Exhibit 99.1

01 | 10 | 2022 ANGIODYNAMICS Needham Healthcare ConferenceJim Clemmer, President & CEO Stephen Trowbridge, EVP & CFO 1
2 Notice Regarding Forward-Looking StatementsThis presentation containsforward-looking statementswithin the meaning of the Private Securities Litigation Reform Act of 1995. All statementsregarding AngioDynamics’ expected future financial position, resultsof operations, cash flows, business strategy, budgets, projected costs, capital expenditures, products, competitive positions, growth opportunities, plans and objectives of management for future operations, as well as statements that include the words such as “expects,” “reaffirms,” “intends,” “anticipates,” “plans,” “projects,” “believes,” “seeks,” “estimates,” “optimistic,” or variations of such words and similar expressions, are forward-looking statements. These forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ materially from AngioDynamics’ expectations, expressed or implied. Factors that may affect the actual results achieved by AngioDynamics include, without limitation, the scale and scope of the COVID-19 global pandemic, the ability of AngioDynamicsto develop its existing and new products, technological advances and patentsattainedby competitors, infringement of AngioDynamics’ technology or assertions thatAngioDynamics’ technology infringes the technology of third parties, the ability of AngioDynamics to effectively compete against competitors that have substantially greater resources, future actions by the FDA or other regulatory agencies, domestic and foreign health care reforms and government regulations, results of pending or future clinical trials, overall economic conditions (including inflation and labor shortages), the results of on-going litigation, challenges with respect to third-party distributors or joint venture partners or collaborators, the results of sales efforts, the effects of product recalls and product liability claims, changes in key personnel, the ability of AngioDynamics to execute on strategic initiatives, the effects of economic, credit and capital market conditions, general market conditions, market acceptance, foreigncurrency exchange rate fluctuations, the effects on pricing from group purchasing organizations and competition, the ability of AngioDynamics to obtain regulatory clearances or approval of its products, or integrate acquired businesses, as well as the risk factors listed from time to time in AngioDynamics’ SEC filings, including but not limited to its Annual Report on Form 10-K for the year ended May 31, 2021. AngioDynamics does not assume any obligation to publicly update or revise any forward-looking statementsfor any reason.In the United States, the NanoKnife System has received a 510(k) clearance by the Food and Drug Administrationfor use in the surgical ablation of soft tissue, and is similarly approved for commercialization inCanada, the European Union and Australia. The NanoKnife System has not been cleared for the treatmentor therapy of a specific disease or condition.Notice Regarding Non-GAAP Financial MeasuresManagement uses non-GAAP measures to establish operational goals and believes that non-GAAP measures may assist investors in analyzing the underlying trends in AngioDynamics’ business over time. Investorsshould consider these non-GAAP measures in addition to, not as a substitute for or as superior to, financial reporting measures prepared in accordance with GAAP. In this presentation, AngioDynamics has included adjusted EBITDA, adjusted net income (loss), and adjusted earnings per share. Management uses these measures in its internal analysis and review of operational performance. Management believes that these measures provide investorswith useful informationin comparing AngioDynamics’ performance over different periods. By using these non-GAAP measures, management believes that investors get a better picture of the performance of AngioDynamics’ underlying business. Management encourages investors to review AngioDynamics’ financial results prepared in accordance with GAAP to understand AngioDynamics’ performance taking into account all relevant factors, including those that may only occur from time to time but have a material impact on AngioDynamics’ financial results.

3 FOCUSED TRANSFORMATION U.S. Total Addressable Markets FY2021Launch of the Auryon System gives us access to the peripheral atherectomy market $3.0B2-5% Mkt CAGR $5.5B3-7% Mkt CAGR $8.0B3-7% Mkt CAGR FY2023Planned Thrombectomy & NanoKnife System portfolio additions & new indications increase market access The planned portfolio additions and new indications are not guarantees of future performance and are subject to risks and uncertaintiesincluding FDA clearance. Investors are cautioned that actual events or results may differ from AngioDynamics’ expectations. FY2025Planned Thrombectomy & PE portfolio additions & new indications increase market access FY2018Began our strategic initiative to become a growth company $1.3B0-3%Mkt CAGR

