Exhibit 99.1

1 RAFT meeting | 6 - 9 NOVEMBER 2022 Dalit Weinstein - Fischer

2 This communication is not a prospectus or offer of securities for subscription or sale in any jurisdiction. This communication contains forward - looking statements within the meaning of the Private Litigation Reform Act of 1995 . Words such as “ expect, ” “ believe, ” “ intend, ” “ plan, ” “ continue, ” “ may, ” “ will, ” “ anticipate, ” and similar expressions are intended to identify forward - looking statements . All statements, other than statements of historical facts, included in this communication regarding strategy, future operations, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward - looking statements . Examples of such statements include, but are not limited to, statements regarding execution of an additional agreement with the Max Planck Society and the University Medical Center Göttingen ; the therapeutic and commercial potential of nanosized antibodies ( NanoAbs ) ; and the timing of proof - of - concept studies and clinical trials in NanoAbs . These forward - looking statements reflect management ’ s current views with respect to certain current and future events and are subject to various risks, uncertainties and assumptions that could cause the results to differ materially from those expected by the management of BiondVax Pharmaceuticals Ltd . Risks and uncertainties include, but are not limited to, the risk that the therapeutic and commercial potential of NanoAbs will not be met ; the risk of a delay in the preclinical and clinical data for NanoAbs, if any ; the risk that BiondVax and EIB will not reach agreement with respect to the restructuring of the loan from European Investment Bank ; the risk that BiondVax may not be able to secure additional capital on attractive terms, if at all ; the risk that the European Investment Bank may accelerate the loans under its finance contract with BiondVax ; risks relating to the COVID - 19 (coronavirus) pandemic ; BiondVax ’ s ability to acquire rights to additional product opportunities ; BiondVax ’ s ability to enter into collaborations on terms acceptable to BiondVax or at all ; timing of receipt of regulatory approval of BiondVax ’ s manufacturing facility in Jerusalem, if at all or when required ; the risk that the manufacturing facility will not be able to be used for a wide variety of applications and other vaccine and treatment technologies, and the risk that drug development involves a lengthy and expensive process with uncertain outcomes . More detailed information about the risks and uncertainties affecting the Company is contained under the heading “ Risk Factors ” in the Company's Annual Report on Form 20 - F filed with the Securities and Exchange Commission on March 28 , 2022 . BiondVax undertakes no obligation to revise or update any forward - looking statement for any reason . SAFE HARBOR STATEMENT

3 C OLLABORATION WITH M AX P LANCK & UMG • World - class science & access to leading scientists • NanoAb platform for development of promising potent therapeutics • Patents covering NanoAbs & their manufacturing • R&D knowledge, capabilities • Biosafety level 3 lab BIONDVAX MAX PLANCK & UMG Capabilities to develop significant clinical and commercial advantages • Manufacturing, quality, int ’ l regulatory experience • GMP NanoAb manufacturing facility • Top - tier big pharma & biotech leadership expertise Exclusive worldwide license for COVID - 19 asset & option to exclusively license additional NanoAbs developed through collaboration

4 T HE SCIENCE OF NANOSIZED ANTIBODIES ( N ANO A BS ) * Nanobody is a trademark by ABLYNX N. V., a wholly owned subsidiary of Sanofi. BiondVax has no affiliation with and is not endorsed by Sanofi. HUMAN ANTIBODY ALPACA - DERIVED NANOAB Alpaca - derived nanosized antibodies (NanoAbs) are also known as nanobodies * Heavy Chain Only Antibody

5 A GLANCE INTO THE COMPLEXITY OF PRODUCING A STRONG N ANO A BS VHH Neutralization test GOOD RESULTS Relevant Lead in E.coli Relevant Lead in P. Pastoris Efficiency test Clone production and selection

6 W HY N ANO A BS ARE BIO - BETTER ‣ Choice from 10 8 library ‣ Extreme affinities ‣ Neutralizing receptor binding ‣ Extreme thermostability ‣ Rapid development ‣ Affordable production ‣ Flexible engineering to meet PK requirementsAR0

