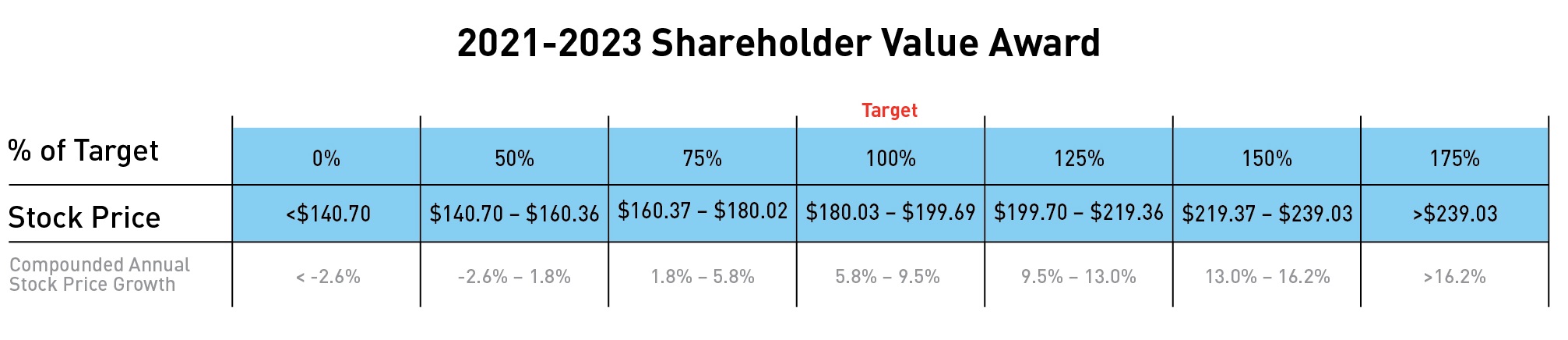
For purposes of establishing the stock price target for the shareholder value awards, the starting price was $72.15$152.16 per share, the average closing stock price for all trading days in November and December 2016.2020. The target share price was established using the expected annual rate of return for large-cap companies (8 percent), less an assumed Lilly dividend yield of 2.88 percent.(2.23 percent). To determine payout, the ending price will be the average of the closing pricesprice of company stock for all trading days in November and December 2019.2023. The award is designed to deliver no payout to executive officers if the shareholder return (including projected dividends) is zero or negative. Possible payouts based on share price ranges are illustrated in the grid below.
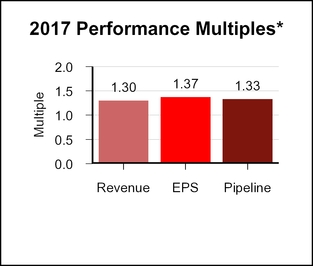
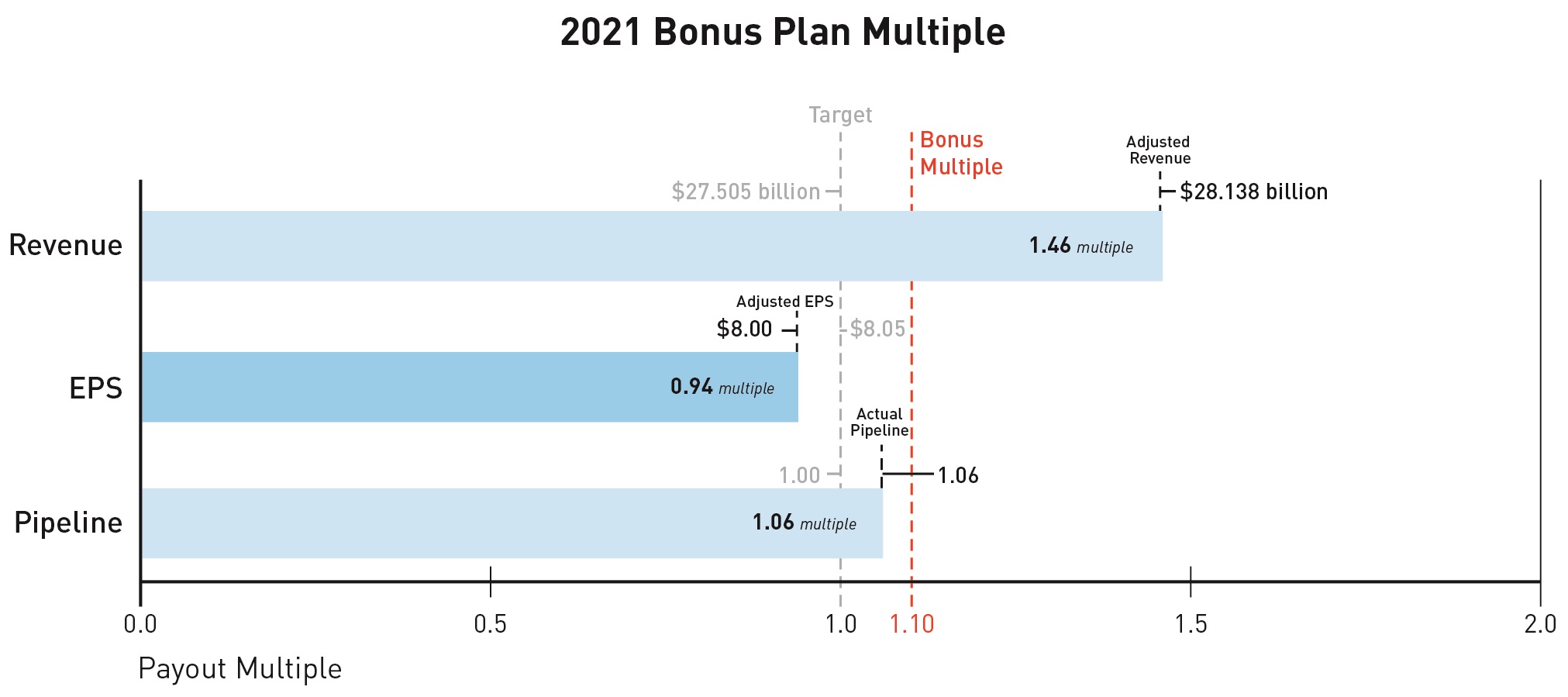
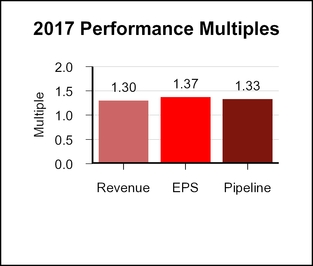
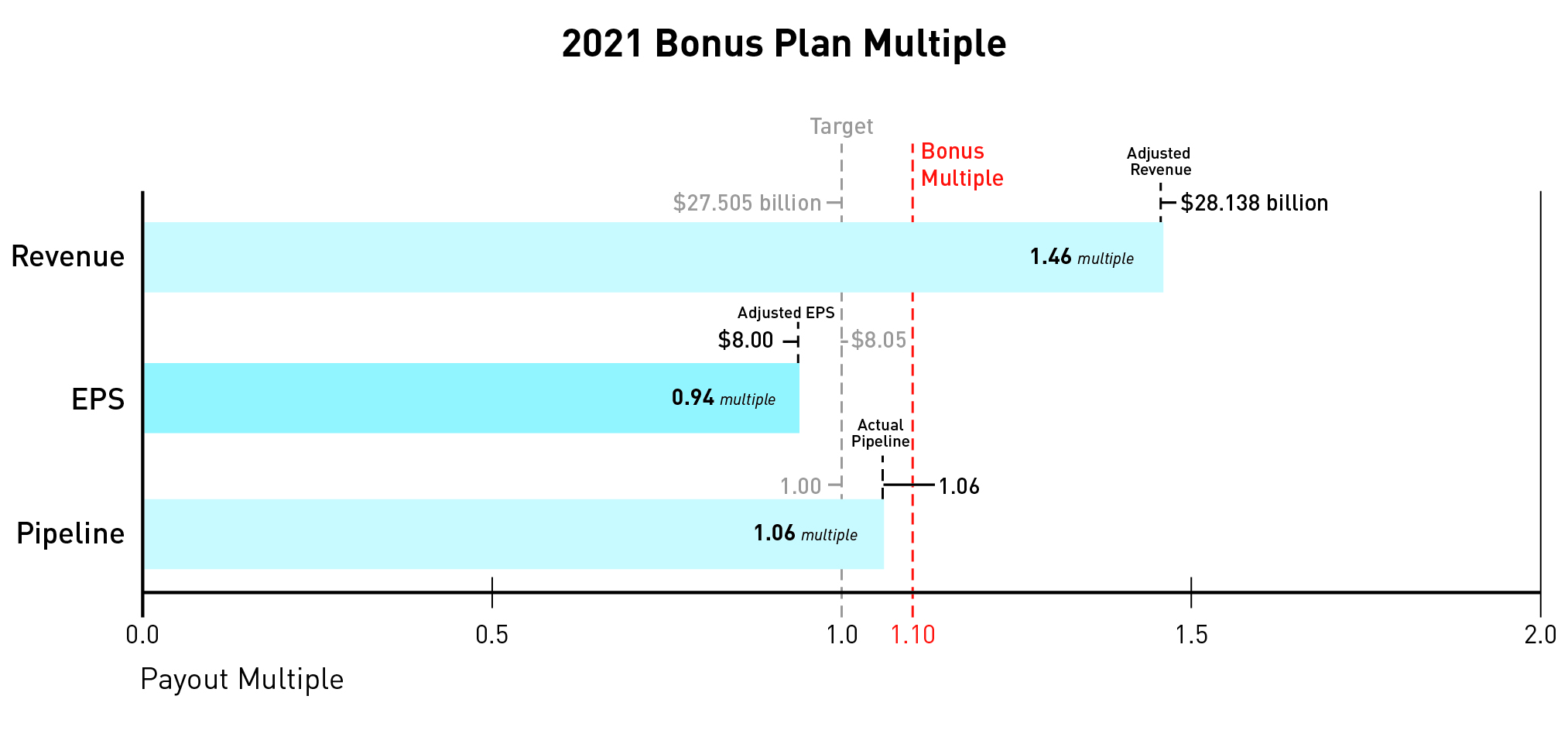
Based on the recommendation of the Science and Technology Committee, the Compensation Committee certified a pipeline score of 3.65, resulting inapproved a pipeline multiple of 1.33.1.06.
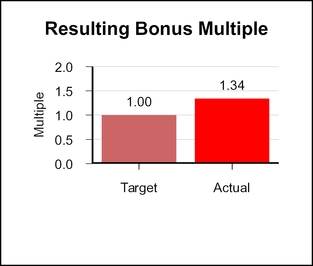
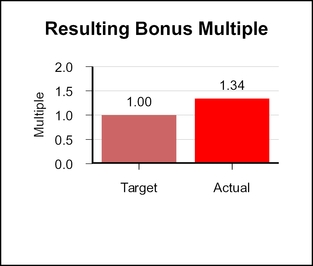
When combined, the revenue, EPS, and pipeline multiples yielded a bonus multiple of 1.34.1.10.
Members of senior management may defer receipt of part or all of their cash compensation under The Lilly Deferred Compensation Plan (Deferred Compensation Plan), which allows executives to save for retirement in a tax-effective way at minimal cost to the company. Under this unfunded plan, amounts deferred by the executive are credited at an interest rate of 120 percent of the applicable federal long-term rate, as described in more detail following the “Nonqualified"Nonqualified Deferred Compensation in 2017”2021" table.
The company has adopted change-in-control severance pay plans for nearly all employees, including the executive officers. The plans are intended to preserve employee morale and productivity and encourage retention in the face of the disruptive impact of an actual or rumored change in control. In addition, the plans are intended to align executive and shareholder interests by enabling executives to evaluate corporate transactions that may be in the best interests of the shareholders and other constituents of the company without undue concern over whether the transactions may jeopardize the executives’ own employment.
Although benefit levels may differ depending on the employee’s job level and seniority, the basic elements of the plans are comparable for all eligible employees:
•Excise taxtax:. In some circumstances, the payments or other benefits received by the employee in connection with a change in control could exceed limits established under Section 280G of the Internal Revenue Code. The employee would then be subject to an excise tax on top of normal federal income tax. The company does not reimburse employees for these taxes. However, the amount of change in control-related benefitsany change-in-control-related benefit will be reduced to the 280G limit if the effect would be to deliver a greater after-tax benefit than the employee would receive with an unreduced benefit.
Share Ownership and Retention Guidelines; Prohibition on Hedging and Pledging Shares
GuidelinesShare ownership and retention guidelines help to fostercreate direct alignment of interests between senior management and shareholders over the longer term. Lilly has established a focus on long-term growth. Theformal share ownership policy under which the CEO isand other senior executives are required to own company stock valued at least six times annualacquire and hold Lilly shares in an amount representing a multiple of base salary. During 2017,2021, the holding requirementCompensation Committee approved an increase to share ownership requirements for the CEO and the other named executive officers ranged from two to three times annual base salary depending on the position. Beginning in 2018, the holding requirement for other executive officers will range from two to four times annual base salary depending on the position. as follows:
| | | | | | | | |
| Previous Share Ownership Requirement | Current Share Ownership Requirement |
| CEO | 6X Base Salary | 12X Base Salary |
| Other NEOs | 4X Base Salary | 6X Base Salary |
Until ownership of the required number of shares is reached, thean executive officer must retainhold 50 percent of all shares, net of taxes receivedtax, from newall equity payouts. Our executives have a long history of maintaining significant levels of company stock. As of December 31, 2017, Mr. Ricks held shares valued at approximately 8 times his annual salary. The following table shows the share requirements for the named executive officers:
|
| | | |
Name | Share Requirement
2017 2018
| Owns Required 2018 Shares |
Mr. Ricks | six times base salary | six times base salary | Yes |
Mr. Conterno | three times base salary | four times base salary | Yes |
Mr. Rice (retired) | three times base salary
| four times base salary | Yes |
Dr. Lundberg | three times base salary
| four times base salary | Yes |
Mr. Harrington | three times base salary
| four times base salary | Yes
|
Executive officers are also required to hold all shares received from equity program payouts, net of acquisition costs and taxes, for at least one year, even once share ownership requirements have been met. For performance awards granted to executive officers, this holding requirement is met by the 13-month service-vesting period that applies after the end of the performance period.
All of the named executive officers are compliant with the share ownership guidelines. The following graphic shows each respective named executive officer's guideline and each named executive officer's holdings as of December 31, 2021, other than Mr. Smiley, who resigned effective as of February 9, 2021.
Prohibition on Hedging and Pledging Shares
Non-employee directors and employees, including executive officers, are not permitted to hedge their economic exposures to company stock through short sales or derivative transactions. Non-employee directors and all members of senior managementemployees, including executive officers, are prohibited from pledging any company stock (i.e., using company stock as collateral for a loan or trading shares on margin).
Executive Compensation Recovery Policy
AllPursuant to our executive compensation recovery policy (the Executive Compensation Recovery Policy), all incentive awards are subject to forfeiture upon termination of employment prior to the end of the performance or vesting period or for disciplinary reasons. In addition, the Compensation Committee has adopted an executive compensation recovery policy that givesupdates to the Executive Compensation Recovery Policy to be effective January 1, 2022 (the Executive Compensation Recovery Policy, as updated and effective January 1, 2022, the Updated Executive Compensation Recovery Policy) which provides the Compensation Committee broad discretion
to claw back incentive payoutsnon-qualified retirement benefits accrued beginning January 1, 2022 from any member of senior management (approximately 150 employees) whose misconduct results in a material violation of law or company policy that causes significant harm to the company or who fails in his or her supervisory responsibility to prevent such misconduct by others.
Additionally,Pursuant to the Executive Compensation Recovery Policy, the company cancould recover all or a portion of any incentive compensation and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, the company could also recover non-qualified retirement benefits accrued beginning January 1, 2022 from an executive officer incentive compensation in the case of materially inaccurate financial statements or material errors in the performance calculation, whether such inaccuracies or not theyerrors result in a restatement and whether or not the executive officer has engaged in wrongful conduct.
The recovery policyExecutive Compensation Recovery Policy covers any incentive compensation awarded or paid to an employee at a time when he or she is a member of senior management.management during the last three years and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, covers non-qualified retirement benefits accrued beginning January 1, 2022. Subsequent changes in status, including retirement or termination of employment, do not affect the company’s rights to recover compensation under the policy. Recoveries under
The company will disclose decisions to take action pursuant to the plan can extend back as far as three years.
Looking AheadUpdated Executive Compensation Recovery Policy in its proxy materials when required by, and in compliance with, SEC rules and regulations and other applicable laws. In addition, when legally permissible to 2018 Compensation
Lilly’s Board of Directors unanimously elected Joshua L. Smiley to assume the role of Senior Vice President and Chief Financial Officer effective January 1, 2018, succeeding Mr. Rice, who retired fromdo so, the company atwill disclose a decision to take action under this policy when the end of 2017. In connection with his appointment, Mr. Smiley will receive a base salary of $875,000facts and will be eligible for an annual cash bonus with a target of 95 percent of base salary. Mr. Smiley received an equity award in February 2018 as partcircumstances of the company’s annual equity incentive program with a grant valuematter that triggered application of $2.3 million. One hundred percent of this grant value was deliveredthe policy have been publicly disclosed in the form of performance-based equity: 60 percent in shareholder value awardscompany’s filings with the SEC and 40 percent in performance awards.
where disclosure can be made without prejudicing the company and its shareholders.
The principles of our robust Updated Executive Compensation Recovery Policy are also incorporated into the terms of our incentive plans and award agreements, which, in the event of misconduct meeting the standards described above, allow the Compensation Committee approved new share ownership guidelines for named executive officers other thanto reduce or cancel awards or payouts that would otherwise have been earned based on company performance. Action by the CEO. WhileCompensation Committee to reduce or cancel awards or payouts can occur during or following the CEO’s requirement remains six times his annual base salary, the named executive officers' requirement increased from three times to four times annual base salary.relevant performance period.
Compensation Committee Matters
Background
Role of the Independent Consultant in Assessing Executive Compensation
SummaryThe Compensation Table
|
| | | | | | | | | | | | | | | | | | | |
| Name and Principal Position | Year | Salary
($) | Bonus
($) | Stock Awards
($) 1 | | Option Awards
($) | Non-Equity Incentive Plan Compensation
($) 2 | Change in
Pension Value
($) 3 |
| All Other Compensation
($) 4 | Total Compensation
($) |
| David A. Ricks | 2017 |
| $1,400,000 |
| $0 | $10,200,000 | | $0 |
| $2,814,000 |
| $1,347,991 |
|
| $84,000 |
|
| $15,845,991 |
|
Chairman, President, and
Chief Executive Officer | 2016 | N/A | N/A | N/A | | N/A | N/A | N/A |
| N/A | N/A |
| 2015 | N/A | N/A | N/A | | N/A | N/A | N/A |
| N/A | N/A |
| Enrique A. Conterno | 2017 |
| $762,002 |
| $0 | $6,000,000 |
| $0 |
| $816,866 |
| $999,426 |
|
| $45,720 |
|
| $8,624,014 |
|
Senior Vice President and
President, Lilly Diabetes and President, Lilly USA | 2016 |
| $727,960 |
| $0 | $2,200,000 | | $0 |
| $681,371 |
| $935,408 |
|
| $43,678 |
|
| $4,588,417 |
|
| 2015 |
| $705,653 |
| $0 | $2,270,000 | | $0 |
| $852,075 |
| $0 | 5 |
| $42,339 |
|
| $3,870,067 |
|
| Derica W. Rice (retired) | 2017 |
| $1,089,134 |
| $0 | $4,560,000 | | $0 |
| $1,459,440 |
| $1,719,690 |
|
| $65,348 |
|
| $8,893,612 |
|
Executive Vice President,
Global Services and
Chief Financial Officer | 2016 |
| $1,067,805 |
| $0 | $3,800,000 | | $0 |
| $1,249,332 |
| $1,739,429 |
|
| $64,068 |
|
| $7,920,634 |
|
| 2015 |
| $1,045,200 |
| $0 | $4,313,000 | | $0 |
| $1,514,495 |
| $0 | 5 |
| $62,712 |
|
| $6,935,407 |
|
| Jan M. Lundberg, Ph.D. | 2017 |
| $1,024,643 |
| $0 | $4,320,000 | | $0 |
| $1,373,021 |
| $618,333 |
|
| $61,479 |
|
| $7,397,476 |
|
Executive Vice President, Science
and Technology and President,
Lilly Research Laboratories | 2016 |
| $1,007,855 |
| $0 | $3,600,000 | | $0 |
| $1,179,190 |
| $627,381 |
|
| $60,471 |
|
| $6,474,897 |
|
| 2015 |
| $1,007,855 |
| $0 | $3,859,000 | | $0 |
| $1,460,382 |
| $390,645 |
|
| $60,471 |
|
| $6,778,353 |
|
| Michael J. Harrington | 2017 |
| $856,130 |
| $0 | $2,760,000 | | $0 |
| $917,771 |
| $1,657,718 |
|
| $51,368 |
|
| $6,242,987 |
|
Senior Vice President and
General Counsel | 2016 |
| $827,400 |
| $0 | $2,300,000 | | $0 |
| $774,446 |
| $1,441,954 |
|
| $49,644 |
|
| $5,393,444 |
|
| 2015 |
| $784,167 |
| $0 | $2,610,500 | | $0 |
| $946,881 |
| $391,899 |
|
| $47,050 |
|
| $4,780,497 |
|
Committee has retained Frederic W. Cook & Co., Inc. (FW Cook) as its independent compensation consultant. FW Cook reports directly to the Compensation Committee, and it is not permitted to have any business or personal relationship with management or members of the Compensation Committee. FW Cook’s responsibilities are to:
1 •This column showsreview the grant date fair valuecompany’s total compensation philosophy, peer group, and target competitive positioning for reasonableness and appropriateness;
•review the company’s executive compensation program and advise the Compensation Committee of evolving best practices;
•provide independent analyses and recommendations to the Compensation Committee on the CEO’s pay;
•review the draft CD&A and related tables for the proxy statement;
•proactively advise the Compensation Committee on best practices for board governance of executive compensation; and
•undertake special projects at the request of the Compensation Committee chair.
FW Cook interacts directly with members of company management only on matters under the Compensation Committee’s oversight and with the knowledge and permission of the Compensation Committee chair.
Role of Executive Officers and Management in Assessing Executive Compensation
With the oversight of the CEO and the senior vice president of human resources and diversity, the company’s global compensation group formulates recommendations on compensation philosophy, plan design, and compensation for executive officers (other than the CEO, as noted below). The CEO provides the Compensation Committee with a performance assessment and compensation recommendation for each of the other executive officers. The Compensation Committee considers those recommendations with the assistance of its compensation consultant. The CEO and the senior vice president of human resources and diversity attend Compensation Committee meetings; however, they are not present for executive sessions or any discussion of their own compensation. Only non-employee directors and the Compensation Committee’s consultant attend executive sessions.
The CEO does not participate in the formulation or discussion of his pay recommendations. He has no prior knowledge of the recommendations that FW Cook makes to the Compensation Committee.
Risk Assessment Process
As part of the company's overall enterprise risk management program, in 2021 (consistent with prior years), the Compensation Committee reviewed the company’s compensation policies and practices and concluded that the programs and practices are not reasonably likely to have a material adverse effect on the company. The Compensation Committee noted numerous policy and design features of the company’s compensation programs and governance structure that reduce the likelihood of inappropriate risk-taking, including, but not limited to:
•Only independent directors serve on the Compensation Committee
•The Compensation Committee engages its own independent compensation consultant
•The Compensation Committee has downward discretion to lower compensation plan payouts
•The Compensation Committee approves all adjustments to financial results that affect compensation calculations
•Different measures and metrics are used across multiple incentive plans that appropriately balance cash/stock, fixed/variable pay, and short-term/long-term incentives
•Incentive plans have predetermined maximum payouts
•Performance objectives are challenging but achievable
•Programs with operational metrics have a continuum of payout multiples based upon achievement of performance awardsmilestones, rather than "cliffs" that might encourage suboptimal or improper behavior
•A compensation recovery policy is in place for all members of senior management; negative compensation consequences can result in cases involving serious compliance violations
•Meaningful share ownership and shareholder value awards computedretention requirements are in accordance with FASB ASC Topic 718. See Note 11place for all members of senior management and the consolidatedboard.
Compensation Committee Report
The Compensation Committee evaluates and establishes compensation for executive officers and oversees the deferred compensation plan, management stock plans, and other management incentive and benefit programs. Management has the primary responsibility for the company’s financial statements and reporting process, including the disclosure of executive compensation in the company’sCD&A. With this in mind, the Compensation Committee has reviewed and discussed the CD&A with management. Based on this discussion, the Compensation Committee recommended to the board that the CD&A be included in this proxy statement and the company's Annual Report on Form 10-K for the fiscal year ended December 31, 20172021, for additional detail regarding assumptions underlyingfiling with the valuationSEC.
Compensation Committee
Ralph Alvarez, Chair
J. Erik Fyrwald
Kimberly H. Johnson
Juan R. Luciano
Karen Walker
Compensation Discussion and Analysis
This CD&A describes our executive compensation philosophy, the Compensation Committee’s process for setting executive compensation, the elements of equity awards. Allour compensation program, the factors the Compensation Committee considered when setting executive compensation for 2021, and how the company’s results affected incentive payouts. This CD&A provides compensation information for our chief executive officer, David Ricks, our chief financial officer, Anat Ashkenazi, and our next three most highly compensated executive officers who were serving as executive officers on December 31, 2021, Daniel Skovronsky, Jacob Van Naarden and Diogo Rau. The CD&A also includes our former chief financial officer, Joshua Smiley, who resigned from his officer position on February 9, 2021.
| | | | | |
| Name and principal occupation |
| David A. Ricks Chair, President and CEO |
| Anat Ashkenazi Senior Vice President and Chief Financial Officer |
| Daniel Skovronsky, M.D., Ph.D. Senior Vice President, Chief Scientific and Medical Officer and President, Lilly Research Laboratories |
| Jacob Van Naarden Senior Vice President, CEO Loxo Oncology at Lilly and President, Lilly Oncology |
| Diogo Rau Senior Vice President and Chief Information and Digital Officer |
Four of our named executive officers, Ms. Ashkenazi, Mr. Van Naarden, Mr. Rau and Mr. Smiley, served in their respective roles for a portion of 2021. Ms. Ashkenazi succeeded Mr. Smiley as senior vice president and CFO on February 9, 2021. Prior to becoming CFO, Ms. Ashkenazi served as senior vice president, controller and chief financial officer, Lilly Research Laboratories.
Mr. Van Naarden became senior vice president, CEO of Loxo Oncology at Lilly and President of Lilly Oncology on September 5, 2021. He previously served as CEO of Loxo Oncology at Lilly. Mr. Rau joined Lilly as senior vice president and chief
information and digital officer on May 17, 2021. Prior to joining Lilly, Mr. Rau served as senior director, information systems and technology for retail and online stores at Apple, Inc.
Our Philosophy on Compensation
At Lilly, our purpose is to unite caring with discovery to create medicines that make life better for people around the world. To do this, we must attract, engage, and retain highly talented individuals from a variety of backgrounds and motivate them to create long-term shareholder value by achieving top-tier corporate performance while embracing the company's core values of integrity, excellence, and respect for people. Our compensation programs are designed to help us achieve these goals while balancing the long-term interests of our shareholders and customers.
Objectives
Our compensation and benefits programs are based on the following objectives:
•Reflect individual and company performance: We reinforce a high-performance culture by linking pay with individual and company performance. As employees assume greater responsibilities, the proportion of total compensation based on absolute company performance, relative company performance and shareholder returns increases. We perform annual reviews to ensure our programs provide an incentive to deliver long-term, sustainable business results while discouraging excessive risk-taking or other adverse behaviors.
•Attract and retain talented employees: Compensation opportunity is market competitive and reflects the level of job impact and responsibilities. Retention of talent is an important factor in the “Stock Awards” column were based upondesign of our compensation and benefit programs.
•Implement broad-based programs: While the probable outcomeamount of performance conditions ascompensation paid to employees varies, the overall structure of our compensation and benefit programs is broadly similar across the organization to encourage and reward all employees who contribute to our success.
•Consider shareholder input: Management and the Compensation Committee consider the results of our annual say-on-pay vote and other sources of shareholder feedback when designing executive compensation and benefit programs.
Say-on-Pay Results
At our 2021 annual meeting of shareholders, greater than 95 percent of the grant date, which varyshares cast voted in favor of the company's say-on-pay proposal on executive compensation. Management and the Compensation Committee view this vote as supportive of the company's overall approach toward executive compensation.
Compensation Committee's Processes and Analyses
Setting Compensation
The Compensation Committee considers individual performance assessments, compensation recommendations from the CEO (with respect to each of the other executive officers), company performance, peer group data, input from its compensation consultant, and its own judgment when determining compensation for the company's executive officers.
•Individual performance: Generally, the independent directors, under the direction of the lead independent director, meet with the CEO at the beginning of each year to year. For 2017,establish the probable outcomeCEO's performance objectives. At the end of the year, the independent directors meet to assess the CEO's achievement of those objectives along with other factors, including contribution to the company’s performance, awardsdiversity, ethics, and integrity. This evaluation is used in setting the CEO's compensation opportunity for the next year.
The Compensation Committee receives individual performance assessments and target compensation recommendations from the CEO for each of the remaining executive officers. Each executive officer’s performance assessment is based on the achievement of objectives established at the start of the year, as well as other factors, including contribution to the company's performance, diversity, ethics, and integrity. The Compensation Committee considers these inputs, its knowledge of and interactions with each executive officer, and its judgment to develop a final individual performance assessment. For new executive officers, target compensation is set by the Compensation Committee at the time of grant was at maximum. Aspromotion or offer.
•Company performance: Lilly performance is considered in multiple ways:
•Overall prior year performance is a result,factor in setting target compensation for the values incoming year.
•At the "Stock Awards" columnbeginning of each calendar year, annual performance goals are above target. For Mr. Conterno, this column shows both the grant date fair value of performance awardsestablished and shareholder value awards, as well as a special retention grant of $3 million he received in recognition of his leadership in delivering the company's strategy and providing continuity in a time of significant transition at the company; this special retention grant will vest on December 11, 2021, and it will be forfeited if Mr. Conterno resigns or retires from the company prior to that date.
For purposes of comparison, the supplemental table below shows the total target grant values approved by the committee:compensation committee. Performance against these annual goals is used to determine the short-term cash incentive payout.
•Prior to the annual equity grant, multi-year performance goals are established and approved by the compensation committee. Performance against these multi-year objectives is used to determine the long-term incentive equity payout.
|
| | | |
| Name | 2015 Total Equity | 2016 Total Equity | 2017 Total Equity |
| Mr. Ricks | N/A | N/A | $8,500,000 |
| Mr. Conterno | $2,000,000 | $2,200,000 | $2,500,000 |
| Mr. Rice (retired) | $3,800,000 | $3,800,000 | $3,800,000 |
| Dr. Lundberg | $3,400,000 | $3,600,000 | $3,600,000 |
| Mr. Harrington | $2,300,000 | $2,300,000 | $2,300,000 |
•Input from independent compensation consultant:The Compensation Committee considers the advice of its independent compensation consultant, FW Cook, when setting executive officer compensation.
Competitive Pay Assessment
Lilly’s peer group is composed of companies that directly compete with Lilly, use a similar business model, and employ people with the unique skills required to operate an established biopharmaceutical company. The Compensation Committee selects a peer group whose median market cap and revenue are broadly like Lilly's. The Compensation Committee reviews the peer group at least every three years. The Compensation Committee established the following peer group in May 2018 for purposes of assessing competitive pay:
| | | | | | | | | | | | | | | | | | | | |
| AbbVie | | Bristol-Myers Squibb | | Merck | | Sanofi |
| Allergan* | | Celgene* | | Novartis | | Shire* |
| Amgen | | Gilead | | Novo Nordisk | | Takeda |
| AstraZeneca | | GlaxoSmithKline | | Pfizer | | |
| Biogen | | Johnson & Johnson | | Roche | | |
| *Market data unavailable for assessing competitive 2021 pay due to business mergers. |
At the time of the review in May 2018, all peer companies were no greater than two times our revenue or market cap except Johnson & Johnson, Novartis, Pfizer, and Roche. The Compensation Committee included these four companies despite their size because they compete directly with Lilly, have similar business models, and seek to hire from the same pool of management and scientific talent.
When determining pay levels for target compensation, the Compensation Committee considers an analysis of market competitive pay for each executive officer position (except CEO), along with internal factors such as the performance and experience of each executive officer. The independent compensation consultant for the Compensation Committee provides a similar analysis when recommending pay levels for the CEO. The CEO analysis includes a comparison of our CEO actual total direct compensation in the prior year to company performance on an absolute and relative basis. The analysis also includes a comparison of current target total direct compensation for our CEO to the most recently available data on CEO target total direct compensation for our peer group with an emphasis on peers that are headquartered in the U.S. On average, the named executive officers' target total direct compensation for 2021 was comparable to the median of the peer group.
Components of Our Compensation
Our 2021 executive compensation was primarily composed of three components:
•base salary
•annual cash bonus, which is generally based on company performance relative to internal targets for revenue, EPS, and the progress of our pipeline
•three different forms of equity incentives:
◦performance award, which is a performance-based equity award that vests over three years. Payout is based on the company's two-year growth in EPS relative to the median external analyst anticipated peer group EPS growth. The earned shares are then subject to a 13-month service-vesting period
◦shareholder value award, which is a performance-based equity award that pays out based on absolute company stock price growth measured over a three-year period, followed by a one-year holding period
◦relative value award, which is a performance-based equity award that pays out based on company TSR results relative to peers measured over a three-year period, followed by a one-year holding period.
Executives also receive a company benefits package, described below under "Other Compensation Practices and Information—Employee Benefits."
Adjustments to Reported Financial Results
The tableCompensation Committee has authority to adjust the company's reported revenue and EPS upon which incentive compensation payouts are determined to eliminate the distorting effect of unusual income or expense items. The adjustments are intended to:
•align award payments with the underlying performance of the core business
•avoid volatile, artificial inflation or deflation of awards due to unusual items in any year in the performance period
•eliminate certain counterproductive short-term incentives—for example, incentives to refrain from acquiring new technologies, to defer disposing of underutilized assets, or to defer settling legacy legal proceedings to protect current bonus payments
•facilitate comparisons with peer companies.
The Compensation Committee considers the adjustments approved by the Audit Committee for reporting non-GAAP EPS and other adjustments, based on guidelines approved by the Compensation Committee prior to the performance period. The Compensation Committee reviews and approves adjustments on a quarterly basis and may adjust payouts for items, including but not limited to, the impact of significant acquisitions or divestitures, the impact of share repurchases that differ significantly from business plan, and large swings in foreign exchange rates. Further details on the adjustments for 2021 and the rationale for making these adjustments are set forth in Appendix A, "Summary of Adjustments Related to the Annual Cash Bonus and Performance Award." For ease of reference, throughout the CD&A and the other compensation disclosures, we refer simply to "revenue" and "EPS," but we encourage you to review the information in Appendix A to understand the adjustments from reported revenue and EPS that were approved.
The Compensation Committee also has general authority to apply downward discretion to bonus, performance award, shareholder value award, and relative value award payouts for individual or all executive officers.
1.Base Salary
In setting salaries, Lilly seeks to retain, motivate, and reward successful performers while maintaining affordability within the company's business plan. Base salaries are reviewed and established annually and may be adjusted upon promotion, following a change in job responsibilities, or to maintain market competitiveness. Salaries are based on each person's level of contribution, responsibility, expertise, and competitiveness and are compared annually with peer group data.
Base salary increases for 2021 were based on competitiveness. Generally, executive base salaries are positioned against market salaries, considering experience and performance over time. If an executive's salary is deemed competitive given their experience and performance, no salary increase is provided.
2.Annual Cash Bonus
The Bonus Plan is designed to reward the achievement of the company’s annual financial and innovation objectives. All the named executive officers participated in the Bonus Plan during 2021. Mr. Van Naarden's participation in the Bonus Plan commenced at the time of his promotion on September 5, 2021. For the portion of the 2021 performance year prior to his promotion, Mr. Van Naarden participated in The Loxo Oncology, Inc. Bonus Plan (Loxo Bonus Plan), and in lieu of the payments he would have been eligible to receive thereunder, the Company paid Mr. Van Naarden a one-time payment, as further described below shows the minimum, target, and maximum payouts (using the grant date fair value) for the 2017-2019 performance awards included in the Summary Compensation Table.
|
| | | | |
| Name | Payout Date | Minimum Payout | Target Payout | Maximum Payout |
| Mr. Ricks | January 2019 | $0 | $3,400,000 | $5,100,000 |
| Mr. Conterno | January 2019 | $0 | $1,000,000 | $1,500,000 |
| Mr. Rice (retired) | January 2019 | $0 | $1,520,000 | $2,280,000 |
| Dr. Lundberg | January 2019 | $0 | $1,440,000 | $2,160,000 |
| Mr. Harrington | January 2019 | $0 | $920,000 | $1,380,000 |
Bonus Plan
The table below shows the minimum, target,Compensation Committee sets performance goals and maximum payouts (using the grant date fair value)individual bonus targets for the 2017-2019 shareholder value awards included in the Summary Compensation Table.
|
| | | | |
| Name | Payout Date | Minimum Payout | Target Payout | Maximum Payout |
| Mr. Ricks | January 2019 | $0 | $5,100,000 | $7,650,000 |
| Mr. Conterno | January 2019 | $0 | $1,500,000 | $2,250,000 |
| Mr. Rice (retired) | January 2019 | $0 | $2,280,000 | $3,420,000 |
| Dr. Lundberg | January 2019 | $0 | $2,160,000 | $3,240,000 |
| Mr. Harrington | January 2019 | $0 | $1,380,000 | $2,070,000 |
2 Payments under the Bonus Plan forat the beginning of each year. The bonus is based on three areas of company performance in each of the respective years. All bonuses paidmeasured relative to named executive officers were part of a non-equity incentive plan.internal targets: revenue, EPS, and pipeline progress. The annual cash bonus payout is calculated as follows:
3 The amounts in this column reflect the change in pension value for each individual, calculated by our actuary, and are affected by additional service accruals and pay earned, as well as actuarial assumption changes. The changes in pension values in 2017 were driven to a large extent by a lower discount rate which increased the net present value of pensions. The design of the pension benefit did not change. See the Pension Benefits in 2017 table below for information about the standard actuarial assumptions used. No named executive officer received preferential or above-market earnings on deferred compensation.
4 The amounts in this column are solely company matching contributions for each individual's 401(k) plan and nonqualified savings plan contributions. The company does not reimburse executives for taxes outside of the limited circumstance of taxes related to employee relocation or a prior international assignment. There were no reportable perquisites or personal benefits.
5 In 2015, the net present value of the pension benefits for Mr. Conterno and Mr. Rice reflect no change from the previous year due to an increase in the discount rate over the prior year. For the other named executive officers, increases in pensionable earnings offset the impact of the 2015 increased discount rate.
Grants of Plan-Based Awards During 2017
The compensation plans under which the grants in the following table were made are described in the CD&A and consist of the bonus plan (a non-equity incentive plan) and the 2002 Lilly Stock Plan (which provides for performance awards, shareholder value awards, and restricted stock units).
To receive a payout under the performance award or the shareholder value award, a participant must remain employed with the company through the end of the relevant award period (except in the case of death, disability, retirement, or redundancy). No dividends accrue on either performance awards or shareholder value awards during the performance period. Non-preferential dividends accrue during the 13-month service-vesting period (following the two-year performance period) and are paid upon vesting.
|
| | | | | | | | | | | | |
| Name | Award | | Grant Date2 | Compensation Committee Action Date | Estimated Future Payouts
Under Non-Equity
Incentive Plan Awards1 | Estimated Future
Payouts Under Equity
Incentive Plan Awards | All Other
Stock or Option Awards:
Number of
Shares of Stock,
Options, or Units | Grant Date
Fair Value
of Equity
Awards |
Threshold
($) | Target
($) | Maximum
($) | Threshold
(# shares) | Target
(# shares) | Maximum
(# shares) |
| Mr. Ricks | Annual Bonus | | __ | __ | $52,500 | $2,100,000 | $4,200,000 | | | | | |
| 2017-2019 PA | 3 | 2/9/2017 | 12/12/2016 | | | | 23,117 | 46,233 | 69,350 | | $5,100,000 |
| 2017-2019 SVA | 4 | 2/9/2017 | 12/12/2016 | | | | 39,045 | 78,089 | 117,134 | | $5,100,000 |
| | | | | | | | | | | 0 | |
| Mr. Conterno | Annual Bonus | | __ | __ | $15,240 | $609,601 | $1,219,203 | | | | | |
| 2017-2019 PA | 3 | 2/9/2017 | 12/12/2016 | | | | 6,799 | 13,598 | 20,397 | | $1,500,000 |
| 2017-2019 SVA | 4 | 2/9/2017 | 12/12/2016 | | | | 11,484 | 22,967 | 34,451 | | $1,500,000 |
| RSU | 5 | 12/11/2017 | 12/11/2017 | | | | | | | 34,615 | $3,000,000 |
| Mr. Rice (retired) | Annual Bonus | | __ | __ | $27,228 | $1,089,134 | $2,178,269 | | | | | |
| 2017-2019 PA | 3 | 2/9/2017 | 12/12/2016 | | | | 10,335 | 20,669 | 31,004 | | $2,280,000 |
| 2017-2019 SVA | 4 | 2/9/2017 | 12/12/2016 | | | | 17,455 | 34,910 | 52,365 | | $2,280,000 |
| | | | | | | | | | | 0 | |
| Dr. Lundberg | Annual Bonus | | __ | __ | $25,616 | $1,024,643 | $2,049,285 | | | | | |
| 2017-2019 PA | 3 | 2/9/2017 | 12/12/2016 | | | | 9,791 | 19,581 | 29,372 | | $2,160,000 |
| 2017-2019 SVA | 4 | 2/9/2017 | 12/12/2016 | | | | 16,537 | 33,073 | 49,610 | | $2,160,000 |
| | | | | | | | | | | 0 | |
| Mr. Harrington | Annual Bonus | | __ | __ | $17,123 | $684,904 | $1,369,808 | | | | | |
| 2017-2019 PA | 3 | 2/9/2017 | 12/12/2016 | | | | 6,255 | 12,510 | 18,765 | | $1,380,000 |
| 2017-2019 SVA | 4 | 2/9/2017 | 12/12/2016 | | | | 10,565 | 21,130 | 31,695 | | $1,380,000 |
| | | | | | | | | | | 0 | |
1 These columns show the threshold, target, and maximumActual payouts for performance under the Bonus Plan. Bonus payoutscan range from 0 to 200 percent of an individual's bonus target. The Compensation Committee references the annual operating plan and pipeline objectives to establish performance targets and to assess the relative weighting for each objective. In 2021, the weightings remain unchanged from the prior year:
| | | | | |
| Lilly Goals | Weighting |
| Revenue performance | 25% |
| EPS performance | 50% |
| Pipeline progress | 25% |
Based on this weighting, the company bonus paymentmultiple is calculated as follows:
Loxo Bonus Plan
Performance goals for 2017 performance was 134the Loxo Bonus Plan are determined by a committee consisting of senior company leadership (the Loxo Oncology Steering Committee) and mirror the goals under the Bonus Plan; provided, however, that eligible employees have an additional bonus opportunity that may be awarded for the attainment of other goals specific to oncology research and development (Additional Cash Award). These other goals can vary annually, but typically include preclinical, clinical, regulatory actions, external innovation and scientific publication milestones. These additional goals associated with molecule advancement and proof of concept milestones typically carry more weight in the Additional Cash Award results.
The annual Loxo Bonus Plan payout is calculated by multiplying a participant's base earnings, the individual bonus target and the sum of the Bonus Plan and Additional Cash Award payout multiples.
Base Earnings x Individual Bonus Target x (Bonus Plan Multiple + Additional Cash Award Multiple)
Actual payouts for the Loxo Bonus Plan can range from 0 to 400 percent of an individual's bonus target (0 to 200 percent for the Bonus Plan performance and 0 to 200 percent for the Additional Cash Award). Goal attainment is includeddetermined by the Loxo Oncology Steering Committee.
3.Equity Incentives
The company grants three types of equity incentives to executives and certain other employees: performance awards that are designed to focus leaders on multi‑year operational performance relative to peer companies, shareholder value awards that are intended to align earned compensation with long-term growth in shareholder value, and relative value awards which encourage TSR outperformance compared to our peer group. These awards, when considered together, align with shareholder interests by incenting long-term operational excellence, shareholder return and peer company outperformance without encouraging excessive risk-taking behaviors. The Compensation Committee has the discretion to adjust payout from an equity award granted to an executive officer downward from the amount yielded by the applicable formula.
Performance Awards
Performance awards vest over three years. Payouts are based on achieving EPS growth targets over a two-year performance period, followed by an additional 13-month service-vesting period for executive officers. During the service-vesting period, the award is held in the form of restricted stock units. The growth-rate targets are set relative to the median external analyst anticipated EPS growth for our peer group over the same performance period. These awards do not accumulate dividends during the two-year performance period, but they do accumulate dividend equivalent units during the service-vesting period.
The Compensation Committee believes EPS growth is an effective measure of operational performance because it is closely linked to shareholder value, is broadly communicated to the public, is understood by Lilly employees, and allows for objective comparisons to the performance of Lilly's peer group. Consistent with the objectives established by the Compensation Committee, Lilly company performance exceeding the expected peer group median results in above‑target payouts, while Lilly company performance lagging the expected peer group median results in below‑target payouts. Possible payouts range from 0 percent to 175 percent of the target number of shares, depending on Lilly EPS growth over the performance period.
The measure of EPS used in the performance award program differs from the measure used in the Bonus Plan in two ways. First, the EPS goal in the Bonus Plan is set with reference to internal goals that align to our annual operating plan, while the EPS goal in the performance award program is set based on the external analyst anticipated EPS growth rates of our peer group. Second, the Bonus Plan measures EPS over a one-year period, while the performance award program measures EPS over a two-year period. In any given year, the Bonus Plan may pay above target while the performance award pays below target (or vice versa).
Shareholder Value Awards
Shareholder value awards are earned based on Lilly's share price performance. Shareholder value awards pay above target if Lilly's stock outperforms an expected rate of return and below target if Lilly's stock underperforms that expected rate of return. The expected rate of return is based on the three-year TSR that a reasonable investor would consider appropriate when investing in a basket of large-cap U.S. companies, as determined by the Compensation Committee. The minimum price to achieve target is calculated by multiplying the starting share price of Lilly's stock by the three-year compounded expected rate of return less Lilly's dividend yield. Shareholder value awards have a three-year performance period, and any shares paid are subject to a one-year holding requirement. No dividends are accrued during the performance period. Possible payouts are based on share price growth and range from 0 to 175 percent of the target number of shares. Executive officers receive no payout if Lilly's TSR for the three-year period is zero or negative.
Relative Value Awards
Relative value awards are earned based on Lilly's TSR performance relative to our peer group. The minimum performance to achieve target is a TSR that is equal to the median TSR performance for the peer group. Relative value awards have a three-year performance period, and any shares paid are subject to a one-year holding requirement. No dividends are accrued during the performance period. Executive officers receive no payout if Lilly's TSR for the three-year period is 30 or more percentage points below the median TSR performance for the peer group over the same time period. Possible payouts range from 0 to 175 percent of the target number of shares.
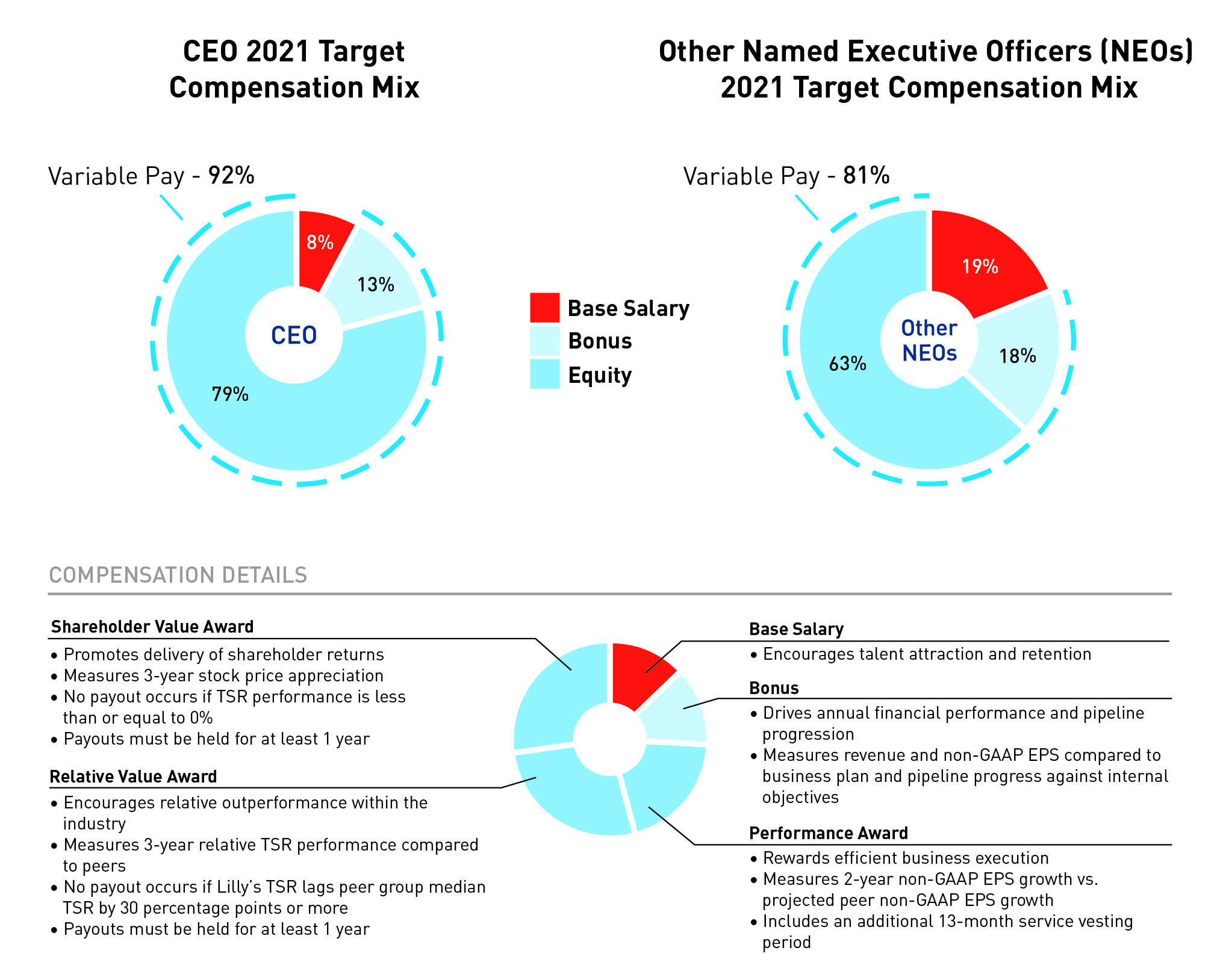
Pay for Performance
The mix of compensation for our named executive officers reflects Lilly’s desire to link executive compensation with individual and company performance. As reflected in the charts below, a substantial portion of the target pay for executive officers is performance-based. The annual cash bonus and equity payouts are contingent upon company performance, with the bonus factoring in performance over a one-year period, and equity compensation factoring in performance over multi-year periods. The charts below depict the annualized mix of target compensation for Lilly’s CEO and the average for the
other named executive officers annualized for Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau. The Other Named Executive Officers chart below excludes Mr. Smiley.
Target Total Compensation
Performance Review Process
In setting 2021 target compensation for the named executive officers, the Compensation Committee considered individual contributions, Lilly's performance during 2020, internal relativity, peer group data, and input from the CEO for named executive officers other than himself.
Evaluation of Individual Named Executive Officer Performance
A summary of the Compensation Committee's review of individual named executive officer performance in 2020 that influenced decisions on 2021 target compensation for these executives is provided below:
David Ricks, Chair, President, and CEO:
In accordance with the company’s corporate governance guidelines, the independent directors conducted an assessment of Mr. Ricks’ performance led by the lead independent director. The independent directors believe the company largely met or exceeded its combined financial and strategic goals for 2020 under Mr. Ricks’ leadership. In 2020, Mr. Ricks and his team:
•Launched several innovative medicines to patients globally. Additionally, 25 new indications for already marketed medicines were launched to patients around the world
•Progressed 17 potential new medicines into Phase I clinical development from both internal research and external sources
•Initiated actions against the global COVID-19 pandemic, including (1) implementing diagnostic testing in our community, (2) discovering and developing SARS-CoV-2 neutralizing antibodies leading to the Emergency Use Authorization for bamlanivimab, and (3) striving to protect the health and safety of Lilly employees while continuing to provide life saving medicines
•Completed the acquisition of Dermira, expanded Lilly's strategic collaboration with Innovent for Tyvyt globally and completed seven preclinical asset transactions
•Continued to drive a cross-company productivity agenda resulting in savings that funded increased investment in research and development
•Implemented a strategy that improved diversity, equity and inclusion across the company, increased the representation of women and minorities in management and conducted pay equity studies to promote equality in pay. The company was ranked #3 on the DiversityInc. top 50 companies list and received Women Business Collaborative CEO recognition for excellence in gender diversity
•Led the development of Lilly's comprehensive Racial Justice Commitment to help improve equity within Lilly and the communities in which it operates
•Achieved certain 2020 environmental performance goals first established in 2013, such as greenhouse gas emissions, energy efficiency, waste efficiency and wastewater
•Established new long-term goals for environmental, safety and governance goals for 2030
In addition, the company was named one of the world’s most ethical companies by the Ethisphere Institute.
