Exhibit 99.1

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Searchable text section of graphics shown above
[LOGO]
Process |
| Polymers |
| Products |
Overview | August 2004
[GRAPHIC]
Safe Harbor Statement
This presentation may include forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 as amended, and Section 21E of the Securities Exchange Act of 1934. We have based these forward-looking statements on our current expectations and projections about future events. These forward-looking statements are subject to known and unknown risks, uncertainties and assumptions about us and our affiliate companies, that may cause our actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by such forward-looking statements. Factors that might cause or contribute to such a discrepancy include, but are not limited to, those identified in our other Securities and Exchange Commission filing and other public documents, including our 2004 Annual Report on Form 10-KSB and our Quarterly Reports on Form 10-QSB, which can be found on our corporate Web site, www.cardiotech-inc.com.
[LOGO]
2
Company Overview
• Recognized leader in pioneering innovative devices that enhance the treatment of cardiovascular disease
• Manufacturing Capability
• Based on a deep knowledge of polymer-based materials (biomaterials)
• Manufactures and markets single-use, disposable cardio pulmonary bypass devices
• Develops, manufactures, and sells a range of biomaterials for use in the manufacture of medical devices
• Manufacturing for medical device companies
• Research and Development
• Advanced wound care products
• Drug Eluting Stents
• Synthetic Artery
3
Key Facts
Symbol |
| CTE (AMEX) |
Corporate Headquarters |
| Wilmington, MA |
Amex Listing |
| June 1996 |
Stock Price (7/20/04) and 52-Week Range |
| $3.89 || $3.20 to $7.20 |
Basic and Fully Diluted Shares Outstanding |
| 17.4mm || 23mm |
Float |
| 14.9mm |
Market Cap |
| $68mm |
Volume (daily 90-day average) |
| 68,102 |
Long-Term Debt (3/31/04) |
| $0 |
Insider Ownership Shares |
| 22% of fully diluted |
Institutional Ownership Shares |
| 12% of fully diluted |
Employees |
| 160 |
Fiscal Year End |
| March 31st |
4
Significant Events
Corporate History |
| |
|
| |
• | Spinoff from PolyMedica (NASDAQ: PLMD) | June 1996 |
| • To develop synthetic artery and wound dressings |
|
|
|
|
• | Acquisition of CDT | May 2001 |
| • To utilize biomaterials expertise in outsource market |
|
|
|
|
• | Started synthetic artery (Coronary Artery Bypass Graft - CABG) clinical trials in Brazil | July 2003 |
| • Goal to obtain approval in Europe and US |
|
|
|
|
Financings |
| |
|
| |
Common Stock | December 1998 | |
| • $2,500,000, proceeds used for working capital |
|
| • 1 common stock, 1 warrant ($2.75 per unit) |
|
5
Capital Structure
$ in 000s |
| FYE |
| |
|
|
|
|
|
Cash and Equivalents |
| $ | 7,117 |
|
|
|
|
| |
DEBT: |
|
|
| |
Senior Credit Facilities - RLOC |
| $ | 730 |
|
Other Senior Debt |
| $ | 0 |
|
|
|
|
| |
NET DEBT(1) |
| $ | 0 |
|
|
|
|
| |
TOTAL EQUITY |
| $ | 19,368 |
|
|
|
|
| |
TOTAL CAPITALIZATION |
| $ | 19,368 |
|
|
|
|
| |
DEBT / EQUITY |
| 0.0 | x | |
(1) Debt net of cash and equivalents
6
CTE Founder – Dr. Michael Szycher
• Expert in Biomaterials
• Recognized international authority on polyurethanes and blood compatible polymers
• Co-invented the HeartMate, the best-selling artificial heart on the market
• Developed the CardioPass synthetic coronary artery bypass graft
• Editor of five books (and the author of three) related to biomaterials and medical devices - widely used in graduate biomedical engineering classrooms
• Co-founded three successful companies
• Thermedics in 1983 (sold to Thoratec)
• PolyMedica in 1988 ($700 million market cap, $420 million revenue)
• CardioTech in 1996
• Educational Background
• BS in Chemical Engineering, University of New Hampshire
• PhD in Cardiac Physiology, Boston University School of Medicine
• MBA, Suffolk University
7
Senior Management
• Dr. Michael Szycher – Chairman & CEO
• Leslie Taeger – CFO
• Thomas Lovett – VP of Finance & Controller
• Douglas Whittaker – President of GISH
• Liann Johnson – General Manager of CDT
8
Corporate Organization
Manufacturing
• GISH Biomedical, Inc.
