UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14D-9
(Rule 14d-101)
Solicitation/Recommendation Statement
Under Section 14(d)(4) of the Securities Exchange Act of 1934
Under Section 14(d)(4) of the Securities Exchange Act of 1934
HOME DIAGNOSTICS, INC.
(Name of Subject Company)
HOME DIAGNOSTICS, INC.
(Name of Person Filing Statement)
Common Stock, par value $.01 per share
(Title of Class of Securities)
(Title of Class of Securities)
437080104
(Cusip Number of Class of Securities)
(Cusip Number of Class of Securities)
Peter Ferola, Esq.
Vice President and General Counsel
Home Diagnostics, Inc.
2400 NW 55th Court
Fort Lauderdale, Florida 33309
(954) 677-9201
Vice President and General Counsel
Home Diagnostics, Inc.
2400 NW 55th Court
Fort Lauderdale, Florida 33309
(954) 677-9201
(Name, address and telephone number of person authorized to
receive notices and communications on behalf of
the person(s) filing statement)
receive notices and communications on behalf of
the person(s) filing statement)
With copies to:
Clifford E. Neimeth, Esq. | Edwin T. Markham, Esq. | |
Greenberg Traurig, LLP | Satterlee Stephens Burke & Burke LLP | |
MetLife Building | 230 Park Avenue | |
200 Park Avenue | New York, New York 10169 | |
New York, New York 10166 | (212) 818-9200 | |
(212) 801-9383 |
| þ | Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer. |

| Home DiagnosticsSpecial MeetingFebruary 3, 2010Joe Capper, CEORon Rubin, CFO |

| Forward Looking Statement The following presentation contains forward-looking statements and is subject to risks and uncertainties. Such statements are only predictions and reflect the Company's expectations and assumptions as of the date of this presentation, based on currently available operating, financial and competitive information. The actual events or results may differ materially from those projected in such forward-looking statements due to a number of factors, including risks and uncertainties identified in the Company's most recent annual report on Form 10-K and its other filings with the Securities and Exchange Commission. |

| Important Information about the Tender Offer The tender offer described in this presentation has not yet commenced and this communication is neither an offer to purchase nor the solicitation of an offer to sell any securities. At an appropriate time and if the tender offer is commenced, Home Diagnostics intends to file a tender offer solicitation / recommendation statement with the SEC. Investors and security holders are urged to read the tender offer solicitation / recommendation statement and any other relevant documents filed with the SEC when they become available, because they will contain important information. Investors and security holders may obtain a free copy of the tender offer solicitation / recommendation statement and other documents (when available) that Home Diagnostics files with the SEC at www.sec.gov. In addition, the tender offer solicitation / recommendation statement and other documents filed by Home Diagnostics with the SEC may be obtained from Home Diagnostics free of charge by directing a request to Home Diagnostics at at 2400 N.W. 55th Court, Ft. Lauderdale, FL 33309, attention Peter Ferola, General Counsel |

| Meeting Agenda Home Diagnostics / Nipro transaction profileWhat does this mean for Home Diagnostics and its employees?Nipro corporate and product profile |
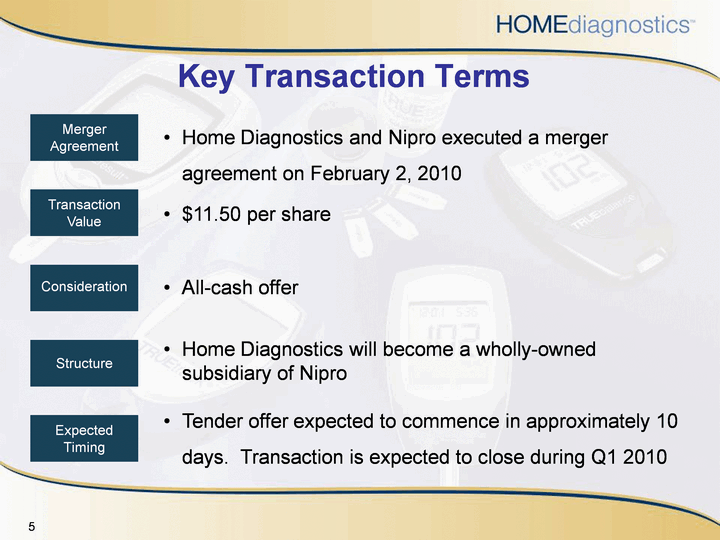
| Key Transaction Terms Home Diagnostics and Nipro executed a merger agreement on February 2, 2010$11.50 per shareAll-cash offerHome Diagnostics will become a wholly-owned subsidiary of NiproTender offer expected to commence in approximately 10 days. Transaction is expected to close during Q1 2010 TransactionValue Consideration Structure ExpectedTiming MergerAgreement |

| Benefits for All Stakeholders Shareholders: Receive a 90% premium over Home Diagnostics previous day's closing price of $6.05Employees: Opportunity to participate as an integral part of growth strategy for company with international platform and distributionCustomers and Patients: Access to a growing product line in the quality-value segment, strengthening our commitment to our customers and the patients they serveVendors and Suppliers: Opportunities for expansion of relationship as Home Diagnostics executes growth strategyStrategic Partners: Opportunity to work with a larger partner with more resources and capabilities |

