Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
TECX similar filings
- 1 May 23 AVROBIO Announces Leadership Transition
- 23 Mar 23 AVROBIO Reports Fourth Quarter and Fiscal Year 2022 Financial Results and Provides Business Update
- 27 Feb 23 Other Events
- 7 Dec 22 Regulation FD Disclosure
- 8 Nov 22 AVROBIO Reports Third Quarter 2022 Financial Results and Provides Business Update
- 7 Oct 22 Notice of Delisting or Failure to Satisfy a Continued Listing Rule or Standard
- 9 Aug 22 AVROBIO Reports Second Quarter 2022 Financial Results and Provides Business Update
Filing view
External links
Exhibit 99.2

CO NF I DE NT I AL Arianna living with Gaucher disease type 3

Disclaimer 2 This presentation has been prepared by AVROBIO, Inc. (“AVROBIO”) for informational purposes only and not for any other purpose. Certain information contained in this presentation and statements made orally during this presentation relate to or are based on studies, publications, surveys, and other data obtained from third-party sources and AVROBIO’s own internal estimates and research. Although AVROBIO believes these third-party sources to be reliable as of the date of this presentation, they have not been independently verified, and AVROBIO makes no representation as to the adequacy, fairness, accuracy. or completeness of any information obtained from third-party sources. Although AVROBIO believes its internal research is reliable, such research has not been verified by any independent source. This presentation may contain forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These statements may be identified by words and phrases such as “aims,” “anticipates,” “believes,” "continue," “could,” “designed to,” “estimates,” “expects,” “forecasts,” “goal,” “intends,” “may,” “plans,” “possible,” “potential,” "predicts," "projects," “seeks,” "strives," "should," and “will,” as well as variations of these words and phrases or similar expressions that are intended to identify forward-looking statements. These forward-looking statements include, without limitation, statements regarding our business strategy for and the potential therapeutic benefits of our current and prospective product candidates; the design, commencement, enrollment, and timing of ongoing or planned clinical trials and regulatory pathways; our plans and expectations with respect to the development of our clinical and preclinical product candidates, including timing, design, and initiation of our potential clinical and registration trials and anticipated interactions with regulatory agencies; the timing of anticipated clinical and regulatory updates; the timing of patient recruitment and enrollment activities, clinical trial results, and product approvals; the timing and results of our ongoing preclinical studies; the anticipated benefits of our gene therapy platform including the potential impact on our commercialization activities, timing and likelihood of success; the expected benefits and results of our manufacturing technology, including the implementation of our plato® platform in our clinical trials and gene therapy programs; the expected safety profile of our investigational gene therapies; and our financial position and cash runway expectations. Any such statements in this presentation that are not statements of historical fact may be deemed to be forward- looking statements. Any forward-looking statements in this presentation are based on our current expectations, estimates, and projections about our industry as well as management’s current beliefs and expectations of future events only as of today and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to, the risk that any one or more of our product candidates will not be successfully developed or commercialized; the risk that regulatory agencies may disagree with our anticipated development approach for any one or more of our product candidates; the risk of cessation or delay of any ongoing or planned clinical trials of AVROBIO or our collaborators; the risk that we may not successfully recruit or enroll a sufficient number of patients for our clinical trials; the risk that we may not realize the intended benefits of our gene therapy platform, including the features of our plato® platform; the risk that our product candidates or procedures in connection with the administration thereof, including our use of busulfan as a conditioning agent or potential use of monoclonal antibody conditioning agents, will not have the safety or efficacy profile that we anticipate; the risk that prior results, such as signals of safety, activity, or durability of effect, observed from preclinical or clinical trials, will not be replicated or will not continue in ongoing or future studies or trials involving our product candidates; the risk that we will be unable to obtain and maintain regulatory approval for our product candidates; the risk that the size and growth potential of the market for our product candidates will not materialize as expected; risks associated with our dependence on third-party suppliers and manufacturers; risks regarding the accuracy of our estimates of expenses and future revenue; risks relating to our capital requirements and needs for additional financing; risks relating to clinical trial and business interruptions resulting from the ongoing COVID-19 pandemic or similar public health crises, including that such interruptions may materially delay our development timeline and/or increase our development costs or that data collection efforts may be impaired or otherwise impacted by such crises; and risks relating to our ability to obtain and maintain intellectual property protection for our product candidates. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause AVROBIO’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in AVROBIO’s most recent Quarterly Report, as well as discussions of potential risks, uncertainties and other important factors in AVROBIO’s subsequent filings with the Securities and Exchange Commission. AVROBIO explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law. Note regarding trademarks: plato® is a registered trademark of AVROBIO. Other trademarks referenced in this presentation are the property of their respective owners. © Copyright 2022 AVROBIO, Inc. All rights reserved.
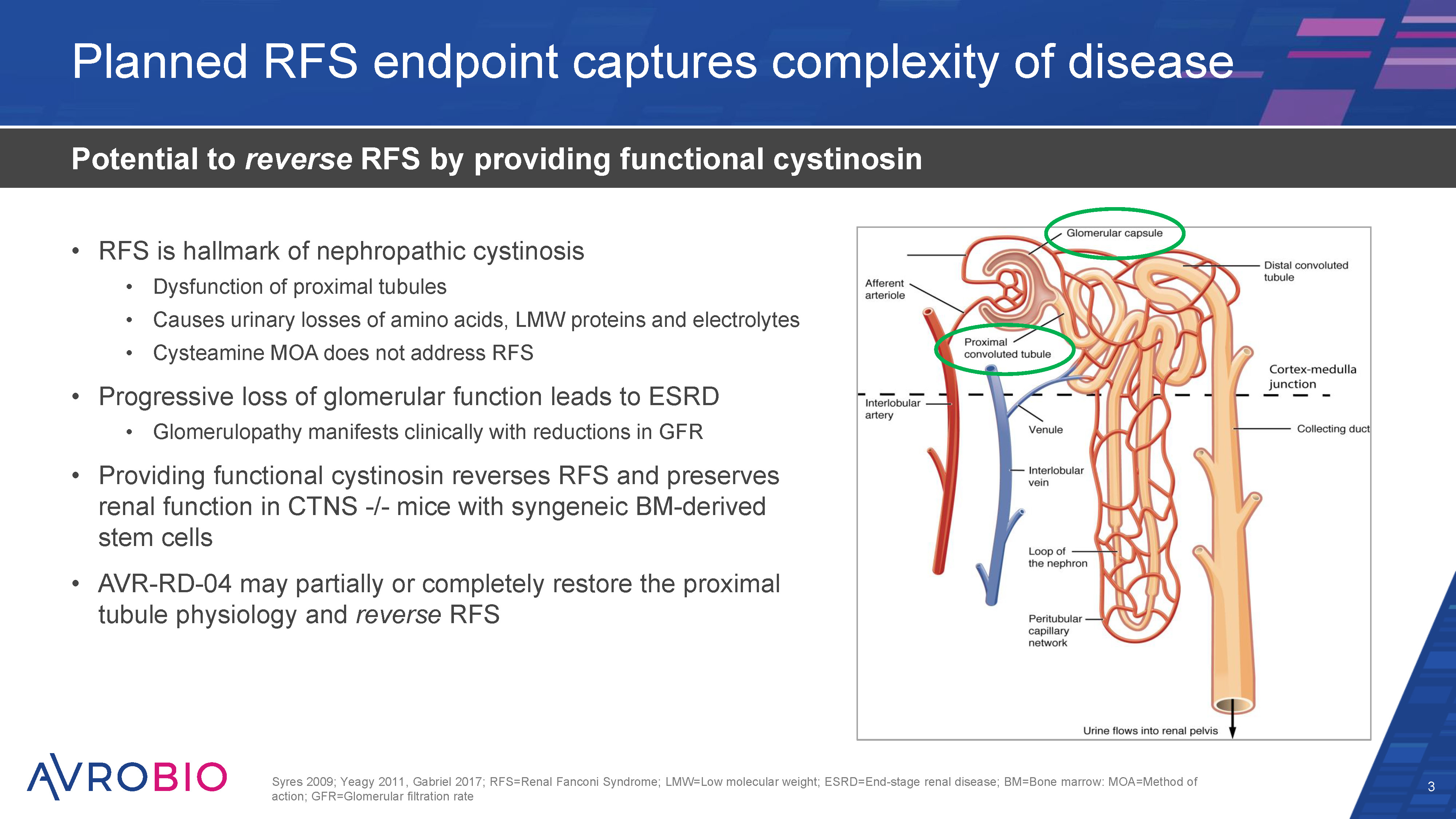
Leading hematopoietic stem cell (HSC) gene therapy company 3 CMC=Chemistry, manufacturing and controls pipeline targeting multi-billion dollar market Strong data generated to date across two clinical-stage programs Lysosomal disorder Late-stage trials in Gaucher disease and cystinosis planned for 2023 plato® platform delivers unrivaled CMC & analytics capabilities Multiple clinical and regulatory milestones anticipated over next 12 months Unique competitive position with first mover advantage in lead programs
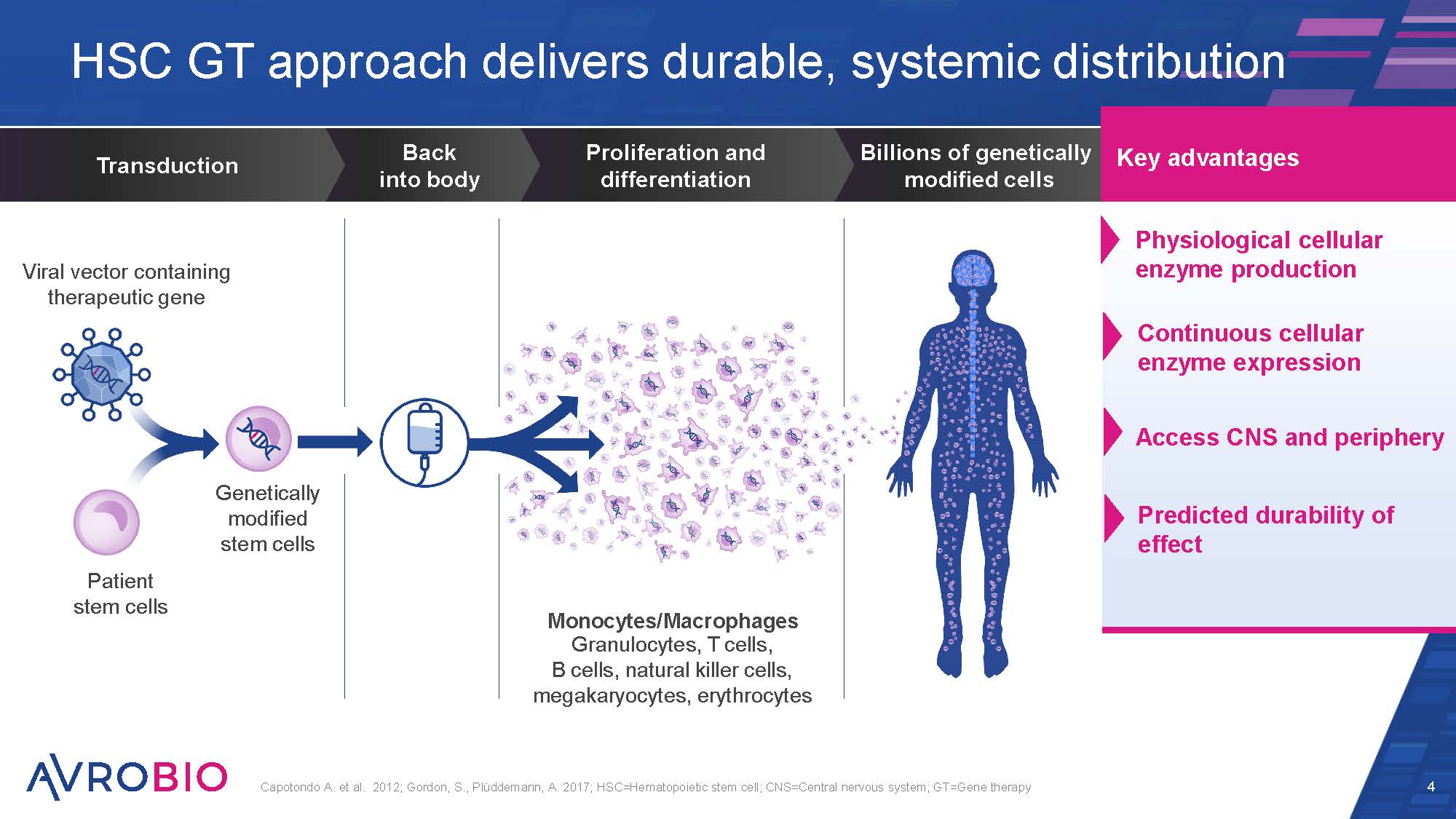
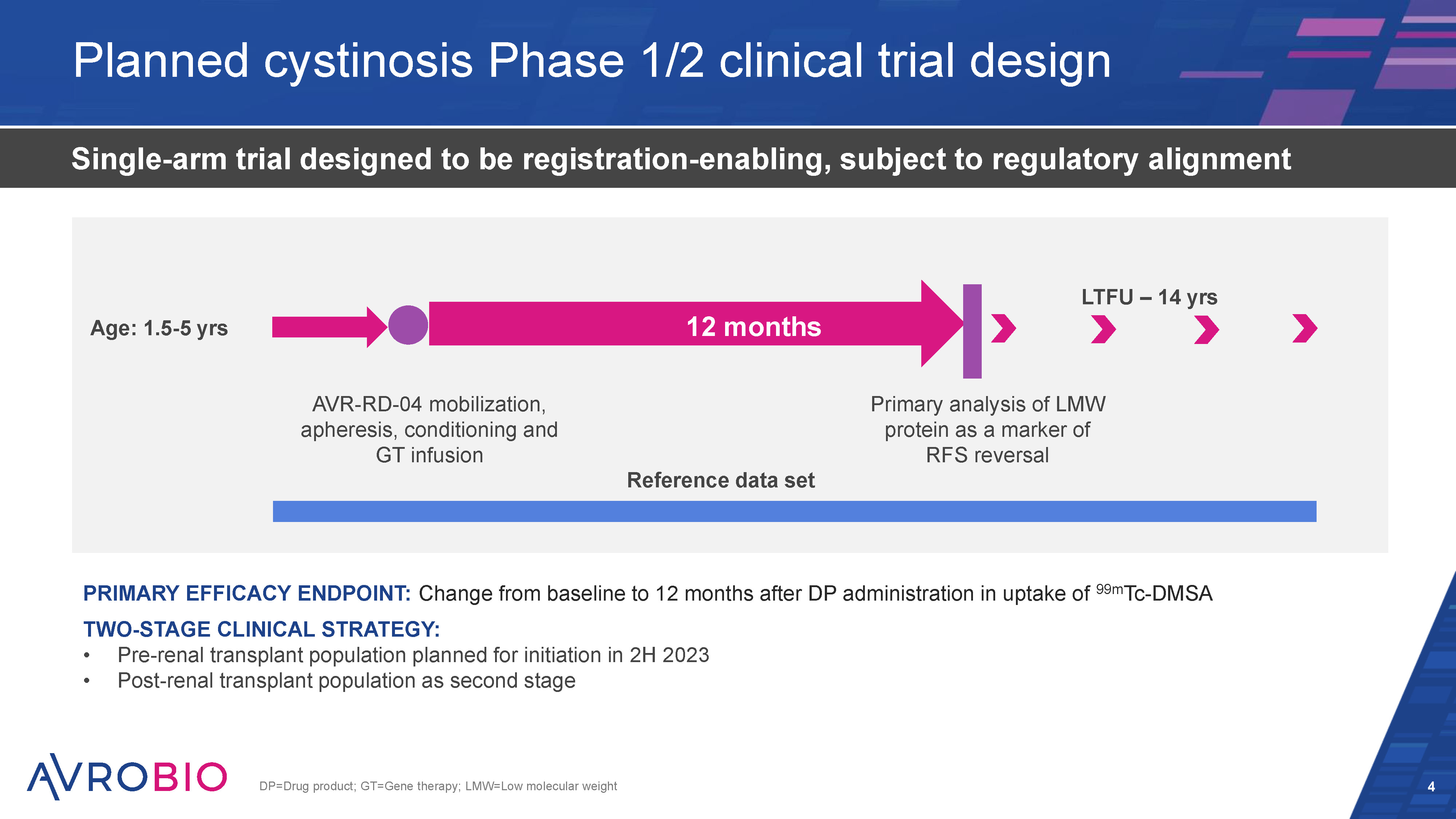