• Cardiopulmonary bypass devices
• FY04 Revenue of $17.7 million, Market Size of $500 million
• Catheter and Disposables Technology, Inc. (CDT)
• Outsourcing for medical device companies
• FY04 Revenue of $2.9 million, Market Size of $2 billion
Research and Development
• Total estimated FY05 R&D Expense of $1.3 million
• Biomaterials
• Development of proprietary biomaterials
• FY04 Revenue of $1.0 million, Market Size of $250 million
• Dermaphylyx, Inc. (Wound Care)
• Wound care and biomaterials
• FY04 Revenue of $25 thousand, Market Size of $600 million
• Drug Eluting Stents
• Development of a coronary drug eluting stent
• Market Size of $1 billion (international)
• Synthetic Artery
• Development of a Coronary Artery Bypass Graft (CABG)
• Market Size of $1 billion
9
GISH Biomedical - Manufacturing
• Acquired in 2003 for $8.7 million in an all stock transaction
• Custom cardiovascular tubing systems, arterial filters, oxygenators
• Competitive Advantages:
• Gish “GBS” heparin coating reduces platelet adhesion and reduces systemic inflammatory response syndrome (SIRS)
• Competition can only do one or the other – not both
• Market Characteristics
• $500 million worldwide market
• Declining market; Gish gaining market share
• Operating Results
($ in 000s) |
| Audited |
| Audited |
| Projected |
| ||
Revenue |
| n/a |
| $ | 17,781 |
| $ | 19,000 |
|
|
|
|
|
|
|
|
|
|
|
Gross Profit |
| n/a |
| $ | 4,582 |
| $ | 5,300 |
|
• Growth Strategy:
• Establish GBS Heparin Coating as “Gold Standard”
• Strategic Partnerships/Acquisitions
• Increase foreign sales
10
CDT - Manufacturing
• Acquired in 2001 for $1.3 million in cash
• Outsourcing for medical device companies: design, development and manufacturing of medical products based on molded and extruded components.
• Products include: sensing, balloon, and drug delivery catheters; disposable endoscopes; and in-vitro diagnostic and surgical disposables
• Customer-base consists of industry leading medical device suppliers (Fortune 100)
• Competitive Advantages:
• Offer unique mix of R&D and manufacturing capabilities
• Sales/Distribution performed by internal sales force
• Market Characteristics
• Highly Fragmented: Potential to cross-pollinate technologies via acquisition
• Operating Results
($ in 000s) |
| Audited |
| Audited |
| Projected |
| |||
Revenue |
| $ | 2,343 |
| $ | 2,966 |
| $ | 3,300 |
|
|
|
|
|
|
|
|
|
|
|
|
Gross Profit |
| $ | 964 |
| $ | 1,092 |
| $ | 1,300 |
|
• Growth Strategy
• Strategic acquisitions
• Partnership opportunities
11
CDT – Acquisition Strategy
• Candidate Attributes
Profitable operation
Outsourcing and manufacturing for the medical device market
Cross-selling opportunities
New customers and branded, quality products
• Transaction Mechanics
Revenue Size: |
| $5M to $10 MM |
Net Profit Margin: |
| 10% to 20% |
EBITDA Margin: |
| 15% to 30% |
Valuations: |
| 4x-5x EBITDA |
Purchase Method: |
| Combination of Cash and Stock |
