Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
RCKT similar filings
- 1 Dec 22 Rocket Pharmaceuticals Completes Acquisition of Renovacor
- 3 Nov 22 Results of Operations and Financial Condition
- 4 Oct 22 Entry into a Material Definitive Agreement
- 3 Oct 22 Other Events
- 20 Sep 22 Entry into a Material Definitive Agreement
- 9 Sep 22 Other Events
- 8 Aug 22 Rocket Pharmaceuticals Reports Second Quarter 2022 Financial Results and Highlights Recent Progress
Filing view
External links
Exhibit 99.1

Update from Phase 1 Trial of RP-A501 Gene Therapy Treatment for Danon DiseaseSeptember 30, 2022

DISCLAIMER Various statements in this release concerning Rocket’s future expectations, plans and prospects, including without limitation, Rocket’s expectations regarding its guidance for 2022, the safety and effectiveness of RP-A501 for the potential treatment of Danon Disease, trends for RP-A501 safety and efficacy based on the adult patients treated to date, the expected timing and outcome of Rocket’s regulatory interactions and planned submissions, including in connection with the potential advancement toward a Phase 2 pivotal study for RP-A501, Rocket’s plans for the advancement of its Danon Disease program and the safety, effectiveness and timing of related pre-clinical studies and clinical trials, may constitute forward-looking statements for the purposes of the safe harbor provisions under the Private Securities Litigation Reform Act of 1995 and other federal securities laws and are subject to substantial risks, uncertainties and assumptions. You should not place reliance on these forward-looking statements, which often include words such as "believe," "expect," "anticipate," "intend," "plan," "will give," "estimate," "seek," "will," "may," "suggest" or similar terms, variations of such terms or the negative of those terms. Although Rocket believes that the expectations reflected in the forward-looking statements are reasonable, Rocket cannot guarantee such outcomes. Actual results may differ materially from those indicated by these forward-looking statements as a result of various important factors, including, without limitation, Rocket’s ability to monitor the impact of COVID-19 on its business operations and take steps to ensure the safety of patients, families and employees, the interest from patients and families for participation in each of Rocket’s ongoing trials, our expectations regarding the delays and impact of COVID-19 on clinical sites, patient enrollment, trial timelines and data readouts, our expectations regarding our drug supply for our ongoing and anticipated trials, actions of regulatory agencies, which may affect