
Copyright © 2022 Zosano Pharma Corporation Corporate Presentation January 2022 1 Exhibit 99.1

Copyright © 2022 Zosano Pharma Corporation Disclaimer 2 This presentation contains forward-looking statements regarding the benefits and potential uses of Zosano’s transdermal microneedle system and product candidates, including M207, the resubmission strategy of the M207 New Drug Application, the potential benefits and availability of M207 for patients, the potential benefits and availability of a Covid-19 vaccine candidate, the pursuit of non-dilutive and partnership opportunities to expand the potential use of our technology and other future events and expectations. Readers are urged to consider statements that include the words "may," "will," "would," "could," "should," "might," "believes," "estimates," "projects," "potential," "expects," "plans," "anticipates," "intends," "continues," "forecast," "designed," "goal," "approximately" or the negative of those words or other comparable words to be uncertain and forward- looking. These statements are subject to risks and uncertainties that are difficult to predict, and actual outcomes may differ materially. These include, without limitation, risks and uncertainties associated with the process of discovering, developing and commercializing products that are safe and effective for use as human therapeutics, risks inherent in the effort to build a business around such products and other risks and uncertainties described under the heading "Risk Factors" in the Company's most recent Annual Report on Form 10-K and its periodic reports filed with the Securities and Exchange Commission. Although Zosano believes that the expectations reflected in these forward-looking statements are reasonable, we cannot in any way guarantee that the future results, level of activity, performance or events and circumstances reflected in forward- looking statements will be achieved or occur. All forward-looking statements are based on information currently available to Zosano and Zosano assumes no obligation to update any such forward-looking statements. The website addresses or links to websites included in this presentation are inactive text references and are not intended to be actual links to the websites. The information contained on such websites, or that can be accessed therein, is not incorporated into or otherwise a part of this presentation.
Copyright © 2022 Zosano Pharma Corporation Zosano Pharma: Working to Transform How Drugs Are Delivered 3 Significant Clinical Evidence Versatile Platform Differentiated Technology OUR MISSION Advancing Patient Care and Transforming Patient Lives Through Therapies Developed Utilizing Our Proprietary Delivery Platform OUR FOCUS Late-Stage Clinical Development Program for the Acute Treatment of Migraine Where Current Therapies Have Significant Limitations OUR FUTURE Transdermal Microneedle System Technology Designed to Allow for Innovative Application of Small Molecules, Proteins, Peptides and Vaccines

Copyright © 2022 Zosano Pharma Corporation Executive Summary and Recent Developments 4 Transformative Delivery Platform Addressing Significant Unmet Needs in Migraine Platform Development Key Developments ▪ Migraine is Ranked Globally as the Seventh Most Disabling Disease1 ▪ Migraine Impacts approximately 37 Million People in the US2 ▪ M207, if Approved, May Have the Potential to Offer Fast, Complete and a Durability of Effect to Patients Suffering From Migraine3 ▪ Technology (formerly MACROFLUX) Developed at ALZA Corp/J&J ▪ 18 Patent Families and IP Protection ▪ Patents with Claims Covering Composition of Matter and Method of Use for M207 with Expirations in 2037 ▪ Completed Pharmacokinetic (PK) Study (CP 2021-001) in October 2021 ▪ Resubmitted M207 NDA in January 2022 under Section 505(b)(2) of FDCA ▪ Application Relies on FDA’s Previous Findings of Safety and Efficacy for ZOMIG® Nasal Spray (NDA 21-450) ▪ Application Includes CP 2021-001 Data and Seeks to Establish a PK Bridge to ZOMIG® Nasal Spray ▪ Successful Formulation of a Vaccine Developer’s COVID-19 Vaccine Candidate Utilizing our Transdermal Microneedle System ▪ Two Ongoing Feasibility Studies with Undisclosed Partners ▪ Actively Seeking Collaborations that Utilize our Proprietary Transdermal Microneedle System Technology 1. Steiner TJ et al. Migraine: the seventh disabler. The Journal of Headache and Pain 2013, 14:1. 2. American Migraine Foundation (2019) Migraine Essentials: What It's Like Living with Migraine. Retrieved from https://americanmigrainefoundation.org/living-with-migraine/migraine-essentials. 3. Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224.
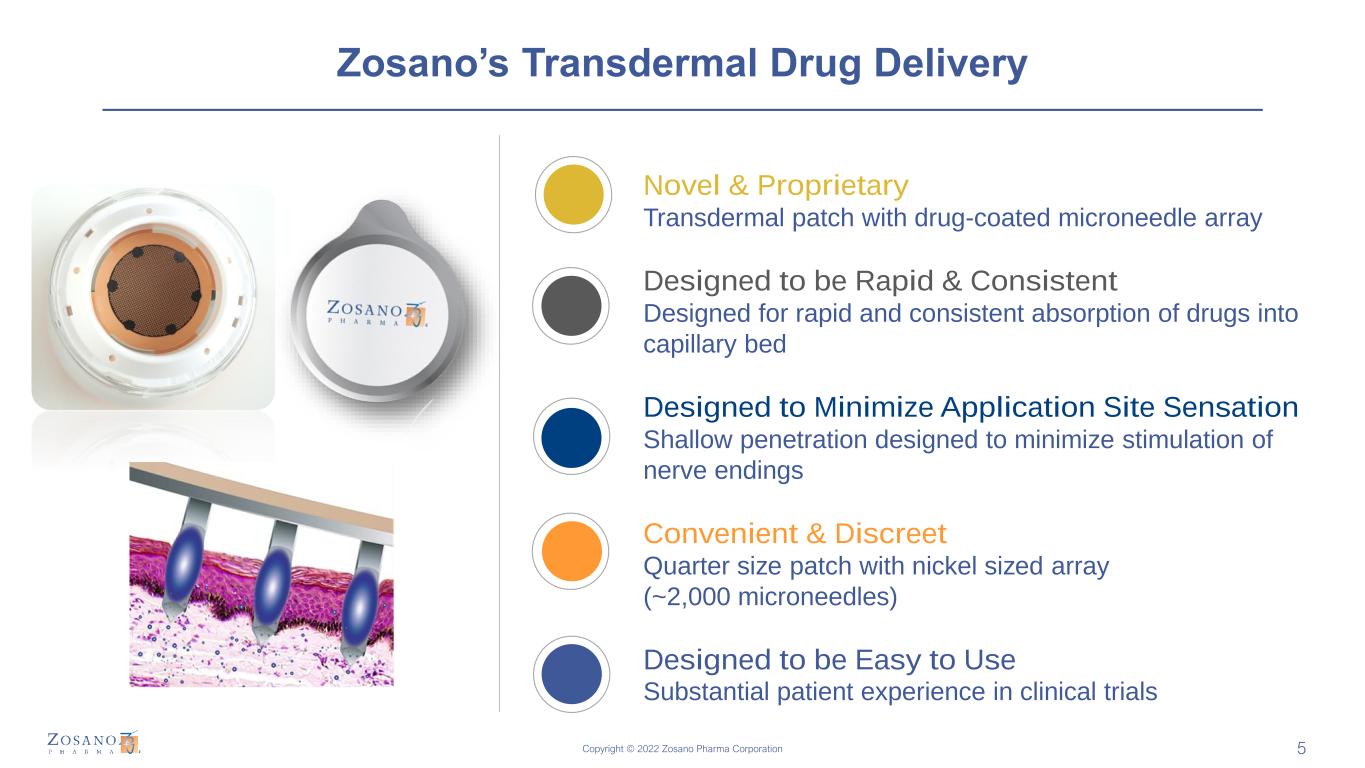
Copyright © 2022 Zosano Pharma Corporation Zosano’s Transdermal Drug Delivery 5 Novel & Proprietary Transdermal patch with drug-coated microneedle array Designed to be Rapid & Consistent Designed for rapid and consistent absorption of drugs into capillary bed Designed to Minimize Application Site Sensation Shallow penetration designed to minimize stimulation of nerve endings Convenient & Discreet Quarter size patch with nickel sized array (~2,000 microneedles) Designed to be Easy to Use Substantial patient experience in clinical trials

