UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
ANNUAL REPORT
PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended December 31, 2007
Commission File Number: 0-27422
ARTHROCARE CORPORATION
(Exact name of Registrant as specified in its charter)
Delaware | 94-3180312 |
(State or other jurisdiction of Incorporation or organization) | (I.R.S. employer Identification number) |
7500 Rialto Blvd., Building Two, Suite 100, Austin, TX 78735
(Address of principal executive offices and zip code)
(512) 391-3900
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12 (b) of the Act: | Common Stock, $0.001 Par Value; Preferred Share Purchase Rights | |
Securities registered pursuant to Section 12 (g) of the Act: | None | |
Indicate by check mark if the Registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes x No ¨
Indicate by check mark if the Registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the Registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of Registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the Registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer (as defined in Rule 12b-2 of the Act). Large accelerated filer x Accelerated filer ¨ Non-accelerated filer ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No x
As of June 30, 2007, the aggregate market value of the voting and non-voting common equity held by non-affiliates of the Registrant was approximately $1,104,895,343 (based upon the closing sales price of such stock as reported by The NASDAQ Stock Exchange on such date). Shares of Common Stock held by each officer, director and holder of 5 percent or more of the outstanding Common Stock on that date have been excluded in that such persons may be deemed to be affiliates. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of February 19, 2008, the number of outstanding shares of the Registrant’s Common Stock was 26,585,219.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the definitive proxy statement for our 2007 annual meeting of stockholders, which we expect to file with the Commission within 120 days after December 31, 2007, are incorporated by reference into Part III of this Annual Report.
ARTHROCARE CORPORATION
FORM 10-K ANNUAL REPORT
TABLE OF CONTENTS
Page | |||
PART I. | |||
| ITEM 1. | Business | 3 | |
| ITEM 1A. | Risk Factors | 13 | |
| ITEM 1B. | Unresolved Staff Comments | 20 | |
| ITEM 2. | Properties | 20 | |
| ITEM 3. | Legal Proceedings | 21 | |
| ITEM 4. | Submission of Matters to a Vote of Security Holders | 21 | |
PART II. | |||
| ITEM 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | 21 | |
| ITEM 6. | Selected Consolidated Financial Data | 23 | |
| ITEM 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 24 | |
| ITEM 7A. | Quantitative and Qualitative Disclosures about Market Risk | 35 | |
| ITEM 8. | Financial Statements and Supplementary Data | 35 | |
| ITEM 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 35 | |
| ITEM 9A. | Controls and Procedures | 36 | |
| ITEM 9B. | Other Information | 36 | |
PART III. | |||
| ITEM 10. | Directors, Executive Officers and Corporate Governance | 37 | |
| ITEM 11. | Executive Compensation | 37 | |
| ITEM 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 37 | |
| ITEM 13. | Certain Relationships and Related Transactions, and Director Independence | 40 | |
| ITEM 14. | Principal Accountant Fees and Services | 40 | |
PART IV. | |||
| ITEM 15. | Exhibits and Financial Statement Schedules | 40 |
2
PART I
This Report on Form 10-K contains certain forward-looking statements regarding future events and our future operating results that are subject to the safe harbors created under the Securities Act of 1933 (the “Securities Act”) and the Securities Exchange Act of 1934 (the “Exchange Act”) with respect to ArthroCare Corporation (“ArthroCare,” “we,” “us,” “our,” and “Company” refer to ArthroCare Corporation, a Delaware corporation, unless the context otherwise requires). Readers are cautioned that these forward-looking statements are only predictions and are subject to risks, uncertainties, and assumptions that are difficult to predict, including those identified below, under “Risk Factors”. Actual events or results could differ materially due to a number of factors, including those described herein and in the documents incorporated herein by reference.
Overview
We are a multi-business medical device company that develops, manufactures and markets minimally invasive surgical products, many of which are based on our patented Coblation® technology. We have grown well beyond our roots in arthroscopy to capitalize on numerous market opportunities across several medical specialties, improving many existing soft-tissue surgical procedures and enabling new minimally invasive procedures. With our innovative technologies, we are committed to improving the lives of individuals suffering from conditions as diverse as torn rotator cuffs and anterior cruciate ligaments, or ACLs, to herniated discs and enlarged tonsils/tonsillitis.
We currently market minimally invasive surgical products across three core business units—ArthroCare Sports Medicine, ArthroCare Spine and ArthroCare Ear, Nose and Throat, or ENT — but also have developed, manufactured and marketed Coblation-based and complementary products for application in neurology, cosmetic surgery, urology and gynecology, with research continuing in additional areas. In each of our core business units, we are focused on driving the application of enabling technologies, primarily for plasma-based soft tissue removal, and increasing the number of minimally invasive procedures being performed.
We focus on executing a mission-driven business strategy featuring the following key elements:
| · | Expanding our product offering to address large and rapidly growing markets; |
| · | Targeting established procedures and replacing current technology with value-added ArthroCare technologies; |
| · | Driving disposable device sales with a direct sales force; |
| · | Augmenting growth with complementary and compatible acquisitions to expand margins and provide additional opportunity to capitalize on emerging and existing business opportunities; and |
| · | Establishing strategic partnerships to further commercialize our minimally invasive technologies. |
ArthroCare was incorporated in California in 1993 and reincorporated in Delaware in 1995. We maintain an internet website at http://www.arthrocare.com. On our website we make available, free of charge, the following filings as soon as reasonably practicable after they are electronically filed with or furnished to the Securities and Exchange Commission, or SEC: our annual reports on Form 10-K, our quarterly reports on Form 10-Q, our current reports on Form 8-K and any amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act of 1934. You may also read and copy any materials that we file with the SEC at the SEC’s Public Reference Room at 100 F Street, NE, Washington DC 20549. You may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. Our reports, proxy statements and other documents filed electronically with the SEC are available at the website maintained by the SEC at http://www.sec.gov.
3
Coblation Technology
Many of our minimally invasive products are based on ArthroCare’s patented Coblation technology, which uses radiofrequency energy to create a precisely focused plasma to ablate soft tissue. The use of Coblation allows surgeons to operate with a high level of precision and accuracy, limiting damage to surrounding healthy tissue and thereby potentially reducing patient pain and recovery times. Our Coblation-based products operate at lower temperatures than traditional electrosurgical or laser surgery tools that use heat to burn away targeted tissue, which often results in thermal damage to tissue surrounding the surgical area. Our Coblation-based systems consist of a controller unit and an assortment of sterile, single-use disposable devices that are specialized for specific types of procedures. We believe our Coblation technology can replace the multiple surgical tools traditionally used in soft-tissue surgery procedures with one multi-purpose surgical system.
We have applied Coblation technology for soft-tissue surgery throughout the body, primarily in the areas of arthroscopy/sports medicine, spinal surgery and ENT applications. We also are exploring the use of Coblation technology in other markets, such as neurosurgery, cosmetic surgery, urology, gynecology, cardiac surgery and general surgical procedures.
The Sports Medicine Market
Overview
Due to patient demand for less invasive procedures, we believe the number of arthroscopic procedures is growing. In addition, a greater emphasis on physical fitness and an aging population are increasing the incidence of joint and soft tissue injuries. Joints are susceptible to injuries from blows, falls or twisting, as well as from natural degeneration and stiffening associated with aging.
Historically, joint injuries have been treated using open surgery involving large incisions, a hospital stay and a prolonged recovery period. In contrast, arthroscopic surgery is performed through several small incisions, called portals, and can be performed on an outpatient basis. We believe arthroscopic surgery has gained widespread market acceptance because it can offer shorter hospital stays and reduced recovery time, which may result in reduced costs and improved medical outcomes.
The advantages of arthroscopic surgery over open surgery can be significant. Due to the smaller incisions and reduced surgical trauma, the patient might experience several benefits, including reduced pain, treatment on an outpatient basis, reduced hospitalization times, smaller scars, immediate joint mobility, less muscle atrophy, less surrounding tissue damage, a lower rate of complication and generally quicker rehabilitation.
Our Solution to Arthroscopic Surgery
Our Arthroscopic System is used to treat a number of orthopedic conditions, including those involving knees, ankles, elbows, wrists, tendons and hips. Our Arthroscopic System is comprised of an assortment of disposable bipolar multi-electrode and single electrode devices, a connecting cable, foot pedal or hand switch and a radiofrequency controller. Our controller delivers radiofrequency energy to the disposable surgical device without the need for a ground or “return” pad, which is required for conventional monopolar electrosurgical systems. A surgeon can use the disposable device for ablation, resection, coagulation of soft tissue and to seal bleeding vessels. The incorporation of ablation, suction and fluid management into a single disposable device potentially reduces operating time and expense.
Complementary Enabling Sports Medicine Technologies
The Opus Collection
In November of 2004, we acquired Opus Medical, Inc., a manufacturer of products designed to facilitate the arthroscopic repair, including the Opus Autocuff System, for knotless repair of the rotator cuff. The Opus AutoCuff System addresses each of the requirements for a total arthroscopic repair through its proprietary family of specialized instruments and devices. These products may enhance efficacy, decrease complications, reduce costs, reduce procedure time and broaden the treatable patient base. In 2005, we introduced a sequel device to the AutoCuff Anchoring System known as the LabraFix System. The LabraFix System provides knotless arthroscopic repair of the labrum.
4
The Atlantech Collection
In October and November of 2002, we acquired two of our European distributors: Atlantech Medical Devices, Ltd., a distributor of ArthroCare products in the United Kingdom, and Atlantech AG, our German distributor. The acquisitions accelerated the implementation of our direct sales strategy in Europe, providing us with an immediate direct sales force in two key markets. We also obtained a complementary line of sports medicine products through the purchase of Atlantech Medical Devices. This product line consists of more than 1,500 individual arthroscopic surgical items.
Distribution for Sports Medicine Products
We sell our Arthroscopic System through our Sports Medicine business unit by means of a network of direct sales representatives and independent orthopedic distributors supported by regional managers. Additionally, we offer certain of our customers the ability to draw on our expertise in billing private third-party insurance companies and other payers for devices that qualify for reimbursement separate from a global fee by assigning the right to bill for such devices to us. This allows facilities that do not have sophisticated reimbursement processes to focus their efforts on providing patient care. Upon reimbursement from the third-party payers, the customer is rebated the reimbursement amount, up to the price they paid for the device.
Competition in the Sports Medicine Market
We compete directly with the providers of tissue removal systems, including conventional electrosurgical systems, manual instruments, power shavers and laser systems. Smith & Nephew Endoscopy, which owns Acufex Microsurgical, Inc., Dyonics, Inc. and Oratec Interventions, Inc., Conmed Corporation, including its Linvatec unit, Arthrex and Stryker Corporation each have large shares of the market for manual instruments, power shavers and arthroscopes.
Johnson & Johnson, including DePuy Mitek, a division of its Ethicon, Inc. unit, Stryker Corporation and Smith & Nephew market bipolar electrosurgical systems that compete directly with our tissue ablation and coagulation technology in Sports Medicine. In addition, the Linvatec unit of Conmed Corporation, Smith & Nephew and Arthrex are marketing monopolar electrosurgical tools for tissue ablation that compete with our products in this field. These same competitors also compete against our Opus collection with rotator cuff repair systems that include suture passers and bone anchors to complete the repair.
The Spinal Surgery Market
Overview
We believe the spine surgery market represents the most rapidly growing segment of the orthopedic surgery market. A high percentage of back pain can be traced to problems associated with intervertebral discs. Intervertebral discs mainly function to cushion and tether the vertebrae, providing flexibility and stability to the patient’s spine. Problems that can occur with intervertebral discs include degeneration and herniation. With degeneration, discs lose their water content and height, bringing the adjoining vertebrae closer together. This results in a weakening of the shock absorption properties of the disc and a narrowing of the nerve openings in the sides of the spine, which may pinch these nerves. With herniation, the outer layer of the disc, which is called the annulus, bulges or ruptures. Disc herniation can eventually cause back pain and pain that radiates to the extremities, including the buttocks, legs, shoulders and arms.
In some cases, disc herniation results in intractable pain, thereby necessitating a microdiscectomy, the removal of a portion of the disc to eliminate the source of inflammation and pressure. In more severe cases, the adjacent vertebral bodies must be stabilized following excision of the disc material to avoid recurrence of the disabling back pain. One approach to stabilizing the vertebrae, termed spinal fusion, is to insert an interbody graft or implant into the space vacated by the degenerative disc. In this procedure, a small amount of bone may be grafted from other portions of the body, such as the hip, and packed into the implants. This process allows the bone to grow through and around the implant, fusing the vertebral bodies, thereby alleviating the pain. More recently, the implantation of an artificial disc, termed disc arthroplasty, has become more popular primarily due to the potential for these artificial discs to preserve the natural motion of the spine and lead to faster recovery than fusion procedures.
5
Our Solution to Spinal Surgery
Disc Decompression
Randomized controlled studies have indicated minimally invasive disc decompression is an effective alternative to microdiscectomy for patients with contained herniated discs who have symptoms of back or radicular pain. Disc decompression is a minimally invasive procedure where disc material is removed without the patient having to undergo major open surgery. The disc is accessed through a narrow cannula and the physician performs the procedure under fluoroscopic (live x-ray) guidance. We created the DLR, DLG and DC SpineWand® Convenience Packs for minimally invasive disc decompression. Plasma Disc Decompression, a minimally invasive disc decompression procedure using our Coblation technology to treat symptomatic patients with contained herniated discs, employs the process of ablation of soft tissue for partial removal of the nucleus of a disc. Plasma Disc Decompression can be performed in both the lumbar and cervical spine either percutaneously or through a small incision with or without an access port. Plasma Disc Decompression can be performed as a stand alone procedure or in conjunction with other surgical procedures such as a spinal fusion.
Microdisectomy
In cases where the surgeon determines that a microdiscectomy is indicated, the surgeon requires a device that can remove a greater amount of tissue than the devices we market for Plasma Disc Decompression. For microdiscectomy cases, we market the MD SpineWand surgical device. The MD SpineWand, which is also based on our Coblation technology, is designed to quickly and efficiently remove disc material through an annulotomy that is typically much smaller than the one required with the mechanical instruments that are used in these procedures.
Complementary Enabling Spinal Surgery Technologies
Vertebral Compression Fractures
In January 2004, we completed the acquisition of Medical Device Alliance, Inc. and its majority-owned subsidiary, Parallax Medical, a leader in products for the minimally invasive treatment of vertebral compression fractures. It is estimated that 750,000 Americans suffer from vertebral compression fractures which are caused by osteoporosis, trauma or, increasingly, metastatic disease. Approximately 260,000 of these patients are treated annually. Parallax’s product line includes vertebral access and bone biopsy needles, cement delivery systems, bone cement and opacifiers and ancillary devices permitting minimally invasive repair of vertebral compression fractures.
In May of 2007 ArthroCare received clearance from the Food and Drug Administration, or FDA, to market the Cavity SpineWand, a Coblation-based device, to reduce malignant lesions within a vertebra in patients suffering from compression fractures secondary to tumors. Medical grade cement is then injected to stabilize the fracture.
Distribution for Spinal Surgery Products
We sell our spinal surgery products through our Spine business unit by means of a network of direct sales representatives and independent orthopedic distributors supported by regional managers. Additionally, prior to December 31, 2007 a third-party billing and reimbursement service provider known as DiscoCare, Inc. would purchase devices from ArthroCare and provide them to customers generally at no charge in return for either the agreement by the facility to assign the right to bill private third-party insurance companies and other payers for the device or a letter of protection in a personal injury case. More specifically, a third-party billing and reimbursement service provider, such as DiscoCare, deals with insurance pre-authorizations, device procurement, inventory management and claims processing to help reduce device costs and the financial collection risk for facilities who use the service in conjunction with a procedure. Many of ArthroCare’s spinal surgery products are leading edge technologies which have not yet received routine coverage decisions from insurance companies and other payers making reimbursement dependent on case by case decisions from various third-party payors. In part because of DiscoCare’s history of success in demonstrating the efficacy of the procedures using the ArthroCare devices and their ability to receive reimbursement for such devices, on December 31, 2007, ArthroCare acquired DiscoCare to augment our internal reimbursement resources. Utilizing DiscoCare’s services allows facilities who do not have sophisticated reimbursement processes to focus their efforts on patient care.
6
Competition in the Spinal Surgery Market
In the Plasma Disc Decompression market, we directly compete with Stryker, Clarus, Smith and Nephew and other small companies. We may indirectly compete with large companies in the spine fusion and discectomy markets such as DePuy Spine, Medtronic Sofamor Danek, Stryker, Zimmer and Synthes. In addition, we are aware of several small companies offering alternative treatments for back pain that may indirectly compete with our products. For example, Smith & Nephew Endoscopy and Radionics, a division of Integra, manufacture and sell catheters that use resistive heating to reduce chronic low back pain caused by degenerative disc disease.
In the vertebral compression fracture market, the primary direct competitors in the vertebroplasty market are Stryker, Cardinal Health/Allegiance and Cook Medical. Kyphon, which was recently acquired by Medtronic, is our leading competitor in the overall vertebral compression fracture market with its balloon kyphoplasty procedure.
The ENT Market
Overview
The most common ENT procedures are placement of ear tubes, and the removal of tonsils and adenoids. We estimate that there are more than 600,000 tonsillectomies performed in the United States each year, many of which are accompanied by an adenoidectomy. The majority of tonsillectomies are performed on pediatric patients, which results in these procedures typically being performed more often in the late spring and winter months of the year. Other commonly performed ENT procedures include endoscopic sinus surgery, septoplasty, turbinate reduction and procedures to remove or stiffen tissue to treat obstructive sleep apnea and snoring. Highly specialized ENT surgeons, typically in an outpatient or ambulatory surgery center, perform these procedures. For decades, monopolar electrosurgical instruments (e.g. the bovie) have been the standard for removal of soft tissue and cauterization in ENT procedures. As in other surgical procedures, the high levels of heat associated with bovies often results in significant post-operative pain and extended recovery periods.
Our Solution to ENT Surgery
We market ENT products through a network of direct sales representatives and independent distributors supported by sales managers for use in general head, neck and oral surgical procedures, including sinus surgery, the treatment of snoring, reduction of nasal turbinates, adenoidectomy and tonsillectomy. We have three categories of disposable devices, or wands, to address the ENT Market—suction wands, channeling wands and excision wands. Suction wands simultaneously ablate and remove tissue in applications such as tonsillectomy, providing enhanced visibility. Channeling wands combine controlled ablation and effective coagulative lesion formation in applications such as turbinate reduction to relieve nasal obstruction and stiffening of the soft palate for the treatment of snoring. Excision wands provide precise dissection of soft tissue with minimal damage to surrounding tissue.
Complementary Enabling ENT Technologies
The Applied Therapeutics Collection
In August 2005, we completed the purchase of substantially all of the assets of Applied Therapeutics, Inc., or ATI, a maker of sinus surgery treatment products. ATI’s carboxymethyl-cellulose based packing and tamponade products promote platelet aggregation directly at the wound site while remaining moist for the duration of insertion. We believe this minimizes patient discomfort and re-bleeding during removal, as compared to traditional treatments. The ATI collection includes the dissolvable Stammberger Sinus Dressing and dissolvable Sinu Knit. These products are used after endoscopic sinus surgery to control minor bleeding, facilitate epithelial healing and preserve the spatial integrity of the sinus.
Competition in the ENT Market
There are large companies, such as Gyrus and Medtronic, which have shares of the market for manual and powered instruments for ENT, head and neck surgical procedures. Gyrus manufactures and sells medical devices that utilize radio frequency energy for the treatment of upper airway disorders, such as turbinate reduction, snoring and obstructive sleep apnea. In addition, Gyrus has introduced a radiofrequency system for treating tonsils that competes directly with some of our products in the ENT business.
7
Additional Markets for Coblation Technology
We developed and commercialized Coblation-based urology products through a strategic partnership with ACMI until the termination of that agreement in August 2005 following Gyrus Medical’s acquisition of ACMI. We also developed gynecology products for commercialization through an agreement with GyneCare, a unit of Johnson & Johnson, until the expiration of that agreement in September 2004. We may establish strategic partnerships in these markets and others, where beneficial, in the future or enter the market directly, as appropriate.
Potential Competition in Non Core-Coblation Markets
We believe Coblation-based technologies will compete effectively against a variety of technologies used in gynecology, urology, laparoscopic and cardiology procedures. There are several large companies, such as Ethicon Endo-Surgery, a division of Johnson & Johnson; Valleylab, a division of U.S. Surgical; and Gyrus and Olympus Medical Systems Group, which have shares of the gynecology market for mechanical, ultrasonic, monopolar and bipolar instruments. In the field of urology, we face competition from companies that market monopolar, bipolar, mechanical, and ultrasonic and laser devices for a variety of urological procedures, including transurethral prostatectomy, or TURP, and transurethral incisions in the prostate, or TUIP. These companies include Karl Storz, Olympus Medical Systems Group, C.R. Bard, Gyrus, Medtronic, Urologix and Laserscope. We expect competition from these and other well-established competitors will increase as will competition from smaller medical device companies in both the field of gynecology and urology. Several large companies, including Edwards Lifescience, Medtronic, Guidant, Johnson & Johnson and St. Jude Medical, dominate cardiology. These companies, including several smaller companies, offer mechanical, powered, laser and electrosurgical systems.
Research and Development
We have focused our research and development efforts in four areas. First, in response to physician feedback, we modify and enhance the performance of our generator/controller platforms and introduce new platforms from time to time. Second, we have continued to design new disposable devices that incorporate added functionalities for faster and easier use. Third, we have developed and continue to explore new applications of our Coblation technology in surgical markets. Fourth, we incorporate complementary technologies and products into our portfolio in order to provide more value to our customers and participate to a greater extent in certain surgical procedures. Research and development expenses were $26.9 million in 2007, $23.2 million in 2006, and $21.0 million in 2005.
We also have undertaken preliminary studies and development for the use of our technology in several new fields. To this end, we continue to explore and develop the plasma physics underlying Coblation technology. We are engaged with a number of doctors, scientists and research institutions to further understand the technology and its additional applications and other technical improvements.
Manufacturing
Our disposable devices are primarily manufactured at our facility located in an industrial park in San Jose, Costa Rica. In 2004, we transferred the manufacturing of our controllers from our Sunnyvale, California facility to our Costa Rica facility. As of December 31, 2005, our Costa Rica facility was also producing essentially all of the disposable products associated with the Opus product line.
In 2003, we transformed our primary Sunnyvale facility from a general manufacturing facility to a facility with an increased focus on research, development and prototype manufacturing. In 2005, the lease for this facility was extended through February 2014. Approximately 50 percent of this facility is devoted to research and new product development. We believe our existing operations will provide adequate capacity for our manufacturing needs at least through 2008.
We have established quality assurance systems in conformance with the FDA’s Quality System Regulation, or QSR. Our facilities in Sunnyvale and Costa Rica have received ISO 13485 and CMDCAS certification and are in conformance with applicable regulatory requirements.
Our products are manufactured from several components, most of which are supplied to us from third parties. Most of the components we use in the manufacture of our products are available from more than one qualified supplier. For some components, however, there are relatively few alternative sources of supply and the establishment of additional or replacement suppliers may not be accomplished quickly. In isolated cases, we rely upon single source suppliers. We also use a single subcontractor to sterilize our disposable devices, but do not believe a major disruption is likely because the supplier has multiple sterilization facilities throughout the United States as well as internationally, and there are competing sterilization companies that offer similar services. Our Atlantech product line of handheld instruments is primarily single sourced. Disruption of this supply source would result in a material disruption of our ability to sell this line of products for an extended time period.
8
DHL Worldwide Express, Inc., or DHL, is our primary provider of logistic services, including warehousing of finished goods, shipment of finished products to customers and field returns, along with management of a portion of our in-bound freight requirements. We have experienced favorable cost performance and have gained an ability to scale our business at a faster pace due to this relationship. However, the complex nature of the underlying support technologies, such as “Electronic Data Interchange,” has, at times, caused disruptions in our ability to deliver products to customers on a timely basis. Failure of these supporting systems could impair our business for the duration of the failure.
Marketing and Sales
We are marketing and selling our arthroscopic/sports medicine, spinal surgery and ENT products using a combination of distributors supported by regional sales managers, a direct sales force and corporate partners to sell our products both domestically and internationally. In the U.S. and Europe, we principally use distributors and field sales representatives to sell our products.
Outside of the United States and Europe, we have established distribution capability in certain countries by means of exclusive and non-exclusive distribution agreements for the distribution of our arthroscopy, spinal surgery and ENT surgery products in Japan. We have also established distribution capability through relationships with distributors in New Zealand, Russia, China, Korea, Taiwan, Canada, Mexico, the Caribbean, North Africa, South Africa, the Middle East, South America and Central America. During the years ended December 31, 2007, 2006 and 2005, approximately 21 percent, 20 percent and 21 percent, respectively, of our product sales were derived internationally. For information regarding product sales in certain product markets and geographic areas, see Note 15, “Segment Information,” in the notes to the consolidated financial statements in this Form 10-K.
Reimbursement Assistance
Many of ArthroCare’s products are leading edge technologies which have not yet received routine coverage decisions from insurance companies and other payers. As a result, reimbursement may be dependent on case by case decisions from various private third-party payers. In 2007, we established a new subsidiary, Device Reimbursement Services (“DRS”), to provide principally our Sports Medicine devices that we sell in exchange for an assignment of the right to bill for such devices. DRS offers certain of our customers the ability to draw on our expertise in billing private third-party insurance companies and other payers for devices that qualify for reimbursement separate from a global fee by assigning the right to bill for such devices to us. Utilizing DRS’ services allows facilities that do not have sophisticated reimbursement processes to focus their efforts on providing patient care. Upon reimbursement from the third-party payers, the customer is typically rebated the reimbursement amount, up to the price they paid for the device.
