UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
☒ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| |
For the fiscal year ended December 31, 2021 | |
| |
or | |
| |
☐ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| |
For the transition period from ______ to ______
Commission File Number: 001-36612

ReWalk Robotics Ltd.
(Exact name of registrant as specified in charter)
Israel |
| Not applicable |
(State or other jurisdiction of incorporation or organization) |
| (I.R.S. employer identification no.) |
|
| |
3 Hatnufa Street, Floor 6, Yokneam Ilit, Israel |
| 2069203 |
(Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: +972.4.959.0123
Securities registered pursuant to Section 12(b) of the Act:
Title of Each Class | Trading Symbol(s) | Name of Each Exchange on Which Registered | ||
Ordinary Shares, par value NIS 0.25 per share | RWLK | Nasdaq Capital Market |
Securities Registered Pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes ☐ No ☒
Indicate by a check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ | Accelerated filer ☐ | |||
Non-accelerated filer ☒ | Smaller reporting company ☒ | |||
Emerging growth company ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes ☐ No ☒
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 USC. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
The aggregate market value of the Ordinary Shares held by non-affiliates of the Registrant based upon the closing price of the Ordinary Shares as reported by the Nasdaq Capital Market on June 30, 2021 (the last business day of the Registrant’s most recently completed second fiscal quarter) was $77,594,282.
As of February 24, 2022, the Registrant had outstanding 62,507,717 Ordinary Shares, par value NIS 0.25 per share.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of our proxy statement for our 2022 Annual Meeting of Shareholders, which is to be filed within 120 days after the end of our 2021 fiscal year, are incorporated by reference into Part III of this annual report on Form 10-K.
Page No | ||
| 1 | ||
| 26 | ||
| 62 | ||
| 62 | ||
| 63 | ||
| 63 | ||
| 64 | ||
| 67 | ||
| 67 | ||
| 82 | ||
| 82 | ||
| 83 | ||
| 83 | ||
| 84 | ||
| 85 | ||
| 85 | ||
| 85 | ||
| 85 | ||
| 85 | ||
| 86 | ||
| 90 | ||
| 91 | ||
| 92 | ||
| F-1 | ||
| • | our expectations regarding future growth, including our ability to increase sales in our existing geographic markets and expand to new markets; |
| • | our ability to maintain and grow our reputation and the market acceptance of our products; |
| • | our ability to achieve reimbursement from third-party payors or advance Centers for Medicare & Medicaid Services (“CMS”) coverage for our products; |
| • | Our ability to maintain compliance with the continued requirements of the Nasdaq Capital Market and the risk that our ordinary shares will be delisted if we do not comply with such requirements; |
| • | the adverse effect that the COVID-19 pandemic has had and continues to have on our business and results of operations; |
| • | our ability to have sufficient funds to meet certain future capital requirements, which could impair our efforts to develop and commercialize existing and new products; |
| • | our limited operating history and our ability to leverage our sales, marketing and training infrastructure; |
| • | our ability to grow our business through acquisitions of businesses, products or technologies, and the failure to manage acquisitions, or the failure to integrate them with our existing business, which could have a material adverse effect on our business, financial condition, and operating results; |
| • | our expectations as to our clinical research program and clinical results; |
| • | our ability to obtain certain components of our products from third-party suppliers and our continued access to our product manufacturers; |
| • | our ability to improve our products and develop new products; |
| • | our compliance with medical device reporting regulations to report adverse events involving our products, which could result in voluntary corrective actions or enforcement actions such as mandatory recalls, and the potential impact of such adverse events on our ability to market and sell our products; |
| • | our ability to gain and maintain regulatory approvals and to comply with any post-marketing requests |
| • | the risk of a cybersecurity attack or breach of our information technology systems significantly disrupting our business operations; |
| • | our ability to maintain adequate protection of our intellectual property and to avoid violation of the intellectual property rights of others; |
| • | the impact of substantial sales of our shares by certain shareholders on the market price of our ordinary shares; |
| • | our ability to use effectively the proceeds of our offerings of securities; |
| • | the risk of substantial dilution resulting from the periodic issuances of our ordinary shares; |
| • | the impact of the market price of our ordinary shares on the determination of whether we are a passive foreign investment company; |
| • | market and other conditions; and |
| • | other factors discussed in “Part I. Item 1A. Risk Factors.” |
| ReWalk Personal 6.0 | ||
● ReWalk Personal: intended for everyday use at home, at work or in the community with a trained companion. We began marketing ReWalk Personal in Europe with CE mark clearance at the end of 2012. We received FDA clearance to market ReWalk Personal in the United States in June 2014. ReWalk Personal units are all manufactured according to the same mechanical specifications. Each unit is then permanently sized to fit the individual user and the software is configured for the user’s specifications by the rehabilitation center, clinic or distributor. We are currently offering our 6th generation device. ● ReWalk Rehabilitation: the current offering for clinics who wish to implement exo-skeleton training is comprised of our Personal 6.0 unit along with multiple sizing of different parts, enabling multiple patient use. The replacement of parts for different sizing is done by the clinic team and can take between 5 to 15 minutes. ReWalk Rehabilitation provides a valuable means of exercise and therapy. It also enables individuals to evaluate their capacity for using ReWalk Personal in the future. We began marketing a unique design for use in hospitals, rehabilitation centers and stand-alone training centers in the United States and Europe in 2011 and in December 2020, we decided to end the production of this unique design. |  |
Spinal Cord Injury |
Spinal cord injury is the result of a direct trauma to the nerves themselves or damage to the surrounding bones and soft tissues which ultimately impacts the spinal cord. Spinal cord damage results in a loss of function, such as mobility or feeling. In most people who have spinal cord injury, the spinal cord is intact. Spinal cord injury is not the same as back injury, which may result from pinched nerves or ruptured disks. Even when a person sustains a break in a vertebra or vertebrae, there may not be any spinal cord injury if the spinal cord itself is not affected. There are two types of spinal cord injury – complete and incomplete. In a complete injury, a person loses all ability to feel and voluntarily move below the level of the injury. In an incomplete injury, there is some functioning below the level of the injury. Upon examination, a patient is assigned a level of injury depending on the location of the spinal cord injury. Cervical level injuries cause paralysis or weakness in both arms and legs and is referred to as quadriplegia. Sometimes this type of injury is accompanied by loss of physical sensation, respiratory issues, bowel, bladder, and sexual dysfunction. Thoracic level injuries can cause paralysis or weakness of the legs (paraplegia) along with loss of physical sensation, bowel, bladder, and sexual dysfunction. In most cases, arms and hands are not affected. Lumbar level injuries result in paralysis or weakness of the legs (paraplegia). Loss of physical sensation, bowel, bladder, and sexual dysfunction can occur. The shoulder, arm, and hand functions are usually unaffected. Sacral level injuries primarily cause loss of bowel and bladder function as well as sexual dysfunction. |
| ● | reduced pain; |
| ● | improved bowel and urinary tract function; |
| ● | reduced spasticity; |
| ● | increases in joint range of motion for the hip and ankle joints; |
| ● | improved sleep and reduced fatigue; |
| ● | increase in oxygen uptake and heart rate as a result of walking as opposed to sitting and standing; |
| ● | ability to ambulate at a speed greater than 0.4 meters per second, which is considered to be conducive to outdoor related community ambulation; and |
| ● | reduced hospitalizations. |
| ● | In September 2017, Barmer confirmed it will provide ReWalk systems to all qualifying beneficiaries. Barmer provides insurance coverage for nearly nine million people in Germany, as a member of the SHI network and one of the most significant national insurers in the country. Exoskeletons are provided to users that meet certain inclusion criteria and assessment by the German Health Insurance Medical Service (Medizinischer Dienst der Krankenversicherungen) before and after training. We remain in discussion with Barmer regarding a contract based on their 2017 decision. |
| ● | In September 2017 Germany’s national social accident insurance provider, DGUV, indicated that the DGUV’s member payers, including the health insurance association Berufsgenossenschaft (also known as BG) and state insurers, will approve the supply of exoskeleton systems for qualifying beneficiaries on a case-by-case basis. DGUV is comprised of 36 different insurers, which provide coverage for more than 80 million individuals in Germany. Per the agreement, eligible individuals go to BG clinics for evaluation as a part of the procurement. In May 2020 the DGUV agreed to a binding offer to the evaluation, training, and supply of the ReWalk Personal 6.0 device to qualified individuals. |
| ● | In February 2018, the GKV-Spitzenverband (Central Federal Association of (the) Statutory Health Insurance Funds) confirmed its decision to list the ReWalk Personal system in the German MDD, a comprehensive list of all medical devices which are principally and regularly reimbursed by German SHI and PHI providers. The ReWalk Personal was added to the official German list of medical aids, code number 23.29.01.2001, in June 2018. This decision means that ReWalk Personal is listed among all medical devices for compensation, which SHI providers can procure for any approved beneficiary on a case-by-case basis. |
| ● | During the year 2020 we have announced several new agreements with SHIs such as TK and DAK-Gesundheit and others as well as the first German Private Health Insurer (“PHI”) that have chosen to enter into an agreement with us that outline the process to obtaining a device for eligible insured patients. |
| ● | In March 2021 we entered into a contract with BKK Mobile Oil health insurance to supply ReWalk’s Personal 6.0 System to eligible persons in Germany. |
| ● | In June 2020, a certain SHI has appealed the decision of the State Social Court, which ordered the supply of the SHI’s insured SCI person with ReWalk. The State Social Court ruled and deemed ReWalk as the medical aid which will directly compensate the plaintiff’s disability. The SHI appealed this ruling with the Federal Social Court (Bundessozialgericht), which now has to decide whether an exoskeleton, as an orthopedic aid that replaces the function of the legs and enables independent walking and standing, serves to directly compensate for disability. The cost-effectiveness of an aid that serves to directly compensate for a disability is generally to be assumed and only examined if two actually equivalent but differently priced aids are available for selection. The 3rd Senate of the Federal Social Court is expected to announce the hearing date in the coming months. |
ReStore In June 2017 we unveiled our lightweight exo-suit ReStore system designed initially for rehabilitation of stroke patients. The patented soft exo-suit technology was originally developed at Harvard University’s Wyss Institute for Biologically Inspired Engineering, (“Harvard”) where it also underwent initial clinical testing that demonstrated potential to improve walking for stroke survivors. ReWalk and Harvard entered into a multi-year research collaboration agreement in 2016 which provides ReWalk license to intellectual property relating to lightweight exo-suit system technologies for lower limb disabilities and provides access to future innovations that emerge from this collaboration and may be relevant to additional stroke products or other therapies. The development and regulatory clearance process for ReStore took us approximately three years. In June 2019, we received FDA clearance following CE clearance in May 2019. Following the regulatory clearances, we began to commercialize the ReStore product. For more information on the collaboration with Harvard, see “Research and Development-Research and Development Collaborations.”. |  ReStore exo-suit |
| ● | establishment registration and device listing; |
| ● | development of a quality assurance system, including establishing and implementing procedures to design and manufacture devices; |
| ● | labeling regulations that prohibit the promotion of products for unapproved or “off-label” uses and impose other restrictions on labeling; |
| ● | FDA’s Unique Device Identification requirements that call for a unique device identifier (UDI) on device labels and packages and submission of data to the FDA’s Global Unique Device Identification Database (GUDID); |
| ● | medical device reporting regulations that require manufacturers to report to the FDA if a device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if it were to recur; and corrections and removal reporting regulations that require manufacturers report to the FDA field corrections and product recalls or removals if undertaken to reduce a risk to health posed by the device or to remedy a violation of the FFDCA that may present a risk to health; and |
| ● | Post-market surveillance. |
| ● | untitled letters, warning letters, fines, injunctions, consent decrees and civil penalties; |
| ● | customer notifications or repair, replacement, or refunds; |
| ● | recalls, withdrawals, or administrative detention or seizure of our products; |
| ● | operating restrictions or partial suspension or total shutdown of production; |
| ● | refusing or delaying requests for approval of pre-market approval applications relating to new products or modified products; |
| ● | withdrawing PMA approval; |
| ● | refusal to grant export approvals for our products; or |
| ● | pursuing criminal prosecution. |
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Revenues based on customer’s location: | ||||||||
| United States | $ | 2,519 | $ | 1,746 | ||||
| Europe | 3,381 | 2,631 | ||||||
| Asia-Pacific | 60 | 8 | ||||||
| Latin America | — | 6 | ||||||
| Africa | 2 | 2 | ||||||
| Total revenues | $ | 5,966 | $ | 4,393 | ||||
| ● | Annual revenue of $6.0 million in 2021 represents 36% year over year growth; |
| ● | Fourth quarter 2021 revenues were $1.2 million, up by 2% compared to previous year quarter; |
| ● | Strong cash position with $88.3 million as of December 31, 2021; |
| ● | New DMEPOS rules issued in December 2021 will advance consideration of the ReWalk benefit category and pricing, and |
| ● | German court case on ReWalk Personal 6.0 direct compensation decision expected later this year. |
While we have regained compliance with the quantitative continued listing rules of the Nasdaq Capital Market, we may not be able to maintain the listing of our ordinary shares on the Nasdaq Capital Market going forward, which could adversely affect our liquidity and the trading volume and market price of our ordinary shares.
