
Innovative Therapies for Skin Diseases N A S D A Q : N O V N | n o v a n . c o m C O R P O R AT E P R E S E N TAT I O N / M AY 2 0 2 2 Exhibit 99.1

NOVAN.COM FORWARD-LOOKING STATEMENTS This presentation contains forward-looking statements including, but not limited to, statements related to the acquisition of EPI Health, LLC (the “EPI Acquisition”), including the potential benefits of the EPI Acquisition, the therapeutic value and benefits of the Company’s marketed products and the potential therapeutic value and benefits of the Company’s Nitricil™ platform technology and its product candidates, the Company’s pharmaceutical development of nitric oxide-releasing product candidates, such as SB206 (berdazimer gel, 10.3%) for molluscum contagiosum, the potential timing of FDA submissions, the potential market opportunity for the Company’s product candidates and promoted products, the plans for launch and commercialization of SB206 (berdazimer gel, 10.3%), if approved, the Company’s expected cash runway, and the Company’s intention to enter into strategic relationships and transactions with third parties. These forward-looking statements are included throughout this presentation, and the Company uses the words “believe,” “expect,” “target,” “anticipate,” “may,” “plan,” “potential,” “will,” and similar expressions to identify forward-looking statements in this presentation. Such statements are based on the Company’s current beliefs and expectations. Forward-looking statements are subject to a number of risks and uncertainties that could cause actual results to differ materially from the Company’s expectations, including, but not limited to, risks related to the EPI Acquisition, including the ability to realize the anticipated benefits of the EPI Acquisition, which may be affected by, among other things, competition, the ability of management to integrate the combined company's business and operations, the ability of the parties to retain key employees, and costs related to the EPI Acquisition; risks related to the regulatory approval process, which is lengthy, time-consuming and inherently unpredictable, including the risk that the FDA will not agree with the Company’s approach to a potential NDA submission, that the Company’s product candidates may not be approved or that additional studies may be required for approval or other delays may occur, that the Company may not have sufficient quantities of drug substance and/or drug product to support regulatory submissions and that the Company may not obtain funding sufficient to complete the regulatory or development process; the Company’s limited experience as a company in obtaining regulatory approvals and commercializing pharmaceutical products and its ability to recruit and retain qualified personnel and key talent; changes in the size and nature of the market for the Company’s product candidates and marketed products, including potential competition, patient and payer perceptions and reimbursement determinations; the Company’s ability to grow revenues from marketed and promoted products; risks and uncertainties in the Company’s ongoing or future product development activities and preclinical studies, which may not prove successful in demonstrating proof-of concept, or may show adverse toxicological findings and, even if successful, may not necessarily predict that subsequent clinical trials will show the requisite safety and efficacy of the Company’s product candidates, or that any of the Company’s product candidates, if approved, will continue to demonstrate requisite safety and efficacy following their commercial launch; any operational or other disruptions as a result of the COVID-19 pandemic; risks related to the manufacture of raw materials, including, but not limited to, the Company’s active pharmaceutical ingredient and drug product components utilized in clinical trial materials, such as supply chain disruptions or delays, failure to transfer technology and processes to third parties effectively or failure of those third parties (or the Company in connection with the upfit of the Company’s facility) to obtain approval of and maintain compliance with the FDA or comparable regulatory authorities; the Company’s ability to obtain additional funding or enter into strategic or other business relationships or transactions necessary or useful for the further development or commercialization of the Company’s product candidates and the expansion of its business, on favorable terms or at all, or if such relationships or transactions are unsuccessful or the Company is unable to realize the potential economic benefits of such relationships or transactions; the Company’s reliance on arrangements with third parties to support its operations and its development, manufacturing and commercialization efforts and the risk that such parties will not successfully carry out their contractual duties or meet expected deadlines; and other risks and uncertainties described in the Company’s annual report filed with the Securities and Exchange Commission on Form 10-K for the twelve months ended December 31, 2021, and in the Company’s subsequent filings with the Securities and Exchange Commission. Such forward-looking statements speak only as of the date of this presentation, and the Company disclaims any intent or obligation to update these forward-looking statements to reflect events or circumstances after the date of such statements, except as may be required by law. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the Company’s own estimates and research. While the Company believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of or that any independent source has verified, any information obtained from third-party sources. 2

A medical dermatology company primarily focused on researching, developing and commercializing innovative therapeutic products for skin diseases

