Exhibit 99.1

Corporate Presentation September 2020 Nasdaq: ADTX Monitoring and Reprogramming the Immune System aditxt.com

2 aditxt.com This presentation contains forward - looking statements that are subject to many risks and uncertainties . Forward looking statements include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things, our ongoing and planned product development ; our intellectual property position ; our ability to develop commercial functions ; expectations regarding product launch and revenue ; our results of operations, cash needs, spending, financial condition, liquidity, prospects, growth and strategies ; the industry in which we operate ; and the trends that may affect the industry or us . Although we believe we have a reasonable basis for each forward - looking statement, we caution you that forward - looking statements are not guarantees of future performance . Actual results may differ materially from those indicated by these forward - looking statements as a result of various important factors, as well as those risks more fully discussed in the section entitled “Risk Factors” in the Company’s prospectus, dated June 29 , 2020 , that was filed with the U . S . Securities and Exchange Commission under File No . 333 - 235933 , as well as discussions of potential risks, uncertainties, and other important factors in the Company’s subsequent filings with the U . S . Securities and Exchange Commission . All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward - looking statements, whether as a result of new information, future events or otherwise .

3 aditxt.com Overview Developing biotechnologies focused on improving the health of the immune system through immune monitoring and reprogramming Market Data * Began trading on June 30, 2020 SYMBOL NASDAQ: ADTX Price as of 09 - 02 - 2020 $2.67 52 Week Range* $2.35 - $9.58 Average Volume ~2.3M Market Capitalization $18.6M Current Shares Outstanding ~7.1M

4 aditxt.com • Advanced diagnostic platform for rapidly monitoring the immune system to anticipate response and possible reaction to viruses, bacteria, allergens, and transplanted organs (Invented at Stanford University and covered by a U.S. Patent. • Professional team with extensive experience in translation of research technologies into clinical practice providing direct access to clinical markets by production of LDTs (physician - based, population health, business - to - business, and direct - to - consumer models) utilizing behavioral health approaches • Commercialization efforts driven by advanced technologies encompassing many different chronic and infectious diseases, empowering consumers to monitor and take more control of their health Two Timely Biotechnologies AditxtMonitoring TM aditxt.com

5 aditxt.com AditxtReprogramming TM • Pioneering Selective Tolerance (ST) therapeutic technology developed over the past 20+ years at Loma Linda University • 7 U.S. patents and 59 foreign patents as of April 2020, 18 additional pending • Demonstrated preclinical efficacy for Skin Grafting, Psoriasis, Type 1 Diabetes, and Alopecia Areata • Clinical Trial pipeline leading to licensed products in multiple therapeutic areas Two Timely Biotechnologies

aditxt.com

7 aditxt.com Challenge and Needs The fact is that every person needs to monitor their immune system as evidenced by the COVID - 19 pandemic We need: More Meaningful… More Timely Information…

