biopharmaceutical, biotechnological and therapeutics companies. Moreover, we may also compete with universities and other research institutions that may be active in oncology research and could be in direct competition with us. We also compete with these organizations to recruit management, scientists and clinical development personnel, and our inability to compete successfully could negatively affect our level of expertise and our ability to execute our business plan. We will also face competition in establishing clinical trial sites, enrolling patients for clinical trials and identifying and in-licensing intellectual property related to future product candidates, as well as entering into collaborations, joint ventures, license agreements and other similar arrangements. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
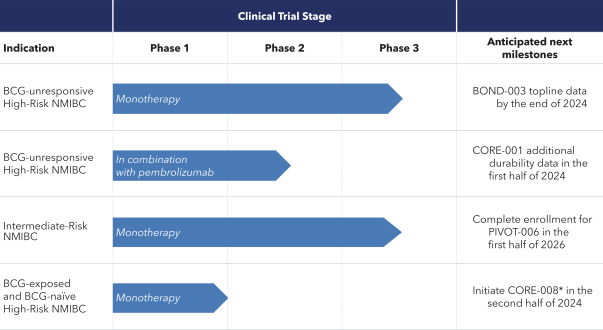
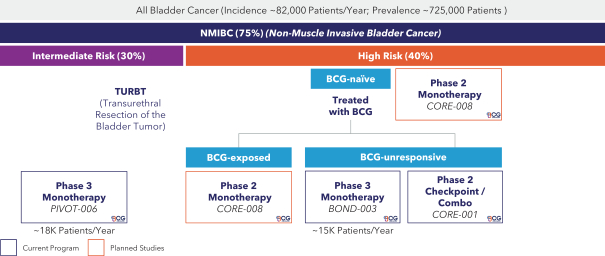
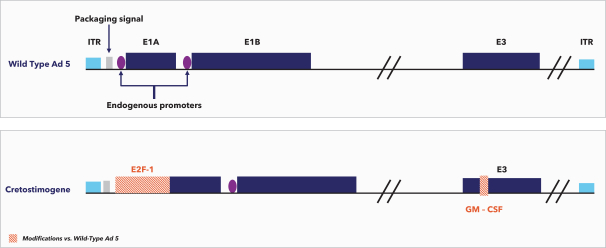
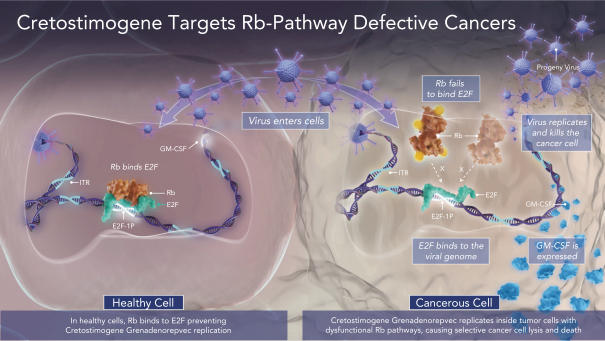
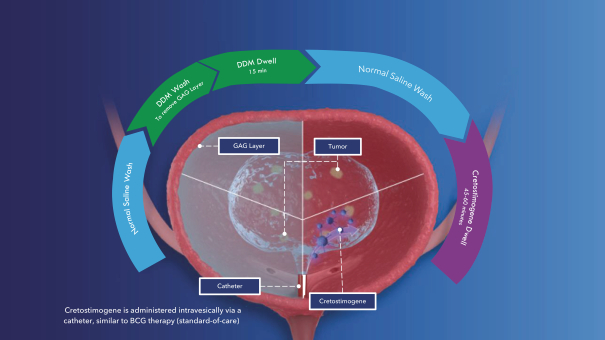
If cretostimogene or any future product candidates are approved, they will compete with surgery, radiation, and drug therapy, including chemotherapy, BCG, hormone therapy, biologic therapy, such as monoclonal and bispecific antibodies, antibody-drug conjugates, radiopharmaceuticals, immunotherapy, cell-based therapy, and targeted therapy, or a combination of any such methods, either approved or under development, which are intended to treat the same indications that we are targeting or may target, including through approaches that may prove to be more effective, have fewer side effects, be less costly to manufacture, be more convenient to administer or have other advantages over cretostimogene and any future product candidates. To the extent Merck & Co. (Merck) or another manufacturer increases the supply of BCG, there may be less demand for alternative treatments such as cretostimogene in BCG-naïve or BCG-exposed patients. There are numerous companies that have commercialized or are developing treatments for NMIBC that we will compete with, including Bristol Meyers Squibb, enGene Inc., Gilead Sciences, Inc., Hoffman-La Roche AG (Roche), ImmunityBio Inc., Johnson & Johnson Inc., Merck, Protara Therapeutics, Inc., Pfizer, Inc. and UroGen Pharma, Inc.
Many of our competitors have significantly greater financial, technical, manufacturing, marketing, sales and supply resources or experience than we do. If we successfully obtain approval for cretostimogene or any future product candidate, we will face competition based on many different factors, including the safety and effectiveness of our product candidates, the ease with which our product candidates can be administered, and the extent to which patients accept relatively new routes of administration, the timing and scope of regulatory approvals for these product candidates, the availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage, and patent position. Competing products could present superior treatment alternatives, including by being more effective, safer, more convenient, less expensive, or marketed and sold more effectively than any products we may develop. Competitive products may make cretostimogene or any future product candidates we develop obsolete or noncompetitive before we recover the expense of their development and commercialization. If we are unable to compete effectively, our opportunity to generate revenue from the sale of cretostimogene or any future product candidates we may develop, if approved, could be adversely affected.
If the market opportunities for cretostimogene or any future product candidates are smaller than we believe they are, our revenue may be adversely affected, and our business may suffer.
Cancer therapies are defined by lines of therapy as well as by treatment-naïve or previously-treated status. Often the initial approval for a new therapy is in later lines and subsequent approval in an earlier line may not be feasible. When cancer is detected early enough, first line therapy is sometimes adequate to cure the cancer or prolong life without a cure. Whenever first line therapy, including surgery, radiation therapy, targeted therapy, immunotherapy, chemotherapy, hormone therapy, or a combination of these, proves unsuccessful, second line therapy may be administered. Second line therapies often consist of additional chemotherapy, radiation, antibody drugs, tumor targeted small molecules, or a combination of these. Third line therapies can include antibody and small molecule targeted therapies, more invasive forms of surgery, and new technologies. In markets with approved therapies, there is no guarantee that cretostimogene or any future product candidate, even if approved, would be approved for second line or first line therapy. This could limit our potential market opportunity. In addition, we may have to conduct additional clinical trials prior to gaining approval for second line or first line therapy.
39