Copyright © 2022 Zosano Pharma Corporation M207 Designed for Optimization of Zolmitriptan Delivery 6

Copyright © 2022 Zosano Pharma Corporation DESIGNED TO ADDRESS UNMET NEEDS IN MIGRAINE 7

Copyright © 2022 Zosano Pharma Corporation Migraine is Highly Prevalent, Debilitating and Costly 8 Prevalent1 ▪ 3rd Most Prevalent Disorder in the World ▪ Impacts ~25% of U.S. Households ▪ Occurs in 12% of U.S. Population ▪ 90% Unable to Function Normally ▪ 1.2 MM ER Visits per Year ▪ 25% of women experience 4+ Migraines/Month ▪ Attacks Last 4-72 Hours CostlyDebilitating1 ▪ $36B in Lost Productivity2 ▪ $5.4B in Treatment Costs1 1. Migraine Research Foundation (2019). Migraine Facts. Retrieved from https://migraineresearchfoundation.org/about-migraine/migraine-facts. 2. Bonafede M, Sapra S, Shah N, Tepper S, Cappell K, Desai P. Direct and indirect healthcare resource utilization and costs among migraine patients in the United States [published online February 15, 2018]. Headache. doi: 10.1111/head.13275
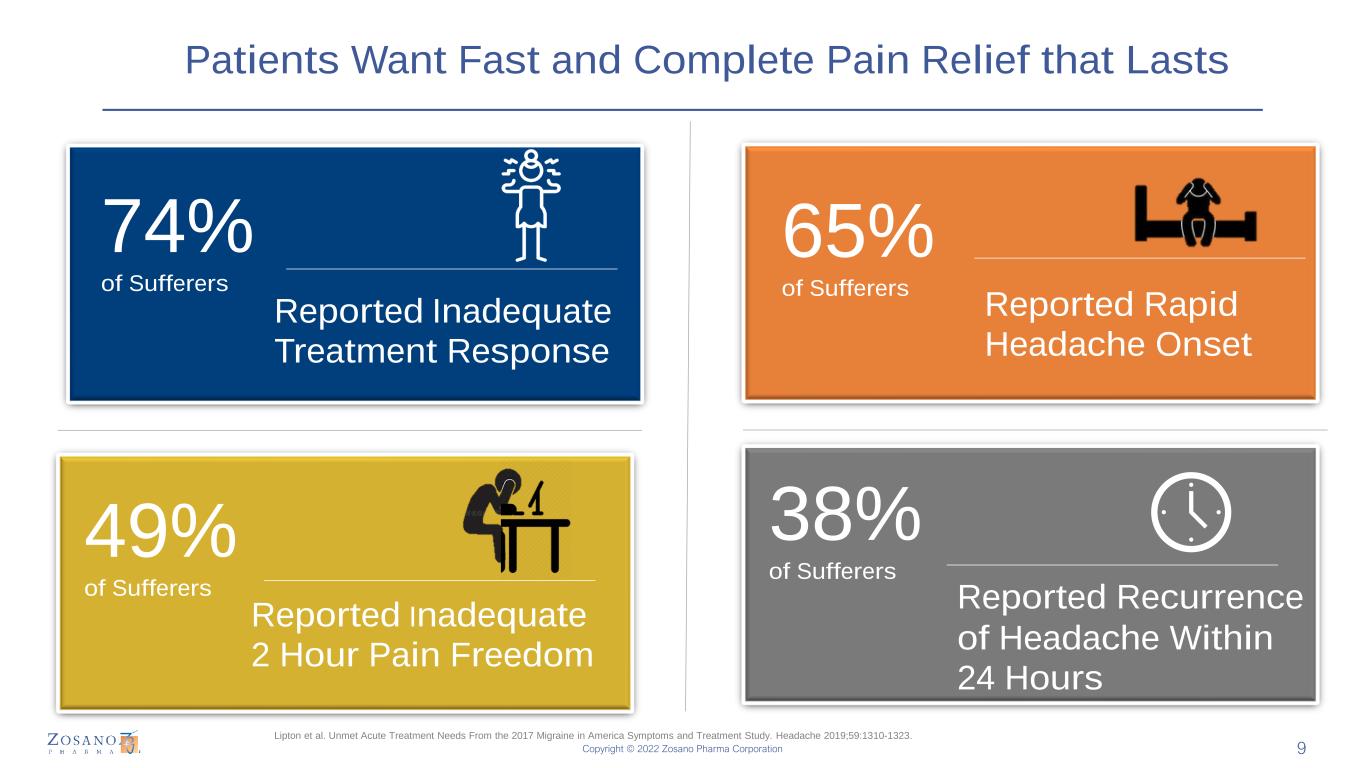
Copyright © 2022 Zosano Pharma Corporation Patients Want Fast and Complete Pain Relief that Lasts 74% of Sufferers 38% of Sufferers 49% of Sufferers 65% of Sufferers Reported Inadequate Treatment Response Reported Rapid Headache Onset Reported Inadequate 2 Hour Pain Freedom Reported Recurrence of Headache Within 24 Hours Lipton et al. Unmet Acute Treatment Needs From the 2017 Migraine in America Symptoms and Treatment Study. Headache 2019;59:1310-1323. 9

Copyright © 2022 Zosano Pharma Corporation What Do Society Guidelines Tell Us About the Goals of Acute Treatment of Migraine? The 2018/2021 American Headache Society Position Statements On Integrating New Migraine Treatments Into Clinical Practice3,4 ▪ Rapid and consistent freedom from pain and associated symptoms, especially the most bothersome symptom, without recurrence4 ▪ Treat early after the onset of a migraine attack3 ▪ Choose a non-oral route of administration for selected patients3 ▪ Account for tolerability and safety issues3 “…and use migraine-specific agents (triptans), for moderate or severe attacks…”3 The International Headache Society Guidelines for the Controlled Trials of Acute Treatment of Migraine Attacks1 ▪ The most clinically relevant information about the efficacy of migraine pharmacotherapy.2 ▪ Freedom from pain (2 hours) before the use of rescue medication is simple, clinically relevant, reflects patients’ expectations1 ▪ Sustained pain free (2-48 hours) is a recommended secondary endpoint, and it is the ideal migraine treatment response1 1. Tfelt-Hansen P, Pascual J, Ramadan N, Dahlöf C, D'Amico D, Diener HC, Hansen JM, Lanteri-Minet M, Loder E, McCrory D, Plancade S, Schwedt T; International Headache Society Clinical Trials Subcommittee. Guidelines for controlled trials of drugs in migraine: third edition. A guide for investigators. Cephalalgia. 2012 Jan;32(1):6-38.. 2. S. D. Silberstein, L. C. Newman, M. J. Marmura, S. J. Nahas & S. J. Farr (2013) Efficacy endpoints in migraine clinical trials: the importance of assessing freedom from pain, Current Medical Research and Opinion, 29:7, 861-867, 3. The American Headache Society Position Statement On Integrating New Migraine Treatments Into Clinical Practice. Headache. 2019 Jan;59(1):1-18.. 4. Ailani J, Burch RC, Robbins MS; Board of Directors of the American Headache Society. The American Headache Society Consensus Statement: Update on integrating new migraine treatments into clinical practice. Headache. 2021 Jul;61(7):1021-1039. 10

