| © 2022 Zynerba Pharmaceuticals, Inc. All rights reserved. NASDAQZYNE Advancing Science.Improving Connections.Next-Generation Transdermal Cannabinoid TherapeuticsNovember 2022 |
| Forward-Looking StatementsThe statements in this presentation may include forward-looking statements within the meaning of the private securities litigation reform act of 1995. These statements, among other things relate to the future operations, opportunities or financial performance of Zynerba pharmaceuticals, inc. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from the company’s current expectations, including the following: the company’s cash and cash equivalents may not be sufficient to support its operating plan for as long as anticipated; the results, cost and timing of the company’s clinical development programs, including any delays to such clinical trials relating to enrollment or site initiation;clinical results for the company’s product candidates may not be replicated or continue to occur in the company’s ongoing or planned clinical trials in FXS, 22q, or ASD, or in any additional trials, and may not otherwise support further development in a specified indication or at all;the company’s planned reconnect trial may not be determined to be sufficient to support a submission for regulatory approval, including an ndaor maa; actions or advice of the U.S. Food and drug administration and foreign regulatory agencies may affect the design, initiation,timing, continuation and/or progress of clinical trials or result in the need for additional clinical trials; the company’s ability to obtain and maintain regulatory approval for its product candidates, and the labeling under any such approval; the company’s expectations regarding its ability to obtain and adequately maintain sufficient intellectual property protection for its product candidates. These and other risks are described in our filings with the securities and exchange commission, available at www.sec.gov. Any forward-looking statements that the company makes in this presentation speak only as of the date of this presentation. The company assumes no obligation to update forward-looking statements whether as a result of new information, future events or otherwise, after the date of this presentation.November 2022Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 2 © 2022 Zynerba Pharmaceuticals, Inc. All rights reserved. Zynerba and Zygel are trademarks of Zynerba Pharmaceuticals, Inc. All other trademarks and registered trademarks are property of their respective owners. trademarks are property of their respective owners. |
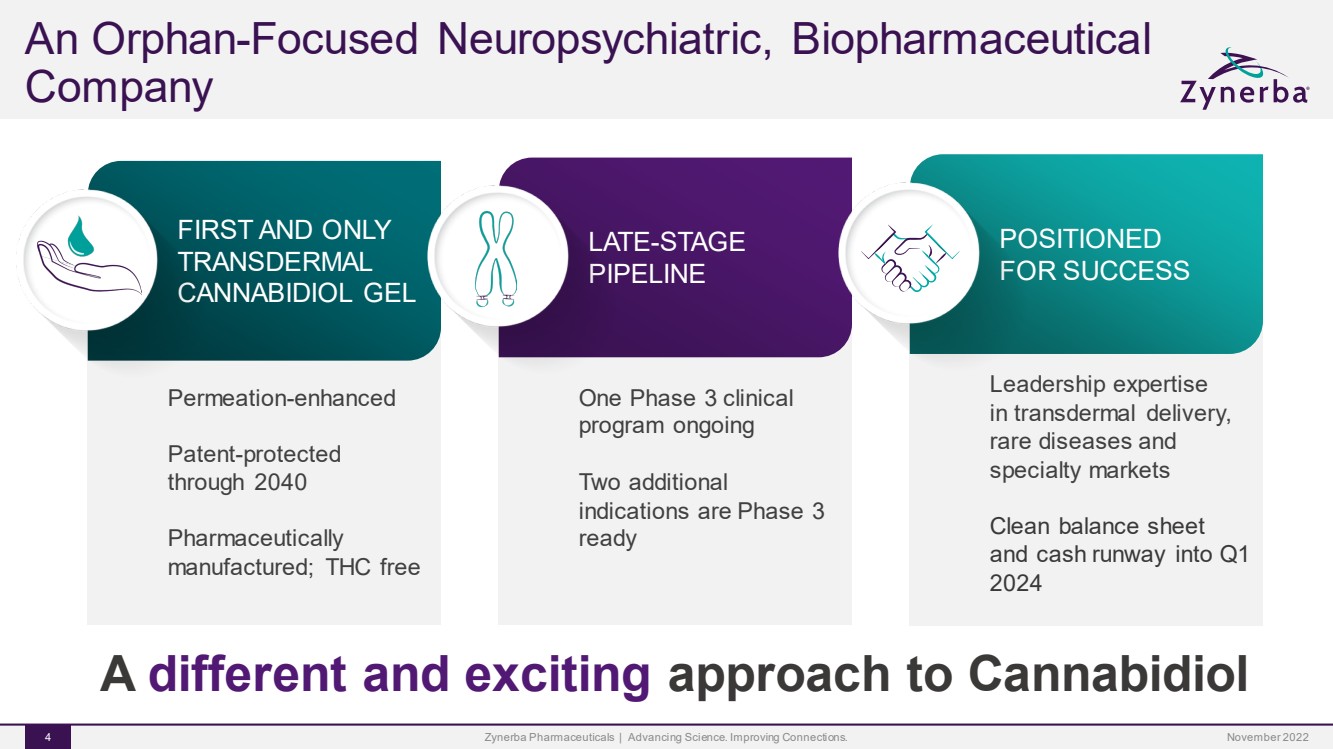
