Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
INSM similar filings
- 3 Jul 24 Regulation FD Disclosure
- 27 Jun 24 Insmed Announces Redemption of all $225 Million of Outstanding 1.75% Convertible Senior Notes Due 2025
- 13 Jun 24 Other Events
- 4 Jun 24 Regulation FD Disclosure
- 30 May 24 Insmed Announces Proposed $500 Million Public Offering of Common Stock
- 28 May 24 Insmed Announces Positive Topline Results from Landmark ASPEN Study of Brensocatib in Patients with Bronchiectasis
- 13 May 24 Departure of Directors or Certain Officers
Filing view
External links
Exhibit 99.1

Insmed Incorporated Commercial Presentation June 4, 2024

Forward Looking Statement Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties. “Forward-looking statements,” as that term is defined in the Private Securities Litigation Reform Act of 1995, are statements that are not historical facts and involve a number of risks and uncertainties. Words herein such as “may,” “will,” “should,” “could,” “would,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects,” “predicts,” “intends,” “potential,” “continues,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) may identify forward-looking statements. The forward-looking statements in this presentation are based upon the Company's current expectations and beliefs, and involve known and unknown risks, uncertainties and other factors, which may cause the Company's actual results, performance and achievements and the timing of certain events to differ materially from the results, performance, achievements or timings discussed, projected, anticipated or indicated in any forward-looking statements. Such risks, uncertainties and other factors include, among others, the following: the risk that the full data set from the ASPEN study or data generated in further clinical trials of brensocatib will not be consistent with the top-line results of the ASPEN study; failure to obtain, or delays in obtaining, regulatory approvals for brensocatib, TPIP or our other product candidates in the U.S., Europe or Japan or regulatory approvals for potential future brensocatib or TPIP indications; failure to successfully commercialize brensocatib, TPIP or our other product candidates, if approved by applicable regulatory authorities, in the U.S., Europe or Japan, or to maintain U.S., European or Japanese approval for brensocatib, TPIP or our other product candidates, if approved; uncertainties or changes in the degree of market acceptance of ARIKAYCE or, if approved, brensocatib or TPIP by physicians, patients, third-party payors and others in the healthcare community; failure to continue to successfully commercialize ARIKAYCE, our only approved product, in the U.S., Europe or Japan (amikacin liposome inhalation suspension, Liposomal 590 mg Nebuliser Dispersion, and amikacin sulfate inhalation drug product, respectively), or to maintain U.S., European or Japanese approval for ARIKAYCE; our inability to obtain full approval of ARIKAYCE from the FDA, including the risk that we will not successfully or in a timely manner validate a patient reported outcome (PRO) tool and complete the confirmatory post-marketing clinical trial required for full approval of ARIKAYCE, or our failure to obtain regulatory approval to expand ARIKAYCE’s indication to a broader patient population; failure to obtain, or delays in obtaining, regulatory approvals for ARIKAYCE outside the U.S., Europe or Japan, including separate regulatory approval for Lamira® in each market and for each usage; failure of third parties on which the Company is dependent, including Esteve Pharmaceuticals, S.A., Thermo Fisher Scientific, Inc. or the Company’s other third-party manufacturers, to manufacture sufficient quantities of ARIKAYCE, brensocatib, or TPIP for commercial or clinical needs, to conduct the Company's clinical trials, or to comply with the Company's agreements or laws and regulations that impact the Company's business or agreements with the Company; our inability to obtain and maintain adequate reimbursement from government or third-party payors for ARIKAYCE or, if approved, brensocatib or TPIP, or acceptable prices for ARIKAYCE or, if approved, brensocatib or TPIP; our inability to create or maintain an effective direct sales and marketing infrastructure or to partner with third parties that offer such an infrastructure for distribution of ARIKAYCE or, if approved, brensocatib or TPIP; risk that our competitors may obtain orphan drug exclusivity for a product that is essentially the same as a product we are developing for a particular indication; inaccuracies in our estimates of the size of the potential markets for ARIKAYCE, brensocatib, TPIP or our other product candidates, in our related estimates of peak sales potential, or in data we have used to identify physicians, expected rates of patient uptake, duration of expected treatment, or expected patient adherence or discontinuation rates; development of unexpected safety or efficacy concerns related to ARIKAYCE, brensocatib, TPIP or our other product candidates; restrictions or other obligations imposed on us by agreements related to brensocatib, including our license agreement with AstraZeneca AB, and failure to comply with our obligations under such agreements; failure to successfully conduct future clinical trials for ARIKAYCE, brensocatib, TPIP or our other product candidates, including due to the Company's potential inability to enroll or retain sufficient patients to conduct and complete the trials or generate data necessary for regulatory approvals, among other things; risks that the Company's clinical studies will be delayed, that serious side effects will be identified during drug development, or that any protocol amendments submitted will be rejected; risks that interim or partial data sets are not representative of a complete or larger data set or that blinded data will not be predictive of unblinded data; delays in the execution of plans to build out an additional third-party manufacturing facility approved by the appropriate regulatory authorities and unexpected expenses associated with those plans; the strength and enforceability of the Company’s intellectual property rights or the rights of third parties; the cost and potential reputational damage resulting from litigation to which the Company may become a party, including product liability claims; changes in laws and regulations applicable to our business, including any pricing reform and laws that impact our ability to utilize certain third parties in the research, development or manufacture of our product candidates, and failure to comply with such laws and regulations; inability to adapt to our highly competitive and changing environment; risk that we are unable to maintain our significant customers; our inability to attract and retain key personnel or to effectively manage our growth; risk that government healthcare reform materially increases our costs and damages our financial condition; risk that our operations are subject to a material disruption in the event of a cybersecurity attack or issue; business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises; deterioration in general economic conditions in the U.S., Europe, Japan and globally, including the effect of prolonged periods of inflation, affecting us, our suppliers, third-party service providers and potential partners; and inability to repay the Company's existing indebtedness and uncertainties with respect to the Company's need and ability to access future capital. The Company may not actually achieve the results, plans, intentions or expectations indicated by the Company's forward-looking statements because, by their nature, forward-looking statements involve risks and uncertainties because they relate to events and depend on circumstances that may or may not occur in the future. For additional information about the risks and uncertainties that may affect the Company's business, please see the factors discussed in Item 1A, "Risk Factors," in the Company's Annual Report on Form 10-K for the year ended December 31, 2023 and any subsequent Company filings with the Securities and Exchange Commission (SEC). The Company cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date of this presentation. The Company disclaims any obligation, except as specifically required by law and the rules of the SEC, to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.

Forward Looking Statement Additional Disclaimers With respect to the blended and blinded data observed from the ongoing TPIP study in pulmonary arterial hypertension, the dose titration and efficacy analyses were based on data available as of April 1, 2024, and the safety analyses were based on data available as of January 25, 2024, respectively. These findings may not be representative of results after the study is completed and all data are collected and analyzed. As a result, later interim data readouts and final data from this study may be materially different than the observations described herein, including with respect to efficacy, safety and tolerability of TPIP. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources, as well as our own internal estimates and research. While we believe the information in these third-party sources to be reliable as of the date of this presentation, we have not independently verified any such information or the underlying assumptions relied on in such third-party sources. In addition, while we believe our internal research is reliable, such research has not been verified by any independent source. Please be aware that brensocatib and TPIP are investigational products that have not been approved for sale or found safe or effective by the FDA or any regulatory authority. In addition, ARIKAYCE has not been approved for the treatment of all patients with MAC lung disease. This presentation is not promotion or advertisement of ARIKAYCE, brensocatib, or TPIP. Insmed and ARIKAYCE are registered trademarks of Insmed Incorporated. All other trademarks are property of their respective owner(s).

This presentation is intended to help frame the commercial potential for TPIP, ARIKAYCE and brensocatib POSITIVE CLINICAL DATA have transformed Insmed’s value In the last 9 months…

Refractory MAC NTM** All MAC NTM Bronchiectasis CRSsNP HS PH-ILD PAH A portfolio of multi-indication programs each with blockbuster potential* TPIP PH-ILD=Pulmonary hypertension due to interstitial lung disease; PAH=Pulmonary arterial hypertension MAC=Mycobacterium avium complex; NTM=nontuberculous mycobacterial Bronchiectasis refers to non-cystic fibrosis bronchiectasis; CRSsNP=Chronic rhinosinusitis sans nasal polyps; HS=Hidradenitis suppurativa *If approved **ARIKAYCE is currently approved for refractory MAC NTM ARIKAYCE® BRENSOCATIB

30K brensocatib Bronchiectasis TPIP PH-ILD Potential addressable patients anticipated to grow substantially with a steady cadence of launches* ARIKAYCE Refractory MAC 1.25M brensocatib CRSsNP 400K TPIP PAH 135K 90K future (through 2030) ~2.5M Bronchiectasis refers to non-cystic fibrosis bronchiectasis; MAC/MAC LD=mycobacterium avium complex lung disease; CRSsNP=chronic rhinosinusitis without nasal polyps, PH-ILD= pulmonary hypertension due to interstitial lung disease; PAH=pulmonary arterial hypertension; HS=hidradenitis suppurativa *If approved Note: Prevalence numbers depicted here are further detailed on slides 9, 10, 13, 17, 22, 24 today 275K ARIKAYCE MAC LD brensocatib HS 600K

