Exhibit 99.2

TWYNEO® PHASE 3 RESULTS

This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “future,” “outlook,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” “continue,” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. The forward-looking statements in this presentation relate to, among other things, statements regarding the commencement of our planned bioequivalence study for a generic product candidate, our expected date to report top-line data from our pivotal Phase III clinical program for Twyneo®, our anticipated NDA submission dates for Epsolay® and Twyneo®, and estimated sales of our product candidates. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties, and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by the forward-looking statement, including but not limited to the following: the fact that we have and expect to continue to incur significant losses; our need for additional funding, which may not be available; our ability to complete the development of our product candidates; our ability to obtain and maintain regulatory approvals for our product candidates in our target markets and the possibility of adverse regulatory or legal actions relating to our product candidates even if regulatory approval is obtained; our ability to commercialize our product candidates; our ability to obtain and maintain adequate protection of our intellectual property; our ability to manufacture our product candidates in commercial quantities, at an adequate quality or at an acceptable cost; our ability to establish adequate sales, marketing, and distribution channels; acceptance of our product candidates by healthcare professionals and patients; the possibility that we may face third-party claims of intellectual property infringement; the timing and results of clinical trials that we may conduct or that our competitors and others may conduct relating to our or their products; intense competition in our industry, with competitors having substantially greater financial, technological, research and development, regulatory and clinical, manufacturing, marketing, and sales, distribution and personnel resources than we do; potential product liability claims; potential adverse federal, state, and local government regulation in the United States, Europe, or Israel; and loss or retirement of key executives and research scientists. These and other important factors discussed in the Company's Annual Report on Form 20-F filed with the Securities and Exchange Commission (“SEC”) on March 21, 2019, and our other reports filed with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management’s estimates as of the date of this presentation. While we may elect to update such forward-looking statements at some point in the future, unless required by applicable law, we disclaim any obligation to do so, even if subsequent events cause our views to change. Thus, one should not assume that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this presentation. This presentation contains trademarks, trade names, and service marks of other companies, which are the property of their respective owners. We do not intend our use or display of other parties' trademarks, trade names, or service marks to imply, and such use or display should not be construed to imply a relationship with, or endorsement or sponsorship of us by, these other parties. Cautionary Note on Forward-looking statements