| A different and exciting approach to Cannabidiol |
| An Orphan-Focused Neuropsychiatric, Biopharmaceutical CompanyZynerba Pharmaceuticals | Advancing Science. Improving Connections. 4 Permeation-enhancedPatent-protected through 2040Pharmaceutically manufactured; THC free FIRST AND ONLY TRANSDERMAL CANNABIDIOL GEL One Phase 3 clinical program ongoingTwo additional indications are Phase 3 ready LATE-STAGE PIPELINE Leadership expertise in transdermal delivery, rare diseases and specialty marketsClean balance sheet and cash runway into Q1 2024 POSITIONED FOR SUCCESS A differentand exciting approach to CannabidiolNovember 2022 |
| Benefits of Our Approach to Cannabidiol Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 5 FDA regulated Consistency of production Purity of ingredients No THC –not a scheduled drug by U.S. DEA Scalable production process PHARMACEUTICAL MANUFACTURINGPHARMACEUTICALDISPENSARY Ease of application for caregivers of patients with behavioral issues Minimizes GI side effects and reduces risk for liver toxicity Lower risk for drug/drug interactions Avoids conversion to THC in stomach TRANSDERMAL DELIVERY November 2022 |
| Clinical Development Programs Fragile X SyndromeFXS 22q Deletion Syndrome22q Autism Spectrum DisorderASD |
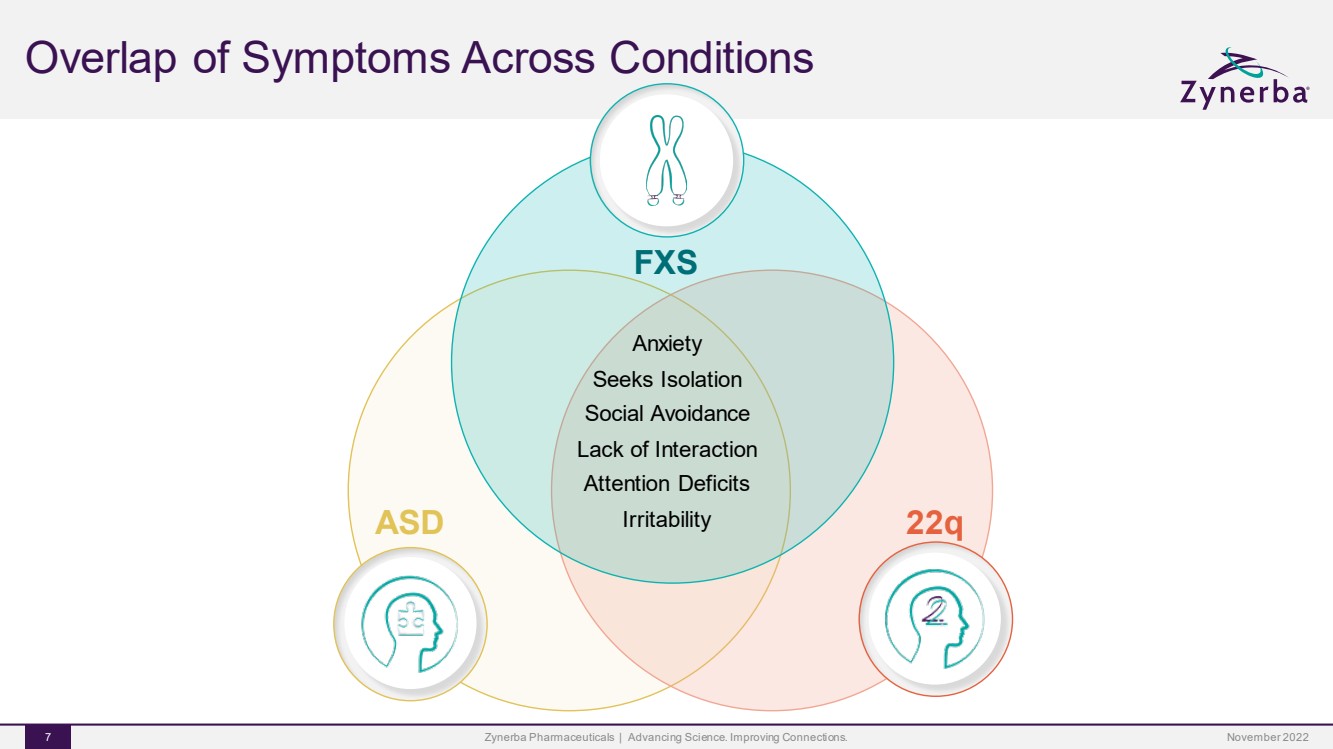
| Overlap of Symptoms Across ConditionsZynerba Pharmaceuticals | Advancing Science. Improving Connections. 7 AnxietySeeks IsolationSocial AvoidanceLack of InteractionAttention DeficitsIrritability FXS22qASDNovember 2022 |
| Fragile X Syndrome (FXS)Focused Clinical Pipeline Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 8 PreclinicalPhase 1Phase 2PivotalMarket 22q Deletion Syndrome (22q) Autism Spectrum Disorder (ASD) Regulatory FilingU.S. Orphan Drug and Fast Track designations; EU Orphan Drug designationU.S. Orphan Drug designation; EU Orphan Drug designation ZygelTM(ZYN002 Cannabidiol Gel) Initiation of Phase 3 Trial DeferredNovember 2022 |
| •Zygel safety database across all clinical studies includes data from over 900 volunteers and patients•Majority of treatment-emergent AEs (TEAE) were mild or moderate •Most common Zygel-related TEAE are application site events, the majority of which were mild and transient •No clinically significant changes in vital signs or ECGs•No Zygel-related clinically significant changes in laboratory values, including liver function testsZynerba Pharmaceuticals | Advancing Science. Improving Connections. 9 Well-Tolerated Safety Profile November 2022 |
| Fragile X Syndrome |
