Exhibit 99.1

Simulations Plus, Inc.
NASDAQ: SLP
First Quarter Fiscal Year 2010
Conference Call
January 13, 2010
NASDAQ: SLP
First Quarter Fiscal Year 2010
Conference Call
January 13, 2010

Introduction and Welcome
• Introduction and Welcome
• Agenda
– First Quarter FY 2010 (1QFY10) Financial
Summary
Summary
– Progress in Business Units
– Strategy Going Forward
– Questions and Answers

With the exception of historical information, the matters discussed in
this presentation are forward-looking statements that involve a number
of risks and uncertainties. The actual results of the Company could
differ significantly from those statements. Factors that could cause or
contribute to such differences include, but are not limited to: continuing
demand for the Company’s products; competitive factors; the
Company’s ability to finance future growth; the Company’s ability to
produce and market new products in a timely fashion; the Company’s
ability to continue to attract and retain skilled personnel; the Company’s
ability to identify, evaluate, and close suitable acquisitions; and the
Company’s ability to sustain or improve current levels of productivity.
Further information on the Company’s risk factors is contained in the
Company’s quarterly and annual reports and filed with the Securities
and Exchange Commission.
this presentation are forward-looking statements that involve a number
of risks and uncertainties. The actual results of the Company could
differ significantly from those statements. Factors that could cause or
contribute to such differences include, but are not limited to: continuing
demand for the Company’s products; competitive factors; the
Company’s ability to finance future growth; the Company’s ability to
produce and market new products in a timely fashion; the Company’s
ability to continue to attract and retain skilled personnel; the Company’s
ability to identify, evaluate, and close suitable acquisitions; and the
Company’s ability to sustain or improve current levels of productivity.
Further information on the Company’s risk factors is contained in the
Company’s quarterly and annual reports and filed with the Securities
and Exchange Commission.
Safe Harbor Statement Under the
Private Securities Litigation Reform Act
of 1995
Private Securities Litigation Reform Act
of 1995

1QFY10 (3 months) Financial Summary
• Gross revenues = $2.437 MM (up 14.2% from 1QFY09)
– Pharmaceutical software and services = $1.735 MM (up 21.3% from $1.430 MM in 1QFY09)
– Words+ subsidiary = $702,000 (down 0.1% from $703,000 1QFY09)
• SG&A = $1,004,000 (up 11.1% from $904,000 in 1QFY09)
– SG&A increased due to travel and show expenses, bonuses to officer, and increases in salaries
and benefits over 1QFY09. As percent of sales, SG&A decreased from 42.4% to 41.2%.
and benefits over 1QFY09. As percent of sales, SG&A decreased from 42.4% to 41.2%.
• R&D Expense = $261,000 (down 2.9% from $269,000 from 1QFY09)
– Greater time on contract studies
• Income Before Income Taxes = $661,000 (up 46.0% from $453,000 in 1QFY09)
– Greater pharmaceutical software and services revenues
• Net Income = $430,000 (up 38.0% from $312,000 in 1QFY09)
– High margin on pharmaceutical business results in percentage earnings growth greater than
percentage revenues growth once breakeven is passed
percentage revenues growth once breakeven is passed
• EPS = $0.03/FD share (up $0.007 from $0.02/FD share in 1QFY09)
– Positive results continue in spite of global economic crisis
– Share repurchase reduced number of fully diluted shares
• Cash = $7.97 MM (up $1.91 MM or 31.5% from $6.06 MM in 1QFY09)
– Cash increased even with >$1 MM share repurchase program spending over $1 MM in FY09
• Shareholders’ Equity = $10.812 MM (up 4.8% from $10.315 MM in 1QFY09)
– Again, in spite of spending ~$1MM for share repurchases

Share Repurchase Program
• Board of directors authorized share repurchase program for up to
$2.5 million from October 2008 through October 2009. Actual
repurchases began December 2, 2008, so the board extended the
repurchase period through December 1, 2009.
$2.5 million from October 2008 through October 2009. Actual
repurchases began December 2, 2008, so the board extended the
repurchase period through December 1, 2009.
• Total of 1.02 MM shares were bought back at average price per
share of about $1.32.
share of about $1.32.
• No plans at this time to repurchase shares; however, the board of
directors may decide to reinstitute repurchasing shares at any
time.
directors may decide to reinstitute repurchasing shares at any
time.