Three therapies with positive clinical data in-hand have potential to be first- and/or best-in-disease* >$2bn Max. dose well-tolerated Nominally stat. sig. benefit on clinical worsening TPIP PH-ILD Phase 2 Bronchiectasis refers to non-cystic fibrosis bronchiectasis PVR reduction including placebo similar to treatment with other prostanoids PAH Phase 2** Peak sales potential *”Best-in-disease/best-in-class" indicates a profile that could be considered more attractive than other treatment options for a particular disease. Head-to-head clinical trials are not anticipated. **Based on blended and blinded data observed from the ongoing Phase 2 study of TPIP in PAH. Efficacy analyses were based on data available as of April 1, 2024 >$1bn Validated a PRO tool Nominally stat. sig. culture conversion benefit at Month 7 compared to active control ARIKAYCE® ARISE >$5bn Potential for a new standard of care in bronchiectasis Unlocks DPP1 mechanism for neutrophil-mediated diseases BRENSOCATIB ASPEN
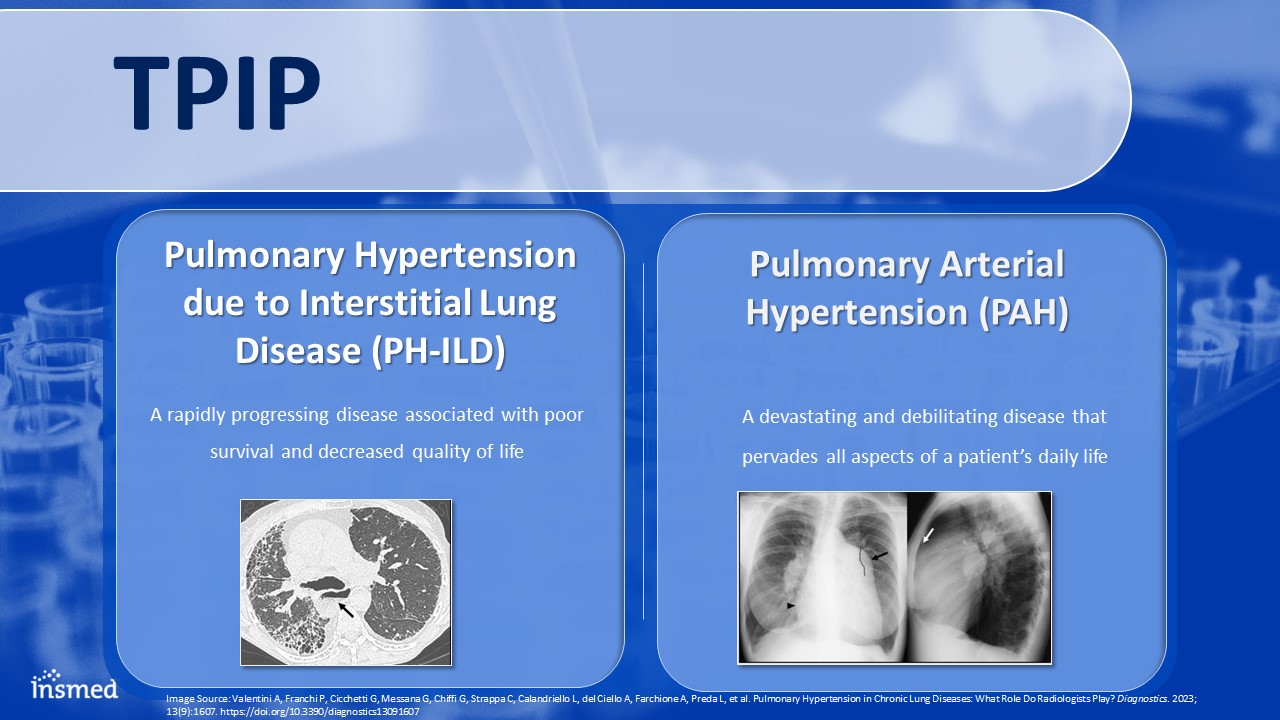
TPIP Pulmonary Arterial Hypertension (PAH) A devastating and debilitating disease that pervades all aspects of a patient’s daily life Pulmonary Hypertension due to Interstitial Lung Disease (PH-ILD) A rapidly progressing disease associated with poor survival and decreased quality of life Image Source: Valentini A, Franchi P, Cicchetti G, Messana G, Chiffi G, Strappa C, Calandriello L, del Ciello A, Farchione A, Preda L, et al. Pulmonary Hypertension in Chronic Lung Diseases: What Role Do Radiologists Play? Diagnostics. 2023; 13(9):1607. https://doi.org/10.3390/diagnostics13091607
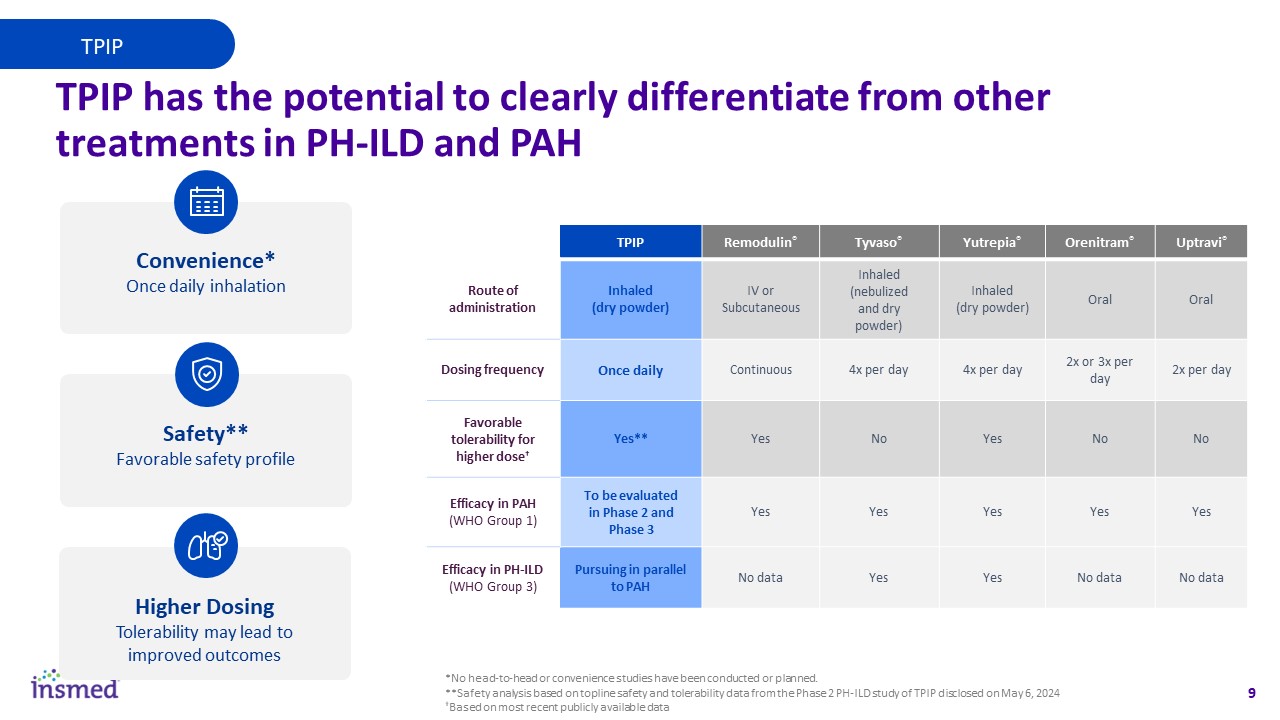
TPIP has the potential to clearly differentiate from other treatments in PH-ILD and PAH Higher Dosing Tolerability may lead to improved outcomes Safety** Favorable safety profile Convenience* Once daily inhalation TPIP TPIP Remodulin® Tyvaso® Yutrepia® Orenitram® Uptravi® Route of administration Inhaled (dry powder) IV orSubcutaneous Inhaled (nebulized and dry powder) Inhaled (dry powder) Oral Oral Dosing frequency Once daily Continuous 4x per day 4x per day 2x or 3x per day 2x per day Favorable tolerability for higher dose† Yes** Yes No Yes No No Efficacy in PAH (WHO Group 1) To be evaluated in Phase 2 and Phase 3 Yes Yes Yes Yes Yes Efficacy in PH-ILD (WHO Group 3) Pursuing in parallel to PAH No data Yes Yes No data No data *No head-to-head or convenience studies have been conducted or planned. **Safety analysis based on topline safety and tolerability data from the Phase 2 PH-ILD study of TPIP disclosed on May 6, 2024 †Based on most recent publicly available data
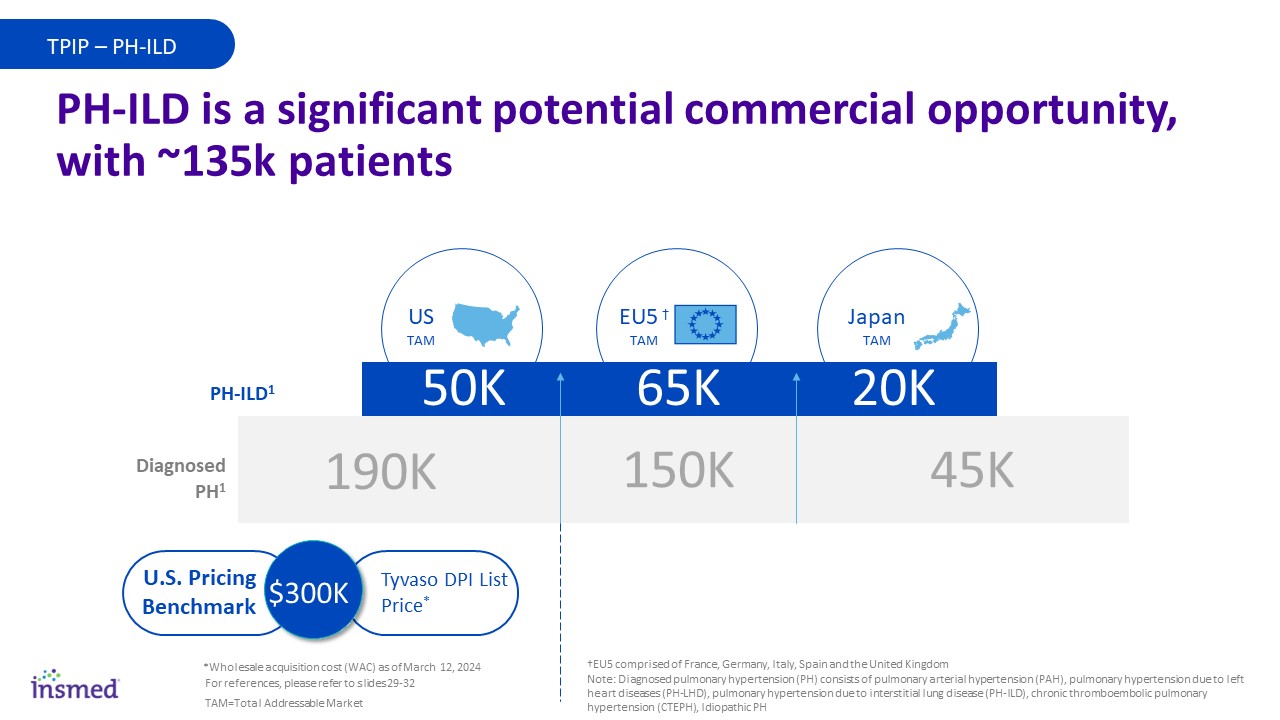
Diagnosed with BE15 TPIP TPIP – PH-ILD PH-ILD is a significant potential commercial opportunity, with ~135k patients 65K 50K 20K 190K 150K 45K PH-ILD1 Diagnosed PH1 Japan TAM US TAM EU5 † TAM Tyvaso DPI List Price* U.S. Pricing Benchmark $300K *Wholesale acquisition cost (WAC) as of March 12, 2024 †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom Note: Diagnosed pulmonary hypertension (PH) consists of pulmonary arterial hypertension (PAH), pulmonary hypertension due to left heart diseases (PH-LHD), pulmonary hypertension due to interstitial lung disease (PH-ILD), chronic thromboembolic pulmonary hypertension (CTEPH), Idiopathic PH For references, please refer to slides 29-32 TAM=Total Addressable Market