the initiation, timing and progress of pre-clinical studies and clinical trials of its product candidates, Rocket’s dependence on third parties for development, manufacture, marketing, sales and distribution of product candidates, the outcome of litigation, and unexpected expenditures, as well as those risks more fully discussed in the section entitled "Risk Factors" in Rocket’s Annual Report on Form 10-K for the year ended December 31, 2021, filed February 28, 2022 with the SEC and subsequent filings with the SEC including our Quarterly Reports on Form 10-Q. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and Rocket undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.

Danon Disease (DD): Serious Condition with Unmet Medical Need Standard of care: Heart transplant (HTx) Limitations: Considerable morbidity and mortality Only ~20% of patients receive HTx Not curative of extracardiac disease Therapeutic Challenges X-linked, dominant, monogenic disease Loss-of-function mutations in LAMP-2B Disease Etiology Impaired autophagy Prominent autophagic vacuoles Myocardial disarray Clinical Manifestations Addressable Market – US and EU Prevalence of 15,000 to 30,000 individuals Annual incidence of 800 to 1,200 individuals Severe cardiomyopathy Mortality secondary to heart failure or arrhythmia Males: Aggressive disease course, median overall survival: 19 years Females: Delayed median presentation (~20 years later) due to additional X chromosome, highly morbid and fatal disorder Other clinical manifestations Skeletal myopathy CNS manifestations Ophthalmologic manifestations CNS, central nervous system; LAMP-2B, lysosome-associated membrane protein 2B. Boucek D et al. Genet Med. 2011;13(6):563-568. Brambatti M et al. Int J Cardiol. 2019;286:92-98. RP-A501: Danon Disease
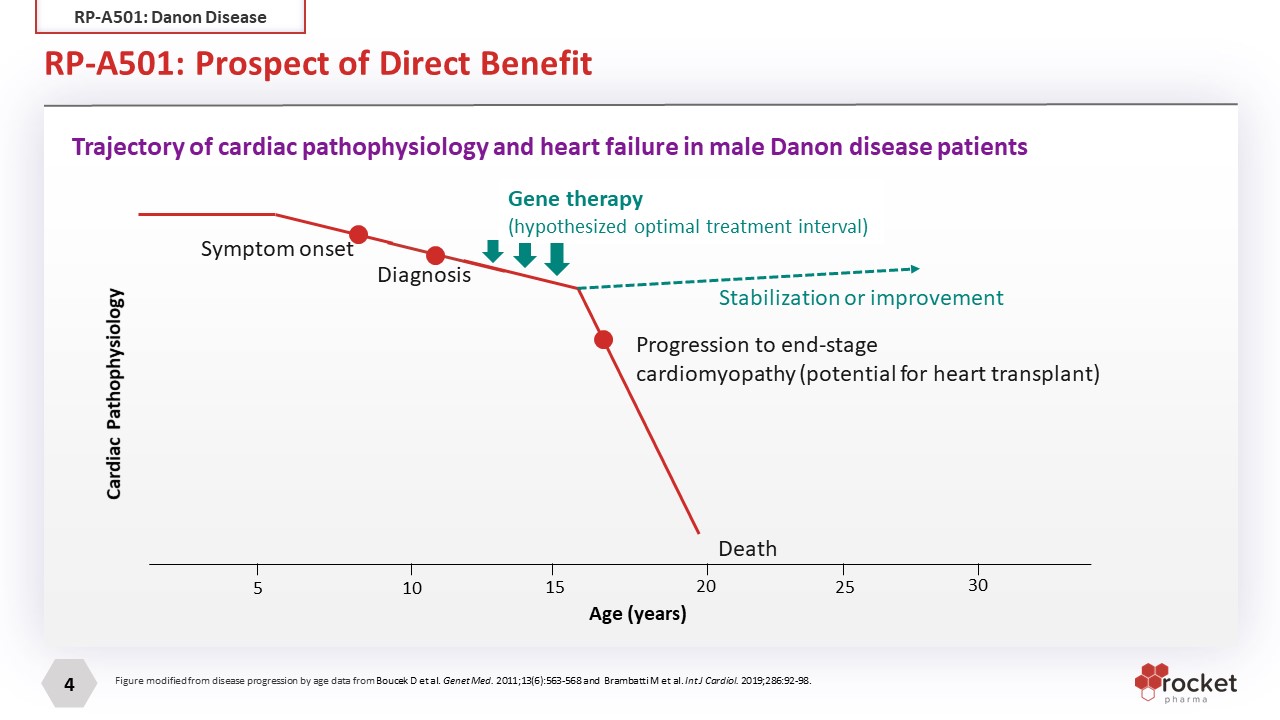
RP-A501: Prospect of Direct Benefit Age (years) 5 10 15 20 25 30 Cardiac Pathophysiology Symptom onset Diagnosis Progression to end-stagecardiomyopathy (potential for heart transplant) Death Gene therapy(hypothesized optimal treatment interval) Stabilization or improvement Trajectory of cardiac pathophysiology and heart failure in male Danon disease patients Figure modified from disease progression by age data from Boucek D et al. Genet Med. 2011;13(6):563-568 and Brambatti M et al. Int J Cardiol. 2019;286:92-98. RP-A501: Danon Disease
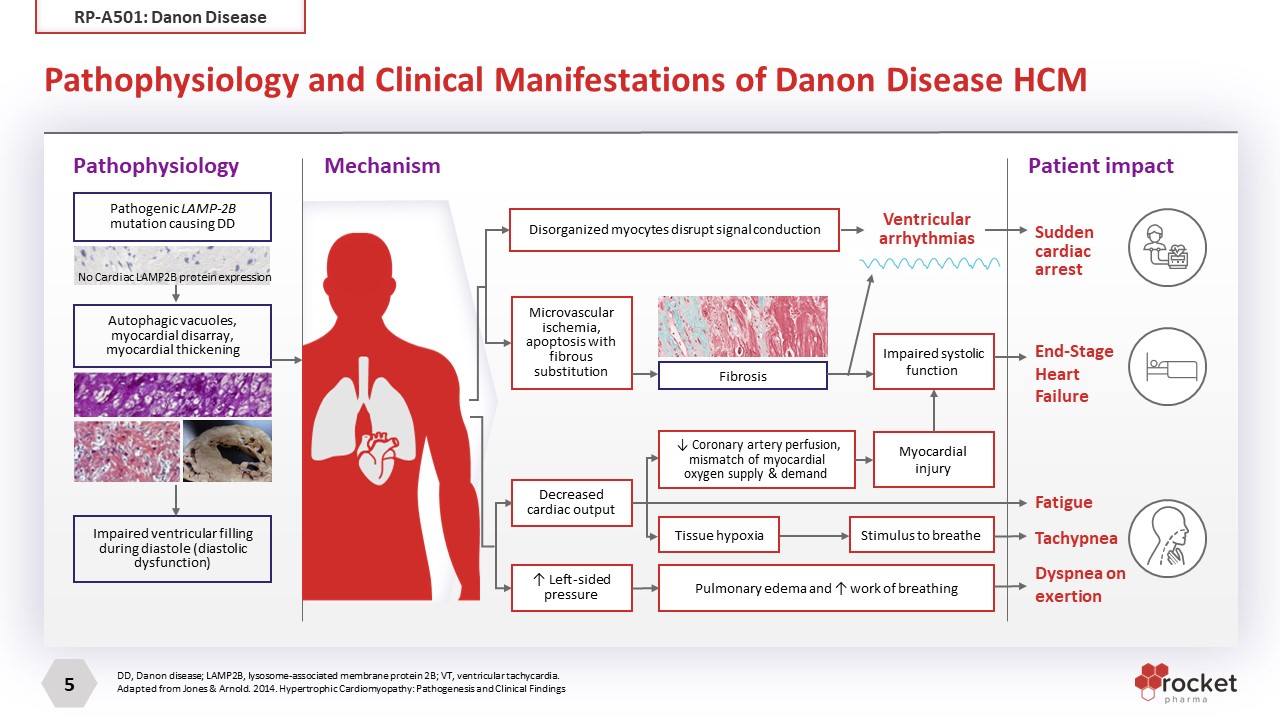
