Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text
Click here for corresponding text

Click here for corresponding text
Click here for corresponding text
Click here for corresponding text

Click here for corresponding text
Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text
Click here for corresponding text
Click here for corresponding text
Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text
Click here for corresponding text

Click here for corresponding text

Click here for corresponding text
Click here for corresponding text
Click here for corresponding text
Click here for corresponding text
Click here for corresponding text
Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text

Click here for corresponding text
A Leading Provider of
Orthopaedic and Urologic
Non-Invasive Medical Devices
and Medical Services
This presentation contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve known and unknown risks, uncertainties or other factors which may cause actual results, performance or achievements of HealthTronics Surgical Services, Inc. to be materially different from any future results, performance or achievements express or implied by such forward-looking statements. As always, these expectations and projections are based on currently available competitive, financial, and economic data, along with operating plans, and are subject to future events and uncertainties. Among the events and uncertainties which could adversely affect future periods are: inability to establish or maintain relationships with physicians and hospitals; health care regulatory developments that prevent certain transactions with health care professionals or facilities; inability of the Company or health care providers to obtain reimbursement for use of the Company’s current or future products; competition or technological change that impacts the market for the Company’s products; and difficulty in managing the Company’s growth. Additional factors that might cause such a difference, include, but are not limited to those discussed in the Management’s Discussion and Analysis of Financial Condition and Results of Operations in the Company’s Annual Report on Form 10-K for the year ended December 31, 2002, and in subsequent documents filed by HealthTronics Surgical Services with the Securities and Exchange Commission.

2
• | Worldwide technology leader in Electrohydraulic Shock Wave medical devices | |
|
| |
• | A leading provider of non-invasive urologic services | |
| • | Lithotripsy – Leading treatment for kidney stones |
| • | TUMT – Treatment of benign prostate enlargement |
| • | Cryotherapy – Treatment for prostate cancer |
| • | Beginning FDA Clinical trials for New Prostate Treatment - HIFU |
|
| |
• | The U.S. pioneer of orthopaedic Extracorporeal Shock Wave Surgery | |
| • | OssaTron® is the first FDA approved device for treating orthopaedic conditions with orthopaedic shock wave surgery |
| • | OssaTron® is the only FDA approved device for multiple indications |
| • | First mover in estimated $2 billion market |

3
• | Leading manufacturer and distributor of lithotripsy and orthopaedic shock wave surgery devices worldwide | |
| • | Distribution in over 35 countries |
| • | Research and Manufacturing Facilities in Switzerland |
| • | In the U.S., approximately 177 devices providing services in 47 states |
|
| |
• | Relationship with approximately 15% of all U.S. urologists | |

4
• | Build infrastructure to support growth of Orthopaedic Extracorporeal Shock Wave Surgery in the U.S. and worldwide |
|
|
• | Continue generating cash flow and earnings from urology operations |
|
|
• | Establish new urology businesses and introduce new urology products by leveraging existing base of U.S. urology network |
|
|
• | Produce world-class products and increase penetration of worldwide medical devices |

5
• | Growing demand for non-invasive and minimally invasive surgical procedures |
|
|
• | Trend from health care facilities to outsource specialized services |
|
|
• | Aging of U.S. population and promotion of healthy, active lifestyle |

6
• | Turnkey Clinical and Technical Services provided to doctors, hospitals and patients |
|
|
• | Sale of Equipment – LithoTron®, LithoDiamond®, OssaTron®, EvoTron®* and VersaTron® |
|
|
• | Sale of Consumables |
|
|
• | Equipment Maintenance and Support |
|
|
• | Management Fees from Partnerships |
*Not approved by the FDA in the U.S. Market