Anat Ashkenazi, Senior Vice President and Chief Financial Officer:
Ms. Ashkenazi became Senior Vice President and Chief Financial Officer (CFO) on February 9, 2021. Prior to becoming CFO, Ms. Ashkenazi served as senior vice president and controller, and led corporate strategy at Lilly Research Laboratories. As controller, Ms. Ashkenazi:
•Played a key role in the company achieving its financial targets in 2020 and in the company’s efforts to reallocate financial resources in the fight against COVID-19
•Led the company’s broad productivity agenda for several years, having been a key partner to the operating units across the value cycle and played a key role in all business development activities
•Led the company’s 2021 strategic plan and 2021 operating plan
•Sponsors Lilly’s Veteran Network, an employee resource group focused on recruiting and advancing people who served in the country’s military
Daniel Skovronsky, M.D., Ph.D., Senior Vice President, Chief Scientific and Medical Officer and President, Lilly Research Laboratories:
Dr. Skovronsky advanced innovation for patients as the company's chief scientific and medical officer. His contributions in 2020 include:
•Advanced innovative medicines through the product pipeline leading to numerous first approvals, including bamlanivimab (under Emergency Use Authorization from the FDA) in partnership with AbCellera. Additionally, 25 new indications for already-marketed medicines were launched to patients globally
•Achieved several significant positive data readouts for Verzenio (Phase III in the adjuvant setting), LOXO-305 (advanced b-cell malignancies), Jardiance (cardiovascular outcomes in heart failure with reduced ejection fraction), mirikizumab (psoriasis head-to-head versus secukinumab) and tirzepatide (type 2 diabetes)
•Co-led the acquisition of Dermira, expanded Lilly's strategic collaboration with Innovent for Tyvyt globally, and completed seven preclinical asset transactions
•Sped research resulting in 17 potential new medicines advancing to Phase I clinical development including both internally discovered molecules and molecules sourced externally via business development
•Led the company’s efforts in the fight against SARS-CoV-2 including (1) implementing diagnostic testing at our headquarters in Indianapolis, Indiana and (2) discovering and developing SARS-CoV-2 neutralizing antibodies leading to the Emergency Use Authorization for bamlanivimab
•Enhanced strategies to further reduce drug discovery and development timelines, leading to earlier product launch
•Led DEI strategies in research and development to improve innovation and productivity.
Jacob Van Naarden, Senior Vice President, CEO Loxo Oncology at Lilly and President, Lilly Oncology:
Mr. Van Naarden served as chief operating officer of Loxo Oncology at Lilly when the company acquired Loxo Oncology, Inc. in 2019 and was later named CEO of Loxo Oncology at Lilly in January 2021. Mr. Van Naarden played an integral role with numerous oncology assets including the approval of Retevmo. As CEO of Loxo Oncology at Lilly, Mr. Van Naarden has built a science-first drug discovery and development organization. He has led numerous business development transactions and contributed to the company’s scientific efforts in other therapeutic areas.
Diogo Rau, Senior Vice President and Chief Information and Digital Officer:
Mr. Rau joined Lilly as senior vice president and chief information and digital officer in May 2021. Prior to joining Lilly, Mr. Rau served as senior director of information systems and technology for retail and online stores and most recently served as the top executive of information technology for retail and online stores at Apple, Inc. (Apple). During his 10 years at Apple, he led the development and implementation of the technology supporting the Apple Online Store and Apple retail stores,
including e-commerce platform, mobile point of sale, the Apple Store App, and systems used by store team members. Prior to joining Apple, Mr. Rau was a partner with McKinsey & Company.
In connection with Mr. Smiley’s resignation as an officer on February 9, 2021, the Compensation Committee took action to reduce his 2020 Bonus Plan payout to zero, reduce his 2018-2020 shareholder value award and cancel all his other outstanding equity incentive awards granted in 2019 and 2020. The foregoing terms were agreed to by Mr. Smiley pursuant to his release agreement effective as of February 9, 2021. The material terms of the agreement were previously disclosed in the company’s 2021 proxy statement.
Target Compensation
The information below reflects total compensation at target for named executive officers for 2021. The actual compensation received is summarized below in "Compensation Results."
Rationale for Changes to Named Executive Officer Target Compensation
The Compensation Committee established the 2021 target total compensation for each named executive officer, except Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau, based on the named executive officer's 2020 performance, internal relativity, and peer group data. The Compensation Committee approved target total compensation for Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau upon their promotion or hire, as applicable, based on job scope, market data, experience, and internal relativity.
Base Salary
The following table shows the approved annualized salary for each named executive officer. Mr. Ricks and Dr. Skovronsky did not receive an adjustment to base pay in 2021, and the remaining executive officers' 2021 annualized salaries were effective on the day they assumed their new responsibilities. Each named executive officer's actual base salary earned during 2021 is reflected in the Summary Compensation Table in the column titled “Non-Equity Incentive Plan Compensation.”"Executive Compensation" section of this proxy statement.
| | | | | | | | | | | | | | | | | |
Name2 | 2020 Annual Base Salary Effective March 1, 2020 | 2021 Annual Base Salary Effective March 7, 2021 | Increase |
| Mr. Ricks | $1,500,000 | | $1,500,000 | | 0 | % |
| Ms. Ashkenazi | N/A | 1 | $900,000 | | N/A |
| Dr. Skovronsky | $1,000,000 | | $1,000,000 | | 0 | % |
| Mr. Van Naarden | N/A | 1 | $675,000 | | N/A |
| Mr. Rau | N/A | 1 | $670,000 | | N/A |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 For the period beginning on January 1, 2021 through February 8, 2021, Mr. Smiley's annual base salary was $1,000,000, and for the transition period beginning on February 9, 2021 through July 31, 2021, Mr. Smiley earned a biweekly base salary of $9,000.
2 To assure grant timing is not manipulated for employee gain, the annual grant date is established in advance byAnnual Cash Bonus Targets
Based on a review of internal relativity, peer group data, and individual performance, the Compensation Committee. Equity awardsCommittee retained the same percent-of-salary bonus target as in 2020 for Mr. Ricks, increased the bonus target for Dr. Skovronsky to new hires100 percent, set Ms. Ashkenazi's bonus target at 100 percent, and other off-cycle grantsset Mr. Van Naarden's and Mr. Rau's bonus targets at 80 percent. Bonus targets are generally effective onshown in the first trading daytable below as a percentage of the following month.each named executive officer’s earnings from base salary in 2021:
| | | | | | | | | | | | | | |
Name3 | 2020 Bonus Target | 2021 Bonus Target |
| Mr. Ricks | 150 | % | | 150 | % | |
| Ms. Ashkenazi | N/A | 1 | 100 | % | |
| Dr. Skovronsky | 95 | % | | 100 | % | |
| Mr. Van Naarden | N/A | 1 | 80 | % | 2 |
| Mr. Rau | N/A | 1 | 80 | % | 2 |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 Mr. Van Naarden's Bonus Plan participation was initiated upon his promotion on September 5, 2021. Prior to his promotion, Mr. Van Naarden participated in the Loxo Bonus Plan, which entitled him to a target bonus equal to 60% of base salary.
3This row shows Pursuant to the possible payoutsterms of his separation agreement with the company, Mr. Smiley was not entitled to an annual bonus for 2017-2019the 2020 and 2021 calendar years. See "Agreement with Former Chief Financial Officer" for additional information.
Equity Incentives - Target Grant Values
For 2021 equity grants, the Compensation Committee set the total target values for named executive officers based on market data, individual performance, award grants ranging from 0 to 150and internal relativity. Named executive officers have 35 percent of target. Thistheir equity target allocated to shareholder value awards and relative value awards, respectively, and 30 percent to performance awards. Total target values for the 2020 and 2021 equity grants to the named executive officers were as follows:
| | | | | | | | | | | | | | |
Name5 | 2020 Annual Equity Grant | 2021 Annual Equity Grant |
| Mr. Ricks | $12,500,000 | | $14,000,000 | |
| Ms. Ashkenazi | N/A | 1 | $2,200,000 | 2 |
| Dr. Skovronsky | $4,100,000 | | $4,700,000 | |
| Mr. Van Naarden | N/A | 1 | $1,300,000 | 3 |
| Mr. Rau | N/A | 1 | N/A | 4 |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 Ms. Ashkenazi’s 2021 award will pay out in January 2020. The grant-date fair value ofwas granted prior to her transition into the performance award reflectsChief Financial Officer role. As such, her 2021 annual equity grant aligns with the probable payout outcome anticipatedterms and conditions associated with her prior position.
3 Mr. Van Naarden was not an executive officer at the time of the 2021 annual equity grant. As such, his 2021 annual equity grant aligns with the terms and conditions associated with his prior position. Mr. Van Naarden also received equity awards of restricted stock units valued at (i) $1,650,000 in February 2021 representing transition equity following Lilly’s acquisition of Loxo Oncology, Inc. and (ii) $1,000,000 in December 2021 associated with Mr. Van Naarden's promotion.
4 Mr. Rau started with Lilly in May 2021, after annual grants were issued; however, he received an equity grant of restricted stock units valued at $4,000,000 upon hire to replace a portion of the outstanding equity he forfeited after leaving Apple and the missed opportunity for a 2021 annual equity award from Lilly.
5 Pursuant to the terms of his separation agreement with the company, Mr. Smiley was not entitled to an annual equity grant in 2021. See "Agreement with Former Chief Financial Officer" for additional information.
Performance Goals for Incentive Programs
Annual Cash Bonus Goals
The Compensation Committee established the company performance targets using the company's 2021 annual operating plan, which was greater thanapproved by the board in 2020. These targets are described below under "Compensation Results."
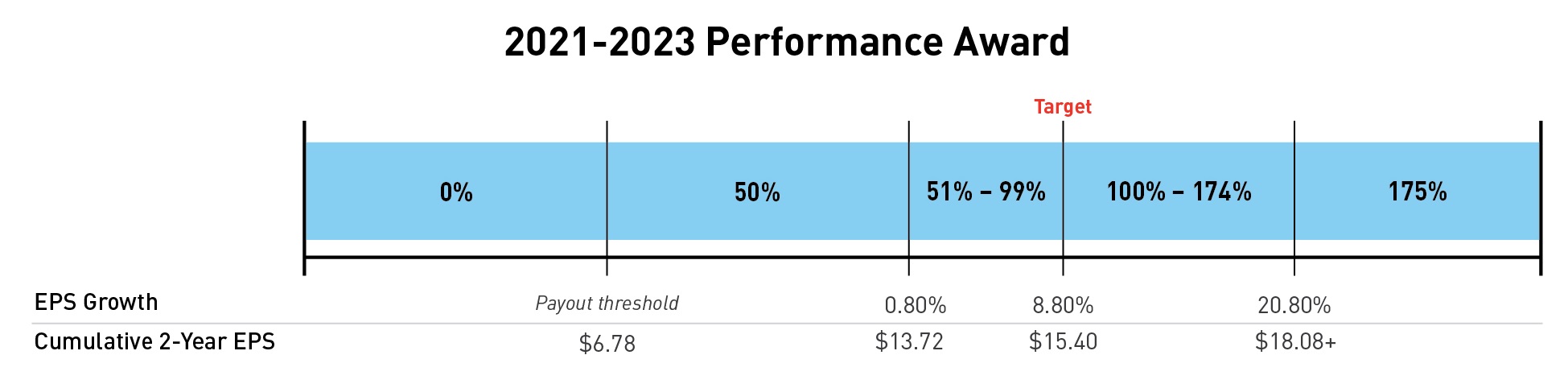
Performance Award
In February 2021, the Compensation Committee established a compounded two-year EPS growth target of 8.8 percent per year based on investment analysts’ EPS growth estimates for our peer group companies at that time. To translate the 8.8 percent per year growth goal into a two-year cumulative EPS target, the Compensation Committee applied the target value.
4 This row showsgrowth to the range2020 adjusted non-GAAP EPS of payouts$6.78 to obtain target 2021 results of $7.38 and then applied the goal growth again to the target 2021 results to obtain target 2022 results of $8.02. The target 2021 and 2022 results were added together to yield a two-year cumulative EPS target of $15.40. Payouts for 2017-2019 shareholder valuethe 2021-2023 performance award grants. This shareholder value award will pay out in January 2020, with payouts rangingcan range from 0 to 150175 percent of target. We measure the fair valuetarget number of shares, as shown below:
Shareholder Value Award
For purposes of establishing the stock price target for the shareholder value award onawards, the grant date using a Monte Carlo simulation model.
5 Mr. Conterno received a special retention grant in recognition of his leadership in delivering the company's strategy and providing continuity in a time of significant transition at the company. The award will vest on December 11, 2021, and it will be forfeited if Mr. Conterno resigns or retires from the company prior to that date.
Outstanding Equity Awards at December 31, 2017
The 2017 closing stockstarting price used to calculate the values in the table below was $84.46.
|
| | | | | | | | | |
| | Stock Awards1 |
| Name | Award | Number of
Shares or
Units of Stock
That Have Not
Vested (#) | Market
Value of
Shares or
Units of
Stock That
Have Not
Vested ($) | Equity Incentive Plan Awards: Number of Unearned Shares, Units, or Other Rights That Have Not Vested (#) | Equity Incentive
Plan Awards:
Market or
Payout Value of Unearned
Shares, Units,
or Other Rights
That Have Not
Vested ($) |
| Mr. Ricks | 2017-2019 SVA | | | | 140,561 |
| 2 | $11,871,782 |
| | 2016-2018 SVA | | | | 57,797 |
| 3 | $4,881,535 |
| | 2017-2019 PA | | | | 69,350 |
| 4 | $5,857,301 |
| | 2016-2018 PA | 12,222 |
| 5 | $1,032,270 | | | |
| | 2015-2017 PA | 21,326 |
| 6 | $1,801,194 | | | |
| Mr. Conterno | 2017-2019 SVA | | | | 41,341 |
| 2 | $3,491,661 |
| | 2016-2018 SVA | | | | 57,797 |
| 3 | $4,881,535 |
| | 2017-2019 PA | | | | 20,397 |
| 4 | $1,722,731 |
| | 2016-2018 PA | 12,222 |
| 5 | $1,032,270 | | | |
| | 2015-2017 PA | 21,326 |
| 6 | $1,801,194 | | | |
| | 2008 RSU Award | 20,000 |
| 7 | $1,689,200 | | | |
| | 2017 RSU Award | 34,615 |
| 8 | $2,923,583 | | | |
| Mr. Rice (retired) | 2017-2019 SVA | | | | 62,838 |
| 2 | $5,307,297 |
| | 2016-2018 SVA | | | | 99,830 |
| 3 | $8,431,642 |
| | 2017-2019 PA | | | | 31,004 |
| 4 | $2,618,598 |
| | 2016-2018 PA | 21,111 |
| 5 | $1,783,035 | | | |
| | 2015-2017 PA | 40,518 |
| 6 | $3,422,150 | | | |
| Dr. Lundberg | 2017-2019 SVA | | | | 59,532 |
| 2 | $5,028,073 |
| | 2016-2018 SVA | | | | 94,576 |
| 3 | $7,987,889 |
| | 2017-2019 PA | | | | 29,372 |
| 4 | $2,480,759 |
| | 2016-2018 PA | 20,000 |
| 5 | $1,689,200 | | | |
| | 2015-2017 PA | 36,252 |
| 6 | $3,061,844 | | | |
| Mr. Harrington | 2017-2019 SVA | | | | 38,034 |
| 2 | $3,212,352 |
| | 2016-2018 SVA | | | | 60,423 |
| 3 | $5,103,327 |
| | 2017-2019 PA | | | | 18,765 |
| 4 | $1,584,892 |
| | 2016-2018 PA | 12,778 |
| 5 | $1,079,230 | | | |
| | 2015-2017 PA | 24,524 |
| 6 | $2,071,297 | | | |
1 The chart no longer includes stock option awards because the company has not awarded stock options to employees since 2006 and there are no outstanding stock option awards.
2 Shareholder value awards granted for the 2017-2019 performance period will vest on December 31, 2019. The number of shares reported reflects the maximum payout, which will be made if$152.16 per share, the average closing stock price for all trading days in November and December 20182020. The target share price was established using the expected annual rate of return for large-cap companies (8 percent), less an assumed Lilly dividend yield (2.23 percent). To determine payout, the ending price will be the average closing price of company stock for all trading days in November and December 2023. The award is designed to deliver no payout to executive officers if the shareholder return (including projected dividends) is zero or negative. Possible payouts based on share price ranges are illustrated in the grid below.
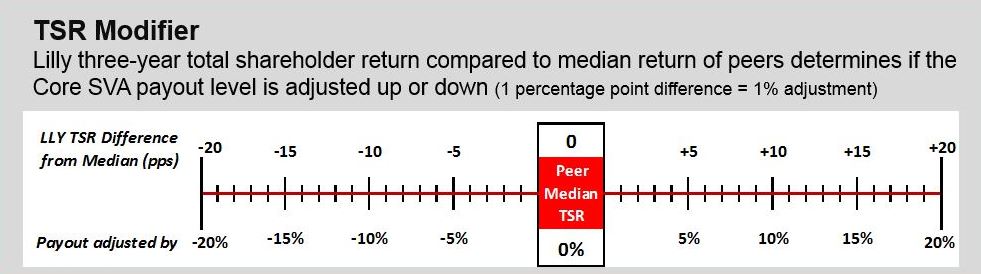
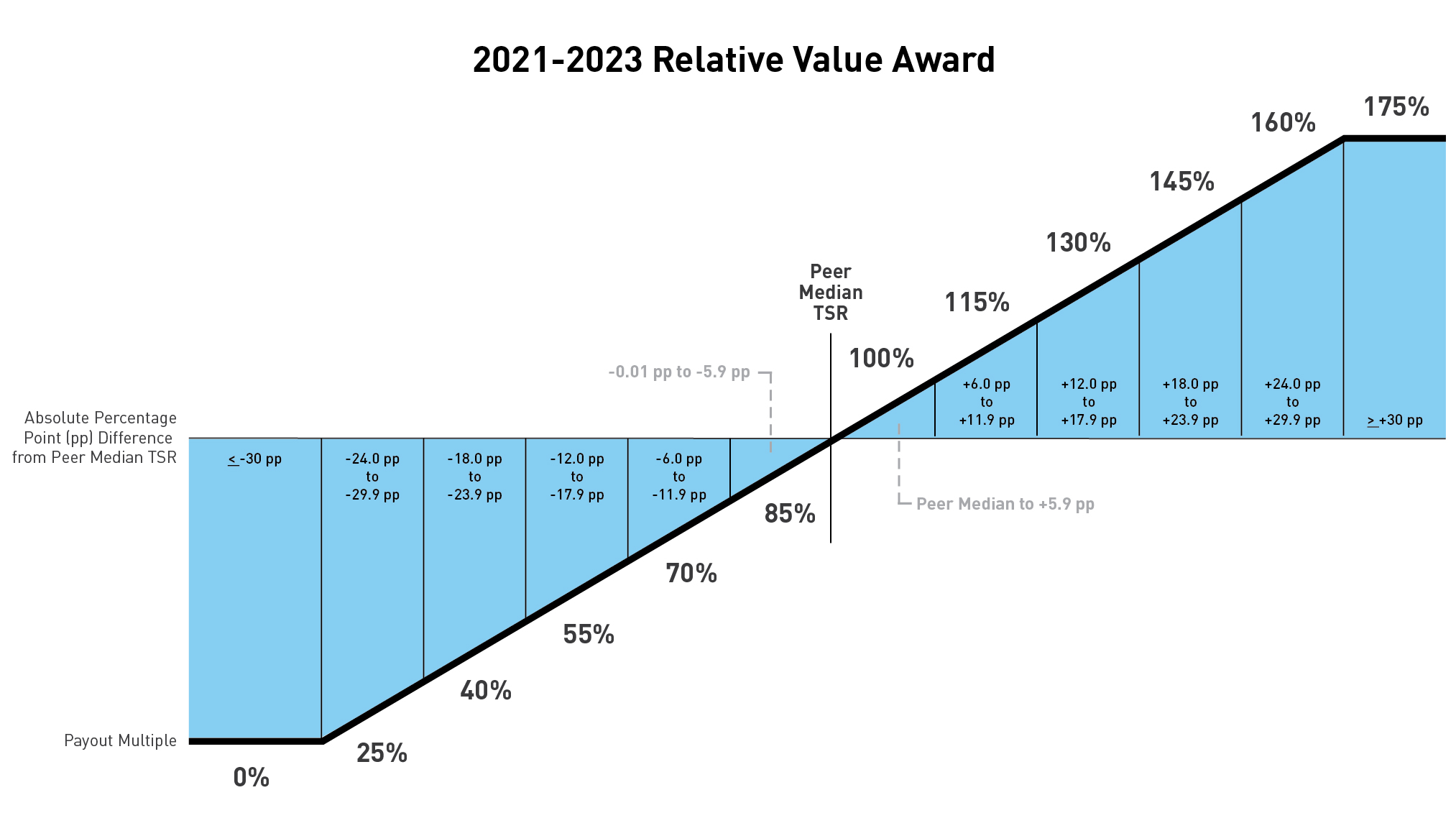
Relative Value Award
The relative value award is based on the three-year Lilly TSR performance compared to our peer group. To determine payout, the TSR performance is calculated for Lilly and its peers. This calculation compares the average closing price of each company’s stock for all trading days in November and December 2020 to the average closing price of each company’s stock for all trading days in November and December 2023, assuming reinvestment of dividends, to obtain the TSR for each company. The median TSR for the peer companies is then subtracted from Lilly’s TSR to determine what payout has been earned. For example, if Lilly’s TSR was 55 percent over $101.79. Actual payouts may varythe three-year performance period and the median peer company performance was 41 percent, Lilly would have outperformed by 14 percentage points (55 percent - 41 percent). This outperformance would have resulted in a 130 percent payout based on the payout ranges depicted below.
Compensation Results
The information in this section reflects the amounts paid to named executive officers under the Bonus Plan and for equity awards granted in prior years for which the relevant performance period ended in 2021.
Lilly Performance
In 2021, we exceeded our revenue target and nearly achieved our EPS target. We also made significant pipeline advances. Key pipeline highlights include U.S. approval of a new indication for Verzenio® for certain people with high-risk, early breast cancer; the Emergency Use Authorization for the administration of bamlanivimab and etesevimab together for the treatment
of COVID-19 in patients at high risk for hospitalization; the regulatory submission for tirzepatide for type 2 diabetes in the U.S. to which Lilly applied a priority review voucher, as well as submission in Europe and Japan; positive topline results from donanemab’s TRAILBLAZER-ALZ, a Phase II study evaluating the efficacy and safety of donanemab compared to placebo in patients with early symptomatic Alzheimer’s disease; updated data for pirtobrutinib, from the LOXO-305 BRUIN Phase I/II clinical trial in mantle cell lymphoma and other non-Hodgkin lymphomas, as well as in chronic lymphocytic leukemia and small lymphocytic lymphoma, at the 2021 American Society of Hematology Annual Meeting; positive topline results from mirikizumab’s Phase III LUCENT-1 and 2 studies evaluating the efficacy and safety of mirikizumab compared to placebo for the treatment of patients with moderately-to-severely active ulcerative colitis; and positive topline results from three studies of lebrikizumab’s Phase III program evaluating the efficacy and safety of lebrikizumab in people with moderate-to-severe atopic dermatitis. See "Proxy Statement Summary—Highlights of 2021 Performance."
By the end of 2021, Lilly exceeded its two-year EPS growth target for the performance award and our three-year stock price growth target for the shareholder value award. The company began granting relative value awards in 2020; therefore, no relative value awards were granted or earned for the 2019-2021 performance period. The discussion below details the measures used in each program, what the performance goal was to obtain target performance, how performance outcomes were assessed and what the Compensation Committee approved as the final payout multiple.
Bonus Plan
The company utilized revenue, EPS, and pipeline progress to incent the achievement of 2021 company objectives. Each measure contributes to the final payout multiple on a weighted basis: revenue (25 percent), EPS (50 percent), and pipeline progress (25 percent). Each performance measure is assessed a payout multiple of 0 to 200 percent.
For purposes of the bonus, the Compensation Committee adjusted revenue and non-GAAP EPS to exclude estimated savings from certain discrete and unplanned performance items from the bonus plan multiple. The Science and Technology Committee's assessment of the company's product pipeline achievements is detailed below:
| | | | | | | | |
| Activity | Objective | Achievement |
| Approvals | 2 new drug first approvals
14-15 other approvals | 0 new drug first approvals
18 other approvals |
| Potential new drug Phase III starts | 3 | 2 |
| Potential new drug Phase I starts | 16-17 | 12 |
| Potential new indication or line extension Phase III starts | 5 | 9 |
| Plan Boldly | Meet industry benchmark for speed of development | Exceeded industry benchmark for speed of development |
| Deliver to Launch | Meet planned project timelines | Met planned project timelines |
| Qualitative Assessment | Assessment of the chief scientific and medical officer's evaluation of performance against strategic objectives |
Based on the recommendation of the Science and Technology Committee, the Compensation Committee approved a pipeline multiple of 1.06.
The company's performance compared to targets as well as the resulting bonus multiple, is illustrated below:
For additional information on financial results, see Appendix A, "Summary of Adjustments Related to the Annual Cash Bonus and Performance Award."
When combined, the revenue, EPS, and pipeline multiples yielded a bonus multiple of 1.10.
Loxo Bonus
The Loxo Bonus Plan Committee assessed the level of oncology research and development goal achievement compared to the preclinical, clinical, regulatory actions, external innovation and scientific disclosure milestones established at the beginning of the year. On a payout continuum between 0 – 200 percent, the Loxo Bonus Plan Committee determined the Additional Cash Award portion of the Loxo Bonus would pay at 200 percent. To determine the final Loxo Bonus payout multiple, the Loxo Bonus Plan Committee added the Bonus Plan multiple to the Additional Cash Award multiple to yield a total payout of 310 percent.
Bonus Payments
The 2021 bonuses paid to the applicable named executive officers under the Bonus Plan were as follows:
| | | | | | | | |
Name3 | 2021 Bonus ($) |
| Mr. Ricks | $2,475,000 | |
| Ms. Ashkenazi | $907,816 | |
| Dr. Skovronsky | $1,100,000 | |
Mr. Van Naarden1 | $1,029,507 | |
Mr. Rau2 | $362,831 | |
1 A portion of the 2021 bonus payable to Mr. Van Naarden was attributable to achievement of the performance goals under the Loxo Bonus Plan. The 2021 Loxo Bonus Plan achievement was 310%. The amount of the 2021 bonus paid to Mr. Van Naarden in the table above reflects his achievement under both the Loxo Bonus Plan and the Bonus Plan, in each case, pro-rated to reflect the period in 2021 during which he was a participant in the respective plan. Mr. Van Naarden also received a one-time cash payment of $218,715 in lieu of compensation and benefits he was no longer eligible to receive following his transition to his new role, including compensation for the difference between the bonus he would have earned under the Loxo Bonus Plan and the Lilly Bonus Plan, and changes in his expected out-of-pocket costs for healthcare expenses.
2 Mr. Rau joined Lilly in May 2021, and the table reflects the pro-rated bonus paid for the portion of the year for which he was employed.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley was not entitled to an annual bonus for calendar year 2021. See "Agreement with Former Chief Financial Officer" for additional Information.
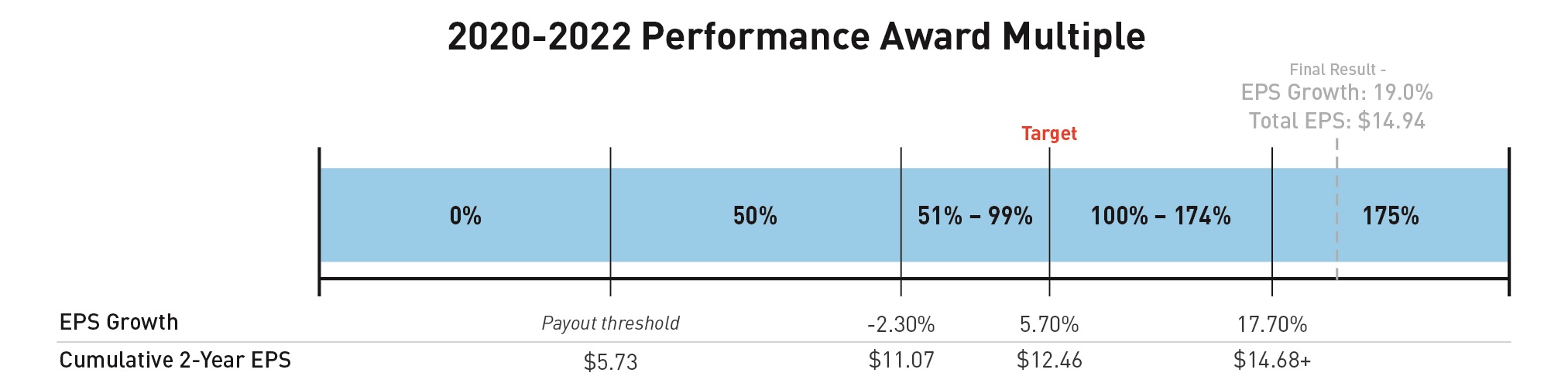
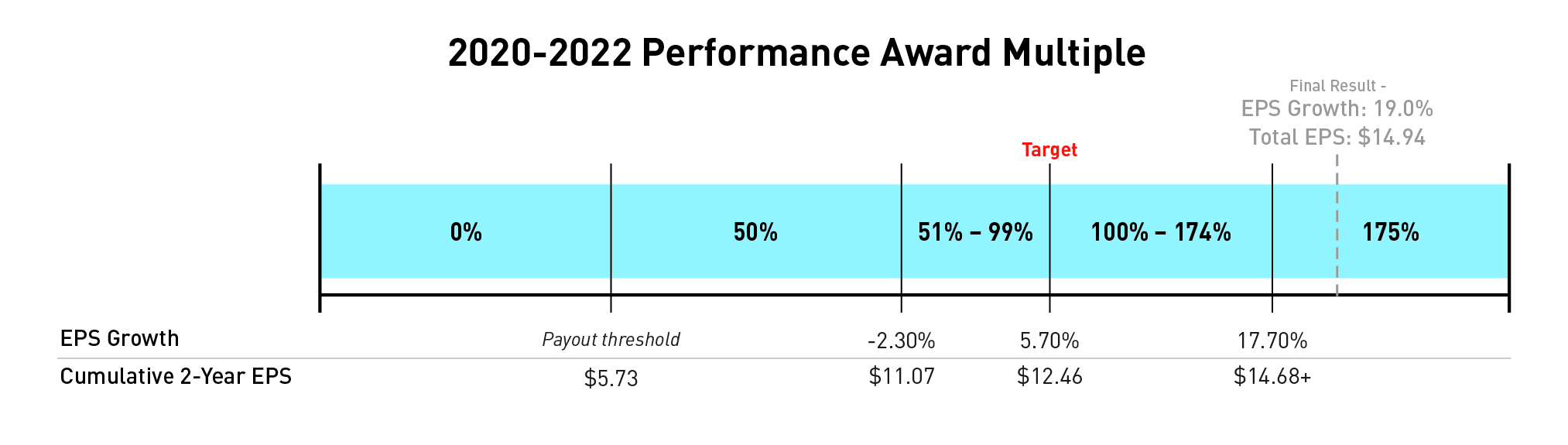
2020-2022 Performance Awards
The target cumulative EPS for the 2020-2022 performance award was originally set in the first quarter of 2020, reflecting expected peer group EPS growth of 5.7 percent each year and a total target EPS of $13.13 over the two-year performance period. In December 2020, the Compensation Committee updated the award to align with Lilly's decision to adjust EPS results to remove the impact of gains and losses on investments in equity securities from its non-GAAP measures and compensation program outcomes. The updated award retained the Compensation Committee's decision to establish a compounded two-year EPS growth target of 5.7 percent per year, but the growth was applied to an adjusted 2019 non-GAAP EPS of $5.73 that excluded gains and losses on investments in equity securities. Using this methodology, the adjusted EPS target became $12.46 for the two-year performance period of 2020-2021. The company's actual adjusted EPS over the performance period was $14.94 representing a growth rate of 19.0 percent.
For Mr. Ricks and Dr. Skovronsky, shares earned for the 2020-2021 performance period are subject to an additional 13-month service-vesting period and are shown in the table below as restricted stock units. Because she was not an executive officer when the award was granted, Ms. Ashkenazi's 2020-2021 performance award was paid in shares of Lilly common stock and not subject to the additional service-vesting period. Mr. Van Naarden was an employee of Loxo Oncology, Inc. and did not receive a performance award grant in 2020. Mr. Rau joined Lilly in May 2021, so he did not receive a performance award grant in 2020.
| | | | | | | | | | | |
Name3 | Target Shares | Shares Earned | RSUs |
| | | |
| Mr. Ricks | 27,306 | N/A | 47,786 |
| Ms. Ashkenazi | 2,731 | 4,779 | N/A |
| Dr. Skovronsky | 8,957 | N/A | 15,675 |
Mr. Van Naarden1 | N/A | N/A | N/A |
Mr. Rau2 | N/A | N/A | N/A |
1 Mr. Van Naarden was an employee of Loxo Oncology, Inc. and did not receive a performance award grant in 2020.
2 Mr. Rau joined Lilly in May 2021, so he did not receive a performance award grant in 2020.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley forfeited his entire 2020-2022 performance award. See "Agreement with Former Chief Financial Officer" for additional information.
2019-2021 Shareholder Value Award
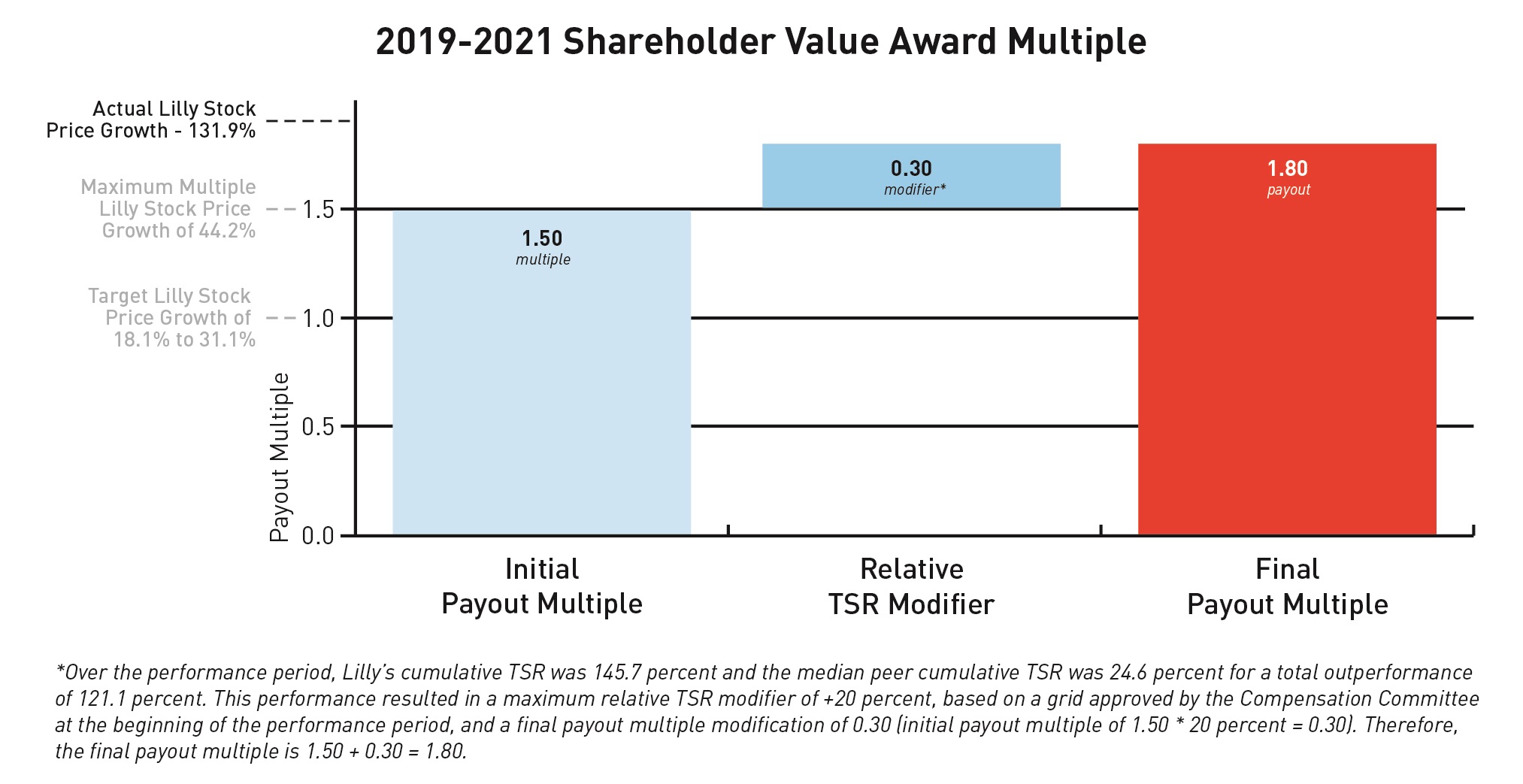
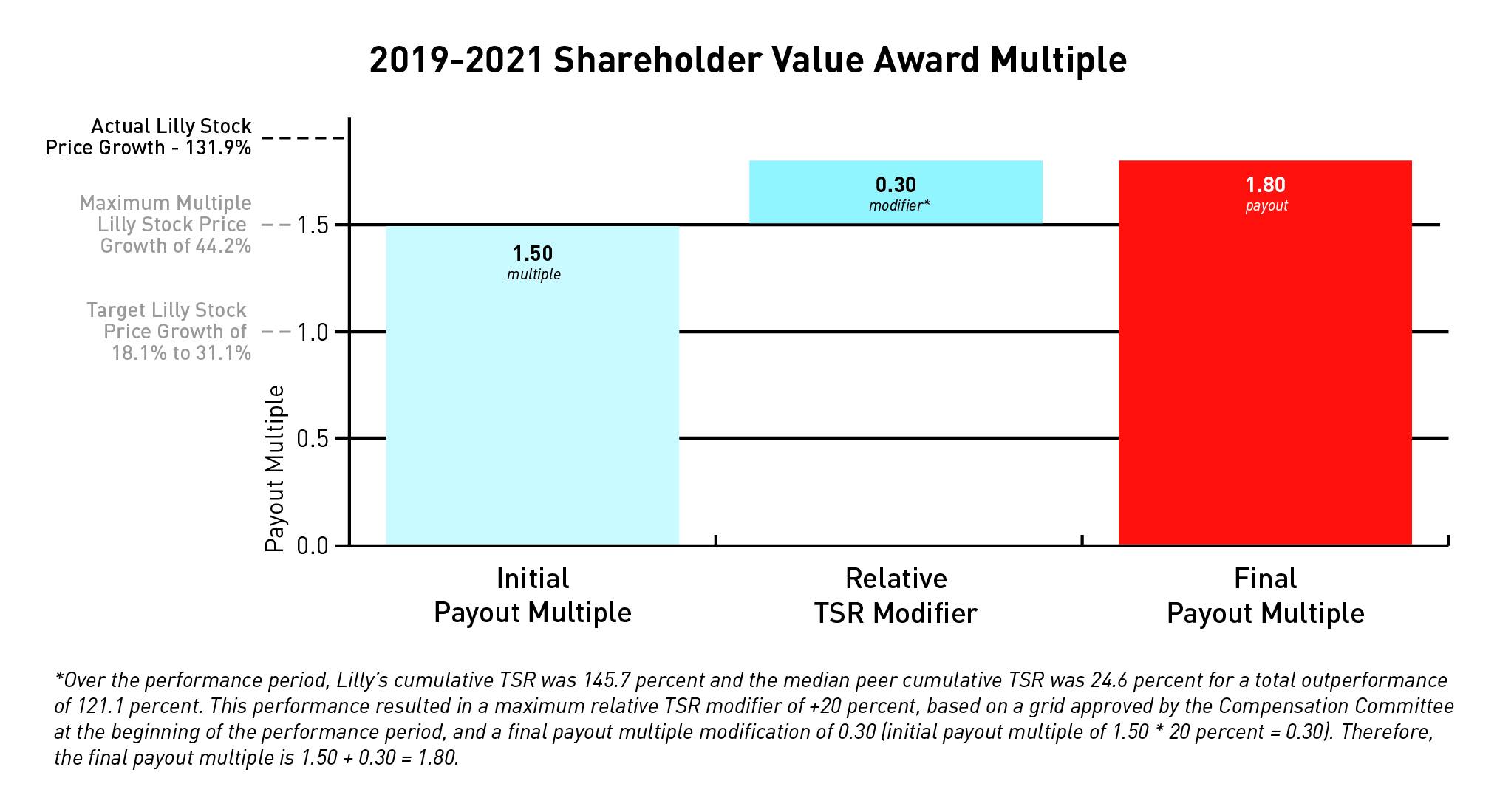
The target stock price range of $132.73 to $147.37 (18.1 percent to 31.1 percent total stock price growth) for the 2019-2021 shareholder value award was set in 2019 based on a beginning stock price of $112.38, which was the average closing price for Lilly stock for all trading days in November and December 2018. The ending stock price of $260.57 represents stock price growth of 131.9 percent over the relevant three-year period resulting in a payout multiple of 1.50.
The relative TSR modifier applies to those individuals who were executive officers when the award was granted. The cumulative TSR median for the company’s peer group was 24.6 percent, and Lilly’s TSR over the same period was 145.7 percent. Given this positive relative performance, Lilly's relative TSR was 121.1 percentage points above the peer group
median resulting in a maximum award payout of 180 percent of target. Net shares from anytarget (SVA payout must be heldmultiple of 150 percent multiplied by executive officers for a minimum of one year. Had the performance period ended December 31, 2017, the payout would have been at target.1.2 modifier = 180 percent final payout).
3 Shareholder value awards granted for the 2016-2018 performance period will vest on December 31, 2018. The number of shares reported reflects the maximum payout, which will be made if the average closing stock price in November and December 2018 is over $119.58. Actual payouts may vary from 0paid to 180 percenteach of target. Net shares from any payout must be held byour named executive officers for a minimum of one year. Had the 2019-2021 performance period ended December 31, 2017, the payout would have been 50 percent of target.were as follows:
| | | | | | | | |
Name3 | Target Shares | Shares Paid Out |
| Mr. Ricks | 68,382 | 123,088 |
Ms. Ashkenazi1 | 3,633 | 5,450 |
| Dr. Skovronsky | 22,794 | 41,029 |
Mr. Van Naarden2 | N/A | N/A |
Mr. Rau2 | N/A | N/A |
4 1 This number represents the maximumThe TSR modifier did not apply to Ms. Ashkenazi’s 2019-2021 shareholder value of performance award shares that could pay out for the 2017-2018 performance period, provided performance goals are met. Once the combined cumulative EPS result and associated payout level is determinedpayouts since she was not an executive officer at the endtime of the performance period, the associated number of shares are restricted stock units vesting in February 2020. Actual payouts may vary from 0 to 150 percent of target. Thegrant.
2 Mr. Van Naarden was an employee of Loxo Oncology, Inc. in 2019 and so he did not receive a shareholder value award grant in 2019. Mr. Rau joined Lilly in May 2021, so he did not receive a shareholder value award grant in 2019.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley forfeited his entire 2019-2021 shareholder value award. See "Agreement with Former Chief Financial Officer" for additional information.
Other Compensation Practices and Information
Employee Benefits
The company offers core employee benefits coverage to:
•provide our workforce with a reasonable level of financial support in the event of illness or injury
•provide post-retirement income
•enhance productivity and job satisfaction through benefit programs that focus on overall well-being.
The benefit programs available to executive officers are offered to all eligible U.S. employees and include benefits such as medical and dental coverage, disability insurance, and life insurance. In addition, the 401(k) Plan and The Lilly Retirement Plan (the Retirement Plan) are intended to provide U.S. employees a reasonable level of retirement income reflecting employees’ careers with the company. To the extent that any employee’s calculated retirement benefit exceeds Internal Revenue Service (IRS) limits for amounts that can be paid through a qualified plan, the company also offers a nonqualified pension plan and a nonqualified savings plan. These plans provide only the difference between the calculated benefits and the applicable IRS limits, and the formula is the same for all participating employees. The cost of employee benefits is partially borne by the employee, including each executive officer.
Perquisites
The company provides limited perquisites to executive officers. In response to the COVID-19 pandemic, the company considered various actions to promote the health and safety of its employees, including its named executive officers, recognizing that the company’s important objectives during this critical time would be significantly disadvantaged without
numberthe full services of shares recorded in the table reflects the payout if the combined cumulative EPS for 2017 and 2018 is at least $8.51.
5 The performance period ending 2017 for the 2016-2018 performance award resulted in a restricted stock unit for 150 percentits employees. As part of target shares. The restricted stock units will vest in February 2019.
6 Restricted stock units vested in February 2018 from the 2015-2017 performance award.
7 This grant was made in 2008 before Mr. Conterno became an executive officer. This award was granted outside of the normal annual cycle and will vest on May 1, 2018.
8 Mr. Conterno received a special retention grant in recognition of his leadership in delivering the company's strategy and providing continuity in a time of significant transition at the company. The award will vest on December 11, 2021, and it will be forfeited if Mr. Conterno resigns or retires from the company prior to that date.
Options Exercised and Stock Vested in 2017
|
| | | | | | | |
| | Option Awards1 | | Stock Awards |
| Name | Number of Shares
Acquired on Exercise (#) | Value Realized
on Exercise ($) | | Number of Shares
Acquired on Vesting (#) | Value Realized
on Vesting ($)2 |
| Mr. Ricks | 0 | $0 | | 10,244 |
| 3 | $789,095 |
| | 22,507 |
| 4 | $1,971,613 |
| | 5,496 |
| 5 | $468,919 |
| Mr. Conterno | 0 | $0 | | 10,244 |
| 3 | $789,095 |
| | 22,507 |
| 4 | $1,971,613 |
| Mr. Rice (retired) | 0 | $0 | | 19,463 |
| 3 | $1,499,235 |
| | 42,764 |
| 4 | $3,746,126 |
| Dr. Lundberg | 0 | $0 | | 15,366 |
| 3 | $1,183,643 |
| | 38,262 |
| 4 | $3,351,751 |
| Mr. Harrington | 0 | $0 | | 9,732 |
| 3 | $749,656 |
| | 25,883 |
| 4 | $2,267,351 |
1 The chart no longer includes stock option awards becausethis process, the company has not awarded stock options to employees since 2006 and there are no outstanding stock option awards.
2 Amounts reflect the market valueencouraged Mr. Ricks’ personal use of the stock on the day the stock vested.
3 Restricted stock units resulting from the 2014-2016 performance award that vested in February 2017.
4 Payoutcorporate aircraft (up to a maximum incremental cost of $60,000) as a means to (i) increase his time available for company business; and (ii) protect his health and safety. The incremental cost of personal use of the 2015-2017 shareholder value award at 100 percent of target.
5 This grant was made in 2007 before Mr. Ricks became an executive officer. This award was granted outside of the normal annual cycle.
Retirement Benefits
We provide retirement income to eligible U.S. employees, including executive officers, through the following plans:
The 401(k) Plan,corporate aircraft is included as a defined contribution plan qualified under Sections 401(a) and 401(k) of the Internal Revenue Code. Participants may elect to contribute a portion of their base salary to the plan, and the company provides matching contributions on employees’ contributions up to 6 percent of base salary up to IRS limits. The employee contributions, company contributions, and earnings thereon are paid out in accordance with elections made by the participant. See the "All Other Compensation" columnperquisite in the Summary Compensation Table for information about company contributions under the 401(k) Plan for the named executive officers.heading "All Other Compensation."
The Retirement Plan, a tax-qualified defined benefit plan that provides monthly benefits to retirees. See the Pension Benefits in 2017 table below for additional information about the value of these pension benefits.
Sections 401 and 415 of the Internal Revenue Code generally limit the amount of annual pension that can be paid from a tax-qualified plan ($270,000 in 2017 and $275,000 in 2018) as well as the amount of annual earnings that can be used to calculate a pension benefit. However, since 1975 the company has maintained a nonqualified pension plan that pays retirees the difference between the amount payable under the Retirement Plan and the amount they would have received without the Internal Revenue Code limits. The nonqualified pension plan is unfunded and subject to forfeiture in the event of bankruptcy. Likewise the company maintains a nonqualified savings plan that allows participants to contribute up to 6 percent of base salary exceeding the IRS limit. The company matches these contributions as described in the 401(k) Plan. For more information, see footnote 3 to the NonqualifiedLilly Deferred Compensation in 2017 table.Plan
The following table shows benefits that the named executive officers have accrued under the Retirement Plan and the nonqualified pension plan.
Pension Benefits in 2017
|
| | | | |
| Name | Plan | Number of Years of Credited Service | Present Value of
Accumulated Benefit ($) 1 | Payments During
Last Fiscal Year ($) |
| Mr. Ricks | retirement plan (pre-2010) | 14 | $570,749 | |
| | retirement plan (post-2009) | 8 | $203,352 | |
| | nonqualified plan (pre-2010) | 14 | $2,816,826 | |
| | nonqualified plan (post-2009) | 8 | $980,129 | |
| | total | | $4,571,056 | $0 |
| Mr. Conterno | retirement plan (pre-2010) | 17 | $849,574 | |
| | retirement plan (post-2009) | 8 | $211,141 | |
| | nonqualified plan (pre-2010) | 17 | $4,074,998 | |
| | nonqualified plan (post-2009) | 8 | $965,590 | |
| | total | | $6,101,303 | $0 |
Mr. Rice (retired)2 | retirement plan (pre-2010) | 20 | $990,838 | |
| | retirement plan (post-2009) | 8 | $219,228 | |
| | nonqualified plan (pre-2010) | 20 | $8,538,391 | |
| | nonqualified plan (post-2009) | 8 | $1,775,668 | |
| | total | | $11,524,125 | $0 |
| Dr. Lundberg | retirement plan (post-2009) | 8 | $344,168 | |
| | nonqualified plan (post-2009) | 8 | $2,660,445 | |
| | total | | $3,004,613 | $0 |
| Mr. Harrington | retirement plan (pre-2010) | 18 | $933,412 | |
| | retirement plan (post-2009) | 8 | $236,344 | |
| | nonqualified plan (pre-2010) | 18 | $4,530,371 | |
| | nonqualified plan (post-2009) | 8 | $1,106,712 | |
| | total | | $6,806,839 | $0 |
1 The following standard actuarial assumptions were used to calculate the present valueMembers of each individual’s accumulated pension benefit:
|
| |
Discount rate: | 3.83 percent for the qualified plan and 3.70 percent for non-qualified plan |
Mortality (post-retirement decrement only): | RP2006 with generational projection using Scale MP2017 |
Pre-2010 joint and survivor benefit (% of pension): | 50% until age 62; 25% thereafter |
Post-2009 benefit payment form: | life annuity |
2 Mr. Rice retired with full retirement benefits under the old plan formula (pre-2010 benefits) and qualified for early retirement under the new plan formula (post-2009 benefits) as described below.
The Retirement Plan benefits shown in the table are net present values. The benefits are not payable as a lump sum; they are generally paid as a monthly annuity for the life of the retiree and, if elected, any qualifying survivor. The annual benefit under the retirement plan is calculated using years of service and the average of the annual earnings (salary plus bonus) for the highest 5 out of the last 10 calendar years of service (final average earnings).
Post-2009 Plan Information:Following amendment of our Retirement Plan formulas, employees hired on or after February 1, 2008, have accrued retirement benefits only under the new plan formula. Employees hired before that date have accrued benefits under both the old and new plan formulas. All eligible employees, including those hired on or after February 1, 2008, can retire at age 65 with at least five years of service and receive an unreduced benefit. The annual benefit under the new plan formula is equal to 1.2 percent of final average earnings multiplied by years of service. Early retirement benefits under this plan formula are reduced 6 percent for each year under age 65. Transition benefits were afforded to employees with 50 points (age plus service) or more as of December 31, 2009. These benefits were intended to ease the transition to the new retirement formula for those employees who were closer to retirement or had been with the company longer at the time the plan was changed. For the transition group, early retirement benefits are reduced 3 percent for each year from age 65 to age 60 and 6 percent for each year under age 60. All named executive officers except Dr. Lundberg are in this transition group.
Pre-2010 Plan Information:Employees hired prior to February 1, 2008, accrued benefits under both plan formulas. For these employees, benefits that accrued before January 1, 2010, were calculated under the old plan formula. The amount of the benefit is calculated using actual years of service through December 31, 2009, while total years of service is used to determine eligibility and early retirement reductions. The benefit amount is increased (but not decreased) proportionately, based on final average earnings at termination compared to final average earnings at December 31, 2009. Full retirement benefits are earned by employees with 90 or more points (the sum of his or her age plus years of service). Employees electing early retirement receive reduced benefits as described below:
The benefit for employees with between 80 and 90 points is reduced by 3 percent for each year under 90 points or age 62.
The benefit for employees who have fewer than 80 points, but who reached age 55 and have at least 10 years of service, is reduced as described above and is further reduced by 6 percent for each year under 80 points or age 65.
Nonqualified Deferred Compensation in 2017
|
| | | | | | | | | | | | | | | |
| Name | Plan | Executive
Contributions in
Last Fiscal Year
($)1 | Registrant
Contributions in
Last Fiscal Year
($)2 | Aggregate Earnings in Last Fiscal Year ($) | Aggregate Withdrawals/ Distributions in Last Fiscal Year ($) | Aggregate
Balance at Last
Fiscal Year End
($)3 |
| Mr. Ricks | nonqualified savings |
| $67,800 |
| | $67,800 |
| $79,349 |
| | $0 |
| $621,637 |
| |
| | deferred compensation |
| $0 |
| | |
| $0 |
| | |
| $0 |
| |
| | total |
| $67,800 |
| | $67,800 |
| $79,349 |
| | $0 |
| $621,637 |
| |
| Mr. Conterno | nonqualified savings |
| $29,520 |
| | $29,520 |
| $133,317 |
| | $0 |
| $902,670 |
| |
| | deferred compensation |
| $100,000 |
| | |
| $40,169 |
| | |
| $1,290,943 |
| |
| | total |
| $129,520 |
| | $29,520 |
| $173,486 |
| | $0 |
| $2,193,613 |
| |
| Mr. Rice (retired) | nonqualified savings |
| $49,148 |
| | $49,148 |
| $313,241 |
| | $0 |
| $1,908,141 |
| |
| | deferred compensation |
| $0 |
| | |
| $0 |
| | |
| $0 |
| |
| | total |
| $49,148 |
| | $49,148 |
| $313,241 |
| | $0 |
| $1,908,141 |
| |
| Dr. Lundberg | nonqualified savings |
| $45,279 |
| | $45,279 |
| $47,378 |
| | $0 |
| $908,217 |
| |
| | deferred compensation |
| $0 |
| | |
| $0 |
| | |
| $0 |
| |
| | total |
| $45,279 |
| | $45,279 |
| $47,378 |
| | $0 |
| $908,217 |
| |
| Mr. Harrington | nonqualified savings |
| $35,168 |
| | $35,168 |
| $66,068 |
| | $0 |
| $519,523 |
| |
| | deferred compensation |
| $0 |
| | |
| $5,704 |
| | |
| $180,677 |
| |
| | total |
| $35,168 |
| | $35,168 |
| $71,772 |
| | $0 |
| $700,199 |
| |
1 The amounts in this column are also included in the Summary Compensation Table, in the “Salary” column (nonqualified savings) or the “Non-Equity Incentive Plan Compensation” column (deferred compensation).
2 The amounts in this column are also included in the Summary Compensation Table, in the “All Other Compensation” column as a portion of the savings plan match.