NOVAN.COM 4 5 NO-Based Product Candidates in Pipeline (Berdazimer gel, 10.3% (SB206), SB204, SB208, SB414, SB019) 3 Potential Near-Term Value Creators (Berdazimer gel, 10.3% (SB206), Rhofade, Wynzora) 1 NDA Targeted for Submission in 4Q 2022 (Berdazimer gel, 10.3% (SB206)) 4 Promoted Dermatology Products (Rhofade, Wynzora, Minolira, Cloderm) 2 Business Units (Recent acquisition combines R&D engine and established commercial capabilities) HIGHLIGHTS BY THE “NUMBERS”

NOVAN.COM A FULLY INTEGRATED MEDICAL DERMATOLOGY COMPANY 5 ✓ Robust NO-Based Drug Discovery Platform ✓ Proven Clinical-Stage Development Capabilities ✓ In-House cGMP Commercial Manufacturing ✓ Supply Chain and Market Access ✓ Sales and Marketing – 42 territories ✓ Financial Management Analytics, and Reporting Commercial CapabilitiesResearch and Development Engine With Acquisition of EPI Health, which on a stand-alone basis, had operating revenues of $17.6 million in 2021 and $5.3 million1 in Q1 2022 1. Represents the unaudited results of operations for the three months ended March 31, 2022, as if EPI Health reported on a stand-alone basis

NOVAN.COM Commercial 6

NOVAN.COM 1. Based on the 2003-2006 and 2009-2010 NHANES and U.S. Census Bureau’s Current Population Survey. NHANES=National Health and Nutrition Examination Surveys 2. Rachakonda TD et aJ Am Acad Dermatol 2014;70:512-6.l; 3. IQVIA, NPA, 2019 3. ”Understanding Rosacea.” Rosacea.org, accessed 10 Jan. 2022, https://www.rosacea.org/patients/materials/understanding-rosacea/what-is-rosacea. 4. Bickers DR, Lim HW, Margolis D, Weinstock MA, Goodman C, Faulkner E et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. Journal of the American Academy of Dermatology 2006;55:490-500 5. https://nationaleczema.org/eczema/types-of-eczema/atopic-dermatitis/ 7 INITIALLY COVERING TOP 4 MEDICAL DERMATOLOGY MARKETS 7.5M 16M 50M 26M Rosacea ~16 million people in the U.S.3 Psoriasis ~7.5 million people in the U.S.1,2 Acne ~50 million people in the U.S.4 Atopic Dermatitis ~9.6 million children and 16.5 million adults in the U.S.5

