Table of Contents
Washington, D.C. 20549
To
UNDER
THE SECURITIES ACT OF 1933
| Delaware (State or other jurisdiction of incorporation or organization) | 5033 (Primary Standard Industrial Classification Code Number) | 04-3559972 (I.R.S. Employer Identification Number) |
Northborough, Massachusetts 01532
(508) 691-1111
President and Chief Executive Officer
Aspen Aerogels, Inc.
30 Forbes Road, Building B
Northborough, Massachusetts 01532
(508) 691-1111
| Sahir Surmeli, Esq. Jonathan L. Kravetz, Esq. Thomas R. Burton, III, Esq. Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. One Financial Center Boston, Massachusetts 02111 (617) 542-6000 | Vincent Pagano, Jr., Esq. Simpson Thacher & Bartlett LLP 425 Lexington Avenue New York, New York 10017 (212) 455-2000 |
Large accelerated filer o | Accelerated filer o | Non-accelerated filer þ (Do not check if a smaller reporting company) | Smaller reporting company o |
Table of Contents
| The information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell nor does it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted. |

| Per Share | Total | |||||||
| Initial public offering price | $ | $ | ||||||
| Underwriting discount | $ | $ | ||||||
| Proceeds, before expenses, to us | $ | $ | ||||||
| Goldman, Sachs & Co. | Morgan Stanley |
Table of Contents

| aspen aerogels |
Page | ||||||||
| 1 | ||||||||
| 12 | ||||||||
| 38 | ||||||||
| 39 | ||||||||
| 40 | ||||||||
| 41 | ||||||||
| 43 | ||||||||
| 45 | ||||||||
| 48 | ||||||||
| 84 | ||||||||
| 110 | ||||||||
| 118 | ||||||||
| 136 | ||||||||
| 144 | ||||||||
| 148 | ||||||||
| 153 | ||||||||
| 155 | ||||||||
| 159 | ||||||||
| 163 | ||||||||
| 167 | ||||||||
| 167 | ||||||||
| 167 | ||||||||
| F-1 | ||||||||
| EX-4.1 | ||||||||
| EX-10.16 | ||||||||
| EX-10.17 | ||||||||
| EX-10.18 | ||||||||
| EX-10.19 | ||||||||
| EX-23.1 | ||||||||
i
Table of Contents
ii
Table of Contents
1
Table of Contents
2
Table of Contents
| • | Best Thermal Performance. Our aerogel blankets provide the best thermal performance of any widely used insulation product available on the market today and excel in applications where available space is constrained or thermal performance targets are aggressive. | |
| • | Wide Temperature Range. We offer insulation products that address the entire range of applications within the cryogenic and sub-ambient (−273oC to 90oC), ambient (0oC to 40oC) and hot process (−25oC to 650oC) temperature ranges. | |
| • | Ease of Installation. Our flexible aerogel blankets install faster than rigid insulation materials in the industrial market, which reduces labor costs and total system costs. | |
| • | Compact Design. Our aerogel blankets reduce insulation system volume by 50% to 80% compared to traditional insulation, enabling a reduction in the footprint, size and structural costs of facilities, systems, vehicles and buildings. | |
| • | High Durability. Our aerogel blankets offer excellent compression resistance, tensile strength and vibration resiliency. Our products allow companies to pre-insulate, stack and transport steel pipes destined for use in harsh environments, which significantly reduces installation labor costs in remote areas. | |
| • | Strong Fire Protection. Our Pyrogel XT and Spaceloft A2 product lines were specifically designed to provide strong fire performance in applications within the industrial and building and construction markets, qualifying our products for use in a variety of applications in our target markets. | |
| • | Moisture Resistance. Our aerogel blankets are durably hydrophobic. Our products offer improved thermal performance in insulation systems exposed to the elements or operating in humid environments compared to traditional insulation. | |
| • | Reduced Corrosion Under Insulation, or CUI. Our Pyrogel XT product line is both durably hydrophobic and vapor permeable. These attributes have the potential to reduce the incidence of CUI in hot process applications, which we believe provides our customers with a significant reduction in long-term operating and capital costs. | |
| • | Simplified Logistics. Our products reduce the volume and weight of material purchased, inventoried, transported and installed in the field. In addition, our products reduce the number of stock-keeping units, or SKUs, required to complete a project. Simplified logistics accelerate project timelines, reduce installation costs and improve worker safety. |
3
Table of Contents
| • | Superior product based on proven technology in commercial production. Our aerogel products provide up to five times the thermal performance of widely used traditional insulation in a thin, easy-to-use blanket form. We believe our array of product attributes provides strong competitive advantages over traditional insulation and will enable us to take a growing share of the existing market for insulation in both the industrial and the building and construction markets. Although competing insulation materials may have one or more comparable attributes, we believe that no single insulation material currently available offers all of the properties of our aerogel insulation. | |
| • | Proven and scalable proprietary manufacturing process. Our manufacturing process is proven and has been replicated to meet increasing demand. Our original line in East Providence, Rhode Island, has operated continuously since mid-2008. We successfully commenced operation of our second production line at this facility in March 2011 and immediately began producing commercial quality aerogel blankets. We believe that our proven ability to produce product that meets our clients’ specifications and our increased production capacity will provide customers with the certainty of supply that is required to expand their use of our products. | |
| • | Strong relationships with industry leaders. Through our relationships with industry leading end-use customers, our products have undergone rigorous testing and are now in use at some of the world’s largest oil producers, refiners and petrochemical companies as ranked by a weighted measure of sales, profits, assets and market value. These relationships have shortened the sales cycle with other customers within the industrial market and have helped to facilitate our market penetration. Within the building and construction market, we have partnered with BASF Construction Chemicals to develop products to meet increasingly stringent building standards for thermal performance of retrofit and new-build wall systems. As part of our relationship with BASF Construction Chemicals, we have recently developed a product, Spaceloft A2, targeted at the building and construction market. | |
| • | Capital efficient business model. To respond to increased demand for our products, we successfully commenced operation in late March 2011 of a second production line at our East Providence facility. The expansion is expected to increase our annual production capacity by 20 to 22 million square feet of aerogel blankets at a total construction cost of approximately $31.5 million. We believe that our second production line, at full capacity and at current prices, would be capable of producing in the range of $50 million to $54 million in annual revenue of aerogel blankets. | |
| • | Experienced management and operations team. Each of our executive officers has over 20 years of experience in global industrial companies, specialty chemical companies or related materials science research. This team has worked closely together at Aspen Aerogels for nearly five years, and we believe our dedicated and experienced workforce is an important competitive asset. As of June 30, 2011, we employed 161 dedicated research scientists, engineers, manufacturing line operators, sales and administrative staff and management. |
4
Table of Contents
| • | Expand our manufacturing capacity to meet market demand. Demand for our aerogel products in 2010 grew by approximately 88% compared to 2009 and exceeded our manufacturing capacity. In response, we constructed a second production line at our East Providence facility designed to double our manufacturing capacity. To meet anticipated future growth in demand for our products, we are engaged in the design, engineering and initial procurement phase of a third production line in our East Providence facility and plan to construct a second manufacturing facility in the United States or Europe. |
| • | Increase industrial insulation market penetration. We plan to focus additional resources to achieve a greater share of the industrial insulation market, both through increased sales to our existing customers and sales to new customers. In addition, we anticipate that our growing maintenance-based business will lead to increasing sales of our products into large capital projects, including the construction of new refineries and petrochemical facilities in emerging markets. | |
| • | Leverage strategic relationships in the building and construction market. We have a joint development arrangement with BASF Construction Chemicals to penetrate the market for energy efficient wall systems. We are pursuing additional market opportunities with other leading building materials manufacturers and distributors across multiple regions to address the increasingly stringent regulatory environment governing the thermal performance of buildings. We believe this approach will enable us to leverage their broad technical and distribution capabilities and facilitate market penetration. | |
| • | Expand our sales force, network of distributors and OEM channels. We plan to expand our sales force and distribution network to support growth in the industrial and building and construction markets. We also intend to expand our network of OEM fabricators to pursue opportunities in the transportation, appliance and apparel markets. | |
| • | Continue to develop advanced aerogel compositions, applications and manufacturing technologies. We believe that we are well positioned to leverage a decade’s worth of research and development to commercialize new products, applications and advanced manufacturing technologies. |
| • | We have incurred net losses since our inception, and we may continue to incur net losses in the future and may never reach profitability; | |
| • | We have yet to achieve positive cash flow, and our ability to generate positive cash flow is uncertain; | |
| • | We have a limited operating history. This may make it difficult to evaluate our business and prospects and may expose us to increased risks and uncertainties; | |
| • | The market for insulation products incorporating aerogel blankets is relatively undeveloped, which makes it difficult to forecast adoption rates and demand for our products; |
5
Table of Contents
| • | We rely on sales to a limited number of distributors and contractors for the substantial majority of our revenue, and the loss of one or more significant distributors or several of our smaller distributors could materially harm our business; |
| • | Any significant disruption to our sole manufacturing facility or the failure of our production lines to operate according to our expectation could have a material adverse effect on our results of operations; |
| • | Our products are very expensive relative to other insulation products, which could make it more difficult for us to grow our revenue and achieve broader adoption of our aerogel products; and |
| • | Our directors, officers and principal stockholders have significant voting power and will continue to control a substantial majority of our common stock after this offering. |
6
Table of Contents
| Common stock offered by us | shares (or shares if the underwriters exercise their option to purchase additional shares in full) | |
| Common stock to be outstanding immediately after this offering | shares (or shares if the underwriters exercise their option to purchase additional shares in full) | |
| Use of proceeds | We estimate that the net proceeds to us from this offering, after deducting estimated underwriting discounts and estimated offering expenses payable by us, will be approximately $ million (or approximately $ million if the underwriters exercise their option to purchase additional shares in full) assuming an initial public offering price of $ per share, which is the mid-point of the estimated price range set forth on the cover page of this prospectus. We intend to use the net proceeds from this offering for general corporate purposes, including the construction of additional manufacturing capacity. See “Use of Proceeds” for more information. | |
| Proposed NYSE symbol | “ASPN” |
| • | 14,110,658 shares of our common stock issuable upon the exercise of stock options outstanding as of June 30, 2011 at a weighted-average exercise price of $0.62 per share; | |
| • | shares of our common stock that will be available for future issuance under our 2011 equity incentive plan to be effective upon completion of this offering; and | |
| • | 1,127,324 shares of common stock issuable upon the exercise of warrants outstanding as of June 30, 2011 at a weighted-average exercise price of $0.002 per share. |
| • | assumes the adoption of our restated certificate of incorporation and restated by-laws in connection with the consummation of the offering made hereby; | |
| • | assumes that the closing of the offering made hereby occurs on , 2011 with an initial public offering price per share of $ , the mid-point of the price range set forth on the cover page of this prospectus; | |
| • | gives effect to the automatic conversion of all outstanding shares of our preferred stock into 68,853,493 shares of our common stock on a 1-for-1 basis; | |
| • | gives effect to the issuance of shares of common stock to the holders of our outstanding preferred stock upon the closing of the offering made hereby in satisfaction of accumulated dividends, as required by the terms of the preferred stock; | |
| • | gives effect to the issuance of shares of common stock issuable upon the automatic conversion of our $30.0 million aggregate principal amount of 8.0% convertible subordinated notes due June 2014, which we refer to as our convertible notes, together with accrued |
7
Table of Contents
| interest thereon, upon the closing of the offering made hereby, at a conversion price equal to 87.5% of the initial offering price per share of the common stock offered hereby; |
| • | gives effect to a for reverse split of our common stock, which will take place prior to the closing of the offering made hereby; and | |
| • | assumes no exercise by the underwriters of their option to purchase additional shares. |
8
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands, except share and per share data) | ||||||||||||||||||||
Consolidated statements of operations data: | ||||||||||||||||||||
| Revenue: | ||||||||||||||||||||
| Product | $ | 17,202 | $ | 24,752 | $ | 38,690 | $ | 16,008 | $ | 24,333 | ||||||||||
| Research services | 2,868 | 3,864 | 4,519 | 2,238 | 1,817 | |||||||||||||||
| Total revenue | 20,070 | 28,616 | 43,209 | 18,246 | 26,150 | |||||||||||||||
| Cost of revenue: | ||||||||||||||||||||
| Product | 32,160 | 30,462 | 35,399 | 16,114 | 21,545 | |||||||||||||||
| Research services | 1,169 | 1,788 | 2,119 | 1,005 | 922 | |||||||||||||||
| Impairment charge | 2,524 | — | — | — | — | |||||||||||||||
| Gross profit (loss) | (15,783 | ) | (3,634 | ) | 5,691 | 1,127 | 3,683 | |||||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 2,134 | 2,524 | 2,985 | 1,719 | 1,728 | |||||||||||||||
| Sales and marketing | 4,034 | 3,994 | 4,526 | 2,318 | 2,557 | |||||||||||||||
| General and administrative | 6,180 | 5,430 | 5,675 | 2,813 | 3,443 | |||||||||||||||
| Total operating expenses | 12,348 | 11,948 | 13,186 | 6,850 | 7,728 | |||||||||||||||
| Income (loss) from operations | (28,131 | ) | (15,582 | ) | (7,495 | ) | (5,723 | ) | (4,045 | ) | ||||||||||
| Other income (expense): | ||||||||||||||||||||
| Interest income | 287 | 18 | 170 | 37 | 51 | |||||||||||||||
| Interest expense | (7,400 | ) | (3,075 | ) | (2,585 | ) | (1,404 | ) | (2,269 | ) | ||||||||||
| Total other expense, net | (7,113 | ) | (3,057 | ) | (2,415 | ) | (1,367 | ) | (2,218 | ) | ||||||||||
| Net income (loss) | (35,244 | ) | (18,639 | ) | (9,910 | ) | (7,090 | ) | (6,263 | ) | ||||||||||
| Dividends and accretion on redeemable convertible preferred stock | (2,351 | ) | (2,984 | ) | (57,007 | ) | (39,264 | ) | (68,604 | ) | ||||||||||
| Net income (loss) attributable to common stockholders | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | $ | (46,354 | ) | $ | (74,867 | ) | |||||
Per share data: | ||||||||||||||||||||
| Net income (loss) per share attributable to common stockholders, basic and diluted | $ | (3,389.12 | ) | $ | (2.21 | ) | $ | (2.62 | ) | $ | (1.81 | ) | $ | (2.88 | ) | |||||
| Weighted-average common shares outstanding, basic and diluted | 11,093 | 9,751,616 | 25,574,286 | 25,573,997 | 26,005,705 | |||||||||||||||
9
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands, except share and per share data) | ||||||||||||||||||||
| Pro forma net income (loss) per share, basic and diluted(1) | ||||||||||||||||||||
| Weighted-average common shares outstanding used in computing pro forma net income (loss) per share, basic and diluted(1) | ||||||||||||||||||||
| Year Ended December 31 | Six Months Ended June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
Other operating data: | ||||||||||||||||||||
| Product shipments in square feet(2) | 6,909 | 10,525 | 16,443 | 7,114 | 10,612 | |||||||||||||||
| Adjusted EBITDA(3) | $ | (17,621 | ) | $ | (9,121 | ) | $ | (2,394 | ) | $ | (3,261 | ) | $ | (238 | ) | |||||
| As of June 30, 2011 | ||||||||
Actual | Pro forma | |||||||
| ($ in thousands) | ||||||||
Consolidated balance sheet data: | ||||||||
| Cash and cash equivalents | $ | 29,633 | ||||||
| Working capital(4) | 30,554 | |||||||
| Total assets | 110,871 | |||||||
| Total debt | 39,264 | |||||||
| Preferred stock | 178,390 | |||||||
| Total stockholders’ (deficit) equity | (132,536 | ) | ||||||
| (1) | Pro forma per share data will be computed based upon the number of shares of common stock outstanding immediately after consummation of this offering applied to our historical net income (loss) amounts and will give retroactive effect to the preferred stock and convertible notes conversions (assuming an initial public offering price of $ , the mid-point of the price range set forth on the cover page of this prospectus) and the issuance of the shares of our common stock offered hereby. | |
| The following table presents the calculation of pro forma basic and diluted net income (loss) per share of common stock attributable to our common stockholders: |
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands, except share and per share data) | ||||||||||||||||||||
| Net income (loss) attributable to common stockholders | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | $ | (46,354 | ) | $ | (74,867 | ) | |||||
| Dividends and accretion on redeemable convertible preferred stock | 2,351 | 2,984 | 57,007 | 39,264 | 68,604 | |||||||||||||||
| Interest expense | ||||||||||||||||||||
| Discount on conversion of convertible notes | ||||||||||||||||||||
| Pro forma net income (loss) attributable to common stockholders | ||||||||||||||||||||
| Weighted-average common shares outstanding, basic and diluted | 11,093 | 9,751,616 | 25,574,286 | 25,573,997 | 26,005,705 | |||||||||||||||
| Common shares issued upon conversion of preferred stock and accrued dividends | ||||||||||||||||||||
| Common shares issued upon conversion of convertible notes and interest thereon | ||||||||||||||||||||
| Weighted-average common shares outstanding used in computing pro forma net income (loss) per share, basic and diluted | ||||||||||||||||||||
| Pro forma net income (loss) per share, basic and diluted | ||||||||||||||||||||
10
Table of Contents
| (2) | We price our products and measure our product shipments in square feet. We believe the square foot operating metric allows us and our investors to measure our sales volume on a uniform and consistent basis. |
| (3) | We use Adjusted EBITDA, a non-GAAP financial measure, as a means to assess our operating performance. We define Adjusted EBITDA as net income (loss) before depreciation and amortization expense, interest expense, stock-based compensation expense and impairment charges. Adjusted EBITDA is a supplemental measure of our performance that is not required by, or presented in accordance with, GAAP. Adjusted EBITDA should not be considered as an alternative to net income (loss) or any other measure of financial performance calculated and presented in accordance with GAAP. In addition, our definition and presentation of Adjusted EBITDA may not be comparable to similarly titled measures presented by other companies. |
| We use Adjusted EBITDA as a measure of operating performance, because it does not include the impact of items that we do not consider indicative of our core operating performance, for planning purposes, including the preparation of our annual operating budget, to allocate resources to enhance the financial performance of our business and as a performance measure under our bonus plan. We also believe that the presentation of Adjusted EBITDA provides useful information to investors with respect to our results of operations and in assessing the performance and value of our business. Various measures of EBITDA are widely used by investors to measure a company’s operating performance without regard to items that can vary substantially from company to company depending upon financing and accounting methods, book values of assets, capital structures and the methods by which assets were acquired. | ||
| We understand that, although measures similar to Adjusted EBITDA are frequently used by investors and securities analysts in their evaluation of companies, Adjusted EBITDA has limitations as an analytical tool, and you should not consider it in isolation or as a substitute for GAAP income from operations or an analysis of our results of operations as reported under GAAP. Some of these limitations are: | ||
| • Adjusted EBITDA does not reflect our historical cash expenditures or future requirements for capital expenditures or other contractual commitments; | ||
| • Adjusted EBITDA does not reflect changes in, or cash requirements for, our working capital needs; | ||
| • Adjusted EBITDA does not reflect stock-based compensation expense; | ||
| • Adjusted EBITDA does not reflect our tax expense or cash requirements to pay our income taxes; | ||
| • Adjusted EBITDA does not reflect our interest expense, or the cash requirements necessary to service interest or principal payments on our debt; | ||
| • Although depreciation, amortization and impairment are non-cash charges, the assets being depreciated, amortized or impaired will often have to be replaced in the future, and Adjusted EBITDA does not reflect any cash requirements for these replacements; and | ||
| • Other companies in our industry may calculate EBITDA or Adjusted EBITDA differently than we do, limiting their usefulness as a comparative measure. | ||
| Because of these limitations, our Adjusted EBITDA should not be considered as a measure of discretionary cash available to us to reinvest in the growth of our business or as a measure of cash available for us to meet our obligations. | ||
| To properly and prudently evaluate our business, we encourage you to review the GAAP financial statements included elsewhere in this prospectus and not to rely on any single financial measure to evaluate our business. | ||
| The following table presents a reconciliation of net income (loss), the most directly comparable GAAP measure, to Adjusted EBITDA for the periods presented: |
| Six Months | ||||||||||||||||||||
| Year Ended December 31 | Ended June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Net income (loss) | (35,244 | ) | (18,639 | ) | (9,910 | ) | (7,090 | ) | (6,263 | ) | ||||||||||
| Depreciation and amortization | 7,059 | 5,630 | 4,633 | 2,261 | 3,365 | |||||||||||||||
| Stock-based compensation | 927 | 831 | 468 | 201 | 442 | |||||||||||||||
| Interest expense, net | 7,113 | 3,057 | 2,415 | 1,367 | 2,218 | |||||||||||||||
| Impairment charge | 2,524 | — | — | — | — | |||||||||||||||
| Adjusted EBITDA | $ | (17,621 | ) | $ | (9,121 | ) | $ | (2,394 | ) | $ | (3,261 | ) | $ | (238 | ) | |||||
| (4) | Working capital means current assets minus current liabilities. |
11
Table of Contents
12
Table of Contents
| • | maintain and expand our current relationships and develop new relationships with direct and end-use customers; | |
| • | increase our penetration of the industrial market, the building and construction market and other markets for our products; | |
| • | maintain and increase our manufacturing capacity to meet existing and anticipated future demand for our products; | |
| • | continue to develop our aerogel insulation products and increase our research and development activities; | |
| • | identify new partnership and market opportunities for our products; | |
| • | develop new applications for our products and our aerogel technology; | |
| • | execute our business and marketing strategies successfully; | |
| • | respond to competitive developments; and | |
| • | attract, integrate, retain and motivate qualified personnel. |
13
Table of Contents
| • | the price of our aerogel insulation compared to traditional insulation materials, including the tendency of end-users in some markets to opt for the frequently lower short-term costs of traditional insulation materials even when the long-term and overall costs of our products are lower and the performance attributes of our products are superior; | |
| • | the regulation of energy efficiency and building standards in the industrial and the building and construction markets; | |
| • | the availability of government funding and incentives to support the use of high-performance insulation materials, such as our aerogel blankets; and | |
| • | fluctuations in economic and market conditions, including changes in the cost of energy, the relative value of energy efficiency measures, foreign exchange rates and the cost of the raw materials for our products. |
14
Table of Contents
15
Table of Contents
16
Table of Contents
17
Table of Contents
18
Table of Contents
19
Table of Contents
20
Table of Contents
21
Table of Contents
22
Table of Contents
23
Table of Contents
24
Table of Contents
25
Table of Contents
26
Table of Contents
27
Table of Contents
28
Table of Contents
29
Table of Contents
30
Table of Contents
| • | foreign currency fluctuations, which could result in reduced revenue and increased operating expense; | |
| • | labor rules and collective bargaining arrangements in foreign jurisdictions, if we seek to expand our manufacturing facilities overseas; | |
| • | the effect of applicable foreign tax structures, including tax rates that may be higher than tax rates in the United States or taxes that may be duplicative of those imposed in the United States; | |
| • | tariffs and trade barriers; | |
| • | general economic and political conditions in each country, which may interfere with, among other things, our supply chain, our customers and all of our activities in a particular location; | |