Diagnosed with BE15 40K 35K 15K 190K 150K 45K PAH2 Diagnosed PH2 TPIP represents potential best-in-class* prostanoid for ~90k patients with PAH Japan TAM US TAM Tyvaso DPI List Price* EU5 † TAM U.S. Pricing Benchmark $300K *Wholesale acquisition cost (WAC) as of March 12, 2024 For references, please refer to slides 29-32 †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom Note: Diagnosed pulmonary hypertension (PH) consists of pulmonary arterial hypertension (PAH), pulmonary hypertension due to left heart diseases (PH-LHD), pulmonary hypertension due to interstitial lung disease (PH-ILD), chronic thromboembolic pulmonary hypertension (CTEPH), Idiopathic PH TPIP – PAH *”Best-in-disease/best-in-class" indicates a profile that could be considered more attractive than other treatment options for a particular disease. Head-to-head clinical trials are not anticipated. TAM=Total Addressable Market

Phase 3 STELLAR trial suggests that sotatercept will be complementary with treprostinil products Note: Prostacyclins are part of the prostacyclin receptor agonist class Prostacyclins expected to remain cornerstone of PAH therapy; TPIP has potential to be prostacyclin of choice Regardless of sotatercept’s efficacy, the prostacyclin pathway will remain important to PAH Thoughts from Key Opinion Leaders in the PAH Space3,4 TPIP really sounds like something that could be very valuable to the patient TPIP – PAH For references, please refer to slides 29-32
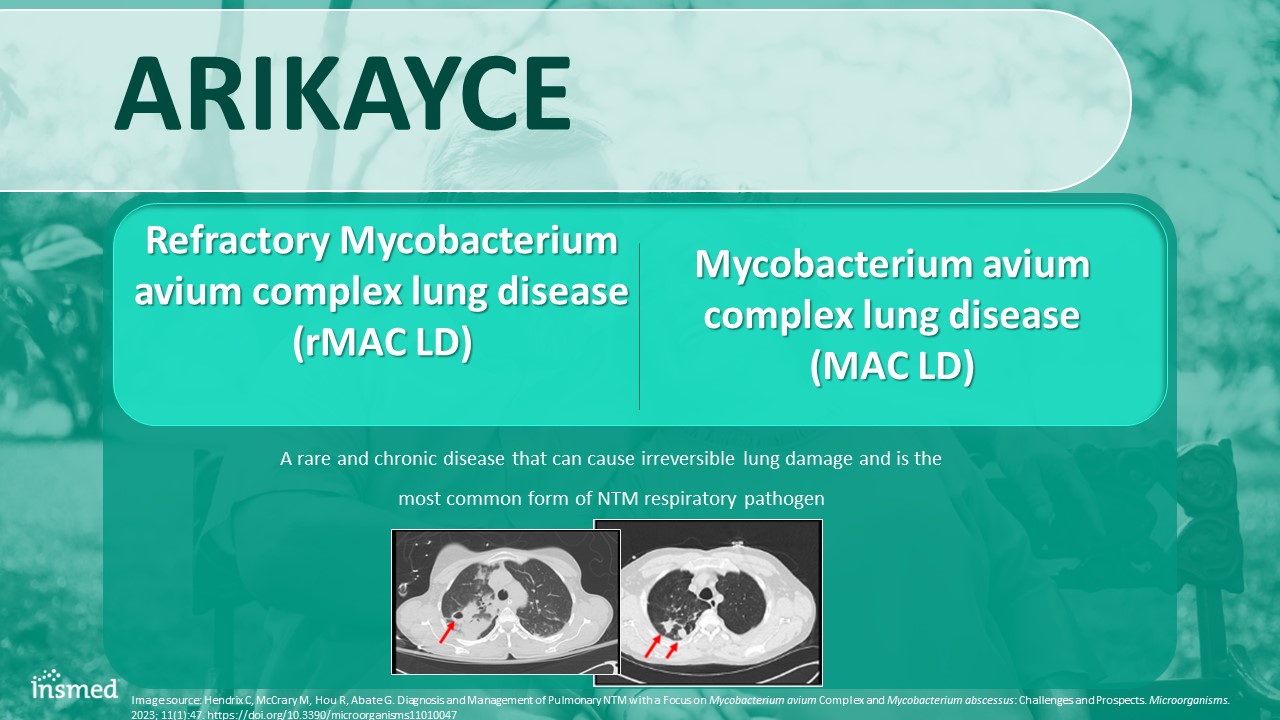
ARIKAYCE Refractory Mycobacterium avium complex lung disease (rMAC LD) A rare and chronic disease that can cause irreversible lung damage and is the most common form of NTM respiratory pathogen Mycobacterium avium complex lung disease (MAC LD) Image source: Hendrix C, McCrary M, Hou R, Abate G. Diagnosis and Management of Pulmonary NTM with a Focus on Mycobacterium avium Complex and Mycobacterium abscessus: Challenges and Prospects. Microorganisms. 2023; 11(1):47. https://doi.org/10.3390/microorganisms11010047
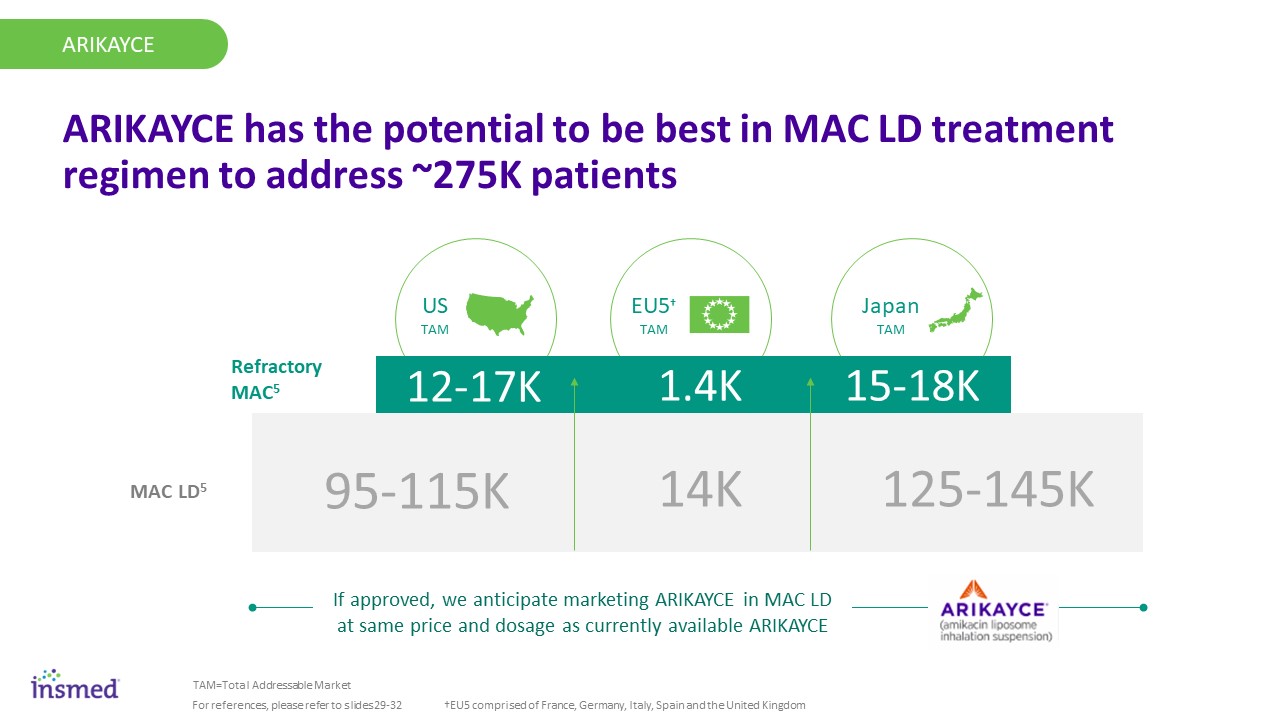
Japan TAM US TAM EU5† TAM Diagnosed with BE15 1.4K 12-17K 15-18K 95-115K 14K 125-145K ARIKAYCE Refractory MAC5 MAC LD5 ARIKAYCE ARIKAYCE has the potential to be best in MAC LD treatment regimen to address ~275K patients TAM=Total Addressable Market For references, please refer to slides 29-32 †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom If approved, we anticipate marketing ARIKAYCE in MAC LD at same price and dosage as currently available ARIKAYCE