8 aditxt.com • A proprietary platform that provides a personalized comprehensive profile of the immune system which can assist the medical community in anticipating responses and possible reaction to viruses, bacteria, allergens, and transplanted organs. • Can be useful in anticipating attacks on the body by having the ability to determine its potential response and for developing a plan to deal with an undesirable reaction by the immune system. • Initial technology was licensed from, invented and used at Stanford University. It encompasses methods, systems and kits for detection and measurement of specific immune responses as described within the company’s U.S. Patent. Introducing AditxtScore™
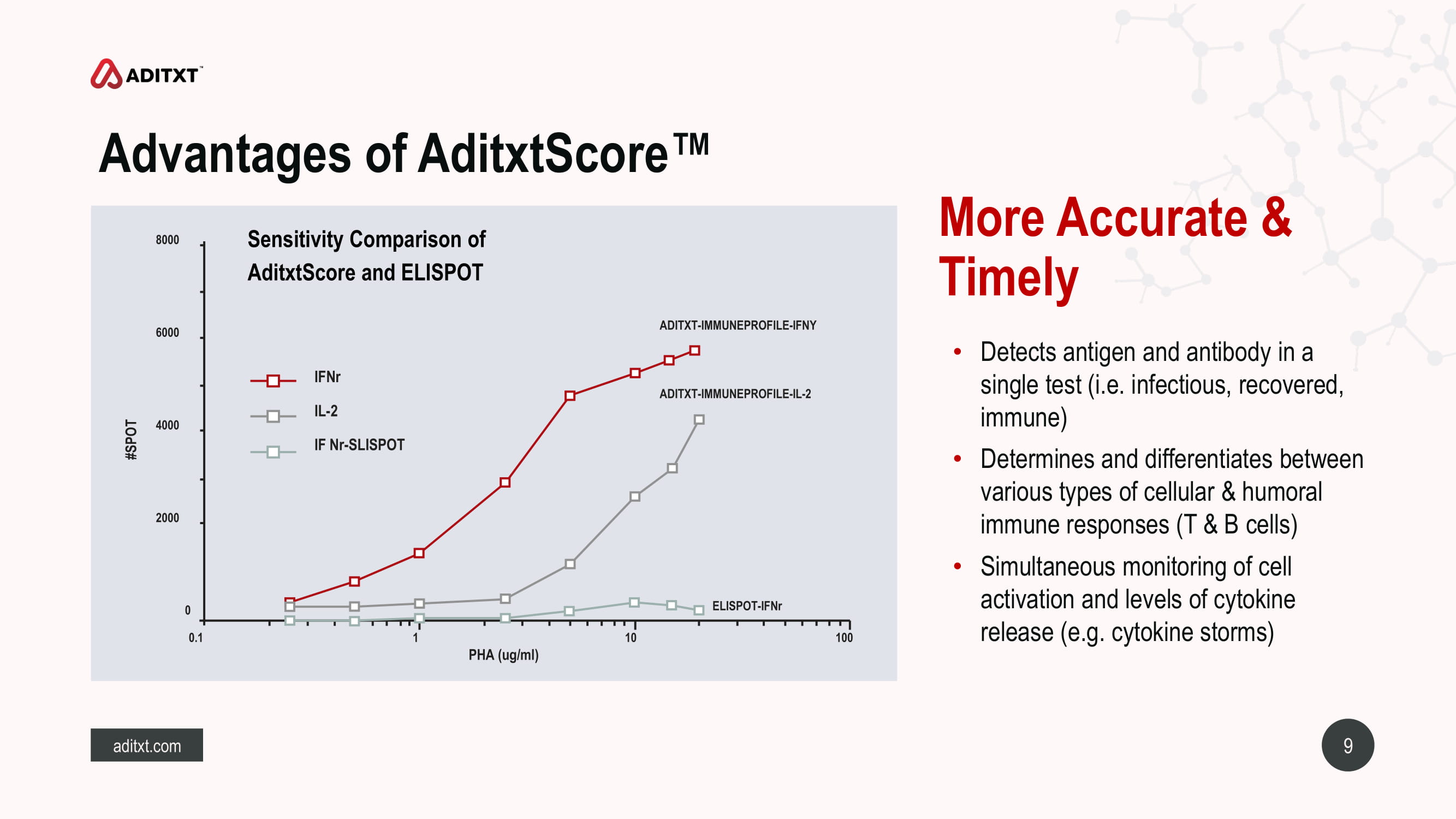
9 aditxt.com • Detects antigen and antibody in a single test (i.e. infectious, recovered, immune) • Determines and differentiates between various types of cellular & humoral immune responses (T & B cells) • Simultaneous monitoring of cell activation and levels of cytokine release (e.g. cytokine storms) Advantages of AditxtScore™ More Accurate & Timely Sensitivity Comparison of AditxtScore and ELISPOT 8000 6000 4000 2000 0 0.1 1 10 100 ELISPOT - IFNr ADITXT - IMMUNEPROFILE - IL - 2 #SPOT PHA (ug/ml) ADITXT - IMMUNEPROFILE - IFNY IFNr IL - 2 IF Nr - SLISPOT
10 aditxt.com AditxtScore™ Applications