| • | inadequate intellectual property protection in foreign countries; | |
| • | the difficulties and increased expense in complying with a variety of domestic and foreign laws, regulations and trade standards, including the Foreign Corrupt Practices Act; and | |
| • | terrorist activity and political unrest, particularly given the use of our products at oil and gas facilities. |
31
Table of Contents
| • | incur additional debt; | |
| • | pay dividends and make distributions; | |
| • | redeem or repurchase capital stock; | |
| • | create liens; | |
| • | enter into transactions with affiliates; and | |
| • | merge or consolidate with or into other entities. |
32
Table of Contents
33
Table of Contents
| • | volume and timing of orders for our products; | |
| • | quarterly and yearly variations in our or our competitors’ results of operations; | |
| • | our announcement or our competitors’ announcements regarding new products, product enhancements, significant contracts, number of distributors, acquisitions or strategic investments; | |
| • | announcements of technological innovations relating to aerogels, thermal management and energy conservation insulation; | |
| • | results of operations that vary from the expectations of securities analysts and investors; | |
| • | the periodic nature of our sales cycles, in particular for capital projects in the industrial market; | |
| • | our ability to develop, obtain regulatory clearance or approval for and market new and enhanced products on a timely basis; | |
| • | future sales of our common stock, including sales by our executive officers, directors and significant stockholders; | |
| • | announcements by third parties of significant claims or proceedings against us, including with regards to intellectual property and product liability; | |
| • | changes in accounting principles; and | |
| • | general U.S. and global economic conditions and other factors, including factors unrelated to our operating performance or the operating performance of our competitors. |
34
Table of Contents
35
Table of Contents
| • | procedures for advance notification of stockholder nominations and proposals; | |
| • | the inability of our stockholders to call a special meeting of the stockholders and the inability of our stockholders to act by written consent; | |
| • | the ability of our board of directors to create new directorships and to fill any vacancies on the board of directors; | |
| • | the ability of our board of directors to amend our restated by-laws without stockholder approval; and | |
| • | the ability of our board of directors to issue up to shares of preferred stock without stockholder approval upon the terms and conditions and with the rights, privileges and preferences as our board of directors may determine. |
36
Table of Contents
37
Table of Contents
| • | our expectations as to the future growth of our business; | |
| • | the expected future growth of the market for aerogel insulation, insulation generally, and energy efficiency solutions; | |
| • | our belief that our products provide strong competitive advantages over traditional insulation materials; |
| • | our expectations that our second production line will be capable of operating at full capacity by the end of 2011 and that our first two production lines will then be capable of an annual production capacity of 40 to 44 million square feet of aerogel blankets, depending on product mix; |
| • | our expectation that upon completion of our third production line, our annual manufacturing capacity will increase by between 20 and 26 million square feet of aerogel blankets; | |
| • | our ability to produce $50 to $54 million in annual revenue of aerogel blankets at current prices on our second production line when operating at full capacity; | |
| • | our expectation that the construction of our third production line will be completed during 2012 and at a cost of approximately $45 million to $50 million; | |
| • | our plans to construct a second manufacturing facility in the United States or Europe, based on proximity to raw material suppliers, proximity to customers, labor and construction costs and available governmental incentives, and our estimate that design, development and construction costs will range from $150 million to $200 million; | |
| • | the expected energy and cost savings of our products; and | |
| • | the expected future development of new aerogel technologies. |
38
Table of Contents
39
Table of Contents
40
Table of Contents
| • | on an actual basis; |
| • | on an unaudited pro forma basis to give effect upon the completion of this offering to (i) the automatic conversion of all shares of our preferred stock into shares of our common stock and the issuance of shares of common stock in satisfaction of accumulated dividends on such preferred stock; (ii) the automatic conversion of our convertible notes and all accrued but unpaid interest thereon into shares of our common stock; and (iii) the sale of shares of our common stock offered by us in this offering at an assumed initial public offering price of $ per share, the mid-point of the price range set forth on the cover page of this prospectus, after deducting estimated underwriting discounts and estimated offering expenses payable by us. |
| As of June 30, 2011 | ||||||||
Actual | Pro forma | |||||||
| (Unaudited) | ||||||||
| (In thousands, except share and per share data) | ||||||||
| Cash and cash equivalents(1) | $ | 29,633 | $ | |||||
| Long-term debt including current portion | $ | 8,724 | $ | |||||
| Convertible notes(2) | 30,540 | |||||||
| Series B redeemable convertible preferred stock, $0.001 par value; 17,000,000 shares authorized, 16,010,292 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma(3) | 42,161 | |||||||
| Series A redeemable convertible preferred stock, $0.001 par value, 52,843,201 shares authorized, issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma(3) | 136,229 | |||||||
| Stockholders’ (deficit) equity: | ||||||||
| Common stock, $0.001 par value; 114,000,000 shares authorized, 26,119,304 shares issued and outstanding, actual; and shares issued and outstanding, pro forma(1)(2)(3) | 26 | |||||||
| Additional paid-in capital(1) | 69,876 | |||||||
| Accumulated deficit | (202,438 | ) | ||||||
| Total stockholders’ (deficit) equity(1) | $ | (132,536 | ) | $ | ||||
| Total capitalization(1) | $ | 85,118 | $ | |||||
| (1) | To the extent we change the number of shares of common stock sold by us in this offering from the shares we expect to sell or we change the initial public offering price from the $ per share assumed initial public offering price, representing the mid-point of the estimated price range set forth on the cover page of this prospectus, or any combination of these events occurs, the net proceeds to us from this offering and consequently the cash and cash equivalents and each of additional paid-in capital, total stockholders’ equity and total capitalization may increase or decrease. A $1.00 increase |
41
Table of Contents
| (decrease) in the assumed initial public offering price per share of the common stock would increase (decrease) the net proceeds that we receive in this offering and each of our unaudited pro forma cash and cash equivalents, additional paid-in capital, total stockholders’ equity and total capitalization by approximately $ million, assuming that the number of shares offered by us under this prospectus remains the same. An increase (decrease) of 1,000,000 shares in the expected number of shares to be sold in the offering, assuming no change in the assumed initial public offering price per share, would increase (decrease) our net proceeds from this offering and consequently the cash and cash equivalents and each of additional paid-in capital, total stockholders’ equity and total capitalization by approximately $ million. | ||
| (2) | The unpaid principal amount of the convertible notes, together with any accrued but unpaid interest thereon, will be automatically converted into common stock upon the closing of the offering made hereby at a conversion price equal to 87.5% of the initial offering price per share to the public in this offering. | |
| A $1.00 increase from the assumed initial public offering price of $ per share would decrease the number of shares of common stock issuable upon the conversion of the convertible notes by shares and $1.00 decrease from the assumed initial public offering price of $ per share would increase the number of shares of common stock issuable upon the conversion of the convertible notes by shares, in each case, assuming a , 2011 closing date of the offering. For each day after the assumed , 2011 closing date, the number of shares of common stock issuable upon the convertible notes would increase by shares, assuming an initial offering price of $ per share. The actual number of shares of common stock to be issued upon the conversion of the convertible notes will be based on the amount of accrued but unpaid interest then outstanding and the actual initial public offering price. |
| (3) | Each series of our preferred stock will convert upon the completion of this offering on a1-for-1 basis. The terms of our existing preferred stock require us, upon the closing of the offering made hereby, to issue additional shares of common stock to the holders of such preferred stock in satisfaction of accumulated dividends on such preferred stock. The accumulated dividends were approximately $5.8 million at June 30, 2011 and accumulate at the rate of approximately $11,000 per day thereafter. The common stock issued in satisfaction of those dividends will be valued at the public offering price per share in this offering. |
| A $1.00 increase from the assumed initial public offering price of $ per share would decrease the number of shares of common stock to be issued to the holders of preferred stock in satisfaction of accumulated dividends on such preferred stock by approximately shares and a $1.00 decrease from the assumed initial public offering price of $ per share would increase the number of shares of common stock to be issued to the holders of our preferred stock in satisfaction of accumulated dividends on such preferred stock by approximately shares, in each case, assuming the closing date of the offering hereby occurs on , 2011. For each day after the assumed , 2011 closing date, the number of shares of common stock issuable to the holders of preferred stock in satisfaction of accumulated dividends on such preferred stock would increase by shares, assuming an initial offering price of $ per share. The actual number of shares of common stock to be issued to the holders of our preferred stock will be based on the amount of accrued dividends then outstanding and the actual initial public offering price. |
42
Table of Contents
| Assumed initial public offering price per share | $ | |||||||
| Pro forma net tangible book value per share as of June 30, 2011 | ||||||||
| Increase per share attributable to cash payments by new investors in this offering | ||||||||
| Pro forma as adjusted net tangible book value per share after this offering | ||||||||
| Dilution in pro forma net tangible book value per share to new investors | $ | |||||||
43
Table of Contents
| Shares Purchased | Total Consideration | Average Price | ||||||||||||||||||
Number | Percentage | Amount | Percentage | per Share | ||||||||||||||||
| Existing stockholders | % | $ | % | $ | ||||||||||||||||
| New investors | ||||||||||||||||||||
| Total | % | $ | % | $ | ||||||||||||||||
44
Table of Contents
45
Table of Contents
| Six Months Ended | ||||||||||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||||||||||
2006 | 2007 | 2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||||||||
| ($ in thousands, except share and per share data) | ||||||||||||||||||||||||||||
Consolidated statements of operations data: | ||||||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||||||
| Product | $ | 5,571 | $ | 9,075 | $ | 17,202 | $ | 24,752 | $ | 38,690 | $ | 16,008 | $ | 24,333 | ||||||||||||||
| Research services | 5,806 | 4,743 | 2,868 | 3,864 | 4,519 | 2,238 | 1,817 | |||||||||||||||||||||
| Total revenue | 11,377 | 13,818 | 20,070 | 28,616 | 43,209 | 18,246 | 26,150 | |||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||||||
| Product | 17,490 | 15,356 | 32,160 | 30,462 | 35,399 | 16,114 | 21,545 | |||||||||||||||||||||
| Research services | 2,316 | 1,573 | 1,169 | 1,788 | 2,119 | 1,005 | 922 | |||||||||||||||||||||
| Impairment charge | — | — | 2,524 | — | — | — | — | |||||||||||||||||||||
| Gross profit (loss) | (8,429 | ) | (3,111 | ) | (15,783 | ) | (3,634 | ) | 5,691 | 1,127 | 3,683 | |||||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||
| Research and development | 6,176 | 3,203 | 2,134 | 2,524 | 2,985 | 1,719 | 1,728 | |||||||||||||||||||||
| Sales and marketing | 5,726 | 4,868 | 4,034 | 3,994 | 4,526 | 2,318 | 2,557 | |||||||||||||||||||||
| General and administrative | 6,678 | 7,158 | 6,180 | 5,430 | 5,675 | 2,813 | 3,443 | |||||||||||||||||||||
| Total operating expenses | 18,580 | 15,229 | 12,348 | 11,948 | 13,186 | 6,850 | 7,728 | |||||||||||||||||||||
| Income (loss) from operations | (27,009 | ) | (18,340 | ) | (28,131 | ) | (15,582 | ) | (7,495 | ) | (5,723 | ) | (4,045 | ) | ||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||||||
| Interest income | 271 | 56 | 287 | 18 | 170 | 37 | 51 | |||||||||||||||||||||
| Interest expense | (10,002 | ) | (10,745 | ) | (7,400 | ) | (3,075 | ) | (2,585 | ) | (1,404 | ) | (2,269 | ) | ||||||||||||||
| Total other expense, net | (9,731 | ) | (10,689 | ) | (7,113 | ) | (3,057 | ) | (2,415 | ) | (1,367 | ) | (2,218 | ) | ||||||||||||||
| Net income (loss) | (36,740 | ) | (29,029 | ) | (35,244 | ) | (18,639 | ) | (9,910 | ) | (7,090 | ) | (6,263 | ) | ||||||||||||||
| Dividends and accretion on redeemable convertible preferred stock | (1,812 | ) | (1,812 | ) | (2,351 | ) | (2,984 | ) | (57,007 | ) | (39,264 | ) | (68,604 | ) | ||||||||||||||
| Net income (loss) attributable to common stockholders | $ | (38,552 | ) | $ | (30,841 | ) | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | $ | (46,354 | ) | $ | (74,867 | ) | |||||||
Per share data: | ||||||||||||||||||||||||||||
| Net income (loss) per share attributable to common stockholders, basic and diluted | $ | (4,163.04 | ) | $ | (3,289.81 | ) | $ | (3,389.12 | ) | $ | (2.21 | ) | $ | (2.62 | ) | $ | (1.81 | ) | $ | (2.88 | ) | |||||||
| Weighted-average common shares outstanding, basic and diluted | 9,261 | 9,375 | 11,093 | 9,751,616 | 25,574,286 | 25,573,997 | 26,005,705 | |||||||||||||||||||||
| Pro forma net income (loss) per share, basic and diluted(1) | ||||||||||||||||||||||||||||
| Weighted-average common shares outstanding used in computing pro forma net income (loss) per share, basic and diluted(1) | ||||||||||||||||||||||||||||
| December 31 | June 30 | |||||||||||||||||||||||
2006 | 2007 | 2008 | 2009 | 2010 | 2011 | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
Consolidated balance sheet data: | ||||||||||||||||||||||||
| Cash and cash equivalents | $ | 5,839 | $ | 794 | $ | 11,988 | $ | 27,502 | $ | 26,800 | $ | 29,633 | ||||||||||||
| Working capital(2) | (34,593 | ) | (55,213 | ) | 9,404 | 21,766 | 24,723 | 30,554 | ||||||||||||||||
| Total assets | 61,273 | 49,296 | 54,624 | 64,735 | 88,795 | 110,871 | ||||||||||||||||||
| Total debt | 26,502 | 29,195 | 755 | 509 | 8,067 | 39,264 | ||||||||||||||||||
| Preferred stock | 61,051 | 62,863 | 184,269 | 31,681 | 109,786 | 178,390 | ||||||||||||||||||
| Total stockholders’ (deficit) equity | (93,185 | ) | (123,055 | ) | (159,655 | ) | 6,153 | (58,103 | ) | (132,536 | ) | |||||||||||||
| (1) | Pro forma per share data will be computed based upon the number of shares of common stock outstanding immediately after consummation of this offering applied to our historical net income (loss) amounts and will give retroactive effect to the preferred stock and convertible notes conversions (assuming an initial public offering price of $ per share, the mid-point |
46
Table of Contents
| of the price range set forth on the cover page of this prospectus) and the issuance of the shares of our common stock offered hereby. |
| Six Months | ||||||||||||||||||||||||||||
| Year Ended December 31 | Ended June 30 | |||||||||||||||||||||||||||
2006 | 2007 | 2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||||||||
| ($ in thousands, except share and per share data) | ||||||||||||||||||||||||||||
| Net income (loss) attributable to common stockholders | $ | (38,552 | ) | $ | (30,841 | ) | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | $ | (46,354 | ) | $ | (74,867 | ) | |||||||
| Dividends and accretion on redeemable convertible preferred stock | 1,812 | 1,812 | 2,351 | 2,984 | 57,007 | 39,264 | 68,604 | |||||||||||||||||||||
| Interest expense | ||||||||||||||||||||||||||||
| Discount on conversion of convertible notes | ||||||||||||||||||||||||||||
| Pro forma net income (loss) attributable to common stockholders | ||||||||||||||||||||||||||||
| Weighted-average common shares outstanding, basic and diluted | 9,261 | 9,375 | 11,093 | 9,751,616 | 25,574,286 | 25,573,997 | 26,005,705 | |||||||||||||||||||||
| Common shares issued upon conversion of preferred stock and accrued dividends | ||||||||||||||||||||||||||||
| Common shares issued upon conversion of convertible notes and interest thereon | ||||||||||||||||||||||||||||
| Weighted-average common shares outstanding used in computing pro forma net income (loss) per share, basic and diluted | ||||||||||||||||||||||||||||
| Pro forma net income (loss) per share, basic and diluted | ||||||||||||||||||||||||||||
| (2) | Working capital means current assets minus current liabilities. |
47
Table of Contents
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
48
Table of Contents
49
Table of Contents
50
Table of Contents
| Six Months | ||||||||||||||||||||
| Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Revenue: | ||||||||||||||||||||
| Product | $ | 17,202 | $ | 24,752 | $ | 38,690 | $ | 16,008 | $ | 24,333 | ||||||||||
| Research services | 2,868 | 3,864 | 4,519 | 2,238 | 1,817 | |||||||||||||||
| Total revenue | $ | 20,070 | $ | 28,616 | $ | 43,209 | $ | 18,246 | $ | 26,150 | ||||||||||
51
Table of Contents
52
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Total costs incurred | $ | 3,193 | $ | 1,672 | $ | 2,005 | $ | 346 | $ | 171 | ||||||||||
| Expenditures offset by cost sharing funding received: | ||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||
| Product | $ | 43 | $ | 20 | $ | 33 | $ | 6 | $ | 7 | ||||||||||
| Research services | 1,227 | 637 | 1,124 | 185 | 51 | |||||||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 1,214 | 620 | 475 | 82 | 79 | |||||||||||||||
| Sales and marketing | 342 | 193 | 170 | 33 | 15 | |||||||||||||||
| General and administrative | 367 | 202 | 203 | 40 | 19 | |||||||||||||||
| Total expenditures offset by cost sharing | $ | 3,193 | $ | 1,672 | $ | 2,005 | $ | 346 | $ | 171 | ||||||||||
53
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| (Square feet in thousands) | ||||||||||||||||||||
| Product shipments in square feet | 6,909 | 10,525 | 16,443 | 7,114 | 10,612 | |||||||||||||||
54
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| ($ in thousands) | ||||||||||||
| Net income (loss) | $ | (35,244 | ) | $ | (18,639 | ) | $ | (9,910 | ) | |||
| Depreciation and amortization | 7,059 | 5,630 | 4,633 | |||||||||
| Stock-based compensation(1) | 927 | 831 | 468 | |||||||||
| Interest expense, net | 7,113 | 3,057 | 2,415 | |||||||||
| Impairment charge | 2,524 | — | — | |||||||||
| Adjusted EBITDA | $ | (17,621 | ) | $ | (9,121 | ) | $ | (2,394 | ) | |||
| (1) | Represents non-cash stock-based compensation related to stock option grants. |
| Three Months Ended | ||||||||||||||||||||||||||||||||||||||||
| 2009 | 2010 | 2011 | ||||||||||||||||||||||||||||||||||||||
| March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | |||||||||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||||||||||
| Net income (loss) | $ | (5,230 | ) | $ | (5,892 | ) | $ | (3,866 | ) | $ | (3,651 | ) | $ | (3,667 | ) | $ | (3,423 | ) | $ | (1,337 | ) | $ | (1,483 | ) | $ | (1,721 | ) | $ | (4,542 | ) | ||||||||||
| Depreciation and amortization | 1,393 | 1,379 | 1,580 | 1,278 | 1,137 | 1,124 | 1,137 | 1,235 | 1,310 | 2,055 | ||||||||||||||||||||||||||||||
| Stock-based compensation(1) | 99 | 91 | 125 | 516 | 96 | 105 | 122 | 145 | 190 | 252 | ||||||||||||||||||||||||||||||
| Interest expense, net | 812 | 773 | 750 | 722 | 696 | 671 | 606 | 442 | 451 | 1,767 | ||||||||||||||||||||||||||||||
| Impairment charge | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Adjusted EBITDA | $ | (2,926 | ) | $ | (3,649 | ) | $ | (1,411 | ) | $ | (1,135 | ) | $ | (1,738 | ) | $ | (1,523 | ) | $ | 528 | $ | 339 | $ | 230 | $ | (468 | ) | |||||||||||||
| (1) | Represents non-cash stock-based compensation related to stock option grants. |
55
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Revenue: | ||||||||||||||||||||
| Product | $ | 17,202 | $ | 24,752 | $ | 38,690 | $ | 16,008 | $ | 24,333 | ||||||||||
| Research services | 2,868 | 3,864 | 4,519 | 2,238 | 1,817 | |||||||||||||||
| Total revenue | 20,070 | 28,616 | 43,209 | 18,246 | 26,150 | |||||||||||||||
| Cost of revenue: | ||||||||||||||||||||
| Product | 32,160 | 30,462 | 35,399 | 16,114 | 21,545 | |||||||||||||||
| Research services | 1,169 | 1,788 | 2,119 | 1,005 | 922 | |||||||||||||||
| Impairment charge | 2,524 | — | — | — | — | |||||||||||||||
| Gross profit (loss) | (15,783 | ) | (3,634 | ) | 5,691 | 1,127 | 3,683 | |||||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 2,134 | 2,524 | 2,985 | 1,719 | 1,728 | |||||||||||||||
| Sales and marketing | 4,034 | 3,994 | 4,526 | 2,318 | 2,557 | |||||||||||||||
| General and administrative | 6,180 | 5,430 | 5,675 | 2,813 | 3,443 | |||||||||||||||
| Total operating expenses | 12,348 | 11,948 | 13,186 | 6,850 | 7,728 | |||||||||||||||
| Income (loss) from operations | (28,131 | ) | (15,582 | ) | (7,495 | ) | (5,723 | ) | (4,045 | ) | ||||||||||
| Other income (expense): | ||||||||||||||||||||
| Interest income | 287 | 18 | 170 | 37 | 51 | |||||||||||||||
| Interest expense | (7,400 | ) | (3,075 | ) | (2,585 | ) | (1,404 | ) | (2,269 | ) | ||||||||||
| Total other expense, net | (7,113 | ) | (3,057 | ) | (2,415 | ) | (1,367 | ) | (2,218 | ) | ||||||||||
| Net income (loss) | $ | (35,244 | ) | $ | (18,639 | ) | $ | (9,910 | ) | $ | (7,090 | ) | $ | (6,263 | ) | |||||
56
Table of Contents
| Six Months Ended June 30 | Six Months Ended June 30 | |||||||||||||||||||||||
2010 | 2011 | $ Change | % Change | 2010 | 2011 | |||||||||||||||||||
| (Percentage of total revenue) | ||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 16,008 | $ | 24,333 | $ | 8,325 | 52 | % | 88 | % | 93 | % | ||||||||||||
| Research services | 2,238 | 1,817 | (421 | ) | (19 | %) | 12 | % | 7 | % | ||||||||||||||
| Total revenue | 18,246 | 26,150 | 7,904 | 43 | % | 100 | % | 100 | % | |||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||
| Product | 16,114 | 21,545 | 5,431 | 34 | % | 88 | % | 82 | % | |||||||||||||||
| Research services | 1,005 | 922 | (83 | ) | (8 | %) | 6 | % | 4 | % | ||||||||||||||
| Gross profit | 1,127 | 3,683 | 2,556 | 227 | % | 6 | % | 14 | % | |||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||
| Research and development | 1,719 | 1,728 | 9 | 1 | % | 9 | % | 7 | % | |||||||||||||||
| Sales and marketing | 2,318 | 2,557 | 239 | 10 | % | 13 | % | 10 | % | |||||||||||||||
| General and administrative | 2,813 | 3,443 | 630 | 22 | % | 15 | % | 13 | % | |||||||||||||||
| Total operating expenses | 6,850 | 7,728 | 878 | 13 | % | 38 | % | 30 | % | |||||||||||||||
| Income (loss) from operations | (5,723 | ) | (4,045 | ) | 1,678 | (29 | %) | (31 | %) | (15 | %) | |||||||||||||
| Other income (expense): | ||||||||||||||||||||||||
| Interest income | 37 | 51 | 14 | 38 | % | 0 | % | 0 | % | |||||||||||||||
| Interest expense | (1,404 | ) | (2,269 | ) | (865 | ) | 62 | % | (8 | %) | (9 | %) | ||||||||||||
| Total other expense, net | (1,367 | ) | (2,218 | ) | (851 | ) | 62 | % | (7 | %) | (8 | %) | ||||||||||||
| Net income (loss) | $ | (7,090 | ) | $ | (6,263 | ) | $ | 827 | (12 | %) | (39 | %) | (24 | %) | ||||||||||
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 16,008 | 88 | % | $ | 24,333 | 93 | % | $ | 8,325 | 52 | % | ||||||||||||
| Research services | 2,238 | 12 | % | 1,817 | 7 | % | (421 | ) | (19 | %) | ||||||||||||||
| Total revenue | $ | 18,246 | 100 | % | $ | 26,150 | 100 | % | $ | 7,904 | 43 | % | ||||||||||||
| Six Months | ||||||||||||||||
| Ended | ||||||||||||||||
| June 30 | Change | |||||||||||||||
2010 | 2011 | Amount | Percentage | |||||||||||||
| (Square feet in thousands) | ||||||||||||||||
| Product shipments in square feet | 7,114 | 10,612 | 3,498 | 49 | % | |||||||||||
57
Table of Contents
| Six Months Ended June 30 | ||||||||||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||||||||||
| Percentage | Percentage | Percentage | Percentage | |||||||||||||||||||||||||||||
| of Related | of Total | of Related | of Total | Change | ||||||||||||||||||||||||||||
Amount | Revenue | Revenue | Amount | Revenue | Revenue | Amount | Percentage | |||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||||||||||
| Product | $ | 16,114 | 101 | % | 88 | % | $ | 21,545 | 89 | % | 82 | % | $ | 5,431 | 34 | % | ||||||||||||||||
| Research services | 1,005 | 45 | % | 6 | % | 922 | 51 | % | 4 | % | (83 | ) | (8 | )% | ||||||||||||||||||
| Total cost of revenue | $ | 17,119 | 94 | % | 94 | % | $ | 22,467 | 86 | % | 86 | % | $ | 5,348 | 31 | % | ||||||||||||||||
58
Table of Contents
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Gross profit | $ | 1,127 | 6 | % | $ | 3,683 | 14 | % | $ | 2,556 | 227 | % | ||||||||||||
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| R&D expenses | $ | 1,719 | 9 | % | $ | 1,728 | 7 | % | $ | 9 | 1 | % | ||||||||||||
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Sales and marketing expenses | $ | 2,318 | 13 | % | $ | 2,557 | 10 | % | $ | 239 | 10 | % | ||||||||||||
59
Table of Contents
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| G&A expenses | $ | 2,813 | 15 | % | $ | 3,443 | 13 | % | $ | 630 | 22 | % | ||||||||||||
| Six Months Ended June 30 | ||||||||||||||||||||||||
| 2010 | 2011 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||
| Interest income | $ | 37 | 0 | % | $ | 51 | 0 | % | $ | 14 | 38 | % | ||||||||||||
| Interest expense | (1,404 | ) | (8 | )% | (2,269 | ) | (9 | )% | (865 | ) | 62 | % | ||||||||||||
| Total other expense, net | $ | (1,367 | ) | (7 | )% | $ | (2,218 | ) | (8 | )% | $ | (851 | ) | 62 | % | |||||||||
60
Table of Contents
| Year Ended December 31 | Year Ended December 31 | |||||||||||||||||||||||
2009 | 2010 | $ Change | % Change | 2009 | 2010 | |||||||||||||||||||
| (Percentage of total revenue) | ||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 24,752 | $ | 38,690 | $ | 13,938 | 56 | % | 86 | % | 90 | % | ||||||||||||
| Research services | 3,864 | 4,519 | 655 | 17 | % | 14 | % | 10 | % | |||||||||||||||
| Total revenue | 28,616 | 43,209 | 14,593 | 51 | % | 100 | % | 100 | % | |||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||