ARIKAYCE brensocatib Physician and patient overlap between NTM and BE is high Insmed has strong relationships in this space, spanning 6+ years Bronchiectasis refers to non-cystic fibrosis bronchiectasis For references, please refer to slides 29-32 Bronchiectasis (BE) and nontuberculous mycobacterial (NTM) lung disease are inextricably linked pathophysiologically6
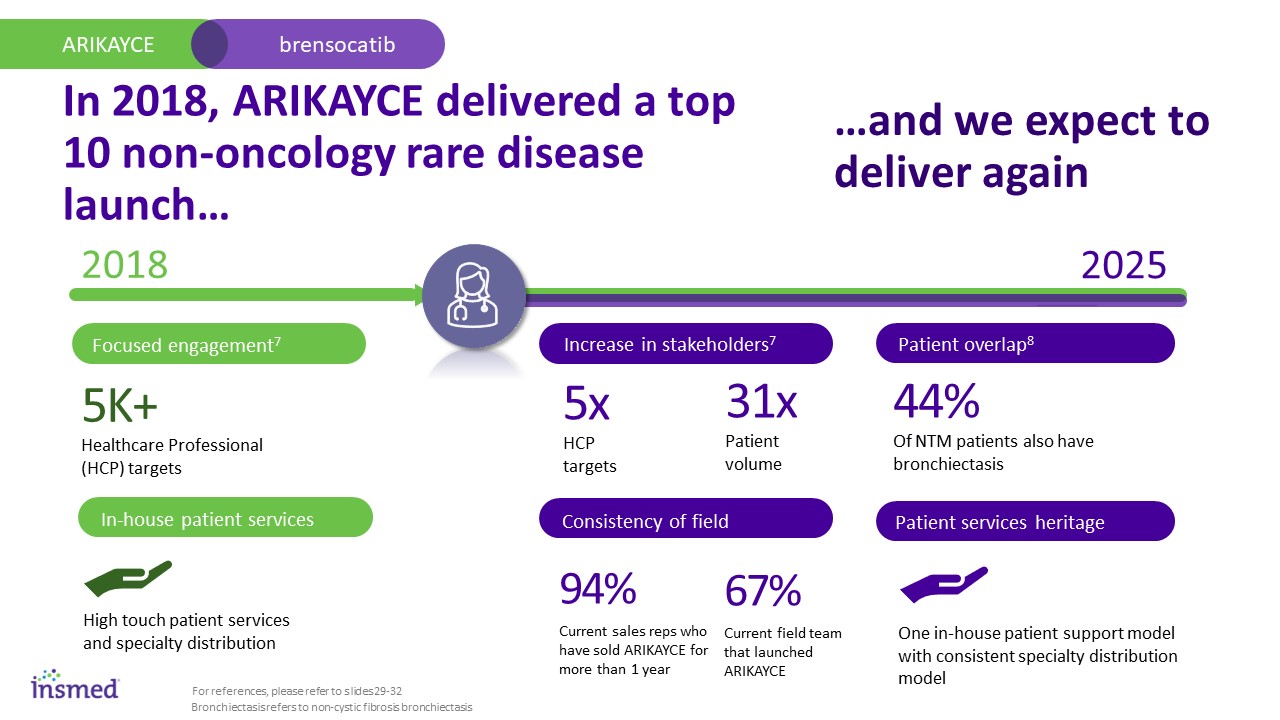
In 2018, ARIKAYCE delivered a top 10 non-oncology rare disease launch… ARIKAYCE brensocatib 2018 2025 94% Current sales reps who have sold ARIKAYCE for more than 1 year 5x HCP targets 31x Patient volume Increase in stakeholders7 Consistency of field 67% Current field team that launched ARIKAYCE Patient overlap8 5K+ Healthcare Professional (HCP) targets Focused engagement7 In-house patient services High touch patient services and specialty distribution Patient services heritage One in-house patient support model with consistent specialty distribution model 44% Of NTM patients also have bronchiectasis For references, please refer to slides 29-32 Bronchiectasis refers to non-cystic fibrosis bronchiectasis …and we expect to deliver again
BRENSOCATIB Chronic Rhinosinusitis without Nasal Polyps (CRSsNP) Hidradenitis Suppurativa (HS) Bronchiectasis (BE) A chronic, progressive inflammatory disease that causes permanent lung damage Bronchiectasis refers to non-cystic fibrosis bronchiectasis Image Source: 1) Fraser CS, José RJ. Insights into Personalised Medicine in Bronchiectasis. Journal of Personalized Medicine. 2023; 13(1):133. https://doi.org/10.3390/jpm13010133. 2) Rethinkbronchiectasis.com
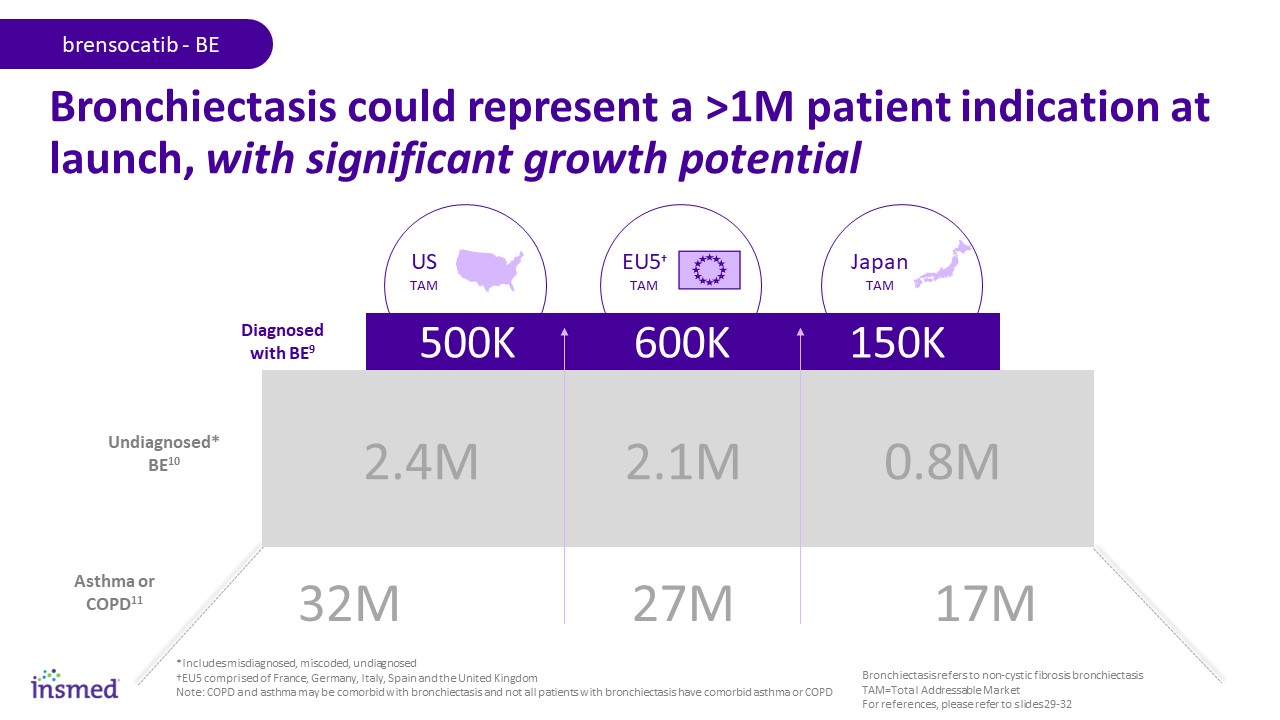
Japan TAM *Includes misdiagnosed, miscoded, undiagnosed †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom Note: COPD and asthma may be comorbid with bronchiectasis and not all patients with bronchiectasis have comorbid asthma or COPD Bronchiectasis could represent a >1M patient indication at launch, with significant growth potential Asthma or COPD11 Undiagnosed* BE10 Diagnosed with BE9 600K 500K 150K US TAM 2.4M 2.1M 0.8M 32M 27M 17M brensocatib - BE EU5† TAM Bronchiectasis refers to non-cystic fibrosis bronchiectasis TAM=Total Addressable Market For references, please refer to slides 29-32
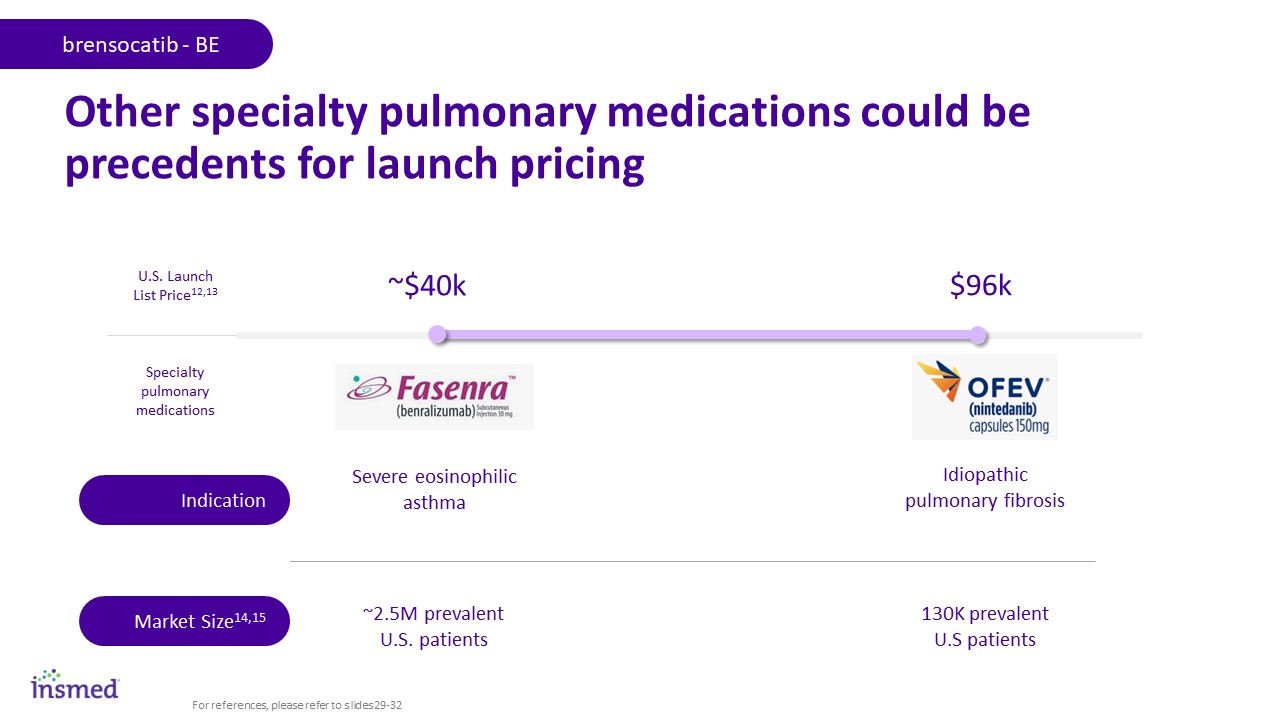
Other specialty pulmonary medications could be precedents for launch pricing ~$40k U.S. Launch List Price12,13 $96k Severe eosinophilic asthma Idiopathic pulmonary fibrosis ~2.5M prevalent U.S. patients 130K prevalent U.S patients Specialty pulmonary medications Indication Market Size14,15 brensocatib - BE For references, please refer to slides 29-32
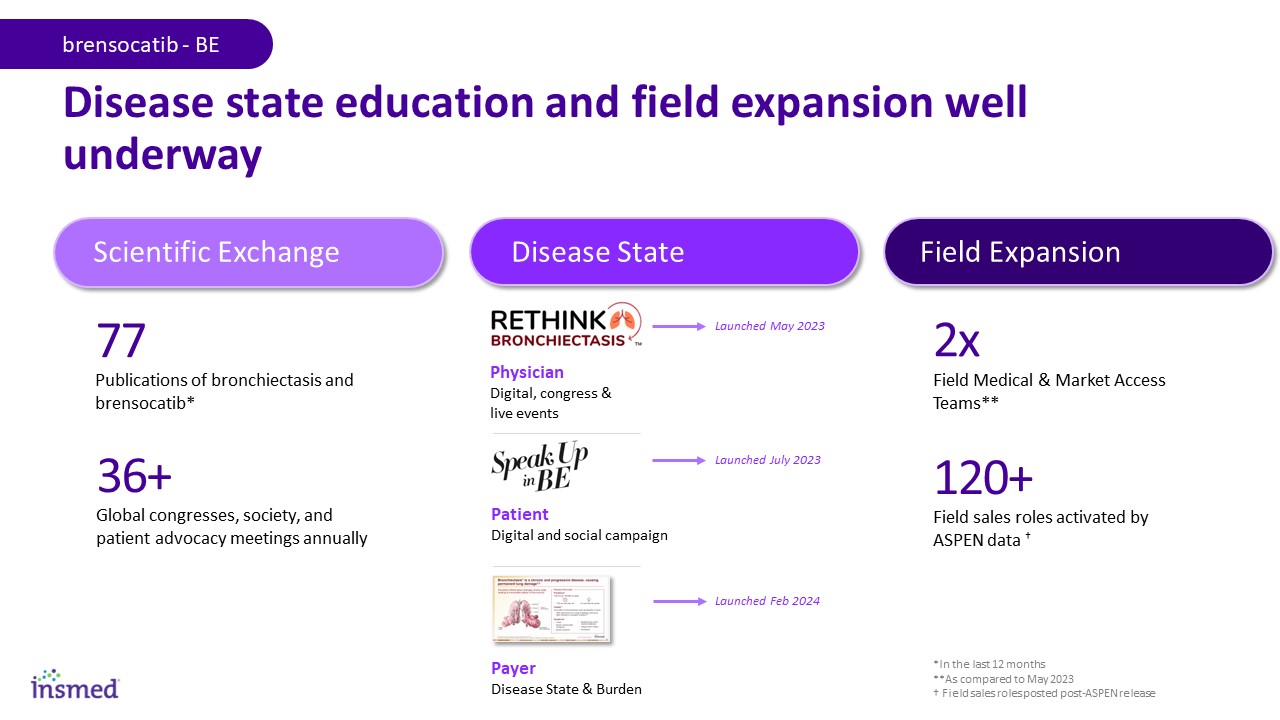
Disease state education and field expansion well underway Scientific Exchange Disease State Field Expansion 77 Publications of bronchiectasis and brensocatib* 36+ Global congresses, society, and patient advocacy meetings annually Physician Digital, congress & live events Patient Digital and social campaign 120+ Field sales roles activated by ASPEN data † 2x Field Medical & Market Access Teams** brensocatib - BE Payer Disease State & Burden Launched May 2023 Launched July 2023 Launched Feb 2024 *In the last 12 months **As compared to May 2023 † Field sales roles posted post-ASPEN release

