Pipeline | | • File investigational new drug, or IND, application for inclacumab • File IND for GBT601 (1) | In connection with his promotion• File inclacumab IND by third quarter
| | 20% (subject to Chief Medical Officer and additional responsibilities in October 2019, Dr. Lehrer-Graiwer’s 2019 target annual bonus was increased from 35%increase to 40% upon achievement of his annual base salary, effective October 1, 2019.the stretch goal) | | | | | Corporate | | • Achieve certain business development goals • Complete year with a cash reserve of at least a certain minimum • Maintain voluntary turnover below industry standard | | | | 10% |
In February 2019, we hired Dr. Cathers as our Chief Scientific Officer. The Compensation Committee considered the factors described in “Governance of Executive Compensation Program—Compensation-Setting Factors” above, particularly market data from our compensation peer group for comparable positions and alignment with our other executive officers at the same level, in approving Dr. Cathers’ 2019 target annual bonus of 40%.
Global Blood Therapeutics, Inc. ï2021 Proxy Statement39
20192020 Annual Cash Bonuses
In January 2020,2021, the Compensation Committee evaluated our achievement of the 20192020 corporate performance goals. The Compensation Committee considered whether we had achieved each goal, the weighting established for each goal, including the weighting for overachievement, management’s self-assessment, and our overall corporate performance in 2019. Based on these considerations, the Compensation Committee approved a 150% achievement level of the 2019 corporate performance goals due in part to certain extraordinary achievements, including: (i) our receipt of approval by the FDA for Oxbryta three months in advance of its PDUFA target action date, (ii) the successful acceleration of our sales force hiring and commercial launch readiness, (iii) our completion of our strategic Syros Collaboration in furtherance of our pipeline and (iv) our success in exceeding our 2019year-end cash goal with our equity and debt financings.2020. | | | | | | | | | | | Goal | | Weighting | | Actual Results | | Bonus Pool
Funding | | | | | Oxbryta Net Sales of $158 million in 2020 (stretch of $178 million in 2020) | | 50%
(75% with stretch) | | $123.6 million in 2020 | | 39.6% | | | | | First dose in clinical study of Oxbryta at doses outside of approved labeling by third quarter | | 5% | | Achieved in September | | 5% | | | | | Regulatory—Gain alignment with FDA on label expansion in ages less than 12 years old and secure scientific advice to inform EU strategy | | 5% | | FDA Type B meeting held in May EU MAA submitted in December | | 5% | | | | | Geographic Expansion—Execute Saudi Arabi partnering/distributor agreement (stretch goal by third quarter) and advance plan for certain other geographies | | 10%
(20% with stretch) | | Distribution Agreement for Gulf Cooperation Council region signed in August, achieving stretch goal of third quarter timing | | 20% | | | | | Pipeline—File IND for inclacumab (stretch goal of filing such IND by third quarter) and IND for GBT601 | | 20%
(40% with stretch) | | Both INDs filed in November | | 20% | | | | | Corporate • Achieve certain business development goals • Complete year with a cash reserve of at least a certain minimum • Maintain voluntary turnover below the industry standard | | 10% | | • Confidential business development goals achieved by year end • Ended 2020 with approximately $580 million in cash, which significantly exceeded the specified minimum • GBT 2020 voluntary turnover at 10.3% compared to life science market at 13.2% | | 10% | | | Total | | 99.6% |
The Compensation Committee also reviewed the 20192020 individual performance of each of our executive officers, other than our CEO, based on an evaluation conducted by our CEO of their performance against their 20192020 individual performance goals.and contribution to the achievement of the corporate objectives above. The Compensation Committee approved an achievement level of 100%105% of the 20192020 individual performance goals for each of our NEOs.NEOs based upon their scope of responsibilities and significant contributions to us meeting our corporate objectives in 2020. The table below sets forth the target annual cash bonuses, the relative weighting of corporate and individual performance, the actual achievement level for corporate and individual performance and the 20192020 annual cash bonuses earned by our NEOs. | | | | | | | | | | | | | | | | | | | | | | | | | NEO | | 2019 Annual
Base Salary
($) | | | Target Annual
Cash Bonus
(% of annual
base salary) | | | Weighting
(corporate/
individual
performance)
(%) | | | Corporate
Performance
(%) | | | Individual
Performance
(%) | | | Annual Cash
Bonus
($) | | Dr. Love | | $ | 600,000 | | | | 60 | % | | | 100%/0 | | | | 150 | % | | | N/A | | | $ | 540,000 | | Mr. Farrow(1) | | $ | 442,500 | | | | 40 | % | | | 75%/25% | | | | 150 | % | | | 100 | % | | $ | 244,000 | | Dr. Cathers (1)(2) | | $ | 375,000 | | | | 40 | % | | | 75%/25% | | | | 150 | % | | | 100 | % | | $ | 172,000 | | Mr. Johnson(1) | | $ | 455,000 | | | | 40 | % | | | 75%/25% | | | | 150 | % | | | 100 | % | | $ | 250,000 | | Dr. Lehrer-Graiwer | | $ | 460,000 | | | | 40 | % | | | 75%/25% | | | | 150 | % | | | 100 | % | | $ | 253,000 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | NEO | | 2020 Annual
Base Salary
($) | | Target Annual
Cash Bonus
(% of annual
base salary) | | Weighting
(corporate/
individual
performance)
(%) | | Corporate
Performance
(%) | | Individual
Performance
(%) | | Annual Cash
Bonus
($) | | | | | | | | Dr. Love | | | $ | 670,000 | | | | | 70 | % | | | | 100 | %/0 | | | | 99.6 | % | | | | N/A | | | | $ | 467,000 | | | | | | | | | Mr. Farrow | | | $ | 460,000 | | | | | 50 | % | | | | 80 | %/20% | | | | 99.6 | % | | | | 105 | % | | | $ | 231,500 | | | | | | | | | Mr. Johnson | | | $ | 465,000 | | | | | 50 | % | | | | 80 | %/20% | | | | 99.6 | % | | | | 105 | % | | | $ | 234,000 | | | | | | | | | Ms. Choi | | | $ | 435,000 | | | | | 50 | % | | | | 80 | %/20% | | | | 99.6 | % | | | | 105 | % | | | $ | 219,000 | | | | | | | | | Ms. Suvari | | | $ | 425,000 | | | | | 50 | % | | | | 80 | %/20% | | | | 99.6 | % | | | | 105 | % | | | $ | 214,000 | |
(1) | Annual cash bonus was immaterially adjusted for rounding.
|
(2) | Dr. Cathers’ employment commencement date was February 25, 2019, and his 2019 annual cash bonus was prorated based on his employment with us for approximately ten months of 2019.
|
402021 Proxy Statementï Global Blood Therapeutics, Inc.
The annual cash bonuses earned by each of our NEOs for 20192020 are set forth in the “Summary Compensation Table” below. Long-Term Incentive Compensation We view long-term incentive compensation in the form of equity awards as an important element of our executive compensation program. The value of equity awards is directly related to stock price appreciation over time, which incentivizes our executive officers to achieve long-term corporate goals and create long-term value for our stockholders. Equity awards also help us attract and retaintop-performing executive officers in a competitive market. At the time of hire, equity awards are granted to our executive officers, including our NEOs, based on the factors described in “Governance of Executive Compensation Program—Compensation-Setting Factors” above. Typically, at the beginning of each year, the Compensation Committee reviews the equity awards for our executive officers, including our NEOs, and determines the size and relative weighting of the annual equity awards it deems reasonable and appropriate based on such factors.the factors described in “Governance of Executive Compensation Program—Compensation-Setting Factors” above. The size and relative weighting is the same for
each of our executive officers, including our NEOs, who are at the same level. In addition, the Compensation Committee may deem it advisable to grant subsequent equity awards to our executive officers, including our NEOs, and may adjust their equity awards in the event of a promotion or significant change in responsibilities. 20192020 Equity Awards
2019 Annual Equity Awards
In January 2019,2020, the Compensation Committee considered the factors described in “Governance of Executive Compensation Program—Compensation-Setting Factors” above, particularly market data from the companies in our compensation peer group and selected broad-based compensation surveys, and approved the 20192020 annual equity awards for our NEOs except Dr. Cathers, below. Mr. Johnson received a larger grant of time-based RSUs than the other non-CEO NEOs, as 5,000 of such RSUs were granted in recognition of his contribution toward us achieving a $4 billion market capitalization milestone. | | NEO | | Stock Options
(Number of Shares) | | | Time-Based RSUs
(Number of Shares) | | | Stock Options
(Number of Shares) | | Time-Based RSUs
(Number of Shares) | | Dr. Love | | | 145,000 | | | | 90,000 | | | 114,190 | | 71,640 | | Mr. Farrow | | | 40,000 | | | | 25,000 | | | 38,500 | | 24,160 | | Mr. Johnson | | | 40,000 | | | | 25,000 | | | 38,500 | | 29,160 | Dr. Lehrer-Graiwer | | | 35,000 | | | | 23,500 | | | | Ms. Choi | | | 38,500 | | 24,160 | | Ms. Suvari | | | 38,500 | | 24,160 |
The stock options vest, and become exercisable, over a four-year period, with 1/16th of the underlying shares vesting on a quarterly basis (every three months) after the vesting commencement date of February 1, 2019,2020, so that all of the underlying shares will be vested on the date four years after the vesting commencement date, so long as the NEO remains an employee or other service provider (including a consultant) of the Company on each such vesting dates.date. The time-based RSUs vest over a four-year period, with 1/8th of the underlying shares vesting on a semi-annual basis (every six months) after the vesting commencement date of February 1, 2019,2020, so that all of the underlying shares will be vested on the date four years after the vesting commencement date, so long as the NEO remains an employee or other service provider (including a consultant) of the Company on each such vesting dates.date. 2019 Equity AwardsShortly after the World Health Organization declared COVID-19 a pandemic in March 2020, the Compensation Committee began to review whether the above annual equity awards approved in January 2020 would be insufficient in light of the pandemic and our reduced stock price to serve as an appropriate motivational and retention vehicle for New Executive Officersour executive officers, particularly as we were entering our first full year of the commercial launch of our first medicine, Oxbryta. To address these concerns, the Compensation Committee decided to grant an additional performance-based equity award to our executive officers, including our NEOs, to provide an opportunity for them to earn equity compensation in line with the 75th percentile of our peer group’s equity compensation for similarly situated executives.
In Februarydetermining the form of additional equity to grant the NEOs, the Compensation Committee revisited the positive impact of the PSUs the Committee granted our executive officers in August 2017, or 2017 PSUs. The 2017 PSUs were scheduled to vest, if ever, upon the first instance of us achieving a market capitalization of certain specified amounts of up to $4 billion on or before December 31, 2019, we hired Dr. Catherswhich the Compensation Committee considered as our Chief Scientific Officer.a significant threshold at the time such PSUs were granted. The Compensation Committee consideredconcluded that the factors described2017 PSUs had significant motivation and retention value, including in “Governanceour achievement of Executivethe highest market capitalization threshold of $4 billion, which resulted in the vesting of the final tranche of the 2017 PSUs in December 2019. Effective June 1, 2020, the Compensation Program—Compensation-Setting Factors” above, particularly market data from our compensation peer group for comparable positions, in approving Dr. Cathers’ new hire equity awards as follows: a stock optionCommittee granted PSUs to purchase 45,000 sharescertain of our commonsenior management, including our NEOs, which are eligible to be earned and vest contingent upon the achievement of three escalating and challenging stock Global Blood Therapeutics, Inc. ï2021 Proxy Statement41
price targets ($109.20, $145.60 and 30,000 time-based RSUs. The stock options vest, and become exercisable,$182.00 per share), based on average closing market price on the NASDAQ Global Select Market over a four-year20 consecutive trading day period, with 1/4ththe percentage of shares allotted to the three tranches increasing with each tranche. Upon the achievement of each respective stock price target, 50% of the underlying shares vestingPSUs allotted to that tranche will vest, while the remaining 50% will vest on the first anniversary of the vesting commencement date of February 25, 2019, and thereafter, 1/12th of the remaining underlying shares vest on a quarterly basis so that all ofstock price target was achieved, subject to the underlying shares will be vested on the date four years after the vesting commencement date, so long as Dr. Cathers remains an employeeexecutive officer’s continued employment or other service provider (including a consultant)relationship with us through such vesting date. The Compensation Committee included such delayed vesting to further foster retention and alignment with stockholder interest. Under the terms of the Companyawards, if the stock price targets are not achieved for all or some of the tranches on such vesting dates.or before June 30, 2024, the unvested awards will be automatically terminate and be forfeited.
The time-based RSUs vestfollowing illustrates the market capitalization we would need to achieve to meet each of the stock price targets and the relative shareholder value created: | | | | | | | | | | | | | | | | | | | | | | Stock Price | | % Increase | | Approx.
Market Cap* | | NEOs
Shares
Earned | | NEOs Value
Earned | | Shareholder
Value
Creation | | Payouts as a % of
Shareholder Value | | | NEOs | | Shareholders | | | | | | | | | $109.20 | | +50% | | $6.7B | | 62,200 | | $6.8M | | +$1.9B | | 0.3% | | 99.7% | | | | | | | | | $145.60 | | +100% | | $9.0B | | 171,050 | | $24.9M | | +$4.2B | | 0.6% | | 99.4% | | | | | | | | | $182.00 | | +150% | | $11.2B | | 311,000 | | $56.6M | | +$6.4B | | 0.9% | | 99.1% |
| * | Approximate market cap is calculated from ~61.7M total common shares outstanding. |
Shareholder Value Created at Maximum Payout 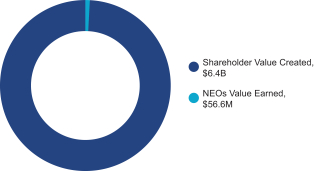
The Compensation Committee granted these performance-based awards as a way to further align the incentives of our senior management, including our NEOs, with our stockholders and to further our pay-for-performance compensation philosophy, as the PSUs will only be earned if our stock price appreciates significantly over a four-year period, with 1/4thperiod. Moreover, the PSUs serve to further retain our executive officers, including our NEOs, as they have a time-based vesting component in addition to the stock price target, which requires the applicable executive officer to remain employed for one year following the achievement of the underlying shares vesting onapplicable stock price target in order to fully receive the first anniversarynumber of the vesting commencement date of March 1, 2019, and thereafter 1/6th of the remaining underlying shares vest on a semi-annual basis (every six months) soPSUs allotted for that all of the underlying shares will be vested on the date four years after the vesting commencement date, so long as Dr. Cathers remains an employee or other service provider (including a consultant) of the Company on such vesting dates. tranche. In connection with his appointment as our Chief Medical Officer in October 2019,addition, the Compensation Committee additionally granted Dr. Lehrer-Graiwer an optiondetermined to purchase 5,000 shares of our common stock and time-based RSUs that may vest and be settled for 1,500 shares of our common stock. The option vests, and becomes exercisable, over a four-year period, with 1/16thtie achievement of the underlying shares vesting onPSUs to our stock price instead of market capitalization (in contrast to the 2017 PSUs) as it viewed increase in stock price as a quarterly basis (every three months) after the vesting commencement date of October 1, 2019, so that allbetter gauge of the underlyingreturn to stockholders than market capitalization, which may simply reflect an increase in the number of shares will be vested on the date four years after the vesting commencement date, so longoutstanding rather than an indicator of an increase in equity value. The PSUs granted to our NEOs are as Dr. Lehrer-Graiwer remains an employee or other service provider (including a consultant) of the Company on such vesting dates. The time-based RSUs vest over a four-year period, with 1/8th of the underlying shares vesting on a semi-annual basis (every six months) after the vesting commencement date of October 1, 2019, so that all of the underlying shares will be vested on the date four years after the vesting commencement date, so long as Dr. Lehrer-Graiwer remains an employee or other service provider (including a consultant) of the Company on such vesting dates. As noted above, Dr. Lehrer-Graiwer resigned from the company effective April 17, 2020, and did not remain in our service (including as a consultant) past that date.follows: | | | | | | | | NEO | | Performance-Based RSUs (Number of Shares) | | | Dr. Love | | | | 141,400 | | | | Mr. Farrow | | | | 42,400 | | | | Mr. Johnson | | | | 42,400 | | | | Ms. Choi | | | | 42,400 | | | | Ms. Suvari | | | | 42,400 | |
The equity awards granted to our NEOs in 20192020 are set forth in the “Summary Compensation Table” and the “Grants of Plan-Based Awards for Fiscal Year 2019”2020” table below. | 422021 Proxy Statementï Global Blood Therapeutics, Inc.