A multifactorial disease of the pilosebaceous unit, involving abnormalities in sebum production, follicular epithelial desquamation, bacterial proliferation, and inflammation Topical BPO, retinoids, antibiotics, and their combinations; isotretinoin and antibiotics are mainstays of systemic therapy Insufficient efficacy negatively affects self-esteem; contributes to antibiotic resistance; systemic side effects What isacne vulgaris? How is it treated? What are the current treatment shortfalls? Acne vulgaris Multifactorial disease requiring powerful combination treatments Our solution: TWYNEO®E-BPO + E-ATRA Cream Encapsulation allows combining 2 highly effective APIs, BPO and ATRA, that have complementary mechanisms of actionEncapsulation may reduce the irritation of both BPO and ATRAPotential to be more effective than existing topical treatments
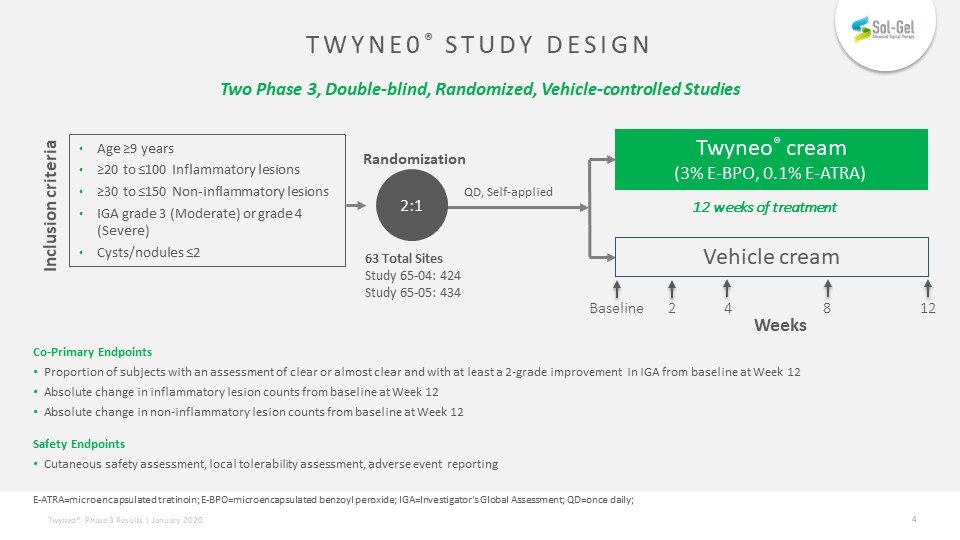
Co-Primary EndpointsProportion of subjects with an assessment of clear or almost clear and with at least a 2-grade improvement in IGA from baseline at Week 12Absolute change in inflammatory lesion counts from baseline at Week 12Absolute change in non-inflammatory lesion counts from baseline at Week 12Safety EndpointsCutaneous safety assessment, local tolerability assessment, adverse event reporting QD, Self-applied Twyneo® cream(3% E-BPO, 0.1% E-ATRA) Vehicle cream Age ≥9 years≥20 to ≤100 Inflammatory lesions ≥30 to ≤150 Non-inflammatory lesionsIGA grade 3 (Moderate) or grade 4 (Severe)Cysts/nodules ≤2 2:1 Randomization Inclusion criteria 12 weeks of treatment E-ATRA=microencapsulated tretinoin; E-BPO=microencapsulated benzoyl peroxide; IGA=Investigator's Global Assessment; QD=once daily; Baseline 2 4 8 12 Weeks 63 Total SitesStudy 65-04: 424Study 65-05: 434 Twyne0® Study design Two Phase 3, Double-blind, Randomized, Vehicle-controlled Studies