12
Research and Development
• Biomaterials (polyurethanes, hyaluronic acid)
• Wound Care
• New Devices under development
• Drug Eluting Stents
• Synthetic Artery: Coronary Artery Bypass Graft (CABG)
13
Biomaterials – Research and Development
• Sells proprietary biomaterials to medical device manufacturers for use in their products
• Strategy
• Introduce biomaterials into the marketplace via customers’ FDA approved medical devices
• Develop a track record of safe usage with the FDA
• Introduce the synthetic artery with our proprietary knowledge
• Operating Results
($ in 000s) |
| Audited |
| Audited |
| Projected |
| |||
Revenue |
| $ | 1,051 |
| $ | 1,028 |
| $ | 1,200 |
|
|
|
|
|
|
|
|
|
|
|
|
Gross Profit |
| $ | 307 |
| $ | 441 |
| $ | 450 |
|
• Intellectual Property
• 42 patents
• Polymer-based knowledge
14
Wound Care – Research & Development
• Started marketing products in beginning of 2004
• Moist wound (for bleeding wounds) and Blister-preventing products
• Competitive Advantages:
• Stops pain from a wound within approximately 2 minutes
• Moist wound products absorb up to 400% of its weight in fluid
• Hydrogels: Intelligent dressing (either absorbs or donates moisture)
• Transparency allows more productive wound monitoring
• Market Characteristics
• $600 million annual worldwide market for Hydrogels and Alginates
• Sales and Distribution are performed in-house
• Manufacturing is outsourced
• Business Operating Results
($ in 000s) |
| Audited |
| Projected |
| |
Revenue |
| n/a |
| $ | 200 |
|
|
|
|
|
|
|
|
Gross Profit |
| n/a |
| $ | 80 |
|
• Growth Strategy
• Medicare Reimbursement
• Expand approvals into other market
15
Drug Eluting Stents – Research & Develop.
• Partnership with CorNova was formed to develop a coronary drug eluting stent using the capabilities and technologies of CorNova (40%), Implant Services (30%), and CTE (30%)
• CTE will contribute shares and technologies (no cash investment)
• Share contribution expected at $750 thousand or less
• Market Characteristics
• $1 billion annual international market
• 450,000 stents used worldwide annually (CTE will market $900 stents)
• Currently only two FDA approved drug eluting stents on the market
• Current competitors market their products in the US for $3,000 – do not compete in Europe due to limited reimbursement of only $900
• Growth Strategy
• Gain CE marking by FY 2007
• International market
16
Drug Eluting Stents – CTE Advantage
|
| Radio |
| In-House |
| Generic Drug |
| Price |
| |||||||
Company |
| Yes |
| Slight |
| Yes |
| No |
| Yes |
| No |
| Amount |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Boston Scientific |
|
|
| X |
|
|
| X |
|
|
| X |
| $ | 3,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Johnson & Johnson |
|
|
| X |
|
|
| X |
|
|
| X |
| $ | 3,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
CardioTech |
| X |
|
|
| X |
|
|
| X |
|
|
| $ | 900 |
|
Why is CTE better?