11 aditxt.com • AditxtScore™ for COVID - 19 is a double - multiplex assay that can be used to detect and differentiate various antibody isotypes (IgG, IgM, IgA) against multiple SARS - CoV - 2 antigens (e.g. RBD, S1, NP) simultaneously in a single reaction • Results validated by Stanford Blood Center • “The AditxtScore™ for COVID - 19 has the potential to be a game changer for our industry. AditxtScore™ for COVID - 19 is a multiplexed platform that has shown tremendous speed and accuracy, which is extremely critical for meeting the ongoing COVID - 19 challenge”. Harpreet Sandhu, CEO of Stanford Blood Center AditxtScore™ for COVID - 19

12 aditxt.com Strategic Collaborative Agreement with Fully Compliant CLIA and CAP Accredited Laboratory Provides 24/7 Operational Monitoring Capabilities and Supports Regulatory Pathway for AditxtScore Launch

13 aditxt.com AditxtMonitoring™ Sales Channels

aditxt.com

15 aditxt.com 1 https://www.prnewswire.com/news - releases/organ - transplant - immunosuppressant - drugs - market - worth - 5 - 88 - billion - by - 2026 - grand - view - research - inc - 300942663.html 2 https://www.prnewswire.com/news - releases/the - global - autoimmune - disease - therapeutics - market - size - is - expected - to - reach - 149 - 4 - bill ion - by - 2025 -- rising - at - a - market - growth - of - 4 - 34 - cagr - during - the - forecast - period - 300902336.html 3 https://www.grandviewresearch.com/press - release/global - allergy - diagnostics - therapeutics - market 4 https://www.grandviewresearch.com/press - release/global - in - vitro - diagnostics - market Market Opportunity Organ Transplant Immunosuppressant Drugs $5.88B 1 by 2026 Allergy Diagnostics & Therapeutics $51.95B 3 by 2026 Autoimmune Disease Therapeutics $149.4B 2 by 2026

16 aditxt.com Immune Reprogramming Background • Immune reprogramming (immune tolerance) therapies reprogram the immune system so that disease - causing immune responses are stopped while maintaining the immune system’s ability to combat pathogenic infection. • Tolerance is achievable as demonstrated clinically via chimerism and cell - based therapy but there is a need for more practical and cost - effective approaches which: x Can be made into a product x Do not require additional hospitalization x Are simple to produce and ship
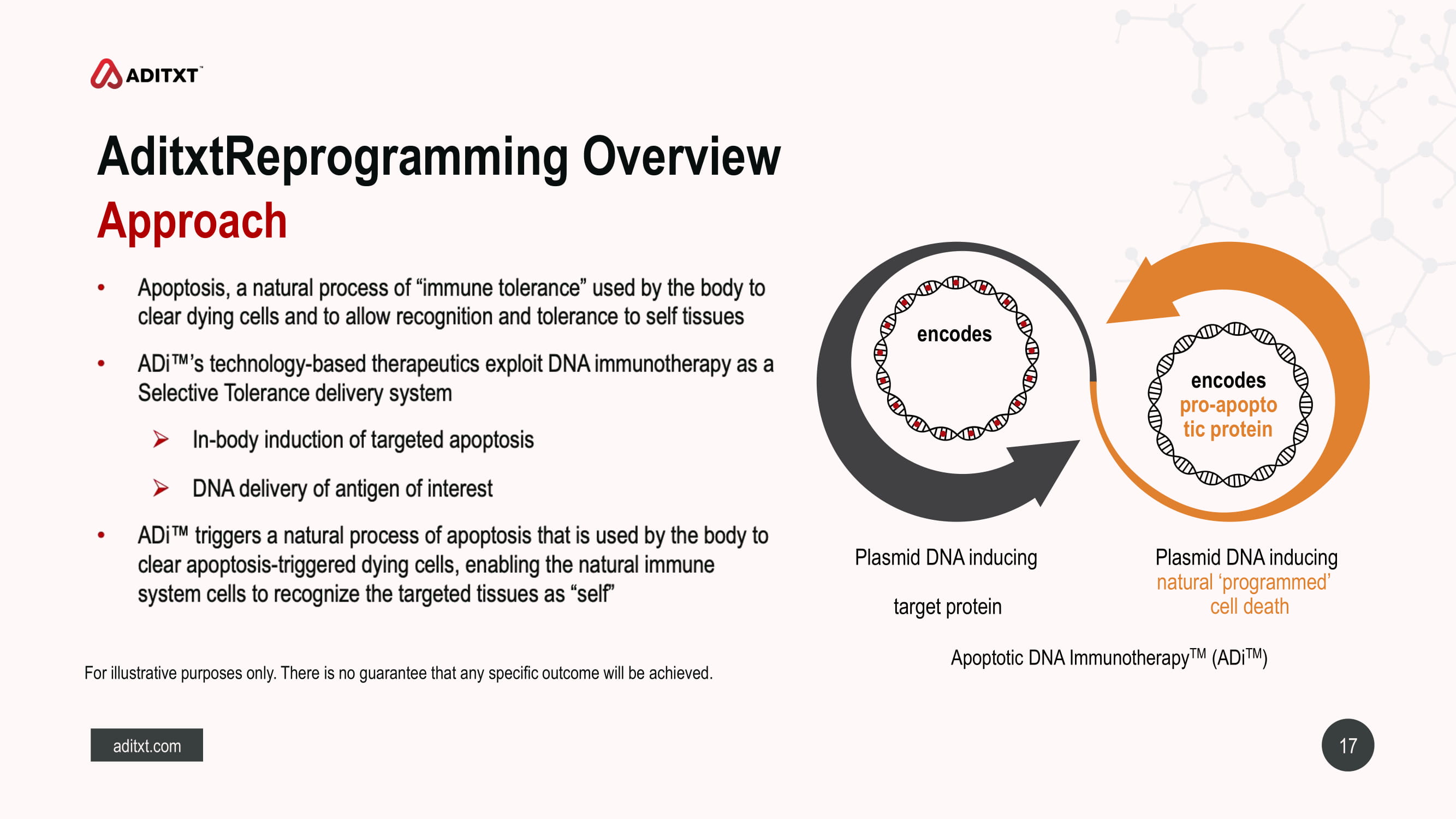
17 aditxt.com AditxtReprogramming Overview For illustrative purposes only. There is no guarantee that any specific outcome will be achieved. Approach encodes encodes pro - apopto tic protein Plasmid DNA inducing natural ‘programmed’ cell death Plasmid DNA inducing target protein Apoptotic DNA Immunotherapy TM ( ADi TM )