Other Employee Benefits Other Employee Benefits
|
Health and Welfare Benefits Our executive officers, including our NEOs, are eligible to participate in the same employee benefit plans that are generally available to all of our employees, subject to the satisfaction of certain eligibility requirements, such as medical, dental, and life and disability insurance plans. We pay, on behalf of our employees, all or a portion of the premiums for health, life and disability insurance. 2015 Employee Stock Purchase Plan Our executive officers, including our NEOs, are eligible to participate in our ESPP on the same basis as our other full-time employees. The ESPP permits eligible employees to set aside a portion of their compensation during offering periods that are generally two years long, with purchase periods generally every six months during each offering period, and use such contributions to purchase shares of our common stock at a purchase price equal to 85% of the lower of the fair market value of the shares on the first business day of the offering period or the last business day of the purchase period. 401(k) Savings Plan Our U.S. executive officers, including our NEOs, are eligible to participate in atax-qualified retirement plan, or 401(k) Plan, on the same basis as our other employees. The 401(k) Plan provides eligible U.S. employees with an opportunity to save for retirement on a tax advantaged basis. Eligible employees are able to defer eligible compensation subject to applicable annual limits of the Internal Revenue Code of 1986, as amended, or the Code. Employees’pre-tax contributions are allocated to each participant’s individual account and are then invested in selected investment alternatives according to the participants’ directions. Employees are immediately and fully vested in their contributions. Our 401(k) Plan is intended to be qualified under Section 401(a) of the Code with our 401(k) Plan’s related trust intended to be tax exempt under Section 501(a) of the Code. As atax-qualified retirement plan, contributions to our 401(k) Plan and earnings and matching amounts on those contributions are not taxable to the employees until distributed from our 401(k) Plan. Since December 2015, the Compensation CommitteeCompany has approved various matching contributions under the 401(k) Plan. Under our current matching policy, approved by the Compensation Committee, effective January 1, 2018, we match in cash 100% of an employee’s 401(k) contributions, subject to an annual cap of $5,000$6,000 per employee. Perquisites Perquisites or other personal benefits are not a significant component of our executive compensation program. Accordingly, we do not provide significant perquisites or other personal benefits to our executive officers, including our NEOs, except for limited travel stipends or limited housing and travel reimbursements for one NEO and certain other executive officers for recruitment and retention purposes. During 2019, none ofNEOs. Employment Arrangements with our NEOs received perquisites or other personal benefits that were, in the aggregate, $10,000 or more for each individual, except Dr. Cathers, for whom we provided a travel allowance of $10,000 per month to cover travel between his residence and our corporate offices in South San Francisco. | Employment Arrangements with our NEOs
|
Post-Employment Compensation We consider it essential to the best interests of our stockholders to foster the continuous employment of our key management personnel. Accordingly, we believe that reasonable and competitive post-employment compensation arrangements are an important part of an executive compensation program to attract and retain highly-qualified senior executives. While the Compensation Committee does not consider the specific amounts payable under these post-employment compensation arrangements when determining the annual compensation of our NEOs, we believe that providing our executivesexecutive officers with post-employment payments and benefits if they lose their position in connection with a change in control are in the best interests of our stockholders because the possibility of a change in control and the related uncertainty may lead to the departure or distraction of senior executives to the detriment of our company and stockholders. In July 2015, our Board of Directors adopted a change in control policy, which has been subsequently amended and restated (the “Amended and Restated Severance and Change in Control Policy”). The Amended and Restated Severance and Change in Control Policy provides our NEOs with certain payments and benefits upon a qualified termination event outside of a Change in Control Period (as defined below). Based on its review of our pre-amended and restated change in control policy compared to the post-employment compensation arrangements of the companies in our compensation peer group, the Compensation Committee determined that these changes were necessary to recruit and retain top talent and to align with market norms. The Amended and Restated Severance and Change in Control Policy applies to our executive officers, including our NEOs, to reinforce and encouragereinforces and encourages the continued attention and dedication of certain key senior executives in the event of a change in control by providing these executivesexecutive officers with certain cash payments, equity acceleration and other benefits upon a qualifying termination of employment event in connection with a change in control. Additionally, prior Pursuant to the January 2020 amendment of our change in control policy (which amendment is in the form of our current Amended and Restated Severance and Change in Control Policy, as described below), our CEO was entitled to certain post-employment compensation upon a qualifying termination event independent of any change in control pursuant to the terms of his employment offer letter with us, as further described below under “Employment Offer Letters—CEO.” Pursuant to our change in control policy, in the event the employment of any of our NEOs is terminated by us or our acquirer or successor without Cause or an NEO resigns for Good Reason (as such
Global Blood Therapeutics, Inc. ï2021 Proxy Statement43
terms are defined in our change in control policy), in either case, within one year after the consummation of a Sale Event (as defined in the 2015 Plan) (suchone-year period, the “Change in Control Period”), he or she will be entitled to receive the following payments and benefits, or CIC Benefits, subject to his or her execution andnon-revocation of a severance agreement within 60 days following the date of such termination, including a general release of claims:claims in our favor: a lump sum cash payment equal to 12 months (or 18 months in the case of our CEO) of the NEO’s then-current“base salary” (i.e., the greater of the NEO’s base salary;salary in effect immediately prior to the termination or the base salary in effect immediately prior to the Sale Event, as applicable); a lump sum cash payment equal to 100% of the NEO’s annual “target incentive bonus” (i.e., the greater of the NEO’s target annual bonus in effect immediately prior to the termination or the target bonus in effect immediately prior to the Sale Event, as applicable) for the year in which the closing of the Sale Event occurred, which, under our Amended and Restated Severance and Change in Control Policy, was increased from 100% to 150% in the case of our CEO; a lump sum cash payment equal to the prorated annual cash bonus payout of the NEO for the portion of the year in which the closing of the Sale Event occurred, based on the NEO’s target annual cash bonus target and the date of termination of his or her employment or other service relationship with the company;us; if the NEO elects to continue his or her group healthcare benefits, a cash payment of an amount equal to the monthly employer contribution we would have made to provide the NEO with health insurance if he or she had remained employed by us until the earlier of (i) 12 months (or 18 months in the case of our CEO) following the date of termination, or (ii) the end of the NEO’s COBRA health continuation period; and | he or she had remained employed by us until the earlier of (i) 12 months (or 18 months in the case of our CEO) following the date of termination, or (ii) the end of the NEO’s COBRA health continuation period; and
|
full acceleration of vesting of all outstanding equity awards under the 2015 Plan, the 2017 Inducement Plan and such additional equity incentive plans and arrangements as may be applicable from time to time, including all performance-based equity awards, which will accelerate and vest based on the deemed achievement of 100% of the target performance levels as of the date of the NEO’s termination. As described above, the CIC Benefits are “double trigger” because the change in control alone does not trigger such payments and benefits. Rather, the CIC Benefits are triggered only if there is a qualifying termination of an NEO’s employment within the Change in Control Period. In the case of the acceleration of vesting of outstanding equity awards, we use this double-trigger arrangement to protect against the loss of retention value following a change in control of the Company and to avoid windfalls, both of which could occur if vesting of either equity or cash-based awards accelerated automatically as a result of the transaction. In addition, upon a Sale Event, to the extent Section 280G of the Internal Revenue Code, of 1986, as amended, or Section 280G, is applicable, each NEO who is then employed with us will be entitled to receive the better treatment of: (i) payment of the full amounts set forth above to which the NEO is entitled or (ii) payment of such lesser amount that does not trigger excise taxes under Section 280G. None of our NEOs are entitled to excise tax payments (or“gross-ups” “gross-ups”) relating to a change in control of the Company. The payments and benefits provided under our change in control policy are designed to be competitive in the market. Accordingly, in January 2020, following its review of our then current change in control policy compared to the post-employment compensation arrangements of the companies in our compensation peer group, the Compensation Committee determined that the policy was in need of enhancement to recruit and retain top talent and to align with market norms. As a result of this review, the Compensation Committee approved our Amended and Restated Severance and Change in Control Policy which,are designed to be competitive in addition to the above described benefits, provides our NEOs with certain payments and benefits upon a qualified termination event outside of the Change in Control Period.market. Pursuant to the Amended and Restated Severance and Change in Control Policy, in the event the employment of any of our NEOs is terminated by us or our acquirer or successor without Cause or an NEO resigns for Good Reason outside of the Change in Control Period, he or she will be entitled to receive the following payments and benefits, subject to his or her execution andnon-revocation of a severance agreement within 60 days following the date of such termination, including a general release of claims:claims in our favor: a lump sum cash payment equal to 12 months of the NEO’s then-current base salary; in the case of our CEO only, a lump sum cash payment equal to (i) 100% of the CEO’s target annualincentive bonus for the year in which the termination of his employment or other service relationship with the company occurred, plus (ii) a prorated annual cash bonus payout for the portion of the year in which the termination of his employment or other service relationship with the company occurred, based on the CEO’s annual cashtarget bonus target and the date of termination of his employment or other service relationship with the company; and if the NEO elects to continue his or her group healthcare benefits, a cash payment of an amount equal to the monthly employer contribution we would have made to provide the NEO with health insurance if he or she had remained employed by us until the earlier of (i) 12 months following the date of termination, or (ii) the end of the NEO’s COBRA health continuation period. For an estimate of the potential payments and benefits that our NEOs would have been eligible to receive under the Amended and Restated Severance and Change in Control Policy if a hypothetical change in control or other trigger event had occurred on December 31, 2019,2020, see “Potential Payments on Termination or Change in Control” below. 442021 Proxy Statementï Global Blood Therapeutics, Inc.
Employment Offer Letters
NEOs
We have entered into a written employment offer letter with each of our NEOs. These offer letters set forth the basic termsOther Compensation Policies and conditions of employment, including initial base salary, eligibility to participate in the Cash Incentive Plan, initial equity awards, eligibility to participate in our standard employee benefits plans and theat-willPractices employment relationship. These offer letters also require that each NEO execute our standard employee confidentiality and assignment agreement. Prior to January 2020, our NEOs were also eligible to receive certain severance and/orchange-in-control payments and benefits in accordance with our change in control policy (and pursuant to his offer letter, for our CEO) as described in “Post-Employment Compensation” above. As of January 2020, our NEOs are eligible to receive certain severance andchange-in-control payments and benefits in accordance with the Amended and Restated Severance and Change in Control Policy as described in “Post-Employment Compensation” above.
CEO
Prior to the adoption of our Amended and Restated Severance and Change in Control Policy, Dr. Love’s written employment offer letter provided for certain termination payments and benefits not in connection with a change in control. In particular, Dr. Love’s employment offer letter provided that if Dr. Love’s employment were terminated by us without Cause (as defined in the letter) not in connection with a change in control, he would be entitled to receive the following payments and benefits, subject to his execution andnon-revocation of a severance agreement, including a general release of claims, his resignation from all positions with us and compliance with our instructions regarding Company property:
continuation of his then-current base salary for nine months after his termination date; and
continuation of group health plan benefits to the extent authorized by and consistent with COBRA, with the cost of the regular premium for such benefits shared in the same relative proportion by us and Dr. Love as in effect on his termination date until the earlier of (i) the date that is nine months after his termination date, and (ii) the date Dr. Love becomes eligible for health benefits through another employer or otherwise becomes ineligible for COBRA.
These payments and benefits were designed to be competitive in the market. As noted above under “Post-Employment Compensation,” following its review of the post-employment compensation arrangements of the companies in our peer compensation group, the Compensation Committee adopted the Amended and Restated Severance and Change in Control Policy in January 2020. The post-employment payments and benefits to which Dr. Love is entitled under such recently adopted policy both during and outside of a Change in Control Period supersede and replace those under Dr. Love’s employment offer letter.