4 MED DEVICEMaintain Positioning Irreversible Electroporation Peripheral Atherectomy MED TECHInvest for GrowthThrombus Management
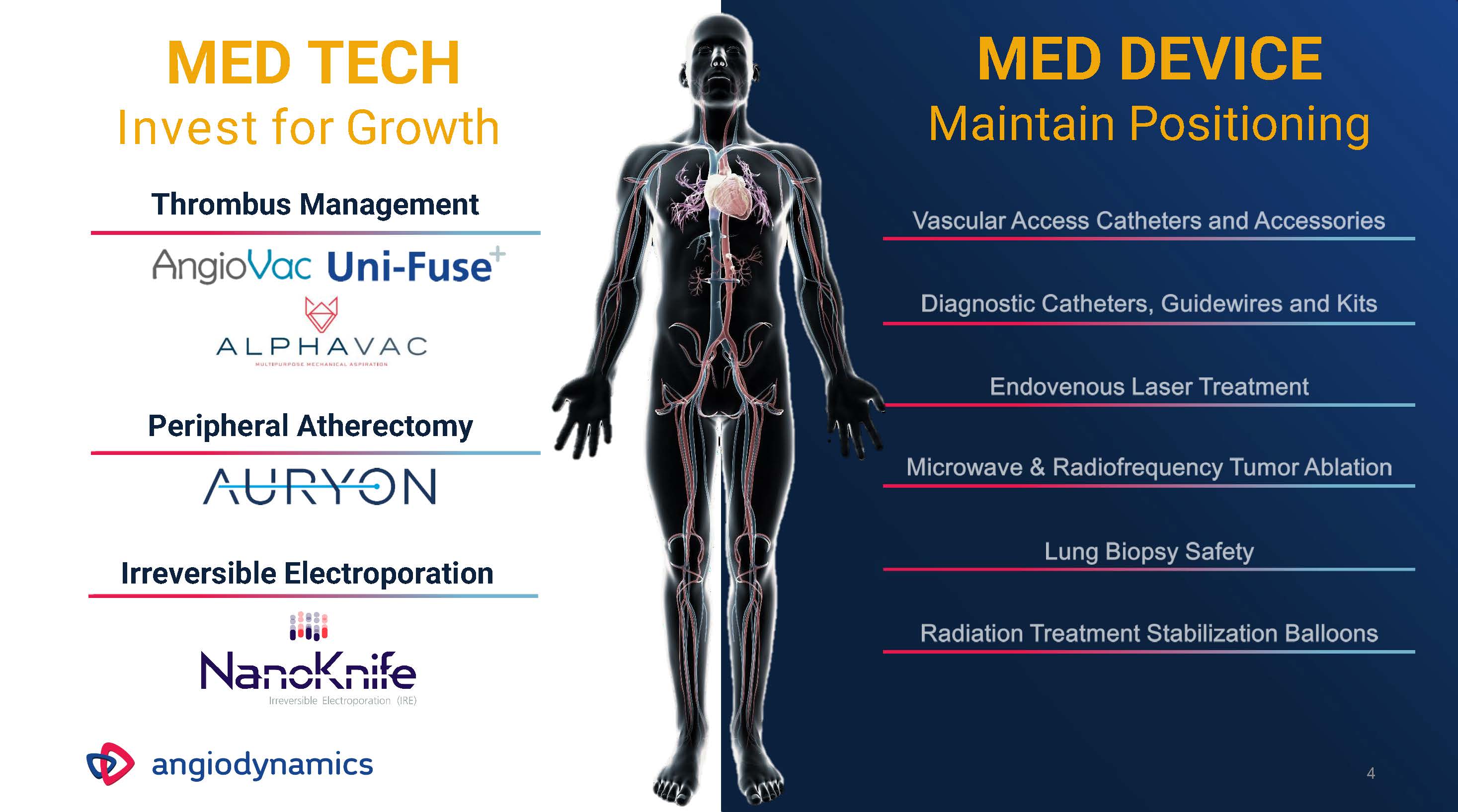
THROMBUS MANAGEMENT MED TECH