Study 65-04 Study 65-05 Number of sites 32 31 Twyneo®(n=281) Vehicle (n=143) Twyneo®(n=290) Vehicle (n=144) Age, yearsMean (SD)Median (range) 20.9 (8.48)18.0 (11-67) 21.4 (8.62)18.0 (10-57) 20.1 (6.96)18.0 (10-51) 20.3 (6.67)18.5 (9-42) Sex, n (%)MaleFemale 106 (37.7%)175 (62.3%) 60 (42.0%)83 (58.0%) 117 (40.3%)173 (59.7%) 67 (46.5%)77 (53.5%) Ethnicity, n (%)Hispanic/LatinoNot Hispanic or LatinoUnknown/Not Reported 102 (36.3%)178 (63.3%)1 (0.4%) 44 (30.8%)98 (68.5%)1 (0.7%) 85 (29.3%)204 (70.3%)1 (0.3%) 56 (38.9%)87 (60.4%)1 (0.7%) IGA severity Moderate Severe 251 (89.3%)30 (10.7%) 132 (92.3%)11 (7.7%) 262 (90.3%)28 (9.7%) 133 (93.0%)10 (7.0%) Inflammatory lesion countMean (SD)Median (range) 33.5 (14.62)28.0 (20-92) 33.5 (14.69)28.0 (20-90) 28.2 (8.70)25.0 (20-62) 27.5 (8.52)25 (20-75) Non-inflammatory lesion countMean (SD)Median (range) 48.6 (20.24)42.0 (30-148) 47.1 (19.97)41.0 (30-140) 44.6 (18.03)39.0 (23-149) 44.9 (18.82)38.0 (30-123) Well-balanced studies at Baseline (ITT)
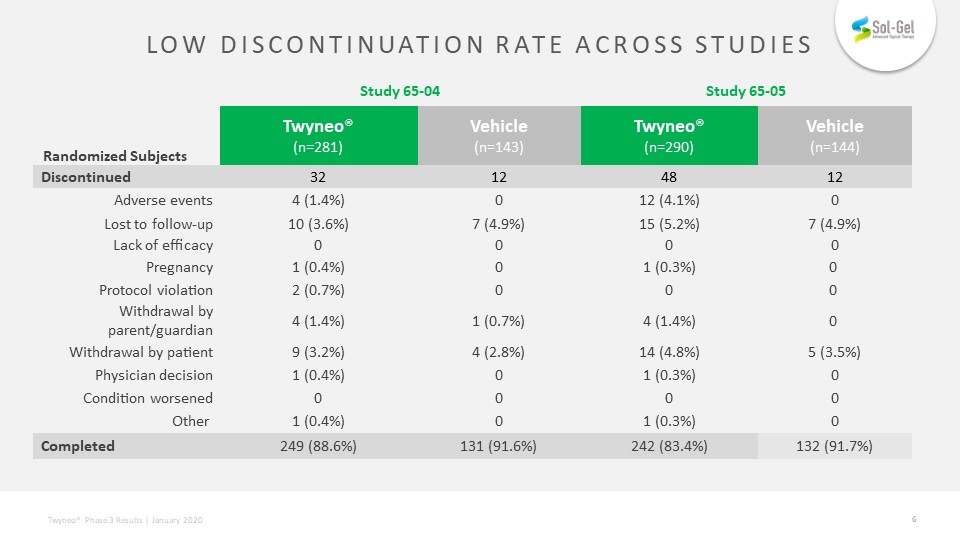
Low discontinuation rate across studies Study 65-04 Study 65-05 Twyneo®(n=281) Vehicle (n=143) Twyneo®(n=290) Vehicle (n=144) Randomized Subjects Discontinued 32 12 48 12 Adverse events 4 (1.4%) 0 12 (4.1%) 0 Lost to follow-up 10 (3.6%) 7 (4.9%) 15 (5.2%) 7 (4.9%) Lack of efficacy 0 0 0 0 Pregnancy 1 (0.4%) 0 1 (0.3%) 0 Protocol violation 2 (0.7%) 0 0 0 Withdrawal by parent/guardian 4 (1.4%) 1 (0.7%) 4 (1.4%) 0 Withdrawal by patient 9 (3.2%) 4 (2.8%) 14 (4.8%) 5 (3.5%) Physician decision 1 (0.4%) 0 1 (0.3%) 0 Condition worsened 0 0 0 0 Other 1 (0.4%) 0 1 (0.3%) 0 Completed 249 (88.6%) 131 (91.6%) 242 (83.4%) 132 (91.7%)
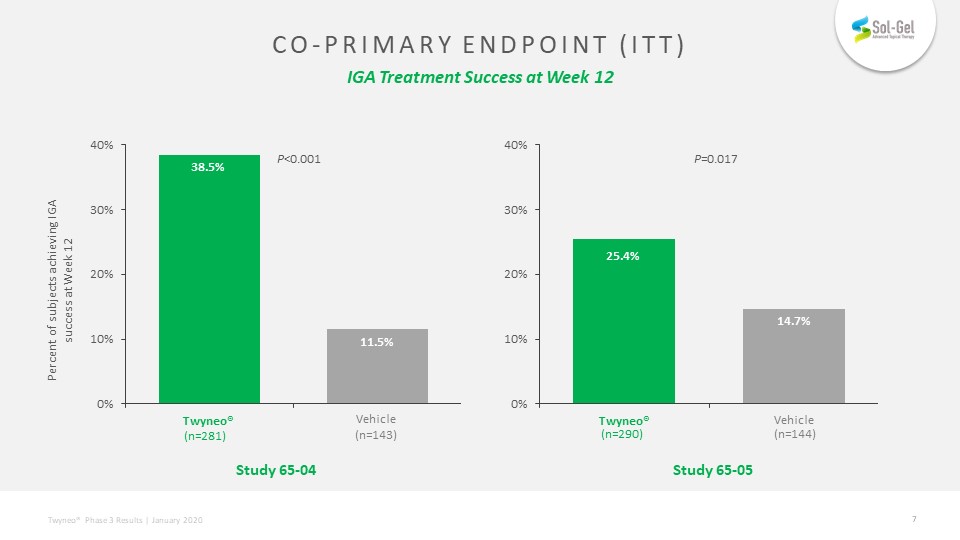
Co-primary endpoint (itt) IGA Treatment Success at Week 12 Percent of subjects achieving IGA success at Week 12 P<0.001 Study 65-04 Vehicle(n=143) Vehicle P=0.017 Study 65-05 (n=290) (n=144) (n=281)

Co-primary endpoint (itt) Absolute Mean Change From Baseline in Inflammatory Lesions at Week 12 P<0.001 P=0.018 Study 65-04 Study 65-05 Mean reduction in inflammatory lesion count from baseline at Week 12 Vehicle(n=143) (n=281) (n=290) Vehicle(n=144)