NOVAN.COM PROMOTED MEDICAL DERMATOLOGY PRODUCT PORTFOLIO 8 Sales Reps currently covering 42 territories across the U.S. with plan to cover 50 by year end
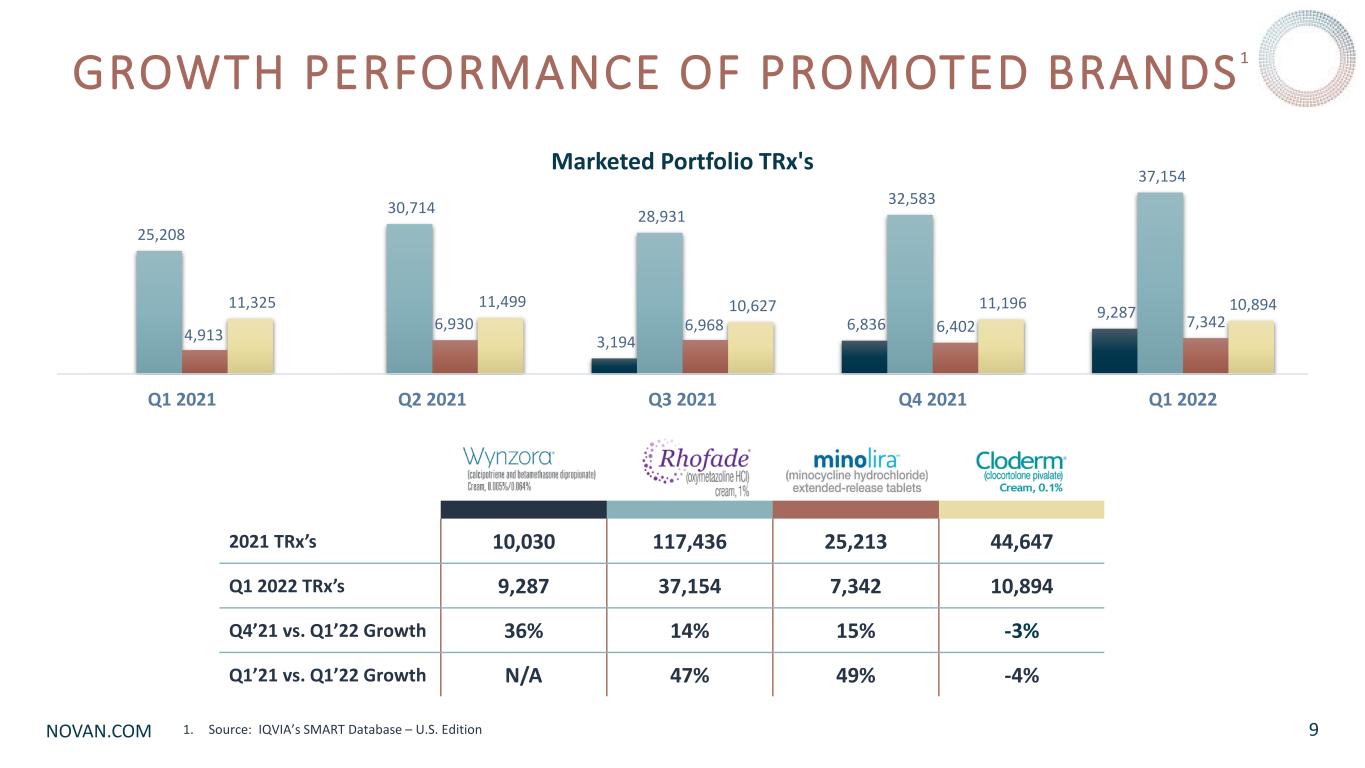
NOVAN.COM 2021 TRx’s 10,030 117,436 25,213 44,647 Q1 2022 TRx’s 9,287 37,154 7,342 10,894 Q4’21 vs. Q1’22 Growth 36% 14% 15% -3% Q1’21 vs. Q1’22 Growth N/A 47% 49% -4% GROWTH PERFORMANCE OF PROMOTED BRANDS1 1. Source: IQVIA’s SMART Database – U.S. Edition 9 3,194 6,836 9,287 25,208 30,714 28,931 32,583 37,154 4,913 6,930 6,968 6,402 7,342 11,325 11,499 10,627 11,196 10,894 Q1 2021 Q2 2021 Q3 2021 Q4 2021 Q1 2022 Marketed Portfolio TRx's

NOVAN.COM COMMERCIAL CAPABILITIES 10 Established Presence Across the Dermatology Community ✓ Supply Chain And Market Access Well established dermatology specialty pharmacy network Comprehensive market access (payor strategy, patient assistance program) Established supply chain with 3rd parties ✓ Sales And Marketing Experienced sales team and leadership Significant launch experience: Rhofade, Minolira, Cloderm, Wynzora Strong KOL relationships and engagement ✓ Financial Management Analytics, and Reporting Alignment of operational and financial reporting Enhanced third-party partnerships: data, sample management, financial drivers ROI analytics on key activities Ready the field. Focus on engagement. Measure team effectiveness.

NOVAN.COM KEY GROWTH MANDATES TO EXPAND PROMOTED PRODUCTS 11 Expand presence at premier dermatology meetings Increase utilization of omni-channel marketing/sales opportunities Raise awareness among target prescribers Strengthen KOL relationships Explore ex-U.S. opportunities Hire experienced sales leader Add reps in targeted territories

NOVAN.COM Research & Development 12

NOVAN.COM DEVELOPMENT PIPELINE 1. Subject to additional funding 2. Programs are not currently in active development 13 Product Candidate Indication Pre-IND Phase 1 Phase 2 Phase 3 Approval Next Targeted Milestone1 PRIORITY DEVELOPMENT PIPELINE SB206 (berdazimer gel, 10.3%) Molluscum NDA Submission 4Q 2022 SB204 Acne Vulgaris Commence Phase 3 study SB019 SARS-CoV-2 Submit IND and Commence Phase 1 POTENTIAL FUTURE VALUE DRIVERS2 SB414 Atopic Dermatitis Initiate Phase 1b study Psoriasis Evaluate potential next step SB208 Tinea Pedis Evaluate potential next step SB207 Genital Warts Initiate Phase 2/3 study