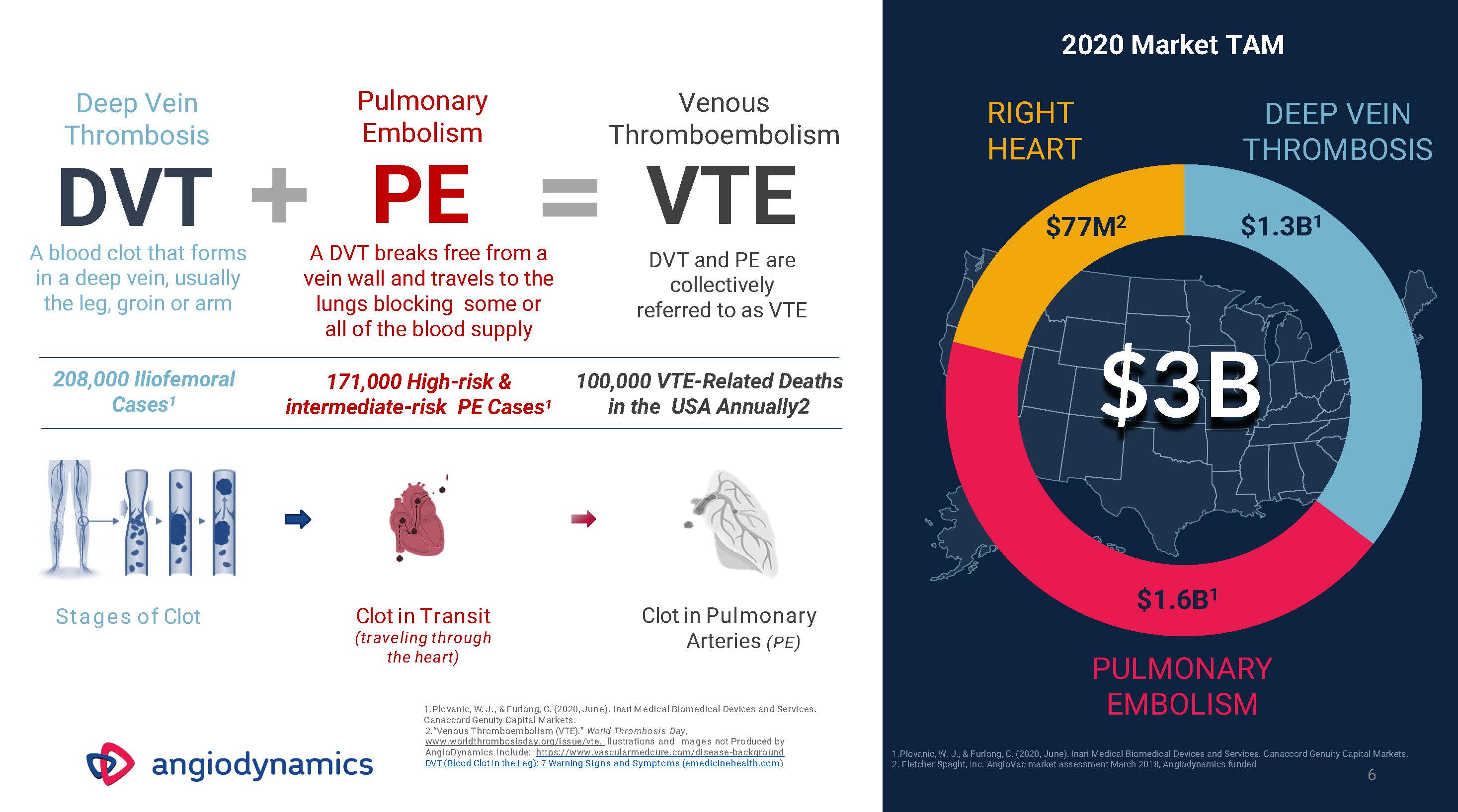
Deep Vein Thrombosis Pulmonary Embolism PE A blood clot that forms in a deep vein, usually the leg, groin or arm A DVT breaks free from a vein wall and travels to the lungs blocking some or all of the blood supply 208,000 Iliofemoral Cases1 171,000 High-risk & intermediate-risk PE Cases1 Stages of Clot Clot in Transit(traveling through the heart) Clot in Pulmonary Arteries (PE) Plovanic, W. J., & Furlong, C. (2020, June). Inari Medical Biomedical Devices and Services. Canaccord Genuity Capital Markets.“Venous Thromboembolism (VTE).” World Thrombosis Day, www.worldthrombosisday.org/issue/vte. Illustrations and Images not Produced by AngioDynamics Include: https://www.vascularmedcure.com/disease-background DVT (Blood Clot In the Leg): 7 Warning Signs and Symptoms (emedicinehealth.com) 6 RIGHT HEART $3B DEEP VEIN THROMBOSIS 2020 Market TAM 2. Fletcher Spaght, Inc. AngioVac market assessment March 2018, Angiodynamics funded DVT + = VTE DVT and PE are collectively referred to as VTE Venous Thromboembolism 100,000 VTE-Related Deaths in the USA Annually2 $77M2 $1.3B1 $1.6B$1.6B1PULMONARYEMBOLISM1.Plovanic, W. J., & Furlong, C. (2020, June). Inari Medical Biomedical Devices and Services. Canaccord Genuity Capital Markets.
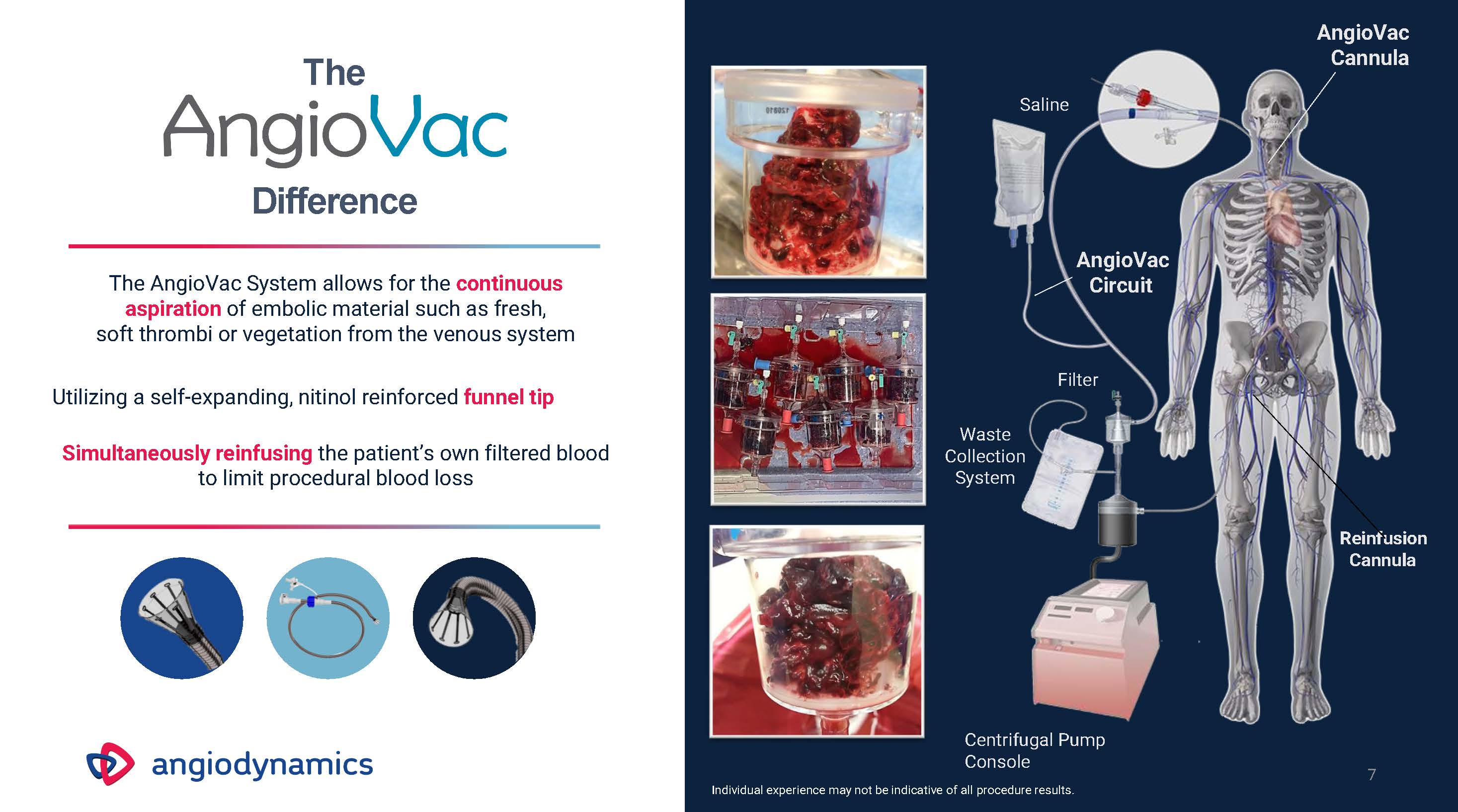
The Difference The AngioVac System allows for the continuous aspiration of embolic material such as fresh,soft thrombi or vegetation from the venous system Utilizing a self-expanding, nitinol reinforced funnel tip Simultaneously reinfusing the patient’s own filtered bloodto limit procedural blood loss Waste Collection System Filter Saline AngioVac Cannula Reinfusion Cannula AngioVacCircuit 7 Centrifugal PumpConsoleIndividual experience may not be indicative of all procedure results.
Control Features for Handle & Cannula SYRINGE BARRELAspirate between 10 & 30cc / pull VOLUME LIMITING SWITCHAllows tailored aspiration volume VACUUM LOCKAllows for continuous aspiration When engaged in clot MANUAL ASPIRATION HANDLEControlled, single-hand design MULTIPLE TIP ANGLES00, 200, 850, 1800 PROPRIETARY FUNNEL DESIGNAllows for Massive Clot Removal | En Bloc RADIOPAQUE MARKERSBetter Tip Visibility LARGE END HOLE ASPIRATION 42FR & 30FROpening COMPLETE CONTROL INTUITIVELY SIMPLE EFFICIENTLY POWERFUL

