Exhibit 99.1

| Creating a World-Class Pharmaceuticals and Chemicals Company Roadshow Presentation, March 2006 |

| Disclaimer The information contained in this document with regard to Schering AG has been obtained from publicly available sources only. Neither Merck KGaA nor any of their representatives have independently verified any of the information contained herein. Merck KGaA therefore cannot assume any responsibility with respect to the completeness and the accuracy of this information. This document includes certain statements, estimates and projections with respect to the anticipated future performance of the Schering AG's and Merck KGaA's business and the market for the Schering AG's and Merck KGaA's business' products and services. Such statements, estimates and projections reflect various assumptions made by Merck KGaA concerning anticipated results, which may or may not prove to be correct. No representations are made as to the accuracy of such statements, estimates and projections. The information contained in this document is selective and does not purport to contain all the information the addressee may require. This document should exclusively be viewed together with the oral briefing provided by Merck KGaA . The addressees of this document should conduct their own investigation and analysis of Schering AG's and Merck KGaA's business and of the information contained in this document and are advised to seek their own professional advice on legal, financial and tax aspects. Note regarding forward-looking statements: The information in this document may contain "forward-looking statements". Forward-looking statements may be identified by words such as "expects", "anticipates", "intends", "plans", "believes", "seeks", "estimates", "will" or words of similar meaning and include, but are not limited to, statements about the expected future business of Schering AG resulting from and following the proposed transaction. These statements are based on the current expectations of management of Merck KGaA and are inherently subject to uncertainties and changes in circumstances. Among the factors that could cause actual results to differ materially from those described in the forward-looking statements are factors relating to satisfaction of the conditions to the proposed transaction, and changes in global, political, economic, business, competitive, market and regulatory forces. Merck KGaA do not undertake any obligation to update the forward-looking statements to reflect actual results, or any change in events, conditions, assumptions or other factors. The information in this document is neither an offer to purchase nor a solicitation of an offer to sell securities of Schering AG nor a prospectus. At the time the tender offer is commenced, a tender offer statement will be filed with the U.S. Securities and Exchange Commission (SEC) with respect to the tender offer. Investors and holders of securities of Schering AG are strongly advised to read the tender offer statement and other relevant documents regarding the tender offer filed with the SEC when they become available because they will contain important information. Investors and holders of securities of Schering AG will be able to receive these documents, when they become available, free of charge at the SEC's web site, www.sec.gov. This is not an offer of Merck KGaA securities for sale in the United States. No such securities have been registered under the U.S. Securities Act of 1933, as amended, and no such securities may be offered or sold in the United States absent registration or an exemption from registration. Any public offering of securities to be made in the United States must be made by means of a prospectus that contains detailed information about the issuer and management as well as financial statements. All pharmaceuticals products mentioned in this presentation are registered trademarks. |

| Key Transaction Terms Acquisition of Schering AG for €77 per share in cash Represents a premium of 35% to the three month average share price and 24% to the unaffected share price (€62.12) Total equity value of €14.6bn Takeover offer to acquire all outstanding shares Subject to regulatory approvals Minimum acceptance threshold of 51% of Schering AG's share capital Expected timetable Launch of voluntary tender offer Early April 2006 Expiration of tender offer period Early May 2006 Completion of public tender offer Early June 2006 Capital increase Q3 2006 |