Motivated to act17 Intend to prescribe16 Recognize disease burden18 *Since website launch in November 2023 **Highly engaged defined as those patients who have downloaded support tools or registered for CRM †From February-May 2024; Commercial lives include VA, Tricare and DOD Strong signals from stakeholders that uptake at launch could be rapid U.S. Physicians Patients Payers 60% See significantly high unmet need in bronchiectasis 80% Likely to prescribe brensocatib 300K Unique visits to disease state website* 26K Highly engaged** visitors who have acted >85% Medicare lives† >85% Commercial lives † brensocatib - BE For references, please refer to slides 29-32 Lives covered by engaged accounts: Bronchiectasis refers to non-cystic fibrosis bronchiectasis
BRENSOCATIB Hidradenitis Suppurativa (HS) Bronchiectasis (BE) Chronic Rhinosinusitis without Nasal Polyps (CRSsNP) A burdensome disease that significantly impairs quality of life Image source: Yang Y, Zhang N, Crombruggen KV, Lan F, Hu G, Hong S, Bachert C. Differential Expression and Release of Activin A and Follistatin in Chronic Rhinosinusitis with and without Nasal Polyps. PLoS One. 2015 Jun 1;10(6):e0128564. doi: 10.1371/journal.pone.0128564. PMID: 26030615; PMCID: PMC4451080
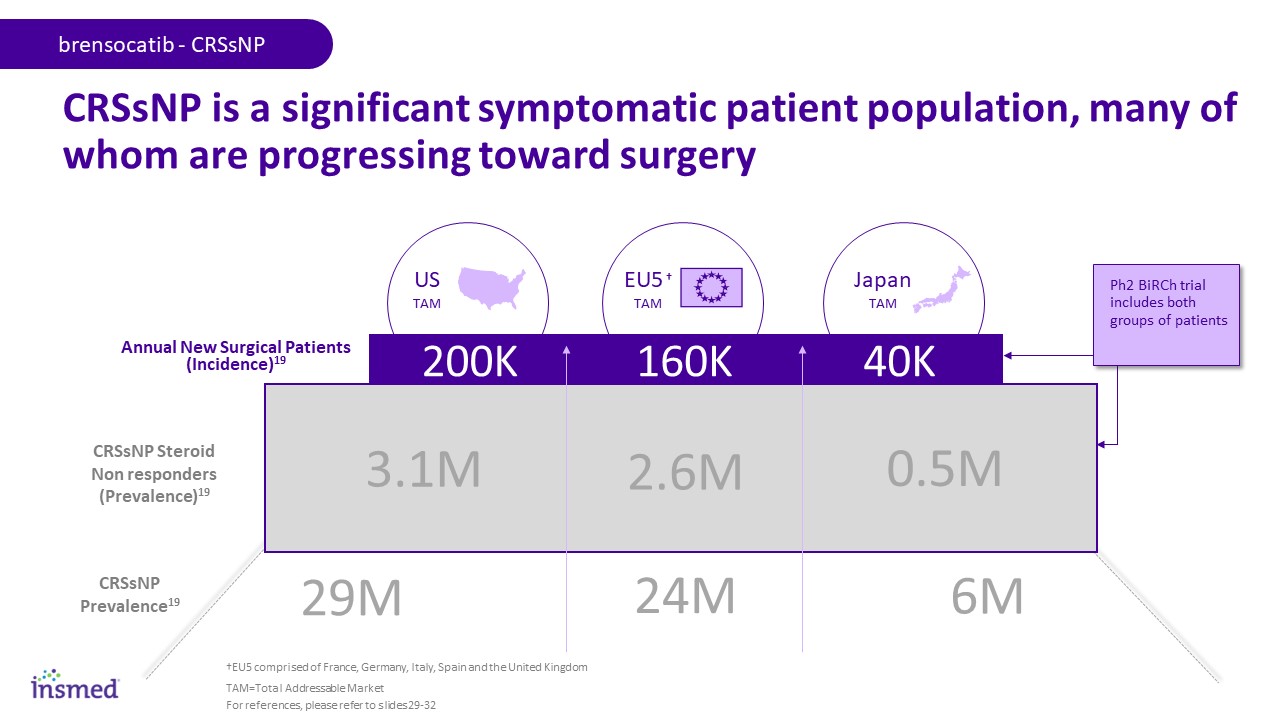
Diagnosed with BE15 Japan TAM CRSsNP is a significant symptomatic patient population, many of whom are progressing toward surgery CRSsNP Prevalence19 CRSsNP Steroid Non responders (Prevalence)19 Annual New Surgical Patients (Incidence)19 160K 200K 40K US TAM 3.1M 2.6M 0.5M 29M 24M 6M brensocatib - CRSsNP EU5 † TAM TAM=Total Addressable Market Ph2 BiRCh trial includes both groups of patients For references, please refer to slides 29-32 †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom
BRENSOCATIB Bronchiectasis (BE) Chronic Rhinosinusitis without Nasal Polyps (CRSsNP) Hidradenitis Suppurativa (HS) A chronic, recurrent and debilitating inflammatory disease with significant treatment challenges Image source: Z.N. Ovadja, M.M. Schuit, C.M.A.M. van der Horst, O. Lapid, Inter‐ and intrarater reliability of Hurley staging for hidradenitis suppurativa, British Journal of Dermatology, Volume 181, Issue 2, 1 August 2019, Pages 344–349, https://doi.org/10.1111/bjd.17588