| Other Compensation Policies and Practices
|
Equity Award Grant Policy We have adopted an Amended and Restated Equity Award Grant Policy in January 2020, as amended in November 2020, that sets forth the process and timing for us to follow when we grant equity awards for shares of our common stock to our employees, including our executive officers, or advisors or consultants to us pursuant to any of our equity compensation plans. Pursuant to the policy, all grants of equity awards must be approved in advance by our Board of Directors, the Compensation Committee or, subject to the delegation requirements in the policy, our CEO or a committee comprised of the CEO and at least one other executive officer of the Company, or Equity Grant Committee. The Equity Grant Committee is currently comprised of our CEO and another executive (which in practice has been our Chief Human Resources Officer.Officer). The equity award granting authority delegated to the Equity Grant Committee applies tonon-executive employees and covers awards of stock options and RSUs within specific ranges set forth in the policy, which will be updated annually by the Compensation Committee. The Amended and Restated Equity Award Grant Policy sets forth that equity awards are generally granted on the following regularly scheduled basis: Equity awards granted in connection with the hiring of a new employee or the engagement of a new consultant are effective on the first trading day of the month following the later of the date on which such individual’s employment or consulting term begins or the date on which such award is approved by the Board, the Compensation Committee or the Equity Grant Committee. Equity awards granted in connection with the promotion of an existing employee are effective on the first trading day of the month following the later of the date on which such individual’s promotion occurs or the date on which such award is approved by the Board of Directors, the Compensation Committee or the Equity Grant Committee; provided, that in the case of any promotion effective on the first trading day of a particular month, the award will be effective on the effective date of such promotion so long as the Board of Directors, the Compensation Committee or the Equity Grant Committee approves the award on or before such date. Equity awards granted to existing employees (other than in connection with a promotion) will generally be granted, if at all, on an annual basis effective on the first trading day of the month following the later of the date on which we complete the focal review process with respect to such individual or the date on which such award is approved by the Board of Directors, the Compensation Committee or the Equity Grant Committee. Our Board of Directors and the Compensation Committee retain the discretion to grant equity awards at other times to the extent appropriate in light of the circumstances of such awards. In addition, the policy sets forth the manner in which our equity awards will be priced. If equity awards arean award of restricted stock or restricted stock units is denominated in dollars, the number of shares subject to the award will be determined by dividing the dollar value by the average closing market price on the NASDAQ Global Market of a share of our common stock over the trailing 20-trading day period ending on the effective date oftrading day immediately preceding the grant or if no closing price is reported for such date, the closing price on the last date preceding such date for which a closing price is reported.date. The exercise price of all stock options will be at least equal to the closing market price on the NASDAQ Global Market of a share of our common stock on the effective date of grant or if no closing price is reported for such date, the closing price on the last date preceding such date for which a closing price is reported. If the amount of a stock option award is to be determined by reference to a fair value calculated under Financial Accounting Standards Board Accounting Standard Codification Topic 718,Stock Compensation (formerly FASB Statement No. 123R), or FASB ASC Topic 718, then the number of shares to be subject to such stock option will be determined based on such fair value, and the exercise price determined in accordance with the preceding sentence and the approved valuation assumptions, subject to any other limits on the number of shares that may be subject to such stock option. Policy Prohibiting Hedging and Pledging Our Insider Trading Policy prohibits our executive officers, thenon-employee members of our Board of Directors and certain other employees from engaging in the following transactions: selling any of our securities that they do not own at the time of the sale (a(referred to as a “short sale”); buying or selling puts, calls, other derivative securities of the Company or any derivative securities that provide the economic equivalent of ownership of any of our securities or an opportunity, direct or indirect, to profit from any change in the value of our securities or engaging in any other hedging transaction with respect to our securities at any time without the prior approval of the Audit Committee of our Board of Directors, or Audit Committee; using our securities as collateral in a margin account; and pledging our securities as collateral for a loan (or modifying an existing pledge) unless the pledge has been approved by the Audit Committee. Global Blood Therapeutics, Inc. ï2021 Proxy Statement45
As of the date of this Proxy Statement, none of our NEOs or non-employee directors had previously sought or obtained approval from the Audit Committee to engage in any hedging or pledging transaction involving our securities. Stock Ownership Policy In March 2020, we adopted a stock ownership policy for our senior executive officers (i.e., our CEO and each member of our senior management team, which includes our NEOs), which requires (i) our CEO to acquire and hold a number of shares of our common stock equal in value to at least six times his or her annual base salary and (ii) each senior management team member to acquire and hold a number of shares of our common stock equal in value to at least two times his or her applicable annual base salary, in each case until such executive’s service as our CEO or senior management team member, respectively, ceases. We only count directly and beneficially owned shares, including shares purchased through our ESPP or 401(k) plan, and 50% of shares underlying vested and unexercisedin-the-money stock options. Each executive has until the later of April 1, 2025, the Initial Determination Date, or the April 1st in the year that is the fifth anniversary of his or her initial appointment in the capacity of an executive to attain the required ownership level. Once an executive satisfies his or her stock ownership requirement, the executive must continue to satisfy such stock ownership requirement as assessed on each Determination Date. If an executive fails to satisfy such stock ownership requirement as of any Determination Date (including the Initial Determination Date, as applicable)Date), then such executive shall be required to come into compliance with his or her applicable stock ownership requirement within two years following the Determination Date on which he or she failed to satisfy such stock ownership requirement. As of April 1, 2021, all of our NEOs met their respective stock ownership requirement: | | | | | | | | | | | | | | Name | | Ownership
Requirement | | Actual
Ownership | | | | Dr. Love | | | | 6.0x | | | 67.7x | | | | | Mr. Farrow | | | | 2.0x | | | 8.2x | | | | | Mr. Johnson | | | | 2.0x | | | 2.4x | | | | | Ms. Choi | | | | 2.0x | | | 24.4x | | | | | Ms. Suvari | | | | 2.0x | | | 6.4x | |
Clawback Policy In March 2021, we adopted a policy for recoupment of executive incentive compensation, or Clawback Policy, which provides that if our financial statements are materially restated, whether in part or in their entirety, due to misconduct by one or more covered officer (meaning any Section 16 officer(s)), then our Board of Directors or Compensation Committee will have the discretion to recoup a portion of any performance-based compensation that has been paid or distributed to the covered officer during the clawback period (i.e., the three-year period preceding the publication of the restated financials), to the extent such compensation paid or distributed was in excess of what would have been paid under the restated financials. Our Board of Directors or the Compensation Committee, in its sole discretion, may also reduce the amount of future compensation, including, without limitation, any bonus or severance, or the future grant or vesting of any equity award, payable to any covered officer by an amount equal to such excess proceeds from performance-based compensation received by the covered officer during the clawback period. The Clawback Policy is applicable to all cash and equity-based compensation predicated on the achievement of financial performance goals or financial metrics (excluding any such compensation based on TSR or similar stock price-based metrics). Tax and Accounting Considerations Deductibility of Executive Compensation Generally, Section 162(m) of the Code, or Section 162(m), disallows a federal income tax deduction for public corporations of remuneration in excess of $1 million paid in any fiscal year to certain specified former or current executive officers. For taxable years beginning before January 1, 2018, (i) these executive officers consisted of a public corporation’s chief executive officer and up to three other executive officers (other Other than the chief financial officer) whose compensation is required to be disclosed to stockholders under the Securities Exchange Act of 1934 because they are our most highly-compensated executive officers and (ii) qualifying “performance-based compensation” was not subject to this deduction limit if specified requirements are met. Pursuant to the Tax Cuts and Jobs Act of 2017, which was signed into law on December 22, 2017, or Tax Act, for taxable years beginning after December 31, 2017, the remuneration of a public corporation’s chief financial officer is also subject to the deduction limit. In addition, subject to certain transition rules (which apply to remuneration provided pursuant to written binding contracts which were in effect on November 2, 2017 and(and which are not subsequently modified in any material respect), for taxable years beginning after December 31, 2017, the exemption from the deduction limit for “performance-based compensation” is no longer available. Consequently, for fiscal years beginning after December 31, 2017, all remuneration in excess of $1 million paid to a specified executive will not be deductible.
In designing our executive compensation program and determining the compensation of our executive officers, including our NEOs, the Compensation Committee considers a variety of factors, including the potential impact of the Section 162(m) deduction limit. However, the Compensation Committee will not necessarily limit executive compensation to that which is or may be deductible under Section 162(m). The Compensation Committee will consider various alternatives to preserving the deductibility of compensation payments and benefits to the extent consistent with its compensation goals.
To maintain flexibility to compensate our executive officers in a manner designed to promote our short- and long-term corporate goals, the Compensation Committee has not adopted a policy that all compensation must be deductible. The Compensation Committee believes that our stockholders’ interests are best served if its discretion and flexibility in awarding compensation is not restricted in order to allow such compensation to be consistent with the goals of our executive compensation program, even though some compensation awards may result innon-deductible compensation expense. Consequently, the Compensation Committee will not necessarily limit executive compensation to that which is or may be deductible under Section 162(m). 462021 Proxy Statementï Global Blood Therapeutics, Inc.
Accounting for Stock-Based Compensation We follow FASB ASC Topic 718 for our stock-based compensation awards. FASB ASC Topic 718 requires us to measure the compensation expense for all share-based payment awards made to our employees andnon-employee members of our Board of Directors, including stock options to purchase shares of our common stock and other stock awards, based on the grant date “fair value” of these awards. This calculation is performed for accounting purposes and reported in the executive compensation tables required by the federal securities laws, even though the recipient of the awards may never realize any value from their awards. Taxation of “Parachute” Payments Sections 280G and 4999 of the Code provide that executive officers and directors who hold significant equity interests and certain other service providers may be subject to significant additional taxes if they receive payments or benefits in connection with a change in control of the company that exceeds certain prescribed limits, and that the company (or a successor) may forfeit a deduction on the amounts subject to this additional tax. We have not agreed to provide any executive officer, including any NEO, with a“gross-up” “gross-up” or other reimbursement payment for any tax liability that the executive officer might owe as a result of the application of Sections 280G or 4999 of the Code. Section 409A of the Internal Revenue Code Section 409A of the Code imposes additional significant taxes in the event that an executive officer, director or service provider receives “deferred compensation” that does not satisfy the requirements of Section 409A of the Code. Although we do not maintain a traditional nonqualified deferred compensation plan, Section 409A of the Code does apply to certain severance arrangements, bonus arrangements and equity awards. We structure all our severance arrangements, bonus arrangements and equity awards in a manner to either avoid the application of Section 409A or, to the extent doing so is not possible, to comply with the applicable requirements of Section 409A of the Code. Compensation Risk Assessment We structure our pay to consist of both fixed and variable compensation to motivate our employees, including our NEOs, to produce superior short- and long-term results that are in the best interests of our company and stockholders to attain our ultimate objective of increasing stockholder value. In addition, we have established, and the Compensation Committee endorses, several controls to address and mitigate compensation-related risk, such as maintaining an anti-hedging and anti-pledging policy and stock ownership guidelines for our executive officers (including our NEOs) and directors. The Compensation Committee, in consultation with its compensation consultant, Compensia, evaluates whether our policies and practices create excessive risk in our compensation programs. In 2019,2020, this risk assessment included, among other things, a review of our cash and equity incentive-based compensation plans to ensure that they are aligned with our corporate performance goals and overall target total direct compensation to ensure an appropriate balance between fixed and variable pay components. Based on this assessment, the Compensation Committee concluded that our compensation policies and practices are not reasonably likely to have a material adverse effect on our company. The Compensation Committee intends to continue to evaluate on an annual basis the potential risks associated with our compensation policies and practices, and has engaged Compensia to conduct an updated assessment of our compensation policies and practices during 2020.2021. As a result of the approval and launch of our first commercial product, Oxbryta, in late 2019, we expect this evaluation will include the potential risks associated with field-based incentive compensation and commercial-related goals and targets. Global Blood Therapeutics, Inc. ï2021 Proxy Statement47
NEO Compensation Tables 20192020 Summary Compensation Table
The following table sets forth information regarding total compensation awarded to, earned by and paid to each of our NEOs during the fiscal years ended December 31, 2020, 2019 2018 and 2017 to the extent he was an NEO in such year.2018. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Name and Principal Position | | Year | | | Salary
($) | | | Bonus
($) | | | Stock Awards
($)(1) | | | Option
Awards
($)(2) | | | Non-Equity
Incentive
Plan
Compensation
($)(3) | | | All Other
Compensation
($) | | | Total
($) | | Ted Love, M.D. | | | 2019 | | | | 596,876 | (4) | | | — | | | | 4,359,600 | | | | 4,595,717 | | | | 540,000 | | | | 5,000 | (5) | | | 10,097,193 | | President, Chief Executive Officer and Director | | | 2018 2017 | | | | 568,750 520,000 | | | | — — | | | | 4,648,800 5,559,125 | | | | 4,623,690 1,557,373 | | | | 414,000 380,000 | | | | 5,000 2,500 | | | | 10,260,240 8,018,998 | | Jeffrey Farrow | | | 2019 | | | | 440,625 | (6) | | | — | | | | 1,211,000 | | | | 1,267,784 | | | | 244,000 | | | | 5,000 | (5) | | | 3,168,409 | | Chief Financial Officer | |
| 2018
2017 |
| |
| 425,938
412,945 |
| |
| —
— |
| |
| 1,370,800
629,030 |
| |
| 1,326,469
397,398 |
| |
| 201,000
200,000 |
| |
| 5,000
11,296 |
| |
| 3,329,207
1,650,669 |
| Brian Cathers | | | 2019 | | | | 319,445 | (7) | | | — | | | | 1,604,400 | | | | 1,571,112 | | | | 172,000 | (7) | | | 100,236 | (5) | | | 3,767,193 | | Chief Scientific Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | David Johnson(8) | | | 2019 | | | | 453,125 | (9) | | | — | | | | 1,211,000 | | | | 1,267,784 | | | | 250,000 | | | | 5,000 | (5) | | | 3,186,909 | | Chief Commercial Officer | | | 2018 | | | | 355,000 | | | | 75,000 | | | | 1,891,750 | | | | 1,898,534 | | | | 169,000 | | | | 5,000 | | | | 4,394,284 | | Joshua Lehrer-Graiwer(10) | | | 2019 | | | | 430,625 | (11) | | | — | | | | 1,208,825 | | | | 1,258,161 | | | | 253,000 | | | | 5,000 | (5) | | | 3,155,611 | | Chief Medical Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Name and Principal Position | | Year | | Salary
($) | | Bonus
($) | | Stock
Awards
($)(1) | | Option
Awards
($)(2) | | Non-equity
incentive plan
compensation
($)(3) | | All other
Compensation
($) | | Total ($) | | | | | | | | | | Ted Love, M.D. President, Chief Executive Officer and Director | | | | 2020 | | | | | 661,250 | (4) | | | | — | | | | | 11,778,840 | (5) | | | | 4,693,666 | | | | | 467,000 | | | | | 5,000 | (6) | | | | 17,605,756 | | | | | 2019 | | | | | 596,876 | | | | | — | | | | | 4,359,600 | | | | | 4,595,717 | | | | | 540,000 | | | | | 5,000 | | | | | 10,097,193 | | | | | 2018 | | | | | 568,750 | | | | | — | | | | | 4,648,800 | | | | | 4,623,690 | | | | | 414,000 | | | | | 5,000 | | | | | 10,260,240 | | | | | | | | | | | David Johnson Chief Commercial Officer | | | | 2020 | | | | | 463,750 | (7) | | | | — | | | | | 4,037,361 | (8) | | | | 1,582,504 | | | | | 234,000 | | | | | 5,000 | (6) | | | | 6,322,615 | | | | | 2019 | | | | | 453,125 | | | | | — | | | | | 1,211,000 | | | | | 1,267,784 | | | | | 250,000 | | | | | 5,000 | | | | | 3,186,909 | | | | | 2018 | | | | | 355,000 | | | | | 75,000 | | | | | 1,897,750 | | | | | 1,898,534 | | | | | 169,000 | | | | | 5,000 | | | | | 4,394,284 | | | | | | | | | | | Jeffrey Farrow Chief Financial Officer | | | | 2020 | | | | | 457,813 | (9) | | | | — | | | | | 3,708,261 | (10) | | | | 1,582,504 | | | | | 231,500 | | | | | 5,000 | (6) | | | | 5,985,078 | | | | | 2019 | | | | | 440,625 | | | | | — | | | | | 1,211,000 | | | | | 1,267,784 | | | | | 244,000 | | | | | 5,000 | | | | | 3,168,409 | | | | | 2018 | | | | | 425,938 | | | | | — | | | | | 1,370,800 | | | | | 1,326,469 | | | | | 201,000 | | | | | 5,000 | | | | | 3,329,207 | | | | | | | | | | | Jung Choi Chief Business and Strategy Officer | | | | 2020 | | | | | 433,125 | (11) | | | | — | | | | | 3,708,261 | (12) | | | | 1,582,504 | | | | | 219,000 | | | | | 5,000 | (6) | | | | 5,947,890 | | | | | 2019 | | | | | 416,875 | | | | | — | | | | | 1,211,000 | | | | | 1,267,784 | | | | | 231,000 | | | | | 5,000 | | | | | 3,131,659 | | | | | 2018 | | | | | 392,063 | | | | | — | | | | | 1,370,800 | | | | | 1,326,469 | | | | | 185,000 | | | | | 5,000 | | | | | 3,279,332 | | | | | | | | | | | Tricia Suvari Chief Legal Officer | | | | 2020 | | | | | 422,499 | (13) | | | | — | | | | | 3,708,261 | (14) | | | | 1,582,504 | | | | | 214,000 | | | | | 5,000 | (6) | | | | 5,932,264 | | | | | 2019 | | | | | 403,125 | | | | | — | | | | | 1,211,000 | | | | | 1,267,784 | | | | | 223,000 | | | | | 5,000 | | | | | 3,109,909 | | | | | 2018 | | | | | 386,875 | | | | | — | | | | | 1,549,600 | | | | | 1,515,964 | | | | | 183,000 | | | | | 5,000 | | | | | 3,640,439 | |
| (1) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the restricted stock and/or RSUs granted during 2017, 2018, 2019 and 2019,2020, as applicable, computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of these amounts are included in Note 10 to our financial statements included in our Annual Report on Form10-K for the fiscal year ended December 31, 2019.2020. These amounts do not reflect the actual economic value that may be realized by the NEOs upon the vesting or settlement of the restricted stock or RSUs as applicable, or the sale of the common stock underlying such awards. For performance-based RSUs, or PSUs, this table assumes the maximum achievement as of December 31, 2020, of all three escalating stock price targets under such awards over a four-year period ending June 30, 2024; based on this assumption, the value of the awards made to our NEOs at the date of grant would be as follows: Dr. Love - $7,063,496; Mr. Johnson - $2,118,050; Mr. Farrow - $2,118,050; Ms. Choi - $2,118,050; and Ms. Suvari - $2,118,050. |
| (2) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the stock option awards granted during 2017, 2018, 2019 and 2019,2020, as applicable, computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of these amounts are included in Note 10 to our financial statements included in our Annual Report on Form10-K for the fiscal year ended December 31, 2019.2020. These amounts do not reflect the actual economic value that may be realized by the NEOs upon the exercise of the stock options or the sale of the common stock underlying such stock options. |
| (3) | The amounts reported reflect the annual cash incentive compensation earned by the NEOs under our Cash Incentive Plan, based on our achievement of certain corporate performance goals and each NEO’s (except for the CEO) achievement of his or her individual performance goals. |
| (4) | From January 1, 2019,2020, Dr. Love’s annual base salary was $575,000,$600,000, which increased to $600,000,$670,000, effective February 16, 2019.2020. |
| (5) | Includes (a) $7,063,496, representing the PSUs granted to Dr. Love in June 2020, and (b) $4,715,344, representing the time-based RSUs granted to Dr. Love in January 2020. |
| (6) | The amounts reported consist of employer matching contributions under our 401(k) of $5,000 each for each NEO other than for Dr. Cathers. The amount reported for Dr. Cathers consists of employer matching contributions under our 401(k) of $236 and monthly stipends to cover his travel expenses in connection with his employment for an aggregate of $100,000. |
(6) | From January 1, 2019, Mr. Farrow’s annual base salary was $427,500, which increased to $442,500, effective February 16, 2019.NEO.
