NOVEMBER 14, 2023

Up
Debt Securities
Common Stock
Preferred Stock
Warrants
Subscription Rights
Purchase Contracts
Depositary Shares
Units
This prospectus relates to the resale of 10,947,371 shares of Common Stock,time, either separately or together in one or more offerings, our debt securities, common stock with a par value of $0.001 per share (“Common Stock”) of Rezolute, Inc. (the “Company”) by Federated Hermes Kaufmann Small Cap Fund, Federated Hermes Kaufmann Fund, Federated Hermes Kaufmann Fund II, CDK Associates, L.L.C., Third Street Holdings LLC, Blackstone Annex Master Fund L.P., Vivo Opportunity Fund Holdings, L.P., and Adage Capital Partners LP (the “Selling Stockholders”). The
Our registration of the securities covered by this prospectus does not mean that the Selling Stockholders will offer or sell any of the shares of Common Stock. The Selling Stockholders may sell the shares of Common Stock offered by this prospectussecurities from time to time in amounts, at prices and on other terms to be determined at the time of sale through ordinary brokerage transactionsoffering. We may offer and sell the securities to or through one or more underwriters, dealers or agents, or directly to purchasers, on a continuous or delayed basis. If any other means describedunderwriters, dealers or agents are involved in the sale of any of the securities, their names, and any applicable purchase price, fee, commission or discount arrangement between or among us and them will be set forth, or will be calculable from the information set forth, in any applicable prospectus supplement. See the sections entitled “About this prospectus under the captionProspectus” and “Plan of Distribution.” TheDistribution” for more information.
Our Common Stock is listed on The Nasdaq Capital Market underon September 13, 2023 (a date within 60 days of the symbol “RZLT”. On June 14, 2022,date hereof), calculated in accordance with General Instruction I.B.6 of Form S-3. During the last reported sale price for12 calendar months prior to and including the date of this prospectus, we have not offered and sold any of our Common Stock was $3.02 per share. securities pursuant to General Instruction I.B.6 of Form S-3.
THESE
| | | Page | | ||||
| | | | | | | ||
| | | | | | | ||
| | | | | | | ||
| | | | | | | ||
| | | | | | | ||
| | | | | 11 | | ||
| | | | | | | ||
| | | | | 20 | | | |
| | | | | 26 | | ||
| | | | | 27 | | | |
| | | | | | | ||
| | | | | | | ||
| | | | | | | ||
| | | | | 37 | | | |
| | | | | 37 | | | |
Neither we nor the Selling Stockholders have done anything that would permit
This prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entiretycovered by the actual documents. Copies of some of the documents referred to herein have been filed, will be filedprospectus supplement. The prospectus supplement may also add, update or will bechange information contained or incorporated by reference asin this prospectus. If there is any inconsistency between the information in this prospectus and any prospectus supplement, you should rely on the information in the prospectus supplement.
Unless the context indicates otherwise, as used in thissecurities unless accompanied by a prospectus the terms “Rezolute,” “we,” “us,” “our,” and “our business” refer to Rezolute, Inc. and its subsidiaries.
Our Pipeline
and orphan diseases.
The RIZE study was conducted primarily in a young pediatric population, average ~6.5 years of age and enrolled 23 patients across diverse genetic types. RZ358 resulted in a > 50% improvement in hypoglycemic events across all doses and approximately 75% improvement at the mid (6 mg/kg) and top (9 mg/kg) doses. Time-in-range by continuous glucose monitoring (“CGM”) improved by 8% across all doses and 16% at the top dose. Expected RZ358 concentrations were achieved and clear dose-exposure responses were observed. There were no adverse drug reactions, dose-limiting toxicities, or drug-related serious adverse events. We believe that these positive results from the RIZE study will be Phase-3 enabling and we plan to interact with the regulatory authorities in the second half of calendar year 2022. If we obtain clearance, we will initiate our Phase 3 study in the first half of calendar year 2023.
In addition, during the first half of calendar year 2020 we had positive interactions with the United States Food and Drug Administration (“FDA”) whereby we were granted Rare Pediatric Disease (“RPD”) designation for RZ358, which qualified us to receive a priority review voucher (“PRV”) upon marketing approval of the drug in congenital HI. Such a voucher could be redeemed to receive a priority review of a subsequent marketing application for any drug candidate in any disease indication.