• More opaque than the competition – more visible on x-rays
• CTE owns the polymer – no royalty payments
• Utilizes a generic drug – no royalty payments
17
Synthetic Artery – Research & Development
• Synthetic Artery (CABG)
[GRAPHIC]
18
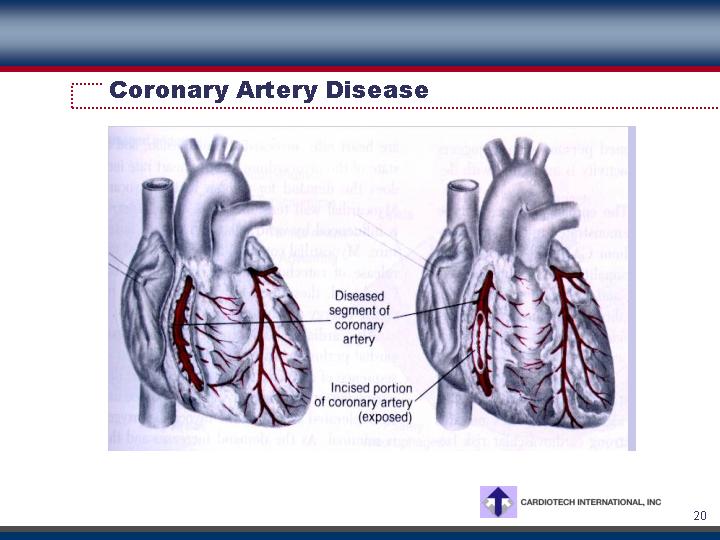
Coronary Artery Disease
Market Characteristics
• 60 million patients in the US
• 1 million deaths annually in the US
• Treatment: diet, lifestyle changes, drugs, angioplasty, stents, and coronary artery bypass graft surgery
• 650,000 annual CABG surgeries performed worldwide
• $1 billion worldwide market
19
Coronary Artery Disease
[GRAPHIC]
20
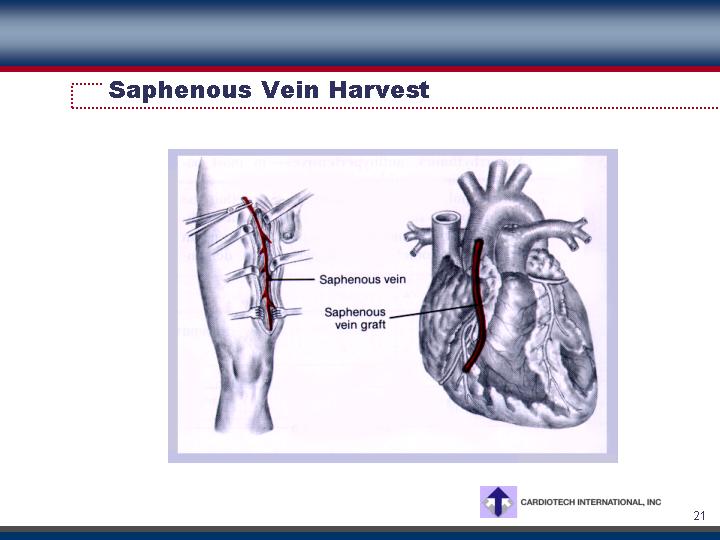
Saphenous Vein Harvest
[GRAPHIC]
21
Coronary Artery Bypass Graft (CABG)
• CABG surgery is performed to treat the impairment of blood flow to portions of the heart by rerouting blood around blocked coronary arteries
• Competitive Landscape:
• CABG Medical - Development stage company, product is reliant on drug combination
• Thoratec - Developer and manufacturer of medical devices, discarded CABG development attempts
• No other direct competitors at this time
• Why will CTE’s synthetic artery (CABG) work?
