Table of Contents
Registration No. 333-159371
 |  |
| • | scientific innovation, with a combined team of top scientists focused on discovering, developing and delivering innovative treatments for patients around the world; | |
| • | a stronger, more diversified product portfolio with an expanded geographic footprint and an industry-leading team of marketing and sales professionals; and | |
| • | a strong financial base, to be further strengthened by synergies expected to be recognized from the combination, to support further investments in research and strategic opportunities to build for the future. |
| Sincerely, | Sincerely, | |
 |  | |
| Richard T. Clark | Fred Hassan | |
| Chairman, President and Chief Executive Officer Merck & Co., Inc. | Chairman and Chief Executive Officer Schering-Plough Corporation |
Table of Contents

| • | Consider and act on a proposal to approve the Agreement and Plan of Merger, dated as of March 8, 2009, by and among Merck & Co., Inc.,Schering-Plough Corporation, SP Merger Subsidiary One, Inc. (formerly Blue, Inc.), and SP Merger Subsidiary Two, Inc. (formerly Purple, Inc.), as it may be amended (the “merger agreement”) and the issuance of shares of common stock in the merger contemplated by the merger agreement. The Board recommends a voteFORthis proposal. | |
| • | Approve any adjournment of the Schering-Plough Special Meeting (including, if necessary, to solicit additional proxies if there are not sufficient votes to approve the merger agreement and the issuance of shares of common stock in the merger). The Board recommends a voteFORthis proposal. |
Table of Contents

Table of Contents
 |  |
is first being mailed to shareholders on or about June 29, 2009.
Table of Contents
Merck & Co., Inc. One Merck Drive P.O. Box 100 Whitehouse Station, NJ 08889 1-908-423-7845 Attention: Stockholder Services Dept, WS3AB-40 www.merck.com/finance | Schering-Plough Corporation 2000 Galloping Hill Road Kenilworth, NJ 07033 1-908-298-7436 Attention: Investor Relations www.schering-plough.com/investor-relations/index.aspx |
IF YOU ARE A MERCK SHAREHOLDER: Laurel Hill Advisory Group, LLC 100 Wall Street, 22nd Floor New York, NY 10005 1-888-742-1305 | IF YOU ARE A SCHERING-PLOUGH SHAREHOLDER: Georgeson Shareholder Communications, Inc. 199 Water Street, 26th Floor New York, NY 10038 1-866-288-2190 For strategic and financial issues: Alex Kelly Group Vice President — Global Communications and Investor Relations Schering-Plough Corporation 2000 Galloping Hill Road Mail Stop: K-1-4-4275 Kenilworth, NJ 07033 Phone:(908) 298-7436 Fax:(908) 298-7082 For governance and social issues: Susan Ellen Wolf Corporate Secretary and Vice President — Corporate Governance Schering-Plough Corporation 2000 Galloping Hill Road Mail Stop: K-1-4-4275 Kenilworth, NJ 07033 Phone:(908) 298-3636 Fax:(908) 298-7303 |
Table of Contents
| Page | ||||
| v | ||||
| 1 | ||||
| 1 | ||||
| 2 | ||||
| 4 | ||||
| 4 | ||||
| 4 | ||||
| 4 | ||||
| 5 | ||||
| 7 | ||||
| 8 | ||||
| 9 | ||||
| 9 | ||||
| 9 | ||||
| 10 | ||||
| 10 | ||||
| 10 | ||||
| 10 | ||||
| 10 | ||||
| 10 | ||||
| 12 | ||||
| 12 | ||||
| 14 | ||||
| 15 | ||||
| 15 | ||||
| 16 | ||||
| 17 | ||||
| 17 | ||||
| 23 | ||||
| 37 | ||||
| 38 | ||||
| 38 | ||||
| 39 | ||||
| 39 | ||||
| 39 | ||||
| 39 | ||||
| 40 | ||||
| 40 | ||||
| 40 | ||||
| 40 | ||||
| 41 | ||||
| 41 | ||||
Table of Contents
| Page | ||||
| 41 | ||||
| 41 | ||||
| 42 | ||||
| 42 | ||||
| 42 | ||||
| 42 | ||||
| 42 | ||||
| 43 | ||||
| 43 | ||||
| 43 | ||||
| 43 | ||||
| 44 | ||||
| 44 | ||||
| 44 | ||||
| 45 | ||||
| 45 | ||||
| 45 | ||||
| 45 | ||||
| 46 | ||||
| 46 | ||||
| 46 | ||||
| 47 | ||||
| 47 | ||||
| 48 | ||||
| 48 | ||||
| 49 | ||||
| 60 | ||||
| 64 | ||||
| 70 | ||||
| 73 | ||||
| 89 | ||||
| 90 | ||||
| 93 | ||||
| 93 | ||||
| 93 | ||||
| 95 | ||||
| 95 | ||||
| 96 | ||||
| 96 | ||||
| 98 | ||||
| 101 | ||||
| 101 | ||||
| 101 | ||||
ii
Table of Contents
| Page | ||||
| 101 | ||||
| 102 | ||||
| 102 | ||||
| 103 | ||||
| 104 | ||||
| 106 | ||||
| 109 | ||||
| 111 | ||||
| 111 | ||||
| 113 | ||||
| 114 | ||||
| 115 | ||||
| 116 | ||||
| 119 | ||||
| 119 | ||||
| 120 | ||||
| 121 | ||||
| 122 | ||||
| 123 | ||||
| 127 | ||||
| 127 | ||||
| 127 | ||||
| 129 | ||||
| 129 | ||||
| 130 | ||||
| 130 | ||||
| 130 | ||||
| 130 | ||||
| 143 | ||||
| 143 | ||||
| 144 | ||||
| 145 | ||||
| 147 | ||||
| 148 | ||||
| 148 | ||||
| 149 | ||||
| 150 | ||||
| 150 | ||||
| 150 | ||||
| 151 | ||||
| 151 | ||||
| 151 | ||||
| 152 | ||||
| 152 | ||||
iii
Table of Contents
| Page | ||||
| 153 | ||||
| 153 | ||||
| 153 | ||||
| 154 | ||||
| 154 | ||||
| 154 | ||||
| 154 | ||||
| 155 | ||||
| 155 | ||||
| 155 | ||||
| 155 | ||||
| 156 | ||||
| 156 | ||||
| 156 | ||||
| 157 | ||||
| 159 | ||||
iv
Table of Contents
| Q: | What is the proposed transaction for which I am being asked to vote? | |
| A: | You are being asked to approve a merger agreement providing for the combination of Merck and Schering-Plough. In order to complete the merger, Merck shareholders must vote to approve the merger agreement and Schering-Plough shareholders must vote to approve the merger agreement and the issuance of shares of common stock of New Merck in the merger. Merck and Schering-Plough will hold separate special shareholders’ meetings to obtain these approvals. This joint proxy statement/prospectus contains important information about the merger, including the special meetings of the respective shareholders of Merck and Schering-Plough. You should read it carefully and in its entirety. The enclosed proxy card or voting instruction card allows you to vote your shares without attending your company’s special meeting. | |
| Your vote is important. We encourage you to vote as soon as possible. | ||
| Q: | When and where will the special meetings be held? | |
| A: | The Merck special meeting is scheduled to be held at 8:30 a.m., local time, on August 7, 2009, at the Bridgewater Marriott located at 700 Commons Way, Bridgewater, NJ. The Schering-Plough special meeting is scheduled to be held at 1:30 p.m., local time, on August 7, 2009, at The Conference Center at Harvard Medical, 77 Avenue Louis Pasteur, Boston, MA. | |
| Q: | Who is entitled to vote at the Merck and Schering-Plough special meetings? | |
| A: | The boards of directors of each of Merck and Schering-Plough has fixed June 22, 2009 as the record date for its respective special meeting. If you were a Merck or Schering-Plough shareholder at the close of business on the record date you are entitled to vote your Merck or Schering-Plough shares at your company’s special meeting. | |
| Q: | How many votes do I have? |
| A: | You are entitled to one vote at the Merck special meeting for each share of Merck common stock that you owned as of the record date. As of the close of business on the record date, there were approximately 2,108,780,449 outstanding shares of Merck common stock. As of that date, less than 1% of the outstanding shares of Merck common stock were held by the directors and executive officers of Merck. |
| You are entitled to one vote at the Schering-Plough special meeting for each share of Schering-Plough common stock that you owned as of the record date. As of the close of business on the record date, there were approximately 1,633,437,974 outstanding shares of Schering-Plough common stock. As of that date, less than 1% of the outstanding shares of Schering-Plough common stock were held by the directors and executive officers of Schering-Plough. |
| Q: | What constitutes a quorum? | |
| A: | Shareholders who hold at least a majority of the outstanding shares of Merck common stock as of the close of business on the record date and who are entitled to vote must be present, either in person or represented by proxy, in order to constitute a quorum to conduct business at the Merck special meeting. | |
| Shareholders who hold at least a majority of the outstanding shares of Schering-Plough common stock as of the close of business on the record date and who are entitled to vote must be present, either in person or represented by proxy, in order to constitute a quorum to conduct business at the Schering-Plough special meeting. | ||
| Q: | What vote is required to approve the merger agreement? | |
| A: | As long as a quorum is present at the companies’ respective special meetings, the affirmative vote of a majority of the votes cast at the special meeting is required for each of Merck and Schering-Plough to approve the merger agreement. Moreover, in the case of Schering-Plough, the rules of the New York Stock Exchange require that holders of at least a majority of the outstanding shares of Schering-Plough common |
v
Table of Contents
| stock actually cast votes on the proposal to approve the merger agreement (whether for or against the proposal). | ||
| Q: | What is the difference between holding shares as a shareholder of record or in “street name”? | |
| A: | If your shares are registered directly in your name with Merck’s transfer agent, Wells Fargo Bank, N.A., or with Schering-Plough’s transfer agent, BNY Mellon, as the case may be, you are considered, with respect to those shares, the “shareholder of record.” If you are a shareholder of record, this joint proxy statement/prospectus and the enclosed proxy card have been sent directly to you by Merck or Schering-Plough. | |
| If your shares are held in a stock brokerage account or by a bank or other nominee, you are considered the beneficial owner of shares held in “street name.” This joint proxy statement/prospectus has been forwarded to you by your broker, bank or nominee who is considered, with respect to those shares, the shareholder of record. As the beneficial owner of shares held in street name, you have the right to direct your broker, bank or nominee how to vote your shares by using the voting instruction card included with this joint proxy statement/prospectus or by following their instructions for voting by telephone or the Internet. | ||
| Q: | How do I vote? | |
| A: | In order to ensure that your vote is recorded, please submit your proxy or voting instructions as instructed below as soon as possible even if you plan to attend your company’s special meeting in person. | |
| Mail. You can vote by mail by completing, signing, dating and mailing your proxy card or voting instruction card in the postage-paid envelope included with this joint proxy statement/prospectus. | ||
| Vote by Telephone or Internet. If you are a shareholder of record (that is, if you hold your shares in your own name), you may vote by telephone (toll-free) or the Internet by following the instructions on your proxy and voting instruction card. If your shares are held in the name of a bank, broker or other holder of record (that is, in “street name”), and if the bank or broker offers telephone and Internet voting, you will receive instructions from them that you must follow in order for your shares to be voted. If you vote by telephone or the Internet, you do not need to return your proxy and voting instruction card. | ||
| In addition, all shareholders may vote in person at their company’s special meeting. You may also be represented by another person at the meeting by executing a proper proxy designating that person. If you are a beneficial owner of shares held in street name, you must obtain a legal proxy from your broker, bank or nominee and present it to the inspectors of election with your ballot when you vote at the meeting. | ||
| Q: | How will my proxy be voted? | |
| A: | If you vote by Internet, by telephone or by completing, signing, dating and mailing your proxy card or voting instruction card, your shares will be voted in accordance with your instructions. If you are a shareholder of record and you sign, date, and return your proxy card but do not indicate how you want to vote or do not indicate that you wish to abstain, your shares will be voted in favor of the approval of the merger agreement. | |
| Q: | Who can attend the Merck and Schering-Plough special meetings? | |
| A: | All Merck shareholders as of the record date may attend the Merck special meeting but must have an admission ticket. If you are a shareholder of record, the ticket attached to the proxy card will admit you and one guest. If you are a beneficial owner of Merck shares held in street name, you may request a ticket by writing to the following address: |
| Office of the Secretary, WS 3AB-05 Merck & Co., Inc. P.O. Box 100 Whitehouse Station, NJ08889-0100 |
| or by faxing your request to908-735-1224. You must provide evidence of your ownership of shares with your ticket request, which you can obtain from your broker, bank or nominee. We encourage you or your |
vi
Table of Contents
| broker, bank or nominee to fax your ticket request and proof of ownership in order to avoid any mail delays. | ||
| All Schering-Plough shareholders as of the record date may attend the Schering-Plough special meeting with an admission ticket and a photo identification. To get an admission ticket, Schering-Plough shareholders must write to Schering-Plough’s transfer agent, BNY Mellon, using the following address: |
| BNY Mellon Shareowner Services 480 Washington Boulevard 29th Floor Jersey City, NJ 07310 Attn: Ann-Marie Webb |
| If you are a record shareholder (your shares are held in your name), you must list your name exactly as it appears on your stock ownership records from BNY Mellon. If you hold shares through a bank, broker or trustee, you must also include a copy of your latest bank or broker statement showing your ownership. | ||
| Q: | Can I change my vote after I have submitted a proxy or voting instruction card? | |
| A: | Yes. If you are a shareholder of record you can change your vote at any time before your proxy is voted at your special meeting. You can do this in one of three ways: | |
| • you can send a signed notice of revocation to the Secretary of Merck or the Corporate Secretary of Schering-Plough, as appropriate; | ||
| • you can submit a revised proxy bearing a later date by Internet, telephone or mail as described above; or | ||
| • you can attend your company’s special meeting and vote in person, which will automatically cancel any proxy previously given, or you may revoke your proxy in person, but your attendance alone will not revoke any proxy that you have previously given. | ||
| If you choose either of the first two methods, you must submit your notice of revocation or your new proxy no later than the beginning of the applicable special meeting. If you are a beneficial owner of shares held in street name, you may submit new voting instructions by contacting your broker, bank or nominee. You may also vote in person at the special meeting if you obtain a legal proxy from your broker, bank or nominee and present it to the inspectors of election with your ballot when you vote at the meeting. |
| Additional information on changing your vote is located on page 41 for Merck and on page 45 for Schering-Plough. |
| Q: | As a participant in Merck’s 401(k) or similar employee retirement plan(s), how do I vote shares held in my plan account? | |
| A: | If you are a participant in the Merck & Co., Inc. Employee Savings and Security Plan, Merck & Co., Inc. Employee Stock Purchase and Savings Plan, Hubbard LLC Employee Savings Plan, Merck Puerto Rico Employee Savings and Security Plan, Merck Frosst Canada Inc. Stock Purchase Plan (Merck Frosst Plan) or Merial 401(k) Savings Plan (Merial Plan), you should have received separate proxy voting instruction cards from the plan trustees and you have the right to provide voting directions to the plan trustee by submitting your voting instruction card for those shares of Merck common stock that are held by your plan and allocated to your plan account on the approval of the merger agreement. | |
| Q: | If I am a participant in one of the Merck retirement plans mentioned above, what happens if the plan trustee does not receive voting instructions from me? | |
| A: | If voting instructions are not received from participants in the Merck Frosst Plan, the plan trustee will vote the shares in accordance with the recommendation of the Merck board of directors. | |
| If voting instructions are not received from participants in the Merial Plan, the plan trustee will vote the shares in the same proportion as it votes shares for which voting instructions are received from plan participants. |
vii
Table of Contents
| If voting instructions are not received from participants in the plans other than the Merck Frosst Plan and the Merial Plan mentioned above, trustees for the other plans will not vote shares for which no voting instructions are received from plan participants. | ||
| Q: | As a participant in Schering-Plough’s employees’ savings plans, how do I vote shares held in my plan account? | |
| A: | If you are a current or former Schering-Plough employee with shares credited to an account under the Schering-Plough employees’ savings plan or the Schering-Plough Puerto Rico employees’ retirement savings plan, you will receive a proxy and voting instruction card. | |
| If you do not give voting instructions to the plan trustee by mailing your proxy and voting instruction card or voting by telephone or the Internet, the trustee will vote shares you hold in the employees’ savings plan or in the Puerto Rico employees’ retirement savings plan in the same proportion as shares held in that plan for which voting instructions were timely received. To allow sufficient time for the trustee to vote your shares under either plan, your voting instructions must be received by 5:00 p.m. (Eastern Time) on Tuesday, August 4, 2009. | ||
| Q: | Should I send in my share certificates now? | |
| A: | No. If you hold Schering-Plough share certificates, after we have completed the transaction, we will send you written instructions informing you how to exchange your share certificates. If you hold Merck share certificates, your share certificates will automatically represent an equal number of shares in New Merck after completion of the transaction. | |
| Q: | If I hold outstanding Merck stock options or restricted stock units, what do I need to do? |
| A: | No action is necessary on your part. Immediately prior to the closing, each of your outstanding stock options and restricted stock units will automatically convert, on a one for one basis, to be a stock option exercisable for, or a restricted stock unit settled in, common shares of New Merck. This conversion is also described on page 102. |
| Q: | If I hold Schering-Plough stock options or deferred stock units, what do I need to do? | |
| A: | No action is necessary on your part. Immediately prior to the closing, your outstanding stock options will automatically convert to options to purchase common shares of New Merck, pursuant to the conversion formulas described on page 102. |
| Q: | When do you expect the merger to be completed? | |
| A: | Schering-Plough and Merck are working to complete the merger in the fourth quarter of 2009. However, the merger is subject to various regulatory approvals and other conditions, and it is possible that factors outside the control of both companies could result in the merger being completed at a later time, or not at all. There may be a substantial amount of time between the respective Schering-Plough and Merck special meetings and the completion of the merger. Schering-Plough and Merck hope to complete the merger as soon as reasonably practicable. | |
| Q: | Who can answer any questions I may have about the special meeting or the transaction? | |
| A: | Merck shareholders may call Laurel Hill Advisory Group, LLC toll-free at 1-888-742-1305 and banks and brokers may call collect at 1-917-338-3181 with any questions they may have. |
viii
Table of Contents
| For logistical questions, such as how to exchange shares, Schering-Plough shareholders may call Georgeson Shareholder Communications, Inc. toll-free at 1-866-288-2190 and banks and brokers may call 1-212-440-9800 with any questions they may have. | ||
| For other questions that Schering-Plough shareholders may have, the officers leading the Schering-Plough Shareholder Engagement Program remain your contacts: |
| For Strategic and Financial Issues: | For Governance and Social Issues: | |
| Alex Kelly Group Vice President — Global Communications and Investor Relations Schering-Plough Corporation 2000 Galloping Hill Road Mail Stop: K-1-4-4275 Kenilworth, NJ 07033 Phone:(908) 298-7436 Fax:(908) 298-7082 | Susan Ellen Wolf Corporate Secretary and Vice President — Corporate Governance Schering-Plough Corporation 2000 Galloping Hill Road Mail Stop: K-1-4-4525 Kenilworth, NJ 07033 Phone: (908) 298-3636 Fax: (908) 298-7303 |
ix
Table of Contents
1
Table of Contents
2
Table of Contents
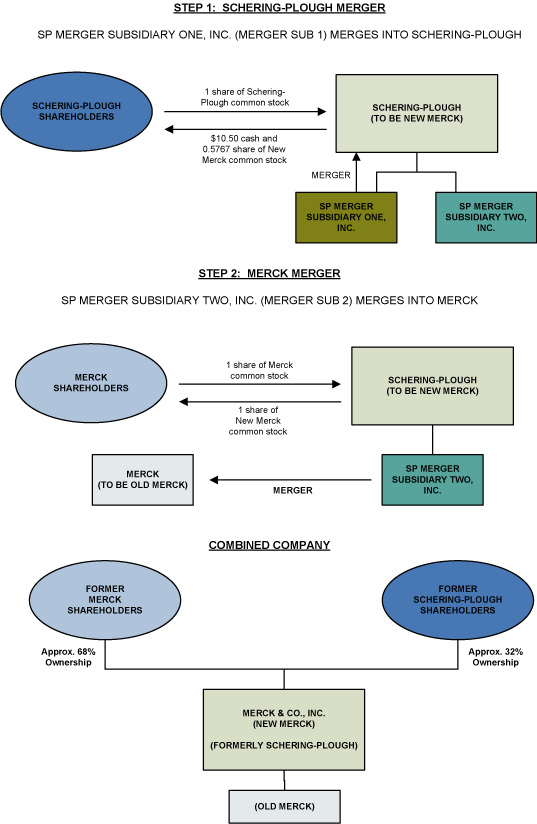
3
Table of Contents
4
Table of Contents
5
Table of Contents
| • | solicit any inquiries or the making of any acquisition proposal; | |
| • | engage in discussions or negotiations regarding an acquisition proposal or furnish to any third party any information in connection with an acquisition proposal; | |