Our second clinical asset, RZ402, is a selective and potent plasma kallikrein inhibitor (“PKI”) being developed as a potential oral therapy for the chronic treatment of diabetic macular edema (“DME”). RZ402 recently completed the Phase 1 development program. In January 2021, we dosed the first subject in the Phase 1a study, and in May 2021 we announced positive topline results whereby single dose oral administration of RZ402 resulted in plasma concentrations that substantially exceeded target pharmacologically active drug levels, demonstrating the potential for once daily dosing. RZ402 was generally safe and well-tolerated at all doses tested, without dose-limiting toxicities. In August 2021, we announced the initiation in the Phase 1b multiple-ascending dose (“MAD”) study and reported positive results in March 2022. The results further validated and supported the potential for once daily oral dosing and showed dose-dependent increases in systemic exposures, with repeat-dosing to steady-state resulting in the highest concentrations of RZ402 explored to date, exceeding 200 ng/mL and 50 ng/mL at peak and 24-hour trough, respectively. The MAD study results showed that RZ402 was generally safe and well-tolerated, including at higher doses than previously tested in the SAD study. There were no serious adverse events, adverse drug reactions or identified risks. We are advancing developmental activities toward a Phase 2a proof-of-concept study, which we plan to initiate during the second half of calendar year 2022.
RZ358
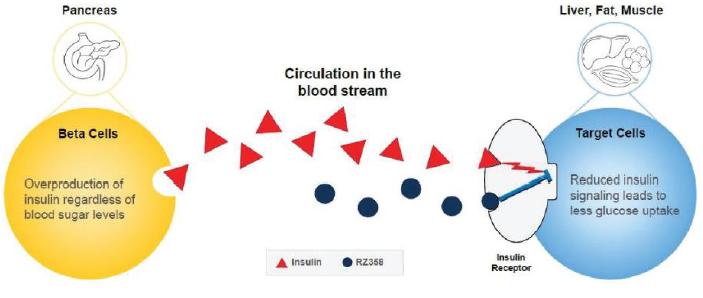
Congenital HI is an ultra-rare pediatric genetic disorder characterized by excessive production of insulin by the pancreas. If untreated, the elevated insulin levels in these patients suffering with congenital HI can induce extreme hypoglycemia (low blood sugar) events, increasing the risk of neurological and developmental complications, including persistent feeding problems, learning disabilities, recurrent seizures, brain damage or even death. There are no FDA approved therapies for all forms of congenital HI and the current standard of care treatments are suboptimal. The current treatments used by physicians includes Glucagon, Diazoxide, Somatostatin Analoguesinclude glucagon, diazoxide, somatostatin analogues and Pancreatectomy. Each of these treatments has the following drawbacks as set forth in the graphic set below.

Our lead candidate, RZ358 is an intravenously administered human monoclonal antibody that binds to a unique site (allosteric) on the insulin receptor throughout the body,in insulin target tissues, such as in the liver, fat, and muscle. The antibody modifiesdown modulates insulin’s binding, signaling, and signalingaction to maintain glucose levels in a normal range which counteractsthereby counteracting the effects of elevated insulin in the body. RZ358 shows dose dependent pharmacokinetics with a half- life greater than two weeks which has the potential for twice or even once monthly dosing. Therefore, we believe that RZ358 is ideally suited as a potential therapy for conditions characterized by excessive insulin levels, and it is being developed to treat the hyperinsulinism and low blood sugar characteristic of diseases such as congenital HI.sugar. As RZ358 acts downstream from the beta cells, it has the potential to be universally effective at treating congenital HI caused by any of the underlying genetic defects.
A summary
Phase 1 pharmaco-kinetic studies of single intravenous doses of RZ358 at 0.1 to 9 mg/kg in healthy volunteers revealed dose-dependent pharmacokinetics with a half-life of 15 days, supporting the biweekly dosing approach. In healthy volunteers, RZ358 prevented hypoglycemia induced by insulin administration, without producing hyperglycemia. This effect showed a pharmacokinetic-pharmacodynamic (dose - response) correlation, with the hypoglycemia-blunting effects of RZ358 lasting for two weeks.