Copyright © 2022 Zosano Pharma Corporation Triptans Are and Will Likely Remain the Standard of Care 11 1.Lipton et al. Unmet Acute Treatment Needs From the 2017 Migraine in America Symptoms and Treatment Study. Headache 2019;59:1310-1323. 2.GlaxoSmithKline. (2013). IMITREX (sumatriptan) Nasal Spray HIGHLIGHTS OF PRESCRIBING INFORMATION. Research Triangle Park, NC: Author. 3.Alam A. et al. Triptan Use and Discontinuation in a Representative Sample of Persons With Migraine: Results From Migraine in America Symptoms and Treatment (MAST) Study Neurology April 09, 2019; 92 (15 Supplement) P4.10-019. 4. D.H.E. 45® (dihydroergotamine mesylate) Injection, USP Prescribing Information. East Hanover, New Jersey: Author. 5. Impel NeuroPharma Inc. TrudhesaTM nasal spray (dihydroergotamine mesylate) [package insert]. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/213436s000lbl.pdf 6.Satsuma Pharmaceuticals (2020 September 10) Satsuma Pharmaceuticals Announces Topline Results from EMERGE Phase 3 Trial of STS101 for the Acute Treatment of Migraine [Press Release] https://investors.satsumarx.com/2020-09-10-Satsuma-Pharmaceuticals-Announces-Topline-Results-from-EMERGE-Phase-3-Trial-of-STS101-for-the-Acute-Treatment-of-Migraine. 7. Smith TR, Winner P, Aurora SK, Jeleva M, Hocevar-Trnka J, Shrewsbury SB. STOP 301: A Phase 3, open-label study of safety, tolerability, and exploratory efficacy of INP104, Precision Olfactory Delivery (POD®) of dihydroergotamine mesylate, over 24/52 weeks in acute treatment of migraine attacks in adult patients. Headache. 2021 Aug 7. DHE4 Yet Current Triptan & DHE Formulations Fail to Meet Patient Needs of Subjects Complained of Bad or Unusual Taste2 24% 25% 82% of Subjects noted Nausea Affected Treatment1 Discontinuation Rate for Triptan Injectables was observed3 ▪ Formulations not Optimal ▪ Associated with Severe Cardiac Events ▪ Not Recommended for Use if Pregnant ▪ Trudhesa Approved 9/21(505b2)5 ▪ Efficacy data from Migranal label* ▪ Black Box Warning – Drug Interactions ▪ Local Irritative Symptoms Reported in 52% of Subjects in Open Label Trial ▪ Multi-step Nasal Application ▪ Satsuma’s STS101 Failed to Meet Clinical Endpoints in Phase 3 Trial6 In Studies Evaluating Triptans: *open label results: PF at 2 hours = 38% & MBS at 2hours = 52.1%7

Copyright © 2022 Zosano Pharma Corporation 1. Biohaven Holding Company Ltd. Nurtec® (Rimegepant) [package insert]. U.S. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212728s000lbl.pdf. Revised 2/2020. Accessed August 31, 2021. 2 Allergan Pharmaceuticals International Limited. Ubrelvy® (Ubrogepant) [package insert]. U.S. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf. Revised 12/2019. Accessed August 31, 2021. 3 Dodick, D., Goadsby, P., Hutchinson, S., Lipton, P. (2018, April 22). Rimegepant Phase 3 Update and Clinical Perspectives [Investor Webcast Event] held concurrently with the American Academy of Neurology, Los Angeles, CA, United States. https://www.biohavenpharma.com/investors/news-events/press-releases/04-19-2018 4.Ready DM., Sbar E. Build Your Migraine Toolbox. Neurology Live. 2020 Oct; Volume 3 (5):34-40. 5.Eli Lilly and Company. Reyvow® (Lasmiditan) [package insert]. U.S. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/211280s001lbl.pdf. Revised 7/ Accessed August 31, 2021. A Closer Look at New Market Entrants GEPANTS DITANS Modest Pain Freedom at 2 Hours in Clinical Study (21.2%)1,2 Observed Slow Onset of Pain Freedom (43% of Subjects at 4 Hours in Clinical Study)3 Metabolized Through the CYP3A4 Liver Pathway1,2 Fatty Meal Delayed Tmax by 1 Hour1,2 in Clinical Study “Although it is exciting to have additional entries in the acute migraine tool kit, these newer medications should not be seen as replacements for current medications but instead as options for patients in whom current medications are contraindicated or ineffective.”4 Crosses the Blood Brain Barrier – Potential for Central Nervous System Side Effects4 Clinical Studies of Lasmiditan showed: 17% Dizziness5 7% Sedation5 8 Hour Post-Dose Driving Restriction and Schedule V Medication5 12

Copyright © 2022 Zosano Pharma Corporation A Stratified Treatment Plan Incorporating a Toolbox Approach is Key to Optimizing Treatment of Acute Attacks “There are foreseeable scenarios in which a patient may have 2 different triptans, an NSAID, and a ditan or gepant in their tool kit”2 ▪ Migraine is a Complex Neurological Disease and Attacks Experienced by One Patient Can Vary Considerably ▪ A Stratified Treatment Approach Based on Severity and Frequency of Attacks has been Recommended1,2 ▪ Requires Multiple Tools to Treat Acute Attacks Since Not Every Patient will Respond to the Same Treatment for Every Attack2 ▪ Stratified Care has Demonstrated Both a Higher Mean Effectiveness and Lower Mean Cost than Stepped Care, as Well as a Higher Patient Satisfaction Rate2 1.Harris L, L'Italien G, O'Connell T, Hasan Z, Hutchinson S, Lucas S. A Framework for Estimating the Eligible Patient Population for New Migraine Acute Therapies in the United States. Adv Ther. 2021 May 31. doi: 10.1007/s12325-021-01781-z. Epub ahead of print. PMID: 34057676. 2.Ready DM., Sbar E. Build Your Migraine Toolbox. Neurology Live. 2020 Oct; Volume 3 (5):34-40. 13

Copyright © 2022 Zosano Pharma Corporation M207 Clinical Results From Phase 2/3 ZOTRIP Study 15 Mins Results 23% of Patients Achieved Pain Relief Efficacy Results Activity 81% 42% Observed Peak Plasma Concentration Reported Pain Freedom at 2 Hours 78% Reported Pain Relief at 24 Hours Reported Pain Relief at 2 Hours Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224. 71% Reported Pain Relief at 48 Hours 14

Copyright © 2022 Zosano Pharma Corporation 41.5% 68.3% 14.3% 42.9% 0% 20% 40% 60% 80% Pain Freedom MBS % S u b je c ts A c h ie v in g P a in F re e d o m o r F re e d o m fr o m M B S a t 2 H o u rs M207 3.8mg Placebo Clinically Significant Results on Pain Freedom and Most Bothersome Symptom (MBS) in Phase 2/3 Study 15 P = 0.0001 TG = 27.2 P = 0.0009 TG = 25.4 Successful Achievement of Co-Primary Endpoints Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224. TG = Therapeutic Gain (difference between M207 and Placebo)