HSC GT approach delivers durable, systemic distribution 4 Capotondo A. et al. 2012; Gordon, S., Plüddemann, A. 2017; HSC=Hematopoietic stem cell; CNS=Central nervous system; GT=Gene therapy Billions of genetically modified cells Proliferation and differentiation Back into body Transduction Viral vector containing therapeutic gene Patient stem cells Genetically modified stem cells Monocytes/Macrophages Granulocytes, T cells, B cells, natural killer cells, megakaryocytes, erythrocytes Physiological cellular enzyme production Continuous cellular enzyme expression Access CNS and periphery Key advantages Predicted durability of effect
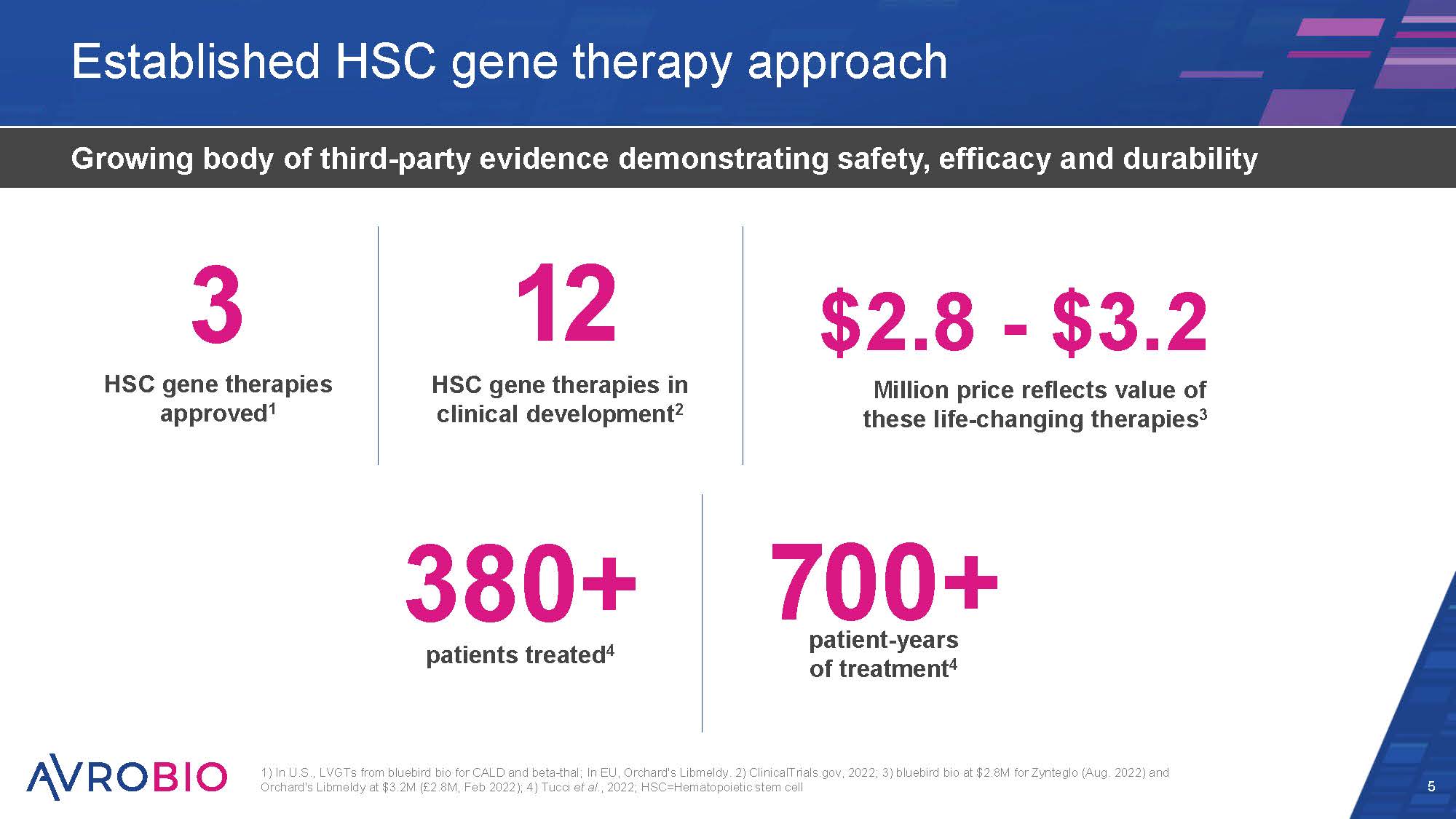
Established HSC gene therapy approach 5 Growing body of third-party evidence demonstrating safety, efficacy and durability 1) In U.S., LVGTs from bluebird bio for CALD and beta-thal; In EU, Orchard's Libmeldy. 2) ClinicalTrials.gov, 2022; 3) bluebird bio at $2.8M for Zynteglo (Aug. 2022) and Orchard's Libmeldy at $3.2M (£2.8M, Feb 2022); 4) Tucci et al., 2022; HSC=Hematopoietic stem cell 380+ patients treated4 3 HSC gene therapies approved1 12 HSC gene therapies in clinical development2 patient-years of treatment4 700+ $2.8 - $3.2 Million price reflects value of these life-changing therapies3
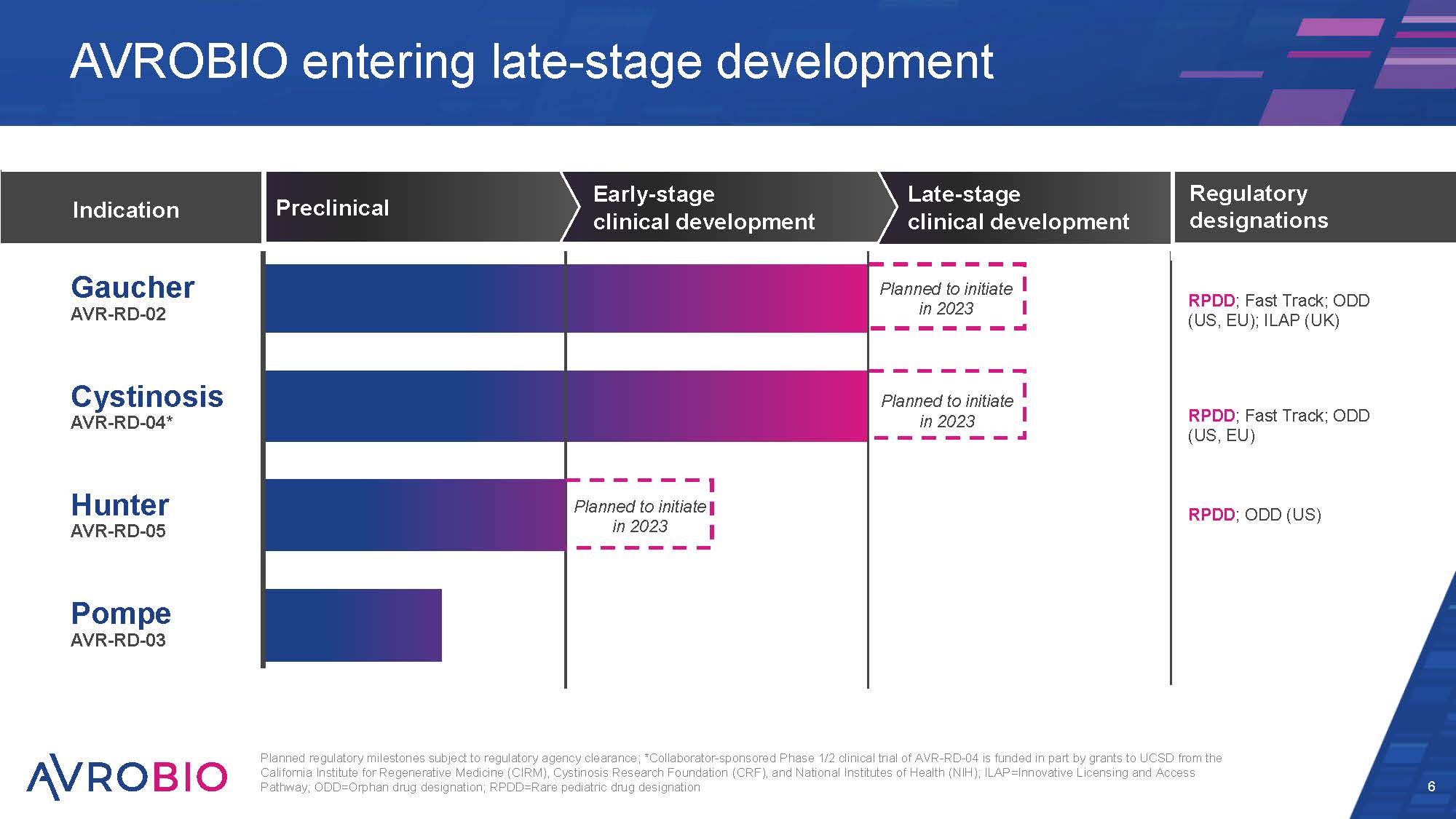
Late-stage clinical development Early-stage clinical development Preclinical Planned regulatory milestones subject to regulatory agency clearance; *Collaborator-sponsored Phase 1/2 clinical trial of AVR-RD-04 is funded in part by grants to UCSD from the California Institute for Regenerative Medicine (CIRM), Cystinosis Research Foundation (CRF), and National Institutes of Health (NIH); ILAP=Innovative Licensing and Access Pathway; ODD=Orphan drug designation; RPDD=Rare pediatric drug designation 6 AVROBIO entering late-stage development Indication Cystinosis AVR-RD-04* Hunter AVR-RD-05 Pompe AVR-RD-03 Gaucher AVR-RD-02 Regulatory designations RPDD; Fast Track; ODD (US, EU); ILAP (UK) RPDD; Fast Track; ODD (US, EU) RPDD; ODD (US) Planned to initiate in 2023 Planned to initiate in 2023 Planned to initiate in 2023
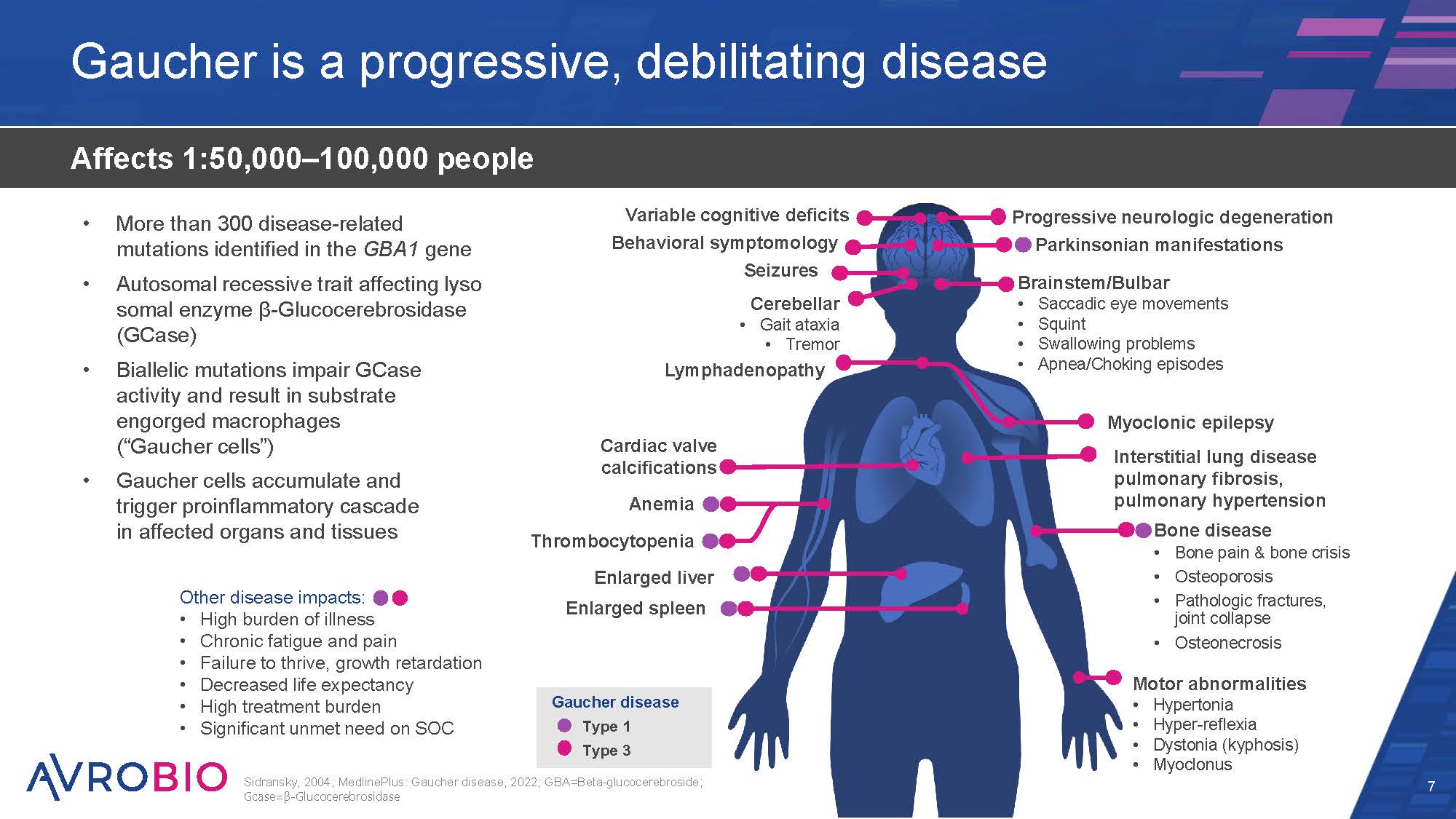
Brainstem/Bulbar Saccadic eye movements Squint Swallowing problems Apnea/Choking episodes Progressive neurologic degeneration Parkinsonian manifestations Gaucher is a progressive, debilitating disease 7 Affects 1:50,000–100,000 people Sidransky, 2004; MedlinePlus: Gaucher disease, 2022; GBA=Beta-glucocerebroside; Gcase=β-Glucocerebrosidase Gaucher disease Type 1 Type 3 Cardiac valve calcifications Anemia Thrombocytopenia Enlarged liver Enlarged spleen Myoclonic epilepsy Interstitial lung disease pulmonary fibrosis, pulmonary hypertension Bone disease Bone pain & bone crisis Osteoporosis Pathologic fractures, joint collapse Osteonecrosis Motor abnormalities Hypertonia Hyper-reflexia Dystonia (kyphosis) Myoclonus Variable cognitive deficits Behavioral symptomology Seizures Cerebellar Gait ataxia Tremor Lymphadenopathy More than 300 disease-related mutations identified in the GBA1 gene Autosomal recessive trait affecting lyso somal enzyme β-Glucocerebrosidase (GCase) Biallelic mutations impair GCase activity and result in substrate engorged macrophages (“Gaucher cells”) Gaucher cells accumulate and trigger proinflammatory cascade in affected organs and tissues Other disease impacts: High burden of illness Chronic fatigue and pain Failure to thrive, growth retardation Decreased life expectancy High treatment burden Significant unmet need on SOC
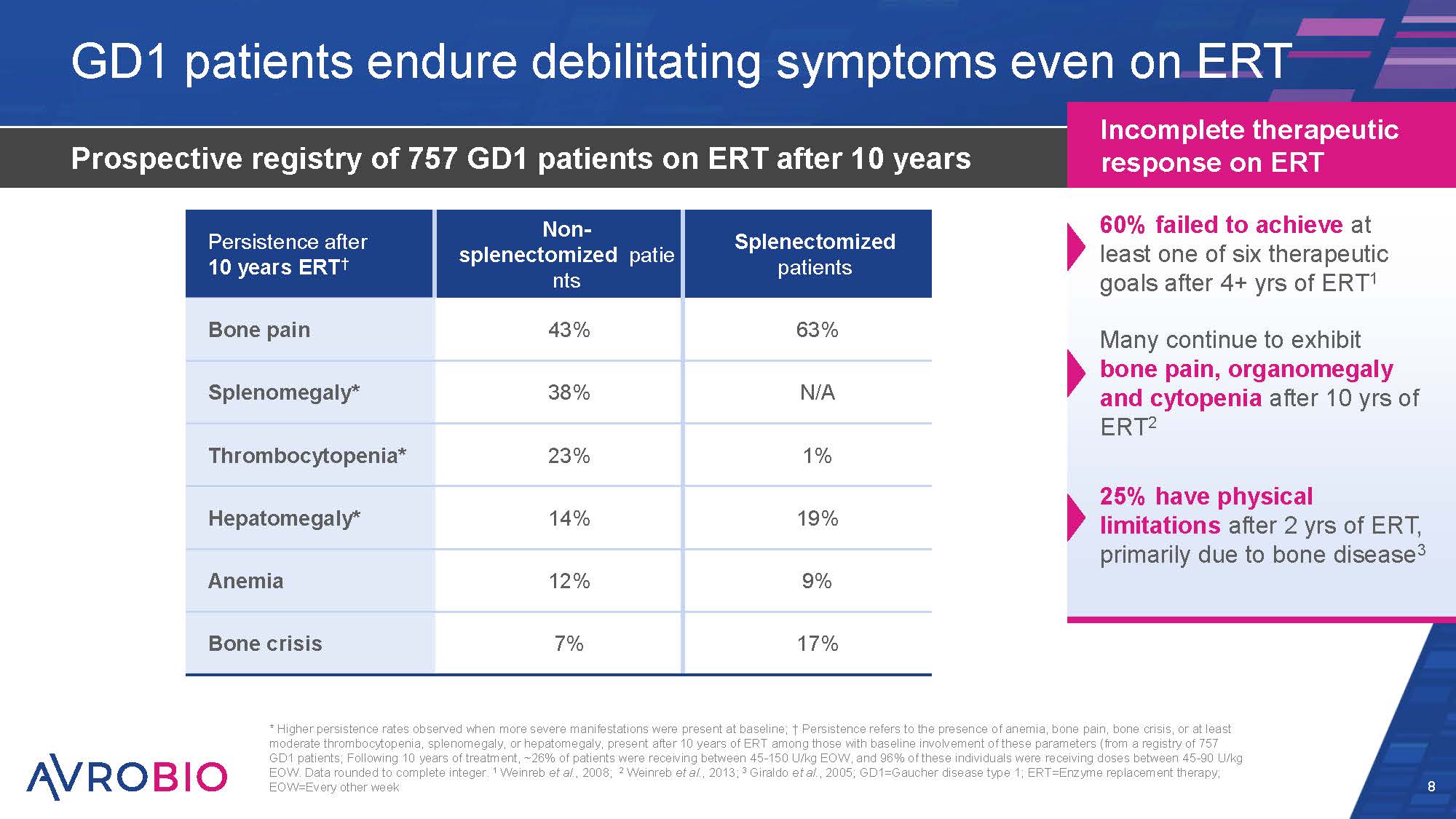
GD1 patients endure debilitating symptoms even on ERT Prospective registry of 757 GD1 patients on ERT after 10 years 8 Incomplete therapeutic response on ERT 60% failed to achieve at least one of six therapeutic goals after 4+ yrs of ERT1 Many continue to exhibit bone pain, organomegaly and cytopenia after 10 yrs of ERT2 25% have physical limitations after 2 yrs of ERT, primarily due to bone disease3 * Higher persistence rates observed when more severe manifestations were present at baseline; † Persistence refers to the presence of anemia, bone pain, bone crisis, or at least moderate thrombocytopenia, splenomegaly, or hepatomegaly, present after 10 years of ERT among those with baseline involvement of these parameters (from a registry of 757 GD1 patients; Following 10 years of treatment, ~26% of patients were receiving between 45-150 U/kg EOW, and 96% of these individuals were receiving doses between 45-90 U/kg EOW. Data rounded to complete integer. 1 Weinreb et al., 2008; 2 Weinreb et al., 2013; 3 Giraldo et al., 2005; GD1=Gaucher disease type 1; ERT=Enzyme replacement therapy; EOW=Every other week Persistence after 10 years ERT† Non- splenectomized patie nts 43% Splenectomized patients 63% Bone pain Splenomegaly* 38% N/A Thrombocytopenia* 23% 1% Hepatomegaly* 14% 19% Anemia 12% 9% Bone crisis 7% 17%
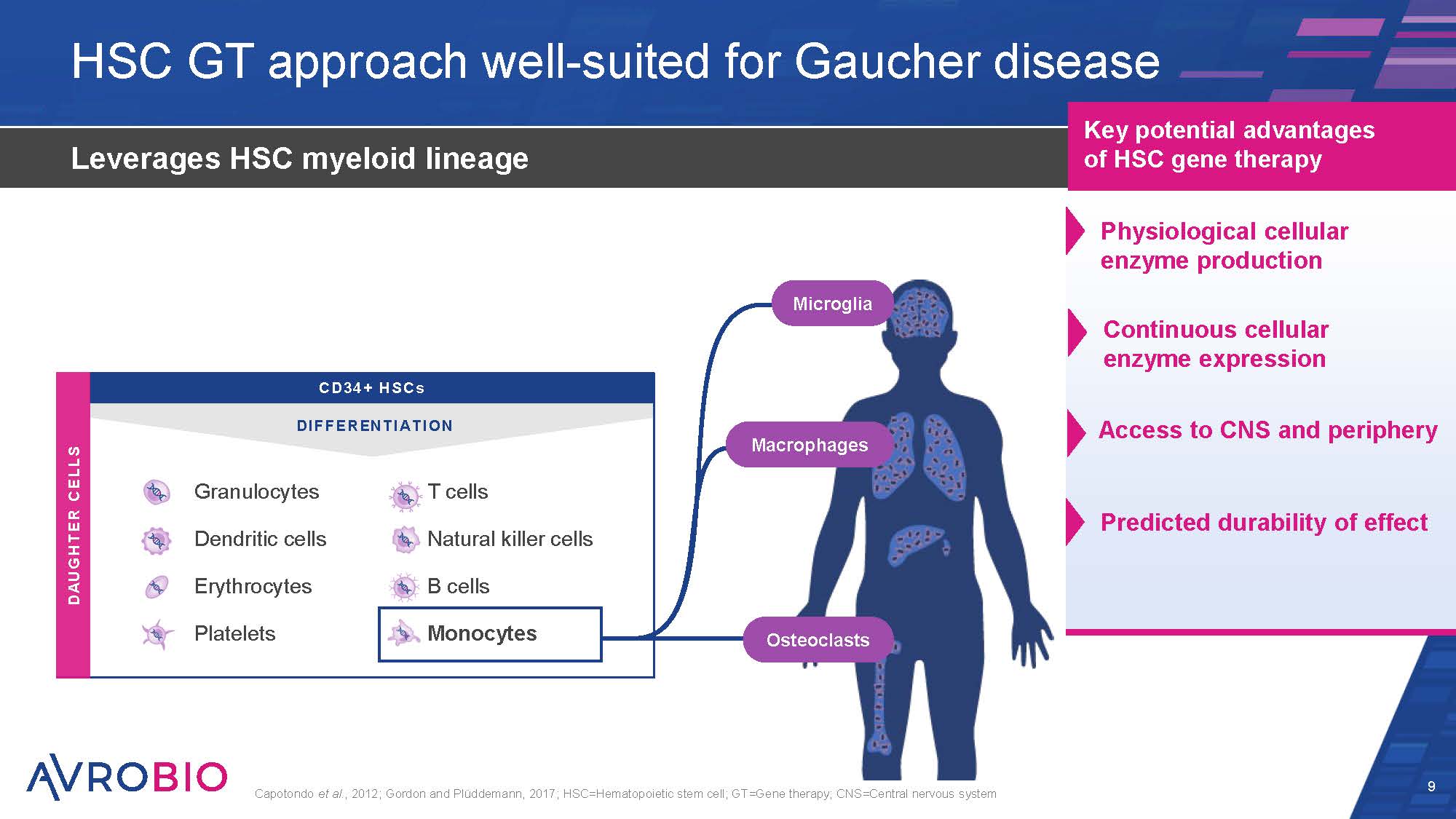
HSC GT approach well-suited for Gaucher disease 9 Leverages HSC myeloid lineage Capotondo et al., 2012; Gordon and Plüddemann, 2017; HSC=Hematopoietic stem cell; GT=Gene therapy; CNS=Central nervous system Key potential advantages of HSC gene therapy T cells Natural killer cells B cells Monocytes CD34 + HSCs DIFFERENTIATION DAUGHTER CELLS Erythrocytes Platelets Granulocytes Dendritic cells Osteoclasts Microglia Macrophages Physiological cellular enzyme production Continuous cellular enzyme expression Access to CNS and periphery Predicted durability of effect
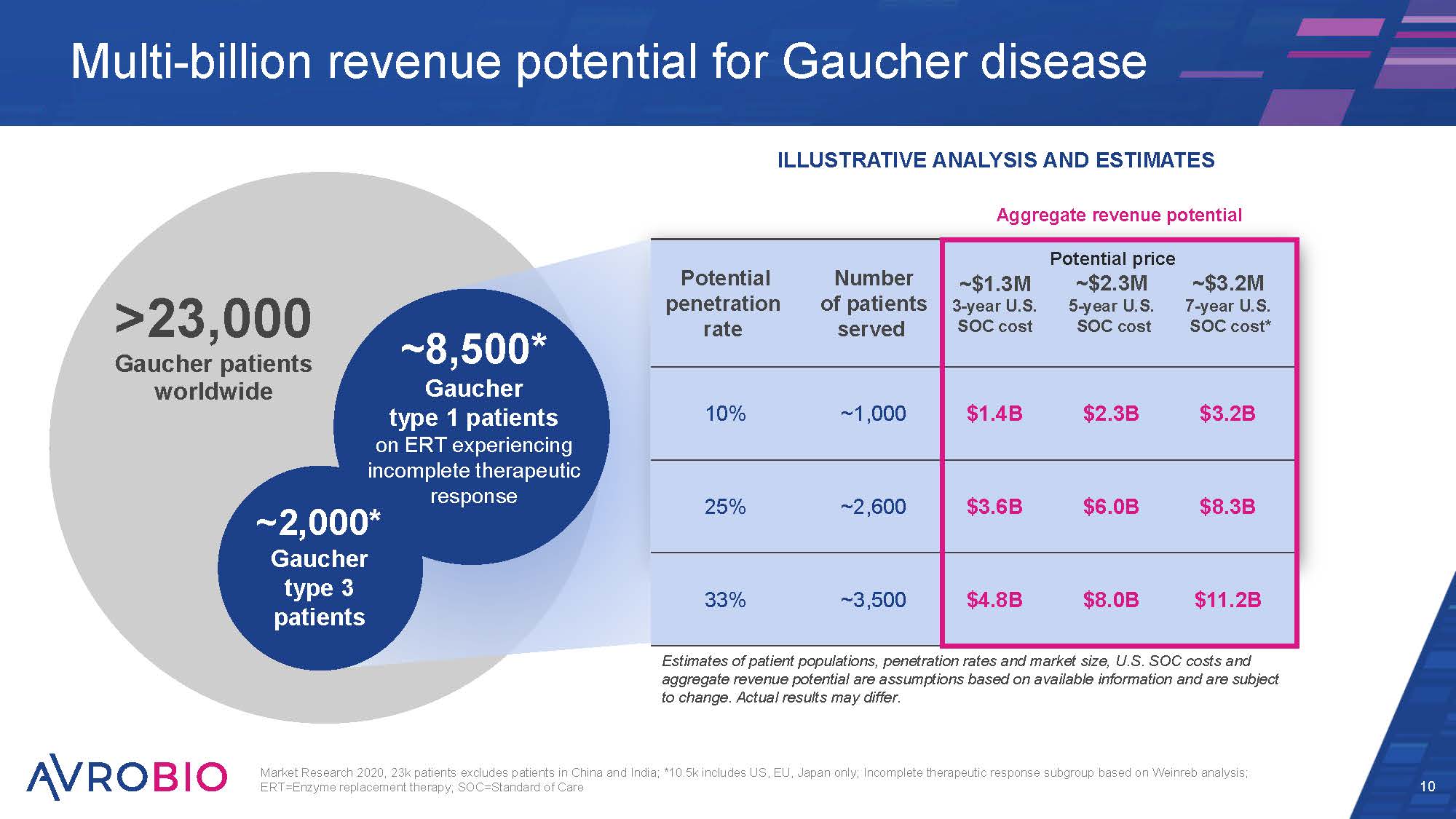
Market Research 2020, 23k patients excludes patients in China and India; *10.5k includes US, EU, Japan only; Incomplete therapeutic response subgroup based on Weinreb analysis; ERT=Enzyme replacement therapy; SOC=Standard of Care 10 Multi-billion revenue potential for Gaucher disease >23,000 Gaucher patients worldwide ~2,000* Gaucher type 3 patients ~8,500* Gaucher type 1 patients on ERT experiencing incomplete therapeutic response ILLUSTRATIVE ANALYSIS AND ESTIMATES Aggregate revenue potential Potential penetration rate Number of patients served ~$1.3M 3-year U.S. SOC cost Potential pric ~$2.3M 5-year U.S. SOC cost e ~$3.2M 7-year U.S. SOC cost* 10% ~1,000 $1.4B $2.3B $3.2B 25% ~2,600 $3.6B $6.0B $8.3B 33% ~3,500 $4.8B $8.0B $11.2B Estimates of patient populations, penetration rates and market size, U.S. SOC costs and aggregate revenue potential are assumptions based on available information and are subject to change. Actual results may differ.
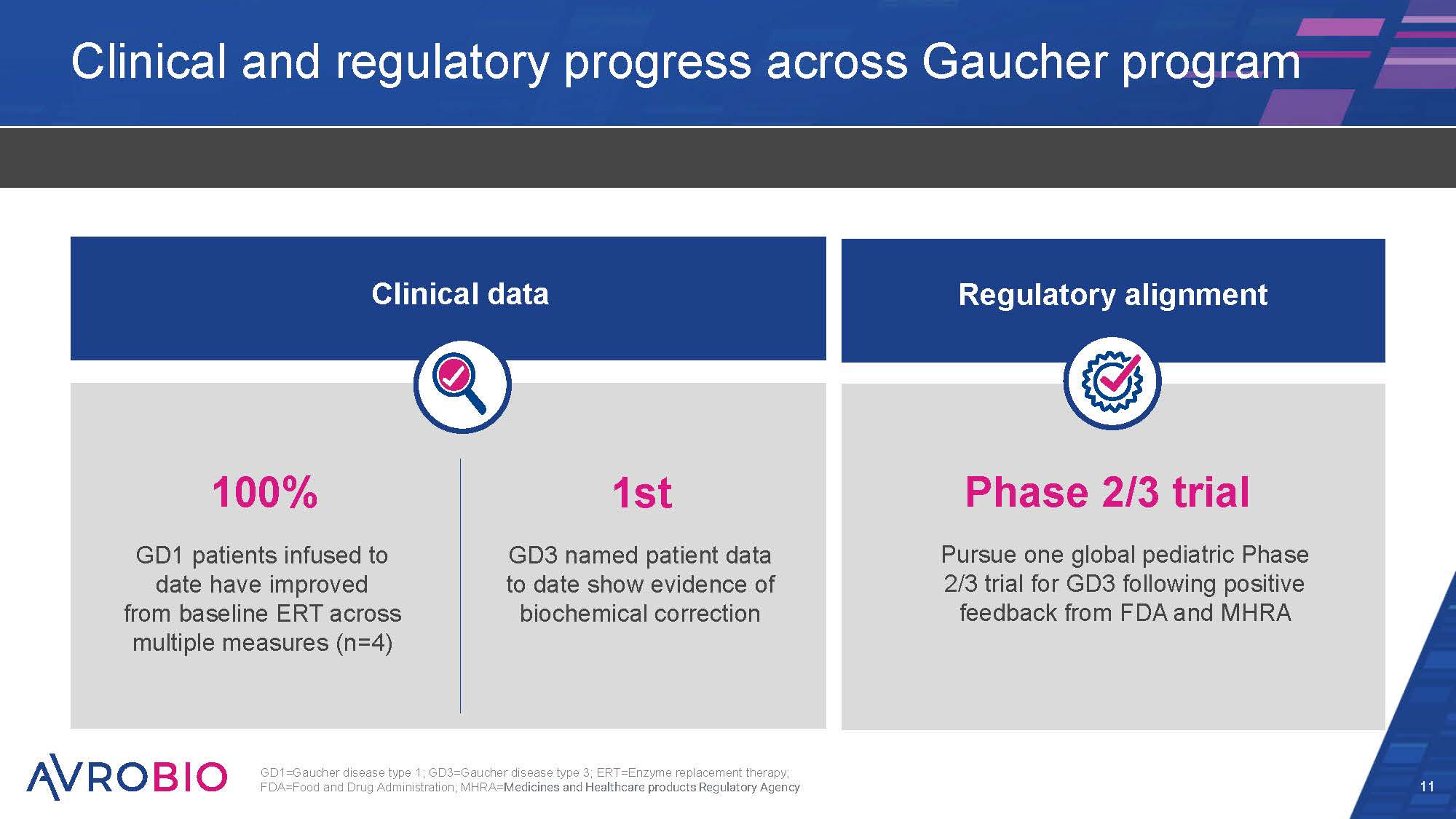
Clinical and regulatory progress across Gaucher program 11 100% GD1 patients infused to date have improved from baseline ERT across multiple measures (n=4) 1st GD3 named patient data to date show evidence of biochemical correction Phase 2/3 trial Pursue one global pediatric Phase 2/3 trial for GD3 following positive feedback from FDA and MHRA Clinical data Regulatory alignment GD1=Gaucher disease type 1; GD3=Gaucher disease type 3; ERT=Enzyme replacement therapy; FDA=Food and Drug Administration; MHRA=Medicines and Healthcare products Regulatory Agency