Progress - - 1
• ADMET Predictor™ (Structure-property prediction program)
– Version 4.0 was released in July with new metabolism and toxicity models
– New Dose Optimization Module developed under estimates dose amount
needed for new molecules based on mini-GastroPlus simulation
needed for new molecules based on mini-GastroPlus simulation
• Developed under Pfizer funding and in active use at Pfizer now
– Expanded graphics capability was added with Miner3D component
– Work continues on our $525,000 SBIR Phase II grant with excellent
progress on advancing the predictions in ADMET Predictor through better
atomic and molecular descriptors. The rapid partial charge calculations we
have developed allow calculation of atomic partial charges at the rate of
hundreds of thousands of molecules per hour rather than one molecule per
day using traditional quantum methods.
progress on advancing the predictions in ADMET Predictor through better
atomic and molecular descriptors. The rapid partial charge calculations we
have developed allow calculation of atomic partial charges at the rate of
hundreds of thousands of molecules per hour rather than one molecule per
day using traditional quantum methods.

Solubility in Biorelevant Fluids
• During FY09 we funded a series of experiments with Northeastern
University to measure the solubility of 160 drugs and drug-like compounds
in three different types of biorelevant fluids:
University to measure the solubility of 160 drugs and drug-like compounds
in three different types of biorelevant fluids:
– Fasted state simulated gastric fluid (FaSGF)
– Fasted state simulated intestinal fluid (FaSSIF)
– Fed state simulated intestinal fluid (FeSSIF)
• These data have been used to build preliminary solubility models for
ADMET Predictor to provide estimates for in vivo solubilities for new
compounds without the need to measure them
ADMET Predictor to provide estimates for in vivo solubilities for new
compounds without the need to measure them
• We presented a scientific poster at the American Association of
Pharmaceutical Scientists meeting in November, which generated very high
interest from scientists at the meeting
Pharmaceutical Scientists meeting in November, which generated very high
interest from scientists at the meeting
• We will soon offer these data as a new product. We also intend to conduct
more of these experiments to expand the database and improve our
predictions. And we are considering funding other types of experiments to
improve different aspects of our modeling and simulation capabilities.
more of these experiments to expand the database and improve our
predictions. And we are considering funding other types of experiments to
improve different aspects of our modeling and simulation capabilities.

Progress - - 2
• ClassPharmer™ (Data Mining and de novo Molecule Design)
– Numerous improvements were being incorporated during the 4th
quarter, and Version 4.7 was released in October.
quarter, and Version 4.7 was released in October.
– We continue to enhance ClassPharmer’s capabilities in data mining and
de novo molecule design (design of new molecules) and to add
additional convenience features requested by our users.
de novo molecule design (design of new molecules) and to add
additional convenience features requested by our users.
– New “scaffold hopping” capability was added in 4.7 - allows chemists
to change the core (or any portion) of a molecule while maintaining
other features and the bond distances between them.
to change the core (or any portion) of a molecule while maintaining
other features and the bond distances between them.
– Current efforts are focused on refactoring the code to make it more
streamlined and efficient, and adding a number of additional
visualization and user convenience features.
streamlined and efficient, and adding a number of additional
visualization and user convenience features.
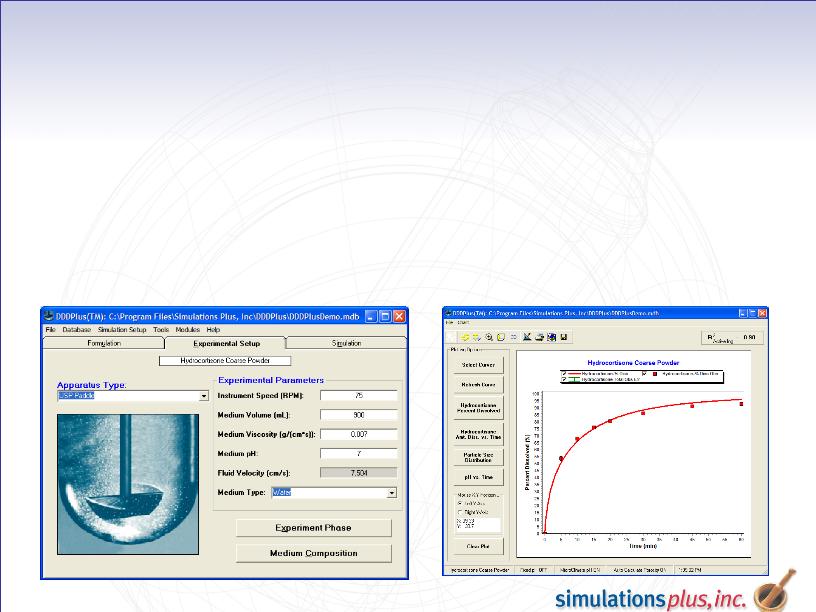
Progress - - 3
DDDPlus - Simulates laboratory dissolution experiments
Sales continue to increase steadily
Version 3.0 was released in Q3 with a number of important
enhancements
enhancements
Remains the only tool of its kind
Further development on hold during 1QFY10 to allow
greater support for contract studies
greater support for contract studies

