
VAX-31 Phase 1/2 Study Topline Results in Adults Aged 50 and Older September 3, 2024 Exhibit 99.2

September 3, 2024 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, statements related to the potential benefits of Vaxcyte’s vaccine candidates, including breadth of coverage, the ability to deliver a potentially best-in-class pneumococcal conjugate vaccine franchise and the potential to improve upon the standard-of-care and raise the immunogenicity threshold; the process and timing of anticipated future development of Vaxcyte’s vaccine candidates; the timing and availability of data for the VAX-24 infant Phase 2 study; the timing and availability of data for the VAX-31 adult Phase 3 studies and infant Phase 2 study; the potential of VAX-31 to provide unrivaled invasive pneumococcal disease coverage; the ability of Vaxcyte’s cell-free platform to deliver the broadest-spectrum PCVs that provide protection against both currently circulating and historically prevalent strains; demand for Vaxcyte’s vaccine candidates; the growth and expansion of the pneumococcal vaccine market; the market opportunity for Vaxcyte’s vaccines; Vaxcyte’s expectations regarding the spectrum coverage, regulatory pathway, adoption speed and immunogenicity of its vaccine candidates; and other statements that are not historical fact. The words “anticipate,” “believe,” “continue,” “could,” “designed,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements are based on Vaxcyte’s current expectations and actual results and timing of events could differ materially from those anticipated in such forward-looking statements as a result of risks and uncertainties, including, without limitation, risks related to Vaxcyte’s product development programs, including development timelines, success and timing of chemistry, manufacturing and controls and related manufacturing activities; potential delays or inability to obtain and maintain required regulatory approvals for its vaccine candidates; the risks and uncertainties inherent with preclinical and clinical development processes; the success, cost and timing of all development activities and clinical trials; and the sufficiency of cash and other funding to support Vaxcyte’s development programs and other operating expenses, any of which could materially and adversely affect Vaxcyte’s business and operations. These and other risks are described more fully in Vaxcyte’s filings with the Securities and Exchange Commission (SEC), including its Quarterly Report on Form 10-Q filed with the SEC on August 6, 2024 or in other documents Vaxcyte subsequently files with or furnishes to the SEC. Vaxcyte undertakes no duty or obligation to update any forward-looking statements contained in this release as a result of new information, future events or changes in its expectations.

September 3, 2024 VAXCYTE MISSION STATEMENT We are on a global mission to engineer high-fidelity vaccines that protect humankind from the consequences of bacterial diseases.

September 3, 2024 Agenda Introduction and vax-31 Results Overview VAX-31 Phase 1/2 Study TOPLINE results in Adults Aged 50 and Older Disposition and Demographics Tolerability and Full Six-Month Safety Data Topline Immunogenicity Data PCV franchise status and Next Steps

Introduction and VAX-31 Results Overview September 3, 2024

Unprecedented Results Support Potential Best-in-Class PCV With Broadest Serotype and Disease Coverage Summary of VAX-31 Adult 50+ Phase 1/2 Study Topline Data Findings September 3, 2024 SAFETY AND TOLERABILITY: At all doses studied, VAX-31 was well tolerated and demonstrated a safety profile similar to Prevnar 20® (PCV20) for all doses IMMUNOGENICITY: At all doses studied, VAX-31 demonstrated robust OPA immune responses for all 31 serotypes (STs) -- all three doses advanceable to Phase 3 High and Middle doses met or exceeded OPA regulatory immunogenicity criteria for all 31 STs, Low dose for 29 of 31 STs For the 20 STs common with PCV20: High dose, 18 had GMR greater than 1.0 and 7 achieved statistically higher immune responses; Middle dose, 13 had GMR greater than 1.0 and 5 achieved statistically higher immune responses; Low dose, 8 had GMR greater than 1.0 and 3 achieved statistically higher immune responses For the 11 additional STs unique to VAX-31: All 11 met the superiority criteria at all doses KEY VAX-31 NEXT STEPS: Adults: VAX-31 selected to advance to Phase 3 with initiation of pivotal, non-inferiority study by mid-2025 and topline safety, tolerability and immunogenicity results in 2026; pursuing Breakthrough Therapy Designation Pediatrics: Plan to initiate VAX-31 infant Phase 2 study in 1Q:2025 following IND submission and clearance PLATFORM: The VAX-31 data further validate the potential of Vaxcyte’s carrier-sparing platform to deliver the broadest-spectrum PCVs that provide protection against both currently circulating and historically prevalent STs GMR = geometric mean ratio; OPA = Opsonophagocytic activity

September 3, 2024 Over 150,000 U.S. hospitalizations annually due to pneumococcal pneumonia Streptococcus pneumoniae is the leading cause of vaccine preventable deaths globally in children under five ~300,000 children under five years old die annually worldwide due to Streptococcus pneumoniae Global Health Impact of Pneumococcal Disease (PD) Remains Significant Streptococcus pneumoniae is among the World Health Organization’s top antibiotic-resistant pathogens to be urgently addressed and the U.S. CDC lists drug-resistant Streptococcus pneumoniae as a “serious threat” https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-17-pneumococcal-disease.html#:~:text=Over%20150%2C000%20hospitalizations%20from%20pneumococcal,younger%20than%20age%205%20years. https://iris.who.int/bitstream/handle/10665/376776/9789240093461-eng.pdf?sequence=1. https://www.cdc.gov/pneumococcal/php/drug-resistance/index.html. GBD 2019 Diseases and Injuries Collaborators.. Lancet 2020; 396: 1204-22. Supplementary Appendix 2. https://www.gavi.org/pneumococcal-disease-leading-vaccine-preventable-cause-death-children-under-five. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8677503/table/T2/. https://www.cdc.gov/pneumococcal/php/surveillance/index.html#:~:text=Global%20trends,deaths%20occur%20in%20developing%20countries.