NOVAN.COM NITRICILTM PLATFORM 14 Active Pharmaceutical Ingredient: Berdazimer Sodium New Chemical Entity Patients treated to date: ~4,000 Compelling safety profile IP until as late as 2035
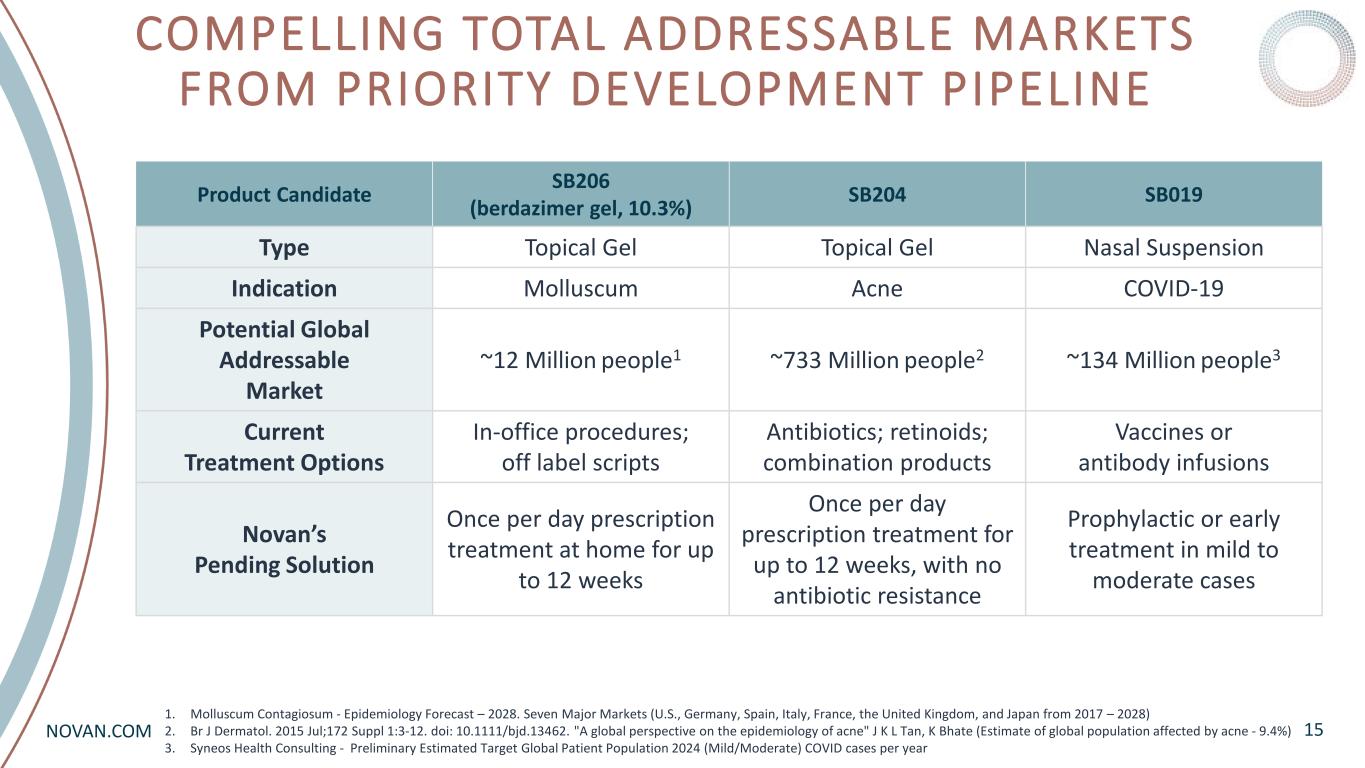
NOVAN.COM COMPELLING TOTAL ADDRESSABLE MARKETS FROM PRIORITY DEVELOPMENT PIPELINE 15 Product Candidate SB206 (berdazimer gel, 10.3%) SB204 SB019 Type Topical Gel Topical Gel Nasal Suspension Indication Molluscum Acne COVID-19 Potential Global Addressable Market ~12 Million people1 ~733 Million people2 ~134 Million people3 Current Treatment Options In-office procedures; off label scripts Antibiotics; retinoids; combination products Vaccines or antibody infusions Novan’s Pending Solution Once per day prescription treatment at home for up to 12 weeks Once per day prescription treatment for up to 12 weeks, with no antibiotic resistance Prophylactic or early treatment in mild to moderate cases 1. Molluscum Contagiosum - Epidemiology Forecast – 2028. Seven Major Markets (U.S., Germany, Spain, Italy, France, the United Kingdom, and Japan from 2017 – 2028) 2. Br J Dermatol. 2015 Jul;172 Suppl 1:3-12. doi: 10.1111/bjd.13462. "A global perspective on the epidemiology of acne" J K L Tan, K Bhate (Estimate of global population affected by acne - 9.4%) 3. Syneos Health Consulting - Preliminary Estimated Target Global Patient Population 2024 (Mild/Moderate) COVID cases per year