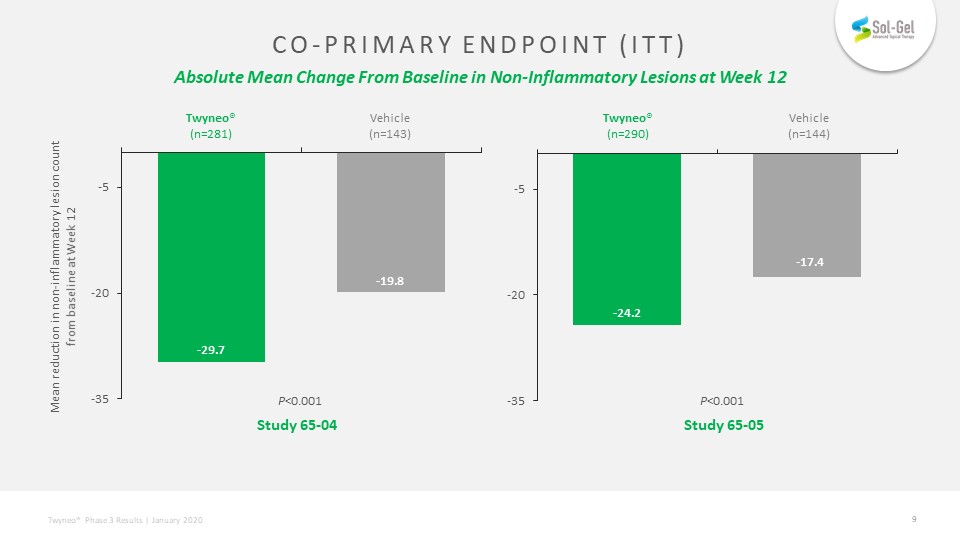
Co-primary endpoint (itt) Absolute Mean Change From Baseline in Non-Inflammatory Lesions at Week 12 Mean reduction in non-inflammatory lesion count from baseline at Week 12 Study 65-04 Study 65-05 P<0.001 P<0.001 Vehicle(n=143) Vehicle(n=144) Twyneo®(n=281) Twyneo®(n=290)
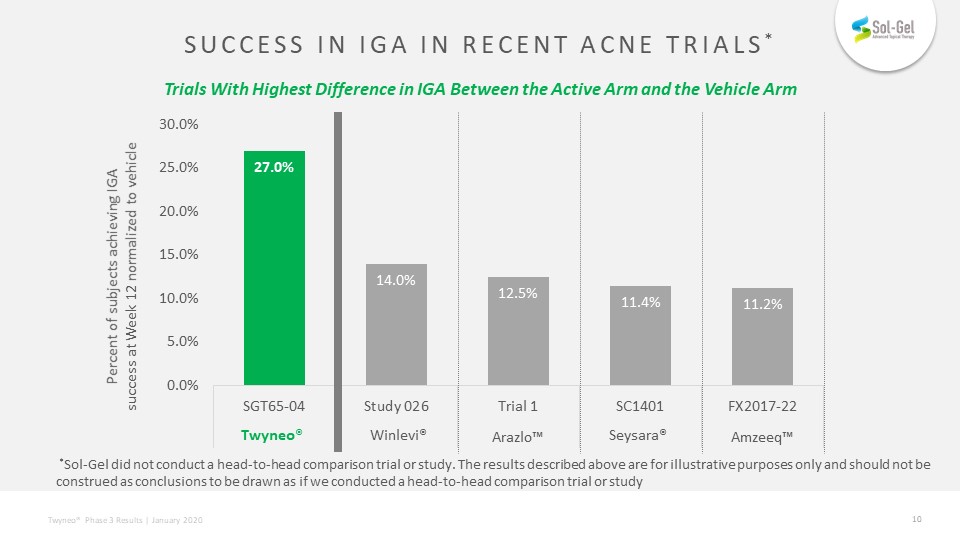
SUCCESS IN IGA IN RECENT ACNE TRIALS* Trials With Highest Difference in IGA Between the Active Arm and the Vehicle Arm *Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study Percent of subjects achieving IGA success at Week 12 normalized to vehicle Twyneo® Winlevi® Seysara®
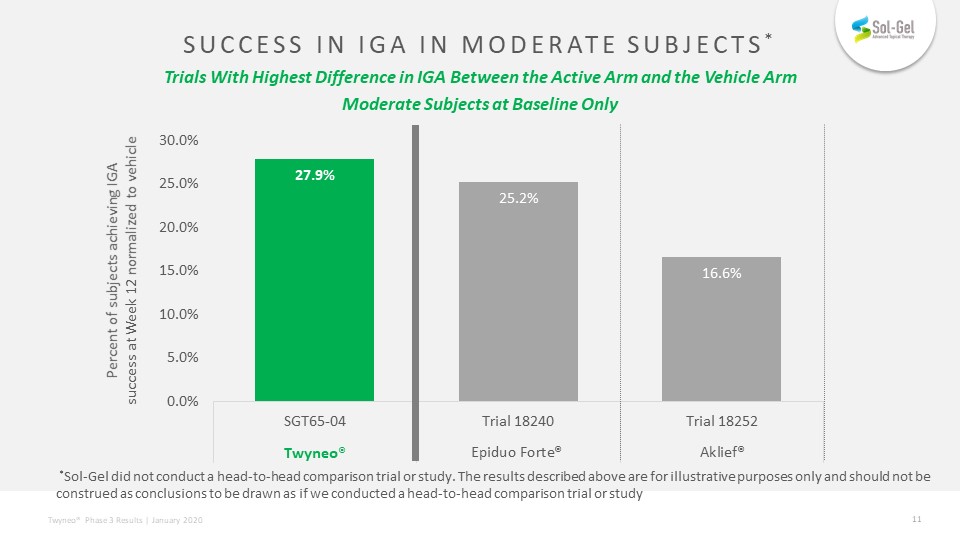
SUCCESS IN IGA IN MODERATE Subjects* Trials With Highest Difference in IGA Between the Active Arm and the Vehicle ArmModerate Subjects at Baseline Only Percent of subjects achieving IGA success at Week 12 normalized to vehicle Twyneo® *Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study
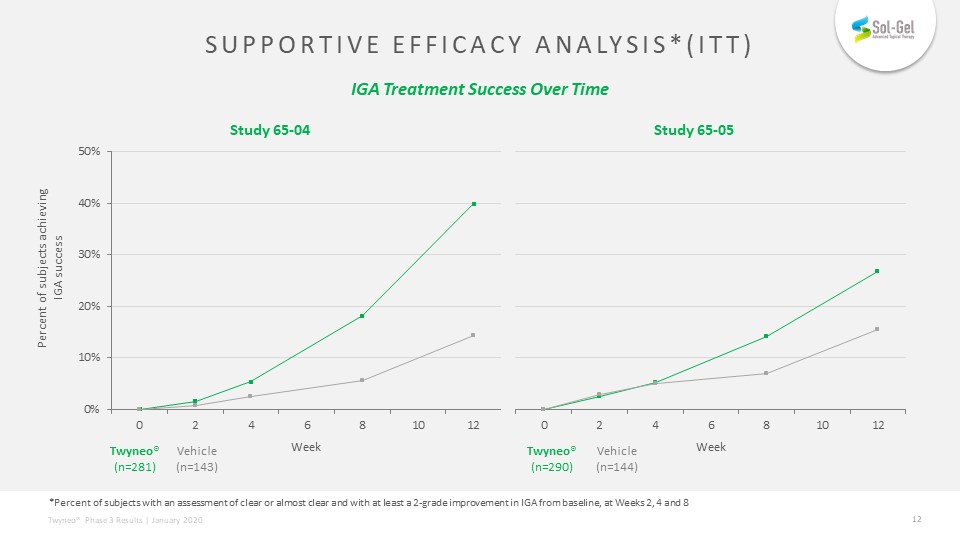
Twyneo®(n=281) Supportive efficacy analysis*(itt) IGA Treatment Success Over Time Percent of subjects achieving IGA success Week Week Study 65-04 Study 65-05 *Percent of subjects with an assessment of clear or almost clear and with at least a 2-grade improvement in IGA from baseline, at Weeks 2, 4 and 8 Vehicle(n=143) Twyneo®(n=290) Vehicle(n=144)
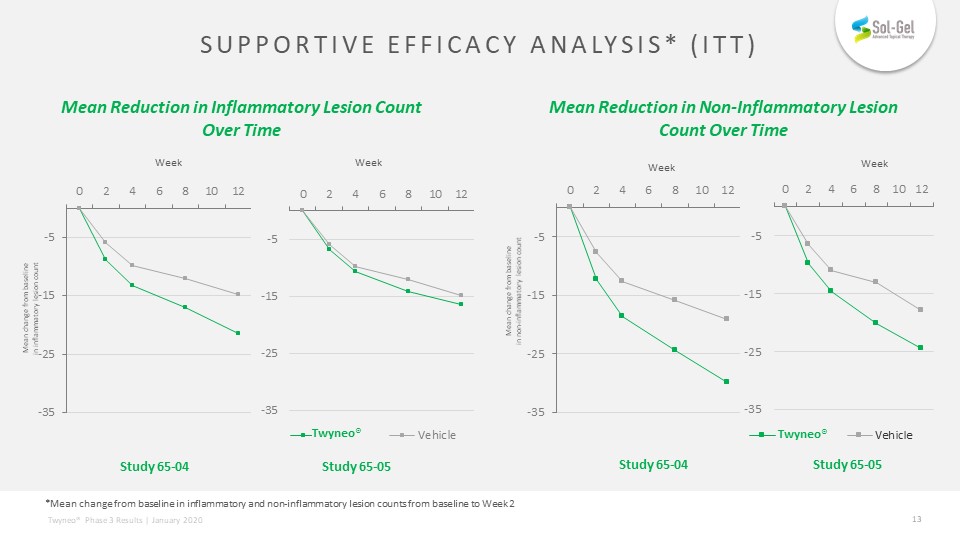
Supportive efficacy analysis* (itt) Mean Reduction in Non-Inflammatory Lesion Count Over Time Mean change from baseline in non-inflammatory lesion count Study 65-04 Study 65-05 Week Week Mean change from baseline in inflammatory lesion count Study 65-04 Study 65-05 Week Week Mean Reduction in Inflammatory Lesion Count Over Time *Mean change from baseline in inflammatory and non-inflammatory lesion counts from baseline to Week 2 Twyneo® Twyneo®