Pathophysiology and Clinical Manifestations of Danon Disease HCM DD, Danon disease; LAMP2B, lysosome-associated membrane protein 2B; VT, ventricular tachycardia. Adapted from Jones & Arnold. 2014. Hypertrophic Cardiomyopathy: Pathogenesis and Clinical Findings Pathophysiology Mechanism Patient impact Pathogenic LAMP-2B mutation causing DD Autophagic vacuoles, myocardial disarray, myocardial thickening Impaired ventricular filling during diastole (diastolic dysfunction) No Cardiac LAMP2B protein expression Disorganized myocytes disrupt signal conduction Microvascular ischemia, apoptosis with fibrous substitution Ventricular arrhythmias Decreased cardiac output ↑ Left-sided pressure ↓ Coronary artery perfusion, mismatch of myocardial oxygen supply & demand Tissue hypoxia Stimulus to breathe Myocardial injury Fibrosis Impaired systolic function Pulmonary edema and ↑ work of breathing Sudden cardiac arrest End-Stage Heart Failure Tachypnea Fatigue Dyspnea on exertion RP-A501: Danon Disease
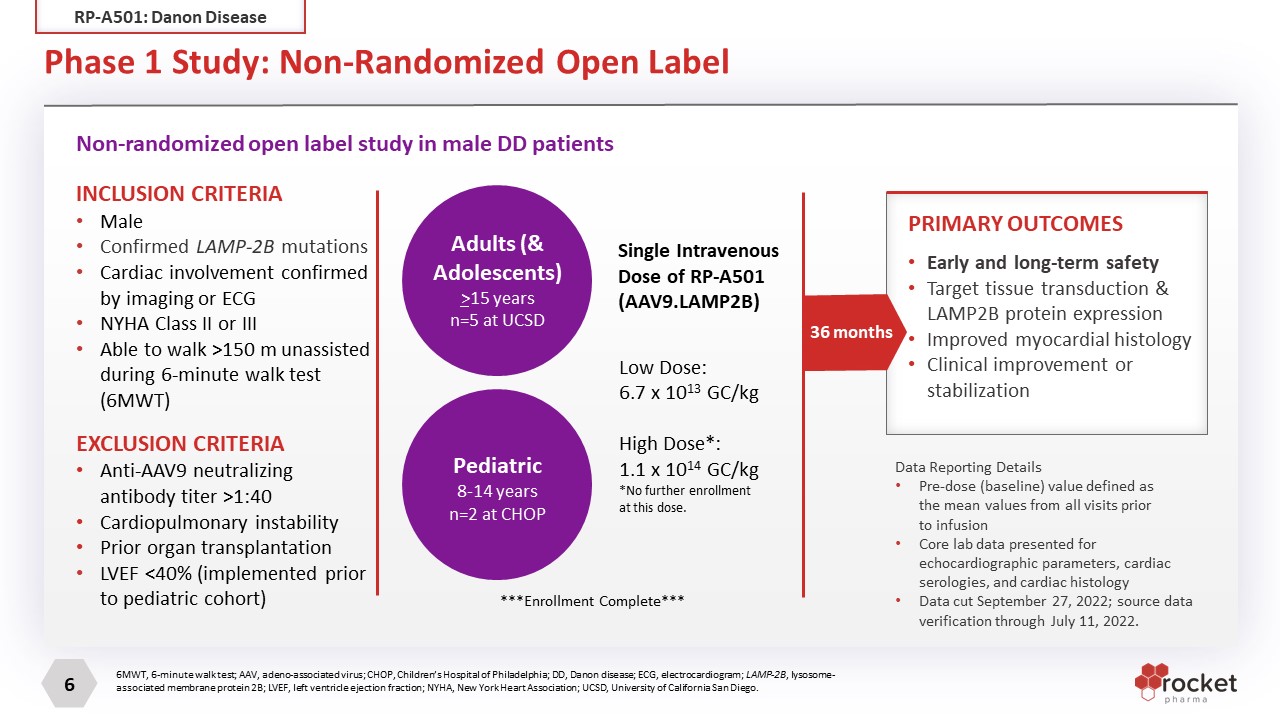
Phase 1 Study: Non-Randomized Open Label Non-randomized open label study in male DD patients INCLUSION CRITERIA Male Confirmed LAMP-2B mutations Cardiac involvement confirmed by imaging or ECG NYHA Class II or III Able to walk >150 m unassisted during 6-minute walk test (6MWT) EXCLUSION CRITERIA Anti-AAV9 neutralizing antibody titer >1:40 Cardiopulmonary instability Prior organ transplantation LVEF <40% (implemented prior to pediatric cohort) Pediatric 8-14 years n=2 at CHOP Adults (& Adolescents) >15 years n=5 at UCSD Single Intravenous Dose of RP-A501 (AAV9.LAMP2B) Low Dose: 6.7 x 1013 GC/kg High Dose*: 1.1 x 1014 GC/kg *No further enrollment at this dose. Early and long-term safety Target tissue transduction & LAMP2B protein expression Improved myocardial histology Clinical improvement or stabilization PRIMARY OUTCOMES 36 months ***Enrollment Complete*** Data Reporting Details Pre-dose (baseline) value defined as the mean values from all visits prior to infusion Core lab data presented for echocardiographic parameters, cardiac serologies, and cardiac histology Data cut September 27, 2022; source data verification through July 11, 2022. 6MWT, 6-minute walk test; AAV, adeno-associated virus; CHOP, Children’s Hospital of Philadelphia; DD, Danon disease; ECG, electrocardiogram; LAMP-2B, lysosome-associated membrane protein 2B; LVEF, left ventricle ejection fraction; NYHA, New York Heart Association; UCSD, University of California San Diego. RP-A501: Danon Disease
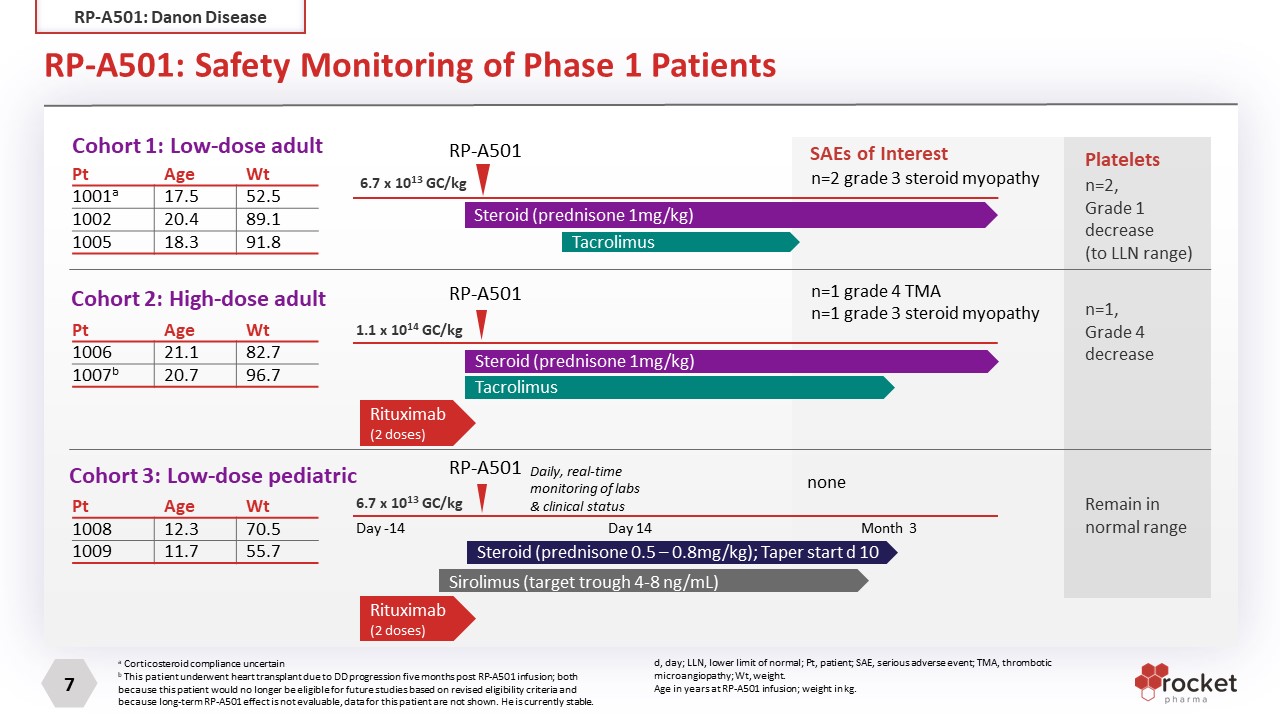