NOVAN.COM SB206 (berdazimer gel, 10.3%) Molluscum Contagiosum 16 Potential to Be First-In-Class Prescription Treatment for Molluscum

NOVAN.COM SB206 BERDAZIMER GEL, 10.3% 17 1. Molluscum Contagiosum - Epidemiology Forecast – 2028. Seven Major Markets (U.S., Germany, Spain, Italy, France, the United Kingdom, and Japan from 2017 – 2028) 2. Syneos Health Consulting Primary (n=40 Pediatricians, n=39 Dermatologists, n=4 Pediatric Dermatologists) and Secondary Research (2019) Note: B-SIMPLE3 was a Phase 1, Maximum-Use PK study in patients and will also be included in the NDA package. B-SIMPLE1 & B-SIMPLE2 will be integrated to support B- SIMPLE4 results Molluscum: Contagious skin infection caused by the molluscipoxvirus Berdazimer gel, 10.3% demonstrated clinical evidence of efficacy and a favorable safety profile across Phase 3 program B-SIMPLE4 was statistically significant for the primary efficacy endpoint (p<0.0001) with robust clinical data Berdazimer gel, 10.3% as a topical prescription therapy with a rapid treatment benefit, if approved, would satisfy an important patient-care need for the treatment of molluscum High unmet medical need with no FDA-approved prescription treatment U.S. addressable market of ~6 million, primarily children 1-14 years old1 Tolerability of existing procedures remains a concern with Pediatricians and Dermatologists2
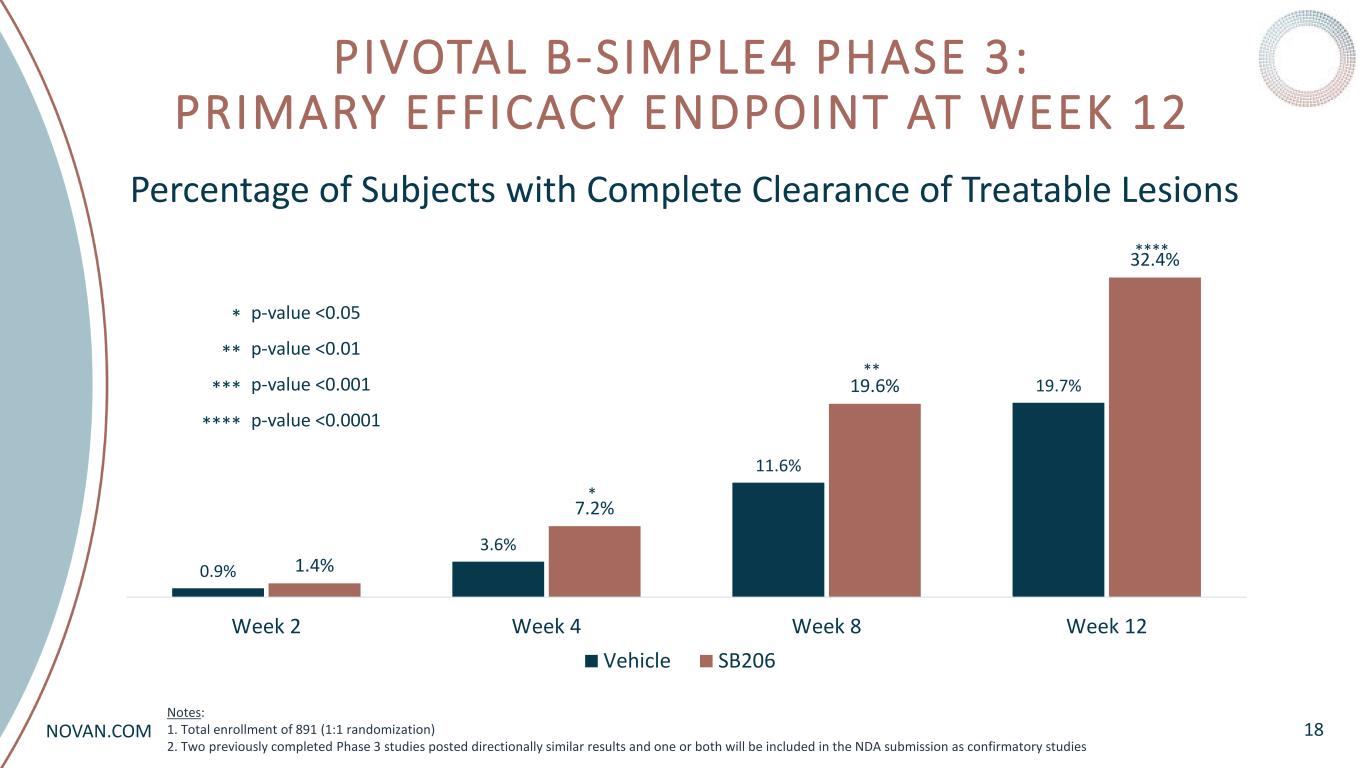
NOVAN.COM PIVOTAL B-SIMPLE4 PHASE 3: PRIMARY EFFICACY ENDPOINT AT WEEK 12 18 Percentage of Subjects with Complete Clearance of Treatable Lesions 0.9% 3.6% 11.6% 19.7% 1.4% 7.2% 19.6% 32.4% Week 2 Week 4 Week 8 Week 12 Vehicle SB206 **** ** * p-value <0.05 p-value <0.01 p-value <0.001 p-value <0.0001 * ** *** **** Notes: 1. Total enrollment of 891 (1:1 randomization) 2. Two previously completed Phase 3 studies posted directionally similar results and one or both will be included in the NDA submission as confirmatory studies

