| | | | | | mg treatment group was 20.83%, 21.74% and 16.67%, respectively, with no statistical significance.
|
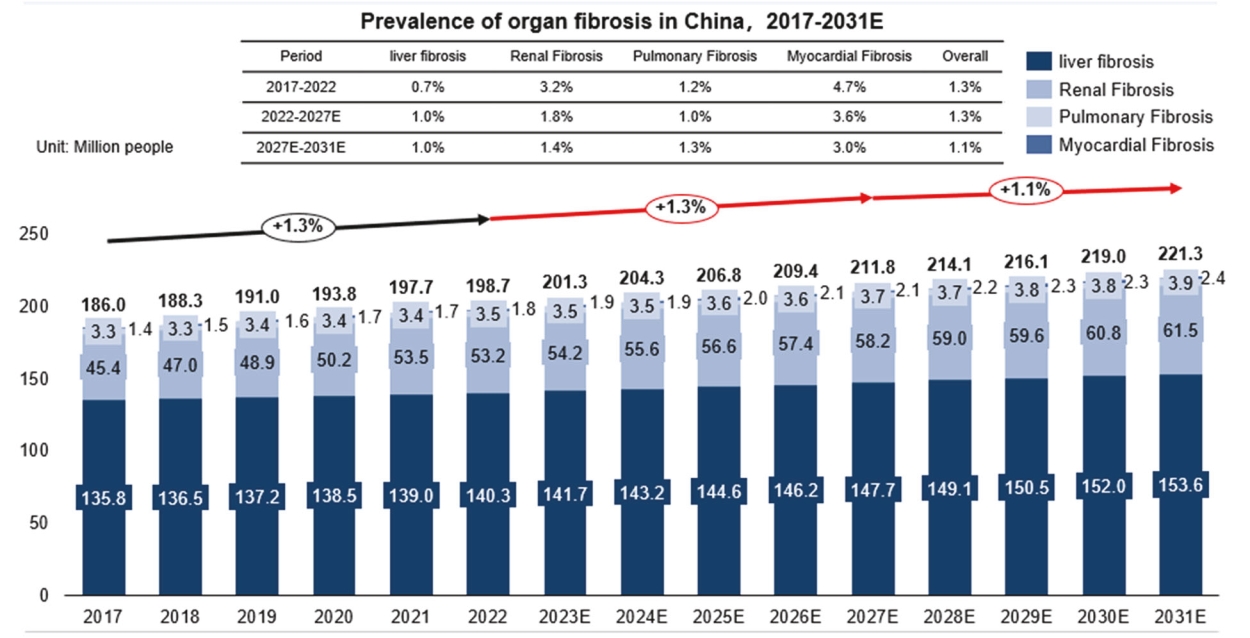
17 patients experienced SAEs, but none were drug-related. The incidenceAccording to Frost & Sullivan, the prevalence of adverse drug reactions (“ADRs”)IPF in the placebo group, 400 mgPRC increased from 83,002 patients in 2017 to 131,654 patients in 2022 at a CAGR of 9.7%, and 600 mg treatment groups was 41.67%, 29.17%it is expected to increase to 214,664 patients by 2027 at a CAGR of 10.3% from 2022 to 2027 and 45.83%, respectively, with no statistical difference between the three groups.to 320,677 patients by 2031 at a CAGR of 10.6% from 2027 to 2031. The incidencetotal market size of rashIPF in the PRC increased from $13.6 million in 2017 to $127.4 in 2022 at a CAGR of 56.3%, and is expected to reach $344.9 million by 2027 at a CAGR of 22.0% from 2022 to 2027 and $698.6 million by 2031 at a CAGR of 19.3% from 2027 to 2031.