| Product | 30,462 | 35,399 | 4,937 | 16 | % | 106 | % | 82 | % | |||||||||||||||
| Research services | 1,788 | 2,119 | 331 | 19 | % | 6 | % | 5 | % | |||||||||||||||
| Gross profit (loss) | (3,634 | ) | 5,691 | 9,325 | 257 | % | (13 | )% | 13 | % | ||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||
| Research and development | 2,524 | 2,985 | 461 | 18 | % | 9 | % | 7 | % | |||||||||||||||
| Sales and marketing | 3,994 | 4,526 | 532 | 13 | % | 14 | % | 10 | % | |||||||||||||||
| General and administrative | 5,430 | 5,675 | 245 | 5 | % | 19 | % | 13 | % | |||||||||||||||
| Total operating expenses | 11,948 | 13,186 | 1,238 | 10 | % | 42 | % | 31 | % | |||||||||||||||
| Income (loss) from operations | (15,582 | ) | (7,495 | ) | 8,087 | 52 | % | (54 | )% | (17 | )% | |||||||||||||
| Other income (expense): | ||||||||||||||||||||||||
| Interest income | 18 | 170 | 152 | 844 | % | 0 | % | 0 | % | |||||||||||||||
| Interest expense | (3,075 | ) | (2,585 | ) | 490 | 16 | % | (11 | )% | (6 | )% | |||||||||||||
| Total other expense, net | (3,057 | ) | (2,415 | ) | 642 | 21 | % | (11 | )% | (6 | )% | |||||||||||||
| Net income (loss) | $ | (18,639 | ) | $ | (9,910 | ) | $ | 8,729 | 47 | % | (65 | )% | (23 | )% | ||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 24,752 | 86 | % | $ | 38,690 | 90 | % | $ | 13,938 | 56 | % | ||||||||||||
| Research services | 3,864 | 14 | % | 4,519 | 10 | % | 655 | 17 | % | |||||||||||||||
| Total revenue | $ | 28,616 | 100 | % | $ | 43,209 | 100 | % | $ | 14,593 | 51 | % | ||||||||||||
| Year Ended | ||||||||||||||||
| December 31 | Change | |||||||||||||||
2009 | 2010 | Amount | Percentage | |||||||||||||
| Product shipments in square feet (in thousands) | 10,525 | 16,443 | 5,918 | 56 | % | |||||||||||
61
Table of Contents
| Year Ended December 31 | ||||||||||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||||||||||
| Percentage | Percentage | Percentage | Percentage | |||||||||||||||||||||||||||||
| of Related | of Total | of Related | of Total | Change | ||||||||||||||||||||||||||||
Amount | Revenue | Revenue | Amount | of Revenue | Revenue | Amount | Percentage | |||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||||||||||
| Product | $ | 30,462 | 123 | % | 106 | % | $ | 35,399 | 91 | % | 82 | % | $ | 4,937 | 16 | % | ||||||||||||||||
| Research services | 1,788 | 46 | % | 6 | % | 2,119 | 47 | % | 5 | % | 331 | 19 | % | |||||||||||||||||||
| Total cost of revenue | $ | 32,250 | 113 | % | 113 | % | $ | 37,518 | 87 | % | 87 | % | $ | 5,268 | 16 | % | ||||||||||||||||
62
Table of Contents
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Gross profit (loss) | $ | (3,634 | ) | (13 | )% | $ | 5,691 | 13 | % | $ | 9,325 | 257 | % | |||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| R&D expenses | $ | 2,524 | 9 | % | $ | 2,985 | 7 | % | $ | 461 | 18 | % | ||||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Sales and marketing expenses | $ | 3,994 | 14 | % | $ | 4,526 | 10 | % | $ | 532 | 13 | % | ||||||||||||
63
Table of Contents
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| G&A expenses | $ | 5,430 | 19 | % | $ | 5,675 | 13 | % | $ | 245 | 5 | % | ||||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2009 | 2010 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||
| Interest income | $ | 18 | 0 | % | $ | 170 | 0 | % | $ | 152 | 844 | % | ||||||||||||
| Interest expense | (3,075 | ) | (11 | )% | (2,585 | ) | (6 | )% | 490 | 16 | % | |||||||||||||
| Total other expense, net | $ | (3,057 | ) | (11 | )% | $ | (2,415 | ) | (6 | )% | $ | 642 | 21 | % | ||||||||||
64
Table of Contents
| Year Ended December 31 | Year Ended December 31 | |||||||||||||||||||||||
2008 | 2009 | $ Change | % Change | 2008 | 2009 | |||||||||||||||||||
| ($ in thousands) | (Percentage of total revenue) | |||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 17,202 | $ | 24,752 | $ | 7,550 | 44 | % | 86 | % | 86 | % | ||||||||||||
| Research services | 2,868 | 3,864 | 996 | 35 | % | 14 | % | 14 | % | |||||||||||||||
| Total revenue | 20,070 | 28,616 | 8,546 | 43 | % | 100 | % | 100 | % | |||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||
| Product | 32,160 | 30,462 | (1,698 | ) | (5 | )% | 160 | % | 106 | % | ||||||||||||||
| Research services | 1,169 | 1,788 | 619 | 53 | % | 6 | % | 6 | % | |||||||||||||||
| Impairment charge | 2,524 | — | (2,524 | ) | (100 | )% | 13 | % | 0 | % | ||||||||||||||
| Gross profit (loss) | (15,783 | ) | (3,634 | ) | 12,149 | 77 | % | (79 | )% | (13 | )% | |||||||||||||
| Operating expenses: | ||||||||||||||||||||||||
| Research and development | 2,134 | 2,524 | 390 | 18 | % | 11 | % | 9 | % | |||||||||||||||
| Sales and marketing | 4,034 | 3,994 | (40 | ) | (1 | )% | 20 | % | 14 | % | ||||||||||||||
| General and administrative | 6,180 | 5,430 | (750 | ) | (12 | )% | 31 | % | 19 | % | ||||||||||||||
| Total operating expenses | 12,348 | 11,948 | (400 | ) | (3 | )% | 62 | % | 42 | % | ||||||||||||||
| Income (loss) from operations | (28,131 | ) | (15,582 | ) | 12,549 | 45 | % | (140 | )% | (54 | )% | |||||||||||||
| Other income (expense): | ||||||||||||||||||||||||
| Interest income | 287 | 18 | (269 | ) | (94 | )% | 1 | % | 0 | % | ||||||||||||||
| Interest expense | (7,400 | ) | (3,075 | ) | 4,325 | 58 | % | (37 | )% | (11 | )% | |||||||||||||
| Total other expense, net | (7,113 | ) | (3,057 | ) | 4,056 | 57 | % | (35 | )% | (11 | )% | |||||||||||||
| Net income (loss) | $ | (35,244 | ) | $ | (18,639 | ) | $ | 16,605 | 47 | % | (176 | )% | (65 | )% | ||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||
| Product | $ | 17,202 | 86 | % | $ | 24,752 | 86 | % | $ | 7,550 | 44 | % | ||||||||||||
| Research services | 2,868 | 14 | % | 3,864 | 14 | % | 996 | 35 | % | |||||||||||||||
| Total revenue | $ | 20,070 | 100 | % | $ | 28,616 | 100 | % | $ | 8,546 | 43 | % | ||||||||||||
65
Table of Contents
| Year Ended December 31 | Change | |||||||||||||||
2008 | 2009 | Amount | Percentage | |||||||||||||
| Product shipments in square feet (in thousands) | 6,909 | 10,525 | 3,616 | 52 | % | |||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||||||||||
| Percentage | Percentage | Percentage | Percentage | |||||||||||||||||||||||||||||
| of Related | of Total | of Related | of Total | Change | ||||||||||||||||||||||||||||
Amount | Revenue | Revenue | Amount | Revenue | Revenue | Amount | Percentage | |||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||||||||||
| Product | $ | 32,160 | 187 | % | 160 | % | $ | 30,462 | 123 | % | 106 | % | $ | (1,698 | ) | (5 | )% | |||||||||||||||
| Research services | 1,169 | 41 | % | 6 | % | 1,788 | 46 | % | 6 | % | 619 | 53 | % | |||||||||||||||||||
| Impairment charge | 2,524 | 13 | % | 13 | % | — | 0 | % | 0 | % | (2,524 | ) | (100 | )% | ||||||||||||||||||
| Total cost of revenue | $ | 35,853 | 179 | % | 179 | % | $ | 32,250 | 113 | % | 113 | % | $ | (3,603 | ) | (10 | )% | |||||||||||||||
66
Table of Contents
| Year Ended December 31 | ||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Gross profit (loss) | $ | (15,783 | ) | (79 | )% | $ | (3,634 | ) | (13 | )% | $ | 12,149 | 77 | % | ||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| R&D expenses | $ | 2,134 | 11 | % | $ | 2,524 | 9 | % | $ | 390 | 18 | % | ||||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| Sales and marketing expenses | $ | 4,034 | 20 | % | $ | 3,994 | 14 | % | $ | (40 | ) | (1 | )% | |||||||||||
67
Table of Contents
| Year Ended December 31 | ||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||
| G&A expenses | $ | 6,180 | 31 | % | $ | 5,430 | 19 | % | $ | (750 | ) | (12 | )% | |||||||||||
| Year Ended December 31 | ||||||||||||||||||||||||||||
| 2008 | 2009 | |||||||||||||||||||||||||||
| Percentage | Percentage | Change | ||||||||||||||||||||||||||
Amount | of Revenue | Amount | of Revenue | Amount | Percentage | |||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||||||
| Interest income | $ | 287 | 1 | % | $ | 18 | 0 | % | $ | (269 | ) | (94 | )% | |||||||||||||||
| Interest expense | (7,400 | ) | (37 | )% | (3,075 | ) | (11 | )% | 4,325 | 58 | % | |||||||||||||||||
| Total other expense, net | $ | (7,113 | ) | (35 | )% | $ | (3,057 | ) | (11 | )% | $ | 4,056 | 57 | % | ||||||||||||||
68
Table of Contents
| Three Months Ended | ||||||||||||||||||||||||||||||||||||||||
| 2009 | 2010 | 2011 | ||||||||||||||||||||||||||||||||||||||
March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | |||||||||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||||||||||
| Revenue: | ||||||||||||||||||||||||||||||||||||||||
| Product | $ | 3,886 | $ | 5,949 | $ | 5,967 | $ | 8,950 | $ | 7,681 | $ | 8,327 | $ | 10,187 | $ | 12,495 | $ | 11,274 | $ | 13,059 | ||||||||||||||||||||
| Research services | 879 | 866 | 1,076 | 1,043 | 1,066 | 1,172 | 1,059 | 1,222 | 1,015 | 802 | ||||||||||||||||||||||||||||||
| Total revenue | 4,765 | 6,815 | 7,043 | 9,993 | 8,747 | 9,499 | 11,246 | 13,717 | 12,289 | 13,861 | ||||||||||||||||||||||||||||||
| Cost of revenue: | ||||||||||||||||||||||||||||||||||||||||
| Product | 6,064 | 8,426 | 6,860 | 9,112 | 7,647 | 8,467 | 8,567 | 10,718 | 9,473 | 12,072 | ||||||||||||||||||||||||||||||
| Research services | 346 | 418 | 498 | 526 | 456 | 549 | 508 | 606 | 544 | 378 | ||||||||||||||||||||||||||||||
| Gross profit (loss) | (1,645 | ) | (2,029 | ) | (315 | ) | 355 | 644 | 483 | 2,171 | 2,393 | 2,272 | 1,411 | |||||||||||||||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||||||||||||||
| Research and development | 614 | 601 | 538 | 771 | 917 | 802 | 535 | 731 | 731 | 997 | ||||||||||||||||||||||||||||||
| Sales and marketing | 749 | 1,276 | 892 | 1,077 | 1,194 | 1,124 | 1,007 | 1,201 | 1,224 | 1,333 | ||||||||||||||||||||||||||||||
| General and administrative | 1,410 | 1,213 | 1,371 | 1,436 | 1,504 | 1,309 | 1,360 | 1,502 | 1,587 | 1,856 | ||||||||||||||||||||||||||||||
| Total operating expenses | 2,773 | 3,090 | 2,801 | 3,284 | 3,615 | 3,235 | 2,902 | 3,434 | 3,542 | 4,186 | ||||||||||||||||||||||||||||||
| Income (loss) from operations | (4,418 | ) | (5,119 | ) | (3,116 | ) | (2,929 | ) | (2,971 | ) | (2,752 | ) | (731 | ) | (1,041 | ) | (1,270 | ) | (2,775 | ) | ||||||||||||||||||||
| Other income (expense): | ||||||||||||||||||||||||||||||||||||||||
| Interest income | 6 | — | 14 | (2 | ) | 10 | 27 | 1 | 132 | 48 | 3 | |||||||||||||||||||||||||||||
| Interest expense | (818 | ) | (773 | ) | (764 | ) | (720 | ) | (706 | ) | (698 | ) | (607 | ) | (574 | ) | (499 | ) | (1,770 | ) | ||||||||||||||||||||
| Total other expense, net | (812 | ) | (773 | ) | (750 | ) | (722 | ) | (696 | ) | (671 | ) | (606 | ) | (442 | ) | (451 | ) | (1,767 | ) | ||||||||||||||||||||
| Net income (loss) | $ | (5,230 | ) | $ | (5,892 | ) | $ | (3,866 | ) | $ | (3,651 | ) | $ | (3,667 | ) | $ | (3,423 | ) | $ | (1,337 | ) | $ | (1,483 | ) | $ | (1,721 | ) | $ | (4,542 | ) | ||||||||||
| Three Months Ended | ||||||||||||||||||||||||||||||||||||||||
| 2009 | 2010 | 2011 | ||||||||||||||||||||||||||||||||||||||
| March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | Sept. 30 | Dec. 31 | March 31 | June 30 | |||||||||||||||||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||||||||||||||||||||||
| Net income (loss) | $ | (5,230 | ) | $ | (5,892 | ) | $ | (3,866 | ) | $ | (3,651 | ) | $ | (3,667 | ) | $ | (3,423 | ) | $ | (1,337 | ) | $ | (1,483 | ) | $ | (1,721 | ) | $ | (4,542 | ) | ||||||||||
| Depreciation and amortization | 1,393 | 1,379 | 1,580 | 1,278 | 1,137 | 1,124 | 1,137 | 1,235 | 1,310 | 2,055 | ||||||||||||||||||||||||||||||
| Stock-based compensation | 99 | 91 | 125 | 516 | 96 | 105 | 122 | 145 | 190 | 252 | ||||||||||||||||||||||||||||||
| Interest expense, net | 812 | 773 | 750 | 722 | 696 | 671 | 606 | 442 | 451 | 1,767 | ||||||||||||||||||||||||||||||
| Impairment charge | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Adjusted EBITDA | $ | (2,926 | ) | $ | (3,649 | ) | $ | (1,411 | ) | $ | (1,135 | ) | $ | (1,738 | ) | $ | (1,523 | ) | $ | 528 | $ | 339 | $ | 230 | $ | (468 | ) | |||||||||||||
69
Table of Contents
70
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| Year Ended December 31 | June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Net cash provided by (used in) : | ||||||||||||||||||||
| Operating activities | $ | (21,973 | ) | $ | (12,972 | ) | $ | (15,126 | ) | $ | (9,426 | ) | $ | (7,242 | ) | |||||
| Investing activities | (952 | ) | (1,783 | ) | (15,707 | ) | (12,767 | ) | (16,845 | ) | ||||||||||
| Financing activities | 34,119 | 30,269 | 30,131 | (424 | ) | 26,920 | ||||||||||||||
| Net increase (decrease) in cash | 11,194 | 15,514 | (702 | ) | (22,617 | ) | 2,833 | |||||||||||||
| Cash and cash equivalents, beginning of period | 794 | 11,988 | 27,502 | 27,502 | 26,800 | |||||||||||||||
| Cash and cash equivalents, end of period | $ | 11,988 | $ | 27,502 | $ | 26,800 | $ | 4,885 | $ | 29,633 | ||||||||||
71
Table of Contents
72
Table of Contents
| Less than | More than | |||||||||||||||||||
Contractual Obligations(1) | Total | 1 Year | 1-3 Years | 3-5 Years | 5 Years | |||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Operating leases | $ | 1,939 | $ | 622 | $ | 1,119 | $ | 100 | $ | 98 | ||||||||||
| Capital leases | 195 | 52 | 122 | 21 | — | |||||||||||||||
| 6% Term Loan | 134 | 134 | — | — | — | |||||||||||||||
| Subordinated Notes | 14,556 | — | 14,556 | — | — | |||||||||||||||
| Accrued ARO | 1,033 | — | 1,033 | — | — | |||||||||||||||
| Cross License Agreement | 15,000 | 6,000 | 9,000 | — | — | |||||||||||||||
| Purchase Commitment | 4,708 | 3,328 | 1,380 | — | — | |||||||||||||||
| Total | $ | 37,565 | $ | 10,136 | $ | 27,210 | $ | 121 | �� | $ | 98 | |||||||||
| (1) | The contractual obligations table excludes repayment of our convertible notes issued in June 2011, all of which will be automatically converted into shares of our common stock upon closing of the offering made hereby. |
73
Table of Contents
74
Table of Contents
75
Table of Contents
| Year Ended December 31 | Six Months Ended June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Product cost of revenue | $ | 178 | $ | 148 | $ | 81 | $ | 36 | $ | 76 | ||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 67 | 96 | 57 | 26 | 47 | |||||||||||||||
| Sales and marketing | 136 | 157 | 87 | 40 | 65 | |||||||||||||||
| General and administrative | 546 | 430 | 243 | 99 | 254 | |||||||||||||||
| Total | $ | 927 | $ | 831 | $ | 468 | $ | 201 | $ | 442 | ||||||||||
76
Table of Contents
| Six Months | ||||||||||||||||||||
| Year Ended December 31 | Ended June 30 | |||||||||||||||||||
2008 | 2009 | 2010 | 2010 | 2011 | ||||||||||||||||
| Weighted-average assumptions: | ||||||||||||||||||||
| Expected term (in years) | 6.08 | 5.60 | 6.04 | 5.94 | 6.05 | |||||||||||||||
| Expected volatility | 33.00 | % | 57.00 | % | 49.75 | % | 49.95 | % | 49.27 | % | ||||||||||
| Risk free rate | 3.65 | % | 2.64 | % | 1.90 | % | 2.76 | % | 2.24 | % | ||||||||||
| Expected dividend yield | 0.00 | % | 0.00 | % | 0.00 | % | 0.00 | % | 0.00 | % | ||||||||||
| • | The expected term represents the period that our stock-based awards are expected to be outstanding and is determined using the simplified method described in ASC 718 for all grants. We believe this is a better representation of the estimated life than our actual limited historical exercise behavior. |
| • | For the six months ended June 30, 2010 and 2011, and for the years ended December 31, 2008, 2009, and 2010, the expected volatility is based on the weighted-average volatility of up to six companies within various industries that the we believe are similar to our own. |
| • | The risk-free interest rate is based on U.S. Treasury zero-coupon issues with a remaining term equal to the expected life assumed at the date of grant. | |
| • | We use an expected dividend yield of zero, since we do not intend to pay cash dividends on our common stock in the foreseeable future, nor have we paid dividends on our common stock in the past. |
77
Table of Contents
| • | current business conditions and projections; | |
| • | the probability and value of future liquidity scenarios, including an initial public offering by the company and the sale of the company; | |
| • | the market performance of comparable publicly traded companies; and | |
| • | U.S. and global capital conditions. |
78
Table of Contents
| Estimated | ||||||||||||
| Number | Per Share | per Share | ||||||||||
| of Options | Exercise | Option | ||||||||||
Date of Grant | Granted | Price(1) | Fair Value(2) | |||||||||
| March 27, 2009 | 2,000 | $ | 0.33 | $ | 0.04 | |||||||
| May 27, 2009 | 9,100 | 0.33 | 0.04 | |||||||||
| July 22, 2009 | 6,000 | 0.33 | 0.04 | |||||||||
| November 11, 2009 | 10,276,078 | 0.22 | 0.09 | |||||||||
| December 18, 2009 | 225,000 | 0.22 | 0.09 | |||||||||
| January 20, 2010 | 27,050 | 0.22 | 0.11 | |||||||||
| March 17, 2010 | 49,430 | 0.22 | 0.11 | |||||||||
| July 21, 2010 | 333,443 | 1.30 | 0.63 | |||||||||
| September 15, 2010 | 54,222 | 1.30 | 0.63 | |||||||||
| November 17, 2010 | 1,257,018 | 1.30 | 0.64 | |||||||||
| January 18, 2011 | 530,000 | 1.46 | 0.74 | |||||||||
| January 19, 2011 | 82,512 | 1.46 | 0.74 | |||||||||
| March 16, 2011 | 124,013 | 1.46 | 0.72 | |||||||||
| May 18, 2011 | 1,318,883 | 2.44 | 1.18 | |||||||||
| August 10, 2011 | 243,310 | 2.49 | 1.17 | |||||||||
| Total | 14,538,059 | |||||||||||
| (1) | The fair value of our common stock and the per share exercise price of options are determined by our board of directors. | |
| (2) | The per share fair value of the options was estimated for the date of grant using the Black-Scholes option pricing model. This model estimates the fair value by applying a series of factors including the exercise price of the option, a risk-free interest rate, the expected term of the option, expected share price volatility of the underlying common stock and expected dividends on the underlying common stock. Additional information regarding our common stock option awards is set forth in Note 9 to our audited consolidated financial statements included elsewhere in this prospectus. |
79
Table of Contents
80
Table of Contents
81
Table of Contents
82
Table of Contents
83
Table of Contents
84
Table of Contents
85
Table of Contents
| • | industrial, including use as covering for pipes, valves and storage tanks; | |
| • | building and construction, in walls, floors and ceilings; and | |
| • | original equipment manufacture, including use in transportation, appliances and apparel applications. |
86
Table of Contents
| • | Thermal Performance. Insulation must deliver the required thermal performance within the space allotted. Higher performance insulation is principally required where space is at a premium. | |
| • | Operating Temperature Limitations. Insulation must be able to perform safely and effectively in a system’s operating temperature range. Insulation is commonly targeted and optimized for applications within the cryogenic andsub-ambient (−273oC to 90oC), ambient (0oC to 40oC) or hot process (−25oC to 650oC) temperature regimes. | |
| • | Form Factor. Insulation must be supplied in a form that meets system specifications. Common insulation forms include flexible blankets, boards, loose fill, rigid pipe covers, sprayed and shaped foam structures. | |
| • | Speed of Installation. Insulation that installs rapidly helps reduce total system costs. Labor costs foron-site installation are a significant component of an insulation system’s total cost. | |
| • | Volume and Weight. The volume and weight of the insulation required to meet system specifications impact capital and operating costs of a facility, system, vehicle or building. | |
| • | Durability. Insulation must meet customer specifications for tensile strength, compressive strength and resiliency. Under cryogenic conditions, insulation should not crack, contract or degrade in response to freeze-thaw cycles. In hot process applications, insulation should not crack, crumble or sag. |
87
Table of Contents
| • | Fire Resistance and Protection. Insulation is commonly tested for fire resistance and fire protection under industry-standard protocols. These tests measure whether insulation contributes to the spread of flame and smoke or acts as a passive fire barrier. In industrial markets, minimum fire ratings are largely determined by individual companies. In the building and construction markets, minimum fire ratings are usually determined by national, regional and local building practices and codes. | |
| • | Moisture Resistance. Insulation is commonly tested for water absorption using industry-standard protocols. Moisture reduces the thermal performance of insulation. The resistance of insulation to moisture is critical to the performance of systems exposed to the elements and operating in humid climates. | |
| • | Vapor Permeability. The rate of flow of vapor through insulation layers is critical to the performance of industrial systems. Insulation systems used in cryogenic applications must be impermeable to water vapor to avoid catastrophic system failures. Many cryogenic insulation systems incorporate vapor barriers. Conversely, high vapor permeability enables optimal system performance in hot process applications. | |
| • | Corrosion Under Insulation, or CUI. In the industrial market, systems operating between −4oC and 175oC are subject to CUI. Insulation materials can trap water and exacerbate corrosion of the underlying metal surfaces. Corrosion of process piping and storage tanks increases the risk of catastrophic system failures. Preventative facility maintenance and shutdowns are estimated to cost the petrochemical industry billions of dollars per year. Insulation that is both hydrophobic and vapor permeable has the potential to significantly reduce the incidence of CUI. | |
| • | Logistics. The logistics associated with purchasing, storing, transporting and installing insulation materials in facilities and on job sites can be complex and costly. Improved logistics on job sites reduces operating costs and enhances worker safety. |
88
Table of Contents
| • | Best Thermal Performance. Our aerogel blankets provide the best thermal performance of any widely used insulation product available on the market today. Our products excel in applications where available space is constrained or thermal performance targets are aggressive. | |
| • | Wide Temperature Range. We offer insulation products that address the entire range of applications within the cryogenic and sub-ambient (−273oC to 90oC), ambient (0oC to 40oC) and hot process (−25oC to 650oC) temperature ranges. | |
| • | Ease of Installation. Our flexible aerogel blankets install faster than rigid insulation materials in the industrial market, which reduces labor costs and total system costs. | |
| • | Compact Design. Our aerogel blankets reduce insulation system volume by 50% to 80% compared to traditional insulation. Our products allow for a reduction in the footprint, size and structural costs of facilities, systems, vehicles and buildings. | |
| • | High Durability. Our aerogel blankets offer excellent compression resistance, tensile strength and vibration resiliency. The compression resistance of our products allows companies to pre-insulate, stack and transport steel pipes destined for use in harsh or remote environments. Pre-insulation of pipes significantly reduces installation labor costs in industrial applications in remote areas such as the oil sands in Alberta, Canada. | |
| • | Strong Fire Protection. Our Pyrogel XT and Spaceloft A2 product lines were specifically designed to provide strong fire performance in applications within the industrial and building and construction markets. Strong fire performance qualifies our products for use in a variety of applications in our target markets. | |
| • | Moisture Resistance. Our aerogel blankets are durably hydrophobic. Our products offer improved thermal performance in insulation systems exposed to the elements or operating in humid environments compared to traditional insulation. | |
| • | Reduced Corrosion Under Insulation. Our Pyrogel XT product line is both durably hydrophobic and vapor permeable. These attributes have the potential to reduce the incidence of CUI in hot process applications. We believe that a reduction in CUI offers industrial customers a significant reduction in long-term operating and capital costs. | |
| • | Simplified Logistics. Our aerogel blankets simplify job site logistics. Our products reduce the volume and weight of material purchased, inventoried, transported and installed in the field. In addition, our products reduce the number of stock-keeping units, or SKUs, required to complete a project. Simplified logistics accelerate project timelines, reduce installation costs and improve worker safety. |
89
Table of Contents
| • | Superior product based on proven technology in commercial production. Our aerogel products provide up to five times the thermal performance of widely used insulation in a thin, easy-to-use blanket form. Our products enable compact design, reduce installation time and costs, promote freight savings, simplify logistics, reduce system weight and required storage space and enhance job site safety. Our products provide excellent compression resistance, are hydrophobic and reduce the incidence of corrosion under insulation, a significant operational cost and safety issue in industrial facilities. Our products also offer strong fire protection which is a critical performance requirement in both the industrial and building and construction markets. We believe our array of product attributes provides strong competitive advantages over traditional insulation and will enable us to take a growing share of the existing market for insulation in both the industrial and the building and construction markets. Although competing insulation materials may have one or more comparable attributes, we believe that no single insulation material currently available offers all of the properties of our aerogel insulation. | |
| • | Proven and scalable proprietary manufacturing process. Our manufacturing process is proven and has been replicated to meet increasing demand. Our original line in East Providence, Rhode Island, has operated continuously since mid-2008. From mid-2008 through June 30, 2011, we have produced and sold in excess of 44 million square feet of aerogel blankets. We successfully commenced operation of our second production line at this facility in March 2011 and immediately began producing commercial quality aerogel blankets. This line was completed on time and on budget and is expected to double our annual production capacity by the end of 2011 to 40 to 44 million square feet of aerogel blankets, depending on product mix. We believe that our proven ability to produce product that meets our clients’ specifications and our increased production capacity will provide customers with the certainty of supply that is required to expand their use of our products. | |