VAX-31 Phase 1/2 Study Topline Results in Adults Aged 50 and Older September 3, 2024
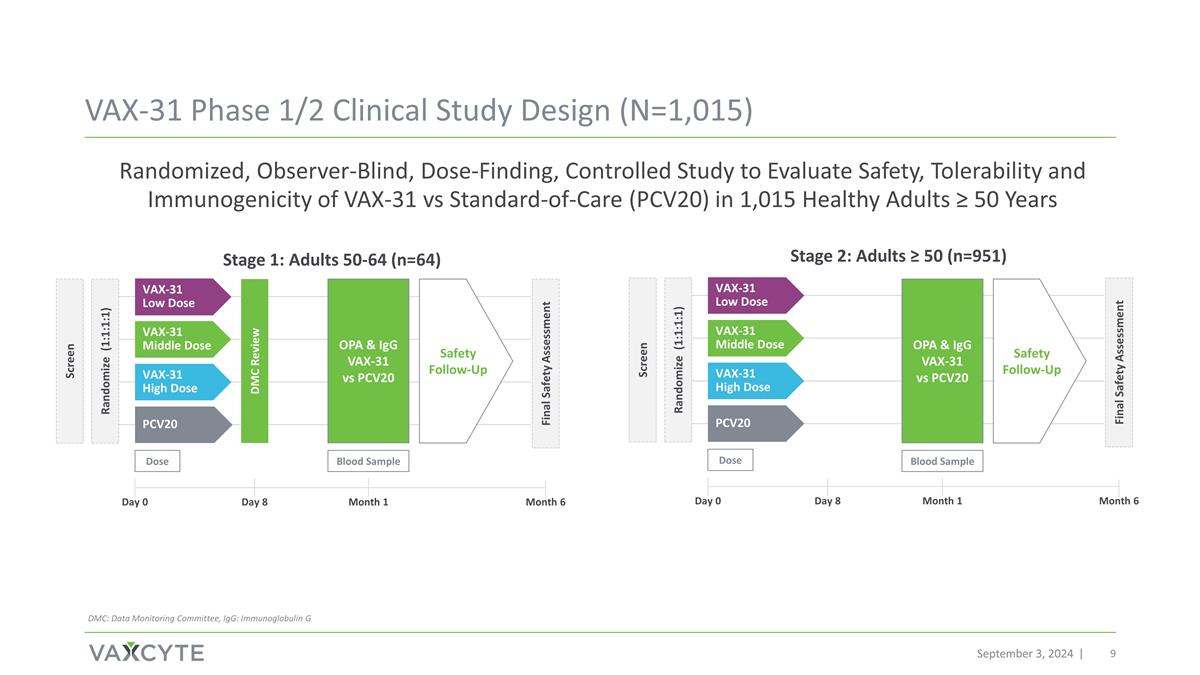
Randomized, Observer-Blind, Dose-Finding, Controlled Study to Evaluate Safety, Tolerability and Immunogenicity of VAX-31 vs Standard-of-Care (PCV20) in 1,015 Healthy Adults ≥ 50 Years Dose Blood Sample PCV20 VAX-31 High Dose VAX-31 Middle Dose VAX-31 Low Dose Safety Follow-Up Randomize (1:1:1:1) Screen DMC Review Final Safety Assessment Dose PCV20 VAX-31 High Dose VAX-31 Middle Dose VAX-31 Low Dose Randomize (1:1:1:1) Screen Final Safety Assessment OPA & IgG VAX-31 vs PCV20 Blood Sample Safety Follow-Up OPA & IgG VAX-31 vs PCV20 Stage 1: Adults 50-64 (n=64) Stage 2: Adults ≥ 50 (n=951) Day 0 Month 1 Month 6 Day 8 Day 0 Month 1 Month 6 Day 8 VAX-31 Phase 1/2 Clinical Study Design (N=1,015) September 3, 2024 DMC: Data Monitoring Committee, IgG: Immunoglobulin G
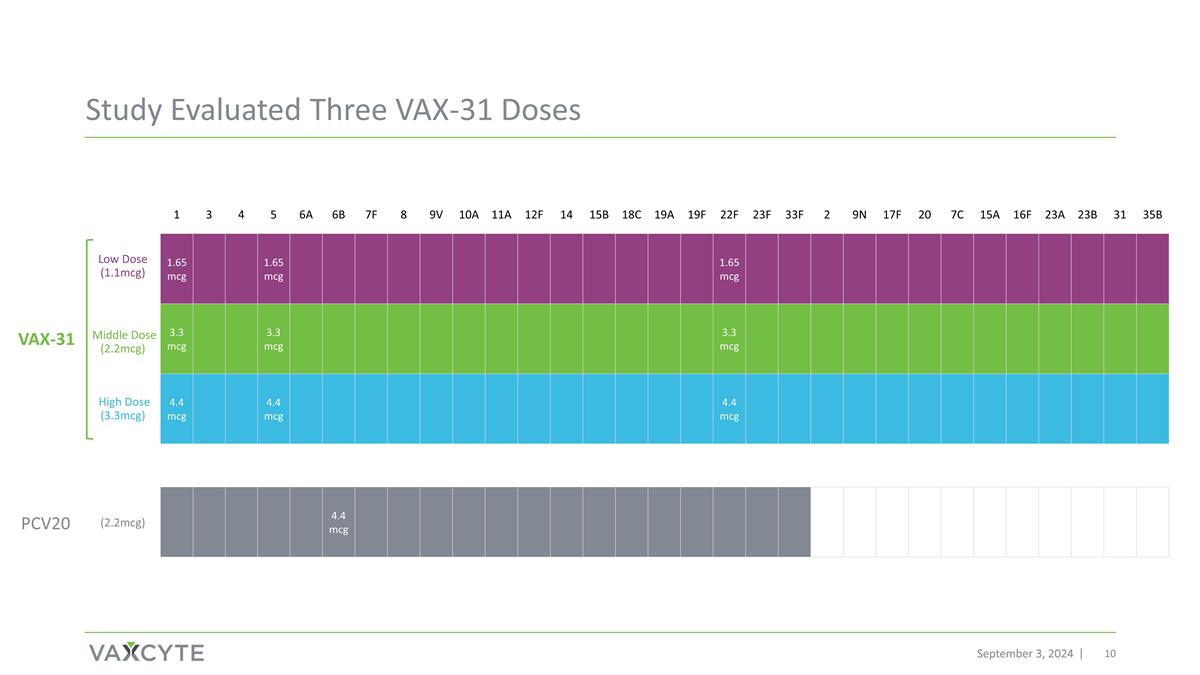
Study Evaluated Three VAX-31 Doses September 3, 2024 Low Dose (1.1mcg) High Dose (3.3mcg) PCV20 VAX-31 (2.2mcg) 1 3 4 5 6A 6B 7F 8 9V 10A 11A 12F 14 15B 18C 19A 19F 22F 23F 33F 2 9N 17F 20 7C 15A 16F 23A 23B 31 35B 1.65 mcg 1.65 mcg 1.65 mcg 3.3 mcg 3.3 mcg 3.3 mcg 4.4 mcg 4.4 mcg 4.4 mcg 4.4 mcg Middle Dose (2.2mcg)
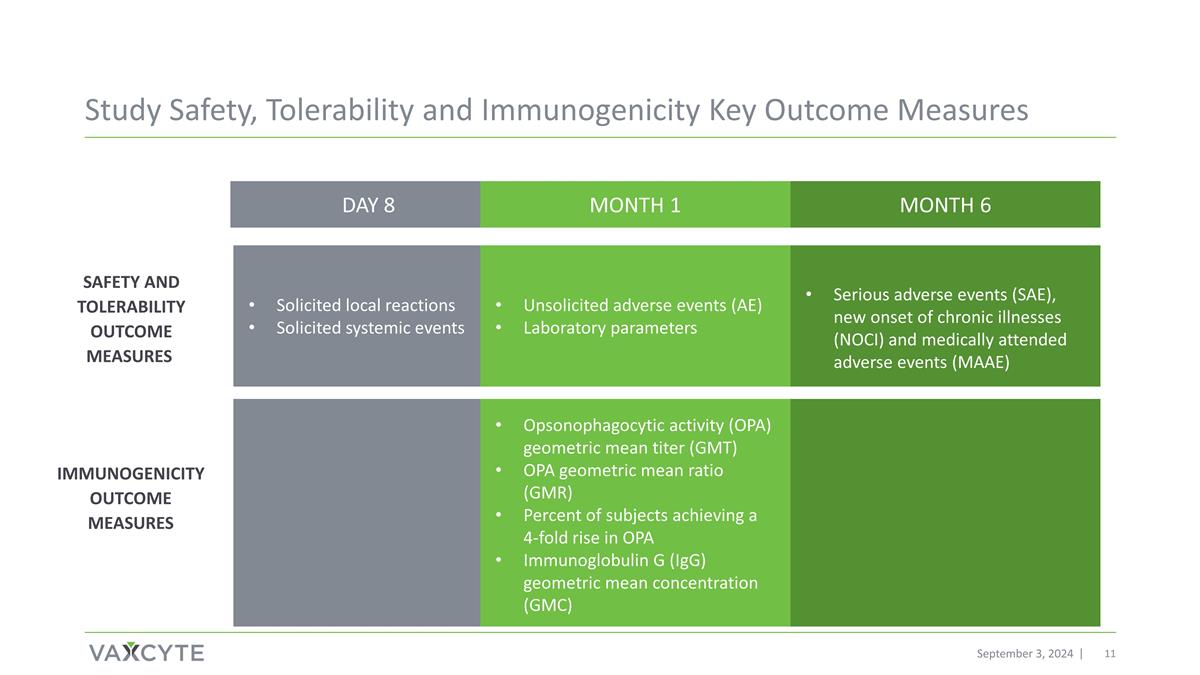
Study Safety, Tolerability and Immunogenicity Key Outcome Measures September 3, 2024 Safety and Tolerability Outcome Measures Immunogenicity Outcome Measures DAY 8 MONTH 1 MONTH 6 Solicited local reactions Solicited systemic events Unsolicited adverse events (AE) Laboratory parameters Serious adverse events (SAE), new onset of chronic illnesses (NOCI) and medically attended adverse events (MAAE) Opsonophagocytic activity (OPA) geometric mean titer (GMT) OPA geometric mean ratio (GMR) Percent of subjects achieving a 4-fold rise in OPA Immunoglobulin G (IgG) geometric mean concentration (GMC)