| • | allow its board of directors to change its recommendation in favor of the merger agreement; or | |
| • | enter into any agreement relating to an acquisition proposal. |
| • | engage in discussions or negotiations with a third party that has made a superior proposal or an acquisition proposal that the board determines in good faith could reasonably lead to a superior proposal and that the board determines in good faith is credible and reasonably capable of consummating a superior proposal; | |
| • | thereafter, furnish to the third party nonpublic information pursuant to a confidentiality agreement with terms no less materially favorable to Merck or Schering-Plough, as the case may be, than those contained in the confidentiality agreement between Merck and Schering-Plough, and including a standstill agreement no more materially favorable to such third party than any standstill or similar agreement applicable to Merck or Schering-Plough, as the case may be (provided that any such standstill or similar provision may allow such third party to make acquisition proposals to Merck or Schering-Plough, as the case may be, in connection with the negotiations or discussions permitted by the merger agreement); and | |
| • | in response to a superior proposal or an intervening event, change its recommendation in favor of the merger agreement. Moreover, each must present the merger agreement to its shareholders for their approval or disapproval, even if it is no longer recommending the transaction. However, the board of directors of Schering-Plough may, in response to an acquisition proposal which the board determines in good faith is a superior proposal, terminate the merger agreement to enter into a definitive agreement with respect to the superior proposal and, therefore, need not hold its shareholder meeting to vote on the merger with Merck. |
6
Table of Contents
| • | a $3 billion364-day bridge loan agreement with respect to the bridge loan facility; | |
| • | a $3 billion364-day asset sale facility agreement with respect to the asset sale facility; and | |
| • | a $1 billion364-day incremental loan agreement with respect to the incremental facility. |
7
Table of Contents
8
Table of Contents
9
Table of Contents
10
Table of Contents
11
Table of Contents
| As of and for the | ||||||||||||||||||||||||||||
| Three Months Ending | ||||||||||||||||||||||||||||
| March 31, | As of and for the Years Ending December 31, | |||||||||||||||||||||||||||
| 2009 | 2008 | 2008(2) | 2007(3) | 2006(4) | 2005(5) | 2004(6) | ||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (In millions, except per share figures) | ||||||||||||||||||||||||||||
Results for Year: | ||||||||||||||||||||||||||||
| Sales | $ | 5,385.2 | $ | 5,822.1 | $ | 23,850.3 | $ | 24,197.7 | $ | 22,636.0 | $ | 22,011.9 | $ | 22,972.8 | ||||||||||||||
| Equity (income) from affiliates | (585.8 | ) | (652.1 | ) | (2,560.6 | ) | (2,976.5 | ) | (2,294.4 | ) | (1,717.1 | ) | (1,008.2 | ) | ||||||||||||||
| Net income attributable to Merck & Co., Inc. | 1,425.0 | 3,302.6 | 7,808.4 | 3,275.4 | 4,433.8 | 4,631.3 | 5,830.1 | |||||||||||||||||||||
| Basic earnings per common share attributable to Merck & Co., Inc. common shareholders | $ | 0.67 | $ | 1.52 | $ | 3.65 | $ | 1.51 | $ | 2.03 | $ | 2.10 | $ | 2.63 | ||||||||||||||
| Diluted earnings per common share attributable to Merck & Co., Inc. common shareholders | $ | 0.67 | $ | 1.52 | $ | 3.63 | $ | 1.49 | $ | 2.02 | $ | 2.10 | $ | 2.62 | ||||||||||||||
| Cash dividends paid per common share | $ | 0.38 | $ | 0.38 | $ | 1.52 | $ | 1.52 | $ | 1.52 | $ | 1.52 | $ | 1.49 | ||||||||||||||
Year-End Position: | ||||||||||||||||||||||||||||
| Total assets | 46,543.1 | 47,041.1 | 47,195.7 | 48,350.7 | 44,569.8 | 44,845.8 | 42,572.8 | |||||||||||||||||||||
| Long-term debt | 3,939.1 | 3,965.0 | 3,943.3 | 3,915.8 | 5,551.0 | 5,125.6 | 4,691.5 | |||||||||||||||||||||
| (1) | Merck’s financial statements have been restated to reflect the retrospective application of Financial Accounting Standards Board (“FASB”) Statement No. 160,Noncontrolling Interests in Consolidated Financial Statements — an amendment of ARB No. 51and FASB Staff Position EITF03-6-1, Determining Whether Instruments Granted in Share-Based Payment Transactions Are Participating Securities, which Merck adopted on January 1, 2009. | |
| (2) | Amounts for 2008 include a gain on distribution from AstraZeneca LP, a gain related to the sale of Merck’s remaining worldwide rights toAggrastat, the favorable impact of certain tax items, the impact of |
12
Table of Contents
| restructuring actions, additional legal defense costs and an expense for a contribution to the Merck Company Foundation. | ||
| (3) | Amounts for 2007 include the impact of Merck’s U.S.VioxxSettlement Agreement charge, restructuring actions, a civil governmental investigations charge, an insurance arbitration settlement gain, acquired research expense resulting from an acquisition, additionalVioxxlegal defense costs, gains on sales of assets and product divestitures, as well as a net gain on the settlements of certain patent disputes. | |
| (4) | Amounts for 2006 include the impact of restructuring actions, acquired research expenses resulting from acquisitions, additionalVioxxlegal defense costs and the adoption of a new accounting standard requiring the expensing of stock options. | |
| (5) | Amounts for 2005 include the impact of the net tax charge primarily associated with the American Jobs Creation Act repatriation, restructuring actions and additionalVioxxlegal defense costs. | |
| (6) | Amounts for 2004 include the impact of the withdrawal ofVioxx,Vioxxlegal defense costs and restructuring actions. |
13
Table of Contents
| As of and for the | ||||||||||||||||||||||||||||
| Three Months Ending | ||||||||||||||||||||||||||||
| March 31, | As of and for the Years Ending December 31, | |||||||||||||||||||||||||||
| 2009 | 2008 | 2008(1) | 2007(1) | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (In millions, except per share figures) | ||||||||||||||||||||||||||||
Operating Results | ||||||||||||||||||||||||||||
| Net sales | $ | 4,393 | $ | 4,657 | $ | 18,502 | $ | 12,690 | $ | 10,594 | $ | 9,508 | $ | 8,272 | ||||||||||||||
| Equity (income) | (400 | ) | (517 | ) | (1,870 | ) | (2,049 | ) | (1,459 | ) | (873 | ) | (347 | ) | ||||||||||||||
| Net income/(loss)(2) | 805 | 314 | 1,903 | (1,473 | ) | 1,143 | 269 | (947 | ) | |||||||||||||||||||
| Basic earnings/(loss) per common share(2) | 0.47 | 0.17 | 1.08 | (1.04 | ) | 0.71 | 0.12 | (0.67 | ) | |||||||||||||||||||
| Diluted earnings/(loss) per common share(2) | 0.46 | 0.17 | 1.07 | (1.04 | ) | 0.71 | 0.12 | (0.67 | ) | |||||||||||||||||||
Financial Position | ||||||||||||||||||||||||||||
| Total assets(3) | 27,718 | 30,120 | 28,117 | 29,156 | 16,071 | 15,469 | 15,911 | |||||||||||||||||||||
| Long-term debt(3) | 7,685 | 9,349 | 7,931 | 9,019 | 2,414 | 2,399 | 2,392 | |||||||||||||||||||||
Other Data | ||||||||||||||||||||||||||||
| Cash dividends per common share | 0.065 | 0.065 | 0.26 | 0.25 | 0.22 | 0.22 | 0.22 | |||||||||||||||||||||
| Cash dividends paid on preferred shares | 38 | 38 | 150 | 99 | 86 | 86 | 30 | |||||||||||||||||||||
| (1) | Operating results and other financial information reflect the operations of the Organon BioSciences’ (OBS) business subsequent to Schering-Plough’s acquisition of OBS on November 19, 2007, including the impacts of purchase accounting in accordance with SFAS No. 141, “Business Combinations.” | |
| (2) | 2008, 2007, 2006, 2005, and 2004 include special and acquisition-related charges and manufacturing streamlining costs of $329 million, $84 million, $248 million, $294 million, and $153 million, respectively. See Note 3, “Special and Acquisition-Related Charges and Manufacturing Streamlining” in the audited financial statements of Schering-Plough included in its Annual Report onForm 10-K for the year ended December 31, 2008 for additional information on these charges that were incurred in 2008, 2007 and 2006. The special charges incurred in 2005 of $294 million included litigation charges of $250 million, employee termination costs of $28 million and asset impairment and other charges of $16 million. The special charges incurred in 2004 included $119 million of employee termination costs and $34 million for asset impairment and related charges. | |
| (3) | The increase in total assets and long-term debt in 2007, as compared to 2006, primarily reflects the purchase of OBS (total assets) and the financing of the OBS acquisition (long-term debt). |
14
Table of Contents
| Implied Value of | ||||||||||||
| Merger Consideration | ||||||||||||
| per Share of | ||||||||||||
| Merck | Schering-Plough | Schering-Plough | ||||||||||
| Common Stock | Common Stock | Common Stock(1) | ||||||||||
March 6, 2009 | $ | 22.74 | $ | 17.63 | $ | 23.61 | ||||||
June 5, 2009 | 26.07 | 23.80 | 25.53 | |||||||||
| (1) | The equivalent implied per share data for Schering-Plough common stock has been determined by multiplying the closing market price of a share of Merck common stock on each of the dates by the exchange ratio of 0.5767 per share and adding the per share cash consideration of $10.50 being paid to Schering-Plough shareholders. Schering-Plough shareholders will not receive the merger consideration until the merger is completed, which may be a substantial period of time after the Schering-Plough shareholder meeting. There can be no assurance as to the trading prices of the Merck common stock at the time of the closing of the merger. Moreover, because of the need to obtain regulatory approvals, the closing of the merger may not occur, if at all, until months after the vote of shareholders on the transaction. |
| As of and for the | ||||||||
| Three Months Ending | For the Year Ending | |||||||
| March 31, 2009 | December 31, 2008 | |||||||
| (In millions, except per share figures) | ||||||||
Pro Forma Statement of Income Data | ||||||||
| Sales | $ | 10,685.6 | $ | 46,749.6 | ||||
| Equity income from affiliates | $ | (294.9 | ) | $ | (1,024.3 | ) | ||
| Net income available to common shareholders | $ | 1,506.0 | $ | 6,565.1 | ||||
| Basic earnings per common share | $ | 0.48 | $ | 2.09 | ||||
| Earnings per common share assuming dilution | $ | 0.48 | $ | 2.09 | ||||
| Cash dividends per common share | $ | 0.38 | $ | 1.52 | ||||
Pro Forma Balance Sheet Data | ||||||||
| Total assets | $ | 116,725.0 | ||||||
| Long-term debt | $ | 16,878.1 | ||||||
15
Table of Contents
| For the | For the | |||||||
| Three Months Ended | Year Ended | |||||||
| March 31, 2009 | December 31, 2008 | |||||||
Basic Earnings Per Share | ||||||||
| Merck historical | $ | 0.67 | $ | 3.65 | ||||
| Schering-Plough historical | $ | 0.47 | $ | 1.08 | ||||
| Pro forma combined | $ | 0.48 | $ | 2.09 | ||||
| Schering-Plough equivalent | $ | 0.28 | $ | 1.21 | ||||
Diluted Earnings Per Share | ||||||||
| Merck historical | $ | 0.67 | $ | 3.63 | ||||
| Schering-Plough historical | $ | 0.46 | $ | 1.07 | ||||
| Pro forma combined | $ | 0.48 | $ | 2.09 | ||||
| Schering-Plough equivalent | $ | 0.28 | $ | 1.21 | ||||
Dividends Per Share | ||||||||
| Merck historical | $ | 0.38 | $ | 1.52 | ||||
| Schering-Plough historical | $ | 0.065 | $ | 0.26 | ||||
| Pro forma combined | $ | 0.38 | $ | 1.52 | ||||
| Schering-Plough equivalent | $ | 0.22 | $ | 0.88 | ||||
Book Value Per Share at Period End | ||||||||
| Merck historical | $ | 10.43 | ||||||
| Schering-Plough historical | $ | 6.30 | ||||||
| Pro forma combined | $ | 17.93 | ||||||
| Schering-Plough equivalent | $ | 10.34 | ||||||
16
Table of Contents
17
Table of Contents
18
Table of Contents
| • | integrating the research and development, manufacturing, distribution, marketing and promotion activities and information technology systems of Merck and Schering-Plough; | |
| • | conforming standards, controls, procedures and accounting and other policies, business cultures and compensation structures between the companies; | |
| • | consolidating corporate and administrative infrastructures; | |
| • | consolidating sales and marketing operations; | |
| • | retaining existing customers and attracting new customers; | |
| • | identifying and eliminating redundant and underperforming operations and assets; | |
| • | coordinating geographically dispersed organizations; | |
| • | managing tax costs or inefficiencies associated with integrating the operations of the combined company; and | |
| • | making any necessary modifications to operating control standards to comply with the Sarbanes-Oxley Act of 2002 and the rules and regulations promulgated thereunder. |
19
Table of Contents
20
Table of Contents
21
Table of Contents
| • | Schering-Plough may be required to pay Merck a termination fee of up to $1.25 billion if the merger agreement is terminated under certain circumstances (plus, in certain circumstances, Schering-Plough also would be obligated to reimburse Merck up to $250 million of Merck’s actual expenses incurred in connection with the merger), or Merck may be required to pay Schering-Plough a termination fee of $1.25 billion if the merger agreement is terminated under certain other circumstances (and, in certain circumstances, Merck also would be obligated to reimburse Schering-Plough up to $150 million of Schering-Plough’s actual expenses incurred in connection with the merger), all as described in the merger agreement and summarized in this joint proxy statement/prospectus; | |
| • | Merck will be required to pay Schering-Plough a termination fee of $2.5 billion and reimburse Schering-Plough’s expenses up to a maximum of $150 million if either Merck or Schering-Plough terminates the merger agreement because the drop-dead date, as it may be extended, has occurred and the merger has not been consummated because the proceeds of the financing are not available in full; | |
| • | Merck and Schering-Plough will be required to pay certain costs relating to the merger, whether or not the merger is completed; and | |
| • | matters relating to the merger (including integration planning) may require substantial commitments of time and resources by Merck and Schering-Plough management, which could otherwise have been devoted to other opportunities that may have been beneficial to Merck and Schering-Plough as independent companies, as the case may be. |
22
Table of Contents
23
Table of Contents
24
Table of Contents
25
Table of Contents
26
Table of Contents
27
Table of Contents
28
Table of Contents
29
Table of Contents
30
Table of Contents
31
Table of Contents
32
Table of Contents
33
Table of Contents
| • | There may be limited access to and supply of normal and diseased tissue samples, cell lines, pathogens, bacteria, viral strains and other biological materials. In addition, government regulations in multiple jurisdictions such as the U.S. and European states within the EU, could result in restricted access to, or transport or use of, such materials. If Schering-Plough, or New Merck after the merger, loses access to sufficient sources of such materials, or if tighter restrictions are imposed on the use of such materials, we may not be able to conduct research activities as planned and may incur additional development costs. |
34
Table of Contents
| • | The development, manufacturing and marketing of biologics are subject to regulation by the FDA, the EMEA and other regulatory bodies. These regulations are often more complex and extensive than the regulations applicable to other pharmaceutical products. For example, in the U.S., a Biologics License Application, including both preclinical and clinical trial data and extensive data regarding manufacturing procedures, is required for human vaccine candidates and FDA approval for the release of each manufactured lot. | |
| • | Manufacturing biologics, especially in large quantities, is often complex and may require the use of innovative technologies to handle living micro-organisms. Each lot of an approved biologic must undergo thorough testing for identity, strength, quality, purity and potency. Manufacturing biologics requires facilities specifically designed for and validated for this purpose, and sophisticated quality assurance and quality control procedures are necessary. Slight deviations anywhere in the manufacturing process, including filling, labeling, packaging, storage and shipping and quality control and testing, may result in lot failures, product recalls or spoilage. When changes are made to the manufacturing process, we may be required to provide pre-clinical and clinical data showing the comparable identity, strength, quality, purity or potency of the products before and after such changes. | |
| • | Biologics are frequently costly to manufacture because production ingredients are derived from living animal or plant material, and most biologics cannot be made synthetically. In particular, keeping up with the demand for vaccines may be difficult due to the complexity of producing vaccines. |
35
Table of Contents
| • | changes in medical reimbursement policies and programs and pricing restrictions in key markets; | |
| • | multiple regulatory requirements that could restrict New Merck’s ability to manufacture and sell its products in key markets; | |
| • | trade protection measures and import or export licensing requirements; | |
| • | diminished protection of intellectual property in some countries; and | |
| • | possible nationalization and expropriation. |
36
Table of Contents
| • | those identified and disclosed in public filings with the SEC made by Merck and Schering-Plough; |
37
Table of Contents
| • | obtaining shareholder approvals required for the Merck merger, the Schering-Plough merger and the issuance of shares of New Merck common stock in connection with the merger; | |
| • | satisfying the conditions to the closing of the merger; | |
| • | successfully integrating the Merck and Schering-Plough businesses, avoiding problems which may result in the combined company not operating as effectively and efficiently as expected; | |
| • | the possibility that the estimated synergies are not realized, or will not be realized within the expected timeframe; | |
| • | unexpected costs or unexpected liabilities, or the effects of purchase accounting varying from the companies’ expectations; | |
| • | the risk that funds invested in research will not generate financial returns because the development of novel drugs requires significant expenditure with a low probability of success; | |
| • | the actual resulting credit ratings of the companies or their respective subsidiaries; | |
| • | the effects on the businesses of the companies resulting from uncertainty surrounding the merger; | |
| • | adverse outcomes of pending or threatened litigation or government investigations; | |
| • | the effects on the companies of future regulatory or legislative actions; | |
| • | conduct and changing circumstances related to third-party relationships on which Merck andSchering-Plough rely for their key products; | |
| • | the extremely volatile and unpredictable current stock market and credit market conditions; | |
| • | market risks from fluctuations in currency exchange rates and interest rates; | |
| • | variations between the stated assumptions on which forward-looking statements are based and Merck’s and Schering-Plough’s actual experience; and | |
| • | other economic, business,and/or competitive factors. |
38
Table of Contents
| • | consider and act on a proposal to approve the merger agreement; and | |
| • | transact any other business that may properly come before the special meeting or any reconvened meeting following an adjournment or postponement of the special meeting. |
39
Table of Contents
40
Table of Contents
| • | sending a signed notice of revocation to the Secretary of Merck; | |
| • | submitting a revised proxy bearing a later date by mail, Internet or telephone; or | |
| • | attending the special meeting and voting in person, which will automatically cancel any proxy previously given, or revoking your proxy in person. Your attendance alone will not revoke any proxy that you have previously given. |
41
Table of Contents
| • | record shareholders wishing to discontinue or begin householding, or any record shareholder residing at a household address wanting to request delivery of a copy of this joint proxy statement/prospectus should contact Merck Stockholder Services, WS3AB-40, P.O. Box 100, Whitehouse Station,NJ 08889-0100 or by calling our toll-free number 1-877-602-7615; and | |
| • | shareholders owning their shares through a bank, broker or other holder of record who wish to either discontinue or begin householding should contact their record holder. |
42
Table of Contents
| • | consider and act on a proposal to approve the merger agreement and the issuance of shares of common stock in the merger contemplated by the merger agreement; | |
| • | approve the adjournment of the Schering-Plough Special Meeting (including, if necessary, to solicit additional proxies if there are not sufficient votes to approve the merger agreement and the issuance of shares of common stock in the merger); and | |
