Exhibit 99.2

ANGIODYNAMICS Third Quarter 2022 Earnings PresentationApril 7, 2022 1

2 Forward-Looking Statement Notice Regarding Forward-Looking StatementsThis presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements regarding AngioDynamics’ expected future financial position, results of operations, cash flows, business strategy, budgets, projected costs, capital expenditures, products, competitive positions, growth opportunities, plans and objectives of management for future operations, as well as statements that include the words such as “expects,” “reaffirms,” “intends,” “anticipates,” “plans,” “projects,” “believes,” “seeks,” “estimates,” “optimistic,” or variations of such words and similar expressions, are forward-looking statements. These forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ materially from AngioDynamics’ expectations, expressed or implied. Factors that may affect the actual results achieved by AngioDynamics include, without limitation, the scale and scope of the COVID-19 global pandemic, the ability of AngioDynamics to develop its existing and new products, technological advances and patents attained by competitors, infringement of AngioDynamics’ technology or assertions that AngioDynamics’ technology infringes the technology of third parties, the ability of AngioDynamics to effectively compete against competitors that have substantially greater resources, future actions by the FDA or other regulatory agencies, domestic and foreign health care reforms and government regulations, results of pending or future clinical trials, overall economic conditions (including inflation, labor shortages and supply chain challenges including the cost and availability of raw materials), the results of on-going litigation, challenges with respect to third-party distributors or joint venture partners or collaborators, the results of sales efforts, the effects of product recalls and product liability claims, changes in key personnel, the ability of AngioDynamics to execute on strategic initiatives, the effects of economic, credit and capital market conditions, general market conditions, market acceptance, foreign currency exchange rate fluctuations, the effects on pricing from group purchasing organizations and competition, the ability of AngioDynamics to obtain regulatory clearances or approval of its products, or to integrate acquired businesses, as well as the risk factors listed from time to time in AngioDynamics’ SEC filings, including but not limited to its Annual Report on Form 10-K for the year ended May 31, 2021. AngioDynamics does not assume any obligation to publicly update or revise any forward-looking statements for any reason.In the United States, the NanoKnife System has received a 510(k) clearance by the Food and Drug Administration for use in the surgical ablation of soft tissue, and is similarly approved for commercialization in Canada, the European Union and Australia. The NanoKnife System has not been cleared for the treatment or therapy of a specific disease or condition.Notice Regarding Non-GAAP Financial MeasuresManagement uses non-GAAP measures to establish operational goals and believes that non-GAAP measures may assist investors in analyzing the underlying trends in AngioDynamics’ business over time. Investors should consider these non-GAAP measures in addition to, not as a substitute for or as superior to, financial reporting measures prepared in accordance with GAAP. In this presentation, AngioDynamics has reported adjusted EBITDA (income before interest, taxes, depreciation and amortization and stock-based compensation); adjusted net income and adjusted earnings per share. Management uses these measures in its internal analysis and review of operational performance. Management believes that these measures provide investors with useful information in comparing AngioDynamics’ performance over different periods. By using these non-GAAP measures, management believes that investors get a better picture of the performance of AngioDynamics’ underlying business. Management encourages investors to review AngioDynamics’ financial results prepared in accordance with GAAP to understand AngioDynamics’ performance taking into account all relevant factors, including those that may only occur from time to time but have a material impact on AngioDynamics’ financial results. Please see the tables that follow for a reconciliation of non-GAAP measures to measures prepared in accordance with GAAP.