NOVAN.COM 19 PIVOTAL PHASE 3: SECONDARY ENDPOINTS 90% Clearance and 0 or 1 Remaining Lesions at Week 12 23.90% 24.60% 43% 43.50% 90% Clearance at Week 12 0 or 1 Lesions at Week 12 Vehicle SB206 ************ p-value <0.0001

NOVAN.COM 20 PIVOTAL PHASE 3: SECONDARY ENDPOINT - CHANGE AT WEEK 4 0% -1.31% -5.55% -14.53% -31.28% -12.91% -27.12% -49.25% -57.48% BL Week 2 Week 4 Week 8 Week 12 Vehicle SB206 p-value <0.05 p-value <0.01 p-value <0.001 p-value <0.0001 * ** *** **** *** **** **** **** % Change From Baseline Lesion Count
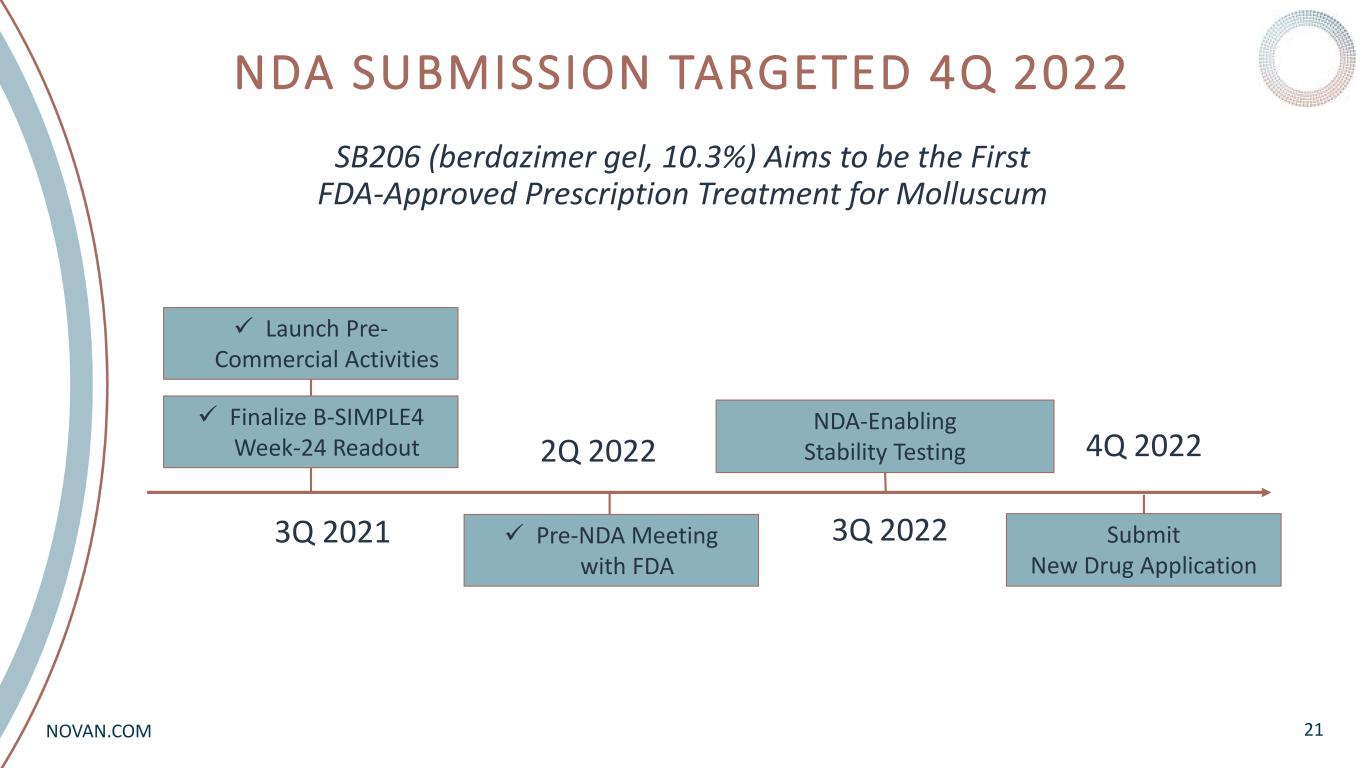
NOVAN.COM NDA SUBMISSION TARGETED 4Q 2022 21 SB206 (berdazimer gel, 10.3%) Aims to be the First FDA-Approved Prescription Treatment for Molluscum Finalize B-SIMPLE4 Week-24 Readout 3Q 2021 2Q 2022 4Q 2022 Launch Pre- Commercial Activities NDA-Enabling Stability Testing Pre-NDA Meeting with FDA Submit New Drug Application 3Q 2022