• | ReWalk. We have sold only a limited number of ReWalk systems, and market acceptance and adoption depend on educating people with limited upright mobility and health care providers as to the distinct features, ease-of-use, positive lifestyle impact and other benefits of ReWalk compared to alternative technologies and treatments. ReWalk may not be perceived to have sufficient potential benefits compared with these alternatives. Users may also choose other therapies due to disadvantages of ReWalk, including the time it takes for a user to put on ReWalk, the slower pace of ReWalk compared to a wheelchair, the weight of ReWalk when carried, which makes it more burdensome for a companion to transport than a wheelchair, the required training, and the requirement that users be accompanied by a trained companion. Also, we believe that healthcare providers tend to be slow to change their medical treatment practices because of perceived liability risks arising from the use of new products and the uncertainty of third-party reimbursement. Accordingly, healthcare providers may not recommend ReWalk until there is sufficient evidence to convince them to alter the treatment methods they typically recommend, such as prominent healthcare providers or other key opinion leaders in the spinal cord injury community recommending ReWalk as effective in providing identifiable immediate and long-term health benefits. |
• | ReStore. The ReStore system is designed to provide advantages to stroke rehabilitation clinics and therapists as compared to other traditional therapies and devices by minimizing setup time, improving patients’ clinical results during therapy, supplying real-time analytics to optimize session productivity, and generating ongoing data reports to assist with tracking patient progress Since the ReStore device is currently being used only in the rehabilitative clinical setting, its market reception will depend heavily on our ability to demonstrate to clinics and therapists the systemic and economic benefits of using the ReStore device, its clinical advantage when compared to other devices or manual therapy, the functionality of the device for a significant portion of the patients that they treat and the overall advantages that the device provides to their patients compared to other technologies. Because the ReStore system is currently being used only in clinical settings, and we received FDA approval and CE clearance in 2019, close in time to the start of the COVID-19 pandemic, the overall sales of the system have been lower than originally anticipated, as many healthcare providers and rehabilitation centers have shifted focus from the clinical setting to at-home therapies and are generally less open for introduction of new technologies such as the ReStore. |
| • | a market will not sufficiently develop for our products; |
| • | we will not be able to develop scalable products and services, or that, although scalable, our products and services will not be economical to market nor will we get sufficient reimbursement coverage; |
| • | we will not be able to establish brand recognition and competitive advantages for our products; |
| • | we will not receive necessary regulatory clearances or approvals for our products; and |
| • | our competitors market an equivalent or superior product or hold proprietary rights that preclude us from marketing our products. |
| • | identify the product features that people with paraplegia or paralysis, their caregivers, and healthcare providers are seeking in a medical device that restores upright mobility and successfully incorporate those features into our products; |
| • | identify the product features that people with stroke, multiple sclerosis or other similar indications require while the products are used at home as well as what items are valuable to the clinics that provide them rehabilitation; |
| • | develop and introduce proposed products in sufficient quantities and in a timely manner; |
| • | adequately protect our intellectual property and avoid infringing upon the intellectual property rights of third-parties; |
| • | demonstrate the safety, efficacy, and health benefits of proposed products; and |
| • | obtain the necessary regulatory clearances and approvals for proposed products. |
| ● | untitled letters, warning letters, fines, injunctions, consent decrees and civil penalties; | |
| ● | customer notifications or repair, replacement, or refunds; |
| ● | operating restrictions or partial suspension or total shutdown of production; | |
| ● | recalls, withdrawals, or administrative detention or seizure of our products; | |
| ● | refusing or delaying requests for approval of pre-market approval applications relating to new products or modified products; | |
| ● | withdrawing a PMA approval; | |
| ● | refusing to provide Certificates for Foreign Government; | |
| ● | refusing to grant export approval for our products; or | |
| ● | pursuing criminal prosecution. |
The FCPA applies to companies, including ours, with a class of securities registered under the Exchange Act. The FCPA and other anti-bribery laws to which various aspects of our operations may be subject generally prohibit companies and their intermediaries from making improper payments to officials for the purpose of obtaining or retaining business. In various jurisdictions, our operations require that we and third parties acting on our behalf routinely interact with government officials, including medical personnel who may be considered government officials for purposes of these laws because they are employees of state-owned or controlled facilities. Other anti-bribery laws to which various aspects of our operations may be subject, including the United Kingdom Bribery Act, also prohibit improper payments to private parties and prohibit receipt of improper payments. Our policies prohibit our employees from making or receiving corrupt payments, including, among other things, to require compliance by third parties engaged to act on our behalf. Our policies mandate compliance with these anti-bribery laws; however, we operate in many parts of the world that have experienced governmental and/or private corruption to some degree. As a result, the existence and implementation of a robust anti-corruption program cannot eliminate all risk that unauthorized reckless or criminal acts have been or will be committed by our employees or agents. Violations of these laws, or allegations of such violations, could disrupt our business and harm our financial condition, results of operations, cash flows and reputation.
| ● | actual or anticipated fluctuations in our growth rate or results of operations or those of our competitors; | |
| ● | customer acceptance of our products; | |
| ● | announcements by us or our competitors of new products or services, commercial relationships, acquisitions, or expansion plans; | |
| ● | announcements by us or our competitors of other material developments; | |
| ● | our involvement in litigation; | |
| ● | changes in government regulation applicable to us and our products; |
| ● | sales, or the anticipation of sales, of our ordinary shares, warrants and debt securities by us, or sales of our ordinary shares by our insiders or other shareholders, including upon expiration of contractual lock-up agreements; | |
| ● | developments with respect to intellectual property rights; | |
| ● | competition from existing or new technologies and products; | |
| ● | changes in key personnel; | |
| ● | the trading volume of our ordinary shares; | |
| ● | changes in the estimation of the future size and growth rate of our markets; | |
| ● | changes in our quarterly or annual forecasts with respect to operating results and financial conditions; | |
| ● | general economic and market conditions and | |
| ● | Announcements regarding business acquisitions. |
| ● | problems assimilating the acquired products or technologies; | |
| ● | issues maintaining uniform standards, procedures, controls and policies; | |
| ● | problems integrating employees from an acquired organization into our company and integrating each company’s accounting, management information, human resources and other administrative systems; | |
| ● | unanticipated costs associated with acquisitions; | |
| ● | diversion of management’s attention from our existing business operations; | |
| ● | potential incurrence of debt, contingent liabilities or amortization expenses, or write-offs of goodwill; |
| ● | risks associated with entering new markets in which we have limited or no experience; and | |
| ● | increased legal and accounting costs relating to the acquisitions or compliance with regulatory matters. |
| Square feet (approximate) | ||||
| Marlborough, Massachusetts | 11,850 | |||
| Yokneam, Israel | 11,500 | |||
| Berlin, Germany | 753 | |||
| Total | 24,103 | |||
Among ReWalk Robotics, the NASDAQ Composite Index
and the NASDAQ Medical Equipment Index
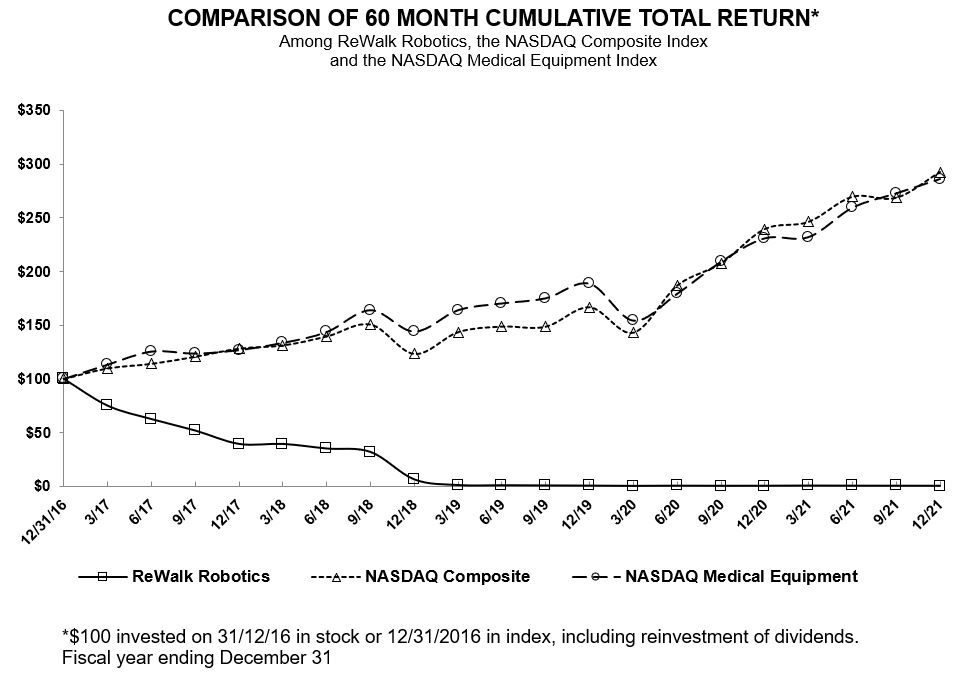
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Personal unit revenues | $ | 4,820 | $ | 4,220 | ||||
| Rehabilitation unit revenues | $ | 1,146 | $ | 173 | ||||
| Revenues | $ | 5,966 | $ | 4,393 | ||||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Gross profit | $ | 2,903 | $ | 2,189 | ||||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Research and development expenses, net | $ | 2,939 | $ | 3,459 | ||||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Sales and marketing expenses | $ | 6,993 | $ | 5,754 | ||||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| General and administrative | $ | 5,626 | $ | 4,980 | ||||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Financial expenses (income), net | $ | (13 | ) | $ | 921 | |||
| Years Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Taxes on income | $ | 94 | $ | 51 | ||||
| Years Ended December 31, | ||||||||||||
| 2021 | 2020 | 2019 | ||||||||||
| Net cash used in operating activities | $ | (11,469 | ) | $ | (12,589 | ) | $ | (14,815 | ) | |||
| Net cash provided by used in investing activities | (47 | ) | (73 | ) | (22 | ) | ||||||
| Net cash provided by financing activities | 79,512 | 16,724 | 21,482 | |||||||||
| Net cash flow | $ | 67,996 | $ | 4,062 | $ | 6,645 | ||||||
| Payments due by period (in dollars, in thousands) | ||||||||||||
| Contractual obligations | Total | Less than 1 year | 1-3 years | |||||||||
| Purchase obligations (1) | $ | 1,457 | $ | 1,457 | $ | — | ||||||
| Collaboration Agreement and License Agreement obligations (2) | 145 | 145 | — | |||||||||
| Operating lease obligations (3) | 1,189 | 689 | 500 | |||||||||
| Total | $ | 2,791 | $ | 2,291 | $ | 500 | ||||||
| (1) | The Company depends on one contract manufacturer, Sanmina Corporation, for both the ReStore products and the SCI Products. We place our manufacturing orders with Sanmina pursuant to purchase orders or by providing forecasts for future requirements |
| (2) | Our Collaboration Agreement was originally signed for a period of six years and as of December 31, 2021 has a remaining term of 0.25 years, it requires us to pay in quarterly installments for the funding of our joint research collaboration with Harvard, subject to a minimum funding commitment under applicable circumstances. Our License Agreement consists of patent reimbursement expenses payments and of a license upfront fee payment. There are also several milestone payments contingent upon the achievement of certain product development and commercialization milestones and royalty payments on net sales from certain patents licensed to Harvard. These product development milestones have been met as of December 31, 2021. There are commercialization milestones which depend on us reaching certain sales amounts some or all of which may not occur. |
| (3) | Our operating leases consist of leases for our facilities and motor vehicles. |
| Change in Average Exchange Rate | ||||||||
| Period | NIS against the U.S. Dollar (%) | Euro against the U.S. Dollar (%) | ||||||
| 2021 | (6.38 | ) | 3.46 | |||||
| 2020 | 3.76 | 2.07 | ||||||
| 2019 | 0.87 | (5.16 | ) | |||||
| ● | pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; |
| ● | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with U.S. GAAP, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and |
| ● | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of our assets that could have a material effect on our financial statements. |
| Name | Age | Position | ||
| Larry Jasinski | 64 | Chief Executive Officer and Director | ||
| Ori Gon | 40 | Chief Financial Officer |
| 10.1 | Letter of Agreement, dated July 11, 2013, between the Company and Sanmina Corporation.* | |
| 10.2 | Research Collaboration Agreement, dated May 16, 2016, between the Company and the President and Fellows of Harvard College.* | |
| 10.3 | License Agreement, dated May 16, 2016, between the Company and the President and Fellows of Harvard College.* | |
| 101.INS | XBRL Instance Document. | |
| 101.SCH | XBRL Taxonomy Extension Schema Document. | |
| 101.PRE | XBRL Taxonomy Presentation Linkbase Document. | |
| 101.CAL | XBRL Taxonomy Calculation Linkbase Document. | |
| 101.LAB | XBRL Taxonomy Label Linkbase Document. | |
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase Document. |
| * | Certain identified information in the exhibit has been omitted because it is the type of information that (i) the Company customarily and actually treats as private and confidential, and (ii) is not material. |
| ** | Management contract or compensatory plan, contract or arrangement. |
| *** | Furnished herewith. |
| ReWalk Robotics Ltd. | ||
| By: | /s/ Larry Jasinski | |
| Name: Larry Jasinski | ||
| Title: Chief Executive Officer | ||
| Signature | Title | Date | ||
| /s/ Larry Jasinski | Director and Chief Executive Officer (Principal Executive Officer) | February 24, 2022 | ||
| Larry Jasinski | ||||
| /s/ Ori Gon | Chief Financial Officer | February 24, 2022 | ||
| Ori Gon | (Principal Financial Officer and Principal Accounting Officer) | |||
| /s/ Jeff Dykan | Chairman of the Board | February 24, 2022 | ||
| Jeff Dykan | ||||
| /s/ Yohanan R Engelhardt | Director | February 24, 2022 | ||
| Yohanan R Engelhardt | ||||
| /s/ Dr. John William Poduska | Director | February 24, 2022 | ||
| Dr. John William Poduska | ||||
| /s/ Wayne B. Weisman | Director | February 24, 2022 | ||
| Wayne B. Weisman | ||||
| /s/ Yasushi Ichiki | Director | February 24, 2022 | ||
| Yasushi Ichiki | ||||
| /s/ Aryeh Dan | Director | February 24, 2022 | ||
| Aryeh Dan | ||||
| /s/ Randel Richner | Director | February 24, 2022 | ||
| Randel Richner |
PART IV
REWALK ROBOTICS LTD
CONSOLIDATED FINANCIAL STATEMENTS
U.S. DOLLARS IN THOUSANDS
INDEX
(PCAOB ID 1281)
F-4 | |
F-6 | |
F-7 | |
F-8 | |
F-10 |
F - 1
| Kost Forer Gabbay & Kasierer Menachem Begin 144, Tel-Aviv 6492102, Israel | Tel: +972-3-6232525 Fax: +972-2-5622555 ey.com |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
REWALK ROBOTICS LTD.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Rewalk Robotics Ltd. and subsidiaries (the Company) as of December 31, 2021 and 2020, the related consolidated statements of operations, changes in shareholders’ equity and cash flows for each of the three years in the period ended December 31, 2021, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2021 and 2020, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2021, in conformity with U.S. generally accepted accounting principles.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the account or disclosure to which it relates.
F - 2
Revenue recognition | |
| |
Description of the Matter | As described in Note 2 to the consolidated financial statements, the Company generates revenues from sales of products. Revenue is recognized when obligations under the terms of a contract with the Company's customers are satisfied. Revenue is measured as the amount of consideration to which the Company expects to be entitled in exchange for transferring products or providing services. In addition, the Company provides a service type warranty which is accounted for as a separate performance obligation. Revenue is then recognized ratably over the life of the warranty.
|
Auditing the Company’s revenue recognition involves subjective assumptions used in determining the standalone selling price of distinct performance obligations. | |
| |
How We Addressed the Matter in Our Audit | Our audit procedures included, among others, reading the executed contract and purchase order to understand the contract, identify the performance obligations and evaluate management’s identification of the distinct performance obligations for a sample of contracts. To test the management’s determination of standalone selling prices for each performance obligation, our audit procedures included, among others, evaluating the methodology applied and testing the calculations as well as the completeness and accuracy of the underlying data and assumptions used by the Company in its estimates. We also evaluated the Company’s disclosures included in notes to the consolidated financial statements. |
KOST FORER GABBAY & KASIERER
A Member of Ernst & Young Global
We have served as the Company’s auditor since 2014.