7
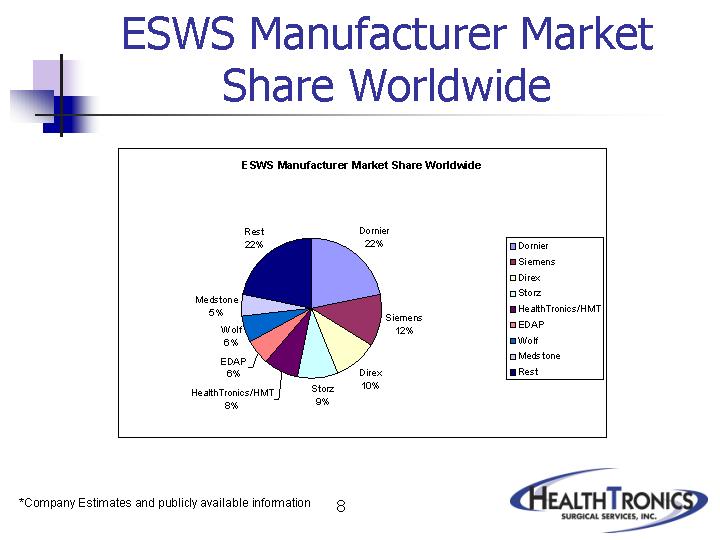
ESWS Manufacturer Market
Share Worldwide
Dornier |
|
| 22% |
|
Siemens |
|
| 12% |
|
Direx |
|
| 10% |
|
Storz |
|
| 9% |
|
HealthTronics/HMT |
|
| 8% |
|
EDAP |
|
| 6% |
|
Wolf |
|
| 6% |
|
Medstone |
|
| 5% |
|
Rest |
|
| 22% |
|
*Company Estimates and publicly available information

8
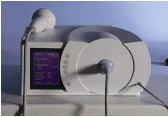
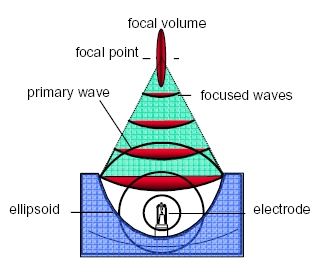
Electrohydraulic shock wave generation
|
|
| |
|
9
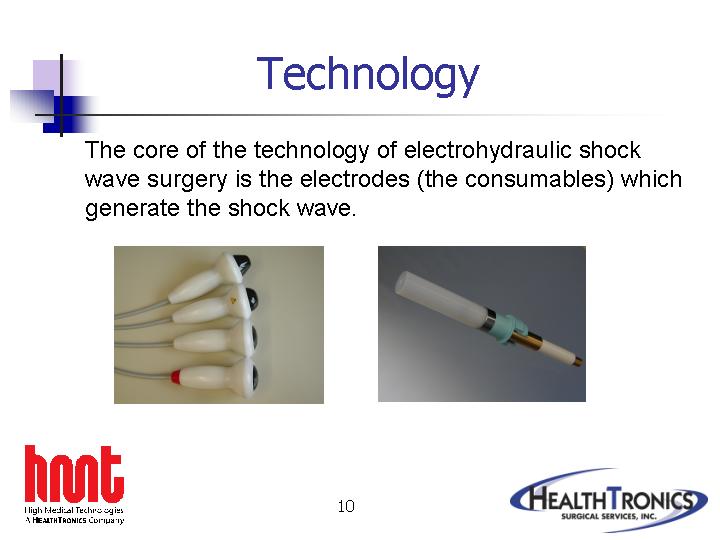
The core of the technology of electrohydraulic shock wave surgery is the electrodes (the consumables) which generate the shock wave.