| • | transact any other business that may properly come before the special meeting or any reconvened meeting following an adjournment or postponement of the special meeting. |
43
Table of Contents
44
Table of Contents
480 Washington Boulevard
29th Floor
Jersey City, NJ 07310
Attn: Ann-Marie Webb
| • | sending a signed notice of revocation to the Corporate Secretary of Schering-Plough; | |
| • | submitting a revised proxy bearing a later date by mail, Internet or telephone; or | |
| • | attending the special meeting and voting in person, which will automatically cancel any proxy previously given, or giving written notice of revocation to the Corporate Secretary before the proxy is voted at the special meeting. Your attendance alone will not revoke any proxy that you have previously given. |
45
Table of Contents
| • | record shareholders wishing to discontinue or begin householding, or any record shareholder residing at a household address wanting to request delivery of a copy of this joint proxy statement/prospectus should contact Schering-Plough’s transfer agent, BNY Mellon, at877-429-1240 (U.S.),201-680-6685 (outside of the U.S.) or www.bnymellon.com/shareowner/isd or may write to them at Schering-Plough Corporation,c/o BNY Mellon Shareowner Services, P.O. Box 358015, Pittsburgh,Pennsylvania 15252-8015; and | |
| • | shareholders owning their shares through a bank, broker or other holder of record who wish to either discontinue or begin householding should contact their record holder. Any shareholder in the household may request prompt delivery of a copy of this joint proxy statement/prospectus by contactingSchering-Plough at908-298-3636 or may write to Schering-Plough at Office of the Corporate Secretary, Schering-Plough Corporation, 2000 Galloping Hill Road, Mail Stop: K-1-4-4525, Kenilworth, New Jersey 07033. |
46
Table of Contents
Whitehouse Station, NJ 08889
Telephone:(908) 423-1000
Mailstop K-1-4525
Kenilworth, NJ 07033
Telephone:(908) 298-4000
47
Table of Contents
Mailstop K-1-4525
Kenilworth, NJ 07033
Telephone:(908) 298-4000
Mailstop K-1-4525
Kenilworth, NJ 07033
Telephone:(908) 298-4000
48
Table of Contents
49
Table of Contents
50
Table of Contents
51
Table of Contents
52
Table of Contents
53
Table of Contents
54
Table of Contents
55
Table of Contents
56
Table of Contents
57
Table of Contents
58
Table of Contents
59
Table of Contents
| • | Expanded Pipeline to Deliver Innovative Medicines for Patients. Merck’s board of directors considered that the combination is expected to: |
| • | increase Merck’s pipeline of early, mid and late stage product candidates, including a doubling from 9 to 18 of the number of potential medicines Merck has in Phase III development; | |
| • | create a combined company having a product pipeline with greater depth and breadth and many promising drug candidates; with greater resources, the combined company is expected to have greater financial flexibility to invest in these development opportunities, as well as external opportunities; and | |
| • | accelerate the expansion into therapeutic areas that Merck has focused on in recent years with the addition of Schering-Plough’s established presence and expertise in oncology, neuroscience and novel biologics. |
60
Table of Contents
| • | Complementary Product Portfolios Focused on Key Therapeutic Areas. The Merck board of directors considered that the combination with Schering-Plough is expected to broaden Merck’s commercial portfolio with leading franchises in key therapeutic areas, including cardiovascular, respiratory, oncology, neuroscience, infectious diseases, immunology and women’s health. In particular, the Merck board of directors considered that: |
| • | Schering-Plough’s products are expected to have long periods of marketing exclusivity; | |
| • | as a result of its expanded product offerings, the combined company is expected to benefit from additional revenue growth opportunities; | |
| • | the combined company is expected to have expanded opportunities for life-cycle management through the introduction of potential new combinations and formulations of existing products of the two companies; | |
| • | the combined company will be well positioned to expand its presence and product offerings; and | |
| • | the combined company is expected to realize potential benefits from Schering-Plough’s strong portfolio of women’s health products, its animal health business and portfolio of consumer health brands, includingClaritin, CoppertoneandDr. Scholl’s. |
| • | Strong Commercial Organization. Merck’s board of directors considered that both companies have teams of talented and experienced employees with strong customer relationships. In particular, the board considered that: |
| • | both Merck and Schering-Plough have made progress in implementing new customer-centric selling models; this is expected to help ensure a smooth and efficient integration of the companies’ commercial operations; and | |
| • | the combined company’s broadened product portfolio is expected to help its sales force be more effective. |
| • | Expanded Global Presence and Geographically Diverse Revenue Base. Merck’s board of directors considered the global reach of the combined company, including that: |
| • | in 2008, Schering-Plough generated approximately 70% of its revenue outside the United States, including more than $2 billion in revenue from newer markets; the combination is expected to accelerate Merck’s international growth efforts, especially in key, high-growth emerging markets; | |
| • | the combined company is expected to have an industry-leading global team of marketing and sales professionals; and | |
| • | the combined company will have a more geographically diverse revenue base with more than 50% of its revenues expected to be generated outside the United States. |
| • | Increased Manufacturing Capabilities. Merck’s board of directors considered the increased manufacturing capabilities that the combined company is expected to have, including that: |
| • | the combination with Schering-Plough will increase Merck’s manufacturing capabilities, particularly in the important growth areas related to biologics and sterile medicines; and | |
| • | the application of Merck’s manufacturing and sourcing strategies across a larger manufacturing base can be expected to create opportunities for synergies and cost savings across the organization. |
| • | Strong Financial Profile. Merck’s board of directors considered the expected financial profile of the combined company. In particular, Merck’s board of directors noted that: |
| • | the combined company’s broad product offerings could be expected to generate strong cash flow; |
61
Table of Contents
| • | the combined revenues of the two companies in 2008 totaled approximately $47 billion (consisting of Schering-Plough’s 2008 reported revenue of $18.502 billion, Merck’s 2008 reported revenue of $23.85 billion, and the Merck/Schering-Plough cholesterol partnership’s 2008 reported revenue of $4.561 billion); | |
| • | the combined company would have had approximately $8 billion in cash and cash equivalents as of December 31, 2008, after giving effect to the cash payable as merger consideration to the Schering-Plough shareholders and to satisfy transaction expenses; and | |
| • | it is expected that Merck would maintain its existing credit rating following completion of the transaction. |
| • | Maintenance of Merck’s Dividend. Merck’s board of directors considered the ability of the combined entity to pay dividends to its shareholders, and confirmed that it expects the combined company to maintain Merck’s existing annual dividend of $1.52 per share. | |
| • | Substantial Cost Savings. Merck’s board of directors considered the potential for cost savings and synergies from the transaction, including that: |
| • | the companies’ shared therapeutic category focus provides opportunities for consolidation in both sales and marketing and research and development; | |
| • | in addition to the ongoing cost reduction initiatives at both companies, up to $3.5 billion in annual cost savings are expected to be realized from the transaction after 2011; | |
| • | to the extent realized, the savings would allow for greater flexibility for continued investment in strategic opportunities, promising pipeline candidates and licensing opportunities; and | |
| • | there is a probability of meaningful value creation for shareholders of both companies as a result of these savings. |
| • | Accretive to Non-GAAP Earnings. Merck’s board of directors considered that the transaction is anticipated to be modestly accretive to Merck’s non-GAAP earnings per share or EPS, in the first full year following completion of the merger and significantly accretive in the following years. For this purpose, non-GAAP EPS means EPS in accordance with GAAP, excluding purchase-accounting adjustments, restructuring costs, acquisition-related costs and certain other significant items. |
| • | the current and future landscape of the pharmaceutical industry, and in light of the regulatory, financial and competitive challenges facing industry participants, the likelihood that the combined company would be better positioned to overcome these challenges if the expected strategic and financial benefits of the transaction were fully realized; | |
| • | the recommendation of Merck’s management in support of the transaction; | |
| • | the opinion of J.P. Morgan, dated March 8, 2009, that, based upon and subject to the factors and assumptions stated in that opinion, as of such date, the consideration to be paid to the Merck shareholders in the Merck merger is fair, from a financial point of view, to such shareholders; | |
| • | the terms of the proposed financing for the transaction and the fact that Merck would not be required to complete the transaction in the event the full proceeds of the financing were not available to it, even though a $2.5 billion fee would be payable in these circumstances; | |
| • | the views of Merck’s management and its financial advisors as to the likelihood that Merck will be able to obtain the necessary financing and that the full proceeds of the financing will be available to Merck; |
62
Table of Contents
| • | the expectation that the Merck merger will qualify as a reorganization for U.S. federal income tax purposes and that, as a result, the exchange by Merck shareholders of Merck common stock for New Merck common stock in the Merck merger generally will be tax-free to the Merck shareholders; | |
| • | the expected percentage ownership interests and voting power of the Merck shareholders following completion of the merger, and the fact that the stock portion of the merger consideration is a fixed ratio and will not be affected by changes in the market price of Merck’s stock; | |
| • | the required regulatory consents and the views of Merck’s advisors that the merger will be approved by the requisite authorities without the imposition of conditions sufficiently material to preclude the merger, and that the transaction would otherwise be completed in accordance with the terms of the merger agreement; | |
| • | the fact that Merck’s directors and senior management prior to closing will be members of the board of directors and management of the combined company following completion of the merger, and the enhanced value of the combined company that may be realized through continuity of management and implementation of Merck’s long-range strategic plans; | |
| • | the fact that, following completion of the merger, New Merck will be entitled to all of the benefits related toVytorin andZetia, the drugs marketed by the Merck/Schering-Plough cholesterol partnership; | |
| • | the scope and results of Merck’s due diligence investigation, which included reviews of organizational, operational, financial, commercial, regulatory, legal, employee and other matters related toSchering-Plough’s business and potential financial, operational and other impacts of the merger on Merck; | |
| • | after reviewing the merger agreement with its legal advisors, that the terms of the merger agreement offered Merck reasonable assurances as to the likelihood of consummation of the transaction; and | |
| • | that the structure of the transaction would permit Schering-Plough’s revolving credit line of up to $2 billion to remain outstanding following completion of the merger. |
| • | Certain risks inherent in Schering-Plough’s business and operations, including, in particular: |
| • | FDA approval prospects for its product candidates, the investment required to develop experimental compounds and the timing of such development efforts; | |
| • | the various contingent liabilities, including pending legal proceedings, to which Schering-Plough is subject; | |
| • | the results of operations and prospects of Schering-Plough’s global animal health business, the possibility that divestiture of all or a portion of Schering-Plough’s animal health business or Merck’s interests in Merial Ltd. may be required in order to obtain regulatory approvals for the merger, and the possibility of New Merck selling all or a portion of the animal health business to Merial Ltd.; | |
| • | to the extent of issues raised concerning the efficacy ofVytorin andZetia in connection with certain clinical trials, the fact that New Merck will be subject to all of the risk associated with these drugs; and | |
| • | the possibility that Centocor, a wholly owned subsidiary of Johnson & Johnson may, through the required arbitration process, seek to terminate Schering-Plough’s distribution agreement with respect toRemicadeand golimumab, prior to, or following completion of, the merger; and the possibility that Centocor could be successful in convincing an arbitrator that Centocor will have the right to effect such termination and the adverse impact that any termination could have. |
63
Table of Contents
| • | Certain provisions of the merger agreement, including in particular: |
| • | the obligation of Merck to pay a $2.5 billion fee if the drop-dead date occurs, the conditions to Merck’s obligations to close have been satisfied and the merger agreement is terminated because the full proceeds of the financing are not available to Merck; | |
| • | restrictions on Merck’s operations until completion of the transactions, and the extent of those restrictions as negotiated between the parties; | |
| • | Schering-Plough’s right to terminate to enter into a transaction representing a superior proposal; | |
| • | restrictions on Merck’s ability to consider alternative transactions except in limited circumstances; | |
| • | the amount of and circumstances in which Merck may be required to pay termination fees to Schering-Plough and reimburse Schering-Plough for its expenses; | |
| • | the requirement that Merck hold a shareholder vote on the merger agreement, even though the board of directors may have withdrawn its recommendation; | |
| • | the challenges inherent in the combination of two business enterprises of the size and scope of Merck and Schering-Plough, including the possibility the anticipated cost savings and synergies and other benefits sought to be obtained from the merger might not be achieved in the time frame contemplated or at all; | |
| • | the possibility of disruption to business and operational relationships and employee morale as a result of the pending transaction and in the event the merger is not completed; | |
| • | the risks associated with the timing of, the possibility that adverse conditions are imposed in connection with, and the possibility of not obtaining, necessary regulatory approvals required for the transaction; | |
| • | the potential length of the regulatory approval process and the period of time Merck may be subject to the merger agreement; | |
| • | in light of the turbulence in the credit markets, the possibility that the financing for the transaction may not be available and that Merck may be required to pay $2.5 billion under those circumstances; | |
| • | the failure of Merck or Schering-Plough shareholders to approve the merger agreement; | |
| • | the risks described under “Risk Factors” located beginning on page 17; and | |
| • | the risks of not satisfying the closing conditions in the merger agreement. |
64
Table of Contents
| • | reviewed a draft dated March 8, 2009 of the merger agreement; | |
| • | reviewed certain publicly available business and financial information concerning Merck andSchering-Plough and the industries in which they operate; | |
| • | compared the proposed financial terms of the transaction with the publicly available financial terms of certain transactions involving companies J.P. Morgan deemed relevant and the consideration received for such companies; | |
| • | compared the financial and operating performance of Merck and Schering-Plough with publicly available information concerning certain other companies J.P. Morgan deemed relevant and reviewed the current and historical market prices of Merck common stock and Schering-Plough common stock and certain publicly traded securities of such other companies; | |
| • | reviewed certain internal financial analyses and forecasts prepared by (i) the management ofSchering-Plough relating to its businesses and (ii) the management of Merck relating to the respective businesses of Merck and Schering-Plough, as well as the estimated amount and timing of the cost savings and related expenses and synergies expected to result from the proposed merger, which J.P. Morgan refers to as the “synergies”; and | |
| • | performed such other financial studies and analyses and considered such other information as J.P. Morgan deemed appropriate for the purposes of its opinion. |
65
Table of Contents
66
Table of Contents
Segment/Company | Metric/Multiple | |
Consumer | 2009E P/E | |
| Procter & Gamble | 10.9x | |
| Colgate Palmolive | 13.0x | |
| Reckitt-Benckiser | 13.4x | |
| Kimberly Clark | 10.6x | |
| Henkel | 9.2x | |
| Clorox | 12.1x | |
| Church & Dwight | 14.4x | |
| Energizer | 6.8x | |
| Alberto Culver | 15.1x | |
Animal Health | 2009E Firm Value/ EBITDA | |
| Virbac S.A. | 9.1x | |
| Vetoquinol | 5.0x | |
Pharmaceutical | 2009E P/E | |
| Johnson & Johnson | 10.7x | |
| Abbott | 12.7x | |
| Pfizer | 7.0x | |
| Wyeth | 10.4x | |
| Eli Lilly | 6.9x | |
| Bristol-Myers Squibb | 9.7x | |
| Roche | 12.1x | |
| GlaxoSmithKline | 8.1 | |
| Novartis | 8.1x | |
| Sanofi-Aventis | 6.3x | |
| AstraZeneca | 5.6x |
67
Table of Contents
| Firm | ||||||||
| Firm | Value/LTM | |||||||
| Value/LTM | EBITDA & | |||||||
Announcement | Acquiror | Target | EBITDA | Synergies | ||||
| 01/26/2009 | Pfizer | Wyeth | 8.3x | 5.5x | ||||
| 01/26/2004 | Sanofi-Synthelabo | Aventis | 10.2x | 7.9x | ||||
| 07/15/2002 | Pfizer | Pharmacia | 19.0x | 10.6x | ||||
| 02/02/2000 | Pfizer | Warner-Lambert | 31.8x | 20.3x |
68
Table of Contents
69
Table of Contents
70
Table of Contents
| • | Increased Scale and Scope. The Schering-Plough board of directors considered the current and prospective competitive climate in the industry in which Schering-Plough and Merck operate, which includes challenging conditions that are likely to persist, and the relatively better position that the combined company would have in facing such challenges, as a result of: |
| • | the advantages presented by the larger scale and greater scope of the combined company in meeting the challenges facing the pharmaceutical industry in light of changes in regulatory, financial and economic conditions affecting the industry, as well as the possibility of future industry consolidation; | |
| • | the greater financial flexibility of the combined company to invest in promising drug candidates and to invest in internal and external research and development opportunities; and | |
| • | the significantly greater scope of the combined company’s operations and product offerings, including the broadening of the portfolio of blockbuster products and expansion of the global footprint for both companies, resulting in a more diverse mix of business. |
| • | Complementary Products and Customer Bases. The Schering-Plough board of directors considered the complementary nature of the respective products and customer bases of Schering-Plough and Merck and the opportunity created by the transaction to enhance the capabilities of both companies to more effectively and efficiently serve customers. | |
| • | Significant Cost Savings. The Schering-Plough board of directors considered the opportunity for the combined company to achieve significant annual cost savings and revenue opportunities, including: |
| • | savings and revenue opportunities from operational efficiencies, including with respect to a consolidation of the Merck/Schering-Plough cholesterol partnership; and | |
| • | savings and revenue opportunities from consolidating other operations, procurement savings, and sharing support infrastructure and best practices. |