Copyright © 2022 Zosano Pharma Corporation M207 Treatment Resulted in High Rates of Pain Freedom from 30 Minutes to 48 Hours 16 7.3% 17.1% 26.8% 41.5% 51.2% 54.9% 62.2% 69.5% 64.6% 0% 20% 40% 60% 30 minutes 45 minutes 1 hour 2 hour 3 hour 4 hour 12 hours 24 hours 48 hours % S u b je c ts A c h ie v in g P a in F re e d o m Pain Freedom 30 Minutes to 48 Hours * ** ** ** ** ** **** Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224. *p=<0.05 **p=0.01

Copyright © 2022 Zosano Pharma Corporation 23.2% 46.3% 56.1% 68.3% 80.5% 81.7% 82.9% 80.5% 78.0% 71.0% 0% 20% 40% 60% 80% 15 minutes 30 minutes 45 minutes 1 hour 2 hour 3 hour 4 hour 12 hours 24 hours 48 hours % S u b je c ts A c h ie v in g P a in R e li e f Pain Relief 15 Minutes to 48 Hours * ** ** ** ** **** Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224. *p=<0.05 **p=0.01 Early Separation From Placebo Observed Within 15 Minutes and was Statistically Significant by 1 Hour 17
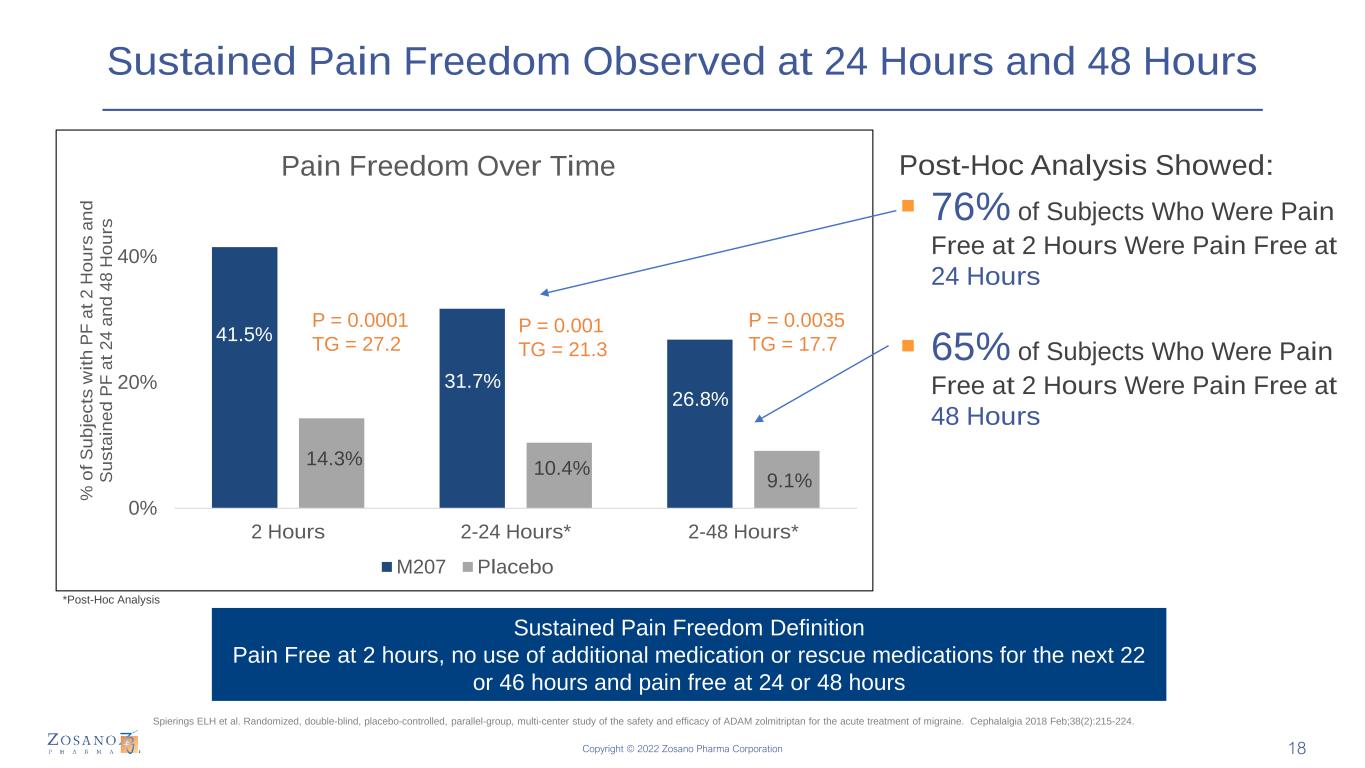
Copyright © 2022 Zosano Pharma Corporation Sustained Pain Freedom Observed at 24 Hours and 48 Hours 41.5% 31.7% 26.8% 14.3% 10.4% 9.1% 0% 20% 40% 2 Hours 2-24 Hours* 2-48 Hours* % o f S u b je c ts w it h P F a t 2 H o u rs a n d S u s ta in e d P F a t 2 4 a n d 4 8 H o u rs Pain Freedom Over Time M207 Placebo P = 0.0001 TG = 27.2 Post-Hoc Analysis Showed: ▪ 76% of Subjects Who Were Pain Free at 2 Hours Were Pain Free at 24 Hours ▪ 65% of Subjects Who Were Pain Free at 2 Hours Were Pain Free at 48 Hours P = 0.001 TG = 21.3 P = 0.0035 TG = 17.7 Sustained Pain Freedom Definition Pain Free at 2 hours, no use of additional medication or rescue medications for the next 22 or 46 hours and pain free at 24 or 48 hours Spierings ELH et al. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018 Feb;38(2):215-224. *Post-Hoc Analysis 18
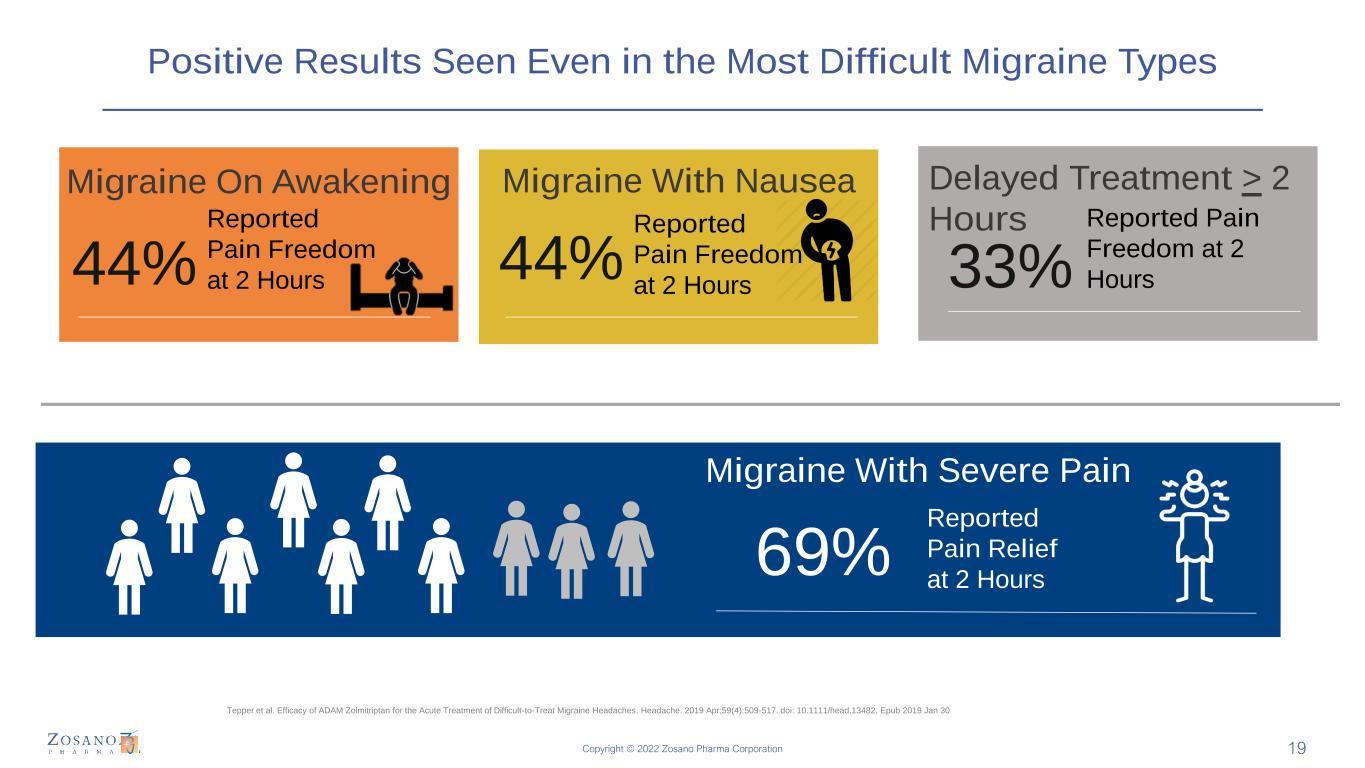
Copyright © 2022 Zosano Pharma Corporation Positive Results Seen Even in the Most Difficult Migraine Types Migraine On Awakening 44% 69% Reported Pain Freedom at 2 Hours Migraine With Severe Pain Reported Pain Relief at 2 Hours 44% 33% Migraine With Nausea Reported Pain Freedom at 2 Hours Delayed Treatment > 2 Hours Reported Pain Freedom at 2 Hours Tepper et al. Efficacy of ADAM Zolmitriptan for the Acute Treatment of Difficult-to-Treat Migraine Headaches. Headache. 2019 Apr;59(4):509-517. doi: 10.1111/head.13482. Epub 2019 Jan 30 19