RP-A501: Safety Monitoring of Phase 1 Patients Pt Age Wt 1001a 17.5 52.5 1002 20.4 89.1 1005 18.3 91.8 Cohort 1: Low-dose adult Cohort 2: High-dose adult Cohort 3: Low-dose pediatric 1.1 x 1014 GC/kg 6.7 x 1013 GC/kg 6.7 x 1013 GC/kg RP-A501 Steroid (prednisone 1mg/kg) Tacrolimus Tacrolimus Rituximab (2 doses) Steroid (prednisone 0.5 – 0.8mg/kg); Taper start d 10 Sirolimus (target trough 4-8 ng/mL) RP-A501 RP-A501 n=2 grade 3 steroid myopathy n=1 grade 3 steroid myopathy n=1 grade 4 TMA SAEs of Interest none Platelets n=2, Grade 1 decrease(to LLN range) n=1, Grade 4 decrease Remain in normal range Steroid (prednisone 1mg/kg) Pt Age Wt 1006 21.1 82.7 1007b 20.7 96.7 Pt Age Wt 1008 12.3 70.5 1009 11.7 55.7 a Corticosteroid compliance uncertain b This patient underwent heart transplant due to DD progression five months post RP-A501 infusion; both because this patient would no longer be eligible for future studies based on revised eligibility criteria and because long-term RP-A501 effect is not evaluable, data for this patient are not shown. He is currently stable. Rituximab (2 doses) Daily, real-time monitoring of labs & clinical status Day -14 Month 3 Day 14 d, day; LLN, lower limit of normal; Pt, patient; SAE, serious adverse event; TMA, thrombotic microangiopathy; Wt, weight. Age in years at RP-A501 infusion; weight in kg. RP-A501: Danon Disease
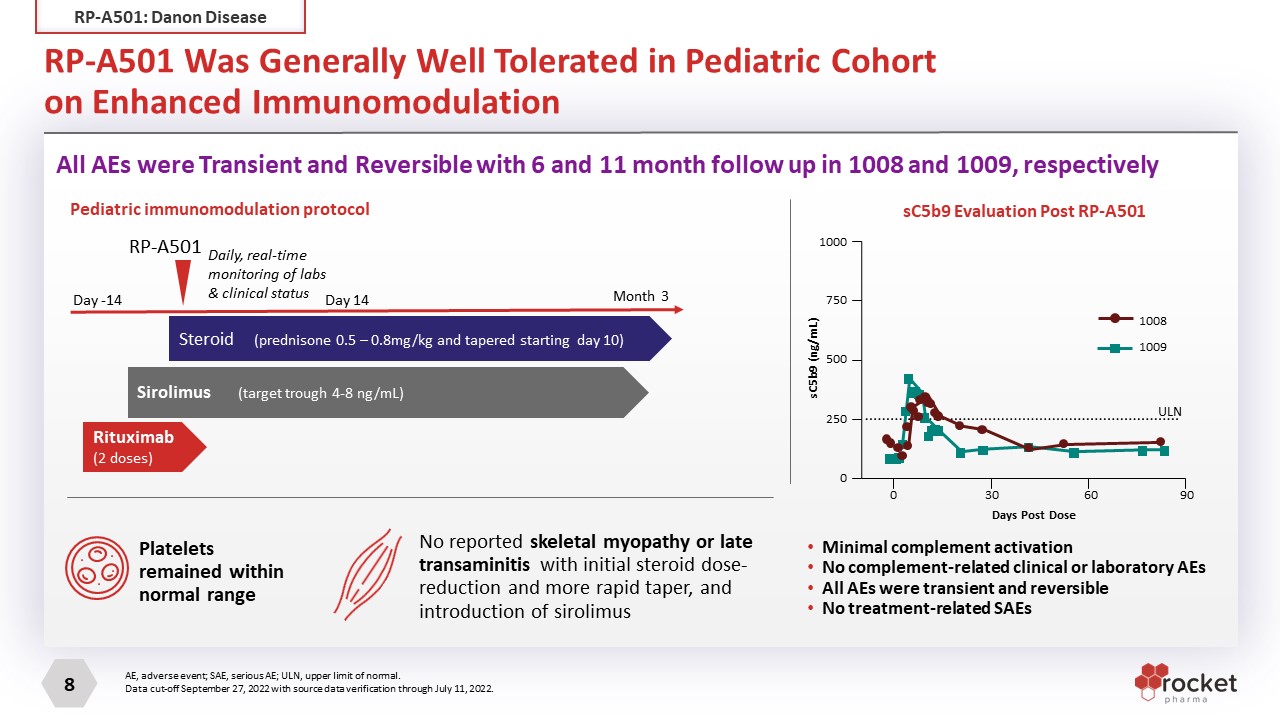
RP-A501 Was Generally Well Tolerated in Pediatric Cohorton Enhanced Immunomodulation Minimal complement activation No complement-related clinical or laboratory AEs All AEs were transient and reversible No treatment-related SAEs Steroid (prednisone 0.5 – 0.8mg/kg and tapered starting day 10) Sirolimus (target trough 4-8 ng/mL) Rituximab (2 doses) RP-A501 Pediatric immunomodulation protocol Daily, real-time monitoring of labs & clinical status Day -14 Month 3 Day 14 Platelets remained within normal range No reported skeletal myopathy or late transaminitis with initial steroid dose-reduction and more rapid taper, and introduction of sirolimus sC5b9 Evaluation Post RP-A501 AE, adverse event; SAE, serious AE; ULN, upper limit of normal. Data cut-off September 27, 2022 with source data verification through July 11, 2022. All AEs were Transient and Reversible with 6 and 11 month follow up in 1008 and 1009, respectively 1008 1009 1000 750 500 250 0 0 30 60 90 ULN sC5b9 (ng/mL) Days Post Dose RP-A501: Danon Disease
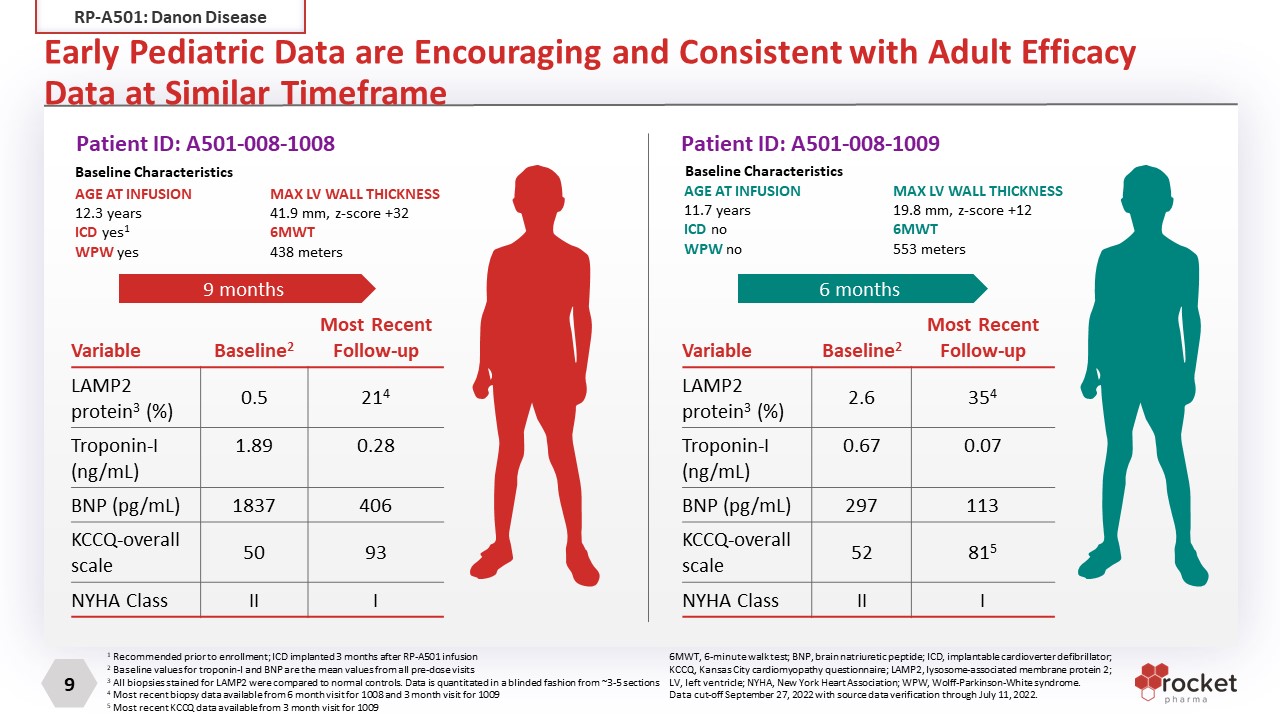
Early Pediatric Data are Encouraging and Consistent with Adult Efficacy Data at Similar Timeframe Variable Baseline2 Most Recent Follow-up LAMP2 protein3 (%) 2.6 354 Troponin-I (ng/mL) 0.67 0.07 BNP (pg/mL) 297 113 KCCQ-overall scale 52 815 NYHA Class II I 6 months AGE AT INFUSION 11.7 years ICD no WPW no Patient ID: A501-008-1008 Patient ID: A501-008-1009 AGE AT INFUSION 12.3 years ICD yes1 WPW yes Variable Baseline2 Most RecentFollow-up LAMP2 protein3 (%) 0.5 214 Troponin-I (ng/mL) 1.89 0.28 BNP (pg/mL) 1837 406 KCCQ-overall scale 50 93 NYHA Class II I 9 months MAX