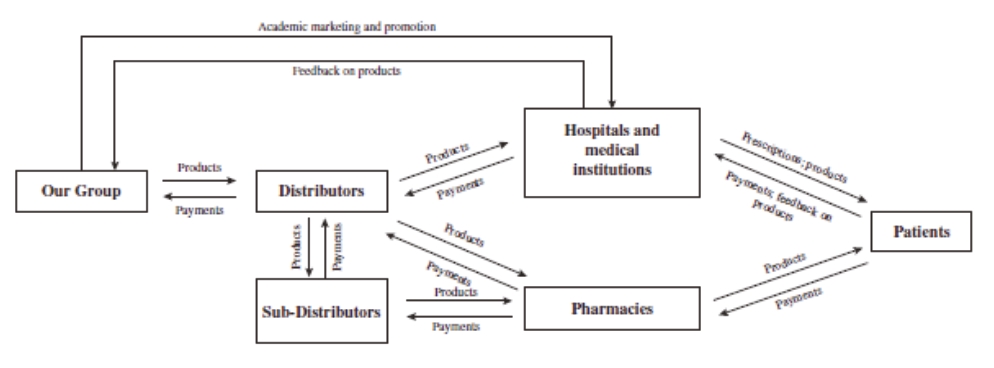
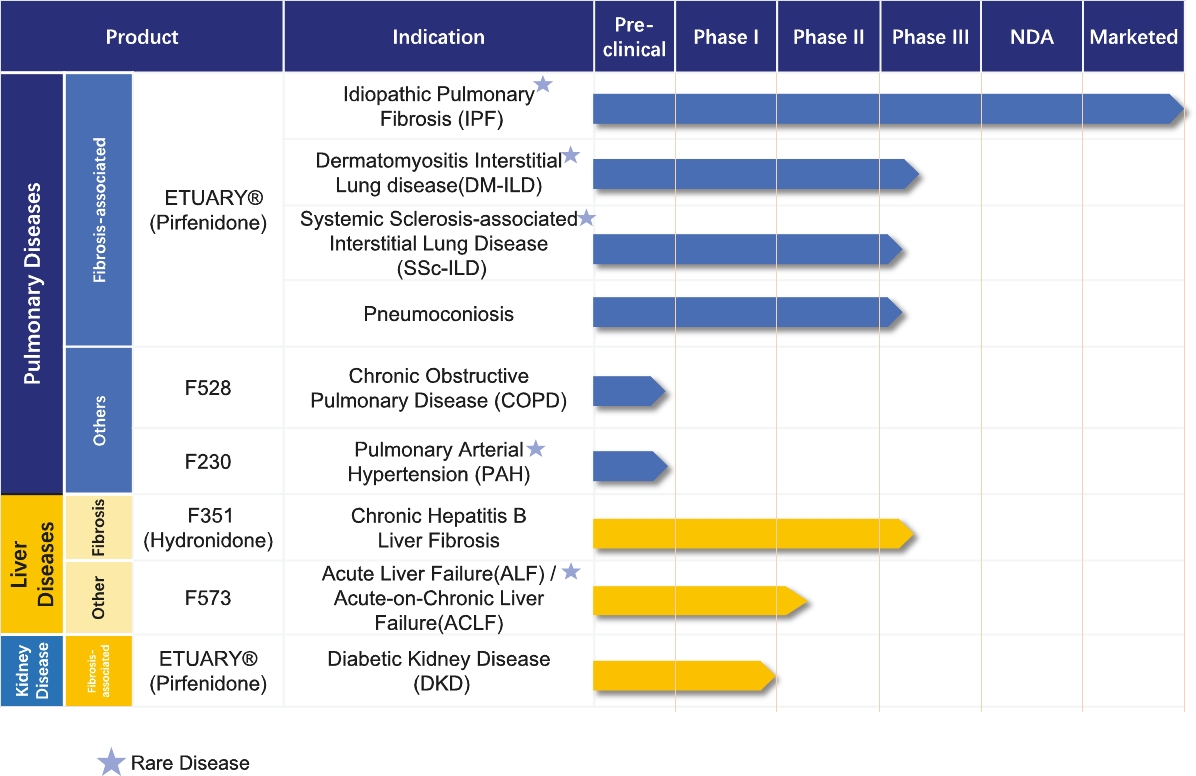
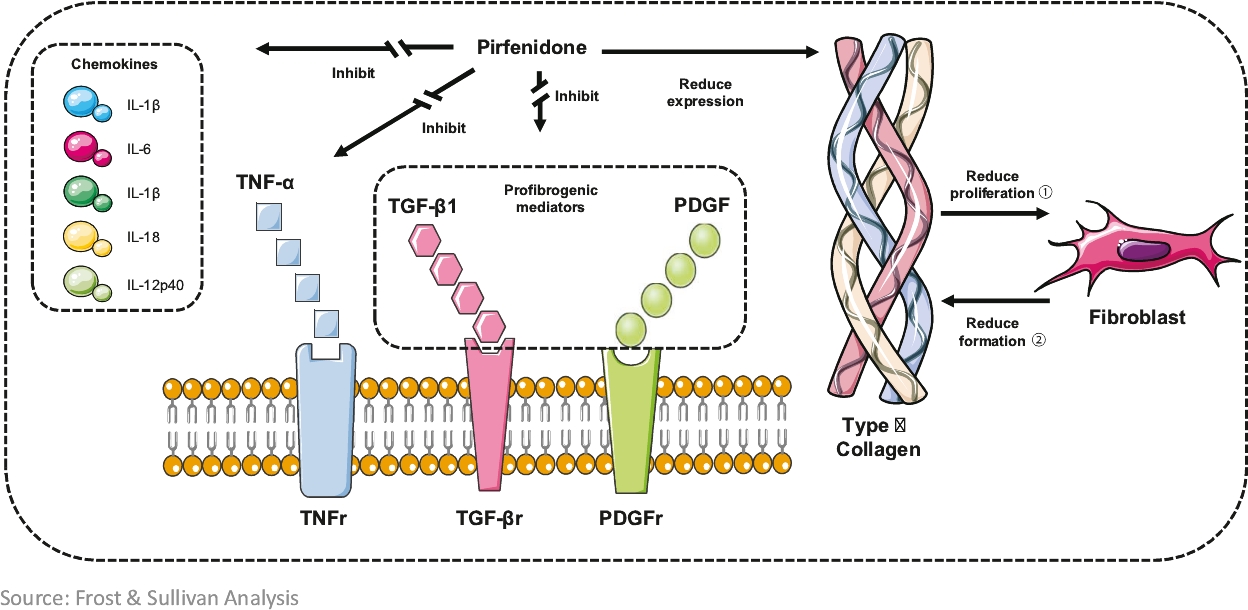
The scarring of lung tissues is irreversible. However, proper treatment groups was statistically different from thatmay slow the rate of fibrosis, increase the patient’s survival rate, alleviate the patient’s symptoms and improve the patient’s quality of life. There are currently two types of IPF drugs approved in the placebo groupPRC: pirfenidone and was present in allnintedanib. They are both clinically shown to slow down the formation of the 600 mg treatment groups with an incidence of 20.83%. The common ADRs included nausea (12.5% in each of the 400 mg treatment group and 600 mg treatment group), photosensitivity (4.17%scar tissue in the 400 mg treatment grouplungs of IPF patients and 12.5% inare the 600 mg treatment group) and drowsiness (8.33% in the 600 mg treatment group), but these were not statistically significant from the placebo group. The incidence of AEs in the placebo group, 400 mg and 600 mg treatment groups was 70.83%, 66.67% and 66.67%, respectively, with no statistical difference between the three groups. The average incidence of significant adverse events in each of the placebo group and the 400 mg and 600 mg treatment groups was 54.17%, with no statistical difference between the three groups. The incidence of SAEs (including mortality and hospitalization) in each of the placebo group and the 400 mg and 600 mg treatment groups was 29.17%, 20.83% and 20.83%, with no statistical difference between the three groups.
After 12 months of treatment, pirfenidone wasonly drugs that are considered effective in slowing down the decline in DLco%, DLco, SaO2 and SpO2 immediately after 6MWT. No drug-related SAEs were observed and rash and nausea were the most common ADRs. The results show that pirfenidone has potential for the treatment of IPF.
Phase 3 clinical trial of pirfenidoneorgan fibrosis in the PRC. According to the latest guideline for the treatment of SSc-ILDIPF issued by the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society and Latin American Thoracic Association (“ATS/ERS/JRS/ALAT”), pirfenidone and nintedanib are the only two types of IPF drug conditionally recommended with moderate-quality evidence.
We are conducting a randomized, double-blind, placebo-controlled, multicenter Phase 3 clinical trial. The purposePirfenidone has been clinically shown to slow down the development of this registration trial is to evaluate the efficacy and safety of pirfenidonescar tissues in the treatmentlungs of SSc-ILD. The primary endpoint isIPF patients. Based on the change in FVC% at 52 weeks of treatment compared to baseline. 144 patients are planned to be enrolled invast clinical needs for pirfenidone, ETUARY was approved as the trial, with 108 in the treatment group and 36 in the control group.