Tel-Aviv, Israel
February 24, 2022
F - 3
REWALK ROBOTICS LTD. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
U.S. dollars in thousands
December 31, | ||||||||
2021 | 2020 | |||||||
ASSETS | ||||||||
CURRENT ASSETS: | ||||||||
Cash and cash equivalents | $ | 88,337 | $ | 20,350 | ||||
Trade receivable, net | 585 | 684 | ||||||
Prepaid expenses and other current assets | 610 | 672 | ||||||
Inventories | 2,989 | 3,542 | ||||||
Total current assets | 92,521 | 25,248 | ||||||
| ||||||||
LONG-TERM ASSETS | ||||||||
| ||||||||
Restricted cash and other long term assets | 1,064 | 1,033 | ||||||
Operating lease right-of-use assets | 881 | 1,349 | ||||||
Property and equipment, net | 284 | 437 | ||||||
Total long-term assets | 2,229 | 2,819 | ||||||
| ||||||||
Total assets | $ | 94,750 | $ | 28,067 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
F - 4
REWALK ROBOTICS LTD. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
U.S. dollars in thousands (except share and per share data)
December 31, | ||||||||
2021 | 2020 | |||||||
LIABILITIES AND SHAREHOLDERS’ EQUITY | ||||||||
CURRENT LIABILITIES: | ||||||||
Current maturities of operating leases liability | $ | 641 | $ | 660 | ||||
Trade payables | 1,384 | 2,268 | ||||||
Employees and payroll accruals | 1,142 | 867 | ||||||
Deferred revenues | 316 | 441 | ||||||
Other current liabilities | 555 | 432 | ||||||
Total current liabilities | 4,038 | 4,668 | ||||||
| ||||||||
LONG-TERM LIABILITIES | ||||||||
| ||||||||
Deferred revenues | 866 | 667 | ||||||
Non-current operating leases liability | 418 | 923 | ||||||
Other long-term liabilities | 45 | 35 | ||||||
Total long-term liabilities | 1,329 | 1,625 | ||||||
| ||||||||
Total liabilities | 5,367 | 6,293 | ||||||
| ||||||||
COMMITMENTS AND CONTINGENT LIABILITIES | ||||||||
Shareholders’ equity: | ||||||||
| ||||||||
Share capital | ||||||||
Ordinary share of NIS 0.25 par value-Authorized: 120,000,000 and 60,000,000 shares at December 31, 2021 and 2020; Issued and outstanding: 62,480,163 and 25,332,225 shares at December 31, 2021 and December 31, 2020, respectively | 4,661 | 1,827 | ||||||
Additional paid-in capital | 278,903 | 201,392 | ||||||
Accumulated deficit | (194,181 | ) | (181,445 | ) | ||||
| ||||||||
Total shareholders’ equity | 89,383 | 21,774 | ||||||
| ||||||||
Total liabilities and shareholders’ equity | $ | 94,750 | $ | 28,067 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
F - 5
REWALK ROBOTICS LTD. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
U.S. dollars in thousands (except share and per share data)
Year ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Revenues | $ | 5,966 | $ | 4,393 | $ | 4,873 | ||||||
Cost of revenues | 3,063 | 2,204 | 2,147 | |||||||||
| ||||||||||||
Gross profit | 2,903 | 2,189 | 2,726 | |||||||||
| ||||||||||||
Operating expenses: | ||||||||||||
Research and development | 2,939 | 3,459 | 5,348 | |||||||||
Sales and marketing | 6,993 | 5,754 | 6,167 | |||||||||
General and administrative | 5,626 | 4,980 | 5,259 | |||||||||
| ||||||||||||
Total operating expenses | 15,558 | 14,193 | 16,774 | |||||||||
| ||||||||||||
Operating loss | (12,655 | ) | (12,004 | ) | (14,048 | ) | ||||||
| ||||||||||||
Financial expenses (income), net | (13 | ) | 921 | 1,496 | ||||||||
| ||||||||||||
Loss before income taxes | (12,642 | ) | (12,925 | ) | (15,544 | ) | ||||||
Taxes on income | 94 | 51 | 7 | |||||||||
| ||||||||||||
Net loss | $ | (12,736 | ) | $ | (12,976 | ) | $ | (15,551 | ) | |||
| ||||||||||||
Net loss per ordinary share, basic and diluted | $ | (0.27 | ) | $ | (0.82 | ) | $ | (2.70 | ) | |||
| ||||||||||||
Weighted average number of shares used in computing net loss per ordinary share, basic and diluted | 47,935,652 | 15,764,980 | 5,763,317 | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F - 6
REWALK ROBOTICS LTD. AND SUBSIDIARIES
STATEMENTS OF CHANGES IN SHAREHOLDERS’ EQUITY
U.S. dollars in thousands (except share data)
| Ordinary Share | Additional paid-in | Accumulated | Total shareholders’ | ||||||||||||||||
Number | Amount | capital | deficit | equity | ||||||||||||||||
Balance as of December 31, 2018 | 2,813,087 | 193 | 154,670 | (152,918 | ) | 1,945 | ||||||||||||||
Share-based compensation to employees and non-employees | — | — | 1,108 | — | 1,108 | |||||||||||||||
Issuance of ordinary shares upon exercise of options to purchase ordinary shares and RSUs by employees and non-employees | 47,473 | 2 | — | — | 2 | |||||||||||||||
Issuance of ordinary shares in a “best effort” offering, net of issuance expenses in the amount of $686 (1) | 760,000 | 52 | 3,632 | 0— | 3,684 | |||||||||||||||
Exercise of pre-funded warrants and warrants (1) | 584,087 | 40 | 1,461 | 0— | 1,501 | |||||||||||||||
Issuance of ordinary shares in a “Registered Direct” offering, net of issuance expenses in the amount of $1,125 (1) | 1,650,248 | 115 | 8,010 | 0— | 8,125 | |||||||||||||||
Issuance of ordinary shares in a “Warrant exercise” agreement, net of issuance expenses in the amount of $1,019 (1) | 1,464,665 | 102 | 9,864 | 0— | 9,966 | |||||||||||||||
Net loss | — | — | — | (15,551 | ) | (15,551 | ) | |||||||||||||
| ||||||||||||||||||||
Balance as of December 31, 2019 | 7,319,560 | 504 | 178,745 | (168,469 | ) | 10,780 | ||||||||||||||
Share-based compensation to employees and non-employees | — | — | 749 | — | 749 | |||||||||||||||
Issuance of ordinary shares upon exercise of options to purchase ordinary shares and RSUs by employees and non-employees | 63,111 | 3 | (3 | ) | — | 0— | ||||||||||||||
Issuance of ordinary shares in a “Best Efforts” offering, net of issuance expenses in the amount of $1,056 (1) | 4,053,172 | 290 | 3,720 | 0— | 4,010 | |||||||||||||||
Exercise of pre-funded warrants and warrants (1)(2) | 3,378,328 | 244 | 3,979 | 0— | 4,223 | |||||||||||||||
Issuance of ordinary shares in a “registered direct” offering, net of issuance expenses in the amount of $1,019 (1) | 4,938,278 | 357 | 7,624 | 0— | 7,981 | |||||||||||||||
Issuance of ordinary shares in a private placement, net of issuance expenses in the amount of $993 (1) | 5,579,776 | 429 | 6,578 | 0— | 7,007 | |||||||||||||||
Net loss | — | — | — | (12,976 | ) | (12,976 | ) | |||||||||||||
Balance as of December 31, 2020 | 25,332,225 | 1,827 | 201,392 | (181,445 | ) | 21,774 | ||||||||||||||
Share-based compensation to employees and non-employees | — | — | 833 | — | 833 | |||||||||||||||
Issuance of ordinary shares upon vesting of RSUs by employees and non-employees | 398,164 | 31 | (31 | ) | — | 0— | ||||||||||||||
Issuance of ordinary shares in a “Best Efforts” offering, net of issuance expenses in the amount of $3,679 (1) | 10,921,502 | 832 | 35,489 | 0— | 36,321 | |||||||||||||||
Exercise of pre-funded warrants and warrants (1)(2) | 10,425,258 | 772 | 14,288 | 0— | 15,060 | |||||||||||||||
Issuance of ordinary shares in a “registered direct” offering, net of issuance expenses in the amount of $3,215 (1) | 15,403,014 | 1,199 | 26,932 | 0— | 28,131 | |||||||||||||||
Net loss | — | — | — | (12,736 | ) | (12,736 | ) | |||||||||||||
Balance as of December 31, 2021 | 62,480,163 | 4,661 | 278,903 | (194,181 | ) | 89,383 | ||||||||||||||
*) | Represents an amount lower than $1. |
(1) | See Note 8b. |
(2) | See Note 8f. |
The accompanying notes are an integral part of these consolidated financial statements.
F - 7
REWALK ROBOTICS LTD. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
U.S. dollars in thousands
| Year ended December 31, | |||||||||||
| 2021 | 2020 | 2019 | |||||||||
Cash flows used in operating activities: | ||||||||||||
Net loss | $ | (12,736 | ) | $ | (12,976 | ) | $ | (15,551 | ) | |||
Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| ||||||||||||
Depreciation | 266 | 285 | 321 | |||||||||
Share-based compensation to employees and non-employees | 833 | 749 | 1,108 | |||||||||
Deferred taxes | (29 | ) | (44 | ) | (57 | ) | ||||||
Gain on PPP forgiveness | 0— | (392 | ) | 0— | ||||||||
| ||||||||||||
Changes in assets and liabilities: | ||||||||||||
| ||||||||||||
Trade receivables, net | 99 | 110 | (36 | ) | ||||||||
Prepaid expenses, operating lease right-of-use assets and other assets | 592 | 166 | 64 | |||||||||
Inventories | 432 | (469 | ) | (1,221 | ) | |||||||
Trade payables | (884 | ) | (506 | ) | 370 | |||||||
Employees and payroll accruals | 275 | 197 | 20 | |||||||||
Deferred revenues | 74 | 264 | 176 | |||||||||
Operating lease liabilities and other liabilities | (391 | ) | 27 | (9 | ) | |||||||
Net cash used in operating activities | (11,469 | ) | (12,589 | ) | (14,815 | ) | ||||||
| ||||||||||||
Cash flows used in investing activities: | ||||||||||||
Purchase of property and equipment | (47 | ) | (73 | ) | (22 | ) | ||||||
Net cash used in investing activities | (47 | ) | (73 | ) | (22 | ) | ||||||
| ||||||||||||
Cash flows from financing activities: | ||||||||||||
Repayment of long-term loan | 0— | (6,965 | ) | (1,722 | ) | |||||||
Proceeds from PPP loan (3) | 0— | 392 | 0— | |||||||||
Issuance of ordinary shares in a “best effort” offering, net of issuance expenses in the amount of $686 (1) | 0— | 0— | 3,684 | |||||||||
Issuance of ordinary shares in a “registered direct” offering, net of issuance expenses in the amount of $1,035 (1) | 0— | 0— | 8,125 | |||||||||
Issuance of ordinary shares in a “warrant exercise” agreement, net of issuance expenses in the amount of $1,019 (1) | 0— | 0— | 9,966 | |||||||||
Issuance of ordinary shares in a “best effort” offering, net of issuance expenses in the amount of $1,056 (1) | 0— | 4,010 | 0— | |||||||||
Issuance of ordinary shares in a “registered direct” offering, net of issuance expenses in the amount of $977 (1) | 0— | 8,023 | 0— | |||||||||
Issuance of ordinary shares in a private placement, net of issuance expenses in the amount of $959 (1) | 0— | 7,041 | 0— | |||||||||
Issuance of ordinary shares in a private placement, net of issuance expenses paid in the amount of $3,679 (1) | 36,321 | 0— | 0— | |||||||||
Issuance of ordinary shares in a “registered direct” offering, net of issuance expenses in the amount of $3,215 (1) | 28,131 | 0— | 0— | |||||||||
Exercise of pre-funded warrants and warrants (1)(2) | 15,060 | 4,223 | 1,429 | |||||||||
Net cash provided by financing activities | 79,512 | 16,724 | 21,482 | |||||||||
| ||||||||||||
Increase in cash, cash equivalents, and restricted cash | 67,996 | 4,062 | 6,645 | |||||||||
Cash, cash equivalents, and restricted cash at beginning of period | 21,054 | 16,992 | 10,347 | |||||||||
Cash, cash equivalents, and restricted cash at end of period | $ | 89,050 | $ | 21,054 | $ | 16,992 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F - 8
REWALK ROBOTICS LTD. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
U.S. dollars in thousands
| Year ended December 31, | |||||||||||
| 2021 | 2020 | 2019 | |||||||||
| ||||||||||||
Supplemental disclosures of non-cash flow information |
|
|
|
|
|
|
|
|
| |||
Expenses related to offerings not yet paid (1) | $ | 0— | $ | 76 | $ | 90 | ||||||
Classification of other current assets to property and equipment, net | $ | 34 | $ | 98 | $ | 0— | ||||||
Classification of inventory to other current assets | $ | 89 |
| $ | 0— |
| $ | 164 | ||||
Classification of inventory to property and equipment | $ | 32 | $ | 50 | $ | 174 | ||||||
Cashless exercise of pre-funded warrants | $ | 0— | $ | 0— | $ | 72 | ||||||
Initial recognition of operating lease right-of-use assets | $ | 0— | $ | 0— | $ | 2,099 | ||||||
Initial recognition of operating lease liabilities | $ | 0— | $ | 0— | $ | (2,249 | ) | |||||
|
| |||||||||||
Supplemental disclosures of cash flow information: | ||||||||||||
Cash and cash equivalents | $ | 88,337 | $ | 20,350 | $ | 16,253 | ||||||
Restricted cash included in other long-term assets | $ | 713 | $ | 704 | $ | 739 | ||||||
Total Cash, cash equivalents, and restricted cash | $ | 89,050 | $ | 21,054 | $ | 16,992 | ||||||
| ||||||||||||
Supplemental disclosures of cash flow information: | ||||||||||||
Cash paid for income taxes | $ | 40 | $ | 13 | $ | 21 | ||||||
Cash paid for interest | $ | 0— | $ | 862 | $ | 1,499 | ||||||
(1) | See Note 8b. |
(2) | See Note 8f. |
(3) | See Note 10. |
The accompanying notes are an integral part of these consolidated financial statements.
