current assets”).
B.Information about the investing activities that were partially paid by cash is provided in Note 6(26).
F-23
Table of Content
C.The details of the amortization charges of intangible assets (recorded in “Operating expenses”) are as follows:
| | Years ended December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | General and administrative expenses | | $ | 3,001 | | | $ | 2,983 | | | $ | 97 | | Research and development expenses | | | 7,569 | | | | 5,161 | | | | 169 | | | | $ | 10,570 | | | $ | 8,144 | | | $ | 266 | |
| D. | The information on income and expense accounts relating to lease agreements is as follows: |
| | | | | | | | | Items affecting profit or loss | | 2019 | | | | | NT$000 | | | US$000 | | Interest expense on lease liabilities | | $ | 2,323 | | | $ | 78 | | Expense on short-term lease contracts | | | 2,272 | | | | 76 | | Expense on leases oflow-value assets | | | 366 | | | | 12 | |
| E. | (7)
| Other non-current assetsFor 2019, the Group’s total cash outflow for leases amounted to NT$$70,416 thousand (US$2,354 thousand).
|
| | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Refundable deposits | | $ | 27,188 | | | $ | 18,930 | | | $ | 619 | | Prepaid expense for medical research-non-current | | | 20,000 | | | | 20,000 | | | | 653 | | Prepayments for equipment | | | 923 | | | | 27,942 | | | | 913 | | | | $ | 48,111 | | | $ | 66,872 | | | $ | 2,185 | |
| A. | (8)The details of intangible assets are as follows:
|
| | | | | | | | | | | | | | | | Professional
technology | | | Computer
software | | | Total | | | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2018 | | | | | | | | | | | | | Cost | | $ | 49,114 | | | $ | 23,522 | | | $ | 72,636 | | Accumulated amortization | | | (46,315 | ) | | | (17,684 | ) | | | (63,999 | ) | | | | | | | | | | | | | | | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | | | | | | | | | | | | | | | 2018 | | | | | | | | | | | | | Opening net book amount | | $ | 2,799 | | | $ | 5,838 | | | $ | 8,637 | | Additions | | | — | | | | 3,537 | | | | 3,537 | | Amortization charges | | | (2,799 | ) | | | (5,345 | ) | | | (8,144 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | | | | | | | | | | | | | | At December 31, 2018 | | | | | | | | | | | | | Cost | | $ | 49,290 | | | $ | 27,058 | | | $ | 76,438 | | Accumulated amortization | | | (49,290 | ) | | | (23,028 | ) | | | (72,318 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | | | | | | | | | | | | | | | | | | | | | Professional
technology | | | Computer
software | | | Total | | | | | NT$000 | | | NT$000 | | | NT$000 | | At January 1, 2019 | | | | | | | | | | | | | Cost | | $ | 49,290 | | | $ | 27,058 | | | $ | 76,438 | | Accumulated amortization | | | (49,290 | ) | | | (23,028 | ) | | | (72,318 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | | | | | | | | | | | | | | 2019 | | | | | | | | | | | | | Opening net book amount | | $ | — | | | $ | 4,030 | | | $ | 4,030 | | Additions | | | — | | | | 4,177 | | | | 4,177 | | Transfers (Note) | | | — | | | | 243 | | | | 243 | | Amortization charges | | | — | | | | (6,648 | ) | | | (6,648 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 1,802 | | | $ | 1,802 | | | | | | | | | | | | | | | At December 31, 2019 | | | | | | | | | | | | | Cost | | $ | 49,136 | | | $ | 31,479 | | | $ | 80,614 | | Accumulated amortization | | | (49,136 | ) | | | (29,677 | ) | | | (78,812 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 1,802 | | | $ | 1,802 | | | | | | | | | | | | | | | At December 31, 2019(US$000) | | | | | | | | | | | | | Cost | | $ | 1,643 | | | $ | 1,052 | | | $ | 2,695 | | Accumulated amortization | | | (1,643 | ) | | | (992 | ) | | | (2,635 | ) | | | | | | | | | | | | | | | | $ | — | | | $ | 60 | | | $ | 60 | | | | | | | | | | | | | | |
| (Note): | Short-term borrowingsTransferred from prepayments for equipment (shown as “Othernon-current assets”).
|
| B. | Information about the investing activities that were partially paid by cash is provided in Note 6(27). |
| C. | The details of the amortization charges of intangible assets (recorded in “Operating expenses”) are as follows: |
| | | | | | | | | | | | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | General and administrative expenses | | $ | 2,983 | | | $ | 2,796 | | | $ | 93 | | Research and development expenses | | | 5,161 | | | | 3,852 | | | | 129 | | | | | | | | | | | | | | | | | $ | 8,144 | | | $ | 6,648 | | | $ | 222 | | | | | | | | | | | | | | |
Type of borrowings | | December 31, | | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Bank unsecured borrowings | | $ | 46,000 | | | $ | 46,000 | | | $ | 1,503 | | Interest rate | | 1.95%~2.10% | | | 1.95% ~2.10% | | | 1.95% ~2.10% | | Credit line | | $ | — | | | $ | — | | | $ | — | |
| (8) | Othernon-current assets |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Refundable deposits | | $ | 18,930 | | | $ | 24,351 | | | $ | 814 | | Prepaid expense for medicalresearch-non-current | | | 20,000 | | | | 20,000 | | | | 669 | | Prepayments for equipment | | | 27,942 | | | | 74,841 | | | | 2,502 | | | | | | | | | | | | | | | | | $ | 66,872 | | | $ | 119,192 | | | $ | 3,985 | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Bank unsecured borrowings | | $ | 46,000 | | | $ | 46,000 | | | $ | 1,538 | | | | | | | | | | | | | | | Interest rate | | | 1.95% ~2.10% | | | | 1.95 | % | | | 1.95 | % | | | | | | | | | | | | | | Credit line | | $ | — | | | $ | 30,000 | | | $ | 1,003 | | | | | | | | | | | | | | |
Interest expense recognized in profit or loss amounted to NT$942 thousand and NT$942917 thousand (US$31 thousand) for the years ended December 31, 20172018 and 2018,2019, respectively. | | | December 31, | | | December 31, | | | | 2017 | | | 2018 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | US$000 | | Research expenses | | $ | 33,742 | | | $ | 97,930 | | | $ | 3,199 | | | $ | 97,930 | | | $ | 72,576 | | | $ | 2,426 | | Salaries and bonuses | | | | 31,049 | | | | 25,405 | | | | 850 | | Service expenses | | | 9,825 | | | | 48,137 | | | | 1,573 | | | | 48,137 | | | | 14,236 | | | | 476 | | Salaries and bonuses | | | 30,539 | | | | 31,049 | | | | 1,014 | | | Payables on machinery, equipment and intangible assets | | | — | | | | 3,677 | | | | 120 | | | Medical research expenses | | | 4,697 | | | | 2,707 | | | | 89 | | | | 2,707 | | | | 4,526 | | | | 151 | | Labor and health insurance | | | 2,218 | | | | 2,191 | | | | 72 | | | | 2,191 | | | | 1,732 | | | | 58 | | Repair expenses | | | 6,668 | | | | 676 | | | | 22 | | | | 676 | | | | 638 | | | | 21 | | Payables on machinery, equipment and intangible assets | | | | 3,677 | | | | 264 | | | | 9 | | Other accrued expenses | | | 5,852 | | | | 19,901 | | | | 650 | | | | 19,901 | | | | 11,687 | | | | 391 | | | | $ | 93,541 | | | $ | 206,268 | | | $ | 6,739 | | | | | | | | | | | | | | $ | 206,268 | | | $ | 131,064 | | | $ | 4,382 | | | | | | | | | | | | |
F-24
Table of Content
| | | | | | | | | | | | | | | | | Type of loans | | Borrowing period
and repayment term | | | Interest
rate | | | Collateral | | | December 31,
2018 | | | | | | | | | | | | | | NT$000 | | Taiwan Cooperative Bank - secured borrowings | | | Note 1 | | | | 1.85 | % | | | Note 4 | | | | 37,277 | | Taiwan Cooperative Bank - secured borrowings | | | Note 2 | | | | 1.85 | % | | | Note 4 | | | | 28,900 | | Cathay Bank - secured borrowings | | | Note 3 | | | | 5.25 | % | | | Note 4 | | | | 368,580 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 434,757 | | Less: Current portion (Shown as “Other current liabilities”) | | | | | | | | | | | | (66,747 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 368,010 | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | Type of loans | | Borrowing period
and repayment term | | | Interest
rate | | | Collateral | | | December 31, 2019 | | | | | | | | | | | | | | NT$000 | | | US$000 | | Taiwan Cooperative Bank - secured borrowings | | | Note 1 | | | | 1.85 | % | | | Note 4 | | | $ | 35,360 | | | $ | 1,182 | | Taiwan Cooperative Bank - secured borrowings | | | Note 2 | | | | 1.85 | % | | | Note 4 | | | | 25,500 | | | | 852 | | Cathay Bank - secured borrowings | | | Note 3 | | | | 4.75 | % | | | Note 4 | | | | 309,793 | | | | 10,358 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 370,653 | | | | 12,392 | | | | | | | | | | | | | | | | | | | | | | | Less: Current portion (Shown as “Other current liabilities”) | | | | | | | | | | | | (315,145 | ) | | | (10,536 | ) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | $ | 55,508 | | | $ | 1,856 | | | | | | | | | | | | | | | | | | | | | | |
Type of loans | | Borrowing period and repayment term | | Interest rate | | | Collateral | | December 31, 2017 | | | | | | | | | | | | NT$000 | | Taiwan Cooperative Bank - secured borrowings | | Note 1 | | 1.85% | | | Note 5 | | $ | 37,750 | | Taiwan Cooperative Bank - secured borrowings | | Note 2 | | 1.85% | | | Note 5 | | | 32,300 | | | | | | | | | | | | | 70,050 | | Less: Current portion (Shown as “Other current liabilities”) | | | | | | | | | | | (3,873 | ) | | | | | | | | | | | $ | 66,177 | |
Type of loans | | Borrowing period and repayment term | | Interest rate | | | Collateral | | December 31, 2018 | | | | | | | | | | | | NT$000 | | | US$000 | | Taiwan Cooperative Bank - secured borrowings | | Note 1 | | 1.85% | | | Note 5 | | $ | 37,277 | | | $ | 1,218 | | Taiwan Cooperative Bank - secured borrowings | | Note 2 | | 1.85% | | | Note 5 | | | 28,900 | | | | 944 | | Cathay Bank - secured borrowings | | Note 3 and 4 | | 5.25% | | | Note 5 | | | 368,580 | | | | 12,041 | | | | | | | | | | | | | 434,757 | | | | 14,203 | | Less: Current portion (Shown as “Other current liabilities”) | | | | | | | | | | | (66,747 | ) | | | (2,180 | ) | | | | | | | | | | | $ | 368,010 | | | $ | 12,023 | |
| Note 1: | The Company entered into a long-term loan contract with Taiwan Cooperative Bank on September 1, 2015 in the amount of NT$37,750 thousand (US$1,2331,262 thousand). The contract period is from September 2015 to September 2035. The interest is payable monthly for the first 3 years and payable monthly along with the same amount of principal starting from the fourth year. |
| Note 2: | The Company entered into amid-term loan contract with Taiwan Cooperative Bank on September 4, 2015 in the amount of NT$34,000 thousand (US$1,1111,137 thousand). The contract period is from September 2015 to September 2022. The interest is payable monthly for the first 2 years and payable semi-annually along with 5% (NT$1,700 thousand (US$5657 thousand)) of the principal starting from September 2017. The remaining 50% of principal (NT$17,000 thousand (US$555568 thousand)) is required to be repaid in September 2022. |
| Note 3: | The Company and its subsidiary, TLC Biopharmaceuticals, Inc. (“TLC US”) entered into a loan and security agreement with Cathay Bank on June 14, 2018 in the amount of US$12 million. The contract period was from June 2018 to December 2020. The interest is payable monthly for the first six months and payable monthly along with the same amount of principal starting from January 2019. The Company repaid the loan in full amount in November 2018 and the agreement was terminated. |
| Note 4:
| The Company and its subsidiary, TLC US entered into amid-term loan and security agreement with Cathay Bank on December 27, 2018 in the amount of US$12 million. The contract period is from December 2018 to June 2020. The interest is payable monthly for the first six months and payable monthly along with the same amount of principal starting from July 2019.
|
F-25
Table of Content
| Note 4: | Information about the collateral provided for the loans is provided in Note 8. |
Note 5: Information about the collateral provided for the loans is provided in Note 8.
| A. | According to the above two bank loan contracts with Taiwan Cooperative Bank, the Company is restricted from paying cash dividends or other distributions on the common sharesstock and Taiwan Cooperative Bank retains the right in requesting the Company to raisepaid-in capital or to improve financial structure if certain conditions are met. |
| B. | According to the above loan and security agreement with Cathay Bank, if the Company or TLC US violate any of the following covenants, Cathay Bank has the right to demand, among other things, that the Company and TLC US repay the outstanding loan early: |
(i)The Group must maintain an adjusted quick ratio (“Adjusted Quick Ratio”) of at least a minimum of 2.25 to 1.00 and (ii) the Group must maintain an adjusted tangible net worth (“Adjusted Tangible Net Worth”) of no less than US$12 million as per its quarterly and yearly consolidated financial statements. “Adjusted “Adjusted Quick Ratio” means a ratio of cash and cash equivalents plus net trade receivables to the amount of principal payments owing to Cathay Bank under this contract for the next 12 months plus all other current liabilities. “Adjusted Tangible Net Worth” means the differences between the value of the capital stock, partnership interests, or limited liability company interests of the companyCompany and TLC US (and their respective subsidiaries), minus intangible assets, plus deferred revenue.