Corporate Developments – Q3 and YTD Highlights 3 Continued focused investment in our 3 key Med Tech platforms: Auryon, Thrombectomy & NanoKnife Revised FY2021 Guidance 3 FY22 Guidance Revenue(unchanged) Adjusted EPS(unchanged) Gross Margin(unchanged) $310 - $315 million COVID-19 related headwinds persist Gross margin impacted by supply chain disruptions, labor shortages and inflation Procedural volume pressures particularly in December and JanuaryBacklog of $9.6 million at quarter end Company Q3 revenue growth of 3.9%Med Tech up 28.6%; Med Device down 2.8%$7.3 million in Auryon sales 13.7% YOY growth in Mechanical Thrombectomy11.2% YOY growth in NanoKnife disposables ($0.02) - $0.02 52% - 54% Company YTD revenue growth of 7.0%Med Tech up 41.8%; Med Device down 0.8% (excluding NHS, up 2.2%)$19.5 million in Auryon sales 18.3% YOY growth in Mechanical Thrombectomy16.9% YOY growth in NanoKnife disposables Subsequent to quarter end: The Company received FDA approval for its AlphaVac F18 thrombectomy system;The Company received FDA approval for its IDE study for the use of AlphaVac F18 to treat pulmonary embolism; and Enrolled first patients in the PRESERVE study for NanoKnife in prostate Manufacturing capacity enhancement initiatives drove a 20% increase in production hours exiting Q3
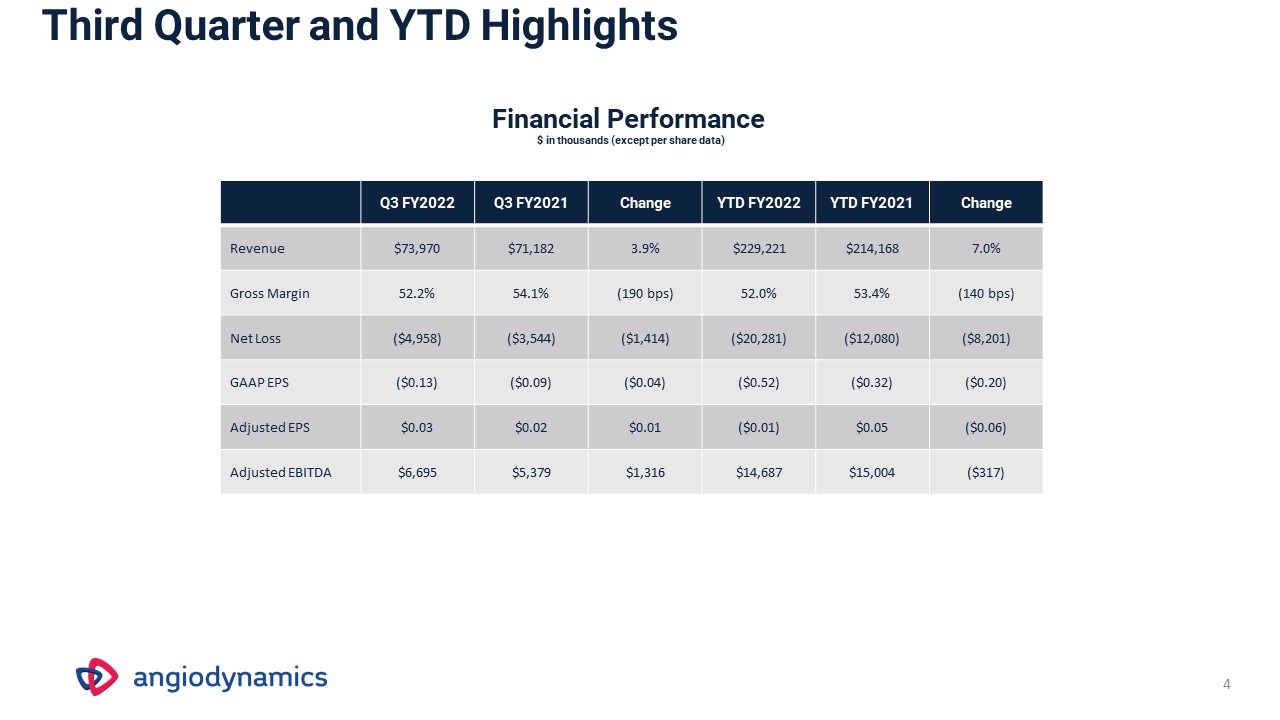
Third Quarter and YTD Highlights Q3 FY2022 Q3 FY2021 Change YTD FY2022 YTD FY2021 Change Revenue $73,970 $71,182 3.9% $229,221 $214,168 7.0% Gross Margin 52.2% 54.1% (190 bps) 52.0% 53.4% (140 bps) Net Loss ($4,958) ($3,544) ($1,414) ($20,281) ($12,080) ($8,201) GAAP EPS ($0.13) ($0.09) ($0.04) ($0.52) ($0.32) ($0.20) Adjusted EPS $0.03 $0.02 $0.01 ($0.01) $0.05 ($0.06) Adjusted EBITDA $6,695 $5,379 $1,316 $14,687 $15,004 ($317) Financial Performance$ in thousands (except per share data) 4
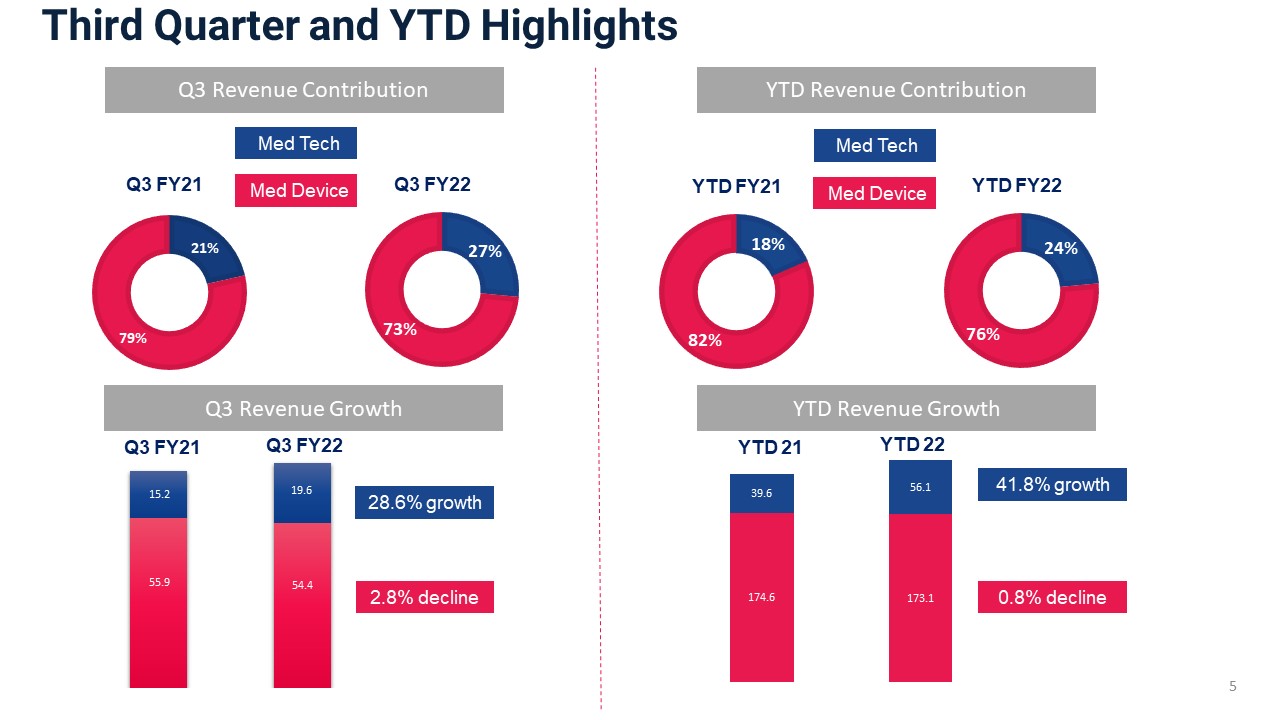
Third Quarter and YTD Highlights 5 Med Device Med Tech $10.5 $17.6 $59.7 $59.4 28.6% growth 2.8% decline Q3 Revenue Growth Q3 FY21 Q3 FY22 YTD Revenue Contribution Med Device Med Tech YTD FY21 YTD FY22 41.8% growth 0.8% decline Q3 Revenue Contribution YTD Revenue Growth 15.2 19.6 54.4
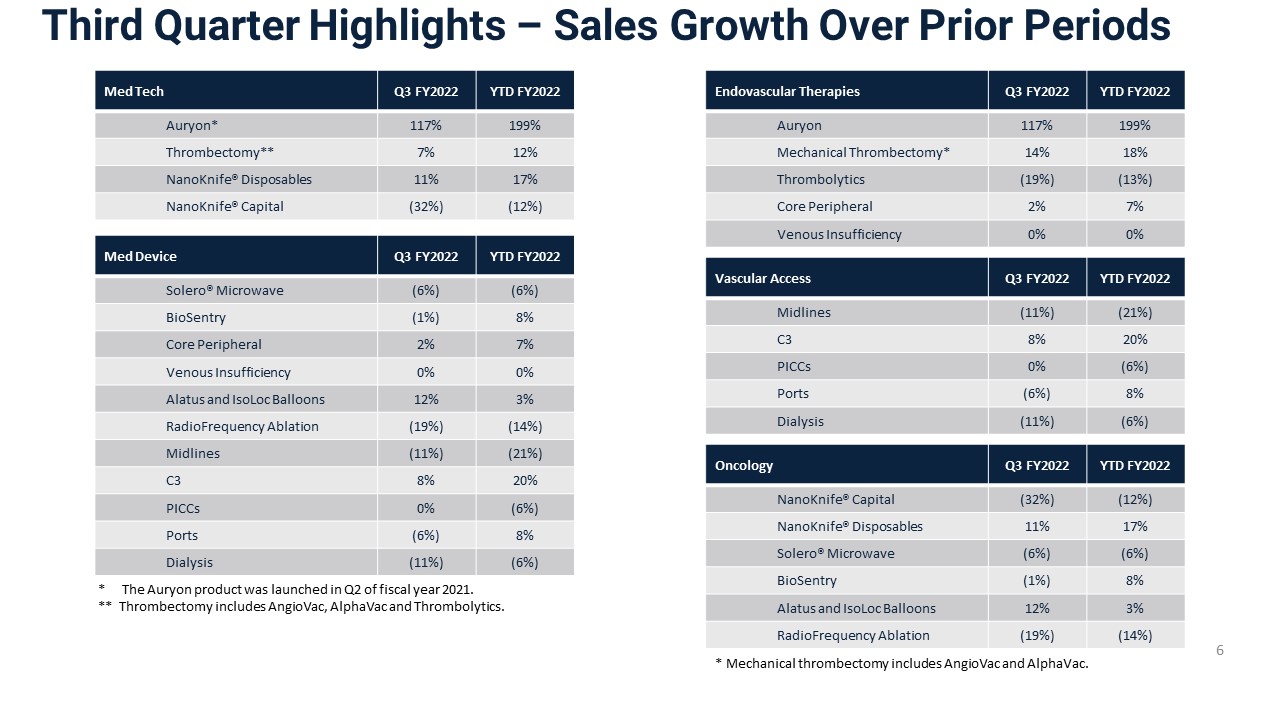