F - 9
NOTE 1:-GENERAL
a.ReWalk Robotics Ltd. (“RRL”, and together with its subsidiaries, the “Company”) was incorporated under the laws of the State of Israel on June 20, 2001 and commenced operations on the same date.
b.RRL has two wholly owned subsidiaries: (i) ReWalk Robotics Inc. (“RRI”) incorporated under the laws of Delaware on February 15, 2012 and (ii) ReWalk Robotics GMBH. (“RRG”) incorporated under the laws of Germany on January 14, 2013.
c.The Company is designing, developing, and commercializing robotic exoskeletons that allow individuals with mobility impairments or other medical conditions the ability to stand and walk once again. The Company has developed and is continuing to commercialize the ReWalk, an exoskeleton designed for individuals with paraplegia that uses its patented tilt-sensor technology and an on-board computer and motion sensors to drive motorized legs that power movement. The ReWalk system consists of a light wearable brace support suit which integrates motors at the joints, rechargeable batteries, an array of sensors and a computer-based control system to power knee and hip movement. Additionally, the Company developed and, in June 2019, started to commercialize the ReStore following receipt of European Union CE mark and United States Food and Drug Administration (“FDA”). The ReStore is a powered, lightweight soft exo-suit intended for use in the rehabilitation of individuals with lower limb disability due to stroke. The Company markets and sells its products directly to institutions and individuals and through third-party distributors. The Company sells its products directly primarily in Germany and the United States, and primarily through distributors in other markets. In its direct markets, the Company has established relationships with rehabilitation centers and the spinal cord injury community, and in its indirect markets, the Company’s distributors maintain these relationships. RRI markets and sells products mainly in the United States. RRG markets and sells the Company’s products mainly in Germany and Europe.
During the second quarter of 2020, we have finalized two separate agreements to distribute additional product lines in the U.S. market. The Company will be the exclusive distributor of the MediTouch Tutor movement biofeedback systems in the United States and will also have distribution rights for the MYOLYN MyoCycle FES cycles to U.S. rehabilitation clinics and personal sales through the U.S. Department of Veterans Affairs (“VA”) hospitals. These new products will improve our product offering to clinics as well as patients within the VA as they both have similar clinician and patient profiles.
d.The Company depends on one contract manufacturer, Sanmina. Reliance on this vendor makes the Company vulnerable to possible capacity constraints and reduced control over component availability, delivery schedules, manufacturing yields and costs.
e.The worldwide spread of COVID-19 has resulted in a global economic slowdown and is expected to continue to disrupt general business operations until the disease is contained. This has had a negative impact on the Company's sales and results of operations since the start of the pandemic, and the Company expects that it will continue to negatively affect its sales and results of operations, but the Company is currently unable to predict the scale and duration of that impact. As of the date of issuance of these financial statements, the Company is not aware of any specific event or circumstance that would require an update of its accounting estimates or judgments or revision of the carrying value of its assets or liabilities. This determination may change as new events occur and additional information is obtained. Actual results could differ from our estimates and judgments, and any such differences may be material to our financial statements.
F - 10
f.For the full year ended December 31, 2021 the Company incurred a consolidated net loss of $12.7 million and has an accumulated deficit in the total amount of $194.2 million. The Company’s negative operating cash flow for the full year ended December 31, 2021 was $11.5 million. Our cash and cash equivalent on December 31, 2021 totaled $88.3 million. The Company has sufficient funds to support its operation for more than 12 months following the approval of its consolidated financial statements for the fiscal year ended December 31, 2021.
The Company expects to incur future net losses and our transition to profitability is dependent upon, among other things, the successful development and commercialization of the Company’s products and product candidates, the achievement of a level of revenues adequate to support the cost structure. Until the Company achieves profitability or generates positive cash flows, it will continue to need to raise additional cash. The Company intends to fund future operations through cash on hand, additional private and/or public offerings of debt or equity securities, cash exercises of outstanding warrants or a combination of the foregoing. In addition, the Company may seek additional capital through arrangements with strategic partners or from other sources and will continue to address its cost structure. Notwithstanding, there can be no assurance that the Company will be able to raise additional funds or achieve or sustain profitability or positive cash flows from operations.
NOTE 2:-SIGNIFICANT ACCOUNTING POLICIES
The consolidated financial statements are prepared according to United States generally accepted accounting principles (“U.S. GAAP”), applied on a consistent basis, as follows:
a.Use of Estimates
The preparation of the consolidated financial statements in conformity with U.S. generally accepted accounting principles requires management to make estimates, judgments, and assumptions. The Company’s management believes that the estimates, judgments, and assumptions used are reasonable based upon information available at the time they are made. These estimates, judgments and assumptions can affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the dates of the financial statements, and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates. On an ongoing basis, the Company’s management evaluates estimates, including those related to inventories, fair values of share-based awards and warrants, contingent liabilities, provision for warranty, allowance for doubtful account and sales return reserve. Such estimates are based on historical experience and on various other assumptions that are believed to be reasonable, the results of which form the basis for making judgments about the carrying values of assets and liabilities.
b.Financial Statements in U.S. Dollars:
Since 2015, most of the Company’s expenses were denominated in United States dollars (“dollars”) and the remaining expenses were denominated in New Israeli Shekel (“NIS”) and Euros. Until 2018 most of the Company’s revenues were denominated in U.S. dollars and the remainder of our revenues was denominated in Euros and British pound whereas in the last three years our Euro revenues are higher than the ones in dollars. However, the selling prices are linked to the Company’s price list which is determined in dollars, the budget is managed in dollars, financing activities including loans and fundraising activities, are made in U.S. dollars and the Company’s management believes that the dollar is the primary currency of the economic environment in which the Company and each of its subsidiaries operate. Thus, the dollar is the Company’s and its subsidiaries’ functional and reporting currency.
Accordingly, transactions denominated in currencies other than the functional currency are re-measured to the functional currency in accordance with Accounting Standards Codification (“ASC”) No. 830, “Foreign Currency Matters” at the exchange rate at the date of the transaction or the average exchange rate in the relevant reporting period. At the end of each reporting period, financial assets and liabilities are re-measured to the functional currency using exchange rates in effect at the balance sheet date. Non-financial assets and liabilities are re-measured at historical exchange rates. All transaction gains and losses of the re-measured monetary balance sheet items are reflected in the consolidated statements of operations.
F - 11
c.Principles of Consolidation:
The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries, RRI and RRG. All intercompany transactions and balances have been eliminated upon consolidation.
d.Cash Equivalents:
Cash equivalents are short-term highly liquid investments that are readily convertible to cash with original maturities of three months or less, at the date acquired.
e.Inventories:
Inventories are stated at the lower of cost or market value. Inventory reserves are provided to cover risks arising from slow-moving items or technological obsolescence.
The Company periodically evaluates the quantities on hand relative to historical, current and projected sales volume. Based on this evaluation, an impairment charge is recorded when required to write-down inventory to its market value.
Cost is determined as follows:
Finished products - on the basis of raw materials and manufacturing costs on an average basis.
Raw materials - The weighted average cost method.
The Company regularly evaluates the ability to realize the value of inventory based on a combination of factors, including historical usage rates and forecasted sales according to outstanding backlogs. Purchasing requirements and alternative usage are explored within these processes to mitigate inventory exposure. When recorded, the reserves are intended to reduce the carrying value of inventory to its net realizable value. In the years ended December 31, 2021, 2020 and 2019, the Company wrote off inventory in the amount of $252 thousand, $215 thousand, and $64 thousand, respectively. The write off inventory were recorded in cost of revenue. If actual demand for the Company’s products deteriorates, or market conditions are less favorable than those projected, additional inventory reserves may be required.
F - 12
f.Balances and transactions with related parties:
The Company has a related party shareholder named Yaskawa Electric Corporation (“YEC”).
In September 2013, the Company entered into a share purchase agreement and a strategic alliance with YEC, pursuant to which YEC has agreed to distribute the Company’s products, in addition to providing sales, marketing, service and training functions, in Japan, China (including Hong-Kong and Macau), Taiwan, South Korea, Singapore and Thailand.
As of December 31, 2021, and 2020, there have been no related party receivable with YEC . Revenues from YEC during the years ended December 31, 2021, 2020, and 2019 amounted to $0 thousand, $0 thousand and $41 thousand, respectively.
g.Property and Equipment:
Property and equipment are stated at cost, net of accumulated depreciation. Depreciation is calculated using the straight-line method over the estimated useful lives of the assets at the following annual rates:
% | |
Computer equipment | 20-33 (mainly 33) |
Office furniture and equipment | 6 - 10 (mainly 10) |
Machinery and laboratory equipment | 15 |
Field service units | 50 |
Leasehold improvements | Over the shorter of the lease term or estimated useful life |
h.Impairment of Long-Lived Assets:
The Company’s long-lived assets are reviewed for impairment in accordance with ASC No. 360, “Property, Plant and Equipment” whenever events or changes in circumstances indicate that the carrying amount of an asset (or asset group) may not be recoverable. Recoverability of assets (or asset group) to be held and used is measured by a comparison of the carrying amount of an asset to the future undiscounted cash flows expected to be generated by the assets. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the carrying amount of the assets exceeds the fair value of the assets. During the years ended December 31, 2021, 2020 and 2019, 000no impairment losses have been recorded.
i.Restricted cash and Other long-term assets:
Other long-term assets include long-term prepaid expenses and restricted cash deposits for offices and cars leasing based upon the term of the remaining restrictions.
j.Revenue Recognition:
The Company generates revenues from sales of products. The Company sells its products directly to end customers and through distributors. The Company sells its products to private individuals (who finance the purchases by themselves, through fundraising or reimbursement coverage from insurance companies), rehabilitation facilities and distributors.
F - 13
The Company recognized revenue in accordance with ASC Topic 606 when, or as, control of the promised goods or services is transferred to the customer, in an amount that reflects the consideration the Company expects to be entitled to in exchange for those goods or services. The Company applies the following five steps:
1. Identify the contract with a customer
The Company generally considers purchase order or a signed quote, to be contracts with customers. In evaluating the contract with a customer, the Company analyzes the customer’s intent and ability to pay the amount of promised consideration (credit risk) and considers the probability of collecting substantially all of the consideration.
2. Identify the performance obligations in the contract
At a contract’s inception, the Company assesses the goods or services promised in a contract with a customer and identifies the performance obligations.
3. Determine the transaction price
The transaction price is determined based on the consideration to which the Company will be entitled in exchange for transferring products or services to the customer.
Shipping and handling costs charged to customers are included in net sales. Determining the transaction price requires significant judgment, which is discussed by revenue category in further detail below.
In practice, the Company does not offer extended payment terms beyond one year to customers.
F - 14
4. Allocate the transaction price to performance obligations in the contract
If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. Contracts that contain multiple performance obligations require an allocation of the transaction price to each performance obligation based on a relative standalone selling price basis unless a portion of the variable consideration related to the contract is allocated entirely to a performance obligation. The Company determines standalone selling price based on the price at which the performance obligation is sold separately.
5. Recognize revenue when or as the Company satisfies a performance obligation
The Company generally satisfies performance obligations at a point in time, once the customer has obtained the legal title to the items purchased or service provided.
For systems sold to rehabilitation facilities, the Company includes training and considers the elements in the arrangement to be a single performance obligation. Therefore, the Company recognizes revenue for the system and training only after delivery in accordance with the agreement's delivery terms to the customer and after the training has been completed.
For sales of Personal systems to end users, and for sales of Personal or Rehabilitation systems to third party distributors, the Company does not provide training to the end user as this training is completed by the Rehabilitation centers or by the distributor that have previously completed the ReWalk Training program. Therefore, the Company recognizes revenue in such sales upon delivery.
Revenue is recognized based on the transaction price at the time the related performance obligation is satisfied by transferring a promised product or service to a customer.
The Company generally does not grant a right of return for its products. In rare circumstances the Company provides a right of return of its products. In those cases, the Company records reductions to revenue for expected future product returns based on the Company’s historical experience and estimates.
Disaggregation of Revenues (in thousands)
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Units placed | $ | 5,449 | $ | 3,620 | $ | 4,385 | ||||||
Spare parts and warranties | 517 | 773 | 488 | |||||||||
Total Revenues | $ | 5,966 | $ | 4,393 | $ | 4,873 | ||||||
Units placed
The Company currently offer five products: (1) ReWalk Personal, (2) ReWalk Rehabilitation, (3) ReStore, (4) MyoCycle and (5) MediTouch.
ReWalk Personal and ReWalk Rehabilitation are units for spinal cord injuries (“SCI Products”). SCI Products are currently designed for everyday use by paraplegic individuals at home and in their communities, and are custom fitted for each user, as well as for use by paraplegic patients in the clinical rehabilitation environment, where they provide individuals access to valuable exercise and therapy. ReWalk Rehabilitation current design is dated and will not be produced in the future.
F - 15
ReStore is a powered, lightweight soft exo-suit intended for use in the rehabilitation of individuals with lower limb disability due to stroke in the clinical rehabilitation environment.
MyoCycle which uses Functional Electrical Stimulation (“FES”) technology and MediTouch tutor movement biofeedback devices (“Distributed Products”). The Company markets the Distributed Products in the United States for use at home or in clinic.
Units placed include revenue from sales of SCI Products, ReStore, and Distributed Products.
For units placed, the Company recognizes revenues when it transfers control and title has passed to the customer. Each unit placed is considered an independent, unbundled performance obligation. The Company also offers a rent-to-purchase model in which the Company recognizes revenue ratably according to the agreed rental monthly fee.
Spare parts and warranties
Spare parts are sold to private individuals, rehabilitation facilities and distributors. Revenue is recognized when the Company satisfies a performance obligation by transferring control over promised goods or services to the customer. Each part sold is considered an independent, unbundled performance obligation.
Warranties are classified as either assurance type or service type warranty. A warranty is considered an assurance type warranty if it provides the consumer with assurance that the product will function as intended for a limited period of time.
In the beginning of 2018, the Company updated its service policy for SCI Products to include a five- year warranty compared to a period of two years that were included in the past for parts and services. The first two years are considered as assurance type warranty and the additional period is considered an extended service arrangement, which is a service type warranty. An assurance type warranty is not accounted for as separate performance obligations under the revenue model. A service type warranty is either sold with a unit or separately for units for which the warranty has expired. Revenue is then recognized ratably over the life of the warranty.
The ReStore device is sold with a two-year warranty which is considered as assurance type warranty.
The Distributed Products are sold with assurance type warranty ranging between one year to ten years depending on the specific product and part.