The Company was in compliance with all of the loan covenants as of December 31, 2018. Accordingly, the entire debt balance for this loan has been classified as non-current liability at December 31, 2018.2018 and 2019. On April 25, 2019,January 9, 2020, Cathay Bank agreed to amend the Company entered into an amendment to the loan and security agreement with Cathay Bank, pursuant to which the applicabilityAdjusted Quick Ratio covenant was amended so that the Adjusted Quick Ratio must be at least 1.75 to 1.00, which became effective beginning January 9, 2020. The Group must maintain an adjusted quick ratio (“Adjusted Quick Ratio”) of at least a minimum of 1.75 to 1.00. “Adjusted Quick Ratio” means a ratio of cash and cash equivalents plus net trade receivables to the above-mentioned loan covenants was deferred until September 30, 2019 (and tested asamount of principal payments owing to Cathay Bank under this contract for the last day of each quarter going forward).next 12 months plus all other current liabilities, but excluding lease liabilities recognized under IFRS 16. The loan and security agreement with Cathay Bank also prohibits the Company from paying cash dividends or making distributions on account of the Company’s capital stock without the consent of Cathay Bank, subject to certain exceptions. | C. | As of December 31, 20172018 and 2018,2019, the undrawn loan facilities amounted to NT$2,7506,623 thousand and NT$6,63211,940 thousand (US$216399 thousand), respectively. The information about the Group’s liquidity risk is provided in Note 12(2)C(c). |
| (12) | (11)
| Finance lease liabilities |
The Group leases testing equipment under finance leases. lease as of December 31, 2018. The leasehold assets are shown as “Property, plant and equipment” as of December 31, 2018 and remeasured and shown as“Right-of-use assets” as of December 31, 2019. Please refer to Notes 6(5) and (6) for details of related information. Future minimum lease payments and their present values as of December 31, 2017 and 2018 are as follows: | | | | | | | | | | | | | | | | December 31, 2018 | | | | | Total finance
lease liabilities | | | Future finance
charges | | | Present value of
finance lease
liabilities | | | | | NT$000 | | | NT$000 | | | NT$000 | | Current | | | | | | | | | | | | | Not later than one year (Note) | | $ | 24,583 | | | ($ | 583 | ) | | $ | 24,000 | | Non-current | | | | | | | | | | | | | Later than one year but not later than two years (Note) | | | 24,198 | | | | (198 | ) | | | 24,000 | | | | | | | | | | | | | | | | | $ | 48,781 | | | ($ | 781 | ) | | $ | 48,000 | | | | | | | | | | | | | | |
| | December 31, 2017 | | | | Total finance lease liabilities | | | Future finance charges | | | Present value of finance lease liabilities | | | | NT$000 | | | NT$000 | | | NT$000 | | Current | | | | | | | | | | | | | Not later than one year (Note) | | $ | 48,466 | | | ($ | 466 | ) | | $ | 48,000 | | Non-current | | | | | | | | | | | | | Later than one year but not later than two years (Note) | | | 4,008 | | | | (8 | ) | | | 4,000 | | | | $ | 52,474 | | | ($ | 474 | ) | | $ | 52,000 | |
F-26
Table of Content
| | December 31, 2018 | | | | Total finance lease liabilities | | | Future finance charges | | | Present value of finance lease liabilities | | | | NT$000 | | | NT$000 | | | NT$000 | | Current | | | | | | | | | | | | | Not later than one year (Note) | | $ | 24,583 | | | ($ | 583 | ) | | $ | 24,000 | | Non-current | | | | | | | | | | | | | Later than one year but not later than two years (Note) | | | 24,198 | | | | (198 | ) | | | 24,000 | | | | $ | 48,781 | | | ($ | 781 | ) | | $ | 48,000 | |
| | December 31, 2018 | | | | Total finance lease liabilities | | | Future finance charges | | | Present value of finance lease liabilities | | | | US$000 | | | US$000 | | | US$000 | | Current | | | | | | | | | | | | | Not later than one year (Note) | | $ | 803 | | | ($ | 19 | ) | | $ | 784 | | Non-current | | | | | | | | | | | | | Later than one year but not later than two years (Note) | | | 791 | | | | (7 | ) | | | 784 | | | | $ | 1,594 | | | ($ | 26 | ) | | $ | 1,568 | |
| Note: | Shown as “Other current liabilities” and “Othernon-current liabilities”, respectively. |
A.Defined benefit plan
| (a) | The Company has a defined benefit pension plan in accordance with the Labor Standards Act, covering all regular employees’ service years prior to the enforcement of the Labor Pension Act on July 1, 2005 and service years thereafter of employees who chose to continue to be subject to the pension mechanism under the Labor Standards Act. Under the defined benefit pension plan, two units are accrued for each year of service for the first 15 years and one unit for each additional year thereafter, subject to a maximum of 45 units. Pension benefits are based on the number of units accrued and the average monthly salaries and wages of the last 6 months prior to retirement. The Company contributes monthly an amount equal to 2% of the employees’ monthly salaries and wages to the retirement fund deposited with Bank of Taiwan, the trustee, under the name of the independent retirement fund committee. Also, the Company would assess the balance in the aforementioned labor pension reserve account by December 31, every year. If the account balance is insufficient to pay the pension calculated by the aforementioned methods to the employees expected to qualify for retirement in the following year, the Company will make contributions to cover for the deficit by the following March. |
F-27
Table of Content
| (b) | (b)
| The amounts recognized in the balance sheet are as follows: |
| | | December 31, | | | December 31, | | | | 2017 | | | 2018 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | US$000 | | Present value of defined benefit obligations | | $ | 6,421 | | | $ | 7,064 | | | $ | 231 | | | $ | 7,064 | | | $ | 7,402 | | | $ | 247 | | Fair value of plan assets | | | (1,319 | ) | | | (1,560 | ) | | | (51 | ) | | | (1,560 | ) | | | (1,805 | ) | | | (60 | ) | | | | | | | | | | | | Net defined benefit liability | | $ | 5,102 | | | $ | 5,504 | | | $ | 180 | | | $ | 5,504 | | | $ | 5,597 | | | $ | 187 | | | | | | | | | | | | |
| (c) | Movements in net defined benefit liabilities are as follows: |
| | | | | | | | | | | | | 2018 | | Present value of
defined benefit
obligations | | | Fair value of
plan assets | | | Net defined
benefit liability | | | | | NT$000 | | | NT$000 | | | NT$000 | | Balance at January 1 | | $ | 6,421 | | | ($ | 1,319 | ) | | $ | 5,102 | | Interest expense / income | | | 83 | | | | (17 | ) | | | 66 | | | | | | | | | | | | | | | | | | 6,504 | | | | (1,336 | ) | | | 5,168 | | | | | | | | | | | | | | | Remeasurements: | | | | | | | | | | | | | Change in financial assumptions | | | 162 | | | | — | | | | 162 | | Experience adjustments | | | 398 | | | | (33 | ) | | | 365 | | | | | | | | | | | | | | | | | | 560 | | | | (33 | ) | | | 527 | | Pension fund contribution | | | — | | | | (191 | ) | | | (191 | ) | | | | | | | | | | | | | | Balance at December 31 | | $ | 7,064 | | | ($ | 1,560 | ) | | $ | 5,504 | | | | | | | | | | | | | | |
Year ended December 31, 2017 | | Present value of defined benefit obligations | | | Fair value of plan assets | | | Net defined benefit liability | | | | 2019 | | | Present value of
defined benefit
obligations | | | Fair value of
plan assets | | | Net defined
benefit liability | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | Balance at January 1 | | $ | 6,141 | | | ($ | 1,086 | ) | | $ | 5,055 | | | $ | 7,064 | | | ($ | 1,560 | ) | | $ | 5,504 | | Current service cost | | | 59 | | | | — | | | | 59 | | | Interest expense / income | | | 105 | | | | (18 | ) | | | 87 | | | | 77 | | | | (17 | ) | | | 60 | | | | | | | | | | | | | | | | | 7,141 | | | | (1,577 | ) | | | 5,564 | | | | | 6,305 | | | | (1,104 | ) | | | 5,201 | | | | | | | | | | | Remeasurements: | | | | | | | | | | | | | | | | | | | Change in financial assumptions | | | 312 | | | | — | | | | 312 | | | | 304 | | | | — | | | | 304 | | Experience adjustments | | | (196 | ) | | | 8 | | | | (188 | ) | | | (43 | ) | | | (50 | ) | | | (93 | ) | | | | 116 | | | | 8 | | | | 124 | | | | | | | | | | | | | | | 261 | | | | (50 | ) | | | 211 | | Pension fund contribution | | | — | | | | (223 | ) | | | (223 | ) | | | — | | | | (178 | ) | | | (178 | ) | | | | | | | | | | | | Balance at December 31 | | $ | 6,421 | | | ($ | 1,319 | ) | | $ | 5,102 | | | $ | 7,402 | | | ($ | 1,805 | ) | | $ | 5,597 | | | | | | | | | | | | | Balance at December 31(US$000) | | | $ | 247 | | | ($ | 60 | ) | | $ | 187 | | | | | | | | | | | | |
Year ended December 31, 2018 | | Present value of defined benefit obligations | | | Fair value of plan assets | | | Net defined benefit liability | | | | NT$000 | | | NT$000 | | | NT$000 | | Balance at January 1 | | $ | 6,421 | | | ($ | 1,319 | ) | | $ | 5,102 | | Interest expense / income | | | 83 | | | | (17 | ) | | | 66 | | | | | 6,504 | | | | (1,336 | ) | | | 5,168 | | Remeasurements: | | | | | | | | | | | | | Change in financial assumptions | | | 162 | | | | — | | | | 162 | | Experience adjustments | | | 398 | | | | (33 | ) | | | 365 | | | | | 560 | | | | (33 | ) | | | 527 | | Pension fund contribution | | | — | | | | (191 | ) | | | (191 | ) | Balance at December 31 | | $ | 7,064 | | | ($ | 1,560 | ) | | $ | 5,504 | | Balance at December 31 (US$000) | | $ | 231 | | | ($ | 51 | ) | | $ | 180 | |
F-28
Table of Content
| (d) | (d)
| The Bank of Taiwan was commissioned to manage the Fund of the Company’s defined benefit pension plan in accordance with the Fund’s annual investment and utilization plan and the “Regulations for Revenues, Expenditures, Safeguard and Utilization of the Labor Retirement Fund” (Article 6: The scope of utilization for the Fund includes deposit in domestic or foreign financial institutions, investment in domestic or foreignlisted, over-the-counter, or private placement equity securities, and investment in domestic or foreign real estate securitization products, etc.). With regard to the utilization of the Fund, its minimum earnings in the annual distributions on the final financial statements shall be no less than the earnings attainable from the amounts accruedfrom two-year time deposits with the interest rates offered by local banks. If the earnings is less than aforementioned rates, government shall make payment for the deficit after being authorized by the Regulator. The Company has no right to participate in managing and operating that fund and hence the Company is unable to disclose the classification of plan asset fair value in accordance with IAS 19 paragraph 142. The composition of fair value of plan assets as of December 31, 20172018 and 20182019 is given in the Annual Labor Retirement Fund Utilization Report announced by the government. |
| (e) | The principal actuarial assumptions used were as follows: |
| | | | | | | | | | | | 2018 | | | 2019 | | Discount rate | | | 1.10 | % | | | 0.70 | % | | | | | | | | | | Future salary increases | | | 2.00 | % | | | 2.00 | % | | | | | | | | | |
| | Years ended December 31, | | | | 2017 | | | 2018 | | Discount rate | | | 1.30 | % | | | 1.10 | % | Future salary increases | | | 2.00 | % | | | 2.00 | % |
Assumptions regarding future mortality experience are set based on actuarial valuation in accordance with the 5th version of Taiwan Standard Ordinary Experience Mortality Tables. The present value of defined benefit obligation is affected whenever there is change in main actuarial assumption. The sensitivity analysis is as follows: | | Discount rate | | | Future salary increases | | | | Increase 0.25% | | | Decrease 0.25% | | | Increase 0.25% | | | Decrease 0.25% | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | December 31, 2017 | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligations | | ($ | 197 | ) | | $ | 205 | | | $ | 186 | | | ($ | 181 | ) |
| | | | | | | | | | | | | | | | | | | | Discount rate | | | Future
salary increases | | | | | Increase
0.25% | | | Decrease
0.25% | | | Increase
0.25% | | | Decrease
0.25% | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | December 31, 2018 | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligations | | ($ | 201 | ) | | $ | 209 | | | $ | 189 | | | ($ | 183 | ) | | | | | | | | | | | | | | | | | |
| | Discount rate | | | Future salary increases | | | | Increase 0.25% | | | Decrease 0.25% | | | Increase 0.25% | | | Decrease 0.25% | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | December 31, 2018 | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligations | | ($ | 201 | ) | | $ | 209 | | | $ | 189 | | | ($ | 183 | ) | December 31, 2018 (US$000) | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligation | | ($ | 7 | ) | | $ | 7 | | | $ | 6 | | | ($ | 6 | ) |
| | | | | | | | | | | | | | | | | | | | Discount rate | | | Future salary increases | | | | | Increase
0.25% | | | Decrease
0.25% | | | Increase
0.25% | | | Decrease
0.25% | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | December 31, 2019 | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligations | | ($ | 192 | ) | | $ | 198 | | | $ | 176 | | | ($ | 172 | ) | | | | | | | | | | | | | | | | | | December 31, 2019(US$000) | | | | | | | | | | | | | | | | | Effect on present value of defined benefit obligation | | ($ | 6 | ) | | $ | 7 | | | $ | 6 | | | ($ | 6 | ) | | | | | | | | | | | | | | | | | |
The sensitivity analysis above was arrived at based on the assumption that other conditions remain unchanged. In practice, more than one assumption may change all at once. The method of analyzing sensitivity and the method of calculating net pension liability in the balance sheet are the same..same. | (f) | Expected contributions to the defined benefit pension plans of the Company for the year ending December 31, 20192020 is NT$194174 thousand (US$6 thousand). |
F-29
Table of Content
| (g) | (g)
| As of December 31, 2018,2019, the weighted average duration of the retirement plan is 1514 years. The analysis of timing of the future pension payment is as follows: |
| | | NT$000 | | | US$000 | | | NT$000 | | | US$000 | | Within 1 year | | $ | — | | | $ | — | | | $ | — | | | $ | — | | 1-2 year(s) | | | — | | | | — | | | | 257 | | | | 9 | | 2-5 years | | | 462 | | | | 15 | | | | 307 | | | | 10 | | 6-10 years | | | 2,753 | | | | 90 | | | | 3,176 | | | | 106 | | | | $ | 3,215 | | | $ | 105 | | | | | | | | | | | $ | 3,740 | | | $ | 125 | | | | | | | | | |
| B. | Defined contribution plans |
Effective July 1, 2005, the Company has established a defined contribution pension plan (the “New Plan”) under the Labor Pension Act (the “Act”), covering all regular employees with R.O.C. nationality. Under the New Plan, the Company contributes monthly an amount based on 6% of the employees’ monthly salaries and wages to the employees’ individual pension accounts at the Bureau of Labor Insurance. The benefits accrued are paid monthly or in lump sum upon termination of employment. The pension costs under the defined contribution pension plan of the Company for the years ended December 31, 20172018 and 20182019 are NT$8,6768,843 thousand and NT$8,843thousand7,231 thousand (US$289242 thousand), respectively. | C. | The subsidiaries have defined contribution plans in accordance with the local regulations, and contributions are based on a certain percentage of employees’ salaries and wages. Other than the yearly contributions, the subsidiaries have no further obligations. The pension costs of the subsidiaries for the years ended December 31, 20172018 and 20182019 were NT$1,4231,487 thousand and NT$1,4871,065 (US$4936 thousand), respectively. |
F-30
Table of Content
| (14) | (13)
| Share-based payment |
| A. | For the years ended December 31, 2016, 2017 , 2018 and 20182019, the Company’s equity-settled share-based payment arrangements are as follows: |
| Type of arrangement | | Grant date | | Quantity granted (in thousands) | | | Contract period | | Vesting conditions | | Grant date | | | Quantity granted
(in thousands) | | | Contract period | | Vesting conditions | Employee stock options | | 2012.05.08 | | | 62.8 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2012.05.08 | | | | 63 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2013.11.14 | | | 883.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2013.11.14 | | | | 883 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2014.03.20 | | | 153.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2014.03.20 | | | | 153 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2014.08.15 | | | 82.3 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2014.08.15 | | | | 82 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2015.02.26 | | | 1,102.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2015.02.26 | | | | 1,102 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2015.04.30 | | | 16.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2015.04.30 | | | | 16 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2015.05.04 | | | 35.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2015.05.04 | | | | 35 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2015.07.30 | | | 50.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2015.07.30 | | | | 50 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2015.10.29 | | | 180.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2015.10.29 | | | | 180 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2016.02.25 | | | 1,391.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2016.02.25 | | | | 1,391 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2016.08.11 | | | 140.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2016.08.11 | | | | 140 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2016.11.03 | | | 73.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2016.11.03 | | | | 73 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2018.06.29 | | | 1,320.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2018.06.29 | | | | 1,320 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | 2018.07.02 | | | 65.0 | | | 5 years | | Gradually vested after 2 year service (Note 1) | | | 2018.07.02 | | | | 65 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Restricted stocks to employees(Note 2) | | 2014.08.15 | | | 307.0 | | | 3 years | | Service and performance (Note 3) | | Restricted stocks to employees(Note 2) | | 2014.11.14 | | | 43.0 | | | 3 years | | Service and performance (Note 3) | | Restricted stocks to employees(Note 2) | | 2017.11.16 | | | 500.0 | | | 3 years | | (Note 4) | | Restricted stocks to employees(Note 2) | | 2018.07.02 | | | 50.0 | | | 3 years | | (Note 4) | | Employee stock options | | | | 2019.03.07 | | | | 115 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Employee stock options | | | | 2019.05.08 | | | | 300 | | | 5 years | | Gradually vested after 2 year service (Note 1) | Restricted stocks to employees (Note 2) | | | | 2017.11.16 | | | | 500 | | | 3 years | | (Note 3) | Restricted stocks to employees (Note 2) | | | | 2018.07.02 | | | | 50 | | | 3 years | | (Note 3) |
| Note 1: | Employees with 2 year service are entitled to 50%; after the 2 year service, the ratio will increase by 1/48 every month for the following 24 months; and employees with 4 year service are entitled to 100%. |
| Note 2: | The restricted stocks issued by the Company cannot be transferred within the vesting period, but voting rights and dividend rights are not restricted on these stocks. Employees are required to return the stocks but not required to return the dividends received if they resign during the vesting period. |
F-31
Table of Content
| Note 3: | For the employees who are currently working in the Company and whose services have reached 1 year, 2 years and 3 years while achieving targeted performance, which are mainly based on the progress of research and development projects, and they have made certain contribution. The applicable accumulated maximum vested share ratio is 30%, 60% and 100%, respectively.
|
| Note 4:
| For the employees who are currently working in the Company and whose services have reached 1 year, 2 years and 3 years without violating the terms of employment agreement entered between the Company and employees, they are entitled to 20%, 30% and 50%, respectively. |
| B. | Details of the share-based payment arrangements are as follows: |
| (a) | Employee stock options |
| | | 2016 | | | 2017 | | Stock options | | No. of units (in thousands) | | | Weighted-average exercise price (in NT dollars) | | | No. of units
(in thousands) | | | Weighted-average
exercise price
(in NT dollars) | | Options outstanding at beginning of the year | | | 3,148 | | | $ | 267 | | | | 4,168 | | | $ | 231 | | Options granted | | | 1,800 | | | | 155 | | | Options exercised | | | (145 | ) | | | 42 | | | Options forfeited | | | (635 | ) | | | 237 | | | | (655 | ) | | | 189 | | | | | | | | | Options outstanding at end of the year | | | 4,168 | | | | 231 | | | | 3,513 | | | | 239 | | | | | | | | | Options exercisable at end of the year | | | 901 | | | | 338 | | | | 1,844 | | | | 301 | | | | | | | | | Options permitted but not yet granted at end of the year | | | — | | | | | | | | — | | | | | | | | | | |
| | | | | | | | | | | | 2018 | | Stock options | | No. of units
(in thousands) | | | Weighted-average
exercise price
(in NT dollars) | | Options outstanding at beginning of the year | | | 3,513 | | | $ | 239 | | Options granted | | | 1,385 | | | | 101 | | Options expired | | | (831 | ) | | | 379 | | Options forfeited | | | (338 | ) | | | 196 | | | | | | | | | | | Options outstanding at end of the year | | | 3,729 | | | | 152 | | | | | | | | | | | Options exercisable at end of the year | | | 1,957 | | | | 188 | | | | | | | | | | | Options permitted but not yet granted at end of the year | | | 415 | | | | | | | | | | | | | | |
| | | 2017 | | | 2019 | | Stock options | | No. of units (in thousands) | | | Weighted-average exercise price (in NT dollars) | | | No. of units
(in thousands) | | | Weighted-average
exercise price
(in NT dollars) | | | Weighted-average
exercise price
(in US dollars) | | Options outstanding at beginning of the year | | | 4,168 | | | $ | 231 | | | | 3,729 | | | $ | 152 | | | $ | 5 | | Options granted | | | | 415 | | | | 93 | | | | 3 | | Options expired | | | | (171 | ) | | | 232 | | | | 8 | | Options forfeited | | | (655 | ) | | | 189 | | | | (749 | ) | | | 138 | | | | 5 | | | | | | | | | | | Options outstanding at end of the year | | | 3,513 | | | | 239 | | | | 3,224 | | | | 134 | | | | 4 | | | | | | | | | | | Options exercisable at end of the year | | | 1,844 | | | | 301 | | | | 1,669 | | | | 167 | | | | 6 | | | | | | | | | | | Options permitted but not yet granted at end of the year | | | — | | | | | | | | — | | | | | | | | | | | | | | |
| | 2018 | | Stock options | | No. of units (in thousands) | | | Weighted-average exercise price (in NT dollars) | | | Weighted-average exercise price (in US dollars) | | Options outstanding at beginning of the year | | | 3,513 | | | $ | 239 | | | $ | 8 | | Options granted | | | 1,385 | | | | 101 | | | | 3 | | Options expired | | | (831 | ) | | | 379 | | | | 12 | | Options forfeited | | | (338 | ) | | | 196 | | | | 6 | | Options outstanding at end of the year | | | 3,729 | | | | 152 | | | | 5 | | Options exercisable at end of the year | | | 1,957 | | | | 188 | | | | 6 | | Options permitted but not yet granted at end of the year | | | 415 | | | | | | | | | |
F-32
Table of Content
| (b) | (b)
| Restricted stocks to employees |
| | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | Shares (in thousands) | | | Shares (in thousands) | | | Shares (in thousands) | | | Shares (in thousands) | | | Shares (in thousands) | | | Shares (in thousands) | | At January 1 | | | 204 | | | | 111 | | | | 500 | | | | 111 | | | | 500 | | | | 422 | | Granted for the year (Note 1) | | | — | | | | 500 | | | | 50 | | | | 500 | | | | 50 | | | | — | | Expired for the year (Note 2) | | | (26 | ) | | | (31 | ) | | | (35 | ) | | | (31 | ) | | | (35 | ) | | | (56 | ) | Vested/restrictions removed for the year | | | (67 | ) | | | (80 | ) | | | (93 | ) | | | (80 | ) | | | (93 | ) | | | (139 | ) | | | | | | | | | | | | At December 31 | | | 111 | | | | 500 | | | | 422 | | | | 500 | | | | 422 | | | | 227 | | | | | | | | | | | | |
| Note 1: | For the restricted stocks granted with the compensation cost accounted for using the fair value method, the fair values on the grant datedates are calculated based on the closing priceprices on the grant datedates subtracting the subscription price of NT$10 (in dollars). |
| Note 2: | Please refer to Note 6(15)D.6(16)E. |
| C. | C.
| The weighted-average stock price of stock options at exercise dates for the year ended December 31, 2016 was NT$134.73 (in dollars). No stock options were exercised for the year ended December 31, 2017, 2018 and 2018.2019.
