Exhibit 99.2
 | Interim report for the first half of 2020 Zealand Pharma August 13, 2020 |
 | This presentation contains information pertaining to Zealand Pharma A/S (“Zealand"). Neither Zealand nor its management, directors, employees or representatives make any representation or warranty, express or implied, as to the accuracy or completeness of any of the information contained in this presentation or any other information transmitted or made available to the viewer or recipient hereof, whether communicated in written or oral form. This presentation does not constitute or form part of, and should not be construed as, an offer to sell or issue or the solicitation of an offer to buy or acquire Zealand securities, in any jurisdiction, or an inducement to enter into investment activity, nor shall there be any sale of Zealand securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. No part of this presentation, nor the fact of its distribution, should form the basis of, or be relied on in connection with, any contract or commitment or investment decision whatsoever. This presentation contains forward-looking statements that reflect management's current views with respect to Zealand's product candidates' development, clinical and regulatory timelines and anticipated results, market opportunity, potential financial performance and other statements of future events or conditions. Although Zealand believes that the expectations reflected in such forward-looking statements are reasonable, no assurance can be given that such expectations will prove to have been correct. Accordingly, results could differ materially from those set out in the forward-looking statements as a result of various factors, many of which are beyond Zealand’s control. No reliance should be made on such forward-looking statements. Zealand does not intend to update the presentation, including the forward-looking statements contained therein, following distribution, beyond what is required by applicable law or applicable stock exchange regulations if and when circumstances arise that lead to changes compared to the date when these statements were provided. This presentation should be read together with announcement of the half year results for 2020 and the safe harbor provisions and forward looking section of that accompanying company announcement of the half year results for 2020 for Zealand Pharma A/S delivered on 13 August 2020. |
 | Business highlights for H1 2020 and subsequent events •Announced FDA filing and acceptance of New Drug Application (NDA) for the dasiglucagon HypoPal® rescue pen for treatment of severe hypoglycemia •Completed enrollment in the first Phase 3 trial of dasiglucagon for the treatment of congenital hyperinsulinism (CHI) •Expansion of U.S. Commercial Operations through acquisition and integration of business assets of Valeritas Holdings, Inc., opening of new Boston office, and appointing of U.S. leadership •Secured a total of DKK gross 657.7 million through a direct issue and private placement of new shares •EUR 20 million milestone triggered in connection with Boehringer Ingelheim dosing the first patient in June of their Phase 2 trial with BI 456906, a long-acting GLP-1/Glucagon dual agonist, for the treatment of Type 2 diabetes and obesity. |