| • | Strong relationships with industry leaders. We have a track record of working with industry leading end-use customers, in order to achieve in-depth knowledge of our target market and to optimize our products to meet the challenges they face. With these relationships, our products have undergone rigorous testing and are now in use at some of the world’s largest oil producers, refiners and petrochemical companies as ranked by a weighted measure of sales, profits, assets and market value (see “ — Our Products — Qualification for Use”). We believe these strong relationships represent a competitive advantage, giving us both a significant source of potential demand and a means of validating our technology, products and value proposition. These relationships have proven to shorten the sales cycle with other customers within the industrial market and have helped to facilitate our market penetration. Within the building and construction market, we have partnered with BASF Construction Chemicals to develop products to meet increasingly stringent building standards for thermal performance of retrofit and new-build wall systems. As part of our relationship with BASF Construction Chemicals, we have recently developed a product, Spaceloft A2, targeted at the building and construction market. BASF Venture Capital made strategic investments in us in September 2010 and June 2011. | |
| • | Capital efficient business model. To respond to increased demand for our products, we successfully commenced operation in late March 2011 of a second production line at the East Providence facility. The expansion is expected to increase our annual production capacity by 20 to 22 million square feet of aerogel blankets at a total construction cost of approximately $31.5 million. We believe that our second production line, at full capacity and at current prices, would be capable of producing in the range of $50 million to $54 million in annual revenue of |
90
Table of Contents
| aerogel blankets. We expect to add production capacity in a capital efficient manner through the planned expansion of our East Providence facility and construction of a second manufacturing facility in the United States or Europe. |
| • | Experienced management and operations team. Each of our executive officers has over 20 years of experience in global industrial companies, specialty chemical companies or related materials science research. This team has worked closely together at Aspen Aerogels for nearly five years and we believe our dedicated and experienced workforce is an important competitive asset. As of June 30, 2011, we employed 161 dedicated research scientists, engineers, manufacturing line operators, sales and administrative staff and management. |
| • | Expand our manufacturing capacity to meet market demand. Demand for our aerogel products in 2010 grew by approximately 88% compared to 2009 and exceeded our manufacturing capacity. In response, we constructed a second production line in our East Providence, Rhode Island, manufacturing facility designed to double our manufacturing capacity. To meet anticipated growth in demand for our products, we are engaged in the design, engineering and initial procurement phase of a third production line in our East Providence facility and plan to construct a second manufacturing facility in the United States or Europe. We believe an expanded and geographically diversified manufacturing base will allow us to satisfy increasing demand for our products and to eliminate the risk associated with single site operations. |
| • | Increase industrial insulation market penetration. We plan to focus additional resources to achieve a greater share of the industrial insulation market, both through increased sales to our existing customers and sales to new customers. We are promoting greater enterprise-wide utilization of our products by existing end-use customers. We believe the validation of our products by these technically sophisticated customers helps shorten the sales cycle with new customers and facilitates market penetration. In addition, we anticipate that our growing maintenance-based business will lead to increasing sales of our products into large capital projects, including the construction of new refineries and petrochemical facilities in emerging markets. | |
| • | Leverage strategic relationships in the building and construction market. We believe the building and construction market represents our largest target market opportunity and is in the midst of a transformation that is increasing demand for high-performance, energy-efficient insulation systems. We have partnered with BASF Construction Chemicals to penetrate the market for energy efficient wall systems. We are pursuing additional market opportunities with other leading building materials manufacturers and distributors across multiple regions to address the increasingly stringent regulatory environment governing the thermal performance of buildings. We believe this approach will enable us to leverage their broad technical and distribution capabilities and facilitate market penetration. | |
| • | Expand our sales force, network of distributors and OEM channels. We plan to expand our sales force and distribution network to support growth in the industrial and building and construction markets. We have identified distributors with proven records of success serving important customers and intend to make selected additions to our existing global distribution network. We also intend to expand our network of OEM fabricators to pursue opportunities in the transportation, appliance and apparel markets. We believe that a strong, global and diverse network of distributors and OEMs will support our goal to expand and further penetrate the global markets for our products. |
91
Table of Contents
| • | Continue to develop advanced aerogel compositions, applications and manufacturing technologies. We believe that we are well positioned to leverage a decade’s worth of research and development to commercialize new products, applications and advanced manufacturing technologies. Examples of new technologies in early stages of development include: |
| • | Refractory aerogel insulation compositions for use in applications with temperatures as high as 1649°C including advanced aerospace thermal protection, high temperature furnaces and boilers and single crystal silicon manufacture. | |
| • | Ultra-low dielectric aerogel films that are mechanically robust and can be readily integrated into electronic devices including circuit boards, cables, connectors and discrete components. | |
| • | Aerogel solid sorbents that reversibly capture carbon dioxide for future use in the power industry. | |
| • | Highly durable and flexible aerogel composite insulation for use in tents, outerwear and safety gear. | |
| • | Advanced gel compositions designed to enable aerogels to be processed rapidly and at ultra-high volumes. | |
| • | Aerogel materials suitable for gas, liquid and solid filtration and separation. | |
| • | Electrically conductive aerogels suitable for power storage applications. |
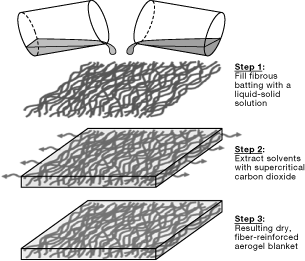
92
Table of Contents
| • | Convection. Heat convection through the gas in nanoporous structures is confined. The mean free path, or average distance traveled, of gas molecules in aerogels is significantly reduced. As a result, thermal convection is severely restricted. | |
| • | Conduction. Heat conduction is correlated to material density. Aerogels are very low density solids. As a result, thermal conductivity is extremely low. | |
| • | Radiation. Radiation requires no medium to transfer heat. Thermal radiation is partially absorbed by aerogels. Our aerogel products also contain infrared absorbing additives to significantly reduce radiant heat transfer. |
| • | Pyrogel XT. Pyrogel XT, our best selling product, is reinforced with a glass-fiber batting and has an upper use temperature of 650oC. Pyrogel XT was initially designed for use in high temperature systems in refineries and petrochemical facilities, and we believe that it has wide applicability throughout the industrial market, including the power generation and district heating sectors. Pyrogel XT’s hydrophobicity and vapor permeability reduce the risk of corrosion under insulation in high temperature operating systems when compared to traditional insulation. | |
| • | Pyrogel XTF. Pyrogel XTF is similar in thermal performance to Pyrogel XT, but is reinforced with a glass- and silica-fiber batting. Pyrogel XTF is specially formulated to provide strong protection against fire. | |
| • | Cryogel Z. Cryogel Z is designed forsub-ambient and cryogenic applications in the industrial market. Cryogel Z is reinforced with a glass- and polyester-fiber batting and is produced with an integral vapor barrier. Cryogel Z is also specially formulated to minimize the incidence of stress corrosion cracking in stainless steel systems. We believe that Cryogel Z’s combination of properties allow for simplified designs and reduced installation costs in cold applications throughout the industrial market when compared to traditional insulation. | |
| • | Spaceloft Subsea. Spaceloft Subsea is reinforced with glass- and polyester-fiber batting and is designed for use inpipe-in-pipe applications in offshore oil production. Spaceloft Subsea can be fabricated and pre-packaged to permit faster installation. Spaceloft Subsea allows for small profile carrier pipelines and associated reductions in capital costs. |
93
Table of Contents
| • | Spaceloft. Spaceloft is reinforced with a glass- and polyester-fiber batting and is designed for use in the building and construction market. Spaceloft is either utilized in roll form by contractors in the field or fabricated by OEMs into strips, panels and systems that meet industry standards. Spaceloft is designed for use in solid wall buildings and where space is at a premium. | |
| • | Spaceloft A2. Spaceloft A2 is reinforced with a glass-fiber batting and specifically designed to meet Euroclass A2 standards, a European classification system based on harmonized standards for fire properties of building and construction products. Spaceloft A2 was developed under a joint development agreement with BASF Construction Chemicals. Under the Euroclass system, the fire characteristics of construction products are described using fire classes A1, A2, B, C, D, E and F. Class A1 comprises materials that do not contribute to a fire. Class A2 contains materials with a very limited contribution to a fire. We believe that achieving the Euroclass A2 classification will permit wider use of Spaceloft A2 in systems subject to European fire performance standards, including hospitals, schools, warehouses, factories, shopping centers and commercial buildings over 15 meters high. |
| • | Pyrogel 2250. Pyrogel 2250 is reinforced with carbon-fiber batting and has an upper use temperature of 200oC. Pyrogel 2250 is our thinnest and our highest tensile strength product. Pyrogel 2250 is designed for use in applications where space is limited. Pyrogel 2250 is targeted to OEMs that design, produce and sell hot appliances, automotive systems and electronic components. | |
| • | Pyrogel 6650. Pyrogel 6650 is reinforced with silica-fiber batting and has an upper use temperature of 650oC. Pyrogel 6650 is our lowest-density product and is designed for use in applications where weight is critical. Pyrogel 6650 is targeted to OEMs that design, produce and sell aerospace systems and components. | |
| • | Cryogel X201. Cryogel X201 is similar in composition to Cryogel Z, but is produced without a vapor barrier. Cryogel X201 is designed for use in cold system designs where space is at a premium. Cryogel X201 is targeted to OEMs that design, produce and sell refrigerated appliances, cold storage equipment and aerospace systems. |
| Nominal | Thermal | Maximum Use | ||||||||||||||||||||||||||||||||||
Product | Thickness | Conductivity | Density | Temperature | Applications | Markets | ||||||||||||||||||||||||||||||
| mm | in | mW/ | Btu-in/ | g/cc | lb/ft3 | °C | °F | |||||||||||||||||||||||||||||
| m-K | hr-ft2-°F | |||||||||||||||||||||||||||||||||||
Pyrogel XT | 5.0 10.0 | 0.20 0.40 | 21.0 | 0.146 | 0.18 | 11.0 | 650° | 1200° | High temperature steam pipes, vessels and equipment, and aerospace and defense systems | Industrial | ||||||||||||||||||||||||||
Pyrogel XTF | 10.0 | 0.40 | 21.0 | 0.146 | 0.18 | 11.0 | 650° | 1200° | High temperature steam pipes, vessels and equipment, aerospace and defense systems, fire barriers and welding blankets | Industrial | ||||||||||||||||||||||||||
Cryogel Z | 5.0 10.0 | 0.20 0.40 | 15.0 | 0.104 | 0.13 | 8.0 | 90° | 194° | Sub-ambient and cryogenic pipelines, vessels and equipment | Industrial | ||||||||||||||||||||||||||
94
Table of Contents
| Nominal | Thermal | Maximum Use | ||||||||||||||||||||||||||||||||||
Product | Thickness | Conductivity | Density | Temperature | Applications | Markets | ||||||||||||||||||||||||||||||
| mm | in | mW/ | Btu-in/ | g/cc | lb/ft3 | °C | °F | |||||||||||||||||||||||||||||
| m-K | hr-ft2-°F | |||||||||||||||||||||||||||||||||||
Spaceloft Subsea | 5.0 | 0.20 | 13.9 | 0.096 | 0.13 | 8.0 | 200° | 390° | Medium to high temperature offshore oil pipelines | Industrial | ||||||||||||||||||||||||||
Spaceloft | 5.0 10.0 | 0.20 0.40 | 14.0 | 0.097 | 0.15 | 9.4 | 200° | 390° | Ambient temperature walls, floors and roofs in commercial, residential and institutional buildings | Building and Construction | ||||||||||||||||||||||||||
Spaceloft A2 | 10.0 | 0.40 | 18.0 | 0.125 | 0.18 | 11.0 | 200° | 390° | Ambient temperature walls, floors and roofs in commercial, residential and institutional buildings; Euroclass A2 Fire Rated | Building and Construction | ||||||||||||||||||||||||||
Pyrogel 2250 | 2.0 | 0.08 | 15.5 | 0.107 | 0.17 | 10.7 | 200° | 390° | Medium to high temperature appliances, transportation and electronics | OEM | ||||||||||||||||||||||||||
Pyrogel 6650 | 6.0 | 0.24 | 14.0 | 0.097 | 0.12 | 7.5 | 650° | 1200° | High temperature aerospace and defense systems | OEM | ||||||||||||||||||||||||||
Cryogel x201 | 5.0 10.0 | 0.20 0.40 | 15.0 | 0.104 | 0.13 | 8.0 | 200° | 390° | Sub-ambient including refrigerated appliances, cold storage and aerospace | OEM | ||||||||||||||||||||||||||
| R-Value | ||||||||||
| per meter of thickness | ||||||||||
Material | Form | From | To | |||||||
| Aerogel | Blankets, Beads | 66 | 100 | |||||||
| Cellulose | Loose Fill | 22 | 26 | |||||||
| Expanded Clay | Loose Fill | 4 | 4 | |||||||
| Expanded Polystyrene (EPS) | Boardstock | 26 | 31 | |||||||
| Extruded Polystyrene (XPS) | Boardstock | 35 | 40 | |||||||
| Fiberglass | Blankets | 22 | 28 | |||||||
| Fiberglass | Loose Fill | 20 | 20 | |||||||
| Fiberglass | Pipe Covering | 25 | 25 | |||||||
| Foamed Glass | Boardstock | 21 | 21 | |||||||
| Mineral Wool | Blankets | 22 | 26 | |||||||
| Mineral Wool | Loose Fill | 17 | 17 | |||||||
| Mineral Wool | Pipe Covering | 19 | 26 | |||||||
| Perlite | Pipe Covering | 17 | 17 | |||||||
| Perlite | Loose Fill | 17 | 21 | |||||||
95
Table of Contents
| R-Value | ||||||||||
| per meter of thickness | ||||||||||
Material | Form | From | To | |||||||
| Perlite | Board | 17 | 19 | |||||||
| Polyurethane | Spray On | 44 | 44 | |||||||
| Polyurethane | Rigid board | 44 | 44 | |||||||
| Polyisocyanurate | Rigid board | 45 | 50 | |||||||
| Vermiculite | Loose Fill | 17 | 26 | |||||||
96
Table of Contents
| • | Distributors: We are currently operating under agreements with a network of over 40 insulation distributors serving industrial and building and construction markets across the globe. The agreements generally provide for exclusivity by geography linked to annual purchase volume minimums. In general, insulation distributors stock, sell and distribute aerogel materials to insulation contractors and end-users. Outside of North America, insulation distributors often will also proactively market and promote aerogel materials across all markets. During 2010, our top distributors by revenue were Thorpe Products, Distribution International, Insulation Fabricators and General Insulation in the United States, Aerogel Korea Co, Ltd. in Asia, Aktarus Group and Sheffield Insulations in Europe, Isolenge Isolantes in South America and Burnaby Insulation in Canada. |
| • | Contractors: We currently sell directly to a number of insulation contractors under multi-year agreements and under project specific contracts. Insulation contractors generally perform insulation installation, inspection and maintenance and project management for end-users. In addition, some insulation contractors provide end-users with project engineering and design services. Several of our agreements with contractors provide for exclusivity by market sector or geography linked to annual purchase volume minimums. During 2010, our top contractor customers by revenue included Technip and Saint-Gobain Wanner. |
| • | OEMs: We currently sell directly to more than ten OEMs that design, fabricate and manufacture insulation components and systems for use in the industrial, building and construction, transportation, appliance and apparel markets. During 2010, our top OEM customers by revenue were Enershield, A. Proctor Group and Shenzhen Naneng Technology Co., Ltd. Enershield manufactures and sells pre-insulated pipes for use in the Canadian oil sands, A. Proctor Group fabricates and sells aerogel insulated wall board in the U.K. market, and Shenzhen Naneng Technology Co., Ltd. supplies insulation systems for use in China’s national high speed railway. |
97
Table of Contents
| • | Oil Refinery and Petrochemical: Our aerogel blankets are in use at ExxonMobil, Hu-Chems Company, SK Energy, Petrobras, Honam Petrochemical, Shell, LyondellBasell, Enagas, Citgo, PetroLogistics and Tupras, among others. We believe these companies are the largest purchasers of our products by revenue during 2010 in the oil refinery and petrochemical sector. Over time, these companies have used our products in an increasing range of applications and throughout an increasing number of their industrial facilities. | |
| • | LNG Production and Storage: Our products are in use in LNG facilities including Gate Import Terminal in the Netherlands, Canaport LNG in Canada and Golden Pass LNG in the United States, among others. These facilities are owned and operated by Qatar Petroleum, ExxonMobil, ConocoPhillips, Repsol, Irving Oil, Gasunie and Vopak. We believe that the size and geographic locations of the these facilities and the owner/operators of these facilities are representative of our end-use customers in the LNG production and storage sector. | |
| • | Oil Production: Our aerogel blankets are in use in several Canadian oil sands facilities owned and operated by Suncor Energy, Devon Energy, Esso, Nexen and Trans Canada Power. We are not aware of any other significant end-users of our aerogel blankets in oil sands facilities. In the offshore oil sector, our products are currently used off the coast of Brazil, in |
98
Table of Contents
| the Gulf of Mexico, in the North Sea and off the west coast of Africa in projects operated by ExxonMobil, Total, BP, Statoil, Dong Energy, Maersk Oil, Anadarko Petroleum, ENI Petroleum, Helix, Marathon Oil and Petrobras. We are not aware of any other significant end-users of our aerogel blankets in offshore oil projects. |
| • | Power Generation: We are targeting operators of gas, coal, nuclear, hydro and solar power generating facilities. We are working with NextEra Energy to optimize our product and approach to the power generation sectors. Our products are currently in use at facilities owned or operated by NextEra Energy, American Electric Power, NRG Energy, Luminant Energy, Nova Scotia Power and Xcel Energy. We are not aware of any other significant end-users of our aerogel blankets in power generation facilities. | |
| • | Other Industrial: Our network of distributors and contractors provides access to additional sectors within the industrial market. Our aerogel blankets are in use in district heating systems owned by Brown University, Massachusetts Institute of Technology, University of Minnesota, The City of Baltimore and the Intercontinental Hotel, Miami. Our products are in use in food processing plants owned by Archer Daniels Midland, Cargill and Pepperidge Farm. We also have products in use at a paper mill owned by Tullis Russell Group. We are not aware of any other significant end-users of our aerogel blankets in this sector. |
| • | Building and Construction: We are targeting leading building products manufacturers with established distribution channels to facilitate penetration of the building and construction market. We have partnered with BASF Construction Chemicals to jointly develop technologically advanced solutions for improved energy efficiency, particularly for the building envelope. In addition, our products have been sold and installed in several hundred residential and commercial new-build and retrofit building projects through the efforts of our network of distributors and OEMs and by means of U.S. federal, state and local sponsored projects. We have products in use today in properties owned or supported by agencies of U.S. federal, state and local governments, private and public schools and universities, hospitals, housing associations, businesses and individuals. |
| • | Other Markets: We rely on the efforts of a small network of OEMs and fabrication houses to target opportunities in the transportation, apparel and appliance markets. Our OEMs are manufacturers of components and systems for refrigerated and hot appliances, cold storage equipment, automobiles, aircraft, trains and electronic sectors and manufacturers of outdoor gear and apparel. While our products have not yet been widely adopted in these markets, we expect that the end-users of our products in these markets will include a wide range of government agencies, institutions, businesses and individuals. We currently have products in use in China’s national high-speed railway, commercial ovens, transport ships, helicopters, motorcycles, hunting and hiking boots, sleeping pads and insulated water bottles. |
| • | Petrobras, a global integrated energy company, needed to replace deteriorating calcium silicate thermal insulation on its REGAP NAPHTHA hydrodesulfurization and treatment unit. Prior to the replacement, the unit was insulated with a combination of calcium silicate for thermal protection and a cementitious surface application for fire protection. However, continued water ingress had deteriorated the calcium silicate over five years of operation. Petrobras chose to replace this insulation with Pyrogel XTF for the project, which offers |
99
Table of Contents
| improved thermal performance and reduced thickness, UL 1709 compliant passive fire protection, hydrophobic properties that prevent water ingress, and rapid installation. As a result of the choice to use Pyrogel XTF, the refurbishment was accomplished with a single insulation product with reduced installation time, which helped reduce disruption at the facility. |
| • | Brown University, a private, Ivy League university in the Northeast, wanted to replace existing fiberglass insulation damaged by moisture that surrounded high-pressure hot water and chilled water lines running throughout its campus. Due to tight spaces and limited access in the underground tunnels where the pipes are housed, the contractor specified Cryogel Z for chilled water lines and Pyrogel XT for hot water lines. The aerogel blankets met thermal performance and corrosion prevention requirements and offered easier installation than bulkier insulation on pipes of all sizes. Additionally, the aerogel blankets reduced insulation volume in comparison to alternative insulation material considered for the project, which improved clearances in the tunnel pipe racks. The contractor has informed us that the aerogel blankets reduced total project costs by 16% in comparison to incumbent materials. | |
| • | NextEra Energy, a leadingU.S.-based power company, has installed Pyrogel XT at four existing fossil fuel-fired power plants, for distinct applications. NextEra Energy determined that Pyrogel XT’s thermal efficiency was superior to competing insulations and its hydrophobicity and vapor permeability would help to prevent corrosion under insulation. Due to Pyrogel XT’s flexible form factor, NextEra Energy achieved accelerated installation times that it determined would result in net cost savings over time versus traditional materials. In May 2011, NextEra Energy placed an order for Pyrogel XT material, for use at a new 250 megawatt solar thermal power plant to be located in Riverside County, California, with product delivery planned for 2012 and 2013. | |
| • | Brock Services Ltd.,a leading insulation contractor in the United States, was hired to replace perlite insulation and associated jacketing on an overhead accumulator drum at aU.S.-based manufacturing facility. Upon removal of the existing insulation, Brock uncovered heavy corrosion of the drum and recommended Pyrogel XT as the preferred alternative due to its hydrophobicity and vapor permeability. Brock informed us that it was impressed by the 75% reduction in material, reduced weight and ease of installation associated with the aerogel blankets. Brock also informed us that Pyrogel XT was more durable and reduced waste and breakage on the job site. Brock estimated that use of the aerogel material reduced total installed costs by 12% in comparison to the traditional material. | |
| • | Framingham Housing Authority, in cooperation with the Massachusetts Department of Housing & Community Development, renovated and re-insulated a110-unit low income housing development in Framingham, Massachusetts. The renovation was a semi-gut rehabilitation that consisted of retrofitting the insulation to the existing building walls and adding insulation to the crawl spaces. Insulating the existing building walls with traditional insulation materials was problematic due to the limited space under a new cement plank siding. Spaceloft’s combination of high thermal performance and compact design allowed our insulation materials to be successfully installed in all 110 housing units. |
100
Table of Contents
| • | Sol Preparation. Mixing of a silica precursor in ethanol, a catalyst and additives in set formulas to deliver the target properties of the resultant aerogel. | |
| • | Casting. Application of the sol into a non-woven batting and initial formation of the gel structure. | |
| • | Aging. Bathing of the gel-saturated blankets in fluids to impart desired physical and thermal properties. | |
| • | Extraction. Supercritcal extraction of the ethanol liquid from the gel-saturated blanket to produce an aerogel blanket. | |
| • | Heat Treatment. Drying to remove trace ethanol, ammonia salts and water from the aerogel blankets. | |
| • | Quality Control. Utilizing statistical process and quality controls to measure thermal conductivity, hydrophobicity and thickness of our aerogel blankets. |
101
Table of Contents
| • | Research: Our research scientists and process engineers explore the chemistry and process technologies of aerogel materials to enhance product performance in response to needs for high temperature thermal stability, high compression strength, low density, low thermal conductivity at low, ambient and high temperatures, high resilience and flexibility and resistance to environmental elements. Our research scientists are investigating aerogel compositions beyond the fiber reinforced silicate aerogel materials which are the backbone of current products. Carbon, ceramic, rubber and hybrid aerogels are all topics of study. A range of form factors are under exploration, including flexible sheets, monoliths, beads and powders. | |