Third Quarter Highlights – Sales Growth Over Prior Periods Endovascular Therapies Q3 FY2022 YTD FY2022 Auryon 117% 199% Mechanical Thrombectomy* 14% 18% Thrombolytics (19%) (13%) Core Peripheral 2% 7% Venous Insufficiency 0% 0% Vascular Access Q3 FY2022 YTD FY2022 Midlines (11%) (21%) C3 8% 20% PICCs 0% (6%) Ports (6%) 8% Dialysis (11%) (6%) Oncology Q3 FY2022 YTD FY2022 NanoKnife® Capital (32%) (12%) NanoKnife® Disposables 11% 17% Solero® Microwave (6%) (6%) BioSentry (1%) 8% Alatus and IsoLoc Balloons 12% 3% RadioFrequency Ablation (19%) (14%) 6 Med Tech Q3 FY2022 YTD FY2022 Auryon* 117% 199% Thrombectomy** 7% 12% NanoKnife® Disposables 11% 17% NanoKnife® Capital (32%) (12%) Med Device Q3 FY2022 YTD FY2022 Solero® Microwave (6%) (6%) BioSentry (1%) 8% Core Peripheral 2% 7% Venous Insufficiency 0% 0% Alatus and IsoLoc Balloons 12% 3% RadioFrequency Ablation (19%) (14%) Midlines (11%) (21%) C3 8% 20% PICCs 0% (6%) Ports (6%) 8% Dialysis (11%) (6%) * The Auryon product was launched in Q2 of fiscal year 2021. ** Thrombectomy includes AngioVac, AlphaVac and Thrombolytics. * Mechanical thrombectomy includes AngioVac and AlphaVac.

