Use these links to rapidly review the documentTABLE OF CONTENTS
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
Proxy Statement Pursuant to Section 14(a) of
the Securities Exchange Act of 1934 (Amendment No. )
| Filed by the Registrantý | ||
Filed by a Party other than the Registranto | ||
Check the appropriate box: | ||
Preliminary Proxy Statement | ||
o | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) | |
Definitive Proxy Statement | ||
o | Definitive Additional Materials | |
o | Soliciting Material under §240.14a-12 | |
| Bristol-Myers Squibb Company | ||||
(Name of Registrant as Specified In Its Charter) | ||||
(Name of Person(s) Filing Proxy Statement, if other than the Registrant) | ||||
Payment of Filing Fee (Check the appropriate box): | ||||
ý | No fee required. | |||
o | Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11. | |||
| (1) | Title of each class of securities to which transaction applies: | |||
| (2) | Aggregate number of securities to which transaction applies: | |||
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): | |||
| (4) | Proposed maximum aggregate value of transaction: | |||
| (5) | Total fee paid: | |||
o | Fee paid previously with preliminary materials. | |||
o | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. | |||
(1) | Amount Previously Paid: | |||
| (2) | Form, Schedule or Registration Statement No.: | |||
| (3) | Filing Party: | |||
| (4) | Date Filed: | |||
![]()
March 19, 2014, 2015
| NOTICE OF MEETING AND PROXY STATEMENT TUESDAY, MAY AT 10:00 A.M. BRISTOL-MYERS SQUIBB COMPANY 777 SCUDDERS MILL RD. PLAINSBORO NEW JERSEY | DEARFELLOWSTOCKHOLDER: You are cordially invited to attend the Annual Meeting of Stockholders of Bristol-Myers Squibb Company at our offices located in Plainsboro, New Jersey, on Tuesday, May These materials include the Notice of Annual Meeting and the Proxy Statement. The Proxy Statement describes the business to be transacted at the meeting and provides other information about the company that you should know when you vote your shares. The principal business of the Annual Meeting will be:
(i) the election of directors;
(ii) an advisory vote to approve the compensation of our named executive officers; (iii) the ratification of the appointment of an independent registered public accounting firm; (iv) the consideration of two amendments to our Amended and
Restated Certificate of Incorporation; and (v) the consideration of one stockholder proposal. We will also review the status of the company's business at the meeting. Last year, over Please follow the instructions in the Proxy Statement on how to attend the Annual Meeting. Admission to the Annual Meeting will be by ticket only.Please bring photo identification. We have provided space on the proxy card for comments from our registered stockholders. We urge you to use it to let us know your feelings about BMS or to bring a particular matter to our attention. If you hold your shares through an intermediary or received the proxy materials electronically, please feel free to write directly to us. | |||||
 |  | |||||
JAMES M. CORNELIUS | LAMBERTO ANDREOTTI | |||||
| Chairman of the Board | Chief Executive Officer and Chairman-Designate |
![]()
345 Park Avenue
New York, New York 10154-0037
NOTICE OF ANNUAL MEETING
OF STOCKHOLDERS
Notice is hereby given that the 20142015 Annual Meeting of Stockholders will be held at Bristol-Myers Squibb Company, 777 Scudders Mill Road, Plainsboro, New Jersey, on Tuesday, May 6, 2014,5, 2015, at 10:00 a.m. for the following purposes as set forth in the accompanying Proxy Statement:
Holders of record of our common and preferred stock at the close of business on March 14, 201413, 2015 will be entitled to vote at the meeting.
| By Order of the Board of Directors | ||
| SANDRALEUNG General Counsel and Corporate Secretary |
Dated: March 19, 2014, 2015
Regardless of the number of shares you own, your vote is important. If you do not attend the Annual Meeting to vote in person, your vote will not be counted unless a proxy representing your shares is presented at the meeting. To ensure that your shares will be voted at the meeting, please vote in one of these ways:
If you do attend the Annual Meeting, you may revoke your proxy and vote by ballot.
2015 Annual Meeting of Stockholders
| Date: | Tuesday, May 5, 2015 | |
| Time: | 10:00 a.m. | |
| Place: | 777 Scudders Mill Road, Plainsboro, New Jersey |
| Voting Matters | ||||||||
|---|---|---|---|---|---|---|---|---|
| | | | | | | | | |
| Item | Proposal | Board Vote Recommendation | Required Vote | Page Number | ||||
| 1 | Election of Directors | FOR ALL | Majority of votes cast | 14 | ||||
| 2 | Advisory vote to approve the compensation of our named executive officers | FOR | Majority of shares voted | 71 | ||||
| 3 | Ratification of the appointment of an independent registered public accounting firm | FOR | Majority of shares voted | 72 | ||||
| 4 | Approval of Amendment to our Amended and Restated Certificate of Incorporation to designate Delaware Chancery Court as the exclusive legal forum for certain legal actions | FOR | Majority of outstanding shares | 75 | ||||
| 5 | Approval of Amendment to our Amended and Restated Certificate of Incorporation to remove the supermajority voting provisions applicable to Preferred Stockholders | FOR | Majority of outstanding sharesAND 2/3 of outstanding preferred shares | 76 | ||||
| 6 | Stockholder proposal on shareholder action by written consent | AGAINST | Majority of shares voted | 77 | ||||
| | | | | | | | | |
| Nominees for Board of Directors | ||||||||
|---|---|---|---|---|---|---|---|---|
| | | | | | | | | |
| Name | Occupation | Independent | Committee Memberships* | Other Public Company Boards | ||||
Lamberto Andreotti | Chief Executive Officer and Chairman-Designate of the Company | No | | 1 | ||||
Giovanni Caforio, M.D. | Chief Operating Officer and CEO-Designate of the Company | No | 0 | |||||
| Former Chairman and Chief Executive Officer of Textron Inc. and Navistar International Corporation | Yes | CDCG (c); CMDC | 2 | ||||
Laurie H. Glimcher, M.D. | Dean of Weill Cornell Medical College and the Cornell University Provost for Medical Affairs | Yes | Audit; S&T | 1 | ||||
Michael Grobstein | Former Vice Chairman of Ernst & Young LLP | Yes | Audit; CMDC | 1 | ||||
Alan J. Lacy | Former Chairman and Chief Executive Officer, Sears, Roebuck and Co. | Yes | Audit (c); CDCG | 1 | ||||
Thomas J. Lynch, Jr., M.D. | Director of Yale Cancer Center; Professor of Internal Medicine, Yale Cancer Center, Yale School of Medicine; and Physician-in-Chief of Smilow Cancer Hospital, Yale-New Haven | Yes | CDCG; S&T | 0 | ||||
Dinesh C. Paliwal | Chairman, President and Chief Executive Officer of Harman International Industries, Inc. | Yes | Audit; CDCG | 1 | ||||
Vicki L. Sato, Ph.D. | Professor of Management Practice at the Harvard Business School | Yes | CMDC; S&T (c) | 2 | ||||
Gerald L. Storch | Chief Executive Officer of Hudson's Bay Company | Yes | Audit; CMDC | 1 | ||||
Togo D. West, Jr. | Chairman of TLI Leadership Group and Former U.S. Secretary of Veterans Affairs | Yes | CDCG; CMDC (c) | 2 | ||||
| | | | | | | | | |
* Audit: Audit Committee | ||||||||
CDCG: Committee on Directors and Corporate Governance | (c): Committee Chair | |||||||
CMDC: Compensation and Management Development Committee | ||||||||
S&T: Science and Technology Committee | ||||||||
| | | | | | | | | |
Corporate Governance Highlights
The Committee on Directors and Corporate Governance continually reviews corporate governance issues and is responsible for identifying and recommending corporate governance initiatives. Below are some significant corporate governance features and best practices that the Company has adopted:
Annual election of Directors |
Semi-annual disclosure of | |||||||
• Majority voting standard and resignation policy for election of Directors | • Director retirement policy (age 75) | |||||||
• Ability for stockholders to call a special meeting (25%) | • Clawback and recoupment policies | |||||||
• No supermajority voting provisions for common stockholders | • Share ownership and retention policy | |||||||
• No stockholder rights plan | • Limit on number of | public company directorships Board members may hold (4) | ||||||
• Extensive related party transaction policies and procedures |
| Active stockholder engagement | ||||||
• Prohibition of speculative and hedging transactions by all employees and directors |
| |||||||
Annual | review of Corporate Governance Guidelines | |||||||
Stockholder Engagement
Bristol-Myers Squibb values the views of its stockholders. In 2014, members of management met with institutional stockholders holding a substantial portion of our outstanding shares to discuss the Company's executive compensation program and general corporate governance issues. We received valuable and generally positive feedback from these meetings, which is described in more detail on page 32.
Executive Compensation
Our Compensation Discussion and Analysis can be found on page 29 of the Proxy Statement.
Performance Graph
The following performance graph compares the performance of Bristol-Myers Squibb for the periods indicated with the performance of the Standard & Poor's 500 Stock Index (S&P 500) and the average performance of our executive compensation extended peer group which is listed on page 35 of this Proxy Statement.
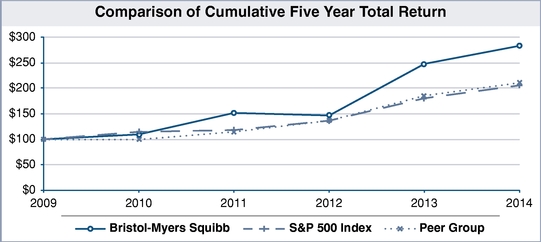
Assumes $100 invested on December 31, 2008 in Bristol-Myers Squibb common stock, S&P 500 Index and the peer group. Values are as of December 31 of specified year assuming dividends are reinvested. Total return indices reflect reinvested dividends and are weighted using beginning-period market capitalization for each of the reported time periods.
![]()
PROXY STATEMENT
| | Page | |
|---|---|---|
GENERAL INFORMATION ABOUT THE ANNUAL MEETING AND VOTING | 1 | |
CORPORATE GOVERNANCE AND BOARD MATTERS | ||
Board's Role in Strategic Planning and Risk Oversight | ||
Director Independence | 7 | |
Board Leadership Structure | ||
Meetings of our Board | ||
Annual Meeting of Stockholders | ||
Committees of our Board | ||
Compensation Committee Interlocks and Insider Participation | ||
Risk Assessment of Compensation Policies and Practices | ||
Criteria for Board Membership | ||
Identification and Selection of Nominees for our Board | ||
Stockholder Nominations for Director | ||
Information on Nominees for Directors | ||
Communications with our Board of Directors | ||
Codes of Conduct | ||
Related Party Transactions | ||
Availability of Corporate Governance Documents | ||
Compensation of Directors | ||
VOTING SECURITIES AND PRINCIPAL HOLDERS | ||
Common Stock Ownership by Directors and Executive Officers | ||
Principal Holders of Voting Securities | ||
Section 16(a) Beneficial Ownership Reporting Compliance | ||
Policy on Hedging and Pledging | ||
EXECUTIVE COMPENSATION | ||
Compensation Discussion and Analysis | ||
Compensation and Management Development Committee Report | ||
Summary Compensation Table | ||
Grants of Plan-Based Awards | ||
Outstanding Equity Awards at Fiscal Year-End | ||
Option Exercises and Stock Vesting | ||
Present Value of Accumulated Pension Benefits | ||
Non-Qualified Deferred Compensation | ||
Post-Termination Benefits | ||
Termination of Employment Obligations (Excluding Vested Benefits) | ||
ITEMS TO BE VOTED UPON | ||
Item 1—Election of Directors | ||
Item 2—Advisory Vote to Approve the Compensation of our Named Executive Officers | 71 | |
Equity Compensation Plan Information | 72 | |
Item 3—Ratification of the Appointment of Independent Registered Public Accounting Firm | ||
Audit and Non-Audit Fees | ||
Pre-Approval Policy for Services Provided by our Independent Registered Public Accounting Firm | ||
Audit Committee Report | ||
| ||
| ||
Item 4— | ||
Item 5—Approval of Amendment to our Amended and Restated Certificate of Incorporation to Remove the Supermajority Provisions Applicable to Preferred Stockholders | 76 | |
Item 6—Stockholder Proposal on Shareholder Action by Written Consent | 77 | |
OTHER MATTERS | ||
Exhibit A—Categorical Standards of Independence | A-1 | |
Exhibit B—Certificate of Amendment to Amended and Restated Certificate of Incorporation—exclusive forum provision | B-1 | |
Exhibit C—Certificate of Amendment to Amended and Restated Certificate of Incorporation—supermajority voting applicable to preferred stockholders | C-1 | |
DIRECTIONS TO OUR PLAINSBORO OFFICE Inside Back Cover | ||
GENERAL INFORMATION ABOUT THE ANNUAL MEETING AND VOTING
Why am I receiving these materials?
This Proxy Statement is being delivered to all stockholders of record as of the close of business on March 14, 201413, 2015 in connection with the solicitation of proxies on behalf of the Board of Directors for use at the Annual Meeting of Stockholders on May 6, 2014.5, 2015. We expect our proxy materials, including this Proxy Statement and the Annual Report, to be first made available to stockholders on or about March 19, 2014., 2015. Although the Annual Report and Proxy Statement are being delivered together, the Annual Report should not be deemed to be part of the Proxy Statement.
What is "Notice and Access" and how does it affect me?
The U.S. Securities and Exchange Commission (SEC) has adopted a "Notice and Access" model which permits us to provide proxy materials to our stockholders electronically by posting the proxy materials on a publicly accessible website. Delivering proxy materials electronically will conserve natural resources and save us money by reducing printing and mailing costs. Accordingly, we have sent to most of our stockholders a "Notice of Internet Availability of Proxy Materials." This Notice provides instructions on how to access our proxy materials online and, if you prefer receiving a paper copy of the proxy materials, how you can request one. Employees and pension plan participants who have given consent to receive materials electronically received a link to access our proxy materials by email. We encourage all of our stockholders who currently receive paper copies of the proxy materials to elect to view future proxy materials electronically if they have Internet access. You can do so by following the instructions when you vote your shares online or, if you are a beneficial holder, by asking your bank, broker or other holder of record how to receive proxy materials electronically.
What is "householding" and how does it work?
"Householding" is a procedure we adopted whereby stockholders of record who have the same last name and address and who receive the proxy materials by mail will receive only one copy of the proxy materials unless we have received contrary instructions from one or more of the stockholders. This procedure reduces printing and mailing costs. If you wish to receive a separate copy of the proxy materials, now or in the future, at the same address, or if you are currently receiving multiple copies of the proxy materials at the same address and wish to receive a single copy, you may contact us by writing to Stockholder Services, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, or by calling us at (212) 546-3309.
If you are a beneficial owner (your shares are held in the name of a bank, broker or other holder of record), the bank, broker or other holder of record may deliver only one copy of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, to stockholders who have the same address unless the bank, broker or other holder of record has received contrary instructions from one or more of the stockholders. If you wish to receive a separate copy of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, now or in the future, you may contact us at the address or phone number above and we will promptly deliver a separate copy. Beneficial owners sharing an address who are currently receiving multiple copies of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, and wish to receive a single copy in the future, should contact their bank, broker or other holder of record to request that only a single copy be delivered to all stockholders at the shared address in the future.
Who can attend the Annual Meeting?
Only stockholders of Bristol-Myers Squibb as of the record date, March 14, 2014,13, 2015, their authorized representatives and guests of Bristol-Myers Squibb may attend the Annual Meeting. Admission will be by ticket only. A form of government-issued photograph identification will be required to enter the meeting. Large bags, backpacks, briefcases, cameras, recording equipment and other electronic devices will not be permitted in the meeting, and attendees will be subject to security inspections. Our
offices are wheelchair accessible. We will provide, upon request, wireless headsets for hearing amplification.
How do I receive an admission ticket?
If you are a registered stockholder (your shares are held in your name) and plan to attend the meeting, you should bring either the Notice of Internet Availability of Proxy Materials or the top portion of the proxy card, both of which will serve as your admission ticket.
If you are a beneficial owner (your shares are held in the name of a bank, broker or other holder of record) and plan to attend the meeting, you can obtain an admission ticket in advance by writing to Stockholder Services, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154. Please be sure to enclose proof of ownership, such as a bank or brokerage account statement. Stockholders who do not obtain tickets in advance may obtain them upon verification of ownership at the Registration Desk on the day of the Annual Meeting.
We may also issue tickets to other individuals at our discretion.
All holders of record of our $0.10 par value common stock and $2.00 convertible preferred stock at the close of business on March 14, 201413, 2015 will be entitled to vote at the 20142015 Annual Meeting. Each share is entitled to one vote on each matter properly brought before the meeting.
How do I vote if I am a registered stockholder?
Proxies are solicited to give all stockholders who are entitled to vote on the matters that come before the meeting the opportunity to do so whether or not they attend the meeting in person. If you are a registered holder, you can vote your shares by proxy in one of the following manners:
Choosing to vote via Internet or calling the toll-free number listed above will save us expense. In order to vote online or via telephone, have the voting form in hand and either call the number or go to the website and follow the instructions. If you vote via the Internet or by telephone, please do not return a signed proxy card.
If you received a paper copy of the proxy materials and choose to vote by mail, specify how you want your shares voted on each proposal by marking the appropriate boxes on the proxy card enclosed with the Proxy Statement, date and sign it, and mail it in the postage-paid envelope.
If you wish to vote in person, you can vote your shares at the Annual Meeting.
How do I vote if I am a beneficial stockholder?
If you are a beneficial stockholder, you have the right to direct your broker or nominee on how to vote the shares. You should complete a voting instruction card which your broker or nominee is obligated to provide you. If you wish to vote in person at the meeting, you must first obtain from the record holder a legal proxy issued in your name.
Under the rules of the New York Stock Exchange (NYSE), brokers that have not received voting instructions from their customers ten days prior to the meeting date may vote their customers' shares in the brokers' discretion on the proposals regarding routine matters, which in most cases includes the ratification of the appointment of the independent registered public accounting firm.
Under NYSE rules, the election of directors, the advisory vote to approve the compensation of our named executive officers, the approval of two amendments to our Amended and Restated Certificate of Incorporation and the approval of any stockholder proposals are considered "non-discretionary" items, which means that your broker cannot vote your shares on these proposals.
What items will be voted upon at the Annual Meeting?
At the Annual Meeting, we will consider and act on the following items of business:
We do not know of any other matter that may be brought before the meeting. However, if other matters are properly presented for action, it is the intention of the named proxies to vote on them according to their best judgment.
What are the Board of Directors' voting recommendations?
For the reasons set forth in more detail later in the Proxy Statement, our Board of Directors recommends a vote FOR the election of each director, FOR the advisory vote to approve the compensation of our named executive officers, FOR the ratification of the appointment of Deloitte & Touche LLP as our independent registered public accounting firm for 20142015, FOR each of the two amendments to our Amended and FOR the advisory vote to approve the compensationRestated Certificate of our named executive officers. The Board of Directors has decided not to take a position FOR orIncorporation and AGAINST the stockholder proposal on simple majority vote.proposal.
How will my shares be voted at the Annual Meeting?
The individuals named as proxies on the proxy card will vote your shares in accordance with your instructions. Please review the voting instructions and read the entire text of the proposals and the positions of the Board of Directors in the Proxy Statement prior to marking your vote.
If your proxy card is signed and returned without specifying a vote or an abstention on a proposal, it will be voted according to the recommendation of the Board of Directors on that proposal. That recommendation is shown for each proposal on the proxy card. With respect to the shareholder proposal on simple majority vote, in the absence of voting instructions to the contrary, shares represented by validly executed proxies will be voted ABSTAIN.
How many votes are needed to elect the directors and to approve each of the proposals?
Director Elections: A majority of votes cast with respect to each director's election at the meeting is required to elect each director. A majority of the votes cast means that the number of votes cast "for" a director must exceed the number of votes cast "against" that director in order for the director to be elected. Abstentions will not be counted as votes cast for or against the director and broker non-votes will have no effect on this proposal.
Ratification of our Auditors: The affirmative vote of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required for the ratification of the appointment of our independent registered public accounting firm. Abstentions will be counted as votes against this proposal. As described above, a broker or other nominee may generally vote on routine matters such as this one, and therefore no broker non-votes are expected to exist in connection with this proposal.
Advisory Vote: The affirmative vote of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required for the approval of the advisory vote to approve the compensation of our named executive officers. Because your vote is advisory, it will not be binding upon our Board of Directors. Abstentions will be counted as votes against this proposal and broker non-votes will have no effect on this proposal.
Ratification of our Auditors: The affirmative vote of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required for the ratification of the appointment of our independent registered public accounting firm. Abstentions will be counted as votes against this
proposal. As described above, a broker or other nominee may generally vote on routine matters such as this one, and therefore no broker non-votes are expected to exist in connection with this proposal.
Exclusive Forum Provision: The affirmative vote of the holders of a majority of our outstanding shares entitled to vote on the matter is required to approve the Certificate of Amendment to our Amended and Restated Certificate of Incorporation to designate the Court of Chancery of the State of Delaware as the sole and exclusive forum for specified legal actions, unless otherwise consented to by the company. Abstentions and broker non-votes will be counted as votes against this proposal.
Removal of Supermajority Voting Provisions: Both the affirmative vote of the holders of a majority of our outstanding shares entitled to vote on the matter and the affirmative vote of the holders of at least two-thirds of our outstanding shares of preferred stock is required to approve the Certificate of Amendment to our Amended and Restated Certificate of Incorporation to eliminate the supermajority voting provisions applicable to preferred stockholders. Abstentions and broker non-votes will be counted as votes against the proposal.
Stockholder Proposal: The affirmative vote of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required for the approval of the stockholder proposal, if presented at the meeting. Abstentions will be counted as votes against this proposal and broker non-votes will have no effect on this proposal.
In accordance with the laws of Delaware, our Amended and Restated Certificate of Incorporation and our Bylaws, for all matters being submitted to a vote of stockholders, only proxies and ballots that indicate votes "FOR," "AGAINST" or "ABSTAIN" on the proposals, or that provide the designated proxies with the right to vote in their judgment and discretion on the proposals are counted to determine the number of shares present and entitled to vote. Broker non-votes are not counted as shares present and entitled to vote but will be counted for purposes of determining quorum (whether enough votes are present to hold the Annual Meeting).
Can I change my vote after I return the proxy card, or after voting by telephone or electronically?
If you are a stockholder of record, you can revoke your proxy at any time before it is voted at the meeting by taking one of the following three actions:
If you are a beneficial owner of shares, you may submit new voting instructions by contacting your bank, broker or other holder of record. You may also vote in person at the Annual Meeting if you obtain a legal proxy.
All shares that have been properly voted and not revoked will be voted at the Annual Meeting.
If you wish to give your proxy to someone other than the persons named as proxies in the enclosed form of proxy, you may do so by crossing out the names of all three persons named as proxies on the proxy card and inserting the name of another person. The signed card must be presented at the meeting by the person you have designated on the proxy card.
An independent agent tabulates the proxies and the votes cast at the meeting. In addition, independent inspectors of election certify the results of the vote tabulation.
Yes, any information that identifies a stockholder or the particular vote of a stockholder is kept confidential.
Who will pay for the costs involved in the solicitation of proxies?
We will pay all costs of preparing, assembling, printing and distributing the proxy materials as well as the solicitation of all proxies. We have retained Georgeson Shareholder Communications Inc. to assist in soliciting proxies for a fee of $18,000, plus reasonable out-of-pocket expenses. We may solicit proxies on behalf of the Board of Directors through the mail, in person, electronically, and by telecommunications. We will, upon request, reimburse brokerage firms and others for their reasonable expenses incurred for forwarding solicitation material to beneficial owners of stock.
CORPORATE GOVERNANCE AND BOARD MATTERS
Our business is managed under the direction of our Board of Directors pursuant to the Delaware General Corporation Law and our Bylaws. The Board has responsibility for establishing broad corporate policies and for the overall performance of our company. The Board is kept advised of company business through regular written reports and analyses and discussions with the CEO and other officers of Bristol-Myers Squibb, by reviewing materials provided to them and by participating in Board and Board Committee meetings.
The Committee on Directors and Corporate Governance continually reviews corporate governance issues and is responsible for identifying and recommending the adoption of corporate governance initiatives. In addition, our Compensation and Management Development Committee regularly reviews compensation issues and recommends adoption of policies and procedures that strengthen our compensation practices. The Compensation Discussion and Analysis beginning on page 2729 discusses many of these policies and procedures.
Listed below are some of the significant corporate governance initiatives we have adopted:
other payments made to trade associations to which we give $50,000 or more that can be attributed to lobbying expenditures.
The Board of Directors has adopted Corporate Governance Guidelines that govern its operation and that of its Committees. FromOur Board annually reviews our Corporate Governance Guidelines and, from time to time, our Board revises the Corporate Governance Guidelinesthem in response to changing regulatory requirements, evolving best practices and the concerns of our stockholders and other constituents. Our Corporate Governance Guidelines may be viewed on our website at www.bms.com/ourcompany/governance.
Board's Role in Strategic Planning and Risk Oversight
Our Board meets regularly to discuss the strategic direction and the issues and opportunities facing our company in light of trends and developments in the biopharmaceutical industry and general business environment. Our Board has been instrumental in determining our strategy to further evolve our business model to become a leadingdiversified specialty biopharmaceutical company. Throughout the year, our Board provides guidance to management regarding our strategy and helps to refine our operating plans to implement our strategy. Each year, typically during the second quarter, the Board holds an extensive meeting with senior management dedicated to discussing and reviewing our long-term operating plans and overall corporate strategy. A discussion of key risks to the plans and strategy as well as risk mitigation plans and activities is led by our Chief Executive Officer as part of the meeting. The involvement of the Board in setting our business strategy is critical to the determination of the types and appropriate levels of risk undertaken by the company. As stated in our Corporate Governance Guidelines, our Board is responsible for risk oversight as part of its fiduciary duty of care to effectively monitor business operations.operations effectively. Our Board administers its strategic planning and risk oversight function as a whole and through its Board Committees. For example, theThe following are examples of how our Board Committees are involved in this process:
It is the policy of our Board that a substantial majority of its members be independent from management, and the Board has adopted independence standards that meet the listing standards of the New York Stock Exchange. In accordance with our Corporate Governance Guidelines, our Board undertook its annual review of director independence. Our Board considered any and all commercial and charitable relationships of directors, including transactions and relationships between each director or any member of his or her immediate family and Bristol-Myers Squibb and its subsidiaries, which are described more fully below. Following the review, our Board determined, by applying the independence standards contained in the Corporate Governance Guidelines, that each of our directors and each
director nominee for election at this Annual Meeting is independent of Bristol-Myers Squibb and its management in that none has a direct or indirect material relationship with our company, except for Lamberto Andreotti.Andreotti and Giovanni Caforio, M.D. Mr. Andreotti isand Dr. Caforio are not considered an independent directordirectors because he isthey are currently employed by our company.
In determining that each of Lewis B. Campbell, James M. Cornelius, Laurie H. Glimcher, M.D., Michael Grobstein, Alan J. Lacy, Thomas J. Lynch, Jr., M.D., Dinesh C. Paliwal, Vicki L. Sato, Ph.D., Gerald L. Storch and Togo D. West, Jr. is independent, the Board considered the following relationships which were deemed immaterial under our categorical standards (see Exhibit A):
Additionally, in determining whether Mr. Cornelius and Mr. Grobstein meetour directors met the applicable independence standards, the Board considered certain payments made to, and certain payments received from, our former subsidiary Mead Johnson Nutrition Company. Mr. Cornelius serves as the non-executive chairman of the board of directors of Mead Johnson and Mr. Grobstein began serving as a member of the board in February 2014. The Board also considered payments we made to a private company where the following relationships which did not fall under our categorical standards:
The Board determined that none of these relationships impairs Mr. Cornelius' or Mr. Grobstein'sthe independence of these directors under the New York Stock Exchange's independence standards or otherwise.
The company's governance documents provide the Board with flexibility to select the appropriate leadership structure for the company. They establish well-defined responsibilities with respect to the Chairman and Lead Independent Director roles, including the requirement that the Board have a Lead Independent Director if the Chairman is not an independent director. This information is set forth in more detail on our website at www.bms.com/ourcompany/governance.
On January 20, 2015, we announced the following organizational and Board leadership changes:
James M. Corneliusthrough August 3, 2015 during which he will be working closely with our new Chief Executive Officer;
The Board has determined to elect a three-year period after his retirement,Lead Independent Director when Mr. Cornelius wasAndreotti becomes our Chairman because Mr. Andreotti will not considered to be an independent director under the independence standards set forth in our Corporate Governance Guidelines (which meet the listing standards of the New York Stock Exchange)Exchange standards of independence due to his priorcurrent service as Chief Executive Officer of the company. During that time, the Board determined that it was appropriate to maintain aThe Lead Independent Director is selected annually by the independent directors. The Lead Independent Director's responsibilities include, among others, presiding at the meetings of independent directors, approving meeting agendas and Lewis B. Campbell,meeting schedules, approving and advising the current ChairChairman as to the quality, quantity and timeliness of information sent to the Board and serving as the principal liaison and facilitator between the independent directors and the Chairman. A more detailed description of the Committee on Directorsroles and Corporate Governance, was annually selected by the Board to serve asresponsibilities of the Lead Independent Director. As noted above, Mr. CorneliusDirector is now independent. As such, the Board determined that aavailable on our website at www.bms.com/ourcompany/governance.
In determining our next Chairman, Chief Executive Officer and Lead Independent Director, role was no longer necessary or required under our Guidelines, and Mr. Campbell stepped down from that role and continues to serve as an independent director. We continue to separate the positions of Chairman and CEO.
In making these Board leadership structure determinations, the Board consideredgave thoughtful and significant consideration to many factors, including the specific needs of the Board and the business, our Corporate Governance Guidelines and the best interests of our stockholders. Our Board believes that in the context of the upcoming transition to a new Chief Executive Officer, it will be in the best interests of the company to have our former Chief Executive Officer become Chairman and work closely with our new Chief Executive Officer to ensure a seamless transition of leadership to support our continued evolution to a diversified specialty biopharma company. In accordance with our Corporate Governance Guidelines, the Board recognized the importance of having a Lead Independent Director to maintain a strong counterbalancing structure to ensure that the Board functions in an appropriately independent manner. The Board believes this structure will continue to provide an effective, high-functioning Board as well as appropriate safeguards and oversight. Our Board will continue to evaluate its leadership structure in light of changing circumstances and will evaluate the Board's leadership structure on at least an annual basis and make changes at such times as it deems appropriate.
Our Board meets on a regularly scheduled basis during the year to review significant developments affecting Bristol-Myers Squibb and to act on matters requiring Board approval. It also holds special meetings when important matters require Board action between scheduled meetings. Members of senior management regularly attend Board meetings to report on and discuss their areas of responsibility. In 2013,2014, the Board of Directors met 7eight times. The average aggregate attendance of directors at Board and committee meetings was over 94%96%. No director attended fewer than 75% of the aggregate number of Board and committee meetings during the period he or she served, except that Mr. Paliwal attended three of the five (60%) Board and Committee on Directors and Corporate Governance meetings held since he joined the Board on July 1, 2013. Both of the meetings Mr. Paliwal missed were held on the same day and Mr. Paliwal had informed the company prior to joining the Board that he would be unable to attend these meetings due to a previously scheduled commitment.served. In addition, our independent directors met six times during 20132014 to discuss such topics as our independent directors determined, including the evaluation of the performance of our current Chief Executive Officer. The Lead Independent Director's responsibilities include, among others, presiding atOfficer as well as the meetingsselection of independent directorsour new Chief Executive Officer, Chairman and Mr. Campbell presided over these sessions while he served as our Lead Independent Director.
Annual Meeting of Stockholders
Directors are strongly encouraged, but not required, to attend the Annual Meeting of Stockholders. All of the 20132014 nominees for director attended our 20132014 Annual Meeting of Stockholders except for Mr. Andreotti (who was attending to a family emergency) and Dr. Glimcher (whoand Mr. Lacy who each had a long-standing previous commitment).commitments.
Our Bylaws specifically provide for an Audit Committee, Compensation and Management Development Committee, and Committee on Directors and Corporate Governance, which are composed entirely of independent directors. Our Board of Directors has determined, in its judgment, that all members of the Audit Committee are financially literate and that all members of the Audit Committee meet additional, heightened independence criteria applicable to directors serving on audit committees under the New York Stock Exchange listing standards. In addition, our Board has determined that Messrs. Grobstein, Lacy and Storch each qualify as an "audit committee financial expert" under the applicable SEC rules.
Our Bylaws also authorize the establishment of additional committees of the Board and, under this authorization, our Board of Directors established the Science and Technology Committee. Our Board has appointed individuals from among its members to serve on these four committees and each committee operates under a written charter adopted by the Board, as amended from time to time. These charters are published on our website at www.bms.com/ourcompany/governance. Each of these Board committees has the necessary resources and authority to discharge its responsibilities, including the authority to retain consultants or experts to advise the committee.