7 D E - RISKED N ANO A B TARGET CHOICE – FAVORABLE PATH TO MARKET MOLECULAR TARGET MECHANISM OF ACTION COMPOSITION OF MATTER COMMERCIAL ض ض + TBD Validated by existing mAb therapies Well understood Assessing safety & efficacy of alpaca - derived NanoAbs Solid demand for available mAb therapies VALIDATED THERAPEUTIC USE First commercial NanoAbs is blood disorder therapy Caplacizuma – by Ablynx , acquired by Sanofi in 2018 for $ 4.8 B AR0

8 Shorter R&D and regulatory pathways V ALIDATED EXISTING M A B TREATMENT SUPPORT N ANO A BS TARGETS CHOICE - sars - anti products/ - antibody - 2 - cov - sars - anti therapies/ gov/ nih. treatmentguidelines. 19 covid www. https:// : Rxs COVID mAbs – More info / antibodies - monoclonal - 2 - cov Asthma mAb therapies include: • Nucala (GSK) • CINQAIR (Teva) • Fasenra (AZ) COVID mAb • Bamlanivimab / etesev imab (Lilly) • Casirivimab (REGEN - COV, Regeneron) • Sotrovimab ( Xuvedy , GSK - Vir ) • Evusheld (AZ) Psoriasis & Psoriatic Arthritis: Humira (AbbVie) for rheumatoid arthritis (RA), psoriatic arthritis, plaque, and other indications.

9 C REATING AN INNOVATIVE N ANO A BS PIPELINE Extending from a promising COVID - 19 therapeutic… • Validated targets of existing mAb treatments • Short time to value generation, lower risk than mAbs • Large markets growing at attractive CAGRs COVID 19 Lead candidate demonstrating strong competitive edge ( 1 ) ASTHMA PSORIASIS PSORIATIC ARTHRITIS MACULAR DEGEN …to multiple, significant de - risked opportunities 1 . Based on laboratory research at Max Planck Institute for Biophysical Chemistry (MPG) and University Medicine Göttingen (UM G);

10 C OVID - 19 MARKET CONTINUES TO GROW -- AUTHORED BY THE SCIENTIFIC LEADS OF OUR MAX PLANCK COLLABORATION ( 3 ) Initial waves Jan 2020 Jun Jan 2021 Jun Jan 2022 1 . New England Journal of Medicine. https://www.nejm.org/doi/full/ 10.1056 /NEJMp 2118468 . | 2 . WHO “ SARS - CoV - 2 is unlikely to be eliminated, let alone eradicated; it will probably continue to circulate indefinitely in periodic outbreaks and endemics. -- ANTHONY FAUCI, December 2021 . ( 1 ) Delta Omicron Worldwide COVID - 19 Cases REPORTED DAILY AS OF November 2 nd , 2022 ( 2 ) BA. 4 / 5

11 I NHALED N ANO A BS FOR C OVID TREATMENT

12 To date, there are limited therapeutic options to treat mild to moderate COVID - 19 • Our NanoAb is engineered to act against the SARS - CoV - 2 receptor binding domain (RBD) • By blocking the surface spike protein/RBD, the virus is neutralized • NanoAbs show high affinity and thermostability • The NanoAb is developed as inhalable therapy using a nebulizer device T ARGETING SARS - C O V - 2 WITH N ANO A BS

13 E XTREME VIRUS - NEUTRALIZING POTENCY EXPLAINED BY X - RAY STRUCTURE NanoAb RBD (surface representation) NanoAb RBD cartoon surface NanoAb RBD & ACE2 Frontal clashes with RBD - bound ACE 2