| ~78K U.S. PATIENTSWhat is Fragile X Syndrome (FXS)Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 11 •Leading known cause of inherited intellectual disability and autism spectrum disorder•Mutation of the FMR1gene causes endocannabinoid system (ECS) dysregulation•Easily identified mutation manifests as multiple CGG repeats onFMR1(full mutation >200 repeats)•Resulting in cognitive, social, and behavioral symptoms•Behavioral Symptoms linked to deficiencies in the ECSWITH FXS EU/UK PATIENTS~121KNovember 2022U.S. & EU Orphan Drug Designation Granted for FXS |
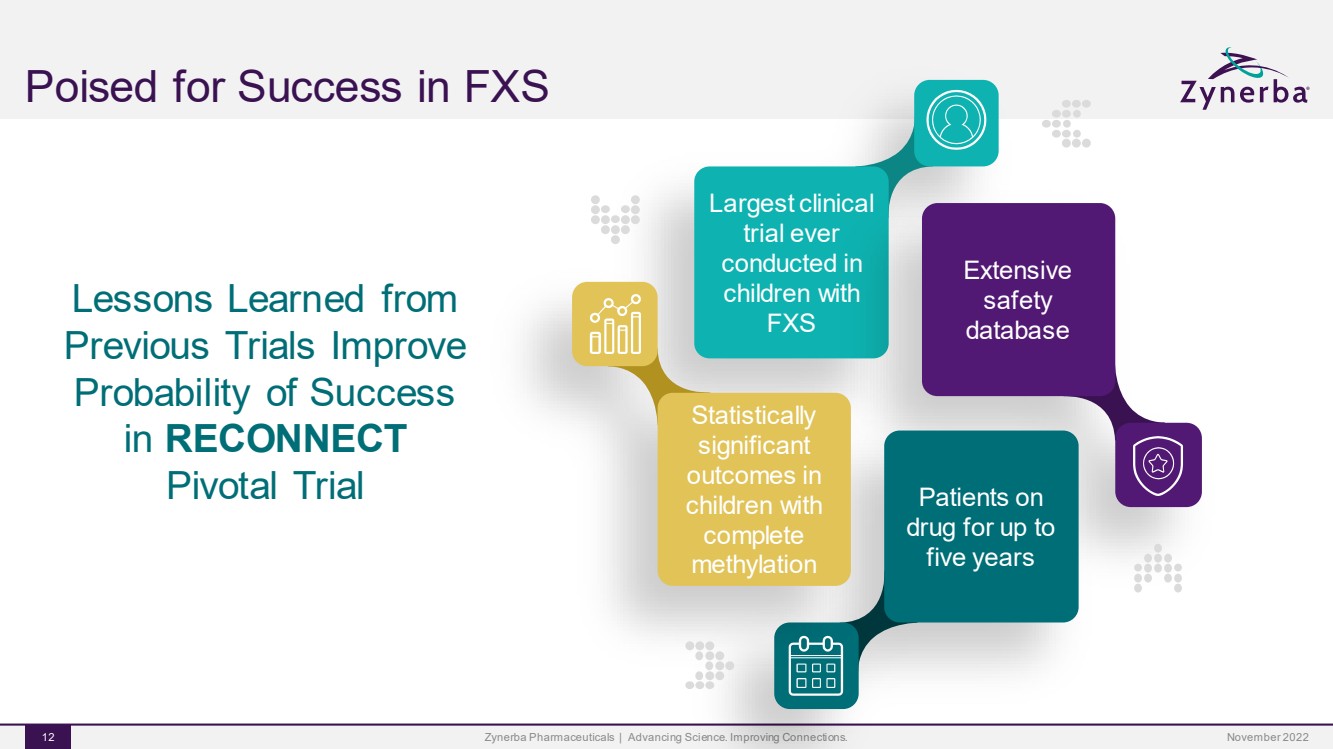
| Poised for Success in FXSZynerba Pharmaceuticals | Advancing Science. Improving Connections. 12 Largest clinical trial ever conducted in children with FXS Extensive safety database Patients on drug for up to five years Statistically significant outcomes in children with complete methylation Lessons Learned from Previous Trials Improve Probability of Success in RECONNECTPivotal Trial November 2022 |
| Role of FMR1Methylation in FXSZynerba Pharmaceuticals | Advancing Science. Improving Connections. 13 •FMR1gene codes for production of FMRP* which is vital to synapse development•Methylation of the FMR1gene plays a role in determining gene function•When methylation of the FMR1gene silences the gene, no FMRP is produced: •Systems and processes affected by FMRP become dysregulated•~60% of FXS patients are believed to be completely methylated•Completely methylated patients are the most severely impacted~47K U.S. PATIENTSWITH COMPLETEMETHYLATION EU/UK PATIENTS~73K*RNA-binding protein that helps regulate synaptic development and plasticityNovember 2022 |
| CONNECT-FXTrialKey Learning: Results with complete methylation of FMR1 geneConsistent Improvements Observed with Zygel vs. Placebo in Patients with Complete MethylationZynerba Pharmaceuticals | Advancing Science. Improving Connections. 14 PRIMARY ENDPOINT CAREGIVER REPORTED BEHAVIOR CHANGE CLINICIAN REPORTED BEHAVIOR IMPROVEMENTS CLINICALLY MEANINGFUL BEHAVIOR IMPROVEMENTS ABC-CFXSSocial Avoidance Subscale Caregiver Global Impression of Change (ZYGEL vs Placebo) Clinical Global Impression of Improvement (anchored)** More Patients Achieved Meaningful Change (ZYGEL vs Placebo)Ad hoc analysis of 136 patients with complete methylation*Statistically significant , **Not specific to Social Avoidance40% median percent improvement in socially avoidant behaviors (p=0.027*)SOCIAL INTERACTION 63% vs 37% (p=0.005*)IRRITABLE/DISRUPTIVE BEHAVIORS54% vs 33% (p=0.027*)SOCIAL AVOIDANCE/ISOLATION58% vs 46% (p=0.195)OVERALL BEHAVIOR61% vs 46% (p=0.100)ANY IMPROVEMENT Zygel vs placebo 50% vs 36%(p=0.128)SOCIAL AVOIDANCE (≥ 3 POINTS)56% vs 37% (p=0.030*)IRRITABILITY (≥ 9 POINTS) 37% vs 26% (p=0.232)November 2022 |
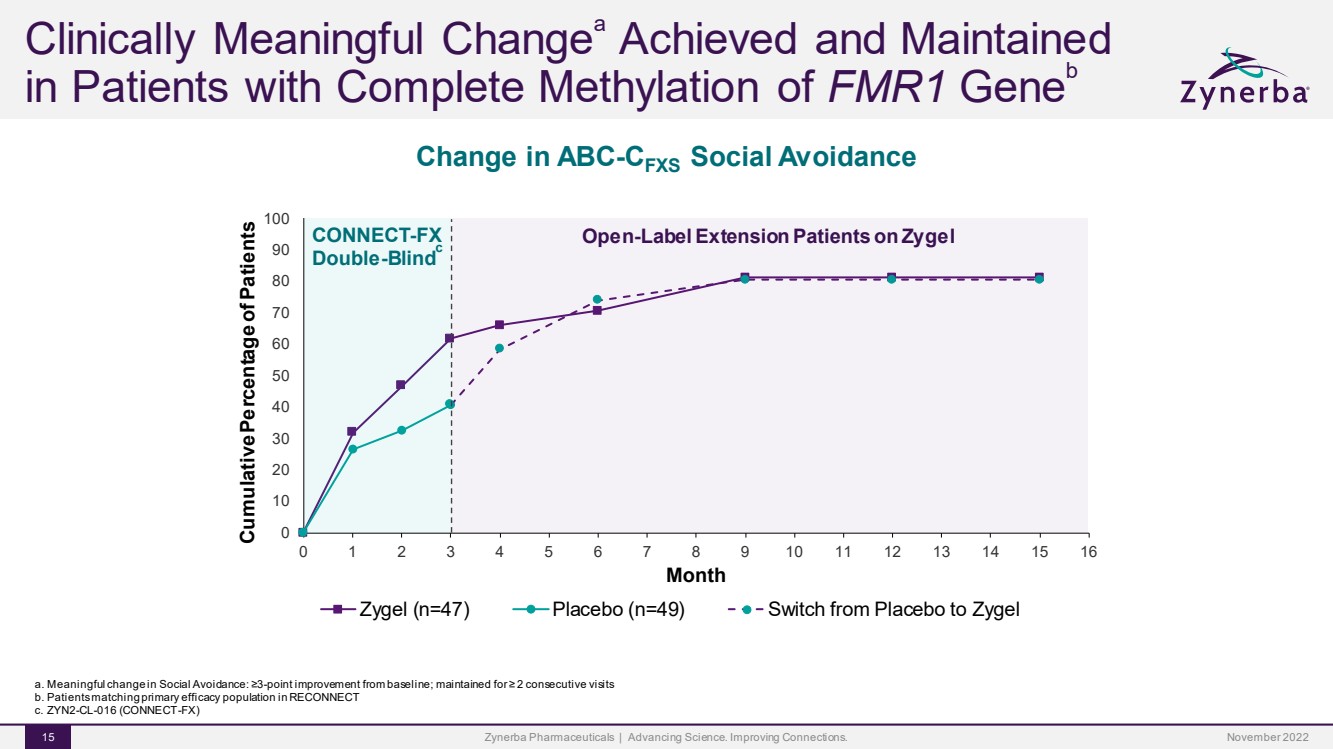