Contract balances (in thousands)
December 31, | December 31, | |||||||
2021 | 2020 | |||||||
Trade receivable, net (1) | $ | 585 | $ | 684 | ||||
Deferred revenues (1) (2) | $ | 1,182 | $ | 1,108 | ||||
(1) | Balance presented net of unrecognized revenues that were not yet collected. |
| |
(2) | $432 thousands of December 31, 2020 deferred revenues balance were recognized as revenues during the year ended December 31, 2021. |
Typical timing of payment
Deferred revenue is comprised mainly of unearned revenue related to service type warranty but also includes other offerings for which the Company has been paid in advance and earns revenue when the Company transfers control of the product or service.
F - 16
The Company's unfilled performance obligations as of December 31, 2021 and the estimated revenue expected to be recognized in the future related to the service type warranty amounts to $1.21 million, which is fulfilled over one to five years.
k.Accounting for Share-Based Compensation:
The Company accounts for share-based compensation in accordance with ASC No. 718, “Compensation-Stock Compensation” (“ASC No. 718”). ASC No. 718 requires companies to estimate the fair value of equity-based payment awards on the date of grant using an Option-Pricing Model (“OPM”). The value of the portion of the award that is ultimately expected to vest is recognized as an expense over the requisite service periods in the Company’s consolidated statements of operations. According to Accounting Standards Update 2016-09, “Compensation-Stock Compensation (Topic 718)” (“ASU 2016-09”) the Company account for forfeitures as they occur.
The Company recognizes compensation expenses for the value of its awards granted based on the straight-line method over the requisite service period of each of the awards.
The Company selected the Black-Scholes-Merton option pricing model as the most appropriate fair value method for its share-option awards. The option-pricing model requires a number of assumptions, of which the most significant are the fair market value of the underlying ordinary share, expected share price volatility and the expected option term. Expected volatility was calculated based upon certain peer companies that the Company considered to be comparable. The expected option term represents the period of time that options granted are expected to be outstanding. The expected option term is determined based on the simplified method in accordance with Staff Accounting Bulletin No. 110, as adequate historical experience is not available to provide a reasonable estimate. The simplified method will continue to apply until enough historical experience is available to provide a reasonable estimate of the expected term. The risk-free interest rate is based on the yield from U.S. treasury bonds with an equivalent term. The Company has historically not paid dividends and has no foreseeable plans to pay dividends.
F - 17
Following the IPO in September 2014, the fair value of ordinary shares is observable as they are publicly traded.
The fair value of Restricted Stock Units (RSUs) granted is determined based on the price of the Company’s ordinary shares on the date of grant.
The fair value for options granted in 2019 is estimated at the date of grant using a Black-Scholes-Merton option pricing model with the following assumptions:
2019 | ||||
Expected volatility | 57.5 | % | ||
Risk-free rate | 2.22 | % | ||
Dividend yield | 0— | % | ||
Expected term (in years) | 6.11 | |||
Share price | $ | 5.37 | ||
There were no options granted during the twelve months ended December 31, 2021, and 2020.
The Company accounts for options granted to consultants and other service providers under ASC No. 718. The fair value of these options was estimated using a Black-Scholes-Merton option-pricing model.
The non-cash compensation expenses related to employees and non-employees for the years ended December 31, 2021, 2020 and 2019 amounted to $833 thousand, $749 thousand, and $1.11 million, respectively.
l.Warrants to Acquire Ordinary Shares:
During the twelve-month ended December 31, 2021, and 2020, respectively, the Company issued warrants to acquire up to 15,083,611 and 11,389,555 ordinary shares. The Company assessed the warrants pursuant to ASC 480 "Distinguishing Liabilities from Equity" and ASC 815 "Derivatives and Hedging" and determine that the warrants should be accounted for as equity and not as a derivative liability. Refer to Note 8f for additional information.
m.Research and Development Costs:
Research and development costs are charged to the consolidated statement of operations as incurred and are presented net of the amount of any grants the Company received for research and development in the period in which the grant was received.
n.Income Taxes
The Company accounts for income taxes in accordance with ASC No. 740, “Income Taxes” (“ASC No. 740”), using the liability method whereby deferred tax assets and liability account balances are determined based on the differences between financial reporting and the tax basis for assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. The Company provides a valuation allowance, if necessary, to reduce deferred tax assets to the amounts that are more likely-than-not to be realized.
ASC No. 740 contains a two-step approach to recognizing and measuring a liability for uncertain tax positions. The first step is to evaluate the tax position taken or expected to be taken in a tax return by determining if the weight of available evidence indicates that it is more likely than not that, on an evaluation of the technical merits, the tax position will be sustained on audit, including resolution of any related appeals or litigation processes. The second step is to measure the tax benefit as the largest amount that is more than 50% likely to be realized upon ultimate settlement. The Company accrues interest and penalties related to unrecognized tax benefits in its taxes on income. As of December 31, 2021, and 2020, the Company did not identify any significant uncertain tax positions.
F - 18
o.Warranty:
The Company provided a two-year standard warranty for its products. In the beginning of 2018, we updated our service policy for new devices sold to include five-year warranties. The Company determined that the first two years of warranty is an assurance-type warranty and records a provision for the estimated cost to repair or replace products under warranty at the time of sale. Factors that affect the Company’s warranty reserve include the number of units sold, historical and anticipated rates of warranty repairs and the cost per repair.
US Dollars in thousands | ||||
Balance at December 31, 2020 | $ | 140 | ||
Provision | 271 | |||
Usage | (299 | ) | ||
Balance at December 31, 2021 | 112 | |||
p.Concentrations of Credit Risks:
Financial instruments that potentially subject the Company to concentrations of credit risk consist principally of cash, cash equivalents and trade receivables.
The Company’s cash and cash equivalents are deposited in major banks in Israel, the United States and Germany. Such deposits in the United States may be in excess of insured limits and are not insured in other jurisdictions. The Company maintains cash and cash equivalents with diverse financial institutions and monitors the amount of credit exposure to each financial institution.
Concentration of credit risk with respect to trade receivable is primarily limited to a customer to which the Company makes substantial sales.
December 31, | ||||||||
2021 | 2020 | |||||||
Customer A | 20 | % | 0* | ) | ||||
Customer B | 18 | % | 0* | ) | ||||
Customer C | 16 | % | 0* | ) | ||||
Customer D | 12 | % | 12 | % | ||||
Customer E | 10 | % | 0* | ) | ||||
Customer F | 0* | ) | 15 | % | ||||
Customer G | 0* | ) | 15 | % | ||||
Customer H | 0* | ) | 15 | % | ||||
Customer I | 0* | ) | 14 | % | ||||
Customer J | 0* | ) | 11 | % | ||||
*) | Less than 10% |
The Company’s trade receivables are geographically diversified and derived primarily from sales to customers in various countries, mainly in the United States and Europe. Concentration of credit risk with respect to trade receivables is limited by credit limits, ongoing credit evaluation and account monitoring procedures. The Company performs ongoing credit evaluations of its distributors based upon a specific review of all significant outstanding invoices. The Company writes off receivables when they are deemed uncollectible and having exhausted all collection efforts. As of December 31, 2021, and 2020 trade receivables are presented net of $42 thousand and $102 thousand allowance for doubtful accounts, respectively, and net of sales return reserve of $43 thousand and $0 thousand, respectively.
F - 19
q.Accrued Severance Pay:
Pursuant to Israel’s Severance Pay Law, Israeli employees are entitled to severance pay equal to one month’s salary for each year of employment, or a portion thereof. All of the employees of the RRL elected to be included under section 14 of the Severance Pay Law, 1963 (“section 14”). According to this section, these employees are entitled only to monthly deposits, at a rate of 8.33% of their monthly salary, made in their name with insurance companies. Payments in accordance with section 14 release the Company from any future severance payments (under the above Israeli Severance Pay Law) in respect of those employees; therefore, related assets and liabilities are not presented in the balance sheet.
Total Company's expenses related to severance pay amounted to $104 thousand, $125 thousand and $156 thousand for the years ended December 31, 2021, 2020 and 2019, respectively.
r.Fair Value Measurements:
Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The Company uses a three-tier fair value hierarchy to classify and disclose all assets and liabilities measured at fair value on a recurring basis, as well as assets and liabilities measured at fair value on a non-recurring basis, in periods subsequent to their initial measurement. The hierarchy requires the Company to use observable inputs when available, and to minimize the use of unobservable inputs when determining fair value. If a financial instrument uses inputs that fall in different levels of the hierarchy, the instrument will be categorized based upon the lowest level of input that is significant to the fair value calculation. The three-tiers are defined as follows:
•
Level 1. Observable inputs based on unadjusted quoted prices in active markets for identical assets or liabilities;
•
Level 2. Inputs, other than quoted prices in active markets, that are observable either directly or indirectly; and
•
Level 3. Unobservable inputs for which there is little or no market data requiring the Company to develop its own assumptions.
The carrying amounts of cash and cash equivalents, short term deposits, trade receivables and trade payables approximate their fair value due to the short-term maturity of such instruments.
F - 20
s.Basic and Diluted Net Loss Per Share:
Basic net loss per share is computed by dividing the net loss by the weighted-average number of shares of ordinary shares outstanding during the period.
Diluted net loss per share is computed by giving effect to all potential shares of ordinary shares, including stock options, convertible preferred share warrants, to the extent dilutive, all in accordance with ASC No. 260, “Earning Per Share”.
The following table sets forth the computation of the Company’s basic and diluted net loss per ordinary share (in thousands, except share and per share data):
Year ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Net loss | $ | (12,736 | ) | $ | (12,976 | ) | $ | (15,551 | ) | |||
| ||||||||||||
Net loss attributable to ordinary shares | (12,736 | ) | (12,976 | ) | (15,551 | ) | ||||||
Shares used in computing net loss per ordinary shares, basic and diluted | 47,935,652 | 15,764,980 | 5,763,317 | |||||||||
| ||||||||||||
Net loss per ordinary share, basic and diluted | $ | (0.27 | ) | $ | (0.82 | ) | $ | (2.70 | ) | |||
Basic and diluted net loss per share was the same for each period presented as the inclusion of all potential shares of ordinary shares and warrants outstanding would have been anti-dilutive.
t.Contingent liabilities
The Company accounts for its contingent liabilities in accordance with ASC No. 450, “Contingencies”. A provision is recorded when it is both probable that a liability has been incurred and the amount of the loss can be reasonably estimated.
With respect to legal matters, provisions are reviewed and adjusted to reflect the impact of negotiations, estimated settlements, legal rulings, advice of legal counsel and other information and events pertaining to a particular matter.
See Note 7e for further information.
u.Government grants
Government grants received by the Company relating to categories of operating expenditures are credited to the consolidated statements of operations during the period in which the expenditure to which they relate is charged. Royalty and non-royalty-bearing grants from the Israel Innovation Authority, or the IIA, (formerly known as the Israeli Office of the Chief Scientist), for funding certain approved research and development projects which are recognized at the time when the Company is entitled to such grants, on the basis of the related costs incurred, and are included as a deduction from research and development expenses (see Note 7c).
000No royalty-bearing grants were recorded for the years ended December 31, 2021, 2020, and 2019.
Total Company expenses related to royalties amounted to $14 thousand, $46 thousand and $15 thousand for the years ended December 31, 2021, 2020 and 2019, respectively.
F - 21
v.Leases
In February 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standard Update (“ASU”), No. 2016-02, Leases (Topic 842), to enhance the transparency and comparability of financial reporting related to leasing arrangements. The Company adopted the standard effective January 1, 2019.
At the inception of an arrangement, the Company determines whether the arrangement is or contains a lease based on the unique facts and circumstances present. Operating lease liabilities and their corresponding right-of-use assets are recorded based on the present value of lease payments over the expected lease term. The interest rate implicit in lease contracts is typically not readily determinable. As such, the Company utilizes its incremental borrowing rate, which is the rate incurred to borrow on a collateralized basis over a similar term an amount equal to the lease payments in a similar economic environment. Certain adjustments to the right-of-use asset may be required for items, such as initial direct costs paid or incentives received.
Lease expense is recognized over the expected lease term on a straight-line basis. Operating leases are recognized on the balance sheet as right-of-use assets, lease liabilities current and lease liabilities non-current. As a result, the Company no longer recognizes deferred rent on the balance sheet.
Leases with an initial term of 12 months or less are not recorded on the balance sheet. The Company recognizes the lease expense for such leases on a straight-line basis over the lease term.
F - 22
w.New Accounting Pronouncements
Recent Accounting Pronouncements Not Yet Adopted
i.Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity
In August 2020, the Financial Accounting Standards Board (“FASB”) issued ASU No. 2020-06, Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity (“ASU 2020-06”), which simplifies the accounting for certain financial instruments with characteristics of liabilities and equity, including convertible instruments and contracts in an entity’s own equity. Among other changes, ASU 2020-06 removes from U.S. GAAP the liability and equity separation model for convertible instruments with a cash conversion feature and a beneficial conversion feature, and as a result, after adoption, entities will no longer separately present in equity an embedded conversion feature for such debt. Similarly, the embedded conversion feature will no longer be amortized into income as interest expense over the life of the instrument. Instead, entities will account for a convertible debt instrument wholly as debt unless (1) a convertible instrument contains features that require bifurcation as a derivative under ASC Topic 815, Derivatives and Hedging, or (2) a convertible debt instrument was issued at a substantial premium. Additionally, ASU 2020-06 requires the application of the if-converted method to calculate the impact of convertible instruments on diluted earnings per share (“EPS”). ASU 2020-06 is effective for fiscal years beginning after December 15, 2021, with early adoption permitted for fiscal years beginning after December 15, 2020 and can be adopted on either a fully retrospective or modified retrospective basis. The adoption of this standard is not expected to result in a material impact to the Company’s financial statements.
ii.Financial Instruments
In June 2016, FASB issued ASU 2016-13, Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments. ASU 2016-13 amends the impairment model to utilize an expected loss methodology in place of the currently used incurred loss methodology, which will result in the more timely recognition of losses. Topic 326 will be effective on the Company beginning on January 1, 2023. The Company is currently evaluating the impact of this new standard on its financial statements.
iii.Income Taxes
In December 2019, the FASB issued ASU 2019-12 to simplify the accounting for income taxes. The guidance eliminates certain exceptions related to the approach for intraperiod tax allocation, the methodology for calculating income taxes in an interim period, and the recognition of deferred tax liabilities for outside basis differences related to changes in ownership of equity method investments and foreign subsidiaries.
The guidance also simplifies aspects of accounting for franchise taxes and enacted changes in tax laws or rates and clarifies the accounting for transactions that result in a step-up in the tax basis of goodwill. The standard will be effective for the Company beginning January 1, 2022. The adoption of ASU 2019-12 is not expected to result in a material impact on the Company's consolidated financial statements.