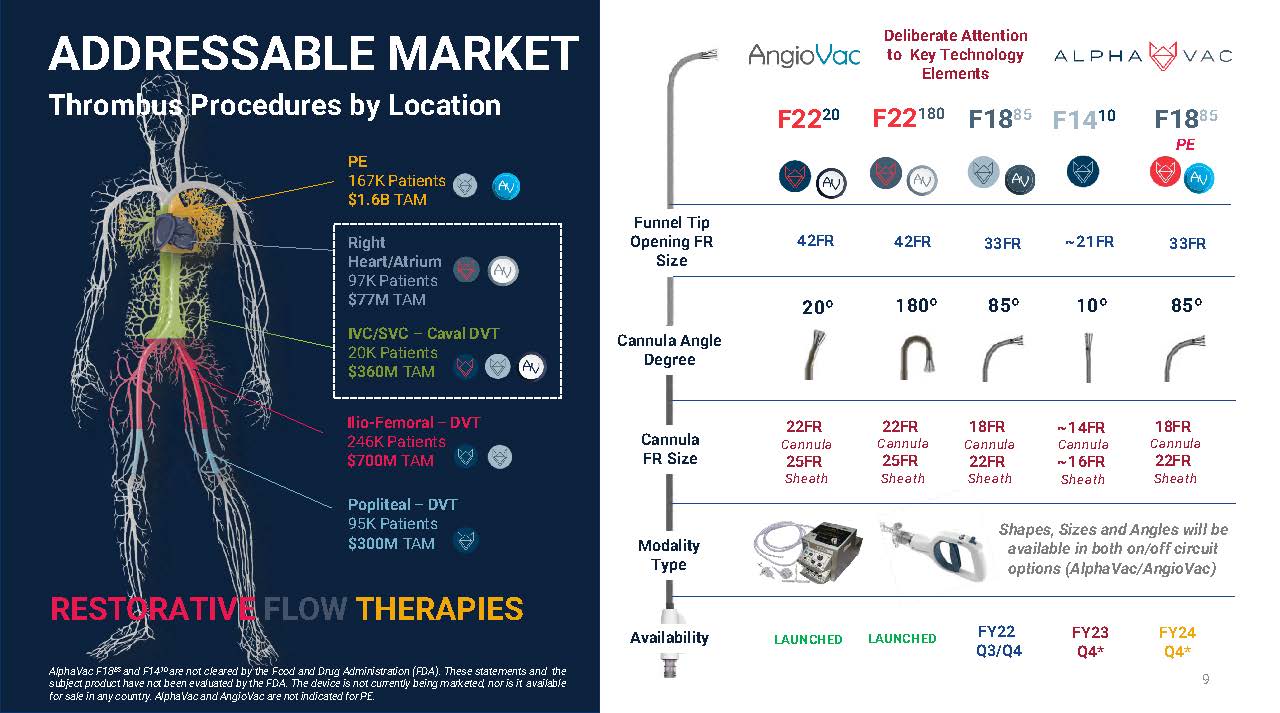
ADDRESSABLE MARKET Thrombus Procedures by Location Right Heart/Atrium 97K Patients$77M TAMIVC/SVC – Caval DVT20K Patients$360M TAM PE167K Patients$1.6B TAM Popliteal – DVT95K Patients$300M TAM Ilio-Femoral – DVT246K Patients$700M TAM RESTORATIVE FLOW THERAPIES Funnel Tip Opening FR Size Cannula AngleDegree Cannula FR Size Modality Type F2220 F22180 F1885 F1410 F1885PE 42FR 42FR 33FR ~21FR 33FR 20º 180º 85º 10º 85º 22FR 22FR 18FR ~14FR 18FR Cannula Cannula Cannula Cannula Cannula 25FR 25FR 22FR ~16FR 22FR Sheath Sheath Sheath Sheath Sheath Deliberate Attention to Key Technology Elements Shapes, Sizes and Angles will be available in both on/off circuit options (AlphaVac/AngioVac) Availability LAUNCHED FY22 Q3/Q4 FY23Q4* FY24Q4* AlphaVac F1885 and F1410 are not cleared by the Food and Drug Administration (FDA). These statements and the subject product have not been evaluated by the FDA. The device is not currently being marketed, nor is it available for sale in any country. AlphaVac and AngioVac are not indicated for PE. 9 LAUNCHED
10 THROMBUS MANAGEMENT Planned Portfolio Additions &U.S. Addressable Markets Expansion FY 2021 FY 2022 FY 2025 Q3 Q4 Q1 Q2 Q4FY 2023 Q1 Q3 Q4FY 2024 $140M $700M $3.6B $1.5B 2 (F2220, F22180)Right Atrium, Illio -caval DVT (F2220 - F22180 - F1885)Entry into Ilio-femoral DVT (F1410)Access to full Ilio-femoral DVT market (F1885)Smaller size should drive further adoption in addressable market Purpose Built, Comprehensive, Thrombus Portfolio F22180 F2220 F1885 F1885 PE F1410 Continuous Aspiration with Simultaneous Reinfusion Multi-purpos e Mechanical/Manual As piration Catheter Directed Thrombolysis with PE Indication F22180 F2220 F1885 F1885 PE F1410 The planned portfolio additions are not guarantees of future performance and are subject to risks and uncertainties, including clearance by the FDA. Investors are cautioned that actual events or results may differ from AngioDynamics’ expectations. (F1885 PE)PE Indication

