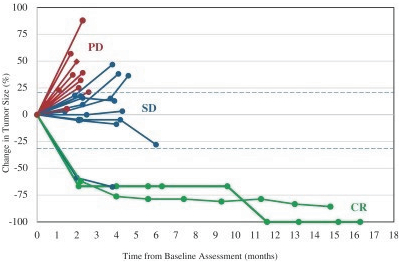
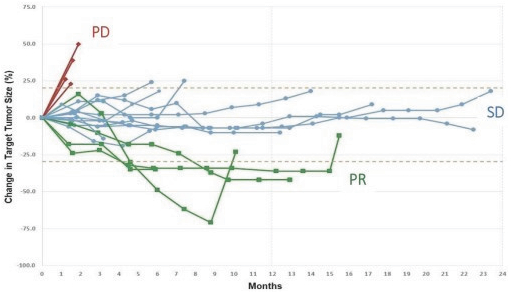
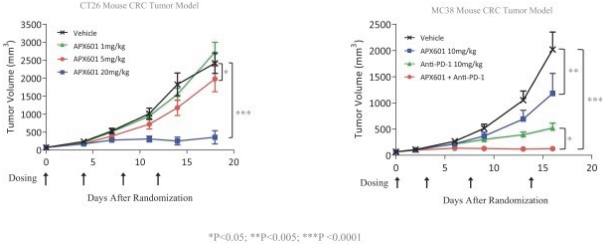
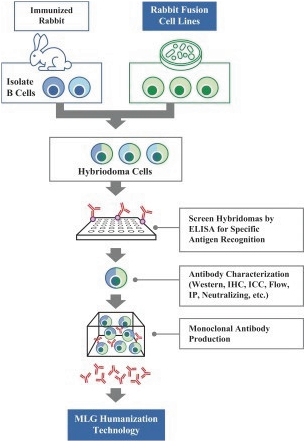
In addition, the distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, or “PDMA,” which regulates the distribution of drugs and drug samples at the federal level, and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution.
Other U.S. Regulatory Matters
Manufacturing, sales, promotion and other activities following product approval are also subject to regulation by numerous regulatory authorities in the United States in addition to the FDA, including the CMS, other divisions of the HHS, the Department of Justice, the Drug Enforcement Administration, the Consumer Product Safety Commission, the Federal Trade Commission, the Occupational Safety & Health Administration, the Environmental Protection Agency, and state and local governments.
For example, in the United States, financial arrangements with healthcare providers and other business arrangements, including, but not limited to, sales, marketing and scientific and educational programs, also must comply with state and federal healthcare fraud and abuse laws. These laws include, without limitation, state and federal anti-kickback, fraud and abuse, false claims, privacy and security, and transparency and reporting laws and regulations related to drug pricing and payments and other transfers of value made to physicians and other healthcare providers. Violation of any of such laws or any other governmental regulations that apply, may result in penalties, including, without limitation, administrative, civil and criminal penalties, damages, fines, disgorgement, the curtailment or restructuring of operations, integrity oversight and reporting obligations, exclusion from participation in federal and state healthcare programs and imprisonment. In particular, the federal Anti-Kickback Statute makes it illegal for any person, including a prescription drug manufacturer (or a party acting on its behalf), to knowingly and willfully solicit, receive, offer or pay any remuneration that is intended to induce or reward referrals, including the purchase, recommendation, order or prescription of a particular drug, for which payment may be made under a federal healthcare program, such as Medicare or Medicaid. Violations of this law are punishable by up to five years in prison, criminal fines, administrative civil money penalties and exclusion from participation in federal healthcare programs. Moreover, the ACA provides that the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act. HIPAA also created additional federal civil and criminal penalties for, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing, or covering up a material fact or making any materially false, fictitious, or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items, or services. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
The ACA, through the Physician Payments Sunshine Act, imposes new reporting requirements on drug manufacturers for payments made by them to physicians and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Drug manufacturers are required to submit annual reports to the government and these reports are posted on a website maintained by CMS. Certain states also mandate implementation of compliance programs, impose restrictions on drug manufacturer marketing practices, and/or require the tracking and reporting of gifts, compensation, and other remuneration to physicians.
We may also be subject to data privacy and security requirements that may impact the way in which we conduct research and operate our business. HIPAA, as amended by HITECH, and their respective implementing regulations, including the final omnibus rule published on January 25, 2013, impose obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security, and transmission of individually identifiable health information on covered entities, including certain healthcare providers, health plans, and
127