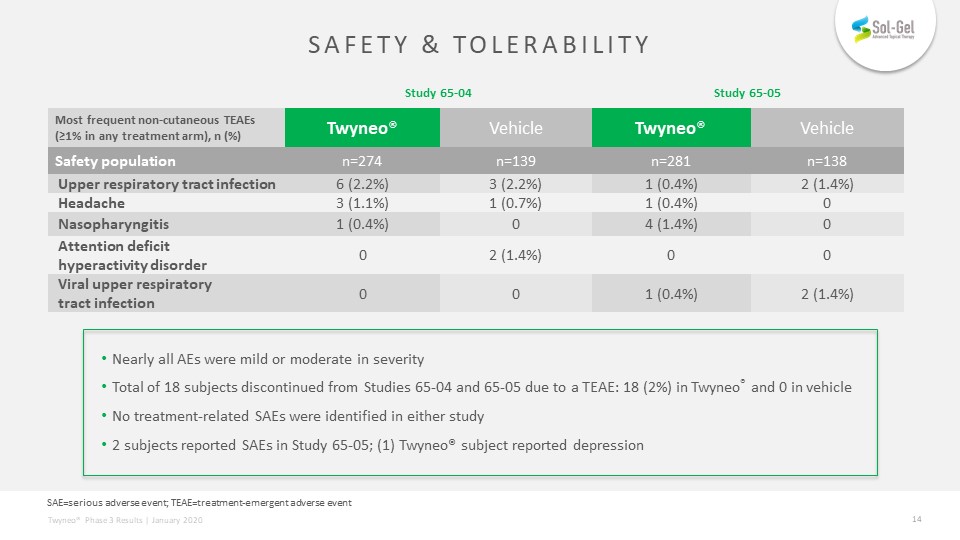
Safety & tolerability Nearly all AEs were mild or moderate in severityTotal of 18 subjects discontinued from Studies 65-04 and 65-05 due to a TEAE: 18 (2%) in Twyneo® and 0 in vehicle No treatment-related SAEs were identified in either study2 subjects reported SAEs in Study 65-05; (1) Twyneo® subject reported depression Study 65-04 Study 65-05 Most frequent non-cutaneous TEAEs (≥1% in any treatment arm), n (%) Twyneo® Vehicle Twyneo® Vehicle Safety population n=274 n=139 n=281 n=138 Upper respiratory tract infection 6 (2.2%) 3 (2.2%) 1 (0.4%) 2 (1.4%) Headache 3 (1.1%) 1 (0.7%) 1 (0.4%) 0 Nasopharyngitis 1 (0.4%) 0 4 (1.4%) 0 Attention deficithyperactivity disorder 0 2 (1.4%) 0 0 Viral upper respiratorytract infection 0 0 1 (0.4%) 2 (1.4%) SAE=serious adverse event; TEAE=treatment-emergent adverse event

Local Skin Tolerability assessment* at week 12 *Safety population Study 65-04 Study 65-05
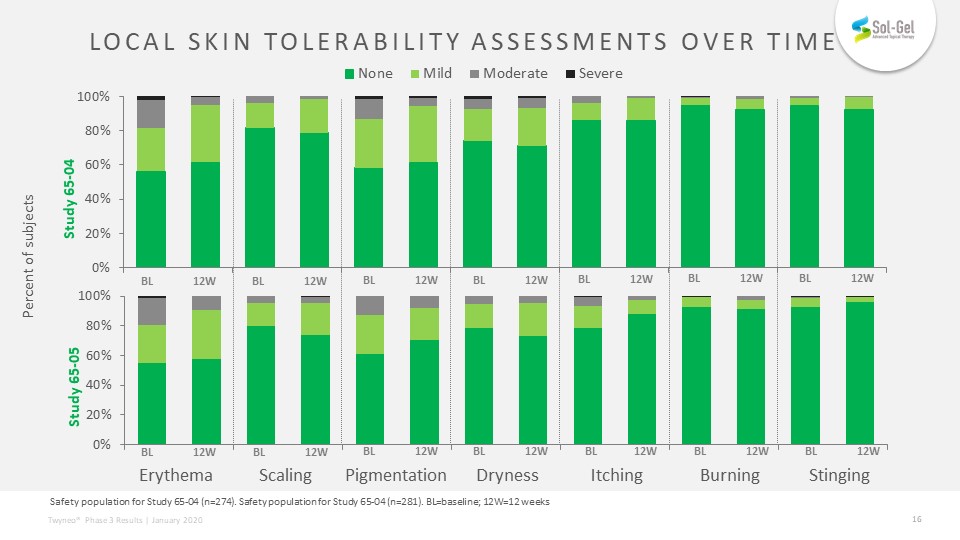
Local skin tolerability assessments over time Safety population for Study 65-04 (n=274). Safety population for Study 65-04 (n=281). BL=baseline; 12W=12 weeks Erythema Scaling Pigmentation Dryness Itching Burning Stinging Percent of subjects BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W BL 12W Study 65-05 Study 65-04 BL 12W BL 12W BL 12W

Twyneo® phase 3 results Successfully met all primary efficacy endpoints demonstrating statistically significant improvements over vehicleNo treatment-related seriousadverse events Well-tolerated, with results similarto vehicle at 12 weeks

Introducing Hilary baldwin, MD Past President of the American Acne and Rosacea Society, Clinical Associate Professor of Dermatology at Rutgers Robert Wood Johnson School of Medicine, and Director of the Acne Treatment and Research Center.