• Polyurethane transparent to the blood and immune system
• Elasticity
• Non-kinking
• Not reliant on drug combination
22
CTE Opportunity - CABG
• Only Company in the world in human trials with CABG, CTE should have the first synthetic artery on the market
• CABG Clinical Trial Approval Process
• Currently in human clinical trials in Brazil
• $1 million spent to date ($1.5 million total cost)
• Enter trials in Europe in late 2004 (two years to approval)
• $5 million budgeted to complete European trials
• FDA approval could be obtained within three years
• $10 million budgeted to complete the US trials
• Once Europe and US approvals received, CTE will pursue 200,000 annual “no option” patients worldwide
• CTE synthetic artery priced at approximately $1,500
• Potential use as substitute for autogenous saphenous veins
23
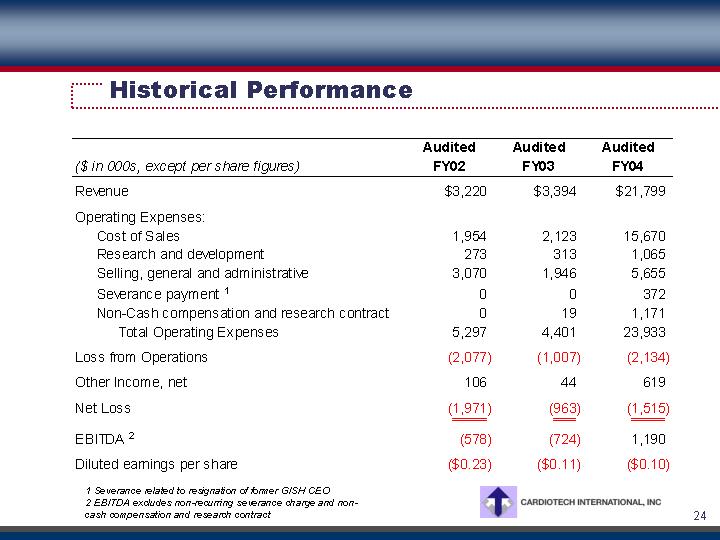
Historical Performance
($ in 000s, except per share figures) |
| Audited |
| Audited |
| Audited |
| |||
Revenue |
| $ | 3,220 |
| $ | 3,394 |
| $ | 21,799 |
|
|
|
|
|
|
|
|
| |||
Operating Expenses: |
|
|
|
|
|
|
| |||
Cost of Sales |
| 1,954 |
| 2,123 |
| 15,670 |
| |||
Research and development |
| 273 |
| 313 |
| 1,065 |
| |||
Selling, general and administrative |
| 3,070 |
| 1,946 |
| 5,655 |
| |||
Severance payment (1) |
| 0 |
| 0 |
| 372 |
| |||
Non-Cash compensation and research contract |
| 0 |
| 19 |
| 1,171 |
| |||
Total Operating Expenses |
| 5,297 |
| 4,401 |
| 23,933 |
| |||
|
|
|
|
|
|
|
| |||
Loss from Operations |
| (2,077 | ) | (1,007 | ) | (2,134 | ) | |||
|
|
|
|
|
|
|
| |||
Other Income, net |
| 106 |
| 44 |
| 619 |
| |||
|
|
|
|
|
|
|
| |||
Net Loss |
| (1,971 | ) | (963 | ) | (1,515 | ) | |||
|
|
|
|
|
|
|
| |||
EBITDA (2) |
| (578 | ) | (724 | ) | 1,190 |
| |||
|
|
|
|
|
|
|
| |||
Diluted earnings per share |
| $ | (0.23 | ) | $ | (0.11 | ) | $ | (0.10 | ) |
(1) Severance related to resignation of former GISH CEO
(2) EBITDA excludes non-recurring severance charge and non-cash compensation and research contract
24
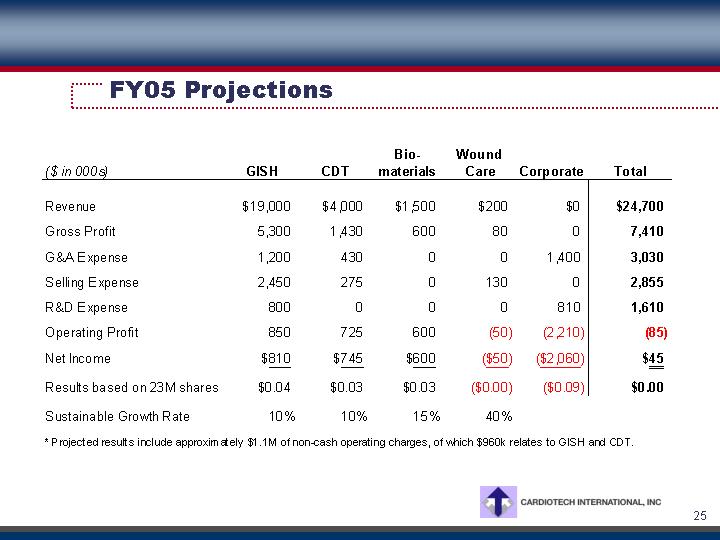
FY05 Projections
($ in 000s) |
| GISH |
| CDT |
| Bio- |
| Wound |
| Corporate |