 | Safety of our people Taking precautions to keep our employees, patients and business and clinical partners safe Research activities Adjusting the way we work so that laboratory-based research and development activities have continued with minimal interruption Clinical trials Continuing enrollment in trials; starting to see previously closed sites opening for new patient enrollment and expect this positive development to continue in the coming months HCP and patient engagement Staying connected with health care providers and patients by leveraging virtual meetings, training, and support. Commercial activities in U.S. focused on continuing to support the business for V-Go® while continuing high level of service |
 | [LOGO] |
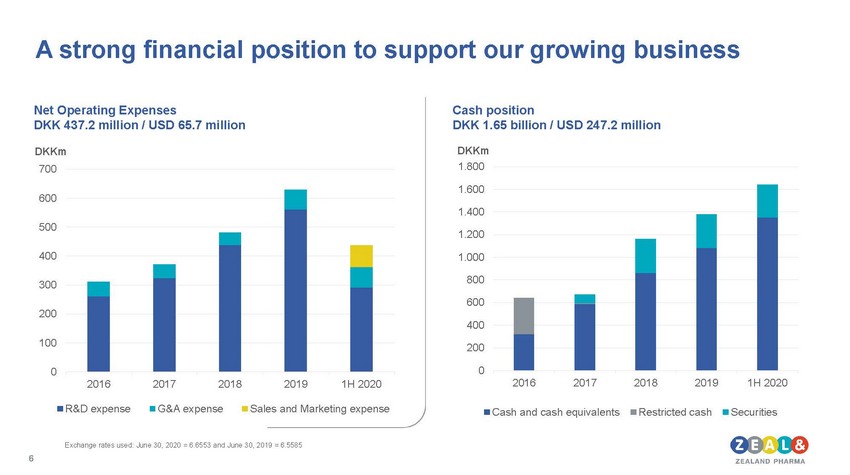 | Net Operating Expenses DKK 437.2 million / USD 65.7 million Cash position DKK 1.65 billion / USD 247.2 million DKKm 700 600 500 400 300 200 100 DKKm 1.800 1.600 1.400 1.200 1.000 800 600 400 200 0 20162017201820191H 2020 R&D expenseG&A expenseSales and Marketing expense 0 20162017201820191H 2020 Cash and cash equivalentsRestricted cashSecurities |
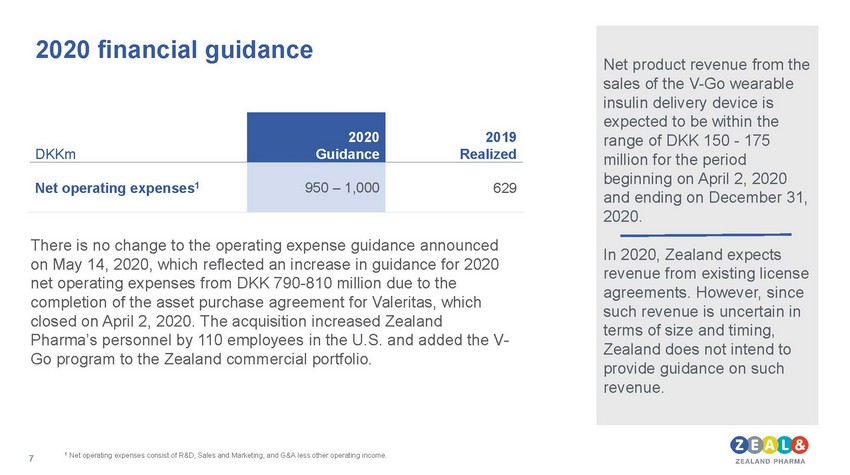 | 2020 financial guidance There is no change to the operating expense guidance announced on May 14, 2020, which reflected an increase in guidance for 2020 net operating expenses from DKK 790-810 million due to the completion of the asset purchase agreement for Valeritas, which closed on April 2, 2020. The acquisition increased Zealand Pharma’s personnel by 110 employees in the U.S. and added the V-Go program to the Zealand commercial portfolio. Net product revenue from the sales of the V-Go wearable insulin delivery device is expected to be within the range of DKK 150 - 175 million for the period beginning on April 2, 2020 and ending on December 31, 2020. In 2020, Zealand expects revenue from existing license agreements. However, since such revenue is uncertain in terms of size and timing, Zealand does not intend to provide guidance on such revenue. |
 | R&D Pipeline Product Candidate Partner Indication Pre-clinicalPhase 1Phase 2Phase 3Registration Upcoming Milestones Dasiglucagon HypoPal® Rescue Pen Dasiglucagon S.C. Continuous Infusion Dasiglucagon Dual-hormone Pump Dasiglucagon Adjustable Mini-Dose Severe hypoglycemia Congenital hyperinsulinism Diabetes management Post bariatric hypoglycemia PDUFA Date March 27, 2021 Q4 2020: Phase 3 Readout 2021: Phase 3 Trial Initiation Glepaglutide GLP-2 Analog ZP7570 GLP-1/GLP-2 Dual Agonist BI 456906 GLP-1/GLU Dual Agonist Amylin Analog Complement C3 Inhibitor ZP10000 α4β7 Integrin Inhibitor Ion Channel Blockers GIP/GLP-1/Glucagon Mono/Dual/Triple Short bowel syndrome Short bowel syndrome Obesity/Type 2 diabetes/NASH1 Undisclosed Undisclosed2 Inflammatory bowel disease3 Undisclosed Undisclosed 2H 2021: Phase 3 Readout Q4 2020: Phase 1a Results 1 Partnered with Boehringer Ingelheim. Zealand eligible for EUR 345m in outstanding milestones; 2 Partnered with Alexion Pharmaceuticals: Zealand eligible for USD 610m in outstanding milestones. 3 8Acquired Encycle Therapeutics, Inc.: future potential earn-outs of up to US $80 million contingent on successful achievement of development, regulatory and commercial milestones; payable in cash and/or ZEAL equity at Zealand’s discretion. |
