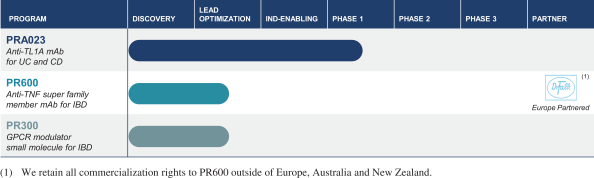
trials by Gilead Sciences, Inc. We are also aware of additional product candidates in clinical trials by AbbVie Inc., Abivax SA, Amgen Inc., Arena Pharmaceuticals, Inc., C.H. Boehringer Sohn AG & Ko. KG, Bristol-Myers Squibb Company, Celgene Corporation, Gilead Sciences, Inc., GlaxoSmithKline plc, Gossamer Bio, Inc., Incyte Corp., Janssen Pharmaceutica N.V., Landos Biopharma, Inc., Protagonist Therapeutics, Inc., Theravance Biopharma, Inc., Applied Molecular Transport Inc., Pandion Therapeutics, Inc., RedHill Biopharma Ltd. and Seres Therapeutics, Inc.
Government Regulation
Government authorities in the United States, at the federal, state and local level, and other countries extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, marketing, post-approval monitoring and reporting, pricing, and export and import of diagnostic tests and pharmaceutical products such as those we are currently marketing and developing. The process of obtaining and maintaining regulatory approvals and the subsequent compliance with appropriate federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources.
U.S. Regulation of Diagnostic Tests and other Medical Devices
In the United States, the laws and regulations governing the marketing of diagnostic products are evolving, extremely complex, and in many instances, there are no significant regulatory or judicial interpretations of these laws and regulations. The Federal Food, Drug and Cosmetic Act (FDCA) defines a medical device to include any instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory, intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals. Among other things, pursuant to the FDCA and its implementing regulations, the FDA regulates the research, testing, manufacturing, safety, labeling, storage, recordkeeping, premarket clearance or approval, marketing and promotion and sales and distribution of medical devices in the United States to ensure that medical products distributed domestically are safe and effective for their intended uses.
PMA Pathway
We are developing companion diagnostics to be used in connection with our therapeutic product candidates, if approved. These companion diagnostics are regulated by the FDA as medical devices. The FDA categorizes medical devices into one of three classes—Class I, II, or III—based on the risks presented by the device and the regulatory controls necessary to provide a reasonable assurance of the device’s safety and effectiveness. Class I includes devices with the lowest risk to the patient and are those for which safety and effectiveness can be assured by adherence to the FDA’s General Controls for medical devices, which include compliance with the applicable portions of the Quality System Regulation (QSR) facility registration and product listing, reporting of adverse medical events, and truthful and non-misleading labeling, advertising, and promotional materials. Class II devices are subject to the FDA’s General Controls, and special controls as deemed necessary by the FDA to ensure the safety and effectiveness of the device. Special controls are established by the FDA for a specific device type and often include specific labeling provisions, performance metrics, and other types of controls that mitigate risks of the device (usually incorrect results for an in-vitro diagnostic device (IVD)). Devices deemed by the FDA to pose the greatest risks, such as life sustaining, life supporting or some implantable devices, or devices that have a new intended use, or use advanced technology that is not substantially equivalent to that of a legally marketed device, are placed in Class III, requiring approval of an application for premarket approval (PMA). Some pre-amendment devices are unclassified, but are subject to the FDA’s premarket notification and clearance process in order to be commercially distributed.
Class III devices, including most companion diagnostics, generally require PMA approval before they can be marketed. Obtaining PMA approval requires the submission of “valid scientific evidence” to the FDA to
- 118 -