PERIPHERAL ATHERECTOMY MED TECH
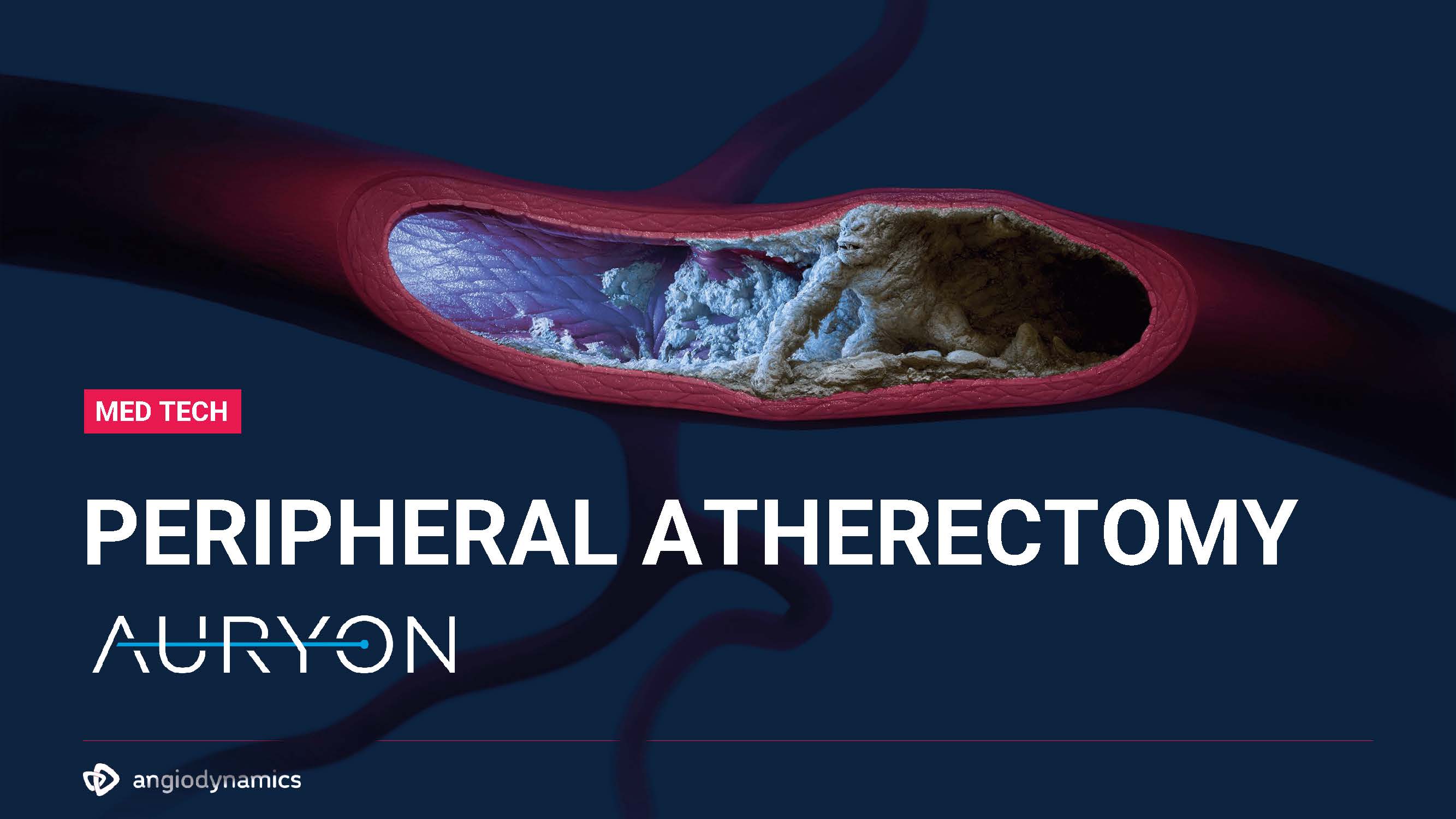
$600M 23%3 77%3 Over 8 Million2 Americans Suffer from P AD Over 150,000 Limbs4 are Lost Every Yearbecause of PAD 50% Mortality Rate4 Associated with PAD after Limb Loss PERIPHERAL ATHERECTOMYUS Addressable Markets & Competitive Landscape Peripheral Vascular Devices Medtech 360 Market Analysis US 2017. (2016, December). Millenium Research Group, Inc.https://www.cookmedical.com/peripheral-intervention/10-facts-about- peripheral-arterial-disease/ 12 2021 Served MarketMECHANICAL LASER

2.35 mmAspiration and Off-Center capabilities and indicated for Peripheral Atherectomy and In-Stent Restenosis (ISR) 2.0 mmAspiration capability and indicated for Peripheral Atherectomy and ISR 1.5 mmIndicated for Peripheral Atherectom y Why wavelength mattersEach type of tissue interacts differently with a given wavelength Why pulse width and amplitude matterGreater amplitude is achieved with shorter pulses, which can deposit energy before thermal diffusion occurs The Auryon System produces a photon energy of 3.5 eV, which is low enough to be nonreactive to vessel endothelium,but high enough to vaporize calcium. 6,7 The Auryon System has a pulse width of10 to 25 ns, ensuring enough power to target the lesion and spare the vessel. 5 0.9 mmIndicated for Peripheral Atherectom y 5. Auryon. Instructions for use. AngioDynamics; 2019. 6. Herzog A, Bogdan S, Glikson M, I s h a a y a AA, Love C. Selective t issue ablation u s in g laser radiation at 355 nm in lead extraction by a hybrid catheter; a preliminary report. Lasers Surg Med. 2016;48(3):281-287.7. Spectranetics Corporation. CVX-300 Excimer Laser System: Operator’s Manual. Version 28. 2019:1-56. 13