Since 2006, DiscoCare, Inc., another third-party billing and reimbursement service provider, has purchased devices from ArthroCare and provided them to their customers generally at no charge in return for either the agreement by the facility to assign to DiscoCare the right to bill private third-party insurance companies and other payers for the device or a letter of protection in a personal injury case. After a procedure is completed, DiscoCare bills either the applicable insurer or other third-party payer. More specifically, a third-party billing and reimbursement service provider, such as DiscoCare, deals with insurance pre-authorizations, device procurement, inventory management and claims processing to help reduce device costs and the financial collection risk for facilities who use the service in conjunction with a procedure. DiscoCare originally worked with ArthroCare on a limited geographical basis and, in the fourth quarter of 2006, DiscoCare approached ArthroCare about providing their services to ArthroCare customers on a national basis. The resulting agreement provided for DiscoCare to purchase the devices and provide the billing and reimbursement services to ArthroCare’s customers and receive a service fee paid by ArthroCare. Many of the ArthroCare products purchased by DiscoCare consisted of leading edge technologies which have not yet received routine coverage decisions from insurance companies and other payers making reimbursement dependent on case by case decisions from various third-party payers. In part because of DiscoCare’s history of success in demonstrating the efficacy of the procedures using the ArthroCare devices and their ability to receive reimbursement for such devices, and in part to augment our internal reimbursement resources, ArthroCare acquired DiscoCare in December 2007.
9
In connection with our reimbursement activities, we have adopted the ArthroCare Corporation Health Care Compliance Program, implementing the key elements of a model health care compliance plan developed by the Office of Inspector General of the United States Department of Health and Human Services, or OIG, which oversees compliance with applicable Federal laws in connection with the payment for products by government funded programs.
Patents and Proprietary Rights
Our ability to compete effectively depends in part on developing and maintaining the proprietary aspects of our technologies, including Coblation technology and our acquired technologies. We own over 160 issued U.S. patents and over 100 issued international patents. In addition, we have over 200 U.S. and international pending patent applications. We believe our issued patents are directed at, among other things, the core technology used in our soft-tissue surgery systems, including both multi-electrode and single electrode configurations of our disposable devices, as well as the use of Coblation technology in many different surgical procedures. In addition, we believe that our issued patents are directed at many of the core features of our acquired technologies from Opus, Atlantech, Parallax and ATI.
We cannot assure you the patents we have obtained, or any patents we may obtain as a result of our U.S. or international patent applications, will provide any competitive advantages for our products or that they will not be successfully challenged, invalidated or circumvented in the future. In addition, we cannot assure you that competitors, many of whom have substantial resources and have made substantial investments in competing technologies, will not seek to apply for and obtain patents that will prevent, limit or interfere with our ability to make, use and sell our products either in the United States or in international markets.
A number of other companies, universities and research institutions have filed patent applications or have issued patents relating to monopolar and/or bipolar electrosurgical methods and apparatus. In addition, we have become aware of, and may become aware of in the future, patent applications and issued patents that relate to our products and/or the surgical application of our issued patents and, in some cases, have obtained internal and/or external opinions of our counsel regarding the relevance of certain issued patents to our products. We do not believe that our products currently infringe any valid and enforceable claims of the issued patents that we have reviewed. However, if third-party patents or patent applications contain claims infringed by our technology and such claims are ultimately determined to be valid, we cannot assure you that we would be able to obtain licenses to those patents at a reasonable cost, if at all, or be able to develop or obtain alternative technology. The inability to do either would have a material adverse effect on our business, financial condition, results of operations and future growth prospects. We cannot assure you that we will not have to defend ourselves in court against allegations of infringement.
In addition to patents, we rely on trade secrets and proprietary know-how, which we seek to protect, in part, through confidentiality and proprietary information agreements. We require our employees and consultants to execute confidentiality agreements upon the commencement of an employment or consulting relationship with us. These agreements generally provide that all confidential information developed or made known to the individual by us during the course of the individual’s relationship with us, is to be kept confidential and not disclosed to third parties. These agreements also generally provide that inventions conceived by the individual in the course of rendering services to us shall be our exclusive property. We cannot assure you that employees will not breach the agreements, that we would have adequate remedies for any breach or that our trade secrets will not otherwise become known to or be independently developed by competitors.
The medical device industry has been characterized by extensive litigation regarding patents and other intellectual property rights, and companies in the medical device industry have employed intellectual property litigation to gain a competitive advantage. We cannot assure you that we will not become subject to patent infringement claims or litigation or reexamination or interference proceedings declared by the United States Patent and Trademark office, or USPTO, to determine the priority of inventions.
The defense and prosecution of this lawsuit and intellectual property suits generally, USPTO reexamination and interference proceedings and related legal and administrative proceedings are both costly and time-consuming. If others violate our proprietary rights, further litigation may be necessary to enforce our patents, to protect trade secrets or know-how owned by us or to determine the enforceability, scope and validity of the proprietary rights of others. Any litigation or reexamination or interference proceedings will be costly and cause significant diversion of effort by our technical and management personnel. An adverse determination in existing litigation, additional litigation or reexamination or interference proceedings to which we may become a party could subject us to significant liabilities to third parties, require disputed rights to be licensed from third parties or require us to cease using certain technology. Furthermore, we cannot be sure that we could obtain necessary licenses on satisfactory terms, if at all. Adverse determinations in judicial or administrative proceedings or failure to obtain necessary licenses could prevent us from manufacturing and selling our products, which would have a material adverse effect on our business, financial condition, results of operations, and future growth prospects.
10
Government Regulation
United States
Our products are considered medical devices and are subject to extensive regulation in the United States. We must comply with FDA regulations to market our products or obtain pre-market clearance or approval by the FDA for each of our products and indications before they can be commercialized.
FDA regulations are wide-ranging and govern, among other things:
| • | Product design and development; |
| • | Product testing; |
| • | Product labeling; |
| • | Product storage; |
| • | Premarket clearance or approval; |
| • | Advertising and promotion; and |
| • | Product sales and distribution. |
Non-compliance with applicable regulatory requirements can result in enforcement action, which may include:
| • | Warning letters; |
| • | Fines, injunctions and civil penalties against us; |
| • | Recall or seizure of our products; |
| • | Operating restrictions, partial suspension or total shutdown of our production; |
| • | Refusing our requests for premarket clearance or approval of new products; |
| • | Withdrawing product approvals already granted; and |
| • | Criminal prosecution. |
Unless an exemption applies, generally, before we can introduce a new medical device into the United States market, we must obtain FDA clearance of a 510(k) premarket notification or approval of a premarket approval application, or PMA. If we can establish that our device is “substantially equivalent” to a “predicate device,” i.e., a legally marketed Class I or Class II device or a preamendment Class III device (i.e., a device that was in commercial distribution before May 28, 1976) for which the FDA has not called for PMAs, we may seek clearance from the FDA to market the device by submitting a 510(k) premarket notification. The 510(k) premarket notification must be supported by appropriate data, including, in some cases, clinical data establishing the claim of substantial equivalence to the satisfaction of the FDA.
11
We have received 510(k) clearance to market our Arthroscopic System for surgery of the knee, shoulder, elbow, wrist, hip, and ankle joints. We have received clearance to market our Spinal Surgery System in the United States for spinal and neurosurgery as well as the treatment of symptomatic patients with contained herniated discs. We have received 510(k) clearance to market our interventional therapy devices for percutaneous, intraoperative and spinal procedures. We have received 510(k) clearance to market our ENT Surgery System in general head, neck and sinus surgical procedures, as well as treatment of snoring, turbinate reduction, submucosal palatal and tissue shrinkage procedures and tonsillectomies. In addition, we have received 510(k) clearance to market our Cosmetic Surgery System in general dermatology and for skin resurfacing for the treatment of wrinkles. We have received 510(k) clearance to market our Coblation-based products for a variety of laparoscopic and open general surgery and gynecology procedures. We have received 510(k) clearance to market our Coblation-based urology products for endoscopic urological procedures, including TURP and TUIP.
We cannot assure you we will be able to obtain necessary clearances or approvals to market any other products, or existing products for new intended uses, on a timely basis, if at all. Delays in receipt or failure to receive clearances or approvals, the loss of previously received clearances or approvals, or failure to comply with existing or future regulatory requirements could have a material adverse effect on our business, financial condition, results of operations and future growth prospects.
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in the intended use of the device, technology, materials, packaging, and certain manufacturing process may require a new 510(k) clearance and the FDA may retroactively require the manufacturer to submit a premarket notification requesting 510(k) clearance. The FDA also can require the manufacturer to cease marketing and/or recall the modified device until 510(k) clearance is obtained. FDA guidance documents define when to submit premarket notifications for new or modified devices. These guidance documents also define modifications for which a new 510(k) is not required. We have modified some of our marketed devices, and have determined that in certain instances new 510(k) clearances are not required. When a 510(k) clearance has been required, premarket notifications have been submitted to the FDA documenting the specific device modifications. No assurance can be given that the FDA would agree with any of our decisions not to seek 510(k) clearance. If the FDA requires us to cease marketing and/or recall the modified device until we obtain a new 510(k) clearance, our business, financial condition, results of operations and future growth prospects could be materially adversely affected.
If we cannot establish that a proposed device is substantially equivalent to a legally marketed device, we must seek premarket approval through submission of a PMA application. A PMA application must be supported by extensive data, including, in many instances, preclinical and clinical trial data, as well as extensive literature to prove the safety and effectiveness of the device. Often, clinical testing will require the submission and approval of an Investigational Device Exemption, or IDE, which defines the scope and conditions of the clinical trial. If necessary, we will file an IDE and seek a PMA application for approval to sell our potential products at the successful conclusion of the study and the submission of the completed IDE. The PMA process can be expensive, uncertain and lengthy. We cannot assure you that we will be able to obtain PMA approvals on a timely basis, if at all, and delays in receipt or failure to receive approvals, could have a material adverse effect on our business, financial condition, results of operations and future growth prospects.
We are also required to demonstrate and maintain compliance with the QSR. The QSR incorporates the requirements of Good Manufacturing Practice and relates to product design, testing, and manufacturing quality assurance, as well as the maintenance of records and documentation. The FDA enforces the QSR through inspections, including post-market surveillance. We cannot assure you that we or our key component suppliers are or will continue to be in compliance, will not encounter any manufacturing difficulties, or that we or any of our subcontractors or key component suppliers will be able to maintain compliance with regulatory requirements. Failure to do so will have a material adverse effect on our business, financial condition, results of operations and future growth prospects.
We may not promote or advertise our products for uses not within the scope of our clearances or approvals or make unsupported safety and effectiveness claims. These determinations can be subjective. We cannot assure you that the FDA would agree that all of our promotional claims are permissible or that the FDA will not require us to revise our promotional claims or take enforcement action against us based upon our labeling and promotional materials.
12
International
International sales of our products are subject to strict regulatory requirements. The regulatory review process varies from country to country. We have obtained regulatory clearance to market our Arthroscopic System in Europe, Japan, Australia, Taiwan, Korea, Canada, China, Israel, the Middle East, South America and Mexico; to market our spinal surgery products in Europe, Canada, Japan, South America, Australia, Korea, Mexico, the Middle East and Taiwan; to market our ENT surgery products in Europe, Australia, Canada, China, Israel, Japan, Middle East, Korea Taiwan, Australia and South America; to market our cosmetic surgery products in Europe, Australia, Canada, the Middle East, Korea, South America and Israel; to market our general surgery products in Europe, Canada, the Middle East, Korea, South America, and Taiwan; and to market neurosurgery products in Europe, Australia, South America and Japan, but we have not obtained any other international regulatory approvals in other international markets.
Product Liability Risk and Insurance Coverage
The development, manufacture and sale of medical products entail significant risk of product liability claims. Our current product liability insurance coverage limits are $10 million per occurrence and $10 million in the aggregate. We cannot assure you that such coverage limits are adequate to protect us from any liabilities we might incur in connection with the development, manufacture and sale of our products. In addition, we may require increased product liability coverage as products are successfully commercialized in additional applications. Product liability insurance is expensive and in the future may not be available to us on acceptable terms, if at all. A successful product liability claim or series of claims brought against us in excess of our insurance coverage could have a material adverse effect on our business, financial condition, results of operations and our ability to attract and retain customers for our products.
Employees
As of December 31, 2007, in North America, we had 413 employees, of which 29 were engaged in manufacturing activities, 96 in research and development activities, 196 in sales and marketing activities, 35 in regulatory affairs and quality assurance and 57 in administration and finance. In Europe, we had 145 employees in sales, marketing, product development, customer service, manufacturing and administration who are responsible for our international business. In Costa Rica, we had 507 employees engaged in manufacturing and administration. In Asia Pacific we had 9 employees engaged in sales and marketing activities. We have no employees covered by collective bargaining agreements, and we believe we maintain good relations with our employees.
ITEM 1A. RISK FACTORS
We operate in a dynamic and rapidly changing industry that involves numerous risks and uncertainties. The risks and uncertainties described below are those that we currently believe may materially affect us. Other risks and uncertainties that we do not presently consider to be material or of which we are not presently aware, may become important factors that affect us in the future.
We Face Intense Competition
The markets for our current products in our core businesses are intensely competitive. These markets include arthroscopy, spinal surgery and ENT surgery. We cannot assure you that other companies will not succeed in developing technologies and products that are more effective than ours, or that would render our technology or products obsolete or uncompetitive in these markets.
In arthroscopy, we compete against companies such as Johnson & Johnson, Smith & Nephew, Inc., Conmed Corporation, Stryker Corp., and Arthrex. Specifically, Johnson & Johnson, Smith & Nephew and Stryker are currently marketing bipolar electrosurgical systems for tissue ablation and shrinkage and Arthrex and Conmed are currently marketing monopolar electrosurgical systems for tissue ablation. Arthrex, Smith & Nephew, Conmed, Stryker and DePuy Mitek, a division of Johnson & Johnson, market products that directly compete with our shoulder anchors, acquired as part of our Opus acquisition. In spinal surgery, we compete against companies that market products to remove tissue and treat spinal disorders. We compete against Stryker, which markets the Dekompressor® device, which uses a mechanical auger, as well as Clarus, which markets the Nucleotome® device, to perform disc decompression procedures. In addition, Smith & Nephew Endoscopy and the Radionics division of Integra are currently marketing percutaneous thermal heating products for treating certain types of disc pain. Our MD SpineWand for microdiscectomy competes indirectly with large spine companies and their mechanical instruments, such as DePuy Spine, Medtronic Sofamor Danek, Zimmer, Stryker Spine and Synthes. Stryker, Cardinal Health, Cook and Kyphon, which was recently acquired by Medtronic, market products that compete directly with our Parallax product line. In ENT surgery, we compete against companies that offer manual instruments, such as Smith & Nephew, Inc., Stryker Corp., Conmed Corporation, and Medtronic Neurosurgery. In addition, we compete with companies that develop and market lasers for various ENT surgery applications, including Lumenis and Gyrus Group International, Inc., also sell medical devices for the treatment of various ENT disorders, including snoring and obstructive sleep apnea. In addition, Gyrus has introduced a bipolar system for tonsillectomy procedures that competes directly with our Coblation-based tonsillectomy products. Medtronic and Gyrus both market sinus surgery packing products that compete with our ATI products.
13
Many of our competitors have significantly greater financial, manufacturing, marketing, distribution and technical resources than we do. Some of these companies offer broad product lines that they may offer as a single package and frequently offer significant discounts as a competitive tactic. For example, in order to compete successfully, we anticipate that we may have to continue to offer substantial discounts on our controllers, place controllers at customers sites at no cost or in return for a minimum purchase commitment of our surgical wands in order to increase demand for our disposable devices, and that this competition could have a material adverse effect on our business, financial condition, results of operations and future growth prospects. Furthermore, some of our competitors utilize purchasing contracts that link discounts on the purchase of one product to purchases of other products in their broad product lines. Many of the hospitals in the United States have purchasing contracts with our competitors. Accordingly, customers may be dissuaded from purchasing our products rather than the products of these competitors to the extent the purchase would cause them to lose discounts on products.
We May Not Be Able to Keep Pace with Technological Change or to Successfully Develop New Products with Wide Market Acceptance, Which Could Cause Us to Lose Business to Competitors
We may not be able to keep pace with technology or to develop viable new products, as we compete in a market characterized by rapidly changing technology. Our future financial performance will depend in part on our ability to develop and manufacture new products in a cost-effective manner, to introduce them to the market on a timely basis, and to achieve market acceptance. Factors which may result in delays of new product introductions or cancellation of our plans to manufacture and market new products include capital constraints, research and development delays or delays in acquiring regulatory approvals. Our new products and new product introductions may fail to achieve expected levels of market acceptance. Factors impacting the level of market acceptance include our ability to successfully implement new technologies, the timeliness of our product introductions, our product pricing strategies, and the financial and technological resources for product promotion and development available.
We Are Dependent Upon Our Core Coblation Technology
We commercially introduced our Coblation technology product lines in December 1995. Since these product lines accounted for over 70 percent of our product sales in 2007, we are highly dependent on its sales. We cannot assure you that we will be able to continue to manufacture arthroscopy products in commercial quantities at acceptable costs, or that we will be able to continue to market such products successfully.
To achieve increasing disposable device sales over time, we believe we must continue to penetrate the market in knee procedures, expand physicians’ education with respect to Coblation technology and continue working on new product development efforts specifically for knee applications. Furthermore, in order to maintain and increase current market penetration we must continue to increase our installed base of controllers to generate increased disposable device revenue. To date, we have placed at no charge or have priced our arthroscopic controllers at substantial discounts in order to stimulate demand for our disposable devices.
We believe that surgeons will not use our products unless they determine, based on experience, clinical data and other factors, that these systems are an attractive alternative to conventional means of tissue ablation. There are only a few independently published clinical reports and limited long-term clinical follow-up to support the marketing efforts for our Arthroscopic System. We believe that if continued recommendations and endorsements by influential surgeons or long-term data do not support our current claims of efficacy, our business, financial condition, results of operations and future growth prospects could be materially adversely affected.
14
We Face Uncertainty Over Reimbursement
Failure by physicians, hospitals and other users of our products to obtain sufficient reimbursement from health care payers for procedures in which our products are used, or adverse changes in environmental and private third-party payers’ policies toward reimbursement for such procedures would have a material adverse effect on our business, financial condition, results of operations and future growth prospects. Reimbursement for arthroscopic, spinal surgery, neurosurgery, ENT surgery, gynecology, urology, cardiology and general surgery procedures performed using our devices that have received FDA clearance has generally been available in the United States. Typically, cosmetic surgery procedures are not reimbursed.
We are unable to predict what changes will be made in the reimbursement methods used by third-party health care payers. In addition, some health care providers are moving toward a managed care system in which providers contract to provide comprehensive health care for a fixed cost per person. Managed care providers are attempting to control the cost of health care by authorizing fewer elective surgical procedures. We anticipate that in a prospective payment system, such as the diagnosis related group system utilized by Medicare, and in many managed care systems used by private health care payers, the cost of our products will be incorporated into the overall cost of the procedure and that there will be no separate, additional reimbursement for our products.
If we obtain the necessary international regulatory approvals, market acceptance of our products in international markets would be dependent, in part, upon the availability of reimbursement within prevailing health care payment systems. Reimbursement and health care payment systems in international markets vary significantly by country and include both government-sponsored health care and private insurance. We intend to seek international reimbursement approvals, although we cannot assure investors that any such approvals will be obtained in a timely manner, if at all.
Plasma Disc Decompression is our version of minimally invasive disc decompression, where tissue is removed from the nucleus to decompress the disc. Several payers consider these procedures, regardless of the method used to remove tissue, to be investigational, and others cover specific methods of disc decompression, but consider Plasma Disc Decompression investigational. DiscoCare, the reimbursement service provider acquired by the Company in December 2007, has had some success in obtaining authorizations for Plasma Disc Decompression procedures, by following a strict medical and documentation regimen, from all major private health insurance carriers and most workers compensation plans, including carriers who consider Plasma Disc Decompression, or disc decompression procedures in general, to be experimental. However, there is no assurance that we will continue to be successful in obtaining authorizations for these cases in the future. In addition, there is no assurance that we will be able to obtain positive coverage decisions from major insurance companies for Plasma Disc Decompression at any point in the future.
We Rely Exclusively on Our Costa Rica Facility to Manufacture Our High-Volume Coblation Products. If Our Costa Rica Facility is Unable to Produce Our Coblation Products in Sufficient Quantities or With Adequate Quality, or if Our Operations at Our Costa Rican Facility are Disrupted, We Will Be Forced to Find Alternative Manufacturing Options, Which Could Cause Sales to be Delayed or Increase Our Manufacturing Costs, Which Could Harm Our Reputation and Profitability
Our high-volume disposable devices and controllers are manufactured at our Company-owned facility in an industrial park in San Jose, Costa Rica. If our Costa Rica facility is not able to produce sufficient quantities of our controllers and other high-volume Coblation products with adequate quality, or if our Costa Rica operations are disrupted for any reason, then we may be forced to locate alternative manufacturing facilities, including facilities operated by third parties. Disruptions may include, but are not limited to, changes in the legal and regulatory environment in Costa Rica; slowdowns or work stoppages within the Costa Rican customs authorities; acts of God and other issues associated with significant operations that are remote from our headquarters and operations centers. Locating alternative facilities would be time-consuming, would disrupt our production and cause shipment delays and could result in damage to our reputation and profitability. We cannot guarantee that alternative manufacturing facilities offering the cost and tax advantages associated with our Costa Rica facility would be available on favorable terms, or at all.
We Are Dependent on Key Suppliers
Some of the key components of our products are purchased from single vendors. If the supply of materials from a sole source supplier were interrupted, replacement or alternative sources might not be readily obtainable due to the regulatory requirements applicable to our manufacturing operations or the availability of certain product drawings and/or specifications. In addition, a new or supplemental filing with applicable regulatory authorities may require clearance prior to our marketing a product containing new material. This clearance process may take a substantial period of time and we cannot assure investors that we would be able to obtain the necessary regulatory approval for a new material to be used in our products on a timely basis, if at all. This could create supply disruptions that would materially adversely affect our business, financial condition, results of operations and future growth prospects.
15
In addition, we currently single source our product sterilization requirements. While there are alternate sources available, we would be required to qualify and validate a new supplier(s), which could lead to a disruption in our operation and ability to supply products for a period of time.
We are also dependent on the performance of DHL, our logistics partner. Should this supplier experience a work stoppage for whatever reason, we would be unable to supply products to our customers on a timely basis. This work stoppage could take the form of labor action, significant weather events or extended systems downtime, which could materially adversely affect our business.
Circumstances Associated with Our Acquisition Strategy and Internal Growth May Adversely Affect Our Operating Results
An important element of our growth strategy has been the pursuit of acquisitions of other businesses that expand or complement our existing products. Integrating businesses, however, involves a number of special risks, including the possibility that management may be distracted from regular business concerns by the need to integrate operations, unforeseen difficulties in integrating operations and systems, problems assimilating and retaining our employees or the employees of the acquired company, accounting issues that could arise in connection with, or as a result of, the acquisition of the acquired company, regulatory or compliance issues that could exist at an acquired company, challenges in retaining our customers or the customers of the acquired company following the acquisition and potential adverse short term effects on operating results through increased costs or otherwise. In addition, we may incur debt to finance future acquisitions and/or may issue securities in connection with future acquisitions which may dilute the holdings of our current and future stockholders.
In addition to the risks associated with acquisition-related growth, our business has grown in size and complexity over the past few years as a result of internal growth. This growth and increase in complexity have placed significant demands on management, systems, internal controls and financial and physical resources. To meet such demands, we intend to continue to invest in new technology, make other capital expenditures and, where appropriate, hire and/or train employees with expertise to handle these particular demands. If we are unable to successfully complete and integrate strategic acquisitions in a timely manner or if we fail to efficiently manage operations in a way that accommodates continued internal growth, our business, financial condition or operating results could be adversely affected.
In 2005, we acquired substantially all of the assets of ATI, a maker of wound care products for ENT indications. In 2004 and 2002, we acquired three companies representing three lines of business independent of our core Coblation platform: Atlantech and its mechanical handheld instruments; MDA and its line of Parallax bone cement and delivery systems; and Opus and its line of tissue suturing and anchoring products. Integration of these businesses has been a significant challenge to our existing resources. At times, these integration issues have restricted sales of one or more product lines as a host of systems, distribution, logistics and product supply issues have materialized. While we are diligently focused on successful integration, the newness of these technologies along with the dispersed nature of the product development and supply chain represents a risk to our business and availability of internal resources. If we are unsuccessful in integrating these businesses, or experience disruptions related to our integration efforts, then our financial condition, results of operations and future growth prospects could be materially adversely affected.
Our Business May Become Increasingly Susceptible to Risks Associated with International Operations
International operations are generally subject to a number of risks, including:
| • | protectionist laws and business practices that favor local competition; | |
| • | changes in jurisdictional tax laws including laws regulating intercompany transactions; | |
| • | dependence on local vendors; | |
| • | multiple, conflicting and changing governmental laws and regulations; | |
| • | difficulties in collecting accounts receivable; | |
16
| • | seasonality of operations; | |
| • | difficulties in staffing and managing foreign operations; | |
| • | licenses, tariffs, and other trade barriers; | |
| • | loss of proprietary information due to piracy, misappropriation or weaker laws regarding intellectual property protection; | |
| • | foreign currency exchange rate fluctuations; | |
| • | political and economic instability; and | |
| • | acquisitions and related IPR&D. |
We derived 21 percent, 20 percent and 21 percent of our total product sales for the years ended December 31, 2007, 2006 and 2005, respectively, from customers located outside of the Americas. We expect international revenue to remain a large percentage of total revenue and we believe that we must continue to expand our international sales activities to be successful. Historically, a majority of our international revenues and costs have been denominated in foreign currencies, and we expect future international revenues and costs will be denominated in foreign currencies. Our international sales growth will be limited if we are unable to establish appropriate foreign operations, expand international sales channel management and support organizations, hire additional personnel, develop relationships with international sales representatives, and establish relationships with additional distributors. In that case, our business, operating results and financial condition could be materially adversely affected. Even if we are able to successfully expand international operations, we cannot be certain that we will be able to maintain or increase international market demand for our products.
We May Be Unable to Effectively Protect Our Intellectual Property
Our ability to compete effectively depends in part on developing and maintaining the proprietary aspects of our Coblation technology and our acquired technologies. We believe that our issued patents are directed at the core technology used in our soft-tissue surgery systems, including both multi-electrode and single electrode configurations of our disposable devices, as well as the use of Coblation technology in specific surgical procedures. In addition, we believe that our issued patents are directed at many of the core features of our acquired technologies from Opus, Parallax, Atlantech and ATI.
There is no assurance that the patents we have obtained, or any patents we may obtain as a result of our pending U.S. or international patent applications, will provide any competitive advantages for our products. We also cannot assure investors that those patents will not be successfully challenged, invalidated or circumvented in the future. In addition, we cannot provide assurance that competitors, many of which have substantial resources and have made substantial investments in competing technologies, have not already applied for or obtained, or will not seek to apply for and obtain, patents that will prevent, limit or interfere with our ability to make, use and sell our products either in the United States or in international markets. Patent applications are maintained in secrecy for a period after filing. We may not be aware of all of the patents and patent applications potentially adverse to our interests.