This trial enrolled thePRC’s first patient in June 2018. Due to the outbreak of COVID-19 and the scarcity of eligible patients, this trial is still in the process of recruiting patients and therefore no clinical results are currently available for analysis.
Phase 3 clinical trial of pirfenidoneNational Class 1.1 New Drug for the treatment of DM-ILDmild to moderate IPF.
We are conductingSince its commercialization, ETUARY has remained a randomized, double-blind, placebo-controlled, multicenter Phase 3 clinical trial. The purposedominant player in the IPF drug market, with a market share of this registration trial isover 90% in 2020 and over 70% in 2021. Sales of ETUARY have continued to evaluategrow rapidly, increasing from $63.3 million in 2020 to $86.8 million in 2021 to $99.3 million in 2022 and we expect our sales growth to continue. In 2021, the efficacymarket share of ETUARY decreased to 78.8%, primarily due to recent inclusion of competitors’ products into NRDL (such as Ofev by Boehringer Ingelheim) and safetythe related increased sales of pirfenidonesuch competitors’ products. In 2022, the market share of ETUARY decreased to 55.3%, primarily due to the market share of new drugs rising rapidly as a result of new drugs having different shelf times and targeting different indications. Despite the recent decrease of market share of ETUARY in the IPF market, we continue to expect a strong sales performance due to: (i) sustained increases in the prevalence of IPF; and (ii) future indication expansion of ETUARY for the potential treatment of DM-ILD. The primary endpointDM-ILD, SSc-ILD, pneumoconiosis, and DKD. In addition, there are various barriers to entry for the potential market entrants. For instance, it is the change in FVC% at 52 weeks of treatment compareddifficult for new entrants to baseline. 152 patients will be enrolledbuild an experienced and specialized sales and marketing team in the trial,short term given that sales and marketing strategies of organ fibrosis drugs significantly differ from that of etiological treatment drugs and long-term and stable collaboration with 114KOLs and hospitals is critical to developing and optimizing product portfolio, effectively educating and penetrating the market and recruiting patients for clinical trials.