LV WALL THICKNESS 41.9 mm, z-score +32 6MWT 438 meters MAX LV WALL THICKNESS 19.8 mm, z-score +12 6MWT 553 meters Baseline Characteristics Baseline Characteristics 6MWT, 6-minute walk test; BNP, brain natriuretic peptide; ICD, implantable cardioverter defibrillator; KCCQ, Kansas City cardiomyopathy questionnaire; LAMP2, lysosome-associated membrane protein 2; LV, left ventricle; NYHA, New York Heart Association; WPW, Wolff-Parkinson-White syndrome. Data cut-off September 27, 2022 with source data verification through July 11, 2022. 1 Recommended prior to enrollment; ICD implanted 3 months after RP-A501 infusion 2 Baseline values for troponin-I and BNP are the mean values from all pre-dose visits 3 All biopsies stained for LAMP2 were compared to normal controls. Data is quantitated in a blinded fashion from ~3-5 sections 4 Most recent biopsy data available from 6 month visit for 1008 and 3 month visit for 1009 5 Most recent KCCQ data available from 3 month visit for 1009 RP-A501: Danon Disease
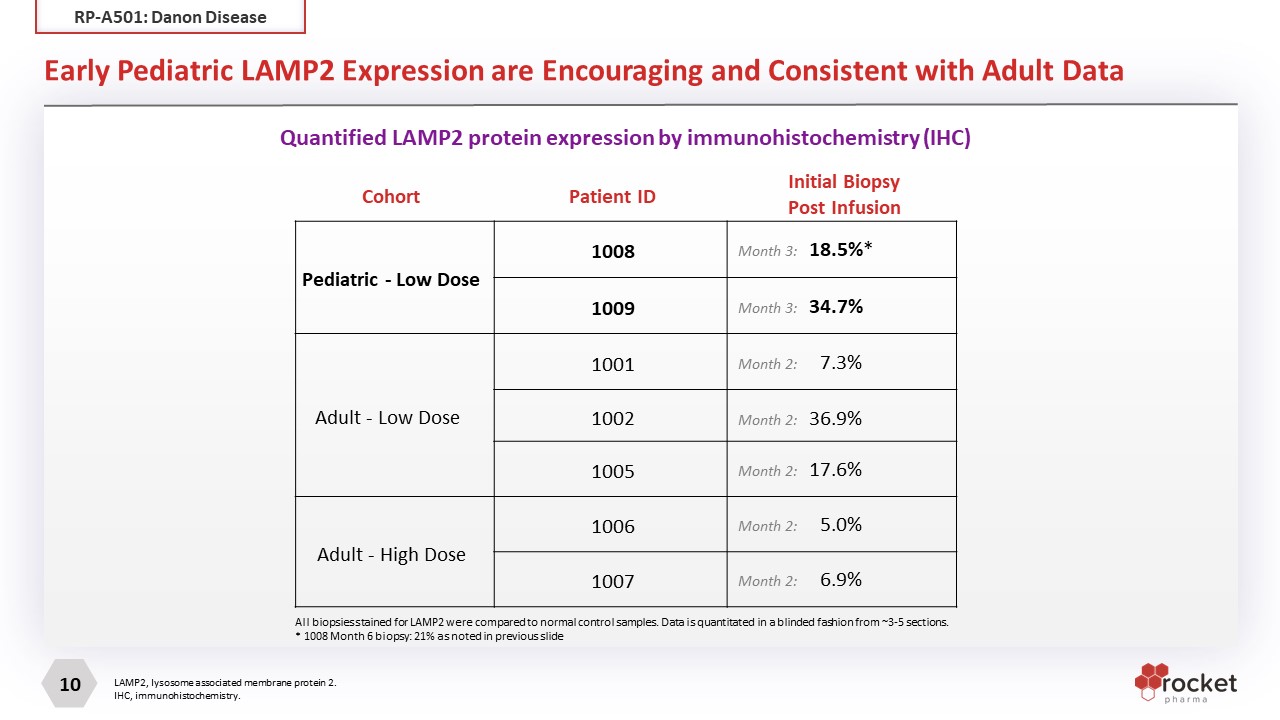
Early Pediatric LAMP2 Expression are Encouraging and Consistent with Adult Data LAMP2, lysosome associated membrane protein 2. IHC, immunohistochemistry. Cohort Patient ID Initial Biopsy Post Infusion Pediatric - Low Dose 1008 Month 3: 18.5%* 1009 Month 3: 34.7% Adult - Low Dose 1001 Month 2: 7.3% 1002 Month 2: 36.9% 1005 Month 2: 17.6% Adult - High Dose 1006 Month 2: 5.0% 1007 Month 2: 6.9% All biopsies stained for LAMP2 were compared to normal control samples. Data is quantitated in a blinded fashion from ~3-5 sections. * 1008 Month 6 biopsy: 21% as noted in previous slide Adapted from Oldfors 2015. A: IHC of an explanted heart of a younger female Danon disease patient with extremely patchy, nonhomogenous LAMP2 staining. C: IHC of an explanted heart of an older female Danon disease patient with more homogenous LAMP2 staining Quantified LAMP2 protein expression by immunohistochemistry (IHC) RP-A501: Danon Disease
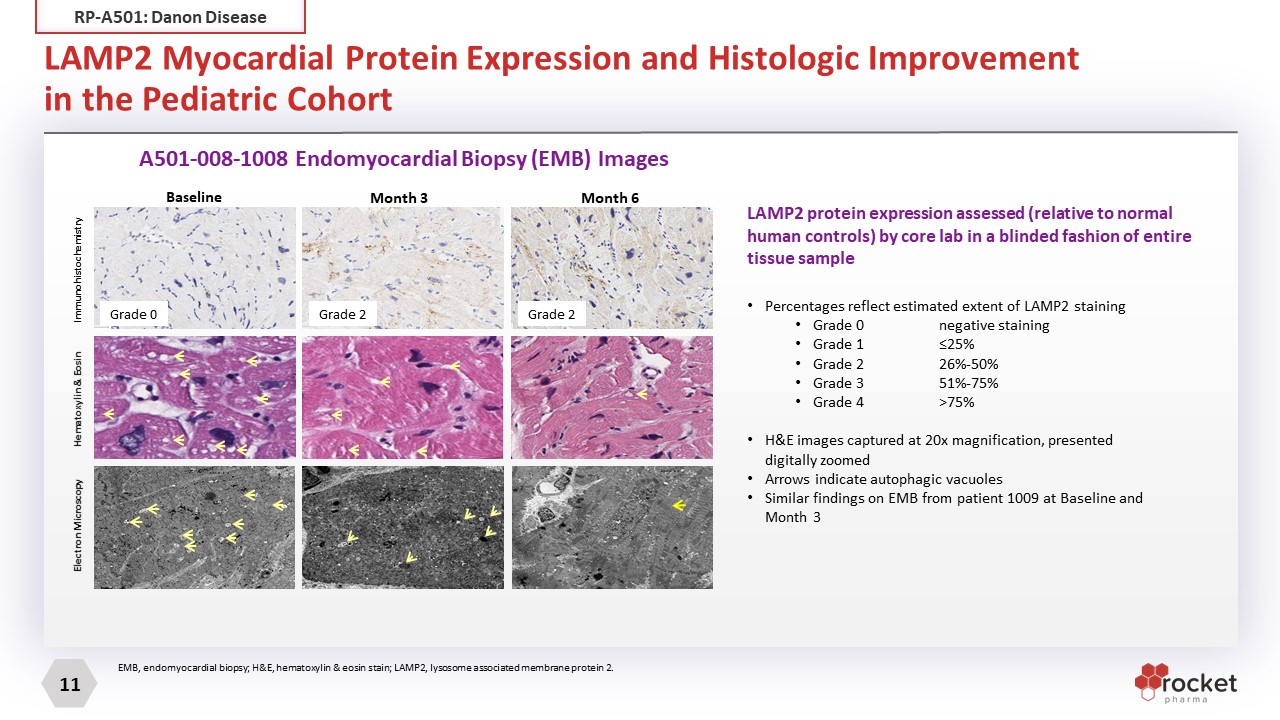
LAMP2 Myocardial Protein Expression and Histologic Improvement in the Pediatric Cohort EMB, endomyocardial biopsy; H&E, hematoxylin & eosin stain; LAMP2, lysosome associated membrane protein 2. A501-008-1008 Endomyocardial Biopsy (EMB) Images LAMP2 protein expression assessed (relative to normal human controls) by core lab in a blinded fashion of entire tissue sample Electron Microscopy Immunohistochemistry Baseline Month 3 Month 6 Hematoxylin & Eosin Grade 0 Grade 2 Grade 2 Percentages reflect estimated extent of LAMP2 staining Grade 0 negative staining Grade 1 ≤25% Grade 2 26%-50% Grade 3 51%-75% Grade 4 >75% H&E images captured at 20x magnification, presented digitally zoomed Arrows indicate autophagic vacuoles Similar findings on EMB from patient 1009 at Baseline and Month 3 RP-A501: Danon Disease