|
|
| |
|
10
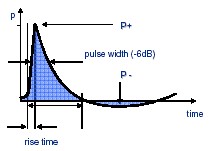
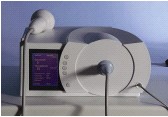
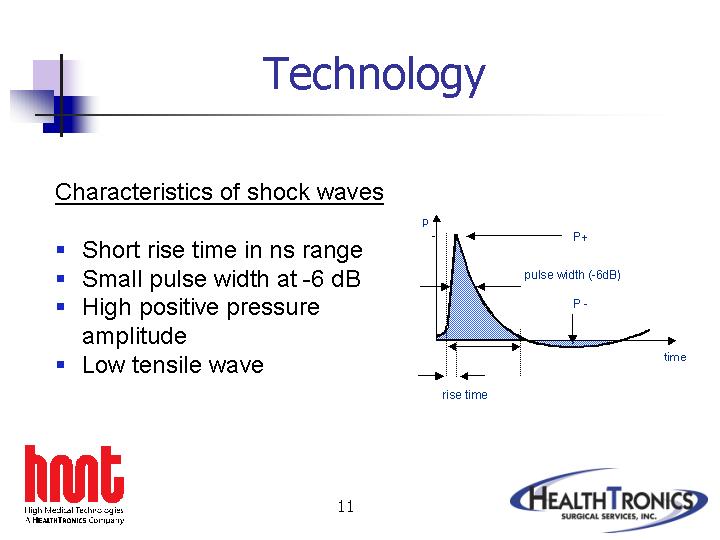
Characteristics of shock waves |
| |
|
| |
|
| |
• | Short rise time in ns range | |
• | Small pulse width at -6 dB | |
• | High positive pressure amplitude | |
• | Low tensile wave | |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
|
|
| |
| ||
| ||
11
Current Scientific and Clinical Studies
• | FDA study on non-union indication OssaTron® |
|
|
• | Pressure measurement (Boston University) |
|
|
• | Lateral epicondylitis study (Baylor University Hospital) |
|
|
• | Chip model study (Colorado State University) |
|
|
• | Multicenter study with LithoDiamond® |
|
|
• | Shoulder study with EvoTron® |
|
|
• | Clinical study and Scintigraphy (Zürich University) |
|
|
• | Clinical study on Osteoarthritis for small animal (dogs, cats) with VersaTron® |

12
|
|
|
LithoTron® |
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
LithoDiamond® |
| |
| ||
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|

13
Total Lithotripters Sold per Region
(HealthTronics and HMT)
GRAPHIC
Americas = 138
Europe = 96
Asia = 42

14
• | Accepted treatment protocol by physicians and insurers for treating kidney stones |
|
|
• | Approximately 250,000 cases per year |
|
|
• | Reimbursement averages $2,000 per procedure for HealthTronics |

15
• | Active with 44 lithotripsy entities (of which we consolidate 30) in 35 states |
|
|
• | Average entity now treating 800 cases per year |
|
|
• | HealthTronics Surgical Services’ average partnership stake is approximately 30% |
|
|
• | HealthTronics Surgical Services’ average equity stake in unconsolidated partnerships is approximately 15% |

16
• | Transurethral Microwave Therapy | |
| • | Treatment of benign prostate enlargement |
| • | Outpatient procedure |
| • | One of the largest providers in the U.S., projected to treat approximately 3,300 patients in 2004 |
| • | Partnership model does not generally apply |
|
| |
• | Cryosurgery (controlled freezing) | |
| • | Minimally invasive treatment of prostate cancer |
| • | Outpatient or limited stay procedure |
| • | 185,000 new cases in the U.S. each year |
| • | Better long-term outcomes compared to radiation; faster and less expensive |
| • | Partnership model applies |

17
BPH –BenignProstaticHypertrophy
• | Non-cancerous enlargement of the prostate |
|
|
• | Affects 50% of men over age 60 |
|
|
• | Symptoms associated with restricted flow of urine |
|
|
• | Multiple treatments available |

18
HealthTronics BPH Treatment Options
• | Trans Urethral Microwave Therapy (TUMT) | |
| • | In office |
| • | Single treatment – 30 minutes |
| • | Good Medicare reimbursement |
|
| |
• | Laser Excision | |
| • | In surgery center |
| • | Single treatment – 1 hour |
| • | Doctors like it (more like surgery) |

19
• | Second most common cancer in males | |
|
| |
• | 230,000 new cases per year in the U.S. | |
|
| |
• | PSA has changed the management of prostate cancer | |
|
| |
• | Five treatment options | |
| • | Surgery |
| • | Radiation |
| • | Cryosurgery |
| • | Medication |
| • | HIFU |

20
| Patient Concerns |
| Payor Concerns | ||
|
| ||||
| • | Efficacy |
| • | Cost |
| • | Potency |
| • | Cost |
| • | Incontinence |
| • | Cost |
| • | Time |
| • | Cost |
| • | Complications |
| • | Cost |