|
| (7) | Dr. Cathers commenced employment with us on February 25, 2019, and hisFrom January 1, 2020, Mr. Johnson’s annual base salary and annual cash incentive compensation amounts were prorated accordingly.was $455,000, which increased to $465,000, effective February 16, 2020.
|
| (8) | Includes (a) $2,118,050, representing the PSUs granted to Mr. Johnson commenced employment with us on March 12, 2018in June 2020, and was not an NEO for 2017.(b) $1,919,311, representing the time-based RSUs granted to Mr. Johnson in January 2020. |
| (9) | From January 1, 2019,2020, Mr. Johnson’sFarrow’s annual base salary was $440,000,$442,500, which increased to $455,000,$460,000, effective February 16, 2019.2020. |
| (10) | Dr. Lehrer-Graiwer was not an NEO for 2018 or 2017.Includes (a) $2,118,050, representing the PSUs granted to Mr. Farrow in June 2020, and (b) $1,590,211, representing the time-based RSUs granted to Mr. Farrow in January 2020.
|
| (11) | From January 1, 2019, Dr. Lehrer-Graiwer’s2020, Ms. Choi’s annual base salary was $400,000,$420,000, which increased to $435,000, effective February 16, 2020. |
| (12) | Includes (a) $2,118,050, representing the PSUs granted to Ms. Choi in June 2020, and (b) $1,590,211, representing the time-based RSUs granted to Ms. Choi in January 2020. |
| (13) | From January 1, 2020, Ms. Suvari’s annual base salary was $405,000, which increased to $425,000, effective February 16, 2019,2020. |
| (14) | Includes (a) $2,118,050, representing the PSUs granted to Ms. Suvari in June 2020, and $460,000, effective October 1, 2019.(b) $1,590,211, representing the time-based RSUs granted to Ms. Suvari in January 2020. |
482021 Proxy Statementï Global Blood Therapeutics, Inc.
Grants of Plan-Based Awards for Fiscal Year 20192020 The following table sets forth the individual awards made to each of our NEOs during 2019.2020. For a description of the types of awards indicated below, please see our “Compensation Discussion and Analysis” above. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Estimated
Future
Payouts
Under
Non-
Equity
Incentive
Plan
Awards(1) | | | All Other
Stock
Awards:
Number of
Shares of
Stock or
Units
(#)(2) | | | All Other
Option
Awards:
Number
of
Securities
Underlying
Options
(#)(3) | | | Exercise
or Base
Price of
Option
Awards
($/sh.) | | | Grant
Date Fair
Market
Value of
Awards
($)(4) | | Name | | | | Grant
Date | | | Target
($) | | Ted Love, M.D. | | Time-Based RSUs | | | 2/1/2019 | | | | — | | | | 90,000 | | | | — | | | | — | | | | 4,359,600 | | | | Time-Based Stock
Options | | | 2/1/2019 | | | | — | | | | — | | | | 145,000 | | | | 48.44 | | | | 4,595,717 | | | | Annual Bonus
Opportunity | | | | | | | 360,000 | | | | — | | | | — | | | | — | | | | — | | | | | | | | | | Jeffrey Farrow | | Time-Based RSUs | | | 2/1/2019 | | | | — | | | | 25,000 | | | | — | | | | — | | | | 1,211,000 | | | | Time-Based Stock
Options | | | 2/1/2019 | | | | — | | | | — | | | | 40,000 | | | | 48.44 | | | | 1,267,784 | | | | Annual Bonus
Opportunity | | | | | | | 177,000 | | | | — | | | | — | | | | — | | | | — | | | | | | | | | | Brian Cathers | | Time-Based RSUs | | | 3/1/2019 | | | | — | | | | 30,000 | | | | — | | | | — | | | | 1,604,400 | | | | Time-Based Stock
Options | | | 3/1/2019 | | | | — | | | | — | | | | 45,000 | | | | 53.48 | | | | 1,571,112 | | | | Annual Bonus
Opportunity | | | | | | | 150,000 | (5) | | | — | | | | — | | | | — | | | | — | | | | | | | | | | David Johnson | | Time-Based RSUs | | | 2/1/2019 | | | | — | | | | 25,000 | | | | — | | | | — | | | | 1,211,000 | | | | Time-Based Stock
Options | | | 2/1/2019 | | | | — | | | | — | | | | 40,000 | | | | 48.44 | | | | 1,267,784 | | | | Annual Bonus
Opportunity | | | | | | | 182,000 | | | | — | | | | — | | | | — | | | | — | | | | | | | | | | Joshua Lehrer-Graiwer | | Time-Based RSUs | | | 2/1/2019 | | | | — | | | | 23,500 | | | | — | | | | — | | | | 1,138,340 | | | | | | | 10/1/2019 | | | | — | | | | 1,500 | | | | — | | | | — | | | | 70,485 | | | | Time-Based Stock
Options | | | 2/1/2019 | | | | — | | | | — | | | | 35,000 | | | | 48.44 | | | | 1,109,311 | | | | | | | 10/1/2019 | | | | — | | | | — | | | | 5,000 | | | | 46.99 | | | | 148,850 | | | | Annual Bonus
Opportunity | | | | | | | 184,000 | | | | — | | | | — | | | | — | | | | — | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Name | | | | Grant
date | | Estimated
Future
Payouts
Under
Non-
Equity
Incentive
Plan
Awards(1) Target ($) | | Estimated Future
Payouts Under
Equity Incentive Pan
Awards(2) | | All
Other
Stock
Awards:
Number
of
Shares
of Stock
or Units
(#)(3) | | All Other Option Awards: Number Of
Securities Underlying Options (#)(4) | | Exercise or Base Price of Option Awards ($/sh.) | | Grant Date Fair Market Value of Awards ($)(5) | | | | | Threshold
(#) | | Maximum
(#) | | | | | | | | | | | Ted Love, M.D. | | Time-based RSUs | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | 71,640 | | | | | — | | | | | — | | | | | 4,715,345 | | | | | | | | | | | | | | Time-based Stock Options | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | — | | | | | 114,190 | | | | | 65.82 | | | | | 4,693,666 | | | | | | | | | | | | | | Performance-based RSUs | | | | 6/1/2020 | | | | | — | | | | | 28,280 | | | | | 141,400 | | | | | | | | | | — | | | | | — | | | | | 7,063,496 | | | | | | | | | | | | | | Annual Bonus Opportunity | | | | | | | | | 469,000 | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | David Johnson | | Time-based RSUs | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | 29,160 | | | | | — | | | | | — | | | | | 1,919,311 | | | | | | | | | | | | | | Time-based Stock Options | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | — | | | | | 38,500 | | | | | 65.82 | | | | | 1,582,504 | | | | | | | | | | | | | | Performance-based RSUs | | | | 6/1/2020 | | | | | — | | | | | 8,480 | | | | | 42,400 | | | | | | | | | | — | | | | | — | | | | | 2,118,050 | | | | | | | | | | | | | | Annual Bonus Opportunity | | | | | | | | | 232,500 | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | Jeffrey Farrow | | Time-based RSUs | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | 24,160 | | | | | — | | | | | — | | | | | 1,590,211 | | | | | | | | | | | | | | Time-based Stock Options | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | — | | | | | 38,500 | | | | | 65.82 | | | | | 1,582,504 | | | | | | | | | | | | | | Performance-based RSUs | | | | 6/1/2020 | | | | | — | | | | | 8,480 | | | | | 42,400 | | | | | | | | | | — | | | | | — | | | | | 2,118,050 | | | | | | | | | | | | | | Annual Bonus Opportunity | | | | | | | | | 230,000 | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | Jung Choi | | Time-based RSUs | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | 24,160 | | | | | — | | | | | — | | | | | 1,590,211 | | | | | | | | | | | | | | Time-based Stock Options | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | — | | | | | 38,500 | | | | | 65.82 | | | | | 1,582,504 | | | | | | | | | | | | | | Performance-based RSUs | | | | 6/1/2020 | | | | | — | | | | | 8,480 | | | | | 42,400 | | | | | | | | | | — | | | | | — | | | | | 2,118,050 | | | | | | | | | | | | | | Annual Bonus Opportunity | | | | | | | | | 217,500 | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | Tricia Suvari | | Time-based RSUs | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | 24,160 | | | | | — | | | | | — | | | | | 1,590,211 | | | | | | | | | | | | | | Time-based Stock Options | | | | 2/3/2020 | | | | | — | | | | | | | | | | | | | | | — | | | | | 38,500 | | | | | 65.82 | | | | | 1,582,504 | | | | | | | | | | | | | | Performance-based RSUs | | | | 6/1/2020 | | | | | — | | | | | 8,480 | | | | | 42,400 | | | | | | | | | | — | | | | | — | | | | | 2,118,050 | | | | | | | | | | | | | | | Annual Bonus Opportunity | | | | | | | | | 212,500 | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | |
| (1) | The amounts shown reflect the target cash incentive compensation for our NEOs, which are disclosed in the “2019“2020 Target Annual Bonus” section of the Compensationabove “Compensation Discussion and Analysis.” The actual amounts paid for 20192020 are disclosed in the“Non-Equity “Non-Equity Incentive Plan Compensation” column of the Summary Compensation Table. There were no threshold or maximum payout levels for the cash incentive compensation.compensation (however, the portion based on corporate performance goals had a maximum of 155% of target for 2020). |
| (2) | The amounts shown reflect the threshold and maximum payout levels associated with PSUs granted pursuant to our 2015 Plan, which amounts will be payable in shares of our common stock if the stock price targets for such PSUs are met, with each such share price based on the average closing market price on the NASDAQ Global Select Market over a 20 consecutive trading day period. The target payout for the PSUs is the maximum payout. 20% of the maximum number of PSUs will be earned if a stock price of $109.20 per share is achieved on or before June 30, 2024; an additional 35% (for a total of 55%) of the maximum number of PSUs will be earned if a stock price of $145.60 per share is achieved on or before June 30, 2024; and an additional 45% (for a total of 100%) of the maximum number of PSUs will be earned if a stock price of $182.00 per share is achieved on or before June 30, 2024. Upon the achievement of each respective stock price target, 50% of the PSUs allotted to that tranche will vest, while the remaining 50% will vest on the first anniversary of the date the stock price target was achieved, subject to the executive officer’s continued employment or other service relationship with us through such vesting date. |
| (3) | The amounts shown represent time-based RSUs granted pursuant to our 2015 Plan or 2017 Inducement Plan, which amounts will be payable in shares of our common stock if the service-based conditions for such time-based RSUs are met. Except for Dr. Cathers’ time-based RSUs, theThe time-based RSUs vest semi-annually over four years, subject to the NEO’s continuous service relationship with us through each applicable vesting date. Dr. Cathers’ time-based RSUs vest over a four-year period, with 1/4th of the underlying shares vesting on the first anniversary of the vesting commencement date of March 1, 2019, and thereafter 1/6th of the remaining underlying shares vest on a semi-annual basis (every six months) so that all of the underlying shares will be vested on the date four years after the vesting commencement date, subject to Dr. Cathers’ continuouscontinued employment or other service relationship with us through each applicable vesting date. |
(3)(4) | The amounts shown represent time-based stock options granted pursuant to our 2015 Plan or 2017 Inducement Plan. Except for Dr. Cathers’ time-based stock options, theThe time-based stock options vest quarterly over four years, subject to the NEO’s continuous service relationship with us through each applicable vesting date. Dr. Cathers’ time-based options vest, and become exercisable, over a four-year period, with 1/4th of the underlying shares vesting on the first anniversary of the vesting commencement date of February 25, 2019, and thereafter, 1/12th of the remaining underlying shares vest on a quarterly basis so that all of the underlying shares will be vested on the date four years after the vesting commencement date, subject to Dr. Cathers continuouscontinued employment or other service relationship with us through each applicable vesting date. |
(4)(5) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the equity awards granted during 2019,2020, computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of these amounts are included in Note 10 to our financial statements included in our Annual Report on Form10-K for the fiscal year ended December 31, 2019.2020. These amounts do not reflect the actual economic value that may be realized by the NEOs upon the vesting or settlement of PSUs or RSUs or the exercise of the stock options, as applicable, or the sale of the common stock underlying such awards. For PSUs, this table assumes the maximum achievement as of December 31, 2020, of all three escalating stock price targets under such awards over a four-year period ending June 30, 2024; based on this assumption, the value of the awards made to our NEOs at the date of grant would be as follows: Dr. Love - $7,063,496; Mr. Johnson - $2,118,050; Mr. Farrow - $2,118,050; Ms. Choi - $2,118,050; and Ms. Suvari - $2,118,050. |
(5) | Dr. Cathers commenced employment with us on February 25, 2019. The amount reflects Dr. Cathers’ annualized target cash incentive compensation.
