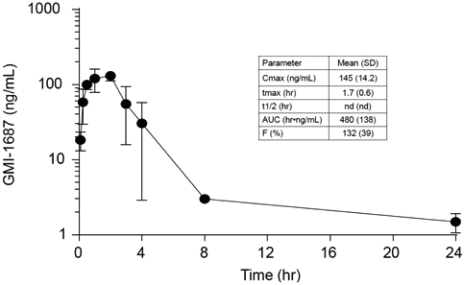
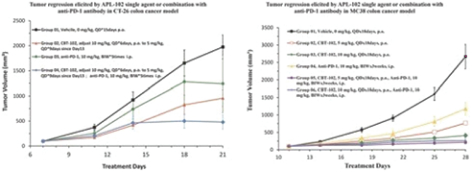
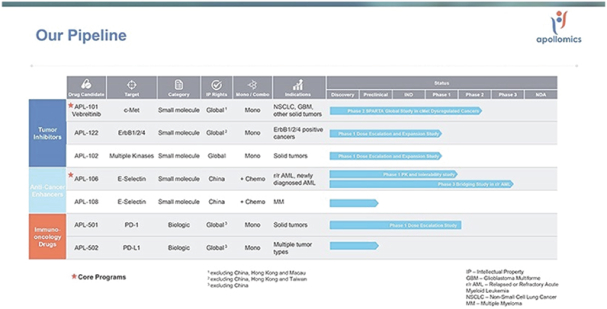
We conduct our business under the brand name of “Apollomics.” As of the date of this prospectus, we had primarily registered 14 trademarks/classes in China, 2 trademarks/classes in the United States, and 24 trademarks/classes in Hong Kong.
We enter into collaboration agreements and other relationships with pharmaceutical companies and other industry participants to leverage our intellectual property and gain access to the intellectual property of others. Please refer to “—Licensing and Collaboration Arrangements” above for further details.
As of the date of this prospectus, we were not involved in any proceedings in respect of, and we had not received notice of any claims of infringement of, any intellectual property rights that were threatened or pending, in which we were a claimant or a respondent.
Our directors confirm that as of the date of this prospectus, there had been no instance in our R&D activities of drug candidates, including APL-101, that may give rise to a claim of infringement of intellectual property rights by any third party for injunctive relief or actual damages because the jurisdictions where we are conducting R&D of drug candidates exempt R&D activities from obtaining regulatory approvals for patent infringements. Such jurisdictions are Australia, Canada, China, Finland, France, Hungary, Italy, New Zealand, Russia, Singapore, Spain, Taiwan, the United Kingdom, Ukraine and the United States.
Government Regulations
Government authorities in the United States, at the federal, state and local level, in China, and in other countries and jurisdictions, extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of pharmaceutical products. The processes for obtaining regulatory approvals in the United States, in China and in other foreign countries and jurisdictions, along with subsequent compliance with applicable statutes and regulations and other regulatory authorities, require the expenditure of substantial time and financial resources.
United States regulation of pharmaceutical product development and approval
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The FDC Act and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Pharmaceutical products—such as small molecule drugs and biological products, or biologics—used for the prevention, treatment, or cure of a disease or condition of a human being are subject to regulation under the FDC Act, with the exception that the section of the FDC Act that governs the approval of drugs via NDAs does not apply to the approval of biologics. In contrast, biologics are approved for marketing under provisions of the Public Health Service Act, or PHS Act, via a BLA. However, the application process and requirements for approval of BLAs are very similar to those for NDAs. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as clinical hold, FDA refusal to approve pending NDAs or BLAs, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal prosecution.
Pharmaceutical product development for a new product or certain changes to an approved product in the United States typically involves nonclinical laboratory and animal tests, the submission to the FDA of an IND, which must become effective before clinical testing may commence in the United States, and adequate and well- controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity, and novelty of the product or disease.
180