21
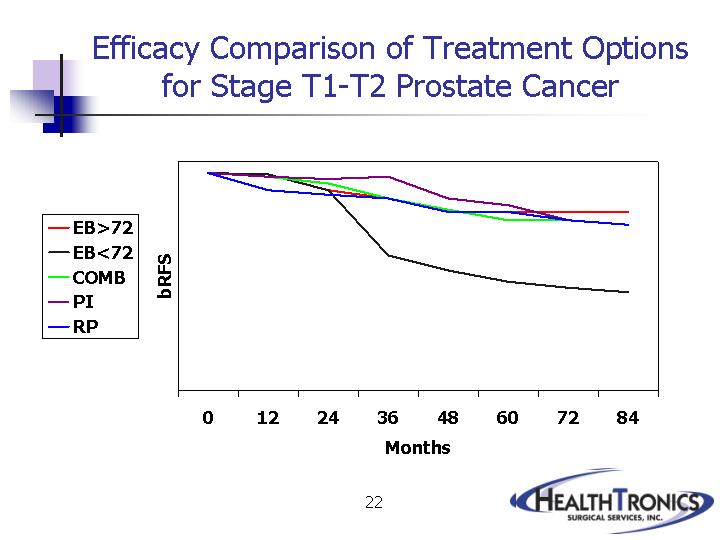
Efficacy Comparison of Treatment Options
for Stage T1-T2 Prostate Cancer
|
|
| MONTHS |
| |||||||||||||||||||||
|
|
|
| ||||||||||||||||||||||
bRFS |
|
| 0 |
|
| 12 |
|
| 24 |
|
| 36 |
|
| 48 |
|
| 60 |
|
| 72 |
|
| 84 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||
EB>72 |
|
| 100 |
|
| 99 |
|
| 92 |
|
| 88 |
|
| 82 |
|
| 82 |
|
| 82 |
|
| 82 |
|
EB<72 |
|
| 100 |
|
| 99 |
|
| 92 |
|
| 62 |
|
| 55 |
|
| 50 |
|
| 47 |
|
| 45 |
|
COMB |
|
| 100 |
|
| 98 |
|
| 95 |
|
| 88 |
|
| 83 |
|
| 78 |
|
| 78 |
|
| 76 |
|
PI |
|
| 100 |
|
| 98 |
|
| 97 |
|
| 98 |
|
| 88 |
|
| 85 |
|
| 78 |
|
| 76 |
|
RP |
|
| 100 |
|
| 92 |
|
| 90 |
|
| 88 |
|
| 82 |
|
| 82 |
|
| 78 |
|
| 76 |
|

22
|
|
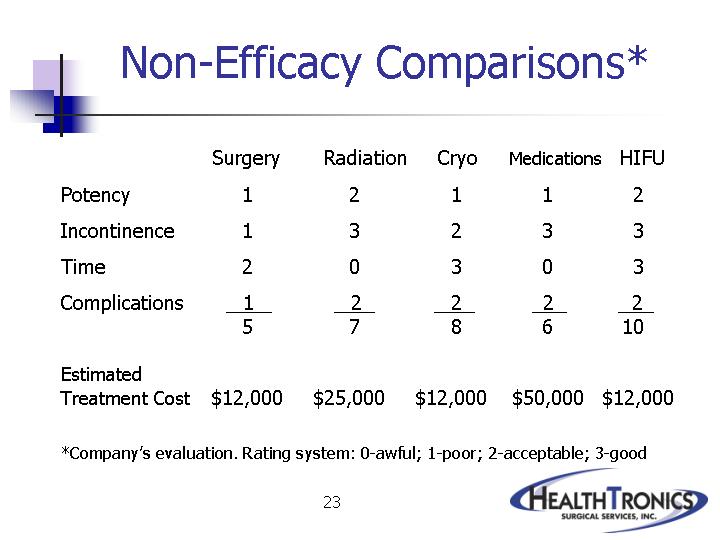
| Surgery |
|
| Radiation |
|
| Cryo |
|
| Medications |
|
| HIFU |
|
|
|
|
|
|
|
|
|
|
|
|
| |||||
Potency |
|
| 1 |
|
| 2 |
|
| 1 |
|
| 1 |
|
| 2 |
|
Incontinence |
|
| 1 |
|
| 3 |
|
| 2 |
|
| 3 |
|
| 3 |
|
Time |
|
| 2 |
|
| 0 |
|
| 3 |
|
| 0 |
|
| 3 |
|
Complications |
|
| 1 |
|
| 2 |
|
| 2 |
|
| 2 |
|
| 2 |
|
|
|
|
|
|
|
|
|
|
|
|
| |||||
|
|
| 5 |
|
| 7 |
|
| 8 |
|
| 6 |
|
| 10 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated |
|
| $ 12,000 |
|
| $ 25,000 |
|
| $ 12,000 |
|
| $ 50,000 |
|
| $ 12,000 |
|
*Company’s evaluation. Rating system: 0-awful; 1-poor; 2-acceptable; 3-good