| Change in ABC-CFXSSocial Avoidance Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 15 Clinically Meaningful ChangeaAchieved and Maintained in Patients with Complete Methylation of FMR1Geneb 01020 3040506070 8090100012345678910111213141516 Cumulative Percentage of Patients Month Zygel (n=47) Placebo (n=49) Switch from Placebo to Zygel CONNECT-FXDouble-Blindc Open-Label Extension Patients on Zygel a. Meaningful change in Social Avoidance: ≥3-point improvement from baseline; maintained for ≥ 2 consecutive visitsb. Patients matching primary efficacy population in RECONNECTc. ZYN2-CL-016 (CONNECT-FX) November 2022 |
| Design Optimized from CONNECT-FX TrialZynerba Pharmaceuticals | Advancing Science. Improving Connections. 16 More patient and family friendlyExtending trial to 18-weeksIncreased dosing option for individuals >50 kgSuccessful completion of Phase 3 pivotal trial expected to satisfy requirements for an NDA submission in the U.S. and a marketing authorization application in the EU. Primary endpoint: Patients with complete methylationNovember 2022 |
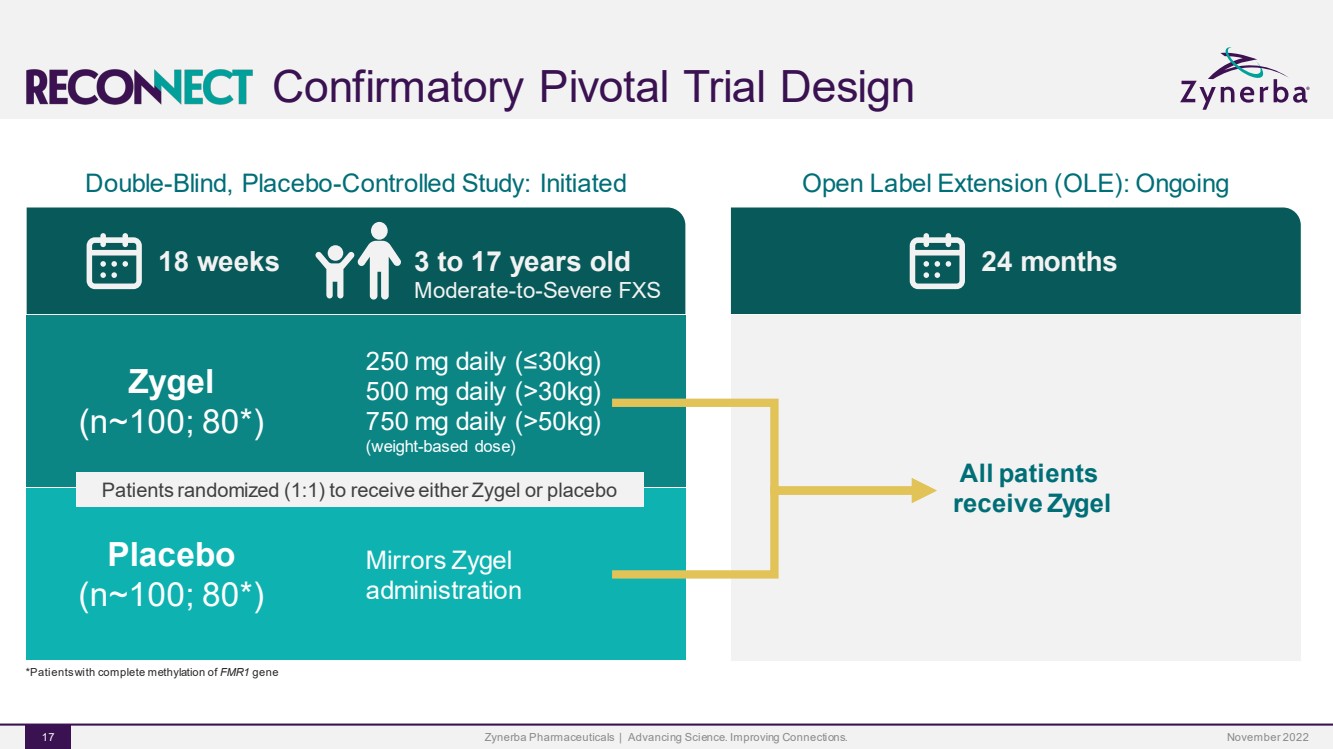
| Confirmatory Pivotal Trial DesignZynerba Pharmaceuticals | Advancing Science. Improving Connections. 17 Double-Blind, Placebo-Controlled Study: Initiated 18 weeks 3 to 17 years oldModerate-to-Severe FXS Zygel (n~100; 80*)250 mg daily (≤30kg)500 mg daily (>30kg)750 mg daily (>50kg)(weight-based dose) Placebo (n~100; 80*)Mirrors Zygel administration*Patients with complete methylation of FMR1gene Patients randomized (1:1) to receive either Zygel or placeboOpen Label Extension (OLE): Ongoing 24 months All patients receive Zygel November 2022 |
| Trial ObjectivesZynerba Pharmaceuticals | Advancing Science. Improving Connections. 18 Change from baseline to end of treatment in ABC-CFXS Social Avoidance subscale in patients who have complete (100%) methylation of their FMR1gene •Change from baseline to end of treatment in:•ABC-CFXS Irritability subscale in patients who have complete methylation•ABC-CFXSSocial Avoidance subscale among all randomized patients (complete and partial methylation)•Percent of patients:•Any improvement on the Caregiver Global Impression of Change (CaGI-C) for Social Interactions among patients with complete methylation•Rated as improved on the Clinical Global Impression-Improvement (CGI-I) scale among patients with complete methylation SECONDARY ENDPOINTS PRIMARY ENDPOINT November 2022 |
| Next Steps in FXS Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 19 Continued RECONNECTpivotal trial enrollmentTopline results expected in 2H 2023November 2022 |