Disposition and Demographics September 3, 2024

Study Disposition High Proportion of Subjects with Safety and Immunogenicity Follow-Up September 3, 2024 Phase 1/2 Study in Adults 50+ 24 subjects (2.4%) discontinued and did not complete the full Month 6 safety follow-up (lost to follow-up (13), withdrawal by subject (11)). Enrolled N = 1,015 Included in Safety Population N = 253 (100.0%) Included in Safety Population N = 253 (100.0%) Included in Safety Population N = 254 (100.0%) Included in Safety Population N = 255 (100.0%) Vaccinated PCV20 N = 253 Vaccinated VAX-31 Low Dose N = 255 Vaccinated VAX-31 Middle Dose N = 254 Vaccinated VAX-31 High Dose N = 253 Included in Immunogenicity Population N = 247 (96.8%) Included in Immunogenicity Population N = 245 (96.4%) Included in Immunogenicity Population N = 244 (96.4%) Included in Immunogenicity Population N = 247 (97.6%)
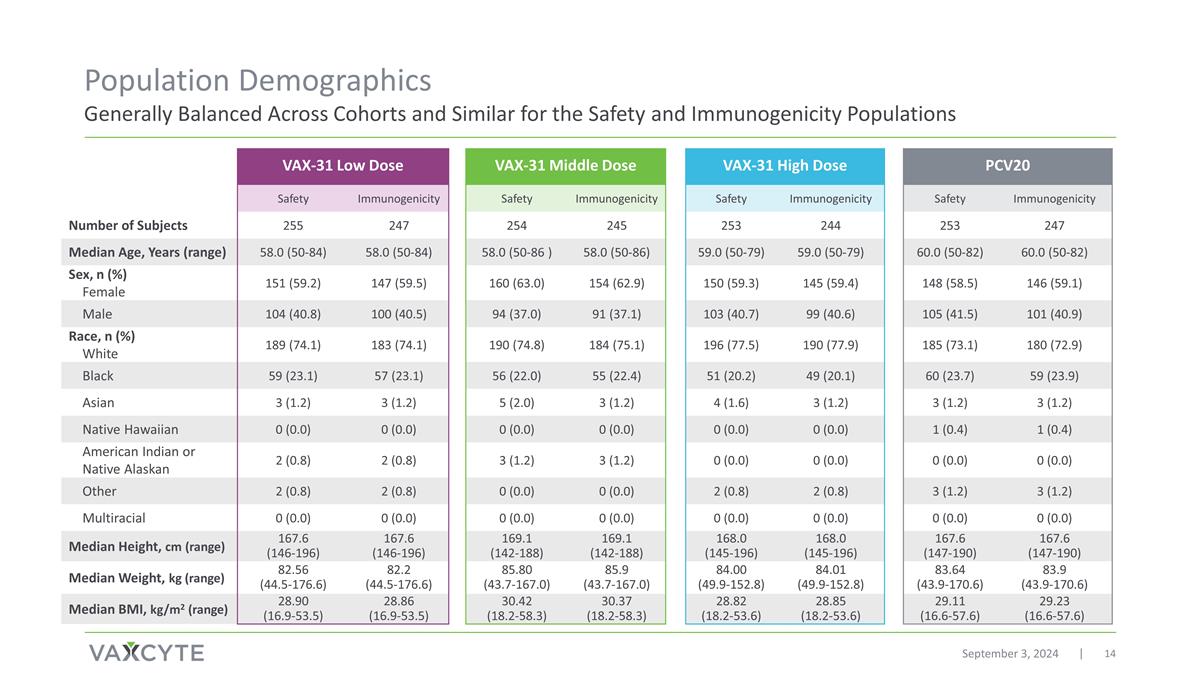
Population Demographics Generally Balanced Across Cohorts and Similar for the Safety and Immunogenicity Populations September 3, 2024 VAX-31 Low Dose VAX-31 Middle Dose VAX-31 High Dose PCV20 Safety Immunogenicity Safety Immunogenicity Safety Immunogenicity Safety Immunogenicity Number of Subjects 255 247 254 245 253 244 253 247 Median Age, Years (range) 58.0 (50-84) 58.0 (50-84) 58.0 (50-86 ) 58.0 (50-86) 59.0 (50-79) 59.0 (50-79) 60.0 (50-82) 60.0 (50-82) Sex, n (%) Female 151 (59.2) 147 (59.5) 160 (63.0) 154 (62.9) 150 (59.3) 145 (59.4) 148 (58.5) 146 (59.1) Male 104 (40.8) 100 (40.5) 94 (37.0) 91 (37.1) 103 (40.7) 99 (40.6) 105 (41.5) 101 (40.9) Race, n (%) White 189 (74.1) 183 (74.1) 190 (74.8) 184 (75.1) 196 (77.5) 190 (77.9) 185 (73.1) 180 (72.9) Black 59 (23.1) 57 (23.1) 56 (22.0) 55 (22.4) 51 (20.2) 49 (20.1) 60 (23.7) 59 (23.9) Asian 3 (1.2) 3 (1.2) 5 (2.0) 3 (1.2) 4 (1.6) 3 (1.2) 3 (1.2) 3 (1.2) Native Hawaiian 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 1 (0.4) 1 (0.4) American Indian or Native Alaskan 2 (0.8) 2 (0.8) 3 (1.2) 3 (1.2) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) Other 2 (0.8) 2 (0.8) 0 (0.0) 0 (0.0) 2 (0.8) 2 (0.8) 3 (1.2) 3 (1.2) Multiracial 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) Median Height, cm (range) 167.6 (146-196) 167.6 (146-196) 169.1 (142-188) 169.1 (142-188) 168.0 (145-196) 168.0 (145-196) 167.6 (147-190) 167.6 (147-190) Median Weight, kg (range) 82.56 (44.5-176.6) 82.2 (44.5-176.6) 85.80 (43.7-167.0) 85.9 (43.7-167.0) 84.00 (49.9-152.8) 84.01 (49.9-152.8) 83.64 (43.9-170.6) 83.9 (43.9-170.6) Median BMI, kg/m2 (range) 28.90 (16.9-53.5) 28.86 (16.9-53.5) 30.42 (18.2-58.3) 30.37 (18.2-58.3) 28.82 (18.2-53.6) 28.85 (18.2-53.6) 29.11 (16.6-57.6) 29.23 (16.6-57.6)