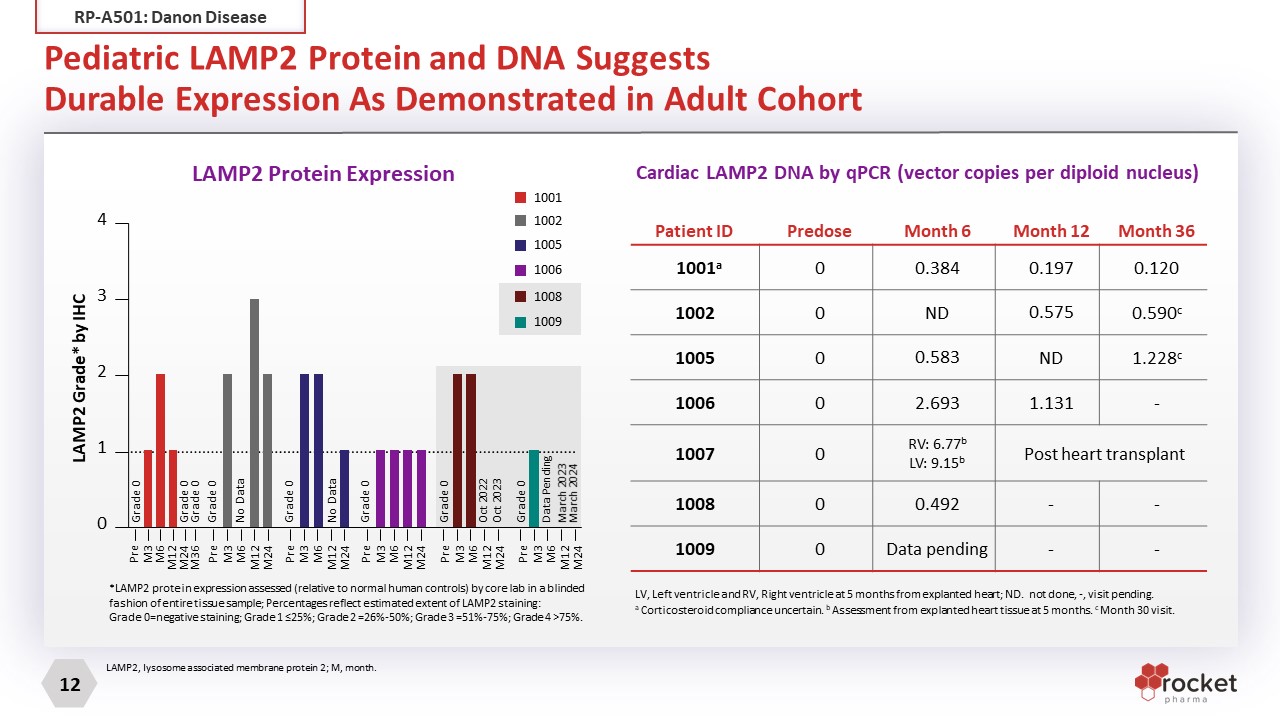
Pediatric LAMP2 Protein and DNA Suggests Durable Expression As Demonstrated in Adult Cohort *LAMP2 protein expression assessed (relative to normal human controls) by core lab in a blinded fashion of entire tissue sample; Percentages reflect estimated extent of LAMP2 staining: Grade 0=negative staining; Grade 1 ≤25%; Grade 2 =26%-50%; Grade 3 =51%-75%; Grade 4 >75%. 1001 1002 1005 1006 1008 1009 4 3 2 1 0 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 M36 Grade 0 Grade 0 Grade 0 Grade 0 No Data Grade 0 No Data Grade 0 Grade 0 Oct 2022 Oct 2023 Grade 0 Data Pending March 2023 March 2024 LAMP2 Grade* by IHC Patient ID Predose Month 6 Month 12 Month 36 1001a 0 0.384 0.197 0.120 1002 0 ND 0.575 0.590c 1005 0 0.583 ND 1.228c 1006 0 2.693 1.131 - 1007 0 RV: 6.77b LV: 9.15b Post heart transplant 1008 0 0.492 - - 1009 0 Data pending - - LAMP2, lysosome associated membrane protein 2; M, month. LAMP2 Protein Expression Cardiac LAMP2 DNA by qPCR (vector copies per diploid nucleus) LV, Left ventricle and RV, Right ventricle at 5 months from explanted heart; ND. not done, -, visit pending. a Corticosteroid compliance uncertain. b Assessment from explanted heart tissue at 5 months. c Month 30 visit. RP-A501: Danon Disease
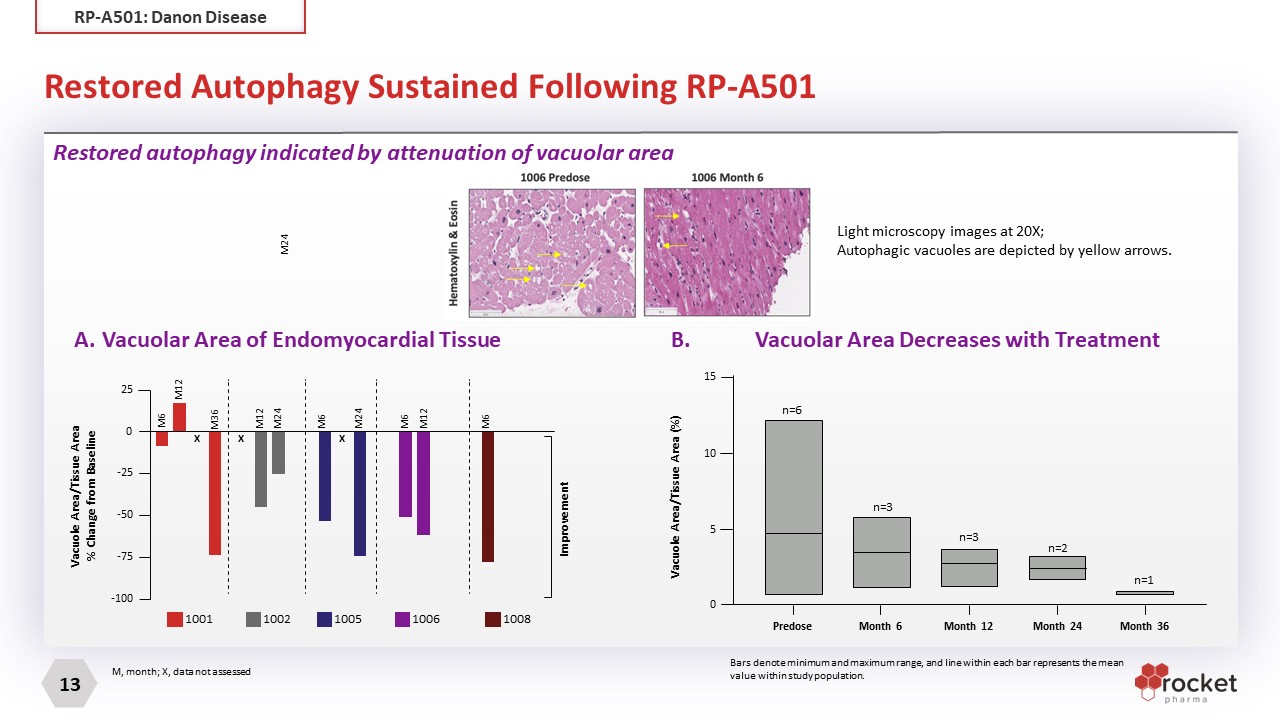
n=1 15 n=6 n=3 n=3 n=2 10 5 0 Predose Month 6 Vacuole Area/Tissue Area (%) Month 12 Month 24 Month 36 Restored Autophagy Sustained Following RP-A501 M, month; X, data not assessed B. 1001 1002 1005 1006 1008 M12 M24 M36 M6 M12 M24 M24 M6 M12 M6 M6 25 0 -25 -50 -75 -100 Improvement Vacuole Area/Tissue Area % Change from Baseline X X X A. Vacuolar Area of Endomyocardial Tissue Restored autophagy indicated by attenuation of vacuolar area B. Vacuolar Area Decreases with Treatment Bars denote minimum and maximum range, and line within each bar represents the mean value within study population. Light microscopy images at 20X; Autophagic vacuoles are depicted by yellow arrows. RP-A501: Danon Disease
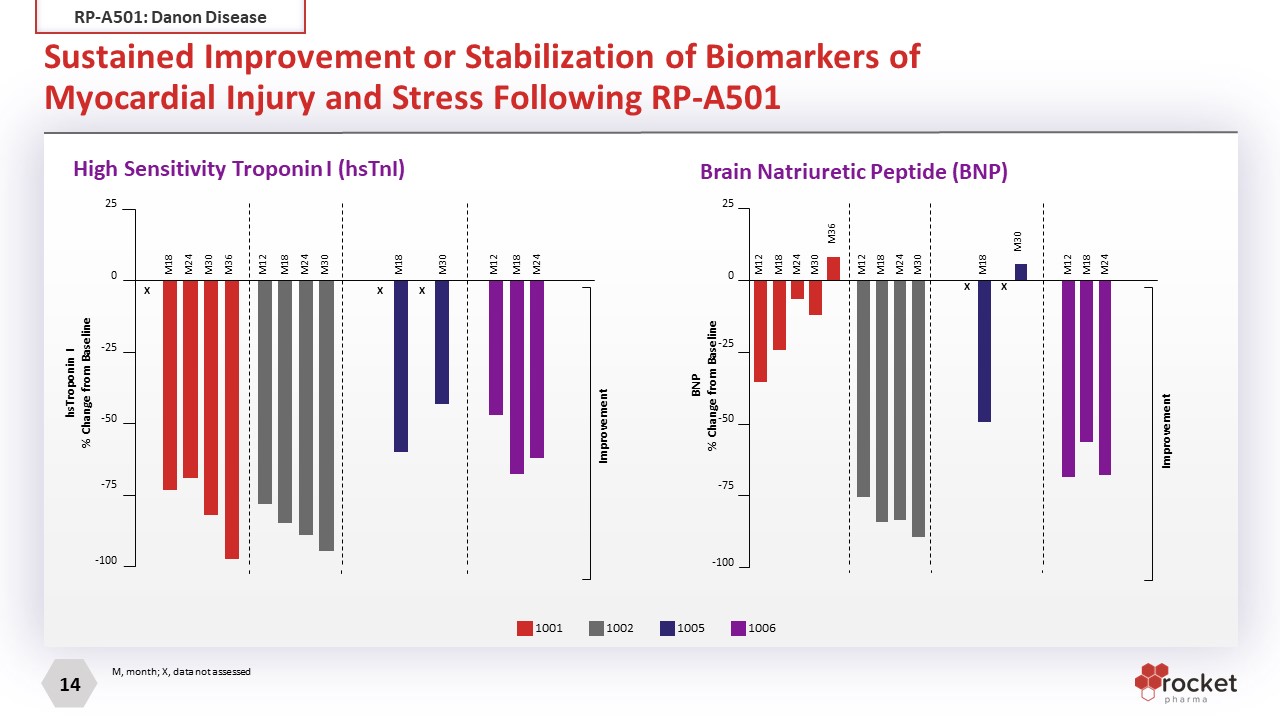
Sustained Improvement or Stabilization of Biomarkers of Myocardial Injury and Stress Following RP-A501 Brain Natriuretic Peptide (BNP) 1001 1002 1005 1006 High Sensitivity Troponin I (hsTnI) 25 0 -25 -50 -75 -100 M18 M24 M30 M36 X X X M18 M24 M30 M12 M18 M30 M18 M24 M12 Improvement hsTroponin I % Change from Baseline 25 0 -25 -50 -75 -100 M18 M24 M30 M36 M12 M18 M24 M30 M12 M18 M30 M18 M24 M12 Improvement BNP % Change from Baseline X X M, month; X, data not assessed RP-A501: Danon Disease