Today’s agenda 12 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

Timothy M. Cox, M.D., MAE, FRCP, FMedSci Professor, University of Cambridge; Cambridge University Hospitals UK Perspective from leading KOLs 13 Simon Jones, M.D., BSc, MRCPCH Professor, Pediatric Inherited Metabolic Diseases at the Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, Manchester University NHS Foundation Trust Robert Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath Professor, Pediatric Hematology at Royal Manchester Children’s Hospital, Manchester University NHS Foundation Trust Profs. Cox, Wynn and Jones are consultants to AVROBIO; KOL: Key Opinion Leader

An Introduction to Gaucher disease Timothy M Cox Department of Medicine University of Cambridge Addenbrooke’s Hospital Cambridge University NHS Hospitals Trust

Dr Gaucher (1854-1918) SV ♀ aged 34 years Splenomegaly from 7 Bleeding and pain Swollen abdomen Necropsy (6 April 1881) Cachexia (31 Kg) Spleen: 4.77 Kg Liver: 3.88 Kg 1882
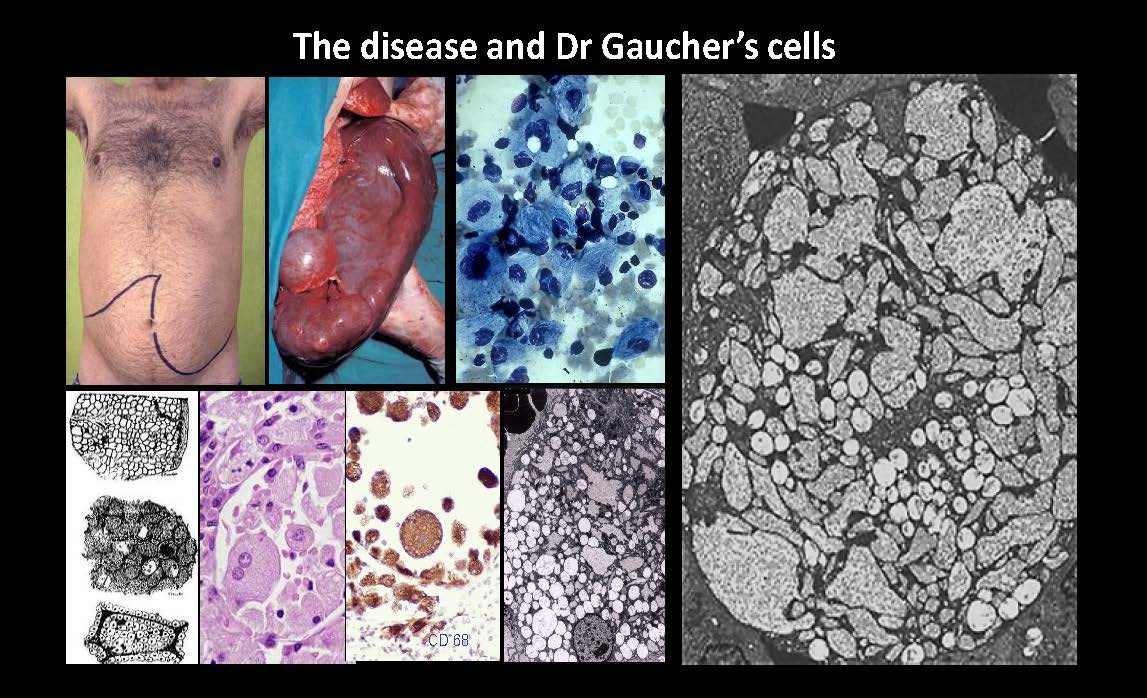
The disease and Dr Gaucher’s cells CD 68
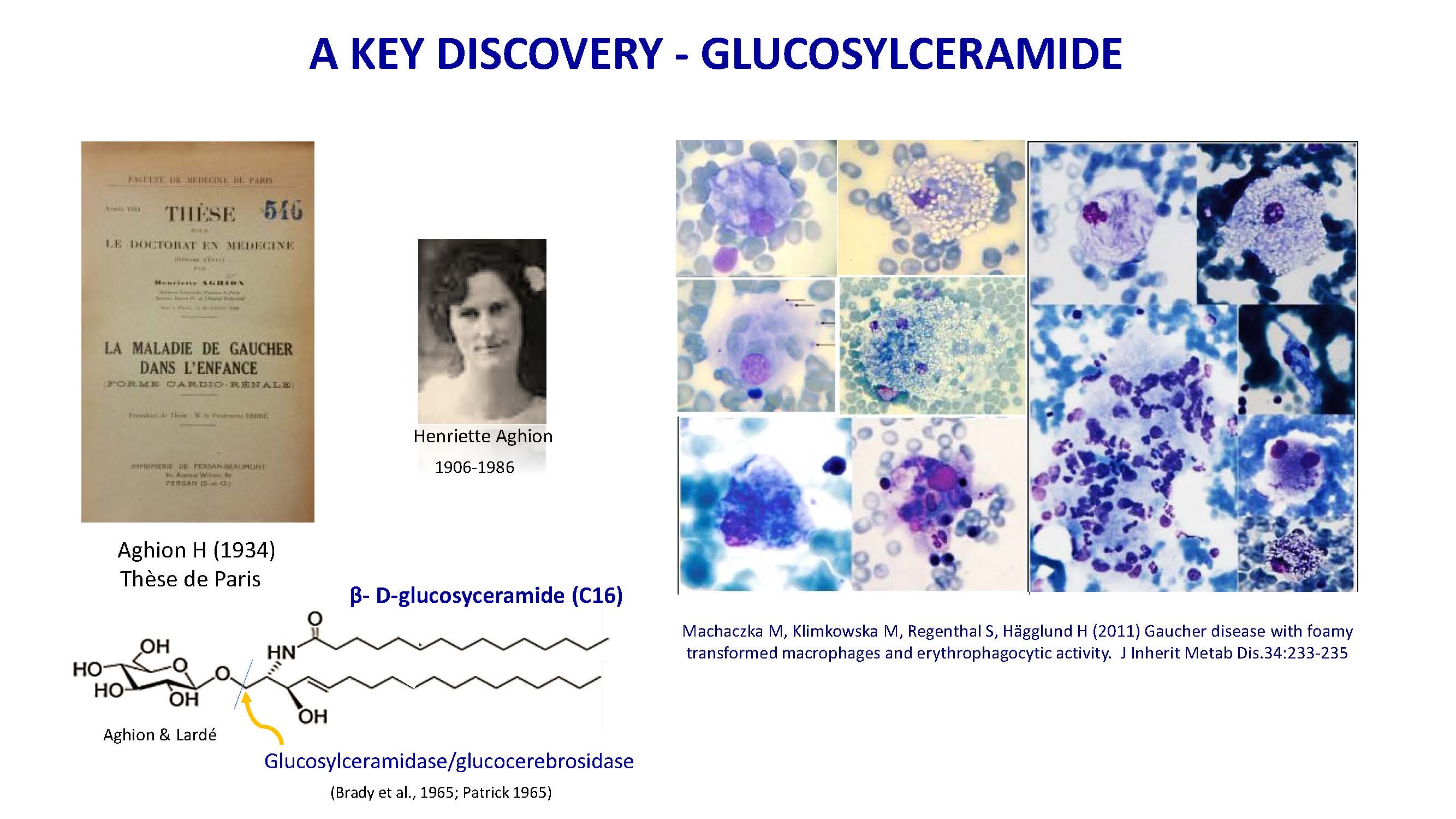
A KEY DISCOVERY - GLUCOSYLCERAMIDE Machaczka M, Klimkowska M, Regenthal S, Hägglund H (2011) Gaucher disease with foamy transformed macrophages and erythrophagocytic activity. J Inherit Metab Dis.34:233-235 Henriette Aghion 1906-1986 β- D-glucosyceramide (C16) Aghion & Lardé Aghion H (1934) Thèse de Paris Glucosylceramidase/glucocerebrosidase (Brady et al., 1965; Patrick 1965)
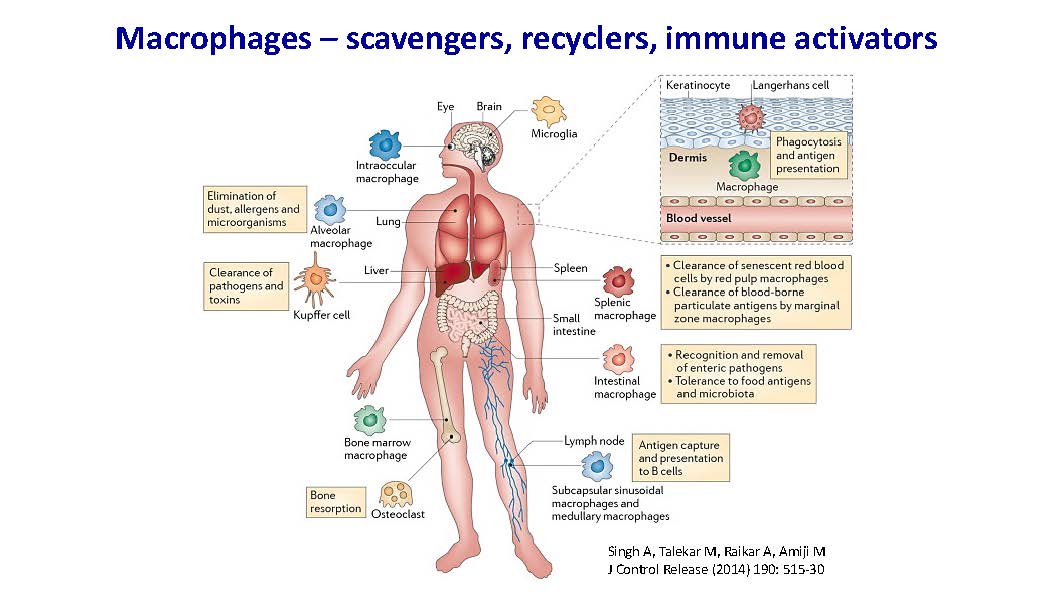
Macrophages – scavengers, recyclers, immune activators Singh A, Talekar M, Raikar A, Amiji M J Control Release (2014) 190: 515-30
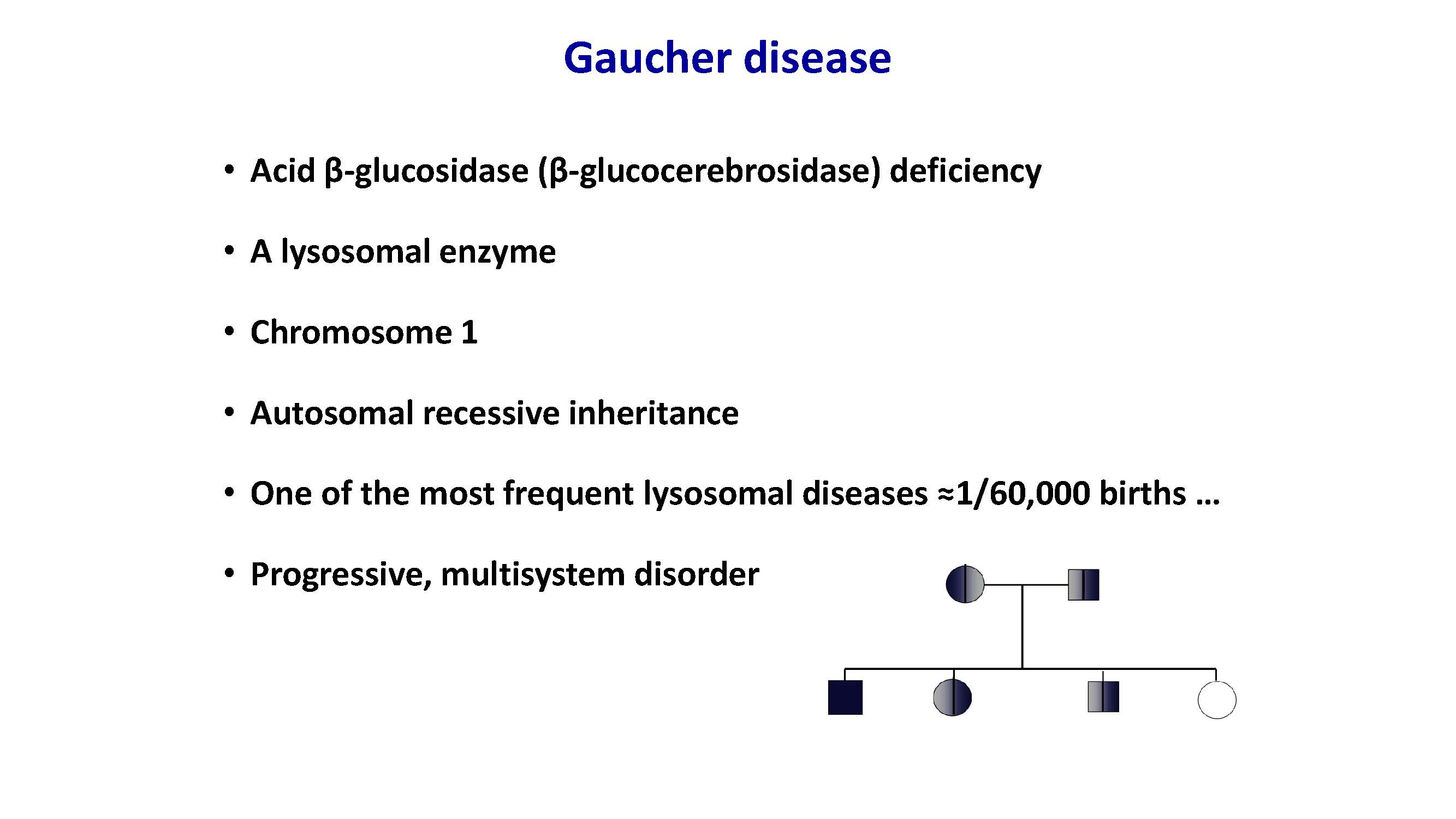
Gaucher disease Acid β-glucosidase (β-glucocerebrosidase) deficiency A lysosomal enzyme Chromosome 1 Autosomal recessive inheritance One of the most frequent lysosomal diseases ≈1/60,000 births … Progressive, multisystem disorder
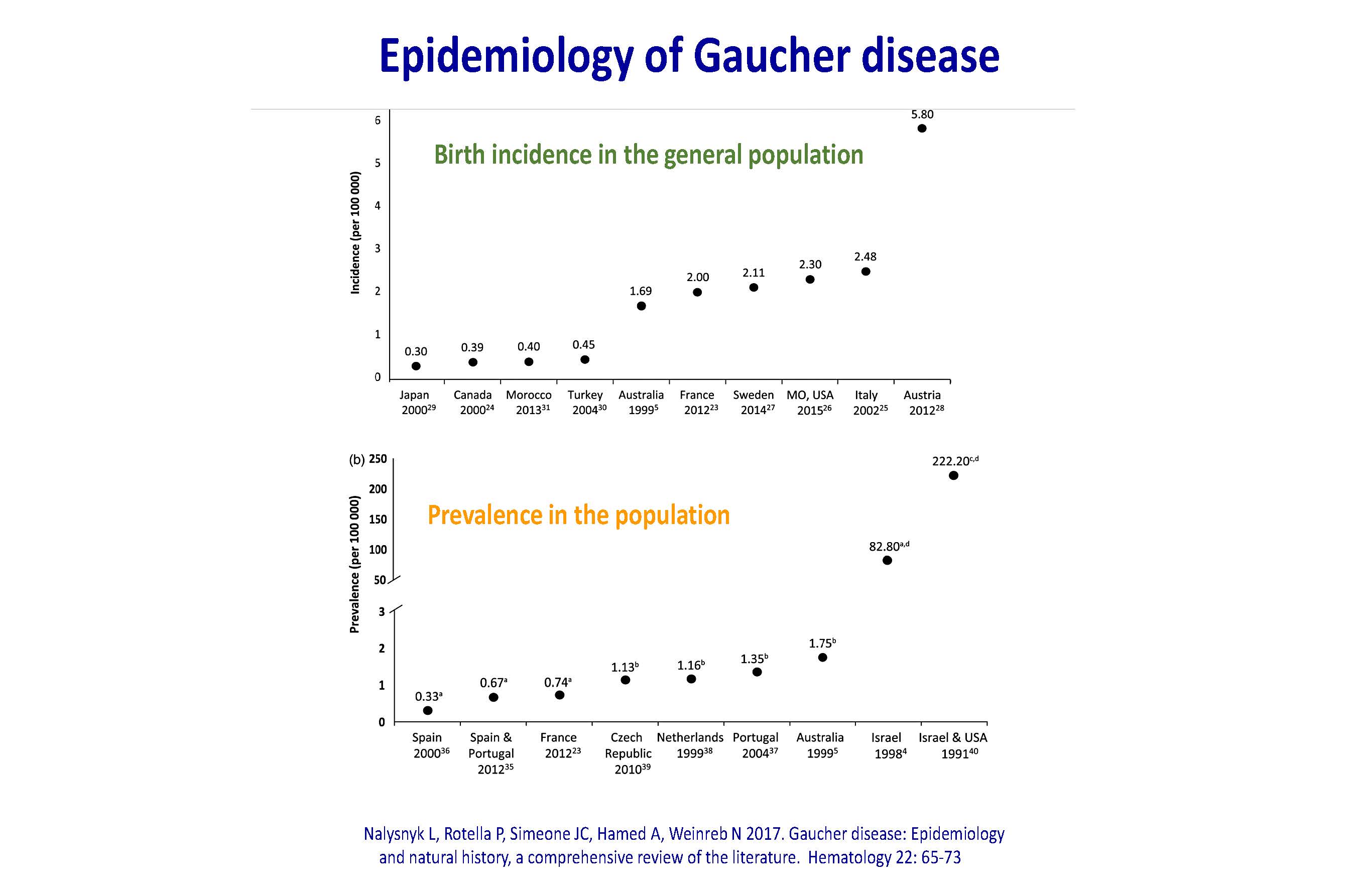
Epidemiology of Gaucher disease Nalysnyk L, Rotella P, Simeone JC, Hamed A, Weinreb N 2017. Gaucher disease: Epidemiology and natural history, a comprehensive review of the literature. Hematology 22: 65-73 Birth incidence in the general population Prevalence in the population
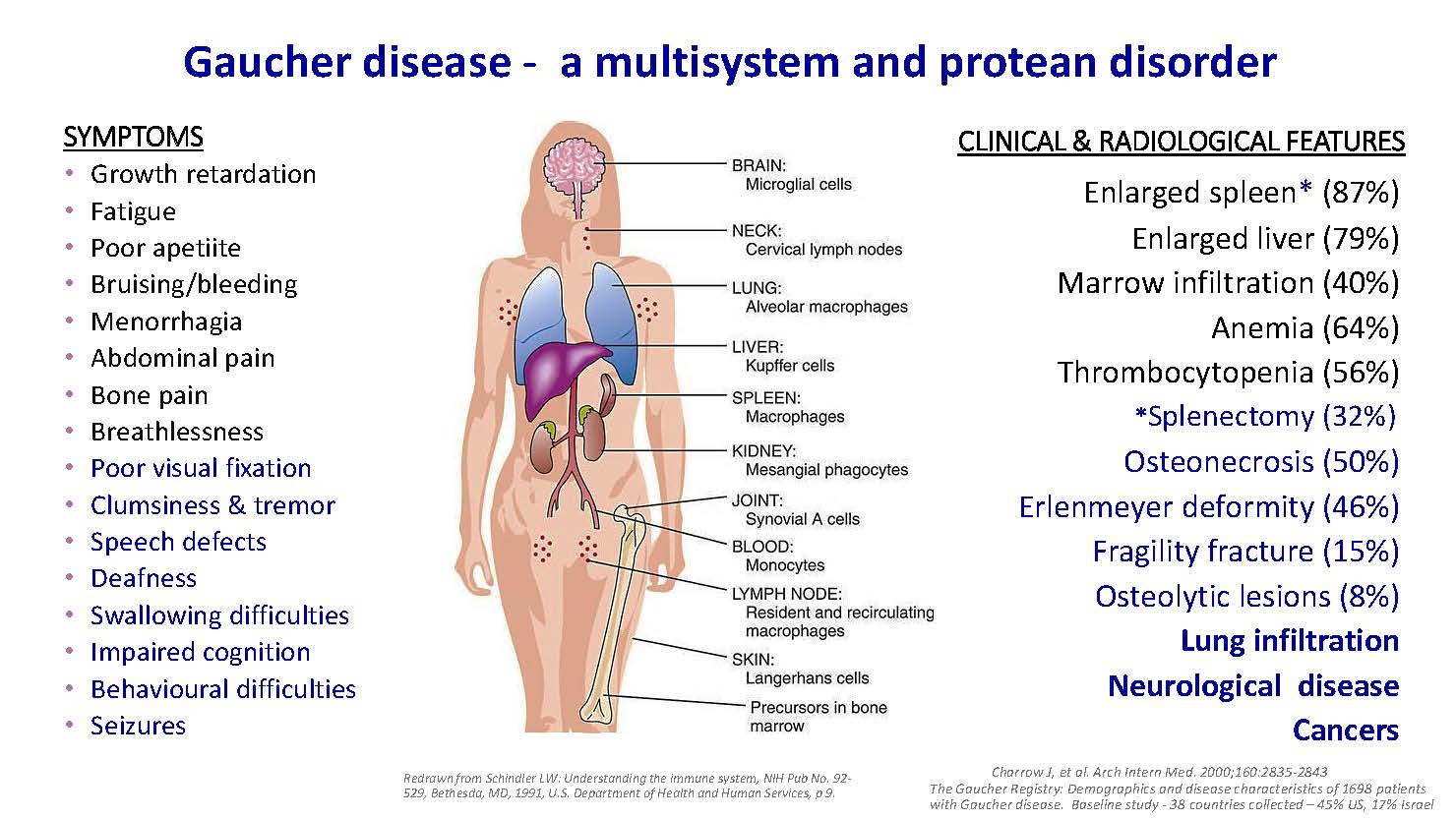
Gaucher disease - a multisystem and protean disorder SYMPTOMS Growth retardation Fatigue Poor apetiite Bruising/bleeding Menorrhagia Abdominal pain Bone pain Breathlessness Poor visual fixation Clumsiness & tremor Speech defects Deafness Swallowing difficulties Impaired cognition Behavioural difficulties Seizures CLINICAL & RADIOLOGICAL FEATURES Enlarged spleen* (87%) Enlarged liver (79%) Marrow infiltration (40%) Anemia (64%) Thrombocytopenia (56%) *Splenectomy (32%) Osteonecrosis (50%) Erlenmeyer deformity (46%) Fragility fracture (15%) Osteolytic lesions (8%) Lung infiltration Neurological disease Cancers Charrow J, et al. Arch Intern Med. 2000;160:2835-2843 The Gaucher Registry: Demographics and disease characteristics of 1698 patients with Gaucher disease. Baseline study - 38 countries collected – 45% US, 17% Israel Redrawn from Schindler LW: Understanding the immune system, NIH Pub No. 92- 529, Bethesda, MD, 1991, U.S. Department of Health and Human Services, p 9.
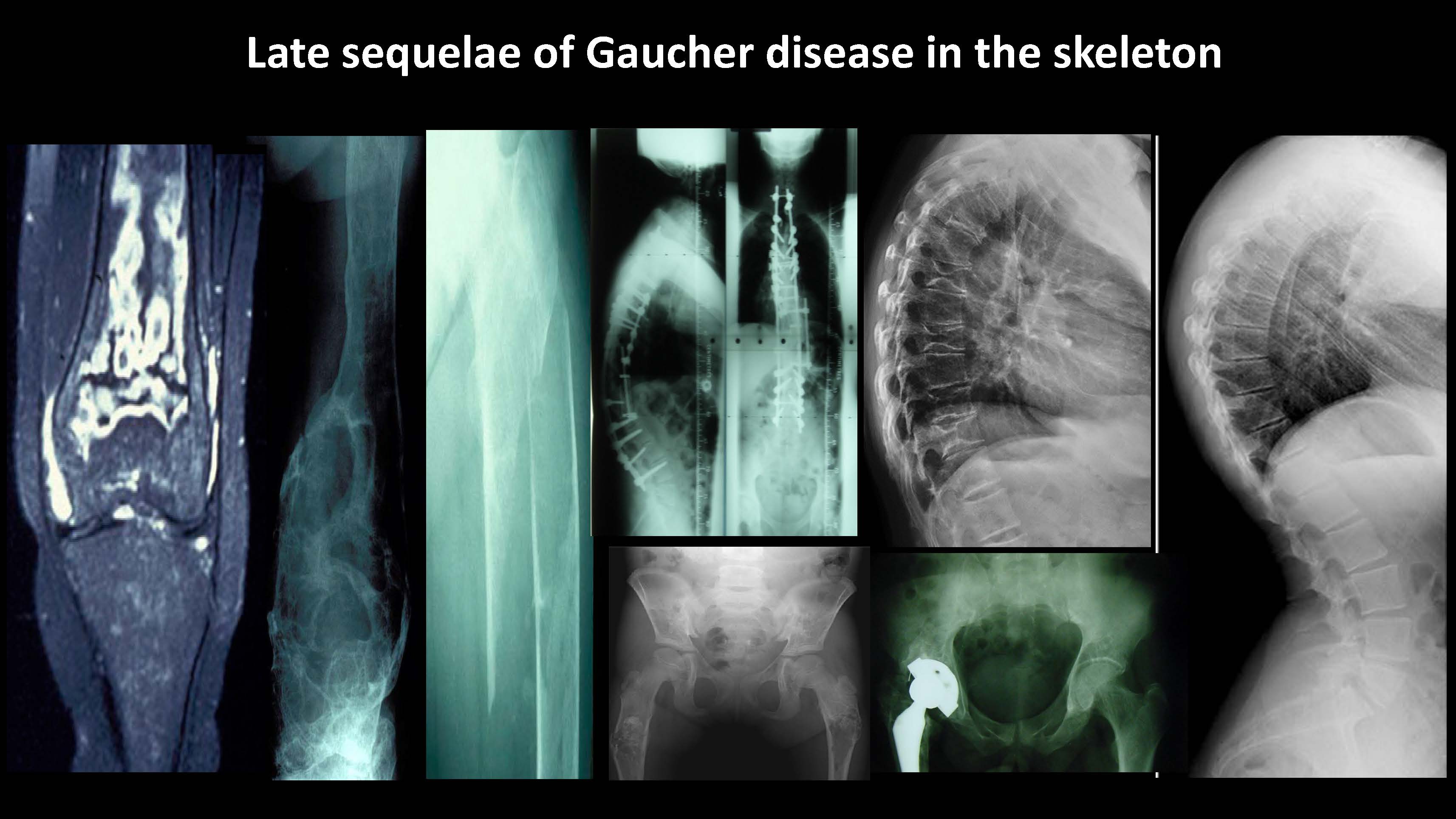
Late sequelae of Gaucher disease in the skeleton
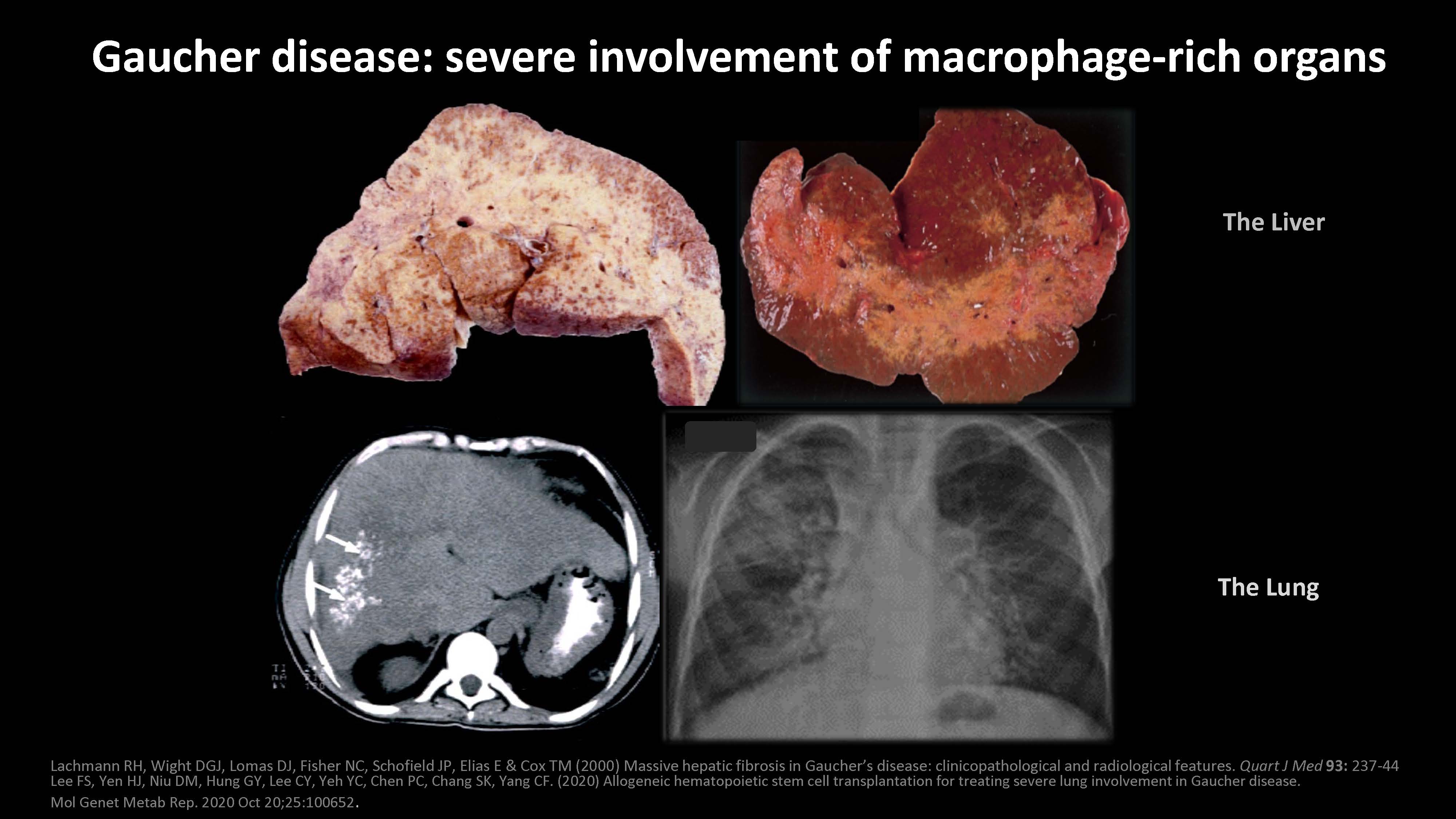
