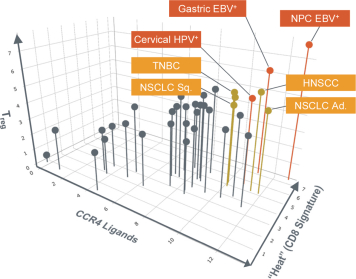
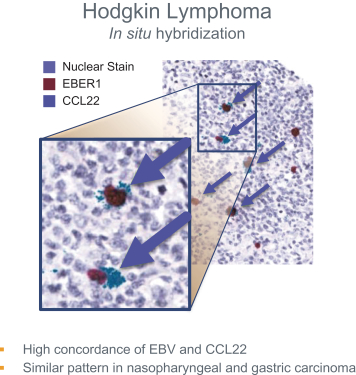
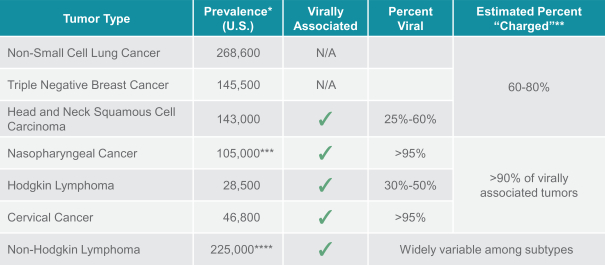
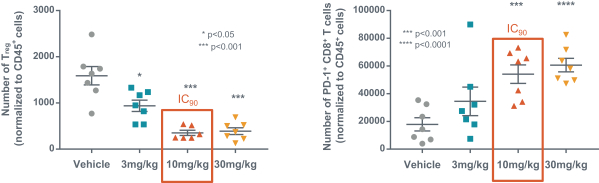
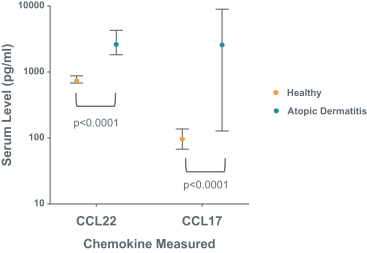
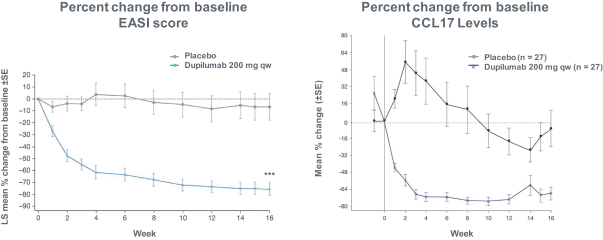
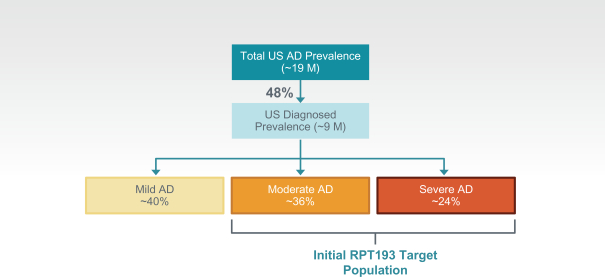
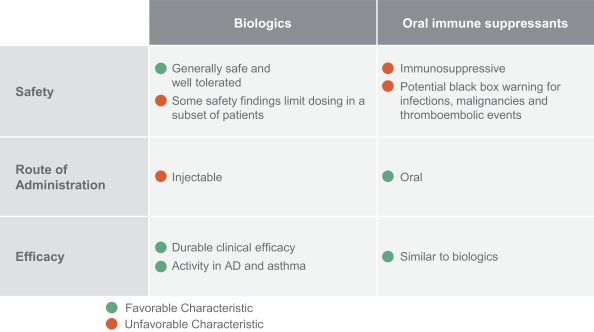
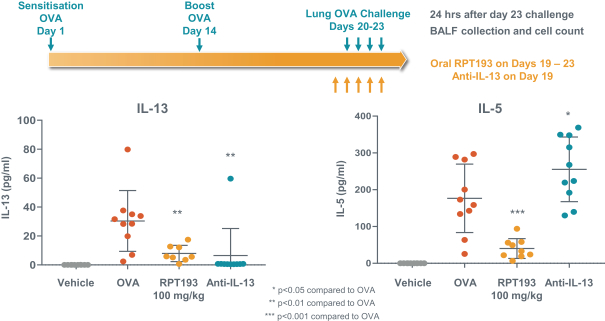
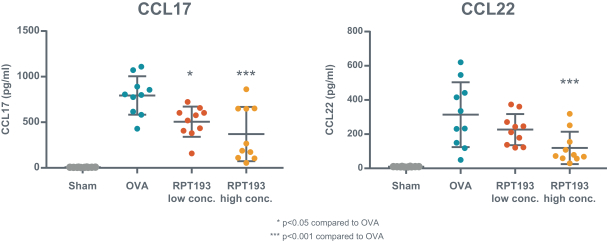
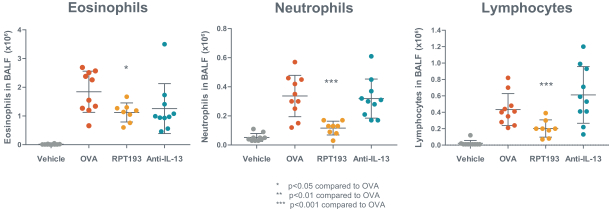
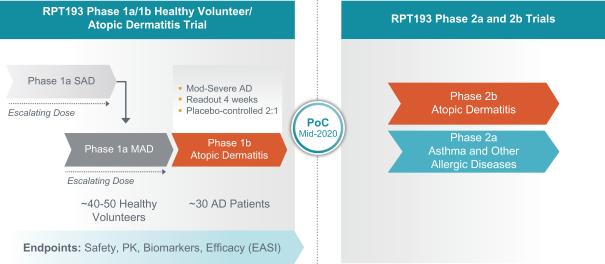
us, have significant clinical experience, and may include our future partners. In addition, these companies compete with us in recruiting scientific and managerial talent. Our success will partially depend on our ability to obtain, maintain, enforce and defend patents and other intellectual property rights with respect to small molecule drugs or biologics that are safer and more effective than competing products. Our commercial opportunity and success will be reduced or eliminated if competing products that are safer, more effective, or less expensive than the drugs we develop are or become available.
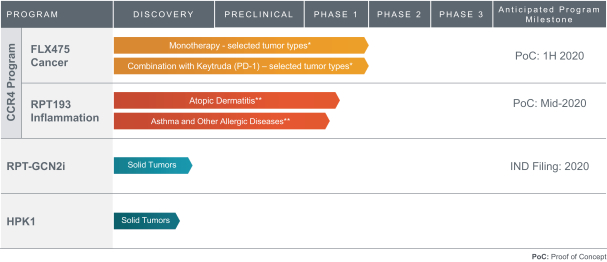
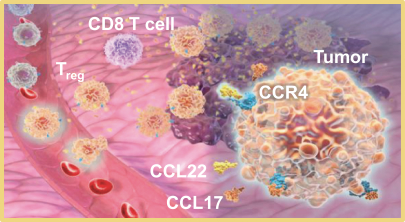
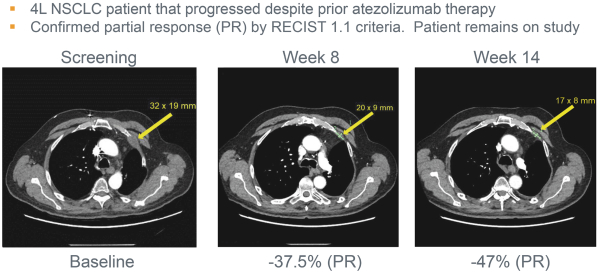
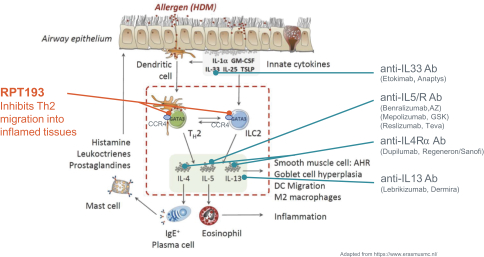
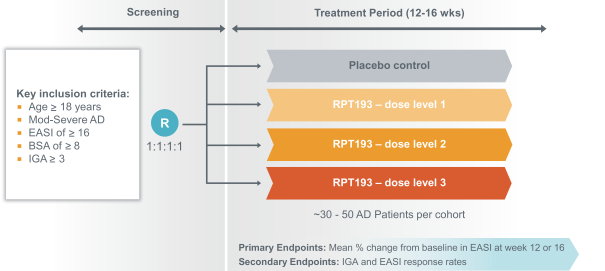
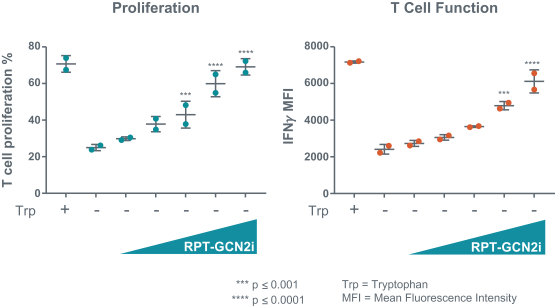
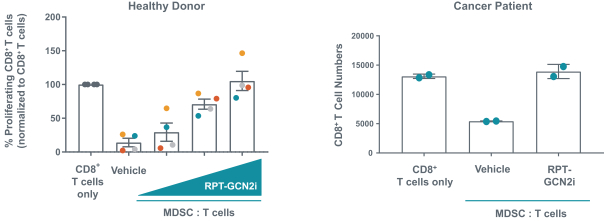
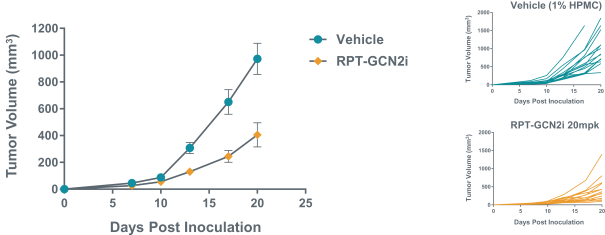
We expect to compete with small molecule, biologics and other therapeutic platforms and development companies, including, but not limited to, companies such as ChemoCentryx, Tusk/Roche and Agenus/Gilead for oncology, and Dermira and AnaptysBio for inflammatory diseases. In addition, we expect to compete with large, multinational pharmaceutical companies that discover, develop and commercialize