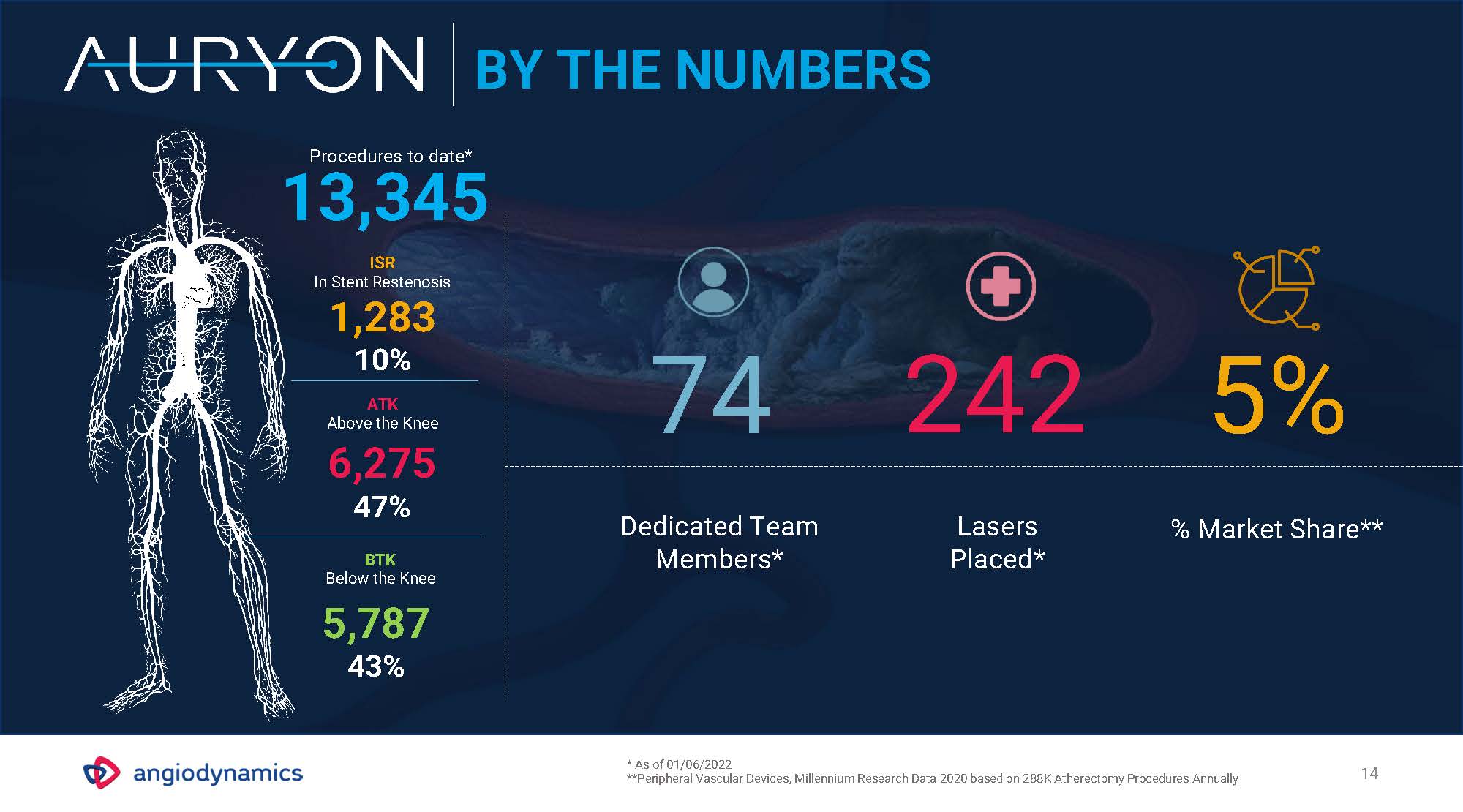
14 BY THE NUMBERS Dedicated Team Members* % Market Share** BTKBelow the Knee5,78743% 74 242 5% Lasers Placed* 13,345ISRIn Stent Restenosis1,283 10% ATKAbove the Knee6,27547% Procedures to date* * As of 01/06/2022**Peripheral Vascular Devices, Millennium Research Data 2020 based on 288K Atherectomy Procedures Annually
Procedural Hub at all Sites of Service OBL/ASC/Hospital Technology Pipeline PAD AtherectomyCurrent Indication Arterial ThrombectomyIn Development Venous ThrombectomyIn Development Coronary AtherectomyAdjacent Expansion Opportunity Lead ExtractionAdjacent Expansion Opportunity ISR ATK BTK Acute PAD Sub-Acute Chronic Laser Debulking Laser Removal Chronic Total OcclusionIn Development CTO crossing without a wire 15The planned portfolio additions are not guarantees of future performance and are subject to risks and uncertainties, includingclearance by the FDA. Investors are cautioned that actual events or results may differ from AngioDynamics’ expectations.

IRREVERSIBLE ELECTROPORATION MED TECH 16

INNOVATION DOCTORS NEEDExpands treatment options and help preserve patient's quality of life DECELLULARIZATIONDestroys targeted tissue with precise treatment margins.10,11 PROBE PLACEMENTNanoKnife can be confidently used in all segments of an organ.10,11 NON-THERMALSpares vital structures by retaining the structural integrity of tissue.12,13 REVASCULARIZATIONFacilitates functional tissue regeneration post-ablation.12,13 Lee EW, Thai S, Kee ST. Irreversible electroporation: a novel image-guided cancer therapy. Gut Liver. (2010);4(SUPPL. 1):99–104. doi: 10.5009/gnl.2010.4.S1.S99Guidance for Selection of NanoKnife Probe Array Configuration and Ablation parameters for the Treatment of Stage III Pancreatic Cancer.Scheltema MJ, Chang JI, van den Bos W, Gielchinsky I, Nguyen TV, Reijke TM, Siriwardana AR, Böhm M, de la Rosette JJ, Stricker PD. Impact on genitourinary function and quality of life following focal irreversible electroporation of different prostate segments. Diagn Interv Radiol. 2018 Sep;24(5):268-275. doi: 10.5152/dir.2018.17374. PMID: 30211680; PMCID: PMC6135060.Li W, Fan Q, Ji Z, Qiu X, Li Z. The effects of irreversible electroporation (IRE) on nerves. PLoS One. 2011 Apr 14;6(4):e18831. doi: 10.1371/journal.pone.0018831. PMID: 21533143; PMCID: PMC3077412. 17