The clinical proof-of-conceptrandomized, double-blind, placebo-controlled, parallel arm evaluation of RZ358 in congenital HI was evaluated in Phase 2a studies in a total of 14 patients with congenital HI (12 years or older). The studies investigated the pharmacokinetics (PK), pharmacodynamics (PD), safety and preliminary efficacy of RZ358. RZ358 was well-tolerated in adult and pediatric patientsparticipants with congenital HI who received single intravenous doses in the phase 2a studies and the PKare not adequately responding to standard of care medical therapies. Topline results from the phase 2a studies were consistent with thosestudy are anticipated to be available in healthy volunteers. There was a durable dose- and disease-dependent normalizationthe first half of blood sugar in patients with congenital HI who had elevated insulin levels/low blood glucose levels at baseline, with an approximate 50% improvement in hypoglycemia and near normalization of glucose control, which was sustained for more than two weeks after dosing. RZ358 did not increase blood sugar levels in patients with normal blood sugar levels at baseline. The increases in blood sugar, depended on and correlated with disease severity and RZ358 concentrations, making RZ358 uniquely suited as a potential therapy for congenital HI, a disease with known heterogeneity and variability.
2025. The Phase 3 study follows the Company’s multinational Phase 2b (RZ358-606) trial, “RIZE” study was(“
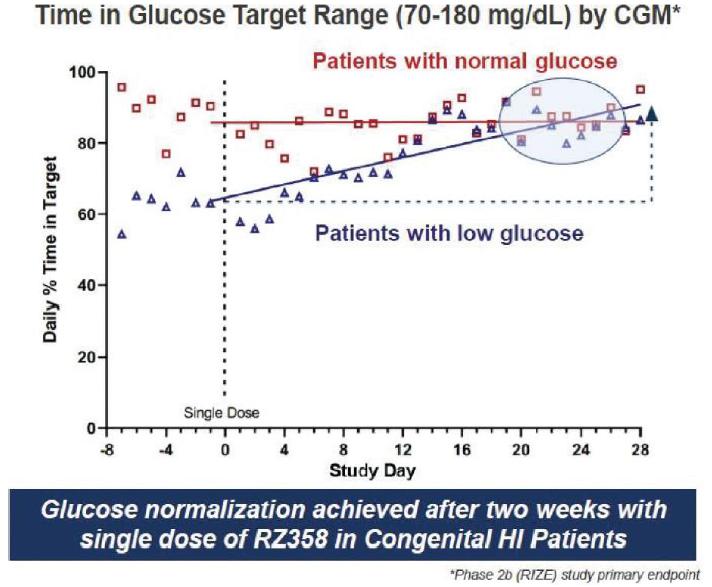
The RIZE study enrolled 23 patients and was primarily in a young pediatric population, average ~6.52 years of age and in a diverse group of patients across gender and genetics. A key entry criterion was for patients to have substantial hypoglycemia to be eligible for enrollment. We observed that patients enrolled on stable background therapies had clinically-significant, and in many cases, substantial residual hypoglycemia and also, some hyperglycemia (> 180 mg/dL) at baseline.
Results from theolder who were failing medical therapies. The RIZE study showeddemonstrated that target and expected RZ358 concentrations were achieved and dose-exposure dependent responses were also observed. RZ358 was generally safe and well-tolerated, as well as highly effective in improving hypoglycemia.
RZ402
. DME is a vascular complication of diabetes and a leading cause of blindness in the U.S. and elsewhere. Chronic exposure to high blood sugar levels can lead to inflammation, cell damage, and the breakdown of blood vessel walls. Specifically, in DME, retinal blood vessels behindat the back of the eye become porous and permeable leading to the unwanted infiltration of fluid into the macula. This fluid leakage creates distorted vision, and if left untreated, could result in blindness.
Results from theDecember 2022, we initiated a Phase 1a Single-Ascending Dose (SAD) Study (RZ402-101) were reported in May 2021. RZ402-101 was a first-in-human single-center,2 multi-center, randomized, double-blind,double-masked, placebo-controlled, SADparallel-arm study in healthy adult volunteers. The study objectives were to characterizeevaluate the safety, profileefficacy, and pharmacokinetics of RZ402 administered as single oral doses.a monotherapy over a twelve week treatment period in participants with DME who are naïve to, or have received limited anti-VEGF injections. The study enrolled 30 subjects inpopulation is comprised of DME patients with mild to moderate non-proliferative diabetic retinopathy. Eligible participants are being randomized equally, to one of three planned sequential dose- level cohortsRZ402 active treatment arms at doses of 2550, 200, and 400 mg, 100 mg, and 250 mg. Within each ten-subject dose cohort, subjects were randomized 8:2or a placebo control arm, to receive either RZ402 oral solution or matched placebo. After receiving single doses, participants remainedstudy drug once daily for twelve weeks, before completing a four-week follow-up. The study is expected to enroll up to approximately 100 patients overall, across approximately 25 investigational sites in the clinic for seven days for serial pharmacokineticUnited States. The principal endpoints of the trial include (i) changes in central subfield thickness of the macula, as measured by Spectral Domain Ocular Coherence Tomography, (ii) changes in visual acuity as measured by the early treatment diabetic retinopathy scale, (iii) the repeat dose pharmacokinetics of RZ402 in patients with DME, and safety assessments, before completing two outpatient follow-up visits at study days 14 and 30. Dose advancement proceeded following blinded reviews of(iv) the safety and pharmacokinetic data from the preceding cohort(s).