| 22q11.2 Deletion Syndrome (22q)Image courtesy Positive Exposure © 2016 Positive Exposure. All rights reserved. www.PositiveExposure.org ..Eliza, living with 22q11.2 deletion syndrome |
| What is 22q11.2 Deletion Syndrome (22q)?Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 21 •Rare disorder and second most common genetic disorder, behind Down syndrome•Midline condition with abnormalities affecting palate, face, heart and other organs; surgically corrected in infancy•Neuropsychiatric illnesses and learning disabilities common•Early onset of neuropsychiatric symptoms disrupts development and quality of life, and heightens risk of later psychotic disorders•No drugs currently approved WITH 22q~83K U.S. PATIENTS~129K EU/UK PATIENTSNovember 2022 |
| U.S. & EU ORPHAN DRUG DESIGNATION GRANTED FOR TREATMENT OF 22qRationale for Zygel in 22qZynerba Pharmaceuticals | Advancing Science. Improving Connections. 22 •Overlapping symptoms with FXS and ASD•Associated with increased anxiety, irritability, social withdrawal and social interaction problems •Cannabidiol may treat neuropsychiatric symptoms due to activity as: •Modulator of ECS•Agonist at serotonin1Areceptors•Antagonist at GPR55 receptorsNovember 2022 |
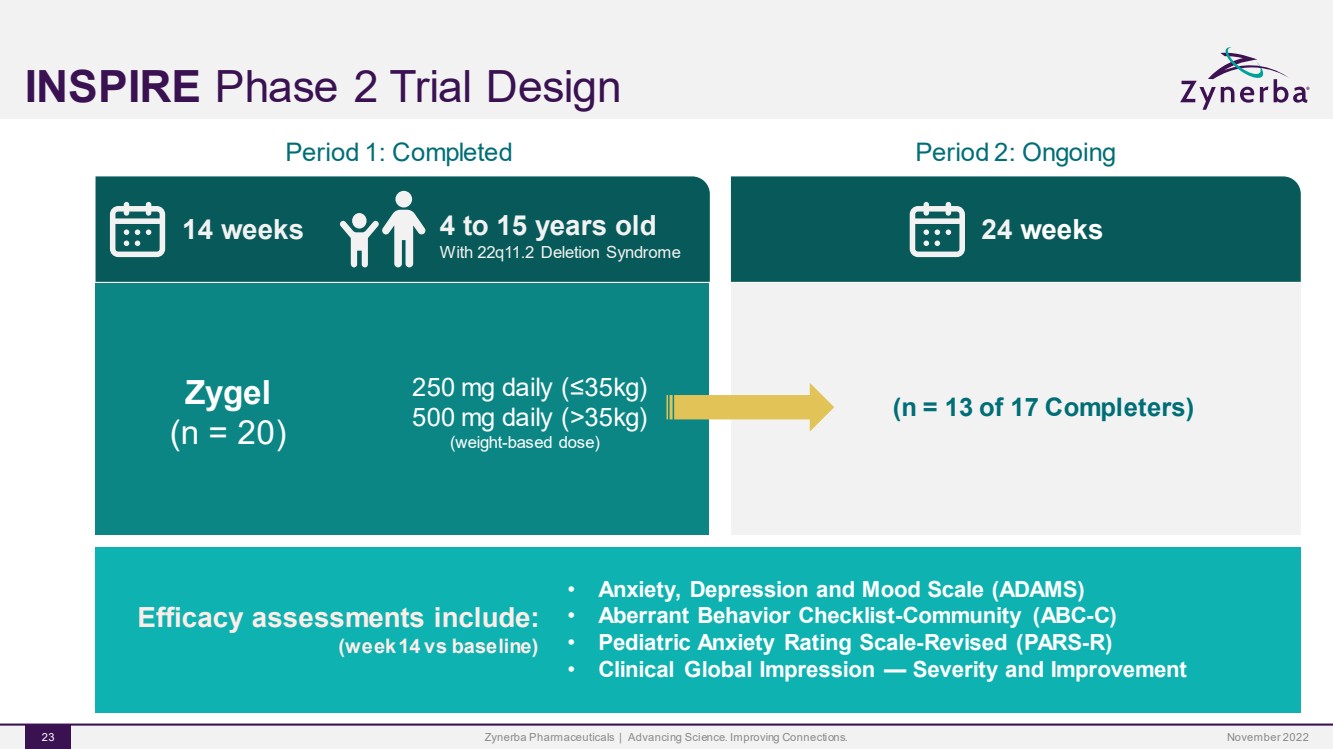
| INSPIREPhase 2 Trial DesignZynerba Pharmaceuticals | Advancing Science. Improving Connections. 23 Period 1: Completed 14 weeks 4 to 15 years oldWith 22q11.2 Deletion Syndrome Zygel(n = 20)250 mg daily (≤35kg)500 mg daily (>35kg)(weight-based dose)Period 2: Ongoing 24 weeks Efficacy assessments include:(week 14 vs baseline) •Anxiety, Depression and Mood Scale (ADAMS)•Aberrant Behavior Checklist-Community (ABC-C)•Pediatric Anxiety Rating Scale-Revised (PARS-R) •Clinical Global Impression —Severity and Improvement (n = 13 of 17 Completers)November 2022 |
| •Statistically significant improvements at 14 weeks of treatment compared to baseline for multiple efficacy assessments:•The total score and all five subscales of the Anxiety, Depression and Mood Scale (ADAMS)•All five subscales of the Aberrant Behavior Checklist –Community (ABC-C)•Pediatric Anxiety Rating Scale -Revised (PARS –R) •The majority of patients showed clinically meaningful improvements at week 14 as demonstrated by the Clinical Global Impression –Improvement (CGI-I) •Seventy-five percent of patients were rated by the clinicians as “improved”, “much improved” or “very much improved” •Nearly two-thirds (62.5%) of the patients being “much improved” or “very much improved” •Zygel was shown to be well tolerated, and the safety profile was consistent with previously released data from other Zygel clinical trials•Three patients reported treatment related adverse events which were all mild application site adverse events•One patient discontinued treatment due to adverse events not related to Zygel 24 Positive Topline Results from INSPIRE TrialZynerba Pharmaceuticals | Advancing Science. Improving Connections.November 2022 |