Lachmann RH, Wight DGJ, Lomas DJ, Fisher NC, Schofield JP, Elias E & Cox TM (2000) Massive hepatic fibrosis in Gaucher’s disease: clinicopathological and radiological features. Quart J Med 93: 237-44 Lee FS, Yen HJ, Niu DM, Hung GY, Lee CY, Yeh YC, Chen PC, Chang SK, Yang CF. (2020) Allogeneic hematopoietic stem cell transplantation for treating severe lung involvement in Gaucher disease. Mol Genet Metab Rep. 2020 Oct 20;25:100652. Gaucher disease: severe involvement of macrophage-rich organs The Liver The Lung
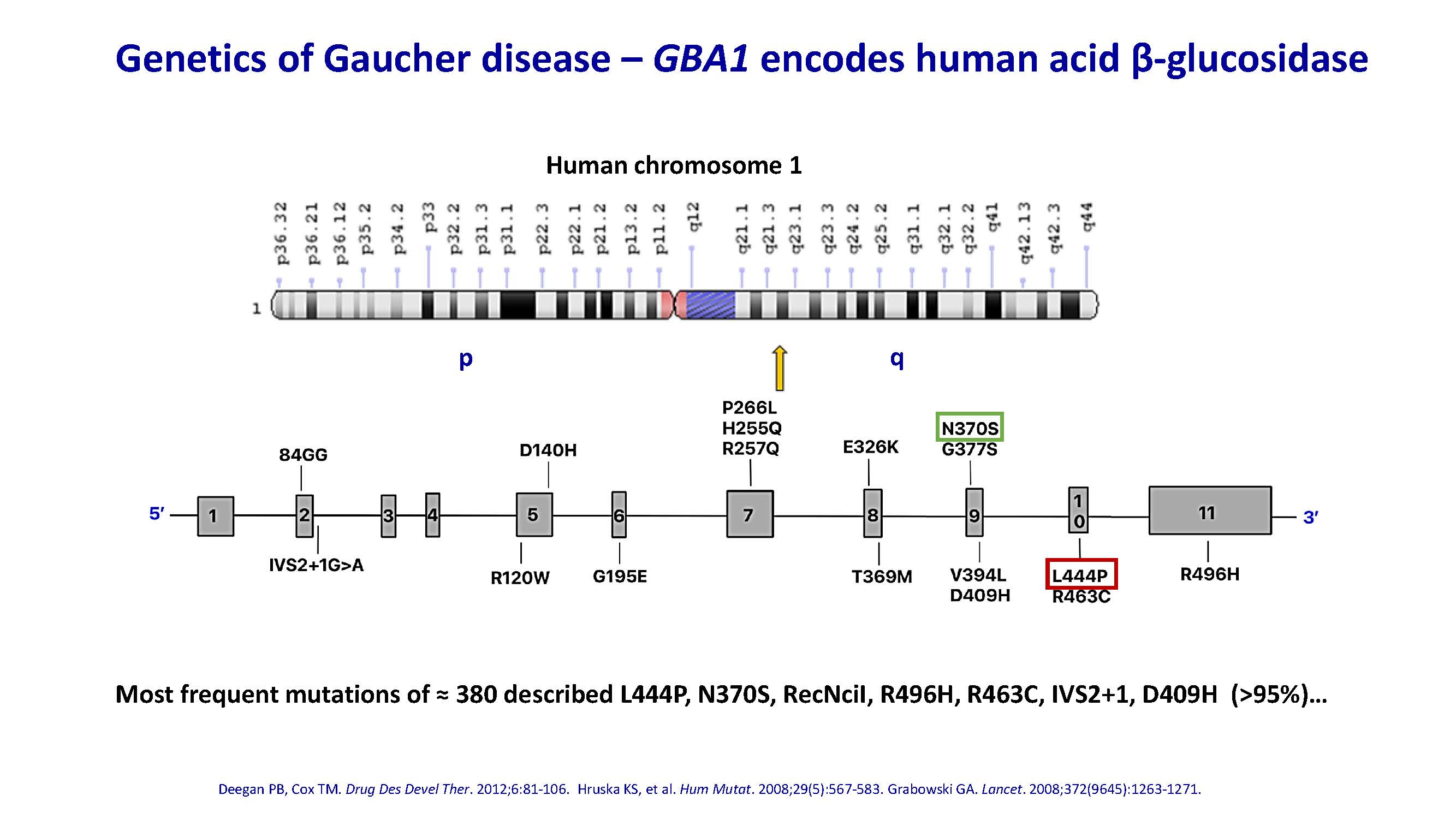
Genetics of Gaucher disease – GBA1 encodes human acid β-glucosidase Deegan PB, Cox TM. Drug Des Devel Ther. 2012;6:81-106. Hruska KS, et al. Hum Mutat. 2008;29(5):567-583. Grabowski GA. Lancet. 2008;372(9645):1263-1271. Most frequent mutations of ≈ 380 described L444P, N370S, RecNciI, R496H, R463C, IVS2+1, D409H (>95%)… Human chromosome 1 p q
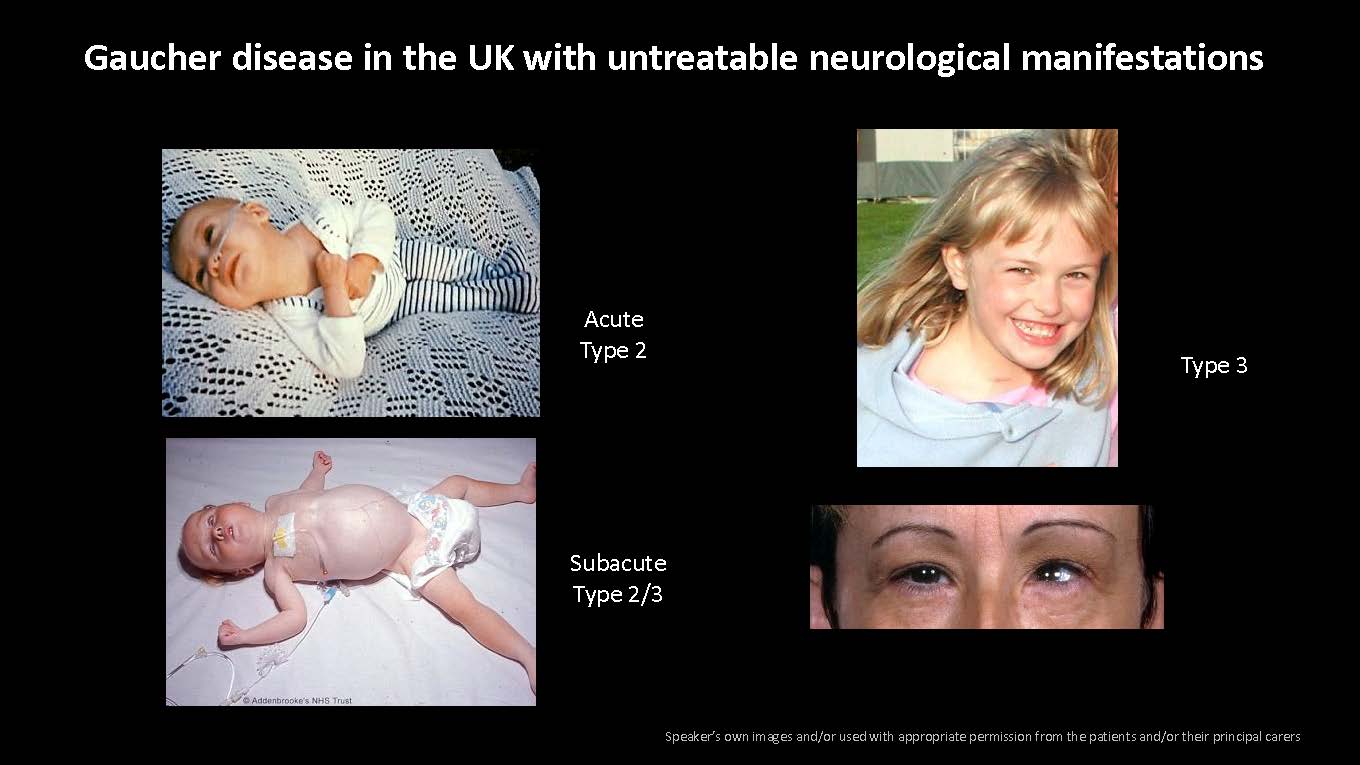
Gaucher disease in the UK with untreatable neurological manifestations Speaker’s own images and/or used with appropriate permission from the patients and/or their principal carers Acute Type 2 Subacute Type 2/3 Type 3
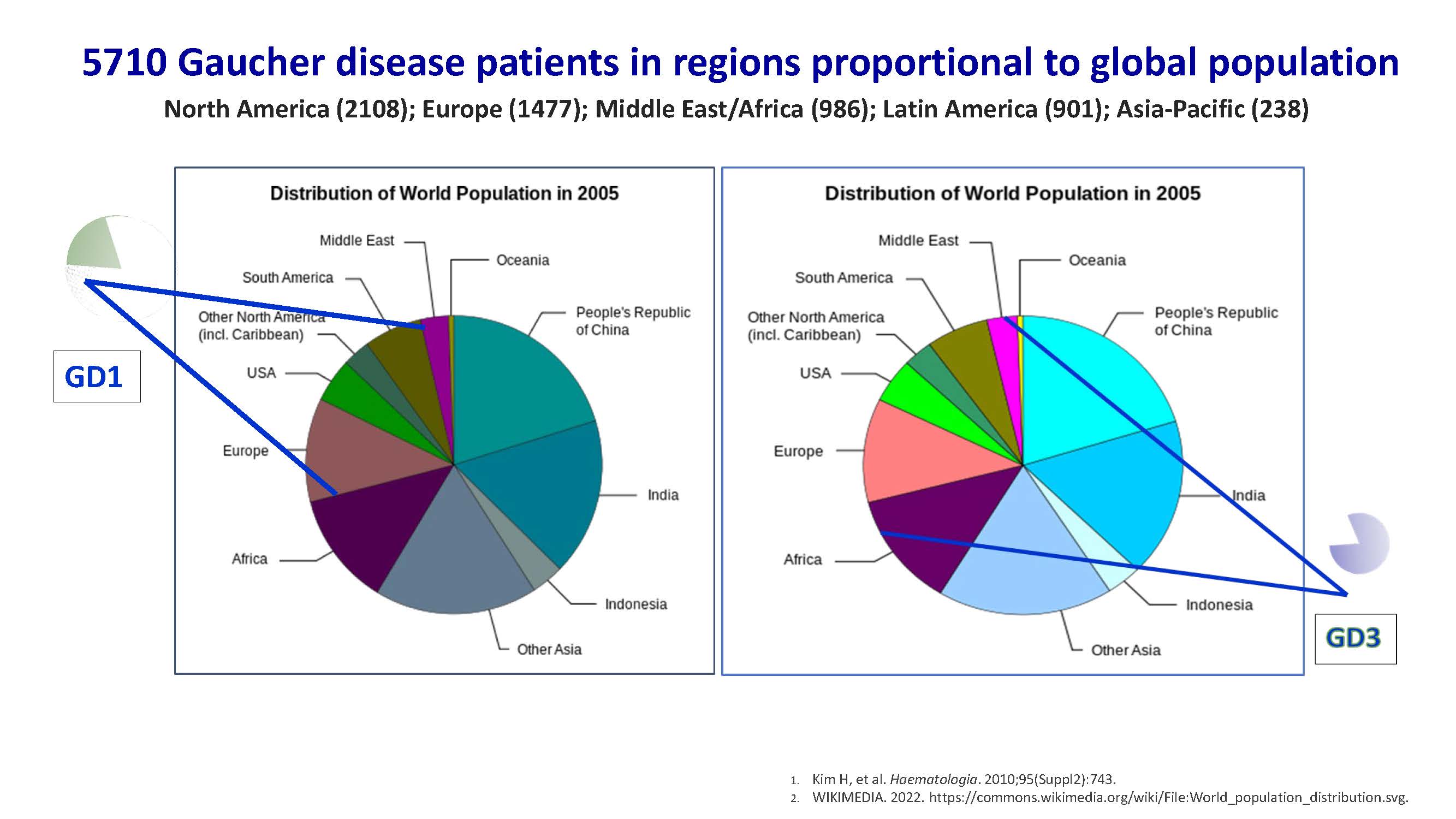
5710 Gaucher disease patients in regions proportional to global population North America (2108); Europe (1477); Middle East/Africa (986); Latin America (901); Asia-Pacific (238) Kim H, et al. Haematologia. 2010;95(Suppl2):743. WIKIMEDIA. 2022. https://commons.wikimedia.org/wiki/File:World_population_distribution.svg. GD1
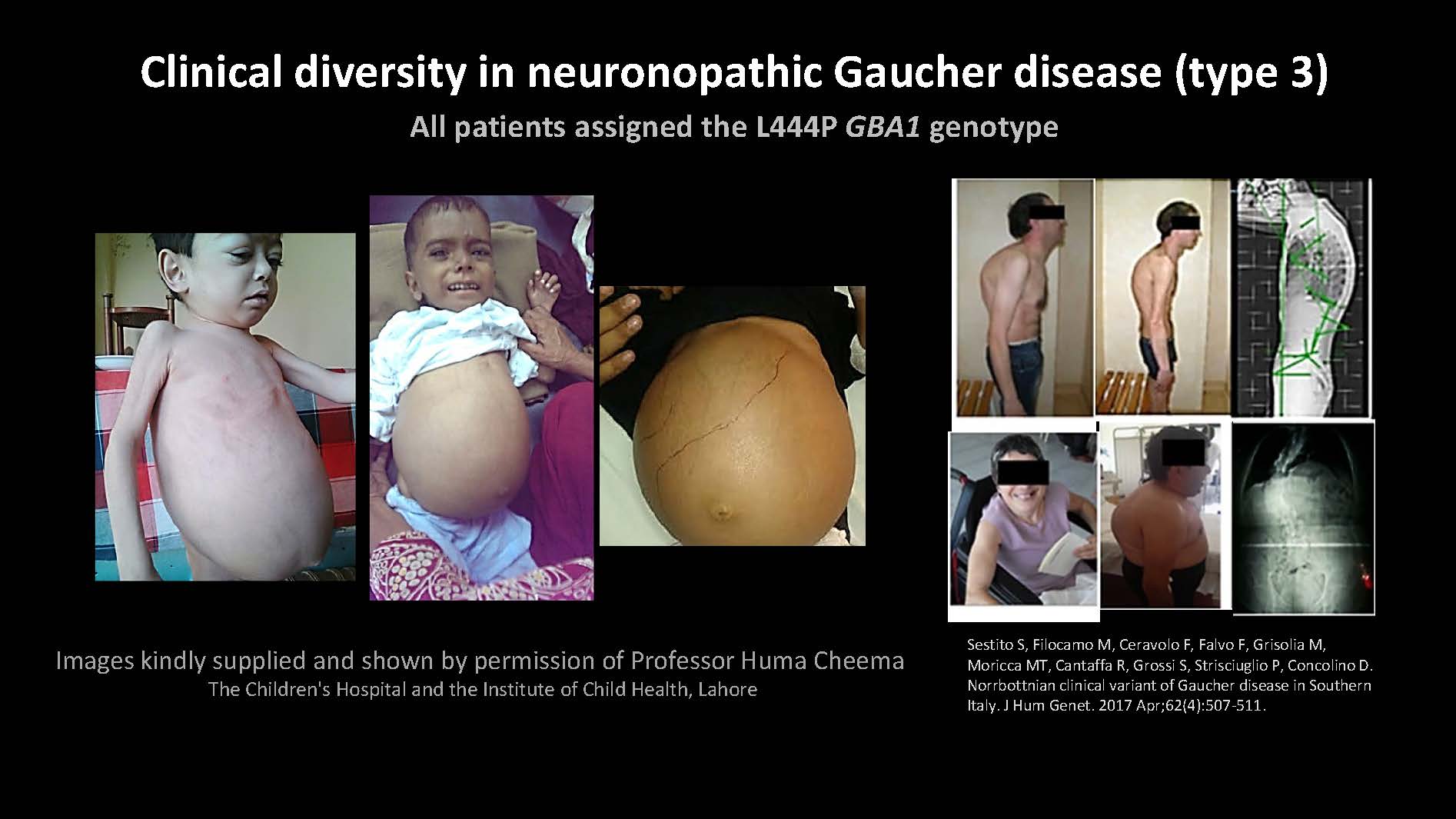
Sestito S, Filocamo M, Ceravolo F, Falvo F, Grisolia M, Moricca MT, Cantaffa R, Grossi S, Strisciuglio P, Concolino D. Norrbottnian clinical variant of Gaucher disease in Southern Italy. J Hum Genet. 2017 Apr;62(4):507-511. Images kindly supplied and shown by permission of Professor Huma Cheema The Children's Hospital and the Institute of Child Health, Lahore Clinical diversity in neuronopathic Gaucher disease (type 3) All patients assigned the L444P GBA1 genotype
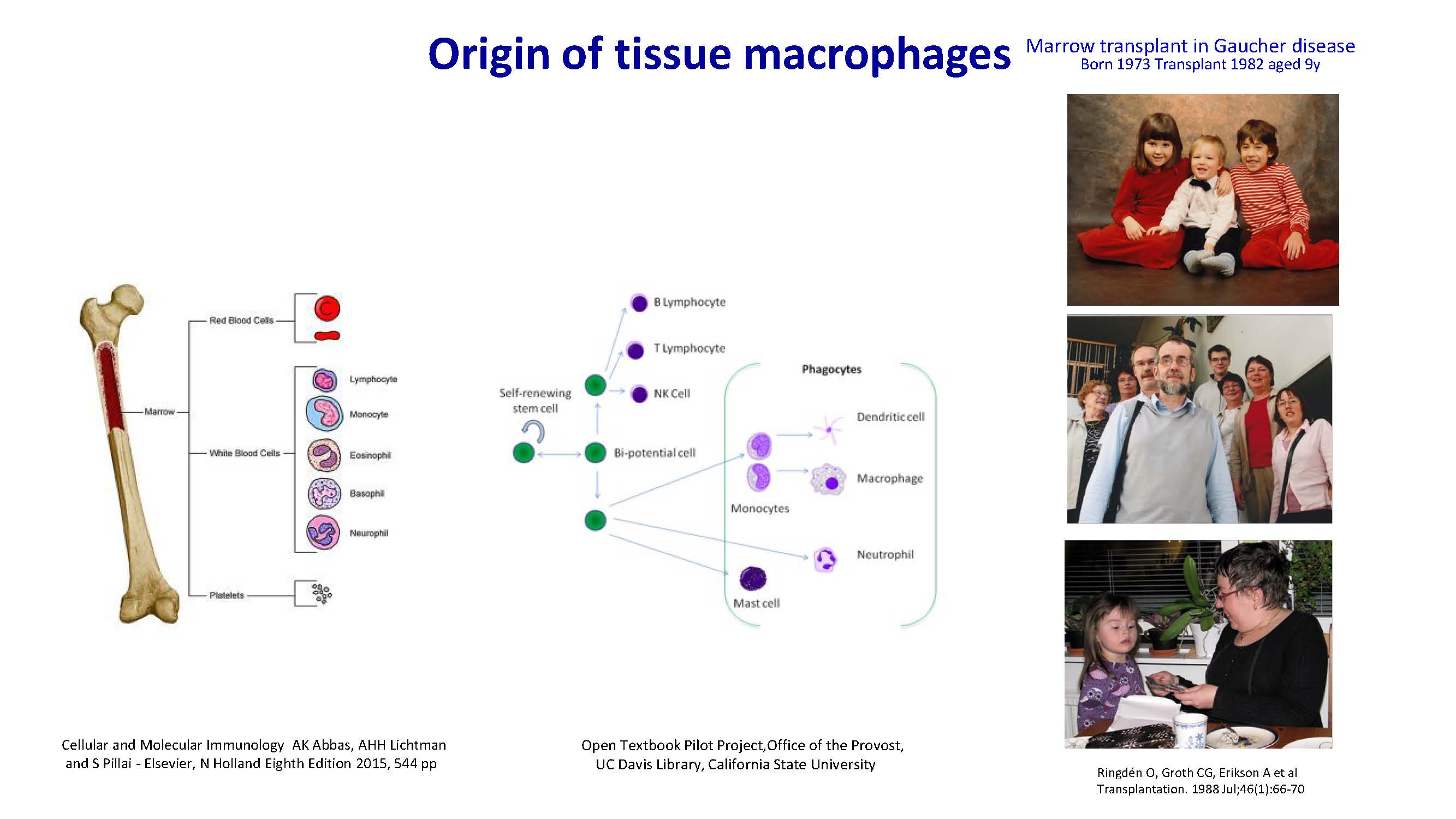
Origin of tissue macrophages Cellular and Molecular Immunology AK Abbas, AHH Lichtman and S Pillai - Elsevier, N Holland Eighth Edition 2015, 544 pp Marrow transplant in Gaucher disease Born 1973 Transplant 1982 aged 9y Ringdén O, Groth CG, Erikson A et al Transplantation. 1988 Jul;46(1):66-70 Open Textbook Pilot Project,Office of the Provost, UC Davis Library, California State University
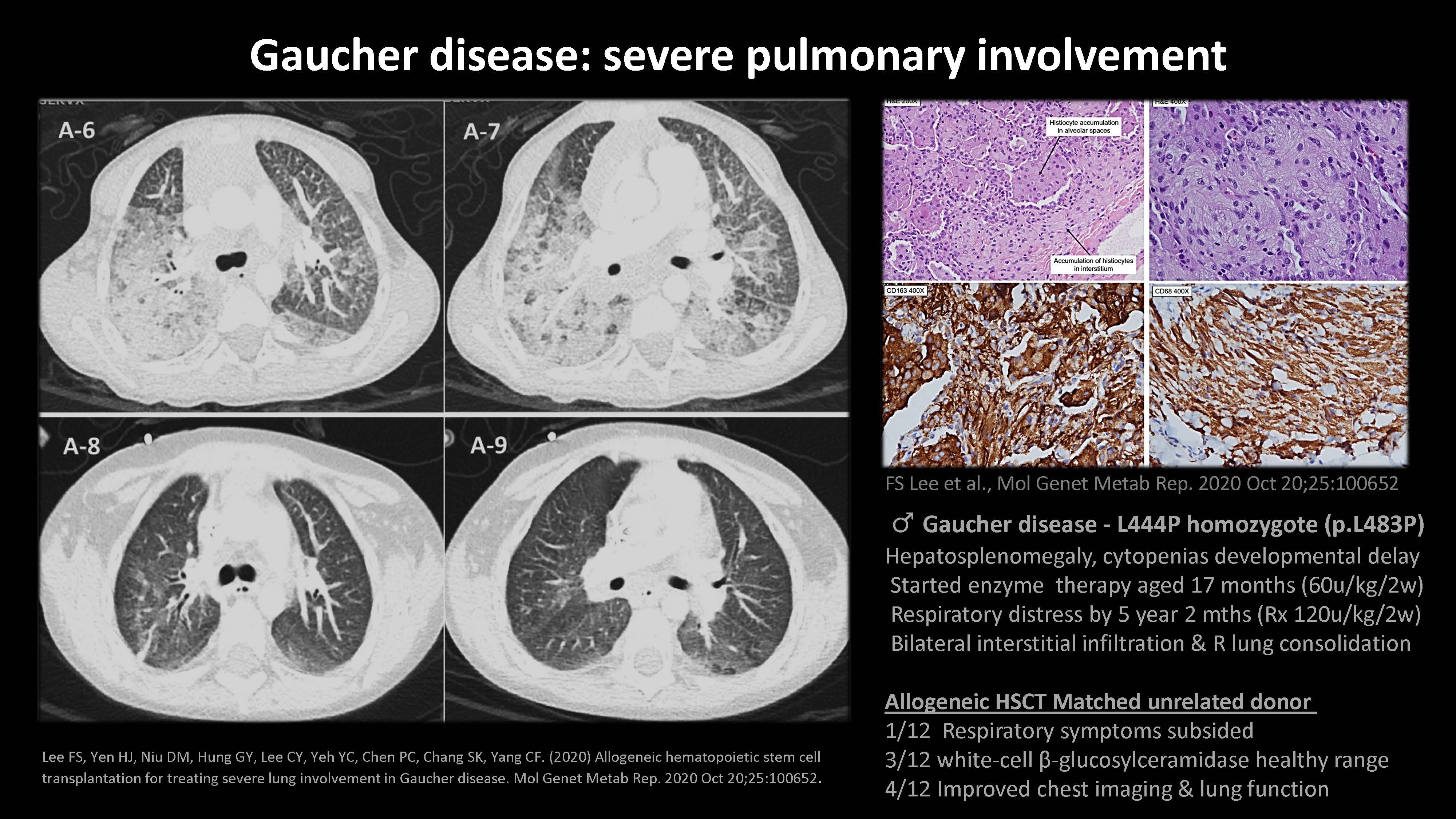
Lee FS, Yen HJ, Niu DM, Hung GY, Lee CY, Yeh YC, Chen PC, Chang SK, Yang CF. (2020) Allogeneic hematopoietic stem cell transplantation for treating severe lung involvement in Gaucher disease. Mol Genet Metab Rep. 2020 Oct 20;25:100652. Gaucher disease: severe pulmonary involvement FS Lee et al., Mol Genet Metab Rep. 2020 Oct 20;25:100652 ♂ Gaucher disease - L444P homozygote (p.L483P) Hepatosplenomegaly, cytopenias developmental delay Started enzyme therapy aged 17 months (60u/kg/2w) Respiratory distress by 5 year 2 mths (Rx 120u/kg/2w) Bilateral interstitial infiltration & R lung consolidation Allogeneic HSCT Matched unrelated donor 1/12 Respiratory symptoms subsided 3/12 white-cell β-glucosylceramidase healthy range 4/12 Improved chest imaging & lung function
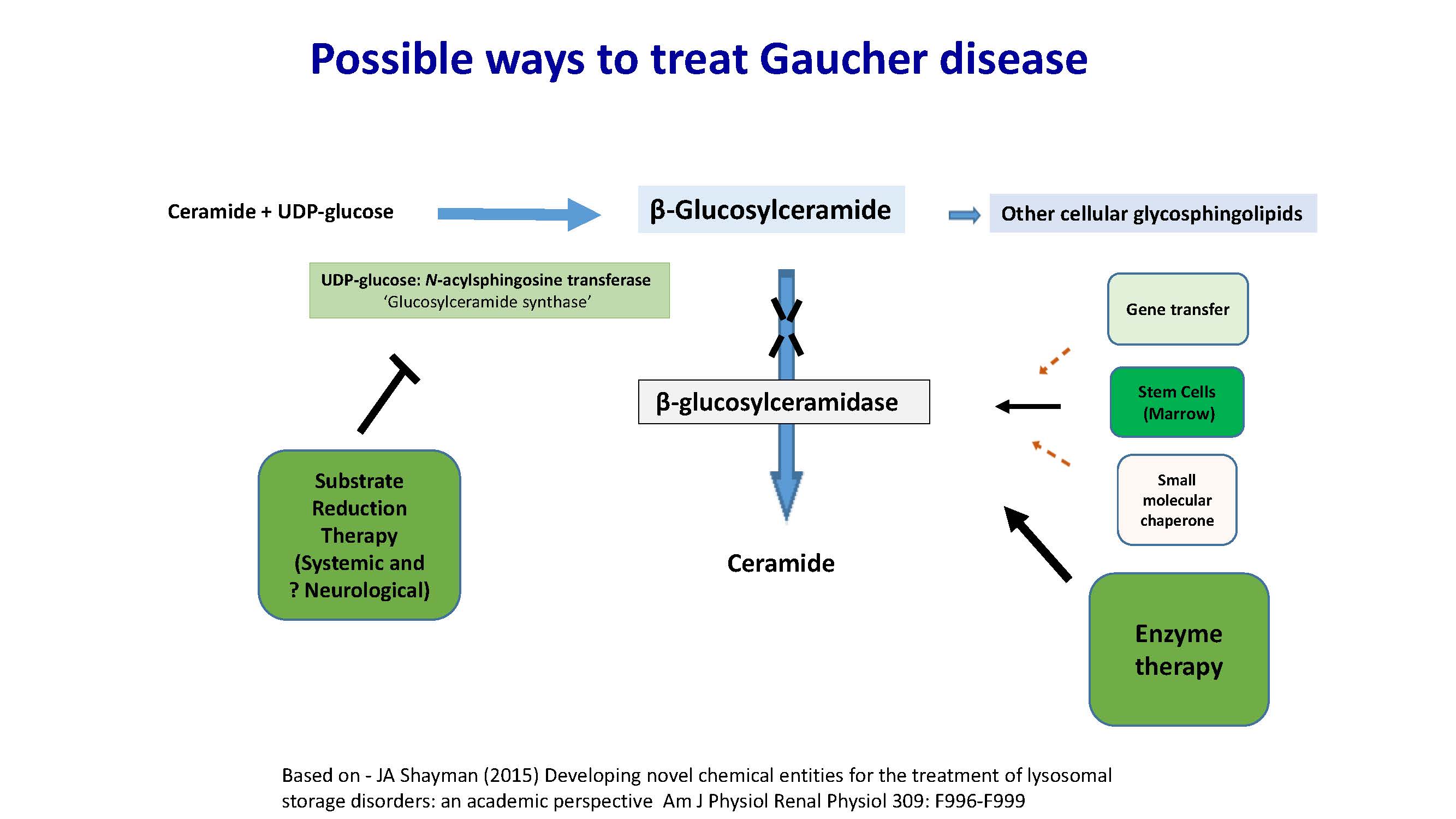
Possible ways to treat Gaucher disease Ceramide + UDP-glucose β-Glucosylceramide Other cellular glycosphingolipids UDP-glucose: N-acylsphingosine transferase ‘Glucosylceramide synthase’ Substrate Reduction Therapy (Systemic and ? Neurological) Small molecular chaperone Gene transfer Stem Cells (Marrow) β-glucosylceramidase Ceramide Enzyme therapy Based on - JA Shayman (2015) Developing novel chemical entities for the treatment of lysosomal storage disorders: an academic perspective Am J Physiol Renal Physiol 309: F996-F999