A number of medical device and other companies, universities and research institutions have filed patent applications or have issued patents relating to monopolar and/or bipolar electrosurgical methods and apparatus. We have received, and we may receive in the future, notifications of potential conflicts of existing patents, pending patent applications and challenges to the validity of existing patents. In addition, we have become aware of, and may become aware of in the future, patent applications and issued patents that relate to our products and/or the surgical applications and issued patents and, in some cases, have obtained internal and/or external opinions of counsel regarding the relevance of certain issued patents to our products. We do not believe that our products currently infringe any valid and enforceable claims of the issued patents that we have reviewed. However, if third-party patents or patent applications contain claims infringed by our technology and such claims are ultimately determined to be valid, we may not be able to obtain licenses to those patents at a reasonable cost, if at all, or be able to develop or obtain alternative technology. Our inability to do either would have a material adverse effect on our business, financial condition, results of operations and prospects. We cannot assure investors that we will not have to defend ourselves in court against allegations of infringement of third-party patents, or that such defense would be successful.
17
In addition to patents, we rely on trade secrets and proprietary know-how, which we seek to protect, in part, through confidentiality and proprietary information agreements. We require our key employees and consultants to execute confidentiality agreements upon the commencement of an employment or consulting relationship with us. These agreements generally provide that all confidential information, developed or made known to the individual during the course of the individual’s relationship with us, is to be kept confidential and not disclosed to third parties. These agreements also generally provide that inventions conceived by the individual in the course of rendering services to us shall be our exclusive property. We cannot assure investors that employees will not breach such agreements, that we would have adequate remedies for any breach or that our trade secrets will not otherwise become known to or be independently developed by competitors.
We May Become Subject to Patent Litigation
The medical device industry has been characterized by extensive litigation regarding patents and other intellectual property rights, and companies in the medical device industry have employed intellectual property litigation to gain a competitive advantage. We cannot assure investors that we will not become subject to patent infringement claims or litigation or interference proceedings declared by the USPTO, to determine the priority of inventions.
We Must Obtain Governmental Clearances or Approvals Before We Can Sell Our Products; We Must Continue to Comply with Applicable Laws and Regulations
Our products are considered medical devices and are subject to extensive regulation in the United States. We must obtain premarket clearance or approval by the FDA for each of our products and indications before they can be commercialized. International sales of our products are also subject to strict regulatory requirements. Information about the U.S. and foreign regulatory requirements pertaining to our products and indications before they can be commercialized can be found under the heading “Government Regulations” in Item 1, above.
Product Liability Claims Could Adversely Impact our Financial Condition and Impair our Reputation
Our business exposes us to potential product liability risks which are inherent in the design, manufacture and marketing of medical devices. In addition, many of the medical devices we manufacture and sell are designed to be implanted in the human body for long periods of time. Component failures, manufacturing flaws, design defects or inadequate disclosure of product-related risks or product-related information with respect to these or other products we manufacture or sell could result in an unsafe condition or injury to a patient. The occurrence of such a problem could result in product liability claims or a recall of one or more of our products, which could ultimately result in the removal from the body of such products and claims regarding costs associated therewith.
We are Dependent Upon a Number of Key Management and Technical Personnel
The loss of the services of one or more key employees or consultants could have a material adverse effect on us. Our success also depends on our ability to attract and retain additional highly qualified management and technical personnel. We face intense competition for qualified personnel, any of whom often receive competing employment offers. We cannot assure you that we will continue to be able to attract and retain such personnel. Furthermore, our scientific advisory board members are all otherwise employed on a full-time basis. As a result, our scientific advisory board members are not available to devote their full time or attention to our affairs.
Our Operating Results May Fluctuate
We achieved profitability in 1999 and, as of December 31, 2007, we had retained earnings of $78.4 million. Results of operations may fluctuate significantly from quarter to quarter due to many factors, including the following:
| • | The introduction of new product lines; | |
| • | Increased penetration in existing applications; |
18
| • | Product returns; | |
| • | Achievement of research and development milestones; | |
| • | The amount and timing of receipt and recognition of license fees; | |
| • | Manufacturing or supply disruptions; | |
| • | Internal systems availability and uptimes; | |
| • | Timing of expenditures; | |
| • | Absence of a backlog of orders; | |
| • | Receipt of necessary regulatory approvals; | |
| • | The level of market acceptance for our products; | |
| • | Acquisition related in-process research and development write-offs; | |
| • | Timing of the receipt of orders and product shipments; and | |
| • | Promotional programs for our products. |
We cannot provide assurance that future quarterly fluctuations will not adversely affect our business, financial condition, results of operations or future growth prospects. Our revenues and profitability will be critically dependent on whether or not we can successfully continue to market our existing Coblation-based technology product lines and our new product introductions. We cannot assure investors that we will maintain or increase our revenues or level of profitability.
The Market Price of Our Stock May Be Highly Volatile
During the fiscal year ended December 31, 2007 our common stock traded in a closing price range of $33.93 to $64.84 per share. The market price of our common stock could continue to fluctuate substantially due to a variety of factors, including:
| • | Quarterly fluctuations in results of our operations; | |
| • | Our ability to successfully commercialize our products; | |
| • | Announcements regarding results of regulatory approval filing, clinical studies or other testing, technological innovations or new products commercialized by us or our competitors; | |
| • | Developments concerning government regulations, proprietary rights or public concern as to the safety of our technology; | |
| • | The execution of new collaborative agreements and material changes in our relationships with our business partners; | |
| • | Market reaction to acquisitions and trends in sales, marketing, and research and development; | |
| • | Rumors or dissemination of false information; | |
| • | Changes in coverage or earnings estimates by analysts; | |
| • | Sales of common stock by existing stockholders; and | |
| • | Economic and political conditions. |
19
The market price for our common stock may also be affected by our ability to meet analysts’ expectations. Any failure to meet such expectations, even slightly, could have an adverse effect on the market price and volume fluctuations. This volatility has had a significant effect on the market prices of securities issued by many companies for reasons unrelated to the operating performance of these companies. In the past, following periods of volatility in the market price of a company’s securities, the risk of securities class action litigation has increased. If similar litigation were instituted against us, it could result in substantial costs and a diversion of our management’s attention and resources, which could have an adverse effect on our business, results of operations and financial condition. See “Item 5: Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities,” for more information regarding fluctuations in the price of our common stock.
Delaware Law and Provisions in Our Charter Could Make the Acquisition of Our Company by Another Company More Difficult
Certain provisions of our certificate of incorporation and bylaws may have the effect of making it more difficult for a third-party to acquire, or of discouraging a third-party from attempting to acquire, control of our company. This could limit the price that certain investors might be willing to pay in the future for shares of our common stock. Some provisions of our certificate of incorporation and bylaws allow us to issue preferred stock without any vote or further action by the stockholders, to eliminate the right of stockholders to act by written consent without a meeting, to specify procedures for director nominations by stockholders and submission of other proposals for consideration at stockholder meetings, and to eliminate cumulative voting in the election of directors. Some provisions of Delaware law applicable to us could also delay or make more difficult a merger, tender offer or proxy contest involving us, including Section 203, which prohibits a Delaware corporation from engaging in any business combination with any interested stockholder for a period of three years unless certain conditions are met. The procedures required for director nominations and stockholder proposals and Delaware law could have the effect of delaying, deferring or preventing a change in control of ArthroCare, including without limitation, discouraging a proxy contest or making more difficult the acquisition of a substantial block of our common stock.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
We lease a facility in Austin, Texas, which serves as our corporate headquarters. The Austin, Texas facility is used for general and administrative purposes as well as for research and development activities. Our lease for this building will expire in June 2016.
We lease a facility in Sunnyvale, California for administrative offices, research and development, general and administrative purposes and prototype manufacturing. Our lease for this building will expire in February 2014. We also lease, on a month-to-month basis, a warehouse in a neighboring building in Sunnyvale, California for warehousing and distribution.
As a result of our acquisition of Opus Medical we lease a facility in Irvine, California, which is used for product development and prototype manufacturing associated with the Opus product line. This lease expires at the end of 2011.
Internationally, we lease a facility in Stockholm, Sweden, which serves as our headquarters for international operations. We lease additional international properties in Harrogate, England, for administrative, sales and marketing purposes as well as contributions to the product development process for a portion of our Sports Medicine product line. In Glenfield, England we lease a plant that is used for manufacturing the Rapid Rhino product set and in Opglabbeek, Belgium we lease a facility that is used for a European repair center. Expiration dates related to these international leases range from two to five years.
In Costa Rica we own a building located in a tax-advantaged business park. This facility serves as our principal manufacturing location for disposable devices.
We believe these facilities, in addition to multiple less significant facilities leased in various countries around the world for sales, marketing and administrative purposes, are sufficient for our operations at least through 2008.
20
ITEM 3. LEGAL PROCEEDINGS
From time to time, we are a defendant in certain lawsuits alleging product liability, patent infringement or other claims incurred in the ordinary course of business. These claims are generally covered by certain insurance policies, subject to certain deductible amounts and maximum policy limits. When there is no insurance coverage, as would typically be the case primarily in lawsuits alleging patent infringement, we establish sufficient reserves to cover probable losses associated with such claims. Except as otherwise described above, we have product liability insurance coverage in amounts we consider necessary to prevent material losses. We recognize losses when they are known or considered probable and the amount can be reasonably estimated.
Defending and prosecuting intellectual property suits, USPTO interference proceedings and related legal and administrative proceedings are costly and time-consuming. Further litigation may be necessary to enforce our patents, to protect our trade secrets or know-how or to determine the enforceability, scope and validity of the proprietary right of others. Any litigation or interference proceedings will be costly and will result in significant diversion of effort by technical and management personnel. An adverse determination in any of the litigation or interference proceedings to which we may become a party could subject us to significant liabilities to third parties, require us to license disputed rights from third parties or require us to cease using such technology, which would have a material adverse effect on our business, financial condition, results of operations and future growth prospects. Patent and intellectual property disputes in the medical device area have often been settled through licensing or similar arrangements, and could include ongoing royalties. We cannot assure provide assurance that we can obtain any necessary licenses on satisfactory terms, if at all.
While we are not aware of any pending investigation concerning DiscoCare, we are responding to a request from the NASDAQ Stock Exchange regarding DiscoCare.
ITEM 4. SUBMISSION OF MATTERS TO A VOTE OF SECURITY HOLDERS
No matters were submitted to a vote of security holders during the fourth quarter of the fiscal year covered by this Annual Report on Form 10-K.
PART II
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Our common stock trades publicly on The NASDAQ Stock Exchange under the symbol ARTC. The following table sets forth, for the periods indicated, the quarterly high and low closing sales prices of our Common stock.
2007 | |||||||||||||
Quarter 1 | Quarter 2 | Quarter 3 | Quarter 4 | ||||||||||
| High | $ | 40.05 | $ | 44.53 | $ | 58.99 | $ | 64.84 | |||||
| Low | 33.93 | 36.68 | 43.77 | 45.97 | |||||||||
2006 | |||||||||||||
Quarter 1 | Quarter 2 | Quarter 3 | Quarter 4 | ||||||||||
| High | $ | 48.05 | $ | 48.64 | $ | 47.88 | $ | 48.06 | |||||
| Low | 41.03 | 38.53 | 39.11 | 39.92 | |||||||||
As of February 19, 2008, there were no outstanding shares of preferred stock and 249 holders of record of 26,585,219 shares of outstanding common stock. We have not paid any cash dividends since our inception and do not anticipate paying cash dividends on our common stock in the foreseeable future.
ISSUER PURCHASES OF EQUITY SECURITIES
In December 2007, our Board of Directors approved a discretionary share repurchase program that allows us to purchase up to $75.0 million of our outstanding common stock on a periodic basis in the open market. Pursuant to the program, in December 2007 we purchased 1,255,982 shares at an average cost of $47.49 per share. In January 2008, we purchased an additional 243,918 shares at an average price of $48.29 per share. We repurchased the shares based on an evaluation of our long-term cash forecast and balance sheet capacity, including anticipated capital requirements.
21
The description of equity compensation plans required by Item 201(d) of Regulation S-K is incorporated herein by reference to Part III, Item 12 of this Form 10-K.
PERFORMANCE GRAPH
The following graph shows a comparison of total stockholder return for holders of our common stock for the five years ended December 31, 2007 compared with the NASDAQ Stock Exchange, U.S. Index and the NASDAQ Health Services Index. This graph is presented pursuant to SEC rules. We believe that while total stockholder return can be an important indicator of corporate performance, the stock prices of medical device companies like ours are subject to a number of market-related factors other than company performance, such as those discussed in “Risk Factors” in Item I above.
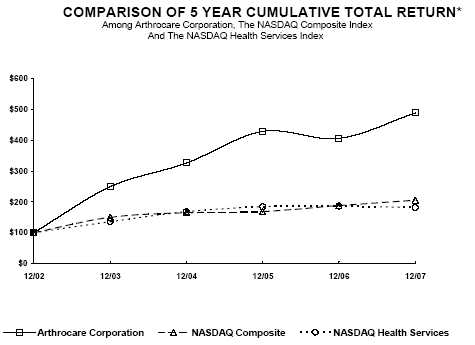
| * | $100 invested on December 31, 2002 in stock or in index - including reinvestment of dividends. Our most recent fiscal year ended December 31, 2007. |
| Cumulative Total Return | |||||||||||||||||||
| 12/02 | 12/03 | 12/04 | 12/05 | 12/06 | �� | 12/07 | |||||||||||||
| ArthroCare Corporation | 100.00 | 248.73 | 325.48 | 427.82 | 405.28 | 487.82 | |||||||||||||
| NASDAQ Stock Exchange (U.S.) | 100.00 | 149.75 | 164.64 | 168.60 | 187.83 | 205.22 | |||||||||||||
| NASDAQ Health Services | 100.00 | 135.61 | 168.24 | 184.41 | 186.06 | 181.42 | |||||||||||||
22
ITEM 6. SELECTED CONSOLIDATED FINANCIAL DATA
The following selected financial data should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and notes thereto included in this Annual Report on Form 10-K. The statements of operations data for the years ended December 31, 2007, 2006 and 2005 and the balance sheet data as of December 31, 2007 and 2006 have been derived from audited consolidated financial statements included elsewhere in this report. The consolidated statement of operations data for the years ended December 31, 2004 and 2003 and the balance sheet data as of December 31, 2005, 2004 and 2003 have been derived from audited consolidated financial statements that are not included in this report. The historical results are not necessarily indicative of the results of operations to be expected in the future. Net income for 2005 and net loss for 2004 reflect significant charges related to acquisitions during those periods.
Year Ended December 31, | ||||||||||||||||
2007 | 2006 | 2005 | 2004 | 2003 | ||||||||||||
(in thousands, except per share data) | ||||||||||||||||
Statements of Operations Data: | ||||||||||||||||
| Product sales | $ | 307,596 | $ | 253,376 | $ | 206,533 | $ | 147,830 | $ | 114,719 | ||||||
| Royalties, fees and other | 11,646 | 9,625 | 7,801 | 6,318 | 4,134 | |||||||||||
| Total revenues | 319,242 | 263,001 | 214,334 | 154,148 | 118,853 | |||||||||||
| Gross profit | 234,518 | 186,163 | 150,128 | 103,048 | 80,912 | |||||||||||
| Operating expenses | 181,343 | 143,693 | 117,069 | 126,801 | 72,767 | |||||||||||
| Net income (loss) | 43,180 | 31,675 | 23,530 | (26,189 | ) | 7,456 | ||||||||||
| Basic net income (loss) per share | $ | 1.57 | $ | 1.21 | $ | 0.97 | $ | (1.21 | ) | $ | 0.36 | |||||
| Diluted net income (loss) per share | $ | 1.50 | $ | 1.14 | $ | 0.89 | $ | (1.21 | ) | $ | 0.34 | |||||
December 31, | ||||||||||||||||
2007 | 2006 | 2005 | 2004 | 2003 | ||||||||||||
(in thousands) | ||||||||||||||||
Balance Sheet Data: | ||||||||||||||||
| Cash, cash equivalents and available-for-sale securities (including long-term portion) | $ | 43,250 | $ | 30,756 | $ | 23,317 | $ | 21,836 | $ | 31,318 | ||||||
| Working capital | 148,348 | 113,744 | 98,841 | 80,912 | 67,670 | |||||||||||
| Total assets | 450,068 | 375,046 | 266,978 | 240,531 | 138,138 | |||||||||||
| Long-term liabilities | 67,393 | 2,870 | 4,092 | 34,290 | 155 | |||||||||||
| Total stockholders’ equity (1) | 340,503 | 322,503 | 228,894 | 175,252 | 120,648 | |||||||||||
| (1) | We have not declared any cash dividends on our common stock since our inception. We do not anticipate paying cash dividends in the foreseeable future. |
23
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following discussion should be read in conjunction with the consolidated financial statements and notes thereto appearing elsewhere in this Form 10-K. Statements in this Management’s Discussion and Analysis of Financial Condition and Results of Operations and elsewhere in this annual report on Form 10-K which express that we “believe,” “anticipate,” “expect” or “plan to” as well as other statements which are not historical fact, are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and are subject to the safe harbors created under the Securities Act of 1933 and the Securities Exchange Act of 1934. Readers are cautioned that these forward-looking statements are only predictions and are subject to risks, uncertainties, and assumptions that are difficult to predict. As such, actual events or results may differ materially as a result of the risks and uncertainties described herein and elsewhere including, but not limited to, those factors discussed in “Risk Factors” set forth in Part I of this Report as well as other risks and uncertainties in the documents incorporated herein by reference.
Overview
We are a multi-business medical device company that develops, manufactures and markets minimally invasive surgical products, many of which are based on our patented Coblation® technology. We currently market minimally invasive surgical products across three core business units — ArthroCare Sports Medicine, ArthroCare Spine, and ArthroCare Ear, Nose and Throat, or ENT — but also have developed, manufactured and marketed Coblation-based and complementary products for application in neurology, cosmetic surgery, urology and gynecology, with research continuing in additional areas. In each of our core business units we are focused on driving the application of enabling technologies, primarily for plasma-based soft tissue removal, and increasing the number of minimally invasive procedures being performed.
In December 1995, we introduced our Arthroscopy System commercially in the United States and have derived a significant portion of our sales from this system. Our strategy includes placing controller units, which enable use of our disposable Coblation products, at substantial discounts or placing controllers at customer sites at no cost in order to generate future disposable product revenue. Our strategy also includes applying our patented Coblation technology to a range of other soft-tissue surgical markets, including the products we have introduced in the fields of spinal surgery, neurosurgery, gynecology, urology, cosmetic surgery, ENT surgery, cardiology and general surgery. We cannot be sure that any of our clinical studies in other fields will lead to 510(k) applications or that the applications will be cleared by the FDA on a timely basis, if at all. In addition, we cannot be sure that the products, if cleared for marketing, will ever achieve commercial acceptance.
In May 1998, we announced that we had entered the ear, nose and throat market and had formed a business unit called ENTec to commercialize Coblation technology in this field. In September 1999, we announced that we had entered the spinal surgery market. In February 2000, we announced that we were expanding our marketing efforts for our spinal surgery system to specifically address selected applications in neurosurgery. We are marketing and selling our spinal surgery products through a network of independent distributors and direct sales representatives supported by regional managers worldwide. In January 2004, we acquired MDA and its majority-owned subsidiary, Parallax, a business focused on the treatment of vertebral compression fractures. In November 2004, we acquired Opus Medical, a business focused on soft tissue to bone repair systems, including systems for the treatment of rotator cuff injuries. In August 2005, we completed the purchase of substantially all of the assets of Applied Therapeutics, Inc., or ATI, a maker of sinus surgery treatment products.
We have received 510(k) clearance from the FDA to market several of our products and several of our products are CE marked, which is a requirement to sell our products in most of Western Europe. Specifically, we have received 510(k) clearance from the FDA to market our Coblation-based Arthroscopic Surgery System, or Arthroscopic System, for use in arthroscopic surgery of the knee, shoulder, ankle, elbow, wrist and hip. In addition, our Arthroscopic System is CE marked for use in arthroscopic surgery. We have also received 510(k) clearances in the United States and a CE mark in Europe to market and sell our Coblation-based Spinal Surgery System for spinal surgery and neurosurgery. We have received 510(k) clearance and a CE mark to market our interventional therapy devices for percutaneous, intraoperative and spinal procedures. Our ENT Surgery System has received 510(k) clearances from the FDA and a CE mark for use in general head, neck, oral and sinus surgery procedures, including tonsillectomy and adenoidectomy, turbinate reduction to relieve nasal obstruction, and soft tissue removal to treat snoring. The FDA also has cleared our Cosmetic Surgery System for general dermatologic procedures and skin resurfacing in connection with wrinkle reduction procedures. In addition, the Cosmetic Surgery System has received a CE Mark. We also have received 510(k) clearance from the FDA, and applied for a CE mark, to market products based on our Coblation technology for use in urology, gynecology, plastic and reconstructive surgery, orthopedic surgery and general surgery.
24
Many of ArthroCare’s products are leading edge technologies which have not yet received routine coverage decisions from insurance companies and other payers. As a result, reimbursement may be dependent on case by case decisions from various private third-party payers. In 2007, we established a new subsidiary, Device Reimbursement Services, to provide certain of the devices we sell in exchange for an assignment of the right to bill for such devices. Additionally, in December 2007 we acquired another third-party billing and reimbursement service provider, DiscoCare, Inc. In addition, we have adopted the ArthroCare Corporation health Care Compliance Program, implementing the key elements of a model health care compliance plan developed by the Office of Inspector General of the United States Department of Health and Human Services, or OIG, which oversees compliance with applicable Federal laws in connection with the payment for products by government funded programs.
Critical Accounting Policies and Estimates
The preparation of financial statements and related disclosures in conformity with accounting principles generally accepted in the United States of America requires management to make judgments, assumptions and estimates that affect the amounts reported in the consolidated financial statements and accompanying notes. Note 2 to the consolidated financial statements describes the significant accounting policies and methods used in the preparation of the consolidated financial statements. The most significant areas involving management judgments and estimates are described below and are impacted significantly by judgments, assumptions, and estimates used in the preparation of the consolidated financial statements. Actual results could differ materially from these estimates.
Revenue Recognition
We recognize product revenue in accordance with SEC Staff Accounting Bulletin No. 104, Revenue Recognition in Financial Statements, or SAB 104. Under SAB 104, revenue is recognized when there is persuasive evidence of an arrangement, delivery has occurred or services have been rendered, the sales price is fixed and determinable and collectibility is reasonably assured. Generally, the criteria are met upon shipment of the Company’s products. For amounts due from third-party payors, revenues are recorded at estimated net amounts due for devices utilized with allowances determined using historical collection amounts for each type of payor. Estimated allowances are adjusted to actual allowances as cash is received and claims are reconciled. We evaluate the following criteria in developing the estimated allowance percentages each month: historical allowance trends based on actual claims paid by third-party payors; consideration and analysis of changes in payor mix reimbursement levels; and other issues that may impact allowances. In certain cases, at the customer’s request, we will enter into bill and hold transactions whereby title and risk of loss transfers to the customer, but the product does not ship until a specified later date. Revenue on such transactions is recognized only after ArthroCare has met all conditions for bill and hold transactions set forth in SAB 104, specifically that ArthroCare has no further performance obligations, that the product is complete and ready for shipment, and that a fixed schedule for delivery of the product is set. As of December 31, 2007, 2006 and 2005, respectively, there were no undelivered bill and hold transactions. Revenue is reported net of a provision for estimated product returns.
We recognize license fees and other revenue over the term of the associated agreement unless the fee is in exchange for products delivered or services performed that represent the culmination of a separate earnings process. Royalties are recognized as earned, generally based on the licensees’ product shipments. The items are classified as royalties, fees and other revenues in the accompanying statements of operations. Amounts billed to customers relating to shipping and handling costs have also been classified as royalties, fees and other revenues and related costs are classified as costs of product sales in the accompanying statements of operations.
We maintain an allowance for doubtful accounts receivable based on our assessment of the collectibility of customer accounts. The Company regularly reviews the allowance, considering such factors as historical collection experience, a customer’s current credit-worthiness, customer concentrations, the age of the receivable balances, both individually and in the aggregate, and general economic conditions that may affect a customer’s ability to pay. Actual customer collections could differ from our estimates. We believe that the allowance for doubtful accounts receivable of $4.0 million at December 31, 2007 is adequate to provide for probable losses associated with accounts receivable.
25
Inventory Allowance
Inventory purchases and commitments are based upon future demand forecasts. We record an allowance against our inventory value to the extent we believe that demand for certain inventory items has decreased or if certain inventory items have become obsolete. If there were to be a sudden and significant decrease in demand for our products, or if there were a higher incidence of inventory obsolescence because of rapidly changing technology and customer requirements, we could be required to increase our inventory allowances and our gross margins could be adversely affected.
Business Combinations
Accounting for our business acquisitions requires extensive accounting estimates and judgments to allocate the purchase price between net tangible assets, in-process research and development, other identifiable intangible assets, and goodwill. Assets and liabilities of acquired businesses are recorded under the purchase method of accounting at their estimated fair value at the date of acquisition.
The Company accounts for business combinations in accordance with Statement of Financial Accounting Standards No. 142, Goodwill and Other Intangible Assets, or FAS 142. As of December 31, 2007, we had recorded goodwill of $166.8 million and other intangible assets of $37.7 million, net of accumulated amortization. In response to changes in industry and market conditions, we may be required to strategically realign our resources and consider restructuring, disposing, or otherwise exiting businesses, which could result in an impairment of goodwill or other intangible assets.