| • | Development: Our development team responds to customer needs for new and broader performance aerogel products. Our efforts to enhance the aerogel product line currently include new grades of flexible aerogel with higher temperature performance, compression resistance, flame and smoke resistance and improved low infrared signature properties. | |
| • | Analytical Services: We have invested in testing and characterization equipment for chemical, mechanical and thermal property measurements. The instrumentation allows in-house measurement of all key properties related to formulation and performance of aerogel products. The analytical laboratory is managed on a service basis with testing prioritized against commercial opportunities. | |
| • | Process Engineering: We maintain a range of pilot equipment that allows rapid examination of formulation and process variables at the research scale. This capability allows the study and continuous improvement of operating processes and provides the ability to produce a range of novel product forms. | |
| • | Program Management: We have a demonstrated record of success in winning and delivering upon government research programs. Research and development performed under contract to NASA, NSF, DARPA, U.S. Army, U.S. Navy, U.S. Air Force and the Department of Energy enables us to develop and leverage technologies into broader commercial applications. | |
| • | Business Development and Technology Transition: We work closely with customers in government and industry to develop potential aerogel solutions that leverage a wide range of attributes of aerogels. We have a long history and demonstrated capability of successfully transitioning technologies from the lab scale into full production. |
102
Table of Contents
| • | Contract Research. We regularly seek funding from a number of federal and non-federal government agencies in support our research and development and manufacturing activities. Research performed under contract to NASA, National Science Foundation, Defense Advanced Research Projects Agency, U.S. Army, U.S. Navy, U.S. Air Force and the Department of Energy allows us to develop and leverage technologies into broader commercial applications. We also work closely with customers in government and industry to develop potential aerogel solutions that leverage not only the thermal insulation performance but other benefits of aerogels as well. |
| • | Title III. Title III of the Defense Production Act was established to create, expand and maintain domestic capacity to produce materials needed for national defense. Title III has accelerated technology adoption by establishing competitive and reliable U.S. sources for key materials. We have received $16.8 million in awards under Title III to build our first production line in East Providence, Rhode Island, to expand effective production capacity, and to reduce manufacturing costs of high temperature aerogel blankets. | |
| • | Department of Energy Loan Guarantee: We have applied to the Department of Energy for a loan guarantee of up to $70.7 million under Section 1703 of Title XVII of the Energy Policy Act of 2005 to fund a portion of our construction costs of our second and third production lines in East Providence, Rhode Island and associated infrastructure. The Department of Energy is currently performing a detailed Phase II due diligence review of our business and financial condition to determine whether the capacity expansion project meets program, statutory and regulatory requirements. Our continued expansion of our East Providence facility is not dependent upon obtaining this guarantee. If awarded, we will assess the opportunity to obtain a loan guaranteed under this program. | |
| • | MassDevelopment: In January 2005, we executed a term loan with the Massachusetts Development Finance Agency for $1.5 million. The proceeds were used to build research and development lab space at our Northborough facility. The Massachusetts Development Finance Agency is a finance and development authority established to strengthen the Massachusetts economy. | |
| • | Rhode Island: Our East Providence facility is located in a Rhode Island Enterprise Zone. We are eligible for tax credits, credit guarantees and job training grants. |
103
Table of Contents
| • | U.S. Patent Nos. 7,078,359 and 7,504,346, as well as related foreign counterparts which are directed to fiber-reinforced aerogel composites and expire on December 21, 2021; | |
| • | U.S. Patent Nos. 7,399,439, 6,989,123 and 7,780,890, as well as related foreign counterparts, all of which are directed to methods of manufacturing fiber-reinforced aerogel composites and expire on June 23, 2024; | |
| • | U.S. Patent No. 7,833,916, which is directed to aerogel-based building materials and expires on July 18, 2025; | |
| • | U.S. Patent No. 7,226,243, which is directed tosub-sea pipeline systems incorporating our Spaceloft® products and expires on May 5, 2024; and | |
| • | U.S. Patent No. 7,691,912, which is directed to organic/inorganic hybrid aerogel composites and expires on October 10, 2027. |
104
Table of Contents
105
Table of Contents
| • | product performance and fitness for purpose; | |
| • | product price, installed cost and lifecycle cost; | |
| • | product availability; and | |
| • | proximity to customer and logistics. |
| • | Foamed Plastics. Armacell, BASF, Bayer, CRH, Dow Chemical, Huntsman, Johns Manville, Kingspan, Knauf Gips, Owens Corning, Recticel and Saint-Gobain. | |
| • | Fiberglass. Johns Manville, Knauf Gips, Owens Corning, Saint-Gobain and Uralita. | |
| • | Mineral Wool. Rockwool, Beijing New Building Materials, CSR, Johns Manville, KCC, Knauf Gips, Nichias, Paroc Group, Saint-Gobain and TechnoNICOL. | |
| • | Aerogels. Aspen Aerogels and Cabot. |
106
Table of Contents
| • | Other. Pittsburgh Corning for foam glass insulation; manufacturers of calcium silicate, perlite and vermiculite tend to be limited to medium- and small-sized, regional suppliers. |
| • | Foamed Plastics. Foamed plastics are the most prevalent form of insulation material in the world and are used broadly in the appliance, transportation, industrial and building and construction markets. Foamed plastic insulation is made from inexpensive raw materials and is a cost-effective solution for many end-users. Foamed plastics provide some of the characteristics of solid insulation and are available in a wide variety of form factors. However, foamed plastics are generally not fire resistant. | |
| • | Fiberglass. Fiberglass is used primarily in building and construction and transportation markets in North America. Fiberglass insulation is made of fine fibers of glass and is typically adhered with a chemical binder to a batt form. Fiberglass will not burn, but will melt under |
107
Table of Contents
| intensive heat. Fiberglass is inexpensive, easy to install, can be cut very easily and is primarily used to fill cavity spaces in building and construction applications. |
| • | Mineral Wool. Mineral wool is commonly used for insulation and fire protection in the building and construction market and in high temperature applications in the industrial market. The two different types of mineral wool are rock wool, which is produced from natural minerals, and slag wool, which is produced from iron ore blast furnace slag. Mineral wool is inexpensive, performs well at high temperatures, and meets fire safety standards in the building and construction market, primarily in Europe. | |
| • | Specialty Materials. Other insulation materials are typically used in specialty applications. Generally, these insulations are either inexpensive, low performance materials or expensive, high-performance materials. Perlite and vermiculite are low performance materials used generally as lightweight, noncombustible insulation. Calcium silicate is a high-performance alternative that can be molded for retrofit industrial applications and can withstand high temperatures. Foamed glass is a high-performance, closed cell, lightweight material that is used in industrial applications where chemical absorption is an issue. Vacuum insulated panels are a relatively new product that provides excellent thermal performance. |
108
Table of Contents
109
Table of Contents
Name | Age | Position | ||||
Executive Officers | ||||||
| Donald R. Young | 54 | President, Chief Executive Officer and Director | ||||
| John F. Fairbanks | 50 | Chief Financial Officer, Vice President and Treasurer | ||||
| George L. Gould, Ph.D. | 48 | Vice President, Research and Development | ||||
| Christopher L. Marlette | 43 | Vice President, Engineering and Design | ||||
| Kevin A. Schmidt | 45 | Vice President, Operations | ||||
| Harry R. Walkoff | 50 | Vice President, Sales and Marketing | ||||
Non-Employee Directors | ||||||
| P. Ramsay Battin | 41 | Director(1)(2) | ||||
| Robert M. Gervis | 51 | Director(3) | ||||
| Craig A. Huff | 47 | Director(1) | ||||
| Steven R. Mitchell | 41 | Director(1)(3) | ||||
| Mark L. Noetzel | 54 | Chairman of the Board(1)(4) | ||||
| William P. Noglows | 53 | Director(2)(4) | ||||
| David J. Prend | 54 | Director(1)(2) | ||||
| Richard F. Reilly | 64 | Director(3)(4) | ||||
| (1) | See “Certain Relationships and Related Person Transactions — Agreements with Stockholders” for a discussion of arrangements among our stockholders pursuant to which this director was selected. | |
| (2) | Member of compensation and leadership development committee. | |
| (3) | Member of nominating and corporate governance committee. | |
| (4) | Member of audit committee. |
110
Table of Contents
111
Table of Contents
112
Table of Contents
113
Table of Contents
| • | the Class I directors will be Messrs. Gervis, Noetzel and Reilly and their terms will expire at the annual meeting of stockholders to be held in 2012; | |
| • | the Class II directors will be Messrs. Battin, Huff and Young and their terms will expire at the annual meeting of stockholders to be held in 2013; and | |
| • | the Class III directors will be Messrs. Mitchell, Noglows and Prend and their terms will expire at the annual meeting of stockholders to be held in 2014. |
114
Table of Contents
| • | approve and retain the independent auditors to conduct the annual audit of our financial statements; | |
| • | review the proposed scope and results of the audit; | |
| • | review and pre-approve audit and non-audit fees and services; | |
| • | review accounting and financial controls with the independent auditors and our financial and accounting staff; | |
| • | review and approve transactions between us and our directors, officers and affiliates; | |
| • | recognize and prevent prohibited non-audit services; | |
| • | establish procedures for complaints received by us regarding accounting matters; and | |
| • | oversee internal audit functions, if any. |
| • | review and recommend the compensation arrangements for management; | |
| • | establish and review general compensation policies with the objective to attract and retain superior talent, to reward individual performance and to achieve our financial goals; | |
| • | administer our stock incentive and purchase plans; | |
| • | ensure appropriate leadership development and succession planning is in place; and | |
| • | oversee the evaluation of management. |
| • | identify and nominate candidates for election to the board of directors; | |
| • | review and recommend the compensation arrangements for certain members of our board of directors; | |
| • | develop and recommend to the board of directors a set of corporate governance principles applicable to our company; and | |
| • | oversee the evaluation of our board of directors. |
115
Table of Contents
| • | our board’s principal responsibility is to oversee the management of Aspen Aerogels; | |
| • | a majority of the members of our board of directors shall be independent directors; | |
| • | directors have full and free access to management and employees of our company, and the right to hire and consult with independent advisors at our expense; | |
| • | all directors are expected to participate in continuing director education on an ongoing basis; and | |
| • | at least annually, our board of directors and its committees will conduct self-evaluations to determine whether they are functioning effectively. |
116
Table of Contents
117
Table of Contents
| • | increased the base salary of each named executive officer by three percent; | |
| • | awarded bonuses above a pre-established target based on revenue performance and awarded discretionary bonuses to our CEO, Mr. Young, and our Vice President Sales and Marketing, Mr. Walkoff, who were instrumental in helping the company achieve its 2010 revenue performance and sales order activity; | |
| • | approved stock option awards to provide additional incentives, satisfy our retention objectives and reward individual performance for 2010; and | |
| • | entered into employment agreements with our executive officers, including our named executive officers, that provide for severance and change of control benefits. |
| • | provide total compensation opportunities which enable us to recruit and retain executives with the experience and skills to manage the development of our products and the profitable growth of our company; | |
| • | create a direct and meaningful link between the compensation we pay and both our business results and each executive’s performance; |
118
Table of Contents
| • | establish a clear alignment between the interests of our executives and the interests of our stockholders; | |
| • | reinforce a culture of ownership, excellence and urgency; and | |
| • | offer total compensation that is competitive and fair. |
119
Table of Contents
120
Table of Contents
121
Table of Contents
122
Table of Contents
| Number of | ||||
Name | Stock Options | |||
| Donald R. Young | 250,000 | |||
| John F. Fairbanks | 100,000 | |||
| Kevin A. Schmidt | 100,000 | |||
| Harry R. Walkoff | 100,000 | |||
| Christopher L. Marlette | 40,000 | |||
123
Table of Contents
124
Table of Contents
| Non-Equity | ||||||||||||||||||||||||
| Option | Incentive Plan | |||||||||||||||||||||||
| Salary | Bonus | Awards | Compensation | Total | ||||||||||||||||||||
Name and Principal Position | Year | ($) | ($)(1) | ($)(2) | ($) | ($) | ||||||||||||||||||
| Donald R. Young | 2010 | $ | 319,202 | $ | 40,000 | $ | 223,265 | $ | 140,503 | $ | 722,970 | |||||||||||||
President and Chief Executive Officer | ||||||||||||||||||||||||
| John F. Fairbanks | 2010 | 236,710 | — | 95,685 | 72,215 | 404,610 | ||||||||||||||||||
Vice President, Finance and Chief Financial Officer | ||||||||||||||||||||||||
| Kevin A. Schmidt | 2010 | 257,027 | — | 95,685 | 78,414 | 431,126 | ||||||||||||||||||
Vice President, Operations | ||||||||||||||||||||||||
| Harry R. Walkoff | 2010 | 230,413 | 20,000 | 95,685 | 70,481 | 416,579 | ||||||||||||||||||
Vice President, Sales and Marketing | ||||||||||||||||||||||||
| Christopher L. Marlette | 2010 | 185,060 | — | 31,895 | 40,728 | 257,683 | ||||||||||||||||||
Vice President, Engineering and Design | ||||||||||||||||||||||||
| (1) | Represents a discretionary bonus amount recommended by our compensation committee and approved by our board of directors in January 2011 for performance in fiscal year 2010. | |
| (2) | These amounts represent the aggregate grant date fair value for option awards granted to our named executive officers, computed in accordance with FASB ASC Topic 718. Valuation assumptions are described in the notes to our financial statements included elsewhere in this prospectus. See our discussion of stock-based compensation under “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Critical Accounting Policies and Estimates — Stock-based Compensation.” |
125
Table of Contents
| All Other | ||||||||||||||||||
| Estimated Future | Option | |||||||||||||||||
| Payouts Under | Awards: | Exercise or | Grant Date | |||||||||||||||
| Non-Equity | Number of | Base Price | Fair Value of | |||||||||||||||
| Incentive Plan | Securities | of Option | Stock and | |||||||||||||||
| Grant | Awards: Target | Underlying | Awards | Option | ||||||||||||||
Name | Date | ($)(1) | Options (#) | ($/Sh) | Awards ($)(2) | |||||||||||||
| Donald R. Young | — | $ | 95,937 | (3) | — | — | — | |||||||||||
| 11/17/2010 | — | 350,000 | $ | 1.30 | $ | 223,265 | ||||||||||||
| John F. Fairbanks | — | 47,429 | (4) | — | — | — | ||||||||||||
| 11/17/2010 | — | 150,000 | 1.30 | 95,685 | ||||||||||||||
| Kevin A. Schmidt | — | 51,500 | (4) | — | — | — | ||||||||||||
| 11/17/2010 | — | 150,000 | 1.30 | 95,685 | ||||||||||||||
| Harry R. Walkoff | — | 46,290 | (4) | — | — | — | ||||||||||||
| 11/17/2010 | — | 150,000 | 1.30 | 95,685 | ||||||||||||||
| Christopher L. Marlette | — | 27,810 | (5) | — | — | — | ||||||||||||
| 11/17/2010 | — | 50,000 | 1.30 | 31,895 | ||||||||||||||
| (1) | Reflects the target non-equity incentive plan award amounts under our 2010 corporate bonus plan. The amount shown above for each named executive officer reflects the target award amount for the full fiscal year which is based on a percentage of that named executive officer’s base salary. The maximum amount of such award cannot be determined because the named executive officer, upon the achievement of certain corporate goals, is entitled to a payout equal to an additional 4% of annual base salary for Mr. Young, an additional 3% of annual base salary for each of Messrs. Fairbanks, Schmidt and Walkoff, and 2% of annual base salary for Mr. Marlette, for every 1% by which the actual revenue exceeded the target amount. The amounts actually paid to our named executive officers under the 2010 corporate bonus plan are shown above in the Summary Compensation Table under the “Non-Equity Incentive Plan Compensation” column. Additionally, see “Compensation Discussion and Analysis — Cash Bonuses” and “— 2010 Corporate Bonus Plan” below for details related to the determination of payments under our 2010 corporate bonus plan. | |
| (2) | These amounts represent the aggregate grant date fair value for option awards granted to our named executive officers, computed in accordance with FASB ASC Topic 718. Valuation assumptions are described in the notes to our financial statements included elsewhere in this prospectus. See our discussion of stock-based compensation under “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Critical Accounting Policies and Estimates — Stock-based Compensation.” | |
| (3) | Represents 30% of Mr. Young’s base salary. See “— 2010 Corporate Bonus Plan” below for details related to the determination of payments under our 2010 corporate bonus plan. | |
| (4) | Represents 20% of the named executive officer’s base salary. See “— 2010 Corporate Bonus Plan” below for details related to the determination of payments under our 2010 corporate bonus plan. | |
| (5) | Represents 15% of Mr. Marlette’s base salary. See “— 2010 Corporate Bonus Plan” below for details related to the determination of payments under our 2010 corporate bonus plan. |
126
Table of Contents
127
Table of Contents
| Option Awards | ||||||||||||||||
| Number of | Number of | |||||||||||||||
| Securities | Securities | |||||||||||||||
| Underlying | Underlying | |||||||||||||||
| Unexercised | Unexercised | |||||||||||||||
| Options | Options | Option | Option | |||||||||||||
| (#) | (#) | Exercise Price | Expiration | |||||||||||||
Name | Exercisable | Unexercisable | ($) | Date | ||||||||||||
| Donald R. Young | 1,854,024 | (1) | 1,705,704 | (2) | $ | 0.22 | 11/11/2019 | |||||||||
| 7,291 | 342,709 | (3) | 1.30 | 11/17/2020 | ||||||||||||
| John F. Fairbanks | 466,576 | 429,250 | (2) | 0.22 | 11/11/2019 | |||||||||||
| 3,124 | 146,876 | (3) | 1.30 | 11/17/2020 | ||||||||||||
| Kevin A. Schmidt | 552,524 | 508,323 | (2) | 0.22 | 11/11/2019 | |||||||||||
| 3,124 | 146,876 | (3) | 1.30 | 11/17/2020 | ||||||||||||
| Harry R. Walkoff | 466,576 | 429,250 | (2) | 0.22 | 11/11/2019 | |||||||||||
| 3,124 | 146,876 | (3) | 1.30 | 11/17/2020 | ||||||||||||
| Christopher L. Marlette | 202,592 | 186,385 | (2) | 0.22 | 11/11/2019 | |||||||||||
| 1,041 | 48,959 | (3) | 1.30 | 11/17/2020 | ||||||||||||
| (1) | Includes options for 58,206 shares of our common stock transferred to Mr. Young’s children in December 2010. | |
| (2) | This option to purchase shares of our common stock vested as to 25% of the shares on November 11, 2009 and thereafter 2.083% of the shares vest in equal monthly installments over 36 months. | |
| (3) | This option to purchase shares of our common stock vests as to 2.083% of the shares in equal monthly installments over the 48 months following the grant date of November 17, 2010. |
128
Table of Contents
129
Table of Contents
130
Table of Contents
| Post- | ||||||||||||||||
| Severance | Bonus | Termination | Total | |||||||||||||
Name | Payments | Payments | Benefits(1) | Benefits | ||||||||||||
| Donald R. Young | $ | 159,895 | $ | 140,503 | $ | 7,113 | $ | 307,511 | ||||||||
| John F. Fairbanks | 59,286 | 72,215 | 4,125 | 135,626 | ||||||||||||
| Kevin A. Schmidt | 64,375 | 78,414 | 3,557 | 146,346 | ||||||||||||
| Harry R. Walkoff | 57,863 | 90,481 | 3,557 | 151,901 | ||||||||||||
| Christopher L. Marlette | 46,350 | 40,728 | 3,557 | 90,635 | ||||||||||||
| (1) | Represents premiums paid by us for continuation of the executive’s medical, dental and vision insurance coverage. |
| Value of | ||||||||||||||||||||
| Additional | Post- | |||||||||||||||||||
| Severance | Bonus | Vested Option | Termination | Total | ||||||||||||||||
Name | Payments | Payments | Awards ($)(1) | Benefits(2) | Benefits | |||||||||||||||
| Donald R. Young | $ | 159,895 | $ | 140,503 | $ | 2,169,906 | $ | 7,113 | $ | 2,477,417 | ||||||||||
| John F. Fairbanks | 59,286 | 72,215 | 555,770 | 4,125 | 691,396 | |||||||||||||||
| Kevin A. Schmidt | 64,375 | 78,414 | 653,821 | 3,557 | 800,167 | |||||||||||||||
| Harry R. Walkoff | 57,863 | 90,481 | 555,770 | 3,557 | 707,671 | |||||||||||||||
| Christopher L. Marlette | 46,350 | 40,728 | 238,951 | 3,557 | 329,586 | |||||||||||||||
| (1) | This represents the amount equal to (a) the number of option shares that would vest, assuming a change of control termination at December 31, 2010, multiplied by (b) the excess of $1.46, which represented our board of directors’ determination of the fair market value of our common stock as of December 31, 2010, over the exercise price of the option. | |
| (2) | Represents premiums paid by us for continuation of the executive’s medical, dental and vision insurance coverage. |
131
Table of Contents
| Fees Earned | Option | All Other | ||||||||||||||
| or Paid | Awards | Compensation | Total | |||||||||||||
Name | in Cash | ($)(1) | ($) | ($) | ||||||||||||
Thomas E. Banahan(2) | $ | — | $ | — | $ | — | $ | — | ||||||||
| P. Ramsay Battin | — | — | — | — | ||||||||||||
| Craig A. Huff | — | — | — | — | ||||||||||||
| Steven R. Mitchell | — | — | — | — | ||||||||||||
| Mark L. Noetzel | — | 63,980 | (3) | 32,625 | (5) | 96,605 | ||||||||||
| David J. Prend | — | — | — | — | ||||||||||||
| Richard F. Reilly | — | 173,313 | (4) | 9,376 | (5) | 182,689 | ||||||||||
| (1) | These amounts represent the aggregate grant date fair value for option awards granted to our directors, computed in accordance with FASB ASC Topic 718. Valuation assumptions are described in the notes to our financial statements included elsewhere in this prospectus. See our discussion of stock-based compensation under “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Critical Accounting Policies and Estimates — Stock-based Compensation.” | |
| (2) | Mr. Banahan resigned as a member of our board of directors on June 17, 2011. | |
| (3) | On November 17, 2010, Mr. Noetzel was granted options to purchase 100,000 shares of our common stock at an exercise price of $1.30. These options vest at the rate of 6.25% per quarter beginning three months after the grant date, provided that upon completion of this offering or a sale of the company, 25% of any unvested options will immediately vest. As of December 31, 2010, Mr. Noetzel held options to purchase 325,000 shares of common stock, 56,250 of which were vested. | |
| (4) | On July 21, 2010 and November 17, 2010, Mr. Reilly was granted options to purchase 225,000 and 50,000, respectively, shares of our common stock, each with an exercise price of $1.30 per share. These options vest at the rate of 6.25% per quarter beginning three months after the applicable grant date, provided that upon completion of this offering or a sale of the company, 25% of any unvested options will immediately vest. As of December 31, 2010, Mr. Reilly held options to purchase 275,000 shares of common stock, 14,062 of which were vested. | |
| (5) | Consists of payments made pursuant to a consulting arrangement with the director that is unrelated to the director’s service on the board. Pursuant to the arrangement, the director was required to attend operating, sales or similar meetings with our management team and received an hourly fee in connection with such service. |
132
Table of Contents
Committee | Chairman | Member | ||||||
| Audit Committee | $ | 14,000 | $ | 5,500 | ||||
| Compensation Committee | 10,000 | 5,000 | ||||||
| Nominating and Governance Committee | 8,000 | 4,000 | ||||||
133
Table of Contents
| • | shares of our common stock; | |
| • | % of the number of shares of our common stock outstanding as of such date; and | |
| • | an amount determined by our board of directors or compensation committee. |
| • | which employees, directors and consultants shall be granted awards; | |
| • | the number of shares of our common stock subject to options and other awards; | |
| • | the exercise price of each option, which generally shall not be less than fair market value on the date of grant; | |
| • | the schedule upon which options become exercisable; | |
| • | the termination or cancellation provisions applicable to options; | |
| • | the terms and conditions of other awards, including conditions for repurchase, termination or cancellation, issue price and repurchase price; and | |
| • | all other terms and conditions upon which each award may be granted in accordance with our plan. |
| • | provide that outstanding options will be assumed or substituted for options of the successor corporation; | |
| • | provide that the outstanding options must be exercised within a certain number of days, either to the extent the options are then exercisable, or at our board of directors’ discretion, any such options being made partially or fully exercisable; | |
| • | terminate outstanding options in exchange for a cash payment of an amount equal to the difference between (a) the consideration payable upon consummation of the corporate transaction to a holder of the number of shares into which such option would have been exercisable to the extent then exercisable (or, in our board of directors’ discretion, any such options being made partially or fully exercisable) and (b) the aggregate exercise price of those options; | |