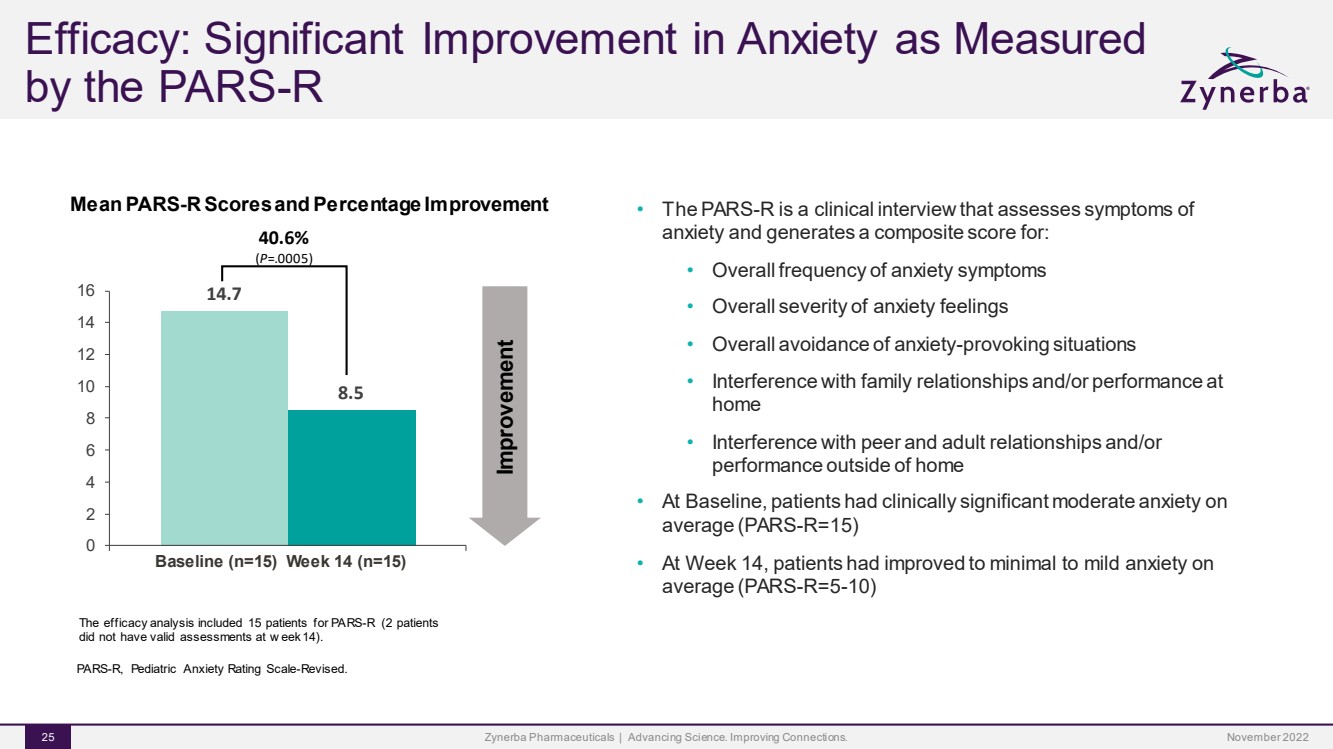
| Efficacy: Significant Improvement in Anxiety as Measured by the PARS-RZynerba Pharmaceuticals | Advancing Science. Improving Connections. 25 Mean PARS-R Scores and Percentage ImprovementPARS-R, Pediatric Anxiety Rating Scale-Revised. 14.78.50246 8101214 1640.6%(P=.0005) ImprovementBaseline (n=15)Week 14 (n=15)The efficacy analysis included 15 patients for PARS-R (2 patients did not have valid assessments at week 14).•The PARS-R is a clinical interview that assesses symptoms of anxiety and generates a composite score for:•Overall frequency of anxiety symptoms•Overall severity of anxiety feelings•Overall avoidance of anxiety-provoking situations•Interference with family relationships and/or performance at home•Interference with peer and adult relationships and/or performance outside of home•At Baseline, patients had clinically significant moderate anxiety on average (PARS-R=15)•At Week 14, patients had improved to minimal to mild anxiety on average (PARS-R=5-10)November 2022 |
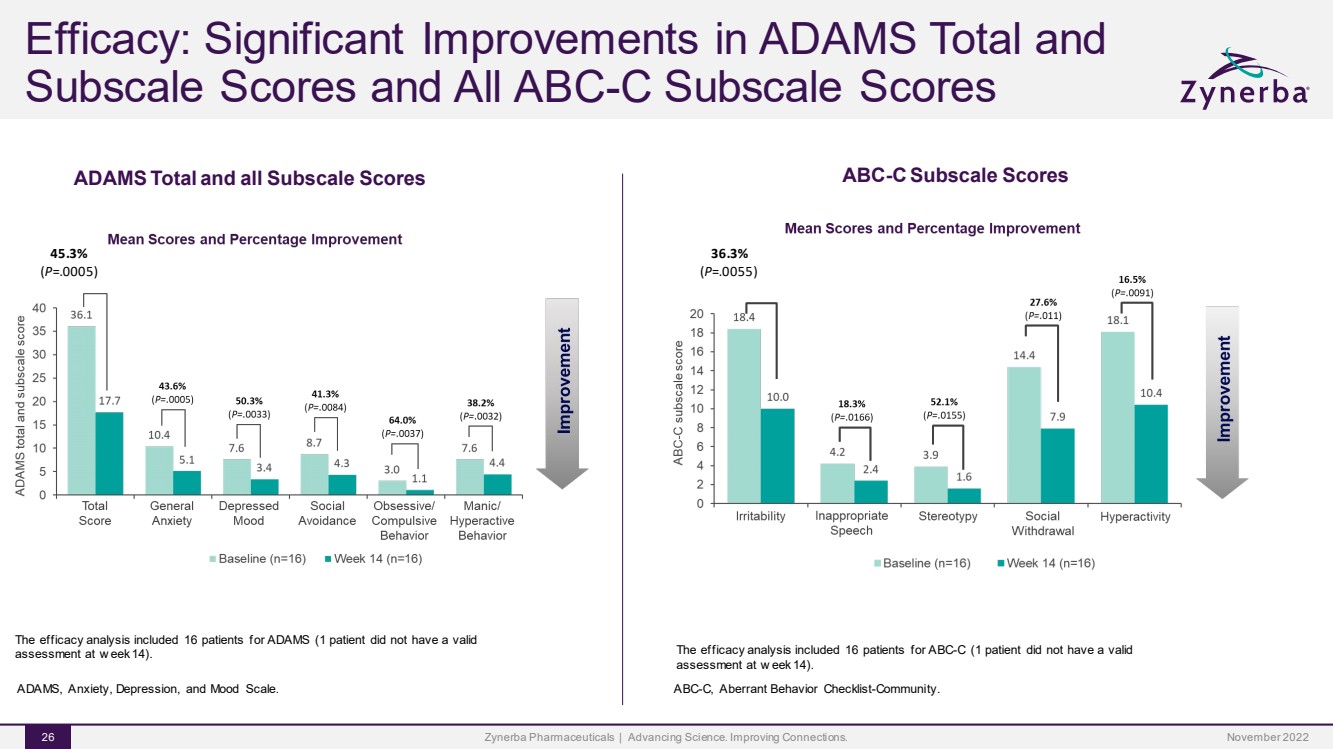
| Efficacy: Significant Improvements in ADAMS Total and Subscale Scores and All ABC-C Subscale ScoresZynerba Pharmaceuticals | Advancing Science. Improving Connections. 26 ADAMS, Anxiety, Depression, and Mood Scale.45.3%(P=.0005)The efficacy analysis included 16 patients for ADAMS (1 patient did not have a valid assessment at week 14). ADAMS Total and all Subscale ScoresABC-C Subscale Scores36.3%(P=.0055)The efficacy analysis included 16 patients for ABC-C (1 patient did not have a valid assessment at week 14).ABC-C, Aberrant Behavior Checklist-Community. November 2022 |
| Next Steps in 22qZynerba Pharmaceuticals | Advancing Science. Improving Connections. 27 Discuss regulatory pathway with FDA Initiate Phase 3 program after FDA discussions and RECONNECT toplineresultsNovember 2022 |
| Autism Spectrum Disorder |
| Rationale for Zygel in Autism Spectrum Disorder (ASD)Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 29 •Results from FXS trials suggested spectrum of activity against behaviors also seen in ASD: irritability, social avoidance and anxiety•Studies suggested ASD is linked to disruption of the ECS•Altered anandamide* signaling may contribute to ASD-related social and communication impairments•The ECS modulates many cellular functions and molecular pathways altered in ASD •Cannabidiol may modulate the ECS and improve certain autism-related behaviors WITH ASD U.S. CHILDREN AND ADOLESCENTS~1.4M* Anandamide is one of two primary endocannabinoidsNovember 2022 |