We test goodwill for impairment at the reporting unit level at least annually during the fourth quarter of each fiscal year and more frequently if impairment indicators are identified. The first step of the goodwill impairment test is a comparison of the fair value of a reporting unit to its carrying value. We estimate the fair values of each reporting unit using discounted cash flow valuation models. These methods require estimates of our future revenues, profits, capital expenditures, working capital, and other relevant factors. We estimate these amounts by evaluating historical trends, current budgets, operating plans, industry data, and other relevant factors. The estimated fair value of each reporting unit exceeded its respective carrying value in fiscal 2007, indicating the underlying goodwill of each reporting unit was not impaired as of December 31, 2007. Accordingly, we were not required to complete the second step of the goodwill impairment test. The timing and frequency of our goodwill impairment test is based on an ongoing assessment of events and circumstances that would more than likely reduce the fair value of a reporting unit below its carrying value. We will continue to monitor our goodwill balance and conduct formal tests on at least an annual basis or more often when impairment indicators are present. There are various assumptions and estimates underlying the determination of an impairment loss, and estimates using different, but each reasonable, assumptions could produce significantly different results and materially affect the determination of fair value and/or goodwill impairment for each reporting unit. Therefore, the timing and recognition of impairment losses by us in the future, if any, may be highly dependent upon our estimates and assumptions. We believe that the assumptions and estimates utilized were appropriate based on the information available to management.
Intangible assets with finite lives are amortized over the estimated useful life of each asset. We monitor conditions related to these assets to determine whether events and circumstances warrant a revision to the remaining amortization period. Under Statement No. 144, Accounting for the Impairment or Disposal of Long-Lived Assets, intangible assets with finite lives must be tested for potential impairment whenever events or changes in circumstances indicate that the carrying amount may not be recoverable. To date, no events or circumstances have been noted that would indicate the potential for impairment of our intangible assets.
In connection with the ATI acquisition, we made an estimate of $2.4 million related to the valuation of purchased in-process research and development, or IPR&D, projects. Our policy defines IPR&D as the value assigned to those projects which have no alternative future use, including those for which the related products have not reached technological feasibility or have not received regulatory approval. Determining the portion of the purchase price allocated to IPR&D requires significant estimates. The amount of the purchase price allocated to IPR&D is determined by estimating the amount and timing of future cash flows of each project or technology and discounting the net cash flows back to their present values. The discount rate used is determined at the time of acquisition, in accordance with accepted valuation methods, and includes consideration of the assessed risk of the project not being developed to a stage of commercial feasibility.
26
On December 31, 2007, we acquired a third-party billing and reimbursement service provider, DiscoCare, Inc. Since 2006, DiscoCare has been providing surgical devices to facilities, generally at no charge in exchange for the right to bill for such device. ArthroCare paid $24.5 million in cash, net of acquisition costs paid and cash acquired with the potential for future earnouts based on achievement of contractually specified milestones. In accordance with Emerging Issues Task Force 04-01, Accounting for Preexisting Relationships between the Parties to a Business Combination, we evaluated the purchase as a multiple element contract to separately account for settlement of the preexisting relationship and for the acquisition. Based on our analysis, there was no gain or loss required to be recognized for settling the preexisting relationship. Therefore, the entire purchase price was allocated to the net assets of DiscoCare based on fair market value as of the date of acquisition, primarily accounts receivable, inventory, identified intangible assets, and liabilities assumed with the balance applied to goodwill.
Contingencies
We are subject to the possibility of various loss contingencies arising in the ordinary course of business. In determining loss contingencies, we consider the likelihood of loss or impairment of an asset or the incurrence of a liability, as well as our ability to reasonably estimate the amount of loss. An estimated loss contingency is accrued when it is probable that an asset has been impaired or a liability has been incurred and the amount of loss can be reasonably estimated. We regularly evaluate current information available to us to determine whether such accrual estimates should be adjusted.
Income Taxes
We account for income taxes under the liability method, whereby deferred tax asset or liability account balances are determined based on the difference between the financial statement and the tax bases of assets and liabilities using current tax laws and rates in effect for the year in which the differences are expected to affect taxable income. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amounts expected to be realized.
In addition, we make an evaluation at the end of each reporting period as to whether or not some or all of the undistributed earnings of our foreign subsidiaries are permanently reinvested (as that term is defined in generally accepted accounting principles). While we may have concluded in the past that some of such undistributed earnings are permanently reinvested, facts and circumstances can change in the future, and it is possible that a change in facts and circumstances, such as a change in the expectation regarding the capital needs of our foreign subsidiaries, could result in a conclusion that some or all of such undistributed earnings are no longer permanently reinvested. In such an event, we would be required to recognize a deferred income tax liability in an amount equal to the estimated incremental U.S. income tax and withholding tax liability that would be generated if all of such previously-considered permanently reinvested undistributed earnings were distributed to the U.S.
In July 2006, the Financial Accounting Standards Board, or FASB, issued Interpretation No. 48, Accounting for Uncertainty in Income Taxes, or FIN 48. FIN 48 clarifies the accounting for uncertainty in income taxes recognized in an enterprise’s financial statements in accordance with Statement of Financial Accounting Standards No. 109, Accounting for Income Taxes. This interpretation prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. FIN 48 also provides guidance on derecognition of tax benefits, classification on the balance sheet, interest and penalties, accounting in interim periods, disclosure, and transition. We adopted FIN 48 effective January 1, 2007. In accordance with FIN 48, paragraph 19, we have decided to classify interest and penalties as a component of tax expense. As a result of the implementation of FIN 48, we recognized a $2.1 million increase in liability for unrecognized tax benefits, which was accounted for as a reduction to the January 1, 2007 balance of retained earnings.
As of December 31, 2007, we had unrecognized tax benefits of approximately $8.4 million, of which $7.6 million, if recognized, would result in a reduction of our effective tax rate. The Company recorded an increase in its unrecognized tax benefits of approximately $0.5 million for the year ended December 31, 2007. Interest and penalties were immaterial as of December 31, 2007 and are included in the unrecognized tax benefits. The Company is subject to audit by the Internal Revenue Service and California Franchise Tax Board for the years 1994 through 2007 and by the Texas State Comptroller for the years 2006 and 2007. As the Company has operations in most other U.S. states, other state tax authorities may assess deficiencies related to prior year activities; however, the years open to assessment vary with each state. The Company also files income tax returns for non-U.S. jurisdictions; the most significant of which are the UK, Sweden, and Germany. The years open to adjustment for the UK and Germany are 2003 through 2007. The years open to adjustment for Sweden are 1999 through 2007.
27
Stock-Based Compensation
On January 1, 2006, we adopted Statement of Financial Accounting Standards No. 123 (revised 2004), Share-Based Payment, or FAS 123R, which requires the measurement and recognition of compensation expense for all share-based payment awards made to employees, including employee stock options, restricted stock awards and employee stock purchases related to the Employee Stock Purchase Plan, or ESPP, based on estimated grant date fair values. Stock-based compensation expense recognized under FAS 123R for the years ended December 31, 2007 and 2006 was $10.2 million and $9.8 million, respectively. Stock-based compensation expense relating to restricted stock awards and non-employee stock options for the year ended December 31, 2005 was $1.4 million. There was no stock-based compensation expense related to employee stock options recognized during the year ended December 31, 2005. See Note 11, “Stockholders’ Equity,” in the notes to the consolidated financial statements for additional information.
Upon adoption of FAS 123R, we began estimating the value of employee stock options on the date of grant using the Black Scholes model, as we had previously used for the purpose of disclosing pro forma financial information. The determination of the fair value of share based payment awards on the date of grant using an option pricing model is affected by our stock price as well as assumptions regarding a number of highly complex and subjective variables. These variables include, but are not limited to, the expected stock price volatility and employee stock option exercise behavior.
The weighted average fair value of stock awards, excluding restricted stock, granted for the year ended December 31, 2007 was $12.21 per share and $6.20 per share for the ESPP, using the Black Scholes model with the following weighted average assumptions:
Year ended December 31, 2007 | |||||||
Options | ESPP | ||||||
| Expected term (in years) (1) | 3.8 | 0.5 | |||||
| Expected volatility (2) | 33 | % | 35 | % | |||
| Risk-free interest rate (3) | 4.7 | % | 4.8 | % | |||
| Expected dividends | - | - | |||||
| (1) | The expected term assumption for options was determined based on historical data, adjusted for the recent reduction of the contractual life for options from 10 to seven years. |
| (2) | The expected volatility was determined using a blend of implied volatility and historical volatility over the expected term, which we consider a better indictor of expected volatility than using only historical volatility. |
| (3) | The risk-free interest rate is based upon observed interest rates appropriate for the term of the Company’s awards. |
As stock-based compensation expense recognized in the consolidated statement of operations for 2007 is based on awards ultimately expected to vest, it has been reduced for estimated forfeitures at a rate of 6.6 percent. FAS 123R requires forfeitures to be estimated at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. Forfeitures were estimated based on historical experience but actual forfeitures could differ materially and result in volatility in our stock-based compensation expense amounts.
28
Overview of 2007 Financial Results
Total revenue for the year ended December 31, 2007 was $319.2 million, an increase of 21.4 percent compared to the year-ended December 31, 2006. The increase was driven by the combined performance of all business units. International product sales represented approximately 20.9 percent of net product sales for the year. No single customer accounted for more than 10 percent of our net product sales for the year ended December 31, 2007.
Total operating expenses were approximately $181.3 million for 2007, compared to $143.7 million in 2006. This $37.6 million increase was driven by a combination of factors: approximately $15.4 million represents increased salaries, commissions and other employee-related expenses due to increased headcount, $12.6 million relates to additional commissions and fees related to increased sales volume and new sales agency agreements, $5.5 million is attributable to increased clinical testing, marketing and promotions related to new product lines, and $1.6 million relates to a net increase in legal, accounting and other outside consulting costs.
Our cash, cash equivalents and short-term investments balance at December 31, 2007 was $43.3 million, an increase of approximately $12.5 million from December 31, 2006.
Results of Operations | ||||||||||||||||||||
Year Ended December 31, | ||||||||||||||||||||
| (Dollar amounts in thousands) | 2007 | 2006 | 2005 | |||||||||||||||||
| Revenues: | ||||||||||||||||||||
| Product sales | $ | 307,596 | 96.4 | % | $ | 253,376 | 96.3 | % | $ | 206,533 | 96.4 | % | ||||||||
| Royalties, fees and other | 11,646 | 3.6 | % | 9,625 | 3.7 | % | 7,801 | 3.6 | % | |||||||||||
| Total revenues | 319,242 | 100.0 | % | 263,001 | 100.0 | % | 214,334 | 100.0 | % | |||||||||||
| Cost of product sales | 84,724 | 26.5 | % | 76,838 | 29.2 | % | 64,206 | 30.0 | % | |||||||||||
| Gross profit | 234,518 | 73.5 | % | 186,163 | 70.8 | % | 150,128 | 70.0 | % | |||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 26,886 | 8.4 | % | 23,247 | 8.9 | % | 21,015 | 9.8 | % | |||||||||||
| Sales and marketing | 123,213 | 38.6 | % | 91,915 | 35.0 | % | 75,302 | 35.1 | % | |||||||||||
| General and administrative | 24,033 | 7.5 | % | 21,355 | 8.1 | % | 12,202 | 5.7 | % | |||||||||||
| Amortization of intangible assets | 7,211 | 2.3 | % | 7,176 | 2.7 | % | 6,150 | 2.9 | % | |||||||||||
| Acquired in-process research and development costs | — | 0.0 | % | — | 0.0 | % | 2,400 | 1.1 | % | |||||||||||
| Total operating expenses | 181,343 | 56.8 | % | 143,693 | 54.7 | % | 117,069 | 54.6 | % | |||||||||||
| Income from operations | 53,175 | 16.7 | % | 42,470 | 16.1 | % | 33,059 | 15.4 | % | |||||||||||
| Interest and other income (expense), net | 2,184 | 0.6 | % | (1,809 | ) | (0.7 | %) | (2,336 | ) | (1.1 | %) | |||||||||
| Income before income taxes | 55,359 | 17.3 | % | 40,661 | 15.4 | % | 30,723 | 14.3 | % | |||||||||||
| Income tax provision | 12,179 | 3.8 | % | 8,986 | 3.4 | % | 7,193 | 3.3 | % | |||||||||||
| Net income | $ | 43,180 | 13.5 | % | $ | 31,675 | 12.0 | % | $ | 23,530 | 11.0 | % | ||||||||
29
Revenues
Product sales consist principally of sales of disposable devices. Product sales for fiscal 2007 were $307.6 million compared to $253.4 million and $206.5 million in fiscal 2006 and 2005, respectively. Product sales by product market for the periods shown were as follows (dollar amounts in thousands):
Year Ended December 31, | |||||||||||||||||||||
2007 | 2006 | 2005 | |||||||||||||||||||
| Sports Medicine | $ | 189,588 | 61.6 | % | $ | 166,665 | 65.8 | % | $ | 139,868 | 67.7 | % | |||||||||
| ENT | 73,065 | 23.8 | % | 59,886 | 23.6 | % | 43,546 | 21.1 | % | ||||||||||||
| Spine | 44,653 | 14.5 | % | 26,635 | 10.5 | % | 23,110 | 11.2 | % | ||||||||||||
| Coblation Technology | 290 | 0.1 | % | 190 | 0.1 | % | 9 | 0.0 | % | ||||||||||||
| Total Product Sales | $ | 307,596 | 100.0 | % | $ | 253,376 | 100.0 | % | $ | 206,533 | 100.0 | % | |||||||||
Product sales by geography for the periods shown were as follows (dollar amounts in thousands):
Year Ended December 31, | |||||||||||||||||||||
2007 | 2006 | 2005 | |||||||||||||||||||
| Americas | $ | 243,209 | 79.1 | % | $ | 202,333 | 79.9 | % | $ | 164,072 | 79.4 | % | |||||||||
| United Kingdom | 17,561 | 5.7 | % | 12,478 | 4.9 | % | 11,863 | 5.7 | % | ||||||||||||
| Germany | 14,179 | 4.6 | % | 11,405 | 4.5 | % | 9,007 | 4.4 | % | ||||||||||||
| Rest of World | 32,647 | 10.6 | % | 27,160 | 10.7 | % | 21,591 | 10.5 | % | ||||||||||||
| Total Product Sales | $ | 307,596 | 100.0 | % | $ | 253,376 | 100.0 | % | $ | 206,533 | 100.0 | % | |||||||||
Our sustained increase in direct sales presence has continued to have a positive effect on product sales since inception, as has the execution of our strategic plan to build market share through promotional programs of controller placements, commercialization of our technology in fields outside of arthroscopy, and the introduction of new products designed to address surgical procedures that have traditionally been difficult to perform. Additionally, our acquisitions of Atlantech, Parallax, ATI and Opus Medical product lines have continued to have a positive effect on sales growth. We anticipate that disposable device sales will remain a key component of our product sales in the near future.
Based upon the estimated number of arthroscopic procedures performed each year, we believe that knee procedures represent the largest segment of the arthroscopic market, while shoulder procedures represent the fastest growing segment. To achieve increasing disposable device sales in arthroscopy over time, we believe we must continue to penetrate the market in knee procedures, expand physicians’ education with respect to Coblation technology, and continue to work on new product development efforts specifically for knee applications. We believe that, in our more than ten years of product shipments, we have penetrated 30 to 35 percent of the facilities that perform arthroscopic procedures in the United States. We believe that approximately 45 percent of our arthroscopy product sales are being generated by the sale of disposables for use in knee procedures. We expect our sales to continue to be strong in 2008, as we continue to increase our presence in Europe and the South Pacific and benefit from the integration of our acquisitions.
Royalties, fees, and other revenues consist mainly of revenue from the licensing of our products and technology, plus shipping and handling costs billed to customers. Royalties, fees, and other revenues increased to $11.6 million in 2007 from $9.6 million and $7.8 million in 2006 and 2005, respectively, as a result of increased sales of our products and growth in the markets for which we receive royalties.
Cost of Product Sales
Cost of product sales consist of manufacturing costs, material costs, labor costs, manufacturing overhead, warranty and other direct product costs. Additionally, cost of product sales includes amortization of controller unit placements under a program whereby we maintain ownership of controller units shipped to customers, with the costs being capitalized and amortized into cost of product sales over the useful life of the controller unit.
Cost of product sales was $84.7 million, or 26.5 percent of total revenues, for 2007, compared to $76.8 million, or 29.2 percent of total revenues, in 2006 and $64.2 million, or 30.0 percent of total revenues, in 2005. Gross product margin as a percentage of sales increased to 72.5 percent in 2007 from 69.7 percent in 2006 and 68.9 percent in 2005. The increase in gross product margin percentage for the year ended December 31, 2007 compared to the same period in 2006 is primarily a result of increased manufacturing efficiencies, a rise in average selling prices, and increased sales volume associated with our higher gross margin products. The favorable increase in gross product margin percentage in 2006 as compared to 2005 is primarily due to an increase in average selling prices accompanied by a decrease in component costs.
30
Amortization of controllers was $7.4 million in 2007, compared to $6.6 million and $5.8 million in 2006 and 2005, respectively. Controller amortization represented 2.4 percent of product sales in 2007. This is compared to 2006 and 2005, when controller amortization represented 2.6 percent and 2.8 percent of product sales, respectively. The decrease as a percent of cost of sales for 2007 is due to lower controller placements with existing customers in anticipation of our new controller launched in the first quarter of 2008. Cost of product sales for the year ended December 31, 2007 includes $0.7 million of stock-based compensation expense related to employee stock options. In 2006, cost of product sales included $0.6 million of stock-based compensation expense.
We expect gross margin to continue to increase as we continue to increase our average selling prices and improve operating efficiencies.
Operating Expenses
Research and development expense increased in 2007 to $26.9 million, or 8.4 percent of total revenues, from $23.2 million, or 8.9 percent of total revenues, in 2006 and $21.0 million, or 9.8 percent of total revenues, in 2005. The absolute dollar increase in research and development expenses in 2007 compared to 2006 is primarily attributable to $1.9 million in increased clinical testing supplies and services associated with our new products and $1.5 million in additional employee compensation due to increased research and development headcount to support new product development initiatives. The increase in research and development costs in 2006 as compared to 2005 is primarily due to $1.3 million in increased stock compensation expense due to our adoption of FAS 123R, along with a continued investment in our Coblation-based products and in the MDA product line, resulting in an increase of $0.6 million in consulting and outside service costs and $0.2 million in patent-related and product development charges.
We expect research and development expenses to increase through continued expenditures on new product development, regulatory affairs, clinical studies and patents. We expect these expenses as a percentage of product sales to remain essentially flat.
Sales and marketing expense increased to $123.2 million, or 38.6 percent of total revenues, in 2007 from $91.9 million, or 35.0 percent of total revenues, in 2006, and from $75.3 million, or 35.1 percent of total revenues, in 2005. The increase for 2007 compared to 2006 related primarily to $12.6 million in additional commissions and fees due to increased sales volume and new sales agency agreements, $7.8 million in additional salaries and employee-related expenses and $5.2 million in additional commissions associated with increased sales and marketing headcount, and $3.6 million in additional marketing costs related to trade shows, samples and demonstrations to promote new product lines. Increased expenses in 2006 as compared to 2005 related primarily to $6.7 million in training and promotion costs, $4.4 million in additional compensation and related expenses associated with our increased direct sales force, $2.7 million in stock-based compensation expense associated with our adoption of FAS 123R, and $2.7 million in settlement fees associated with the Settlement and License Agreement between ArthroCare and MarcTec, LLC (formerly Bonutti IP, LLC).
We anticipate that sales and marketing spending will continue to increase as a result of the expansion of our distribution capabilities to address the spinal surgery and ear, nose and throat markets, higher dealer commissions from increased sales, the additional cost of penetrating international markets, higher promotional, demonstration and sample expenses, and additional investments in the sales, marketing and support staff necessary to commercialize and market future products. This increase will be partially offset by a reduction in service fees previously paid to DiscoCare which we acquired on December 31, 2007.
General and administrative expense increased in absolute dollars to $24.0 million, or 7.5 percent of total revenues, in 2007, from $21.4 million, or 8.1 percent of total revenues, in 2006 and $12.2 million, or 5.7 percent of total revenues, in 2005. The absolute dollar increase is primarily due to a $1.6 million net increase in legal, accounting and other outside service costs and a $0.9 million increase in employee-related expenses associated with increased headcount in administrative functions. The increase in general and administrative expenses in 2006 as compared to 2005 is primarily due to a net increase of $3.5 million in legal, accounting and other outside services costs, $3.3 million of stock-based compensation expense associated with our adoption of FAS 123R, and $1.6 million of additional compensation and related expenses associated with increasing our headcount in our accounting and other administrative functions.
31
We expect that general and administrative expenses will remain essentially flat as a percentage of total revenues during 2008 due to continued investments in our infrastructure to support further revenue growth.
Amortization of intangible assets was $7.2 million, or 2.3 percent of total revenues, in 2007, compared with $7.2 million, or 2.7 percent of total revenues, in 2006, and $6.2 million, or 2.9 percent of total revenues, in 2005. The decrease as a percentage of total revenues over the last two years is primarily due to fewer acquisitions of intangible assets during periods of increasing revenues each year since 2005.
Acquired in-process research and development costs (IPR&D) were not incurred during the years ended December 31, 2007 or 2006. During the third quarter of 2005 we incurred $2.4 million of acquired in-process research and development costs in connection with the research and development efforts acquired in our acquisition of substantially all of the assets of ATI. Prior to the acquisition, we did not have a comparable product under development. This expense related to the development of technology for arresting bleeding that had not yet reached technological feasibility at ATI and had no alternative future use. As of December 31, 2005, the research and development of this technology was completed and the Stammberger Foam product, which utilizes this technology, was commercialized for use in ENT procedures.
Interest and Other Income (Expense), Net
Interest and other income (expense), net was a net benefit of $2.2 million in 2007 compared to a net expense of $1.8 million in 2006 and a net expense of $2.3 million in 2005. The $4.0 million increase in net interest income in 2007 is primarily attributable to the funding of our operations through operating cash flows for the majority of 2007 as opposed to borrowing against our credit facility. The $0.5 million decrease in expense in 2006 was primarily attributable to foreign exchange gains in 2006, whereas in 2005 we had foreign exchange losses. We expect to see a net interest expense in 2008 as a result of borrowing $60.0 million against our credit facility in December 2007.
Income Tax Provision
The provision for income taxes was $12.2 million for 2007, compared to $9.0 million for 2006 and $7.2 million for 2005. The effective tax rate was 22.0 percent in 2007, 22.1 percent in 2006 and 23.4 percent in 2005. The yearly improvement in our tax rate from 2005 to 2007 is primarily due to the continued expansion of business activities in our Costa Rica operations and tax credits for research and development activities.
Liquidity and Capital Resources
As of December 31, 2007, we had $148.3 million in working capital, compared to $113.7 million at December 31, 2006. Our principal sources of liquidity consisted of $43.3 million in cash, cash equivalents, and short-term investments at December 31, 2007. Cash equivalents are highly liquid with original maturities of ninety days or less. Our short-term investments consist primarily of corporate bonds.
Cash generated by operating activities in 2007 was $52.7 million, as compared to $43.8 million in 2006 and $37.2 million in 2005. The cash generated in 2007 was primarily attributable to $43.2 million of net income, $21.2 million of depreciation and amortization and $10.2 million of stock based compensation, partially offset by $23.8 million in increased accounts receivable and $6.2 million in increased inventory. The cash generated in 2006 was primarily attributable to $31.7 million of net income and non-cash items such as $21.3 million of depreciation and amortization and stock-based compensation of $9.8 million, partially offset by a $17.2 million increase in accounts receivable and $8.6 million in increased inventory. The cash generated in 2005 was primarily attributable to $23.5 million in net income adjusted for non-cash items, including $16.8 million of depreciation and amortization and $9.6 million in income tax benefits related to employee stock options, partially offset by increases in accounts receivable and inventories of $14.8 million and $7.7 million, respectively.
32
Cash used in investing activities was $49.9 million in 2007, as compared to $86.5 million in 2006 and $16.4 million in 2005. Cash used in investing activities in 2007 was primarily due to $26.5 million for purchases of intangible assets and other purchase business combinations, $20.2 million for purchases of property and equipment, and $14.8 million in payments to ATI shareholders in satisfaction of acquisition-related contractual contingencies, partially offset by $12.8 million in sales of available-for-sale securities. Cash used in investing activities in 2006 was primarily due to a $55.2 million payment to former Opus Medical shareholders, $17.2 million for the purchase of property and equipment, and $26.9 million for the purchase of available-for-sale securities, partially offset by $15.0 million of sales of available-for-sale securities. Cash used in investing activities in 2005 was primarily due to the $10.0 million payment made for the purchase of ATI’s assets in August 2005, accompanied by $9.6 million associated with the capitalization of controllers and $3.7 million in fixed asset investments, offset by net sales of $7.4 million in available for sale securities.
The fair value of our investments in marketable securities at December 31, 2007 was $3.9 million. Investments with maturities beyond one year may be classified as short-term based on their highly liquid nature and because such marketable securities represent the investment of cash that is available for current operations. Our investment policy is to manage our investment portfolio to preserve principal and liquidity while maximizing the return on the investment portfolio through the full investment of available funds.
Cash provided by financing activities was $22.8 million in 2007, as compared to $37.7 million in 2006 and $11.2 million in cash used in financing activities in 2005. Cash provided by financing activities in 2007 was due primarily to $60.0 million in proceeds from borrowings against our revolving line of credit, $14.3 million of proceeds from stock option purchases, and $7.8 million in tax benefits related to stock option expenses, partially offset by $59.6 million used for the purchase of shares of our common stock on the open market. Cash provided by financing activities in 2006 was primarily due to $28.1 million of proceeds from stock option purchases and a $9.5 million tax benefit related to the stock option expenses. Cash used in financing activities in 2005 was primarily due to $28.9 million of loan repayments, partially offset by $16.5 million of proceeds from stock option exercises.
On January 13, 2006, the Company entered into a Revolving Credit Agreement (the “Credit Agreement”) with a syndicate of banks (collectively, the “Lenders”). Under the terms of the Credit Agreement, we may borrow up to $100.0 million under a revolving line of credit from the named Lenders at the lead Lender’s Prime Rate or the British Bankers Association LIBOR Rate, plus a spread. The spread over the Prime Rate or LIBOR Rate is determined by the Company’s leverage ratio, as defined in the Credit Agreement. In December 2007, in connection with our purchase of shares of our common stock on the open market, we borrowed $60.0 million against the line of credit. At December 31, 2007, we had $60.0 million in outstanding balances under the Credit Agreement and the weighted average interest rate applicable to borrowings under the Credit Agreement was 5.5 percent.
The Credit Agreement has a five-year maturity and includes additional terms under which we may request an increase of up to $75.0 million in commitments from the Lenders, as business needs dictate. The Credit Agreement also contains various covenants that specify minimum or maximum financial ratios. We were in compliance with all such covenants at December 31, 2007.