| • | provide that outstanding stock grants will be substituted for shares of the successor corporation or consideration payable with respect to our outstanding stock in connection with the corporate transaction; and |
134
Table of Contents
| • | terminate outstanding stock grants in exchange for payment of an amount equal to the consideration payable upon consummation of the corporate transaction to a holder of the same number of shares comprising the stock grant, to the extent the stock grant is no longer subject to any forfeiture or repurchase rights (or, at our board of directors’ discretion, all forfeiture and repurchase rights being waived upon the corporate transaction). |
135
Table of Contents
Director | Affiliation with Principal Stockholder | |
| P. Ramsay Battin | Director of Arcapita Inc., an affiliate of Arcapita Ventures I Limited | |
| Craig A. Huff | Senior Managing Member of RCGM, LLC, the ultimate general partner of Reservoir Capital Partners, L.P. and Reservoir Capital Master Fund, L.P. | |
| Steven R. Mitchell | Managing Director of Argonaut Private Equity, LLC, the manager of Argonaut Ventures I, LLC | |
| David J. Prend | Managing Member of the general partners of RockPort Capital Partners, L.P. and affiliated entities |
| Date of Issuance | Original Principal | |||||
Name of Beneficial Owner | of Debt | Amount of Debt | ||||
| Reservoir Capital Partners, L.P. and affiliated funds: | ||||||
| Reservoir Capital Partners, L.P. | January 30, 2008 | $ | 220,450.00 | |||
| February 11, 2008 | $ | 220,450.00 | ||||
| February 26, 2008 | $ | 661,350.00 | ||||
| March 11, 2008 | $ | 661,350.00 | ||||
| March 25, 2008 | $ | 440,900.00 | ||||
| April 22, 2008 | $ | 440,900.00 | ||||
| May 6, 2008 | $ | 440,900.00 | ||||
| May 27, 2008 | $ | 456,001.71 | ||||
| Reservoir Capital Master Fund, L.P. | January 30, 2008 | $ | 29,550.00 | |||
| February 11, 2008 | $ | 29,550.00 | ||||
| February 26, 2008 | $ | 88,650.00 | ||||
| March 11, 2008 | $ | 88,650.00 | ||||
| March 25, 2008 | $ | 59,100.00 | ||||
| April 22, 2008 | $ | 59,100.00 | ||||
| May 6, 2008 | $ | 59,100.00 | ||||
| May 27, 2008 | $ | 61,124.29 | ||||
136
Table of Contents
| Type of | ||||||||||||||||
| Type of | Preferred | Number of | ||||||||||||||
| Preferred | Number of | Shares | Shares | |||||||||||||
| Shares | Shares | Received in | Received in | |||||||||||||
Name of Beneficial Owner | Converted | Converted | Conversion | Conversion | ||||||||||||
| Reservoir Capital Partners, L.P. and affiliated funds | Series D | 838.8141(1 | ) | Series A-1 | 1,863,372(2 | ) | ||||||||||
| RockPort Capital Partners, L.P. and affiliated funds | Series A | 279.9500(3 | ) | Series A-3 | 229,867(3 | ) | ||||||||||
| Series C | 235.8491(4 | ) | Series A-2 | 315,874(5 | ) | |||||||||||
| Series D | 67.7626(6 | ) | Series A-1 | 150,530(7 | ) | |||||||||||
| (1) | Consists of 739.6663 shares held by Reservoir Capital Partners, L.P. and 99.1478 shares held by Reservoir Capital Master Fund, L.P. | |
| (2) | Consists of 1,643,121 shares held by Reservoir Capital Partners, L.P. and 220,251 shares held by Reservoir Capital Master Fund, L.P. | |
| (3) | Consists of shares held by RockPort Capital Partners, L.P. | |
| (4) | Consists of 146.2264 shares held by RockPort Capital Partners, L.P. and 89.6227 shares held by RP Co-Investment Fund I, L.P. | |
| (5) | Consists of 195,842 shares held by RockPort Capital Partners, L.P. and 120,032 shares held by RP Co-Investment Fund I, L.P. | |
| (6) | Consists of 32.0654 shares held by RockPort Capital Partners, L.P. and 35.6972 shares held by RP Co-Investment Fund I, L.P. | |
| (7) | Consists of 71,231 shares held by RockPort Capital Partners, L.P. and 79,299 shares held by RP Co-Investment Fund I, L.P. |
137
Table of Contents
| Type of | ||||||||||||||||
| Original | Preferred | Number of | ||||||||||||||
| Description of | Date of | Principal | Shares | Shares | ||||||||||||
| Debt | Issuance of | Amount | Received in | Received in | ||||||||||||
Name of Beneficial Owner | Converted | Debt | of Debt | Conversion | Conversion | |||||||||||
| Reservoir Capital Partners, L.P. and | Demand loans | (1) | $ | 16,686,082.39(1 | ) | Series B-3 | 8,312,260(2 | ) | ||||||||
| affiliated funds | 14% senior secured notes due 2010 | (3) | $ | 16,480,460.48(3 | ) | Series B-2 | 11,847,136(4 | ) | ||||||||
| Demand loans | (5) | $ | 5,017,126.00(5 | ) | Series B-1 | 2,353,601(6 | ) | |||||||||
| RockPort Capital Partners, L.P. and | Demand loans | (7) | $ | 14,506,944.47(7 | ) | Series B-3 | 7,234,907(8 | ) | ||||||||
| affiliated funds | 14% senior secured notes due 2010 | (9) | $ | 1,331,354.42(9 | ) | Series B-2 | 950,910(10 | ) | ||||||||
| (1) | Consists of demand notes held by: Reservoir Capital Partners, L.P. dated January 19, 2007 in the original principal amount of $10,966,137.45, Reservoir Capital Partners, L.P. dated February 26, 2007 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated March 28, 2007 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated April 25, 2007 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated June 6, 2007 in the original principal amount of $881,800, Reservoir Capital Partners, L.P. dated August 1, 2007 in the original principal amount of $881,800, Reservoir Capital Partners, L.P. dated September 24, 2007 in the original principal amount of $661,350, Reservoir Capital Master Fund, L.P. dated January 19, 2007 in the original principal amount of $1,469,944.94, Reservoir Capital Master Fund, L.P. dated February 26, 2007 in the original principal amount of $59,100, Reservoir Capital Master Fund, L.P. dated March 28, 2007 in the original principal amount of $59,100, Reservoir Capital Master Fund, L.P. dated April 25, 2007 in the original principal amount of $59,100, Reservoir Capital Master Fund, L.P. dated June 6, 2007 in the original principal amount of $118,200, Reservoir Capital Master Fund, L.P. dated August 1, 2007 in the original principal amount of $118,200 and Reservoir Capital Master Fund, L.P. dated September 24, 2007 in the original principal amount of $88,650. | |
| (2) | Consists of 7,329,751 shares held by Reservoir Capital Partners, L.P. and 982,509 shares held by Reservoir Capital Master Fund, L.P. | |
| (3) | Consists of 14% senior secured notes due 2010 held by: Reservoir Capital Partners, L.P. dated February 24, 2005 in the original principal amount of $8,818,000, Reservoir Capital Partners, L.P. dated March 23, 2005 in the original principal amount of $5,714,470.05, Reservoir Capital Master Fund, L.P. dated February 24, 2005 in the original principal amount of $1,182,000 and Reservoir Capital Master Fund, L.P. dated March 23, 2005 in the original principal amount of $765,990.43. | |
| (4) | Consists of 10,446,805 shares held by Reservoir Capital Partners, L.P. and 1,400,331 shares held by Reservoir Capital Master Fund, L.P. | |
| (5) | Consists of demand notes held by: Reservoir Capital Partners, L.P. dated November 30, 2007 in the original principal amount of $661,350, Reservoir Capital Partners, L.P. dated December 28, 2007 in the original principal amount of $220,450, Reservoir Capital Partners, L.P. dated January 30, 2008 in the original principal amount of $220,450, Reservoir Capital Partners, L.P. dated February 11, 2008 in the original principal amount of $220,450, Reservoir Capital Partners, L.P. dated February 26, 2008 in the original principal amount of $661,350, Reservoir Capital Partners, L.P. dated March 11, 2008 in the original principal amount of $661,350, Reservoir Capital Partners, L.P. dated March 25, 2008 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated April 22, 2008 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated May 6, 2008 in the original principal amount of $440,900, Reservoir Capital Partners, L.P. dated May 27, 2008 in the original principal amount of $456,001.71, Reservoir Capital Master Fund, L.P. dated November 30, 2007 in the original principal amount of $88,650, Reservoir Capital Master Fund, L.P. dated December 28, 2007 in the original principal amount of $29,550, Reservoir Capital Master Fund, L.P. dated January 30, 2008 in the original principal amount of $29,550, Reservoir Capital Master Fund, L.P. dated February 11, 2008 in the original principal amount of $29,550, Reservoir Capital Master Fund, L.P. dated February 26, 2008 in the original principal amount of $88,650, Reservoir Capital Master Fund, L.P. dated March 11, 2008 in the original principal amount of $88,650, Reservoir Capital Master Fund, L.P. dated March 25, 2008 in the original principal amount of $59,100, Reservoir Capital Master Fund, L.P. dated April 22, 2008 in the original principal amount of $59,100, Reservoir Capital Master Fund, L.P. dated May 6, 2008 in the original principal amount of $59,100 and Reservoir Capital Master Fund, L.P. dated May 27, 2008 in the original principal amount of $61,124.29. | |
| (6) | Consists of 2,075,405 shares held by Reservoir Capital Partners, L.P. and 278,196 shares held by Reservoir Capital Master Fund, L.P. | |
| (7) | Consists of demand notes held by: RockPort Capital Partners, L.P. dated January 19, 2007 in the original principal amount of $1,023,763.65, RP Co-Investment Fund I, L.P. dated January 19, 2007 in the original principal amount of $713,117.81, RockPort SII, LLC dated January 19, 2007 in the original principal amount of $3,157,624.65, RockPort Capital Partners II, L.P. dated January 19, 2007 in the original principal amount of $6,112,438.36, RockPort Capital Partners II, L.P. dated February 26, 2007 in the original principal amount of $500,000, RockPort Capital Partners II, L.P. dated March 28, 2007 in |
138
Table of Contents
| the original principal amount of $500,000, RockPort Capital Partners II, L.P. dated April 25, 2007 in the original principal amount of $500,000, RockPort Capital Partners II, L.P. dated June 6, 2007 in the original principal amount of $1,000,000 and RockPort Capital Partners II, L.P. dated August 1, 2007 in the original principal amount of $1,000,000. | ||
| (8) | Consists of 512,953 shares held by RockPort Capital Partners, L.P., 357,305 shares held by RP Co-Investment Fund I, L.P., 1,582,116 shares held by Rockport SII, LLC and 4,782,533 shares held by RockPort Capital Partners II, L.P. | |
| (9) | Consists of 14% senior secured notes due 2010 held by RockPort Capital Partners, L.P. dated March 23, 2005 in the original principal amount of $630,000 and RP Co-Investment Fund I, L.P. dated March 23, 2005 in the original principal amount of $701,354.42. | |
| (10) | Consists of 449,973 shares held by RockPort Capital Partners, L.P. and 500,937 shares held by RP Co-Investment Fund I, L.P. |
| Number of | Approximate | |||||||||||
| Shares of Series | Aggregate | |||||||||||
| B-1 Convertible Preferred Stock | Purchase | Date of | ||||||||||
Name of Beneficial Owner | Purchased | Price | Purchase | |||||||||
| Arcapita Ventures I Limited | 4,617,847 | $ | 10,000,000 | June 10, 2008 | ||||||||
| Reservoir Capital Partners, L.P. and affiliated funds | 1,385,355 | (1) | $ | 3,000,000 | June 10, 2008 | |||||||
| RockPort Capital Partners, L.P. and affiliated funds | 2,078,031 | (2) | $ | 4,500,000 | June 10, 2008 | |||||||
| (1) | Consists of 1,221,606 shares purchased by Reservoir Capital Partners, L.P. and 163,749 shares purchased by Reservoir Capital Master Fund, L.P. | |
| (2) | Consists of shares purchased by RockPort Capital Partners II, L.P. |
139
Table of Contents
| Number of | ||||||||||||||||||||||||||||
| Shares of | ||||||||||||||||||||||||||||
| Common | ||||||||||||||||||||||||||||
| Number of Shares of Preferred Stock Converted | Stock | |||||||||||||||||||||||||||
| Series | Series | Series | Series | Series | Series | Received in | ||||||||||||||||||||||
Name of Beneficial Owner | A-3 | A-2 | A-1 | B-3 | B-2 | B-1 | Conversion | |||||||||||||||||||||
| Arcapita Ventures I Limited | — | — | — | — | — | 4,617,847 | 9,235,694 | |||||||||||||||||||||
| Tenaya Capital and affiliated funds | — | 97,031 | (1) | 19,221 | (1) | 957,450 | (1) | 121,423 | (1) | 359,027 | (1) | 1,913,179 | (1) | |||||||||||||||
| Reservoir Capital Partners, L.P. and affiliated funds | — | — | 1,863,372 | (2) | 8,312,260 | (3) | 11,847,136 | (4) | 3,738,956 | (5) | 29,500,680 | (6) | ||||||||||||||||
| RockPort Capital Partners, L.P. and affiliated funds | 229,867 | (7) | 315,874 | (8) | 150,530 | (9) | 7,234,907 | (10) | 950,910 | (11) | 2,078,031 | (12) | 13,039,094 | (13) | ||||||||||||||
| (1) | Consists of shares held by Tenaya Capital IV, LP. | |
| (2) | Consists of 1,643,121 shares held by Reservoir Capital Partners, L.P. and 220,251 shares held by Reservoir Capital Master Fund, L.P. | |
| (3) | Consists of 7,329,751 shares held by Reservoir Capital Partners, L.P. and 982,509 shares held by Reservoir Capital Master Fund, L.P. | |
| (4) | Consists of 10,446,805 shares held by Reservoir Capital Partners, L.P. and 1,400,331 shares held by Reservoir Capital Master Fund, L.P. | |
| (5) | Consists of 3,297,011 shares held by Reservoir Capital Partners, L.P. and 441,945 shares held by Reservoir Capital Master Fund, L.P. | |
| (6) | Consists of 26,013,699 shares held by Reservoir Capital Partners, L.P. and 3,486,981 shares held by Reservoir Capital Master Fund, L.P. | |
| (7) | Consists of shares held by RockPort Capital Partners, L.P. | |
| (8) | Consists of 195,842 shares held by RockPort Capital Partners, L.P. and 120,032 shares held by RP Co-Investment Fund I, L.P. | |
| (9) | Consists of 71,231 shares held by RockPort Capital Partners, L.P. and 79,299 shares held by RP Co-Investment Fund I, L.P. | |
| (10) | Consists of 512,953 shares held by RockPort Capital Partners, L.P., 357,305 shares held by RP Co-Investment Fund I, L.P., 4,782,533 shares held by RockPort Capital Partners II, L.P. and 1,582,116 shares held by RockPort SII, LLC. | |
| (11) | Consists of 449,973 shares held by RockPort Capital Partners, L.P. and 500,937 shares held by RP Co-Investment Fund I, L.P. | |
| (12) | Consists of shares held by RockPort Capital Partners II, L.P. | |
| (13) | Consists of 1,460,810 shares held by RockPort Capital Partners, L.P., 1,057,573 shares held by RP Co-Investment Fund I, L.P., 8,938,595 shares held by RockPort Capital Partners II, L.P. and 1,582,116 shares held by RockPort SII, LLC. |
140
Table of Contents
| Type of | Approximate | |||||||||||||||
| Preferred | Number of | Aggregate | Date of | |||||||||||||
Name of Beneficial Owner | Shares | Shares | Purchase Price | Purchase | ||||||||||||
5% or Greater Stockholders | ||||||||||||||||
Arcapita Ventures I Limited(1) | Series A | 5,140,488 | $ | 3,000,000 | 8/14/09 | |||||||||||
| Series A | 5,063,381 | $ | 2,955,000 | 9/14/09 | ||||||||||||
| Series B | 1,569,961 | $ | 2,098,727 | 9/22/10 | ||||||||||||
| Argonaut Ventures I, LLC | Series A | 12,851,220 | $ | 7,500,000 | 8/14/09 | |||||||||||
| Series A | 2,499,517 | $ | 1,458,724 | 9/14/09 | ||||||||||||
| Series B | 1,831,721 | $ | 2,448,649 | 9/22/10 | ||||||||||||
| Tenaya Capital IV, L.P. and affiliated funds | Series A | 1,720,198 | (2) | $ | 1,003,911 | 9/14/09 | ||||||||||
| Series B | 625,531 | (3) | $ | 836,211 | 9/22/10 | |||||||||||
| Reservoir Capital Partners, L.P. and affiliated funds | Series A | 5,997,236 | (4) | $ | 3,500,000 | 8/14/09 | ||||||||||
| Series A | 7,710,733 | (5) | $ | 4,500,000 | 9/14/09 | |||||||||||
| Series B | 2,809,085 | (6) | $ | 3,755,191 | 9/22/10 | |||||||||||
| RockPort Capital Partners, L.P. and affiliated funds | Series A | 3,426,992 | (7) | $ | 2,000,000 | 8/14/09 | ||||||||||
| Series A | 3,769,692 | (8) | $ | 2,200,000 | 9/14/09 | |||||||||||
| Series B | 1,377,332 | (9) | $ | 1,841,221 | 9/22/10 | |||||||||||
Officers and Directors | ||||||||||||||||
| P. Ramsay Battin | Series A | 25,702 | $ | 15,000 | 9/14/09 | |||||||||||
| Series B | 3,067 | $ | 4,100 | 10/20/10 | ||||||||||||
| John F. Fairbanks | Series A | 12,919 | $ | 7,540 | 9/10/09 | |||||||||||
| Series B | 1,541 | $ | 2,061 | 10/20/10 | ||||||||||||
| Donald R. Young | Series A | 12,919 | $ | 7,540 | 9/10/09 | |||||||||||
| Series B | 1,541 | $ | 2,061 | 10/20/10 | ||||||||||||
| (1) | Does not include shares of preferred stock purchased by P. Ramsay Battin, a director of Arcapita Inc., which is an affiliate of Arcapita Ventures I Limited, and referenced under the “Officers and Directors” section of this table. | |
| (2) | Consists of 676,380 shares of Series A preferred stock purchased by Tenaya Capital IV-P, LP, 649,052 shares of Series A preferred stock purchased by Tenaya Capital IV-C, LP and 394,766 shares of Series A preferred stock purchased by Tenaya Capital IV, LP. | |
| (3) | Consists of 211,090 shares of Series B preferred stock purchased by Tenaya Capital IV-P, LP, 202,561 shares of Series B preferred stock purchased by Tenaya Capital IV-C, LP and 211,880 shares of Series B preferred stock purchased by Tenaya Capital IV, LP. | |
| (4) | Consists of 5,222,427 shares of Series A preferred stock purchased by Reservoir Capital Partners, L.P. and 774,809 shares of Series A preferred stock purchased by Reservoir Capital Master Fund, L.P. | |
| (5) | Consists of 6,799,324 shares of Series A preferred stock purchased by Reservoir Capital Partners, L.P. and 911,409 shares of Series A preferred stock purchased by Reservoir Capital Master Fund, L.P. | |
| (6) | Consists of shares of Series B preferred stock purchased by Reservoir Capital Partners, L.P. | |
| (7) | Consists of shares of Series A preferred stock purchased by RockPort Capital Partners II, L.P. |
141
Table of Contents
| (8) | Consists of 2,398,894 shares of Series A preferred stock purchased by RockPort Capital Partners, L.P., 965,348 shares of Series A preferred stock purchased by RockPort S II, LLC and 405,449 shares of Series A preferred stock purchased by RockPort Capital Partners II, L.P. | |
| (9) | Consists of 344,313 shares of Series B preferred stock purchased by RockPort Capital Partners, L.P., 812,836 shares of Series B preferred stock purchased by RockPort Capital Partners II, L.P., 178,118 shares of Series B preferred stock purchased by RockPort S II, LLC and 42,065 shares of Series B preferred stock purchased by RP Co-Investment Fund I, L.P. |
142
Table of Contents
143
Table of Contents
| • | each existing stockholder we know to beneficially own 5% or more of our common stock, which we call our principal stockholders; | |
| • | each of our directors; | |
| • | each of our named executive officers; and | |
| • | all of our current directors and executive officers as a group. |
144
Table of Contents
| After this Offering | After this Offering | |||||||||||||||||||||||
| Assuming the Underwriters’ | Assuming the Underwriters’ | |||||||||||||||||||||||
| Prior to this Offering | Option is not Exercised | Option is Exercised in Full | ||||||||||||||||||||||
| Shares of | Percentage | Shares of | Percentage | Shares of | Percentage | |||||||||||||||||||
| Common | of Common | Common | of Common | Common | of Common | |||||||||||||||||||
| Stock | Stock | Stock | Stock | Stock | Stock | |||||||||||||||||||
Name of Beneficial Owner | ||||||||||||||||||||||||
Principal Stockholders: | ||||||||||||||||||||||||
Arcapita Ventures I Limited(1) | 14,852,396 | 15.6 | % | |||||||||||||||||||||
Argonaut Ventures I, LLC(2) | 17,182,459 | 18.1 | % | |||||||||||||||||||||
Reservoir Capital Partners, L.P. and affiliated funds(3) | 26,354,045 | 27.7 | % | |||||||||||||||||||||
RockPort Capital Partners, L.P. and affiliated funds(4) | 12,937,266 | 13.6 | % | |||||||||||||||||||||
Tenaya Capital and affiliated funds(5) | 5,869,742 | 6.2 | % | |||||||||||||||||||||
Entities affiliated with Fidelity Investments(6) | % | |||||||||||||||||||||||
Directors and Executive Officers: | ||||||||||||||||||||||||
Donald R. Young(7) | 2,543,019 | 2.6 | % | |||||||||||||||||||||
John F. Fairbanks(8) | 664,713 | * | ||||||||||||||||||||||
Christopher L. Marlette(9) | 279,294 | * | ||||||||||||||||||||||
Kevin A. Schmidt(9) | 763,705 | * | ||||||||||||||||||||||
Harry R. Walkoff(9) | 650,253 | * | ||||||||||||||||||||||
P. Ramsay Battin(10) | 28,769 | * | ||||||||||||||||||||||
Robert M. Gervis(9) | 33,125 | * | ||||||||||||||||||||||
Craig A. Huff(11) | 26,354,045 | 27.7 | % | |||||||||||||||||||||
Steven R. Mitchell(12) | 17,182,459 | 18.1 | % | |||||||||||||||||||||
Mark L. Noetzel(9) | 103,125 | * | ||||||||||||||||||||||
William P. Noglows(9) | 33,125 | * | ||||||||||||||||||||||
David J. Prend(13) | 12,937,266 | 13.6 | % | |||||||||||||||||||||
Richard F. Reilly(9) | 65,625 | * | ||||||||||||||||||||||
All executive officers and directors as a group (14 persons)(14) | 61,917,817 | 61.7 | % | |||||||||||||||||||||
| * | Indicates beneficial ownership of less than 1%. | |
| (1) | Consists of shares held by Arcapita Ventures I Limited (“AVIL”). AVIL is managed and advised by Arcapita Investment Management Limited and Arcapita Inc., two wholly-owned subsidiaries of Arcapita Bank B.S.C.(c), which exercises the voting and investment power over the shares held of record by AVIL. The address for AVIL isc/o Arcapita Inc., 75 Fourteenth Street, 24th Floor, Atlanta, Georgia 30309. | |
| (2) | Consists of shares of common stock held by Argonaut Ventures I, LLC (“Argonaut”), a limited liability company, which is managed by Ken Levit and Argonaut Private Equity, LLC (“APE”). Steven R. Mitchell, one of our directors, Jason Martin, Don Millican, Fred Dorwart and Ken Kinnear are managers, and the George B. Kaiser Family Foundation (“GKFF”), an Oklahomanot-for-profit corporation, is the majority owner, of APE. Messrs. Mitchell, Martin, Millican, Dorwart and Kinnear and GKFF may be deemed to share and voting and investment control over the shares, which are beneficially owned by Argonaut. Each of these individuals and GKFF disclaims beneficial ownership of the reported securities except to the extent of his or its pecuniary interest therein. The address of Argonaut Ventures I, LLC is 6733 South Yale, Tulsa, OK 74136. | |
| (3) | Consists of 23,502,071 shares of common stock and 3,025 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by Reservoir Capital Partners, L.P. (“RCP”) and 2,848,544 shares of common stock and 405 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by Reservoir Capital Master Fund, L.P. (“RCMF”). The securities held by RCP may be deemed to be beneficially owned by Daniel Stern and one of our directors, Craig A. Huff, who are the senior managing members (the “Reservoir Members”) of RCGM, LLC (“RCGM”). RCGM is the managing member of Reservoir Capital Group, L.L.C. (“RCG”), which is in turn the general partner of Reservoir Capital Partners (Cayman), L.P. (“RCP Cayman”), which is in turn the sole member of RCP GP, LLC (“RCP GP”), and which is in turn the general partner of RCP. The securities held by RCMF may be deemed to be beneficially owned by the Reservoir Members, who are the senior managing members of RCGM. RCGM is the managing member of RCG, which is in turn the general partner of RCMF. As a result, Messrs. Stern and Huff share voting and |
145
Table of Contents
| investment control over the shares held by RCP and RCMF. Each of the Reservoir Members, RCGM, RCG, RCP Cayman and RCP GP disclaims beneficial ownership of the reported securities except to the extent of his or its pecuniary interest therein. The address for RCP and RCMF isc/o Reservoir Capital Group, L.L.C., 650 Madison Avenue, 24th Floor, New York, New York 10022. | ||
| (4) | Consists of 3,231,129 shares of common stock and 13,972 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by RockPort Capital Partners, L.P. (“RockPort I”); 7,624,809 shares of common stock and 1,368 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by RockPort Capital Partners II, L.P. (“RockPort II”); 1,670,838 shares of common stock and 446 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by RockPort SII, LLC (“RSII”); and 394,589 shares of common stock and 115 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by RP Co-Investment Fund I, LP (“RPCIF”). The securities held by RockPort I, RockPort II, RSII and RPCIF may be deemed to be beneficially owned by RockPort Capital I, LLC, RockPort Capital II, LLC, RockPort SGII, LLC and RP Co-Investment Fund I GP, LLC, respectively, each of which is the general partner of the respective entity. The securities held by RockPort I, RockPort II, RSII and RPCIF may also be deemed to be beneficially owned by Alexander Ellis III, Janet B. James, William E. James, Charles J. McDermott, Stoddard M. Wilson and one of our directors, David J. Prend, who are the managing members (the “Rockport Members”) of the general partners of each of RockPort I, RockPort II, RSII and RPCIF, all of which may be deemed to share voting and investment control with respect to such shares. Each of the general partners of RockPort I, RockPort II, RSII and RPCIF and the Rockport Members (the “Rockport Reporting Persons”) disclaim beneficial ownership of the reported securities except to the extent of his, her or its pecuniary interest therein. The address for the Rockport Reporting Persons isc/o RockPort Capital Partners, 160 Federal Street, 18th Floor, Boston, Massachusetts 02110. | |
| (5) | Consists of 1,987,541 shares of common stock and 661 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by Tenaya Capital IV, LP (“TC IV”); 1,980,126 shares of common stock and 658 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by Tenaya Capital IV-P, LP (“TC IV-P”); 1,900,123 shares of common stock and 633 shares issuable upon the exercise of warrants exercisable within 60 days of June 30, 2011 held by Tenaya Capital IV-C, LP (“TC IV-C”). The general partner of TC IV is Tenaya Capital IV Annex GP, LLC (“TC IV Annex”). The general partner of both TC IV-P and TC IV-C is Tenaya Capital IV GP, LP, whose general partner is Tenaya Capital IV GP, LLC (“TC IV LLC”). The managing members of TC IV LLC and TC IV Annex are Thomas E. Banahan, Benjamin Boyer, Stewart Gollmer, Brian Melton and Brian Paul (collectively, the “TC Managing Members”), all of which share voting and dispositive power over these shares. The TC Managing Members disclaim beneficial of such shares except to the extent of his pecuniary interest, if any. The address for TC IV, TC IV-P, TC IV-C and the TC Managing Members isc/o Tenaya Capital, 2965 Woodside Road, Suite A, Woodside, California 94062. | |