Tolerability and Full Six-Month Safety Data September 3, 2024
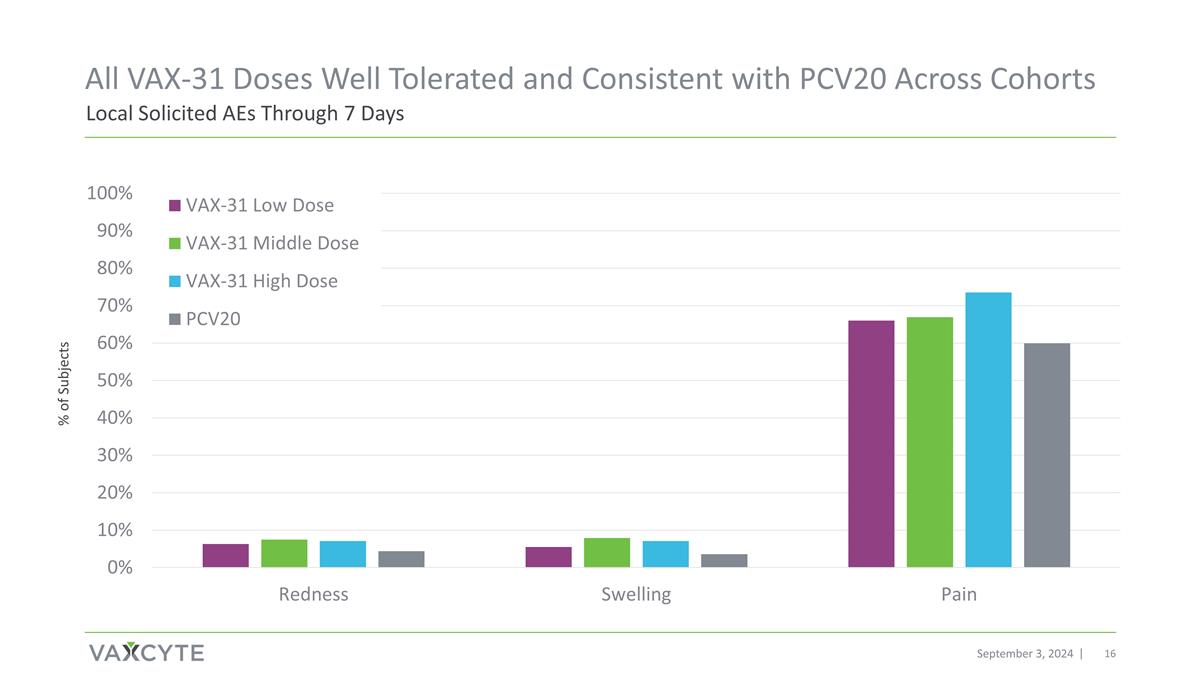
September 3, 2024 Local Solicited AEs Through 7 Days All VAX-31 Doses Well Tolerated and Consistent with PCV20 Across Cohorts % of Subjects
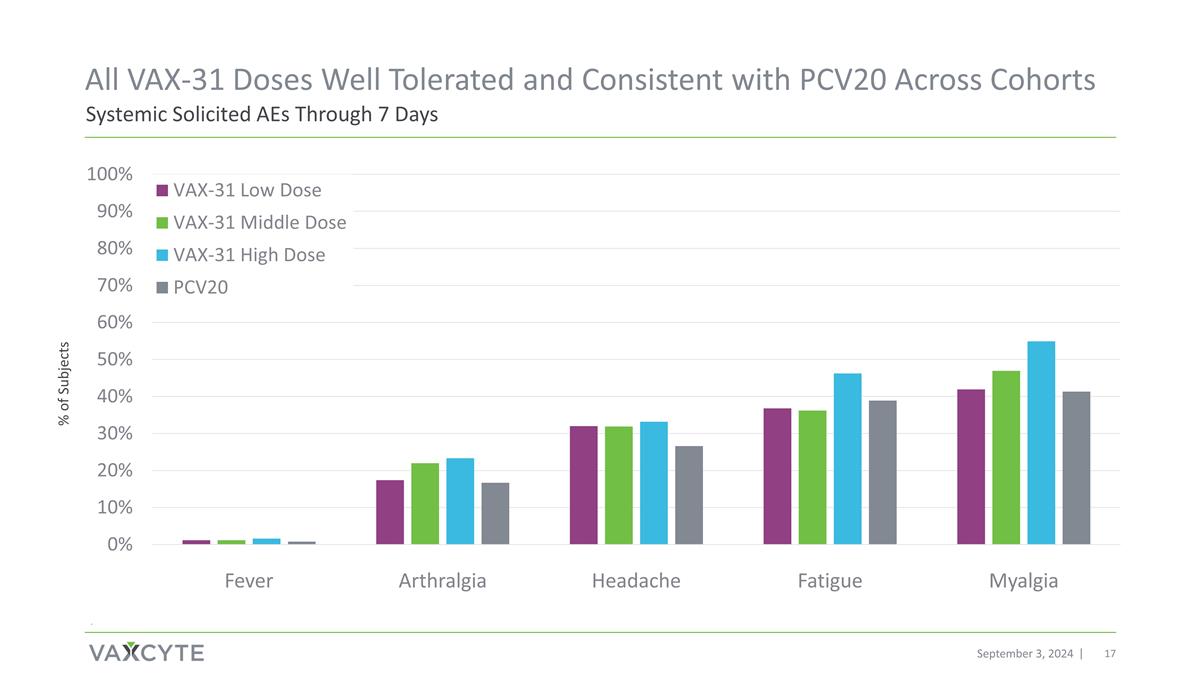
September 3, 2024 . Systemic Solicited AEs Through 7 Days All VAX-31 Doses Well Tolerated and Consistent with PCV20 Across Cohorts % of Subjects
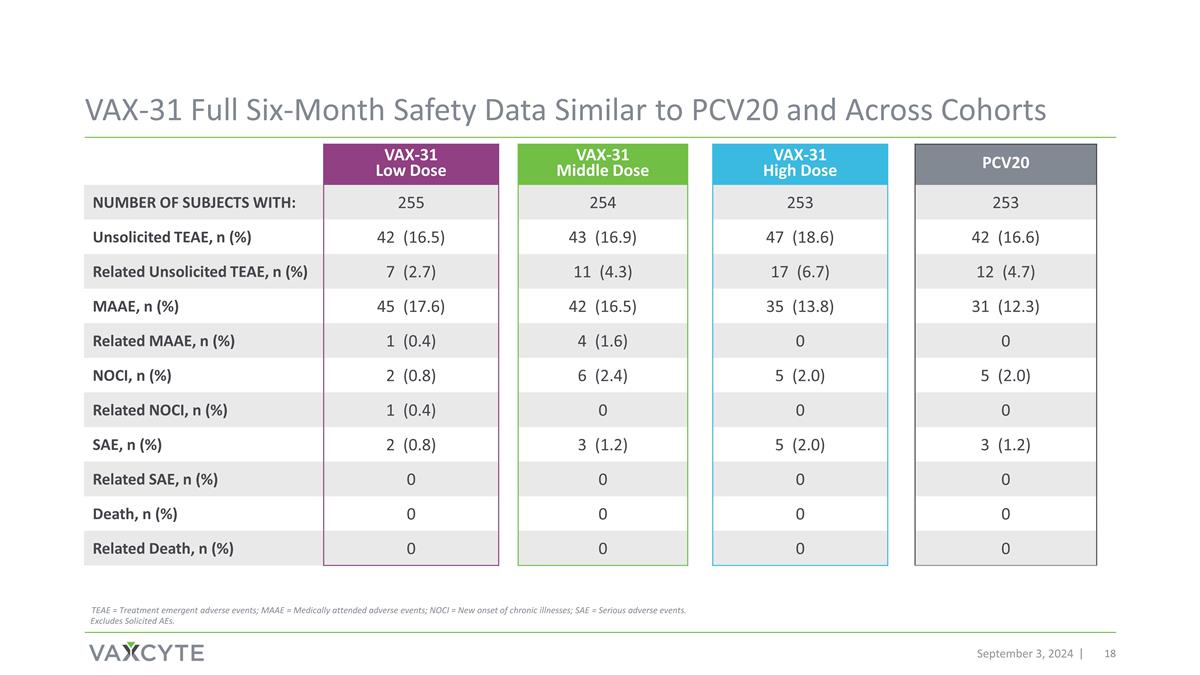
VAX-31 Full Six-Month Safety Data Similar to PCV20 and Across Cohorts September 3, 2024 VAX-31 Low Dose VAX-31 Middle Dose VAX-31 High Dose PCV20 Number of Subjects with: 255 254 253 253 Unsolicited TEAE, n (%) 42 (16.5) 43 (16.9) 47 (18.6) 42 (16.6) Related Unsolicited TEAE, n (%) 7 (2.7) 11 (4.3) 17 (6.7) 12 (4.7) MAAE, n (%) 45 (17.6) 42 (16.5) 35 (13.8) 31 (12.3) Related MAAE, n (%) 1 (0.4) 4 (1.6) 0 0 NOCI, n (%) 2 (0.8) 6 (2.4) 5 (2.0) 5 (2.0) Related NOCI, n (%) 1 (0.4) 0 0 0 SAE, n (%) 2 (0.8) 3 (1.2) 5 (2.0) 3 (1.2) Related SAE, n (%) 0 0 0 0 Death, n (%) 0 0 0 0 Related Death, n (%) 0 0 0 0 TEAE = Treatment emergent adverse events; MAAE = Medically attended adverse events; NOCI = New onset of chronic illnesses; SAE = Serious adverse events. Excludes Solicited AEs.