| Transaction Highlights Creating a world-class pharmaceuticals and chemicals company Significant strengthening of Ethicals business built on strong product portfolio, enhanced pipeline and excellent business fit Leading market positions in core businesses (Generics, Liquid Crystals, Life Science & Analytics, Pigments and Diagnostic Imaging) Significant value creation Opportunities for accelerated growth through enhanced global platform in R&D and M&S €500m of annual cost synergies Immediately accretive to adjusted EPS1 before synergies Fully financed transaction Full commitment by Merck Family with equity contribution of €1bn Adjusted EPS before transaction related charges relating to purchase accounting and restructuring costs. |
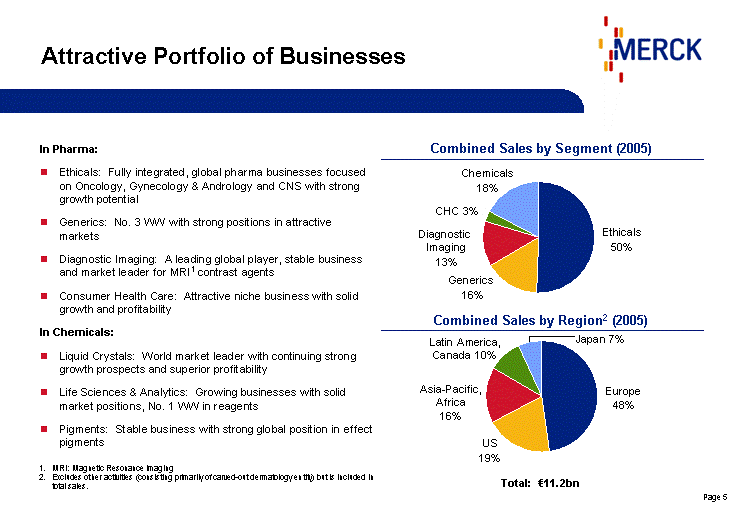
| Europe 47.7 USA 19.5 Asia-Pacific, Afria 16.1 Latin America, Canada 9.6 Japan 7 Ethicals 50 Generics 16 Diagnostics 13 CHC 3 Chemicals 17 Attractive Portfolio of Businesses In Pharma: Ethicals: Fully integrated, global pharma businesses focused on Oncology, Gynecology & Andrology and CNS with strong growth potential Generics: No. 3 WW with strong positions in attractive markets Diagnostic Imaging: A leading global player, stable business and market leader for MRI1 contrast agents Consumer Health Care: Attractive niche business with solid growth and profitability In Chemicals: Liquid Crystals: World market leader with continuing strong growth prospects and superior profitability Life Sciences & Analytics: Growing businesses with solid market positions, No. 1 WW in reagents Pigments: Stable business with strong global position in effect pigments Ethicals 50% Generics 16% Diagnostic Imaging 13% Chemicals 18% CHC 3% Europe 48% US 19% Asia-Pacific, Africa 16% Latin America, Canada 10% Japan 7% Total: €11.2bn MRI: Magnetic Resonance Imaging Excludes other activities (consisting primarily of carved-out dermatology entity) but is included in total sales. Combined Sales by Segment (2005) Combined Sales by Region2 (2005) |