33
Disclosures about Contractual Obligations and Commercial Commitments
Our cash flows from operations are dependent on a number of factors, including fluctuations in our operating results, accounts receivable collections, inventory management, expensing of stock options, and the timing of tax and other payments. As a result, the impact of contractual obligations on our liquidity and capital resources in future periods should be analyzed in conjunction with such factors. The following table aggregates all material contractual obligations and commercial commitments that affected our financial condition and liquidity as of December 31, 2007:
Payments Due by Period (in thousands) | ||||||||||||||||
Total | Less than 1 Year | 1-3 Years | 4-5 Years | After 5 Years | ||||||||||||
| Long-term debt obligations | $ | 60,000 | $ | — | $ | — | $ | 60,000 | $ | — | ||||||
| Operating lease obligations | 15,368 | 3,158 | 4,651 | 3,582 | 3,977 | |||||||||||
| Purchase commitments with suppliers (1) | 12,169 | 6,297 | 4,088 | 1,784 | — | |||||||||||
| Total | $ | 87,537 | $ | 9,455 | $ | 8,739 | $ | 65,366 | $ | 3,977 | ||||||
(1) Represents agreements to purchase products that are enforceable, legally binding and specify terms, including: fixed or minimum quantities to be purchased and the approximate timing of the payments.
We believe that the above-described credit and term facilities, in addition to cash generated from operations, as well as our existing cash balances and short-term investments, will be sufficient to fund our operations through fiscal year 2008 and in the near future. Our future liquidity and capital requirements will depend on numerous factors, including our success in commercializing our products, development and commercialization of products in fields other than arthroscopy, the ability of our suppliers to continue to meet our demands at current prices, obtaining and enforcing patents important to our business, the status of regulatory approvals and competition.
Recently Issued Accounting Pronouncements
In September 2006, the FASB issued Statement of Financial Accounting Standards No. 157, Fair Value Measurements, or FAS 157. FAS 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles, and expands disclosures about fair value measurements. FAS 157 is effective for us beginning January 1, 2008 for financial assets and liabilities and January 1, 2009 for non-financial assets and liabilities. We do not believe that FAS 157 will have a material effect on our operating results or financial position.
In February 2007, the FASB issued Statement of Financial Accounting Standards No. 159, The Fair Value Option for Financial Assets and Financial Liabilities - Including an amendment of FASB No. 115, or FAS 159. FAS 159 permits companies to choose to measure many financial instruments and certain other items at fair value in order to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without having to apply complex hedge accounting provisions. FAS 159 is effective for us beginning January 1, 2008. We are currently evaluating the impact of FAS 159 on our operating results and financial position. We do not believe that FAS 159 will have a material effect on our operating results or financial position.
In December 2007, the FASB issued Statement of Financial Accounting Standards No. 141(R), Business Combination, or FAS 141R. FAS 141R is effective for us as of January 1, 2009 and is applied prospectively for business combinations completed after January 1, 2009. Under FAS 141, organizations utilized the announcement date as the measurement date for the purchase price of the acquired entity. FAS 141R requires measurement at the date the acquirer obtains control of the acquiree, generally referred to as the acquisition date. FAS 141R will have a significant impact on the accounting for transaction costs, restructuring costs as well as the initial recognition of contingent assets and liabilities assumed during a business combination completed after January 1, 2009.
34
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Our exposure to market risk for changes in interest rates relates primarily to our investment portfolio, including cash and cash equivalents. We do not use derivative financial instruments in our investment portfolio.
We are subject to fluctuating interest rates that may impact, adversely or otherwise, our results of operations or cash flows for our cash and cash equivalents.
The table below presents principal amounts and related weighted average interest rates as of December 31, 2007 for our cash, cash equivalents, and short-term investments (in thousands):
| Cash, cash equivalents, and short-term investments | $ | 43,250 | ||
| Average interest rate | 3.9 | % | ||
Although payments under the operating leases for our facilities are tied to market indices, we are not exposed to material interest rate risk associated with operating leases.
Borrowings under our Credit Agreement incur interest based on current market interest rates. To the extent that these rates fluctuate, our results of operations and cash flows, as well as our ability to borrow needed capital at a critical time, could be significantly affected.
Foreign Currency Risk
A significant portion of our international sales and operating expenses are denominated in currencies other than the U.S. Dollar. In 2007, most of these currencies strengthened relative to the U.S. Dollar. To the extent that the exchange rates for these currencies fluctuate against the U.S. Dollar, we will experience variations in our results of operations and financial condition.
Our cash and cash equivalents in 2007 are denominated primarily in U.S. Dollars; however, we also maintain significant balances in Euros, British Pounds, Swedish Krona, Australian Dollars and Costa Rican Colones. A 10 percent change in the December 31, 2007 exchange rates for these currencies would have an impact on pre-tax income of approximately $0.8 million.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
Certain information required by this Item is included in Item 6 of Part II of this Report and is incorporated herein by reference. All other information required by this Item is included in Item 15 of this Report and is incorporated herein by reference.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
35
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) adopted by the SEC under the Securities Exchange Act of 1934, as amended (the “Exchange Act”)) that are designed to ensure that information required to be disclosed in our filings under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to management, including our Chief Executive Officer, or CEO, and Chief Financial Officer, or CFO, as appropriate, to allow timely decisions regarding required disclosure.
We carried out an evaluation under the supervision and with the participation of our management, including our CEO and CFO, of the effectiveness of the design and operation of our disclosure controls and procedures as of December 31, 2007. Based upon this evaluation, our CEO and CFO have concluded that the design and operation of our disclosure controls and procedures were effective as of December 31, 2007. Accordingly, management believes that the financial statements included in this report fairly present in all material respects our financial condition, results of operations and cash flows for the periods presented.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act and for our assessment of the effectiveness of internal control over financial reporting. Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness of internal control over financial reporting to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Management, including our CEO and CFO, has assessed the effectiveness of our internal control over financial reporting as of December 31, 2007. In making its assessment of internal control over financial reporting, management used the criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission, or COSO. This assessment included an evaluation of the design of our internal control over financial reporting and testing of the operational effectiveness of those controls. Based on the results of this assessment, management has concluded that our internal control over financial reporting was effective as of December 31, 2007.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting during the quarter ended December 31, 2007 that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
None.
36
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
We have adopted a code of business conduct and ethics, or code of conduct, containing general guidelines for conducting our business consistent with the highest standards of business ethics. The code of conduct is designed to qualify as a “code of ethics” within the meaning of Section 406 of the Sarbanes-Oxley Act of 2002 and the rules promulgated there under as well as under applicable rules of the NASDAQ Stock Exchange. Our code of conduct is available on the Investor Relations section of our website (www.arthrocare.com). To the extent required by law or the rules of the NASDAQ Stock Exchange, any amendments to, or waivers from, any provision of the code will be promptly disclosed publicly. To the extent permitted by such requirements, we intend to make such public disclosure by posting the relevant material on the Investor Relations section of our website in accordance with SEC rules.
All additional information required by this item is incorporated by reference to our definitive Proxy Statement to be filed pursuant to Regulation 14A under the Exchange Act of 1934, as amended, in connection with our annual meeting of stockholders.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this item is incorporated by reference into our definitive Proxy Statement to be filed pursuant to Regulation 14A under the Exchange Act in connection with our annual meeting of stockholders.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
Except as set forth below regarding securities authorized for issuance under equity compensation plans, the information required by this item is incorporated by reference into our definitive Proxy Statement to be filed pursuant to Regulation 14A under the Exchange Act in connection with our annual meeting of stockholders.
EQUITY COMPENSATION PLAN INFORMATION
The following table provides information as of December 31, 2007 regarding the common stock that may be issued upon the exercise of options, warrants and rights under our existing equity compensation plans.
Plan category | Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) | Weighted-average exercise price of outstanding options, warrants and rights (b) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) | |||||||
Equity compensation plans approved by security holders (1) | 2,249,851 | $ | 29.11 | 804,001 | ||||||
Equity compensation plans not approved by security holders (2) | 745,394 | $ | 27.79 | 19,906 | ||||||
Total | 2,995,245 | $ | 28.78 | 823,907 | ||||||
| (1) | Includes 72,756 shares remaining available for future issuance under the Company’s Employee Stock Purchase Plan. |
| (2) | Consists of shares issuable under our Amended and Restated Non-statutory Option Plan, which does not require the approval of, and has not been approved by, our stockholders. |
37
AMENDED AND RESTATED NONSTATUTORY OPTION PLAN
The Company’s Amended and Restated Nonstatutory Option Plan (the “NSO Plan”) provides for the grant of options to employees and consultants of the Company. The Board of Directors of the Company initially adopted the NSO Plan in May 1999. As of December 31, 2007, a total of 3,550,000 shares of common stock are reserved for issuance under the NSO Plan, options for 893,295 were outstanding under the NSO Plan, and 19,906 shares remained available for future grants. The NSO Plan is not required to be and has not been approved by the Company’s stockholders.
Options granted under the NSO Plan will be non-statutory stock options. Incentive stock options, within the meaning of Section 422 of the Code, may not be granted under the NSO Plan. The NSO Plan also authorizes the grant of stock purchase rights to our employees and consultants.
Eligibility; Administration. The NSO Plan provides that awards may be granted to employees and consultants of the Company or any parent or subsidiary of the Company. However, officers and directors of the Company are not eligible to receive awards under the NSO Plan. As of December 31, 2007, approximately 1,074 people were eligible to participate in the NSO Plan. The NSO Plan may be administered by the Board or Board Committees (the “Administrator”). The Administrator of the NSO Plan will have full power to select, from among the employees and consultants of the Company eligible for awards, the individuals to whom awards will be granted, to make any combination of awards to any participant and to determine the specific terms of each grant, subject to the provisions of the NSO Plan. The interpretation and construction of any provision of the NSO Plan by the Administrator will be final and conclusive.
Exercise Price. The exercise price of options granted under the NSO Plan is determined by the Administrator. Non-statutory options may be granted with a per share exercise price below the fair market value per share of the common stock at the time of grant.
Exercisability. The Administrator has discretion in determining the vesting schedule for each option granted. Options granted to new optionees under the NSO Plan generally become exercisable starting one year after the date of grant with 25 percent of the shares covered thereby becoming exercisable at that time and with an additional 1/48 of the total number of options becoming exercisable at the beginning of each full month thereafter, with full vesting occurring on the fourth anniversary of the date of grant. The term of an option will be determined by the Administrator.
Exercisability Following Termination of Employment. If an optionee’s employment or consulting relationship terminates for any reason (other than death or disability), then all options held by the optionee under the NSO Plan expire on the earlier of (1) the date set forth in his or her notice of grant, or (2) the expiration date of such option. In the absence of a specified time in the notice of grant, an option will remain exercisable for 90 days following an optionee’s termination (other than for death or disability). To the extent the option is exercisable at the time of the optionee’s termination, the optionee may exercise all or part of his or her option at any time before it expires.
Exercisability Following Disability. If an optionee’s employment or consulting relationship terminates as a result of disability, then all options held by such optionee under the NSO Plan expire on the earlier of (1) 12 months from the date of such termination or (2) the expiration date of such option. The optionee (or the optionee’s estate or a person who has acquired the right to exercise the option by bequest or inheritance) may exercise all or part of the option at any time before such expiration to the extent that the option was exercisable at the time of such termination.
Exercisability Following Death. In the event of an optionee’s death, the option may be exercised at any time within 12 months of the date of death (but no later than the expiration date of such option) by the optionee’s estate or a person who has acquired the right to exercise the option by bequest or inheritance, but only to the extent that the option was exercisable at the time of such death.
Stock Purchase Rights. The NSO Plan permits the Company to grant rights to purchase common stock. After the Administrator determines that it will offer stock purchase rights under the NSO Plan, it will advise the offeree in writing or electronically of the terms, conditions and restrictions related to the offer, including the number of shares that the offeree will be entitled to purchase, and the time within which the offeree must accept such offer. The Administrator will establish the purchase price, if any, and form of payment for each stock purchase right. The offer will be accepted by execution of a stock purchase agreement or a stock bonus agreement in the form determined by the Administrator.
38
Unless the Administrator determines otherwise, the stock purchase agreement or a stock bonus agreement will grant the Company a repurchase option exercisable upon the voluntary or involuntary termination of the purchaser’s employment with the Company for any reason. The purchase price for shares repurchased pursuant to the stock purchase agreement or a stock bonus agreement will be the original price paid by the purchaser and may be paid by cancellation of any indebtedness of the purchaser to the Company. The repurchase option will lapse at such rate as the Administrator may determine.
Transferability of Awards. No award may be transferred by the holder other than by will or the laws of descent or distribution. Each award may be exercised, during the lifetime of the holder only by such holder.
Adjustments Upon Changes in Common Stock. If any change is made in the common stock subject to the NSO Plan, without receipt of consideration by the Company (through merger, consolidation, reorganization, recapitalization, stock dividend, dividend in property other than cash, stock split, liquidating dividend, combination of shares, exchange of shares, change in corporate structure or otherwise), the number of shares reserved for issuance under the NSO Plan and awards outstanding thereunder and the class, number of shares and price per share of common stock subject to outstanding awards will be appropriately adjusted. The conversion of convertible securities of the Company will not be deemed to have been “effected without receipt of consideration.”
In the event of the dissolution or liquidation of the Company, each outstanding award will terminate immediately prior to the consummation of such proposed action. The Administrator may provide that the vesting of outstanding awards will be accelerated and such awards will be fully exercisable prior to such transaction.
In the event of a merger of the Company with or into another corporation or the sale of all or substantially all of the assets of the Company, then outstanding options will be assumed or substituted for by the acquiring company or one of its affiliates. In the absence of or in lieu of any such assumption or substitution, the Administrator may provide that the vesting of outstanding awards will be accelerated and such awards will be fully exercisable for a period of at least 15 days prior to such transaction. Awards that are not assumed or substituted for and accelerated options not exercised before the closing of such a transaction will terminate automatically at the closing of the transaction.
Amendment and Termination of the NSO Plan. The Board may amend or terminate the NSO Plan at any time. However, no action by the Board may alter or impair any option previously granted under the NSO Plan. The Administrator may accelerate any option or waive any condition or restriction pertaining to such option at any time. Any options outstanding under the Plan at the time of its termination will remain outstanding until they expire by their terms.
FEDERAL INCOME TAX CONSEQUENCES OF NSO PLAN
Non-Statutory Stock Options. An optionee does not recognize any taxable income at the time he or she is granted a non-statutory stock option. Upon exercise, the optionee recognizes taxable income generally measured by the excess of the current fair market value of the shares over the exercise price. Any taxable income recognized in connection with an option exercise by an employee of the Company is subject to tax withholding by the Company. The Company is generally entitled to a deduction in the same amount as the ordinary income recognized by the optionee. Upon a disposition of such shares by the optionee, any difference between the sale price and the optionee’s exercise price, to the extent not recognized as taxable income as provided above, is treated as long-term or short-term capital gain or loss, depending on the holding period.
Stock Purchase Rights. For federal income tax purposes, if an individual is granted a stock purchase right, the recipient generally will recognize taxable ordinary income equal to the excess of the common stock’s fair market value over the purchase price, if any. However, to the extent the common stock is subject to certain types of restrictions, such as a repurchase right in favor of the Company, the taxable event will be delayed until the vesting restrictions lapse unless the recipient makes a valid election under Section 83(b) of the Code. If the recipient makes a valid election under Section 83(b) of the Code with respect to restricted stock, the recipient generally will recognize ordinary income at the date of acquisition of the restricted stock in an amount equal to the difference, if any, between the fair market value of the shares at that date over the purchase price for the restricted stock. If, however, a valid Section 83(b) election is not made by the recipient, the recipient will generally recognize ordinary income when the restrictions on the shares of restricted stock lapse, in an amount equal to the difference between the fair market value of the shares at the date such restrictions lapse over the purchase price for the restricted stock. With respect to employees, the Company is generally required to withhold from regular wages or supplemental wage payments an amount based on the ordinary income recognized. Generally, the Company will be entitled to a business expense deduction equal to the taxable ordinary income realized by the recipient. Upon disposition of the common stock, the recipient will recognize a capital gain or loss equal to the difference between the selling price and the sum of the amount paid for such common stock, if any, plus any amount recognized as ordinary income upon acquisition (or the lapse of restrictions) of the common stock. Such gain or loss will be long-term or short-term depending on how long the common stock was held.
39
The foregoing is only a summary of the effect of federal income taxation upon optionees, holders of restricted stock awards or stock bonus awards and the Company with respect to the grant and exercise of awards under the NSO Plan. It does not purport to be complete, and does not discuss the tax consequences of the employee’s or consultant’s death or the provisions of the income tax laws of any municipality, state or foreign country in which the employee or consultant may reside.
ITEM 13. CERTAIN RELATIONSHIPS, RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required by this item is incorporated by reference into our definitive Proxy Statement to be filed pursuant to Regulation 14A under the Exchange Act in connection with our annual meeting of stockholders.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required by this item is incorporated by reference into our definitive Proxy Statement to be filed pursuant to Regulation 14A under the Exchange Act in connection with our annual meeting of stockholders.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a) The following documents are filed as part of this Report.
1. Financial Statements. The following financial statements of the company and the Report of Independent Registered Public Accounting Firm are included in this Report on the pages indicated.
Page | |
| Report of Independent Registered Public Accounting Firm | 46 |
| Consolidated Balance Sheets as of December 31, 2007 and December 31, 2006 | 47 |
| Consolidated Statements of Operations for the years ended December 31, 2007, 2006 and 2005 | 48 |
| Consolidated Statements of Stockholders’ Equity for the years ended December 31, 2007, 2006 and 2005 | 49 |
| Consolidated Statements of Cash Flows for the years ended December 31, 2007, 2006 and 2005 | 50 |
| Notes to the Consolidated Financial Statements | 51 |
2. Financial Statement Schedule. The following financial statement schedule of the company as of and for the years ended December 31, 2007, 2006 and 2005, is included in Part IV of this Report on the pages indicated. This financial statement schedule should be read in conjunction with the consolidated financial statements, and notes thereto, of the Company.
40
Schedule | Title | Page | ||
| II | Valuation and Qualifying Accounts | 69 |
Schedules not listed above have been omitted because they are not applicable, not required, or the information required to be set forth therein is included in the consolidated financial statements or notes thereto.
3. Exhibits (in accordance with Item 601 of Regulation S-K).
| 2.1 | Agreement and Plan of Merger, dated as of September 3, 2004, by and among ArthroCare Corporation, Opus Medical, Inc., OC Merger Sub Corporation, OC Acquisition Sub LLC, and James W. Hart and Steven L. Gex, as the Shareholders’ Agents (Incorporated herein by reference to Exhibit 2.1 to the Registrant’s Current Report on Form 8-K filed on November 18, 2004). |
| 2.2 | Amendment No. 1, dated as of October 6, 2004, to the Agreement and Plan of Merger, dated as of September 3, 2004, by and among ArthroCare Corporation, Opus Medical, Inc., OC Merger Sub Corporation, OC Acquisition Sub LLC, and James W. Hart and Steven L. Gex, as the Shareholders’ Agents (Incorporated herein by reference to Exhibit 2.2 to the Registrant’s Current Report on Form 8-K filed on November 18, 2004). |
| 2.3 | Asset Purchase Agreement, dated as of August 16, 2005, by and among ArthroCare Corporation, a Delaware corporation, ArthroCare (Deutschland) GmbH, a corporation organized under the laws of Germany and a wholly-owned subsidiary of ArthroCare, ArthroCare UK, Ltd., a corporation registered in England & Wales and a wholly-owned subsidiary of ArthroCare, Applied Therapeutics, Inc., a Florida corporation, Applied Therapeutics, Ltd., a corporation registered in England & Wales, Applied Therapeutics GmbH, a corporation organized under the laws of Germany and BHK Holding, a corporation organized under the laws of the Cayman Islands. (Incorporated herein by reference to Exhibit 2.1 to the Registrant’s Current Report on Form 8-K filed on August 22, 2005). |
| 3.1 | Restated Certificate of Incorporation of the Registrant. (Incorporated herein by reference to Exhibit 3.1 filed previously with the Registrant’s Annual Report on Form 10-K for the period ended December 30, 2000). |
| 3.2 | Amended and Restated Bylaws of the Registrant. (Incorporated herein by reference to Exhibit 3.2 filed previously with the Registrant’s Quarterly Report on Form 10-Q for the period ended October 3, 1998). |
| 3.3 | Certificate of Amendment of the Amended and Restated Bylaws of the Registrant (Incorporated herein by reference to Exhibit 3.1 filed previously with the Registrant’s Current Report on Form 8-K filed on December 13, 2006). |
| 4.1 | Specimen Common Stock Certificate. (Incorporated herein by reference to Exhibit 4.1 filed previously with the Registrant’s Registration Statement on Form 8-A (Registration No. 000-27422)). |
| 10.1* | Form of Indemnification Agreement between the registrant and each of its directors and officers. (Incorporated herein by reference to Exhibit 10.1 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.2* | Incentive Stock Plan and form of Stock Option Agreement there under. (Incorporated herein by reference to Exhibit 10.2 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.3* | Director Option Plan and form of Director Stock Option Agreement there under. (Incorporated herein by reference to Exhibit 10.3 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.4* | Employee Stock Purchase Plan and forms of agreements there under. (Incorporated herein by reference to Exhibit 10.4 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.5 | Form of Exclusive Distribution Agreement. (Incorporated herein by reference to Exhibit 10.5 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.6 | Form of Exclusive Sales Representative Agreement. (Incorporated herein by reference to Exhibit 10.6 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
41
| 10.7 | Consulting Agreement dated May 10, 1993, between the Registrant and Philip E. Eggers, and amendment thereto. (Incorporated herein by reference to Exhibit 10.7 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.8 | Consulting Agreement dated May 20, 1993, between the Registrant and Eggers & Associates, Inc., and amendment thereto. (Incorporated herein by reference to Exhibit 10.8 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.9 | Lease Agreement, dated May 20, 1993, between the Registrant and Eggers & Associates, Inc., and amendment thereto. (Incorporated herein by reference to Exhibit 10.9 filed previously with the Registrant’s Registration Statement on Form S-1 (registration No. 33-80453)). |
| 10.10 | Amended and Restated Stockholder Right Agreement dated October 16, 1995, between the Registrant and certain holders of the Registrant’s securities. (Incorporated herein by reference to Exhibit 10.20 filed previously with the Registrant’s Registration Statement on form S-1 (Registration No. 33-80453)). |
| 10.11 | Contribution Agreement, dated March 31, 1995, by and among Philip E. Eggers, Robert S. Garvie, Anthony J. Manlove, Hira V. Thapliyal and the Registrant. (Incorporated herein by reference to Exhibit 10.21 filed previously with the Registrant’s Registration Statement on Form S-1 (Registration No. 33-80453)). |
| 10.12 | Amended and Restated Stockholder Rights Agreement dated October 2, 1998, between the Registrant and Norwest Bank Minnesota, N.A. (Incorporated herein by reference to Exhibit 10.20 filed previously with the Registrant’s Registration Statement on Form 8-A filed October 21, 1998 (Registration No. 000-27422)). |