NOVAN.COM 22 STRATEGIC INITIATIVES FOR LAUNCH READINESS Ongoing Activities to Support Successful Potential Launch and Commercialization of Berdazimer Gel, 10.3% (SB206) Scale commercial manufacturing Pricing and reimbursement strategy Medical affairs outreach Sales force training and alignment

NOVAN.COM MOLLUSCUM THERAPEUTIC LANDSCAPE 231. Syneos Health Consulting Market Opportunity Assessment for SB206 in Molluscum Contagiosum (2019) ~1.3 million new U.S. cases diagnosed annually1 Potential Therapy Characteristics Topical – SB206 (berdazimer gel, 10.3%) Prescription treatment at home Safety profile Tolerability Primary care or pediatrician prescribed Therapy Characteristics Procedures – (cryo, curettage, cantharidin) Multiple in-office treatments Burning / Blistering Pain / Scarring Primarily performed by Dermatologist or Pediatric Dermatologist

NOVAN.COM SB204 Acne Vulgaris 24 Late-stage asset with two completed Phase 3 studies
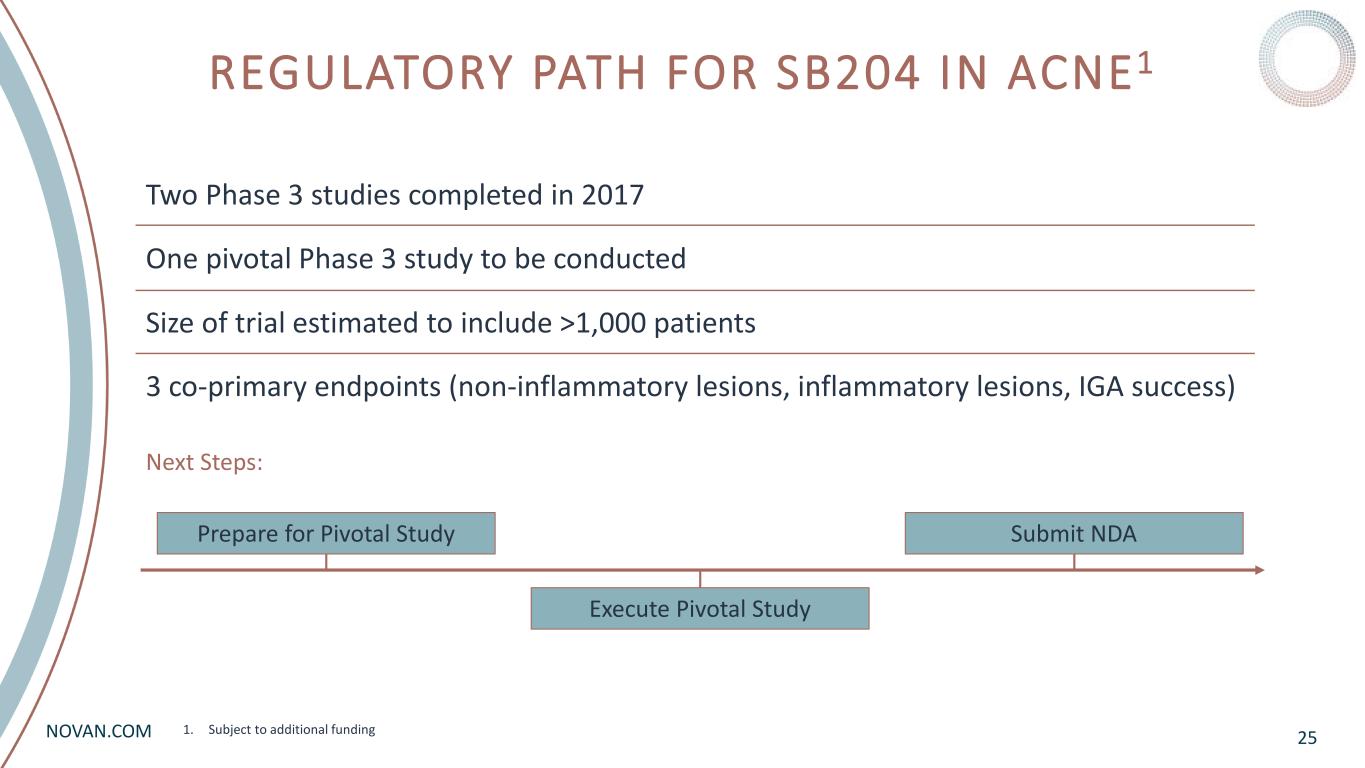
NOVAN.COM REGULATORY PATH FOR SB204 IN ACNE1 25 Next Steps: Two Phase 3 studies completed in 2017 One pivotal Phase 3 study to be conducted Size of trial estimated to include >1,000 patients 3 co-primary endpoints (non-inflammatory lesions, inflammatory lesions, IGA success) Prepare for Pivotal Study Execute Pivotal Study Submit NDA 1. Subject to additional funding