| Larger Scale and Critical Mass in Ethicals Business Larger scale to compete €5.6bn pro-forma Ethicals sales in 2005 (3.3x Merck KGaA Ethicals sales) €1.3bn pro-forma R&D budget, 30 projects in clinical development 9,000+ pro-forma sales force, present in all major regions "Right size" to focus on specialty-type markets Large enough to compete on a global scale Remain entrepreneurial and responsive Target expertise and investments |
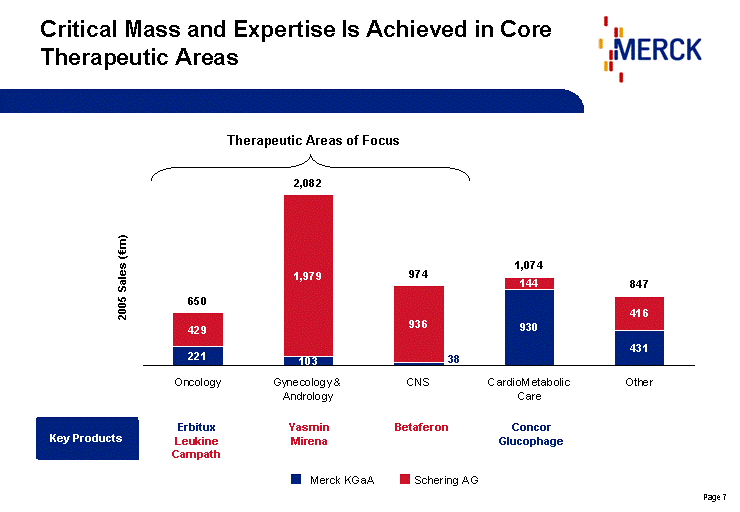
| Key Products Critical Mass and Expertise Is Achieved in Core Therapeutic Areas Oncology Gynecology & Andrology CNS CardioMetabolic Care Other Generics Magnificent 221 103 38 930 431 1597 Superb 429 1979 936 144 416 0 Total 650 2082 974 1074 847 1597 2005 Sales (€m) Erbitux Leukine Campath Yasmin Mirena Betaferon Concor Glucophage Therapeutic Areas of Focus Schering AG Merck KGaA |
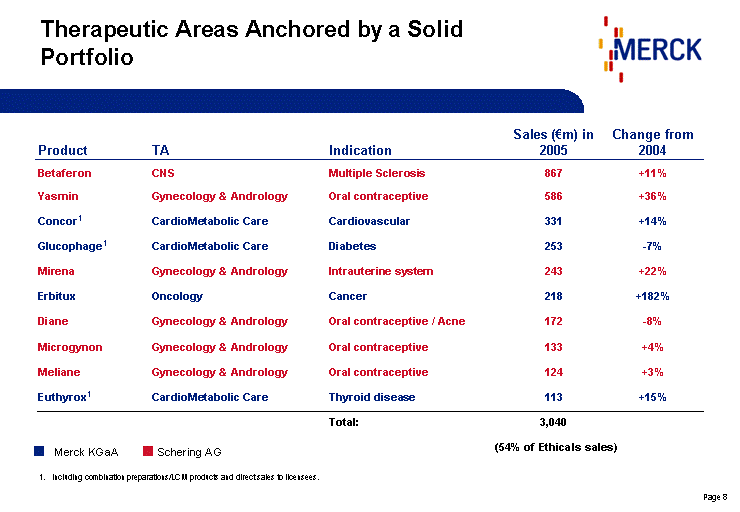
| Therapeutic Areas Anchored by a Solid Portfolio Product TA Indication Sales (€m) in 2005 Change from 2004 Betaferon CNS Multiple Sclerosis 867 +11% Yasmin Gynecology & Andrology Oral contraceptive 586 +36% Concor1 CardioMetabolic Care Cardiovascular 331 +14% Glucophage1 CardioMetabolic Care Diabetes 253 -7% Mirena Gynecology & Andrology Intrauterine system 243 +22% Erbitux Oncology Cancer 218 +182% Diane Gynecology & Andrology Oral contraceptive / Acne 172 -8% Microgynon Gynecology & Andrology Oral contraceptive 133 +4% Meliane Gynecology & Andrology Oral contraceptive 124 +3% Euthyrox1 CardioMetabolic Care Thyroid disease 113 +15% Total: 3,040 (54% of Ethicals sales) Including combination preparations/LCM products and direct sales to licensees. Schering AG Merck KGaA |
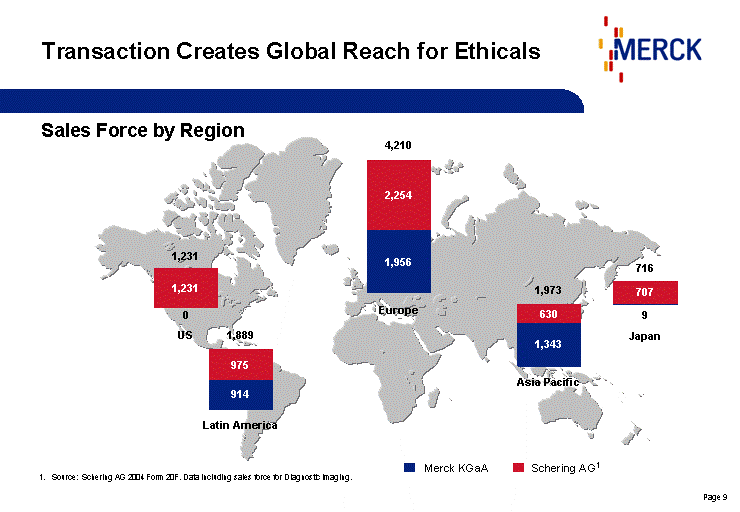
| Transaction Creates Global Reach for Ethicals Schering AG1 Merck KGaA 2,254 1,956 4,210 Europe 630 1,343 1,973 Asia Pacific 707 9 716 Japan 975 914 1,889 Latin America 0 1,231 1,231 US Source: Schering AG 2004 Form 20F. Data including sales force for Diagnostic Imaging. Sales Force by Region |