Today’s agenda 31 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

CO NF I DE NT I AL Arianna living with Gaucher disease type 3 Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Key takeaways GD1 patient data to date has improved from baseline ERT with some clinically significant reductions in liver (n=3) and spleen volume (n=2) GD3 named patient data to date show evidence of biochemical correction, with lymphadenopathy and enteropathy improvements and neurological stabilization Continued favorable safety profile to date

Guard1 - Phase 1/2 AVR-RD-02 Gaucher type 1 Phase 1/2 has 6 patients enrolled to date 33 Objectives Patients Safety Efficacy Engraftment Enrollment goal 8-16 patients Gaucher disease type 1 18-45-year-old males and females patients who are: ERT-stable for >24 months or Have a confirmed diagnosis of GD1 based on: Treatment-naïve or GBA biallelic mutations on genetic sequencing Deficient glucocerebrosidase enzyme activity • Have not received ERT or SRT in the last 12 months GD1=Gaucher disease type 1; ERT=Enzyme replacement therapy; SRT=Substrate reduction therapy Actively recruiting Guard1
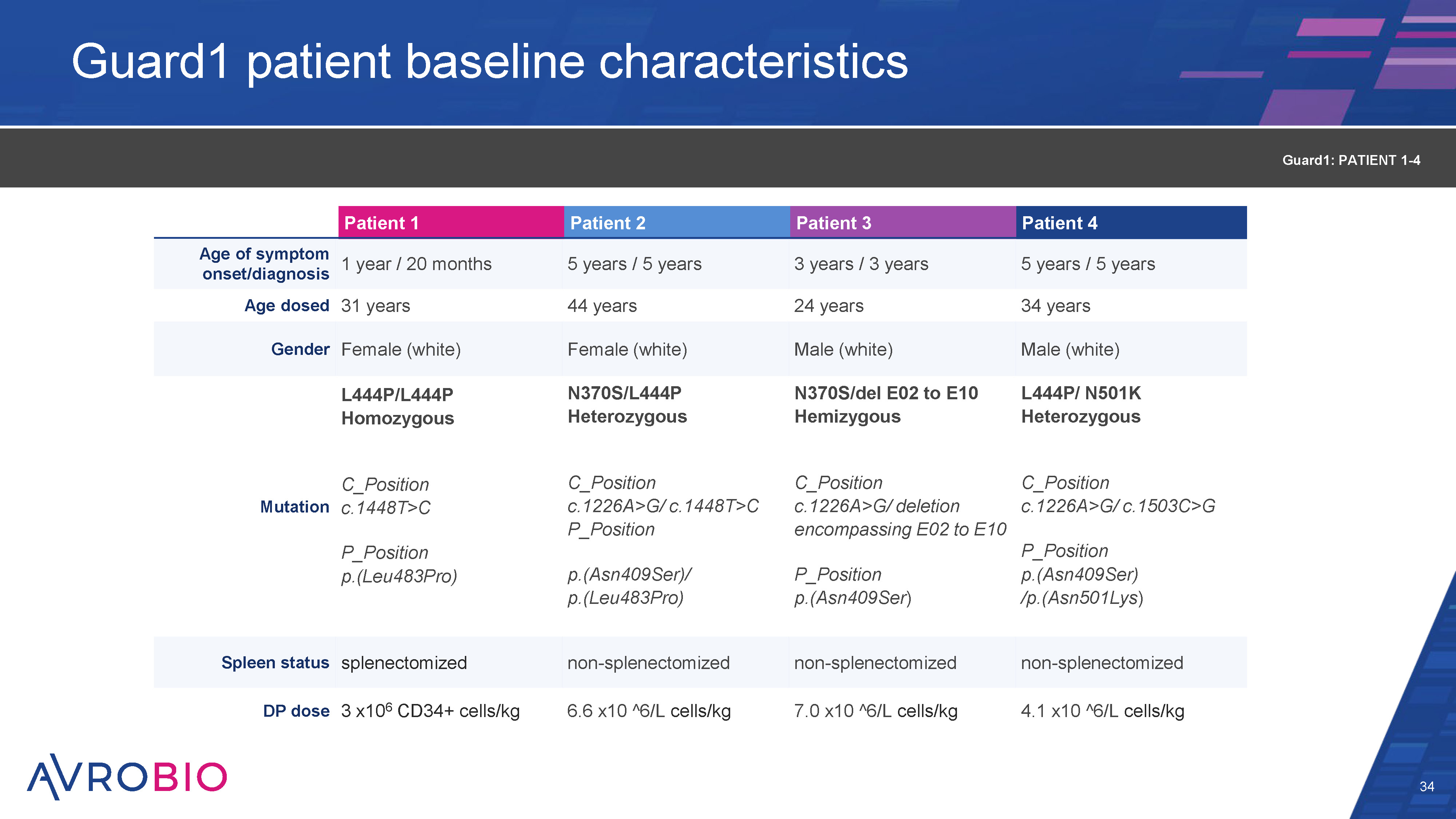
Guard1 patient baseline characteristics 34 Patient 1 Patient 2 Patient 3 Patient 4 Age of symptom onset/diagnosis 1 year / 20 months 5 years / 5 years 3 years / 3 years 5 years / 5 years Age dosed 31 years 44 years 24 years 34 years Gender Female (white) Female (white) Male (white) Male (white) L444P/L444P Homozygous N370S/L444P Heterozygous N370S/del E02 to E10 Hemizygous L444P/ N501K Heterozygous Mutation C_Position c.1448T>C C_Position c.1226A>G/ c.1448T>C P_Position C_Position c.1226A>G/ deletion encompassing E02 to E10 C_Position c.1226A>G/ c.1503C>G P_Position p.(Leu483Pro) p.(Asn409Ser)/ p.(Leu483Pro) P_Position p.(Asn409Ser) P_Position p.(Asn409Ser) /p.(Asn501Lys) Spleen status splenectomized non-splenectomized non-splenectomized non-splenectomized DP dose 3 x106 CD34+ cells/kg 6.6 x10 ^6/L cells/kg 7.0 x10 ^6/L cells/kg 4.1 x10 ^6/L cells/kg Guard1: PATIENT 1-4
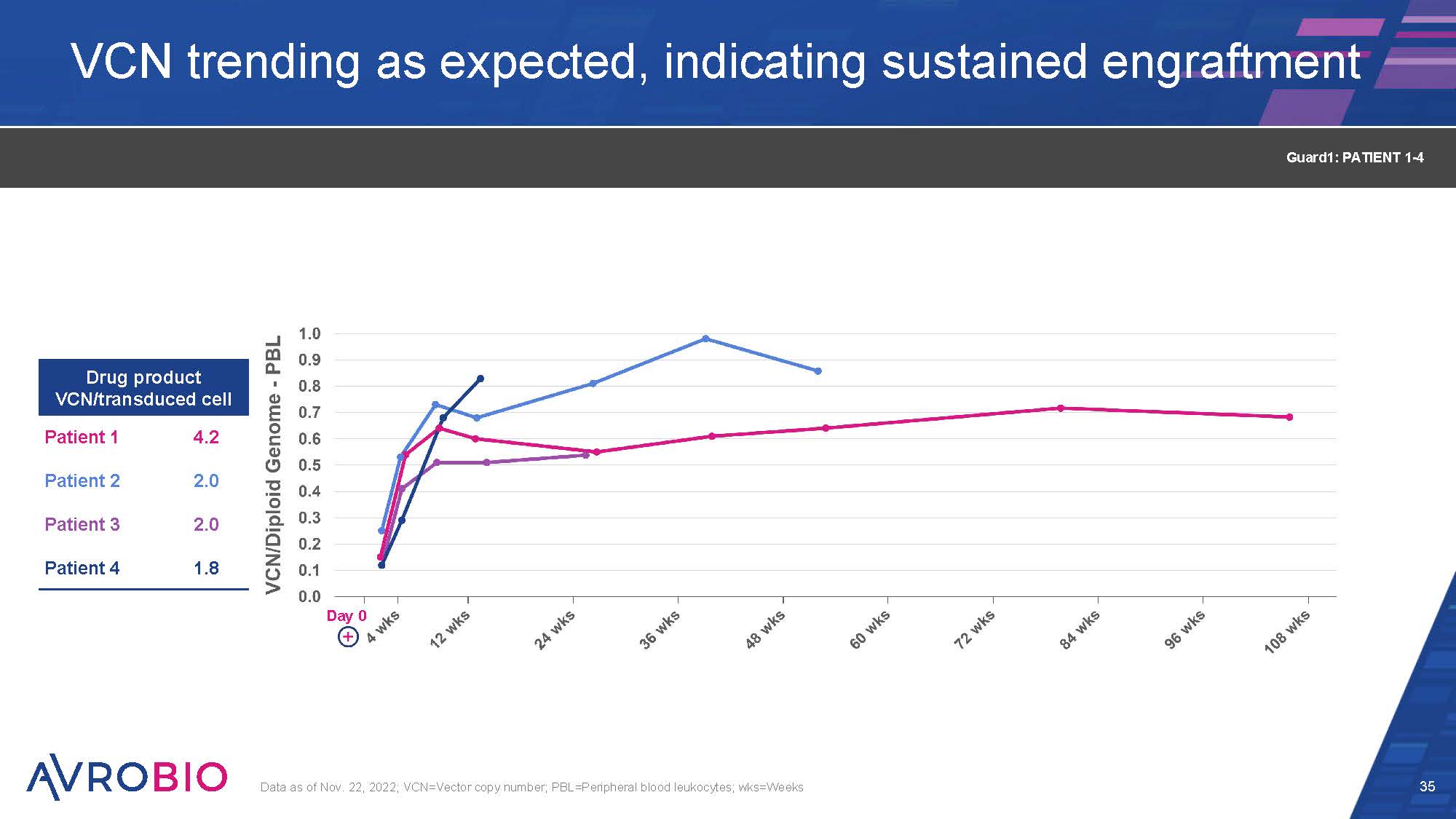
VCN trending as expected, indicating sustained engraftment 35 Data as of Nov. 22, 2022; VCN=Vector copy number; PBL=Peripheral blood leukocytes; wks=Weeks Drug product VCN/transduced cell Patient 1 4.2 Patient 2 2.0 Patient 3 2.0 Patient 4 1.8 Guard1: PATIENT 1-4 Day 0
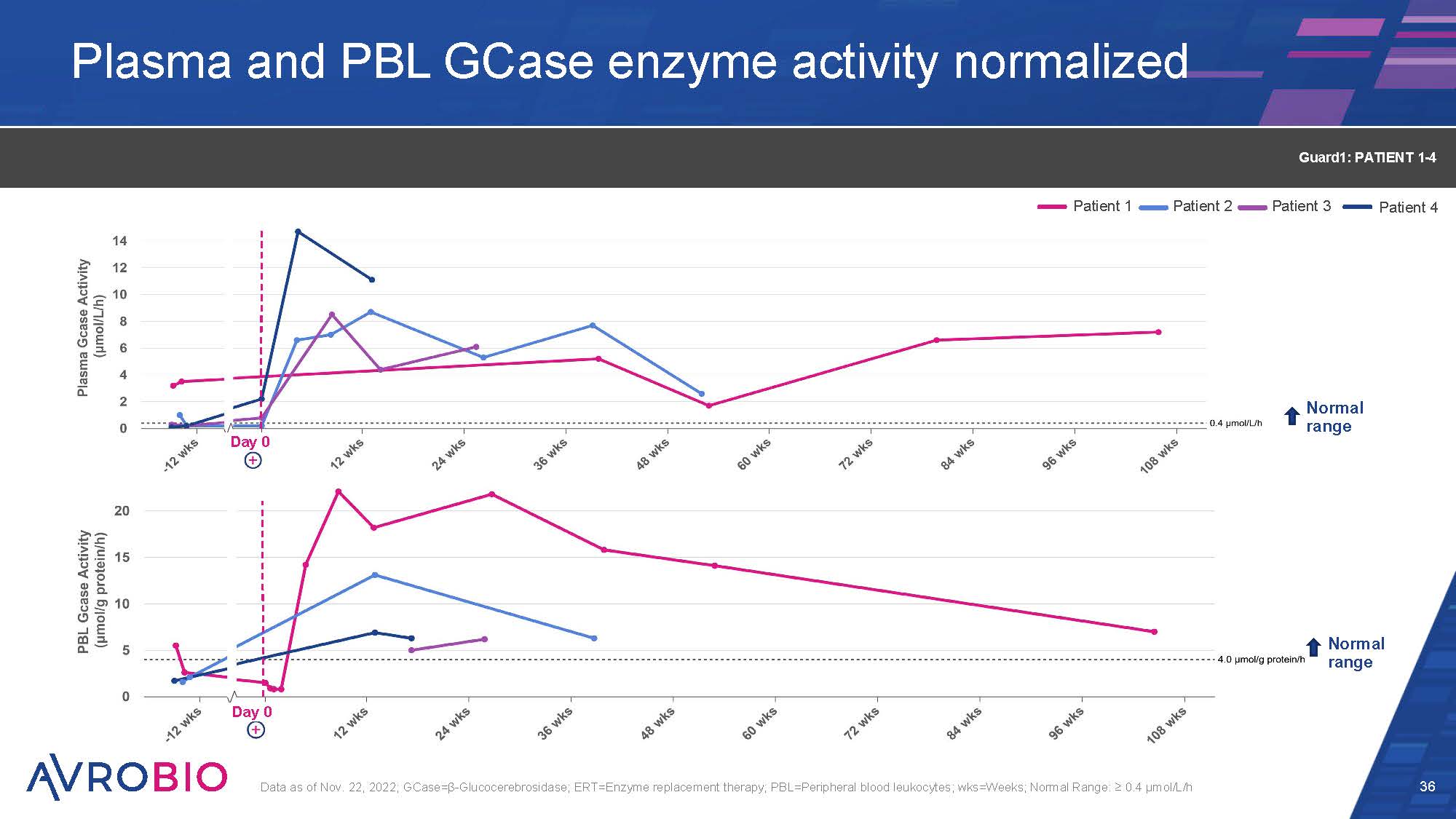
Plasma and PBL GCase enzyme activity normalized 36 Data as of Nov. 22, 2022; GCase=β-Glucocerebrosidase; ERT=Enzyme replacement therapy; PBL=Peripheral blood leukocytes; wks=Weeks; Normal Range: ≥ 0.4 µmol/L/h Guard1: PATIENT 1-4 Normal range Day 0 Day 0 Normal range Patient 1 Patient 2 Patient 3 Patient 4
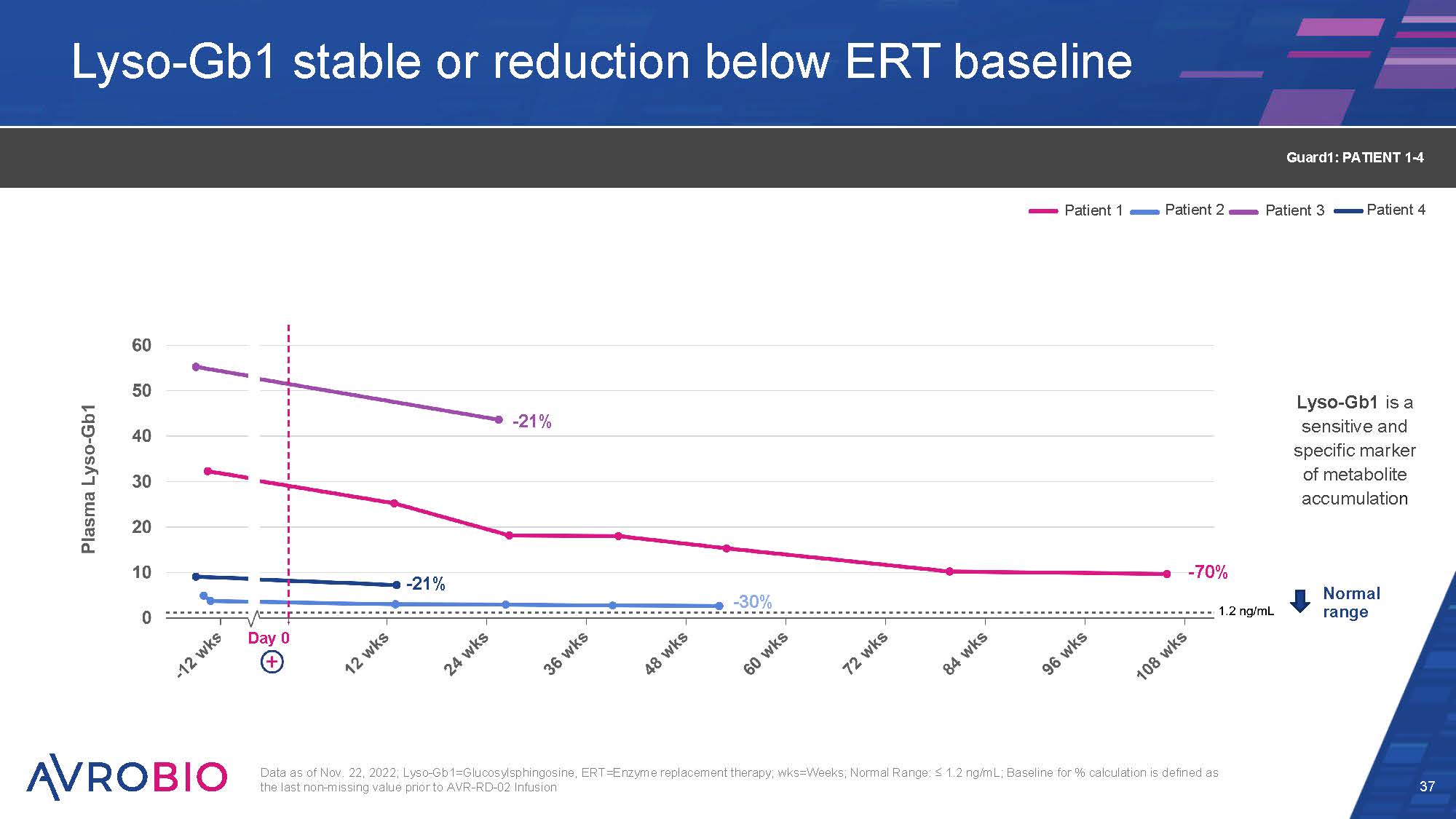
Lyso-Gb1 stable or reduction below ERT baseline 37 Data as of Nov. 22, 2022; Lyso-Gb1=Glucosylsphingosine, ERT=Enzyme replacement therapy; wks=Weeks; Normal Range: ≤ 1.2 ng/mL; Baseline for % calculation is defined as the last non-missing value prior to AVR-RD-02 Infusion Normal range Lyso-Gb1 is a sensitive and specific marker of metabolite accumulation Patient 1 Patient 2 Patient 3 Patient 4 Guard1: PATIENT 1-4 Day 0 -70% -30% -21% -21%
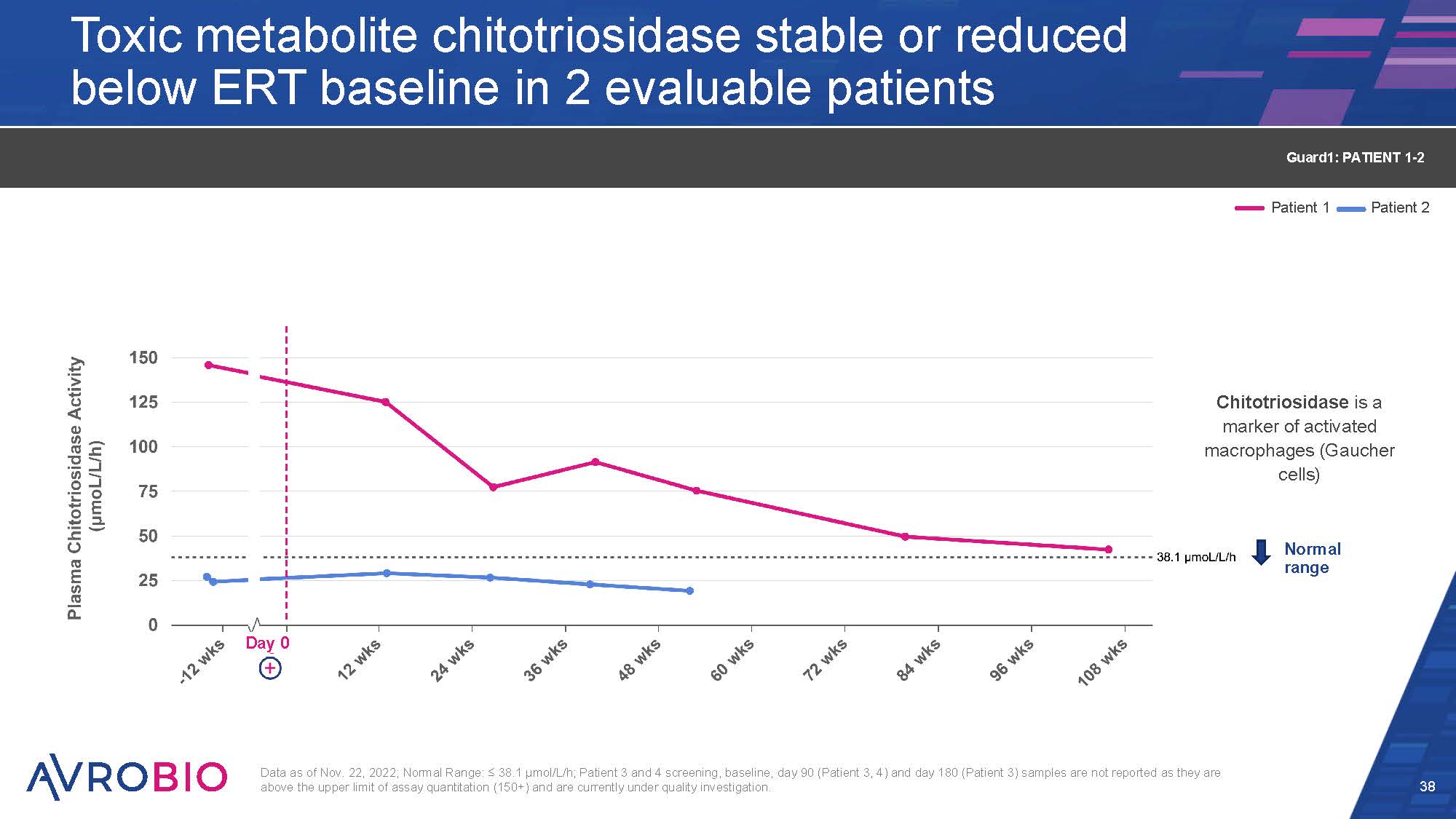
Toxic metabolite chitotriosidase stable or reduced below ERT baseline in 2 evaluable patients 38 Data as of Nov. 22, 2022; Normal Range: ≤ 38.1 µmol/L/h; Patient 3 and 4 screening, baseline, day 90 (Patient 3, 4) and day 180 (Patient 3) samples are not reported as they are above the upper limit of assay quantitation (150+) and are currently under quality investigation. Normal range Patient 1 Patient 2 Guard1: PATIENT 1-2 Day 0 Chitotriosidase is a marker of activated macrophages (Gaucher cells)
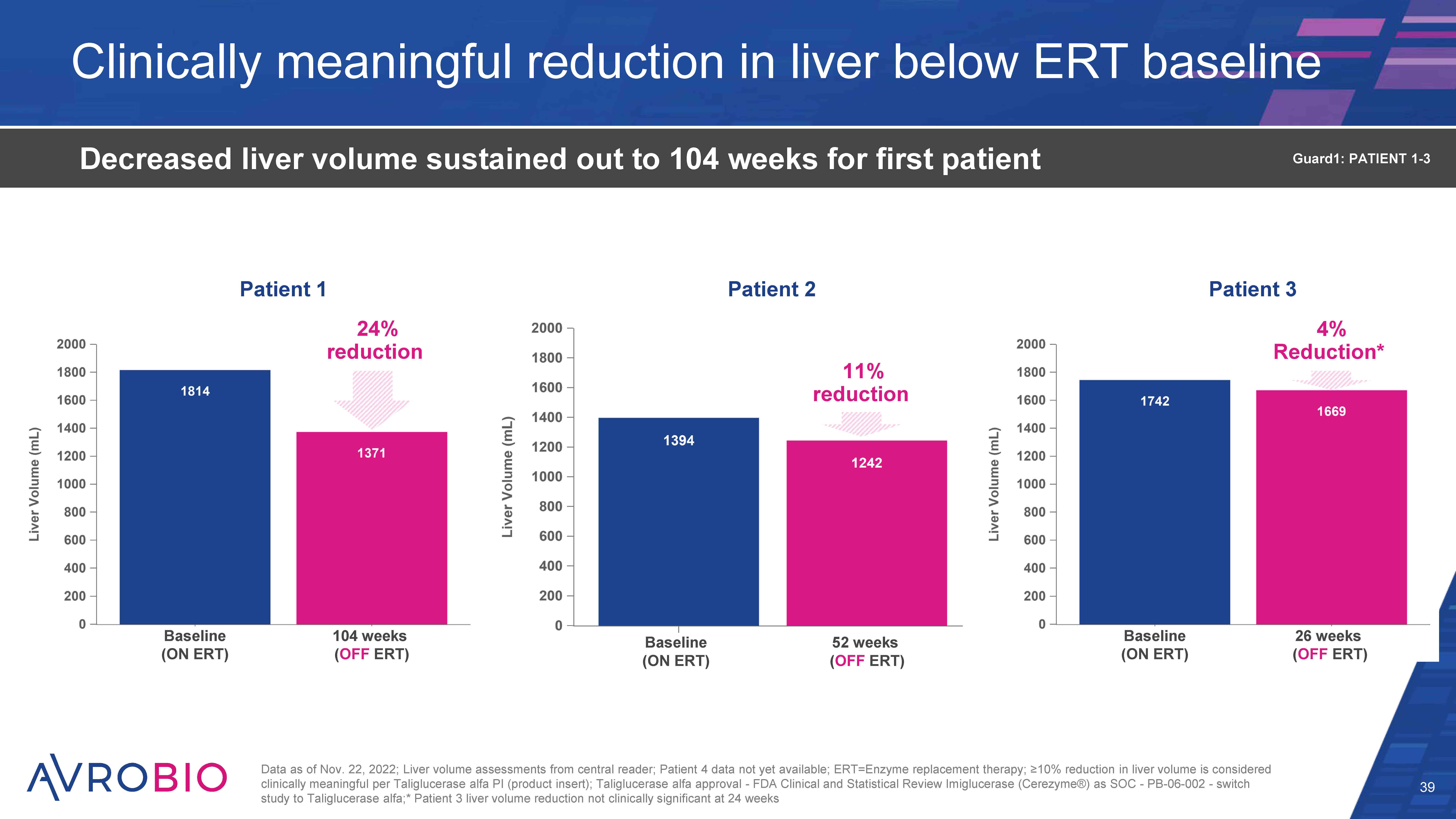
Clinically meaningful reduction in liver below ERT baseline 39 Decreased liver volume sustained out to 104 weeks for first patient Data as of Nov. 22, 2022; Liver volume assessments from central reader; Patient 4 data not yet available; ERT=Enzyme replacement therapy; ≥10% reduction in liver volume is considered clinically meaningful per Taliglucerase alfa PI (product insert); Taliglucerase alfa approval - FDA Clinical and Statistical Review Imiglucerase (Cerezyme®) as SOC - PB-06-002 - switch study to Taliglucerase alfa;* Patient 3 liver volume reduction not clinically significant at 24 weeks Guard1: PATIENT 1-3 Patient 1 Patient 2 Patient 3 24% reduction 4% Reduction* Baseline (ON ERT) Baseline (ON ERT) 104 weeks (OFF ERT) 24 weeks (OFF ERT) Baseline (ON ERT) 52 weeks (OFF ERT) 11% reduction
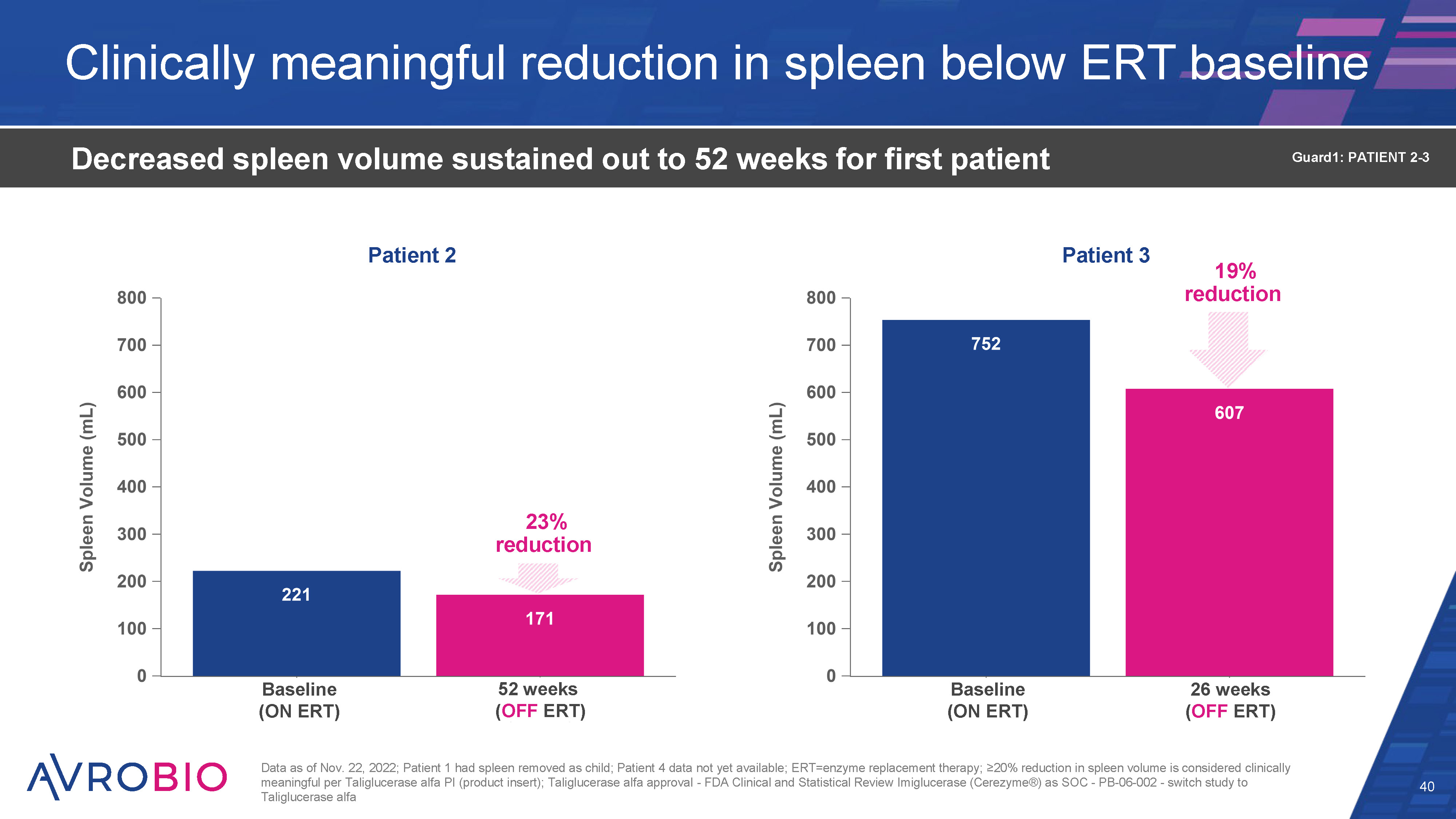
Clinically meaningful reduction in spleen below ERT baseline 40 Decreased spleen volume sustained out to 52 weeks for first patient Data as of Nov. 22, 2022; Patient 1 had spleen removed as child; Patient 4 data not yet available; ERT=enzyme replacement therapy; ≥10% reduction in spleen volume is considered clinically meaningful per Taliglucerase alfa PI (product insert); Taliglucerase alfa approval - FDA Clinical and Statistical Review Imiglucerase (Cerezyme®) as SOC - PB-06-002 - switch study to Taliglucerase alfa Guard1: PATIENT 2-3 23% reduction Patient 2 Patient 3 19% reduction Baseline (ON ERT) Baseline (ON ERT) 52 weeks (OFF ERT) 26 weeks (OFF ERT)
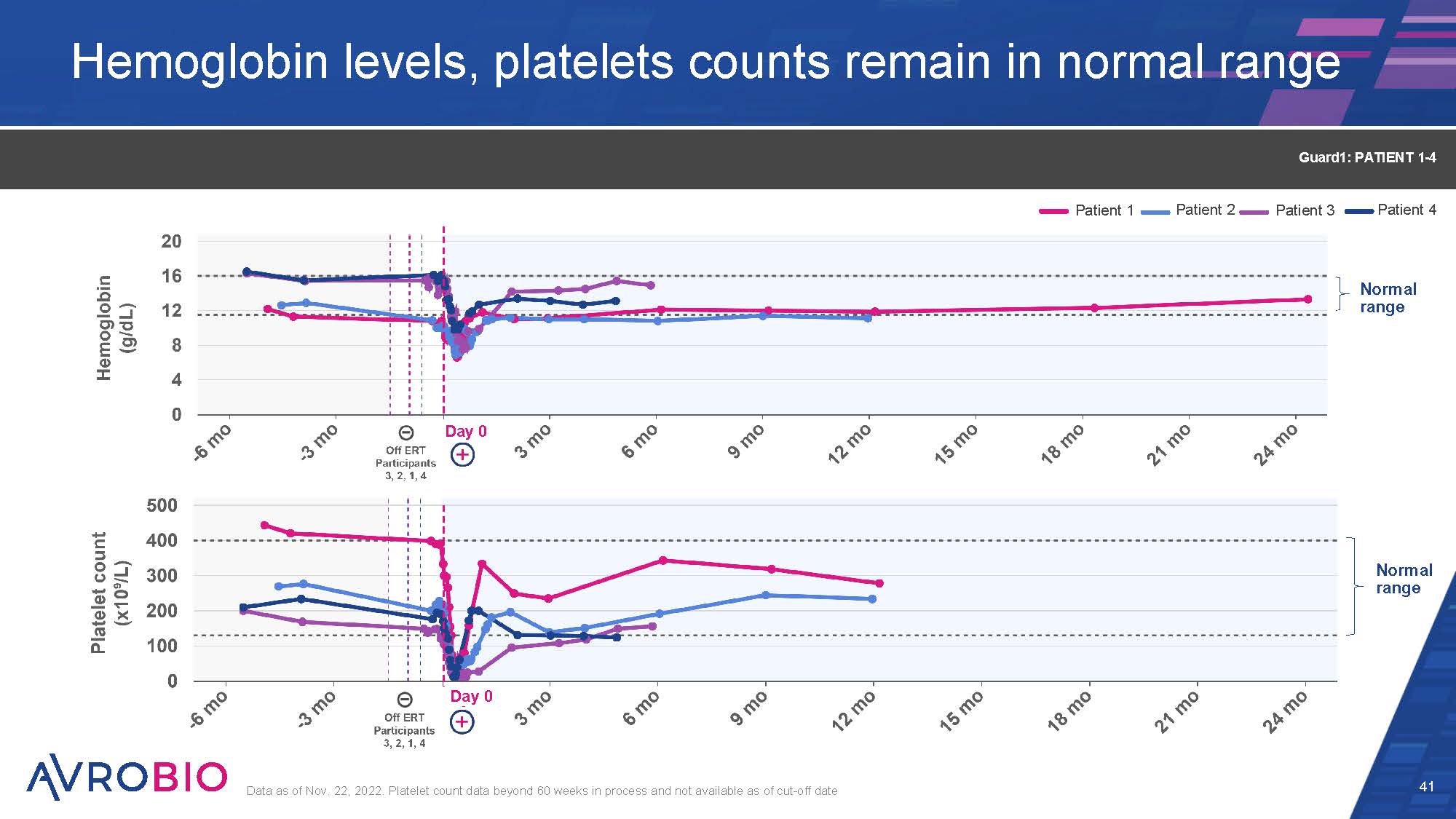
Hemoglobin levels, platelets counts remain in normal range 41 Data as of Nov. 22, 2022. Platelet count data beyond 60 weeks in process and not available as of cut-off date Guard1: PATIENT 1-4 Normal range Normal range Patient 1 Patient 2 Patient 3 Patient 4 Day 0 Day 0