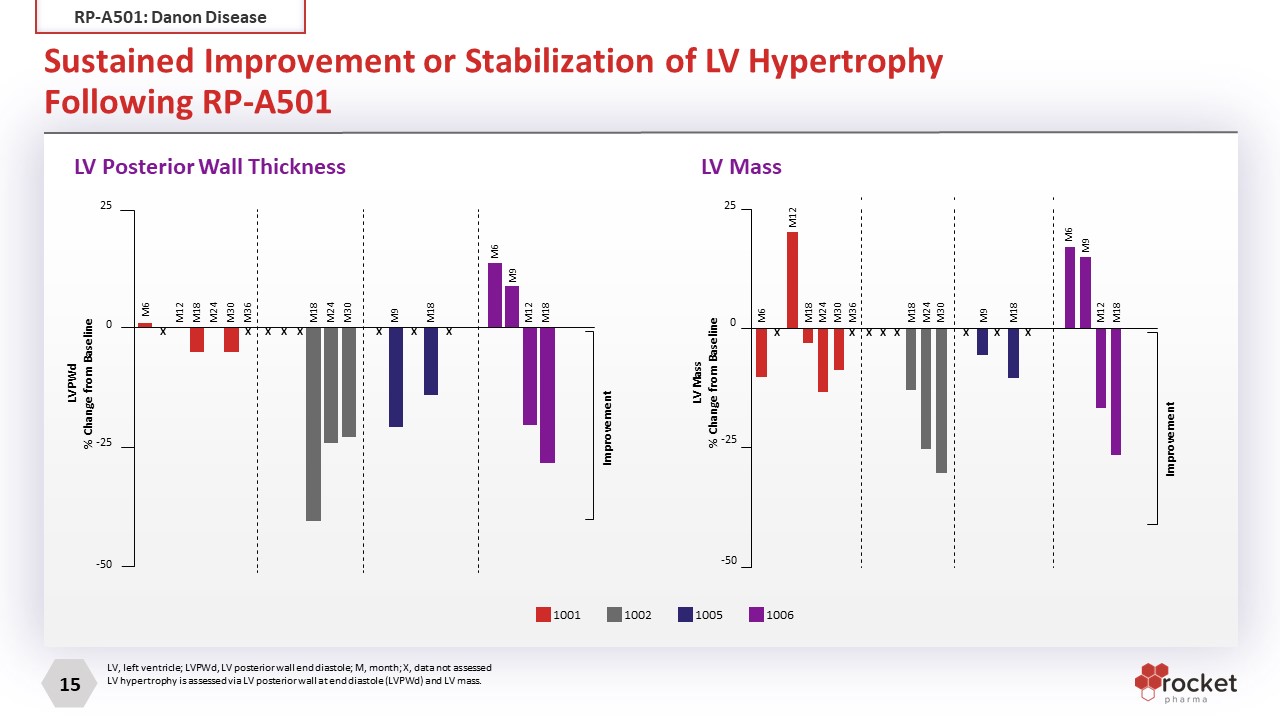
Sustained Improvement or Stabilization of LV HypertrophyFollowing RP-A501 LV, left ventricle; LVPWd, LV posterior wall end diastole; M, month; X, data not assessed LV hypertrophy is assessed via LV posterior wall at end diastole (LVPWd) and LV mass. LV Posterior Wall Thickness LV Mass M18 M24 M30 M36 M12 M6 M18 M24 M30 M18 M9 M18 M12 M9 M6 25 0 -25 -50 Improvement X X X X X X X X LVPWd % Change from Baseline LV Mass % Change from Baseline M18 M24 M30 M36 M12 M6 M18 M24 M30 M18 M9 M18 M12 M9 M6 Improvement X X X X X X X X 25 0 -25 -50 1001 1002 1005 1006 RP-A501: Danon Disease
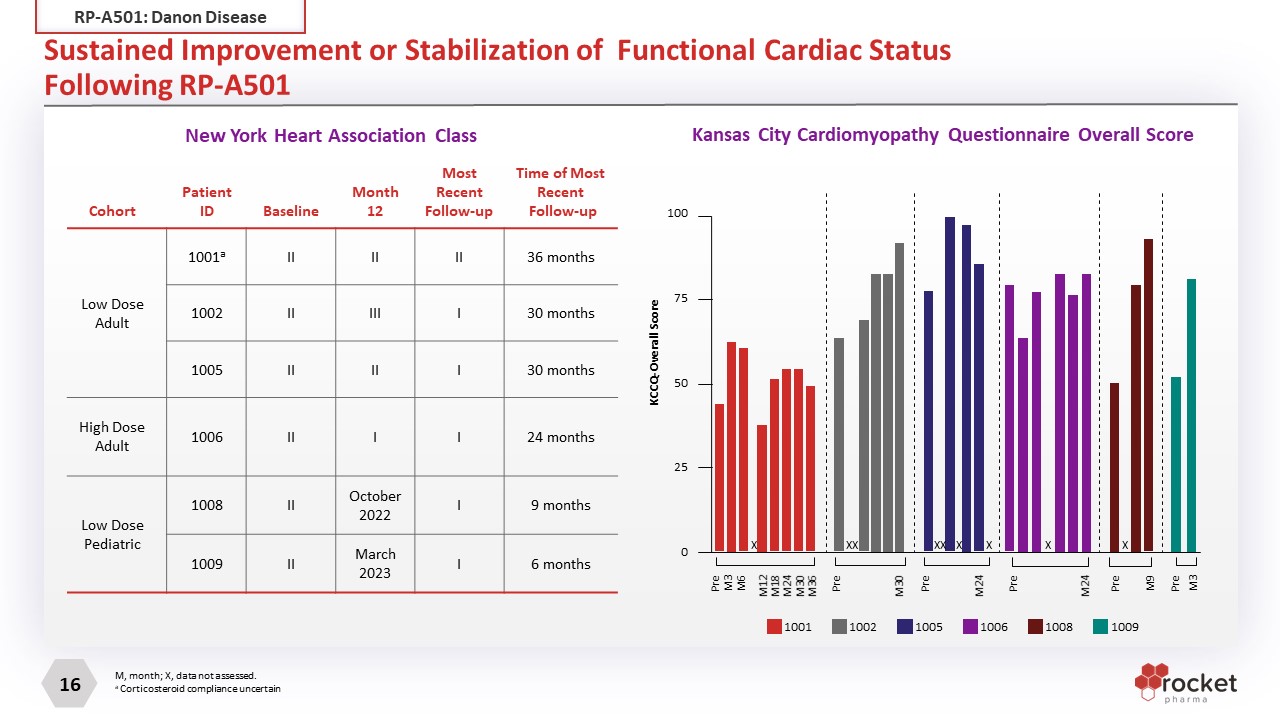
Sustained Improvement or Stabilization of Functional Cardiac StatusFollowing RP-A501 Cohort Patient ID Baseline Month 12 Most Recent Follow-up Time of Most Recent Follow-up Low Dose Adult 1001a II II II 36 months 1002 II III I 30 months 1005 II II I 30 months High Dose Adult 1006 II I I 24 months Low Dose Pediatric 1008 II October 2022 I 9 months 1009 II March 2023 I 6 months New York Heart Association Class 100 75 50 25 0 KCCQ-Overall Score 1001 1002 1005 1006 1008 M, month; X, data not assessed. a Corticosteroid compliance uncertain Pre M3 Pre M9 X Pre M24 X Pre M24 X X X X Pre M30 X X Pre M3 M6 M12 M18 M24 M30 M36 X 1009 Kansas City Cardiomyopathy Questionnaire Overall Score RP-A501: Danon Disease

Summary of Results PEDIATRIC COHORT ADULT COHORT Low-dose continues to be generally well tolerated at 2-3 years post-treatment Increased LAMP2B protein expression was associated with durable disease improvement or stabilization including clinical status (NYHA class, KCCQ), LV hypertrophy (LV wall thickness and mass), biomarkers of myocardial injury and stress (hsTroponin I and BNP), and cardiac histology All patients are alive in their early 20s, whereas median survival in DD males is 19 years old* RP-A501 was well tolerated No immediate, early or delayed RP-A501 related SAEs observed to date with enhanced immunomodulation Minimal complement activation Platelets remained within normal range Absent or limited worsening of skeletal myopathy with reduced steroid dose and more rapid taper, and introduction of sirolimus Increased LAMP2B protein expression was associated with early signals of improved cardiac histology, as well as serological evidence of decreased myocardial injury and stress Early improvement in NYHA class and KCCQ for both patients BNP, brain natriuretic peptide; hs, high sensitivity; KCCQ, Kansas City cardiomyopathy questionnaire; LAMP2B, lysosome-associated membrane protein 2B; LV, left ventricle; NYHA, New York Heart Association; SAE, serious adverse event. *Except for patient 1007 who received a heart transplant at 5 months due to Danon Disease progression. He is currently stable. Brambatti M et al. Int J Cardiol. 2019;286:92-98 Data cut-off September 27, 2022 with source data, verification through July 11, 2022. RP-A501: Danon Disease