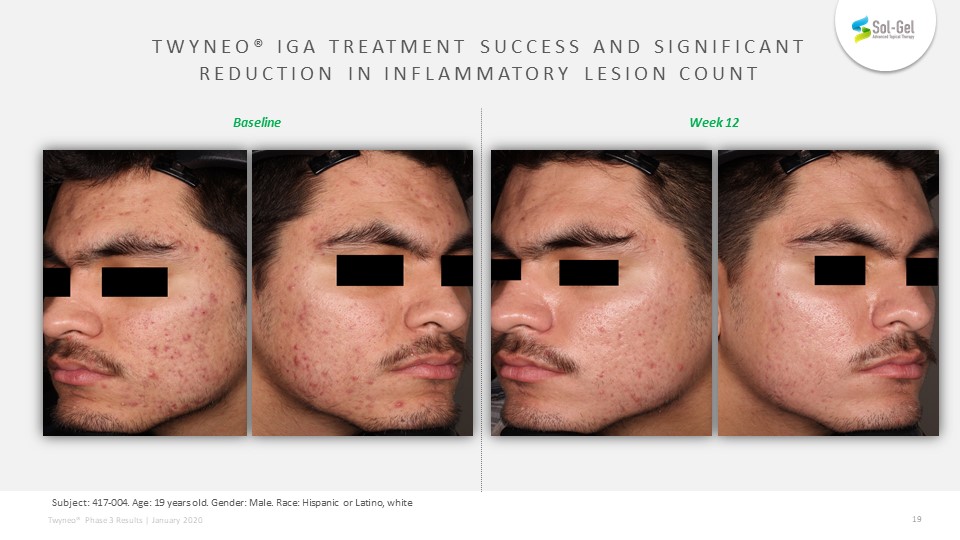
TWYNEO® IGA treatment success and significantreduction in Inflammatory Lesion Count TEAE definition: Subject: 417-004. Age: 19 years old. Gender: Male. Race: Hispanic or Latino, white Week 12 Baseline
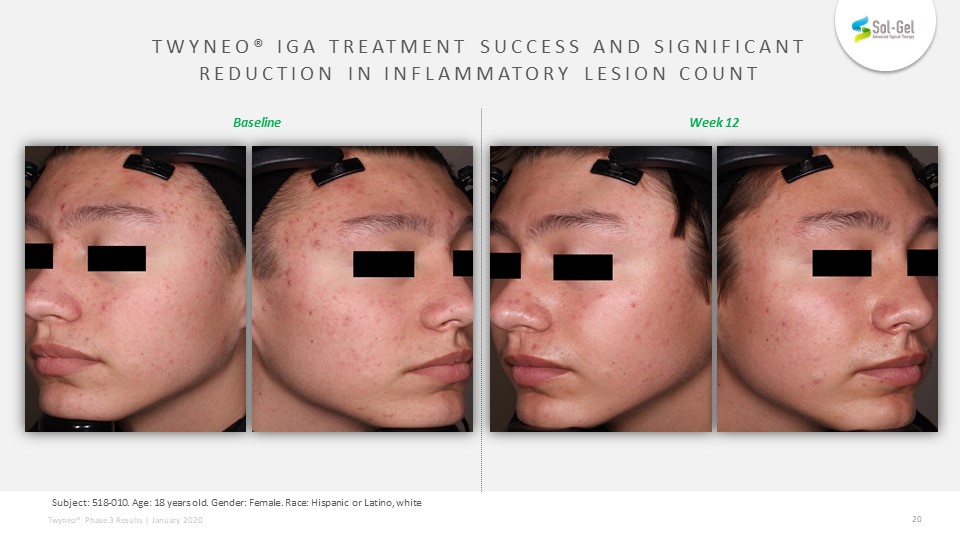
TEAE definition: Subject: 518-010. Age: 18 years old. Gender: Female. Race: Hispanic or Latino, white Week 12 Baseline TWYNEO® IGA treatment success and significantreduction in Inflammatory Lesion Count