18 aditxt.com Apoptotic DNA Immunotherapy (ADi TM ) CLICK TO PLAY • Over 20 years of R&D • $5M total funding including $3M in DOD grant* *Funding was received prior to Aditxt through LLU • Apoptotic DNA Immunotherapy TM (ADi TM ) was developed under the leadership of the late Dr. Leonard Bailey at Loma Linda University (LLU) • Dr. Bailey was a Surgeon - in - Chief of LLU Children’s Hospital and a pioneer in organ transplantation ▪ Developed vision for infant heart transplantation for babies with hypoplastic left heart syndrome ▪ Performed first successful human to human heart transplantation in a newborn baby ▪ Headed LLU’s transplant team that conducted preclinical development of therapeutic solutions to modulate the immune system in order to accept transplanted organs Co - founder and Former Chairman ADi™ Technology background (pre Aditxt™):

19 aditxt.com Intellectual Property Portfolio ADi™ technology is protected by 7 patent families, including: • 7 U.S. patents, 3 U.S. pending patent applications, 59 foreign patents, and 15 foreign pending patent applications (EU, Australia, Canada, Japan, China, India, Hong Kong) ADi™ patents are broadly categorized into 3 groups: • Autoimmune diseases and type 1 diabetes • Organ transplantation and a method of producing plasmid DNA that is mammalian - like to prevent immune activation • Composition of matter for a tolerance delivery system for antigens of interest (basis for a platform allowing development of a new class of immunotherapeutics for various indications) The projected expiration dates for the ADi™ patents range from 2021 to 2032. We also possess and/or in - license substantial know - how and trade secrets relating to the development and commercialization of our product candidates, including related manufacturing processes and technology.