Topline Immunogenicity Data September 3, 2024

Criteria for 11 Incremental Serotypes in VAX-31: Criteria for 20 Serotypes Common to VAX-31 and PCV20: Precedent Immunogenicity Regulatory Criteria for Phase 2/3 PCV Studies September 3, 2024 CI = confidence interval . Non-inferiority: Lower bound of the 2-sided 95% CI of the OPA GMR is greater than 0.5 Superiority: Lower bound of the 2-sided 95% CI of the difference in the proportions of participants with a ≥4-fold increase from Day 1 to Month 1 is greater than 10% Lower bound of the 2-sided 95% CI of the OPA GMR is greater than 2.0
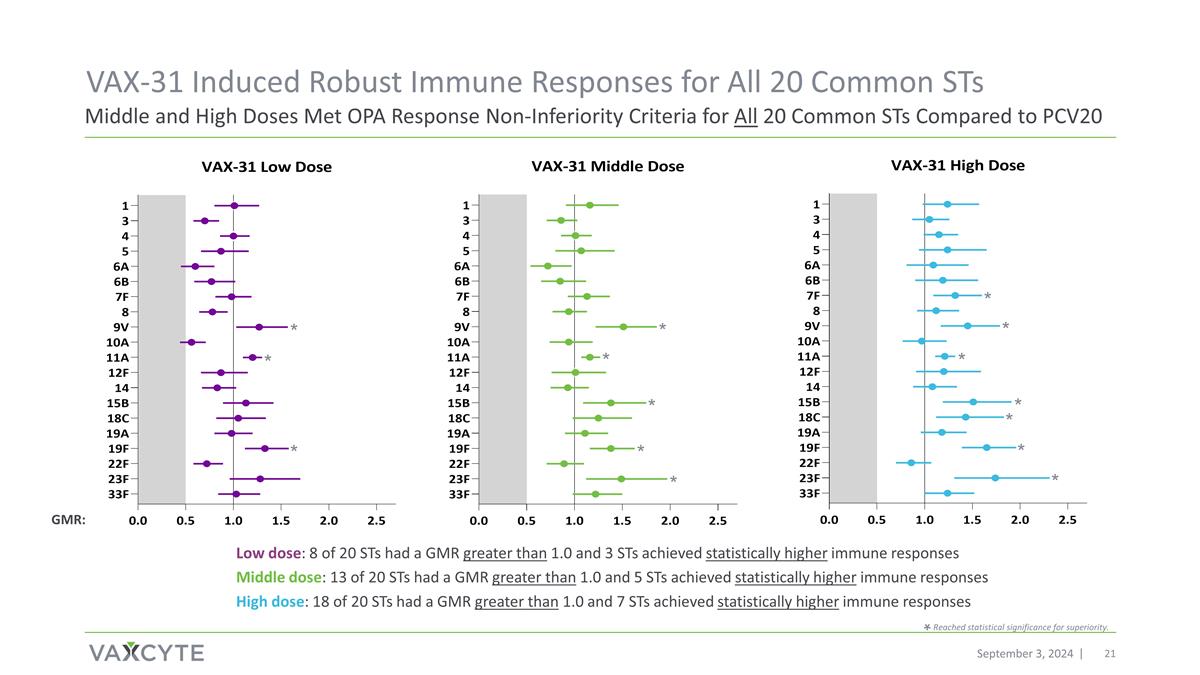
September 3, 2024 Middle and High Doses Met OPA Response Non-Inferiority Criteria for All 20 Common STs Compared to PCV20 VAX-31 Induced Robust Immune Responses for All 20 Common STs GMR: * * * * * * * * * * * * * Reached statistical significance for superiority. Low dose: 8 of 20 STs had a GMR greater than 1.0 and 3 STs achieved statistically higher immune responses Middle dose: 13 of 20 STs had a GMR greater than 1.0 and 5 STs achieved statistically higher immune responses High dose: 18 of 20 STs had a GMR greater than 1.0 and 7 STs achieved statistically higher immune responses * * *