| R&D Power Greatly Expanded by Larger Investment Capacity More competitive position compared to other mid-size companies Significant benefits of increased scale Higher probability of product approvals Allows acceleration of most promising products Complementary R&D strengths and capabilities Industry-competitive pipeline focused on three therapeutic areas Combined portfolio has a more balanced risk/reward profile Est. No. of Projects % of Sales 2005 estimated R&D budgets (€m) of select mid- size pharmaceuticals companies Eli Lilly Wyeth Bristol Myers Boehringer New Merck Group1 Schering-Plough Takeda Schering AG1 Bayer Novo Nordisk UCB Merck KGaA Organon Forest Shire a 2436 2059 1818 1320 1127 1059 743 682 483 434 236 225 b 1302 c 860 d 442 19.3% 23.1% 18.5% 14.8% 22.0% 19.0% 15.1% 21.1% 25.7% 17.9% 9.4% 17.8% 12 30 20 18 19 22 17 10 11 13 11 7 14.8% 26 17.9% 39 22.0% 29 Excluding Diagnostic Imaging. Source: Wall Street research, public filings and company websites |
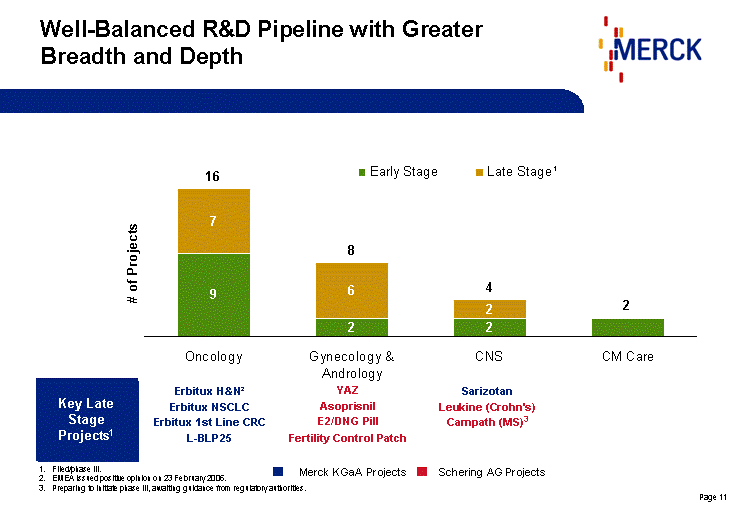
| Well-Balanced R&D Pipeline with Greater Breadth and Depth Oncology Gynecology & Andrology CNS CM Care Early Stage 9 2 2 2 Late Stage 7 6 2 0 Magnificent Product Superb Product Total 16 8 4 2 Key Late Stage Products1: Erbitux H&N Erbitux NSCLC Erbitux 1st Line CRC L-BLP25 Vatalanib Sarizotan Leukine (Crohn's) YAZ Asoprisnil E2/DNG Pill Fertility Control Patch Oncology Gynecology & Andrology CNS Cardio Metabolics Other Generics Magnificent Product 0 0 0 0 0 0 Superb Product 0 0 0 0 0 0 Total 0 0 0 0 0 0 Key Late Stage Projects1 Oncology Gynecology & Andrology CNS CM Care Early Stage 9 2 2 2 Late Stage1 7 6 2 0 Magnificent Product Superb Product Total 16 8 4 2 Erbitux H&N2 Erbitux NSCLC Erbitux 1st Line CRC L-BLP25 YAZ Asoprisnil E2/DNG Pill Fertility Control Patch Sarizotan Leukine (Crohn's) Campath (MS)3 Filed/phase III. EMEA issued positive opinion on 23 February 2006. Preparing to initiate phase III, awaiting guidance from regulatory authorities. Schering AG Projects Merck KGaA Projects |