| • | its knowledge of Schering-Plough’s business, operations, financial condition, earnings and prospects and of Merck’s business, operations, financial condition, earnings and prospects, taking into account the results of Schering-Plough’s due diligence review of Merck; | |
| • | the adequacy of the merger consideration and the other value provided to Schering-Plough shareholders including: |
| • | the “spot” implied per share price of the merger consideration on various measurement dates and the premium to the price of Schering-Plough common stock as of various dates represented by such implied prices; | |
| • | the stock component of the merger consideration, which would give Schering-Plough shareholders an equity interest in the combined entity providing the Schering-Plough shareholders an opportunity to |
71
Table of Contents
| benefit from the future performance of the combined Merck and Schering-Plough businesses and synergies resulting from the merger; |
| • | the cash component of the merger consideration, which would allow Schering-Plough shareholders to diversify a portion of their current exposure to the evolving U.S. pharmaceutical industry; | |
| • | the Merck dividend rate, which Merck has stated it will maintain at the combined company and which is three times the current Schering-Plough dividend; and | |
| • | the current market price of Schering-Plough’s common stock, as well as the historical, present and anticipated future earnings of Schering-Plough and the anticipated future earnings of the combined companies; |
| • | the analyses and presentations of Goldman Sachs, including the opinion of Goldman Sachs, dated March 8, 2009, to the Schering-Plough board of directors to the effect that, as of that date, and based upon the factors and subject to the assumptions set forth in such opinion, the merger consideration is fair, from a financial point of view, to Schering-Plough shareholders (other than Merck and its affiliates), as more fully described below under the caption “— Opinions of Schering-Plough’s Financial Advisors — Opinion of Goldman, Sachs & Co.”; | |
| • | the analyses and presentations of Morgan Stanley, including the opinion of Morgan Stanley, dated March 8, 2009, to the Schering-Plough board of directors to the effect that, as of that date, and based upon the factors and subject to the assumptions set forth in such opinion, the merger consideration is fair, from a financial point of view, to Schering-Plough shareholders, as more fully described below under the caption “— Opinions of Schering-Plough’s Financial Advisors — Opinion of Morgan Stanley & Co. Incorporated”; | |
| • | the efforts made to negotiate a merger agreement favorable to Schering-Plough and its shareholders and the financial and other terms and conditions of the merger agreement, including the fact thatSchering-Plough is permitted to terminate the merger agreement in order to approve an alternative transaction proposed by a third party that is a “Superior Proposal” as defined in the merger agreement, upon the payment of a $1.25 billion termination fee, and its belief that such termination fee was reasonable in the context ofbreak-up fees that were payable in other transactions and should not preclude another party from making a competing proposal; | |
| • | the extent of the commitments to obtain required antitrust regulatory approvals that Merck has made under the merger agreement; | |
| • | the fact that Merck has firmly committed financing from a reputable financing source for the merger, the efforts that Merck is required to make under the merger agreement to obtain the proceeds of the financing on the terms and conditions described in the financing commitment letters, and the resulting likelihood that Merck will have the financing available to complete the merger despite the difficulties in the financial markets, including if such difficulties increase in the coming months; | |
| • | the knowledge that another company with a potentially strong strategic fit, and similar capability to effect a transaction such as the one proposed by Merck, and which Schering-Plough’s financial advisors advised may be the only other logical potential bidder for the company, declined to make an offer for a business combination or other arrangement; and | |
| • | the opportunity to combine two strong teams with compatible corporate cultures and a strong and successful existing relationship in connection with the Merck/Schering-Plough cholesterol partnership and the inclusion of three Schering-Plough directors on the board of directors of the combined company. |
| • | the challenges inherent in the combination of two businesses of the size and scope of Schering-Plough and Merck and the size of the companies relative to each other, including the risk that integration costs |
72
Table of Contents
| may be greater than anticipated and the possible diversion of management attention for an extended period of time; |
| • | the risk that changes in the regulatory or competitive landscape may adversely affect the business benefits anticipated to result from the transaction; | |
| • | the risk of not capturing all the anticipated cost savings and operational synergies between Schering-Plough and Merck and the risk that other anticipated benefits might not be realized; | |
| • | the risk that regulatory agencies may not approve the transaction or may impose terms and conditions on their approvals that adversely affect the financial results of the combined company, including divestitures of key businesses (see the section entitled “— Regulatory Approvals” beginning on page 98); | |
| • | the challenges in the financial markets and the risk that the required financing will not be available to Merck; | |
| • | the various contingent liabilities, including pending legal proceedings with respect toSingulair andVioxx, to which Merck is subject; | |
| • | the possibility that Centocor, a wholly owned subsidiary of Johnson & Johnson, would challenge the right of the combined company to maintain its rights under Schering-Plough’s distribution agreement with Centocor with respect toRemicadeand golimumab; | |
| • | the potential impact of the merger announcement and the consummation of the transaction on employees, however the board recognized the overall benefits of the greater scale and size of the combined entity, given the challenges in the pharmaceutical industry, and in light of changes in the regulatory and financial conditions and broader economic changes affecting the industry; and | |
| • | the risks of the type and nature described under “Risk Factors” beginning on page 17 and the matters described under “Cautionary Statement Regarding Forward-Looking Statements” beginning on page 37. |
73
Table of Contents
| • | the merger agreement; | |
| • | annual reports to shareholders and Annual Reports onForm 10-K of Schering-Plough and Merck; | |
| • | certain interim reports to shareholders and Quarterly Reports onForm 10-Q of Schering-Plough and Merck; | |
| • | certain other communications from Schering-Plough and Merck to their respective shareholders; | |
| • | certain publicly available research analyst reports for Schering-Plough and Merck; and | |
| • | certain internal financial analyses and forecasts for Schering-Plough prepared by its management and for Merck by its management, in each case, as approved for Goldman Sachs’s use by Schering-Plough, including certain cost savings and operating synergies projected by the managements of Schering-Plough and Merck to result from the transaction. |
74
Table of Contents
| • | a premium of 33.9% based on the March 6, 2009 market price of $17.63 per share of Schering-Plough common stock and $22.74 per share of Merck common stock; | |
| • | a premium of 43.8% based on the latest one month’s average market price of $18.08 per share of Schering-Plough common stock and $26.87 per share of Merck common stock; | |
| • | a premium of 64.3% based on the latest twelve months’ average market price of $18.06 per share of Schering-Plough common stock and $33.25 per share of Merck common stock; | |
| • | a premium of 50.4% based on the latest two years’ average market price of $23.24 per share of Schering-Plough common stock and $42.42 per share of Merck common stock. |
| • | U.S.: Abbott Laboratories, Bristol-Myers Squibb Company, Eli Lilly and Company, Johnson & Johnson, Pfizer Inc. | |
| • | U.K.: AstraZeneca PLC, GlaxoSmithKline plc | |
| • | Continental Europe: Bayer AG, Merck KgaA, Novartis AG, Novo Nordisk A/S, Roche Holdings Ltd. |
75
Table of Contents
Price/Earnings Multiples | 2009E | |
| Novo Nordisk A/S | 15.4x | |
| Abbott Laboratories | 12.7x | |
| Roche Holdings Ltd. | 11.2x | |
| Johnson & Johnson | 10.7x | |
Schering-Plough | 10.4x | |
| Merck KGaA | 9.6x | |
| Bristol-Myers Squibb Company | 9.5x | |
| Bayer AG | 9.2x | |
| GlaxoSmithKline plc | 8.8x | |
| Novartis AG | 8.5x | |
Merck | 7.0x | |
| Eli Lilly and Company | 6.7x | |
| Sanofi-Aventis | 6.6x | |
| Pfizer Inc. | 6.5x | |
| AstraZeneca PLC | 5.7x |
| Illustrative Discounted Cash Flow | ||||||||||||
| Value (per Share) of Schering-Plough | ||||||||||||
| (IncludingMerck/Schering-Plough | ||||||||||||
| Cholesterol Partnership) | ||||||||||||
| Discount Rate | ||||||||||||
Perpetuity Growth Rate | 8.0% | 8.5% | 9.0% | |||||||||
0.0% | $ | 20.93 | $ | 19.78 | $ | 18.75 | ||||||
1.0% | $ | 22.76 | $ | 21.36 | $ | 20.13 | ||||||
2.0% | $ | 25.20 | $ | 23.43 | $ | 21.90 | ||||||
76
Table of Contents
| Illustrative Pro Forma | ||||||||||||
| Discounted Cash Flow Value | ||||||||||||
| (per Share) of the Combined Company | ||||||||||||
| Discount Rate | ||||||||||||
Perpetuity Growth Rate | 8.0% | 8.5% | 9.0% | |||||||||
(1.0)% | $ | 43.47 | $ | 41.16 | $ | 39.07 | ||||||
0.0% | $ | 47.23 | $ | 44.46 | $ | 41.99 | ||||||
1.0% | $ | 52.06 | $ | 48.64 | $ | 45.62 | ||||||
| • | 2009: Pfizer Inc. — Wyeth | |
| • | 2004: Sanofi SA — Aventis | |
| • | 2002: Pfizer Inc. — Pharmacia Corporation | |
| • | 2000: Pfizer Inc. — Warner-Lambert Company; Glaxo Wellcome plc — SmithKline Beecham plc | |
| • | 1999 — Monsanto Company — Pharmacia & Upjohn, Inc.; American Home Products Corporation — Warner-Lambert Company |
77
Table of Contents
78
Table of Contents
| Date | Type of | Premium | ||||||||
Target | Acquirer | Announced | Consideration | Paid | ||||||
| Schering-Plough | Merck | March 2009 | cash/stock | 44.7 | % | |||||
| Synthelabo SA | Sanofi SA | December 1998 | all stock | 39.6 | % | |||||
| Pharmacia Corporation | Pfizer Inc. | July 2002 | all stock | 34.0 | % | |||||
| Schering AG | Bayer AG | March 2006 | all cash | 33.4 | % | |||||
| Aventis | Sanofi SA | January 2004 | cash/stock | 31.2 | % | |||||
| Warner-Lambert Company | Pfizer Inc. | November 1999 | all stock | 29.8 | % | |||||
| Wyeth | Pfizer Inc. | January 2009 | cash/stock | 28.8 | % | |||||
79
Table of Contents
80
Table of Contents
| • | reviewed certain publicly available financial statements and other business and financial information of Schering-Plough and Merck, respectively; | |
| • | reviewed certain internal financial statements and other financial and operating data concerning Schering-Plough and Merck, respectively; | |
| • | reviewed certain financial projections prepared by the managements of Schering-Plough and Merck, respectively; | |
| • | reviewed information relating to certain strategic, financial and operational benefits anticipated from the merger, prepared by the managements of Schering-Plough and Merck, respectively; | |
| • | discussed the past and current operations and financial condition and the prospects of Schering-Plough, including information relating to certain strategic, financial and operational benefits anticipated from the transaction, with senior executives of Schering-Plough; | |
| • | discussed the past and current operations and financial condition and the prospects of Merck, including information relating to certain strategic, financial and operational benefits anticipated from the transaction, with senior executives of Merck; | |
| • | reviewed the pro forma impact of the transaction on Merck’s earnings per share, cash flow, consolidated capitalization and financial ratios; | |
| • | reviewed the reported prices and trading activity for Schering-Plough’s common stock and Merck’s common stock; | |
| • | compared the financial performance of Schering-Plough and Merck and the prices and trading activity of Schering-Plough’s common stock and Merck’s common stock with that of certain other publicly traded companies comparable with Schering-Plough and Merck, respectively, and their securities; | |
| • | reviewed the financial terms, to the extent publicly available, of certain comparable acquisition transactions; | |
| • | participated in certain discussions and negotiations among representatives of Schering-Plough and Merck and their financial and legal advisors; | |
| • | reviewed the draft merger agreement dated March 7, 2009, the draft commitment letter from J.P. Morgan Securities Inc. and JPMorgan Chase Bank, N.A. substantially in the form of the draft dated March 6, 2009 and certain related documents; and | |
| • | performed such other analyses, reviewed such other information and considered such other factors as Morgan Stanley had deemed appropriate. |
81
Table of Contents
82
Table of Contents
| Schering-Plough | ||||||||||||||||
| Merck Average | Average Price per | Implied Premium to | ||||||||||||||
| Price per Share of | Implied | Share of Common | Schering-Plough | |||||||||||||
Range | Common Stock | Offer Value | Stock | Average Price | ||||||||||||
| Unaffected | $ | 22.74 | $ | 23.61 | $ | 16.32 | 45 | % | ||||||||
| 1 calendar month | $ | 27.29 | $ | 26.24 | $ | 18.19 | 44 | % | ||||||||
| 3 calendar months | $ | 28.13 | $ | 26.72 | $ | 17.82 | 50 | % | ||||||||
| 6 calendar months | $ | 28.64 | $ | 27.01 | $ | 16.85 | 60 | % | ||||||||
| 1 calendar year | $ | 33.37 | $ | 29.74 | $ | 18.09 | 64 | % | ||||||||
| 2 calendar years | $ | 42.48 | $ | 35.00 | $ | 23.26 | 50 | % | ||||||||
| 3 calendar years | $ | 41.64 | $ | 34.51 | $ | 22.53 | 53 | % | ||||||||
| Implied Exchange | ||||||||
Time Period | Exchange Ratio | Ratio Premium | ||||||
| 3 calendar months | 0.635 | x | 64 | % | ||||
| 6 calendar months | 0.589 | x | 76 | % | ||||
| 1 calendar year | 0.551 | x | 89 | % | ||||
| 2 calendar years | 0.552 | x | 88 | % | ||||
| 3 calendar years | 0.544 | x | 91 | % | ||||
| Premium to | ||||
| Price 1-Day | ||||
| Prior to | ||||
Precedent Transactions Premia | Announcement | |||
| Mean | 26.6 | % | ||
| Median | 28.8 | % | ||
| High | 52.9 | % | ||
| Low | (1.1 | )% | ||
83
Table of Contents
| Representative | Implied Value | |||
Precedent Transaction Financial Statistic | Premium Range | per Share | ||
| Premium to Unaffected Stock Price | 25% - 35% | $20.40 - $22.03 |
| • | Abbott Laboratories Inc. | |
| • | Amgen Inc. | |
| • | Bristol-Myers Squibb Co. | |
| • | Eli Lilly & Co. | |
| • | Johnson & Johnson | |
| • | Merck & Co. Inc. | |
| • | Pfizer Inc. |
| • | AstraZeneca P.L.C. | |
| • | GlaxoSmithKline P.L.C. | |
| • | Novartis A.G. | |
| • | Roche Holding A.G. | |
| • | Sanofi-Aventis S.A. |
84
Table of Contents
| • | the closing stock price divided by the estimated IBES consensus EPS for calendar year 2009, referred to below as the P/E multiple; and | |
| • | the annual dividend divided by the closing stock price, referred to below as the dividend yield. |
85
Table of Contents
86
Table of Contents
87
Table of Contents
| 2009E P/E Multiple | ||||||||||||
| Merck | Blended | DCF | ||||||||||
Valuation Basis | (7.0x) | (7.7x) | Value | |||||||||
| (In billions of dollars) | ||||||||||||
| Total Value of Synergies | $ | 17.1 | $ | 19.0 | $ | 17.3 | ||||||
| Premium Paid as a Percentage of Total Value of Synergies | 75.8 | % | 68.2 | % | 74.6 | % | ||||||
88
Table of Contents
89
Table of Contents
90
Table of Contents
91
Table of Contents
92
Table of Contents
93
Table of Contents
94
Table of Contents
| • | Any change or termination of the Distribution Agreement with Centocor is excluded by the merger agreement from the definition of “material adverse effect” both with respect to Merck and Schering-Plough and is excluded from the definition of “material adverse effect” in the credit agreements for the credit facilities entered into in connection with financing the merger. | |
| • | The estimated annual cost savings of $3.5 billion expected to be realized from the transaction annually after 2011 is not dependent on the retention of the rights to distributeRemicadeand golimumab, although the loss of these rights would reduce the amount of sales expected to be generated by the combined company. | |
| • | The anticipated continued payment by the combined company of the current Merck dividend of $1.52 per share annually is not conditioned on the retention of the rights to distributeRemicadeand golimumab. |
95
Table of Contents
| • | a $3 billion364-day bridge loan agreement which we refer to as the bridge loan facility; | |
| • | a $3 billion364-day asset sale facility agreement which we refer to as the asset sale facility; and |
96
Table of Contents
| • | a $1 billion364-day incremental loan agreement which we refer to as the incremental facility. |
| • | the consummation of the merger in accordance with the merger agreement; | |
| • | the absence, since December 31, 2008, of any material adverse change (as defined in the new credit facilities) with respect to Merck and Schering-Plough taken as a whole; | |
| • | the execution of definitive documentation with respect to the new credit facilities and, if applicable, the amendment to Merck’s existing revolving credit facility (which condition has been satisfied); | |
| • | certification by the chief financial officer of Merck that the ratio of total debt to capitalization of the combined company on a pro forma basis as of the last fiscal quarter ended at least 45 days before closing does not exceed 60%; and | |
| • | other customary closing conditions each as more fully described in the new credit facilities. |
97
Table of Contents
98
Table of Contents
99
Table of Contents
100
Table of Contents
101
Table of Contents
102
Table of Contents
103
Table of Contents
| • | organization and qualification; | |
| • | significant subsidiaries; | |
| • | corporate authority to enter into the merger agreement and the other transactions contemplated by the merger agreement; | |
| • | required governmental approvals; | |
| • | capital stock; | |
| • | SEC filings, financial statements and compliance with the Sarbanes-Oxley Act of 2002; | |
| • | absence of changes or events that have had or could reasonably be expected to have a material adverse effect; | |
| • | existence and validity of, and compliance with, material contracts; | |
| • | intellectual property; | |
| • | litigation; | |
| • | possession of requisite permits and compliance with laws; | |
| • | regulatory compliance; | |
| • | certain employee benefits matters; | |
| • | taxes and other tax matters; | |
| • | maintenance of adequate insurance; | |
| • | opinions of their respective financial advisors; | |
| • | interested shareholders; and | |
| • | inapplicability of state anti-takeover statutes and rights agreements. |
| • | certain labor and employee relations matters; | |
| • | compliance with environmental laws and certain other environmental matters; | |
| • | transactions with affiliates; | |
| • | brokers’ and finders’ fees; |
104
Table of Contents
| • | ownership and operation of Merger Sub 1 and Merger Sub 2; and | |
| • | certain intercompany notes. |
| • | any change or development in United States financial, credit or securities markets, general economic or business conditions, or political or regulatory conditions, to the extent such changes do not have a disproportionate effect on the business, financial condition or results of operations of the party and its subsidiaries, taken as a whole, relative to other participants in the pharmaceutical (including animal health, biotechnology and consumer health) industry; | |
| • | any act of war, armed hostilities or terrorism or any worsening thereof, to the extent such changes do not have a disproportionate effect on the business, financial condition or results of operations of the party and its subsidiaries, taken as a whole, relative to other participants in the pharmaceutical (including animal health, biotechnology and consumer health) industry; | |
| • | any change in law or U.S. GAAP or the interpretation or enforcement of either, to the extent such changes do not have a disproportionate effect on the business, financial condition or results of operations of the party and its subsidiaries, taken as a whole, relative to other participants in the pharmaceutical (including animal health, biotechnology and consumer health) industry; | |
| • | any change in the pharmaceutical (including animal health, biotechnology and consumer health) industry, to the extent such changes do not have a disproportionate effect on the business, financial condition or results of operations of the party and its subsidiaries, taken as a whole, relative to other participants in the pharmaceutical (including animal health, biotechnology and consumer health) industry; | |