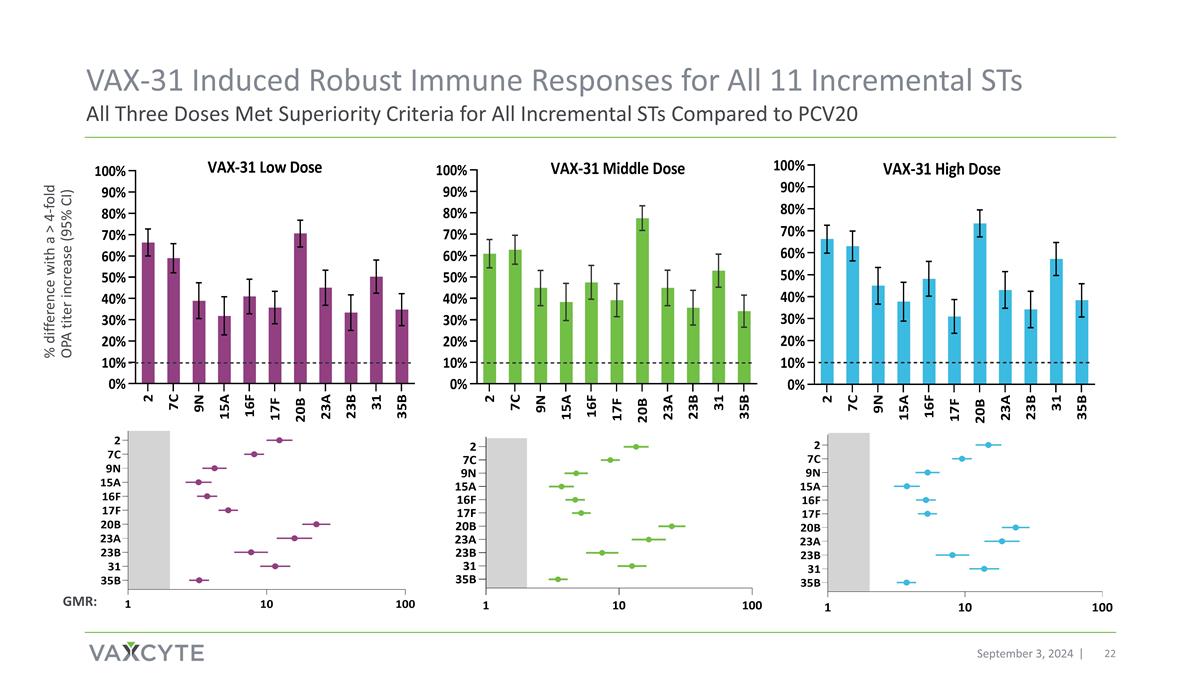
VAX-31 Induced Robust Immune Responses for All 11 Incremental STs All Three Doses Met Superiority Criteria for All Incremental STs Compared to PCV20 September 3, 2024 % difference with a > 4-fold OPA titer increase (95% CI) GMR:
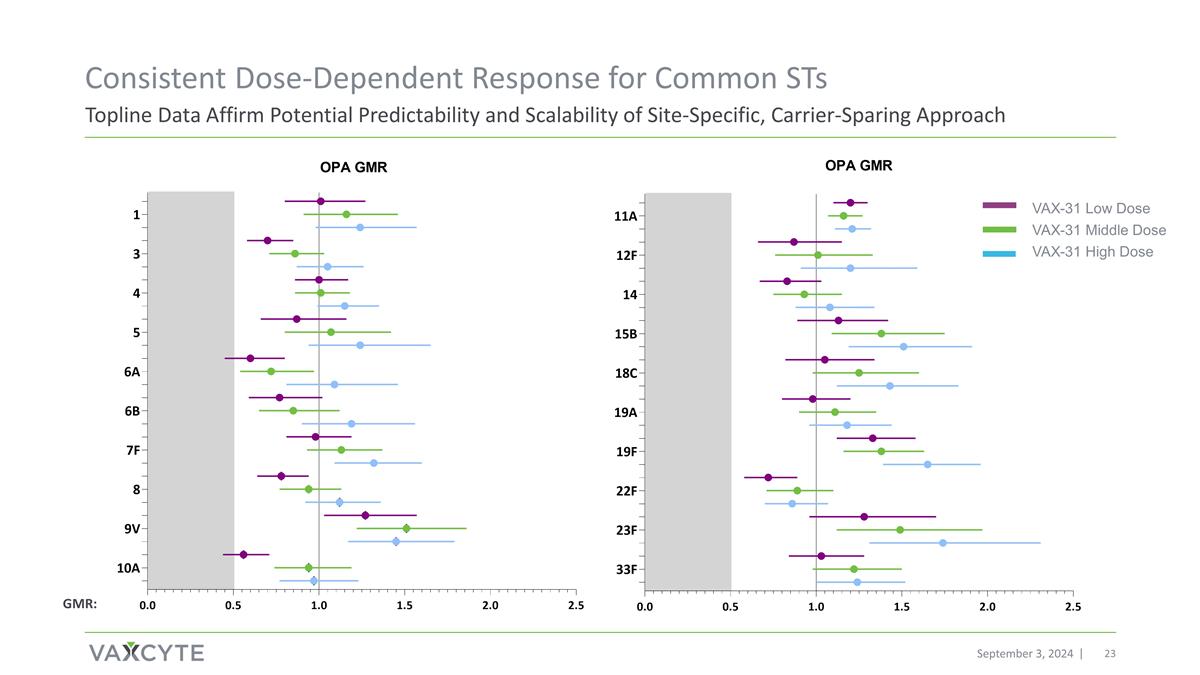
Topline Data Affirm Potential Predictability and Scalability of Site-Specific, Carrier-Sparing Approach September 3, 2024 Consistent Dose-Dependent Response for Common STs VAX-31 Low Dose VAX-31 Middle Dose VAX-31 High Dose GMR:
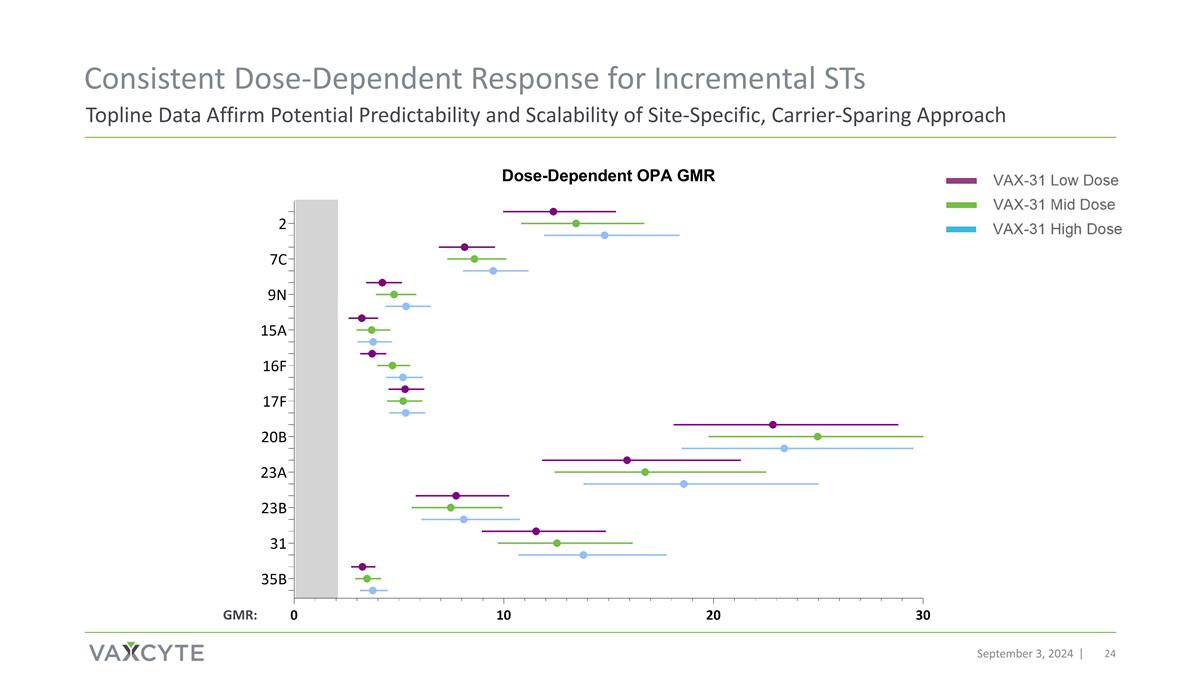
September 3, 2024 Topline Data Affirm Potential Predictability and Scalability of Site-Specific, Carrier-Sparing Approach Consistent Dose-Dependent Response for Incremental STs GMR:
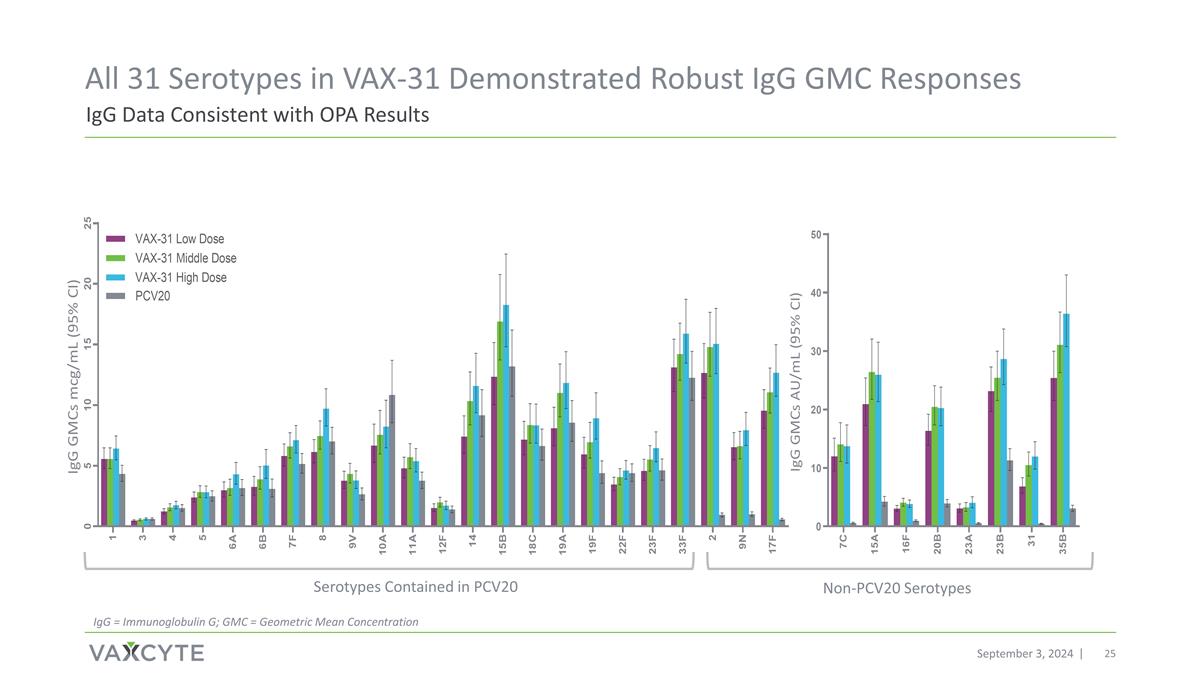
IgG Data Consistent with OPA Results All 31 Serotypes in VAX-31 Demonstrated Robust IgG GMC Responses September 3, 2024 Non-PCV20 Serotypes Serotypes Contained in PCV20 IgG = Immunoglobulin G; GMC = Geometric Mean Concentration