Progress - - 4
• GastroPlus™
– Development of Drug-drug Interaction module continues under funded
collaboration with Roche - initial capability is now in use at Roche. Full
capability to be released with Version 7.0.
collaboration with Roche - initial capability is now in use at Roche. Full
capability to be released with Version 7.0.
– Development of ocular delivery capability under a funded collaboration
with Pfizer is complete - will be released with Version 7.0.
with Pfizer is complete - will be released with Version 7.0.
– Development of a nasal-pulmonary delivery module under a funded
collaboration with GlaxoSmithKline is complete and in use at
GlaxoSmithKline. Full capability will be released with Version 7.0.
collaboration with GlaxoSmithKline is complete and in use at
GlaxoSmithKline. Full capability will be released with Version 7.0.
– Consulting contracts continue to come in to assist pharmaceutical
companies worldwide in analyzing preclinical and clinical data with
GastroPlus. Multiple contracts currently underway with three top-5
pharmaceutical companies.
companies worldwide in analyzing preclinical and clinical data with
GastroPlus. Multiple contracts currently underway with three top-5
pharmaceutical companies.
– At AAPS meeting in November, GastroPlus was mentioned by every
speaker in at least 3 different sessions as the tool used to run the analyses
that were being presented. One presentation by Novartis noted that
GastroPlus simulation results submitted to the FDA resulted in the FDA
agreeing to eliminate a clinical trial that would have otherwise been
required.
speaker in at least 3 different sessions as the tool used to run the analyses
that were being presented. One presentation by Novartis noted that
GastroPlus simulation results submitted to the FDA resulted in the FDA
agreeing to eliminate a clinical trial that would have otherwise been
required.
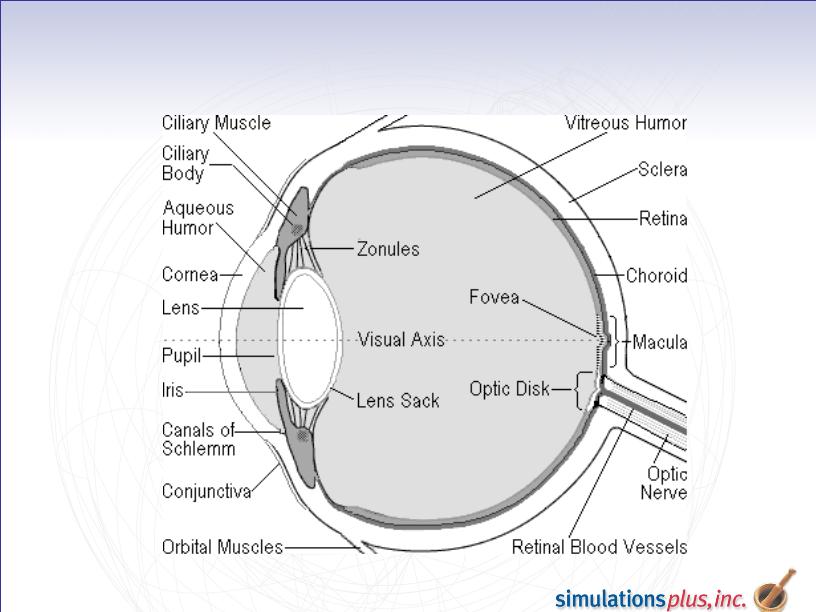
GastroPlus Ocular Delivery - Funded
Collaboration with Pfizer
Collaboration with Pfizer
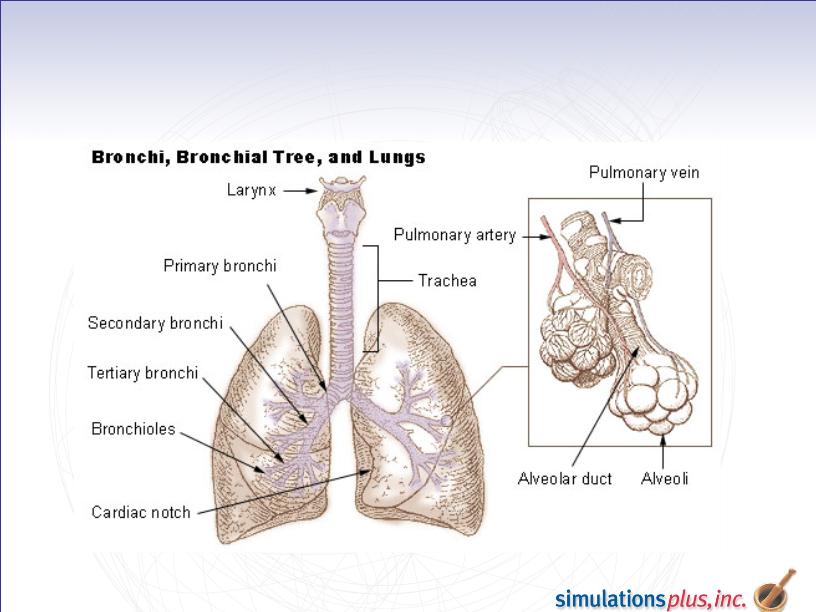
GastroPlus Pulmonary Delivery - Funded
Collaboration with GlaxoSmithKline
Collaboration with GlaxoSmithKline