3 Of the totals in this column, the following amounts have previously been reported in the Summary Compensation Table for this year and for previous years:
|
| | | | | | |
| Name | 2017 ($) | Previous Years ($) | Total ($) |
| Mr. Ricks | $135,600 | | N/A | | $135,600 | |
| Mr. Conterno | $159,040 | | $760,600 | | $919,640 | |
| Mr. Rice (retired) | $98,296 | | $895,298 | | $993,594 | |
| Dr. Lundberg | $90,557 | | $616,819 | | $707,376 | |
| Mr. Harrington | $70,336 | | $276,588 | | $346,924 | |
The Nonqualified Deferred Compensation in 2017 table above shows information about two company programs: the nonqualified savings plan and the Deferred Compensation Plan. The nonqualified savings plan is designed to allow each employee to contribute up to 6 percent of his or her base salary and receive a company match, beyond the contribution limits prescribed by the IRS with regard to 401(k) plans. This plan is administered in the same manner as the 401(k) Plan, with the same participation and investment elections. Executive officers and other U.S. executivessenior management may also defer receipt of allpart or partall of their cash compensation under theThe Lilly Deferred Compensation Plan. AmountsPlan (Deferred Compensation Plan), which allows executives to save for retirement in a tax-effective way at minimal cost to the company. Under this unfunded plan, amounts deferred by executives under this planthe executive are credited withat an interest atrate of 120 percent of the applicable federal long-term rate, as establisheddescribed in more detail following the preceding December by the U.S. Treasury Department under Section 1274(d) of the Internal Revenue Code with monthly compounding, which was 2.7 percent for 2017 and is 3.1 percent for 2018. Participants may elect to receive the funds"Nonqualified Deferred Compensation in a lump sum or in up to 10 annual installments following termination of employment, but may not make withdrawals while employed by the company, except2021" table.
Severance Benefits
Except in the eventcase of hardship as approved by the Compensation Committee. All deferral elections and associated distribution schedules are irrevocable. Both plans are unfunded and subject to forfeiture in the event of bankruptcy.
Payments Upon Termination or Change in Control (as of December 31, 2017)
The following table describes the potential payments and benefits under the company’s compensation and benefit plans and arrangements to which the named executive officers would be entitled upon termination of employment. Except for certain terminations following a change in control of the company, as described below, there are no agreements, arrangements, or plans that entitle namedthe company is generally not obligated to pay severance to executive officers to severance, perquisites, or other enhanced benefits upon termination of their employment. Any agreement to provideemployment; any such payments or benefits to a terminating executive officer (other than following a change in control) would beare at the discretion of the Compensation Committee.
|
| | | | | |
| | | Cash
Severance
Payment 1 | Continuation
of Medical /
Welfare
Benefits
(present
value) 2 | Value of
Acceleration
of Equity
Awards | Total
Termination
Benefits |
| Mr. Ricks |
|
|
|
|
| • | Voluntary retirement | $0 | $0 | $0 | $0 |
| • | Involuntary retirement or termination | $0 | $0 | $0 | $0 |
| • | Involuntary or good reason termination after change in control | $7,000,000 | $39,903 | $6,206,019 | $13,245,922 |
| Mr. Conterno |
|
|
|
|
| • | Voluntary termination | $0 | $0 | $0 | $0 |
| • | Involuntary retirement or termination | $0 | $0 | $0 | $0 |
| • | Involuntary or good reason termination after change in control | $2,765,160 | $296,844 | $4,347,980 | $7,409,984 |
| Mr. Rice (retired) |
|
|
|
|
| • | Voluntary retirement | $0 | $0 | $0 | $0 |
| • | Involuntary retirement or termination | $0 | $0 | $0 | $0 |
| • | Involuntary or good reason termination after change in control | $4,370,800 | $45,916 | $4,159,289 | $8,576,005 |
| Dr. Lundberg |
|
|
|
|
| • | Voluntary retirement | $0 | $0 | $0 | $0 |
| • | Involuntary retirement or termination | $0 | $0 | $0 | $0 |
| • | Involuntary or good reason termination after change in control | $4,112,000 | $63,472 | $3,938,307 | $8,113,779 |
| Mr. Harrington |
|
|
|
|
| • | Voluntary termination | $0 | $0 | $0 | $0 |
| • | Involuntary retirement or termination | $0 | $0 | $0 | $0 |
| • | Involuntary or good reason termination after change in control | $3,097,080 | $45,916 | $2,517,449 | $5,660,445 |
1 See “Change-in-Control Severance Pay Plan—Cash Severance Payment” below.
2 See “Accrued Pay and Regular Retirement Benefits” and “Change-in-Control Severance Pay Plan—Continuation of medical and welfare benefits” below.
Accrued Pay and Regular Retirement Benefits.The amounts shown in the table above do not include certain payments and benefits to the extent they are provided on a non-discriminatory basis to salaried employees generally upon termination of employment. These include:
accrued salary and vacation pay.
regular pension benefits under the Retirement Plan and the nonqualified pension plan. See “Retirement Benefits” above.
welfare benefits provided to all U.S. retirees, including retiree medical and dental insurance. The amounts shown in the table above as “Continuation of Medical / Welfare Benefits” are explained below.
distributions of plan balances under the 401(k) Plan, the nonqualified savings plan, and the Deferred Compensation Plan. See the narrative following the Nonqualified Deferred Compensation in 2017 table for information about these plans.
Deferred Compensation. The amounts shown in the table do not include distributions of plan balances under the deferred compensation plan. Those balances are shown in the Nonqualified Deferred Compensation in 2017 table.
Death and Disability. A termination of employment due to death or disability does not entitle named executive officers to any payments or benefits that are not available to U.S. salaried employees generally.
Termination for Cause. Executives terminated for cause receive no severance or enhanced benefits and forfeit any unvested equity grants.
Change-in-Control Severance Pay Plan. As described in the CD&A under “Severance Benefits,” the company maintains ahas adopted change-in-control severance pay planplans for nearly all employees, including the named executive officers. The change-in-control plan definesplans are intended to preserve employee morale and productivity and encourage retention in the face of the disruptive impact of an actual or rumored change in control. In addition, the plans are intended to align executive and shareholder interests by enabling executives to evaluate corporate transactions that may be in the best interests of the shareholders and other constituents of the company without undue concern over whether the transactions may jeopardize the executives’ own employment.
Highlights of Our Change-in-Control Severance Plans
•all regular employees are covered
•double trigger required
•no tax gross-ups
•up to two-year pay protection
•18-month benefit continuation
Although benefit levels may differ depending on the employee’s job level and seniority, the basic elements of the plans are comparable for all eligible employees:
•Double trigger:Unlike "single trigger" plans that pay out immediately upon a change in control, very specifically, but generallyour plans require a "double trigger"—a change in control followed by an involuntary loss of employment within two years. This is consistent with the terms include the occurrenceplans' intent to provide employees with financial protection upon loss of one of the following: (i) acquisition of 20 percent or more of the company’s stock; (ii) replacement by the shareholders of one half or more of the Board of Directors; (iii) consummation of a merger, share exchange, or consolidation of the company (other than a transaction that results in the Lilly shareholders prioremployment. With respect to unvested equity, performance to the transaction continuing to hold more than 60 percentdate of the voting stock of the combined entity); or (iv) liquidation of the company or sale or disposition of all or substantially all of its assets. The amounts shown in the table for “involuntary or good-reason termination after change in control” are based on the following assumptions and plan provisions:
Covered terminations. The table assumes a termination of employment that is eligible for severance under the terms of the plan, based on the named executive officer’s compensation, benefits, age, and service credit at December 31, 2017. Eligible terminations include an involuntary termination for reasons other than for cause or a voluntary termination by the executive for good reason, within two years following the change in control.
A termination of an executive officer by the company is for cause if it is for any of the following reasons: (i) the employee’s willful and continued refusal to perform, without legal cause, his or her material duties, resulting in demonstrable economic harm to the company; (ii) any act of fraud, dishonesty, or gross misconduct resulting in significant economic harm or other significant harm to the business reputation of the company; or (iii) conviction of or the entering of a plea of guilty or nolo contendere to a felony.
A termination by the executive officer is for good reason if it results from: (i) a material diminution in the nature or status of the executive’s position, title, reporting relationship, duties, responsibilities, or authority, or the assignment to him or her of additional responsibilities that materially increase his or her workload; (ii) any reduction in the executive’s then-current base salary; (iii) a material reduction in the executive’s opportunities to earn incentive bonuses below those in effect for the year prior to the change in control; (iv) a material reduction in the executive’s employee benefits from the benefit levels in effect immediately prior to the change in control; (v) the failure to grant to the executive stock options, stock units, performance shares, or similar incentive rights during each 12-month period following the change in control on the basis of a number of shares or units and all other material terms at least as favorable to the executive as those rights granted to him or her on an annualized average basis for the three-year period immediately prior to the change in control; or (vi) relocation of the executive by more than 50 miles.
Cash severance payment. The cash severance payment amounts to two times the executive officer's annual base salary plus two times the executive officer’s bonus target for that year under the bonus plan.
Continuation of medical and welfare benefits. This amount represents the present value of the change-in-control plan’s provision, following a covered termination, of 18 months of continued coverage equivalent to the company’s current active employee medical, dental, life, and long-term disability insurance. Similar actuarial assumptions to thosewill be used to calculate incremental pension benefits apply to the calculation for continuation of medical and welfare benefits, with the addition of actual COBRA rates based on current benefit elections.
Acceleration of equity awards. Upon a covered termination, any unvested equity awards would convert into restricted stock units of the new company, withdetermine the number of shares earned under an award, but vesting does not accelerate immediately upon a change in control. Rather, the performance-adjusted awards based on accrued performance at the time of the transaction. Thewill convert to time-based restricted stock units willthat continue to vest andwith the new company. Shares will pay out upon the earlier of the completion of the original award period; upon a covered termination; or if the successor entity does not assume, substitute, or otherwise replace the award. The amount in this column represents the valueawards.
•Covered terminations: Employees are eligible for payments if, within two years of the accelerationchange in control, their employment is terminated (i) without cause by the company or (ii) for good reason by the employee, each as defined in the plans. See "Compensation—Executive Compensation—Payments upon Termination or Change in Control" for a more detailed discussion, including a discussion of what constitutes a change in control.
•Employees who suffer a covered termination receive up to two years of pay and 18 months of benefits protection:These provisions ensure employees a reasonable period of protection of their income and core employee benefits.
•Severance payment. Eligible terminated employees would receive a severance payment ranging from six months' to two years’ base salary. Executives are all eligible for two years’ base salary plus two times the then-current year’s target bonus.
•Benefit continuation. Basic employee benefits such as health, dental, life insurance and long-term disability would continue for 18 months following termination of employment, unless the individual becomes eligible for coverage with a new employer. All employees would receive an additional two years of both age and years-of-service credit for purposes of determining eligibility for retiree medical and dental benefits.
•Accelerated vesting of equity awards: Any unvested equity grants, hadawards would vest at the time of a qualifying termination occurred on December 31, 2017. covered termination.
•Excise taxestax:. UponIn some circumstances, the payments or other benefits received by the employee in connection with a change in control employees may be subject to certain excise taxescould exceed limits established under Section 280G of the Internal Revenue Code. The employee would then be subject to an excise tax on top of normal federal income tax. The company does not reimburse the affected employees for those excisethese taxes. However, the amount of any change-in-control-related benefit will be reduced to the 280G limit if the effect would be to deliver a greater after-tax benefit than the employee would receive with an unreduced benefit.
Share Ownership and Retention Guidelines
Share ownership and retention guidelines help create direct alignment of interests between senior management and shareholders over the longer term. Lilly has established a formal share ownership policy under which the CEO and other senior executives are required to acquire and hold Lilly shares in an amount representing a multiple of base salary. During 2021, the Compensation Committee approved an increase to share ownership requirements for the CEO and the other named executive officers as follows:
| | | | | | | | |
| Previous Share Ownership Requirement | Current Share Ownership Requirement |
| CEO | 6X Base Salary | 12X Base Salary |
| Other NEOs | 4X Base Salary | 6X Base Salary |
Until ownership of the required number of shares is reached, an executive officer must hold 50 percent of all shares, net of tax, from all equity payouts. Executive officers are also required to hold all shares received from equity program payouts, net of taxes, or any income taxes payablefor at least one year, even once share ownership requirements have been met. For performance awards granted to executive officers, this holding requirement is met by the employee. To reduce13-month service-vesting period after the employee's exposureend of the performance period.
All of the named executive officers are compliant with the share ownership guidelines. The following graphic shows each respective named executive officer's guideline and each named executive officer's holdings as of December 31, 2021, other than Mr. Smiley, who resigned effective as of February 9, 2021.
Prohibition on Hedging and Pledging Shares
Non-employee directors and employees, including executive officers, are not permitted to excise taxes, the employee’s change-in-control benefit may be decreasedhedge their economic exposures to maximize the after-tax benefitcompany stock through short sales or derivative transactions. Non-employee directors and employees, including executive officers, are prohibited from pledging any company stock (i.e., using company stock as collateral for a loan or trading shares on margin).
Executive Compensation Recovery Policy
Pursuant to the individual.
Payments Upon Change in Control Alone. In general, the change-in-control plan is a “double trigger” plan, meaning paymentsour executive compensation recovery policy (the Executive Compensation Recovery Policy), all incentive awards are made only if the employee suffers a coveredsubject to forfeiture upon termination of employment within two years followingprior to the changeend of the performance or vesting period or for disciplinary reasons. In addition, the Compensation Committee adopted updates to the Executive Compensation Recovery Policy to be effective January 1, 2022 (the Executive Compensation Recovery Policy, as updated and effective January 1, 2022, the Updated Executive Compensation Recovery Policy) which provides the Compensation Committee broad discretion to claw back non-qualified retirement benefits accrued beginning January 1, 2022 from any member of senior management whose misconduct results in control,a material violation of law or company policy that causes significant harm to the company or who fails in his or her supervisory responsibility to prevent such misconduct by others.
Pursuant to the Executive Compensation Recovery Policy, the company could recover all or a portion of any incentive compensation and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, the company could also recover non-qualified retirement benefits accrued beginning January 1, 2022 from an executive officer in the case of equitymaterially inaccurate financial statements or material errors in the performance calculation, whether such inaccuracies or errors result in a restatement and whether or not the executive officer has engaged in wrongful conduct.
The Executive Compensation Recovery Policy covers any incentive compensation awarded or paid to a member of senior management during the last three years and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, covers non-qualified retirement benefits accrued beginning January 1, 2022. Subsequent changes in status, including retirement or termination of employment, do not affect the company’s rights to recover compensation under the policy.
The company will disclose decisions to take action pursuant to the Updated Executive Compensation Recovery Policy in its proxy materials when required by, and in compliance with, SEC rules and regulations and other applicable laws. In addition, when legally permissible to do so, the company will disclose a decision to take action under this policy when the facts and circumstances of the matter that triggered application of the policy have been publicly disclosed in the company’s filings with the SEC and where disclosure can be made without prejudicing the company and its shareholders.
The principles of our robust Updated Executive Compensation Recovery Policy are also incorporated into the terms of our incentive plans and award agreements, which, in the event of misconduct meeting the standards described above, allow the Compensation Committee to reduce or cancel awards ifor payouts that would otherwise have been earned based on company performance. Action by the successor entity does not assume, substitute,Compensation Committee to reduce or otherwise replacecancel awards or payouts can occur during or following the awards.relevant performance period.
Compensation Committee Matters
Background
Role of the Independent Consultant in assessingAssessing Executive Compensation
The Compensation Committee has retained Cimi B. Silverberg of Frederic W. Cook & Co., Inc., (FW Cook) as its independent compensation consultant. Ms. SilverbergFW Cook reports directly to the committee. Neither she nor her firmCompensation Committee, and it is not permitted to have any business or personal relationship with management or the members of the committee. The consultant’sCompensation Committee. FW Cook’s responsibilities are to:
•review the company’s total compensation philosophy, peer group, and target competitive positioning for reasonableness and appropriatenessappropriateness;
•review the company’s executive compensation program and advise the committeeCompensation Committee of evolving best practicespractices;
•provide independent analyses and recommendations to the committeeCompensation Committee on the CEO’s paypay;
•review the draft CD&A and related tables for the proxy statementstatement;
•proactively advise the committeeCompensation Committee on best practices for board governance of executive compensationcompensation; and
•undertake special projects at the request of the committeeCompensation Committee chair.
Ms. SilverbergFW Cook interacts directly with members of company management only on matters under the committee’sCompensation Committee’s oversight and with the knowledge and permission of the committeeCompensation Committee chair.
Role of Executive Officers and Management in assessingAssessing Executive Compensation
With the oversight of the CEO and the senior vice president of human resources and diversity, the company’s global compensation group formulates recommendations on compensation philosophy, plan design, and compensation for executive officers (other than the CEO, as noted below). The CEO provides the committeeCompensation Committee with a performance assessment and compensation recommendation for each of the other executive officers. The committeeCompensation Committee considers those recommendations with the assistance of its compensation consultant. The CEO and the senior vice president of human resources and diversity attend committeeCompensation Committee meetings; however, they are not present for executive sessions or any discussion of their own compensation. Only non-employee directors and the committee’sCompensation Committee’s consultant attend executive sessions.
The CEO does not participate in the formulation or discussion of his pay recommendations. He has no prior knowledge of the recommendations that the consultantFW Cook makes to the committee.
Compensation Committee.
Risk Assessment Process
As part of the company's overall enterprise risk management program, in 20172021 (consistent with prior years), the committeeCompensation Committee reviewed the company’s compensation policies and practices and concluded that the programs and practices are not reasonably likely to have a material adverse effect on the company. The committeeCompensation Committee noted numerous policy and design features of the company’s compensation programs and governance structure that reduce the likelihood of inappropriate risk-taking, including, but not limited to:
The committee comprises of•Only independent directors onlyserve on the Compensation Committee
•The committeeCompensation Committee engages its own independent compensation consultant
•The committeeCompensation Committee has downward discretion to lower compensation plan payouts
•The committeeCompensation Committee approves all adjustments to financial results that affect compensation calculations
•Different measures and metrics are used across multiple incentive plans that appropriately balance cash/stock, fixed/variable pay, and short-term/long-term incentives
•Incentive plans have predetermined maximum payouts
•Performance objectives are challenging but achievable
•Programs with operational metrics have a continuum of payout multiples based upon achievement of performance milestones, rather than "cliffs" that might encourage suboptimal or improper behavior
•A compensation recovery policy is in place for all members of senior management; negative compensation consequences can be appliedresult in cases ofinvolving serious compliance violations
•Meaningful share ownership and retention requirements are in place for all members of senior management and the board.
Compensation Committee Report
The Compensation Committee evaluates and establishes compensation for executive officers and oversees the deferred compensation plan, the company’s management stock plans, and other management incentive and benefit programs. Management has the primary responsibility for the company’s financial statements and reporting process, including the disclosure of executive compensation.compensation in the CD&A. With this in mind, the Compensation Committee has reviewed and discussed with management the CD&A above. The committeewith management. Based on this discussion, the Compensation Committee recommended to the Board of Directorsboard that the CD&A be included in this proxy statement and the company's Annual Report on Form 10-K for the fiscal year ended December 31, 2021, for filing with the SEC.
Compensation Committee
Ralph Alvarez, Chair
Michael L. EskewJ. Erik Fyrwald
EllenKimberly H. Johnson
Juan R. MarramLuciano
Kathi P. SeifertKaren Walker
Compensation Discussion and Analysis
This CD&A describes our executive compensation philosophy, the Compensation Committee’s process for setting executive compensation, the elements of our compensation program, the factors the Compensation Committee considered when setting executive compensation for 2021, and how the company’s results affected incentive payouts. This CD&A provides compensation information for our chief executive officer, David Ricks, our chief financial officer, Anat Ashkenazi, and our next three most highly compensated executive officers who were serving as executive officers on December 31, 2021, Daniel Skovronsky, Jacob Van Naarden and Diogo Rau. The CD&A also includes our former chief financial officer, Joshua Smiley, who resigned from his officer position on February 9, 2021.
| | | | | |
| Name and principal occupation |
| David A. Ricks Chair, President and CEO |
| Anat Ashkenazi Senior Vice President and Chief Financial Officer |
| Daniel Skovronsky, M.D., Ph.D. Senior Vice President, Chief Scientific and Medical Officer and President, Lilly Research Laboratories |
| Jacob Van Naarden Senior Vice President, CEO Loxo Oncology at Lilly and President, Lilly Oncology |
| Diogo Rau Senior Vice President and Chief Information and Digital Officer |
Four of our named executive officers, Ms. Ashkenazi, Mr. Van Naarden, Mr. Rau and Mr. Smiley, served in their respective roles for a portion of 2021. Ms. Ashkenazi succeeded Mr. Smiley as senior vice president and CFO on February 9, 2021. Prior to becoming CFO, Ms. Ashkenazi served as senior vice president, controller and chief financial officer, Lilly Research Laboratories.
Mr. Van Naarden became senior vice president, CEO Pay Ratioof Loxo Oncology at Lilly and President of Lilly Oncology on September 5, 2021. He previously served as CEO of Loxo Oncology at Lilly. Mr. Rau joined Lilly as senior vice president and chief
information and digital officer on May 17, 2021. Prior to joining Lilly, Mr. Rau served as senior director, information systems and technology for retail and online stores at Apple, Inc.
Our Philosophy on Compensation
At Lilly, our purpose is to unite caring with discovery to create medicines that make life better for people around the world. To do this, we must attract, engage, and retain highly talented individuals from a variety of backgrounds and motivate them to create long-term shareholder value by achieving top-tier corporate performance while embracing the company's core values of integrity, excellence, and respect for people. Our compensation programs are designed to help us achieve these goals while balancing the long-term interests of our shareholders and customers.
Objectives
Our compensation and benefits philosophyprograms are based on the following objectives:
•Reflect individual and company performance: We reinforce a high-performance culture by linking pay with individual and company performance. As employees assume greater responsibilities, the proportion of total compensation based on absolute company performance, relative company performance and shareholder returns increases. We perform annual reviews to ensure our programs provide an incentive to deliver long-term, sustainable business results while discouraging excessive risk-taking or other adverse behaviors.
•Attract and retain talented employees: Compensation opportunity is market competitive and reflects the level of job impact and responsibilities. Retention of talent is an important factor in the design of our compensation and benefit programs.
•Implement broad-based programs: While the amount of compensation paid to employees varies, the overall structure of our compensation and benefit programs areis broadly similar across the organization to encourage and reward all employees who contribute to our success.
•Consider shareholder input: Management and the Compensation Committee consider the results of our annual say-on-pay vote and other sources of shareholder feedback when designing executive compensation and benefit programs.
Say-on-Pay Results
At our 2021 annual meeting of shareholders, greater than 95 percent of the shares cast voted in favor of the company's say-on-pay proposal on executive compensation. Management and the Compensation Committee view this vote as supportive of the company's overall approach toward executive compensation.
Compensation Committee's Processes and Analyses
Setting Compensation
The Compensation Committee considers individual performance assessments, compensation recommendations from the CEO (with respect to each of the other executive officers), company performance, peer group data, input from its compensation consultant, and its own judgment when determining compensation for the company's executive officers.
•Individual performance: Generally, the independent directors, under the direction of the lead independent director, meet with the CEO at the beginning of each year to establish the CEO's performance objectives. At the end of the year, the independent directors meet to assess the CEO's achievement of those objectives along with other factors, including contribution to the company’s performance, diversity, ethics, and integrity. This evaluation is used in setting the CEO's compensation opportunity for the next year.
The Compensation Committee receives individual performance assessments and target compensation recommendations from the CEO for each of the remaining executive officers. Each executive officer’s performance assessment is based on the achievement of objectives established at the start of the year, as well as other factors, including contribution to the company's performance, diversity, ethics, and integrity. The Compensation Committee considers these inputs, its knowledge of and interactions with each executive officer, and its judgment to develop a final individual performance assessment. For new executive officers, target compensation is set by the Compensation Committee at the time of promotion or offer.
•Company performance: Lilly performance is considered in multiple ways:
•Overall prior year performance is a factor in setting target compensation for the coming year.
•At the beginning of each calendar year, annual performance goals are established and approved by the compensation committee. Performance against these annual goals is used to determine the short-term cash incentive payout.
•Prior to the annual equity grant, multi-year performance goals are established and approved by the compensation committee. Performance against these multi-year objectives is used to determine the long-term incentive equity payout.
•Peer group analysis: The Compensation Committee uses data from the peer group described below as a market check for compensation decisions but does not use this data as the sole basis for its compensation targets and does not target a specific position within that range of market data.
•Input from independent compensation consultant:The Compensation Committee considers the advice of its independent compensation consultant, FW Cook, when setting executive officer compensation.
Competitive Pay Assessment
Lilly’s peer group is composed of companies that directly compete with Lilly, use a similar business model, and employ people with the unique skills required to operate an established biopharmaceutical company. The Compensation Committee selects a peer group whose median market cap and revenue are broadly like Lilly's. The Compensation Committee reviews the peer group at least every three years. The Compensation Committee established the following peer group in May 2018 for purposes of assessing competitive pay:
| | | | | | | | | | | | | | | | | | | | |
| AbbVie | | Bristol-Myers Squibb | | Merck | | Sanofi |
| Allergan* | | Celgene* | | Novartis | | Shire* |
| Amgen | | Gilead | | Novo Nordisk | | Takeda |
| AstraZeneca | | GlaxoSmithKline | | Pfizer | | |
| Biogen | | Johnson & Johnson | | Roche | | |
| *Market data unavailable for assessing competitive 2021 pay due to business mergers. |
At the time of the review in May 2018, all peer companies were no greater than two times our revenue or market cap except Johnson & Johnson, Novartis, Pfizer, and Roche. The Compensation Committee included these four companies despite their size because they compete directly with Lilly, have similar business models, and seek to hire from the same pool of management and scientific talent.
When determining pay levels for target compensation, the Compensation Committee considers an analysis of market competitive pay for each executive officer position (except CEO), along with internal factors such as the performance and experience of each executive officer. The independent compensation consultant for the Compensation Committee provides a similar analysis when recommending pay levels for the CEO. The CEO analysis includes a comparison of our CEO actual total direct compensation in the prior year to company performance on an absolute and relative basis. The analysis also includes a comparison of current target total direct compensation for our CEO to the most recently available data on CEO target total direct compensation for our peer group with an emphasis on peers that are headquartered in the U.S. On average, the named executive officers' target total direct compensation for 2021 was comparable to the median of the peer group.
Components of Our Compensation
Our 2021 executive compensation was primarily composed of three components:
•base salary
•annual cash bonus, which is generally based on company performance relative to internal targets for revenue, EPS, and the progress of our pipeline
•three different forms of equity incentives:
◦performance award, which is a performance-based equity award that vests over three years. Payout is based on the company's two-year growth in EPS relative to the median external analyst anticipated peer group EPS growth. The earned shares are then subject to a 13-month service-vesting period
◦shareholder value award, which is a performance-based equity award that pays out based on absolute company stock price growth measured over a three-year period, followed by a one-year holding period
◦relative value award, which is a performance-based equity award that pays out based on company TSR results relative to peers measured over a three-year period, followed by a one-year holding period.
Executives also receive a company benefits package, described below under "Other Compensation Practices and Information—Employee Benefits."
Adjustments to Reported Financial Results
The Compensation Committee has authority to adjust the company's reported revenue and EPS upon which incentive compensation payouts are determined to eliminate the distorting effect of unusual income or expense items. The adjustments are intended to:
•align award payments with the underlying performance of the core business
•avoid volatile, artificial inflation or deflation of awards due to unusual items in any year in the performance period
•eliminate certain counterproductive short-term incentives—for example, incentives to refrain from acquiring new technologies, to defer disposing of underutilized assets, or to defer settling legacy legal proceedings to protect current bonus payments
•facilitate comparisons with peer companies.
The Compensation Committee considers the adjustments approved by the Audit Committee for reporting non-GAAP EPS and other adjustments, based on guidelines approved by the Compensation Committee prior to the performance period. The Compensation Committee reviews and approves adjustments on a quarterly basis and may adjust payouts for items, including but not limited to, the impact of significant acquisitions or divestitures, the impact of share repurchases that differ significantly from business plan, and large swings in foreign exchange rates. Further details on the adjustments for 2021 and the rationale for making these adjustments are set forth in Appendix A, "Summary of Adjustments Related to the Annual Cash Bonus and Performance Award." For ease of reference, throughout the CD&A and the other compensation disclosures, we refer simply to "revenue" and "EPS," but we encourage you to review the information in Appendix A to understand the adjustments from reported revenue and EPS that were approved.
The Compensation Committee also has general authority to apply downward discretion to bonus, performance award, shareholder value award, and relative value award payouts for individual or all executive officers.
1.Base Salary
In setting salaries, Lilly seeks to retain, motivate, and reward successful performers while maintaining affordability within the company's business plan. Base salaries are reviewed and established annually and may be adjusted upon promotion, following a change in job responsibilities, or to maintain market competitiveness. Salaries are based on each person's level of contribution, responsibility, expertise, and competitiveness and are compared annually with peer group data.
Base salary increases for 2021 were based on competitiveness. Generally, executive base salaries are positioned against market salaries, considering experience and performance over time. If an executive's salary is deemed competitive given their experience and performance, no salary increase is provided.
2.Annual Cash Bonus
The Bonus Plan is designed to reward the achievement of the company’s annual financial and innovation objectives. All the named executive officers participated in the Bonus Plan during 2021. Mr. Van Naarden's participation in the Bonus Plan commenced at the time of his promotion on September 5, 2021. For the portion of the 2021 performance year prior to his promotion, Mr. Van Naarden participated in The Loxo Oncology, Inc. Bonus Plan (Loxo Bonus Plan), and in lieu of the payments he would have been eligible to receive thereunder, the Company paid Mr. Van Naarden a one-time payment, as further described below in the Summary Compensation Table.
Bonus Plan
The Compensation Committee sets performance goals and individual bonus targets for the Bonus Plan at the beginning of each year. The bonus is based on three areas of company performance measured relative to internal targets: revenue, EPS, and pipeline progress. The annual cash bonus payout is calculated as follows:
Actual payouts can range from 0 to 200 percent of an individual's bonus target. The Compensation Committee references the annual operating plan and pipeline objectives to establish performance targets and to assess the relative weighting for each objective. In 2021, the weightings remain unchanged from the prior year:
| | | | | |
| Lilly Goals | Weighting |
| Revenue performance | 25% |
| EPS performance | 50% |
| Pipeline progress | 25% |
Based on this weighting, the company bonus multiple is calculated as follows:
Loxo Bonus Plan
Performance goals for the Loxo Bonus Plan are determined by a committee consisting of senior company leadership (the Loxo Oncology Steering Committee) and mirror the goals under the Bonus Plan; provided, however, that eligible employees have an additional bonus opportunity that may be awarded for the attainment of other goals specific to oncology research and development (Additional Cash Award). These other goals can vary annually, but typically include preclinical, clinical, regulatory actions, external innovation and scientific publication milestones. These additional goals associated with molecule advancement and proof of concept milestones typically carry more weight in the Additional Cash Award results.
The annual Loxo Bonus Plan payout is calculated by multiplying a participant's base earnings, the individual bonus target and the sum of the Bonus Plan and Additional Cash Award payout multiples.
Base Earnings x Individual Bonus Target x (Bonus Plan Multiple + Additional Cash Award Multiple)
Actual payouts for the Loxo Bonus Plan can range from 0 to 400 percent of an individual's bonus target (0 to 200 percent for the Bonus Plan performance and 0 to 200 percent for the Additional Cash Award). Goal attainment is determined by the Loxo Oncology Steering Committee.
3.Equity Incentives
The company grants three types of equity incentives to executives and certain other employees: performance awards that are designed to focus leaders on multi‑year operational performance relative to peer companies, shareholder value awards that are intended to align earned compensation with long-term growth in shareholder value, and relative value awards which encourage TSR outperformance compared to our peer group. These awards, when considered together, align with shareholder interests by incenting long-term operational excellence, shareholder return and peer company outperformance without encouraging excessive risk-taking behaviors. The Compensation Committee has the discretion to adjust payout from an equity award granted to an executive officer downward from the amount yielded by the applicable formula.
Performance Awards
Performance awards vest over three years. Payouts are based on achieving EPS growth targets over a two-year performance period, followed by an additional 13-month service-vesting period for executive officers. During the service-vesting period, the award is held in the form of restricted stock units. The growth-rate targets are set relative to the median external analyst anticipated EPS growth for our peer group over the same performance period. These awards do not accumulate dividends during the two-year performance period, but they do accumulate dividend equivalent units during the service-vesting period.
The Compensation Committee believes EPS growth is an effective measure of operational performance because it is closely linked to shareholder value, is broadly communicated to the public, is understood by Lilly employees, and allows for objective comparisons to the performance of Lilly's peer group. Consistent with the objectives established by the Compensation Committee, Lilly company performance exceeding the expected peer group median results in above‑target payouts, while Lilly company performance lagging the expected peer group median results in below‑target payouts. Possible payouts range from 0 percent to 175 percent of the target number of shares, depending on Lilly EPS growth over the performance period.
The measure of EPS used in the performance award program differs from the measure used in the Bonus Plan in two ways. First, the EPS goal in the Bonus Plan is set with reference to internal goals that align to our annual operating plan, while the EPS goal in the performance award program is set based on the external analyst anticipated EPS growth rates of our peer group. Second, the Bonus Plan measures EPS over a one-year period, while the performance award program measures EPS over a two-year period. In any given year, the Bonus Plan may pay above target while the performance award pays below target (or vice versa).
Shareholder Value Awards
Shareholder value awards are earned based on Lilly's share price performance. Shareholder value awards pay above target if Lilly's stock outperforms an expected rate of return and below target if Lilly's stock underperforms that expected rate of return. The expected rate of return is based on the three-year TSR that a reasonable investor would consider appropriate when investing in a basket of large-cap U.S. companies, as determined by the Compensation Committee. The minimum price to achieve target is calculated by multiplying the starting share price of Lilly's stock by the three-year compounded expected rate of return less Lilly's dividend yield. Shareholder value awards have a three-year performance period, and any shares paid are subject to a one-year holding requirement. No dividends are accrued during the performance period. Possible payouts are based on share price growth and range from 0 to 175 percent of the target number of shares. Executive officers receive no payout if Lilly's TSR for the three-year period is zero or negative.
Relative Value Awards
Relative value awards are earned based on Lilly's TSR performance relative to our peer group. The minimum performance to achieve target is a TSR that is equal to the median TSR performance for the peer group. Relative value awards have a three-year performance period, and any shares paid are subject to a one-year holding requirement. No dividends are accrued during the performance period. Executive officers receive no payout if Lilly's TSR for the three-year period is 30 or more percentage points below the median TSR performance for the peer group over the same time period. Possible payouts range from 0 to 175 percent of the target number of shares.
Pay for Performance
The mix of compensation for our named executive officers reflects Lilly’s desire to link executive compensation with individual and company performance. As reflected in the charts below, a substantial portion of the target pay for executive officers is performance-based. The annual cash bonus and equity payouts are contingent upon company performance, with the bonus factoring in performance over a one-year period, and equity compensation factoring in performance over multi-year periods. The charts below depict the annualized mix of target compensation for Lilly’s CEO and the average for the
other named executive officers annualized for Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau. The Other Named Executive Officers chart below excludes Mr. Smiley.
Target Total Compensation
Performance Review Process
In setting 2021 target compensation for the named executive officers, the Compensation Committee considered individual contributions, Lilly's performance during 2020, internal relativity, peer group data, and input from the CEO for named executive officers other than himself.
Evaluation of Individual Named Executive Officer Performance
A summary of the Compensation Committee's review of individual named executive officer performance in 2020 that influenced decisions on 2021 target compensation for these executives is provided below:
David Ricks, Chair, President, and CEO:
In accordance with the company’s corporate governance guidelines, the independent directors conducted an assessment of Mr. Ricks’ performance led by the lead independent director. The independent directors believe the company largely met or exceeded its combined financial and strategic goals for 2020 under Mr. Ricks’ leadership. In 2020, Mr. Ricks and his team:
•Launched several innovative medicines to patients globally. Additionally, 25 new indications for already marketed medicines were launched to patients around the world
•Progressed 17 potential new medicines into Phase I clinical development from both internal research and external sources
•Initiated actions against the global COVID-19 pandemic, including (1) implementing diagnostic testing in our community, (2) discovering and developing SARS-CoV-2 neutralizing antibodies leading to the Emergency Use Authorization for bamlanivimab, and (3) striving to protect the health and safety of Lilly employees while continuing to provide life saving medicines
•Completed the acquisition of Dermira, expanded Lilly's strategic collaboration with Innovent for Tyvyt globally and completed seven preclinical asset transactions
•Continued to drive a cross-company productivity agenda resulting in savings that funded increased investment in research and development
•Implemented a strategy that improved diversity, equity and inclusion across the company, increased the representation of women and minorities in management and conducted pay equity studies to promote equality in pay. The company was ranked #3 on the DiversityInc. top 50 companies list and received Women Business Collaborative CEO recognition for excellence in gender diversity
•Led the development of Lilly's comprehensive Racial Justice Commitment to help improve equity within Lilly and the communities in which it operates
•Achieved certain 2020 environmental performance goals first established in 2013, such as greenhouse gas emissions, energy efficiency, waste efficiency and wastewater
•Established new long-term goals for environmental, safety and governance goals for 2030
In addition, the company was named one of the world’s most ethical companies by the Ethisphere Institute.
Anat Ashkenazi, Senior Vice President and Chief Financial Officer:
Ms. Ashkenazi became Senior Vice President and Chief Financial Officer (CFO) on February 9, 2021. Prior to becoming CFO, Ms. Ashkenazi served as senior vice president and controller, and led corporate strategy at Lilly Research Laboratories. As controller, Ms. Ashkenazi:
•Played a key role in the company achieving its financial targets in 2020 and in the company’s efforts to reallocate financial resources in the fight against COVID-19
•Led the company’s broad productivity agenda for several years, having been a key partner to the operating units across the value cycle and played a key role in all business development activities
•Led the company’s 2021 strategic plan and 2021 operating plan
•Sponsors Lilly’s Veteran Network, an employee resource group focused on recruiting and advancing people who served in the country’s military
Daniel Skovronsky, M.D., Ph.D., Senior Vice President, Chief Scientific and Medical Officer and President, Lilly Research Laboratories:
Dr. Skovronsky advanced innovation for patients as the company's chief scientific and medical officer. His contributions in 2020 include:
•Advanced innovative medicines through the product pipeline leading to numerous first approvals, including bamlanivimab (under Emergency Use Authorization from the FDA) in partnership with AbCellera. Additionally, 25 new indications for already-marketed medicines were launched to patients globally
•Achieved several significant positive data readouts for Verzenio (Phase III in the adjuvant setting), LOXO-305 (advanced b-cell malignancies), Jardiance (cardiovascular outcomes in heart failure with reduced ejection fraction), mirikizumab (psoriasis head-to-head versus secukinumab) and tirzepatide (type 2 diabetes)
•Co-led the acquisition of Dermira, expanded Lilly's strategic collaboration with Innovent for Tyvyt globally, and completed seven preclinical asset transactions
•Sped research resulting in 17 potential new medicines advancing to Phase I clinical development including both internally discovered molecules and molecules sourced externally via business development
•Led the company’s efforts in the fight against SARS-CoV-2 including (1) implementing diagnostic testing at our headquarters in Indianapolis, Indiana and (2) discovering and developing SARS-CoV-2 neutralizing antibodies leading to the Emergency Use Authorization for bamlanivimab
•Enhanced strategies to further reduce drug discovery and development timelines, leading to earlier product launch
•Led DEI strategies in research and development to improve innovation and productivity.
Jacob Van Naarden, Senior Vice President, CEO Loxo Oncology at Lilly and President, Lilly Oncology:
Mr. Van Naarden served as chief operating officer of Loxo Oncology at Lilly when the company acquired Loxo Oncology, Inc. in 2019 and was later named CEO of Loxo Oncology at Lilly in January 2021. Mr. Van Naarden played an integral role with numerous oncology assets including the approval of Retevmo. As CEO of Loxo Oncology at Lilly, Mr. Van Naarden has built a science-first drug discovery and development organization. He has led numerous business development transactions and contributed to the company’s scientific efforts in other therapeutic areas.
Diogo Rau, Senior Vice President and Chief Information and Digital Officer:
Mr. Rau joined Lilly as senior vice president and chief information and digital officer in May 2021. Prior to joining Lilly, Mr. Rau served as senior director of information systems and technology for retail and online stores and most recently served as the top executive of information technology for retail and online stores at Apple, Inc. (Apple). During his 10 years at Apple, he led the development and implementation of the technology supporting the Apple Online Store and Apple retail stores,
including e-commerce platform, mobile point of sale, the Apple Store App, and systems used by store team members. Prior to joining Apple, Mr. Rau was a partner with McKinsey & Company.
In connection with Mr. Smiley’s resignation as an officer on February 9, 2021, the Compensation Committee took action to reduce his 2020 Bonus Plan payout to zero, reduce his 2018-2020 shareholder value award and cancel all his other outstanding equity incentive awards granted in 2019 and 2020. The foregoing terms were agreed to by Mr. Smiley pursuant to his release agreement effective as of February 9, 2021. The material terms of the agreement were previously disclosed in the company’s 2021 proxy statement.
Target Compensation
The information below reflects total compensation at target for named executive officers for 2021. The actual compensation received is summarized below in "Compensation Results."
Rationale for Changes to Named Executive Officer Target Compensation
The Compensation Committee established the 2021 target total compensation for each named executive officer, except Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau, based on the named executive officer's 2020 performance, internal relativity, and peer group data. The Compensation Committee approved target total compensation for Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau upon their promotion or hire, as applicable, based on job scope, market data, experience, and internal relativity.
Base Salary
The following table shows the approved annualized salary for each named executive officer. Mr. Ricks and Dr. Skovronsky did not receive an adjustment to base pay in 2021, and the remaining executive officers' 2021 annualized salaries were effective on the day they assumed their new responsibilities. Each named executive officer's actual base salary earned during 2021 is reflected in the Summary Compensation Table in the "Executive Compensation" section of this proxy statement.
| | | | | | | | | | | | | | | | | |
Name2 | 2020 Annual Base Salary Effective March 1, 2020 | 2021 Annual Base Salary Effective March 7, 2021 | Increase |
| Mr. Ricks | $1,500,000 | | $1,500,000 | | 0 | % |
| Ms. Ashkenazi | N/A | 1 | $900,000 | | N/A |
| Dr. Skovronsky | $1,000,000 | | $1,000,000 | | 0 | % |
| Mr. Van Naarden | N/A | 1 | $675,000 | | N/A |
| Mr. Rau | N/A | 1 | $670,000 | | N/A |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 For the period beginning on January 1, 2021 through February 8, 2021, Mr. Smiley's annual base salary was $1,000,000, and for the transition period beginning on February 9, 2021 through July 31, 2021, Mr. Smiley earned a biweekly base salary of $9,000.
Annual Cash Bonus Targets
Based on a review of internal relativity, peer group data, and individual performance, the Compensation Committee retained the same percent-of-salary bonus target as in 2020 for Mr. Ricks, increased the bonus target for Dr. Skovronsky to 100 percent, set Ms. Ashkenazi's bonus target at 100 percent, and set Mr. Van Naarden's and Mr. Rau's bonus targets at 80 percent. Bonus targets are shown in the table below as a percentage of each named executive officer’s earnings from base salary in 2021:
| | | | | | | | | | | | | | |
Name3 | 2020 Bonus Target | 2021 Bonus Target |
| Mr. Ricks | 150 | % | | 150 | % | |
| Ms. Ashkenazi | N/A | 1 | 100 | % | |
| Dr. Skovronsky | 95 | % | | 100 | % | |
| Mr. Van Naarden | N/A | 1 | 80 | % | 2 |
| Mr. Rau | N/A | 1 | 80 | % | 2 |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 Mr. Van Naarden's Bonus Plan participation was initiated upon his promotion on September 5, 2021. Prior to his promotion, Mr. Van Naarden participated in the Loxo Bonus Plan, which entitled him to a target bonus equal to 60% of base salary.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley was not entitled to an annual bonus for the 2020 and 2021 calendar years. See "Agreement with Former Chief Financial Officer" for additional information.
Equity Incentives - Target Grant Values
For 2021 equity grants, the Compensation Committee set the total target values for named executive officers based on market data, individual performance, and internal relativity. Named executive officers have 35 percent of their equity target allocated to shareholder value awards and relative value awards, respectively, and 30 percent to performance awards. Total target values for the 2020 and 2021 equity grants to the named executive officers were as follows:
| | | | | | | | | | | | | | |
Name5 | 2020 Annual Equity Grant | 2021 Annual Equity Grant |
| Mr. Ricks | $12,500,000 | | $14,000,000 | |
| Ms. Ashkenazi | N/A | 1 | $2,200,000 | 2 |
| Dr. Skovronsky | $4,100,000 | | $4,700,000 | |
| Mr. Van Naarden | N/A | 1 | $1,300,000 | 3 |
| Mr. Rau | N/A | 1 | N/A | 4 |
1 Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not named executive officers in 2020.
2 Ms. Ashkenazi’s 2021 award was granted prior to her transition into the Chief Financial Officer role. As such, her 2021 annual equity grant aligns with the terms and conditions associated with her prior position.
3 Mr. Van Naarden was not an executive officer at the time of the 2021 annual equity grant. As such, his 2021 annual equity grant aligns with the terms and conditions associated with his prior position. Mr. Van Naarden also received equity awards of restricted stock units valued at (i) $1,650,000 in February 2021 representing transition equity following Lilly’s acquisition of Loxo Oncology, Inc. and (ii) $1,000,000 in December 2021 associated with Mr. Van Naarden's promotion.
4 Mr. Rau started with Lilly in May 2021, after annual grants were issued; however, he received an equity grant of restricted stock units valued at $4,000,000 upon hire to replace a portion of the outstanding equity he forfeited after leaving Apple and the missed opportunity for a 2021 annual equity award from Lilly.
5 Pursuant to the terms of his separation agreement with the company, Mr. Smiley was not entitled to an annual equity grant in 2021. See "Agreement with Former Chief Financial Officer" for additional information.
Performance Goals for Incentive Programs
Annual Cash Bonus Goals
The Compensation Committee established the company performance targets using the company's 2021 annual operating plan, which was approved by the board in 2020. These targets are described below under "Compensation Results."
Performance Award
In February 2021, the Compensation Committee established a compounded two-year EPS growth target of 8.8 percent per year based on investment analysts’ EPS growth estimates for our peer group companies at that time. To translate the 8.8 percent per year growth goal into a two-year cumulative EPS target, the Compensation Committee applied the target growth to the 2020 adjusted non-GAAP EPS of $6.78 to obtain target 2021 results of $7.38 and then applied the goal growth again to the target 2021 results to obtain target 2022 results of $8.02. The target 2021 and 2022 results were added together to yield a two-year cumulative EPS target of $15.40. Payouts for the 2021-2023 performance award can range from 0 to 175 percent of the target number of shares, as shown below:
Shareholder Value Award
For purposes of establishing the stock price target for the shareholder value awards, the starting price was $152.16 per share, the average closing stock price for all trading days in November and December 2020. The target share price was established using the expected annual rate of return for large-cap companies (8 percent), less an assumed Lilly dividend yield (2.23 percent). To determine payout, the ending price will be the average closing price of company stock for all trading days in November and December 2023. The award is designed to deliver no payout to executive officers if the shareholder return (including projected dividends) is zero or negative. Possible payouts based on share price ranges are illustrated in the grid below.
Relative Value Award
The relative value award is based on the three-year Lilly TSR performance compared to our peer group. To determine payout, the TSR performance is calculated for Lilly and its peers. This calculation compares the average closing price of each company’s stock for all trading days in November and December 2020 to the average closing price of each company’s stock for all trading days in November and December 2023, assuming reinvestment of dividends, to obtain the TSR for each company. The median TSR for the peer companies is then subtracted from Lilly’s TSR to determine what payout has been earned. For example, if Lilly’s TSR was 55 percent over the three-year performance period and the median peer company performance was 41 percent, Lilly would have outperformed by 14 percentage points (55 percent - 41 percent). This outperformance would have resulted in a 130 percent payout based on the payout ranges depicted below.
Compensation Results
The information in this section reflects the amounts paid to named executive officers under the Bonus Plan and for equity awards granted in prior years for which the relevant performance period ended in 2021.
Lilly Performance
In 2021, we exceeded our revenue target and nearly achieved our EPS target. We also made significant pipeline advances. Key pipeline highlights include U.S. approval of a new indication for Verzenio® for certain people with high-risk, early breast cancer; the Emergency Use Authorization for the administration of bamlanivimab and etesevimab together for the treatment
of COVID-19 in patients at high risk for hospitalization; the regulatory submission for tirzepatide for type 2 diabetes in the U.S. to which Lilly applied a priority review voucher, as well as submission in Europe and Japan; positive topline results from donanemab’s TRAILBLAZER-ALZ, a Phase II study evaluating the efficacy and safety of donanemab compared to placebo in patients with early symptomatic Alzheimer’s disease; updated data for pirtobrutinib, from the LOXO-305 BRUIN Phase I/II clinical trial in mantle cell lymphoma and other non-Hodgkin lymphomas, as well as in chronic lymphocytic leukemia and small lymphocytic lymphoma, at the 2021 American Society of Hematology Annual Meeting; positive topline results from mirikizumab’s Phase III LUCENT-1 and 2 studies evaluating the efficacy and safety of mirikizumab compared to placebo for the treatment of patients with moderately-to-severely active ulcerative colitis; and positive topline results from three studies of lebrikizumab’s Phase III program evaluating the efficacy and safety of lebrikizumab in people with moderate-to-severe atopic dermatitis. See "Proxy Statement Summary—Highlights of 2021 Performance."
By the end of 2021, Lilly exceeded its two-year EPS growth target for the performance award and our three-year stock price growth target for the shareholder value award. The company began granting relative value awards in 2020; therefore, no relative value awards were granted or earned for the 2019-2021 performance period. The discussion below details the measures used in each program, what the performance goal was to obtain target performance, how performance outcomes were assessed and what the Compensation Committee approved as the final payout multiple.
Bonus Plan
The company utilized revenue, EPS, and pipeline progress to incent the achievement of 2021 company objectives. Each measure contributes to the final payout multiple on a weighted basis: revenue (25 percent), EPS (50 percent), and pipeline progress (25 percent). Each performance measure is assessed a payout multiple of 0 to 200 percent.
For purposes of the bonus, the Compensation Committee adjusted revenue and non-GAAP EPS to exclude estimated savings from certain discrete and unplanned performance items from the bonus plan multiple. The Science and Technology Committee's assessment of the company's product pipeline achievements is detailed below:
| | | | | | | | |
| Activity | Objective | Achievement |
| Approvals | 2 new drug first approvals
14-15 other approvals | 0 new drug first approvals
18 other approvals |
| Potential new drug Phase III starts | 3 | 2 |
| Potential new drug Phase I starts | 16-17 | 12 |
| Potential new indication or line extension Phase III starts | 5 | 9 |
| Plan Boldly | Meet industry benchmark for speed of development | Exceeded industry benchmark for speed of development |
| Deliver to Launch | Meet planned project timelines | Met planned project timelines |
| Qualitative Assessment | Assessment of the chief scientific and medical officer's evaluation of performance against strategic objectives |
Based on the recommendation of the Science and Technology Committee, the Compensation Committee approved a pipeline multiple of 1.06.
The company's performance compared to targets as well as the resulting bonus multiple, is illustrated below:
For additional information on financial results, see Appendix A, "Summary of Adjustments Related to the Annual Cash Bonus and Performance Award."
When combined, the revenue, EPS, and pipeline multiples yielded a bonus multiple of 1.10.
Loxo Bonus
The Loxo Bonus Plan Committee assessed the level of oncology research and development goal achievement compared to the preclinical, clinical, regulatory actions, external innovation and scientific disclosure milestones established at the beginning of the year. On a payout continuum between 0 – 200 percent, the Loxo Bonus Plan Committee determined the Additional Cash Award portion of the Loxo Bonus would pay at 200 percent. To determine the final Loxo Bonus payout multiple, the Loxo Bonus Plan Committee added the Bonus Plan multiple to the Additional Cash Award multiple to yield a total payout of 310 percent.
Bonus Payments
The 2021 bonuses paid to the applicable named executive officers under the Bonus Plan were as follows:
| | | | | | | | |
Name3 | 2021 Bonus ($) |
| Mr. Ricks | $2,475,000 | |
| Ms. Ashkenazi | $907,816 | |
| Dr. Skovronsky | $1,100,000 | |
Mr. Van Naarden1 | $1,029,507 | |
Mr. Rau2 | $362,831 | |
1 A portion of the 2021 bonus payable to Mr. Van Naarden was attributable to achievement of the performance goals under the Loxo Bonus Plan. The 2021 Loxo Bonus Plan achievement was 310%. The amount of the 2021 bonus paid to Mr. Van Naarden in the table above reflects his achievement under both the Loxo Bonus Plan and the Bonus Plan, in each case, pro-rated to reflect the period in 2021 during which he was a participant in the respective plan. Mr. Van Naarden also received a one-time cash payment of $218,715 in lieu of compensation and benefits he was no longer eligible to receive following his transition to his new role, including compensation for the difference between the bonus he would have earned under the Loxo Bonus Plan and the Lilly Bonus Plan, and changes in his expected out-of-pocket costs for healthcare expenses.
2 Mr. Rau joined Lilly in May 2021, and the table reflects the pro-rated bonus paid for the portion of the year for which he was employed.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley was not entitled to an annual bonus for calendar year 2021. See "Agreement with Former Chief Financial Officer" for additional Information.
2020-2022 Performance Awards
The target cumulative EPS for the 2020-2022 performance award was originally set in the first quarter of 2020, reflecting expected peer group EPS growth of 5.7 percent each year and a total target EPS of $13.13 over the two-year performance period. In December 2020, the Compensation Committee updated the award to align with Lilly's decision to adjust EPS results to remove the impact of gains and losses on investments in equity securities from its non-GAAP measures and compensation program outcomes. The updated award retained the Compensation Committee's decision to establish a compounded two-year EPS growth target of 5.7 percent per year, but the growth was applied to an adjusted 2019 non-GAAP EPS of $5.73 that excluded gains and losses on investments in equity securities. Using this methodology, the adjusted EPS target became $12.46 for the two-year performance period of 2020-2021. The company's actual adjusted EPS over the performance period was $14.94 representing a growth rate of 19.0 percent.