|
| D. | The expiry date and exercise price of stock options outstanding at the balance sheet dates are as follows: |
| | | | | | | | December 31, 2017 | | | | | | | | | | Options outstanding at end of year | | | Options exercisable at end of year | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Remaining contractual life (years) | | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Exercise price (in dollars) | | NT$ | | | | | | | | | | | NT$ | | | | | | | NT$ | | $ | 379 | | | | 831 | | | | 0.87 | | | $ | 379 | | | | 831 | | | $ | 379 | | | 272 | | | | 150 | | | | 1.22 | | | | 272 | | | | 142 | | | | 272 | | | 206 | | | | 60 | | | | 1.62 | | | | 206 | | | | 51 | | | | 206 | | | 246.5 | | | | 955 | | | | 2.16 | | | | 246.5 | | | | 688 | | | | 246.5 | | | 225 | | | | 16 | | | | 2.33 | | | | 225 | | | | 11 | | | | 225 | | | 225 | | | | 35 | | | | 2.34 | | | | 225 | | | | 23 | | | | 225 | | | 148 | | | | 38 | | | | 2.58 | | | | 148 | | | | 23 | | | | 148 | | | 141 | | | | 138 | | | | 2.83 | | | | 141 | | | | 75 | | | | 141 | | | 159 | | | | 1,148 | | | | 3.15 | | | | 159 | | | | — | | | | — | | | 128.5 | | | | 69 | | | | 3.61 | | | | 128.5 | | | | — | | | | — | | | 122 | | | | 73 | | | | 3.84 | | | | 122 | | | | — | | | | — | | | | | | | 3,513 | | | | | | | | | | | | 1,844 | | | | | |
F-33
Table of Content
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | December 31, 2018 | | | | | | | | | Options outstanding
at end of year | | | Options exercisable
at end of year | | Exercise price (in dollars) | | | Quantity
(in thousands) | | | Remaining
contractual
life (years) | | | Exercise price
(in dollars) | | | Quantity
(in thousands) | | | Exercise price
(in dollars) | | | NT$ | | | | | | | | | NT$ | | | | | | NT$ | | | | $ 249.7 | | | | 119 | | | | 0.22 | | | $ | 249.7 | | | | 119 | | | $ | 249.7 | | | | 191.7 | | | | 52 | | | | 0.62 | | | | 191.7 | | | | 52 | | | | 191.7 | | | | 227.3 | | | | 820 | | | | 1.16 | | | | 227.3 | | | | 787 | | | | 227.3 | | | | 208.4 | | | | 15 | | | | 1.33 | | | | 208.4 | | | | 14 | | | | 208.4 | | | | 208.4 | | | | 35 | | | | 1.34 | | | | 208.4 | | | | 31 | | | | 208.4 | | | | 140.8 | | | | 38 | | | | 1.58 | | | | 140.8 | | | | 33 | | | | 140.8 | | | | 134.7 | | | | 138 | | | | 1.83 | | | | 134.7 | | | | 109 | | | | 134.7 | | | | 150.5 | | | | 1,063 | | | | 2.15 | | | | 150.5 | | | | 758 | | | | 150.5 | | | | 123.7 | | | | 28 | | | | 2.61 | | | | 123.7 | | | | 16 | | | | 123.7 | | | | 118.0 | | | | 61 | | | | 2.84 | | | | 118.0 | | | | 38 | | | | 118.0 | | | | 99.2 | | | | 1,295 | | | | 4.49 | | | | 99.2 | | | | — | | | | — | | | | 101.2 | | | | 65 | | | | 4.50 | | | | 101.2 | | | | — | | | | — | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 3,729 | | | | | | | | | | | | 1,957 | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | December 31, 2019 | | | | | | | | | Options outstanding
at end of year | | | Options exercisable
at end of year | | Exercise price (in dollars) | | | Quantity
(in thousands) | | | Remaining
contractual
life (years) | | | Exercise price
(in dollars) | | | Remaining
contractual
life (years) | | | Exercise price
(in dollars) | | | NT$ | | | | | | | | | NT$ | | | | | | NT$ | | | | $ 207.3 | | | | 671 | | | | 0.16 | | | $ | 207.3 | | | | 671 | | | $ | 207.3 | | | | 191.0 | | | | 8 | | | | 0.33 | | | | 191.0 | | | | 8 | | | | 191.0 | | | | 191.0 | | | | 35 | | | | 0.34 | | | | 191.0 | | | | 35 | | | | 191.0 | | | | 132.7 | | | | 30 | | | | 0.58 | | | | 132.7 | | | | 30 | | | | 132.7 | | | | 127.5 | | | | 120 | | | | 0.83 | | | | 127.5 | | | | 120 | | | | 127.5 | | | | 141.1 | | | | 792 | | | | 1.16 | | | | 141.1 | | | | 760 | | | | 141.1 | | | | 118.0 | | | | 8 | | | | 1.62 | | | | 118.0 | | | | 7 | | | | 118.0 | | | | 113.1 | | | | 50 | | | | 1.85 | | | | 113.1 | | | | 38 | | | | 113.1 | | | | 98.4 | | | | 1,215 | | | | 3.50 | | | | 98.4 | | | | — | | | | — | | | | 96.6 | | | | 75 | | | | 4.19 | | | | 96.6 | | | | — | | | | — | | | | 90.6 | | | | 220 | | | | 4.36 | | | | 90.6 | | | | — | | | | — | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 3,224 | | | | | | | | | | | | 1,669 | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | December 31 , 2019 | | | | | | | | | Options outstanding
at end of year | | | Options exercisable
at end of year | | Exercise price (in dollars) | | | Quantity
(in thousands) | | | Remaining
contractual
life (years) | | | Exercise price
(in dollars) | | | Quantity
(in thousands) | | | Exercise price
(in dollars) | | | US$ | | | | | | | | | US$ | | | | | | US$ | | | | $ 6.93 | | | | 671 | | | | 0.16 | | | $ | 6.93 | | | | 671 | | | $ | 6.93 | | | | 6.39 | | | | 8 | | | | 0.33 | | | | 6.39 | | | | 8 | | | | 6.39 | | | | 6.39 | | | | 35 | | | | 0.34 | | | | 6.39 | | | | 35 | | | | 6.39 | | | | 4.44 | | | | 30 | | | | 0.58 | | | | 4.44 | | | | 30 | | | | 4.44 | | | | 4.26 | | | | 120 | | | | 0.83 | | | | 4.26 | | | | 120 | | | | 4.26 | | | | 4.72 | | | | 792 | | | | 1.16 | | | | 4.72 | | | | 760 | | | | 4.72 | | | | 3.95 | | | | 8 | | | | 1.62 | | | | 3.95 | | | | 7 | | | | 3.95 | | | | 3.78 | | | | 50 | | | | 1.85 | | | | 3.78 | | | | 39 | | | | 3.78 | | | | 3.29 | | | | 1,215 | | | | 3.50 | | | | 3.29 | | | | — | | | | — | | | | 3.23 | | | | 75 | | | | 4.19 | | | | 3.23 | | | | — | | | | — | | | | 3.03 | | | | 220 | | | | 4.36 | | | | 3.03 | | | | — | | | | — | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 3,224 | | | | | | | | | | | | 1,669 | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | December 31, 2018 | | | | | | | | | | Options outstanding at end of year | | | Options exercisable at end of year | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Remaining contractual life (years) | | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Exercise price (in dollars) | | NT$ | | | | | | | | | | | NT$ | | | | | | | NT$ | | $ | 249.7 | | | | 119 | | | | 0.22 | | | $ | 249.7 | | | | 119 | | | $ | 249.7 | | | 191.7 | | | | 52 | | | | 0.62 | | | | 191.7 | | | | 52 | | | | 191.7 | | | 227.3 | | | | 820 | | | | 1.16 | | | | 227.3 | | | | 787 | | | | 227.3 | | | 208.4 | | | | 15 | | | | 1.33 | | | | 208.4 | | | | 14 | | | | 208.4 | | | 208.4 | | | | 35 | | | | 1.34 | | | | 208.4 | | | | 31 | | | | 208.4 | | | 140.8 | | | | 38 | | | | 1.58 | | | | 140.8 | | | | 33 | | | | 140.8 | | | 134.7 | | | | 138 | | | | 1.83 | | | | 134.7 | | | | 109 | | | | 134.7 | | | 150.5 | | | | 1,063 | | | | 2.15 | | | | 150.5 | | | | 758 | | | | 150.5 | | | 123.7 | | | | 28 | | | | 2.61 | | | | 123.7 | | | | 16 | | | | 123.7 | | | 118.0 | | | | 61 | | | | 2.84 | | | | 118.0 | | | | 38 | | | | 118.0 | | | 99.2 | | | | 1,295 | | | | 4.49 | | | | 99.2 | | | | — | | | | — | | | 101.2 | | | | 65 | | | | 4.50 | | | | 101.2 | | | | — | | | | — | | | | | | | 3,729 | | | | | | | | | | | | 1,957 | | | | | |
| | | | | | | | December 31, 2018 | | | | | | | | | | Options outstanding at end of year | | | Options exercisable at end of year | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Remaining contractual life (years) | | | Exercise price (in dollars) | | | Quantity (in thousands) | | | Exercise price (in dollars) | | US$ | | | | | | | | | | | US$ | | | | | | | US$ | | $ | 8.16 | | | | 119 | | | | 0.22 | | | $ | 8.16 | | | | 119 | | | | 8.16 | | | 6.26 | | | | 52 | | | | 0.62 | | | | 6.26 | | | | 52 | | | | 6.26 | | | 7.43 | | | | 820 | | | | 1.16 | | | | 7.43 | | | | 787 | | | | 7.43 | | | 6.81 | | | | 15 | | | | 1.33 | | | | 6.81 | | | | 14 | | | | 6.81 | | | 6.81 | | | | 35 | | | | 1.34 | | | | 6.81 | | | | 31 | | | | 6.81 | | | 4.60 | | | | 38 | | | | 1.58 | | | | 4.60 | | | | 33 | | | | 4.60 | | | 4.40 | | | | 138 | | | | 1.83 | | | | 4.40 | | | | 109 | | | | 4.40 | | | 4.92 | | | | 1,063 | | | | 2.15 | | | | 4.92 | | | | 758 | | | | 4.92 | | | 4.04 | | | | 28 | | | | 2.61 | | | | 4.04 | | | | 16 | | | �� | 4.04 | | | 3.85 | | | | 61 | | | | 2.84 | | | | 3.85 | | | | 38 | | | | 3.85 | | | 3.24 | | | | 1,295 | | | | 4.49 | | | | 3.24 | | | | — | | | | — | | | 3.31 | | | | 65 | | | | 4.50 | | | | 3.31 | | | | — | | | | | | | | | | | 3,729 | | | | | | | | | | | | 1,957 | | | | | |
F-34
Table of Content
| E. | E.
| The fair value of stock options granted on grant date is measured using the Black-ScholesBlack Scholes option-pricing model. Relevant information is as follows: |
Employee stock options | | | | | | Grant date | | June 29, 2018 | | July 2, 2018 | Dividend yield | | — | | — | Expected volatility | | 43.59%~44.04% | | 43.60%~44.03% | Risk-free interest rate | | 0.65%~0.69% | | 0.65%~0.70% | Expected life (years) | | 3.5~4.5 | | 3.5~4.5 | Per share exercise price (in NT dollars) | | $ 100.5 | | $ 102.5 | Weighted average stock options fair value (in NT dollars) | | $ 33~37 | | $ 33~38 | | | | | Grant date | | March 7, 2019 | | May 8, 2019 | Dividend yield | | — | | — | Expected volatility | | 41.76%~42.66% | | 38.85%~41.63% | Risk-free interest rate | | 0.62%~0.65% | | 0.57%~0.60% | Expected life (years) | | 3.5~4.5 | | 3.5~4.5 | Per share exercise price (in NT dollars) | | $ 97.4 | | $ 91.3 | | | (US$3.26 dollars) | | (US$3.05 dollars) | Weighted average stock options fair value (in NT dollars) | | $31~$35 | | $27~$32 | | | (US$1.0~1.2 dollars) | | (US$0.9~1.1 dollars) |
Grant date
| | | June 29, 2018
| | | July 2, 2018
| | Dividend yield
| | | | —
| | | | —
| | Expected volatility
| | | 43.59%~44.04%
| | | 43.60%~44.03
| | Risk-free interest rate
| | | 0.65%~0.69%
| | | 0.65%~0.70%
| | Expected life (years)
| | | 3.5~4.5
| | | 3.5~4.5
| | Per share exercise price (in NT
dollars)
| | | $ 100.5
(US$3.28 dollars)
| | | $ 102.5
(US$3.35 dollars)
| | Weighted average stock
options fair value
(in NT dollars)
| | | $ 33~37
(US$1.1~1.2 dollars)
| | | $ 33~38
(US$1.1~1.2 dollars)
| |
| F. | Expenses incurred on share-based payment transactions are shown below: |
| | Years ended December 31, | | | | 2016 | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Equity-settled | | $ | 100,573 | | | $ | 57,149 | | | $ | 41,386 | | | $ | 1,352 | |
| | | | | | | | | | | | | | | | | | | | 2017 | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Equity-settled | | $ | 57,149 | | | $ | 41,386 | | | $ | 26,793 | | | $ | 896 | | | | | | | | | | | | | | | | | | |
| (15) | (14)
| Provisions (decommissioning liabilities) |
| | 2018 | | | | | NT$000 | | | At January 1 | | $ | 6,922 | | | Used during the year | | | — | | | At December 31 | | $ | 6,922 | | | At December 31 (US$000) | | $ | 226 | | |
| | | | | | | | 2019 | | | | | NT$000 | | At January 1 | | $ | 6,922 | | Unused amount reversed | | | (490 | ) | | | | | | At December 31 | | $ | 6,432 | | | | | | | At December 31 (US$000) | | $ | 215 | | | | | | |
Analysis of total provisions isare shown below: | | | | | | December 31, 2017 | | | December 31, 2018 | | | | NT$000 | | | NT$000 | | | US$000 | | Non-current | | $ | 6,922 | | | $ | 6,922 | | | $ | 226 | |
| | | | | | | | | | | | | | | | December 31, | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Non-current | | $ | 6,922 | | | $ | 6,432 | | | $ | 215 | | | | | | | | | | | | | | |
In accordance with the requirements specified in the agreements, the Group bears the obligation for the costs of dismantling, removing the asset and restoring the site of its rented office in the future. A provision is recognized for the present value of costs to be incurred for dismantling, removing the asset and restoring the site. It is expected that the provision will be used in 1~2 to 4 years. | (16) | (15)
| Common stockshares |
| A. | A.
| As of December 31, 2018,2019, the Company’s authorized capital was NT$2,000,000 thousand (US$65,33866,867 thousand), and thepaid-in capital was NT$640,451741,939 thousand (US$20,923 24,806 thousand) with a par value of NT$10 (in dollars) (US$0.3 (in dollars)) per share. All proceeds from shares issued have been collected. |
F-35
Table of Content
Movements in the number of the Company’s common shares outstanding are as follows (Unit: thousand shares): | | 2016 | | | 2017 | | | 2018 | | At January 1 | | | 55,620 | | | | 55,730 | | | | 56,199 | | Cash capital increase – issuance of American Depositary Shares | | | — | | | | — | | | | 7,831 | | Issuance of employee restricted stocks | | | — | | | | 500 | | | | 50 | | Employee stock options exercised | | | 145 | | | | — | | | | — | | Cancellation of employee restricted stocks | | | (35 | ) | | | (31 | ) | | | (35 | ) | At December 31 | | | 55,730 | | | | 56,199 | | | | 64,045 | |
| | | | | | | | | | | | | | | | 2017 | | | 2018 | | | 2019 | | Issued common shares at January 1 | | | 55,730 | | | | 56,199 | | | | 64,045 | | Cash capital increase | | | — | | | | — | | | | 10,200 | | Cash capital increase – issuance of American Depositary Shares | | | — | | | | 7,831 | | | | — | | Issuance of employee restricted stocks | | | 500 | | | | 50 | | | | — | | Cancellation of employee restricted stocks | | | (31 | ) | | | (35 | ) | | | (51 | ) | | | | | | | | | | | | | | Issued common shares at December 31 | | | 56,199 | | | | 64,045 | | | | 74,194 | | Restricted stocks retrieved from employees and to be cancelled | | | — | | | | — | | | | (5 | ) | | | | | | | | | | | | | | Outstanding common shares at December 31 | | | 56,199 | | | | 64,045 | | | | 74,189 | | | | | | | | | | | | | | |
| B. | To increase the Company’s working capital, the stockholders at their extraordinary stockholders’ meeting on March 10, 2011 adopted a resolution to raise additional cash through private placement with the effective date set on March 25, 2011. The maximum number of shares to be issued through the private placement was 4,711 thousand shares at a subscription price of NT$42.45 (in dollars) per share. The amount of capital raised through the private placement was NT$200,000 thousand which had been registered. Pursuant to the Securities and Exchange Act of the ROC, the common shares raised through the private placement are subject to certain transfer restrictions and cannot be listed on the stock exchange until three years after they have been issued and have applied for retroactive handling of public issuance procedures. Other than these restrictions, the rights and obligations of the common shares raised through the private placement are the same as other issued common shares. |
| C. | In February 2018, the Company filed thea registration statement on FormF-1, with the U.S. Securities and Exchange Commission (“SEC”) for the initial public offering in the United States of its American Depositary Shares (“ADS”ADSs”) representing common shares. The registration statement for listing its ADSs in the Nasdaq Global Market was declared effective by the SEC inon November 21, 2018, and the Company’ s ADSs began trading on the Nasdaq Global Market under the Ticker symbol “TLC”. |
The Company sold an aggregateactual units of 3,915,550 ADSs infor this offering were 3,915,550, and each ADS represents representing two of the Company'sCompany’s common shares, which in the aggregate represents 7,831,100 common shares. The offering price per ADS was US$5.80 (in dollars), equivalent to a price per common share of NT$89.32 (in dollars). As of December 31, 2019, the outstanding ADRs were 3,915,550 units, or 7,831,100 common shares, representing 10.55% of the Company’s issued common shares. The terms of ADS are as follows: ADSs holders may, pursuant to the Depositary Agreement and the relevant laws and regulations of the R.O.C., exercise the voting rights pertaining to the underlying common sharesstock represented by the ADSs. | (b) | Dividends, stock warrants and other rights |
ADSs holders and common shareholders are all entitled to receive dividends. The Depositary may issue new ADSs in proportion to ADSs holding ratios or raise the number of shares of common shareshares represented by each unit of ADSs or sell stock dividends on behalf of ADSs holders and distribute proceeds to them in proportion to their ADSs holding ratios. | D. | In order to raise funds for drug development, on July 30, 2019 the Board of Directors resolved to increase its capital for cash, which was approved by the FSC on September 17, 2019. For this capital increase for cash, the 10,200,000 common shares were issued with an offering price of NT$82 (in dollars) (US$2.74 dollars) per share. The totalpaid-in capital is NT$836,400 thousand (US$27,964 thousand), which has been fully collected. The Company set the record date of the capital increase on October 24, 2019. |
| D.