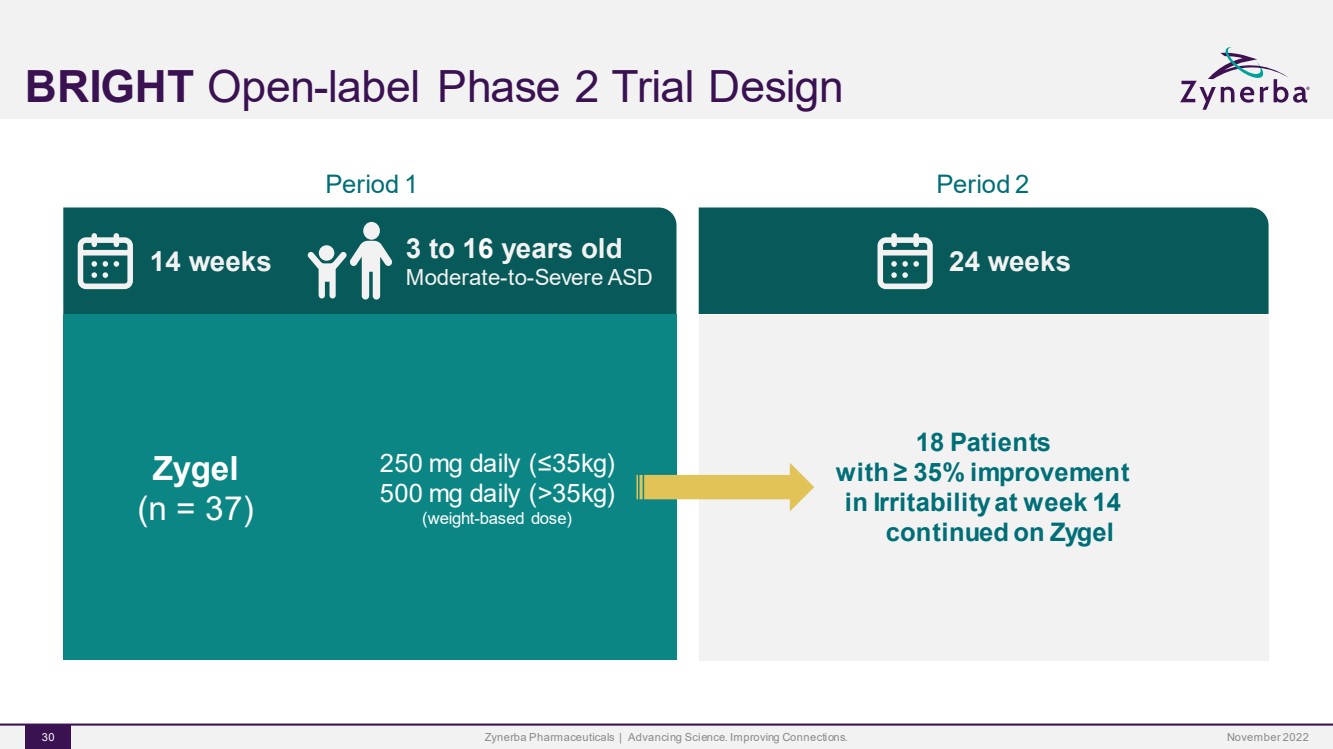
| BRIGHTOpen-label Phase 2 Trial DesignZynerba Pharmaceuticals | Advancing Science. Improving Connections. 30 Period 1 14 weeks 3 to 16 years oldModerate-to-Severe ASD Zygel(n = 37)250 mg daily (≤35kg)500 mg daily (>35kg)(weight-based dose)Period 2 24 weeks 18 Patientswith ≥ 35% improvementin Irritability at week 14continued on ZygelNovember 2022 |
| BRIGHTPhase 2 Trial Results: Period 1Statistically Significant Results at Week 14 Compared to Baseline (Completers in Efficacy Population n=28†)Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 31 †n=26 for ABC-C inappropriate speech; *Statistically significant; Full data available in May 26, 2020 and October 15, 2020 press releases Irritability: 39%(p<0.0001*)Inappropriate Speech: 43%(p=0.0002*)Stereotypy: 39%(p<0.0001*)Social Withdrawal: 36%(p<0.0001*)Hyperactivity: 36% (p<0.0001*) Atypical behavior: 34%(p<0.001*)Communication: 20%(p<0.001*)Peer interaction: 20%(p<0.001*)Repetitive behavior: 33%(p<0.001*)Social reciprocity: 11%(p=0.0053*) Mean improvement:39% (p<0.0001*) Behavioral: 69% improvedSocial: 63% improved Emotional: 72% improved Mean improvement: 46% (p<0.0001*) Aberrant Behavior Checklist —Community (ABC-C) subscales% improvement Qualitative Caregiver Behavioral Problems Survey % improvements Autism Parenting Stress Index Autism Impact Measure (AIM)% improvement Parent Rated Anxiety Scale for ASD (PRAS-ASD)November 2022 |
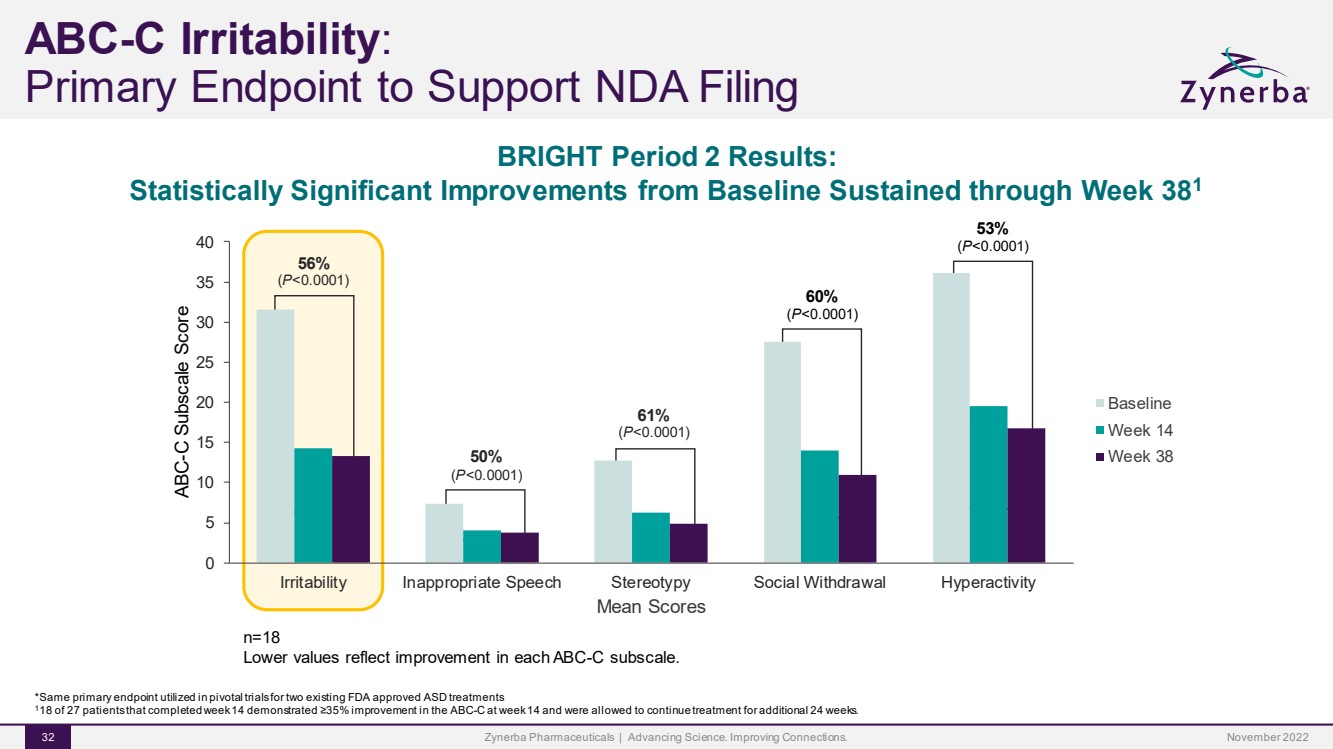
| 50%(P<0.0001)61%(P<0.0001)60%(P<0.0001)53%(P<0.0001)56%(P<0.0001) 0510 1520253035 40IrritabilityInappropriate SpeechStereotypySocial WithdrawalHyperactivityABC-C Subscale ScoreMean Scores Baseline Week 14 Week 38BRIGHT Period 2 Results: Statistically Significant Improvements from Baseline Sustained through Week 381Zynerba Pharmaceuticals | Advancing Science. Improving Connections. 32 ABC-C Irritability: Primary Endpoint to Support NDA Filingn=18 Lower values reflect improvement in each ABC-C subscale.*Same primary endpoint utilized in pivotal trials for two existing FDA approved ASD treatments118 of 27 patients that completed week 14 demonstrated ≥35% improvement in the ABC-C at week 14 and were allowed to continue treatment for additional 24 weeks.November 2022 |
| Next Steps in ASDZynerba Pharmaceuticals | Advancing Science. Improving Connections. 33 Submit Investigational New Drug (IND) application specific to ASD with finalized clinical protocolASD is now third in priority and initiation of Phase 3 program is deferred at this timeNovember 2022 |