For Mr. Ricks and Dr. Skovronsky, shares earned for the 2020-2021 performance period are subject to an additional 13-month service-vesting period and are shown in the table below as restricted stock units. Because she was not an executive officer when the award was granted, Ms. Ashkenazi's 2020-2021 performance award was paid in shares of Lilly common stock and not subject to the additional service-vesting period. Mr. Van Naarden was an employee of Loxo Oncology, Inc. and did not receive a performance award grant in 2020. Mr. Rau joined Lilly in May 2021, so he did not receive a performance award grant in 2020.
| | | | | | | | | | | |
Name3 | Target Shares | Shares Earned | RSUs |
| | | |
| Mr. Ricks | 27,306 | N/A | 47,786 |
| Ms. Ashkenazi | 2,731 | 4,779 | N/A |
| Dr. Skovronsky | 8,957 | N/A | 15,675 |
Mr. Van Naarden1 | N/A | N/A | N/A |
Mr. Rau2 | N/A | N/A | N/A |
1 Mr. Van Naarden was an employee of Loxo Oncology, Inc. and did not receive a performance award grant in 2020.
2 Mr. Rau joined Lilly in May 2021, so he did not receive a performance award grant in 2020.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley forfeited his entire 2020-2022 performance award. See "Agreement with Former Chief Financial Officer" for additional information.
2019-2021 Shareholder Value Award
The target stock price range of $132.73 to $147.37 (18.1 percent to 31.1 percent total stock price growth) for the 2019-2021 shareholder value award was set in 2019 based on a beginning stock price of $112.38, which was the average closing price for Lilly stock for all trading days in November and December 2018. The ending stock price of $260.57 represents stock price growth of 131.9 percent over the relevant three-year period resulting in a payout multiple of 1.50.
The relative TSR modifier applies to those individuals who were executive officers when the award was granted. The cumulative TSR median for the company’s peer group was 24.6 percent, and Lilly’s TSR over the same period was 145.7 percent. Given this positive relative performance, Lilly's relative TSR was 121.1 percentage points above the peer group
median resulting in a maximum award payout of 180 percent of target (SVA payout multiple of 150 percent multiplied by the 1.2 modifier = 180 percent final payout).
The number of shares paid to each of our named executive officers for the 2019-2021 performance period were as follows:
| | | | | | | | |
Name3 | Target Shares | Shares Paid Out |
| Mr. Ricks | 68,382 | 123,088 |
Ms. Ashkenazi1 | 3,633 | 5,450 |
| Dr. Skovronsky | 22,794 | 41,029 |
Mr. Van Naarden2 | N/A | N/A |
Mr. Rau2 | N/A | N/A |
1 The TSR modifier did not apply to Ms. Ashkenazi’s 2019-2021 shareholder value award payouts since she was not an executive officer at the time of grant.
2 Mr. Van Naarden was an employee of Loxo Oncology, Inc. in 2019 and so he did not receive a shareholder value award grant in 2019. Mr. Rau joined Lilly in May 2021, so he did not receive a shareholder value award grant in 2019.
3 Pursuant to the terms of his separation agreement with the company, Mr. Smiley forfeited his entire 2019-2021 shareholder value award. See "Agreement with Former Chief Financial Officer" for additional information.
Other Compensation Practices and Information
Employee Benefits
The company offers core employee benefits coverage to:
•provide our workforce with a reasonable level of financial support in the event of illness or injury
•provide post-retirement income
•enhance productivity and job satisfaction through benefit programs that focus on overall well-being.
The benefit programs available to executive officers are offered to all eligible U.S. employees and include benefits such as medical and dental coverage, disability insurance, and life insurance. In addition, the 401(k) Plan and The Lilly Retirement Plan (the Retirement Plan) are intended to provide U.S. employees a reasonable level of retirement income reflecting employees’ careers with the company. To the extent that any employee’s calculated retirement benefit exceeds Internal Revenue Service (IRS) limits for amounts that can be paid through a qualified plan, the company also offers a nonqualified pension plan and a nonqualified savings plan. These plans provide only the difference between the calculated benefits and the applicable IRS limits, and the formula is the same for all participating employees. The cost of employee benefits is partially borne by the employee, including each executive officer.
Perquisites
The company provides limited perquisites to executive officers. In response to the COVID-19 pandemic, the company considered various actions to promote the health and safety of its employees, including its named executive officers, recognizing that the company’s important objectives during this critical time would be significantly disadvantaged without
the full services of its employees. As part of this process, the company has encouraged Mr. Ricks’ personal use of the corporate aircraft (up to a maximum incremental cost of $60,000) as a means to (i) increase his time available for company business; and (ii) protect his health and safety. The incremental cost of personal use of the corporate aircraft is included as a perquisite in the Summary Compensation Table under the heading "All Other Compensation."
The Lilly Deferred Compensation Plan
Members of senior management may defer receipt of part or all of their cash compensation under The Lilly Deferred Compensation Plan (Deferred Compensation Plan), which allows executives to save for retirement in a tax-effective way at minimal cost to the company. Under this unfunded plan, amounts deferred by the executive are credited at an interest rate of 120 percent of the applicable federal long-term rate, as described in more detail following the "Nonqualified Deferred Compensation in 2021" table.
Severance Benefits
Except in the case of certain terminations following a change in control of the company, the company is generally not obligated to pay severance to executive officers upon termination of their employment; any such payments are at the discretion of the Compensation Committee.
The company has adopted change-in-control severance pay plans for nearly all employees, including executive officers. The plans are intended to preserve employee morale and productivity and encourage retention in the face of the disruptive impact of an actual or rumored change in control. In addition, the plans are intended to align executive and shareholder interests by enabling executives to evaluate corporate transactions that may be in the best interests of the shareholders and other constituents of the company without undue concern over whether the transactions may jeopardize the executives’ own employment.
Highlights of Our Change-in-Control Severance Plans
•all regular employees are covered
•double trigger required
•no tax gross-ups
•up to two-year pay protection
•18-month benefit continuation
Although benefit levels may differ depending on the employee’s job level and seniority, the basic elements of the plans are comparable for all eligible employees:
•Double trigger:Unlike "single trigger" plans that pay out immediately upon a change in control, our plans require a "double trigger"—a change in control followed by an involuntary loss of employment within two years. This is consistent with the plans' intent to provide employees with financial protection upon loss of employment. With respect to unvested equity, performance to the date of the change in control will be used to determine the number of shares earned under an award, but vesting does not accelerate immediately upon a change in control. Rather, the performance-adjusted awards will convert to time-based restricted stock units that continue to vest with the new company. Shares will pay out upon the earlier of the completion of the original award period; upon a covered termination; or if the successor entity does not assume, substitute, or otherwise replace the awards.
•Covered terminations: Employees are eligible for payments if, within two years of the change in control, their employment is terminated (i) without cause by the company or (ii) for good reason by the employee, each as defined in the plans. See "Compensation—Executive Compensation—Payments upon Termination or Change in Control" for a more detailed discussion, including a discussion of what constitutes a change in control.
•Employees who suffer a covered termination receive up to two years of pay and 18 months of benefits protection:These provisions ensure employees a reasonable period of protection of their income and core employee benefits.
•Severance payment. Eligible terminated employees would receive a severance payment ranging from six months' to two years’ base salary. Executives are all eligible for two years’ base salary plus two times the then-current year’s target bonus.
•Benefit continuation. Basic employee benefits such as health, dental, life insurance and long-term disability would continue for 18 months following termination of employment, unless the individual becomes eligible for coverage with a new employer. All employees would receive an additional two years of both age and years-of-service credit for purposes of determining eligibility for retiree medical and dental benefits.
•Accelerated vesting of equity awards: Any unvested equity awards would vest at the time of a covered termination.
•Excise tax:In some circumstances, the payments or other benefits received by the employee in connection with a change in control could exceed limits established under Section 280G of the Internal Revenue Code. The employee would then be subject to an excise tax on top of normal federal income tax. The company does not reimburse employees for these taxes. However, the amount of any change-in-control-related benefit will be reduced to the 280G limit if the effect would be to deliver a greater after-tax benefit than the employee would receive with an unreduced benefit.
Share Ownership and Retention Guidelines
Share ownership and retention guidelines help create direct alignment of interests between senior management and shareholders over the longer term. Lilly has established a formal share ownership policy under which the CEO and other senior executives are required to acquire and hold Lilly shares in an amount representing a multiple of base salary. During 2021, the Compensation Committee approved an increase to share ownership requirements for the CEO and the other named executive officers as follows:
| | | | | | | | |
| Previous Share Ownership Requirement | Current Share Ownership Requirement |
| CEO | 6X Base Salary | 12X Base Salary |
| Other NEOs | 4X Base Salary | 6X Base Salary |
Until ownership of the required number of shares is reached, an executive officer must hold 50 percent of all shares, net of tax, from all equity payouts. Executive officers are also required to hold all shares received from equity program payouts, net of taxes, for at least one year, even once share ownership requirements have been met. For performance awards granted to executive officers, this holding requirement is met by the 13-month service-vesting period after the end of the performance period.
All of the named executive officers are compliant with the share ownership guidelines. The following graphic shows each respective named executive officer's guideline and each named executive officer's holdings as of December 31, 2021, other than Mr. Smiley, who resigned effective as of February 9, 2021.
Prohibition on Hedging and Pledging Shares
Non-employee directors and employees, including executive officers, are not permitted to hedge their economic exposures to company stock through short sales or derivative transactions. Non-employee directors and employees, including executive officers, are prohibited from pledging any company stock (i.e., using company stock as collateral for a loan or trading shares on margin).
Executive Compensation Recovery Policy
Pursuant to our executive compensation recovery policy (the Executive Compensation Recovery Policy), all incentive awards are subject to forfeiture upon termination of employment prior to the end of the performance or vesting period or for disciplinary reasons. In addition, the Compensation Committee adopted updates to the Executive Compensation Recovery Policy to be effective January 1, 2022 (the Executive Compensation Recovery Policy, as updated and effective January 1, 2022, the Updated Executive Compensation Recovery Policy) which provides the Compensation Committee broad discretion to claw back non-qualified retirement benefits accrued beginning January 1, 2022 from any member of senior management whose misconduct results in a material violation of law or company policy that causes significant harm to the company or who fails in his or her supervisory responsibility to prevent such misconduct by others.
Pursuant to the Executive Compensation Recovery Policy, the company could recover all or a portion of any incentive compensation and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, the company could also recover non-qualified retirement benefits accrued beginning January 1, 2022 from an executive officer in the case of materially inaccurate financial statements or material errors in the performance calculation, whether such inaccuracies or errors result in a restatement and whether or not the executive officer has engaged in wrongful conduct.
The Executive Compensation Recovery Policy covers any incentive compensation awarded or paid to a member of senior management during the last three years and, effective as of January 1, 2022 pursuant to the Updated Executive Compensation Recovery Policy, covers non-qualified retirement benefits accrued beginning January 1, 2022. Subsequent changes in status, including retirement or termination of employment, do not affect the company’s rights to recover compensation under the policy.
The company will disclose decisions to take action pursuant to the Updated Executive Compensation Recovery Policy in its proxy materials when required by, and in compliance with, SEC rules and regulations and other applicable laws. In addition, when legally permissible to do so, the company will disclose a decision to take action under this policy when the facts and circumstances of the matter that triggered application of the policy have been publicly disclosed in the company’s filings with the SEC and where disclosure can be made without prejudicing the company and its shareholders.
The principles of our robust Updated Executive Compensation Recovery Policy are also incorporated into the terms of our incentive plans and award agreements, which, in the event of misconduct meeting the standards described above, allow the Compensation Committee to reduce or cancel awards or payouts that would otherwise have been earned based on company performance. Action by the Compensation Committee to reduce or cancel awards or payouts can occur during or following the relevant performance period.
Executive Compensation
Summary Compensation Table
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name and Principal Position | Year | Salary
($) | | Bonus
($) | | Stock Awards
($) 1 | | Non-Equity Incentive Plan Compensation
($) 2 | | Change in
Pension Value
($) 3 | | All Other Compensation
($) 4 | | Total Compensation
($) |
| David A. Ricks | 2021 | $1,500,000 | | | $0 | | $ | 14,966,000 | | | $2,475,000 | | | $2,442,235 | | $126,750 | | $21,509,985 | |
Chair, President, and
Chief Executive Officer | 2020 | $1,483,333 | | | $0 | | $ | 13,587,500 | | | $2,625,500 | | | $5,883,924 | | $128,372 | | $23,708,629 | |
| 2019 | $1,400,000 | | | $0 | | $ | 12,222,000 | | | $2,919,000 | | | $4,658,242 | | $84,000 | | $21,283,242 | |
| Anat Ashkenazi | 2021 | $850,432 | | | $0 | | $2,453,000 | | 7 | $907,816 | | | $395,335 | | $51,026 | | $4,657,609 | |
| Senior Vice President and Chief Financial Officer | 2020 | N/A | | N/A | | N/A | 7 | N/A | | N/A | | N/A | | N/A |
| 2019 | N/A | | N/A | | N/A | 7 | N/A | | N/A | | N/A | | N/A |
| Daniel M. Skovronsky, M.D., Ph.D. | 2021 | $1,000,000 | | | $0 | | $5,024,300 | | | $1,100,000 | | | $342,792 | | $60,000 | | $7,527,092 | |
Senior Vice President,
Chief Scientific and Medical Officer and President, Lilly Research Laboratories | 2020 | $983,333 | | | $0 | | $4,456,700 | | | $1,102,317 | | | $751,223 | | $58,539 | | $7,352,112 | |
| 2019 | $900,000 | | | $0 | | $4,074,000 | | | $1,188,450 | | | $446,521 | | $54,000 | | $6,662,971 | |
| Jacob Van Naarden | 2021 | $745,649 | | 5 | $218,715 | | 6 | $4,099,500 | | | $1,029,507 | | | $16,909 | | $12,462 | | | $6,122,742 | |
| Senior Vice President, CEO Loxo Oncology at Lilly and President, Lilly Oncology | 2020 | N/A | | N/A | | N/A | | N/A | | N/A | | N/A | | N/A |
| 2019 | N/A | | N/A | | N/A | | N/A | | N/A | | N/A | | N/A |
Diogo Rau7 | 2021 | $412,308 | | | $250,000 | 8 | $4,000,000 | | $362,831 | | | $44,519 | | $31,670 | | $5,101,328 | |
| Senior Vice President and Chief Information and Digital Officer | 2020 | N/A | | N/A | | N/A | | N/A | | N/A | | N/A | | N/A |
| 2019 | N/A | | N/A | | N/A | | N/A | | N/A | | N/A | | N/A |
| Joshua L. Smiley | 2021 | $230,831 | | | $0 | | $0 | 10 | $0 | 9 | $0 | | $13,850 | | | $244,681 | |
| Former Senior Vice President and Chief Financial Officer | 2020 | $983,333 | | | $0 | | $3,587,100 | | 10 | $0 | 9 | $2,685,276 | | $58,539 | | | $7,314,248 | |
| 2019 | $895,833 | | | $0 | | $3,142,800 | | 10 | $1,182,948 | | | $2,073,070 | | $53,750 | | | $7,348,401 | |
1 This column shows the grant date fair value of performance awards, shareholder value awards and relative value awards for all named executive officers and additional restricted stock unit awards for Mr. Van Naarden and Mr. Rau computed in accordance with FASB ASC Topic 718. See Note 12 of the consolidated financial statements in the company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2021 for additional detail regarding assumptions underlying the valuation of equity awards. All values in the "Stock Awards" column were based upon the probable outcome of performance conditions as of the grant date, which vary year to year.
For purposes of comparison, the supplemental table below shows the total target grant values of stock awards approved by the Compensation Committee:
| | | | | | | | | | | | | | | | | | | | |
| Name | 2019 Total Equity | 2020 Total Equity | 2021 Total Equity |
| Mr. Ricks | $10,500,000 | | $12,500,000 | | $14,000,000 | |
| Ms. Ashkenazi | N/A | * | N/A | * | $2,200,000 | |
| Dr. Skovronsky | $3,500,000 | | $4,100,000 | | $4,700,000 | |
| Mr. Van Naarden | N/A | * | N/A | * | $4,099,500 | ** |
| Mr. Rau | N/A | * | N/A | * | $4,000,000 | *** |
| Mr. Smiley | $2,700,000 | **** | $3,300,000 | **** | $0 | **** |
|
| *Ms. Ashkenazi, Mr. Van Naarden and Mr. Rau were not executive officers in 2019 and 2020. |
| **Mr. Van Naarden's total equity includes $1,300,000 non-executive officer annual equity grant when he held his former role with Loxo Oncology, Inc. and $2,650,000 of restricted stock units ($1,650,000 granted in February 2021 representing transition equity following Lilly’s acquisition of Loxo Oncology, Inc.and $1,000,000 granted in December 2021 associated with Mr. Van Naarden's promotion). |
| ***Mr. Rau received a sign-on restricted stock unit to replace a portion of the outstanding equity he forfeited after leaving Apple and the missed opportunity for a 2021 annual equity award from Lilly. |
| ****As a result of Mr. Smiley's resignation as an officer on February 9, 2021, his 2019 and 2020 equity awards were cancelled and he did not receive an equity grant in 2021. |
The table below shows the minimum, target, and maximum payouts (valuing the number of shares that would vest at each payout level using the grant date fair value of a share of Lilly common stock on the date of grant) for the 2021-2023 performance award included in the Summary Compensation Table, which will pay out in February 2024.
| | | | | | | | | | | |
| Name | Minimum Payout | Target Payout | Maximum Payout |
| Mr. Ricks | $0 | $4,200,000 | $7,350,000 |
| Ms. Ashkenazi | $0 | $1,100,000 | $1,925,000 |
| Dr. Skovronsky | $0 | $1,410,000 | $2,467,500 |
| Mr. Van Naarden | $0 | $650,000 | $1,137,500 |
| Mr. Rau* | N/A | N/A | N/A |
| Mr. Smiley** | N/A | N/A | N/A |
|
| *Mr. Rau joined Lilly in May 2021 and did not receive a 2021-2023 performance award grant. |
| **As a result of Mr. Smiley's resignation as an officer on February 9, 2021, Mr. Smiley did not receive a 2021-2023 performance award grant. |
The table below shows the minimum, target, and maximum payouts (valuing the number of shares that would vest at each payout level using the grant date fair value of a share of Lilly common stock on the date of grant) for the 2021-2023 shareholder value award included in the Summary Compensation Table, which will pay out in February 2024.
| | | | | | | | | | | |
| Name | Minimum Payout | Target Payout | Maximum Payout |
| Mr. Ricks | $0 | $4,900,000 | $8,575,000 |
| Ms. Ashkenazi | $0 | $550,000 | $962,500 |
| Dr. Skovronsky | $0 | $1,645,000 | $2,878,750 |
| Mr. Van Naarden | $0 | $325,000 | $568,750 |
| Mr. Rau* | N/A | N/A | N/A |
| Mr. Smiley** | N/A | N/A | N/A |
|
| *Mr. Rau joined Lilly in May 2021 and did not receive a 2021-2023 shareholder value award grant. |
| **As a result of Mr. Smiley's resignation as an officer on February 9, 2021, Mr. Smiley did not receive a 2021-2023 shareholder value award grant. |
The table below shows the minimum, target, and maximum payouts (valuing the number of shares that would vest at each payout level using the grant date fair value of a share of Lilly common stock on the date of grant) for the 2021-2023 relative value award included in the Summary Compensation Table, which will pay out in February 2024.
| | | | | | | | | | | |
| Name | Minimum Payout | Target Payout | Maximum Payout |
| Mr. Ricks | $0 | $4,900,000 | $8,575,000 |
| Ms. Ashkenazi | $0 | $550,000 | $962,500 |
| Dr. Skovronsky | $0 | $1,645,000 | $2,878,750 |
| Mr. Van Naarden | $0 | $325,000 | $568,750 |
| Mr. Rau* | N/A | N/A | N/A |
| Mr. Smiley** | N/A | N/A | N/A |
|
| *Mr. Rau joined Lilly in May 2021 and did not receive a 2021-2023 relative value award grant. |
| ** As a result of Mr. Smiley's resignation as an officer on February 9, 2021, Mr. Smiley did not receive a 2021-2023 relative value award grant. |
2 Payments under the Bonus Plan for performance in the years represented. For Mr. Van Naarden, the amount in this column reflects payments under the Bonus Plan and the Loxo Bonus Plan for 2021.
3 The amounts in this column reflect the change in pension value for each individual, calculated by an actuary. The changes in pension values in 2021 were driven by pay changes, an additional year of credited service, offset by changes in actuarial assumptions. The design of the pension benefit plan did not change. See the Pension Benefits in 2021 table below for information about the standard actuarial assumptions used. No named executive officer received preferential or above-market earnings on deferred compensation.
4 The amounts in this column are company matching contributions into each individual's 401(k) and nonqualified savings plan contributions. For Mr. Ricks, the amount in this column reflects $90,000 of company matching contributions and nonqualified savings plan contributions and $36,750 of aggregate incremental cost for his personal use of the corporate aircraft. The aggregate incremental costs for personal use of our aircraft are calculated based on our variable operating costs, which include crew travel expenses, on-board catering, landing fees, trip-related hangar/parking costs, fuel, trip-related maintenance, and other smaller variable costs. Because the vast majority of the use of the corporate aircraft is for business purposes, fixed costs such as aircraft purchase costs, maintenance not related to personal trips, and flight crew salaries are not included. For Mr. Rau, the amount in this column reflects $18,914 of company matching contributions and nonqualified savings plan contributions and $12,756 of moving expense reimbursements. The company does not reimburse executives for taxes outside of the limited circumstance of taxes related to a domestic employee relocation or a prior international assignment.
5 For Mr. Van Naarden, this column reflects $668,399 in salary payments and $77,250 of unused vacation pay from Loxo Oncology, Inc.
6 This column includes the amount which represents a one-time cash payment of $218,715 in lieu of compensation and benefits Mr. Van Naarden was no longer eligible to receive following his transition to his new role, including compensation for the difference between the bonus he would have earned under the Loxo Bonus Plan and the Lilly Bonus Plan, and changes in his expected out-of-pocket costs for healthcare expenses.
7 Mr. Rau joined Lilly in May 2021, and the amount in this column reflects the pro-rated annual incentive paid for the portion of the year for which he was employed.
8 Mr. Rau received a cash sign-on award of $250,000 to replace a portion of the outstanding equity with near-term vesting provisions that he forfeited after leaving Apple and to cover other incidental expenses associated with his transition to the role with Lilly.
9 As a result of Mr. Smiley’s resignation as an officer on February 9, 2021, he did not receive a bonus payout for 2020 or 2021.
10 As a result of Mr. Smiley’s resignation as an officer on February 9, 2021, his 2019 and 2020 equity awards were cancelled and he did not receive a 2021 equity grant. Mr. Smiley realized $0 from these awards.
Grants of Plan-Based Awards During 2021
The compensation plans under which the grants in the following table were made are described in the CD&A above and consist of the Bonus Plan (a non-equity incentive plan) and the Amended and Restated 2002 Lilly Stock Plan which provides for performance awards, shareholder value awards, relative value awards and restricted stock units, among others.
To receive a payout under the performance award, the shareholder value award and the relative value award issued annually to executive officers, the participant must remain employed with the company through the end of the relevant award period, retire after December 31 of the year in which the award was issued or be subject to termination events outside of the control of the employee (death, disability, plant closing or reduction in workforce). Annual awards issued to executive officers are also generally subject to a one-year non-compete post company exit, other than the award of restricted stock units granted to Mr. Van Naarden on December 28, 2021, which does not include a non-compete covenant. The annual awards issued to executive officers (other than Mr. Van Naarden's award of restricted stock units granted on December 28, 2021) provide that if the former executive officer works for a direct competitor within the one-year restriction period, all annual cycle awards issued during the 2021 annual cycle or later would be forfeited. Awards issued to employees who were not executive officers at the time of grant generally do not include the non-compete or vesting into retirement provisions.
No dividends accrue on either performance awards, shareholder value awards, or relative value awards during the performance period. During the performance award 13-month service-vesting period and restricted stock unit award restriction period, non-preferential dividends accrue and are paid upon vesting.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | Award | | Grant Date2 | Compensation Committee Action Date | Estimated Possible Payouts
Under Non-Equity
Incentive Plan Awards 1 | Estimated Possible and Future
Payouts Under Equity
Incentive Plan Awards | All Other
Stock or Option Awards:
Number of
Shares of Stock,
Options, or Units | Grant Date
Fair Value
of Equity
Awards |
Threshold
($) | Target
($) | Maximum
($) | Threshold
(# shares) | Target
(# shares) | Maximum
(# shares) |
| Mr. Ricks | | __ | __ | $562,500 | $2,250,000 | $4,500,000 | | | | | |
| 2021-2023 PA | 3 | 2/8/2021 | 12/11/2020 | | | | 10,576 | | 21,151 | | 37,014 | | | $ | 5,166,000 | |
| 2021-2023 SVA | 4 | 2/8/2021 | 12/11/2020 | | | | 11,271 | | 22,541 | | 39,447 | | | $ | 4,900,000 | |
| 2021-2023 RVA | 5 | 2/8/2021 | 12/11/2020 | | | | 8,545 | | 17,090 | | 29,908 | | | $ | 4,900,000 | |
| | | | | | | | | | | — | |
| Ms. Ashkenazi | | __ | __ | $206,322 | $825,287 | $1,650,575 | | | | | |
| 2021-2023 PA | 3 | 2/8/2021 | 12/11/2020 | | | | 2,770 | | 5,540 | | 9,695 | | | $ | 1,353,000 | |
| 2021-2023 SVA | 4 | 2/8/2021 | 12/11/2020 | | | | 1,176 | | 2,351 | | 4,114 | | | $ | 550,000 | |
| 2021-2023 RVA | 5 | 2/8/2021 | 12/11/2020 | | | | 959 | | 1,918 | | 3,357 | | | $ | 550,000 | |
| | | | | | | | | | | — | |
| Dr. Skovronsky | | __ | __ | $250,000 | $1,000,000 | $2,000,000 | | | | | |
| 2021-2023 PA | 3 | 2/8/2021 | 12/11/2020 | | | | 3,551 | | 7,101 | | 12,427 | | | $ | 1,734,300 | |
| 2021-2023 SVA | 4 | 2/8/2021 | 12/11/2020 | | | | 3,784 | | 7,567 | | 13,242 | | | $ | 1,645,000 | |
| 2021-2023 RVA | 5 | 2/8/2021 | 12/11/2020 | | | | 2,869 | | 5,738 | | 10,042 | | | $ | 1,645,000 | |
| | | | | | | | | | | — | |
| Mr. Van Naarden | | __ | __ | $111,164 | $444,655 | $1,429,696 | | | | | |
| 2021-2023 PA | 3 | 2/8/2021 | 12/11/2020 | | | | 1,637 | | 3,273 | | 5,728 | | | $ | 799,500 | |
| 2021-2023 SVA | 4 | 2/8/2021 | 12/11/2020 | | | | 695 | | 1,389 | | 2,431 | | | $ | 325,000 | |
| 2021-2023 RVA | 5 | 2/8/2021 | 12/11/2020 | | | | 567 | | 1,134 | | 1,985 | | | $ | 325,000 | |
| RSU | 6 | 2/8/2021 | 10/15/2020 | | | | | | | 5,461 | | $ | 1,100,000 | |
| RSU | 6 | 2/8/2021 | 10/15/2020 | | | | | | | 2,785 | | $ | 550,000 | |
| RSU | 6 | 12/28/2021 | 12/21/2021 | | | | | | | 3,618 | | $ | 1,000,000 | |
| | | | | | | | | | | | |
| Mr. Rau | | | __ | __ | $82,462 | $329,846 | $659,692 | | | | | |
| RSU | 7 | 6/1/2021 | 3/10/2021 | | | | | | | 20,160 | | $ | 4,000,000 | |
1 These columns show the threshold, target and maximum payouts for performance under the Bonus Plan. For Mr. Van Naarden, the columns show the threshold, target and maximum payouts for performance under both the Bonus Plan and the Loxo Bonus, pro-rated for the portion of the year he was participating in each plan. The Bonus Plan payouts range from 0 to 200 percent of target and the Loxo Bonus payouts range from 0 to 400 percent of target. The Bonus Plan and Loxo Bonus payments for 2021 performance were 110 percent of target and 310 percent of target, respectively. Actual payouts are shown in the Summary Compensation Table in the column titled "Non-Equity Incentive Plan Compensation."
2 To assure grant timing is not manipulated for employee gain, the annual grant date is established in advance by the Compensation Committee.
3 This row shows the possible payouts for the 2021-2023 performance awards ranging from 0 to 175 percent of target. This performance award will pay out in February 2024. Ms. Ashkenazi and Mr. Van Naarden were not executive officers at the time of grant and, therefore, their performance awards will not be subject to the 13-month service vesting period and will payout in February 2023.
4 This row shows the range of payouts for the 2021-2023 shareholder value awards. This shareholder value award will pay out in February 2024, with payouts ranging from 0 to 175 percent of target. We measure the fair value of the shareholder value award on the grant date using a Monte Carlo simulation model.
5 This row shows the range of payouts for the 2021-2023 relative value awards. This relative value award will pay out in February 2024, with payouts ranging from 0 to 175 percent of target. We measure the fair value of the relative value award on the grant date using a Monte Carlo simulation model.
6 These grants were made outside of the normal annual cycle in 2021, and 5,461 units will vest on February 16, 2022, 2,785 units will vest on February 16, 2023, and 3,618 units will vest on February 1, 2024.
7 This grant was made outside of the normal annual cycle in 2021, and 7,560 units will vest on July 1, 2022, 6,300 units will vest on July 1, 2023 and 6,300 units will vest on July 1, 2024.
Outstanding Equity Awards on December 31, 2021
The 2021 closing stock price used to calculate the values in the table below was $276.22.
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | |
| | | Stock Awards |
| Name | Award | | Number of Shares or Units of Stock That Have Not Vested (#) | Market Value of Shares or Units of Stock That Have Not Vested ($) | Equity Incentive Plan Awards: Number of Unearned Shares, Units, or Other Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units, or Other Rights That Have Not Vested ($) |
| Mr. Ricks | 2021-2023 SVA | | | | | 39,447 | 1 | $10,896,050 |
| 2020-2022 SVA | | | | | 59,063 | 2 | $16,314,382 |
| 2021-2023 RVA | | | | | 17,090 | 3 | $4,720,600 |
| 2020-2022 RVA | | | | | 24,319 | 4 | $6,717,394 |
| 2021-2023 PA | | | | | 37,014 | 5 | $10,224,007 |
| 2020-2022 PA | | 47,786 | 6 | $13,199,449 | |
| |
| 2019-2021 PA | | 56,205 | 7 | $15,524,945 | |
| |
| Ms. Ashkenazi | 2121-2023 SVA | | | | | 4,114 | 1 | $1,136,369 |
| 2020-2022 SVA | | | | | 2,305 | 2 | $636,687 |
| 2021-2023 RVA | | | | | 1,918 | 3 | $529,790 |
| 2020-2022 RVA | | | | | 1,042 | 4 | $287,821 |
| 2021-2023 PA | | | | | 9,695 | 5 | $2,677,953 |
| RSU | | 11,499 | 8 | $3,176,254 | | | |
| Dr. Skovronsky | 2021-2023 SVA | | | | | 13,242 | 1 | $3,657,705 |
| 2020-2022 SVA | | | | | 19,373 | 2 | $5,351,210 |
| 2021-2023 RVA | | | | | 5,738 | 3 | $3,657,705 |
| 2020-2022 RVA | | | | | 7,977 | 4 | $2,203,407 |
| 2021-2023 PA | | | | | 12,427 | 5 | $3,432,586 |
| 2020-2022 PA | | 15,675 | 6 | $4,329,749 | | | |
| 2019-2021 PA | | 18,735 | 7 | $5,174,982 | | | |
| Mr. Van Naarden | 2021-2023 SVA | | | | | 2,431 | 1 | $671,491 |
| 2021-2023 RVA | | | | | 1,134 | 3 | $671,491 |
| 2021-2023 PA | | | | | 5,728 | 5 | $1,582,188 |
| RSU | | 11,864 | 9 | $3,277,074 | | | |
| Mr. Rau | RSU | | 20,160 | 10 | $5,568,595 | | | |
| Mr. Smiley | | | 0 | | $0 | 0 | | $0 |
1 Shareholder value awards granted for the 2021-2023 performance period will vest on December 31, 2023. The number of shares reported reflects the maximum payout, which will be made if the average closing stock price in November and December 2023 is over $239.03. Actual payouts may vary from 0 to 175 percent of target. Net shares from any payout must be held by executive officers for a minimum of one year. Had the performance period ended December 31, 2021, the payout would have been at 175 percent of target.
2 Shareholder value awards granted for the 2020-2022 performance period will vest on December 31, 2022. The number of shares reported reflects the maximum payout, which will be made if the average closing stock price in November and December 2022 is over $187.17. Actual payouts may vary from 0 to 180 percent of target. Net shares from any payout must be held by executive officers for a minimum of one year. Had the performance period ended December 31, 2021, the payout would have been 175 percent of target.
3 Relative value awards granted for the 2021-2023 performance period will vest on December 31, 2023. The number of shares reported reflects the target payout, which will be paid if Lilly's absolute TSR is at or up to 5.9 percentage points above the actual peer median TSR. Net shares from any payout must be held by executive officers for a minimum of one year. Had the performance period ended December 31, 2021, the payout would have been 175 percent of target.
4 Relative value awards granted for the 2020-2022 performance period will vest on December 31, 2022. The number of shares reported reflects the target payout, which will be paid if Lilly's absolute TSR is at or up to 5.9 percentage points above the actual peer median TSR. Net shares from any payout must be held by executive officers for a minimum of one year. Had the performance period ended December 31, 2021, the payout would have been 175 percent of target.
5 This number represents the maximum value of performance award shares that could pay out for the 2021-2023 performance awards, provided performance goals are met. Once the combined cumulative EPS result and associated payout level are determined at the end of the 2021-2022 performance period, the associated number of shares will be granted as restricted stock units, vesting in February 2024. Actual payouts may vary from 0 to 175 percent of target. The number of shares recorded in the table reflects the payout if the combined cumulative EPS for 2021 and 2022 is at least $18.08 under the 2021-2023 performance award grid (see "Compensation Discussion and Analysis—Performance Goals for 2021 Incentive Programs—2021-2023 Performance Award"). Ms. Ashkenazi and Mr. Van Naarden were not executive officers at the time of grant and, therefore, their 2021-2023 performance awards will not be subject to the 13-month service vesting period and will payout in February 2023.
6 The performance period ended December 31, 2021, for the 2020-2022 performance award, resulting in the issuance of restricted stock units for 175 percent of target shares for Mr. Ricks and Dr. Skovronsky. These restricted stock units will vest in February 2023.
7 For Mr. Ricks and Dr. Skovronsky, restricted stock units vested from the 2019-2021 performance award on February 1, 2022.
8 These grants were made outside of the normal annual cycle in 2020, and 4,600 units will vest on November 1, 2024 and 6,899 units will vest on November 1, 2026.
9 These grants were made outside of the normal annual cycle in 2021, and 5,461 units will vest on February 16, 2022, 2,785 units will vest on February 16, 2023, and 3,618 units will vest on February 1, 2024.
10 This grant was made outside of the normal annual cycle in 2021. A total of 7,560 units will vest on July 1, 2022, 6,300 units will vest on July 1, 2023 and 6,300 units will vest on July 1, 2024.
Options Exercised and Stock Vested in 2021
| | | | | | | | | | | | | | | | | | | | | | | |
| Option Awards | | Stock Awards |
| Name | Number of Shares Acquired on Exercise (#) | | Value-Realized on Exercise ($) | Number of Shares Acquired on Vesting (#) | Value Realized on Vesting ($)1 |
| Mr. Ricks | 0 | | $0 | | 75,387 | | 2 | $15,678,234 |
| | 123,088 | | 3 | $29,943,618 |
| Ms. Ashkenazi | 0 | | $0 | | 5,450 | | 3 | $1,325,822 |
| | 4,779 | | 4 | $1,162,587 |
| Dr. Skovronsky | 0 | | $0 | | 41,029 | | 3 | $9,981,125 |
| Mr. Van Naarden | 0 | | $0 | | 7,929 | | 5 | $1,624,573 |
Mr. Rau6 | 0 | | $0 | | 0 | | $0 |
| Mr. Smiley | 0 | | $0 | | 19,266 | | 2 | $4,006,750 |
1 Amounts reflect the market value of Lilly stock on the day value is realized.
2 Restricted stock units resulting from the 2018-2020 performance award that vested in February 2021.
3 Payout of the 2019-2021 shareholder value award at 150 percent of target, adjusted by Lilly’s three-year cumulative TSR (145.7 percent) relative to its peer companies’ median cumulative TSR of 24.6 percent, resulting in a maximum TSR modifier of 20 percent and a final payout of 180 percent of target. Since Ms. Ashkenazi was not an executive officer when the 2019-2021 shareholder value award was granted, her award was not subject to the TSR modifier. As a result, her payout multiple was 150 percent of target.
4 Payout of the 2020-2022 performance award at 175% of target for Ms. Ashkenazi. She was not an executive officer in 2020 at the time of grant; therefore, no additional 13-month service-vesting period applied.
5 This grant was made in 2020 before Mr. Van Naarden became an executive officer.
6 Mr. Rau joined Lilly in 2021 and had no option exercise or stock awards vest during 2021.
Retirement Benefits
We provide retirement income to eligible U.S. employees, including executive officers, through the following plans:
•The 401(k) Plan, a defined contribution plan qualified under Sections 401(a) and 401(k) of the Internal Revenue Code. Participants may elect to contribute a portion of their base salary to the plan, and the company provides matching contributions on employees’ contributions up to 6 percent of base salary up to IRS limits. The employee contributions, company contributions, and earnings thereon are paid out in accordance with elections made by the participant and the terms of the plan. See the "All Other Compensation" column in the Summary Compensation Table for information about company contributions under the 401(k) Plan for the named executive officers.
•The Retirement Plan, a tax-qualified defined benefit plan that provides monthly benefits to retirees. See the Pension Benefits in 2021 table below for additional information about the value of these pension benefits.
Sections 401 and 415 of the Internal Revenue Code generally limit the amount of annual pension that can be paid from a tax-qualified plan ($230,000 in 2021 and $245,000 in 2022) as well as the amount of annual earnings that can be used to calculate a pension benefit ($290,000 in 2021 and $305,000 in 2022). However, since 1975 the company has maintained a nonqualified pension plan that pays eligible retirees the difference between the amount payable under the Retirement Plan and the amount they would have received without the Internal Revenue Code limits. The nonqualified pension plan is unfunded and subject to forfeiture in the event of bankruptcy. Likewise, the company maintains a nonqualified savings plan that allows participants to contribute up to 6 percent of base salary exceeding the IRS limit. The company matches these contributions in the same manner as described in the 401(k) Plan. For more information, see footnote 2 to the "Nonqualified Deferred Compensation in 2021" table.
The following table shows benefits that the named executive officers have accrued under the Retirement Plan and the nonqualified pension plan.
Pension Benefits in 2021
| | | | | | | | | | | | | | |
| Name | Plan | Number of Years of Credited Service | Present Value of
Accumulated Benefit ($) 1 | Payments During
Last Fiscal Year ($) |
| Mr. Ricks | retirement plan (pre-2010) | 14 | $827,458 | |
| retirement plan (post-2009) | 12 | $455,777 | |
| nonqualified plan (pre-2010) | 14 | $11,555,253 | |
| nonqualified plan (post-2009) | 12 | $6,246,306 | |
| total | | $19,084,794 | $0 |
| Ms. Ashkenazi | retirement plan (pre-2010) | 9 | $473,739 | |
| retirement plan (post-2009) | 12 | $396,301 | |
| retirement plan (post-2009) | 9 | $1,015,918 | |
| nonqualified plan (post-2009) | 12 | $856,088 | |
| total | | $2,742,046 | $0 |
| Dr. Skovronsky | retirement plan (post-2009) | 9 | $294,986 | |
| nonqualified plan (post-2009) | 9 | $1,676,195 | |
| total | | $1,971,181 | $0 |
| Mr. Van Naarden | retirement plan (post-2009) | 0.3 | $7,555 | |
| nonqualified plan (post-2009) | 0.3 | $9,354 | |
| total | | $16,909 | $0 |
| Mr. Rau | retirement plan (post-2009) | 1 | $30,790 | |
| nonqualified plan (post-2009) | 1 | $13,729 | |
| total | | $44,519 | $0 |
| Mr. Smiley | retirement plan (pre-2010) | 14 | $877,998 | |
| retirement plan (post-2009) | 12 | $408,343 | |
| retirement plan (post-2009) | 14 | $4,002,852 | |
| nonqualified plan (post-2009) | 12 | $1,832,029 | |
| total | | $7,121,222 | $0 |
1 The following standard actuarial assumptions were used to calculate the present value of each individual’s accumulated pension benefit:
| | | | | |
| Discount rate: | 3.11 percent for the qualified plan and 2.91 percent for nonqualified plan |
| Mortality (post-retirement decrement only): | Private 2012 white collar table with generational projection using Scale MP-2021 |
| Pre-2010 joint and survivor benefit (% of pension): | 50 percent until age 62; 25 percent thereafter |
| Post-2009 benefit payment form: | Life annuity |
The Retirement Plan benefits shown in the table are net present values. The benefits are not payable as a lump sum; they are generally paid as a monthly annuity for the life of the retiree and, if elected, any qualifying survivor. The annual benefit under the Retirement Plan is calculated using years of service and the average of the annual earnings (salary plus bonus) for the highest five out of the last ten calendar years of service (final average earnings).
Post-2009 Plan Information:Following amendment of our Retirement Plan formulas, employees first hired on or after February 1, 2008, have accrued retirement benefits only under the new plan formula. Employees hired before that date have accrued benefits under both the old and new plan formulas. All eligible employees, including those hired on or after February 1, 2008, can currently retire at age 65 with at least five years of service and receive an unreduced benefit. The annual benefit under the new plan formula is equal to 1.2 percent of final average earnings multiplied by years of service. Early retirement benefits under this plan formula are reduced six percent for each year under age 65. Transition benefits were afforded to employees with 50 points (age plus service) or more as of December 31, 2009. These benefits were intended to ease the transition to the new retirement formula for those employees who were closer to retirement or had been with the company longer at the time the plan was changed. For the transition group, early retirement benefits are reduced three percent for each year from age 65 to age 60 and six percent for each year under age 60. Mr. Ricks, Ms. Ashkenazi and Mr. Smiley are in this transition group.
Pre-2010 Plan Information:Employees hired prior to February 1, 2008 accrued benefits under both plan formulas. For these employees, benefits that accrued before January 1, 2010 were calculated under the old plan formula. The amount of the benefit is calculated using actual years of service through December 31, 2009, while total years of service are used to determine eligibility and early retirement reductions. The benefit amount is increased (but not decreased) proportionately based on final average earnings at termination compared to final average earnings at December 31, 2009. Full retirement benefits are earned by employees with 90 or more points (the sum of his or her age plus years of service). Employees
electing early retirement receive reduced benefits as described below:
•The benefit for employees with between 80 and 90 points is reduced by three percent for each year before the earlier of 90 points or age 62.
•The benefit for employees who have fewer than 80 points, but who reached age 55 and have at least ten years of service, is reduced as described above and is further reduced by six percent for each year before the earlier of 80 points or age 65.
Nonqualified Deferred Compensation in 2021
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | Plan | Executive
Contributions in
Last Fiscal Year
($)1 | Registrant
Contributions in
Last Fiscal Year
($)2 | Aggregate Earnings in Last Fiscal Year ($) | Aggregate Withdrawals/ Distributions in Last Fiscal Year ($) | Aggregate
Balance at Last
Fiscal Year End
($)3 |
| Mr. Ricks | nonqualified savings | $72,600 | | | $72,600 | | $1,140,638 | | | $0 | | $3,131,383 | | |
| deferred compensation | $0 | | | $0 | | $0 | | | $0 | | $0 | | |
| total | $72,600 | | | $72,600 | | $1,140,638 | | | $0 | | $3,131,383 | | |
| Ms. Ashkenazi | nonqualified savings | $33,626 | | | $33,626 | | $20,659 | | | $0 | | $212,556 | | |
| deferred compensation | $0 | | | $0 | | $0 | | | $0 | | $0 | | |
| total | $33,626 | | | $33,626 | | $20,659 | | | $0 | | $212,556 | | |
| Dr. Skovronsky | nonqualified savings | $42,600 | | | $42,600 | | $174,864 | | | $0 | | $920,647 | | |
| deferred compensation | $0 | | | $0 | | $0 | | | $0 | | $0 | | |
| total | $42,600 | | | $42,600 | | $174,864 | | | $0 | | $920,647 | | |
| Mr. Van Naarden | nonqualified savings | $12,462 | | $12,462 | $544 | | $0 | $25,467 | |
| deferred compensation | $0 | | $0 | $0 | | $0 | $0 | |
| total | $12,462 | | $12,462 | $544 | | $0 | $25,467 | |
| Mr. Rau | nonqualified savings | $7,339 | | $7,339 | $229 | | $0 | $14,906 | |
| deferred compensation | $0 | | $0 | $0 | | $0 | $0 | |
| total | $7,339 | | $7,339 | $229 | | $0 | $14,906 | |
| Mr. Smiley | nonqualified savings | $0 | | $0 | $189,951 | | $0 | $802,988 | |
| deferred compensation | $0 | | $0 | $0 | | $0 | $0 | |
| total | $0 | | $0 | $189,951 | | $0 | $802,988 | |
1 The amounts in this column are also included in the Summary Compensation Table in the "Salary" column (nonqualified savings) or the "Non-Equity Incentive Plan Compensation" column (deferred compensation).
2 The amounts in this column are also included in the Summary Compensation Table in the "All Other Compensation" column as a portion of the savings plan match.
3 Of the totals in this column, the following amounts have previously been reported in the Summary Compensation Table for this year and for previous years:
| | | | | | | | | | | |
| Name | 2021 ($) | Previous Years ($) | Total ($) |
| Mr. Ricks | $145,200 | $547,415 | $692,615 |
| Ms. Ashkenazi | $67,252 | N/A | $67,252 |
| Dr. Skovronsky | $85,200 | $224,777 | $309,977 |
| Mr. Van Naarden | $24,923 | N/A | $24,923 |
| Mr. Rau | $14,677 | N/A | $14,677 |
| Mr. Smiley | $0 | $228,777 | $228,777 |
The Nonqualified Deferred Compensation in 2021 table above shows information about two company programs: the nonqualified savings plan and the Deferred Compensation Plan. The nonqualified savings plan is designed to allow each employee to contribute up to 6 percent of his or her base salary and receive a company match, beyond the contribution limits prescribed by the IRS with regards to 401(k) plans. This plan is administered in the same manner as the 401(k) Plan, with the same participation and investment elections. Executive officers and other U.S. executives may also defer receipt of all or part of their cash compensation under the Deferred Compensation Plan. Amounts deferred by executives under this plan are credited with interest at 120 percent of the applicable federal long-term rate as established the preceding December by the U.S. Treasury Department under Section 1274(d) of the Internal Revenue Code with monthly compounding, which was 1.6 percent for 2021 and is 2.3 percent for 2022. Participants may elect to receive the funds in a lump sum or in up to 10 annual installments following termination of employment but may not make withdrawals while employed by the company, except in the event of hardship as approved by the Compensation Committee. All deferral elections and associated
distribution schedules are irrevocable. Both plans are unfunded and subject to forfeiture in the event of company bankruptcy.
Payments upon Termination or Change in Control (as of December 31, 2021)
The following table describes the potential payments and benefits under the company’s compensation and benefit plans and arrangements to which the named executive officers would be eligible upon qualifying termination of employment. Except for certain terminations following a change in control of the company, as described below, there are no agreements, arrangements, or plans that entitle named executive officers to severance, perquisites, or other enhanced benefits upon termination of their employment. Any agreement to provide such payments or benefits to a terminating executive officer (other than following a change in control) would be at the discretion of the Compensation Committee.
| | | | | | | | | | | | | | | | | | | | |
| Name | Cash Severance Payment 1 | Continuation of Medical / Welfare Benefits (present value) 2 | Acceleration and Continuation of Equity Awards as of 12/31/2021 | | Total Termination Benefits |
| Mr. Ricks | | | | | |
| • | Involuntary retirement or termination | $0 | $0 | $28,724,256 | | $28,724,256 |
| • | Involuntary or good-reason termination after change in control | $7,500,000 | $44,054 | $83,838,115 | | $91,382,169 |
| Ms. Ashkenazi | | | | | |
| • | Involuntary retirement or termination | $0 | $0 | $4,496,378 | | $4,496,378 |
| • | Involuntary or good-reason termination after change in control | $3,600,000 | $52,130 | $9,766,103 | | $13,418,233 |
| Dr. Skovronsky | | | | | |
| • | Involuntary retirement or termination | $0 | $0 | $9,504,661 | | $9,504,661 |
| • | Involuntary or good-reason termination after change in control | $4,000,000 | $44,242 | $27,791,074 | | $31,835,316 |
| Mr. Van Naarden | | | | | |
| • | Involuntary retirement or termination | $0 | $0 | $3,277,074 | | $3,277,074 |
| • | Involuntary or good-reason termination after change in control | $2,430,000 | $51,818 | $5,717,146 | | $8,198,964 |
| Mr. Rau | | | | | |
| • | Involuntary retirement or termination | $0 | $0 | $5,568,595 | | $5,568,595 |
| • | Involuntary or good-reason termination after change in control | $2,412,000 | $39,340 | $5,568,595 | | $8,019,935 |
1 See "Change-in-Control Severance Pay Plan—Cash Severance Payment" below.
2 See "Accrued Pay and Regular Retirement Benefits" and "Change-in-Control Severance Pay Plan—Continuation of medical and welfare benefits" below.
Accrued Pay and Regular Retirement Benefits:The amounts shown in the table above do not include certain payments and benefits to the extent they are provided on a non-discriminatory basis to salaried employees generally upon termination of employment. These include:
•accrued salary, vacation pay, and, if applicable, equity payouts prorated for time worked in the performance period and adjusted for company performance
•regular pension benefits under the Retirement Plan and the nonqualified pension plan. See "Retirement Benefits" above
•welfare benefits provided to eligible U.S. retirees, including retiree medical and dental insurance. The amounts shown in the table above as "Continuation of Medical/Welfare Benefits" are explained below; and
•distributions of plan balances under the 401(k) Plan, the nonqualified savings plan, and the Deferred Compensation Plan. See the narrative following the Nonqualified Deferred Compensation in 2020 table for information about these plans.
Death and Disability:A termination of employment due to death or disability does not entitle named executive officers to any payments or benefits that are not available to U.S. employees generally.
Termination for Cause:Executives terminated for cause receive no severance or enhanced benefits and forfeit any unvested equity grants.
Change-in-Control Severance Pay Plan: As described in the CD&A under "Other Compensation Practices and Information— Severance Benefits," the company maintains a change-in-control severance pay plan for nearly all employees, including the named executive officers. The change-in-control plan for executive officers defines a change in control specifically, but generally the terms include the occurrence of one of the following: (i) acquisition by any person of 20 percent or more of the company’s stock; (ii) replacement by the shareholders of one-half or more of the board of directors; (iii) consummation of a merger, share exchange, or consolidation of the company (other than a transaction that results in the Lilly shareholders prior to the transaction continuing to hold more than 60 percent of the voting stock of the combined entity); or (iv) complete
liquidation of the company or sale or disposition of all or substantially all of its assets. The amounts shown in the table for "involuntary or good-reason termination after change in control" are based on the following assumptions and plan provisions:
•Covered terminations. The table assumes a termination of employment that is eligible for severance under the terms of the plan, based on the named executive officer’s compensation, benefits, age, and service credit at December 31, 2021. Eligible terminations include an involuntary termination for reasons other than "cause" or a voluntary termination by the executive for "good reason," each as defined in the plan, within two years following the change in control.
•A termination of an executive officer by the company is for "cause," in general, if it is for any of the following reasons: (i) the employee’s willful refusal to perform, without legal cause, his or her material duties, resulting in demonstrable economic harm to the company or participating employers; (ii) any act of fraud, dishonesty, or gross misconduct resulting in significant economic harm or other significant harm to the company or participating employers or to the business reputation of the company or participating employers; or (iii) conviction by a court of competent jurisdiction of any crime (or the entering of a plea of guilty or nolo contendere to a charge of any crime) constituting a felony.
•A termination by the executive officer is for "good reason," in general, if it results from: (i) a material diminution in the nature or status of the executive’s position, title, reporting relationship, duties, responsibilities, or authority, or the assignment to him or her of additional responsibilities that materially increase his or her workload; (ii) any reduction in the executive’s then-current base salary; (iii) a material reduction in the executive’s opportunities to earn incentive bonuses below those in effect for the year prior to the change in control; (iv) a material reduction in the executive’s employee benefits from the benefit levels in effect immediately prior to the change in control; (v) the failure to grant to the executive stock options, stock units, performance shares, or similar incentive rights during each 12-month period following the change in control on the basis of a number of shares or units and all other material terms at least as favorable to the executive as those rights granted to him or her on an annualized average basis for the three-year period immediately prior to the change in control; (vi) relocation of the executive by more than 50 miles from his or her regularly assigned workplace in effect immediately prior to the change in control; or (vii) any failure by a successor entity to the company in connection with a change in control to assume the obligations of the company under the plan, or any attempted amendment, termination or repudiation of the plan by such successor entity.
•Cash severance payment. The cash severance payment amounts, in general, to two times the executive officer's annual base salary plus two times the executive officer’s bonus target for that year under the Bonus Plan.
•Continuation of medical and welfare benefits. This amount represents the present value of the change-in-control plan’s provision, following a covered termination, of 18 months of continued coverage equivalent to the company’s current active employee medical, dental, group life, company-provided death benefit, and long-term disability insurance. Similar actuarial assumptions to those used to calculate incremental pension benefits apply to the calculation for continuation of medical and welfare benefits, with the addition of actual COBRA rates based on current benefit elections.
•Acceleration of equity awards. Upon a covered termination, any unvested equity awards would convert into restricted stock units of the new company, with the number of shares earned under the awards based on accrued performance at the time of the transaction. The restricted stock units will continue to vest and pay out upon the earlier of the completion of the original award period; upon a covered termination; or if the successor entity does not assume, substitute, or otherwise replace the award. The amount in this column represents the value of the acceleration of unvested equity grants had a qualifying termination occurred on December 31, 2021.
•Excise taxes. Upon a change in control, employees may be subject to certain excise taxes under Section 280G of the Internal Revenue Code. The company does not reimburse the affected employees for those excise taxes or any income taxes payable by the employee. To reduce the employee's exposure to potential excise taxes, the employee’s change-in-control benefit will be decreased to maximize the after-tax benefit to the individual.
Payments upon Change in Control Alone:The change-in-control plan is a "double trigger" plan, meaning payments are made only if the employee suffers a covered termination of employment within two years following the change in control, or in the case of equity awards, if the successor entity does not assume, substitute, or otherwise replace the awards.