E. | Employee restricted stocks |
| (a) | (a)
| TheAs 2,000 shares of employee restricted stocks granted to certain employees did not meet the vesting conditions in accordance with the terms of restricted stocks in March 2017, the Board of Directors during itsresolved on May 11, 2017 to buy back the restricted stocks to retire for capital reduction. The transaction was completed on June 23, 2017.
|
| (b) | As 15,000 shares of employee restricted stocks granted to certain employees did not meet the vesting conditions in accordance with the terms of restricted stocks in July and August 2017, the Board of Directors resolved on August 10, 2017 to buy back the restricted stocks to retire for capital reduction. The transaction was completed on September 7, 2017. |
| (c) | As 14,000 shares of employee restricted stocks granted to certain employees did not meet the vesting conditions in accordance with the terms of restricted stocks in September and November 2017, the Board of Directors resolved on November 1, 2017 to buy back the restricted stocks to retire for capital reduction. The transaction was completed on December 8, 2017. |
| (d) | As 25,000 shares of employee restricted stocks granted to certain employees did not meet the vesting conditions in accordance with the terms of restricted stocks in June and July 2018, the Board of Director resolved on August 1, 2018 to buy back the restricted stocks to retire for capital reduction. The transaction was completed on August 29, 2018. |
| (e) | As 10,000 shares of employee restricted stocks granted to certain employees did not meet the vesting conditions in accordance with the terms of restricted stocks in November 2018, the Board of Directors resolved on October 31, 2018 to buy back the restricted stocks to retire for capital reduction. The transaction was completed on January 3, 2019. |
| (f) | The stockholders at their annual stockholders’ meeting on June 18, 2014May 31, 2017 adopted a resolution to issue employee restricted stocks (see Note 6(13)6(14)) with the effective date set on August 21, 2014November 16, 2017 and November 20, 2014, respectively.July 2, 2018. The subscription price was NT$10is $10 (in dollars) per share. The employee restricted stocks issued are subject to certain restrictions on selling, pledging as collateral, transfer, donation or other methods to dispose before their vesting conditions are met. Other than these restrictions, the rights and obligations of these shares issued are the same as other issued common shares. |
F-36
Table of Content
| (g) | (b)
| As 9,00017,600 shares of employee restricted stocks distributedgranted to certain employees in November 2015 did not meet the vesting conditions in accordance with the terms of restricted stocks in February and April 2019, the Board of Directors has resolved on February 25, 2016May 8, 2019 to buy back the restricted stocks to retire for capital reduction. The registrationtransaction was completed on April 22, 2016.June 25, 2019. |
| (c)
(h) | As 14,00033,000 shares of employee restricted stocks distributedgranted to certain employees in April 2016 did not meet the vesting conditions in accordance with the terms of restricted stocks in July 2019, the Board of Directors has resolved on May 5, 2016November 13, 2019 to buy back the restricted stocks to retire for capital reduction. The registrationtransaction was completed on July 18, 2016.December 25, 2019. |
| (d)
(i) | As 12,0005,000 shares of employee restricted stocks distributedgranted to certain employees in July and August 2016 did not meet the vesting conditions in accordance with the terms of restricted stocks in December 2019, the Board of Directors has resolved on August 11, 2016 to buy back the restricted stock to retire for capital reduction. The registration was completed on October 11, 2016. |
| (e)
| As 2,000 shares of employee restricted stocks granted to certain employees in March 2017 did not meet the vesting conditions in accordance with the terms of restricted stocks, the Board of Directors has resolved on May 11, 2017 to buy back the restricted stocks to retire for capital reduction. The registration was completed on June 23, 2017.
|
| (f)
| As 15,000 shares of employee restricted stocks granted to certain employees in July and August 2017 did not meet the vesting conditions in accordance with the terms of restricted stocks, the Board of Directors has resolved on August 10, 2017 to buy back the restricted stocks to retire for capital reduction. The registration was completed on September 7, 2017.
|
| (g)
| As 14,000 shares of employee restricted stocks granted to certain employees in September and November 2017 did not meet the vesting conditions in accordance with the terms of restricted stocks, the Board of Directors has resolved on November 1, 2017 to buy back the restricted stocks to retire for capital reduction. The registration was completed on December 8, 2017.
|
| (h)
| As 25,000 shares of employee restricted stocks granted to certain employees in June and July 2018 did not meet the vesting conditions in accordance with the terms of restricted stocks, the Board of Directors has resolved on August 1, 201820, 2019 to buy back the restricted stocks to retire for capital reduction. The registrationtransaction was completed on August 29, 2018.February 4, 2020.
|
| (17) | (i)
| As 10,000 shares of employee restricted stocks granted to certain employees in November 2018 did not meet the vesting conditions in accordance with the terms of restricted stocks, the Board of Directors has resolved on October 31, 2018 to buy back the restricted stocks to retire for capital reduction. The registration was completed on January 3, 2019.Capital surplus
|
| (j)
| The stockholders at their annually stockholders’ meeting on May 31, 2017 adopted a resolution to issue employee restricted stocks (see Note 6(13)) with the effective date set on November 16, 2017 and July 2, 2018. The subscription price is NT$10 (in dollars) (US$0.3 (in dollars)) per share. The employee restricted stocks issued are subject to certain restrictions on selling, pledging as collateral, transfer, donation or other methods to dispose before their vesting conditions are met. Other than these restrictions, the rights and obligations of these shares issued are the same as other issued common shares.
|
| (a)
| Reason for share repurchase and movements in the number of the Company’s treasury stock are as follows:
|
| | 2016 | | Reason for repurchase | | January 1 | | | Additions | | | Disposal | | | December 31 | | To be reissued to employees | | | | | | | | | | | | | | | | | —Number of shares (in thousands) | | | 276 | | | | — | | | | (276 | ) | | | — | | —Carrying amount (NT$000) | | $ | 36,893 | | | $ | — | | | $ | (36,893 | ) | | $ | — | |
For the year ended December 31, 2017 and 2018: None.
F-37
Table of Content
| (b)
| Pursuant to the R.O.C. Securities and Exchange Act, the number of shares repurchased as treasury stocks should not exceed 10% of the number of the Company’s issued and outstanding shares and the amount repurchased should not exceed the sum of retained earnings, paid-in capital in excess of par value and realized capital surplus.
|
| (c)
| Pursuant to the R.O.C. Securities and Exchange Act, treasury stocks should not be pledged as collateral and is not entitled to dividends before it is reissued.
|
| (d)
| Pursuant to the R.O.C. Securities and Exchange Act, treasury stocks should be reissued to the employees within three years from the repurchase date and shares not reissued within the three-year period are to be retired.
|
Pursuant to the R.O.C. Company Act, capital reservesurplus arising frompaid-in capital in excess of par value on issuance of common stocksstock and donations can be used to cover accumulated deficit or to issue new stocks or cash to shareholders in proportion to their share ownership, provided that the Company has no accumulated deficit. Further, the R.O.C. Securities and Exchange Act requires that the amount of capital reservesurplus to be capitalized mentioned above should not exceed 10% of thepaid-in capital each year. Capital surplus should not be used to cover accumulated deficit unless the legal reserve is insufficient. | (18) | (17)
| Retained earnings / accumulated deficit |
| A. | Under the Company’s Articles of Incorporation, the current earnings, if any, shall be distributed in the following order: |
| (a) | Payment of taxes and duties.duties; |
| (b) | Cover prior years’ accumulated deficit, if any.any; |
| (c) | (c)
| After deducting items a and b, set aside 10% of the remaining amount as legal reserve.reserve; |
| (d) | Appropriate or reverse special reserve in accordance with the relevant laws and regulations, if necessary; |
| (e) | After deducting items a to d, the remainder, if any, to be retained or to be appropriated shall be resolved by the shareholders at the shareholders’ meeting. |
| B. | The Company’s dividend policy is summarized below: |
As the Company operates in a volatile business environment and is in the growth stage, the residual dividend policy is adopted taking into consideration the Company’s financial structure, operating results and future expansion plans. According to the dividend policy adopted by the Board of Directors, cash dividends shall account for at least 10% of the total dividends distributed. | C. | Under the R.O.C. Company Act, when the accumulated deficit exceeds 50% of the capital, the directors should convene a meeting of the shareholders and report the situation. |
| D. | The shareholders during their meetings on June 21, 2016, May 31, 2017 and June 26, 2018 adopted a resolution to use capital surplus amounting to NT$673,652 thousand, NT$824,662 thousand and NT$874,086 thousand (US$28,555 thousand) to cover accumulated deficit, respectively. |
| E. | As of December 31, 20172018 and 2018,2019, the Company had an accumulated deficit.deficits. Therefore, the earnings distribution information disclosure is not applicable. |
| F. | For the information relating to employees’ compensation and directors’ and supervisors’ remuneration, please refer to Note 6(23)6(24). |
| (19) | (18)
| Operating revenue |
| Years ended December 31, 2018 | | | | | NT$000 | | | US$000 | | Revenue from contracts with customers | | | $ | 62,324 | | | $ | 2,036 | |
| | | | | | | | | | | | | | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | US$000 | | Revenue from contracts with customers | | $ | 62,324 | | | $ | 209,140 | | | $ | 6,992 | | | | | | | | | | | | | | |
F-38
Table of Content
| A. | A.
| Disaggregation of revenue from contracts with customers |
The Group derives revenue from the transfer of goods and services over time and at a point in time in the following types: | | | | | | | | | | | | | | | | | 2018 | | Royalty
revenue | | | Authorization
collaboration and
development revenue | | | Other | | | Total | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | Timing of revenue recognition | | | | | | | | | | | | | | | | | At a point in time | | $ | 52,100 | | | $ | — | | | $ | — | | | $ | 52,100 | | Over time | | | — | | | | 10,224 | | | | — | | | | 10,224 | | | | | | | | | | | | | | | | | | | Total | | $ | 52,100 | | | $ | 10,224 | | | $ | — | | | $ | 62,324 | | | | | | | | | | | | | | | | | | |
Year ended December 31, 2018 | | Royalty Revenue | | | Authorization collaboration and development revenue | | | Total | | | | 2019 | | | Royalty revenue | | | Authorization
collaboration and
development revenue | | | Other | | | Total | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | Timing of revenue recognition | | | | | | | | | | | | | | | | | | | | | At a point in time | | $ | 52,100 | | | $ | — | | | $ | 52,100 | | | $ | 58,970 | | | $ | 149,056 | | | $ | 304 | | | $ | 208,330 | | over time | | | — | | | | 10,224 | | | | 10,224 | | | Over time | | | | — | | | | — | | | | 810 | | | | 810 | | | | | | | | | | | | | | | | Total | | $ | 52,100 | | | $ | 10,224 | | | $ | 62,324 | | | $ | 58,970 | | | $ | 149,056 | | | $ | 1,114 | | | $ | 209,140 | | | | | | | | | | | | | | | | Total (US$000) | | $ | 1,702 | | | 334 | | | $ | 2,036 | | | $ | 1,972 | | | $ | 4,983 | | | $ | 37 | | | $ | 6,992 | | | | | | | | | | | | | | | |
| B. | Contract assets and liabilities |
The Group has recognized the following revenue-related contract liabilitiesassets and assets:liabilities: | December 31 | | | 2017 (Note) | | | 2018 | | | NT$000 | | | NT$000 | | | US$000 | | Contract liabilities – authorization collaboration and development revenue | $ | 7,941 | | | | | | | | | | Contract assets- authorization collaboration and development revenue | | | | | $ | 2,283 | | | $ | 75 | |
| | | | | | | | | | | | | | | | | | | | January 1, | | | December 31, | | | | | 2018 | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Contract assets - authorization collaboration and development revenue | | $ | — | | | $ | 2,283 | | | $ | — | | | $ | — | | | | | | | | | | | | | | | | | | | Non-contract liabilities - authorization collaboration and development revenue | | $ | — | | | $ | — | | | $ | 10,760 | | | $ | 360 | | | | | | | | | | | | | | | | | | |
Note: Please refer to Note 3(1)(b) for the reclassified amount.
| (a) | Revenue recognized that was included in the contract liability balance at the beginning of the year:year. |
| | | December 31, 2018 | | | 2018 | | | 2019 | | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | US$000 | | Revenue recognized that was included in the contract liability balance at the beginning of the year | | | | | | | | | | | | | | | Authorization collaboration and development contracts | | $ | 7,941 | | | $ | 259 | | | $ | 7,941 | | | $ | — | | | $ | — | | | | | | | | | | | | |
| (b) | Unfulfilled long-term authorization collaboration and development contracts |
Aggregate amount of the transaction price allocated to long-term authorization collaboration and development contracts that are partially unsatisfied as of December 31, 2018 and 2019 amounted to NT$37,716 thousand and NT$10,760 thousand (US$1,232 360 thousand)., respectively. The above transaction prices disclosed do not include any estimated amounts of variable consideration that are constrained. Considering the progress of research and development, management expects that the transaction price allocated to the unsatisfied contracts amounting to NT$37,716 thousand and NT$10,760 thousand (US$1,232 360 thousand), as of December 31, 2018 and 2019, respectively, will be recognized as revenue in the next three years, which is subject to the management’s plan in terms of completion of clinical and bioequivalence study. | C. | C.
| Related disclosures in relation to operating revenue for the year ended December 31, 2017 are provided in Note 12(5)B.
|
F-39
Table of Content
| D.
| Authorization collaboration and development revenue is the revenue arising from authorization collaboration and development of generic drugs. The details are as follows: |
| (a) | In December 2013, the Company entered into a license and collaboration agreement with Sandoz AG. (“Sandoz”) for the development and commercialization of products in Europe and in the USA. Under the terms of the agreement, the Company received an upfront,non-refundable payment of US$100 thousand in January 2014, and aggregated milestone payments of US$2,275 thousand related to the Europe submission in March 2014, April 2014, and January 2015, respectively. |
| | Once the new drug is launched in the market, a royalty fee will be received by the Company, which is equal to an agreed upon percentage of net sales. The Company is also eligible to receive performance-based milestone payment upon entering the local market in Europe and in the USA, and upon achieving a certain net sales volume and market position within five years. |
| | The Company is in negotiation with Sandoz in relation to the termination of the above agreement. |
| (b) | In March 2019, the Company entered into a commercialization agreement with Hong Kong Sansheng Medical Limited (“3SBio”) to commercialize two liposomal products utilizing the Company’s NanoX technology platform in mainland China, excluding Hong Kong and Macau. Under the terms of the agreement, the Company received an upfront,non-refundable payment in March 2019, and the Company may receive development and sales milestones for a total of up to US$25 million. In addition, the Company is also eligible to receive double-digit profit shares from the potential sales of products. |
| (c) | In June 2019, the Company entered into a development and license agreement with Birdie Biopharmaceuticals, Inc. (“Birdie”). Under the terms of the development and license agreement, the Company granted Birdie the right to use its intellectual property of NanoX technology to develop and commercialize Birdie’s liposomal formulated dual agonist product. Birdie will be responsible for the product’s preclinical and clinical development, regulatory filing and commercialization. The Company will be assisting with Birdie in the formulation development and manufacturing process of Birdie’s product. Birdie will fund all the development and manufacturing costs. The Company is eligible to receive upfront payment and future milestone payments up to US$49 million. Once the new drug is launched in the market, Birdie will pay the Company a royalty payment based on a certain percentage of the net sales. |
In December 2013, the Company entered into a license and collaboration agreement with Sandoz AG. (“Sandoz”) for the development and commercialization of products in Europe and in the USA. Under the terms of the agreement, the Company received an upfront, non-refundable payment of US$0.1 million in January 2014, and aggregated milestone payments of US$2.275 million related to the Europe submission in March and April 2014, and January 2015, respectively.
Once the new drug is launched in the market, a royalty fee will be received by the Company, which is equal to an agreed upon percentage of net sales. The Company is also eligible to receive performance-based milestone payment upon entering the local market in Europe and in the USA, and upon achieving a certain net sales volume and market position within five years.
| E.
D. | The details of royalty revenue are as follows: |
| (a) | The Company granted TTY Biopharm Company Limited (“TTY”) the exclusive right in Taiwan to produce and promoteLIPO-DOX, a medicinal product developed by the Company. Under the agreement,contract, royalty payments are based on 12% of the net product sales. |
| (b) | The Company authorizes Yung Shin Pharm. Ind. Co., Ltd. (“YSP”) the exclusive right in Taiwan to produce and promote generic drugs. Under the agreement,contract, the Company will receive a royalty payment based on a certain percentage of the net sales. |
| (20) | (19)
| Other income and expenses |
| | Years ended December 31, | | | | | | | | | | | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Government subsidy income (Note 1 and 2) | | $ | 1,810 | | | $ | 14,206 | | | $ | 21,100 | | | $ | 689 | | | $ | 14,206 | | | $ | 21,100 | | | $ | 1,420 | | | $ | 47 | | Others | | | 3,765 | | | | 6,942 | | | | 5,128 | | | | 168 | | | | 6,942 | | | | 5,128 | | | | 14,629 | | | | 489 | | | | $ | 5,575 | | | $ | 21,148 | | | $ | 26,228 | | | $ | 857 | | | | | | | | | | | | | | | | | $ | 21,148 | | | $ | 26,228 | | | $ | 16,049 | | | $ | 536 | | | | | | | | | | | | | | | |
Note 1: The Company has entered into contracts of “A phase IIa trial of lipid-based investigational drug TLC399 in the subjects with macular edema due to retinal vein occlusion in the United States” and “A phase I/II trial of lipid-based, sustained release investigational drug TLC399 (ProDex®) for treating macular edema due to retinal vein occlusion” with the Institute for Information Industry in 2017 and 2014, respectively. The Company has accrued government subsidy income in accordance with the progress of the plans. The aforesaid subsidy plans has recognized income of NT$1,810 thousand, NT$14,206 thousand and NT$7,615 thousand (US$249 thousand) for the years ended December 31, 2016, 2017 and 2018, respectively. As of December 31, 2018, the Company has not received the government subsidy of NT$2,661 thousand (US$87
| Note 1: | The Company has entered into contracts of “A phase IIa trial of lipid-based investigational drug TLC399 in the subjects with macular edema due to retinal vein occlusion in the United States” and “A phase I/II trial of lipid-based, sustained release investigational drug TLC399 (ProDex®) for treating macular edema due to retinal vein occlusion” with the Institute for Information Industry in 2017 and 2014, respectively. The Company has accrued government subsidy income in accordance with the progress of the plans. The aforesaid subsidy plans has recognized income of NT$14,206 thousand, NT$7,615 thousand and NT$1,420 thousand (US$47 thousand) for 2017, 2018 and 2019, respectively. As of December 31, 2019, the Company has not received the government subsidy of NT$1,420 thousand (US$47 thousand) (shown as “Other receivables”). |
Note 2: The Company’s subsidiary, TLC Biopharmaceuticals Pty Ltd. received the financial incentives from Australian government in August 2018 of NT$13,485 thousand (US$441 thousand) for its research and development activities.