Global Blood Therapeutics, Inc. ï2021 Proxy Statement49 |
20192020 Outstanding Equity Awards at Fiscal Year End
The following table sets forth certain information with respect to outstanding equity awards held by each of our NEOs as of December 31, 2019.2020. | | | | | | | | | | | | | | | | | | | | | | Option Awards(1) | | | | Stock Awards(1) | | | | Option Awards(1) | | Stock Awards(1) | | | Name | | Number of
Securities
Underlying
Unexercised
Options (#)
Exercisable | | Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable | | Equity
Incentive
Plan
Awards:
Number of
Securities
Underlying
Unexercised
Unearned
Options
(#) | | Option
Exercise
Price
($) | | Option
Expiration
Date | | Number
of
shares
or units
of stock
that
have
not
vested
(#)(1) | | Market
value of
shares or
units of
stock that
have
not
vested
($)(2) | | Equity
Incentive
Plan
Awards:
Number of
unearned
shares, units
or
other rights
that have
not vested
(#) | | Equity
Incentive
Plan
Awards:
Market
value or
payout
value of
unearned
shares, units
or other
rights
that have
not
vested
($)(2) | | | Number of Securities Underlying Unexercised Options (#) Exercisable | | Number of Securities Underlying Unexercised Options (#) Unexercisable | | Equity
Incentive
Plan
Awards: Number of Securities Underlying Unexercised Unearned Options (#) | | Option Exercise Price ($) | | | | Option Expiration Date | | Number of shares or units of
stock that have not vested (#)(1) | | Market value of shares or units of stock that have not vested ($)(2) | | Equity Incentive Plan Awards: Number of unearned shares, units or other
rights that have not vested (#) | | Equity Incentive Plan Awards: Market value or payout value of unearned shares, units or other rights that have not vested ($)(2) | | | (a) | | (b) | | (c) | | (d) | | (e) | | (f) | | (g) | | (h) | | (i) | | (j) | | | (b) | | (c) | | (d) | | (e) | | | | (f) | | (g) | | (h) | | (i) | | (j) | | Ted Love, M.D. | | | — | | | | — | | | | — | | | | — | | | | — | | | 36,188 | (3) | | 2,876,584 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 12,063 | (3) | | | 522,449 | | | | — | | | | — | | | | | | | 123,743 | (4) | | | 9,063 | (4) | | | — | | | | 16.40 | | | | | | 1/17/2027 | | | | — | | | | — | | | | — | | | | — | | | | | | | 83,875 | (5) | | | 38,125 | (5) | | | — | | | | 59.60 | | | | | | 2/1/2028 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 29,250 | (6) | | | 1,266,818 | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 56,250 | (7) | | | 2,436,188 | | | | — | | | | — | | | | | | | 63,437 | (8) | | | 81,563 | (8) | | | — | | | | 48.44 | | | | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 62,685 | (9) | | | 2,714,887 | | | | — | | | | — | | | | | | | 21,410 | (10) | | | 92,780 | (10) | | | — | | | | 65.82 | | | | | | 2/3/2030 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | 141,400 | (11) | | | 6,124,034 | | | David Johnson | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 13,125 | (12) | | | 568,444 | | | | — | | | | — | | | | | | | 37,812 | (13) | | | 17,188 | (13) | | | — | | | | 54.05 | | | | | | 3/12/2028 | | | | — | | | | — | | | | — | | | | — | | | | | 87,493 | (4) | | 45,313 | (4) | | | — | | | 16.40 | | | 1/17/2027 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 15,625 | (7) | | | 676,719 | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 48,750 | (5) | | 3,875,138 | | | | — | | | | — | | | | 17,500 | (8) | | | 22,500 | (8) | | | — | | | | 48.44 | | | | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | | 53,375 | (6) | | 68,625 | (6) | | | — | | | 59.60 | | | 2/1/2028 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 25,515 | (9) | | | 1,105,055 | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 78,750 | (7) | | 6,259,838 | | | | — | | | | — | | | | 7,218 | (10) | | | 31,282 | (10) | | | — | | | | 65.82 | | | | | | 2/3/2030 | | | | — | | | | — | | | | — | | | | — | | | | | 27,187 | (8) | | 117,813 | (8) | | | — | | | 48.44 | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | 42,400 | (11) | | | 1,836,344 | | | Jeffrey Farrow | | 105,000 | (9) | | 15,000 | (9) | | | — | | | 14.96 | | | 2/24/2026 | | | | — | | | | — | | | | — | | | | — | | | | 120,000 | (14) | | | — | | | | — | | | | 14.96 | | | | | | 2/24/2026 | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 9,188 | (3) | | 730,354 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 3,063 | (3) | | | 132,659 | | | | — | | | | — | | | | | 22,389 | (4) | | 11,563 | (4) | | | — | | | 16.40 | | | 1/17/2027 | | | | — | | | | — | | | | — | | | | — | | | | 31,639 | (4) | | | 2,313 | (4) | | | — | | | | 16.40 | | | | | | 1/17/2027 | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 14,375 | (5) | | 1,142,669 | | | | — | | | | — | | | | 24,062 | (5) | | | 10,938 | (5) | | | — | | | | 59.60 | | | | | | 2/1/2028 | | | | — | | | | — | | | | — | | | | — | | | | | 15,312 | (6) | | 19,688 | (6) | | | — | | | 59.60 | | | 2/1/2028 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 8,625 | (6) | | | 373,549 | | | | — | | | | — | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 21,875 | (7) | | 1,738,844 | | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 15,625 | (7) | | | 676,719 | | | | — | | | | — | | | | | 7,500 | (8) | | 32,500 | (8) | | | — | | | 48.44 | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | 17,500 | (8) | | | 22,500 | (8) | | | — | | | | 48.44 | | | | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | Brian Cathers | | | — | | | | — | | | | — | | | | — | | | | — | | | 30,000 | (10) | | 2,384,700 | | | | — | | | | — | | | | | | — | (11) | | 45,000 | (11) | | | — | | | 53.48 | | | 3/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | David Johnson | | | — | | | | — | | | | — | | | | — | | | | — | | | 21,875 | (12) | | 1,738,844 | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | | | — | | | | 21,140 | (9) | | | 915,573 | | | | — | | | | — | | | | 24,062 | (13) | | 30,938 | (13) | | | — | | | 54.05 | | | 3/12/2028 | | | | — | | | | — | | | | — | | | | — | | | | | | | 7,218 | (10) | | | 31,282 | (10) | | | — | | | | 65.82 | | | | | | 2/3/2030 | | | | — | | | | — | | | | — | | | | — | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 21,875 | (7) | | 1,738,844 | | | | — | | | | — | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | — | | | | | — | | | | | — | | | | | 42,400 | (11) | | | | 1,836,344 | | | | 7,500 | (8) | | 32,500 | (8) | | | — | | | 48.44 | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | Joshua Lehrer-Graiwer | | — | (14) | | 625 | (14) | | | — | | | 12.95 | | | 2/9/2026 | | | | — | | | | — | | | | — | | | | — | | | | | | — | (15) | | 2,813 | (15) | | | — | | | 19.89 | | | 9/8/2026 | | | | — | | | | — | | | | — | | | | — | | | | | | | | | | | | | | | | 5,250 | (16) | | 417,323 | | | | | | | | | | — | (17) | | 6,563 | (17) | | | | 26.90 | | | 2/17/2027 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 4,150 | (18) | | 329,884 | | | | — | | | | — | | | | | | — | (19) | | 5,469 | (19) | | | | 27.90 | | | 7/26/2027 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 10,625 | (5) | | 844,581 | | | | — | | | | — | | | | | | — | (6) | | 14,907 | (6) | | | — | | | 59.60 | | | 2/1/2028 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 20,563 | (7) | | 1,634,553 | | | | — | | | | — | | | | | | — | (8) | | 28,438 | (8) | | | — | | | 48.44 | | | 2/1/2029 | | | | — | | | | — | | | | — | | | | — | | | | | | | — | | | | — | | | | — | | | | — | | | | — | | | 1,500 | (20) | | 119,235 | | | | — | | | | — | | | | | | — | (21) | | 5,000 | (21) | | | | 46.99 | | | 10/1/2029 | | | | — | | | | — | | | | — | | | | — | | |
502021 Proxy Statementï Global Blood Therapeutics, Inc.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Option Awards(1) | | | | Stock Awards(1) | | | | | | | | | | | | Name | | Number of Securities Underlying Unexercised Options (#) Exercisable | | Number of Securities Underlying Unexercised Options (#) Unexercisable | | Equity
Incentive
Plan
Awards: Number of Securities Underlying Unexercised Unearned Options (#) | | Option Exercise Price ($) | | | | Option Expiration Date | | Number of shares or units of
stock that have not vested (#)(1) | | Market value of shares or units of stock that have not vested ($)(2) | | Equity Incentive Plan Awards: Number of unearned shares, units or other
rights that have not vested (#) | | Equity Incentive Plan Awards: Market value or payout value of unearned shares, units or other rights that have not vested ($)(2) | | | | | | | | | | | | (a) | | (b) | | (c) | | (d) | | (e) | | | | (f) | | (g) | | (h) | | (i) | | (j) | | | | | | | | | | | | Jung Choi | | | | 74,964 | (14) | | | | — | | | | | — | | | | | 3.40 | | | | | | | | | | 4/9/2025 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 5,350 | (14) | | | | — | | | | | — | | | | | 3.40 | | | | | | | | | | 4/9/2025 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 4,063 | (3) | | | | 175,969 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 45,937 | (4) | | | | 3,063 | (4) | | | | — | | | | | 16.40 | | | | | | | | | | 1/17/2027 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 24,062 | (5) | | | | 10,938 | (5) | | | | — | | | | | 59.60 | | | | | | | | | | 2/1/2028 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 8,625 | (6) | | | | 373,549 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 15,625 | (7) | | | | 676,719 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 17,500 | (8) | | | | 22,500 | (8) | | | | — | | | | | 48.44 | | | | | | | | | | 2/1/2029 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 21,140 | (9) | | | | 915,573 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 7,218 | (10) | | | | 31,282 | (10) | | | | — | | | | | 65.82 | | | | | | | | | | 2/3/2030 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | — | | | | | — | | | | | 42,400 | (11) | | | | 1,836,344 | | | | | | | | | | | | | Tricia Suvari | | | | 100,000 | (14) | | | | — | | | | | — | | | | | 18.25 | | | | | | | | | | 10/11/2026 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 27,500 | (5) | | | | 12,500 | (5) | | | | — | | | | | 59.60 | | | | | | | | | | 2/1/2028 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 9,750 | (6) | | | | 422,273 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 15,625 | (7) | | | | 676,719 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 17,500 | (8) | | | | 22,500 | (8) | | | | — | | | | | 48.44 | | | | | | | | | | 2/1/2029 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | 21,140 | (9) | | | | 915,573 | | | | | — | | | | | — | | | | | | | | | | | | | | | | | 7,218 | (10) | | | | 31,282 | (10) | | | | — | | | | | 65.82 | | | | | | | | | | 2/3/2030 | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | | | | | | | | — | | | | | — | | | | | — | | | | | — | | | | | | | | | | — | | | | | — | | | | | — | | | | | 42,400 | (11) | | | | 1,836,344 | |
| (1) | All of the outstanding equity awards held by our NEOs will become fully vested and exercisable ornon-forfeitable, as applicable, if the NEO is terminated without Cause or resigns for Good Reason, in either case, within 12 months following a Sale Event. The vesting acceleration of the outstanding equity awards held by our NEOs is described in greater detail in “Employment Arrangements with Our Named Executive Officers—Change in Control Policy”. |
| (2) | Computed in accordance with SEC rules as the number of unvested shares or units multiplied by the closing market price of a share of our common stock on December 31, 2019,2020, which was $79.49.$43.31. The actual value (if any) to be realized by the NEO depends on whether the shares or units vest and the future performance of our common stock. |
| (3) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of January 17, 2017, such that all of the RSUs will bewere fully vested on January 17, 2021, providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
| (4) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of January 17, 2017, such that all of the shares will bewere fully vested on January 17, 2021, providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
| (5) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date February 1, 2018, such that all of the RSUs will be fully vested on February 1, 2022, provided the NEO remains in continuous service through each such vesting date.
|
(6) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of February 1, 2018, such that all of the shares will be fully vested on February 1, 2022 providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each applicable vesting date. |
| (6) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of February 1, 2018, such that all of the RSUs will be fully vested on February 1, 2022, subject to the NEO’s continued employment or other service relationship with us through each applicable vesting date. |
| (7) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of February 1, 2019, such that all of the RSUs will be fully vested on February 1, 2023, providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
Global Blood Therapeutics, Inc. ï2021 Proxy Statement51
| (8) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of February 1, 2019, such that all of the shares will be fully vested on February 1, 2023, providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
| (9) | 25% of the shares subject to the stock option vested on April 4, 2017, and the remaining shares vest quarterly over the following three years thereafter, such that all of the shares will be fully vested on April 4, 2020, subject to the NEO’s continuous service through each such vesting date.
|
(10) | 25% of the RSUs vested on March 1, 2020, and 1/8th8th of the RSUs vest on a semi-annual basis from suchthe vesting commencement date of February 3, 2020, such that all of the RSUs will be fully vested on March 1, 2023, providedFebruary 3, 2024, subject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date.
|
(11)(10) | 25%1/16th of the shares subject to the stock option vestedvest on the first anniversary ofa quarterly basis from the vesting commencement date of February 25, 2019, and the remaining shares vest quarterly over the following three years thereafter,3, 2020, such that all of the shares will be fully vested on February 25, 2023,3, 2024, subject to the NEO’s continuouscontinued employment or other service relationship with us through each applicable vesting date.