Copyright © 2022 Zosano Pharma Corporation Long Term Safety Results Consistent with Placebo-Controlled Study Endpoint ZOTRIP Pivotal Study (Single Dose) Open-Label Long-Term Placebo (N = 77) M207 3.8 mg (N = 82) M207 3.8 mg (N = 5,617 attacks) Pain Freedom at 2 hours 14% 42% 44% Pain Relief at 2 hours 57% 81% 81% Sustained Pain Freedom 2-24 hour 10% 32% 38% Sustained Pain Freedom 2-48 hour 9% 27% 35% Sustained Pain Relief 2-24 hour 38% 68% 70% Sustained Pain Relief 2-48 hour 33% 63% 65% * For sustained endpoints, data from all timepoints 2-24 (or 48) hours had to be present Spierings E.L.H., et al. (2020 June 13th). Comparison of a single dose and repeat dose of M207 for pain freedom, pain relief, sustained pain freedom and sustained pain relief for the acute treatment of migraine [Virtual Oral Presentation] American Headache Society 2020 Virtual Annual Scientific Meeting, US. https://americanheadachesociety.org/events/virtual-annual-scientific-meeting/ 20

Copyright © 2022 Zosano Pharma Corporation M207 was Well Tolerated in the Long-Term Safety Study 21 ▪ Most Common Adverse Events were Application Site Redness/Swelling. ▪ 95% were Mild ▪ 80% Resolved Within 48 hours ▪ Less than 2% of Patients Reported Neurological Triptan-like Side Effects Such as Dizziness and Paresthesia. Spierings E.L.H., et al. (2020 June 13th). Comparison of a single dose and repeat dose of M207 for pain freedom, pain relief, sustained pain freedom and sustained pain relief for the acute treatment of migraine [Virtual Oral Presentation] American Headache Society 2020 Virtual Annual Scientific Meeting, US. https://americanheadachesociety.org/events/virtual-annual-scientific-meeting/
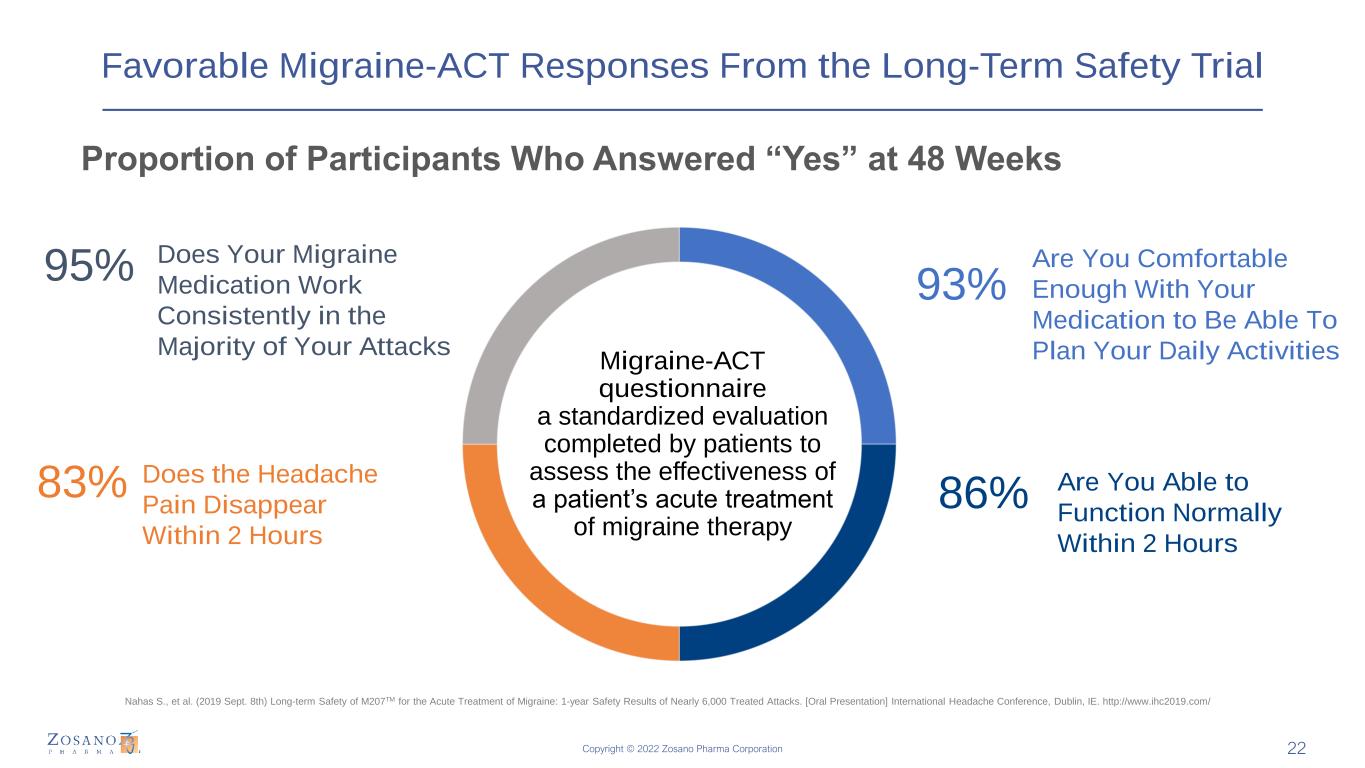
Copyright © 2022 Zosano Pharma Corporation Favorable Migraine-ACT Responses From the Long-Term Safety Trial Proportion of Participants Who Answered “Yes” at 48 Weeks 22 Migraine-ACT questionnaire a standardized evaluation completed by patients to assess the effectiveness of a patient’s acute treatment of migraine therapy Does the Headache Pain Disappear Within 2 Hours Are You Able to Function Normally Within 2 Hours 95% Are You Comfortable Enough With Your Medication to Be Able To Plan Your Daily Activities Does Your Migraine Medication Work Consistently in the Majority of Your Attacks 83% 93% 86% Nahas S., et al. (2019 Sept. 8th) Long-term Safety of M207TM for the Acute Treatment of Migraine: 1-year Safety Results of Nearly 6,000 Treated Attacks. [Oral Presentation] International Headache Conference, Dublin, IE. http://www.ihc2019.com/