NOTE 3:-PREPAID EXPENSES AND OTHER CURRENT ASSETS
The components of prepaid expenses and other current assets are as follows (in thousands):
December 31, | ||||||||
2021 | 2020 | |||||||
Government institutions | $ | 207 | $ | 87 | ||||
Prepaid expenses | 335 | 311 | ||||||
Advances to vendors | 5 | 241 | ||||||
Other assets | 63 | 33 | ||||||
| ||||||||
$ | 610 | $ | 672 | |||||
NOTE 4:-INVENTORIES
The components of inventories are as follows (in thousands):
December 31, | ||||||||
2021 | 2020 | |||||||
Finished products | $ | 2,284 | $ | 2,764 | ||||
Raw materials | 705 | 778 | ||||||
| ||||||||
$ | 2,989 | $ | 3,542 | |||||
F - 23
NOTE 5:-PROPERTY AND EQUIPMENT, NET
The components of property and equipment, net are as follows (in thousands):
December 31, | ||||||||
2021 | 2020 | |||||||
Cost: | ||||||||
Computer equipment | $ | 741 | $ | 725 | ||||
Office furniture and equipment | 301 | 298 | ||||||
Machinery and laboratory equipment | 620 | 612 | ||||||
Field service units | 1,712 | 1,626 | ||||||
Leasehold improvements | 333 | 333 | ||||||
| ||||||||
$ | 3,707 | $ | 3,594 | |||||
December 31, | ||||||||
2021 | 2020 | |||||||
Accumulated depreciation | 3,423 | 3,157 | ||||||
| ||||||||
Property and equipment, net | $ | 284 | $ | 437 | ||||
Depreciation expenses amounted to $266 thousand, $285 thousand, and $321 thousand for the years ended December 31, 2021, 2020 and 2019, respectively.
F - 24
NOTE 6: -LOAN AGREEMENT WITH KREOS AND RELATED WARRANT TO PURCHASE ORDINARY SHARES
On December 30, 2015, the Company entered into the loan agreement (the “Loan Agreement”) with Kreos Capital V (Expert Fund) Limited (“Kreos”), pursuant to which Kreos extended a line of credit to us in the amount of $20 million, with interest payable monthly in arrears on any amounts drawn down at a rate of 10.75% per year from the applicable drawdown date through the date on which all principal is repaid. As of June 30, 2017, the Company raised more than $20 million in connection with the issuance of its share capital and, therefore, in accordance with the terms of the Loan Agreement, the repayment period was extended from 24 months to 36 months. The principal was also reduced in connection with the issuance of the Kreos Convertible Note on June 9, 2017. Pursuant to the Loan Agreement, we granted Kreos a first priority security interest over all of our assets, including certain intellectual property and equity interests in its subsidiaries, subject to certain permitted security interests.
Pursuant to the terms of the warrant, in connection with the $20.0 million drawdown under the Loan Agreement on January 4, 2016, we issued to Kreos the warrant to purchase up to 4,771 of our ordinary shares at an exercise price of $241.0 per share, increased to 6,679 ordinary shares on December 28, 2016. Subject to the terms of the warrant, the warrant is exercisable, in whole or in part, at any time prior to the earlier of (i) December 30, 2025, or (ii) immediately prior to the consummation of a merger, consolidation, or reorganization of us with or into, or the sale or license of all or substantially all our assets or shares to, any other entity or person, other than a wholly-owned subsidiary of us, excluding any transaction in which our shareholders prior to the transaction will hold more than 50% of the voting and economic rights of the surviving entity after the transaction.
On June 9, 2017, the Company and Kreos entered into the First Amendment, under which $3.0 million of the outstanding principal under the Loan Agreement became subject to repayment pursuant to the senior secured Kreos Convertible Note issued on June 9, 2017.
On November 20, 2018, the Company and Kreos entered into the Second Amendment of the Loan Agreement, in which the Company repaid Kreos the $3.6 million other related payments, including prepayment costs and end of loan payments, terminating the Kreos Note, by issuing to Kreos 192,000 units and 288,000 pre-funded units as part of an underwritten public offering at the public offering prices, and the parties agreed to revise the principal and the repayment schedule under the Kreos Loan. Additionally, Kreos and the Company entered into the Kreos Warrant Amendment, which amended the exercise price of the warrant to purchase 6,679 ordinary shares currently held by Kreos from $241.0 to $7.50.
On June 5, 2019, and June 6, 2019, the Company entered into warrant exercise agreements with certain institutional investors of warrants to purchase the Company’s ordinary shares, pursuant to which, Kreos agreed to exercise in cash their November 2018 warrants at the existing exercise price of $7.50 per share. Under the exercise agreements, the Company also agreed to issue to Kreos new warrants to purchase up to 480,000 ordinary shares at an exercise price of $7.50 per share and exercise period of five years.
On December 29, 2020, the Company repaid in full the remaining loan principal amount to Kreos including the end of loan payments, and by that discharged all of its obligations to Kreos and as of December 31, 2020, the outstanding principal amount under the Kreos Loan Agreement was zero.
The Company recorded interest expense in the amount of $907 thousand during the fiscal year ended December 31, 2020.
F - 25
NOTE 7:-COMMITMENTS AND CONTINGENT LIABILITIES
a.Purchase commitment:
The Company has contractual obligations to purchase goods from its contract manufacturer as well as raw materials from different vendors. Purchase obligations do not include contracts that may be canceled without penalty. As of December 31, 2021, non-cancelable outstanding obligations amounted to approximately $1.5 million.
b.Operating lease commitment:
(i)The Company operates from leased facilities in Israel, the United States and Germany. These leases expire between 2022 and 2023. A portion of the Company’s facilities leases is generally subject to annual changes in the Consumer Price Index (CPI). The changes to the CPI are treated as variable lease payments and recognized in the period in which the obligation for those payments was incurred.
(ii)RRL and RRG lease cars for their employees under cancelable operating lease agreements expiring at various dates in between 2022 and 2023. A subset of the Company’s cars leases is considered variable. The variable lease payments for such cars leases are based on actual mileage incurred at the stated contractual rate. RRL and RRG have an option to be released from these agreements, which may result in penalties in a maximum amount of approximately $20 thousand as of December 31, 2021.
The Company’s future lease payments for its facilities and cars, which are presented as current maturities of operating leases and non-current operating leases liabilities on the Company’s consolidated balance sheets as of December 31, 2021 are as follows (in thousands):
2022 | $ | 689 | ||
2023 | 500 | |||
Total lease payments | 1,189 | |||
Less: imputed interest | (130 | ) | ||
Present value of future lease payments | 1,059 | |||
Less: current maturities of operating leases | (641 | ) | ||
Non-current operating leases | $ | 418 | ||
Weighted-average remaining lease term (in years) | 1.74 | |||
Weighted-average discount rate | 12.6 | % |
Total rent expenses for the years ended December 31, 2021, 2020 and 2019 were $730 thousand, $764 thousand, and $739 thousand, respectively.
F - 26
c.Royalties:
The Company's research and development efforts are financed, in part, through funding from the IIA. Since the Company's inception through December 31, 2021, the Company received funding from the IIA in the total amount of $1.97 million. Out of the $1.97 million in funding from the IIA, a total amount of $1.57 million were royalty-bearing grants (as of December 31, 2021, the Company paid royalties to the IIA in the total amount of $99 thousand), while a total amount of $400 thousand was received in consideration of 209 convertible preferred A shares, which converted after the Company's initial public offering in September 2014 into ordinary shares in a conversion ratio of 1 to 1. The Company is obligated to pay royalties to the IIA, amounting to 3% of the sales of the products and other related revenues generated from such projects, up to 100% of the grants received. The royalty payment obligations also bear interest at the LIBOR rate. The obligation to pay these royalties is contingent on actual sales of the applicable products and in the absence of such sales, no payment is required.
Additionally, the License Agreement requires the Company to pay Harvard royalties on net sales, see Note 9 below for more information about the Collaboration Agreement and the License Agreement.
Royalties expenses in cost of revenue were $14 thousand, $46 thousand and $15 thousand for the years ended December 31, 2021, 2020 and 2019, respectively.
As of December 31, 2021, the contingent liability to the IIA amounted to $1.5 million. The Israeli Research and Development Law provides that know-how developed under an approved research and development program may not be transferred to third parties without the approval of the IIA. Such approval is not required for the sale or export of any products resulting from such research or development. The IIA, under special circumstances, may approve the transfer of IIA-funded know-how outside Israel, in the following cases:
(a) the grant recipient pays to the IIA a portion of the sale price paid in consideration for such IIA-funded know-how or in consideration for the sale of the grant recipient itself, as the case may be, which portion will not exceed six times the amount of the grants received plus interest (or three times the amount of the grant received plus interest, in the event that the recipient of the know-how has committed to retain the R&D activities of the grant recipient in Israel after the transfer); (b) the grant recipient receives know-how from a third party in exchange for its IIA-funded know-how; (c) such transfer of IIA-funded know-how arises in connection with certain types of cooperation in research and development activities; or (d) If such transfer of know-how arises in connection with a liquidation by reason of insolvency or receivership of the grant recipient.
d.Liens
As part of the Company’s Restricted cash and other long-term assets, as of December 31, 2021, an amount of $713 thousand has been pledged as security in respect of a guarantee granted to a third party. Such deposit cannot be pledged to others or withdrawn without the consent of such third party.
F - 27
e.Legal Claims:
Occasionally, the Company is involved in various claims such as product liability claims, lawsuits, regulatory examinations, investigations, and other legal matters arising, for the most part, in the ordinary course of business. While the outcome of any pending or threatened litigation and other legal matters is inherently uncertain, the Company does not believe the outcome of any of the matters will have a material adverse effect on the Company’s consolidated results of operation, liquidity or financial condition.
F - 28
NOTE 8: -SHAREHOLDERS’ EQUITY
a.Reverse share split:
On March 27, 2019, the Company’s shareholders approved (i) a reverse share split within a range of 1:8 to 1:32, to be effective at the ratio and on a date to be determined by the Board of Directors, and (ii) amendments to the Company’s Articles of Association authorizing an increase in the Company’s authorized share capital (and corresponding authorized number of ordinary shares, proportionally adjusting such number for the reverse share split) by up to NIS 17.5 million. Following the shareholder approval, an authorized committee of the Board of Directors of the Company approved a one-for-twenty-five reverse share split of the Company’s ordinary shares, and the Company filed the Third Amended and Restated Articles of Association of the Company with the Israeli Corporations Authority to affect the reverse share split and to increase the Company’s authorized share capital after the effect of the reverse share split. The reverse share split became effective on April 1, 2019. Additionally, effective at the same time, the total number of ordinary shares the Company is authorized to issue changed from 250,000,000 shares to 60,000,000 shares, the par value per share of the ordinary shares changed to NIS 0.25 and the authorized share capital of the Company changed from NIS 2,500,000 to NIS 15,000,000. All share and per share data included in these consolidated financial statements, for periods before December 31, 2019, give retroactive effect to the reverse stock split.
Upon the effectiveness of the reverse share split, every twenty-five shares were automatically combined and converted into one ordinary share. Appropriate adjustments were also made to all outstanding derivative securities of the Company, including all outstanding equity awards and warrants.
No fractional shares were issued in connection with the reverse share split. Instead, all fractional shares (including shares underlying outstanding equity awards and warrants) were rounded down to the nearest whole number.
F - 29
b.Equity raise:
1.Follow-on offerings
In February 2019, the Company entered into an exclusive placement agent agreement with H.C. Wainwright, on a reasonable best-efforts basis in connection with a public offering of 760,000 ordinary shares at a price of $5.75 per share.
The total gross proceeds received from the February 2019 follow-on public offering, before deducting commissions, discounts, and expenses, were $4.37 million. The Company also issued to H.C Wainwright and/or its designees warrants to purchase up to 45,600 ordinary shares, which are immediately exercisable starting on February 25, 2019, until February 21, 2024 at $7.1875 per share.
In April 2019, the Company entered into securities purchase agreements with certain institutional purchasers whereby the Company issued 816,914 ordinary shares at $5.2025 per ordinary share and warrants to purchase up to 408,457 ordinary shares with an exercise price of $5.14 per share, exercisable from April 5, 2019 until October 7, 2024, in a private placement that took place concurrently with the Company’s registered direct offering of ordinary shares in April 2019. Additionally, the Company issued warrants to purchase up to 49,015 ordinary shares, with an exercise price of $6.503125 per share, exercisable from April 5, 2019 until April 3, 2024, to representatives of H.C. Wainwright as compensation for its role as the placement agent in the Company’s April 2019 registered direct offering and concurrent private placement of warrants.
On June 5, 2019, and June 6, 2019, the Company entered into warrant exercise agreements with certain institutional investors whereby the Company issued warrants to purchase up to 1,464,665 ordinary shares with an exercise price of $7.50 per share, exercisable from June 5, 2019 or June 6, 2019 until June 5, 2024 or June 6, 2024, respectively. Additionally, the Company issued warrants to purchase up to 87,880 ordinary shares, with an exercise price of $9.375 per share, exercisable from June 5, 2019, until June 5, 2024, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in the Company’s June 2019 warrant exercise agreement and concurrent private placement of warrants.
On June 12, 2019, the Company entered into a purchase agreement with certain institutional investors for the issuance and sale of 833,334 ordinary shares, par value NIS 0.25 per share at $6.00 per ordinary share and warrants to purchase up to 416,667 ordinary shares with an exercise price of $6.00 per share, exercisable from June 12, 2019 until December 12, 2024, in a private placement that took place concurrently with the Company’s registered direct offering of ordinary shares in June 2019. Additionally, the Company issued warrants to purchase up to 50,000 ordinary shares, with an exercise price of $7.50 per share, exercisable from June 12, 2019, until June 10, 2024, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in the Company’s June 2019 registered direct offering and concurrent private placement of warrants.
On February 10, 2020, the Company closed a “best efforts” public offering whereby the Company issued an aggregate of 5,600,000 of common units and pre-funded units at a public offering price of $1.25 per common unit and $1.249 per pre-funded unit. As part of the public offering, the Company entered into a securities purchase agreement with certain institutional purchasers. Each common unit consisted of one ordinary share, par value NIS 0.25 per share, and one common warrant to purchase one ordinary share. Each of the 1,546,828 pre-funded unit consisted of one pre-funded warrant to purchase one ordinary share and one common warrant. Additionally, the Company issued warrants to purchase up to 336,000 ordinary shares, with an exercise price of $1.5625 per share, to representatives of H.C. Wainwright as compensation for its role as the placement agent in the Company’s February 2020 offering. During the three months ended March 31, 2020, all pre-funded warrants to purchase ordinary shares were exercised. As of December 31, 2021, a total of 5,571,600 common warrants to purchase ordinary shares were exercised, additionally 230,160 common warrants to purchase ordinary shares were exercised to representatives of H.C. Wainwright.