small molecule drugs and other therapeutics for use in treating cancer and inflammatory diseases such as AbbVie, Amgen, AstraZeneca plc, Bristol-Myers Squibb, GlaxoSmithKline, Incyte, Kyowa Hakko Kirin, Merck, Novartis, Pfizer, Roche/Genentech and Sanofi/Regeneron. If FLX475, RPT193 or anRPT-GCN2i or other future drug candidate is eventually approved, it will compete with a range of treatments that are either in development or currently marketed.
Many of our competitors have significantly greater financial, technical, manufacturing, marketing, sales and supply resources or experience than we do. If we successfully obtain approval for any drug candidate, we will face competition based on many different factors, including the safety and effectiveness of our products, the ease with which our products can be administered, the timing and scope of regulatory approvals for these products, the availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage and patent position. Competing products could present superior treatment alternatives, including by being more effective, safer, less expensive or marketed and sold more effectively than any products we may develop. Competitive products may make any product we develop obsolete or noncompetitive before we recover the expense of developing and commercializing such product. Such competitors could also recruit our employees, which could negatively impact our level of expertise and our ability to execute our business plan.
Any inability to attract and retain qualified key management, technical personnel and employees would impair our ability to implement our business plan.
Our success largely depends on the continued service of key management, advisors and other specialized personnel, including Brian Wong, M.D., Ph.D., our President and Chief Executive Officer, William Ho, M.D., Ph.D., our Chief Medical Officer, Dirk Brockstedt, Ph.D., our Chief Scientific Officer, David Wustrow, Ph.D., our Senior Vice President of Drug Discovery and Preclinical Development, Paul Kassner, Ph.D., our Vice President, Quantitative and Computational Biology, and Karen C. Lam, our Vice President, Finance and Corporate Controller, as well as our ability to attract and retain other highly qualified personnel. We have a written employment agreement with each of Dr. Wong, Dr. Ho, Dr. Brockstedt, Dr. Wustrow, Dr. Kassner, and Ms. Lam. The loss of one or more members of our executive team, management team or other key employees or advisors could delay our research and development programs and have a material and adverse effect on our business, financial condition, results of operations and prospects.
The relationships that our key managers have cultivated within our industry make us particularly dependent upon their continued employment with us. We are dependent on the continued service of our technical personnel because of the highly technical nature of our drug candidates and technologies and the specialized nature of the regulatory approval process. Because our management team and key employees are not obligated to provide us with continued service, they could terminate their employment with us at any time without penalty.
Our future success will also depend in large part on our ability to attract and retain other highly qualified scientific, technical and management personnel, as well as personnel with expertise in clinical testing, manufacturing, governmental regulation and commercialization. We face significant competition for personnel from other companies, universities, public and private research institutions, government entities and other organizations.
26