$1.6B 18 22%1,2 8%1,2 PROSTATE 70%1,2 2021 Total Addressable Market (TAM)PANCREAS LIVER THE NANOKNIFE SYSTEMEstimated # of US Patients Diagnosed in 20211 Liver42,230 Pancreas60,430 Prostate248,530

Prostate cancer is the only solid tumor without a standardized local treatment option1Advances in genetics, imaging, and methodology support a local treatment option for PCa2But existing technology has less than ideal outcomes for cancer control and quality of life3,4 19 Approximately 100,000men are estimated to be candidates for focal therapy each year5 PROSTATE INITIATIVE Effectively destroys targeted tissue with precise treatment margins6 Spares vital structures within the ablation zone7 Enables treatment to be performed in all segments of an organ8 Bridge the gap between an active surveillance strategy and whole-gland treatment NCCN Guidelines for Patients Early Stage Prostate Cancer. https://www.nccn.org/patients/guidelines/content/PDF/prostate-early-patient.pdf.Lee, Byron H., et al. “Changing Landscape of Prostate Cancer Favoring Low-Risk Prostate Cancer: Implications for Active Surveillance Versus Focal Therapy.” Imaging and Focal Therapy of Early Prostate Cancer, 2012, pp. 17–36., doi:10.1007/978-1-62703-182-0_2. 3). Worrell Design. Next Gen Voice of Customer, 2020.Sivaraman A, Barret E. Focal Therapy for Prostate Cancer: An "À la Carte" Approach. Eur Urol. 2016;69(6):973-975. doi:10.1016/j.eururo.2015.12.015Cancer Facts & Figures 2021. American Cancer Society. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2021.html. Accessed January 4, 2022. 6). Lee EW, Thai S, Kee ST. Irreversible electroporation: a novel image-guided cancer therapy. Gut Liver. (2010);4(SUPPL. 1):99–104. doi: 10.5009/gnl.2010.4.S1.S997). Li W, Fan Q, Ji Z, Qiu X, Li Z. The effects of irreversible electroporation (IRE) on nerves. PLoS One. 2011 Apr 14;6(4):e18831. doi: 10.1371/journal.pone.0018831. PMID: 21533143; PMCID: PMC3077412. 8.) Scheltema MJ, Chang JI, van den Bos W, Gielchinsky I, Nguyen TV, Reijke TM, Siriwardana AR, Böhm M, de la Rosette JJ, Stricker PD. Impact on genitourinary function and quality of life following focal irreversible electroporation of different prostate segments. Diagn Interv Radiol. 2018 Sep;24(5):268-275. doi: 10.5152/dir.2018.17374. PMID: 30211680; PMCID: PMC6135060.

20 PRESERVE Prostate IDE 37 SUO-CTC US sites responded to Call for Sites 20 Sites selected, focused on geographic and demographic diversity, high-volume focal therapy institutions 118 Intermediate-risk patients to be enrolled through 1-year follow up SUO-CTC is a clinical research investigator network of 500+ members from more than 250 clinical sites in the US and Canada. Primary Endpoint: Rate of negative in-field biopsy at 1 yearFirst Enrollment: Expected Q3 Enrollment Period: 12 monthsDate of last update: Jan 7, 2022