F - 30
On July 6, 2020, the Company entered into a purchase agreement with certain institutional investors for the issuance and sale of (i) 4,938,278 ordinary shares, par value NIS 0.25 per share, at a price of $1.8225 per ordinary share and (ii) warrants to purchase up to 2,469,139 ordinary shares with an exercise price of $1.76 per share, exercisable from July 6, 2020, until January 6, 2026. Additionally, the Company issued warrants to purchase up to 296,297 ordinary shares, with an exercise price of $2.2781 per share, exercisable from July 6, 2020, until July 2, 2025, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in its July 2020 registered direct offering. As of December 31, 2021, a total of 2,020,441 common warrants to purchase ordinary shares were exercised.
On December 3, 2020, the Company entered into a purchase agreement with certain institutional investors for the issuance and sale of (i) 5,579,776 ordinary shares, par value NIS 0.25 per share, at a price of $1.4337 per ordinary share and (ii) warrants to purchase up to 4,184,832 ordinary shares with an exercise price of $1.34 per share, exercisable from December 8, 2020, until June 8, 2026. Additionally, the Company issued warrants to purchase up to 334,787 ordinary shares, with an exercise price of $1.7922 per share, exercisable from December 8, 2020, until June 8, 2026, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in its December 2020 registered direct offering. As of December 31, 2021, a total of 3,598,072 common warrants to purchase ordinary shares were exercised, additionally 225,981 common warrants to purchase ordinary shares were exercised to representatives of H.C. Wainwright.
On February 19, 2021, the Company entered into a purchase agreement with certain institutional and other accredited investors for the issuance and sale of 10,921,502 ordinary shares, par value NIS 0.25 per share at $3.6625 per ordinary share and warrants to purchase up to an aggregate of 5,460,751 ordinary shares with an exercise price of $3.6 per share, exercisable from February 19, 2021, until August 26, 2026. Additionally, the Company issued warrants to purchase up to 655,290 ordinary shares, with an exercise price of $4.578125 per share, exercisable from February 19, 2021, until August 26, 2026, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in our February 2021 private placement offering.
On September 27, 2021, the Company signed a purchase agreement with certain institutional investors for the issuance and sale of 15,403,014 ordinary shares, par value NIS 0.25 per share, pre-funded warrants to purchase up to an aggregate of 610,504 ordinary shares and ordinary warrants to purchase up to an aggregate of 8,006,759 ordinary shares at an exercise price of $2.00 per share. The Pre-Funded Warrants have an exercise price of $0.001 per Ordinary Share and are immediately exercisable and can be exercised at any time after their original issuance until such pre-funded warrants are exercised in full. Each ordinary shares was sold at an offering price of $2.035 and each pre-funded warrant was sold at an offering price of $2.034 (equal to the purchase price per ordinary share minus the exercise price of the pre-funded warrant). The offering of the ordinary shares, the pre-funded warrants and the ordinary shares that are issuable from time to time upon exercise of the pre-funded warrants was made pursuant to the Company's shelf registration statement on Form S-3 initially filed with the Securities and Exchange Commission (“SEC”) on May 9, 2019, and declared effective by the SEC on May 23, 2019, and the ordinary warrants were issued in a concurrent private placement. The ordinary warrants are exercisable at any time and from time to time, in whole or in part, following the date of issuance and ending five and one-half years from the date of issuance. All of the pre-funded warrants were exercised in full on September 27, 2021, and the offering closed on September 29, 2021. Additionally, the Company issued warrants to purchase up to 960,811 ordinary shares, with an exercise price of $2.5438 per share, exercisable from September 27, 2021, until September 27, 2026, to certain representatives of H.C. Wainwright as compensation for its role as the placement agent in our September 2021 registered direct offering.
During the twelve months ended December 31, 2021, we received a total of 9,814,754 outstanding warrants exercises with exercise prices ranging from $1.25 to $1.79 were exercised, for total gross proceeds of approximately $13.8 million.
F - 31
c.Share option plans:
On March 30, 2012, the Company’s board of directors adopted the ReWalk Robotics Ltd. 2012 Equity Incentive Plan.
On August 19, 2014, the Company’s board of directors adopted the ReWalk Robotics Ltd. 2014 Incentive Compensation Plan or the “Plan”. The Plan provides for the grant of stock options, stock appreciation rights, restricted stock awards, restricted stock units, cash-based awards, other stock-based awards and dividend equivalents to the Company’s and its affiliates’ respective employees, non-employee directors and consultants.
Starting in 2014, the Company grants to directors and employees also Restricted Stock Units (“RSUs’’) under this Plan. An RSU award is an agreement to issue shares of the company’s ordinary shares at the time the award is vested.
As of December 31, 2021 and 2020, the Company had reserved 233,957 and 604,320 shares of ordinary shares, respectively, available for issuance to employees, directors, officers, and non-employees of the Company.
F - 32
The options generally vest over four years, with certain options granted to non-employee directors during the fiscal year ended December 31, 2019, vesting over one year.
Any option or RSUs that are forfeited or canceled before expiration becomes available for future grants under the Plan.
A summary of employee and non-employee shares options activity during the fiscal year ended 2021 is as follows:
Number | Average exercise price | Average remaining contractual life (years) | Aggregate intrinsic value (in thousands) | |||||||||||||
Options outstanding at the beginning of the year | 69,606 | $ | 37.9 | 5.59 | $ | 0— | ||||||||||
Granted | 0— | 0— | — | — | ||||||||||||
Exercised | 0— | 0— | — | — | ||||||||||||
Forfeited | (7,774 | ) | 34.68 | — | — | |||||||||||
| ||||||||||||||||
Options outstanding at the end of the year | 61,832 | $ | 38.34 | 4.55 | $ | 0— | ||||||||||
| ||||||||||||||||
Options exercisable at the end of the year | 55,747 | $ | 41.09 | 4.30 | $ | 0— | ||||||||||
A summary of employee and non-employee RSUs activity during the fiscal year ended 2021 is as follows:
Number of shares underlying outstanding RSUs | Weighted- average grant date fair value | |||||||
Unvested RSUs at the beginning of the year | 1,251,311 | 1.69 | ||||||
Granted | 721,216 | 1.69 | ||||||
Vested | (398,164 | ) | 1.74 | |||||
Forfeited | (218,079 | ) | 1.50 | |||||
| ||||||||
Unvested RSUs at the end of the year | 1,356,284 | 1.61 | ||||||
The weighted average grant date fair values of options granted during the fiscal year ended December 31, 2019, were $2.98, there were no options granted during the fiscal year ended December 31, 2021, and 2020. The weighted average grant date fair values of RSUs granted during the fiscal year ended December 31, 2021, 2020 and 2019, were $1.69, $1.44 and $4.67, respectively.
The aggregate intrinsic value in the table above represents the total intrinsic value that would have been received by the option holders had all option holders, which hold options with positive intrinsic value, exercised their options on the last date of the exercise period. During the years ended December 31, 2021, 2020 and 2019, no options were exercised. Total fair value of shares vested during the year ended December 31, 2021, 2020 and 2019 were $802 thousand, $676 thousand, and $1.18 million, respectively. As of December 31, 2021, there were $1.8 million of total unrecognized compensation cost related to non-vested share-based compensation arrangements granted under the 2014 Plan. This cost is expected to be recognized over a period of approximately 2.8 years.
F - 33
The number of options and RSUs outstanding as of December 31, 2021 is set forth below, with options separated by range of exercise price:
Range of exercise price | Options and RSUs Outstanding as of December 31, 2021 | Weighted average remaining contractual life (years) (1) | Options Exercisable as of December 31, 2021 | Weighted average remaining contractual life (years) (1) | ||||||||||||
RSUs only | 1,356,284 | — | 0— | — | ||||||||||||
$5.37 | 12,425 | 7.24 | 8,542 | 7.24 | ||||||||||||
$20.42- $33.75 | 31,155 | 4.08 | 28,953 | 3.91 | ||||||||||||
$37.14-$38.75 | 8,946 | 1.98 | 8,946 | 1.98 | ||||||||||||
$50-$52.5 | 6,731 | 5.46 | 6,731 | 5.46 | ||||||||||||
$182.5-$524.25 | 2,575 | 3.85 | 2,575 | 3.85 | ||||||||||||
1,418,116 | 4.55 | 55,747 | 4.30 | |||||||||||||
| |
(1) | Calculation of weighted average remaining contractual term does not include the RSUs that were granted, which have an indefinite contractual term. |
d.Equity compensation issued to consultants:
The Company granted 6,680 fully vested RSUs during the fiscal year ended December 31, 2019, to non-employee consultants. As of December 31, 2021, there are no outstanding options or RSUs held by non-employee consultants.
e.Share-based compensation expense for employees and non-employees:
The Company recognized non-cash share-based compensation expense in the consolidated statements of operations as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Cost of revenues | $ | 10 | $ | 8 | $ | 13 | ||||||
Research and development, net | 55 | 136 | 204 | |||||||||
Sales and marketing, net | 171 | 163 | 166 | |||||||||
General and administrative | 597 | 442 | 725 | |||||||||
| ||||||||||||
Total | $ | 833 | $ | 749 | $ | 1,108 | ||||||
F - 34
f.Warrants to purchase ordinary shares:
The following table summarizes information about warrants outstanding and exercisable as of December 31, 2021:
Issuance date | Warrants outstanding | Exercise price per warrant | Warrants outstanding and exercisable | Contractual term | |||||||||
(number) | (number) | ||||||||||||
December 31, 2015 (1) | 4,771 | $ | 7.500 | 4,771 | See footnote (1) | ||||||||
December 28, 2016 (2) | 1,908 | $ | 7.500 | 1,908 | See footnote (1) | ||||||||
November 20, 2018 (3) | 126,839 | $ | 7.500 | 126,839 | November 20, 2023 | ||||||||
November 20, 2018 (4) | 106,680 | $ | 9.375 | 106,680 | November 15, 2023 | ||||||||
February 25, 2019 (5) | 45,600 | $ | 7.187 | 45,600 | February 21, 2024 | ||||||||
April 5, 2019 (6) | 408,457 | $ | 5.140 | 408,457 | October 7, 2024 | ||||||||
April 5, 2019 (7) | 49,015 | $ | 6.503 | 49,015 | April 3, 2024 | ||||||||
June 5, 2019, and June 6, 2019 (8) | 1,464,665 | $ | 7.500 | 1,464,665 | June 5, 2024 | ||||||||
June 5, 2019 (9) | 87,880 | $ | 9.375 | 87,880 | June 5, 2024 | ||||||||
June 12, 2019 (10) | 416,667 | $ | 6.000 | 416,667 | December 12, 2024 | ||||||||
June 10, 2019 (11) | 50,000 | $ | 7.500 | 50,000 | June 10, 2024 | ||||||||
February 10, 2020 (12) | 28,400 | $ | 1.250 | 28,400 | February 10, 2025 | ||||||||
February 10, 2020 (13) | 105,840 | $ | 1.563 | 105,840 | February 10, 2025 | ||||||||
July 6, 2020 (14) | 448,698 | $ | 1.760 | 448,698 | January 2, 2026 | ||||||||
July 6, 2020 (15) | 296,297 | $ | 2.278 | 296,297 | January 2, 2026 | ||||||||
December 8, 2020 (16) | 586,760 | $ | 1.340 | 586,760 | June 8, 2026 | ||||||||
December 8, 2020 (17) | 108,806 | $ | 1.792 | 108,806 | June 8, 2026 | ||||||||
February 26, 2021 (18) | 5,460,751 | $ | 3.600 | 5,460,751 | August 26, 2026 | ||||||||
February 26, 2021 (19) | 655,290 | $ | 4.578 | 655,290 | August 26, 2026 | ||||||||
September 29, 2021 (20) | 8,006,759 | $ | 2.000 | 8,006,759 | March 29, 2027 | ||||||||
September 29, 2021 (21) | 960,811 | $ | 2.544 | 960,811 | September 27, 2026 | ||||||||
19,420,894 | 19,420,894 | ||||||||||||
| |
(1) | Represents warrants for ordinary shares issuable upon an exercise price of $7.500 per share, which were granted on December 31, 2015 to Kreos Capital V (Expert) Fund Limited (“Kreos”) in connection with a loan made by Kreos to the Company and are currently exercisable (in whole or in part) until the earlier of (i) December 30, 2025 or (ii) immediately prior to the consummation of a merger, consolidation, or reorganization of the Company with or into, or the sale or license of all or substantially all the assets or shares of the Company to, any other entity or person, other than a wholly owned subsidiary of the Company, excluding any transaction in which the Company’s shareholders prior to the transaction will hold more than 50% of the voting and economic rights of the surviving entity after the transaction. None of these warrants had been exercised as of December 31, 2021. |
| |
(2) | Represents common warrants that were issued as part of the $8.0 million drawdown under the Loan Agreement which occurred on December 28, 2016. See footnote 1 for exercisability terms. |
| |
(3) | Represents common warrants that were issued as part of the Company’s follow-on public offering in November 2018. |
| |
(4) | Represents common warrants that were issued to the underwriters as compensation for their role in the Company’s follow-on public offering in November 2018. |
| |
(5) | Represents warrants that were issued to the exclusive placement agent as compensation for its role in the Company’s follow-on public offering in February 2019. |
F - 35
(6) | Represents warrants that were issued to certain institutional purchasers in a private placement in the Company’s registered direct offering of ordinary shares in April 2019. |
| |
(7) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s April 2019 registered direct offering. |
| |
(8) | Represents warrants that were issued to certain institutional investors in a warrant exercise agreement on June 5, 2019, and June 6, 2019, respectively. |
| |
(9) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s June 2019 warrant exercise agreement and concurrent private placement of warrants. |
| |
(10) | Represents warrants that were issued to certain institutional investors in a warrant exercise agreement in June 2019. |
| |
(11) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s June 2019 registered direct offering and concurrent private placement of warrants. |
| |
(12) | Represents warrants that were issued to certain institutional purchasers in a private placement in the Company’s best efforts offering of ordinary shares in February 2020. During the year ended December 31, 2021, 3,740,100 warrants were exercised for total consideration of $4,675,125. |
| |
(13) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s February 2020 best efforts offering. During the year ended December 31, 2021, 230,160 warrants were exercised for total consideration of $359,625. |
| |
(14) | Represents warrants that were issued to certain institutional purchasers in a private placement in our registered direct offering of ordinary shares in July 2020. During the year ended December 31, 2021, 2,020,441 warrants were exercised for total consideration of $3,555,976. |
| |
(15) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s July 2020 registered direct offering. |
| |
(16) | Represents warrants that were issued to certain institutional purchasers in a private placement in our private placement offering of ordinary shares in December 2020. During the year ended December 31, 2021, 3,598,072 warrants were exercised for total consideration of $4,821,416. |
| |
(17) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s December 2020 private placement. During the year ended December 31, 2021, 225,981 warrants were exercised for total consideration of $405,003. |
| |
(18) | Represents warrants that were issued to certain institutional purchasers in a private placement in our private placement offering of ordinary shares in February 2021. |
| |
(19) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s February 2021 private placement. |
| |
(20) | Represents warrants that were issued to certain institutional purchasers in a private placement in our registered direct offering of ordinary shares in September 2021. |
| |
(21) | Represents warrants that were issued to the placement agent as compensation for its role in the Company’s September 2021 registered direct offering. |
F - 36
NOTE 9:-RESEARCH COLLABORATION AGREEMENT AND LICENSE AGREEMENT
On May 16, 2016, the Company entered into a Research Collaboration Agreement (“Collaboration Agreement”) and an Exclusive License Agreement (“License Agreement”) with Harvard. The Research Collaboration Agreement was amended on May 1, 2017, and April 1, 2018 (as amended, the “Collaboration Agreement”), and the Exclusive License Agreement was amended on April 1, 2018 (as amended, the “License Agreement”), to extend the term of the Collaboration Agreement by one year to May 16, 2022 and reallocate the Company’s quarterly installment payments to Harvard through such date, and to make certain technical changes. On April 30, 2020, the Company and Harvard amended the Collaboration Agreement, which included certain adjustments to the quarterly installments and extended the term an additional three quarters until February 2023.