|
| (11) | Represents the maximum number of PSUs that may be earned. The vesting of the PSUs is contingent upon the achievement of three escalating stock price targets, with each such share price based on the average closing market price on the NASDAQ Global Select Market over a 20 consecutive trading day period. 20% of the maximum number of PSUs will be earned if a stock price of $109.20 per share is achieved on or before June 30, 2024; an additional 35% (for a total of 55%) of the maximum number of PSUs will be earned if a stock price of $145.60 per share is achieved on or before June 30, 2024; and an additional 45% (for a total of 100%) of the maximum number of PSUs will be earned if a stock price of $182.00 per share is achieved on or before June 30, 2024. Upon the achievement of the respective stock price targets, 50% of the PSUs allotted to that tranche will vest, while the remaining 50% will vest on the first anniversary of the date the stock price target was achieved, subject to the NEO’s continued employment or other service relationship with us through such vesting date. |
| (12) | 25% of the RSUs vested on April 1, 2019 and 1/8th8th of the RSUs vest on a semi-annual basis from such date, such that all of the RSUs will be fully vested on April 1, 2022, providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
| (13) | 25% of the shares subject to the stock option vested on March 12, 2019 and 1/16th16th of the shares subject to the stock option vest on a quarterly basis from such date, such that all of the shares will be fully vested on March 12, 2022 providedsubject to the NEO remains in continuousNEO’s continued employment or other service relationship with us through each suchapplicable vesting date. |
| (14) | 1/16th of the sharesShares subject to the stock option vest on a quarterly basis from the vesting commencement date of February 9, 2016, such that all of the shares vested on February 9, 2020.have fully vested.
|
(15) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of September 8, 2016, such that all of the shares will be fully vested on September 8, 2020, provided the NEO remains in continuous service through each such vesting date.
|
(16) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of March 1, 2017, such that all of the RSUs will be fully vested on March 1, 2021, provided the NEO remains in continuous service through each such vesting date.
|
(17) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of February 17, 2017, such that all of the shares will be fully vested on February 17, 2021, provided the NEO remains in continuous service through each such vesting date.
|
(18) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of August 1, 2017, such that all of the RSUs will be fully vested on August 1, 2021, provided the NEO remains in continuous service through each such vesting date.
|
(19) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of July 26, 2017, such that all of the shares will be fully vested on July 26, 2021, provided the NEO remains in continuous service through each such vesting date.
|
(20) | 1/8th of the RSUs vest on a semi-annual basis from the vesting commencement date of October 1, 2019, such that all of the RSUs will be fully vested on October 1, 2023, provided the NEO remains in continuous service through each such vesting date.
|
(21) | 1/16th of the shares subject to the stock option vest on a quarterly basis from the vesting commencement date of October 1, 2019, such that all of the shares will be fully vested on October 1, 2023, provided the NEO remains in continuous service through each such vesting date.
|
Option Exercises and Stock Vested in Fiscal Year 20192020 The following table sets forth the number of shares acquired and the value realized upon exercises of stock options and vesting of RSUs during the fiscal year ended December 31, 20192020, by each of our NEOs. | | | | | | | | | | | | | | Option Awards | | | | Stock Awards | | | | Option Awards | | | Stock Awards | | | | | Name | | Number of Shares
Acquired on
Exercise
(#) | | | Value Realized
on Exercise
($)(1) | | | Number of Shares
Acquired on
Vesting
(#) | | | Value Realized on
Vesting
($)(2) | | | | | Number of
Shares
Acquired on
Exercise (#) | | Value Realized
on Exercise ($)(1) | | | | Number of
Shares
Acquired on
Vesting (#) | | Value
Realized on
Vesting ($)(2) | | Ted Love, M.D. | | | — | | | | — | | | | 202,553 | | | | 12,889,776 | | | | | — | | — | | | | | 75,080 | | | | 4,992,999 | | | David Johnson | | | | | — | | — | | | | | 18,645 | | | | 1,127,546 | | | Jeffrey Farrow | | | — | | | | — | | | | 22,000 | | | | 1,280,670 | | | | | — | | — | | | | | 21,145 | | | | 1,406,745 | | | Brian Cathers | | | — | | | | — | | | | — | | | | — | | | Jung Choi | | | | | — | | — | | | | | 23,145 | | | | 1,539,485 | | | David Johnson | | | — | | | | — | | | | 16,250 | | | | 837,988 | | | | Joshua Lehrer-Graiwer | | | 50,354 | | | | 1,204,657 | | | | 18,362 | | | | 1,059,283 | | | Tricia Suvari | | | | | — | | — | | | | | 15,770 | | | | 1,050,007 | |
| (1) | The value realized upon the exercise of stock options is calculated by (a) subtracting the stock option exercise price from the market price on the date of exercise to get the realized value per share, and (b) multiplying the realized value per share by the number of shares underlying the stock options exercised. |
| (2) | The value realized upon vesting of restricted stock and RSUs is calculated by multiplying the number of shares of restricted stock and RSUs vested by the market price on the vest date. |
522021 Proxy Statementï Global Blood Therapeutics, Inc.
Potential Payments on Termination or Change in Control Our Amended and Restated Severance and Change in Control Policy provides for certain payments and benefits to each of our NEOs if the NEO is terminated by us or our acquirer or successor without Cause or resigns for Good Reason (as such terms are defined in such policy), both during and outside of the Change in Control Period (assuming that all outstanding equity awards were assumed, continued or substituted by an acquirer or successor in such change in control), subject to the NEO’s execution andnon-revocation of a severance agreement, including a general release of claims. The table below quantifies the potential payments and benefits that would have become due to our NEOs assuming that Amended and Restated Severance and Change in Control Policy had been in effect as of December 31, 20192020, and that one of the triggering events above occurred as of December 31, 2019.2020. The market price of a share of our common stock on December 31, 20192020, was $79.49. Prior to the adoption of the Amended and Restated Severance and Change in Control Policy in January 2020, none of our NEOs except for our CEO was entitled to termination benefits outside of the Change in Control Period.$43.31. | | Name | | Qualifying
Termination
Not in
Connection
with a Change
in Control
($) | | Qualifying
Termination
in
Connection
with a Change
in Control
($) | | | Qualifying
termination
Not in
connection
with a Change
in Control ($) | | Qualifying
Termination in
Connection
with a Change
in Control ($) | | Ted Love, M.D. | | | | | | | | | | Cash Severance Payment | | | 600,000 | (1) | | | 900,000 | (2) | | | 670,000 | (1) | | | 1,005,000 | (2) | | | | | Cash Incentive Bonus Payment | | | 360,000 | (3) | | | 540,000 | (4) | | | 469,000 | (3) | | | 703,500 | (4) | | | | | COBRA Premiums | | | 16,154 | (5) | | | 24,231 | (6) | | | 21,103 | (5) | | | 31,655 | (6) | | | | | Accelerated Equity Vesting (Time-Based) | | | — | | | | 17,235,308 | (7) | | | — | | | | 7,184,226 | (7) | | | | | Accelerated Equity Vesting (Performance-Based) | | | | — | | | | — | | | David Johnson | | | | | | | Cash Severance Payment | | | | 465,000 | (1) | | | 465,000 | (1) | | | | | Cash Incentive Bonus Payment | | | | — | | | | 232,500 | (3) | | | | | COBRA Premiums | | | | 21,783 | (5) | | | 21,783 | (5) | | | | | Accelerated Equity Vesting (Time-Based) | | | | — | | | | 2,350,217 | (7) | | | | | Accelerated Equity Vesting (Performance-Based) | | | | — | | | | — | | | Jeffrey Farrow | | | | | | | | | | Cash Severance Payment | | | 442,500 | (1) | | | 442,500 | (1) | | | 460,000 | (1) | | | 460,000 | (1) | | | | | Cash Incentive Bonus Payment | | | — | | | | 177,000 | (3) | | | — | | | | 230,000 | (3) | | | | | COBRA Premiums | | | 11,285 | (5) | | | 11,285 | (5) | | | 21,103 | (5) | | | 21,103 | (5) | | | | | Accelerated Equity Vesting (Time-Based) | | | — | | | | 6,710,046 | (7) | | | — | | | | 2,160,742 | (7) | Brian Cathers | | | | | | | | | | Accelerated Equity Vesting (Performance-Based) | | | | — | | | | — | | | Jung Choi | | | | | | | Cash Severance Payment | | | 375,000 | (1) | | | 375,000 | (1) | | | 435,000 | (1) | | | 435,000 | (1) | | | | | Cash Incentive Bonus Payment | | | — | | | | 150,000 | (3) | | | — | | | | 217,500 | (3) | | | | | COBRA Premiums | | | 11,719 | (5) | | | 11,719 | (5) | | | — | (5) | | | — | (5) | | | | | Accelerated Equity Vesting (Time-Based) | | | — | | | | 3,555,150 | (7) | | | — | | | | 2,224,235 | (7) | David Johnson | | | | | | | | | | Accelerated Equity Vesting (Performance-Based) | | | | — | | | | — | | | Tricia Suvari | | | | | | | Cash Severance Payment | | | 455,000 | (1) | | | 455,000 | (1) | | | 425,000 | (1) | | | 425,000 | (1) | | | | | Cash Incentive Bonus Payment | | | — | | | | 182,000 | (3) | | | — | | | | 212,500 | (3) | | | | | COBRA Premiums | | | 11,719 | (5) | | | 11,719 | (5) | | | 17,740 | (5) | | | 17,740 | (5) | | | | | Accelerated Equity Vesting (Time-Based) | | | — | | | | 5,273,875 | (7) | | | — | | | | 2,014,565 | (7) | | | | | Accelerated Equity Vesting (Performance-Based) | | | | — | | | | — | |
| (1) | Represents 12 months of the NEO’s base salary. |
| (2) | Represents 18 months of Dr. Love’s base salary. |
| (3) | Represents 100% of the NEO’s target annual bonus opportunity. The table does not include the prorated annual bonus opportunity that the NEO would be entitled to since, on December 31, 2019,2020, the full annual bonus, which is shown in the “Summary Compensation Table,” was earned. |
| (4) | Represents 150% of Dr. Love’s target annual bonus opportunity. The table does not include the prorated annual bonus opportunity that Dr. Love would be entitled to since, on December 31, 2019,2020, the full annual bonus, which is shown in the “Summary Compensation Table,” was earned. |
Global Blood Therapeutics, Inc. ï2021 Proxy Statement53
| (5) | Represents 12 months of our contribution towards health insurance, based on our actual costs to provide health insurance to the NEO as of the date of termination. Since Ms. Choi did not participate in the Company’s health insurance plan, she will receive zero months of payment of COBRA premiums. |
| (6) | Represents 18 months of our contribution towards health insurance, based on our actual costs to provide health insurance to the NEODr. Love as of the date of termination. |
| (7) | Represents the value of acceleration of vesting of 100% of the NEO’s unvested and outstanding equity awards, other than PSUs, based on the market price of a share of our common stock on December 31, 2019,2020, which was $79.49.$43.31. For the PSUs, represents the value of acceleration of vesting of 0% of the NEO’s unvested and outstanding PSUs, based on an assumed sale price of a share of our common stock in the change in control of $43.31. For stock options with a per share exercise price greater than $79.49,$43.31, no amount was included with respect to such stock options. |
In February 2020, Dr. Lehrer-Graiwer resigned from his employment with us, effective April 17, 2020. In connection with Dr. Lehrer-Graiwer’s resignation, he did not receive any severance benefits or payments or acceleration of vesting of equity awards.
Securities Authorized for Issuance under Equity Compensation Plans Equity Compensation Plans The following table sets forth information as of December 31, 2019,2020, regarding shares of common stock that may be issued under our equity compensation plans, consisting of our 2012 Stock Option and Grant Plan (or 2012 Plan), our 2015 Plan, our 2017 Inducement Plan and our ESPP. | | Plan Category | | Number of Securities
to be Issued upon Exercise
of Outstanding Options,
Warrants and
Rights (#)(a) | | Weighted Average Exercise
Price of Outstanding
Options, Warrants and
Rights (b)(1) | | Number of Securities
Remaining Available for
Future Issuance under
Equity Compensation Plans
(Excluding Securities
Reflected in Column (a))(c) | | | Number of
Securities to be
Issued upon
Exercise of
Outstanding
Options,
Warrants and
Rights (#) (a) | | Weighted
Average Exercise
Price of
Outstanding
Options,
Warrants and
Rights (b)(1) | | Number of
Securities
Remaining
Available for
Future Issuance
under Equity
Compensation
Plans (Excluding
Securities
Reflected in
Column (a)) (c) | | Equity compensation plans approved by security holders(2) | | | 3,963,354 | (3) | | $ | 33.72 | (3) | | | 3,893,761 | (4) | | Equity compensation plans approved by security holders(2) | | | | 4,408,439 | (3) | | | $40.38 | (3) | | | 4,995,682 | (4) | | | | Equity compensation plans not approved by security holders(5) | | | 1,459,278 | | | $ | 48.18 | | | | 837,550 | | | Equity compensation plans not approved by security holders(5) | | | | 1,543,947 | | | | $50.44 | | | | 1,277,475 | | | Total | | | 5,422,632 | | | $ | 36.24 | | | | 4,731,311 | | | | 5,952,386 | | | | $42.07 | | | | 6,233,157 | |
| (1) | The weighted average exercise price is calculated based solely on outstanding stock options. |
| (2) | Includes the 2012 Plan, the 2015 Plan and the ESPP. Our 2015 Plan provides that the number of shares reserved and available for issuance under the plan will automatically increase each January 1, beginning on January 1, 2016 until January 1, 2025, by 4% of the outstanding number of shares of common stock on the immediately preceding December 31 or such lesser number of shares as determined by the compensation committee of our Board of Directors. Our ESPP provides that the number of shares reserved and available for issuance will automatically increase each January 1, from January 1, 2016 until January 1, 2025, by the lesser of (i) 3,000,000 shares of common stock, (ii) 1% of the outstanding number of shares of common stock on the immediately preceding December 31 or (iii) such lesser number of shares as determined by the compensation committee of our Board of Directors. On January 1, 2020, the number of shares available for issuance under our 2015 Plan and our ESPP increased by 2,425,694 shares and 100,000 shares, respectively, pursuant to these provisions. These increases are not reflected in the table above. We no longer grant new awards under the 2012 Plan, and any awards previously granted under such plan prior to our initial public offering that are forfeited, canceled, reacquired by us prior to vesting satisfied without the issuance of stock or otherwise terminated (other than by exercise) are added to shares available for issuance under the 2015 Plan. |
| (3) | Does not include purchase rights accruing under the ESPP as of December 31, 2020, because the purchase rightrights (and, therefore, the number of shares to be purchased) willwere not be determined until the end of the purchase period.periods on January 31, 2021, and February 28, 2021. |
| (4) | As of December 31, 2019,2020, there were 3,641,1064,741,092 shares of common stock available for issuance under the 2015 Plan and 252,655254,590 shares of common stock available for issuance under the ESPP (which number includes shares subject to purchase during the current purchase period,periods which commenced on February 1, 2020,2021 and March 1, 2021, since the exact number of which will not be known until the end of the purchase periodperiods on JulyAugust 31, 2020. Subject to the number of shares remaining in the share reserve, the maximum number of shares purchasable by any participant on any one purchase date for any purchase period, including the current period, may not exceed 2,500 shares).2021. |