Third Quarter and YTD FY2022 Results (unaudited) $ in thousands (except per share data) Q3 FY2022 Q3 FY2021 Change YTD FY2022 YTD FY2021 Change Revenue $73,970 $71,182 3.9% $229,221 $214,168 7.0% Med Tech Med Device $19,612$54,358 $15,246$55,936 28.6%(2.8%) $56,117$173,104 $39,581$174,587 41.8%(0.8%) Endovascular Therapies Vascular Access Oncology $38,083$23,431$12,456 $33,251$24,813$13,118 14.5%(5.6%)(5.0%) $115,799$73,459$39,963 $97,008$76,848$40,312 19.4%(4.4%)(0.9%) United States International $62,445$11,525 $58,654$12,528 6.5%(8.0%) $192,259$36,962 $173,446$40,722 10.8%(9.2%) Net LossNon-GAAP Adjusted Net Income (Loss) ($4,958)$1,307 ($3,544)$738 ($1,414)$569 ($20,281)($436) ($12,080)$1,919 ($8,201)($2,355) GAAP EPSNon-GAAP Adjusted EPS ($0.13)$0.03 ($0.09)$0.02 ($0.04)$0.01 ($0.52)($0.01) ($0.32)$0.05 ($0.20)($0.06) Gross Margin 52.2% 54.1% (190 bps) 52.0% 53.4% (140 bps) Adjusted EBITDA $6,695 $5,379 $1,316 $14,687 $15,004 ($317) $ in thousands Q3 FY2022 Q4 FY2021 Change Cash $23,890 $48,161 ($24,271) Debt $25,000 $20,000 $5,000 Net (Debt) Cash ($1,110) $28,161 ($29,271) 7
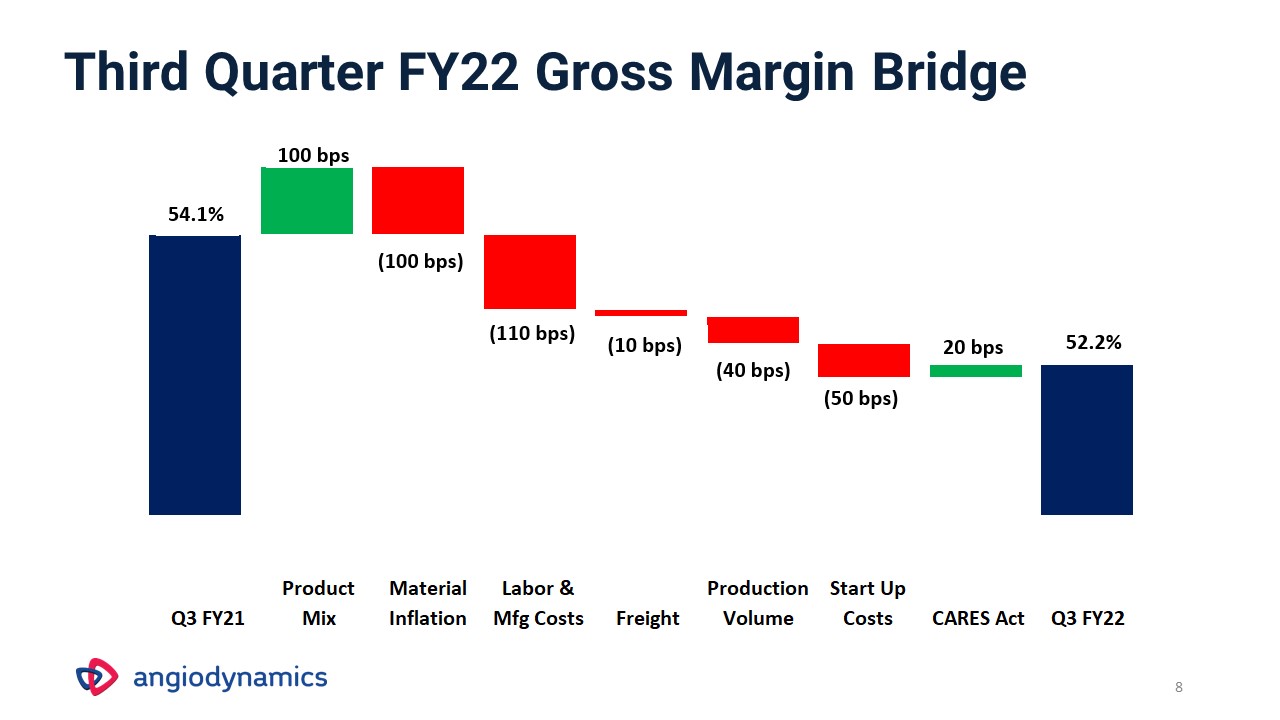
8 Third Quarter FY22 Gross Margin Bridge

GAAP to Non-GAAP Reconciliation 9

Reconciliation of GAAP to Non-GAAP Net Income (Loss) and EPS Reconciliation of Net Loss to Adjusted EBITDA Includes costs related to merger and acquisition activities, restructurings, and unusual items, including asset impairments and write-offs, certain litigation, and other items. Adjustment to reflect the income tax provision on a non-GAAP basis has been calculated assuming no valuation allowance on the Company's U.S. deferred tax assets and an effective tax rate of 23% for the periods ended February 28, 2022 and February 28, 2021.Diluted shares may differ for non-GAAP measures as compared to GAAP due to a GAAP loss. 10 Includes costs related to merger and acquisition activities, restructurings, and unusual items, including asset impairments and write-offs, certain litigation, and other items. (in thousands, except per share data) (in thousands)