| Impact on Home Diagnostics Plan is for Home Diagnostics to continue to operate on a standalone basis as a wholly owned subsidiary of Nipro No anticipated changes in operations or business locationsContinued employment of the current HDI work force anticipatedAssumption of role as leader of Nipro's blood glucose monitoring growth strategyAbility to expand distribution capabilities of our products to international markets through Nipro's geographic footprintDiscontinuation of Home Diagnostics' status as a public company and delisting of Home Diagnostics' stock on the NASDAQ Global Select marketAll stock options accelerate and are exchanged for the transaction price, net of the exercise price |

| Impact on Home Diagnostics' Employees Operations should remain substantially unchanged as Home Diagnostics will continue to function as a stand-alone business unitCompany to remain in Florida; Nipro's U.S. and Latin America operations are based in Miami and Nipro's insulin pump subsidiary is located in MiramarSubstantially all functions and locations are expected to continue to remain in operationNo significant reduction in headcount is expected as a result of the transactionOpportunity for career advancement with a company with an international platform and distribution |

| Overview of Nipro Corporation Nipro is highly regarded as a global leader in terms of technological innovation and superior quality in the development of artificial organs, regenerative medicine, generic drug manufacturing and related products.Nipro is recognized as one of the leading manufacturers of dialyzers and dialysis related products, and the Company aims to be the world's leading manufacturer of artificial organs.Nipro also markets an insulin pump for individuals with type one or type two diabetes and other diabetes related products. |

| Nipro Corporate Profile Established: 1954Headquarters: Osaka, JapanEmployees: More than 8,200 globallyStock Exchange: TokyoFY2009 Sales: ¥ 176 billion ($1.8 billion)FY2009 Net Income: ¥ 4.5 billion ($46.1 million)Market Cap: ¥ 122 billion ($1.3 billion)Geographic Presence: Europe, North America, South America and Asia |
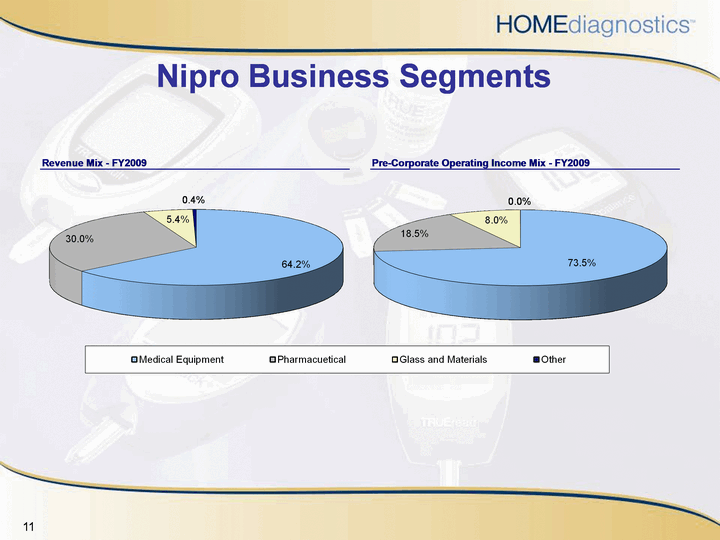
| Nipro Business Segments |

| Nipro Medical Equipment Segment Manufactures and markets medical equipment including:Dialyzers, blood lines for dialysis and dialysis machinesInfusion sets, syringes and needles and a variety of cathetersBlood tests including developments in regenerative medicine and in vitro diagnoticsNipro's U.S. and Latin America operations headquarters are in MiamiNipro's subsidiary, Nipro Diabetes Systems, located in Miramar, FL, markets "The Amigo", an insulin pump for individuals with type one or type two diabetes |

| Nipro Other Business Segments PharmaceuticalsOffers largest contract manufacturing platform in JapanInvolved in the development of generic drugsManufactures a variety of kit products for safety and operability of pharmaceuticalsGlass and MaterialsSells internal glass sections for vacuum flasks, glass tubes for ampoule and vial production, and glass tube vials |
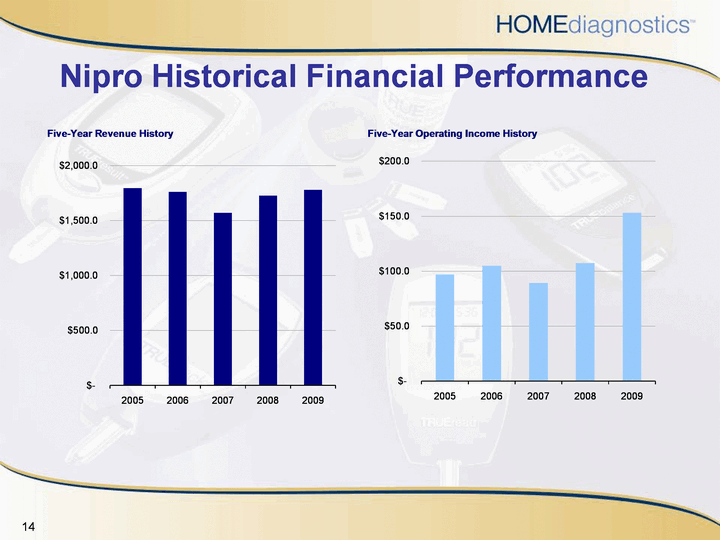
| Nipro Historical Financial Performance |

| Questions |