On March 7, 2013, the Board established a Securities Issuance Committee to determine and approve the terms and provisions of securities issued by the company during the fourth quarter of 2013. The members of the Securities Issuance Committee were James M. Cornelius, Lamberto Andreotti and Lewis B. Campbell. The Securities Issuance Committee met once during 2013.
The table below indicates the current members of our Board Committees and the number of meetings held in 2014:
Director |
| Audit | Committee on Directors and Corporate Governance | Compensation and Management Development | Science and Technology(4) | |||||
| | | | | | | | | | | |
| | | | | | | | | | | |
Lamberto Andreotti(1) | ||||||||||
Giovanni Caforio, M.D. | ||||||||||
Lewis B. Campbell | | C | X | | ||||||
James M. Cornelius(2) | ||||||||||
Laurie H. Glimcher, M.D. | X | | | X | ||||||
Michael Grobstein | X | X | ||||||||
Alan J. Lacy | C | X | | | ||||||
Thomas J. Lynch, Jr., M.D. | X | X | ||||||||
Dinesh C. Paliwal | X | X | | | ||||||
Vicki L. Sato, Ph.D. | X | C | ||||||||
Gerald L. Storch | X | | X | | ||||||
Togo D. West, Jr.(3) | X | C | ||||||||
Number of 2014 Meetings | 6 | 3 | 6 | 8 | ||||||
Audit Committee
The primary functions of this Committee are:
| |||||||||||||||||
| |||||||||||||||||
related work regarding our financial statements and the effectiveness of our internal control over financial reporting;
| |||||||||||||||||
| |||||||||||||||||
| |||||||||||||||||
Our Board of Directors has determined, in its judgment, that all members of the Audit Committee are financially literate and that all members of the Audit Committee meet additional, heightened independence criteria applicable to directors serving on audit committees under the New York Stock Exchange listing standards. In addition, our Board has determined that Messrs. Grobstein, Lacy and Storch each qualify as an "audit committee financial expert" under the applicable SEC rules.
Committee on Directors and Corporate Governance until his retirement from
The primary functions of this Committee are:
Compensation and Management Development Committee
The primary functions of this Committee are:
Science and Technology Committee until their retirements from the
The primary functions of this Committee are:
Beginning on May 6, 2014, the members and chairs of the Board's four committees will be as follows:
Compensation Committee Interlocks and Insider Participation
There were no Compensation and Management Development Committee interlocks or insider (employee) participation in 2013.2014.
Risk Assessment of Compensation Policies and Practices
We annually conduct a worldwide review of our material compensation policies and practices. Based on this review, we have concluded that our material compensation policies and practices are not reasonably likely to have a material adverse effect on the company. On a global basis, our compensation programs contain many design features that mitigate the likelihood of inducing excessive risk-taking behavior. These features include:
Our Corporate Governance Guidelines contain Board membership criteria that apply to nominees for a position on our Board of Directors, including candidates recommended by stockholders in accordance with the procedures described below. Under these criteria, members of our Board should be persons with broad experience in areas important to the operation of our company such as business, science, medicine, finance/accounting, law, business strategy, crisis management, corporate governance, education or government and should possess qualities reflecting integrity, independence, leadership, good business judgment, wisdom, an inquiring mind, vision, a proven record of accomplishment and an ability to work well with others. The Board believes that its membership should continue to reflect a diversity of gender, race and ethnicity.
Identification and Selection of Nominees for our Board
The Committee on Directors and Corporate Governance periodicallyregularly assesses the appropriate size and composition of our Board,Board. The Committee also considers succession planning for directors. When looking to identify and whether any vacancies on our Board are expected due to retirement or otherwise. In the event that vacancies are anticipated or otherwise arise,select a potential new director, the Committee on Directors and Corporate Governance considers candidates for Board membership. Candidates may come to the attention of the Committee on Directors and Corporate Governance through current Board members, third-party search firms, management, stockholders or others. The Chair of the Committee on Directors and Corporate Governance, in consultation with the Chairman, conducts an initial evaluation of the prospective nominees against the established Board membership criteria discussed above. The Committee reviews the skills of the current directors and compares them to the particular skills of potential candidates, keeping in mind its commitment to maintain a Board with members of diverse experience and background. Candidates may come to the attention of the Committee on Directors and Corporate Governance through current Board members, third-party search firms, management, stockholders or others. The Committee on Directors and Corporate Governance, in consultation with the Chairman, conducts an initial evaluation of the prospective nominees against the established Board membership criteria discussed above. Additional information relevant to the qualifications of prospective nominees may be requested from third-party search firms, other directors, management or other sources. After this initial evaluation, prospective nominees may be interviewed by telephone or in person by the Chairmembers of the Committee on Directors and Corporate Governance, the Chairman, the Lead Independent Director and other directors, as applicable. After completing this evaluation and interview, the Committee on Directors and Corporate Governance makes a recommendation to the full Board as to the persons who should be nominated by our Board, and the full Board determines the nominees after considering the recommendation and any additional information it may deem appropriate. Mr. Paliwal and Dr. Lynch, who joined the Board on July 1, 2013 and January 1, 2014 respectively, were initially identified as potential candidates for election to our Board by a third-party search firm retained by the Committee on Directors and Corporate Governance.
Stockholder Nominations for Director
The Committee on Directors and Corporate Governance considers and evaluates stockholder recommendations of nominees for election to our Board of Directors in the same manner as other director nominees. Stockholder recommendations must be accompanied by disclosure including written information about the recommended nominee's business experience and background with consent in writing signed by the recommended nominee that he or she is willing to be considered as a nominee and, if nominated and elected, he or she will serve as a director. Stockholders should send their written recommendations of nominees accompanied by the required documents to the principal executive offices of the company addressed to: Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Corporate Secretary.
Our Board of Directors has nominated eleven current directors, Lamberto Andreotti, Giovanni Caforio, M.D., Lewis B. Campbell, James M. Cornelius, Laurie H. Glimcher, M.D., Michael Grobstein, Alan J. Lacy, Thomas J. Lynch, Jr., M.D., Dinesh C. Paliwal, Vicki L. Sato, Ph.D., Gerald L. Storch and Togo D. West, Jr., to serve as directors of Bristol-Myers Squibb. The directors will hold office from election until the 20152016 Annual Meeting.
A majority of the votes cast is required to elect directors. Any current director who does not receive a majority of votes cast must tender his or her resignation as a director within 10 business days after the certification of the stockholder vote. The Committee on Directors and Corporate Governance, without participation by any director tendering his or her resignation, will consider the resignation offer and recommend to the Board whether to accept it. The Board, without participation by any director tendering his or her resignation, will act on the Committee's recommendation at its next regularly scheduled meeting to be held within 60 days after the certification of the stockholder vote. We will promptly disclose the Board's decision and the reasons for that decision in a broadly disseminated press release that will also be furnished to the SEC on Form 8-K. If any nominee is unable to serve, proxies will be voted in favor of the remainder of those nominated and may be voted for substitute nominees, unless our Board of Directors provides for a lesser number of directors.
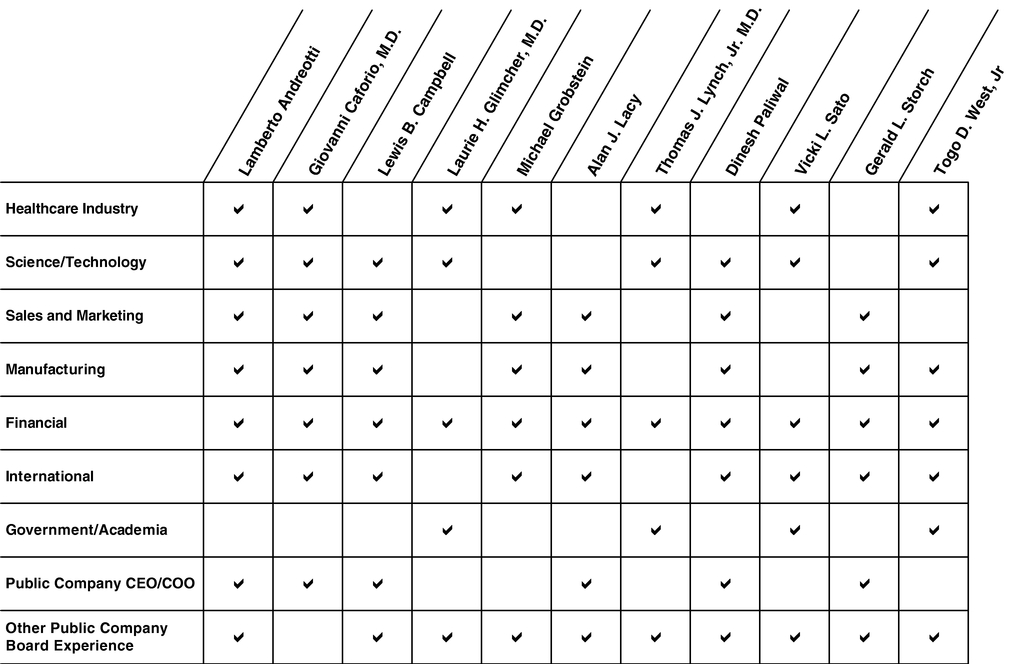
Listed below isThe following pages contain certain biographical information offor each of the nominees for election including his or her principal occupation and current directorships and former public company directorships held during the past 5 years of public companies and registered investment advisors and other business affiliations.years. Also included is a description of the specific experience, qualifications, attributes and skills of each nominee that led the Board to conclude that each nominee is well-qualified to serve as a member of our Board of Directors. The matrix below summarizes the variety of experiences, qualifications, attributes and skills of our nominees:
All Director Nominees Possess: | ||||||
• Leadership | • Sound business judgment | • Innovative thinking | • Integrity | |||
 | ||||||
| | | |
| LAMBERTO ANDREOTTI Mr. Andreotti, age With his Mr. Andreotti is a Director of E. I. du Pont de Nemours and Company. | |
| | | |
| GIOVANNI CAFORIO, M.D. Dr. Caforio, age 50, has been our Chief Executive Officer-Designate since January 2015 and our Chief Operating Officer since June 2014. He served as Executive Vice President and Chief Commercial Officer from November 2013 to June 2014. From October 2011 to November 2013, he served as President, U.S. He held the With over 25 years of pharmaceutical industry experience, including more than 14 years at the company, Dr. Caforio has overseen the creation of a fully integrated worldwide commercial organization as part of our continued evolution into a diversified specialty biopharma company. A physician by training, Dr. Caforio has worked across many businesses within the company, including Europe and the U.S., and has a proven record of developing talented leaders with the diverse experiences and competencies needed for the continued success of the company. | |
| | | |
| | | |
| LEWIS B. CAMPBELL Mr. Campbell, age Mr. Campbell is a demonstrated leader with keen business understanding. With his executive level experience at Textron and Navistar, Mr. Campbell is uniquely positioned to help guide the company as we continue to build a strong foundation for success as a biopharmaceutical company. Furthermore, his first-hand knowledge of the many issues facing public companies and his current and past service as a member of each of our independent Board Committees position him well to serve as the Chair of the Committee on Directors and Corporate Mr. Campbell is on the Board of Directors of Sensata Technologies Holding N.V. | |
| | | |
|
| |
| LAURIE H. GLIMCHER, M.D. Dr. Glimcher, age Dr. Glimcher serves on the Board of Trustees of Cornell University, the Board of Overseers of Weill Cornell Medical College, the Memorial Sloan-Kettering Cancer Center Board of Overseers and on the Board of Trustees of the New York Blood Foundation. Dr. Glimcher also serves on the Scientific Advisory Boards of Cancer Research Institute, Health Care Ventures, Inc. Dr. Glimcher is an internationally known immunologist and physician who brings a unique perspective to our Board on a variety of healthcare related issues. Her expertise in the immunology area and her extensive experience in the medical field position her well to serve as a member of our Science and Technology Committee. Additionally, her experience as the Dean of a major medical school positions her well to serve as a member of our Audit Committee. Dr. Glimcher is a Director of Waters Corporation. | |
| | | |
| | | |
| MICHAEL GROBSTEIN Mr. Grobstein, age With over 30 years experience at a major auditing firm, Mr. Grobstein has extensive knowledge and background relating to accounting and financial reporting rules and regulations as well as the evaluation of financial results, internal controls and business processes. Mr. Grobstein's depth and breadth of financial expertise and his experience handling complex financial issues position him well to serve as a member of our Audit and Compensation and Management Development Committees. Mr. Grobstein is a Director of Mead Johnson Nutrition Company. During the last five years, Mr. Grobstein was a Director of Given Imaging Ltd. | |
| |
| | ||
| | ||
| ALAN J. LACY Mr. Lacy, age Mr. Lacy is a highly respected business leader with a proven record of accomplishment. He brings to the Board extensive business understanding and demonstrated management expertise having served in key leadership positions at Sears Holdings Corporation, including Chief Executive Officer. In addition, his experience as a senior financial officer of three large public companies provides him with a comprehensive understanding of the complex financial, legal and corporate governance issues facing large companies and positions him well to serve as Chair of our Audit Committee and a member of our Committee on Directors and Corporate Governance. Mr. Lacy is | |
| | | |
| | | |
| THOMAS J. LYNCH, JR., M.D. Dr. Lynch, age Dr. Lynch is an internationally recognized oncologist known for his leadership in the treatment of lung cancer with a special interest in personalized medicine. His experience as a practicing physician, During the last 5 years, Dr. Lynch was a Director of Infinity Pharmaceuticals, Inc. | |
| |
| | ||
| | ||
| DINESH C. PALIWAL Mr. Paliwal, age Mr. Paliwal brings to the Board extensive leadership, business and governance experience having served as a public company chief executive officer and a senior executive officer of various divisions of a multinational corporation. His engineering and financial background, together with his worldwide experience, particularly in emerging markets, provide him with a heightened understanding of the complex issues which arise in the global marketplace. In addition, Mr. Paliwal's prior service During the last 5 years, he was a Director of ADT Corporation and Tyco International, Ltd. | |
| | | |
| | | |
| VICKI L. SATO, PH.D. Dr. Sato, age Dr. Sato's extensive and distinctive experience in business, academia and science over more than Dr. Sato is a Director of PerkinElmer Corporation, | |
| |
| | ||
| | ||
| GERALD L. STORCH Mr. Storch, age A retail veteran with more than 20 years of experience, Mr. Storch provides the Board with valuable business, leadership and management insight, including expertise leading an organization with global operations, giving him a keen understanding of the issues facing a multinational business. These qualities make him a valued member of our Audit Committee. Additionally, his prior service on the compensation committee of another public company positions him well to serve as a member of our Compensation and Management Development Committee. Mr. Storch is the Non-Executive Chairman of the Board of Directors of Supervalu, Inc. | |
| | | |
| | | |
| TOGO D. WEST, JR. Secretary West, age Secretary West's legal, business and government experience provides the Board with a unique perspective of the issues facing our company. In his position as Secretary of Veterans Affairs, he was a member of the President's Cabinet, and oversaw the largest healthcare system in the country; and as Secretary of the Army, he was responsible for all Army activities, including the extensive system of Army medical centers around the world. In 2007, Secretary West was asked to co-chair the review of the delivery of healthcare at Walter Reed Army Medical Center and the National Naval Medical Center at Bethesda. With his keen understanding of the need to attract and retain talented employees and the public policy issues facing the healthcare industry, Secretary West is positioned well to serve as Chair of our Compensation and Management Development Committee and as a member of our Committee on Directors and Corporate Governance. Furthermore, his first-hand knowledge of the many issues facing public companies positions him well to serve as our Lead Independent Director effective May 5, 2015. Secretary West is a Director of FuelCell Energy, Inc. and Krispy Kreme Doughnuts, Inc. During the last 5 years, he was a Director of AbitibiBowater Inc. | |
| | | |
Communications with our Board of Directors
The Committee on Directors and Corporate GovernanceOur Board has created a process by which an interested party mayfor anyone to communicate directly with our Board, any committee of the Board, the non-management directors.directors of the Board collectively or any individual director, including our Chairman and Lead Independent Director, if any. Any interested party wishing to contact our non-management directorsBoard may do so in writing by sending a letter c/o Corporate Secretary, Bristol-Myers Squibb Company, 345 Park Avenue, New York, NY 10154.
Any matter relating to our financial statements, accounting practices or internal controls should be addressed to the Chair of the Audit Committee. All other matters should be addressed to the Chair of the Committee on Directors and Corporate Governance. Our Corporate Secretary or her designee reviews all correspondence and forwards to the addressee all correspondence determined to be appropriate for delivery. The Board has determined that certain items which are of a personal nature or not related to the duties and responsibilities of the Board should not be forwarded, including, but not limited to, junk mail and mass mailings (except those that may involve a reputational risk to the company); customer correspondence, including product complaints and inquiries; new product suggestions; resumes and other forms of job inquiries; opinion surveys or polls; business solicitations or advertisements; or obscene, threatening, harassing or similarly inappropriate materials. Our Corporate Secretary periodically forwards to the Chair of our BoardCommittee on Directors and Corporate Governance a summary of all such correspondence and copies of all correspondence that, in the opinion of our Corporate Secretary, deals with the functions of our Board or its committees, or that our Corporate Secretary otherwise determines requires Board attention.received. Directors may at any time review a log of the correspondence we receive that is addressed to members of the Board and request copies of any such correspondence. Our process for handling communications to our Board has been approved by the independent directors.
OurThe Principles adopted by our Board of Directors has adopted the Standards of Business Conduct and Ethics that setsset forth important company policies and procedures in conducting our business in a legal, ethical and responsible manner. These standards are applicable to all of our employees, including the Chief Executive Officer, the Chief Financial Officer and the Controller. In addition, the Audit Committee has adopted the Code of Ethics for Senior Financial Officers that supplements the Standards of Business Conduct and EthicsPrinciples by providing more specific requirements and guidance on certain topics. The Code of Ethics for Senior Financial Officers applies to the Chief Executive Officer, the Chief Financial Officer, the Controller, the Treasurer and the heads of major operating units. Our Board has also adopted the Code of Business Conduct and Ethics for Directors that applies to all directors and sets forth guidance with respect to recognizing and handling areas of ethical issues. The Standards of Business Conduct and Ethics,Principles, the Code of Ethics for Senior Financial Officers and the Code of Business Conduct and Ethics for Directors are available on our website at www.bms.com/ourcompany/governance. We will post any substantive amendments to, or waivers from, applicable provisions of our Standards of Business Conduct and Ethics,Principles, our Code of Ethics for Senior Financial Officers, and our Code of Business Conduct and Ethics for Directors on our website at www.bms.com/ourcompany/governance within two days following the date of such amendment or waiver.
Employees are required to report any conduct they believe in good faith to be an actual or apparent violation of our Codes of Conduct. In addition, as required under the Sarbanes-Oxley Act of 2002, the Audit Committee has established procedures to receive, retain and treat complaints received regarding accounting, internal accounting controls, or auditing matters and the confidential, anonymous submission by company employees of concerns regarding questionable accounting or auditing matters.
The Board has adopted written policies and procedures for the review and approval of transactions involving the company and related parties, such as directors, executive officers and their immediate family members. The policy covers any transaction or series of transactions (an "interested transaction") in which the amount involved exceeds $120,000, the company is a participant, and a related party has a direct or indirect material interest (other than solely as a result of being a director or less than 10 percent beneficial owner of another entity). All interested transactions are subject to approval or ratification in accordance with the following procedures:
As disclosed inThere were no reportable related party transactions during 2014 with any of our proxy statements fordirectors, executive officers or any of their immediate family members. However, BlackRock, Inc. (BlackRock), Wellington Management Group, LLP (Wellington) and The Vanguard Group (Vanguard) are each considered a "Related Party" under our 2012 and 2013 Annual Meetings, the Chairrelated party transaction policy because they each beneficially own more than 5% of our outstanding common stock. The Committee on Directors and Corporate Governance in consultation with the General Counsel,ratified and approved the following related party transaction in 2011 with our former director R. Sanders Williams, M.D., which approval was subsequently ratified by the Committee in 2012 (with Dr. Williams recusing himself from that portion of the meeting),transactions in accordance with our policy and Bylaws:
The Committee on Directors and validate novel targets in Alzheimer's disease. Dr. Williams did not participate inCorporate Governance ratified the above relationships on the basis that these projects while he was a memberentities' ownership of our Board. The agreement was negotiated on an arm's length basis and Dr. Williams was not involvedstock plays no role in the decision-making processbusiness relationship between us and them, and that the engagement of either partyeach entity was on terms no more favorable to them than terms that would be available to unaffiliated third parties under the transaction nor was he involved in any communications between the company and Gladstone.
Additionally, as disclosed in our proxy statement for our 2013 Annual Meeting, the following related party transaction with our former director Louis J. Freeh was deemed to be pre-approved in accordance with our policy and Bylaws:
Pepper Hamilton in 2012 and approximately $5.5 million in 2013. Mr. Freeh did not initiatesame or negotiate the services provided by Pepper Hamilton and the relationship with Pepper Hamilton was entered into in the ordinary course of business prior to Mr. Freeh becoming a partner of the firm. In February 2013, Mr. Freeh was named Chair of Pepper Hamilton.similar circumstances.
Availability of Corporate Governance Documents
Our Corporate Governance Guidelines (including the standards of director independence), Standards of Business Conduct and Ethics,Principles, Code of Ethics for Senior Financial Officers, Code of Business Conduct and Ethics for Directors, additional policies and guidelines, committee charters and links to Reports of Insider Transactions are available on our corporate governance webpage at www.bms.com/ourcompany/governance and are available to any interested partyanyone who requests them by writing to: Corporate Secretary, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154.
2013 Director Compensation Program
We aim to provide a competitive compensation program to attract and retain high quality directors. The Committee on Directors and Corporate Governance annually reviews our directors' compensation practices and compares them against the practices of the companies in our peer group.practices. The Committee submits its recommendations for director compensation to the full Board for approval. Our employee directors do not receive any additional compensation for serving as directors.
In 2012,2013, management engaged an outside consultant, Frederic W. Cook & Co., Inc. (FWC), to review market data and competitive information on director compensation. The Committee requested that FWC analyze the appropriateness of continuing to use the company's executive compensation peer group as the primary data point in determining director compensation. FWC recommended that the executive compensation peer group should be the primary source for determining director compensation. The following companies were in our peer group: AbbVie Inc., Amgen Inc., Biogen Idec Inc., Celgene Corporation, Eli Lilly & Company, Gilead Sciences Inc., Johnson & Johnson, Merck & Co. and Pfizer, Inc.
As further described below, our director compensation program in 2014 was positioned at the median of the executive compensation peer group. Consistent with our desire to attract and retain highly skilled and experienced directors, the Committee on Directors and Corporate Governance, in consultation with FWC, determined that it was appropriate to continue to target director compensation at the median of the companies in our executive compensation peer group for 2013. The following companies were in our peer group: Abbott Laboratories (prior to AbbVie Inc. separation), Amgen Inc., Biogen Idec Inc., Eli Lilly & Co., Gilead Sciences Inc., Johnson & Johnson, Merck & Co. and Pfizer, Inc. As further described below, our director compensation program in 2013 was positioned at the median.2014. The Committee believes the total compensation package for directors we offered in 20132014 was reasonable, and appropriately aligned the interests of directors to stockholders by ensuring directors have a proprietary stake in our company.
In December 2014, the Committee on Directors and Corporate Governance engaged FWC to review market data and prepare analyses that compared our director compensation program against our executive compensation peer group. The componentscompanies in the executive compensation peer group are the same as the companies used to evaluate 2014 director compensation set forth above. Our director compensation program in 2014 was positioned at the median for the executive compensation peer group. Based on this analysis and FWC's recommendation in January 2015 that no changes need to be made to the compensation program for our non-employee directors in 2015, the Committee determined that no changes were necessary at that time. In connection with the organizational and Board leadership changes described beginning on page 8, FWC was subsequently asked to consider Non-Executive Chairman and Lead Independent Director compensation for Mr. Andreotti and Secretary West, respectively. FWC recommended, and the Board approved, compensation as further described below.
The Components of our standard non-management directors' compensation for 2013 were as follows:Director Compensation Program
Cash Compensation
In 2013,2014, our non-management directors were entitled to receive the following cash compensation:
In addition, Mr. Campbell, as the Lead Independent Director through March 3, 2014, received a pro rata amount of an annual cash retainer of $30,000. As of May 5, 2015, Secretary West will be entitled to receive a pro rata amount of an annual cash retainer of $35,000 as Lead Independent Director, which is the median among our peers. The amount of the annual cash retainer paid to Mr. Campbell was set in 2010 and the increase for Secretary West is based on a review of current market data of our peer group.
Deferral Program
A non-management director may elect to defer payment of all or part of the cash compensation received as a director under our company's 1987 Deferred Compensation Plan for Non-Employee Directors. The election to defer is made in the year preceding the calendar year in which the compensation is earned. Deferred funds may be credited to one or more of the following funds: a 6-month United States Treasury bill equivalent fund, a fund based on the return on the company's invested cash or a fund based on the return on our common stock. Deferred portions are payable in a lump sum or in a maximum of ten annual installments. Payments under the Plan begin when a participant ceases to be a director or at a future date previously specified by the director.
Equity Compensation
On February 1, 2013,2014, all non-management directors serving on the Board at that time received an annual award of deferred common share units valued at $160,000 under the 1987 Deferred Compensation Plan for Non-Employee Directors. These deferred common share units are non-forfeitable at grant and are settleable solely in shares of company common stock. A new member of the Board who is eligible to participate in the Plan receives, on the date the Director joins the Board, a pro-rata number of deferred common share units based on the number of share units payable to participants as of the prior February 1.
Share Retention Requirements
All non-management directors are required to acquire at least $300,000 worth of BMS shares and/or deferred share units within three years of joining the Board and to maintain this ownership level throughout their service as a director. We require that at least 25% of the annual retainer be deferred and credited to a deferred compensation account, the value of which is determined by the value of our common stock, until a non-management director has attained our share retention requirements.
Charitable Contribution Programs
Each director who joined the Board prior to December 2009 participates in our Directors' Charitable Contribution Program. Upon the death of a director, we will donate up to an aggregate of $500,000 to up to five qualifying charitable organizations designated by the director. Individual directors derive no financial or tax benefit from this program since the tax benefit of all charitable deductions relating to the contributions accrues solely to us. In December 2009, the Board eliminated the Charitable Contributions Program for all new directors.
Also, each director was able to participate in our company-wide matching gift program in 2013.2014. We matched dollar for dollar a director's contribution to qualified charitable and educational organizations up to $30,000. This benefit was also available to all company employees. In 2013,2014, each of the following directors participated in our matching gift programs as indicated in the Director Compensation Table below: Messrs. Campbell, Cornelius, Freeh,Campbell, Grobstein, Lacy and WestPaliwal and Dr. Williams.Glimcher.
Compensation of the Non-Executive Chairman
On May 4, 2010, Mr. Cornelius retired as our CEO and became ourOur Non-Executive Chairman of the Board. As Non-Executive Chairman, Mr. Cornelius has significantly greater responsibilities than other directors, including chairing the Office of the Chairman to meet on a regular basis with the CEO on the most critical strategic issues and transactions, serving as a liaison between the CEO and the independent directors, frequently discussing the strategy and direction of the company with senior management, and serving as a non-voting member, ex-officio, of the Audit Committee, Committee on Directors and Corporate Governance and the Compensation and Management Development Committee.
Mr. Cornelius has served as our Non-Executive Chairman since May 4, 2010, when he retired as our CEO. In addition to the standard Board compensation that all non-employee directors receive, Mr. Cornelius receives an annual Non-Executive Chairman retainer of $200,000, paid quarterly, of which 50% is paid in cash and 50% in shares of company common stock.
2014 Director Compensation
As described in more detail beginning on page 8, Mr. Cornelius will retire as our Non-Executive Chairman and a member of our Board at our 2015 Annual Meeting of Stockholders on May 5, 2015. The Board has elected Mr. Andreotti to become Executive Chairman of the Board effective May 5, 2015 and Non-Executive Chairman of the Board effective August 3, 2015 following his retirement as an officer of the Company. In December 2013,addition to the Committee on Directorsregular cash Board retainer and Corporate Governance engaged FWC to review market data and prepare analyses that compared our director compensation program against our peer group. The companies in this peer group are the same as the companies in the primary peer group thatannual equity award, Mr. Andreotti will receive an annual Non-Executive Chairman retainer of $200,000, paid quarterly, of which 50% will be usedpaid in cash and 50% in shares of the Company's common stock. As our Non-Executive Chairman, Mr. Andreotti will continue to work closely with the new Chief Executive Officer for evaluating 2014 executive compensationa transition period and are listedhe will receive a Transitional Non-Executive Chairman retainer of $225,000 on page 32. Our director compensation programan annualized basis, paid quarterly, of which 50% will be paid in 2013 was positioned atcash and 50% in shares of the medianCompany's common stock. The Company will also provide Mr. Andreotti with office space, supplies and administrative support for the executive compensation peer group. Based on this analysis and FWC's recommendation in January 2014 that no changes need to be made to the compensation program for our non-employee directors in 2014, the Committee determined that no changes were necessary at that time.Company-related work.
Director Compensation Table
The following table sets forth information regarding the compensation earned by our non-employee directors in 2013. Dr. Lynch, who joined our board on January 1, 2014, did not receive any compensation for 2013.2014.
Name | Fees Earned or Paid in Cash(1) | Stock Awards(2) | Option Awards(3) | All Other Compensation(4) | Total |
| | Fees Earned or Paid in Cash(1) | | Stock Awards(2) | | Option Awards(3) | | All Other Compensation(4) | | Total | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | ||||||||||||||||
| | | | | | | | | | | | | | | | | | | | ||||||||||||||||
L. B. Campbell | $ | 160,000 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 350,000 | L. B. Campbell | $ | 135,242 | $ | 160,000 | $ | 0 | $ | 23,000 | $ | 318,242 | |||||||||||||
J. M. Cornelius(5) | $ | 190,000 | $ | 260,000 | $ | 0 | $ | 30,000 | $ | 480,000 | J. M. Cornelius(5) | $ | 190,000 | $ | 260,000 | $ | 0 | $ | 30,000 | $ | 480,000 | |||||||||||||
L. J. Freeh(6) | $ | 17,823 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 207,823 | ||||||||||||||||||||||||
L. H. Glimcher, M.D. | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 0 | $ | 280,000 | L. H. Glimcher, M.D. | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 20,000 | $ | 300,000 | |||||||||||||
M. Grobstein | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 310,000 | M. Grobstein | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 310,000 | |||||||||||||
A. J. Lacy | $ | 122,500 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 312,500 | A. J. Lacy | $ | 122,500 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 312,500 | |||||||||||||
D. C. Paliwal(7) | $ | 48,750 | $ | 93,336 | $ | 0 | $ | 0 | $ | 142,086 | ||||||||||||||||||||||||
T. J. Lynch, Jr., M.D.(6) | T. J. Lynch, Jr., M.D.(6) | $ | 109,896 | $ | 173,320 | $ | 0 | $ | 0 | $ | 283,216 | |||||||||||||||||||||||
D. C. Paliwal | D. C. Paliwal | $ | 107,292 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 297,292 | |||||||||||||||||||||||
V. L. Sato, Ph.D. | $ | 130,000 | $ | 160,000 | $ | 0 | $ | 0 | $ | 290,000 | V. L. Sato, Ph.D. | $ | 130,000 | $ | 160,000 | $ | 0 | $ | 0 | $ | 290,000 | |||||||||||||
G. L. Storch | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 0 | $ | 280,000 | G. L. Storch | $ | 120,000 | $ | 160,000 | $ | 0 | $ | 0 | $ | 280,000 | |||||||||||||
T. D. West, Jr. | $ | 122,500 | $ | 160,000 | $ | 0 | $ | 15,000 | $ | 297,500 | T. D. West, Jr. | $ | 122,500 | $ | 160,000 | $ | 0 | $ | 0 | $ | 282,500 | |||||||||||||
R. S. Williams, M.D.(8) | $ | 36,753 | $ | 160,000 | $ | 0 | $ | 30,000 | $ | 226,753 | ||||||||||||||||||||||||
Name | Dollar Amount Deferred | Percentage of Deferred Amount Allocated to U.S. Treasury Bill Fund | Percentage of Deferred Amount Allocated to Deferred Share Units | Number of Deferred Share Units Acquired |
| | Dollar Amount Deferred | | Percentage of Deferred Amount Allocated to U.S. Treasury Bill Fund | | Percentage of Deferred Amount Allocated to Company Investment Return Fund | | Percentage of Deferred Amount Allocated to Deferred Share Units | | Number of Deferred Share Units Acquired | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | |||||||||||||
| | | | | | | | | | | | | | | | | | | | |||||||||||||
L. H. Glimcher, M.D. | $ | 120,000 | 100 | % | 0 | % | 0 | L. H. Glimcher, M.D. | $ | 120,000 | | 100 | % | | 0 | % | | 0 | % | | 0 | ||||||||||
M. Grobstein | $ | 60,000 | 0 | % | 100 | % | 1,303 | M. Grobstein | $ | 60,000 | 0 | % | 0 | % | 100 | % | 1,139 | ||||||||||||||
A. J. Lacy | A. J. Lacy | $ | 122,500 | | 100 | % | | 0 | % | | 0 | % | | 0 | |||||||||||||||||
T. J. Lynch, Jr., M.D. | T. J. Lynch, Jr., M.D. | $ | 27,474 | 0 | % | 0 | % | 100 | % | 521 | |||||||||||||||||||||
D. C. Paliwal | $ | 48,750 | 0 | % | 50 | % | 989 | D. C. Paliwal | $ | 107,292 | | 0 | % | | 50 | % | | 50 | % | | 1,017 | ||||||||||
G. L. Storch | $ | 120,000 | 0 | % | 100 | % | 2,606 | G. L. Storch | $ | 120,000 | 0 | % | 0 | % | 100 | % | 2,278 | ||||||||||||||
deferred share units held by each of these directors as of December 31, 20132014 is set forth below. In some cases, these figures include deferred share units acquired through elective deferrals of cash compensation.