23
Orthopaedic Extracorporeal
Shock Wave Surgery
|
|
|
| OssaTron® |
|
|
|
|
| ||
|
|
|
| ||
EvoTron® |
|
| |||
|
|
| |||
|
|
|
| ||
|
|
|
| ||

24
Total OssaTrons® Sold per Region
GRAPHIC
Americas = 71
Europe = 35
Asia = 11

25
Total EvoTrons® Sold per Region
GRAPHIC
Americas = 0*
Europe = 77
Asia = 6
*EvoTron® is not FDA approved for use in the U.S.

26
• | Active with 51 Partnerships in 47 States |
|
|
• | Average location now treating 150 cases per year |
|
|
• | Orthopaedic Division is losing money overall but pre-tax net income loss decreased from$1.5 million in 2002 to $700,000 in 2003 |
|
|
• | HealthTronics Surgical Services’ average partnership stake is approximately 50-60% |

27
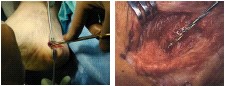
OssaTron® Treatment for
Chronic Plantar Fasciitis
• | American Podiatric Medical Association estimates over 17 million cases of heel pain in U.S. per year |
|
|
• | Over 17,000 patients treated to date in U.S. by HealthTronics |
|
|
• | HealthTronics patient history (2000 to present) demonstrates are-treatment rate of <10% |
|
|
• | Based on current data, outcomes appear to be better than surgery |

28
OssaTron® Treatment for
Chronic Lateral Epicondylitis
• | More than “tennis” elbow |
|
|
• | Opens orthopaedic market for OssaTron® |
|
|
• | Procedure easily learned by doctor |
|
|
• | Minimal complications |
|
|
• | In double-blind, placebo-controlled clinical trial, the FDA concluded treatment is safe and effective |
|
|
• | Training of physicians now underway |

29
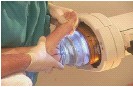
Veterinary Extracorporeal Shock
Wave Surgery VersaTron®
|
| |
| ||
| ||
| ||
| ||
VersaTron® | ||
|
| |
• | Introduced in 2001 in USA for equine specialists | |
|
| |
• | 5.5 Million Horses in the U.S. | |
|
| |
• | 145 Units sold by year-end 2003 | |
|
| |
• | Excellent results in several indications | |
|
| |
• | FDA accepted study with CSU in Denver | |
|
| |
• | Provides excellent data that can be used by Orthopaedics | |

30
Total VersaTrons® Sold per Region
GRAPHIC
Americas = 145
Europe = 25
Asia = 9

31
Orthopaedic US Market
Opportunity
Condition |
| Estimated # of |
| Estimated |
| ||||
|
|
| |||||||
*Heel Spurs |
|
| 250,000 |
|
| $ | 500 Million |
| |
*Tennis Elbow |
|
| 250,000 |
|
| $ | 500 Million |
| |
Non-Union Fractures |
|
| 75,000 |
|
| $ | 300 Million |
| |
Achilles Tendonitis |
|
| 100,000 |
|
| $ | 250 Million |
| |
Patellar Tendonitis |
|
| 100,000 |
|
| $ | 250 Million |
| |
Shoulder Tendonitis |
|
| 100,000 |
|
| $ | 250 Million |
| |
AVN of the Femoral Head |
|
| 25,000 |
|
| $ | 100 Million |
| |
|
|
|
|
|
|
| |||
|
|
| Total |
| $ | 2.15 BILLION |
| ||
*FDA approved indications