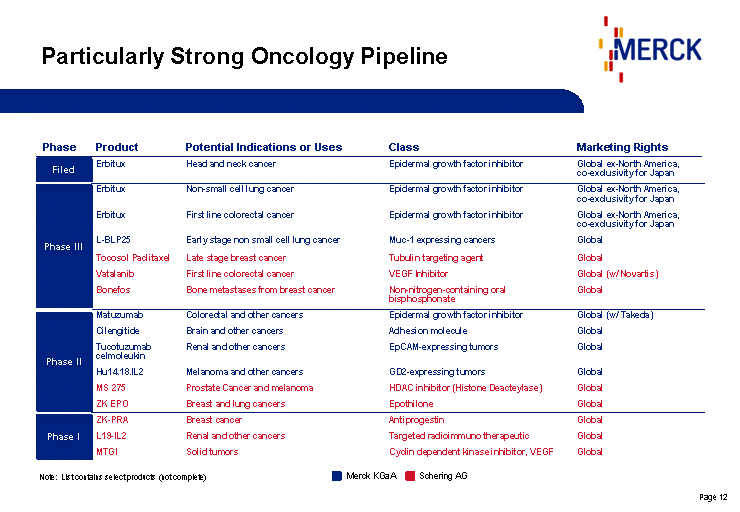
| Particularly Strong Oncology Pipeline Phase Product Potential Indications or Uses Class Marketing Rights Erbitux Head and neck cancer Epidermal growth factor inhibitor Global ex-North America, co-exclusivity for Japan Erbitux Non-small cell lung cancer Epidermal growth factor inhibitor Global ex-North America, co-exclusivity for Japan Erbitux First line colorectal cancer Epidermal growth factor inhibitor Global ex-North America, co-exclusivity for Japan L-BLP25 Early stage non small cell lung cancer Muc-1 expressing cancers Global Tocosol Paclitaxel Late stage breast cancer Tubulin targeting agent Global Vatalanib First line colorectal cancer VEGF Inhibitor Global (w/ Novartis) Bonefos Bone metastases from breast cancer Non-nitrogen-containing oral bisphosphonate Global Matuzumab Colorectal and other cancers Epidermal growth factor inhibitor Global (w/ Takeda) Cilengitide Brain and other cancers Adhesion molecule Global Tucotuzumab celmoleukin Renal and other cancers EpCAM-expressing tumors Global Hu14.18.IL2 Melanoma and other cancers GD2-expressing tumors Global MS 275 Prostate Cancer and melanoma HDAC inhibitor (Histone Deacteylase) Global ZK EPO Breast and lung cancers Epothilone Global ZK-PRA Breast cancer Antiprogestin Global L19-IL2 Renal and other cancers Targeted radioimmuno therapeutic Global MTGI Solid tumors Cyclin dependent kinase inhibitor, VEGF Global Note: List contains select products (not complete) Schering AG Merck KGaA Filed Phase III Phase II Phase I |