20 aditxt.com ADi TM Proof - of - Concept for Skin Allograft Transplantation 21 Days 58 Days Allograft lost in 21 days on average Immunosuppression alone 3 - fold increase in the longevity of the skin allografts ADi™ with minimal immunosuppression Mouse Model: Donor and recipient mice were genetically mismatched “A therapeutic DNA vaccination strategy for autoimmunity and transplantation,” published in Vaccine 28 (2010) 1897 - 1904. Studies conducted at Loma Linda University. IMMUNOSUPPRESION ALONE TREATMENT WITH ADi TM TECHNOLOGY (minimal immunosupresion)

21 aditxt.com ADi TM Proof - of - Concept for Psoriasis (Autoimmunity) • Psoriasis causes increased skin thickness and scaling in an established 10 - day psoriasis model • ADi TM treatment resulted in a 69% reduction in skin thickening and 38% reduction in scaling over the 10 - day study period* Change in thickness (mm) Scaling Score (range = 0 - 4) *Studies conducted at BioMedCode, a Contract Research Organization specialized in immune and psoriasis preclinical studies.

22 aditxt.com • Type 1 or autoimmune diabetes is a condition where the body’s immune system mistakenly attacks cells in the pancreas resulting in diminished production of insulin • ADi TM incorporates an antigen (GAD) expressed in the pancreas • Administration of ADi TM using GAD as the antigen over an 8 - week period in animals with T1D restores insulin production and reverses hyperglycemia ADi TM Proof - of - Concept for T1D (Autoimmunity) 10 15 20 25 30 35 40 0 100 200 300 400 500 600 Untreated NOD Mice Age (weeks) F a s t i n g B l o o d G l u c o s e ( m g / d L ) 1 2 3 4 5 6 7 8 9 10 11 12 10 15 20 25 30 35 40 0 100 200 300 400 500 600 ADi Tx of NOD mice at >200 mg/dL Age (weeks) F a s t i n g B l o o d G l u c o s e ( m g / d L ) 1 2 3 4 5 6 7 8 9 10 11 12 90% of female NOD mice developed spontaneous autoimmune diabetes. Disease progression may be different for individual animals. ADi TM was administered once a week for 8 weeks after each animal developed hyperglycemia. All animals responded with 80% showing durable response for the entire 40 - week study period. “Reversal of Hyperglycemia and Suppression of Type1 Diabetes in the NOD Mouse with Apoptotic DNA Immunotherapy™ (ADi™), ADi - 100” published in Biomedicines (2020) A B A B

23 aditxt.com ADi TM Proof - of - Concept for Alopecia Areata (Autoimmunity) Methylated vector Methylated BAX Unmethylated BAX ADi TM protects hair follicles from autoimmune attack Studies conducted at Loma Linda University.