Copyright © 2022 Zosano Pharma Corporation COMMERCIAL STRATEGY 23
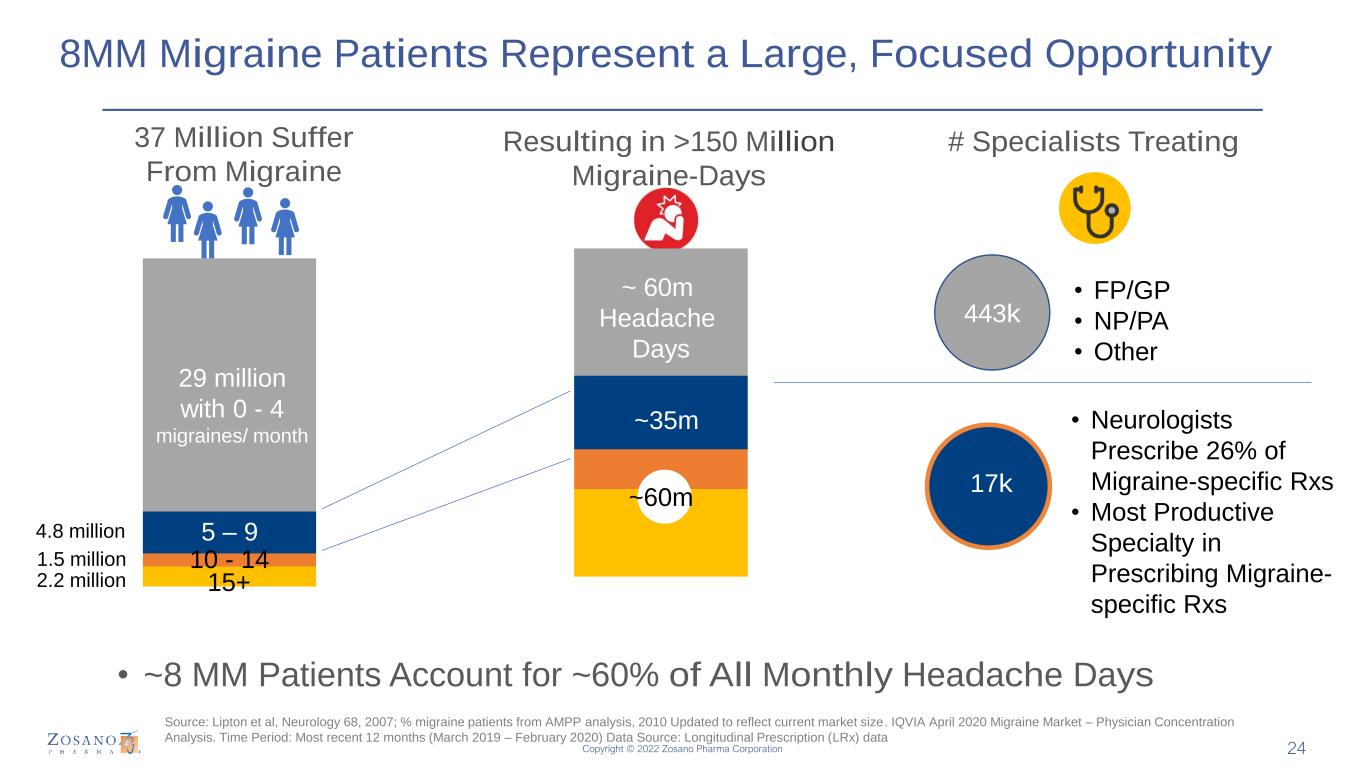
Copyright © 2022 Zosano Pharma Corporation 8MM Migraine Patients Represent a Large, Focused Opportunity 37 Million Suffer From Migraine • ~8 MM Patients Account for ~60% of All Monthly Headache Days Resulting in >150 Million Migraine-Days 29 million with 0 - 4 migraines/ month 5 – 9 10 - 14 15+ # Specialists Treating ~ 60m Headache Days ~35m 443k 17k • FP/GP • NP/PA • Other • Neurologists Prescribe 26% of Migraine-specific Rxs • Most Productive Specialty in Prescribing Migraine- specific Rxs 4.8 million 1.5 million 2.2 million ~60m Source: Lipton et al, Neurology 68, 2007; % migraine patients from AMPP analysis, 2010 Updated to reflect current market size. IQVIA April 2020 Migraine Market – Physician Concentration Analysis. Time Period: Most recent 12 months (March 2019 – February 2020) Data Source: Longitudinal Prescription (LRx) data 24

Copyright © 2022 Zosano Pharma Corporation Patients Continue to Suffer Even with New Market Entrants 25 Neurologists Have a Difficult-To-Treat Population 67% Of Physicians’ Patients are on Preventive Therapy Experiencing > 4 Headaches Monthly 300+ Migraine Patients Treated Monthly (Avg.) 51% of These Patients Have High Unmet Needs AND… Strongly Agree They Have a Substantial “Difficult-To-Treat” Migraine Patient Population 45% Trinity Market Research (2020). Zosano Pharma Proprietary Neurologist and Headache Specialist Market Research. (N=100)

Copyright © 2022 Zosano Pharma Corporation Neurologists Strongly Agree That… 79% Still an UNMET NEED in the acute setting even in patients that respond to preventive therapy 70% Would offer a NON- ORAL triptan that offers fast and complete pain relief that is sustained and well tolerated 63% Using a NON-ORAL therapy in the difficult to treat population is ideal since many migraine patients have nausea and vomiting Patients with morning migraine need a therapy with FAST ONSET and high success of PAIN RELIEF 79% Trinity Market Research (2020). Zosano Pharma Proprietary Neurologist and Headache Specialist Market Research. (N=100) 26

Copyright © 2022 Zosano Pharma Corporation Physician Market Research: Strong Positive Reaction to M207’s Potential Product Profile Differentiated Potential Product Attributes Key Drivers for HCP Adoption ZS Associates May 2020 Market Research: Insights drawn from n=19 Neurologists, n=4 NP/PAs, n=11 PCPs, and n=5 KOLs Positive Impression of Clinical Data; Comfortable With Molecule and Using New Route of Administration Based on Clinical Data POTENTIAL IDEAL PATIENT TYPES OVERALL PERCEPTION OF POTENTIAL PRODUCT PROFILE KEY DRIVERS ▪ Potential Rapid Onset of Action ▪ Potential Duration of Response ▪ Patch Route of Administration ▪ Safety and Tolerability Data ▪ Experiences Early Nausea and/or Early Vomiting Due to Migraine ▪ Averse to Injections Due to Fear of Needles ▪ Experiences Rapid Onset of Migraine ▪ Averse to Nasal Sprays Due to Bad Taste 27