| 10.13 | Exclusive Distributor Agreement dated August 21, 1997, between the Registrant and Kobyashi Pharmaceutical Company, Ltd. (Incorporated herein by reference to Exhibit 10.25 filed previously with the Registrant’s Quarterly Report on Form 10-Q for the period ended September 27, 1997). | |
| 10.14 | License Agreement dated February 9, 1998, between the Registrant and Boston Scientific Corporation. (Incorporated herein by reference to Exhibit 10.26 filed previously with the Registrant’s Annual Report on form 10-K for the period ended January 3, 1998). | |
| 10.15 | Development and Supply Agreement dated February 9, 1998, between the Registrant and Boston Scientific Corporation. (Incorporated herein by reference to Exhibit 10.27 filed previously with the Registrant’s Annual report on form 10-K for the period ended January 3, 1998). | |
| 10.16* | Change of Control Agreement between the Registrant and the CEO. (Incorporated herein by reference to Exhibit 10.28 filed previously with the Registrant’s Annual Report on Form 10-K for the period ended January 2, 1999). | |
| 10.17* | The form of “VP Continuity Agreement” between the Registrant and its Vice Presidents. (Incorporated herein by reference to Exhibit 10.29 filed previously with the Registrant’s Annual Report on Form 10-K for the period ended January 2, 1999). | |
| 10.18 | Letter Agreement dated February 9, 1999 between the Registrant and Collagen Aesthetics. (Incorporated herein by reference to Exhibit 10.30 filed previously with the Registrant’s Annual Report on Form 10-K/A for the period ended January 2, 1999). | |
| 10.19* | Employment Letter Agreement, between the Registrant and John R. Tighe dated January 26, 1999. (Incorporated herein by reference to Exhibit 10.30 filed previously with the registrant’s Quarterly Report on Form 10-Q for the period ended April 3, 1999). | |
| 10.20* | Reserved. | |
| 10.21* | Employment Letter Agreement, between the Registrant and Bruce P. Prothro amended May 19, 1999. (Incorporated herein by reference to Exhibit 10.32 filed previously with the Registrant’s Quarterly Report on Form 10-Q for the period ended April 3, 1999). | |
| 10.22† | Litigation Settlement Agreement, between the Registrant and ETHICON Inc. dated June 24, 1999 (Incorporated herein by reference to Exhibit 10.33 previously filed with the Registrant’s Quarterly Report on Form 10-Q for the period ended July 3, 1999). | |
42
| 10.23 | Relocation Loan Agreement, between the Registrant and John. R. Tighe dated May 1, 1999. (Incorporated herein by reference to Exhibit 10.34 previously filed with the Registrant’s Quarterly Report on Form 10-Q for the period ended July 3, 1999). |
| 10.24 | Line of Credit Agreement with Silicon Valley Bank dated June 11, 1999. (Incorporated herein by reference to Exhibit 10.35 previously filed with the Registrant’s Quarterly Report on Form 10-Q for the period ended July 3, 1999). |
| 10.25† | Amendment to License Agreement between ArthroCare Corporation and Inamed Corporation dated October 1, 1999. (Incorporated herein by reference to Exhibit 10.33 previously filed with the Registrant’s Registration Statement on Form S-3 (Registration No. 333-87187)). |
| 10.26 | First Amendment to Rights Agreement between the ArthroCare Corporation and Norwest Bank Minnesota, N.A. (the “Rights Agent”) dated March 10, 2000. (Incorporated herein by reference to Exhibit 99.1 previously filed with the Registrant’s Form 8-K filed March 10, 2000.) |
| 10.27* | Nonstatutory Stock Option Plan and form of Stock Option Agreement there under. (Incorporated herein by reference to Exhibit 10.35 filed previously with this the Registrant’s Annual Report on Form 10-K for the period ended December 31, 1999). |
| 10.28† | License Agreement between ArthroCare Corporation and Stryker Corporation, dated June 28, 2000. (Incorporated herein by reference to Exhibit 10.36 filed previously with this the Registrant’s Quarterly Report on Form 10-Q for the period ended July 1, 2000). |
| 10.29* | Change of Control Agreement between the Registrant and Michael Baker, CEO, dated September 25, 2001. (Incorporated herein by reference to Exhibit 10.37 filed previously with the Registrant’s Quarterly Report on Form 10-Q for the period ended September 29, 2001). |
| 10.30* | Amendment to the 1993 Incentive Plan (Incorporated herein by reference to Exhibit 4.2 to the Registrant’s Registration Statement on Form S-8 filed on August 8, 2000). |
| 10.31* | Amendment to the 1995 Director Option Plan (Incorporated herein by reference to Exhibit 4.3 to the Registrant’s Statement on Form S-8 filed on August 8, 2000). |
| 10.32† | Share Purchase Agreement relating to the entire issued share capital of Atlantech Medical Devices Limited and Atlantech Medical Devices (UK), Limited, dated October 21, 2002. |
| 10.33* | Amended and Restated Nonstatutory Option Plan (Incorporated herein by reference to Exhibit 4.5 to the Registrant’s Registration Statement on Form S-8 filed on May 8, 2003). |
| 10.34* | 2003 Incentive Stock Plan (Incorporated herein by reference to Exhibit 4.4 to the Registrant’s Registration Statement on Form S-8 filed with the Securities and Exchange Commission on June 24, 2003). |
| 10.35* | Second Amendment to the 1995 Director Option Plan (Incorporated herein by reference to Exhibit 4.3 to the Registrant’s Registration Statement on Form S-8 filed with the Securities and Exchange Commission on May 8, 2003). |
| 10.36 | Agreement and Plan of Merger, dated as of October 23, 2003, as amended by Amendment No. 1 to Agreement and Plan of Merger, dated as of January 5, 2004, by and among ArthroCare Corporation, Alpha Merger Sub Corporation and Medical Device Alliance Inc. (Incorporated herein by reference to Exhibit 2.1 to the Registrant’s Current Report on Form 8-K filed on February 11, 2004). |
| 10.37 | Contingent Value Rights Agreement, dated as of January 28, 2004, by and among ArthroCare Corporation, Alpha Merger Sub Corporation, Medical Device Alliance Inc., Wells Fargo Bank, N.A. and Frank Bumstead (Incorporated herein by reference to Exhibit 2.2 to the Registrant’s Current Report on Form 8-K filed on February 11, 2004). |
43
| 10.38 | Form of Stockholder Waiver Agreement, dated as of October 23, 2003, by each of Vegas Ventures, LLC, Jeffrey Barber and Howard Preissman (Incorporated herein by reference to Exhibit 99.3 to the Registrant’s Current Report on Form 8-K filed on October 31, 2003). |
| 10.39 | Stockholder Waiver Agreement, dated as of October 23, 2003, by the McGhan Entities (Incorporated herein by reference to Exhibit 99.4 to the Registrant’s Current Report on Form 8-K filed on October 31, 2003). |
| 10.40† | Credit Agreement between the Registrant, Bank of America, N.A. and Wells Fargo Bank, National Association, dated December 19, 2003. |
| 10.41 | Form of “Amendment to VP Continuity Agreement” between ArthroCare Corporation and its vice presidents (Incorporated herein by reference to Exhibit 10.41 to the Registrant’s Quarterly Report on Form 10-Q filed on August 6, 2004). |
| 10.42 | Form of “Senior VP Continuity Agreement” between ArthroCare Corporation and its senior vice presidents (Incorporated herein by reference to Exhibit 10.42 to the Registrant’s Quarterly Report on Form 10-Q filed on August 6, 2004). |
| 10.43 | Form of “Amendment to Senior VP Continuity Agreement” between ArthroCare Corporation and its senior vice presidents (Incorporated herein by reference to Exhibit 10.43 to the Registrant’s Quarterly Report on Form 10-Q filed on August 6, 2004). |
| 10.44 | Employment Agreement between the Registrant and Michael Baker dated January 1, 2003 (Incorporated herein by reference to Exhibit 10.44 to the Registrant’s Quarterly Report on Form 10-Q filed on November 26, 2004). |
| 10.45† | Amended and Restated Credit Agreement between the Registrant, Bank of America, N.A. and Wells Fargo Bank, National Association, dated October 15, 2004 (Incorporated herein by reference to Exhibit 10.45 to the Registrant’s Quarterly Report on Form 10-Q filed on November 26, 2004 and Exhibit 10.44 to the Registrant’s Quarterly Report on Form 10-Q/A filed on May 11, 2006.) |
| 10.46 | 2004/2005 Executive Officer Bonus Plan (Incorporated herein by reference to Exhibit 10.46 to the Registrant’s Current Report on Form 8-K/A filed on February 22, 2005). |
| 10.47 | 2005 Executive Officer Bonus Plan (Incorporated herein by reference to Exhibit 10.47 to the Registrant’s Current Report on Form 8-K filed on February 22, 2005). |
| 10.48 | Form of Option Agreement under the Amended and Restated 2003 Incentive Plan (Incorporated herein by reference to Exhibit 10.48 to the Registrant’s Current Report on Form 8-K filed on February 22, 2005). |
| 10.49 | Form of Restricted Stock Bonus Agreement under the Amended and Restated Director Option Plan (Incorporated herein by reference to Exhibit 10.49 to the Registrant’s Current Report on Form 8-K filed on February 22, 2005). |
| 10.50 | Form of Restricted Stock Bonus Agreement under the Amended and Restated 2003 Incentive Stock Plan (Incorporated herein by reference to Exhibit 10.50 to the Registrant’s Current Report on Form 8-K filed on February 22, 2005). |
| 10.51† | First Amendment to Amended and Restated Credit Agreement between the Registrant, Bank of America, N.A. and Wells Fargo Bank, National Association, dated March 30, 2005. |
| 10.52† | Settlement and License Agreement between the Registrant, ArthroCare Caymans, a corporation organized under the laws of the Cayman Islands and a wholly-owned subsidiary of the Registrant and Smith & Nephew, Inc., dated September 2, 2005 (Incorporated herein by reference to Exhibit 10.52 to the Registrant’s Quarterly Report on Form 10-Q filed November 3, 2005). |
| 10.53† | Settlement and Distribution Agreement between the Registrant, ArthroCare Caymans, a corporation organized under the laws of the Cayman Islands and a wholly-owned subsidiary of the Registrant and Smith & Nephew, Inc., dated September 2, 2005 (Incorporated herein by reference to Exhibit 10.53 to the Registrant’s Quarterly Report on Form 10-Q filed November 3, 2005). |
| 10.54 * | 2006 Executive Officer Bonus Plan (Incorporated herein by reference to Exhibit 10.54 to the Registrant’s Current Report on Form 8-K filed on February 24, 2006). |
44
| 10.55 * | 2006 Director Compensation Plan (Incorporated herein by reference to Exhibit 10.55 to the Registrant’s Current Report on Form 8-K filed on February 24, 2006). |
| 10.56 | Credit Agreement between the Registrant, Bank of America, N.A., Wells Fargo Bank National Association, and certain other lenders dated January 13, 2006. |
| 10.57 | Amended and Restated 2003 Incentive Stock Plan (Incorporated herein by reference to Exhibit 10.58 to the Registrant’s Current Report on Form 8-K filed on May 25, 2006). |
| 10.58 | Lease Agreement between Lantana Office properties I, L.P. and the Registrant (Incorporated herein by reference to Exhibit 10.57 to the Registrant’s Quarterly Report on 10-Q filed on June 30, 2006). |
| 10.59 | Employment Agreement between the Registrant and Mike Baker, effective January 1, 2007 (Incorporated herein by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed on September 1, 2006). |
| 10.60† | Settlement and License Agreement between the Registrant, ArthroCare Corporation Cayman Islands and MarcTec, LLC effective January 3, 2007. |
| 10.61 * | 2007 Executive Officer Bonus Plan (Incorporated herein by reference to Exhibit 10.61 to the Registrant’s Current Report on Form 8-K filed on March 1, 2007). |
| 10.62 | Certificate of Amendment of the Amended and Restated Bylaws of the Registrant (Incorporated herein by reference to Exhibit 3.4 to the Registrant’s Current Report on Form 8-K filed on April 5, 2007). |
| 10.63 * | Form of “Amended VP Continuity Agreement” between ArthroCare Corporation and its vice presidents (Incorporated herein by reference to Exhibit 10.61 to the Registrant’s Current Report on Form 8-K filed on April 5, 2007. |
| 10.64 * | Form of “Amended Senior VP Continuity Agreement” between ArthroCare Corporation and its senior vice presidents (Incorporated herein by reference to Exhibit 10.62 to the Registrant’s Current Report on Form 8-K filed on April 5, 2007). |
| 10.65†† | Stock Purchase Agreement, dated as of December 31, 2007 by and among ArthroCare Corporation and DiscoCare, Inc. |
| 21.1 | Subsidiaries of the Registrant. |
| 23.1 | Consent of PricewaterhouseCoopers LLP, Independent Registered Public Accounting Firm. |
| 31.1 | Certification of the Chief Executive Officer pursuant to Rule 13a-14(a) of the Exchange Act, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| 31.2 | Certification of the Chief Financial Officer pursuant to Rule 13a-14(a) of the Exchange Act, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| 32.1 | Certification of the Chief Executive Officer pursuant to Rule 13a-14(b) of the Exchange Act and 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| 32.2 | Certification of the Chief Financial Officer pursuant to Rule 13a-14(b) of the Exchange Act and 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| † | Confidential treatment has been granted as to portions of this exhibit. |
| †† | Confidential treatment has been requested as to portions of this exhibit. |
| * | Management contract or compensatory plan or arrangement. |
45
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of ArthroCare Corporation
In our opinion, the consolidated financial statements listed in the index appearing under Item 15(a)(1) present fairly, in all material respects, the financial position of ArthroCare Corporation and its subsidiaries at December 31, 2007 and 2006, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2007 in conformity with accounting principles generally accepted in the United States of America. In addition, in our opinion, the financial statement schedule listed in the index appearing under Item 15(a)(2) presents fairly, in all material respects, the information set forth therein when read in conjunction with the related consolidated financial statements. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2007, based on criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company's management is responsible for these financial statements and financial statement schedule, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in Management's Report on Internal Control over Financial Reporting appearing under Item 9A. Our responsibility is to express opinions on these financial statements, on the financial statement schedule, and on the Company's internal control over financial reporting based on our integrated audits. We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audits of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
As discussed in Note 2 to the consolidated financial statements, the Company changed the manner in which it accounts for uncertain tax positions in 2007 and the manner in which it accounts for stock-based compensation in 2006.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ PricewaterhouseCoopers LLP
Austin, Texas
February 29, 2008
46
ARTHROCARE CORPORATION
CONSOLIDATED BALANCE SHEETS
(in thousands, except per share data)
December 31, | |||||||
2007 | 2006 | ||||||
ASSETS | |||||||
| Current assets: | |||||||
| Cash and cash equivalents | $ | 39,375 | $ | 15,531 | |||
| Short-term investments | 3,875 | 15,225 | |||||
| Accounts receivable, net of allowances of $3,980 and $2,701 at 2007 and 2006, respectively | 69,924 | 61,935 | |||||
| Inventories, net | 61,776 | 51,542 | |||||
| Deferred tax assets | 10,406 | 13,795 | |||||
| Prepaid expenses and other current assets | 5,164 | 5,389 | |||||
| Total current assets | 190,520 | 163,417 | |||||
| Property and equipment, net | 43,405 | 36,071 | |||||
| Related party receivables | — | 500 | |||||
| Intangible assets, net | 37,705 | 35,982 | |||||
| Goodwill | 166,771 | 137,831 | |||||
| Deferred tax assets | 4,940 | — | |||||
| Other assets | 6,727 | 1,245 | |||||
| Total assets | $ | 450,068 | $ | 375,046 | |||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |||||||
| Current liabilities: | |||||||
| Accounts payable | $ | 15,876 | $ | 12,993 | |||
| Accrued liabilities | 12,909 | 26,347 | |||||
| Accrued compensation | 12,727 | 7,906 | |||||
| Deferred tax liabilities | 530 | — | |||||
| Income taxes payable | 130 | 2,427 | |||||
| Total current liabilities | 42,172 | 49,673 | |||||
| Notes payable | 60,000 | — | |||||
| Deferred tax liabilities | 383 | 1,991 | |||||
| Other non-current liabilities | 7,010 | 879 | |||||
| Total liabilities | 109,565 | 52,543 | |||||
| Commitments and contingencies (Note 9) | |||||||
| Stockholders’ equity: | |||||||
| Preferred stock, par value $0.001; Authorized: 5,000 shares; | |||||||
| Issued and outstanding: none | — | — | |||||
| Common stock, par value $0.001: Authorized: 75,000 shares; | |||||||
| Issued and outstanding: 26,808 shares at 2007 and 27,394 shares at 2006 | 27 | 27 | |||||
| Treasury stock: 3,936 shares at 2007 and 2,692 shares at 2006 | (100,682 | ) | (41,644 | ) | |||
| Additional paid-in capital | 360,374 | 324,823 | |||||
| Accumulated other comprehensive income | 2,382 | 1,992 | |||||
| Retained earnings | 78,402 | 37,305 | |||||
| Total stockholders’ equity | 340,503 | 322,503 | |||||
| Total liabilities and stockholders’ equity | $ | 450,068 | $ | 375,046 | |||
The accompanying notes are an integral part of these consolidated financial statements.
47
ARTHROCARE CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share data)
Year Ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Revenues: | ||||||||||
| Product sales | $ | 307,596 | $ | 253,376 | $ | 206,533 | ||||
| Royalties, fees and other | 11,646 | 9,625 | 7,801 | |||||||
| Total revenues | 319,242 | 263,001 | 214,334 | |||||||
| Cost of product sales | 84,724 | 76,838 | 64,206 | |||||||
| Gross profit | 234,518 | 186,163 | 150,128 | |||||||
| Operating expenses: | ||||||||||
| Research and development | 26,886 | 23,247 | 21,015 | |||||||
| Sales and marketing | 123,213 | 91,915 | 75,302 | |||||||
| General and administrative | 24,033 | 21,355 | 12,202 | |||||||
| Amortization of intangible assets | 7,211 | 7,176 | 6,150 | |||||||
| Acquired in-process research and development costs | — | — | 2,400 | |||||||
| Total operating expenses | 181,343 | 143,693 | 117,069 | |||||||
| Income from operations | 53,175 | 42,470 | 33,059 | |||||||
| Interest income | 2,101 | 1,376 | 631 | |||||||
| Interest expense | (977 | ) | (2,423 | ) | (1,593 | ) | ||||
| Other income (expense), net | 1,060 | (762 | ) | (1,374 | ) | |||||
| Income before income taxes | 55,359 | 40,661 | 30,723 | |||||||
| Income tax provision | 12,179 | 8,986 | 7,193 | |||||||
| Net income | $ | 43,180 | $ | 31,675 | $ | 23,530 | ||||
| Basic net income per share | $ | 1.57 | $ | 1.21 | $ | 0.97 | ||||
| Shares used in computing basic net income per share | 27,452 | 26,207 | 24,375 | |||||||
| Diluted net income per share | $ | 1.50 | $ | 1.14 | $ | 0.89 | ||||
| Shares used in computing diluted income per share | 28,870 | 27,900 | 26,407 | |||||||
The accompanying notes are an integral part of these consolidated financial statements.
48
ARTHROCARE CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands)
Common Stock Shares | Common Stock Amount | Treasury Stock Amount | Additional Paid-In Capital | Deferred Stock Compensation | Accumulated Other Comprehensive Income (Loss) | Retained Earnings (Deficit) | Total Stockholders’ Equity | Comprehensive Income | ||||||||||||||||||||
Balances, December 31, 2004 | 24,086 | $ | 24 | $ | (42,158 | ) | $ | 238,355 | $ | (2,477 | ) | $ | (592 | ) | $ | (17,900 | ) | $ | 175,252 | |||||||||
| Issuance of common stock through: | ||||||||||||||||||||||||||||
| Exercise of options | 1,020 | 1 | — | 16,459 | — | — | — | 16,460 | ||||||||||||||||||||
| Employee stock purchase plan | 48 | — | — | 1,291 | — | — | — | 1,291 | ||||||||||||||||||||
| Issuance of restricted stock | 101 | — | — | 3,392 | (3,392 | ) | — | — | — | |||||||||||||||||||
| Stock compensation | — | — | — | 90 | 1,326 | — | — | 1,416 | ||||||||||||||||||||
| Income tax benefit resulting from exercise of stock options | — | — | — | 9,583 | — | — | — | 9,583 | ||||||||||||||||||||
| Currency translation adjustment | — | — | — | — | — | 1,362 | — | 1,362 | $ | 1,362 | ||||||||||||||||||
| Net income | — | — | — | — | — | — | 23,530 | 23,530 | 23,530 | |||||||||||||||||||
Balances, December 31, 2005 | 25,255 | 25 | (42,158 | ) | 269,170 | (4,543 | ) | 770 | 5,630 | 228,894 | $ | 24,892 | ||||||||||||||||
| Issuance of common stock through: | ||||||||||||||||||||||||||||
| Exercise of options and restricted stock | 1,955 | 2 | — | 28,095 | — | — | — | 28,097 | ||||||||||||||||||||
| Employee stock purchase plan | 4 | — | — | 151 | — | — | — | 151 | ||||||||||||||||||||
| Shares issued for Opus Medical | 180 | — | — | 8,054 | — | — | — | 8,054 | ||||||||||||||||||||
| Contributions to employee benefit plan | — | — | 514 | — | — | — | — | 514 | ||||||||||||||||||||
| Stock compensation | — | — | — | 10,052 | — | — | — | 10,052 | ||||||||||||||||||||
| Income tax benefit resulting from exercise of stock options | — | — | — | 13,844 | — | — | — | 13,844 | ||||||||||||||||||||
| Reclassification at adoption of FAS 123R | — | — | — | (4,543 | ) | 4,543 | — | — | — | |||||||||||||||||||
| Currency translation adjustment | — | — | — | — | — | 1,222 | — | 1,222 | $ | 1,222 | ||||||||||||||||||
| Net income | — | — | — | — | — | — | 31,675 | 31,675 | 31,675 | |||||||||||||||||||
Balances, December 31, 2006 | 27,394 | 27 | (41,644 | ) | 324,823 | — | 1,992 | 37,305 | 322,503 | $ | 32,897 | |||||||||||||||||
| Issuance of common stock through: | ||||||||||||||||||||||||||||
| Exercise of options and restricted stock | 638 | 1 | — | 14,297 | — | — | — | 14,298 | ||||||||||||||||||||
| Employee stock purchase plan | 7 | — | — | 306 | — | — | — | 306 | ||||||||||||||||||||
| Contributions to employee benefit plan | 25 | — | 607 | — | — | — | — | 607 | ||||||||||||||||||||
| Stock compensation | — | — | — | 10,279 | — | — | — | 10,279 | ||||||||||||||||||||
| Income tax benefit resulting from exercise of stock options | — | — | — | 10,669 | — | — | — | 10,669 | ||||||||||||||||||||
| Purchase of common stock | (1,256 | ) | (1 | ) | (59,645 | ) | — | — | — | — | (59,646 | ) | ||||||||||||||||
| Reclassification at adoption of FIN 48 | — | — | — | — | — | — | (2 ,083 | ) | (2,083 | ) | ||||||||||||||||||
| Currency translation adjustment | — | — | — | — | — | 390 | — | 390 | $ | 390 | ||||||||||||||||||
| Net income | — | — | — | — | — | — | 43,180 | 43,180 | 43,180 | |||||||||||||||||||
Balances, December 31, 2007 | 26,808 | $ | 27 | $ | (100,682 | ) | $ | 360,374 | $ | — | $ | 2,382 | $ | 78,402 | $ | 340,503 | $ | 43,570 | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
49
ARTHROCARE CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
Year Ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Cash flows from operating activities: | ||||||||||
| Net income | $ | 43,180 | $ | 31,675 | $ | 23,530 | ||||
| Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||||
| Acquired in-process research and development projects | — | — | 2,400 | |||||||
| Depreciation and amortization | 21,203 | 21,339 | 16,768 | |||||||
| Provision for doubtful accounts receivable and product returns | 2,498 | 1,887 | 1,111 | |||||||
| Provision for inventory reserves | 3,760 | 4,191 | 856 | |||||||
| Non-cash stock compensation expense | 10,215 | 9,824 | 1,416 | |||||||
| Deferred taxes | (630 | ) | (4,741 | ) | (5,871 | ) | ||||
| Income tax benefit relating to employee stock options | (7,811 | ) | (9,476 | ) | 9,583 | |||||
| Other | (714 | ) | 351 | 1,135 | ||||||
| Changes in operating assets and liabilities, net of assets acquired and liabilities assumed in business combinations: | ||||||||||
| Accounts receivable | (23,765 | ) | (17,200 | ) | (14,753 | ) | ||||
| Inventories | (6,155 | ) | (8,617 | ) | (7,655 | ) | ||||
| Prepaid expenses and other current assets | 220 | (2,049 | ) | 1,290 | ||||||
| Other assets | (4,965 | ) | 197 | 28 | ||||||
| Accounts payable | 2,736 | 1,121 | 3,397 | |||||||
| Accrued liabilities | 4,568 | 3,131 | 317 | |||||||
| Income taxes payable | 8,372 | 12,167 | 3,626 | |||||||
| Net cash provided by operating activities | 52,712 | 43,800 | 37,178 | |||||||
| Cash flows from investing activities: | ||||||||||
| Payments for purchases of property and equipment | (20,198 | ) | (17,240 | ) | (13,281 | ) | ||||
| Payments for business combinations and purchases of intangible assets | (41,267 | ) | (57,307 | ) | (10,521 | ) | ||||
| Payments for purchases of available-for-sale securities | (1,301 | ) | (26,926 | ) | — | |||||
| Proceeds from sales of available-for-sale securities | 12,839 | 15,000 | 7,400 | |||||||
| Net cash used in investing activities | (49,927 | ) | (86,473 | ) | (16,402 | ) | ||||
| Cash flows from financing activities: | ||||||||||
| Payments on notes payable | — | (38,500 | ) | (28,928 | ) | |||||
| Proceeds from notes payable | 60,000 | 38,500 | — | |||||||
| Payments for purchases of treasury stock | (59,646 | ) | — | — | ||||||
| Proceeds from issuance of common stock, net of issuance costs | 306 | 151 | 1,291 | |||||||
| Proceeds from exercise of options to purchase common stock, net of issuance costs | 14,298 | 28,097 | 16,460 | |||||||
| Income tax benefit relating to employee stock options | 7,811 | 9,476 | — | |||||||
| Net cash provided by (used in) financing activities | 22,769 | 37,724 | (11,177 | ) | ||||||
| Effect of exchange rate changes on cash and cash equivalents | (1,710 | ) | (237 | ) | (718 | ) | ||||
| Net increase (decrease) in cash and cash equivalents | 23,844 | (5,186 | ) | 8,881 | ||||||
| Cash and cash equivalents, beginning of year | 15,531 | 20,717 | 11,836 | |||||||
| Cash and cash equivalents, end of year | $ | 39,375 | $ | 15,531 | $ | 20,717 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
50
ARTHROCARE CORPORATION
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 - FORMATION AND BUSINESS OF THE COMPANY
ArthroCare Corporation (“ArthroCare” or the “Company”) was incorporated on April 29, 1993 and its principal operations commenced in August 1995. The Company designs, develops, manufactures and markets medical devices for use in soft-tissue surgery. Its products are based on the Company’s patented soft-tissue surgical controlled ablation technology, which it calls Coblation technology. Coblation technology involves an innovative use of a non-thermal process and has the capability of performing at temperatures lower than traditional electrosurgical tools. ArthroCare’s strategy includes applying Coblation technology to a broad range of soft-tissue surgical markets, including sports medicine, spinal surgery, cavity surgery, neurosurgery, cosmetic surgery, ear, nose and throat, or ENT, surgery, gynecology, urology, general surgery and various cardiology applications. ArthroCare is a global company with manufacturing facilities in Costa Rica and the United States and sales offices in the United States, Europe and the South Pacific.
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation. The consolidated financial statements include the accounts of ArthroCare and all of its wholly-owned subsidiaries. All significant intra-company transactions and accounts have been eliminated. The Company uses the calendar year as its fiscal year.
Use of Estimates. The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities in the consolidated financial statements and the reported amount of revenue and expenses during the reporting period. Actual results could differ from those estimates.
Cash and Cash Equivalents. The Company considers all highly liquid investments purchased with original maturities of ninety days or less to be cash equivalents. Cash and cash equivalents include money market funds and various deposit accounts.
Short-term Investments. Short-term investments consist primarily of corporate bonds with readily determinable fair market values. The Company’s investments are classified as “available-for-sale” and accordingly are reported at fair value, with unrealized gains and losses reported as a component of stockholders’ equity. Unrealized losses are charged against income when a decline in the fair market value of an individual security is determined to be other than temporary. Realized gains and losses on investments are included in interest income and are determined on the specific identification method. For the years ended December 31, 2007, 2006 and 2005, the Company had no significant realized gains or losses.
Inventories. The Company’s inventories, which include material and labor costs, are stated at standard cost, which approximates actual cost determined on a first-in, first-out basis, not in excess of market value. The Company records reserves, when necessary, to reduce the carrying value of excess or obsolete inventories to their net realizable value.
Property and Equipment. Property and equipment is stated at cost and is depreciated on a straight-line basis over the estimated useful lives of three to five years, except buildings which have an estimated useful life of 30 years. The Company places the majority of its manufactured controller units with customers in order to facilitate the sale of disposable devices. Controller units placed with customers are capitalized at cost and amortized to cost of product sales over a three or four-year period. Leasehold improvements are amortized over the shorter of the estimated useful lives or the lease term. Maintenance and repair costs are charged to operations as incurred. Upon retirement or sale, the cost of disposed assets and their accumulated depreciation are removed from the balance sheet and any gain or loss is recognized in current operations.