Name | | | # of Deferred Share Units | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | |||||
| | | | | | | | |||||
L. B. Campbell | | ||||||||||
J. M. Cornelius | |||||||||||
L. H. Glimcher, M.D. | | ||||||||||
M. Grobstein | |||||||||||
A. J. Lacy | | ||||||||||
T. J. Lynch, Jr., M.D. | 4,045 | ||||||||||
D. C. Paliwal | | ||||||||||
V. L. Sato, Ph.D. | |||||||||||
G. L. Storch | | ||||||||||
T. D. West, Jr. | |||||||||||
| |||||||||||
Name | | | # of Stock Options | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | |||||
| | | | | | | | |||||
L. B. Campbell | | 5,000 | |||||||||
Award Date | Value | Fair Market Value | Shares of Common Stock Acquired | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
3/31/2013 | $ | 25,000 | $ | 41.19 | 606 | |||||
6/30/2013 | $ | 25,000 | $ | 44.69 | 559 | |||||
9/30/2013 | $ | 25,000 | $ | 46.28 | 540 | |||||
12/31/2013 | $ | 25,000 | $ | 53.15 | 470 | |||||
Award Date |
| | Value | | Fair Market Value | | Shares of Common Stock Acquired | |||||
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
3/31/2014 | $ | 25,000 | $ | 51.95 | | 481 | ||||||
6/30/2014 | $ | 25,000 | $ | 48.51 | 515 | |||||||
9/30/2014 | $ | 25,000 | $ | 51.18 | | 488 | ||||||
12/31/2014 | $ | 25,000 | $ | 59.03 | 423 | |||||||
VOTING SECURITIES AND PRINCIPAL HOLDERS
At the close of business on March 14, 2014,13, 2015, there were 1,656,972,493 shares of $0.10 par value common stock and 4,342 shares of $2.00 convertible preferred stock outstanding and entitled to vote.
Common Stock Ownership by Directors and Executive Officers
The following table sets forth, as of March 12, 2014,February 11, 2015, beneficial ownership of shares of our common stock by each director, each of the named executive officers and all directors and executive officers as a group. Shares are beneficially owned when an individual has voting and/or investment power over the shares or could obtain voting and/or investment power over the shares within 60 days. Voting power includes the power to direct the voting of the shares and investment power includes the power to direct the disposition of the shares. Unless otherwise noted, shares listed below are owned directly or indirectly with sole voting and investment power. None of our directors and executive officers, individually or as a group, beneficially owns greater than 1% of our outstanding shares of common or preferred stock.
| | Bristol-Myers Squibb Company | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| | Bristol-Myers Squibb Company | |||||||||||||||||||
| | | | | | | | | | | | | | ||||||||||
Name | Total Common Shares Owned(1) | Common Shares Underlying Options or Stock Units(2) | Common Shares Underlying Deferred Share Units(3) |
| | Total Common Shares Owned(1) | | Common Shares Underlying Options or Stock Units(2) | | Common Shares Underlying Deferred Share Units(3) | ||||||||||||
| | | | | | | | | | | | | | ||||||||||
| | | | | | | | | | | | | | ||||||||||
L. Andreotti | 2,099,304 | 1,450,825 | 0 | L. Andreotti | | 2,019,437 | | 1,636,374 | | 0 | ||||||||||||
C. Bancroft | 277,127 | 112,942 | 0 | C. Bancroft | 291,716 | 193,447 | 0 | |||||||||||||||
G. Caforio, M.D. | 138,026 | 70,580 | 0 | G. Caforio, M.D. | | 150,743 | | 105,344 | | 0 | ||||||||||||
L. B. Campbell | 42,725 | 5,000 | 35,615 | L. B. Campbell | 43,843 | 2,500 | 39,233 | |||||||||||||||
B. Cazala(4) | 254,965 | 0 | 0 | |||||||||||||||||||
J. M. Cornelius | 1,335,908 | 1,035,000 | 27,515 | J. M. Cornelius | | 1,145,378 | | 1,035,000 | | 30,914 | ||||||||||||
F. Cuss, MB BChir, FRCP | 416,123 | 177,677 | 0 | F. Cuss, MB BChir, FRCP | 391,023 | 149,368 | 0 | |||||||||||||||
L. H. Glimcher, M.D. | 78,655 | 0 | 78,655 | L. H. Glimcher, M.D. | | 83,438 | | 0 | | 83,438 | ||||||||||||
M. Grobstein | 53,720 | 0 | 50,337 | M. Grobstein | 58,894 | 0 | 55,511 | |||||||||||||||
A. J. Lacy | 43,107 | 0 | 40,802 | A. J. Lacy | | 46,865 | | 0 | | 44,560 | ||||||||||||
S. Leung | 838,262 | 572,135 | 0 | S. Leung | 899,594 | 633,324 | 0 | |||||||||||||||
T. J. Lynch, Jr, M.D. | 3,447 | 0 | 3,447 | T. J. Lynch, Jr, M.D. | | 6,724 | | 0 | | 6,724 | ||||||||||||
D. C. Paliwal | 6,290 | 0 | 6,290 | D. C. Paliwal | 10,165 | 0 | 10,165 | |||||||||||||||
V. L. Sato, Ph.D. | 42,890 | 0 | 42,890 | V. L. Sato, Ph.D. | | 46,705 | | 0 | | 46,705 | ||||||||||||
E. Sigal, M.D., Ph.D.(5) | 231,263 | 101,478 | 0 | |||||||||||||||||||
G. L. Storch | 18,344 | 0 | 18,344 | G. L. Storch | 23,810 | 0 | 23,810 | |||||||||||||||
T. D. West, Jr. | 38,382 | 0 | 38,382 | T. D. West, Jr. | | 42,075 | | 0 | | 42,075 | ||||||||||||
All Directors and Executive Officers as a Group(6) | 6,638,300 | 3,867,738 | 342,278 | |||||||||||||||||||
All Directors and Executive Officers as a Group(4) | All Directors and Executive Officers as a Group(4) | 5,740,769 | 3,984,800 | 383,136 | ||||||||||||||||||
Principal Holders of Voting Securities
The following table sets forth information regarding beneficial owners of more than 5 percent of the outstanding shares of our common stock. There are no beneficial owners of more than 5 percent of the outstanding shares of our preferred stock.
Name | Number of Shares Beneficially Owned | Percent of Class | | |||||
|---|---|---|---|---|---|---|---|---|
| Capital World Investors 333 South Hope Street Los Angeles, CA 90071 | 108,984,410 | (1) | 6.6% | (1) | ||||
| BlackRock, Inc. 40 East 52nd Street New York, NY 10022 | 105,137,363 | (2) | 6.4% | (2) | ||||
Name |
| Number of Shares Beneficially Owned | Percent of Class | |||||
| | | | | | | | | |
| | | | | | | | | |
Wellington Management Group LLP | 135,151,992(1) | 8.2%(1) | ||||||
BlackRock, Inc. | 109,114,094(2) | 6.6%(2) | ||||||
The Vanguard Group | 91,322,860(3) | 5.5%(3) | ||||||
Section 16(a) Beneficial Ownership Reporting Compliance
Under Section 16(a) of the Securities Exchange Act of 1934, our directors, executive officers and the beneficial holders of more than 10% of our common stock are required to file reports of ownership and changes in ownership with the SEC. To the best of our knowledge, during 20132014 all applicable Section 16(a) filing requirements were met.
Policy on Hedging and Pledging
Our insider trading policy prohibits all employees, including directors and executive officers, from engaging in any speculative or hedging transactions. Our insider trading policy also prohibits all employees, including directors and executive officers, from holding our securities in a margin account or pledging our securities as collateral for a loan except in certain limited circumstances pre-approved by our Corporate Secretary when a person wishes to pledge our securities as collateral for a loan and clearly demonstrates the ability to repay the loan without selling such securities. None of our directors or executive officers (including our NEOs) has pledged shares of our stock as collateral for a loan or holds shares of our stock in a margin account.
Compensation Discussion and Analysis
INTRODUCTION
This Compensation Discussion and Analysis (CD&A) is intended to explain to our stockholders how our compensation program is designed and how it operates with respect tofor our Named Executive Officers (our current CEO, CFO and the three other most highly paid executives of our company and twocompany).
EXECUTIVE SUMMARY
Executive Compensation Program Highlights
Our executive compensation program is designed to align the long-term economic interests of our former executives). Elliott Sigal, M.D., Ph.D.,executives with those of our former Executive Vice Presidentstockholders by rewarding the successful execution of our business strategy and, Chief Scientific Officer, retired fromultimately, the company effective June 30, 2013. On November 13, 2013, Beatrice Cazala ceased serving as Executive Vice President, Commercial Operationsfulfillment of our mission to help patients prevail over serious diseases. In 2014, we enhanced this alignment by placing greater emphasis on the long-term incentive component of pay, adding new metrics to our annual incentive and long-term incentive programs and driving a more accountable culture throughout our leadership to accelerate our ongoing evolution to a diversified specialty biopharmaceutical company. In addition, we engaged in chargedialogue with stockholders to facilitate their understanding of Global Commercialization, Europe and China. As previously announced, Ms. Cazala will be departing from the company on June 9, 2014.
Our CD&A first describes our executive compensation philosophyprogram and to gain insight into how we design our compensation program, with a discussion focusing on the main components of our compensation program. We then detail the process for, and analyze the determination by the Compensation and Management Development Committee (Committee) of, the resultant compensation of our Named Executive Officers. Finally, we discuss 2014 changes to our compensation program, outline other benefits we provide to our Named Executive Officers and describe several of our keythey view executive compensation policies.in general, and our program in particular.
EXECUTIVE SUMMARY
Highlights of our Executive Compensation Program Key 2014 executive compensation highlights:
In addition to the significant changes we made to our executive compensation program in 2014, we continue to retain the following best practices:
As described in more detail below, our Named Executive Officers were compensated based on their individual performance including their continued focus on evolving the company into a specialty care biopharmaceutical business with a focus on innovative products for significant unmet medical needs in
oncology, virology, immunology
2014 Company Performance
2014 was a foundation for the future by shiftingtransformational year in which we achieved significant pipeline and regulatory milestones, realized strong sales growth of our research and development focus, evolving our commercial model, growing our marketedpriority products and investing significantlydivested our global diabetes business. We realigned our organizational structure to simplify operations, increase efficiency, and focus on priority markets. Our transformation to a diversified specialty biopharmaceutical company is well underway and we are leveraging that model to accelerate the speed at which we can bring new medicines to patients while maintaining quality, safety and cost efficiency, all while preserving high ethical standards.
Business development remains a core element of our strategy and in 2014, we executed a substantial number of transactions across multiple disease areas and drug platforms. We also entered into a number of licensing and option transactions in fibrosis and oncology and nine collaborations in immuno-oncology to further study the potential of our pipeline. In addition, we alignedportfolio in combination with other approved products and investigational compounds.
Our strong 2014 performance was attributable to a number of factors, including:
2013 Financial Performance
Our executive compensation program and the compensation of our Named Executive Officers are directly tied to the financial and operational performance of our company. Our solid 2013 financialThe performance was attributable to a number of factors, including:
The financial measures used in our compensation arrangements in 2013 were applicable to the determination of incentive awards for all eligible employees, including our Named Executive Officers. These financial measures are detailed below:2014 were:
| | | | | | | | | | | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Performance Measure | | | Target | | | Actual(1) | | | Actual after Adjustment | | | % of Target | ||||||||||
| | | | | | | | | | | | | | | | | | | | | | | |
Non-GAAP Diluted Earnings Per Share(2) | | $ | 1.72 | | $ | 1.85 | | $ | 1.89 | (3) | | | 109.9 | % | ||||||||
Total Revenues, Net of Foreign Exchange ($=MM)(4) | $ | 15,374 | $ | 16,047 | $ | 16,154 | (3) | 105.1 | % | |||||||||||||
Pipeline Score(5) | | | 3 | | | 4.3 | | | | | | 143.3 | % | |||||||||
Financial Measure | Target | Actual | Actual after Adjustment | Percent of Target | | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Non-GAAP Diluted Earnings Per Share(1) | $ | 1.80 | $ | 1.82 | 101.1 | % | ||||||||
Total Revenues, Net of Foreign Exchange ($=MM)(2)(3) | $ | 16,355 | $ | 16,286 | 99.6 | % | ||||||||
Adjusted Net Cash Flow from Operating Activities ($=MM)(4) | $ | 3,417 | $ | 3,137 | $ | 3,266 | (5) | 95.6 | %(4) | |||||
Total Shareholder Return
Our executive compensation program is designed to align the long-term economic interests of our executives with the interests of our stockholders. This alignment is achieved through our pay-for-performance philosophy that balances strategic, financial, operational, and behavioral objectives. The equity awards granted to all of our executives include performance and service requirements, and our senior executives are required to retain substantial stock ownership in the company.
As shown below, our total shareholder return (stock price appreciation plus dividends), or TSR, for the three and five-year periods ended in 20132014 exceeded that of our peers (includes companies in our primary and extended peer groups) and the S&P 500 Index. In 2013,2014, for the fifthsixth year in a row, we increased our dividend.
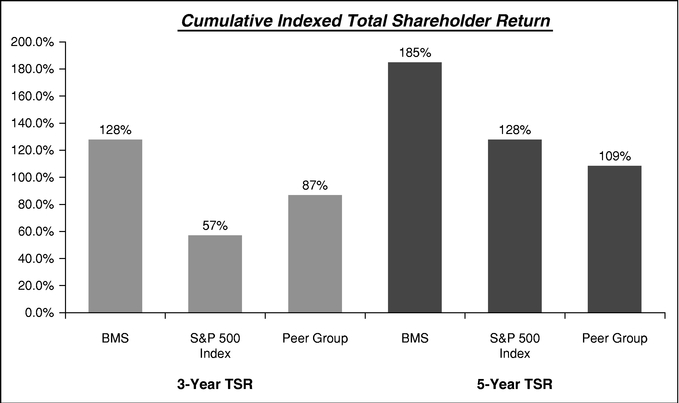
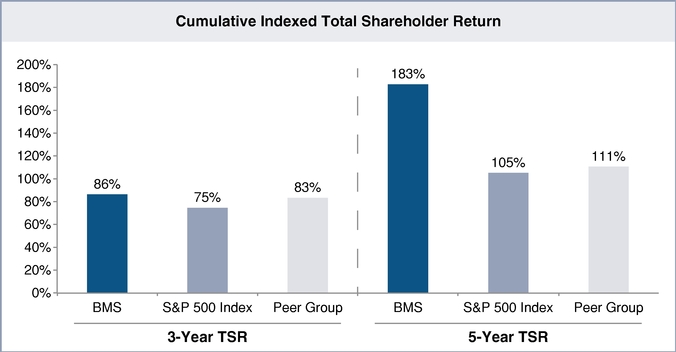
Role of Advisory Vote to Approve Compensation of our Named Executive OfficersOfficers; Stockholder Engagement
We provide our stockholders with the opportunity to cast an annual advisory vote to approve compensation of our Named Executive Officers. At our annual meeting of stockholders held in May 2013,2014, approximately 96.1%86.0% of the votes cast on the proposal at that meeting voted in favor of the proposal. The Compensation and Management Development Committee believes this affirms stockholders' support(the Committee) respects and seeks input from our stockholders and takes their perspective and potential concerns seriously. In 2014, under the direction of the company's approachCommittee, we engaged our largest institutional stockholders holding in the aggregate approximately one-third of our outstanding shares in significant outreach efforts to discuss, among other things, how our executive compensation program is linked to our business strategy and to listen to our stockholders' perspective and obtain feedback on executive compensation generally and BMS' executive compensation policies and practices specifically. In particular, throughout these meetings, management provided the strategic context for the design changes the Committee made to our 2014 executive compensation program. We also analyzed guidance from and held meetings with the two major proxy advisory firms that prepare reports reviewed by certain of our institutional stockholders, and consulted with the Committee's independent compensation consultant regarding evolving market practice. The feedback received on our executive compensation program was summarized and presented to the Committee and much of the input received is reflected in this proxy statement.
These outreach efforts provided us with valuable insights regarding stockholders' views of our compensation program, and we believe provided our stockholders an understanding of how our programs are aligned and support our strategy. The feedback was generally positive. Highlights of these discussions are as follows:
Many of the views expressed by our stockholders reinforced the actions the Committee took with respect to our 2014 executive compensation program, such as the addition of a relative 3-year TSR modifier to our PSU awards and the addition of a pipeline metric to our annual bonus plan, which together have reduced duplication of performance metrics between our annual and long-term incentive plans. We believe that these changes reflect and incorporate many of our key stockholders' views and have helped drive performance and a more accountable culture throughout our leadership. As a result of our stockholder outreach program, we have sought to simplify and enhance certain aspects of our disclosure to assist the reader in more fully understanding our executive compensation program. Since the Committee approved changes to our 2014 executive compensation program prior to the 2014 Annual Meeting, and these changes were generally well received by the stockholders we met with, the Committee did not implement any changes to the 2015 executive compensation program as a direct result of the vote. However, as disclosed in more detail on page 43,vote or the Committee implemented some enhancements to our annual and long-term incentive programs to mitigate the overlap of performance metrics between those two programs and to further align the interests of our executives with the interests of our stockholders. The Committee will continue to consider the outcome of the annual advisory vote to approve compensation when making future compensation decisions for the Named Executive Officers.stockholder outreach.
CEO SUCCESSION
As previously announced, on May 5, 2015, the date of the Annual Meeting, Mr. Andreotti will retire as Chief Executive Officer of the company and Mr. Cornelius will retire as our Non-Executive Chairman. On January 20, 2015, the Board of Directors appointed Giovanni Caforio, M.D. as Chief Executive Officer-Designate and Mr. Andreotti as Chairman-Designate. On May 5, 2015, Dr. Caforio will become the Chief Executive Officer of the company and Mr. Andreotti will become the Executive Chairman of the Board. Mr. Andreotti will also remain an officer of the company for a transition period through August 3, 2015 during which he will work closely with our new Chief Executive Officer. Mr. Andreotti's annual salary and bonus opportunity will remain unchanged during this transition period. Mr. Andreotti did not receive an annual long-term incentive award on March 10, 2015 nor will he receive any further long-term incentive awards in connection with his role as Executive Chairman. On August 3, 2015, Mr. Andreotti will become our Non-Executive Chairman and his Non-Executive Chairman compensation package is described under "Compensation of the Chairman" beginning on page 24.
The Board of Directors has approved Dr. Caforio's new compensation package as Chief Executive Officer, effective May 5, 2015, as detailed below:
Dr. Caforio's new total compensation package is positioned at approximately the 25th percentile among Chief Executive Officers within our current proxy peer group. The Committee believes Dr. Caforio's new compensation package positions him appropriately among his peers when taking multiple factors into consideration, including Dr. Caforio's new tenure as Chief Executive Officer, peers that hold the dual role of Chairman and Chief Executive Officer, and relative company size.
EXECUTIVE COMPENSATION PHILOSOPHY
Our executive compensation philosophy is based on two core elements: to pay for performance and to provide a competitive compensation package and design. Each of these elements is described below:
Pay for Performance: We structure our compensation program to align the interests of our executives with the interests of our stockholders. We believe that an executive's compensation should be directly tied to helping us achieve our mission and deliver value to our stockholders. Therefore, a substantial portion of an executive's compensation is variable and at risk in the form of annual bonus and equity awards that vary in value based on company financial results, our progress against the goals of our strategic and operating plans and our TSR over one or more years. In addition, a significant portion of each executive's pay depends on his or her individual performance against pre-determined strategic, financial and operational objectives as well as key behaviors.behaviors necessary to accelerate our ongoing evolution into a diversified specialty biopharmaceutical company. During 2014, we enhanced our performance-based compensation programs to place a greater emphasis on long-term performance and value-drivers, including the introduction of a relative 3-year TSR modifier in our PSU award plan and a pipeline metric in our annual incentive plan.
Competitive Pay Program: We believe that a competitive compensation program is important to help attract and retain talented employeesexecutives capable of leading our business in the highly complex and competitive business environment in which we operate. We intend to pay our executives at approximately the median level of pay of our peer group when targeted levels of performance are achieved. In certain circumstances, we may target pay above or below the competitive median to help attract or retain executives, as necessary, or to recognize differences in their competencies, qualifications, experiences, responsibilities, contribution, individual performance, role criticality and/or potential. By providing compensation that is competitive with our peer companies, we reduce the risk that our competitors can successfully recruit our executives.executives as well as maintain the highest ongoing level of engagement of these talented executives to facilitate and sustain high performance.
In addition, our compensation program is designed with the following principles in mind:
Additionally,Moreover, the Committee annually reviews the compensation programs from a risk perspective. Based on that review of our executive compensation arrangements as detailed beginning on page 11,12, our Committee believes that our compensation program does not encourage executives to take unreasonable risks that may harm stockholder value. Our compensation program achieves this by striking an appropriate balance between short-term and long-term incentives, using a diversity of metrics to assess performance under our incentive programs, using different forms of long-term incentives, placing caps on our incentive award payout opportunities, following equity grant practices that limit potential for timing awards and having stock ownership and retention requirements.
OUR COMPENSATION PROGRAM DESIGN
This section will explain how we determine the design of our executive compensation program.
Compensation and Management Development Committee
The Committee is responsible for providing oversight of our executive compensation program for the Named Executive Officers as well as other members of senior management. The Committee is responsible for setting the compensation of the Chief Executive Officer and approving the compensation of all of the other Named Executive Officers.
The Committee annually reviews and evaluates the executive compensation program to ensure that the program is aligned with our compensation philosophy and with our performance. The "Committees of Our Board" section on page 8 discusses the duties and responsibilities of the Committee in more detail.
Independent Compensation Consultant
The Committee has retained Compensation Advisory Partners, LLC (CAP) on an annual basis as its independent compensation consultant to provide executive compensation services to the Committee. CAP reports directly to the Committee, and the Committee directly oversees the fees paid for services provided by CAP. The Committee instructs CAP to give advice to the Committee independent of management and to provide such advice for the benefit of our company and stockholders. CAP does not provide any consulting services to BMS beyond its role as consultant to the Committee.
In 2013, CAP provided the following services:
Role of Company Management
The CEO makes recommendations to the Committee concerning the compensation of the other Named Executive Officers and other members of senior management. In addition, the CEO and CFO are involved in recommending for the Committee's approval the financial and business goals that are used as the performance goals for the annual and long-term incentive plans. The head of Human Resources works closely with the Committee, the Committee's independent compensation consultant and management to (i) ensure that the Committee is provided with the appropriate information to make its decisions, (ii) propose recommendations for Committee consideration, and (iii) communicate those decisions to management for implementation.
Peer Group and Benchmarking Analysis
OurIn general, our executive compensation program seeks to provide total direct compensation, when targeted levels of performance are achieved, at the median of the pay levels of our primary peer group, a designated peer group of U.S. companies further described below. In any given year, however, we may target total direct compensation for an executive above or below the median of our primary peer group due to multiple factors, including individual performance results, scope of responsibility and retention. We define total direct compensation as base salary plus target annual incentive bonus plus the fair value of annual long-term incentive awards on the date of grant. With the change in our pay mix and the increased focus on long-term incentives, grantedwe believe the total direct compensation of our Named Executive Officers, as a group, more closely approximates the median of our primary peer group. In any given year, however, we may target total direct compensation for a particular executive above or below the median of our primary peer group due to an executive.multiple factors, including competencies, qualifications, experiences, responsibilities, contribution, individual performance, role criticality and/or potential. We may also target total direct compensation above or below the median of our primary peer group to attract and retain talent within the competitive biopharmaceutical industry marketplace. The Committee's independent compensation consultant annually conducts a review of the compensation for our Named Executive Officers using compensation information compiled from the proxy statementpublicly filed disclosures made by our primary and extended peer groups to assess our overall program. We use pay levels of our peers as a reference point, among others factors, when determining individual pay decisions (i.e., base salary levels, the size of salary adjustments, target bonus amounts and the size of long-term incentive awards). Paying at levels competitive with our peers when targeted levels of performance are achieved allows us to attract and retain the talent we need to run our businesscontinue to drive performance while also enabling us to maintain a competitive cost base with respect to compensation expense. Resulting target compensation for our current Named Executive Officers was between median and 75th percentile of our primary peer group for Mr. Andreotti, Dr. Caforio and approximately atMr. Bancroft, and approximated the 25th percentileand 75th percentiles of our primary peer group for Mr. Bancroft and Dr. Caforio and below the 25th percentile for Dr. Cuss and Ms. Leung.Leung, respectively.