Copyright © 2022 Zosano Pharma Corporation Payer Feedback on Market Access for Product X, if Approved ▪ Recognition that Migraine Patients May Need Non-oral Treatment Options ▪ Speed of Onset and Sustained Response Noted as Positives ▪ Likely Formulary Placement Will be Tier 3 or Non-preferred After Failure of Two Generic Triptans ▪ Formulary Placement is Comparable to Current Branded Triptans ▪ Price Expectations in the Range of the Branded Triptans ▪ Payer Access is Not Expected to be a Barrier 1. Precision Xtract Market Research (2020). Zosano Pharma Proprietary National Payer Market Research. (100MM Lives) 2. ZS Associates Market Research (2020). Zosano Pharma Proprietary National Payer Market Research. (~90MM Lives) . Top Line National, Regional Payer and PBM Findings 28

Copyright © 2022 Zosano Pharma Corporation Strong Market Opportunity with a Compelling Value Proposition Large and Concentrated Marketplace Differentiated Potential Clinical Profile ▪ Fast Onset of Action ▪ Significant Pain Freedom ▪ Lack of Recurrence ▪ Low Percentage of Triptan-like Side Effects ▪ Large Migraine Population ▪ Small Percentage of HCPs Treat the Majority of Patients ▪ Neurologists are the Most Productive Specialty Competitive Managed Care Access Anticipated ▪ Tier 3 Non- Preferred Status ▪ Formulary Status Comparable to Branded Triptans ▪ Pricing Expectations in Range of Branded Triptans 29

Copyright © 2022 Zosano Pharma Corporation ▪ October: CRL from FDA ▪ December: Ended Enrollment in Phase 2/3 Cluster Study Development of M207 ▪ June: 1st Subject Enrolled In Pivotal Study ▪ November: Completion of Enrollment 2016 2017 20202019 30 2021 ▪ October: Publication of Pivotal Data in Cephalalgia ▪ November: Initiation of 1 Year LTSS ▪ February: Completion of LTSS ▪ October: Initiation of Phase 2/3 Study in Cluster ▪ December: M207 NDA Submission ▪ January: Completed Type A Meeting with FDA to Discuss Requirements for Resubmission ▪ February: Received Official Type A Meeting Minutes ▪ October: Completed PK Study and Submitted Type C Briefing Book to FDA ▪ December: Received Type C WRO Feedback from FDA 2022 ▪ January: Resubmitted NDA to FDA

Copyright © 2022 Zosano Pharma Corporation CLUSTER HEADACHE 31

Copyright © 2022 Zosano Pharma Corporation ▪ Cluster headaches, which occur in cyclical patterns or cluster periods, are one of the most painful types of headache ▪ 15%-22% Have Suicidal Ideations2 Significant Unmet Need in Cluster Headache 32 . Highly Disabling Chronic Neurological Condition1 Short-lasting Headache Attacks Characterized By3 Prevalence of Cluster Headache ▪ Severe Unilateral Temporal/orbital Pain ▪ Ipsilateral Autonomic Symptoms ▪ Lasting 15–180 Minutes (when untreated) ▪ Sense of Restlessness or Agitation 1. Jürgens T, Gaul C, et al. Impairment in episodic and chronic cluster headache. Cephalalgia 2010; 31:671–682.2. Rozen TD, Fishman RS. Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache 2012;52:99-113. 3. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018: 38, 1–211. 4. Fischera M, Marziniak M, Gralow I, Evers S. The incidence and prevalence of cluster headache: a meta-analysis of population-based studies. Cephalalgia 2008;28:614-8. 5. Ford HL, Gerry Em Johnson M et al. A prospective study of the incidence, prevalence and mortality of multiple sclerosis in Leeds. J Neurol 2002; 249:260-5. 6. Klapper, et all, (2011) Cluster Headache. Headache, Face, Neck Pain Science, Evaluation and Management. Retrieved from https” books.Google.com. ▪ Approximately 1 in 1000 Suffer From the Condition ▪ Prevalence Similar to Parkinson's and Multiple Sclerosis4,5 ▪ Over 300,000 People Suffer From Cluster Headache in the US Limited Clinical Development ▪ Last FDA Approved Acute Treatment Occurred in the 1990s

Copyright © 2022 Zosano Pharma Corporation Cluster Headache (C213) Clinical Trial Program Update 33 Terminated Phase 2/3 Clinical Trial Design Cluster Patients: 120 Population: Chronic or episodic cluster headache sufferers Design: Each cluster headache sufferer will take a single dose to treat a qualifying headache Dosing: 1.9 mg and 3.8 mg vs placebo (1:1:1) Co-primary Endpoints: • Proportion of subjects with pain relief at 15 minutes • Proportion whose pain relief is sustained from 15 minutes to 60 minutes 20202019 2021 ▪ March: Enrollment suspended due to COVID; resumed in July ▪ December: Ended Enrollment in Phase 2/3 Cluster Study ▪ October: Initiation of Phase 2/3 Study in Cluster; Planned enrollment of 120 subjects ▪ August: Announced preliminary results: 42 subjects were randomized, 22 had post- treatment self-reported diary data
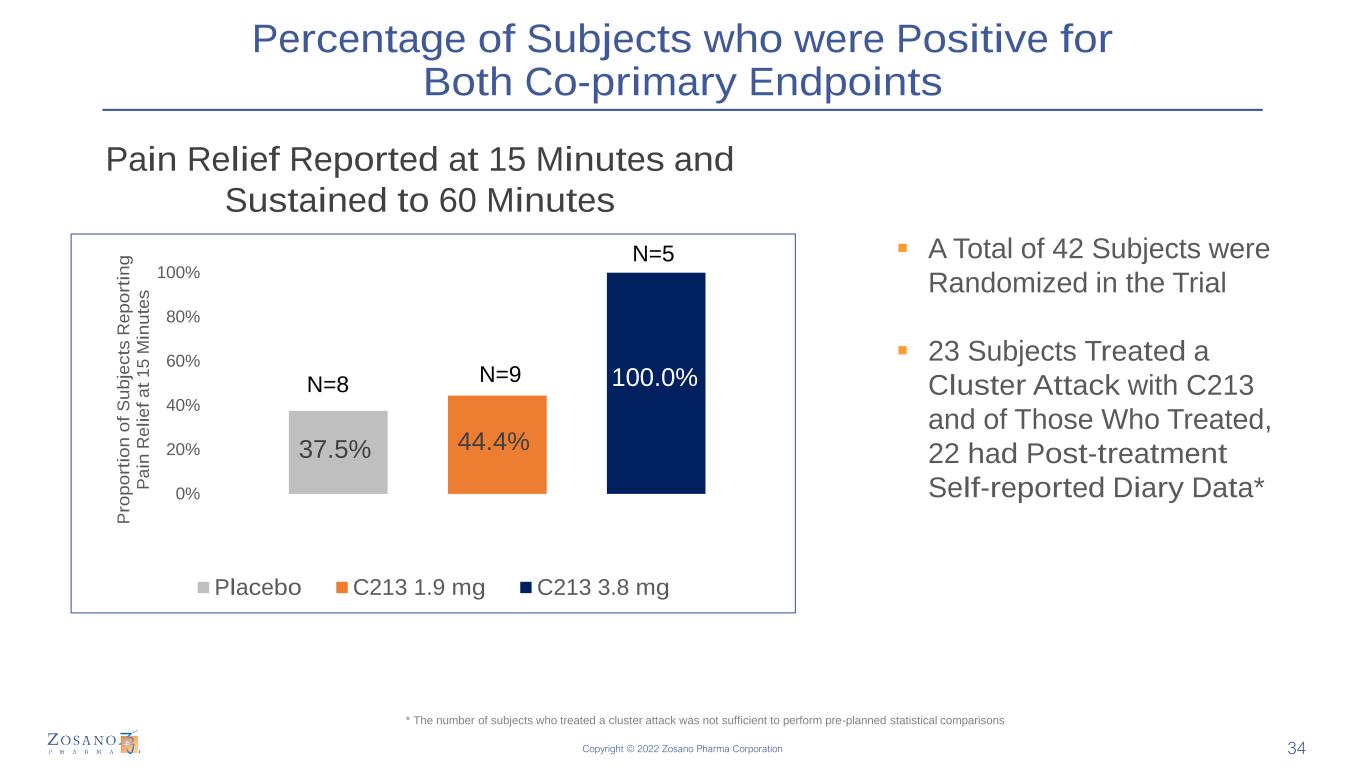
Copyright © 2022 Zosano Pharma Corporation Percentage of Subjects who were Positive for Both Co-primary Endpoints 34 37.5% 44.4% 100.0% 0% 20% 40% 60% 80% 100% 15 MinuteP ro p o rt io n o f S u b je c ts R e p o rt in g P a in R e li e f a t 1 5 M in u te s Placebo C213 1.9 mg C213 3.8 mg N=5 ▪ A Total of 42 Subjects were Randomized in the Trial ▪ 23 Subjects Treated a Cluster Attack with C213 and of Those Who Treated, 22 had Post-treatment Self-reported Diary Data* n=5 * The number of subjects who treated a cluster attack was not sufficient to perform pre-planned statistical comparisons Pain Relief Reported at 15 Minutes and Sustained to 60 Minutes N=8 N=9