14 E XTREME CROSS - STRAIN POTENCY OF THE N ANO A BS 72 h Infection + NanoAb NanoAb Concentration I NFECTION AND TREATMENT OF V ERO CELLS N EUTRALIZATION AT P ICOMOLAR RANGE W IDE COVERAGE
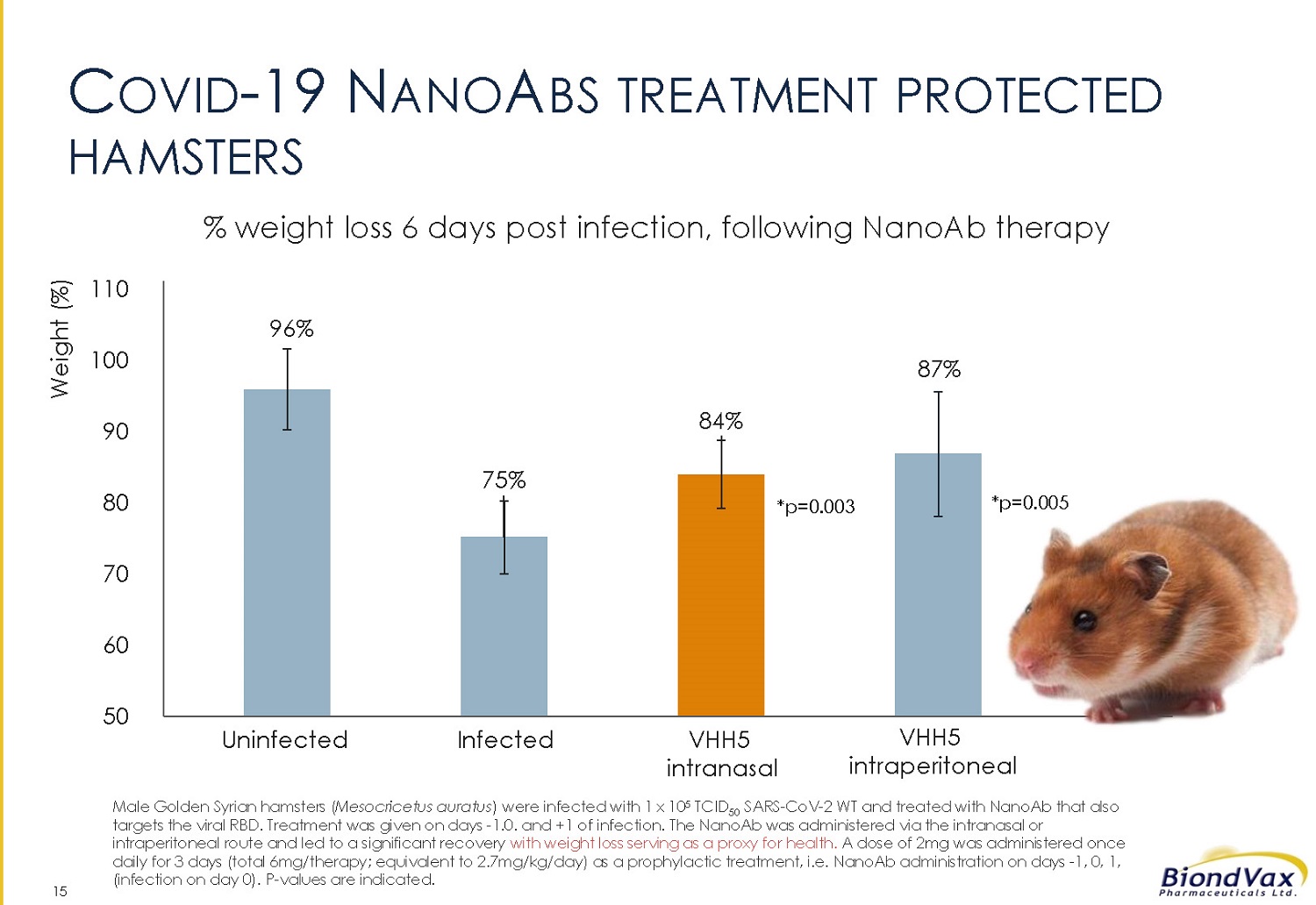
15 96 % 75 % 84 % 87 % 50 60 70 80 90 100 110 Weight (%) *p= 0.003 *p= 0.005 Uninfected Infected VHH5 intranasal VHH 5 intraperitoneal C OVID - 19 N ANO A BS TREATMENT PROTECTED HAMSTERS % weight loss 6 days post infection, following NanoAb therapy Male Golden Syrian hamsters ( Mesocricetus auratus ) were infected with 1 x 10 5 TCID 50 SARS - CoV - 2 WT and treated with NanoAb that also targets the viral RBD. Treatment was given on days - 1.0 . and + 1 of infection. The NanoAb was administered via the intranasal or intraperitoneal route and led to a significant recovery with weight loss serving as a proxy for health. A dose of 2 mg was administered once daily for 3 days (total 6 mg/therapy; equivalent to 2.7 mg/kg/day) as a prophylactic treatment, i.e. NanoAb administration on days - 1 , 0 , 1 , (infection on day 0 ). P - values are indicated.