| • | the negotiation, execution, delivery, performance, consummation, potential consummation or public announcement of the merger agreement or the transactions contemplated by the merger agreement, including any litigation resulting therefrom or with respect thereto, and any adverse change in customer, distributor, employee, supplier, financing source, licensor, licensee,sub-licensee, shareholder, co-promotion, collaboration or joint venture partner or similar relationships resulting therefrom or with respect thereto, including as a result of the identity of the other party to the merger; | |
| • | any failure of Schering-Plough or Merck, as applicable, or any of its subsidiaries to meet, with respect to any period or periods, any internal or industry analyst projections, forecasts, estimates of earnings or revenues, or business plans (however, the facts and circumstances giving rise to such failure that are not otherwise excluded from the definition of material adverse effect may be taken into account in determining whether a material adverse effect has occurred); | |
| • | any change, in and of itself, in the market price or trading volume of the Schering-Plough or Merck common stock, as applicable (however, the facts and circumstances giving rise to such change that are not otherwise excluded from the definition of material adverse effect may be taken into account in determining whether a material adverse effect has occurred); | |
| • | the taking of any action required by the merger agreement; and | |
| • | certain specified events related to or arising out of or in connection with the business(es) of Merck and Schering-Plough. |
105
Table of Contents
| • | propose or adopt any changes to its organizational documents or the organizational documents of its subsidiaries; | |
| • | declare or pay any dividend or distribution on any shares of capital stock other than dividends paid by a wholly owned subsidiary of Schering-Plough to its parent corporation and regular quarterly dividends of not more than $0.065 per share paid on the common stock of Schering-Plough and $3.75 per share on the Schering-Plough 6% preferred stock; | |
| • | adjust, split, combine, reclassify or otherwise amend the terms of its capital stock or equity interests or issue, grant, deliver, sell, repurchase, redeem, acquire, encumber, pledge, dispose of or otherwise transfer any shares of its capital stock or equity interests, or any securities convertible into or exchangeable or exercisable for its capital stock or equity interests, other than pursuant to the exercise of options and settlement of restricted stock units, deferred stock units and performance awards or pursuant to the conversion of outstanding convertible preferred stock, provided that Schering-Plough may continue to grant equity awards in the ordinary course of business consistent with past practice to its and its subsidiaries’ directors, officers and employees; | |
| • | increase the compensation to directors, officers or employees, except for, with respect to officers and employees who are not senior employees, certain increases made in the ordinary and usual course of business consistent with past practice in timing and amount and increases made in connection with the completion of Schering-Plough’s integration of global compensation and benefits and, with respect to certain senior employees, certain pre-approved increases; increase the benefits of any past or present director, officer, employees or service providers who provide services exclusively to Schering-Plough except for increases made in the ordinary and usual course of business consistent with past practice and increases made in connection with the completion of Schering-Plough’s integration of global compensation and benefits; with certain exceptions, grant any severance or termination pay to any of its past or present directors, officers, employees or other service providers; enter into, amend or modify any employment, severance, consulting or change in control agreement with any person; with certain |
106
Table of Contents
| exceptions, establish, adopt, enter into, amend or take any action to accelerate rights under any Schering-Plough employee plans; pay, accrue or certify performance level achievements at levels in excess of actually achieved performance in respect of any component of an incentive-based award that requires achievement at a specified level; take any affirmative action to amend or waive performance or vesting criteria or accelerate distribution, settlement or funding under any compensation or benefit plan; take any action with respect to salary, compensation, benefits or other terms of employment that would result in a senior employee having “good reason” to terminate employment and collect severance benefits and payments pursuant to any change in control agreement; and, without consulting with Merck, terminate the employment of a senior employee in a manner that would cause the senior employee to collect severance payments; |
| • | merge or consolidate or effect any share exchange involving any class of the capital stock ofSchering-Plough with any person; | |
| • | sell, dispose of, lease, guarantee or encumber any property or assets except (i) for the sale of goods and services in the ordinary and usual course of business, (ii) involving property or assets having a value no greater than $100 million in the aggregate, (iii) in connection with any waiver, release or settlement of litigation otherwise permitted by the merger agreement, (iv) in connection with cash management or investment portfolio activities, (v) in connection with the sale or pledge of accounts receivable, or (vi) in connection with the pledge of cash for letters of credit purposes; | |
| • | make any acquisitions in excess of $25 million individually or $50 million in the aggregate by purchase or other acquisition of assets, stock or other equity interests, or by merger, consolidation or other business combination; | |
| • | enter into any strategic licensing, joint venture or similar contract involving non-contingent consideration valued in excess of $50 million individually or $100 million in the aggregate; | |
| • | with respect to its corporate cash investment portfolio and not to its pension plan or similar benefit plans, purchase material financial instruments that qualify as Level III assets (as defined by FASB Statement No. 157), change in a material manner the average duration of its investment portfolio to more than six months, materially change investment guidelines with respect to Schering-Plough’s investment portfolio, hypothecate, enter into repurchase agreements with respect to or otherwise pledge assets in its investment portfolio, or invest surplus cash from operations in securities other than short term liquid securities; | |
| • | enter into material interest rate swaps, foreign exchange or commodity agreements and other similar hedging arrangements; | |
| • | renew, extend, materially amend, terminate, or grant material waivers under any material contract or enter into any new contract that would constitute a material contract; | |
| • | incur any indebtedness or issue any debt securities or other rights to acquire debt securities or indebtedness of Schering-Plough, in each case in excess of $200 million; | |
| • | prepay any long term indebtedness or change the terms or extend the maturity thereof; | |
| • | make any loans, capital contributions to, or investment in any person; | |
| • | make capital expenditures in excess of $750 million during fiscal year 2009 or in excess of $200 million during any quarter in 2010, or undertake or enter into commitments in 2009 that will require capital expenditures beyond 2009 in excess of $25 million for any one capital project; | |
| • | change its financial accounting policies or procedures in effect as of December 31, 2008, other than as required by law or GAAP, or write up, write down or write off the book value of any material assets; | |
| • | waive, release, assign, settle or compromise any proceeding that would involve the payment, whether or not covered or reimbursed by insurance, of an amount in excess of $250 million or injunctive relief that would materially limit or restrict the business of Schering-Plough and its subsidiaries, taken as a whole, or, to the knowledge of Schering-Plough, the business of New Merck and its subsidiaries, taken as a |
107
Table of Contents
| whole, following the effective time of the merger, or enter into certain settlements of patent infringement claims brought by Schering-Plough or its subsidiaries against a generic manufacturer; |
| • | adopt a plan of complete or partial liquidation or resolutions providing for a complete or partial liquidation, dissolution or recapitalization of the shares of Schering-Plough; | |
| • | make or change any material tax election (except in the ordinary course of business consistent with past practice), file any material amendment to a material tax return, settle or compromise any material tax audit or enter into any material closing agreement, change any annual tax accounting period, adopt or change any tax accounting method, surrender any right to claim a material refund of taxes or consent to any extension or waiver of the limitation period applicable to any material tax claim or assessment relating to Schering-Plough or its subsidiaries; | |
| • | enter into, renew, extend, amend, grant a waiver under or terminate any affiliate transaction; | |
| • | enter into, modify, amend or terminate any contract, or waive, release or assign any rights or claims under a contract, which would be reasonably likely to impair the ability of Schering-Plough to perform its obligations under the merger agreement in any material respect or prevent or materially delay the consummation of the transaction; or take any other action that would reasonably be expected to impede or interfere with the consummation of the merger or the other transactions contemplated by the merger agreement; | |
| • | amend, modify, terminate, prepay, repay or satisfy any portion of certain intercompany notes, except required interest payments in accordance with the terms of such intercompany notes; | |
| • | enter into a noncompetition contract or any other contract containing a provision that expressly limits or otherwise restricts Schering-Plough or its subsidiaries or would, after the effective time of the Merck merger, expressly limit or otherwise restrict New Merck or its subsidiaries, from engaging in any line of business or in any geographic area or from developing or commercializing any compounds, any therapeutic area, class of drugs or mechanism of action, in a manner that would be reasonably likely to be material to Schering-Plough and its subsidiaries, taken as a whole, or New Merck and its subsidiaries, taken as a whole (as the case may be); or | |
| • | authorize or commit or agree to take any of the foregoing actions. |
| • | propose or adopt any changes to its organizational documents or the organizational documents of its subsidiaries; | |
| • | declare or pay any dividend or distribution on any shares of capital stock other than dividends paid by a wholly owned subsidiary of Merck to its parent corporation and regular quarterly dividends of not more than $0.38 per share paid on the common stock of Merck; | |
| • | adjust, split, combine, reclassify or otherwise amend the terms of its capital stock or equity interests or issue, grant, deliver, sell, acquire, encumber, dispose of or otherwise transfer any shares of its capital stock or equity interests, or any securities convertible into or exchangeable or exercisable for its capital stock or equity interests, other than pursuant to the exercise of options and settlement of other equity awards of Merck; | |
| • | merge or consolidate or effect any share exchange with, or sell substantially all of its assets to, any person; | |
| • | enter into, modify, amend or terminate any contract, or waive, release or assign any rights or claims under a contract, which would be reasonably likely to impair the ability of Merck to perform its obligations under the merger agreement in any material respect or prevent or materially delay the consummation of the merger; or take any other action that would reasonably be expected to impede or interfere with the consummation of the merger or the other transactions contemplated by the merger agreement; |
108
Table of Contents
| • | adopt a plan of complete or partial liquidation or resolutions providing for a complete or partial liquidation, dissolution or recapitalization; or | |
| • | authorize or commit or agree to take any of the foregoing actions. |
| • | solicit, initiate, encourage or knowingly facilitate any inquiries or the making of any proposal or offer that constitutes or could reasonably be expected to lead to an acquisition proposal; | |
| • | engage in discussions or negotiations regarding, or that could reasonably be expected to lead to, an acquisition proposal, furnish to any third party any information in connection with an acquisition proposal, or otherwise cooperate with or assist a third party that has made or is seeking to make an acquisition proposal; | |
| • | fail to make, or withdraw or amend its board of directors’ recommendation that its shareholders vote in favor of the merger or recommend, adopt or approve another acquisition proposal, or take any action or make any statement inconsistent with the board of director’s recommendation that its shareholders vote in favor of the merger; | |
| • | take any action to make any anti-takeover statute or anti-takeover provision in its certificate of incorporation or bylaws inapplicable to any transaction contemplated by an acquisition proposal; | |
| • | except with respect to a confidentiality agreement permitted to be entered into as described below, enter into any agreement, term sheet or other instrument constituting or relating to an acquisition proposal or which compels the termination or breach of the merger agreement or the failure to consummate the merger; | |
| • | enter into any confidentiality agreement which prohibits them from making available, within certain time periods, to the other party, Merck or Schering-Plough, as the case may be, any information to be provided to a third party; | |
| • | grant or permit any party a waiver under any confidentiality or standstill agreement; and | |
| • | resolve or propose to do any of the foregoing. |
| • | Engage in discussions or negotiations with a third party that has made a superior proposal or an acquisition proposal that the board determines in good faith could reasonably lead to a superior proposal and that the board determines in good faith is credible and reasonably capable of consummating a superior proposal; | |
| • | Thereafter, furnish to such third party nonpublic information pursuant to a confidentiality agreement with terms no less materially favorable than those contained in the confidentiality agreement between Merck and Schering-Plough and which contains a standstill provision on terms no more materially favorable than any standstill provision applicable to Merck and Schering-Plough, as the case may be, provided that any such standstill or similar provision may allow such third party to make acquisition proposals to Merck or Schering-Plough, as the case may be, in connection with the negotiations or discussions permitted by the merger agreement, and provided that such information must be provided to the other party, Merck or Schering-Plough, as the case may be, prior to or substantially concurrently with the time that such information is provided to the third party; and | |
| • | In response to a superior proposal or an intervening event, change its recommendation to its shareholders that they vote in favor of the merger agreement. |
109
Table of Contents
| • | It first notifies the other party, at least three business days prior to taking such action, of its intent to take such action; | |
| • | If requested by the other party, it negotiates during the three business day period described above with respect to a revised proposal from the other party; and | |
| • | In the case of a change of recommendation in response to a superior proposal, the other party does not make a proposal within the three business day period that is at least as favorable to shareholders as such superior proposal. |
| • | A merger or similar transaction pursuant to which such third party would own 15% or more of the assets, revenues or net income of Merck or Schering-Plough, as applicable, and its subsidiaries, as a whole; | |
| • | A sale, lease or other disposition of assets of Merck or Schering-Plough, as applicable (including equity interests of their respective subsidiaries) representing 15% or more of the assets, revenues or net income of Merck or Schering-Plough, as applicable, and its subsidiaries, as a whole; | |
| • | The issuance, sale or other disposition of equity interests representing 15% or more of the voting power of Merck or Schering-Plough, as applicable; | |
| • | A transaction or series of transactions in which any third party would acquire beneficial ownership of equity interests representing 15% or more of the voting power of Merck or Schering-Plough, as applicable; or | |
| • | Any combination of the foregoing. |
| • | Actions, and the consequences of any such actions, taken pursuant to the provisions of the merger agreement with respect to seeking and gaining regulatory approvals; | |
| • | Receipt of, existence of or terms of an acquisition proposal or any inquiry related thereto or the consequences thereof; and | |
| • | Any event or events that has or have an adverse effect on the business, financial condition, assets, liabilities, results of operations or market price of the other party or its subsidiaries unless such event or events has had or would reasonably be expected to have a material adverse effect on the other party. |
110
Table of Contents
| • | Schering-Plough employees will continue to participate in the short-term incentive program for the 2009 performance year that is already in place at Schering-Plough. If the closing of theSchering-Plough merger does not occur in 2009, awards will generally be paid in the ordinary course. If the closing of the Schering-Plough merger occurs in 2009, the performance and amount of the awards for Schering-Plough employees who are employed by New Merck on December 31, 2009 will be |
111
Table of Contents
| determined at the end of 2009, using the performance metrics originally set by Schering-Plough but calculated on the actual performance through the closing date of the Schering-Plough merger (or a good faith estimate of actual performance), and will be payable by March 15, 2010. A Schering-Plough employee who is terminated in 2009 will be entitled to a pro-rata portion of his or her 2009 target bonus based on his or her termination date. |
| • | If the closing of the Schering-Plough merger has not occurred before January 1, 2010, the Schering-Plough employees will be paid a pro-rated 2010 target bonus for the portion of the 2010 performance period occurring between January 1, 2010 and the closing of the Schering-Plough merger. After the closing of the Schering-Plough merger, for the remaining portion of 2010 (or the whole portion of 2010, if the closing of the Schering-Plough merger occurs on or before January 1, 2010), the Schering-Plough employees will participate in the short term incentive award programs of New Merck in the same manner as such programs apply to the participants who were formerly Merck employees. A Schering-Plough employee who is terminated in 2010 will be entitled to a pro-rata portion of his or her 2010 target bonus based on his or her termination date. | |
| • | Beginning January 1, 2010, Schering-Plough employees will be eligible to participate in any equity compensation plans of New Merck to the same extent and on the same terms and conditions as similarly situated participants who were formerly Merck employees. In the event that theSchering-Plough merger is completed after the date on which equity compensation awards were granted to Merck employees for 2010, Schering-Plough employees will be granted equity compensation awards from New Merck as soon as practicable following completion of the Schering-Plough merger to the same extent and on the same terms and conditions as similarly situated participants who were formerly Merck employees. | |
| • | New Merck will maintain Schering-Plough’s severance benefit plan through December 31, 2010 (or the period required under the terms of the plan, if longer) and will provide any Schering-Plough employee who qualifies for benefits under the plan with the severance payments and benefits required under the plan. New Merck will not be prohibited from terminating the employment of any Schering-Plough employee for any reason. |
112
Table of Contents
113