| Enhanced Global Platform Creates Opportunities for Accelerated Growth The potential launch success of Merck KGaA's Sarizotan will be accelerated by Schering AG's experience in CNS New indications for Merck KGaA's Erbitux will benefit from added Schering AG promotional support Attractive global development and commercialization partner to compete for licensing opportunities Full value from every R&D Euro - ability to retain global rights and launch in all major markets Creating access for Merck KGaA to two large markets, North America and Japan Strengthening presence in Europe, South America and Asia-Pacific |

| Sizable Cost Savings €500m in annual cost savings expected from integration of the two companies Annual savings represent 8% of the relevant cost base1 Need to retain R&D scale of both companies to be competitive Limited synergies in North America and Japan Therapeutic Area overlap in Oncology only, which is viewed as attractive Management anticipates that full synergies will be reached in 2009 Note: Full integration requires implementation of a merger or a domination agreement. Schering AG total cost + Merck KGaA Ethicals cost + Merck KGaA corporate cost |

| Manageable Integration German combination Complementary nature of businesses Little-to-no integration required in North America and Japan Highly complementary therapeutic areas with little overlap Integration strategy to be completed together with Schering AG1 Implementation of integration is subject to a Domination Agreement being in place. |

| New Merck Group Will Have Strong Financials Merck KGaA Schering AG New Merck Group1 Total Sales (2005) €5.9bn €5.3bn €11.2bn Pharma as % of Total2 60% 100% 79% Ethicals as % of Total 29% 74% 50% Operating Income (2005) €0.9bn €0.9bn €1.8bn Operating Margin (%) 15.0% 17.5% 16.2% Pharma Margin (%) 11.5% 17.5% 15.1% Chemicals Margin (%) 25.9% NA 25.9% Net Income (2005) €0.6bn €0.6bn €0.9bn3 Earnings per Share (diluted) €3.00 €3.26 €3.453 Sources of Growth LC, Oncology Gynecology & Andrology LC, Oncology, G&A Pro-forma combined before synergies Pharma including total Schering AG plus Merck KGaA Pharma (Ethicals, Generics and CHC). Before transaction related charges relating to purchase accounting and restructuring costs, 100% take up in tender offer and based on post refinancing capital structure. EPS assumes capital increase at current market price. [PF Net Income = 572.1+619 - post-tax interest for full year on acquisition debt net of cap increase] [PF adj. EPS = PF Net Income / (190.8+5.25/83.50) |
| Financing of Transaction Tender offer initially funded through existing Merck KGaA cash and bridge financing provided by Bear Stearns, Deutsche Bank and Goldman Sachs All-cash transaction will be refinanced through a combination of existing cash, a syndicated loan, a bond, a hybrid instrument and a capital increase Merck KGaA is committed to retain investment-grade rating Rapid de-leveraging driven by strong operating cash flow Equity financing through capital increase post completion of tender offer (expected Q3 2006) Rights Issue to public shareholders of €0.5bn to €4.0bn based on 51% to 100% take-up Merck Family is committed to invest €1bn |

| Financial Guidance for 2006 Sales: Single digit growth Adjusted EBIT1: Double digit growth Adjusted EPS1: Accretive in first year Free Cash Flow: Strong Adjusted EBIT and EPS before transaction related charges relating to purchase accounting and restructuring costs. |

| In Conclusion.... Creating a world-class pharmaceuticals and chemicals company Significant strengthening of Ethicals business built on a strong product portfolio, enhanced pipeline and excellent business fit Leading market positions in core businesses (Generics, Liquid Crystals, Life Science & Analytics, Pigments and Diagnostic Imaging) Significant value creation Opportunities for accelerated growth through enhanced global platform in R&D and M&S €500m of annual cost synergies Immediately accretive to adjusted EPS1 before synergies Fully financed transaction Full commitment by Merck Family with equity contribution of €1bn Adjusted EPS excludes transaction related amortization and one-time restructuring costs. |

| Your Contact Sascha Becker Head of Investor Relations +49 6151 72-3706 Dr. Monika Buttkereit Manager Investor Relations +49 6151 72-2584 Susanne Zeichner Assistant Investor Relations +49 6151 72-3315 Merck KGaA Investor Relations Frankfurter Str. 250 64271 Darmstadt Germany Fax: +49 6151 72-913321 investor.relations@merck.de www.investors.merck.de |

| Creating a World-Class Pharmaceuticals and Chemicals Company |