Progress - - 5
• Words+ subsidiary
– General economy appears to be affecting this business -
many states are struggling, and a large percentage of our
sales come from 3rd-party sources (Medicaid, Medicare,
schools, state vocational rehabilitation programs, etc.)
many states are struggling, and a large percentage of our
sales come from 3rd-party sources (Medicaid, Medicare,
schools, state vocational rehabilitation programs, etc.)
– New Conversa™ communication device selling well
– Full-time national sales manager working to improve
marketing and sales activities and dealer relationships
marketing and sales activities and dealer relationships
– Two new employee sales reps added in California during
2009.
2009.
– Significant progress on a new product that includes a
patent disclosure with IP owned by Words+
patent disclosure with IP owned by Words+

Miscellaneous Items
• Potential acquisition we were working on for a number of months did not go
through - - seller decided to go with another buyer. Potential remains to acquire
software part of the business, which was our primary interest.
through - - seller decided to go with another buyer. Potential remains to acquire
software part of the business, which was our primary interest.
• Due diligence currently in progress on three acquisition possibilities for the
pharmaceutical side of the business.
pharmaceutical side of the business.
• Two new Ph.D.s started recently and are making excellent contributions
already. Another has just accepted our offer and will join the team on March 1.
Our elite team continues to grow!
already. Another has just accepted our offer and will join the team on March 1.
Our elite team continues to grow!
• Dr. Robert Clark will join the company on January 19 as Director, Life
Sciences. Experience as VP of Research of a public pharmaceutical software
company will provide valuable leadership for the Life Sciences team.
Sciences. Experience as VP of Research of a public pharmaceutical software
company will provide valuable leadership for the Life Sciences team.

Strategy Going Forward
• Continue to expand product line and services
– Pharmaceutical software and services
• Continue to expand Life Sciences team
– For several new product development projects in our corporate strategic plan
– To increase capacity to conduct consulting studies and funded collaborations
• Seek and complete strategic acquisitions
– Goal is to add both new products & services as well as additional scientific staff
– Negotiations continue at slow pace, but we expect results in 2010
• Continue to examine additional SBIR opportunities
– Words+ subsidiary
• Continue product improvements and more aggressive marketing and sales
• Added staff and new billing software to process prior authorizations and accounts
receivable more quickly - now collecting old receivables from state Medicaid
agencies
receivable more quickly - now collecting old receivables from state Medicaid
agencies
• Seek SBIR funding opportunities that fit our expertise
• Continue our expanded marketing and sales activities - increasing number
of conferences and meetings worldwide - lead list is growing
of conferences and meetings worldwide - lead list is growing
– China appears to be significant potential market for pharmaceutical business
• Two new dealers in China now signed up
– Continue our push into the environmental toxicology area

Additional Factors
• State of the pharmaceutical industry
– Mergers have had no adverse effect on business to date - in fact licenses
have increased at the larger companies, academic and government
organizations.
have increased at the larger companies, academic and government
organizations.
–Large companies continue to adopt in silico (software) technologies in
various departments, and Simulations Plus software is independently
ranked as best-in-class.
various departments, and Simulations Plus software is independently
ranked as best-in-class.
–Government agencies expanding use (FDA added 10 more GastroPlus
licenses recently.)
licenses recently.)
–Smaller companies are recognizing the value of in silico tools as a result
of the many scientific presentations that are now being presented by our
customers at meetings around the world.
of the many scientific presentations that are now being presented by our
customers at meetings around the world.
– Outsourcing of consulting studies is on the rise. We expect to see
continued growth in this area of business.
continued growth in this area of business.

Summary
• Simulations Plus financial strength continues
– Excellent cash position that continues to grow
– No debt
• Record first quarter! Strong marketing and sales efforts starting to show
results
results
– Pharmaceutical software and services revenues up 21.3% from 1QFY09
– Words+ subsidiary flat for the quarter, but new product developments are expected
to improve performance of this division in 2010
to improve performance of this division in 2010
• Focus is on maintaining best-in-class position in product quality and superior
customer service in both business units
customer service in both business units
– Version 7.0 of GastroPlus is expected to generate significant additional interest in this
industry-leading program
industry-leading program
– Internal projects expected to add new products during FY2010
– Price increases and reduced discounts expected to further add to revenues and earnings
• Continue to actively seek strategic acquisitions.
• Slides available by e-mail on request through info@simulations-plus.com

Questions?