Agreement with Former Chief Financial Officer
In connection with Mr. Smiley’s resignation, he and the Company entered into a Separation Agreement (the Separation Agreement), which provided that Mr. Smiley immediately resign from his position as senior vice president and chief financial officer of the Company, as well as forfeit or forego compensatory arrangements totaling $20,133,375 (using Lilly's closing share price at the time of such forfeiture or the intended grant date value for such awards that had been previously approved but not yet granted prior to Mr. Smiley's resignation, as applicable), including all of his earned 2020 cash bonus (which would have otherwise been $1,102,317 based on company performance), his target 2021 bonus of $1,000,000, 25 percent of his earned 2018-2020 shareholder value award (representing a forfeiture of $3,100,954 in value based on Lilly's closing share price at the time of such forfeiture), and all other outstanding and previously approved but not yet granted equity incentive awards totaling $14,930.104. Mr. Smiley also agreed to remain available to the company’s chief executive officer and his successor as chief financial officer through July 2021 to facilitate the transition of his responsibilities, at reduced cash compensation of $9,000 every two weeks. The Separation Agreement includes customary provisions regarding confidentiality
and a release of claims against the company, as well as a 24-month non-solicitation agreement and an 18-month non-competition agreement.
CEO Pay Ratio
Lilly’s compensation and benefits philosophy across the organization is to encourage and reward all employees who contribute to our success. We strive to ensure that the pay of every Lilly employee reflects the level of their job impact and responsibilities and is competitive within our peer group. Compensation rates are benchmarked and set to be market-competitive in the country in which the jobs are performed. Lilly’s ongoing commitment to pay equity is critical to our success in supporting a diverse workforce with opportunities for all employees to grow, develop, and contribute. Lilly
Below is a global company that employs over 40,000 people with more than halfthe 2021 annual total compensation of our workforce located outside of the U.S.
Under rules adopted pursuant to the Dodd-Frank Act of 2010, Lilly is required to calculateCEO and disclose the total compensation paid to itsour median paid employee as well asand the ratio of the annual total compensation paidof our CEO to thethat of our median employee as compared to the total compensation paid to Lilly’s CEO. The paragraphs that follow describe our methodology and the resulting CEO Pay ratio.employee.
| | | | | |
| CEO Pay Ratio: |
| CEO Annual Total Compensation* | $21,509,985 |
| Median Employee Annual Total Compensation | $97,130 |
| CEO to Median Employee Pay Ratio | 221:1 |
| *This annual total compensation is the "Summary Compensation Table" amount. |
Methodology:
•Measurement Date
Date: We identified the median employee using our employee population on November 1, 2017.October 31, 2021. On this date, Lilly employed approximately 37,100 people, with approximately 16,100 members of our workforce located in the U.S. and approximately 21,000 members of our workforce located outside of the U.S.
Consistently Applied Compensation Measure (CACM)
Under the relevant•Identification of Median Employee: In a manner consistent with SEC rules, we were required to identifyidentified the median employee by use of a “consistently"consistently applied compensation measure,”" or CACM. We chose a CACM that closely approximates the annual total direct compensation of our employees. Specifically, we identified the median employee by looking at annual base pay, bonus opportunity at target, and the grant date fair value for standard equity awards. We did not perform adjustments toadjust the compensation paid to part-time employees to calculate what they would have been paid on a full-time basis.
•De Minimis Exception
Exception: Lilly has employees in 8670 countries. In identifying the median employee, we excluded 386 workers in the following 10 countries, totaling 639 workers (approximately 1.5which represent approximately one percent of our workforce).workforce: Bahrain, Ecuador, Egypt, Greece, Indonesia, Kuwait, Pakistan, Panama, Qatar, and United Arab Emirates. We excluded these employees because they are affiliated with joint ventures or third-party distributors, and Lilly does not set their compensation philosophy.
We excluded the following number of workers from the following countries in the identification of the median employee:
|
| |
Countries Excluded | Workers Excluded |
Bahrain | 2 |
Greece | 270 |
Indonesia | 30 |
Kuwait | 17 |
Oman | 2 |
Pakistan | 33 |
Qatar | 7 |
Saudi Arabia | 145 |
United Arab Emirates | 100 |
Vietnam | 33 |
Total | 639 |
Methodology and•Calculated CEO Pay Ratio
Ratio: After applying our CACM methodology and excluding the employees listed above, we identified the median employee. Once the median employee was identified, we calculated the median employee’s total annual compensation in accordance with the requirements of the Summary Compensation Table.
Our median employee compensation as calculated using Summary Compensation Table requirements was $134,003. Our CEO’s compensation as reported in the Summary Compensation Table was $15,845,991. Therefore, our CEO to median employee pay ratio is 118:1. Our median employee’s total compensation included the amount of a pension enhancement offered under our 2017 voluntary early retirement program. If we eliminated the change in pension value from our median employee and CEO’s total compensation, our CEO to median employee pay ratio would have been 171:1.
This information is being provided for compliance purposes. Neither the Compensation Committee nor management of the company used the pay ratio measure in making compensation decisions.
Audit Matters
Item 3. Ratification of the Appointment of Principalthe Independent Auditor
Audit Committee Oversight of the Independent Auditor
The Audit Committee is responsible for the appointment, compensation, retention, and oversight of the independent auditor and oversees the process for selecting, reviewing and evaluating the lead audit partner. Further information regarding the committee's oversight of the independent auditor can be found in the Audit Committee charter, available online at https://www.lilly.com/who-we-are/lilly.com/leadership/governance or upon request to the company's corporate secretary.
.
In connection with the decision regarding whether to reappoint the independent auditor each year (subject to shareholder ratification), the committee assesses the independent auditor's performance. This assessment examines three primary criteria: (1) the independent auditor's qualifications and experience; (2) the communication and interactions with the auditor over the course of the year; and (3) the auditor's independence, objectivity, and professional skepticism. These criteria are assessed against an internal and an external scorecard and are discussed with management during a private session as well as in executive session. The committee also periodically considers whether a rotation of the company's independent auditor is advisable.
Ernst & Young LLP (EY) has served as the principal independent auditor for the company since 1940. Based on this year'sthe Audit Committee’s assessment of EY's performance during 2021, the Audit Committee believes that the continued retention of EY to serve as the company's principal independent auditor is in the best interests of the company and its shareholders and has therefore reappointed the firm of EY as principalthe company’s independent auditor for the company for 2018.2022. In addition to
this year's favorable assessment of EY's performance, we recognize that there are several benefits of retaining a longer-tenured independent auditor. EY has gained institutional knowledge and expertise regarding the company's global operations, accounting policies and practices, and internal controls over financial reporting. Audit and other fees are also competitive with peer companies because of EY's
familiarity with the company and its operations. In accordance with the bylaws, this appointment is being submitted to the shareholders for ratification.
Representatives of EY are expected to be present atparticipate in the annual meetingAnnual Meeting and will be available to respond to questions. Those representatives will have the opportunity to make a statement if they wish to do so.
Board Recommendation on Item 3
The Board of Directorsboard recommends that you vote FOR ratifying the appointment of Ernst & Young LLPEY as principalthe independent auditor for 2018.2022.
Audit Committee Report
The Audit Committee reviews the company’s financial reporting process on behalf of the Board.board. Management has the primary responsibility for the financial statements and the reporting process, including the systems of internal controls and disclosure controls. In this context, the committeeAudit Committee has met and held discussions with management and the independent auditor. Management represented to the committeeAudit Committee that the company’s consolidated financial statements for the year ended December 31, 2021 were prepared in accordance with generally accepted accounting principles (GAAP),GAAP, and the committeeAudit Committee has reviewed and discussed the audited financial statements and related disclosures with management and the independent auditor, including a review of the significant management judgments underlying the financial statements and disclosures.
The independent auditor reports directly to the Audit Committee, which has sole authority to appoint and to replace the independent auditor (subject to shareholder ratification).
The committeeAudit Committee has discussed with the independent auditor the matters required to be discussed with the Audit Committee by the standards of the Public Company Accounting Oversight Board (PCAOB), the SEC, and the NYSE, including the quality, not just the acceptability, of the accounting principles, the reasonableness of significant judgments, and the clarity of the disclosures in the financial statements. In addition, the committeeAudit Committee has received the written disclosures and the letter from the independent auditor required by applicable requirements of the PCAOB rules regarding communications with the Audit Committee concerning independence and has discussed with the independent auditor the auditor’s independence from the company and its management. In concluding that the auditor is independent, the committeeAudit Committee determined, among other things, that the nonauditnon-audit services provided by EY (as described below) were compatible with its independence. Consistent with the requirements of the Sarbanes-Oxley Act of 2002 (the Sarbanes-Oxley Act), the committeeAudit Committee has adopted policies to ensure the independence of the independent auditor, such as prior committee approval of nonauditnon-audit services and required audit partner rotation.
The committeeAudit Committee discussed with the company’s internal and independent auditors the overall scope and plans for their respective audits, including internal control testing under Section 404 of the Sarbanes-Oxley Act. The committeeAudit Committee periodically meets with the internal and independent auditors, with and without management present, and in private sessions with members of senior management (such as the chief financial officer and the chief accounting officer) to discuss the results of their examinations, their evaluations of the company’s internal controls, and the overall quality of the company’s financial reporting. The committeeAudit Committee also periodically meets in executive session.
In reliance on the reviews and discussions referred to above, the committeeAudit Committee recommended to the Boardboard (and the Boardboard subsequently approved the recommendation) that the audited consolidated financial statements be included in the company’s annual reportAnnual Report on Form 10-K for the year ended December 31, 2017,2021, for filing with the SEC. The committeeAudit Committee has also appointed EY as the company’s independent auditor, subject to shareholder ratification, for 2018.
2022.
Audit Committee
Jamere Jackson, Chair
Ralph Alvarez
Michael L. Eskew Chair
Katherine Baicker, Ph.D.
Jamere Jackson
Kathi P. SeifertGabrielle Sulzberger
Jackson P. Tai
Karen Walker
Services Performed by the Independent Auditor
The Audit Committee pre-approves all services performed by the independent auditor, in part to assess whether the provision of such services might impair the auditor’s independence. The committee’sAudit Committee’s policy and procedures are as follows:
•Audit services:The committeeAudit Committee approves the annual audit services engagement and, if necessary, any changes in terms, conditions, and fees resulting from changes in audit scope, company structure, or other matters. Audit services include internal controls attestation work under Section 404 of the Sarbanes-Oxley Act. The committeeAudit Committee may also preapprovepre-approve other audit services, which are those services that only the independent auditor reasonably can provide.
•Audit-related services: Audit-related services are assurance and related services that are reasonably related to the performance of the audit or reviews of the financial statements, and that are traditionally performed by the independent
auditor. The committeeAudit Committee believes that the provision of these services does not impair the independence of the auditor.
•Tax services: The committeeAudit Committee believes that, in appropriate cases, the independent auditor can provide tax compliance services, tax planning, and tax advice without impairing the auditor’s independence.
•Other services: The committeeAudit Committee may approve other services to be provided by the independent auditor if (i) the services are permissible under SEC and PCAOB rules, (ii) the committeeAudit Committee believes the provision of the services would not impair the independence of the auditor, and (iii) management believes that the auditor is the best choice to provide the services.
•Approval process: At the beginning of each audit year, management requests prior committee approvalpre-approval from the Audit Committee of the annual audit, statutory audits, and quarterly reviews for the upcoming audit year as well as any other services known at that time. Management will also present at that time an estimate of all fees for the upcoming audit year.year and known services. As specific engagements are identified thereafter that were not initially approved, they are brought forward to the committeeAudit Committee for approval. To the extent approvals are required between regularly scheduled committeeAudit Committee meetings, preapprovalpre-approval authority is delegated to the committee chair.
For each engagement, management provides the committeeAudit Committee with information about the services and fees, sufficiently detailed to allow the committee to make an informed judgment about the nature and scope of the services and the potential for the services to impair the independence of the auditor.
After the end of the audit year, management provides the committee with a summary of the actual fees incurred for the completed audit year.
Independent Auditor Fees
The following table shows the fees incurred for services rendered on a worldwide basis by Ernst & YoungEY in 20172021 and 2016.2020. All such services were pre-approved by the committeeAudit Committee in accordance with the pre-approval policy.
| | | | | | | | | | | | | | |
| 2021 ($ millions)* | 2020 ($ millions)* |
| Audit Fees | Annual audit of consolidated and subsidiary financial statements, including Sarbanes-Oxley 404 attestation | $13.9 | $13.7 |
| | |
| Reviews of quarterly financial statements | | |
| Audit-Related Fees | | $0.7 | $0.9 |
| Primarily related to assurance and related services reasonably related to the performance of the audit or reviews of the financial statements primarily related to employee benefit plan and other ancillary audits, and due diligence services on potential acquisitions | | |
| | |
| Tax Fees | | | $2.1 | $2.7 |
| Tax compliance services, tax planning, tax advice
Primarily related to consulting and compliance services | | |
| Total | | | $16.7 | $17.3 |
| *Numbers may not add due to rounding |
|
| | | | |
| | 2017 ($ millions) | 2016 ($ millions) |
| Audit Fees | | | $14.8 | $12.8 |
| | Annual audit of consolidated and subsidiary financial statements, including Sarbanes-Oxley 404 attestation | | |
| | Reviews of quarterly financial statements | | |
| | Other services normally provided by the auditor in connection with statutory and regulatory filings | | |
| Audit-Related Fees | | $0.5 | $0.6 |
| | Primarily related to assurance and related services reasonably related to the performance of the audit or reviews of the financial statements primarily related to employee benefit plan and other ancillary audits, and due diligence services on potential acquisitions | | |
| | | |
| Tax Fees | | | $4.8 | $6.7 |
| |
Tax compliance services, tax planning, tax advice Primarily related to consulting and compliance services | | |
| Total | | | $20.1 | $20.2 |
*Numbers may not add due to rounding
| | |
Management Proposals
Item 4. Proposal to Amend the Company'sCompany’s Articles of Incorporation to Eliminate the Classified Board Structure
The company’s articles of incorporation provide that the board of directors is divided into three classes, with each class elected every three years. On the recommendation of theThe board, after review by its Directors and Corporate Governance Committee, the board has approved, and recommends that the shareholders approve, amendments to eliminate the classified board structure in order to provide for the declassificationannual election of all directors (the Declassification Amendments). From 2010 through 2012 and again from 2018 through 2021, the company's board. Thisboard submitted this management proposal was brought beforeto shareholders each year between 2007 and 2012, receivingseeking approval to eliminate the votecompany’s classified board structure; however, under the company’s articles of a strong majority of the outstanding shares at each meeting; however,incorporation the proposal requires the vote of 80 percent of the outstanding shares to pass.
be approved and on each prior occasion failed to receive the required vote.
If approved, this proposalthe Declassification Amendments would become effective following the Annual Meeting upon the filingcompany making the required filings of amended and restated articles of incorporationthe Declassification Amendments with the Secretary of State of Indiana, which the company would do promptly after shareholder approval is obtained.Indiana. Directors elected prior to the effectiveness of the amendmentsDeclassification Amendments would stand for election forserve the remainder of their respective three-year terms and each director elected after the Annual Meeting would serve a one-year terms once their then-current terms expire. This means that directors whose terms expireterm, ending at the 2019 and 2020next annual meetingsmeeting of shareholdersshareholders. Thereafter, the company’s classified board structure would be elected for one-year terms, and beginningfully eliminated starting with the 20212025 annual meeting all directors would be elected for one-year terms at each annual meeting.of shareholders. In the case of any vacancy on the board occurring after the 2018 annual meetingAnnual Meeting created by an increase
in the number of directors, the vacancy would be filled through an interim electionappointment by the board, with the new director to serve a term ending at the next annual meeting.meeting of shareholders. Vacancies created by resignation, removal or death would be filled by interim election ofappointment by the board forof a termnew director to serve until the end of the term of the director being replaced. This proposal would not change the present number of directors or the board’s authority to change that number and to fill any vacancies or newly-creatednewly created directorships.
Background of Proposal
As part of its ongoing review of corporate governance matters, the board, assisted bytaking into account the input of the Directors and Corporate Governance Committee, considered the advantages and disadvantages of maintaining the classified board structure and eliminating supermajority voting provisions of the articles of incorporation (see Item 5 below).structure. The board considered the view of somecertain shareholders who believe that classified boards have the effect of reducing the accountability of directors to shareholders because shareholders are unable to evaluate and electconsider all directors for election on an annual basis. The board gave considerable weight to the approval at the 2006 annual meeting of a shareholder proposal requesting that the board take all necessary steps to elect the directors annually, and to the favorable votes of a strong majority of the outstanding shares for management’s proposals in each of the following sixprior years.
The board also considered the benefits of retaining the classified board structure, which has a long history in corporate law.structure. A classified structure may providepromote shareholder value by providing continuity and stability in the management of the business and affairs of the company, becauseas a majority of directorsthe board always has prior experience as directors of the company. In someaddition, under certain circumstances, classified boards may enhanceprotect shareholder value by forcing an entity seeking control of the company to initiate discussions at arm’s lengtharm’s-length with the board of the company, because the entity cannot replace the entiremajority of the board in a single election. The board also considered that even without a classified board (and without the supermajority voting requirements, which the board also recommends eliminating), the company has defenses that work togetherwould have appropriate safeguards to protect the interests of all shareholders and discourage a would-be acquirer from proceeding with a proposal that undervalues the company and to assist the board in responding to such proposals.or is opportunistic. These defenses include other provisions of the company’s articles of incorporation and bylaws, as well as certain provisions of Indiana corporation law.
The board believes it is important to maintain appropriate defenses to inadequate takeover bids, but also important to retain shareholder confidence by demonstrating that the board is accountable and responsive to shareholders. After balancing these interests, the board has decided to resubmit this proposal to eliminate the classified board structure.
Text of the Amendments
Article 9(b) of the company’s articles of incorporation contains the provisions that will be affected if this proposal is adopted. This article, set forth in Appendix B to this proxy statement, shows the proposed changes, with deletions indicated by strikeouts and additions indicated by underlining. The board has also adopted conforming amendments to the company’s bylaws, to be effective immediately upon, and subject to, the effectiveness of the amendments to the articles of incorporation.
Vote Required
The affirmative vote of at least 80 percent of the outstanding shares of common stock is needed to approve this proposal. Unless such vote is received, the present classification of the board will continue.
Board Recommendation on Item 4
The board recommends that you vote FOR amending the company’s articles of incorporation to eliminate the classified board structure.
Item 5. Proposal to Amend the Company’s Articles of Incorporation to Eliminate Supermajority Voting Provisions
The company’s articles of incorporation provide that nearly all matters submitted to a vote of shareholders can be adopted by a majority of the votes cast. However, the company’s articles of incorporation require certain fundamental corporate actions to be approved by the holders of 80 percent of the outstanding shares of common stock. Those actions are:
•amending certain provisions of the articles of incorporation that relate to the number and terms of office of directors:
◦the company’s classified board structure (as described under Item 4)
◦a provision that the number of directors shall be specified solely by resolution of the board
•removing directors prior to the end of their elected term
•entering into mergers, consolidations, recapitalizations, or certain other business combinations with a “related person”—a party who has acquired at least five percent of the company’s stock (other than the Endowment or a company benefit plan) — without the prior approval of such action or transaction by the directors not affiliated with such shareholder
•modifying or eliminating any of the above supermajority voting requirements.
The board, after review by the Directors and Corporate Governance Committee, has approved, and recommends that the shareholders approve, amendments to eliminate the supermajority voting requirements (the Supermajority Voting Amendments). From 2010 through 2012 and again from 2018 through 2021, the board submitted this management proposal to shareholders seeking approval to eliminate these supermajority voting requirements; however, under the company’s articles of incorporation the proposal requires the vote of 80 percent of the outstanding shares to be approved and on each prior occasion failed to receive the required vote.
Background of Proposal
As part of its ongoing review of corporate governance matters, the board, taking into account the input of the Directors and Corporate Governance Committee, considered the advantages and disadvantages of maintaining the supermajority voting requirements. The board considered the potential adverse consequences of maintaining the supermajority voting requirements. The board believes it is important to maintain shareholder confidence by demonstrating that the board is responsive and accountable to shareholders and committed to strong corporate governance. This requires the board to carefully balance sometimes competing interests. In this regard, the board gave considerable weight to the favorable votes of a strong majority of the outstanding shares for management’s proposal in the previous four years. Many shareholders believe that supermajority voting requirements impede accountability to shareholders and contribute to board and management entrenchment. The board also considered that, even without the supermajority vote (and without the classified board, which the board also recommends eliminating), the company has appropriate safeguards to protect the interests of all shareholders and to discourage a would-be acquirer from proceeding with a proposal that undervalues the company or is opportunistic and to assist the board in responding to such proposals. These include other provisions of the company’s articles of incorporation and bylaws as well as certain provisions of Indiana corporation law.
The board also considered that under certain circumstances, supermajority voting requirements can provide benefits to the company and all its shareholders by making it more difficult for one or a few large shareholders to facilitate a takeover of the company or implement certain significant changes to the company without more widespread shareholder support.
After balancing these interests, the board has decided to resubmit this proposal to eliminate the supermajority voting requirements.
Text of Amendments
Article 9(b)Articles 9(c), 9(d), and 13 of the company’s articles of incorporation contain the provisions that will be affected if this proposal is adopted. These articles, set forth in Appendix B to this proxy statement, show the proposed changes with deletions indicated by strikeouts and additions indicated by underlining. The board has also adopted conforming amendments to the company’s bylaws, to be effective immediately upon, and subject to, the effectiveness of the amendments to the articles of incorporation.
Vote Required
The affirmative vote of at least 80 percent of the outstanding shares of common stock is needed to approve this proposal. Unless such vote is received, the supermajority voting requirements will continue to be in effect.
Board Recommendation on Item 5
The board recommends that you vote FOR amending the company's articles of incorporation to eliminate supermajority voting requirements.
Item 6. Proposal to Amend the Company's Articles of Incorporation to Give Shareholders the Ability to Amend the Company’s Bylaws
The company’s articles of incorporation do not allow shareholders to amend the company’s bylaws. This position reflects the default under Indiana law, which reserves the exclusive authority to amend bylaws to the board. The board, after review by the Directors and Corporate Governance Committee, has approved, and recommends that the shareholders approve, amendments to the articles of incorporation to allow shareholders to amend the company’s bylaws. Pursuant to Indiana law, approval of the proposal requires that the votes cast in favor of the proposal exceed the votes cast against the proposal.
Background of Proposal
As part of its ongoing review of corporate governance matters, the board, taking into account the input of the Directors and Corporate Governance Committee, considered the advantages and disadvantages of maintaining the Indiana default position that reserves the exclusive right to amend the bylaws to the board. The board considered the potential adverse consequences of maintaining the Indiana default position that does not allow shareholders to amend the bylaws. The board believes it is important to maintain shareholder confidence by demonstrating that the board is responsive and accountable to shareholders and committed to strong corporate governance. This requires the board to carefully balance sometimes competing interests. In this regard, the board gave considerable weight to shareholder feedback supporting the ability of
shareholders to amend the bylaws. The board also considered that even if shareholders have the authority to amend the bylaws, the company has appropriate safeguards to protect the interests of all shareholders and discourage a would-be acquirer from proceeding with a proposal that undervalues the company or is opportunistic. These include other provisions of the company’s articles of incorporation and bylaws, as well as certain provisions of Indiana corporation law.
The board also considered that under certain circumstances, allowing the bylaws to be amended by shareholders would not be in the best long-term interests of the company because of the potential risk of abuse by a single shareholder or special interest group of shareholders with no fiduciary duties to other shareholders, and accordingly, the board, which is accountable to all shareholders, may be better positioned to ensure that bylaw amendments are appropriate.
After balancing these interests, the board has decided to submit this proposal to allow shareholders holding a majority of the company’s issued and outstanding common stock to amend the company’s bylaws.
Proposed amendments to the bylaws:
Article 10 of the company’s articles of incorporation contains the provisions that will be affected if this proposal is adopted. The amendments to the company's amended articles of incorporation,This article, set forth in Appendix BC to this proxy statement, shows the proposed changes, with deletions indicated by strike-outs and additions indicated by underlining. The board has also adopted conforming amendments to the company’s bylaws, to be effective immediately upon, and subject to, the effectiveness of the amendments to the amended articles of incorporation.
Vote Required
The affirmativeTo approve this proposal, the votes cast in favor of this proposal must exceed the votes cast against this proposal. Unless such vote of at least 80 percent ofis received, the outstanding common shares is needed to pass this proposal.
present prohibitions against shareholders amending the company’s bylaws will continue.
Board Recommendation on Item 4
6The Board of Directorsboard recommends that you vote FOR amending the company'scompany’s articles of incorporation to eliminateallow shareholders to amend the classified board structure.company’s bylaws.
Item 5.7. Proposal to Amend the Company's ArticlesBylaws to Require an Independent Board Chair
Legal & General Investment Management America, Inc. ("LGIM America"), 71 South Wacker Drive, Suite 800, Chicago, Illinois 60606, beneficial owner of Incorporation to Eliminate Supermajority Voting Provisions
Under the company’s articles of incorporation, nearly all matters submitted to a vote of shareholders can be adopted by a majority of the votes cast. However, our articles require a few fundamental corporate actions to be approved by the holders of 80 percent of the outstanding shares of common stock (a “supermajority vote”). Those actions are:
|
| | | | |
• | | amending certain provisions of the articles of incorporation that relate to the number and terms of office of directors: |
| | —the company's classified board structure (as described under Item 4, above)
|
| | —a provision that the number of directors shall be specified solely by resolution of the board of directors |
• | | removing directors prior to the end of their elected term |
• | | entering into mergers, consolidations, recapitalizations, or certain other business combinations with a "related person"—a party who has acquired at least five percent of the company's stock (other than the Lilly Endowment or a company benefit plan) without the prior approval of the board of directors |
• | | modifying or eliminating any of the above supermajority voting requirements. |
Background of Proposal
This proposal is the result of the board’s ongoing review of corporate governance matters. In 2007 through 2009, shareholder proposals requesting that the board take action to eliminate all supermajority voting provisions were supported by a majority of votes cast. In 2010 through 2012, the board responded by submitting proposals seeking shareholder approval to eliminate the provisions. In all three years, the proposal received the votes of a strong majority of the outstanding shares, but fell short of the required 80 percent.
Assisted by the Directors and Corporate Governance Committee, the board considered the advantages and disadvantages of maintaining the supermajority voting requirements. The board considered that under certain circumstances, supermajority voting provisions can provide benefits to the company. The provisions can make it more difficult for one or a few large shareholders to take over or restructure the company without negotiating with the board. In the event of an unsolicited bid to take over or restructure the company, supermajority voting provisions may encourage bidders to negotiate with the board and increase the board’s negotiating leverage on behalf of the shareholders. They can also give the board time to consider alternatives that might provide greater value for all shareholders.
The board also considered the potential benefits of eliminating the supermajority voting provisions. While it is important to the company’s long-term success for the board to maintain appropriate defenses against inadequate takeover bids, it is also important for the board to maintain shareholder confidence by demonstrating that it is responsive and accountable to shareholders and committed to strong corporate governance. This requires the board to carefully balance sometimes competing interests. In this regard, the board gave considerable weight to the fact that a substantial majority of shares voted have supported eliminating the supermajority voting provisions. Many shareholders believe that supermajority voting provisions impede accountability to shareholders and contribute to board and management entrenchment.
The board also considered that even without the supermajority vote (and without the classified board, which the board also recommends eliminating), the company has defenses that work together to discourage a would-be acquirer from proceeding with a proposal that undervalues the company and to assist the board in responding to such proposals. These defenses include other provisions of the company’s articles of incorporation and bylaws as well as certain provisions of Indiana corporation law.
Therefore, the board believes the balance of interests is best served by recommending to shareholders that the articles of incorporation be amended to eliminate the supermajority voting provisions. By recommending these amendments, the board is demonstrating its accountability and willingness to take steps that address shareholder-expressed concerns.
Text of Amendments
Articles 9(c), 9(d), and 13 of the company’s amended articles of incorporation contain the provisions that will be affected if this proposal is adopted. The amendments to the company’s amended articles of incorporation set forth in Appendix B to this proxy statement show the proposed changes with deletions indicated by strike-outs and additions indicated by underlining.
Vote Required
The affirmative vote of at least 80 percent of the outstanding common shares is needed to pass this proposal.
Board Recommendation on Item 5
The Board of Directors recommends that you vote FOR amending the company's articles of incorporation to eliminate supermajority voting provisions.
Item 6. Proposal to Approve the Amended and Restated 2002 Lilly Stock Plan
Background of Proposal
As the 2002 Lilly Stock Plan (the “2002 Plan”) nears its April 20, 2020 expiration date, we are asking our shareholders to approve an amendment and restatement of the 2002 Plan (the “Amended 2002 Plan”). The Amended 2002 Plan provides for a decrease – not an increase – in the number of shares of common stock available for issuance. Further, the Amended 2002 Plan eliminates certain provisions related to Section 162(m) of the Internal Revenue Code that are no longer applicable in light of tax legislation that was recently enacted while retaining a framework to continue to grant performance-based awards.
As of February 16, 2018, there were no options outstanding under the 2002 Plan, and 11,753,977full value awards that were unvested and outstanding.
Material Changes to the 2002 Plan
The following summary highlights the proposed material changes to the 2002 Plan.
53,000,000 shares of common stock would be available for issuance pursuant to future awards granted on or following the effective date of the Amended 2002 Plan. This represents a decrease in the number of shares reserved for issuance under the plan:
|
| | |
| | Immediately Prior to Shareholder Approval | After Shareholder Approval |
| Authorized Shares | 119,000,000* | 75,657,296* |
| Shares Available to Grant | 96,342,704
| 53,000,000 |
*plus shares available for issuance from prior plans, as approved by shareholders at the inception of the 2002 Plan
Amendments have been made to improve our corporate governance and to comply with some of the policies recommended by shareholder advisors, including:
| |
o | Provisions to preclude the payment of dividends or dividend equivalents on unvested restricted stock, restricted stock units or other share-based awards that are full-value awards; and |
| |
o | Imposition of a minimum one-year vesting period for all awards other than a carve-out for up to 5% of the shares that are available for issuance as of the effective date of the Amended 2002 Plan. |
In light of the tax legislation that was recently enacted by Congress eliminating the performance-based exception from the deductibility limitations under Section 162(m) of the Internal Revenue Code, the requirements applicable to equity awards that were intended to constitute “qualified performance-based compensation” under Section 162(m) of the Internal Revenue Code have been eliminated. However, the Amended 2002 Plan retains a framework to grant performance-based awards that provides for requirements similar to those previously imposed on awards intended to constitute qualified performance-based compensation under Section 162(m).
An annual limit has been imposed on the size of equity awards that may be granted to any non-employee director during a calendar year. The accounting value of equity awards, when aggregated with cash compensation, granted to a non-employee director in any calendar year may not exceed $800,000.
A “clawback” provision has been added, permitting us to recover awards or payments from participants, including as may be required under the Dodd-Frank Act.
To achieve consistency with our other change in control arrangements, the “Change in Control” definition has been revised to reflect a definition that is more consistent with the definition in the Eli Lilly and Company Change in Control Severance Pay Plan.
The single-trigger change in control vesting acceleration provision applicable to time-based awards has been eliminated. The Amended 2002 Plan now provides that time-based awards will not vest in connection with a Change in Control unless they are not assumed, substituted or otherwise replaced.
The change in control treatment applicable to performance-based awards has been revised to provide that the vesting of performance-based awards upon a Change in Control will not occur at a rate that is greater than the actual level of attainment and/or provide for pro-rated vesting of the award based on any reduction to the performance period.
An amendment to add the authority to grant other share-based awards, which would include other potential types of awards denominated or based on the stock of the Company that may not fall into the category of awards that currently may be granted.
An amendment to eliminate the automatic expiration date. Instead, the Amended 2002 Plan provides that it will continue in effect until it is terminated by our Board of Directors.
Key Terms of the Amended 2002 Plan at a Glance
The following is a summary of the key provisions of the Amended 2002 Plan, as set forth and stated herein.
|
| |
Plan Term: | The Amended 2002 Plan was adopted by the Board of Directors on February 20, 2018, subject to obtaining shareholder approval and will continue in effect until terminated by the Board of Directors. Shares available under the Amended 2002 Plan are expected to last at least five years.
|
Eligible Participants: | Employees and directors of the Company and its affiliates generally are eligible to receive non-qualified stock options, restricted stock, stock appreciation rights, restricted stock units and other share-based awards under the Amended 2002 Plan.
Only employees of the Company or a subsidiary meeting the requirements of the Internal Revenue Code are eligible to receive “incentive stock options,” within the meaning of Section 422 of the Internal Revenue Code (ISOs) under the Amended 2002 Plan.
|
Shares Available for Awards: | 53,000,000 shares would be available for future awards granted on or following the effective date of the Amended 2002 Plan. The Amended 2002 Plan provides for a decrease in the number of shares of common stock reserved for issuance under the plan (including previously granted awards) to 75,657,296 shares plus shares available for issuance under prior plans immediately prior to the effective date of the 2002 Plan.
|
Award Types: | (1) Non-Qualified Stock Options and Incentive Stock Options
(2) Restricted Stock
(3) Stock Appreciation Rights
(4) Restricted Stock Units
(5) Dividend Equivalent Rights
(6) Other Share-Based Awards
(7) Performance-Based Awards
|
|
| |
Award Terms
(Exercisability Period):
| Options, Stock Appreciation Rights (SARs), and Other Share-Based Awards have a term of no longer than 10 years.
ISOs granted to ten percent owners will have a term of no longer than five years.
|
ISO Limits: | No more than 30,000,000 shares reserved for issuance may be issued upon the exercise of ISOs granted under the Amended 2002 Plan.
|
Minimum Vesting: | Vesting is generally determined by the Compensation Committee within limits set forth in the Amended 2002 Plan, except that no award may fully vest before the first anniversary of the grant date other than a carve-out for up to 5% of the number of shares that are reserved for issuance pursuant to future awards as of the effective date of the Amended 2002 Plan.
|
Not Permitted: | 1) Repricing or reducing the exercise price of a share option or SAR below the per share exercise price as of the date of grant without shareholder approval.
2) Canceling, surrendering or substituting any outstanding option or SAR in exchange for (i) the grant of a new option or SAR with a lower exercise price, or (ii) other awards or a cash payment at a time when the exercise price of the option or SAR is greater than the fair market value of a share.
3) Adding shares back to the number of shares available for issuance when shares are repurchased on the open market with the proceeds of the exercise of an option.
4) Single-trigger change in control vesting acceleration.
5) Payment of dividend or dividend equivalent rights prior to the vesting of the underlying awards.
|
Summary of the Amended 2002 Plan
The following summary of certain material features of the Amended 2002 Plan is qualified in its entirety by reference to the Amended 2002 Plan, which is attached to this proxy statement as Appendix C.
Purpose of the Amended 2002 Plan
The purpose of the Amended 2002 Plan is to benefit the Company’s shareholders by allowing the Company to attract, motivate and retain the best available employees and directors and by providing those employees and directors stock-based incentives to strengthen the alignment of interests between those persons and the Company’s shareholders.
Shares Reserved for Issuance under Amended 2002 Plan
Shares Reserved. As proposed, the total number of212,515 shares of our common stock that are authorized and available for issuance pursuant to awards granted under the Amended 2002 Plan is 75,657,296shares plus shares available for issuance under prior plans immediately prior to the effective date of the 2002 Plan, subject to adjustment in the event of certain changes in the capitalization of the Company. However, only 53,000,000 shares would be available for future awards as of the effective date of the Amended 2002 Plan. As of February 16, 2018, 96,342,704 shares of common stock were available for the grant of future awards under the 2002 Plan. The Amended 2002 Plan provides for a decrease – rather than an increase – in the number of shares of common stock reserved for issuance under the plan.
Shares Reissuable Under Amended 2002 Plan. The following shares are reissuable pursuant to new awards granted under the Amended 2002 Plan: shares that are not issued as a result of the termination, expiration or lapsing of an award for any reason; shares subject to a full value award that are not issued because the award is settled in cash; shares covered by an option surrendered in payment of the exercise or purchase price or in satisfaction of any tax-related items incident to the exercise of an option; or shares that are surrendered in satisfaction of obligations for tax-related items incident to the vesting or settlement of a full value award.
Shares Not Reissuable Under Amended 2002 Plan. Shares repurchased by the Company on the open market with the proceeds of the exercise price from options will be deducted from the aggregate number of shares available for future awards.
Shares Not Counted Against Share Reserve Pool Under Amended 2002 Plan. To the extent permitted by applicable law or any stock exchange rule, shares issued in assumption of, or in substitution for, any outstanding awards of any entity acquired in any form of combination by the Company or an affiliate will not be counted against shares available for grant pursuant to the Amended 2002 Plan. The payment of a dividend equivalent right in cash in conjunction with any outstanding awards will not be counted against the shares available for issuance under the Amended 2002 Plan.
Award Limits
In any calendar year, the maximum number of shares that may be granted to any one participant under the Amended 2002 Plan is 1,500,000 shares, subject to adjustment in the event of specified capitalization events of our company.
Awards
Under the Amended 2002 Plan,November 9, 2021, has submitted the following awards may be granted: stock options (including “incentive stock options” withinproposal:
RESOLVED, Eli Lilly and Company ("Lilly" or the meaning of Section 422 of the Internal Revenue Code), restricted stock, stock appreciation rights, restricted stock units, dividend equivalent rights, other share-based awards, and performance-based awards.
Eligibility
Incentive stock options may be granted only to our employees and to employees of any of our subsidiaries meeting the requirements of the Internal Revenue Code. Awards other than incentive stock options may be granted to our non-employee directors and to employees of the Company and any of its affiliates. As of February 16, 2018,13 non-employee directors and 31,531 employees were eligible to participate in the 2002 Plan.
Administration
The Amended 2002 Plan provides that it will be administered by our Board of Directors, unless"Company") shareholders request the Board of Directors electsadopt as policy (the "Policy"), and amend the bylaws as necessary, to delegate administration responsibilities to a committee. (In this Proxy Statement, we will refer to the Board of Directors or the committee to which administration of the Amended 2002 Plan has been delegated as the “Committee”). The Committee has the sole authority to grant awards, and sole and exclusive discretion to interpret and administer the Amended 2002 Plan. The Committee determines the eligible individuals who will receive grants and the precise terms of the grants (including accelerations or waivers of any restrictions, and the conditions under which such accelerated vesting or waivers occur, such as in connection with a participant’s death). The Committee has the authority to amend or modify the terms of an outstanding award, except that an amendment that materially and adversely impacts the rights under an outstanding award will require prior written consent from the participant, unless the amendment is necessary or desirable to facilitate compliance with applicable law or to avoid adverse tax consequences under Section 409A of the Internal Revenue Code. The decisions of the Committee will be final and binding on all holders of awards. To the extent permitted by applicable law, our Board of Directors also may delegate to a committee of one or more members of our Board of Directors or one or more officers of our company the authority to grant or amend awards to participants other than employees who are subject to Section 16 of the Securities Exchange Act of 1934, as amended, or officers or directors of our company to whom authority to grant or amend awards has been delegated.
Stock Options
The Amended 2002 Plan authorizes the grant of incentive stock options, which are intended to satisfy the requirements of Section 422 of the Internal Revenue Code, and non-qualified stock options, which do not satisfy the requirements of Section 422 of the Code. The exercise price of stock options granted under the Amended 2002 Plan may not be less than 100% (or higher in the case of certain incentive stock options) of the fair market value of a share of our common stock on the date of grant. As of February 16, 2018, the fair market value of a share of our common stock was $78.97. Options granted under the Amended 2002 Plan will vest at the rate specified by the Committee. No stock option will be exercisable more than ten years after the date it is granted.
The Committee determines the methods by which the exercise price of options is paid, including the following: in cash or check, in shares, through a broker-dealer sale and remittance procedure pursuant to which the participant effects a same-day exercise of the option and sale of the purchased shares in order to cover the exercise price for the purchased shares and the applicable withholding taxes, a “net exercise” arrangement pursuant to which the number of shares issuable upon exercise of the option is reduced by a number of shares having a fair market value that would cover the exercise price and tax withholding. In addition, the Committee may provide financial assistance
to a participant who wishes to exercise his or her outstanding options, providedhenceforth that the participant is not an executive officer or memberChair of the Board of Directors, by allowing the participant to deliverwhenever possible, be an interest-bearing full recourse promissory note or through a third-party loan guaranteed by the Company in the amountindependent member of the exercise price andboard. The Policy shall apply prospectively so as not to violate any associated withholding taxes.
Until the shares are issued, no right to vote or receive dividends or any other rights as a shareholder will exist with respect to the shares subject to an option, notwithstanding the exercise of the option. No adjustment will be made for a dividend or other right for which the record date is prior to the date the shares are issued, except in the case of a capitalization event of the Company as provided under the terms of the Amended 2002 Plan.
Restricted Stock Unit
A restricted stock unit represents the equivalent of one share and this type of award is typically awarded to participants without payment of consideration. Restricted stock units may be subject to vesting conditions based upon the passage of time or the attainment of performance-based conditions as determined in the discretion of the Committee. Except as otherwise determined by the Committee at the time of the grant of the award or thereafter, any restricted stock units that are not vested as of the date of the participant’s termination of service will be forfeited. Unlike restricted stock, the stock underlying restricted stock units will not be issued until the restricted stock units have vested. In addition, recipients of restricted stock units generally have no voting or dividend rights until the vesting conditions are satisfied and the underlying shares are issued. Restricted stock units may be settled in shares, cash or a combination of both. The Committee may authorize dividend equivalents to be granted with respect to restricted stock units.
Restricted Stock Awards
An award of restricted stock is a direct grant of common stock, subject to such restrictions on transferability and other restrictions as the Committee may impose (including, without limitation, limitations on the right to vote the underlying shares or the right to receive dividends with respect to the underlying shares). These restrictions may lapse separately or in combination at such times, pursuant to such circumstances, in such installments, or otherwise, as the Committee determines at the time of the grant of the award or thereafter. Restrictions may be based on the passage of time or the attainment of performance-based conditions. Generally, any shares subject to restrictions are forfeited upon termination of employment. The price, if any, that participants are required to pay for each share of restricted stock will be set by the Committee and will be paid in a form approved by the Committee, which may be cash, services rendered or to be rendered to our company or an affiliate of our company, or in another form of payment.
Stock Appreciation Rights
Stock appreciation rights, or “SARs,” typically provide for payments to the holder based upon increases in the price of our shares from the date the SAR was granted to the date that the right is exercised. The Committee will generally determine when the SAR will vest and become exercisable. The grant price of a SAR may not be less than the fair market value of a share on the date of grant of the SAR. The Committee determines the term of a SAR, but no SAR will be exercisable more than ten years after the date it is granted.
A SAR may be granted in connection with an option, either at the time of grant or at any time thereafter during the term of the option. Upon exercise, a SAR granted in connection with an option will entitle the holder to surrender the option or any portion thereof to the extent unexercised, with respect to the number of shares as to which such SAR is exercised. The option will, to the extent and when surrendered, cease to be exercisable. If a related option is exercised in whole or in part, then the SAR related to the shares purchased terminates as of the date of such exercise.
The Committee may elect to settle exercised SARs in cash, in shares, or in a combination of cash and shares. Until the shares are issued, no right to vote or receive dividends or any other rights as a shareholder will exist with respect to the shares subject to a SAR, notwithstanding the exercise of the SAR. No adjustment will be made for a dividend or other right for which the record date is prior to the date the shares are issued, except in the case of a capitalization event as provided under the terms of the Amended 2002 Plan.
Other Share-Based Awards
The Committee is authorized under the Amended 2002 Plan to make any other award that is not inconsistent with the provisions of the Amended 2002 Plan and that by its terms involves or might involve the issuance of shares, or of a right vesting based on the passage of time, the occurrence of one or more events, or the satisfaction of performance criteria or other conditions, or the issuance of any other security with the value derived from the value
of the shares. The Committee may elect to settle these awards in cash, in shares, or in a combination of cash and shares. The Committee may establish the exercise price, if any, of any other share-based awards granted under the Plan, except that the exercise price may not be less than the fair market value of a share on the date of grant for an award that is intended to be exempt from Section 409A of the Internal Revenue Code. The Committee may establish the term of other share-based awards, but it may not exceed ten years. The Committee may authorize dividend equivalents to be paid on other share-based awards.
Performance-Based Awards
The Committee may grant to eligible participants awards that are paid, vest or become exercisable upon the attainment of company performance goals which include, but are not limited to, one or more of the following performance criteria: cash flow (including, without limitation, operating cash flow and free cash flow), earnings per share, gross or net profit margin, net income (either before or after interest, taxes, amortization, and/or depreciation), operating income (either before or after restructuring and amortization charges), return on capital or return on invested capital, return on equity, return on operating assets or net assets, return on sales, sales or revenue, stock price goals or total shareholder return. The Committee intends to define objectively the manner of calculating the performance criteria it selects to use for any applicable performance period.
At the time of grant, the Committee may specify one or more objectively determinable adjustments to one or more of the performance goals. For all performance-based awards, the Committee intends that such determinations shall be made within the first twenty-five percent (25%) of the applicable performance period. No performance-based award may have a performance period with a duration that is less than twelve (12) months.
Notwithstanding the foregoing, while the Committee intends to grant performance-based awards subject to the conditions and procedures outlined above, the Committee may in its discretion grant awards that do not meet such conditions and procedures.
Transferability of Awards
Except as otherwise provided by the Committee, no award granted under the Amended 2002 Plan may be assigned, transferred, or otherwise disposed of by a participant other than by will or the laws of descent and distribution.
Minimum One-Year Vesting Requirement
No award may vest before the first anniversary of the date of grant with the exception of (i) up to five percent (5%) of the number of shares reserved under the Amended 2002 Plan for future awards as of the date the Amended 2002 Plan becomes effective, (ii) awards granted in connection with the assumption or substitution of awards as part of a transaction, and (iii) awards that may be settled only in cash.
Dividends/Dividend Equivalents
To the extent that any dividends or dividend equivalents are payable with respect to a full value award, the dividend or dividend equivalents, as applicable, will not be paid unless the underlying award vests.
Changes in Control
Provided that any applicable award agreement does not preclude the following from applying, in the event of a change in control of our company, each outstanding award that vests solely on the passage of time under the Amended 2002 Plan will immediately vest in the event the award is not converted, assumed, substituted or replaced by the successor corporation, and following the change in control, the awards will immediately terminate. Awards that vest based on the attainment of performance-based conditions shall be subject to the change in control provisions in the applicable award agreement, provided that the agreement does not permit vesting at a rate that is greater than the actual level of attainment and/or provide for pro-rated vesting based on any reduction to the performance period resulting from the change in control. Where awards are assumed, substituted or otherwise continued after a change in control of our company, the Committee may provide that one or more awards will automatically accelerate upon an involuntary termination of the participant’s employment or service within a designated period in connection with the change of control. “Change in control” has a special meaning that is defined in the Amended 2002 Plan.
Adjustments upon Changes in Capitalization
In the event of any stock dividend, stock split, combination or exchange of shares, merger, consolidation, or other distribution (other than normal cash dividends) of assets to our shareholders or any other similar event or change in capitalization affecting our shares other than certain equity restructurings identified in the Amended 2002 Plan, the
Committee has discretion to make appropriate adjustments in the number and type of shares subject to the Amended 2002 Plan, the terms and conditions of any award outstanding under the Amended 2002 Plan, and the grant or exercise price of any such award. In the case of certain equity restructurings as specified in the Amended 2002 Plan, the number and type of securities subject to each outstanding award and the grant or exercise price, if applicable, will be equitably adjusted.
Amendment and Termination of Plan
With the approval of our Board of Directors, at any time and from time to time, the Committee may terminate, amend or modify the Amended 2002 Plan, except that the Board may not, without prior shareholder approval, amend the Amended 2002 Plan in any manner that would require shareholder approval to comply with any applicable laws, rules or regulations. Except as may be required to avoid adverse tax consequences under Section 409A of the Internal Revenue Code or as may be required or desirable to facilitate compliance with applicable law, no termination, amendment or modification of the Amended 2002 Plan may adversely affect in any material way any award granted under the Amended 2002 Plan without the consent of the participant.
Furthermore, absent approval of our shareholders and except as permitted under the provisions of the Amended 2002 Plan dealing with certain capitalization adjustments and change in control, no option or SAR may be amended to reduce the exercise price or grant price of the shares subject to such option or SAR and no option or SAR may be cancelled in exchange for the grant of an option or SAR having a lower per share exercise price or for a cash payment or another award at a time when the option or SAR has a per share exercise price that is higher than the fair market value of the shares.
Clawback/Recovery
Awards are subject to recoupment under any “clawback” policy that the Company adopts for the recovery of awards or payments thereunder in the event of fraud or as required by applicable law or governance considerations or in other similar circumstances.
Plan Term
The Amended 2002 Plan will continue in effect until terminated by our Board of Directors, but no incentive stock options may be granted under the Amended 2002 Plan after the tenth anniversary of the date the amendments to the Amended 2002 Plan were approved by our Board of Directors. Any awards that are outstanding at the time the Amended 2002 Plan terminates will remain in force according to the terms of the Amended 2002 Plan and the applicable agreement evidencing the award.
Federal Income Tax Consequences
The following is a summary of the U.S. federal income tax consequences applicable to equity awards under the Amended 2002 Plan based on current U.S. federal income tax laws. The Amended 2002 Plan is not qualified under Section 401(a) of the Internal Revenue Code. The summary is general in nature and is not intended to cover all tax consequences that may apply to a particular employee, director or to our company. The provisions of the Internal Revenue Code and regulations thereunder relating to these matters are complicated, may change and their impact in any one case may depend upon the particular circumstances. Further, this summary does not discuss the tax consequences of a participant’s death or the provisions of any income tax laws of any municipality, state or foreign country in which a participant may reside.
Nonqualified Stock Options. With respect to nonqualified stock options: (i) no income is recognized by the participant at the time the nonqualified stock option is granted; (ii) generally, at exercise, ordinary income is recognized by the participant in an amount equal to the difference between the option exercise price paid for the shares and the fair market value of the shares on the date of exercise and we are entitled to a tax deduction in the same amount (subject to the restrictions on deductibility described under “Section 162(m) Limitation” below); and (iii) upon disposition of the shares, any gain or loss is treated as capital gain or loss.contractual obligations. If the options are exercised andboard determines that a Chair who was independent when selected is no longer independent, the shares acquired are sold on the same date, generally, the difference between the option exercise price paid for the shares and the sale price is recognized as ordinary income and no capital gain or loss is reported. If required, income tax must be withheld from the participant on the income recognized by the participant upon exercise ofboard shall select a nonqualified stock option.
Incentive Stock Options. The grant of an incentive stock option under the Amended 2002 Plan will not result in any federal income tax consequences to the participant or to our company. A participant recognizes no federal taxable income upon exercising an incentive stock option (subject to the alternative minimum tax rules discussed below), and we receive no deduction at the time of exercise. In the event of a disposition of common stock acquired upon
exercise of an incentive stock option, the tax consequences depend upon how long the participant has held the shares of common stock. If the participant does not dispose of the shares within two years after the incentive stock option was granted, nor within one year after the incentive stock option was exercised, the participant will recognize a long-term capital gain (or loss) equal to the difference between the sale price of the shares and the exercise price. We are not entitled to any deduction under these circumstances.
If the participant fails to satisfy either of these holding periods, he or she must recognize ordinary income in the year of the disposition (referred to as a “disqualifying disposition”). The amount of such ordinary income generally is the lesser of (A) the difference between the amount realized on the disposition and the exercise price or (B) the difference between the fair market value of the common stock on the exercise date and the exercise price. Any gain in excess of the amount taxed as ordinary income will be treated as a long- or short-term capital gain, depending on whether the common stock was held for more than one year. In the year of the disqualifying disposition, we are entitled to a deduction equal to the amount of ordinary income recognized by the participant, subject to possible limitations imposed by Section 162(m) of the Internal Revenue Code.
The “spread” under an incentive stock option (i.e., the difference between the fair market value of the shares at exercise and the exercise price) is classified as an item of adjustment in the year of exercise for purposes of the alternative minimum tax. If a participant’s alternative minimum tax liability exceeds such participant’s regular income tax liability, the participant will owe the larger amount of taxes. The alternative minimum tax will not apply with respect to incentive stock options if the participant sells the shares within the same calendar year in which the incentive stock options are exercised. However, such a sale of shares within the same year of exercise will constitute a disqualifying disposition, as described above.
Stock Appreciation Rights. Upon exercise of a SAR, the participant will recognize ordinary income (treated as compensation) in an amount equal to the excess of the aggregate fair market value of the shares on the date the SAR is exercised over the aggregate exercise price of the SAR. We generally will be entitled to a business expense deduction in the same amount and at the same time as the participant recognizes ordinary compensation income (subject to the limits of Section 162(m) of the Internal Revenue Code). If required, income tax will be withheld from the participant on the income recognized by the participant upon exercise of a SAR.
Restricted Stock. In the absence of a Section 83(b) election (as described below), a participantnew Chair who receives restricted stock will recognize no income at the time of grant. When the restrictions lapse, a participant will recognize ordinary income (treated as compensation) equal to the excess of the fair market value of the stock when the restrictions lapse over the amount paid (if any) for the stock. As the restrictions applicable to a grant of restricted stock lapse (for example, if the restrictions on 20% of a grant lapse on each anniversary of the grant date), the participant will include the applicable portion of the shares that vests as ordinary income (treated as compensation). The participant’s basis in the common stock is equal to the amount included in income on the expiration of the restrictions and the amount paid (if any), and the holding period will begin when the restrictions end. Any disposition of the restricted stock will result in a long- or short-term capital gain or loss, depending on the time the common stock is held after the restrictions end. We generally will be entitled to a deduction equal to the fair market value of the common stock when it is included in the participant’s income, and will also be entitled to a business expense deduction for dividends paid to the participant (if any) on common stock that remains subject to restrictions (in each case subject to the limits of Section 162(m) of the Internal Revenue Code).
If a Section 83(b) election is made within 30 days of the grant of the award, the participant must recognize the fair market value of the restricted stock on the date of grant as ordinary income (treated as compensation) as of the date of grant, and the holding period would begin at the time the restricted stock is granted. We generally would be entitled to a corresponding business expense deduction for the grant, but dividends on the stock would not be deductible. Any subsequent disposition of the stock by the participant, other than by forfeiture, would result in capital gain or loss, which would be long- or short-term, depending on the length of the holding period. Upon a subsequent forfeiture of restricted stock with respect to which a Section 83(b) election has been made, no deduction will be allowed in respect of the amount included as income at the time the Section 83(b) election was made; however, the participant will generally be allowed a loss deduction equal to the amount (if any) the participant paid for the restricted stock over the amount (if any) we paid the participant for the restricted stock at the time it is forfeited.