Revenue Recognition and Allowance for Doubtful Accounts. The Company recognizes product revenue in accordance with SEC Staff Accounting Bulletin No. 104, Revenue Recognition in Financial Statements, or SAB 104. Under SAB 104, revenue is recognized when there is persuasive evidence of an arrangement, delivery has occurred or services have been rendered, the sales price is fixed and determinable and collectibility is reasonably assured. Generally, the criteria are met upon shipment of the Company’s products. For amounts due from third-party payors, revenues are recorded at estimated net amounts due for devices utilized with allowances determined using historical collection amounts for each type of payor. Estimated allowances are adjusted to actual allowances as cash is received and claims are reconciled. We evaluate the following criteria in developing the estimated allowance percentages each month: historical allowance trends based on actual claims paid by third-party payors; consideration and analysis of changes in payor mix reimbursement levels; and other issues that may impact allowances. In certain cases, at the customer’s request, we will enter into bill and hold transactions whereby title and risk of loss transfers to the customer, but the product does not ship until a specified later date. Revenue on such transactions is recognized only after ArthroCare has met all conditions for bill and hold transactions set forth in SAB 104, specifically that ArthroCare has no further performance obligations, that the product is complete and ready for shipment, and that a fixed schedule for delivery of the product is set. As of December 31, 2007, 2006 and 2005, there were no undelivered bill and hold transactions. Revenue is reported net of a provision for estimated product returns.
51
The Company recognizes license fees and other revenue over the term of the associated agreement unless the fee is in exchange for products delivered or services performed that represent the culmination of a separate earnings process. Royalties are recognized as earned, generally based on the licensees’ product shipments. These items are classified as royalties, fees and other revenues in the accompanying condensed consolidated statements of operations. Amounts billed to customers relating to shipping and handling costs have also been classified as royalties, fees and other revenues and related costs are classified as cost of product sales in the accompanying condensed consolidated statements of operations.
The Company maintains an allowance for doubtful accounts receivable based on our assessment of the collectibility of customer accounts. The Company regularly reviews the allowance, considering such factors as historical collection experience, a customer’s current credit-worthiness, customer concentrations, the age of the receivable balances, both individually and in the aggregate, and general economic conditions that may affect a customer’s ability to pay. Actual customer collections could differ from our estimates. The Company believes that the allowance for doubtful accounts of $4.0 million at December 31, 2007 is adequate to provide for probable losses associated with accounts receivable.
Goodwill. The Company’s methodology for allocating the purchase price relating to business acquisitions is consistent with established valuation techniques in the medical device industry. Goodwill is measured as the excess of the cost of the business acquired over the sum of the amounts assigned to tangible and identifiable intangible assets acquired less liabilities assumed.
In accordance with Statement of Financial Accounting Standards No. 142, Goodwill and Other Intangible Assets, or FAS 142, the Company performs an annual impairment test of goodwill. The Company evaluates goodwill in the fourth quarter of each year or more frequently if events or changes in circumstances indicate that goodwill might be impaired. As required by FAS 142, the impairment test is accomplished using a two-stepped approach. The first step screens for impairment and, when impairment is indicated, a second step is employed to measure the impairment. Using data as of December 31, 2007, the first step did not indicate an impairment of goodwill and based on other risk factors reviewed by the Company, no impairment of the goodwill balance was considered necessary.
Long-Lived Assets. Long-lived assets and certain identifiable intangible assets to be disposed of are reported at the lower of carrying amount or fair value less costs to sell. Purchased intangible assets are carried at cost less accumulated amortization. Amortization is computed over the estimated useful lives of the respective assets, which are generally less than ten years. Long-lived assets and certain identifiable intangible assets to be held and used are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable. Determination of recoverability is based on an estimate of undiscounted future cash flows resulting from the use of the group of assets and their eventual disposition. Measurement of an impairment loss for long-lived assets and certain identifiable intangible assets that management expects to hold and use is based on the fair value of the asset.
Treasury Stock. Treasury stock is accounted for under the cost method and is included as a component of stockholders’ equity.
52
Research and Development. Research and development costs consist mostly of payroll expenses and prototype development costs and are charged to operations as incurred.
Advertising Expense. Advertising expenses are charged to operations as sales and marketing expenses as incurred. Advertising expense was $5.0 million, $4.0 million and $3.9 million in 2007, 2006 and 2005, respectively.
Stock-Based Compensation. Effective January 1, 2006, the Company adopted the provisions of Statement of Financial Accounting Standards No. 123 (revised 2004), Share-Based Payment, or FAS 123R, which requires the measurement and recognition of compensation expense for all share-based payment awards made to employees, including employee stock options and employee stock purchases related to the Company’s Employee Stock Purchase Plan, or ESPP, based on estimated fair values. Under the provisions of FAS 123R, share-based compensation cost is measured at the grant date, based on the calculated fair value of the award, and is recognized as an expense over the employee’s requisite service period. Prior to January 1, 2006, the Company accounted for share-based compensation to employees in accordance with Accounting Principles Board Opinion No. 25, Accounting for Stock Issued to Employees, or APB 25, and related interpretations. Under APB 25, when the Company issued options to its employees with an exercise price equal to the market value of the underlying common stock on the date of grant or issuance, no stock-based compensation costs were recorded. In the event stock-based awards were issued with an exercise price that was less than the market value of the underlying common stock on the date of grant or issuance, the Company recorded deferred compensation expense in an amount equivalent to the difference between the market value and the exercise price of the award.
The Company elected to adopt the modified prospective transition method as provided by FAS 123R and, accordingly, financial statement amounts for the prior periods presented in this Form 10-K have not been restated to reflect the fair value method of expensing share-based compensation. Under this transition method, stock-based compensation expense for the year ended December 31, 2005 includes compensation expense for all stock-based compensation awards granted prior to, but not yet vested as of January 1, 2006, based on the grant date fair value estimated in accordance with the original provisions of FAS 123, Accounting for Stock-Based Compensation. Stock-based compensation for all stock-based compensation awards granted subsequent to January 1, 2006 was based on the grant-date fair value estimated in accordance with the provisions of FAS 123R. The Company recognizes compensation expense for stock option awards on a straight-line basis over the requisite service period of the award.
Foreign Currency Translation. Certain of the Company’s wholly-owned subsidiaries have functional currencies other than the U.S. dollar. Accordingly, all balance sheet accounts of these operations are translated into U.S. dollars using the current exchange rate in effect at the balance sheet date, and revenues and expenses are translated using the average exchange rate in effect during the period. The gains and losses from foreign currency translation of these subsidiaries’ financial statements are recorded directly as a separate component of stockholders’ equity under the caption “accumulated other comprehensive income (loss).”
For our other non-U.S. operations which have a functional currency of the U.S. dollar, all monetary assets and liabilities of these foreign operations are remeasured into U.S. dollars at current period-end exchange rates and non-monetary assets and related elements of expense are remeasured using historical rates of exchange. Income and expense elements are remeasured into U.S. dollars using average exchange rates in effect during the period.
Concentration of Risks and Uncertainties. A majority of the Company’s cash and cash equivalents are maintained at financial institutions in the United States. Deposits at these institutions may exceed the amount of insurance provided on such deposits. The Company has not experienced any losses on deposits of cash and cash equivalents.
The Company purchases certain key components of its products from sole, single or limited source suppliers. For some of these components there are few alternative sources. A reduction or stoppage in supply of sole-source components would limit the Company’s ability to manufacture certain products. There can be no assurance that an alternate supplier could be established if necessary or that available inventories would be adequate to meet ArthroCare’s production needs during any prolonged interruption of supply.
The Company’s products require approval from the United States Food and Drug Administration, or FDA, and international regulatory agencies prior to the commencement of commercial sales. There can be no assurance that its products will receive any of these required approvals. If the Company were denied such approvals, or if such approvals were delayed, it would have a material adverse impact on its business.
53
Sales to both international and domestic customers are generally made on open credit terms. Management performs ongoing credit evaluations of the Company’s customers and maintains an allowance for potential credit losses when needed, but historically has not experienced any significant losses related to individual customers or a group of customers in any particular geographic area.
Income Taxes. The Company accounts for income taxes under the liability method, whereby deferred tax asset or liability account balances are determined based on the difference between the financial statement and the tax bases of assets and liabilities using the tax laws and rates in effect for the year in which the differences are expected to affect taxable income. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amounts expected to be realized.
In July 2006, the Financial Accounting Standards Board, or FASB, issued Interpretation No. 48, Accounting for Uncertainty in Income Taxes, or FIN 48. FIN 48 clarifies the accounting for uncertainty in income taxes recognized in an enterprise’s financial statements in accordance with Statement of Financial Accounting Standards No. 109, Accounting for Income Taxes. This interpretation prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. FIN 48 also provides guidance on derecognition of tax benefits, classification on the balance sheet, interest and penalties, accounting in interim periods, disclosure, and transition. We adopted FIN 48 effective January 1, 2007. In accordance with FIN 48, paragraph 19, we have decided to classify interest and penalties as a component of tax expense. As a result of the implementation of FIN 48, we recognized a $2.1 million increase in liability for unrecognized tax benefits, which was accounted for as a reduction to the January 1, 2007 balance of retained earnings.
Reclassifications. Certain reclassifications have been made to prior year balances to conform to the current year’s presentation. The effect of such reclassifications is not material to the consolidated financial statements.
Accounting Pronouncements. In September 2006, the FASB issued Statement of Financial Accounting Standards No. 157, Fair Value Measurements, or FAS 157. FAS 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles, and expands disclosures about fair value measurements. FAS 157 is effective for the Company beginning January 1, 2008 for financial assets and liabilities and January 1, 2009 for non-financial assets and liabilities. The Company is currently in the process of evaluating the impact of adopting FAS 157 on its operating results and financial position. The Company does not believe that FAS 157 will have a material effect on its operating results or financial position.
In February 2007, the FASB issued Statement of Financial Accounting Standards No. 159, The Fair Value Option for Financial Assets and Financial Liabilities - Including an amendment of FASB No. 115, or FAS 159. The Statement permits companies to choose to measure many financial instruments and certain other items at fair value in order to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without having to apply complex hedge accounting provisions. FAS 159 is effective for the Company beginning January 1, 2008. The Company does not believe that FAS 159 will have a material effect on its operating results or financial position.
In December 2007, the FASB issued Statement of Financial Accounting Standards No. 141(R), Business Combinations, or FAS 141R. FAS 141R is effective for the Company as of January 1, 2009 and is applied prospectively for business combinations completed after January 1, 2009. Under FAS 141, organizations utilized the announcement date as the measurement date for the purchase price of the acquired entity. FAS 141R requires measurement at the date the acquirer obtains control of the acquiree, generally referred to as the acquisition date. FAS 141R will have a significant impact on the accounting for transaction costs, restructuring costs as well as the initial recognition of contingent assets and liabilities assumed during a business combination completed after January 1, 2009.
54
NOTE 3 - SHORT-TERM INVESTMENTS
Short-term investments were acquired at an aggregate cost of $3.9 million and $15.2 million at December 31, 2007 and 2006, respectively. At December 31, 2007 and 2006, the Company’s short-term investments consisted primarily of corporate bonds. The fair values of these instruments are based on market interest rates and other market information available to management as of each balance sheet date presented.
Management classifies investments in marketable securities at the time of purchase and reevaluates such classification at each balance sheet date. Securities classified as available-for-sale are stated at fair value, and all those which have original maturities beyond one year have interest reset maturities of less than thirty-five days.
NOTE 4 - COMPUTATION OF NET INCOME PER SHARE
Basic net income per common share is computed using the weighted average number of shares of common stock outstanding. Diluted net income per common share is computed using the weighted average number of shares of common stock outstanding and potential shares of common stock when they are dilutive. The following is a reconciliation of the numerator, net income, and the denominator, number of shares, used in the calculation of basic and diluted net income per share (in thousands, except per share data):
Year Ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Net income | $ | 43,180 | $ | 31,675 | $ | 23,530 | ||||
| Basic: | ||||||||||
| Weighted-average common shares outstanding | 27,452 | 26,207 | 24,375 | |||||||
| Basic net income per share | $ | 1.57 | $ | 1.21 | $ | 0.97 | ||||
| Diluted: | ||||||||||
| Weighted-average shares outstanding used in basic calculation | 27,452 | 26,207 | 24,375 | |||||||
| Dilutive effect of options | 1,282 | 1,453 | 1,933 | |||||||
| Dilutive effect of unvested restricted stock | 136 | 240 | 99 | |||||||
| Weighted-average common stock and common stock equivalents | 28,870 | 27,900 | 26,407 | |||||||
| Diluted net income per share | $ | 1.50 | $ | 1.14 | $ | 0.89 | ||||
| Options excluded from calculation as their effect would be anti-dilutive | 585 | 370 | 142 | |||||||
| Price range of excluded options | $ | 46.82 - $54.15 | $ | 43.68-$48.56 | $ | 34.03-$48.56 | ||||
NOTE 5 - INVENTORY
The following summarizes the Company’s inventories (in thousands):
December 31, | |||||||
2007 | 2006 | ||||||
| Raw materials | $ | 11,201 | $ | 12,126 | |||
| Work-in-progress | 4,308 | 4,949 | |||||
| Finished goods | 52,040 | 38,605 | |||||
| 67,549 | 55,680 | ||||||
| Inventory reserves | (5,773 | ) | (4,138 | ) | |||
| Inventories, net | $ | 61,776 | $ | 51,542 | |||
The provision for inventory reserves for the years ended December 31, 2007, 2006 and 2005 was $3.8 million, $4.2 million and $0.9 million, respectively.
55
NOTE 6 - PROPERTY AND EQUIPMENT
The following summarizes the Company’s property and equipment (in thousands, except lives):
Estimated | ||||||||||
December 31, | Useful Lives | |||||||||
2007 | 2006 | (Years) | ||||||||
| Controller placements | $ | 59,593 | $ | 54,909 | 3 to 4 | |||||
| Computer equipment and software | 15,578 | 16,795 | 3 to 5 | |||||||
| Machinery and equipment | 11,665 | 10,994 | 5 | |||||||
| Furniture, fixtures and leasehold improvements | 10,386 | 6,478 | 5 (a | ) | ||||||
| Construction in process | 8,520 | 3,161 | — | |||||||
| Building and improvements | 5,991 | 5,287 | 30 | |||||||
| Tooling and molds | 5,430 | 1,922 | 5 | |||||||
| Land | 745 | 745 | — | |||||||
| 117,908 | 100,291 | |||||||||
| Less accumulated depreciation | (74,503 | ) | (64,220 | ) | ||||||
| Property and equipment, net | $ | 43,405 | $ | 36,071 | ||||||
| (a) | The estimated useful life for leasehold improvements is the shorter of 5 years or the life of the lease. |
Depreciation expense related to ArthroCare’s property and equipment was $13.8 million, $14.2 million and $11.2 million for the years ended December 31, 2007, 2006 and 2005, respectively.
NOTE 7 - GOODWILL AND INTANGIBLE ASSETS
Goodwill
The changes in the carrying amount of goodwill are as follows (in thousands):
| Balance at December 31, 2005 | $ | 59,170 | ||
| Acquisitions and purchase accounting adjustments | 77,122 | |||
| Translation adjustments and other | 1,539 | |||
| Balance at December 31, 2006 | 137,831 | |||
| Acquisitions and purchase accounting adjustments | 28,675 | |||
| Translation adjustments and other | 265 | |||
| Balance at December 31, 2007 | $ | 166,771 |
In 2006, the Company adjusted the purchase price of the ATI acquisition by $13.3 million for the expected additional payments that were to be made to ATI shareholders in 2007 based on the net revenues earned by the Company from the acquired business. The additional accrual increased the goodwill balance associated with the ATI acquisition. In addition, during 2006, the goodwill balances related to the MDA and Opus acquisitions increased by $0.4 million and $63.2 million, respectively, to reflect payments made to former shareholders based on the satisfaction of contractual contingencies. In 2007, the Company adjusted the goodwill balance associated with its acquisition of ATI by an additional $1.5 million and paid the shareholders of ATI $14.8 million in satisfaction of the remaining contractual obligations associated with the acquisition. The remainder of the increases in goodwill related to the 2007 DiscoCare acquisition as well as other acquisitions, which were immaterial both individually and in the aggregate, and foreign currency translation adjustments related to goodwill.
56
Intangible assets with definite lives
Intangible assets are originally recorded at historical cost, in the case of separately purchased intangible assets, or at estimated fair value, in the case of assets acquired in the purchase of a business, and all of the Company’s intangible assets are subject to amortization ratably over their estimated useful lives. Intangible assets consist of the following (in thousands):
December 31, | Estimated | |||||||||
2007 | 2006 | Useful Life | ||||||||
| Intellectual property rights | $ | 30,826 | $ | 31,300 | 8 years | |||||
| Patents | 11,700 | 11,700 | 8 years | |||||||
| Trade name/trademarks | 6,800 | 4,800 | 7-8 years | |||||||
| Distribution/customer relationships and backlog | 6,042 | 5,785 | 1-5 years | |||||||
| Licensing, employment and non-competition agreements | 2,588 | 1,685 | 1-10 years | |||||||
| 57,956 | 55,270 | |||||||||
| Accumulated amortization | (20,251 | ) | (19,288 | ) | ||||||
| Net intangible assets | $ | 37,705 | $ | 35,982 | ||||||
Total amortization expense for the years ended December 31, 2007, 2006 and 2005 was approximately $7.2 million, $7.2 million and $6.2 million, respectively.
Estimated future amortization expense is as follows (in thousands):
| 2008 | $ | 10,538 | ||
| 2009 | 6,737 | |||
| 2010 | 6,630 | |||
| 2011 | 6,608 | |||
| 2012 | 5,205 | |||
| Thereafter | 1,987 | |||
| $ | 37,705 | |||
NOTE 8 - ACCRUED LIABILITIES
The following summarizes the Company’s accrued liabilities (in thousands):
December 31, | |||||||
2007 | 2006 | ||||||
| Accrued liabilities: | |||||||
| Accrued acquisition costs | $ | 1,639 | $ | 14,992 | |||
| Accrued dealer commissions | 2,592 | 2,816 | |||||
| Accrued legal fees | 502 | 3,225 | |||||
| Accrued professional fees and other | 8,176 | 5,314 | |||||
| $ | 12,909 | $ | 26,347 | ||||
57
NOTE 9 - COMMITMENTS AND CONTINGENCIES
Operating Leases
The Company leases certain facilities and equipment under operating leases. The Company recognizes rent expense on a straight-line basis over the lease term. At December 31, 2007, total future minimum lease payments were as follows (in thousands):
| 2008 | $ | 3,158 | ||
| 2009 | 2,499 | |||
| 2010 | 2,152 | |||
| 2011 | 1,977 | |||
| 2012 | 1,605 | |||
| Thereafter | 3,977 | |||
| $ | 15,368 | |||
Rent expense was $5.6 million, $3.5 million and $2.1 million in 2007, 2006 and 2005, respectively.
Purchase Commitments
In the ordinary course of business, the Company enters into agreements to purchase materials in the future that are enforceable, legally binding and specify terms with fixed or minimum quantities to be purchased and the approximate timing of the payments. At December 31, 2007, total future minimum purchase commitments with suppliers were as follows (in thousands):
| 2008 | $ | 6,297 | ||
| 2009 | 2,174 | |||
| 2010 | 1,914 | |||
| 2011 | 1,784 | |||
| 2012 | — | |||
| $ | 12,169 | |||
Warranties
The Company generally provides customers with a limited 90-day warranty on devices sold and a limited one-year warranty on controller units sold. ArthroCare accrues for the estimated cost of product warranties at the time revenue is recognized. The Company’s warranty obligation is affected by product failure rates, material usage and service delivery costs incurred in correcting a product failure. ArthroCare periodically evaluates and adjusts the warranty reserve to the extent actual warranty expense varies from the original estimates.
The following table describes the activity in ArthroCare’s warranty accrual (in thousands):
Year ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Balance at beginning of year | $ | 250 | $ | 175 | $ | 207 | ||||
| Accruals for warranties issued during the period | 1,856 | 1,078 | 929 | |||||||
| Settlements made during the period | (1,665 | ) | (1,003 | ) | (961 | ) | ||||
| Balance at end of year | $ | 441 | $ | 250 | $ | 175 | ||||
Litigation
From time to time, the Company is a defendant in certain lawsuits alleging product liability, patent infringement or other claims incurred in the ordinary course of business. These claims are generally covered by certain insurance policies, subject to certain deductible amounts and maximum policy limits. The Company establishes sufficient reserves to cover probable losses associated with such claims. Except as otherwise described above, the Company has product liability insurance coverage in amounts it considers necessary to prevent material losses. The Company recognizes losses when they are known or considered probable and the amount can be reasonably estimated.
58
Defending and prosecuting intellectual property suits, United States Patent and Trademark office, or USPTO, interference proceedings and related legal and administrative proceedings is generally costly and time-consuming. Further litigation may be necessary to enforce the Company’s patents, to protect its trade secrets or know-how or to determine the enforceability, scope and validity of the proprietary right of others. Any litigation or interference proceedings will be costly and will result in significant diversion of effort by technical and management personnel. An adverse determination in any of the litigation or interference proceedings to which ArthroCare may become a party could subject us to significant liabilities to third parties, require us to license disputed rights from third parties or require us to cease using such technology, which would have a material adverse effect on the Company’s business, financial condition, results of operations and future growth prospects. Patent and intellectual property disputes in the medical device area have often been settled through licensing or similar arrangements, and could include ongoing royalties. There can be no assurance ArthroCare can obtain any necessary licenses on satisfactory terms, if at all.
Contingent Consideration
Certain of the Company’s agreements for purchase business combinations and intangible asset purchases include provisions to provide additional consideration if contractually specified conditions related to the acquired business or assets are achieved. At December 31, 2007, liabilities have been recorded for conditions which have been met or are expected to be met based on current information available.
NOTE 10 - DEBT
On January 13, 2006, the Company entered into a Revolving Credit Agreement (the “Credit Agreement”) with a syndicate of banks (collectively, the “Lenders”). Under the terms of the Credit Agreement, the Company may borrow up to $100.0 million under a revolving line of credit from the named Lenders at the lead Lender’s Prime Rate or the British Bankers Association LIBOR Rate, plus a spread. The spread over the Prime Rate or LIBOR Rate is determined by the Company’s leverage ratio, as defined in the Credit Agreement. In connection with the Company’s repurchase of outstanding common stock, the Company borrowed $60.0 million in December 2007. At December 31, 2007, the Company had $60.0 million in outstanding balances under the Credit Agreement and the weighted average interest rate applicable to borrowings under the Credit Agreement was 5.5 percent. The estimated fair value of this debt approximates the carrying value presented in our consolidated balance sheet based on discounting the expected future cash flows using current market rates as of December 31, 2007.
Future contractual debt payments are as follows:
| 2008 | $ | — | ||
| 2009 | — | |||
| 2010 | — | |||
| 2011 | 60,000 | |||
| 2012 | — | |||
| $ | 60,000 | |||
The Credit Agreement has a five-year maturity and includes additional terms under which the Company may request an increase of up to $75.0 million in commitments from the Lenders, as business needs dictate. The Credit Agreement also contains various covenants that specify minimum or maximum financial ratios. The Company was in compliance with all such covenants at December 31, 2007.
NOTE 11 - STOCKHOLDERS’ EQUITY
Preferred Stock
As of December 31, 2007, 5,000,000 shares of preferred stock were authorized and no preferred stock was issued and outstanding.
59
Treasury Stock
In 2007, the Board of Directors authorized the purchase of up to $75.0 million in shares of the Company’s common stock. In December 2007, the Company purchased 1,255,982 treasury shares at a cost of $59.6 million and in January 2008, the Company purchased an additional 243,918 treasury shares at a cost of $11.8 million. The Company repurchased shares based on an evaluation of the Company’s long-term cash forecast and balance sheet capacity, including anticipated capital requirements.
In 2007 and 2006, the Company issued 12,102 shares at a value of $0.6 million and 12,633 shares at a value of $0.5 million, respectively, in order to fulfill the Company’s obligations to match employees’ 401(k) contributions with shares of Company stock.
Stock Option Plans
In August 1999, the Company adopted the Nonstatutory Option Plan, or the 1999 Plan. In June 2001, it authorized an amendment to the plan, effective April 26, 2001, increasing the aggregate number of shares authorized under the 1999 Plan to 3,550,000. As of December 31, 2007, 19,906 shares remained available for future grant under the 1999 Plan.
In May 2003, the Company adopted the 2003 Incentive Stock Plan, or the 2003 Plan, under which the Board of Directors is authorized to grant incentive and nonstatutory stock option awards and restricted stock awards to employees and consultants. In May 2004, the Company’s stockholders approved an amendment to the 2003 Plan to increase the number of shares available for issuance by 750,000 shares, to a total of 1,250,000 shares. In May 2006, the Company’s stockholders approved an amendment to the 2003 plan to increase the number of shares available for issuance by 1,250,000, to a total of 2,500,000. Options granted under the 2003 Plan generally become exercisable over a 48-month period and restricted stock awards generally become exercisable over a 60-month period. As of December 31, 2007, the Company had 731,245 shares available for future grant under the 2003 Plan.
The 1999 Plan and the 2003 Plan authorize the Company to issue Stock Appreciation Rights, or SARs, to directors, employees and consultants of the Company. SARs granted under the plans have an expiration term of seven years and generally become exercisable over a 48 month period.
All options and awards granted under each of the Company’s plans have a legal life of seven years, effective February 2005. Prior to that, options granted had a legal life of 10 years from the grant date.
Employee Stock Purchase Plan. In December 1995, ArthroCare approved the ESPP and reserved 300,000 shares of common stock for issuance thereunder. In May 2004, the Company’s stockholders approved an amendment to the ESPP to increase the number of shares available for issuance by 150,000 shares, to a total of 450,000 shares. Under the plan, regular full-time employees (subject to certain exceptions) may contribute up to 10 percent of base compensation to the semi-annual purchase of shares of ArthroCare common stock. In February 2006, the Company amended the ESPP to increase the purchase price from 85 percent to 95 percent of the fair market value at certain plan-defined dates. During the years ended December 31, 2007 approximately 7,461 shares were issued under the Company’s ESPP. As of December 31, 2007, approximately 72,756 shares remained available for future grant under the ESPP.