Peer Group
Our primary peer groupgroups in 20132014 consisted of the following companies:
| Primary Peer Group | | Extended Peer Group(1) | ||||
| | | | | | | |
| AbbVie Inc. | Gilead Sciences Inc. | | AstraZeneca PLC | |||
| Amgen Inc. | Johnson & Johnson | GlaxoSmithKline PLC | ||||
| Biogen Idec Inc. | Merck & Co. | | Roche Holding AG | |||
| Celgene Corporation | Pfizer, Inc. | Novartis AG | ||||
| Eli Lilly and Company | | Sanofi | ||||
We believe the companies included in our 2014 primary peer group DETERMINING THE INDIVIDUAL COMPENSATION OF OUR EXECUTIVES Our executive compensation program is designed to provide value to the executive through (i) TSR and share price performance over one or more years, (ii) company performance versus annual financial targets and pipeline goals, and (iii) individual performance against key strategic, financial, and operational objectives, as well as our specified BMS BioPharma Behaviors described below. We believe this approach, with a significant emphasis on long-term and performance-based compensation, serves to focus the efforts of our executives on our long-term business strategy, our mission to help patients prevail over serious diseases, and the attainment of sustained long-term When determining individual award levels, the Committee considers (i) individual performance against strategic, financial and operational objectives that support our long-term business strategy and creating stockholder value ("Results") and (ii) an executive's demonstration of the values and behaviors defined in the Bristol-Myers Squibb Commitment and our BMS BioPharma Behaviors ("Behaviors") identified in the box to the right. The Commitment can be found on our website (www.bms.com). Individual Performance In 2014, we have placed additional emphasis on assessing and promoting individual performance and accountability for all employees, including our Named Executive Officers. Our performance management Each executive is assessed on both "Results" and "Behaviors". The Committee uses these assessments as the basis for making individual compensation decisions. The assessments described below pertain to When determining the individual For Mr. Andreotti, the Committee considered his strong leadership in: (i) delivering For Mr. Bancroft, the Committee considered: (i) his role in the achievement of For Dr. Caforio, the Committee considered (i) his successful transition to the role of For Dr. Cuss, the Committee considered (i) his role in meeting 2014 budget targets while delivering strong pipeline progression, exceeding corporate pipeline goals for registration study starts and submissions includingOpdivoin lung cancer; (ii) bringing several new medicines, additional indications, and key formulations to market including approvals forOpdivoin melanoma in the U.S.,DaklinzaandSunveprain Japan, For Ms. Leung, the Committee considered: (i) her THE COMPONENTS OF OUR The main components of our executive compensation program in the changes the Committee made to our 2014 executive compensation program, the Committee focused on emphasizing variable, or "at risk," compensation for our executives. Our long-term incentive guidelines for executives (other than the CEO) are now expressed as a percentage of salary rather than a fixed dollar amount to sustain our desired pay mix (fixed to variable) over time. This change generally resulted in increases in the size of long-term incentive awards to our Named Executive Officers (other than the CEO). The Committee in conjunction with the changes to our long-term incentive guidelines elected not to provide 2014 annual base salary merit increases to our most senior executive officers, including all of our Named Executive Officers, to further enhance the proportion of pay subject to performance-based incentives. As a result, the proportion of total direct compensation for our Named Executive Officers (other than the CEO) that is subject to performance (i.e., annual cash incentive plus long-term incentive) increased from 71% to 82%, on average. The following This target mix supports the core elements of our executive compensation philosophy by emphasizing long-term, stock-based incentives while providing competitive Base Salary Base salaries are used to help In Annual our mission to help patients prevail over serious diseases. The bonus plan aligns with our business strategy and mission by sharpening management's focus on key financial and Performance Metrics Our 2014 bonus plan design has the following corporate-wide measures, which apply to all employees eligible to participate in the annual incentive plan, including our Named Executive Officers: Pipeline Metric Components We have added the new pipeline metric as a performance measure to highlight the importance of pipeline delivery to the near-term and long-term success of the company. This metric replaces our cash flow metric and incorporates both quantitative and qualitative elements. The metric measures the sustainability and output of our R&D pipeline portfolio and is comprised of goals in two categories, Near-Term Value and Long-Term Growth Potential: The Science and Technology Committee of our Board of Directors provides assistance to the Committee in setting the pipeline performance metric and reviewing the performance results. At the beginning of the year, the Science and Technology Committee recommends to the Committee specific goals in each category in the above chart (for example, a number of regulatory submissions within a given range) and other relevant qualitative factors, including progression of high value assets. Factors considered in the initial determination of a high value asset may include a compound's degree of scientific innovation, its strategic value to the company as well as its financial valuation. After the performance period is completed, the Science and Technology Committee reviews our performance in these categories, including a qualitative assessment of results, and determines a performance score using a scale of 1 to 5, with 3 being target. Once the metric has been determined, the Committee provides final approval of the pipeline performance metric together with the other metrics. Annual Incentive Award A Named Executive Officer's annual incentive award opportunity is expressed as a percentage of base The table below shows the performance and resulting payout percentage of the financial measures used for our Financial Measure Non-GAAP Diluted Earnings Per Share Total Revenues, Net of Foreign Exchange ($=MM) Pipeline Score Total Non-GAAP Diluted Earnings Per Share Total Revenues, Net of Foreign Exchange ($=MM) Adjusted Net Cash Flow from Operating Activities ($=MM) Total The Science and Technology Committee recommended, and the Committee approved, a high value milestone being achieved, including all registrational study starts being initiated with several for nivolumab and two in hepatitis C. The Science and Technology Committee, in the exercise of its discretion in applying the qualitative assessment, also considered other factors, including breakthrough therapy designation granted to nivolumab (Hodgkin lymphoma, melanoma) and elotuzumab (multiple myeloma), as well as promising nivolumab data in multiple tumor types (melanoma, renal cell carcinoma, lung and hematological malignancies). Assuming the achievement of the financial and pipeline measures, the actual bonus an executive receives is then further modified by the executive's individual performance. As described above, individual performance is assessed on The actual annual incentives paid to our Named Executive Officers are shown in the table below and also reported in the Summary Compensation Table in the Non-Equity Incentive Plan Compensation column: Executive Mr. Andreotti Mr. Bancroft Dr. Caforio Dr. Cuss Ms. Leung Mr. Andreotti Mr. Bancroft Dr. Cuss Dr. Caforio Ms. Leung Dr. Sigal(3) Ms. Cazala As set forth in the table above, each Named Executive Officer's target bonus was earned at Long-Term Incentive Program We believe our long-term incentive program serves the best interests of our stockholders by focusing the efforts of our executives on key Annual Equity Award Grants Annual equity awards are typically approved on the date the Committee and full Board meet during the first week of March with a grant effective date of March 10. We believe that consistent timing of equity award grants is a good corporate governance practice that reduces the risk of selecting a grant date with a preferential stock price. Based upon individual performance, an executive may receive a long-term incentive award ranging from 0% to 150% of the target award. In determining the size of the individual long-term incentive awards granted to our Named Executive Officers in March awards ranging between In addition to the new design, the Committee changed the metrics and the weightings to reduce the duplication of the performance metrics used between • Target number of PSUs granted in year one (2013) of the 3-year performance period were divided into three tranches with performance metrics set at the beginning of each performance year • Earned awards banked each year and paid out at end of three-year performance period • Target number of PSUs granted in year one (2014) with performance metrics applicable to the first year only • Earned award, if any, banked after year one and subject to two year holding period • After completion of 3-year performance period, TSR modifier adjusts banked award up or down by up to 20% • EPS (50%) • Total Revenues (25%) • Adjusted Net Cash Flows from Operating Activities (25%) • EPS (70%) • Total Revenues (30%) • BMS extended peer group (see page 35 for listing of 14 companies) • Three-year TSR Modifier based on BMS' percentile rank vs. extended peer group: • Fair value for 2014 and 2015 tranches of award not determined until performance conditions set and communicated in later years • Results in significantly higher accounting expense in later years if stock price is higher than when award was initially granted • Fair value for entire award fixed on grant date, taking into account the TSR modifier • However, for 2014, our reported values are significantly higher than in prior years due to the recognition of the entire 2014-2016 PSU award rather than only one-third of that award, as was the case under our prior design This new PSU design simplifies the administration, communication and executive understanding of the award by measuring financial performance in the first year only, while also mitigating the volatility in total cost of the award because the fair value for the entire award is fixed on the grant date. Under our old PSU awards, the fair value for accounting purposes could not be determined for the later tranches of the award until performance conditions were set in later years. The following is an illustration of the grant date fair value under FASB ASC Topic 718 of the stock awards we are required to report for accounting purposes in our 2014 compensation tables for our CEO as compared to the grant date fair value under FASB ASC Topic 718 of the 2014 stock awards (i.e., the 2014-2016 PSU award and the 2014 MSU award) approved and granted to our CEO by the Committee: The $18.0 million that we are required to disclose as stock award value is $6.4 million higher than the $11.6 million approved and granted by the Committee in 2014 due to the phasing out of the old PSU design and corresponding recognition of prior year award values. In addition, the values of the 3rd tranche of the 2012-2014 PSU award and the 2nd tranche of the 2013-2015 PSU award included in the reported amount is significantly higher than the initial values from their original grant in 2012 and 2013, respectively. This is because the accounting rules require that their value for reporting purposes be determined using the company's current stock price on the date that the performance goals are set (March 10, 2014) for those tranches rather than the stock price on the initial grant dates in March 2012 and March 2013, respectively, which was the original value the Committee intended to deliver at the time the award was granted. Performance Results The Committee established a The following table shows the MSUs vest 25% per year over For illustrative purposes only, the following chart shows the payouts of the first MSU award we granted on March 2, 2010, with a grant date share price of $24.62 (10-day average closing price) and assuming a $1 million award value which is divided The total pre-tax value realized from the MSU over the life of the award was $2,358,523 (total value paid out on all tranches), or a Performance Results We first granted Vesting Date # of Years in Payout Factor March 2, 2010 March 2, 2014 4 200.00% May 4, 2010 May 4, 2010 (Mr. Andreotti only) May 4, 2014 4 200.00% March 1, 2011 March 1, 2014 3 200.00% March 6, 2012 March 6, 2014 2 167.90% March 10, 2013 March 10, 2014 1 145.39% Restricted Stock Units and Stock Options In OTHER ELEMENTS OF In addition to the components set forth above, our senior executives, including all of our Named Executive Officers, were entitled to participate in the following plans or arrangements in Post-Employment Benefits We offer certain plans which provide compensation and benefits to employees who have terminated their employment. These plans are periodically reviewed by the Committee to ensure that they are consistent with competitive practice. The plans offered are common within our primary peer group and enhance our ability to attract and retain key talent. Change-in-Control Arrangements We have entered into change-in-control agreements with certain executives including the CEO and other Named Executive Officers. These agreements enable management to evaluate and support potential transactions that might be beneficial to stockholders even though the result would be a change in control of BMS. Additionally, the agreements provide for continuity of management in the event of a change in control. Our agreements require a "double-trigger" before any payments are made to an executive. This means that payments are only made in the event of a change in control and subsequent involuntary termination or termination for good reason of the employee within 36 months after a change in control for executives who became eligible for change-in-control benefits prior to September 1, 2010, or within 24 months after a change in control for executives who became eligible for change-in-control benefits after September 1, 2010. With respect to our Named Executive Officers, if payments made to a covered officer are subject to excise tax as excess parachute payments by the Internal Revenue Code, then the covered officer is eligible to have the compensation grossed up to fully offset the excise taxes. However, if the payment does not exceed the excise tax threshold by more than 10%, we will reduce the payment so that no portion of the payment is subject to excise tax and no gross-up would be made. As of September 1, 2010, we no longer gross up compensation on excess parachute payments for newly eligible executives. In December 2014, the Committee determined that it would eliminate the remaining excise tax gross-ups provisions in change-in-control agreements for grandfathered executives, including all of our Named Executive Officers. This change will become effective as of January 1, 2016. If a change in control occurs during the term of the agreement, the agreement will continue in effect for either 36 months or 24 months beyond the month in which such change in control occurred depending on whether the executive became eligible for change-in-control benefits before or after September 1, 2010. The value of this benefit for our Named Executive Officers is provided in the "Post-Termination Benefits" section. Severance Plan The Bristol-Myers Squibb Senior Executive Severance Plan provides a competitive level of severance protection for certain senior executives (including the Named Executive Officers) to help us attract and retain key talent necessary to run our company. The value of this benefit for our Named Executive Officers is shown in the "Post-Termination Benefits" section beginning on page 64. Defined Benefit Pension Plans Our defined benefit plans provide income for employees following retirement. The Retirement Income Plan is a tax-qualified plan, as defined under IRS regulations, and the Benefit Equalization Plan relating to the Retirement Income Plan is a non-qualified plan that provides pension benefits above those allowed under the contribution limits for tax-qualified plans. The Summary Compensation Table reflects the annual increase in the actuarial value of these benefits. Current accrued benefits for each of the participating Named Executive Officers are provided in the Pension Benefit Table. As of December 31, 2009, we discontinued service accruals under our qualified and non-qualified pension plans in the U.S. and Puerto Rico for active plan participants, including all of our Named Executive Officers, and the plans to new On September 29, 2014, BMS agreed to transfer certain U.S. pension assets under the Retirement Income Plan to The Prudential Insurance Company of America (Prudential) to settle approximately $1.4 billion of pension obligations. BMS has purchased a group annuity contract from Prudential, which has irrevocably assumed the obligation to make future annuity payments to certain BMS retirees. The transaction will not change the amount of the monthly pension benefit received by affected retirees and surviving beneficiaries. Savings Plans Our savings plans allow employees to defer a portion of their total eligible cash compensation and to receive matching contributions from BMS to supplement their savings and retirement income. The Savings and Investment Program is a tax-qualified 401(k) plan, as defined under IRS regulations, and the Benefit Equalization Plan for the Savings and Investment Program is a non-qualified deferred compensation plan that allows employees to defer a portion of their total eligible cash compensation and to receive matching contributions from BMS in excess of the contributions allowed under the Savings and Investment Program. The savings plans are designed to allow employees to accumulate savings for retirement on a tax-advantaged basis. The company matching contribution under our savings plans equals 100% of the employee's contribution on the first 6% of eligible compensation that an employee elects to contribute. Employees are eligible for an additional automatic company contribution that is based on a point system of an employee's age plus service as follows: below 40 points, the automatic contribution is an additional 3% of total cash; between 40 and 59 points, the contribution is 4.5%; and at 60 points and above, the contribution is 6%. For those employees with 60 or more points who had 10 or more years of service at year-end 2009, we provided an additional automatic contribution of 2% Annual Incentive Deferral Plan We maintain a non-qualified deferred compensation plan for our executives, including our Named Executive Officers. Until we discontinued new deferrals under the plan, effective January 1, 2010, the plan permitted executives to defer up to 100% of their annual cash incentive awards into a choice of two funds: a Bristol-Myers Squibb common stock unit fund and a U.S. Treasury Bill fund. Although we no longer permit new deferrals under the plan, we maintain the plan for executives who made deferrals prior to 2010. We do not pay above-market interest rates on these investments. Upon retirement or termination, plan participants are eligible to receive their deferred amounts based on a previously-selected payout schedule. The Other Compensation OUR COMPENSATION PROGRAM DESIGN PROCESS Compensation and Management Development Committee The Committee is responsible for providing oversight of our executive compensation program for the Named Executive Officers as well as other members of senior management. The Committee is responsible for setting the compensation of the Chief Executive Officer and approving the compensation of all of the other Named Executive Officers and certain other members of senior management. The Committee annually reviews and evaluates the executive compensation program to ensure that the program is aligned with our compensation philosophy and with our performance. The "Committees of our Board" section on page 10 discusses the duties and responsibilities of the Committee in more detail. Independent Compensation Consultant The Committee has retained Compensation Advisory Partners, LLC (CAP) on an annual basis as its independent compensation consultant to provide executive compensation services to the Committee. CAP reports directly to the Committee, and the Committee directly oversees the fees paid for services provided by CAP. The Committee instructs CAP to give advice to the Committee independent of management and to provide such advice for the benefit of our company and stockholders. CAP does not provide any consulting services to BMS beyond its role as consultant to the Committee. In 2014, CAP provided the following services: The Role of Company Management The CEO makes recommendations to the Committee concerning the compensation of the other Named Executive Officers and other members of senior management. In addition, the CEO, CFO and, in the case of our new pipeline performance metric, the Chief Scientific Officer, are involved in recommending for the Committee's approval the performance goals for the annual and long-term incentive Table of CORPORATE POLICIES COVERING EXECUTIVE COMPENSATION Share Ownership and Retention Policy In order to preserve the link between the interests of the Named Executive Officers and those of stockholders, executives are expected to use the shares acquired upon the executives. Our current Named Executive Officers must comply with the following ownership and retention requirements: L. Andreotti C. Bancroft F. Cuss In 2013, we removed Abbott Laboratories from ourand added AbbVie Inc. Abbott spun-off its pharmaceutical business as AbbVie as of January 1, 2013. Additionally,plus these five companies based outside the Committee approved adding Celgene Corporation toU.S.for evaluating 2014 compensation. Celgene is a biopharmaceutical company with a similar business and product focus as BMS and will provide a balance against some of our larger peer companies. We believe this peer group wasare appropriate given the unique nature of the pharmaceutical/biotechnology industry. TheThese companies in our primary peer group varied in size. BMS approximated between the 25th percentile and the median in revenue and at the median in market capitalization amongst our primary peer group. We believe that company size, however, should not be the only factor in determining a peer group. Instead, we believe emphasis should be placed on whether a company competes directly with us for unique pharmaceutical/biotechnology talent. The companies in our 2013 primary peer group represented our primary competitors for executive talent and operated in a similarly complex regulatory and research-driven environment. In determining our primary peer group, we believe emphasis should be placed on whether a company competes directly with us for specialized pharmaceutical/biotechnology talent necessary to progress our evolution to a diversified specialty biopharmaceutical company. We also consider company size in determining our peer group. The companies in our primary peer group varied in size, but all have annual revenues of at least $7 billion for 2014. BMS was slightly below the 25th percentile in revenue and approximated between the 25th percentile and the median in market capitalization amongst our primary peer group. We also reviewed an extended peer group, which is comprised of the nine companies in our primary peer group plus five companies based outside the U.S. The five non-U.S. companies included in our extended peer group are: AstraZeneca PLC, GlaxoSmithKline PLC, Roche Holding Ltd., Novartis AG and Sanofi-Aventis SA. This extended peer group serves as an additional reference point for the Committee given the global nature of our business and the fact that we compete for talent on a global basis. We monitor the composition of our peer groups regularly and make changes when appropriate. This section will explain how the 2013 compensation was determined for each of our Named Executive Officers.growth and profitabilityvalue creation for the benefit of our company and our long-term stockholders while demonstrating high ethical standards.
When determining individual award levels, the Committee considers (i) individual performance against strategic, financial and operational objectives that support our long-term business strategy and TSR ("Results") and (ii) an executive's demonstration of the values and behaviors defined in the Bristol-Myers Squibb Commitment and our BMS BioPharma Behaviors ("Behaviors") and identified in the box to the right. The Commitment can be found on our website (www.bms.com).2013 BMS BioPharma BehaviorsDecide and ActConnect and CollaborateInnovate and ImproveGrow and EngagePerformance Management System
system involvespractices, known as "Three Powerful Conversations," provide an annual review of all executives, includingongoing focus for managers and employees to connect individual objectives and behaviors to the Named Executive Officers, which measures individual performance over the courseevolution of the previous year. This review includesbusiness. Leaders are encouraged to apply a balanced approach to setting objectives: driving results today, while laying the foundation for tomorrow—which is at the heart of our BioPharma strategy. Our objectives define how we are driving the company strategy while our powerful conversations throughout the year create an evaluation ofengaged, empowered and enriched workforce as we continue our evolution into a world class diversified specialty biopharmaceutical company. In particular, the individual goals set by each executive on an annual basis. These individual goals are set within the framework of the company's strategic goals. The system assists in ensuring that each executive's compensation is tied to the key strategic, financial and operational objectives of our company, to stockholder return, and to the executive's demonstration of the BMS BioPharma Behaviors and the values embodied in the BMS Commitment. The Committee conducts the assessment process for our CEO. The CEO conducts the assessment for all of our other Named Executive Officers.Officers and other members of senior management. The assessment for each Named Executive Officer isand the other members of senior management are then reviewed and approved by the Committee.20132014 performance and were used to help the Committee determine the size of each Named Executive Officer's 20132014 annual bonus payment. Prior-year assessments, as disclosed in our 20132014 proxy statement, were used byan input into the Committee to determineCommittee's decision on the size of the 20132014 long-term incentive awards granted in March of 20132014 to each Named Executive Officer.Individual PerformanceTable of Contents20132014 annual incentive payments and the 20142015 long-term incentive awards, the Committee considered each executive's contributions to our company's strategic achievements and financial and operational performance, including their continued focus on evolving the company into a diversified specialty care biopharmaceutical business,company, growing our marketed products, and investing significantly in our pipeline.exceptionalexcellent TSR that exceeded that of many of our peers (includes companies in our primary and extended peer groups) and the S&P 500 Index for bothsince the three and five-year periods ended in 2013, which wasbeginning of his tenure as CEO, supported by solidstrong financial results; (ii) continuing to emphasize organizational management in connection with the company's evolution to a specialty care BioPharma business, as well as developing and recruiting topleadership talent through, among other things, the addition of two new members to the Senior Management Team in 2013 through promotion or external hire; (iii)significantly enhancing the value of our pipeline; (iii) executing robust CEO and key senior management succession plans; (iv) evolving the company's strategy throughfinalizing the divestiture of the diabetes business;business and driving our structural and operating model changes to further enable our company's evolution; and (v) continuing to drive a culture of integrity, andbusiness ethics, compliance, continuous improvement and cost management.solidstrong financial results, including our TSR that exceededcontinues to exceed that of many of our peers (includes companies in our primary and extended peer groups) and the S&P 500 Index for both the three and five-year periods ended in 2013;during his tenure as CFO; (ii) his leadership in the divestitureexecution of the diabetes business to strengthen the company's overall strategy;divestiture, including achieving all milestones and associated financials at or above plan; (iii) his continued role in drivingoversight of a business development screening process and exceeding continuous improvement goals;establishment of a venture capital investment and partnership strategy; (iv) his successful management of the Intercontinental and Japan businesses. The Committee also considered that Mr. Bancroft took on new responsibilities for business development and strategy during 2013 throughteams, including the previously announced management changes. Accordingly, Mr. Bancroft now leads an expanded global organization that integrates financial planning, enterprise-wide strategysigning of six immuno-oncology collaborations and business development. For Dr. Cuss, the Committee considered (i)seven other strategic transactions in 2014; and (v) his successful transition to theongoing role of Executive Vice President and Chief Scientific Officer; (ii) progressing several new product indications, key formulations and geographic submissions including securing EU approval forYervoyin first-line metastatic melanoma, early submission in the EU for daclatasvir, an investigational compound being studied for the treatment of hepatitis C, and accelerated initiation of the Phase III program for an all-oral combination of three BMS investigational products also for the treatment of hepatitis C; (iii) achieving four first-in-human starts and ten Early Candidate Nominations; and (iv) leading and executing the shift of our R&D strategy by, among other things, simplifying and streamlining research governance and evolving the R&D strategy and organization to strengthen our position for future growth.driving continuous productivity initiatives.Executive Vice President and Chief CommercialOperating Officer; (ii) his role in the achievement of solid financial results; (iii) the continued growth of our keydriving strong business performance across brands particularlyYervoy, Sprycel, Orencia, Onglyza/Kombiglyze andBaraclude; (iv) his leadership and role in the launch of(e.g., Eliquis globally,,Yervoy, andSprycel) and regions (e.g., U.S., Japan and Germany) in terms of both sales above budget and competitive in-market performance; (iii) tailoring of effective HCV launches in Japan and Europe; and (iv) delivery of a new, innovative organization that better prepares the company for future launches. As Chief Operating Officer, Dr. Caforio was also instrumental in driving a culture of results, integrity, business ethics, compliance, continuous improvement and cost management. Germany,Daklinzain the U.K.EU,Farxiga*andMyalept*in the U.S., and Canada;a new indication forEliquisin the U.S. and (v) successfully driving significant improvement in our employees' engagementEU; (iii) completion of key business development transactions, strategic immuno-oncology collaborations (e.g., Ono) and the development of his Global Commercial team.multiple immuno-oncology clinical collaborations (e.g., Celldex, Incyte); and (iv) creating a flatter, more accountable R&D organization, simplifying governance and accelerating key programs.performancerole in providing consistently sound legal advice to senior management and the Board of Directors, including critical support related to CEO and Board succession, governance and business development; (ii) promotion and defense of our intellectual property rights; (iii) aligning the divestitureBMS Foundation with the increased complexity of the diabetes businessproduct portfolio; and significant contributions toward(iv) the additionestablishment of twoa new Directorsglobal environmental, health, safety and sustainability structure. Ms. Leung also continues to play a key senior leadership role beyond her areas of direct accountability influencing our Board; (ii) securing, protectingongoing evolution to a diversified specialty biopharmaceutical company and defendingis recognized as a leader within our industry and the Company'sbroader legal rights and interests globally, including overseeing favorable resolutions and outcomes in a numbercommunity.20132014 COMPENSATION PROGRAM20132014 were:in year following endon the third anniversary of three-yearthe award date, where the number of shares banked for an executive will be increased or decreased by up to 20% upon vesting depending on the performance cycle)of BMS' 3-year TSR relative to the TSR of our extended peer group)three- andthree-and four-year performance cycles)periods)chart showscharts provide a comparison of the 2014 and 2013 compensation mixmixes for these elementsthe main components of our executive compensation program for our CEO as well as for our other Named Executive Officers (which is based on the average of targeted compensation for ourthe other Named Executive Officers (other than Dr. Sigal and Ms. Cazala)Officers):
short-term components. Additionally,annual cash components, which further aligns our executive compensation realizedprogram with our biopharmaceutical strategy by our Named Executive Officers hasproviding a significant relationship to the company's stock price because a considerable portionlarger percentage of each Named Executive Officer's compensation is delivered in stock.total pay as at-risk, performance-based compensation. Below, we explain how each of these components is set and how our compensation program continues to be aligned with the core elements of our compensation philosophy. The specific pay decisions with respect to our Named Executive Officers are also detailed.keep us attract and retain talent in a highly competitive and to help us retain talent.labor market. The base salaries of our executives are set based primarily upon the pay levels of comparable positions within our primary peer group and the uniquespecialized qualifications, experience and experiencerole criticality of the individual executives.executive and/or his or her role. Merit increases for our executives are determined based upon both the performance of an individual and the size of our merit increase budget in a given year. We review results of surveys that forecast what other companies' salary increase budgets will be.for other companies. We typically set our annual salary increase budgets based upon the median of such forecasts. Salary adjustments may also be granted from time to time during the year, such as when an executive assumes significant increases in responsibility.2013,2014, under our company-wide salary increase program, employees, including the Named Executive Officers, were eligible for a merit increase provided their performance fully met or exceeded expectations on both Results and Behaviors. Employees rated below the fully-performing level typically receive a reduced merit increase or receive no salary increase depending on the extent to which they were rated below the fully-performing level. Consistent with this policy, Mr. Andreotti, Drs. Sigal and Cuss, and Mmes. Cazala and Leung each received a 3% salary increase effective April 1, 2013. Mr. Bancroft received a 4% salary increase effective April 1, 2013For 2014, the Committee elected not to bring him closerprovide annual merit increases to competitive market levels.any of our most senior executives, including all of our Named Executive Officers, to support the shifting pay mix, as discussed above. Dr. Caforio received a 7%9% salary increase effective January 1, 2013 to bring him closer to competitive market levels and a 3% salary increaseJune 5, 2014 in connection with his promotion effective February 15, 2013 in lieu of a merit increase. Dr. Caforio alsofrom Chief Commercial Officer to Chief Operating Officer. Ms. Leung, our General Counsel and Corporate Secretary, received a 19%1.8% salary increase effective November 13, 2013December 9, 2014 in connection with hisher promotion to Executive Vice President and Chief Commercial Officer. Dr. Cuss received a 42% salary increase effective July 1, 2013 in connection with his promotion to Executive Vice President and Chief Scientific Officer.President.IncentivesIncentive ProgramAnnualOur annual incentive awards areprogram is designed to reward the Named Executive Officers for achieving short-termperformance that supports our business strategy, including our continued evolution to a diversified specialty biopharmaceutical company andoperationalpipeline goals and to reward theiras well as rewarding individual performance, consistent with our pay-for-performance philosophy.2014 Metric and Weighting What It Is Why It's Important
Earnings Per Share (EPS)
(50%)
Non-GAAP Diluted EPS
Net Incomedividedby outstanding shares of common stock
Bottom-Line
A critical measure of annual profitability aligning our employees with our stockholders
Total Revenues
(25%)
Total Revenues, net of foreign exchange
Total revenues minus reserves for returns, discounts, rebates and other adjustments
Top-Line
The foundation of long-term sustainable growth and competitive superiority
Pipeline (New for 2014)
(25%)
• Near-Term Value
(submissions and approvals)
• Long-Term Growth
Potential
Value and Growth
Increases BMS-wide focus on delivery of our late-stage pipeline and development of a robust earlier pipeline through both internal efforts and external sourcesMetric What It Is Why It's Important
Near-Term Value (50%)
Regulatory submissions and approvals for new medicines and marketed products in U.S., EU, and Japan
Recognizes delivery of the late-stage pipeline, which drives near-term value
Long-Term Growth Potential (50%)
• Development Candidates
• First in Human
• Proof of Confidence
• Registrational Study Starts
Recognizes the progression and successes of the R&D pipeline at various stages of development including internal and externally sourced compoundssalary as determined by the individual's grade level. Under our 2013 bonus plan design, three corporate-wide measures—non-GAAP diluted earnings per share (weighted 50%), total revenues, net of foreign exchange (weighted 25%) and adjusted net cash flow from operating activities (weighted 25%)—determine our bonus pool.salary. Individual target bonuses, in turn, may be increased or decreased based upon our company's performance against thesethe corporate-wide measures.performance measures described above. Overall individual results can modify an award from 0% to 150%, and if a pre-specified amount above target plan is achieved, individual results can range from 0% to 165%. The maximum bonus opportunity for a Named Executive Officer based on company and individual results is therefore 251% of target.target (152% × 165% = 251%).
Current Structure of Annual Incentive Award for Named Executive Officers

20132014 bonus plan: Target Performance % of
Target Resulting
Payout
Percentage $ 1.72 $ 1.89 (1) 109.9 % 132.15% $ 15,374 $ 16,154 (1) 105.1 % 152.17% 3 4.3 143.3 % 133.91% 137.60% Financial Measure Target Actual Percent of Target Resulting Payout Percentage $ 1.80 $ 1.82 101.1 % 101.93% $ 16,355 $ 16,286 99.6 % 98.53% $ 3,417 $ 3,266 (1) 95.6 % 97.94% 100.08% AdjustedNon-GAAP diluted earnings per share and total revenues, net cash flow of foreign exchange, were each adjusted $0.04 and $107 million, respectively, due to the loss of exclusivity ofBaracludefrom operating activitiesTeva's launch of generic entecavir on September 4, 2014. Teva's launch was adjusted by the result of a court decision invalidatingBaraclude'scomposition of matter patent in the United States. The Committee determined that it was appropriate to neutralizeexclude the impact of the early loss of exclusivity because this event adversely impacted performance in an amount that was not determinable when the target was set in the first quarter of 2014.cash flow reclassification triggered by stock compensation accounting rules relatedpipeline score of 4.3 (on a scale of 1 to an excess tax benefit.5) based on the following results:the two dimensions of our performance management system—dimensions—Results and Behaviors. We believe this approach for determining incentive award payments balances the need to consider overall company performance, results specific to an executive's functional area of responsibility, and the executive's ability to achieve results vs. objectives on an individual level while also demonstrating the BMS BioPharma Behaviors. The recommended payments are reviewed and approved by the Committee in the first quarter of the year following the performance year, and the awards are paid byno later than March 15th. Target Bonus Adjusted
Target Bonus(1) Actual
Payout(2) $ 2,550,000 $ 3,508,800 $ 5,614,080 $ 910,520 $ 1,252,876 $ 1,566,095 $ 1,029,575 $ 1,416,695 $ 2,125,043 $ 875,000 $ 1,204,000 $ 1,685,600 $ 695,228 $ 956,634 $ 1,291,456 Executive Target Bonus Adjusted
Target
Bonus(1) Actual
Payout(2) % of
Target $ 2,531,250 $ 2,533,275 $ 3,799,913 150.12% $ 901,765 $ 902,486 $ 1,128,108 125.10% $ 650,238 $ 650,758 $ 748,372 115.09% $ 630,348 $ 630,852 $ 788,565 125.10% $ 674,850 $ 675,390 $ 844,238 125.10% $ 652,612 $ 653,134 $ 653,134 100.08% $ 895,199 $ 895,915 $ 895,915 100.08% 100.08%137.60%.(3)Dr. Sigal's target bonus was prorated to reflect his service through June 30, 2013.100.08%137.60% based on their continued focus on our transformation into a leading specialty BioPharma company.overall company performance. Then, an individual performance payout factor was applied to each of the adjusted target bonuses. In determining the individual annual incentives paid to our Named Executive Officers, the Committee considered the 20132014 performance of each executive as described under "Individual Performance" on page 33.36. The Committee approved individual performance factors ranging between 115%125% and 150%160% for eachour Named Executive Officer, other than Dr. Sigal and Ms. Cazala, based on the strong individual performance results described above.Officer.Long-term incentives areLike our annual incentive plan, our long-term incentive program is designed to tiereward performance that supports our evolution to a diversified specialty biopharmaceutical company. It also ties executive interests to the interests of our stockholders. Thestockholders because the ultimate value of long-term awards is driven by the company's stock price, and dividends, which provideprovides a direct link to the creation of stockholder value. In addition, our long-term incentive program is designed to promote retention as well as reward individual performance. In 2013,2014, we offeredcontinued to offer two long-term award vehicles, each of which served a different purpose:internal financial goals;goals and further align executive compensation with the interests of our stockholders through the addition of the new 3-year TSR modifier discussed in more detail below; andvalue and help us retain key talent.value.financial drivers of both short- and long-term success and on TSR while enabling us to reduce expenses.stockholder value. Our long-term incentive program is designedaligned with our strategy and ongoing evolution to achieve the following objectives that support oura diversified specialty biopharmaceutical business strategy:company:entiredesign of our long-term incentive program for executives is performance-based.•The design applies to all levels ofour executives, thus promoting organizational alignment with our biopharmaceutical strategy.WithOur long-term incentive program serves as a retention lever, through vesting and payout spread over several years, including any payouts relating to dividend equivalents, the program promotes greater retention of our executives.•Dividend equivalents that may accrue on performance share units and market share units are earned only when the underlying award is earned.years.Under our long-term incentive program,Beginning with the equity awards granted in March 2014, the Committee established annual equity award guidelines expressed in dollars,for all executives at each grade level underthe company, including our Named Executive Officers other than the CEO, as a percentage of salary rather than a fixed dollar amount. The CEO's long-term incentive program. The value of these guidelines, taken in conjunction withaward level is assessed by the other componentsCommittee annually. In addition, we eliminated dividend equivalents under all of our pay program, enables us2014 annual equity awards, including our PSUs and MSUs, and all equity awards granted thereafter. Our equity award guidelines were adjusted upward to achieve our overall target level of competitive compensation.compensate for the lost dividend value.Typically, onceOnce the grant value is established for each executive, 60% of the value is converted into performance share unitsPSUs and 40% into market share units.MSUs.2013,2014, the Committee considered the prior-year performance of each executive as well as ways to motivate our Named Executive Officers to focus on the company's long-term performance over the next three years and beyond. Each Named Executive Officer other than the CEO had a target value for their long-term incentive award granted in March 2013.2014. The Committee approved individual120%125% and 130%135% of the target value for these Named Executive Officers based on strong individual performance during 2012. The 2013-2015 performance share unit award and the market share unit award granted to Dr. Sigal in March 2013 were forfeited in their entirety in connection with his retirement from the company on June 30, 2013. The CEO's long-term incentive award is not based on a target value and is determined annually by the Committee based on competitive benchmarks and individual performance and contributions. His award took into account his strong performance during 2012.2013.Performance Results Under Our Outstanding Performance Share Unit Awards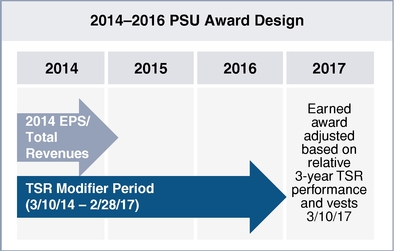
The payoutFor 2014, we revamped our PSU award to simplify the design to enhance executive understanding of performance share unitthe award, as well as to provide additional leverage and further align our executives' interests with those of our stockholders. When the PSU awards is based on three-year performance cycles. Theare granted, the Committee approves the annual performance targets atfor the beginning of eachfirst year of the three-year performance cycle, and awards are payable in the year following the endaward. After completion of the three-year cycle. This design providesfirst year, the target PSUs granted are adjusted based on the company's performance during the year. The PSUs are then banked for a better linktwo-year holding period. Prior to vesting on the third anniversary of the grant date, the award is further adjusted upward or downward by up to 20% based on BMS' three-year TSR relative to its extended peer group. The relative TSR is measured from the award date through the February 28 immediately preceding the vesting date. See the chart showing the new design of our 2014-2016 PSU.
performanceour annual incentive and payout because it eliminates the need to project performance beyond one year. This closer line of sight helps to avoid situations where unforeseen events lead to performance targets that are either overstated or understated and do not appropriately support our pay-for-performance philosophy and retention objectives.long-term incentive plans. Given that the shares underlying our performance share unitPSU awards are not paid out until after the three-year performance cycle has been completed,third anniversary of the grant date and are subject to a relative 3-year TSR modifier, the new awards create an effective balance between short-term motivation to maximize annual financial performance and long-term motivation to create shareholderstockholder value, as well as promote greater retention of our executives. The following chart provides a comparison of our old and new PSU awards: Old (2013-2015) PSU Award New (2014-2016) PSU Award Performance
Period
Performance
Metrics and
Weightings 2013 tranche(1) TSR
Comparator
Group
N/A TSR
Payout Scale N/A Percentile
Rank < 20th 20th < 40th 40th < 60th 60th < 80th ³ 80th TSR
Modifier –20% –10% 0% 10% 20% Accounting
Treatment
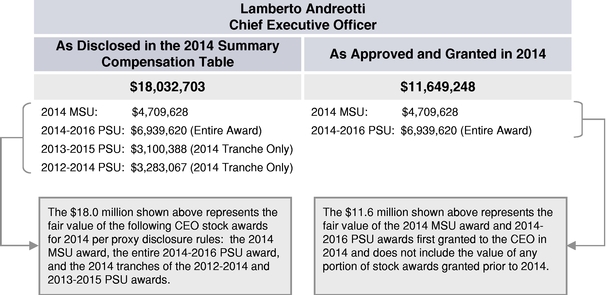
20132014 non-GAAP pretax earnings goal of $2,649.75$2,598 million for the purpose of preserving tax deductibility of 20132014 payouts under this award pursuant to Section 162(m) of the Internal Revenue Code. The company's actual non-GAAP pretax earnings for 20132014 of $3,552$3,835 million exceeded the established goal, thus enabling the 20132014 portion of the 2011-2013, 2012-2014 and 2013-2015 performance share unitPSU awards, and the entire 2014-2016 PSU award, to be earned.2011-2013 Award: The 2013 portion of the 2011-2013 award has the same performance metrics and weights as the 2012 portion of the award: non-GAAP diluted earnings per share (weighted 50%), total revenues, net of foreign exchange (weighted 25%) and adjusted net cash flow from operating activities (weighted 25%). In 2011, working capital and capital expenditures as a percent of net sales was used as our cash flow metric. Upon grant in 2011, the value of our 2011-2013 performance share unit award was enhanced by 50%. All of the enhanced value was built into the 2013 portion of the award. This enhancement represented a one-time increase in total long-term incentive value of 30% (when taking both the value of performance share units and market share units into consideration). The purpose of this enhanced award was to: (a) provide extra incentive for our executives to realize key strategic opportunities over the three-year performance cycle, particularly in light of patent expirations on our largest products; and (b) help retain and reward the talent we need to become a premier growth company in the biopharmaceutical industry in 2014 and beyond. Under the 2013 earnings per share metric, achievement of target yields a payout of 100% of target for that portion of the award; achievement below 85% of target will yield no payout; and achievement levels of 115% or higher of target yields a maximum payout of 167.5% for that portion of the award. Under the 2013 total revenues metric, achievement of target yields a payout of 100% of target for that portion of the award; achievement below 95% of target will yield no payout; and achievement levels of 105% or higher of target will yield a maximum payout of 167.5% for that portion of the award. Under the 2013 adjusted net cash flow from operating activities, achievement of target yields a payout of 100% of target for that portion of the award; achievement below 70% of target will yield no payout; and achievement levels of 130% or higher of target yields a maximum payout of 167.5% for that portion of the award. The 2011-2013 award is payable at 114.26% of target award.banked. The following table summarizes the performance and payout results relating to this award:the 2014 performance metrics applicable to these awards:Year Measure Target Actual % of Target % Payout 2011 EPS $ 2.14 $ 2.28 106.5 % 127.79% Sales ($=MM) $ 20,004 $ 20,583 (1) 102.9 % 137.06% Work Cap + CapEx (%) 11.0 % 8.5 % 122.7 %(2) 167.50% Annual Total 140.04% 2012 EPS $ 1.95 $ 2.01 (3) 103.1 % 111.91% Sales ($=MM) $ 18,018 $ 17,922 (3) 99.5 % 97.66% Adj. NCF from Oper. Act. ($=MM) $ 3,361 $ 3,875 (3) 115.3 % 159.47% Annual Total 120.24% 2013 EPS $ 1.80 $ 1.82 101.1 % 105.00% Total Revenues ($=MM) $ 16,355 $ 16,286 99.6 % 98.42% Adj. NCF from Oper. Act. ($=MM) $ 3,417 $ 3,266 (4) 95.6 % 97.79% Annual Total 101.55% Three-Year Total 114.26% Year Measure Target Performance % of Target % Payout 2014 EPS $ 1.72 $ 1.89 (1) 109.9 % 144.48 % Total Revenues ($=MM) $ 15,374 $ 16,154 (1) 105.1 % 167.50 % Annual Total 151.38 % Reflects the Committee's use of negative discretion to adjust this financial measure downward because theAvalide* supply interruption did not adversely impact performance to the extent originally projected when the target was set in January 2011.(2)Percent of target exceeded maximum performance threshold of 117.1% set forth on the payout curve for our performance share unit awards, resulting in maximum payout percentage.(3)Actuals exclude the net impact of the acquisition of Amylin and related expansion of our AstraZeneca alliance, which was a decrease in non-GAAPNon-GAAP diluted earnings per share of $0.014, an increase in net sales of $262 million and a decrease in adjusted net cash flow from operating activities of $64 million. The Committee also adjusted net sales,total revenues, net of foreign exchange, downward by $1were each adjusted $0.04 and $107 million, becauserespectively, due to the loss of exclusivity ofBaraclude from Teva's launch of generic entecavir on September 4, 2014. Teva's launch was the result of a court decision invalidatingBaraclude's composition of matter patent in the United States. The Committee determined that it was appropriate to exclude the impact of the early loss of exclusivity ofPlavix*because this event adversely impacted performance in Canada didan amount that was not adversely impact performance to the extent originally projecteddeterminable when the target was set in the first quarter of 2012.