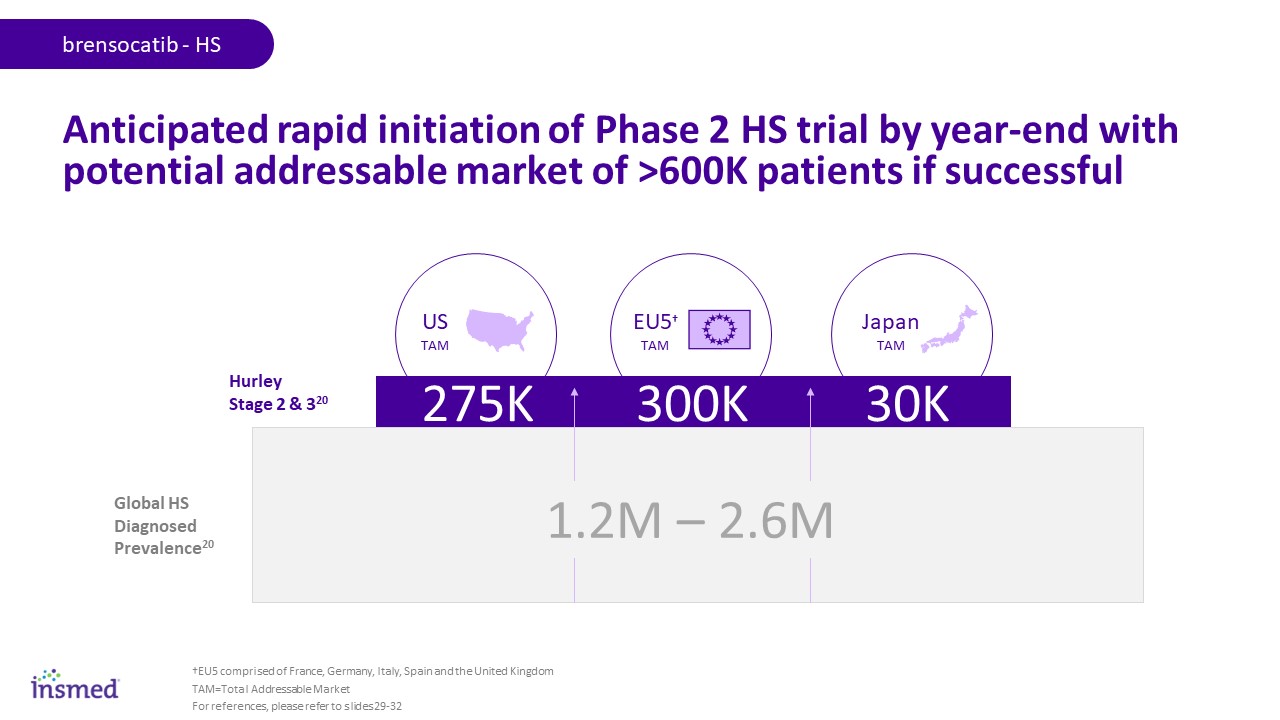
Diagnosed with BE15 300K 275K 30K 1.2M – 2.6M Global HS Diagnosed Prevalence20 Hurley Stage 2 & 320 Japan TAM US TAM EU5† TAM brensocatib - HS Anticipated rapid initiation of Phase 2 HS trial by year-end with potential addressable market of >600K patients if successful TAM=Total Addressable Market For references, please refer to slides 29-32 †EU5 comprised of France, Germany, Italy, Spain and the United Kingdom
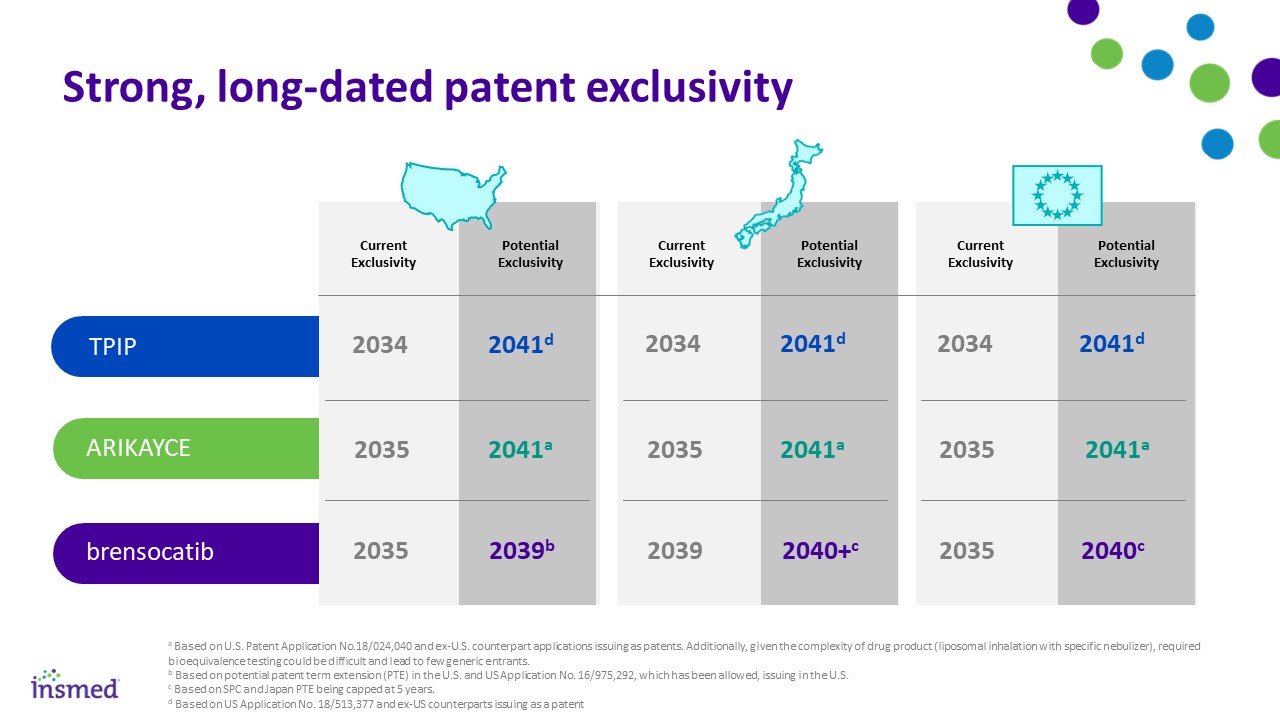
brensocatib a Based on U.S. Patent Application No.18/024,040 and ex-U.S. counterpart applications issuing as patents. Additionally, given the complexity of drug product (liposomal inhalation with specific nebulizer), required bioequivalence testing could be difficult and lead to few generic entrants. b Based on potential patent term extension (PTE) in the U.S. and US Application No. 16/975,292, which has been allowed, issuing in the U.S. c Based on SPC and Japan PTE being capped at 5 years. d Based on US Application No. 18/513,377 and ex-US counterparts issuing as a patent Strong, long-dated patent exclusivity Current Exclusivity Potential Exclusivity Current Exclusivity Potential Exclusivity Current Exclusivity Potential Exclusivity 2035 2039b 2039 2040+c 2035 2040c ARIKAYCE 2035 2041a 2035 2041a 2035 2041a TPIP 2034 2041d 2034 2041d 2034 2041d
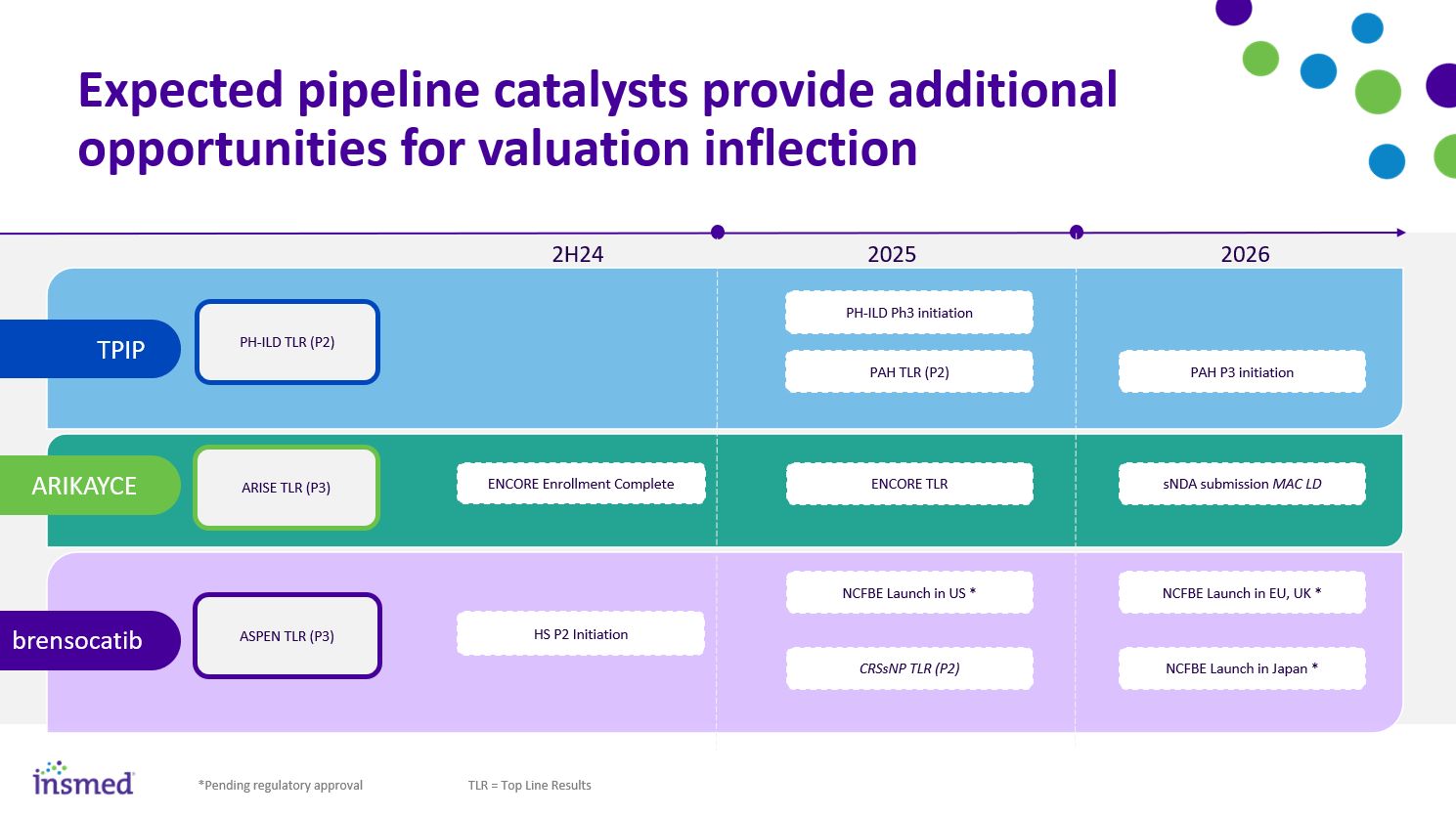
sNDA submission MAC LD ENCORE Enrollment Complete ASPEN TLR (P3) NCFBE Launch in US * NCFBE Launch in EU, UK * NCRBE Launch in Japan * ARIKAYCE 2025 2026 brensocatib Expected pipeline catalysts provide additional opportunities for valuation inflection ARISE TLR (P3) HS P2 Initiation ENCORE TLR CRSsNP TLR (P2) 2H24 *Pending regulatory approval PH-ILD TLR (P2) PH-ILD Ph3 initiation PAH P3 initiation TPIP PAH TLR (P2) TLR = Top Line Results