SSc-ILD and DM-ILD
Connective tissue disease associated with interstitial lung disease (“CTD-ILD”) is non-idiopathic interstitial pneumonia. CTD is a type of autoimmune disease that causes damage to various organs throughout the body based on chronic inflammation of blood vessels and connective tissue. ILD is one of the most serious pulmonary complications and can result in significant morbidity and mortality when associated with CTD.
SSc is a CTD characterized by degenerative microvascular phenomena and immune system activation, leading to fibrosis of the skin and internal organs. ILD is very frequent in patients affected by SSc, reaching about 50% prevalence, representing the leading SSc-related cause of death. DM is characterized by proximal skeletal muscle weakness and muscle inflammation. Among patients with DM, ILD is a major cause of morbidity and mortality. The frequency of ILD in DM has been reported to range between 5% and 45% depending on the diagnostic method.
According to Frost & Sullivan, the prevalence of CTD-ILD in the treatment groupPRC increased from approximately 2.3 million patients in 2017 to 2.4 million patients in 2022, and 38is expected to reach 2.5 million patients in 2027 and 2.6 million patients in 2031. Among the control group.CTD-ILD patients, approximately 8.4% are SSc-ILD and DM-ILD patients in 2022. The market size of anti-fibrosis drugs for SSc-ILD/DM-ILD patients was $9.1 million in 2022 and is expected to reach $53.1 million by 2027 and $117.6 million by 2031 at a CAGR of 42.4% from 2022 to 2027 and a CAGR of 22.0% from 2027 to 2031.
This trial enrolled the first patient in June 2018. Due to the outbreak of COVID-19SSc-ILD and the scarcity of eligible patients, this trial is still in the process of recruiting patientsDM-ILD are induced by known factors, including specific exposure or autoimmune diseases (such as scleroderma and no clinical results are currently available for analysis.
Phase 3 clinical trial of pirfenidone for therheumatoid arthritis). Symptoms include chronic cough, expectoration, hemoptysis, progressive dyspnea and intermittent fever. The treatment of pneumoconiosis
We are conductingCTD-ILD (including SSc-ILD and DM-ILD) is a randomized, double-blind, placebo-controlled, multi-center Phase 3 clinical trial. The purposecombination of this registration trial is to evaluate the efficacy and safety of pirfenidone in the treatment of pneumoconiosis. The primary endpoint is the change in force vital capacity at 52 weeks of treatment compared to baseline. 272 patients will be enrolled in the trial, with 136 in the treatment group and 136 in the control group.
We obtained ethics committee approval as of January 2022 and enrolled the first patient in June 2022.
Phase 1 clinical trial of pirfenidone for the treatment of DKD
We are conducting an open-label, parallel-controlled, single-center clinical trial. The purpose of this registration trial is to evaluate the safety and PK of a single dose of pirfenidone capsules in patients with CKD stages G2 and G3a. 24 subjects were enrolled, consisting of 12 patients with renal insufficiency and 12 healthy volunteers.
The Phase 1 clinical trial was completed in March 2022. In this trial, pirfenidone was tolerated when used in patients with chronic kidney disease G2 and G3a, there is no significant change in the main pharmacokinetic parameters compared with healthy controls, and no dose adjustment is required.






 ”. As of March 31, 2023, we owned four registered artwork copyrights, 13 registered software copyrights and 34 registered trademarks in the PRC. We also owned seven registered trademarks in Hong Kong, one international trademark of “ETUARY” and the trademark application of “ETUARY” in seven countries and regions including the United States, EU and Japan. As of the same date, we are also the registered owner of 13 domain names.
”. As of March 31, 2023, we owned four registered artwork copyrights, 13 registered software copyrights and 34 registered trademarks in the PRC. We also owned seven registered trademarks in Hong Kong, one international trademark of “ETUARY” and the trademark application of “ETUARY” in seven countries and regions including the United States, EU and Japan. As of the same date, we are also the registered owner of 13 domain names.