2014.(4)Adjusted net cash flow from operating activities was adjusted byPSU Award Payouts EPS (70%) Total Revenues (30%) Performance Description Achievement Payout Achievement Payout Maximum 115% 167.5% 105% 167.5% Target 100% 100% 100% 100% Threshold 85% 42.5% 95% 42.5% Below Threshold <85% 0% <95% 0% Committee to neutralize the impact of a cash flow reclassification triggered by stock compensation accounting rules related to an excess tax benefit.2012-2014 Award: The 2013 portion ofperformance and payout results for the 2012-2014 PSU award has the same performance metrics and weights, and the same performance goals and payout schedules as the 2013 portion of the 2011-2013 award: non-GAAP diluted earnings per share (weighted 50%), total revenues, net of foreign exchange (weighted 25%) and adjusted net cash flow from operating activities (weighted 25%). Below arewell as the performance and banked payout results for the first two years2013-2015 and 2014-2016 PSU awards. For a description of the 2012-2014 award:Year Measure Target Actual % of Target % Payout 2012 EPS $ 1.95 $ 2.01 (1) 103.1 % 111.91% Sales ($=MM) $ 18,018 $ 17,922 (1) 99.5 % 97.66% Adj. NCF from Oper. Act. ($=MM) $ 3,361 $ 3,875 (1) 115.3 % 159.47% Annual Total 120.24% 2013 EPS $ 1.80 $ 1.82 101.1 % 105.00% Total Revenues ($=MM) $ 16,355 $ 16,286 99.6 % 98.42% Adj. NCF from Oper. Act. ($=MM) $ 3,417 $ 3,266 (2) 95.6 % 97.79% Annual Total 101.55% (1)Actuals exclude the net impact of the acquisition of Amylin and related expansion of our AstraZeneca alliance, which was a decrease in non-GAAP diluted earnings per share of $0.014, an increase in net sales of $262 million and a decrease in adjusted net cash flow from operating activities of $64 million. The Committee also adjusted net sales, net of foreign exchange, downward by $1 million because the early loss of exclusivity ofPlavix* in Canada did not adversely impact performance to the extent originally projected when the target was set in the first quarter of 2012.(2)Adjusted net cash flow from operating activities was adjusted by the Committee to neutralize the impact of a cash flow reclassification triggered by stock compensation accounting rules related to an excess tax benefit.2013-2015 Award: The 2013 portion of the 2013-2015 award has the same performance metrics and weights, and the same performance goals and payout schedules as the 2013 portion of the 2011-2013 award: non-GAAP diluted earnings per share (weighted 50%), total revenues, net of foreign exchange (weighted 25%) and adjusted net cash flow from operating activities (weighted 25%). Below are the performance and banked payout results for the first yearperformance metrics relating to the portions of the 2013-2015 award:PSU awards for years prior to 2014, see the proxy statement for our 2014 Annual Meeting available on our website at www.bms.com/ir under the "Financial Information" tab.Year Measure Target Actual % of Target % Payout 2013 EPS $ 1.80 $ 1.82 101.1 % 105.00% Total Revenues ($=MM) $ 16,355 $ 16,286 99.6 % 98.42% Adj. NCF from Oper. Act. ($=MM) $ 3,417 $ 3,266 (1) 95.6 % 97.79% Annual Total 101.55% (1)Adjusted net cash flow from operating activities was adjusted by the Committee to neutralize the impact of a cash flow reclassification triggered by stock compensation accounting rules related to an excess tax benefit. PSUs Banked Performance Period 2012 2013 2014 2015 2016 2017 Total 2012 – 2014
Performance Share Units 120.24% 101.55% 151.38% 124.39%
2013 – 2015
Performance Share Units
101.55%
151.38%
TBD
2015
TBD
2014 – 2016
Performance Share Units
151.38%
HOLD
2015-2016
TSR Modifier
applied
in March
TBD
Performance Results Under Our Outstanding Market Share Unit AwardsMarket share units will
a four-year period,four years, where the number of shares received by an executive upon payout is increased or decreased depending on the performance of our stock price during the one-, two-, three- and four-year performance cycles. On eachperiods. Upon vesting, date, a payout factor is derived as a ratio of the average closing price (i.e., an average of the closing price on the vesting date plus the nine prior trading days) divided by the average stock price on the grant date (also a 10-day average). The payout factor is applied to the target number of unitsMSUs vesting on a given date inclusive of accrued dividend equivalents, to determine the total number of units paid out. If our stock price increases during the performance period, both the number of units and dividend equivalents payable. Novalue of shares that vests increases. If our stock price declines during the performance period, both the number of units and value of shares that are eligible to vest will be reduced. The payout may exceed 200%factor is a ratio of the target units and accrued dividend equivalents payable. If the vesting date stock price (a 10-day average closing price) falls below 60% ofprice on the measurement date divided by the 10-day average closing price on the grant price (also a 10-day average), the target units, inclusive of the accrued dividend equivalents, are forfeited.date. Beginning with our 2013 annual market share unitMSU award grant, on each vestingthe measurement date the payout factor is derived as a ratio of the average of the closing price on the February 28 immediately preceding the vesting date. For MSUs granted in prior years, the measurement date plusis the nine prior trading daysapplicable anniversary of the grant date. The minimum payout performance factor that must be achieved to earn any payout is 60% and the maximum payout factor is 200%. The following chart shows the performance periods for the MSU award granted to our executives in March 2014:
by the average stock price oninto four equal tranches of $250,000: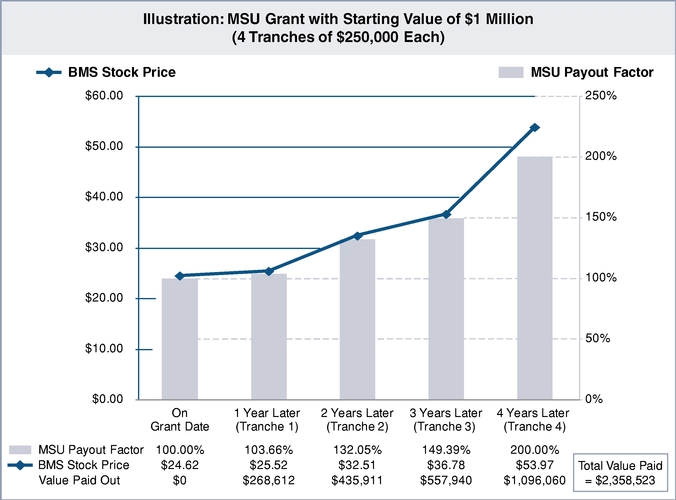
(alsoof March 2, 2010 and the measurement date of March 2, 2011, our stock price rose 3.66%, so the number of MSUs in thefirst tranche that vested on March 2, 2011 was adjusted upward by 3.66%.10-day average).136% increase over the initial pre-tax value. This compares to our TSR of 156% over the same period.market share unitsMSUs to our Named Executive Officers on March 2, 2010. Mr. Andreotti was granted an additional market share unitMSU award on May 4, 2010 in connection with his promotion to the position of Chief Executive Officer. The following table summarizes the payout factors relating to the tranches that vested in 2013 and earlier2014 for ourMSU awards outstanding market share unit awards:at that time:Grant Date Vesting Date # of Months in
Performance Cycle Payout Factor
Performance
Period March 2, 2011 12 103.66% March 2, 2012 24 132.05% March 2, 2013 36 149.39% May 4, 2011 12 113.53% May 4, 2012 24 135.47% May 4, 2013 36 163.33% March 1, 2012 12 127.09% March 1, 2013 24 144.01% March 6, 2013 12 113.82% 2013,2014, we did not grant any service-based restricted stock units to executives as part of our annual long-term incentive program. Restricted stock units were onlymay be granted selectively to executives at other times of the year for purposes of attracting, retaining and providing special recognition. Among our Named Executive Officers, the Committee granted tworecognition, such as when an employee assumes significant increases in responsibility. During 2014, no special restricted stock unit awards in 2013. Dr. Cuss received an award on July 1, 2013 valued at $500,000 in connection with his promotionwere granted to any of our Named Executive Vice President and Chief Scientific Officer. Mr. Bancroft received an award on December 2, 2013 valued at $1,000,000 to recognize his taking on an expanded role that includes the Business Development and Strategy groups. Each of Dr. Cuss's and Mr. Bancroft's awards will vest one-third per year on the third, fourth and fifth anniversaries of the grant date.Officers. We have not granted any stock options to our executives since 2009.20132014 COMPENSATION2013:2014:•Severance Plan•Change-in-Control Arrangements
we stopped addingclosedparticipants to our plans.participants. For active plan participants at year-end 2009, we are allowingallowed five additional years of pay growth in our pension plans. Accordingly, 2014 was the last year of pay growth under our pension plans. These actions were taken to align our retirement program with our new biopharmaceutical business strategy and culture, to mitigate volatility risk to the Company, to respond to the competitiveness of a changing industry, and to meet the mobility and career expectations of an evolving workforce.is provided for a five-year period. Accordingly, contributions with respect to 2014 are the last year of this additional 2% automatic contribution for this group. As of December 31, 2009, each Named Executive Officer other than Drs. Cuss and Caforio had earned over 60 points and had more than 10 years of service. All U.S. employees are eligible to participate in both savings plans. The Summary Compensation Table reflects company contributions to these plans during 20132014 in the All Other Compensation column. The Non-Qualified Deferred Compensation Table provides more detail on the Benefit Equalization Plan for the Savings and Investment Program.Committee may approve accelerated distributions in the event of an unforeseeable emergency. The Non-Qualified Deferred Compensation Table provides more detail on this plan for those Named Executive Officers who participated in previous years.Severance Plan The Bristol-Myers Squibb Senior Executive Severance Plan provides a competitive level of severance protection for certain senior executives (including the Named Executive Officers) to help us attract and retain key talent necessary to run our company. The value of this benefit for our Named Executive Officers is shown in the "Post-Termination Benefits" section on page 59.Change-in-Control Arrangements We have entered into change-in-control agreements with certain executives including the CEO and other Named Executive Officers. These agreements enable management to evaluate and support potential transactions that might be beneficial to stockholders even though the result would be a change in control of BMS. Additionally, the agreements provide for continuity of management in the event of a change in control. Our agreements require a "double-trigger" before any payments are made to an executive. This means that payments are only made in the event of a change in control and subsequent involuntary termination or termination for good reason of the employee within 36 months after a change in control for executives who became eligible for change-in-control benefits prior to September 1, 2010, or within 24 months after a change in control for executives who became eligible for change-in-control benefits after September 1, 2010. With respect to our Named Executive Officers, if payments made to a covered officer are subject to excise tax as excess parachute payments by the Internal Revenue Code, then the covered officer is eligible to have the compensation grossed up to fully offset the excise taxes. However, if the payment does not exceed the excise tax threshold by more than 10%, we will reduce the payment so that no portion of the payment is subject to excise tax and no gross-up would be made. As of September 1, 2010, we no longer gross up compensation on excess parachute payments for newly eligible executives. If a change in control occurs during the term of the agreement, the agreement will continue in effect for either 36 months or 24 months beyond the month in which such change in controloccurred depending on whether the executive became eligible for change-in-control benefits before or after September 1, 2010. The value of this benefit for our Named Executive Officers is provided in the "Post-Termination Benefits" section.Except as set forth below, weWe did not provide perquisites and other personal benefits to our Named Executive Officers that were not otherwise available to all salaried employees. On February 21, 2011, Ms. Cazala, our Executive Vice President, Commercial Operations, began a long-term assignment in the United States. Prior to that time, she had been working in the United States on a short-term assignment as a French expatriate. Under the employment letter agreement we entered into with Ms. Cazala in connection with her long-term assignment, the company made payments to continue certain of Ms. Cazala's social insurance and company-sponsored health and welfare benefits in France. Additionally, Ms. Cazala received reimbursement of tax preparation fees related to her past expatriate arrangement and a tax gross up on such amount during 2013, which is a benefit generally available to any of our salaried employees who had been working under an expatriate arrangement. The amounts relating to these benefits are disclosed in the All Other Compensation column in the Summary Compensation Table. As further discussed on page 61, we have entered into a Settlement Agreement and General Release with Ms. Cazala and her employment letter agreement has been terminated.TAX IMPLICATIONS OF EXECUTIVE COMPENSATION PROGRAM Section 162(m) of the Internal Revenue Code includes potential limitations on the deductibility of compensation in excess of $1 million paid to certain Named Executive Officers. A significant portion of the compensation we pay to our Named Executive Officers qualifies as "performance-based compensation" for purposes of Section 162(m) and is, therefore, eligible to be fully deducted by BMS for federal income tax purposes. We view preserving tax deductibility as an important objective, but not the sole objective, in establishing executive compensation. In specific instances, we may authorize compensation arrangements that are not fully tax deductible, but which promote other important objectives. To the extent that compensation paid in 2013 to certain Named Executive Officers, such as salary and distributions pursuant to the vesting of restricted stock units awarded without performance-based vesting conditions, does not qualify for an exception under Section 162(m) and exceeds $1 million in the aggregate, we will not be able to deduct such excess for federal income tax purposes.COMPENSATION PROGRAM CHANGES FOR 2014 We implemented certain changes to our compensation program for 2014 to support the continued evolution of our biopharmaceutical business strategy and culture. These changes are described below:•Added Pipeline Performance Metric to Annual Incentive Awards: Beginning in 2014, we replaced the adjusted net cash flow from operating activities metric under our bonus plan with a pipeline performance metric. This new metric measures the sustainability and output of our pipeline portfolio and consists of two categories: (1) submission and approvals; and (2) long-term growth potential. The Board's Science and Technology Committee will assist the Committee in setting the pipeline performance metric and reviewing the performance results. The metric will be evaluated both quantitatively and qualitatively.•Change in Long-Term Incentive Program Guidelines: Beginning with the equity awards granted in March 2014, our annual equity award guidelines for all executives at the company, including our Named Executive Officers other than the CEO, are expressed as a percentage of salary rather than a fixed dollar amount for each grade level. The CEO's long-term incentive award level will continue to be assessed by the Committee annually.•Elimination of Dividend Equivalents: We have eliminated dividend equivalents under all of our 2014 annual equity awards and all equity awards granted thereafter. Our equity award guidelines were adjusted upward to compensate for the lost value.New Performance Share Unit Award Designreviewed and advised on the composition of the peer group used for 2014 Including a New TSR Modifier: Our 2014-2016 performance share unit award is now tied to two performance metrics with new weightings: non-GAAP diluted earnings per share (weighted 70%) and non-GAAP total revenues, net of foreign exchange (weighted 30%). These performance metrics are established at the beginning of 2014 and measure our financial performance during 2014 only. In March 2015, the Committee will certify the performance metrics, the performance factor will be applied and the resulting performance share units will be banked for a two-year holding period. Prior to vesting in March 2017, a TSR modifier will be applied to the performance share unit award which can increase or decrease the banked shares by up to 20%. The TSR measurement period will be approximately three years and run from the grant date (March 10, 2014) through February 28, 2017. The TSR modifier will be determined by measuring BMS's TSR relative to the TSR of our extended peer group. TSR reflects the change in the value of a company's publicly traded stock over the TSR measurement period, taking into account both stock price appreciation (or depreciation) and the reinvestment of dividends.competitive benchmarking;New Performance Metricsparticipated in the design and Weighting for Outstanding Performance Share Unit Awards: The 2014 portionsdevelopment of the outstanding 2012-2014 and 2013-2015 performance share unit awards will have the same performance metrics and weights as the 2014-2016 performance share unit award: non-GAAP diluted earnings per share (weighted 70%) and non-GAAP total revenues, net of foreign exchange (weighted 30%). Through these changes, we were able to achieve several objectives that support our biopharmaceutical business strategy:•The metrics and weightings used for the Annual Bonus Plan and the performance share unit awards are no longer duplicative.executive compensation program;The new pipeline performance metric balances short-term results with sustainable long-term value creation, while reinforcing the foundational importanceprovided an assessment of pipelineBMS senior executive pay levels and practices relative to all stakeholders—patients, stockholders,peers and employees.other competitive market data;The TSR modifier on our performance share unit awards further improves alignment with stockholders.provided an annual analysis of industry trends among the peers and best practices related to pay program design and other program elements;The new 2014-2016 performance share unit awardreviewed and elimination of dividend equivalents simplifies employee communicationsadvised on all materials provided to the Committee for discussion and administration to further engageapproval; and retain employees.changeCommittee reviews the independence of CAP annually in accordance with the Committee's charter and applicable SEC rules and NYSE listing requirements. After review and consultation with CAP, the Committee has determined that CAP is independent and there is no conflict of interest resulting from retaining CAP currently or during the year ended December 31, 2014.programplans, as applicable. The head of Human Resources works closely with the Committee, the Committee's independent compensation consultant and management to express equity award guidelines as a percentage(i) ensure that the Committee is provided with the appropriate information to make its decisions, (ii) propose recommendations for Committee consideration, and (iii) communicate those decisions to management for implementation.salary further focuses our executives on the company's long-term performance.Contentsexercisevesting of their previously granted(i) restricted stock options,unit awards, (ii) market share unit awards and (iii) performance share unit awards, after satisfying the cost of exercise andapplicable taxes, to establish and maintain a significant level of direct ownership. This same expectation applies to shares acquired upon the vestingexercise of (i) restrictedtheir previously granted stock units, (ii) market share units and (iii) performance share unit awards granted in 2011 and beyond.options. We continue to maintain longstanding share-ownership expectations for our senior Stock Ownership Share Retention Policy—applied to all
shares acquired, net of taxes
2014 ComplianceExecutive Stock OwnershipExecutive Guideline as a
Multiple of Salary Prior to
Achieving Guideline After
Achieving Guideline2013 Compliance with Share Ownership
and Retention Policy 6 x 100% 75% for 1 year Yes 3 x 100% 75% for 1 year Yes C. Bancroft G. Caforio 3 x 100% 75% for 1 year Yes 3 x 100% 75% for 1 year Yes G. CaforioS. Leung 3 x 100% 75% for 1 year Yes F. Cuss 3 x100%75% for 1 yearYesS. Leung
Recoupment of Compensation
We maintain clawback provisions relating to stock options, restricted stock units, performance share units and market share units. Under these clawback provisions, executives that violate non-competition or non-solicitation agreements, or otherwise act in a manner detrimental to our interests, forfeit any outstanding awards, and any accrued and unpaid dividend equivalents underlying these awards, as of the date such violation is discovered and have to return any gains realized in the twelve months prior to the violation. These provisions serve to protect our intellectual property and human capital, and help ensure that executives act in the best interest of BMS and our stockholders.
In 2005, the Board adopted a policy wherein the Board will seek reimbursement of annual incentives paid to an executive if such executive engaged in misconduct that caused or partially caused a restatement of financial results. In such an event, we will seek to claw back the executive's entire annual incentive for the relevant period, plus a reasonable rate of interest. This policy may be viewed on our website atwww.bms.com. www.bms.com.
In December 2012, the Board adopted a policy that BMS will seek recoupment of any bonus and/or other compensation paid to executives and certain other employees after December 4, 2012 where:
In any instance where the employee misconduct occurred in a prior year, the Committee may elect to reduce a current or future bonus and/or other compensation award in lieu of requiring reimbursement of past compensation previously paid to such executive or other employee. This policy may be viewed on our website atwww.bms.com. www.bms.com.
Once the SEC has implemented Dodd-Frank legislation on clawback provisions, we will review and revise our policies, as appropriate, based on such rules.
Equity Grant Policy
The Committee's policy covering equity grants for the Named Executive Officers is as follows.
Approval of Awards
Grant Effective Date
Annual Awards
All Other Awards
In no case whatsoever will the grant effective date precede the approval date of a given award.
Grant Price
Policy Against the Repricing of Stock Options
We have always maintained a consistent policy against the repricing of stock options. We believe this is a critical element in maintaining the integrity of the equity compensation program and ensuring alignment of senior executives' interests with the interests of stockholders. The Board of Directors has adopted a formal policy prohibiting the repricing of stock options without stockholder approval. This policy may be viewed on our website atwww.bms.com. www.bms.com.
Policy Regarding Stockholder Approval of Severance
The Board has approved a policy that requires stockholder approval of any future agreements that provide for cash severance payments in excess of 2.99 times the sum of an executive's base salary plus annual incentive. "Cash severance payments" exclude accrued incentive payments, the value of equity acceleration, benefits continuation or the increase in retirement benefits triggered by severance provisions or tax gross-up payments. This policy may be viewed on our website atwww.bms.com. www.bms.com.
TAX IMPLICATIONS OF EXECUTIVE COMPENSATION PROGRAM
Section 162(m) of the Internal Revenue Code includes potential limitations on the deductibility of compensation in excess of $1 million paid to certain Named Executive Officers. A significant portion of the compensation we pay to our Named Executive Officers qualifies as "performance-based compensation" for purposes of Section 162(m) and is, therefore, eligible to be fully deducted by BMS for federal income tax purposes. We view preserving tax deductibility as an important objective, but not the sole objective, in establishing executive compensation. In specific instances, we may authorize compensation arrangements that are not fully tax deductible, but which promote other important objectives. To the extent that compensation paid in 2014 to certain Named Executive Officers, such as salary and distributions pursuant to the vesting of restricted stock units awarded without performance-based vesting conditions, does not qualify for an exception under Section 162(m) and exceeds $1 million in the aggregate, we will not be able to deduct such excess for federal income tax purposes.
Compensation and Management Development Committee Report
The Compensation and Management Development Committee (Committee) of Bristol-Myers Squibb Company has reviewed and discussed with management the Compensation Discussion and Analysis on pages 2729 to 4654 of this Proxy Statement as required under Item 402(b) of Regulation S-K. Based on its review and discussions with management, the Committee recommended to the full Board that the Compensation Discussion and Analysis be included in this Proxy Statement.
Compensation and Management Development Committee
Togo D. West, Jr., Chair
Lewis B. Campbell
Michael Grobstein
Vicki L. Sato, Ph.D.
Gerald L. Storch
The following tables and notes present the compensation provided to Lamberto Andreotti, Chief Executive Officer, Charles A. Bancroft, Executive Vice President and Chief Financial Officer, and the three other most highly compensated Executive Officers. The tables and notes also include two additional highly compensated executives: Elliott Sigal, M.D., Ph.D., our former Executive Vice President and Chief Scientific Officer, who retired from the company effective June 30, 2013, and Beatrice Cazala, our former Executive Vice President, Commercial Operations in charge of Global Commercialization, Europe and China, who ceased serving in that Executive Officer role effective November 13, 2013.
Summary Compensation TableforFor Fiscal Years Ended December 31, 2014, 2013 2012 and 20112012
Name and Principal Position | Year (1) | Salary (2) | Stock Awards (3) | Non-Equity Incentive Plan Compensation (4) | Change in Pension Value and Non- Qualified Deferred Compensation Earnings (5) | All Other Compensation (6) | Total | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Lamberto Andreotti | 2013 | $ | 1,686,539 | $ | 14,586,898 | $ | 3,799,913 | $ | 0 | $ | 774,396 | $ | 20,847,746 | |||||||||
Chief Executive Officer | 2012 | $ | 1,623,077 | $ | 9,722,004 | $ | 3,844,864 | $ | 1,193,957 | $ | 818,078 | $ | 17,201,980 | |||||||||
| 2011 | $ | 1,510,192 | $ | 7,351,673 | $ | 4,220,340 | $ | 1,179,899 | $ | 649,843 | $ | 14,911,947 | |||||||||
Charles Bancroft | 2013 | $ | 901,092 | $ | 4,778,079 | $ | 1,128,108 | $ | 759,507 | $ | 311,230 | $ | 7,878,016 | |||||||||
EVP and Chief Financial | 2012 | $ | 868,635 | $ | 2,395,753 | $ | 1,321,979 | $ | 3,662,332 | $ | 318,614 | $ | 8,567,313 | |||||||||
Officer | 2011 | $ | 809,712 | $ | 2,151,058 | $ | 1,407,181 | $ | 2,754,087 | $ | 250,363 | $ | 7,372,400 | |||||||||
Francis Cuss, MB, | 2013 | $ | 736,102 | $ | 2,016,197 | $ | 748,372 | $ | 65,331 | $ | 153,035 | $ | 3,719,037 | |||||||||
BChir, FRCP | ||||||||||||||||||||||
Giovanni Caforio, M.D. | 2013 | $ | 748,320 | $ | 1,587,106 | $ | 788,565 | $ | 0 | $ | 177,861 | $ | 3,301,852 | |||||||||
EVP and Chief Commercial Officer | ||||||||||||||||||||||
Sandra Leung | 2013 | $ | 843,087 | $ | 2,883,914 | $ | 844,238 | $ | 0 | $ | 245,048 | $ | 4,816,287 | |||||||||
General Counsel | 2012 | $ | 768,738 | $ | 2,020,399 | $ | 907,256 | $ | 1,953,216 | $ | 243,163 | $ | 5,892,772 | |||||||||
& Corporate Secretary | 2011 | $ | 724,587 | $ | 1,827,335 | $ | 968,140 | $ | 1,758,498 | $ | 213,113 | $ | 5,491,673 | |||||||||
Elliott Sigal, M.D., Ph.D.(7) | 2013 | $ | 564,451 | $ | 5,890,044 | $ | 653,134 | $ | 0 | $ | 340,796 | $ | 7,448,425 | |||||||||
Former EVP and Chief | 2012 | $ | 1,063,209 | $ | 3,219,625 | $ | 1,869,807 | $ | 1,740,960 | $ | 450,082 | $ | 8,343,683 | |||||||||
Scientific Officer | 2011 | $ | 1,034,986 | $ | 2,908,753 | $ | 2,151,663 | $ | 2,518,327 | $ | 418,268 | $ | 9,031,998 | |||||||||
Beatrice Cazala(8) | 2013 | $ | 894,694 | $ | 3,611,980 | $ | 895,915 | $ | 0 | $ | 75,609 | $ | 5,478,198 | |||||||||
Former EVP, Commercial | 2012 | $ | 868,635 | $ | 2,445,800 | $ | 1,126,130 | $ | 3,522,012 | $ | 68,448 | $ | 8,031,025 | |||||||||
Operations | 2011 | $ | 895,100 | $ | 2,602,126 | $ | 1,353,436 | $ | 957,938 | $ | 132,567 | $ | 5,941,167 | |||||||||
| Name and Principal Position | | Year (1) | | Salary (2) | | Stock Awards (3) | | Non-Equity Incentive Plan Compensation (4) | | Change in Pension Value and Non- Qualified Deferred Compensation Earnings (5) | | All Other Compensation (6) | | Total | ||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | |
| Lamberto Andreotti | | 2014 | $ | 1,700,000 | $ | 18,032,703 | $ | 5,614,080 | $ | 945,611 | $ | 769,988 | $ | 27,062,382 | ||||||||||
Chief Executive Officer | | 2013 | $ | 1,686,539 | $ | 14,586,898 | $ | 3,799,913 | $ | 0 | $ | 774,396 | $ | 20,847,746 | ||||||||||
| | | | 2012 | $ | 1,623,077 | $ | 9,722,004 | $ | 3,844,864 | $ | 1,193,957 | $ | 818,078 | $ | 17,201,980 | |||||||||
| Charles Bancroft | 2014 | $ | 910,520 | $ | 5,287,786 | $ | 1,566,095 | $ | 4,004,475 | $ | 285,408 | $ | 12,054,284 | |||||||||||
EVP and Chief Financial | 2013 | $ | 901,092 | $ | 4,778,079 | $ | 1,128,108 | $ | 759,507 | $ | 311,230 | $ | 7,878,016 | |||||||||||
Officer | 2012 | $ | 868,635 | $ | 2,395,753 | $ | 1,321,979 | $ | 3,662,332 | $ | 318,614 | $ | 8,567,313 | |||||||||||
| Giovanni Caforio, M.D.(7) | | 2014 | $ | 915,962 | $ | 3,999,630 | $ | 2,125,043 | $ | 0 | $ | 204,543 | $ | 7,245,178 | ||||||||||
Chief Operating Officer | | 2013 | $ | 748,320 | $ | 1,587,106 | $ | 788,565 | $ | 0 | $ | 177,861 | $ | 3,301,852 | ||||||||||
| Francis Cuss, MB BChir, FRCP | 2014 | $ | 875,000 | $ | 3,541,409 | $ | 1,685,600 | $ | 782,167 | $ | 194,805 | $ | 7,078,981 | |||||||||||
EVP and Chief Scientific | 2013 | $ | 736,102 | $ | 2,016,197 | $ | 748,372 | $ | 65,331 | $ | 153,035 | $ | 3,719,037 | |||||||||||
Officer | ||||||||||||||||||||||||
| Sandra Leung | | 2014 | $ | 849,750 | $ | 3,981,588 | $ | 1,291,456 | $ | 1,694,853 | $ | 237,158 | $ | 8,054,805 | ||||||||||
EVP, General Counsel | | 2013 | $ | 843,087 | $ | 2,883,914 | $ | 844,238 | $ | 0 | $ | 245,048 | $ | 4,816,287 | ||||||||||
& Corporate Secretary | | 2012 | $ | 768,738 | $ | 2,020,399 | $ | 907,256 | $ | 1,953,216 | $ | 243,163 | $ | 5,892,772 | ||||||||||
these awards is disclosed in the Grants of Plan-Based Awards Table in the Proxy Statements for the specified years. For performance share unit awards, the following represents the aggregate value based on the maximum number of shares that can be earned for the awards granted in the specified years.
| | Performance Share Units | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| | Performance Share Units | |||||||||||||||||||
| | | | | | | | | | | | | | ||||||||||
Name | 2011 | 2012 | 2013 |
| | 2012 | | 2013 | | 2014 | ||||||||||||
| | | | | | | | | | | | | | ||||||||||
| | | | | | | | | | | | | | ||||||||||
Lamberto Andreotti | $ | 6,511,186 | $ | 10,044,739 | $ | 17,513,725 | Lamberto Andreotti | $ | 10,044,739 | $ | 17,513,725 | $ | 22,316,150 | |||||||||
Charles Bancroft | $ | 1,428,360 | $ | 2,450,141 | $ | 4,741,036 | Charles Bancroft | $ | 2,450,141 | $ | 4,741,036 | $ | 6,320,423 | |||||||||
Giovanni Caforio, M.D.(1) | Giovanni Caforio, M.D.(1) | | n.a. | $ | 1,436,011 | $ | 4,581,476 | |||||||||||||||
Francis Cuss, MB BChir, FRCP(1) | n.a. | n.a. | $ | 1,867,635 | Francis Cuss, MB BChir, FRCP(1) | n.a. | $ | 1,867,635 | $ | 3,970,800 | ||||||||||||
Giovanni Caforio, M.D.(1) | n.a. | n.a. | $ | 1,436,011 | ||||||||||||||||||
Sandra Leung | $ | 1,777,063 | $ | 2,194,865 | $ | 3,608,164 | Sandra Leung | $ | 2,194,865 | $ | 3,608,164 | $ | 4,767,532 | |||||||||
Elliott Sigal, M.D., Ph.D. | $ | 2,844,839 | $ | 3,514,622 | $ | 5,660,978 | ||||||||||||||||
Beatrice Cazala | $ | 1,895,405 | $ | 2,533,970 | $ | 4,567,447 | ||||||||||||||||
earnings, (ii) a decrease in discount rates, and (iii) updated mortality assumptions. Additionally, for Mr. Bancroft and Ms. Leung, the increase reflects the fact that they are one year closer to age 60, the earliest age at which participants are eligible for an unreduced benefit. For Mr. Andreotti and Dr. Cuss, the increase was partially off-set by one less year of payments, as both are over age 60. Dr. Caforio in not a participant in any of the company's pension plans. The company does not pay above-market interest rates on deferred compensation. For 2013, the changes in the pension values were negative for the following Named Executive Officers:
| ||||
| ||||
| ||||
| ||||
For 2013, for Mr. Bancroft and Dr. Cuss, the rise in interest rates was offset by increases in their Final Average Compensation, which is defined as the average of the five consecutive years out of the last ten years, ending December 31, 2014, in which the employee's compensation was the highest. Ms. Cazala was a participant in the U.S. pension plan during her U.S. assignment from June 1, 1987 through April 30, 1991. Additionally, Ms. Cazala is a participant in our Supplementary Pension Plan payable in Euros. The change in value relating to the Supplementary Pension Plan also reflects the difference in exchange rates used to convert the 2011, 2012 and 2013 amounts from Euros into US dollars. These exchange rates were 1.3928, 1.2861 and 1.3279 for 2011, 2012 and 2013, respectively. Dr. Caforio is not a participant in any company pension plans.
| ||||
| ||||
| ||||
| ||||
| ||||
| ||||
| ||||
On occasion, a family member accompanied Mr. Bancroft and Dr. Caforio when traveling HeliFlight/NetJets on the company's HeliFlite account for business travel.business. Mr. Bancroft and Dr. Caforio paid the taxes on the imputed income as calculated using the Standard Industry Fare Level (SIFL) rate. We did not reimburse Mr. Bancroft and Dr. Caforio for taxes hethey paid.