24 aditxt.com Timeline for First - in - Human Trials Organ Transplantation (Skin) and Autoimmunity (Psoriasis) There can be no assurance that the projected product development plan can be successfully executed. Received Letter of Interest to conduct and fund Phase II trials in Psoriasis from the Immune Tolerance Network – leading research organization funded by the U.S. National Institute of Health Begin Phase I/IIA 20 - 30 Patients Q1,2021 Q2,2022 Q3,2022 Q4,2023 Complete Phase I/IIA Begin Phase II Complete Phase II
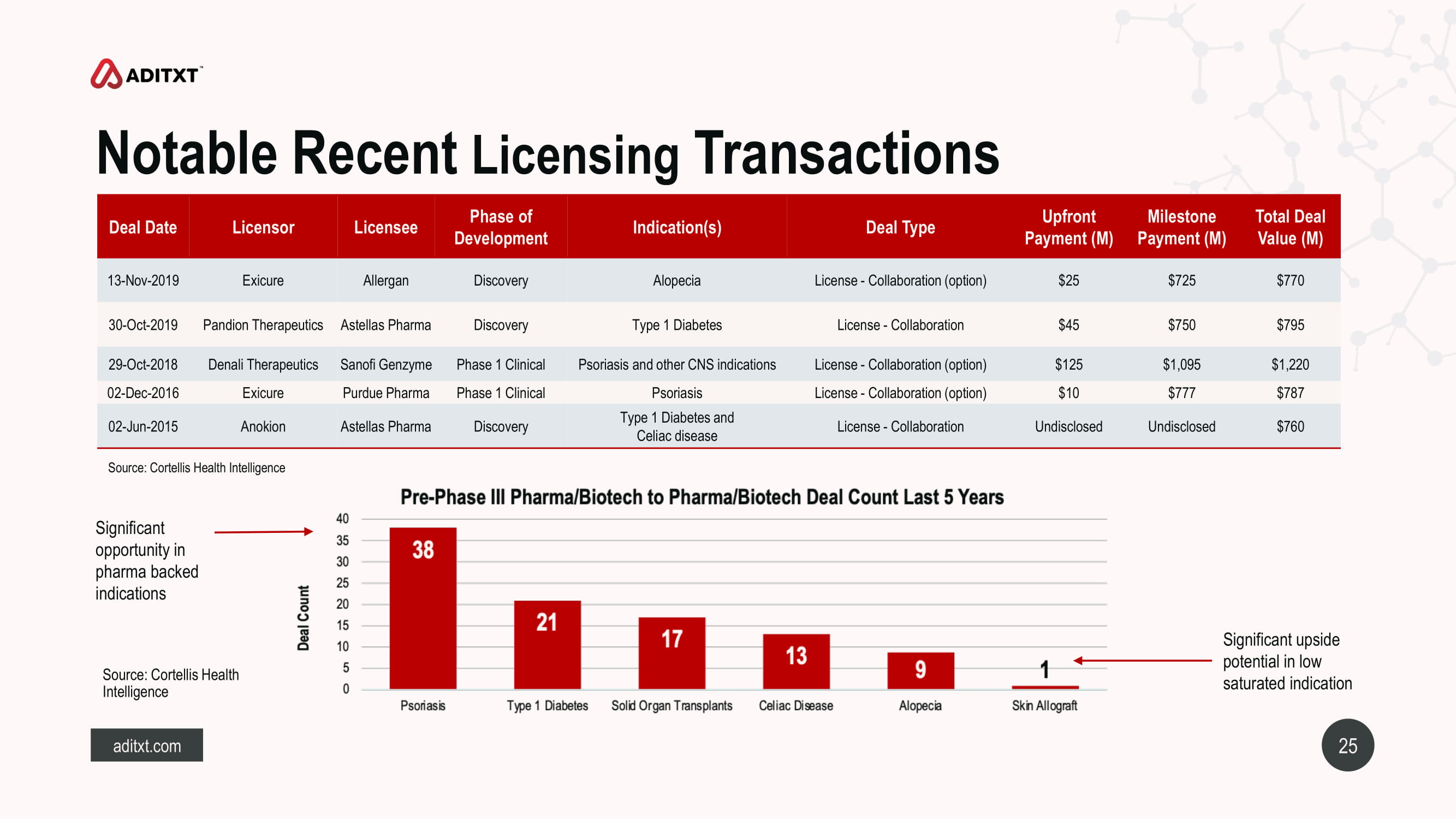
25 aditxt.com Notable Recent Licensing Transactions Significant upside potential in low saturated indication Source: Cortellis Health Intelligence Significant opportunity in pharma backed indications Source: Cortellis Health Intelligence Deal Date Licensor Licensee Phase of Development Indication(s) Deal Type Upfront Payment (M) Milestone Payment (M) Total Deal Value (M) 13 - Nov - 2019 Exicure Allergan Discovery Alopecia License - Collaboration (option) $25 $725 $770 30 - Oct - 2019 Pandion Therapeutics Astellas Pharma Discovery Type 1 Diabetes License - Collaboration $45 $750 $795 29 - Oct - 2018 Denali Therapeutics Sanofi Genzyme Phase 1 Clinical Psoriasis and other CNS indications License - Collaboration (option) $125 $1,095 $1,220 02 - Dec - 2016 Exicure Purdue Pharma Phase 1 Clinical Psoriasis License - Collaboration (option) $10 $777 $787 02 - Jun - 2015 Anokion Astellas Pharma Discovery Type 1 Diabetes and Celiac disease License - Collaboration Undisclosed Undisclosed $760