| Note 2: | The Company’s subsidiary, TLC Biopharmaceuticals Pty Ltd. received the financial incentives from Australian government in August 2018 of NT$13,485 thousand for its research and development activities. |
| (21) | (20)
| Other gains and losses |
| | Years ended December 31, | | | | | | | | | | | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Net currency exchange gain (loss) | | $ | 417 | | | $ | 2,632 | | | $ | (2,986 | ) | | $ | (97 | ) | | $ | 2,632 | | | ($ | 2,986 | ) | | $ | 14,515 | | | $ | 486 | | Gain on disposal of property, plant and equipment | | | — | | | | 20 | | | | 1,478 | | | | 48 | | | Other gains (losses) | | | | 20 | | | | 1,478 | | | | (53 | ) | | | (2 | ) | | | $ | 417 | | | $ | 2,652 | | | $ | (1,508 | ) | | $ | (49 | ) | | | | | | | | | | | | | | | | $ | 2,652 | | | ($ | 1,508 | ) | | $ | 14,462 | | | $ | 484 | | | | | | | | | | | | | | | |
F-40
Table of Content
| | Years ended December 31, | | | | | | | | | | | | | | | | | | | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Bank borrowings | | $ | 2,376 | | | $ | 2,255 | | | $ | 9,379 | | | $ | 306 | | | $ | 2,255 | | | $ | 9,379 | | | $ | 21,333 | | | $ | 713 | | Financial lease liabilities | | | 564 | | | | 1,130 | | | | 507 | | | | 17 | | | Finance lease liabilities | | | | 1,130 | | | | 507 | | | | — | | | | — | | Lease liabilities | | | | — | | | | — | | | | 2,323 | | | | 78 | | | | $ | 2,940 | | | $ | 3,385 | | | $ | 9,886 | | | $ | 323 | | | | | | | | | | | | | | | | | $ | 3,385 | | | $ | 9,886 | | | $ | 23,656 | | | $ | 791 | | | | | | | | | | | | | | | |
| (23) | (22)
| Expenses by nature (Shown as operating expenses)“Operating expenses”) |
| | Years ended December 31, | | | | | | | | | | | | | | | | | | | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Employee benefit expenses | | $ | 346,111 | | | $ | 323,991 | | | $ | 314,655 | | | $ | 10,280 | | | $ | 323,991 | | | $ | 314,655 | | | $ | 268,381 | | | $ | 8,973 | | | | | | | | | | | | | | | | Depreciation charges | | $ | 63,571 | | | $ | 41,926 | | | $ | 39,315 | | | $ | 1,284 | | | $ | 41,926 | | | $ | 39,315 | | | $ | 64,754 | | | $ | 2,165 | | | | | | | | | | | | | | | | Amortization charges | | $ | 11,668 | | | $ | 10,570 | | | $ | 8,144 | | | $ | 266 | | | $ | 10,570 | | | $ | 8,144 | | | $ | 6,648 | | | $ | 222 | | | | | | | | | | | | | | | |
| (24) | (23)
| Employee benefit expenses |
| | Years ended December 31, | | | | | | | | | | | | | | | | | | | | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Wages and salaries | | $ | 207,591 | | | $ | 225,633 | | | $ | 234,735 | | | $ | 7,669 | | | $ | 225,633 | | | $ | 234,735 | | | $ | 210,140 | | | $ | 7,026 | | Share-based payment compensation costs | | | 100,573 | | | | 57,149 | | | | 41,386 | | | | 1,352 | | | | 57,149 | | | | 41,386 | | | | 26,793 | | | | 896 | | Labor and health insurance fees | | | 17,194 | | | | 19,117 | | | | 19,026 | | | | 621 | | | | 19,117 | | | | 19,026 | | | | 16,177 | | | | 541 | | Pension costs | | | 9,369 | | | | 10,245 | | | | 10,396 | | | | 340 | | | | 10,245 | | | | 10,396 | | | | 8,356 | | | | 279 | | Other personnel expenses | | | 11,384 | | | | 11,847 | | | | 9,112 | | | | 298 | | | | 11,847 | | | | 9,112 | | | | 6,915 | | | | 231 | | | | $ | 346,111 | | | $ | 323,991 | | | $ | 314,655 | | | $ | 10,280 | | | | | | | | | | | | | | | | | $ | 323,991 | | | $ | 314,655 | | | $ | 268,381 | | | $ | 8,973 | | | | | | | | | | | | | | | |
According to the Articles of Incorporation of the Company, a ratio of distributable profit of the current year, after covering accumulated deficit, shall be distributed as employees’ compensation and directors’ and supervisors’ remuneration. The ratio shall be 2%~8% for employees’ compensation and shall not be higher than 2% for directors’ remuneration. As of December 31, 2016, 2017, 2018 and 2018,2019, the Company had accumulated deficits and did not accrue employees’ compensation and directors’ and supervisors’ remuneration. | A. | Components of income tax expense: |
| | | | | | | | | | | | | | | | | | | | 2017 | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Current income tax: | | | | | | | | | | | | | | | | | Current income tax on profits for the year | | $ | 735 | | | $ | 538 | | | $ | 906 | | | $ | 30 | | Prior year income tax underestimation | | | 133 | | | | 329 | | | | 3,214 | | | | 108 | | | | | | | | | | | | | | | | | | | Total current income tax | | | 868 | | | | 867 | | | | 4,120 | | | | 138 | | | | | | | | | | | | | | | | | | | Deferred income tax: | | | | | | | | | | | | | | | | | Origination and reversal of temporary differences | | | 83 | | | | — | | | | — | | | | — | | | | | | | | | | | | | | | | | | | | | $ | 951 | | | $ | 867 | | | $ | 4,120 | | | $ | 138 | | | | | | | | | | | | | | | | | | |
| | Years ended December 31, | | | | 2016 | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Current income tax: | | | | | | | | | | | | | | | | | Current income tax on profits for the year | | $ | 535 | | | $ | 735 | | | $ | 538 | | | $ | 17 | | Prior year income tax underestimation | | | 117 | | | | 133 | | | | 329 | | | | 11 | | Total current income tax | | | 652 | | | | 868 | | | | 867 | | | | 28 | | Deferred income tax: | | | | | | | | | | | | | | | | | Origination and reversal of temporary differences | | | (89 | ) | | | 83 | | | | — | | | | — | | | | $ | 563 | | | $ | 951 | | | $ | 867 | | | $ | 28 | |
F-41
Table of Content
| B. | B.
| Reconciliation between income tax expense and accounting profit: |
| | | | | | | | | | | | | | | | | | | | 2017 | | | 2018 | | | 2019 | | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Tax calculated based on profit (loss) before tax and statutory tax rate (Note) | | ($ | 148,412 | ) | | ($ | 180,141 | ) | | ($ | 160,680 | ) | | ($ | 5,372 | ) | Effect of different tax rates in countries in which the Group operates | | | 573 | | | | 300 | | | | 83 | | | | 3 | | Tax effect of amounts which are not (taxable) deductible in calculating taxable income | | | 83 | | | | — | | | | — | | | | — | | Taxable loss not recognized as deferred tax assets | | | 148,574 | | | | 180,379 | | | | 161,503 | | | | 5,400 | | Prior year income tax underestimation | | | 133 | | | | 329 | | | | 3,214 | | | | 107 | | | | | | | | | | | | | | | | | | | Income tax expense | | $ | 951 | | | $ | 867 | | | $ | 4,120 | | | $ | 138 | | | | | | | | | | | | | | | | | | |
| Note: | The basis for computing the applicable tax rate is the rate applicable of 17% in the parent company’s country in 2017, and 20% and 20% in 2018 and 2019, respectively. |
| | Years ended December 31, | | | | 2016 | | | 2017 | | | 2018 | | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Tax calculated based on profit (loss) before tax and statutory tax rate (Note) | | ($ | 140,038 | ) | | ($ | 148,412 | ) | | ($ | 180,141 | ) | | ($ | 5,885 | ) | Effect of different tax rates in countries in which the group operates | | | 439 | | | | 573 | | | | 300 | | | | 10 | | Tax effect of amounts which are not (taxable) deductible in calculating taxable income | | | (89 | ) | | | 83 | | | | — | | | | — | | Taxable loss not recognized as deferred tax assets | | | 140,134 | | | | 148,574 | | | | 180,379 | | | | 5,892 | | Prior year income tax underestimation | | | 117 | | | | 133 | | | | 329 | | | | 11 | | Income tax expense | | $ | 563 | | | $ | 951 | | | $ | 867 | | | $ | 28 | |
Note: The basis for computing the applicable tax rate is the rate applicable of 17% and 17% in the parent company’s country in 2016 and 2017, and 20% in 2018, respectively.
| C. | Amounts of deferred income tax assets or liabilities as a result of temporary differences are as follows: |
| | | | | | | | | | | | | | | | | | | | | | | | 2017 | | | | | January 1 | | | Recognized
in profit or
loss | | | Recognized in
other
comprehensive
income | | | Recognized
in equity | | | December 31 | | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | Temporary differences: | | | | | | | | | | | | | | | | | | | | | —Deferred income tax assets: | | | | | | | | | | | | | | | | | | | | | Unrealized expenses | | $ | 164 | | | ($ | 83 | ) | | $ | — | | | $ | — | | | $ | 81 | | | | | | | | | | | | | | | | | | | | | | |
| | 2016 | | | | | | | | | | | | Reason for reacquisition | | January 1 | | | Recognized in profit or loss | | | Recognized in other comprehensive income | | | Recognized in equity | | | December 31 | | | | | | | 2018 | | | | | | January 1 | | | Recognized
in profit or
loss | | | Recognized in
other
comprehensive
income | | Recognized
in equity | | | December 31 | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | NT$000 | | | NT$000 | | Temporary differences: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | —Deferred income tax assets: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Unrealized expenses | | $ | 75 | | | $ | 89 | | | $ | — | | | $ | — | | | $ | 164 | | | $ | 81 | | | $ | — | | | ($ | 2 | ) | | $ | — | | | $ | 79 | | | | | | | | | | | | | | | | | | |
| | 2017 | | | | | | | | | | | | | | Reason for reacquisition | | January 1 | | | Recognized in profit or loss | | | Recognized in other comprehensive income | | | Recognized in equity | | | December 31 | | | | | | | 2019 | | | | | | January 1 | | | Recognized
in profit or
loss | | | Recognized in
other
comprehensive
income | | Recognized
in equity | | | December 31 | | | December 31 | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | NT$000 | | | NT$000 | | | US$000 | | Temporary differences: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | —Deferred income tax assets: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Unrealized expenses | | $ | 164 | | | ($ | 83 | ) | | $ | — | | | $ | — | | | $ | 81 | | | $ | 79 | | | $ | — | | | ($ | 3 | ) | | $ | — | | | $ | 76 | | | $ | 3 | | | | | | | | | | | | | | | | | | | | | |
| | 2018 | | Reason for reacquisition | | January 1 | | | Recognized in profit or loss | | | Recognized in other comprehensive income | | | Recognized in equity | | | December 31 | | | December 31 | | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | Temporary differences: | | | | | | | | | | | | | | | | | | | | | | | | | —Deferred income tax assets: | | | | | | | | | | | | | | | | | | | | | | | | | Unrealized expenses | | $ | 81 | | | $ | — | | | ($ | 2 | ) | | $ | — | | | $ | 79 | | | $ | 3 | |
F-42
Table of Content
| D. | D.
| Details of investment tax credits and unrecognized deferred tax assets are as follows: |
| | | | | | | | | | | | | | | | December 31, 2018 | | | | | Unused credits | | | Unrecognized deferred
income tax assets | | | Final year tax
credits are due | | | | | NT$000 | | | NT$000 | | | | | Qualifying items | | | | | | | | | | | | | Research and development expenditure | | $ | 358,394 | | | $ | 358,394 | | | | Note | | Employees’ development and training | | | 72 | | | | 72 | | | | Note | |
| | | | | | | | | | | | | | | | December 31, 2019 | | | | | Unused credits | | | Unrecognized deferred
income tax assets | | | Final year tax
credits are due | | | | | NT$000 | | | NT$000 | | | | | Qualifying items | | | | | | | | | | | | | Research and development expenditure | | $ | 549,466 | | | $ | 549,466 | | | | Note | | Employees’ development and training | | | 72 | | | | 72 | | | | Note | |
| | | | | | | | | | | | | | | | December 31, 2019 | | | | | Unused credits | | | Unrecognized deferred
income tax assets | | | Final year tax
credits are due | | | | | US$000 | | | US$000 | | | | | Qualifying items | | | | | | | | | | | | | Research and development expenditure | | $ | 18,371 | | | $ | 18,371 | | | | Note | | Employees’ development and training | | | 2 | | | | 2 | | | | Note | |
| Note: | In accordance with the Ministry of Economic Affairs (MOEA)Jing-Shou-Gong-Zi Letter No. 10020409420 dated June 10, 2011, Letter No. 10320407210 dated April 3, 2014, and Letter No. 1072042611 dated September 26, 2018, the Company was approved as a biotech pharmaceuticals company. Accordingly, the Company and its shareholders are eligible for investment tax credits under the Statute for Development of Biotech New Pharmaceuticals Industry. Relevant investment tax credits can be used to offset against the Company’s income tax within five years from the year in which the Company starts to have income tax payable. |
| | December 31, 2017 | | | Unused credits | | | Unrecognized deferred income tax assets | | | Final year tax credits are due | | | NT$000 | | | NT$000 | | | | Qualifying items | | | | | | | | | | | Research and development expenditure | | $ | 307,403 | | | $ | 307,403 | | | Note | Employees’ development and training | | | 72 | | | | 72 | | | Note |
| | December 31, 2018 | | | Unused credits | | | Unrecognized deferred income tax assets | | | Final year tax credits are due | | | NT$000 | | | NT$000 | | | | Qualifying items | | | | | | | | | | | Research and development expenditure | | $ | 358,394 | | | $ | 358,394 | | | Note | Employees’ development and training | | | 72 | | | | 72 | | | Note |
| | December 31, 2018 | | | Unused tax credits | | | Unrecognized deferred income tax assets | | | Final year tax credits are due | | | US$000 | | | US$000 | | | | Qualifying items | | | | | | | | | | | Research and development expenditure | | $ | 11,708 | | | $ | 11,708 | | | Note | Employees’ development and training | | | 2 | | | | 2 | | | Note |
Note: In accordance with the Ministry of Economic Affairs (MOEA) Jing-Shou-Gong-Zi Letter No. 10020409420 dated June 10, 2011 and Letter No. 10320407210 dated April 3, 2014, the Company was approved as a biotech pharmaceuticals company. Accordingly, the Company and its shareholders are eligible for investment tax credits under the Statute for Development of Biotech New Pharmaceuticals Industry. Relevant investment tax credits can be used to offset against the Company’s income tax within five years from the year in which the Company starts to have income tax payable.
F-43
Table of Content
| E. | E.
| Expiration dates of unused loss carryforward and amounts of unrecognized deductible amounts of the Company are as follows: |
| | | | | | | | | | | | | | | | | | | | December 31, 2018 | | Year incurred | | Amount filed /
assessed | | | Unused amount | | | Unrecognized
amount (Note) | | | Expiry year | | | | | NT$000 | | | NT$000 | | | NT$000 | | | | | 2009 | | $ | 136,642 | | | $ | 136,642 | | | $ | 136,642 | | | | 2019 | | 2010 | | | 196,215 | | | | 196,215 | | | | 196,215 | | | | 2020 | | 2011 | | | 212,903 | | | | 212,903 | | | | 212,903 | | | | 2021 | | 2012 | | | 187,946 | | | | 187,946 | | | | 187,946 | | | | 2022 | | 2013 | | | 407,816 | | | | 407,816 | | | | 407,816 | | | | 2023 | | 2014 | | | 632,283 | | | | 632,283 | | | | 632,283 | | | | 2024 | | 2015 | | | 649,799 | | | | 649,799 | | | | 649,799 | | | | 2025 | | 2016 | | | 792,388 | | | | 792,388 | | | | 792,388 | | | | 2026 | | 2017 | | | 832,622 | | | | 832,622 | | | | 832,622 | | | | 2027 | | 2018 | | | 918,113 | | | | 918,113 | | | | 918,113 | | | | 2028 | | | | | | | | | | | | | | | | | | | | | $ | 4,966,727 | | | $ | 4,966,727 | | | $ | 4,966,727 | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2017 | Year incurred | | Amount filed / assessed | | | Unused amount | | | Unrecognized amount (Note) | | | Expiry year | | | NT$000 | | | NT$000 | | | NT$000 | | | | 2008 | | $ | 200,442 | | | $ | 200,442 | | | $ | 200,442 | | | 2018 | 2009 | | | 136,642 | | | | 136,642 | | | | 136,642 | | | 2019 | 2010 | | | 196,215 | | | | 196,215 | | | | 196,215 | | | 2020 | 2011 | | | 212,903 | | | | 212,903 | | | | 212,903 | | | 2021 | 2012 | | | 187,946 | | | | 187,946 | | | | 187,946 | | | 2022 | 2013 | | | 407,816 | | | | 407,816 | | | | 407,816 | | | 2023 | 2014 | | | 632,283 | | | | 632,283 | | | | 632,283 | | | 2024 | 2015 | | | 649,799 | | | | 649,799 | | | | 649,799 | | | 2025 | 2016 | | | 792,388 | | | | 792,388 | | | | 792,388 | | | 2026 | 2017 | | | 869,479 | | | | 869,479 | | | | 869,479 | | | 2027 | | | $ | 4,285,913 | | | $ | 4,285,913 | | | $ | 4,285,913 | | | |
| Note: | Unrecognized amount represents unused tax losses for which no deferred income tax asset has been recognized. |
| | | | | | | | | | | | | | | | | | | | December 31, 2019 | | Year incurred | | Amount filed/
assessed | | | Unused amount | | | Unrecognized
amount (Note) | | | Expiry year | | | | | NT$000 | | | NT$000 | | | NT$000 | | | | | 2010 | | $ | 196,215 | | | $ | 196,215 | | | $ | 196,215 | | | | 2020 | | 2011 | | | 212,903 | | | | 212,903 | | | | 212,903 | | | | 2021 | | 2012 | | | 187,946 | | | | 187,946 | | | | 187,946 | | | | 2022 | | 2013 | | | 407,816 | | | | 407,816 | | | | 407,816 | | | | 2023 | | 2014 | | | 632,283 | | | | 632,283 | | | | 632,283 | | | | 2024 | | 2015 | | | 649,799 | | | | 649,799 | | | | 649,799 | | | | 2025 | | 2016 | | | 792,388 | | | | 792,388 | | | | 792,388 | | | | 2026 | | 2017 | | | 832,622 | | | | 832,622 | | | | 832,622 | | | | 2027 | | 2018 | | | 870,584 | | | | 870,584 | | | | 870,584 | | | | 2028 | | 2019 | | | 754,611 | | | | 754,611 | | | | 754,611 | | | | 2029 | | | | | | | | | | | | | | | | | | | | | $ | 5,537,167 | | | $ | 5,537,167 | | | $ | 5,537,167 | | | | | | | | | | | | | | | | | | | | | | |
| Note: | Unrecognized amount represents unused tax losses for which no deferred income tax asset has been recognized. |
| | | | | | | | | | | | | | | | |
| | | December 31, 2019 | |
Year incurred | | Amount filed/
assessed | | | Unused amount | | | Unrecognized
amount
(Note) | | | Expiry year | |
| | | US$000 | | | US$000 | | | US$000 | | | | |
2010 | | $ | 6,560 | | | $ | 6,560 | | | $ | 6,560 | | | | 2020 | |
2011 | | | 7,118 | | | | 7,118 | | | | 7,118 | | | | 2021 | |
2012 | | | 6,284 | | | | 6,284 | | | | 6,284 | | | | 2022 | |
2013 | | | 13,635 | | | | 13,635 | | | | 13,635 | | | | 2023 | |
2014 | | | 21,140 | | | | 21,140 | | | | 21,140 | | | | 2024 | |
2015 | | | 21,725 | | | | 21,725 | | | | 21,725 | | | | 2025 | |
2016 | | | 26,492 | | | | 26,492 | | | | 26,492 | | | | 2026 | |
2017 | | | 27,838 | | | | 27,838 | | | | 27,838 | | | | 2027 | |
2018 | | | 29,107 | | | | 29,107 | | | | 29,107 | | | | 2028 | |
2019 | | | 25,229 | | | | 25,229 | | | | 25,229 | | | | 2029 | |
| | | | | | | | | | | | | | | | |
| | $ | 185,128 | | | $ | 185,128 | | | $ | 185,128 | | | | | |
| | | | | | | | | | | | | | | | |
| | December 31, 2018 |
Year incurred | | Amount filed / assessed | | | Unused amount | | | Unrecognized amount (Note) | | | Expiry year |
| | NT$000 | | | NT$000 | | | NT$000 | | | |
2009 | | $ | 136,642 | | | $ | 136,642 | | | $ | 136,642 | | | 2019 |
2010 | | | 196,215 | | | | 196,215 | | | | 196,215 | | | 2020 |
2011 | | | 212,903 | | | | 212,903 | | | | 212,903 | | | 2021 |
2012 | | | 187,946 | | | | 187,946 | | | | 187,946 | | | 2022 |
2013 | | | 407,816 | | | | 407,816 | | | | 407,816 | | | 2023 |
2014 | | | 632,283 | | | | 632,283 | | | | 632,283 | | | 2024 |
2015 | | | 649,799 | | | | 649,799 | | | | 649,799 | | | 2025 |
2016 | | | 792,388 | | | | 792,388 | | | | 792,388 | | | 2026 |
2017 | | | 832,622 | | | | 832,622 | | | | 832,622 | | | 2027 |
2018 | | | 918,113 | | | | 918,113 | | | | 918,113 | | | 2028 |
| | $ | 4,966,727 | | | $ | 4,966,727 | | | $ | 4,966,727 | | | |
| Note: | Unrecognized amount represents unused tax losses for which no deferred income tax asset has been recognized. |
F-44
Table of Content
| | December 31, 2018 |
Year incurred | | Amount filed / assessed | | | Unused amount | | | Unrecognized amount (Note) | | | Expiry year |
| | US$000 | | | US$000 | | | US$000 | | | |
2009 | | $ | 4,464 | | | $ | 4,464 | | | $ | 4,464 | | | 2019 |
2010 | | | 6,410 | | | | 6,410 | | | | 6,410 | | | 2020 |
2011 | | | 6,955 | | | | 6,955 | | | | 6,955 | | | 2021 |
2012 | | | 6,140 | | | | 6,140 | | | | 6,140 | | | 2022 |
2013 | | | 13,323 | | | | 13,323 | | | | 13,323 | | | 2023 |
2014 | | | 20,656 | | | | 20,656 | | | | 20,656 | | | 2024 |
2015 | | | 21,228 | | | | 21,228 | | | | 21,228 | | | 2025 |
2016 | | | 25,887 | | | | 25,887 | | | | 25,887 | | | 2026 |
2017 | | | 27,201 | | | | 27,201 | | | | 27,201 | | | 2027 |
2018 | | | 29,994 | | | | 29,994 | | | | 29,994 | | | 2028 |
| | $ | 162,258 | | | $ | 162,258 | | | $ | 162,258 | | | |
| Note:
| Unrecognized amount represents unused tax losses for which no deferred income tax asset has been recognized.