16 B IOND V AX ’ S PROJECT AND RESULTS

17 B IOND V AX ’ S GMP M ANUFACTURING AND R&D F ACILITY • Equipped to produce recombinant protein products such as NanoAb • Single - use equipment enables: - Adaptable manufacturing processes for the entire NanoAb pipeline - Quicker lead times - Faster time - to - market for new products • Designed to meet FDA and EMA reqs & approved cGMP by Israel MoH & EU QP BiondVax ’ s GMP Biologics Manufacturing Facility | Jerusalem Well - suited for NanoAb drug development, clinical supplies AND commercial production

18 L ARGE - S CALE PRODUCTION IN P ICHIA P ASTORIS • Fed - Batch fermentation: up to 10 gr/L culture (pending on P.P strain) • Kg yields in a standard 1000 L fermenter NanoAb 10 15 20 25 30 40 50 60 70 85 100 kDa 0.5 µl Supernatant

19 - 70 C O.N growth Centrifuge Supernatant Carbon Source Glycerol/Methanol 120 hr process U PSTREAM D OWNSTREAM Clarification 1 d Chromatography 2 d Buffer exchange & concentration 1d Filtration 1 d P RODUCTION PROCESS FLOW

20 N ANO A BS ACCUMULATION THROUGHOUT THE FERMENTATION – L OW HCP CONTENT IPC - SDS Page assay 95 h post induction 98 h post induction 115 h post induction Size Marker Size Marker NanoAb

21 S TRAIGHT FORWARD DSP O PTIMIZED PURIFICATION PROCESS WITH ~ 75 % EFFICIENCY Filtration Clarification Chromatography Formulation Size removal of large particles, impurities, aggregates, etc.) Size removal of small impurities, media components, etc. and buffer exchange Purification according to VHH protein properties Reaching VHH concentration and buffer)

22 This communication is not a prospectus or offer of securities for subscription or sale in any jurisdiction. 2 2 H IGH PURITY OF FINAL PRODUCT Harvest (pool) Main peak Impurities Final protein RP - HPLC R ESULTS • Downstream process efficiency ~ 75 % • Purity (HPLC - RP) ~ 98 %

23 S TABILITY STUDY – T HE ACTIVITY OF OUR N ANO A BS IS UNAFFECTED FOLLOWING 60 DAYS AT 25 C

24 I NHALED ROUTE OF ADMINISTRATION & CONSIDERATIONS OF DEVICE CHOICE

25 N OT ALL INHALER TECHNOLOGIES SUIT BIOLOGICAL MOLECULES • S UITABLE FOR L ARGE BIOLOGICAL MOLECULES ( PROTEINS ) • L OW PRESSURE • R EDUCE SHEAR FORCE • T YPE OF MIST PLUME GENERATED BY THE DEVICE • A DJUSTED FORMULATION DEVELOPMENT TO KEEP STABILITY AT HIGH CONCENTRATIONS • S INGLE - USE INHALER FOR C OVID TREATMENT ( ACUTE AND INFECTIOUS ) • L OW - COST DEVICE FOR SHORT TREATMENT

26 S UMMARY

27 K EY M ESSAGES : • D EVELOP SELF INHALED N ANO A BS TREATMENT FOR COVID - 19 • F IRST I N H UMAN CLINICAL TRIAL PLANNED FOR Q 3 2023 • P ICHIA P ASTORIS EXPRESSION SYSTEM • T ECHNOLOGY PLATFORM FOR ADDITIONAL N ANO A BS TARGETING OTHER DISEASES SUCH AS A STHMA AND PSORIASIS • CUTTING - EDGE TECH GMP MANUFACTURING FACILITY • EXTRA CAPACITY TO PROVIDE CDMO SERVICES

28 T HANK YOU BIONDVAX.COM Contact: Dalit Weinstein Fischer, VP TRD dalit@biondvax.com