32
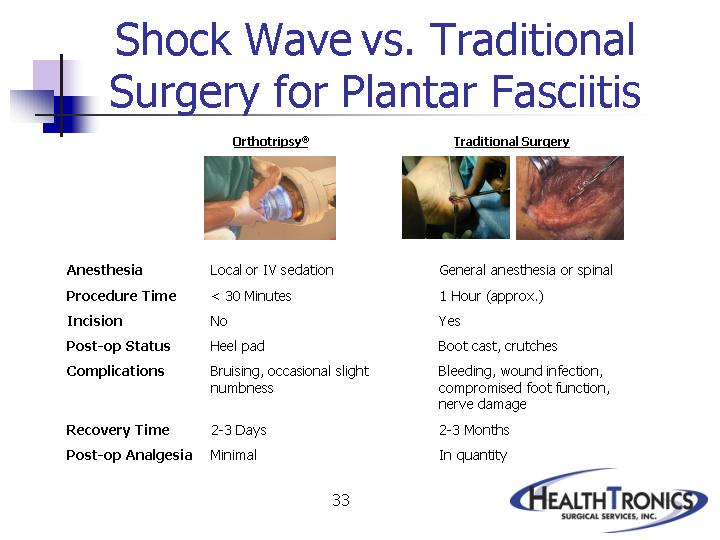
Shock Wave vs. Traditional
Surgery for Plantar Fasciitis
|
| Orthotripsy® |
| Traditional Surgery |
| ||
|
|
|
| ||||
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
Anesthesia |
|
| Local or IV sedation |
|
| General anesthesia or spinal |
|
|
|
|
|
|
|
|
|
Procedure Time |
|
| < 30 Minutes |
|
| 1 Hour (approx.) |
|
|
|
|
|
|
|
|
|
Incision |
|
| No |
|
| Yes |
|
|
|
|
|
|
|
|
|
Post-op Status |
|
| Heel pad |
|
| Boot cast, crutches |
|
|
|
|
|
|
|
|
|
Complications |
|
| Bruising, occasional slight |
|
| Bleeding, wound infection, |
|
|
|
|
|
|
|
|
|
Recovery Time |
|
| 2-3 Days |
|
| 2-3 Months |
|
|
|
|
|
|
|
|
|
Post-op Analgesia |
|
| Minimal |
|
| In quantity |
|

33
Growing Clinical Acceptance
and Clinical Data
|
|
|
|
|
|
| |
| • | Significant number of positive clinical studies published worldwide | |
|
|
| |
| • | Limited number of negative studiespublished worldwide | |
|
|
| |
| • | NO negative studiesusing HealthTronics’current treatment protocol | |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
|