| (5) | Consists solely of the 2017 Inducement Plan. |
542021 Proxy Statementï Global Blood Therapeutics, Inc.
| CEO PAY RATIOCEO Pay Ratio
|
As required by Section 953(b) of the Dodd-Frank Act, and Item 402(u) of RegulationS-K, we are disclosing the ratio of the annual total compensation of our CEO to the median of the annual total compensation of all our median employee.employees (other than our CEO). For 2019:2020: the annual total compensation of our CEO was $10,097,193$17,605,756 (as disclosed in the Summary Compensation Table above); the median of the annual total compensation of all our median employeeemployees was $321,142;$373,366; and the ratio of the annual total compensation of our CEO to the annual total compensation of our median employee was 3147 to 1. In 2020, there was no change in our employee population or employee compensation arrangements that we reasonably believe would result in a significant change to our pay ratio disclosure. However, in 2020 there was a change in the circumstances of the employee identified as the median employee for 2019 as that individual was promoted and received a special promotional award. Accordingly, it is no longer appropriate to use that median employee because we reasonably believe that the change in such employee’s circumstances would result in a significant change in our pay ratio disclosure. As permitted by the SEC rules, we have elected to use another employee, whose 2019 compensation was substantially similar to the original median employee’s 2019 compensation based on the same consistently applied compensation measure used to select the original median employee, as our median employee for 2020. After identifying our median employee and ensuring this employee did not have anomalous compensation in 2020, we then calculated the annual total compensation of our median employee using the same methodology we used for our CEO, and our other NEOs, in the 2020 Summary Compensation Table above. In addition, in 2020, our CEO was granted an award of performance-based RSUs with a grant date fair value of $7,063,496 to be earned and vest contingent upon the achievement of three escalating stock price targets over a four-year period ending June 30, 2024. This one-time award that the Compensation Committee granted to our CEO (as well as other members of our senior management) as a special motivational and retention vehicle had the effect of increasing our CEO’s annual total compensation by nearly 75% year-over-year (based on the calculations and assumptions described above), resulting in a significantly higher pay ratio than in 2019. In 2019, we identified our median employee as of December 31, 2019, by: (i) calculating for each full-time employee, except our CEO, and part-time employee on that date (a) actual annual base salary in 2019 (annualized for new hires who were not employed for the entire year and for permanent employees on an unpaid leave of absence for a portion of the year), (b) actual annual bonus earned in 2019 (annualized for new hires who were not employed for the entire year and for permanent employees on an unpaid leave of absence for a portion of the year), and (c) the grant date fair value of all equity awards granted in 2019; and (ii) ranking this compensation from lowest to highest to identify our median employee. For purposes of this disclosure, earnings of any employee outside the U.S. were converted to U.S. dollars using the monthly average Swiss franc to U.S. dollar exchange rate for each month in 2019, based on exchange rates used by us for various purposes. We used this compensation as our consistently applied compensation measure because we believe it was representative of our employee compensation. After identifying our median employee and ensuring this employee did not have anomalous compensation in 2019, we then calculated the annual total compensation of our median employee using the same methodology we used for our CEO, and our other NEOs, in the Summary Compensation Table above. The pay ratio above is a reasonable estimate calculated in a manner consistent with the SEC rules. The SEC rules provide companies with significant flexibility in identifying the median employee and calculating the pay ratio, including flexibility to adopt a variety of methodologies, to apply certain exclusions and to make reasonable estimates and assumptions that reflect their employee populations and compensation practices. Accordingly, the pay ratio above may not be comparable with the pay ratios of other companies, even companies within our industry. Global Blood Therapeutics, Inc. ï2021 Proxy Statement55
COMPENSATION COMMITTEE REPORT
The following Compensation Committee Report is not considered proxy solicitation material and is not deemed filed with the Securities and Exchange Commission. Notwithstanding anything to the contrary set forth in any of our filings made under the Securities Act of 1933 or the Exchange Act that might incorporate our filings under those statutes, the Compensation Committee Report shall not be incorporated by reference into any of our prior filings or into any of our future filings under those statutes. The Compensation Committee of the Board of Directors has reviewed and discussed the Compensation Discussion and Analysis required by Item 402(b) of RegulationS-K with the Company’s management. Based on this review and discussion, the Compensation Committee recommended to the Board of Directors, and the Board of Directors approved, that the Compensation Discussion and Analysis be included in this Proxy Statement for the Annual Meeting and incorporated by reference in the Company’s Annual Report on Form10-K for the fiscal year ended December 31, 2019.2020. COMPENSATION COMMITTEE WENDY L. YARNO, CHAIRPERSONCHAIR SCOTT W. MORRISON MARK L. PERRY 562021 Proxy Statementï Global Blood Therapeutics, Inc.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS Other than the compensation agreements and other arrangements with our directors and NEOs described above, and the transactions described below, since January 1, 2019,2020, there has not been and there is not currently proposed, any transaction or series of similar transactions to which we were, or will be, a party in which the amount involved exceeded, or will exceed, $120,000 and in which any director, executive officer, holder of five percent or more of any class of our capital stock or any member of the immediate family of, or entities affiliated with, any of the foregoing persons, had, or will have, a direct or indirect material interest, except as described in the next paragraph.interest. Dr. Homcy resigned from our Board of Directors, effective June 18, 2019. Effective June 19, 2019, we entered into a consulting agreement with Dr. Homcy, pursuant to which Dr. Homcy serves as a strategic consultant to the Company and as an observer on the Research and Development Committee of our Board of Directors. As consideration for such consulting services, Dr. Homcy is entitled to receive a cash retainer of $2,500 per month and at the start of the consultancy received (i) aone-time,up-front payment of $7,500 and (ii) an award of RSUs for 4,000 shares of the Company’s common stock, which were granted on June 19, 2019. The fair market value for such RSUs as of the date of grant was $234,200. The RSUs will vest on the first anniversary of the grant date, subject to Dr. Homcy’s continued service under the consulting agreement.
Indemnification Agreements We have entered into agreements to indemnify our directors and executive officers. These agreements will, among other things, require us to indemnify these individuals for certain expenses (including attorneys’ fees), judgments, fines and settlement amounts reasonably incurred by such person in any action or proceeding, including any action by or in our right, on account of any services undertaken by such person on behalf of our company or that person’s status as a member of our board of directors to the maximum extent allowed under Delaware law. Procedures for Approval of Related Person Transactions The Audit Committee conducts an appropriate review of all related party transactions for potential conflict of interest situations on an ongoing basis, and the approval of the Audit Committee is required for all such transactions. The Audit Committee follows the policies and procedures set forth in our Related Person Transaction Policy in order to facilitate such review. The Related Person Transaction Policy is written. Global Blood Therapeutics, Inc. ï2021 Proxy Statement57
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS The following table sets forth the beneficial ownership information of our common stock by: each person known to us to be the beneficial owner of more than 5% of our common stock as of March 31, 2020;2021; each current director; and all current executive officers and directors as a group. We have based our calculation of the percentage of beneficial ownership of 60,886,05862,240,050 shares of common stock outstanding on March 31, 2020.2021. Each individual or entity shown in the table has furnished information with respect to beneficial ownership. The information with respect to each individual or entity is as of March 31, 2020,2021, unless otherwise noted. The information with respect to certain significant stockholders is based on filings by the beneficial owners with the SEC pursuant to Sections 13(d) and 13(g) of the Exchange Act. We have determined beneficial ownership in accordance with the SEC’s rules. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities. In addition, the rules include shares of common stock issuable pursuant to the exercise of stock options that are either immediately exercisable or exercisable on or before May 30, 2020,2021, which is 60 days after March 31, 2020.2021. These shares are deemed to be outstanding and beneficially owned by the person holding those stock options for the purpose of computing the percentage ownership of that person, but they are not treated as outstanding for the purpose of computing the percentage ownership of any other person. Unless otherwise indicated, the persons or entities identified in this table have sole voting and investment power with respect to all shares shown as beneficially owned by them, subject to applicable community property laws. | | | | | | | | | Beneficial Owner(1) | | Number of Shares
Beneficially Owned | | | Percentage of Shares
Beneficially Owned | | 5% or Greater Stockholders: | | | | | | | | | Entities affiliated with Fidelity(2) 245 Summer Street, Boston, MA 02210 | | | 6,651,295 | | | | 10.9 | % | Entities affiliated with Perceptive(3) 51 Astor Place, 10th Floor, New York, NY 10003 | | | 5,865,726 | | | | 9.6 | % | Entities affiliated with T. Rowe Price Associates, Inc.(4) 100 E. Pratt Street, Baltimore, MD 21202 | | | 5,656,185 | | | | 9.3 | % | Entities affiliated with The Vanguard Group(5) 100 Vanguard Blvd. Malvern, PA 19355 | | | 5,471,462 | | | | 9.0 | % | Entities affiliated with BlackRock(6) 55 East 52nd Street, New York, NY 10055 | | | 4,855,173 | | | | 8.0 | % | Entities affiliated with Bank of America Corporation(7) 100 N Tryon Street, Charlotte, NC 28255 | | | 4,698,143 | | | | 7.7 | % |
| | | | | | | | | | | | | | Beneficial Owner(1) | | Number of Shares
Beneficially Owned | | Percentage of Shares
Beneficially Owned | | | | 5% or Greater Stockholders: | | | | | | | | | | | | | | Entities affiliated with Janus Henderson Group plc(2) 201 Bishopsgate EC2M 3AE, United Kingdom | | | | 5,966,862 | | | | | 9.6 | % | | | | Entities affiliated with Bank of America Corporation(3) 100 N Tryon Street Charlotte, NC 28255 | | | | 5,883,578 | | | | | 9.5 | % | | | | Entities affiliated with Perceptive(4) 51 Astor Place, 10th Floor New York, NY 10003 | | | | 5,674,711 | | | | | 9.1 | % | | | | Entities affiliated with The Vanguard Group(5) 100 Vanguard Blvd. Malvern, PA 19355 | | | | 5,460,482 | | | | | 8.8 | % | | | | Entities affiliated with Fidelity(6) 245 Summer Street Boston, MA 02210 | | | | 4,407,065 | | | | | 7.1 | % | | | | Entities affiliated with T. Rowe Price Associates, Inc.(7) 100 E. Pratt Street Baltimore, MD 21202 | | | | 4,063,123 | | | | | 6.5 | % | | | | Entities affiliated with BlackRock(8) 55 East 52nd Street New York, NY 10055 | | | | 3,683,260 | | | | | 5.9 | % |
582021 Proxy Statementï Global Blood Therapeutics, Inc.
| | | | | | | | | Beneficial Owner(1) | | Number of Shares
Beneficially Owned | | | Percentage of Shares
Beneficially Owned | | Named Executive Officers and Directors: | | | | | | | | | Ted W. Love, M.D.(8) | | | 1,309,141 | | | | 2.1 | % | Jeffrey Farrow(9) | | | 215,071 | * | | | * | | Brian Cathers(10) | | | 21,647 | * | | | * | | David Johnson(11) | | | 55,591 | * | | | * | | Joshua Lehrer-Graiwer(12) | | | 3,220 | * | | | * | | Willie L. Brown, Jr.(13) | | | 106,903 | * | | | * | | Scott W. Morrison(14) | | | 81,875 | * | | | * | | Mark L. Perry(15) | | | 101,903 | * | | | * | | Glenn F. Pierce, M.D., Ph.D.(16) | | | 81,875 | * | | | * | | Philip A. Pizzo, M.D.(17) | | | 51,875 | * | | | * | | Dawn Svoronos(18) | | | 21,041 | * | | | * | | Wendy Yarno(19) | | | 46,041 | * | | | * | | All directors and executive officers as a group (16 persons)(20) | | | 2,615,993 | | | | 4.2 | % |
| | | | | | | | | | | | | | Beneficial Owner(1) | | Number of Shares
Beneficially Owned | | Percentage of Shares
Beneficially Owned | | | | Named Executive Officers and Directors: | | | | | | | | | | | | | | Ted W. Love, M.D.(9) | | | | 1,434,488 | | | | | 2.3 | % | | | | Jeffrey Farrow(10) | | | | 232,322 | * | | | | * | | | | | Jung E. Choi(11) | | | | 394,372 | * | | | | * | | | | | David L. Johnson(12) | | | | 103,188 | * | | | | * | | | | | Tricia Suvari, Esq.(13) | | | | 186,596 | * | | | | * | | | | | Willie L. Brown, Jr.(14) | | | | 117,137 | * | | | | * | | | | | Scott W. Morrison(15) | | | | 92,109 | * | | | | * | | | | | Deval L. Patrick(16) | | | | 7,834 | * | | | | * | | | | | Mark L. Perry(17) | | | | 58,537 | * | | | | * | | | | | Glenn F. Pierce, M.D., Ph.D.(18) | | | | 92,109 | * | | | | * | | | | | Philip A. Pizzo, M.D.(19) | | | | 62,109 | * | | | | * | | | | | Dawn A. Svoronos(20) | | | | 38,731 | * | | | | * | | | | | Alexis A. Thompson, M.D., M.P.H.(21) | | | | 622 | * | | | | * | | | | | Wendy L. Yarno(22) | | | | 62,109 | * | | | | * | | | | | All directors and executive officers as a group (16 persons)(23) | | | | 2,964,928 | | | | | 4.7 | % |
| * | Represents beneficial ownership of less than 1% of the shares of common stock. |
| (1) | Unless otherwise indicated, the address for each beneficial owner is c/o Global Blood Therapeutics, Inc., 181 Oyster Point Boulevard, South San Francisco, CA 94080. |
| (2) | Based solely on a report on Schedule 13G filed with the SEC on February 12, 2021, which indicates that Janus Henderson Group plc (“Janus Henderson”) had shared voting power and shared dispositive power over 5,966,862 shares of common stock. Janus Henderson has an indirect 97% ownership stake in Intech Investment Management LLC (“Intech”) and a 100% ownership stake in Janus Capital Management LLC (“JCM”), Perkins Investment Management LLC, Henderson Global Investors Limited and Janus Henderson Investors Australia Institutional Funds Management Limited (each an “Asset Manager”). Each Asset Manager is an investment adviser registered or authorized in its relevant jurisdiction and each furnishing investment advice to various fund, individual and/or institutional clients (collectively referred to herein as “Managed Portfolios”). As a result of its role as investment adviser or sub-adviser to the Managed Portfolios, JCM may be deemed to be the beneficial owner of 5,897,668 shares of common stock, and as a result of its role as investment adviser or sub-adviser to the Managed Portfolios, Intech may be deemed to be the beneficial owner of 69,194 shares of common stock. |
| (3) | Based solely on a report on Schedule 13G/A filed with the SEC on February 8, 2021, which indicates that Bank of America Corporation had shared voting power with respect to 5,778,958 shares of common stock and had shared dispositive power over 5,883,578 shares of common stock. |
| (4) | Based solely on a report on Schedule 13G/A filed with the SEC on February 16, 2021, which indicates that Perceptive Advisors LLC and Joseph Edelman had shared voting power and shared dispositive power over 5,674,711 shares of common stock, all of which are held by Perceptive Life Sciences Master Fund Ltd. (the “Fund”). Perceptive Advisors LLC serves as the investment manager to the Fund. Joseph Edelman is the managing member of Perceptive Advisors LLC. |
| (5) | Based solely on a report on Schedule 13G/A filed with the SEC on February 10, 2020,2021, which indicates that The Vanguard Group had (i) shared voting power with respect to 46,133 shares of common stock, (ii) sole dispositive power over 5,368,569 shares of common stock and (iii) shared dispositive power over 91,913 shares of common stock. |
| (6) | Based solely on a report on Schedule 13G/A filed with the SEC on February 8, 2021, which indicates that FMR LLC had sole voting power with respect to 1,549,966463,185 shares of common stock and had sole dispositive power over 6,651,2954,407,065 shares of common stock. Abigail P. Johnson is a Director, the Chairman, and the Chief Executive Officer of FMR LLC. Members of the Johnson family, including Abigail P. Johnson, are the predominant owners, directly or through trusts, of Series B voting common shares of FMR LLC, representing 49% of the voting power of FMR LLC. The Johnson family group and all other Series B shareholders have entered into a shareholders’ voting agreement under which all Series B voting common shares will be voted in accordance with the majority vote of Series B voting common shares. Accordingly, through their ownership of voting common shares and the execution of the shareholders’ voting agreement, members of the Johnson family may be deemed, under the Investment Company Act of 1940, to form a controlling group with respect to FMR LLC. Neither FMR LLC nor Abigail P. Johnson has the sole power to vote or direct the voting of the shares owned directly by the various investment companies registered under the Investment Company Act (“Fidelity Funds”) advised by Fidelity Management & Research Company (“FMR Co”), a wholly owned subsidiary of FMR LLC, which power resides with the Fidelity Funds’ Boards of Trustees. Fidelity Management & Research Company carries out the voting of the shares under written guidelines established by the Fidelity Funds’ Boards of Trustees. |
(3)(7) | Based solely on a report on Schedule 13G/A filed with the SEC on February 14, 2020,16, 2021, which indicates that Perceptive Advisors LLC and Joseph EdelmanT. Rowe Price Associates, Inc. had sharedsole voting power and dispositive power over 5,865,726with respect to 837,735 shares of common stock alland had sole dispositive power over 4,063,123 shares of which are held by Perceptive Life Sciences Master Fund Ltd (the “Fund”). Perceptive Advisors LLC serves as the investment manager to the Fund. Joseph Edelman is the managing member of Perceptive Advisors LLC.common stock. |
(4)(8) | Based solely on a report on Schedule 13G/A filed with the SEC on February 14, 2020, which indicates that T. Rowe Price Associates, Inc. had sole voting power with respect to 897,781 shares of common stock and had sole dispositive power over 5,656,185 shares of common stock. |
(5) | Based solely on a report on Schedule 13G/A filed with the SEC on February 12, 2020, which indicates that The Vanguard Group had (i) sole voting power with respect to 122,827 shares of common stock, (ii) shared voting power with respect to 7,775 shares of common stock, (iii) sole dispositive power over 5,348,702 shares of common stock and (iv) shared dispositive power over 122,760 shares of common stock.