| (6) | Consists of shares of our common stock to be issued upon the automatic conversion of $29.0 million principal amount and all accrued but unpaid interest on the convertible notes upon the closing of the offering made hereby at a conversion price equal to 87.5% of the initial offering price, assuming an initial public offering price of $ per share, the mid-point of the price range set forth on the cover page of this prospectus, and that the closing of the offering made hereby occurs on , 2011, of which shares of our common stock will be issued to Fidelity Select Portfolios: Environment and Alternative Energy Portfolio; shares of our common stock will be issued to Fidelity Puritan Trust: Fidelity Puritan Fund; shares of our common stock will be issued to Variable Insurance Products Fund III: Balanced Portfolio; shares of our common stock will be issued to Fidelity Securities Fund: Fidelity Dividend Growth Fund; shares of our common stock will be issued to Fidelity Advisor Series I: Fidelity Advisor Dividend Growth Fund; shares of our common stock will be issued to Fidelity Central Investment Portfolios LLC: Fidelity Materials Central Fund; shares of our common stock will be issued to Variable Insurance Products Fund IV: Materials Portfolio; shares of our common stock will be issued to Fidelity Select Portfolios: Materials Portfolio; shares of our common stock will be issued to Fidelity Select Portfolios: Industrials Portfolio; shares of our common stock will be issued to Fidelity Advisor Series VII: Fidelity Advisor Industrials Fund; shares of our common stock will be issued to Variable Insurance Products Fund IV: Industrials Portfolio; and shares of our common stock will be issued to Fidelity Central Investment Portfolios LLC: Fidelity Industrials Central Fund. The address for each of these entities isc/o Fidelity Investments, 82 Devonshire Street, Boston, MA 02109. |
| (7) | Consists of 14,460 shares of common stock and 2,528,559 shares issuable upon the exercise of options exercisable within 60 days of June 30, 2011. These options include 116,412 shares issuable upon the exercise of options held by Mr. Young’s children, of which Mr. Young has sole voting power. | |
| (8) | Consists of 14,460 shares of common stock and 650,253 shares issuable upon the exercise of options exercisable within 60 days of June 30, 2011. | |
| (9) | Consists of shares issuable upon the exercise of options exercisable within 60 days of June 30, 2011. | |
| (10) | Reflects securities beneficially owned individually by Mr. Battin. Mr. Battin is a director of Arcapita Inc., an affiliate of AVIL, but is not deemed to beneficially own any of the shares held by AVIL. |
146
Table of Contents
| (11) | Reflects securities beneficially owned by RCP and RCMF as set forth in footnote 3, for which Mr. Huff is a senior managing member of the ultimate general partner of both entities. Mr. Huff disclaims beneficial ownership of such shares except to the extent of his pecuniary interest therein, if any. | |
| (12) | Reflects securities beneficially owned by Argonaut as set forth in footnote 2. Mr. Mitchell is a manager of APE, a manager of Argonaut. Mr. Mitchell disclaims beneficial ownership of such shares except to the extent of his pecuniary interest therein, if any. | |
| (13) | Reflects securities beneficially owned by Rockport Capital Partners, L.P. and affiliated funds as set forth in footnote 4, for which Mr. Prend is the managing general partner. Mr. Prend disclaims beneficial ownership of such shares except to the extent of his pecuniary interest therein, if any. |
| (14) | See footnotes 7 through 13. Also includes options to purchase 279,294 shares of common stock held by George L. Gould, Ph.D., which are exercisable within 60 days following June 30, 2011. |
147
Table of Contents
148
Table of Contents
| Number of | Weighted-Average | |||||||
Expiration Date | Shares | Exercise Price | ||||||
February 24, 2013(1)(3)(4)(5) | 224 | $ | 0.003 | |||||
March 23, 2013(1)(3)(4)(5) | 237 | $ | 0.003 | |||||
October 20, 2014(1)(2)(3)(4)(5) | 56 | $ | 4.525 | |||||
October 28, 2014(1)(2)(3)(4)(5) | 28 | $ | 4.525 | |||||
January 12, 2015(1)(2)(3)(4)(5) | 25 | $ | 4.525 | |||||
| June 10, 2016 | 133,339 | $ | 0.003 | |||||
December 29, 2017(3)(5) | 993,415 | $ | 0.001 | |||||
Total: | 1,127,324 | $ | 0.002 | |||||
| (1) | Each of these warrants contains anti-dilution provisions providing for adjustments to the exercise price upon the issuance of shares of our common stock at a price less than the exercise price, excluding shares of our common stock issuable upon exercise of options, warrants, conversion of convertible securities and certain issuances approved in advance by a majority of our board of directors. | |
| (2) | Each of these warrants expires on the earlier of this date or a corporate event, which is defined in the warrant to include (i) the sale, transfer, exchange or other disposition of all or substantially all of our assets; (ii) a merger or reorganization that results in our stockholder immediately prior to such transaction holding less than 50% of the voting power of the surviving entity of such transaction; (iii) a dissolution or liquidation; and (iv) the sale, transfer, exchange or other disposition of all or substantially all of our common stock. | |
| (3) | The shares underlying each of these warrants are entitled to certain registration rights set forth in our registration rights agreement. See “— Registration Rights” below for a description of these registration rights. | |
| (4) | Each of these warrants provides that immediately before its expiration or termination, if the fair market value of one share of our common stock is greater than the exercise price, the warrant will be automatically exercised pursuant to its net exercise provision. | |
| (5) | Each of these warrants has net exercise provisions under which the holder may, in lieu of payment of the exercise price in cash, surrender the warrant and receive a net amount of shares of our common based on the fair market value of the underlying shares of our common stock at the time of exercise of the warrant, after deduction of the aggregate exercise price. |
149
Table of Contents
150
Table of Contents
151
Table of Contents
152
Table of Contents
153
Table of Contents
154
Table of Contents
| Shares Eligible | ||||||
Date Available for Sale | for Sale | Comment | ||||
| Date of prospectus | Shares sold in the offering and shares that can be sold under Rule 144 that are not subject to a lock-up | |||||
| 180 days* after date of prospectus | Lock-up released; shares can be sold under Rule 144 | |||||
| * | 180 days corresponds to thelock-up period described below in“— Lock-up Agreements.” Thislock-up period may be extended or shortened under certain circumstances as described in that section. However, Goldman, Sachs & Co. and Morgan Stanley & Co. LLC, may in their sole discretion, at any time without prior notice, release all or any portion of the shares from the restrictions in any of these agreements. |
155
Table of Contents
| • | one percent of the number of the total number of shares of our common stock then outstanding, which will equal approximately shares immediately after this offering; and | |
| • | the average weekly trading volume in our common stock on the stock exchange where our common stock is traded during the four calendar weeks preceding either (i) to the extent that the seller is required to file a notice on Form 144 with respect to such sale, the date of filing such notice, (ii) date of receipt of the order to execute the transaction by the broker or (iii) the date of execution of the transaction with the market maker. |
156
Table of Contents
| • | if, during the last 17 days of the180-day restricted period, we release earnings results or announce material news or a material event, then the restrictions described in the preceding paragraph will continue to apply until the expiration of the18-day period beginning on the date of release of the earnings results or the announcement of the material news or material event, as applicable; or | |
| • | if, prior to the expiration of the180-day restricted period, we announce that we will release earnings results during the15-day period following the last day of the180-day restricted period, then the restrictions described in the preceding paragraph will continue to apply until the expiration of the18-day period beginning on the date of release of the earnings results or the announcement of the material news or material event, as applicable. |
157
Table of Contents
158
Table of Contents
| • | an individual who is a citizen or resident of the United States; | |
| • | a corporation, or other entity treated as a corporation for U.S. federal income tax purposes, created or organized in or under the laws of the United States, any state thereof or the District of Columbia; | |
| • | an estate the income of which is subject to U.S. federal income taxation regardless of its source; or | |
| • | a trust, if a U.S. court is able to exercise primary supervision over the administration of the trust and one or more U.S. persons have authority to control all substantial decisions of the trust or if the trust has a valid election to be treated as a U.S. person under applicable U.S. Treasury Regulations. |
| • | insurance companies; | |
| • | tax-exempt organizations; | |
| • | financial institutions; | |
| • | brokers or dealers in securities; | |
| • | regulated investment companies; | |
| • | pension plans; | |
| • | controlled foreign corporations; | |
| • | passive foreign investment companies; |
159
Table of Contents
| • | owners that hold our common stock as part of a straddle, hedge, conversion transaction, synthetic security or other integrated investment; and | |
| • | certain U.S. expatriates. |
160
Table of Contents
| • | the gain is effectively connected with thenon-U.S. holder’s conduct of a trade or business in the United States, and, if an applicable income tax treaty so provides, the gain is attributable to a permanent establishment maintained by thenon-U.S. holder in the United States; in these cases, thenon-U.S. holder will be taxed on a net income basis at the regular graduated rates and in the manner applicable to United States persons (as defined in the Code), and, if thenon-U.S. holder is a foreign corporation, an additional branch profits tax at a rate of 30%, or a lower rate as may be specified by an applicable income tax treaty, may also apply; | |
| • | thenon-U.S. holder is an individual present in the United States for 183 days or more in the taxable year of the disposition and certain other conditions are met, in which case thenon-U.S. holder will be subject to a 30% tax (or such lower rate as may be specified by an applicable income tax treaty) on the net gain derived from the disposition (which may be offset by United States source capital losses, even though the individual is not considered a resident of the United States); or | |
| • | we are or have been, at any time during the five-year period preceding such disposition (or thenon-U.S. holder’s holding period, if shorter) a “U.S. real property holding corporation” for U.S. federal income tax purposes, unless our common stock is regularly traded on an established securities market and thenon-U.S. holder held no more than five percent of our outstanding common stock, directly or indirectly, during the shorter of the five-year period ending on the date of the disposition or the period that thenon-U.S. holder held our common stock. Generally, a corporation is a “U.S. real property holding corporation” if the fair market value of its “U.S. real property interests” equals or exceeds 50% of the sum of the fair market value of its worldwide real property interests plus its other assets used or held for use in a trade or business. Although valuations are inherently uncertain, and no assurance can be given that our valuations of our “U.S. real property interests” and our other assets used or held for use in a trade or business will not change, we nevertheless believe that we are not currently, and we do not anticipate becoming, a “U.S. real property holding corporation” for U.S. federal income tax purposes. Furthermore, even if we were to become a “U.S. real property holding corporation,” so long as our common stock continues to be regularly traded on an established securities market, only a non-U.S. holder who holds or held at any time during the five year period preceding the date of disposition (or such non-U.S. holder’s holding period, if shorter) more than 5% of our common stock will be subject to U.S. federal income tax on the disposition of our common stock. |
161
Table of Contents
162
Table of Contents
| Number of | ||||
Underwriters | Shares | |||
| Goldman, Sachs & Co. | ||||
| Morgan Stanley & Co. LLC | ||||
| Total | ||||
No Exercise | Full Exercise | |||||||
| Per Share | $ | $ | ||||||
| Total | $ | $ | ||||||
163
Table of Contents
164
Table of Contents
165
Table of Contents
166
Table of Contents
167
Table of Contents
| F-2 | ||||
| Financial Statements: | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 | ||||
Index To Consolidated Financial Statements (unaudited) | ||||
| F-33 | ||||
| F-34 | ||||
| F-35 | ||||
| F-36 |
F-1
Table of Contents
June 24, 2011
F-2
Table of Contents
| December 31 | ||||||||
2009 | 2010 | |||||||
| (In thousands, except | ||||||||
| share and per share data) | ||||||||
Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 27,502 | $ | 26,800 | ||||
| Marketable securities | — | 4,020 | ||||||
| Accounts receivable, net of allowance for doubtful accounts | 4,234 | 10,205 | ||||||
| Costs in excess of billings | 100 | 124 | ||||||
| Inventories | 1,601 | 2,253 | ||||||
| Prepaid expenses and other current assets | 354 | 419 | ||||||
| Total current assets | 33,791 | 43,821 | ||||||
| Restricted cash | 556 | 857 | ||||||
| Property, plant, and equipment, net | 29,763 | 42,622 | ||||||
| Other assets | 625 | 1,495 | ||||||
| Total assets | $ | 64,735 | $ | 88,795 | ||||
Liabilities, Redeemable Convertible Preferred Stock and Stockholders’ (Deficit) Equity | ||||||||
| Current liabilities: | ||||||||
| Long-term debt, current portion | $ | 262 | $ | 247 | ||||
| Capital leases, current portion | 15 | 20 | ||||||
| Accounts payable | 2,927 | 5,597 | ||||||
| Accrued expenses | 791 | 5,987 | ||||||
| Deferred revenue | 630 | 447 | ||||||
| Other current liabilities | 7,400 | 6,800 | ||||||
| Total current liabilities | 12,025 | 19,098 | ||||||
| Long-term debt, excluding current portion | 247 | 7,820 | ||||||
| Capital leases, excluding current portion | 51 | 52 | ||||||
| Other long-term liabilities | 14,578 | 10,142 | ||||||
| Total liabilities | 26,901 | 37,112 | ||||||
Commitments and contingencies (Note 14) | ||||||||
| Series B redeemable convertible preferred stock, $0.001 par value; authorized 17,000,000 shares; issued and outstanding 0 and 16,010,292 shares at December 31, 2009 and 2010, at redemption and liquidation value | — | 28,799 | ||||||
| Series A redeemable convertible preferred stock, $0.001 par value; authorized, issued and outstanding 52,843,201 shares at December 31, 2009 and 2010, at redemption and liquidation value | 31,681 | 80,987 | ||||||
| Stockholders’ (deficit) equity: | ||||||||
| Common stock, $0.001 par value; authorized 114,000,000 shares; issued and outstanding 25,567,499 and 25,574,570 shares at December 31, 2009 and 2010 | 26 | 26 | ||||||
| Additional paid-in capital | 192,392 | 138,038 | ||||||
| Accumulated deficit | (186,265 | ) | (196,175 | ) | ||||
| Accumulated other comprehensive income | — | 8 | ||||||
| Total stockholders’ (deficit) equity | 6,153 | (58,103 | ) | |||||
| Total liabilities, redeemable convertible preferred stock and stockholders’ (deficit) equity | $ | 64,735 | $ | 88,795 | ||||
F-3
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| (In thousands, except share and per share data) | ||||||||||||
| Revenue: | ||||||||||||
| Product | $ | 17,202 | $ | 24,752 | $ | 38,690 | ||||||
| Research services | 2,868 | 3,864 | 4,519 | |||||||||
| Total revenue | 20,070 | 28,616 | 43,209 | |||||||||
| Cost of revenue: | ||||||||||||
| Product | 32,160 | 30,462 | 35,399 | |||||||||
| Research services | 1,169 | 1,788 | 2,119 | |||||||||
| Impairment charge | 2,524 | — | — | |||||||||
| Gross profit (loss) | (15,783 | ) | (3,634 | ) | 5,691 | |||||||
| Operating expenses: | ||||||||||||
| Research and development | 2,134 | 2,524 | 2,985 | |||||||||
| Sales and marketing | 4,034 | 3,994 | 4,526 | |||||||||
| General and administrative | 6,180 | 5,430 | 5,675 | |||||||||
| Total operating expenses | 12,348 | 11,948 | 13,186 | |||||||||
| Income (loss) from operations | (28,131 | ) | (15,582 | ) | (7,495 | ) | ||||||
| Other income (expense): | ||||||||||||
| Interest income | 287 | 18 | 170 | |||||||||
| Interest expense | (7,400 | ) | (3,075 | ) | (2,585 | ) | ||||||
| Total other expense, net | (7,113 | ) | (3,057 | ) | (2,415 | ) | ||||||
| Net income (loss) | (35,244 | ) | (18,639 | ) | (9,910 | ) | ||||||
| Dividends and accretion of redeemable convertible preferred stock | (2,351 | ) | (2,984 | ) | (57,007 | ) | ||||||
| Net income (loss) attributable to common stockholders | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | |||
| Net income (loss) per common share attributable to common stockholders, basic and diluted | $ | (3,389.12 | ) | $ | (2.21 | ) | $ | (2.62 | ) | |||
| Weighted-average common shares outstanding, basic and diluted | 11,093 | 9,751,616 | 25,574,286 | |||||||||
F-4
Table of Contents
Consolidated Statements of Stockholders’ (Deficit) Equity and Comprehensive Income (Loss)
| Accumulated | ||||||||||||||||||||||||
| Common Stock | Additional | Other | Total | |||||||||||||||||||||
| $0.001 Par Value | Paid-in | Accumulated | Comprehensive | Stockholders’ | ||||||||||||||||||||
Shares | Value | Capital | Deficit | Income | (Deficit)/Equity | |||||||||||||||||||
| (In thousands, except share and per share data) | ||||||||||||||||||||||||
| Balance at December 31, 2007 | 9,408 | $ | — | $ | 9,327 | $ | (132,382 | ) | $ | — | $ | (123,055 | ) | |||||||||||
| Net income (loss) and total comprehensive income (loss) | — | — | — | (35,244 | ) | — | (35,244 | ) | ||||||||||||||||
| Issuance of common stock warrants | — | — | 73 | — | — | 73 | ||||||||||||||||||
| Stock compensation expense | — | — | 927 | — | — | 927 | ||||||||||||||||||
| Dividends on redeemable convertible preferred stock | — | — | (2,512 | ) | — | — | (2,512 | ) | ||||||||||||||||
| Exercise of common stock warrants | 14,287 | — | — | — | — | — | ||||||||||||||||||
| Exchange of redeemable convertible preferred stock | — | — | 55,968 | — | — | 55,968 | ||||||||||||||||||
| Accretion of redeemable convertible preferred stock to redemption value | — | — | (55,807 | ) | — | — | (55,807 | ) | ||||||||||||||||
| Balance at December 31, 2008 | 23,695 | — | 7,976 | (167,626 | ) | — | (159,650 | ) | ||||||||||||||||
| Net income (loss) and total comprehensive income (loss) | — | — | — | (18,639 | ) | — | (18,639 | ) | ||||||||||||||||
| Issuance of common stock | 2,272 | — | — | — | — | — | ||||||||||||||||||
| Stock compensation expense | — | — | 831 | — | — | 831 | ||||||||||||||||||
| Dividends on redeemable convertible preferred stock | — | — | (2,682 | ) | — | — | (2,682 | ) | ||||||||||||||||
| Conversion of preferred stock and related dividends to | 25,541,532 | 26 | 186,084 | — | — | 186,110 | ||||||||||||||||||
| common stock | ||||||||||||||||||||||||
| Cancellation of warrant liability | — | — | 485 | — | — | 485 | ||||||||||||||||||
| Accretion of redeemable convertible preferred stock to redemption value | — | — | (302 | ) | — | — | (302 | ) | ||||||||||||||||
| Balance at December 31, 2009 | 25,567,499 | 26 | 192,392 | (186,265 | ) | — | 6,153 | |||||||||||||||||
| Comprehensive income (loss): | ||||||||||||||||||||||||
| Net income (loss) | — | — | — | (9,910 | ) | — | (9,910 | ) | ||||||||||||||||
Unrealized gain onavailable-for-sale securities | — | — | — | — | 8 | 8 | ||||||||||||||||||
| Total comprehensive income (loss) | (9,902 | ) | ||||||||||||||||||||||
| Issuance of common stock | 7,071 | — | 2 | — | — | 2 | ||||||||||||||||||
| Issuance of common stock warrant | — | — | 2,183 | — | — | 2,183 | ||||||||||||||||||
| Stock compensation expense | — | — | 468 | — | — | 468 | ||||||||||||||||||
| Dividends on redeemable convertible preferred stock | — | — | (2,928 | ) | — | — | (2,928 | ) | ||||||||||||||||
| Accretion of redeemable convertible preferred stock to redemption value | — | — | (54,079 | ) | — | — | (54,079 | ) | ||||||||||||||||
| Balance at December 31, 2010 | 25,574,570 | $ | 26 | $ | 138,038 | $ | (196,175 | ) | $ | 8 | $ | (58,103 | ) | |||||||||||
F-5
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| (In thousands) | ||||||||||||
| Cash flows from operating activities: | ||||||||||||
| Net income (loss) | $ | (35,244 | ) | $ | (18,639 | ) | $ | (9,910 | ) | |||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 7,059 | 5,630 | 4,633 | |||||||||
| Asset impairment charge | 2,524 | — | — | |||||||||
| Imputed interest | 3,507 | 3,000 | 2,425 | |||||||||
Paid-in-kind interest | 3,446 | — | — | |||||||||
| Loss on sale of marketable securities | — | — | 53 | |||||||||
| Stock compensation expense | 927 | 831 | 468 | |||||||||
| Change in fair value of preferred stock warrant liability | 165 | — | — | |||||||||
| Settlement of asset retirement obligation | — | — | (40 | ) | ||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Accounts receivable | (1,069 | ) | 232 | (5,971 | ) | |||||||
| Costs in excess of billings | 209 | (20 | ) | (24 | ) | |||||||
| Inventories | (1,812 | ) | 1,591 | (652 | ) | |||||||
| Prepaid expenses and other assets | (54 | ) | 47 | (348 | ) | |||||||
| Accounts payable | 1,931 | (1,295 | ) | 181 | ||||||||
| Accrued expenses | (41 | ) | 142 | 1,642 | ||||||||
| Deferred revenue | 479 | (91 | ) | (183 | ) | |||||||
| Other long-term liabilities | (4,000 | ) | (4,400 | ) | (7,400 | ) | ||||||
| Net cash used in operating activities | (21,973 | ) | (12,972 | ) | (15,126 | ) | ||||||
| Cash flows from investing activities: | ||||||||||||
| Capital expenditures | (1,121 | ) | (1,644 | ) | (11,341 | ) | ||||||
| Proceeds from sale of equipment | 31 | — | — | |||||||||
| Decrease (increase) in restricted cash | 128 | (139 | ) | (301 | ) | |||||||
| Purchase of marketable securities | — | — | (15,037 | ) | ||||||||
| Proceeds from maturities and sales of marketable securities | 10 | — | 10,972 | |||||||||
| Net cash used in investing activities | (952 | ) | (1,783 | ) | (15,707 | ) | ||||||
| Cash flows from financing activities: | ||||||||||||
| Proceeds from issuance of long-term debt | 8,000 | — | 10,000 | |||||||||
| Repayment of borrowings under long-term debt | (483 | ) | (249 | ) | (265 | ) | ||||||
| Deferred financing costs | — | — | (687 | ) | ||||||||
| Repayment of obligations under capital lease | (3 | ) | (20 | ) | (17 | ) | ||||||
| Proceeds from issuance of preferred stock | 26,605 | 30,538 | 21,098 | |||||||||
| Proceeds from issuance of common stock | — | — | 2 | |||||||||
| Net cash provided by financing activities | 34,119 | 30,269 | 30,131 | |||||||||
| Net increase (decrease) in cash | 11,194 | 15,514 | (702 | ) | ||||||||
| Cash at beginning of period | 794 | 11,988 | 27,502 | |||||||||
| Cash at end of period | $ | 11,988 | $ | 27,502 | $ | 26,800 | ||||||
| Supplemental disclosures of cash flow information: | ||||||||||||
| Interest paid | $ | 56 | $ | 41 | $ | 33 | ||||||
| Income taxes paid | $ | — | $ | — | $ | — | ||||||
| Supplemental disclosures of non-cash activities: | ||||||||||||
| Accrued dividends on preferred stock | $ | 2,512 | $ | 2,682 | $ | 2,928 | ||||||
| Accretion of preferred stock | $ | 55,807 | $ | 302 | $ | 54,079 | ||||||
| Issuance of common stock warrants | $ | — | $ | — | $ | 2,183 | ||||||
| Issuance of preferred stock warrants | $ | 321 | $ | — | $ | — | ||||||
| Exchange of preferred stock and related dividends | $ | 55,968 | $ | 186,110 | $ | — | ||||||
| Conversion of debt | $ | 92,771 | $ | — | $ | — | ||||||
| Cancellation of preferred stock warrant liability | $ | — | $ | 485 | $ | — | ||||||
| Accrued capital expenditures | $ | — | $ | — | $ | 6,044 | ||||||
| Capital lease | $ | — | $ | 87 | $ | 23 | ||||||
F-6
Table of Contents
| (1) | Description of Business |
F-7
Table of Contents
| (2) | Summary of Basis of Presentation and Significant Accounting Policies |
F-8
Table of Contents
F-9
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Costs incurred | $ | 3,193 | $ | 1,672 | $ | 2,005 | ||||||
| Expenditures offset by cost sharing funding received: | ||||||||||||
| Cost of revenue: | ||||||||||||
| Product | 43 | 20 | 33 | |||||||||
| Research services | 1,227 | 637 | 1,124 | |||||||||
| Operating expenses: | ||||||||||||
| Research and development | 1,214 | 620 | 475 | |||||||||
| Sales and marketing | 342 | 193 | 170 | |||||||||
| General and administrative | 367 | 202 | 203 | |||||||||
| Total expenditures offset by cost sharing | $ | 3,193 | $ | 1,672 | $ | 2,005 | ||||||
F-10
Table of Contents
| Year Ended | ||||||||||||
| December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Product cost of revenue | $ | 178 | $ | 148 | $ | 81 | ||||||
| Research and development expenses | 67 | 96 | 57 | |||||||||
| Sales and marketing expenses | 136 | 157 | 87 | |||||||||
| General and administrative expenses | 546 | 430 | 243 | |||||||||
| Total stock-based compensation | $ | 927 | $ | 831 | $ | 468 | ||||||
F-11
Table of Contents
F-12
Table of Contents
F-13
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Revenue: | ||||||||||||
| U.S. | 10,427 | 12,668 | 20,056 | |||||||||
| International | 9,643 | 15,948 | 23,153 | |||||||||
| $ | 20,070 | $ | 28,616 | $ | 43,209 | |||||||
F-14
Table of Contents
| (3) | Related Party Transactions |
| (4) | Property, Plant, and Equipment |
| December 31 | ||||||||||
2009 | 2010 | Useful life | ||||||||
| Construction in progress | $ | 191 | $ | 13,215 | — | |||||
| Buildings | 12,884 | 12,884 | 30 years | |||||||
| Machinery and equipment | 31,237 | 35,606 | 5 — 7 years | |||||||
| Computer equipment and software | 1,416 | 1,431 | 3 years | |||||||
| Total | 45,728 | 63,136 | ||||||||
| Accumulated depreciation and amortization | (15,965 | ) | (20,514 | ) | ||||||
| Property, plant and equipment, net | $ | 29,763 | $ | 42,622 | ||||||
F-15
Table of Contents
| December 31 | ||||||||
2009 | 2010 | |||||||
| Office equipment, at cost | $ | 62 | $ | 94 | ||||
| Vehicles, at cost | 84 | 73 | ||||||
| Total capital leases | 146 | 167 | ||||||
| Accumulated amortization | (70 | ) | (88 | ) | ||||
| Capital leases, net | $ | 76 | $ | 79 | ||||
| (5) | Inventories |
| December 31 | ||||||||
2009 | 2010 | |||||||
| Raw material | $ | 1,288 | $ | 1,657 | ||||
| Finished goods | 313 | 596 | ||||||
| Total | $ | 1,601 | $ | 2,253 | ||||
| (6) | Recapitalization and Reorganization |
F-16
Table of Contents
| (7) | Redeemable Convertible Preferred Stock |
| Series A (2001) | Series C | Series D | ||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | |||||||||||||||||||
| Balance at December 31, 2007 | 293.95 | $ | 4,032 | 1,116.1831 | $ | 23,648 | 1,024.7362 | $ | 35,183 | |||||||||||||||
| Dividends | — | — | — | — | — | 850 | ||||||||||||||||||
| Exchange of redeemable convertible preferred stock and related dividends | (293.95 | ) | (4,032 | ) | (1,116.1831 | ) | (23,648 | ) | (1,024.7362 | ) | (36,033 | ) | ||||||||||||
| Balance at December 31, 2008, 2009 and 2010 | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||
F-17
Table of Contents
| Series A-1 | Series A-2 | Series A-3 | ||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | |||||||||||||||||||
| Balance at December 31, 2007 | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||
| Exchange of redeemable convertible preferred stock and related dividends at fair value | 758,795 | 5,349 | 498,303 | 2,048 | 80,454 | 348 | ||||||||||||||||||