Employment Letter Agreement
On February 11, 2011, we entered into an employment letter agreement with Ms. Cazala in connection with the commencement of her long-term assignment in the United States, which took effect on February 21, 2011. At the same time, Ms. Cazala's employment contract with our French subsidiary, Bristol-Myers Squibb SARL, was suspended. Ms. Cazala's employment letter agreement was terminated by the company in January 2014, resulting in the automatic reinstatement of her French employment contract. As previously announced, Ms. Cazala's French contract is being terminated effective June 9, 2014. In connection with Ms. Cazala's departure from the company, we entered into a Settlement Agreement and General Release with her, which is further described on page 61.
During the term of the U.S. employment letter agreement, Ms. Cazala was eligible to participate in the company's U.S. benefit plans. The company had also agreed to continue certain of Ms. Cazala's social insurance and company-sponsored health and welfare benefits in France under the same cost-sharing arrangements that applied before Ms. Cazala's long-term assignment in the United States. During the term of her U.S. assignment, Ms. Cazala's participation in our Supplementary Pension Plan was suspended, although she will receive credit under the plan for her service in the United States. Ms. Cazala was not eligible to participate in the Savings and Investment Program.
We do not have employment agreements with any of our other Named Executive Officers.
Grants of Plan-Based Awards20132014 Fiscal Year
| | | | | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(2) | | Estimated Future Payouts Under Equity Incentive Plan Awards (shares) | | Grant Date Fair Value of Stock | |||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name | | Award Type | | Grant Date(1) | | Approval Date | | Threshold ($) | | Target ($) | | Maximum ($) | | Threshold (#) | | Target (#) | | Maximum (#) | | and Option Awards | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Lamberto Andreotti | | AIP | | | $ | 296,565 | $ | 2,550,000 | $ | 6,402,553 | | | | | |||||||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 7,456 | | 58,480 | | 97,954 | (3)(6) | $ | 3,283,067 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 7,041 | | 55,226 | | 92,504 | (4)(6) | $ | 3,100,388 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 12,997 | | 127,426 | | 256,126 | (5)(7) | $ | 6,939,620 | (10) | ||||||||||||
| | MSU | | 03/10/14 | | 03/03/14 | | | | | 50,970 | | 84,950 | | 169,900 | (8) | $ | 4,709,628 | (10) | ||||||||||||
Charles Bancroft | AIP | $ | 105,893 | $ | 910,520 | $ | 2,286,138 | ||||||||||||||||||||||||
| PSU | 03/10/14 | 03/03/14 | 1,867 | 14,647 | 24,534 | (3)(6) | $ | 822,283 | (9) | ||||||||||||||||||||||
| PSU | 03/10/14 | 03/03/14 | 1,635 | 12,820 | 21,474 | (4)(6) | $ | 719,715 | (9) | ||||||||||||||||||||||
| PSU | 03/10/14 | 03/03/14 | 4,179 | 40,973 | 82,356 | (5)(7) | $ | 2,231,390 | (10) | ||||||||||||||||||||||
| MSU | 03/10/14 | 03/03/14 | 16,390 | 27,316 | 54,632 | (8) | $ | 1,514,399 | (10) | ||||||||||||||||||||||
Giovanni Caforio, M.D. | | AIP | | | $ | 119,740 | $ | 1,029,575 | $ | 2,585,062 | | | | | |||||||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 737 | | 5,778 | | 9,678 | (3)(6) | $ | 324,377 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 1,244 | | 9,756 | | 16,341 | (4)(6) | $ | 547,702 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 3,490 | | 34,211 | | 68,764 | (5)(7) | $ | 1,863,131 | (10) | ||||||||||||
| | MSU | | 03/10/14 | | 03/03/14 | | | | | 13,684 | | 22,807 | | 45,614 | (8) | $ | 1,264,420 | (10) | ||||||||||||
Francis Cuss, | AIP | $ | 101,763 | $ | 875,000 | $ | 2,196,954 | ||||||||||||||||||||||||
MB BChir, FRCP | PSU | 03/10/14 | 03/03/14 | 767 | 6,019 | 10,082 | (3)(6) | $ | 337,907 | (9) | |||||||||||||||||||||
| PSU | 03/10/14 | 03/03/14 | 699 | 5,479 | 9,177 | (4)(6) | $ | 307,591 | (9) | ||||||||||||||||||||||
| PSU | 03/10/14 | 03/03/14 | 3,231 | 31,677 | 63,671 | (5)(7) | $ | 1,725,129 | (10) | ||||||||||||||||||||||
| MSU | 03/10/14 | 03/03/14 | 12,671 | 21,118 | 42,236 | (8) | $ | 1,170,782 | (10) | ||||||||||||||||||||||
Sandra Leung | | AIP | | | $ | 80,855 | $ | 695,228 | $ | 1,745,582 | | | | | |||||||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 1,421 | | 11,147 | | 18,671 | (3)(6) | $ | 625,793 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 1,244 | | 9,756 | | 16,341 | (4)(6) | $ | 547,702 | (9) | ||||||||||||
| | PSU | | 03/10/14 | | 03/03/14 | | | | | 3,133 | | 30,716 | | 61,739 | (5)(7) | $ | 1,672,793 | (10) | ||||||||||||
| | MSU | | 03/10/14 | | 03/03/14 | | | | | 12,287 | | 20,478 | | 40,956 | (8) | $ | 1,135,300 | (10) | ||||||||||||
| | | | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(3) | Estimated Future Payouts Under Equity Incentive Plan Awards (shares) | | | | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | All Other Stock Awards: # of Shares of Stock or Units | Grant Date Fair Value of Stock and Option Awards(4) | | |||||||||||||||||||||||||||
Name | Grant Date(1) | Approval Date | Threshold ($) | Target ($) | Maximum ($) | Threshold (#) | Target (#) | Maximum (#) | | |||||||||||||||||||||||
Lamberto Andreotti | $ | 294,384 | $ | 2,531,250 | $ | 6,355,475 | ||||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 17,631 | 165,865 | 277,824 | (5)(8) | $ | 6,203,351 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 6,216 | 58,480 | 97,954 | (6)(8) | $ | 2,187,152 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 5,871 | 55,226 | 92,504 | (7)(8) | $ | 2,065,452 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 66,272 | 110,453 | 220,906 | (9) | $ | 4,130,942 | ||||||||||||||||||||||||
Charles Bancroft | $ | 104,875 | $ | 901,765 | $ | 2,264,156 | ||||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 5,125 | 48,215 | 80,760 | (5)(8) | $ | 1,803,241 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,557 | 14,647 | 24,534 | (6)(8) | $ | 547,798 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,363 | 12,819 | 21,472 | (7)(8) | $ | 479,431 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 15,384 | 25,640 | 51,280 | (9) | $ | 958,936 | |||||||||||||||||||||||||
| 12/02/13 | 11/13/13 | 19,205 | (10) | $ | 988,673 | |||||||||||||||||||||||||||
Francis Cuss, | $ | 75,623 | $ | 650,238 | $ | 1,632,621 | ||||||||||||||||||||||||||
MB BChir, FRCP | 03/10/13 | 03/07/13 | 1,947 | 18,316 | 30,679 | (5)(8) | $ | 685,018 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 640 | 6,019 | 10,082 | (6)(8) | $ | 225,111 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 582 | 5,478 | 9,176 | (7)(8) | $ | 204,877 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 6,575 | 10,958 | 21,916 | (9) | $ | 409,829 | ||||||||||||||||||||||||
| 07/01/13 | 03/29/13 | (2) | 10,963 | (10) | $ | 491,362 | |||||||||||||||||||||||||
Giovanni Caforio, | $ | 73,309 | $ | 630,348 | $ | 1,582,681 | ||||||||||||||||||||||||||
M.D. | 03/10/13 | 03/07/13 | 785 | 7,389 | 12,377 | (5)(8) | $ | 276,349 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 614 | 5,778 | 9,678 | (6)(8) | $ | 216,097 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,037 | 9,756 | 16,341 | (7)(8) | $ | 364,874 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 11,708 | 19,513 | 39,026 | (9) | $ | 729,786 | |||||||||||||||||||||||||
Sandra Leung | $ | 78,485 | $ | 674,850 | $ | 1,694,417 | ||||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 3,901 | 36,694 | 61,462 | (5)(8) | $ | 1,372,356 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,185 | 11,147 | 18,671 | (6)(8) | $ | 416,898 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,037 | 9,756 | 16,341 | (7)(8) | $ | 364,874 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 11,708 | 19,513 | 39,026 | (9) | $ | 729,786 | ||||||||||||||||||||||||
Elliott Sigal, M.D., | $ | 152,919 | $ | 1,314,868 | $ | 3,301,376 | ||||||||||||||||||||||||||
Ph.D.(11) | 03/10/13 | 03/07/13 | 6,160 | 57,948 | 97,063 | (5)(8) | $ | 2,167,255 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,871 | 17,603 | 29,485 | (6)(8) | $ | 658,352 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,575 | 14,815 | 24,815 | (7)(8) | $ | 554,081 | |||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 17,778 | 29,630 | 59,260 | (9) | $ | 1,108,162 | |||||||||||||||||||||||||
| 06/30/13 | 05/07/13 | 31,376 | $ | 1,402,193 | (12) | |||||||||||||||||||||||||||
Beatrice Cazala | $ | 104,112 | $ | 895,199 | $ | 2,247,670 | ||||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 4,936 | 46,430 | 77,770 | (5)(8) | $ | 1,736,482 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,557 | 14,647 | 24,534 | (6)(8) | $ | 547,798 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 1,258 | 11,833 | 19,820 | (7)(8) | $ | 442,554 | ||||||||||||||||||||||||
| 03/10/13 | 03/07/13 | 14,200 | 23,667 | 47,334 | (9) | $ | 885,146 | ||||||||||||||||||||||||
based on the grant date closing price of $44.69 on June 28, 2013 (the last trading date preceding the grant date of June 30) for Dr. Sigal's award, $44.82 on July 1, 2013 for Dr. Cuss' award, and $51.48 on December 2, 2013 for Mr. Bancroft's award. These values are consistent with the grant date estimate of compensation costs to be recognized over the service period, excluding the effect of forfeitures.
Outstanding Equity Awards at Fiscal Year-End20132014 Fiscal Year
| | | Option Awards | Stock Awards | | | Option Awards | | Stock Awards | ||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ||||||||||||||||||||||||||||
| | | | | | | | | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That Have Not Vested ($)(5) | | | Grant Date/ | | Number of Securities Underlying Unexercised Options (#) | | Option | | Option | | Number of Shares or Units of Stock That Have Not | | Market Value of Shares or Units of Stock That Have Not | | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That | | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That | ||||||||||||||||||||||||||||||||
| | | Number of Securities Underlying Unexercised Options (#) | | | Number of Shares or Units of Stock That Have Not Vested (#)(3) | Market Value of Shares or Units of Stock That Have Not Vested ($)(3)(5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ||||||||||||||||||||||||||||
Name | Grant Date/ Performance Award Period | Exer- cisable(2) | Unexer- cisable(1)(2) | Option Exercise Price | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#)(3) | Market Value of Shares or Units of Stock That Have Not Vested ($)(3)(5) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That Have Not Vested ($)(5) | | Performance Award Period | | Exer- cisable(1) | | Unexer- cisable | | Exercise Price | | Expiration Date | | Vested (#)(2) | | Vested ($)(2)(3) | | Have Not Vested (#) | | Have Not Vested ($)(3) | |||||||||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ||||||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | ||||||||||||||||||||||||||||
Lamberto Andreotti | 3/2/2004 | 108,000 | 0 | $ | 28.11 | 3/1/2014 | Lamberto Andreotti | | 3/1/2005 | | 112,500 | | 0 | (4) | $ | 25.45 | | 2/28/2015 | | | | | | | | | ||||||||||||||||||||||||||||||||
| 3/1/2005 | 112,500 | 0 | (8) | $ | 25.45 | 2/28/2015 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/7/2006 | 115,000 | 0 | (8) | $ | 22.73 | 3/6/2016 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 12/1/2006 | 300,000 | 0 | (8) | $ | 24.74 | 11/30/2016 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2007 | 234,720 | 0 | (8) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/4/2008 | 305,909 | 0 | (8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/3/2009 | 368,706 | 0 | (8) | $ | 17.51 | 3/2/2019 | 10,344 | (4) | $ | 549,784 | |||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2011-12/31/2013 | 360,641 | (9) | $ | 19,168,088 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 135,637 | (10) | $ | 7,209,129 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 57,407 | (11) | $ | 3,051,163 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/2/2010 | 32,698 | (6) | $ | 1,737,899 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5/4/2010 | 27,980 | (6) | $ | 1,487,137 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/1/2011 | 134,124 | (6) | $ | 7,128,691 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2012 | 175,440 | (6) | $ | 9,324,636 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/10/2013 | 66,272 | (7) | $ | 3,522,346 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| | | 3/7/2006 | | 115,000 | | 0 | (4) | $ | 22.73 | | 3/6/2016 | | | | | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 12/1/2006 | | 300,000 | | 0 | (4) | $ | 24.74 | | 11/30/2016 | | | | | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 3/6/2007 | | 234,720 | | 0 | (4) | $ | 27.01 | | 3/5/2017 | | | | | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 3/4/2008 | | 305,909 | | 0 | (4) | $ | 22.14 | | 3/3/2018 | | | | | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 3/3/2009 | | 368,706 | | 0 | (4) | $ | 17.51 | | 3/2/2019 | | | | | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2012-12/31/2014 | | | | | | | | | | 229,867 | (5) | $ | 13,569,068 | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2013-12/31/2015 | | | | | | | | | | 144,384 | (6) | $ | 8,522,990 | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2014-2/28/2017 | | | | | | | | | | | | | | 154,318 | (7) | $ | 9,109,368 | ||||||||||||||||||||||||||||||||||||||
| | | 3/1/2011 | | | | | | | | | | | | | | 67,062 | (8) | $ | 3,958,670 | ||||||||||||||||||||||||||||||||||||||
| | | 3/6/2012 | | | | | | | | | | | | | | 116,960 | (8) | $ | 6,904,149 | ||||||||||||||||||||||||||||||||||||||
| | | 3/10/2013 | | | | | | | | | | | | | | 165,680 | (8) | $ | 9,780,090 | ||||||||||||||||||||||||||||||||||||||
| | | 3/10/2014 | | | | | | | | | | | | | | 50,970 | (9) | $ | 3,008,759 | ||||||||||||||||||||||||||||||||||||||
Charles Bancroft | 3/1/2005 | 16,931 | 0 | $ | 25.45 | 2/28/2015 | Charles Bancroft | 3/6/2007 | 22,598 | 0 | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||||||||
| 3/7/2006 | 19,320 | 0 | $ | 22.73 | 3/6/2016 | 3/4/2008 | 37,460 | 0 | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2007 | 22,598 | 0 | $ | 27.01 | 3/5/2017 | 3/3/2009 | 52,884 | 0 | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||||||||||
| 3/4/2008 | 37,460 | 0 | $ | 22.14 | 3/3/2018 | 11/1/2011 | 6,200 | (10) | $ | 365,986 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 3/3/2009 | 52,884 | 0 | $ | 17.51 | 3/2/2019 | 12/2/2013 | 19,205 | (10) | $ | 1,133,671 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 11/1/2011 | 9,302 | (4) | $ | 494,401 | 1/1/2012-12/31/2014 | 57,572 | (5) | $ | 3,398,481 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 12/2/2013 | 19,205 | (4) | $ | 1,020,746 | 1/1/2013-12/31/2015 | 33,516 | (6) | $ | 1,978,464 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2011-12/31/2013 | 104,835 | (9) | $ | 5,571,964 | 1/1/2014-2/28/2017 | 49,620 | (7) | $ | 2,929,069 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 33,971 | (10) | $ | 1,805,547 | 3/1/2011 | 19,494 | (8) | $ | 1,150,731 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 13,325 | (11) | $ | 708,249 | 3/6/2012 | 29,294 | (8) | $ | 1,729,225 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 3/2/2010 | 11,316 | (6) | $ | 601,445 | 3/10/2013 | 38,460 | (8) | $ | 2,270,294 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 3/1/2011 | 38,988 | (6) | $ | 2,072,212 | 3/10/2014 | 16,390 | (9) | $ | 967,478 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2012 | 43,940 | (6) | $ | 2,335,411 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/10/2013 | 15,384 | (7) | $ | 817,660 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
Giovanni Caforio, M.D. | Giovanni Caforio, M.D. | | 3/7/2006 | | 20,125 | | 0 | $ | 22.89 | | 3/6/2016 | | | | | | | | | |||||||||||||||||||||||||||||||||||||||
| | | 3/6/2007 | | 21,615 | | 0 | $ | 27.01 | | 3/5/2017 | | | | | | | | | |||||||||||||||||||||||||||||||||||||||
| | | 3/4/2008 | | 28,840 | | 0 | $ | 23.12 | | 3/3/2018 | | | | | | | | | |||||||||||||||||||||||||||||||||||||||
| | | 1/3/2011 | | | | | | | | | | 7,500 | (10) | $ | 442,725 | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2012-12/31/2014 | | | | | | | | | | 22,712 | (5) | $ | 1,340,689 | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2013-12/31/2015 | | | | | | | | | | 25,506 | (6) | $ | 1,505,647 | | | | | ||||||||||||||||||||||||||||||||||||||
| | | 1/1/2014-2/28/2017 | | | | | | | | | | | | | | 41,431 | (7) | $ | 2,445,684 | ||||||||||||||||||||||||||||||||||||||
| | | 3/1/2011 | | | | | | | | | | | | | | 2,988 | (8) | $ | 176,382 | ||||||||||||||||||||||||||||||||||||||
| | | 3/6/2012 | | | | | | | | | | | | | | 11,556 | (8) | $ | 682,151 | ||||||||||||||||||||||||||||||||||||||
| | | 3/10/2013 | | | | | | | | | | | | | | 29,270 | (8) | $ | 1,727,808 | ||||||||||||||||||||||||||||||||||||||
| | | 3/10/2014 | | | | | | | | | | | | | | 13,684 | (9) | $ | 807,778 | ||||||||||||||||||||||||||||||||||||||
Francis Cuss, MB | 3/2/2004 | 38,667 | 0 | $ | 28.11 | 3/1/2014 | Francis Cuss, MB | 12/1/2006 | 75,000 | 0 | $ | 24.74 | 11/30/2016 | |||||||||||||||||||||||||||||||||||||||||||||
BChir, FRCP | 3/1/2005 | 38,667 | 0 | $ | 25.45 | 2/28/2015 | BChir, FRCP | 3/6/2007 | 39,010 | 0 | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||||||||
| 12/1/2006 | 100,000 | 0 | $ | 24.74 | 11/30/2016 | 7/1/2013 | 10,963 | (10) | $ | 647,146 | |||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2007 | 39,010 | 0 | $ | 27.01 | 3/5/2017 | 1/1/2012-12/31/2014 | 23,658 | (5) | $ | 1,396,523 | |||||||||||||||||||||||||||||||||||||||||||||||
| 11/2/2009 | 6,666 | (4) | $ | 354,298 | 1/1/2013-12/31/2015 | 14,323 | (6) | $ | 845,507 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 7/1/2013 | 10,963 | (4) | $ | 582,683 | 1/1/2014-2/28/2017 | 38,362 | (7) | $ | 2,264,532 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2011-12/31/2013 | 39,825 | (9) | $ | 2,116,706 | 3/1/2011 | 7,406 | (8) | $ | 437,176 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 13,959 | (10) | $ | 741,917 | 3/6/2012 | 12,038 | (8) | $ | 710,603 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 5,694 | (11) | $ | 302,657 | 3/10/2013 | 16,438 | (8) | $ | 970,335 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 3/2/2010 | 8,262 | (6) | $ | 439,125 | 3/10/2014 | 12,671 | (9) | $ | 747,957 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 3/1/2011 | 14,812 | (6) | $ | 787,258 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2012 | 18,058 | (6) | $ | 959,783 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/10/2013 | 6,575 | (7) | $ | 349,451 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
Giovanni Caforio, | 3/7/2006 | 20,125 | 0 | $ | 22.89 | 3/6/2016 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
M.D. | 3/6/2007 | 21,615 | 0 | $ | 27.01 | 3/5/2017 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/4/2008 | 28,840 | 0 | $ | 23.12 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/3/2009 | 39,363 | 0 | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/3/2011 | 15,000 | (4) | $ | 797,250 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2011-12/31/2013 | 16,066 | (9) | $ | 853,908 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 13,402 | (10) | $ | 712,291 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 10,141 | (11) | $ | 538,994 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/2/2010 | 2,394 | (6) | $ | 127,241 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/1/2011 | 5,976 | (6) | $ | 317,624 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/6/2012 | 17,334 | (6) | $ | 921,302 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3/10/2013 | 11,708 | (7) | $ | 622,270 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| | | Option Awards | Stock Awards | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That Have Not Vested ($)(5) | |||||||||||||||||||
| | | Number of Securities Underlying Unexercised Options (#) | | | Number of Shares or Units of Stock That Have Not Vested (#)(3) | Market Value of Shares or Units of Stock That Have Not Vested ($)(3)(5) | ||||||||||||||||||||||
Name | Grant Date/ Performance Award Period | Exer- cisable(2) | Unexer- cisable(1)(2) | Option Exercise Price | Option Expiration Date | |||||||||||||||||||||||
Sandra Leung | 3/2/2004 | 15,000 | 0 | $ | 28.11 | 3/1/2014 | ||||||||||||||||||||||
| 3/1/2005 | 15,000 | 0 | $ | 25.45 | 2/28/2015 | ||||||||||||||||||||||
| 3/7/2006 | 14,560 | 0 | $ | 22.73 | 3/6/2016 | ||||||||||||||||||||||
| 12/1/2006 | 100,000 | 0 | (8) | $ | 24.74 | 11/30/2016 | |||||||||||||||||||||
| 3/6/2007 | 116,100 | 0 | (8) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||
| 3/4/2008 | 156,582 | 0 | (8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||
| 3/3/2009 | 169,893 | 0 | (8) | $ | 17.51 | 3/2/2019 | 4,793 | (4) | $ | 254,748 | |||||||||||||||||
| 1/1/2011-12/31/2013 | 79,782 | (9) | $ | 4,240,432 | |||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 25,854 | (10) | $ | 1,374,114 | |||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 10,141 | (11) | $ | 538,994 | |||||||||||||||||||||||
| 3/2/2010 | 14,710 | (6) | $ | 781,837 | |||||||||||||||||||||||
| 3/1/2011 | 29,672 | (6) | $ | 1,577,067 | |||||||||||||||||||||||
| 3/6/2012 | 33,440 | (6) | $ | 1,777,336 | |||||||||||||||||||||||
| 3/10/2013 | 11,708 | (7) | $ | 622,270 | |||||||||||||||||||||||
Elliott Sigal, M.D., | 3/6/2007 | 25,000 | 0 | (8) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||
Ph.D. | 3/4/2008 | 76,478 | 0 | (8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||
| 1/1/2011-12/31/2013 | 95,631 | (9) | $ | 5,082,805 | ||||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 31,604 | (10) | $ | 1,679,758 | ||||||||||||||||||||||||
Beatrice Cazala | 3/6/2009 | 169,893 | 0 | (8) | $ | 18.35 | 9/2/2018 | |||||||||||||||||||||
| 3/1/2011 | 19,577 | (4) | $ | 1,040,518 | |||||||||||||||||||||||
| 1/1/2011-12/31/2013 | 100,952 | (9) | $ | 5,365,606 | |||||||||||||||||||||||
| 1/1/2012-12/31/2014 | 33,971 | (10) | $ | 1,805,547 | |||||||||||||||||||||||
| 1/1/2013-12/31/2015 | 12,300 | (11) | $ | 653,735 | |||||||||||||||||||||||
| 3/2/2010 | 13,578 | (6) | $ | 721,671 | |||||||||||||||||||||||
| 3/1/2011 | 37,544 | (6) | $ | 1,995,464 | |||||||||||||||||||||||
| 3/6/2012 | 43,940 | (6) | $ | 2,335,411 | |||||||||||||||||||||||
| 3/10/2013 | 14,200 | (7) | $ | 754,741 | |||||||||||||||||||||||
| | | Option Awards | | Stock Awards | ||||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Grant Date/ | | Number of Securities Underlying Unexercised Options (#) | | Option | | Option | | Number of Shares or Units of Stock That Have Not | | Market Value of Shares or Units of Stock That Have Not | | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That | | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That | ||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name | | | Performance Award Period | | Exer- cisable(1) | | Unexer- cisable | | Exercise Price | | Expiration Date | | Vested (#)(2) | | Vested ($)(2)(3) | | Have Not Vested (#) | | Have Not Vested ($)(3) | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Sandra Leung | | 3/1/2005 | | 15,000 | | 0 | $ | 25.45 | | 2/28/2015 | | | | | | | | | ||||||||||||
| | 3/7/2006 | | 14,560 | | 0 | $ | 22.73 | | 3/6/2016 | | | | | | | | | ||||||||||||
| | 12/1/2006 | | 100,000 | | 0 | (4) | $ | 24.74 | | 11/30/2016 | | | | | | | | | |||||||||||
| | 3/6/2007 | | 116,100 | | 0 | (4) | $ | 27.01 | | 3/5/2017 | | | | | | | | | |||||||||||
| | 3/4/2008 | | 156,582 | | 0 | (4) | $ | 22.14 | | 3/3/2018 | | | | | | | | | |||||||||||
| | 3/3/2009 | | 169,893 | | 0 | (4) | $ | 17.51 | | 3/2/2019 | | | | | | | | | |||||||||||
| | 1/1/2012-12/31/2014 | | | | | | | | | | 43,815 | (5) | $ | 2,586,371 | | | | | |||||||||||
| | 1/1/2013-12/31/2015 | | | | | | | | | | 25,506 | (6) | $ | 1,505,647 | | | | | |||||||||||
| | 1/1/2014-2/28/2017 | | | | | | | | | | | | | | 37,198 | (7) | $ | 2,195,822 | |||||||||||
| | 3/1/2011 | | | | | | | | | | | | | | 14,836 | (8) | $ | 875,769 | |||||||||||
| | 3/6/2012 | | | | | | | | | | | | | | 22,294 | (8) | $ | 1,316,015 | |||||||||||
| | 3/10/2013 | | | | | | | | | | | | | | 29,270 | (8) | $ | 1,727,808 | |||||||||||
| | 3/10/2014 | | | | | | | | | | | | | | 12,287 | (9) | $ | 725,290 | |||||||||||
Grant Date | Vesting | ||
|---|---|---|---|
| 1/3/2011 | Four equal installments of 25% on each of the first four | ||
| 11/1/2011 | Three equal installments on each of the third, fourth, and fifth anniversaries of the grant date | ||
| 7/1/2013 | Three equal installments on each of the third, fourth, and fifth anniversaries of the grant date | ||
| 12/2/2013 | Three equal installments on each of the third, fourth, and fifth anniversaries of the grant date | ||
Option Exercises and Stock Vesting20132014 Fiscal Year
| | Option Awards | Stock Awards | | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| | Option Awards | | Stock Awards | |||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | ||||||||||||||
Name | Number of Shares Acquired on Exercise (#) | Value Realized On Exercise (1)($) | Number of Shares Acquired on Vesting (#) | Value Realized On Vesting (1)($) | | | | Number of Shares Acquired on Exercise (#) | | Value Realized On Exercise(1) ($) | | Number of Shares Acquired on Exercise (#) | | Value Realized On Exercise(2) ($) | ||||||||||||||||
| | | | | | | | | | | | | | | | | | ||||||||||||||
| | | | | | | | | | | | | | | | | | ||||||||||||||
Lamberto Andreotti | 0 | $ | 0 | 17,797 | $ | 659,593 | (2) | Lamberto Andreotti | | 108,000 | $ | 2,651,227 | | 10,344 | $ | 553,301 | (3) | |||||||||||||
| 128,838 | $ | 4,896,294 | (3) | ||||||||||||||||||||||||||
| 251,044 | $ | 9,421,691 | (4) | ||||||||||||||||||||||||||
| | | | | | | 216,981 | $ | 11,711,082 | (4) | ||||||||||||||||||||
| | | | | | | 363,310 | $ | 19,433,446 | (5) | ||||||||||||||||||||
Charles Bancroft | 0 | $ | 0 | 2,487 | $ | 92,392 | (2) | Charles Bancroft | 36,251 | $ | 1,100,030 | 3,102 | $ | 180,505 | (3) | |||||||||||||||
| 30,823 | $ | 1,156,787 | (3) | 52,424 | $ | 2,854,306 | (4) | |||||||||||||||||||||||
| 46,817 | $ | 1,757,044 | (4) | 105,611 | $ | 5,649,115 | (5) | |||||||||||||||||||||||
Giovanni Caforio, M.D. | Giovanni Caforio, M.D. | | 39,363 | $ | 1,450,660 | | 7,500 | $ | 396,375 | (3) | ||||||||||||||||||||
| | | | | | | 17,325 | $ | 955,550 | (4) | ||||||||||||||||||||
| | | | | | | 16,185 | $ | 865,738 | (5) | ||||||||||||||||||||
Francis Cuss, MB BChir, FRCP | 84,952 | $ | 1,246,668 | 10,419 | $ | 489,256 | (2) | Francis Cuss, MB BChir, FRCP | 102,334 | $ | 2,654,393 | 6,666 | $ | 387,895 | (3) | |||||||||||||||
| 14,928 | $ | 560,248 | (3) | 24,704 | $ | 1,342,431 | (4) | ||||||||||||||||||||||
| 34,177 | $ | 1,282,666 | (4) | 40,120 | $ | 2,146,026 | (5) | ||||||||||||||||||||||
Giovanni Caforio, M.D. | 43,833 | $ | 613,896 | 9,351 | $ | 318,065 | (2) | |||||||||||||||||||||||
| 7,228 | $ | 271,267 | (3) | |||||||||||||||||||||||||||
| 9,904 | $ | 371,685 | (4) | |||||||||||||||||||||||||||
Sandra Leung | 19,310 | $ | 265,128 | 8,773 | $ | 325,081 | (2) | Sandra Leung | | 15,000 | $ | 392,522 | | 4,793 | $ | 256,378 | (3) | |||||||||||||
| 28,014 | $ | 1,051,365 | (3) | ||||||||||||||||||||||||||
| 60,859 | $ | 2,284,021 | (4) | ||||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | 914,877 | $ | 15,552,623 | 100,185 | $ | 4,069,915 | (2)(5) | |||||||||||||||||||||||
| 63,338 | $ | 2,509,041 | (3)(6) | |||||||||||||||||||||||||||
| 99,812 | $ | 3,745,957 | (4) | |||||||||||||||||||||||||||
Beatrice Cazala | 216,412 | $ | 3,974,876 | 3,595 | $ | 135,747 | (2) | |||||||||||||||||||||||
| 31,994 | $ | 1,200,735 | (3) | ||||||||||||||||||||||||||
| 56,181 | $ | 2,108,474 | (4) | ||||||||||||||||||||||||||
| | | | | | | 45,995 | $ | 2,498,435 | (4) | ||||||||||||||||||||
| | | | | | | 80,373 | $ | 4,299,142 | (5) | ||||||||||||||||||||
Retirement Plan
As of December 31, 2009, we discontinued service accruals under the Retirement Income Plan and Benefit Equalization Plan—Plan (BEP)—Retirement Plan in the U.S. and Puerto Rico for active plan participants and we stopped addingclosed the plans to new participants to those plans.entrants. For active plan participants at year-end 2009, we are allowingprovided five additional years of pay growth in the pension plans. Accordingly, 2014 was the last year of pay growth under our pension plans.