NOVAN.COM 26 SB019 COVID-19 Pre-IND Filed April 2022

NOVAN.COM REGULATORY PATH FOR SB019 IN COVID-191 27 Pre-clinical IND enabling studies completed2 Candidate formulation administered intranasally in animals Berdazimer sodium well-tolerated up to the maximum dose tested Drug-device combination in development for potential Phase 2/3 study(s) 1. Subject to additional funding 2. Assumes pre-clinical studies completed to date are all that is required to initiate Phase 1 in human volunteers Next Steps: Targeting Phase 1 Study in Healthy Volunteers Targeting Phase 2/3 Study(s) in Patients Submit New Drug Application

NOVAN.COM Corporate Overview 28

NOVAN.COM FINANCIAL SNAPSHOT 1. Existing cash runway into early fourth quarter of 2022 based on Management’s projections and planned development and operating activities described in the Company’s March 31, 2022 Form 10-Q as filed with the SEC on May 16, 2022 29 $35.5M Cash as of March 31, 20221 20 Days of Commercial Operations for 1st Quarter 2022 56% Growth in Promoted Product TRx’s from Q1’21 COMMERCIAL OPERATIONS March 11, 2022 – March 31, 2022 Revenue of $1.2 million Net Loss of $0.7 million R&D OPERATIONS January 1, 2022 – March 31, 2022 Revenue of $0.7 million Net Loss of $12.7 million

NOVAN.COM 30 PROVEN TEAM WITH EXPERTISE ACROSS DRUG DEVELOPMENT AND COMMERCIALIZATION Paula Brown Stafford, MPH President, Chief Executive Officer and Chairman Tomoko Maeda-Chubachi, MD, Ph.D., MBA Chief Medical Officer Carri Geer, Ph.D. SVP, Chief Technology Officer Brian M. Johnson, MBA Chief Commercial Officer John M. Gay, CPA Chief Financial Officer John Donofrio EVP, Chief Operating Officer; President, EPI Health

NOVAN.COM 31 A medical dermatology company primarily focused on researching, developing and commercializing innovative therapeutic products for skin diseases Grow sales and expand commercial organization through multiple initiatives Advance berdazimer gel, 10.3% (SB206) towards NDA, submission targeted before end of 2022 Execute pre-commercial activities to support potential launch of berdazimer gel, 10.3% (SB206) Pursue multiple business development initiatives, including out-licensing products ex-U.S. Explore development of our pipeline to potentially expand commercial product portfolio FOCUS GOING FORWARD

Innovative Therapies for Skin Diseases N A S D A Q : N O V N | n o v a n . c o m Thank You!