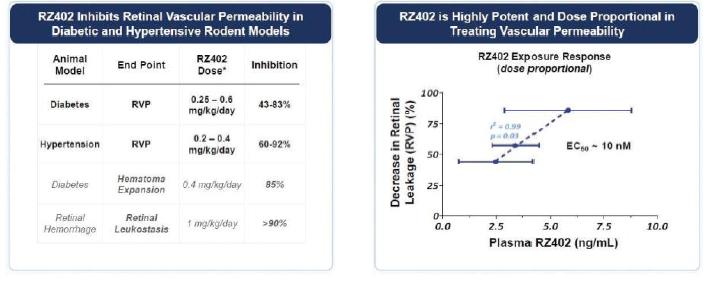
Single doses of RZ402 resulted in dose-dependent increases in systemic exposure. Plasma concentrations of RZ402 significantly exceeded the 3.5 ng/mL target concentration that was pharmacologically active in animal models of DME for a 24-hour period after receipttolerability of RZ402. AcrossWe expect to complete enrollment in 2023 and to provide an update on the dosestudy prior to year end.
Resultsconducted in participants with congenital HI who were twelve years of age and older in countries governed by the regulatory authorities in the European Union and elsewhere in Europe. In the US, the FDA restricted enrollment in Phase 2a to participants eighteen years of age and older and, based on the rat findings, imposed a human drug exposure limit equating to repeat doses of approximately 3 mg/kg per week (“
MAD study showed dose-dependent increases in systemic exposures, with repeat-dosing to steady-state resultingbe available in the highest concentrationsfirst half of RZ402 explored to date, exceeding 200 ng/mL and 50 ng/mL at peak and 24-hour trough, respectively. Following the precedent established in systemic deliveries of PKIs in vascular diseases such as hereditary angioedema, steady-state plasma kallikrein activity in human plasma was measured on Day 14 as a biomarker of RZ402 target engagement. Daily dosing with RZ402 inhibited plasma kallikrein in a dose and concentration-dependent manner (r=0.74; p < 0.001). Given that the in-vivo EC90 for RZ402 in animal models of DME is ~6 ng/mL, the results at both peak and 24-hour trough substantially exceeded target concentrations based on a combination of in-vitro and in-vivo profiling. RZ402 was generally safe and well-tolerated, including at higher doses than previously tested in the SAD study. There were no serious adverse events, adverse drug reactions or identified risks.
2025.
and Zealand Pharma.
Ocuphire Pharma, Oxurion and Verseon.
We
Employees
Employees
All of the shares of Common Stock offered by the Selling Stockholders pursuant to this prospectus will be sold by the Selling Stockholders for its own account. We will not receive any of the proceeds from these sales.
We will receive up to an aggregate of approximately $10,000 from the exercise of the Class B Warrants, assuming the exercise in full of all of the Class B Warrantsdata produced by such contractors in support of our marketing applications;
On May 1, 2022, we entered into an underwriting agreement with Jefferies LLC, as representativeDEBT SECURITIES
The Registered Direct Offering closed on May 4, 2022. The net proceeds of the Registered Direct Offering to the Company, after deducting the underwriting discounts and commissions and offering expenses payable by the Company were approximately $110.5 million.
The Class B Warrants issued in the Registered Direct Offering have an exercise price of $0.001 per share, which is subject to adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting the Common Stock and also upon any distributions for no consideration of assets to the Company's stockholders. In the event of certain corporate transactions,our other securities that the holders of the Class B Warrantsseries of debt securities receive would be subject to adjustment.