If required, income tax will be withheld from the participant on the income recognized by the participant at the time the restrictions on the restricted stock lapse (or grant of the restricted stock, in the event the participant makes a Section 83(b) election).
Restricted Stock Units. A participant will not recognize any income at the time a restricted stock unit is granted, nor will we be entitled to a deduction at that time. When payment on a restricted stock unit is made, the participant will recognize ordinary income in an amount equal to the difference between the fair market value of the common stock received (or if the restricted stock unit is settled in cash, the cash amount) and the amount paid as consideration for the units, which will typically be nil. If required, income tax will be withheld on the income recognized by the participant. We will receive a deduction for federal income tax purposes equal to the ordinary income recognized by the participant, subject to the limits of Section 162(m) of the Code.
Performance-Based Awards. A participant will generally not recognize income at the time an award based on achievement of performance objectives is granted, nor will we be entitled to a deduction at that time. When payment on the performance award is made, the participant generally will recognize ordinary income in an amount equal to the fair market value of the common stock received (or if the award is settled in cash, the cash amount). If required, income tax must be withheld on the income recognized by the participant. We will receive a deduction for federal income tax purposes equal to the ordinary income recognized by the participant, subject to the limits of Section 162(m) of the Code.
Dividend Equivalents. A recipient of dividend equivalents generally will recognize ordinary income at the time the dividend equivalent is paid. If required, income tax will be withheld on the income recognized by the participant. We will receive a deduction for federal income tax purposes equal to the ordinary income recognized by the participant, subject to the limits of Section 162(m) of the Code.
Section 162(m) Limitation. Under Section 162(m) of the Internal Revenue Code, income tax deductions of publicly-held corporations are generally limited to the extent total compensation (including base salary, annual bonus, stock option exercises and non-qualified benefits paid) for specified executive officers exceeds $1 million (less the amount of any “excess parachute payments” as defined in Section 280G of the Internal Revenue Code) in any one year.
Section 409A. Section 409A of the Code imposes certain requirements on non-qualified deferred compensation arrangements. These include requirements on an individual’s election to defer compensation and the individual’s selection of the timing and form of distribution of the deferred compensation. If an award under the Amended 2002 Plan is subject to and fails to satisfysatisfies the requirements of Section 409A, the recipientpolicy within a reasonable amount of that award may recognize ordinary income on the amounts deferred under the award,time. Compliance with this policy is waived if no independent director is available and willing to the extent vested, which mayserve as Chair. This policy would be prior to when the compensation is actually or constructively received. Also, if an award that is subject to Section 409A fails to comply with the requirements of Section 409A, Section 409A imposes an additional 20% federal penalty tax on compensation recognized as ordinary income, as well as interest on such deferred compensation.
Future Plan Benefits
Future awards to employees, officers, and directors under the Amended 2002 Plan are generally made at the discretion of the Committee. Therefore, the benefits and amounts that will be received or allocated under the Amended 2002 Plan, as amended,phased in the future are not determinable at this time.
Past Grants under the 2002 Plan
As of February 16, 2018, awards covering 44,257,285 shares of the common stock had been granted under the 2002 Plan. The following table shows information regarding the grants of those awards among the persons and groups identified below.
PRIOR GRANTS UNDER THE 2002 PLAN
|
| | | | | |
| | Options | Restricted Stock
| RSUs | Performance RSUs |
No. of Shares | No. of Shares | No. of Shares | Target No. of Shares | Maximum No. of Shares |
David A. Ricks Chairman, President, and CEO
| 17,779 | — | 20,466 | 590,166 | 877,744 |
Enrique A. Conterno Senior VP and President, Lilly Diabetes and President, Lilly USA
| 35,429 | 11,000 | 76,837 | 524,315 | 783,226 |
Derica W. Rice (retired) Executive VP, Global Services and Chief Financial Officer
| 116,385 | — | — | 993,511 | 1,518,504 |
Jan M. Lundberg, Ph.D. Executive VP, Science and Technology and President, Lilly Research Laboratories
| — | — | 127,871 | 620,024 | 901,590 |
Michael Harrington Senior VP and General Counsel
| 17,746 | 5,000 | 22,778 | 313,188 | 466,151 |
| Current Executive Officers, as a Group | 392,978 | 30,000 | 443,736 | 4,053,170 | 6,045,121 |
Non-Employee Directors, as a Group
| 19,600 | — | — | — | — |
| All current employees who are not executive officers, as a group | 9,201,253 | 55,700 | 10,760,750 | 18,190,202 | 29,334,005 |
Vote Required
The affirmative vote of at least a majority of the outstanding common shares present in person or by proxy at the annual meeting is needed to pass this proposal.
Board Recommendation on Item 6
The Board of Directors recommends that you vote FOR approving the Amended and Restated 2002 Lilly Stock Plan.
Shareholder Proposals
Item 7. Shareholder Proposal Seeking Support for the Descheduling of Cannabis
Fred Pfenninger, 9247 N. Meridian Street, Suite 219, Indianapolis, Indiana, beneficial owner of 79 shares of common stock of Eli Lilly and Company, has submitted the following proposal:
Shareholder Proposal
The proponent requests that the Company announce its support for the descheduling of cannabis.
next CEO transition.
Supporting Statement
Eli Lilly, who wasThe board is responsible for overseeing the Third Presidentactions of Lilly from 1932management. They are expected to 1948, graduated fromact as a counter-power and constructively challenge the Philadelphia Collegeexecutive directors. Conflicts of Pharmacy in 1907interest may arise when one person holds both the Chair and wrote his Doctoral Thesis on "The Comparative Physiological Effects of Several Varieties of Cannabis Sativa." Lilly was a world leader in cannabis based pharmaceutical products in the early 1900s. Lilly sold 23 different cannabis entries in its medical catalog in 1935 before the 1937 Marijuana Tax Act and Reefer Madness halted sales. Parke Davis worked with Lilly to create its own strain Cannabis Americana which Lilly grew in Greenfield, Indiana.
Federal prohibitions outlawing cannabis recreational, industrial and therapeutic use were first imposed by Congress under the Marijuana Tax Act of 1937 and later reaffirmed by federal lawmakers' decision to "temporarily" classify marijuanaCEO positions. In our view, shareholders, as well as the plant's organic compounds known as cannabinoids ascompany, are best served by an independent board Chair who can provide a Schedule I substance underbalance of power between the Controlled Substances ActCEO and the board.
While the CEO's insights and communication can and should be shared with an independently led board, the difference, should the shareholder proposal be implemented, is that those insights will then be subjected to review and oversight by a board led by an independent chair, rather than by a board led by the same person whose insights are being considered. We believe that Lilly's board should adopt best practice governance policies, including having an independent board chair.
In 2019 PricewaterhouseCoopers surveyed over 700 directors, 57% of 1970. This classification, which categorizesdirectors who sat on a board with a combined Chair/CEO stated that it was difficult to voice dissent - a 37% higher result than on boards with an independent Chair.
Pharmaceutical companies are particularly in need of effective and unconflicted oversight because of the plant by statute alongside heroin, defines cannabisindustry's high legal and its cannabinoids as possessingregulatory risks related to product safety and the industry's commercial practices. Eli Lilly is not immune to litigation and regulatory attention. Only in 2021, the company has been repeatedly cited in a high potential for abuse, no currently accepted medical useUS Senate report on significant insulin price increase; the Mississippi Attorney General launched a lawsuit against Lilly, and a lackothers, alleging collusion to keep insulin prices high; and the City of accepted safetyMiami, Florida initiated litigation asserting antitrust and other claims against the company.
The risk of lawsuits, sustained public controversy and regulatory intervention, whether ultimately found to be justified or not, are strong arguments for the useneed for continuous, effective and unconflicted board oversight of the drug.corporate management.
The Controlled Substances Act of 1970 calledIn order to ensure that our board can provide rigorous oversight for the creation ofour Company and management with greater independence and accountability, we urge a special federal commission appointed by Congress and President Nixon to study all aspects of cannabis and report their findings. After 2 years of scientific study the National Commission on Marijuana and Drug Abuse ("Schafer Commission") report "Marijuana: A Signal of Misunderstanding" reported that there was little proven danger of physical or psychological harm, it does not lead to physical dependence, it is not a gateway drug, and no one should go to jail for the private possession of cannabis.
Despite the US Government's nearly century long prohibition of the plant, cannabis is one of the most investigated therapeutically active substances in history. To date there are approximately 22,000 published studies or reviews in the scientific literature referencing the cannabis plant and its cannabinoids, nearly half of which were published within 10 years according to a key work search on the search engine PubMed Central.
The late 1980s discovery of the endogenous cannabinoid system, with specific receptors and ligands, has progressed our understanding of the therapeutic actions of cannabis. The cannabinoid system evolved with our species and is intricately involved in normal physiology -control of movement; pain, reproduction, memory, appetite.
Cannabis oil kills cancer, prevents and reverses dementia, prevents epileptic seizures, and extends longevity among other things. Cannabis is the most medicinal plant on the planet.
vote FOR this shareholder proposal.
Statement in Opposition to the Shareholder Proposal Regarding Support forto Amend the Descheduling of Cannabis
Bylaws to Require an Independent Board ChairThe Public Policyboard, after review by its Directors and ComplianceCorporate Governance Committee, has reviewed and recommends a vote against this proposal. We
If implemented, the proposal would lock in a mandatory board leadership structure that eliminates our board’s flexibility to evaluate and adopt what it believes to be the most effective leadership structure for Lilly under the relevant facts and circumstances at any given point in time. Unlike the proponent, the board believes, whether in the present or after the next CEO transition, that there is no “one-size-fits-all” approach to board leadership and recognizes that two of its key responsibilities are to evaluate and implement the leadership structure best suited to achieve the company’s objectives and to promote the long-term interests of its shareholders with due regard for all our stakeholders. In 2021, the board again undertook an assessment of its leadership structure in the context of our business, long-term strategy and industry environment, and developments in corporate governance, and believes that a combined chair and CEO, coupled with a strong lead independent director position, continues to be in the best interest of the company and our shareholders.
Lilly’s board leadership structure is consistent with market practice and our flexible approach was strongly endorsed by Lilly’s shareholders last year.
There is no singular approach to independent board leadership across S&P 500 companies. Notably, as of December 31, 2021, only 36% of companies in the S&P 500 have finite resourcesindependent chairs and, like Lilly, 57% of companies in the S&P 500 instead have lead independent directors,1 and six of the eight U.S.-incorporated companies from our peer group (see “Compensation Discussion and Analysis—Compensation Committee’s Processes and Analyses—Competitive Pay Assessment” for advocacy,a list of our peer group companies)2 have a combined board chair and CEO. In addition, Lilly’s shareholders decidedly rejected similar proposals seeking to mandate an independent board chair at both our 2020 and 2021 annual meetings of shareholders, with nearly two-thirds of the votes cast against the proposal, thus endorsing our flexible approach.
Lilly’s current board leadership structure and corporate governance practices provide effective, independent oversight of management.
Lilly has a strong independent board that operates under sound principles of corporate governance. See “Governance—Highlights of the Company’s Corporate Governance—Leadership Structure; Oversight of Chair, CEO, and Senior Management” for a description of the board’s governance principles. Although the chair and CEO roles are combined, we ensure independent oversight of the company through a counterbalancing governance structure, which we must limithave had since 2006 through either a lead independent director or presiding director. Further bolstering independent oversight, each of our current board members other than the CEO is independent (12 out of 13 directors), and focusall standing board committees are made up solely of independent directors and led by independent committee chairs.
Lilly’s lead independent director is elected annually by and from among the independent directors of the board.Our strong lead independent director is empowered with clearly defined responsibilities including:
•leading the board’s processes for selecting the CEO;
•overseeing the independent directors’ annual performance evaluation of the chair and CEO;
•serving as a liaison between the chair and the independent directors;
•presiding at all meetings of the board at which the chair is not present;
•presiding at executive sessions of the independent directors;
•calling meetings of the independent directors, as appropriate;
•approving meeting agendas and schedules and reviewing information to be provided to the board;
•being available for consultation and direct communication with shareholders, as appropriate;
1 Source: EY Center for Board Matters, Corporate Governance by the Numbers. December 31, 2021.
2 Biogen has an independent board chair and Merck’s former CEO and Chair retired as CEO in 2021 and remains on Merck’s board of directors as executive chair.
•together with the chair and the chair of the Directors and Corporate Governance Committee, conducting the annual board assessment process;
•together with the Directors and Corporate Governance Committee, leading the director succession planning process; and
•retaining advisors for the independent directors, as appropriate.
Furthermore, the board has instituted a number of governance best practices to ensure effective independent oversight, including:
•executive sessions of the independent directors held after every regular board meeting that are presided over by our lead independent director;
•an annual performance evaluation of the chair and deschedulingCEO conducted by the independent directors, the results of cannabiswhich are reviewed with the CEO and considered by the Compensation Committee and independent directors in establishing the CEO’s compensation for the next year;
•an annual performance evaluation of the lead independent director by the board as part of the annual board assessment process;
•independent director access to management whenever deemed necessary by the independent directors; and
•the ability of independent directors and all committees to retain their own independent advisors, at the company’s expense, whenever they deem it desirable to do so.
Lilly’s current governance structure provides effective, independent oversight over key matters that are important to our stakeholders, including drug pricing and access.
Our independent directors are deeply engaged in key matters important to Lilly and our stakeholders, including the oversight over the company’s approach to drug pricing and access. Guided by this active oversight, Lilly already has taken numerous steps to promote drug affordability and access. For example, Lilly introduced two additional lower-priced versions of Humalog in January 2020 and added the Lilly Insulin Value Program to Lilly’s comprehensive suite of insulin affordability solutions in September 2020, which now enables customers with commercial insurance or no insurance to purchase their monthly prescription of all Lilly insulins for $35. In addition, in September 2021, Lilly announced that it would lower the list price of all Lilly’s non-branded insulins, including Insulin Lispro Injection, by an additional 40% in the U.S. That price reduction took effect January 1, 2022, effectively reducing the list price to 2008 levels. These examples, among others, demonstrate Lilly’s commitment to providing effective oversight over drug pricing and access.
Our board of directors believes that our shareholders are best served by preserving the flexibility to determine the appropriate leadership structure for the company in light of the circumstances at the relevant time.
We believe the proposal would unnecessarily restrict the board’s ability to exercise its fiduciary duty to determine the board leadership structure most appropriate for the company given the specific circumstances and leadership needs at any particular point in time. The company’s robust governance framework ensures that board leadership is balanced with independent participation given the extensive involvement and approval authority of the lead independent director and his oversight. Our independent directors also collectively bring to the board vast leadership experience, industry expertise, and other critical skills, and individually have demonstrated the willingness to think and act independently on behalf of shareholders. Therefore, adopting a proposal that would limit the board’s ability to exercise decision-making on the appropriate leadership is not in shareholders’ best interests.
We believe independence is essential to strong corporate governance. The combination of a chair who is also the CEO and a lead independent director assigned specific duties and empowered with robust authority achieves the delivery of multiple balanced inputs to the board. Having one individual serve as both chair and CEO provides the board with deep insights to drive long-term strategy and execution and allows consistent communication throughout the company. This is vital to our innovative research and development business with prolonged product development cycles. Further, the lead independent director, currently a sitting CEO, drives an “outside in” analysis of company decisions and performance, maintains frequent contact with our chair and CEO to ensure a productive partnership between our independent directors and management and leads our independent directors in their important oversight function. This leadership structure has served our shareholders well.
Our current governance structure enables us to be responsive to shareholders’ preferences—for example, this year we are submitting a management proposal to allow shareholders to amend the company’s bylaws.
Our current governance structure enables us to be responsive and accountable to shareholders. For example, this year we have submitted a management proposal to amend our articles of incorporation to give shareholders the ability to amend the company’s bylaws (see Item 6) with a majority vote standard. In addition, as we have done each of the past four years, we are also resubmitting management proposals to eliminate the classified board structure and supermajority voting requirements (see Items 4 and 5), which reflects our continued commitment to the interests of our core priorities. We focusshareholders.
Our current governance structure appropriately balances the company’s need for continuity in its leadership with independent and effective oversight.
Lilly’s independent directors have determined that Mr. Ricks is eminently qualified to serve as both chair and CEO, and the board believes that having him fill that combined role, complemented by Mr. Luciano, a strong lead independent director, strikes an appropriate balance between consistent leadership and independent and effective oversight that is optimal for the company and our resourcesshareholders. For additional information on the particular qualities of Mr. Ricks and why he is best suited to support organizations that champion public policies that contribute to pharmaceutical innovation, healthy patients,serve as chair at this time, as well as information on the leadership provided by Mr. Luciano, the lead independent director, please see “Governance—Highlights of the Company’s Corporate Governance—Leadership Structure; Oversight of Chair, CEO, and Senior Management.”
For these reasons, we believe a healthy business climate. Thepolicy requiring an independent board chair is not necessary and not in the best interests of the company is also actively engaged in public policy discussions that relate to our current products and other important topics related to drug pricing.
its shareholders.
Board Recommendation on Item 7
The Board of Directorsboard recommends athat you vote AGAINST thethis proposal.
Item 8. Shareholder Proposal Requestingto Publish an Annual Report Regarding Direct and Indirect Political ContributionsDisclosing Lobbying Activities
The Comptroller of the State of New York, Thomas P. DiNapoli, trustee of the New York State Common Retirement Fund and the administrative head of the New York State and Local Retirement System,SEIU Master Trust, 1800 Massachusetts Ave. NW, Suite 301, Washington, D.C. 20036-1202, beneficial owner of 2,967,282 shares of our common stock having a market value in excess of $2000, has submitted the following proposal:
Shareholder Proposal
The proponent seeks a report from the company regarding its direct and indirect political contributions.
Supporting Statement
Whereas, we believe in full disclosure of Eli Lilly and Company's ("Lilly"Company’s (“Lilly’s”) direct and indirect lobbying activities and expenditures to assess whether Lilly'sits lobbying is consistent with its expressed goals and in the best interests of shareholders.
Resolved,the shareholders of Lilly request the preparation of a report, updated annually, disclosing:
1.Company policies and procedures governing lobbying, both direct and indirect, and grassroots lobbying communications.
2.Payments by Lilly used for (a) direct or indirect lobbying or (b) grassroots lobbying communications, in each case including the amount of the payment and the recipient.
| |
1. | Company policy and procedures governing lobbying, both direct and indirect, and grassroots lobbying communications. |
3.Lilly’s membership in and payments to any tax-exempt organization that writes and endorses model legislation.
| |
2. | Payments by Lilly used for (a) direct or indirect lobbying or (b) grassroots lobbying communications, in each case including the amount of the payment and the recipient. |
| |
3. | Lilly's membership in and payments to any tax-exempt organization that writes and endorses model legislation. |
| |
4. | Description of the decision making process and oversight by management and the Board for making payments described in section 2 and 3 above. |
4.Description of management’s and the Board’s decision-making process and oversight for making payments described in sections 2 and 3 above.
For purposes of this proposal, a "grassroots“grassroots lobbying communication"communication” is a communication directed to the general public that (a) refers to specific legislation or regulation, (b) reflects a view on the legislation or regulation and (c) encourages the recipient of the communication to take action with respect to the legislation or regulation. "Indirect lobbying"“Indirect lobbying” is lobbying engaged in by a trade association or other organization of which Lilly is a member.
Both "direct“direct and indirect lobbying” and "grassroots“grassroots lobbying communications"communications” include efforts at the territory, local, state and federal levels.
The report shall be presented to the AuditPublic Policy and Compliance Committee or other relevant oversight committees and posted on Lilly'sLilly’s website.
Supporting Statement
We encourage transparency in the use of corporate fundsDrugmakers spend more to influence legislation and regulation, both directly and indirectly. Sincelobby Washington than any other industry.1 Lilly spent $88,362,000 from 2010 Lilly has spent over $64 million- 2020 on federal lobbying (opensecrets.org). This figure does not include lobbying expenditures to influence legislation in states,lobbying. Lilly lobbies extensively at the state level where Lilly also lobbies in 48 states ("Amid Federal Gridlock, Lobbying Rises in the States," Center for Public Integrity, February 11, 2016), but disclosure is uneven or absent.absent, with at least 144 lobbyists in 44 states in 2020 (followthemoney.org). Lilly lobbies abroad, spending between €700,000-799,000 on lobbying in Europe.
Lilly is a member offails to disclose its third-party payments to trade associations and social welfare organizations, or the amounts used for lobbying, to shareholders. Companies can give unlimited amounts to third party groups that spend millions on lobbying and often undisclosed grassroots activity, and these groups may be spending “at least double what’s publicly reported.”2 For example, Pharmaceutical Research and Manufacturers of America (PhRMA), which spent over $100 million fighting a California drug pricing initiative ("Big Pharma Fights 'Tooth and Nail' against California Drug Vote," has given millions to controversial “dark money” social welfare groups like the American Action Network. Bloomberg, 3
October 25, 2016),Lilly chairs the board of PhRMA and belongs to the U.S. Chamber of Commerce, which hastogether have spent over $1.3$2.1 billion on lobbying since 1998. Lilly does not disclose its payments1998, and supports social welfare organizations that lobby, like the Alliance for Patient Access, “which claims to trade associations, or the amounts used for lobbying. be pro-consumer but consistently advocates against policies to lower drug prices.”4
1 https://www.opensecrets.org/news/2021/04/vaccine-access-pharma-lobbying-fight/.
2 https://theintercept.com/2019/08/06/business-group-spending-on-lobbying-in-washington-is-at-least-double-whats-publicly-reported/.
3 https://www.opensecrets.org/news/2019/ll/big-phanna-bankrolled-conservative-groups-tax-retums-show/.
4https://prospect.org/power/astroturf-campaign-attacks-discount-drug-program-for-poor/.
5https://www.opensecrets.org/news/2021/09/phannaceutical-industrv-backs-democratic-holdouts-on-drug-pricing-plan/.
We are concerned that Lilly'sLilly’s lack of trade association lobbying disclosure presents reputational risks.risk when its lobbying contradicts company public positions. For example, Lilly believes in providingstates it supports more affordable medicines, yet helps fund PhRMA'sfunds PhRMA’s opposition to lower prescription drug price initiatives, andprices.5 And while Lilly supports smoking cessation, yet the Chamber worksopposed Indiana voter restrictions, groups have asked Lilly to block global smoking laws.
And Lilly does not disclose its contributions to tax-exempt organizations that write and endorse model legislation, such as its membership incut ties with the American Legislative Exchange Council (ALEC). Lilly's ALEC membership has drawn media scrutiny ("Kendall: Businesses Should Cut Ties with Union-busting Lobbyists," “because of its voter restriction efforts.”Indianapolis Star6, July 27, 2016). Over 100 companies have publicly left ALEC, including Allergan, Amgen, AstraZeneca, GlaxoSmithKline, Medtronic and Merck.
Statement in Opposition to the Shareholder Proposal Requestingto Publish an Annual Report Regarding Direct and Indirect Political Contributions
Disclosing Lobbying ActivitiesThe Public Policyboard, after review by its Directors and ComplianceCorporate Governance Committee, of the board has reviewed this proposal and recommends a vote against this proposal.
The proposal calls for the board to publish an annual report to shareholders disclosing the company’s direct and indirect lobbying activities and expenditures to assess whether its lobbying is consistent with its expressed goals and in the best interest of shareholders.
The report requested by the proposal is unnecessary because Lilly already provides shareholders with extensive disclosures regarding the company’s direct and indirect lobbying activities and expenditures, and information necessary to assess the consistency of the company’s lobbying activities with its expressed goals through existing disclosures. Given the focus of our stakeholders on this topic, in November 2021 we substantially enhanced our disclosures with respect to Lilly’s direct and indirect lobbying activities, for example, disclosures regarding:
•our board’s oversight of political expenditures and lobbying activities;
•Lilly’s political contributions to candidates for public office (directly from Lilly and through the company’s exclusively employee-led political action committee (the “LillyPAC”));
•recipients of Lilly’s political contributions;
•Lilly’s federal and state lobbying activities; and
•Lilly’s trade association memberships to which Lilly pays annual membership dues of $50,000 or more (including those trade associations where Lilly has a board seat).
These extensive disclosures already enable shareholders to assess whether the company’s lobbying activities are consistent with its expressed goals and in the best interest of shareholders.
In addition, the company already directs its public policy engagement toward offering Lilly’s perspective in a manner that supports access to innovative medicines and advocates for improvements to the U.S. healthcare system and, accordingly, Lilly’s policies and procedures relating to its engagement in political activities already seek to align the company’s lobbying activities with its expressed goals. Moreover, contrary to the proposal’s assertions, Lilly does not fund opposition to lowering prescription drug prices. Lilly does, however, oppose legislative policies that would hinder medical discovery for patients. For example, we opposed H.R. 3 because it would not only stifle innovation but there are better policy solutions to help patients with their out of pockets costs such as Medicare Part D modernization and not requiring deductibles for insulin coverage. Similarly, we currently publish mosthave opposed the drug pricing aspects of the Build Back Better Act that would also have negative consequences to drug discovery without directly helping patients—such as Medicare negotiation. Our political expenditures, however, cover the political spectrum and reflect our approach to engaging stakeholders on various complex issues to help preserve incentives and ensure an environment conducive to finding new treatments.
Lilly already publicly discloses the information requested by the shareholder.proposal regarding the company’s policies and procedures governing direct and indirect lobbying activities.
In this proxy statement, the political participation page of Lilly’s website (the “Political Participation Website”), and Lilly’s corporate governance guidelines, the company discloses its extensive policies and procedures governing direct and indirect lobbying activities. For example, the Political Participation Website discloses:
•Lilly’s commitment, through public policy engagement, to provide a way for all our locations globally to offer Lilly’s perspective on the political environment in a manner that supports access to innovative medicines, as well as to look for ways to engage on issues specific to local business environments;
•that Lilly employees must comply with our global policies, core values and legal obligations, which are outlined in our written code of business conduct, The additional reporting requirementsRed Book;
•that when engaging in lobbying activities Lilly complies with the laws that govern such activities, Lilly’s vice president, U.S. government affairs is responsible for overseeing these activities and the company’s general counsel and chief financial officer, or their designees, also approve all corporate political contributions before they are unnecessary, asmade; and
•that, where permitted, Lilly makes lawful political contributions in the United States to political candidate committees, political parties, political action committees, ballot measure committees, associations and other political organizations operating under section 527 of the Internal Revenue Code.
6 https://www.thenation.com/article/politics/alec-corporations-democracv/.
Lilly already publicly discloses the information requested isby the proposal regarding our direct and indirect lobbying expenditures.
Lilly makes extensive disclosures regarding its direct and indirect lobbying expenditures in its Political Participation Website, proxy materials, the environmental, social and governance (“ESG”) page of its website, and through other publicly available disclosures regarding its political activities. The trade associations through which Lilly conducts its indirect lobbying activities (which are evaluated annually by the Company’s U.S. government affairs leaders) also publicly disclose their lobbying expenditures. As noted in the bullet points above, in November 2021 Lilly substantially enhanced its disclosures related to its direct and this reporting would placeindirect lobbying activities, including lobbying expenditures. Lilly voluntarily discloses its corporate political contributions and expenditures on an undue administrative burdenannual basis on the company.
BeginningPolitical Participation Website. The proposal also requests we disclose our grassroots lobbying communications, however, Lilly does not engage in 2005, the company has published the following information on our website (www.lilly.com) for both direct company contributions and employee political action committee (PAC) contributions to support candidates for political office, political parties, officials, or committees in the U.S.:
policies and procedures for company and PAC contributions
contributions to candidates, including information about the candidate's office (for example, state, local, or federal; House or Senate), party affiliation, and state
contributions to political organizations and Section 527 organizations reported by state.
This information is updated annually. In additiongrassroots lobbying communications to the information available on our website, detailedgeneral public. The proposal also notes that groups have asked Lilly to cut ties with the American Legislative Exchange Council (“ALEC”). Lilly is no longer a member of ALEC. Additional details regarding the company’s corporate contributions, PACLillyPAC’s contributions (which are also voluntarily disclosed on an annual basis), contribution data, and the company’s direct lobbying expenses are also already made available to the public onfrom numerous public sources.
Lilly already publicly discloses the Federal Election Committee website (https://www.fec.gov/data/)information requested by the proposal regarding our membership in, and through individualpayments to, any tax-exempt organizations that writes and endorses model legislation.
As noted above, Lilly’s Political Participation Website also contains information regarding the company’s U.S. trade association memberships for which the company pays annual dues of $50,000 or more and information regarding the percentage of dues collected by such trade associations utilized for federal and state agencies. The company’s direct lobbying expenses are also available toand political expenditures. In the public on the Lobbying Disclosure page of the U.S. House website (http://disclosures.house.gov/ld/ldsearch.aspx) and through individual state agencies.
In addition to direct political contributions,Political Participation Website, Lilly maintainsalready discloses Lilly’s memberships in certain 501(c)(6)s - trade associationsorganizations that report lobbying activity to the U.S. government. We maintain memberships in trade associations and other tax-exempt organizations specific to business and pharmaceutical industry interests, such as PhRMA (Pharmaceuticalfederal government, including the Pharmaceutical Research and Manufacturers Association), BIO (Biotechnology Association), andof America, the National Association of Manufacturers. We support organizations that champion public policies that contribute to pharmaceutical innovation, healthy patients, and a healthy business climate.
Information relating to Lilly’s memberships in trade associations to which we contribute $50,000 per year or more, and any such organizations where Lilly has a board seat can be found at https://www.lilly.com/LillyPAC.
These tax exempt organizations are also required to disclose their lobbying expenditures underManufacturers, the Lobbying Act of 1995: they report their lobbying expenditures toBiotechnology Innovation Organization, the U.S. Senate.Chamber of Commerce and the Business Roundtable.
Lilly already publicly discloses the information requested by the proposal regarding its decision-making process and the board’s oversight over direct and indirect lobbying activities.
As weIn this proxy statement and the Political Participation Website, Lilly already discloses its decision-making process and the board’s oversight over the company’s political expenditures and direct and indirect lobbying activities to ensure the company fulfills its commitment to stewardship of corporate funds and risk minimization with respect to such activities.
For example, the board receives regular updates from the senior vice president, corporate affairs, and communications regarding public policy issues and the company’s direct and indirect lobbying activities and receives semi-annual updates on direct and indirect lobbying activities and trade association memberships. See “Governance—Highlights of the Company’s Corporate Governance—Political and Policy Participation” for additional information.
Requiring Lilly to prepare a separate report with this information would place an undue administrative burden on the company and would not provide meaningful additional information to shareholders.
We do not control what portion ofbelieve the organization’s budget is spent on lobbying, it is the fact of company membership in and support for the trade association, and the trade association’s total lobbying expenditure, that reveals the most about Lilly's political activities. As a result, the board of directors does not believe any value providedadditional disclosures contemplated by the requestedproposal would provide meaningful additional disclosures meritsinformation to shareholders to merit the resources required to produce such aprovide the requested report.
For these reasons, we believe the proposal is not in the best interests of the company and its shareholders.
Board Recommendation on Item 8
The Board of Directorsboard recommends athat you vote AGAINST thethis proposal.
Item 9. Shareholder Proposal Requesting Report on Policiesto Disclose Lobbying Activities and Practices Regarding Contract Animal LaboratoriesAlignment with Public Policy Positions and Statements
People for the Ethical Treatment of Animals (PETA), 1536 16thCommonSpirit Health, 444 West Lake Street, N.W., Washington, D.C.,Chicago, IL 60606, beneficial owner of 56 shares of our common stock having a market value in excess of Eli Lilly and Company,$2000, has submitted the following proposal:
Establish Accountability for Animal Welfare
RESOLVED, in light of disturbing mistreatment of animals at external research organizations with which our Company has conducted business,Resolved: Shareholders request that the Board of Directors commission and publish a third party review within the next year (at reasonable cost, omitting proprietary information) of whether Eli Lilly and Company’s (“Lilly’s”) lobbying activities (direct and through trade associations) align with Lilly’s public policy position and public statements, particularly supporting “making medicines more accessible and affordable to patients” and “fairness and transparency in the biopharma industry.”1 The report should strengthen our Company's policydiscuss how Lilly addresses the risks presented by any misaligned lobbying and practices regarding contract animal laboratories and issue a reportits plans, if any, to shareholders.mitigate these risks.
1https://www.lilly.com/policies-reports/public-policy.
Supporting Statement
In spite of its "commitment to the ethical treatment of animals," which extends to external laboratories, our Company has repeatedly conducted business with contract laboratories where substandard animal care practices have been documented by government agencies.
Our Company's animal care policy states that “animals used in research shall be treated humanely, with pain or distress eliminated or minimized." Additionally, our Company requires all contract research organizations "to adhere to [its animal welfare] policies and principles." Yet our Company has paid for services conducted at and purchased animals from at least three contract laboratories—Liberty Research, Inc. (Liberty), Professional Laboratory and Research Services (PLRS), and Covance—with serious violations of federal animal welfare laws.
A 2017 exposé of Liberty conducted by People for the Ethical Treatment of Animals (PETA) documented, including on video, living and dying conditions for dogs and cats marked by pain and misery. Workers failed to provide adequate anesthesia to dogs whose skulls were opened during invasive surgery and failed to administer humane euthanasia. Liberty used animals in multiple tests despite the long-term effects of experimental compounds and possible interactions with other medications. Cats were forced to live in severely crowded, barren, windowless pens where recently, some suffocated under litter pans; and dogs suffered severe injuries after being confined with incompatible cagemates.
Supporting Statement:
OurLilly’s commitment to Health Above All is in opposition to its lobbying efforts. Lilly says, “We’re dedicated to making our medicines more equitable, accessible and affordable,” and clearly states, “no one should have to ration their insulin.”2 Yet, Lilly is among three insulin manufacturers explicitly called out for price collusion in a 2017 class action lawsuit.3
Lilly states, “Now more than ever, it’s vitally important that we demonstrate accountability and trustworthiness so we can continue to earn the confidence of patients, healthcare providers and other customers, as well as society as a whole.” However, Lilly has directly lobbied against drug pricing reform that advances affordability,4 hiring three lobbyists in March 2021 to defeat Democratic drug pricing proposals even while Lilly was under intense scrutiny for insulin price hikes.5
Lilly’s CEO Dave Ricks is now the Board Chair for Pharmaceutical Research and Manufacturers of America (“PhRMA”), which raised nearly $527 million in 2020 and spent roughly $506 million, including donating millions to numerous other organizations for use in opposing congressional drug pricing reform efforts.6 PhRMA also sits on the board of the American Legislative Exchange Council, which has actively opposed H.R. 3 and its moderate counterpart S. 2534 (both 116th Congress) - bills to lower the costs of pharmaceuticals.7
Lilly is the fourth largest lobbying spender ($166.2M) and the third highest campaign contributor ($13.3M) between 1999 and 2018. Lilly was among several pharmaceutical companies that gave $1.6M to lawmakers in the first half of 2021, targeting legislators who were likely to oppose drug pricing reforms in the Build Back Better Act.8
The positions Lilly adopts should not be undermined by lobbying efforts undertaken by organizations the Company supports financially. A company also contracts with Covance, which was cited and finedmay not support every position taken by the U.S. Departmenttrade associations to which it belongs, but proper risk management requires that the board be aware of Agriculture (USDA) in 2016 when negligence resulted in thirteen monkeys dying of hyperthermia.inconsistencies and evaluate salient risks that would require mitigation.
Given Lilly’s extensive direct and indirect lobbying against measures that would make drugs more affordable, we are concerned that the misalignment between Lilly’s lobbying and its stated position with regard to equity, access and affordability creates reputational risk.1 According to recent federal inspections, beagles and monkeys at Covance were denied adequate veterinary care for numerous ailments: monkeys sustained limb fractures and beagles were not adequately treated for inflamed and painful skin. A rabbit was euthanized after she was found with a bell stuck in her mouth. Another rabbit was euthanized after she sustained a spinal injury.9
Apparent carelessness in choosing outside laboratories is a long-standing issue for our Company. A 2010 PETA video exposé of PLRS documented repeated violations of federal laws. Workers yelled profanities at cowering, frightened dogs and cats. Employees kicked, threw, dropped, and dragged dogs, and violently threw cats into cages. Animals at PLRS were forced to live in their own feces and urine and suffered constantly from burns and sores-but received no veterinary care for their wounds. Following the release of the video, and inspection by the USDA, this laboratory was forced to close.
Shareholders cannot monitor what goes on inside animal testing laboratories, but our Company can and must review federal records and conduct frequent and extensive visits to contract laboratories. The Board must ensure that animal welfare measures are an integral part of our Company's corporate stewardship.
WeFor these reasons, we urge shareholders to vote in favor of this socially and ethically importantsupport the proposal.
1http://www.mediapeta.com/peta/PDF/Covance_Research_Products.Stip.July2016.pdf
Statement in Opposition to the Shareholder Proposal Requestingto Publish an Annual Report on PoliciesDisclosing Lobbying Activities
The board, after review by its Directors and Practices Regarding Contract Animal Laboratories
We share the concerns raised in this shareholder proposal. We abhor mistreatment of animals and we are committed to the appropriate treatment of animals in research. However, for reasons stated below, the Public Policy and ComplianceCorporate Governance Committee, of the board has reviewed this proposal and recommends a vote against it.this proposal.
The proposal calls for the board to publish a report to shareholders disclosing whether the company’s lobbying activities align with its public policy positions and statements, particularly regarding Lilly’s goals of promoting access and affordability of medicines and fairness and transparency in the biopharmaceutical industry.
OfThe report requested by the violations cited by PETAproposal is unnecessary because Lilly already provides shareholders with sufficient information to assess the alignment of the company’s lobbying activities with its public positions and statements through existing disclosures. Given the focus of our stakeholders on this topic, in their proposal,November 2021 we substantially enhanced our disclosures with respect to Lilly’s direct and indirect lobbying activities, including, without limitation, disclosures regarding:
•our board’s oversight of political expenditures and lobbying activities;
•Lilly’s political contributions to candidates for public office (directly from Lilly and through the company’s exclusively employee-led political action committee (the “LillyPAC”));
•recipients of Lilly’s political contributions;
•Lilly’s federal and state lobbying activities; and
•Lilly’s trade association memberships to which Lilly pays annual membership dues of $50,000 or more (including those trade associations where Lilly has terminated relationshipsa board seat).
These extensive disclosures already enable shareholders to assess whether the company’s lobbying activities align with oneits public policy positions and statements, as well as to assess the risks presented by any potential misalignment.
2https://www.lilly.com/who-we-are/health-above-all.
3https://www.fiercepharma.com/pharma/novo-faces-new-shareholder-suit-for-alleged-collusive-price-fixing.
4https://www.wfyi.org/news/articles/eli-lilly-says-legislation-to-address-drug-prices-would-hurt-its-current-future-operations.
5https://www.opensecrets.org/news/2021/04/vaccine-access-pharma-lobbying-fight/.
6https://www.opensecrets.org/news/2020/12/pharma-lobby-poured-millions-into-darkmoney-groups/.
7https://www.alecaction.org/update/20-alec-lawmakers-say-no-to-importing-price-controls-and-socialized-medicine-to-america.
8https://www.tampabay.com/news/health/2021/10/27/big-pharma-spends-big-to-block-attempts-to-control-drug-prices/.
9https://www.opensecrets.org/orgs/eli-lilly-co/lobbying?id=d000000166
In addition, the company already directs its public policy engagement toward offering Lilly’s perspective in a manner that supports access to innovative medicines and advocates for improvements to the U.S. healthcare system and, accordingly, Lilly’s policies and procedures relating to its engagement in political activities already seek to align the company’s lobbying activities with its expressed goals. Moreover, contrary to the proposal’s assertions, Lilly does not fund opposition to lowering prescription drug prices. Lilly does, however, oppose legislative policies that would hinder medical discovery for patients. For example, we opposed H.R. 3 because it would not only stifle innovation but there are better policy solutions to help patients with their out of pockets costs such as Medicare Part D modernization and not requiring deductibles for insulin coverage. Similarly, we have opposed the drug pricing aspects of the three laboratories.Build Back Better Act that would also have negative consequences to drug discovery without directly helping patients—such as Medicare negotiation. Our political expenditures, however, cover the political spectrum and reflect our approach to engaging stakeholders on various complex issues to help preserve incentives and ensure an environment conducive to finding new treatments.
Lilly already publicly discloses the information requested by the proposal regarding the company’s lobbying activities.
Lilly makes extensive disclosures regarding its lobbying activities in the political participation page of its website (the “Political Participation Website”), proxy materials, the environmental, social and governance (“ESG”) page of its website, and through other publicly available disclosures regarding its political activities. As noted in the bullet points above, in November 2021 Lilly substantially enhanced its disclosures related to its lobbying activities. In addition, Lilly voluntarily discloses its corporate political contributions on an annual basis. In addition to the public disclosures of lobbying expenditures by trade associations through which Lilly conducts its indirect lobbying activities (which are evaluated annually by the company’s U.S. government affairs leaders), Lilly’s Political Participation Website also contains information regarding the company’s trade association memberships for which the company pays annual dues of $50,000 or more, as well as information regarding the percentage of dues collected by such trade associations utilized for federal and state lobbying and political expenditures. Additional details regarding the company’s corporate contributions, LillyPAC’s contributions (which are also voluntarily disclosed on an annual basis), contribution data, and the company’s direct lobbying expenses are also already made available to the public from numerous public sources.
Lilly already publicly discloses the information requested by the proposal regarding its public policy positions and statements with regard to accessibility and affordability and fairness and transparency.
In this proxy statement, the Political Participation Website and Lilly’s public policy website, the company discloses its public policy positions regarding accessibility and affordability as well as fairness and transparency in the biopharmaceutical industry. These disclosures include Lilly’s commitment to advocate for improvements to the U.S. health care system that align with the company’s core principles of: (1) encouraging and protecting innovation; (2) fairness and transparency in the biopharmaceutical industry and all of health care; and (3) lowering costs at the pharmacy counter for patients who use our medicine. Lilly’s lobbying activities are aligned with these public policy positions and our actions also clearly demonstrate this alignment. For example, Lilly advocates for insurers to pass through its negotiated rebates directly to consumers at the second laboratory, work has been curtailedpharmacy counter and confinedis supportive of efforts to exempt health care services for chronic conditions from a single sitehealth insurance plan’s deductible. On the ESG page of its website, Lilly also discloses its support for various transparency initiatives globally, and the company seeks to collaborate with policy makers, industry colleagues and key stakeholders to align on approaches that achieve these objectives. As demonstrations of delivering on its commitment to its public policy positions regarding drug pricing and access, Lilly introduced two additional lower-priced versions of Humalog in January 2020 and added the Lilly Insulin Value Program to Lilly’s comprehensive suite of insulin affordability solutions in September 2020, which now enables customers with commercial insurance or no insurance to purchase their monthly prescription of all Lilly insulins for $35. In addition, in September 2021, Lilly announced that it would lower the list price of all Lilly’s non-branded insulins, including Insulin Lispro Injection, by an additional 40% in the U.S. That price reduction took effect January 1, 2022, effectively reducing the list price to 2008 levels. These examples, among others, demonstrate Lilly’s commitment to providing effective oversight over drug pricing and access.
Lilly already publicly discloses the information requested by the proposal regarding its oversight and remediation.plans to mitigate alleged risks presented by any misaligned lobbying activities.
In this proxy statement and the Political Participation Website, Lilly already discloses its governance and risk mitigation procedures that are in place to avoid and address any misalignment between Lilly’s lobbying activities and its public policy positions and statements.
For example, the board exercises oversight of Lilly’s political expenditures and lobbying activities to ensure the company fulfills its commitment to stewardship of corporate funds and risk minimization with respect to such activities. The third laboratory self-reportedboard receives regular updates from the incidentssenior vice president, corporate affairs, and took immediate actioncommunications regarding public policy issues and the company’s direct and indirect lobbying activities and receives semi-annual updates on direct and indirect lobbying activities and trade association memberships.
Therefore, the board already takes steps to addressoversee and mitigate the cited issues. potential risks of misalignment between Lilly’s lobbying activities and public policy positions and statements.
Requiring Lilly to prepare a separate report with this information would place an undue administrative burden on the company and would not provide meaningful additional information to shareholders.
We do not condone, in any form,believe the mistreatment of research animals, andadditional disclosures contemplated by the proposal would provide meaningful additional information to shareholders to merit the resources required to provide the requested report. For these reasons, we recognize our fundamental ethical and scientific obligation to ensurebelieve the appropriate treatment of animals used in research. We have processes and procedures in place to ensure humane treatment of animals, including programs for oversight by an internal corporate Animal Welfare Board, Institutional Animal Care and Use Committees, or an equivalent ethical review board, as well as veterinary oversight at every site—both ours and contract laboratories. We are committed to quality research-animal care and use, the responsible use of animals in medical research, and the use of alternative methods whenever possible and appropriate.
We adhere to standards set forthproposal is not in the U.S. Animal Welfare Act. We have been accredited bybest interests of the Association for Assessmentcompany and Accreditation of Laboratory Animal Care (AAALAC). AAALAC accreditation rules and standards can be found on the AAALAC website (www.aaalac.org). This accreditation is a voluntary process that includes a detailed, comprehensive review of our research-animal program including animal care and use policies and procedures, animal environment, housing and management, veterinary medical care, and physical plant operations. We consider our policies and practices to be very much in line with leading industry standards as evidenced by our engagement in industry consortia and professional societies focused on the use of animals in biomedical research (National Association of Biomedical Research, IQ Consortium, Foundation of Biomedical Research). We currently
publish information detailing our commitment to responsible animal research as well as an overview of our policies and procedures on our website (www.lilly.com).
For safe and effective medicines to be available to patients, U.S. and foreign regulatory agencies have mandated that a defined amount of research be performed in animals. Where animals must be used, we take every measure to assure that the lowest number of animals is used and that discomfort and distress are either eliminated or minimized.
As a global company, we develop contractual relationships with select laboratory-animal research and animal-supply companies inside and outside the U.S. We seek to do business only with those companies that share our commitment to animal welfare. We require these companies to maintain a quality animal care and use program. To ensure animal welfare, we assess third-party organization adherence to these expectations. If events suggest a laboratory has failed to meet our standards, we promptly investigate and act upon the allegations. These actions may include termination of a business relationship.
its shareholders.
Board Recommendation on Item 9
The Board of Directorsboard recommends athat you vote AGAINST thethis proposal.
Item 10. Shareholder Proposal Requestingto Report on the Extent to WhichOversight of Risks Related to Public Concern Over DrugAnticompetitive Pricing Strategies are Integrated into Incentive Compensation Arrangements
Mercy Investment Services, Inc., 2039 North Geyer Road, St. Louis, Missouri, Trinity Health, 766 Brady Avenue, Apt. #635, Bronx, New York 10462, beneficial owner of 73 shares of our common stock having a market value in excess of Eli Lilly and Company,$2000, has submitted the following proposal:
RESOLVED, that shareholders of Eli Lilly and Company (“Eli Lilly”) urgeask the Compensation Committee (the “Committee”)board of directors to report annually to shareholders on the extent to whichhow it oversees risks related to anticompetitive practices, including whether the full board or board committee has oversight responsibility, whether and how consideration of such risks is incorporated into board deliberations regarding strategy, and the board’s role in Eli Lilly’ s public concern over drug pricing strategies are integrated into Eli Lilly’s incentive compensation policies, plans and programs (together, “arrangements”) for senior executives.policy activities related to such risks. The report should include, but need not be limited to, discussionprepared at reasonable expense and should omit confidential or proprietary information, as well as information about existing litigation and claims of whether incentive compensation arrangements reward, or not penalize, senior executives for (i) adopting pricing strategies, or making and honoring commitments about pricing, that incorporate public concern regarding the level or rate of increase in prescription drug prices; and (ii) considering risks related to drug pricing when allocating capital.which Eli Lilly has notice.
SUPPORTING STATEMENT
As long-termThe anticompetitive practices of companies within the pharmaceutical supply chain, including insulin manufacturers such as Eli Lilly, are receiving increasing scrutiny from the public, regulators, and enforcers. The criticism of Eli Lilly has focused on the company’s insulin pricing strategy, which has resulted in massive price hikes for everyday consumers.1
In response, regulators and legislators have increasingly focused on the pricing strategies of insulin manufacturers. In early 2021, the Senate Finance Committee issued a Staff Report on the rising cost of insulin, noting that Eli Lilly’s Humalog 50-50 Kwikpen had seen a 64% price increase between 2013 and 2017, and that insulin manufacturers had “aggressively raised the [wholesale acquisition cost] of their insulin products absent significant advances in the efficacy of the drugs.”2 Additionally, in response to high insulin prices, eight states (CO, IL, ME, NM, NY, UT, WA, WV) have capped the price of insulin within their jurisdiction and others are considering adopting similar policies.3
Separately, the company is facing a multitude of lawsuits around the pricing of its insulin products, including claims from various state attorney generals, class action lawsuits, and other market participants.4 The allegations include claims that Eli Lilly violated both state and federal RICO statutes, was unjustly enriched, and violated various state level consumer protection laws.5 The number and magnitude of lawsuits continues to mount, with the Mississippi Attorney General recently filing a lawsuit against Eli Lilly, alleging that the company colluded with other insulin manufacturers and pharmacy benefit managers to artificially keep insulin prices high:6
Eli Lilly’s pricing strategies and the resulting mounting pressure on the company increase the likelihood of new regulation, increase risk for investors, and have a substantial impact on the public at large. Given the widespread concern and rapidly changing environment the company finds itself in, we believe that senior executive incentive compensation arrangements should rewardrobust board oversight would improve Eli Lilly’s management of the creation of sustainable long-term value. To that end, it is important that those arrangements align with company strategy and encourage responsible risk management.
A key risk facing pharmaceutical companies is potential backlash against high drug prices. Public outrage over high prices and their impact on patient access may force price rollbacks and harm corporate reputation. Legislative or regulatory investigations regarding pricing of prescription medicines may bring about broader changes, with some favoring allowing Medicarerisks related to bargain over drug prices. (E.g., https://democrats-oversight.house.gov/news/press-releases/cummings-and-welch-launch-investigation-of-drug-companies-skyrocketing-prices; https://democrats-oversight.house.gov/news/press-releases/cummings-and-welch-propose-medicare-drug-negotiation-bill-in-meeting-with) An October 2017 report indicated that five states and federal prosecutors are investigating insulin makers, including Eli Lilly, forits anticompetitive practices related to pricing. (https://medcitynews.com/2017/10/insulin-prices-soar/)and that shareholders would benefit from more information about the board’s role.
We applaudTherefore, we urge Eli Lilly for improving transparency on drug pricing and supporting alternative pricing approaches. We are concerned, however, that the incentive compensation arrangements applicable to Eli Lilly’s senior executives may not encourage senior executives to take actions that result in lower short-term financial performance even when those actions may be in Eli Lilly’s best long-term financial interests.
Eli Lilly uses revenue and earnings per share (EPS) as metrics for the annual bonus and EPS growth as the metric for performance awards. (2017 Proxy Statement, at 41-42) A recent Credit Suisse analyst report stated that “US drug price rises contributed 100% of industry EPS growth in 2016” and characterized that fact as “the most important issue for a Pharma investor today.” The report identified Eli Lilly as a company where price increases accounted for at least 100% of EPS growth in 2016. (Global Pharma and Biotech Sector Review: Exploring Future US Pricing Pressure, Apr. 18, 2017, at 1)
In our view, excessive dependence on drug price increases is a risky and unsustainable strategy, especially when price hikes drive large senior executive payouts. For example, media coverage of the skyrocketing cost of Mylan’s EpiPen noted that a 600% rise in Mylan’s CEO’s total compensation accompanied the 400% EpiPen price increase. (See, e.g., https://www.nbcnews.com/business/consumer/mylan-execs-gave-themselves-raises-they-hiked-epipen-prices-n636591; https://www.wsj.com/articles/epipen-maker-dispenses-outsize-pay-1473786288; https://www.marketwatch.com/story/mylan-top-executive-pay-was-second-highest-in-industry-just-as-company-raised-epipen-prices-2016-09-13)
The disclosure we request would allow shareholders to better assess the extent to which compensation arrangements encourage senior executives to responsibly manage risks relating to drug pricing and contribute to long-term value creation. We urge shareholders to vote forFOR this Proposal.proposal.
Statement in Opposition to the Shareholder Proposal Requesting to Report on the Extent to WhichOversight of Risks Related to Public Concern Over DrugAnticompetitive Pricing Strategies are Integrated into Incentive Compensation Arrangements
The Public Policyboard, after review by its Directors and Corporate Governance Committee, recommends a vote against this proposal.
The proposal calls for the board to produce a report on how it oversees risks from alleged anticompetitive practices. The supporting statement makes reference to, among other things, competition-related lawsuits and regulatory investigations that relate to the pricing of insulin from several years ago.
1https://www.finance.senate.gov/imo/media/doc/Grassley-Wyden%20lnsulin%20Report%20(FINAL%201).pdf.
2 Id.
3https://www.nbcnews.com/news/us-news/states-are-trying-cap-price-insulin-pharmaceutical-companies-arepushing-n1236766
4See Eli Lilly and Company 2020 Form 10-K, at 108-109 (https://investor.lilly.com/static-files/e724cf2c-3a2e-4180-beaf-944ecd588323).
5Id.
6https://seekingalpha.com/news/3706905-eli-lilly-sanofi-and-novo-nordisk-sued-over-insulin-prices
The proposal is unnecessary because Lilly already provides shareholders information that can be used to assess the board’s oversight over risks from alleged anticompetitive practices, including relating to the pricing of its medicines. This proxy statement and the company’s environmental, social and governance (“ESG”) page of its website include extensive disclosures describing how the board actively oversees and approves Lilly’s strategy, approves significant management initiatives, and structures its board and board committee agendas to engage directors in informed reviews of strategic and forward-looking issues, and Lilly’s independent directors are deeply engaged in overseeing Lilly’s approach to drug pricing and access.
These materials disclose that under the active oversight from the board and its committees, Lilly devotes substantial time and resources to implementing industry-leading initiatives in furtherance of insulin affordability. While the proposal attempts to articulate alleged anticompetitive risks by relying on “wholesale acquisition cost” data from 2017, the fact is that through numerous affordability solutions, combined with insurance coverage, Lilly has lowered the average monthly out-of-pocket cost for a prescription of Lilly insulin (regardless of the number of vials or pens) by 42% since 2017, to $22.59. Today, with various affordability options now available, anyone is eligible to purchase their monthly prescription of Lilly insulin for $35, whether they are uninsured, use commercial insurance, or are enrolled in a participating Medicare Part D plan.
In the 2021 10-K, this proxy statement and board committee charters, we already publicly disclose risks regarding regulatory and pricing strategy and the mechanisms by which the board and its committees actively oversee such risks.
In our 2021 10-K, we already disclose that there continues to be considerable public and government scrutiny of pharmaceutical pricing, and that measures to address the perceived high cost of pharmaceuticals are being considered at
various levels of state and federal government. Our risk factors address drug pricing risk, noting that public and private payers continue to take steps to control expenditures for pharmaceuticals by placing restrictions on pricing and reimbursement for, and patient access to, our medications.