34


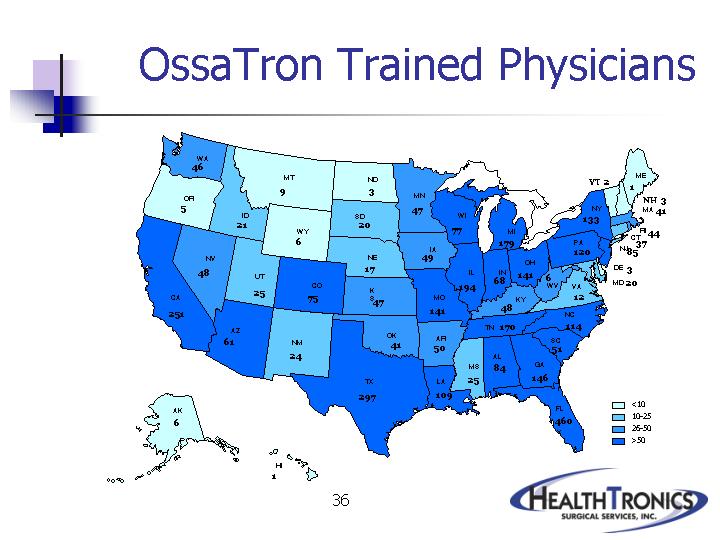
35

36
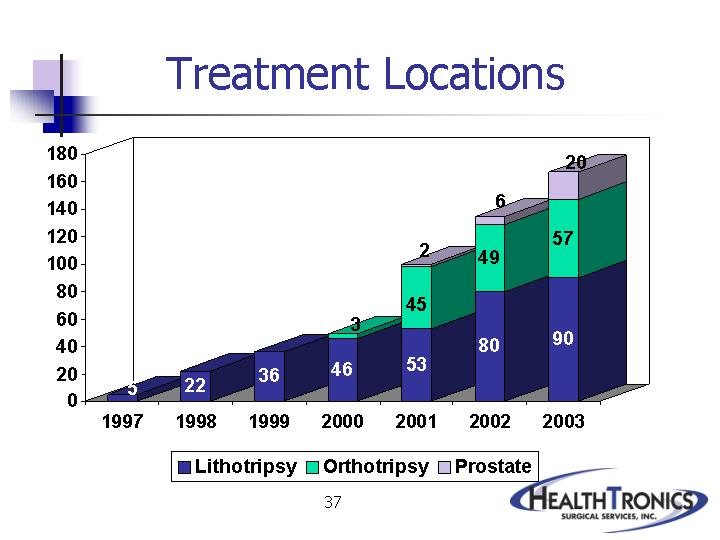
|
|
| 1997 |
|
| 1998 |
|
| 1999 |
|
| 2000 |
|
| 2001 |
|
| 2002 |
|
| 2003 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||
Lithotripsy |
|
| 5 |
|
| 22 |
|
| 36 |
|
| 46 |
|
| 53 |
|
| 80 |
|
| 90 |
|
Orthotripsy |
|
|
|
|
|
|
|
|
|
|
| 3 |
|
| 45 |
|
| 49 |
|
| 57 |
|
Prostate |
|
|
|
|
|
|
|
|
|
|
|
|
|
| 2 |
|
| 6 |
|
| 20 |
|

37

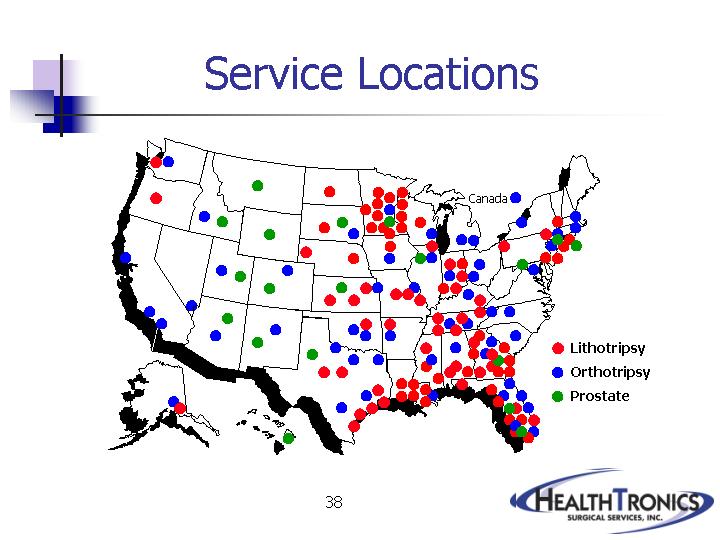
38
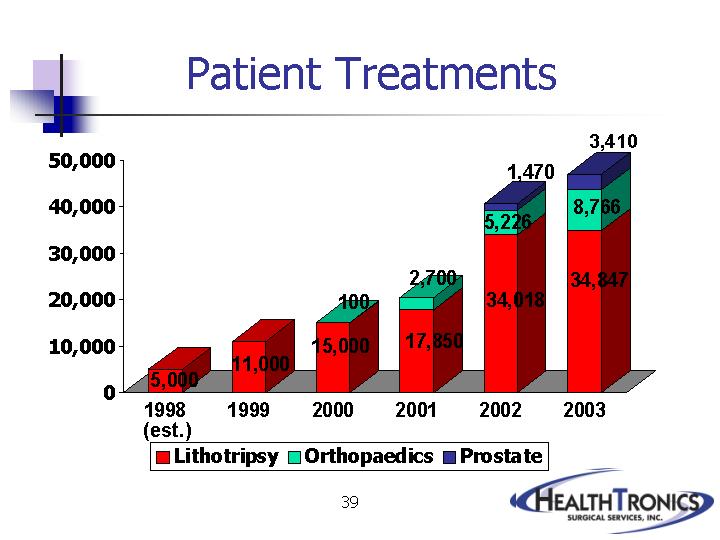
|
|
| 1998 |
|
| 1999 |
|
| 2000 |
|
| 2001 |
|
| 2002 |
|
| 2003 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Lithotripsy |
|
| 5,000 |
|
| 11,000 |
|
| 15,000 |
|
| 17,850 |
|
| 34,018 |
|
| 34,847 |
|
Orthopaedics |
|
|
|
|
|
|
|
| 100 |
|
| 2,700 |
|
| 5,226 |
|
| 8,766 |
|
Prostate |
|
|
|
|
|
|
|
|
|
|
|
|
|
| 1,470 |
|
| 3,410 |
|