Inthe applicable series of debt securities, unless such holders have offered the trustee reasonable indemnity. The holders of a majority in principal amount of the outstanding debt securities of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy available to the trustee, or exercising any trust or power conferred on the trustee, with respect to the debt securities of that series, provided that:
Common Stock
Ourrestated articles of incorporation as(our “
The table below lists the Selling Stockholders and other information regarding the beneficial ownershipaggregate number of the shares of our common stock beneficially owned by eachthe holder (together with its affiliates) to exceed 4.99%, 9.99% or 19.99%, as applicable to the holder, of the Selling Stockholders. The second column lists the number of shares of our common stock outstanding immediately after giving effect to the exercise, or (ii) the combined voting power of our securities beneficially owned by each Selling Stockholder, based on its ownershipthe holder of the shares of common stock and warrants,Class B Pre-Funded Warrant (together with its affiliates) to exceed 4.99%, 9.99% or 19.99%, as of June 14, 2022, assuming exerciseapplicable to the holder, of the warrants held bycombined voting power of all of our securities then outstanding immediately after giving effect to the Selling Stockholders on that date, without regard to any limitations on exercises.
The third column lists the shares of common stock being offered by this prospectus by Selling Stockholders.
Inexercise, as such percentage ownership is determined in accordance with the terms of the Class B Pre-Funded Warrants. However, any holder may increase or decrease such percentage to any other percentage upon at least 61 days’ prior notice from the holder to us; provided, that a registration rights agreement with the Selling Stockholders, this prospectus generally covers the resaleholder of a Class B Pre-Funded Warrants may not increase such percentage to a percentage in excess of 19.99%.The exercise price of the maximumClass B Pre-Funded Warrants and the number of shares of our common stock issuable upon exercise of the Class B Pre-Funded Warrants determinedis subject to appropriate adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and also upon any distributions of assets, including cash, stock or other property to our stockholders. The Class B Pre-Funded Warrants also contain provisions that provide certain rights to holders in the event of a fundamental transaction, including a merger or consolidation with or into another entity, such as if the outstanding warrants were exercisedright to receive the same amount and kind of consideration paid to the holders of our common stock in full asthe fundamental transaction. The Class B Pre-Funded Warrants do not entitle the holders to any voting rights or any of the trading day immediately precedingother rights or privileges to which holders of common stock are entitled.
Underexercise, as such percentage ownership is determined in accordance with the terms of the Class B2021 Pre-Funded Warrants. However, any holder may increase or decrease such percentage to any other percentage upon at least 61 days’ prior notice from the holder to us; provided, that a holder of a 2021 Pre-Funded Warrants a Selling Stockholder may not increase such percentage to a percentage in excess of 19.99%.The exercise price of the warrants to2021 Pre-Funded Warrants and the extent such exercise would cause such Selling Stockholder, together with its affiliates and attribution parties, to beneficially own a number of shares of common stock which would exceed 19.99% (or, at the election of the Selling Stockholder, 4.99% or 9.99%) of our then outstanding common stock following such exercise, excluding for purposes of such determination shares of common stock issuable upon exercise of the warrants which have not been exercised. The number of shares2021 Pre-Funded Warrants is subject to appropriate adjustment in the secondevent of certain stock dividends and third columns,distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and the percentage ownershipalso upon any distributions of assets, including cash, stock or other property to our stockholders. The 2021 Pre-Funded Warrants also contain provisions that provide certain rights to holders in the fourth column, do not reflect this limitation. The Selling Stockholders may sell all, someevent of a fundamental transaction, including a merger or noneconsolidation with or into another entity, such as the right to receive the same amount and kind of their shares in this offering. See "Plan of Distribution."
| Name of Selling Stockholder | Number of Shares of Common Stock Owned Prior to Offering | Maximum Number of Shares of Common Stock to be Sold Pursuant to this Prospectus | Number of Shares of Common Stock Owned After Offering |
Percentage of Shares | ||||||||||||
| Federated Hermes Kaufmann Funds (3) | 10,554,327 | 3,421,053 | 7,133,274 | 21.2 | % | |||||||||||
| CDK Associates, L.L.C. (4) | 2,490,306 | 1,039,739 | 1,450,567 | 4.3 | % | |||||||||||
| Third Street Holdings LLC (5) | 158,956 | 66,366 | 92,590 | * | ||||||||||||
| Blackstone Annex Master Fund L.P. (6) | 2,631,579 | 1,093,895 | 1,537,684 | 4.6 | % | |||||||||||
| Vivo Opportunity Fund Holdings, L.P. (7) | 3,289,473 | 2,800,000 | 489,473 | 1.5 | % | |||||||||||
| Adage Capital Partners LP (8) | 3,158,008 | 2,526,318 | 631,690 | 1.9 | % | |||||||||||
| Total | 22,282,649 | 10,947,371 | 11,335,278 | 33.8 | % | |||||||||||
* Represents ownership of less than 1%.