|
| F. | The U.S. subsidiary, TLC Biopharmaceuticals, Inc., has not recognized unused tax losses as deferred income tax assets. As of December 31, 20172018 and 2018,2019, the amounts of unused tax losses not recognized as deferred income tax assets are NT$48,73229,984 thousand and NT$1,96828,438 thousand (US$64951 thousand), respectively. |
| G. | The amounts of deductible temporary differences that wereare not recognized as deferred income tax assets are as follows: |
| | December 31, | |
| | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | |
Deductible temporary differences | | $ | 104,865 | | | $ | 85,337 | | | $ | 2,788 | |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
Deductible temporary differences | | $ | 85,337 | | | $ | 84,234 | | | $ | 2,816 | |
| | | | | | | | | | | | |
| H. | The Company’s income tax returns through 20162017 have been assessed and approved by the Tax Authority. |
F-45
Table of Content
| | | Year ended December 31, 2016 | | | 2017 | |
| | Amount after tax | | | Weighted average number of common shares outstanding (in thousands of shares) | | | Loss per share (in dollars) | | | Amount
after tax | | | Weighted
average
number
of common
shares
outstanding
(in thousands
of shares) | | | Loss per
share
(in dollars) | |
| | NT$000 | | | | | | | NT$ | | | NT$000 | | | | | | NT$ | |
Basic loss per share | | | | | | | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 824,316 | ) | | | 55,361 | | | ($ | 14.89 | ) | | ($ | 873,962 | ) | | | 55,489 | | | ($ | 15,75 | ) |
| | | | | | | | | |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | | | | | | | |
Employees’ stock options | | | — | | | (Note) | | | | | | | | — | | | | (Note | ) | | |
Restricted stocks | | | — | | | (Note) | | | | | | | | — | | | | (Note | ) | | |
| | | | | | | | | |
Diluted loss per share | | | | | | | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 824,316 | ) | | | 55,361 | | | ($ | 14.89) | | | ($ | 873,962 | ) | | | 55,489 | | | ($ | 15,75 | ) |
| | | | | | | | | | |
| | | | | | | | | | | | |
| | | 2018 | |
| | | Amount
after tax | | | Weighted
average
number
of common
shares
outstanding
(in thousands
of shares) | | | Loss per
share
(in dollars) | |
| | | NT$000 | | | | | | NT$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 901,574 | ) | | | 62,719 | | | ($ | 14.37 | ) |
| | | | | | | | | | | | |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | | (Note | ) | | | | |
Restricted stocks | | | — | | | | (Note | ) | | | | |
| | | | | | | | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 901,574 | ) | | | 62,719 | | | ($ | 14.37 | ) |
| | | | | | | | | | | | |
| | Year ended December 31, 2017 | |
| | Amount after tax | | | Weighted average number of common shares outstanding (in thousands of shares) | | | Loss per share (in dollars) | |
| | NT$000 | | | | | | | NT$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 873,962 | ) | | | 55,489 | | | ($ | 15.75 | ) |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | (Note) | | | | | |
Restricted stocks | | | — | | | (Note) | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 873,962 | ) | | | 55,489 | | | ($ | 15.75 | ) |
F-46
Table of Content
| | | | | | | | | | | | |
| | | 2019 | |
| | | Amount
after tax | | | Weighted
average
number
of common
shares
outstanding
(in thousands
of shares) | | | Loss per
share
(in dollars) | |
| | | NT$000 | | | | | | NT$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 807,522 | ) | | | 65,545 | | | ($ | 12.32 | ) |
| | | | | | | | | | | | |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | | (Note | ) | | | | |
Restricted stocks | | | — | | | | (Note | ) | | | | |
| | | | | | | | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 807,522 | ) | | | 65,545 | | | ($ | 12.32 | ) |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | 2019 | |
| | | Amount
after tax | | | Weighted
average
number
of common
shares
outstanding
(in thousands
of shares) | | | Loss per
share
(in dollars) | |
| | | US$000 | | | | | | US$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 26,999 | ) | | | 65,545 | | | ($ | 0.41 | ) |
| | | | | | | | | | | | |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | | (Note | ) | | | | |
Restricted stocks | | | — | | | | (Note | ) | | | | |
| | | | | | | | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 26,999 | ) | | | 65,545 | | | ($ | 0.41 | ) |
| | | | | | | | | | | | |
| | Year ended December 31, 2018 | |
| | Amount after tax | | | Weighted average number of common shares outstanding (in thousands of shares) | | | Loss per share (in dollars) | |
| | NT$000 | | | | | | | NT$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 901,574 | ) | | | 62,719 | | | ($ | 14.37 | ) |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | (Note) | | | | | |
Restricted stocks | | | — | | | (Note) | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 901,574 | ) | | | 62,719 | | | ($ | 14.37 | ) |
| | Year ended December 31, 2018 | |
| | Amount after tax | | | Weighted average number of common shares outstanding (in thousands of shares) | | | Loss per share (in dollars) | |
| | US$000 | | | | | | | UST$ | |
Basic loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company | | ($ | 29,454 | ) | | | 62,719 | | | ($ | 0.47 | ) |
Dilutive effect of common shares equivalents: | | | | | | | | | | | | |
Employees’ stock options | | | — | | | (Note) | | | | | |
Restricted stocks | | | — | | | (Note) | | | | | |
Diluted loss per share | | | | | | | | | | | | |
Loss attributable to common shareholders of the Company plus assumed conversion of all dilutive potential common shares | | ($ | 29,454 | ) | | | 62,719 | | | ($ | 0.47 | ) |
| Note: | Employee stock options and employee restricted stocks have no dilutive effect for any periodperiods due to the fact that the Company was in loss position for all periods presented. |
F-47
Table of Content
| (27) | (26)
| Cash flow information |
Investing activities with partial cash payments
| | | | | | | | | | | | | | | | |
| | | 2017 | | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | |
Acquisition of property, plant and equipment (including transfers) | | $ | 17,237 | | | $ | 42,213 | | | $ | 5,337 | | | $ | 179 | |
Add: Opening balance of payables on machinery, equipment and intangible assets | | | 1,229 | | | | — | | | | 3,303 | | | | 110 | |
Ending balance of prepayments for equipment | | | 923 | | | | 27,942 | | | | 74,841 | | | | 2,502 | |
Opening balance of prepayments for equipment being transferred to other expenses | | | — | | | | 780 | | | | — | | | | — | |
Opening balance of prepayments for equipment being transferred to intangible assets | | | 227 | | | | — | | | | 243 | | | | 8 | |
Less: Ending balance of payables on machinery, equipment and intangible assets | | | — | | | | (3,303 | ) | | | (190 | ) | | | (6 | ) |
Opening balance of prepayments for equipment | | | (1,483 | ) | | | (923 | ) | | | (27,942 | ) | | | (934 | ) |
| | | | | | | | | | | | | | | | |
Cash paid | | $ | 18,133 | | | $ | 66,709 | | | $ | 55,592 | | | $ | 1,859 | |
| | | | | | | | | | | | | | | | |
| | Years ended December 31, | |
| | 2016 | | | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | |
Acquisition of property, plant and equipment (including transfers) | | $ | 20,497 | | | $ | 17,237 | | | $ | 42,213 | | | $ | 1,379 | |
Add: Opening balance of payables on machinery, equipment, and intangible assets | | | 3,826 | | | | 1,229 | | | | — | | | | — | |
Ending balance of prepayments for equipment | | | 1,483 | | | | 923 | | | | 27,942 | | | | 913 | |
Opening balance of prepayments for equipment being transferred to other expenses | | | — | | | | — | | | | 780 | | | | 25 | |
Opening balance of prepayments for equipment being transferred to intangible assets | | | 71 | | | | 227 | | | | — | | | | — | |
Less: Ending balance of payables on machinery, equipment, and intangible assets | | | (1,229 | ) | | | — | | | | (3,303 | ) | | | (108 | ) |
Opening balance of prepayments for equipment | | | (71 | ) | | | (1,483 | ) | | | (923 | ) | | | (30 | ) |
Provisions | | | (3,150 | ) | | | — | | | | — | | | | — | |
Cash paid | | $ | 21,427 | | | $ | 18,133 | | | $ | 66,709 | | | $ | 2,179 | |
| | | | | | | | | | | | | | | | |
| | | 2017 | | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | |
Acquisition of intangible assets (including transfers) | | $ | 5,933 | | | $ | 3,537 | | | $ | 4,420 | | | $ | 148 | |
Add: Opening balance of payable on machinery, equipment and intangible assets | | | 1,495 | | | | — | | | | 374 | | | | 12 | |
Less: Ending balance of payables on machinery, equipment and intangible assets | | | — | | | | (374 | ) | | | (74 | ) | | | (2 | ) |
Opening balance of prepayments for equipment | | | (227 | ) | | | — | | | | (243 | ) | | | (8 | ) |
| | | | | | | | | | | | | | | | |
Cash paid | | $ | 7,201 | | | $ | 3,163 | | | $ | 4,477 | | | $ | 150 | |
| | | | | | | | | | | | | | | | |
| | Years ended December 31, | |
| | 2016 | | | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | |
Acquisition of intangible assets (including transfers) | | $ | 6,126 | | | $ | 5,933 | | | $ | 3,537 | | | $ | 115 | |
Add: Opening balance of payable on machinery, equipment, and intangible assets | | | 84 | | | | 1,495 | | | | — | | | | — | |
Less: Ending balance of payables on machinery, equipment, and intangible assets | | | (1,495 | ) | | | — | | | | (374 | ) | | | (12 | ) |
Opening balance of prepayments for equipment | | | (71 | ) | | | (227 | ) | | | — | | | | — | |
Cash paid | | $ | 4,644 | | | $ | 7,201 | | | $ | 3,163 | | | $ | 103 | |
F-48
Table of Content
| (28) | (27)
| Changes in liabilities from financing activities |
| | | | | | | | | | | | | | | | |
| | | 2017 | |
| | | Short-term
borrowings | | | Long-term
borrowings
(including
current
portion) | | | Financial
lease
liabilities
(including
current
portion) | | | Liabilities
from
financing
activities | |
| | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
At January 1 | | $ | 46,000 | | | $ | 71,750 | | | $ | 50,500 | | | $ | 168,250 | |
Changes in cash flow from financing activities | | | — | | | | (1,700 | ) | | | 1,500 | | | | (200 | ) |
| | | | | | | | | | | | | | | | |
At December 31 | | $ | 46,000 | | | $ | 70,050 | | | $ | 52,000 | | | $ | 168,050 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | 2018 | |
| | | Short-term
borrowings | | | Long-term
borrowings
(including
current
portion) | | | Financial
lease
liabilities
(including
current
portion) | | | Liabilities
from
financing
activities | |
| | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
At January 1 | | $ | 46,000 | | | $ | 70,050 | | | $ | 52,000 | | | $ | 168,050 | |
Changes in cash flow from financing activities | | | — | | | | 364,107 | | | | (4,000 | ) | | | 360,107 | |
Changes in othernon-cash items | | | — | | | | 600 | | | | — | | | | 600 | |
| | | | | | | | | | | | | | | | |
At December 31 | | $ | 46,000 | | | $ | 434,757 | | | $ | 48,000 | | | $ | 528,757 | |
| | | | | | | | | | | | | | | | |
| | | 2017 | | | 2019 | |
| | Short-term borrowings | | | Long-term borrowings (including current portion) | | | Financial lease liabilities (including current portion) | | | Liabilities from financing activities | | | Short-term
borrowings | | | Long-term
borrowings
(including current portion) | | | Lease liabilities-2019
Financial lease
liabilities-2018
(including current portion) | | | Liabilities from
financing
activities | |
| | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
At January 1 | | $ | 46,000 | | | $ | 71,750 | | | $ | 50,500 | | | $ | 168,250 | | | $ | 46,000 | | | $ | 434,757 | | | $ | 48,000 | | | $ | 528,757 | |
Effect on initial application of IFRS16 (Note) | | | | — | | | | — | | | | 73,021 | | | | 73,021 | |
| | | | | | | | | | | | | |
Adjusted balance at January 1, 2019 | | | | 46,000 | | | | 434,757 | | | | 121,021 | | | | 601,778 | |
Changes in cash flow from financing activities | | | — | | | | (1,700 | ) | | | 1,500 | | | | (200 | ) | | | — | | | | (56,425 | ) | | | (37,778 | ) | | | (94,203 | ) |
Additions | | | | — | | | | — | | | | 1,770 | | | | 1,770 | |
Changes in othernon-cash items | | | | — | | | | (7,679 | ) | | | 7,496 | | | | (183 | ) |
| | | | | | | | | | | | | |
At December 31 | | $ | 46,000 | | | $ | 70,050 | | | $ | 52,000 | | | $ | 168,050 | | | $ | 46,000 | | | $ | 370,653 | | | $ | 92,509 | | | $ | 509,162 | |
| | | | | | | | | | | | | |
At December 31 (US$000) | | | $ | 1,538 | | | $ | 12,392 | | | $ | 3,093 | | | $ | 17,023 | |
| | | | | | | | | | | | | |
| | 2018 | |
| | Short-term borrowings | | | Long-term borrowings (including current portion) | | | Financial lease liabilities (including current portion) | | | Liabilities from financing activities | |
| | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
At January 1 | | $ | 46,000 | | | $ | 70,050 | | | $ | 52,000 | | | $ | 168,050 | |
Changes in cash flow from financing activities | | | — | | | | 364,107 | | | | (4,000 | ) | | | 360,107 | |
Net exchange differences | | | — | | | | 600 | | | | — | | | | 600 | |
At December 31 | | $ | 46,000 | | | $ | 434,757 | | | $ | 48,000 | | | $ | 528,757 | |
At December 31 (US$000) | | $ | 1,503 | | | $ | 14,203 | | | $ | 1,568 | | | $ | 17,274 | |
| Note: | Please refer to Note 3(1) for the initial application of IFRS 16. |
7. | RELATED PARTY TRANSACTIONS |
| (1) | (1)
| Names of related parties and relationship |
| | |
Names of related parties | | Relationship with the Group |
Keelung Hong | | The Group'sGroup’s Chairman |
George Yeh | | The Group’s General Manager |
| (2) | | The Group's General Manager
|
| (2)
| Significant transactions and balances with related parties |
| A. | The Company’s Chairman provided guarantees for the Company’s long-term and short-term borrowings with Taiwan Cooperative Bank. |
| B. | The Company’s Chairman and General Manager provided guarantees for the Company’s short-term borrowings with E.SUN Commercial Bank. |
| C. | The Company’s Chairman provided guarantees for the Company’s short-term borrowings with Taishin International Bank. |
| C.
D. | The Company’s Chairman provided guarantees to Taiwan Cooperative Bank for the Company’s government grant in relation to the research and development program from the Institute of Information Industry. |
As of December 31, 20172018 and 2018,2019, details of loans are described in Notes 6(8)6(9) and 6(10)6(11).
F-49
Table of Content
| (3) | (3)
| Key management personnel compensation |
| | Years ended December 31, | | | | | | | | | |
| | 2016 | | | 2017 | | | 2018 | | | 2017 | | | 2018 | | | 2019 | |
| | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | US$000 | |
Salaries and other short-term employee benefits | | $ | 26,810 | | | $ | 33,072 | | | $ | 38,227 | | | $ | 1,249 | | | $ | 33,072 | | | $ | 38,227 | | | $ | 38,684 | | | $ | 1,293 | |
Post-employment benefits | | | 350 | | | | 432 | | | | 486 | | | | 16 | | | | 432 | | | | 486 | | | | 522 | | | | 18 | |
Share-based payments | | | 11,823 | | | | 8,120 | | | | 9,366 | | | | 306 | | | | 8,120 | | | | 9,366 | | | | 6,264 | | | | 209 | |
| | $ | 38,983 | | | $ | 41,624 | | | $ | 48,079 | | | $ | 1,571 | | | | | | | | | | | | | |
| | | $ | 41,624 | | | $ | 48,079 | | | $ | 45,470 | | | $ | 1,520 | |
| | | | | | | | | | | | | |
(1)
| | December 31, | | | |
Assets pledged | | 2017 | | | 2018 | | | Pledge purpose |
| | NT$000 | | | NT$000 | | | US$000 | | | |
Shown as “Property, plant and equipment” | | | | | | | | | | | | | | |
Land | | $ | 14,962 | | | $ | 14,962 | | | $ | 489 | | | Note |
Buildings | | | 24,172 | | | | 23,516 | | | | 768 | | | Note |
| | $ | 39,134 | | | $ | 38,478 | | | $ | 1,257 | | | |
Note: Provided as collateral for borrowings.
| | | | | | | | | | | | | | | | |
| | | December 31, | | | | |
Assets pledged | | 2018 | | | 2019 | | | Pledge purpose | |
| | | NT$000 | | | NT$000 | | | US$000 | | | | |
Shown as “Property, plant and equipment” | | | | | | | | | | | | | | | | |
Land | | $ | 14,962 | | | $ | 14,962 | | | $ | 500 | | | | Note | |
Buildings | | | 23,516 | | | | 22,860 | | | | 764 | | | | Note | |
| | | | | | | | | | | | | | | | |
| | $ | 38,478 | | | $ | 37,822 | | | $ | 1,264 | | | | | |
| | | | | | | | | | | | | | | | |
| Note: | Provided as collateral for long-term borrowings with Taiwan Cooperative Bank. |
| (2) | Pursuant to the loan and security agreement entered between Cathay Bank and the Company and its subsidiary, TLC US, on December 27, 2018, except for the Intellectual Property, including trademarks, patents, copyrights, servicemarks, technology, trade secrets, and etc., defined in the loan agreement, all other personal property, including tangible and intangible assets, of the Company and its subsidiary, TLC US are pledged as collateral for borrowings. Cathay Bank constitutes a first priority security interest in the personal property of the Company and TLC US located in the United States and does not constitute a first priority security interest in the personal property of the Company located outside of the United States. |
9. | SIGNIFICANT CONTINGENT LIABILITIES AND UNRECOGNIZED CONTRACT COMMITMENTS |
Under a certain special generic product agreements, the Company is required to have a certain market supply capacity before the launch of the products in the market. Otherwise, the Company is obligated to pay a certain amount as compensation.