26 aditxt.com Product Development Team Shahrokh Shabahang, MS, PhD – Co - founder, Chief Innovation Officer, Board Member 20+ years of experience commercializing life science technologies focused on product and clinical development in the fields of microbiology and immunology. Gilles Benichou, PhD – Immunology Consultant Associate Professor, Harvard Medical School, and Director of Molecular and Cellular Immunology Laboratory at Massachusetts General Hospital focused on immune tolerance in transplantation. Dolly Tyan, PhD – Incoming VP of Clinical Development, Transplant 45 years of HLA immunology experience as past Medical Director of Stanford Histocompatibility, Immunogenetics & Disease Profiling Laboratory. David Schwarz, PhD – Molecular Biology Consultant CEO of Advanced Cellular Dynamics leading design, development, validation and deployment of cell - based assay technologies. Ge Chen, MD – Incoming VP of Preclinical R&D Supervisor of Innovation in Histocompatibility and Immunogenetics at Stanford with 30 years experience developing technology platforms for immune monitoring. Research & Development

27 aditxt.com Waldo Concepcion, MD – Incoming VP of Clinical Development, Transplantation Professor of surgery and pediatrics (nephrology) at Stanford University Medical Center with 31 years experience. Joachim - Friedrich Kapp, MD, PhD – Incoming VP of Clinical Development, Autoimmunity Former President Global Business Unit (GBU) Specialized Therapeutics at Schering AG with 30+ years of experience with regulatory agencies (FDA & EMA) and product registration. Daniel Alam, MD – Transplant Advisor Performed first face transplant in the US, which was world's first near - total facial transplant and fourth known facial transplant to have been successfully performed to - date. SeedIP – IP Counsel Founded in 1962. A leading IP law firm with clients ranging in size from start - up companies to established multinational corporations with IP needs ranging from trademark clearance to protecting complex chemical and biotechnology patents. Leif Eldevik – Director of Analytics 20+ years experience in quantitative analytics. Contributing author to A Mathematical Model for DC Vaccine Treatment of Type I Diabetes published in Front. Physiol., 2019. Clinical, Regulatory and IP

28 aditxt.com Business Development AditxtReprogramming Michael Casey Cozart – Head of Business Development Managing Director and Co - Head of LifeSci Advisors Partnering and Analytics division. Advised both publicly - traded as well as privately held life sciences companies on transactions totaling over $2.6 billion in total deal value. Received a B.A. in Economics and En glish from the University of North Carolina at Chapel Hill. LifeSci Advisors – Partnering and Analytics The LifeSci Partnering and Analytics team has executed 80+ mandates that have supported strategic transactions, financing activities, internal capital rationing and development decisions for companies spanning all therapeutic areas and development stages. The te am has advised on 20+ transactions with a total deal value exceeding $2.3 billion. Their extensive global network of contacts co mpr ised of C - level executives, business and corporate development executives, and R&D teams allows their team to complete transactions expeditiously and under the most advantageous terms for their clients.

29 aditxt.com Capital Markets & Communications Team Jeff Ramson – Senior Advisor, Capital Markets & Communications Founder & CEO of PCG Advisory. Has 25+ years of Wall Street experience, investing in and raising money for emerging public a nd private companies in various stages of development. Through traditional and digital IR, he has helped communicate each of their stories accurately an d effectively to maximize exposure to current and potential stakeholders. Kerry Corbit – Digital Investor Relations Oversees PCG Digital, a division of PCG Advisory that focuses on multimedia, investor - focused marketing. Developed two successful entrepreneurial businesses with exits. Kerry is focused on working with innovative companies and crafting campaigns designed to reach stakeholder and shareholder audiences. Chuck Harbey – Investor Relations Managing Director, Corporate Advisory & Private Markets at PCG Advisory. Has 20+ year career as a securities professional serving the industry as a wealth manager, VP of trading, placement agent, branch manager of boutique NYC firms. Kirin Smith – Investor Relations President at PCG Advisory. Brings wealth of capital markets expertise including advisory banking, portfolio management, retail/institutional sales and fundamental research analysis. Former investment banker where he advised, structured and raised capital for middle market companies in multiple sectors. Investor Relations Digital Investor Relations