| Total |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Revenue |
| $ | 19,000 |
| $ | 4,000 |
| $ | 1,500 |
| $ | 200 |
| $ | 0 |
| $ | 24,700 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Gross Profit |
| 5,300 |
| 1,430 |
| 600 |
| 80 |
| 0 |
| 7,410 |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
G&A Expense |
| 1,200 |
| 430 |
| 0 |
| 0 |
| 1,400 |
| 3,030 |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Selling Expense |
| 2,450 |
| 275 |
| 0 |
| 130 |
| 0 |
| 2,855 |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
R&D Expense |
| 800 |
| 0 |
| 0 |
| 0 |
| 810 |
| 1,610 |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Operating Profit |
| 850 |
| 725 |
| 600 |
| (50 | ) | (2,210 | ) | (85 | ) | ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Net Income |
| $ | 810 |
| $ | 745 |
| $ | 600 |
| $ | (50 | ) | $ | (2,060 | ) | $ | 45 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Results based on 23M shares |
| $ | 0.04 |
| $ | 0.03 |
| $ | 0.03 |
| $ | (0.00 | ) | $ | (0.09 | ) | $ | 0.00 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Sustainable Growth Rate |
| 10 | % | 10 | % | 15 | % | 40 | % |
|
|
|
| ||||||
* Projected results include approximately $1.1M of non-cash operating charges, of which $960k relates to GISH and CDT.
25
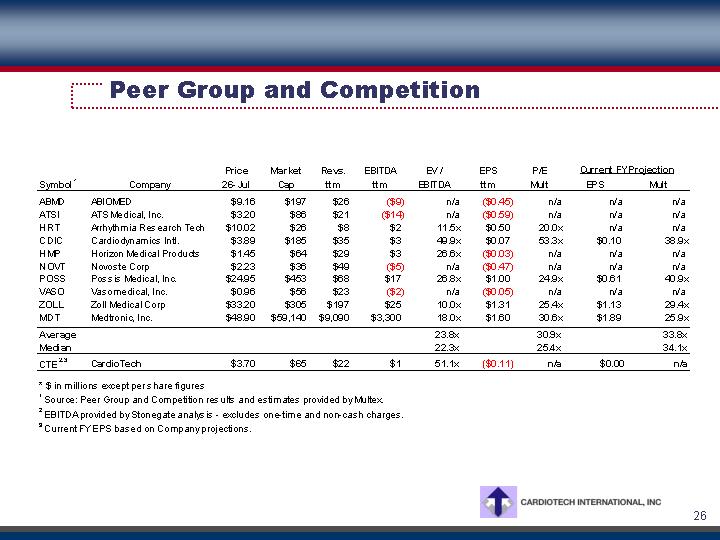
Peer Group and Competition
Symbol (1) |
| Company |
| Price |
| Market |
| Revs. |
| EBITDA |
| EV / |
| EPS |
| P/E |
| Current FY Projection |
| ||||||||
EPS |
| Mult | |||||||||||||||||||||||||
ABMD |
| ABIOMED |
| $ | 9.16 |
| $ | 197 |
| $ | 26 |
| $ | (9 | ) | n/a |
| $ | (0.45 | ) | n/a |
| n/a |
| n/a |
| |
ATSI |
| ATS Medical, Inc. |
| $ | 3.20 |
| $ | 86 |
| $ | 21 |
| $ | (14 | ) | n/a |
| $ | (0.59 | ) | n/a |
| n/a |
| n/a |
| |
HRT |
| Arrhythmia Research Tech |
| $ | 10.02 |
| $ | 26 |
| $ | 8 |
| $ | 2 |
| 11.5 | x | $ | 0.50 |
| 20.0 | x | n/a |
| n/a |
| |
CDIC |
| Cardiodynamics Intl. |
| $ | 3.89 |
| $ | 185 |
| $ | 35 |
| $ | 3 |
| 49.9 | x | $ | 0.07 |
| 53.3 | x | $ | 0.10 |
| 38.9 | x |
HMP |
| Horizon Medical Products |
| $ | 1.45 |
| $ | 64 |
| $ | 29 |
| $ | 3 |
| 26.6 | x | $ | (0.03 | ) | n/a |
| n/a |
| n/a |