The Retirement Income Plan is a tax-qualified defined benefit pension plan under Section 401(a) of the Internal Revenue Code that provides income for employees after retirement. The Retirement Income Plan is a tax-qualified plan, as defined under Section 401(a) of the Internal Revenue Code. The benefit is calculated based on the employee's final average compensation and years of service. All U.S. employees hired before January 1, 2010 who arewere not participants in a pension plan through a collective bargaining agreement arewere eligible for the Retirement Income Plan if they workworked at least 1,000 hours per year. Employees whose pay or benefits exceedexceeded the IRS qualified plan limits arewere eligible for the Benefit Equalization Plan—BEP—Retirement Plan.
The key plan provisions of the Retirement Income Plan are as follows:
The Benefit Equalization Plan—BEP—Retirement Plan is a non-qualified deferred compensation plan that provides income for employees after retirement in excess of the benefits payable under the qualified Retirement Income Plan. The benefit is calculated using the same formula as the Retirement Income Plan, but without the limits on compensation and benefits imposed under Section 401(a)(17) and Section 415(b) of the Internal Revenue Code. Employees whose pay or benefits exceedexceeded the IRS qualified plan limits arewere eligible for the Benefit Equalization Plan—BEP—Retirement Plan.
The provisions are the same as those above for the Retirement Income Plan, except for the following:
Supplementary Pension Plan
We maintain a defined benefit pension plan which was closed to new participants in January 2010. Participation in the plan was limited to certain eligible employees in Europe. Ms. Cazala is the only Named Executive Officer who was eligible to participate in the plan. The key plan provisions of the Supplementary Pension Plan are as follows:
Present Value of Accumulated Pension Benefits20132014 Fiscal Year
Name | Plan Name | # of Years of Credited Service(5) | Present Value of Accumulated Benefits(1) | Payments During Last Fiscal Year | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lamberto Andreotti(2)(3) | Retirement Income Plan | 4.3 | $ | 248,924 | $ | 0 | ||||||
| Benefit Equalization Plan | 4.3 | $ | 5,821,254 | $ | 0 | |||||||
| Charles Bancroft | Retirement Income Plan | 25.6 | $ | 1,213,060 | $ | 0 | ||||||
| Benefit Equalization Plan | 25.6 | $ | 9,540,694 | $ | 0 | |||||||
| Francis Cuss, MB BChir, FRCP(3) | Retirement Income Plan | 6.5 | $ | 404,988 | $ | 0 | ||||||
| Benefit Equalization Plan | 6.5 | $ | 1,942,580 | $ | 0 | |||||||
| Giovanni Caforio, M.D.(2) | Retirement Income Plan | 0.0 | $ | 0 | $ | 0 | ||||||
| Benefit Equalization Plan | 0.0 | $ | 0 | $ | 0 | |||||||
| Sandra Leung | Retirement Income Plan | 17.8 | $ | 859,669 | $ | 0 | ||||||
| Benefit Equalization Plan | 17.8 | $ | 6,000,701 | $ | 0 | |||||||
| Elliott Sigal, M.D., Ph.D.(3)(6) | Retirement Income Plan | 12.4 | $ | 0 | $ | (823,663) | ||||||
| Benefit Equalization Plan | 12.4 | $ | 10,822,866 | $ | 0 | |||||||
| Beatrice Cazala(2)(3)(4) | Retirement Income Plan | 4.4 | $ | 216,216 | $ | 0 | ||||||
| Benefit Equalization Plan | 4.4 | $ | 1,818,118 | $ | 0 | |||||||
| Supplementary Pension Plan | 31.9 | $ | 8,409,591 | $ | 0 | |||||||
Name | | Plan Name | | # of Years of Credited Service(1) | | Present Value of Accumulated Benefits(2) | Payments During Last Fiscal Year | |||||
| | | | | | | | | | | | | |
Lamberto Andreotti(3)(4) | Retirement Income Plan | | 4.3 | $ | 275,207 | $0 | ||||||
| | Benefit Equalization Plan | | 4.3 | $ | 6,740,582 | $0 | |||||
Charles Bancroft(4) | Retirement Income Plan | 25.6 | $ | 1,503,950 | $0 | |||||||
| Benefit Equalization Plan | 25.6 | $ | 13,254,279 | $0 | ||||||||
Giovanni Caforio, M.D.(3) | Retirement Income Plan | | 0.0 | $ | 0 | $0 | ||||||
| | Benefit Equalization Plan | | 0.0 | $ | 0 | $0 | |||||
Francis Cuss, MB BChir, FRCP(4) | Retirement Income Plan | 6.5 | $ | 465,759 | $0 | |||||||
| Benefit Equalization Plan | 6.5 | $ | 2,663,976 | $0 | ||||||||
Sandra Leung | Retirement Income Plan | | 17.8 | $ | 1,076,466 | $0 | ||||||
| | Benefit Equalization Plan | | 17.8 | $ | 7,478,757 | $0 | |||||
These assumptions are the same as those disclosed in conformity with generally accepted accounting principles. For active executives, payments are assumed to begin at age 60, the earliest age that employees are eligible for an unreduced pension, or current age if over age 60. The actual benefit received will vary based on a number of factors including final pay, years of serviceage and interest rates at the time of retirement.
Non-Qualified Deferred Compensation Plan
The Benefit Equalization Plan—Plan (BEP)—Savings Plan is a non-qualified deferred compensation plan that allows employees to defer a portion of their total eligible cash compensation and to receive company matching contributions in excess of contributions allowed under the Savings and Investment Program. The Savings and Investment Program is a tax-qualified plan, as defined under Section 401(a) and Section 401(k) of the Internal Revenue Code. Employees who are eligible to participate in the Bristol-Myers Squibb Savings and Investment Program, and whose pay or benefits exceed the IRS qualified plan limits, are eligible for the Benefit Equalization Plan—BEP—Savings Plan. The key provisions of the BEP-Savings Plan are as follows:
Non-Qualified Deferred Compensation20132014 Fiscal Year
Name | Executive Contributions in 2013(1) | Registrant Contributions in 2013(2) | Aggregate Earnings in 2013(3) | Aggregate Withdrawals/ Distributions in 2013 | Aggregate Balance at December 31, 2013(2)(4) |
| | Executive Contributions in 2014(1) | | Registrant Contributions in 2014(2) | | Aggregate Earnings in 2014(3) | | Aggregate Withdrawals/ Distributions in 2014 | | Aggregate Balance at December 31, 2014(2)(4) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | ||||||||||||||||
Lamberto Andreotti(5) | $ | 316,584 | $ | 738,696 | $ | 1,265,722 | $ | 0 | $ | 5,814,391 | Lamberto Andreotti(5) | $ | 314,395 | $ | 733,588 | $ | 600,086 | $ | 0 | $ | 7,462,460 | |||||||||||||
Charles Bancroft(5) | $ | 118,084 | $ | 280,630 | $ | 308,128 | $ | 0 | $ | 1,777,255 | Charles Bancroft(5) | $ | 106,718 | $ | 254,208 | $ | 242,627 | $ | 0 | $ | 2,380,808 | |||||||||||||
Giovanni Caforio, M.D.(5) | Giovanni Caforio, M.D.(5) | $ | 86,672 | $ | 173,343 | $ | 75,686 | $ | 0 | $ | 924,502 | |||||||||||||||||||||||
Francis Cuss, MB BChir, FRCP(5) | $ | 61,218 | $ | 122,435 | $ | 167,840 | $ | 0 | $ | 1,101,995 | Francis Cuss, MB BChir, FRCP(5) | $ | 81,802 | $ | 163,605 | $ | (26,562 | ) | $ | 0 | $ | 1,320,840 | ||||||||||||
Francis Cuss, MB BChir, FRCP(6) | $ | 0 | $ | 0 | $ | 1,609 | $ | 0 | $ | 2,195,283 | Francis Cuss, MB BChir, FRCP(6) | $ | 0 | $ | 0 | $ | 1,477 | $ | 0 | $ | 2,196,760 | |||||||||||||
Giovanni Caforio, M.D.(5) | $ | 85,903 | $ | 147,261 | $ | 73,597 | $ | 0 | $ | 588,801 | ||||||||||||||||||||||||
Sandra Leung(5) | $ | 237,874 | $ | 235,177 | $ | 388,109 | $ | 0 | $ | 2,539,345 | Sandra Leung(5) | $ | 228,937 | $ | 227,094 | $ | 105,414 | $ | 0 | $ | 3,100,789 | |||||||||||||
Elliott Sigal M.D., Ph.D.(5)(9) | $ | 130,755 | $ | 307,646 | $ | 84,995 | $ | 0 | $ | 3,498,571 | ||||||||||||||||||||||||
Elliott Sigal M.D., Ph.D.(6) | $ | 0 | $ | 0 | $ | 345,691 | $ | (244,820 | ) | $ | 1,783,774 | |||||||||||||||||||||||
Elliott Sigal M.D., Ph.D.(7) | $ | 2,895,823 | $ | 0 | $ | 561,093 | $ | 0 | $ | 3,456,915 | ||||||||||||||||||||||||
Beatrice Cazala(5)(8) | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | ||||||||||||||||||||||||
The following illustrates the potential payments and benefits under the company's plans and programs to the Named Executive Officers upon a termination of employment assuming an effective date of December 31, 2013. To the extent payments and benefits are generally available to salaried employees on a non-discriminatory basis, they are excluded from the table.
Termination of Employment Obligations (Excluding Vested Benefits)2013 Fiscal Year
Name | Cash Severance (1) | Restricted Stock Units (2) | Market Share Units (3)(5) | Performance Share Units (4)(5) | Retirement (6) | Health (7) | Retiree Medical (8) | Total | Gross-Up on Excise Taxes (9) | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Voluntary Termination for Good Reason | ||||||||||||||||||||||||||||
Lamberto Andreotti(10) | $ | 3,400,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 20,227 | $ | 0 | $ | 3,420,227 | $ | 0 | ||||||||||
Charles Bancroft | $ | 1,821,040 | $ | 0 | $ | 0 | $ | 0 | $ | 4,248,877 | $ | 15,686 | $ | 129,677 | $ | 6,215,280 | $ | 0 | ||||||||||
Francis Cuss, MB BChir, FRCP(10) | $ | 1,750,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 32,188 | $ | 0 | $ | 1,782,188 | $ | 0 | ||||||||||
Giovanni Caforio, M.D.(11) | $ | 1,750,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 22,094 | $ | 88,687 | $ | 1,860,781 | $ | 0 | ||||||||||
Sandra Leung | $ | 1,699,500 | $ | 0 | $ | 0 | $ | 0 | $ | 2,705,594 | $ | 22,470 | $ | 109,069 | $ | 4,536,633 | $ | 0 | ||||||||||
Involuntary Termination Not for Cause | ||||||||||||||||||||||||||||
Lamberto Andreotti(10) | $ | 3,400,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 20,227 | $ | 0 | $ | 3,420,227 | $ | 0 | ||||||||||
Charles Bancroft | $ | 1,821,040 | $ | 118,897 | $ | 1,878,427 | $ | 8,085,759 | $ | 4,248,877 | $ | 15,686 | $ | 129,677 | $ | 16,298,363 | $ | 0 | ||||||||||
Francis Cuss, MB BChir, FRCP(10) | $ | 1,750,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 32,188 | $ | 0 | $ | 1,782,188 | $ | 0 | ||||||||||
Giovanni Caforio, M.D.(11) | $ | 1,750,000 | $ | 395,436 | $ | 442,474 | $ | 2,105,193 | $ | 0 | $ | 22,094 | $ | 88,687 | $ | 4,803,884 | $ | 0 | ||||||||||
Sandra Leung | $ | 1,699,500 | $ | 210,208 | $ | 1,698,036 | $ | 6,153,540 | $ | 2,705,594 | $ | 22,470 | $ | 109,069 | $ | 12,598,418 | $ | 0 | ||||||||||
Qualifying Termination Within 3 Years Following a Change in Control | ||||||||||||||||||||||||||||
Lamberto Andreotti(12) | $ | 12,707,500 | $ | 96,042 | $ | 18,915,288 | $ | 0 | $ | 2,261,598 | $ | 30,868 | $ | 0 | $ | 34,011,296 | $ | 16,868,569 | ||||||||||
Charles Bancroft | $ | 5,444,910 | $ | 1,515,147 | $ | 6,530,062 | $ | 8,085,759 | $ | 9,614,370 | $ | 48,175 | $ | 106,612 | $ | 31,345,035 | $ | 13,129,810 | ||||||||||
Francis Cuss, MB BChir, FRCP(12) | $ | 5,232,500 | $ | 879,845 | $ | 1,938,912 | $ | 0 | $ | 2,180,670 | $ | 98,860 | $ | 0 | $ | 10,330,787 | $ | 4,876,116 | ||||||||||
Giovanni Caforio, M.D.(11) | $ | 5,232,500 | $ | 797,250 | $ | 2,680,514 | $ | 2,105,193 | $ | 0 | $ | 68,016 | $ | 90,326 | $ | 10,973,799 | $ | 4,451,440 | ||||||||||
Sandra Leung | $ | 4,573,355 | $ | 254,748 | $ | 5,293,793 | $ | 6,153,540 | $ | 5,454,669 | $ | 69,139 | $ | 107,341 | $ | 21,906,585 | $ | 8,270,710 | ||||||||||
The following information presents the post-termination benefits for Dr. Sigal and Ms. Cazala in connection with their terminations. To the extent payments and benefits are generally available to salaried employees on a non-discriminatory basis or are vested benefits, we have not quantified the value of these payments or benefits.
Elliott Sigal, M.D., Ph.D.
Dr. Sigal retired from the company on June 30, 2013. In accordance with our various benefit plans and compensation programs, Dr. Sigal was entitled to and received the following benefits on account of his retirement:
Additionally, on May 7, 2013 the Board approved the acceleration of the final tranches of Dr. Sigal's unvested restricted stock unit awards scheduled to vest in March 2014 as of his retirement date. As a result of the acceleration, Dr. Sigal received 31,376 shares of our common stock with an incremental fair value of $1.4 million.
Beatrice Cazala Post-Termination Benefits
As previously announced, Ms. Cazala ceased to serve as an executive officer of the company on November 13, 2013 and was going to transition to a new role. As part of that transition, in January 2014, we terminated Ms. Cazala's employment letter agreement, dated as of February 11, 2011, covering her long-term U.S. assignment, resulting in the automatic reinstatement of her suspended French employment contract with our French subsidiary Bristol-Myers Squibb SARL. Ms. Cazala was offered a position in France which she has declined and, as a result, her French contract is being terminated effective as of June 9, 2014, her separation date.
In connection with Ms. Cazala's departure, the company and our French subsidiary have entered into a Settlement Agreement and General Release with Ms. Cazala, effective as of February 14, 2014. Pursuant to the agreement, Ms. Cazala remains an active employee in the United States in the position of Executive Vice President through her separation date at her current salary and she will continue to be eligible to participate in all applicable benefit plans. She received her 2013 annual incentive award and remains eligible for the continued vesting of all previously awarded but unvested stock unit awards in accordance with the applicable plan documents. Ms. Cazala is not eligible for 2014 annual or long-term incentive awards.
Ms. Cazala will receive the following benefits to which she is entitled consistent with her French employment contract and all applicable benefit plans as an employee who is retirement eligible and involuntarily terminated without cause and who executes a general release:
Under the Agreement, Ms. Cazala also received in February 2014 an aggregate of $1.15 million in settlement of any claims Ms. Cazala may have had against the company or our French Subsidiary. Ms. Cazala will be entitled to the following payments in connection with her signing another general release prior to her termination:
Additionally, under the agreement, Ms. Cazala has agreed to release any claims she may have against the company and our French subsidiary and any of their affiliates, subsidiaries, officers, directors, employees and agents, among others, except for those that may relate to the administration of any applicable benefit plan or those that cannot be lawfully waived. The agreement also contains covenants imposing on Ms. Cazala certain obligations with respect to confidentiality, proprietary information and non-disparagement, and restricting her ability to engage in certain activities in competition with the company for a period of six months after her separation date. Ms. Cazala has agreed to cooperate and to generally make herself available in connection with any investigation or legal proceedings involving, or any matter that relates to her employment with, the company or our French subsidiary.
Following is a description of payments and benefits available under different termination scenarios:
Voluntary Termination
The company does not offer any payments or benefits to salaried employees, including the Named Executive Officers, upon a voluntary termination other than those that are vested at the time of termination unless the applicable plan or award agreement provides otherwise.
Voluntary Termination for Good Reason
Under the Bristol-Myers Squibb Senior Executive Severance Plan, certain senior executives (including the Named Executive Officers) are eligible to receive severance payments and benefits if they voluntarily terminate their employment for "good reason," where "good reason" is defined as:
A terminated executive who signs a general release will be eligible for the following:
Retirement and Death
The following benefits are generally available to all salaried employees including the Named Executive Officers:
Annual Incentive—Employees are eligible for a pro-rata award based on the number of months worked in the performance period.
Stock Options—Employees are eligible for accelerated vesting of stock options held at least one year from the grant date and have the full term to exercise vested stock options. Upon retirement, exercise thresholds, as described in the Outstanding Equity Awards Table, remain in effect, where applicable. Upon death, exercise thresholds lapse. All outstanding options held by our employees are vested as of December 31, 2013.
Restricted Stock Units—Employees are eligible to vest in a pro-rata portion of restricted stock unit awards held at least one year from the grant date; provided that if an employee turns 65 on or prior
to their retirement or death, then any unvested Restricted Stock Units held for at least one year will have vestedvest in full prior to their retirement or death.
Market Share Units—Employees are eligible to vest in a pro-rata portion of market share unit awards held at least one year from the grant date, subject to performance provisions; provided that if an employee turns 65 on or prior to their retirement or death, then any unvested Market Share Units held for at least one year will vest in full upon their retirement or death;death, subject to performance provisions.
Performance Share Units—If
Defined Benefit Pension Plans—Employees are eligible for benefits accrued under the Retirement Income Plan and the Benefit Equalization Plan—BEP—Retirement Plan.
Savings Plans—Employees are eligible for benefits accumulated under the Savings and Investment Program and the Benefit Equalization Plan—BEP—Savings Plan, as well as a pro-rata annual and transition (if applicable) contribution on eligible compensation paid in the year of separation from service or death.
Post-Retirement Medical and Life Insurance—Employees age 55 or older with ten years of service are eligible for post-retirement medical and life insurance benefits.
Involuntary Termination Not for Cause
The following benefits are generally available to all salaried employees including the Named Executive Officers:
Annual Incentive—Employees are eligible for a pro-rata award based on the number of months worked in the performance period if the termination occurs on or after September 30th of the plan year. If an employee is eligible to retire, or the employee's age plus years of service equal or exceed 70, and the employee has at least 10 years of service, the employee is eligible for a pro-rata award based on the number of months worked in the performance period.
Stock Options—Upon signing a general release, an employee is eligible for accelerated vesting of stock options held at least one year from the grant date and has three months to exercise. If an employee is eligible to retire, or the employee's age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee will have the full term to exercise. Exercise thresholds, as described in the Outstanding Equity Awards Table, remain in effect, where applicable. All outstanding options held by our employees are vested as of December 31, 2013.
Restricted Stock Units—Upon signing a general release, employees are eligible to vest in a pro-rata portion of restricted stock unit awards held at least one year from the grant date; provided that if an employee turns 65 on or prior to their involuntary termination not for cause, then any unvested Restricted Stock Units held for at least one year will have vested in full prior to their involuntary termination not for cause.
Market Share Units—Upon signing a general release, employees are eligible to vest in a pro-rata portion of unvested market share unit awards held at least one year from the grant date, subject to performance provisions; provided that if an employee turns 65 on or prior to their involuntary termination not for cause, then any unvested Market Share Units held for at least one year will vest in full upon their involuntary termination not for cause;cause, subject to performance provisions.
Performance Share Units—If
employees are eligible to vest (1) in any performance share units for which the performance year has been completed before the employee's involuntary termination not for cause and (2) in a
proportionate amount of the performance share units for the performance year during which the employee is involuntarily terminated not for cause, subject to performance provisions.
Defined Benefit Pension Plans—Employees are eligible for benefits accrued under the Retirement Income Plan and the Benefit Equalization Plan—BEP—Retirement Plan. If the employee's age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee is not eligible for early retirement, and the employee signs a general release, the retirement benefits are payable following termination of employment based upon enhanced adjustment factors similar to those applied to employees eligible for early retirement.
Savings Plans—Employees are eligible for benefits accumulated under the Savings and Investment Program and the Benefit Equalization Plan—BEP—Savings Plan. If the employee is involuntarily terminated not for cause on or after September 30th and the employee signs a general release, or the employee's age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee is not eligible for early retirement, and the employee signs a general release, the employee is eligible for a pro-rata annual and transition (if applicable) contribution based on eligible compensation paid in the year of separation from service.
Post-Retirement Medical Insurance—If the employee's age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee is not eligible for early retirement, and the employee signs a general release, the employee is eligible for continued medical coverage beyond the severance and COBRA period, as long as no other group medical coverage is available, without company subsidy until age 55. At age 55, they become eligible for company-subsidized, post-retirement medical benefits starting at age 55.
Under the Bristol-Myers Squibb Senior Executive Severance Plan, certain senior executives (including the Named Executive Officers) are eligible to receive severance payments and benefits if they are involuntarily terminated not for "cause," where "cause" is defined as:
A terminated executive who signs a general release will be eligible for the following:
Change in Control
As disclosed in the CD&A, the company has entered into change-in-control agreements with certain senior executives, including all of the Named Executive Officers. The current agreements will expire on December 31, 2014,2015, and may be extended with revisions, as appropriate, beginning on January 1, 2015, 2016,
in one-year increments unless either the company or the executive gives prior notice of termination of the agreement or a change in control shall have occurred prior to January 1 of such year.
To trigger benefits, there must be both a change in control of the company and either (i) a subsequent involuntary termination without cause by the company or (ii) a good reason termination by the employee. Good reason includes a reduction in job responsibilities or changes in pay and benefits as well as relocation beyond 50 miles. The executive has 120 days to assert a claim for payments under this provision. This protection extends for 36 months following a change in control for executives who became eligible for change-in-control benefits prior to September 1, 2010 (including all of the Named Executive Officers), or 24 months following a change in control for executives who became eligible for change-in-control benefits after September 1, 2010.
"Change in Control" means the earliest to occur of any one of the following dates:
Each of our Named Executive Officers is eligible to receive the following benefits if he or she is terminated in connection with a change in control:
payout of a proportionate amount of the target performance share units for the performance year during which the change in control occurs.
longer provide any pension subsidy or enhancement for newly eligible executives. In lieu of such subsidy or enhancement, we provide under the non-qualified savings plan a continuation of company matching contributions and automatic year-end contributions equal to the length of the severance period.
The following illustrates the potential payments and benefits under the company's plans and programs to the Named Executive Officers upon a termination of employment assuming an effective date of December 31, 2014. To the extent payments and benefits are generally available to salaried employees on a non-discriminatory basis, they are excluded from the table.
Termination of Employment Obligations (Excluding Vested Benefits)
2014 Fiscal Year
Name | | Cash Severance (1) | | Restricted Stock Units (2)(5) | | Market Share Units (3)(5) | | Performance Share Units (4)(5) | | Retirement (6) | | Health (7) | | Retiree Medical (8) | | Total | | Gross-Up on Excise Taxes (9) | ||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Voluntary Termination for Good Reason | ||||||||||||||||||||||||||||||
Lamberto Andreotti(10) | $ | 3,400,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 19,316 | $ | 0 | $ | 3,419,316 | $ | 0 | ||||||||||||
Charles Bancroft (10) | $ | 1,821,040 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 17,608 | $ | 0 | $ | 1,838,648 | $ | 0 | ||||||||||||
Giovanni Caforio, M.D.(11) | $ | 1,900,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 23,624 | $ | 90,660 | $ | 2,014,284 | $ | 0 | ||||||||||||
Francis Cuss, MB BChir, FRCP(10) | $ | 1,750,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 25,694 | $ | 0 | $ | 1,775,694 | $ | 0 | ||||||||||||
Sandra Leung | $ | 1,730,000 | $ | 0 | $ | 0 | $ | 0 | $ | 2,813,253 | $ | 23,270 | $ | 116,142 | $ | 4,682,665 | $ | 0 | ||||||||||||
Involuntary Termination Not for Cause | | |||||||||||||||||||||||||||||
Lamberto Andreotti(10) | $ | 3,400,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 19,316 | $ | 0 | $ | 3,419,316 | $ | 0 | ||||||||||||
Charles Bancroft(10) | $ | 1,821,040 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 17,608 | $ | 0 | $ | 1,838,648 | $ | 0 | ||||||||||||
Giovanni Caforio, M.D.(11) | $ | 1,900,000 | $ | 439,183 | $ | 777,838 | $ | 3,864,426 | $ | 0 | $ | 23,624 | $ | 90,660 | $ | 7,095,732 | $ | 0 | ||||||||||||
Francis Cuss, MB BChir, FRCP(10) | $ | 1,750,000 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 25,694 | $ | 0 | $ | 1,775,694 | $ | 0 | ||||||||||||
Sandra Leung | $ | 1,730,000 | $ | 0 | $ | 1,597,293 | $ | 5,006,097 | $ | 2,813,253 | $ | 23,270 | $ | 116,142 | $ | 11,286,055 | $ | 0 | ||||||||||||
Qualifying Termination Within 3 Years Following a Change in Control | | |||||||||||||||||||||||||||||
Lamberto Andreotti(12) | $ | 12,707,500 | $ | 0 | $ | 15,662,666 | $ | 0 | $ | 961,256 | $ | 10,013 | $ | 0 | $ | 29,341,435 | $ | 0 | ||||||||||||
Charles Bancroft(12) | $ | 5,444,910 | $ | 1,333,842 | $ | 4,232,628 | $ | 0 | $ | 4,587,429 | $ | 53,640 | $ | 0 | $ | 15,652,448 | $ | 7,577,024 | ||||||||||||
Giovanni Caforio, M.D.(11) | $ | 6,249,100 | $ | 442,725 | $ | 3,665,291 | $ | 3,864,426 | $ | 0 | $ | 72,142 | $ | 99,575 | $ | 14,393,259 | $ | 5,596,741 | ||||||||||||
Francis Cuss, MB BChir, FRCP(12) | $ | 5,232,500 | $ | 539,475 | $ | 2,391,955 | $ | 0 | $ | 2,493,269 | $ | 78,300 | $ | 0 | $ | 10,735,499 | $ | 4,822,112 | ||||||||||||
Sandra Leung | $ | 5,172,700 | $ | 0 | $ | 4,798,608 | $ | 5,006,097 | $ | 6,143,900 | $ | 71,084 | $ | 113,195 | $ | 21,305,584 | $ | 8,127,159 | ||||||||||||
parachute payment subject to excise tax. We have committed to eliminate remaining excise tax gross ups in change-in-control agreements for grandfathered executives, including all of the Named Executive Officers listed above, effective January 1, 2016.
ITEM 2—ADVISORY VOTE TO APPROVE THE COMPENSATION OF OUR NAMED
EXECUTIVE OFFICERS
As required by Section 14A of the Securities Exchange Act of 1934, as amended, we are providing stockholders the opportunity to advise the Compensation and Management Development Committee and the Board of Directors regarding the compensation of our Named Executive Officers, as such compensation is described in the Compensation Discussion and Analysis (CD&A) section, the tabular disclosure regarding such compensation and the accompanying narrative disclosure, beginning on page 29. We strongly encourage you to read these sections for a detailed description of our executive compensation philosophy and programs, the compensation decisions the Committee has made under those programs, the factors considered in making those decisions, the changes made to such programs in 2014 and the feedback we received from our stockholder engagement. Accordingly, we are requesting your nonbinding vote on the following resolution:
"RESOLVED, that the stockholders of Bristol-Myers Squibb Company approve, on an advisory basis, the compensation of the Company's Named Executive Officers, as described in the Compensation Discussion and Analysis section, the tabular disclosure regarding such compensation and the accompanying narrative disclosure set forth in the Company's 2015 Proxy Statement."
Our executive compensation programs are designed to enable us to attract and retain talented employees capable of leading our business in the highly complex and competitive business environment in which we operate. We seek to accomplish this goal in a way that rewards performance and is aligned with our stockholders' long-term interests. A significant portion of each executive's pay depends on his or her individual performance against financial and operational objectives as well as key behaviors necessary to accelerate our ongoing evolution into a diversified specialty biopharmaceutical company. In addition, a substantial portion of an executive's compensation is in the form of equity awards that tie the executive's compensation directly to creating stockholder value and achieving financial and operational results.
We value input from our stockholders as expressed through their votes and other communications. As an advisory vote, this proposal is not binding on the company. However, consistent with our record of shareholder responsiveness, the Compensation and Management Development Committee will consider the outcome of the vote when making future executive compensation decisions.
Accordingly, the Board of Directors unanimously recommends a vote "FOR" the approval, on an advisory basis, of the compensation of our Named Executive Officers.
Equity Compensation Plan Information
The following table summarizes information concerning the company's equity compensation plans and outstanding and exercisable options as of December 31, 2014:
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights (in millions) | Weighted-average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (in millions) | |||||
| | | | | | | | | |
| (a) | (b) | (c) | ||||||
| Equity compensation plans approved by security holders | 26.2(1) | $22.28(1) | 111.9 | |||||
| Equity compensation plans not approved by security holders(2) | 0.01 | $25.51 | 28.5 | |||||
| | | | | | | | | |
| 26.2 | $22.29 | 140.3 | ||||||
| | | | | | | | | |
| | | | | | | | | |
| | | | | | | | | |
ITEM 3—RATIFICATION OF THE APPOINTMENT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
Our Board of Directors, upon the recommendation of its Audit Committee, has ratified the Audit Committee's appointment of Deloitte & Touche LLP (D&T) as our independent registered public accounting firm for the year 2014.2015. The Audit Committee and the Board believe that the continued retention of D&T to serve as our independent registered public accounting firm is in the best interests of the company and its stockholders. As a matter of good corporate governance, we are asking stockholders to ratify such appointment. In the event our stockholders fail to ratify the appointment, the Board of Directors and the Audit Committee will reconsider such appointment. It is understood that even if the appointment is ratified, the Audit Committee at its discretion, may direct the appointment of a new independent registered public accounting firm at any time during the year if the Audit Committee feels that such a change would be in the best interests of our company and our stockholders.