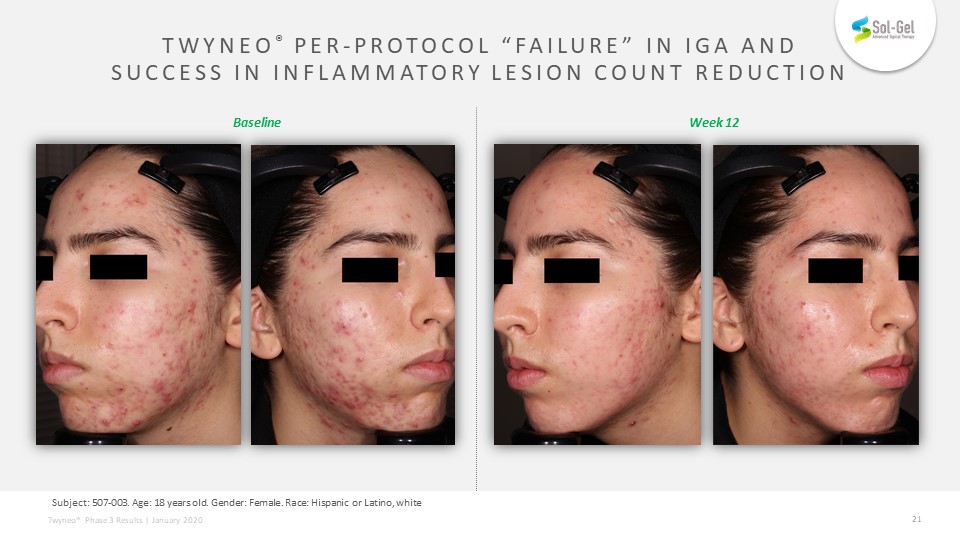
TEAE definition: Subject: 507-003. Age: 18 years old. Gender: Female. Race: Hispanic or Latino, white TWYNEO® Per-Protocol “Failure” in IGA and Success in Inflammatory Lesion Count Reduction Week 12 Baseline
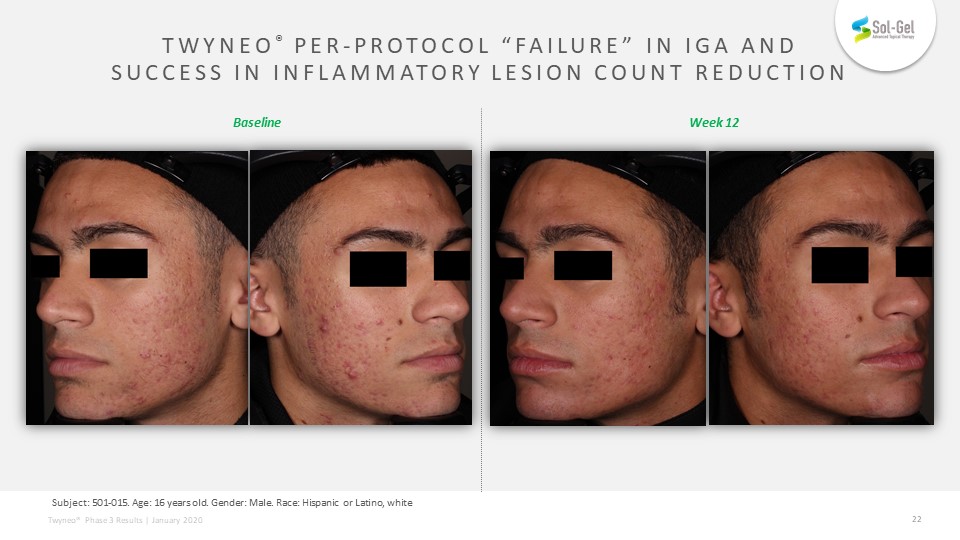
TWYNEO® Per-Protocol “Failure” in IGA and Success in Inflammatory Lesion Count Reduction TEAE definition: Subject: 501-015. Age: 16 years old. Gender: Male. Race: Hispanic or Latino, white Week 12 Baseline

Questions