| Positioned for Success |
| LeadershipZynerba Pharmaceuticals | Advancing Science. Improving Connections. 35 Armando AnidoChairman of the Board and CEO Terri B. SebreePresident Jim FickenscherCFO and VP, Corporate Development Paul KirschVP, Regulatory Affairs and Quality Assurance Joe ApostolicoVP, Human Resources Terry HurstGM, Zynerba Pharmaceuticals Pty Ltd (Australia) Stephen O’Quinn, PharmDVP, Medical Affairs Albert P. ParkerChief Legal Officer Brian RosenbergerVP, Commercial and Business Development Nancy Tich, Ph.D.VP, Clinical Ray MannionVP, Manufacturing Carol O’NeillVP, Development November 2022 |
| Financial StrengthZynerba Pharmaceuticals | Advancing Science. Improving Connections. 36 CASH AND CASH EQUIVALENTS$55.9Mas of September 30, 2022;expected to be sufficient to fund operations and capital requirements into Q1 2024BALANCE SHEET CLEANNo debt, 47.1M shares outstanding (as of November 9, 2022) November 2022 |
| A Year of Clinical Progress AheadZynerba Pharmaceuticals | Advancing Science. Improving Connections. 37 Fragile X Syndrome (FXS) Autism Spectrum Disorder (ASD) 22q Deletion Syndrome (22q) U.S. Orphan Drug and Fast Track designations; EU Orphan Drug designationU.S. and EU Orphan Drug designations Pivotal trial results expected in 2H 2023 Finalize regulatory pathwaywith FDA; Initiate Phase 3 after RECONNECT topline results Submit IND with finalized clinical protocol; initiation of Phase 3 program deferredNovember 2022 |
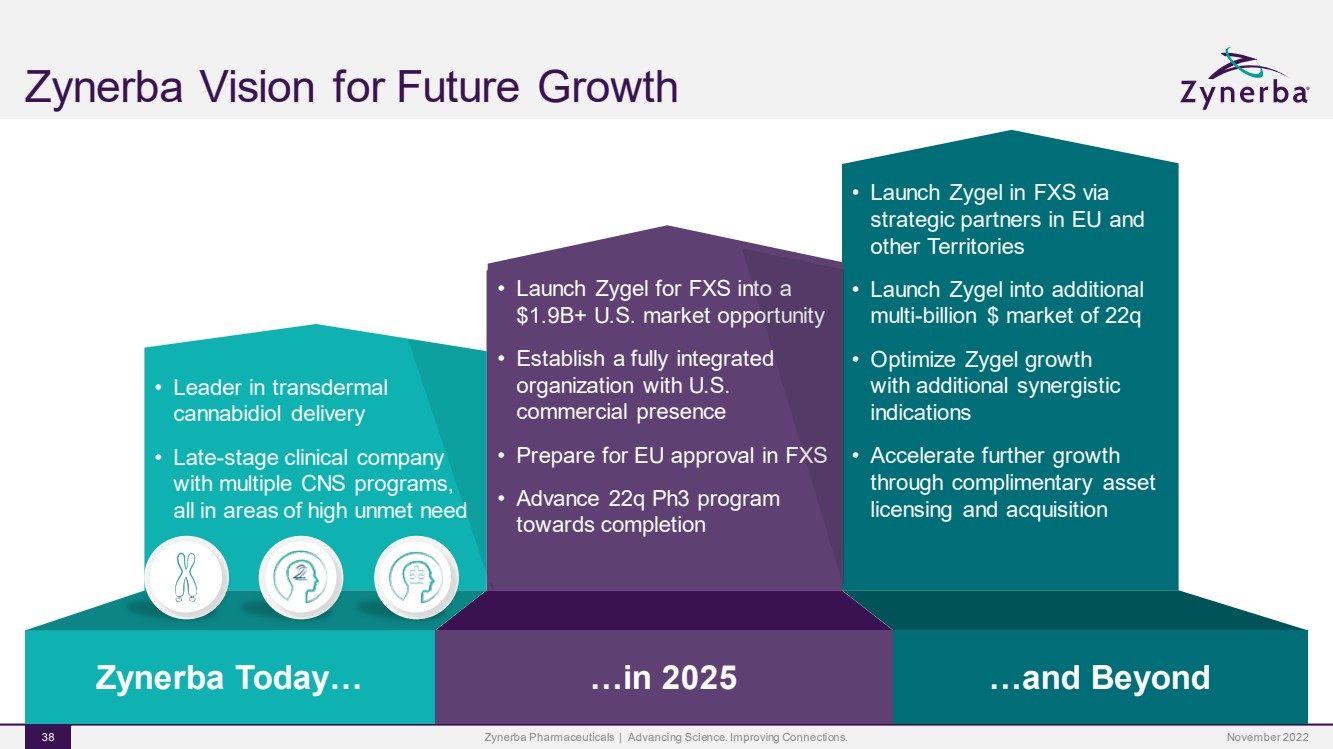
| •Launch Zygel for FXS into a $1.9B+ U.S. market opportunity•Establish a fully integrated organization with U.S. commercial presence•Preparing for EU approval in FXS•Advance 22q and ASD Ph3 programs towards completion •Leader in transdermal cannabidiol delivery •Late-stage clinical company with multiple CNS programs, all in areas of high unmet need •Launch Zygel in FXS via strategic partners in EU and other Territories•Launch Zygel into additional multi-billion $ market of 22q •Optimize Zygel growth with additional synergistic indications•Accelerate further growth through complimentary asset licensing and acquisition …and Beyond Zynerba Today… …in 2025 Zynerba Vision for Future GrowthZynerba Pharmaceuticals | Advancing Science. Improving Connections. 38 •Launch Zygel for FXS into a $1.9B+ U.S. market opportunity•Establish a fully integrated organization with U.S. commercial presence•Prepare for EU approval in FXS•Advance 22q Ph3 program towards completion November 2022 |
| A different and exciting approach to Cannabidiol |
| © 2022 Zynerba Pharmaceuticals, Inc. All rights reserved. NASDAQZYNE Thank you! |