|
(6) | Based solely on a report on Schedule 13G/A filed with the SEC on February 5, 2020,January 29, 2021, which indicates that BlackRock, Inc. had sole voting power with respect to 4,708,1513,470,400 shares of common stock and had sole dispositive power over 4,855,1733,683,260 shares of common stock.
|
Global Blood Therapeutics, Inc. ï2021 Proxy Statement59
(7) | Based solely on a report on Schedule 13G filed with the SEC on February 14, 2020, which indicates that Bank of America Corporation had shared voting power with respect to 4,697,667 shares of common stock and had shared dispositive power over 4,698,143 shares of common stock.
|
(8)(9) | Includes (i) 1,077,450774,311 shares of common stock held by Dr. Love; (ii) 5,000 shares of common stock held by Dr. Love’s two daughters, as to which Dr. Love disclaims beneficial ownership; (iii) 306,000 shares of common stock held by three trusts for the benefit of Dr. Love’s daughters; and (iii) 226,691(iv) 349,177 shares of common stock that Dr. Love has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(9)(10) | Includes (i) 33,46415,402 shares of common stock held by Mr. Farrow; and (ii) 181,607216,920 shares of common stock that Mr. Farrow has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(10)(11) | Includes (i) 5,179177,090 shares of common stock held by Mr. Cathers;Ms. Choi; (ii) 25,000 shares of common stock held in The 2005 William Park and (ii) 16,468Jung Choi Family Trust; and (iii) 192,282 shares of common stock that Mr. CathersMs. Choi has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(11)(12) | Includes (i)13,185 27,407 shares of common stock held by Mr. Johnson; and (ii) 42,40675,781 shares of common stock that Mr. Johnson has the right to acquire from us within 60 days of March 31, 2020 pursuant to the exercise of stock options. |
(12) | Includes (i) 2,595 shares of common stock held by Mr. Lehrer-Graiwer and (ii) 625 shares of common stock that Mr. Lehrer-Graiwer has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options.
|
| (13) | Includes (i) 31,42819,565 shares of common stock held by Ms. Suvari; and (ii) 167,031 shares of common stock that Ms. Suvari has the right to acquire from us within 60 days of March 31, 2021 pursuant to the exercise of stock options. |
| (14) | Includes (i) 36,228 shares of common stock held by Mr. Brown; and (ii) 75,47580,909 shares of common stock that Mr. Brown has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(14)(15) | Includes 81,875(i) 4,800 shares of common stock held by Mr. Morrison; and (ii) 87,309 shares of common stock that Mr. Morrison has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(15)(16) | Includes (i) 26,4282,400 shares of common stock held by Mr. Patrick; and (ii) 5,434 shares of common stock that Mr. Patrick has the right to acquire from us within 60 days of March 31, 2021 pursuant to the exercise of stock options. |
| (17) | Includes (i) 31,228 shares of common stock held by Mr. Perry; and (ii) 75,47527,309 shares of common stock that Mr. Perry has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(16)(18) | Includes 81,875(i) 19,800 shares of common stock held by Dr. Pierce; and (ii) 72,309 shares of common stock that Dr. Pierce has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(17)(19) | Includes 51,875(i) 4,800 shares of common stock held by Dr. Pizzo; and (ii) 57,309 shares of common stock that Dr. Pizzo has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(18)(20) | Includes 21,041(i) 2,256 shares of common stock held by Ms. Svoronos; and (ii) 36,475 shares of common stock that Ms. Svoronos has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(19)(21) | Includes 46,041622 shares of common stock that Dr. Thompson has the right to acquire from us within 60 days of March 31, 2021 pursuant to the exercise of stock options. |
| (22) | Includes (i) 4,800 shares of common stock held by Ms. Yarno; and (ii) 57,309 shares of common stock that Ms. Yarno has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
(20)(23) | Includes the number of shares beneficially owned by the named executive officers and directors listed in the table above, as well as: (i) 3,52712,005 shares of common stock held by Lesley CalhounBrian Cathers, Ph.D. and 36,22337,343 shares of common stock that Ms. CalhounDr. Cathers has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options; and (ii) 161,5084,661 shares of common stock held by Ms. Choi, 25,000 shares of common stock held in The 2005 William ParkEric Fink and Jung Choi Family Trust and 154,719 shares of common stock that Ms. Choi has the right to acquire from us within 60 days of March 31, 2020 pursuant to the exercise of stock options; (iii) 2,40628,656 shares of common stock that Mr. Fink has the right to acquire from us within 60 days of March 31, 2020 pursuant to the exercise of stock options; and (iv) 11,521 shares of common stock held by Ms. Suvari and 124,906 shares of common stock that Ms. Suvari has the right to acquire from us within 60 days of March 31, 20202021 pursuant to the exercise of stock options. |
602021 Proxy Statementï Global Blood Therapeutics, Inc.
HOUSEHOLDING OF PROXY MATERIALS We have made available a procedure approved by the SEC known as “householding.” This procedure allows multiple stockholders residing at the same address the convenience of receiving a single copy of our Notice, Annual Report and proxy materials, as applicable. This allows us to save money by reducing the number of documents we must print and mail, and helps protect the environment as well. Householding is available to both registered stockholders (i.e., those stockholders with certificates registered in their name) and streetnamestreet name holders (i.e., those stockholders who hold their shares through a brokerage). Registered Stockholders If you are a registered stockholder and would like to consent to a mailing of proxy materials and other stockholder information only to one account in your household, as identified by you, we will deliver or mail a single copy of our Annual Report and proxy materials for all registered stockholders residing at the same address. Your consent will be perpetual unless you revoke it, which you may do at any time by contacting the Proxy Department of Continental Stock Transfer & Trust Company, LLC, or Continental, at 1 State Street, 30th30th Floor, New York, NY 10004. Registered stockholders who have not consented to householding will continue to receive copies of Annual Reports and proxy materials for each registered stockholder residing at the same address. As a registered stockholder, you may elect to participate in householding and receive only a single copy of Annual Reports or proxy statements for all registered stockholders residing at the same address by contacting Continental as outlined above. Street Name Holders Stockholders who hold their shares through a brokerage may elect to participate in householding or revoke their consent to participate in householding by contacting their respective brokers. Global Blood Therapeutics, Inc. ï2021 Proxy Statement61
OTHER MATTERS We are not aware of any matters that may come before the meeting other than those referred to in the Notice. If any other matter shall properly come before the Annual Meeting, however, the persons named in the accompanying proxy intend to vote all proxies in accordance with their best judgment. Accompanying this Proxy Statement is our Annual Report. Copies of our Annual Report are available free of charge on our website athttps://ir.gbt.com/financial-information/sec-filings or you can request a copy free of charge by calling Investor Relations at 650.741.7730 or sending ane-mail request to investor@gbt.com. Please include your contact information with the request. By Order of the Board of Directors, | Global | By Order of the Board of Directors, | Global Blood Therapeutics,Inc. |
| Ted W. Love, M.D. | Ted W. Love, M.D. | President and Chief Executive Officer |
Ted W. Love, M.D. President and Chief Executive Officer April 28, 20202021 622021 Proxy Statementï Global Blood Therapeutics, Inc.
 GLOBAL BLOOD THERAPEUTICS, INC. 181 OYSTER POINT BOULEVARD SOUTH SAN FRANCISCO, CA 94080 VOTE BY INTERNET Before The Meeting - Go to www.proxyvote.com Use the Internet to transmit your voting instructions and for electronic delivery of information up until 11:59 p.m. Eastern Time on June 16, 2021. Have your proxy card in hand when you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form. During The Meeting - Go to www.virtualshareholdermeeting.com/GBT2021 You may attend the meeting via the Internet and vote during the meeting. Have the information that is printed in the box marked by the arrow available and follow the instructions. VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions up until 11:59 p.m. Eastern Time on June 16, 2021. Have your proxy card in hand when you call and then follow the instructions. VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. | | | 
GLOBAL BLOOD THERAPEUTICS, INC.
181 OYSTER POINT BOULEVARD
SOUTH SAN FRANCISCO, CA 94080
| | VOTE BY INTERNET
Before The Meeting -Go towww.proxyvote.com
Use the Internet to transmit your voting instructions and for electronic delivery of information up until 11:59 p.m. Eastern Time on June 16, 2020. Have your proxy card in hand when you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form.
| | During The Meeting -Go towww.virtualshareholdermeeting.com/GBT2020
You may attend the meeting via the Internet and vote during the meeting. Have the information that is printed in the box marked by the arrow available and follow the instructions.
| | VOTE BY PHONE - 1-800-690-6903
Use any touch-tone telephone to transmit your voting instructions up until 11:59 p.m. Eastern Time on June 16, 2020. Have your proxy card in hand when you call and then follow the instructions.
| | | | | VOTE BY MAIL
Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717.
|
| | | | TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: | | | D15136-P35794 | | D52148-P49635 | | KEEP THIS PORTION FOR YOUR RECORDS | | — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — —— — —— — —— — —— — —— — — — |
— — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — —
| | | | | DETACH AND RETURN THIS PORTION ONLY | | THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED. |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | GLOBAL BLOOD THERAPEUTICS, INC. | | For
All
| | | Withhold
All
| | | | For All
Except
| | | For All | | Withhold All | | For All Except | | | | To withhold authority to vote for any individual nominee(s), mark “For All Except” and write the number(s) of the nominee(s) on the line below. | | | | | | | | | | | | | | | | | | | The Board of Directors recommends you vote FOR
the following Class II Directors in Proposal 1:
| | ☐ | | | ☐ | | | | ☐ | | | | | | | | | | | | | | | | | | | | | | 1. Election of Directors.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Nominees:
The Board of Directors recommends you vote FOR the following Class III Directors in Proposal 1: | | ☐ | | | | ☐ | | ☐ | | | | | | | | | | | | | | | | | | | | | | | 01) Willie L. Brown, Jr.
02) Philip A. Pizzo, M.D.
03) Wendy Yarno
| | | | | | | | | | | | | | | | | | | | | 1. | | Election of Directors. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Nominees: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 01) | | Scott W. Morrison | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 02) | | Deval L. Patrick | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 03) | | Mark L. Perry | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | The Board of Directors recommends you vote FOR Proposals 2 and 3: | | | | | | | | | | | | | For | | | Against | | | Abstain | | Abstain | | | | | | | | | | 2. | | 2. Approval, on a non-binding, advisory basis, of the compensation of the Company’s named executive officers as disclosed in the proxypoxy statement.
| | | | ☐ | | | ☐ | | | | ☐ | | | | | | | | | | | | | 3. | | Ratification of the appointment of KPMG LLP as the independent registered public accounting firm of the Company for its fiscal year ending December 31, 2020.2021. | | | | ☐ | | | ☐ | | | | ☐ | | | | | | | | | | | | | | 4. | | Transaction of such other business as may properly come before the meeting or any adjournment or postponement thereof. | | | | | | | | | | | | | | | | | | | | | | | | | | NOTE:This proxy, when properly executed, will be voted as directed herein by the undersigned Stockholder. If no direction is made, this proxy will be voted “FOR ALL” in Proposal 1 and “FOR” Proposals 2 and 3. | | | | | | | | | | |
| | | | | | | | | Please sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name by authorized officer. office. | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | Signature [PLEASE SIGN WITHIN BOX] | | Date | | |
| | | | | | | | Signature (Joint Owners) | | | | | Date | | | | | | | | | | | | | | | | | | | Signature [PLEASE SIGN WITHIN BOX]
| | Date
| | | | | | | | | | | | Signature (Joint Owners)
| | Date
| | | | | | | | | | | | | | | | |
Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice and Proxy Statement and Annual Report and Form 10-K are available at www.proxyvote.com. — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — —
| | — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — — |
D15137-P35794D52149-P49635
| | | GLOBAL BLOOD THERAPEUTICS, INC. Annual Meeting of Stockholders June 17, 20202021 8:00 AM This proxy is solicited by the Board of Directors The stockholder(s) hereby appoint(s) Ted Love, M.D. and Jeffrey Farrow, or either of them, as proxies, each with the power to appoint his substitute, and hereby authorize(s) them to represent and to vote, as designated on the reverse side of this ballot, all of the shares of Common Stock of GLOBAL BLOOD THERAPEUTICS, INC. that the stockholder(s) is/are entitled to vote at the Annual Meeting of Stockholders to be held at 8:00 AM PDT on June 17, 2020,2021, held virtually at www.virtualshareholdermeeting.com/GBT2020,GBT2021, and any adjournment or postponement thereof. This proxy, when properly executed, will be voted in the manner directed herein. If no such direction is made, this proxy will be voted in accordance with the Board of Directors’ recommendations. Continued and to be signed on reverse side | | |
|