The Audit Committee is directly responsible for appointing, compensating and providing oversight of the performance of our independent registered public accounting firm for the purpose of issuing audit reports and related work regarding our financial statements and the effectiveness of our internal control over financial reporting. The Audit Committee is also responsible for approving the audit fee of our independent registered public accounting firm. In order to assure continuing auditor independence, the Audit Committee periodically considers whether there should be a regular rotation of the independent registered public accounting firm. Further, in conjunction with the mandated rotation of the audit firm's lead engagement partner, every five years, the Audit Committee and its chairperson participate in the process for the selection of D&T's new lead engagement partner.
Representatives from D&T will be present at the Annual Meeting to respond to appropriate questions and to make any statements as they may desire.
The Board of Directors unanimously recommends a vote "FOR" the ratification of the appointment of Deloitte & Touche LLP as Bristol-Myers Squibb's independent registered public accounting firm for 2014.2015.
The following table presents aggregate fees for professional audit services rendered by D&T for the years ended December 31, 20132014 and 20122013 for the audits of our annual financial statements and internal control over financial reporting, and fees billed for other services rendered by D&T during those periods.
| | 2012 | 2013 | | 2013 | | 2014 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | |||||||
| | (in millions) | | (in millions) | |||||||||||
| | | | | | | | | |||||||
Audit | $ | 11.43 | $ | 10.98 | $ | 10.98 | $ | 10.78 | ||||||
Audit Related | 1.61 | 1.21 | 1.21 | 0.99 | ||||||||||
Tax | 7.33 | 7.40 | | 7.40 | | 6.48 | ||||||||
All Other | 0.05 | 0.66 | 0.66 | 0.44 | ||||||||||
| | | | | | | | | | | | | | ||
Total | $ | 20.42 | $ | 20.25 | $ | 20.25 | $ | 18.69 | ||||||
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | |||||||
Audit fees for 20122013 and 20132014 were for professional services rendered for the audits of our consolidated financial statements, including accounting consultation, and of our internal control over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act, statutory and subsidiary audits, timely reviews of quarterly financial statements, consents, and assistance with review of documents filed with the SEC.
Audit Related fees for 20122013 and 20132014 were primarily for agreed-upon procedures, special purpose financial statement audits, due diligence related to acquisitions, and other audit-related services that are not required by statute or regulation.
Tax fees for 20122013 and 20132014 were for services related to tax compliance, including the preparation of tax returns and claims for refund, tax planning (excluding planning related to transactions or proposals for which the sole purpose may be tax avoidance or for which tax treatment may not be supported by the Internal Revenue Code) and tax advice, including assistance with tax audits and appeals, advice related to acquisitions, preparation of individual income tax returns (excluding those of executive officers) and consultations relating to our international compensation matters, and requests for rulings or technical advice from tax authorities.
All Other fees for 20122013 and 20132014 related to subscription fees to an accounting and reporting research library and a pharmaceutical alliance database, as well as surveys, benchmarking, commercial strategy and training programs.
Pre-Approval Policy for Services Provided by our Independent Registered Public Accounting Firm
The Audit Committee has established a policy to pre-approve all audit and permissible non-audit services provided by our independent registered public accounting firm consistent with applicable SEC rules. Our independent registered public accounting firm is prohibited from providing tax consulting services relating to transactions or proposals in which the sole purpose may be tax avoidance or for which the tax treatment may not be supported by the Internal Revenue Code. Prior to the engagement of our independent registered public accounting firm for the next year's audit, a schedule of the aggregate of services expected to be rendered during that year for each of the four categories of services described above is submitted to the Audit Committee for approval. Prior to engagement, the Audit Committee pre-approves these services by category of service. The fees are budgeted by category of service and the Audit Committee receives periodic reports from management and our independent registered public accounting firm on actual fees versus the budget by category of service. During the year, circumstances may arise when it may become necessary to engage our independent registered public accounting firm for additional services not contemplated in the pre-approval. In those instances, the Audit Committee requires specific pre-approval before engaging our independent registered public accounting firm.
The Audit Committee may delegate pre-approval authority to one or more of its members. The member or members to whom such authority is delegated is required to report, for informational purposes, any pre-approval decisions to the Audit Committee at its next regularly scheduled meeting. During 2014, the Audit Committee did not delegate pre-approval authority to any of its members.
As the Audit Committee of the Board of Directors, we are composed of independent directors as required by and in compliance with the listing standards of the New York Stock Exchange. We operate pursuant to a written charter adopted by the Board of Directors that is published on the company's website.
Management has primary responsibility for the company's financial reporting process, principles and internal controls as well as preparation of its consolidated financial statements. The independent registered public accounting firm is responsible for performing an audit in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB) to obtain reasonable assurance that Bristol-Myers Squibb's consolidated financial statements are free from material misstatement and expressing an opinion on the conformity of such financial statements with accounting principles generally accepted in the United States. We are responsible for overseeing and monitoring D&T's auditing process on behalf of the Board of Directors.
As part of the oversight of the company's financial statements, we review and discuss with both management and D&T all annual and quarterly financial statements prior to their issuance. Management advised us that each set of financial statements reviewed was prepared in accordance with accounting principles generally accepted in the United States. We have reviewed with management significant accounting and disclosure issues and reviewed with D&T matters required to be discussed pursuant to auditing standards adopted by the PCAOB.
In addition, we have received the written disclosures and the letter from D&T required by PCAOB Ethics and Independence Rule 3526, "Communication with Audit Committees Concerning Independence", and have discussed with D&T their independence from Bristol-Myers Squibb and its management. We have determined that D&T's provision of non-audit services in 20132014 was compatible with, and did not impair, its independence. We have also received written materials addressing D&T's internal quality control procedures and other matters, as required by the New York Stock Exchange listing standards.
We have discussed with our internal auditors and D&T the overall scope and plans for their respective audits. We have met with the internal auditors and D&T, with and without management present, to discuss their evaluations of the company's internal control over financial reporting, and the overall quality of the company's financial reporting.
Based on the reviews and discussions described above, we recommended to the Board of Directors, and the Board has approved, that the audited consolidated financial statements for the year ended December 31, 20132014 be included in Bristol-Myers Squibb's Annual Report on Form 10-K for the year ended December 31, 20132014 for filing with the Securities and Exchange Commission.
In addition, we have confirmed there have been no new circumstances or developments since our respective appointments to the Committee that would impair any of our member's ability to act independently.
The Audit Committee
Alan J. Lacy, Chair
Laurie H. Glimcher, M.D.
Michael Grobstein
Dinesh C. Paliwal
Gerald L. Storch
ITEM 3—ADVISORY VOTE4—APPROVAL OF AMENDMENT TO APPROVEOUR AMENDED AND RESTATED CERTIFICATE OF INCORPORATION TO DESIGNATE DELAWARE CHANCERY COURT AS THE COMPENSATION OF OUR NAMED EXECUTIVE OFFICERSEXCLUSIVE FORUM FOR CERTAIN LEGAL ACTIONS
As required by Section 14A of the Securities Exchange Act of 1934, as amended, we are providing stockholders the opportunity to advise the Compensation and Management Development Committee and theOur Board of Directors regardinghas approved and recommends that our stockholders approve an amendment to our Amended and Restated Certificate of Incorporation to add a new Article FOURTEENTH designating the compensationCourt of Chancery of the State of Delaware, to the fullest extent permitted by law, as the sole and exclusive forum for specified legal actions unless otherwise consented to by the Company. This designation of the Court of Chancery would apply to (1) any derivative action or proceeding brought on behalf of the Company, (2) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee of the Company to the Company or the Company's stockholders, creditors or other constituents, (3) any action asserting a claim arising pursuant to any provision of the General Corporation Law of the State of Delaware or the Company's Amended and Restated Certificate of Incorporation or Bylaws (as either may be amended from time to time), or (4) any action asserting a claim against the Company or any director, officer or other employee of the Company governed by the internal affairs doctrine.
Plaintiffs seeking to bring claims against the Company for the matters to which the proposed amendment relates could use the Company's diverse operations to bring duplicative suits in multiple jurisdictions or to choose a forum state that may not apply the law of Delaware, our Named Executive Officers, as such compensation is describedstate of incorporation, to the Company's internal affairs in the Compensation Discussionsame manner as the Court of Chancery of the State of Delaware would be expected to do so. The proposed amendment is intended to avoid subjecting the Company to multiple lawsuits in multiple jurisdictions on the same matter. The ability to require such actions to be brought in a single forum provides numerous benefits to the Company and Analysis (CD&A) section,our stockholders.
Specifically, the tabular disclosure regarding such compensationCompany and our stockholders benefit from having disputes resolved by the Delaware Court of Chancery, which is widely regarded as the preeminent court for the determination of disputes involving a corporation's internal affairs in terms of precedent, experience and focus. The Delaware Chancery Court has experienced jurists who have a deep understanding of Delaware corporate law and the accompanying narrative disclosure, beginning on page 27. Accordingly,duties of directors and officers. Delaware's well-developed body of case law provides stockholders with more certainty about the outcome of intra-corporate disputes. By ensuring that intra-corporate disputes are heard in a Delaware court, we are requesting your nonbinding vote onand our stockholders avoid costly and duplicative litigation, the following resolution:
"RESOLVED,risk that the stockholders of Bristol-Myers Squibb Company approve, on an advisory basis, the compensation of the Company's Named Executive Officers, as describedDelaware law would be misapplied by a court in the Compensation Discussion and Analysis section, the tabular disclosure regarding such compensationanother jurisdiction and the accompanying narrative disclosure set forthrisk of inconsistent outcomes when two similar cases proceed in different courts. Lastly, Delaware offers a system of specialized Chancery Courts to deal with corporate law questions, with streamlined procedures and processes that help provide relatively quick decisions. This accelerated schedule can limit the Company's 2014 Proxy Statement."time, cost and uncertainty of protracted litigation for all parties.
Our executive compensation programs are designed to enable us to attract and retain talented employees capableFor these reasons, the Board believes that providing for Delaware as the exclusive forum for the types of leading our business in the highly complex and competitive business environment in which we operate. We seek to accomplish this goal in a way that rewards performance and is aligned with our stockholders' long-term interests. A significant portion of each executive's pay depends on his or her individual performance against financial and operational objectives as well as key behaviors. In addition, a substantial portion of an executive's compensationdisputes described above is in the form of equity awards that tie the executive's compensation directly to creating stockholder value and achieving financial and operational results.
At our annual meeting of stockholders held in May 2013, approximately 96.1%best interests of the votes castCompany and its stockholders. At the same time, the proposed amendment still gives the Company the flexibility to consent to an alternative forum on a case-by-case basis where the proposal atCompany determines that meeting votedits interests and those of its stockholders are best served by permitting a dispute to proceed in favora forum other than Delaware.
It is important to note that this action by the Board is not being taken in reaction to any specific litigation confronting the Company; rather, it is being taken to prevent potential future harm to the Company and its stockholders. In recent years, we have experienced a number of the proposal. The Committee believes this affirms stockholders' support of the company's approach to executive compensation. Nevertheless, as disclosedlitigation matters that were each filed in more detail on page 43, the Committee implemented some enhancements to our annual and long-term incentive programs to mitigate the overlap of performance metrics between those two programs and to further align the interests of our executivesthan one jurisdiction, primarily in connection with the interestsacquisition of our stockholders.
We strongly urge youanother public company. For example, when we acquired Amylin Pharmaceuticals, Inc. in 2013, we and Amylin were sued by Amylin's stockholders in both Delaware and California. The facts and claims raised in both jurisdictions were similar, but we and Amylin had to readincur additional costs to defend ourselves in two different forums. These costs included fees for additional local counsel in California and legal fees and expenses associated with litigating in both forums to determine where the CD&A, which describes our compensation program in greater detail. Highlights include:litigation should proceed. We
faced similar multi-forum litigation when we acquired other companies too, such as Zymogenetics, Inc. and Inhibitex, Inc. Although the proposed amendment would not have covered these cases because they are outside the scope described above, an exclusive forum provision would help reduce our exposure to the potential harm of multi-forum litigation. The proposed amendment, when applicable, would not only help reduce legal fees associated with proceeding in our change-in-control agreements for executives who became eligible for change-in-control benefits after September 1, 2010;two forums, but it would also limit the risk of inconsistent judicial rulings, prevent witnesses from needing to appear in multiple jurisdictions, and
We value inputmaintain strong corporate governance practices, many of which are described in this proxy statement, including a Board that is substantially comprised of independent directors elected on an annual basis, a majority vote standard in uncontested director elections, stockholders' ability to call special meetings and the absence of a stockholder rights plan (poison pill). Additionally, the Board's decision to seek stockholder approval of this exclusive forum provision was influenced by feedback received from our stockholdersthe Company's stockholders.
Although exclusive forum provisions such as expressed through their votesthe one we are proposing are becoming increasingly common, and other communications. As an advisory vote,we know of no reason a court in another state would not be willing to enforce its terms, no assurance can be given that all courts outside of Delaware will enforce the terms of the amendment and transfer any covered proceeding to the Delaware courts.
Exhibit B of this proxy statement sets forth the proposed Certificate of Amendment to the Amended and Restated Certificate of Incorporation. If this proposal is not binding onapproved by our stockholders, the company. However, consistentCertificate of Amendment will be effective upon filing with our recordthe Secretary of shareholder responsiveness, the Compensation and Management Development Committee will consider the outcomeState of the vote when making future executive compensation decisions.State of Delaware, which we intend to do promptly after stockholder approval is obtained.
Accordingly, theThe Board of Directors unanimously recommends a vote "FOR" the approval on an advisory basis, of the compensationamendment to our Amended and Restated Certificate of our Named Executive Officers.Incorporation to designate Delaware Chancery Court as the exclusive forum for certain legal actions.
Equity Compensation Plan InformationITEM 5—APPROVAL OF AMENDMENT TO OUR AMENDED AND RESTATED CERTIFICATE
OF INCORPORATION TO REMOVE THE SUPERMAJORITY VOTING PROVISIONS
APPLICABLE TO PREFERRED STOCKHOLDERS
Our Amended and Restated Certificate of Incorporation includes two supermajority voting provisions applicable solely to our preferred stockholders. At our 2014 Annual Meeting of Stockholders, a stockholder proposal requesting the elimination of all supermajority voting provisions in our Amended and Restated Certificate of Incorporation received support from a majority of shares cast. The following table summarizes information concerning the company's equity compensation plansBoard, in its continuing review of corporate governance best practices and outstanding and exercisable options as of December 31, 2013:
Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights (in millions) | Weighted-average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (in millions) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | (a) | (b) | (c) | |||||||
Equity compensation plans approved by security holders | 35.2 | (1) | $ | 22.87 | (1) | 114.4 | ||||
Equity compensation plans not approved by security holders(2) | 0.1 | $ | 25.39 | 28.4 | ||||||
| | | | | | | | | | | |
| 35.3 | $ | 22.88 | 142.8 | |||||||
| | | | | | | | | | | |
| | | | | | | | | | | |
The text of the first paragraph immediately following the heading "Preferred Stock" in Article FOURTH as proposed to be amended is set forth below with additions indicated by underlining and deletions by strike-out:
The affirmative vote of the holders of at leasttwo-thirdsa majority of the Preferred Stock at the time outstanding voting only as a class shall be required to make effective any amendment to the Certificate of Incorporation or by-laws of the corporation altering materially any existing provisions of the Preferred Stock, or authorizing a class
of preferred stock ranking prior to the Preferred Stock as to dividends or assets, and the affirmative vote of the holders of at least a majority of the Preferred Stock at the time outstanding voting only as a class shall be required to make effective any amendment to the Certificate of Incorporation of the corporation authorizing the issuance of or any increase in the authorized amount of any class of preferred stock ranking on a parity with or increasing the number of authorized shares issuable upon vesting of the Preferred Stock.
In addition, the text of the second paragraph of Section (c) of the Certificate of Designation as proposed to be amended is set forth below with additions indicated by underlining and deletions by strike-out:
So long as any shares of such series are outstanding, restricted stock units, market share unitsthe consent of the holders of at leasttwo-thirdsa majority of the shares of such series at the time outstanding, given in person or performance share unitsby proxy, either in writing or at a meeting at which have no exercise price. At December 31, 2013, there werethe holders of the shares of such series shall vote separately as a totalclass, shall be necessary for effecting the amendment, alteration or repeal of approximately 6.6 millionany provision of the Certificate of Incorporation of the Corporation, any certificate amendatory thereof or supplemental thereto, or the by-laws of the Corporation so as to affect materially any of the powers, preferences and rights of the shares subject to restricted stock units, approximately 1.8 million shares subject to market share units and approximately 3.8 million shares subject to performance share units.
If our stockholders approve this plan since 2006 and no future awardsproposal, all supermajority voting provisions will be made undereliminated from our Amended and Restated Certificate of Incorporation. Exhibit C of this plan.proxy statement sets forth the proposed Certificate of Amendment to the Amended and Restated Certificate of Incorporation. If this proposal is approved by both the holders of a majority of our outstanding shares and at least two-thirds of our outstanding shares of preferred stock, the Certificate of Amendment will be effective upon filing with the Secretary of State of the State of Delaware, which we intend to do promptly after stockholder approval is obtained. At our 2010 Annual Meeting, the Board also asked our stockholders to approve an amendment to our Amended and Restated Certificate of Incorporation to remove the supermajority voting requirements applicable to the preferred stockholders. The proposal was not approved because less than two-thirds of the outstanding preferred shares voted in favor of the proposal. We currently have only 4,191 shares of our $2.00 Convertible Preferred Stock outstanding as of February 6, 2015. At last year's annual meeting, the number of preferred shares present in person or by proxy and entitled to vote would not have constituted a quorum for this type of proposal. We intend to target our preferred stockholders with additional solicitations with respect to this proposal, however, there can be no assurance that a sufficient number of preferred stockholders will be present in person or by proxy to constitute a quorum at the 2015 Annual Meeting or that the proposal will be approved.
The Board of Directors unanimously recommends a vote "FOR" the approval of the amendment to our Amended and Restated Certificate of Incorporation to remove the supermajority provisions applicable to preferred stockholders.
We expect the following stockholder proposal (Item 4)6) to be presented at the 20142015 Annual Meeting. The Board of Directors is not recommendinghas recommended a vote "FOR" or "AGAINST" Item 4against this proposal for the policy reasons as set forth following the proposal. The stock holdings of thea proponent will be provided upon request to the Corporate Secretary of Bristol-Myers Squibb.
ITEM 4—6—STOCKHOLDER PROPOSAL ON SIMPLE MAJORITY VOTESHAREHOLDER ACTION BY WRITTEN CONSENT
The proponent of this resolution is Kenneth Steinerthe Trust for the International Brotherhood of 14 Stoner Ave., 2M, Great Neck, New York 11021.Electrical Workers' Pension Benefit Fund of 900 Seventh Street, NW, Washington, D.C. 20001.
RESOLVED, Resolved: Shareholders request that our board takeof directors undertake such steps as may be necessary to permit written consent by shareholders entitled to cast the steps necessary sominimum number of votes that each voting requirement in our charter and bylaws that calls for a greater than simple majority vote be eliminated, and replaced by a requirement for a majority of the votes cast for and against applicable proposals, or a simple majority in compliance with applicable laws. If necessary this means the closest standard to a majority of the votes cast for and against such proposals consistent with applicable laws. This includes the 75% provision in our Charter.
Shareowners are willingwould be necessary to payauthorize the action at a premium for shares of corporations that have excellent corporate governance. Supermajority voting requirements have been foundmeeting at which all shareholders entitled to vote thereon were present and voting. This written consent is to be oneconsistent with giving shareholders the fullest power to act by written consent in accordance with applicable law. This includes shareholder ability to initiate any topic for written consent consistent with applicable law.
Supporting statement:
Shareholders have taken a renewed interest in companies providing them the right to act by written consent. In the past two years, investors filed 55 proposals on this topic with high average support levels: 39% in favor in 2014 and 41% in favor in 2013. In addition, this proposal received majority support at three firms in the past two years: Allergan, Occidental Petroleum and Duke Energy Corporation.
This issue is of six entrenching mechanisms that are negatively relatedparticular concern at Bristol-Myers Squibb where the bar for shareholders' ability to call a special meeting is particularly high at 25%. A more workable threshold to grant shareholders this ability would be 10%. Shareholder action by written consent could save our company performance according to "What Matters in Corporate Governance" by Lucien Bebchuk, Alma Cohen and Allen Ferrellthe cost of the Harvard Law School. Supermajority requirements are arguably most often used to block initiatives supported by most shareowners but opposed byholding a status quo management.physical meeting between annual meetings.
This proposal topic won 74%would empower shareholders by giving them the ability to 88% supporteffect change at Weyerhaeuser, Alcoa, Waste Management, Goldman Sachs, FirstEnergy, McGraw-Hill and Macy's. The proponents of these proposals included Ray T. Chevedden and William Steiner. Currentlyour company without being forced to wait until an annual shareholder meeting. Shareholders should be able to express their views on a 1%-minority can frustrate the will of our 74%-shareholder majority.more frequent basis than once a year.
This proposal should also be more favorably evaluated dueTherefore, we urge shareholders to our Company's clearly improvable environmental, social and corporate governance performance as reported in 2013:
In regard to executive pay there was $17 million for Lamberto Andreotti. GMI Ratings, an independent investment research firm, said Bristol-Myers could give long-term incentive pay to our CEO for below-median performance. Unvested equity pay would not lapse upon CEO termination.
Lead Director Lewis Campbell had 15-years independence-detracting tenure and received our highest negative votes—into double digits. Togo West chaired our executive pay committee. Mr. West's resume included director experience at Krispy Kreme and AbitibiBowater leading up to their bankruptcies. We also had a long-tenured former CEO on our executive pay committee, Lewis Campbell. A former CEO on an executive pay committee is a practice not associated with moderation.
Bristol-Myers admitted the SEC had launched an investigation under the Foreign Corrupt Practices Act into its sales and marketing practices in various countries. Louisiana Attorney General James Caldwell's office said Bristol Myers [sic] would pay $7 million to resolve allegations they misreported drug price information to the state's Medicaid program.
GMI said there were forensic accounting ratios related to asset-liability valuation that had extreme values either relative to industry peers or to the company's own history. GMI said BMY was rated as having Very Aggressive Accounting & Governance Risk. This indicated higher accounting and governance risk than 96% of companies. Bristol-Myers had a higher shareholder class action litigation risk than 99% of all rated companies.
Returning to the core topic ofvote FOR this proposal from the context of our clearly improvable corporate climate, please vote to protect shareholder value:proposal.
Simple Majority Vote—Proposal 4
The Board of Directors is not recommendingrecommends a vote "FOR" or "AGAINST" this proposal for the following reasons:
OurThe Board believes that generally,this proposal is not in the best interests of the stockholders because, unlike meetings of stockholders, action by written consent would disenfranchise certain stockholders by denying them the ability to vote or otherwise have a say on proposed stockholder actions. Action by written consent would enable the holders of just a majority of our governing documents shouldoutstanding shares to take action on a proposal without the benefit of hearing the views, questions and arguments of other stockholders or the company. In addition, action by written consent eliminates the need for advance notice to be given to stockholders about a proposed action, and therefore, certain stockholders may not contain supermajority voting provisions. Atbe informed about the proposed action until after the action has already been taken. This would deny these stockholders the ability to determine whether to exercise their rights. The Board, therefore, believes that this proposal could adversely affect the conduct of stockholder business by resulting in our 2010 Annual Meeting,taking of an action that we otherwise would not have taken if all of our stockholders were afforded the opportunity to discuss, debate and vote on the matter.
The Board in its continuing reviewalso believes that adoption of this proposal is unnecessary because the company is committed to high standards of corporate governance bestand has already taken a number of steps to achieve greater transparency and accountability to stockholders. These corporate governance practices and after careful consideration, askedpolicies cover a wide range of matters and are described beginning on page 6 of this Proxy Statement under the heading "Corporate Governance and Board Matters." Our Board continually reassesses our corporate governance practices to identify additional steps to further benefit our stockholders. In recent years, our Board recommended, and our stockholders to approveapproved, amendments to our Amended and Restated Certificate of Incorporation to removeeliminate all of the supermajority voting requirements remaining in our Certificate. These requirements consisted of a provisionprovisions applicable to our common stockholders that requiredand to permit stockholders holding a voteminimum of at least 75% of the holderstwenty-five percent of our outstanding shares of common stock to eliminatecall a special meeting of stockholders. Furthermore, the Board's Committee on Directors and Corporate Governance has created a process for stockholders to communicate directly with our non-management directors outside the annual electionmeeting cycle, which is described on page 20 of directors and approve a classifiedthis Proxy Statement under the heading "Communications with our Board structure, as well as two provisions applicable to our preferred stockholders that require a two-thirds vote of preferred stock on any proposed amendments to the Certificate or by-laws that would materially alter the existing provisions or powers, preferences or rights of the preferred stock.Directors."
The proposalPermitting stockholder action by written consent has the potential to removecreate substantial confusion and disruption, and the 75% supermajority voting provision applicableBoard does not believe it is part of an appropriate corporate governance model for a widely-held public company. Multiple groups of stockholders would be able to common stockholders referencedsolicit written consents at any time and as frequently as they choose on a range of special or self-interested issues. There also is the possibility that consent solicitations may conflict with one another, be duplicative, or not be in the above proposal and the prior paragraph was approved, and we made the applicable state filings to remove this provision from our Certificate. However, the proposal to remove the supermajority voting provisions applicable to preferred stockholders was not approved because less than two-thirdsbest interests of the outstanding preferredcompany or the stockholders as a whole. The written consent process that this proposal seeks to authorize can be cumbersome, time consuming, and may lead to a chaotic state of corporate affairs. The Board believes that holding meetings whereby all stockholders may discuss the proposed actions and vote their shares votedis the best way for stockholders to take action and helps to ensure the accuracy and completeness of information presented to stockholders to obtain their approval. The Board, therefore, believes that action by written consent is not in favorthe best interests of the proposal as required by our Amended and Restated Certificate of Incorporation. In view of the stockholder vote in 2010, the Board has decided not to take a position for or against the stockholder resolution this year so it can better assess stockholder interest in this issue. Our Bylaws do not have any supermajority voting provisions.stockholders.
Accordingly, the Board of Directors is not recommendingunanimously recommends a vote "FOR" or "AGAINST" thisthe proposal.
Advance Notice Procedures
As set forth in our Bylaws, if you wish to propose any action, including the nomination of directors, at next year's annual meeting, you must deliver notice to BMS containing certain information set forth in our Bylaws, not less than 90 but not more than 120 days before the anniversary of the prior year's annual meeting. For our 20152016 Annual Meeting, we must receive this notice between January 6, 20152016 and February 5, 2015.2016. These requirements are separate and distinct from the SEC requirements that a stockholder must meet to have a stockholder proposal included in our proxy statement. For further information on how a stockholder may nominate a candidate to serve as a director, please see page 12.13.
Our Bylaws are available on our website at www.bms.com/ourcompany/governance. In addition, a copy of the Bylaw provisions discussed above may be obtained by writing to us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Corporate Secretary.
20152016 Stockholder Proposals
Stockholder proposals relating to our 20152016 Annual Meeting of Stockholders must be received by us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Corporate Secretary, no later than November 19, 2014.24, 2015. Such proposals must comply with SEC regulations under Rule 14a-8 regarding the inclusion of stockholder proposals in company sponsored proxy materials. Stockholders are encouraged to contact the Office of the Corporate Secretary prior to submitting a stockholder proposal or any time they have a concern. At the direction of the Board of Directors, the Office of the Corporate Secretary acts as corporate governance liaison to stockholders.
Categorical Standards of Independence
In determining director independence, the Board has adopted the following categorical standards to assist it in determining which relationships will be considered immaterial:
CERTIFICATE OF AMENDMENT
TO THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION
OF
BRISTOL-MYERS SQUIBB COMPANY
Pursuant to Section 242
of the General Corporation Law of the State of Delaware
Bristol-Myers Squibb Company, a corporation duly organized and existing under the General Corporation Law of the State of Delaware (the "Corporation"), does hereby certify that:
1. The Amended and Restated Certificate of Incorporation of the Corporation (the "Certificate of Incorporation") is hereby amended by inserting the following as a new Article FOURTEENTH immediately following Article THIRTEENTH:
"FOURTEENTH: Unless the corporation consents in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware shall, to the fullest extent permitted by law, be the sole and exclusive forum for (1) any derivative action or proceeding brought on behalf of the corporation, (2) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee of the corporation to the corporation or the corporation's stockholders, creditors or other constituents, (3) any action asserting a claim arising pursuant to any provision of the General Corporation Law of the State of Delaware or this certificate of incorporation or the by-laws (as either may be amended from time to time), or (4) any action asserting a claim against the corporation or any director, officer or other employee of the corporation governed by the internal affairs doctrine; provided, however, that, if the Court of Chancery of the State of Delaware lacks jurisdiction over any such action or proceeding, the sole and exclusive forum for such action or proceeding shall be another state or federal court located within the State of Delaware. Any person or entity purchasing or otherwise acquiring or holding any interest in shares of capital stock of the corporation shall be deemed to have notice of and consented to the provisions of this Article FOURTEENTH."
2. The foregoing amendment was duly adopted in accordance with the provisions of Section 242 of the General Corporation Law of the State of Delaware.
IN WITNESS WHEREOF, the Corporation has caused this Certificate of Amendment to be executed by a duly authorized officer on this day of , 20 .
BRISTOL-MYERS SQUIBB COMPANY | ||||
By: | ||||
| Name: | ||||
| Title: | ||||
CERTIFICATE OF AMENDMENT
TO THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION
OF
BRISTOL-MYERS SQUIBB COMPANY
Pursuant to Section 242
of the General Corporation Law of the State of Delaware
Bristol-Myers Squibb Company, a corporation duly organized and existing under the General Corporation Law of the State of Delaware (the "Corporation"), does hereby certify that:
1. The Amended and Restated Certificate of Incorporation of the Corporation (the "Certificate of Incorporation") is hereby amended by deleting "two-thirds" from the first paragraph immediately following the heading "Preferred Stock" in Article FOURTH of the Certificate of Incorporation and inserting "a majority" in lieu thereof.
2. The Certificate of the Designation, Preferences and Relative, Participating, Optional or Other Special Rights of the $2.00 Convertible Preferred Stock of the Corporation (the "Certificate of Designation"), which is attached as Appendix A to the Certificate of Incorporation of the Corporation, is hereby amended by deleting "two-thirds" from the second paragraph of Section (c) of the Certificate of Designation and inserting "a majority" in lieu thereof.
3. The foregoing amendments were duly adopted in accordance with the provisions of Section 242 of the General Corporation Law of the State of Delaware.
IN WITNESS WHEREOF, the Corporation has caused this Certificate of Amendment to be executed by a duly authorized officer on this day of , 20 .
BRISTOL-MYERS SQUIBB COMPANY | ||||
By: | ||||
| Name: | ||||
| Title: | ||||
DIRECTIONS TO OUR PLAINSBORO OFFICE AT
777 SCUDDERS MILL ROAD
PLAINSBORO, NJ 08536
By Car:
From New York:
Take the Lincoln Tunnel.
Take the New Jersey Turnpike South/Newark Exit.
Exit Left onto I-95 South.
Merge onto NJ-18 North Via Exit 9 toward US-1/New Brunswick/Princeton.
Merge onto US-1 South toward Trenton.
Take ramp onto Scudders Mill Rd.
Our offices are approximately 11/2 mile on the left side of the road.
From Philadelphia:
Take I-95 North.
Merge onto US-1 North via Exit 67A toward New Brunswick.
Turn Slight Right onto Scudders Mill Road.
Our offices are approximately 11/2 mile on the left side of the road.
By Train:
New Jersey Transit and Amtrak train service is available to Princeton Junction, New Jersey. Our Plainsboro office is approximately a 10 minute car drive from the station.
Parking:
Free parking for stockholders attending the 20142015 Annual Meeting is available. Please go directly to the parking area reserved for stockholders.
Y O U R V O T E I S I M P O R T A N T
P L E A S E V O T E Y O U R P R O X Y
![]()
![]()
|
|
| ADMISSION TICKET |