In addition to the commitment mentioned in Note 6(10)6(11)A and B, the Group’s significant commitments are as follows:
| A. | Capital expenditures contracted for at the balance sheet date but not yet incurred and are cancelable without cause are as follows: |
| | December 31, | |
| | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | |
Property, plant and equipment | | $ | 1,781 | | | $ | 11,037 | | | $ | 361 | |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
Property, plant and equipment | | $ | 11,037 | | | $ | 9,109 | | | $ | 305 | |
| | | | | | | | | | | | |
F-50
Table of Content
| B. | B.
| Operating lease commitments |
2018
The Group leases offices with lease terms between 1 and 5 years, and the majority of lease agreements are renewable at the end of the lease terms at market rate. The future aggregate minimum lease payments are as follows:
| | December 31, | | | |
| | 2017 | | | 2018 | | | December 31, 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | |
Not later than one year | | $ | 32,999 | | | $ | 31,787 | | | $ | 1,038 | | | $ | 31,787 | |
Later than one year but not later than five years | | | 75,450 | | | | 46,315 | | | | 1,513 | | | | 46,315 | |
| | $ | 108,449 | | | $ | 78,102 | | | $ | 2,551 | | | | |
| | | $ | 78,102 | |
| | | | |
| C. | The Company has outstanding commitments on purchase agreements for the research and manufacturing of medicines which are cancelable without cause as follows: |
| | December 31, | |
| | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | |
| | $ | 31,577 | | | $ | 120,707 | | | $ | 3,943 | |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
| | $ | 120,707 | | | $ | 130,089 | | | $ | 4,349 | |
| | | | | | | | | | | | |
| D. | The Company has outstanding commitments on research and development which are cancelable without cause as follows: |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
| | $ | 603,178 | | | $ | 1,311,875 | | | $ | 43,861 | |
| | | | | | | | | | | | |
| | December 31, | |
| | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | |
| | $ | 478,977 | | | $ | 603,178 | | | $ | 19,705 | |
| E. | The Company has signed a licensing agreement for technology transition with TWI Pharmaceuticals, Inc. with maximum royalty charges of US$5,000 thousand according to the R&D achievement.achievement on October 1, 2013. Once the new drug is launched in the market, the Company will pay a royalty fee based on a certain percentage of the net product sales. |
| F. | The Company’s subsidiary entered into a synthesis technology of novel camptothecin derivative transfer agreement with Sutter West Bay Hospitals (SWBH, formerly, California Pacific Medical Center). on June 24, 2005. Under the agreement, SWBH charges the Company’s subsidiary a patent usage fee of US$10 thousand per annum, royalty fees up to US$300 thousand according to the R&D achievement and royalty fees to a certain percentage of relevant product sales volume. Through December 31, 2018,2019, the Company’s subsidiary has paid US$100 thousand. |
10. | SIGNIFICANT DISASTER LOSS |
None.
F-51
Table of Content
11. | EVENTS AFTER THE REPORTING PERIOD |
A. On March 1,December 20, 2019, the Company entered into a commercialization agreement with Hong Kong Sansheng Medical Limited (“3SBio”)Company’s Board of Directors resolved to commercialize two liposomal products utilizingincrease the Company’s NanoX technology platform in mainland China, excluding Hong Kong and Macau (the “Territory”). Undercapital by issuing within 30,000,000 common shares with par value of NT$10 (in dollars) (US$0.33 dollar) per share. The proposal for issuance of common shares was approved by the terms ofCompany’s shareholders at the agreement, the Company has received an upfront payment in March 2019 and is eligible to receive further development and sales milestones for a total of up to US$25 million. In addition, the Company is also eligible to receive double-digit profit shares from the potential sales of products. The Company and 3SBio will be jointly responsible for obtaining the regulatory approvals for the products in the Territory. The Company will manufacture the productsCompany’s extraordinary general meeting on its own cost. 3SBio will be responsible to import, promote, market, sell and distribute the products in the Territory at its own responsibilities and expenses.February 11, 2020.
B. On April 25, 2019, the Company entered into an amendment to the loan and security agreement with Cathay Bank. Please refer to Note 6(10) for details.
| (1) | (1)
| Capital risk management |
The Group’s objectives when managing capital are to safeguard the Group’s ability to continue as a going concern in order to provide returns for shareholders and to maintain an optimal capital structure to reduce the cost of capital. In order to improve the Group’s capital structure, the Group may issue new shares or sell assets to reduce debt ratio. The Group monitors capital on the basis of the gearingdebt ratio. This ratio is calculated as total debt divided by total capital.
As of December 31, 20172018 and 2018,2019, the Group’s gearing ratios are as follows:
| | | December 31, | | | December 31, | |
| | 2017 | | | 2018 | | | 2018 | | 2019 | |
| | NT$000 | | | NT$000 | | | US$000 | | | NT$000 | | NT$000 | | US$000 | |
Total debt | | $ | 275,255 | | | $ | 748,725 | | | $ | 24,460 | | | $ | 748,725 | | | $ | 664,068 | | | $ | 22,202 | |
| | | | | | | | | | |
Total capital | | $ | 561,990 | | | $ | 640,451 | | | $ | 20,923 | | | $ | 640,451 | | | $ | 741,939 | | | $ | 24,806 | |
| | | | | | | | | | |
Debt ratio | | | 48.98 | % | | | 116.91 | % | | | 116.91 | % | | | 116.91 | % | | 89.50 | % | | 89.50 | % |
| | | | | | | | | | |
F-52
Table of Content
| (2) | (2)
| Financial instruments |
| A. | Financial instruments by category |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
Financial assets | | | | | | | | | | | | |
Financial assets at amortized cost/ loans and receivables | | | | | | | | | | | | |
Cash and cash equivalents | | $ | 807,484 | | | $ | 1,023,874 | | | $ | 34,232 | |
Current financial assets at amortized cost | | | 307,150 | | | | — | | | | — | |
Accounts receivables, net | | | 9,343 | | | | 15,120 | | | | 506 | |
Other receivables | | | 5,811 | | | | 4,654 | | | | 156 | |
Refundable deposits | | | 18,930 | | | | 24,351 | | | | 814 | |
| | | | | | | | | | | | |
| | $ | 1,148,718 | | | $ | 1,067,999 | | | $ | 35,708 | |
| | | | | | | | | | | | |
| | December 31, | |
| | 2017 | | | 2018 | |
| | NT$000 | | | NT$000 | | | US$000 | |
Financial assets | | | | | | | | | | | | |
Financial assets at amortized cost/ loans and receivables | | | | | | | | | | | | |
Cash and cash equivalents | | $ | 951,713 | | | $ | 807,484 | | | $ | 26,380 | |
Current financial assets at amortized cost | | | — | | | | 307,150 | | | | 10,034 | |
Accounts receivables, net | | | 8,622 | | | | 9,343 | | | | 305 | |
Other receivables | | | 19,726 | | | | 5,811 | | | | 190 | |
Refundable deposits | | | 27,188 | | | | 18,930 | | | | 618 | |
| | $ | 1,007,249 | | | $ | 1,148,718 | | | $ | 37,527 | |
Financial liabilities | | | | | | | | | | | | |
Financial liabilities at amortized cost | | | | | | | | | | | | |
Short-term borrowings | | $ | 46,000 | | | $ | 46,000 | | | $ | 1,503 | |
Other payables | | | 93,541 | | | | 206,268 | | | | 6,739 | |
Financial lease liabilities (including current portion) | | | 52,000 | | | | 48,000 | | | | 1,568 | |
Long-term borrowings (including current portion) | | | 70,050 | | | | 434,757 | | | | 14,203 | |
| | $ | 261,591 | | | $ | 735,025 | | | $ | 24,013 | |
| | | | | | | | | | | | |
| | | December 31, | |
| | | 2018 | | | 2019 | |
| | | NT$000 | | | NT$000 | | | US$000 | |
Financial liabilities | | | | | | | | | | | | |
Financial liabilities at amortized cost | | | | | | | | | | | | |
Short-term borrowings | | $ | 46,000 | | | $ | 46,000 | | | $ | 1,538 | |
Other payables | | | 206,268 | | | | 131,064 | | | | 4,382 | |
Financial lease liabilities (including current portion) | | | 48,000 | | | | — | | | | — | |
Lease liabilities (including current portion) | | | — | | | | 92,509 | | | | 3,093 | |
Long-term borrowings (including current portion) | | | 434,757 | | | | 370,653 | | | | 12,392 | |
| | | | | | | | | | | | |
| | $ | 735,025 | | | $ | 640,226 | | | $ | 21,405 | |
| | | | | | | | | | | | |
| B. | Financial risk management policies |
| (a) | The Group’s activities expose it to a variety of financial risks: market risk, credit risk, liquidity risk and cash flow interest rate risk. The Group adopts overall risk management program and control system to identify all financial risks and seeks to control and balance potential adverse effects from those aforesaid financial risks. |
| (b) | The goal of market risk management is to appropriately consider the impacts of economic environment, competition and market value risk, in order to achieve the best risk position, to maintain appropriate liquidity position and to centrally manage all market risks. |
| (c) | To meet its risk management objectives, the Group’s procedures of hedge focus on market risk and cash flow interest rate risk. |
F-53
Table of Content
| C. | C.
| Significant financial risks and degree of financial risks |
| i. | i.
| Foreign exchange risk |
| (i) | The Group’s businesses involve somenon-functional currency operations (the Company’s functional currency: NTD; the subsidiaries’ functional currencies: USD, EUR, HKD, RMB, AUD and JPY). The information on assets and liabilities denominated in foreign currencies whose values would be materially affected by the exchange rate fluctuations is as follows: |
| | | | | | | | | | | | |
| | | December 31, 2018 | |
| (Foreign currency: functional currency) | | Foreign
currency amount | | | Exchange Rate | | | Book value | |
| | | $000 | | | | | | NT$000 | |
Financial assets Monetary items | | | | | | | | | | | | |
USD : NTD | | US$ | 29,933 | | | | 30.715 | | | $ | 919,392 | |
Non-monetary items | | | | | | | | | | | | |
USD : NTD | | US$ | 1,529 | | | | 30.715 | | | | 46,963 | |
AUD : NTD | | AUD | 1,754 | | | | 21.665 | | | | 38,000 | |
Financial liabilities Monetary items | | | | | | | | | | | | |
USD : NTD | | US$ | 17,886 | | | | 30.715 | | | | 549,368 | |
AUD : NTD | | AUD | 2,544 | | | | 21.665 | | | | 55,116 | |
| | | December 31, 2017 | | | December 31, 2019 | |
(Foreign currency: functional currency) | | Foreign currency amount | | | Exchange Rate | | | Book value | | | Foreign
currency amount | | | Exchange Rate | | | Book value | | | Book value | |
| | $000 | | | | | | | NT$000 | | | $000 | | | | | | NT$000 | | | US$000 | |
Financial assets Monetary items | | | | | | | | | | | | | |
Financial assets | | | | | | | | | |
Monetary items | | | | | | | | | |
USD : NTD | US | $ | 575 | | | | 29.76 | | | $ | 17,112 | | | US$ | 3,126 | | | | 29.980 | | | $ | 93,717 | | | $ | 3,133 | |
Non-monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | US | $ | 1,437 | | | | 29.76 | | | | 42,762 | | | US$ | 1,671 | | | | 29.980 | | | | 50,097 | | | | 1,675 | |
AUD : NTD | AUD | $ | 1,084 | | | | 23.71 | | | | 25,135 | | | AUD | 1,650 | | | | 21.005 | | | | 34,658 | | | | 1,159 | |
Financial liabilities Monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | US | $ | 741 | | | | 29.76 | | | | 22,052 | | | US$ | 13,493 | | | | 29.980 | | | | 404,520 | | | | 13,525 | |
AUD : NTD | AUD | $ | 1,466 | | | | 23.71 | | | | 34,752 | | | AUD | 1,687 | | | | 21.005 | | | | 35,435 | | | | 1,185 | |
| | December 31, 2018 | |
(Foreign currency: functional currency) | | Foreign currency amount | | | Exchange Rate | | | Book value | | | Book value | |
| | $000 | | | | | | | NT$000 | | | US$000 | |
Financial assets Monetary items | | | | | | | | | | | | | | | | |
USD : NTD | US | $ | 29,333 | | | | 30.715 | | | $ | 919,392 | | | $ | 30,036 | |
Non-monetary items | | | | | | | | | | | | | | | | |
USD : NTD | US | $ | 1,529 | | | | 30.715 | | | | 46,963 | | | | 1,534 | |
AUD : NTD | AUD | $ | 1,754 | | | | 21.665 | | | | 38,000 | | | | 1,241 | |
Financial liabilities Monetary items | | | | | | | | | | | | | | | | |
USD : NTD | US | $ | 17,886 | | | | 30.715 | | | | 549,368 | | | | 17,947 | |
AUD : NTD | AUD | $ | 2,544 | | | | 21.665 | | | | 55,116 | | | | 1,801 | |
| (ii) | Analysis of foreign currency market risk arising from significant foreign exchange variation: |
| | Year ended December 31, 2017 | |
| | Sensitivity analysis | |
(Foreign currency: functional currency) | | Extent of variation | | | Effect on profit or loss | | | Effect on other comprehensive income | |
| | | | | | NT$000 | | | NT$000 | |
Financial assets Monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | $ | 171 | | | $ | — | |
Non-monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | — | | | | 428 | |
AUD:NTD | | | 1 | % | | | — | | | | 251 | |
Financial liabilities Monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | 221 | | | | — | |
AUD:NTD | | | 1 | % | | | 348 | | | | — | |
F-54
Table of Content
| | | | | | | | | | | | | | | | |
| | | 2018 | |
| | | Sensitivity analysis | |
| (Foreign currency: functional currency) | | Extent of
variation | | | Effect on
profit or loss | | | Effect on other
comprehensive
income | |
| | | | | | NT$000 | | | NT$000 | |
Financial assets Monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | $ | 9,194 | | | $ | — | |
Non monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | — | | | | 470 | |
AUD : NTD | | | 1 | % | | | — | | | | 380 | |
Financial liabilities Monetary items | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | 5,494 | | | | — | |
AUD : NTD | | | 1 | % | | | 551 | | | | — | |
| | | | | | | | | | | | | | | | | | | | |
| | | 2019 | |
| | | Sensitivity analysis | |
| (Foreign currency: functional currency) | | Extent of
variation | | | Effect on
profit or loss | | | Effect on
profit or loss | | | Effect on other
comprehensive
income | | | Effect on other
comprehensive
income | |
| | | | | | NT$000 | | | US$000 | | | NT$000 | | | US$000 | |
Financial assets Monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | $ | 937 | | | $ | 31 | | | $ | — | | | $ | — | |
Non monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | — | | | | — | | | | 501 | | | | 17 | |
AUD : NTD | | | 1 | % | | | — | | | | — | | | | 347 | | | | 12 | |
Financial liabilities Monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | 4,045 | | | | 135 | | | | — | | | | — | |
AUD : NTD | | | 1 | % | | | 354 | | | | 12 | | | | — | | | | — | |
| | Year ended December 31, 2018 | |
| | Sensitivity analysis | |
(Foreign currency: functional currency) | | Extent of variation | | | Effect on profit or loss | | | Effect on profit or loss | | | Effect on other comprehensive income | | | Effect on other comprehensive income | |
| | | | | | NT$000 | | | US$000 | | | NT$000 | | | US$000 | |
Financial assets Monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | $ | 9,194 | | | $ | 300 | | | $ | — | | �� | $ | — | |
Non-monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | — | | | | — | | | | 470 | | | | 15 | |
AUD : NTD | | | 1 | % | | | — | | | | — | | | | 380 | | | | 12 | |
Financial liabilities Monetary items | | | | | | | | | | | | | | | | | | | | |
USD : NTD | | | 1 | % | | | 5,494 | | | | 179 | | | | — | | | | — | |
AUD : NTD | | | 1 | % | | | 551 | | | | 18 | | | | — | | | | — | |
| (iii) | The unrealized exchange gain (loss) arising from significant foreign exchange variation on the monetary items held by the Group for the years ended December 31, 2016, 2017, 2018 and 20182019 are (NT$461) thousand, NT$329 thousand, and NT$24 thousand and NT$12,271 thousand (US$1410 thousand), respectively. |
| ii. | ii.
| Cash flow and fair value Interestinterest rate risk |
The Group’s interest rate risk arises from short-term and long-term borrowings. Borrowings issued at floating interest rates expose the Group to cash flow interest rate risk. During the years ended December 31, 2016, 2017, 2018 and 2018,2019, the Group’s borrowings at floating interest rate were denominated in the NT dollars and US dollars.