PCV Franchise Status and Next Steps September 3, 2024

Potential to Set New Standard-of-Care with Broadest Coverage and Raise Immunogenicity Threshold September 3, 2024 SAFETY AND TOLERABILITY: At all doses studied, VAX-31 was well tolerated and demonstrated a safety profile similar to PCV20 IMMUNOGENICITY: At all doses studied, VAX-31 demonstrated robust OPA immune responses for all 31 STs High and middle doses met or exceeded regulatory immunogenicity criteria for all 31 STs PCV FRANCHISE STRATEGY: Adult: VAX-31 selected to advance to Phase 3; dose to be chosen prior to study initiation Pediatric: Advance both VAX-24 and VAX-31 Next-generation readiness PLATFORM: The data further validate potential of Vaxcyte’s site-specific, carrier-sparing platform to deliver the broadest-spectrum PCVs that provide protection against both currently circulating and historically prevalent STs

Potential Best-in-Class PCV Franchise for Adult and Infant Segments September 3, 2024 24-valent PCV candidate VAX-24 Infants Announce topline safety, tolerability and immunogenicity data from primary three-dose immunization series of Phase 2 study, which is fully enrolled with 802 healthy infants, by end of 1Q:2025, followed by topline data from booster dose by end of 2025. 31-valent PCV candidate VAX-31 Adults Following FDA End-of-Phase 2 meeting, initiate Phase 3 pivotal, non-inferiority study by mid-2025 and announce topline safety, tolerability and immunogenicity data in 2026. Initiate remaining Phase 3 studies in 2025 and 2026. Population Investigational PCV Key Anticipated Milestones 1Guidance as of September 3, 2024. 31-valent PCV candidate VAX-31 Initiate Phase 2 study in 1Q:2025 following IND submission and clearance. Announce topline safety, tolerability and immunogenicity data from VAX-31 infant Phase 2 study primary three-dose immunization series in mid-2026, followed by topline data from booster dose approximately nine months later. Clinical Development Next Steps and Anticipated Milestones1
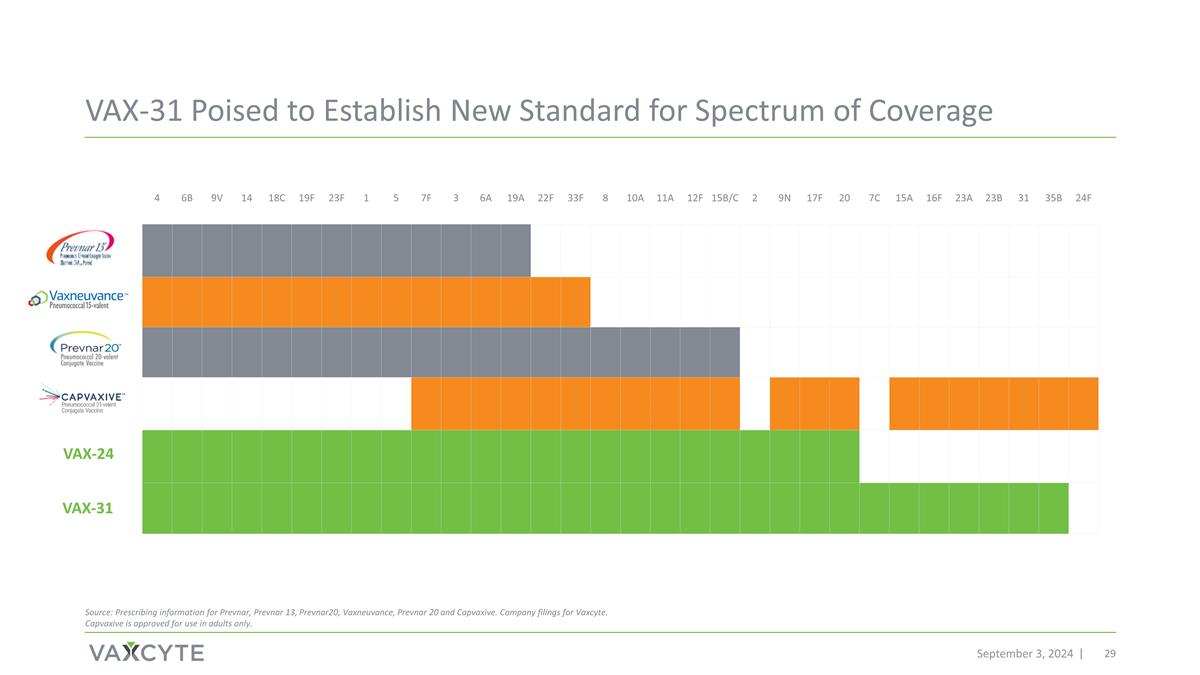
VAX-31 Poised to Establish New Standard for Spectrum of Coverage 4 6B 9V 14 18C 19F 23F 1 5 7F 3 6A 19A 22F 33F 8 10A 11A 12F 15B/C 2 9N 17F 20 7C 15A 16F 23A 23B 31 35B 24F Source: Prescribing information for Prevnar, Prevnar 13, Prevnar20, Vaxneuvance, Prevnar 20 and Capvaxive. Company filings for Vaxcyte. Capvaxive is approved for use in adults only. VAX-31 September 3, 2024 VAX-24