No adverse events related to AVR-RD-02 drug product 42 0 SAEs or AEs related to AVR-RD-02 drug product Guard1: PATIENTS 1-4 All AEs and SAEs related* to: Myeloablative conditioning Drugs mandated by protocol or study procedures Underlying disease Pre-existing conditions * AEs/SAEs as determined by investigator. Of the non-AVR-RD-02 drug product AEs/SAEs observed, 71 are AEs and 2 are SAEs, including anemia, leukopenia, neutropenia, thrombocytopenia, eye pain, decreased appetite, dehydration, headache, hypophosphatemia, amenorrhea (unresolved and ongoing as of safety database cut date). AVR-RD-02 has not been approved by FDA or by any other regulatory body and its safety and efficacy has not been established; SAE=serious adverse event; AE=adverse event DATA AS OF SEPT. 27, 2022

Today’s agenda 43 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

Named Patient AVR-RD-02 Patient Primary disease complications 12-year-old male with GD3 Diagnosed at 10 months –lymphadenopathy; hepatosplenomegaly Commenced ERT at 17 months Seizures developed age 10 years Biomarkers and clinical signs of Gaucher disease have never normalized despite maximal multimodal therapies Primary disease complications: Mesenteric lymphadenopathy Protein-losing enteropathy Commenced compassionate use SRT at 4.5 years Neurology Saccadic eye movement defect Intellectual impairment (FSIQ 66 – Low) Seizures (2 x antiepileptic medications) Modified Severity Scoring Tool*: 1.5-12.5 (2016-2021) 44 First pediatric patient with GD3 dosed Manchester University NHS Foundation Trust, UK ERT=Enzyme replacement therapy; SRT=Substrate reduction therapy; AEs=Adverse events, FSIQ=Full-scale intelligence quotient; Bu90=Cumulative busulfan AUC of 90mg.hr/L; GT=Gene therapy; GD3=Gaucher disease type 3

45 "Having a child with Gaucher type 3 disease can, at times, feel hopeless and helpless. Our son was on ERT and developed seizures and protein- losing enteropathy (PLE) which required additional steroids and medications. I was always worried about the long-term use of the steroids specifically as he is still growing. The process to receive his medications was overwhelming and time consuming. He was declining cognitively, and he developed seizures that kept getting worse despite anti-epileptic medications. Our son's cognitive decline and seizures were very scary and devastating to all of us, and I was looking for new treatment options online when I found gene therapy. We finally had a glimpse of hope.” Parent of Named Patient

HSC gene therapy well tolerated to date Age at treatment: 11 years Underwent mobilization and apheresis of HSCs which were then transduced ex-vivo with LV-containing GBA gene to produce AVR-RD-02 drug product Received conditioning with busulfan (Bu90-TCI) which was uncomplicated Engraftment achieved at Day 9 (neutrophils > 1× 109/L and platelets > 50 × 109/L) Required no blood products No AEs related to drug product Minimal AEs of low grade/severity Single episode of febrile neutropenia which was culture negative and resolved within 48 hours without sequalae 46 Data as of June 2022; HSC=Hematopoietic stem cell; AEs=Adverse events, Bu90=Cumulative busulfan AUC of 90mg.hr/L; GT=Gene therapy; GD3=Gaucher disease type 3 Named Patient AVR-RD-02 GD3: Named Patient
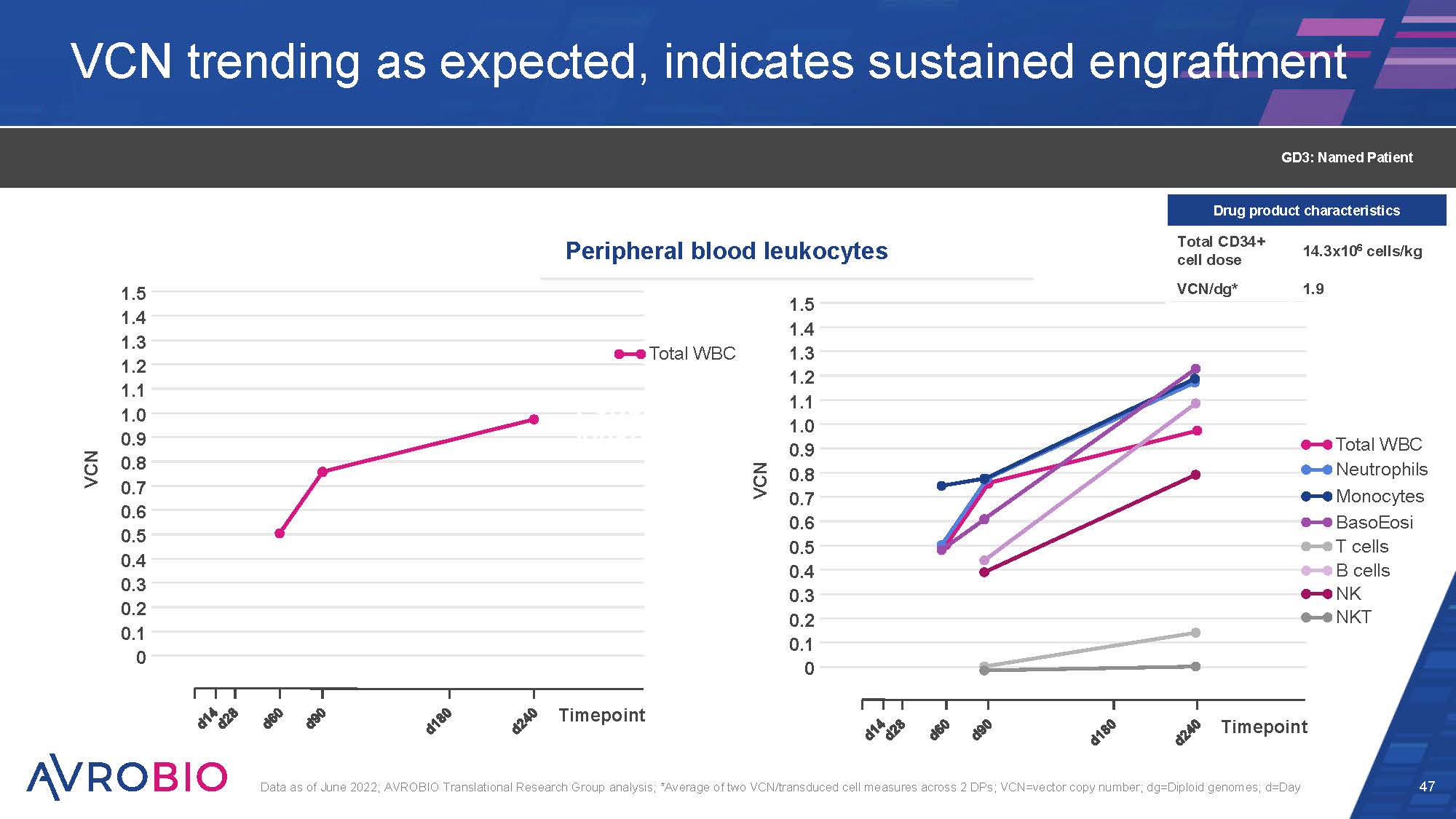
VCN trending as expected, indicates sustained engraftment 47 Data as of June 2022; AVROBIO Translational Research Group analysis; *Average of two VCN/transduced cell measures across 2 DPs; VCN=vector copy number; dg=Diploid genomes; d=Day 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 VCN Timepoint Total WBC Perip leuko 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 VCN Timepoint Total WBC Neutrophils Monocytes BasoEosi T cells B cells NK NKT Peripheral blood leukocytes GD3: Named Patient Drug product characteristics Total CD34+ cell dose 14.3x106 cells/kg VCN/dg* 1.9
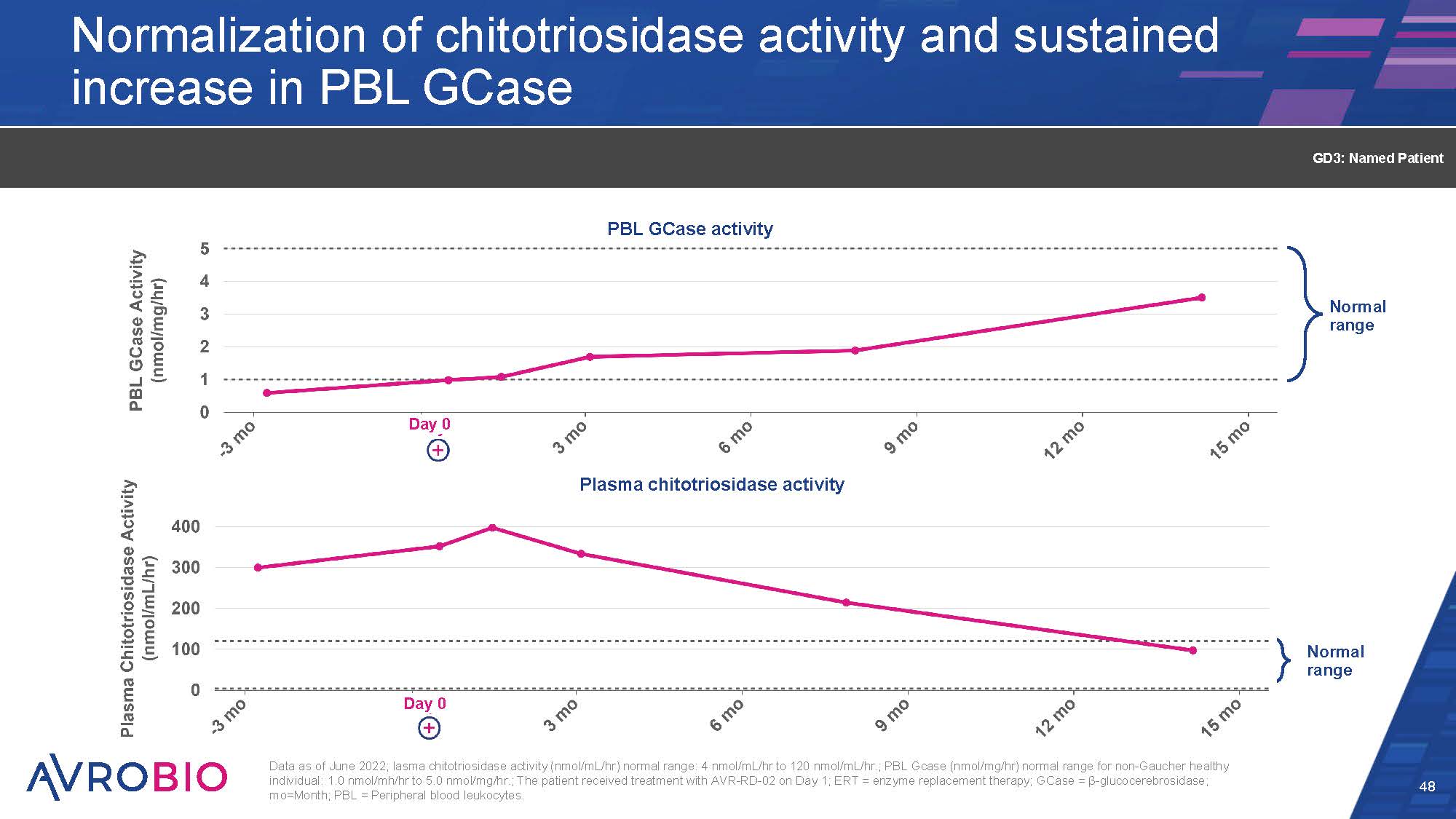
Normal range Normalization of chitotriosidase activity and sustained increase in PBL GCase 48 GD3: Named Patient Normal range Plasma chitotriosidase activity P BL GCase activity Day 0 Day 0 Data as of June 2022; lasma chitotriosidase activity (nmol/mL/hr) normal range: 4 nmol/mL/hr to 120 nmol/mL/hr.; PBL Gcase (nmol/mg/hr) normal range for non-Gaucher healthy individual: 1.0 nmol/mh/hr to 5.0 nmol/mg/hr.; The patient received treatment with AVR-RD-02 on Day 1; ERT = enzyme replacement therapy; GCase = β-glucocerebrosidase; mo=Month; PBL = Peripheral blood leukocytes.
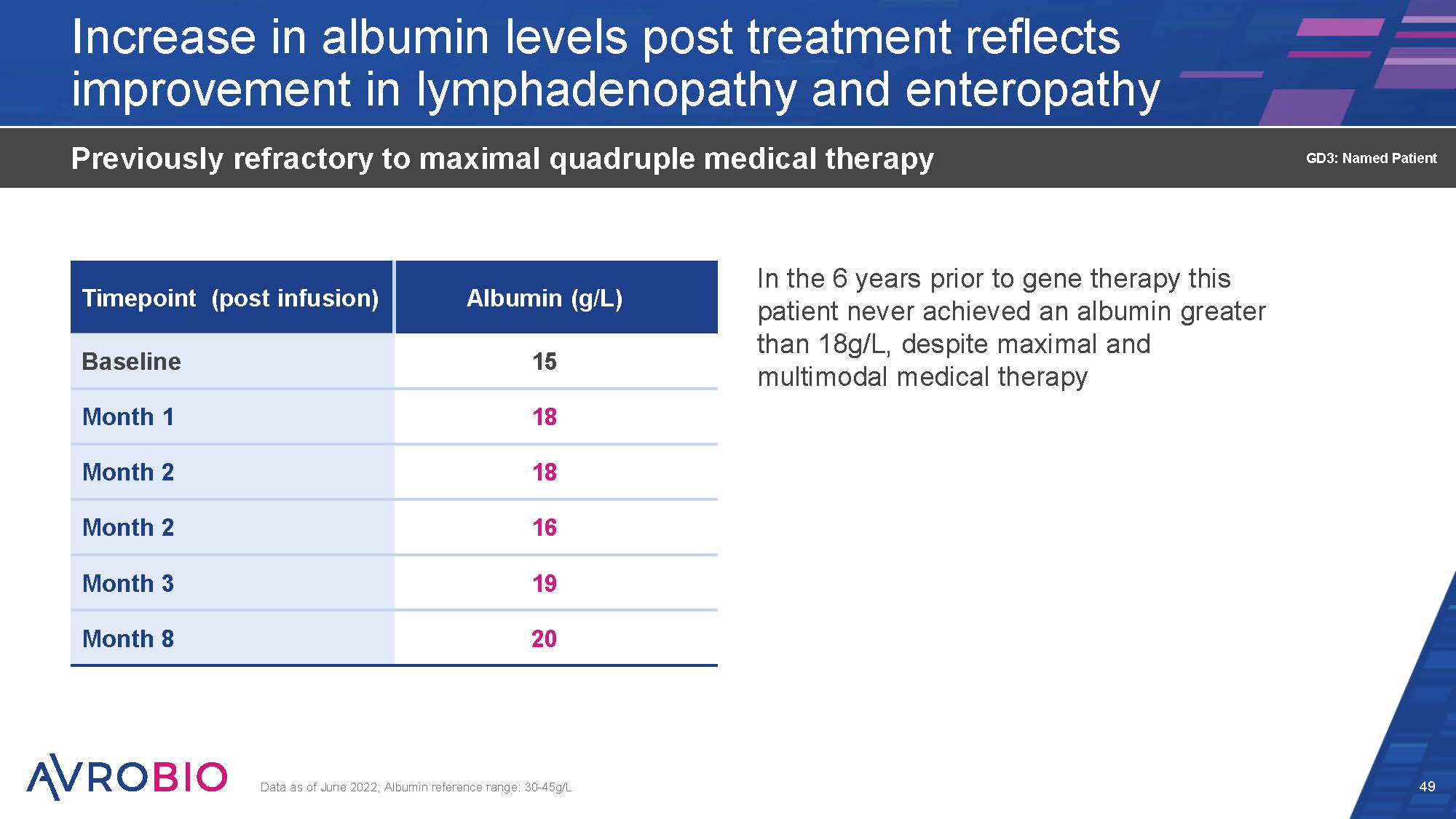
Increase in albumin levels post treatment reflects improvement in lymphadenopathy and enteropathy In the 6 years prior to gene therapy this patient never achieved an albumin greater than 18g/L, despite maximal and multimodal medical therapy Previously refractory to maximal quadruple medical therapy Data as of June 2022; Albumin reference range: 30-45g/L 49 GD3: Named Patient Timepoint (post infusion) Albumin (g/L) Baseline 15 Month 1 18 Month 2 18 Month 2 16 Month 3 19 Month 8 20

Biomarkers normalized 15 months post gene therapy Normalized peripheral GCase enzyme activity and plasma chitotriosidase (ERT and SRT free) Lymphadenopathy – reduction on MRI, with highest albumin levels achieved in parallel with stopping enteropathy-oriented therapy MRI brain – no new lesions post-gene therapy when previously they were developing rapidly No clinically detectable change in neurological status (mSST) No new neurological manifestations post gene therapy No adverse events related to AVR-RD-02 drug product Reported AEs and SAEs consistent with myeloablative conditioning, drugs mandated by protocol or study procedures, underlying disease or pre- existing conditions 50 GD3: Named Patient GD3=Gaucher disease type 3; GCase=β-Glucocerebrosidase, ERT=Enzyme replacement therapy; SRT=Substrate reduction therapy; MRI=Magnetic resonance imaging; mSST=Modified severity scoring tool Patient remains OFF ERT, SRT, enteral steroids, dietary restrictions and intermittent albumin infusions DATA AS OF JUNE, 2022

51 “Following gene therapy, we have seen real changes in our life and our son's life. The first few weeks were a bit rough in terms of mucosal inflammation, hair loss and skin changes, but overall, he appeared to respond to the treatment very well. He is off ERT, steroids and SRT completely, with no return of PLE symptoms, such as edema and GI distress. He still has seizures but no further change in cognitive abilities. My son now is sleeping throughout the night, while he used to wake up often. Our family gained freedom as we are no longer tied to a challenging medication schedule and many hospital visits.” Parent of Named Patient AVR-RD-02 remains investigational and is not approved by FDA or any other regulatory authority for the treatment of Gaucher disease

Today’s agenda 52 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

CO NF I DE NT I AL Arianna living with Gaucher disease type 3 Development and design of clinical trials for Gaucher disease Key takeaways Pursue one global Phase 2/3 trial for GD3 following positive feedback from FDA and MHRA Clinical development approach intends to use combined data set for GD1 and GD3 based on common underlying pathology of disease

First RCT in HSC gene therapy Open-label, parallel-arm, randomized controlled, pediatric Phase 2/3 study evaluating efficacy and safety of AVR-RD-02 Regulatory alignment based on guidance received during MHRA advice meeting and FDA type C meeting; CTA and IND amendments to follow in 2023; Multi-component endpoint: Weighting on liver and spleen endpoints and 52 weeks trial length subject to regulatory alignment; GT=Gene therapy; LTFU=Long term follow-up; pts=Patients; CSF=Cerebrospinal fluid 54 Planned GD3 Phase 2/3 registrational clinical trial design Primary efficacy endpoint – Multi-domain endpoint Primary endpoint includes: Scale for the assessment and rating of ataxia (SARA) Diffusing capacity of the lung for carbon monoxide (DLCO) Liver volume Spleen volume Key secondary endpoint: Lyso-Gb1 level in CSF Change from Baseline to Week 52 (length TBC) in multi-domain endpoint Primary inference based on treatment comparison at Week 52 GD3 study design n=~20 n=~20
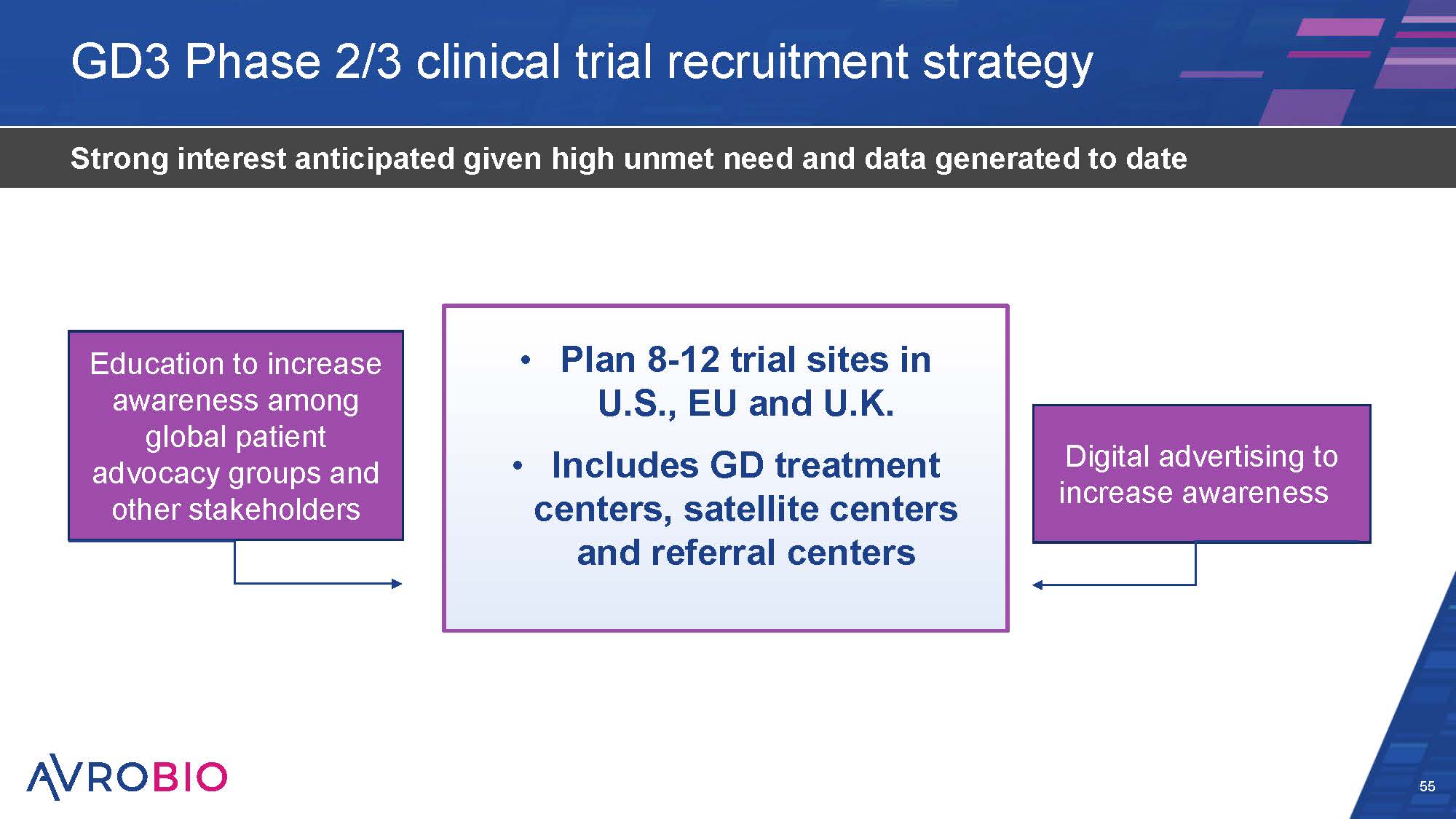
GD3 Phase 2/3 clinical trial recruitment strategy 55 Strong interest anticipated given high unmet need and data generated to date Plan 8-12 trial sites in U.S., EU and U.K. Includes GD treatment centers, satellite centers and referral centers Education to increase awareness among global patient advocacy groups and other stakeholders Digital advertising to increase awareness
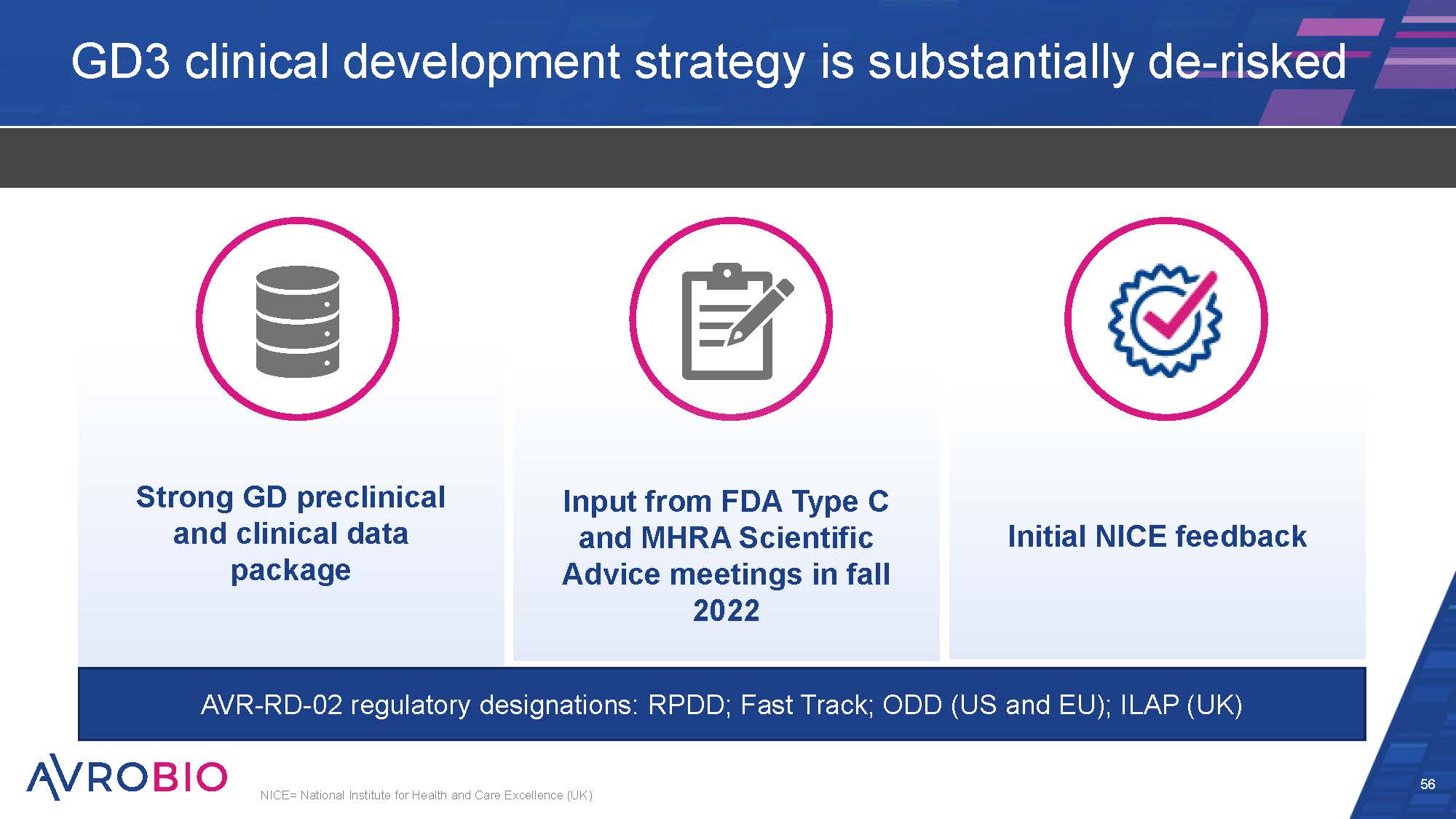
Input from FDA Type C and MHRA Scientific Advice meetings in fall 2022 GD3 clinical development strategy is substantially de-risked 56 NICE= National Institute for Health and Care Excellence (UK) Strong GD preclinical and clinical data package Initial NICE feedback AVR-RD-02 regulatory designations: RPDD; Fast Track; ODD (US and EU); ILAP (UK)
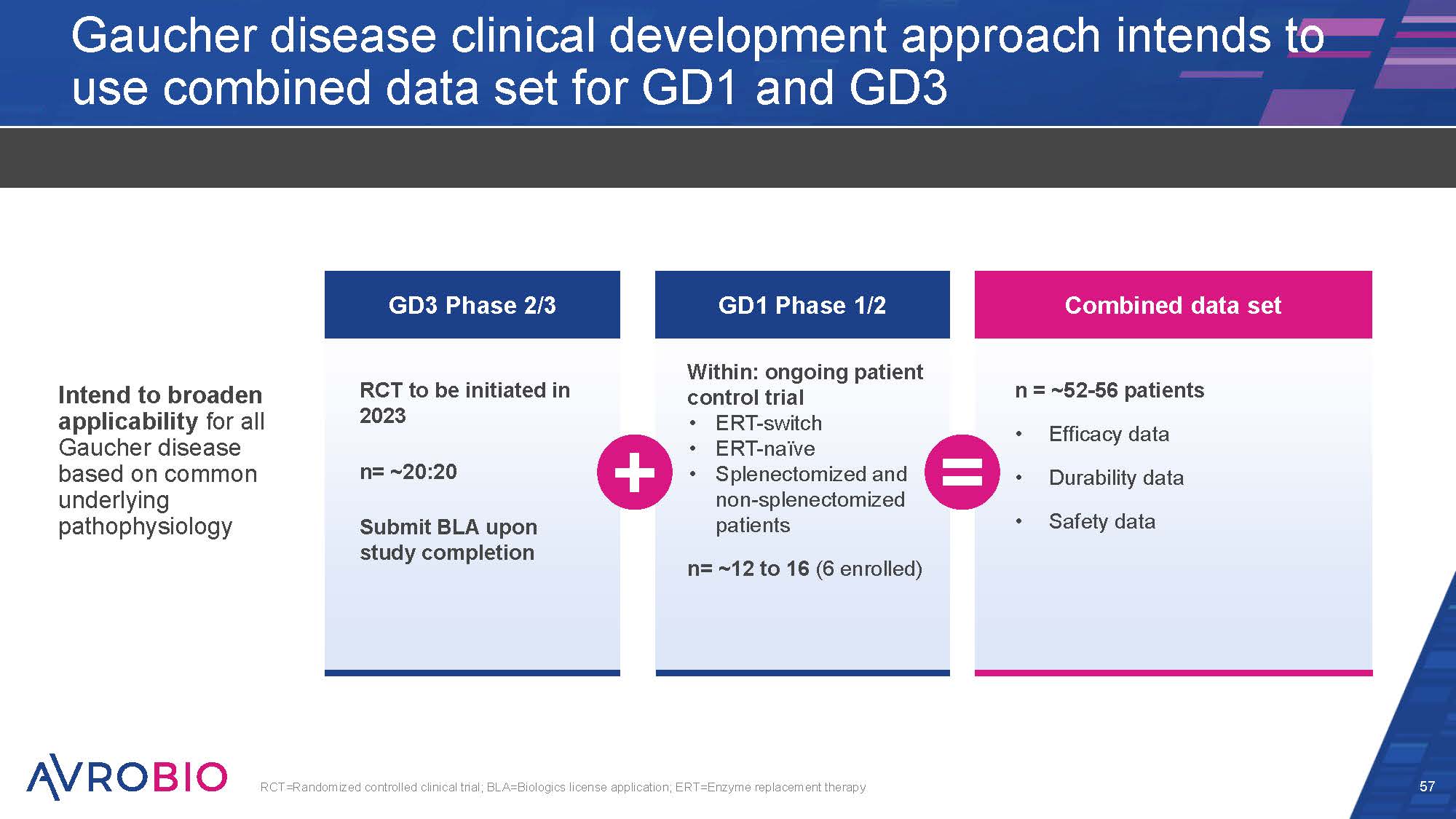
Gaucher disease clinical development approach intends to use combined data set for GD1 and GD3 Intend to broaden applicability for all Gaucher disease based on common underlying pathophysiology 57 RCT=Randomized controlled clinical trial; BLA=Biologics license application; ERT=Enzyme replacement therapy GD3 Phase 2/3 RCT to be initiated in 2023 n= ~20:20 Submit BLA upon study completion GD1 Phase 1/2 Within: ongoing patient control trial ERT-switch ERT-naïve Splenectomized and non-splenectomized patients n= ~12 to 16 (6 enrolled) Combined data set n = ~52-56 patients Efficacy data Durability data Safety data