Table of Contents
| • | Merck shareholder approval of the merger agreement and Schering-Plough shareholder approval of the merger agreement and the issuance of shares of common stock of New Merck in connection with the merger; | |
| • | the termination or expiration of the waiting period under the HSR Act, the approval of the merger by the European Commission, and the approval, or termination or expiration of certain other antitrust waiting periods in, specified jurisdictions outside the United States and the European Union; | |
| • | the absence of an injunction, order or law issued or enacted by a governmental entity in the United States, the European Union or certain other specified jurisdictions that enjoins or otherwise makes illegal the consummation of the merger; | |
| • | the effectiveness of the registration statement onForm S-4 of which this joint proxy statement/prospectus is a part and the absence of a stop order suspending its effectiveness; | |
| • | the approval for listing on the NYSE of the shares of common stock of New Merck issuable to holders of Merck common stock and Schering-Plough common stock; | |
| • | the accuracy of the representations of the other party (with certain exceptions for inaccuracies that are de minimis, not material or would not have a material adverse effect on that other party) and receipt of an officer’s certificate to that effect; | |
| • | the performance in all material respects by the other party of its material agreements and covenants in the merger agreement and receipt of an officer’s certificate to that effect; and | |
| • | the absence of the occurrence of an event or events that have had or would be reasonably expected to have a material adverse effect on the other party, and receipt of an officer’s certificate to that effect. |
| • | there shall not have been any commitment, undertaking, agreement or law or order (1) requiring the license, sale, divestiture or other disposition of any equity interests of a subsidiary of Merck or |
114
Table of Contents
| Schering-Plough, or business, assets or products of Merck, Schering-Plough or their respective subsidiaries or (2) that otherwise places a limitation on the freedom of action of Merck orSchering-Plough with respect to, or their ability to retain, any business, asset or product, which in the case of (1) and (2) would result in, individually or in the aggregate, a one-year loss of net sales revenues (based on 2008 net sales revenues) in excess of $1 billion (excluding any loss of net sales revenues related to the license, sale, divestiture or other disposition or holding separate ofSchering-Plough’s animal health segment and Merck’s direct or indirect interest in Merial Ltd.); and |
| • | Merck shall have received a written opinion from its counsel that the Merck merger will be treated for U.S. federal income tax purposes as a reorganization within the meaning of Section 368(a) of the Code. |
| • | by mutual written consent of Merck and Schering-Plough; | |
| • | by either Merck or Schering-Plough if a governmental entity in the United States, European Union or certain other specified jurisdictions has issued a final order, decree, or injunction or taken any other action permanently restraining or otherwise prohibiting the consummation of the merger or if any law has been adopted in the United States, European Union or certain other specified jurisdictions that makes the consummation of the merger illegal or otherwise prohibited (provided that the party seeking to terminate under these circumstances must have fulfilled its obligations under the merger agreement with respect to obtaining regulatory approvals); | |
| • | by Merck or Schering-Plough if the merger has not been consummated by a “drop-dead date” of December 8, 2009, provided that the drop-dead date on which the merger agreement may be terminated will be extended to March 8, 2010 if, on December 8, 2009: |
| • | the closing conditions dealing with antitrust approvals, laws or injunctions prohibiting the merger and regulatory divestitures have not been satisfied but all other conditions to the merger have been satisfied; or | |
| • | the proceeds of the financing are not available to Merck in full but all other conditions to the merger have been satisfied. |
| • | by Merck if Schering-Plough’s representations or warranties were or become inaccurate or Schering-Plough breaches a covenant such that the closing conditions relating to the accuracy of Schering-Plough’s representations and warranties or Schering-Plough’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date; | |
| • | by Schering-Plough if Merck’s representations or warranties were or become inaccurate or Merck breaches a covenant such that the closing conditions relating to the accuracy of Merck’s representations and warranties or Merck’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date; | |
| • | by Merck if: |
| • | Schering-Plough’s board of directors changes its recommendation; | |
| • | following any bona fide acquisition proposal first being publicly announced or disclosed prior to Schering-Plough shareholder approval, Schering-Plough fails to issue a press release reaffirming its recommendation within ten business days of Merck’s request to do so; | |
| • | any tender offer or exchange offer by a third party with respect to Schering-Plough’s common stock constituting an acquisition proposal is commenced and within ten business days of commencement of |
115
Table of Contents
| the offer, Schering-Plough’s board of directors does not recommend that Schering-Plough’s shareholders reject such offer or issue a press release reaffirming its recommendation in favor of the merger agreement and the issuance of shares; |
| • | Schering-Plough or its board of directors approves, endorses, recommends, adopts or enters into an acquisition proposal or a contract relating to an acquisition proposal; | |
| • | Schering-Plough materially breaches its non-solicitation covenants; | |
| • | Schering-Plough materially breaches its obligation to hold its shareholder meeting; or | |
| • | Schering-Plough or its board of directors announces, resolves or proposes to do any of the foregoing; |
| • | by either Merck or Schering-Plough if Schering-Plough does not receive shareholder approval at its shareholder meeting; | |
| • | by either Merck or Schering-Plough if Merck does not receive shareholder approval at its shareholder meeting; | |
| • | by Schering-Plough if: |
| • | Merck’s board of directors changes its recommendation; | |
| • | following any bona fide acquisition proposal first being publicly announced or disclosed prior to Merck shareholder approval, Merck fails to issue a press release reaffirming its recommendation within ten business days of Schering-Plough’s request to do so; | |
| • | any tender offer or exchange offer by a third party with respect to Merck’s common stock constituting an acquisition proposal is commenced and within ten business days of commencement of the offer, Merck’s board of directors does not recommend that Merck’s shareholders reject such offer or issue a press release reaffirming its recommendation in favor of the merger agreement; | |
| • | Merck or its board of directors approves, endorses, recommends, adopts or enters into an acquisition proposal or a contract relating to an acquisition proposal; | |
| • | Merck materially breaches its non-solicitation covenants; | |
| • | Merck materially breaches its obligation to hold its shareholder meeting; or | |
| • | Merck or its board of directors announces, resolves or proposes to do any of the foregoing; |
| • | by Schering-Plough at any time prior to the time at which it receives its shareholder approval, if the board of directors of Schering-Plough determines to enter into a definitive agreement with respect to a superior proposal, provided it pays to Merck the termination fee, and reimburses Merck’s out of pocket expenses, as described below. |
116
Table of Contents
| • | Merck terminates the merger agreement because: |
| • | Schering-Plough’s board of directors changes its recommendation; |
| • | following any bona fide acquisition proposal first being publicly announced or disclosed prior to Schering-Plough shareholder approval, Schering-Plough fails to issue a press release reaffirming its recommendation within ten business days of Merck’s request to do so; | |
| • | any tender offer or exchange offer by a third party with respect to Schering-Plough’s common stock constituting an acquisition proposal is commenced and, within ten business days of commencement of the offer, Schering-Plough’s board of directors does not recommend that Schering-Plough’s shareholders reject such offer or issue a press release reaffirming its recommendation in favor of the merger and the issuance of shares; | |
| • | Schering-Plough or its board of directors approves, endorses, recommends, adopts or enters into an acquisition proposal or a contract relating to an acquisition proposal; | |
| • | Schering-Plough materially breaches its non-solicitation covenants; | |
| • | Schering-Plough materially breaches its obligation to hold its shareholder meeting; or | |
| • | Schering-Plough or its board of directors announces, resolves or proposes to do any of the foregoing; |
| • | Either Merck or Schering-Plough terminates the merger agreement because: |
| • | the merger has not been consummated by the drop-dead date and an acquisition proposal was made for Schering-Plough prior to such termination, whether or not publicly announced, and within twelve months after such termination, Schering-Plough enters into a definitive agreement with respect to, or consummates an acquisition proposal (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%); or | |
| • | the Schering-Plough shareholders do not approve the merger and the share issuance and an acquisition proposal for Schering-Plough was publicly announced prior to the Schering-Plough shareholders meeting and within twelve months after such termination, Schering-Plough enters into a definitive agreement with respect to, or consummates, an acquisition proposal (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%); |
| • | Merck terminates the merger agreement because Schering-Plough’s representations or warranties were or become inaccurate or Schering-Plough breaches a covenant such that the closing conditions relating to the accuracy of Schering-Plough’s representations and warranties or Schering-Plough’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date and an acquisition proposal was made for Schering-Plough prior to such termination, whether or not publicly announced, and within twelve months after such termination, Schering-Plough enters into a definitive agreement with respect to or consummates an acquisition proposal (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%); or | |
| • | Schering-Plough terminates the merger agreement at any time prior to the time at which it receives its shareholder approval if the board of directors of Schering-Plough determines to enter into a definitive agreement with respect to a superior proposal. |
| • | Schering-Plough terminates the agreement because: |
| • | Merck’s board of directors changes its recommendation; |
117
Table of Contents
| • | following any bona fide acquisition proposal first being publicly announced or disclosed, Merck fails to issue a press release reaffirming its recommendation within ten business days of Schering-Plough’s request to do so; | |
| • | any tender offer or exchange offer by a third party with respect to Merck’s common stock constituting an acquisition proposal is commenced and within ten business days of commencement of the offer, Merck’s board of directors does not recommend that Merck’s shareholders reject such offer or issue a press release reaffirming its recommendation in favor of the merger agreement; | |
| • | Merck or its board of directors approves, endorses, recommends, adopts or enters into an acquisition proposal or a contract relating to an acquisition proposal; | |
| • | Merck materially breaches its non-solicitation covenants; | |
| • | Merck materially breaches its obligation to hold its shareholder meeting; or | |
| • | Merck or its board of directors announces, resolves or proposes to do any of the foregoing. |
| • | Either Merck or Schering-Plough terminates the merger agreement because: |
| • | the merger has not been consummated by the drop-dead date and an acquisition proposal was made for Merck prior to such termination, whether or not publicly announced, and within twelve months after such termination, Merck enters into a definitive agreement with respect to, or consummates, an acquisition proposal (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%); | |
| • | the Merck shareholders do not approve the Merck merger and an acquisition proposal for Merck was publicly announced prior to the Merck shareholders meeting and within twelve months after such termination, Merck enters into a definitive agreement with respect to or consummates an acquisition proposal (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%); |
| • | Schering-Plough terminates the merger agreement because Merck’s representations or warranties were or become inaccurate or Merck breaches a covenant such that the closing conditions relating to the accuracy of Merck’s representations and warranties or Merck’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date and an acquisition proposal was made for Merck prior to such termination, whether or not publicly announced, and within twelve months after such termination, Merck enters into a definitive agreement with respect to or consummates an acquisition proposal for 50% (where references in the definition of acquisition proposal to 15% are deemed to be references to 50%). |
118
Table of Contents
| • | Merck or Schering-Plough terminates the merger agreement because the Schering-Plough shareholders do not approve the merger and the issuance of common stock of New Merck in connection with the merger; | |
| • | Merck terminates the merger agreement because Schering-Plough’s representations or warranties were or become inaccurate or Schering-Plough breaches a covenant such that the closing conditions relating to the accuracy of Schering-Plough’s representations and warranties or Schering-Plough’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date; and | |
| • | Either Merck or Schering-Plough terminates the merger agreement because the drop-dead date shall have occurred and Merck’s closing conditions dealing with the accuracy of Schering-Plough’s representations and warranties, performance by Schering-Plough of its agreements and covenants or lack of a material adverse effect on Schering-Plough are not then capable of being satisfied. |
| • | Merck or Schering-Plough terminate the merger agreement because the Merck shareholders do not approve the merger agreement; | |
| • | Schering-Plough terminates the merger agreement because Merck’s representations or warranties become inaccurate or Merck breaches a covenant such that the closing conditions relating to the accuracy of Merck’s representations and warranties or Merck’s compliance with its covenants, as the case may be, cannot be then satisfied and the inaccuracy or breach is not capable of being cured or, if curable, is not cured by the drop-dead date; and | |
| • | Either Merck or Schering-Plough terminates the merger agreement because the drop-dead date shall have occurred and Schering-Plough’s closing conditions dealing with the accuracy of Merck’s representations and warranties, performance by Merck of its agreements and covenants or lack of a material adverse effect on Merck are not then capable of being satisfied. |
119
Table of Contents
| • | The American Heart Association. Founded in 1924, the American Heart Association (AHA) is a national voluntary health agency whose mission is to reduce disability and death from cardiovascular diseases and stroke. Since the AHA became a voluntary health organization in 1948, it has spent more that $2.7 billion dollars for projects that explore the prevention, detection and treatment of heart disease and stroke. The AHA’s goal is to provide credible heart disease and stroke information for effective prevention and treatment. | |
| • | The American Cancer Society. The American Cancer Society is a nationwide community-based voluntary health organization dedicated to eliminating cancer as a major health problem by preventing cancer, saving lives, and diminishing suffering from cancer through research, education, advocacy, and service. With more than two million volunteers nationwide, the American Cancer Society is one of the oldest and largest voluntary health agencies in the United States. | |
| • | Juvenile Diabetes Research Foundation International. The Juvenile Diabetes Research Foundation International (JDRF) is the leading charitable funder and advocate of type 1 (juvenile) diabetes research worldwide. Founded in 1970, the JDRF’s mission is to find a cure for diabetes, a disease which strikes children suddenly and requires multiple injections or infusions of insulin daily. Since inception, JDRF has awarded more than $1.16 billion to diabetes research, including more than $137 million in fiscal year 2007. | |
| • | Alzheimer’s Association. Alzheimer’s Association, National Office, was founded in 1980 and is the leading voluntary health organization in Alzheimer care, support and research. Its mission is to eliminate Alzheimer’s disease through the advancement of research; to provide and enhance care and support for all affected; and to reduce the risk of dementia through the promotion of brain health. | |
| • | The Conservation Fund. Since 1985, The Conservation Fund and its partners have protected more than 5 million acres of America’s outdoor heritage including wildlife habitat, working landscapes, rivers and wetlands and community open space. This conservation legacy ranges from national parks and wildlife refuges to private ranches, state forests and historic sites. The Conservation Fund has a dual-purpose mission to promote economic development and environmental protection. Using market-based approaches, the Fund works with the private sector to provide comprehensive real estate solutions and sustainable technologies. | |
| • | Feeding America. Feeding America, formerly America’s Second Harvest, feeds America’s hungry and engages in the fight to end hunger. It is the nation’s largest charitable hunger-relief organization. Feeding America supports approximately 50,000 local charitable agencies including food pantries, soup kitchens, emergency shelters, after-school programs, and Kids Cafes. Each year it provides food assistance to more than 25 million low-income hungry people in the United States. | |
| • | Teach for America. Teach For America is the national corps of recent college graduates and professionals who commit two years to teach in urban and rural public schools. Its mission is to build the movement to eliminate educational inequity. Since 1990, 17,000 individuals have participated in Teach for America, impacting the lives of more than 2.5 million students. Today, some 5,000 Teach For America corps members work in 26 urban and rural areas. |
120
Table of Contents
| • | a citizen or resident of the United States; | |
| • | a corporation, or other entity taxable as a corporation for U.S. federal income tax purposes, created or organized under the laws of the United States or any state thereof (or the District of Columbia); | |
| • | a trust if it (1) is subject to the primary supervision of a court within the United States and one or more U.S. persons have the authority to control all substantial decisions of the trust, or (2) has a valid election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person; or | |
| • | an estate that is subject to U.S. federal income tax on its income regardless of its source. |
121
Table of Contents
122
Table of Contents
| • | a U.S. holder will not recognize gain or loss upon receipt of shares of New Merck common stock solely in exchange for shares of Merck common stock in the Merck merger; | |
| • | a U.S. holder’s aggregate tax basis in the shares of New Merck common stock received in the Merck merger will be equal to the U.S. holder’s aggregate tax basis in the shares of Merck common stock surrendered; and | |
| • | a U.S. holder’s holding period of the shares of New Merck common stock received in the Merck merger will include the U.S. holder’s holding period of the shares of Merck common stock surrendered. |
123
Table of Contents
124
Table of Contents
| • | Substantially Disproportionate Test. The Schering-Plough merger generally will result in a “substantially disproportionate” redemption with respect to a U.S. holder of shares of Schering-Plough common stock if, among other things, the percentage of the outstanding shares of New Merck common stock actually and constructively owned by the U.S. holder immediately after the Schering-Plough merger and the Merck merger is less than 80% of the percentage of the shares of Schering-Plough common stock actually and constructively owned by the U.S. holder before the Schering-Plough merger and the Merck merger. | |