 | We are pursuing multiple opportunities to improve patients’ lives with dasiglucagon via different product modalities Dasiglucagon Novel stable glucagon analog Fast onset-of-action Unique stability in liquid formulation Suitable for multiple indications HypoPal® rescue pen for severe hypoglycemia NDA filed Infusion pump therapy for recurrent low blood glucose in congenital hyperinsulinism Phase 3 ongoing Dual hormone artificial pancreas for automated diabetes management Phase 3 ready Adjustable mini dose pen for hypoglycemia in type I diabetes and following bariatric surgery Phase 2a results ~300,000 hospitalizations annually in the U.S. 1 ~300 newborns diagnosed every year in the U.S. and EU2,3 ~400,000 patients in 20184 ~500 patients in 20185,6 1 National Diabetes Statistics Report. CDC. 2014 2 https://www.orpha.net/consor/cgi-bin/ (not including transient cases due to perinatal stress or diabetic mother) 3 Congenital Hyperinsulinism International. Available at: http://congenitalhi.org 9 5 https://asmbs.org/resources/estimate-of-bariatric-surgery-numbers |
 | Severe hypoglycemia is one of the most feared complications for diabetes patients on insulin therapy1 Our Solution Dasiglucagon HypoPal® – a stable glucagon analog for fast rescue treatment of severe hypoglycemia For illustration only Fast onset of action: Three Phase 3 trials met all primary and key secondary endpoints, with median time to recovery of only 10 minutes Preferred mode of administration by patients, care givers and HCPs1 New Drug Application filed with U.S. FDA; expected approval decision March 27, 2021 Expected U.S. launch in 2021 |
 | Congenital hyperinsulinism (CHI) is an ultra-rare and devastating congenital disorder in newborns Our Solution A potential first-in-class glucagon analog for short-and long-term glycemic stabilization of children with CHIFor illustration only Phase 3 trials in children with CHI: First Phase 3 with 32 children age 3 months to 12 years •Completed enrollment in the first Phase 3 trial; results expected late this year Second Phase 3 in up to 12 neonates age 7 days to 1 year •First patients enrolled Extension study for all children •Ongoing EU and U.S. orphan drug designation granted •Less hypoglycemic events •Longer fasting intervals •Less dependence on i.v. glucose •Long-term safety and efficacy outcomes |
 | Short bowel syndrome is a chronic and debilitating disease affecting up to 40,000 people in the U.S. and Europe1,2 Rare and severe disease that impairs intestinal absorption, diarrhea and metabolic complications3 Life-long dependency Complex parenteral support to survive and risk of life-threatening infections and extra-organ impairment4 Need for better treatments Faster and reliable treatment for reduction of parenteral support needs Our Solution Glepaglutide: a long-acting GLP-2 analog for treatment of short bowel syndromeFor illustration only Effective and well-tolerated in Phase 2, suggesting a fast and reliable treatment5 Targets once-weekly dosing via autoinjector •Long acting with effective half-life of ~50 hours Phase 3 ongoing with results expected in 2nd Half of 2021 |
 | 2020 is a significant year of Zealand 5x25 5 commercialized products by 2025 2020 Commercial foundation established 4 Late stage assets and robust early pipeline 2 License partnerships 313 Employees Fully integrated biotech with U.S. commercial presence Commercial platform in place to launch metabolic and gastrointestinal franchises Three late-stage assets for metabolic diseases, one for GI diseases Boehringer Ingelheim and Alexion Pharmaceuticals Offices in Copenhagen, New York, Boston, and Marlborough |
 | ZEALAND PHARNA |