Gaucher disease - AVR-RD-02 Anticipated next steps 58 Initiate global GD3 registrational trial in 2H 2023 Complete GD1 Phase 1/2 patient enrollment in YE 2023 GD3=Gaucher disease type 3; GD1=Gaucher disease type 1

Today’s agenda 59 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

luca plato® –– AVROBIO’s platform for global gene therapy commercialization and pipeline expansion Redefines safety best practices Reinvents manufacturing best practices Photo depicts multiple Miltenyi Biotec Prodigy® units in a cleanroom; Photo courtesy of Miltenyi Biotec

CO NF I DE NT I AL Deploying the plato® advantage Arianna living with Gaucher disease type 3 Key takeaways Late-stage ready with no major CMC changes anticipated Scalable to support commercialization globally Designed to reduce COGs

Path through BLA is well understood Clarity on regulators’ expectations regarding Potency assay Product release and characterization (LV and DP) Comparability Traceability Stability AVROBIO has obtained feedback from multiple regulatory agencies and is working to incorporate it US Canada Japan Israel Brazil UK 62 BLA=Biologics license application; LV=Lentiviral; DP=Drug product; FDA=Food and Drug Administration; CMC=Chemistry, manufacturing and controls Oct 20, 2022 Positive FDA Type C meeting on proposed GD3 Phase 2/3 Trial No major CMC changes anticipated for Phase 2/3 trial
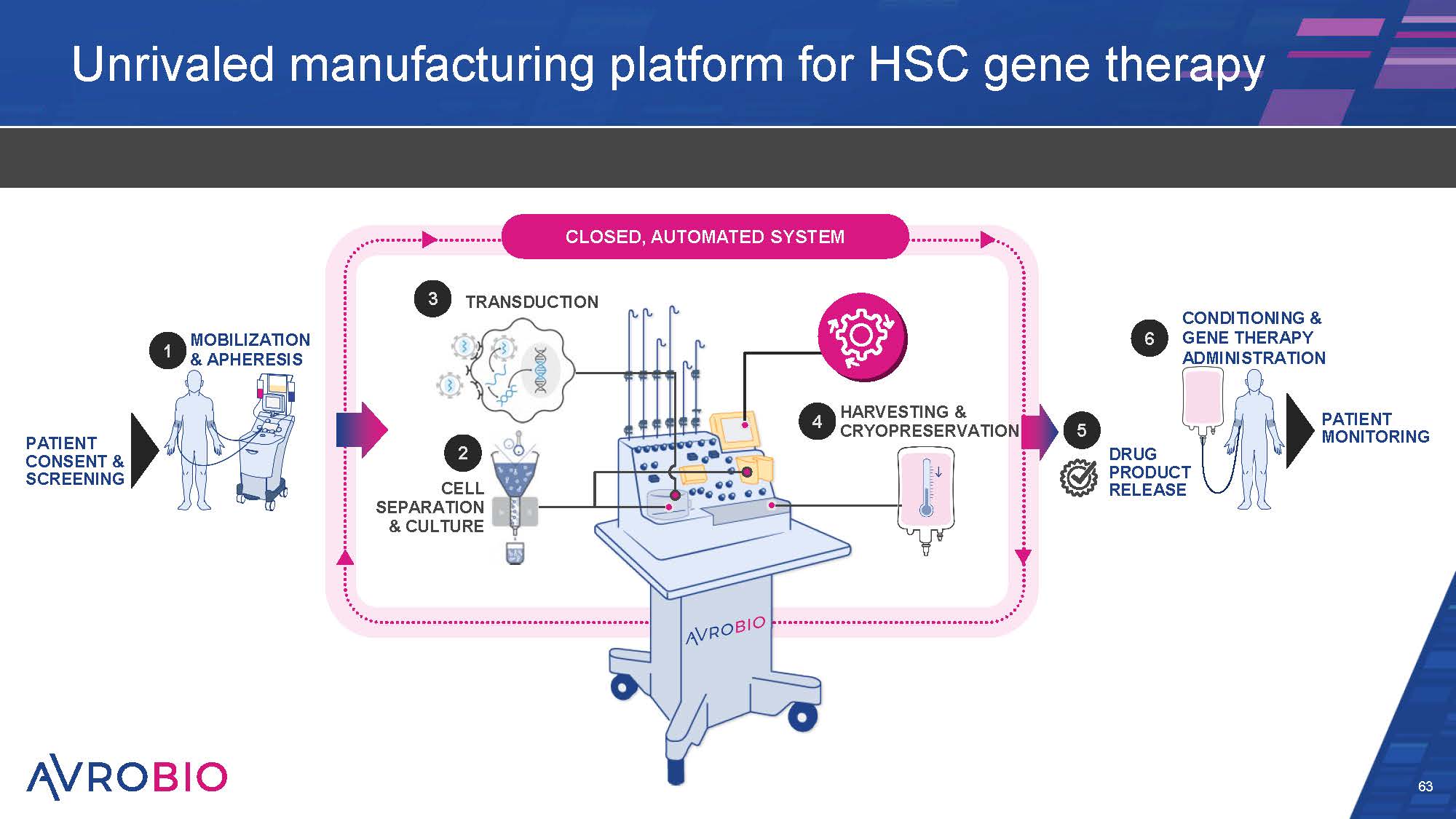
Unrivaled manufacturing platform for HSC gene therapy 63 MOBILIZATION & APHERESIS PATIENT CONSENT & SCREENING PATIENT MONITORING CONDITIONING & GENE THERAPY ADMINISTRATION TRANSDUCTION CLOSED, AUTOMATED SYSTEM HARVESTING & CRYOPRESERVATION 2 CELL SEPARATION & CULTURE DRUG PRODUCT RELEASE 1 3 4 5 6
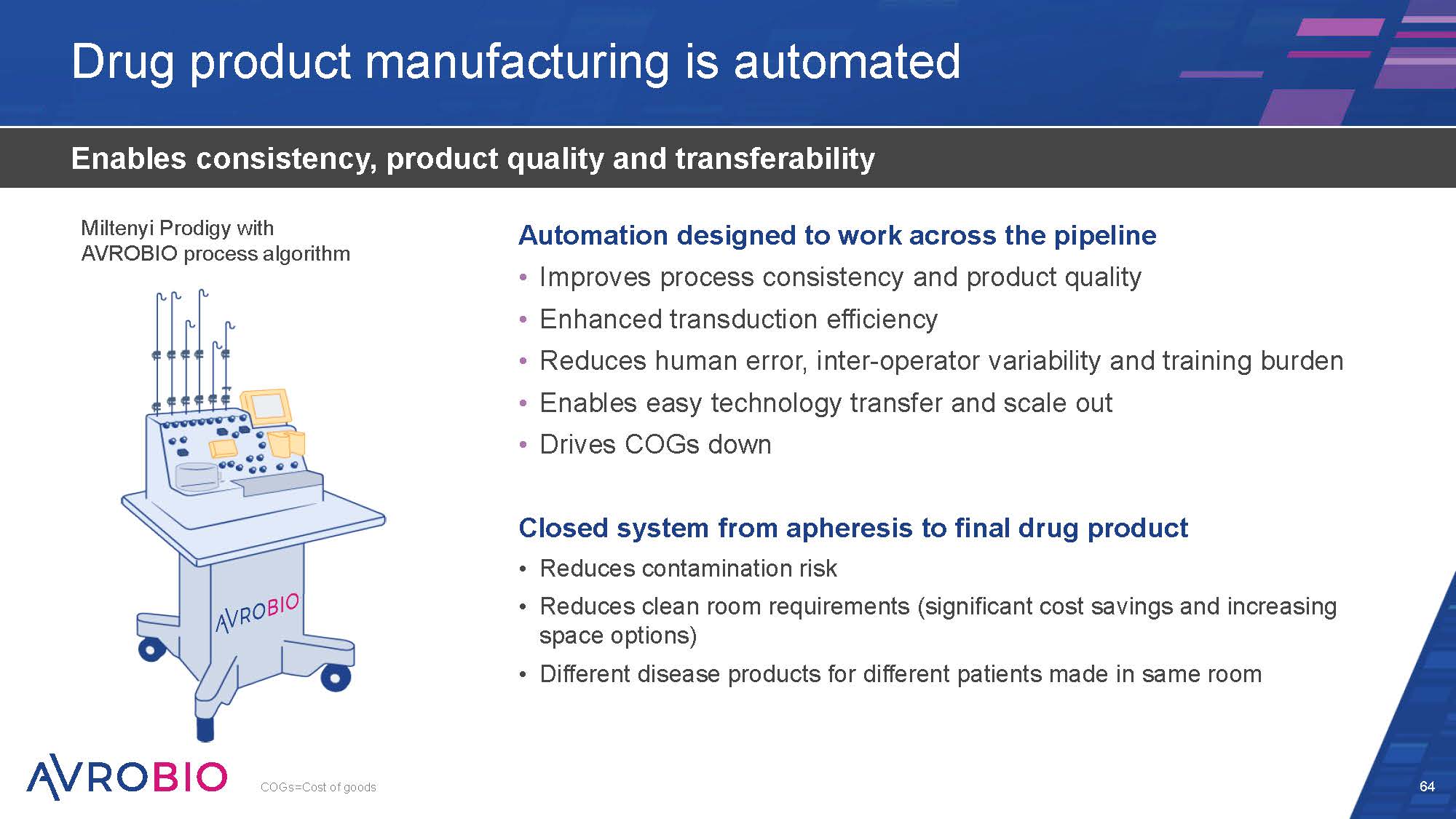
Drug product manufacturing is automated 64 Enables consistency, product quality and transferability Automation designed to work across the pipeline Improves process consistency and product quality Enhanced transduction efficiency Reduces human error, inter-operator variability and training burden Enables easy technology transfer and scale out Drives COGs down Closed system from apheresis to final drug product Reduces contamination risk Reduces clean room requirements (significant cost savings and increasing space options) Different disease products for different patients made in same room Miltenyi Prodigy with AVROBIO process algorithm COGs=Cost of goods
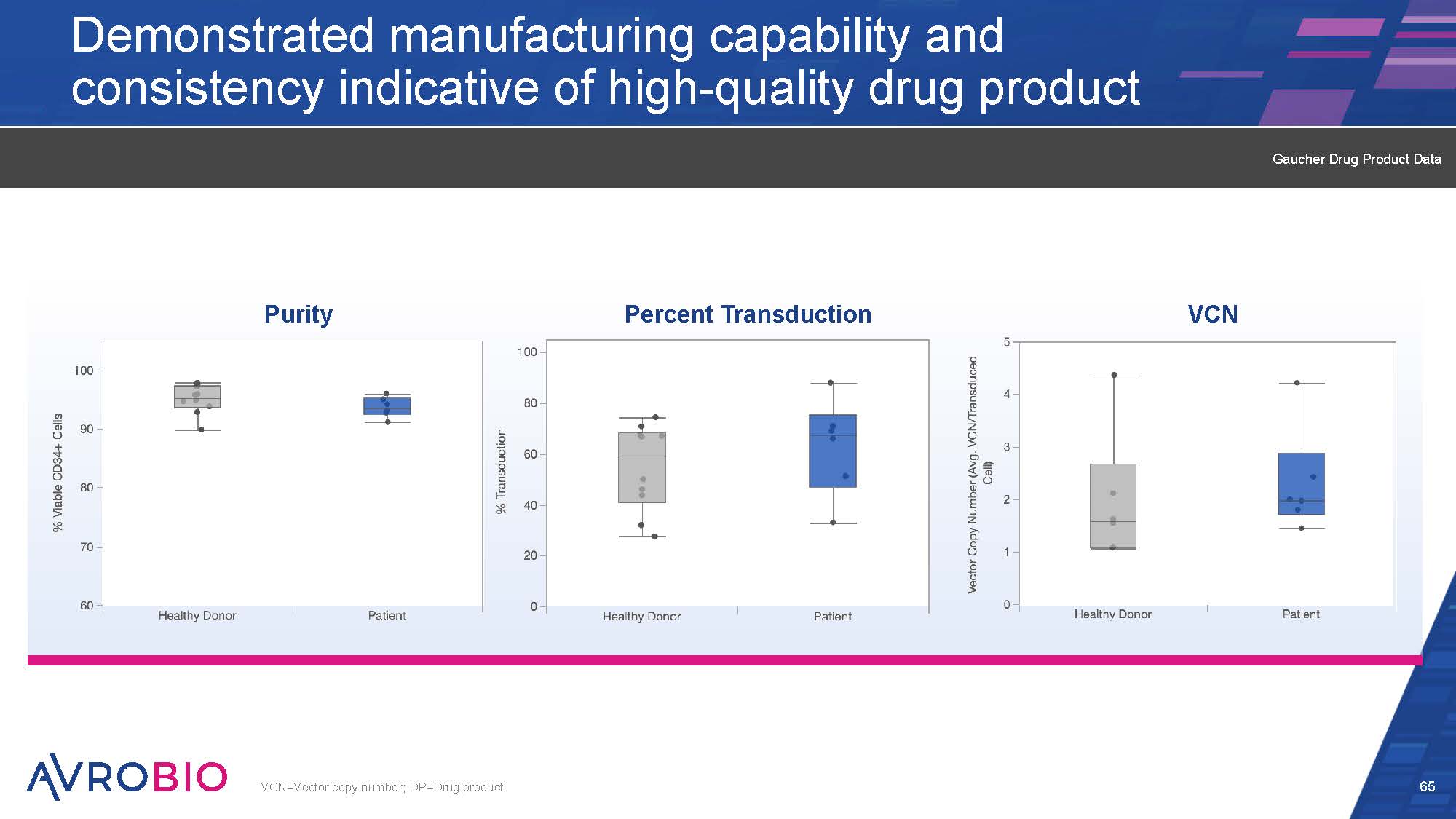
Demonstrated manufacturing capability and consistency indicative of high-quality drug product 65 VCN=Vector copy number; DP=Drug product VCN Purity Percent Transduction Gaucher Drug Product Data
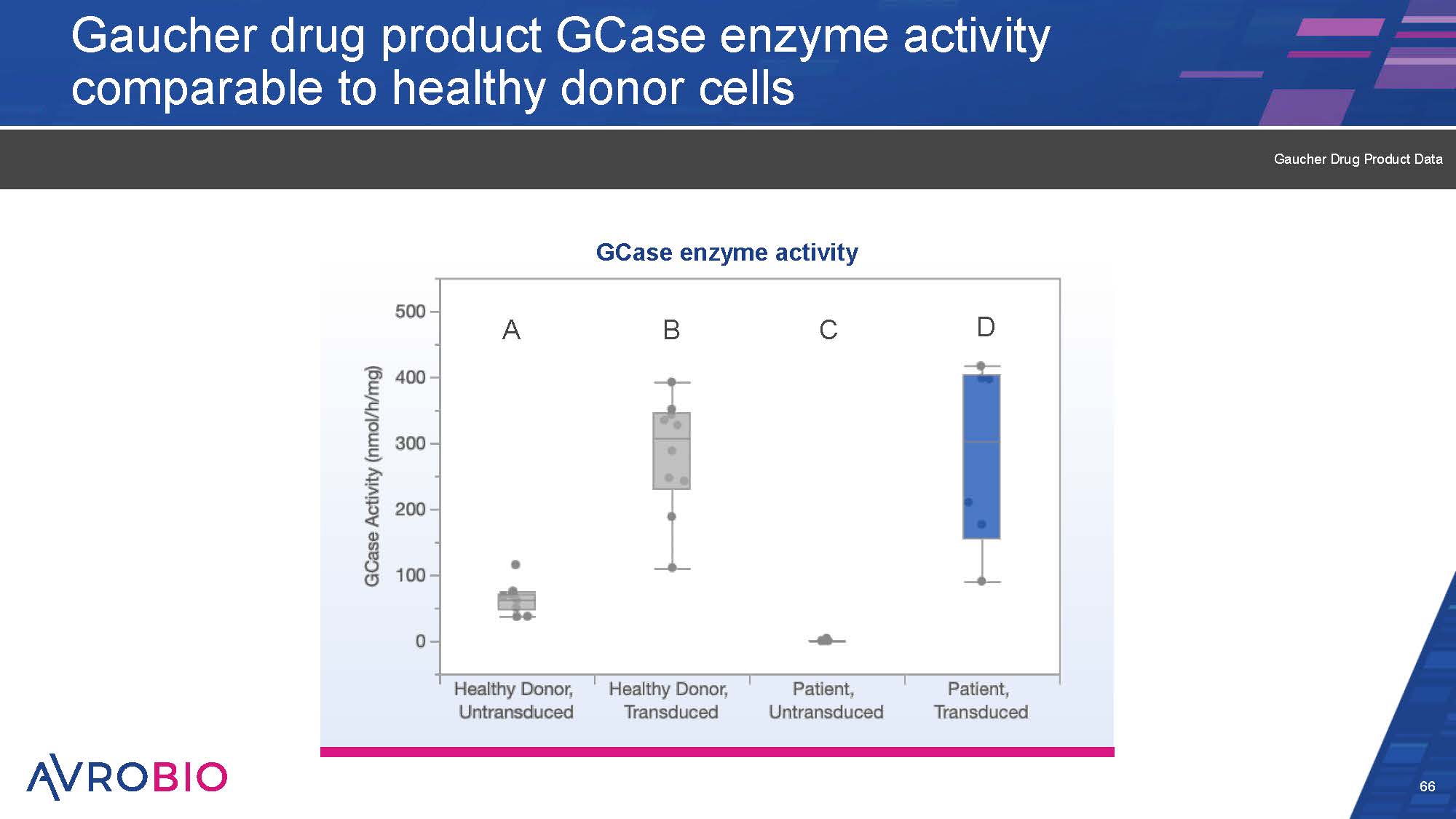
Gaucher drug product GCase enzyme activity comparable to healthy donor cells 66 Gaucher Drug Product Data A GCase enzyme activity B C D