| • | Not Essentially Equivalent to a Dividend Test. The receipt of cash in the Schering-Plough merger will be treated as “not essentially equivalent to a dividend” if the reduction in a U.S. holder’s proportionate interest in New Merck as a result of the Schering-Plough merger and the Merck merger (when compared to the U.S. holder’s proportionate interest in Schering-Plough immediately prior to the Schering-Plough merger and the Merck merger) constitutes a “meaningful reduction” of the U.S. holder’s proportionate interest given the U.S. holder’s particular facts and circumstances. The IRS has indicated in a published revenue ruling that even a small reduction in the percentage interest of a shareholder whose relative stock interest in a publicly held corporation is minimal and who exercises no control over corporate affairs should constitute a “meaningful reduction.” | |
| • | Complete Termination Test. A U.S. holder of shares of Schering-Plough common stock may be able to satisfy the “complete termination” test if such holder sells or otherwise disposes of all of such holder’s shares of Schering-Plough common stock contemporaneously with the completion of theSchering-Plough merger and as part of a single integrated plan which includes participation by such holder in the Schering-Plough merger, as discussed below. However, there is some uncertainty as to whether the “complete termination” test applies in such circumstances. U.S. holders should consult their own tax advisors as to this matter in light of their particular circumstances and the applicable law. |
125
Table of Contents
126
Table of Contents
127
Table of Contents
128
Table of Contents
Name, Age | Position | |
| Richard T. Clark, 63 | Chairman, President and Chief Executive Officer | |
| Adele D. Ambrose, 52 | Vice President and Chief Communications Officer | |
| John Canan, 52 | Senior Vice President and Controller | |
| Celia A. Colbert, 52 | Senior Vice President, Secretary and Assistant General Counsel | |
| Willie A. Deese, 53 | Executive Vice President and President, Merck Manufacturing Division | |
| Kenneth C. Frazier, 54 | Executive Vice President, Global Human Health | |
| Mirian M. Graddick-Weir, 54 | Executive Vice President, Human Resources | |
| Peter N. Kellogg, 53 | Executive Vice President and Chief Financial Officer | |
| Peter S. Kim, 51 | Executive Vice President and President, Merck Research Laboratories | |
| Bruce N. Kuhlik, 52 | Executive Vice President and General Counsel | |
| Mark E. McDonough, 44 | Vice President and Treasurer | |
| Margaret G. McGlynn, 49 | President, Merck Vaccines and Infectious Diseases | |
| Stefan Oschmann, 51 | President, Europe, Middle East, Africa and Canada | |
| J. Chris Scalet, 50 | Executive Vice President, Global Services and Chief Information Officer | |
| Adam H. Schechter, 44 | President, Global Pharmaceuticals |
129
Table of Contents
130
Table of Contents
| • | The merger with Schering-Plough for aggregate consideration of approximately $44.6 billion; | |
| • | The issuance of common shares and debt, as well as the use of existing cash and the sale or redemption of short-term investments to fund the merger; and | |
| • | The consolidation of the Merck/Schering-Plough cholesterol partnership joint venture which will be owned 100% by New Merck upon consummation of the transaction. |
131
Table of Contents
132
Table of Contents
March 31, 2009
| Merck/Schering- | ||||||||||||||||||||||||||||
| Plough Cholesterol | ||||||||||||||||||||||||||||
| Historical | Partnership | Pro Forma Adjustments (Note 3) | ||||||||||||||||||||||||||
| Schering- | Adjustments | Purchase | Pro Forma | |||||||||||||||||||||||||
| Merck | Plough | (Note 2) | Reclassifications | Financing | Accounting | As Adjusted | ||||||||||||||||||||||
| (In millions) | ||||||||||||||||||||||||||||
ASSETS | ||||||||||||||||||||||||||||
| Current Assets | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 6,017.1 | $ | 2,845.0 | $ | 225.0 | $ | — | $ | 8,500.0 | (a) | $ | (11,846.9 | )(b) | $ | 5,440.2 | ||||||||||||
| (100.0 | )(c) | |||||||||||||||||||||||||||
| (200.0 | )(d) | |||||||||||||||||||||||||||
| Short-term investments | 6,564.5 | 708.0 | — | — | — | (6,564.5 | )(b) | 708.0 | ||||||||||||||||||||
| Accounts receivable | 3,569.7 | 2,953.0 | 99.3 | — | — | — | 6,622.0 | |||||||||||||||||||||
| Inventories | 2,127.8 | 3,158.0 | 84.0 | — | — | 1,873.0 | (e) | 7,242.8 | ||||||||||||||||||||
| Deferred income taxes and other current assets | 7,905.5 | 1,680.0 | (99.0 | ) | — | — | (711.7 | )(f) | 8,870.2 | |||||||||||||||||||
| 95.4 | (d) | |||||||||||||||||||||||||||
| Total current assets | 26,184.6 | 11,344.0 | 309.3 | — | 8,500.0 | (17,454.7 | ) | 28,883.2 | ||||||||||||||||||||
| Investments | 78.7 | — | — | — | — | — | 78.7 | |||||||||||||||||||||
| Property, Plant and Equipment, at cost, net | 11,826.1 | 6,662.0 | — | 222.0 | (g) | — | — | (h) | 18,710.1 | |||||||||||||||||||
| Goodwill | 1,439.0 | 2,667.0 | — | — | — | 15,551.3 | (i) | 16,990.3 | ||||||||||||||||||||
| (2,667.0 | )(i) | |||||||||||||||||||||||||||
| Other Intangibles, Net | 614.2 | 5,761.0 | — | — | — | 36,693.0 | (j) | 44,861.2 | ||||||||||||||||||||
| 7,554.0 | (j)(r) | |||||||||||||||||||||||||||
| (5,761.0 | )(j) | |||||||||||||||||||||||||||
| Other Assets | 6,400.5 | 1,284.0 | (261.0 | ) | (222.0 | )(g) | — | — | 7,201.5 | |||||||||||||||||||
Total Assets | $ | 46,543.1 | $ | 27,718.0 | $ | 48.3 | $ | — | $ | 8,500.0 | $ | 33,915.6 | $ | 116,725.0 | ||||||||||||||
LIABILITIES AND EQUITY | ||||||||||||||||||||||||||||
| Current Liabilities | ||||||||||||||||||||||||||||
| Loans payable and current portion of long-term debt | $ | 2,798.9 | $ | 204.0 | $ | — | $ | — | $ | 3,500.0 | (a) | $ | — | $ | 6,502.9 | |||||||||||||
| Trade accounts payable | 622.5 | 1,670.0 | (3.3 | ) | — | — | — | 2,289.2 | ||||||||||||||||||||
| Accrued and other current liabilities | 8,710.7 | 2,925.0 | 70.0 | (125.0 | )(g) | — | 100.0 | (k) | 11,741.2 | |||||||||||||||||||
| 165.0 | (l) | |||||||||||||||||||||||||||
| (104.5 | )(d) | |||||||||||||||||||||||||||
| Income taxes payable | 480.2 | 162.0 | — | — | — | — | 642.2 | |||||||||||||||||||||
| Dividends payable | 803.6 | — | — | 125.0 | (g) | — | — | 928.6 | ||||||||||||||||||||
| Total current liabilities | 13,415.9 | 4,961.0 | 66.7 | — | 3,500.0 | 160.5 | 22,104.1 | |||||||||||||||||||||
| Long-Term Debt | 3,939.1 | 7,685.0 | — | — | 5,000.0 | (a) | 254.0 | (m) | 16,878.1 | |||||||||||||||||||
| Deferred Income Taxes and Noncurrent Liabilities | 7,186.6 | 4,237.0 | — | — | — | 10,904.1 | (f) | 22,228.7 | ||||||||||||||||||||
| (99.0 | )(n) | |||||||||||||||||||||||||||
| Equity | ||||||||||||||||||||||||||||
| Mandatory convertible preferred shares | — | 2,500.0 | — | — | — | (2,500.0 | )(o) | — | ||||||||||||||||||||
| Common shares | 29.8 | 1,060.0 | — | — | — | 494.0 | (b) | 1,548.4 | ||||||||||||||||||||
| (8.7 | )(s) | |||||||||||||||||||||||||||
| 1,033.3 | (p) | |||||||||||||||||||||||||||
| (1,060.0 | )(o) | |||||||||||||||||||||||||||
| Other paid-in capital | 8,386.8 | 5,128.0 | — | — | — | 25,263.2 | (b) | 33,014.5 | ||||||||||||||||||||
| 397.8 | (q) | |||||||||||||||||||||||||||
| (1,033.3 | )(p) | |||||||||||||||||||||||||||
| (5,128.0 | )(o) | |||||||||||||||||||||||||||
| Retained earnings | 44,320.4 | 9,842.0 | (18.4 | ) | — | — | 7,576.0 | (r) | 20,985.5 | |||||||||||||||||||
| (100.0 | )(c) | |||||||||||||||||||||||||||
| (100.0 | )(k) | |||||||||||||||||||||||||||
| (30,692.5 | )(s) | |||||||||||||||||||||||||||
| (9,842.0 | )(o) | |||||||||||||||||||||||||||
| Accumulated other comprehensive loss | (2,472.8 | ) | (2,352.0 | ) | — | — | — | 2,352.0 | (o) | (2,472.8 | ) | |||||||||||||||||
| Treasury stock, at cost | (30,701.2 | ) | (5,343.0 | ) | — | — | — | 30,701.2 | (s) | — | ||||||||||||||||||
| 5,343.0 | (o) | |||||||||||||||||||||||||||
| Total stockholders’ equity | 19,563.0 | 10,835.0 | (18.4 | ) | — | — | 22,696.0 | 53,075.6 | ||||||||||||||||||||
| Noncontrolling interests | 2,438.5 | — | — | — | — | — | 2,438.5 | |||||||||||||||||||||
| Total equity | 22,001.5 | 10,835.0 | (18.4 | ) | — | — | 22,696.0 | 55,514.1 | ||||||||||||||||||||
Total Liabilities and Stockholders’ Equity | $ | 46,543.1 | $ | 27,718.0 | $ | 48.3 | $ | — | $ | 8,500.0 | $ | 33,915.6 | $ | 116,725.0 | ||||||||||||||
133
Table of Contents
Three Months Ended March 31, 2009
| Merck/Schering- | ||||||||||||||||||||||||||||
| Historical | Plough Cholesterol | Pro Forma Adjustments (Note 3) | ||||||||||||||||||||||||||
| Schering- | Partnership | Purchase | Pro Forma | |||||||||||||||||||||||||
| Merck | Plough | Adjustments (Note 2) | Reclassifications | Financing | Accounting | As Adjusted | ||||||||||||||||||||||
| (In millions except per share amounts) | ||||||||||||||||||||||||||||
| Sales | $ | 5,385.2 | $ | 4,393.0 | $ | 907.4 | $ | — | $ | — | $ | — | $ | 10,685.6 | ||||||||||||||
| Costs, Expenses and Other | ||||||||||||||||||||||||||||
| Materials and production | 1,333.8 | 1,399.0 | 8.6 | — | — | 972.1 | (t1) | 3,584.5 | ||||||||||||||||||||
| (129.0 | )(t2) | |||||||||||||||||||||||||||
| Marketing and administrative | 1,632.9 | 1,493.0 | 162.0 | — | — | (7.0 | )(v) | 3,280.9 | ||||||||||||||||||||
| Research and development | 1,224.2 | 804.0 | 43.0 | — | — | — | 2,071.2 | |||||||||||||||||||||
| Restructuring costs | 64.3 | 75.0 | — | — | — | (19.0 | )(v) | 120.3 | ||||||||||||||||||||
| Equity income from affiliates | (585.8 | ) | (400.0 | ) | 690.9 | — | — | — | (294.9 | ) | ||||||||||||||||||
| Other (income) expense, net | (67.2 | ) | 88.0 | — | — | 69.6 | (w2) | 70.3 | (w3) | 141.7 | ||||||||||||||||||
| (12.0 | )(w5) | (7.0 | )(w4) | |||||||||||||||||||||||||
| 3,602.2 | 3,459.0 | 904.5 | — | 57.6 | 880.4 | 8,903.7 | ||||||||||||||||||||||
| Income Before Taxes | 1,783.0 | 934.0 | 2.9 | — | (57.6 | ) | (880.4 | ) | 1,781.9 | |||||||||||||||||||
| Taxes on Income | 327.2 | 129.0 | — | — | (21.9 | )(x) | (189.2 | )(x) | 245.1 | |||||||||||||||||||
| Net Income | 1,455.8 | 805.0 | 2.9 | — | (35.7 | ) | (691.2 | ) | 1,536.8 | |||||||||||||||||||
| Less: Net Income Attributable to Noncontrolling Interests | 30.8 | — | — | — | — | — | 30.8 | |||||||||||||||||||||
| Net Income Attributable to Controlling Interests | 1,425.0 | 805.0 | 2.9 | — | (35.7 | ) | (691.2 | ) | 1,506.0 | |||||||||||||||||||
| Preferred Stock Dividends | — | 38.0 | — | — | — | (38.0 | )(y) | — | ||||||||||||||||||||
| Net Income Available to Common Shareholders | $ | 1,425.0 | $ | 767.0 | $ | 2.9 | $ | — | $ | (35.7 | ) | $ | (653.2 | ) | $ | 1,506.0 | ||||||||||||
| Basic Earnings per Common Share | $ | 0.67 | $ | 0.47 | $ | 0.48 | ||||||||||||||||||||||
| Earnings per Common Share Assuming Dilution | $ | 0.67 | $ | 0.46 | $ | 0.48 | ||||||||||||||||||||||
| Weighted average common shares used to calculate earnings per share: | ||||||||||||||||||||||||||||
| Basic | 2,107.9 | 1,627.0 | 3,095.9 | (z) | ||||||||||||||||||||||||
| Diluted | 2,109.2 | 1,720.0 | 3,098.4 | (z) | ||||||||||||||||||||||||
134
Table of Contents
| Merck/Schering- | ||||||||||||||||||||||||||||
| Historical | Plough Cholesterol | Pro Forma Adjustments (Note 3) | ||||||||||||||||||||||||||
| Schering- | Partnership | Purchase | Pro Forma | |||||||||||||||||||||||||
| Merck | Plough | Adjustments (Note 2) | Reclassifications | Financing | Accounting | As Adjusted | ||||||||||||||||||||||
| (In millions except per share amounts) | ||||||||||||||||||||||||||||
| Sales | $ | 23,850.3 | $ | 18,502.0 | $ | 4,397.3 | $ | — | $ | — | $ | — | $ | 46,749.6 | ||||||||||||||
| Costs, Expenses and Other | ||||||||||||||||||||||||||||
| Materials and production | 5,582.5 | 7,307.0 | 38.0 | — | — | 3,888.4 | (t1) | 16,245.9 | ||||||||||||||||||||
| (570.0 | )(t2) | |||||||||||||||||||||||||||
| Marketing and administrative | 7,377.0 | 6,823.0 | 839.0 | 54.0 | (u) | — | — | 15,093.0 | ||||||||||||||||||||
| Research and development | 4,805.3 | 3,529.0 | 168.0 | — | — | — | 8,502.3 | |||||||||||||||||||||
| Restructuring costs | 1,032.5 | 329.0 | — | (54.0 | )(u) | — | — | 1,307.5 | ||||||||||||||||||||
| Equity income from affiliates | (2,560.6 | ) | (1,870.0 | ) | 3,406.3 | — | — | — | (1,024.3 | ) | ||||||||||||||||||
| Other (income) expense, net | (2,318.1 | ) | 335.0 | — | — | 200.0 | (w1) | 281.4 | (w3) | (1,251.5 | ) | |||||||||||||||||
| 278.5 | (w2) | (28.3 | )(w4) | |||||||||||||||||||||||||
| 13,918.6 | 16,453.0 | 4,451.3 | — | 478.5 | 3,571.5 | 38,872.9 | ||||||||||||||||||||||
| Income Before Taxes | 9,931.7 | 2,049.0 | (54.0 | ) | — | (478.5 | ) | (3,571.5 | ) | 7,876.7 | ||||||||||||||||||
| Taxes on Income | 1,999.4 | 146.0 | — | — | (181.8 | )(x) | (775.9 | )(x) | 1,187.7 | |||||||||||||||||||
| Net Income | 7,932.3 | 1,903.0 | (54.0 | ) | — | (296.7 | ) | (2,795.6 | ) | 6,689.0 | ||||||||||||||||||
| Less: Net Income Attributable to Noncontrolling Interests | 123.9 | — | — | — | — | — | 123.9 | |||||||||||||||||||||
| Net Income Attributable to Controlling Interests | 7,808.4 | 1,903.0 | (54.0 | ) | — | (296.7 | ) | (2,795.6 | ) | 6,565.1 | ||||||||||||||||||
| Preferred Stock Dividends | — | 150.0 | — | — | — | (150.0 | )(y) | — | ||||||||||||||||||||
| Net Income Available to Common Shareholders | $ | 7,808.4 | $ | 1,753.0 | $ | (54.0 | ) | $ | — | $ | (296.7 | ) | $ | (2,645.6 | ) | $ | 6,565.1 | |||||||||||
| Basic Earnings per Common Share | $ | 3.65 | $ | 1.08 | $ | 2.09 | ||||||||||||||||||||||
| Earnings per Common Share Assuming Dilution | $ | 3.63 | $ | 1.07 | $ | 2.09 | ||||||||||||||||||||||
| Weighted average common shares used to calculate earnings per share: | ||||||||||||||||||||||||||||
| Basic | 2,135.8 | 1,625.0 | 3,123.8 | (z) | ||||||||||||||||||||||||
| Diluted | 2,142.5 | 1,635.0 | 3,132.5 | (z) | ||||||||||||||||||||||||
135
Table of Contents
| (1) | Calculation of Estimated Consideration Transferred and Preliminary Allocation of Consideration Transferred to Net Assets Acquired |
| (In millions | ||||
| except per share amounts) | ||||
| Schering-Plough common stock shares outstanding at March 31, 2009 (net of treasury shares) | 1,628.0 | |||
| Conversion of Schering-Plough 6% Mandatory Convertible Preferred Stock | 85.5 | (a) | ||
| Shares eligible for conversion | 1,713.5 | |||
| Cash per share | $ | 10.50 | ||
| Cash consideration for outstanding shares | $ | 17,991.8 | ||
| Schering-Plough 6% Mandatory Convertible Preferred Stock make-whole dividend payments | $ | 144.5 | (a) | |
| Value of Schering-Plough deferred stock units to be settled in cash | 275.1 | (b) | ||
| Total cash consideration | $ | 18,411.4 | ||
| Shares eligible for conversion | 1,713.5 | |||
| Common stock exchange ratio per share | 0.5767 | |||
| Equivalent new company shares (par value $0.50) | 988 | |||
| Merck common stock share price on June 5, 2009 | $ | 26.07 | (c) | |
| Common stock equity consideration | $ | 25,757.2 | ||
| Fair value of share-based compensation awards | $ | 397.8 | (b) | |
| Total estimated consideration transferred | $ | 44,566.4 | ||
| (a) | For purposes of preparing these unaudited pro forma condensed combined financial statements, Merck has assumed conversion of all outstanding Schering-Plough 6% Mandatory Convertible Preferred Stock in connection with the merger at the make-whole conversion rate as Merck believes this would be most advantageous to those holders. Holders converting their preferred stock will be entitled to receive a make-whole dividend payment equal to the present value of the dividends that would otherwise be payable in respect of those shares during the period from the conversion date through the mandatory conversion date on August 13, 2010 using a discount rate of 6.75%. Each share of preferred stock not so converted would result in a reduction of 8.6 shares of common stock issued and a per share reduction of $14.45 in the make-whole dividend payment. This conversion factor assumes the transaction closes in the fourth quarter of 2009. | |
| (b) | Represents the fair value of Schering-Plough stock option, performance-based deferred stock unit and certain deferred stock unit replacement awards and the cash consideration to be paid to holders of certain other deferred stock units for precombination services. FAS 141(R) requires that the fair value of replacement awards and cash payments made to settle vested awards attributable to precombination service be included in the consideration transferred. Holders of Schering-Plough stock options and performance-based deferred stock units will receive replacement awards. Holders of Schering-Plough deferred stock units issued after 2007 will receive replacement awards and holders of deferred stock units for 2007 awards and prior will be converted into the right to receive cash as specified in the merger agreement. The fair value of outstanding Schering-Plough stock options, deferred stock units and performance-based deferred stock units for 2007 awards and prior, which will immediately vest at the effective time of the merger, has been attributed to precombination service and included in the consideration transferred. Awards for 2008 and 2009 will not immediately vest upon completion of the merger. For these awards, the fair value of the |
136
Table of Contents
| awards attributed to precombination services is included as part of the consideration transferred and the fair value attributed to postcombination services will be recorded as compensation cost in the postcombination financial statements of the combined entity. | ||
| (c) | In accordance with FAS 141(R), the fair value of equity securities issued as part of the consideration transferred will be measured using the market price of Merck common stock on the closing date. Assuming a $1 change in Merck’s closing common stock price, the estimated consideration transferred would increase or decrease by approximately $1 billion which would have a corresponding offset to estimated goodwill. |
| Identifiable intangible assets | $ | 36,693.0 | ||
| Property, plant and equipment | 6,662.0 | |||
| Inventories | 5,009.0 | |||
| Other non-current assets | 1,284.0 | |||
| Net working capital, excluding inventories and deferred taxes | 2,575.0 | |||
| Deferred income taxes, net | (12,572.9 | ) | ||
| Long-term debt | (7,939.0 | ) | ||
| Other long-term liabilities | (2,696.0 | ) | ||
| Goodwill | 15,551.3 | |||
| Estimated purchase price to be allocated | $ | 44,566.4 | ||
| (2) | Merck/Schering-Plough Cholesterol Partnership Adjustments |
137
Table of Contents
| (3) | Pro Forma Adjustments |
138
Table of Contents
139
Table of Contents
| Weighted | ||||||||
| Average | ||||||||
| Estimated | Estimated | |||||||
| Fair Value | Useful Life | |||||||
| Products and product rights | $ | 35,730 | 9 years | |||||
| Tradenames | 705 | 14 years | ||||||
| Tradenames — indefinite lived | 420 | — | ||||||
| In-process research and development | 7,392 | Unknown* | ||||||
| $ | 44,247 | |||||||
| * | These amounts will be capitalized and accounted for as indefinite-lived intangible assets, subject to impairment testing until completion or abandonment of the projects. Upon successful completion of each project, Merck will make a separate determination as to the useful life of the assets and begin amortization. |
140
Table of Contents
141
Table of Contents
142
Table of Contents
| • | 20,000,000 shares of preferred stock, par value $1.00 per share, issuable in one or more series; and | |
| • | 6,500,000,000 shares of common stock, par value $0.50 per share. |
| • | adoption of a proposed amendment to the certificate of incorporation; | |
| • | approval of a proposed plan of merger or consolidation; | |
| • | approval of a sale, lease, exchange or other disposition of all, or substantially all, the assets of New Merck, not in the usual and regular course of business; | |
| • | approval of a proposed plan of exchange; and | |
| • | approval of a proposed plan of dissolution. |
143
Table of Contents
144
Table of Contents
| • | Dividends. 6% annual cumulative dividends on the liquidation preference of $250 per share of New Merck 6% preferred stock will be payable quarterly in cash of $3.75 on February 15, May 15, August 15 and November 15 each year until the final dividend payment of $3.67 on August 13, 2010 (the mandatory conversion date). | |
| • | Redemption. The New Merck 6% preferred stock is not redeemable. | |
| • | Mandatory Conversion. On the mandatory conversion date, each share of New Merck 6% preferred stock will automatically convert into a number of units of the merger consideration equal to the conversion rate described below and all accrued, cumulated and unpaid dividends that have not been declared for all dividend periods up to and excluding the mandatory conversion date will be due and payable at such time. | |
| • | Merger Consideration Unit. Each unit of merger consideration will consist of $10.50 in cash and 0.5767 of a share of New Merck common stock. | |