On October 14, 2021, the Company and Harvard further amended the Collaboration Agreement, to make certain adjustments to the quarterly installments and technical changes and establish that the term of the Collaboration Agreement will conclude on March 31, 2022. The Company and Harvard might consider new arrangement to support our research efforts in the future.
Under the Collaboration Agreement, Harvard and the Company have agreed to collaborate on research regarding the development of lightweight “soft suit” exoskeleton system technologies for lower limb disabilities, which are intended to treat stroke, multiple sclerosis, mobility limitations for the elderly and other medical applications. The Company has committed to pay in quarterly installments for the funding of this research.
Under the License Agreement, Harvard has granted the Company an exclusive, worldwide royalty-bearing license under certain patents of Harvard relating to lightweight “soft suit” exoskeleton system technologies for lower limb disabilities, a royalty-free license under certain related know-how and the option to obtain a license under certain inventions conceived under the joint research collaboration.
The License Agreement required the Company to pay Harvard an upfront fee, reimbursements for expenses that Harvard incurred in connection with the licensed patents, royalties on net sales and several milestone payments contingent upon the achievement of certain product development and commercialization milestones. The Harvard License Agreement will continue in full force and effect until the expiration of the last-to-expire valid claim of the licensed patents. As of December 31, 2021, the Company achieved three of the milestones which represent all development milestones under the License Agreement. The Company continues to evaluate the likelihood that the other milestones will be achieved on a quarterly basis.
The Company has recorded expenses in the amount of $293 thousand, $762 thousand, and $1.6 million for the years ended December 31, 2021, 2020, and 2019, respectively, as research and development expenses related to the Harvard License Agreement and to the Collaboration Agreement. No withholding tax was deducted from the Company’s payments to Harvard in respect of the Collaboration Agreement and License Agreement since this is not taxable income in Israel in accordance with Section 170 of the Israel Income Tax Ordinance 1961-5721.
F - 37
NOTE 10: -PAYCHECK PROTECTION PROGRAM LOAN
On April 21, 2020, RRI received an unsecured loan in the principal amount of $392 thousand under the Paycheck Protection Program (the “PPP”) administered by the U.S. Small Business Administration, or the SBA, pursuant to the Coronavirus Aid, Relief, and Economic Security Act (the “CARES Act”), or the PPP loan. The terms of the PPP Loan were subsequently revised in accordance with the provisions of the Paycheck Protection Flexibility Act of 2020, or the PPP Flexibility Act, which was enacted on June 5, 2020. The PPP loan provides for an interest rate of 1.00% per year and matures two years after the date of initial disbursement, with initial principal and interest payments coming due late in fiscal 2021. The PPP loan may be used for payroll costs, costs related to certain group health care benefits and insurance premiums, rent payments, utility payments, mortgage interest payments and interest payments on any other debt obligation that were incurred before February 15, 2020. Under the terms of the CARES Act and the PPP Flexibility Act, the Company may apply for and be granted forgiveness for all or a portion of loan granted under the PPP loan, with such forgiveness to be determined, subject to limitations (including where employees of the Company have been terminated and not re-hired by a certain date), based on the use of the loan proceeds for payment of payroll costs and any payments of mortgage interest, rent, and utilities. The terms of any forgiveness may also be subject to further requirements in regulations and guidelines adopted by the SBA.
On September 29, 2020, the Company applied for loan forgiveness and on November 6, 2020, the Company received confirmation of its PPP Note forgiveness.
Forgiveness is booked as other income within the marketing and sales expenses because it was granted and used for payroll, rent, and utility costs related to sales efforts.
NOTE 11: -INCOME TAXES
The Company’s subsidiaries are separately taxed under the domestic tax laws of the jurisdiction of incorporation of each entity.
a.Corporate tax rates in Israel:
Presented hereunder are the tax rates relevant to the Company in the years 2019-2021:
The Israeli statutory corporate tax rate and real capital gains were 23% in the years 2019-2021.
b.Income (loss) before taxes on income is comprised as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Domestic | $ | (12,780 | ) | $ | (12,992 | ) | $ | (15,599 | ) | |||
Foreign | 138 | 67 | 55 | |||||||||
$ | (12,642 | ) | $ | (12,925 | ) | $ | (15,544 | ) | ||||
F - 38
c.Taxes on income are comprised as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Current | $ | 123 | $ | 95 | $ | 64 | ||||||
Deferred | (29 | ) | (44 | ) | (57 | ) | ||||||
| ||||||||||||
$ | 94 | $ | 51 | $ | 7 | |||||||
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Domestic | $ | 0— | $ | 0— | $ | 0— | ||||||
Foreign | 94 | 51 | 7 | |||||||||
| ||||||||||||
$ | 94 | $ | 51 | $ | 7 | |||||||
d.Deferred income taxes (in thousands):
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. The Company’s deferred tax assets as of December 31, 2021 and 2020 are derived from temporary differences.
In assessing the realization of deferred tax assets, the Company considers whether it is more likely than not that all or some portion of the deferred tax assets will not be realized. Based on the Company’s history of losses, the Company established a full valuation allowance for RRL.
Undistributed earnings of certain subsidiaries as of December 31, 2021 were immaterial. The Company intends to reinvest these earnings indefinitely in the foreign subsidiaries. As a result, the Company has not provided for any deferred income taxes.
December 31, | ||||||||
2021 | 2020 | |||||||
Deferred tax assets: | ||||||||
Carry forward tax losses | $ | 47,323 | $ | 41,941 | ||||
Research and development carry forward expenses-temporary differences | 718 | 946 | ||||||
Accrual and reserves | 373 | 341 | ||||||
Lease liabilities | 261 | 390 | ||||||
Total deferred tax assets | 48,675 | 43,618 | ||||||
Deferred tax liabilities: | ||||||||
Right-of-use asset | (261 | ) | (390 | ) | ||||
Net deferred tax assets | 48,414 | 43,228 | ||||||
Valuation allowance | (48,098 | ) | (42,941 | ) | ||||
| ||||||||
Net deferred tax assets | $ | 316 | $ | 287 | ||||
F - 39
The net changes in the total valuation allowance for each of the years ended December 31, 2021, 2020 and 2019, are comprised as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Balance at beginning of year | $ | (42,941 | ) | $ | (36,392 | ) | $ | (29,655 | ) | |||
Changes due to exchange rate differences | (1,488 | ) | (2,929 | ) | (2,055 | ) | ||||||
Adjustment previous year loss | 0— | 0— | (735 | ) | ||||||||
Additions during the year | (3,669 | ) | (3,620 | ) | (3,947 | ) | ||||||
| ||||||||||||
Balance at end of year | $ | (48,098 | ) | $ | (42,941 | ) | $ | (36,392 | ) | |||
e.Reconciliation of the theoretical tax expenses:
A reconciliation between the theoretical tax expense, assuming all income is taxed at the statutory tax rate applicable to income of the Company, and the actual tax expense (benefit) as reported in the consolidated statements of operations is as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Loss before taxes, as reported in the consolidated statements of operations | $ | (12,642 | ) | $ | (12,925 | ) | $ | (15,544 | ) | |||
| ||||||||||||
Statutory tax rate | 23.0 | % | 23.0 | % | 23.0 | % | ||||||
| ||||||||||||
Theoretical tax benefits on the above amount at the Israeli statutory tax rate | $ | (2,908 | ) | $ | (2,973 | ) | $ | (3,575 | ) | |||
Income tax at rate other than the Israeli statutory tax rate | 7 | 3 | (1 | ) | ||||||||
Non-deductible expenses including equity-based compensation expenses and other | 102 | 185 | 255 | |||||||||
Operating losses and other temporary differences for which valuation allowance was provided | 3,669 | 3,620 | 3,947 | |||||||||
Permanent differences | (784 | ) | (706 | ) | (651 | ) | ||||||
Other | 8 | (78 | ) | 32 | ||||||||
| ||||||||||||
Actual tax expense | $ | 94 | $ | 51 | $ | 7 | ||||||
f.Foreign tax rates:
Taxable income of RRI was subject to tax at the rate of 21% in 2021, 2020 and 2019.
Taxable income of RRG was subject to tax at the rate of 30% in 2021, 2020, and 2019.
g.Tax benefits under the Law for the Encouragement of Capital Investments, 1959 (the “Investment Law”):
Conditions for entitlement to the benefits:
Under the Investment Law, in 2012 the Company elected “Beneficiary Enterprise” status which provides certain benefits, including tax exemptions and reduced tax rates. Income not eligible for Beneficiary Enterprise benefits is taxed at a regular rate.
F - 40
Income derived from Beneficiary Enterprise from productive activity will be exempt from tax for ten years from the year in which the Company first has taxable income, providing that 12 years have not passed from the beginning of the year of election. In the event of a dividend distribution from income that is exempt from company tax, as aforementioned, the Company will be required to pay tax of 10%-25% on that income.
In the event of distribution of dividends from the said tax-exempt income, the amount distributed will be subject to corporate tax at the rate ordinarily applicable to the Beneficiary Enterprise’s income. Tax-exempt income generated under the Company’s “Beneficiary Enterprise” program will be subject to taxes upon dividend distribution or complete liquidation.
The entitlement to the above benefits is conditional upon the Company’s fulfilling the conditions stipulated by the Law and regulations published thereunder.
On December 29, 2010, the Knesset approved an additional amendment to the Law for the Encouragement of Capital Investments, 1959. According to the amendment, a reduced uniform corporate tax rate for exporting industrial enterprises (over 25%) was established. The reduced tax rate will not be program dependent and will apply to the industrial enterprise’s entire income. The tax rates for industrial enterprises have been reduced. In August 2013, the Israeli Knesset approved an amendment to the Investment Law, pursuant to which the rates for development area A will be 9% and for the rest of the country- 16% in 2014 and thereafter. The Amendment also prescribes that any dividends distributed to individuals or foreign residents from a preferred enterprise’s earnings as above will be subject to taxes at a rate of 20% (subject to tax treaty benefits)
In December 2016, the Economic Efficiency Law (Legislative Amendments for Applying the Economic Policy for the 2017 and 2018 Budget Years), 2016 which includes Amendment 73 to the Law for the Encouragement of Capital Investments (“the Amendment”) was published. According to the Amendment, a preferred enterprise located in development area A will be subject to a tax rate of 7.5% instead of 9% effective from January 1, 2017 (and thereafter the tax rate applicable to preferred enterprises located in other areas remains at 16%).
The Company has examined the effect of the adoption of the Amendment on its financial statements, and as of the date of the publication of the financial statements, the Company estimates that it will not apply the Amendment. The Company’s estimate may change in the future.
h.Tax assessments:
RRL has had final tax assessments up to and including the 2016 tax year.
Each RRI and RRG have not had a final tax assessment since its inception.
i.Net operating carry-forward losses for tax purposes:
As of December 31, 2021, RRL has carry-forward losses amounting to approximately $205.8 million, which can be carried forward for an indefinite period, and RRI has carry-forward losses amounting to approximately $74 thousands, which can be carried forward for a period of 20 years.
F - 41
NOTE 12: - FINANCIAL EXPENSES (INCOME), NET
The components of financial expenses (income), net were as follows (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Foreign currency transactions and other | $ | (38 | ) | $ | (9 | ) | $ | (34 | ) | |||
Financial expenses related to loan agreement with Kreos | 0— | 907 | 1,499 | |||||||||
Bank commissions | 25 | 23 | 31 | |||||||||
| ||||||||||||
$ | (13 | ) | $ | 921 | $ | 1,496 | ||||||
NOTE 13: - GEOGRAPHIC INFORMATION AND MAJOR CUSTOMER AND PRODUCT DATA
Summary information about geographic areas:
ASC 280, “Segment Reporting” establishes standards for reporting information about operating segments. Operating segments are defined as components of an enterprise about which separate financial information is available that is evaluated regularly by the chief operating decision maker in deciding how to allocate resources and in assessing performance. The Company manages its business on the basis of 1 reportable segment and derives revenues from selling systems and services (see Note 1 for a brief description of the Company’s business). The following is a summary of revenues within geographic areas (in thousands):
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Revenues based on customer’s location: | ||||||||||||
Israel | $ | 0— | $ | 0— | $ | 2 | ||||||
United States | 2,519 | 1,746 | 2,003 | |||||||||
Europe | 3,381 | 2,631 | 2,832 | |||||||||
Asia-Pacific | 60 | 8 | 36 | |||||||||
Latin America | 0— | 6 | 0— | |||||||||
Africa | 6 | 2 | 0— | |||||||||
| ||||||||||||
Total revenues | $ | 5,966 | $ | 4,393 | $ | 4,873 | ||||||
December 31, | ||||||||
2021 | 2020 | |||||||
Long-lived assets by geographic region: | ||||||||
Israel | $ | 629 | $ | 953 | ||||
United States | 493 | 790 | ||||||
Germany | 43 | 43 | ||||||
| ||||||||
$ | 1,165 | $ | 1,786 | |||||
| |
(*) | Long-lived assets are comprised of property and equipment, net, and operating lease right-of-use assets. |
|
F - 42
Major customers data as a percentage of total revenues:
Year Ended December 31, | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Customer A | 11.0 | % | * | ) | * | ) | ||||||
Customer B | * | ) | 10.0 | % | 15.0 | % | ||||||
*) | Less than 10% |
F - 43