Commercial scale lentiviral vector manufacturing 67 Robust vector production platform for the pipeline Commercial scale 200L serum free, suspension culture 50 patient doses per batch Optimized process, including fill/finish Minimal lot to lot variability Validated analytics Strong quality profile Low impurities No “empty” capsids with lentiviral vectors Consistent, high titer Biostat STR Bioreactor STR=Stirred tank bioreactor
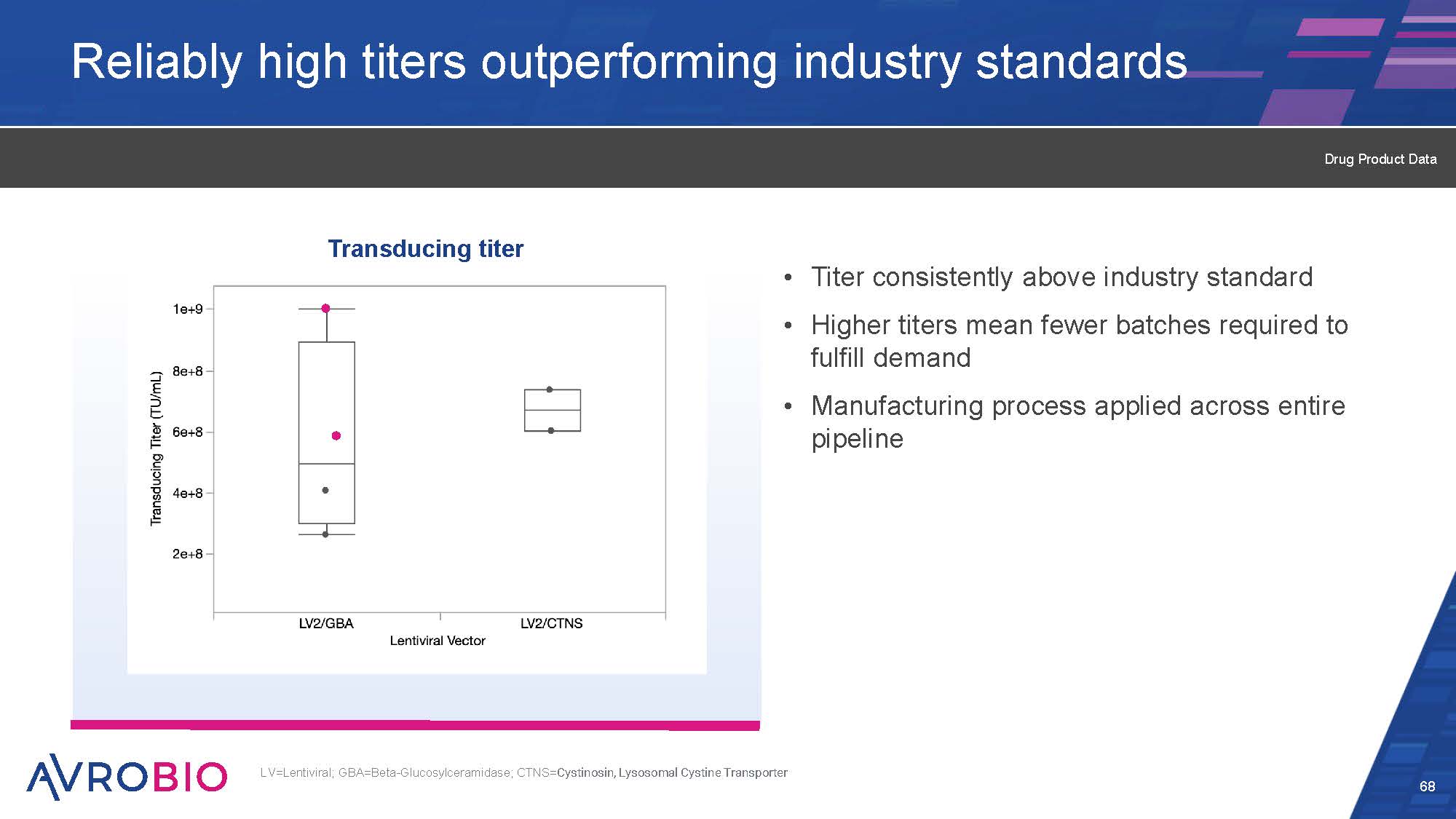
Reliably high titers outperforming industry standards Titer consistently above industry standard Higher titers mean fewer batches required to fulfill demand Manufacturing process applied across entire pipeline 68 LV=Lentiviral; GBA=Beta-Glucosylceramidase; CTNS=Cystinosin, Lysosomal Cystine Transporter Transducing titer Drug Product Data
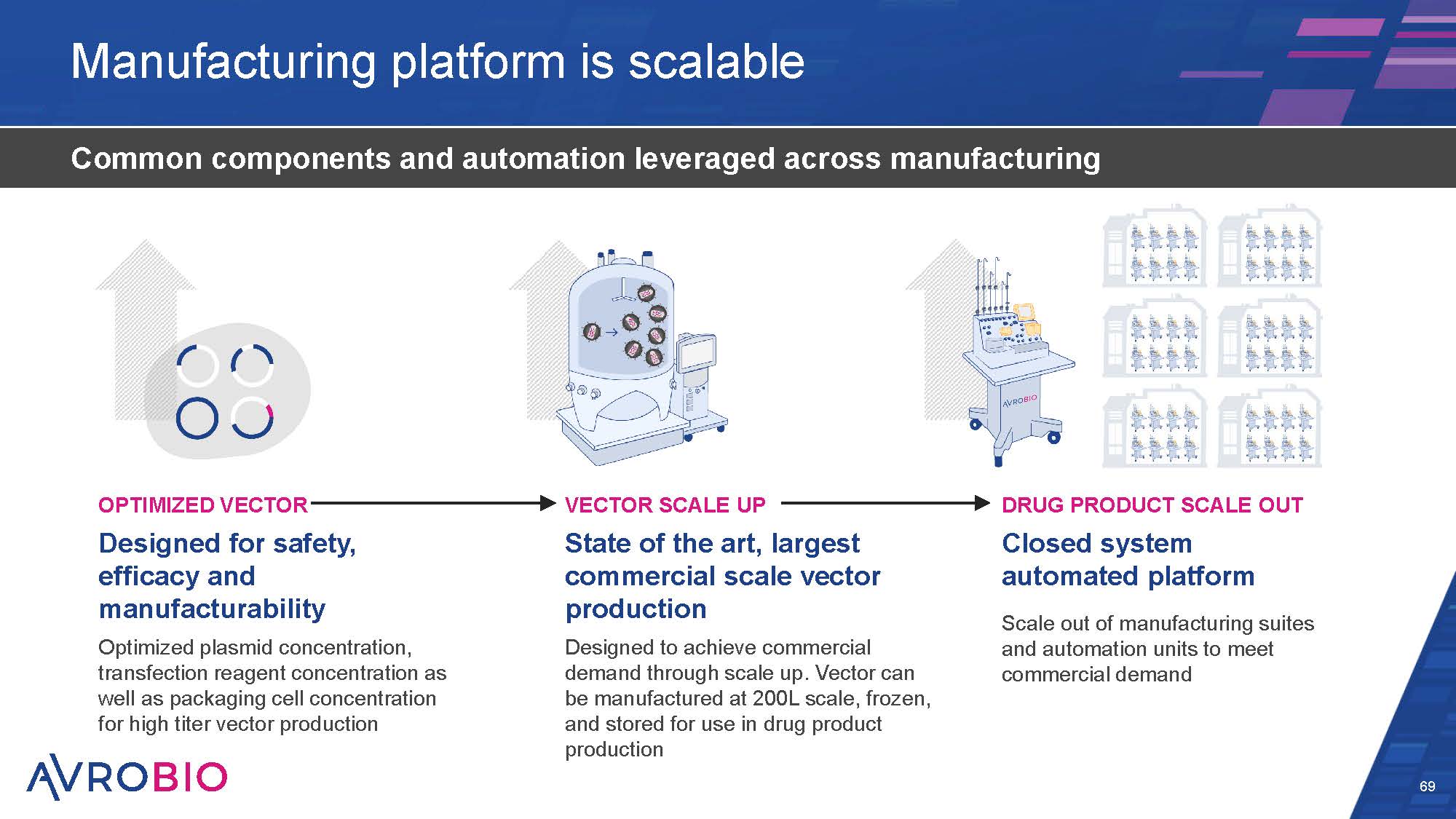
Manufacturing platform is scalable 69 Common components and automation leveraged across manufacturing DRUG PRODUCT SCALE OUT Closed system automated platform Scale out of manufacturing suites and automation units to meet commercial demand OPTIMIZED VECTOR Designed for safety, efficacy and manufacturability Optimized plasmid concentration, transfection reagent concentration as well as packaging cell concentration for high titer vector production VECTOR SCALE UP State of the art, largest commercial scale vector production Designed to achieve commercial demand through scale up. Vector can be manufactured at 200L scale, frozen, and stored for use in drug product production
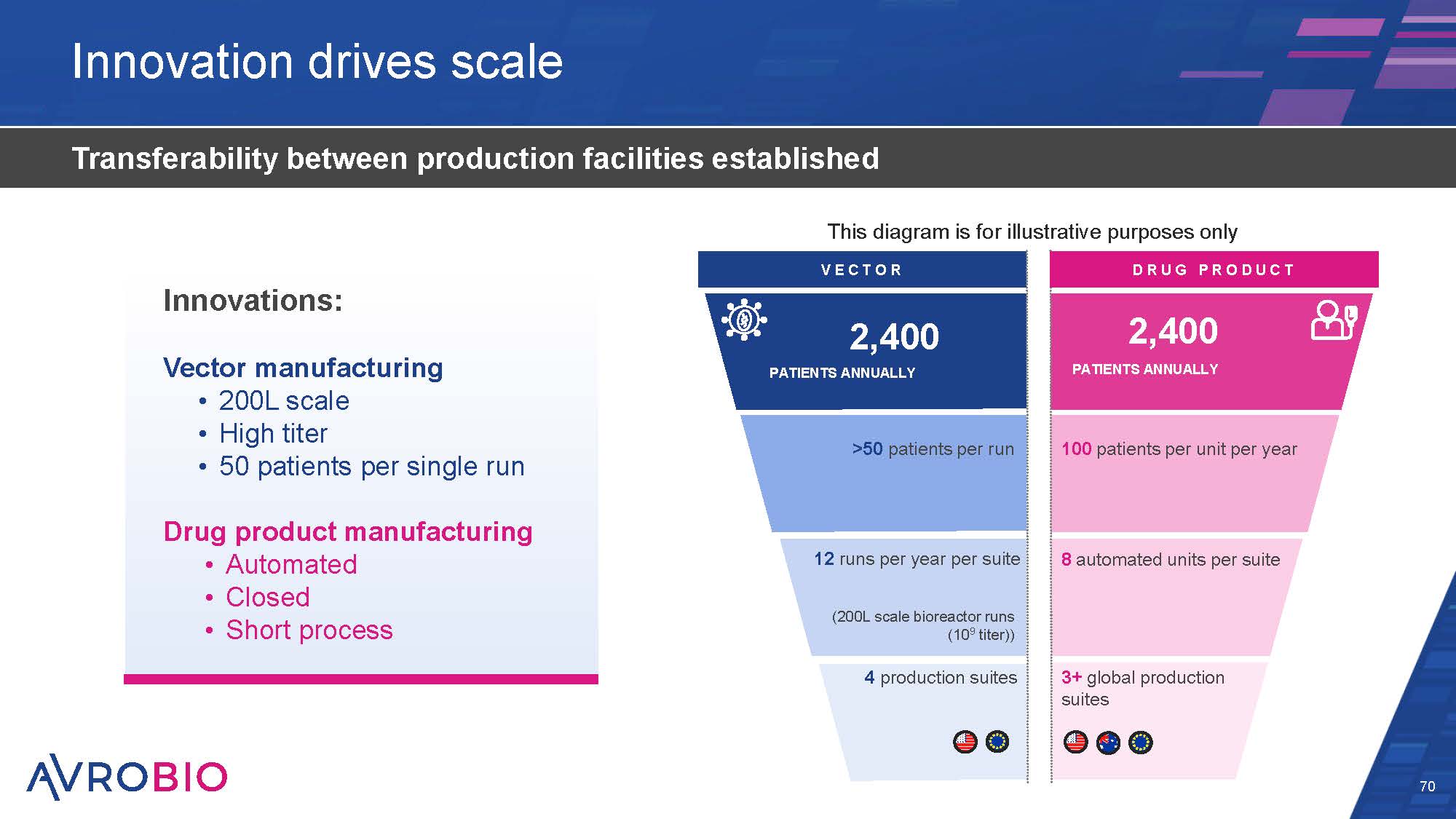
Innovation drives scale 70 Transferability between production facilities established DRUG P RO DUCT VE C T O R (200L scale bioreactor runs (109 titer)) 4 production suites 12 runs per year per suite >50 patients per run 2,400 PATIENTS ANNUALLY 3+ global production suites 8 automated units per suite 100 patients per unit per year 2,400 PATIENTS ANNUALLY This diagram is for illustrative purposes only Innovations: Vector manufacturing 200L scale High titer 50 patients per single run Drug product manufacturing Automated Closed Short process
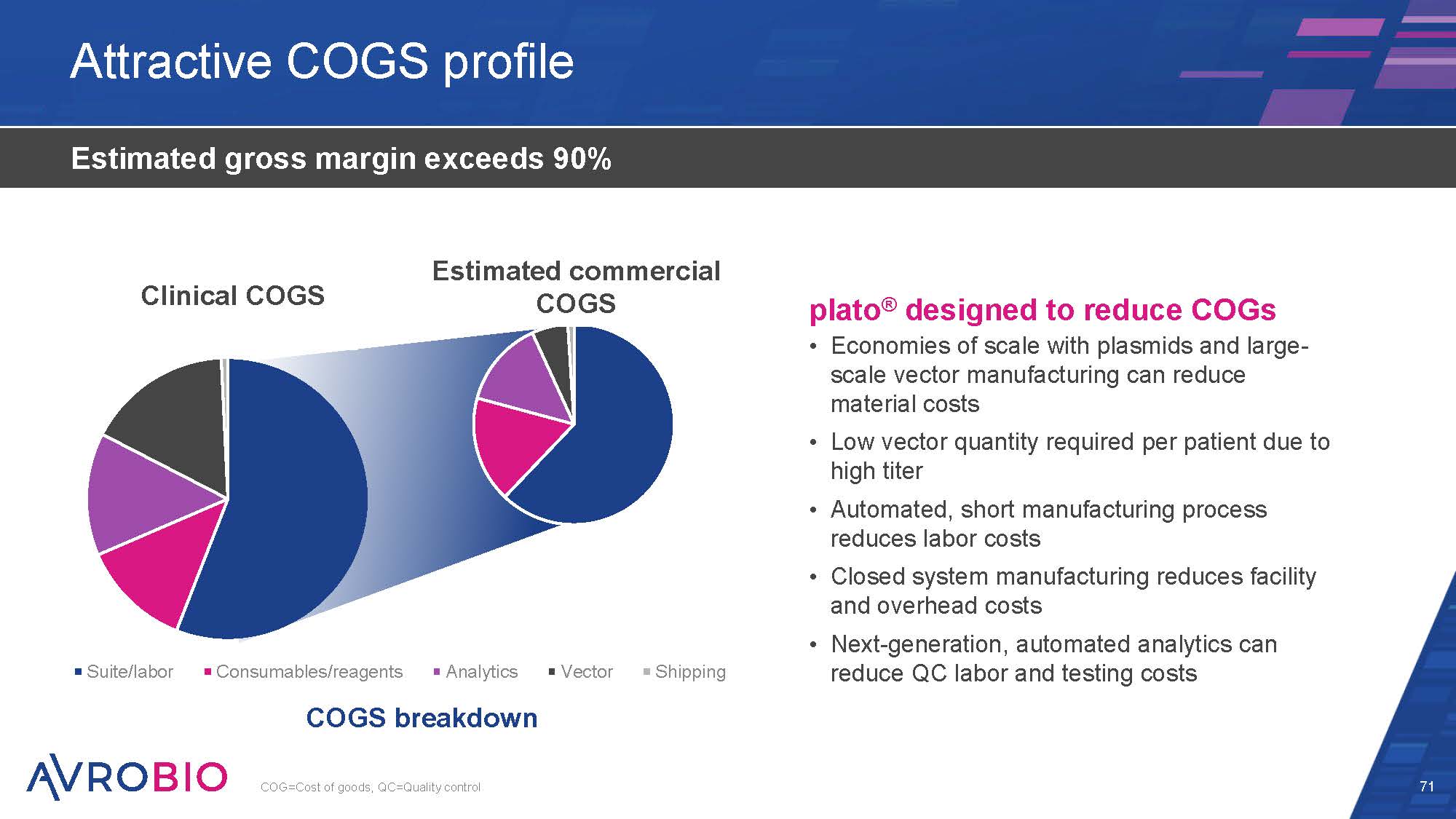
Actual clinical COGS ~$265k Suite/labor Consumables/reagents Analytics COGS breakdown Vector Shipping Attractive COGS profile 71 Estimated gross margin exceeds 90% plato® designed to reduce COGs Economies of scale with plasmids and large- scale vector manufacturing can reduce material costs Low vector quantity required per patient due to high titer Automated, short manufacturing process reduces labor costs Closed system manufacturing reduces facility and overhead costs Next-generation, automated analytics can reduce QC labor and testing costs COG=Cost of goods, QC=Quality control Estimated commercial COGS ~$150k Clinical COGS Estimated commercial COGS

Strategic investment in technology laid foundation for our manufacturing platform 72 Manufacturing Robust production platform Unrivalled LV manufacturing Scalable from plasmid to drug product Global footprint Cleared for the clinic from multiple agencies Cost effective Intended to address key COGs issues Analytics Robust platform analytics Best-in-class VCN assay First-in-class transduction assay Deep product characterization First-in-class single cell analytics Potency assay matrix Intended to accelerate regulatory approvals VCN=Vector copy number; LV-Lentiviral; COGs=Cost of goods

CO NF I DE NT I AL Arianna living with Gaucher disease type 3 No reported cases of insertional oncogenesis in AVROBIO clinical trials No evidence of persistent dominant clonal expansion in any AVROBIO clinical trials AVROBIO used state-of-the-art vectors and assessed vector safety before entering clinic Recent Advances in Vector Safety Key takeaways

No reported cases of insertional oncogenesis across lentiviral HSC gene therapy programs outside of CALD 3 insertional oncogenic events 0 insertional oncogenic events 1 vector construct + indication 67 patients LVV-MND-ABCD1 for CALD … All other gene therapies using lentiviral vectors … 16 vector constructs + indications 55 clinical trials 314 patients 72nd Cellular, Tissue and Gene Therapies Advisory Committee June 9-10, 2022; Tucci et al., 2022 (updated manually for the last two years with publicly available patient numbers from bluebird, Orchard Therapeutics, Rocket Pharma, and AVROBIO) CALD=Cerebral adrenoleukodystrophy; LVV=Lentiviral vector 74

AVROBIO state-of-the-art vector Vector design elements Replication incompetent SIN modified to abolish viral LTR promoter/enhancer activity EF1ɑ/EFS nonviral promoter with greatly reduced enhancer activity Used in at least 6 indications with 75 patients, out up to 10 years Kozak sequence to direct correct start of translation Codon optimization to optimize expression and remove cryptic splice sites WPRE Increase transgene expression and reduce readthrough to neighboring genes Modified to reduce potential toxicity of regulatory element caused by WHV X protein 75 Designed with the highest safety standards and tested extensively ΔU3 R U5 EFS Codon-optimized Transgene WPRE ΔU3 R U5 5’ LTR 3’ LTR RRE AVROBIO’s plato® vector Tucci et al., 2022; SIN=Self inactivated; LTR=Long terminal repeat; EFS=Factor 1alpha binding sequence; WPRE=Woodchuck Hepatitis Virus (WHP) posttranscriptional regulatory element
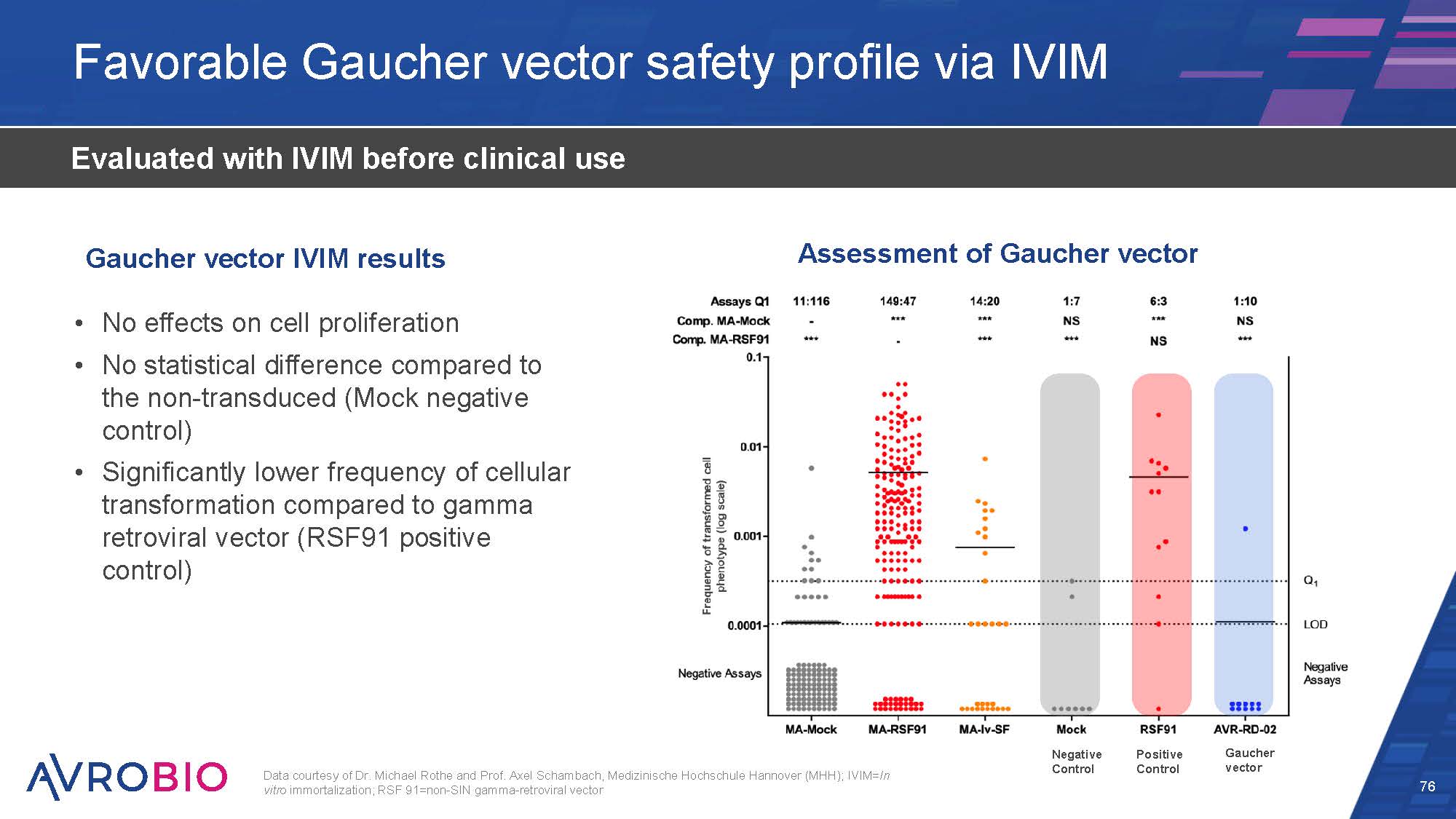
Favorable Gaucher vector safety profile via IVIM 76 Evaluated with IVIM before clinical use No effects on cell proliferation No statistical difference compared to the non-transduced (Mock negative control) Significantly lower frequency of cellular transformation compared to gamma retroviral vector (RSF91 positive control) Gaucher vector IVIM results Data courtesy of Dr. Michael Rothe and Prof. Axel Schambach, Medizinische Hochschule Hannover (MHH); IVIM=In vitro immortalization; RSF 91=non-SIN gamma-retroviral vector Assessment of Gaucher vector Gaucher vector Positive Control Negative Control
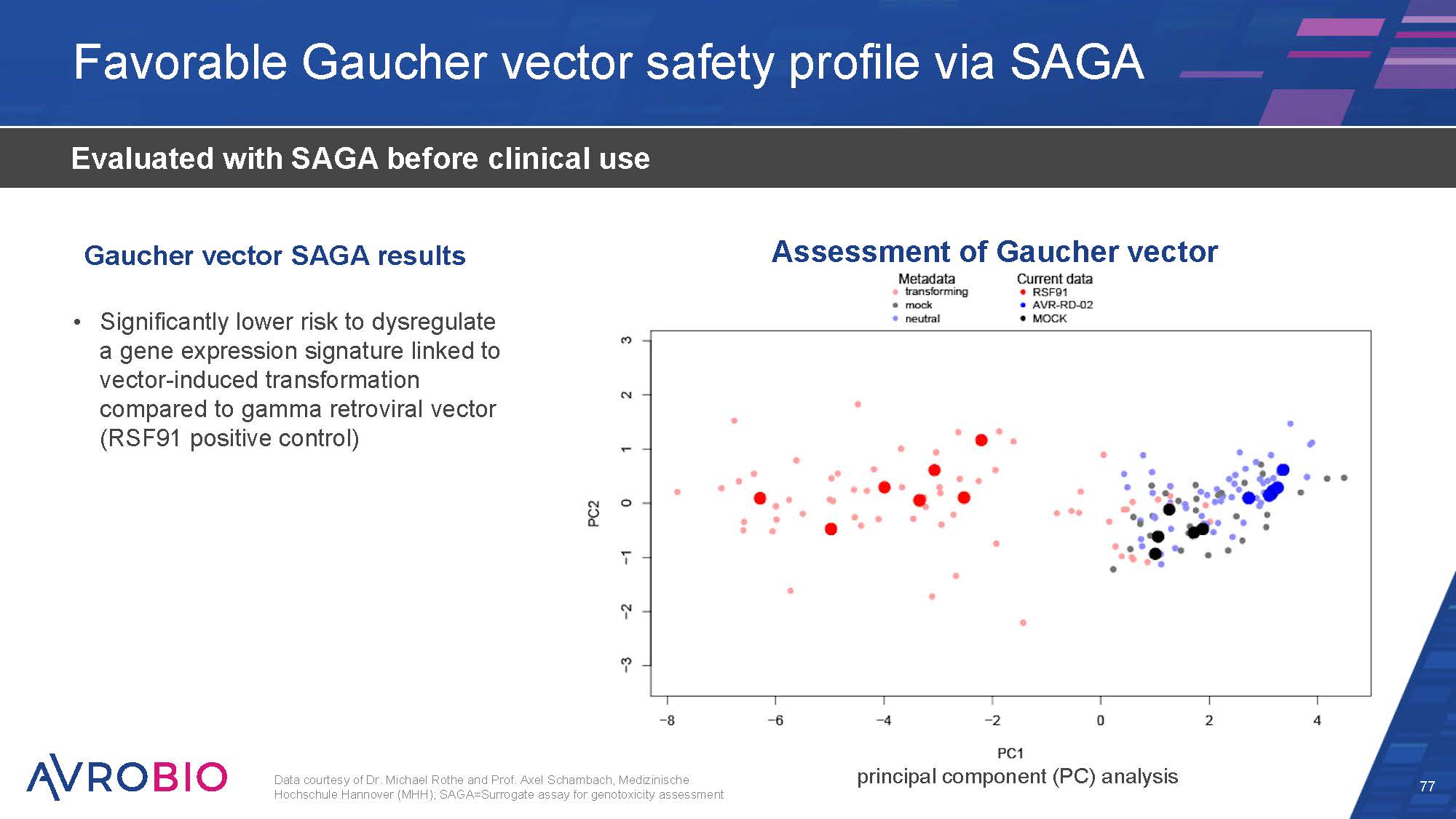
Favorable Gaucher vector safety profile via SAGA 77 Evaluated with SAGA before clinical use Significantly lower risk to dysregulate a gene expression signature linked to vector-induced transformation compared to gamma retroviral vector (RSF91 positive control) Data courtesy of Dr. Michael Rothe and Prof. Axel Schambach, Medizinische Hochschule Hannover (MHH); SAGA=Surrogate assay for genotoxicity assessment Assessment of Gaucher vector Gaucher vector SAGA results principal component (PC) analysis

Commitment to vector safety across platform 78 No reported cases of insertional oncogenesis in AVROBIO clinical trials No evidence of persistent dominant clonal expansion in AVROBIO clinical trials Developed and used state-of-the-art vector designed with safety features Rigorously test using state-of-the-art vector safety assays to assess risk of insertional oncogenesis before entering clinic

Today’s agenda 79 What if one gene could change your life?: The GBA gene and Gaucher disease Welcome and opening remarks – Geoff MacKay, AVROBIO Arianna and Veronica’s story: Living with Gaucher disease type 3 The role of GBA in Gaucher Disease – Timothy Cox, M.D., MAE, FRCP, FMedSci, University of Cambridge, UK Fulfilling the one-gene promise: AVROBIO’s Gaucher disease program Gaucher disease type 1 data – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Gaucher disease type 3 data – Rob Wynn, M.D. (Camb), MB BChir, MRCP, FRCPath, Royal Manchester Children’s Hospital, and Simon Jones, M.D., BSc, MRCPCH, Manchester Centre for Genomic Medicine at Saint Mary’s Hospital, UK Paving a clinical path: AVROBIO’s strategy for advancing AVR-RD-02 Development and design of clinical trials for Gaucher disease – Essra Ridha, M.D., MRCP, FFPM, AVROBIO Delivering for patients: CMC and analytics to execute on the one-gene promise Deploying the plato® advantage – Azadeh Golipour, Ph.D., AVROBIO Recent advances in vector safety – Azadeh Golipour, Ph.D., AVROBIO Closing remarks and Q&A

CO NF I DE NT I AL Closing remarks Arianna living with Gaucher disease type 3
Building a leading Gaucher disease program 81 First mover advantage Program targeting multi-billion dollar market opportunity AVROBIO transitioning into a late-stage company in 2023 Key takeaways from today: GD1 – expanding positive data set GD3 – initial data with early signs of clinical activity GD3 – pursue one global pediatric Phase 2/3 trial Plan to utilize combined data set for GD1 and GD3 for Gaucher program development approach Manufacturing late-stage trial ready, no CMC changes anticipated Attractive commercial opportunity with large, pre-identified patient population

Late-stage clinical development Early-stage clinical development Preclinical AVROBIO entering late-stage development 82 Planned regulatory milestones subject to regulatory agency clearance; *Collaborator-sponsored Phase 1/2 clinical trial of AVR-RD-04 is funded in part by grants to UCSD from the California Institute for Regenerative Medicine (CIRM), Cystinosis Research Foundation (CRF), and National Institutes of Health (NIH); ILAP=Innovative Licensing and Access Pathway; ODD=Orphan drug designation; RPDD=Rare pediatric drug designation Indication Cystinosis AVR-RD-04* Hunter AVR-RD-05 Pompe AVR-RD-03 Gaucher AVR-RD-02 Regulatory designations RPDD; Fast Track; ODD (US, EU); ILAP (UK) RPDD; Fast Track; ODD (US, EU) RPDD; ODD (US) Planned to initiate in 2023 Planned to initiate in 2023 Planned to initiate in 2023
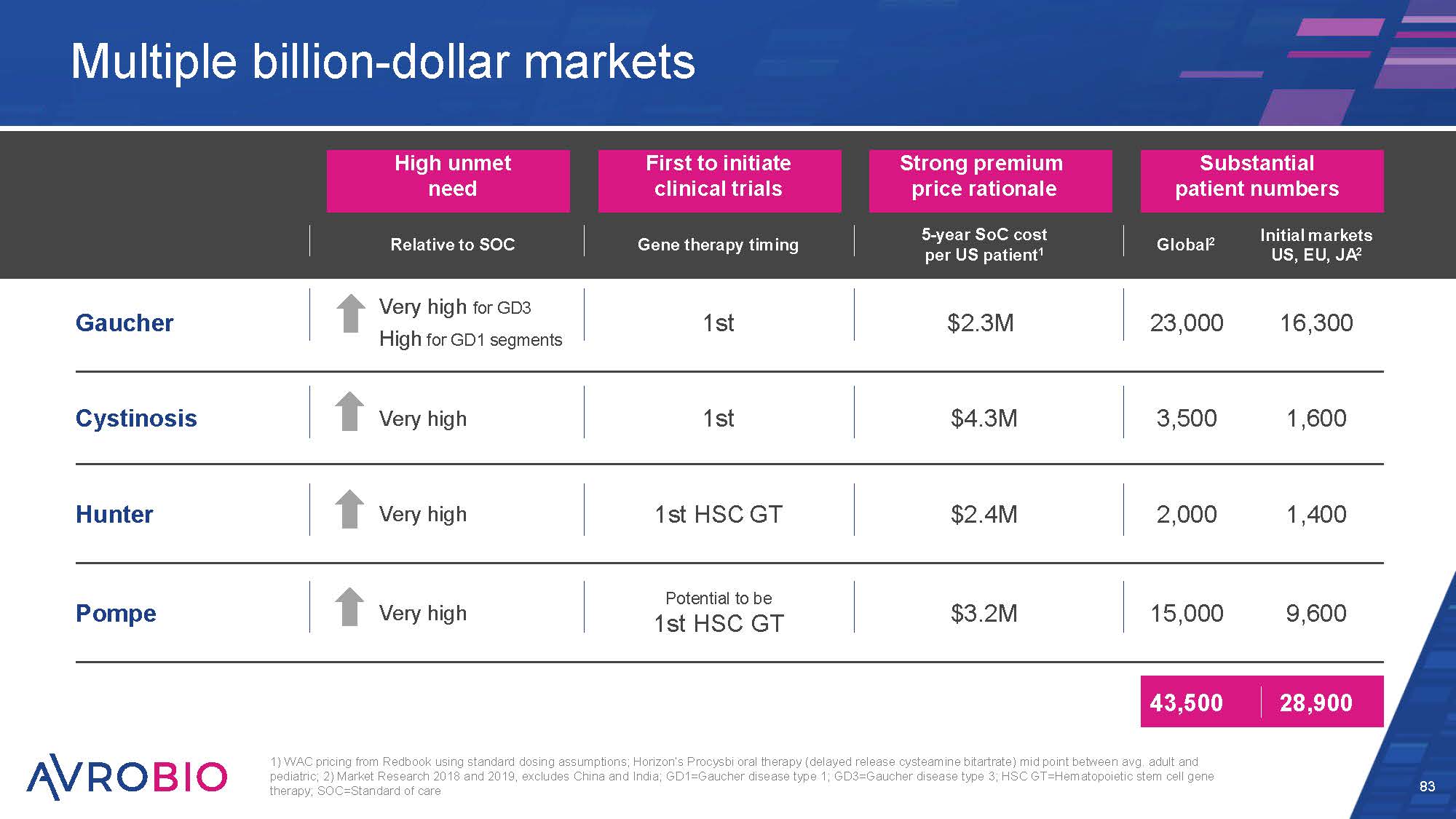
High unmet need First to initiate clinical trials Strong premium price rationale Substantial patient numbers Relative to SOC Gene therapy timing 5-year SoC cost per US patient1 Global2 Initial markets US, EU, JA2 Gaucher Very high for GD3 High for GD1 segments 1st $2.3M 23,000 16,300 Cystinosis Very high 1st $4.3M 3,500 1,600 Hunter Very high 1st HSC GT $2.4M 2,000 1,400 Pompe Very high Potential to be 1st HSC GT $3.2M 15,000 9,600 43,500 28,900 Multiple billion-dollar markets 83 1) WAC pricing from Redbook using standard dosing assumptions; Horizon’s Procysbi oral therapy (delayed release cysteamine bitartrate) mid point between avg. adult and pediatric; 2) Market Research 2018 and 2019, excludes China and India; GD1=Gaucher disease type 1; GD3=Gaucher disease type 3; HSC GT=Hematopoietic stem cell gene therapy; SOC=Standard of care

Key anticipated 2023 milestones 84 FDA=Food and Drug Administration; 2H=Second Half; MHRA=Medicines and Healthcare products Regulatory Agency Gaucher AVR-RD-02 Cystinosis AVR-RD-04 Hunter AVR-RD-05 Initiate Phase 2/3 clinical trial for GD3 in 2H 2023 Complete enrollment in Guard1 by year end 2023 Dose first patient in collaborator-sponsored Phase 1/2 trial early 2023 Share initial patient data in 2H 2023 Engage with MHRA on clinical trial design in 1Q 2023 Initiate late-stage clinical trial activities in 2H 2023

FDA: Food and Drug Administration; 2H: Se cond Half THANK YOU