| • | Conversion Rate. The conversion rate for each share of New Merck 6% preferred stock applicable on the mandatory conversion date will be, if the applicable market value of a unit of merger consideration is equal to or greater than $33.69, 7.4206 units of merger consideration and, if the applicable market value of a unit of merger consideration is less than or equal to $27.50, 9.0909 units. If the applicable market value of a unit of merger consideration is greater than $27.50 and less than $33.69, the conversion rate will be determined by dividing $250 by the applicable market value of a unit of merger consideration. These conversion rates will be subject to an anti-dilution adjustment in the event of, among other things, stock dividends or distributions in common stock by New Merck or subdivisions, splits and combinations of the common stock of New Merck. The conversion rates will also be subject to adjustment in the event of any quarterly dividend above a dividend threshold amount per share of New Merck common stock, certain self tender offers by New Merck or third-party tender offers for shares of New Merck common stock. In connection with a mandatory conversion, the applicable market value of a unit of merger consideration shall be equal to (i) $10.50 plus (ii) the average of the closing |
145
Table of Contents
| prices of the shares of New Merck common stock over the twenty consecutive trading days ending on the third trading date before the mandatory conversion date, multiplied by 0.5767. |
| • | Optional Conversion. At any time prior to the mandatory conversion date, a holder of New Merck 6% preferred stock may elect to convert each share at the minimum conversion rate of 7.4206 units of merger consideration for each share of New Merck 6% preferred stock subject to anti-dilution adjustments, and also receive all accrued, cumulated and unpaid dividends that have not been declared for all prior dividend periods ending on or prior to the dividend payment date immediately preceding the early conversion date. | |
| • | Provisional Conversion at New Merck’s Option. If, at any time prior to the mandatory conversion date, the market value of one unit of merger consideration exceeds $50.53, subject to anti-dilution adjustments, for at least twenty trading days within a period of thirty trading days, New Merck may elect to cause the conversion of all, but not less than all, of the New Merck 6% preferred stock then outstanding at the minimum conversion rate of 7.4206 units of merger consideration for each share of New Merck 6% preferred stock subject to anti-dilution adjustments, only if New Merck is then legally permitted to pay in cash an amount equal to the sum of: |
| • | all accrued, cumulated and unpaid dividends on the New Merck 6% preferred stock that have not been declared for all dividend periods up to, but excluding, the conversion date; plus | |
| • | if the conversion date is prior to the record date for any declared dividend, all accrued, cumulated and unpaid dividends on the New Merck 6% preferred stock that have been declared for all dividend periods to, but excluding, the conversion date; plus | |
| • | the present value of all remaining future dividend payments on the New Merck 6% preferred stock through and including the mandatory conversion date. |
| • | Early Conversion and Dividend Make-Whole Payment Upon Certain Acquisitions. If prior to the earlier of the mandatory conversion date and New Merck providing notice of a provisional conversion at its option, New Merck is the subject of any acquisition in which 90% or more of New Merck’s common shares are exchanged for, converted into or constitute solely the right to receive cash, securities or other property, and more than 10% of such cash, securities or property is cash or securities or other property that is not, or will not be upon issuance, shares of common equity traded on the NYSE, Nasdaq Global Select Market or the Nasdaq Global Market (a make-whole transaction), then New Merck will: |
| • | permit conversion of the New Merck 6% preferred stock, for a period beginning on the effective date of the make-whole transaction and ending fifteen days after, at a specified make-whole conversion rate determined by reference to the market value of a unit of merger consideration ($10.50 plus the price of 0.5767 of a common share of New Merck) and the consideration to be paid in the make-whole transaction; | |
| • | pay, upon conversion, in cash an amount equal to the sum of: |
| • | all accrued, cumulated and unpaid dividends on the New Merck 6% preferred stock that have not been declared for all dividend periods up to, but excluding, the conversion date; plus | |
| • | if the conversion date is prior to the record date for any declared dividend, all accrued, cumulated and unpaid dividends on the New Merck 6% preferred stock that have been declared for all dividend periods to, but excluding, the conversion date; plus | |
| • | the present value of all remaining future dividend payments on the New Merck 6% preferred stock through and including the mandatory conversion date. |
| • | Reorganization Events. In the event of a consolidation or merger of New Merck with or into another person, a sale, transfer, lease or conveyance of all or substantially all the property and assets of New |
146
Table of Contents
| Merck, certain reclassifications of common shares or statutory exchanges of securities of New Merck (each a reorganization event), then: |
| • | each share of New Merck 6% preferred stock outstanding immediately prior to the reorganization event and convertible into units of merger consideration will, with respect to the non-cash portion of a unit of merger consideration (i.e., 0.5767 of a share of New Merck common stock), become convertible into the kind of securities, cash and other property receivable in the reorganization event by a holder of a share of common stock of New Merck. |
| • | Liquidation Preference. $250 per share of New Merck 6% preferred stock, plus an amount equal to the sum of all accrued, cumulated and unpaid dividends. | |
| • | Voting Rights. Holders of New Merck 6% preferred stock will not be entitled to any voting rights, except as required by New Jersey law and as described below. |
| • | New Merck may not issue any class of capital stock, the terms of which provide that such class will rank senior to the New Merck 6% preferred stock as to payment of dividends or distribution of assets upon dissolution or otherwise amend its certificate if incorporation in a manner that would adversely affect the rights of the New Merck 6% preferred stock without the approval of the holders of at least two-thirds of the New Merck 6% preferred stock voting together as a single class. |
| • | Ranking. The New Merck 6% preferred stock will rank as to payment of dividends and distributions of assets upon dissolution, liquidation or winding up: |
| • | junior to all existing and future debt obligations of New Merck and its subsidiaries; | |
| • | junior to any class or series of capital stock of New Merck, the terms of which provide that such class or series will rank senior to the New Merck 6% preferred stock; | |
| • | senior to the common shares and any other class or series of capital stock of New Merck, the terms of which provide that such class or series will rank junior to the New Merck 6% preferred stock; and | |
| • | on a parity with any other class or series of New Merck capital stock. |
147
Table of Contents
148
Table of Contents
| • | with respect to the annual meeting, 120 days prior to the anniversary of the preceding year’s annual meeting; and | |
| • | with respect to a special meeting, the close of business on the seventh day following the date on which notice of such meeting is first given to shareholders. |
| • | the name and address of the shareholder making the nomination and the person to be nominated; | |
| • | a representation that the shareholder is a holder of record entitled to vote at such meeting and intends to appear in person or by proxy at the meeting to nominate the person or persons specified in the notice; | |
| • | a description of all arrangements or understandings between the shareholder and each nominee; | |
| • | such other information regarding each nominee as would have been required under the rules of the SEC to be included in a proxy statement had each nominee been nominated, or intended to be nominated, by the board of directors; and | |
| • | the consent of each nominee to serve as a director of Merck or New Merck, as applicable, if so elected. |
| • | the name, age, business address, and residence address of each nominee proposed; |
149
Table of Contents
| • | the principal occupation or employment of each nominee; | |
| • | the number of shares of capital stock of Schering-Plough beneficially owned by each nominee; | |
| • | such other information regarding each nominee as would have been required under the rules of the SEC to be included in a proxy statement; and | |
| • | a signed consent to serve as a director if so elected. |
150
Table of Contents
151
Table of Contents
| • | the name and address of the shareholder presenting the business at the meeting, a brief description of such business to be presented, the reasons for conducting such business and any material interest of the shareholder in such business; | |
| • | a representation that the shareholder is a holder of record of stock of the company entitled to vote on such business and intends to appear in person or by proxy at the meeting to present the business specified in the notice; | |
| • | a description of all arrangements or understandings between the shareholder and any other person with respect to the business presented; and | |
| • | such other information regarding the business to be presented as would be required to be included in a proxy statement filed pursuant to the proxy rules of the SEC had such business been presented, or intended to be presented, by the board of directors. |
| • | a brief description of the business desired to be brought before the meeting, the reasons for conducting such business and any material interest of the shareholder in such business; | |
| • | the name and address of such shareholder as they appear on Schering-Plough’s books and the beneficial owner, if any, on whose behalf such proposal is made; and |
152
Table of Contents
| • | the class and number of shares of Schering-Plough which are owned beneficially and of record by the shareholder or the beneficial owner. |
153
Table of Contents
| • | constitute a breach of the duty of loyalty to the corporation or its shareholders; | |
| • | are not in good faith or involving a knowing violation of law; or | |
| • | result in receipt by such person of an improper personal benefit. |
154
Table of Contents
| • | the effects of the action on the corporation’s employees, suppliers and customers; | |
| • | the effects of the action on the community in which the corporation operates; and | |
| • | the long-term as well as short-term interests of the corporation and its shareholders, including the possibility that these interests may best be served by the continued independence of the corporation. |
155
Table of Contents
156
Table of Contents
Merck SEC Filings | Period | |
Annual Report onForm 10-K | Year ended December 31, 2008 filed February 27, 2009 | |
Quarterly Report onForm 10-Q | Period ended March 31, 2009 filed May 4, 2009 | |
Current Reports onForm 8-K | Filed February 3, 2009, February 11, 2009, February 24, 2009, March 2, 2009, March 9, 2009, March 10, 2009, April 21, 2009, May 4, 2009(Form 8-K/A), May 12, 2009 May 20, 2009 and June 22, 2009 |
157
Table of Contents
Schering-Plough SEC Filings | Period | |
Annual Report onForm 10-K | Year ended December 31, 2008 filed February 27, 2009 | |
Quarterly Report onForm 10-Q | Period ended March 31, 2009 filed May 1, 2009 | |
Current Reports onForm 8-K | Filed January 20, 2009, February 3, 2009, March 9, 2009, March 11, 2009 and April 21, 2009 |
Merck & Co., Inc. | Schering-Plough Corporation | |
| P.O. Box 100 — WS 3AB-40 Whitehouse Station, NJ08889-0100 (908) 423-7845 Attention: Stockholder Services Dept. | 2000 Galloping Hill Road Kenilworth, NJ 07033-0530 (908) 298-7436 Attention: Investor Relations |
158
Table of Contents
Annex A: | Agreement and Plan of Merger | |
Annex B: | Opinion of J.P. Morgan Securities Inc. | |
Annex C: | Opinion of Goldman, Sachs & Co. | |
Annex D: | Opinion of Morgan Stanley & Co. Incorporated |
159
Table of Contents
by and among
MERCK & CO., INC.,
SCHERING-PLOUGH CORPORATION,
BLUE, INC.,
and
PURPLE, INC.
March 8, 2009
Table of Contents
| Page | ||||||||
ARTICLE I THE MERGERS | ||||||||
| 1.1 | The Mergers | A-1 | ||||||
| 1.2 | Closing | A-2 | ||||||
| 1.3 | Certificates of Incorporation and Bylaws | A-2 | ||||||
| 1.4 | Directors and Officers | A-3 | ||||||
ARTICLE II EFFECT ON THE CAPITAL STOCK OF THE CONSTITUENT CORPORATIONS | ||||||||
| 2.1 | Conversion of Securities | A-3 | ||||||
| 2.2 | Exchange of Certificates | A-5 | ||||||
| 2.3 | Stock Transfer Books | A-8 | ||||||
| 2.4 | Saturn Options and Other Equity Awards | A-8 | ||||||
| 2.5 | Mercury Options and Other Equity Awards | A-9 | ||||||
| 2.6 | Reservation of Shares; Registration | A-9 | ||||||
ARTICLE III REPRESENTATIONS AND WARRANTIES OF SATURN AND MERGER SUB 1 AND MERGER SUB 2 | ||||||||
| 3.1 | Organization and Qualification | A-10 | ||||||
| 3.2 | Significant Subsidiaries | A-10 | ||||||
| 3.3 | Authorization; Validity of Agreement; Necessary Action | A-10 | ||||||
| 3.4 | Governmental Filings; No Violations; Consents and Waivers | A-11 | ||||||
| 3.5 | Capital Stock | A-12 | ||||||
| 3.6 | Saturn SEC Reports | A-13 | ||||||
| 3.7 | Absence of Certain Changes or Events | A-15 | ||||||
| 3.8 | Material Contracts | A-15 | ||||||
| 3.9 | Intellectual Property | A-16 | ||||||
| 3.10 | Litigation | A-16 | ||||||
| 3.11 | Permits; Compliance with Laws | A-16 | ||||||
| 3.12 | Regulatory Compliance | A-17 | ||||||
| 3.13 | Saturn Employee Benefit Plans | A-17 | ||||||
| 3.14 | Labor and Employment Matters | A-19 | ||||||
| 3.15 | Taxes | A-20 | ||||||
| 3.16 | Tax Matters | A-20 | ||||||
| 3.17 | Insurance | A-20 | ||||||
| 3.18 | Environmental Liability | A-20 | ||||||
| 3.19 | Affiliated Transactions | A-21 | ||||||
| 3.20 | Brokerage | A-21 | ||||||
| 3.21 | Opinion of Saturn’s Financial Advisor | A-21 | ||||||
| 3.22 | Interested Stockholder | A-21 | ||||||
| 3.23 | Ownership and Operations of Merger Sub 1 and Merger Sub 2 | A-21 | ||||||
| 3.24 | Rights Agreement; Takeover Statutes | A-22 | ||||||
| 3.25 | Intercompany Notes | A-22 | ||||||
| 3.26 | No Additional Representations | A-22 | ||||||
Table of Contents
| Page | ||||||||
ARTICLE IV REPRESENTATIONS AND WARRANTIES OF MERCURY | ||||||||
| 4.1 | Organization and Qualification | A-23 | ||||||
| 4.2 | Significant Subsidiaries | A-23 | ||||||
| 4.3 | Authorization; Validity of Agreement; Necessary Action | A-23 | ||||||
| 4.4 | Governmental Filings; No Violations; Consents and Waivers | A-24 | ||||||
| 4.5 | Capital Stock | A-24 | ||||||
| 4.6 | Mercury SEC Reports | A-25 | ||||||
| 4.7 | Absence of Certain Changes or Events | A-27 | ||||||
| 4.8 | Material Contracts | A-27 | ||||||
| 4.9 | Intellectual Property | A-27 | ||||||
| 4.10 | Litigation | A-28 | ||||||
| 4.11 | Permits; Compliance with Laws | A-28 | ||||||
| 4.12 | Regulatory Compliance | A-28 | ||||||
| 4.13 | Mercury Employee Benefit Plans | A-29 | ||||||
| 4.14 | Taxes | A-29 | ||||||
| 4.15 | Tax Matters | A-30 | ||||||
| 4.16 | Insurance | A-30 | ||||||
| 4.17 | Opinion of Mercury’s Financial Advisor | A-30 | ||||||
| 4.18 | Interested Stockholder | A-30 | ||||||
| 4.19 | Rights Agreement; Takeover Statutes | A-30 | ||||||
| 4.20 | Financing | A-30 | ||||||
| 4.21 | No Additional Representations | A-31 | ||||||
ARTICLE V CERTAIN PRE-CLOSING COVENANTS | ||||||||
| 5.1 | Conduct of the Business of Saturn | A-31 | ||||||
| 5.2 | Conduct of the Business of Mercury | A-35 | ||||||
| 5.3 | No Control of Other Party’s Business | A-36 | ||||||
ARTICLE VI ADDITIONAL AGREEMENTS | ||||||||
| 6.1 | Preparation of the Joint Proxy Statement and the Registration Statement | A-37 | ||||||
| 6.2 | Shareholders Meetings; Recommendations | A-38 | ||||||
| 6.3 | Access to Information; Confidentiality | A-38 | ||||||
| 6.4 | No Solicitation | A-39 | ||||||
| 6.5 | Efforts to Consummate; Notification | A-45 | ||||||
| 6.6 | Certain Notices | A-47 | ||||||
| 6.7 | Public Announcements | A-47 | ||||||
| 6.8 | Indemnification of Directors and Officers | A-48 | ||||||
| 6.9 | Employee Benefits | A-49 | ||||||
| 6.10 | Financing | A-53 | ||||||
| 6.11 | Takeover Statutes | A-56 | ||||||
| 6.12 | Transaction Litigation | A-56 | ||||||
| 6.13 | NYSE Listing | A-56 | ||||||
| 6.14 | Overseas Financing | A-56 | ||||||
| 6.15 | Convertible Preferred Stock Conversion | A-56 | ||||||
A-ii
Table of Contents
| Page | ||||||||
| 6.16 | Dividends | A-57 | ||||||
| 6.17 | Tax-Free Qualification | A-57 | ||||||
| 6.18 | Tax Treatment of Specified Subsidiaries | A-57 | ||||||
| 6.19 | Tax Representation Letters | A-57 | ||||||
| 6.20 | Environmental Matters | A-57 | ||||||
ARTICLE VII CONDITIONS | ||||||||
| 7.1 | Conditions to Obligations of Each Party under this Agreement | A-58 | ||||||
| 7.2 | Conditions to Mercury’s Obligations | A-58 | ||||||
| 7.3 | Conditions to Saturn’s, Merger Sub 1’s and Merger Sub 2’s Obligations | A-59 | ||||||
ARTICLE VIII TERMINATION AND EXPENSES | ||||||||
| 8.1 | Termination | A-59 | ||||||
| 8.2 | Notice of Termination; Effect of Termination | A-61 | ||||||
| 8.3 | Expenses and Other Payments | A-62 | ||||||
ARTICLE IX DEFINITIONS | ||||||||
| 9.1 | Definitions | A-64 | ||||||
| 9.2 | Construction | A-74 | ||||||
ARTICLE X MISCELLANEOUS | ||||||||
| 10.1 | Non-Survival of Representations and Warranties | A-74 | ||||||
| 10.2 | Notices | A-74 | ||||||
| 10.3 | Severability | A-75 | ||||||
| 10.4 | Entire Agreement | A-75 | ||||||
| 10.5 | Assignment; Merger Subs | A-75 | ||||||
| 10.6 | Extension; Waiver | A-75 | ||||||
| 10.7 | Third Party Beneficiaries | A-75 | ||||||
| 10.8 | No Strict Construction | A-75 | ||||||
| 10.9 | Governing Law; Consent to Jurisdiction | A-75 | ||||||
| 10.10 | Disclosure Letters | A-76 | ||||||
| 10.11 | Specific Performance | A-76 | ||||||
| 10.12 | WAIVER OF TRIAL BY JURY | A-76 | ||||||
| 10.13 | Counterparts | A-77 | ||||||
| 10.14 | Amendment | A-77 | ||||||
| Exhibit A | Restated Certificate of Incorporation of Saturn Merger Surviving Corporation | |
| Exhibit B | Bylaws of Saturn Merger Surviving Corporation | |
| Exhibit C | Certificate of Incorporation of Mercury Merger Surviving Corporation | |
| Exhibit D | Bylaws of Mercury Merger Surviving Corporation |
A-iii
Table of Contents
A-1
Table of Contents
A-2
Table of Contents
A-3
Table of Contents
A-4
Table of Contents
A-5
Table of Contents
A-6
Table of Contents
A-7
Table of Contents
A-8
Table of Contents
A-9
Table of Contents
A-10
Table of Contents
A-11
Table of Contents
A-12
Table of Contents
A-13
Table of Contents
A-14
Table of Contents
A-15
Table of Contents
A-16
Table of Contents
A-17
Table of Contents
A-18
Table of Contents
A-19
Table of Contents
A-20
Table of Contents
A-21
Table of Contents
A-22
Table of Contents
A-23
Table of Contents
A-24
Table of Contents
A-25
Table of Contents
A-26
Table of Contents
A-27
Table of Contents
A-28
Table of Contents
A-29
Table of Contents
A-30
Table of Contents
A-31
Table of Contents
A-32
Table of Contents
A-33
Table of Contents
A-34
Table of Contents
A-35
Table of Contents
A-36
Table of Contents
A-37
Table of Contents
A-38
Table of Contents
A-39
Table of Contents
A-40
Table of Contents
A-41
Table of Contents
A-42
Table of Contents
A-43
Table of Contents
A-44
Table of Contents
A-45
Table of Contents
A-46
Table of Contents
A-47
Table of Contents
A-48
Table of Contents
A-49
Table of Contents
A-50
Table of Contents
A-51
Table of Contents
A-52
Table of Contents
A-53
Table of Contents
A-54
Table of Contents
A-55
Table of Contents
A-56
Table of Contents
A-57
Table of Contents
A-58
Table of Contents
A-59
Table of Contents
A-60
Table of Contents
A-61
Table of Contents
A-62
Table of Contents
A-63
Table of Contents
A-64
Table of Contents
A-65
Table of Contents
A-66
Table of Contents
A-67
Table of Contents
A-68
Table of Contents
A-69
Table of Contents
A-70
Table of Contents
A-71
Table of Contents
A-72
Table of Contents
A-73
Table of Contents
| Attn: | David N. Shine Philip Richter |
A-74
Table of Contents
| Attn: | Thomas Sabatino K.C. Lam |
| Attn: | Andrew R. Brownstein Gavin D. Solotar |
A-75
Table of Contents
A-76
Table of Contents
A-77
Table of Contents
| By: |  |
| Title: | Chairman, President and Chief |
| By: |  |
| Title: | Chairman and Chief Executive Officer |
| By: |  |
| Title: | Vice President |
| By: |  |
| Title: | Vice President |
A-78
Table of Contents
B-1
Table of Contents
B-2
Table of Contents
B-3
Table of Contents

C-1
Table of Contents
C-2
Table of Contents
C-3
Table of Contents

D-1
Table of Contents
D-2
Table of Contents
| By: |
D-3