At December 31, 20172018 and 2018,2019, if interest ratesrate had been 0.2% higher/lower with all other conditions held constant, net loss for the years ended December 31, 20172018 and 20182019 would have been NT$232653 thousand and NT$653833 thousand (US$2128 thousand) higher/lower, respectively. The main factor is that the floating-rate borrowings resulted in changes in interest expense.
| i. | Credit risk refers to the risk of financial loss to the Group arising from cash and deposits with banks and financial institutions, as well as default by the customers on the contract obligations. The main factor is that counterparties could not repay in full the accounts receivable based on the agreed termsterms. |
| ii. | The Group manages their credit risk taking into consideration the entire Group’s concern. For banks and financial institutions, only financial institutions with a good credit rating are accepted. According to the Group’s credit policy, each entity in the Group is responsible for managing and analyzing the credit risk for each of their new customers before entering into license contracts. Internal risk control assesses the credit quality of the customers, taking into account their financial positions, past experience and other factors. |
| iii. | The default occurs when the contract payments are past due based on the agreed terms. |
| iv. | The following indicators are used to determine whether the credit impairment of debt instruments has occurred: |
| (i) | iv.It becomes probable that the issuer will enter bankruptcy or other financial reorganization due to their financial difficulties;
|
| (ii) | The disappearance of an active market for that financial asset because of financial difficulties; |
| (iii) | Default or delinquency in interest or principal repayments. |
| v. | The Group classifies customers’ accounts receivable in accordance with credit rating of customers. The Group applies the simplified approach to estimate expected credit loss under the provision matrix basis. |
| v.
vi. | The Group wrote off the financial assets, which cannot be reasonably expected to be recovered, after initiating recourse procedures. However, the Group will continue executing the recourse procedures to secure their rights. |
F-55
Table of Content
| vii. | vi.
| The Group assesses the expected credit losses based on the payment terms stipulated in the contracts with the customers. To measure the expected credit losses, trade receivables have been grouped based on shared credit risk characteristics and the days past due. The historical loss rates are adjusted to reflect current and forward-looking information on factors affecting the ability of the customers to settle the receivables. Except for loss allowance of NT$18,132 thousand (US$592 5housand) established based on the delay payment of the balances due from one customer, the Group has not identified impairments on the trade receivables. The Group has therefore concluded that the expected loss rates for the trade receivables and contract assets is very low, and the loss allowance for the trade receivables and contract assets recognized is immaterial as of December 31, 2018.2018 and 2019, respectively. As of December 31, 2018 and 2019, the balance of allowance for doubtful accounts was both NT$18,132 thousand. |
Movements in relation to the group applying the simplified approach to provide loss allowance for accounts receivable and contract assets are as follows:
| | 2018 | |
| | Accounts receivable | | | Contact assets | |
| | NT$000 | | | US$000 | | | NT$000 | | | US$000 | |
At January 1_IAS 39 | | $ | 18,132 | | | $ | 592 | | | $ | — | | | $ | — | |
Adjustments under new standards | | — | | | — | | | — | | | — | |
At January 1_IFRS 9 | | | 18,132 | | | | 592 | | | — | | | — | |
Provision for impairment | | — | | | — | | | — | | | — | |
At December 31 | | $ | 18,132 | | | $ | 592 | | | $ | — | | | $ | — | |
| viii. | i.
| Credit risk information for the years endedAs of December 31, 20162018 and 2017 is provided2019, with no collateral held or other credit enhancements, maximum exposure to credit risk in Note 12(4).respect of the Group’s accounts receivable and contract assets was NT$11,626 thousand and NT$15,120 thousand (US$506 thousand), respectively.
|
| i. | Cash flow forecasting is performed in the operating entities of the Group and aggregated by Group treasury. Group treasury monitors rolling forecasts of the Group’s liquidity requirements to ensure it has sufficient cash to meet operational needs. Additionally, the Group closely monitors the execution and covenants compliance of the loan and security agreement and intitiates discussing with the banks as needed. |
| ii. | The table below analyzes the Group’snon-derivative financial liabilities based on the remaining period at the balance sheet date to the contractual maturity date. The amounts disclosed in the table are the contractual undiscounted cash flows. |
| | | | | | | | | | | | | | | | | | | | |
| | | December 31, 2018 | |
| | | Within
1 year | | | Between 1
and 2 years | | | Between 2
and 3 years | | | Between 3
and 5 years | | | Over 5
years | |
| | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
Short-term borrowings | | $ | 46,761 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Other payables | | | 206,268 | | | | — | | | | — | | | | — | | | | — | |
Finance lease liabilities (including current portion) | | | 24,583 | | | | 24,198 | | | | — | | | | — | | | | — | |
Long-term borrowings (including current portion) | | | 82,864 | | | | 322,928 | | | | 8,351 | | | | 24,210 | | | | 30,429 | |
| | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2017 | | | December 31, 2019 | |
| | Within 1 year | | | Between 1 and 2 years | | | Between 2 and 3 years | | | Between 3 and 5 years | | | Over 5 years | | | Within
1 year | | | Between 1
and 2 years | | | Between 2
and 3 years | | | Between 3
and 5 years | | | Over 5
years | |
| | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
Short-term borrowings | | $ | 46,062 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | | | $ | 46,897 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Other payables | | | 93,541 | | | | — | | | | — | | | | — | | | | — | | | | 131,064 | | | | — | | | | — | | | | — | | | | — | |
Finance lease liabilities (including current portion) | | | 48,466 | | | | 4,008 | | | | — | | | | — | | | | — | | |
Lease liabilities (including current portion) | | | | 64,827 | | | | 20,352 | | | | 7,523 | | | | 2,055 | | | | — | |
Long-term borrowings (including current portion) | | | 5,137 | | | | 6,493 | | | | 6,430 | | | | 27,901 | | | | 33,019 | | | | 323,437 | | | | 6,367 | | | | 21,534 | | | | 5,180 | | | | 27,839 | |
| | December 31, 2018 | |
| | Within 1 year | | | Between 1 and 2 years | | | Between 2 and 3 years | | | Between 3 and 5 years | | | Over 5 years | |
| | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
Short-term borrowings | | $ | 46,761 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Other payables | | | 206,268 | | | | — | | | | — | | | | — | | | | — | |
Finance lease liabilities (including current portion) | | | 24,583 | | | | 24,198 | | | | — | | | | — | | | | — | |
Long-term borrowings (including current portion) | | | 82,864 | | | | 322,928 | | | | 8,351 | | | | 24,210 | | | | 30,429 | |
F-56
Table of Content
| | | | | | | | | | | | | | | | | | | | |
| | | December 31, 2019 | |
| | | Within
1 year | | | Between 1
and 2 years | | | Between 2
and 3 years | | | Between 3
and 5 years | | | Over 5
years | |
| | | US$000 | | | US$000 | | | US$000 | | | US$000 | | | US$000 | |
Short-term borrowings | | $ | 1,568 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Other payables | | | 4,382 | | | | — | | | | — | | | | — | | | | — | |
Lease liabilities (including current portion) | | | 2,167 | | | | 680 | | | | 252 | | | | 69 | | | | — | |
Long-term borrowings (including current portion) | | | 10,814 | | | | 213 | | | | 720 | | | | 173 | | | | 931 | |
| | December 31, 2018 | |
| | Within 1 year | | | Between 1 and 2 years | | | Between 2 and 3 years | | | Between 3 and 5 years | | | Over 5 years | |
| | US$000 | | | US$000 | | | US$000 | | | US$000 | | | US$000 | |
Short-term borrowings | | $ | 1,528 | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Other payables | | | 6,739 | | | | — | | | | — | | | | — | | | | — | |
Finance lease liabilities (including current portion) | | | 803 | | | | 791 | | | | — | | | | — | | | | — | |
Long-term borrowings (including current portion) | | | 2,707 | | | | 10,550 | | | | 273 | | | | 791 | | | | 994 | |
| (3) | (3)
| Fair value estimationinformation |
| A. | The Group had no financial instruments measured at fair value, by valuation method, as of December 31, 20172018 and 2018.2019. |
| B. | B.
| The managementManagement considers that the carrying amounts of financial assets and liabilities not measured at fair value are approximate to their fair values, including cash and cash equivalents, financial assets at amortized cost, receivables, refundable deposits, short-term borrowings, long-term borrowings (including current portion), payables, and finance lease liabilities (including current portion).
|
| (4)
| Effects on initial application of IFRS 9 and information in relation to the application of IAS 39 for the years ended December 31, 2016lease liabilities (including current and 2017
|
| A.
| The significant accounting policies of accounts receivable, other receivables, and impairment of financial assets adopted in the years ended December 31, 2016 and 2017 are as follows:
|
| (a)
| Accounts receivable and other receivables
|
Accounts receivable are claims resulting from the sale of goods or services. Other receivables are those arising from transactions other than the sale of goods or services. Accounts receivable and other receivables are recognized initially at fair value and subsequently measured at amortized cost using the effective interest method, less provision for impairment. However, short-term accounts receivable without bearing interest are subsequently measured at initial invoice amount as the effect of discounting is immaterial.
| (b)
| Impairment of financial assets
|
| i.
| The Group assesses at each balance sheet date whether there is objective evidence that a financial asset or a group of financial assets is impaired as a result of one or more events that occurred after the initial recognition of the asset (a ‘loss event’) and that loss event (or events) has an impact on the estimated future cash flows of the financial asset or group of financial assets that can be reliably estimated.
|
| ii.
| The criteria that the Group uses to determine whether there is objective evidence of an impairment loss is as follows:
|
| (a)
| Significant financial difficulty of the issuer or debtor;
|
| (b)
| A breach of contract, such as a default or delinquency in interest or principal payments;
|
| (c)
| The Group, for economic or legal reasons relating to the borrower’s financial difficulty, granted the borrower a concession that a lender would not otherwise consider;
|
| (d)
| It becomes probable that the borrower will enter bankruptcy or other financial reorganization;
|
| (e)
| Information about significant changes with an adverse effect that have taken place in the technology, market, economic or legal environment in which the issuer operates, and indicates that the cost of the investment in the equity instrument may not be recovered.
|
| iii.
| When the Group assesses that there has been objective evidence of impairment and an impairment loss has occurred on financial assets measured at amortized cost, the amount of the impairment loss is measured as the difference between the asset’s carrying amount and the present value of estimated future cash flows discounted using the financial asset’s original effective interest rate, and is recognized in profit or loss. If, in
|
F-57
Table of Content
| | a subsequent period, the amount of the impairment loss decreases and the decrease can be related objectively to an event occurring after the impairment loss was recognized, the previously recognized impairment loss is reversed through profit or loss to the extent that the carrying amount of the asset does not exceed its amortized cost that would have been at the date of reversal had the impairment loss not been recognized previously. Impairment loss is recognized and reversed by adjusting the carrying amount of the asset through the use of an impairment allowance account.
|
| B.
| The reconciliation of allowance for impairment and provision from December 31, 2017, as these are impaired under IAS 39, to January 1, 2018, as these are expected to be impaired under IFRS 9, are as follows:
|
| | Accounts receivable | |
| | NT$000 | | | US$000 | |
IAS 39 | | $ | 18,132 | | | $ | 592 | |
Impairment loss adjustment | | | — | | | | — | |
IFRS 9 | | $ | 18,132 | | | $ | 592 | |
| C.
| Credit risk information for the years ended December 31, 2016 and 2017 are as follows:
|
| (a)
| Credit risk refers to the risk of financial loss to the Group arising from default by the customers on the contract obligations. According to the Group’s credit policy, each entity in the Group is responsible for managing and analysing the credit risk for each of their new customers before signing the license agreement. Internal risk control assesses the credit quality of the customers, taking into account their financial position, past experience and other factors. Credit risk arises from cash and deposits with banks and financial institutions, as well as credit exposures to corporate pharmaceutical companies, including outstanding receivables. For banks and financial institutions, only financial institutions with a good credit rating are accepted.
|
| (b)
| For the years ended December 31, 2016 and 2017, no credit limits were exceeded during the reporting periods, and management does not expect any significant losses from non-performance by these counterparties.
|
| (c)
| The Group’s accounts receivable that were neither past due nor impaired were fully performing in line with the credit standards prescribed based on counterparties’ industrial characteristics, scale of business and profitability. As of December 31, 2017, the Group’s accounts receivable that were neither past due nor impaired amounted to NT$8,622 thousand. The Group deals with counterparties with good credit reputation and has policies in place to ensure that customers have an appropriate credit history when signing the contracts.
|
| (d)
| The ageing analysis of financial assets that were past due but not impaired is as follows:
|
| | December 31, 2017
| |
| | NT$000
| |
Up to 30 days
| | $
| —
| |
31 to 90 days
| | | —
| |
91 to 180 days
| | | —
| |
Over 181 days
| | | —
| |
| | $
| —
| |
The above ageing analysis were based on past due date.
| D.
| Movement analysis of accounts receivable that were impaired is as follows:
|
| (a)
| As of December 31, 2017, the Group’s accounts receivable that were individually determined to be impaired amounted to NT$18,132 thousand.
|
F-58
Table of Content
| (b)
| Movements on the Group’s provision for impairment of accounts receivable for the year ended December 31, 2017 are as follows:non-current).
|
| | 2017 | |
| | Individually assessed for impairment | | | Collectively assessed for impairment | | | Total | |
| | NT$000 | | | NT$000 | | | NT$000 | |
At January 1 | | $ | 9,067 | | | $ | — | | | $ | 9,067 | |
Provision for impairment | | | 9,065 | | | | — | | | | 9,065 | |
At December 31 | | $ | 18,132 | | | $ | — | | | $ | 18,132 | |
| 13. | (5)
| Effects of initial application of IFRS 15 and information in relation to the application of IAS 18 for the years ended December 31, 2016 and 2017SEGMENT INFORMATION
|
| (1) | A.
| The significant accounting policies of revenue recognition for the years ended December 31, 2016 and 2017 are as follows:General information
|
| (a)
| Out-licensing development collaboration revenue includes up-front fees and milestone payments. Upfront fees are recognized in a reasonable and systematic approach over the development period, and is not recognized in full, if the authorization contract of the Group does not meet all of the following criteria:
|
| i.
| The amount of royalty is fixed or non-refundable.
|
| ii.
| The contract is irrevocable.
|
| iii.
| Relevant rights may be at the authorized party’s own disposition.
|
| iv.
| The party granting authority has no further obligations after passing on the rights to the authorized party.
|
Milestone payments are contractual payments due to the Group upon the achievement of certain additional events. The entire milestone payment due is recognized as revenue, in full, at the time the milestone set forth in the respective agreements has been achieved and the amount is reasonably assured of collection.
| (b)
| Royalty revenues are recognized based on the substance of contracts when the earning process is substantially completed and are realized or realizable, which is in the same period that the licensee makes a qualifying sale of licensed products.
|
| B.
| The revenue recognized by using previous accounting policies for the years ended December 31, 2016 and 2017 are as follows:
|
| | 2016 | | | 2017 | |
| | NT$000 | | | NT$000 | |
Royalty revenue | | $ | 40,571 | | | $ | 49,635 | |
Co-development revenue | | | 1,103 | | | | — | |
| | $ | 41,674 | | | $ | 49,635 | |
F-59
Table of Content
| C.
| The effects and description of current balance sheets and comprehensive income statements items if the Company continued adopting previous accounting policies as of and for the year ended December 31, 2018 are as follows:
|
| | | | As of and for the year ended December 31, 2018 | |
Items | | Description | | Balance by using IFRS 15 | | | Balance by using previous accounting policies | | | Effects from changes in accounting policies | |
| | | | NT$000 | | | NT$000 | | | NT$000 | |
Contract assets | | Note | | $ | 2,283 | | | $ | — | | | $ | 2,283 | |
Operating revenue | | Note | | $ | 10,224 | | | $ | — | | | $ | 10,224 | |
Accumulated deficit | | Note | | $ | 7,941 | | | $ | — | | | $ | 7,941 | |
| | | | | | | | | | | | | | |
| | | | As of and for the year ended December 31, 2018 | |
Items | | Description | | Balance by using IFRS 15 | | | Balance by using previous accounting policies | | | Effects from changes in accounting policies | |
| | | | US$000 | | | US$000 | | | US$000 | |
Contract assets | | Note | | $ | 75 | | | $ | — | | | $ | 75 | |
Operating revenue | | Note | | $ | 334 | | | $ | — | | | $ | 334 | |
Accumulated deficit | | Note | | $ | 259 | | | $ | — | | | $ | 259 | |
Note: The Group accounts for all the authorization and subsequent research and development services provided by the Group as one performance obligation, and recognizes revenue based on the transaction price, and the stage of completion, which is measured by the proportion of contract costs incurred for research and development services performed as of the financial reporting date to the estimated total research and development costs for the authorization collaboration and development contracts.
The Group’s major business is research and development for new medicine and operates only in one single industry. The Chief Operating Decision-Maker, who allocates resources and assesses performance of the Group as a whole, has identified that the Group has only one reportable operating segment.
| (2) | (2)
| Information about segment profit or loss, assets and liabilities |
The Group has only one reportable operating segment, and therefore, the reportable segment information is the same as the financial statements.
The adoption of IFRS 16, ‘Leases’, had impact on the segment information in 2019, which resulted in increase in depreciation and interest expenses. Please refer to Note 6(6) for the related information.
| (3) | (3)
| Reconciliation for segment income (loss) |
The segment income (loss) reported to the chief operating decision-maker is measured in a manner consistent with that in the statement of comprehensive income. There is no reconciliation because the report provided to the chief operating decision-maker for business decisions has no difference to the segment income (loss) information.
| (4) | (4)
| Information on product and service |
Please refer to Note 6(18)6(19) for the related information.
F-60
Table of Content
| (5) | (5)
| Geographical information |
Geographical information for the years ended December 31, 2016, 2017, 2018 and 20182019 is as follows:
| | | Year ended December 31, 2016 | | | Year ended December 31, 2017 | | | 2017 | | | 2018 | |
| | Revenue | | | Non-current assets (Note) | | | Revenue | | | Non-current assets (Note) | | | Revenue | | | Non-current
assets (Note) | | | Revenue | | | Non-current
assets (Note) | |
| | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | | | NT$000 | |
Taiwan | | $ | 40,571 | | | $ | 199,916 | | | $ | 49,635 | | | $ | 176,727 | | | $ | 49,635 | | | $ | 176,727 | | | $ | 52,100 | | | $ | 202,724 | |
Europe | | | 1,003 | | | | — | | | | — | | | | — | | |
China | | | — | | | | 35 | | | | — | | | | 23 | | | | — | | | | 23 | | | | — | | | | 11 | |
Others | | | 100 | | | | 13,788 | | | | — | | | | 6,645 | | | | — | | | | 6,645 | | | | 10,224 | | | | 7,482 | |
| | $ | 41,674 | | | $ | 213,739 | | | $ | 49,635 | | | $ | 183,395 | | | | | | | | | | | | | |
| | | $ | 49,635 | | | $ | 183,395 | | | $ | 62,324 | | | $ | 210,217 | |
| | | | | | | | | | | | | |
| | | Year ended December 31, 2018 | | | Year ended December 31, 2018 | | | 2019 | | | 2019 | |
| | Revenue | | | Non-current assets (Note) | | | Revenue | | | Non-current assets (Note) | | | Revenue | | | Non-current
assets (Note) | | | Revenue | | | Non-current
assets (Note) | |
| | NT$000 | | | NT$000 | | | US$000 | | | US$000 | | | NT$000 | | | NT$000 | | | US$000 | | | US$000 | |
Taiwan | | $ | 52,100 | | | $ | 202,724 | | | $ | 1,702 | | | $ | 6,623 | | | $ | 58,970 | | | $ | 246,719 | | | $ | 1,972 | | | $ | 8,249 | |
Europe | | | 10,224 | | | | — | | | | 334 | | | | — | | |
China | | | — | | | | 11 | | | | — | | | | — | | | | 142,840 | | | | — | | | | 4,775 | | | | — | |
Others | | | — | | | | 7,482 | | | | — | | | | 245 | | | | 7,330 | | | | 19,218 | | | | 245 | | | | 642 | |
| | $ | 62,324 | | | $ | 210,217 | | | $ | 2,036 | | | $ | 6,868 | | | | | | | | | | | | | |
| | | $ | 209,140 | | | $ | 265,937 | | | $ | 6,992 | | | $ | 8,891 | |
| | | | | | | | | | | | | |
| Note: | Deferred tax assets and refundable deposits are excluded fromnon-current assets. |
| (6) | (6)
| Major customer information |
Details of sales to individual customers exceeding 10% of the Group’s revenue for the years ended December 31, 2016, 2017, 2018 and 20182019 are as follows:
| | Year ended December 31, | | | | |
| | 2016 | | | 2017 | | 2017 | | | 2018 | |
Customer | | Revenue | | | Revenue | | | | Revenue | | | Revenue | |
| | NT$000 | | | NT$000 | | | | NT$000 | | | NT$000 | |
A | | $ | 34,357 | | | $ | 40,385 | | | | $ | 40,385 | | | $ | 40,765 | |
B | | | 6,214 | | | | 9,250 | | | | | 9,250 | | | | 11,335 | |
F | | | 1,003 | | | | — | | | | | — | | | | 10,224 | |
| | Year ended December 31, | | | | | |
| | 2018 | | | 2018 | | | 2019 | | | 2019 | |
Customer | | Revenue | | | Revenue | | | Revenue | | | Revenue | |
| | NT$000 | | | US$000 | | | NT$000 | | | US$000 | |
D | | | $ | 142,840 | | | $ | 4,776 | |
A | | $ | 40,765 | | | $ | 1,332 | | | | 45,828 | | | | 1,532 | |
B | | | 11,335 | | | | 370 | | | | 13,142 | | | | 439 | |
F | | | 10,224 | | | | 334 | | |
F-73
F-61