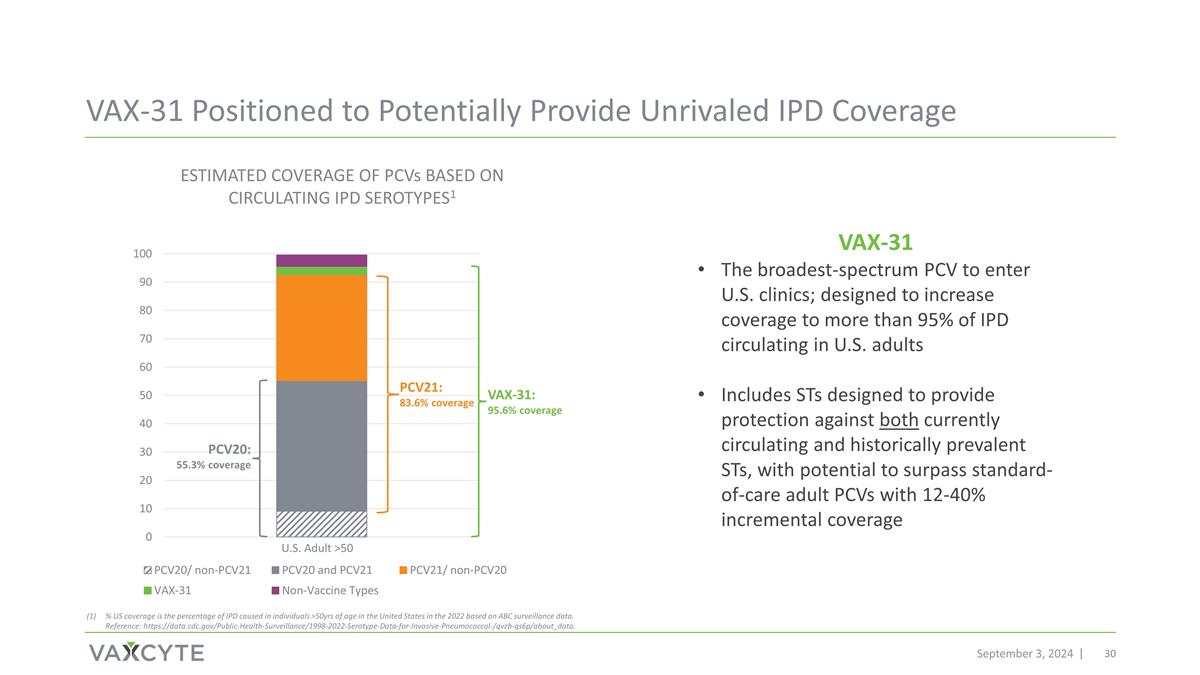
PCV20: 55.3% coverage PCV21: 83.6% coverage VAX-31: 95.6% coverage September 3, 2024 ESTIMATED COVERAGE OF PCVs BASED ON CIRCULATING IPD SEROTYPES1 VAX-31 Positioned to Potentially Provide Unrivaled IPD Coverage % US coverage is the percentage of IPD caused in individuals >50yrs of age in the United States in the 2022 based on ABC surveillance data. Reference: https://data.cdc.gov/Public-Health-Surveillance/1998-2022-Serotype-Data-for-Invasive-Pneumococcal-/qvzb-qs6p/about_data. U.S. Adult >50 VAX-31 The broadest-spectrum PCV to enter U.S. clinics; designed to increase coverage to more than 95% of IPD circulating in U.S. adults Includes STs designed to provide protection against both currently circulating and historically prevalent STs, with potential to surpass standard-of-care adult PCVs with 12-40% incremental coverage
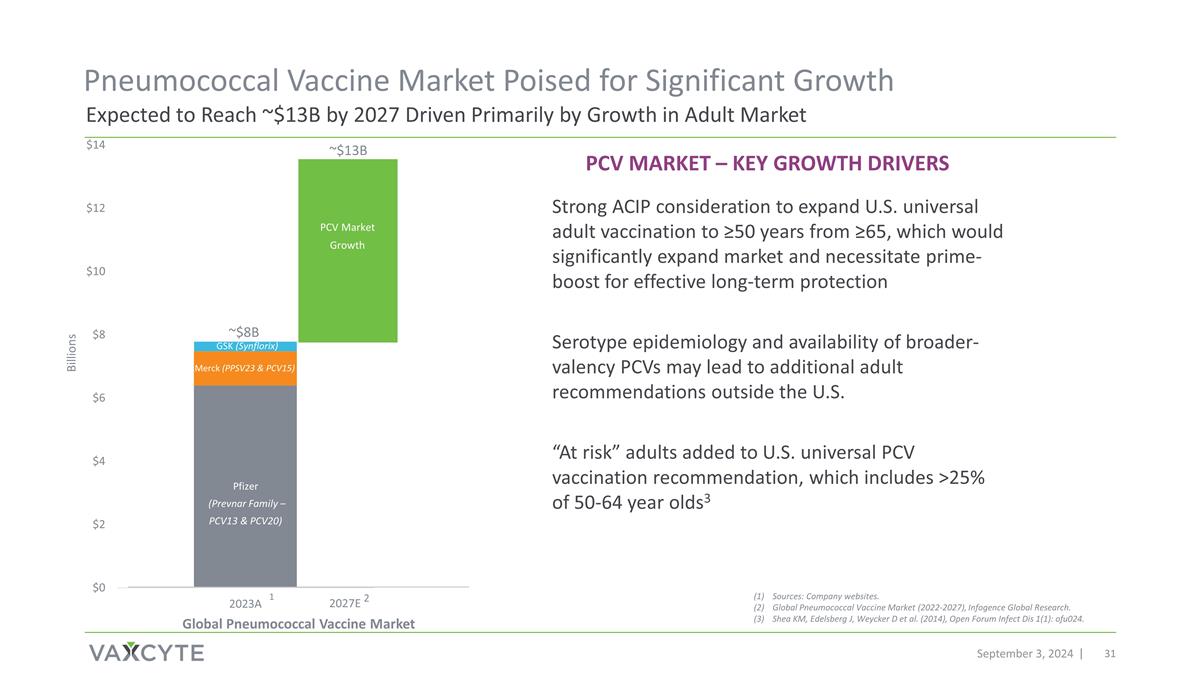
September 3, 2024 ~$13B ~$8B Pfizer (Prevnar Family – PCV13 & PCV20) Merck (PPSV23 & PCV15) GSK (Synflorix) Billions Other Sources: Company websites. Global Pneumococcal Vaccine Market (2022-2027), Infogence Global Research. Shea KM, Edelsberg J, Weycker D et al. (2014), Open Forum Infect Dis 1(1): ofu024. 1 PCV Market – Key Growth Drivers 2027E 2 Global Pneumococcal Vaccine Market PCV Market Growth Expected to Reach ~$13B by 2027 Driven Primarily by Growth in Adult Market Pneumococcal Vaccine Market Poised for Significant Growth Strong ACIP consideration to expand U.S. universal adult vaccination to ≥50 years from ≥65, which would significantly expand market and necessitate prime-boost for effective long-term protection Serotype epidemiology and availability of broader-valency PCVs may lead to additional adult recommendations outside the U.S. “At risk” adults added to U.S. universal PCV vaccination recommendation, which includes >25% of 50-64 year olds3

September 3, 2024 VAXCYTE MISSION STATEMENT We are on a global mission to engineer high-fidelity vaccines that protect humankind from the consequences of bacterial diseases.

September 3, 2024 Q&A with Management Grant Pickering Chief Executive Officer, Director and Founder Jim Wassil Executive Vice President and Chief Operating Officer Andrew Guggenhime President and Chief Financial Officer