| |
NOVT |
| Novoste Corp |
| $ | 2.23 |
| $ | 36 |
| $ | 49 |
| $ | (5 | ) | n/a |
| $ | (0.47 | ) | n/a |
| n/a |
| n/a |
| |
POSS |
| Possis Medical, Inc. |
| $ | 24.95 |
| $ | 453 |
| $ | 68 |
| $ | 17 |
| 26.8 | x | $ | 1.00 |
| 24.9 | x | $ | 0.61 |
| 40.9 | x |
VASO |
| Vasomedical, Inc. |
| $ | 0.96 |
| $ | 56 |
| $ | 23 |
| $ | (2 | ) | n/a |
| $ | (0.05 | ) | n/a |
| n/a |
| n/a |
| |
ZOLL |
| Zoll Medical Corp |
| $ | 33.20 |
| $ | 305 |
| $ | 197 |
| $ | 25 |
| 10.0 | x | $ | 1.31 |
| 25.4 | x | $ | 1.13 |
| 29.4 | x |
MDT |
| Medtronic, Inc. |
| $ | 48.90 |
| $ | 59,140 |
| $ | 9,090 |
| $ | 3,300 |
| 18.0 | x | $ | 1.60 |
| 30.6 | x | $ | 1.89 |
| 25.9 | x |
Average |
|
|
|
|
|
|
|
|
|
|
| 23.8 | x |
|
| 30.9 | x |
|
| 33.8 | x | ||||||
Median |
|
|
|
|
|
|
|
|
|
|
| 22.3 | x |
|
| 25.4 | x |
|
| 34.1 | x | ||||||
CTE (2),(3) |
| CardioTech |
| $ | 3.70 |
| $ | 65 |
| $ | 22 |
| $ | 1 |
| 51.1 | x | $ | (0.11 | ) | n/a |
| $ | 0.00 |
| n/a |
|
* $ in millions except per share figures
(1) Source: Peer Group and Competition results and estimates provided by Multex.
(2) EBITDA provided by Stonegate analysis - excludes one-time and non-cash charges.
(3) Current FY EPS based on Company projections.
26
Valuation Approach
• Manufacturing Valuation (GISH and CDT):
• EV / FY05 EBITDA multiple in-line with comparables
• Organic manufacturing growth of 10% annually
• Accretive acquisitions
• Valuation Upside:
• New products
• Potential blockbuster with synthetic artery (CABG)
• CABG Medical, Inc.: Pre-clinical development stage
• Based on recent S-1 filing, IPO is expected to raise $30+ million for 34% ownership (total company valued at $85+ million)
• Drug eluting stent
• Wound care
• Valuable Intellectual Property
• Includes 42 patents
27
Summary
• Biomaterials expertise
• Profitable niche manufacturing business
• Valuable Intellectual Property – includes 42 patents
• Manufacturing Valuation (GISH and CDT):
• EV / FY05 EBITDA multiple in-line with comparables
• Accretive manufacturing acquisitions
• Valuation Upside with Developed Products:
• Potential blockbuster with synthetic artery (CABG)
• Drug eluting stents
• Wound care
28