References

Internal assessment of published epidemiology, including: Andersen, C. U., Mellemkjær, S., Hilberg, O., Nielsen-Kudsk, J. E., Simonsen, U., & Bendstrup, E. (2012). Pulmonary hypertension in interstitial lung disease: prevalence, prognosis and 6 min walk test. Respiratory medicine, 106(6), 875-882. Ryu, Jay H., et al. "Pulmonary hypertension in patients with interstitial lung diseases." Mayo Clinic Proceedings. Vol. 82. No. 3. Elsevier, 2007 Duchemann et al., “Prevalence and incidence of interstitial lung diseases in a multi-ethnic county of Greater Paris.” European Respiratory Journal, 2017 Diagnosed prevalence for PH-LHD, CTEPH and PH-Idiopathic sourced from “Patient-Based Forecast Model Pulmonary Hypertension”, Datamonitor, September 2023. Internal assessment of published epidemiology and US patient level claims data analysis, including: Kirson, N. Y., Birnbaum, H. G., Ivanova, J. I., Waldman, T., Joish, V., & Williamson, T. (2011). Prevalence of pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension in the United States. Current Medical Research and Opinion, 27(9), 1763–1768. https://doi.org/10.1185/03007995.2011.604310 2019 National Audit of Pulmonary Hypertension Great Britain; Humbert M et al, “Pulmonary arterial hypertension in France: results from a national registry”, Feb 2006 Ling Y, Johnson MK, Kiely DG, Condliffe R, Elliot CA, Gibbs JS, Howard LS, Pepke-Zaba J, Sheares KK, Corris PA, Fisher AJ, Lordan JL, Gaine S, Coghlan JG, Wort SJ, Gatzoulis MA, Peacock AJ. Changing demographics, epidemiology, and survival of incident pulmonary arterial hypertension: results from the pulmonary hypertension registry of the United Kingdom and Ireland. Am J Respir Crit Care Med. 2012 Oct 15;186(8):790-6. doi: 10.1164/rccm.201203-0383OC. Epub 2012 Jul 12. PMID: 22798320. Escribano-Subias P, Blanco I, López-Meseguer M, Lopez-Guarch CJ, Roman A, Morales P, Castillo-Palma MJ, Segovia J, Gómez-Sanchez MA, Barberà JA; REHAP investigators. Survival in pulmonary hypertension in Spain: insights from the Spanish registry. Eur Respir J. 2012 Sep;40(3):596-603. doi: 10.1183/09031936.00101211. Epub 2012 Feb 23. PMID: 22362843. Hoeper MM, Huscher D, Pittrow D. Incidence and prevalence of pulmonary arterial hypertension in Germany. Int J Cardiol. 2016 Jan 15;203:612-3. doi: 10.1016/j.ijcard.2015.11.001. Epub 2015 Nov 9. PMID: 26580339. Humbert M, Sitbon O, Chaouat A, Bertocchi M, Habib G, Gressin V, Yaici A, Weitzenblum E, Cordier JF, Chabot F, Dromer C, Pison C, Reynaud-Gaubert M, Haloun A, Laurent M, Hachulla E, Simonneau G. Pulmonary arterial hypertension in France: results from a national registry. Am J Respir Crit Care Med. 2006 May 1;173(9):1023-30. doi: 10.1164/rccm.200510-1668OC. Epub 2006 Feb 2. PMID: 16456139. Secondary research: Japan’s Intractable Disease Database 2021 Diagnosed prevalence for PH-LHD, CTEPH and PH-Idiopathic sourced from “Patient-Based Forecast Model Pulmonary Hypertension”, Datamonitor, September 2023. Cowen & Company – UTHR PH-ILD PAH Buying Process Research References (1 of 4)