Each Selling Stockholder of the securities and any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities covered hereby on the principal Trading Market or any other stock exchange, market or trading facility on which the securities are traded or in private transactions. These sales may be at fixed or negotiated prices. A Selling Stockholder may use any one or more of the following methods when selling securities:
The Selling Stockholders may also sell securities under Rule 144 or any other exemption from registration under the Securities Act, if available, rather than under this prospectus.
Broker-dealers engaged by the selling stockholders may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the selling stockholders (or, if any broker-dealer acts as agent for the purchaserholders of our common stock from the purchaser) in amounts to be negotiated, but, except as set forth in a supplement to this prospectus, in the case of an agency transactionfundamental transaction. The 2021 Pre-Funded Warrants do not in excess of a customary brokerage commission in compliance with Financial Industry Regulatory Authority,entitle the holders to any voting rights or FINRA, Rule 5110; and in the case of a principal transaction a markup or markdown in compliance with FINRA Rule 2121.
In connection with the saleany of the securitiesother rights or interests therein,privileges to which holders of common stock are entitled.
The Selling Stockholders and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act. Each Selling Stockholder has informed the Company that it does not have any written or oral agreement or understanding, directly or indirectly, with any person to distribute the securities.
The Company is required to pay certain fees and expenses incurred by the Company incident to the registration of the securities. The Company has agreed to indemnify the Selling Stockholders against certain losses, claims, damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus effective until the date that allpurchase 0.33 shares of Common Stock covered by this propsectus (i) have been sold, thereunderour common stock at an exercise price of $19.50 per share of our common stock. Each Participating Warrant is exercisable on or pursuantafter October 9, 2020 and will expire on or prior to Rule 144, or (ii) may be sold without volume or manner-of-sale restrictions pursuant to Rule 144 and without the requirement for the Company to be in compliance with the current public information requirement under Rule 144, as determined by the counsel to the Company pursuant to a written opinion letter to such effect, addressed and acceptable to the Transfer Agent and the affected holders The Company shall telephonically request effectiveness of a Registration Statement as of 5:00 p.m. (New York City time) on October 9, 2027. The Participating Warrants were subsequently registered for resale by certain selling stockholders pursuant to a Trading Day. The resale securities will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition, in certain states,registration statement on Form S-3.
Under applicable rules and regulations under the Exchange Act, any person engaged in the distributionholder, of the resale securitiesnumber of shares of our common stock outstanding immediately after giving effect to the exercise. However, any holder may increase or decrease such percentage to any other percentage; provided, that a holder of Participating Warrant may not simultaneously engageincrease such percentage to a percentage in market making activities with respect to the common stock for the applicable restricted period, as defined in Regulation M, prior to the commencementexcess of 9.99% of the distribution. In addition, the Selling Stockholders will be subject to applicable provisionsnumber of the Exchange Act and the rules and regulations thereunder, including Regulation M, which may limit the timing of purchases and salesshares of the common stock outstanding immediately after giving effect to the issuance of shares of common stock upon exercise of the Participating Warrant held by the Selling Stockholdersholder. Any increase in such percentage will not be effective until the 61st day after such notice is delivered to the company. The exercise price of the Participating Warrants and the number of shares of our common stock issuable upon exercise of the Participating Warrant is subject to appropriate adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and also upon any distributions of assets, including cash, stock or other property to our stockholders. The Participating Warrants also contain provisions that provide certain rights to holders in the event of a fundamental transaction, including a merger or consolidation with or into another entity, such as (i) the right to receive the same amount and kind of consideration paid to the holders of our common stock in the fundamental transaction and (ii) the right to require the company to repurchase the unexercised portion of certain warrants at the warrant’s respective fair value using the Black Scholes option pricing formula. The Participating Warrants do not entitle the holders to any voting rights or any of the other rights or privileges to which holders of common stock are entitled.
At the time a particular offer of securities is made, if required, a prospectus supplement may differ from the terms we describe below, you should rely solely on information in the applicable prospectus supplement if that summary is different from the summary in this prospectus.
Plante & Moran, PLLC has audited our consolidated financial statements included
the ordinary course of business for which they receive compensation. We will describe the terms of such arrangements in the applicable prospectus supplement.
prospectus.
201 Redwood Shores Parkway,
275 Shoreline Drive, Suite
500
Redwood City, CA 94065
(650) 206-4507