We already disclose to shareholders the mechanisms by which the board oversees such risks in this proxy statement and in our committee charters, which we make publicly available on our website. This proxy statement includes extensive information about how our independent directors are deeply engaged in key matters important to Lilly and our stakeholders, including oversight over the company’s approach to drug pricing and access. For example, the full board oversees the state of our compliance program and reviews key enterprise-level risks, and the Audit Committee oversees enterprise risk management processes and procedures.
The Audit Committee charter charges the Audit Committee with monitoring legal and regulatory requirements, Lilly’s compliance with legal and regulatory requirements and processes and procedures to identify and mitigate enterprise-level risks. The Ethics and Compliance Committee charter charges the Ethics and Compliance Committee with reviewing, identifying and, when appropriate, bringing to the board’s attention legal and regulatory trends and issues. The Ethics and Compliance Committee meets at least four times a year, including semi-annual private sessions with the company’s chief ethics and compliance officer, general auditor, and senior vice president, global quality. On an annual basis, the full board reviews the company’s overall state of compliance and the Ethics and Compliance Committee receives an update on compliance at each meeting. In addition, the Audit Committee and the Ethics and Compliance Committee meet jointly at least annually to review significant legal or regulatory compliance exposure and material reports or inquiries from regulators. Guided by this active oversight and as described in the next section, Lilly provides sufficient information to shareholders to assess that the company has already taken numerous industry-leading initiatives to promote drug affordability and access.
In this proxy statement and on the ESG page of our website, we already disclose how the board incorporates pricing decisions into its strategic considerations designed in furtherance of medicine accessibility.
This proxy statement discloses that the board actively oversees our corporate strategy and provides that each year the board and executive management closely examine the company’s strategy, including key risks and decisions facing the company.
The ESG page of our website describes that Lilly aims to strike a balance between access and affordability for patients while sustaining investments in life-changing treatments for some of today’s most serious diseases. When making pricing decisions, we use a value-based approach, taking into account the following:
•Customer perspective – The unmet needs that medicines can fulfill for patients and caregivers and how people can affordably access the treatment;
•Company considerations – The costs of research, development, manufacturing and support services for customers; business trends and other economic factors; as well as the medicine’s potential market size, patent life and place within our larger portfolio of medicines;
•Competitive landscape –The benefits of our medicine compared to alternative medicines, where our medicine fits in treating conditions and existing contracts between payers and our competitors;
•Other external factors – Such as health system changes and policy guidelines.
As described in this proxy statement, under the active oversight of the board, we have taken substantial actions to address drug pricing and access concerns. For example, Lilly introduced two additional lower-priced versions of Humalog in January
2020 and added the Lilly Insulin Value Program to Lilly’s comprehensive suite of insulin affordability solutions in September 2020, which now enables customers with commercial insurance or no insurance to purchase their monthly prescription of all Lilly insulins for $35. In addition, in September 2021, Lilly announced that it would lower the list price of all Lilly’s non-branded insulins, including Insulin Lispro Injection, by an additional 40% in the U.S. That price reduction took effect January 1, 2022, effectively reducing the list price to 2008 levels. These examples, among others, demonstrate Lilly’s commitment to providing effective oversight over drug pricing and access.
In addition, for the last five years Lilly has reviewedpublished on the company’s website U.S. list price increases and net price decreases for certain Lilly products, as well as specific additional data for Humalog and Insulin Lispro. Our board approved this proposalmeasure five years ago and recommends against it.
Lilly was among the first companies to provide this information. In addition to other actions that actually lower the price of our drugs, such as those referenced above, we routinely disclose our drug price changes, in some cases several months in advance, to stakeholders and multiple state and federal agencies.
The company’s annualboard’s role in public policy assessments is disclosed on the Political Participation Website, stating our approach to overseeing current and emerging environmental, social, political, and governance trends and public policy issues that may affect business operations, including overseeing political activities.
As noted in this proxy statement provides detailedand the Political Participation Website, as a biopharmaceutical company that develops treatments for serious diseases, we believe it is important for our company to be a responsible participant in global political and public policy debates. Our engagement in the political arena helps ensure that patients have access to needed medicines. Through public policy engagement, we provide a way for all our locations globally to offer Lilly’s perspective on the political environment in a manner that supports access to innovative medicines and a way to engage on issues specific to local business environments.
The Political Participation Website describes that the board exercises governance oversight of our political expenditures and lobbying activities ensuring our commitment to stewardship of corporate funds and risk minimization with respect to such activities. As discussed in the Political Participation Website, the board receives regular updates from our senior vice president, corporate affairs and communications regarding public policy issues and the company’s direct and indirect lobbying activities and receives semi-annual updates on political engagement, including information on the contributions made by our exclusively employee-led political action committee (LillyPAC) and the company, as well as trade association memberships. Lilly’s vice president, U.S. government affairs reviews and approves all corporate political contributions to ensure these contributions are consistent with the company’s policies, plans,guidelines and practices relatingin accordance with applicable laws. Our general counsel and our chief financial officer, or their designees, also approve all corporate political contributions before they are made.
Therefore, the board already takes steps to executive compensation. Each year,oversee and mitigate the CD&A sectionpotential risks of public policy issues that may affect our business operations, including overseeing political activities.
Requiring Lilly to prepare a separate report with this document describes our executive compensation philosophy,information would place an undue administrative burden on the Board of Director’s Compensation Committee's process for setting executive compensation,company and would not provide meaningful additional information to shareholders.
We do not believe the elements of our compensation program,additional disclosures contemplated by the factorsproposal would provide meaningful additional information to shareholders to merit the committee considered when setting executive compensation forresources required to provide the previous year, and howrequested report. For these reasons, we believe the company's results affected incentive payouts forproposal is not in the previous year’s performance.
The proxy statement includes a detailed summarybest interests of the Compensation Committee’s review of individual named executive officers. These summaries provide all relevant information regarding the factors considered in those executive’s compensation. These summaries include information regarding specific individualized performance inputs, including inputs relating to strategic effortscompany and decisions. This is information is broadly available to shareholders and the general public.
Given that information relating to executive compensation programs, plans, and practices is already disclosed as part of the proxy statement, we believe an annual report is unnecessary.
its shareholders.
Board Recommendation on Item 10
The Board of Directorsboard recommends athat you vote AGAINST thethis proposal.
Other Information
Meeting and Voting Logistics
Additional Items of Business
We do not expect any items of business to be submitted to shareholders at the Annual Meeting other than those above because the deadline for shareholder proposals and nominations has passed.referred to in this proxy statement. Nonetheless, if necessary, the accompanying proxy gives discretionary authority to the persons named on the proxy have discretionary authority to vote the shares represented thereby with respect to any other matters that might be brought before the meeting. Those persons intend to vote that proxyon any such matters in accordance with their best judgment.
Voting
Shareholders as of the close of business on March 12, 2018February 22, 2022 (the record date) may vote or have their shares voted at the annual meeting.Annual Meeting. You have one vote for each share of common stock you held on the record date, including shares:
•held directly in your name as the shareholder of recordrecord;
•held for you in an account with a broker, bank, or other nomineenominee; and
•attributed to your account in the company's 401(k) plan.Plan.
You may vote your shares in person at the meeting. However, weWe encourage you to vote by mail, by telephone, or online even if you plan to attend the meeting.Annual Meeting. Shareholders who hold their shares in the 401(k) Plan must vote by April 27, 2022 so the plan trustee can vote their shares accordingly. See
"—Voting Shares Held in the Company 401(k) Plan" for more information.
Required Vote
Below are the vote requirements for the various proposals:
•The fivethree nominees for director will be elected if the votes cast for the nominee exceed the votes cast against the nominee. Abstentions and broker non-votes will not count as votes cast either for or against a nominee.
•The following items of business will be approved if the votes cast for the proposal exceed thosethe votes cast against the proposal:
•an advisory approval of compensation paid to the named executive compensationofficers presented in this proxy statement;
•ratification of the appointment of principalthe independent auditorauditor;
approve •the amended proposal to amend the company’s articles of incorporation to give shareholders the ability to amend the bylaws;and restated 2002 Lilly stock plan
•four shareholder proposals.
Abstentions and broker non-votes will not be counted as votes cast either for or against these proposals. As discussed below in "Meeting and Voting Logistics—Voting Shares Held by a Broker," broker non-votes are not expected in connection with the ratification of the appointment of the independent auditor.
The proposals to amend the articles of incorporation for declassification ofto eliminate the classified board structure and to eliminate supermajority voting provisions require the vote of 80 percent of the outstanding shares.shares of our common stock. For these items, abstentions and broker nonvotesnon-votes have the same effect as a vote against the proposals.
Quorum
A majority of the outstanding shares entitled to vote, present or represented by proxy, constitutes a quorum for the annual meeting.Annual Meeting. As of the record date, XXXXXXFebruary 22, 2022, 952,347,126 shares of company common stock were issued and outstanding.
Voting By Proxyby Shareholders of Record
If you are a shareholder of record, you may vote your proxy by any one of the following methods:
| | | | | |
8 | OnlineOnline. . You may vote online at www.proxyvote.com.ProxyVote.com. Follow the instructions on your proxy card or notice. If you received these materials electronically, follow the instructions in the e-mailemail message that notified you of their availability. Voting online has the same effect as voting by mail. If you vote online, do not return your proxy card.
|
| |
) | By telephonetelephone. . Shareholders in the U.S., Puerto Rico,Call 1-800-690-6903 using a touch-tone phone and Canada may vote by telephone by followingfollow the instructions on your proxy card or notice.provided. |
| By mail. If you received these materials electronically, follow the instructions in the e-mail message that notified youor requested paper copies of their availability. Voting by telephone has the same effect as voting by mail. If you vote by telephone, do not return your proxy card. |
| |
* | By mail. Signmaterials, sign, date, and datereturn each proxy card you receive and return it in the prepaid envelope. Sign your name exactly as it appears on the proxy.appears. If you are signing in a representative capacity (for example, as an attorney-in-fact, executor, administrator, guardian, trustee, or the officer or agent of a corporation or partnership), please indicate your name and your title or capacity. If the stock is held in custody for a minor (for example, under the Uniform Transfers to Minors Act), the custodian should sign, not the minor. If the stock is held in joint ownership, one owner may sign on behalf of all owners. If you return your signed proxy but do not indicate your voting preferences, wethe proxy holder will vote on your behalf withbased upon the board’s recommendations.
|
You may vote your shares prior to the Annual Meeting until 11:59 p.m. EDT on May 1, 2022 online or by telephone. If you are voting by mail, your marked, signed, and dated proxy card must be received by May 1, 2022. Shareholders of record may also opt to vote at the Annual Meeting, which will be held online via live webcast at virtualshareholdermeeting.com/LLY2022. See "—Attending the Annual Meeting" for more information on attending the Annual Meeting.
You have the right to change your vote or revoke your proxy before it is voted at any time before the meetingAnnual Meeting by (i) timely notifying the company’s secretaryGeneral Counsel and Secretary in writing, or (ii) timely delivering a later-dated proxy online, by mail, or (iii) timely casting a new vote online or by telephone. If you are a shareholderShareholders of record you may also revoke your proxytheir proxies by voting in person at the meeting.Annual Meeting.
Voting Shares Held Byby a Broker
If your shares are held by a broker, the broker will ask you how you want your shares to be voted. You may instruct your broker or other nominee to vote your shares by following instructions that the broker or nominee provides to
you. Most brokers offer voting by mail, by telephone, and online.
You may submit new voting instructions by contacting your broker or other nominee or by voting at the Annual Meeting.
If you give the broker instructions, your shares will be voted as you direct. If you do not giveprovide voting instructions, oneyour shares will not be voted on any proposal on which the broker does not have discretionary authority to vote. This is called a "broker non-vote." In these cases, the broker can register your shares as being present at the Annual Meeting for purposes of two things can happen, dependingdetermining the presence of a quorum but will not be able to vote on the typethose matters for which specific authorization is required under NYSE rules. If you are a beneficial owner whose shares are held of proposal. Forrecord by a broker, your broker has discretionary voting authority under NYSE rules to vote your shares on the ratification of EY as the principal independent auditor for 2022, even if the broker maydoes not receive voting instructions from you. However, your broker does not have discretionary authority to vote on the election of directors, the advisory approval of executive compensation, or the shareholder or management proposals without instructions from you, in which case a broker non-vote will occur, and your shares in its discretion. For all other proposals, the broker maywill not vote your shares at all.be voted on these matters.
Voting Shares Held in the Company 401(k) Plan
You may instruct the plan trustee on how to vote your shares in the 401(k) planPlan online, by mail, or by telephone as described above in "Meeting and Voting Logistics—Voting by Shareholders of Record," except that if you vote by mail, the card that you use will be a voting instruction form rather than a proxy card.
In addition, unless you decline, your vote will apply to a proportionate number of other shares held by participants in the 401(k) planPlan for which voting directions are not received (except for a small number of shares from a prior stock ownership plan, which can be voted only on the directions of the participants to whose accounts the shares are credited).
All participants are named fiduciaries under the terms of the 401(k) planPlan and under the Employee Retirement Income Security Act (ERISA) for the limited purpose of voting shares credited to their accounts and the portion of undirected shares to which their vote applies. Under ERISA, fiduciaries are required to act prudently in making voting decisions.
If you do not want to have your vote applied to the undirected shares, you must so indicate when you vote. Otherwise, the trustee will automatically apply your voting preferences to the undirected shares proportionally with all other participants who elected to have their votes applied in this manner.
If you do not vote online or by telephone by 11:59 p.m. EDT on April 27, 2022, or if your mailed ballot is not received by April 27, 2022, your shares will be voted byin accordance with instructions received from other plan participants who have elected to have their voting preferences applied proportionally to all shares for which voting instructions are not otherwise received.
You will not be able to vote your shares personally at the Annual Meeting.
Multiple Notices, Proxy Cards and NoticesMaterials, or Emails
If you received more than one notice, full set of proxy card, notice,materials, or e-mailemail related to proxy materials, you hold shares in more than one account. To ensure that all your shares are voted, sign and return each card. Alternatively, if you vote by telephone or online, youYou will need to cast a vote once for each notice, full set of proxy card, notice,materials, or e-mailemail you receive. If you do not receive a proxy card, you may have elected to receive your proxy statement electronically, in which case you should have received an e-mailemail with directions on how to access thethis proxy statement and how to vote your shares. If you wish to request a paper copy of these materials and a proxy card, please call 800-579-1639.1-800-579-1639 on or before April 18, 2022 to facilitate timely delivery.
Vote Tabulation
Votes are tabulated by an independent inspector of election, Broadridge Financial Solutions, Inc.
Attending the Annual Meeting
The Annual Meeting will be held on Monday, May 2, 2022, at 11:00 a.m. EDT, and all shareholders as of close of business on February 22, 2022 are entitled to participate.
The Annual Meeting will be held virtually via live webcast.
Although you will not be able to attend the Annual Meeting at a physical location, we have designed the Annual Meeting live webcast to provide shareholders the opportunity to participate virtually to facilitate shareholder attendance and provide a consistent experience to all shareholders, regardless of location.
The live webcast of the Annual Meeting can be accessed by shareholders on the day of the meeting at virtualshareholdermeeting.com/LLY2022 and will begin promptly at 11:00 a.m. EDT. To attend the Annual Meeting, you will need to log in to virtualshareholdermeeting.com/LLY2022 using the 16-digit control number found on the proxy card, voting instruction form, or notice you previously received. This website can be accessed on a computer, tablet, or phone with internet connection. Online access to the webcast will open 15 minutes prior to the start of the Annual Meeting to allow time to log in and test your device’s audio system. We encourage you to access the meeting in advance of the designated start time.
To submit questions in advance of the Annual Meeting, visit ProxyVote.com before May 2, 2022 and enter your 16-digit control number. During the meeting, if you wish to submit a question, log into the virtual meeting website atvirtualshareholdermeeting.com/LLY2022, click on "Q&A," type your question into the "Submit a Question" field, and click "Submit." In order to provide an opportunity to as many shareholders as possible who wish to ask a question, each shareholder will be limited to one question. Shareholders may ask a second question if all other shareholders have had an opportunity to ask a question and if time allows. The Annual Meeting is scheduled to begin at 11:00 a.m. EDT and end at 11:45 a.m. EDT, and time remaining after agenda items are addressed will be available for shareholder questions. We will endeavor to answer as many questions submitted by shareholders as time permits. We reserve the right to edit profanity or other inappropriate language and to exclude questions regarding topics that are not pertinent to meeting matters or company business. If we receive substantially similar questions, we may group such questions together and provide a single response to avoid repetition. Responses to questions relevant to meeting matters that we do not have time to respond to during the meeting will be limitedposted to shareholders of record, those holding proxies from shareholders of record, and invited guests from the media and financial community. All shareholders of record as of the record date may attend by presenting the admission ticket that appears at the end of this proxy statement. Please fill it out and bring it with you toour website following the meeting. TheQuestions regarding topics that are not pertinent to meeting matters or company business will not be held at the Lilly Center Auditorium. Please use the Lilly Center entrance to the south of the fountain at the intersection of Delaware and McCarty streets. You will need to pass through security, including a metal detector. Present your ticket to an usher at the meeting.answered.
ParkingSupport staff will be available on a first-come, first-served basisshould you experience any technical difficulties in accessing the garage indicated onvirtual meeting. Instructions for requesting technical assistance will be available atvirtualshareholdermeeting.com/LLY2022.
List of Shareholders of Record
A list of the mapnames of shareholders entitled to vote at the end of this report. If you have questions about admittance or parking, please call 855-731-6026 (toll free) or 317-433-5112 (priorAnnual Meeting will be available to shareholders for five business days prior to the annual meeting).
The 2019 Annual Meeting for any purpose germane to the Annual Meeting. Please contact us at shareholderproposals@lilly.com if you wish to examine the list prior to the Annual Meeting. The shareholder list will also be available during the virtual Annual Meeting for examination by shareholders who access the Annual Meeting using their 16-digit control number at virtualshareholdermeeting.com/LLY2022.
The 2023 Annual Meeting
The company’s 20192023 annual meeting of shareholders is currently scheduled for May 6, 2019.1, 2023.
Other Matters
Notice and AccessWe distribute proxy materials to many shareholders via the internet under the SEC’s "Notice and Access" rules to reduce production and mailing costs and to help preserve environmental resources. Using this method of distribution, on or about March 18, 2022, we mailed the Notice Regarding the Availability of Proxy Materials that contains basic information about the Annual Meeting and instructions on how to view all proxy materials and vote. If you receive the notice and prefer to receive proxy materials by regular mail or email, follow the instructions in the notice for making this request, and the materials will be sent promptly to you via the preferred method. If you prefer to vote by phone rather than online, the website listed on the notice (ProxyVote.com) has instructions for voting by phone.
Householding
We have adopted a procedure approved by the SEC called "householding." Under the householding procedure, certain shareholders, whether they own registered shares or shares in street name, who have the same address and who receive either notices or paper copies of the proxy materials in the mail will receive only one setcopy of our proxy materials, or a single notice, for all shareholders at that address, unless one or more of the shareholders at that address has previously notified us that they want to receive separate copies. Each 401(k) Plan participant will continue to receive a copy of all of the proxy materials. Regardless of how you own your shares, if you received a single set of proxy materials as a result of householding, and one or more shareholders at your address would like to have separate copies of these materials with respect to the 2018 annual meetingAnnual Meeting or in the future, or if you would like to request that only a single set of proxy materials be sent to the household, please contact Broadridge Financial Solutions, Inc., at 866-540-7095.1-866-540-7095 or 51 Mercedes Way, Edgewood, NY 11717.
Other information regardingInformation Regarding the company’s proxy solicitationCompany’s Proxy Solicitation
The board of directors is soliciting proxies for the 2018 annual meeting.Annual Meeting. We will pay all expenses in connection with our solicitation of proxies. We will pay brokers, nominees, fiduciaries, or other custodians their reasonable expenses for sending proxy material to and obtaining instructions from persons for whom they hold stock of the company. We expect to solicit proxies primarily by mail and email, but directors, officers, and other employees of the company may also solicit in person or by telephone, fax, or email. We have retained Georgeson LLC to assist in the distribution and solicitation of proxies. Georgeson may solicit proxies by personal interview, telephone, fax, mail, and email. We expect that the fee for those services will not exceed $17,500 plus reimbursement of customary out-of-pocket expenses.
Corporate Governance Materials
Section 16(a) beneficial ownership reporting compliance
Under SEC rules,The company’s main corporate website address is lilly.com. We also make available through our directors and executive officers are required to fileinvestor relations website, free of charge, our company filings with the SEC reports of holdings and changes in beneficial ownership of company stock. We have reviewed copies of reports providedas soon as reasonably practicable after we electronically file them with, or furnish them to, the company,SEC. The reports we make available include annual reports on Form 10-K, quarterly reports on Form
10-Q, current reports on Form 8-K, proxy statements, registration statements, and any amendments to those documents. The website link to our SEC filings is investor.lilly.com/sec.cfm. This proxy statement and the annual report to shareholders are also available at ProxyVote.com as well as other recordson our website at lilly.com/policies-reports/annual-report, and information. Basedthe articles of incorporation, bylaws, and all committee charters are available online at lilly.com/leadership/governance. Certain documents and information referenced in this proxy statement are available on our website. However, except as otherwise expressly provided in this proxy statement, we are not including the information contained on our website, or any information that review, we concluded that all reports were timely filed, except that, due to administrative errors, Dr. Carolyn Bertozzi was late in filing Form 3, due to absencemay be accessed by links on our website, as part of, SEC Edgar access codes; Christi Shaw was late in filing a Form 4 to report a restricted stock unit grant; and Maria Crowe was late in filing a Form 4 to report a 2016 charitable gift contribution. Each filing was made promptly after the issue was discovered.or incorporating it by reference into, this proxy statement.
By order of the Board of Directors,
Ms. Anat Hakim
Bronwen L. Mantlo
Senior Vice President, General Counsel and Secretary
March 19, 201818, 2022
Appendix A - Summary of Adjustments Related to the Annual Cash Bonus and Performance Award
Consistent with past practice, the Compensation Committee adjusted the reported financial results on which the 20172021 annual cash bonus and the 2016-20182020-2022 performance awards were determined to eliminate the distorting effect of certain unusual items on incentive compensation performance measures. The adjustments are intended to:
•align award payments with the underlying performance of the core business
•avoid volatile, artificial inflation or deflation of awards due to unusual items during any year in the award year,performance period, and, where relevant, the previous (comparator) year
•eliminate certain counterproductive short-term incentives—for example, incentives to refrain from acquiring new technologies, to defer disposing of underutilized assets, or to defer settling legacy legal proceedings to protect current bonus payments
•facilitate comparisons with peer companies.
To ensure the integrity of the adjustments, the Compensation Committee establishes adjustment guidelines in the first 90 days of the performance period. These guidelines are generally consistent with the company guidelines for reporting non-GAAP financial measures to the investment community, which are reviewed by the Audit Committee. The adjustments apply equally to income and expense items. The Compensation Committee reviews all adjustments and retains downward discretion, i.e., discretion to reduce compensation below the amounts that are yielded by the adjustment guidelines.
Adjustments for 20172021 Bonus Plan
For 20172021 bonus calculations, the Compensation Committee made the following adjustments to reported EPS consistent with our external reporting of non-GAAP financial measures:
•Eliminated the impact of the charges recognized for acquired in-process research and development
•Eliminated the impact of amortization of intangible assets
•Eliminated the impact of a charge related to U.S. tax reform legislationthe repurchase of higher-cost debt
•Eliminated the impact of asset impairments, restructuring, and other special charges
•Eliminated the impact of the chargeCOVID-19 antibodies inventory charges that were recognized for acquired in-process researchprimarily due to changes in forecasted demand from U.S. and developmentinternational governments
•Eliminated the impact of amortizationnet gains on investments in equity securities
In addition to the adjustments consistent with our reporting of non-GAAP financial measures, the Compensation Committee made the following adjustment:
•When the Compensation Committee set 2021 bonus targets, the revenue and EPS goal did not contemplate estimated savings from certain intangible assetsdiscrete and unplanned performance items. The Compensation Committee reduced revenue and non-GAAP EPS for the purposes of the bonus calculation to exclude the savings from these items.
Eliminated the impactA reconciliation of inventory step-up costs associated with the acquisitionadjustments to our reported revenue is below:
| | | | | |
| 2021 |
| Revenue as reported | $28,318.4 |
| Adjustment for estimated savings from certain discrete and unplanned performance items | (180.8) | |
| Adjusted revenue | $28,137.6 |
A reconciliation of Boehringer Ingelheim Vetmedica’s U.S. feline, canine, and rabies vaccine portfolio.
Reconciliations of these adjustments to our reported EPS areis below:
| | | | | |
| |
| 20172021 |
| EPS as reported | $(0.19)6.12 |
Eliminate U.S. tax reform legislation charge
Acquired in-process research and development charges | $1.810.77 |
Eliminate assetAmortization of intangible assets
| 0.53 |
| Charge related to repurchase of higher-cost debt | 0.35 |
| Asset impairments, restructuring, and other special charges
| $1.230.28 |
Eliminate acquired in-process research and development chargeCOVID-19 antibodies inventory charges | $0.970.25 |
Eliminate amortization of certain intangible assetsNet gains on investments in equity securities | $0.44 |
Eliminate inventory step-up costs associated with the acquisition of
Boehringer Ingelheim Vetmedica’s U.S. feline, canine, and rabies vaccine portfolio
| $0.03(0.16) |
| Non-GAAP EPS | $4.288.16 |
| Adjustment for estimated savings from certain discrete and unplanned performance items | (0.16) |
| Adjusted Non-GAAP EPS | $8.00 |
*Numbers may not add due to rounding
Adjustments for 2016-20182020-2022 Performance Award
For the 2016-20182020-2022 performance award payout calculations, the Compensation Committee made the following adjustments to reported EPS consistent with our reporting of non-GAAP financial measures:
2017:•2019: Eliminated Elanco discontinued operations
•2021, 2020, and 2019: Eliminated the impact of the charge related to U.S. tax reform legislation
2017: Eliminated the impact of inventory step-up costs associated with the acquisition of Boehringer Ingelheim Vetmedica’s U.S. feline, canine, and rabies vaccine portfolio
2017, 2016 and 2015: Eliminated the impact of the charges recognized for acquired in-process research and development
2017, 2016•2021, 2020, and 2015:2019: Eliminated the impact of amortization of intangible assets
•2021 and 2019: Eliminated the impact of charges related to the repurchase of debt
•2021, 2020, and 2019: Eliminated the impact of asset impairments, restructuring, and other special charges
2017, 2016 and 2015:•2021: Eliminated the impact of amortizationCOVID-19 antibodies inventory charges that were recognized primarily due to changes in forecasted demand from U.S. and international governments
•2021, 2020, and 2019: Eliminated the impact of certain intangible assets.net gains on investments in equity securities
2016:•2019: Eliminated the impact of the Venezuelan financial crisisgain on sale of the China antibiotics business
2015:•2019: Eliminated the impact of the debt extinguishment lossLartruvo charges
2015:•2019: Eliminated the impact of inventory step-up for Novartis Animal Health.reduced shares outstanding from the Elanco exchange offer
In addition to the adjustments consistent with our reporting of non-GAAP financial measures, the Compensation Committee made the following other adjustments:
When the Compensation Committee set 2016-2018 performance award targets, the EPS goals were set assuming the transfer of the commercialization rights for Erbitux® in North America to Lilly (which occurred in October 2015). To make effective comparisons, the committee adjusted the base year 2015 results to include•2019: Eliminated the impact of the transfercertain income tax items
A reconciliation of the commercialization rights of Erbitux as if the transfer had occurred as of January 1, 2015.
Reconciliations of these adjustments to our reported EPS is below:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2021 | 2020 | % Growth
2021 vs. 2020 | 2019 | % Growth 2020 vs. 2019 |
| EPS as reported | $6.12 | | $6.79 | | (9.9) | % | | $8.89 | | (23.6) | % | |
| Elanco discontinued operations | — | | — | | | | (3.93) | | | |
| EPS as reported from continuing operations | 6.12 | | 6.79 | | | | 4.96 | | | |
| Acquired in-process research and development charges | 0.77 | | 0.64 | | | | 0.21 | | | |
| Amortization of intangible assets | 0.53 | | 0.36 | | | | 0.18 | | | |
| Charge related to repurchase of debt | 0.35 | | — | | | | 0.22 | | | |
| Asset impairment, restructuring, and other
special charges | 0.28 | | 0.14 | | | | 0.58 | | | |
| COVID-19 antibodies inventory charges | 0.25 | | — | | | | — | | | |
| Net gains on investments in equity securities(a) | (0.16) | | (1.15) | | | | (0.31) | | | |
| Gain on sale of China antibiotics business | — | | — | | | | (0.26) | | | |
| Lartruvo charges | — | | — | | | | 0.14 | | | |
| Impact of reduced shares outstanding for non-GAAP reporting(b) | — | | — | | | | 0.07 | | | |
| Impact of certain tax items(c) | — | | — | | | | (0.05) | | | |
| Non-GAAP EPS | $8.16 | | $6.78 | | 20.4 | % | | $5.73 | | 18.3 | % | |
*Numbers may not add due to rounding
(a) In 2021, the company began excluding gains and losses on investments in equity securities from non-GAAP measures. Prior to 2021, the Compensation Committee made a $0.98 adjustment to 2020 non-GAAP EPS to offset the impact of the net gains on investments in equity securities on EPS results in an amount that significantly exceeded the expected net gains on investments originally included in the EPS plan during 2020. For the 2020-2021 performance award, the Compensation Committee adjusted the 2019 base-year results to which the expected industry growth rates are below.applied to derive the two-year cumulative EPS goals and recast 2020 non-GAAP EPS to be consistent with the 2021 non-GAAP EPS treatment of gains and losses on investments in equity securities. Those adjusted 2020 and 2019 results are presented in this table for comparability.
(b)Non-GAAP EPS assume that the disposition of Elanco occurred at the beginning of 2019 and, therefore, exclude the approximately 65.0 million shares of Lilly common stock retired in the Elanco exchange offer.
(c) Amount relates to a tax benefit from a capital loss on the disposition of subsidiary stock.
|
| | | | | |
| | 2017 | 2016 | % Growth 2017 vs. 2016 | 2015 | % Growth 2016 vs. 2015 |
| EPS as reported | $(0.19) | $2.58 | NM | $2.26 | 14.2% |
Eliminate impact of U.S. tax reform legislation
| $1.81 | — | | — | |
Eliminate inventory step-up for Vetmedica
| $0.03 | — | | — | |
Eliminate impact of the Venezuelan financial crisis
| — | $0.19 | | — | |
Eliminate acquired in-process research and development charges
| $0.97 | $0.02 | | $0.33 | |
Eliminate asset impairments, restructuring and other special charges
| $1.23 | $0.29 | | $0.25 | |
Eliminate amortization of certain intangible assets
| $0.44 | $0.44 | | $0.39 | |
Eliminate debt extinguishment loss
| — | — | | $0.09 | |
Eliminate inventory step-up for Novartis Animal Health
| — | — | | $0.10 | |
| Non-GAAP EPS | $4.28 | $3.52 | 21.6% | $3.43 | 2.6% |
| Transfer of Erbitux commercialization rights adjustment | — | — | | $0.09 | |
| Adjusted Non-GAAP EPS | $4.28 | $3.52 | 26.6% | $3.52 | — |
| *Numbers may not add due to rounding | | | | | |
Appendix B - Proposed Amendments to the Company's Articles of Incorporation
Proposed changes to the company’s articles of incorporation are shown below related to Items 4 and 5,, “Items "Items of Business To Be Acted Upon at the Meeting.”Business." The changes shown to Article 9(b) will be effective if Item 4,, "Proposal to Amend the Company’s Articles of Incorporation to Eliminate the Classified Board Structure,”" receives the vote of at least 80 percent of the outstanding shares. The changes to Articles 9(c), 9(d), and 13 will be effective if Item 5,, "Proposal to Amend the Company’s Articles of Incorporation to Eliminate Supermajority Voting Provisions,”" receives the vote of at least 80 percent of the outstanding shares. Additions are indicated by underlining and deletions are indicated by strike-outs. The full text of the company'scompany’s Articles of Incorporation can be found on our website at: https://www.lilly.com/who-we-are/governance.at lilly.com/leadership/governance.
. . . . .
9. The following provisions are inserted for the management of the business and for the conduct of the affairs of the Corporation, and it is expressly provided that the same are intended to be in furtherance and not in limitation or exclusion of the powers conferred by statute:
(a) The number of directors of the Corporation, exclusive of directors who may be elected by the holders of any one or more series of Preferred Stock pursuant to Article 7(b) (the "Preferred“Preferred Stock Directors"Directors”), shall not be less than nine, the exact number to be fixed from time to time solely by resolution of the Board of Directors, acting by not less than a majority of the directors then in office.
(b) Prior to the 20192023 annual meeting of directors, T thethe Board of Directors (exclusive of Preferred Stock Directors) shall be divided into three classes, with the term of office of one class expiring each year. At the annual meeting of shareholders in 1985, five directors of the first class shall be elected to hold office for a term expiring at the 1986 annual meeting, five directors of the second class shall be elected to hold office for a term expiring at the 1987 annual meeting, and six directors of the third class shall be elected to hold office for a term expiring at the 1988 annual meeting.Commencing with the annual meeting of shareholders in 198620192023, each class of directors whose term shall then expire shall be elected to hold office for a threeone-year term.expiring at the next annual meeting of shareholders.shareholders. In the case of any vacancy on the Board of Directors, including a vacancy created by an increase in the numberof Directors,Ddirectors, the vacancy shall be filled by election of the Board of Directors with the director so elected to serve for the remainder of the term of the director being replaced or, in the case of an additional director, for the remainder of the term of the class to which the director has been assigned.assigned until the next annual meeting of shareholders. All directors shall continue in office until the election and qualification of their respective successors in office. When the number of directors is changed, any newly created directorships or any decrease in directorships shall be so assigned among the classes by a majority of the directors then in office, though less than a quorum, as to make all classes as nearly equal in number as possible.No decrease in the number of directors shall have the effect of shortening the term of any incumbent director. Election of directors need not be by written ballot unless the By-laws so provide.
(c) Any director or directors (exclusive of Preferred Stock Directors) may be removed from office at any time, but only for cause and only by the affirmative vote of at least 80%a majority of the votes entitled to becastby theholders of all the outstanding shares of Voting Stock (as defined in Article 13 hereof), voting together as a single class.
(d) Notwithstanding any other provision of these Amended Articles of Incorporation or of law which might otherwise permit a lesser vote or no vote, but in addition to any affirmative vote of the holders of any particular class of Voting Stock required by law or these Amended Articles of Incorporation, the affirmative vote of at least 80% of the votes entitled to be cast by holders of all the outstanding shares of Voting Stock, voting together as a single class, shall be required to alter, amend or repeal this Article 9.
13. In addition to all other requirements imposed by law and these Amended Articles and except as otherwise expressly provided in paragraph (c) of this Article 13,, none of the actions or transactions listedin paragraph (a) below shall be effected by the Corporation, or approved by the Corporation as a shareholder of any majority-owned subsidiary of the Corporation if, as of the record date for the determination of the shareholders entitled to vote thereon, any Related Person (as hereinafter defined) exists, unless the applicable requirements of paragraphs (b),
(c), (d), (e), and (fe)(f) of this Article 13 are satisfied.
(a) The actions or transactions within the scope of this Article 13 are as follows:
(i) any merger or consolidation of the Corporation or any of itsthe Corporation’s subsidiaries into or with such Related Person;
(ii) any sale, lease, exchange, or other disposition of all or any substantial part of the assets of the Corporation or any of itsitsthe Corporation’s majority-owned subsidiaries to or with such Related Person;
(iii) the issuance or delivery of any Voting Stock (as hereinafter defined) or of voting securities of any of the Corporation'sCorporation’s majority-owned subsidiaries to such Related Person in exchange for cash, other assets or securities, or a combination thereof;
(iv) any voluntary dissolution or liquidation of the Corporation;
(v)(iv) any reclassification of securities (including any reverse stock split), or recapitalization of the Corporation, or any merger or consolidation of the Corporation with any of its subsidiaries, or any other transaction (whether or not with or otherwise involving a Related Person) that has the effect, directly or indirectly, of increasing the proportionate share of any class or series of capital stock of the Corporation, or any securities convertible into capital stock of the Corporation or into equity securities of any subsidiary, that is beneficially owned by any Related Person; or
(vi)(vi) any agreement, contract, or other arrangement providing for any one or more of the actions specified in the foregoing clauses (i) through (v)(iv).
(b) The actions and transactions described in paragraph (a) of this Article 13 shall have been authorized by the affirmative vote of at least 80% of alla majority of the votes entitled to be cast by holders of all the outstanding shares of Voting Stock, voting together as a single class.
(c) Notwithstanding paragraph (b) of this Article 13, the 80% voting special shareholder approvalrequirementset forth in paragraph (b) shall not be applicable if any action or transaction specified in paragraph (a) is approved by the Corporation'sCorporation’s Board of Directors and by a majority of the Continuing Directors (as hereinafter defined).
(d(c)(d) Unless approved by a majority of the Continuing Directors, after becoming a Related Person and prior to consummation of such action or transaction.transaction.:
(i) the Related Person shall not have acquired from the Corporation or any of its subsidiaries any newly issued or treasury shares of capital stock or any newly issued securities convertible into capital stock of the Corporation or any of its majority-owned subsidiaries, directly or indirectly (except upon conversion of convertible securities acquired by it prior to becoming a Related Person or as a result of a pro rata stock dividend or stock split or other distribution of stock to all shareholders pro rata);
(ii) such Related Person shall not have received the benefit, directly or indirectly (except proportionately as a shareholder), of any loans, advances, guarantees, pledges, or other financial assistance or tax credits provided by the Corporation or any of its majority-owned subsidiaries, or made any major changes in the Corporation'sCorporation’s or any of its majority-owned subsidiaries'subsidiaries’ businesses or capital structures or reduced the current rate of dividends payable on the Corporation'sCorporation’s capital stock below the rate in effect immediately prior to the time such Related Person became a Related Person; and
(iii) such Related Person shall have taken all required actions within its power to ensure that the Corporation'sCorporation’s Board of Directors included representation by Continuing Directors at least proportionate to the voting power of the shareholdings of Voting Stock of the Corporation'sCorporation’s Remaining Public Shareholders (as hereinafter defined), with a Continuing Director to occupy an additional Board position if a fractional right to a director results and, in any event, with at least one Continuing Director to serve on the Board so long as there are any Remaining Public Shareholders.
(ed)(e) A proxy statement responsive to the requirements of the Securities Exchange Act of 1934, as amended, whether or not the Corporation is then subject to such requirements, shall be mailed to the
shareholders of the Corporation for the purpose of soliciting shareholder approval of such action or transaction and shall contain at the front thereof, in a prominent place, any recommendations as to the advisability or inadvisability of the action or transaction which the Continuing Directors may choose to state and, if deemed advisable by a majority of the Continuing Directors, the opinion of an investment banking firm selected by a majority of the Continuing Directors as to the fairness (or not) of the terms of the action or transaction from a financial point of view to the Remaining Public Shareholders, such investment banking firm to be paid a reasonable fee for its services by the Corporation. The requirements of this paragraph (ed)(e) shall not apply to any such action or transaction which is approved by a majority of the Continuing Directors.
(fe)(f) For the purpose of this Article 13
13:
(i) the term "Related Person"“Related Person” shall mean any other corporation, person, or entity which beneficially owns or controls, directly or indirectly, 5% or more of the outstanding shares of Voting Stock, and any Affiliate or Associate (as those terms are defined in the General Rules and Regulations under the Securities Exchange Act of 1934) of a Related Person; provided however, , however, that the term Related Person shall not include (a) the Corporation or any of its subsidiaries, (b) any profit-sharing, employee stock ownership or other employee benefit plan of the Corporation or any subsidiary of the Corporation or any trustee of or fiduciary with respect to any such plan when acting in such capacity, or (c) Lilly Endowment, Inc.; and furtherprovided, that no corporation, person, or entity shall be deemed to be a Related Person solely by reason of being an Affiliate or Associate of Lilly Endowment, Inc.;
(ii) a Related Person shall be deemed to own or control, directly or indirectly, any outstanding shares of Voting Stock owned by it or any Affiliate or Associate of record or beneficially, including, without limitation, shares
a. which it has the right to acquire pursuant to any agreement, or upon exercise of conversion rights, warrants, or options, or otherwiseotherwise; or
b. which are beneficially owned, directly or indirectly (including shares deemed owned through application of clause a. above), by any other corporation, person, or other entity with which it or its Affiliate or Associate has any agreement, arrangement, or understanding for the purpose of acquiring, holding, voting, or disposing of Voting Stock, or which is its Affiliate (other than the Corporation) or Associate (other than the Corporation);
(iii) the term "Voting Stock"“Voting Stock” shall mean all shares of any class of capital stock of the Corporation which are entitled to vote generally in the election of directors;
(iv) the term "Continuing Director"“Continuing Director” shall mean a director who is not an Affiliate or Associate or representative of a Related Person and who was a member of the Board of Directors of the Corporation immediately prior to the time that any Related Person involved in the proposed action or transaction became a Related Person or a director who is not an Affiliate or Associate or representative of a Related Person and who was nominated by a majority of the remaining Continuing Directors; and
(v) the term "Remaining“Remaining Public Shareholders"Shareholders” shall mean the holders of the Corporation'sCorporation’s capital stock other than the Related Person.
(gf)(g) A majority of the Continuing Directors of the Corporation shall have the power and duty to determine for the purposes of this Article 13, on the basis of information then known to the Continuing Directors, whether
(i) any Related Person exists or is an Affiliate or an Associate of another and (ii) any proposed sale, lease, exchange, or other disposition of part of the assets of the Corporation or any majority-owned subsidiary involves a substantial part of the assets of the Corporation or any of its subsidiaries. Any such determination by the Continuing Directors shall be conclusive and binding for all purposes.
(hg)(h) Nothing contained in this Article 13 shall be construed to relieve any Related Person or any Affiliate or Associate of any Related Person from any fiduciary obligation imposed by law.
(ih)(i) The fact that any action or transaction complies with the provisions of this Article 13 shall not be construed to waive or satisfy any other requirement of law or these Amended Articles of Incorporation or to impose any fiduciary duty, obligation, or responsibility on the Board of Directors or any member thereof, to
approve such action or transaction or recommend its adoption or approval to the shareholders of the Corporation, nor shall such compliance limit, prohibit, or otherwise restrict in any manner the Board of Directors, or any member thereof, with respect to evaluations of or actions and responses taken with respect to such action or transaction. The Board of Directors of the Corporation, when evaluating any actions or transactions described in paragraph (a) of this Article 13, shall, in connection with the exercise of its judgment in determining what is in the best interests of the Corporation and its shareholders, give due consideration to all relevant factors, including, without limitation, the social and economic effects on the employees, customers, suppliers, and other constituents of the Corporation and its subsidiaries and on the communities in which the Corporation and its subsidiaries operate or are located.
(j)Notwithstanding any other provision of these Amended Articles of Incorporation or of law which might otherwise permit a lesser vote or no vote, but in addition to any affirmative vote of the holders of any particular class of Voting Stock required by law or these Amended Articles of Incorporation, the affirmative vote of the holders of at least 80% of the votes entitled to be cast by holders of all the outstanding shares of Voting Stock, voting together as a single class, shall be required to alter, amend, or repeal this Article 13.
Appendix C - Proposed Amended and Restated 2002 Lilly Stock Plan
AMENDED AND RESTATED
2002 LILLY
STOCK PLAN
(Effective May 07, 2018)
ARTICLE 1. PURPOSES OF THE PLAN
The Company believes that this Amended and Restated 2002 Lilly Stock Plan, as amended from timeAmendments to time (the “Plan”), will benefit the Company’s shareholders by allowing the Company to attract, motivate and retain the best available Employees and Directors and by providing those Employees and Directors stock-based incentives to strengthen the alignment of interests between those persons and the Company’s shareholders.
ARTICLE 2. DEFINITIONS
Wherever the following terms are used in the Plan, they shall have the meanings specified below, unless the context clearly indicates otherwise. The singular pronoun shall include the plural where the context so indicates.
2.1 “Affiliate” shall have the meaning given to such term in Rule 12b-2 promulgated under the Exchange Act. The Board shall have the authority to determine the time or times at which “Affiliate” status is determined within the foregoing definition.
2.2 “Applicable Laws” means the requirements relating to the administration of equity-based and cash-based awards, as applicable, and the related issuance of Shares under U.S. state corporate laws, U.S. federal and state and non-U.S. securities laws, the Code, any stock exchange or quotation system on which the Common Stock is listed or quoted and the applicable laws of any non-U.S. country or jurisdiction where Awards are, or will be, granted under the Plan.
2.3 “Award” means an Option, Restricted Stock Units, Restricted Stock, a Stock Appreciation Right, Dividend Equivalent Rights, an Other Share-Based Award or a Performance-Based Award granted to a Participant pursuant to the Plan.
2.4 “Award Agreement” means any written agreement, contract, or other instrument or document evidencing the terms and conditions of an Award, including through electronic medium.
2.5 “Board” means the board of directors of the Company.
2.6 “Change in Control” means and includes each of the following:
(a) the acquisition by any “person,” as that term is used in Sections 13(d) and 14(d) of the Exchange Act (other than (i) the Company, (ii) any subsidiary of the Company, (iii) any employee benefit plan or employee stock plan of the Company or a subsidiary of the Company or any trustee or fiduciary with respect to any such plan when acting in that capacity, or (iv) Lilly Endowment, Inc.) of “beneficial ownership,” as defined in Rule 13d-3 under the Exchange Act, directly or indirectly, of twenty percent (20%) or more of the shares of the Company’s capital stock the holders of which have general voting power under ordinary circumstances to elect at least a majority of the Board (or which would have such voting power but for the application of the Indiana Control Shares Statute) (“Voting Stock”); provided, however, that an acquisition of Voting Stock directly from the Company shall not constitute a Change in Control under this Section 2.6(a);
(b) the first day on which less than one-half of the total membership of the Board shall be Continuing Directors (as that term is defined in Section 13(f) of the Company's Articles of Incorporation);Incorporation
(c) consummationProposed changes to the company’s articles of a merger, share exchange, or consolidationincorporation are shown below related to Item 6, "Items of Business." The changes shown to Article 10 will be effective if the votes cast in favor of Item 6, "Proposal to Amend the Company’s Articles of Incorporation to Give Shareholders the Ability to Amend the Company's Bylaws," exceeds the votes cast against this proposal. Additions are indicated by underlining and deletions are indicated by strike-outs. The full text of the Company (a “Transaction”), other than a Transaction which would result in the Voting Stockcompany’s Articles of Incorporation can be found on our website at lilly.com/leadership/governance.
10. The Board of Directors Subject to any express provision of the Company outstanding immediately prior thereto continuingIndiana BusinessCorporation is exclusively authorized (a) to represent (either by remaining outstandingadopt, repeal, alter or by being converted into voting
securities of the surviving entity) more than sixty percent (60%) of the Voting Stock of the Company or such surviving entity immediately after such Transaction;
(d) a complete liquidation of the Company or a sale or disposition of all or substantially all the assets of the Company, other than a sale or disposition of assets to any subsidiary of the Company.
For purposes of this Section 2.6(a) only, the term “subsidiary” means a corporation or limited liability company of which the Company owns directly or indirectly fifty percent (50%) or more of the voting power.
2.7 “amendCode” means the U.S. Internal Revenue Code of 1986, as amended. All references herein to specific sections of the Code shall include any successor provisions of the Code or corresponding sections of any future U.S. federal tax code.
2.8 “Committee” means the committee of the Board appointed or described in Article 3 to administer the Plan.
2.9 “Common Stock” means the common stock of the Company, no par value, and such other securities of the Company that may be substituted for the Common Stock pursuant to Article 13.
2.10 “Company” means Eli Lilly and Company, an Indiana corporation, and any successor corporation thereto.
2.11 “Director” means a member of the Board.
2.12 “Disability”means, unless otherwise provided in an Award Agreement, that the Participant would qualify to receive benefit payments under the long-term disability plan or policy, as it may be amended from time to time, of the Company or the Affiliate to which the Participant provides Service regardless of whether the Participant is covered by such policy. If the Company or the Affiliate to which the Participant provides Service does not have a long-term disability policy, “Disability” means that a Participant is unable to carry out the responsibilities and functions of the position held by the Participant by reason of any medically determined physical or mental impairment for a period of not less than ninety (90) consecutive days. A Participant shall not be considered to have incurred a Disability unless he or she furnishes proof of such impairment sufficient to satisfy the Committee in its discretion. Notwithstanding the foregoing, (a) for purposes of Incentive Stock Options granted under the Plan, “Disability” means that the Participant is disabled within the meaning of Section 22(e)(3) of the Code, and (b) with respect to an Award that is subject to Section 409A of the Code where the payment or settlement of the Award will accelerate as a result of the Participant’s Disability, solely for purposes of determining the timing of payment, no such event will constitute a Disability for purposes of the Plan or any Award Agreement unless such event also constitutes a “disability” as defined under Section 409A of the Code.
2.13 “Dividend Equivalent Right” means a right to receive the equivalent value of dividends paid on the Shares with respect to Shares underlying Restricted Stock Units or an Other Share-Based Award that is a Full Value Award prior to vesting of the Award in accordance with the provision of Section 12.4.
2.14 “Effective Date” means the date that the shareholders approved the amendment and restatement of the Plan.
2.15 “Eligible Individual” means any natural person who is an Employee or a Director determined by the Committee as eligible to participate in the Plan.
2.16 “Employee” means an individual, including an officer or Director, who is treated as an employee in the personnel records of the Company or an Affiliate and providing Service to the Company or the Affiliate. Neither services as a Director nor payment of a director’s fee by the Company or an Affiliate shall be sufficient to constitute “employment” by the Company or an Affiliate.
2.17 “Equity Restructuring” shall mean a nonreciprocal transaction between the Company and its shareholders, such as a stock dividend, stock split, spin-off, rights offering or recapitalization through a large, nonrecurring cash dividend, that affects the Shares (or other securities of the Company) or the price of Shares (or other securities) and causes a change in the per-share value of the Shares underlying outstanding Awards.
2.18 “Exchange Act” means the U.S. Securities Exchange Act of 1934, as amended.
2.19 “Fair Market Value” means, as of any given date, (a) if Shares are traded on any established stock exchange, the closing price of a Share as quoted on the principal exchange on which the Shares are listed, as reported in The Wall Street Journal (or such other source as the Company may deem reliable for such purposes) for such date, or if no sale occurred on such date, the first trading date immediately prior to such date during which a sale occurred; or (b) if Shares are not traded on an exchange but are regularly quoted on a national market or other quotation system, the closing sales price on such date as quoted on such market or system, or if no sales occurred on such date, then on the date immediately prior to such date on which sales prices are reported; or (c) in the absence of an established market for the Shares of the type described in (a) or (b) of this Section 2.19, the fair market value established by the Committee acting in good faith, under a reasonable methodology and reasonable application in compliance with Section 409A of the Code to the extent such determination is necessary for Awards under the Plan to comply with, or be exempt from, Section 409A of the Code.
Notwithstanding the foregoing, for income tax reporting purposes under U.S. federal, state, local or non-US law and for such other purposes as the Committee deems appropriate, including, without limitation, where Fair Market Value is used in reference to exercise, vesting, settlement or payout of an Award, the Fair Market Value shall be determined by the Company in accordance with uniform and nondiscriminatory standards adopted by it from time to time.
2.20 “Full Value Award” means any Award other than an (i) Option, (ii) Stock Appreciation Right or (iii) other Award for which the Participant pays (or the value or amount payable under the Award is reduced by) an amount equal to or exceeding the Fair Market Value of the Shares, determined as of the date of grant.
2.21 “Incentive Stock Option” means an Option that is intended to meet the requirements of Section 422 of the Code.
2.22 “Non-Employee Director” means a Director of the Company who is not an Employee.
2.23 “Non-Qualified Stock Option” means an Option that is not intended to be an Incentive Stock Option.
2.24 “Option” means a right granted to a Participant pursuant to Article 6 to purchase a specified number of Shares at a specified price during specified time periods. An Option may be either an Incentive Stock Option or a Non-Qualified Stock Option.
2.25 “Other Share-Based Award” shall mean an Award granted pursuant to Article 10.
2.26 “Outstanding Qualified Performance-Based Awards” shall mean any Awards granted prior to November 3, 2017 and that are outstanding as of the Effective Date and that are intended to constitute “qualified performance-based compensation” as described in Section 162(m)(4)(C) of the Code. For the avoidance of any doubt, all provisions of the Plan governing Outstanding Qualified Performance Awards that were in effect prior to the Effective Date shall continue in effect with respect to Outstanding Qualified Performance-Based Awards, notwithstanding the elimination of such provisions from the Plan as of the Effective Date.
2.27 “Participant” means any Eligible Individual who, as an Employee or Director, has been granted an Award pursuant to the Plan.
2.28 “Performance-Based Award” means an Award that are subject, in whole or in part, to Performance Goals and are granted pursuant to Article 10.
2.29 “Performance Criteria” means the criteria that the Committee selects for purposes of establishing the Performance Goal or Performance Goals for a Participant for a Performance Period. The Performance Criteria that will be used to establish Performance Goals include, but are not limited to, the following: cash flow (including, without limitation, operating cash flow and free cash flow), earnings per share, gross or net profit margin, net income (either before or after interest, taxes, amortization, and/or depreciation), operating income (either before or after restructuring and amortization charges), return on capital or return on invested capital, return on equity, return on operating assets or net assets, return on sales, sales or revenue, stock price goals, total shareholder return. The Committee shall define objectively the manner of calculating the Performance Criteria it selects to use for such Performance Period for such Participant.
2.30 “Performance Goals” means, for a Performance Period, the goals established in writing by the Committee for the Performance Period based upon the Performance Criteria that the Committee, in its sole discretion, selects. The Committee, in its sole discretion, may provide that one or more objectively determinable adjustments shall be made to one or more of the Performance Goals.
2.31 “Performance Period” means the one or more periods of time, which may be of varying and overlapping durations, as the Committee may select, over which the attainment of one or more Performance Goals will be measured for the purpose of determining a Participant’s right to, and the payment of, a Performance-Based Award, provided that the duration of any Performance Period shall not be less than twelve (12) months.
2.32 “Plan” means this Amended and Restated 2002 Lilly Stock Plan, as it may be amended from time to time.
2.33 “Prior Plans” means the 1989, 1994 and 1998 Lilly Stock Plans, as amended from time to time.