Internal analysis of published NTM epidemiology, including internal market research and US patient level claims data analysis: Jennifer Adjemian, Kenneth N Olivier, Amy E Seitz, Steven M Holland, D Rebecca Prevots: Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries Am J Respir Crit Care Med. 2012 Apr 15; 185(8):881-6 DOI: 10.1164/rccm.201111-2016OC Jennifer Adjemian, D Rebecca Prevots, Jack Gallagher, Kylee Heap, Renu Gupta, David Griffith: Lack of adherence to evidence-based treatment guidelines for nontuberculous mycobacterial lung disease Ann Am Thorac Soc. 2014 Jan; 11(1): 9–16 DOI: 10.1513/AnnalsATS.201304-085OC Sara E. Strollo , Jennifer Adjemian, Michael K. Adjemian, and D. Rebecca Prevots: The Burden of Pulmonary Nontuberculous Mycobacterial Disease in the United States Ann Am Thorac Soc Vol 12, No 10, pp 1458–1464, Oct 2015 DOI: 10.1513/AnnalsATS.201503-173OC https://www.kff.org/medicare/state-indicator/total-medicare-beneficiaries/?currentTimeframe=0&sortModel=%7B%22colId%22:%22Location%22,%22sort%22:%22asc%22%7D Felix C. Ringshausen, Dirk Wagner, Andrés de Roux, Roland Diel, David Hohmann, Lennart Hickstein, Tobias Welte, Jessica Rademacher: Prevalence of Nontuberculous Mycobacterial Pulmonary Disease, Germany, 2009–2014 Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 22, No. 6, June 2016 DOI: http://dx.doi.org/10.3201/eid2206.151642 Jonathan E Moore, Michelle E Kruijshaar, L Peter Ormerod, Francis Drobniewski , Ibrahim Abubakar: Increasing reports of non-tuberculous mycobacteria in England, Wales and Northern Ireland, 1995-2006 BMC Public Health 2010, 10:612 http://www.biomedcentral.com/1471-2458/10/612 Hoefsloot, Van Ingen et al, The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples, AN NTM-NET collaborative study; 2013, European Respiratory Journal 2013 42: 1604-1613; DOI: 10.1183/09031936.00149212 Kozo Morimoto , Kazuro Iwai , Kazuhiro Uchimura , Masao Okumura , Takashi Yoshiyama , Kozo Yoshimori, Hideo Ogata , Atsuyuki Kurashima , Akihiko Gemma, and Shoji Kudoh: A Steady Increase in Nontuberculous Mycobacteriosis Mortality and Estimated Prevalence in Japan Ann Am Thorac Soc Vol 11, No 1, pp 1–8, Jan 2014, DOI: 10.1513/AnnalsATS.201303-067OC Griffith D et al. Clinics in Chest Medicine. 2012; 33(2):283-295. Based on internal field sizing exercise. Adjemian J et al. Am J Respir Crit Care Med. 2022; 185(8):881-886. Internal analysis of published BE epidemiology, including internal market research and US patient level claims data analysis: Weycker, et al. Prevalence and incidence of NCFBE among US adults in 2013. Chronic Respiratory Disease. 2017 BE Patient Level Claims Data Analysis. Source: swoop/ipm.ai Trinity Epidemiology Assessment; 2020 (for Japan epi) Ringausen et al 2019 Growth (Germany) Aliberti 2016; quality standards for the management of bronchiectasis in Italy Snell et al. United Kingdom; 2019 Internal Insmed NCFBE market sizing EU5 report References (2 of 4)

References (3 of 4) Internal analysis and estimations based on internal market research and US patient level claims data analysis: Insmed Analysis 2022: Potential Undiagnosed or Misdiagnosed (with COPD, Asthma) BE patients in US estimated based on Medical Experts driven insights, applied to Patient Level Claims Data -using advanced analytics / statistical methods Potential Undiagnosed or Co-morbid (with COPD) BE patients in US derived based on internal Insmed meta-analysis of 16 epi studies that look at BE prevalence in COPD patients; Ex-US estimates are based on extrapolation of US focused claims and epi data analysis Internal analysis and estimations based on published epidemiology studies: National Health Interview Survey (NHIS) Data (2021) Alshabanat A, Zafari Z, Albanyan O, Dairi M, FitzGerald JM (2015) Asthma and COPD Overlap Syndrome (ACOS): A Systematic Review and Meta Analysis. PLoS ONE 10(9): e0136065. doi:10.1371/ journal.pone.0136065 OECD/European Union (2016), “Asthma and COPD prevalence”, in Health at a Glance: Europe 2016: State of Health in the EU Cycle, OECD Publishing, Paris. Hosseini, M., Almasi-Hashiani, A., Sepidarkish, M. et al. Global prevalence of asthma-COPD overlap (ACO) in the general population: a systematic review and meta-analysis. Respir Res 20, 229 (2019). https://doi.org/10.1186/s12931-019-1198-4 Blanco I, Diego I, Bueno P, et al. Geographic distribution of COPD prevalence in the world displayed by Geographic Information System maps. Eur Respir J 2019; 54: 1900610 [https://doi.org/ 10.1183/13993003.00610-2019]. R. de Marco et al. Eur Respir J 2012; 39:883-892. DOI: 10.1183/09031936.000611. Iwanaga T, Tohda Y. [Epidemiology of asthma in Japan]. Nihon Rinsho. 2016 Oct;74(10):1603-1608. Japanese. PMID: 30551268 Massoth L, Anderson C, McKinney KA. Asthma and Chronic Rhinosinusitis: Diagnosis and Medical Management. Med Sci (Basel). 2019 Mar 27;7(4):53. doi: 10.3390/medsci7040053. PMID: 30934800; PMCID: PMC6524348. Hashimoto S, Yoshida Y, Makita N, Sorimachi R, Sugaya S, Arita Y, Hayashi N, Tashiro N, Ichinose M. Real-World Evidence on the Diagnostic and Clinical Characteristics of Asthma in Japanese Patients with COPD: The ACO Japan Cohort Study. Int J Chron Obstruct Pulmon Dis. 2023;18:37-46 Awad MT, Sankari A. Asthma and COPD Overlap. [Updated 2023 Jun 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK592422/ https://www.cdc.gov/asthma/most_recent_national_asthma_data.htm https://www.cdc.gov/copd/php/case-reporting/national-trends-in-copd.html Minakata Y, Ichinose M. [Epidemiology of COPD in Japan]. Nihon Rinsho. 2011 Oct;69(10):1721-6. Japanese. PMID: 22073563

References (4 of 4) Reuters; Nov 15 2017. AstraZeneca pricing new severe asthma drug Fasenra below rivals Pulmonary Fibrosis News.com/Two New IP Treatments Reportedly Will Cost 94K-96K American Lung Association/Severe Asthma Raghu G, Weycker D, Edelsberg J, Bradford WZ, Oster G. Incidence and prevalence of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2006;174(7):810-816. doi:10.1164/rccm.200602-163OC. Brensocatib Competitive Impact Demand Assessment; 2022. Adelphi Research. Patient website metrics dashboard; as of May 2024 Payer KPI metrics; as of May 2024 Internal assessment of published epidemiology and US patient level claims data analysis : Cho et. al, Chronic Rhinosinusitis without Nasal Polyps J Allergy Clin Immunol Pract. 2016 ; 4(4): 575–582. doi:10.1016/j.jaip.2016.04.015 Benjamin et. al, Clinical Characteristics of Patients with Chronic Rhinosinusitis without Nasal Polyps in an Academic Setting, J ALLERGY CLIN IMMUNOL PRACT VOLUME 7, NUMBER 3, MARCH 2019 Komodo Health: CRS patient level claims data analysis 2022 - US only, Extrapolated to Europe5 and Japan Palmer JN, Messina JC, Biletch R, Grosel K, Mahmoud RA. A cross-sectional, population-based survey of U.S. adults with symptoms of chronic rhinosinusitis. Allergy Asthma Proc. 2019 Jan 14;40(1):48-56. doi: 10.2500/aap.2019.40.4182. PMID: 30582496 Internal assessment of market research, published epidemiology and US patient level claims data analysis : Hidradenitis Suppurativa (HS) - Market Insights, Epidemiology, and Market Forecast Report (2019-2032). Source: DelveInsight Komodo Health: HS patient level claims data analysis 2024: Potential HS Patients (1+ HS Dx in claims history 2016-23) - US only, Extrapolated to Europe5 and Japan