60
Plan Activity
Stock option and stock appreciation rights activity for all plans for the years ended December 31, 2007, 2006 and 2005 is as follows (in thousands, except per share data):
Awards Outstanding | |||||||
Number of Shares | Weighted-Average Exercise Price | ||||||
| Balance as of December 31, 2004 | 5,361,319 | $ | 17.40 | ||||
| Awards granted | 609,384 | 31.55 | |||||
| Awards exercised | (1,020,303 | ) | 16.12 | ||||
| Awards canceled/forfeited | (131,345 | ) | 18.30 | ||||
| Balance as of December 31, 2005 | 4,819,055 | 19.50 | |||||
| Awards granted | 459,034 | 45.23 | |||||
| Awards exercised | (1,877,759 | ) | 14.93 | ||||
| Awards canceled/forfeited | (224,069 | ) | 31.43 | ||||
| Balance as of December 31, 2006 | 3,176,261 | 24.94 | |||||
| Awards granted | 647,264 | 37.92 | |||||
| Awards exercised | (747,827 | ) | 20.48 | ||||
| Awards canceled/forfeited | (80,453 | ) | 28.74 | ||||
| Balance as of December 31, 2007 | 2,995,245 | 28.78 |
Net cash proceeds from the exercise of stock option awards were $14.3 million, $28.1 million, and $16.5 million for the years ended December 31, 2007, 2006 and 2005, respectively.
For the year ended December 31, 2007, there was $13.8 million of total unrecognized compensation expense related to unvested share-based compensation arrangements granted under the Company’s stock plans related to stock option awards. That cost is expected to be recognized over a weighted-average period of 1.4 years. As of December 31, 2007, 2.8 million awards were vested or expected to vest with a weighted average exercise price of $28.26. At December 31, 2007, the aggregate intrinsic value associated with these options was $56.3 million.
Awards outstanding and currently exercisable by exercise price for all plans at December 31, 2007 were as follows:
Outstanding Awards | Exercisable Awards | |||||||||||||||
Exercise Price | Number Outstanding | Weighted Average Remaining Contractual Life | Weighted Average Exercise Price | Number Exercisable As of December 31, 2007 | Weighted Average Exercise Price | |||||||||||
| $5.56 - $13.29 | 278,564 | 4.23 | $ | 11.28 | 278,564 | $ | 11.28 | |||||||||
| $13.31 - $17.30 | 256,937 | 4.85 | $ | 14.41 | 256,937 | $ | 14.41 | |||||||||
| $17.38 - $22.46 | 260,598 | 3.46 | $ | 19.31 | 252,976 | $ | 19.25 | |||||||||
| $22.60 - $24.38 | 320,449 | 4.25 | $ | 23.36 | 308,675 | $ | 23.33 | |||||||||
| $24.50 - $27.56 | 336,102 | 4.99 | $ | 26.33 | 318,715 | $ | 26.32 | |||||||||
| $27.75 - $31.00 | 297,159 | 5.99 | $ | 30.14 | 206,078 | $ | 30.24 | |||||||||
| $31.50 - $35.71 | 198,932 | 4.31 | $ | 33.10 | 134,875 | $ | 33.39 | |||||||||
| $36.08 - $39.92 | 563,317 | 6.11 | $ | 37.04 | 96,303 | $ | 37.17 | |||||||||
| $39.96 - $46.63 | 296,967 | 5.70 | $ | 42.96 | 94,217 | $ | 43.20 | |||||||||
| $46.84 - $54.15 | 186,220 | 5.33 | $ | 47.42 | 65,306 | $ | 46.92 | |||||||||
| 2,995,245 | 5.05 | $ | 28.78 | 2,012,646 |
61
During the year ended December 31, 2007, the Company issued 142,614 restricted common stock shares, which were valued at $6.1 million when reduced by estimated forfeitures. These awards vest over a period of five years. Restricted stock awards and units activity for the years ended December 31, 2007, 2006 and 2005, is as follows:
Number of Shares | Weighted- Average Grant Date Fair Value | ||||||
| Unvested at December 31, 2004 | 191,430 | $ | 16.12 | ||||
| Awards granted | 106,731 | 31.68 | |||||
| Awards vested | (22,699 | ) | 23.76 | ||||
| Awards canceled/forfeited | (6,000 | ) | 12.61 | ||||
| Unvested at December 31, 2005 | 269,462 | 20.84 | |||||
| Awards granted | 127,429 | 45.34 | |||||
| Awards vested | (65,400 | ) | 24.78 | ||||
| Awards canceled/forfeited | (42,767 | ) | 21.92 | ||||
| Unvested at December 31, 2006 | 288,724 | 19.21 | |||||
| Awards granted | 142,614 | 46.17 | |||||
| Awards vested | (63,919 | ) | 45.66 | ||||
| Awards canceled/forfeited | (8,549 | ) | 31.01 | ||||
| Unvested at December 31, 2007 | 358,870 | 16.46 | |||||
At December 31, 2007, there was total unrecognized compensation expense of $9.7 million, net of estimated forfeitures, related to unvested restricted stock awards and units. At December 31, 2007, unrecognized compensation expense was expected to be recognized over a weighted-average period of 2.9 years. The aggregate intrinsic value associated with the restricted stock awards was $14.6 million.
Valuation and Expense Information
In the year ended December 31, 2007, the Company recognized total stock-based compensation expense of approximately $10.2 million in our consolidated financial statements, which included approximately $7.5 million for employee stock options and stock appreciation rights and approximately $2.7 million for restricted stock awards and units. In the year ended December 31, 2006, the Company recognized total stock-based compensation expense of approximately $9.8 million, which included approximately $8.0 million for employee stock options and stock appreciation rights and approximately $1.8 million for restricted stock awards and units. Prior to its adoption of FAS 123R on January 1, 2006, the Company did not recognize expense related to employee stock options having exercise prices equal to the fair value of the stock.
62
The following table summarizes the total stock-based compensation expense resulting from employee stock options, including stock appreciation rights, and restricted stock awards and units that the Company recorded in accordance with the provisions of FAS 123R for the years ended December 31, 2007 and 2006. (in thousands):
Year Ended December 31, | |||||||||||||
2007 | 2006 | ||||||||||||
Employee Stock Options | Restricted Stock Awards and Units | Employee Stock Options | Restricted Stock Awards and Units | ||||||||||
| Cost of product sales | $ | 709 | $ | 210 | $ | 618 | $ | 150 | |||||
| Research and development | 1,111 | 498 | 1,308 | 320 | |||||||||
| Sales and marketing | 2,170 | 856 | 2,720 | 412 | |||||||||
| General and administrative | 3,491 | 1,152 | 3,315 | 942 | |||||||||
| Stock-based compensation expense before income taxes | 7,481 | 2,716 | 7,961 | 1,824 | |||||||||
| Income tax benefit | 2,522 | 918 | 2,581 | 677 | |||||||||
| Total stock-based compensation expense after income taxes | $ | 4,959 | $ | 1,798 | $ | 5,380 | $ | 1,147 | |||||
At December 31, 2007 and 2006, approximately $0.3 million and $0.2 million, respectively, of stock-based compensation cost related to employee awards was capitalized into inventory based on units produced and will be amortized to cost of product sales based on units sold.
The income tax benefit realized from stock option exercises was $10.7 million for the year ended December 31, 2007 and $13.8 million for the year ended December 31, 2006. In accordance with FAS 123R, the Company presents excess tax benefits from the exercise of stock options, if any, as financing cash flows rather than operating cash flows.
The weighted-average fair value of options granted to employees during the years ended December 31, 2007 and 2006 were $12.21 and $15.39, respectively. The fair value of each stock option was estimated on the grant date using the Black-Scholes option-pricing model with the following weighted-average assumptions for the years ended December 31, 2007 and 2006:
2007 | 2006 | ||||||
| Expected term (in years) (1) | 3.8 | 3.8 | |||||
| Expected volatility (2) | 33 | % | 36 | % | |||
| Risk-free interest rate (3) | 4.7 | % | 4.7 | % | |||
| Expected dividends | — | — | |||||
| (1) | The expected term assumption for options was determined based on historical data, adjusted for the recent reduction of the contractual life for options from 10 to seven years. |
| (2) | The expected volatility was determined using a blend of implied volatility and historical volatility over the expected term, which the Company considers a better indictor of expected volatility than using only historical volatility. |
| (3) | The risk-free interest rate is based upon observed interest rates appropriate for the term of the Company’s awards. |
Stock-based compensation expense recognized in the consolidated statement of operations is based on awards ultimately expected to vest. Consequently, it has been reduced for estimated forfeitures at a rate of 6.6 percent. FAS 123R requires forfeitures to be estimated at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. Forfeitures have been estimated based on the Company’s historical experience. Prior to fiscal 2006, the expected term and expected volatility of stock options were primarily based upon historical data. Forfeitures of employee stock options were accounted for on an as-incurred basis.
63
The weighted average fair value of purchase rights granted under the ESPP during the years ended December 31, 2007, 2006 and 2005 were $6.20, $6.79 and $8.93, respectively. The following assumptions were used in determining the fair value associated with shares granted under the ESPP as estimated on the date of grant using the Black-Scholes model with the following weighted average assumptions, or range of assumptions:
2007 | 2006 | 2005 | ||||||||
| Expected term (in years) | 0.5 years | 0.5 years | 0.5 years | |||||||
| Expected volatility | 35 | % | 35 | % | 46 | % | ||||
| Risk-free interest rate | 4.8 | % | 5.2 | % | 2.4 - 3.1 | % | ||||
| Expected dividends | — | — | — | |||||||
The following table illustrates net income and net income per share as if the Company had adopted the fair value recognition provision of FAS 123 for the year ended December 31, 2005 (in thousands, except per share data):
2005 | ||||
| Net income—as reported | $ | 23,530 | ||
| Add: Stock-based employee compensation expense recognized in net income (loss), net of tax effects | 751 | |||
| Less: Total employee stock-based compensation expense determined under the fair value method for all awards, net of related tax effects | (8,462 | ) | ||
| Pro forma net income | $ | 15,819 | ||
| Net income per share: | ||||
| Basic-as reported | $ | 0.97 | ||
| Basic-pro forma | 0.65 | |||
| Diluted-as reported | 0.89 | |||
| Diluted-pro forma | 0.60 | |||
The weighted-average fair value of options granted to employees during the year ended December 31, 2005 was $17.59 per share. In determining the pro forma net income for the year ended December 31, 2005, the fair value of each employee stock option grant was estimated on the date of grant using the Black-Scholes model with the following weighted average assumptions, or range of assumptions:
2005 | ||||
| Risk-free interest rate | 3.6% - 4.1 | % | ||
| Expected life | 5 years | |||
| Expected dividends | — | |||
| Expected volatility | 63 | % | ||
NOTE 12 - RELATED PARTIES
In June 1997, the Company loaned an officer $500,000 pursuant to a provision in the officer’s employment agreement. The promissory note, which bore no interest, was collateralized by a mortgage on the officer’s residence and was due and payable upon either the officer’s termination of employment or the sale of the officer’s residence. If the Company terminated the officer or if it is acquired, the loan was due and payable within 12 months thereafter. In January 2007, the officer repaid the loan in its entirety.
64
NOTE 13 - INCOME TAXES
The income tax provision consisted of the following (in thousands):
Year Ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Current | ||||||||||
| Federal | $ | 10,897 | $ | 11,241 | $ | 1,852 | ||||
| State | 1,246 | 2,353 | 766 | |||||||
| Foreign | 607 | 971 | 3,379 | |||||||
| Total current | 12,750 | 14,565 | 5,997 | |||||||
| Deferred | ||||||||||
| Federal | 162 | (5,686 | ) | 4,571 | ||||||
| State | (992 | ) | (1,578 | ) | (1,402 | ) | ||||
| Foreign | 259 | 1,685 | (1,973 | ) | ||||||
| Total deferred | (571 | ) | (5,579 | ) | 1,196 | |||||
| Total income tax provision | $ | 12,179 | $ | 8,986 | $ | 7,193 | ||||
The income tax provision differed from a provision computed at the U.S. statutory tax rate as follows (in thousands):
Year Ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Statutory rate tax provision | $ | 19,376 | $ | 14,231 | $ | 10,754 | ||||
| State income taxes | 1,291 | 1,040 | 870 | |||||||
| Differences in foreign tax rates | (8,363 | ) | (5,934 | ) | (4,088 | ) | ||||
| Nondeductible expenses | 257 | 214 | 172 | |||||||
| Research and development credits | (1,291 | ) | (1,269 | ) | (1,304 | ) | ||||
| Stock compensation | 435 | 456 | — | |||||||
| Charges for acquired IPR&D projects | — | — | 840 | |||||||
| Other | 474 | 248 | (51 | ) | ||||||
| Total income tax provision | $ | 12,179 | $ | 8,986 | $ | 7,193 | ||||
Of the Company’s 2007 pre-tax earnings of $55.4 million, $26.7 million was related to foreign tax jurisdictions. For 2006, these amounts were $40.7 million and $21.4 million, respectively. For 2005, these amounts were $30.7 million and $12.8 million, respectively.
The Company’s 2007 tax expense was significantly reduced due to a tax holiday for its Costa Rica manufacturing operations. Under its holiday, the Company will not be subject to Costa Rican income tax until 2010. At that time, the Company will be subject to normal Costa Rican corporate taxes, which are currently imposed at a rate of 30 percent.
Had the Company not benefited from this tax holiday, its 2007 tax liability would have been increased by $9.0 million and diluted earnings per share would have been decreased by $0.31. For 2006, the income related to Costa Rica would have increased the Company’s tax liability by $5.5 million and decreased earnings per share by $0.20. For 2005, the income related to Costa Rica would have increased the Company’s tax liability by $2.6 million and decreased earnings per share by $0.11.
The Company’s cash tax payments during 2007 were limited to $4.9 million due primarily to stock option benefits and prior year overpayments that have been applied to estimated taxes.
65
At December 31, 2007, the Company had federal net operating loss carryforwards of approximately $17.6 million, $14.1 million of which will be subject to an annual utilization limitation of approximately $1.7 million under Section 382 of the Internal Revenue Code. These loss carryforwards will expire during the period 2011 through 2023 if not utilized.
At December 31, 2007, the Company had federal and state research and development credit carryforwards of approximately $7.7 million and $7.9 million, respectively. These federal credits will expire during the period 2009 through 2027; the state credits are not subject to expiration.
Under the Internal Revenue Code, certain substantial changes in the Company’s ownership could result in an annual limitation on the amount of net operating loss carryforwards and income tax credits that could be utilized to offset future tax liabilities.
The Company’s deferred tax assets and liabilities consist of the following (in thousands):
December 31, | |||||||
2007 | 2006 | ||||||
| Deferred tax assets (liabilities): | |||||||
| Net operating loss carryforwards | $ | 4,923 | $ | 10,151 | |||
| R&D and MIC credits | 9,878 | 8,723 | |||||
| Allowances and reserves | 2,636 | 2,051 | |||||
| Deferred intercompany transactions | 130 | 472 | |||||
| Alternative minimum tax credits | 840 | 362 | |||||
| Stock compensation | 4,028 | 3,130 | |||||
| Cost recovery | 222 | (307 | ) | ||||
| Non-goodwill intangible assets | (8,702 | ) | (11,343 | ) | |||
| Other | 478 | (1,435 | ) | ||||
| Net deferred tax assets | $ | 14,433 | $ | 11,804 | |||
Income tax benefits resulting from the exercise of employee stock options of $10.7 million, $13.8 million and $9.6 million were credited to additional paid-in capital for 2007, 2006 and 2005, respectively.
Deferred U.S. income taxes and foreign withholding taxes are not provided on the undistributed cumulative earnings of foreign subsidiaries because those earnings are considered to be permanently reinvested in those operations. The permanently reinvested undistributed earnings were approximately $58.2 million as of December 31, 2007. These earnings could become subject to additional tax if remitted (or deemed remitted) as a dividend to the United States parent company; the resulting tax liability would be approximately $19.6 million.
A reconciliation of the Company’s beginning and ending amount of unrecognized tax benefits is as follows (in thousands):
| Balance at January 1, 2007 | $ | 7,924 | ||
| Additions for tax positions of current period | 527 | |||
| Balance at December 31, 2007 | $ | 8,451 |
66
The Company is subject to audit by the IRS and California Franchise Tax Board for the years 1994 through 2007 and by the Texas State Comptroller for the years 2006 and 2007. As the Company has operations in most other US states, other state tax authorities may assess deficiencies related to prior year activities; however, the years open to assessment vary with each state. The Company also files income tax returns for non-US jurisdictions; the most significant of which are the UK, Sweden, and Germany. The years open to adjustment for the UK and Germany are 2003 through 2007. The years open to adjustment for Sweden are 1999 through 2007.
In February 2008, the Company submitted an application with the IRS for an Advanced Pricing Agreement, or APA, relating to the transfer pricing policies between the Company and its foreign subsidiaries. Discussions and conclusions reached with the IRS may impact the Company’s determination of related services for prior periods.
NOTE 14 - EMPLOYEE BENEFIT PLAN
ArthroCare maintains a Retirement Savings and Investment Plan (“401(k) Plan”), which covers all United States based employees. Eligible employees may defer salary (before tax) up to a federally specified maximum. Management, at its discretion, may make matching contributions on behalf of the participants in the 401(k) Plan. The Company matched approximately $0.6 million, $0.5 million and $0.1 million of employee contributions to the 401(k) Plan in 2007, 2006 and 2005, respectively.
NOTE 15 - SEGMENT INFORMATION
ArthroCare has organized its marketing and sales efforts based on four operating segments which are aggregated into one reportable segment—the development, manufacture and marketing of disposable devices for less invasive surgical procedures. Each of the Company’s business units has similar economic characteristics, technology, manufacturing processes, customers, distribution and marketing strategies, regulatory environments, and shared infrastructures. These business units are Sports Medicine (shoulder and knee arthroscopic products), ENT (to include ear, nose, throat and the Visage® cosmetic products), Spine (to include spinal and neuro surgery products) and Coblation Technology (to include gynecology, urology, laparoscopic, general surgical and cardiology products).
Product sales by business unit for the periods shown were as follows (in thousands):
Year Ended December 31, | |||||||||||||||||||||
2007 | 2006 | 2005 | |||||||||||||||||||
| Sports Medicine | $ | 189,588 | 61.6 | % | $ | 166,665 | 65.8 | % | $ | 139,868 | 67.7 | % | |||||||||
| ENT | 73,065 | 23.8 | % | 59,886 | 23.6 | % | 43,546 | 21.1 | % | ||||||||||||
| Spine | 44,653 | 14.5 | % | 26,635 | 10.5 | % | 23,110 | 11.2 | % | ||||||||||||
| Coblation Technology | 290 | 0.1 | % | 190 | 0.1 | % | 9 | 0.0 | % | ||||||||||||
| Total Product Sales | $ | 307,596 | 100.0 | % | $ | 253,376 | 100.0 | % | $ | 206,533 | 100.0 | % | |||||||||
67
Internationally, the Company markets and supports its products primarily through its subsidiaries and various distributors. Revenues attributed to geographic areas are based on the country in which subsidiaries are domiciled. Product sales by geography for the periods shown were as follows (in thousands):
2007 | 2006 | 2005 | |||||||||||||||||||
Year ended December 31: | |||||||||||||||||||||
| Product sales: | |||||||||||||||||||||
| Americas | $ | 243,209 | 79.1 | % | $ | 202,333 | 79.9 | % | $ | 164,072 | 79.4 | % | |||||||||
| United Kingdom | 17,561 | 5.7 | % | 12,478 | 4.9 | % | 11,863 | 5.7 | % | ||||||||||||
| Germany | 14,179 | 4.6 | % | 11,405 | 4.5 | % | 9,007 | 4.4 | % | ||||||||||||
| Rest of World | 32,647 | 10.6 | % | 27,160 | 10.7 | % | 21,591 | 10.5 | % | ||||||||||||
| Total product sales | $ | 307,596 | 100.0 | % | $ | 253,376 | 100.0 | % | $ | 206,533 | 100.0 | % | |||||||||
December 31: | |||||||||||||||||||||
| Long-lived assets: | |||||||||||||||||||||
| United States | $ | 36,843 | 73.5 | % | $ | 26,851 | 71.0 | % | $ | 23,305 | 68.4 | % | |||||||||
| Costa Rica | 9,400 | 18.8 | % | 7,685 | 20.3 | % | 5,347 | 15.7 | % | ||||||||||||
| Rest of World | 3,889 | 7.7 | % | 3,280 | 8.7 | % | 5,422 | 15.9 | % | ||||||||||||
| Total long-lived assets | $ | 50,132 | 100.0 | % | $ | 37,816 | 100.0 | % | $ | 34,074 | 100.0 | % | |||||||||
NOTE 16 - QUARTERLY FINANCIAL INFORMATION (Unaudited)
The following tables present certain unaudited consolidated quarterly financial information for each quarter in the years ended December 31, 2007 and 2006. In the Company’s opinion, this unaudited quarterly information has been prepared on the same basis as the consolidated financial statements and includes all adjustments necessary to present fairly the information for the periods presented.
| (in thousands, except per share data) | |||||||||||||
2007 | 1st Quarter | 2nd Quarter | 3rd Quarter | 4th Quarter | |||||||||
| Total revenues | $ | 73,743 | $ | 79,542 | $ | 78,461 | $ | 87,496 | |||||
| Gross profit | 52,987 | 57,839 | 57,261 | 66,431 | |||||||||
| Net income | 7,136 | 10,413 | 11,122 | 14,509 | |||||||||
| Basic net income per share | 0.26 | 0.38 | 0.40 | 0.52 | |||||||||
| Diluted net income per share | 0.25 | 0.37 | 0.39 | 0.50 | |||||||||
2006 | 1st Quarter | 2nd Quarter | 3rd Quarter | 4th Quarter | |||||||||
| Total revenues | $ | 62,481 | $ | 66,005 | $ | 64,695 | $ | 69,820 | |||||
| Gross profit | 44,776 | 46,249 | 44,762 | 50,376 | |||||||||
| Net income | 7,133 | 7,699 | 8,676 | 8,167 | |||||||||
| Basic net income per share | 0.28 | 0.29 | 0.33 | 0.31 | |||||||||
| Diluted net income per share | 0.26 | 0.28 | 0.31 | 0.29 | |||||||||
NOTE 17 - SUPPLEMENTAL CASH FLOW INFORMATION
Information below is in thousands:
Year ended December 31, | ||||||||||
2007 | 2006 | 2005 | ||||||||
| Cash paid for interest | $ | — | $ | 1,926 | $ | 1,027 | ||||
| Cash paid for income taxes | 4,902 | 1,254 | 114 | |||||||
| Non-cash investing and financing activities: | ||||||||||
| Stock issued for acquisition of a business | — | 8,054 | — | |||||||
68
Schedule II
ARTHROCARE CORPORATION
VALUATION AND QUALIFYING ACCOUNTS
(in thousands)
Balance at Beginning of Period | Additional Charged to Costs and Expenses | Deductions | Balance at End of Period | ||||||||||
Year Ended December 31, 2007 | |||||||||||||
| Deducted from asset accounts: | |||||||||||||
| Allowance for doubtful accounts and product returns | $ | 2,701 | $ | 2,498 | $ | (1,219 | ) | $ | 3,980 | ||||
| Inventory reserves | 4,138 | 3,760 | (2,125 | ) | 5,773 | ||||||||
Year Ended December 31, 2006 | |||||||||||||
| Deducted from asset accounts: | |||||||||||||
| Allowance for doubtful accounts and product returns | $ | 1,365 | $ | 1,887 | $ | (551 | ) | $ | 2,701 | ||||
| Inventory reserves | 1,455 | 4,191 | (1,508 | ) | 4,138 | ||||||||
Year Ended December 31, 2005 | |||||||||||||
| Deducted from asset accounts: | |||||||||||||
| Allowance for doubtful accounts and product returns | $ | 400 | $ | 1,111 | $ | (146 | ) | $ | 1,365 | ||||
| Inventory reserves | 1,000 | 856 | (401 | ) | 1,455 | ||||||||
69
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized:
| ARTHROCARE CORPORATION | ||
| a Delaware Corporation | ||
| By: | /s/ Michael A. Baker | |
Michael A. Baker | ||
President and Chief Executive Officer | ||
Date: February 29, 2008
Pursuant to the requirements of the Securities Exchange Act of 1934, the following persons on behalf of the Registrant and in the capacities and on the dates indicated have signed this Report below:
Signature | Title | Date | ||
| /s/ Michael A. Baker | President, Chief Executive Officer and | February 29, 2008 | ||
Michael A. Baker | Director (Principal Executive Officer) | |||
| /s/ Michael Gluk | Senior Vice President Finance, Chief | February 29, 2008 | ||
Michael Gluk | Financial Officer and Assistant Secretary | |||
| (Principal Financial and Accounting | ||||
| Officer) | ||||
| /s/ David F. Fitzgerald | Director | February 29, 2008 | ||
David F. Fitzgerald | ||||
| /s/ James Foster | Director | February 29, 2008 | ||
James Foster | ||||
| /s/ Peter L. Wilson | Director | February 29, 2008 | ||
Peter L. Wilson | ||||
| /s/ Tord B. Lendau | Director | February 29, 2008 | ||
Tord B. Lendau | ||||
| /s/ Barbara D. Boyan | Director | February 29, 2008 | ||
Barbara D. Boyan | ||||
| /s/ Terrence E. Geremski | Director | February 29, 2008 | ||
Terrence E. Geremski |
70
ARTHROCARE CORPORATION
INDEX TO EXHIBITS*
Exhibit Number | Exhibit Name | |
10.65 †† | Stock Purchase Agreement, dated as of December 31, 2007, by and among ArthroCare Corporation and DiscoCare, Inc. | |
| 21.1 | Subsidiaries of the Registrant. | |
| 23.1 | Consent of PricewaterhouseCoopers LLP, Independent Registered Public Accounting Firm. | |
| 31.1 | Certification of the Chief Executive Officer pursuant to Rule 13a-15(f) of the Exchange Act, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 31.2 | Certification of the Chief Financial Officer pursuant to Rule 13a-15(f) of the Exchange Act, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 32.1 | Certification of the Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | |
| 32.2 | Certification of the Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| †† | Confidential treatment has been requested as to portions of this exhibit. |
| * | Only exhibits actually filed are listed. Exhibits incorporated by reference are set forth in the exhibit listing included in Item 15 of the Report on Form 10-K. |