39
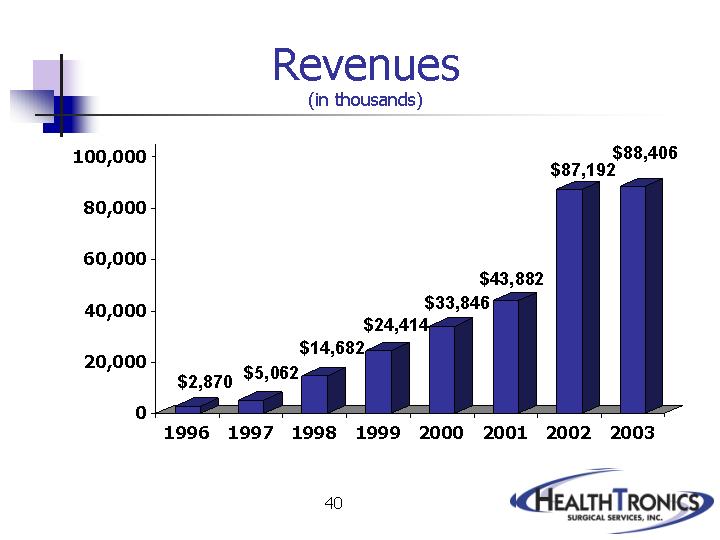
|
| 1996 |
| 1997 |
| 1998 |
| 1999 |
| 2000 |
| 2001 |
| 2002 |
| 2003 |
| ||||||||
|
|
|
|
|
|
|
|
|
| ||||||||||||||||
Revenues |
| $ | 2,870 |
| $ | 5,062 |
| $ | 14,682 |
| $ | 24,414 |
| $ | 33,846 |
| $ | 43,882 |
| $ | 87,192 |
| $ | 88,406 |
|

40
• | Argil Wheelock, M.D. –Chairman &CEO since 1996 |
| 20 years experience in physician-related business activities. Practicing urologist, 1979-1996. Public company consultant, 1991-1996. |
|
|
• | Martin McGahan – President and COO; Previous CFO since 2001 |
| 15 years experience in healthcare finance, business development and corporate finance with various public and private companies. |
|
|
• | Price Dunaway – COO of Lithotripsy Operations since 2003 |
| 20 years experience in healthcare. Former CEO of biotech company and former CFO of urological device and services company. |
|
|
• | Andreas Baenziger, M.D. – COO of Sales & Services since 2004 |
| Orthopaedic surgeon with 16 years experience in healthcare companies. Former CEO of HMT Holding AG. |
|
|
• | Ron Gully – SR VP of Orthopaedic Division since 2001 |
| 17 years experience in healthcare services. Former CEO of Ascension Corporation, a healthcare billing and management firm. |

41
Nasdaq: |
| HTRN |
Share Price: |
| $6.00 |
52 Week Range: |
| $11.67 – $5.10 |
Shares Outstanding: |
| 11.6 Million |
Market Capitalization: |
| $69.6 Million |
Average Daily Volume: |
| 96,000 |
2001 EPS: |
| $0.28 |
2002 EPS: |
| $0.55* |
2003 EPS: |
| $0.45 |
*GAAP earnings were $.73, which includes gain on sale of $.18 from disposition of USL

42
• | Rapidly growing innovator with the first FDA-approved device for orthopaedic shock wave surgery |
|
|
• | Addressing large untapped orthopaedic market that could exceed $2 billion |
|
|
• | Steady cash flow from kidney lithotripsy business financing new opportunities in orthopaedics and urology |
|
|
• | Physician network that will be leveraged to offer additional products and services |
|
|
• | Record of strong growth in sales and earnings |
|
|
• | Globally experienced management team |
|
|
• | Strong technology and product pipeline |
|
|
• | Promising new scientific activities |

43
A Leading Provider of
Orthopaedic and Urologic
Non-Invasive Medical Devices
and Medical Services