Connecting Surrogate Endpoints to Functional Outcomes for Pivotal Study* Molecular Expression Cellular Structure Cardiac Structure and Function Cardiac Structure and Function Clinical Status Endomyocardial Bx: LAMP2 protein expression Endomyocardial Bx: Vacuolar area Serology: Troponin-I BNP Echo Parameters: Wall thickness (LVPWd, MLVWT) LVEF LV mass Hemodynamics: PCWp Cardiac output Functional parameters: NYHA Class KCCQ Overall Score Patient Screening Predose Month 3 Month 6 Month 12 Month 24 Month 36 RP-A501 Post Gene Therapy BNP, brain natriuretic peptide; Bx, biopsy; KCCQ, Kansas City cardiomyopathy questionnaire; LAMP2, lysosome-associated membrane protein 2; LV, left ventricle; LVEF, LV ejection fraction; LVPWd, LV posterior wall end diastole; MLVWT, maximal LV wall thickness; NYHA, New York Heart Association; PCWp, pulmonary capillary wedge pressure. *Pending regulatory feedback RP-A501: Danon Disease

Summary of Results Phase 1 enrollment and treatment are complete The enhanced immunomodulatory regimen was generally well tolerated and has mitigated adverse events in the pediatric cohort, who are currently 6 and 11 months post treatment The early LAMP2 expression data from the pediatric cohort are encouraging and consistent with that seen in the adult patients at the same timepoints The early clinical trends for the pediatric cohort are encouraging and consistent with the sustained clinical responses seen in the adults at 24-36 months Study design and endpoints have been identified for the planned Phase 2 pivotal study* and endorsed by an International Scientific and Clinical Advisory Board; FDA discussion planned at the end of this year *Pending regulatory feedback RP-A501: Danon Disease
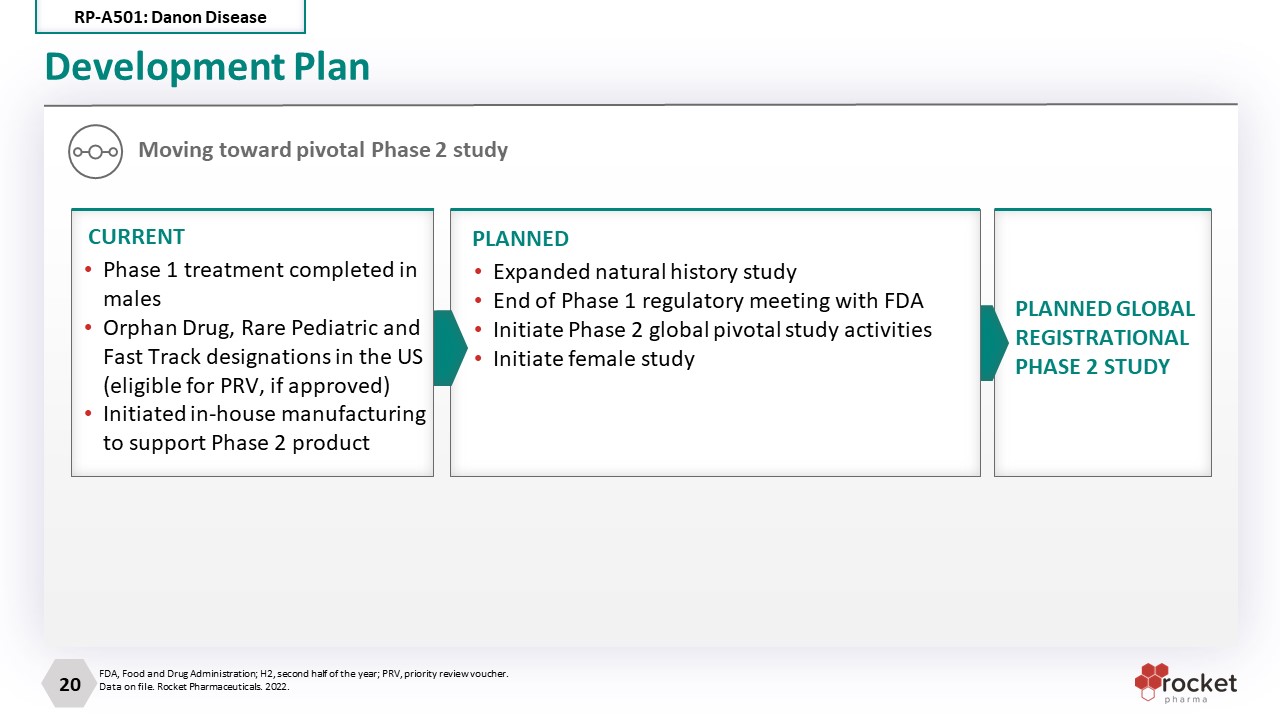
Development Plan Moving toward pivotal Phase 2 study PLANNED GLOBAL REGISTRATIONAL PHASE 2 STUDY Expanded natural history study End of Phase 1 regulatory meeting with FDA Initiate Phase 2 global pivotal study activities Initiate female study PLANNED Phase 1 treatment completed in males Orphan Drug, Rare Pediatric and Fast Track designations in the US (eligible for PRV, if approved) Initiated in-house manufacturing to support Phase 2 product CURRENT FDA, Food and Drug Administration; H2, second half of the year; PRV, priority review voucher. Data on file. Rocket Pharmaceuticals. 2022. RP-A501: Danon Disease