2.34 “Restricted Stock” means Shares awarded to a Participant pursuant to Article 8 that are subject to certain restrictions and may be subject to risk of forfeiture.
2.35 “Restricted Stock Unit” means an Award granted pursuant to Article 7 that shall be evidenced by a bookkeeping entry representing the equivalent of one Share.
2.36 “Securities Act” means the U.S. Securities Act of 1933, as amended.
2.37 “Service” means service as an Employee or Non-Employee Director. Except as otherwise determined by the Committee in its sole discretion, a Participant’s Service terminates when the Participant ceases to actively provide services to the Company or an Affiliate and shall not be extended by any notice period mandated under applicable employment laws or the terms of the Participant’s employment or service contract, if any. The Committee shall determine which leaves shall count toward Service and when Service terminates for all purposes under the Plan. Further, unless otherwise determined by the Committee, a Participant’s Service shall not be deemed to have terminated merely because of a change in the capacity in which the Participant provides Service to the Company or an Affiliate, or a transfer between entities (i.e.Law, the Company or any Affiliates), providedBy-laws that there is no interruption or other termination of Service in connection with the Participant’s change in capacity or transfer between entities (except asCorporation may be required to effectadopted, repealed, altered or amended by either (i) the change in capacity or transfer between entities). For purposesBoard of determining whether an Option is entitled to Incentive Stock Option status, an Employee’s Service shall be treated as terminated ninety (90) days after such Employee goes on leave, unless such Employee’s right to return to active work is guaranteed by law or by a contract.
2.38 “DirectorsShare” means a share of Common Stock.
2.39 “Stock Appreciation Right” or “SAR” means a right granted pursuant to Article 9 to receive a payment equal to the excess of the Fair Market Value of a specified number of Shares on the date the SAR is exercised over the exercise price of the SAR, as set forth in the applicable Award Agreement.
2.40 “CorporationTax-Related Items” means any U.S. federal, state, and/or local taxes and any taxes imposed by a jurisdiction outside of the U.S. (including, without limitation, income tax, social insurance and similar contributions, payroll tax, fringe benefits tax, payment on account, employment tax, stamp tax and any other taxes related to participation in the Plan and legally applicable to a Participant, including any employer liability for which the Participant is liable pursuant to Applicable Laws or the applicable Award Agreement).
ARTICLE 3. ADMINISTRATION
3.1 Committee. The Board, at its discretion or as otherwise necessary to comply with the requirements of Section 162(m) of the Code (with respect to Outstanding Qualified Performance-Based Awards), Rule 16b-3 promulgated under the Exchange Act or to the extent required by any other Applicable Law or regulation, may delegate administration of the Plan to a Committee consisting of two or more members of the Board. Unless otherwise determined by the Board, the Committee shall consist solely of two or more Non-Employee Directors, each of whom is an “outside director,” within the meaning of Section 162(m) of the Code (with respect to Outstanding Qualified Performance-Based Awards), a “non-employee director” within the meaning of Rule 16b-3(b)(3) under the Exchange Act, or any successor rule, and an “independent director” under the applicable New York Stock Exchange rules (or other principal securities
market on which Shares are traded). Notwithstanding the foregoing: (a) the full Board, acting by a majority of its members in office, shall conduct the general administration of the Plan with respect to all Awards granted to Non-Employee Directors and for purposes of such Awards the term “Committee” as used in this Plan shall be deemed to refer to the Board and (b) the Committee may delegate its authority hereunder to the extent permitted by Section 3.5 hereof. Unless and until the Board delegates administration of the Plan to a Committee as set forth below, the Plan shall be administered by the full Board, and for such purposes the term “Committee” as used in this Plan shall be deemed to refer to the Board. In its sole discretion, the Board may at any time and from time to time exercise any and all rights and duties of the Committee under the Plan, except with respect to matters which under Rule 16b-3 under the Exchange Act or Section 162(m) of the Code (with respect to Outstanding Qualified Performance-Based Awards), or any regulations or rules issued thereunder, are required to be determined in the sole discretion of the Committee.
3.2 Action by the Committee. Unless otherwise established by the Board or in any charter of the Committee, a majority of the Committee shall constitute a quorum and the actsvote of a majority of the members present atentire Board of Directors and, or (bii) toadopt any meeting atBy-laws which a quorum is present, and acts approved in writing by a majoritythe Board of the Committee in lieu of a meeting, shall be deemed the acts of the Committee. Each member of the Committee is entitled to, in good faith, rely or act upon any report or other information furnished to that member by any officer or other employee of the Company or any Affiliate, the Company’s independent certified public accountants, or any executive compensation consultant or other professional retained by the Company to assist in the administration of the Plan.
3.3 Authority of Committee. Subject to any specific designation in the Plan, the Committee has the exclusive power, authority and discretion to:
(a) designate Participants to receive Awards;
(b) determine the type or types of Awards to be granted to each Participant;
(c) determine the number of Awards to be granted and the number of Shares to which an Award will relate;
(d) determine the terms and conditions of any Award granted pursuant to the Plan, including, without limitation, the exercise price, grant price, or purchase price, any restrictions or limitations on the Award, any schedule for lapse of forfeiture restrictions or restrictions on the exercisability of an Award, and accelerations or waivers thereof, any provisions related to recoupment of gain on an Award, based in each case on such considerations as the Committee in its sole discretion determines;
(e) determine whether, to what extent, and pursuant to what circumstances an Award may be settled in, or the exercise price of an Award may be paid in, cash, Shares, other Awards, or other property, or an Award may be cancelled, forfeited, or surrendered;
(f) prescribe the form of each Award Agreement, which need not be identical for each Participant and may vary for Participants within and outside of the U.S.;
(g) decide all other matters that must be determined in connection with an Award;
(h) establish, adopt or revise any rules and regulations, including adopting sub-plans to the Plan, for the purposes of facilitating compliance with foreign laws, easing the administration of the Plan and/or taking advantage of tax-favorable treatment for Awards granted to Participants outside the U.S., in each case as itDirectors may deem necessary or advisable;
(i) suspend or terminatedesirable for the Plan at any time, subject to Article 15;
(j) amend or modifyefficient conduct of the termsaffairs of an Award,the Corporation, including, without limitation, accelerateprovisions governing the vesting and/or exercisabilityconduct of, any Award for any reason, including, without limitation,and the Participant's retirement or other termination; provided, however, that no amendment or modification of an outstanding Award other than the following types of amendments or modifications shall affect adversely, in any material way, any Award previously granted pursuant to the Plan without the prior written consentmatters which may properly be brought before, meetings of the Participant: (i) an amendment or modification that may cause an Incentive Stock Optionshareholders and provisions specifying the manner and extent to becomewhich prior notice shall be giventhe affirmative vote of at least a Non-Qualified Stock Option; (ii) an amendment made or other action taken pursuant to Section 16.14 majority of the Plan; (iii) any amendment or other action that mayvotes entitled to be required or desirablecast by holders of all the submission of proposals to facilitate compliance with Applicable Laws, as determined in the sole discretion of the Committee .
(k) interpret the terms of, and any matter arising pursuant to, the Plan or any Award Agreement; and
(l) make all other decisions and determinations that may be required pursuant to the Plan or that the Committee deems necessary or advisable to administer the Plan.
3.4 Decisions Binding. The Committee’s interpretation of the Plan, any Awards granted pursuant to the Plan, and any Award Agreement and all decisions and determinations by the Committee with respect to the Plan are final, binding, and conclusive on all parties.
3.5 Delegation of Authority. To the extent permitted by Applicable Laws, the Board, from time to time, may delegate to a Committee of one or more members of the Board (pursuant to delegation that does not meet the requirement of Section 3.1 hereof) or to one or more officers of the Company the authority to grant Awards to Participants other than (a) Employees who are subject to Section 16 of the Exchange Act, or (b) officers of the Company (or Directors) to whom authority to grant or amend Awards has been delegated hereunder. Furthermore, if the authority to grant or amend Awards has been delegated to the Committee pursuant and subject to the preceding sentence, such authority may be further delegated by the Committee to one or more officers of the Company. For the avoidance of doubt, provided it meets the limitations of this Section 3.5, any delegation hereunder shall include the right to modify Awards as necessary to accommodate changes in Applicable Laws or regulations, including in jurisdictions outside the U.S. Furthermore, any delegation hereunder shall be subject to the restrictions and limitations that the Board (or, as applicable, the Committee) specifies at the time of such delegation, and the Board (or, as applicable, the Committee) may rescindsubmitted at any time the authority so delegated and/or appoint a new delegatee. At all times, the delegatee appointed under this Section 3.5 shall serve in such capacity at the pleasureoutstanding shares of the Board (or, as applicable, the Committee).
ARTICLE 4. SHARES SUBJECT TO THE PLAN
4.1 Number of Shares. Subject to Article 13 hereof, the aggregate number of Shares that may be issued or transferred pursuant to Awards under the Plan shall be the sum of (i) 75,657,296 Shares, plus (ii) the number of Shares available for issuance under the Prior Plans (including Shares subject to awards granted under the Prior Plans that otherwise subsequently became available for issuance under the Prior Plans upon forfeiture, cancellation, or termination of the awards or any other reason under the terms of the Prior Plans); provided, however, that only 53,000,000 Shares may be issued or transferred pursuant to new Awards granted on or following the Effective Date. Subject to Article 13, the aggregate number of Shares that may be issued or transferred pursuant to the exercise of IncentiveVoting Stock, Options shall be 30,000,000.
(a) Shares Reissuable under Plan. The following Shares shall again be available for the grant of an Award pursuant to the Plan: (i) Shares that are not issuedvoting together as a resultsingle class at a duly called and constituted meeting of the termination, expirationshareholders or lapsing of any Award for any reason; (ii) Shares subject to a Full Value Award that are not issued because the Award is settled in cash; (iii) Shares covered by an Option which are surrendered in paymentnominations of the Option exercise or purchase price or in satisfactionelections of obligations for Tax-Related Items incident to the exercise of an Option; (iv) Shares covered by an Award which are surrendered in satisfaction of obligations for Tax-Related Items incident to the vesting or settlement of a Full Value Award. Notwithstanding the provisions of this Section 4.1, no Shares may again be optioned, granted or awarded if such action would cause an Incentive Stock Option to fail to qualify as an Incentive Stock Option.
(b) Shares Not Reissuable under Plan. Notwithstanding the foregoing, Shares that are repurchased on the open market with the proceeds of the exercise of an Option shall be counted against the maximum number of Shares available for issuance pursuant to Section 4.1 hereof and shall not be returned to the Plan.
(c) Shares Not Counted Against Share Pool Reserve. To the extent permitted by Applicable Laws, Shares issued in assumption of, or in substitution for, any outstanding awards of any entity acquired in any form of combination by the Company or an Affiliate shall not be counted against Shares available for grant pursuant to this Plan. Additionally, to the extent permitted by Applicable Laws, in the event that a company acquired by (or combined with) the Company or an Affiliate has shares available under a pre-existing plan approved by its shareholders and not adopted in contemplation of such acquisition or combination, the shares available for grant pursuant to the terms of such pre-existing plan (as adjusted, to the extent appropriate, using the exchange ratio or other adjustment or valuation ratio or formula used in such acquisition or combination to determine the consideration payable to the shareholders of the entities party to such acquisition or combination) may, at the discretion of the Committee, be used for Awards under the Plan in lieu of awards under the applicable pre-existing plan of the other company and
shall not reduce the Shares authorized for grant under the Plan; provided that Awards using such available shares shall not be made after the date awards or grants could have been made under the terms of the pre-existing plan absent the acquisition or combination, and shall only be made to individuals who were not employees or directors of the Company or any Affiliate in existence prior to such acquisition or combination by the Company or an Affiliate. The payment of Dividend Equivalent Rights in cash in conjunction with any outstanding Awards shall not be counted against the Shares available for issuance under the Plan.
4.2 Shares Distributed. Any Shares distributed pursuant to an Award may consist, in whole or in part, of authorized and unissued Shares, treasury Shares or Shares purchased on the open market, subject to Section 4.1(b)(ii) hereof.
4.3 Limitation on Number of Shares Subject to Awards. Notwithstanding any provision in the Plan to the contrary, and subject to Article 13, the maximum number of Shares subject to all Awards that may be granted to any one Participant during any calendar year shall be 1,500,000 Shares.
4.4 Non-Employee Director Award Limit. Notwithstanding any provision to the contrary in the Plan or in any policy of the Company regarding compensation payable to a Non-Employee Director, the sum of the grant date fair value (determined as of the grant date in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718, or any successor thereto) of all Awards payable in Common Stock to an individual as compensation for services as a Non-Employee Director, together with cash compensation earned by the Non-Employee Director during any calendar year, shall not exceed $800,000 in any calendar year.
ARTICLE 5. ELIGIBILITY AND PARTICIPATION
5.1 Eligibility. Each Eligible Individual shall be eligible to be granted one or more Awards pursuant to the Plan. An Eligible Individual who is subject to taxation in the U.S. and who is providing Services to an Affiliate may be granted Options or SARs under this Plan only if the Affiliate qualifies as an “eligible issuer of service recipient stock” within the meaning of the U.S. Department of Treasury regulations promulgated under Section 409A of the Code.
5.2 Participation. Subject to the provisions of the Plan, the Committee, from time to time, may select from among all Eligible Individuals those to whom Awards shall be granted, and shall determine the nature and amount of each Award. No Eligible Individual shall haveheld at any right to be granted an Award pursuant to this Plan and the grant of an Award to an Eligible Individual shall not imply any entitlement to receive future Awards.
ARTICLE 6. STOCK OPTIONS
6.1 General. The Committee is authorized to grant Options to Eligible Individuals on the following terms and conditions, and the Committee may specify such additional terms and conditions as:
(a) Exercise Price. The exercise price per Share subject to an Option shall be determined by the Committee and set forth in the Award Agreement; provided that, subject to Section 6.2(c) hereof, the per-Share exercise price for any Option shall not be less than 100% of the Fair Market Value of a Share on the date of grant.
(b) Time and Conditions of Exercise. The Committee shall determine the time or times at which an Option may be exercised in whole or in part; provided that the term of any Option granted under the Plan shall not exceed ten (10) years. Subject to Section 12.3, the Committee also shall specify the vesting conditions, if any, as it deems appropriate that must be satisfied before all or part of an Option may be exercised. The vesting conditions, if any, may be based on, among other conditions, a Participant’s continued Service, the attainment of performance conditions, or a combination of both.
(c) Payment. The Committee shall determine the methods by which the exercise price of an Option may be paid, including the following methods: (i) cash or check; (ii) surrender of Shares or delivery of a properly executed form of attestation of ownership of Shares as the Committee may require (including withholding of Shares otherwise deliverable upon exercise of the Option) which have a Fair Market Value on the date or surrender of attestation equal to the aggregate exercise price of the Shares as to which the Option is to be exercised; (iii) promissory note from a Participant to the Company or a third-party loan guaranteed by the Company (in either case, with such loan bearing interest at no less than such rate as shall then preclude the imputation of interest under the Code); (iv) through the delivery of a notice that the Participant has placed a market sell order with a broker with respect to Shares then issuable upon exercise of the Option, and that the broker has been directed to pay a sufficient
portion of the net proceeds of the sale to the Company in satisfaction of the Option exercise price, provided that payment of such proceeds is then made to the Company upon settlement of such sale; (v) by a “net exercise” arrangement pursuant to which the number of Shares issuable upon exercise of the Option shall be reduced by the largest whole number of Shares having an aggregate fair market value that does not exceed the aggregate exercise price (plus withholding taxes, if applicable) and any remaining balance of the aggregate exercise price (and/or applicable withholding taxes) not satisfied by such reduction in the number of whole Shares to be issued shall be paid by Participant in cash or other form of payment approved by the Committee; (vi) other property acceptable to the Committee; or (vii) any combination of the foregoing methods of payment. The Award Agreement will specify the methods of paying the exercise price available to each Participant. The Committee also shall determine the methods by which Shares shall be delivered or deemed to be delivered to Participants. Notwithstanding any other provision of the Plan to the contrary, no Participant who is a Director or an “executive officer” of the Company within the meaning of Section 13(k) of the Exchange Act shall be permitted to pay the exercise price of an Option, or continue any extension of credit with respect to the exercise price of an Option, with a loan from the Company or a loan arranged by the Company in violation of Section 13(k) of the Exchange Act.
(d) Exercise of Optionmeeting.
(i) Procedure for Exercise; Rights as a Shareholder. An Option may not be exercised for a fraction of a Share. An Option shall be deemed exercised when the Company receives: (A) a notice of exercise (in such form as the Committee may specify from time to time) from the person entitled to exercise the Option, and (B) full payment for the Shares with respect to which the Option is exercised (together with applicable withholding taxes). Full payment may consist of any consideration and method of payment authorized by the Committee and permitted by the Award Agreement and the Plan. Shares issued upon exercise of an Option shall be issued in the name of the Participant. Until the Shares are issued (as evidenced by the appropriate entry on the books of the Company or of a duly authorized transfer agent of the Company), no dividends or Dividend Equivalent Right shall be paid, and no right to vote or receive dividends or Dividend Equivalent Rights or any other rights as a shareholder shall exist with respect to the Shares subject to an Option, notwithstanding the exercise of the Option. The Company shall issue (or cause to be issued) such Shares promptly after the Option is exercised. No adjustment shall be made for a dividend or other right for which the record date is prior to the date the Shares are issued, except as provided in Section 13.1 of the Plan.
(ii) Termination of Participant’s Service. If a Participant ceases to provide Service, including as a result of the Participant’s death or Disability, the Participant may exercise his or her Option within such period of time as is specified in the Award Agreement to the extent that the Option is vested on the date of termination (but in no event later than the expiration of the term of such Option as set forth in the Award Agreement). Unless otherwise provided by the Committee, if on the date of termination of Service the Participant is not vested as to his or her entire Option, the unvested portion of the Option shall be forfeited and the Shares covered by the unvested portion of the Option shall revert to the Plan. If, after termination of Service, the Participant does not exercise his or her Option within the time specified by the Committee, the Option shall terminate, and the Shares covered by such Option shall revert to the Plan. To the extent the Option is exercisable following a Participant’s death, the Option may be exercised by such persons as may be specified in the Award Agreement, which may include any of the following: (i) the Participant’s designated beneficiary, provided that such designation is permitted under Applicable Laws and that such beneficiary has been designated before the Participant’s death in a form acceptable to the Company; (ii) the Participant’s legal representative or representatives; (iii) the person or persons entitled to do so pursuant to the Participant’s last will and testament; or (iv) if the Participant fails to make testamentary disposition of the Option or dies intestate, by the person or persons entitled to receive the Option pursuant to the applicable laws of descent and distribution.
6.2 Incentive Stock Options. Incentive Stock Options shall be granted only to Employees of the Company or any “subsidiary corporation,” as defined in Section 424(f) of the Code and any applicable U.S. Department of Treasury regulations promulgated thereunder, of the Company, and the terms of any Incentive Stock Options granted pursuant to the Plan, in addition to the requirements of Section 6.1 hereof, must comply with the provisions of this Section 6.2.
(a) Expiration. Subject to Section 6.2(c) hereof, an Incentive Stock Option shall expire and may not be exercised to any extent by anyone after the first to occur of the following events:
(i) Ten (10) years from the date of grant, unless an earlier time is set in the Award Agreement;
(ii) Three (3) months after the date of the Participant’s termination of Service on account of any reason other than death or Disability (within the meaning of Section 22(e)(3) of the Code); and
(iii) One (1) year after the date of the Participant’s termination of Service on account of death or Disability (within the meaning of Section 22(e)(3) of the Code).
(b) Dollar Limitation. The aggregate Fair Market Value (determined as of the time the Option is granted) of all Shares with respect to which Incentive Stock Options are first exercisable by a Participant in any calendar year may not exceed US$100,000 or such other limitation as imposed by Section 422(d) of the Code, or any successor provision. To the extent that Incentive Stock Options are first exercisable by a Participant in excess of such limitation, the excess shall be considered Non-Qualified Stock Options.
(c) Ten Percent Owners. An Incentive Stock Option shall be granted to any individual who, at the date of grant, owns stock possessing more than ten percent (10%) of the total combined voting power of all classes of Shares of the Company only if such Option is granted at a price that is not less than 110% of Fair Market Value on the date of grant and the Option is exercisable for no more than five (5) years from the date of grant.
(d) Notice of Disposition. The Participant shall give the Company prompt notice of any disposition of Shares acquired by exercise of an Incentive Stock Option within (i) two (2) years from the date of grant of such Incentive Stock Option or (ii) one (1) year after the transfer of such Shares to the Participant.
(e) Right to Exercise. During a Participant’s lifetime, only the Participant may exercise an Incentive Stock Option.
(f) Failure to Meet Requirements. Any Option (or portion thereof) purported to be an Incentive Stock Option, which, for any reason, fails to meet the requirements of Section 422 of the Code shall be considered a Non-Qualified Stock Option.
ARTICLE 7. RESTRICTED STOCK UNITS
7.1 Restricted Stock Units. The Committee is authorized to grant Restricted Stock Units to Eligible Individuals in such amounts and subject to such terms and conditions not inconsistent with the Plan as the Committee shall impose.
7.2 Vesting Conditions. Subject to Section 12.3, the Committee shall specify the date or dates on which the Restricted Stock Units shall become fully vested and nonforfeitable, and may specify such conditions to vesting, if any, as it deems appropriate. The vesting conditions, if any, may be based on among other conditions, a Participant’s continued Service, the attainment of performance conditions, or a combination of both.
7.3 Form and Timing of Payment. The Committee shall specify the settlement date applicable to each grant of Restricted Stock Units, which date shall not be earlier than the date or dates on which the Restricted Stock Units shall become fully vested and nonforfeitable, or such settlement date may be deferred to any later date, subject to compliance with Section 409A of the Code, as applicable. On the settlement date, the Company shall, subject to Section 12.6(a) hereof and satisfaction of applicable Tax-Related Items (as further set forth in Section 16.3 hereof), transfer to the Participant one Share for each Restricted Stock Unit scheduled to be paid out on such date and not previously forfeited. Alternatively, settlement of a Restricted Stock Unit may be made in cash (in an amount reflecting the Fair Market Value of the Shares that otherwise would have been issued) or any combination of cash and Shares, as determined by the Committee, in its sole discretion, in either case, less applicable Tax-Related Items (as further set forth in Section 16.3 hereof). Until a Restricted Stock Unit is settled, the number of Restricted Stock Units shall be subject to adjustment pursuant to Article 13 hereof.
7.4 Forfeiture. Except as otherwise determined by the Committee at the time of the grant of the Award or thereafter, any Restricted Stock Units that are not vested as of the date of the Participant’s termination of Service shall be forfeited.
7.5 General Creditors. A Participant who has been granted Restricted Stock Units shall have no rights other than those of a general creditor of the Company. Restricted Stock Units represent an unfunded and unsecured obligation of the Company, subject to the terms and conditions of the applicable Award Agreement evidencing the grant of the Restricted Stock Units.
ARTICLE 8. RESTRICTED STOCK AWARDS
8.1 Grant of Restricted Stock. The Committee is authorized to grant Restricted Stock to Eligible Individuals selected by the Committee in such amounts and subject to such terms and conditions not inconsistent with the Plan as the Committee shall impose.
8.2 Purchase Price. At the time of the grant of Restricted Stock, the Committee shall determine the price, if any, to be paid by the Participant for each Share subject to the Award. The purchase price of Shares acquired pursuant to the Award shall be paid either: (i) in cash at the time of purchase; (ii) at the sole discretion of the Committee, by Service rendered or to be rendered to the Company or an Affiliate; or (iii) in any other form of legal consideration that may be acceptable to the Committee in its sole discretion and in compliance with Applicable Laws.
8.3 Issuance and Restrictions. Subject to Section 12.3 hereof, Restricted Stock shall be subject to such restrictions, if any, on transferability and other restrictions as the Committee may impose (including, without limitation, limitations on the right to vote Restricted Stock or the right to receive dividends on the Restricted Stock). The restrictions, if any, may be based on, among other conditions, a Participant’s continued Service, the attainment of performance conditions, or a combination of both. These restrictions, if any, may lapse separately or in combination at such times, pursuant to such circumstances, in such installments, or otherwise, as the Committee determines at the time of the grant of the Award or thereafter.
8.4 Dividends. Any dividends that are distributed with respect to Shares of Restricted Stock shall be paid in accordance with the applicable Award Agreement, subject to the provisions of Section 12.4(b).
8.5 Forfeiture. Except as otherwise determined by the Committee at the time of the grant of the Award or thereafter, upon termination of Service during the applicable restriction period, Restricted Stock that is at that time subject to restrictions shall be forfeited.
8.6 Certificates for Restricted Stock. Restricted Stock granted pursuant to the Plan may be evidenced in such manner as the Committee shall determine. If certificates representing shares of Restricted Stock are registered in the name of the Participant, certificates shall bear an appropriate legend referring to the terms, conditions, and restrictions applicable to such Restricted Stock, and the Company may, at its discretion, retain physical possession of the certificate until such time as all applicable restrictions lapse.
ARTICLE 9. STOCK APPRECIATION RIGHTS
9.1 Grant of Stock Appreciation Rights. The Committee is authorized to grant SARs to Eligible Individuals on the following terms and conditions, and the Committee may specify such additional terms and conditions as:
(a) Exercise Price. The exercise price per Share subject to a SAR shall be determined by the Committee and set forth in the Award Agreement; provided that the exercise price per Share for any SAR shall not be less than 100% of the Fair Market Value of a Share on the date of grant.
(b) Time and Conditions of Exercise. The Committee shall determine the time or times at which a SAR may be exercised in whole or in part; provided that the term of any SAR granted under the Plan shall not exceed ten (10) years. Subject to Section 12.3, the Committee also shall specify the vesting conditions, if any, as it deems appropriate that must be satisfied before all or part of a SAR may be exercised. The vesting conditions, if any, may be based on, among other conditions, a Participant’s continued Service, the attainment of performance conditions, or a combination of both.
(c) A SAR may not be exercised for a fraction of a Share. A SAR shall be deemed exercised when the Company receives a notice of exercise (in such form as the Committee may specify from time to time) from the person entitled to exercise the SAR.
9.2 Tandem Stock Appreciation Rights. A SAR may be granted in connection with an Option, either at the time of grant or at any time thereafter during the term of the Option. A SAR granted in connection with an Option will entitle the holder, upon exercise, to surrender the Option or any portion thereof to the extent unexercised, with respect to the number of Shares as to which such SAR is exercised, and to receive payment of an amount computed as described in Section 9.3. The Option shall, to the extent and when surrendered, cease to be exercisable. A SAR granted in connection with an Option hereunder will have an exercise price per share equal to the per share exercise price of the Option, will be exercisable at such time or times, and only to the extent, that the related Option is exercisable, and will expire no later than the related Option expires. If a related Option is exercised in whole or in part, then the SAR related to the Shares purchased terminates as of the date of such exercise.
9.3 Payment and Limitations on Exercise.
(a) A SAR shall entitle the Participant (or other person entitled to exercise the SAR pursuant to the Plan) to exercise all or a specified portion of the SAR (to the extent then exercisable pursuant to its terms) and to receive from the Company an amount equal to the excess of the aggregate Fair Market Value of the Shares on the date the SAR is exercised over the aggregate exercise price of the SAR, less applicable Tax-Related Items (as further set forth in Section 16.3 hereof), subject to any limitations the Committee may impose.
(b) Payment of the amounts determined under Section 9.3(a) hereof shall be in cash, in Shares (based on the Fair Market Value of the Shares as of the date the SAR is exercised) or a combination of both, as determined by the Committee in the Award Agreement. To the extent Shares are issued upon exercise of a SAR, the Shares shall be issued in the name of the Participant. Until the Shares are issued (as evidenced by the appropriate entry on the books of the Company or of a duly authorized transfer agent of the Company), no dividends or Dividend Equivalent Right shall be paid, and no right to vote or receive dividends or Dividend Equivalent Rights or any other rights as a shareholder shall exist with respect to the Shares subject to a SAR, notwithstanding the exercise of the SAR. The Company shall issue (or cause to be issued) such Shares promptly after the SAR is exercised. No adjustment shall be made for a dividend or other right for which the record date is prior to the date the Shares are issued, except as provided in Section 13.1 of the Plan. The provisions of Section 6.1(d)(ii) regarding the treatment of a termination of the Participant’s Service shall also apply to SARs.
ARTICLE 10. OTHER SHARE-BASED AWARDS
10.1 Grants of Other Share-Based Awards. Subject to limitation under Applicable Laws, the Committee is authorized under the Plan to grant Awards (other than Options, Restricted Stock Units, Restricted Stock and SARs) to Eligible Individuals subject to the terms and conditions set forth in this Article 10 and such other terms and conditions as may be specified by the Committee that are not inconsistent with the provisions of the Plan and that, by their terms, involve or might involve the issuance of, consist of, or are denominated in, payable in, valued in whole or in part by reference to, or otherwise relate to, Shares. The Committee may also grant Shares as a bonus, or may grant other Awards in lieu of obligations of the Company or an Affiliate to pay cash or other property under the Plan or other plans or compensatory arrangements. The terms and conditions applicable to such other Awards shall be determined from time to time by the Committee and set forth in an applicable Award Agreement. The Committee may establish one or more separate programs under the Plan for the purpose of issuing particular forms of Awards to one or more classes of Participants on such terms and conditions as determined by the Committee from time to time.
10.2 Exercise Price. The Committee may establish the exercise price, if any, of any Other Share-Based Award granted pursuant to this Article 10; provided that such exercise price shall not be less than the Fair Market Value of a Share on the date of grant for an Award that is intended to be exempt from Section 409A of the Code.
10.3 Form of Payment. Payments with respect to any Awards granted under Section 10.1 shall be made in cash or cash equivalent, in Shares or any combination of the foregoing, as determined by the Committee.
10.4 Vesting Conditions. Subject to Section 12.3, the Committee shall specify the date or dates on which the Awards granted pursuant to this Article 10 shall become fully vested and nonforfeitable, and may specify such conditions to vesting as it deems appropriate. The vesting conditions may be based on, among other vesting conditions, a Participant’s continued Service, the attainment of performance conditions, or a combination of both.
10.5 Term. Except as otherwise provided herein, the Committee shall set, in its discretion, the term of any Award granted pursuant to this Article 10; provided that the term of any Award granted pursuant to this Article 10 shall not exceed ten (10) years.
ARTICLE 11. PERFORMANCE-BASED AWARDS
11.1 Purpose. If the Committee, in its discretion, decides to grant a Performance-Based Award to an Eligible Individual, the provisions of this Article 11 shall control over any contrary provision contained in Articles 6 through 10; provided that the Committee may in its discretion grant Awards to Eligible Individuals that are based on Performance Criteria or other performance conditions but that do not satisfy the requirements of this Article 11.
11.2 Applicability. This Article 11 shall apply only to those Eligible Individuals selected by the Committee to receive Performance-Based Awards. The designation of an Eligible Individual as a Participant for a Performance Period shall not entitle the Participant, in any manner, to receive an Award for the period. Moreover, the designation of an Eligible Individual as a Participant for a particular Performance Period shall not require designation of such Eligible Individual as a Participant in any subsequent Performance Period and designation of one Eligible Individual as a Participant shall not require designation of any other Eligible Individuals as a Participant in such period or in any other Performance Period.
11.3 Procedures with Respect to Performance-Based Awards. With respect to any Performance-Based Awards, which may be granted to one or more Eligible Individuals, within the first twenty-five percent (25%) of the Performance Period in question or period of Service, the Committee, in writing (a) shall designate one or more Eligible Individuals as eligible for an Award, (b) shall designate the Performance Period over which the Performance Goals shall be measured; (c) shall select the Performance Criteria applicable to the Performance Period, (d) shall establish the Performance Goals, and amounts of such Awards, as applicable, which may be earned for such Performance Period, and (e) shall specify the relationship between Performance Criteria and the Performance Goals and the amounts of such Awards, as applicable, to be earned by each Eligible Individuals for such Performance Period. Following the completion of each Performance Period, the Committee shall certify in writing whether the applicable Performance Goals have been achieved for such Performance Period. In determining the amount earned by an Eligible Individual, the Committee shall have the right to adjust or eliminate the amount payable at a given level of performance to take into account additional factors that the Committee may deem relevant to the assessment of individual or corporate performance for the Performance Period.
11.4 Payment of Performance-Based Awards. Unless otherwise provided in the applicable Award Agreement, a Participant must be providing Service on the day a Performance-Based Award for the appropriate Performance Period is paid to the Participant. Furthermore, unless otherwise provided in the applicable Award Agreement, a Participant shall be eligible to receive payment pursuant to a Performance-Based Award for a Performance Period only if the Performance Goals for such period are achieved.
ARTICLE 12.PROVISIONS APPLICABLE TO AWARDS
12.1 Stand-Alone and Tandem Awards. Awards granted pursuant to the Plan may, in the discretion of the Committee, be granted either alone, in addition to, or in tandem with, any other Award granted pursuant to the Plan. Awards granted in addition to or in tandem with other Awards may be granted either at the same time as or at a different time from the grant of such other Awards.
12.2 Award Agreement. Awards under the Plan shall be evidenced by Award Agreements that set forth the terms, conditions and limitations for each Award, not inconsistent with the Plan, which may include, without limitation, the term of an Award, the provisions applicable in the event the Participant’s Service terminates, and the Company’s authority to unilaterally or bilaterally amend, modify, suspend, cancel or rescind an Award.
12.3 Minimum Vesting Requirements. Notwithstanding any other provision of the Plan, except in connection with Awards granted in connection with assumption or substitution of awards as part of a transaction as contemplated under Section 4.1(c) or Awards that may be settled only in cash, no portion of an Award granted on or after the Effective Date may vest before the first anniversary of the date of grant, subject to accelerated vesting as contemplated under Section 3.3(j) and ARTICLE 13; provided, however, that the Company may grant Awards with respect to up to five percent (5%) of the number of Shares reserved under Section 4.1 as of the Effective Date without regard to the minimum vesting period set forth in this Section 12.3.
12.4 Dividends and Dividend Equivalent Rights.
(a) Any Participant selected by the Committee may be granted Dividend Equivalent Rights based on the dividends declared on the Shares that are subject to any Restricted Stock Unit or an Other Share-
Based Award that is a Full Value Award, to be credited as of dividend payment dates, during the period between the date the Award is granted and the date the Award vests or is settled, as determined by the Committee and set forth in the applicable Award Agreement. Such Dividend Equivalent Rights shall be converted to cash or additional Shares by such formula and at such time and subject to such limitations as may be determined by the Committee.
(b) To the extent Shares subject to an Award (other than Restricted Stock) are subject to vesting conditions, any Dividend Equivalent Rights relating to such Shares shall either (i) not be paid or credited or (ii) be accumulated and subject to restrictions and risk of forfeiture to the same extent as the underlying Award with respect to which such cash, stock or other property has been distributed. For Shares of Restricted Stock that are subject to vesting, dividends shall be accumulated and subject to any restrictions and risk of forfeiture to which the underlying Restricted Stock is subject.
12.5 Limits on Transfer. No right or interest of a Participant in any Award may be pledged, encumbered, or hypothecated to or in favor of any party other than the Company or an Affiliate, or shall be subject to any lien, obligation, or liability of such Participant to any other party other than the Company or an Affiliate. Except as otherwise provided by the Committee, no Award shall be assigned, transferred, or otherwise disposed of by a Participant other than by will or the laws of descent and distribution.
12.6 Stock Certificates; Book Entry Procedures.
(a) Notwithstanding anything herein to the contrary, the Company shall not be required to issue or deliver any certificates evidencing Shares pursuant to the exercise or vesting, as applicable, of any Award, unless and until the Board has determined, with advice of counsel, that the issuance and delivery of such certificates is in compliance with all Applicable Laws, regulations of governmental authorities and, if applicable, the requirements of any exchange on which the Shares are listed or traded. All certificates evidencing Shares delivered pursuant to the Plan are subject to any stop-transfer orders and other restrictions as the Committee deems necessary or advisable to comply with federal, state, or local securities or other laws, including laws of jurisdictions outside of the U.S., rules and regulations and the rules of any national securities exchange or automated quotation system on which the Shares are listed, quoted, or traded. The Committee may place legends on any certificate evidencing Shares to reference restrictions applicable to the Shares. In addition to the terms and conditions provided herein, the Board may require that a Participant make such reasonable covenants, agreements, and representations as the Board, in its discretion, deems advisable in order to comply with any such laws, regulations, or requirements. The Committee shall have the right to require any Participant to comply with any timing or other restrictions with respect to the settlement or exercise of any Award, including, without limitation, a window-period limitation, as may be imposed in the discretion of the Committee.
(b) Notwithstanding any other provision of the Plan, unless otherwise determined by the Committee or required by any Applicable Laws, rule or regulation, the Company shall not deliver to any Participant certificates evidencing Shares issued in connection with any Award and instead such Shares shall be recorded in the books of the Company (or, as applicable, its transfer agent or stock plan administrator).
12.7 Paperless Administration. In the event that the Company establishes, for itself or using the services of a third party, an automated system for the documentation, granting or exercise of Awards, such as a system using an internet website, intranet or interactive voice response, then the paperless documentation, granting or exercise of Awards by a Participant may be permitted through the use of such an automated system.
ARTICLE 13. CHANGES IN CAPITAL STRUCTURE
13.1 Adjustments.
(a) In the event of any stock dividend, stock split, combination or exchange of shares, merger, consolidation or other distribution (other than normal cash dividends) of Company assets to shareholders, or any other similar event or other change related to a corporate event affecting the Shares or the price of the Shares other than an Equity Restructuring, the Committee shall make such adjustments, if any, as the Committee in its discretion may deem appropriate to reflect such change with respect to (a) the aggregate number and kind of shares that may be issued under the Plan (including, without limitation, adjustments of the limitations in Sections 4.1 and 4.3 hereof); (b) the terms and conditions of any outstanding Awards (including, without limitation, the number and kind of shares that may be issued, or any applicable performance goals or criteria with respect thereto); and (c) the grant or exercise price per Share for any outstanding Awards under the Plan.
(b) In the event of any transaction or event described in Section 13.1(a) hereof or any unusual or infrequently occurring items or nonrecurring transactions or events affecting the Company, any affiliate of the Company, or the financial statements of the Company or any affiliate, or of changes in Applicable Laws, regulations or accounting principles, the Committee, in its sole and absolute discretion, and on such terms and conditions as it deems appropriate, either by the terms of the Award or by action taken prior to the occurrence of such transaction or event and either automatically or upon the Participant’s request, is hereby authorized to take any one or more of the following actions whenever the Committee determines that such action is appropriate in order to prevent dilution or enlargement of the benefits or potential benefits intended to be made available under the Plan or with respect to any Award under the Plan, to facilitate such transactions or events or to give effect to such changes in laws, regulations or principles:
(i) to provide for either (A) termination of any such Award in exchange for an amount of cash, if any, equal to the amount that would have been attained upon the exercise of such Award or realization of the Participant’s rights (and, for the avoidance of doubt, if as of the date of the occurrence of the transaction or event described in this Section 13.1 the Committee determines in good faith that no amount would have been attained upon the exercise of such Award or realization of the Participant’s rights, then such Award may be terminated by the Company without payment) or (B) the replacement of such Award with other rights or property selected by the Committee in its sole discretion;
(ii) to provide that such Award be assumed by the successor or survivor corporation, or a parent or subsidiary thereof, or shall be substituted for by similar options, rights or awards covering the stock of the successor or survivor corporation, or a parent or subsidiary thereof, with appropriate adjustments as to the number and kind of shares and prices;
(iii) to make adjustments in the number and type of Shares (or other securities or property) subject to outstanding Awards, and/or in the terms and conditions of (including the grant or exercise price), and the criteria included in, outstanding options, rights and awards;
(iv) to provide that such Award shall be exercisable or payable or fully vested with respect to all Shares covered thereby, notwithstanding anything to the contrary in the Plan or the applicable Award Agreement; and
(v) to provide that the Award cannot vest, be exercised or become payable after such event.
(c) In connection with the occurrence of any Equity Restructuring, and notwithstanding anything to the contrary in Sections 13.1(a) and 13.1(b) hereof:
(i) the number and type of securities subject to each outstanding Award and the exercise price or grant price thereof, if applicable, shall be equitably adjusted. The adjustments provided under this Section 13.1(c)(i) shall be final and binding on the affected Participant and the Company.
(ii) the Committee shall make such equitable adjustments, if any, as the Committee in its discretion may deem appropriate to reflect such Equity Restructuring with respect to the aggregate number and kind of shares that may be issued under the Plan (including, without limitation, adjustments of the limitations in Sections 4.1 and 4.3 hereof).
13.2 Change in Control.
(a) Notwithstanding Section 13.1 hereof, and provided that any applicable Award Agreement does not expressly preclude the following from applying, if a Change in Control occurs and Awards that vest solely on the Participant’s continued Service are not converted, assumed, substituted or replaced by a successor or survivor corporation, or a parent or subsidiary thereof, then immediately prior to the Change in Control such Awards shall become fully exercisable and all forfeiture restrictions on such Awards shall lapse and, immediately following the consummation of such Change in Control, all such Awards shall terminate and cease to be outstanding.
(b) Notwithstanding Section 13.1 hereof, Awards that vest based on the attainment of performance-based conditions shall be subject to the provisions of the Award Agreement governing the impact of a Change in Control, provided that any such provisions in the Award Agreement shall (i) not permit the vesting of
Awards at a rate that is greater than the actual level of attainment and/or (ii) provide for pro-rated vesting of the Award based on any reduction to the performance period resulting from the Change in Control.
(c) Where Awards are assumed or continued after a Change in Control, the Committee may provide that the vesting of one or more Awards will automatically accelerate upon an involuntary termination of the Participant’s employment or service within a designated period following the effective date of such Change in Control. Any such Award shall accordingly, upon an involuntary termination of the Participant’s employment or service in connection with a Change in Control, become fully exercisable and all forfeiture restrictions on such Award shall lapse.
(d) The portion of any Incentive Stock Option accelerated in connection with a Change in Control shall remain exercisable as an Incentive Stock Option only to the extent the applicable $100,000 limitation is not exceeded. To the extent such U.S. dollar limitation is exceeded, the accelerated portion of such Option shall be exercisable as a Non-Statutory Option under the U.S. federal tax laws.
13.3 No Other Rights. Except as expressly provided in the Plan, no Participant shall have any rights by reason of any subdivision or consolidation of Shares of any class, the payment of any dividend, any increase or decrease in the number of Shares of any class or any dissolution, liquidation, merger, or consolidation of the Company or any other corporation. Except as expressly provided in the Plan or pursuant to action of the Committee under the Plan, no issuance by the Company of Shares of any class, or securities convertible into Shares of any class, shall affect, and no adjustment by reason thereof shall be made with respect to, the number of Shares subject to an Award or the grant or the exercise price of any Award.
ARTICLE 14. EFFECTIVE AND EXPIRATION DATE
14.1 Plan Effective Date. The Plan was approved by the Board on February 20, 2018 and shall become effective upon approval of the shareholders of the Company.
14.2 Expiration Date. The Plan will continue in effect until it is terminated by the Board pursuant to Section 15.1 hereof, except that no Incentive Stock Options may be granted under the Plan after February 20, 2028. Any Awards that are outstanding on the date the Plan terminates shall remain in force according to the terms of the Plan and the applicable Award Agreement.
ARTICLE 15. AMENDMENT, MODIFICATION, AND TERMINATION
15.1 Amendment, Modification, and Termination. Subject to Section 16.14 hereof, with the approval of the Board, at any time and from time to time, the Committee may terminate, amend or modify the Plan; provided, however, that to the extent necessary and desirable to comply with any Applicable Laws, the Company shall obtain shareholder approval of any Plan amendment in such a manner and to such a degree as required. Notwithstanding any provision in this Plan to the contrary, absent approval of the shareholders of the Company, and except as permitted by Article 13, no Option or SAR may be amended to reduce the per-Share exercise price of the Shares subject to such Option or SAR below the per-Share exercise price as of the date the Option or SAR is granted and (a) no Option or SAR may be granted in exchange for, or in connection with, the cancellation, surrender or substitution of an Option or SAR having a higher per-Share exercise price and (b) no Option or SAR may be cancelled in exchange for, or in connection with, the payment of a cash amount or another Award at a time when the Option or SAR has a per-Share exercise price that is higher than the Fair Market Value of a Share.
15.2 Awards Previously Granted. Except with respect to amendments made or other actions taken pursuant to Section 16.14 hereof or any amendment or other action with respect to an outstanding Award that may be required or desirable to facilitate compliance with Applicable Laws, as determined by the Committee in its sole discretion, no termination, amendment, or modification of the Plan shall affect adversely, in any material way, any Award previously granted pursuant to the Plan without the prior written consent of the Participant; provided, however, that an amendment or modification that may cause an Incentive Stock Option to become a Non-Qualified Stock Option shall not be treated as adversely affecting the rights of the Participant.
ARTICLE 16. GENERAL PROVISIONS
16.1 No Rights to Awards. No Eligible Individual or other person shall have any claim to be granted any Award pursuant to the Plan, and neither the Company nor the Committee is obligated to treat Eligible Individuals, Participants or any other persons uniformly.
16.2 No Shareholders Rights. Except as otherwise provided herein, a Participant shall have none of the rights of a shareholder with respect to Shares covered by any Award, including the right to vote or receive dividends, until the Participant becomes the record owner of such Shares, notwithstanding the exercise of an Option or SAR or vesting of another Award.
16.3 Tax-Related Items. The Company or any Affiliate, as applicable, shall have the authority to require a Participant to remit to the Company or an Affiliate, an amount sufficient to satisfy the withholding obligations for Tax-Related Items or to take such other action as may be necessary or appropriate in the opinion of the Company or an Affiliate, as applicable, to satisfy withholding obligations for Tax-Related Items, including one or a combination of the following: (a) withholding from the Participant’s wages or other cash compensation payable to the Participant by the Company or an Affiliate; (b) withholding from the proceeds of the sale of Shares acquired pursuant to an Award, either through a voluntary sale or a mandatory sale arranged by the Company on the Participant’s behalf, without need of further authorization; or (c) in the Committee’s sole discretion, by withholding Shares otherwise issuable under an Award (or allowing the return of Shares) sufficient, as determined by the Committee in its sole discretion, to satisfy such Tax-Related Items. No Shares shall be delivered pursuant to an Award to any Participant or other person until the Participant or such other person has made arrangements acceptable to the Committee to satisfy the withholding obligations for Tax-Related Items.
16.4 No Right to Employment or Services. Nothing in the Plan or any Award Agreement shall interfere with or limit in any way the right of the Company or any Affiliate to terminate any Participant’s Service at any time, nor confer upon any Participant any right to continue in the Service of the Company or any Affiliate.
16.5 Unfunded Status of Awards. The Plan is intended to be an “unfunded” plan for incentive compensation. With respect to any payments not yet made to a Participant pursuant to an Award, nothing contained in the Plan or any Award Agreement shall give the Participant any rights that are greater than those of a general creditor of the Company or any Affiliate.
16.6 Indemnification. To the extent allowable pursuant to Applicable Laws, each member of the Committee and the Board shall be indemnified and held harmless by the Company from any loss, cost, liability, or expense that may be imposed upon or reasonably incurred by such member in connection with or resulting from any claim, action, suit, or proceeding to which he or she may be a party or in which he or she may be involved by reason of any action or failure to act pursuant to the Plan and against and from any and all amounts paid by him or her in satisfaction of judgment in such action, suit, or proceeding against him or her; provided he or she gives the Company an opportunity, at its own expense, to handle and defend the same before he or she undertakes to handle and defend it on his or her own behalf. The foregoing right of indemnification shall not be exclusive of any other rights of indemnification to which such persons may be entitled pursuant to the Company’s Certificate of Incorporation or Bylaws, as a matter of law, or otherwise, or any power that the Company may have to indemnify them or hold them harmless.
16.7 Relationship to other Benefits. No payment pursuant to the Plan shall be taken into account in determining any benefits pursuant to any pension, retirement, savings, profit sharing, group insurance, termination programs and/or indemnities or severance payments, welfare or other benefit plan of the Company or any Affiliate, except to the extent otherwise expressly provided in writing in such other plan or an agreement thereunder.
16.8 Expenses. The expenses of administering the Plan shall be borne by the Company and/or its Affiliates.
16.9 Titles and Headings. The titles and headings of the sections in the Plan are for convenience of reference only and, in the event of any conflict, the text of the Plan, rather than such titles or headings, shall control.
16.10 Fractional Shares. No fractional Shares shall be issued and the Committee shall determine, in its discretion, whether cash shall be given in lieu of fractional shares or whether such fractional shares shall be eliminated by rounding up or down as appropriate.
16.11 Limitations Applicable to Section 16 Persons. Notwithstanding any other provision of the Plan, the Plan, and any Award granted or awarded to any Participant who is then subject to Section 16 of the Exchange Act, shall be subject to any additional limitations set forth in any applicable exemptive rule under Section 16 of the Exchange Act (including any amendment to Rule 16b-3 under the Exchange Act) that are requirements for the application of such exemptive rule. To the extent permitted by Applicable Laws, the Plan and Awards granted or awarded hereunder shall be deemed amended to the extent necessary to conform to such applicable exemptive rule.
16.12 Government and Other Regulations. The obligation of the Company to make payment of awards in Shares or otherwise shall be subject to all Applicable Laws, and to such approvals by government agencies, including government agencies in jurisdictions outside of the U.S., in each case as may be required or as the Company deems necessary or advisable. Without limiting the foregoing, the Company shall have no obligation to issue or deliver evidence of title for Shares subject to Awards granted hereunder prior to: (i) obtaining any approvals from governmental agencies that the Company determines are necessary or advisable, and (ii) completion of any registration or other qualification with respect to the Shares under any Applicable Laws in the U.S. or in a jurisdiction outside of the U.S. or ruling of any governmental body that the Company determines to be necessary or advisable or at a time when any such registration or qualification is not current, has been suspended or otherwise has ceased to be effective. The inability or impracticability of the Company to obtain or maintain authority from any regulatory body having jurisdiction, which authority is deemed by the Company’s counsel to be necessary to the lawful issuance and sale of any Shares hereunder, shall relieve the Company of any liability in respect of the failure to issue or sell such Shares as to which such requisite authority shall not have been obtained and shall constitute circumstances in which the Committee may determine to amend or cancel Awards pertaining to such Shares, with or without consideration to the affected Participant. The Company shall be under no obligation to register, pursuant to the Securities Act or otherwise, any offering of Shares issuable under the Plan. If, in certain circumstances, the Shares paid pursuant to the Plan may be exempt from registration pursuant to the Securities Act, the Company may restrict the transfer of such Shares in such manner as it deems advisable to ensure the availability of any such exemption.
16.13 Governing Law. The Plan and all Award Agreements shall be construed in accordance with and governed by the laws of the State of Indiana.
16.14 Section 409A. Except as provided in Section 16.15 hereof, to the extent that the Committee determines that any Award granted under the Plan is subject to Section 409A of the Code, the Award Agreement evidencing such Award shall incorporate the terms and conditions required by Section 409A of the Code. To the extent applicable, the Plan and Award Agreements shall be interpreted in accordance with Section 409A of the Code and U.S. Department of Treasury regulations and other interpretive guidance issued thereunder, including without limitation any such regulations or other guidance that may be issued after the Effective Date. Notwithstanding any provision of the Plan to the contrary, in the event that following the date an Award is granted the Committee determines that the Award may be subject to Section 409A of the Code and related U.S. Department of Treasury guidance (including such guidance as may be issued after the Effective Date), the Committee may adopt such amendments to the Plan and the applicable Award Agreement or adopt other policies and procedures (including amendments, policies and procedures with retroactive effect), or take any other actions, including amendments or actions that would result in a reduction to the benefits payable under an Award, in each case, without the consent of the Participant, that the Committee determines are necessary or appropriate to (a) exempt the Award from Section 409A of the Code and/or preserve the intended tax treatment of the benefits provided with respect to the Award, or (b) comply with the requirements of Section 409A of the Code and related U.S. Department of Treasury guidance and thereby avoid the application of any penalty taxes under such Section or mitigate any additional tax, interest and/or penalties or other adverse tax consequences that may apply under Section 409A of the Code if compliance is not practical.
16.15 No Representations or Covenants with respect to Tax Qualification. Although the Company may endeavor to (a) qualify an Award for favorable or specific tax treatment under the laws of the U.S. (e.g., Incentive Stock Options under Section 422 of the Code) or jurisdictions outside of the U.S. or (b) avoid adverse tax treatment (e.g., under Section 409A of the Code), the Company makes no representation to that effect and expressly disavows any covenant to maintain favorable or avoid unfavorable tax treatment, notwithstanding anything to the contrary in this Plan, including Section 16.14 hereof. The Company shall be unconstrained in its corporate activities without regard to the potential negative tax impact on Participants under the Plan. Nothing in this Plan or in an Award Agreement shall provide a basis for any person to take any action against the Company or any Affiliate based on matters covered by Section 409A of the Code, including the tax treatment of any Awards, and neither the Company nor any Affiliate will have any liability under any circumstances to the Participant or any other party if the Award that is intended to be exempt from, or compliant with, Section 409A of the Code, is not so exempt or compliant or for any action taken by the Committee with respect thereto.
16.16 Clawback/Recovery. All Awards granted under the Plan will be subject to recoupment in accordance with any clawback policy adopted by the Company providing for the recovery of Awards, shares, proceeds, or payments to Participants in the event of fraud or as required by Applicable Laws or governance considerations or in other similar circumstances.
16.17 Severability. If any provision of the Plan or the application of any provision hereof to any person or circumstance is held to be invalid or unenforceable, the remainder of the Plan and the application of such provision to any other person or circumstance shall not be affected, and the provisions so held to be unenforceable shall be reformed to the extent (and only to the extent) necessary to make it enforceable and valid.
* * * *
Annual Meeting Admission Ticket
Eli Lilly and Company 2018 Annual Meeting of Shareholders
Monday, May 7, 2018
11:00 a.m. EDT
Lilly Center Auditorium
Lilly Corporate Center
Indianapolis, Indiana 46285
The top portion of this page will be required for admission to the meeting.
Please write your name and address in the space provided below and present this ticket when you enter the Lilly Center.
Doors open at 10:15 a.m.
|
|
Name |
|
Address |
|
City, State, and Zip Code |
Parking Pass
Directions and Parking
From I-70 take Exit 79B; follow signs to McCarty Street. Turn right (east) on McCarty Street; go straight into Lilly Corporate Center. You will be directed to parking. Be sure to take the admission ticket (the top portion of this page) with you to the meeting and leave this parking pass on your dashboard.
Take the top portion of this page with you to the meeting.
Eli Lilly and Company
Annual Meeting of Shareholders
May 7, 2018
Please place this identifier on the dashboard of your car as you enter Lilly Corporate Center so it can be clearly seen by security and parking personnel.