FINANCIALS 21

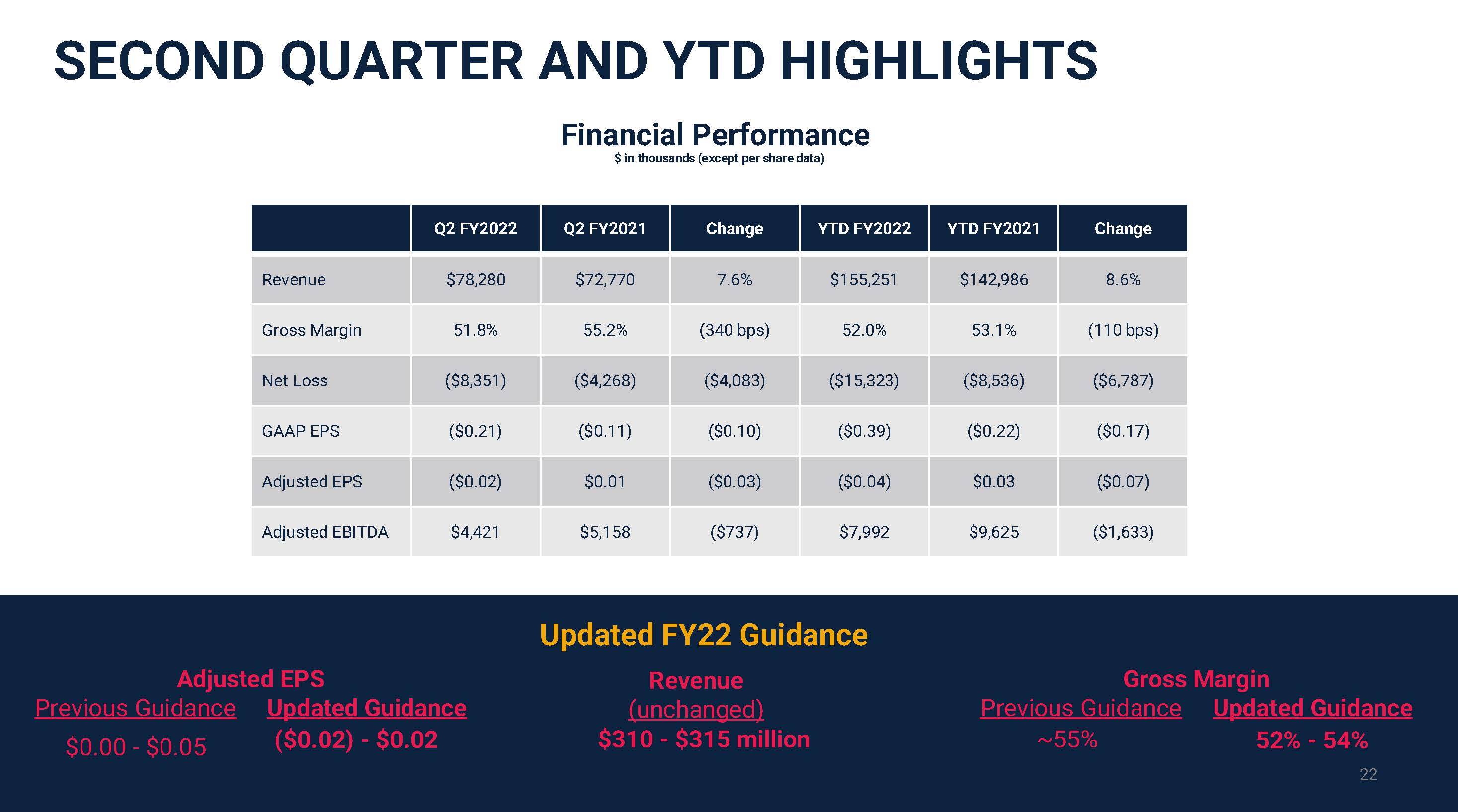
SECOND QUARTER AND YTD HIGHLIGHTS Q2 FY2022 Q2 FY2021 Change YTD FY2022 YTD FY2021 Change Revenue $78,280 $72,770 7.6% $155,251 $142,986 8.6% Gross Margin 51.8% 55.2% (340 bps) 52.0% 53.1% (110 bps) Net Loss ($8,351) ($4,268) ($4,083) ($15,323) ($8,536) ($6,787) GAAP EPS ($0.21) ($0.11) ($0.10) ($0.39) ($0.22) ($0.17) Adjusted EPS ($0.02) $0.01 ($0.03) ($0.04) $0.03 ($0.07) Adjusted EBITDA $4,421 $5,158 ($737) $7,992 $9,625 ($1,633) Financial Performance$ in thousands (except per share data) 22 22 Adjusted EPS Previous Guidance$0.00 - $0.05 Gross Margin Previous Guidance~55% Updated FY22 GuidanceRevenue(unchanged)$310 - $315 million Updated Guidance ($0.02) - $0.02 Updated Guidance 52% - 54%22
24% 76% SECOND QUARTER AND YTD HIGHLIGHTS 23 Med Device Med Tech 36.4% growth 0.8% growth Q2 Revenue Growth Q2 FY21 Q2 FY22 19% 81% YTD Revenue Contribution Med Device Med Tech YTD FY21 YTD FY22 17% 83% 24% 76% 50.0% growth 0.1% growth Q2 Revenue Contribution YTD Revenue Growth 24.3 36.5 118.7 118.7 YTD 21 YTD 22 13.8 18.9 58.9 59.4 Q2 FY21 Q2 FY22

SECOND QUARTER AND YTD FY2022 RESULTS (unaudited) $ in thousands (except per share data) Q2 FY2022 Q2 FY2021 Change YTD FY2022 YTD FY2021 Change Revenue $78,280 $72,770 7.6% $155,251 $142,986 8.6% Med TechMed Device $18,886$59,394 $13,849$58,921 36.4%0.8% $36,504$118,747 $24,335$118,651 50.0%0.1% Endovascular Therapies Vascular Access Oncology $39,660$25,070$13,550 $33,900$23,930$14,940 17.0%4.8%(9.3%) $77,718$50,026$27,507 $63,757$52,035$27,194 21.9%(3.9%)1.2% United States International $65,350$12,930 $60,684$12,086 7.7%7.0% $129,814$25,437 $114,792$28,194 13.1%(9.8%) Net LossNon-GAAP Adjusted Net Income (Loss) ($8,351)($856) ($4,268)$564 ($4,083)($1,420) ($15,323)($1,743) ($8,536)$1,181 ($6,787)($2,924) GAAP EPSNon-GAAP Adjusted EPS ($0.21)($0.02) ($0.11)$0.01 ($0.10)($0.03) ($0.39)($0.04) ($0.22)$0.03 ($0.17)($0.07) Gross Margin 51.8% 55.2% (340 bps) 52.0% 53.1% (110 bps) Adjusted EBITDA $4,421 $5,158 ($737) $7,992 $9,625 ($1,633) 24

25 PANDEMIC LANDSCAPETaking Actions to Address Short-term Supply Chain Disruption & Long-term Ability to Supply Customers HeadwindsTight Labor MarketDrives increasing labor costsImpacts absorptionRaw Materials Inflationary PressureIncreasing Freight Costs Operational FocusIncreasing and Qualifying Additional Manufacturing CapacityImproving EfficienciesSKU rationalizationRobust supply chain planningPricing and Shipping Adjustments

01 | 10 | 2022 ANGIODYNAMICS Needham Healthcare ConferenceJim Clemmer, President & CEO Stephen Trowbridge, EVP & CFO 26


APPENDIX 27
GAAP to Non-GAAP Reconciliation 28

Reconciliation of GAAP to Non-GAAPNet Income (Loss) and EPS Reconciliation of Net Loss toAdjusted EBITDA (1) (2) (3) Includes costs related to merger and acquisition activities, restructurings, and unusual items, including asset impairments and write- offs, certain litigation, and other items.Adjustment to reflect the income tax provision on a non-GAAP basis has been calculated assuming no valuation allowance on the Company's U.S. deferred tax assets and an effective tax rate of 23% for the periods ended November 30, 2021 and November 30, 2020.Diluted shares may differ for non-GAAP measures as compared to GAAP due to a GAAP loss. 29 (1) Includes costs related to merger and acquisition activities, restructurings, and unusual items, including asset impairments and write-offs,certain litigation, and other items. (in thousands, except per share data) (in thousands)