| Accretion to redemption value | — | 12,410 | — | 4,456 | — | 759 | ||||||||||||||||||
| Balance at December 31, 2008 | 758,795 | $ | 17,759 | 498,303 | $ | 6,504 | 80,454 | $ | 1,107 | |||||||||||||||
| Conversion of redeemable convertible preferred stock and related dividends | (758,795 | ) | (17,759 | ) | (498,303 | ) | (6,504 | ) | (80,454 | ) | (1,107 | ) | ||||||||||||
| Balance at December 31, 2009 and 2010 | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||
F-18
Table of Contents
| Series B-1 | Series B-2 | Series B-3 | ||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | |||||||||||||||||||
| Balance at December 31, 2007 | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||
| Conversion of debt into redeemable convertible preferred stock | 1,563,681 | 10,159 | 4,786,982 | 30,822 | 7,971,886 | 51,790 | ||||||||||||||||||
Issuance ofSeries B-1 | 4,158,875 | 26,284 | — | — | — | — | ||||||||||||||||||
| Accretion to redemption value | — | 37,906 | — | 276 | — | — | ||||||||||||||||||
| Dividends | — | 1,662 | — | — | — | — | ||||||||||||||||||
| Balance at December 31, 2008 | 5,722,556 | $ | 76,011 | 4,786,982 | $ | 31,098 | 7,971,886 | $ | 51,790 | |||||||||||||||
| Dividends | — | 1,841 | — | — | — | — | ||||||||||||||||||
| Conversion of redeemable convertible preferred stock and related dividends | (5,722,556 | ) | (77,852 | ) | (4,786,982 | ) | (31,098 | ) | (7,971,886 | ) | (51,790 | ) | ||||||||||||
| Balance at December 31, 2009 and 2010 | — | $ | — | — | $ | — | — | $ | — | |||||||||||||||
F-19
Table of Contents
| Series B (2010) | Series A (2009) | |||||||||||||||
Shares | Amount | Shares | Amount | |||||||||||||
| Balance at December 31, 2008 | — | $ | — | — | $ | — | ||||||||||
| Issuance of Series A (2009) | 52,843,201 | 30,538 | ||||||||||||||
| Accretion to redemption value | — | — | — | 302 | ||||||||||||
| Dividends | — | — | — | 841 | ||||||||||||
| Balance at December 31, 2009 | — | — | 52,843,201 | 31,681 | ||||||||||||
| Issuance of Series B (2010) | 16,010,292 | 21,098 | — | — | ||||||||||||
| Accretion to redemption value | — | 7,240 | — | 46,839 | ||||||||||||
| Dividends | — | 461 | — | 2,467 | ||||||||||||
| Balance at December 31, 2010 | 16,010,292 | $ | 28,799 | 52,843,201 | $ | 80,987 | ||||||||||
F-20
Table of Contents
F-21
Table of Contents
| (8) | Stockholders’ (Deficit) Equity |
| (9) | Employee Stock Options |
F-22
Table of Contents
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Weighted average assumptions: | ||||||||||||
| Expected term (in years) | 6.08 | 5.60 | 6.04 | |||||||||
| Expected volatility | 33.00 | % | 57.00 | % | 49.75 | % | ||||||
| Risk free rate | 3.65 | % | 2.64 | % | 1.90 | % | ||||||
| Expected dividend yield | 0.00 | % | 0.00 | % | 0.00 | % | ||||||
| Weighted average grant-date fair value of options granted | $ | 0.14 | $ | 0.09 | $ | 0.61 | ||||||
| Weighted average grant-date fair value of options vested | $ | 0.43 | $ | 0.12 | $ | 0.12 | ||||||
| Aggregate intrinsic value of options exercised | — | — | — | |||||||||
F-23
Table of Contents
| Weighted | ||||||||||||||||||||
| Weighted | Weighted | Average | ||||||||||||||||||
| Average | Average | Remaining | Aggregate | |||||||||||||||||
| Number of | Grant Date | Exercise | Contractual | Intrinsic | ||||||||||||||||
Shares | Fair Value | Price | Term | Value | ||||||||||||||||
| Options outstanding at December 31, 2009 | 10,513,805 | $ | 0.12 | $ | 0.26 | 9.86 | ||||||||||||||
| Granted | 1,721,163 | $ | 0.61 | $ | 1.25 | |||||||||||||||
| Forfeited | (79,172 | ) | $ | 0.24 | $ | 0.48 | ||||||||||||||
| Exercised | (7,071 | ) | $ | 0.12 | $ | 0.22 | — | |||||||||||||
| Options outstanding at December 31, 2010 | 12,148,725 | $ | 0.19 | $ | 0.40 | 8.97 | $ | 13,288,975 | ||||||||||||
| Exercisable at December 31, 2010 | 5,473,821 | $ | 0.12 | $ | 0.30 | 8.81 | $ | 6,730,867 | ||||||||||||
| Expected to vest at December 31, 2010 | 6,474,669 | $ | 0.24 | $ | 0.48 | 9.10 | $ | 6,361,370 | ||||||||||||
| Nonvested options at December 31, 2009 | 7,683,662 | $ | 0.12 | |||||||||||||||||
| Granted | 1,721,163 | |||||||||||||||||||
| Vested | (2,662,583 | ) | $ | 3,264,487 | ||||||||||||||||
| Forfeited | (67,338 | ) | ||||||||||||||||||
| Nonvested options at December 31, 2010 | 6,674,904 | $ | 0.24 | |||||||||||||||||
F-24
Table of Contents
| (10) | Net Income (Loss) Per Share |
| Year Ended December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Net income (loss) | $ | (35,244 | ) | $ | (18,639 | ) | $ | (9,910 | ) | |||
| Deemed dividends on redeemable convertible preferred stock (inclusive of issuance costs and accretion to redemption value): | ||||||||||||
| Series D | (850 | ) | — | — | ||||||||
| Exchange of Series A (2001), C and D | 55,968 | — | — | |||||||||
Series A-1,A-2 andA-3 | (17,625 | ) | — | — | ||||||||
Series B-1 and B-2 | (39,844 | ) | (1,841 | ) | — | |||||||
| Series A (2009) | — | (1,143 | ) | (49,306 | ) | |||||||
| Series B (2010) | — | — | (7,701 | ) | ||||||||
| Total deemed dividends | (2,351 | ) | (2,984 | ) | (57,007 | ) | ||||||
| Net income (loss) attributable to common stockholders | $ | (37,595 | ) | $ | (21,623 | ) | $ | (66,917 | ) | |||
| Weighted average shares outstanding | 11,093 | 9,751,616 | 25,574,286 | |||||||||
| Net income (loss) per share attributable to common stockholders, basic and diluted | $ | (3,389.12 | ) | $ | (2.21 | ) | $ | (2.62 | ) | |||
F-25
Table of Contents
| December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| Common stock options | 3,444,077 | 10,513,805 | 12,148,725 | |||||||||
| Common stock warrants | 137,213 | 134,941 | 1,631,048 | |||||||||
| Preferred stock warrant | 384,721 | — | — | |||||||||
| Series A-1 | 758,795 | — | — | |||||||||
| Series A-2 | 498,303 | — | — | |||||||||
| Series A-3 | 80,455 | — | — | |||||||||
| Series B-1 | 11,445,112 | — | — | |||||||||
| Series B-2 | 4,786,982 | — | — | |||||||||
| Series B-3 | 7,971,886 | — | — | |||||||||
| Series A (2009) | — | 52,843,201 | 52,843,201 | |||||||||
| Series B (2010) | — | — | 16,010,292 | |||||||||
| Total | 29,507,544 | 63,491,947 | 82,633,266 | |||||||||
| (11) | Long-Term Debt |
| December 31 | ||||||||
2009 | 2010 | |||||||
| 6% Term Loan | $ | 509 | $ | 247 | ||||
| Subordinated Notes, net of discount | — | 7,820 | ||||||
| Total long-term debt | 509 | 8,067 | ||||||
| Current maturities of long-term debt | (262 | ) | (247 | ) | ||||
| Long-term debt, less current maturities | $ | 247 | $ | 7,820 | ||||
F-26
Table of Contents
| (12) | Other Long-term Liabilities |
| December 31 | ||||||||
2009 | 2010 | |||||||
| ARO | $ | 964 | $ | 953 | ||||
| Cross License Agreement | 21,014 | 15,989 | ||||||
| 21,978 | 16,942 | |||||||
| Current maturities of other long-term liabilities | (7,400 | ) | (6,800 | ) | ||||
| Other long-term liabilities, less current maturities | $ | 14,578 | $ | 10,142 | ||||
F-27
Table of Contents
| Year Ended December 31 | ||||||||
2009 | 2010 | |||||||
| Balance at beginning of period | $ | 1,085 | $ | 964 | ||||
| Revisions in the estimated cash flow | (145 | ) | — | |||||
| Accretion of discount expense | 24 | 29 | ||||||
| Settlement costs | — | (40 | ) | |||||
| Balance at end of period | $ | 964 | $ | 953 | ||||
Year | Payment | |||
| 2011 | $ | 6,800 | ||
| 2012 | 6,000 | |||
| 2013 | 6,000 | |||
| Total payments | $ | 18,800 | ||
| (13) | Income Taxes |
F-28
Table of Contents
| December 31 | ||||||||||||
2008 | 2009 | 2010 | ||||||||||
| U.S. federal income tax statutory rate | 34 | % | 34 | % | 34 | % | ||||||
| Changes in valuation allowance for deferred tax assets | 15 | % | (33 | )% | (32 | )% | ||||||
| Debt forgiveness income | (47 | )% | 0 | % | 0 | % | ||||||
| Other | (2 | )% | (1 | )% | (2 | )% | ||||||
| Effective tax rate | 0 | % | 0 | % | 0 | % | ||||||
| December 31 | ||||||||||||
2009 | 2010 | |||||||||||
| Deferred tax assets: | ||||||||||||
| Net operating loss carryforwards | $ | 45,165 | $ | 41,540 | ||||||||
| Other long-term liabilities | 4,116 | 3,099 | ||||||||||
| Property and equipment | 1,010 | 561 | ||||||||||
| Tax credit carryforwards | 437 | 453 | ||||||||||
| Other | 1,087 | 1,199 | ||||||||||
| Total gross deferred tax assets | 51,815 | 46,852 | ||||||||||
| Valuation allowance | (51,815 | ) | (46,852 | ) | ||||||||
| Net deferred tax asset | $ | — | $ | — | ||||||||
F-29
Table of Contents
| (14) | Commitments and Contingencies |
| Capital Lease | ||||
Year | Obligations | |||
| 2011 | $ | 26 | ||
| 2012 | 26 | |||
| 2013 | 21 | |||
| 2014 | 11 | |||
| 2015 | 2 | |||
| Total | 86 | |||
| Less portion representing interest | (14 | ) | ||
| Present value of future minimum payments | 72 | |||
| Current maturities of capital lease payments | (20 | ) | ||
| Long-term capital lease obligations | $ | 52 | ||
F-30
Table of Contents
| Operating | ||||
Year | Leases | |||
| 2011 | $ | 519 | ||
| 2012 | 540 | |||
| 2013 | 549 | |||
| Total minimum lease payments | $ | 1,608 | ||
| (15) | Employee Benefit Plan |
| (16) | Accrued Expenses |
| December 31 | ||||||||
2009 | 2010 | |||||||
| Accrued capital expenditures | $ | — | $ | 3,554 | ||||
| Employee compensation and related taxes | 567 | 1,860 | ||||||
| Other accrued expenses | 224 | 573 | ||||||
| $ | 791 | $ | 5,987 | |||||
| (17) | Marketable Securities |
| As of December 31, 2010 | ||||||||||||||||
| Gross | Gross | |||||||||||||||
| Amortized | Unrealized | Unrealized | Fair | |||||||||||||
Description | Cost | Gain | Losses | Value | ||||||||||||
| Government agency bonds | $ | 4,012 | $ | 8 | $ | — | $ | 4,020 | ||||||||
F-31
Table of Contents
| (18) | Subsequent Events |
F-32
Table of Contents
| December 31 | June 30 | |||||||
| 2010 | 2011 | |||||||
| (In thousands, except share and per share data) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 26,800 | $ | 29,633 | ||||
| Marketable securities | 4,020 | — | ||||||
| Accounts receivable, net of allowance for doubtful accounts | 10,205 | 12,844 | ||||||
| Costs in excess of billings | 124 | 279 | ||||||
| Inventories | 2,253 | 5,112 | ||||||
| Prepaid expenses and other current assets | 419 | 390 | ||||||
| Total current assets | 43,821 | 48,258 | ||||||
| Restricted cash | 857 | 858 | ||||||
| Property, plant, and equipment, net | 42,622 | 57,691 | ||||||
| Other assets | 1,495 | 4,064 | ||||||
| Total assets | $ | 88,795 | $ | 110,871 | ||||
| Liabilities, Redeemable Convertible Preferred Stock and Stockholders’ Deficit | ||||||||
| Current liabilities: | ||||||||
| Long-term debt, current portion | $ | 247 | $ | 110 | ||||
| Capital leases, current portion | 20 | 36 | ||||||
| Accounts payable | 5,597 | 5,399 | ||||||
| Accrued expenses | 5,987 | 4,760 | ||||||
| Deferred revenue | 447 | 1,399 | ||||||
| Other current liabilities | 6,800 | 6,000 | ||||||
| Total current liabilities | 19,098 | 17,704 | ||||||
| Convertible notes | — | 30,540 | ||||||
| Long-term debt, excluding current portion | 7,820 | 8,614 | ||||||
| Capital leases, excluding current portion | 52 | 118 | ||||||
| Other long-term liabilities | 10,142 | 8,041 | ||||||
| Total liabilities | 37,112 | 65,017 | ||||||
Commitments and contingencies (Note 13) | ||||||||
| Series B redeemable convertible preferred stock, $0.001 par value; authorized 17,000,000 shares; issued and outstanding 16,010,292 shares at December 31, 2010 and June 30, 2011, at redemption and liquidation value | 28,799 | 42,161 | ||||||
| Series A redeemable convertible preferred stock, $0.001 par value; authorized, issued and outstanding 52,843,201 shares at December 31, 2010 and June 30, 2011, at redemption and liquidation value | 80,987 | 136,229 | ||||||
| Stockholders’ deficit: | ||||||||
| Common stock, $0.001 par value; authorized 114,000,000 shares; issued and outstanding 25,574,570 and 26,119,304 shares at December 31, 2010 and June 30, 2011 | 26 | 26 | ||||||
| Additional paid-in capital | 138,038 | 69,876 | ||||||
| Accumulated deficit | (196,175 | ) | (202,438 | ) | ||||
| Accumulated other comprehensive income | 8 | — | ||||||
| Total stockholders’ deficit | (58,103 | ) | (132,536 | ) | ||||
| Total liabilities, redeemable convertible preferred stock and stockholders’ deficit | $ | 88,795 | $ | 110,871 | ||||
F-33
Table of Contents
| Six Months Ended | ||||||||
| June 30 | ||||||||
2010 | 2011 | |||||||
| (In thousands, except share and per share data) | ||||||||
| Revenue: | ||||||||
| Product | $ | 16,008 | $ | 24,333 | ||||
| Research services | 2,238 | 1,817 | ||||||
| Total revenue | 18,246 | 26,150 | ||||||
| Cost of revenue: | ||||||||
| Product | 16,114 | 21,545 | ||||||
| Research services | 1,005 | 922 | ||||||
| Gross profit | 1,127 | 3,683 | ||||||
| Operating expenses: | ||||||||
| Research and development | 1,719 | 1,728 | ||||||
| Selling and marketing | 2,318 | 2,557 | ||||||
| General and administrative | 2,813 | 3,443 | ||||||
| Total operating expenses | 6,850 | 7,728 | ||||||
| Income (loss) from operations | (5,723 | ) | (4,045 | ) | ||||
| Other income (expense): | ||||||||
| Interest income | 37 | 51 | ||||||
| Interest expense | (1,404 | ) | (2,269 | ) | ||||
| Total other expense, net | (1,367 | ) | (2,218 | ) | ||||
| Net income (loss) | (7,090 | ) | (6,263 | ) | ||||
| Dividends and accretion of redeemable convertible preferred stock | (39,264 | ) | (68,604 | ) | ||||
| Net income (loss) attributable to common stockholders | $ | (46,354 | ) | $ | (74,867 | ) | ||
| Net income (loss) per common share attributable to common stockholders, basic and diluted | $ | (1.81 | ) | $ | (2.88 | ) | ||
| Weighted-average common shares outstanding, basic and diluted | 25,573,997 | 26,005,705 | ||||||
F-34
Table of Contents
| Six Months Ended | ||||||||
| June 30 | ||||||||
2010 | 2011 | |||||||
| (In thousands) | ||||||||
| Cash flows from operating activities: | ||||||||
| Net income (loss) | $ | (7,090 | ) | $ | (6,263 | ) | ||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 2,276 | 3,365 | ||||||
| Imputed interest | 1,311 | 1,379 | ||||||
| Accretion of convertible notes to fair value | — | 540 | ||||||
| Gain on sale of marketable securities | — | (14 | ) | |||||
| Stock compensation expense | 201 | 442 | ||||||
| Settlement of asset retirement obligation | (15 | ) | (12 | ) | ||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (2,036 | ) | (2,639 | ) | ||||
| Costs in excess of billings | (442 | ) | (155 | ) | ||||
| Inventories | (1,183 | ) | (2,859 | ) | ||||
| Other current assets | (38 | ) | 252 | |||||
| Accounts payable | (9 | ) | 664 | |||||
| Accrued expenses | 1,116 | 906 | ||||||
| Deferred revenue | 183 | 952 | ||||||
| Other long-term liabilities | (3,700 | ) | (3,800 | ) | ||||
| Net cash used in operating activities | (9,426 | ) | (7,242 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Capital expenditures | (1,633 | ) | (20,870 | ) | ||||
| Increase in restricted cash | (92 | ) | (1 | ) | ||||
| Purchase of marketable securities | (11,042 | ) | — | |||||
| Proceeds from maturities and sales of marketable securities | — | 4,026 | ||||||
| Net cash used in investing activities | (12,767 | ) | (16,845 | ) | ||||
| Cash flows from financing activities: | ||||||||
| Proceeds from issuance of convertible notes | — | 30,000 | ||||||
| Repayment of borrowings under long-term debt | (131 | ) | (139 | ) | ||||
| Deferred financing costs | (276 | ) | (551 | ) | ||||
| Deferred offering costs | — | (2,372 | ) | |||||
| Repayment of obligations under capital lease | (19 | ) | (18 | ) | ||||
| Payment of stock option exercise | 2 | — | ||||||
| Net cash (used in) / provided by financing activities | (424 | ) | 26,920 | |||||
| Net (decrease) / increase in cash | (22,617 | ) | 2,833 | |||||
| Cash at beginning of period | 27,502 | 26,800 | ||||||
| Cash at end of period | $ | 4,885 | $ | 29,633 | ||||
| Supplemental disclosures of cash flow information: | ||||||||
| Interest paid | $ | 14 | $ | 315 | ||||
| Income taxes paid | $ | — | $ | — | ||||
| Supplemental disclosures of non-cash activities: | ||||||||
| Accrued dividends on preferred stock | $ | 1,223 | $ | 2,074 | ||||
| Accretion of preferred stock | $ | 38,041 | $ | 66,530 | ||||
| Change in accrued capital expenditures | $ | 2,184 | $ | (2,995 | ) | |||
| Capital lease | $ | — | $ | 100 | ||||
F-35
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(Unaudited)
(In thousands, except share and per share data)
| (1) | Description of Business |
F-36
Table of Contents
| (2) | Significant Accounting Policies |
F-37
Table of Contents
F-38
Table of Contents
| (3) | Related Party Transactions |
| (4) | Inventories |
| December 31 | June 30 | |||||||
2010 | 2011 | |||||||
| Raw material | $ | 1,657 | $ | 1,927 | ||||
| Work in Process | — | 69 | ||||||
| Finished goods | 596 | 3,116 | ||||||
| Total | $ | 2,253 | $ | 5,112 | ||||
| (5) | Redeemable Convertible Preferred Stock |
| Series B (2010) | Series A (2009) | |||||||||||||||
Shares | Amount | Shares | Amount | |||||||||||||
| Balance at December 31, 2010 | 16,010,292 | $ | 28,799 | 52,843,201 | $ | 80,987 | ||||||||||
| Accretion to redemption value | — | 12,512 | — | 54,018 | ||||||||||||
| Dividends | — | 850 | — | 1,224 | ||||||||||||
| Balance at June 30, 2011 | 16,010,292 | $ | 42,161 | 52,843,201 | $ | 136,229 | ||||||||||
F-39
Table of Contents
| (6) | Employee Stock Options |
| (7) | Debt |
| December 31 | June 30 | |||||||
2010 | 2011 | |||||||
| 6% Term Loan | $ | 247 | $ | 110 | ||||
| Subordinated Notes, net of discount | 7,820 | 8,614 | ||||||
| Total long-term debt | 8,067 | 8,724 | ||||||
| Current maturities of long-term debt | (247 | ) | (110 | ) | ||||
| Long-term debt, less current maturities | $ | 7,820 | $ | 8,614 | ||||
F-40
Table of Contents
F-41
Table of Contents
| (8) | Fair Value Option and Fair Value Measurements |
| Aggregate | ||||||||||||
| Aggregate | unpaid principal | Fair value | ||||||||||
| fair value | balance under FVO | carrying amount | ||||||||||
| June 30, | June 30, | over unpaid | ||||||||||
| 2011 | 2011 | principal | ||||||||||
| Convertible Notes | $ | 30,540 | $ | 30,000 | $ | 540 | ||||||
| June 30, 2011 | ||||||||||||||||
Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Convertible Notes | $ | — | $ | — | $ | 30,540 | $ | 30,540 | ||||||||
| $ | — | $ | — | $ | 30,540 | $ | 30,540 | |||||||||
F-42
Table of Contents
| Balance at January 1, 2011 | $ | — | ||
| Additions | 30,000 | |||
| Change in fair value | 540 | |||
| Balance at June 30, 2011 | $ | 30,540 | ||
| (9) | Net Income (Loss) Per Share |
| Six Months Ended | ||||||||
| June 30 | ||||||||
2010 | 2011 | |||||||
| Net income (loss) | $ | (7,090 | ) | $ | (6,263 | ) | ||
| Deemed dividends on participating preferred stock (inclusive of issuance cost and accretion to redemption value): | ||||||||
| Series A (2009) | (39,264 | ) | (55,242 | ) | ||||
| Series B (2010) | — | (13,362 | ) | |||||
| Total preferred stock deemed dividends | (39,264 | ) | (68,604 | ) | ||||
| Net income (loss) attributable to common stockholders | $ | (46,354 | ) | $ | (74,867 | ) | ||
| Weighted average shares outstanding | 25,573,997 | 26,005,705 | ||||||
| Net income (loss) per share attributable to common stockholders, basic and diluted | $ | (1.81 | ) | $ | (2.88 | ) | ||
| June 30 | ||||||||
2010 | 2011 | |||||||
| Common stock options | 10,579,089 | 14,110,658 | ||||||
| Common stock warrants | 134,941 | 1,127,324 | ||||||
| Series A (2009) | 52,843,201 | 52,843,201 | ||||||
| Series B (2010) | — | 16,010,292 | ||||||
| Total | 63,557,231 | 84,091,475 | ||||||
F-43
Table of Contents
| (10) | Other Long-term Liabilities |
| December 31 | June 30 | |||||||
2010 | 2011 | |||||||
| ARO | $ | 953 | $ | 956 | ||||
| Cross License Agreement | 15,989 | 13,085 | ||||||
| 16,942 | 14,041 | |||||||
| Current maturities of other long-term liabilities | (6,800 | ) | (6,000 | ) | ||||
| Other long-term liabilities, less current maturities | $ | 10,142 | $ | 8,041 | ||||
| Six Months Ended | ||||||||
| June 30 | ||||||||
2010 | 2011 | |||||||
| Balance at beginning of period | $ | 964 | $ | 953 | ||||
| Accretion of discount expense | 15 | 15 | ||||||
| Settlement costs | (15 | ) | (12 | ) | ||||
| Balance at end of period | $ | 964 | $ | 956 | ||||
F-44
Table of Contents
| (12) | Accrued Expenses |
| December 31 | June 30 | |||||||
2010 | 2011 | |||||||
| Accrued capital expenditures | $ | 3,554 | $ | 1,421 | ||||
| Employee compensation and related taxes | 1,860 | 913 | ||||||
| Accrued professional fees | 474 | 1,352 | ||||||
| Other accrued expenses | 99 | 1,074 | ||||||
| $ | 5,987 | $ | 4,760 | |||||
| (13) | Commitments and Contingencies |
F-45
Table of Contents

| Goldman, Sachs & Co. | Morgan Stanley |
Table of Contents
| Item 13. | Other Expenses of Issuance and Distribution. |
| Amount to | ||||
be paid | ||||
| SEC registration fee | $ | 13,352 | ||
| NYSE listing fee | * | |||
| FINRA filing fee | 12,000 | |||
| Printing and mailing | * | |||
| Legal fees and expenses | * | |||
| Accounting fees and expenses | * | |||
| Blue sky fees and expenses | * | |||
| Transfer agent and registrar | * | |||
| Miscellaneous | * | |||
| Total | $ | * | ||
| * | To be provided by amendment. |
| Item 14. | Indemnification of Directors and Officers. |
II-1
Table of Contents
| • | from any breach of the director’s duty of loyalty to us or our stockholders; | |
| • | from acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law; | |
| • | under Section 174 of the Delaware General Corporation Law; and | |
| • | from any transaction from which the director derived an improper personal benefit. |
| Item 15. | Recent Sales of Unregistered Securities. |
II-2
Table of Contents
II-3
Table of Contents
II-4
Table of Contents
| Item 16. | Exhibits and Financial Statement Schedules. |
| Item 17. | Undertakings |
II-5
Table of Contents
| By: | /s/ Donald R. Young |
Signature | Title | Date | ||||
/s/ Donald R. Young Donald R. Young | President, Chief Executive Officer and Director (principal executive officer) | September 1, 2011 | ||||
/s/ John F. Fairbanks John F. Fairbanks | Chief Financial Officer, Vice President and Treasurer (principal financial and accounting officer) | September 1, 2011 | ||||
* Mark L. Noetzel | Chairman of the Board | September 1, 2011 | ||||
* P. Ramsay Battin | Director | September 1, 2011 | ||||
* Robert M. Gervis | Director | September 1, 2011 | ||||
* Craig A. Huff | Director | September 1, 2011 | ||||
* Steven R. Mitchell | Director | September 1, 2011 | ||||
* William P. Noglows | Director | September 1, 2011 | ||||
* David J. Prend | Director | September 1, 2011 | ||||
II-6
Table of Contents
Signature | Title | Date | ||||
* Richard F. Reilly | Director | September 1, 2011 | ||||
| *By: | /s/ Donald R. Young Donald R. Young Attorney-in-fact | September 1, 2011 | ||||
II-7
Table of Contents
| Exhibit | ||||
number | Description of Exhibit | |||
| 1 | .1* | Form of underwriting agreement. | ||
| 3 | .1.1+ | Third amended and restated certificate of incorporation of the Registrant, as amended. | ||
| 3 | .1.2* | Certificate of amendment to the third amended and restated certificate of incorporation, as amended, of the Registrant. | ||
| 3 | .2* | Form of restated certificate of incorporation of the Registrant to be filed with the Secretary of State of the State of Delaware upon completion of this offering. | ||
| 3 | .3+ | By-laws of the Registrant, as amended. | ||
| 3 | .4* | Form of restated by-laws of the Registrant to be effective upon completion of this offering. | ||
| 4 | .1 | Form of common stock certificate. | ||
| 4 | .2+ | Form of warrant to purchase common stock issued by the Registrant in connection with 2004 and 2005 financing arrangements, as amended and restated. | ||
| 4 | .3+ | Form of warrant to purchase common stock issued by the Registrant in connection with the 2005 equity financing, as amended and restated. | ||
| 4 | .4+ | Form of warrant to purchase common stock issued by the Registrant in connection with the 2008 reorganization. | ||
| 4 | .5+ | Form of warrant to purchase common stock issued by the Registrant in connection with the 2008 financing. | ||
| 4 | .6+ | Form of warrant to purchase common stock issued by the Registrant in connection with the 2010 subordinated note and warrant financing. | ||
| 5 | .1* | Opinion of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C., counsel to the Registrant, with respect to the legality of securities being registered. | ||
| 10 | .1.1+@ | 2001 equity incentive plan, as amended. | ||
| 10 | .1.2+@ | Form of incentive stock option agreement granted under 2001 equity incentive plan, as amended. | ||
| 10 | .1.3+@ | Form of non-qualified stock option agreement granted under 2001 equity incentive plan, as amended. | ||
| 10 | .2.1*@ | 2011 employee, director and consultant equity incentive plan. | ||
| 10 | .2.2*@ | Form of stock option agreement granted under 2011 employee, director and consultant equity incentive plan. | ||
| 10 | .3+ | Multi-tenant industrial net lease, dated August 20, 2001, by and between the Registrant and Cabot II — MA1M03, LLC (as successor landlord to TMT290 Industrial Park, Inc.), as amended. | ||
| 10 | .4+ | Loan and security agreement by and between the Registrant and Silicon Valley Bank, dated as of March 31, 2011, as amended. | ||
| 10 | .5+ | Form of subordinated note issued by the Registrant in the 2010 subordinated note and warrant financing. | ||
| 10 | .6+ | Form of convertible note issued by the Registrant in the 2011 convertible note financing. | ||
| 10 | .7+@ | Executive agreement by and between the Registrant and Donald R. Young. | ||
| 10 | .8+@ | Executive agreement by and between the Registrant and John F. Fairbanks. | ||
| 10 | .9+@ | Executive agreement by and between the Registrant and Harry R. Walkoff. | ||
| 10 | .10+@ | Executive agreement by and between the Registrant and Kevin A. Schmidt. | ||
| 10 | .11+@ | Executive agreement by and between the Registrant and George L. Gould, Ph.D. | ||
| 10 | .12+@ | Executive agreement by and between the Registrant and Christopher L. Marlette. | ||
| 10 | .13+@ | 2010 Corporate Bonus Plan. | ||
| 10 | .14+@ | Director compensation policy. | ||
| 10 | .15+ | Fifth amended and restated registration rights agreement, dated as of September 22, 2010, by and among the Registrant and Investors (as defined therein), as amended. | ||
| 10 | .16# | Joint development agreement dated as of March 1, 2010 by and between BASF Construction Chemicals GmbH and the Registrant, as amended. | ||
Table of Contents
| Exhibit | ||||
number | Description of Exhibit | |||
| 10 | .17# | Cross license agreement dated as of April 1, 2006 by and between Cabot Corporation and the Registrant, as amended. | ||
| 10 | .18# | Supply agreement, dated as of January 1, 2011, by and between Silbond Corporation and the Registrant. | ||
| 10 | .19@ | 2011 Corporate Bonus Plan. | ||
| 21 | .1+ | Subsidiaries of the Registrant. | ||
| 23 | .1 | Consent of KPMG LLP. | ||
| 23 | .2* | Consent of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. (see Exhibit 5.1). | ||
| 23 | .3+ | Consent of Freedonia Custom Research, Inc. | ||
| 24 | .1+ | Powers of Attorney (see signature page to initial filing). | ||
| + | Previously filed. |
| * | To be filed by amendment. |
| # | Confidential treatment has been requested for portions of this exhibit. |
| @ | Denotes management compensation plan or contract. |