30 aditxt.com Corporate Team Amro Albanna - Co - founder, President and CEO Founded multiple startups to commercialize innovations in various industries including enterprise software, research incubati on, GPS & wireless, nano sensors, consumer health and biotech. Led multiple M&A and going - public transactions as a founder, co - founder and senior executive. Foun ded Timely Technology Corporation ( TTC ) which was one of the early developers of e - commerce applications for clients including Mercury Insurance, Reuters, and Boeing. TTC was acquired by a NASDAQ - listed company. Founded and sold an innovation incubator which was developed in partnership with the University of Calif ornia, Riverside Research Park. Was a co - founder of Digital Angel Corporation (DAC), in charge of commercializing its GPS/wireless technologies. DAC reached a market capitalization of $233M on AMEX. Bachelor of Science in Business Administration and Computer Info. Systems, graduate coursework in Computer Science and En gineering, and coursework in Immunology and Genetics at Harvard Medical School HMX online learning platform. Sheppard, Mullin, Richter & Hampton LLP Full service Global 100 firm handling corporate and technology matters with presence in North America, Europe and Asia . Corinne Pankovcin, CPA, MBA – Chief Financial Officer Served as Chief Financial Officer for several world class organizations including; Business Development Corporation of Americ a ( “BDCA”), BlackRock Kelso Capital (NASDAQ BKCC ) and AIG Capital Partners. She was responsible for executing portfolio investments and managing significant M&A transactions th roughout her career. She has extensive experience with compliance with the Securities Act of 1933 as well as managing the reporting requi rem ents under the Securities Exchange Act of 1934. She began her career as a Senior Manager in the consumer products and manufacturing practice with Pricewaterhous eCo opers LLP. Brio Financial Group – Accounting Full service financial and accounting firm . dbbmckennon – Audit Certified public accountants (registered firm PCAOB) Accounting & Audit Corporate and Securities Legal

31 aditxt.com Independent Board Members Brian Brady, CFA, MBA – Current Director, and Chair of Audit Committee Nominee Former Sr. Portfolio Manager/VP at Northern Trust who led the investment practice in the Fort Worth and West Texas marketplac e. Extensive experience with financial markets and investment due diligence. Currently serves as Director of Investments for a large hospital system. Namvar Kiaie – Director and Chair of Compensation Committee Nominee Senior R&D leader and Head of R&D for the New Analyte Ventures at Abbott Diabetes Care, managing up to 80 R&D Engineers and S cie ntists in developing Abbott’s next generation biosensing technologies. Responsible for the successful development of 40+ novel diabetes management related products, including FreeStyle Libre. Laura Anthony – Director and Chair of Nominating and Corporate Governance Committee Nominee Securities attorney providing corporate counsel to small - cap and middle market private and public companies. Focus includes comp liance with the Securities Act of 1933 offer sale and registration requirements, including private and public offerings; IPOs; compliance with the NASDAQ and NYSE America n i nitial and continued listing requirements; compliance with the registration and reporting requirements under the Securities Exchange Act of 1934; and mergers and acquis iti ons. Jeffrey Runge, MD, FACEP – Incoming Director Former Chief Medical Officer & Assistant Secretary for Health Affairs of the U.S. Department of Homeland Security. Has over 3 5 y ears of experience as a physician, board certified in emergency medicine, with two decades of experience as a Federal government and healthcare executive. Has experti se across the fields of homeland defense, threat mitigation and prevention, medical preparedness, and medical services delivery. Also served as the head of two federal ag encies (2001 - 2008).

32 aditxt.com Two key biotechnologies with strong IP Portfolio • Advanced diagnostic platform for rapid Monitoring of the immune system • Pioneering Selective Tolerance (ST) therapeutic technology for Reprogramming the immune system • Protected by 67 issued patents and 19 pending applications AditxtMonitoring for immune systems meets significant needs in the marketplace • Simple, rapid and accurate solution • Provides individuals with personal knowledge of their vulnerabilities, responses, and reactions to a spectrum of diseases, bacteria and viruses • Estimated commercialization Q4 2020

Amro Albanna, CEO, Aditx Therapeutics, Inc. Prolonging Life and Enhancing its Quality by Monitoring and Reprogramming the Immune System Contact Information Jeff Ramson, Senior Advisor, Capital Markets & Communications albanna@aditxt.com 909 - 488 - 0844 jramson@pcgadvisory.com 646 - 863 - 6341