Copyright © 2022 Zosano Pharma Corporation PIPELINE EXPANSION 35

Copyright © 2022 Zosano Pharma Corporation Leveraging Platform & Clinical Validation for Pipeline Expansion PRE-CLINICAL & CLINICAL PROOF-OF-CONCEPT STUDIES ▪ Initiated Three Collaborations in 2020 - Two Ongoing Feasibility Studies with Undisclosed Partners o Assessing feasibility for undisclosed molecule - Successfully formulated and coated a COVID-19 Vaccine Candidate ▪ Small Molecules − Zolmitriptan ▪ Hormones, Peptides & Proteins − Clinical: Glucagon (Phase 2), PTH (Phase 2), Desmopressin (Phase 1) − Preclinical: EPO, HGH ▪ Prophylactic Vaccines − Undisclosed Compound (Phase 1) FUTURE : POTENTIAL PROGRAMS ▪ Cancer Vaccines ▪ COVID-19 Vaccine ▪ Immuno-modulation / Immuno-therapy ▪ Local / Systemic Delivery Applications ▪ Novel Compounds 36

Copyright © 2022 Zosano Pharma Corporation Partnering Opportunity for Development of Microneedle COVID-19 Vaccine ▪ Ongoing Work with COVID-19 Vaccine Developers ▪ Pursuing Non-dilutive Funding Opportunities for COVID -19 Programs Through Governmental Agencies Potential Benefits of Utilizing Zosano’s Microneedle Patch Technology EASY DISPOSAL: Once Applied the User Would Dispose of the Unit in Standard Trash Receptacles STABLE: Formulations Designed to be Stable at Room Temperature Potentially Enabling Distribution Using Mail Distribution Systems SAFE FOR HANDLING: Patch is Designed to Prevent the User From Inadvertently Touching the Microneedles and Potentially Removing Some of the Vaccine PROOF OF CONCEPT: Phase 1 Clinical Data for Influenza Vaccine Showed That the Microneedle Coated Tri-valent Flu Vaccine was Comparable in Immune Response to the Commercial IM Injection EXTENSIVE CLINICAL EVALUATIONS: Over 40,000 patch applications With No Incidences of Infection POTENTIALLY FAVORABLE DELIVERY METHOD: Coated Microneedles That Deliver Directly to Epidermal/dermal Skin Layers Which May Lead to an Improved Response and Potential for Dose Sparing Therapies 37

Copyright © 2022 Zosano Pharma Corporation Zosano Pharma Microneedle Array Drug Delivery System Ideal solution for delivery of peptides, proteins and hydrophilic drugs ▪ First generation transdermal patches limited to hydrophobic molecules ▪ Low bioburden manufacturing and terminal sterilization mitigates need for aseptic manufacturing ▪ Room temperature stable and no reconstitution required: eliminates need for cold supply chain ▪ Simple reusable applicator with unit-dose patch enables single-step drug delivery ▪ Accessible and discreet site of application ▪ Rapid drug delivery bypasses GI tract eliminating first-pass metabolism ▪ Short patch wear time (30 min) ▪ Band-Aid®-like ease of patch removal and convenient disposal 38

Copyright © 2022 Zosano Pharma Corporation Dosage Flexibility Enabled by Formulation, Process and Design 39 1. Formulation: Product formulation defines capability of coating on the microneedle substrate. Kept constant within a product family 2. Process: Multiple dips increase coated amount per microneedle - once passes (& coating morphology) are set, process is kept constant within a product family 3. Individual Microneedle design: Length, width, and features can be used to tune coated area – kept constant within a product family 4. Design of Array (Density): Density of microneedles can be optimized for the drug – kept constant across dosages within a product family 5. Design of Array (Size): Large patch templates can be produced to vary dosages across a product family - linear absorption demonstrated with 2cm2, 3cm2 & 5 cm2 in clinical studies 2cm2 3cm2 5.5 cm2 Formulation & Coating Low Dose 160 ug/3cm2 Higher Dose Up to 2 mg/3 cm2 Microneedle- array / Patch

Copyright © 2022 Zosano Pharma Corporation CORPORATE OVERVIEW 40

Copyright © 2022 Zosano Pharma Corporation Select Financials (unaudited) ▪ Cash and cash equivalents as of September 30, 2021: $17.1m ▪ Total shares outstanding as of November 5, 2021: 118.3m ▪ Select financial information: 41 Three Months Ended September 30, Nine Months Ended September 30, 2021 2020 2021 2020 (in millions, except per share data) Research & development expenses $6.0 $5.8 $16.3 $16.3 General & administrative expenses $2.5 $2.7 $8.3 $8.6 Net loss* $(8.7) $(8.7) $(23.0) $(25.3) Net loss per common share – basic and dilutive* $(0.07) $(0.11) $(0.21) $(0.45) Weighted-average shares 115.8 77.9 109.7 56.4 *Net loss and Net loss per common share – basic and diluted included a $1.6 million gain on forgiveness of our Paycheck Protection Loan for the nine months ended September 30, 2021

Copyright © 2022 Zosano Pharma Corporation Name Title Experience Steven Lo Chief Executive Officer Christine Matthews Chief Financial Officer Don Kellerman VP, Clinical Development & Medical Affairs Mahmoud Ameri VP, Research and Development Lu Liu VP, Technical Operations Greg Meyer VP, Quality Assurance & Regulatory Affairs Kristina Warga VP, Corporate Controller Experienced Management Team 42
Copyright © 2022 Zosano Pharma Corporation Board of Directors 43 Name Title Experience Steven Elms Managing Partner, Aisling Capital Linda Grais, MD, JD Director, Zosano Pharma Kenneth R. Greathouse Director, Zosano Pharma Joseph Hagan President and Chief Executive Officer, Regulus Therapeutics Inc. Steven Lo President and Chief Executive Officer, Zosano Pharma Kathy McGee Chief Operating Officer, AVITA Medical Elaine Yang Chief Revenue Officer, Lyra Health

44